WO2020002090A1 - Substituted thiazolylpyrrolones, salts thereof and the use thereof as herbicidal agents - Google Patents
Substituted thiazolylpyrrolones, salts thereof and the use thereof as herbicidal agents Download PDFInfo
- Publication number
- WO2020002090A1 WO2020002090A1 PCT/EP2019/066198 EP2019066198W WO2020002090A1 WO 2020002090 A1 WO2020002090 A1 WO 2020002090A1 EP 2019066198 W EP2019066198 W EP 2019066198W WO 2020002090 A1 WO2020002090 A1 WO 2020002090A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- phenyl
- methyl
- alkyl
- butenyl
- dimethyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC(OC(C(C)=C1)N(*)C1=O)=O Chemical compound CC(OC(C(C)=C1)N(*)C1=O)=O 0.000 description 13
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
Definitions
- the invention relates to the technical field of crop protection agents, in particular that of herbicides for the selective control of weeds and weeds in crops of useful plants.
- this invention relates to substituted thiazolylpyrrolones and their salts, processes for their preparation and their use as herbicides.
- Crop crops or active ingredients to combat undesirable plant growth sometimes have disadvantages when used, either because they (a) have no or an inadequate herbicidal activity against certain harmful plants, (b) the spectrum of the harmful plants is too small to combat with an active ingredient can be (c) insufficient selectivity in crops and / or (d) have a toxicologically unfavorable profile.
- active ingredients which can be used as plant growth regulators in some crop plants lead to undesirably reduced crop yields in other crop plants or are incompatible or only compatible with the crop plant in a narrow range of application rates.
- Some of the known active ingredients cannot be economically manufactured on an industrial scale because of precursors and reagents that are difficult to access, or they have insufficient chemical stabilities. For other active substances, the effect depends too much on environmental conditions such as weather and soil conditions.
- Isoxazolines Substituted pyrrolones and their herbicidal or pesticidal properties are also described in CH633678, DE 2735841, EP0297378, EP0334133, EP0339390 and EP0286816.
- WO2016 / 071361, WO2016 / 071362, WO2016 / 071363 and WO2016 / 071364 also describe substituted hydantoins which also carry heterocyclic substituents on the nitrogen, for example optionally further substituted isoxazolines.
- Selected specially substituted 1,3,4-thiadiazolyl and l, 2,4-thiadiazolyl-2,5-dioxoimidazolines and their herbicidal activity are described in DE2247266 described.
- Active ingredients are described, for example, in WO2015 / 018434.
- X and Y independently of one another represent CH or the grouping CR 1 , where
- X stands for CH if Y stands for the grouping CR 1 and
- X stands for the grouping CR 1 , if Y stands for CH,
- R 1 for halogen, cyano, (Ci-Cg) -alkyl, (Ci-C 8 ) -haloalkyl, (Ci-Cg) -hydroxyalkyl, (C i - C s) -
- a 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
- a 1 stands for CH if A 2 stands for the grouping CR 2 and
- a 1 stands for the grouping CR 2 , if A 2 stands for CH,
- R 2 for hydrothio, hydroxy, halogen, (Ci-Cg) alkyl, (Ci-Cg) haloalkyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) cycloalkyl (Ci-Cg) alkyl , Aryl, heteroaryl, heterocyclyl, aryl- (Ci-Cg) -alkyl,
- R 3 for hydroxy, hydrothio, halogen, NR 5 R 6 , (Ci-Cs) alkoxy, (C 3 -Cio) cycloalkyl- (Ci-C 8 ) alkoxy, aryl- (Ci-C 8 ) alkoxy , (Ci-C 8 ) -alkoxy- (Ci-C 8 ) -alkoxy, arylcarbonyloxy, (Ci-C 8 ) -alkylcarbonyloxy, aryl- (Ci-C 8 ) -alkylcarbonyloxy, heteroarylcarbonyloxy, (C 3 -C 10 ) - Cycloalkylcarbonyloxy, heterocyclylcarbonyloxy, (Ci-C 8 ) -haloalkyl-carbonyloxy, (C 2 -C 8 ) - alkenylcarbonyloxy, OC (0) OR 5 , OC (0) SR 9 , OC (S)
- R 4 represents hydrogen, (Ci-Cs) -alkyl
- R 5 and R 6 are identical or different and independently of one another are hydrogen, (C 1 -C 8 ) -alkyl
- R 7 is H, (Ci-C 8 ) alkyl, (C 2 -C 8 ) alkenyl, (C 2 -C 8 ) alkynyl, (Ci-C 8 ) cyanoalkyl, (Ci-Cio) haloalkyl , (C 2 -C 8 ) haloalkenyl, (C 2 -C 8 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -C 10 ) halocycloalkyl, (C 4 -Cio) cycloalkenyl, ( C 4 -Cio) halocycloalkenyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) alkyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) haloalkyl, aryl, aryl- (Ci -C 8 ) -alky
- R 8 and R 9 are identical or different and independently of one another for (Ci-C 8 ) alkyl, (C 2 -C 8 ) alkenyl, (C 2 -C 8 ) alkynyl, (Ci-C 8 ) cyanoalkyl , (Ci-Cio) haloalkyl, (C 2 -C 8 ) haloalkenyl, (C 2 -C 8 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -Cio) halocycloalkenyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) alkyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) haloalkyl , Aryl, aryl- (Ci
- the compounds of general formula (1) can be added by adding a suitable one
- inorganic or organic acid such as mineral acids such as HCl, HBr, H2SO4, H3P04 or HNO3, or organic acids, e.g. B. carboxylic acids such as formic acid, acetic acid, propionic acid, oxalic acid, lactic acid or salicylic acid or sulfonic acids, such as p-toluenesulfonic acid, to a basic group such as e.g. Amino, alkylamino, dialkylamino, piperidino, morpholino or pyridino, form salts. These salts then contain the conjugate base of the acid as an anion. Suitable substituents which are in deprotonated form, e.g. Sulfonic acids, certain
- Sulphonic acid amides or carboxylic acids are present, inner salts with protonatable groups such as amino groups can form. Salt formation can also be caused by exposure to a base
- Suitable bases are, for example, organic amines, such as trialkylamines, morpholine, piperidine and pyridine, and ammonium, alkali or
- Potassium hydroxide, sodium and potassium carbonate and sodium and potassium hydrogen carbonate are compounds in which the acidic hydrogen is replaced by a cation suitable for agriculture, for example metal salts, in particular alkali metal salts or
- Alkaline earth metal salts especially sodium and potassium salts, or also ammonium salts, salts with organic amines or quaternary ammonium salts, for example with cations of the formula
- R a to R d each independently represent an organic radical, in particular alkyl, aryl, arylalkyl or alkylaryl.
- Alkylsulfoxonium salts such as (Ci-C4) -trialkylsulfonium and (Ci-C4) -trialkylsulfoxonium salts.
- the substituted thiazolylpyrrolones of the general formula (1) according to the invention can be present in various tautomeric structures, all of which are intended to be encompassed by the general formula (1).
- the compounds of the formula (1) and their salts used according to the invention are referred to below as "compounds of the general formula (1)".
- Preferred subject matter of the invention are compounds of the general formula (1), in which
- X and Y independently of one another represent CH or the grouping CR 1 , where
- X stands for CH if Y stands for the grouping CR 1 and
- X stands for the grouping CR 1 , if Y stands for CH,
- R 1 for halogen, cyano, (Ci-Cv) alkyl, (Ci-C 7 ) haloalkyl, (Ci-Cv) hydroxyalkyl, (C 1 -C 7 ) -
- a 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
- a 1 stands for CH if A 2 stands for the grouping CR 2 and
- a 1 stands for the grouping CR 2 , if A 2 stands for CH,
- R 2 for hydrothio, hydroxy, halogen, (Ci-C 7 ) alkyl, (Ci-C 7 ) haloalkyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) cycloalkyl- (Ci-C 7 ) alkyl, aryl, heteroaryl, heterocyclyl, aryl (Ci-C 7 ) alkyl, heteroaryl (Ci-C 7 ) alkyl, heterocyclyl (Ci-C 7 ) alkyl, (C 2 -C 7 ) Alkenyl, (C 2 -C 7 ) alkynyl, (C 2 -C 7 ) haloalkenyl, (C 2 -C 7 ) haloalkynyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl , (C 4 -Cio)
- R 3 for hydroxy, hydrothio, halogen, NR 5 R 6 , (Ci-C 7 ) alkoxy, (C3-Cio) cycloalkyl- (Ci-C 7 ) alkoxy, aryl- (Ci-C 7 ) alkoxy , (Ci-C 7 ) alkoxy- (Ci-C 7 ) alkoxy, arylcarbonyloxy, (Ci-C 7 ) alkylcarbonyloxy, aryl- (Ci-C 7 ) alkylcarbonyloxy, heteroarylcarbonyloxy, (C3-C10) - cycloalkylcarbonyloxy , Heterocyclylcarbonyloxy, (Ci-C 7 ) -haloalkyl-carbonyloxy, (C 2 -C 7 ) - alkenylcarbonyloxy, OC (0) OR 5 , OC (0) SR 9 , OC (S) OR 8 , OC (
- R 4 represents hydrogen, (Ci-C 7 ) alkyl
- R 5 and R 6 are identical or different and independently of one another are hydrogen, (C 1 -C 7 ) -alkyl,
- R 7 for H, (Ci-C 7 ) alkyl, (C 2 -C 7 ) alkenyl, (C 2 -C 7 ) alkynyl, (Ci-C 7 ) cyanoalkyl, (Ci-C 7 ) - Haloalkyl, (C 2 -C 7 ) haloalkenyl, (C 2 -C 7 ) haloalkynyl, (C3-Cio) cycloalkyl, (C3-Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -Cio) halocycloalkenyl, (Ci-C 7 ) alkoxy- (Ci-C 7 ) alkyl, (Ci-C 7 ) - alkoxy- (Ci-C 7 ) haloalkyl, aryl, aryl- (Ci-C 7 ) -alkyl
- R 8 and R 9 are identical or different and independently of one another for (Ci-C 7 ) -alkyl, (C 2 -C 7 ) -
- X and Y independently of one another represent CH or the grouping CR 1 , where
- X stands for CH if Y stands for the grouping CR 1 and
- X stands for the grouping CR 1 , if Y stands for CH,
- R 1 halogen, cyano, (Ci-C 6 ) -alkyl, (Ci-Cej-haloalkyl, (Ci-Cej-hydroxyalkyl, (GG) -
- a 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
- a 1 stands for CH if A 2 stands for the grouping CR 2 and A 1 stands for the grouping CR 2 , if A 2 stands for CH,
- R 2 for cyano, halogen, (Ci-C6) alkyl, (G-C6) haloalkyl, (Ci-C6) hydroxyalkyl, (GG) -
- R 3 for hydroxy, hydrothio, halogen, NR 5 R 6 , (Ci-Ce) alkoxy, (C 3 -Cio) cycloalkyl- (Ci-C 6 ) alkoxy, aryl- (Ci-C6) alkoxy, (Ci-C6) alkoxy- (Ci-C6) alkoxy, arylcarbonyloxy, (GG) - alkylcarbonyloxy, aryl- (Ci-C6) alkylcarbonyloxy, heteroarylcarbonyloxy, (C 3 -G 0 ) - cycloalkylcarbonyloxy, heterocyclylcarbonyloxy, (Ci -C6) haloalkylcarbonyloxy, (C 2 -C 6 ) alkenylcarbonyloxy, OC (0) OR 5 , OC (0) SR 9 , OC (S) OR 8 , OC (S) SR 9 , OSO 2 R 8 , OSO
- R 4 represents hydrogen, (G-Ce) alkyl
- R 5 and R 6 are identical or different and independently of one another are hydrogen, (G-Ce) -alkyl,
- R 7 for H, (Ci-C 6 ) alkyl, (C 2 -C 6 ) alkenyl, (C 2 -C 6 ) alkynyl, (Ci-C 6 ) cyanoalkyl, (Ci-C 6 ) - Haloalkyl, (C 2 -C 6 ) haloalkenyl, (C 2 -C 6 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, ( C 4 -Cio) halocycloalkenyl, (GC 6 ) alkoxy- (GC 6 ) alkyl, (Ci-C 6 ) alkoxy- (Ci-C6) haloalkyl, aryl, aryl- (Ci-C6) alkyl , Heteroaryl, heteroaryl-
- X and Y independently of one another represent CH or the grouping CR 1 , where
- X stands for CH if Y stands for the grouping CR 1 and
- X stands for the grouping CR 1 , if Y stands for CH,
- R 1 for fluorine, chlorine, bromine, iodine, cyano, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1 -
- Cyclohexylmethyl methylcarbonyl, ethylcarbonyl, n-propylcarbonyl, iso-propylcarbonyl, n-butylcarbonyl, tert-butylcarbonyl, ethenyl, l-propenyl, 2-propenyl, 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-l-propenyl, 2-methyl-l-propenyl, l-methyl-2-propenyl, 2-methyl-2-propenyl, l-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1 - Methyl 1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl, 2-methyl-3-butenyl, 3-methyl
- a 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
- a 1 stands for CH if A 2 stands for the grouping CR 2 and
- a 1 stands for the grouping CR 2 , if A 2 stands for CH,
- R 2 for fluorine, chlorine, bromine, iodine, hydroxy, hydrothio, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl, 1,1-dimethylethyl, n-pentyl, 1 -Methylbutyl, 2-methylbutyl, 3 -methylbutyl, 1,1-dimethylpropyl, 1, 2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, n-hexyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4 -Methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbuty
- R 3 for hydroxy, hydrothio, fluorine, chlorine, bromine, iodine, amino, methoxy, ethoxy, n-propyloxy, 1-methylethoxy, n-butyloxy, 1-methylpropyloxy, 2-methylpropyloxy, l, l-dimethylethoxy, n-pentyloxy , 1-methylbutyloxy, 2-methylbutyloxy, 3-methylbutyloxy, l, l-dimethylpropyloxy,
- Difluoromethylcarbonyloxy methoxycarbonyloxy, ethoxycarbonyloxy, n-propyloxycarbonyloxy, iso-propyloxycarbonyloxy, n-butyloxycarbonyloxy, 1,1-dimethylethyloxycarbonyloxy, 2,2-dimethylpropyloxycarbonyloxy, benzyloxycarbonyloxy, allylcarbonyloxy, methylsulfonyloxy, ethylylsulfonyloxy, ethylylsulfonyloxy, Cyclopentylsulfonyloxy Cyclohexylsulfonyloxy, phenylsulfonyloxy, p-chlorophenylsulfonyloxy, m-chlorophenylsulfonyloxy, o-chlorophenylsulfonyloxy, p-fluorophenylsulfonyloxy
- R 4 represents hydrogen, methyl, ethyl.
- X and Y independently of one another represent CH or the grouping CR 1 , where
- X stands for CH if Y stands for the grouping CR 1 and
- X stands for the grouping CR 1 , if Y stands for CH,
- R 1 for chlorine, bromine, iodine, cyano, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl, l, l-dimethylethyl, n-pentyl, 1-methylbutyl, 2 -Methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1, 2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, n-hexyl, 1 - methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, l , l-dimethylbutyl, 1,2-dimethylbutyl, l, 3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl,
- R 3 for hydroxy, chlorine, amino, methoxy, ethoxy, n-propyloxy, 1-methylethoxy, n-butyloxy, 1 -
- Methyl propyloxy 2-methyl propyloxy, l, l-dimethylethoxy, n-pentyloxy, cyclopropyl methoxy, cyclobutyl methoxy, cyclopentyl methoxy, cyclohexyl methoxy, benzyloxy, p-chlorophenyl methoxy, m-chlorophenyl methoxy, o-chlorophenyl methoxy, p-methoxyphenyl methoxy, methoxy methoxy, methoxymoxy meth propyloxy,
- Difluoromethylcarbonyloxy methoxycarbonyloxy, ethoxycarbonyloxy, n-propyloxycarbonyloxy, iso-propyloxycarbonyloxy, n-butyloxycarbonyloxy, 1,1-dimethylethyloxycarbonyloxy, benzyloxycarbonyloxy, 2,2-dimethylpropyloxycarbonyloxy, allylcarbonyloxy, methylsulfonyloxy, ethylylsulfonyloxy, ethylylsulfonyl
- R 4 represents hydrogen
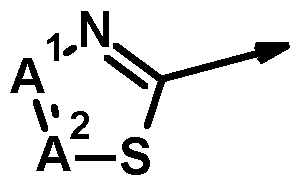
- Q stands for one of the groupings Q-1 .1 to Q-1 .68 specifically mentioned in the following table:
- X and Y independently of one another represent CH or the grouping CR 1 , where
- X stands for CH if Y stands for the grouping CR 1 and
- X stands for the grouping CR 1 , if Y stands for CH,
- R 1 for chlorine, bromine, iodine, cyano, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl, l, l-dimethylethyl, n-pentyl, 1-methylbutyl, 2 -Methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1, 2-dimethylpropyl, 2,2-dimethylpropyl, 1 -ethylpropyl, n-hexyl, 1 - Methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, l, l-dimethylbutyl, 1,2-dimethylbutyl, l, 3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3- dimethylbutyl,
- Pentafluoroethylthio phenyl, phenyloxy, p-Cl-phenyloxy, thiophene-2-yl, thiophene-3-yl, pyridin-2-yl, pyridin-3-yl, pyridin-4-yl, furan-2-yl, furan 3-yl, cyclopropylmethyl,
- R 3 for hydroxy, methylcarbonyloxy, ethylcarbonyloxy, n-propylcarbonyloxy, 1 -
- R 4 represents hydrogen
- X and Y independently of one another represent CH or the grouping CR 1 , where
- X stands for CH if Y stands for the grouping CR 1 and
- X stands for the grouping CR 1 , if Y stands for CH,
- R 1 for chlorine, bromine, methyl, ethyl, 1-methylethyl, l, l-dimethylethyl, methoxy, ethoxy,
- R 3 for hydroxy, methylcarbonyloxy, ethylcarbonyloxy, n-propylcarbonyloxy, 1 -
- R 4 represents hydrogen
- Q stands for one of the groupings Q-1 .1 to Q-1 .68 specifically mentioned in the table above.
- alkylsulfonyl alone or as part of a chemical group - stands for straight-chain or branched alkylsulfonyl, preferably with 1 to 8, or with 1 to 6
- Carbon atoms for example (but not limited to) (Ci-C 6 ) alkylsulfonyl such as methylsulfonyl, ethylsulfonyl, propylsulfonyl, 1-methylethylsulfonyl, butylsulfonyl, 1-methylpropylsulfonyl, 2-methylpropylsulfonyl, l, l-dimethylethylsulfonyl, pentyl 1-methylbutylsulfonyl, 2-methylbutylsulfonyl, 3-methylbutylsulfonyl, l, l-dimethylpropylsulfonyl, 1, 2-dimethylpropylsulfonyl, 2,2-dimethylpropylsulfonyl, 1-ethylpropylsulfonyl, hexylsulfonylsulfonyl, 1-methyls
- heteroarylsulfonyl represents optionally substituted pyridylsulfonyl
- Heteroarylsulfonyl here in particular optionally substituted quinolinylsulfonyl, for example substituted by fluorine, chlorine, bromine, iodine, cyano, nitro, alkyl, haloalkyl, haloalkoxy, amino, alkylamino, alkylcarbonylamino, dialkylamino or alkoxy groups.
- alkylthio alone or as part of a chemical group - stands for straight-chain or branched S-alkyl, preferably with 1 to 8, or with 1 to 6
- Carbon atoms such as (Ci-Cio) -, (C I -C ⁇ ) - or (Ci-C i) -alkylthio, for example (but not limited to) (Ci-C 6 ) -alkylthio such as methylthio, ethylthio, propylthio, 1-methylethylthio, butylthio, 1-methylpropylthio, 2-methylpropylthio, l, l-dimethylethylthio, pentylthio, 1-methylbutylthio, 2-methylbutylthio, 3-methylbutylthio, l, l-dimethylpropylthio, 1, 2-dimethylpropylthio, 2,2- Dimethylpropylthio, 1-ethylpropylthio, hexylthio, 1-methylpentylthio, 2-methylpentylthio, 3-methylpentylthio, 4-methylpent
- alkenylthio means an alkenyl radical bonded via a sulfur atom
- alkynylthio means an alkynyl radical bonded via a sulfur atom
- cycloalkylthio means a cycloalkyl radical bonded via a sulfur atom
- cycloalkenylthio means one via a
- (but not limited to) (Ci-C 6 ) alkylsulfinyl such as methylsulfinyl, ethylsulfinyl, propylsulfinyl, 1-methylethylsulfinyl, butylsulfinyl, 1-methylpropylsulfinyl, 2-methylpropylsulfinyl,
- Alkoxy means an alkyl radical bonded via an oxygen atom, eg. B. (but not limited to) (Ci-C 6 ) alkoxy such as methoxy, ethoxy, propoxy, 1-methylethoxy, butoxy, 1-methylpropoxy, 2-methylpropoxy, l, l-dimethylethoxy, pentoxy, 1-methylbutoxy, 2 -Methylbutoxy, 3-methylbutoxy, 1,1-dimethylpropoxy, 1, 2-dimethylpropoxy, 2,2-dimethylpropoxy, 1-ethylpropoxy, hexoxy, 1 - methylpentoxy, 2-methylpentoxy, 3-methylpentoxy, 4-methylpentoxy, l, l -Dimethylbutoxy, l, 2-dimethylbutoxy, l, 3-dimethylbutoxy, 2,2-dimethylbutoxy, 2,3-dimethylbutoxy, 3,3-dimethylbutoxy, 1-ethylbutoxy, 2-ethylbutoxy, l
- Alkenyloxy means an alkenyl radical bonded via an oxygen atom
- alkynyloxy means an alkynyl radical bonded via an oxygen atom, such as (C2-C10) -, (C2-C6) - or (C2-C 4 ) -alkenoxy or (C3-C10) -, ( C3-C6) or (C3-C 4 ) alkynoxy.
- Cycloalkyloxy means a cycloalkyl radical bonded via an oxygen atom and cycloalkenyloxy means a cycloalkenyl radical bonded via an oxygen atom.
- the number of carbon atoms refers to the alkyl radical in the
- the number of carbon atoms relates to the alkenyl or alkynyl radical in the alkenyl or alkynylcarbonyl group.
- the number of carbon atoms relates to the alkyl radical in the alkoxycarbonyl group.
- the number of carbon atoms relates to the alkenyl or alkynyl radical in the alkene or alkynyloxycarbonyl group.
- the number of carbon atoms relates to the alkyl radical in the alkylcarbonyloxy group.
- the number of carbon atoms relates to the alkenyl or alkynyl radical in the alkenyl or alkynylcarbonyloxy group.
- aryl means an optionally substituted mono-, bi- or polycyclic aromatic system with preferably 6 to 14, in particular 6 to 10 ring carbon atoms, for example phenyl, naphthyl, anthryl, phenanthrenyl, and the like, preferably phenyl.
- optionally substituted aryl also includes multi-cyclic systems such as
- Preferred aryl substituents here are, for example, hydrogen, halogen, alkyl, cycloalkyl, cycloalkylalkyl, cycloalkenyl, halocycloalkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, heteroaryl, heteroarylalkyl, heterocyclyl, heterocyclylalkyl, alkoxyalkyl, alkylthio, haloalkyl, haloalkyl, haloalkyl Haloalkoxy, cycloalkoxy, cycloalkylalkoxy, aryloxy, heteroraryloxy, alkoxyalkoxy, alkynylalkoxy, alkenyloxy, bis-alkylaminoalkoxy, tris [alkyl] silyl, bis [alkyl] arylsilyl, bis [alkyl] alkylsilyl, tris [alky
- Alkylaminocarbonyl cycloalkylaminocarbonyl, bis-alkylaminocarbonyl, heteroarylalkoxy,
- heterocyclic radical contains at least one heterocyclic ring
- ( carbocyclic ring in which at least one C atom is replaced by a hetero atom, preferably by a hetero atom from the group N, O, S, P) which is saturated, unsaturated, partially saturated or heteroaromatic and can be unsubstituted or substituted, where the binding site is located on a ring atom.
- the heterocyclyl radical or the heterocyclic ring is optionally substituted, it can be fused with other carbocyclic or heterocyclic rings.
- heterocyclyl In the case of optionally substituted heterocyclyl, multi-cyclic systems are also included, such as 8-aza-bicyclo [3.2.l] octanyl, 8-aza- bicyclo [2.2.2] octanyl or l-azabicyclo [2.2.l] heptyl. In the case of optionally substituted heterocyclyl also
- the heterocyclic ring preferably contains 3 to 9 ring atoms, in particular 3 to 6 ring atoms, and one or more, preferably 1 to 4, in particular 1, 2 or 3, heteroatoms in the heterocyclic ring, preferably from the group N, O, and S, but not two
- Oxygen atoms are said to be directly adjacent, for example with a heteroatom from the group N, O and S 1- or 2- or 3-pyrrolidinyl, 3,4-dihydro-2H-pyrrol-2- or 3-yl, 2,3- dihydro-lH-pyrrole
- 3-ring and 4-ring heterocycles are, for example, 1- or 2-aziridinyl, oxiranyl, thiiranyl, 1- or 2- or 3-azetidinyl,
- heterocyclyl are a partially or fully hydrogenated heterocyclic radical with two heteroatoms from the group N, O and S, such as 1- or 2- or 3- or 4-pyrazolidinyl; 4,5-dihydro-3H-pyrazol-3- or 4- or 5-yl; 4,5-dihydro-1H-pyrazole-l- or 3- or 4- or 5-yl; 2,3-dihydro-1H-pyrazole-1 or 2- or
- 1,2-dithiolan-3- or 4-yl 1,2-dithiolan-3- or 4-yl; 3H-l, 2-dithiol-3- or 4- or 5-yl; l, 3-dithiolan-2- or 4-yl; l, 3-dithiol-2- or 4-yl; 1,2-dithian-3- or 4-yl; 3,4-dihydro-l, 2-dithiin-3- or 4- or 5- or 6-yl; 3,6-dihydro-
- 1,2-dithiin-3- or 4-yl 1,2-dithiin-3- or 4-yl; 1,2-dithiin-3- or 4-yl; l, 3-dithian-2- or 4- or 5-yl; 4H-l, 3-dithiin-2- or 4- or 5- or 6-yl; Isoxazolidin-2- or 3- or 4- or 5-yl; 2,3-dihydroisoxazole-2- or 3- or
- 6- or 7-yl 2,5-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 2,7-dihydro-l, 3-oxazepine 2- or 4- or 5- or 6- or 7-yl; 4,5-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 4,7-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 6,7-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 1, 4-oxazepan-2- or 3- or 5- or 6- or 7-yl; 2,3,4,5-tetrahydro-l, 4-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2, 3, 4, 7-tetrahydro-l, 4-oxazepin-2
- heterocyclyl are a partially or fully hydrogenated heterocyclic radical with 3 heteroatoms from the group N, O and S, such as l, 4,2-dioxazolidin-2- or 3- or 5-yl; l, 4,2-dioxazol-3- or 5-yl; 1, 4,2-dioxazinan-2- or -3- or 5- or 6-yl; 5,6-dihydro-l, 4,2-dioxazin-3- or 5- or 6-yl; l, 4,2-dioxazin-3- or 5- or 6-yl; l, 4,2-dioxazepan-2- or 3- or 5- or 6- or 7-yl; 6,7-dihydro-5H-l, 4,2-dioxazepin-3- or 5- or 6- or 7-yl; 2,3-dihydro-7H-l, 4,2-dioxazepin-2- or 3- or 5- or 6- or 7-yl; 2,3-dihydro-5H-l, 4,2-dioxa
- heterocycles listed above are preferably, for example, by hydrogen, halogen, alkyl, haloalkyl, hydroxy, alkoxy, cycloalkoxy, aryloxy, alkoxyalkyl, alkoxyalkoxy, cycloalkyl,
- Alkylaminocarbonyl bis-alkylaminocarbonyl, cycloalkylaminocarbonyl,
- the oxo group as a substituent on a ring carbon atom then means, for example, a carbonyl group in the heterocyclic ring. This preferably also includes lactones and lactams.
- the oxo group can also occur on the hetero ring atoms, which can exist in different oxidation states, for example in the case of N and S, and then form, for example, the divalent groups N (O), S (O) (also SO for short) and S (0) 2 (also briefly SO2) in the heterocyclic ring.
- N (O), S (O) also SO for short
- S (0) 2 also briefly SO2
- heteroaryls are, for example, 1H-pyrrol-l-yl; lH-pyrrol-2-yl; lH-pyrrole
- Carbon atoms are part of a further aromatic ring, so they are fused heteroaromatic systems, such as benzo-fused or multiply fused heteroaromatics.
- quinolines e.g. quinolin-2-yl, quinolin-3-yl, quinolin-4-yl, quinolin-5-yl, quinolin-6-yl, quinolin-7-yl, quinolin-8-yl
- Isoquinolines e.g. quinolin-2-yl, quinolin-3-yl, quinolin-4-yl, quinolin-5-yl, quinolin-6-yl, quinolin-7-yl, quinolin-8-yl
- Isoquinolines e.g.
- heteroaryl are also 5- or 6-membered benzo-fused rings from the group lH-indol-l-yl, lH-indol-2-yl, lH-indol-3-yl, lH-indol-4-yl, lH- Indol-5-yl, 1H-indol-6-yl, lH-indol-7-yl, l-benzofuran-2-yl, l-benzofuran-3-yl, l-benzofuran-4-yl, l-benzofuran 5- yl, l-benzofuran-6-yl, l-benzofuran-7-yl, l-benzothiophene-2-yl, l-benzothiophene-3-yl, 1-benzothiophene-4-yl, l-benzothiophene-5- yl, l-benzothiophene-6-yl, l-benz
- halogen means, for example, fluorine, chlorine, bromine or iodine.
- halogen means for example a fluorine, chlorine, bromine or iodine atom.
- alkyl means a straight-chain or branched, open-chain, saturated hydrocarbon radical which is optionally substituted one or more times and is referred to in the latter case as “substituted alkyl”.
- Preferred substituents are halogen atoms, alkoxy, haloalkoxy, cyano, alkylthio, haloalkylthio, amino or nitro groups; methoxy, methyl, fluoroalkyl, cyano, nitro, fluorine, chlorine, bromine or iodine are particularly preferred.
- the prefix "bis” also includes the combination of different alkyl residues, e.g. B. methyl (ethyl) or ethyl (methyl).
- Haloalkyl means alkyl or alkenyl or alkynyl which is partially or completely substituted by identical or different halogen atoms, e.g. monohaloalkyl
- ( Monohalogenalkyl) such as B. CH 2 CH 2 CI, CH 2 CH 2 Br, CHCICH 3 , CH 2 CI, CH 2 F; Perhaloalkyl such as B. CCI3, CCIF 2 , CFC1 2 , CF 2 CC1F 2 , CF 2 CCIFCF3; Polyhaloalkyl such as B. CH 2 CHFC1, CF 2 CC1FH, CF 2 CBrFH, CH 2 CF 3 ;
- perhaloalkyl also includes the term perfluoroalkyl.
- Partially fluorinated alkyl means a straight-chain or branched, saturated hydrocarbon which is substituted one or more times by fluorine, and the corresponding fluorine atoms can be located as substituents on one or more different carbon atoms of the straight-chain or branched hydrocarbon chains, such as, for example, B. CHFCH 3 , CH 2 CH 2 F, CH 2 CH 2 CF 3 , CHF 2 , CH 2 F, CHFCF 2 CF 3
- Partially fluorinated haloalkyl means a straight-chain or branched, saturated
- Hydrocarbon which is substituted by various halogen atoms with at least one fluorine atom, all other halogen atoms which may be present being selected from the group consisting of fluorine, chlorine or bromine, iodine.
- the corresponding halogen atoms can be located as substituents on one or more different carbon atoms of the straight-chain or branched hydrocarbon chain.
- Partially fluorinated haloalkyl also includes the complete substitution of the straight or branched ketene by halogen with the participation of at least one fluorine atom.
- Haloalkoxy is, for example, OCF 3 , OCHF 2 , OCH 2 F, OCF 2 CF 3 , OCH 2 CF 3 and OCH 2 CH 2 CI; The same applies to haloalkenyl and other halogen-substituted radicals.
- (Ci-C4) -alkyl mentioned here by way of example means a shorthand notation for straight-chain or branched alkyl having one to 4 carbon atoms corresponding to the
- Range for C atoms, d. H. includes the residues methyl, ethyl, 1-propyl, 2-propyl, 1-butyl,
- hydrocarbon radicals such as alkyl, alkenyl and alkynyl radicals, even in composite radicals, are the lower carbon skeletons, e.g. with 1 to 6 carbon atoms or in the case of unsaturated groups with 2 to 6 carbon atoms, preferred.
- Alkenyl and alkynyl radicals have the meaning of the possible unsaturated radicals corresponding to the alkyl radicals, at least one double bond or triple bond being present. Residues with a double bond or
- alkenyl also includes straight-chain or branched open-chain ones
- Hydrocarbon radicals with more than one double bond such as 1,3-butadienyl and 1,4-pentadienyl, but also allenyl or cumulenyl radicals with one or more cumulative double bonds, such as, for example, allenyl (1,2-propadienyl), 1,1 2-butadienyl and l, 2,3-pentatrienyl.
- Alkenyl means e.g.
- Vinyl which may optionally be substituted by further alkyl radicals, for example (but not limited to) (C2-C6) alkenyl such as ethenyl, l-propenyl, 2-propenyl, 1-methylethenyl, l-butenyl, 2-butenyl , 3-butenyl, 1-methyl-1-propenyl, 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4 -Pentenyl, 1-methyl-1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl , l-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1, 1-dimethyl-2-propenyl, 1, 2-dimethyl-l-propen
- alkynyl also includes straight-chain or branched open-chain ones
- C2-C6 alkynyl means e.g. Ethynyl, l-propynyl, 2-propynyl, l-butynyl, 2-butynyl, 3-butynyl, 1-methyl-2-propynyl, l-pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, l-methyl- 2-butynyl, 1-methyl-3-butynyl, 2-methyl-3-butynyl, 3-methyl-1-butynyl, 1, 1-dimethyl-2-propynyl, 1-ethyl-2-propynyl, 1-hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, 5-hexynyl, 1-methyl-2-pentynyl, 1-methyl-2-pentynyl
- cycloalkyl means a carbocyclic, saturated ring system with preferably 3-8 ring C atoms, e.g. Cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl, which is optionally further substituted, preferably by hydrogen, alkyl, alkoxy, cyano, nitro, alkylthio, haloalkylthio, halogen, alkenyl, alkynyl, haloalkyl, AMino, alkylamino, bisalkylamino, alkocycarbonyl,
- Cycloalkylaminocarbonyl In the case of optionally substituted cycloalkyl, cyclic systems with substituents are included, substituents also having a double bond on
- Cycloalkyl radical e.g. B. an alkylidene group such as methylidene are included.
- alkylidene group such as methylidene
- multi-cyclic aliphatic systems are also included, such as, for example, bicyclo [1.0] butan-1-yl, bicyclo [1.0] butan-2-yl, bicyclo [2.1.0] pentan-1 - yl, bicyclo [l.
- (C3-C7) cycloalkyl means a shorthand notation for cycloalkyl of three to seven
- Carbon atoms according to the range for carbon atoms.
- spirocyclic aliphatic systems are also included, such as, for example, spiro [2.2] pent-l-yl, spiro [2.3] hex-l-yl, spiro [2.3] hex-4-yl, 3-spiro [2.3] hex-5-yl,
- Cycloalkenyl means a carbocyclic, non-aromatic, partially unsaturated ring system with preferably 4-8 C atoms, for example 1-cyclobutenyl, 2-cyclobutenyl, 1-cyclopentenyl, 2-cyclopentenyl, 3-cyclopentenyl, or 1-cyclohexenyl, 2- Cyclohexenyl, 3-cyclohexenyl, l, 3-cyclohexadienyl or 1, 4-cyclohexadienyl, where also substituents with a double bond on the cycloalkenyl radical, e.g. B. an alkylidene group such as methylidene are included.
- substituents with a double bond on the cycloalkenyl radical e.g.
- an alkylidene group such as methylidene are included.
- alkylidene e.g. B. also in the form (Ci-Cio) alkylidene, means the remainder of a straight-chain or branched open-chain hydrocarbon radical which is bonded via a double bond.
- Cycloalkylidene means a
- Cycloalkylalkyloxy means a cycloalkylalkyl radical bonded via an oxygen atom and “arylalkyloxy” means an arylalkyl radical bonded via an oxygen atom.
- Alkoxyalkyl stands for an alkoxy radical bonded via an alkyl group and "alkoxyalkoxy” means an alkoxyalkyl radical bonded via an oxygen atom, e.g. (but not limited to) methoxymethoxy, methoxyethoxy, ethoxyethoxy, methoxy-n-propyloxy.
- Alkylthioalkyl stands for an alkylthio radical bonded via an alkyl group
- Alkylthioalkylthio means an alkylthioalkyl radical bonded via an oxygen atom.
- Arylalkoxyalkyl stands for an aryloxy radical bonded via an alkyl group
- Heteroaryloxyalkyl means a heteroaryloxy radical bonded via an alkyl group.
- Haloalkoxyalkyl stands for a bound haloalkoxy radical and “Haloalkylthioalkyl” means a haloalkylthio radical bound via an alkyl group.
- Arylalkyl stands for an aryl radical bonded via an alkyl group
- heteroarylalkyl means a heteroaryl radical bonded via an alkyl group
- heterocyclylalkyl means a heterocyclyl radical bonded via an alkyl group.
- Cycloalkylalkyl stands for a cycloalkyl radical bonded via an alkyl group, eg. B.
- cyclopropylmethyl (but not limited to) cyclopropylmethyl, cyclobutylmethyl, cyclopentylmethyl, cyclohexylmethyl, 1 - cyclopropyleth-l -yl, 2-cyclopropyleth-l-yl, l-cyclopropylprop-l-yl, 3-cyclopropylprop-l-yl.
- Arylalkenyl stands for an aryl radical bonded via an alkenyl group
- heteroarylalkenyl means a heteroaryl radical bonded via an alkenyl group
- heterocyclylalkenyl means a heterocyclyl radical bonded via an alkenyl group
- Arylalkynyl stands for an aryl group bonded via an alkynyl group
- heteroarylalkynyl means a heteroaryl group bonded via an alkynyl group
- heterocyclylalkynyl means a heterocyclyl group bonded via an alkynyl group.
- haloalkylthio alone or as part of a chemical group - stands for straight-chain or branched S -haloalkyl, preferably with 1 to 8, or with 1 to 6
- Carbon atoms such as (Ci-Cs) -, (C I -C ⁇ ) - or (Ci-C4) -haloalkylthio, for example (but not limited to) trifluoromethylthio, pentafluoroethylthio, difluoromethyl, 2,2-difluoroeth-1-ylthio, 2,2,2-difluoroeth-l-ylthio, 3,3,3-prop-l-ylthio.
- Halocycloalkyl and “Halocycloalkenyl” mean by identical or different halogen atoms, such as. B. F, CI and Br, or by haloalkyl, such as. B. trifluoromethyl or difluoromethyl partially or fully substituted cycloalkyl or cycloalkenyl, e.g.
- l-fluorocycloprop-l-yl 2-fluorocycloprop-l-yl, 2,2-difluorocycloprop-l-yl, l-fluorocyclobut-l-yl, l-trifluoromethylcycloprop-l-yl, 2-trifluoromethylcycloprop-1-yl, 1-chloro-cycloprop-1-yl, 2-chlorocycloprop-1-yl, 2,2-dichlorocycloprop-1-yl, 3,3-difluorocyclobutyl.
- the compounds of the general formula (I) can be present as stereoisomers.
- the possible stereoisomers defined by their specific spatial shape, such as enantiomers, diastereomers, Z and E isomers, are all encompassed by the formula (I). If, for example, one or more alkenyl groups are present, diastereomers (Z and E Isomers) occur. For example, if one or more asymmetric carbon atoms are present, enantiomers and diastereomers can occur.
- Stereoisomers can be obtained from the mixtures obtained in the preparation by customary separation methods.
- the chromatographic separation can be carried out on an analytical scale to determine the enantiomeric excess or the diastereomeric excess, as well as on a preparative scale for the production of test samples for biological testing.
- stereoisomers can be produced selectively by using stereoselective reactions using optically active starting materials and / or auxiliary substances.
- the invention thus also relates to all stereoisomers which are encompassed by the general formula (I) but are not specified with their specific stereoform, and to mixtures thereof.
- the cleaning can also be carried out by
- Methods for isolation, purification and stereoisomer separation of compounds of the general formula (I) are those which are generally known to the person skilled in the art from analogous cases, e.g. by physical processes such as crystallization, chromatography processes, especially column chromatography and HPLC (high pressure liquid chromatography), distillation, optionally under reduced pressure, extraction and other processes, any remaining mixtures can usually be separated by chromatographic separation, e.g. on chiral solid phases.
- the substituted thiazolylpyrrolones of the general formula (I) according to the invention can be prepared starting from known processes.
- the used and examined Synthesis routes are based on commercially available or easily manufactured substituted 2-aminothiazoles and on appropriately substituted furanones or furandions.
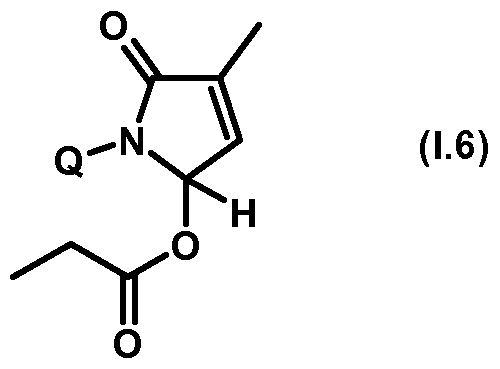
- the compounds of the general formula (Ib) according to the invention are synthesized via a reaction of a hydroxypyrrolone of the general formula (Ia) with a halogen formic acid derivative of the general formula (II).
- the reaction preferably takes place in the temperature range between -20 ° C and 80 ° C, in an adequate solvent such as dichloromethane, acetonitrile, NN-
- the compounds of the general formula (Ic) according to the invention are synthesized by reacting a hydroxypyrrolone of the general formula (Ia) with an acid halide derivative of the general formula (III).
- the reaction preferably takes place in the temperature range between -20 ° C and 80 ° C, in an adequate solvent such as dichloromethane, acetonitrile, N, N-dimethylformamide or ethyl acetate and in the presence of a base such as triethylamine, V, V - diisopropylethylamine or 1,8-diazabicyclo [5.4.0] undec-7-cene instead (see Scheme 2).
- the synthesis of the compounds of the general formula (Id) according to the invention takes place via a reaction of a hydroxypyrrolone of the general formula (Ia) with an isocyanate of the general formula (IV).
- the reaction preferably takes place in the temperature range between -20 ° C and 120 ° C, in an adequate solvent such as dichloromethane, acetonitrile,, V,, V-D imct hy 1 - form am id or ethyl acetate and in the presence of a base such as triethylamine, / V,, V- diisopro py 1 cthy 1 amine or l, 8-diazabicyclo [5.4.0] undec-7-cene instead (see Scheme 3).
- R (Ci-C 4 ) alkyl
- the compounds of the general formula (Ia) and (Ie) according to the invention are synthesized by reducing a substituted maleimide of the general formula (V) with a
- Reducing agents such as sodium hydride, lithium aluminum hydride, sodium borohydride or other hydrogen-generating metal hydrides.
- a transition metal-mediated hydrogenation can also be carried out (cf. CH633678, DE2247266, WO2015 / 018434). Since the groups X and Y are different, the reduction of the carbonyl group can be mixtures of
- the reaction preferably takes place in the temperature range between -20 ° C and 120 ° C in an adequate solvent such as tetrahydrofuran, methanol or ethanol.
- Substituted maleimides of the general formula (V) can be obtained by condensation of a substituted aminothiazole of the general formula (VI) with a substituted furan-2,5-dione of the general formula (VII) in the presence of a Broensted acid such as acetic acid or
- the monosubstituted furan-2,5-diones (VII), which are also referred to as maleic anhydrides, can be prepared via synthesis instructions known from the literature (cf. J. Chem. Soc., Perkin Trans. 1, 1982, 215; EP1426365; J. Org Chem. 1998, 63, 2646; WO2015 / 018431; Tetrahedron 2012, 68, 5863; Russian J. Org. Chem. 2007, 43, 801), for example compounds (VII. A) and (Vll.b) im
- Mono-substituted thiazoles can be prepared by literature known synthetic procedures (see Bioorg & MedChem Let 2004, 14, 5521-5525;.... Heterocyclic Chemistry, Ed JA Joule and K. Mills, 4th Edition, Blackwell Publishing, 2000. 418-420; W02008 / 55840; US2012 / 22067;
- the compound (Vl.a) can be made from a haloketone (X) and thiourea (XI).
- the aminothiazole (Vl.b) can be prepared from the haloaldehyde (XII) and thiourea (XI) (Scheme 7).
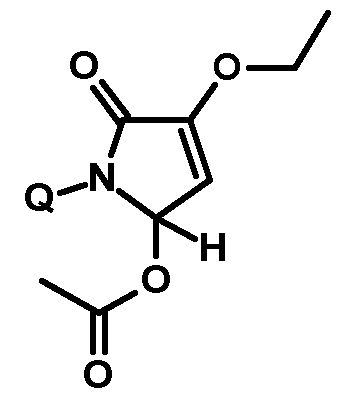
- Acetic anhydride using a suitable base forms the monosubstituted thiazolylpyrrolone of the general formula (XV) with O-acetyl group, which can be obtained by heating under acidic conditions (e.g. in a mixture of acetic acid and Water) can be converted into the corresponding monosubstituted thiazolylpyrrolone of the general formula (Ia) with a free OH group.
- a suitable base e.g. pyridine
- the reaction preferably takes place in the temperature range between -20 ° C and 80 ° C, in an adequate solvent such as dichloromethane, acetonitrile, NN-dimethylformamide or ethyl acetate and in the presence of a base such as triethylamine, V, V-diisopropylethylamine or 1,8-diazabicyclo [5.4.0] undec-7-cene instead (see Scheme 9).
- Synthesis stage 1 2-bromo-l- (l -methylcyclopropyl) ethanone l- (l-Methylcyclopropyl) ethanone (6.00 g, 61.13 mmol, 1.0 equiv.) was dissolved in methanol (40 mL) and cooled to approx. 4 ° C with an ice bath. Bromine (3.15 mL, 61.13 mmol,
- reaction temperature does not exceed 10 ° C.
- the reaction mixture was stirred at about 4 ° C. for 45 min and then water (10 mL) was added. The mixture was then stirred at room temperature for 1 h, during which the reaction solution decolorized.
- the reaction mixture was diluted with water and diethyl ether was added and extracted. The combined organic phases were then dried over magnesium sulfate, filtered and the solvent was distilled off via a short path column. The distillation still contained the desired product 2-bromo-1- (1-methylcyclopropyl) ethanone in the form of a slightly yellow liquid (11.00 g, 90%, 91% of theory). The crude product was used in the next step without further purification.
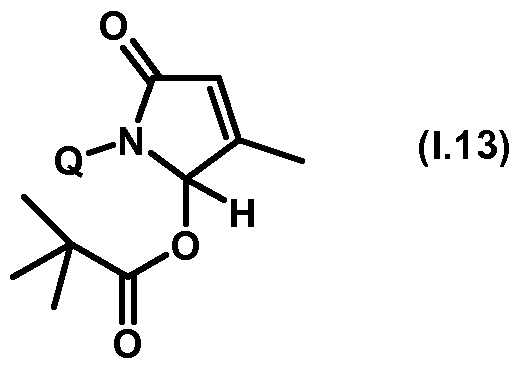
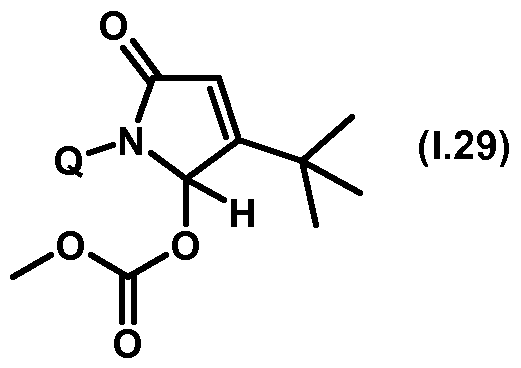
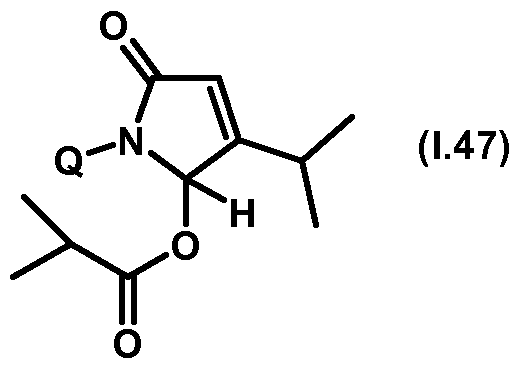
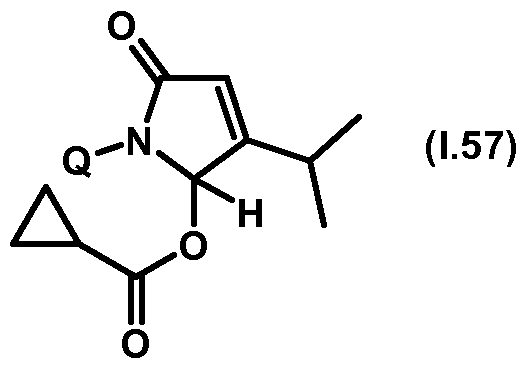
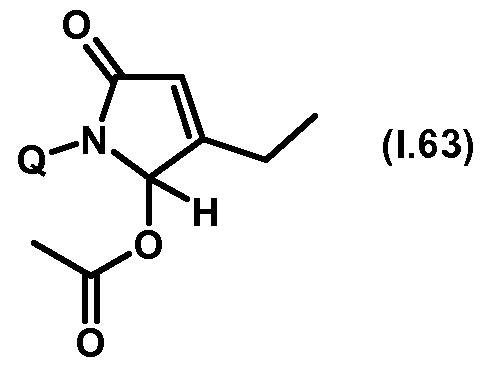
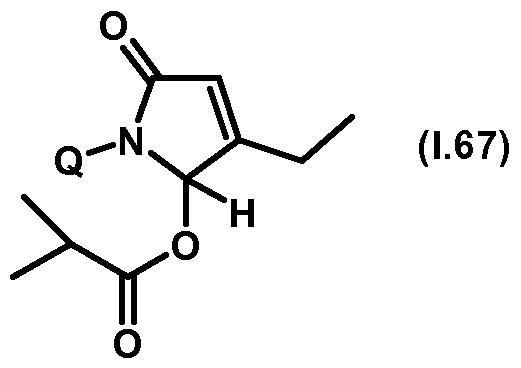
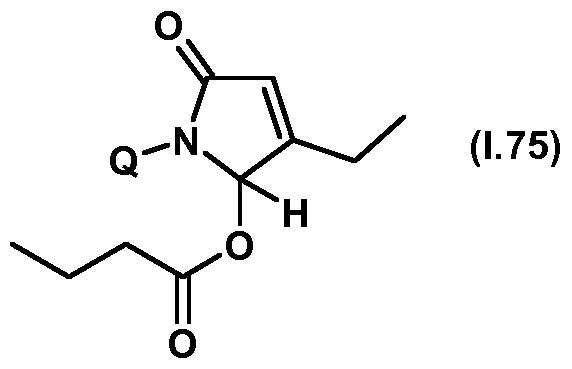
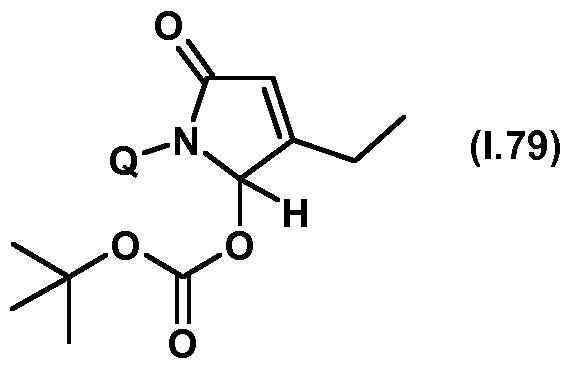
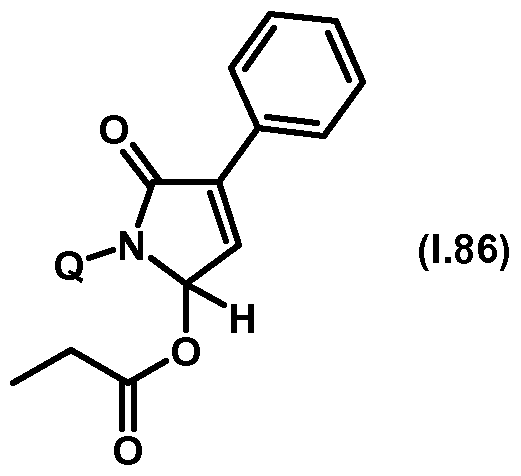
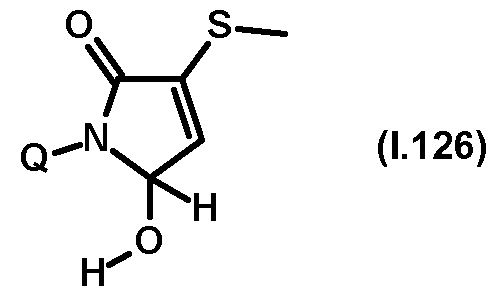
- Thiazolylpyrrolone gives the compounds mentioned below. If in Table 1 a structural element is defined by a structural formula which contains a dashed line, this dashed line means that the group in question is connected to the rest of the molecule at this position.
- Table 1.1 Preferred compounds of the formula (1.1) are the compounds 1.1-1 to 1.1-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.1-1 to 1.1- 68 of Table 1.1 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.2 Preferred compounds of the formula (1.2) are the compounds 1.2-1 to 1.2-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.2-1 to 1.2- 68 of table 1.2 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.3 Preferred compounds of the formula (1.3) are the compounds 1.3-1 to 1.3-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.3-1 to 1.3- 68 of table 1.3 are thus by the meaning of the respective entries no. 1 to 68 defined for Q of Table 1.
- Table 1.4 Preferred compounds of the formula (1.4) are the compounds 1.4-1 to 1.4-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.4-1 to 1.4- 68 of Table 1.4 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.5 Preferred compounds of the formula (1.5) are the compounds 1.5-1 to 1.5-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.5-1 to 1.5- 68 of Table 1.5 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.6 Preferred compounds of the formula (1.6) are the compounds 1.6-1 to 1.6-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.6-1 to 1.6- 68 of table 1.6 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.7 Preferred compounds of the formula (1.7) are the compounds 1.7-1 to 1.7-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.7-1 to 1.7- 68 of table 1.7 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.8 Preferred compounds of the formula (1.8) are the compounds 1.8-1 to 1.8-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.8-1 to 1.8- 68 of Table 1.8 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.9 Preferred compounds of the formula (1.9) are the compounds 1.9-1 to 1.9-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.9-1 to 1.9- 68 of Table 1.9 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.10 Preferred compounds of the formula (1.10) are the compounds 1.10-1 to 1.10-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.10-1 to 1.10-68 of table 1.10 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.11 Preferred compounds of the formula (1.11) are the compounds 1.11-1 to 1.11-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.11-1 to 1.11-68 of table 1.11 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.12 Preferred compounds of the formula (1.12) are the compounds 1.12-1 to 1.12-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.12-1 to 1.12-68 of table 1.12 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.13 Preferred compounds of the formula (1.13) are the compounds 1.13-1 to 1.13-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.13-1 to 1.13-68 of table 1.13 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.14 Preferred compounds of the formula (1.14) are the compounds 1.14-1 to 1.14-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.14-1 to 1.14-68 of table 1.14 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.15 Preferred compounds of the formula (1.15) are the compounds 1.15-1 to 1.15-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.15-1 to 1.15-68 of table 1.15 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.16 Preferred compounds of the formula (1.16) are the compounds 1.16-1 to 1.16-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.16-1 to 1.16-68 of table 1.16 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.17 Preferred compounds of the formula (1.17) are the compounds 1.17-1 to 1.17-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.17-1 to 1.17-68 of table 1.17 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.18 Preferred compounds of the formula (1.18) are the compounds 1.18-1 to 1.18-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.18-1 to 1.18-68 of table 1.18 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.19 Preferred compounds of the formula (1.19) are the compounds 1.19-1 to 1.19-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.19-1 to 1.19-68 of table 1.19 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.20 Preferred compounds of the formula (1.20) are the compounds 1.20-1 to 1.20-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.20-1 to 1.20-68 of Table 1.20 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.21 Preferred compounds of the formula (1.21) are the compounds 1.21-1 to 1.21-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.21-1 to 1.21-68 of table 1.21 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.22 Preferred compounds of the formula (1.22) are the compounds 1.22-1 to 1.22-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.22-1 to 1.22-68 of table 1.22 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.23 Preferred compounds of the formula (1.23) are the compounds 1.23-1 to 1.23-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.23-1 to 1.23-68 of table 1.23 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.24 Preferred compounds of the formula (1.24) are the compounds 1.24-1 to 1.24-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.24-1 to 1.24-68 of table 1.24 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.25 Preferred compounds of the formula (1.25) are the compounds 1.25-1 to 1.25-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.25-1 to 1.25-68 of table 1.25 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.26 Preferred compounds of the formula (1.26) are the compounds 1.26-1 to 1.26-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.26-1 to 1.26-68 of table 1.26 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.27 Preferred compounds of the formula (1.27) are the compounds 1.27-1 to 1.27-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.27-1 to 1.27-68 of table 1.27 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.28 Preferred compounds of the formula (1.28) are the compounds 1.28-1 to 1.28-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.28-1 to 1.28-68 of table 1.28 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.29 Preferred compounds of the formula (1.29) are the compounds 1.29-1 to 1.29-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.29-1 to 1.29-68 of table 1.29 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.30 Preferred compounds of the formula (1.30) are the compounds 1.30-1 to 1.30-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.30-1 to 1.30-68 of table 1.30 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.31 Preferred compounds of the formula (1.31) are the compounds 1.31-1 to 1.31-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.31-1 to 1.31-68 of table 1.31 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.32 Preferred compounds of the formula (1.32) are the compounds 1.32-1 to 1.32-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.32-1 to 1.32-68 of table 1.32 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.33 Preferred compounds of the formula (1.33) are the compounds 1.33-1 to 1.33-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.33-1 to 1.33-68 of table 1.33 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.34 Preferred compounds of the formula (1.34) are the compounds 1.34-1 to 1.34-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.34-1 to 1.34-68 of table 1.34 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.35 Preferred compounds of the formula (1.35) are the compounds 1.35-1 to 1.35-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.35-1 to 1.35-68 of table 1.35 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.36 Preferred compounds of the formula (1.36) are the compounds 1.36-1 to 1.36-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.36-1 to 1.36-68 of table 1.36 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.37 Preferred compounds of the formula (1.37) are the compounds 1.37-1 to 1.37-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.37-1 to 1.37-68 of table 1.37 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.38 Preferred compounds of the formula (1.38) are the compounds 1.38-1 to 1.38-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.38-1 to 1.38-68 of table 1.38 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.39 Preferred compounds of the formula (1.39) are the compounds 1.39-1 to 1.39-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.39-1 to 1.39-68 of table 1.39 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.40 Preferred compounds of the formula (1.40) are the compounds 1.40-1 to 1.40-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.40-1 to 1.40-68 of table 1.40 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.41 Preferred compounds of the formula (1.41) are the compounds 1.41-1 to 1.41-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.41-1 to 1.41-68 of Table 1.41 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.42 Preferred compounds of the formula (1.42) are the compounds 1.42-1 to 1.42-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.42-1 to 1.42-68 of table 1.42 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.43 Preferred compounds of the formula (1.43) are the compounds 1.43-1 to 1.43-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.43-1 to 1.43-68 of table 1.43 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.44 Preferred compounds of the formula (1.44) are the compounds 1.44-1 to 1.44-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.44-1 to 1.44-68 in Table 1.44 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.45 Preferred compounds of the formula (1.45) are the compounds 1.45-1 to 1.45-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.45-1 to 1.45-68 of table 1.45 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.46 Preferred compounds of the formula (1.46) are the compounds 1.46-1 to 1.46-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.46-1 to 1.46-68 in Table 1.46 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.47 Preferred compounds of the formula (1.47) are the compounds 1.47-1 to 1.47-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.47-1 to 1.47-68 in Table 1.47 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.48 Preferred compounds of the formula (1.48) are the compounds 1.48-1 to 1.48-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.48-1 to 1.48-68 of table 1.48 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.49 Preferred compounds of the formula (1.49) are the compounds 1.49-1 to 1.49-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.49-1 to 1.49-68 of table 1.49 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.50 Preferred compounds of the formula (1.50) are the compounds 1.50-1 to 1.50-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.50-1 to 1.50-68 of table 1.50 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.51 Preferred compounds of the formula (1.51) are the compounds 1.51-1 to 1.51-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.51-1 to 1.51-68 of table 1.51 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.52 Preferred compounds of the formula (1.52) are the compounds 1.52-1 to 1.52-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.52-1 to 1.52-68 of table 1.52 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.53 Preferred compounds of the formula (1.53) are the compounds 1.53-1 to 1.53-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.53-1 to 1.53-68 of table 1.53 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.54 Preferred compounds of the formula (1.54) are the compounds 1.54-1 to 1.54-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.54-1 to 1.54-68 of table 1.54 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.55 Preferred compounds of the formula (1.55) are the compounds 1.55-1 to 1.55-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.55-1 to 1.55-68 of table 1.55 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.56 Preferred compounds of the formula (1.56) are the compounds 1.56-1 to 1.56-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.56-1 to 1.56-68 of table 1.56 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.57 Preferred compounds of the formula (1.57) are the compounds 1.57-1 to 1.57-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.57-1 to 1.57-68 of table 1.57 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.58 Preferred compounds of the formula (1.58) are the compounds 1.58-1 to 1.58-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.58-1 to 1.58-68 of table 1.58 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.59 Preferred compounds of the formula (1.59) are the compounds 1.59-1 to 1.59-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.59-1 to 1.59-68 of table 1.59 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.60 Preferred compounds of the formula (1.60) are the compounds 1.60-1 to 1.60-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.60-1 to 1.60-68 of table 1.60 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.61 Preferred compounds of the formula (1.61) are the compounds 1.61-1 to 1.61-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.61-1 to 1.61-68 of table 1.61 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.62 Preferred compounds of the formula (1.62) are the compounds 1.62-1 to 1.62-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.62-1 to 1.62-68 of table 1.62 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.63 Preferred compounds of the formula (1.63) are the compounds 1.63-1 to 1.63-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.63-1 to 1.63-68 of table 1.63 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.64 Preferred compounds of the formula (1.64) are the compounds 1.64-1 to 1.64-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.64-1 to 1.64-68 of table 1.64 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.65 Preferred compounds of the formula (1.65) are the compounds 1.65-1 to 1.65-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.65-1 to 1.65-68 of table 1.65 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.66 Preferred compounds of the formula (1.66) are the compounds 1.66-1 to 1.66-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.66-1 to 1.66-68 of table 1.66 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.67 Preferred compounds of the formula (1.67) are the compounds 1.67-1 to 1.67-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.67-1 to 1.67-68 in Table 1.67 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.68 Preferred compounds of the formula (1.68) are the compounds 1.68-1 to 1.68-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.68-1 to 1.68-68 of table 1.68 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.69 Preferred compounds of the formula (1.69) are the compounds 1.69-1 to 1.69-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.69-1 to 1.69-68 of table 1.69 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.70 Preferred compounds of the formula (1.70) are the compounds 1.70-1 to 1.70-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.70-1 to 1.70-68 of table 1.70 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.71 Preferred compounds of the formula (1.71) are the compounds 1.71-1 to 1.71-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.71-1 to 1.71-68 of table 1.71 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.72 Preferred compounds of the formula (1.72) are the compounds 1.72-1 to 1.72-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.72-1 to 1.72-68 of table 1.72 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.73 Preferred compounds of the formula (1.73) are the compounds 1.73-1 to 1.73-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.73-1 to 1.73-68 of table 1.73 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.74 Preferred compounds of the formula (1.74) are the compounds 1.74-1 to 1.74-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.74-1 to 1.74-68 of table 1.74 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.75 Preferred compounds of the formula (1.75) are the compounds 1.75-1 to 1.75-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.75-1 to 1.75-68 of table 1.75 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.76 Preferred compounds of the formula (1.76) are the compounds 1.76-1 to 1.76-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.76-1 to 1.76-68 of table 1.76 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.77 Preferred compounds of the formula (1.77) are the compounds 1.77-1 to 1.77-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.77-1 to 1.77-68 of table 1.77 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.78 Preferred compounds of the formula (1.78) are the compounds 1.78-1 to 1.78-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.78-1 to 1.78-68 of table 1.78 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.79 Preferred compounds of the formula (1.79) are the compounds 1.79-1 to 1.79-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.79-1 to 1.79-68 of table 1.79 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.80 Preferred compounds of the formula (1.80) are the compounds 1.80-1 to 1.80-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.80-1 to 1.80-68 of table 1.80 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.81 Preferred compounds of the formula (1.81) are the compounds 1.81-1 to 1.81-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.81-1 to 1.81-68 of table 1.81 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.82 Preferred compounds of the formula (1.82) are the compounds 1.82-1 to 1.82-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.82-1 to 1.82-68 of table 1.82 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.83 Preferred compounds of the formula (1.83) are the compounds 1.83-1 to 1.83-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.83-1 to 1.83-68 of table 1.83 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.84 Preferred compounds of the formula (1.84) are the compounds 1.84-1 to 1.84-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.84-1 to 1.84-68 of table 1.84 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.85 Preferred compounds of the formula (1.85) are the compounds 1.85-1 to 1.85-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.85-1 to 1.85-68 of table 1.85 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.86 Preferred compounds of the formula (1.86) are the compounds 1.86-1 to 1.86-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.86-1 to 1.86-68 of table 1.86 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.87 Preferred compounds of the formula (1.87) are the compounds 1.87-1 to 1.87-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.87-1 to 1.87-68 of table 1.87 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.88 Preferred compounds of the formula (1.88) are the compounds 1.88-1 to 1.88-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.88-1 to 1.88-68 of table 1.88 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.89 Preferred compounds of the formula (1.89) are the compounds 1.89-1 to 1.89-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.89-1 to 1.89-68 of table 1.89 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.90 Preferred compounds of the formula (1.90) are the compounds 1.90-1 to 1.90-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.90-1 to 1.90-68 of table 1.90 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.91 Preferred compounds of the formula (1.91) are the compounds 1.91-1 to 1.91-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.91-1 to 1.91-68 of table 1.91 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.92 Preferred compounds of the formula (1.92) are the compounds 1.92-1 to 1.92-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.92-1 to 1.92-68 in Table 1.92 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.93 Preferred compounds of the formula (1.93) are the compounds 1.93-1 to 1.93-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.93-1 to 1.93-68 in Table 1.93 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.94 Preferred compounds of the formula (1.94) are the compounds 1.94-1 to 1.94-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.94-1 to 1.94-68 of table 1.94 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.95 Preferred compounds of the formula (1.95) are the compounds 1.95-1 to 1.95-68, in which Q has the meanings of Table 1 given in the respective row. The connections
- Table 1.96 Preferred compounds of the formula (1.96) are the compounds 1.96-1 to 1.96-68, in which Q has the meanings of Table 1 given in the respective row. The connections
- Table 1.97 Preferred compounds of the formula (1.97) are the compounds 1.97-1 to 1.97-68, in which Q has the meanings of Table 1 given in the respective row. The connections
- Table 1.98 Preferred compounds of the formula (1.98) are the compounds 1.98-1 to 1.98-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.98-1 to 1.98-68 of Table 1.98 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.99 Preferred compounds of the formula (1.99) are the compounds 1.99-1 to 1.99-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.99-1 to 1.99-68 of table 1.99 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.100 Preferred compounds of the formula (I.100) are the compounds I.100-1 to I.100-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 100-1 to I. 100-68 of Table I. 100 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.101 Preferred compounds of the formula (1.101) are the compounds 1. 101-1 to 1. 101- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 101-1 to I. 101-68 of Table I. 101 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1. (1,102)
- Preferred compounds of the formula (I. 102) are the compounds I. 102-1 to I. 102- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 102-1 to I. 102-68 of table I. 102 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.103 Preferred compounds of the formula (1.103) are the compounds I. 103-1 to I. 103- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 103-1 to I. 103-68 of Table I. 103 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.104 Preferred compounds of the formula (1. 104) are the compounds 1. 104-1 to 1. 104- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1. 104-1 to 1. 104-68 of Table 1. 104 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.105 Preferred compounds of the formula (1. 105) are the compounds 1. 105-1 to 1. 105- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1. 105-1 to 1. 105-68 of Table 1. 105 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Preferred compounds of the formula (I. 106) are the compounds I. 106-1 to I. 106- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 106-1 to I. 106-68 of Table I. 106 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Preferred compounds of the formula (I. 107) are the compounds I. 107-1 to I. 107- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 107-1 to I. 107-68 of Table I. 107 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1.108)
- Table 1.108 Preferred compounds of the formula (I.108) are the compounds I.108-1 to I.108-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 108-1 to I. 108-68 of Table I. 108 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.109 Preferred compounds of the formula (1. 109) are the compounds 1. 109-1 to 1. 109- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1. 109-1 to 1. 109-68 of table 1. 109 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.110 Preferred compounds of the formula (I.110) are the compounds I.110-1 to I.110-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I.110-1 to I.110-68 of Table I.110 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.111 Preferred compounds of the formula (1.111) are the compounds 1.111-1 to 1.111-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1. 111-1 to 1. 111-68 of Table 1. 111 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.112 Preferred compounds of the formula (I.112) are the compounds I.112-1 to I.112-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 112-1 to I. 112-68 of Table I. 112 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.113 Preferred compounds of the formula (I. 113) are the compounds I. 113-1 to I. 113- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 113-1 to I. 113-68 of Table I. 113 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1,114)
- Table 1.114 Preferred compounds of the formula (1. 114) are the compounds 1. 114-1 to 1. 114- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1. 114-1 to 1. 114-68 of Table 1. 114 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.115 Preferred compounds of the formula (I. 115) are the compounds I. 115-1 to I. 115- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 115-1 to I. 115-68 of Table I. 115 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.116 Preferred compounds of the formula (I. 116) are the compounds I. 116-1 to I. 116- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 116-1 to I. 116-68 of Table I. 116 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Preferred compounds of the formula (I. 117) are the compounds I. 117-1 to I. 117- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 117-1 to I. 117-68 of Table I. 117 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.118 Preferred compounds of the formula (I. 118) are the compounds I. 118-1 to I. 118- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 118-1 to I. 118-68 of Table I. 118 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1,119 Preferred compounds of the formula (1,119) are the compounds 1,119-1 to 1,119-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1. 119-1 to 1. 119-68 of Table 1. 119 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1,120)
- Table 1.120 Preferred compounds of the formula (I.120) are the compounds I.120-1 to I.120-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 120-1 to I. 120-68 of Table I. 120 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1,121 Preferred compounds of the formula (I. 121) are the compounds I. 121-1 to I. 121- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 121-1 to I. 121-68 of Table I. 121 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.122 Preferred compounds of the formula (I. 122) are the compounds I. 122-1 to I. 122- 68, in which Q has the meanings of Table 1 given in the respective row.
- the connections I. 122-1 to I. 122-68 of Table I. 122 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1.123)
- Table 1.123 Preferred compounds of the formula (1.123) are the compounds 1.123-1 to 1.123-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.123-1 to 1.123-68 of table 1.123 are thus by the meaning of the respective entries No. 1 to
- Table 1.124 Preferred compounds of the formula (1.124) are the compounds 1.124-1 to 1.124-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.124-1 to 1.124-68 of table 1.124 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- Table 1.125 Preferred compounds of the formula (1.125) are the compounds 1.125-1 to 1.125-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.125-1 to 1.125-68 of table 1.125 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1,126 Preferred compounds of the formula (1,126) are the compounds 1,126-1 to 1,126-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.126-1 to 1.126-68 of table 1.126 are thus by the meaning of the respective entries No. 1 to
- Table 1,127 Preferred compounds of the formula (1,127) are the compounds 1,127-1 to 1,127-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.127-1 to 1.127-68 of table 1.127 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
- Table 1.128 Preferred compounds of the formula (1.128) are the compounds 1.128-1 to 1.128-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.128-1 to 1.128-68 of table 1.128 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1,129)
- Table 1,129 Preferred compounds of the formula (1,129) are the compounds 1,129-1 to 1,129-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.129-1 to 1.129-68 of table 1.129 are thus by the meaning of the respective entries No. 1 to
- Table 1.130 Preferred compounds of the formula (1.130) are the compounds 1.130-1 to 1.130-68, in which Q has the meanings of Table 1 given in the respective row.
- the connections 1.130-1 to 1.130-68 of table 1.130 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
- the present invention furthermore relates to the use of one or more
- herbicide and / or plant growth regulator preferably in crops of useful and / or ornamental plants.
- the present invention furthermore relates to a process for controlling harmful plants and / or for regulating the growth of plants, characterized in that an effective amount of one or more compounds of the general formula (I) and / or their salts, as defined above, preferably in one those marked as preferred or particularly preferred
- the present invention also relates to a process for controlling unwanted plants, preferably in crops, characterized in that an effective amount of one or more compounds of the general formula (1) and / or their salts, as defined above, preferably in one of the as preferably or particularly preferably marked Design, in particular one or more compounds of the formulas (1.1) to (1.130) and / or their salts, in each case as defined above, or an agent according to the invention, as defined below, on undesired plants (for example harmful plants such as mono- or dicotyledon weeds or undesirable plants Crops), the seeds of the unwanted plants (ie plant seeds, e.g.
- the soil in or on which the unwanted plants grow e.g. the soil of cultivated or non-cultivated land
- the cultivated area ie area on which the unwanted plants will grow
- the present invention also relates to methods for combating
- Propagation organs such as tubers or shoots with buds
- the soil in or on which the plants grow e.g. the soil of cultivated or non-cultivated land
- the area under cultivation i.e. the area on which the plants will grow
- agents according to the invention e.g. in the pre-sowing (possibly also by incorporation into the soil), pre-emergence and / or post-emergence methods.
- Some representatives of the monocotyledonous and dicotyledonous weed flora can be mentioned by way of example, which can be controlled by the compounds according to the invention without being mentioned a restriction to certain types should be made.
- one or more compounds of the general formula (1) and / or their salts are preferably used for controlling harmful plants or for regulating growth in crops of useful plants or ornamental plants, the useful plants or ornamental plants in a preferred embodiment being transgenic plants.
- the compounds of the general formula (I) and / or their salts according to the invention are suitable for combating the following genera of monocotyledonous and dicotyledonous harmful plants:
- the compounds of the general formula (I) according to the invention are applied to the earth's surface before the germination of the harmful plants (grasses and / or weeds) (pre-emergence method), either the weed or weed seedlings are prevented from emerging completely or these grow up to the cotyledon stage However, they stop growing and eventually die completely after three to four weeks.
- the compounds of the general formula (1) according to the invention have an excellent herbicidal activity against mono- and dicotyledon weeds
- crop plants of economically important crops are e.g. dicotyledon cultures of the genera Arachis, Beta, Brassica, Cucumis, Cucurbita, Helianthus, Daucus, Glycine, Gossypium, lpomoea, Lactuca, Linum,
- the compounds of the general formula (1) according to the invention have excellent properties (depending on their structure and the application rate applied)
- Phytonutrients and to ease the crop, e.g. by triggering desiccation and stunted growth. Furthermore, they are also suitable for general control and inhibition of undesirable vegetative growth without killing the plants. Inhibiting vegetative growth plays a major role in many monocotyledonous and dicotyledonous crops, since, for example, this can reduce or completely prevent stock formation.
- the active compounds can also be used to control harmful plants in crops of plants modified by genetic engineering or by conventional mutagenesis.
- the transgenic plants are generally distinguished by special advantageous properties, for example resistance to certain pesticides, especially certain herbicides, resistance to plant diseases or pathogens of plant diseases such as certain insects or microorganisms such as fungi, bacteria or viruses.
- Other special properties concern e.g. the crop in terms of quantity, quality, shelf life, composition and special ingredients. So are transgenic plants with increased starch content or changed starch quality or those with others
- transgenic cultures is the use of the compounds according to the invention and / or their salts in economically important transgenic cultures of useful and ornamental plants, e.g. of cereals such as wheat, barley, rye, oats, millet, rice and corn or also crops of sugar beet, cotton, soybeans, rapeseed, potatoes, tomatoes, peas and other vegetables.
- cereals such as wheat, barley, rye, oats, millet, rice and corn or also crops of sugar beet, cotton, soybeans, rapeseed, potatoes, tomatoes, peas and other vegetables.
- the compounds according to the invention can preferably also be used as herbicides
- Crop crops are used which are resistant to the phytotoxic effects of the herbicides or have been made genetically resistant.
- the active compounds can also be used to control harmful plants in crops of known or still to be developed genetically modified plants.
- the transgenic plants are generally distinguished by special advantageous properties, for example by resistance to them certain pesticides, especially certain herbicides, resistance to plant diseases or pathogens such as certain insects or microorganisms such as fungi, bacteria or viruses.
- Other special properties relate to the crop, for example, in terms of quantity, quality, shelf life, composition and special ingredients. So are transgenic plants with increased starch content or changed starch quality or those with others
- Other special properties can include tolerance or resistance to abiotic stressors e.g. There is heat, cold, dryness, salt and ultraviolet radiation.
- cereals such as wheat, barley, rye, oats, triticale, millet, rice, manioc and corn or also crops of sugar beet, cotton, soybean, rapeseed, potato, tomato, pea and other vegetables.
- the compounds of the general formula (I) can preferably be used as herbicides in
- Crop crops are used which are resistant to the phytotoxic effects of the herbicides or have been made genetically resistant.
- nucleic acid molecules can be introduced into plasmids which allow mutagenesis or a sequence change by recombining DNA sequences. With the help of standard procedures e.g. Base exchanges made, partial sequences removed or natural or synthetic sequences added. For connecting the DNA fragments
- adapters or linkers can be attached to the fragments.
- DNA molecules can be used that comprise the entire coding sequence of a gene product, including any flanking sequences that may be present, as well as DNA molecules that only comprise parts of the coding sequence, these parts having to be long enough to be in the cells to cause an antisense effect. It is also possible to use DNA sequences which have a high degree of homology to the coding sequences of a gene product, but which are not completely identical.
- the synthesized protein When nucleic acid molecules are expressed in plants, the synthesized protein can be located in any compartment of the plant cell. However, in order to achieve localization in a certain compartment, e.g. the coding region is linked to DNA sequences which ensure localization in a specific compartment. Such sequences are known to the person skilled in the art (see, for example, Braun et al., EMBO J. 11 (1992), 3219-3227). The expression of the nucleic acid molecules can also take place in the organelles of the plant cells.
- the transgenic plant cells can be regenerated into whole plants using known techniques.
- the transgenic plants can be any plants
- the compounds (I) according to the invention can preferably be used in transgenic cultures which are active against growth substances, e.g. Dicamba or against herbicides, the essential
- Plant enzymes e.g. Inhibit acetolactate synthases (ALS), EPSP synthases, glutamine synthases (GS) or hydoxyphenyl pyruvate dioxygenases (HPPD), or are resistant to herbicides from the group of sulfonylureas, glyphosate, glufosinate or benzoylisoxazole and analogous active ingredients.
- ALS Inhibit acetolactate synthases
- EPSP synthases glutamine synthases
- HPPD hydoxyphenyl pyruvate dioxygenases
- the active compounds according to the invention are used in transgenic crops, in addition to the effects on harmful plants which can be observed in other crops, effects often occur which are specific to the application in the respective transgenic culture, for example a changed or specially expanded weed spectrum which can be controlled changed Application rates that can be used for the application, preferably good combinability with the herbicides to which the transgenic culture is resistant, and influencing the growth and yield of the transgenic crop plants.
- the invention therefore also relates to the use of the compounds of the general formula (I) according to the invention and / or their salts as herbicides for controlling harmful plants in crops of useful or ornamental plants, optionally in transgenic crop plants.
- cereals preferably maize, wheat, barley, rye, oats, millet or rice, in the pre- or post-emergence.
- Pre-or post-soya use is also preferred.
- Plant growth regulation also includes the case in which the active ingredient of the general formula (1) or its salt is formed from a precursor substance (“prodrug”) only after application to the plant, in the plant or in the soil.
- the invention also relates to the use of one or more compounds of the formula (1) or their salts or an agent according to the invention (as defined below) (in one
- Process for controlling harmful plants or for regulating the growth of plants, characterized in that an effective amount of one or more compounds of the general formula (I) or their salts are applied to the plants (harmful plants, if appropriate together with the useful plants) plant seeds, the soil , in or on which the plants grow, or the cultivated area is applied.
- the invention also relates to a herbicidal and / or plant growth-regulating agent, characterized in that the agent
- (a) contains one or more compounds of the general formula (I) and / or their salts as defined above, preferably in one of those identified as preferred or particularly preferred
- insecticides acaricides, nematicides, further herbicides (ie those which do not correspond to the general formula (I) defined above), fungicides, safeners, fertilizers and / or further growth regulators, (ii) one or more formulation auxiliaries customary in crop protection.
- component (i) of an agent according to the invention are preferably selected from the group of substances described in "The Pesticide Manual”, 16th edition, The British Crop Protection Council and the Royal Soc. of Chemistry, 2012.
- a herbicidal or plant growth-regulating agent according to the invention preferably comprises one, two, three or more formulation auxiliaries (ii) customary in crop protection selected from the group consisting of surfactants, emulsifiers, dispersants, film formers, thickeners, inorganic salts, dusts, at 25 ° C. and 1013 mbar solid carriers, preferably adsorbable, granulated inert materials, wetting agents, antioxidants, stabilizers, buffer substances, antifoams, water, organic solvents, preferably at 25 ° C. and 1013 mbar, organic solvents miscible with water in any ratio.
- formulation auxiliaries customary in crop protection selected from the group consisting of surfactants, emulsifiers, dispersants, film formers, thickeners, inorganic salts, dusts, at 25 ° C. and 1013 mbar solid carriers, preferably adsorbable, granulated inert materials, wetting agents, antioxidants, stabilizers
- the compounds of the general formula (I) according to the invention can be used in the form of wettable powders, emulsifiable concentrates, sprayable solutions, dusts or granules in the customary formulations.
- the invention therefore also relates to herbicidal and plant growth-regulating compositions which comprise compounds of the general formula (I) and / or their salts.
- the compounds of general formula (I) and / or their salts can be formulated in various ways, depending on which biological and / or chemical-physical parameters are specified. Possible formulation options are, for example: wettable powder (WP), water-soluble powder (SP), water-soluble concentrates, emulsifiable concentrates (EC), emulsions (EW), such as oil-in-water and water-in-oil emulsions, sprayable solutions .
- WP wettable powder
- SP water-soluble powder
- EC emulsifiable concentrates
- EW emulsions
- sprayable solutions such as oil-in-water and water-in-oil emulsions, sprayable solutions .
- SC Suspension concentrates
- CS Capsule suspensions
- DP dusts
- pickling agents granules for the litter
- granules in the form of micro, spray, elevator and adsorption granules, water-dispersible granules (WG), water-soluble granules (SG), ULV formulations,
- Microcapsules and waxes are Microcapsules and waxes.
- Spray powders are preparations which are uniformly dispersible in water and which, in addition to the active substance, contain not only a diluent or an inert substance, but also ionic and / or nonionic surfactants (wetting agents, dispersing agents), e.g.
- the herbicidal active ingredients are, for example, finely ground in customary equipment, such as hammer mills, fan mills and air jet mills, and mixed at the same time or subsequently with the formulation auxiliaries.
- Emulsifiable concentrates are made by dissolving the active ingredient in an organic solvent e.g. Butanol, cyclohexanone, dimethylformamide, xylene or higher-boiling aromatics or hydrocarbons or mixtures of organic solvents with the addition of one or more surfactants of ionic and / or nonionic type (emulsifiers).
- organic solvent e.g. Butanol, cyclohexanone, dimethylformamide, xylene or higher-boiling aromatics or hydrocarbons or mixtures of organic solvents
- surfactants of ionic and / or nonionic type emulsifiers
- alkylarylsulfonic acid calcium salts such as
- Ca-dodecylbenzenesulfonate or nonionic emulsifiers such as fatty acid polyglycol esters
- Alkylaryl polyglycol ether fatty alcohol polyglycol ether, propylene oxide-ethylene oxide condensation products, alkyl polyether, sorbitan esters such as e.g. Sorbitan fatty acid esters or
- Polyoxethylene sorbitan esters such as e.g. Polyoxyethylene.
- Dusts are obtained by grinding the active ingredient with finely divided solid substances, e.g.
- Talc natural clays such as kaolin, bentonite and pyrophyllite, or diatomaceous earth.
- Suspension concentrates can be water or oil based. You can, for example, by wet grinding using commercially available bead mills and optionally adding surfactants such as those e.g. already listed above for the other types of formulation.
- Emulsions e.g. Oil-in-water emulsions (EW) can be used, for example, using stirrers, colloid mills and / or static mixers using aqueous organic ones
- Granules can either be produced by spraying the active ingredient onto adsorbable, granulated inert material or by applying active ingredient concentrates using adhesives, for example polyvinyl alcohol, polyacrylic acid sodium or mineral oils, to the surface of carriers such as sand, kaolinite or granulated inert material. Also suitable ones
- Active ingredients are granulated in the manner customary for the production of fertilizer granules - if desired in a mixture with fertilizers.
- Water-dispersible granules are generally produced using the customary processes, such as spray drying, fluidized bed granulation, plate granulation, mixing with high-speed mixers and extrusion without solid inert material.
- the agrochemical preparations preferably herbicidal or plant growth-regulating agents of the present invention preferably contain a total amount of 0.1 to 99% by weight, preferably 0.5 to 95% by weight, more preferably 1 to 90% by weight, particularly preferably 2 to 80% by weight of active compounds of the general formula (I) and their salts.
- the active substance concentration in wettable powders is e.g. about 10 to 90 wt .-%, the rest of 100 wt .-% consists of conventional formulation components. In the case of emulsifiable concentrates, the active substance concentration can be about 1 to 90, preferably 5 to 80,% by weight. Powdery
- Formulations contain 1 to 30% by weight of active ingredient, preferably mostly 5 to 20% by weight of active ingredient, sprayable solutions contain about 0.05 to 80, preferably 2 to 50% by weight of active ingredient.
- the active ingredient content depends in part on whether the active compound is in liquid or solid form and which granulating aids, fillers, etc. are used.
- the active ingredient content of the water-dispersible granules is, for example, between 1 and 95% by weight, preferably between 10 and 80% by weight.
- the active ingredient formulations mentioned may contain the customary adhesives, wetting agents, dispersants, emulsifiers, penetrants, preservatives, antifreezes and solvents, fillers, carriers and dyes, defoamers, evaporation inhibitors and the pH and Agents influencing viscosity.
- formulation auxiliaries are described, inter alia, in “Chemistry and Technology of Agrochemical Formulations", ed. DA Knowles, Kluwer Academic Publishers (1998).
- the compounds of general formula (I) or their salts can be used as such or in the form of their preparations (formulations) with other pesticidally active substances, e.g. Insecticides, acaricides, nematicides, herbicides, fungicides, safeners, fertilizers and / or
- Growth regulators can be used in combination, e.g. as a finished formulation or as
- Tank mixes The combination formulations can be prepared on the basis of the formulations mentioned above, taking into account the physical properties and stabilities of the active compounds to be combined.
- Treatment conditions according to the invention can also lead to superadditive ("synergistic") effects in growth conditions (soils, climate, growing season, nutrition).
- superadditive additive
- the following effects are possible that go beyond the effects that are actually to be expected: reduced application rates and / or expanded spectrum of activity and / or increased effectiveness of the active compounds and compositions which can be used according to the invention, better plant growth, increased tolerance to high or low Temperatures, increased tolerance to
- Dryness or water or soil salt content increased flowering performance, easier harvesting,
- Mixture formulations or in the tank mix are, for example, known active ingredients which are based on an inhibition of, for example, acetolactate synthase, acetyl-CoA carboxylase, cellulose synthase, enolpyruvylshikimate-3-phosphate synthase, glutamine synthetase, p-hydroxyphenylpyruvate dioxygenase, and , Photosystem I, Photosystem II, protoporphyrinogen oxidase, can be used, as described, for example, in Weed Research 26 (1986) 441-445 or "The Pesticide Manual", 16th edition, The British Crop Protection Council and the Royal Soc. of Chemistry, 2012 and the literature cited therein.
- Compounds (1) according to the invention of particular interest which contain the compounds (1) or their combinations with other herbicides or pesticides and safeners.
- the safeners which are used in an antidotically effective content, reduce the phytotoxic side effects of the herbicides / pesticides used, e.g. in economically important crops such as cereals (wheat, barley, rye, corn, rice, millet), sugar beet, sugar cane, rapeseed, cotton and soybeans, preferably cereals.
- the weight ratio of herbicide (mixture) to safener generally depends on the
- herbicide and the effectiveness of the respective safener can vary within wide limits, for example in the range from 200: 1 to 1: 200, preferably 100: 1 to 1: 100, in particular 20: 1 to 1:20.
- the safeners can be formulated analogously to the compounds of the general formula (1) or their mixtures with further herbicides / pesticides and as
- the herbicide or herbicide safener formulations present in commercially available form are optionally diluted in the customary manner, for example for wettable powders, emulsifiable concentrates, dispersions and water-dispersible granules using water.
- Preparations in the form of dust, ground granules or granules as well as sprayable solutions are usually no longer diluted with other inert substances before use.
- the application rate can vary within wide limits.
- the total amount of compounds of the general formula (1) and their salts is preferably in the range from 0.001 to 10.0 kg / ha, preferably in the range from 0.005 to 5 kg / ha, more preferably in Range from 0.01 to 1.5 kg / ha, particularly preferably in the range from 0.05 to 1 kg / ha. This applies to both pre-emergence and post-emergence applications.
- Plant growth regulator for example as a straw shortener in crop plants as mentioned above, preferably in cereal plants such as wheat, barley, rye, triticale, millet, rice or corn, the total application rate is preferably in the range from 0.001 to 2 kg / ha, preferably in the range from 0.005 to 1 kg / ha, in particular in the range from 10 to 500 g / ha, very particularly preferably in the range from 20 to 250 g / ha. This applies both to the application in
- the application as a straw shortener can take place in different stages of the growth of the plants. For example, use after planting at the beginning of the
- the treatment of the seed which includes the different seed dressing and coating techniques, can also be used.
- the application rate depends on the individual techniques and can be determined in preliminary tests.
- compositions according to the invention for example mixture formulations or in a tank mix
- active substances which are based on inhibition of, for example, acetolactate synthase, acetyl-CoA carboxylase, cellulose synthase, enolpyruvylshikimate -3-phosphate synthase, glutamine synthetase, p-hydroxyphenylpyruvate dioxygenase, phytoendesaturase, Photosystem I, Photosystem II or
- Protoporphyrinogen oxidase are used, such as those e.g. from Weed Research 26 (1986) 441-445 or "The Pesticide Manual", 16th edition, The British Crop Protection Council and the Royal Soc. of Chemistry, 2012 and the literature cited therein.
- Known herbicides or plant growth regulators are mentioned below by way of example, which can be combined with the compounds according to the invention, these active compounds either with their "common name" in the English-language variant according to the International Organization for Standardization (ISO) or with the chemical name or with the code number are designated.
- herbicidal mixture partners examples include:
- flucarbazone flucarbazone-sodium, flucetosulfuron, fluchloralin, flufenacet, flufenpyr, flufenpyr-ethyl, flumetsulam, flumiclorac, flumiclorac-pentyl, flumioxazin, fluometuron, flurenol, flurenol-butyl, -dimethylammonoglypane, and -methyl-fluorine-cyanogen flupyrsulfuron, flupyrsulfuron-methyl-sodium, fluridone, flurochloridone, fluroxypyr, fluroxypyr-meptyl, flurtamone, fluthiacet, fluthiacet-methyl, fomesafen, fomesafen-sodium, foramsulfuron, fosamine, glufosinate, glufosinate ammonium, pufosinate ammonium P-ammonium, glufosinate-P-sodium
- metdicazthiazuron metam, metamifop, metamitron, metazachlor, metazosulfuron,
- plant growth regulators as possible mixing partners are:
- Sl d compounds of the triazole carboxylic acid type (Sl d ), preferably compounds such as
- Fenchlorazole ethyl ester
- ethyl ester i.e. 1- (2,4-dichlorophenyl) -5-trichloromethyl- (lH) -l, 2,4-triazole-3-carboxylic acid ethyl ester (S1-7), and related compounds as described in EP-A-174562 and EP- A-346620;
- Sl e compounds of the type of 5-benzyl- or 5-phenyl-2-isoxazoline-3-carboxylic acid, or of 5,5-diphenyl-2-isoxazoline-3-carboxylic acid (Sl e ), preferably compounds such as
- R-29148 (3-dichloroacetyl-2,2,5-trimethyl-1,3-oxazolidine) from Stauffer (S3-2)
- R-28725" (3-dichloroacetyl-2,2, -dimethyl- 1,3-oxazolidine) from Stauffer (S3-3)
- PPG-1292 N-allyl-N - [(1,3-dioxolan-2-yl) methyl] dichloroacetamide
- TI-35 (l-dichloroacetyl-azepan) from TRI-Chemical RT (S3-8),
- RA 2 halogen (Ci-C 4 ) alkyl, (Ci-C 4 ) alkoxy, CF 3; m A 1 or 2;
- VA is 0, 1, 2 or 3;
- RB 1 , RB 2 independently of one another hydrogen, (Ci-C 6 ) alkyl, (C3-C6) cycloalkyl, (C3-C 6 ) alkenyl, (C3-C 6 ) alkynyl,
- RB 3 represents halogen, (Ci-C4) alkyl, (Ci-C4) haloalkyl or (Ci-C4) alkoxy and ms 1 or 2, for example those in which
- RB 1 cyclopropyl
- RB 2 hydrogen
- (RB 3 ) 2-OMe
- RB 1 cyclopropyl
- R B 1 ethyl
- R B 1 isopropyl
- R B 2 hydrogen
- (R B 3 ) 5-Cl-2-OMe is (S4-4) and
- R B 1 isopropyl
- R B 2 hydrogen
- (R B 3 ) 2-OMe is (S4-5);
- Rc 1 , Rc 2 independently of one another hydrogen, (Ci-Cg) alkyl, (C3-Cg) cycloalkyl, (C3-
- Rc 3 is halogen, (Ci-C i) alkyl, (Ci-C4) alkoxy, CF 3 and m c 1 or 2; for example 1- [4- (N-2-methoxybenzoylsulfamoyl) phenyl] -3-methylurea,
- R D 4 halogen, (Ci-C 4 ) alkyl, (Ci-C 4 ) alkoxy, CF 3; m D 1 or 2; RD 5 is hydrogen, (Ci-C 6 ) alkyl, (C 3 -C 6 ) cycloalkyl, (C 2 -C 6 ) alkenyl, (C 2 -C 6 ) alkynyl, (C 5 - C 6 ) cycloalkenyl.
- Carboxylic acid derivatives (S5) e.g.
- Dihydroxybenzoic acid 4-hydroxysalicylic acid, 4-fluorosalicyclic acid, 2-hydroxycinnamic acid, 2,4-dichlorocinnamic acid, as described in WO-A-2004/084631, WO-A-2005/015994, WO-A-2005/016001.
- RD 1 is halogen, (Ci-C i) alkyl, (Ci-C i) haloalkyl, (Ci-C i) alkoxy, (Ci-C4) haloalkoxy,
- R D 2 is hydrogen or (Ci-C i) alkyl
- R D 3 is hydrogen, (Ci-Csjalkyl, (C 2 -C4) alkenyl, (C 2 -C4) alkynyl, or aryl, where each of the abovementioned C-containing radicals is unsubstituted or by one or more, preferably up to three of the same or various radicals from the group consisting of halogen and alkoxy, or their salts, n D is an integer from 0 to 2.
- S9 active substances from the class of 3- (5-tetrazolylcarbonyl) -2-quinolones (S9), e.g.
- Y E , Z E independently of one another O or S, he is an integer from 0 to 4,
- RE 2 (Ci-Ci 6 ) alkyl, (C2-C6) alkenyl, (C3-C6) cycloalkyl, aryl; Benzyl, halobenzyl, RE 3 is hydrogen or (Ci-C 6 ) alkyl.
- Sl 1 Active substances of the oxyimino compound type (Sl 1), which are known as seed dressings, such as. B.
- Oxabetrinil ((Z) -l, 3-dioxolan-2-ylmethoxyimino (phenyl) acetonitrile) (Sl l-l), which is known as a seed dressing safener for millet against damage to metolachlor,
- Seed pickle safener is known for millet against damage from metolachlor.
- Naphthalic anhydride (1,8-naphthalenedicarboxylic anhydride) (S13-1), which is known as a seed dressing safener for maize against damage from thiocarbamate herbicides,
- Cyanamide which is known as a safener for corn against damage to imidazolinones
- MG 191 (CAS Reg. No. 96420-72-3) (2-dichloromethyl-2-methyl-1,3-dioxolane) (S13-5) from Nitrokemia, which is known as a safener for corn,
- Mephenate (4-chlorophenyl methyl carbamate) (S13-9).
- Active ingredients which, in addition to a herbicidal action against harmful plants, also have safener action on crop plants such as rice, such as, for. B.
- COD l-bromo-4- (chloromethylsulfonyl) benzene
- RH 1 represents a (Ci-C 6 ) haloalkyl radical and R H 2 represents hydrogen or halogen and
- R H 3 , R H 4 independently of one another are hydrogen, (Ci-Ci 6 ) alkyl, (C2-Ci6) alkenyl or
- (C 2 -Ci 6 ) alkynyl each of the latter 3 residues being unsubstituted or by one or more residues from the group halogen, hydroxy, cyano, (Ci-C i) alkoxy, (Ci-C i) haloalkoxy, (Ci C ij alkylthio, (Ci-C ij alkylamino, di [(Ci-C4) alkyl] amino, [(Ci-C i) alkoxy] carbonyl, [(Ci-C t j haloalkoxyj-carbonyl, (C3-C6) cycloalkyl, which is unsubstituted or substituted, phenyl which is unsubstituted or substituted and heterocyclyl which is unsubstituted or substituted, or (C3-C6) cycloalkyl, (C4-C6) cycloalkenyl, (C3-C6) cycloalkyl
- R H 3 is (Ci-C 4 ) alkoxy, (C2-C 4 ) alkenyloxy, (C2-C6) alkynyloxy or (C2-C 4 ) haloalkoxy and R H 4 is hydrogen or (Ci-C 4 ) alkyl or
- R H 3 and R H 4 together with the directly bound N atom form a four- to eight-membered group
- heterocyclic ring which, in addition to the N atom, can also contain further hetero ring atoms, preferably up to two further hetero ring atoms from the group N, O and S, and which is unsubstituted or by one or more radicals from the group halogen, cyano, nitro, (Ci C 4 ) alkyl, (Ci-C 4 ) haloalkyl, (Ci-C 4 ) alkoxy, (Ci-C 4 ) haloalkoxy and (Ci-C 4 ) alkylthio is substituted.
- Preferred safeners in combination with the compounds according to the invention of the formula (I) and / or their salts, in particular with the compounds of the formulas (1.1) to (1.130) and / or their salts are: cloquintocet-mexyl, cyprosulfamide, fenchlorazole ethyl ester, isoxadifene -ethyl, mefenpyr-diethyl, fenclorim, cumyluron, S4-1 and S4-5, and particularly preferred safeners are: cloquintocet-mexyl, cyprosulfamide, isoxadifen-ethyl and mefenpyr-diethyl.
- Biological examples are:
- the amount of water applied is equivalent to 2200 liters per hectare. After 9 to 12 days
- Seeds of monocotyledonous or dicotyledonous weeds and crops were placed in plastic or organic plant pots and covered with soil.
- the compounds according to the invention formulated in the form of wettable powders (WP) or as emulsion concentrates (EC) were then applied to the surface of the covering earth as an aqueous suspension or emulsion with the addition of 0.5% additive with a water application rate of the equivalent of 600 l / ha.
- WP wettable powders
- EC emulsion concentrates
- the following tablets B1 to B5 show the effects of selected compounds of the general formula (I) according to Tables 1.1 to 1.130 on various harmful plants and an application rate corresponding to 320 g / ha or less, which were obtained in accordance with the aforementioned test instructions.
- Compounds 1.2-8, 1.2-1 and 1.4-8 and other compounds of the general formula (I) according to the invention have a good herbicidal activity against harmful plants, e.g. B. against harmful plants such as Abutilon theophrasti, Alopecurus myosuroides, Setaria viridis, Avena fatua, Amaranthus retroflexus, Polygonum convolvulus and Viola tricolor at a rate of 320 g of active ingredient per hectare.
- harmful plants such as Abutilon theophrasti, Alopecurus myosuroides, Setaria viridis, Avena fatua, Amaranthus retroflexus, Polygonum convolvulus and Viola tricolor at a rate of 320 g of active ingredient per hectare.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Substituierte Thiazolylpyrrolone sowie deren Salze und ihre Verwendung als herbizide Wirkstoffe Beschreibung Substituted thiazolylpyrrolones and their salts and their use as herbicidal active ingredients Description
Die Erfindung betrifft das technische Gebiet der Pflanzenschutzmittel, insbesondere das der Herbizide zur selektiven Bekämpfung von Unkräutern und Ungräsem in Nutzpflanzenkulturen. The invention relates to the technical field of crop protection agents, in particular that of herbicides for the selective control of weeds and weeds in crops of useful plants.
Speziell betrifft diese Erfindung substituierte Thiazolylpyrrolone sowie deren Salze, Verfahren zu ihrer Herstellung und ihre Verwendung als Herbizide. In particular, this invention relates to substituted thiazolylpyrrolones and their salts, processes for their preparation and their use as herbicides.
Bisher bekannte Pflanzenschutzmittel zur selektiven Bekämpfung von Schadpflanzen in Known crop protection products for the selective control of harmful plants in
Nutzpflanzenkulturen oder Wirkstoffe zur Bekämpfung von unerwünschtem Pflanzenwuchs weisen bei ihrer Anwendung teilweise Nachteile auf, sei es, dass sie (a) keine oder aber eine unzureichende herbizide Wirkung gegen bestimmte Schadpflanzen, (b) ein zu geringes Spektrum der Schadpflanzen, das mit einem Wirkstoff bekämpft werden kann, (c) zu geringe Selektivität in Nutzpflanzenkulturen und/oder (d) ein toxikologisch ungünstiges Profil besitzen. Weiterhin führen manche Wirkstoffe, die als Pflanzenwachstumsregulatoren bei einigen Nutzpflanzen eingesetzt werden können, bei anderen Nutzpflanzen zu unerwünscht verminderten Emteerträgen oder sind mit der Kulturpflanze nicht oder nur in einem engen Aufwandmengenbereich verträglich. Einige der bekannten Wirkstoffe lassen sich wegen schwer zugänglicher Vorprodukte und Reagenzien im industriellen Maßstab nicht wirtschaftlich hersteilen oder besitzen nur unzureichende chemische Stabilitäten. Bei anderen Wirkstoffen hängt die Wirkung zu stark von Umweltbedingungen, wie Wetter- und Bodenverhältnissen ab. Crop crops or active ingredients to combat undesirable plant growth sometimes have disadvantages when used, either because they (a) have no or an inadequate herbicidal activity against certain harmful plants, (b) the spectrum of the harmful plants is too small to combat with an active ingredient can be (c) insufficient selectivity in crops and / or (d) have a toxicologically unfavorable profile. Furthermore, some active ingredients which can be used as plant growth regulators in some crop plants lead to undesirably reduced crop yields in other crop plants or are incompatible or only compatible with the crop plant in a narrow range of application rates. Some of the known active ingredients cannot be economically manufactured on an industrial scale because of precursors and reagents that are difficult to access, or they have insufficient chemical stabilities. For other active substances, the effect depends too much on environmental conditions such as weather and soil conditions.
Die herbizide Wirkung dieser bekannten Verbindungen, insbesondere bei niedrigen Aufwandmengen, bzw. deren Verträglichkeit gegenüber Kulturpflanzen bleiben verbesserungswürdig. The herbicidal activity of these known compounds, especially at low application rates, or their compatibility with crop plants remains in need of improvement.
Verschiedene Schriften beschreiben substituierte Pyrrolone und Hydantoine mit herbiziden Various publications describe substituted pyrrolones and hydantoins with herbicides
Eigenschaften. Aus WO2016/071359 und W02016/071360 sind Pyrrolone bekannt, die am Stickstoff heterocyclische Substituenten tragen, beispielsweise auch gegebenenfalls weiter substituierte Characteristics. From WO2016 / 071359 and W02016 / 071360 pyrrolones are known which carry heterocyclic substituents on the nitrogen, for example also optionally further substituted
Isoxazoline. Weiterhin sind substituierte Pyrrolone und ihre herbiziden oder pestiziden Eigenschaften in CH633678, DE 2735841, EP0297378, EP0334133, EP0339390 und EP0286816 beschrieben. In WO2016/071361, WO2016/071362, WO2016/071363 und WO2016/071364 werden weiterhin substituierte Hydantoine beschrieben, die am Stickstoff ebenfalls heterocyclische Substituenten tragen, beispielsweise gegebenenfalls weiter substituierte Isoxazoline. Ausgewählte speziell substituierte 1,3,4- Thiadiazolyl- und l,2,4-Thiadiazolyl-2,5-Dioxoimidazoline und ihre herbizide Wirkung werden in DE2247266 beschrieben. Substituierte Pyrazolylpyrrolone und ihre Verwendung als herbizide Isoxazolines. Substituted pyrrolones and their herbicidal or pesticidal properties are also described in CH633678, DE 2735841, EP0297378, EP0334133, EP0339390 and EP0286816. WO2016 / 071361, WO2016 / 071362, WO2016 / 071363 and WO2016 / 071364 also describe substituted hydantoins which also carry heterocyclic substituents on the nitrogen, for example optionally further substituted isoxazolines. Selected specially substituted 1,3,4-thiadiazolyl and l, 2,4-thiadiazolyl-2,5-dioxoimidazolines and their herbicidal activity are described in DE2247266 described. Substituted pyrazolylpyrrolones and their use as herbicides
Wirkstoffe werden beispielsweise in WO2015/018434 beschrieben. Active ingredients are described, for example, in WO2015 / 018434.
Es ist weiterhin bekannt, daß bestimmte substituierte Carbamoyloxypyrrolone und Phenyloxypyrrolone als Wuchsregulatoren oder Keimungsstimulatoren eingesetzt werden können (vgl. WO2014/131843 und WO2015/128321). Das Strukturmotiv der Hydroxypyrrolone findet sich auch in Substanzen, die aus Lilium candidum isoliert wurden, z. B. l,5-Dihydro-5-hydroxy-3,4‘-dimethyl-[l,2‘-bi-2H-pyrrolo]- 2,5‘(l’H)-dion (vgl. Ceska a Slovenska Farmacie, 2007, 56, 27; Neoplasma, 2000, 47, 313). Die Verwendung von an der C-C-Doppelbindung substituierten Thiazolylpyrrolone oder deren Salze als herbizide Wirkstoffe ist dagegen noch nicht beschrieben. Überraschenderweise wurde nun gefunden, daß an der C-C-Doppelbindung substituierte Thiazolylpyrrolone oder deren Salze als Herbizide besonders gut geeignet sind. Gegenstand der vorliegenden Erfindung sind damit substituierte Thiazolylpyrrolone der allgemeinen Formel (I) oder deren Salze It is also known that certain substituted carbamoyloxypyrrolones and phenyloxypyrrolones can be used as growth regulators or germination stimulators (cf. WO2014 / 131843 and WO2015 / 128321). The structural motif of hydroxypyrrolone is also found in substances isolated from Lilium candidum, e.g. B. 1,5-Dihydro-5-hydroxy-3,4'-dimethyl- [1,2'-bi-2H-pyrrolo] - 2,5 '(l'H) -dione (cf. Ceska a Slovenska Farmacie , 2007, 56, 27; Neoplasma, 2000, 47, 313). However, the use of thiazolylpyrrolones substituted on the C-C double bond or their salts as herbicidal active ingredients has not yet been described. Surprisingly, it has now been found that thiazolylpyrrolones or their salts substituted on the C-C double bond are particularly suitable as herbicides. The present invention thus relates to substituted thiazolylpyrrolones of the general formula (I) or salts thereof
worin Q für die Gruppe where Q for the group
steht X und Y unabhängig voneinander für C-H oder die Gruppierung C-R1 stehen, wobei X and Y independently of one another represent CH or the grouping CR 1 , where
X für C-H steht, wenn Y für die Gruppierung C-R1 steht und X stands for CH if Y stands for the grouping CR 1 and
X für die Gruppierung C-R1 steht, wenn Y für C-H steht, X stands for the grouping CR 1 , if Y stands for CH,
R1 für Halogen, Cyano, (Ci-Cg)-Alkyl, (Ci-C8)-Haloalkyl, (Ci-Cg)-Hydroxyalkyl, ( C i - C s ) -R 1 for halogen, cyano, (Ci-Cg) -alkyl, (Ci-C 8 ) -haloalkyl, (Ci-Cg) -hydroxyalkyl, (C i - C s) -
Alkoxyalkyl, (Ci-Cg)-Alkoxy, (Ci-Cg)-Haloalkoxy, (Ci-Cg)-Alkylthio, (Ci-C8)-Haloalkylthio, Aryl, Heteroaryl, Aryloxy, Heteroaryloxy, Heterocyclyl, (C3-Cio)-Cycloalkyl, (C3-C10)- Cycloalkyl-(Ci-C8)-alkyl, (C3-C8)-Halocycloalkyl, (C3-C8)-Halocycloalkyl-(Ci-C8)-alkyl, (Ci-C8)-Alkylcarbonyl, (C2-Cg)-Alkenyl, (C2-Cg)-Alkinyl, steht, Alkoxyalkyl, (Ci-Cg) -alkoxy, (Ci-Cg) -haloalkoxy, (Ci-Cg) -alkylthio, (Ci-C 8 ) -haloalkylthio, Aryl, heteroaryl, aryloxy, heteroaryloxy, heterocyclyl, (C 3 -Cio) cycloalkyl, (C 3 -C 10 ) cycloalkyl- (Ci-C8) alkyl, (C 3 -C 8 ) -halocycloalkyl, (C 3 -C 8 ) halocycloalkyl (Ci-C 8 ) alkyl, (Ci-C 8 ) alkylcarbonyl, (C 2 -Cg) alkenyl, (C 2 -Cg) alkynyl,
A1 und A2 unabhängig voneinander für C-H oder die Gruppierung C-R2 stehen, wobei A 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
A1 für C-H steht, wenn A2 für die Gruppierung C-R2 steht und A 1 stands for CH if A 2 stands for the grouping CR 2 and
A1 für die Gruppierung C-R2 steht, wenn A2 für C-H steht, A 1 stands for the grouping CR 2 , if A 2 stands for CH,
R2 für Hydrothio, Hydroxy, Halogen, (Ci-Cg)-Alkyl, (Ci-Cg)-Haloalkyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Cycloalkyl-(Ci-Cg)-alkyl, Aryl, Heteroaryl, Heterocyclyl, Aryl-(Ci-Cg)-alkyl, R 2 for hydrothio, hydroxy, halogen, (Ci-Cg) alkyl, (Ci-Cg) haloalkyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) cycloalkyl (Ci-Cg) alkyl , Aryl, heteroaryl, heterocyclyl, aryl- (Ci-Cg) -alkyl,
Heteroaryl-(Ci-Cg)-alkyl, Heterocyclyl-(Ci-Cg)-alkyl, (C2-Cg)-Alkenyl, (C2-Cg)-Alkinyl, (C2-Cg)-Haloalkenyl, (C2-Cg)-Haloalkinyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, Aryl-(C2-Cg)-alkenyl, Heteroaryl-(C2-Cg)-alkenyl, Heterocyclyl- (C2-Cg)-alkenyl, Aryl-(C2-Cg)-alkinyl, Heteroaryl-(C2-Cg)-alkinyl, Heterocyclyl-(C2-Cg)-alkinyl, (C3-Cio)-Cycloalkyl-(C2-Cg)-alkinyl, Arylcarbonyl-(Ci-Cg)-alkyl, (Ci-C8)-Alkylcarbonyl- (Ci-Cg)-alkyl, Heteroarylcarbonyl-(Ci-Cg)-alkyl, (C3-Cio)-Cycloalkylcarbonyl-(Ci-Cg)-alkyl, Aryl-(Ci-Cg)-alkoxycarbonyl-(Ci-Cg)-alkyl, (Ci-Cg)-Alkoxycarbonyl-(Ci-Cg)-alkyl, ( C 1 - C s ) - Alkoxy-(Ci-Cg)-alkyl, Arylcarbonyloxy-(Ci-Cg)-alkyl, Heteroarylcarbonyloxy-(Ci-Cg)-alkyl, Heterocyclylcarbonyloxy-(Ci-Cg)-alkyl, (Ci-Cg)-Alkylcarbonyloxy-(Ci-Cg)-alkyl, (C3-C8)- Cycloalkylcarbonyloxy-(Ci-Cg)-alkyl, (Ci-Cg)-Haloalkoxy-(Ci-Cg)-alkyl, Aryl-(Ci-C8)-alkoxy- (Ci-Cg)-alkyl, Heteroaiyl-(Ci-C8)-alkoxy-(Ci-C8)-alkyl, CHO, C(0)R5, C(0)OR5, C(0)NR6R7, OR8, SR9, SOR9, SO2R9, NR5R6, R5R6N-(Ci-C8)-alkyl, Cyano-(Ci-Cg)-alkyl, Hydroxycarbonyl- (Ci-Cg)-alkyl, (Ci-C8)-Haloalkoxy-(Ci-C8)-alkylthio, (Ci-C8)-Alkylthio-(Ci-C8)-alkylen, (Ci-C8)-Haloalkylthio-(Ci-C8)-alkylthio, (Ci-C8)-Alkylthio-(Ci-C8)-alkylthio, Aminocarbonyl, Aminocarbonyl-(Ci-C8)-alkyl, (Ci-C8)-Alkylaminocarbonyl-(Ci-C8)-alkyl, (C3-C8)- Cycloalkylaminocarbonyl-(Ci-C8)-alkyl, (C2-C8)-Alkenyloxycarbonyl-(Ci-C8)-alkyl, (C3-C8)- Cycloalkyl- (Ci-C8)-alkoxycarbonyl-(Ci-C8)-alkyl, Cyano, Hydroxy-(Ci-C8)-alkyl, (C2-C8)- Alkenyloxy-(Ci-C8)-alkyl, C(0)NR5R6 steht, Heteroaryl (Ci-Cg) alkyl, heterocyclyl (Ci-Cg) alkyl, (C 2 -Cg) alkenyl, (C 2 -Cg) alkynyl, (C 2 -Cg) haloalkenyl, (C 2 -Cg) haloalkynyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -Cio) halocycloalkenyl, aryl- (C 2 -Cg) alkenyl, heteroaryl- (C 2 - Cg) -alkenyl, heterocyclyl- (C 2 -Cg) -alkenyl, aryl- (C 2 -Cg) -alkynyl, heteroaryl- (C 2 -Cg) -alkynyl, heterocyclyl- (C 2 -Cg) -alkynyl, ( C 3 -Cio) cycloalkyl- (C 2 -Cg) alkynyl, arylcarbonyl- (Ci-Cg) -alkyl, (Ci-C 8 ) -alkylcarbonyl- (Ci-Cg) -alkyl, heteroarylcarbonyl- (Ci-Cg ) -alkyl, (C 3 -Cio) -cycloalkylcarbonyl- (Ci-Cg) -alkyl, aryl- (Ci-Cg) -alkoxycarbonyl- (Ci-Cg) -alkyl, (Ci-Cg) -alkoxycarbonyl- (Ci- Cg) -alkyl, (C 1 - C s) - alkoxy- (Ci-Cg) -alkyl, arylcarbonyloxy- (Ci-Cg) -alkyl, heteroarylcarbonyloxy- (Ci-Cg) -alkyl, heterocyclylcarbonyloxy- (Ci-Cg) -alkyl, (Ci-Cg) -alkylcarbonyloxy- (Ci-Cg) -alkyl, (C 3 -C 8 ) - cycloalkylcarbonyloxy- (Ci-Cg) -alkyl, (Ci-Cg) -haloalkoxy- (Ci-Cg) -alkyl, aryl- (Ci-C 8 ) -alkoxy- (Ci-Cg) -alkyl, heteroaiyl- (Ci-C 8 ) -alkoxy- (Ci- C 8 ) -alkyl, CHO, C (0) R 5 , C (0) OR 5 , C (0) NR 6 R 7 , OR 8 , SR 9 , SOR 9 , SO 2 R 9 , NR 5 R 6 , R 5 R 6 N- (Ci-C 8 ) -alkyl, cyano- (Ci-Cg) -alkyl, hydroxycarbonyl- (Ci-Cg) -alkyl, (Ci-C 8 ) -haloalkoxy- (Ci-C 8 ) -alkylthio, (Ci-C 8 ) -alkylthio- (Ci-C 8 ) -alkylene, (Ci-C 8 ) -haloalkylthio- (Ci-C 8 ) -alkylthio, (Ci-C 8 ) -alkylthio- (Ci -C 8 ) -alkylthio, aminocarbonyl, aminocarbonyl- (Ci-C 8 ) -alkyl, (Ci-C8) -alkylaminocarbonyl- (Ci-C 8 ) -alkyl, (C 3 -C 8 ) - cycloalkylaminocarbonyl- (Ci C 8) alkyl, (C 2 -C 8) -Alkenyloxycarbonyl- (Ci-C 8) alkyl, (C 3 -C 8) - cycloalkyl (Ci-C8) alkoxycarbonyl (Ci-C 8) -alkyl, cyano, hydroxy- (Ci-C 8 ) -alkyl, (C 2 -C 8 ) -alkenyloxy- (Ci-C 8 ) -alkyl, C (0) NR 5 R 6 ,
R3 für Hydroxy, Hydrothio, Halogen, NR5R6, (Ci-Cs)-Alkoxy, (C3-Cio)-Cycloalkyl-(Ci-C8)-alkoxy, Aryl-(Ci-C8)-alkoxy, (Ci-C8)-Alkoxy-(Ci-C8)-alkoxy, Arylcarbonyloxy, (Ci-C8)- Alkylcarbonyloxy, Aryl-(Ci-C8)-alkylcarbonyloxy, Heteroarylcarbonyloxy, (C3-C10)- Cycloalkylcarbonyloxy, Heterocyclylcarbonyloxy, (Ci-C8)-Haloalkyl-carbonyloxy, (C2-C8)- Alkenylcarbonyloxy, OC(0)OR5, OC(0)SR9, OC(S)OR8, OC(S)SR9, OSO2R9, OSO2OR8, OCHO steht, R 3 for hydroxy, hydrothio, halogen, NR 5 R 6 , (Ci-Cs) alkoxy, (C 3 -Cio) cycloalkyl- (Ci-C 8 ) alkoxy, aryl- (Ci-C 8 ) alkoxy , (Ci-C 8 ) -alkoxy- (Ci-C 8 ) -alkoxy, arylcarbonyloxy, (Ci-C 8 ) -alkylcarbonyloxy, aryl- (Ci-C 8 ) -alkylcarbonyloxy, heteroarylcarbonyloxy, (C 3 -C 10 ) - Cycloalkylcarbonyloxy, heterocyclylcarbonyloxy, (Ci-C 8 ) -haloalkyl-carbonyloxy, (C 2 -C 8 ) - alkenylcarbonyloxy, OC (0) OR 5 , OC (0) SR 9 , OC (S) OR 8 , OC (S ) SR 9 , OSO 2 R 9 , OSO 2 OR 8 , OCHO,
R4 für Wasserstoff, (Ci-Cs)-Alkyl steht, R5 und R6 gleich oder verschieden sind und unabhängig voneinander für Wasserstoff, (Ci-C8)-Alkyl,R 4 represents hydrogen, (Ci-Cs) -alkyl, R 5 and R 6 are identical or different and independently of one another are hydrogen, (C 1 -C 8 ) -alkyl,
(C2-C8)-Alkenyl, (C2-C8)-Alkinyl, (Ci-C8)-Cyanoalkyl, (Ci-Cio)-Haloalkyl, (C2-C8)-Haloalkenyl, (C2-C8)-Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, (Ci-C8)-Alkoxy-(Ci-C8)-alkyl, (Ci-C8)-Haloalkoxy-(Ci-C8)-alkyl, (Ci-C8)-Alkylthio-(Ci-C8)-alkyl, (Ci-C8)Haloalkylthio-(Ci-C8)-alkyl, (Ci-C8)-Alkoxy-(Ci-C8)- haloalkyl, Aryl, Aryl-(Ci-C8)-alkyl, Heteroaryl, Heteroaryl-(Ci-C8)-alkyl, (C3-Cio)-Cycloalkyl- (Ci-C8)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C8)-alkyl, Heterocyclyl, (Ci-C8)-Alkoxycarbonyl- (Ci-C8)-alkyl, Aryl-(Ci-C8)-Alkoxycarbonyl-(Ci-C8)-alkyl und Heterocyclyl-(Ci-C8)-alkyl stehen, (C 2 -C 8 ) alkenyl, (C 2 -C 8 ) alkynyl, (Ci-C 8 ) cyanoalkyl, (Ci-Cio) haloalkyl, (C 2 -C 8 ) haloalkenyl, (C 2 -C 8 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -Cio) halocycloalkenyl, (Ci-C 8 ) - Alkoxy- (Ci-C 8 ) -alkyl, (Ci-C 8 ) -haloalkoxy- (Ci-C 8 ) -alkyl, (Ci-C 8 ) -alkylthio- (Ci-C 8 ) -alkyl, (Ci- C 8 ) haloalkylthio- (Ci-C 8 ) -alkyl, (Ci-C 8 ) -alkoxy- (Ci-C 8 ) -haloalkyl, aryl, aryl- (Ci-C 8 ) -alkyl, heteroaryl, heteroaryl- ( Ci-C 8 ) -alkyl, (C 3 -Cio) -cycloalkyl- (Ci-C 8 ) -alkyl, (C 4 -Cio) -cycloalkenyl- (Ci-C 8 ) -alkyl, heterocyclyl, (Ci-C 8 ) -alkoxycarbonyl- (Ci-C 8 ) -alkyl, aryl- (Ci-C 8 ) -alkoxycarbonyl- (Ci-C 8 ) -alkyl and heterocyclyl- (Ci-C 8 ) -alkyl,
R7 für H, (Ci-C8)-Alkyl, (C2-C8)-Alkenyl, (C2-C8)-Alkinyl, (Ci-C8)-Cyanoalkyl, (Ci-Cio)- Haloalkyl, (C2-C8)-Haloalkenyl, (C2-C8)-Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-C10)- Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, (Ci-C8)-Alkoxy-(Ci-C8)- alkyl, (Ci-C8)-Alkoxy-(Ci-C8)-haloalkyl, Aryl, Aryl-(Ci-C8)-alkyl, Heteroaryl, Heteroaryl- (Ci-C8)-alkyl, (C3-Cio)-Cycloalkyl-(Ci-C8)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C8)-alkyl, (Ci-C8)- Alkoxycarbonyl-(Ci-C8)-alkyl, (C2-C8)-Alkenyloxycarbonyl-(Ci-C8)-alkyl, Aryl-(Ci-C8)- Alkoxycarbonyl-(Ci-C8)-alkyl, Hydroxycarbonyl-(Ci-C8)-alkyl, Heterocyclyl, Heterocyclyl- (Ci-C8)-alkyl, (Ci-C8)-Alkoxycarbonyl, Aryl-(Ci-C8)-Alkoxycarbonyl, Heteroaryl-(Ci-C8)- alkoxycarbonyl, (C2-C8)-Alkenyloxycarbonyl und (C2-C8)-Alkinyloxycarbonyl steht, und R 7 is H, (Ci-C 8 ) alkyl, (C 2 -C 8 ) alkenyl, (C 2 -C 8 ) alkynyl, (Ci-C 8 ) cyanoalkyl, (Ci-Cio) haloalkyl , (C 2 -C 8 ) haloalkenyl, (C 2 -C 8 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -C 10 ) halocycloalkyl, (C 4 -Cio) cycloalkenyl, ( C 4 -Cio) halocycloalkenyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) alkyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) haloalkyl, aryl, aryl- (Ci -C 8 ) -alkyl, heteroaryl, heteroaryl- (Ci-C 8 ) -alkyl, (C 3 -Cio) -cycloalkyl- (Ci-C 8 ) -alkyl, (C 4 -Cio) -cycloalkenyl- (Ci- C 8 ) alkyl, (Ci-C 8 ) alkoxycarbonyl- (Ci-C 8 ) -alkyl, (C 2 -C 8 ) alkenyloxycarbonyl- (Ci-C 8 ) -alkyl, aryl- (Ci-C 8 ) - Alkoxycarbonyl- (Ci-C 8 ) -alkyl, hydroxycarbonyl- (Ci-C 8 ) -alkyl, heterocyclyl, heterocyclyl- (Ci-C 8 ) -alkyl, (Ci-C 8 ) -alkoxycarbonyl, aryl- (Ci -C 8 ) alkoxycarbonyl, heteroaryl- (Ci-C 8 ) alkoxycarbonyl, (C 2 -C 8 ) alkenyloxycarbonyl and (C 2 -C 8 ) alkynyloxycarbonyl, and
R8 und R9 gleich oder verschieden sind und unabhängig voneinander für (Ci-C8)-Alkyl, (C2-C8)- Alkenyl, (C2-C8)-Alkinyl, (Ci-C8)-Cyanoalkyl, (Ci-Cio)-Haloalkyl, (C2-C8)-Haloalkenyl, (C2-C8)-Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, (Ci-C8)-Alkoxy-(Ci-C8)-alkyl, (Ci-C8)-Alkoxy-(Ci-C8)-haloalkyl, Aryl, Aryl-(Ci-C8)-alkyl, Heteroaryl, Heteroaryl-(Ci-C8)-alkyl, Heterocyclyl-(Ci-C8)-alkyl, (C3-Cio)-Cycloalkyl-(Ci-C8)-alkyl und (C4-Cio)-Cycloalkenyl-(Ci-C8)-alkyl stehen, und wobei die cyclischen Strukturelemente, insbesondere die Strukturelemente Aryl, Cycloalkyl, Cycloalkenyl, Heteroaryl und Heterocyclyl, der jeweils in A1, A2, R1, R2, R3, R4, R5, R6, R7, R8 und R9 genannten Reste unsubstituiert sind, oder durch einen oder mehrere Reste, ausgewählt aus der Gruppe bestehend aus Halogen, Nitro, Hydroxy, Cyano, NR5R6, (Ci-C4)-Alkyl, (Ci-C4)-Haloalkyl, (C1-C4)- Alkoxy, (Ci-C4)-Haloalkoxy, (Ci-C4)-Alkylthio, (Ci-C4)-Alkylsulfoxy, (Ci-C4)-Alkylsulfon, (C1-C4)- Haloalkylthio, (Ci-C4)-Haloalkylsulfoxy, (Ci-C4)-Haloalkylsulfon, (Ci-C4)-Alkoxy-carbonyl, (C1-C4)- Haloalkoxy-carbonyl, (Ci-C4)-Alkylcarboxy, (C3-C6)-Cycloalkyl, (C3-C6)-Cycloalkyl-(Ci-C6)-alkyl, (Ci- C4)-Alkoxy-carbonyl-(Ci-C4)-alkyl, Hydroxycarbonyl, Hydroxycarbonyl-(Ci-C4)-alkyl, C(0)NR6R7, und wobei die Strukturelemente Cycloalkyl bzw. Heterocyclyl n Oxogruppen aufweisen, wobei n = 0, 1 oder 2 ist, substituiert sind. R 8 and R 9 are identical or different and independently of one another for (Ci-C 8 ) alkyl, (C 2 -C 8 ) alkenyl, (C 2 -C 8 ) alkynyl, (Ci-C 8 ) cyanoalkyl , (Ci-Cio) haloalkyl, (C 2 -C 8 ) haloalkenyl, (C 2 -C 8 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -Cio) halocycloalkenyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) alkyl, (Ci-C 8 ) alkoxy- (Ci-C 8 ) haloalkyl , Aryl, aryl- (Ci-C 8 ) -alkyl, heteroaryl, heteroaryl- (Ci-C 8 ) -alkyl, heterocyclyl- (Ci-C 8 ) -alkyl, (C 3 -Cio) -cycloalkyl- (Ci- C 8 ) -alkyl and (C 4 -Cio) -cycloalkenyl- (Ci-C 8 ) -alkyl, and where the cyclic structural elements, in particular the structural elements aryl, cycloalkyl, cycloalkenyl, heteroaryl and heterocyclyl, which are each in A 1 , A 2 , R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 and R 9 are unsubstituted, or by one or more radicals selected from the group consisting of halogen, nitro , Hydroxy, cyano, NR 5 R 6 , (Ci-C 4 ) alkyl, (Ci-C 4 ) haloal kyl, (C 1 -C 4 ) alkoxy, (Ci-C 4 ) haloalkoxy, (Ci-C 4 ) alkylthio, (Ci-C 4 ) alkyl sulfoxy, (Ci-C 4 ) alkyl sulfone, (C 1 -C 4 ) haloalkylthio, (Ci-C 4 ) haloalkylsulfoxy, (Ci-C 4 ) haloalkylsulfone, (Ci-C 4 ) alkoxycarbonyl, (C 1 -C 4 ) haloalkoxycarbonyl, ( Ci-C 4 ) -alkylcarboxy, (C 3 -C 6 ) -cycloalkyl, (C 3 -C 6 ) -cycloalkyl- (Ci-C 6 ) -alkyl, (Ci- C4) -alkoxy-carbonyl- (Ci-C4) -alkyl, hydroxycarbonyl, hydroxycarbonyl- (Ci-C4) -alkyl, C (0) NR 6 R 7 , and wherein the structural elements cycloalkyl or heterocyclyl have n oxo groups, where n = 0, 1 or 2 are substituted.
Die Verbindungen der allgemeinen Formel (1) können durch Anlagerung einer geeigneten The compounds of general formula (1) can be added by adding a suitable one
anorganischen oder organischen Säure, wie beispielsweise Mineralsäuren, wie beispielsweise HCl, HBr, H2SO4, H3P04 oder HNO3, oder organische Säuren, z. B. Carbonsäuren, wie Ameisensäure, Essigsäure, Propionsäure, Oxalsäure, Milchsäure oder Salicylsäure oder Sulfonsäuren, wie zum Beispiel p- Toluolsulfonsäure, an eine basische Gruppe, wie z.B. Amino, Alkylamino, Dialkylamino, Piperidino, Morpholino oder Pyridino, Salze bilden. Diese Salze enthalten dann die konjugierte Base der Säure als Anion. Geeignete Substituenten, die in deprotonierter Form, wie z.B. Sulfonsäuren, bestimmte inorganic or organic acid, such as mineral acids such as HCl, HBr, H2SO4, H3P04 or HNO3, or organic acids, e.g. B. carboxylic acids such as formic acid, acetic acid, propionic acid, oxalic acid, lactic acid or salicylic acid or sulfonic acids, such as p-toluenesulfonic acid, to a basic group such as e.g. Amino, alkylamino, dialkylamino, piperidino, morpholino or pyridino, form salts. These salts then contain the conjugate base of the acid as an anion. Suitable substituents which are in deprotonated form, e.g. Sulfonic acids, certain
Sulfonsäureamide oder Carbonsäuren, vorliegen, können innere Salze mit ihrerseits protonierbaren Gruppen, wie Aminogruppen bilden. Salzbildung kann auch durch Einwirkung einer Base auf Sulphonic acid amides or carboxylic acids are present, inner salts with protonatable groups such as amino groups can form. Salt formation can also be caused by exposure to a base
Verbindungen der allgemeinen Formel (1) erfolgen. Geeignete Basen sind beispielsweise organische Amine, wie Trialkylamine, Morpholin, Piperidin und Pyridin sowie Ammonium-, Alkali- oder Compounds of the general formula (1) are made. Suitable bases are, for example, organic amines, such as trialkylamines, morpholine, piperidine and pyridine, and ammonium, alkali or
Erdalkalimetallhydroxide, -carbonate und -hydrogencarbonate, insbesondere Natrium- und Alkaline earth metal hydroxides, carbonates and bicarbonates, especially sodium and
Kaliumhydroxid, Natrium- und Kaliumcarbonat und Natrium- und Kaliumhydrogencarbonat. Diese Salze sind Verbindungen, in denen der azide Wasserstoff durch ein für die Landwirtschaft geeignetes Kation ersetzt wird, beispielsweise Metallsalze, insbesondere Alkalimetallsalze oder Potassium hydroxide, sodium and potassium carbonate and sodium and potassium hydrogen carbonate. These salts are compounds in which the acidic hydrogen is replaced by a cation suitable for agriculture, for example metal salts, in particular alkali metal salts or
Erdalkalimetallsalze, insbesondere Natrium- und Kaliumsalze, oder auch Ammoniumsalze, Salze mit organischen Aminen oder quartäre Ammoniumsalze, zum Beispiel mit Kationen der Formel Alkaline earth metal salts, especially sodium and potassium salts, or also ammonium salts, salts with organic amines or quaternary ammonium salts, for example with cations of the formula
[NRaRbRcRd]+, worin Ra bis Rd jeweils unabhängig voneinander einen organischen Rest, insbesondere Alkyl, Aryl, Arylalkyl oder Alkylaryl darstellen lnfrage kommen auch Alkylsulfonium- und [NR a R b R c R d ] + , in which R a to R d each independently represent an organic radical, in particular alkyl, aryl, arylalkyl or alkylaryl. Alkylsulfonium and
Alkylsulfoxoniumsalze, wie (Ci-C4)-Trialkylsulfonium- und (Ci-C4)-Trialkylsulfoxoniumsalze. Alkylsulfoxonium salts such as (Ci-C4) -trialkylsulfonium and (Ci-C4) -trialkylsulfoxonium salts.
Die erfindungsgemäßen substituierten Thiazolylpyrrolone der allgemeinen Formel (1) können in Abhängigkeit von äußeren Bedingungen, wie pH-Wert, Lösungsmittel und Temperatur in verschiedenen tautomeren Strukturen vorliegen, die alle von der allgemeinen Formel (1) umfasst sein sollen. lm Folgenden werden die erfindungsgemäß verwendeten Verbindungen der Formel (1) und ihre Salze als "Verbindungen der allgemeinen Formel (1)“ bezeichnet. Depending on external conditions such as pH, solvent and temperature, the substituted thiazolylpyrrolones of the general formula (1) according to the invention can be present in various tautomeric structures, all of which are intended to be encompassed by the general formula (1). The compounds of the formula (1) and their salts used according to the invention are referred to below as "compounds of the general formula (1)".
Bevorzugter Erfindungsgegenstand sind Verbindungen der allgemeinen Formel (1), worin Preferred subject matter of the invention are compounds of the general formula (1), in which
Q für die Gruppe steht, Q for the group stands,
X und Y unabhängig voneinander für C-H oder die Gruppierung C-R1 stehen, wobei X and Y independently of one another represent CH or the grouping CR 1 , where
X für C-H steht, wenn Y für die Gruppierung C-R1 steht und X stands for CH if Y stands for the grouping CR 1 and
X für die Gruppierung C-R1 steht, wenn Y für C-H steht, X stands for the grouping CR 1 , if Y stands for CH,
R1 für Halogen, Cyano, (Ci-Cv)-Alkyl, (Ci-C7)-Haloalkyl, (Ci-Cv)-Hydroxyalkyl, (C1-C7)-R 1 for halogen, cyano, (Ci-Cv) alkyl, (Ci-C 7 ) haloalkyl, (Ci-Cv) hydroxyalkyl, (C 1 -C 7 ) -
Alkoxyalkyl, (Ci-C7)-Alkoxy, (Ci-C7)-Haloalkoxy, (Ci-C7)-Alkylthio, (Ci-C7)-Haloalkylthio, Aryl, Heteroaryl, Aryloxy, Heteroaryloxy, Heterocyclyl, (C3-Cio)-Cycloalkyl, (C3-C10)- Cycloalkyl-(Ci-C7)-alkyl, (C3-C8)-Halocycloalkyl, (C3-C8)-Halocycloalkyl-(Ci-C7)-alkyl, (Ci-C7)-Alkylcarbonyl, (C2-C7)-Alkenyl, (C2-C7)-Alkinyl steht, Alkoxyalkyl, (Ci-C 7 ) alkoxy, (Ci-C 7 ) haloalkoxy, (Ci-C 7 ) alkylthio, (Ci-C 7 ) haloalkylthio, aryl, heteroaryl, aryloxy, heteroaryloxy, heterocyclyl, (C 3 -Cio) cycloalkyl, (C 3 -C 10 ) cycloalkyl- (Ci-C 7 ) alkyl, (C 3 -C 8 ) halocycloalkyl, (C 3 -C 8 ) halocycloalkyl- (Ci-C 7) alkyl, (Ci-C 7) alkylcarbonyl, (C 2 -C 7) -alkenyl, (C 2 -C 7) alkynyl,
A1 und A2 unabhängig voneinander für C-H oder die Gruppierung C-R2 stehen, wobei A 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
A1 für C-H steht, wenn A2 für die Gruppierung C-R2 steht und A 1 stands for CH if A 2 stands for the grouping CR 2 and
A1 für die Gruppierung C-R2 steht, wenn A2 für C-H steht, A 1 stands for the grouping CR 2 , if A 2 stands for CH,
R2 für Hydrothio, Hydroxy, Halogen, (Ci-C7)-Alkyl, (Ci-C7)-Haloalkyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Cycloalkyl-(Ci-C7)-alkyl, Aryl, Heteroaryl, Heterocyclyl, Aryl-(Ci-C7)-alkyl, Heteroaryl-(Ci-C7)-alkyl, Heterocyclyl-(Ci-C7)-alkyl, (C2-C7)-Alkenyl, (C2-C7)-Alkinyl, (C2-C7)-Haloalkenyl, (C2-C7)-Haloalkinyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, Aryl-(C2-C7)-alkenyl, Heteroaryl-(C2-C7)-alkenyl, Heterocyclyl- (C2-C7)-alkenyl, Aryl-(C2-C7)-alkinyl, Heteroaryl-(C2-C7)-alkinyl, Heterocyclyl-(C2-C7)-alkinyl, (C3-Cio)-Cycloalkyl-(C2-C7)-alkinyl, Arylcarbonyl-(Ci-C7)-alkyl, (Ci-C7)-Alkylcarbonyl- (Ci-C7)-alkyl, Heteroarylcarbonyl-(Ci-C7)-alkyl, (C3-Cio)-Cycloalkylcarbonyl-(Ci-C7)-alkyl, Aryl-(Ci-C7)-alkoxycarbonyl-(Ci-C7)-alkyl, (Ci-C7)-Alkoxycarbonyl-(Ci-C7)-alkyl, (C1-C7)- Alkoxy-(Ci-C7)-alkyl, Arylcarbonyloxy-(Ci-C8)-alkyl, Heteroarylcarbonyloxy-(Ci-C7)-alkyl, Heterocyclylcarbonyloxy-(Ci-C7)-alkyl, (Ci-C7)-Alkylcarbonyloxy-(Ci-C7)-alkyl, (C3-C8)- Cycloalkylcarbonyloxy-(Ci-C7)-alkyl, (Ci-C7)-Haloalkoxy-(Ci-C7)-alkyl, Aryl-(Ci-C7)-alkoxy- (Ci-C7)-alkyl, Heteroaryl-(Ci-C7)-alkoxy-(Ci-C7)-alkyl, CHO, C(0)R5, C(0)OR5, C(0)NR6R7, OR8, SR9, SOR9, SO2R9, NR6R7, R6R7N-(Ci-C7)-alkyl, Cyano-(Ci-C8)-alkyl, Hydroxycarbonyl- (Ci-C7)-alkyl, (Ci-C8)-Haloalkoxy-(Ci-C8)-alkylthio, (Ci-C7)-Alkylthio-(Ci-C7)-alkylen, (Ci-C7)-Haloalkylthio-(Ci-C7)-alkylthio, (Ci-C7)-Alkylthio-(Ci-C7)-alkylthio, Aminocarbonyl, Aminocarbonyl-(Ci-C7)-alkyl, (Ci-C7)-Alkylaminocarbonyl-(Ci-C7)-alkyl, (C3-C8)- Cycloalkylaminocarbonyl-(Ci-C7)-alkyl, (C2-C7)-Alkenyloxycarbonyl-(Ci-C7)-alkyl, (C3-C8)- Cycloalkyl- (Ci-C7)-alkoxycarbonyl-(Ci-C7)-alkyl, Cyano, Hydroxy-(Ci-C7)-alkyl, (C2-C7)- Alkenyloxy-(Ci-C7)-alkyl steht, R 2 for hydrothio, hydroxy, halogen, (Ci-C 7 ) alkyl, (Ci-C 7 ) haloalkyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) cycloalkyl- (Ci-C 7 ) alkyl, aryl, heteroaryl, heterocyclyl, aryl (Ci-C 7 ) alkyl, heteroaryl (Ci-C 7 ) alkyl, heterocyclyl (Ci-C 7 ) alkyl, (C 2 -C 7 ) Alkenyl, (C 2 -C 7 ) alkynyl, (C 2 -C 7 ) haloalkenyl, (C 2 -C 7 ) haloalkynyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl , (C 4 -Cio) halocycloalkenyl, aryl- (C 2 -C 7 ) alkenyl, heteroaryl- (C 2 -C 7 ) alkenyl, heterocyclyl- (C 2 -C 7 ) alkenyl, aryl- (C 2 -C 7 ) alkynyl, heteroaryl- (C 2 -C 7 ) alkynyl, heterocyclyl- (C 2 -C 7 ) alkynyl, (C 3 -Cio) cycloalkyl- (C 2 -C 7 ) alkynyl , Arylcarbonyl- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -alkylcarbonyl- (Ci-C 7 ) -alkyl, heteroarylcarbonyl- (Ci-C 7 ) -alkyl, (C 3 -Cio) -cycloalkylcarbonyl- (Ci-C 7 ) -alkyl, aryl- (Ci-C 7 ) -alkoxycarbonyl- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -alkoxycarbonyl- (Ci-C 7 ) -alkyl, (C 1 -C 7 ) - alkoxy- (Ci-C 7 ) -alkyl, arylcarbonyloxy- (Ci-C 8 ) -alkyl, heteroarylcarbonyloxy- (Ci-C 7 ) -alk yl, heterocyclylcarbonyloxy- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -alkylcarbonyloxy- (Ci-C 7 ) -alkyl, (C 3 -C 8 ) - cycloalkylcarbonyloxy- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -haloalkoxy- (Ci-C 7 ) -alkyl, aryl- (Ci-C 7 ) -alkoxy- (Ci-C 7 ) -alkyl, heteroaryl- (Ci-C 7 ) -alkoxy- ( Ci-C 7 ) -alkyl, CHO, C (0) R 5 , C (0) OR 5 , C (0) NR 6 R 7 , OR 8 , SR 9 , SOR 9 , SO 2 R 9 , NR 6 R 7 , R 6 R 7 N- (Ci-C 7 ) -alkyl, cyano- (Ci-C 8 ) -alkyl, hydroxycarbonyl- (Ci-C 7 ) -alkyl, (Ci-C 8 ) -haloalkoxy- (Ci -C 8 ) -alkylthio, (Ci-C 7 ) -alkylthio- (Ci-C 7 ) -alkylene, (Ci-C 7 ) -haloalkylthio- (Ci-C 7 ) -alkylthio, (Ci-C 7 ) - Alkylthio- (Ci-C 7 ) -alkylthio, aminocarbonyl, Aminocarbonyl- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -alkylaminocarbonyl- (Ci-C 7 ) -alkyl, (C 3 -C 8 ) - cycloalkylaminocarbonyl- (Ci-C 7 ) -alkyl, (C 2 -C 7 ) alkenyloxycarbonyl- (Ci-C 7 ) -alkyl, (C 3 -C 8 ) - cycloalkyl- (Ci-C 7 ) -alkoxycarbonyl- (Ci-C 7 ) -alkyl, cyano, hydroxy- ( Ci-C 7 ) -alkyl, (C 2 -C 7 ) - alkenyloxy- (Ci-C 7 ) -alkyl,
R3 für Hydroxy, Hydrothio, Halogen, NR5R6, (Ci-C7)-Alkoxy, (C3-Cio)-Cycloalkyl-(Ci-C7)-alkoxy, Aryl-(Ci-C7)-alkoxy, (Ci-C7)-Alkoxy-(Ci-C7)-alkoxy, Arylcarbonyloxy, (Ci-C7)- Alkylcarbonyloxy, Aryl-(Ci-C7)-alkylcarbonyloxy, Heteroarylcarbonyloxy, (C3-C10)- Cycloalkylcarbonyloxy, Heterocyclylcarbonyloxy, (Ci-C7)-Haloalkyl-carbonyloxy, (C2-C7)- Alkenylcarbonyloxy, OC(0)OR5, OC(0)SR9, OC(S)OR8, OC(S)SR9, 0S02R9, 0S020R8, OCHO steht, R 3 for hydroxy, hydrothio, halogen, NR 5 R 6 , (Ci-C 7 ) alkoxy, (C3-Cio) cycloalkyl- (Ci-C 7 ) alkoxy, aryl- (Ci-C 7 ) alkoxy , (Ci-C 7 ) alkoxy- (Ci-C 7 ) alkoxy, arylcarbonyloxy, (Ci-C 7 ) alkylcarbonyloxy, aryl- (Ci-C 7 ) alkylcarbonyloxy, heteroarylcarbonyloxy, (C3-C10) - cycloalkylcarbonyloxy , Heterocyclylcarbonyloxy, (Ci-C 7 ) -haloalkyl-carbonyloxy, (C 2 -C 7 ) - alkenylcarbonyloxy, OC (0) OR 5 , OC (0) SR 9 , OC (S) OR 8 , OC (S) SR 9 , 0S0 2 R 9 , 0S0 2 0R 8 , OCHO stands,
R4 für Wasserstoff, (Ci-C7)-Alkyl steht, R 4 represents hydrogen, (Ci-C 7 ) alkyl,
R5 und R6 gleich oder verschieden sind und unabhängig voneinander für Wasserstoff, (Ci-C7)-Alkyl,R 5 and R 6 are identical or different and independently of one another are hydrogen, (C 1 -C 7 ) -alkyl,
(C2-C7)-Alkenyl, (C2-C7)-Alkinyl, (Ci-C7)-Cyanoalkyl, (Ci-C7)-Haloalkyl, (C2-C7)-Haloalkenyl, (C2-C7)-Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, (Ci-C7)-Alkoxy-(Ci-C7)-alkyl, (Ci-C7)-Haloalkoxy-(Ci-C7)-alkyl, (Ci-C7)-Alkylthio-(Ci-C7)-alkyl, (Ci-C7)Haloalkylthio-(Ci-C7)-alkyl, (Ci-C7)-Alkoxy-(Ci-C7)- haloalkyl, Aryl, Aryl-(Ci-C7)-alkyl, Heteroaryl, Heteroaryl-(Ci-C7)-alkyl, (C3-Cio)-Cycloalkyl- (Ci-C7)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C7)-alkyl, Heterocyclyl, (Ci-C7)-Alkoxycarbonyl- (Ci-C7)-alkyl Aryl-(Ci-C7)-Alkoxycarbonyl-(Ci-C7)-alkyl und Heterocyclyl-(Ci-C7)-alkyl stehen, (C 2 -C 7 ) alkenyl, (C 2 -C 7 ) alkynyl, (Ci-C 7 ) cyanoalkyl, (Ci-C 7 ) haloalkyl, (C 2 -C 7 ) haloalkenyl, (C 2 -C 7 ) -haloalkynyl, (C3-Cio) -cycloalkyl, (C3-Cio) -halocycloalkyl, (C4-Cio) -cycloalkenyl, (C4-Cio) -halocycloalkenyl, (Ci-C 7 ) -alkoxy- ( Ci-C 7 ) -alkyl, (Ci-C 7 ) -haloalkoxy- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -alkylthio- (Ci-C 7 ) -alkyl, (Ci-C 7 ) Haloalkylthio- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -alkoxy- (Ci-C 7 ) -haloalkyl, aryl, aryl- (Ci-C 7 ) -alkyl, heteroaryl, heteroaryl- (Ci-C 7 ) -alkyl, (C3-Cio) -cycloalkyl- (Ci-C 7 ) -alkyl, (C 4 -Cio) -cycloalkenyl- (Ci-C 7 ) -alkyl, heterocyclyl, (Ci-C 7 ) -alkoxycarbonyl - (Ci-C 7 ) -alkyl aryl- (Ci-C 7 ) -alkoxycarbonyl- (Ci-C 7 ) -alkyl and heterocyclyl- (Ci-C 7 ) -alkyl,
R7 für H, (Ci-C7)-Alkyl, (C2-C7)-Alkenyl, (C2-C7)-Alkinyl, (Ci-C7)-Cyanoalkyl, (Ci-C7)-Haloalkyl, (C2-C7)-Haloalkenyl, (C2-C7)-Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, (Ci-C7)-Alkoxy-(Ci-C7)-alkyl, (Ci-C7)- Alkoxy-(Ci-C7)-haloalkyl, Aryl, Aryl-(Ci-C7)-alkyl, Heteroaryl, Heteroaryl-(Ci-C7)-alkyl, (C3-Cio)-Cycloalkyl-(Ci-C7)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C7)-alkyl, (Ci-C7)- Alkoxycarbonyl-(Ci-C7)-alkyl, (C2-C7)-Alkenyloxycarbonyl-(Ci-C7)-alkyl, Aryl-(Ci-C7)- Alkoxycarbonyl-(Ci-C7)-alkyl, Hydroxycarbonyl-(Ci-C7)-alkyl, Heterocyclyl, Heterocyclyl- (Ci-C7)-alkyl steht, R 7 for H, (Ci-C 7 ) alkyl, (C 2 -C 7 ) alkenyl, (C 2 -C 7 ) alkynyl, (Ci-C 7 ) cyanoalkyl, (Ci-C 7 ) - Haloalkyl, (C 2 -C 7 ) haloalkenyl, (C 2 -C 7 ) haloalkynyl, (C3-Cio) cycloalkyl, (C3-Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -Cio) halocycloalkenyl, (Ci-C 7 ) alkoxy- (Ci-C 7 ) alkyl, (Ci-C 7 ) - alkoxy- (Ci-C 7 ) haloalkyl, aryl, aryl- (Ci-C 7 ) -alkyl, heteroaryl, heteroaryl- (Ci-C 7 ) -alkyl, (C 3 -Cio) -cycloalkyl- (Ci-C 7 ) -alkyl, (C 4 -Cio) -cycloalkenyl- (Ci-C 7 ) -alkyl, (Ci-C 7 ) -alkoxycarbonyl- (Ci-C 7 ) -alkyl, (C 2 -C 7 ) -alkenyloxycarbonyl- (Ci-C 7 ) -alkyl, aryl- (Ci-C 7 ) - Alkoxycarbonyl- (Ci-C 7 ) -alkyl, hydroxycarbonyl- (Ci-C 7 ) -alkyl, heterocyclyl, heterocyclyl- (Ci-C 7 ) -alkyl,
R8 und R9 gleich oder verschieden sind und unabhängig voneinander für (Ci-C7)-Alkyl, (C2-C7)-R 8 and R 9 are identical or different and independently of one another for (Ci-C 7 ) -alkyl, (C 2 -C 7 ) -
Alkenyl, (C2-C7)-Alkinyl, (Ci-C7)-Cyanoalkyl, (Ci-C7)-Haloalkyl, (C2-C7)-Haloalkenyl, (C2-C7)- Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-C10)- Halocycloalkenyl, (Ci-C7)-Alkoxy-(Ci-Cv)-alkyl, (Ci-C7)-Alkoxy-(Ci-Cv)-haloalkyl, Aryl, Aryl- (Ci-C7)-alkyl, Heteroaryl, Heteroaryl-(Ci-C7)-alkyl, Heterocyclyl-(Ci-C7)-alkyl, (C3-C10)- Cycloalkyl-(Ci-C7)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C7)-alkyl und NR5R6 stehen, und wobei die cyclischen Strukturelemente, insbesondere die Strukturelemente Aryl, Cycloalkyl, Cycloalkenyl, Heteroaryl und Heterocyclyl, der jeweils in A1, A2, R1, R2, R3, R4, R5, R6, R7, R8 und R9 genannten Reste unsubstituiert sind, oder durch einen oder mehrere Reste, ausgewählt aus der Gruppe bestehend aus Halogen, Nitro, Hydroxy, Cyano, NR5R6, (Ci-C4)-Alkyl, (Ci-C4)-Haloalkyl, (C1-C4)- Alkoxy, (Ci-C4)-Haloalkoxy, (Ci-C4)-Alkylthio, (Ci-C4)-Alkylsulfoxy, (Ci-C4)-Alkylsulfon, (C1-C4)- Haloalkylthio, (Ci-C4)-Haloalkylsulfoxy, (Ci-C4)-Haloalkylsulfon, (Ci-C4)-Alkoxy-carbonyl, (C1-C4)- Haloalkoxy-carbonyl, (Ci-C4)-Alkylcarboxy, (C3-C6)-Cycloalkyl, (C3-C6)-Cycloalkyl-(Ci-C6)-alkyl, (Ci- C4)-Alkoxy-carbonyl-(Ci-C4)-alkyl, Hydroxycarbonyl, Hydroxycarbonyl-(Ci-C4)-alkyl, C(0)NR6R7, und wobei die Strukturelemente Cycloalkyl bzw. Heterocyclyl n Oxogruppen aufweisen, wobei n = 0, 1 oder 2 ist, substituiert sind. Alkenyl, (C 2 -C 7 ) alkynyl, (Ci-C 7 ) cyanoalkyl, (Ci-C 7 ) haloalkyl, (C 2 -C 7 ) haloalkenyl, (C 2 -C 7 ) - Haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -C 10 ) halocycloalkenyl, (Ci-C 7 ) alkoxy- (Ci -Cv) alkyl, (Ci-C 7 ) alkoxy- (Ci-Cv) haloalkyl, aryl, aryl- (Ci-C 7 ) alkyl, heteroaryl, heteroaryl- (Ci-C 7 ) alkyl, heterocyclyl - (Ci-C 7 ) alkyl, (C 3 -C 10 ) - cycloalkyl- (Ci-C 7 ) alkyl, (C 4 -Cio) cycloalkenyl- (Ci-C 7 ) alkyl and NR 5 R 6 , and wherein the cyclic structural elements, in particular the structural elements aryl, cycloalkyl, cycloalkenyl, heteroaryl and heterocyclyl, each of which is in A 1 , A 2 , R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 and R 9 radicals are unsubstituted, or by one or more radicals selected from the group consisting of halogen, nitro, hydroxy, cyano, NR 5 R 6 , (Ci-C 4 ) alkyl, (Ci C 4 ) haloalkyl, (C 1 -C 4 ) alkoxy, (Ci-C 4 ) haloalkoxy, (Ci-C 4 ) alkylthio, (Ci-C 4 ) alkylsulfoxy, (Ci-C 4 ) - Alkyl sulfone, (C 1 -C 4 ) haloalkylthio, (Ci-C 4 ) haloalkylsulfoxy, (Ci-C 4 ) haloalkyl sulfone, ( Ci-C 4 ) alkoxy carbonyl, (C 1 -C 4 ) haloalkoxy carbonyl, (Ci-C 4 ) alkyl carboxy, (C 3 -C 6 ) cycloalkyl, (C 3 -C 6 ) cycloalkyl - (Ci-C 6 ) -alkyl, (Ci-C 4 ) -alkoxy-carbonyl- (Ci-C 4 ) -alkyl, hydroxycarbonyl, hydroxycarbonyl- (Ci-C 4 ) -alkyl, C (0) NR 6 R 7 , and wherein the structural elements cycloalkyl or heterocyclyl have n oxo groups, where n = 0, 1 or 2, are substituted.
Besonders bevorzugter Erfindungsgegenstand sind Verbindungen der allgemeinen Formel (I), worin Particularly preferred subject matter of the invention are compounds of the general formula (I), in which
Q für die Gruppe Q for the group
steht stands
X und Y unabhängig voneinander für C-H oder die Gruppierung C-R1 stehen, wobei X and Y independently of one another represent CH or the grouping CR 1 , where
X für C-H steht, wenn Y für die Gruppierung C-R1 steht und X stands for CH if Y stands for the grouping CR 1 and
X für die Gruppierung C-R1 steht, wenn Y für C-H steht, X stands for the grouping CR 1 , if Y stands for CH,
R1 Halogen, Cyano, (Ci-C6)-Alkyl, (Ci-Cej-Haloalkyl, (Ci-Cej-Hydroxyalkyl, (G-G)-R 1 halogen, cyano, (Ci-C 6 ) -alkyl, (Ci-Cej-haloalkyl, (Ci-Cej-hydroxyalkyl, (GG) -
Alkoxyalkyl, (G-G)-Alkoxy, (Ci-C6)-Haloalkoxy, (Ci-C6)-Alkylthio, (Ci-C6)-Haloalkylthio, Aryl, Heteroaryl, Aryloxy, Heteroaryloxy, Heterocyclyl, (C3-Cio)-Cycloalkyl, (C3-C10)- Cycloalkyl-(Ci-C6)-alkyl, (C3-C6)-Halocycloalkyl, (C3-C6)-Halocycloalkyl-(Ci-C6)-alkyl, (Ci-C6)-Alkylcarbonyl, (C2-G,)-Alkcnyl, (C2-C6)-Alkinyl steht, Alkoxyalkyl, (GG) -alkoxy, (Ci-C 6 ) -haloalkoxy, (Ci-C 6 ) -alkylthio, (Ci-C 6 ) -haloalkylthio, aryl, heteroaryl, aryloxy, heteroaryloxy, heterocyclyl, (C 3 -Cio ) -Cycloalkyl, (C 3 -C 10 ) - cycloalkyl- (Ci-C 6 ) -alkyl, (C 3 -C 6 ) -halocycloalkyl, (C 3 -C 6 ) -halocycloalkyl- (Ci-C 6 ) - alkyl, (Ci-C 6 ) -alkylcarbonyl, (C 2 -G,) -alkynyl, (C 2 -C 6 ) -alkynyl,
A1 und A2 unabhängig voneinander für C-H oder die Gruppierung C-R2 stehen, wobei A 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
A1 für C-H steht, wenn A2 für die Gruppierung C-R2 steht und A1 für die Gruppierung C-R2 steht, wenn A2 für C-H steht, A 1 stands for CH if A 2 stands for the grouping CR 2 and A 1 stands for the grouping CR 2 , if A 2 stands for CH,
R2 für Cyano, Halogen, (Ci-C6)-Alkyl, (G-C6)-Haloalkyl, (Ci-C6)-Hydroxyalkyl, (G-G)-R 2 for cyano, halogen, (Ci-C6) alkyl, (G-C6) haloalkyl, (Ci-C6) hydroxyalkyl, (GG) -
Alkoxyalkyl, (Ci-Ce)-Alkoxy, (Ci-C6)-Haloalkoxy, (Ci-C6)-Alkylthio, (Ci-C6)-Haloalkylthio, Aryl, Heteroaryl, Aryloxy, Heteroaryloxy, Heterocyclyl, (C3-Cio)-Cycloalkyl, (C3-C10)- Cycloalkyl-(Ci-C6)-alkyl, (C3-C8)-Halocycloalkyl, (C3-C8)-Halocycloalkyl-(Ci-C6)-alkyl, (Ci-C6)-Alkylcarbonyl, (C2-Ce)-Alkenyl, (C2-C6)-Alkinyl steht, Alkoxyalkyl, (Ci-Ce) -alkoxy, (Ci-C6) -haloalkoxy, (Ci-C6) -alkylthio, (Ci-C6) -haloalkylthio, aryl, heteroaryl, aryloxy, heteroaryloxy, heterocyclyl, (C 3 -Cio) -Cycloalkyl, (C 3 -C 10 ) -cycloalkyl- (Ci-C6) -alkyl, (C 3 -C 8 ) -halocycloalkyl, (C 3 -C 8 ) -halocycloalkyl- (Ci-C 6 ) -alkyl, (Ci-C6) alkylcarbonyl, (C 2 -Ce) alkenyl, (C 2 -C 6 ) alkynyl,
R3 für Hydroxy, Hydrothio, Halogen, NR5R6, (Ci-Ce)-Alkoxy, (C3-Cio)-Cycloalkyl-(Ci-C6)-alkoxy, Aryl-(Ci-C6)-alkoxy, (Ci-C6)-Alkoxy-(Ci-C6)-alkoxy, Arylcarbonyloxy, (G-G)- Alkylcarbonyloxy, Aryl-(Ci-C6)-alkylcarbonyloxy, Heteroarylcarbonyloxy, (C3-G0)- Cycloalkylcarbonyloxy, Heterocyclylcarbonyloxy, (Ci-C6)-Haloalkyl-carbonyloxy, (C2-C6)- Alkenylcarbonyloxy, OC(0)OR5, OC(0)SR9, OC(S)OR8, OC(S)SR9, OSO2R8, OSO2OR7, OCHO steht, R 3 for hydroxy, hydrothio, halogen, NR 5 R 6 , (Ci-Ce) alkoxy, (C 3 -Cio) cycloalkyl- (Ci-C 6 ) alkoxy, aryl- (Ci-C6) alkoxy, (Ci-C6) alkoxy- (Ci-C6) alkoxy, arylcarbonyloxy, (GG) - alkylcarbonyloxy, aryl- (Ci-C6) alkylcarbonyloxy, heteroarylcarbonyloxy, (C 3 -G 0 ) - cycloalkylcarbonyloxy, heterocyclylcarbonyloxy, (Ci -C6) haloalkylcarbonyloxy, (C 2 -C 6 ) alkenylcarbonyloxy, OC (0) OR 5 , OC (0) SR 9 , OC (S) OR 8 , OC (S) SR 9 , OSO 2 R 8 , OSO 2 OR 7 , OCHO stands,
R4 für Wasserstoff, (G-Ce)-Alkyl steht, R 4 represents hydrogen, (G-Ce) alkyl,
R5 und R6 gleich oder verschieden sind und unabhängig voneinander für Wasserstoff, (G-Ce)-Alkyl,R 5 and R 6 are identical or different and independently of one another are hydrogen, (G-Ce) -alkyl,
(C2-C6)-Alkenyl, (C2-C6)-Alkinyl, (Ci-C6)-Cyanoalkyl, (Ci-C6)-Haloalkyl, (C2-C6)-Haloalkenyl, (C2-C6)-Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Go)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, (Ci-C6)-Alkoxy-(Ci-C6)-alkyl, (Ci-C6)-Haloalkoxy-(Ci-C6)-alkyl, (Ci-C6)-Alkylthio-(Ci-C6)-alkyl, (Ci-C6)Haloalkylthio-(Ci-C6)-alkyl, (G-C6)-Alkoxy-(G-C6)- haloalkyl, Aryl, Aryl-(Ci-C6)-alkyl, Heteroaryl, Heteroaryl-(G-C6)-alkyl, (C3-Go)-Cycloalkyl- (Ci-C6)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C6)-alkyl, Heterocyclyl, (G-Cv)-Alkoxycarbonyl- (Ci-C6)-alkyl, Aryl-(Ci-C6)-Alkoxycarbonyl-(Ci-C6)-alkyl und Heterocyclyl-(Ci-C6)-alkyl stehen, (C 2 -C 6 ) alkenyl, (C 2 -C 6 ) alkynyl, (Ci-C 6 ) cyanoalkyl, (Ci-C 6 ) haloalkyl, (C 2 -C 6 ) haloalkenyl, (C 2 -C 6 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Go) cycloalkenyl, (C 4 -Cio) halocycloalkenyl, (Ci-C6) - alkyl alkoxy (Ci-C6) alkyl, (Ci-C6) haloalkoxy (Ci-C6) alkyl, (Ci-C6) alkylthio (Ci-C 6), (Ci-C6) Haloalkylthio- (Ci-C 6 ) alkyl, (GC 6 ) alkoxy- (GC 6 ) haloalkyl, aryl, aryl- (Ci-C6) alkyl, heteroaryl, heteroaryl- (G-C6) alkyl, (C 3 -Go) -cycloalkyl- (Ci-C6) -alkyl, (C 4 -Cio) -cycloalkenyl- (Ci-C 6 ) -alkyl, heterocyclyl, (G-Cv) -alkoxycarbonyl- (Ci-C6) -alkyl, Aryl- (Ci-C6) -alkoxycarbonyl- (Ci-C6) -alkyl and heterocyclyl- (Ci-C6) -alkyl,
R7 für H, (Ci-C6)-Alkyl, (C2-C6)-Alkenyl, (C2-C6)-Alkinyl, (Ci-C6)-Cyanoalkyl, (Ci-C6)-Haloalkyl, (C2-C6)-Haloalkenyl, (C2-C6)-Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-Cio)-Halocycloalkenyl, (G-C6)-Alkoxy-(G-C6)-alkyl, (Ci-C6)- Alkoxy-(Ci-C6)-haloalkyl, Aryl, Aryl-(Ci-C6)-alkyl, Heteroaryl, Heteroaryl-(G-C6)-alkyl, (C3-Cio)-Cycloalkyl-(Ci-C6)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C6)-alkyl, (Ci-C6)- Alkoxycarbonyl-(G-C6)-alkyl, (C2-C6)-Alkenyloxycarbonyl-(Ci-C6)-alkyl, Aryl-(G-G)- Alkoxycarbonyl-(Ci-C6)-alkyl, Hydroxycarbonyl-(Ci-C6)-alkyl, Heterocyclyl, Heterocyclyl- (Ci-C6)-alkyl steht, und R8 und R9 gleich oder verschieden sind und unabhängig voneinander für (Ci-C6)-Alkyl, (C2-CÖ)-R 7 for H, (Ci-C 6 ) alkyl, (C 2 -C 6 ) alkenyl, (C 2 -C 6 ) alkynyl, (Ci-C 6 ) cyanoalkyl, (Ci-C 6 ) - Haloalkyl, (C 2 -C 6 ) haloalkenyl, (C 2 -C 6 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, ( C 4 -Cio) halocycloalkenyl, (GC 6 ) alkoxy- (GC 6 ) alkyl, (Ci-C 6 ) alkoxy- (Ci-C6) haloalkyl, aryl, aryl- (Ci-C6) alkyl , Heteroaryl, heteroaryl- (G-C6) -alkyl, (C 3 -Cio) -cycloalkyl- (Ci-C 6 ) -alkyl, (C 4 -Cio) -cycloalkenyl- (Ci-C 6 ) -alkyl, ( Ci-C 6 ) alkoxycarbonyl- (G-C6) -alkyl, (C 2 -C 6 ) -alkenyloxycarbonyl- (Ci-C 6 ) -alkyl, aryl- (GG) -alkoxycarbonyl- (Ci-C6) -alkyl , Hydroxycarbonyl- (Ci-C6) -alkyl, heterocyclyl, heterocyclyl- (Ci-C6) -alkyl, and R 8 and R 9 are the same or different and independently of one another for (Ci-C 6 ) alkyl, (C 2 -C Ö ) -
Alkenyl, (C2-C6)-Alkinyl, (Ci-C6)-Cyanoalkyl, (Ci-C6)-Haloalkyl, (C2-C6)-Haloalkenyl, (C2-C6)- Haloalkinyl, (C3-Cio)-Cycloalkyl, (C3-Cio)-Halocycloalkyl, (C4-Cio)-Cycloalkenyl, (C4-C10)- Halocycloalkenyl, (Ci-C6)-Alkoxy-(Ci-C6)-alkyl, (Ci-C6)-Alkoxy-(Ci-C6)-haloalkyl, Aryl, Aryl- (Ci-C6)-alkyl, Heteroaryl, Heteroaryl-(Ci-C6)-alkyl, Heterocyclyl-(Ci-C6)-alkyl, (C3-C10)- Cycloalkyl-(Ci-C6)-alkyl, (C4-Cio)-Cycloalkenyl-(Ci-C6)-alkyl stehen, und wobei die cyclischen Strukturelemente, insbesondere die Strukturelemente Aryl, Cycloalkyl, Cycloalkenyl, Heteroaryl und Heterocyclyl, der jeweils in A1, A2, R1, R2, R3, R4, R5, R6, R7, R8 und R9 genannten Reste unsubstituiert sind, oder durch einen oder mehrere Reste, ausgewählt aus der Gruppe bestehend aus Halogen, Nitro, Hydroxy, Cyano, NR5R6, (Ci-C4)-Alkyl, (Ci-C4)-Haloalkyl, (C1-C4)- Alkoxy, (Ci-C4)-Haloalkoxy, (Ci-C4)-Alkylthio, (Ci-C4)-Alkylsulfoxy, (Ci-C4)-Alkylsulfon, (C1-C4)- Haloalkylthio, (Ci-C4)-Haloalkylsulfoxy, (Ci-C4)-Haloalkylsulfon, (Ci-C4)-Alkoxy-carbonyl, (C1-C4)- Haloalkoxy-carbonyl, (Ci-C4)-Alkylcarboxy, (C3-C6)-Cycloalkyl, (C3-C6)-Cycloalkyl-(Ci-C6)-alkyl, (Ci- C4)-Alkoxy-carbonyl-(Ci-C4)-alkyl, Hydroxycarbonyl, Hydroxycarbonyl-(Ci-C4)-alkyl, C(0)NR6R7, und wobei die Strukturelemente Cycloalkyl bzw. Heterocyclyl n Oxogruppen aufweisen, wobei n = 0, 1 oder 2 ist, substituiert sind. Alkenyl, (C 2 -C 6 ) alkynyl, (Ci-C 6 ) cyanoalkyl, (Ci-C 6 ) haloalkyl, (C 2 -C 6 ) haloalkenyl, (C 2 -C 6 ) haloalkynyl, (C 3 -Cio) cycloalkyl, (C 3 -Cio) halocycloalkyl, (C 4 -Cio) cycloalkenyl, (C 4 -C 10 ) halocycloalkenyl, (Ci-C 6 ) alkoxy- (Ci-C 6 ) -alkyl, (Ci-C 6 ) -alkoxy- (Ci-C 6 ) -haloalkyl, aryl, aryl- (Ci-C 6 ) -alkyl, heteroaryl, heteroaryl- (Ci-C 6 ) -alkyl, heterocyclyl - (Ci-C 6 ) -alkyl, (C 3 -C 10 ) - cycloalkyl- (Ci-C 6 ) -alkyl, (C 4 -Cio) -cycloalkenyl- (Ci-C 6 ) -alkyl, and wherein the cyclic structural elements, in particular the structural elements aryl, cycloalkyl, cycloalkenyl, heteroaryl and heterocyclyl, each of which is in A 1 , A 2 , R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 and R 9 radicals are unsubstituted, or by one or more radicals selected from the group consisting of halogen, nitro, hydroxy, cyano, NR 5 R 6 , (Ci-C 4 ) alkyl, (Ci-C 4 ) haloalkyl , (C 1 -C 4 ) alkoxy, (Ci-C 4 ) haloalkoxy, (Ci-C 4 ) alkylthio, (Ci-C 4 ) alkylsulfoxy, (Ci-C 4 ) alkyl sulfone, (C 1 -C 4 ) haloalkylthio, (Ci-C 4 ) haloalkylsulfoxy, (Ci-C 4 ) haloalkyl sulfone, (Ci-C 4 ) alkoxycarbonyl, (C 1 -C 4 ) - Haloalkoxy-carbonyl, (Ci-C 4 ) -alkylcarboxy, (C 3 -C 6 ) -cycloalkyl, (C 3 -C 6 ) -cycloalkyl- (Ci-C 6 ) -alkyl, (Ci-C 4 ) -Alkoxy-carbonyl- (Ci-C 4 ) -alkyl, hydroxycarbonyl, hydroxycarbonyl- (Ci-C 4 ) -alkyl, C (0) NR 6 R 7 , and wherein the structural elements cycloalkyl or heterocyclyl have n oxo groups, where n = 0, 1 or 2 are substituted.
Ganz besonders bevorzugter Erfindungsgegenstand sind Verbindungen der allgemeinen Formel (I), worin Very particularly preferred subject matter of the invention are compounds of the general formula (I), in which
Q für die Gruppe Q for the group
steht stands
X und Y unabhängig voneinander für C-H oder die Gruppierung C-R1 stehen, wobei X and Y independently of one another represent CH or the grouping CR 1 , where
X für C-H steht, wenn Y für die Gruppierung C-R1 steht und X stands for CH if Y stands for the grouping CR 1 and
X für die Gruppierung C-R1 steht, wenn Y für C-H steht, X stands for the grouping CR 1 , if Y stands for CH,
R1 für Fluor, Chlor, Brom, Iod, Cyano, Methyl, Ethyl, n-Propyl, 1 -Methylethyl, n-Butyl, 1 -R 1 for fluorine, chlorine, bromine, iodine, cyano, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1 -
Methylpropyl, 2-Methylpropyl, l,l-Dimethylethyl, n-Pentyl, 1 -Methylbutyl, 2-Methylbutyl, 3- Methylbutyl, 1 , 1 -Dimethylpropyl, 1 ,2-Dimethylpropyl, 2,2-Dimethylpropyl, 1 -Ethylpropyl, n- Hexyl, 1 -Methylpentyl, 2-Methylpentyl, 3-Methylpentyl, 4-Methylpentyl, l,l-Dimethylbutyl,Methylpropyl, 2-methylpropyl, l, l-dimethylethyl, n-pentyl, 1-methylbutyl, 2-methylbutyl, 3- Methylbutyl, 1, 1-dimethylpropyl, 1, 2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, n-hexyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, l, l-dimethylbutyl,
1.2-Dimethylbutyl, l,3-Di-methylbutyl, 2,2-Dimethylbutyl, 2,3-Dimethylbutyl, 3,3- Dimethylbutyl, 1 -Ethylbutyl, 2-Ethylbutyl, 1 , 1 ,2-Trimethylpropyl, 1 ,2,2-Trimethylpropyl, 1 - Ethyl-l-methylpropyl, 1 -Ethyl-2-methylpropyl, Cyclopropyl, Cyclobutyl, Cyclopentyl, Cyclohexyl, Spiro[2.2]pent-l-yl, Spiro[2.3]hex-l-yl, Spiro[2.3]hex-4-yl, 3-Spiro[2.3]hex-5-yl, Spiro[3.3]hept-l-yl, Spiro[3.3]hept-2-yl, Bicyclo[l.l.0]butan-l-yl, Bicyclo[l.l.0]butan-2-yl, Bicyclo[2.1.0]pentan-l -yl, Bicyclo[l .1.1 ]pentan-l -yl, Bicyclo[2.1 0]pentan-2-yl, 1,2-dimethylbutyl, l, 3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2-ethylbutyl, 1, 1, 2-trimethylpropyl, 1, 2, 2-trimethylpropyl, 1 - ethyl-l-methylpropyl, 1 -ethyl-2-methylpropyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, spiro [2.2] pent-l-yl, spiro [2.3] hex-l-yl, spiro [ 2.3] hex-4-yl, 3-spiro [2.3] hex-5-yl, spiro [3.3] hept-l-yl, spiro [3.3] hept-2-yl, bicyclo [ll0] butan-l-yl , Bicyclo [ll0] butan-2-yl, bicyclo [2.1.0] pentan-l -yl, bicyclo [l .1.1] pentan-l -yl, bicyclo [2.1 0] pentan-2-yl,
Bicyclo[2.l.0]pentan-5-yl, Bicyclo[2.l.l]hexyl, Bicyclo[2.2.l]hept-2-yl, Bicyclo[2.2.2]octan-2- yl, Bicyclo[3.2.l]octan-2-yl, Bicyclo[3.2.2]nonan-2-yl, Adamantan-l-yl, Adamantan-2-yl, 1- Methylcyclopropyl, 2-Methylcyclopropyl, 2,2-Dimethylcyclopropyl, 2,3-Dimethylcyclopropyl, l,l'-Bi(cyclopropyl)-l-yl, 1 , l'-Bi(cyclopropyl)-2-yl, 2'-Methyl-l,l'-bi(cyclopropyl)-2-yl, 1- Cyanocyclopropyl, 2-Cyanocyclopropyl, 1 -Methylcyclobutyl, 2-Methylcyclobutyl, 3- Methylcyclobutyl, 3,3-Dimethylcyclobut-l-yl, 1 -Cyanocyclobutyl, 2-Cyanocyclobutyl, 3- Cyanocyclobutyl, 3,3-Difluorcyclobut-l-yl, 3-Fluorcyclobut-l-yl, 2,2-Difluorcycloprop-l-yl, 1- Fluorcycloprop-l-yl, 2-Fluorcycloprop-l-yl, 1 - Allylcyclopropyl, 1 -Vinylcyclobutyl, 1- Vinylcyclopropyl, 1 -Ethylcyclopropyl, 1 -Methylcyclohexyl, 2-Methylcyclohexyl, 3- Methylcyclohexyl, 1 -Methoxycyclohexyl, 2-Methoxycyclohexyl, 3-Methoxycyclohexyl, Trifluormethyl, Pentafluorethyl, l,l,2,2-Tetrafluorethyl, Heptafluorpropyl, Nonafluorbutyl, Chlordifluormethyl, Bromdifluormethyl, Dichlorfluormethyl, Ioddifluormethyl, Bicyclo [2.l.0] pentan-5-yl, bicyclo [2.ll] hexyl, bicyclo [2.2.l] hept-2-yl, bicyclo [2.2.2] octan-2-yl, bicyclo [3.2. l] octan-2-yl, bicyclo [3.2.2] nonan-2-yl, adamantan-l-yl, adamantan-2-yl, 1-methylcyclopropyl, 2-methylcyclopropyl, 2,2-dimethylcyclopropyl, 2,3- Dimethylcyclopropyl, l, l'-Bi (cyclopropyl) -l-yl, 1, l'-Bi (cyclopropyl) -2-yl, 2'-methyl-l, l'-bi (cyclopropyl) -2-yl, 1 - Cyanocyclopropyl, 2-cyanocyclopropyl, 1-methylcyclobutyl, 2-methylcyclobutyl, 3-methylcyclobutyl, 3,3-dimethylcyclobut-l-yl, 1-cyanocyclobutyl, 2-cyanocyclobutyl, 3-cyanocyclobutyl, 3,3-difluorocyclobut-l-yl , 3-fluorocyclobut-l-yl, 2,2-difluorocycloprop-l-yl, 1-fluorocycloprop-l-yl, 2-fluorocycloprop-l-yl, 1 - allylcyclopropyl, 1-vinylcyclobutyl, 1-vinylcyclopropyl, 1-ethylcyclopropyl , 1-methylcyclohexyl, 2-methylcyclohexyl, 3-methylcyclohexyl, 1-methoxycyclohexyl, 2-methoxycyclohexyl, 3-methoxycyclohexyl, trifluoromethyl, pentafluoroethyl, l, l, 2,2-tetrafluoroethyl, heptafluoropropyl, nonafluorobutyl, chlorodifluoromethyl l, bromodifluoromethyl, dichlorofluoromethyl, iododifluoromethyl,
Bromfluormethyl, l-Fluorethyl, 2-Fluorethyl, Fluormethyl, Difluormethyl, 2,2-Difluorethyl,Bromofluoromethyl, l-fluoroethyl, 2-fluoroethyl, fluoromethyl, difluoromethyl, 2,2-difluoroethyl,
2.2.2-Trifluorethyl, Difluor-tert.-butyl, Chlormethyl, Brommethyl, Hydroxymethyl, 2.2.2-trifluoroethyl, difluoro-tert-butyl, chloromethyl, bromomethyl, hydroxymethyl,
Hydroxyethyl, Hydroxy-n-propyl, Methoxy, Ethoxy, n-Propyloxy, iso-Propyloxy, n-Butyloxy, tert-Butyloxy, Methoxymethyl, Ethoxymethyl, n-Propyloxymethyl, iso-Propyloxymethyl, Methoxyethyl, Ethoxyethyl, n-Propyloxyethyl, iso-Propyloxyethyl, Trifluormethoxy, Hydroxyethyl, hydroxy-n-propyl, methoxy, ethoxy, n-propyloxy, iso-propyloxy, n-butyloxy, tert-butyloxy, methoxymethyl, ethoxymethyl, n-propyloxymethyl, iso-propyloxymethyl, methoxyethyl, ethoxyethyl, n-propyloxyethyl, iso- Propyloxyethyl, trifluoromethoxy,
Difluormethoxy, Pentafluorethoxy, 2,2,l,l-Tetrafluorethoxy, 2,2,2-Trifluorethoxy, 2,2- Difluorethoxy, Methylthio, Ethylthio, n-Propylthio, iso-Propylthio, Trifluormethylthio, Pentafluorethylthio, Phenyl, 2-Fluor-Phenyl, 3 -Fluor-Phenyl, 4-Fluor-Phenyl, 2,4-Difluor- Phenyl, 2,5-Difluor-Phenyl, 2,6-Difluor-Phenyl, 2,3-Difluor-Phenyl, 3,4-Difluor-Phenyl, 3,5- Difluor-Phenyl, 2,4,5-Trifluor-Phenyl, 3,4,5-Trifluor-Phenyl, 2-Chlor-Phenyl, 3 -Chlor-Phenyl, 4-Chlor-Phenyl, 2,4-Dichlor-Phenyl, 2,5-Dichlor-Phenyl, 2,6-Dichlor-Phenyl, 2,3-Dichlor- Phenyl, 3,4-Dichlor-Phenyl, 3,5-Dichlor-Phenyl, 2,4,5-Trichlor-Phenyl, 3,4,5-Trichlor-Phenyl, 2,4,6-Trichlor-Phenyl, 2-Brom-Phenyl, 3 -Brom-Phenyl, 4-Brom-Phenyl, 2-Iod-Phenyl, 3-Iod- Phenyl, 4-Iod-Phenyl, 2-Brom-4-Fluor-Phenyl, 2-Brom-4-Chlor-Phenyl, 3-Brom-4-Fluor- Phenyl, 3-Brom-4-Chlor-Phenyl, 3-Brom-5-Fluor-Phenyl, 3 -Brom-5 -Chlor-Phenyl, 2-Fluor-4- Brom-Phenyl, 2-Chlor-4-Brom-Phenyl, 3-Fluor-4-Brom-Phenyl, 3-Chlor-4-Brom-Phenyl, 2- Chlor-4-Fluor-Phenyl, 3-Chlor-4-Fluor-Phenyl, 2-Fluor-3 -Chlor-Phenyl, 2-Fluor-4-Chlor- Phenyl, 2-Fluor-5-Chlor-Phenyl, 3-Fluor-4-Chlor-Phenyl, 3-Fluor-5-Chlor-Phenyl, 2-Fluor-6- Chlor-Phenyl, 2-Methyl-Phenyl, 3 -Methyl-Phenyl, 4-Methyl-Phenyl, 2,4-Dimethyl-Phenyl, 2,5- Dimethyl-Phenyl, 2,6-Dimethyl-Phenyl, 2,3-Dimethyl-Phenyl, 3,4-Dimethyl-Phenyl, 3,5- Dimethyl-Phenyl, 2,4,5-Trimethyl-Phenyl, 3,4,5-Trimethyl-Phenyl, 2,4,6-Trimethyl-Phenyl, 2- Methoxy-Phenyl, 3-Methoxy-Phenyl, 4-Methoxy-Phenyl, 2,4-Dimethoxy-Phenyl, 2,5- Dimethoxy-Phenyl, 2,6-Dimethoxy-Phenyl, 2,3-Dimethoxy-Phenyl, 3,4-Dimethoxy-Phenyl, 3,5- Dimethoxy-Phenyl, 2,4,5-Trimethoxy-Phenyl, 3,4,5-Trimethoxy-Phenyl, 2,4,6-Trimethoxy- Phenyl, 2-Trifluormethoxy-Phenyl, 3 -Trifluormethoxy- Phenyl, 4-Trifluormethoxy-Phenyl, 2- Difluormethoxy-Phenyl, 3-Difluormethoxy-Phenyl, 4-Difluormethoxy-Phenyl, 2- Trifluormethyl-Phenyl, 3-Trifluormethyl-Phenyl, 4-Trifluormethyl-Phenyl, 2-Difluormethyl- Phenyl, 3-Difluormethyl-Phenyl, 4-Difluormethyl-Phenyl, 3,5-Bis(Trifluormethyl)-Phenyl, 3- Trifluormethyl-5-Fluor-Phenyl, 3-Trifluormethyl-5-Chlor-Phenyl, 3 -Methyl-5 -Fluor-Phenyl, 3- Methyl-5-Chlor-Phenyl, 3-Methoxy-5-Fluor-Phenyl, 3-Methoxy-5-Chlor-Phenyl, 3- Trifluormethoxy-5-Chlor-Phenyl, 2-Ethoxy-Phenyl, 3-Ethoxy-Phenyl, 4-Ethoxy-Phenyl, 2- Methylthio-Phenyl, 3-Methylthio-Phenyl, 4-Methylthio-Phenyl, 2-Trifluormethylthio-Phenyl, 3- Trifluormethylthio-Phenyl, 4-Trifluormethylthio-Phenyl, Phenyloxy, p-Cl-Phenyloxy, Difluoromethoxy, pentafluoroethoxy, 2,2, l, l-tetrafluoroethoxy, 2,2,2-trifluoroethoxy, 2,2-difluoroethoxy, methylthio, ethylthio, n-propylthio, iso-propylthio, trifluoromethylthio, pentafluoroethylthio, phenyl, 2-fluoro Phenyl, 3-fluorophenyl, 4-fluorophenyl, 2,4-difluorophenyl, 2,5-difluorophenyl, 2,6-difluorophenyl, 2,3-difluorophenyl, 3,4- Difluorophenyl, 3,5-difluorophenyl, 2,4,5-trifluorophenyl, 3,4,5-trifluorophenyl, 2-chlorophenyl, 3-chlorophenyl, 4-chlorophenyl, 2,4-dichlorophenyl, 2,5-dichlorophenyl, 2,6-dichlorophenyl, 2,3-dichlorophenyl, 3,4-dichlorophenyl, 3,5-dichlorophenyl, 2, 4,5-trichlorophenyl, 3,4,5-trichlorophenyl, 2,4,6-trichlorophenyl, 2-bromophenyl, 3-bromophenyl, 4-bromophenyl, 2-iodine Phenyl, 3-iodophenyl, 4-iodophenyl, 2-bromo-4-fluoro-phenyl, 2-bromo-4-chlorophenyl, 3-bromo-4-fluoro-phenyl, 3-bromo-4- Chloro-phenyl, 3-bromo-5-fluoro-phenyl, 3-bromo-5-chloro-phenyl, 2-fluoro-4-bromo-phenyl, 2-chloro-4-bromo-phenyl, 3-fluoro-4- Bromophenyl, 3-chloro-4-bromophenyl, 2-chloro-4-fluoro- Phenyl, 3-chloro-4-fluorophenyl, 2-fluoro-3-chlorophenyl, 2-fluoro-4-chloro Phenyl, 2-fluoro-5-chlorophenyl, 3-fluoro-4-chlorophenyl, 3-fluoro-5-chlorophenyl, 2-fluoro-6-chlorophenyl, 2-methylphenyl, 3 - Methyl-phenyl, 4-methyl-phenyl, 2,4-dimethyl-phenyl, 2,5-dimethyl-phenyl, 2,6-dimethyl-phenyl, 2,3-dimethyl-phenyl, 3,4-dimethyl-phenyl, 3,5-dimethyl-phenyl, 2,4,5-trimethyl-phenyl, 3,4,5-trimethyl-phenyl, 2,4,6-trimethyl-phenyl, 2-methoxy-phenyl, 3-methoxy-phenyl, 4-methoxy-phenyl, 2,4-dimethoxy-phenyl, 2,5-dimethoxy-phenyl, 2,6-dimethoxy-phenyl, 2,3-dimethoxy-phenyl, 3,4-dimethoxy-phenyl, 3,5- Dimethoxy-phenyl, 2,4,5-trimethoxy-phenyl, 3,4,5-trimethoxy-phenyl, 2,4,6-trimethoxy-phenyl, 2-trifluoromethoxy-phenyl, 3-trifluoromethoxy-phenyl, 4-trifluoromethoxy- Phenyl, 2-difluoromethoxy-phenyl, 3-difluoromethoxy-phenyl, 4-difluoromethoxy-phenyl, 2-trifluoromethyl-phenyl, 3-trifluoromethyl-phenyl, 4-trifluoromethyl-phenyl, 2-difluoromethyl-phenyl, 3-difluoromethyl-phenyl, 4-difluoromethylphenyl, 3,5-bis (trifluoromethyl) phenyl, 3-trifluoromethyl-5-fluorophenyl, 3-trifl uormethyl-5-chlorophenyl, 3-methyl-5-fluorophenyl, 3-methyl-5-chlorophenyl, 3-methoxy-5-fluorophenyl, 3-methoxy-5-chlorophenyl, 3- Trifluoromethoxy-5-chlorophenyl, 2-ethoxy-phenyl, 3-ethoxy-phenyl, 4-ethoxy-phenyl, 2-methylthio-phenyl, 3-methylthio-phenyl, 4-methylthio-phenyl, 2-trifluoromethylthio-phenyl, 3- trifluoromethylthio-phenyl, 4-trifluoromethylthio-phenyl, phenyloxy, p-Cl-phenyloxy,
Thiophen-2-yl, Thiophen-3-yl, Pyridin-2-yl, Pyridin-3-yl, Pyridin-4-yl, Furan-2-yl, Furan-3-yl, Tetrahydrofuran-2-yl, Cyclopropylmethyl, Cyclobutylmethyl, Cyclopentylmethyl, Thiophene-2-yl, thiophene-3-yl, pyridin-2-yl, pyridin-3-yl, pyridin-4-yl, furan-2-yl, furan-3-yl, tetrahydrofuran-2-yl, cyclopropylmethyl, Cyclobutylmethyl, cyclopentylmethyl,
Cyclohexylmethyl, Methylcarbonyl, Ethylcarbonyl, n-Propylcarbonyl, iso-Propylcarbonyl, n- Butylcarbonyl, tert-Butylcarbonyl, Ethenyl, l-Propenyl, 2-Propenyl, 1 -Methyl- ethenyl, 1- Butenyl, 2-Butenyl, 3-Butenyl, 1 -Methyl- l-propenyl, 2-Methyl- l-propenyl, l-Methyl-2- propenyl, 2-Methyl-2-propenyl, l-Pentenyl, 2-Pentenyl, 3-Pentenyl, 4-Pentenyl, 1 -Methyl- 1- butenyl, 2-Methyl- l-butenyl, 3 -Methyl- l-butenyl, 1 -Methyl-2-butenyl, 2-Methyl-2-butenyl, 3- Methyl-2-butenyl, l-Methyl-3-butenyl, 2-Methyl-3-butenyl, 3-Methyl-3-butenyl, 1 , 1 -Dimethyl- 2-propenyl, 1 ,2-Dimethyl- l-propenyl, 1 ,2-Dimethyl-2-propenyl, l-Ethyl- l-propenyl, l-Ethyl-2- propenyl, l-Hexenyl, 2-Hexenyl, 3-Hexenyl, 4-Hexenyl, 5-Hexenyl, 1 -Methyl- l-pentenyl, 2- Methyl- l-pentenyl, 3 -Methyl- l-pentenyl, 4-Methyl- l-pentenyl, 1 -Methyl-2-pentenyl, 2-Methyl- 2-pentenyl, 3-Methyl-2-pentenyl, 4-Methyl-2-pentenyl, l-Methyl-3-pentenyl, 2-Methyl-3- pentenyl, 3-Methyl-3-pentenyl, 4-Methyl-3-pentenyl, 1 -Methyl-4-pentenyl, 2-Methyl-4- pentenyl, 3-Methyl-4-pentenyl, 4-Methyl-4-pentenyl, 1 , 1 -Dimethyl-2-butenyl, l,l-Dimethyl-3- butenyl, 1 ,2-Dimethyl- l-butenyl, 1 ,2-Dimethyl-2-butenyl, l,2-Dimethyl-3-butenyl, 1,3- Dimethyl- l-butenyl, l,3-Dimethyl-2-butenyl, l,3-Dimethyl-3-butenyl, 2,2-Dimethyl-3-butenyl, 2,3-Dimethyl-l-butenyl, 2,3-Dimethyl-2-butenyl, 2,3-Dimethyl-3-butenyl, 3,3-Dimethyl-l- butenyl, 3,3-Dimethyl-2-butenyl, l-Ethyl- l-butenyl, 1 -Ethyl-2-butenyl, l-Ethyl-3-butenyl, 2- Ethyl- l-butenyl, 2-Ethyl-2-butenyl, 2-Ethyl-3-butenyl, l,l,2-Trimethyl-2-propenyl, l-Ethyl-l- methyl-2-propenyl, 1 -Ethyl-2-methyl- l-propenyl und 1 -Ethyl-2-methyl-2-propenyl, Ethinyl, 1- Propinyl, 2-Propinyl, l-Butinyl, 2-Butinyl, 3-Butinyl, l-Methyl-2-propinyl, l-Pentinyl, 2- Pentinyl, 3-Pentinyl, 4-Pentinyl, 1 -Methyl-2-butinyl, l-Methyl-3-butinyl, 2-Methyl-3-butinyl, 3- Methyl-l-butinyl, l,l-Dimethyl-2-propinyl, 1 -Ethyl-2-propinyl, l-Hexinyl, 2-Hexinyl, 3- Hexinyl, 4-Hexinyl, 5-Hexinyl, 1 -Methyl-2-pentinyl, l-Methyl-3 -pentinyl, 1 -Methyl-4-pentinyl,Cyclohexylmethyl, methylcarbonyl, ethylcarbonyl, n-propylcarbonyl, iso-propylcarbonyl, n-butylcarbonyl, tert-butylcarbonyl, ethenyl, l-propenyl, 2-propenyl, 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-l-propenyl, 2-methyl-l-propenyl, l-methyl-2-propenyl, 2-methyl-2-propenyl, l-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1 - Methyl 1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl, 1-methyl- 3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1, 1-dimethyl-2-propenyl, 1, 2-dimethyl-1-propenyl, 1, 2-dimethyl-2-propenyl, l-ethyl-l-propenyl, l-ethyl-2-propenyl, l-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 1-methyl-l-pentenyl, 2-methyl-l- pentenyl, 3-methyl-1-pentenyl, 4-methyl-1-pentenyl, 1-methyl-2-pentenyl, 2-methyl-2-pentenyl, 3-methyl-2-pentenyl, 4-methyl-2-pentenyl, l-methyl-3-pentenyl, 2-methyl-3-pentenyl, 3-methyl-3-pentenyl, 4-methyl-3-pentenyl, 1-methyl-4-pentenyl, 2 -Methyl-4-pentenyl, 3-methyl-4-pentenyl, 4-methyl-4-pentenyl, 1, 1-dimethyl-2-butenyl, l, l-dimethyl-3-butenyl, 1, 2-dimethyl-l -butenyl, 1,2-dimethyl-2-butenyl, 1,2-dimethyl-3-butenyl, 1,3-dimethyl-1-butenyl, 1,3-dimethyl-2-butenyl, 1,3-dimethyl-3 -butenyl, 2,2-dimethyl-3-butenyl, 2,3-dimethyl-l-butenyl, 2,3-dimethyl-2-butenyl, 2,3-dimethyl-3-butenyl, 3,3-dimethyl-l - Butenyl, 3,3-dimethyl-2-butenyl, 1-ethyl-1-butenyl, 1-ethyl-2-butenyl, 1-ethyl-3-butenyl, 2-ethyl-1-butenyl, 2-ethyl-2 -butenyl, 2-ethyl-3-butenyl, l, l, 2-trimethyl-2-propenyl, l-ethyl-l-methyl-2-propenyl, 1-ethyl-2-methyl-l-propenyl and 1-ethyl -2-methyl-2-propenyl, ethynyl, 1-propynyl, 2-propynyl, l-butynyl, 2-butynyl, 3-butynyl, l-methyl-2-propynyl, l-pentynyl, 2- Pentynyl, 3-pentynyl, 4-pentynyl, 1-methyl-2-butynyl, l-methyl-3-butynyl, 2-methyl-3-butynyl, 3-methyl-l-butynyl, l, l-dimethyl-2- propynyl, 1-ethyl-2-propynyl, l-hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, 5-hexynyl, 1-methyl-2-pentynyl, l-methyl-3-pentynyl, 1-methyl- 4-pentynyl,
2-Methyl-3 -pentinyl, 2-Methyl-4-pentinyl, 3 -Methyl- 1 -pentinyl, 3-Methyl-4-pentinyl, 4-Methyl-2-methyl-3-pentynyl, 2-methyl-4-pentynyl, 3-methyl-1-pentynyl, 3-methyl-4-pentynyl, 4-methyl-
1 -pentinyl, 4-Methyl-2-pentinyl, 1 , 1 -Dimethyl-2-butinyl, l,l-Dimethyl-3-butinyl, 1 ,2-Dimethyl-1-pentynyl, 4-methyl-2-pentynyl, 1, 1-dimethyl-2-butynyl, l, l-dimethyl-3-butynyl, 1, 2-dimethyl-
3-butinyl, 2,2-Dimethyl-3-butinyl, 3,3-Dimethyl-l-butinyl, 1 -Ethyl-2-butinyl, l-Ethyl-3-butinyl,3-butynyl, 2,2-dimethyl-3-butynyl, 3,3-dimethyl-l-butynyl, 1-ethyl-2-butynyl, l-ethyl-3-butynyl,
2-Ethyl-3-butinyl, l-Ethyl-l-methyl-2-propinyl steht, 2-ethyl-3-butynyl, l-ethyl-l-methyl-2-propynyl,
A1 und A2 unabhängig voneinander für C-H oder die Gruppierung C-R2 stehen, wobei A 1 and A 2 independently of one another represent CH or the grouping CR 2 , where
A1 für C-H steht, wenn A2 für die Gruppierung C-R2 steht und A 1 stands for CH if A 2 stands for the grouping CR 2 and
A1 für die Gruppierung C-R2 steht, wenn A2 für C-H steht, A 1 stands for the grouping CR 2 , if A 2 stands for CH,
R2 für Fluor, Chlor, Brom, lod, Hydroxy, Hydrothio, Methyl, Ethyl, n-Propyl, 1 -Methylethyl, n- Butyl, 1 -Methylpropyl, 2-Methylpropyl, 1,1-Dimethylethyl, n-Pentyl, 1 -Methylbutyl, 2- Methylbutyl, 3 -Methylbutyl, 1,1-Dimethylpropyl, 1 ,2-Dimethylpropyl, 2,2-Dimethylpropyl, 1- Ethylpropyl, n-Hexyl, 1 -Methylpentyl, 2-Methylpentyl, 3-Methylpentyl, 4-Methylpentyl, 1,1- Dimethylbutyl, 1 ,2-Dimethylbutyl, 1,3-Di-methylbutyl, 2,2-Dimethylbutyl, 2,3-Dimethylbutyl, 3,3-Dimethylbutyl, 1 -Ethylbutyl, 2-Ethylbutyl, 1,1,2-Trimethylpropyl, 1 ,2,2-Trimethylpropyl,R 2 for fluorine, chlorine, bromine, iodine, hydroxy, hydrothio, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl, 1,1-dimethylethyl, n-pentyl, 1 -Methylbutyl, 2-methylbutyl, 3 -methylbutyl, 1,1-dimethylpropyl, 1, 2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, n-hexyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4 -Methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2-ethylbutyl, 1st , 1,2-trimethylpropyl, 1, 2,2-trimethylpropyl,
1 -Ethyl- 1 -methylpropyl, l-Ethyl-2-methylpropyl, Trifluormethyl, Pentafluorethyl, 1, 1,2,2- Tetrafluorethyl, Heptafluor-n-propyl, Heptafluor-iso-propyl, Nonafluorbutyl, 1-ethyl-1-methylpropyl, l-ethyl-2-methylpropyl, trifluoromethyl, pentafluoroethyl, 1, 1,2,2-tetrafluoroethyl, heptafluoro-n-propyl, heptafluoroisopropyl, nonafluorobutyl,
Chlordifluormethyl, Bromdifluormethyl, Dichlorfluormethyl, loddifluormethyl, Chlorodifluoromethyl, bromodifluoromethyl, dichlorofluoromethyl, iododifluoromethyl,
Bromfluormethyl, 1-Fluorethyl, 2-Fluorethyl, Fluormethyl, Difluormethyl, 2,2-Difluorethyl, 2,2,2-Trifluorethyl, Difluor-tert.-butyl, Chlormethyl, Brommethyl, Fluormethyl, 3,3,3-Trifluor- n-propyl, 1-Fluorprop-l-yl, 1-Trifluormethylprop-l-yl, 2-Trifluormethylprop-2-yl, 1-Fluorprop- 1 -yl, 2-Fluorprop-2-yl, 2-Chlorprop-2-yl, Cyclopropyl, Cyclobutyl, Cyclopentyl, Cyclohexyl, 1 - Methylcycloprop-l-yl, 2-Methylcycloprop-l-yl, 2,2-Dimethylcycloprop-l-yl, 2,3- Dimethylcyclopropyl, 1-Cyanocycloprop-l-yl, 2-Cyanocycloprop-l-yl, 1 -Methylcyclobutyl, 2- Methylcyclobutyl, 3 -Methylcyclobutyl, 3,3-Dimethylcyclobutyl, 1 -Cyanocyclobutyl, 2- Cyanocyclobutyl, 3 -Cyanocyclobutyl, 1 -Allylcyclopropyl, 1 -Vinylcyclobutyl, 1- Vinylcyclopropyl, 1 -Ethylcyclopropyl, 1 -Methylcyclohexyl, 2-Methylcyclohexyl, 3- Methylcyclohexyl, 1 -Methoxycyclohexyl, 2-Methoxycyclohexyl, 3-Methoxycyclohexyl, Spiro[2.2]pent-l-yl, Spiro[2.3]hex-l-yl, Spiro[2.3]hex-4-yl, 3-Spiro[2.3]hex-5-yl, Bromofluoromethyl, 1-fluoroethyl, 2-fluoroethyl, fluoromethyl, difluoromethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, difluoro-tert-butyl, chloromethyl, bromomethyl, fluoromethyl, 3,3,3-trifluoro- n -propyl, 1-fluoroprop-l-yl, 1-trifluoromethylprop-l-yl, 2-trifluoromethylprop-2-yl, 1-fluoroprop-1-yl, 2-fluoroprop-2-yl, 2-chloroprop-2-yl , Cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, 1 - methylcycloprop-l-yl, 2-methylcycloprop-l-yl, 2,2-dimethylcycloprop-l-yl, 2,3-dimethylcyclopropyl, 1-cyanocycloprop-l-yl, 2 -Cyanocycloprop-l-yl, 1-methylcyclobutyl, 2-methylcyclobutyl, 3-methylcyclobutyl, 3,3-dimethylcyclobutyl, 1-cyanocyclobutyl, 2-cyanocyclobutyl, 3-cyanocyclobutyl, 1-allyl cyclopropyl, 1-vinyl cyclobutyl, 1-vinylcyclopropyl, 1 -Ethylcyclopropyl, 1-methylcyclohexyl, 2-methylcyclohexyl, 3-methylcyclohexyl, 1-methoxycyclohexyl, 2-methoxycyclohexyl, 3-methoxycyclohexyl, spiro [2.2] pent-l-yl, spiro [2.3] hex-l-yl, spiro [2.3 ] hex-4-yl, 3-spiro [2.3] hex-5-yl,
Spiro[3.3]hept-l-yl, Spiro[3.3]hept-2-yl, Bicyclo[1.1.0]butan-l-yl, Bicyclo[1.1.0]butan-2-yl, Bicyclo[2.1.0]pentan-l -yl, Bicyclo[l .1.1 ]pentan-l -yl, Bicyclo[2.1 0]pentan-2-yl, Spiro [3.3] hept-l-yl, spiro [3.3] hept-2-yl, bicyclo [1.1.0] butan-l-yl, bicyclo [1.1.0] butan-2-yl, bicyclo [2.1.0] pentan-1-yl, bicyclo [1.1.1] pentan-1-yl, bicyclo [2.1 0] pentan-2-yl,
Bicyclo[2.1.0]pentan-5-yl, Bicyclo[2.1.1]hexyl, Bicyclo[2.2.1]hept-2-yl, Bicyclo[2.2.2]octan-2- yl, Bicyclo[3.2.1]octan-2-yl, Bicyclo[3.2.2]nonan-2-yl, Adamantan-l-yl, Adamantan-2-yl, Cyclopropylmethyl, Cyclobutylmethyl, Cyclopentylmethyl, Cyclohexylmethyl, Phenyl, 2-Fluor- Phenyl, 3 -Fluor-Phenyl, 4-Fluor-Phenyl, 2,4-Difluor-Phenyl, 2,5-Difluor-Phenyl, 2,6-Difluor- Phenyl, 2,3-Difluor-Phenyl, 3,4-Difluor-Phenyl, 3,5-Difluor-Phenyl, 2,4,5-Trifluor-Phenyl,Bicyclo [2.1.0] pentan-5-yl, bicyclo [2.1.1] hexyl, bicyclo [2.2.1] hept-2-yl, bicyclo [2.2.2] octan-2-yl, bicyclo [3.2.1] octan-2-yl, bicyclo [3.2.2] nonan-2-yl, adamantan-l-yl, adamantan-2-yl, cyclopropylmethyl, cyclobutylmethyl, cyclopentylmethyl, cyclohexylmethyl, phenyl, 2-fluoro Phenyl, 3-fluorophenyl, 4-fluorophenyl, 2,4-difluorophenyl, 2,5-difluorophenyl, 2,6-difluorophenyl, 2,3-difluorophenyl, 3,4- Difluorophenyl, 3,5-difluorophenyl, 2,4,5-trifluorophenyl,
3.4.5-Trifluor-Phenyl, 2-Chlor-Phenyl, 3 -Chlor-Phenyl, 4-Chlor-Phenyl, 2,4-Dichlor-Phenyl,3.4.5-trifluorophenyl, 2-chlorophenyl, 3-chlorophenyl, 4-chlorophenyl, 2,4-dichlorophenyl,
2.5-Dichlor-Phenyl, 2,6-Dichlor-Phenyl, 2,3-Dichlor-Phenyl, 3,4-Dichlor-Phenyl, 3,5-Dichlor- Phenyl, 2,4,5-Trichlor-Phenyl, 3,4,5-Trichlor-Phenyl, 2,4,6-Trichlor-Phenyl, 2-Brom-Phenyl, 3- Brom-Phenyl, 4-Brom-Phenyl, 2-Iod-Phenyl, 3-Iod-Phenyl, 4-Iod-Phenyl, 2-Brom-4-Fluor- Phenyl, 2-Brom-4-Chlor-Phenyl, 3-Brom-4-Fluor-Phenyl, 3-Brom-4-Chlor-Phenyl, 3-Brom-5- Fluor-Phenyl, 3-Brom-5-Chlor-Phenyl, 2-Fluor-4-Brom-Phenyl, 2-Chlor-4-Brom-Phenyl, 3- Fluor-4-Brom-Phenyl, 3-Chlor-4-Brom-Phenyl, 2-Chlor-4-Fluor-Phenyl, 3-Chlor-4-Fluor- Phenyl, 2-Fluor-3 -Chlor-Phenyl, 2-Fluor-4-Chlor-Phenyl, 2-Fluor-5-Chlor-Phenyl, 3-Fluor-4- Chlor-Phenyl, 3-Fluor-5-Chlor-Phenyl, 2-Fluor-6-Chlor-Phenyl, 2-Methyl-Phenyl, 3-Methyl- Phenyl, 4-Methyl-Phenyl, 2,4-Dimethyl-Phenyl, 2,5-Dimethyl-Phenyl, 2,6-Dimethyl-Phenyl, 2,3-Dimethyl-Phenyl, 3,4-Dimethyl-Phenyl, 3,5-Dimethyl-Phenyl, 2,4,5-Trimethyl-Phenyl,2,5-dichlorophenyl, 2,6-dichlorophenyl, 2,3-dichlorophenyl, 3,4-dichlorophenyl, 3,5-dichlorophenyl, 2,4,5-trichlorophenyl, 3, 4,5-trichlorophenyl, 2,4,6-trichlorophenyl, 2-bromophenyl, 3-bromophenyl, 4-bromophenyl, 2-iodophenyl, 3-iodophenyl, 4- Iodo-phenyl, 2-bromo-4-fluoro-phenyl, 2-bromo-4-chloro-phenyl, 3-bromo-4-fluoro-phenyl, 3-bromo-4-chloro-phenyl, 3-bromo-5- Fluoro-phenyl, 3-bromo-5-chloro-phenyl, 2-fluoro-4-bromo-phenyl, 2-chloro-4-bromo-phenyl, 3-fluoro-4-bromo-phenyl, 3-chloro-4- Bromophenyl, 2-chloro-4-fluorophenyl, 3-chloro-4-fluorophenyl, 2-fluoro-3-chlorophenyl, 2-fluoro-4-chlorophenyl, 2-fluoro-5- Chloro-phenyl, 3-fluoro-4-chloro-phenyl, 3-fluoro-5-chloro-phenyl, 2-fluoro-6-chloro-phenyl, 2-methyl-phenyl, 3-methyl-phenyl, 4-methyl Phenyl, 2,4-dimethyl-phenyl, 2,5-dimethyl-phenyl, 2,6-dimethyl-phenyl, 2,3-dimethyl-phenyl, 3,4-dimethyl-phenyl, 3,5-dimethyl-phenyl, 2,4,5-trimethyl-phenyl,
3.4.5-Trimethyl-Phenyl, 2,4,6-Trimethyl-Phenyl, 2-Methoxy-Phenyl, 3-Methoxy-Phenyl, 4- Methoxy-Phenyl, 2,4-Dimethoxy-Phenyl, 2,5-Dimethoxy-Phenyl, 2,6-Dimethoxy-Phenyl, 2,3- Dimethoxy-Phenyl, 3,4-Dimethoxy-Phenyl, 3,5-Dimethoxy-Phenyl, 2,4,5-Trimethoxy-Phenyl,3.4.5-trimethyl-phenyl, 2,4,6-trimethyl-phenyl, 2-methoxy-phenyl, 3-methoxy-phenyl, 4-methoxy-phenyl, 2,4-dimethoxy-phenyl, 2,5-dimethoxy- Phenyl, 2,6-dimethoxy-phenyl, 2,3-dimethoxy-phenyl, 3,4-dimethoxy-phenyl, 3,5-dimethoxy-phenyl, 2,4,5-trimethoxy-phenyl,
3.4.5-Trimethoxy-Phenyl, 2,4,6-Trimethoxy-Phenyl, 2-Trifluormethoxy-Phenyl, 3- Trifluormethoxy-Phenyl, 4-Trifluormethoxy-Phenyl, 2-Difluormethoxy-Phenyl, 3- Difluormethoxy-Phenyl, 4-Difluormethoxy-Phenyl, 2-Trifluormethyl-Phenyl, 3-Trifluormethyl- Phenyl, 4-Trifluormethyl-Phenyl, 2-Difluormethyl-Phenyl, 3-Difluormethyl-Phenyl, 4- Difluormethyl-Phenyl, 3,5-Bis(Trifluormethyl)-Phenyl, 3-Trifluormethyl-5-Fluor-Phenyl, 3- Trifluormethyl-5-Chlor-Phenyl, 3 -Methyl-5 -Fluor-Phenyl, 3-Methyl-5-Chlor-Phenyl, 3- Methoxy-5-Fluor-Phenyl, 3-Methoxy-5-Chlor-Phenyl, 3-Trifluormethoxy-5-Chlor-Phenyl, 2- Ethoxy-Phenyl, 3-Ethoxy-Phenyl, 4-Ethoxy-Phenyl, 2-Methylthio-Phenyl, 3-Methylthio-Phenyl, 4-Methylthio-Phenyl, 2-Trifluormethylthio-Phenyl, 3-Trifluormethylthio-Phenyl, 4- Trifluormethylthio-Phenyl, 2-Ethyl-Phenyl, 3-Ethyl-Phenyl, 4-Ethyl-Phenyl, 2- Methoxycarbonyl-Phenyl, 3-Methoxycarbonyl-Phenyl, 4-Methoxycarbonyl-Phenyl, 2- Ethoxycarbonyl-Phenyl, 3-Ethoxycarbonyl-Phenyl, 4-Ethoxycarbonyl-Phenyl, Pyridin-2-yl, Pyridin-3-yl, Pyridin-4-yl, Pyrazin-2-yl, Pyridazin-3-yl, Pyridazin-4-yl, Pyrimidin-2-yl, Pyrimidin-5-yl, Pyrimidin-4-yl, Pyridazin-3-ylmethyl, Pyridazin-4-ylmethyl, Pyrimidin-2- ylmethyl, Pyrimidin-5-ylmethyl, Pyrimidin-4-ylmethyl, Pyrazin-2-ylmethyl, 3-Chlor-Pyrazin-2- yl, 3-Brom-Pyrazin-2-yl, 3-Methoxy-Pyrazin-2-yl, 3-Ethoxy-Pyrazin-2-yl, 3- Trifluormethylpyrazin-2-yl, 3-Cyanopyrazin-2-yl, Naphth-2-yl, Naphth-l-yl, Chinolin-4-yl, Chinolin-6-yl, Chinolin-8-yl, Chinolin-2-yl, Chinoxalin-2-yl, 2-Naphthylmethyl, 1- Naphthylmethyl, Chinolin-4-ylmethyl, Chinolin-6-ylmethyl, Chinolin-8-ylmethyl, Chinolin-2- ylmethyl, Chinoxalin-2-ylmethyl, Pyrazin-2-ylmethyl, 4-Chloropyridin-2-yl, 3-Chloropyridin-4- yl, 2-Chloropyridin-3-yl, 2-Chloropyridin-4-yl, 2-Chlorpyridin-5-yl, 2,6-Dichlorpyridin-4-yl, 3- Chlorpyridin-5-yl, 3,5-Dichlorpyridin-2-yl, 3-Chlor-5-Trifluormethylpyridin-2-yl, (4- Chloropyridin-2-yl)methyl, (3-Chloropyridin-4-yl)methyl, (2-Chloropyridin-3-yl)methyl, (2- Chloropyridin-4-yl)methyl, (2-Chlorpyridin-5-yl)methyl, (2,6-Dichlorpyridin-4-yl)methyl, (3- Chlorpyridin-5-yl)methyl, (3,5-Dichlorpyridin-2-yl)methyl, Thiophen-2-yl, Thiophen-3-yl, 5- Methylthiophen-2-yl, 5-Ethylthiophen-2-yl, 5-Chlorthiophen-2-yl, 5-Bromthiophen-2-yl, 4- Methylthiophen-2-yl, 3-Methylthiophen-2-yl, 5-Fluorthiophen-3-yl, 3,5-Dimethylthiophen-2-yl, 3-Ethylthiophen-2-yl, 4,5-Dimethylthiophen-2-yl, 3,4-Dimethylthiophen-2-yl, 4-Chlorthiophen- 2-yl, Furan-2-yl, 5-Methylfuran-2-yl, 5-Ethylfuran-2-yl, 5-Methoxycarbonylfuran-2-yl, 5- Chlorfuran-2-yl, 5-Bromfuran-2-yl, Thiophan-2-yl, Thiophan-3-yl, Sulfolan-2-yl, Sulfolan-3-yl, Tetrahydrothiopyran-4-yl, Tetrahydropyran-4-yl, Tetrahydrofuran-2-yl, Tetrahydrofuran-3-yl, 4- Methyl-l,3-thiazol-2-yl, l,3-Thiazol-2-yl, Ethenyl, l-Propenyl, 2-Propenyl, 1 -Methyl- ethenyl,3.4.5-trimethoxy-phenyl, 2,4,6-trimethoxy-phenyl, 2-trifluoromethoxy-phenyl, 3-trifluoromethoxy-phenyl, 4-trifluoromethoxy-phenyl, 2-difluoromethoxy-phenyl, 3-difluoromethoxy-phenyl, 4- Difluoromethoxy-phenyl, 2-trifluoromethyl-phenyl, 3-trifluoromethyl-phenyl, 4-trifluoromethyl-phenyl, 2-difluoromethyl-phenyl, 3-difluoromethyl-phenyl, 4-difluoromethyl-phenyl, 3,5-bis (trifluoromethyl) -phenyl , 3-trifluoromethyl-5-fluorophenyl, 3-trifluoromethyl-5-chlorophenyl, 3-methyl-5-fluorophenyl, 3-methyl-5-chlorophenyl, 3-methoxy-5-fluorophenyl , 3-methoxy-5-chlorophenyl, 3-trifluoromethoxy-5-chlorophenyl, 2-ethoxy-phenyl, 3-ethoxy-phenyl, 4-ethoxy-phenyl, 2-methylthio-phenyl, 3-methylthio-phenyl , 4-methylthio-phenyl, 2-trifluoromethylthio-phenyl, 3-trifluoromethylthio-phenyl, 4-trifluoromethylthio-phenyl, 2-ethyl-phenyl, 3-ethyl-phenyl, 4-ethyl-phenyl, 2-methoxycarbonyl-phenyl, 3 -Methoxycarbonyl-phenyl, 4-methoxycarbonyl-phenyl, 2-ethoxycarbonyl-phenyl, 3-ethoxycarbonyl-phenyl, 4-ethoxycarbonyl-phenyl, Py ridin-2-yl, pyridin-3-yl, pyridin-4-yl, pyrazin-2-yl, pyridazin-3-yl, pyridazin-4-yl, pyrimidin-2-yl, pyrimidin-5-yl, pyrimidin 4-yl, pyridazin-3-ylmethyl, pyridazin-4-ylmethyl, pyrimidin-2-ylmethyl, pyrimidin-5-ylmethyl, pyrimidin-4-ylmethyl, pyrazin-2-ylmethyl, 3-chloro-pyrazin-2-yl, 3-bromo-pyrazin-2-yl, 3-methoxy-pyrazin-2-yl, 3-ethoxy-pyrazin-2-yl, 3-trifluoromethylpyrazin-2-yl, 3-cyanopyrazin-2-yl, naphth-2- yl, naphth-l-yl, quinolin-4-yl, quinolin-6-yl, quinolin-8-yl, quinolin-2-yl, quinoxalin-2-yl, 2-naphthylmethyl, 1-naphthylmethyl, quinolin-4- ylmethyl, quinolin-6-ylmethyl, quinolin-8-ylmethyl, quinolin-2-ylmethyl, quinoxalin-2-ylmethyl, pyrazin-2-ylmethyl, 4-chloropyridin-2-yl, 3-chloropyridin-4-yl, 2- Chloropyridin-3-yl, 2-chloropyridin-4-yl, 2-chloropyridin-5-yl, 2,6-dichloropyridin-4-yl, 3- Chloropyridin-5-yl, 3,5-dichloropyridin-2-yl, 3-chloro-5-trifluoromethylpyridin-2-yl, (4-chloropyridin-2-yl) methyl, (3-chloropyridin-4-yl) methyl, (2-chloropyridin-3-yl) methyl, (2-chloropyridin-4-yl) methyl, (2-chloropyridin-5-yl) methyl, (2,6-dichloropyridin-4-yl) methyl, (3-chloropyridine -5-yl) methyl, (3,5-dichloropyridin-2-yl) methyl, thiophene-2-yl, thiophene-3-yl, 5- methylthiophene-2-yl, 5-ethylthiophene-2-yl, 5- Chlorothiophen-2-yl, 5-bromothiophen-2-yl, 4-methylthiophen-2-yl, 3-methylthiophen-2-yl, 5-fluorothiophen-3-yl, 3,5-dimethylthiophen-2-yl, 3- Ethylthiophene-2-yl, 4,5-dimethylthiophene-2-yl, 3,4-dimethylthiophene-2-yl, 4-chlorothiophene-2-yl, furan-2-yl, 5-methylfuran-2-yl, 5- Ethylfuran-2-yl, 5-methoxycarbonylfuran-2-yl, 5-chlorofuran-2-yl, 5-bromfuran-2-yl, thiophan-2-yl, thiophan-3-yl, sulfolan-2-yl, sulfolane 3-yl, tetrahydrothiopyran-4-yl, tetrahydropyran-4-yl, tetrahydrofuran-2-yl, tetrahydrofuran-3-yl, 4-methyl-l, 3-thiazol-2-yl, l, 3-thiazol-2- yl, ethenyl, l-propenyl, 2-propenyl, 1-methyl l- ethenyl,
1-Butenyl, 2-Butenyl, 3-Butenyl, 1 -Methyl- l-propenyl, 2-Methyl- l-propenyl, l-Methyl-2- propenyl, 2-Methyl-2-propenyl, , l-Pentenyl, 2-Pentenyl, 3-Pentenyl, 4-Pentenyl, 1 -Methyl- 1- butenyl, 2-Methyl- l-butenyl, 3 -Methyl- l-butenyl, 1 -Methyl-2-butenyl, 2-Methyl-2-butenyl, 3- Methyl-2-butenyl, l-Methyl-3-butenyl, 2-Methyl-3-butenyl, 3-Methyl-3-butenyl, 1 , 1 -Dimethyl-1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-1-propenyl, 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl,, 1-pentenyl, 2 -Pentenyl, 3-pentenyl, 4-pentenyl, 1-methyl-1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl , 3-methyl-2-butenyl, l-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1, 1-dimethyl-
2-propenyl, 1 ,2-Dimethyl- l-propenyl, 1 ,2-Dimethyl-2-propenyl, l-Ethyl- l-propenyl, l-Ethyl-2- propenyl, l-Hexenyl, 2-Hexenyl, 3-Hexenyl, 4-Hexenyl, 5-Hexenyl, 1 -Methyl- l-pentenyl, 2- Methyl- l-pentenyl, 3 -Methyl- l-pentenyl, 4-Methyl- l-pentenyl, 1 -Methyl-2-pentenyl, 2-Methyl- 2-pentenyl, 3-Methyl-2-pentenyl, 4-Methyl-2-pentenyl, l-Methyl-3-pentenyl, 2-Methyl-3- pentenyl, 3-Methyl-3-pentenyl, 4-Methyl-3-pentenyl, 1 -Methyl-4-pentenyl, 2-Methyl-4- pentenyl, 3-Methyl-4-pentenyl, 4-Methyl-4-pentenyl, 1 , 1 -Dimethyl-2-butenyl, l,l-Dimethyl-3- butenyl, 1 ,2-Dimethyl- l-butenyl, 1 ,2-Dimethyl-2-butenyl, l,2-Dimethyl-3-butenyl, 1,3- Dimethyl- l-butenyl, l,3-Dimethyl-2-butenyl, l,3-Dimethyl-3-butenyl, 2,2-Dimethyl-3-butenyl, 2,3-Dimethyl-l-butenyl, 2,3-Dimethyl-2-butenyl, 2,3-Dimethyl-3-butenyl, 3,3-Dimethyl-l- butenyl, 3,3-Dimethyl-2-butenyl, l-Ethyl- l-butenyl, 1 -Ethyl-2-butenyl, l-Ethyl-3-butenyl, 2- Ethyl- l-butenyl, 2-Ethyl-2-butenyl, 2-Ethyl-3-butenyl, l,l,2-Trimethyl-2-propenyl, l-Ethyl-l- methyl-2-propenyl, 1 -Ethyl-2-methyl- l-propenyl und 1 -Ethyl-2-methyl-2-propenyl, Ethinyl, 1- Propinyl, 2-Propinyl, l-Butinyl, 2-Butinyl, 3-Butinyl, l-Methyl-2-propinyl, l-Pentinyl, 2- Pentinyl, 3-Pentinyl, 4-Pentinyl, 1 -Methyl-2-butinyl, l-Methyl-3-butinyl, 2-Methyl-3-butinyl, 3- Methyl-l-butinyl, l,l-Dimethyl-2-propinyl, 1 -Ethyl-2-propinyl, l-Hexinyl, 2-Hexinyl, 3- Hexinyl, 3,3-Difluorcyclobut-l-yl, 3-Fluorcyclobut-l-yl, l-Fluorcyclobut-l-yl, 2,2- Difluorcycloprop-l-yl, l-Fluorcycloprop-l-yl, 2-Fluorcycloprop-l-yl, 4-Fluorcyclohexyl, 4,4- Difluorcyclohexyl, Methoxy, Ethoxy, n-Propyloxy, iso-Propyloxy, Phenyloxy, p- Chlorphenyloxy, m-Chlorphenyloxy, o-Chlorphenyloxy, p-Fluorphenyloxy, m-Fluorphenyloxy, o-Fluorphenyloxy, p-Methoxyphenyloxy, m-Methoxyphenyloxy, o-Methoxyphenyloxy, p- Trifluormethylphenyloxy, m-Trifluormethylphenyloxy, o-Trifluormethylphenyloxy, 2-propenyl, 1, 2-dimethyl-l-propenyl, 1, 2-dimethyl-2-propenyl, l-ethyl-l-propenyl, l-ethyl-2-propenyl, l-hexenyl, 2-hexenyl, 3- Hexenyl, 4-hexenyl, 5-hexenyl, 1-methyl-1-pentenyl, 2-methyl-1-pentenyl, 3-methyl-1-pentenyl, 4-methyl-1-pentenyl, 1-methyl-2-pentenyl, 2-methyl-2-pentenyl, 3-methyl-2-pentenyl, 4-methyl-2-pentenyl, l-methyl-3-pentenyl, 2-methyl-3-pentenyl, 3-methyl-3-pentenyl, 4- Methyl-3-pentenyl, 1-methyl-4-pentenyl, 2-methyl-4-pentenyl, 3-methyl-4-pentenyl, 4-methyl-4-pentenyl, 1, 1-dimethyl-2-butenyl, l, l-dimethyl-3-butenyl, 1, 2-dimethyl-l-butenyl, 1, 2-dimethyl-2-butenyl, l, 2-dimethyl-3-butenyl, 1,3-dimethyl-l-butenyl, l, 3-dimethyl-2-butenyl, l, 3-dimethyl-3-butenyl, 2,2-dimethyl-3-butenyl, 2,3-dimethyl-l-butenyl, 2,3-dimethyl-2-butenyl, 2, 3-dimethyl-3-butenyl, 3,3-dimethyl-1-butenyl, 3,3-dimethyl-2-butenyl, 1-ethyl-1-butenyl, 1-ethyl-2-butenyl, 1-ethyl-3- butenyl, 2-ethyl-1-butenyl, 2-ethyl-2-butenyl, 2-ethyl-3-butenyl, 1, 1, 2-trimethyl-2-propenyl , l-ethyl-l-methyl-2-propenyl, 1-ethyl-2-methyl-l-propenyl and 1-ethyl-2-methyl-2-propenyl, ethynyl, 1-propynyl, 2-propynyl, l-butynyl , 2-butynyl, 3-butynyl, l-methyl-2-propynyl, l-pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 1-methyl-2-butynyl, l-methyl-3-butynyl, 2nd -Methyl-3-butynyl, 3-methyl-l-butynyl, l, l-dimethyl-2-propynyl, 1-ethyl-2-propynyl, l-hexynyl, 2-hexynyl, 3-hexynyl, 3,3-difluorocyclobut -l-yl, 3-fluorocyclobut-l-yl, l-fluorocyclobut-l-yl, 2,2-difluorocycloprop-l-yl, l-fluorocycloprop-l-yl, 2-fluorocycloprop-l-yl, 4-fluorocyclohexyl , 4,4-difluorocyclohexyl, methoxy, ethoxy, n-propyloxy, iso-propyloxy, phenyloxy, p-chlorophenyloxy, m-chlorophenyloxy, o-chlorophenyloxy, p-fluorophenyloxy, m-fluorophenyloxy, o-fluorophenyloxy, p-methoxyphenyloxy, m Methoxyphenyloxy, o-methoxyphenyloxy, p-trifluoromethylphenyloxy, m-trifluoromethylphenyloxy, o-trifluoromethylphenyloxy,
Cyanomethyl, Cyanoethyl, 3-Cyanoprop-l-yl, 2-Cyanoprop-l-yl, l-Cyanoprop-l-yl, 2- Cyanoprop-2-yl, 2-Cyano-l,l-dimethyleth-l-yl, l-(Cyanomethyl)-l-methylprop-l-yl, 2- (Methoxymethyl)prop-2-yl, 2-(Ethoxymethyl)prop-2-yl, Cyano, Hydroxymethyl, Hydroxyethyl, 2-Hydroxyprop-2-yl stehen, Cyanomethyl, cyanoethyl, 3-cyanoprop-l-yl, 2-cyanoprop-l-yl, l-cyanoprop-l-yl, 2- Cyanoprop-2-yl, 2-cyano-l, l-dimethyleth-l-yl, l- (cyanomethyl) -l-methylprop-l-yl, 2- (methoxymethyl) prop-2-yl, 2- (ethoxymethyl) prop-2-yl, cyano, hydroxymethyl, hydroxyethyl, 2-hydroxyprop-2-yl,
R3 für Hydroxy, Hydrothio, Fluor, Chlor, Brom, Iod, Amino, Methoxy, Ethoxy, n-Propyloxy, 1 - Methylethoxy, n-Butyloxy, 1 -Methylpropyloxy, 2-Methylpropyloxy, l,l-Dimethylethoxy, n- Pentyloxy, 1 -Methylbutyloxy, 2-Methylbutyloxy, 3-Methylbutyloxy, l,l-Dimethylpropyloxy,R 3 for hydroxy, hydrothio, fluorine, chlorine, bromine, iodine, amino, methoxy, ethoxy, n-propyloxy, 1-methylethoxy, n-butyloxy, 1-methylpropyloxy, 2-methylpropyloxy, l, l-dimethylethoxy, n-pentyloxy , 1-methylbutyloxy, 2-methylbutyloxy, 3-methylbutyloxy, l, l-dimethylpropyloxy,
1.2-Dimethylpropyloxy, 2,2-Dimethylpropyloxy, 1 -Ethylpropyloxy, n-Hexyloxy, 1 - Methylpentyloxy, 2-Methylpentyloxy, 3-Methylpentyloxy, 4-Methylpentyloxy, 1,1- Dimethylbutyloxy, 1 ,2-Dimethylbutyloxy, l,3-Di-methylbutyloxy, 2,2-Dimethylbutyloxy, 2,3- Dimethylbutyloxy, 3,3-Dimethylbutyloxy, 1 -Ethylbutyloxy, 2-Ethylbutyloxy, 1,1,2- Trimethylpropyloxy, 1 ,2,2-Trimethylpropyloxy, l-Ethyl-l -methylpropyloxy, l-Ethyl-2- methylpropyloxy, Cyclopropylmethoxy, Cyclobutylmethoxy, Cyclopentylmethoxy, 1,2-dimethylpropyloxy, 2,2-dimethylpropyloxy, 1-ethylpropyloxy, n-hexyloxy, 1 - methylpentyloxy, 2-methylpentyloxy, 3-methylpentyloxy, 4-methylpentyloxy, 1,1-dimethylbutyloxy, 1, 2-dimethylbutyloxy, l, 3- Dimethylbutyloxy, 2,2-dimethylbutyloxy, 2,3-dimethylbutyloxy, 3,3-dimethylbutyloxy, 1-ethylbutyloxy, 2-ethylbutyloxy, 1,1,2-trimethylpropyloxy, 1, 2,2-trimethylpropyloxy, l-ethyl- l -methylpropyloxy, l-ethyl-2-methylpropyloxy, cyclopropylmethoxy, cyclobutylmethoxy, cyclopentylmethoxy,
Cyclohexylmethoxy, Benzyloxy, p-Chlorphenylmethoxy, m-Chlorphenylmethoxy, o- Chlorphenylmethoxy, p-Methoxyphenylmethoxy, p-Nitrophenylmethoxy, Methoxymethoxy, Methoxyethoxy, Methoxy-n-propyloxy, Methoxy-n-butyloxy, Ethoxymethoxy, Ethoxyethoxy, Ethoxy-n-propyloxy, Ethoxy-n-butyloxy, n-Propyloxymethoxy, iso-Propyloxymethoxy, Methylcarbonyloxy, Ethylcarbonyloxy, n-Propylcarbonyloxy, 1 -Methylethylcarbonyloxy, n- Butylcarbonyloxy, 1 -Methylpropylcarbonyloxy, 2-Methylpropylcarbonyloxy, 1,1- Dimethylethylcarbonyloxy, n-Pentylcarbonyloxy, 1 -Methylbutylcarbonyloxy, 2- Methylbutylcarbonyloxy, 3 -Methylbutylcarbonyloxy, l,l-Dimethylpropylcarbonyloxy, 1,2- Dimethylpropylcarbonyloxy, 2,2-Dimethylpropylcarbonyloxy, 1 -Ethylpropylcarbonyloxy, n- Hexylcarbonyloxy, 1 -Methylpentylcarbonyloxy, 2-Methylpentylcarbonyloxy, 3- Methylpentylcarbonyloxy, 4-Methylpentylcarbonyloxy, 1 , 1 -Dimethylbutylcarbonyloxy, 1 ,2- Dimethylbutylcarbonyloxy, 1 ,3-Di-methylbutylcarbonyloxy, 2,2-Dimethylbutylcarbonyloxy, Cyclohexylmethoxy, benzyloxy, p-chlorophenylmethoxy, m-chlorophenylmethoxy, o-chlorophenylmethoxy, p-methoxyphenylmethoxy, p-nitrophenylmethoxy, methoxymethoxy, methoxyethoxy, methoxy-n-propyloxy, methoxy-n-butyloxy, ethoxymethoxy, ethoxyethoxy, ethoxy-noxy Ethoxy-n-butyloxy, n-propyloxymethoxy, iso-propyloxymethoxy, methylcarbonyloxy, ethylcarbonyloxy, n-propylcarbonyloxy, 1-methylethylcarbonyloxy, n-butylcarbonyloxy, 1-methylpropylcarbonyloxy, 2-methylpropylcarbonyloxy, 1,1-dimethylethylcarbonyloxy, n-pentylcarbonate Methylbutylcarbonyloxy, 2-methylbutylcarbonyloxy, 3-methylbutylcarbonyloxy, l, l-dimethylpropylcarbonyloxy, 1,2-dimethylpropylcarbonyloxy, 2,2-dimethylpropylcarbonyloxy, 1-ethylpropylcarbonyloxy, n-hexylcarbonyloxy, 1-methylpentylcarbonyloxy, 2-methylpentylcarbonyl, 2-methylpentylcarbonyl Methylpentylcarbonyloxy, 1, 1-dimethylbutylcarbonyloxy, 1, 2-dimethylbutylcarbonyloxy, 1, 3-dimethylbutylcarbonyloxy, 2,2-dimethylbutylcarbonyloxy,
2.3-Dimethylbutylcarbonyloxy, 3, 3 -Dimethylbutylcarbonyloxy, 1 -Ethylbutylcarbonyloxy, 2- Ethylbutylcarbonyloxy, 1 , 1 ,2-Trimethylpropylcarbonyloxy, 1 ,2,2-Trimethylpropylcarbonyloxy, 1 -Ethyl- 1 -methylpropylcarbonyloxy, 1 -Ethyl-2-methylpropylcarbonyloxy, Phenylcarbonyloxy, p-Chlorphenylcarbonyloxy, m-Chlorphenylcarbonyloxy, o-Chlorphenylcarbonyloxy, p- Fluorphenylcarbonyloxy, m-Fluorphenylcarbonyloxy, o-Fluorphenylcarbonyloxy, 2,3-dimethylbutylcarbonyloxy, 3, 3 -dimethylbutylcarbonyloxy, 1 -ethylbutylcarbonyloxy, 2-ethylbutylcarbonyloxy, 1, 1, 2-trimethylpropylcarbonyloxy, 1, 2,2-trimethylpropylcarbonyloxy, 1-ethyl-1-methylpropylcarbonyloxy, 1-ethylpropyloxy, 1-ethylpropyloxy Phenylcarbonyloxy, p-chlorophenylcarbonyloxy, m-chlorophenylcarbonyloxy, o-chlorophenylcarbonyloxy, p-fluorophenylcarbonyloxy, m-fluorophenylcarbonyloxy, o-fluorophenylcarbonyloxy,
Benzylcarbonyloxy, Cyclopropylcarbonyloxy, Cyclobutylcarbonyloxy, Benzylcarbonyloxy, cyclopropylcarbonyloxy, cyclobutylcarbonyloxy,
Cyclopentylcarbonyloxy, Cyclohexylcarbonyloxy, Trifluormethylcarbonyloxy, Cyclopentylcarbonyloxy, cyclohexylcarbonyloxy, trifluoromethylcarbonyloxy,
Difluormethylcarbonyloxy, Methoxycarbonyloxy, Ethoxycarbonyloxy, n- Propyloxycarbonyloxy, iso-Propyloxycarbonyloxy, n-Butyloxycarbonyloxy, 1,1- Dimethylethyloxycarbonyloxy, 2,2-Dimethylpropyloxycarbonyloxy, Benzyloxycarbonyloxy, Allylcarbonyloxy, Methylsulfonyloxy, Ethylsulfonyloxy, n-Propylsulfonyloxy, 1 - Methylethylsulfonyloxy, Cyclopropylsulfonyloxy Cyclobutylsulfonyloxy, Cyclopentylsulfonyloxy Cyclohexylsulfonyloxy, Phenylsulfonyloxy, p-Chlorphenylsulfonyloxy, m-Chlorphenylsulfonyloxy, o-Chlorphenylsulfonyloxy, p-Fluorphenylsulfonyloxy, m- Fluorphenylsulfonyloxy, o-Fluorphenylsulfonyloxy, p-Methoxyphenylsulfonyloxy, m- Methoxyphenylsulfonyloxy, o-Methoxyphenylsulfonyloxy, p-Methylphenylsulfonyloxy, m- Methylphenylsulfonyloxy, o-Methylphenylsulfonyloxy steht, und Difluoromethylcarbonyloxy, methoxycarbonyloxy, ethoxycarbonyloxy, n-propyloxycarbonyloxy, iso-propyloxycarbonyloxy, n-butyloxycarbonyloxy, 1,1-dimethylethyloxycarbonyloxy, 2,2-dimethylpropyloxycarbonyloxy, benzyloxycarbonyloxy, allylcarbonyloxy, methylsulfonyloxy, ethylylsulfonyloxy, ethylylsulfonyloxy, Cyclopentylsulfonyloxy Cyclohexylsulfonyloxy, phenylsulfonyloxy, p-chlorophenylsulfonyloxy, m-chlorophenylsulfonyloxy, o-chlorophenylsulfonyloxy, p-fluorophenylsulfonyloxy, m- fluorophenylsulfonyloxy, o-fluorophenylsulfonyloxy, p-methoxyphenylsulfonyloxy, m- methoxyphenylsulfonyloxy, o-methoxyphenylsulfonyloxy, p-methylphenylsulfonyloxy, m- methylphenylsulfonyloxy, o -Methylphenylsulfonyloxy stands, and
R4 für Wasserstoff, Methyl, Ethyl steht. R 4 represents hydrogen, methyl, ethyl.
Im Speziellen bevorzugter Erfindungsgegenstand sind Verbindungen der allgemeinen Formel (I), worin Particularly preferred subject matter of the invention are compounds of general formula (I), wherein
X und Y unabhängig voneinander für C-H oder die Gruppierung C-R1 stehen, wobei X and Y independently of one another represent CH or the grouping CR 1 , where
X für C-H steht, wenn Y für die Gruppierung C-R1 steht und X stands for CH if Y stands for the grouping CR 1 and
X für die Gruppierung C-R1 steht, wenn Y für C-H steht, X stands for the grouping CR 1 , if Y stands for CH,
R1 für Chlor, Brom, Iod, Cyano, Methyl, Ethyl, n-Propyl, 1 -Methylethyl, n-Butyl, 1 -Methylpropyl, 2- Methylpropyl, l,l-Dimethylethyl, n-Pentyl, 1 -Methylbutyl, 2-Methylbutyl, 3-Methylbutyl, 1,1- Dimethylpropyl, 1 ,2-Dimethylpropyl, 2,2-Dimethylpropyl, 1 -Ethylpropyl, n-Hexyl, 1 - Methylpentyl, 2-Methylpentyl, 3-Methylpentyl, 4-Methylpentyl, l,l-Dimethylbutyl, 1,2- Dimethylbutyl, l,3-Di-methylbutyl, 2,2-Dimethylbutyl, 2,3-Dimethylbutyl, 3,3-Dimethylbutyl,R 1 for chlorine, bromine, iodine, cyano, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl, l, l-dimethylethyl, n-pentyl, 1-methylbutyl, 2 -Methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1, 2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, n-hexyl, 1 - methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, l , l-dimethylbutyl, 1,2-dimethylbutyl, l, 3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl,
1 -Ethylbutyl, 2-Ethylbutyl, 1 , 1 ,2-Trimethylpropyl, 1 ,2,2-Trimethylpropyl, 1 -Ethyl- 1- methylpropyl, 1 -Ethyl-2-methylpropyl, Cyclopropyl, Cyclobutyl, Cyclopentyl, Cyclohexyl, Adamantan-l-yl, Adamantan-2-yl, 1 -Methylcyclopropyl, 2-Methylcyclopropyl, 2,2- Dimethylcyclopropyl, 2,3-Dimethylcyclopropyl, l,l'-Bi(cyclopropyl)-l-yl, 1,1'- Bi(cyclopropyl)-2-yl, 2'-Methyl-l,l'-bi(cyclopropyl)-2-yl, 1 -Cyanocyclopropyl, 2- Cyanocyclopropyl, 1 -Methylcyclobutyl, 2-Methylcyclobutyl, 3-Methylcyclobutyl, 3,3- Dimethylcyclobut-l-yl, 3,3-Difluorcyclobut-l-yl, 3-Fluorcyclobut-l-yl, 2,2-Difluorcycloprop-l- yl, 1-Fluorcycloprop-l-yl, 2-Fluorcycloprop-l-yl, 1 -Ethylcyclopropyl, 1 -Methylcyclohexyl, 2- Methylcyclohexyl, 3 -Methylcyclohexyl, 1 -Methoxycyclohexyl, 2-Methoxycyclohexyl, 3- Methoxycyclohexyl, Trifluormethyl, Pentafluorethyl, 1,1,2,2-Tetrafluorethyl, Heptafluorpropyl, Nonafluorbutyl, Chlordifluormethyl, Difluormethyl, 2,2-Difluorethyl, 2,2,2-Trifluorethyl, Difluor-tert.-butyl, Chlormethyl, Brommethyl, Methoxy, Ethoxy, n-Propyloxy, iso-Propyloxy, n-Butyloxy, tert-Butyloxy, Methoxymethyl, Ethoxymethyl, Methoxyethyl, Ethoxyethyl, Trifluormethoxy, Difluormethoxy, 2,2,2-Trifluorethoxy, 2,2-Difluorethoxy, Methylthio, Ethylthio, Trifluormethylthio, Pentafluorethylthio, Phenyl, Phenyloxy, p-Cl-Phenyloxy, Thiophen-2-yl, Thiophen-3-yl, Pyridin-2-yl, Pyridin-3-yl, Pyridin-4-yl, Furan-2-yl, Furan-3-yl, Cyclopropylmethyl, Cyclobutylmethyl, Cyclopentylmethyl, Cyclohexylmethyl, Ethenyl, 1 - Propenyl, 2-Propenyl, 1 -Methyl- ethenyl, l-Butenyl, 2-Butenyl, 3-Butenyl, l-Methyl-l- propenyl, 2-Methyl-l -propenyl, l-Methyl-2-propenyl, 2-Methyl-2-propenyl, l-Pentenyl, 2- Pentenyl, 3-Pentenyl, 4-Pentenyl, Ethinyl, l-Propinyl, 2-Propinyl, l-Butinyl, l-Pentinyl, 1- Hexinyl steht, 1-ethylbutyl, 2-ethylbutyl, 1, 1, 2-trimethylpropyl, 1, 2,2-trimethylpropyl, 1-ethyl-1-methylpropyl, 1-ethyl-2-methylpropyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, adamantane l-yl, adamantan-2-yl, 1-methylcyclopropyl, 2-methylcyclopropyl, 2,2-dimethylcyclopropyl, 2,3-dimethylcyclopropyl, l, l'-bi (cyclopropyl) -l-yl, 1,1'-bi (cyclopropyl) -2-yl, 2'-methyl-l, l'-bi (cyclopropyl) -2-yl, 1-cyanocyclopropyl, 2-cyanocyclopropyl, 1-methylcyclobutyl, 2-methylcyclobutyl, 3-methylcyclobutyl, 3.3 - Dimethylcyclobut-l-yl, 3,3-difluorocyclobut-l-yl, 3-fluorocyclobut-l-yl, 2,2-difluorocycloprop-l-yl, 1-fluorocycloprop-l-yl, 2-fluorocycloprop-l-yl , 1-ethylcyclopropyl, 1-methylcyclohexyl, 2-methylcyclohexyl, 3-methylcyclohexyl, 1-methoxycyclohexyl, 2-methoxycyclohexyl, 3-methoxycyclohexyl, trifluoromethyl, pentafluoroethyl, 1,1,2,2-tetrafluoroethyl, heptafluoropropyl, nonafluorobluoromethyl, chlafluorobluoromethyl , 2,2-difluoroethyl, 2,2,2-trifluoroethyl, difluoro-tert-butyl, chloromethyl, Bromomethyl, methoxy, ethoxy, n-propyloxy, iso-propyloxy, n-butyloxy, tert-butyloxy, methoxymethyl, ethoxymethyl, methoxyethyl, ethoxyethyl, trifluoromethoxy, difluoromethoxy, 2,2,2-trifluoroethoxy, 2,2-difluoroethoxy, methylthio, Ethylthio, trifluoromethylthio, pentafluoroethylthio, phenyl, phenyloxy, p-Cl-phenyloxy, Thiophene-2-yl, thiophene-3-yl, pyridin-2-yl, pyridin-3-yl, pyridin-4-yl, furan-2-yl, furan-3-yl, cyclopropylmethyl, cyclobutylmethyl, cyclopentylmethyl, cyclohexylmethyl, Ethenyl, 1-propenyl, 2-propenyl, 1-methyl-ethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-1-propenyl, 2-methyl-1-propenyl, 1-methyl-2- propenyl, 2-methyl-2-propenyl, l-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, ethynyl, l-propynyl, 2-propynyl, l-butynyl, l-pentynyl, 1-hexynyl,
R3 für Hydroxy, Chlor, Amino, Methoxy, Ethoxy, n-Propyloxy, 1 -Methylethoxy, n-Butyloxy, 1 -R 3 for hydroxy, chlorine, amino, methoxy, ethoxy, n-propyloxy, 1-methylethoxy, n-butyloxy, 1 -
Methylpropyloxy, 2-Methylpropyloxy, l,l-Dimethylethoxy, n-Pentyloxy, Cyclopropylmethoxy, Cyclobutylmethoxy, Cyclopentylmethoxy, Cyclohexylmethoxy, Benzyloxy, p- Chlorphenylmethoxy, m-Chlorphenylmethoxy, o-Chlorphenylmethoxy, p- Methoxyphenylmethoxy, Methoxymethoxy, Methoxyethoxy, Methoxy-n-propyloxy, Methyl propyloxy, 2-methyl propyloxy, l, l-dimethylethoxy, n-pentyloxy, cyclopropyl methoxy, cyclobutyl methoxy, cyclopentyl methoxy, cyclohexyl methoxy, benzyloxy, p-chlorophenyl methoxy, m-chlorophenyl methoxy, o-chlorophenyl methoxy, p-methoxyphenyl methoxy, methoxy methoxy, methoxymoxy meth propyloxy,
Ethoxymethoxy, Ethoxyethoxy, Methylcarbonyloxy, Ethylcarbonyloxy, n-Propylcarbonyloxy, Ethoxymethoxy, ethoxyethoxy, methylcarbonyloxy, ethylcarbonyloxy, n-propylcarbonyloxy,
1 -Methylethylcarbonyloxy, n-Butylcarbonyloxy, 1 -Methylpropylcarbonyloxy, 2- Methylpropylcarbonyloxy, 1 , 1 -Dimethylethylcarbonyloxy, n-Pentylcarbonyloxy, 1-methylethylcarbonyloxy, n-butylcarbonyloxy, 1-methylpropylcarbonyloxy, 2-methylpropylcarbonyloxy, 1, 1-dimethylethylcarbonyloxy, n-pentylcarbonyloxy,
Phenylcarbonyloxy, p-Chlorphenylcarbonyloxy, m-Chlorphenylcarbonyloxy, o- Chlorphenylcarbonyloxy, p-Fluorphenylcarbonyloxy, m-Fluorphenylcarbonyloxy, o- Fluorphenylcarbonyloxy, Cyclopropylcarbonyloxy, Cyclobutylcarbonyloxy, Phenylcarbonyloxy, p-chlorophenylcarbonyloxy, m-chlorophenylcarbonyloxy, o-chlorophenylcarbonyloxy, p-fluorophenylcarbonyloxy, m-fluorophenylcarbonyloxy, o-fluorophenylcarbonyloxy, cyclopropylcarbonyloxy, cyclobutylcarbonyloxy,
Cyclopentylcarbonyloxy, Cyclohexylcarbonyloxy, Trifluormethylcarbonyloxy, Cyclopentylcarbonyloxy, cyclohexylcarbonyloxy, trifluoromethylcarbonyloxy,
Difluormethylcarbonyloxy, Methoxycarbonyloxy, Ethoxycarbonyloxy, n- Propyloxycarbonyloxy, iso-Propyloxycarbonyloxy, n-Butyloxycarbonyloxy, 1,1- Dimethylethyloxycarbonyloxy, Benzyloxycarbonyloxy, 2,2-Dimethylpropyloxycarbonyloxy, Allylcarbonyloxy, Methylsulfonyloxy, Ethylsulfonyloxy, n-Propylsulfonyloxy, 1 - Methylethylsulfonyloxy, Cyclopropylsulfonyloxy steht, Difluoromethylcarbonyloxy, methoxycarbonyloxy, ethoxycarbonyloxy, n-propyloxycarbonyloxy, iso-propyloxycarbonyloxy, n-butyloxycarbonyloxy, 1,1-dimethylethyloxycarbonyloxy, benzyloxycarbonyloxy, 2,2-dimethylpropyloxycarbonyloxy, allylcarbonyloxy, methylsulfonyloxy, ethylylsulfonyloxy, ethylylsulfonyl
R4 für Wasserstoff steht, R 4 represents hydrogen,
Q für eine der in der folgenden Tabelle spezifisch genannten Gruppierungen Q-l .1 bis Q-l .68 steht: Q stands for one of the groupings Q-1 .1 to Q-1 .68 specifically mentioned in the following table:
Im besonders Speziellen bevorzugter Erfindungsgegenstand sind Verbindungen der allgemeinen Formel (I), worin Particularly particularly preferred subject matter of the invention are compounds of the general formula (I), in which
X und Y unabhängig voneinander für C-H oder die Gruppierung C-R1 stehen, wobei X and Y independently of one another represent CH or the grouping CR 1 , where
X für C-H steht, wenn Y für die Gruppierung C-R1 steht und X stands for CH if Y stands for the grouping CR 1 and
X für die Gruppierung C-R1 steht, wenn Y für C-H steht, X stands for the grouping CR 1 , if Y stands for CH,
R1 für Chlor, Brom, lod, Cyano, Methyl, Ethyl, n-Propyl, 1 -Methylethyl, n-Butyl, 1 -Methylpropyl, 2- Methylpropyl, l,l-Dimethylethyl, n-Pentyl, 1 -Methylbutyl, 2-Methylbutyl, 3-Methylbutyl, 1,1- Dimethylpropyl, 1 ,2-Dimethylpropyl, 2,2-Dimethylpropyl, 1 -Ethylpropyl, n-Hexyl, 1 - Methylpentyl, 2-Methylpentyl, 3-Methylpentyl, 4-Methylpentyl, l,l-Dimethylbutyl, 1,2- Dimethylbutyl, l,3-Di-methylbutyl, 2,2-Dimethylbutyl, 2,3-Dimethylbutyl, 3,3-Dimethylbutyl,R 1 for chlorine, bromine, iodine, cyano, methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl, l, l-dimethylethyl, n-pentyl, 1-methylbutyl, 2 -Methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1, 2-dimethylpropyl, 2,2-dimethylpropyl, 1 -ethylpropyl, n-hexyl, 1 - Methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, l, l-dimethylbutyl, 1,2-dimethylbutyl, l, 3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3- dimethylbutyl,
1 -Ethylbutyl, 2-Ethylbutyl, 1 , 1 ,2-Trimethylpropyl, 1 ,2,2-Trimethylpropyl, l-Ethyl-l- methylpropyl, 1 -Ethyl-2-methylpropyl, Cyclopropyl, Cyclobutyl, Cyclopentyl, Cyclohexyl, Adamantan-l-yl, Adamantan-2-yl, 1 -Methylcyclopropyl, 2-Methylcyclopropyl, 2,2- Dimethylcyclopropyl, 2,3-Dimethylcyclopropyl, l,l'-Bi(cyclopropyl)-l-yl, 1,1'- Bi(cyclopropyl)-2-yl, 2'-Methyl-l,l'-bi(cyclopropyl)-2-yl, 1 -Cyanocyclopropyl, 2- Cyanocyclopropyl, 1 -Methylcyclobutyl, 2-Methylcyclobutyl, 3-Methylcyclobutyl, 3,3- Dimethylcyclobut-l-yl, 3,3-Difluorcyclobut-l-yl, 3-Fluorcyclobut-l-yl, 2,2-Difluorcycloprop-l- yl, l-Fluorcycloprop-l-yl, 2-Fluorcycloprop-l-yl, 1 -Ethylcyclopropyl, 1 -Methylcyclohexyl, 2- Methylcyclohexyl, 3 -Methylcyclohexyl, 1 -Methoxycyclohexyl, 2-Methoxycyclohexyl, 3- Methoxycyclohexyl, Trifluormethyl, Pentafluorethyl, l,l,2,2-Tetrafluorethyl, Heptafluorpropyl, Nonafluorbutyl, Chlordifluormethyl, Difluormethyl, 2,2-Difluorethyl, 2,2,2-Trifluorethyl, Difluor-tert.-butyl, Methoxy, Ethoxy, n-Propyloxy, iso-Propyloxy, n-Butyloxy, tert-Butyloxy, Methoxymethyl, Ethoxymethyl, Methoxyethyl, Ethoxyethyl, Trifluormethoxy, Difluormethoxy, 2,2,2-Trifluorethoxy, 2,2-Difluorethoxy, Methylthio, Ethylthio, Trifluormethylthio, 1-ethylbutyl, 2-ethylbutyl, 1, 1, 2-trimethylpropyl, 1, 2,2-trimethylpropyl, l-ethyl-l-methylpropyl, 1-ethyl-2-methylpropyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, adamantane l-yl, adamantan-2-yl, 1-methylcyclopropyl, 2-methylcyclopropyl, 2,2-dimethylcyclopropyl, 2,3-dimethylcyclopropyl, l, l'-bi (cyclopropyl) -l-yl, 1,1'-bi (cyclopropyl) -2-yl, 2'-methyl-l, l'-bi (cyclopropyl) -2-yl, 1-cyanocyclopropyl, 2-cyanocyclopropyl, 1-methylcyclobutyl, 2-methylcyclobutyl, 3-methylcyclobutyl, 3.3 - Dimethylcyclobut-l-yl, 3,3-difluorocyclobut-l-yl, 3-fluorocyclobut-l-yl, 2,2-difluorocycloprop-l-yl, l-fluorocycloprop-l-yl, 2-fluorocycloprop-l-yl , 1-ethylcyclopropyl, 1-methylcyclohexyl, 2-methylcyclohexyl, 3-methylcyclohexyl, 1-methoxycyclohexyl, 2-methoxycyclohexyl, 3-methoxycyclohexyl, trifluoromethyl, pentafluoroethyl, l, l, 2,2-tetrafluoroethyl, heptafluoropropyl, nonafluorobluoromethyl , 2,2-difluoroethyl, 2,2,2-trifluoroethyl, difluoro-tert-butyl, methoxy, ethox y, n-propyloxy, iso-propyloxy, n-butyloxy, tert-butyloxy, methoxymethyl, ethoxymethyl, methoxyethyl, ethoxyethyl, trifluoromethoxy, difluoromethoxy, 2,2,2-trifluoroethoxy, 2,2-difluoroethoxy, methylthio, ethylthio, trifluoromethylthio,
Pentafluorethylthio, Phenyl, Phenyloxy, p-Cl-Phenyloxy, Thiophen-2-yl, Thiophen-3-yl, Pyridin-2-yl, Pyridin-3-yl, Pyridin-4-yl, Furan-2-yl, Furan-3-yl, Cyclopropylmethyl, Pentafluoroethylthio, phenyl, phenyloxy, p-Cl-phenyloxy, thiophene-2-yl, thiophene-3-yl, pyridin-2-yl, pyridin-3-yl, pyridin-4-yl, furan-2-yl, furan 3-yl, cyclopropylmethyl,
Cyclobutylmethyl, Cyclopentylmethyl, Cyclohexylmethyl, Ethenyl, 1 -Propenyl, 2-Propenyl, 1 - Methyl- ethenyl, l-Butenyl, 2-Butenyl, 3-Butenyl, 1 -Methyl- 1 -propenyl, 2-Methyl-l -propenyl, Cyclobutylmethyl, cyclopentylmethyl, cyclohexylmethyl, ethenyl, 1-propenyl, 2-propenyl, 1-methyl-ethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-1-propenyl, 2-methyl-1-propenyl,
1 -Methyl-2-propenyl, 2-Methyl-2-propenyl, l-Pentenyl, 2-Pentenyl, 3-Pentenyl, 4-Pentenyl, Prop-2-en-l-yloxy, But-3-en-l-yloxy, Pent-4-en-l-yloxy, Ethinyl, l-Propinyl, 2-Propinyl, 1- Butinyl, 1 -Pentinyl, 1 -Hexinyl steht, 1-methyl-2-propenyl, 2-methyl-2-propenyl, l-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, prop-2-en-l-yloxy, but-3-en-l- yloxy, pent-4-en-l-yloxy, ethynyl, l-propynyl, 2-propynyl, 1-butynyl, 1-pentynyl, 1-hexynyl,
R3 für Hydroxy, Methylcarbonyloxy, Ethylcarbonyloxy, n-Propylcarbonyloxy, 1 -R 3 for hydroxy, methylcarbonyloxy, ethylcarbonyloxy, n-propylcarbonyloxy, 1 -
Methylethylcarbonyloxy, Cyclopropylcarbonyloxy, 1 , 1 -Dimethylethylcarbonyloxy, Methylethylcarbonyloxy, cyclopropylcarbonyloxy, 1, 1 -dimethylethylcarbonyloxy,
Methoxycarbonyloxy, Ethoxycarbonyloxy, 1 , 1 -Dimethylethyloxycarbonyloxy und Methoxycarbonyloxy, ethoxycarbonyloxy, 1, 1-dimethylethyloxycarbonyloxy and
Benzyloxycarbonyloxy steht, Benzyloxycarbonyloxy stands,
R4 für Wasserstoff steht, R 4 represents hydrogen,
Q für eine der in der oben stehenden Tabelle spezifisch genannten Gruppierungen Q-l .1 bis Q-l .68 steht. Im extrem Speziellen bevorzugter Erfindungsgegenstand sind Verbindungen der allgemeinen Formel (I), worin Q stands for one of the groupings Ql .1 to Ql .68 specifically mentioned in the table above. Particularly extremely preferred subject matter of the invention are compounds of the general formula (I), wherein
X und Y unabhängig voneinander für C-H oder die Gruppierung C-R1 stehen, wobei X and Y independently of one another represent CH or the grouping CR 1 , where
X für C-H steht, wenn Y für die Gruppierung C-R1 steht und X stands for CH if Y stands for the grouping CR 1 and
X für die Gruppierung C-R1 steht, wenn Y für C-H steht, X stands for the grouping CR 1 , if Y stands for CH,
R1 für Chlor, Brom, Methyl, Ethyl, 1 -Methylethyl, l,l-Dimethylethyl, Methoxy, Ethoxy, R 1 for chlorine, bromine, methyl, ethyl, 1-methylethyl, l, l-dimethylethyl, methoxy, ethoxy,
Trifluormethyl, Methylthio, Ethylthio, Phenyl steht, Trifluoromethyl, methylthio, ethylthio, phenyl,
R3 für Hydroxy, Methylcarbonyloxy, Ethylcarbonyloxy, n-Propylcarbonyloxy, 1 -R 3 for hydroxy, methylcarbonyloxy, ethylcarbonyloxy, n-propylcarbonyloxy, 1 -
Methylethylcarbonyloxy, Cyclopropylcarbonyloxy, 1 , 1 -Dimethylethylcarbonyloxy, Methylethylcarbonyloxy, cyclopropylcarbonyloxy, 1, 1 -dimethylethylcarbonyloxy,
Methoxycarbonyloxy, Ethoxycarbonyloxy, 1 , 1 -Dimethylethyloxycarbonyloxy und Methoxycarbonyloxy, ethoxycarbonyloxy, 1, 1-dimethylethyloxycarbonyloxy and
Benzyloxycarbonyloxy steht, Benzyloxycarbonyloxy stands,
R4 für Wasserstoff steht, und R 4 represents hydrogen, and
Q für eine der in der oben stehenden Tabelle spezifisch genannten Gruppierungen Q-l .1 bis Q-l .68 steht. Q stands for one of the groupings Q-1 .1 to Q-1 .68 specifically mentioned in the table above.
Die oben aufgeführten allgemeinen oder in Vorzugsbereichen aufgeführten Restedefinitionen gelten sowohl für die Endprodukte der allgemeinen Formel (1) als auch entsprechend für die jeweils zur Herstellung benötigten Ausgangs- oder Zwischenprodukte. Diese Restedefinitionen können The general or preferred radical definitions listed above apply both to the end products of the general formula (1) and correspondingly to the starting or intermediate products required in each case for the preparation. These residual definitions can
untereinander, also auch zwischen den angegebenen bevorzugten Bereichen beliebig kombiniert werden. can be combined with one another, i.e. also between the specified preferred ranges.
Vor allem aus den Gründen der höheren herbiziden Wirkung, besseren Selektivität und/oder besseren Herstellbarkeit sind erfindungsgemäße Verbindungen der genannten allgemeinen Formel (1) oder deren Salze bzw. deren erfindungsgemäße Verwendung von besonderem lnteresse, worin einzelne Reste eine der bereits genannten oder im folgenden genannten bevorzugten Bedeutungen haben, oder insbesondere solche, worin eine oder mehrere der bereits genannten oder im Folgenden genannten bevorzugten Bedeutungen kombiniert auftreten. lm Hinblick auf die erfindungsgemäßen Verbindungen der allgemeinen Formel (1) werden die vorstehend und weiter unten verwendeten Bezeichnungen erläutert. Diese sind dem Fachmann geläufig und haben insbesondere die im Folgenden erläuterten Bedeutungen: Sofern nicht anders definiert, gilt generell für die Bezeichnung von chemischen Gruppen, dass die Anbindung an das Gerüst bzw. den Rest des Moleküls über das zuletzt genannte Strukturelement der betreffenden chemischen Gruppe erfolgt, d.h. beispielsweise im Falle von (C2-Cg)-Alkenyloxy über das Sauerstoffatom, und im Falle von Heterocyclyl-(Ci-C8)-alkyl oder R50(0)C-(Ci -Cs)- Alkyl jeweils über das C-Atom der Alkylgruppe. Especially for the reasons of the higher herbicidal activity, better selectivity and / or better manufacturability, compounds of the general formula (1) according to the invention or their salts or their use according to the invention are of particular interest, in which individual radicals are one of those already mentioned or mentioned below have preferred meanings, or in particular those in which one or more of the preferred meanings already mentioned or mentioned below occur in combination. With regard to the compounds of the general formula (1) according to the invention, the terms used above and below are explained. These are familiar to the person skilled in the art and in particular have the meanings explained below: Unless otherwise defined, it generally applies to the designation of chemical groups that the connection to the framework or the rest of the molecule takes place via the last-mentioned structural element of the chemical group in question, ie for example in the case of (C2-Cg) -alkenyloxy via the oxygen atom, and in the case of heterocyclyl- (Ci-C 8 ) -alkyl or R 5 0 (0) C- (Ci-Cs) - alkyl each via the C-atom of the alkyl group.
Erfindungsgemäß steht "Alkylsulfonyl" - in Alleinstellung oder als Bestandteil einer chemischen Gruppe - für geradkettiges oder verzweigtes Alkylsulfonyl, vorzugsweise mit 1 bis 8, oder mit 1 bis 6 According to the invention, "alkylsulfonyl" - alone or as part of a chemical group - stands for straight-chain or branched alkylsulfonyl, preferably with 1 to 8, or with 1 to 6
Kohlenstoffatomen, z.B. (aber nicht beschränkt auf) (Ci-C6)-Alkylsulfonyl wie Methylsulfonyl, Ethyl- sulfonyl, Propylsulfonyl, 1 -Methylethylsulfonyl, Butylsulfonyl, 1 -Methylpropylsulfonyl, 2-Methyl- propylsulfonyl, l,l-Dimethylethylsulfonyl, Pentylsulfonyl, 1 -Methylbutylsulfonyl, 2-Methylbutyl- sulfonyl, 3 -Methylbutylsulfonyl, l,l-Dimethylpropylsulfonyl, 1 ,2-Dimethylpropylsulfonyl, 2,2-Di- methylpropylsulfonyl, 1 -Ethylpropylsulfonyl, Hexylsulfonyl, 1 -Methylpentylsulfonyl, 2-Methyl- pentylsulfonyl, 3 -Methylpentylsulfonyl, 4-Methylpentylsulfonyl, l,l-Dimethylbutylsulfonyl, l,2-Di- methylbutylsulfonyl, 1 ,3-Dimethylbutylsulfonyl, 2,2-Dimethylbutylsulfonyl, 2,3-Dimethylbutylsulfonyl, 3 ,3 -Dimethylbutylsulfonyl, 1 -Ethylbutylsulfonyl, 2-Ethylbutylsulfonyl, 1 , 1 ,2-Trimethylpropylsulfonyl,Carbon atoms, for example (but not limited to) (Ci-C 6 ) alkylsulfonyl such as methylsulfonyl, ethylsulfonyl, propylsulfonyl, 1-methylethylsulfonyl, butylsulfonyl, 1-methylpropylsulfonyl, 2-methylpropylsulfonyl, l, l-dimethylethylsulfonyl, pentyl 1-methylbutylsulfonyl, 2-methylbutylsulfonyl, 3-methylbutylsulfonyl, l, l-dimethylpropylsulfonyl, 1, 2-dimethylpropylsulfonyl, 2,2-dimethylpropylsulfonyl, 1-ethylpropylsulfonyl, hexylsulfonylsulfonyl, 1-methylsulfonyl, 1-methyl 3-methylpentylsulfonyl, 4-methylpentylsulfonyl, l, l-dimethylbutylsulfonyl, l, 2-dimethylbutylsulfonyl, 1, 3-dimethylbutylsulfonyl, 2,2-dimethylbutylsulfonyl, 2,3-dimethylbutylsulfonyl, 3, 3-dimethylbutylsylonyl, 2-ethylbutylsulfonyl, 1, 1, 2-trimethylpropylsulfonyl,
1 ,2,2-Trimethylpropylsulfonyl, 1 -Ethyl- 1 -methylpropylsulfonyl und 1 -Ethyl-2-methylpropylsulfonyl. 1, 2,2-trimethylpropylsulfonyl, 1-ethyl-1-methylpropylsulfonyl and 1-ethyl-2-methylpropylsulfonyl.
Erfindungsgemäß steht "Heteroarylsulfonyl" für gegebenenfalls substituiertes Pyridylsulfonyl, According to the invention, "heteroarylsulfonyl" represents optionally substituted pyridylsulfonyl,
Pyrimidinylsulfonyl, Pyrazinylsulfonyl oder gegebenenfalls substituiertes polycyclisches Pyrimidinylsulfonyl, pyrazinylsulfonyl or optionally substituted polycyclic
Heteroarylsulfonyl, hier insbesondere gegebenenfalls substituiertes Chinolinylsulfonyl, beispielsweise substituiert durch Fluor, Chlor, Brom, lod, Cyano, Nitro, Alkyl-, Haloalkyl-, Haloalkoxy-, Amino-, Alkylamino-, Alkylcarbonylamino-, Dialkylamino- oder Alkoxygruppen. Heteroarylsulfonyl, here in particular optionally substituted quinolinylsulfonyl, for example substituted by fluorine, chlorine, bromine, iodine, cyano, nitro, alkyl, haloalkyl, haloalkoxy, amino, alkylamino, alkylcarbonylamino, dialkylamino or alkoxy groups.
Erfindungsgemäß steht "Alkylthio" - in Alleinstellung oder als Bestandteil einer chemischen Gruppe - für geradketiges oder verzweigtes S-Alkyl, vorzugsweise mit 1 bis 8, oder mit 1 bis 6 According to the invention, "alkylthio" - alone or as part of a chemical group - stands for straight-chain or branched S-alkyl, preferably with 1 to 8, or with 1 to 6
Kohlenstoffatomen, wie (Ci-Cio)-, (CI-CÖ)- oder (Ci-C i)-Alkylthio, z.B. (aber nicht beschränkt auf) (Ci- C6)-Alkylthio wie Methylthio, Ethylthio, Propylthio, 1 -Methylethylthio, Butylthio, 1 -Methylpropylthio, 2-Methylpropylthio, l,l-Dimethylethylthio, Pentylthio, 1 -Methylbutylthio, 2-Methylbutylthio, 3- Methylbutylthio, l,l-Dimethylpropylthio, 1 ,2-Dimethylpropylthio, 2,2-Dimethylpropylthio, 1- Ethylpropylthio, Hexylthio, 1 -Methylpentylthio, 2-Methylpentylthio, 3-Methylpentylthio, 4-Methyl- pentylthio, l,l-Dimethylbutylthio, 1 ,2-Dimethylbutylthio, l,3-Dimethylbutylthio, 2,2-Dimethylbutyl- thio, 2,3-Dimethylbutylthio, 3,3-Dimethylbutylthio, 1 -Ethylbutylthio, 2-Ethylbutylthio, l,l,2-Tri- methylpropylthio, 1 ,2,2-Trimethylpropylthio, 1 -Ethyl- 1 -methylpropylthio und 1 -Ethyl-2-methyl- propylthio. „Alkenylthio“ bedeutet erfindungsgemäßt ein über ein Schwefelatom gebundenen Alkenylrest, Alkinylthio bedeutet ein über ein Schwefelatom gebundenen Alkinylrest, Cycloalkylthio bedeutet ein über ein Schwefelatom gebundenen Cycloalkylrest und Cycloalkenylthio bedeutet ein über ein Carbon atoms, such as (Ci-Cio) -, (C I -C Ö ) - or (Ci-C i) -alkylthio, for example (but not limited to) (Ci-C 6 ) -alkylthio such as methylthio, ethylthio, propylthio, 1-methylethylthio, butylthio, 1-methylpropylthio, 2-methylpropylthio, l, l-dimethylethylthio, pentylthio, 1-methylbutylthio, 2-methylbutylthio, 3-methylbutylthio, l, l-dimethylpropylthio, 1, 2-dimethylpropylthio, 2,2- Dimethylpropylthio, 1-ethylpropylthio, hexylthio, 1-methylpentylthio, 2-methylpentylthio, 3-methylpentylthio, 4-methylpentylthio, l, l-dimethylbutylthio, 1, 2-dimethylbutylthio, l, 3-dimethylbutylthio, 2,2-dimethylbutylthio thio, 2,3-dimethylbutylthio, 3,3-dimethylbutylthio, 1-ethylbutylthio, 2-ethylbutylthio, l, l, 2-trimethylpropylthio, 1, 2,2-trimethylpropylthio, 1-ethyl-1-methylpropylthio and 1- Ethyl 2-methylpropylthio. According to the invention, “alkenylthio” means an alkenyl radical bonded via a sulfur atom, alkynylthio means an alkynyl radical bonded via a sulfur atom, cycloalkylthio means a cycloalkyl radical bonded via a sulfur atom and cycloalkenylthio means one via a
Schwefelatom gebundenen Cycloalkenylrest. Sulfur atom-bonded cycloalkenyl radical.
„Alkylsulfinyl (Alkyl-S(=0)-)“, soweit nicht an anderer Stelle anders definiert steht erfindungsgemäß für Alkylreste, die über -S(=0)- an das Gerüst gebunden sind, wie (Ci-Cio)-, (Ci-Cr,)- oder (Ci-C4)- Alkylsulfinyl, z. B. (aber nicht beschränkt auf) (Ci-C6)-Alkylsulfinyl wie Methylsulfinyl, Ethylsulfinyl, Propylsulfinyl, 1 -Methylethylsulfinyl, Butylsulfinyl, 1 -Methylpropylsulfinyl, 2-Methylpropylsulfinyl,"Alkylsulfinyl (alkyl-S (= 0) -)", unless otherwise defined elsewhere, according to the invention stands for alkyl radicals which are bonded to the structure via -S (= 0) -, such as (Ci-Cio) -, ( Ci-Cr,) - or (Ci-C 4 ) - alkylsulfinyl, e.g. B. (but not limited to) (Ci-C 6 ) alkylsulfinyl such as methylsulfinyl, ethylsulfinyl, propylsulfinyl, 1-methylethylsulfinyl, butylsulfinyl, 1-methylpropylsulfinyl, 2-methylpropylsulfinyl,
1 , 1 -Dimethylethylsulfinyl, Pentylsulfinyl, 1 -Methylbutylsulfinyl, 2-Methylbutylsulfinyl, 3- Methylbutylsulfinyl, l,l-Dimethylpropylsulfinyl, 1 ,2-Dimethylpropylsulfinyl, 2,2-Di- methylpropylsulfinyl, 1 -Ethylpropylsulfinyl, Hexylsulfinyl, 1 -Methylpentylsulfinyl, 2-Methylpentyl- sulfinyl, 3 -Methylpentylsulfinyl, 4-Methylpentylsulfinyl, l,l-Dimethylbutylsulfinyl, 1 ,2-Dimethyl- butylsulfinyl, l,3-Dimethylbutylsulfinyl, 2,2-Dimethylbutylsulfinyl, 2,3-Dimethylbutylsulfinyl, 3,3- Dimethylbutylsulfinyl, 1 -Ethylbutylsulfinyl, 2-Ethylbutylsulfinyl, l,l,2-Trimethylpropylsulfinyl, 1,2,2- Trimethylpropylsulfinyl, l-Ethyl-l -methylpropylsulfinyl und 1 -Ethyl-2-methylpropylsulfinyl. 1, 1-dimethylethylsulfinyl, pentylsulfinyl, 1-methylbutylsulfinyl, 2-methylbutylsulfinyl, 3-methylbutylsulfinyl, l, l-dimethylpropylsulfinyl, 1, 2-dimethylpropylsulfinyl, 2,2-dimethylpropylsulfinyl, 1-ethylpropylsulfinyl, 1 2-methylpentylsulfinyl, 3-methylpentylsulfinyl, 4-methylpentylsulfinyl, l, l-dimethylbutylsulfinyl, 1, 2-dimethylbutylsulfinyl, l, 3-dimethylbutylsulfinyl, 2,2-dimethylbutylsulfinyl, 2,3-dimethylbutylsulfinyl, 3,3- Dimethylbutylsulfinyl, 1-ethylbutylsulfinyl, 2-ethylbutylsulfinyl, l, l, 2-trimethylpropylsulfinyl, 1,2,2-trimethylpropylsulfinyl, l-ethyl-l-methylpropylsulfinyl and 1-ethyl-2-methylpropylsulfinyl.
Analog sind„Alkenylsulfinyl“ und„Alkinylsulfinyl“, erfindungsgemäß definiert als Alkenyl- bzw. Alkinylreste, die über -S(=0)- an das Gerüst gebunden sind, wie (C2-C10)-, (C2-C6)- oder (C2-C4)- Alkenylsulfinyl bzw. (C3-C10)-, (C3-C6)- oder (C3-C4)-Alkinylsulfinyl. Analogous are “alkenylsulfinyl” and “alkynylsulfinyl”, defined according to the invention as alkenyl or alkynyl radicals which are bonded to the structure via -S (= 0) -, such as (C2-C10) -, (C2-C6) - or ( C2-C4) - alkenylsulfinyl or (C3-C10) -, (C3-C6) - or (C3-C 4 ) -alkynylsulfinyl.
Analog sind„Alkenylsulfonyl“ und„Alkinylsulfonyl“ erfindungsgemäß definiert als Alkenyl- bzw. Alkinylreste, die über -S(=0)2- an das Gerüst gebunden sind, wie (C2-C10)-, (C2-C6)- oder (C2-C4)- Alkenylsulfonyl bzw. (C3-C10)-, (C3-C6)- oder (C3-C4)-Alkinylsulfonyl. Analogously, “alkenylsulfonyl” and “alkynylsulfonyl” are defined according to the invention as alkenyl or alkynyl radicals which are bonded to the structure via -S (= 0) 2 -, such as (C2-C10) -, (C2-C6) - or ( C2-C4) - alkenylsulfonyl or (C3-C10) -, (C3-C6) - or (C3-C 4 ) -alkynylsulfonyl.
„Alkoxy“ bedeutet ein über ein Sauerstoffatom gebundenen Alkylrest, z. B. (aber nicht beschränkt auf) (Ci-C6)-Alkoxy wie Methoxy, Ethoxy, Propoxy, 1 -Methylethoxy, Butoxy, 1 -Methylpropoxy, 2- Methylpropoxy, l,l-Dimethylethoxy, Pentoxy, 1 -Methylbutoxy, 2-Methylbutoxy, 3-Methylbutoxy, 1,1- Dimethylpropoxy, 1 ,2-Dimethylpropoxy, 2,2-Dimethylpropoxy, 1 -Ethylpropoxy, Hexoxy, 1 - Methylpentoxy, 2-Methylpentoxy, 3-Methylpentoxy, 4-Methylpentoxy, l,l-Dimethylbutoxy, l,2-Di- methylbutoxy, l,3-Dimethylbutoxy, 2,2-Dimethylbutoxy, 2,3-Dimethylbutoxy, 3,3-Dimethylbutoxy, 1- Ethylbutoxy, 2-Ethylbutoxy, l,l,2-Trimethylpropoxy, 1 ,2,2-Trimethylpropoxy, l-Ethyl-l -methyl propoxy und 1 -Ethyl-2-methylpropoxy. Alkenyloxy bedeutet ein über ein Sauerstoffatom gebundenen Alkenylrest, Alkinyloxy bedeutet ein über ein Sauerstoffatom gebundenen Alkinylrest wie (C2-C10)-, (C2-C6)- oder (C2-C4)-Alkenoxy bzw. (C3-C10)-, (C3-C6)- oder (C3-C4)-Alkinoxy. „Cycloalkyloxy“ bedeutet ein über ein Sauerstoffatom gebundenen Cycloalkylrest und Cycloalkenyloxy bedeutet ein über ein Sauerstoffatom gebundenen Cycloalkenylrest. “Alkoxy” means an alkyl radical bonded via an oxygen atom, eg. B. (but not limited to) (Ci-C 6 ) alkoxy such as methoxy, ethoxy, propoxy, 1-methylethoxy, butoxy, 1-methylpropoxy, 2-methylpropoxy, l, l-dimethylethoxy, pentoxy, 1-methylbutoxy, 2 -Methylbutoxy, 3-methylbutoxy, 1,1-dimethylpropoxy, 1, 2-dimethylpropoxy, 2,2-dimethylpropoxy, 1-ethylpropoxy, hexoxy, 1 - methylpentoxy, 2-methylpentoxy, 3-methylpentoxy, 4-methylpentoxy, l, l -Dimethylbutoxy, l, 2-dimethylbutoxy, l, 3-dimethylbutoxy, 2,2-dimethylbutoxy, 2,3-dimethylbutoxy, 3,3-dimethylbutoxy, 1-ethylbutoxy, 2-ethylbutoxy, l, l, 2-trimethylpropoxy , 1, 2,2-trimethylpropoxy, l-ethyl-l-methyl propoxy and 1-ethyl-2-methylpropoxy. Alkenyloxy means an alkenyl radical bonded via an oxygen atom, alkynyloxy means an alkynyl radical bonded via an oxygen atom, such as (C2-C10) -, (C2-C6) - or (C2-C 4 ) -alkenoxy or (C3-C10) -, ( C3-C6) or (C3-C 4 ) alkynoxy. “Cycloalkyloxy” means a cycloalkyl radical bonded via an oxygen atom and cycloalkenyloxy means a cycloalkenyl radical bonded via an oxygen atom.
„Alkylcarbonyl“ (Alkyl-C(=0)-), soweit nicht an anderer Stelle anders definiert, steht erfindungsgemäß für Alkylreste, die über -C(=0)- an das Gerüst gebunden sind, wie (Ci-Cio)-, (Ci-Cr,)- oder (Ci-C4)- Alkylcarbonyl. Die Anzahl der C-Atome bezieht sich dabei auf den Alkylrest in der "Alkylcarbonyl" (alkyl-C (= 0) -), unless otherwise defined elsewhere, stands according to the invention for alkyl radicals which are bonded to the structure via -C (= 0) -, such as (Ci-Cio) -, (Ci-Cr,) - or (Ci-C 4 ) - alkylcarbonyl. The number of carbon atoms refers to the alkyl radical in the
Alkylcarbonylgruppe. Alkylcarbonyl.
Analog stehen„Alkenylcarbonyl“ und„Alkinylcarbonyl“, soweit nicht an anderer Stelle anders definiert, erfindungsgemäß für Alkenyl- bzw. Alkinylreste, die über -C(=0)- an das Gerüst gebunden sind, wie (C2-C 10)-, (C2-C6)- oder (C2-C4)-Alkenylcarbonyl bzw. (C2-C 10)-, (C2-C6)- oder (C2-C4)- Alkinylcarbonyl. Die Anzahl der C-Atome bezieht sich dabei auf den Alkenyl- bzw. Alkinylrest in der Alkenyl- bzw. Alkinylcarbonylgruppe. Analogously, “alkenylcarbonyl” and “alkynylcarbonyl”, unless otherwise defined elsewhere, stand according to the invention for alkenyl or alkynyl radicals which are bonded to the structure via -C (= 0) -, such as (C2-C 10) -, (C2-C6) - or (C2-C4) -alkenylcarbonyl or (C2-C10) -, (C2-C6) - or (C2-C4) - alkynylcarbonyl. The number of carbon atoms relates to the alkenyl or alkynyl radical in the alkenyl or alkynylcarbonyl group.
„Alkoxycarbonyl (Alkyl-0-C(=0)-)“, soweit nicht an anderer Stelle anders definiert: Alkylreste, die über -0-C(=0)- an das Gerüst gebunden sind, wie (C1-C10)-, (CI-CÖ)- oder (Ci-C4)-Alkoxycarbonyl. Die Anzahl der C-Atome bezieht sich dabei auf den Alkylrest in der Alkoxycarbonylgruppe. Analog stehen „Alkenyloxycarbonyl“ und„Alkinyloxycarbonyl“, soweit nicht an anderer Stelle anders definiert, erfindungsgemäß für Alkenyl- bzw. Alkinylreste, die über -0-C(=0)- an das Gerüst gebunden sind, wie (C2-C 10)-, (C2-C6)- oder (C2-C4)-Alkenyloxycarbonyl bzw. (C3-C 10)-, (C3-C6)- oder (C3-C4)- Alkinyloxycarbonyl. Die Anzahl der C-Atome bezieht sich dabei auf den Alkenyl- bzw. Alkinylrest in der Alken- bzw. Alkinyloxycarbonylgruppe. "Alkoxycarbonyl (alkyl-0-C (= 0) -)", unless otherwise defined elsewhere: alkyl residues which are bonded to the structure via -0-C (= 0) -, such as (C1-C10) - , (C I -C Ö ) - or (Ci-C 4 ) alkoxycarbonyl. The number of carbon atoms relates to the alkyl radical in the alkoxycarbonyl group. Analogously, "alkenyloxycarbonyl" and "alkynyloxycarbonyl", unless otherwise defined elsewhere, according to the invention stand for alkenyl or alkynyl radicals which are bonded to the structure via -0-C (= 0) -, such as (C2-C 10) -, (C2-C6) - or (C2-C 4 ) alkenyloxycarbonyl or (C3-C 10) -, (C3-C6) - or (C3-C4) - alkynyloxycarbonyl. The number of carbon atoms relates to the alkenyl or alkynyl radical in the alkene or alkynyloxycarbonyl group.
Der Begriff„Alkylcarbonyloxy“ (Alkyl-C(=0)-0-) steht erfindungsgemäß, soweit nicht an anderer Stelle anders definiert, für Alkylreste, die über eine Carbonyloxygruppe (-C(=0)-0-) mit dem Sauerstoff an das Gerüst gebunden sind, wie (C1-C10)-, (CI-CÖ)- oder (Ci-C4)-Alkylcarbonyloxy. Die Anzahl der C- Atome bezieht sich dabei auf den Alkylrest in der Alkylcarbonyloxygruppe. The term “alkylcarbonyloxy” (alkyl-C (= 0) -0-) according to the invention, unless otherwise defined elsewhere, stands for alkyl radicals which are present with the oxygen via a carbonyloxy group (-C (= 0) -0-) the backbone are bound, such as (C1-C10) -, (C I -C Ö ) - or (Ci-C 4 ) -alkylcarbonyloxy. The number of carbon atoms relates to the alkyl radical in the alkylcarbonyloxy group.
Analog sind„Alkenylcarbonyloxy“ und„Alkinylcarbonyloxy“ erfindungsgemäß definiert als Alkenyl- bzw. Alkinylreste, die über (-C(=0)-0-) mit dem Sauerstoff an das Gerüst gebunden sind, wie (C2-C10)-, (C2-C6)- oder (C2-C4)-Alkenylcarbonyloxy bzw. (C2-C10)-, (C2-C6)- oder (C2-C4)-Alkinylcarbonyloxy. Die Anzahl der C-Atome bezieht sich dabei auf den Alkenyl- bzw. Alkinylrest in der Alkenyl- bzw. Alkinylcarbonyloxygruppe. ln Kurzformen wie z.B. C(0)R5, C(0)OR5, 0C(0)NR6R7, oder C(0)NR6R7 steht die in Klammem aufgeführte Kurzform O für ein über eine Doppelbindung an das benachbarte Kohlenstoffatom gebundenes Sauerstoffatom. In Kurzformen wie z.B. OC(S)OR6, OC(S)SR9, OC(S)NR6R7, steht die in Klammem aufgeführte Kurzform S für ein über eine Doppelbindung an das benachbarte Kohlenstoffatom gebundenes Analogously, “alkenylcarbonyloxy” and “alkynylcarbonyloxy” are defined according to the invention as alkenyl or alkynyl radicals which are bonded to the structure via (-C (= 0) -0-) with the oxygen, such as (C2-C10) -, (C 2 -C 6 ) - or (C2-C 4 ) -alkenylcarbonyloxy or (C2-C10) -, (C2-C 6 ) - or (C2-C 4 ) -alkynylcarbonyloxy. The number of carbon atoms relates to the alkenyl or alkynyl radical in the alkenyl or alkynylcarbonyloxy group. In short forms such as C (0) R 5 , C (0) OR 5 , 0C (0) NR 6 R 7 , or C (0) NR 6 R 7 , the short form O in brackets stands for a via a double bond to the adjacent carbon atom bound oxygen atom. In short forms such as OC (S) OR 6 , OC (S) SR 9 , OC (S) NR 6 R 7 , the short form S shown in brackets stands for one bonded to the adjacent carbon atom via a double bond
Schwefelatom. Sulfur atom.
Der Begriff„Aryl“ bedeutet ein gegebenenfalls substituiertes mono-, bi- oder polycyclisches aromatisches System mit vorzugsweise 6 bis 14, insbesondere 6 bis 10 Ring-C- Atomen, beispielsweise Phenyl, Naphthyl, Anthryl, Phenanthrenyl, und ähnliches, vorzugsweise Phenyl. The term “aryl” means an optionally substituted mono-, bi- or polycyclic aromatic system with preferably 6 to 14, in particular 6 to 10 ring carbon atoms, for example phenyl, naphthyl, anthryl, phenanthrenyl, and the like, preferably phenyl.
Vom Begriff„gegebenenfalls substituiertes Aryl“ sind auch mehrcyclische Systeme, wie The term “optionally substituted aryl” also includes multi-cyclic systems such as
Tetrahydronaphtyl, lndenyl, lndanyl, Fluorenyl, Biphenylyl, umfasst, wobei die Bindungsstelle am aromatischen System ist. Von der Systematik her ist„Aryl“ in der Regel auch von dem Begriff „gegebenenfalls substituiertes Phenyl“ umfasst. Bevorzugte Aryl-Substituenten sind hier zum Beispiel Wasserstoff, Halogen, Alkyl, Cycloalkyl, Cycloalkylalkyl, Cycloalkenyl, Halocycloalkyl, Alkenyl, Alkinyl, Aryl, Arylalkyl, Arylalkenyl, Heteroaryl, Heteroarylalkyl, Heterocyclyl, Heterocyclylalkyl, Alkoxyalkyl, Alkylthio, Haloalkylthio, Haloalkyl, Alkoxy, Haloalkoxy, Cycloalkoxy, Cycloalkylalkoxy, Aryloxy, Heteroraryloxy, Alkoxyalkoxy, Alkinylalkoxy, Alkenyloxy, Bis-alkylaminoalkoxy, Tris- [alkyl] silyl, Bis-[alkyl]arylsilyl, Bis-[alkyl]alkylsilyl, Tris-[alkyl]silylalkinyl, Arylalkinyl, Tetrahydronaphthyl, indyl, indanyl, fluorenyl, biphenylyl, wherein the binding site is on the aromatic system. Systematically, “aryl” is usually also included in the term “optionally substituted phenyl”. Preferred aryl substituents here are, for example, hydrogen, halogen, alkyl, cycloalkyl, cycloalkylalkyl, cycloalkenyl, halocycloalkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, heteroaryl, heteroarylalkyl, heterocyclyl, heterocyclylalkyl, alkoxyalkyl, alkylthio, haloalkyl, haloalkyl, haloalkyl Haloalkoxy, cycloalkoxy, cycloalkylalkoxy, aryloxy, heteroraryloxy, alkoxyalkoxy, alkynylalkoxy, alkenyloxy, bis-alkylaminoalkoxy, tris [alkyl] silyl, bis [alkyl] arylsilyl, bis [alkyl] alkylsilyl, tris [alkyl] silylalkynyl, arylalkynyl,
Heteroarylalkinyl, Alkylalkinyl, Cycloalkylalkinyl, Haloalkylalkinyl, Heterocyclyl-N-alkoxy, Nitro, Cyano, Amino, Alkylamino, Bis-alkylamino, Alkylcarbonylamino, Cycloalkylcarbonylamino, Heteroarylalkynyl, alkylalkynyl, cycloalkylalkynyl, haloalkylalkynyl, heterocyclyl-N-alkoxy, nitro, cyano, amino, alkylamino, bis-alkylamino, alkylcarbonylamino, cycloalkylcarbonylamino,
Arylcarbonylamino, Alkoxycarbonylamino, Alkoxycarbonylalkylamino, Arylcarbonylamino, alkoxycarbonylamino, alkoxycarbonylalkylamino,
Arylalkoxycarbonylalkylamino, Hydroxycarbonyl, Alkoxycarbonyl, Aminocarbonyl, Arylalkoxycarbonylalkylamino, hydroxycarbonyl, alkoxycarbonyl, aminocarbonyl,
Alkylaminocarbonyl, Cycloalkylaminocarbonyl, Bis-Alkylaminocarbonyl, Heteroarylalkoxy, Alkylaminocarbonyl, cycloalkylaminocarbonyl, bis-alkylaminocarbonyl, heteroarylalkoxy,
Arylalkoxy. Arylalkoxy.
Ein heterocyclischer Rest (Heterocyclyl) enthält mindestens einen heterocyclischen Ring A heterocyclic radical (heterocyclyl) contains at least one heterocyclic ring
(=carbocyclischer Ring, in dem mindestens ein C-Atom durch ein Heteroatom ersetzt ist, vorzugsweise durch ein Heteroatom aus der Gruppe N, O, S, P) der gesättigt, ungesättigt, teilgesättigt oder heteroaromatisch ist und dabei unsubstituiert oder substituiert sein kann, wobei die Bindungsstelle an einem Ringatom lokalisiert ist. lst der Heterocyclylrest oder der heterocyclische Ring gegebenenfalls substituiert, kann er mit anderen carbocyclischen oder heterocyclischen Ringen annelliert sein lm Falle von gegebenenfalls substituiertem Heterocyclyl werden auch mehrcyclische Systeme umfasst, wie beispielsweise 8-Aza-bicyclo[3.2.l]octanyl, 8-Aza-bicyclo[2.2.2]octanyl oder l-Aza- bicyclo[2.2.l]heptyl. lm Falle von gegebenenfalls substituiertem Heterocyclyl werden auch (= carbocyclic ring in which at least one C atom is replaced by a hetero atom, preferably by a hetero atom from the group N, O, S, P) which is saturated, unsaturated, partially saturated or heteroaromatic and can be unsubstituted or substituted, where the binding site is located on a ring atom. If the heterocyclyl radical or the heterocyclic ring is optionally substituted, it can be fused with other carbocyclic or heterocyclic rings. In the case of optionally substituted heterocyclyl, multi-cyclic systems are also included, such as 8-aza-bicyclo [3.2.l] octanyl, 8-aza- bicyclo [2.2.2] octanyl or l-azabicyclo [2.2.l] heptyl. In the case of optionally substituted heterocyclyl also
spirocyclische Systeme umfasst, wie beispielsweise l-Oxa-5-aza-spiro[2.3]hexyl. Wenn nicht anders definiert, enthält der heterocyclische Ring vorzugsweise 3 bis 9 Ringatome, insbesondere 3 bis 6 Ringatome, und ein oder mehrere, vorzugsweise 1 bis 4, insbesondere 1 , 2 oder 3 Heteroatome im heterocyclischen Ring, vorzugsweise aus der Gruppe N, O, und S, wobei jedoch nicht zwei includes spirocyclic systems such as l-oxa-5-aza-spiro [2.3] hexyl. Unless otherwise defined, the heterocyclic ring preferably contains 3 to 9 ring atoms, in particular 3 to 6 ring atoms, and one or more, preferably 1 to 4, in particular 1, 2 or 3, heteroatoms in the heterocyclic ring, preferably from the group N, O, and S, but not two
Sauerstoffatome direkt benachbart sein sollen, wie beispielsweise mit einem Heteroatom aus der Gruppe N, O und S 1- oder 2- oder 3-Pyrrolidinyl, 3,4-Dihydro-2H-pyrrol-2- oder 3-yl, 2,3-Dihydro-lH-pyrrol-Oxygen atoms are said to be directly adjacent, for example with a heteroatom from the group N, O and S 1- or 2- or 3-pyrrolidinyl, 3,4-dihydro-2H-pyrrol-2- or 3-yl, 2,3- dihydro-lH-pyrrole
1- oder 2- oder 3- oder 4- oder 5-yl; 2,5-Dihydro-lH-pyrrol-l- oder 2- oder 3-yl, 1- oder 2- oder 3- oder 4-Piperidinyl; 2,3,4,5-Tetrahydropyridin-2- oder 3- oder 4- oder 5-yl oder 6-yl; 1 ,2,3,6- Tetrahydropyridin-l- oder 2- oder 3- oder 4- oder 5- oder 6-yl; l,2,3,4-Tetrahydropyridin-l- oder 2- oder 3- oder 4- oder 5- oder 6-yl; l,4-Dihydropyridin-l- oder 2- oder 3- oder 4-yl; 2,3-Dihydropyridin-1- or 2- or 3- or 4- or 5-yl; 2,5-dihydro-1H-pyrrole-1- or 2- or 3-yl, 1- or 2- or 3- or 4-piperidinyl; 2,3,4,5-tetrahydropyridin-2- or 3- or 4- or 5-yl or 6-yl; 1, 2,3,6-tetrahydropyridin-l- or 2- or 3- or 4- or 5- or 6-yl; l, 2,3,4-tetrahydropyridin-l- or 2- or 3- or 4- or 5- or 6-yl; l, 4-dihydropyridin-l- or 2- or 3- or 4-yl; 2,3-dihydropyridine
2- oder 3- oder 4- oder 5- oder 6-yl; 2,5-Dihydropyridin-2- oder 3- oder 4- oder 5- oder 6-yl, 1- oder 2- oder 3- oder 4-Azepanyl; 2,3,4,5-Tetrahydro-lH-azepin-l- oder 2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3,4,7-Tetrahydro-lH-azepin-l- oder 2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2, 3, 6, 7- Tetrahydro-lH-azepin-l- oder 2- oder 3- oder 4-yl; 3,4,5,6-Tetrahydro-2H-azepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 4,5-Dihydro-lH-azepin-l- oder 2- oder 3- oder 4-yl; 2,5-Dihydro-lH-azepin-2- or 3- or 4- or 5- or 6-yl; 2,5-dihydropyridin-2- or 3- or 4- or 5- or 6-yl, 1- or 2- or 3- or 4-azepanyl; 2,3,4,5-tetrahydro-lH-azepine-1- or 2- or 3- or 4- or 5- or 6- or 7-yl; 2,3,4,7-tetrahydro-lH-azepine-1- or 2- or 3- or 4- or 5- or 6- or 7-yl; 2, 3, 6, 7-tetrahydro-lH-azepine-l- or 2- or 3- or 4-yl; 3,4,5,6-tetrahydro-2H-azepin-2- or 3- or 4- or 5- or 6- or 7-yl; 4,5-dihydro-lH-azepine-l- or 2- or 3- or 4-yl; 2,5-dihydro-lH-azepin-
1- oder -2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,7-Dihydro-lH-azepin-l- oder -2- oder 3- oder 4- yl; 2,3-Dihydro-lH-azepin-l- oder -2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 3,4-Dihydro-2H-azepin-1- or -2- or 3- or 4- or 5- or 6- or 7-yl; 2,7-dihydro-lH-azepine-l- or -2- or 3- or 4-yl; 2,3-dihydro-lH-azepine-l- or -2- or 3- or 4- or 5- or 6- or 7-yl; 3,4-dihydro-2H-azepine
2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 3,6-Dihydro-2H-azepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 5,6-Dihydro-2H-azepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 4,5-Dihydro-3H-azepin-2- or 3- or 4- or 5- or 6- or 7-yl; 3,6-dihydro-2H-azepin-2- or 3- or 4- or 5- or 6- or 7-yl; 5,6-dihydro-2H-azepin-2- or 3- or 4- or 5- or 6- or 7-yl; 4,5-dihydro-3H-azepine
2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; lH-Azepin-l- oder -2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2H-Azepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 3H-Azepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 4H-Azepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl, 2- oder 3-Oxolanyl (= 2- oder 3- Tetrahydrofuranyl); 2,3-Dihydrofuran-2- oder 3- oder 4- oder 5-yl; 2,5-Dihydrofuran-2- oder 3-yl, 2- oder 3- oder 4-Oxanyl (= 2- oder 3- oder 4-Tetrahydropyranyl); 3,4-Dihydro-2H-pyran-2- oder 3- oder 4- oder 5- oder 6-yl; 3,6-Dihydro-2H-pyran-2- oder 3-oder 4- oder 5- oder 6-yl; 2H-Pyran-2- oder 3- oder 4- oder 5- oder 6-yl; 4H-Pyran-2- oder 3- oder 4-yl, 2- oder 3- oder 4-Oxepanyl; 2, 3,4,5- Tetrahydrooxepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3,4,7-Tetrahydrooxepin-2- oder 3- oder2- or 3- or 4- or 5- or 6- or 7-yl; 1H-azepine-1- or -2- or 3- or 4- or 5- or 6- or 7-yl; 2H-azepin-2- or 3- or 4- or 5- or 6- or 7-yl; 3H-azepin-2- or 3- or 4- or 5- or 6- or 7-yl; 4H-azepin-2- or 3- or 4- or 5- or 6- or 7-yl, 2- or 3-oxolanyl (= 2- or 3-tetrahydrofuranyl); 2,3-dihydrofuran-2- or 3- or 4- or 5-yl; 2,5-dihydrofuran-2- or 3-yl, 2- or 3- or 4-oxanyl (= 2- or 3- or 4-tetrahydropyranyl); 3,4-dihydro-2H-pyran-2- or 3- or 4- or 5- or 6-yl; 3,6-dihydro-2H-pyran-2- or 3- or 4- or 5- or 6-yl; 2H-pyran-2- or 3- or 4- or 5- or 6-yl; 4H-pyran-2- or 3- or 4-yl, 2- or 3- or 4-oxepanyl; 2, 3,4,5-tetrahydrooxepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,3,4,7-tetrahydrooxepin-2 or 3 or
4- oder 5- oder 6- oder 7-yl; 2,3,6,7-Tetrahydrooxepin-2- oder 3- oder 4-yl; 2,3-Dihydrooxepin-2- oder4- or 5- or 6- or 7-yl; 2,3,6,7-tetrahydrooxepin-2- or 3- or 4-yl; 2,3-dihydrooxepin-2 or
3- oder 4- oder 5- oder 6- oder 7-yl; 4,5-Dihydrooxepin-2- oder 3- oder 4-yl; 2,5-Dihydrooxepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; Oxepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2- oder 3- Tetrahydrothiophenyl; 2,3-Dihydrothiophen-2- oder 3- oder 4- oder 5-yl; 2,5-Dihydrothiophen-2- oder 3-yl; Tetrahydro-2H-thiopyran-2- oder 3- oder 4-yl; 3,4-Dihydro-2H-thiopyran-2- oder 3- oder 4- oder3- or 4- or 5- or 6- or 7-yl; 4,5-dihydrooxepin-2- or 3- or 4-yl; 2,5-dihydrooxepin-2- or 3- or 4- or 5- or 6- or 7-yl; Oxepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2- or 3-tetrahydrothiophenyl; 2,3-dihydrothiophene-2- or 3- or 4- or 5-yl; 2,5-dihydrothiophene-2- or 3-yl; Tetrahydro-2H-thiopyran-2- or 3- or 4-yl; 3,4-dihydro-2H-thiopyran-2- or 3- or 4- or
5- oder 6-yl; 3,6-Dihydro-2H-thiopyran-2- oder 3- oder 4- oder 5- oder 6-yl; 2H-Thiopyran-2- oder 3- oder 4- oder 5- oder 6-yl; 4H-Thiopyran-2- oder 3- oder 4-yl. Bevorzugte 3-Ring und 4-Ring- Heterocyclen sind beispielsweise 1- oder 2-Aziridinyl, Oxiranyl, Thiiranyl, 1- oder 2- oder 3-Azetidinyl,5- or 6-yl; 3,6-dihydro-2H-thiopyran-2- or 3- or 4- or 5- or 6-yl; 2H-thiopyran-2- or 3- or 4- or 5- or 6-yl; 4H-thiopyran-2- or 3- or 4-yl. Preferred 3-ring and 4-ring heterocycles are, for example, 1- or 2-aziridinyl, oxiranyl, thiiranyl, 1- or 2- or 3-azetidinyl,
2- oder 3-Oxetanyl, 2- oder 3-Thietanyl, l,3-Dioxetan-2-yl. Weitere Beispiele für“Heterocyclyl“ sind ein partiell oder vollständig hydrierter heterocyclischer Rest mit zwei Heteroatomen aus der Gruppe N, O und S, wie beispielsweise 1- oder 2- oder 3- oder 4-Pyrazolidinyl; 4,5-Dihydro-3H-pyrazol- 3- oder 4- oder 5-yl; 4,5-Dihydro-lH-pyrazol-l- oder 3- oder 4- oder 5-yl; 2,3-Dihydro-lH-pyrazol-l- oder 2- oder2- or 3-oxetanyl, 2- or 3-thietanyl, l, 3-dioxetan-2-yl. Further examples of “heterocyclyl” are a partially or fully hydrogenated heterocyclic radical with two heteroatoms from the group N, O and S, such as 1- or 2- or 3- or 4-pyrazolidinyl; 4,5-dihydro-3H-pyrazol-3- or 4- or 5-yl; 4,5-dihydro-1H-pyrazole-l- or 3- or 4- or 5-yl; 2,3-dihydro-1H-pyrazole-1 or 2- or
3- oder 4- oder 5-yl; 1- oder 2- oder 3- oder 4- Imidazolidinyl; 2,3-Dihydro-lH-imidazol-l- oder 2- oder 3- oder 4-yl; 2,5-Dihydro-lH-imidazol-l- oder 2- oder 4- oder 5-yl; 4,5-Dihydro-lH-imidazol-l- oder 2- oder 4- oder 5-yl; Hexahydropyridazin-l- oder 2- oder 3- oder 4-yl; l,2,3,4-Tetrahydropyridazin-l- oder 2- oder 3- oder 4- oder 5- oder 6-yl; l,2,3,6-Tetrahydropyridazin-l- oder 2- oder 3- oder 4- oder 5- oder 6-yl; l,4,5,6-Tetrahydropyridazin-l- oder 3- oder 4- oder 5- oder 6-yl; 3,4,5,6-Tetrahydropyridazin-3- oder 4- oder 5-yl; 4,5-Dihydropyridazin-3- oder 4-yl; 3,4-Dihydropyridazin-3- oder 4- oder 5- oder 6-yl; 3,6-Dihydropyridazin-3- oder 4-yl; l,6-Dihydropyriazin-l- oder 3- oder 4- oder 5- oder 6-yl; 3- or 4- or 5-yl; 1- or 2- or 3- or 4-imidazolidinyl; 2,3-dihydro-lH-imidazole-l- or 2- or 3- or 4-yl; 2,5-dihydro-1H-imidazole-1- or 2- or 4- or 5-yl; 4,5-dihydro-1H-imidazole-1- or 2- or 4- or 5-yl; Hexahydropyridazin-l- or 2- or 3- or 4-yl; l, 2,3,4-tetrahydropyridazin-l- or 2- or 3- or 4- or 5- or 6-yl; l, 2,3,6-tetrahydropyridazin-l- or 2- or 3- or 4- or 5- or 6-yl; l, 4,5,6-tetrahydropyridazin-l- or 3- or 4- or 5- or 6-yl; 3,4,5,6-tetrahydropyridazin-3- or 4- or 5-yl; 4,5-dihydropyridazin-3- or 4-yl; 3,4-dihydropyridazin-3- or 4- or 5- or 6-yl; 3,6-dihydropyridazin-3- or 4-yl; l, 6-dihydropyriazin-l- or 3- or 4- or 5- or 6-yl;
Hexahydropyrimidin-l- oder 2- oder 3- oder 4-yl; l,4,5,6-Tetrahydropyrimidin-l- oder 2- oder 4- oder 5- oder 6-yl; l,2,5,6-Tetrahydropyrimidin-l- oder 2- oder 4- oder 5- oder 6-yl; 1 ,2,3,4- Tetrahydropyrimidin-l- oder 2- oder 3- oder 4- oder 5- oder 6-yl; l,6-Dihydropyrimidin-l- oder 2- oderHexahydropyrimidin-l- or 2- or 3- or 4-yl; l, 4,5,6-tetrahydropyrimidin-l- or 2- or 4- or 5- or 6-yl; l, 2,5,6-tetrahydropyrimidin-l- or 2- or 4- or 5- or 6-yl; 1, 2,3,4-tetrahydropyrimidin-l- or 2- or 3- or 4- or 5- or 6-yl; l, 6-dihydropyrimidine-l- or 2- or
4- oder 5- oder 6-yl; l,2-Dihydropyrimidin-l- oder 2- oder 4- oder 5- oder 6-yl; 2,5-Dihydropyrimidin-4- or 5- or 6-yl; l, 2-dihydropyrimidin-l- or 2- or 4- or 5- or 6-yl; 2,5-dihydropyrimidine
2- oder 4- oder 5-yl; 4,5-Dihydropyrimidin- 4- oder 5- oder 6-yl; l,4-Dihydropyrimidin-l- oder 2- oder 4- oder 5- oder 6-yl; 1- oder 2- oder 3-Piperazinyl; l,2,3,6-Tetrahydropyrazin-l- oder 2- oder 3- oder 5- oder 6-yl; l,2,3,4-Tetrahydropyrazin-l- oder 2- oder 3- oder 4- oder 5- oder 6-yl; l,2-Dihydropyrazin-l- oder 2- oder 3- oder 5- oder 6-yl; l,4-Dihydropyrazin-l- oder 2- oder 3-yl; 2,3-Dihydropyrazin-2- oder2- or 4- or 5-yl; 4,5-dihydropyrimidin-4- or 5- or 6-yl; l, 4-dihydropyrimidin-l- or 2- or 4- or 5- or 6-yl; 1- or 2- or 3-piperazinyl; l, 2,3,6-tetrahydropyrazine-1- or 2- or 3- or 5- or 6-yl; l, 2,3,4-tetrahydropyrazine-1- or 2- or 3- or 4- or 5- or 6-yl; l, 2-dihydropyrazine-1- or 2- or 3- or 5- or 6-yl; l, 4-dihydropyrazine-1- or 2- or 3-yl; 2,3-dihydropyrazine-2- or
3- oder 5- oder 6-yl; 2,5-Dihydropyrazin-2- oder 3-yl; l,3-Dioxolan-2- oder 4- oder 5-yl; l,3-Dioxol-2- oder 4-yl; l,3-Dioxan-2- oder 4- oder 5-yl; 4H-l,3-Dioxin-2- oder 4- oder 5- oder 6-yl; l,4-Dioxan-2- oder 3- oder 5- oder 6-yl; 2,3-Dihydro-l,4-dioxin-2- oder 3- oder 5- oder 6-yl; l,4-Dioxin-2- oder 3-yl;3- or 5- or 6-yl; 2,5-dihydropyrazin-2- or 3-yl; l, 3-dioxolan-2- or 4- or 5-yl; l, 3-dioxol-2- or 4-yl; l, 3-dioxan-2- or 4- or 5-yl; 4H-l, 3-dioxin-2- or 4- or 5- or 6-yl; l, 4-dioxan-2- or 3- or 5- or 6-yl; 2,3-dihydro-l, 4-dioxin-2- or 3- or 5- or 6-yl; 1,4-dioxin-2- or 3-yl;
1.2-Dithiolan-3- oder 4-yl; 3H-l,2-Dithiol-3- oder 4- oder 5-yl; l,3-Dithiolan-2- oder 4-yl; l,3-Dithiol- 2- oder 4-yl; l,2-Dithian-3- oder 4-yl; 3,4-Dihydro-l,2-dithiin-3- oder 4- oder 5- oder 6-yl; 3,6-Dihydro-1,2-dithiolan-3- or 4-yl; 3H-l, 2-dithiol-3- or 4- or 5-yl; l, 3-dithiolan-2- or 4-yl; l, 3-dithiol-2- or 4-yl; 1,2-dithian-3- or 4-yl; 3,4-dihydro-l, 2-dithiin-3- or 4- or 5- or 6-yl; 3,6-dihydro-
1.2-dithiin-3- oder 4-yl; l,2-Dithiin-3- oder 4-yl; l,3-Dithian-2- oder 4- oder 5-yl; 4H-l,3-Dithiin-2- oder 4- oder 5- oder 6-yl; Isoxazolidin-2- oder 3- oder 4- oder 5-yl; 2,3-Dihydroisoxazol-2- oder 3- oder1,2-dithiin-3- or 4-yl; 1,2-dithiin-3- or 4-yl; l, 3-dithian-2- or 4- or 5-yl; 4H-l, 3-dithiin-2- or 4- or 5- or 6-yl; Isoxazolidin-2- or 3- or 4- or 5-yl; 2,3-dihydroisoxazole-2- or 3- or
4- oder 5-yl; 2,5-Dihydroisoxazol-2- oder 3- oder 4- oder 5-yl; 4,5-Dihydroisoxazol-3- oder 4- oder 5-yl;4- or 5-yl; 2,5-dihydroisoxazol-2- or 3- or 4- or 5-yl; 4,5-dihydroisoxazol-3- or 4- or 5-yl;
1.3-Oxazolidin-2- oder 3- oder 4- oder 5-yl; 2,3-Dihydro-l,3-oxazol-2- oder 3- oder 4- oder 5-yl; 2,5- Dihydro-l,3-oxazol-2- oder 4- oder 5-yl; 4,5-Dihydro-l,3-oxazol-2- oder 4- oder 5-yl; l,2-Oxazinan-2- oder 3- oder 4- oder 5- oder 6-yl; 3,4-Dihydro-2H-l,2-oxazin-2- oder 3- oder 4- oder 5- oder 6-yl; 3,6- Dihydro-2H-l,2-oxazin-2- oder 3- oder 4- oder 5- oder 6-yl; 5,6-Dihydro-2H-l,2-oxazin-2- oder 3- oder 4- oder 5- oder 6-yl; 5,6-Dihydro-4H-l,2-oxazin-3- oder 4- oder 5- oder 6-yl; 2H-l,2-Oxazin-2- oder 3- oder 4- oder 5- oder 6-yl; 6H-l,2-Oxazin-3- oder 4- oder 5- oder 6-yl; 4H-l,2-Oxazin-3- oder 4- oder 5- oder 6-yl; l,3-Oxazinan-2- oder 3- oder 4- oder 5- oder 6-yl; 3,4-Dihydro-2H-l,3-oxazin-2- oder 3- oder 4- oder 5- oder 6-yl; 3,6-Dihydro-2H-l,3-oxazin-2- oder 3- oder 4- oder 5- oder 6-yl; 5,6-Dihydro-2H-1,3-oxazolidin-2- or 3- or 4- or 5-yl; 2,3-dihydro-l, 3-oxazol-2- or 3- or 4- or 5-yl; 2,5-dihydro-l, 3-oxazol-2- or 4- or 5-yl; 4,5-dihydro-l, 3-oxazol-2- or 4- or 5-yl; l, 2-oxazinan-2- or 3- or 4- or 5- or 6-yl; 3,4-dihydro-2H-l, 2-oxazin-2- or 3- or 4- or 5- or 6-yl; 3,6-dihydro-2H-l, 2-oxazin-2- or 3- or 4- or 5- or 6-yl; 5,6-dihydro-2H-l, 2-oxazin-2- or 3- or 4- or 5- or 6-yl; 5,6-dihydro-4H-l, 2-oxazin-3- or 4- or 5- or 6-yl; 2H-l, 2-oxazin-2- or 3- or 4- or 5- or 6-yl; 6H-l, 2-oxazin-3- or 4- or 5- or 6-yl; 4H-l, 2-oxazin-3- or 4- or 5- or 6-yl; l, 3-oxazinan-2- or 3- or 4- or 5- or 6-yl; 3,4-dihydro-2H-l, 3-oxazin-2- or 3- or 4- or 5- or 6-yl; 3,6-dihydro-2H-l, 3-oxazin-2- or 3- or 4- or 5- or 6-yl; 5,6-dihydro-2H
1.3-oxazin-2- oder 4- oder 5- oder 6-yl; 5,6-Dihydro-4H-l,3-oxazin-2- oder 4- oder 5- oder 6-yl; 2H-1,3-oxazin-2- or 4- or 5- or 6-yl; 5,6-dihydro-4H-l, 3-oxazin-2- or 4- or 5- or 6-yl; 2H
1.3-Oxazin-2- oder 4- oder 5- oder 6-yl; 6H-l,3-Oxazin-2- oder 4- oder 5- oder 6-yl; 4H-l,3-Oxazin-2- oder 4- oder 5- oder 6-yl; Morpholin-2- oder 3- oder 4-yl; 3,4-Dihydro-2H-l,4-oxazin-2- oder 3- oder 4- oder 5- oder 6-yl; 3,6-Dihydro-2H-l,4-oxazin-2- oder 3- oder 5- oder 6-yl; 2H-l,4-oxazin-2- oder 3- oder 5- oder 6-yl; 4H-l,4-oxazin-2- oder 3-yl; 1 ,2-Oxazepan-2- oder 3- oder 4- oder 5- oder 6- oder 7- yl; 2,3,4,5-Tetrahydro-l,2-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3,4,7-Tetrahydro-l,2- oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3,6,7-Tetrahydro-l,2-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,5,6,7-Tetrahydro-l,2-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7- yl; 4,5,6,7-Tetrahydro-l,2-oxazepin-3- oder 4- oder 5- oder 6- oder 7-yl; 2,3-Dihydro-l,2-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,5-Dihydro-l,2-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,7-Dihydro-l,2-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 4,5-Dihydro-l,2- oxazepin-3- oder 4- oder 5- oder 6- oder 7-yl; 4,7-Dihydro-l,2-oxazepin-3- oder 4- oder 5- oder 6- oder 7-yl; 6,7-Dihydro-l,2-oxazepin-3- oder 4- oder 5- oder 6- oder 7-yl; l,2-Oxazepin-3- oder 4- oder 5- oder 6- oder 7-yl; l,3-Oxazepan-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3,4,5-Tetrahydro-l,3- oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3,4,7-Tetrahydro-l,3-oxazepin-2- oder 3- oder1,3-oxazin-2- or 4- or 5- or 6-yl; 6H-l, 3-oxazin-2- or 4- or 5- or 6-yl; 4H-l, 3-oxazin-2- or 4- or 5- or 6-yl; Morpholin-2- or 3- or 4-yl; 3,4-dihydro-2H-l, 4-oxazin-2- or 3- or 4- or 5- or 6-yl; 3,6-dihydro-2H-l, 4-oxazin-2- or 3- or 5- or 6-yl; 2H-l, 4-oxazin-2- or 3- or 5- or 6-yl; 4H-l, 4-oxazin-2- or 3-yl; 1, 2-oxazepan-2- or 3- or 4- or 5- or 6- or 7- yl; 2,3,4,5-tetrahydro-l, 2-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,3,4,7-tetrahydro-l, 2-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,3,6,7-tetrahydro-l, 2-oxazepine-2- or 3- or 4- or 5- or 6- or 7-yl; 2,5,6,7-tetrahydro-l, 2-oxazepine-2- or 3- or 4- or 5- or 6- or 7- yl; 4,5,6,7-tetrahydro-l, 2-oxazepin-3- or 4- or 5- or 6- or 7-yl; 2,3-dihydro-l, 2-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,5-dihydro-l, 2-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,7-dihydro-l, 2-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 4,5-dihydro-l, 2-oxazepin-3- or 4- or 5- or 6- or 7-yl; 4,7-dihydro-l, 2-oxazepin-3- or 4- or 5- or 6- or 7-yl; 6,7-dihydro-l, 2-oxazepin-3- or 4- or 5- or 6- or 7-yl; l, 2-oxazepin-3- or 4- or 5- or 6- or 7-yl; l, 3-oxazepan-2- or 3- or 4- or 5- or 6- or 7-yl; 2,3,4,5-tetrahydro-l, 3-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,3,4,7-tetrahydro-l, 3-oxazepine-2- or 3- or
4- oder 5- oder 6- oder 7-yl; 2,3,6,7-Tetrahydro-l,3-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7- yl; 2,5,6,7-Tetrahydro-l,3-oxazepin-2- oder 4- oder 5- oder 6- oder 7-yl; 4,5,6,7-Tetrahydro-l,3- oxazepin-2- oder 4- oder 5- oder 6- oder 7-yl; 2,3-Dihydro-l,3-oxazepin-2- oder 3- oder 4- oder 5- oder4- or 5- or 6- or 7-yl; 2,3,6,7-tetrahydro-l, 3-oxazepine-2- or 3- or 4- or 5- or 6- or 7- yl; 2,5,6,7-tetrahydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 4,5,6,7-tetrahydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 2,3-dihydro-l, 3-oxazepine-2- or 3- or 4- or 5- or
6- oder 7-yl; 2,5-Dihydro-l,3-oxazepin-2- oder 4- oder 5- oder 6- oder 7-yl; 2,7-Dihydro-l,3-oxazepin- 2- oder 4- oder 5- oder 6- oder 7-yl; 4,5-Dihydro-l,3-oxazepin-2- oder 4- oder 5- oder 6- oder 7-yl; 4,7- Dihydro-l,3-oxazepin-2- oder 4- oder 5- oder 6- oder 7-yl; 6,7-Dihydro-l,3-oxazepin-2- oder 4- oder 5- oder 6- oder 7-yl; l,3-Oxazepin-2- oder 4- oder 5- oder 6- oder 7-yl; 1 ,4-Oxazepan-2- oder 3- oder 5- oder 6- oder 7-yl; 2,3,4,5-Tetrahydro-l,4-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2, 3, 4, 7- Tetrahydro-l,4-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3,6,7-Tetrahydro-l,4-oxazepin-6- or 7-yl; 2,5-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 2,7-dihydro-l, 3-oxazepine 2- or 4- or 5- or 6- or 7-yl; 4,5-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 4,7-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 6,7-dihydro-l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; l, 3-oxazepin-2- or 4- or 5- or 6- or 7-yl; 1, 4-oxazepan-2- or 3- or 5- or 6- or 7-yl; 2,3,4,5-tetrahydro-l, 4-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2, 3, 4, 7-tetrahydro-l, 4-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,3,6,7-tetrahydro-l, 4-oxazepin
2- oder 3- oder 5- oder 6- oder 7-yl; 2,5,6,7-Tetrahydro-l,4-oxazepin-2- oder 3- oder 5- oder 6- oder 7- yl; 4,5,6,7-Tetrahydro-l,4-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7-yl; 2,3-Dihydro-l,4- oxazepin-2- oder 3- oder 5- oder 6- oder 7-yl; 2,5-Dihydro-l,4-oxazepin-2- oder 3- oder 5- oder 6- oder2- or 3- or 5- or 6- or 7-yl; 2,5,6,7-tetrahydro-l, 4-oxazepine-2- or 3- or 5- or 6- or 7- yl; 4,5,6,7-tetrahydro-l, 4-oxazepin-2- or 3- or 4- or 5- or 6- or 7-yl; 2,3-dihydro-l, 4-oxazepin-2- or 3- or 5- or 6- or 7-yl; 2,5-dihydro-l, 4-oxazepine-2- or 3- or 5- or 6- or
7-yl; 2,7-Dihydro-l,4-oxazepin-2- oder 3- oder 5- oder 6- oder 7-yl; 4,5-Dihydro-l,4-oxazepin-2- oder7-yl; 2,7-dihydro-l, 4-oxazepin-2- or 3- or 5- or 6- or 7-yl; 4,5-dihydro-l, 4-oxazepine-2- or
3- oder 4- oder 5- oder 6- oder 7-yl; 4,7-Dihydro-l,4-oxazepin-2- oder 3- oder 4- oder 5- oder 6- oder 7- yl; 6,7-Dihydro-l,4-oxazepin-2- oder 3- oder 5- oder 6- oder 7-yl; 1 ,4-Oxazepin-2- oder 3- oder 5- oder 6- oder 7-yl; Isothiazolidin-2- oder 3- oder 4- oder 5-yl; 2,3-Dihydroisothiazol-2- oder 3- oder 4- oder 5- yl; 2,5-Dihydroisothiazol-2- oder 3- oder 4- oder 5-yl; 4,5-Dihydroisothiazol-3- oder 4- oder 5-yl; 1,3- Thiazolidin-2- oder 3- oder 4- oder 5-yl; 2,3-Dihydro-l ,3-thiazol-2- oder 3- oder 4- oder 5-yl; 2,5- Dihydro-l,3-thiazol-2- oder 4- oder 5-yl; 4,5-Dihydro-l,3-thiazol-2- oder 4- oder 5-yl; l,3-Thiazinan-2- oder 3- oder 4- oder 5- oder 6-yl; 3,4-Dihydro-2H-l,3-thiazin-2- oder 3- oder 4- oder 5- oder 6-yl; 3,6- Dihydro-2H-l,3-thiazin-2- oder 3- oder 4- oder 5- oder 6-yl; 5,6-Dihydro-2H-l,3-thiazin-2- oder 4- oder3- or 4- or 5- or 6- or 7-yl; 4,7-dihydro-l, 4-oxazepine-2- or 3- or 4- or 5- or 6- or 7- yl; 6,7-dihydro-l, 4-oxazepin-2- or 3- or 5- or 6- or 7-yl; 1, 4-oxazepin-2- or 3- or 5- or 6- or 7-yl; Isothiazolidin-2- or 3- or 4- or 5-yl; 2,3-dihydroisothiazol-2- or 3- or 4- or 5-yl; 2,5-dihydroisothiazol-2- or 3- or 4- or 5-yl; 4,5-dihydroisothiazol-3- or 4- or 5-yl; 1,3-thiazolidin-2- or 3- or 4- or 5-yl; 2,3-dihydro-l, 3-thiazol-2- or 3- or 4- or 5-yl; 2,5-dihydro-l, 3-thiazol-2- or 4- or 5-yl; 4,5-dihydro-l, 3-thiazol-2- or 4- or 5-yl; l, 3-thiazinan-2- or 3- or 4- or 5- or 6-yl; 3,4-dihydro-2H-l, 3-thiazin-2- or 3- or 4- or 5- or 6-yl; 3,6-dihydro-2H-l, 3-thiazin-2- or 3- or 4- or 5- or 6-yl; 5,6-dihydro-2H-l, 3-thiazine-2- or 4- or
5- oder 6-yl; 5,6-Dihydro-4H-l,3-thiazin-2- oder 4- oder 5- oder 6-yl; 2H-l,3-Thiazin-2- oder 4- oder 5- oder 6-yl; 6H-l,3-Thiazin-2- oder 4- oder 5- oder 6-yl; 4H-l,3-Thiazin-2- oder 4- oder 5- oder 6-yl. Weitere Beispiele für“Heterocyclyl“ sind ein partiell oder vollständig hydrierter heterocyclischer Rest mit 3 Heteroatomen aus der Gruppe N, O und S, wie beispielsweise l,4,2-Dioxazolidin-2- oder 3- oder 5-yl; l,4,2-Dioxazol-3- oder 5-yl; 1 ,4,2-Dioxazinan-2- oder -3- oder 5- oder 6-yl; 5,6-Dihydro-l,4,2- dioxazin-3- oder 5- oder 6-yl; l,4,2-Dioxazin-3- oder 5- oder 6-yl; l,4,2-Dioxazepan-2- oder 3- oder 5- oder 6- oder 7-yl; 6,7-Dihydro-5H-l,4,2-Dioxazepin-3- oder 5- oder 6- oder 7-yl; 2,3-Dihydro-7H-l,4,2- Dioxazepin-2- oder 3- oder 5- oder 6- oder 7-yl; 2,3-Dihydro-5H-l,4,2-Dioxazepin-2- oder 3- oder 5- oder 6- oder 7-yl; 5H-l,4,2-Dioxazepin-3- oder 5- oder 6- oder 7-yl; 7H-l,4,2-Dioxazepin-3- oder 5- oder 6- oder 7-yl. Strukturbeispiele für gegebenenfalls weiter substituierte Heterocyclen sind auch im Folgenden aufgeführt: 5- or 6-yl; 5,6-dihydro-4H-l, 3-thiazin-2- or 4- or 5- or 6-yl; 2H-l, 3-thiazin-2- or 4- or 5- or 6-yl; 6H-l, 3-thiazin-2- or 4- or 5- or 6-yl; 4H-l, 3-thiazin-2- or 4- or 5- or 6-yl. Further examples of “heterocyclyl” are a partially or fully hydrogenated heterocyclic radical with 3 heteroatoms from the group N, O and S, such as l, 4,2-dioxazolidin-2- or 3- or 5-yl; l, 4,2-dioxazol-3- or 5-yl; 1, 4,2-dioxazinan-2- or -3- or 5- or 6-yl; 5,6-dihydro-l, 4,2-dioxazin-3- or 5- or 6-yl; l, 4,2-dioxazin-3- or 5- or 6-yl; l, 4,2-dioxazepan-2- or 3- or 5- or 6- or 7-yl; 6,7-dihydro-5H-l, 4,2-dioxazepin-3- or 5- or 6- or 7-yl; 2,3-dihydro-7H-l, 4,2-dioxazepin-2- or 3- or 5- or 6- or 7-yl; 2,3-dihydro-5H-l, 4,2-dioxazepine-2- or 3- or 5- or 6- or 7-yl; 5H-l, 4,2-dioxazepin-3- or 5- or 6- or 7-yl; 7H-l, 4,2-dioxazepin-3- or 5- or 6- or 7-yl. Structural examples of optionally further substituted heterocycles are also listed below:
31 31
Die oben aufgeführten Heterocyclen sind bevorzugt beispielsweise durch Wasserstoff, Halogen, Alkyl, Haloalkyl, Hydroxy, Alkoxy, Cycloalkoxy, Aryloxy, Alkoxyalkyl, Alkoxyalkoxy, Cycloalkyl, The heterocycles listed above are preferably, for example, by hydrogen, halogen, alkyl, haloalkyl, hydroxy, alkoxy, cycloalkoxy, aryloxy, alkoxyalkyl, alkoxyalkoxy, cycloalkyl,
Halocycloalkyl, Aryl, Arylalkyl, Heteroaryl, Heterocyclyl, Alkenyl, Alkylcarbonyl, Cycloalkylcarbonyl, Arylcarbonyl, Heteroarylcarbonyl, Alkoxycarbonyl, Hydroxycarbonyl, Cycloalkoxycarbonyl, Halocycloalkyl, aryl, arylalkyl, heteroaryl, heterocyclyl, alkenyl, alkylcarbonyl, cycloalkylcarbonyl, arylcarbonyl, heteroarylcarbonyl, alkoxycarbonyl, hydroxycarbonyl, cycloalkoxycarbonyl,
Cycloalkylalkoxycarbonyl, Alkoxycarbonylalkyl, Arylalkoxycarbonyl, Arylalkoxycarbonylalkyl, Alkinyl, Alkinylalkyl, Alkylalkinyl, Tris-alkylsilylalkinyl, Nitro, Amino, Cyano, Haloalkoxy, Cycloalkylalkoxycarbonyl, alkoxycarbonylalkyl, arylalkoxycarbonyl, arylalkoxycarbonylalkyl, alkynyl, alkynylalkyl, alkylalkynyl, tris-alkylsilylalkynyl, nitro, amino, cyano, haloalkoxy,
Haloalkylthio, Alkylthio, Hydrothio, Hydroxyalkyl, Oxo, Heteroarylalkoxy, Arylalkoxy, Haloalkylthio, alkylthio, hydrothio, hydroxyalkyl, oxo, heteroarylalkoxy, arylalkoxy,
Heterocyclylalkoxy, Heterocyclylalkylthio, Heterocyclyloxy, Heterocyclylthio, Heteroaryloxy, Bis- alkylamino, Alkylamino, Cycloalkylamino, Hydroxycarbonylalkylamino, Alkoxycarbonylalkylamino,Heterocyclylalkoxy, heterocyclylalkylthio, heterocyclyloxy, heterocyclylthio, heteroaryloxy, bis-alkylamino, alkylamino, cycloalkylamino, hydroxycarbonylalkylamino, alkoxycarbonylalkylamino,
Arylalkoxycarbonylalkylamino, Alkoxycarbonylalkyl(alkyl)amino, Aminocarbonyl, Arylalkoxycarbonylalkylamino, alkoxycarbonylalkyl (alkyl) amino, aminocarbonyl,
Alkylaminocarbonyl, Bis-alkylaminocarbonyl, Cycloalkylaminocarbonyl, Alkylaminocarbonyl, bis-alkylaminocarbonyl, cycloalkylaminocarbonyl,
Hydroxycarbonylalkylaminocarbonyl, Alkoxycarbonylalkylaminocarbonyl, Hydroxycarbonylalkylaminocarbonyl, alkoxycarbonylalkylaminocarbonyl,
Arylalkoxycarbonylalkylaminocarbonyl substituiert. Arylalkoxycarbonylalkylaminocarbonyl substituted.
Wenn ein Grundkörper "durch einen oder mehrere Reste" aus einer Aufzählung von Resten (= Gruppe) oder einer generisch definierten Gruppe von Resten substituiert ist, so schließt dies jeweils die gleichzeitige Substitution durch mehrere gleiche und/oder strukturell unterschiedliche Reste ein. Handelt es sich es sich um einen teilweise oder vollständig gesättigten Stickstoff-Heterocyclus, so kann dieser sowohl über Kohlenstoff als auch über den Stickstoff mit dem Rest des Moleküls verknüpft sein. If a basic body is substituted “by one or more residues” from an enumeration of residues (= group) or a generically defined group of residues, this includes the simultaneous substitution by several identical and / or structurally different residues. If the nitrogen heterocycle is partially or fully saturated, this can be linked to the rest of the molecule via carbon as well as nitrogen.
Als Substituenten für einen substituierten heterocyclischen Rest kommen die weiter unten genannten Substituenten in Frage, zusätzlich auch Oxo und Thioxo. Die Oxogruppe als Substituent an einem Ring- C-Atom bedeutet dann beispielsweise eine Carbonylgruppe im heterocyclischen Ring. Dadurch sind vorzugsweise auch Lactone und Lactame umfasst. Die Oxogruppe kann auch an den Heteroringatomen, die in verschiedenen Oxidationsstufen existieren können, z.B. bei N und S, auftreten und bilden dann beispielsweise die divalenten Gruppen N(O) , S(O) (auch kurz SO) und S(0)2 (auch kurz SO2) im heterocyclischen Ring. lm Fall von -N(O)- und -S(0)-Gruppen sind jeweils beide Enantiomere umfasst. Erfindungsgemäß steht der Ausdruck„Heteroaryl“ für heteroaromatische Verbindungen, d. h. Possible substituents for a substituted heterocyclic radical are the substituents mentioned below, and also oxo and thioxo. The oxo group as a substituent on a ring carbon atom then means, for example, a carbonyl group in the heterocyclic ring. This preferably also includes lactones and lactams. The oxo group can also occur on the hetero ring atoms, which can exist in different oxidation states, for example in the case of N and S, and then form, for example, the divalent groups N (O), S (O) (also SO for short) and S (0) 2 ( also briefly SO2) in the heterocyclic ring. In the case of -N (O) - and -S (0) groups, both enantiomers are included. According to the invention, the term “heteroaryl” stands for heteroaromatic compounds, ie
vollständig ungesättigte aromatische heterocyclische Verbindungen, vorzugsweise für 5- bis 7-gliedrige Ringe mit 1 bis 4, vorzugsweise 1 oder 2 gleichen oder verschiedenen Heteroatomen, vorzugsweise O, S oder N. Erfindungsgemäße Heteroaryle sind beispielsweise lH-Pyrrol-l-yl; lH-Pyrrol-2-yl; lH-Pyrrol-completely unsaturated aromatic heterocyclic compounds, preferably for 5- to 7-membered rings with 1 to 4, preferably 1 or 2 identical or different heteroatoms, preferably O, S or N. Heteroaryls according to the invention are, for example, 1H-pyrrol-l-yl; lH-pyrrol-2-yl; lH-pyrrole
3-yl; Furan-2-yl; Furan-3-yl; Thien-2-yl; Thien-3-yl, lH-Imidazol-l-yl; lH-Imidazol-2-yl; lH-Imidazol-3-yl; Furan-2-yl; Furan-3-yl; Thien-2-yl; Thien-3-yl, lH-imidazol-l-yl; lH-imidazol-2-yl; lH-imidazole
4-yl; lH-Imidazol-5-yl; lH-Pyrazol-l-yl; lH-Pyrazol-3-yl; lH-Pyrazol-4-yl; lH-Pyrazol-5-yl, lH-l,2,3- Triazol-l-yl, lH-l,2,3-Triazol-4-yl, lH-l,2,3-Triazol-5-yl, 2H-l,2,3-Triazol-2-yl, 2H-l,2,3-Triazol-4-yl, lH-l,2,4-Triazol-l-yl, lH-l,2,4-Triazol-3-yl, 4H-l,2,4-Triazol-4-yl, l,2,4-Oxadiazol-3-yl, 1,2,4- Oxadiazol-5-yl, l,3,4-Oxadiazol-2-yl, l,2,3-Oxadiazol-4-yl, l,2,3-Oxadiazol-5-yl, l,2,5-Oxadiazol-3-yl, Azepinyl, Pyridin-2-yl, Pyridin-3-yl, Pyridin-4-yl, Pyrazin-2-yl, Pyrazin-3-yl, Pyrimidin-2-yl, 4-yl; lH-imidazol-5-yl; lH-pyrazol-l-yl; lH-pyrazol-3-yl; lH-pyrazol-4-yl; lH-pyrazol-5-yl, lH-l, 2,3-triazol-l-yl, lH-l, 2,3-triazol-4-yl, lH-l, 2,3-triazol-5-yl, 2H-l, 2,3-triazol-2-yl, 2H-l, 2,3-triazol-4-yl, lH-l, 2,4-triazol-l-yl, lH-l, 2,4- Triazol-3-yl, 4H-l, 2,4-triazol-4-yl, l, 2,4-oxadiazol-3-yl, 1,2,4-oxadiazol-5-yl, l, 3,4- Oxadiazol-2-yl, l, 2,3-oxadiazol-4-yl, l, 2,3-oxadiazol-5-yl, l, 2,5-oxadiazol-3-yl, azepinyl, pyridin-2-yl, Pyridin-3-yl, pyridin-4-yl, pyrazin-2-yl, pyrazin-3-yl, pyrimidin-2-yl,
Pyrimidin-4-yl, Pyrimidin-5-yl, Pyridazin-3-yl, Pyridazin-4-yl, l,3,5-Triazin-2-yl, l,2,4-Triazin-3-yl, l,2,4-Triazin-5-yl, 1 ,2,4-Triazin-6-yl, l,2,3-Triazin-4-yl, l,2,3-Triazin-5-yl, 1,2,4-, 1,3,2-, 1,3,6- und l,2,6-Oxazinyl, Isoxazol-3-yl, Isoxazol-4-yl, Isoxazol-5-yl, l,3-Oxazol-2-yl, l,3-Oxazol-4-yl, 1,3- Oxazol-5-yl, Isothiazol-3-yl, Isothiazol-4-yl, Isothiazol-5-yl, l,3-Thiazol-2-yl, l,3-Thiazol-4-yl, 1,3- Thiazol-5-yl, Oxepinyl, Thiepinyl, 1 ,2,4-Triazolonyl und 1 ,2,4-Diazepinyl, 2H-l,2,3,4-Tetrazol-5-yl, lH-l,2,3,4-Tetrazol-5-yl, l,2,3,4-Oxatriazol-5-yl, l,2,3,4-Thiatriazol-5-yl, l,2,3,5-Oxatriazol-4-yl, l,2,3,5-Thiatriazol-4-yl. Die erfindungsgemäßen Heteroarylgruppen können ferner mit einem oder mehreren, gleichen oder verschiedenen Resten substituiert sein. Sind zwei benachbarte Pyrimidin-4-yl, pyrimidin-5-yl, pyridazin-3-yl, pyridazin-4-yl, l, 3,5-triazin-2-yl, l, 2,4-triazin-3-yl, l, 2,4-triazin-5-yl, 1, 2,4-triazin-6-yl, l, 2,3-triazin-4-yl, l, 2,3-triazin-5-yl, 1,2, 4-, 1,3,2-, 1,3,6- and l, 2,6-oxazinyl, isoxazol-3-yl, isoxazol-4-yl, isoxazol-5-yl, l, 3-oxazol-2 -yl, 1,3-oxazol-4-yl, 1,3-oxazol-5-yl, isothiazol-3-yl, isothiazol-4-yl, isothiazol-5-yl, 1,3-thiazol-2-yl , l, 3-thiazol-4-yl, 1,3-thiazol-5-yl, oxepinyl, thiepinyl, 1, 2,4-triazolonyl and 1, 2,4-diazepinyl, 2H-l, 2,3,4 -Tetrazol-5-yl, lH-l, 2,3,4-tetrazol-5-yl, l, 2,3,4-oxatriazol-5-yl, l, 2,3,4-thiatriazol-5-yl , l, 2,3,5-oxatriazol-4-yl, l, 2,3,5-thiatriazol-4-yl. The heteroaryl groups according to the invention can furthermore be substituted by one or more, identical or different radicals. Are two neighboring
Kohlenstoffatome Bestandteil eines weiteren aromatischen Rings, so handelt es sich um annellierte heteroaromatische Systeme, wie benzokondensierte oder mehrfach annellierte Heteroaromaten. Carbon atoms are part of a further aromatic ring, so they are fused heteroaromatic systems, such as benzo-fused or multiply fused heteroaromatics.
Bevorzugt sind beispielsweise Chinoline (z. B. Chinolin-2-yl, Chinolin-3-yl, Chinolin-4-yl, Chinolin-5- yl, Chinolin-6-yl, Chinolin-7-yl, Chinolin-8-yl); Isochinoline (z. B. Isochinolin- l-yl, Isochinolin-3-yl, Isochinolin-4-yl, Isochinolin-5-yl, Isochinolin-6-yl, Isochinolin-7-yl, Isochinolin-8-yl); Chinoxalin; Chinazolin; Cinnolin; l,5-Naphthyridin; l,6-Naphthyridin; l,7-Naphthyridin; l,8-Naphthyridin; 2,6- Naphthyridin; 2,7-Naphthyridin; Phthalazin; Pyridopyrazine; Pyridopyrimidine; Pyridopyridazine; Pteridine; Pyrimidopyrimidine. Beispiele für Heteroaryl sind auch 5- oder 6-gliedrige benzokondensierte Ringe aus der Gruppe lH-Indol-l-yl, lH-Indol-2-yl, lH-Indol-3-yl, lH-Indol-4-yl, lH-Indol-5-yl, 1H- Indol-6-yl, lH-Indol-7-yl, l-Benzofüran-2-yl, l-Benzofüran-3-yl, l-Benzofüran-4-yl, l-Benzofüran-5- yl, l-Benzofüran-6-yl, l-Benzofüran-7-yl, l-Benzothiophen-2-yl, l-Benzothiophen-3-yl, 1- Benzothiophen-4-yl, l-Benzothiophen-5-yl, l-Benzothiophen-6-yl, l-Benzothiophen-7-yl, lH-Indazol-For example, quinolines (e.g. quinolin-2-yl, quinolin-3-yl, quinolin-4-yl, quinolin-5-yl, quinolin-6-yl, quinolin-7-yl, quinolin-8-yl ); Isoquinolines (e.g. isoquinolin-l-yl, isoquinolin-3-yl, isoquinolin-4-yl, isoquinolin-5-yl, isoquinolin-6-yl, isoquinolin-7-yl, isoquinolin-8-yl); quinoxaline; quinazoline; cinnoline; l, 5-naphthyridine; l, 6-naphthyridine; l, 7-naphthyridine; l, 8-naphthyridine; 2,6-naphthyridine; 2,7-naphthyridine; phthalazine; Pyridopyrazine; pyridopyrimidines; Pyridopyridazine; pteridines; Pyrimidopyrimidine. Examples of heteroaryl are also 5- or 6-membered benzo-fused rings from the group lH-indol-l-yl, lH-indol-2-yl, lH-indol-3-yl, lH-indol-4-yl, lH- Indol-5-yl, 1H-indol-6-yl, lH-indol-7-yl, l-benzofuran-2-yl, l-benzofuran-3-yl, l-benzofuran-4-yl, l-benzofuran 5- yl, l-benzofuran-6-yl, l-benzofuran-7-yl, l-benzothiophene-2-yl, l-benzothiophene-3-yl, 1-benzothiophene-4-yl, l-benzothiophene-5- yl, l-benzothiophene-6-yl, l-benzothiophene-7-yl, lH-indazole
1-yl, lH-Indazol-3-yl, lH-Indazol-4-yl, lH-Indazol-5-yl, lH-Indazol-6-yl, lH-Indazol-7-yl, 2H-Indazol-1-yl, 1H-indazol-3-yl, 1H-indazol-4-yl, 1H-indazol-5-yl, 1H-indazol-6-yl, 1H-indazol-7-yl, 2H-indazole
2-yl, 2H-Indazol-3-yl, 2H-Indazol-4-yl, 2H-Indazol-5-yl, 2H-Indazol-6-yl, 2H-Indazol-7-yl, 2H- Isoindol-2-yl, 2H-Isoindol-l-yl, 2H-Isoindol-3-yl, 2H-Isoindol-4-yl, 2H-Isoindol-5-yl, 2H-Isoindol-6-yl; 2H-Isoindol-7-yl, lH-Benzimidazol-l-yl, lH-Benzimidazol-2-yl, lH-Benzimidazol-4-yl, 1H- Benzimidazol-5-yl, lH-Benzimidazol-6-yl, lH-Benzimidazol-7-yl, l,3-Benzoxazol-2-yl, 1,3- Benzoxazol-4-yl, l,3-Benzoxazol-5-yl, l,3-Benzoxazol-6-yl, l,3-Benzoxazol-7-yl, l,3-Benzthiazol-2-yl, l,3-Benzthiazol-4-yl, l,3-Benzthiazol-5-yl, l,3-Benzthiazol-6-yl, l,3-Benzthiazol-7-yl, 1,2- Benzisoxazol-3-yl, l,2-Benzisoxazol-4-yl, l,2-Benzisoxazol-5-yl, l,2-Benzisoxazol-6-yl, 1,2- Benzisoxazol-7-yl, l,2-Benzisothiazol-3-yl, l,2-Benzisothiazol-4-yl, l,2-Benzisothiazol-5-yl, 1,2- Benzisothiazol-6-yl, l,2-Benzisothiazol-7-yl. 2-yl, 2H-indazol-3-yl, 2H-indazol-4-yl, 2H-indazol-5-yl, 2H-indazol-6-yl, 2H-indazol-7-yl, 2H-isoindol-2- yl, 2H-isoindol-1-yl, 2H-isoindol-3-yl, 2H-isoindol-4-yl, 2H-isoindol-5-yl, 2H-isoindol-6-yl; 2H-isoindol-7-yl, 1H-benzimidazol-1-yl, 1H-benzimidazol-2-yl, 1H-benzimidazol-4-yl, 1H-benzimidazol-5-yl, 1H-benzimidazol-6-yl, 1H- Benzimidazol-7-yl, l, 3-benzoxazol-2-yl, 1,3-benzoxazol-4-yl, l, 3-benzoxazol-5-yl, l, 3-benzoxazol-6-yl, l, 3- Benzoxazol-7-yl, l, 3-benzthiazol-2-yl, l, 3-benzothiazol-4-yl, l, 3-benzothiazol-5-yl, l, 3-benzothiazol-6-yl, l, 3-benzothiazol-7-yl, 1,2-benzisoxazol-3-yl, l, 2-benzisoxazol-4-yl, l, 2-benzisoxazol-5-yl, l, 2-benzisoxazol-6-yl, 1,2-benzisoxazol-7-yl, l, 2-benzisothiazol-3-yl, 1,2-benzisothiazol-4-yl, 1,2-benzisothiazol-5-yl, 1,2-benzisothiazol-6-yl, 1,2-benzisothiazol-7-yl.
Die Bezeichnung "Halogen" bedeutet beispielsweise Fluor, Chlor, Brom oder Iod. Wird die The term "halogen" means, for example, fluorine, chlorine, bromine or iodine. Will the
Bezeichnung für einen Rest verwendet, dann bedeutet "Halogen" beispielsweise ein Fluor-, Chlor-, Brom- oder Iodatom. Name used for a radical, then "halogen" means for example a fluorine, chlorine, bromine or iodine atom.
Erfindungsgemäß bedeutet„Alkyl“ einen geradkettigen oder verzweigten offenketigen, gesättigten Kohlenwasserstoffrest, der gegebenenfalls ein- oder mehrfach substituiert ist und im letzteren Falle als „substituiertes Alkyl“ bezeichnet wird. Bevorzugte Substituenten sind Halogenatome, Alkoxy-, Haloalkoxy-, Cyano-, Alkylthio, Haloalkylthio-, Amino- oder Nitrogruppen, besonders bevorzugt sind Methoxy, Methyl, Fluoralkyl, Cyano, Nitro, Fluor, Chlor, Brom oder lod. Die Vorsilbe„Bis“ schließt auch die Kombination unterschiedlicher Alkylreste ein, z. B. Methyl(Ethyl) oder Ethyl(Methyl). According to the invention, “alkyl” means a straight-chain or branched, open-chain, saturated hydrocarbon radical which is optionally substituted one or more times and is referred to in the latter case as “substituted alkyl”. Preferred substituents are halogen atoms, alkoxy, haloalkoxy, cyano, alkylthio, haloalkylthio, amino or nitro groups; methoxy, methyl, fluoroalkyl, cyano, nitro, fluorine, chlorine, bromine or iodine are particularly preferred. The prefix "bis" also includes the combination of different alkyl residues, e.g. B. methyl (ethyl) or ethyl (methyl).
„Haloalkyl“, ,,-alkenyl“ und ,,-alkinyl“ bedeuten durch gleiche oder verschiedene Halogenatome, teilweise oder vollständig substituiertes Alkyl, Alkenyl bzw. Alkinyl, z.B. Monohaloalkyl "Haloalkyl", "- alkenyl" and "- alkynyl" mean alkyl or alkenyl or alkynyl which is partially or completely substituted by identical or different halogen atoms, e.g. monohaloalkyl
(= Monohalogenalkyl) wie z. B. CH2CH2CI, CH2CH2Br, CHCICH3, CH2CI, CH2F; Perhaloalkyl wie z. B. CCI3, CCIF2, CFC12,CF2CC1F2, CF2CCIFCF3; Polyhaloalkyl wie z. B. CH2CHFC1, CF2CC1FH, CF2CBrFH, CH2CF3; Der Begriff Perhaloalkyl umfasst dabei auch den Begriff Perfluoralkyl. (= Monohalogenalkyl) such as B. CH 2 CH 2 CI, CH 2 CH 2 Br, CHCICH 3 , CH 2 CI, CH 2 F; Perhaloalkyl such as B. CCI3, CCIF 2 , CFC1 2 , CF 2 CC1F 2 , CF 2 CCIFCF3; Polyhaloalkyl such as B. CH 2 CHFC1, CF 2 CC1FH, CF 2 CBrFH, CH 2 CF 3 ; The term perhaloalkyl also includes the term perfluoroalkyl.
„Teilfluoriertes Alkyl“ bedeutet einen geradkettigen oder verzweigten, gesättigten Kohlenwasserstoff, der einfach oder mehrfach durch Fluor substituiert ist, wobei sich die entsprechenden Fluoratome als Substituenten an einem oder mehreren verschiedenen Kohlenstoffatomen der geradkettigen oder verzweigten Kohlenwasserstoffkete befinden können, wie z. B. CHFCH3, CH2CH2F, CH2CH2CF3, CHF2, CH2F, CHFCF2CF3 “Partially fluorinated alkyl” means a straight-chain or branched, saturated hydrocarbon which is substituted one or more times by fluorine, and the corresponding fluorine atoms can be located as substituents on one or more different carbon atoms of the straight-chain or branched hydrocarbon chains, such as, for example, B. CHFCH 3 , CH 2 CH 2 F, CH 2 CH 2 CF 3 , CHF 2 , CH 2 F, CHFCF 2 CF 3
„Teilfluoriertes Haloalkyl“ bedeutet einen geradkettigen oder verzweigten, gesättigten “Partially fluorinated haloalkyl” means a straight-chain or branched, saturated
Kohlenwasserstoff, der durch verschiedenene Halogenatomen mit mindestens einem Fluoratom substituiert ist, wobei alle anderen gegebenenfalls vorhandenen Halogenatome ausgewählt sind aus der Gruppe Fluor, Chlor oder Brom, lod. Die entsprechenden Halogenatome können sich dabei als Substituenten an einem oder mehreren verschiedenen Kohlenstoffatomen der geradkettigen oder verzweigten Kohlenwasserstoffkette befinden. Teilfluoriertes Haloalkyl schließt auch die vollständige Substitution der geradkettigen oder verzweigten Kete durch Halogen unter Beteiligung von mindestens einem Fluoratom ein. „Haloalkoxy“ ist z.B. OCF3, OCHF2, OCH2F, OCF2CF3, OCH2CF3 und OCH2CH2CI; Entsprechendes gilt für Haloalkenyl und andere durch Halogen substituierten Reste. Hydrocarbon which is substituted by various halogen atoms with at least one fluorine atom, all other halogen atoms which may be present being selected from the group consisting of fluorine, chlorine or bromine, iodine. The corresponding halogen atoms can be located as substituents on one or more different carbon atoms of the straight-chain or branched hydrocarbon chain. Partially fluorinated haloalkyl also includes the complete substitution of the straight or branched ketene by halogen with the participation of at least one fluorine atom. “Haloalkoxy” is, for example, OCF 3 , OCHF 2 , OCH 2 F, OCF 2 CF 3 , OCH 2 CF 3 and OCH 2 CH 2 CI; The same applies to haloalkenyl and other halogen-substituted radicals.
Der hier beispielhaft genannte Ausdruck "(Ci-C4)-Alkyl" bedeutet eine Kurzschreibweise für geradkettiges oder verzweigtes Alkyl mit einem bis 4 Kohlenstoffatomen entsprechend der The expression "(Ci-C4) -alkyl" mentioned here by way of example means a shorthand notation for straight-chain or branched alkyl having one to 4 carbon atoms corresponding to the
Bereichsangabe für C-Atome, d. h. umfasst die Reste Methyl, Ethyl, 1 -Propyl, 2-Propyl, 1 -Butyl,Range for C atoms, d. H. includes the residues methyl, ethyl, 1-propyl, 2-propyl, 1-butyl,
2-Butyl, 2-Methylpropyl oder tert-Butyl. Allgemeine Alkylreste mit einem größeren angegebenen Bereich von C-Atomen, z. B. "(Ci-C6)-Alkyl", umfassen entsprechend auch geradkettige oder verzweigte Alkylreste mit einer größeren Zahl von C-Atomen, d. h. gemäß Beispiel auch die Alkylreste mit 5 und 6 C-Atomen. 2-butyl, 2-methylpropyl or tert-butyl. General alkyl radicals with a larger specified range of carbon atoms, e.g. B. "(Ci-C 6 ) alkyl", also include straight-chain or branched alkyl radicals with a larger number of carbon atoms, ie, according to the example, also the alkyl radicals with 5 and 6 carbon atoms.
Wenn nicht speziell angegeben, sind bei den Kohlenwasserstoffresten wie Alkyl-, Alkenyl- und Alkinylresten, auch in zusammengesetzten Resten, die niederen Kohlenstoffgerüste, z.B. mit 1 bis 6 C-Atomen bzw. bei ungesättigten Gruppen mit 2 bis 6 C-Atomen, bevorzugt. Alkylreste, auch in den zusammengesetzten Resten wie Alkoxy, Haloalkyl usw., bedeuten z.B. Methyl, Ethyl, n- oder i-Propyl, n-, i-, t- oder 2-Butyl, Pentyle, Hexyle, wie n-Hexyl, i-Hexyl und l,3-Dimethylbutyl, Heptyle, wie n-Heptyl, 1 -Methylhexyl und 1 ,4-Dimethylpentyl; Alkenyl- und Alkinylreste haben die Bedeutung der den Alkylresten entsprechenden möglichen ungesättigten Reste, wobei mindestens eine Doppelbindung bzw. Dreifachbindung enthalten ist. Bevorzugt sind Reste mit einer Doppelbindung bzw. Unless specifically stated, the hydrocarbon radicals such as alkyl, alkenyl and alkynyl radicals, even in composite radicals, are the lower carbon skeletons, e.g. with 1 to 6 carbon atoms or in the case of unsaturated groups with 2 to 6 carbon atoms, preferred. Alkyl radicals, also in the composite radicals such as alkoxy, haloalkyl etc., mean e.g. Methyl, ethyl, n- or i-propyl, n-, i-, t- or 2-butyl, pentyls, hexyls, such as n-hexyl, i-hexyl and 1,3-dimethylbutyl, heptyls, such as n-heptyl, 1-methylhexyl and 1, 4-dimethylpentyl; Alkenyl and alkynyl radicals have the meaning of the possible unsaturated radicals corresponding to the alkyl radicals, at least one double bond or triple bond being present. Residues with a double bond or
Dreifachbindung. Triple bond.
Der Begriff„Alkenyl“ schließt insbesondere auch geradkettige oder verzweigte offenkettige The term “alkenyl” also includes straight-chain or branched open-chain ones
Kohlenwasserstoffreste mit mehr als einer Doppelbindung ein, wie l,3-Butadienyl und 1 ,4-Pentadienyl, aber auch Allenyl- oder Kumulenyl-reste mit einer bzw. mehreren kumulierten Doppelbindungen, wie beispielsweise Allenyl (l,2-Propadienyl), 1 ,2-Butadienyl und l,2,3-Pentatrienyl. Alkenyl bedeutet z.B. Vinyl, welches ggf. durch weitere Alkylreste substituiert sein kann, z B. (aber nicht beschränkt auf) (C2-C6)- Alkenyl wie Ethenyl, l-Propenyl, 2-Propenyl, 1 -Methylethenyl, l-Butenyl, 2-Butenyl, 3- Butenyl, 1 -Methyl- l-propenyl, 2-Methyl- l-propenyl, 1 -Methyl-2-propenyl, 2-Methyl-2-propenyl, 1- Pentenyl, 2-Pentenyl, 3-Pentenyl, 4-Pentenyl, 1 -Methyl- l-butenyl, 2-Methyl- l-butenyl, 3 -Methyl- 1- butenyl, 1 -Methyl-2-butenyl, 2-Methyl-2-butenyl, 3-Methyl-2-butenyl, l-Methyl-3-butenyl, 2-Methyl-3- butenyl, 3-Methyl-3-butenyl, 1 , 1 -Dimethyl-2-propenyl, 1 ,2-Dimethyl- l-propenyl, 1 ,2-Dimethyl-2- propenyl, 1 -Ethyl- l-propenyl, 1 -Ethyl-2-propenyl, l-Hexenyl, 2-Hexenyl, 3-Hexenyl, 4-Hexenyl, 5- Hexenyl, 1 -Methyl- l-pentenyl, 2-Methyl- l-pentenyl, 3-Methyl-l-pentenyl, 4-Methyl-l-pentenyl, 1- Methyl-2-pentenyl, 2-Methyl-2-pentenyl, 3-Methyl-2-pentenyl, 4-Methyl-2-pentenyl, l-Methyl-3- pentenyl, 2-Methyl-3-pentenyl, 3-Methyl-3-pentenyl, 4-Methyl-3-pentenyl, 1 -Methyl-4-pentenyl, 2- Methyl-4-pentenyl, 3-Methyl-4-pentenyl, 4-Methyl-4-pentenyl, 1 , 1 -Dimethyl-2-butenyl, 1 , 1 -Dimethyl-Hydrocarbon radicals with more than one double bond, such as 1,3-butadienyl and 1,4-pentadienyl, but also allenyl or cumulenyl radicals with one or more cumulative double bonds, such as, for example, allenyl (1,2-propadienyl), 1,1 2-butadienyl and l, 2,3-pentatrienyl. Alkenyl means e.g. Vinyl, which may optionally be substituted by further alkyl radicals, for example (but not limited to) (C2-C6) alkenyl such as ethenyl, l-propenyl, 2-propenyl, 1-methylethenyl, l-butenyl, 2-butenyl , 3-butenyl, 1-methyl-1-propenyl, 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4 -Pentenyl, 1-methyl-1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl , l-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1, 1-dimethyl-2-propenyl, 1, 2-dimethyl-l-propenyl, 1, 2-dimethyl -2-propenyl, 1-ethyl-l-propenyl, 1-ethyl-2-propenyl, l-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 1-methyl-l-pentenyl, 2 -Methyl-1-pentenyl, 3-methyl-1-pentenyl, 4-methyl-1-pentenyl, 1-methyl-2-pentenyl, 2-methyl-2-pentenyl, 3-methyl-2-pentenyl, 4-methyl -2-pentenyl, l-methyl-3-pentenyl, 2-methyl-3-pentenyl, 3-methyl-3-pentenyl, 4-methyl-3-pentenyl, 1-methyl-4-pentenyl, 2-met hyl-4-pentenyl, 3-methyl-4-pentenyl, 4-methyl-4-pentenyl, 1, 1 -dimethyl-2-butenyl, 1, 1 -dimethyl-
3-butenyl, 1 ,2-Dimethyl- l-butenyl, 1 ,2-Dimethyl-2-butenyl, l,2-Dimethyl-3-butenyl, l,3-Dimethyl-l- butenyl, l,3-Dimethyl-2-butenyl, l,3-Dimethyl-3-butenyl, 2,2-Dimethyl-3-butenyl, 2,3-Dimethyl-l- butenyl, 2,3-Dimethyl-2-butenyl, 2,3-Dimethyl-3-butenyl, 3,3-Dimethyl-l-butenyl, 3,3-Dimethyl-2- butenyl, l-Ethyl-l -butenyl, l-Ethy 1-2 -butenyl, l-Ethyl- 3 -butenyl, 2-Ethyl-l -butenyl, 2-Ethyl-2-butenyl, 2-Ethyl-3 -butenyl, 1 , 1 ,2-Trimethyl-2-propenyl, 1 -Ethyl- 1 -methyl-2-propenyl, 1 -Ethyl-2-methyl- 1 - propenyl und 1 -Ethyl-2-methyl-2-propenyl. 3-butenyl, 1,2-dimethyl-1-butenyl, 1,2-dimethyl-2-butenyl, 1,2-dimethyl-3-butenyl, 1,3-dimethyl-1- butenyl, l, 3-dimethyl-2-butenyl, l, 3-dimethyl-3-butenyl, 2,2-dimethyl-3-butenyl, 2,3-dimethyl-l-butenyl, 2,3-dimethyl-2- butenyl, 2,3-dimethyl-3-butenyl, 3,3-dimethyl-1-butenyl, 3,3-dimethyl-2-butenyl, 1-ethyl-1-butenyl, 1-ethyl 1-2 -butenyl, 1 -Ethyl-3-butenyl, 2-ethyl-1-butenyl, 2-ethyl-2-butenyl, 2-ethyl-3-butenyl, 1, 1, 2-trimethyl-2-propenyl, 1-ethyl-1-methyl -2-propenyl, 1-ethyl-2-methyl-1-propenyl and 1-ethyl-2-methyl-2-propenyl.
Der Begriff„Alkinyl“ schließt insbesondere auch geradkettige oder verzweigte offenkettige The term “alkynyl” also includes straight-chain or branched open-chain ones
Kohlenwasserstoffreste mit mehr als einer Dreifachbindung oder auch mit einer oder mehreren Hydrocarbon residues with more than one triple bond or with one or more
Dreifachbindungen und einer oder mehreren Doppelbindungen ein, wie beispielsweise l,3-Butatrienyl bzw. 3-Penten-l-in-l-yl. (C2-C6)-Alkinyl bedeutet z.B. Ethinyl, l-Propinyl, 2-Propinyl, l-Butinyl, 2- Butinyl, 3-Butinyl, 1 -Methyl-2-propinyl, l-Pentinyl, 2-Pentinyl, 3-Pentinyl, 4-Pentinyl, l-Methyl-2- butinyl, l-Methyl-3-butinyl, 2-Methyl-3-butinyl, 3 -Methyl- l-butinyl, 1 , 1 -Dimethyl-2-propinyl, l-Ethyl- 2-propinyl, l-Hexinyl, 2-Hexinyl, 3-Hexinyl, 4-Hexinyl, 5-Hexinyl, 1 -Methyl-2-pentinyl, l-Methyl-3- pentinyl, 1 -Methyl-4-pentinyl, 2-Methyl-3-pentinyl, 2-Methyl-4-pentinyl, 3 -Methyl- l-pentinyl, 3- Methyl-4-pentinyl, 4-Methyl- l-pentinyl, 4-Methyl-2-pentinyl, 1 , 1 -Di-methyl-2-butinyl, l,l-Dimethyl-3- butinyl, l,2-Dimethyl-3-butinyl, 2,2-Dimethyl-3-butinyl, 3, 3 -Dimethyl- l-butinyl, 1 -Ethyl-2-butinyl, 1- Ethyl-3-butinyl, 2-Ethyl-3-butinyl und 1 -Ethyl- 1 -methyl-2-propinyl. Triple bonds and one or more double bonds, such as 1, 3-butatrienyl or 3-penten-l-in-l-yl. (C2-C6) alkynyl means e.g. Ethynyl, l-propynyl, 2-propynyl, l-butynyl, 2-butynyl, 3-butynyl, 1-methyl-2-propynyl, l-pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, l-methyl- 2-butynyl, 1-methyl-3-butynyl, 2-methyl-3-butynyl, 3-methyl-1-butynyl, 1, 1-dimethyl-2-propynyl, 1-ethyl-2-propynyl, 1-hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, 5-hexynyl, 1-methyl-2-pentynyl, l-methyl-3-pentynyl, 1-methyl-4-pentynyl, 2-methyl-3-pentynyl, 2- Methyl-4-pentynyl, 3-methyl-1-pentynyl, 3-methyl-4-pentynyl, 4-methyl-1-pentynyl, 4-methyl-2-pentynyl, 1, 1-di-methyl-2-butynyl, l, l-dimethyl-3-butynyl, l, 2-dimethyl-3-butynyl, 2,2-dimethyl-3-butynyl, 3, 3-dimethyl-l-butynyl, 1-ethyl-2-butynyl, 1- Ethyl 3-butynyl, 2-ethyl-3-butynyl and 1-ethyl-1-methyl-2-propynyl.
Der Begriff„Cycloalkyl“ bedeutet ein carbocyclisches, gesättigtes Ringsystem mit vorzugsweise 3-8 Ring-C-Atomen, z.B. Cyclopropyl, Cyclobutyl, Cyclopentyl oder Cyclohexyl, das gegebenenfalls weiter substituiert ist, bevorzugt durch Wasserstoff, Alkyl, Alkoxy, Cyano, Nitro, Alkylthio, Haloalkylthio, Halogen, Alkenyl, Alkinyl, Haloalkyl, AMino, Alkylamino, Bisalkylamino, Alkocycarbonyl, The term "cycloalkyl" means a carbocyclic, saturated ring system with preferably 3-8 ring C atoms, e.g. Cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl, which is optionally further substituted, preferably by hydrogen, alkyl, alkoxy, cyano, nitro, alkylthio, haloalkylthio, halogen, alkenyl, alkynyl, haloalkyl, AMino, alkylamino, bisalkylamino, alkocycarbonyl,
Hydroxycarbonyl, Arylalkoxycarbonyl, Aminocarbonyl, Alkylaminocarbonyl, Hydroxycarbonyl, arylalkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl,
Cycloalkylaminocarbonyl. Im Falle von gegebenenfalls substituiertem Cycloalkyl werden cyclische Systeme mit Substituenten umfasst, wobei auch Substituenten mit einer Doppelbindung am Cycloalkylaminocarbonyl. In the case of optionally substituted cycloalkyl, cyclic systems with substituents are included, substituents also having a double bond on
Cycloalkylrest, z. B. eine Alkylidengruppe wie Methyliden, umfasst sind. Im Falle von gegebenenfalls substituiertem Cycloalkyl werden auch mehrcyclische aliphatische Systeme umfasst, wie beispielsweise Bicyclo[l .1.0]butan-l -yl, Bicyclo[l .1 0]butan-2-yl, Bicyclo[2.1.0]pentan-l -yl, Bicyclo[l .1.1 ]pentan-l - yl, Bicyclo[2.l.0]pentan-2-yl, Bicyclo[2.l.0]pentan-5-yl, Bicyclo[2.l.l]hexyl, Bicyclo[2.2.l]hept-2-yl, Bicyclo[2.2.2]octan-2-yl, Bicyclo[3.2.l]octan-2-yl, Bicyclo[3.2.2]nonan-2-yl, Adamantan-l-yl und Adamantan-2-yl, aber auch Systeme wie z. B. l,l'-Bi(cyclopropyl)-l-yl, l,l'-Bi(cyclopropyl)-2-yl. Der Ausdruck "(C3-C7)-Cycloalkyl" bedeutet eine Kurzschreibweise für Cycloalkyl mit drei bis 7 Cycloalkyl radical, e.g. B. an alkylidene group such as methylidene are included. In the case of optionally substituted cycloalkyl, multi-cyclic aliphatic systems are also included, such as, for example, bicyclo [1.0] butan-1-yl, bicyclo [1.0] butan-2-yl, bicyclo [2.1.0] pentan-1 - yl, bicyclo [l. 1.1] pentan-1-yl, bicyclo [2.l.0] pentan-2-yl, bicyclo [2.l.0] pentan-5-yl, bicyclo [2.ll] hexyl, Bicyclo [2.2.l] hept-2-yl, bicyclo [2.2.2] octan-2-yl, bicyclo [3.2.l] octan-2-yl, bicyclo [3.2.2] nonan-2-yl, adamantane l-yl and adamantan-2-yl, but also systems such as B. l, l'-Bi (cyclopropyl) -l-yl, l, l'-Bi (cyclopropyl) -2-yl. The term "(C3-C7) cycloalkyl" means a shorthand notation for cycloalkyl of three to seven
Kohlenstoffatomen entsprechend der Bereichsangabe für C-Atome. Carbon atoms according to the range for carbon atoms.
Im Falle von substituiertem Cycloalkyl werden auch spirocyclische aliphatische Systeme umfasst, wie beispielsweise Spiro[2.2]pent-l-yl, Spiro[2.3]hex-l-yl, Spiro[2.3]hex-4-yl, 3-Spiro[2.3]hex-5-yl,In the case of substituted cycloalkyl, spirocyclic aliphatic systems are also included, such as, for example, spiro [2.2] pent-l-yl, spiro [2.3] hex-l-yl, spiro [2.3] hex-4-yl, 3-spiro [2.3] hex-5-yl,
Spiro [3.3 ]hept- 1 -yl, Spiro[3.3 ]hept-2-yl. „Cycloalkenyl“ bedeutet ein carbocyclisches, nicht aromatisches, partiell ungesättigtes Ringsystem mit vorzugsweise 4-8 C-Atomen, z.B. 1 -Cyclobutenyl, 2-Cyclobutenyl, 1 -Cyclopentenyl, 2-Cyclopentenyl, 3-Cyclopentenyl, oder 1 -Cyclohexenyl, 2-Cyclohexenyl, 3-Cyclohexenyl, l,3-Cyclohexadienyl oder 1 ,4-Cyclohexadienyl, wobei auch Substituenten mit einer Doppelbindung am Cycloalkenylrest, z. B. eine Alkylidengruppe wie Methyliden, umfasst sind. Im Falle von gegebenenfalls substituiertem Cycloalkenyl gelten die Erläuterungen für substituiertes Cycloalkyl entsprechend. Spiro [3.3] hept-1-yl, Spiro [3.3] hept-2-yl. “Cycloalkenyl” means a carbocyclic, non-aromatic, partially unsaturated ring system with preferably 4-8 C atoms, for example 1-cyclobutenyl, 2-cyclobutenyl, 1-cyclopentenyl, 2-cyclopentenyl, 3-cyclopentenyl, or 1-cyclohexenyl, 2- Cyclohexenyl, 3-cyclohexenyl, l, 3-cyclohexadienyl or 1, 4-cyclohexadienyl, where also substituents with a double bond on the cycloalkenyl radical, e.g. B. an alkylidene group such as methylidene are included. In the case of optionally substituted cycloalkenyl, the explanations for substituted cycloalkyl apply accordingly.
Der Begriff„Alkyliden“, z. B. auch in der Form (Ci-Cio)-Alkyliden, bedeutet den Rest eines geradkettigen oder verzweigten offenkettigen Kohlenwasserstoffrests, der über eine Zweifachbindung gebunden ist. Als Bindungsstelle für Alkyliden kommen naturgemäß nur Positionen am Grundkörper in Frage, an denen zwei H- Atome durch die Doppelbindung ersetzt werden können; Reste sind z. B. =CH2, =CH-CH3, =C(CH3)-CH3, =C(CH3)-C2H5 oder =C(C2FF)-C'2FF. Cycloalkyliden bedeutet ein The term "alkylidene", e.g. B. also in the form (Ci-Cio) alkylidene, means the remainder of a straight-chain or branched open-chain hydrocarbon radical which is bonded via a double bond. As a binding site for alkylidene there are naturally only positions on the base in which two H atoms can be replaced by the double bond; Leftovers are e.g. B. = CH2, = CH-CH3, = C (CH3) -CH3, = C (CH 3 ) -C 2 H5 or = C (C2FF) -C ' 2FF . Cycloalkylidene means a
carbocyclischer Rest, der über eine Zweifachbindung gebunden ist. carbocyclic radical which is bonded via a double bond.
„Cycloalkylalkyloxy“ bedeutet ein über ein Sauerstoffatom gebundenen Cycloalkylalkylrest und „Arylalkyloxy“ bedeutet ein über ein Sauerstoffatom gebundenen Arylalkylrest. “Cycloalkylalkyloxy” means a cycloalkylalkyl radical bonded via an oxygen atom and “arylalkyloxy” means an arylalkyl radical bonded via an oxygen atom.
„Alkoxyalkyl“ steht für einen über eine Alkylgruppe gebundenen Alkoxyrest und„Alkoxyalkoxy“ bedeutet einen über ein Sauerstoffatom gebundenen Alkoxyalkylrest, z.B. (aber nicht beschränkt auf) Methoxymethoxy, Methoxyethoxy, Ethoxyethoxy, Methoxy-n-propyloxy. "Alkoxyalkyl" stands for an alkoxy radical bonded via an alkyl group and "alkoxyalkoxy" means an alkoxyalkyl radical bonded via an oxygen atom, e.g. (but not limited to) methoxymethoxy, methoxyethoxy, ethoxyethoxy, methoxy-n-propyloxy.
„Alkylthioalkyl“ steht für einen über eine Alkylgruppe gebundenen Alkylthiorest und “Alkylthioalkyl” stands for an alkylthio radical bonded via an alkyl group and
„Alkylthioalkylthio“ bedeutet einen über ein Sauerstoffatom gebundenen Alkylthioalkylrest. “Alkylthioalkylthio” means an alkylthioalkyl radical bonded via an oxygen atom.
„Arylalkoxyalkyl“ steht für einen über eine Alkylgruppe gebundenen Aryloxyrest und "Arylalkoxyalkyl" stands for an aryloxy radical bonded via an alkyl group and
„Heteroaryloxyalkyl“ bedeutet einen über eine Alkylgruppe gebundenen Heteroaryloxyrest. “Heteroaryloxyalkyl” means a heteroaryloxy radical bonded via an alkyl group.
„Haloalkoxyalkyl“ steht für einen gebundenen Haloalkoxyrest und„Haloalkylthioalkyl“ bedeutet einen über eine Alkylgruppe gebundenen Haloalkylthiorest. “Haloalkoxyalkyl” stands for a bound haloalkoxy radical and “Haloalkylthioalkyl” means a haloalkylthio radical bound via an alkyl group.
„Arylalkyl“ steht für einen über eine Alkylgruppe gebundenen Arylrest,„Heteroarylalkyl“ bedeutet einen über eine Alkylgruppe gebundenen Heteroarylrest, und„Heterocyclylalkyl“ bedeutet einen über eine Alkylgruppe gebundenen Heterocyclylrest. „Cycloalkylalkyl“ steht für einen über eine Alkylgruppe gebundenen Cycloalkylrest, z. B. (aber nicht beschränkt auf) Cyclopropylmethyl, Cyclobutylmethyl, Cyclopentylmethyl, Cyclohexylmethyl, 1 - Cyclopropyleth-l -yl, 2-Cyclopropyleth-l-yl, l-Cyclopropylprop-l-yl, 3-Cyclopropylprop-l-yl. “Arylalkyl” stands for an aryl radical bonded via an alkyl group, “heteroarylalkyl” means a heteroaryl radical bonded via an alkyl group, and “heterocyclylalkyl” means a heterocyclyl radical bonded via an alkyl group. “Cycloalkylalkyl” stands for a cycloalkyl radical bonded via an alkyl group, eg. B. (but not limited to) cyclopropylmethyl, cyclobutylmethyl, cyclopentylmethyl, cyclohexylmethyl, 1 - cyclopropyleth-l -yl, 2-cyclopropyleth-l-yl, l-cyclopropylprop-l-yl, 3-cyclopropylprop-l-yl.
„Arylalkenyl“ steht für einen über eine Alkenylgruppe gebundenen Arylrest,„Heteroarylalkenyl“ bedeutet einen über eine Alkenylgruppe gebundenen Heteroarylrest, und„Heterocyclylalkenyl“ bedeutet einen über eine Alkenylgruppe gebundenen Heterocyclylrest. “Arylalkenyl” stands for an aryl radical bonded via an alkenyl group, “heteroarylalkenyl” means a heteroaryl radical bonded via an alkenyl group, and “heterocyclylalkenyl” means a heterocyclyl radical bonded via an alkenyl group.
„Arylalkinyl“ steht für einen über eine Alkinylgruppe gebundenen Arylrest,„Heteroarylalkinyl“ bedeutet einen über eine Alkinylgruppe gebundenen Heteroarylrest, und„Heterocyclylalkinyl“ bedeutet einen über eine Alkinylgruppe gebundenen Heterocyclylrest. “Arylalkynyl” stands for an aryl group bonded via an alkynyl group, “heteroarylalkynyl” means a heteroaryl group bonded via an alkynyl group, and “heterocyclylalkynyl” means a heterocyclyl group bonded via an alkynyl group.
Erfindungsgemäß steht "Haloalkylthio" - in Alleinstellung oder als Bestandteil einer chemischen Gruppe - für geradkettiges oder verzweigtes S -Halogenalkyl, vorzugsweise mit 1 bis 8, oder mit 1 bis 6 According to the invention, "haloalkylthio" - alone or as part of a chemical group - stands for straight-chain or branched S -haloalkyl, preferably with 1 to 8, or with 1 to 6
Kohlenstoffatomen, wie (Ci-Cs)-, (CI-CÖ)- oder (Ci-C4)-Haloalkylthio, z.B. (aber nicht beschränkt auf) Trifluormethylthio, Pentafluorethylthio, Difluormethyl, 2,2-Difluoreth-l-ylthio, 2,2,2-Difluoreth-l- ylthio, 3,3,3-prop-l-ylthio. Carbon atoms, such as (Ci-Cs) -, (C I -C Ö ) - or (Ci-C4) -haloalkylthio, for example (but not limited to) trifluoromethylthio, pentafluoroethylthio, difluoromethyl, 2,2-difluoroeth-1-ylthio, 2,2,2-difluoroeth-l-ylthio, 3,3,3-prop-l-ylthio.
„Halocycloalkyl“ und„Halocycloalkenyl“ bedeuten durch gleiche oder verschiedene Halogenatome, wie z. B. F, CI und Br, oder durch Haloalkyl, wie z. B. Trifluormethyl oder Difluormethyl teilweise oder vollständig substituiertes Cycloalkyl oder Cycloalkenyl , z.B. l-Fluorcycloprop-l-yl, 2-Fluorcycloprop- l-yl, 2,2-Difluorcycloprop-l-yl, l-Fluorcyclobut-l-yl, l-Trifluormethylcycloprop-l-yl, 2- Trifluormethylcycloprop- 1 -yl, 1 -Chlor-cycloprop- 1 -yl, 2-Chlorcycloprop- 1 -yl, 2,2-Dichlorcycloprop- 1 - yl, 3,3-Difluorcyclobutyl. "Halocycloalkyl" and "Halocycloalkenyl" mean by identical or different halogen atoms, such as. B. F, CI and Br, or by haloalkyl, such as. B. trifluoromethyl or difluoromethyl partially or fully substituted cycloalkyl or cycloalkenyl, e.g. l-fluorocycloprop-l-yl, 2-fluorocycloprop-l-yl, 2,2-difluorocycloprop-l-yl, l-fluorocyclobut-l-yl, l-trifluoromethylcycloprop-l-yl, 2-trifluoromethylcycloprop-1-yl, 1-chloro-cycloprop-1-yl, 2-chlorocycloprop-1-yl, 2,2-dichlorocycloprop-1-yl, 3,3-difluorocyclobutyl.
Wenn die Verbindungen durch Wasserstoffverschiebung Tautomere bilden können, welche strukturell formal nicht durch die Formel (I) erfasst würden, so sind diese Tautomere gleichwohl von der Definition der erfindungsgemäßen Verbindungen der Formel (I) umfasst, sofern nicht ein bestimmtes Tautomer Gegenstand der Betrachtung ist. So können beispielsweise viele Carbonylverbindungen sowohl in der Ketoform wie auch in der Enolform vorliegen, wobei beide Formen durch die Definition der If the compounds can form tautomers by hydrogen shift, which would not be structurally formally covered by the formula (I), these tautomers are nevertheless included in the definition of the compounds of the formula (I) according to the invention, unless a specific tautomer is the subject of consideration. For example, many carbonyl compounds can exist both in the keto form and in the enol form, both forms being defined by the
Verbindung der Formel (I) umfasst werden. Compound of formula (I) are included.
Die Verbindungen der allgemeinen Formel (I) können je nach Art und Verknüpfung der Substituenten als Stereoisomere vorliegen. Die durch ihre spezifische Raumform definierten möglichen Stereoisomere, wie Enantiomere, Diastereomere, Z- und E-Isomere sind alle von der Formel (I) umfasst. Sind beispielsweise eine oder mehrere Alkenylgruppen vorhanden, so können Diastereomere (Z- und E- Isomere) auftreten. Sind beispielsweise ein oder mehrere asymmetrische Kohlenstoffatome vorhanden, so können Enantiomere und Diastereomere auftreten. Stereoisomere lassen sich aus den bei der Herstellung anfallenden Gemischen nach üblichen Trennmethoden erhalten. Die chromatographische Trennung kann sowohl im analytischen Maßstab zur Feststellung des Enantiomerenüberschusses bzw. des Diastereomerenüberschusses, wie auch im präparativen Maßstab zur Herstellung von Prüfmustern für die biologische Ausprüfung erfolgen. Ebenso können Stereoisomere durch Einsatz stereoselektiver Reaktionen unter Verwendung optisch aktiver Ausgangs- und/oder Hilfsstoffe selektiv hergestellt werden. Die Erfindung betrifft somit auch alle Stereoisomeren, die von der allgemeinen Formel (I) umfasst, jedoch nicht mit ihrer spezifischen Stereoform angegeben sind, sowie deren Gemische. Depending on the nature and linkage of the substituents, the compounds of the general formula (I) can be present as stereoisomers. The possible stereoisomers defined by their specific spatial shape, such as enantiomers, diastereomers, Z and E isomers, are all encompassed by the formula (I). If, for example, one or more alkenyl groups are present, diastereomers (Z and E Isomers) occur. For example, if one or more asymmetric carbon atoms are present, enantiomers and diastereomers can occur. Stereoisomers can be obtained from the mixtures obtained in the preparation by customary separation methods. The chromatographic separation can be carried out on an analytical scale to determine the enantiomeric excess or the diastereomeric excess, as well as on a preparative scale for the production of test samples for biological testing. Likewise, stereoisomers can be produced selectively by using stereoselective reactions using optically active starting materials and / or auxiliary substances. The invention thus also relates to all stereoisomers which are encompassed by the general formula (I) but are not specified with their specific stereoform, and to mixtures thereof.
Sofern die Verbindungen als Feststoffe erhalten werden, kann die Reinigung auch durch If the compounds are obtained as solids, the cleaning can also be carried out by
Umkristallisieren oder Digerieren erfolgen. Sofern einzelne Verbindungen der allgemeinen Formel (I) nicht auf den nachstehend beschriebenen Wegen zufriedenstellend zugänglich sind, können sie durch Derivatisierung anderer Verbindungen der allgemeinen Formel (I) hergestellt werden. Recrystallize or digest. If individual compounds of the general formula (I) are not satisfactorily accessible in the ways described below, they can be prepared by derivatizing other compounds of the general formula (I).
Als Isolierungs-, Reinigungs- und Stereoisomerenauftrennungsverfahren von Verbindungen der allgemeinen Formel (I) kommen Methoden in Frage, die dem Fachmann aus analogen Fällen allgemein bekannt sind, z.B. durch physikalische Verfahren wie Kristallisation, Chromatographieverfahren, vor allem Säulenchromatographie und HPLC (Hochdruckflüssigchromatographie), Destillation, gegebenenfalls unter reduziertem Druck, Extraktion und andere Verfahren, können gegebenfalls verbleibende Gemische in der Regel durch chromatographische Trennung, z.B. an chiralen Festphasen, getrennt werden. Für präparative Mengen oder im industriellen Maßstab kommen Verfahren in Frage wie Kristallisation, z.B. diastereomerer Salze, die aus den Diastereomerengemischen mit optisch aktiven Säuren und gegebenenfalls bei vorhandenen sauren Gruppen mit optisch aktiven Basen erhalten werden können. Methods for isolation, purification and stereoisomer separation of compounds of the general formula (I) are those which are generally known to the person skilled in the art from analogous cases, e.g. by physical processes such as crystallization, chromatography processes, especially column chromatography and HPLC (high pressure liquid chromatography), distillation, optionally under reduced pressure, extraction and other processes, any remaining mixtures can usually be separated by chromatographic separation, e.g. on chiral solid phases. For preparative quantities or on an industrial scale, methods such as crystallization, e.g. diastereomeric salts which can be obtained from the diastereomer mixtures with optically active acids and, if appropriate, with acid groups present with optically active bases.
Synthese von substituierten Thiazolylpyrrolonen der allgemeinen Formel (I). Synthesis of substituted thiazolylpyrrolones of the general formula (I).
Die erfindungsgemäßen substituierten Thiazolylpyrrolonen der allgemeinen Formel (I) können ausgehend von bekannten Verfahren hergestellt werden. Die eingesetzten und untersuchten Syntheserouten gehen dabei von kommerziell erhältlichen oder leicht herstellbaren substituierten 2- Aminothiazolen und von entsprechend substituierten Furanonen oder Furandionen aus. Die The substituted thiazolylpyrrolones of the general formula (I) according to the invention can be prepared starting from known processes. The used and examined Synthesis routes are based on commercially available or easily manufactured substituted 2-aminothiazoles and on appropriately substituted furanones or furandions. The
Gruppierungen Q, X, Y, A1, A2, R1, R2, R3, R4, R5, R6, R7, R8 und R9 der allgemeinen Formel (I) haben in den nachfolgenden Schemata die zuvor definierten Bedeutungen, sofern nicht beispielhafte, aber nicht einschränkende, Definitionen erfolgen. Groupings Q, X, Y, A 1 , A 2 , R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 and R 9 of the general formula (I) have in the schemes below the previously defined meanings, unless exemplary but not restrictive definitions.
Die Synthese der erfindungsgemäßen Verbindungen der allgemeinen Formel (Ib) erfolgt über eine Reaktion eines Hydroxypyrrolons der allgemeinen Formel (Ia) mit einem Halogenameisensäure-Derivat der allgemeinen Formel (II). Die Reaktion findet bevorzugt in dem Temperaturbereich zwischen -20 °C und 80 °C, in einem adäquaten Lösungsmittel wie zum Beispiel Dichlormethan, Acetonitril, NN-The compounds of the general formula (Ib) according to the invention are synthesized via a reaction of a hydroxypyrrolone of the general formula (Ia) with a halogen formic acid derivative of the general formula (II). The reaction preferably takes place in the temperature range between -20 ° C and 80 ° C, in an adequate solvent such as dichloromethane, acetonitrile, NN-
Dimethylformamid oder Ethylacetat und in Gegenwart eine Base wie zum Beispiel Triethylamin, N,N- Diisopropylethylamin oder l,8-Diazabicyclo[5.4.0]undec-7-cen statt (siehe Schema 1). Dimethylformamide or ethyl acetate and in the presence of a base such as triethylamine, N, N-diisopropylethylamine or l, 8-diazabicyclo [5.4.0] undec-7-cene instead (see scheme 1).
(la) (II) (Ib) Mit R = (Ci-C i)-Alkyl und X' = Halogen. ( la ) (II) (Ib) With R = (Ci-C i) alkyl and X '= halogen.
Schema 1 Scheme 1
Die Synthese der erfindungsgemäßen Verbindungen der allgemeinen Formel (Ic) erfolgt über eine Reaktion eines Hydroxypyrrolons der allgemeinen Formel (Ia) mit einem Säurehalogenid-Derivat der allgemeinen Formel (III). Die Reaktion findet bevorzugt in dem Temperaturbereich zwischen -20 °C und 80 °C, in einem adäquaten Lösungsmittel wie zum Beispiel Dichlormethan, Acetonitril, N,N- Dimethyl-formamid oder Ethylacetat und in Gegenwart eine Base wie zum Beispiel Triethylamin, V,V- Diisopropylethylamin oder l,8-Diazabicyclo[5.4.0]undec-7-cen statt (siehe Schema 2). The compounds of the general formula (Ic) according to the invention are synthesized by reacting a hydroxypyrrolone of the general formula (Ia) with an acid halide derivative of the general formula (III). The reaction preferably takes place in the temperature range between -20 ° C and 80 ° C, in an adequate solvent such as dichloromethane, acetonitrile, N, N-dimethylformamide or ethyl acetate and in the presence of a base such as triethylamine, V, V - diisopropylethylamine or 1,8-diazabicyclo [5.4.0] undec-7-cene instead (see Scheme 2).
(la) (III) (Ic) ( la ) (III) (Ic)
Mit R = (Ci-C4)-Alkyl und X' = Halogen. With R = (Ci-C4) alkyl and X '= halogen.
Schema 2 Die Synthese der erfindungsgemäßen Verbindungen der allgemeinen Formel (Id) erfolgt über eine Reaktion eines Hydroxypyrrolons der allgemeinen Formel (Ia) mit einem Isocyanat der allgemeinen Formel (IV). Die Reaktion findet bevorzugt in dem Temperaturbereich zwischen -20 °C und 120 °C, in einem adäquaten Lösungsmittel wie zum Beispiel Dichlormethan, Acetonitril, ,V, ,V- D i m c t hy 1 - fo rm am i d oder Ethylacetat und in Gegenwart eine Base wie zum Beispiel Triethylamin, /V, ,V- D i i s o p ro p y 1 c t h y 1 a m i n oder l,8-Diazabicyclo[5.4.0]undec-7-cen statt (siehe Schema 3). The synthesis of the compounds of the general formula (Id) according to the invention takes place via a reaction of a hydroxypyrrolone of the general formula (Ia) with an isocyanate of the general formula (IV). The reaction preferably takes place in the temperature range between -20 ° C and 120 ° C, in an adequate solvent such as dichloromethane, acetonitrile,, V,, V-D imct hy 1 - form am id or ethyl acetate and in the presence of a base such as triethylamine, / V,, V- diisopro py 1 cthy 1 amine or l, 8-diazabicyclo [5.4.0] undec-7-cene instead (see Scheme 3).
(la) (IV) (Id) ( la ) (IV) (Id)
Mit R = (Ci-C4)-Alkyl. With R = (Ci-C 4 ) alkyl.
Schema 3 Scheme 3
Die Synthese der erfindungsgemäßen Verbindungen der allgemeinen Formel (Ia) und (Ie) erfolgt über eine Reduktion eines substituierten Maleinsäureimids der allgemeinen Formel (V) mit einem The compounds of the general formula (Ia) and (Ie) according to the invention are synthesized by reducing a substituted maleimide of the general formula (V) with a
Reduktionsmittel wie zum Beispiel Natriumhydrid, Lithiumaluminiumhydrid, Natriumborhydrid oder andere Wasserstoff entwickelnde Metallhydride. Es kann alternativ auch eine Übergangsmetall- vermittelte Hydrierung durchgeführt werden (vgl. CH633678, DE2247266, WO2015/018434). Da die Gruppen X und Y verschieden sind, kann die Reduktion der Carbonylgruppe Gemische von Reducing agents such as sodium hydride, lithium aluminum hydride, sodium borohydride or other hydrogen-generating metal hydrides. Alternatively, a transition metal-mediated hydrogenation can also be carried out (cf. CH633678, DE2247266, WO2015 / 018434). Since the groups X and Y are different, the reduction of the carbonyl group can be mixtures of
Regioisomeren liefern, daher sind im nachfolgenden Schema 4 beide möglichen Regioisomere abgebildet, um dies zu verdeutlichen. Die Reaktion findet bevorzugt in dem Temperaturbereich zwischen -20 °C und 120 °C, in einem adäquaten Lösungsmittel wie zum Beispiel Tetrahydrofuran, Methanol oder Ethanol statt. Provide regioisomers, therefore in scheme 4 below both are possible regioisomers to illustrate this. The reaction preferably takes place in the temperature range between -20 ° C and 120 ° C in an adequate solvent such as tetrahydrofuran, methanol or ethanol.
(V) (la) (le)(V) (la) (le)
Schema 4 Scheme 4
Substituierte Maleinimide der allgemeinen Formel (V) lassen sich durch Kondensation eines substituierten Aminothiazols der allgemeinen Formel (VI) mit einem substituierten Furan-2,5-dion der allgemeinen Formel (VII) in Gegenwart einer Broensted-Säure wie zum Beispiel Essigsäure bzw. Substituted maleimides of the general formula (V) can be obtained by condensation of a substituted aminothiazole of the general formula (VI) with a substituted furan-2,5-dione of the general formula (VII) in the presence of a Broensted acid such as acetic acid or
Essigsäureanhydrid oder p-Toluolsulfonsäure bzw. Amberlyst 15, in einem geeigneten Lösungsmittel wie zum Beispiel Toluol hersteilen (Schema 5). Prepare acetic anhydride or p-toluenesulfonic acid or Amberlyst 15 in a suitable solvent such as toluene (Scheme 5).
(VI) (VII) (V) Schema 5 (VI) (VII) (V) Scheme 5
Die monosubstituierte Furan-2,5-dione (VII), die auch als Maleinsäureanhydride bezeichnet werden, können über literaturbekannte Syntheseorschriften hergestellt werden (vgl. J. Chem. Soc., Perkin Trans. 1, 1982, 215; EP1426365; J. Org. Chem. 1998, 63, 2646; WO2015/018431; Tetrahedron 2012, 68, 5863; Russian J. Org. Chem. 2007, 43, 801), beispielsweise Verbindungen (VII. a) und (Vll.b) im The monosubstituted furan-2,5-diones (VII), which are also referred to as maleic anhydrides, can be prepared via synthesis instructions known from the literature (cf. J. Chem. Soc., Perkin Trans. 1, 1982, 215; EP1426365; J. Org Chem. 1998, 63, 2646; WO2015 / 018431; Tetrahedron 2012, 68, 5863; Russian J. Org. Chem. 2007, 43, 801), for example compounds (VII. A) and (Vll.b) im
nachfolgenden Schema 6 in mehreren Schritten aus einem geeigneten Acetylendicarbonsäureester Scheme 6 below in several steps from a suitable acetylenedicarboxylic acid ester
(VIII). Im nachfolgenden Schema 6 steht X beispielhaft, aber nicht einschränkend jeweils für C-CFL und C-C2H5, und Y steht beispielhaft, aber nicht einschränkend, für C-H. 1.NaOH, H20, EtOH (VIII). In Scheme 6 below, X is exemplary but not limiting for C-CFL and CC 2 H 5 , and Y is exemplary but not limiting for CH. 1. NaOH, H 2 0, EtOH
°gg 2. HCl, H20 ° gg 2. HCl, H 2 0
1 .Grignard-Reaktion 1. Grignard reaction
2.NH4CI, H20 2.NH 4 CI, H 2 0
Schema 6 Scheme 6
Monosubstituierte Thiazole (VI) können über literaturbekannte Synthesevorschriften hergestellt werden (vgl. Bioorg. & MedChem. Let. 2004, 14, 5521-5525; Heterocyclic Chemistry, Ed. J.A. Joule and K. Mills, 4th Edition, Blackwell Publishing, 2000, 418-420; W02008/55840; US2012/22067; Mono-substituted thiazoles (VI) can be prepared by literature known synthetic procedures (see Bioorg & MedChem Let 2004, 14, 5521-5525;.... Heterocyclic Chemistry, Ed JA Joule and K. Mills, 4th Edition, Blackwell Publishing, 2000. 418-420; W02008 / 55840; US2012 / 22067;
WO2012/93809 US2009/143448; Name Reactions in Heterocyclic Chemistry, Ed. J.J. Li, Wiley, 2005, 275283; The Chemistry of Heterocycles, Ed. T. Eicher and S. Hauptmann, 2nd Edition, 2003, 149-155). Die Verbindung (Vl.a) lässt sich ausgehend von einem Haloketon (X) und Thiohamstoff (XI) hersteilen. Das Aminothiazol (Vl.b) ist aus dem Haloaldehyd (XII) und Thiohamstoff (XI) herstellbar (Schema 7). WO2012 / 93809 US2009 / 143448; Name Reactions in Heterocyclic Chemistry, Ed. JJ Li, Wiley, 2005, 275283; The Chemistry of Heterocycles, Ed. T. Eicher and S. Hauptmann, 2 nd Edition, 2003, 149-155). The compound (Vl.a) can be made from a haloketone (X) and thiourea (XI). The aminothiazole (Vl.b) can be prepared from the haloaldehyde (XII) and thiourea (XI) (Scheme 7).
(XII) (XI) (Vl b) (XII) (XI) ( Vl b )
Mit R = (Ci-C4)-Alkyl, (C3-Cv)-Cycloalyl, Aryl, Hetaryl, Heterocyclyl und X' = Halogen. With R = (Ci-C4) alkyl, (C3-Cv) cycloalyl, aryl, hetaryl, heterocyclyl and X '= halogen.
Schema 7 Alternativ können einfach substituierte Thiazolylpyrrolone der allgemeinen Formel (Ia) ausgehend von der Umsetzung eines Hydroxy- oder Bromolactons (XIII) mit einem geeigneten gegebenenfalls substituierten Thiazolylamin (VI) in einem geeigneten Lösemittel (z. B. Toluol) bei erhöhter Temperatur hergestellt werden. Durch weitere Reaktion des resultierenden Intermediates (XIV) mit Scheme 7 Alternatively, simply substituted thiazolylpyrrolones of the general formula (Ia) can be prepared starting from the reaction of a hydroxy- or bromolactone (XIII) with a suitable optionally substituted thiazolylamine (VI) in a suitable solvent (for example toluene) at elevated temperature. By further reaction of the resulting intermediate (XIV) with
Essigsäureanhydrid unter Verwendung einer geeigneten Base (z. B. Pyridin) bei erhöhter Temperatur wird das einfach substituierte Thiazolylpyrrolon der allgemeinen Formel (XV) mit O-Acetylgruppe gebildet, das durch Erhitzen unter sauren Bedingungen (z. B. in einem Gemisch aus Essigsäure und Wasser) in das entsprechende einfach substituierte Thiazolylpyrrolone der allgemeinen Formel (Ia) mit freier OH-Gruppe überführt werden kann. Im nachfolgenden Schema 6 haben X, Y, A1 und A2 die zuvor definierten Bedeutungen. Acetic anhydride using a suitable base (e.g. pyridine) at elevated temperature forms the monosubstituted thiazolylpyrrolone of the general formula (XV) with O-acetyl group, which can be obtained by heating under acidic conditions (e.g. in a mixture of acetic acid and Water) can be converted into the corresponding monosubstituted thiazolylpyrrolone of the general formula (Ia) with a free OH group. In the following scheme 6, X, Y, A 1 and A 2 have the meanings defined above.
Schema 8. Die Synthese der erfindungsgemäßen Verbindungen der allgemeinen Formel (If) erfolgt über eine Scheme 8. The synthesis of the compounds of general formula (If) according to the invention takes place via a
Reaktion eines Hydroxypyrrolons der allgemeinen Formel (Ia) mit einem Halosulfonsäure-Derivat der allgemeinen Formel (XVI). Die Reaktion findet bevorzugt in dem Temperaturbereich zwischen -20 °C und 80 °C, in einem adäquaten Lösungsmittel wie zum Beispiel Dichlormethan, Acetonitril, NN- Dimethyl-formamid oder Ethylacetat und in Gegenwart eine Base wie zum Beispiel Triethylamin, V,V- Diisopropylethylamin oder l,8-Diazabicyclo[5.4.0]undec-7-cen statt (siehe Schema 9). Reaction of a hydroxypyrrolone of the general formula (Ia) with a halosulfonic acid derivative of the general formula (XVI). The reaction preferably takes place in the temperature range between -20 ° C and 80 ° C, in an adequate solvent such as dichloromethane, acetonitrile, NN-dimethylformamide or ethyl acetate and in the presence of a base such as triethylamine, V, V-diisopropylethylamine or 1,8-diazabicyclo [5.4.0] undec-7-cene instead (see Scheme 9).
(la) (XVI) (If) (la) (XVI) (If)
Mit R = (Ci-C i)-Alkyl, (C3-Cv)-Cycloalkyl und X' = Halogen. With R = (Ci-C i) alkyl, (C3-Cv) cycloalkyl and X '= halogen.
Schema 9 Ausgewählte detaillierte Synthesebeispiele für die erfindungsgemäßen Verbindungen der allgemeinen Formel (1) sind im Folgenden aufgeführt. Die angegebenen Beispielnummem entsprechen den in den nachstehenden Tabellen 1.1 bis 1.124 genannten Nummerierungen. Die H-NMR-, 13C-NMR- und 19F-Scheme 9 Selected detailed synthesis examples for the compounds of the general formula (1) according to the invention are listed below. The example numbers given correspond to the numbering given in Tables 1.1 to 1.124 below. The H-NMR, 13 C-NMR and 19 F-
NMR-spektroskopischen Daten, die für die in den nachfolgenden Abschnitten beschriebenen chemischen Beispiele angegeben sind, (400 MHz bei *H-NMR und 150 MHz bei 13C-NMR und 375 MHz bei 19F-NMR, Lösungsmittel CDCL, CD3OD oder ck-DMSO, interner Standard: Tetramethylsilan d = 0.00 ppm), wurden mit einem Gerät der Firma Broker erhalten, und die bezeichneten Signale haben die nachfolgend aufgeführten Bedeutungen: br = breit(es); s = Singulett, d = Dublett, t = Triplet, dd = Doppeldublet, ddd = Dublett eines Doppeldubletts, m = Multiplet, q = Quartet, quint = Quintett, sext = Sextett, sept = Septett, dq = Doppelquartet, dt = Doppeltriplet. Bei Diastereomerengemischen werden entweder die jeweils signifikanten Signale beider Diastereomere oder das charakteristische Signal des Hauptdiastereomers angegeben. Die verwendeten Abkürzungen für chemische Gruppen haben beispielsweise die nachfolgenden Bedeutungen: Me = CH3, Et = CH2CH3, t-Hex = C(CH3)2CH(CH3)2, t- Bu = C(CH3)3, n-Bu = unverzweigtes Butyl, n-Pr = unverzweigtes Propyl, i-Pr = verzweigtes Propyl, c- Pr = Cyclopropyl, c-Hex = Cyclohexyl. NMR spectroscopic data given for the chemical examples described in the following sections (400 MHz for * H NMR and 150 MHz for 13 C NMR and 375 MHz for 19 F NMR, solvent CDCL, CD3OD or ck- DMSO, internal standard: tetramethylsilane d = 0.00 ppm), were obtained with a Broker device, and the signals indicated have the meanings given below: br = broad (es); s = singlet, d = doublet, t = triplet, dd = double doublet, ddd = doublet of a double doublet, m = multiplet, q = quartet, quint = quintet, sext = sextet, sept = septet, dq = double quartet, dt = double triplet. In the case of mixtures of diastereomers, either the respectively significant signals of both diastereomers or the characteristic signal of the main diastereomer are given. The abbreviations for chemical groups used have, for example, the following meanings: Me = CH3, Et = CH2CH3, t-Hex = C (CH3) 2CH (CH3) 2, t-Bu = C (CH3) 3, n-Bu = unbranched butyl , n-Pr = unbranched propyl, i-Pr = branched propyl, c-Pr = cyclopropyl, c-Hex = cyclohexyl.
Synthesebeispiele: Synthesis Examples:
Synthesebeispiel: No. 1.2-20: 5-Hydroxy-3-methyl-l-[4-(l-methylcyclopropyl)-l,3-thiazol-2-yl]-l,5- dihydro-2H-pyrrol-2-on Synthesis example: No. 1.2-20: 5-Hydroxy-3-methyl-1- [4- (l-methylcyclopropyl) -1, 3-thiazol-2-yl] -1, 5-dihydro-2H-pyrrol-2-one
Synthesestufe 1 : 2-Brom-l-(l -methylcyclopropyl)ethanon l-(l-Methylcyclopropyl)ethanon (6,00 g, 61,13 mmol, 1,0 equiv.) wurde in Methanol (40 mL) gelöst und mit einem Eisbad auf ca. 4 °C gekühlt. Zu der Lösung tropfte man Brom (3,15 mL, 61,13 mmol,Synthesis stage 1: 2-bromo-l- (l -methylcyclopropyl) ethanone l- (l-Methylcyclopropyl) ethanone (6.00 g, 61.13 mmol, 1.0 equiv.) was dissolved in methanol (40 mL) and cooled to approx. 4 ° C with an ice bath. Bromine (3.15 mL, 61.13 mmol,
1,0 equiv.) zu, so dass die Reaktionstemperatur 10 °C nicht überschritt. Das Reaktionsgemisch wurde bei ca. 4 °C für 45 min nachgerührt und anschließend mit Wasser (10 mL) versetzt. Anschließend rührte man 1 h bei Raumtemperatur nach, wobei sich die Reaktionslösung entfärbte. Das Reaktionsgemisch wurde mit Wasser verdünnt und mit Diethylether versetzt und extrahiert. Die vereinigten organischen Phasen wurden danach über Magnesiumsulfat getrocknet, filtriert und das Lösungsmittel über eine Kurzwegkolonne abdestilliert. Der Destillationsumpf enthielt das gewünschte Produkt 2-Brom-l-(l- methylcyclopropyl)ethanon in Form einer leicht gelben Flüssigkeit (11,00 g, 90 %ig, 91 % der Theorie). Das Rohprodukt wurde ohne weitere Aufreinigung in der nächsten Stufe eingesetzt. *H-NMR (400 MHz, CDCL d, ppm) 4.01 (s, 2H), 1.45 (s, 3H), 1.34 (m, 2H), 0.84 (m, 2H). Synthesestufe 2: 4-(l-Methylcyclopropyl)-l,3-thiazol-2-amin 2-Brom-l-(l-methylcyclopropyl)ethanon (10,81 g, 61,10 mmol, 1,05 equiv.) und Thiohamstoff (4,43 g, 58,19 mmol, 1,00 equiv.) wurden in Ethanol (50 mL) gelöst und 6 h zum Sieden erhitzt. Nach dem Abkühlen auf Raumtemperatur entfernte man das Lösungsmittel im Vakuum. Der Rückstand wurde mit 2 M Natronlauge und Dichlormethan extrahiert. Die Phasen trennte man über einen Phasenseparator. Durch abschließende säulenchromatographische Reinigung (Gradient Essigester/Heptan) des resultierenden Rohproduktes konnte 4-(l-Methylcyclopropyl)-l,3-thiazol-2-amin in Form eines gelben Öls isoliert werden (9,90 g, 99% der Theorie). 'H-NMR (400 MHz, CDCh d, ppm) 6.09 (s, 1H), 4.77 (bs, 2H), 1.38 (s, 3H), 1.04 (m, 2H), 0.66 (m, 2H). 1.0 equiv.) So that the reaction temperature does not exceed 10 ° C. The reaction mixture was stirred at about 4 ° C. for 45 min and then water (10 mL) was added. The mixture was then stirred at room temperature for 1 h, during which the reaction solution decolorized. The reaction mixture was diluted with water and diethyl ether was added and extracted. The combined organic phases were then dried over magnesium sulfate, filtered and the solvent was distilled off via a short path column. The distillation still contained the desired product 2-bromo-1- (1-methylcyclopropyl) ethanone in the form of a slightly yellow liquid (11.00 g, 90%, 91% of theory). The crude product was used in the next step without further purification. * H NMR (400 MHz, CDCL d, ppm) 4.01 (s, 2H), 1.45 (s, 3H), 1.34 (m, 2H), 0.84 (m, 2H). Synthesis stage 2: 4- (l-methylcyclopropyl) -l, 3-thiazol-2-amine 2-Bromo-l- (l-methylcyclopropyl) ethanone (10.81 g, 61.10 mmol, 1.05 equiv.) And thiourea (4.43 g, 58.19 mmol, 1.00 equiv.) Were added in Dissolved ethanol (50 mL) and heated to boiling for 6 h. After cooling to room temperature, the solvent was removed in vacuo. The residue was extracted with 2 M sodium hydroxide solution and dichloromethane. The phases were separated using a phase separator. By final column chromatography purification (gradient ethyl acetate / heptane) of the resulting crude product, 4- (l-methylcyclopropyl) -l, 3-thiazol-2-amine could be isolated in the form of a yellow oil (9.90 g, 99% of theory). 'H NMR (400 MHz, CDCh d, ppm) 6.09 (s, 1H), 4.77 (bs, 2H), 1.38 (s, 3H), 1.04 (m, 2H), 0.66 (m, 2H).
Synthesestufe 3 : 3-Methyl-l -[4-(l -methylcyclopropyl)- 1 ,3-thiazol-2-yl]-lH-pyrrol-2,5-dion Synthesis step 3: 3-methyl-l - [4- (l -methylcyclopropyl) - 1, 3-thiazol-2-yl] -lH-pyrrole-2,5-dione
4-(l-Methylcyclopropyl)-l,3-thiazol-2-amin (500 mg, 3,24 mmol, 1,00 equiv.) und 3-Methylfuran-2,5- dion (436 g, 3,89 mmol, 1,20 equiv.) wurden in Chloroform (10 mL) gelöst und über Nacht bei Raumtemperatur gerührt. Anschließend entfernte man das Lösungsmittel im Vakuum und versetzte den Rückstand mit Essigsäureanhydrid (lOmL) und Natriumacetat (11 mg, 0,13 mmol, 0,08 equiv.). Das Reaktionsgemisch wurde 4 h zum Sieden erhitzt. Nach dem Abkühlen auf Raumtemperatur entfernte man das Lösungsmittel im Vakuum. Den Rückstand extrahierte man mit Wasser und Dichlormethan und trennte die Phasen über einen Phasenseparator. Durch abschließende säulenchromatographische Reinigung (Gradient Essigester/Heptan) des resultierenden Rohproduktes konnte 3 -Methyl- l-[4-(l- methylcyclopropyl)-l,3-thiazol-2-yl]-lH-pyrrol-2,5-dion in Form eines gelben Öls isoliert werden (413 mg, 99% der Theorie). ‘H-NMR (400 MHz, CDCI3 d, ppm) 6.84 (s, 1H), 6.52 (s, 1H), 2.18 (s, 3H), 1.47 (s, 3H), 1.19 (m, 2H), 0.77 (m, 2H). Synthesestufe 4: 5-Hydroxy-3-methyl-l-[4-(l-methylcyclopropyl)-l,3-thiazol-2-yl]-l,5-dihydro-2H- pyrrol-2-on (1.2-20) 4- (l-methylcyclopropyl) -1, 3-thiazol-2-amine (500 mg, 3.24 mmol, 1.00 equiv.) And 3-methylfuran-2,5-dione (436 g, 3.89 mmol , 1.20 equiv.) Were dissolved in chloroform (10 mL) and stirred overnight at room temperature. The solvent was then removed in vacuo and the residue was treated with acetic anhydride (10 ml) and sodium acetate (11 mg, 0.13 mmol, 0.08 equiv.). The reaction mixture was heated to boiling for 4 h. After cooling to room temperature, the solvent was removed in vacuo. The residue was extracted with water and dichloromethane and the phases were separated using a phase separator. By final column chromatography purification (gradient ethyl acetate / heptane) of the resulting crude product, 3-methyl-l- [4- (1-methylcyclopropyl) -l, 3-thiazol-2-yl] -lH-pyrrole-2,5-dione in Form a yellow oil can be isolated (413 mg, 99% of theory). 'H NMR (400 MHz, CDCI 3 d, ppm) 6.84 (s, 1H), 6.52 (s, 1H), 2.18 (s, 3H), 1.47 (s, 3H), 1.19 (m, 2H), 0.77 (m, 2H). Synthesis stage 4: 5-hydroxy-3-methyl-l- [4- (l-methylcyclopropyl) -l, 3-thiazol-2-yl] -l, 5-dihydro-2H-pyrrol-2-one (1.2-20 )
3-Methyl-l-[4-(l-methylcyclopropyl)-l,3-thiazol-2-yl]-lH-pyrrol-2,5-dion (476 mg, 1,92 mmol, 1,00 equiv.) und Cer(lll)chlorid Heptahydrat (1,43 g, 3,84 mmol, 2,00 equiv.) wurden in Methanol (40 mL) gelöst und mit einem Eisbad auf 0 °C gekühlt. Anschließend versetzte man die Lösung portionsweise mit Natriumborhydrid (145 mg, 3,84 mmol, 2,00 equiv.) und rührte 1 h bei 0 °C nach. Das 3-methyl-l- [4- (l-methylcyclopropyl) -1, 3-thiazol-2-yl] -1 H-pyrrole-2,5-dione (476 mg, 1.92 mmol, 1.00 equiv.) and cerium (III) chloride heptahydrate (1.43 g, 3.84 mmol, 2.00 equiv.) were dissolved in methanol (40 mL) and cooled to 0 ° C with an ice bath. Then the solution was mixed in portions with sodium borohydride (145 mg, 3.84 mmol, 2.00 equiv.) And the mixture was stirred at 0 ° C. for 1 h. The
Reaktionsgemisch wurde dann mit Eisessig (1,65 mL, 28,76 mmol, 15,00 equiv.) versetzt und 30 Min. nachgerührt. Das Reaktionsgemisch extrahierte man mit Wasser und Dichlormethan und trennte die Phasen über einen Phasenseparator. Durch abschließende säulenchromatographische Reinigung (Gradient Essigester/Heptan) des resultierenden Rohproduktes konnte 5-Hydroxy-3-methyl-l-[4-(l- methylcyclopropyl)-l,3-thiazol-2-yl]-l,5-dihydro-2H-pyrrol-2-on in Form eines farblosen Feststoffes isoliert werden (240 mg, 47% der Theorie). 'H-NMR (600 MHz, CDCL d, ppm) 6.78 (m, 1H), 6.10 (s, 1H), 4.99 (s, 1H), 1.99 (s, 3H), 1.45 (s, 3H), 1.12 (m, 1H), 1.04 (m, 1H), 0.77 (m, 2H). Glacial acetic acid (1.65 mL, 28.76 mmol, 15.00 equiv.) Was then added to the reaction mixture, and the mixture was stirred for 30 minutes. The reaction mixture was extracted with water and dichloromethane and the phases were separated using a phase separator. By final column chromatography purification (gradient ethyl acetate / heptane) of the resulting crude product, 5-hydroxy-3-methyl-l- [4- (l-methylcyclopropyl) -l, 3-thiazol-2-yl] -l, 5-dihydro- 2H-pyrrol-2-one can be isolated in the form of a colorless solid (240 mg, 47% of theory). 'H NMR (600 MHz, CDCL d, ppm) 6.78 (m, 1H), 6.10 (s, 1H), 4.99 (s, 1H), 1.99 (s, 3H), 1.45 (s, 3H), 1.12 ( m, 1H), 1.04 (m, 1H), 0.77 (m, 2H).
In Analogie zu den oben angeführten und an entsprechender Stelle rezitierten Herstellungsbeispielen und unter Berücksichtigung der allgemeinen Angaben zur Herstellung von substituierten In analogy to the production examples given above and recited at the appropriate place and taking into account the general information on the production of substituted ones
Thiazolylpyrrolone erhält man die nachfolgend genannten Verbindungen. Wenn in Tabelle 1 ein Strukturelement durch eine Strukurformel definiert ist, welches eine gestrichelte Linie enthält, so bedeutet diese gestrichelte Linie, dass an dieser Position die betreffende Gruppe mit dem Rest des Moleküls verbunden ist. Thiazolylpyrrolone gives the compounds mentioned below. If in Table 1 a structural element is defined by a structural formula which contains a dashed line, this dashed line means that the group in question is connected to the rest of the molecule at this position.
Tabelle 1.1 : Bevorzugte Verbindungen der Formel (1.1) sind die Verbindungen 1.1-1 bis 1.1-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.1-1 bis 1.1- 68 der Tabelle 1.1 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.1: Preferred compounds of the formula (1.1) are the compounds 1.1-1 to 1.1-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.1-1 to 1.1- 68 of Table 1.1 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1 : Table 1 :
Tabelle 1.2: Bevorzugte Verbindungen der Formel (1.2) sind die Verbindungen 1.2-1 bis 1.2-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.2-1 bis 1.2- 68 der Tabelle 1.2 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.2: Preferred compounds of the formula (1.2) are the compounds 1.2-1 to 1.2-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.2-1 to 1.2- 68 of table 1.2 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.3: Bevorzugte Verbindungen der Formel (1.3) sind die Verbindungen 1.3-1 bis 1.3-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.3-1 bis 1.3- 68 der Tabelle 1.3 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.3: Preferred compounds of the formula (1.3) are the compounds 1.3-1 to 1.3-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.3-1 to 1.3- 68 of table 1.3 are thus by the meaning of the respective entries no. 1 to 68 defined for Q of Table 1.
Tabelle 1.4: Bevorzugte Verbindungen der Formel (1.4) sind die Verbindungen 1.4-1 bis 1.4-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.4-1 bis 1.4- 68 der Tabelle 1.4 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.4: Preferred compounds of the formula (1.4) are the compounds 1.4-1 to 1.4-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.4-1 to 1.4- 68 of Table 1.4 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.5: Bevorzugte Verbindungen der Formel (1.5) sind die Verbindungen 1.5-1 bis 1.5-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.5-1 bis 1.5- 68 der Tabelle 1.5 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.5: Preferred compounds of the formula (1.5) are the compounds 1.5-1 to 1.5-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.5-1 to 1.5- 68 of Table 1.5 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.6: Bevorzugte Verbindungen der Formel (1.6) sind die Verbindungen 1.6-1 bis 1.6-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.6-1 bis 1.6- 68 der Tabelle 1.6 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.6: Preferred compounds of the formula (1.6) are the compounds 1.6-1 to 1.6-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.6-1 to 1.6- 68 of table 1.6 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.7: Bevorzugte Verbindungen der Formel (1.7) sind die Verbindungen 1.7-1 bis 1.7-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.7-1 bis 1.7- 68 der Tabelle 1.7 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.7: Preferred compounds of the formula (1.7) are the compounds 1.7-1 to 1.7-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.7-1 to 1.7- 68 of table 1.7 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.8: Bevorzugte Verbindungen der Formel (1.8) sind die Verbindungen 1.8-1 bis 1.8-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.8-1 bis 1.8- 68 der Tabelle 1.8 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.8: Preferred compounds of the formula (1.8) are the compounds 1.8-1 to 1.8-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.8-1 to 1.8- 68 of Table 1.8 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.9: Bevorzugte Verbindungen der Formel (1.9) sind die Verbindungen 1.9-1 bis 1.9-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.9-1 bis 1.9- 68 der Tabelle 1.9 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.9: Preferred compounds of the formula (1.9) are the compounds 1.9-1 to 1.9-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.9-1 to 1.9- 68 of Table 1.9 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.10: Bevorzugte Verbindungen der Formel (1.10) sind die Verbindungen 1.10-1 bis 1.10-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.10-1 bis 1.10-68 der Tabelle 1.10 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.10: Preferred compounds of the formula (1.10) are the compounds 1.10-1 to 1.10-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.10-1 to 1.10-68 of table 1.10 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.11 : Bevorzugte Verbindungen der Formel (1.11) sind die Verbindungen 1.11-1 bis 1.11-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.11-1 bis 1.11-68 der Tabelle 1.11 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.11: Preferred compounds of the formula (1.11) are the compounds 1.11-1 to 1.11-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.11-1 to 1.11-68 of table 1.11 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.12: Bevorzugte Verbindungen der Formel (1.12) sind die Verbindungen 1.12-1 bis 1.12-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.12-1 bis 1.12-68 der Tabelle 1.12 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.12: Preferred compounds of the formula (1.12) are the compounds 1.12-1 to 1.12-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.12-1 to 1.12-68 of table 1.12 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.13: Bevorzugte Verbindungen der Formel (1.13) sind die Verbindungen 1.13-1 bis 1.13-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.13-1 bis 1.13-68 der Tabelle 1.13 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.13: Preferred compounds of the formula (1.13) are the compounds 1.13-1 to 1.13-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.13-1 to 1.13-68 of table 1.13 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.14: Bevorzugte Verbindungen der Formel (1.14) sind die Verbindungen 1.14-1 bis 1.14-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.14-1 bis 1.14-68 der Tabelle 1.14 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.14: Preferred compounds of the formula (1.14) are the compounds 1.14-1 to 1.14-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.14-1 to 1.14-68 of table 1.14 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.15: Bevorzugte Verbindungen der Formel (1.15) sind die Verbindungen 1.15-1 bis 1.15-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.15-1 bis 1.15-68 der Tabelle 1.15 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.15: Preferred compounds of the formula (1.15) are the compounds 1.15-1 to 1.15-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.15-1 to 1.15-68 of table 1.15 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.16: Bevorzugte Verbindungen der Formel (1.16) sind die Verbindungen 1.16-1 bis 1.16-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.16-1 bis 1.16-68 der Tabelle 1.16 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.16: Preferred compounds of the formula (1.16) are the compounds 1.16-1 to 1.16-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.16-1 to 1.16-68 of table 1.16 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.17: Bevorzugte Verbindungen der Formel (1.17) sind die Verbindungen 1.17-1 bis 1.17-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.17-1 bis 1.17-68 der Tabelle 1.17 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.17: Preferred compounds of the formula (1.17) are the compounds 1.17-1 to 1.17-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.17-1 to 1.17-68 of table 1.17 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.18: Bevorzugte Verbindungen der Formel (1.18) sind die Verbindungen 1.18-1 bis 1.18-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.18-1 bis 1.18-68 der Tabelle 1.18 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.18: Preferred compounds of the formula (1.18) are the compounds 1.18-1 to 1.18-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.18-1 to 1.18-68 of table 1.18 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.19: Bevorzugte Verbindungen der Formel (1.19) sind die Verbindungen 1.19-1 bis 1.19-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.19-1 bis 1.19-68 der Tabelle 1.19 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.19: Preferred compounds of the formula (1.19) are the compounds 1.19-1 to 1.19-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.19-1 to 1.19-68 of table 1.19 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.20: Bevorzugte Verbindungen der Formel (1.20) sind die Verbindungen 1.20-1 bis 1.20-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.20-1 bis 1.20-68 der Tabelle 1.20 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.20: Preferred compounds of the formula (1.20) are the compounds 1.20-1 to 1.20-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.20-1 to 1.20-68 of Table 1.20 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.21 : Bevorzugte Verbindungen der Formel (1.21) sind die Verbindungen 1.21-1 bis 1.21-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.21-1 bis 1.21-68 der Tabelle 1.21 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.21: Preferred compounds of the formula (1.21) are the compounds 1.21-1 to 1.21-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.21-1 to 1.21-68 of table 1.21 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.22: Bevorzugte Verbindungen der Formel (1.22) sind die Verbindungen 1.22-1 bis 1.22-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.22-1 bis 1.22-68 der Tabelle 1.22 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.22: Preferred compounds of the formula (1.22) are the compounds 1.22-1 to 1.22-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.22-1 to 1.22-68 of table 1.22 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.23: Bevorzugte Verbindungen der Formel (1.23) sind die Verbindungen 1.23-1 bis 1.23-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.23-1 bis 1.23-68 der Tabelle 1.23 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.23: Preferred compounds of the formula (1.23) are the compounds 1.23-1 to 1.23-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.23-1 to 1.23-68 of table 1.23 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.24: Bevorzugte Verbindungen der Formel (1.24) sind die Verbindungen 1.24-1 bis 1.24-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.24-1 bis 1.24-68 der Tabelle 1.24 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.24: Preferred compounds of the formula (1.24) are the compounds 1.24-1 to 1.24-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.24-1 to 1.24-68 of table 1.24 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.25: Bevorzugte Verbindungen der Formel (1.25) sind die Verbindungen 1.25-1 bis 1.25-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.25-1 bis 1.25-68 der Tabelle 1.25 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.25: Preferred compounds of the formula (1.25) are the compounds 1.25-1 to 1.25-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.25-1 to 1.25-68 of table 1.25 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.26: Bevorzugte Verbindungen der Formel (1.26) sind die Verbindungen 1.26-1 bis 1.26-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.26-1 bis 1.26-68 der Tabelle 1.26 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.26: Preferred compounds of the formula (1.26) are the compounds 1.26-1 to 1.26-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.26-1 to 1.26-68 of table 1.26 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.27: Bevorzugte Verbindungen der Formel (1.27) sind die Verbindungen 1.27-1 bis 1.27-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.27-1 bis 1.27-68 der Tabelle 1.27 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.27: Preferred compounds of the formula (1.27) are the compounds 1.27-1 to 1.27-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.27-1 to 1.27-68 of table 1.27 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.28: Bevorzugte Verbindungen der Formel (1.28) sind die Verbindungen 1.28-1 bis 1.28-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.28-1 bis 1.28-68 der Tabelle 1.28 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.28: Preferred compounds of the formula (1.28) are the compounds 1.28-1 to 1.28-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.28-1 to 1.28-68 of table 1.28 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.29: Bevorzugte Verbindungen der Formel (1.29) sind die Verbindungen 1.29-1 bis 1.29-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.29-1 bis 1.29-68 der Tabelle 1.29 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.29: Preferred compounds of the formula (1.29) are the compounds 1.29-1 to 1.29-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.29-1 to 1.29-68 of table 1.29 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
(1-30)(1-30)
Tabelle 1.30: Bevorzugte Verbindungen der Formel (1.30) sind die Verbindungen 1.30-1 bis 1.30-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.30-1 bis 1.30-68 der Tabelle 1.30 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.30: Preferred compounds of the formula (1.30) are the compounds 1.30-1 to 1.30-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.30-1 to 1.30-68 of table 1.30 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.31 : Bevorzugte Verbindungen der Formel (1.31) sind die Verbindungen 1.31-1 bis 1.31-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.31-1 bis 1.31-68 der Tabelle 1.31 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.31: Preferred compounds of the formula (1.31) are the compounds 1.31-1 to 1.31-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.31-1 to 1.31-68 of table 1.31 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.32: Bevorzugte Verbindungen der Formel (1.32) sind die Verbindungen 1.32-1 bis 1.32-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.32-1 bis 1.32-68 der Tabelle 1.32 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.32: Preferred compounds of the formula (1.32) are the compounds 1.32-1 to 1.32-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.32-1 to 1.32-68 of table 1.32 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.33: Bevorzugte Verbindungen der Formel (1.33) sind die Verbindungen 1.33-1 bis 1.33-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.33-1 bis 1.33-68 der Tabelle 1.33 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.33: Preferred compounds of the formula (1.33) are the compounds 1.33-1 to 1.33-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.33-1 to 1.33-68 of table 1.33 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.34: Bevorzugte Verbindungen der Formel (1.34) sind die Verbindungen 1.34-1 bis 1.34-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.34-1 bis 1.34-68 der Tabelle 1.34 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.34: Preferred compounds of the formula (1.34) are the compounds 1.34-1 to 1.34-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.34-1 to 1.34-68 of table 1.34 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.35: Bevorzugte Verbindungen der Formel (1.35) sind die Verbindungen 1.35-1 bis 1.35-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.35-1 bis 1.35-68 der Tabelle 1.35 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.35: Preferred compounds of the formula (1.35) are the compounds 1.35-1 to 1.35-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.35-1 to 1.35-68 of table 1.35 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.36: Bevorzugte Verbindungen der Formel (1.36) sind die Verbindungen 1.36-1 bis 1.36-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.36-1 bis 1.36-68 der Tabelle 1.36 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.36: Preferred compounds of the formula (1.36) are the compounds 1.36-1 to 1.36-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.36-1 to 1.36-68 of table 1.36 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.37: Bevorzugte Verbindungen der Formel (1.37) sind die Verbindungen 1.37-1 bis 1.37-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.37-1 bis 1.37-68 der Tabelle 1.37 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.37: Preferred compounds of the formula (1.37) are the compounds 1.37-1 to 1.37-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.37-1 to 1.37-68 of table 1.37 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.38: Bevorzugte Verbindungen der Formel (1.38) sind die Verbindungen 1.38-1 bis 1.38-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.38-1 bis 1.38-68 der Tabelle 1.38 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.38: Preferred compounds of the formula (1.38) are the compounds 1.38-1 to 1.38-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.38-1 to 1.38-68 of table 1.38 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.39: Bevorzugte Verbindungen der Formel (1.39) sind die Verbindungen 1.39-1 bis 1.39-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.39-1 bis 1.39-68 der Tabelle 1.39 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.39: Preferred compounds of the formula (1.39) are the compounds 1.39-1 to 1.39-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.39-1 to 1.39-68 of table 1.39 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.40: Bevorzugte Verbindungen der Formel (1.40) sind die Verbindungen 1.40-1 bis 1.40-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.40-1 bis 1.40-68 der Tabelle 1.40 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.40: Preferred compounds of the formula (1.40) are the compounds 1.40-1 to 1.40-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.40-1 to 1.40-68 of table 1.40 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.41 : Bevorzugte Verbindungen der Formel (1.41) sind die Verbindungen 1.41-1 bis 1.41-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.41-1 bis 1.41-68 der Tabelle 1.41 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.41: Preferred compounds of the formula (1.41) are the compounds 1.41-1 to 1.41-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.41-1 to 1.41-68 of Table 1.41 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.42: Bevorzugte Verbindungen der Formel (1.42) sind die Verbindungen 1.42-1 bis 1.42-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.42-1 bis 1.42-68 der Tabelle 1.42 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.42: Preferred compounds of the formula (1.42) are the compounds 1.42-1 to 1.42-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.42-1 to 1.42-68 of table 1.42 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.43: Bevorzugte Verbindungen der Formel (1.43) sind die Verbindungen 1.43-1 bis 1.43-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.43-1 bis 1.43-68 der Tabelle 1.43 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.43: Preferred compounds of the formula (1.43) are the compounds 1.43-1 to 1.43-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.43-1 to 1.43-68 of table 1.43 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.44: Bevorzugte Verbindungen der Formel (1.44) sind die Verbindungen 1.44-1 bis 1.44-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.44-1 bis 1.44-68 der Tabelle 1.44 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.44: Preferred compounds of the formula (1.44) are the compounds 1.44-1 to 1.44-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.44-1 to 1.44-68 in Table 1.44 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.45: Bevorzugte Verbindungen der Formel (1.45) sind die Verbindungen 1.45-1 bis 1.45-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.45-1 bis 1.45-68 der Tabelle 1.45 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.45: Preferred compounds of the formula (1.45) are the compounds 1.45-1 to 1.45-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.45-1 to 1.45-68 of table 1.45 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.46: Bevorzugte Verbindungen der Formel (1.46) sind die Verbindungen 1.46-1 bis 1.46-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.46-1 bis 1.46-68 der Tabelle 1.46 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.46: Preferred compounds of the formula (1.46) are the compounds 1.46-1 to 1.46-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.46-1 to 1.46-68 in Table 1.46 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.47: Bevorzugte Verbindungen der Formel (1.47) sind die Verbindungen 1.47-1 bis 1.47-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.47-1 bis 1.47-68 der Tabelle 1.47 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.47: Preferred compounds of the formula (1.47) are the compounds 1.47-1 to 1.47-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.47-1 to 1.47-68 in Table 1.47 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.48: Bevorzugte Verbindungen der Formel (1.48) sind die Verbindungen 1.48-1 bis 1.48-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.48-1 bis 1.48-68 der Tabelle 1.48 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.48: Preferred compounds of the formula (1.48) are the compounds 1.48-1 to 1.48-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.48-1 to 1.48-68 of table 1.48 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.49: Bevorzugte Verbindungen der Formel (1.49) sind die Verbindungen 1.49-1 bis 1.49-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.49-1 bis 1.49-68 der Tabelle 1.49 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.49: Preferred compounds of the formula (1.49) are the compounds 1.49-1 to 1.49-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.49-1 to 1.49-68 of table 1.49 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.50: Bevorzugte Verbindungen der Formel (1.50) sind die Verbindungen 1.50-1 bis 1.50-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.50-1 bis 1.50-68 der Tabelle 1.50 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.50: Preferred compounds of the formula (1.50) are the compounds 1.50-1 to 1.50-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.50-1 to 1.50-68 of table 1.50 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.51 : Bevorzugte Verbindungen der Formel (1.51) sind die Verbindungen 1.51-1 bis 1.51-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.51-1 bis 1.51-68 der Tabelle 1.51 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.51: Preferred compounds of the formula (1.51) are the compounds 1.51-1 to 1.51-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.51-1 to 1.51-68 of table 1.51 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.52: Bevorzugte Verbindungen der Formel (1.52) sind die Verbindungen 1.52-1 bis 1.52-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.52-1 bis 1.52-68 der Tabelle 1.52 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.52: Preferred compounds of the formula (1.52) are the compounds 1.52-1 to 1.52-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.52-1 to 1.52-68 of table 1.52 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.53: Bevorzugte Verbindungen der Formel (1.53) sind die Verbindungen 1.53-1 bis 1.53-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.53-1 bis 1.53-68 der Tabelle 1.53 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.53: Preferred compounds of the formula (1.53) are the compounds 1.53-1 to 1.53-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.53-1 to 1.53-68 of table 1.53 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.54: Bevorzugte Verbindungen der Formel (1.54) sind die Verbindungen 1.54-1 bis 1.54-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.54-1 bis 1.54-68 der Tabelle 1.54 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.54: Preferred compounds of the formula (1.54) are the compounds 1.54-1 to 1.54-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.54-1 to 1.54-68 of table 1.54 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.55: Bevorzugte Verbindungen der Formel (1.55) sind die Verbindungen 1.55-1 bis 1.55-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.55-1 bis 1.55-68 der Tabelle 1.55 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.55: Preferred compounds of the formula (1.55) are the compounds 1.55-1 to 1.55-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.55-1 to 1.55-68 of table 1.55 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.56: Bevorzugte Verbindungen der Formel (1.56) sind die Verbindungen 1.56-1 bis 1.56-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.56-1 bis 1.56-68 der Tabelle 1.56 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.56: Preferred compounds of the formula (1.56) are the compounds 1.56-1 to 1.56-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.56-1 to 1.56-68 of table 1.56 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.57: Bevorzugte Verbindungen der Formel (1.57) sind die Verbindungen 1.57-1 bis 1.57-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.57-1 bis 1.57-68 der Tabelle 1.57 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.57: Preferred compounds of the formula (1.57) are the compounds 1.57-1 to 1.57-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.57-1 to 1.57-68 of table 1.57 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.58: Bevorzugte Verbindungen der Formel (1.58) sind die Verbindungen 1.58-1 bis 1.58-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.58-1 bis 1.58-68 der Tabelle 1.58 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.58: Preferred compounds of the formula (1.58) are the compounds 1.58-1 to 1.58-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.58-1 to 1.58-68 of table 1.58 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.59: Bevorzugte Verbindungen der Formel (1.59) sind die Verbindungen 1.59-1 bis 1.59-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.59-1 bis 1.59-68 der Tabelle 1.59 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.59: Preferred compounds of the formula (1.59) are the compounds 1.59-1 to 1.59-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.59-1 to 1.59-68 of table 1.59 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.60: Bevorzugte Verbindungen der Formel (1.60) sind die Verbindungen 1.60-1 bis 1.60-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.60-1 bis 1.60-68 der Tabelle 1.60 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.60: Preferred compounds of the formula (1.60) are the compounds 1.60-1 to 1.60-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.60-1 to 1.60-68 of table 1.60 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.61 : Bevorzugte Verbindungen der Formel (1.61) sind die Verbindungen 1.61-1 bis 1.61-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.61-1 bis 1.61-68 der Tabelle 1.61 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.61: Preferred compounds of the formula (1.61) are the compounds 1.61-1 to 1.61-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.61-1 to 1.61-68 of table 1.61 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.62: Bevorzugte Verbindungen der Formel (1.62) sind die Verbindungen 1.62-1 bis 1.62-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.62-1 bis 1.62-68 der Tabelle 1.62 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.62: Preferred compounds of the formula (1.62) are the compounds 1.62-1 to 1.62-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.62-1 to 1.62-68 of table 1.62 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.63: Bevorzugte Verbindungen der Formel (1.63) sind die Verbindungen 1.63-1 bis 1.63-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.63-1 bis 1.63-68 der Tabelle 1.63 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.63: Preferred compounds of the formula (1.63) are the compounds 1.63-1 to 1.63-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.63-1 to 1.63-68 of table 1.63 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.64: Bevorzugte Verbindungen der Formel (1.64) sind die Verbindungen 1.64-1 bis 1.64-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.64-1 bis 1.64-68 der Tabelle 1.64 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.64: Preferred compounds of the formula (1.64) are the compounds 1.64-1 to 1.64-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.64-1 to 1.64-68 of table 1.64 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.65: Bevorzugte Verbindungen der Formel (1.65) sind die Verbindungen 1.65-1 bis 1.65-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.65-1 bis 1.65-68 der Tabelle 1.65 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.65: Preferred compounds of the formula (1.65) are the compounds 1.65-1 to 1.65-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.65-1 to 1.65-68 of table 1.65 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.66: Bevorzugte Verbindungen der Formel (1.66) sind die Verbindungen 1.66-1 bis 1.66-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.66-1 bis 1.66-68 der Tabelle 1.66 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.66: Preferred compounds of the formula (1.66) are the compounds 1.66-1 to 1.66-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.66-1 to 1.66-68 of table 1.66 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.67: Bevorzugte Verbindungen der Formel (1.67) sind die Verbindungen 1.67-1 bis 1.67-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.67-1 bis 1.67-68 der Tabelle 1.67 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.67: Preferred compounds of the formula (1.67) are the compounds 1.67-1 to 1.67-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.67-1 to 1.67-68 in Table 1.67 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.68: Bevorzugte Verbindungen der Formel (1.68) sind die Verbindungen 1.68-1 bis 1.68-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.68-1 bis 1.68-68 der Tabelle 1.68 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.68: Preferred compounds of the formula (1.68) are the compounds 1.68-1 to 1.68-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.68-1 to 1.68-68 of table 1.68 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.69: Bevorzugte Verbindungen der Formel (1.69) sind die Verbindungen 1.69-1 bis 1.69-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.69-1 bis 1.69-68 der Tabelle 1.69 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.69: Preferred compounds of the formula (1.69) are the compounds 1.69-1 to 1.69-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.69-1 to 1.69-68 of table 1.69 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.70: Bevorzugte Verbindungen der Formel (1.70) sind die Verbindungen 1.70-1 bis 1.70-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.70-1 bis 1.70-68 der Tabelle 1.70 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.70: Preferred compounds of the formula (1.70) are the compounds 1.70-1 to 1.70-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.70-1 to 1.70-68 of table 1.70 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.71 : Bevorzugte Verbindungen der Formel (1.71) sind die Verbindungen 1.71-1 bis 1.71-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.71-1 bis 1.71-68 der Tabelle 1.71 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.71: Preferred compounds of the formula (1.71) are the compounds 1.71-1 to 1.71-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.71-1 to 1.71-68 of table 1.71 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.72: Bevorzugte Verbindungen der Formel (1.72) sind die Verbindungen 1.72-1 bis 1.72-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.72-1 bis 1.72-68 der Tabelle 1.72 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.72: Preferred compounds of the formula (1.72) are the compounds 1.72-1 to 1.72-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.72-1 to 1.72-68 of table 1.72 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.73: Bevorzugte Verbindungen der Formel (1.73) sind die Verbindungen 1.73-1 bis 1.73-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.73-1 bis 1.73-68 der Tabelle 1.73 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.73: Preferred compounds of the formula (1.73) are the compounds 1.73-1 to 1.73-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.73-1 to 1.73-68 of table 1.73 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.74: Bevorzugte Verbindungen der Formel (1.74) sind die Verbindungen 1.74-1 bis 1.74-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.74-1 bis 1.74-68 der Tabelle 1.74 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.74: Preferred compounds of the formula (1.74) are the compounds 1.74-1 to 1.74-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.74-1 to 1.74-68 of table 1.74 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.75: Bevorzugte Verbindungen der Formel (1.75) sind die Verbindungen 1.75-1 bis 1.75-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.75-1 bis 1.75-68 der Tabelle 1.75 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.75: Preferred compounds of the formula (1.75) are the compounds 1.75-1 to 1.75-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.75-1 to 1.75-68 of table 1.75 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.76: Bevorzugte Verbindungen der Formel (1.76) sind die Verbindungen 1.76-1 bis 1.76-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.76-1 bis 1.76-68 der Tabelle 1.76 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.76: Preferred compounds of the formula (1.76) are the compounds 1.76-1 to 1.76-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.76-1 to 1.76-68 of table 1.76 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.77: Bevorzugte Verbindungen der Formel (1.77) sind die Verbindungen 1.77-1 bis 1.77-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.77-1 bis 1.77-68 der Tabelle 1.77 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.77: Preferred compounds of the formula (1.77) are the compounds 1.77-1 to 1.77-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.77-1 to 1.77-68 of table 1.77 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.78: Bevorzugte Verbindungen der Formel (1.78) sind die Verbindungen 1.78-1 bis 1.78-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.78-1 bis 1.78-68 der Tabelle 1.78 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.78: Preferred compounds of the formula (1.78) are the compounds 1.78-1 to 1.78-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.78-1 to 1.78-68 of table 1.78 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.79: Bevorzugte Verbindungen der Formel (1.79) sind die Verbindungen 1.79-1 bis 1.79-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.79-1 bis 1.79-68 der Tabelle 1.79 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.79: Preferred compounds of the formula (1.79) are the compounds 1.79-1 to 1.79-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.79-1 to 1.79-68 of table 1.79 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.80: Bevorzugte Verbindungen der Formel (1.80) sind die Verbindungen 1.80-1 bis 1.80-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.80-1 bis 1.80-68 der Tabelle 1.80 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.80: Preferred compounds of the formula (1.80) are the compounds 1.80-1 to 1.80-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.80-1 to 1.80-68 of table 1.80 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.81 : Bevorzugte Verbindungen der Formel (1.81) sind die Verbindungen 1.81-1 bis 1.81-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.81-1 bis 1.81-68 der Tabelle 1.81 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.81: Preferred compounds of the formula (1.81) are the compounds 1.81-1 to 1.81-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.81-1 to 1.81-68 of table 1.81 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.82: Bevorzugte Verbindungen der Formel (1.82) sind die Verbindungen 1.82-1 bis 1.82-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.82-1 bis 1.82-68 der Tabelle 1.82 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.82: Preferred compounds of the formula (1.82) are the compounds 1.82-1 to 1.82-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.82-1 to 1.82-68 of table 1.82 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.83: Bevorzugte Verbindungen der Formel (1.83) sind die Verbindungen 1.83-1 bis 1.83-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.83-1 bis 1.83-68 der Tabelle 1.83 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.83: Preferred compounds of the formula (1.83) are the compounds 1.83-1 to 1.83-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.83-1 to 1.83-68 of table 1.83 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.84: Bevorzugte Verbindungen der Formel (1.84) sind die Verbindungen 1.84-1 bis 1.84-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.84-1 bis 1.84-68 der Tabelle 1.84 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.84: Preferred compounds of the formula (1.84) are the compounds 1.84-1 to 1.84-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.84-1 to 1.84-68 of table 1.84 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.85: Bevorzugte Verbindungen der Formel (1.85) sind die Verbindungen 1.85-1 bis 1.85-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.85-1 bis 1.85-68 der Tabelle 1.85 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.85: Preferred compounds of the formula (1.85) are the compounds 1.85-1 to 1.85-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.85-1 to 1.85-68 of table 1.85 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.86: Bevorzugte Verbindungen der Formel (1.86) sind die Verbindungen 1.86-1 bis 1.86-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.86-1 bis 1.86-68 der Tabelle 1.86 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.86: Preferred compounds of the formula (1.86) are the compounds 1.86-1 to 1.86-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.86-1 to 1.86-68 of table 1.86 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.87: Bevorzugte Verbindungen der Formel (1.87) sind die Verbindungen 1.87-1 bis 1.87-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.87-1 bis 1.87-68 der Tabelle 1.87 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.87: Preferred compounds of the formula (1.87) are the compounds 1.87-1 to 1.87-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.87-1 to 1.87-68 of table 1.87 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.88: Bevorzugte Verbindungen der Formel (1.88) sind die Verbindungen 1.88-1 bis 1.88-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.88-1 bis 1.88-68 der Tabelle 1.88 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.88: Preferred compounds of the formula (1.88) are the compounds 1.88-1 to 1.88-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.88-1 to 1.88-68 of table 1.88 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.89: Bevorzugte Verbindungen der Formel (1.89) sind die Verbindungen 1.89-1 bis 1.89-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.89-1 bis 1.89-68 der Tabelle 1.89 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.89: Preferred compounds of the formula (1.89) are the compounds 1.89-1 to 1.89-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.89-1 to 1.89-68 of table 1.89 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.90: Bevorzugte Verbindungen der Formel (1.90) sind die Verbindungen 1.90-1 bis 1.90-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.90-1 bis 1.90-68 der Tabelle 1.90 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.90: Preferred compounds of the formula (1.90) are the compounds 1.90-1 to 1.90-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.90-1 to 1.90-68 of table 1.90 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.91 : Bevorzugte Verbindungen der Formel (1.91) sind die Verbindungen 1.91-1 bis 1.91-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.91-1 bis 1.91-68 der Tabelle 1.91 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.91: Preferred compounds of the formula (1.91) are the compounds 1.91-1 to 1.91-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.91-1 to 1.91-68 of table 1.91 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.92: Bevorzugte Verbindungen der Formel (1.92) sind die Verbindungen 1.92-1 bis 1.92-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.92-1 bis 1.92-68 der Tabelle 1.92 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.92: Preferred compounds of the formula (1.92) are the compounds 1.92-1 to 1.92-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.92-1 to 1.92-68 in Table 1.92 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.93: Bevorzugte Verbindungen der Formel (1.93) sind die Verbindungen 1.93-1 bis 1.93-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.93-1 bis 1.93-68 der Tabelle 1.93 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.93: Preferred compounds of the formula (1.93) are the compounds 1.93-1 to 1.93-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.93-1 to 1.93-68 in Table 1.93 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.94: Bevorzugte Verbindungen der Formel (1.94) sind die Verbindungen 1.94-1 bis 1.94-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.94-1 bis 1.94-68 der Tabelle 1.94 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.94: Preferred compounds of the formula (1.94) are the compounds 1.94-1 to 1.94-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.94-1 to 1.94-68 of table 1.94 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.95: Bevorzugte Verbindungen der Formel (1.95) sind die Verbindungen 1.95-1 bis 1.95-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen Table 1.95: Preferred compounds of the formula (1.95) are the compounds 1.95-1 to 1.95-68, in which Q has the meanings of Table 1 given in the respective row. The connections
1.95-1 bis 1.95-68 der Tabelle 1.95 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. 1.95-1 to 1.95-68 of Table 1.95 are therefore by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.96: Bevorzugte Verbindungen der Formel (1.96) sind die Verbindungen 1.96-1 bis 1.96-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die VerbindungenTable 1.96: Preferred compounds of the formula (1.96) are the compounds 1.96-1 to 1.96-68, in which Q has the meanings of Table 1 given in the respective row. The connections
1.96-1 bis 1.96-68 der Tabelle 1.96 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. 1.96-1 to 1.96-68 of Table 1.96 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.97: Bevorzugte Verbindungen der Formel (1.97) sind die Verbindungen 1.97-1 bis 1.97-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die VerbindungenTable 1.97: Preferred compounds of the formula (1.97) are the compounds 1.97-1 to 1.97-68, in which Q has the meanings of Table 1 given in the respective row. The connections
1.97-1 bis 1.97-68 der Tabelle 1.97 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. 1.97-1 to 1.97-68 of Table 1.97 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.98: Bevorzugte Verbindungen der Formel (1.98) sind die Verbindungen 1.98-1 bis 1.98-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.98-1 bis 1.98-68 der Tabelle 1.98 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.98: Preferred compounds of the formula (1.98) are the compounds 1.98-1 to 1.98-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.98-1 to 1.98-68 of Table 1.98 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.99: Bevorzugte Verbindungen der Formel (1.99) sind die Verbindungen 1.99-1 bis 1.99-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.99-1 bis 1.99-68 der Tabelle 1.99 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.99: Preferred compounds of the formula (1.99) are the compounds 1.99-1 to 1.99-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.99-1 to 1.99-68 of table 1.99 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
(1.100)(1.100)
Tabelle 1.100: Bevorzugte Verbindungen der Formel (I. 100) sind die Verbindungen I. 100-1 bis I. 100- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 100-1 bis I. 100-68 der Tabelle I. 100 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.100: Preferred compounds of the formula (I.100) are the compounds I.100-1 to I.100-68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 100-1 to I. 100-68 of Table I. 100 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.101 : Bevorzugte Verbindungen der Formel (1.101) sind die Verbindungen 1. 101-1 bis 1. 101- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 101-1 bis I. 101-68 der Tabelle I. 101 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. (1.102) Table 1.101: Preferred compounds of the formula (1.101) are the compounds 1. 101-1 to 1. 101- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 101-1 to I. 101-68 of Table I. 101 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1. (1,102)
Tabelle 1.102: Bevorzugte Verbindungen der Formel (I. 102) sind die Verbindungen I. 102-1 bis I. 102- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 102-1 bis I. 102-68 der Tabelle I. 102 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.102: Preferred compounds of the formula (I. 102) are the compounds I. 102-1 to I. 102- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 102-1 to I. 102-68 of table I. 102 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.103: Bevorzugte Verbindungen der Formel (1.103) sind die Verbindungen I. 103-1 bis I. 103- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 103-1 bis I. 103-68 der Tabelle I. 103 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.103: Preferred compounds of the formula (1.103) are the compounds I. 103-1 to I. 103- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 103-1 to I. 103-68 of Table I. 103 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
(1.104)(1,104)
Tabelle 1.104: Bevorzugte Verbindungen der Formel (1. 104) sind die Verbindungen 1. 104-1 bis 1. 104- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1. 104-1 bis 1. 104-68 der Tabelle 1. 104 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.104: Preferred compounds of the formula (1. 104) are the compounds 1. 104-1 to 1. 104- 68, in which Q has the meanings of Table 1 given in the respective row. The connections 1. 104-1 to 1. 104-68 of Table 1. 104 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.105: Bevorzugte Verbindungen der Formel (1. 105) sind die Verbindungen 1. 105-1 bis 1. 105- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1. 105-1 bis 1. 105-68 der Tabelle 1. 105 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.105: Preferred compounds of the formula (1. 105) are the compounds 1. 105-1 to 1. 105- 68, in which Q has the meanings of Table 1 given in the respective row. The connections 1. 105-1 to 1. 105-68 of Table 1. 105 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
(1.106)(1.106)
Tabelle 1.106: Bevorzugte Verbindungen der Formel (I. 106) sind die Verbindungen I. 106-1 bis I. 106- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 106-1 bis I. 106-68 der Tabelle I. 106 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.106: Preferred compounds of the formula (I. 106) are the compounds I. 106-1 to I. 106- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 106-1 to I. 106-68 of Table I. 106 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.107: Bevorzugte Verbindungen der Formel (I. 107) sind die Verbindungen I. 107-1 bis I. 107- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 107-1 bis I. 107-68 der Tabelle I. 107 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. (1.108)Table 1.107: Preferred compounds of the formula (I. 107) are the compounds I. 107-1 to I. 107- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 107-1 to I. 107-68 of Table I. 107 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1.108)
Tabelle 1.108: Bevorzugte Verbindungen der Formel (I. 108) sind die Verbindungen I. 108-1 bis I. 108- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 108-1 bis I. 108-68 der Tabelle I. 108 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.108: Preferred compounds of the formula (I.108) are the compounds I.108-1 to I.108-68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 108-1 to I. 108-68 of Table I. 108 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.109: Bevorzugte Verbindungen der Formel (1. 109) sind die Verbindungen 1. 109-1 bis 1. 109- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1. 109-1 bis 1. 109-68 der Tabelle 1. 109 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.109: Preferred compounds of the formula (1. 109) are the compounds 1. 109-1 to 1. 109- 68, in which Q has the meanings of Table 1 given in the respective row. The connections 1. 109-1 to 1. 109-68 of table 1. 109 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
(1.110)(1.110)
Tabelle 1.110: Bevorzugte Verbindungen der Formel (I. 110) sind die Verbindungen I. 110-1 bis I. 110- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 110-1 bis I. 110-68 der Tabelle I. 110 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.110: Preferred compounds of the formula (I.110) are the compounds I.110-1 to I.110-68, in which Q has the meanings of Table 1 given in the respective row. The connections I.110-1 to I.110-68 of Table I.110 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.111 : Bevorzugte Verbindungen der Formel (1. 111) sind die Verbindungen 1. 111-1 bis 1. 111- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1. 111-1 bis 1. 111-68 der Tabelle 1. 111 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.111: Preferred compounds of the formula (1.111) are the compounds 1.111-1 to 1.111-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1. 111-1 to 1. 111-68 of Table 1. 111 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.112: Bevorzugte Verbindungen der Formel (I. 112) sind die Verbindungen I. 112-1 bis I. 112- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 112-1 bis I. 112-68 der Tabelle I. 112 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.112: Preferred compounds of the formula (I.112) are the compounds I.112-1 to I.112-68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 112-1 to I. 112-68 of Table I. 112 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.113: Bevorzugte Verbindungen der Formel (I. 113) sind die Verbindungen I. 113-1 bis I. 113- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 113-1 bis I. 113-68 der Tabelle I. 113 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. (1.114)Table 1.113: Preferred compounds of the formula (I. 113) are the compounds I. 113-1 to I. 113- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 113-1 to I. 113-68 of Table I. 113 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1,114)
Tabelle 1.114: Bevorzugte Verbindungen der Formel (1. 114) sind die Verbindungen 1. 114-1 bis 1. 114- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1. 114-1 bis 1. 114-68 der Tabelle 1. 114 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.114: Preferred compounds of the formula (1. 114) are the compounds 1. 114-1 to 1. 114- 68, in which Q has the meanings of Table 1 given in the respective row. The connections 1. 114-1 to 1. 114-68 of Table 1. 114 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.115: Bevorzugte Verbindungen der Formel (I. 115) sind die Verbindungen I. 115-1 bis I. 115- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 115-1 bis I. 115-68 der Tabelle I. 115 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.115: Preferred compounds of the formula (I. 115) are the compounds I. 115-1 to I. 115- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 115-1 to I. 115-68 of Table I. 115 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
(1.116)(1.116)
Tabelle 1.116: Bevorzugte Verbindungen der Formel (I. 116) sind die Verbindungen I. 116-1 bis I. 116- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 116-1 bis I. 116-68 der Tabelle I. 116 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.116: Preferred compounds of the formula (I. 116) are the compounds I. 116-1 to I. 116- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 116-1 to I. 116-68 of Table I. 116 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.117: Bevorzugte Verbindungen der Formel (I. 117) sind die Verbindungen I. 117-1 bis I. 117- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 117-1 bis I. 117-68 der Tabelle I. 117 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.117: Preferred compounds of the formula (I. 117) are the compounds I. 117-1 to I. 117- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 117-1 to I. 117-68 of Table I. 117 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.118: Bevorzugte Verbindungen der Formel (I. 118) sind die Verbindungen I. 118-1 bis I. 118- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 118-1 bis I. 118-68 der Tabelle I. 118 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.118: Preferred compounds of the formula (I. 118) are the compounds I. 118-1 to I. 118- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 118-1 to I. 118-68 of Table I. 118 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.119: Bevorzugte Verbindungen der Formel (1. 119) sind die Verbindungen 1. 119-1 bis 1. 119- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1. 119-1 bis 1. 119-68 der Tabelle 1. 119 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. (1.120)Table 1,119: Preferred compounds of the formula (1,119) are the compounds 1,119-1 to 1,119-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1. 119-1 to 1. 119-68 of Table 1. 119 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1,120)
Tabelle 1.120: Bevorzugte Verbindungen der Formel (I. 120) sind die Verbindungen I. 120-1 bis I. 120- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 120-1 bis I. 120-68 der Tabelle I. 120 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.120: Preferred compounds of the formula (I.120) are the compounds I.120-1 to I.120-68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 120-1 to I. 120-68 of Table I. 120 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.121 : Bevorzugte Verbindungen der Formel (I. 121) sind die Verbindungen I. 121-1 bis I. 121- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 121-1 bis I. 121-68 der Tabelle I. 121 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1,121: Preferred compounds of the formula (I. 121) are the compounds I. 121-1 to I. 121- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 121-1 to I. 121-68 of Table I. 121 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
(1.122)(1,122)
Tabelle 1.122: Bevorzugte Verbindungen der Formel (I. 122) sind die Verbindungen I. 122-1 bis I. 122- 68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen I. 122-1 bis I. 122-68 der Tabelle I. 122 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. (1.123)Table 1.122: Preferred compounds of the formula (I. 122) are the compounds I. 122-1 to I. 122- 68, in which Q has the meanings of Table 1 given in the respective row. The connections I. 122-1 to I. 122-68 of Table I. 122 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1.123)
Tabelle 1.123: Bevorzugte Verbindungen der Formel (1.123) sind die Verbindungen 1.123-1 bis 1.123-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.123-1 bis 1.123-68 der Tabelle 1.123 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bisTable 1.123: Preferred compounds of the formula (1.123) are the compounds 1.123-1 to 1.123-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.123-1 to 1.123-68 of table 1.123 are thus by the meaning of the respective entries No. 1 to
68 für Q der Tabelle 1 definiert. 68 defined for Q of Table 1.
(1.124)(1,124)
Tabelle 1.124: Bevorzugte Verbindungen der Formel (1.124) sind die Verbindungen 1.124-1 bis 1.124-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.124-1 bis 1.124-68 der Tabelle 1.124 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.124: Preferred compounds of the formula (1.124) are the compounds 1.124-1 to 1.124-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.124-1 to 1.124-68 of table 1.124 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Tabelle 1.125: Bevorzugte Verbindungen der Formel (1.125) sind die Verbindungen 1.125-1 bis 1.125-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.125-1 bis 1.125-68 der Tabelle 1.125 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1.125: Preferred compounds of the formula (1.125) are the compounds 1.125-1 to 1.125-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.125-1 to 1.125-68 of table 1.125 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
Tabelle 1.126: Bevorzugte Verbindungen der Formel (1.126) sind die Verbindungen 1.126-1 bis 1.126-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.126-1 bis 1.126-68 der Tabelle 1.126 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bisTable 1,126: Preferred compounds of the formula (1,126) are the compounds 1,126-1 to 1,126-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.126-1 to 1.126-68 of table 1.126 are thus by the meaning of the respective entries No. 1 to
68 für Q der oben stehenden Tabelle 1 definiert. 68 for Q of Table 1 above.
Tabelle 1.127: Bevorzugte Verbindungen der Formel (1.127) sind die Verbindungen 1.127-1 bis 1.127-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.127-1 bis 1.127-68 der Tabelle 1.127 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der Tabelle 1 definiert. Table 1,127: Preferred compounds of the formula (1,127) are the compounds 1,127-1 to 1,127-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.127-1 to 1.127-68 of table 1.127 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1.
(1.128)(1,128)
Tabelle 1.128: Bevorzugte Verbindungen der Formel (1.128) sind die Verbindungen 1.128-1 bis 1.128-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.128-1 bis 1.128-68 der Tabelle 1.128 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. (1.129)Table 1.128: Preferred compounds of the formula (1.128) are the compounds 1.128-1 to 1.128-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.128-1 to 1.128-68 of table 1.128 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above. (1,129)
Tabelle 1.129: Bevorzugte Verbindungen der Formel (1.129) sind die Verbindungen 1.129-1 bis 1.129-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.129-1 bis 1.129-68 der Tabelle 1.129 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bisTable 1,129: Preferred compounds of the formula (1,129) are the compounds 1,129-1 to 1,129-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.129-1 to 1.129-68 of table 1.129 are thus by the meaning of the respective entries No. 1 to
68 für Q der Tabelle 1 definiert. 68 defined for Q of Table 1.
Tabelle 1.130: Bevorzugte Verbindungen der Formel (1.130) sind die Verbindungen 1.130-1 bis 1.130-68, worin Q die in der jeweiligen Zeile angegebenen Bedeutungen der Tabelle 1 hat. Die Verbindungen 1.130-1 bis 1.130-68 der Tabelle 1.130 sind somit durch die Bedeutung der jeweiligen Einträge No. 1 bis 68 für Q der oben stehenden Tabelle 1 definiert. Table 1.130: Preferred compounds of the formula (1.130) are the compounds 1.130-1 to 1.130-68, in which Q has the meanings of Table 1 given in the respective row. The connections 1.130-1 to 1.130-68 of table 1.130 are thus by the meaning of the respective entries No. 1 to 68 defined for Q of Table 1 above.
Spektroskopische Daten ausgewählter Tabellenbeispiele: Spectroscopic data from selected table examples:
Die nachfolgend aufgeführten spektroskopischen Daten ausgewählter Tabellenbeispiele wurden über klassische H-NMR-Intcrprctation ausgewertet. The spectroscopic data of selected table examples listed below were evaluated using classic H-NMR interpretation.
Klassische H-NMR-Intcrprctation Beispiel No. 1.2-3: Classic H-NMR interpretation Example No. 1.2-3:
‘H-NMR (600 MHz, CDC13 d, ppm) 6.81 (t, 1H), 6.53 (s, 1H), 6.16 (s, 1H), 5.23 (d, 1H), 2.98 (quin, 1H), 2.01 (s, 3H), 1.27 (dd, 6H). Beispiel No. 1.1-3: 'H NMR (600 MHz, CDC1 3 d, ppm) 6.81 (t, 1H), 6.53 (s, 1H), 6.16 (s, 1H), 5.23 (d, 1H), 2.98 (quin, 1H), 2.01 (s, 3H), 1.27 (dd, 6H). Example No. 1.1-3:
‘H-NMR (600 MHz, CDC13 d, ppm) 6.51 (t, 1H), 6.03 (br s, 1H), 5,99 (br s, 1H), 5.28 (br s, 1H), 2.98 (m, 1H), 2.19 (s, 3H), 1.26 (dd, 6H). 'H NMR (600 MHz, CDC1 3 d, ppm) 6.51 (t, 1H), 6.03 (br s, 1H), 5.99 (br s, 1H), 5.28 (br s, 1H), 2.98 (m , 1H), 2.19 (s, 3H), 1.26 (dd, 6H).
Beispiel No. 1.2-7: Example No. 1.2-7:
‘H-NMR (600 MHz, CDCI3 d, ppm) 6.81 (t, 1H), 6.55 (s, 1H), 6.17 (br s, 1H), 5.24(d, 1H), 2.01 (t, 3H), 1.31 (s, 9H). 'H NMR (600 MHz, CDCI 3 d, ppm) 6.81 (t, 1H), 6.55 (s, 1H), 6.17 (br s, 1H), 5.24 (d, 1H), 2.01 (t, 3H), 1.31 (s, 9H).
Beispiel No. 1.1-7: Example No. 1.1-7:
‘H-NMR (600 MHz, CDCI3 d, ppm) 6.52 (s, 1H), 6.04 (s, 1H), 5.98 (br s, 1H), 5.32 (br s, 1H), 2.19 (s, 3H), 1.31 (s, 9H). 'H NMR (600 MHz, CDCI 3 d, ppm) 6.52 (s, 1H), 6.04 (s, 1H), 5.98 (br s, 1H), 5.32 (br s, 1H), 2.19 (s, 3H) , 1.31 (s, 9H).
Beispiel No. 1.2-10: Example No. 1.2-10:
‘H-NMR (600 MHz, CDCI3 d, ppm) 6.79 (t, 1H), 6.51 (s, 1H), 6.12 (br s, 1H), 5.01 (br s, 1H), 2.00 (s, 3H), 1.97 (m, 1H), 0.92 (m, 2H), 0.84 (m, 1H), 0.80 (m, 1H). 'H NMR (600 MHz, CDCI 3 d, ppm) 6.79 (t, 1H), 6.51 (s, 1H), 6.12 (br s, 1H), 5.01 (br s, 1H), 2.00 (s, 3H) , 1.97 (m, 1H), 0.92 (m, 2H), 0.84 (m, 1H), 0.80 (m, 1H).
Beispiel No. 1.1-10: Example No. 1.1-10:
‘H-NMR (600 MHz, CDCI3 d, ppm) 6.49 (s, 1H), 5.98 (s, 2H), 5.07 (br s, 1H), 2.17 (s, 3H), 1.97 (m, 1H), 0.92 (m, 2H), 0.84 (m, 1H), 0.80 (m, 1H). 'H NMR (600 MHz, CDCI 3 d, ppm) 6.49 (s, 1H), 5.98 (s, 2H), 5.07 (br s, 1H), 2.17 (s, 3H), 1.97 (m, 1H), 0.92 (m, 2H), 0.84 (m, 1H), 0.80 (m, 1H).
Beispiel No. 1.1-20: Example No. 1.1-20:
‘H-NMR (600 MHz, CDCI3 d, ppm) 6.51 (s, 1H), 5.98 (m, 2H), 5.06 (br s, 1H), 2.17 (s, 3H), 1.45 (s, 3H), 1.07 (m, 1H), 0.78 (m, 1H). 'H NMR (600 MHz, CDCI 3 d, ppm) 6.51 (s, 1H), 5.98 (m, 2H), 5.06 (br s, 1H), 2.17 (s, 3H), 1.45 (s, 3H), 1.07 (m, 1H), 0.78 (m, 1H).
Beispiel No. 1.100-7: Example No. 1100-7:
‘H-NMR (600 MHz, CDCI3 d, ppm) 7.09 (s, 1H), 6.61 (s, 1H), 6.28 (br s, 1H), 5.31 (br s, 1H), 1.32 (s, 9H). 'H NMR (600 MHz, CDCI 3 d, ppm) 7.09 (s, 1H), 6.61 (s, 1H), 6.28 (br s, 1H), 5.31 (br s, 1H), 1.32 (s, 9H) ,
Beispiel No. 1.4-20: Example No. 1.4-20:
‘H-NMR (400 MHz, CDCI3 d, ppm) 7.24 (m, 1H), 6.72 (s, 1H), 6.54 (s, 1H), 2.11 (s, 3H), 2.01 (s, 3H), 1.43 (s, 3H), 1.16 (s, 1H), 0.97 (m, 1H), 0.71 (m, 2H). 'H NMR (400 MHz, CDCI3 d, ppm) 7.24 (m, 1H), 6.72 (s, 1H), 6.54 (s, 1H), 2.11 (s, 3H), 2.01 (s, 3H), 1.43 ( s, 3H), 1.16 (s, 1H), 0.97 (m, 1H), 0.71 (m, 2H).
Beispiel No. 1.2-26: Example No. 1.2-26:
‘H-NMR (400 MHz, CDCI3 d, ppm) 6.97 (s, 1H), 6.80 (s, 1H), 6.12 (s, 1H), 4.74 (s, 1H), 2.01 (s, 3H), 0.54-0.44 (m, 4H). Beispiel No. 1.99-12: 'H NMR (400 MHz, CDCI3 d, ppm) 6.97 (s, 1H), 6.80 (s, 1H), 6.12 (s, 1H), 4.74 (s, 1H), 2.01 (s, 3H), 0.54- 0.44 (m, 4H). Example No. 1.99-12:
‘H-NMR (600 MHz, CDC13 d, ppm) 7.44 (s, 1H), 6.36 (s, 1H), 6.22 (s, 1H), 5.12 (br s, 1H). 'H NMR (600 MHz, CDC1 3 d, ppm) 7.44 (s, 1H), 6.36 (s, 1H), 6.22 (s, 1H), 5.12 (br s, 1H).
Beispiel No. 1.100-12: Example No. 1100-12:
‘H-NMR (600 MHz, CDCI3 d, ppm) 7.42 (s, 1H), 7.07 (s, 1H), 6.29 (s, 1H), 4.80 (br s, 1H). ’H-NMR (600 MHz, CDCI3 d, ppm) 7.42 (s, 1H), 7.07 (s, 1H), 6.29 (s, 1H), 4.80 (br s, 1H).
Beispiel No. 1.1-12: Example No. 1.1-12:
‘H-NMR (600 MHz, CDCI3 d, ppm) 7.42 (d, 1H), 6.86 (d, 1H), 6.24 (t, 1H), 4.77 (d, 1H), 2.03 (s, 3 H). Beispiel No. 1.2-48: ’H NMR (600 MHz, CDCI3 d, ppm) 7.42 (d, 1H), 6.86 (d, 1H), 6.24 (t, 1H), 4.77 (d, 1H), 2.03 (s, 3 H). Example No. 1.2-48:
‘H-NMR (600 MHz, CDCI3 d, ppm) 7.86-7.83 (m, 2H), 7.45-7.41 (m, 2H), 7.37-7.32 (m, 1H), 7.17 (s, 1H), 6.86 (s, 1H), 6.29 (s, 1H), 5.14 (s, 1H), 2.04 (s, 3H). 'H NMR (600 MHz, CDCI3 d, ppm) 7.86-7.83 (m, 2H), 7.45-7.41 (m, 2H), 7.37-7.32 (m, 1H), 7.17 (s, 1H), 6.86 (s , 1H), 6.29 (s, 1H), 5.14 (s, 1H), 2.04 (s, 3H).
Beispiel No. 1.2-9: Example No. 1.2-9:
‘H-NMR (400 MHz, CDCI3 d, ppm) 7.09 (s, 1H), 6.78 (m, 1H), 6.13 (s, 1H), 5.04 (s, 1H), 2.00 (s, 3H), 1.96 (m, 1H), 1.00 (m, 2H), 0.72 (m, 2H). 'H NMR (400 MHz, CDCI3 d, ppm) 7.09 (s, 1H), 6.78 (m, 1H), 6.13 (s, 1H), 5.04 (s, 1H), 2.00 (s, 3H), 1.96 ( m, 1H), 1.00 (m, 2H), 0.72 (m, 2H).
Beispiel No. 1.1-9: Example No. 1.1-9:
‘H-NMR (600 MHz, CDCI3 d, ppm) 7.07 (s, 1H), 5.99 (s, 1H), 5.98 (s, 1H), 5.17 (br,s, 1H), 2.17 (s,'H NMR (600 MHz, CDCI 3 d, ppm) 7.07 (s, 1H), 5.99 (s, 1H), 5.98 (s, 1H), 5.17 (br, s, 1H), 2.17 (s,
3H), 1.97 (m, 1H), 0.99 (m, 2H), 0.73 (m, 2H). 3H), 1.97 (m, 1H), 0.99 (m, 2H), 0.73 (m, 2H).
Beispiel No. 1.2-5: Example No. 1.2-5:
‘H-NMR (400 MHz, CDCI3 d, ppm) 7.10 (s, 1H), 6.79 (s, 1H), 6.15 (s, 1H), 5.06 (br s, 1H), 2.81 (q,'H NMR (400 MHz, CDCI 3 d, ppm) 7.10 (s, 1H), 6.79 (s, 1H), 6.15 (s, 1H), 5.06 (br s, 1H), 2.81 (q,
2H), 2.01 (s, 3H), 1.31 (t, 3H). 2H), 2.01 (s, 3H), 1.31 (t, 3H).
Beispiel No. 1.1-5: Example No. 1.1-5:
‘H-NMR (600 MHz, CDCI3 d, ppm) 7.07 (s, 1H), 6.00 (d,2H), 5.14 (br s, 1H), 2.80 (q, 2H), 2.18 (s, 3H), 1.31 (t, 3H). 'H NMR (600 MHz, CDCI3 d, ppm) 7.07 (s, 1H), 6.00 (d, 2H), 5.14 (br s, 1H), 2.80 (q, 2H), 2.18 (s, 3H), 1.31 (t, 3H).
Beispiel No. 1.2-4: Example No. 1.2-4:
‘H-NMR (400 MHz, CDCI3 d, ppm) 7.07 (s, 1H), 5.99 (m, 2H), 5.11 (br, s, 1H), 3.14 (quintet, 1H), 2.18 (s, 3H), 1.35 (s, 3H), 1.34 (s, 3H). 'H NMR (400 MHz, CDCI 3 d, ppm) 7.07 (s, 1H), 5.99 (m, 2H), 5.11 (br, s, 1H), 3.14 (quintet, 1H), 2.18 (s, 3H) , 1.35 (s, 3H), 1.34 (s, 3H).
Beispiel No. 1.2-41 : Example No. 1.2-41:
‘H-NMR (400 MHz, CDCI3 d, ppm) 6.53 (s, 1H), 6.05 (d, 1H), 6.00(d, 1H), 5.31 (br s, 1H), 3.57 (m, ’H-NMR (400 MHz, CDCI3 d, ppm) 6.53 (s, 1H), 6.05 (d, 1H), 6.00 (d, 1H), 5.31 (br s, 1H), 3.57 (m,
1H), 2.35-2.28 (m, 2H), 2.27-2.17 (m, 2H), 2.19 (s , 3H), 2.03 (m, 1H), 1.90 (m, 1H). Beispiel No. 1.1-41 : 1H), 2.35-2.28 (m, 2H), 2.27-2.17 (m, 2H), 2.19 (s, 3H), 2.03 (m, 1H), 1.90 (m, 1H). Example No. 1.1-41:
‘H-NMR (400 MHz, CDC13 d, ppm) 6.81 (s, 1H), 6.55 (s, 1H), 6.00(d, 1H), 5.23 (br s, 1H), 3.56 (m, 1H), 2.36-2.29 (m, 2H), 2.25-2.17 (m, 2H), 2.05 (s , 3H), 2.03 (m, 1H), 1.90 (m, 1H). 'H NMR (400 MHz, CDC1 3 d, ppm) 6.81 (s, 1H), 6.55 (s, 1H), 6.00 (d, 1H), 5.23 (br s, 1H), 3.56 (m, 1H), 2.36-2.29 (m, 2H), 2.25-2.17 (m, 2H), 2.05 (s, 3H), 2.03 (m, 1H), 1.90 (m, 1H).
Beispiel No. 1.99-12: Example No. 1.99-12:
‘H-NMR (400 MHz, CDCI3 d, ppm) 7.49 (s, 1H), 7.14 (s, 1H), 6.35 (m, 1H), 4.76 (d, 1H). ’H NMR (400 MHz, CDCI3 d, ppm) 7.49 (s, 1H), 7.14 (s, 1H), 6.35 (m, 1H), 4.76 (d, 1H).
Beispiel No. 1.2-12: Example No. 1.2-12:
‘H-NMR (400 MHz, CDCI3 d, ppm) 7.42 (s, 1H), 6.87 (s, 1H), 6.24 (s, 1H), 4.71 (d, 1H), 2.03 (s, 3H). H NMR (400 MHz, CDCI3 d, ppm) 7.42 (s, 1H), 6.87 (s, 1H), 6.24 (s, 1H), 4.71 (d, 1H), 2.03 (s, 3H).
Gegenstand der vorliegenden Erfindung ist weiterhin die Verwendung einer oder mehrerer The present invention furthermore relates to the use of one or more
Verbindungen der allgemeinen Formel (I) und/oder deren Salzen, wie oben definiert, vorzugsweise in einer der als bevorzugt bzw. besonders bevorzugt gekennzeichneten Ausgestaltung, insbesondere einer oder mehrerer Verbindungen der Formeln (1.1) bis (1.130) und/oder deren Salze, jeweils wie oben definiert, Compounds of the general formula (I) and / or their salts, as defined above, preferably in one of the configurations characterized as preferred or particularly preferred, in particular one or more compounds of the formulas (1.1) to (1,130) and / or their salts, each as defined above,
als Herbizid und/oder Pflanzenwachstumsregulator, vorzugsweise in Kulturen von Nutz- und/oder Zierpflanzen. as a herbicide and / or plant growth regulator, preferably in crops of useful and / or ornamental plants.
Gegenstand der vorliegenden Erfindung ist ferner ein Verfahren zur Bekämpfung von Schadpflanzen und/oder zur Wachstumsregulierung von Pflanzen, dadurch gekennzeichnet, dass eine wirksame Menge einer oder mehrerer Verbindungen der allgemeinen Formel (I) und/oder deren Salzen, wie oben definiert, vorzugsweise in einer der als bevorzugt bzw. besonders bevorzugt gekennzeichneten The present invention furthermore relates to a process for controlling harmful plants and / or for regulating the growth of plants, characterized in that an effective amount of one or more compounds of the general formula (I) and / or their salts, as defined above, preferably in one those marked as preferred or particularly preferred
Ausgestaltung, insbesondere einer oder mehrerer Verbindungen der Formeln (1.1) bis (1.130) und/oder deren Salze, jeweils wie oben definiert, oder eines erfindungsgemäßen Mittels, wie nachstehend definiert, auf die (Schad)Pflanzcn, (Schad)Pflanzensamen, den Boden, in dem oder auf dem die (Schad) Pflanzen wachsen, oder die Anbaufläche appliziert wird. Design, in particular one or more compounds of the formulas (1.1) to (1.130) and / or their salts, each as defined above, or an agent according to the invention, as defined below, onto the (harmful) plants, (harmful) plant seeds, the soil , in or on which the (harmful) plants grow, or the cultivated area is applied.
Gegenstand der vorliegenden Erfindung ist auch ein Verfahren zur Bekämpfung von unerwünschten Pflanzen, vorzugsweise in Nutzpflanzenkulturen, dadurch gekennzeichnet, dass eine wirksame Menge einer oder mehrerer Verbindungen der allgemeinen Formel (1) und/oder deren Salzen, wie oben definiert, vorzugsweise in einer der als bevorzugt bzw. besonders bevorzugt gekennzeichneten Ausgestaltung, insbesondere einer oder mehrerer Verbindungen der Formeln (1.1) bis (1.130) und/oder deren Salze, jeweils wie oben definiert, oder eines erfindungsgemäßen Mittels, wie nachstehend definiert, auf unerwünschte Pflanzen (z.B. Schadpflanzen wie mono- oder dikotyle Unkräuter oder unerwünschte Kulturpflanzen), das Saatgut der unerwünschten Pflanzen (d.h. Pflanzensamen, z.B. Körner, Samen oder vegetative Vermehrungsorgane wie Knollen oder Sprossteile mit Knospen), den Boden, in dem oder auf dem die unerwünschte Pflanzen wachsen, (z.B. den Boden von Kulturland oder Nicht-Kulturland) oder die Anbaufläche (d.h. Fläche, auf der die unerwünschte Pflanzen wachsen werden) appliziert wird. The present invention also relates to a process for controlling unwanted plants, preferably in crops, characterized in that an effective amount of one or more compounds of the general formula (1) and / or their salts, as defined above, preferably in one of the as preferably or particularly preferably marked Design, in particular one or more compounds of the formulas (1.1) to (1.130) and / or their salts, in each case as defined above, or an agent according to the invention, as defined below, on undesired plants (for example harmful plants such as mono- or dicotyledon weeds or undesirable plants Crops), the seeds of the unwanted plants (ie plant seeds, e.g. grains, seeds or vegetative propagation organs such as tubers or shoots with buds), the soil in or on which the unwanted plants grow (e.g. the soil of cultivated or non-cultivated land ) or the cultivated area (ie area on which the unwanted plants will grow) is applied.
Gegenstand der vorliegenden Erfindung ist ferner auch Verfahren zur Bekämpfung zur The present invention also relates to methods for combating
Wachstumsregulierung von Pflanzen, vorzugsweise von Nutzpflanzen, dadurch gekennzeichnet, dass eine wirksame Menge einer oder mehrerer Verbindungen der allgemeinen Formel (I) und/oder deren Salzen, wie oben definiert, vorzugsweise in einer der als bevorzugt bzw. besonders bevorzugt gekennzeichneten Growth regulation of plants, preferably of useful plants, characterized in that an effective amount of one or more compounds of the general formula (I) and / or their salts, as defined above, preferably in one of the preferred or particularly preferred
Ausgestaltung, insbesondere einer oder mehrerer Verbindungen der Formeln (1.1) bis (1.130) und/oder deren Salze, jeweils wie oben definiert, oder eines erfindungsgemäßen Mittels, wie nachstehend definiert, die Pflanze, das Saatgut der Pflanze (d.h. Pflanzensamen, z.B. Körner, Samen oder vegetative Design, in particular one or more compounds of the formulas (1.1) to (1.130) and / or their salts, each as defined above, or an agent according to the invention, as defined below, the plant, the seed of the plant (ie plant seeds, for example grains, Seeds or vegetative
Vermehrungsorgane wie Knollen oder Sprossteile mit Knospen), den Boden, in dem oder auf dem die Pflanzen wachsen, (z.B. den Boden von Kulturland oder Nicht-Kulturland) oder die Anbaufläche (d.h. Fläche, auf der die Pflanzen wachsen werden) appliziert wird. Propagation organs such as tubers or shoots with buds), the soil in or on which the plants grow (e.g. the soil of cultivated or non-cultivated land) or the area under cultivation (i.e. the area on which the plants will grow) is applied.
Dabei können die erfindungsgemäßen Verbindungen der allgemeinen Formel (I) bzw. die The compounds of general formula (I) or
erfindungsgemäßen Mittel z.B. im Vorsaat- (ggf. auch durch Einarbeitung in den Boden), Vorauflauf und/oder Nachauflaufverfahren ausgebracht werden lm einzelnen seien beispielhaft einige Vertreter der mono- und dikotylen Unkrautflora genannt, die durch die die erfindungsgemäßen Verbindungen kontrolliert werden können, ohne dass durch die Nennung eine Beschränkung auf bestimmte Arten erfolgen soll. agents according to the invention e.g. in the pre-sowing (possibly also by incorporation into the soil), pre-emergence and / or post-emergence methods. Some representatives of the monocotyledonous and dicotyledonous weed flora can be mentioned by way of example, which can be controlled by the compounds according to the invention without being mentioned a restriction to certain types should be made.
Vorzugsweise werden in einem erfindungsgemäßen Verfahren zur Bekämpfung von Schadpflanzen oder zur Wachstumsregulierung von Pflanzen eine oder mehrere Verbindungen der allgemeinen Formel (1) und/oder deren Salze zur Bekämpfung von Schadpflanzen oder zur Wachstumsregulierung in Kulturen von Nutzpflanzen oder Zierpflanzen eingesetzt, wobei die Nutzpflanzen oder Zierpflanzen in einer bevorzugten Ausgestaltung transgene Pflanzen sind. In a process according to the invention for controlling harmful plants or for regulating the growth of plants, one or more compounds of the general formula (1) and / or their salts are preferably used for controlling harmful plants or for regulating growth in crops of useful plants or ornamental plants, the useful plants or ornamental plants in a preferred embodiment being transgenic plants.
Die erfindungsgemäßen Verbindungen der allgemeinen Formel (I) und/oder deren Salze eignen sich zur Bekämpfung der folgenden Gattungen von monokotylen und dikotylen Schadpflanzen: The compounds of the general formula (I) and / or their salts according to the invention are suitable for combating the following genera of monocotyledonous and dicotyledonous harmful plants:
Monokotyle Schadpflanzen der Gattungen: Aegilops, Agropyron, Agrostis, Alopecurus, Apera, Avena, Brachiaria, Bromus, Cenchrus, Commelina, Cynodon, Cyperus, Dactyloctenium, Digitaria, Echinochloa, Eleocharis, Eleusine, Eragrostis, Eriochloa, Festuca, Fimbristylis, Heteranthera, Imperata, Ischaemum, Leptochloa, Lolium, Monochoria, Panicum, Paspalum, Phalaris, Phleum, Poa, Rottboellia, Sagittaria, Scirpus, Setaria, Sorghum. Monocotyledonous harmful plants of the genera: Aegilops, Agropyron, Agrostis, Alopecurus, Apera, Avena, Brachiaria, Bromus, Cenchrus, Commelina, Cynodon, Cyperus, Dactyloctenium, Digitaria, Echinochloa, Eleocharis, Eleusine, Eragrostis, Festoochaantheristant, Eraochisimeterist, Eraochisimeterist, Eriochisimantherist, Eriochisimantheristant, Eriochisimantheristant, Eriochisimantheristant, Eriochisimeterist, Eriochisimeterim , Ischaemum, Leptochloa, Lolium, Monochoria, Panicum, Paspalum, Phalaris, Phleum, Poa, Rottboellia, Sagittaria, Scirpus, Setaria, Sorghum.
Dikotyle Schadpflanzen der Gattungen: Abutilon, Amaranthus, Ambrosia, Anoda, Anthemis, Aphanes, Artemisia, Atriplex, Bellis, Bidens, Capsella, Carduus, Cassia, Centaurea, Chenopodium, Cirsium, Convolvulus, Datura, Desmodium, Emex, Erysimum, Euphorbia, Galeopsis, Galinsoga, Galium, Hibiscus, Ipomoea, Kochia, Lamium, Lepidium, Lindemia, Matricaria, Mentha, Mercurialis, Mullugo, Myosotis, Papaver, Pharbitis, Plantago, Polygonum, Portulaca, Ranunculus, Raphanus, Rorippa, Rotala, Rumex, Salsola, Senecio, Sesbania, Sida, Sinapis, Solanum, Sonchus, Sphenoclea, Stellaria, Taraxacum, Thlaspi, Trifolium, Urtica, Veronica, Viola, Xanthium. Dicotyledonous harmful plants of the genera: Abutilon, Amaranthus, Ambrosia, Anoda, Anthemis, Aphanes, Artemisia, Atriplex, Bellis, Bidens, Capsella, Carduus, Cassia, Centaurea, Chenopodium, Cirsium, Convolvulus, Datura, Desmodium, Emex, Erysimum, Euphor , Galinsoga, Galium, Hibiscus, Ipomoea, Kochia, Lamium, Lepidium, Lindemia, Matricaria, Mentha, Mercurialis, Mullugo, Myosotis, Papaver, Pharbitis, Plantago, Polygonum, Portulaca, Ranunculus, Raphanus, Rorippa, Rotala, Rumex, Salsola, Senec , Sesbania, Sida, Sinapis, Solanum, Sonchus, Sphenoclea, Stellaria, Taraxacum, Thlaspi, Trifolium, Urtica, Veronica, Viola, Xanthium.
Werden die erfindungsgemäßen Verbindungen der allgemeinen Formel (I) vor dem Keimen der Schadpflanzen (Ungräser und/oder Unkräuter) auf die Erdoberfläche appliziert (Vorauflaufverfahren), so wird entweder das Auflaufen der Ungras- bzw. Unkrautkeimlinge vollständig verhindert oder diese wachsen bis zum Keimblattstadium heran, stellen jedoch dann ihr Wachstum ein und sterben schließlich nach Ablauf von drei bis vier Wochen vollkommen ab. If the compounds of the general formula (I) according to the invention are applied to the earth's surface before the germination of the harmful plants (grasses and / or weeds) (pre-emergence method), either the weed or weed seedlings are prevented from emerging completely or these grow up to the cotyledon stage However, they stop growing and eventually die completely after three to four weeks.
Bei Applikation der Wirkstoffe auf die grünen Pflanzenteile im Nachauflaufverfahren tritt nach der Behandlung Wachstumsstop ein und die Schadpflanzen bleiben in dem zum Applikationszeitpunkt vorhandenen Wachstumsstadium stehen oder sterben nach einer gewissen Zeit ganz ab, so dass auf diese Weise eine für die Kulturpflanzen schädliche Unkrautkonkurrenz sehr früh und nachhaltig beseitigt wird. When the active ingredients are applied to the green parts of the plants in the post-emergence process, growth stops after the treatment and the harmful plants remain at the growth stage at the time of application or die completely after a certain time, so that weed competition which is harmful to the crop plants is very early and is permanently eliminated.
Obgleich die erfindungsgemäßen Verbindungen der allgemeinen Formel (1) eine ausgezeichnete herbizide Aktivität gegenüber mono- und dikotylen Unkräutern aufweisen, werden Kulturpflanzen wirtschaftlich bedeutender Kulturen z.B. dikotyler Kulturen der Gattungen Arachis, Beta, Brassica, Cucumis, Cucurbita, Helianthus, Daucus, Glycine, Gossypium, lpomoea, Lactuca, Linum, Although the compounds of the general formula (1) according to the invention have an excellent herbicidal activity against mono- and dicotyledon weeds, crop plants of economically important crops are e.g. dicotyledon cultures of the genera Arachis, Beta, Brassica, Cucumis, Cucurbita, Helianthus, Daucus, Glycine, Gossypium, lpomoea, Lactuca, Linum,
Lycopersicon, Miscanthus, Nicotiana, Phaseolus, Pisum, Solanum, Vicia, oder monokotyler Kulturen der Gattungen Allium, Ananas, Asparagus, Avena, Hordeum, Oryza, Panicum, Saccharum, Secale, Sorghum, Triticale, Triticum, Zea, abhängig von der Struktur der jeweiligen erfindungsgemäßen Verbindung und deren Aufwandmenge nur unwesentlich oder gar nicht geschädigt. Die vorliegenden Verbindungen eignen sich aus diesen Gründen sehr gut zur selektiven Bekämpfung von unerwünschtem Pflanzenwuchs in Pflanzenkulturen wie landwirtschaftlichen Nutzpflanzungen oder Zierpflanzungen. Lycopersicon, Miscanthus, Nicotiana, Phaseolus, Pisum, Solanum, Vicia, or monocotyledon cultures of the genera Allium, Pineapple, Asparagus, Avena, Hordeum, Oryza, Panicum, Saccharum, Secale, Sorghum, Triticale, Triticum, Zea, depending on the structure of the respective according to the invention Connection and their application rate is only slightly or not at all damaged. For these reasons, the present compounds are very suitable for the selective control of undesired plant growth in plant crops, such as agricultural crops or ornamental crops.
Darüberhinaus weisen die erfindungsgemäßen Verbindungen der allgemeinen Formel (1) (abhängig von ihrer jeweiligen Struktur und der ausgebrachten Aufwandmenge) hervorragende In addition, the compounds of the general formula (1) according to the invention have excellent properties (depending on their structure and the application rate applied)
wachstumsregulatorische Eigenschaften bei Kulturpflanzen auf. Sie greifen regulierend in den pflanzeneigenen Stoffwechsel ein und können damit zur gezielten Beeinflussung von growth regulatory properties in crops. They intervene regulating the plant's own metabolism and can thus be used to influence
Pflanzeninhaltsstoffen und zur Emteerleichterung wie z.B. durch Auslösen von Desikkation und Wuchsstauchung eingesetzt werden. Desweiteren eignen sie sich auch zur generellen Steuerung und Hemmung von unerwünschtem vegetativem Wachstum, ohne dabei die Pflanzen abzutöten. Eine Hemmung des vegetativen Wachstums spielt bei vielen mono- und dikotylen Kulturen eine große Rolle, da beispielsweise die Lagerbildung hierdurch verringert oder völlig verhindert werden kann. Phytonutrients and to ease the crop, e.g. by triggering desiccation and stunted growth. Furthermore, they are also suitable for general control and inhibition of undesirable vegetative growth without killing the plants. Inhibiting vegetative growth plays a major role in many monocotyledonous and dicotyledonous crops, since, for example, this can reduce or completely prevent stock formation.
Aufgrund ihrer herbiziden und pflanzenwachstumsregulatorischen Eigenschaften können die Wirkstoffe auch zur Bekämpfung von Schadpflanzen in Kulturen von gentechnisch oder durch konventionelle Mutagenese veränderten Pflanzen eingesetzt werden. Die transgenen Pflanzen zeichnen sich in der Regel durch besondere vorteilhafte Eigenschaften aus, beispielsweise durch Resistenzen gegenüber bestimmten Pestiziden, vor allem bestimmten Herbiziden, Resistenzen gegenüber Pflanzenkrankheiten oder Erregern von Pflanzenkrankheiten wie bestimmten Insekten oder Mikroorganismen wie Pilzen, Bakterien oder Viren. Andere besondere Eigenschaften betreffen z.B. das Emtegut hinsichtlich Menge, Qualität, Lagerfähigkeit, Zusammensetzung und spezieller Inhaltsstoffe. So sind transgene Pflanzen mit erhöhtem Stärkegehalt oder veränderter Qualität der Stärke oder solche mit anderer Due to their herbicidal and plant growth regulatory properties, the active compounds can also be used to control harmful plants in crops of plants modified by genetic engineering or by conventional mutagenesis. The transgenic plants are generally distinguished by special advantageous properties, for example resistance to certain pesticides, especially certain herbicides, resistance to plant diseases or pathogens of plant diseases such as certain insects or microorganisms such as fungi, bacteria or viruses. Other special properties concern e.g. the crop in terms of quantity, quality, shelf life, composition and special ingredients. So are transgenic plants with increased starch content or changed starch quality or those with others
Fettsäurezusammensetzung des Emteguts bekannt. Fatty acid composition of the crop known.
Bevorzugt bezüglich transgener Kulturen ist die Anwendung der erfindungsgemäßen Verbindungen und/oder deren Salze in wirtschaftlich bedeutenden transgenen Kulturen von Nutz und Zierpflanzen, z.B. von Getreide wie Weizen, Gerste, Roggen, Hafer, Hirse, Reis und Mais oder auch Kulturen von Zuckerrübe, Baumwolle, Soja, Raps, Kartoffel, Tomate, Erbse und anderen Gemüsesorten. Preferred for transgenic cultures is the use of the compounds according to the invention and / or their salts in economically important transgenic cultures of useful and ornamental plants, e.g. of cereals such as wheat, barley, rye, oats, millet, rice and corn or also crops of sugar beet, cotton, soybeans, rapeseed, potatoes, tomatoes, peas and other vegetables.
Vorzugsweise können die erfindungsgemäßen Verbindungen auch als Herbizide in The compounds according to the invention can preferably also be used as herbicides
Nutzpflanzenkulturen eingesetzt werden, welche gegenüber den phytotoxischen Wirkungen der Herbizide resistent sind bzw. gentechnisch resistent gemacht worden sind. Crop crops are used which are resistant to the phytotoxic effects of the herbicides or have been made genetically resistant.
Aufgrund ihrer herbiziden und pflanzenwachstumsregulatorischen Eigenschaften können die Wirkstoffe auch zur Bekämpfung von Schadpflanzen in Kulturen von bekannten oder noch zu entwickelnden gentechnisch veränderten Pflanzen eingesetzt werden. Die transgenen Pflanzen zeichnen sich in der Regel durch besondere vorteilhafte Eigenschaften aus, beispielsweise durch Resistenzen gegenüber bestimmten Pestiziden, vor allem bestimmten Herbiziden, Resistenzen gegenüber Pflanzenkrankheiten oder Erregern von Pflanzenkrankheiten wie bestimmten Insekten oder Mikroorganismen wie Pilzen, Bakterien oder Viren. Andere besondere Eigenschaften betreffen z.B. das Emtegut hinsichtlich Menge, Qualität, Lagerfähigkeit, Zusammensetzung und spezieller Inhaltsstoffe. So sind transgene Pflanzen mit erhöhtem Stärkegehalt oder veränderter Qualität der Stärke oder solche mit anderer Because of their herbicidal and plant growth regulatory properties, the active compounds can also be used to control harmful plants in crops of known or still to be developed genetically modified plants. The transgenic plants are generally distinguished by special advantageous properties, for example by resistance to them certain pesticides, especially certain herbicides, resistance to plant diseases or pathogens such as certain insects or microorganisms such as fungi, bacteria or viruses. Other special properties relate to the crop, for example, in terms of quantity, quality, shelf life, composition and special ingredients. So are transgenic plants with increased starch content or changed starch quality or those with others
Fettsäurezusammensetzung des Emteguts bekannt. Weitere besondere Eigenschaften können in einer Toleranz oder Resistenz gegen abiotische Stressoren z.B. Hitze, Kälte, Trockenheit, Salz und ultraviolette Strahlung liegen. Fatty acid composition of the crop known. Other special properties can include tolerance or resistance to abiotic stressors e.g. There is heat, cold, dryness, salt and ultraviolet radiation.
Bevorzugt ist die Anwendung der erfindungsgemäßen Verbindungen der allgemeinen Formel (I) oder deren Salze in wirtschaftlich bedeutenden transgenen Kulturen von Nutz-und Zierpflanzen, z.B. von Getreide wie Weizen, Gerste, Roggen, Hafer, Triticale, Hirse, Reis, Maniok und Mais oder auch Kulturen von Zuckerrübe, Baumwolle, Soja, Raps, Kartoffel, Tomate, Erbse und anderen Gemüsesorten. Preference is given to using the compounds of the general formula (I) according to the invention or their salts in economically important transgenic crops of useful and ornamental plants, e.g. of cereals such as wheat, barley, rye, oats, triticale, millet, rice, manioc and corn or also crops of sugar beet, cotton, soybean, rapeseed, potato, tomato, pea and other vegetables.
Vorzugsweise können die Verbindungen der allgemeinen Formel (I) als Herbizide in The compounds of the general formula (I) can preferably be used as herbicides in
Nutzpflanzenkulturen eingesetzt werden, welche gegenüber den phytotoxischen Wirkungen der Herbizide resistent sind bzw. gentechnisch resistent gemacht worden sind. Crop crops are used which are resistant to the phytotoxic effects of the herbicides or have been made genetically resistant.
Herkömmliche Wege zur Herstellung neuer Pflanzen, die im Vergleich zu bisher vorkommenden Pflanzen modifizierte Eigenschaften aufweisen, bestehen beispielsweise in klassischen Conventional ways of producing new plants which have modified properties in comparison to previously occurring plants are, for example, classic ones
Züchtungsverfahren und der Erzeugung von Mutanten. Alternativ können neue Pflanzen mit veränderten Eigenschaften mit Hilfe gentechnischer Verfahren erzeugt werden. Breeding methods and the generation of mutants. Alternatively, new plants with modified properties can be created using genetic engineering methods.
Zahlreiche molekularbiologische Techniken, mit denen neue transgene Pflanzen mit veränderten Eigenschaften hergestellt werden können, sind dem Fachmann bekannt. Für derartige gentechnische Manipulationen können Nucleinsäuremoleküle in Plasmide eingebracht werden, die eine Mutagenese oder eine Sequenzveränderung durch Rekombination von DNA-Sequenzen erlauben. Mit Hilfe von Standardverfahren können z.B. Basenaustausche vorgenommen, Teilsequenzen entfernt oder natürliche oder synthetische Sequenzen hinzugefügt werden. Für die Verbindung der DNA-Fragmente Numerous molecular biological techniques with which new transgenic plants with modified properties can be produced are known to the person skilled in the art. For such genetic engineering manipulations, nucleic acid molecules can be introduced into plasmids which allow mutagenesis or a sequence change by recombining DNA sequences. With the help of standard procedures e.g. Base exchanges made, partial sequences removed or natural or synthetic sequences added. For connecting the DNA fragments
untereinander können an die Fragmente Adaptoren oder Linker angesetzt werden. among themselves, adapters or linkers can be attached to the fragments.
Die Herstellung von Pflanzenzellen mit einer verringerten Aktivität eines Genprodukts kann The production of plant cells with a reduced activity of a gene product can
beispielsweise erzielt werden durch die Expression mindestens einer entsprechenden antisense-RNA, einer sense-RNA zur Erzielung eines Cosuppressionseffektes oder die Expression mindestens eines entsprechend konstruierten Ribozyms, das spezifisch Transkripte des obengenannten Genprodukts spaltet. Hierzu können zum einen DNA-Moleküle verwendet werden, die die gesamte codierende Sequenz eines Genprodukts einschließlich eventuell vorhandener flankierender Sequenzen umfassen, als auch DNA- Moleküle, die nur Teile der codierenden Sequenz umfassen, wobei diese Teile lang genug sein müssen, um in den Zellen einen antisense-Effekt zu bewirken. Möglich ist auch die Verwendung von DNA- Sequenzen, die einen hohen Grad an Homologie zu den codiereden Sequenzen eines Genprodukts aufweisen, aber nicht vollkommen identisch sind. can be achieved, for example, by the expression of at least one corresponding antisense RNA, a sense RNA to achieve a cosuppression effect or the expression of at least one appropriately constructed ribozyme which specifically cleaves transcripts of the above-mentioned gene product. For this purpose, DNA molecules can be used that comprise the entire coding sequence of a gene product, including any flanking sequences that may be present, as well as DNA molecules that only comprise parts of the coding sequence, these parts having to be long enough to be in the cells to cause an antisense effect. It is also possible to use DNA sequences which have a high degree of homology to the coding sequences of a gene product, but which are not completely identical.
Bei der Expression von Nucleinsäuremolekülen in Pflanzen kann das synthetisierte Protein in jedem beliebigen Kompartiment der pflanzlichen Zelle lokalisiert sein. Um aber die Lokalisation in einem bestimmten Kompartiment zu erreichen, kann z.B. die codierende Region mit DNA-Sequenzen verknüpft werden, die die Lokalisierung in einem bestimmten Kompartiment gewährleisten. Derartige Sequenzen sind dem Fachmann bekannt (siehe beispielsweise Braun et al., EMBO J. 11 (1992), 3219- 3227). Die Expression der Nukleinsäuremoleküle kann auch in den Organellen der Pflanzenzellen stattfinden. When nucleic acid molecules are expressed in plants, the synthesized protein can be located in any compartment of the plant cell. However, in order to achieve localization in a certain compartment, e.g. the coding region is linked to DNA sequences which ensure localization in a specific compartment. Such sequences are known to the person skilled in the art (see, for example, Braun et al., EMBO J. 11 (1992), 3219-3227). The expression of the nucleic acid molecules can also take place in the organelles of the plant cells.
Die transgenen Pflanzenzellen können nach bekannten Techniken zu ganzen Pflanzen regeneriert werden. Bei den transgenen Pflanzen kann es sich prinzipiell um Pflanzen jeder beliebigen The transgenic plant cells can be regenerated into whole plants using known techniques. In principle, the transgenic plants can be any plants
Pflanzenspezies handeln, d.h. sowohl monokotyle als auch dikotyle Pflanzen. Trade plant species, i.e. both monocot and dicot plants.
So sind transgene Pflanzen erhältlich, die veränderte Eigenschaften durch Überexpression, Suppression oder Inhibierung homologer (= natürlicher) Gene oder Gensequenzen oder Expression heterologer (= fremder) Gene oder Gensequenzen aufweisen. Thus, transgenic plants are available which have changed properties due to overexpression, suppression or inhibition of homologous (= natural) genes or gene sequences or expression of heterologous (= foreign) genes or gene sequences.
Vorzugsweise können die erfindungsgemäßen Verbindungen (I) in transgenen Kulturen eingesetzt werden, welche gegen Wuchsstoffe, wie z.B. Dicamba oder gegen Herbizide, die essentielle The compounds (I) according to the invention can preferably be used in transgenic cultures which are active against growth substances, e.g. Dicamba or against herbicides, the essential
Pflanzenenzyme, z.B. Acetolactatsynthasen (ALS), EPSP Synthasen, Glutaminsynthasen (GS) oder Hydoxyphenylpyruvat Dioxygenasen (HPPD) hemmen, respektive gegen Herbizide aus der Gruppe der Sulfonylharnstoffe, der Glyphosate, Glufosinate oder Benzoylisoxazole und analogen Wirkstoffe, resistent sind. Plant enzymes, e.g. Inhibit acetolactate synthases (ALS), EPSP synthases, glutamine synthases (GS) or hydoxyphenyl pyruvate dioxygenases (HPPD), or are resistant to herbicides from the group of sulfonylureas, glyphosate, glufosinate or benzoylisoxazole and analogous active ingredients.
Bei der Anwendung der erfindungsgemäßen Wirkstoffe in transgenen Kulturen treten neben den in anderen Kulturen zu beobachtenden Wirkungen gegenüber Schadpflanzen oftmals Wirkungen auf, die für die Applikation in der jeweiligen transgenen Kultur spezifisch sind, beispielsweise ein verändertes oder speziell erweitertes Unkrautspektrum, das bekämpft werden kann, veränderte Aufwandmengen, die für die Applikation eingesetzt werden können, vorzugsweise gute Kombinierbarkeit mit den Herbiziden, gegenüber denen die transgene Kultur resistent ist, sowie Beeinflussung von Wuchs und Ertrag der transgenen Kulturpflanzen. Gegenstand der Erfindung ist deshalb auch die Verwendung der erfindungsgemäßen Verbindungen der allgemeinen Formel (I) und/oder deren Salze als Herbizide zur Bekämpfung von Schadpflanzen in Kulturen von Nutz- oder Zierpflanzen, gegebenenfalls in transgenen Kulturpflanzen. When the active compounds according to the invention are used in transgenic crops, in addition to the effects on harmful plants which can be observed in other crops, effects often occur which are specific to the application in the respective transgenic culture, for example a changed or specially expanded weed spectrum which can be controlled changed Application rates that can be used for the application, preferably good combinability with the herbicides to which the transgenic culture is resistant, and influencing the growth and yield of the transgenic crop plants. The invention therefore also relates to the use of the compounds of the general formula (I) according to the invention and / or their salts as herbicides for controlling harmful plants in crops of useful or ornamental plants, optionally in transgenic crop plants.
Bevorzugt ist die Verwendung in Getreide, dabei vorzugsweise Mais, Weizen, Gerste, Roggen, Hafer, Hirse, oder Reis, im Vor- oder Nachauflauf. Preference is given to use in cereals, preferably maize, wheat, barley, rye, oats, millet or rice, in the pre- or post-emergence.
Bevorzugt ist auch die Verwendung in Soja im Vor- oder Nachauflauf. Pre-or post-soya use is also preferred.
Die erfindungsgemäße Verwendung zur Bekämpfung von Schadpflanzen oder zur The use according to the invention for controlling harmful plants or
Wachstumsregulierung von Pflanzen schließt auch den Fall ein, bei dem der Wirkstoff der allgemeinen Formel (1) oder dessen Salz erst nach der Ausbringung auf der Pflanze, in der Pflanze oder im Boden aus einer Vorläufersubstanz ("Prodrug") gebildet wird. Plant growth regulation also includes the case in which the active ingredient of the general formula (1) or its salt is formed from a precursor substance (“prodrug”) only after application to the plant, in the plant or in the soil.
Gegenstand der Erfindung ist auch die Verwendung einer oder mehrerer Verbindungen der Formel (1) oder deren Salzen bzw. eines erfindungsgemäßen Mittels (wie nachstehend definiert) (in einem The invention also relates to the use of one or more compounds of the formula (1) or their salts or an agent according to the invention (as defined below) (in one
Verfahren) zur Bekämpfung von Schadpflanzen oder zur Wachstumsregulierung von Pflanzen, dadurch gekennzeichnet, dass man eine wirksame Menge einer oder mehreren Verbindungen der allgemeinen Formel (I) oder deren Salzen auf die Pflanzen (Schadpflanzen, ggf. zusammen mit den Nutzpflanzen) Pflanzensamen, den Boden, in dem oder auf dem die Pflanzen wachsen, oder die Anbaufläche appliziert. Process) for controlling harmful plants or for regulating the growth of plants, characterized in that an effective amount of one or more compounds of the general formula (I) or their salts are applied to the plants (harmful plants, if appropriate together with the useful plants) plant seeds, the soil , in or on which the plants grow, or the cultivated area is applied.
Gegenstand der Erfindung ist auch ein herbizides und/oder pflanzenwachstumsregulierendes Mittel, dadurch gekennzeichnet, dass das Mittel The invention also relates to a herbicidal and / or plant growth-regulating agent, characterized in that the agent
(a) eine oder mehrere Verbindungen der allgemeinen Formel (I) und/oder deren Salze enthält wie oben definiert, vorzugsweise in einer der als bevorzugt bzw. besonders bevorzugt gekennzeichneten (a) contains one or more compounds of the general formula (I) and / or their salts as defined above, preferably in one of those identified as preferred or particularly preferred
Ausgestaltung, insbesondere eine oder mehrere Verbindungen der Formeln (1.1) bis (1.130) und/oder deren Salze, jeweils wie oben definiert, Design, in particular one or more compounds of the formulas (1.1) to (1.130) and / or their salts, each as defined above,
und and
(b) ein oder mehrere weitere Stoffe ausgewählt aus den Gruppen (i) und/oder (ii): (b) one or more further substances selected from groups (i) and / or (ii):
(i) ein oder mehrere weitere agrochemisch wirksame Stoffe, vorzugsweise ausgewählt aus der(i) one or more further agrochemically active substances, preferably selected from the
Gruppe bestehend aus Insektiziden, Akariziden, Nematiziden, weiteren Herbiziden (d.h. solche, die nicht der oben definierten allgemeinen Formel (I) entsprechen), Fungiziden, Safenern, Düngemitteln und/oder weiteren Wachstumsregulatoren, (ii) ein oder mehrere im Pflanzenschutz übliche Formulierungshilfsmittel. Group consisting of insecticides, acaricides, nematicides, further herbicides (ie those which do not correspond to the general formula (I) defined above), fungicides, safeners, fertilizers and / or further growth regulators, (ii) one or more formulation auxiliaries customary in crop protection.
Die weiteren agrochemischen wirksamen Stoffe des Bestandteils (i) eines erfindungsgemäßen Mittels sind dabei vorzugsweise ausgewählt aus der Gruppe der Stoffe, die in "The Pesticide Manual", l6th edition, The British Crop Protection Council und the Royal Soc. of Chemistry, 2012 genannt sind. The further agrochemically active substances of component (i) of an agent according to the invention are preferably selected from the group of substances described in "The Pesticide Manual", 16th edition, The British Crop Protection Council and the Royal Soc. of Chemistry, 2012.
Ein erfindungsgemäßes herbizides oder pflanzenwachstumsregulierendes Mittel, umfasst vorzugsweise ein, zwei, drei oder mehr im Pflanzenschutz übliche Formulierungshilfsmittel (ii) ausgewählt aus der Gruppe bestehend aus Tensiden, Emulgatoren, Dispergiermitteln, Filmbildnem, Verdickungsmitteln, anorganischen Salzen, Stäubemitteln, bei 25 °C und 1013 mbar festen Trägerstoffen, vorzugsweise adsorptionsfähigen, granulierten Inertmaterialien, Netzmitteln, Antioxidationsmitteln, Stabilisatoren, Puffersubstanzen, Antischaummitteln, Wasser, organischen Lösungsmitteln, vorzugsweise bei 25 °C und 1013 mbar mit Wasser in jedem beliebigen Verhältnis mischbare organische Lösungsmittel. A herbicidal or plant growth-regulating agent according to the invention preferably comprises one, two, three or more formulation auxiliaries (ii) customary in crop protection selected from the group consisting of surfactants, emulsifiers, dispersants, film formers, thickeners, inorganic salts, dusts, at 25 ° C. and 1013 mbar solid carriers, preferably adsorbable, granulated inert materials, wetting agents, antioxidants, stabilizers, buffer substances, antifoams, water, organic solvents, preferably at 25 ° C. and 1013 mbar, organic solvents miscible with water in any ratio.
Die erfindungsgemäßen Verbindungen der allgemeinen Formel (I) können in Form von Spritzpulvem, emulgierbaren Konzentraten, versprühbaren Lösungen, Stäubemitteln oder Granulaten in den üblichen Zubereitungen angewendet werden. Gegenstand der Erfindung sind deshalb auch herbizide und pflanzenwachstumsregulierende Mittel, die Verbindungen der allgemeinen Formel (I) und/oder deren Salze enthalten. The compounds of the general formula (I) according to the invention can be used in the form of wettable powders, emulsifiable concentrates, sprayable solutions, dusts or granules in the customary formulations. The invention therefore also relates to herbicidal and plant growth-regulating compositions which comprise compounds of the general formula (I) and / or their salts.
Die Verbindungen der allgemeinen Formel (I) und/oder deren Salze können auf verschiedene Art formuliert werden, je nachdem welche biologischen und/oder chemisch-physikalischen Parameter vorgegeben sind. Als Formulierungsmöglichkeiten kommen beispielsweise in Frage: Spritzpulver (WP), wasserlösliche Pulver (SP), wasserlösliche Konzentrate, emulgierbare Konzentrate (EC), Emulsionen (EW), wie Öl-in-Wasser- und Wasser-in-Öl-Emulsionen, versprühbare Lösungen, The compounds of general formula (I) and / or their salts can be formulated in various ways, depending on which biological and / or chemical-physical parameters are specified. Possible formulation options are, for example: wettable powder (WP), water-soluble powder (SP), water-soluble concentrates, emulsifiable concentrates (EC), emulsions (EW), such as oil-in-water and water-in-oil emulsions, sprayable solutions .
Suspensionskonzentrate (SC), Dispersionen auf Öl- oder Wasserbasis, ölmischbare Lösungen, Suspension concentrates (SC), dispersions based on oil or water, oil-miscible solutions,
Kapselsuspensionen (CS), Stäubemittel (DP), Beizmittel, Granulate für die Streu- und Capsule suspensions (CS), dusts (DP), pickling agents, granules for the litter and
Bodenapplikation, Granulate (GR) in Form von Mikro-, Sprüh-, Aufzugs- und Adsorptionsgranulaten, wasserdispergierbare Granulate (WG), wasserlösliche Granulate (SG), ULV-Formulierungen, Soil application, granules (GR) in the form of micro, spray, elevator and adsorption granules, water-dispersible granules (WG), water-soluble granules (SG), ULV formulations,
Mikrokapseln und Wachse. Microcapsules and waxes.
Diese einzelnen Formulierungstypen und die Formulierungshilfsmittel wie Inertmaterialien, Tenside, Lösungsmittel und weitere Zusatzstoffe sind dem Fachmann bekannt, und werden beispielsweise beschrieben in: Watkins, "Handbook of Insecticide Dust Diluents and Carriers", 2nd Ed., Darland Books, Caldwell N.J., H.v. Olphen, "Introduction to Clay Colloid Chemistry"; 2nd Ed., J. Wiley & Sons, N.Y.; C. Marsden, "Solvents Guide"; 2nd Ed., Interscience, N.Y. 1963; McCutcheon's "Detergents and Emulsifiers Annual", MC Publ. Corp., Ridgewood N.J.; Sisley and Wood, "Encyclopedia of Surface Active Agents", Chem. Publ. Co. Inc., N.Y. 1964; Schönfeldt, "Grenzflächenaktive These individual formulation types and the formulation auxiliaries such as inert materials, surfactants, solvents and other additives are known to the person skilled in the art and are described, for example, in: Watkins, "Handbook of Insecticide Dust Diluents and Carriers", 2nd Ed., Darland Books, Caldwell NJ, Hv Olphen , "Introduction to Clay Colloid Chemistry"; 2nd Ed., J. Wiley & Sons, NY; C. Marsden, "Solvents Guide"; 2nd Ed., Interscience, NY 1963; McCutcheon's Detergents and Emulsifiers Annual, MC Publ. Corp., Ridgewood NJ; Sisley and Wood, "Encyclopedia of Surface Active Agents ", Chem. Publ. Co. Inc., NY 1964; Schönfeldt," interfacial
Äthylenoxidaddukte", Wiss. Verlagsgesellschaft, Stuttgart 1976; Winnacker-Küchler, "Chemische Technologie", Band 7, C. Hanser Verlag München, 4. Aufl. 1986. Äthylenoxidaddukte ", Wiss. Verlagsgesellschaft, Stuttgart 1976; Winnacker-Küchler," Chemische Technologie ", Volume 7, C. Hanser Verlag Munich, 4th ed. 1986.
Spritzpulver sind in Wasser gleichmäßig dispergierbare Präparate, die neben dem Wirkstoff außer einem Verdünnungs- oder Inertstoff noch Tenside ionischer und/oder nichtionischer Art (Netzmittel, Dispergiermittel), z.B. polyoxyethylierte Alkylphenole, polyoxethylierte Fettalkohole, polyoxethylierte Fettamine, Fettalkoholpolyglykolethersulfate, Alkansulfonate, Alkylbenzolsulfonate, ligninsulfonsaures Natrium, 2,2'-dinaphthylmethan-6,6'-disulfonsaures Natrium, dibutylnaphthalin-sulfonsaures Natrium oder auch oleoylmethyltaurinsaures Natrium enthalten. Zur Herstellung der Spritzpulver werden die herbiziden Wirkstoffe beispielsweise in üblichen Apparaturen wie Hammermühlen, Gebläsemühlen und Luftstrahlmühlen feingemahlen und gleichzeitig oder anschließend mit den Formulierungshilfsmitteln vermischt. Spray powders are preparations which are uniformly dispersible in water and which, in addition to the active substance, contain not only a diluent or an inert substance, but also ionic and / or nonionic surfactants (wetting agents, dispersing agents), e.g. polyoxyethylated alkylphenols, polyoxethylated fatty alcohols, polyoxethylated fatty amines, fatty alcohol polyglycol ether sulfates, alkanesulfonates, alkylbenzenesulfonates, ligninsulfonic acid sodium, 2,2'-dinaphthylmethane-6,6'-disulfonic acid sodium, sodium dibutylnaphthalene-sulfonic acid sodium or else sodium. To produce the wettable powders, the herbicidal active ingredients are, for example, finely ground in customary equipment, such as hammer mills, fan mills and air jet mills, and mixed at the same time or subsequently with the formulation auxiliaries.
Emulgierbare Konzentrate werden durch Auflösen des Wirkstoffes in einem organischen Lösungsmittel z.B. Butanol, Cyclohexanon, Dimethylformamid, Xylol oder auch höhersiedenden Aromaten oder Kohlenwasserstoffen oder Mischungen der organischen Lösungsmittel unter Zusatz von einem oder mehreren Tensiden ionischer und/oder nichtionischer Art (Emulgatoren) hergestellt. Als Emulgatoren können beispielsweise verwendet werden: Alkylarylsulfonsaure Calcium-Salze wie Emulsifiable concentrates are made by dissolving the active ingredient in an organic solvent e.g. Butanol, cyclohexanone, dimethylformamide, xylene or higher-boiling aromatics or hydrocarbons or mixtures of organic solvents with the addition of one or more surfactants of ionic and / or nonionic type (emulsifiers). Examples of emulsifiers that can be used are: alkylarylsulfonic acid calcium salts such as
Ca-dodecylbenzolsulfonat oder nichtionische Emulgatoren wie Fettsäurepolyglykolester, Ca-dodecylbenzenesulfonate or nonionic emulsifiers such as fatty acid polyglycol esters,
Alkylarylpolyglykolether, F ettalkoholpolyglykolether, Propylenoxid-Ethylenoxid- Kondensationsprodukte, Alkylpolyether, Sorbitanester wie z.B. Sorbitanfettsäureester oder Alkylaryl polyglycol ether, fatty alcohol polyglycol ether, propylene oxide-ethylene oxide condensation products, alkyl polyether, sorbitan esters such as e.g. Sorbitan fatty acid esters or
Polyoxethylensorbitanester wie z.B. Polyoxyethylensorbitanfettsäureester. Polyoxethylene sorbitan esters such as e.g. Polyoxyethylene.
Stäubemittel erhält man durch Vermahlen des Wirkstoffes mit fein verteilten festen Stoffen, z.B. Dusts are obtained by grinding the active ingredient with finely divided solid substances, e.g.
Talkum, natürlichen Tonen, wie Kaolin, Bentonit und Pyrophyllit, oder Diatomeenerde. Talc, natural clays such as kaolin, bentonite and pyrophyllite, or diatomaceous earth.
Suspensionskonzentrate können auf Wasser- oder Ölbasis sein. Sie können beispielsweise durch Naß- Vermahlung mittels handelsüblicher Perlmühlen und gegebenenfalls Zusatz von Tensiden, wie sie z.B. oben bei den anderen Formulierungstypen bereits aufgeführt sind, hergestellt werden. Suspension concentrates can be water or oil based. You can, for example, by wet grinding using commercially available bead mills and optionally adding surfactants such as those e.g. already listed above for the other types of formulation.
Emulsionen, z.B. Öl-in-Wasser-Emulsionen (EW), lassen sich beispielsweise mittels Rührern, Kolloidmühlen und/oder statischen Mischern unter Verwendung von wäßrigen organischen Emulsions, e.g. Oil-in-water emulsions (EW) can be used, for example, using stirrers, colloid mills and / or static mixers using aqueous organic ones
Lösungsmitteln und gegebenenfalls Tensiden, wie sie z.B. oben bei den anderen Formulierungstypen bereits aufgeführt sind, hersteilen. Granulate können entweder durch Verdüsen des Wirkstoffes auf adsorptionsfähiges, granuliertes Inertmaterial hergestellt werden oder durch Aufbringen von Wirkstoffkonzentraten mittels Klebemitteln, z.B. Polyvinylalkohol, polyacrylsaurem Natrium oder auch Mineralölen, auf die Oberfläche von Trägerstoffen wie Sand, Kaolinite oder von granuliertem Inertmaterial. Auch können geeignete Manufacture solvents and optionally surfactants, such as those already listed above for the other types of formulation. Granules can either be produced by spraying the active ingredient onto adsorbable, granulated inert material or by applying active ingredient concentrates using adhesives, for example polyvinyl alcohol, polyacrylic acid sodium or mineral oils, to the surface of carriers such as sand, kaolinite or granulated inert material. Also suitable ones
Wirkstoffe in der für die Herstellung von Düngemittelgranulaten üblichen Weise - gewünschtenfalls in Mischung mit Düngemitteln - granuliert werden. Active ingredients are granulated in the manner customary for the production of fertilizer granules - if desired in a mixture with fertilizers.
Wasserdispergierbare Granulate werden in der Regel nach den üblichen Verfahren wie Sprühtrocknung, Wirbelbett-Granulierung, Teller-Granulierung, Mischung mit Hochgeschwindigkeitsmischem und Extrusion ohne festes Inertmaterial hergestellt. Water-dispersible granules are generally produced using the customary processes, such as spray drying, fluidized bed granulation, plate granulation, mixing with high-speed mixers and extrusion without solid inert material.
Zur Herstellung von Teller-, Fließbett-, Extruder- und Sprühgranulaten siehe z.B. Verfahren in "Spray- Drying Handbook" 3rd ed. 1979, G. Goodwin Ltd., London; J.E. Browning, "Agglomeration", Chemical and Engineering 1967, Seiten 147 ff; "Perry's Chemical Engineer's Handbook", 5th Ed., McGraw-Hill, New York 1973, S. 8-57. For the production of plate, fluidized bed, extruder and spray granules, see e.g. Procedure in "Spray-Drying Handbook" 3rd ed. 1979, G. Goodwin Ltd., London; J.E. Browning, "Agglomeration", Chemical and Engineering 1967, pages 147 ff; "Perry's Chemical Engineer's Handbook," 5th Ed., McGraw-Hill, New York 1973, pp. 8-57.
Für weitere Einzelheiten zur Formulierung von Pflanzenschutzmitteln siehe z.B. G.C. Klingman, "Weed Control as a Science", John Wiley and Sons, Inc., New York, 1961, Seiten 81-96 und J.D. Freyer, S.A. Evans, "Weed Control Handbook", 5th Ed., Blackwell Scientific Publications, Oxford, 1968, Seiten 101-103. For more details on the formulation of crop protection products see e.g. G.C. Klingman, "Weed Control as a Science," John Wiley and Sons, Inc., New York, 1961, pp. 81-96, and J.D. Freyer, S.A. Evans, "Weed Control Handbook," 5th Ed., Blackwell Scientific Publications, Oxford, 1968, pages 101-103.
Die agrochemischen Zubereitungen, vorzugsweise herbizide oder pflanzenwachstumsregulierende Mittel der vorliegenden Erfindung enthalten vorzugsweise eine Gesamtmenge von 0,1 bis 99 Gew.-%, bevorzugt 0,5 bis 95 Gew.-%, weiter bevorzugt 1 bis 90 Gew.-%, insbesondere bevorzugt 2 bis 80 Gew.-%, an Wirkstoffen der allgemeinen Formel (I) und deren Salzen. The agrochemical preparations, preferably herbicidal or plant growth-regulating agents of the present invention preferably contain a total amount of 0.1 to 99% by weight, preferably 0.5 to 95% by weight, more preferably 1 to 90% by weight, particularly preferably 2 to 80% by weight of active compounds of the general formula (I) and their salts.
In Spritzpulvem beträgt die Wirkstoffkonzentration z.B. etwa 10 bis 90 Gew.-%, der Rest zu 100 Gew.-% besteht aus üblichen Formulierungsbestandteilen. Bei emulgierbaren Konzentraten kann die Wirkstoffkonzentration etwa 1 bis 90, vorzugsweise 5 bis 80 Gew.-% betragen. Staubförmige The active substance concentration in wettable powders is e.g. about 10 to 90 wt .-%, the rest of 100 wt .-% consists of conventional formulation components. In the case of emulsifiable concentrates, the active substance concentration can be about 1 to 90, preferably 5 to 80,% by weight. Powdery
Formulierungen enthalten 1 bis 30 Gew.-% Wirkstoff, vorzugsweise meistens 5 bis 20 Gew.-% an Wirkstoff, versprühbare Lösungen enthalten etwa 0,05 bis 80, vorzugsweise 2 bis 50 Gew.-% Wirkstoff. Bei wasserdispergierbaren Granulaten hängt der Wirkstoffgehalt zum Teil davon ab, ob die wirksame Verbindung flüssig oder fest vorliegt und welche Granulierhilfsmittel, Füllstoffe usw. verwendet werden. Bei den in Wasser dispergierbaren Granulaten liegt der Gehalt an Wirkstoff beispielsweise zwischen 1 und 95 Gew.-%, vorzugsweise zwischen 10 und 80 Gew.-%. Daneben enthalten die genannten Wirkstofformulierungen gegebenenfalls die jeweils üblichen Haft-, Netz-, Dispergier-, Emulgier-, Penetrations-, Konservierungs-, Frostschutz- und Lösungsmittel, Füll-, Träger- und Farbstoffe, Entschäumer, Verdunstungshemmer und den pH-Wert und die Viskosität beeinflussende Mittel. Beispiele für Formulierungshilfsmittel sind unter anderem in "Chemistry and Technology of Agrochemical Formulations", ed. D. A. Knowles, Kluwer Academic Publishers (1998) beschrieben. Formulations contain 1 to 30% by weight of active ingredient, preferably mostly 5 to 20% by weight of active ingredient, sprayable solutions contain about 0.05 to 80, preferably 2 to 50% by weight of active ingredient. In the case of water-dispersible granules, the active ingredient content depends in part on whether the active compound is in liquid or solid form and which granulating aids, fillers, etc. are used. The active ingredient content of the water-dispersible granules is, for example, between 1 and 95% by weight, preferably between 10 and 80% by weight. In addition, the active ingredient formulations mentioned may contain the customary adhesives, wetting agents, dispersants, emulsifiers, penetrants, preservatives, antifreezes and solvents, fillers, carriers and dyes, defoamers, evaporation inhibitors and the pH and Agents influencing viscosity. Examples of formulation auxiliaries are described, inter alia, in "Chemistry and Technology of Agrochemical Formulations", ed. DA Knowles, Kluwer Academic Publishers (1998).
Die Verbindungen der allgemeinen Formel (I) oder deren Salze können als solche oder in Form ihrer Zubereitungen (Formulierungen) mit anderen pestizid wirksamen Stoffen, wie z.B. Insektiziden, Akariziden, Nematiziden, Herbiziden, Fungiziden, Safenern, Düngemitteln und/oder The compounds of general formula (I) or their salts can be used as such or in the form of their preparations (formulations) with other pesticidally active substances, e.g. Insecticides, acaricides, nematicides, herbicides, fungicides, safeners, fertilizers and / or
Wachstumsregulatoren kombiniert eingesetzt werden, z.B. als Fertigformulierung oder als Growth regulators can be used in combination, e.g. as a finished formulation or as
Tankmischungen. Die Kombinationsformulierungen können dabei auf Basis der obengenannten Formulierungen hergestellt werden, wobei die physikalischen Eigenschaften und Stabilitäten der zu kombinierenden Wirkstoffe zu berücksichtigen sind. Tank mixes. The combination formulations can be prepared on the basis of the formulations mentioned above, taking into account the physical properties and stabilities of the active compounds to be combined.
In Abhängigkeit von den Pflanzenarten oder Pflanzensorten, ihrem Standort und ihren Depending on the plant species or plant varieties, their location and their
Wachstumsbedingungen (Böden, Klima, Vegetationsperiode, Ernährung) kann die erfindungsgemäße Behandlung auch zu überadditiven ("synergistischen") Effekten führen. So sind zum Beispiel die folgenden Effekte möglich, die über die eigentlich zu erwartenden Effekte hinausgehen: verringerte Aufwandmengen und/oder erweitertes Wirkungsspektrum und/oder erhöhte Wirksamkeit der Wirkstoffe und Zusammensetzungen, die erfindungsgemäß eingesetzt werden können, besseres Pflanzenwachstum, erhöhte Toleranz gegenüber hohen oder niedrigen Temperaturen, erhöhte Toleranz gegenüber Treatment conditions according to the invention can also lead to superadditive ("synergistic") effects in growth conditions (soils, climate, growing season, nutrition). For example, the following effects are possible that go beyond the effects that are actually to be expected: reduced application rates and / or expanded spectrum of activity and / or increased effectiveness of the active compounds and compositions which can be used according to the invention, better plant growth, increased tolerance to high or low Temperatures, increased tolerance to
Trockenheit oder Wasser- oder Bodensalzgehalt, erhöhte Blühleistung, Emteerleichterung, Dryness or water or soil salt content, increased flowering performance, easier harvesting,
Reifebeschleunigung, höhere Erträge, größere Früchte, größere Pflanzenhöhe, intensiver grüne Farbe des Blatts, frühere Blüte, höhere Qualität und/oder höherer Nährwert der Emteprodukte, höhere Zuckerkonzentration in den Früchten, bessere Lagerfähigkeit und/oder Verarbeitbarkeit der Accelerated ripening, higher yields, larger fruits, higher plant height, more intense green color of the leaf, earlier flowering, higher quality and / or higher nutritional value of the edible products, higher sugar concentration in the fruits, better storage stability and / or processability of the
Emteprodukte. Harvested products.
Als Kombinationspartner für die erfindungsgemäßen Verbindungen der Formel (1) in As a combination partner for the compounds of formula (1) according to the invention in
Mischungsformulierungen oder im Tank-Mix sind beispielsweise bekannte Wirkstoffe, die auf einer lnhibition von beispielsweise Acetolactat-Synthase, Acetyl-CoA-Carboxylase, Cellulose-Synthase, Enolpyruvylshikimat-3-phosphat-Synthase, Glutamin-Synthetase, p-Hydroxyphenylpyruvat- Dioxygenase, Phytoendesaturase, Photosystem I, Photosystem II, Protoporphyrinogen-Oxidase beruhen, einsetzbar, wie sie z.B. in Weed Research 26 (1986) 441-445 oder "The Pesticide Manual", l6th edition, The British Crop Protection Council and the Royal Soc. of Chemistry, 2012 und der dort zitierten Literatur beschrieben sind. Von besonderem Interesse ist die selektive Bekämpfung von Schadpflanzen in Kulturen von Nutz- und Zierpflanzen. Obgleich die erfindungsgemäßen Verbindungen der allgemeinen Formel (I) bereits in vielen Kulturen sehr gute bis ausreichende Selektivität aufweisen, können prinzipiell in einigen Kulturen und vor allem auch im Falle von Mischungen mit anderen Herbiziden, die weniger selektiv sind, Phytotoxizitäten an den Kulturpflanzen auftreten. Diesbezüglich sind Kombinationen Mixture formulations or in the tank mix are, for example, known active ingredients which are based on an inhibition of, for example, acetolactate synthase, acetyl-CoA carboxylase, cellulose synthase, enolpyruvylshikimate-3-phosphate synthase, glutamine synthetase, p-hydroxyphenylpyruvate dioxygenase, and , Photosystem I, Photosystem II, protoporphyrinogen oxidase, can be used, as described, for example, in Weed Research 26 (1986) 441-445 or "The Pesticide Manual", 16th edition, The British Crop Protection Council and the Royal Soc. of Chemistry, 2012 and the literature cited therein. The selective control of harmful plants in crops of useful and ornamental plants is of particular interest. Although the compounds of the general formula (I) according to the invention already have very good to sufficient selectivity in many crops, phytotoxicity can occur on the crop plants in principle in some crops and especially in the case of mixtures with other herbicides which are less selective. In this regard, combinations are
erfindungsgemäßer Verbindungen (1) von besonderem lnteresse, welche die Verbindungen (1) bzw. deren Kombinationen mit anderen Herbiziden oder Pestiziden und Safenern enthalten. Die Safener, welche in einem antidotisch wirksamen Gehalt eingesetzt werden, reduzieren die phytotoxischen Nebenwirkungen der eingesetzten Herbizide/Pestizide, z.B. in wirtschaftlich bedeutenden Kulturen wie Getreide (Weizen, Gerste, Roggen, Mais, Reis, Hirse), Zuckerrübe, Zuckerrohr, Raps, Baumwolle und Soja, vorzugsweise Getreide. Compounds (1) according to the invention of particular interest which contain the compounds (1) or their combinations with other herbicides or pesticides and safeners. The safeners, which are used in an antidotically effective content, reduce the phytotoxic side effects of the herbicides / pesticides used, e.g. in economically important crops such as cereals (wheat, barley, rye, corn, rice, millet), sugar beet, sugar cane, rapeseed, cotton and soybeans, preferably cereals.
Die Gewichtsverhältnisse von Herbizid(mischung) zu Safener hängt im Allgemeinen von der The weight ratio of herbicide (mixture) to safener generally depends on the
Aufwandmenge an Herbizid und der Wirksamkeit des jeweiligen Safeners ab und kann innerhalb weiter Grenzen variieren, beispielsweise im Bereich von 200:1 bis 1 :200, vorzugsweise 100:1 bis 1 :100, insbesondere 20:1 bis 1 :20. Die Safener können analog den Verbindungen der allgemeinen Formel (1) oder deren Mischungen mit weiteren Herbiziden/Pestiziden formuliert werden und als Application rate of herbicide and the effectiveness of the respective safener and can vary within wide limits, for example in the range from 200: 1 to 1: 200, preferably 100: 1 to 1: 100, in particular 20: 1 to 1:20. The safeners can be formulated analogously to the compounds of the general formula (1) or their mixtures with further herbicides / pesticides and as
Fertigformulierung oder Tankmischung mit den Herbiziden bereitgestellt und angewendet werden. Ready formulation or tank mix with the herbicides are provided and applied.
Zur Anwendung werden die in handelsüblicher Form vorliegenden Herbizid- oder Herbizid- Safener- Formulierungen gegebenenfalls in üblicher Weise verdünnt z.B. bei Spritzpulvem, emulgierbaren Konzentraten, Dispersionen und wasserdispergierbaren Granulaten mittels Wasser. Staubförmige Zubereitungen, Boden- bzw. Streugranulate sowie versprühbare Lösungen werden vor der Anwendung üblicherweise nicht mehr mit weiteren inerten Stoffen verdünnt. For use, the herbicide or herbicide safener formulations present in commercially available form are optionally diluted in the customary manner, for example for wettable powders, emulsifiable concentrates, dispersions and water-dispersible granules using water. Preparations in the form of dust, ground granules or granules as well as sprayable solutions are usually no longer diluted with other inert substances before use.
Äußere Bedingungen wie Temperatur, Feuchtigkeit etc. beeinflussen zu einem gewissen Teil die Aufwandmenge der Verbindungen der allgemeinen Formel (1) und/oder deren Salze. Die External conditions such as temperature, humidity etc. influence to a certain extent the application rate of the compounds of the general formula (1) and / or their salts. The
Aufwandmenge kann dabei innerhalb weiter Grenzen variieren. Für die Anwendung als Herbizid zur Bekämpfung von Schadpflanzen liegt die Gesamtmenge an Verbindungen der allgemeinen Formel (1) und deren Salze vorzugsweise im Bereich von 0,001 bis 10,0 kg/ha, bevorzugt im Bereich von 0,005 bis 5 kg/ha, weiter bevorzugt im Bereich von 0,01 bis 1,5 kg/ha, insbesondere bevorzugt im Bereich von 0,05 bis 1 kg/ha. Dies gilt sowohl für die Anwendung im Vorauflauf oder im Nachauflauf. The application rate can vary within wide limits. For use as a herbicide for controlling harmful plants, the total amount of compounds of the general formula (1) and their salts is preferably in the range from 0.001 to 10.0 kg / ha, preferably in the range from 0.005 to 5 kg / ha, more preferably in Range from 0.01 to 1.5 kg / ha, particularly preferably in the range from 0.05 to 1 kg / ha. This applies to both pre-emergence and post-emergence applications.
Bei der Anwendung von Verbindungen der allgemeinen Formel (1) und/oder deren Salzen als When using compounds of general formula (1) and / or their salts as
Pflanzenwachstumsregulator, beispielsweise als Halmverkürzer bei Kulturpflanzen, wie sie oben genannt worden sind, vorzugsweise bei Getreidepflanzen wie Weizen, Gerste, Roggen, Triticale, Hirse, Reis oder Mais, liegt die Gesamt- Aufwandmenge vorzugsweise im Bereich von 0,001 bis 2 kg/ha, vorzugsweise im Bereich von 0,005 bis 1 kg/ha, insbesondere im Bereich von 10 bis 500 g/ha, ganz besonders bevorzugt im Bereich von 20 bis 250 g/ha. Dies gilt sowohl für die Anwendung im Plant growth regulator, for example as a straw shortener in crop plants as mentioned above, preferably in cereal plants such as wheat, barley, rye, triticale, millet, rice or corn, the total application rate is preferably in the range from 0.001 to 2 kg / ha, preferably in the range from 0.005 to 1 kg / ha, in particular in the range from 10 to 500 g / ha, very particularly preferably in the range from 20 to 250 g / ha. This applies both to the application in
Vorauflauf oder im Nachauflauf. Pre-emergence or post-emergence.
Die Applikation als Halmverkürzer kann in verschiedenen Stadien des Wachstums der Pflanzen erfolgen. Bevorzugt ist beispielsweise die Anwendung nach der Bestockung am Beginn des The application as a straw shortener can take place in different stages of the growth of the plants. For example, use after planting at the beginning of the
Längenwachstums . Linear growth.
Alternativ kommt bei der Anwendung als Pflanzenwachstumsregulator auch die Behandlung des Saatguts in Frage, welche die unterschiedlichen Saatgutbeiz- und Beschichtungstechniken einschließt. Die Aufwandmenge hängt dabei von den einzelnen Techniken ab und kann in Vorversuchen ermittelt werden. Alternatively, when used as a plant growth regulator, the treatment of the seed, which includes the different seed dressing and coating techniques, can also be used. The application rate depends on the individual techniques and can be determined in preliminary tests.
Als Kombinationspartner für die erfindungsgemäßen Verbindungen der allgemeinen Formel (I) in erfindungsgemäßen Mitteln (z.B. Mischungsformulierungen oder im Tank-Mix) sind beispielsweise bekannte Wirkstoffe, die auf einer Inhibition von beispielsweise Acetolactat-Synthase, Acetyl-CoA- Carboxylase, Cellulose-Synthase, Enolpyruvylshikimat-3-phosphat-Synthase, Glutamin-Synthetase, p- Hydroxyphenylpyruvat-Dioxygenase, Phytoendesaturase, Photosystem I, Photosystem II oder As a combination partner for the compounds of the general formula (I) according to the invention in compositions according to the invention (for example mixture formulations or in a tank mix), there are, for example, known active substances which are based on inhibition of, for example, acetolactate synthase, acetyl-CoA carboxylase, cellulose synthase, enolpyruvylshikimate -3-phosphate synthase, glutamine synthetase, p-hydroxyphenylpyruvate dioxygenase, phytoendesaturase, Photosystem I, Photosystem II or
Protoporphyrinogen-Oxidase beruhen, einsetzbar, wie sie z.B. aus Weed Research 26 (1986) 441-445 oder "The Pesticide Manual", 16th edition, The British Crop Protection Council und the Royal Soc. of Chemistry, 2012 und dort zitierter Literatur beschrieben sind. Nachfolgend werden beispielhaft bekannte Herbizide oder Pflanzenwachstumsregulatoren genannt, die mit den erfindungsgemäßen Verbindungen kombiniert werden können, wobei diese Wirkstoffe entweder mit ihrem "common name" in der englischsprachigen Variante gemäß International Organization for Standardization (ISO) oder mit dem chemischen Namen bzw. mit der Codenummer bezeichnet sind. Dabei sind stets sämtliche Protoporphyrinogen oxidase are used, such as those e.g. from Weed Research 26 (1986) 441-445 or "The Pesticide Manual", 16th edition, The British Crop Protection Council and the Royal Soc. of Chemistry, 2012 and the literature cited therein. Known herbicides or plant growth regulators are mentioned below by way of example, which can be combined with the compounds according to the invention, these active compounds either with their "common name" in the English-language variant according to the International Organization for Standardization (ISO) or with the chemical name or with the code number are designated. There are always everyone
Anwendungsformen wie beispielsweise Säuren, Salze, Ester sowie auch alle isomeren Formen wie Stereoisomere und optische Isomere umfaßt, auch wenn diese nicht explizit erwähnt sind. Application forms such as acids, salts, esters and also all isomeric forms such as stereoisomers and optical isomers, even if these are not explicitly mentioned.
Beispiele für solche herbiziden Mischungspartner sind: Examples of such herbicidal mixture partners are:
Acetochlor, acifluorfen, acifluorfen-sodium, aclonifen, alachlor, allidochlor, alloxydim, alloxydim- sodium, ametryn, amicarbazone, amidochlor, amidosulfüron, 4-amino-3-chloro-6-(4-chloro-2-fluoro-3- methylphenyl)-5-fluoropyridine-2-carboxylic acid, aminocyclopyrachlor, aminocyclopyrachlor- potassium, aminocyclopyrachlor-methyl, aminopyralid, amitrole, ammoniumsulfamate, anilofos, asulam, atrazine, azafenidin, azimsulfuron, beflubutamid, benazolin, benazolin-ethyl, benfluralin, benfüresate, bensulfüron, bensulfüron-methyl, bensulide, bentazone, benzobicyclon, benzofenap, bicyclopyron, bifenox, bilanafos, bilanafos-sodium, bispyribac, bispyribac-sodium, bromacil, bromobutide, bromofenoxim, bromoxynil, bromoxynil-butyrate, -potassium, -heptanoate und -octanoate, busoxinone, butachlor, butafenacil, butamifos, butenachlor, butralin, butroxydim, butylate, cafenstrole, carbetamide, carfentrazone, carfentrazone-ethyl, chloramben, chlorbromuron, chlorfenac, chlorfenac- sodium, chlorfenprop, chlorflurenol, chlorflurenol-methyl, chloridazon, chlorimuron, chlorimuron-ethyl, chlorophthalim, chlorotoluron, chlorthal-dimethyl, chlorsulfuron, cinidon, cinidon-ethyl, cinmethylin, cinosulfuron, clacyfos, clethodim, clodinafop, clodinafop-propargyl, clomazone, clomeprop, clopyralid, cloransulam, cloransulam-methyl, cumyluron, cyanamide, cyanazine, cycloate, cyclopyrimorate, cyclosulfamuron, cycloxydim, cyhalofop, cyhalofop-butyl, cyprazine, 2,4-D, 2,4-D-butotyl, -butyl, - dimethylammonium, -diolamin, -ethyl, 2-ethylhexyl, -isobutyl, -isooctyl, -isopropylammonium, - potassium, -triisopropanolammonium und -trolamine, 2,4-DB, 2,4-DB-butyl, -dimethylammonium, isooctyl, -potassium und -sodium, daimuron (dymron), dalapon, dazomet, n-decanol, desmedipham, detosyl-pyrazolate (DTP), dicamba, dichlobenil, 2-(2,4-dichlorobenzyl)-4,4-dimethyl-l,2-oxazolidin-3- one, 2-(2,5-dichlorobenzyl)-4,4-dimethyl-l,2-oxazolidin-3-one, dichlorprop, dichlorprop-P, diclofop, diclofop-methyl, diclofop-P-methyl, diclosulam, difenzoquat, diflufenican, diflufenzopyr, diflufenzopyr- sodium, dimefuron, dimepiperate, dimethachlor, dimethametryn, dimethenamid, dimethenamid-P, dimetrasulfuron, dinitramine, dinoterb, diphenamid, diquat, diquat-dibromid, dithiopyr, diuron, DNOC, endothal, EPTC, esprocarb, ethalfluralin, ethametsulfuron, ethametsulfuron-methyl, ethiozin, ethofumesate, ethoxyfen, ethoxyfen- ethyl, ethoxysulfuron, etobenzanid, F-9600, F-5231, i.e. N-[2- Chlor-4-fluor-5-[4-(3-fluorpropyl)-4,5-dihydro-5-oxo-lH-tetrazol-l-yl]-phenyl]-ethansulfonamid, F- 7967, i.e. 3-[7-Chlor-5-fluor-2-(trifluormethyl)-lH-benzimidazol-4-yl]-l-methyl-6- (trifluormethyl)pyrimidin-2,4(lH,3H)-dion, fenoxaprop, fenoxaprop-P, fenoxaprop-ethyl, fenoxaprop-P- ethyl, fenoxasulfone, fenquinotrione, fentrazamide, flamprop, flamprop-M-isopropyl, flamprop-M- methyl, flazasulfuron, florasulam, fluazifop, fluazifop-P, fluazifop-butyl, fluazifop-P-butyl, Acetochlor, acifluorfen, acifluorfen-sodium, aclonifen, alachlor, allidochlor, alloxydim, alloxydim-sodium, ametryn, amicarbazone, amidochlor, amidosulfüron, 4-amino-3-chloro-6- (4-chloro-2-fluoro-3-methylphenyl ) -5-fluoropyridine-2-carboxylic acid, aminocyclopyrachlor, aminocyclopyrachlor-potassium, aminocyclopyrachlor-methyl, aminopyralid, amitrole, ammonium sulfamate, anilofos, asulam, atrazine, azafenidin, azimsulfuron, beflubutamid, benazolin, benuralone, benazolin, benazolin , bensulfüron-methyl, bensulide, bentazone, benzobicyclon, benzofenap, bicyclopyron, bifenox, bilanafos, bilanafos-sodium, bispyribac, bispyribac-sodium, bromacil, bromobutide, bromofenoxim, bromoxynil, bromoxanoilate-butyrhe, -ctano-nano-butyrate, -borate-borate busoxinone, butachlor, butafenacil, butamifos, butenachlor, butralin, butroxydim, butylate, cafenstrole, carbetamide, carfentrazone, carfentrazone-ethyl, chloramben, chlorbromuron, chlorfenac, chlorfenac-sodium, chlorfenprop, chlorflurenol, chlorflurenazonon, chloruron-imur, methyluron, chloruronazon, methyluronimone ethyl, chlorophthalim, chlorotoluron, chlorthal-dimethyl, chlorsulfuron, cinidon, cinidon-ethyl, cinmethylin, cinosulfuron, clacyfos, clethodim, clodinafop, clodinafop-propargyl, clomazone, clomeprop, clopyralid, cloransulam, cloransulam-methyl, cumyluron, cyanamide, cyanazine, cycloate, cyclopyrimorate, cyclosulfamuron, cycloxydim, cyhalofop, cyhalofop-butyl, cyprazine, 2,4-D, 2,4-D-butotyl, -butyl, - dimethylammonium, -diolamine, -ethyl, 2-ethylhexyl, -isobutyl, - isooctyl, isopropylammonium, potassium, triisopropanolammonium and trolamine, 2,4-DB, 2,4-DB-butyl, dimethylammonium, isooctyl, potassium and sodium, daimuron (dymron), dalapon, dazomet, n- decanol, desmedipham, detosyl-py razolate (DTP), dicamba, dichlobenil, 2- (2,4-dichlorobenzyl) -4,4-dimethyl-1,2-oxazolidin-3-one, 2- (2,5-dichlorobenzyl) -4,4-dimethyl -l, 2-oxazolidin-3-one, dichloroprop, dichloroprop-P, diclofop, diclofop-methyl, diclofop-P-methyl, diclosulam, difenzoquat, diflufenican, diflufenzopyr, diflufenzopyr-sodium, dimefuron, dimepipereneth, dimethetrynlor, dimethetrynlor, dimethetrynlor , dimethenamid-P, dimetrasulfuron, dinitramine, dinoterb, diphenamid, diquat, diquat-dibromid, dithiopyr, diuron, DNOC, endothal, EPTC, esprocarb, ethalfluralin, ethametsulfuron, ethametsulfuron-methyl, ethiozin, ethofumesoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxy-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-ethoxysulfuron-methyl , etobenzanide, F-9600, F-5231, ie N- [2-chloro-4-fluoro-5- [4- (3-fluoropropyl) -4,5-dihydro-5-oxo-lH-tetrazole-l- yl] -phenyl] -ethanesulfonamide, F- 7967, ie 3- [7-chloro-5-fluoro-2- (trifluoromethyl) -lH-benzimidazol-4-yl] -l-methyl-6- (trifluoromethyl) pyrimidine- 2,4 (lH, 3H) -dione, fenoxaprop, fenoxaprop-P, fenoxaprop-ethyl, fenoxaprop-P-ethyl, fenoxasulfone, fenquinotrione, fentrazamide, flamprop, flamprop-M-isopropyl, flamprop-M-methyl, flazasulfuron, florasulam, fluazifop, fluazifop-P, fluazifop-butyl, fluazifop-P-butyl,
flucarbazone, flucarbazone-sodium, flucetosulfuron, fluchloralin, flufenacet, flufenpyr, flufenpyr-ethyl, flumetsulam, flumiclorac, flumiclorac-pentyl, flumioxazin, fluometuron, flurenol, flurenol-butyl, - dimethylammonium und -methyl, fluoroglycofen, fluoroglycofen-ethyl, flupropanate, flupyrsulfuron, flupyrsulfuron-methyl-sodium, fluridone, flurochloridone, fluroxypyr, fluroxypyr-meptyl, flurtamone, fluthiacet, fluthiacet-methyl, fomesafen, fomesafen-sodium, foramsulfuron, fosamine, glufosinate, glufosinate-ammonium, glufosinate-P-sodium, glufosinate-P-ammonium, glufosinate-P-sodium, glyphosate, glyphosate-ammonium, -isopropylammonium, -diammonium, -dimethylammonium, - potassium, -sodium und -trimesium, H-9201, i.e. 0-(2,4-Dimethyl-6-nitrophenyl)-0-ethyl- isopropylphosphoramidothioat, halauxifen, halauxifen-methyl, halosafen, halosulfuron, halosulfuron- methyl, haloxyfop, haloxyfop-P, haloxyfop-ethoxyethyl, haloxyfop-P-ethoxyethyl, haloxyfop-methyl, haloxyfop-P-methyl, hexazinone, HW-02, i.e. l-(Dimethoxyphosphoryl)-ethyl-(2,4- dichlorphenoxy)acetat, imazamethabenz, Imazamethabenz-methyl, imazamox, imazamox-ammonium, imazapic, imazapic-ammonium, imazapyr, imazapyr-isopropylammonium, imazaquin, imazaquin- ammonium, imazethapyr, imazethapyr-immonium, imazosulfuron, indanofan, indaziflam, iodosulfuron, iodosulfuron-methyl-sodium, ioxynil, ioxynil-octanoate, -potassium und sodium, ipfencarbazone, isoproturon, isouron, isoxaben, isoxaflutole, karbutilate, KUH-043, i.e. 3-({[5-(Difluormethyl)-l- methyl-3-(trifluormethyl)- 1 H-pyrazol-4-yl]methyl} sulfonyl)-5,5-dimethyl-4,5-dihydro- 1 ,2-oxazol, ketospiradox, lactofen, lenacil, linuron, MCPA, MCPA-butotyl, -dimethylammonium, -2-ethylhexyl, - isopropylammonium, -potassium und -sodium, MCPB, MCPB-methyl, -ethyl und -sodium, mecoprop, mecoprop-sodium, und -butotyl, mecoprop-P, mecoprop-P-butotyl, -dimethylammonium, -2-ethylhexyl und -potassium, mefenacet, mefluidide, mesosulfuron, mesosulfuron-methyl, mesotrione, flucarbazone, flucarbazone-sodium, flucetosulfuron, fluchloralin, flufenacet, flufenpyr, flufenpyr-ethyl, flumetsulam, flumiclorac, flumiclorac-pentyl, flumioxazin, fluometuron, flurenol, flurenol-butyl, -dimethylammonoglypane, and -methyl-fluorine-cyanogen flupyrsulfuron, flupyrsulfuron-methyl-sodium, fluridone, flurochloridone, fluroxypyr, fluroxypyr-meptyl, flurtamone, fluthiacet, fluthiacet-methyl, fomesafen, fomesafen-sodium, foramsulfuron, fosamine, glufosinate, glufosinate ammonium, pufosinate ammonium P-ammonium, glufosinate-P-sodium, glyphosate, glyphosate-ammonium, -isopropylammonium, -diammonium, -dimethylammonium, - potassium, -sodium and -trimesium, H-9201, ie 0- (2,4-dimethyl-6- nitrophenyl) -0-ethyl-isopropylphosphoramidothioat, halauxifen, halauxifen-methyl, halosafen, halosulfuron, halosulfuron-methyl, haloxyfop, haloxyfop-P, haloxyfop-ethoxyethyl, haloxyfop-P-ethoxyethyl, haloxyfop-methyl, haloxyfop-P-methyl , HW-0 2, ie l- (dimethoxyphosphoryl) ethyl (2,4-dichlorophenoxy) acetate, imazamethabenz, imazamethabenz-methyl, imazamox, imazamox-ammonium, imazapic, imazapic-ammonium, imazapyr, imazapyr-isopropylammonium, imazaquin, imazaquine imazethapyr, imazethapyr-immonium, imazosulfuron, indanofan, indaziflam, iodosulfuron, iodosulfuron-methyl-sodium, ioxynil, ioxynil-octanoate, -potassium and sodium, ipfencarbazone, isoproturon, isouron, isoxaben, isoxaflutole, carbutilate, KUH-043, ie 3 - ({[5- (difluoromethyl) -l-methyl-3- (trifluoromethyl) - 1 H-pyrazol-4-yl] methyl} sulfonyl) - 5,5-dimethyl-4,5-dihydro-1,2-oxazole, ketospiradox, lactofen, lenacil, linuron, MCPA, MCPA-butotyl, dimethylammonium, -2-ethylhexyl, isopropylammonium, potassium and sodium, MCPB , MCPB-methyl, -ethyl and -sodium, mecoprop, mecoprop-sodium, and -butotyl, mecoprop-P, mecoprop-P-butotyl, -dimethylammonium, -2-ethylhexyl and -potassium, mefenacet, mefluidide, mesosulfuron, mesosulfuron- methyl, mesotrione,
methabenzthiazuron, metam, metamifop, metamitron, metazachlor, metazosulfuron, methabenzthiazuron, metam, metamifop, metamitron, metazachlor, metazosulfuron,
methabenzthiazuron, methiopyrsulfuron, methiozolin, methyl isothiocyanate, metobromuron, metolachlor, S-metolachlor, metosulam, metoxuron, metribuzin, metsulfuron, metsulfuron-methyl, molinat, monolinuron, monosulfuron, monosulfuron-ester, MT-5950, i.e. N-[3-chlor-4-(l-methylethyl)- phenyl]-2-methylpentanamid, NGGC-011, napropamide, NC-310, i.e. 4-(2,4-Dichlorbenzoyl)-l-methyl- 5-benzyloxypyrazol, neburon, nicosulfuron, nonanoic acid (Pelargonsäure), norflurazon, oleic acid (fatty acids), orbencarb, orthosulfamuron, oryzalin, oxadiargyl, oxadiazon, oxasulfuron, oxaziclomefon, oxyfluorfen, paraquat, paraquat dichloride, pebulate, pendimethalin, penoxsulam, pentachlorphenol, pentoxazone, pethoxamid, petroleum oils, phenmedipham, picloram, picolinafen, pinoxaden, piperophos, pretilachlor, primisulfuron, primisulfuron-methyl, prodiamine, profoxydim, prometon, prometryn, propachlor, propanil, propaquizafop, propazine, propham, propisochlor, propoxycarbazone, propoxycarbazone-sodium, propyrisulfuron, propyzamide, prosulfocarb, prosulfuron, pyraclonil, pyraflufen, pyraflufen- ethyl, pyrasulfotole, pyrazolynate (pyrazolate), pyrazosulfuron, pyrazosulfuron- ethyl, pyrazoxyfen, pyribambenz, pyribambenz-isopropyl, pyribambenz-propyl, pyribenzoxim, pyributicarb, pyridafol, pyridate, pyriftalid, pyriminobac, pyriminobac-methyl, pyrimisulfan, pyrithiobac, pyrithiobac-sodium, pyroxasulfone, pyroxsulam, quinclorac, quinmerac, quinoclamine, quizalofop, quizalofop-ethyl, quizalofop-P, quizalofop-P-ethyl, quizalofop-P-tefuryl, rimsulfuron, saflufenacil, sethoxydim, siduron, simazine, simetryn, SL-261, sulcotrion, sulfentrazone, sulfometuron, sulfometuron-methyl, sulfosulfuron, , SYN-523, SYP-249, i.e. l-Ethoxy-3-methyl-l-oxobut-3-en-2-yl- 5-[2-chlor-4-(trifluormethyl)phenoxy]-2-nitrobenzoat, SYP-300, i.e. l-[7-Fluor-3-oxo-4-(prop-2-in-l- yl)-3,4-dihydro-2H-l,4-benzoxazin-6-yl]-3-propyl-2-thioxoimidazolidin-4,5-dion, 2,3,6-TBA, TCA (Trifluoressigsäure), TCA-sodium, tebuthiuron, tefuryltrione, tembotrione, tepraloxydim, terbacil, terbucarb, terbumeton, terbuthylazin, terbutryn, thenylchlor, thiazopyr, thiencarbazone, thiencarbazone- methyl, thifensulfuron, thifensulfuron-methyl, thiobencarb, tiafenacil, tolpyralate, topramezone, tralkoxydim, triafamone, tri-allate, triasulfuron, triaziflam, tribenuron, tribenuron-methyl, triclopyr, trietazine, trifloxysulfuron, trifloxysulfuron-sodium, trifludimoxazin, trifluralin, triflusulfuron, triflusulfuron-methyl, tritosulfuron, urea sulfate, vemolate, XDE-848, ZJ-0862, i.e. 3,4-Dichlor-N-{2- [(4,6-dimethoxypyrimidin-2-yl)oxy]benzyl}anilin, sowie die folgenden Verbindungen: methabenzthiazuron, methiopyrsulfuron, methiozolin, methyl isothiocyanate, metobromuron, metolachlor, S-metolachlor, metosulam, metoxuron, metribuzin, metsulfuron, metsulfuron-methyl, molinat, monolinuron, monosulfuron, MT-59-chloro-3-ester -4- (l-methylethyl) phenyl] -2-methylpentanamide, NGGC-011, napropamide, NC-310, ie 4- (2,4-dichlorobenzoyl) -l-methyl-5-benzyloxypyrazole, neburon, nicosulfuron, nonanoic acid (pelargonic acid), norflurazon, oleic acid (fatty acids), orbencarb, orthosulfamuron, oryzalin, oxadiargyl, oxadiazon, oxasulfuron, oxaziclomefon, oxyfluorfen, paraquat, paraquat dichloride, pebulate, pendimethalin, penoxsulone, pentachlorone, pentachlorone, pentachlorone, pentachlorone phenmedipham, picloram, picolinafen, pinoxaden, piperophos, pretilachlor, primisulfuron, primisulfuron-methyl, prodiamine, profoxydim, prometon, prometryn, propachlor, propanil, propaquizafop, propazine, propham, propisochlor, propoxycarbazone, propoxycarbazone, propoxycarbazone furon, Propyzamide, prosulfocarb, prosulfuron, pyraclonil, pyraflufen, pyraflufen- ethyl, pyrasulfotole, pyrazolynate (pyrazolate), pyrazosulfuron, pyrazosulfuron- ethyl, pyrazoxyfen, pyribambenz, pyribambenz-isopropyl, pyribambenz-propyl, pyribenzoxim, pyributicarb, pyridafol, pyridate, pyriftalid , pyriminobac, pyriminobac-methyl, pyrimisulfan, pyrithiobac, pyrithiobac-sodium, pyroxasulfone, pyroxsulam, quinclorac, quinmerac, quinoclamine, quizalofop, quizalofop-ethyl, quizalofop-P, quizalofop-P-ethyl, quizalofonofulfonyl , sethoxydim, siduron, simazine, simetryn, SL-261, sulcotrione, sulfentrazone, sulfometuron, sulfometuron-methyl, sulfosulfuron,, SYN-523, SYP-249, ie l-ethoxy-3-methyl-l-oxobut-3-en -2-yl- 5- [2-chloro-4- (trifluoromethyl) phenoxy] -2-nitrobenzoate, SYP-300, ie l- [7-fluoro-3-oxo-4- (prop-2-in-l yl) -3,4-dihydro-2H-l, 4-benzoxazin-6-yl] -3-propyl-2-thioxoimidazolidin-4,5-dione, 2,3,6-TBA, TCA (trifluoroacetic acid), TCA-sodium, tebuthiuron, tefuryltrione , Tembotrione, tepraloxydim, terbacil, terbucarb, terbumeton, terbuthylazine, terbutryn, thenylchlor, thiazopyr, thiencarbazone, thiencarbazone- methyl, thifensulfuron, thifensulfuron-methyl, thiobencarb, tiafenacil, tolpyralate, topramezone, tralkoxydim, triafamone, tri-allate, triasulfuron, triaziflam , tribenuron, tribenuron-methyl, triclopyr, trietazine, trifloxysulfuron, trifloxysulfuron-sodium, trifludimoxazin, trifluralin, triflusulfuron, triflusulfuron-methyl, tritosulfuron, urea sulfate, vemolate, ZJ-08-chlorine, XDE-848 - {2- [(4,6-dimethoxypyrimidin-2-yl) oxy] benzyl} aniline, and the following compounds:
Beispiele für Pflanzenwachstumsregulatoren als mögliche Mischungspartner sind: Examples of plant growth regulators as possible mixing partners are:
Acibenzolar, acibenzolar-S-methyl, 5-Aminolävulinsäure, ancymidol, 6-benzylaminopurine, Acibenzolar, acibenzolar-S-methyl, 5-aminolevulinic acid, ancymidol, 6-benzylaminopurine,
Brassinolid, Catechin, chlormequat chloride, cloprop, cyclanilide, 3-(Cycloprop-l-enyl)propionsäure, daminozide, dazomet, n-decanol, dikegulac, dikegulac-sodium, endothal, endothal- dipotassium, -disodium, und mono(N,N-dimethylalkylammonium), ethephon, flumetralin, flurenol, flurenol-butyl, flurprimidol, forchlorfenuron, gibberellic acid, inabenfide, indol-3-acetic acid (IAA), 4- indol-3-ylbutyric acid, isoprothiolane, probenazole, Jasmonsäure, Jasmonsäuremethylester, maleic hydrazide, mepiquat chloride, 1 -methylcyclopropene, 2-(l-naphthyl)acetamide, 1 -naphthylacetic acid, 2- naphthyloxyacetic acid, nitrophenolate-mixture, 4-Oxo-4[(2-phenylethyl)amino]buttersäure, paclobutrazol, N-phenylphthalamic acid, prohexadione, prohexadione-calcium, prohydrojasmone, Salicylsäure, Strigolacton, tecnazene, thidiazuron, triacontanol, trinexapac, trinexapac-ethyl, tsitodef, uniconazole, uniconazole-P. Ebenfalls als Kombinationspartner für die erfindungsgemäßen Verbindungen der allgemeinen Formel (I) kommen beispielsweise die folgenden Safener in Frage: Brassinolide, catechin, chlormequat chloride, cloprop, cyclanilide, 3- (cycloprop-l-enyl) propionic acid, daminozide, dazomet, n-decanol, dikegulac, dikegulac-sodium, endothal, endothal-dipotassium, -disodium, and mono (N, N-dimethylalkylammonium), ethephon, flumetralin, flurenol, flurenol-butyl, flurprimidol, forchlorfenuron, gibberellic acid, inabenfide, indol-3-acetic acid (IAA), 4-indol-3-ylbutyric acid, isoprothiolane, probenazole, jasmonic acid, jasmonic acid , maleic hydrazide, mepiquat chloride, 1 -methylcyclopropene, 2- (l-naphthyl) acetamide, 1 -naphthylacetic acid, 2-naphthyloxyacetic acid, nitrophenolate-mixture, 4-oxo-4 [(2-phenylethyl) amino] butyric acid, paclobutrazole , N-phenylphthalamic acid, prohexadione, prohexadione-calcium, prohydrojasmone, salicylic acid, strigolactone, tecnazene, thidiazuron, triacontanol, trinexapac, trinexapac-ethyl, tsitodef, uniconazole, uniconazole-P. The following safeners, for example, are also suitable as combination partners for the compounds of the general formula (I) according to the invention:
Sl) Verbindungen aus der Gruppe heterocyclischer Carbonsäurederivate: Sl) compounds from the group of heterocyclic carboxylic acid derivatives:
S 1 a) V erbindungen vom Typ der Dichlorphenylpyrazolin-3 -carbonsäure (S 1 a), vorzugsweise S 1 a ) Compounds of the dichlorophenylpyrazoline-3-carboxylic acid type (S 1 a ), preferably
Verbindungen wie l-(2,4-Dichlorphenyl)-5-(ethoxycarbonyl)-5-methyl-2-pyrazolin-3-carbonsäure, Compounds such as 1- (2,4-dichlorophenyl) -5- (ethoxycarbonyl) -5-methyl-2-pyrazoline-3-carboxylic acid,
1 -(2,4-Dichlorphenyl)-5-(ethoxycarbonyl)-5-methyl-2-pyrazolin-3-carbonsäureethylester (S 1 - 1 ) ("Mefenpyr-diethyl"), und verwandte Verbindungen, wie sie in der WO-A-91/07874 beschrieben sind; 1 - (2,4-dichlorophenyl) -5- (ethoxycarbonyl) -5-methyl-2-pyrazoline-3-carboxylic acid ethyl ester (S 1 - 1) ("Mefenpyr-diethyl"), and related compounds as described in WO-A-91/07874;
S 1 b) Derivate der Dichlorphenylpyrazolcarbonsäure (S 1 b), vorzugsweise V erbindungen wie S 1 b ) derivatives of dichlorophenylpyrazole carboxylic acid (S 1 b ), preferably compounds such as
1 -(2,4-Dichlorphenyl)-5-methylpyrazol-3 -carbonsäureethylester (S 1 -2), 1 - (2,4-dichlorophenyl) -5-methylpyrazole-3-carboxylic acid ethyl ester (S 1 -2),
1 -(2,4-Dichlorphenyl)-5-isopropylpyrazol-3-carbonsäureethylester (S 1 -3), 1 - (2,4-dichlorophenyl) -5-isopropylpyrazole-3-carboxylic acid ethyl ester (S 1 -3),
1 -(2,4-Dichlorphenyl)-5-(l , 1 -dimethyl-ethyl)pyrazol-3-carbonsäureethylester (S 1 -4) und verwandte Verbindungen, wie sie in EP-A-333131 und EP-A-269806 beschrieben sind; 1 - (2,4-dichlorophenyl) -5- (1,1-dimethyl-ethyl) pyrazole-3-carboxylic acid ethyl ester (S 1 -4) and related compounds as described in EP-A-333131 and EP-A-269806 are described;
Slc) Derivate der l,5-Diphenylpyrazol-3-carbonsäure (Slc), vorzugsweise Verbindungen wie Sl c ) derivatives of l, 5-diphenylpyrazole-3-carboxylic acid (Sl c ), preferably compounds such as
l-(2,4-Dichlorphenyl)-5-phenylpyrazol-3-carbonsäureethylester (Sl-5), ethyl 1- (2,4-dichlorophenyl) -5-phenylpyrazole-3-carboxylate (S1-5),
l-(2-Chlorphenyl)-5-phenylpyrazol-3-carbonsäuremethylester (Sl-6) und verwandte 1- (2-Chlorophenyl) -5-phenylpyrazole-3-carboxylic acid methyl ester (S1-6) and related
Verbindungen wie sie beispielsweise in der EP-A-268554 beschrieben sind; Compounds as described for example in EP-A-268554;
Sld) Verbindungen vom Typ der Triazolcarbonsäuren (Sld), vorzugsweise Verbindungen wie Sl d ) compounds of the triazole carboxylic acid type (Sl d ), preferably compounds such as
Fenchlorazol(-ethylester), d.h. l-(2,4-Dichlorphenyl)-5-trichlormethyl-(lH)-l,2,4-triazol-3- carbonsäureethylester (Sl-7), und verwandte Verbindungen, wie sie in EP-A-174562 und EP-A-346620 beschrieben sind; Fenchlorazole (ethyl ester), i.e. 1- (2,4-dichlorophenyl) -5-trichloromethyl- (lH) -l, 2,4-triazole-3-carboxylic acid ethyl ester (S1-7), and related compounds as described in EP-A-174562 and EP- A-346620;
Sle) Verbindungen vom Typ der 5-Benzyl- oder 5-Phenyl-2-isoxazolin-3- carbonsäure, oder der 5,5- Diphenyl-2-isoxazolin-3-carbonsäure(Sle), vorzugsweise Verbindungen wie Sl e ) compounds of the type of 5-benzyl- or 5-phenyl-2-isoxazoline-3-carboxylic acid, or of 5,5-diphenyl-2-isoxazoline-3-carboxylic acid (Sl e ), preferably compounds such as
5-(2,4-Dichlorbenzyl)-2-isoxazolin-3 -carbonsäureethylester (Sl-8) oder 5- (2,4-dichlorobenzyl) -2-isoxazoline-3-carboxylic acid ethyl ester (Sl-8) or
5-Phenyl-2-isoxazolin-3-carbonsäureethylester (Sl-9) und verwandte Verbindungen, wie sie in WO-A-91/08202 beschrieben sind, bzw. 5,5-Diphenyl-2-isoxazolin-carbonsäure (S1-10) oder 5,5-Diphenyl-2-isoxazolin-3-carbonsäureethylester (Sl-l 1) ("Isoxadifen-ethyl") 5-phenyl-2-isoxazoline-3-carboxylic acid ethyl ester (S1-9) and related compounds, as described in WO-A-91/08202, or 5,5-diphenyl-2-isoxazoline-carboxylic acid (S1-10 ) or 5,5-Diphenyl-2-isoxazoline-3-carboxylic acid ethyl ester (S1-L 1) ("Isoxadifen-ethyl")
oder -n-propylester (S1-12) oder 5-(4-Fluorphenyl)-5-phenyl-2-isoxazolin-3-carbon- säureethylester (S 1-13), wie sie in der Patentanmeldung WO-A-95/07897 beschrieben sind. or -n-propyl ester (S1-12) or 5- (4-fluorophenyl) -5-phenyl-2-isoxazoline-3-carboxylic acid ethyl ester (S 1-13), as described in patent application WO-A-95 / 07897 are described.
S2) Verbindungen aus der Gruppe der 8-Chinolinoxyderivate (S2): S2) Compounds from the group of 8-quinoline oxy derivatives (S2):
S2a) Verbindungen vom Typ der 8-Chinolinoxyessigsäure (S2a), vorzugsweise S2 a ) Compounds of the 8-quinolinoxyacetic acid (S2 a ) type, preferably
(5-Chlor-8-chinolinoxy)essigsäure-(l-methylhexyl)-ester ("Cloquintocet-mexyl") (S2-1), (5-Chlor-8-chinolinoxy)essigsäure-(l,3-dimethyl-but-l-yl)-ester (S2-2), (5-chloro-8-quinolinoxy) acetic acid (1-methylhexyl) ester ("Cloquintocet-mexyl") (S2-1), (5-chloro-8-quinolinoxy) acetic acid (1,3-dimethyl-but -l-yl) -ester (S2-2),
(5-Chlor-8-chinolinoxy)essigsäure-4-allyl-oxy-butylester (S2-3), (5-chloro-8-quinolinoxy) 4-allyl-oxy-butyl acetate (S2-3),
(5-Chlor-8-chinolinoxy)essigsäure-l-allyloxy-prop-2-ylester (S2-4), (5-chloro-8-quinolinoxy) acetic acid l-allyloxy prop-2-yl ester (S2-4),
(5-Chlor-8-chinolinoxy)essigsäureethylester (S2-5), (5-chloro-8-quinolinoxy) ethyl acetate (S2-5),
(5-Chlor-8-chinolinoxy)essigsäuremethylester (S2-6), (5-chloro-8-quinolinoxy) methyl acetate (S2-6),
(5-Chlor-8-chinolinoxy)essigsäureallylester (S2-7), (5-chloro-8-quinolinoxy) allyl acetate (S2-7),
(5-Chlor-8-chinolinoxy)essigsäure-2-(2-propyliden-iminoxy)-l-ethylester (S2-8), (5-Chlor-8-chinolinoxy)essigsäure-2-oxo-prop-l-ylester (S2-9) und verwandte Verbindungen, wie sie in EP-A-86750, EP-A-94349 und EP-A-191736 oder EP-A-0 492 366 beschrieben sind, sowie (5-Chlor-8-chinolinoxy)essigsäure (S2-10), deren Hydrate und Salze, beispielsweise deren Lithium-, Natrium- Kalium-, Kalzium-, Magnesium-, Aluminium-, Eisen-, Ammonium-, quartäre Ammonium-, Sulfonium-, oder Phosphoniumsalze wie sie in der WO-A-2002/34048 beschrieben sind; (5-chloro-8-quinolinoxy) acetic acid 2- (2-propylidene-iminoxy) -l-ethyl ester (S2-8), (5-Chloro-8-quinolinoxy) 2-oxo-prop-l-yl acetate (S2-9) and related compounds as described in EP-A-86750, EP-A-94349 and EP-A-191736 or EP-A-0 492 366 are described, and (5-chloro-8-quinolinoxy) acetic acid (S2-10), their hydrates and salts, for example their lithium, sodium, potassium, calcium, magnesium, aluminum , Iron, ammonium, quaternary ammonium, sulfonium, or phosphonium salts as described in WO-A-2002/34048;
S2b) Verbindungen vom Typ der (5-Chlor-8-chinolinoxy)malonsäure (S2b), vorzugsweise S2 b ) Compounds of the type of (5-chloro-8-quinolinoxy) malonic acid (S2 b ), preferably
Verbindungen wie (5-Chlor-8-chinolinoxy)malonsäurediethylester, Compounds such as (5-chloro-8-quinolinoxy) diethyl malonate,
(5-Chlor-8-chinolinoxy)malonsäurediallylester, (5-chloro-8-quinolinoxy) malonate,
(5-Chlor-8-chinolinoxy)malonsäure-methyl-ethylester und verwandte Verbindungen, wie sie in EP-A-0 582 198 beschrieben sind. Methyl (5-chloro-8-quinolinoxy) malonic acid and related compounds as described in EP-A-0 582 198.
53) Wirkstoffe vom Typ der Dichloracetamide (S3), die häufig als Vorauflaufsafener 53) Active substances of the type of dichloroacetamide (S3), often as pre-emergence safeners
(bodenwirksame Safener) angewendet werden, wie z. B. (Soil-effective safeners), such as B.
"Dichlormid" (N,N-Diallyl-2,2-dichloracetamid) (S3-1), "Dichlormid" (N, N-diallyl-2,2-dichloroacetamide) (S3-1),
"R-29148" (3-Dichloracetyl-2,2,5-trimethyl-l,3-oxazolidin) der Firma Stauffer (S3-2), "R-28725" (3-Dichloracetyl-2,2,-dimethyl-l,3-oxazolidin) der Firma Stauffer (S3-3), "R-29148" (3-dichloroacetyl-2,2,5-trimethyl-1,3-oxazolidine) from Stauffer (S3-2), "R-28725" (3-dichloroacetyl-2,2, -dimethyl- 1,3-oxazolidine) from Stauffer (S3-3),
"Benoxacor" (4-Dichloracetyl-3,4-dihydro-3-methyl-2H-l,4-benzoxazin) (S3-4), "Benoxacor" (4-dichloroacetyl-3,4-dihydro-3-methyl-2H-l, 4-benzoxazine) (S3-4),
"PPG-1292" (N-Allyl-N-[(l,3-dioxolan-2-yl)-methyl]-dichloracetamid) der Firma PPG "PPG-1292" (N-allyl-N - [(1,3-dioxolan-2-yl) methyl] dichloroacetamide) from PPG
Industries (S3-5), Industries (S3-5),
"DKA-24" (N-Allyl-N-[(allylaminocarbonyl)methyl]-dichloracetamid) der Firma Sagro-Chem (S3-6), "DKA-24" (N-allyl-N - [(allylaminocarbonyl) methyl] dichloroacetamide) from Sagro-Chem (S3-6),
"AD-67" oder "MON 4660" (3-Dichloracetyl-l-oxa-3-aza-spiro[4,5]decan) der Firma "AD-67" or "MON 4660" (3-dichloroacetyl-l-oxa-3-aza-spiro [4,5] decane) from the company
Nitrokemia bzw. Monsanto (S3-7), Nitrokemia or Monsanto (S3-7),
"TI-35" (l-Dichloracetyl-azepan) der Firma TRI-Chemical RT (S3-8), "TI-35" (l-dichloroacetyl-azepan) from TRI-Chemical RT (S3-8),
"Diclonon" (Dicyclonon) oder "BAS145138" oder "LAB145138" (S3-9) "Diclonon" (dicyclonone) or "BAS145138" or "LAB145138" (S3-9)
((RS)-l-Dichloracetyl-3,3,8a-trimethylperhydropyrrolo[l,2-a]pyrimidin-6-on) der Firma BASF, "Furilazol" oder "MON 13900" ((RS)-3-Dichloracetyl-5-(2-furyl)-2,2-dimethyloxazolidin) (S3-10), sowie dessen (R)-Isomer (S3-11). ((RS) -l-dichloroacetyl-3,3,8a-trimethylperhydropyrrolo [1,2-a] pyrimidin-6-one) from BASF, "Furilazole" or "MON 13900" ((RS) -3-dichloroacetyl- 5- (2-furyl) -2,2-dimethyloxazolidine) (S3-10), as well as its (R) -isomer (S3-11).
54) Verbindungen aus der Klasse der Acylsulfonamide (S4): 54) Compounds from the class of acylsulfonamides (S4):
S4a) N- Acylsulfonamide der Formel (S4a) und deren Salze wie sie in der WO-A-97/45016 S4 a ) N-acylsulfonamides of the formula (S4 a ) and their salts as described in WO-A-97/45016
beschrieben sind, wonn are described Wonn
RA1 (Ci-C6)Alkyl, (C3-C6)Cycloalkyl, wobei die 2 letztgenannten Reste durch VA RA 1 (Ci-C 6 ) alkyl, (C3-C6) cycloalkyl, the latter 2 residues by VA
Substituenten aus der Gruppe Halogen, (Ci-C4)Alkoxy, (Ci-C6)Haloalkoxy und (Ci- C4)Alkylthio und im Falle cyclischer Reste auch durch (Ci-C4)Alkyl und (Ci-C4)Haloalkyl substituiert sind; Substituents from the group halogen, (Ci-C4) alkoxy, (Ci-C 6) haloalkoxy and (Ci- C4) alkylthio and in case of cyclic radicals are substituted by (Ci-C4) alkyl and (Ci-C4) haloalkyl;
RA2 Halogen, (Ci-C4)Alkyl, (Ci-C4)Alkoxy, CF3; mA 1 oder 2; RA 2 halogen, (Ci-C 4 ) alkyl, (Ci-C 4 ) alkoxy, CF 3; m A 1 or 2;
VA ist 0, 1 , 2 oder 3 bedeuten; VA is 0, 1, 2 or 3;
S4b) Verbindungen vom Typ der 4-(Benzoylsulfamoyl)benzamide der Formel (S4b) und deren Salze, wie sie in der WO-A-99/16744 beschrieben sind, S4 b ) compounds of the 4- (benzoylsulfamoyl) benzamide type of the formula (S4 b ) and their salts, as described in WO-A-99/16744,
wonn Wonn
RB1, RB2 unabhängig voneinander Wasserstoff, (Ci-C6)Alkyl, (C3-C6)Cycloalkyl, (C3- C6)Alkenyl, (C3-C6)Alkinyl, RB 1 , RB 2 independently of one another hydrogen, (Ci-C 6 ) alkyl, (C3-C6) cycloalkyl, (C3-C 6 ) alkenyl, (C3-C 6 ) alkynyl,
RB3 Halogen, (Ci-C4)Alkyl, (Ci-C4)Haloalkyl oder (Ci-C4)Alkoxy und ms 1 oder 2 bedeuten, z.B. solche worin RB 3 represents halogen, (Ci-C4) alkyl, (Ci-C4) haloalkyl or (Ci-C4) alkoxy and ms 1 or 2, for example those in which
RB1 = Cyclopropyl, RB2 = Wasserstoff und (RB3) = 2-OMe ist ("Cyprosulfamide", S4-1),RB 1 = cyclopropyl, RB 2 = hydrogen and (RB 3 ) = 2-OMe ("Cyprosulfamide", S4-1),
RB1 = Cyclopropyl, RB2 = Wasserstoff und (RB3) = 5-Cl-2-OMe ist (S4-2), RB 1 = Ethyl, RB 2 = Wasserstoff und (RB 3) = 2-OMe ist(S4-3), RB 1 = cyclopropyl, RB 2 = hydrogen and (RB 3 ) = 5-Cl-2-OMe is (S4-2), R B 1 = ethyl, R B 2 = hydrogen and (R B 3 ) = 2-OMe is (S4-3),
RB 1 = Isopropyl, RB 2 = Wasserstoff und (RB 3) = 5-Cl-2-OMe ist (S4-4) und R B 1 = isopropyl, R B 2 = hydrogen and (R B 3 ) = 5-Cl-2-OMe is (S4-4) and
RB 1 = Isopropyl, RB 2 = Wasserstoff und (RB 3) = 2-OMe ist(S4-5); R B 1 = isopropyl, R B 2 = hydrogen and (R B 3 ) = 2-OMe is (S4-5);
S4C) Verbindungen aus der Klasse der Benzoylsulfamoylphenylhamstoffe der Formel (S4C), wie sie in der EP-A-365484 beschrieben sind, S4 C ) compounds from the class of the benzoylsulfamoylphenylureas of the formula (S4 C ) as described in EP-A-365484,
wonn Wonn
Rc1, Rc2 unabhängig voneinander Wasserstoff, (Ci-Cg)Alkyl, (C3-Cg)Cycloalkyl, (C3-Rc 1 , Rc 2 independently of one another hydrogen, (Ci-Cg) alkyl, (C3-Cg) cycloalkyl, (C3-
C6)Alkenyl, (C3-C6)Alkinyl, C 6 ) alkenyl, (C 3 -C 6 ) alkynyl,
Rc3 Halogen, (Ci-C i)Alkyl, (Ci-C4)Alkoxy, CF3 und mc 1 oder 2 bedeuten; beispielsweise l-[4-(N-2-Methoxybenzoylsulfamoyl)phenyl]-3-methylhamstoff, Rc 3 is halogen, (Ci-C i) alkyl, (Ci-C4) alkoxy, CF 3 and m c 1 or 2; for example 1- [4- (N-2-methoxybenzoylsulfamoyl) phenyl] -3-methylurea,
l-[4-(N-2-Methoxybenzoylsulfamoyl)phenyl]-3,3-dimethylhamstoff, l- [4- (N-2-Methoxybenzoylsulfamoyl) phenyl] -3,3-dimethylurea,
l-[4-(N-4,5-Dimethylbenzoylsulfamoyl)phenyl]-3-methylhamstoff; l- [4- (N-4,5-Dimethylbenzoylsulfamoyl) phenyl] -3-methylhamstoff;
S4d) Verbindungen vom Typ der N-Phenylsulfonylterephthalamide der Formel (S4d) und deren Salze, die z.B. bekannt sind aus CN 101838227, S4 d ) compounds of the N-phenylsulfonylterephthalamide type of the formula (S4 d ) and their salts, which are known, for example, from CN 101838227,
wonn Wonn
RD 4 Halogen, (Ci-C4)Alkyl, (Ci-C4)Alkoxy, CF3; mD 1 oder 2; RD5 Wasserstoff, (Ci-C6)Alkyl, (C3-C6)Cycloalkyl, (C2-C6)Alkenyl, (C2-C6)Alkinyl, (C5- C6)Cycloalkenyl bedeutet. R D 4 halogen, (Ci-C 4 ) alkyl, (Ci-C 4 ) alkoxy, CF 3; m D 1 or 2; RD 5 is hydrogen, (Ci-C 6 ) alkyl, (C 3 -C 6 ) cycloalkyl, (C 2 -C 6 ) alkenyl, (C 2 -C 6 ) alkynyl, (C 5 - C 6 ) cycloalkenyl.
55) Wirkstoffe aus der Klasse der Hydroxyaromaten und der aromatisch-aliphatischen 55) Active ingredients from the class of the hydroxyaromatics and the aromatic-aliphatic
Carbonsäurederivate (S5), z.B. Carboxylic acid derivatives (S5), e.g.
3,4,5-Triacetoxybenzoesäureethylester, 3,5-Dimethoxy-4-hydroxybenzoesäure, 3,5- 3,4,5-triacetoxybenzoic acid ethyl ester, 3,5-dimethoxy-4-hydroxybenzoic acid, 3,5-
Dihydroxybenzoesäure, 4-Hydroxysalicylsäure, 4-Fluorsalicyclsäure, 2-Hydroxyzimtsäure, 2,4- Dichlorzimtsäure, wie sie in der WO-A-2004/084631, WO-A-2005/015994, WO-A- 2005/016001 beschrieben sind. Dihydroxybenzoic acid, 4-hydroxysalicylic acid, 4-fluorosalicyclic acid, 2-hydroxycinnamic acid, 2,4-dichlorocinnamic acid, as described in WO-A-2004/084631, WO-A-2005/015994, WO-A-2005/016001.
56) Wirkstoffe aus der Klasse der l,2-Dihydrochinoxalin-2-one (S6), z.B. 56) Active substances from the class of 1,2-dihydroquinoxalin-2-one (S6), e.g.
1 -Methyl-3-(2-thienyl)-l ,2-dihydrochinoxalin-2-on, 1 -Methyl-3-(2-thienyl)-l ,2-dihydro- chinoxalin-2-thion, l-(2-Aminoethyl)-3-(2-thienyl)-l,2-dihydro-chinoxalin-2-on-hydrochlorid, l-(2-Methylsulfonylaminoethyl)-3-(2-thienyl)-l,2-dihydro-chinoxalin-2-on, wie sie in der WO- A-2005/112630 beschrieben sind. 1-methyl-3- (2-thienyl) -l, 2-dihydroquinoxalin-2-one, 1-methyl-3- (2-thienyl) -l, 2-dihydroquinoxalin-2-thione, l- (2nd -Aminoethyl) -3- (2-thienyl) -l, 2-dihydro-quinoxalin-2-one hydrochloride, l- (2-methylsulfonylaminoethyl) -3- (2-thienyl) -l, 2-dihydro-quinoxalin- 2-one, as described in WO-A-2005/112630.
57) Verbindungen aus der Klasse der Diphenylmethoxyessigsäurederivate (S7), z.B. 57) Compounds from the class of diphenylmethoxyacetic acid derivatives (S7), e.g.
Diphenylmethoxyessigsäuremethylester (CAS-Reg.Nr. 41858-19-9) (S7-1), Diphenylmethoxyacetic acid methyl ester (CAS Reg. No. 41858-19-9) (S7-1),
Diphenylmethoxyessigsäureethylester oder Diphenylmethoxyessigsäure wie sie in der WO-A- 98/38856 beschrieben sind. Diphenylmethoxyacetic acid ethyl ester or Diphenylmethoxyacetic acid as described in WO-A-98/38856.
58) Verbindungen der Formel (S8), wie sie in der WO-A-98/27049 beschrieben sind, 58) compounds of the formula (S8), as described in WO-A-98/27049,
worin die Symbole und Indizes folgende Bedeutungen haben: where the symbols and indices have the following meanings:
RD1 ist Halogen, (Ci-C i)Alkyl, (Ci-C i)Haloalkyl, (Ci-C i)Alkoxy, (Ci-C4)Haloalkoxy, RD 1 is halogen, (Ci-C i) alkyl, (Ci-C i) haloalkyl, (Ci-C i) alkoxy, (Ci-C4) haloalkoxy,
RD 2 ist Wasserstoff oder (Ci-C i)Alkyl, R D 2 is hydrogen or (Ci-C i) alkyl,
RD 3 ist Wasserstoff, (Ci-CsjAlkyl, (C2-C4)Alkenyl, (C2-C4)Alkinyl, oder Aryl, wobei jeder der vorgenannten C-haltigen Reste unsubstituiert oder durch einen oder mehrere, vorzugsweise bis zu drei gleiche oder verschiedene Reste aus der Gruppe, bestehend aus Halogen und Alkoxy substituiert ist; oder deren Salze, nD ist eine ganze Zahl von 0 bis 2. R D 3 is hydrogen, (Ci-Csjalkyl, (C 2 -C4) alkenyl, (C 2 -C4) alkynyl, or aryl, where each of the abovementioned C-containing radicals is unsubstituted or by one or more, preferably up to three of the same or various radicals from the group consisting of halogen and alkoxy, or their salts, n D is an integer from 0 to 2.
S9) Wirkstoffe aus der Klasse der 3-(5-Tetrazolylcarbonyl)-2-chinolone (S9), z.B. S9) active substances from the class of 3- (5-tetrazolylcarbonyl) -2-quinolones (S9), e.g.
l,2-Dihydro-4-hydroxy-l-ethyl-3-(5-tetrazolylcarbonyl)-2-chinolon (CAS-Reg.Nr.: 219479-18- 2), l,2-Dihydro-4-hydroxy-l-methyl-3-(5-tetrazolyl-carbonyl)-2-chinolon (CAS-Reg.Nr. l, 2-Dihydro-4-hydroxy-l-ethyl-3- (5-tetrazolylcarbonyl) -2-quinolone (CAS Reg.No .: 219479-18-2), l, 2-dihydro-4-hydroxy- l-methyl-3- (5-tetrazolyl-carbonyl) -2-quinolone (CAS reg. no.
95855-00-8), wie sie in der WO-A-1999/000020 beschrieben sind. 95855-00-8), as described in WO-A-1999/000020.
S10) Verbindungen der Formeln (Sl0a) oder (Sl0b), wie sie in der WO-A-2007/023719 und WO-A-2007/023764 beschrieben sind, S10) compounds of the formulas (Sl0 a ) or (Sl0 b ) as described in WO-A-2007/023719 and WO-A-2007/023764,
wonn Wonn
RE1 Halogen, (Ci-C i)Alkyl, Methoxy, Nitro, Cyano, CF3, OCF3 RE 1 halogen, (Ci-C i) alkyl, methoxy, nitro, cyano, CF3, OCF3
YE, ZE unabhängig voneinander O oder S, he eine ganze Zahl von 0 bis 4, Y E , Z E independently of one another O or S, he is an integer from 0 to 4,
RE2 (Ci-Ci6)Alkyl, (C2-C6)Alkenyl, (C3-C6)Cycloalkyl, Aryl; Benzyl, Halogenbenzyl, RE3 Wasserstoff oder (Ci-C6)Alkyl bedeuten. RE 2 (Ci-Ci 6 ) alkyl, (C2-C6) alkenyl, (C3-C6) cycloalkyl, aryl; Benzyl, halobenzyl, RE 3 is hydrogen or (Ci-C 6 ) alkyl.
Sl 1) Wirkstoffe vom Typ der Oxyimino- Verbindungen (Sl 1), die als Saatbeizmittel bekannt sind, wie z. B. Sl 1) Active substances of the oxyimino compound type (Sl 1), which are known as seed dressings, such as. B.
"Oxabetrinil" ((Z)-l,3-Dioxolan-2-ylmethoxyimino(phenyl)acetonitril) (Sl l-l), das als Saatbeiz-Safener für Hirse gegen Schäden von Metolachlor bekannt ist, "Oxabetrinil" ((Z) -l, 3-dioxolan-2-ylmethoxyimino (phenyl) acetonitrile) (Sl l-l), which is known as a seed dressing safener for millet against damage to metolachlor,
"Fluxofenim" (l-(4-Chlorphenyl)-2,2,2-trifluor-l-ethanon-0-(l,3-dioxolan-2-ylmethyl)-oxim) (Sl 1-2), das als Saatbeiz-Safener für Hirse gegen Schäden von Metolachlor bekannt ist, und "Cyometrinil" oder "CGA-43089" ((Z)-Cyanomethoxyimino(phenyl)acetonitril) (Sl l-3), das als"Fluxofenim" (1- (4-chlorophenyl) -2,2,2-trifluoro-l-ethanone-0- (1,3-dioxolan-2-ylmethyl) -oxime) (Sl 1-2), which is used as a seed dressing -Safeer for millet against damage from metolachlor is known, and "Cyometrinil" or "CGA-43089" ((Z) -cyanomethoxyimino (phenyl) acetonitrile) (Sl l-3), which as
Saatbeiz-Safener für Hirse gegen Schäden von Metolachlor bekannt ist. ) Wirkstoffe aus der Klasse der Isothiochromanone (S12), wie z.B. Methyl- [(3 -oxo-lH-2- benzothiopyran-4(3H)-yliden)methoxy]acetat (CAS-Reg.Nr. 205121-04-6) (S12-1) und verwandte Verbindungen aus WO-A-1998/13361. ) Eine oder mehrere Verbindungen aus Gruppe (S13): Seed pickle safener is known for millet against damage from metolachlor. ) Active substances from the class of isothiochromanones (S12), such as methyl- [(3-oxo-lH-2-benzothiopyran-4 (3H) -ylidene) methoxy] acetate (CAS reg. No. 205121-04-6) (S12-1) and related compounds from WO-A-1998/13361. ) One or more connections from group (S13):
"Naphthalic anhydrid" (l,8-Naphthalindicarbonsäureanhydrid) (S13-1), das als Saatbeiz-Safener für Mais gegen Schäden von Thiocarbamatherbiziden bekannt ist, "Naphthalic anhydride" (1,8-naphthalenedicarboxylic anhydride) (S13-1), which is known as a seed dressing safener for maize against damage from thiocarbamate herbicides,
"Fenclorim" (4,6-Dichlor-2-phenylpyrimidin) (S13-2), das als Safener für Pretilachlor in gesätem Reis bekannt ist, "Fenclorim" (4,6-dichloro-2-phenylpyrimidine) (S13-2), which is known as a safener for pretilachlor in sown rice,
"Flurazole" (Benzyl-2-chlor-4-trifluormethyl-l,3-thiazol-5-carboxylat) (S13-3), das als Saatbeiz-Safener für Hirse gegen Schäden von Alachlor und Metolachlor bekannt ist, "Flurazole" (benzyl-2-chloro-4-trifluoromethyl-l, 3-thiazole-5-carboxylate) (S13-3), which is known as a seed dressing safener for millet against damage to alachlor and metolachlor,
"CL 304415" (CAS-Reg.Nr. 31541-57-8) "CL 304415" (CAS reg.no.31541-57-8)
(4-Carboxy-3,4-dihydro-2H-l-benzopyran-4-essigsäure) (S13-4) der Firma American (4-Carboxy-3,4-dihydro-2H-1-benzopyran-4-acetic acid) (S13-4) from American
Cyanamid, das als Safener für Mais gegen Schäden von lmidazolinonen bekannt ist, Cyanamide, which is known as a safener for corn against damage to imidazolinones,
"MG 191" (CAS-Reg.Nr. 96420-72-3) (2-Dichlormethyl-2-methyl-l,3-dioxolan) (S13-5) der Firma Nitrokemia, das als Safener für Mais bekannt ist, "MG 191" (CAS Reg. No. 96420-72-3) (2-dichloromethyl-2-methyl-1,3-dioxolane) (S13-5) from Nitrokemia, which is known as a safener for corn,
"MG 838" (CAS-Reg.Nr. 133993-74-5) "MG 838" (CAS Reg. No. 133993-74-5)
(2-propenyl l-oxa-4-azaspiro[4.5]decan-4-carbodithioat) (S13-6) der Firma Nitrokemia "Disulfoton" (O,O-Diethyl S-2-ethylthioethyl phosphordithioat) (S13-7), (2-propenyl l-oxa-4-azaspiro [4.5] decane-4-carbodithioate) (S13-6) from Nitrokemia "Disulfoton" (O, O-Diethyl S-2-ethylthioethyl phosphorodithioate) (S13-7),
"Dietholate" (O,O-Diethyl-O-phenylphosphorothioat) (S13-8), "Dietholate" (O, O-Diethyl-O-phenylphosphorothioat) (S13-8),
"Mephenate" (4-Chlorphenyl-methylcarbamat) (S13-9). ) Wirkstoffe, die neben einer herbiziden Wirkung gegen Schadpflanzen auch Safenerwirkung an Kulturpflanzen wie Reis aufweisen, wie z. B. "Mephenate" (4-chlorophenyl methyl carbamate) (S13-9). ) Active ingredients which, in addition to a herbicidal action against harmful plants, also have safener action on crop plants such as rice, such as, for. B.
"Dimepiperate" oder "MY-93" (S- 1 -Methyl- 1 -phcnylcthyl-pipcridin- 1 -carbothioat), das als Safener für Reis gegen Schäden des Herbizids Molinate bekannt ist, "Dimepiperate" or "MY-93" (S- 1 -methyl-1-phcnylcthyl-pipcridin-1-carbothioate), which is known as a safener for rice against damage to the herbicide Molinate,
"Daimuron" oder "SK 23" (l-(l-Methyl-l-phenylethyl)-3-p-tolyl-hamstoff), das als Safener für Reis gegen Schäden des Herbizids Imazosulfuron bekannt ist, "Daimuron" or "SK 23" (l- (l-methyl-l-phenylethyl) -3-p-tolylurea), which is known as a safener for rice against damage to the herbicide imazosulfuron,
"Cumyluron" = "JC-940" (3-(2-Chlorphenylmethyl)-l-(l-methyl-l-phenyl-ethyl)hamstoff, siehe JP-A-60087254), das als Safener für Reis gegen Schäden einiger Herbizide bekannt ist, "Methoxyphenon" oder "NK 049" (3,3'-Dimethyl-4-methoxy-benzophenon), das als Safener für Reis gegen Schäden einiger Herbizide bekannt ist, "Cumyluron" = "JC-940" (3- (2-chlorophenylmethyl) -l- (l-methyl-l-phenyl-ethyl) urea, see JP-A-60087254), which acts as a safener for rice against damage to some herbicides is known "Methoxyphenone" or "NK 049"(3,3'-dimethyl-4-methoxy-benzophenone), which is known as a safener for rice against damage by some herbicides,
"CSB" (l-Brom-4-(chlormethylsulfonyl)benzol) von Kumiai, (CAS-Reg.Nr. 54091-06-4), das als Safener gegen Schäden einiger Herbizide in Reis bekannt ist. S 15) V erbindungen der F ormel (S 15) oder deren T automere, "COD" (l-bromo-4- (chloromethylsulfonyl) benzene) from Kumiai, (CAS Reg. No. 54091-06-4), which is known as a safener against damage to some herbicides in rice. S 15) compounds of the formula (S 15) or their T automere,
wie sie in der WO-A-2008/131861 und WO-A-2008/131860 beschrieben sind, worin as described in WO-A-2008/131861 and WO-A-2008/131860, in which
RH1 einen (Ci-C6)Haloalkylrest bedeutet und RH 2 Wasserstoff oder Halogen bedeutet und RH 1 represents a (Ci-C 6 ) haloalkyl radical and R H 2 represents hydrogen or halogen and
RH 3, RH 4 unabhängig voneinander Wasserstoff, (Ci-Ci6)Alkyl, (C2-Ci6)Alkenyl oder R H 3 , R H 4 independently of one another are hydrogen, (Ci-Ci 6 ) alkyl, (C2-Ci6) alkenyl or
(C2-Ci6)Alkinyl, wobei jeder der letztgenannten 3 Reste unsubstituiert oder durch einen oder mehrere Reste aus der Gruppe Halogen, Hydroxy, Cyano, (Ci-C i)Alkoxy, (Ci-C i)Haloalkoxy, (Ci-C ijAlkylthio, (Ci-C ijAlkylamino, Di[(Ci-C4)alkyl]-amino, [(Ci-C i)Alkoxy]- carbonyl, [(Ci-C tjHaloalkoxyj-carbonyl, (C3-C6)Cycloalkyl, das unsubstituiert oder substituiert ist, Phenyl, das unsubstituiert oder substituiert ist, und Heterocyclyl, das unsubstituiert oder substituiert ist, substituiert ist, oder (C3-C6)Cycloalkyl, (C4-C6)Cycloalkenyl, (C3-C6)Cycloalkyl, das an einer Seite des Rings mit einem 4 bis 6-gliedrigen gesätigten oder ungesättigten carbocyclischen Ring kondensiert ist, oder (C4-C6)Cycloalkenyl, das an einer Seite des Rings mit einem 4 bis 6-gliedrigen gesättigten oder ungesättigten carbocyclischen Ring kondensiert ist, wobei jeder der letztgenannten 4 Reste unsubstituiert oder durch einen oder mehrere Reste aus der Gruppe Halogen, Hydroxy, Cyano, (Ci-C4)Alkyl, (Ci-C4)Haloalkyl, (Ci-C4)Al oxy, (Ci-C4)Haloalkoxy, (Ci-C4)Alkylthio, (Ci-C4)Alkylamino, Di[(Ci- C4)alkyl]-amino, [(Ci-C4)Alkoxy]-carbonyl, [(Ci-C4)Haloalkoxy]-carbonyl, (C 2 -Ci 6 ) alkynyl, each of the latter 3 residues being unsubstituted or by one or more residues from the group halogen, hydroxy, cyano, (Ci-C i) alkoxy, (Ci-C i) haloalkoxy, (Ci C ij alkylthio, (Ci-C ij alkylamino, di [(Ci-C4) alkyl] amino, [(Ci-C i) alkoxy] carbonyl, [(Ci-C t j haloalkoxyj-carbonyl, (C3-C6) cycloalkyl, which is unsubstituted or substituted, phenyl which is unsubstituted or substituted and heterocyclyl which is unsubstituted or substituted, or (C3-C6) cycloalkyl, (C4-C6) cycloalkenyl, (C3-C6) cycloalkyl, which is one side of the ring is fused with a 4-6 membered saturated or unsaturated carbocyclic ring, or (C4-C6) cycloalkenyl fused to one side of the ring with a 4-6 membered saturated or unsaturated carbocyclic ring, each of the latter 4 residues unsubstituted or by one or more residues from the group halogen, hydroxy, cyano, (Ci-C4) alkyl, (Ci-C4) Hal O-alkyl, (Ci-C 4 ) Al oxy, (Ci-C 4 ) haloalkoxy, (Ci-C 4 ) alkylthio, (Ci-C 4 ) alkylamino, di [(Ci-C 4 ) alkyl] -amino, [(Ci C 4 ) alkoxy] carbonyl, [(Ci-C 4 ) haloalkoxy] carbonyl,
(C3-C6)Cycloalkyl, das unsubstituiert oder substituiert ist, Phenyl, das unsubstituiert oder substituiert ist, und Heterocyclyl, das unsubstituiert oder substituiert ist, substituiert ist, bedeutet oder (C3-C6) cycloalkyl which is unsubstituted or substituted, phenyl which is unsubstituted or substituted and heterocyclyl which is unsubstituted or substituted is substituted or
RH 3 (Ci-C4)-Alkoxy, (C2-C4)Alkenyloxy, (C2-C6)Alkinyloxy oder (C2-C4)Haloalkoxy bedeutet und RH 4 Wasserstoff oder (Ci-C4)-Alkyl bedeutet oder R H 3 is (Ci-C 4 ) alkoxy, (C2-C 4 ) alkenyloxy, (C2-C6) alkynyloxy or (C2-C 4 ) haloalkoxy and R H 4 is hydrogen or (Ci-C 4 ) alkyl or
RH 3 und RH 4 zusammen mit dem direkt gebundenen N-Atom einen vier- bis achtgliedrigen R H 3 and R H 4 together with the directly bound N atom form a four- to eight-membered group
heterocyclischen Ring, der neben dem N-Atom auch weitere Heteroringatome, vorzugsweise bis zu zwei weitere Heteroringatome aus der Gruppe N, O und S enthalten kann und der unsubstituiert oder durch einen oder mehrere Reste aus der Gruppe Halogen, Cyano, Nitro, (Ci- C4)Alkyl, (Ci-C4)Haloalkyl, (Ci-C4)Alkoxy, (Ci-C4)Haloalkoxy und (Ci-C4)Alkylthio substituiert ist, bedeutet. heterocyclic ring which, in addition to the N atom, can also contain further hetero ring atoms, preferably up to two further hetero ring atoms from the group N, O and S, and which is unsubstituted or by one or more radicals from the group halogen, cyano, nitro, (Ci C 4 ) alkyl, (Ci-C 4 ) haloalkyl, (Ci-C 4 ) alkoxy, (Ci-C 4 ) haloalkoxy and (Ci-C 4 ) alkylthio is substituted.
S16) Wirkstoffe, die vorrangig als Herbizide eingesetzt werden, jedoch auch Safenerwirkung auf Kulturpflanzen aufweisen, z. B. S16) Active ingredients which are primarily used as herbicides, but also have safener action on crops, for. B.
(2,4-Dichlorphenoxy)essigsäure (2,4-D), (2,4-dichlorophenoxy) acetic acid (2,4-D),
(4-Chlorphenoxy)essigsäure, (4-chlorophenoxy) acetic acid,
(R,S)-2-(4-Chlor-o-tolyloxy)propionsäure (Mecoprop), (R, S) -2- (4-chloro-o-tolyloxy) propionic acid (mecoprop),
4-(2,4-Dichlorphenoxy)buttersäure (2,4-DB), 4- (2,4-dichlorophenoxy) butyric acid (2,4-DB),
(4-Chlor-o-tolyloxy)essigsäure (MCPA), (4-chloro-o-tolyloxy) acetic acid (MCPA),
4-(4-Chlor-o-tolyloxy)buttersäure, 4- (4-chloro-o-tolyloxy) butyric acid,
4-(4-Chlorphenoxy)buttersäure, 4- (4-chlorophenoxy) butyric acid,
3,6-Dichlor-2-methoxybenzoesäure (Dicamba), 3,6-dichloro-2-methoxybenzoic acid (dicamba),
l-(Ethoxycarbonyl)ethyl-3,6-dichlor-2-methoxybenzoat (Lactidichlor-ethyl). 1- (Ethoxycarbonyl) ethyl 3,6-dichloro-2-methoxybenzoate (lactidichloro-ethyl).
Bevorzugte Safener in Kombination mit den erfindungsgemäßen Verbindungend der Formel (I) und/oder deren Salze, insbesondere mit den Verbindungen der Formeln (1.1) bis (1.130) und/oder deren Salze sind: Cloquintocet-mexyl, Cyprosulfamid, Fenchlorazol-ethylester, Isoxadifen-ethyl, Mefenpyr- diethyl, Fenclorim, Cumyluron, S4-1 und S4-5, und besonders bevorzugte Safener sind: Cloquintocet- mexyl, Cyprosulfamid, Isoxadifen-ethyl und Mefenpyr-diethyl. Biologische Beispiele: Preferred safeners in combination with the compounds according to the invention of the formula (I) and / or their salts, in particular with the compounds of the formulas (1.1) to (1.130) and / or their salts, are: cloquintocet-mexyl, cyprosulfamide, fenchlorazole ethyl ester, isoxadifene -ethyl, mefenpyr-diethyl, fenclorim, cumyluron, S4-1 and S4-5, and particularly preferred safeners are: cloquintocet-mexyl, cyprosulfamide, isoxadifen-ethyl and mefenpyr-diethyl. Biological examples:
A. Herbizide Wirkung im frühen Nachauflauf A. Early post-herbicidal effects
Samen von mono- bzw. dikotylen Unkrautpflanzen wurden in 96-well Mikrotiterplatten in Quarzsand ausgelegt und in der Klimakammer unter kontrollierten Wachstumsbedingungen angezogen. 5 bis 7 Tage nach der Aussaat wurden die Versuchspflanzen im Keimblattstadium behandelt. Die in Form von Emulsionskonzentraten (EC) formulierten erfindungsgemäßen Verbindungen wurden mit einer Seeds of monocotyledonous or dicotyledonous weed plants were placed in 96-well microtiter plates in quartz sand and grown in the climatic chamber under controlled growth conditions. 5 to 7 days after sowing, the test plants were treated at the cotyledon stage. The compounds according to the invention formulated in the form of emulsion concentrates (EC) were treated with a
Wasseraufwandmenge von umgerechnet 2200 Liter pro Hektar appliziert. Nach 9 bis 12 Tagen The amount of water applied is equivalent to 2200 liters per hectare. After 9 to 12 days
Standzeit der Versuchspflanzen in der Klimakammer unter optimalen Wachstumsbedingungen, wurde die Wirkung der Präparate visuell im Vergleich zu unbehandelten Kontrollen bonitiert. Beispielsweise bedeutet 100% Wirkung = Pflanzen sind abgestorben, 0% Wirkung = wie Kontrollpflanzen. If the test plants remained in the climatic chamber under optimal growth conditions, the effect of the preparations was rated visually in comparison to untreated controls. For example, 100% activity = plants have died, 0% activity = like control plants.
In den nachstehenden Tabllen Al bis A7 sind die Wirkungen ausgewählter Verbindungen der allgemeinen Formel (I) gemäß der Tabellen 1.1 bis 1.130 auf verschiedene Schadpflanzen und einer Aufwandmenge entsprechend 1900 g/ha, die gemäß zuvor genannter Versuchvorschrift erhalten wurden, dargestellt. The following tables A1 to A7 show the effects of selected compounds of the general formula (I) according to Tables 1.1 to 1.130 on various harmful plants and an application rate corresponding to 1900 g / ha, which were obtained in accordance with the aforementioned test instructions.
Tabelle Al Table Al
Tabelle A2 Table A2
Tabelle A3 Table A3
Tabelle A4 Tabelle A5 Table A4 Table A5
Tabelle A6 Table A6
Tabelle A7 Table A7
Wie die Ergebnisse zeigten, weisen erfindungsgemäße Verbindungen, wie beispielsweise die As the results showed, compounds according to the invention, such as the
Verbindungen Nr. 1.99.7, 1.1-1, 1.2-10, 1.2-3, 1.1-2, 1.1-31, 1.1-8, 1.106-8, 1.1-10, 1.2-1, 1.1-3 und andere erfindungsgemäße Verbindungen der allgemeinen Formel (I), bei Behandlung im Nachauflauf eine gute herbizide Wirksamkeit gegen Schadpflanzen auf. Compounds No. 1.99.7, 1.1-1, 1.2-10, 1.2-3, 1.1-2, 1.1-31, 1.1-8, 1.106-8, 1.1-10, 1.2-1, 1.1-3 and other compounds according to the invention the general formula (I), when treated post-emergence, has good herbicidal activity against harmful plants.
Beispielsweise haben dabei die Verbindungen Nr. 1.99.7, 1.1-1, 1.2-10, 1.2-3, 1.1-2, 1.1-31, 1.1-8, 1.106-8, 1.1-10, 1.2-1, 1.1-3 im Nachauflaufverfahren eine sehr gute herbizide Wirkung (80% bis 100% herbizide Wirkung) gegen Schadpflanzen wie Setaria viridis, Matricaria chamomilla, Stellaria media, Veronica persica, Poa annua, Agrostis capillaris und Diplotaxis tenuifolia bei einer Aufwandmenge von 1900 Aktivsubstanz pro Hektar. B. Herbizide Wirkung und Kulturverträglichkeit im Vorauflauf For example, compounds No. 1.99.7, 1.1-1, 1.2-10, 1.2-3, 1.1-2, 1.1-31, 1.1-8, 1.106-8, 1.1-10, 1.2-1, 1.1-3 in the post-emergence process a very good herbicidal activity (80% to 100% herbicidal activity) against harmful plants such as Setaria viridis, Matricaria chamomilla, Stellaria media, Veronica persica, Poa annua, Agrostis capillaris and Diplotaxis tenuifolia at an application rate of 1900 active substances per hectare. B. Pre-emergence herbicidal activity and crop tolerance
Samen von mono- bzw. dikotylen Unkraut und Kulturpflanzen wurden in Kunststoff- oder organischen Pflanztöpfen ausgelegt und mit Erde abgedeckt. Die in Form von benetzbaren Pulvern (WP) oder als Emulsionskonzentrate (EC) formulierten erfindungsgemäßen Verbindungen wurden dann als wässrige Suspension bzw. Emulsion unter Zusatz von 0,5% Additiv mit einer Wasseraufwandmenge von umgerechnet 600 1/ha auf die Oberfläche der Abdeckerde appliziert. Nach der Behandlung wurden die Töpfe im Gewächshaus aufgestellt und unter guten Wachstumsbedingungen für die Testpflanzen gehalten. Nach ca. 3 Wochen wird die Wirkung der Präparate visuell im Vergleich zu unbehandelten Kontrollen in Prozentwerten bonitiert. Beispielsweise bedeutet 100% Wirkung = Pflanzen sind abgestorben, 0% Wirkung = wie Kontrollpflanzen. Seeds of monocotyledonous or dicotyledonous weeds and crops were placed in plastic or organic plant pots and covered with soil. The compounds according to the invention formulated in the form of wettable powders (WP) or as emulsion concentrates (EC) were then applied to the surface of the covering earth as an aqueous suspension or emulsion with the addition of 0.5% additive with a water application rate of the equivalent of 600 l / ha. After the treatment, the pots were placed in the greenhouse and kept under good growth conditions for the test plants. After approx. 3 weeks, the effect of the preparations is rated visually in percentages compared to untreated controls. For example, 100% activity = plants have died, 0% activity = like control plants.
In den nachstehenden Tabllen Bl bis B5 sind die Wirkungen ausgewählter Verbindungen der allgemeinen Formel (I) gemäß der Tabellen 1.1 bis 1.130 auf verschiedene Schadpflanzen und einer Aufwandmenge entsprechend 320 g/ha oder niedriger, die gemäß zuvor genannter Versuchvorschrift erhalten wurden, dargestellt. The following tablets B1 to B5 show the effects of selected compounds of the general formula (I) according to Tables 1.1 to 1.130 on various harmful plants and an application rate corresponding to 320 g / ha or less, which were obtained in accordance with the aforementioned test instructions.
Tabelle Bl Table Bl
Tabelle B2 Table B2
Tabelle B3 Table B3
Tabelle B4 Table B4
Tabelle B5 Table B5
Wie die Ergebnisse zeigten, weisen erfindungsgemäße Verbindungen, wie beispielsweise die As the results showed, compounds according to the invention, such as the
Verbindungen 1.2-8, 1.2-1 und 1.4-8 und weitere erfindungsgemäße Verbindungender allgemeinen Formel (I) bei Behandlung im Vorauflauf eine gute herbizide Wirksamkeit gegen Schadpflanzen auf, z. B. gegen Schadpflanzen wie Abutilon theophrasti, Alopecurus myosuroides, Setaria viridis, Avena fatua, Amaranthus retroflexus, Polygonum convolvulus und Viola tricolor bei einer Aufwandmenge von 320 g Aktivsubstanz pro Hektar. Compounds 1.2-8, 1.2-1 and 1.4-8 and other compounds of the general formula (I) according to the invention have a good herbicidal activity against harmful plants, e.g. B. against harmful plants such as Abutilon theophrasti, Alopecurus myosuroides, Setaria viridis, Avena fatua, Amaranthus retroflexus, Polygonum convolvulus and Viola tricolor at a rate of 320 g of active ingredient per hectare.
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18179531.1 | 2018-06-25 | ||
| EP18179531 | 2018-06-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020002090A1 true WO2020002090A1 (en) | 2020-01-02 |
Family
ID=62778772
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2019/066198 Ceased WO2020002090A1 (en) | 2018-06-25 | 2019-06-19 | Substituted thiazolylpyrrolones, salts thereof and the use thereof as herbicidal agents |
Country Status (2)
| Country | Link |
|---|---|
| AR (1) | AR115627A1 (en) |
| WO (1) | WO2020002090A1 (en) |
Citations (53)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2247266A1 (en) | 1971-09-30 | 1973-04-05 | Ciba Geigy Ag | 2,5-DIOXO-IMIDAZOLIDINE AS SELECTIVE HERBICIDES |
| DE2735841A1 (en) | 1976-08-12 | 1978-02-16 | Ciba Geigy Ag | 3,4-DIMETHYL-2-HYDROXY-5-OXO-2,5-DIHYDRO-PYRROLE COMPOUNDS, PROCESS FOR THEIR PRODUCTION AND PEST CONTROL AGENTS |
| CH633678A5 (en) | 1977-07-20 | 1982-12-31 | Ciba Geigy Ag | Pesticide |
| EP0086750A2 (en) | 1982-02-17 | 1983-08-24 | Ciba-Geigy Ag | Use of quinoline derivatives in the protection of crop plants |
| EP0094349A2 (en) | 1982-05-07 | 1983-11-16 | Ciba-Geigy Ag | Use of quinoline derivatives for the protection of cultivated plants |
| JPS6087254A (en) | 1983-10-19 | 1985-05-16 | Japan Carlit Co Ltd:The | Novel urea compound and herbicide containing the same |
| EP0174562A2 (en) | 1984-09-11 | 1986-03-19 | Hoechst Aktiengesellschaft | Plant protecting agents based on 1,2,4 - triazole derivatives as well as 1,2,4-triazole derivatives |
| EP0191736A2 (en) | 1985-02-14 | 1986-08-20 | Ciba-Geigy Ag | Use of quinoline derivatives for the protection of crop plants |
| EP0268554A2 (en) | 1986-10-22 | 1988-05-25 | Ciba-Geigy Ag | 1,5-Diphenyl pyrazole-3-carbonic-acid derivatives for the protection of cultured plants |
| EP0269806A1 (en) | 1986-10-04 | 1988-06-08 | Hoechst Aktiengesellschaft | Phenylpyrazole carbonic acid derivatives, their preparation and use as plant growth regulators and antidotes |
| EP0286816A1 (en) | 1987-03-13 | 1988-10-19 | Nihon Tokushu Noyaku Seizo K.K. | 2,5-Dihydropyrroles |
| EP0297378A2 (en) | 1987-06-29 | 1989-01-04 | Nihon Tokushu Noyaku Seizo K.K. | 2,5-Dihydropyrroles |
| EP0333131A1 (en) | 1988-03-17 | 1989-09-20 | Hoechst Aktiengesellschaft | Plant protective agent based on pyrazolecarboxylic acid derivatives |
| EP0334133A1 (en) | 1988-03-25 | 1989-09-27 | Bayer Ag | Pyrazolyl pyrrolinones |
| EP0339390A1 (en) | 1988-04-27 | 1989-11-02 | Nihon Tokushu Noyaku Seizo K.K. | Herbicidal 2,5-dihydropyrroles |
| EP0346620A1 (en) | 1988-05-20 | 1989-12-20 | Hoechst Aktiengesellschaft | Plant protection agents containing 1,2,4-triazole derivatives, and the 1,2,4-triazole derivatives |
| EP0365484A1 (en) | 1988-10-20 | 1990-04-25 | Ciba-Geigy Ag | Sulfamoyl phenyl ureas |
| WO1991007874A1 (en) | 1989-11-30 | 1991-06-13 | Hoechst Aktiengesellschaft | Pyrazolines for the protection of crops against herbicides |
| WO1991008202A1 (en) | 1989-11-25 | 1991-06-13 | Hoechst Aktiengesellschaft | Isoxazolines, method of preparation thereof, and their use as plant-protection agents |
| EP0492366A2 (en) | 1990-12-21 | 1992-07-01 | Hoechst Schering AgrEvo GmbH | New 5-chloroquinolin-8-oxyalkanecarbonic acid derivatives, process for their preparation and their use as antidotes for herbicides |
| EP0582198A2 (en) | 1992-08-01 | 1994-02-09 | Hoechst Schering AgrEvo GmbH | Substituted (hetero-)aryle compounds, process for their preparation, those containing compositions and their use as safeners |
| WO1995007897A1 (en) | 1993-09-16 | 1995-03-23 | Hoechst Schering Agrevo Gmbh | Substituted isoxazolines, process for producing them, agents containing them and their use as safeners |
| WO1997045016A1 (en) | 1996-05-29 | 1997-12-04 | Hoechst Schering Agrevo Gmbh | Novel n-acyl sulphonamides, novel mixtures of herbicides and antidotes and their use |
| WO1998013361A1 (en) | 1996-09-26 | 1998-04-02 | Novartis Ag | Herbicidal composition |
| WO1998027049A1 (en) | 1996-12-19 | 1998-06-25 | Hoechst Schering Agrevo Gmbh | Novel 2-fluoroacrylic acid derivatives, novel mixtures of herbicides and antidotes and the use thereof |
| WO1998038856A1 (en) | 1997-03-04 | 1998-09-11 | Zeneca Limited | Compositions for safening rice against acetochlor |
| WO1999000020A1 (en) | 1997-06-27 | 1999-01-07 | Hoechst Schering Agrevo Gmbh | 3-(5-tetrazolyl carbonyl)-2-quinolones and products for protecting useful plants containing the same |
| WO1999016744A1 (en) | 1997-09-29 | 1999-04-08 | Aventis Cropscience Gmbh | Acylsulfamoyl benzoic acid amides, plant protection agents containing said acylsulfamoyl benzoic acid amides, and method for producing the same |
| WO2002034048A1 (en) | 2000-10-23 | 2002-05-02 | Syngenta Participations Ag | Agrochemical compositions with quinoline safeners |
| EP1426365A1 (en) | 2001-08-17 | 2004-06-09 | Sankyo Agro Company, Limited | 3-phenoxy-4-pyridazinol derivative and herbicide composition containing the same |
| WO2004084631A1 (en) | 2003-03-26 | 2004-10-07 | Bayer Cropscience Gmbh | Use of aromatic hydroxy compounds as safeners |
| WO2005015994A1 (en) | 2003-08-05 | 2005-02-24 | Bayer Cropscience Gmbh | Use of aromatic compounds as safeners |
| WO2005016001A1 (en) | 2003-08-05 | 2005-02-24 | Bayer Cropscience Gmbh | Safener based on aromatic-aliphatic carboxylic acid derivatives |
| WO2005112630A1 (en) | 2004-05-12 | 2005-12-01 | Bayer Cropscience Gmbh | Quinoxalin-2-one derivatives crop protection agents comprising the same and method for production and use therof |
| WO2007023719A1 (en) | 2005-08-22 | 2007-03-01 | Kumiai Chemical Industry Co., Ltd. | Agent for reducing chemical injury and herbicide composition with reduced chemical injury |
| WO2007023764A1 (en) | 2005-08-26 | 2007-03-01 | Kumiai Chemical Industry Co., Ltd. | Agent for reduction of harmful effect of herbicide and herbicide composition having reduced harmful effect |
| WO2008055840A1 (en) | 2006-11-09 | 2008-05-15 | F. Hoffmann-La Roche Ag | Thiazole and oxazole-substituted arylamides |
| WO2008131860A2 (en) | 2007-04-30 | 2008-11-06 | Bayer Cropscience Ag | Pyridonecarboxamides crop protection agents containing the same method for production and use thereof |
| WO2008131861A1 (en) | 2007-04-30 | 2008-11-06 | Bayer Cropscience Ag | Use of pyridine-2-oxy-3-carbonamides as safeners |
| US20090143448A1 (en) | 2007-11-30 | 2009-06-04 | Paul Hebeisen | New aminothiazoles as fbpase inhibitors for diabetes |
| CN101838227A (en) | 2010-04-30 | 2010-09-22 | 孙德群 | Safener of benzamide herbicide |
| US20120122067A1 (en) | 2010-11-15 | 2012-05-17 | Age Of Learning, Inc. | Online educational system with mentor guided learning |
| WO2012093809A2 (en) | 2011-01-03 | 2012-07-12 | Hanmi Pharm Co., Ltd. | New bicyclic compound for modulating g protein-coupled receptors |
| WO2014131843A1 (en) | 2013-02-28 | 2014-09-04 | Syngenta Participations Ag | Plant growth regulating compounds |
| WO2015018434A1 (en) | 2013-08-05 | 2015-02-12 | Syngenta Participations Ag | Pyrazolyl pyrrolinones and their use as herbicides |
| WO2015018431A1 (en) | 2013-08-05 | 2015-02-12 | Syngenta Participations Ag | Pyrazolyl pyrrolinones and their use as herbicides |
| WO2015128321A1 (en) | 2014-02-26 | 2015-09-03 | Syngenta Participations Ag | Plant growth regulating compounds |
| WO2016071363A1 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071361A2 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071359A1 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071362A2 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071360A1 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal pyrrolone derivatives |
| WO2016071364A2 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
-
2019
- 2019-06-19 WO PCT/EP2019/066198 patent/WO2020002090A1/en not_active Ceased
- 2019-06-25 AR ARP190101767A patent/AR115627A1/en unknown
Patent Citations (53)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2247266A1 (en) | 1971-09-30 | 1973-04-05 | Ciba Geigy Ag | 2,5-DIOXO-IMIDAZOLIDINE AS SELECTIVE HERBICIDES |
| DE2735841A1 (en) | 1976-08-12 | 1978-02-16 | Ciba Geigy Ag | 3,4-DIMETHYL-2-HYDROXY-5-OXO-2,5-DIHYDRO-PYRROLE COMPOUNDS, PROCESS FOR THEIR PRODUCTION AND PEST CONTROL AGENTS |
| CH633678A5 (en) | 1977-07-20 | 1982-12-31 | Ciba Geigy Ag | Pesticide |
| EP0086750A2 (en) | 1982-02-17 | 1983-08-24 | Ciba-Geigy Ag | Use of quinoline derivatives in the protection of crop plants |
| EP0094349A2 (en) | 1982-05-07 | 1983-11-16 | Ciba-Geigy Ag | Use of quinoline derivatives for the protection of cultivated plants |
| JPS6087254A (en) | 1983-10-19 | 1985-05-16 | Japan Carlit Co Ltd:The | Novel urea compound and herbicide containing the same |
| EP0174562A2 (en) | 1984-09-11 | 1986-03-19 | Hoechst Aktiengesellschaft | Plant protecting agents based on 1,2,4 - triazole derivatives as well as 1,2,4-triazole derivatives |
| EP0191736A2 (en) | 1985-02-14 | 1986-08-20 | Ciba-Geigy Ag | Use of quinoline derivatives for the protection of crop plants |
| EP0269806A1 (en) | 1986-10-04 | 1988-06-08 | Hoechst Aktiengesellschaft | Phenylpyrazole carbonic acid derivatives, their preparation and use as plant growth regulators and antidotes |
| EP0268554A2 (en) | 1986-10-22 | 1988-05-25 | Ciba-Geigy Ag | 1,5-Diphenyl pyrazole-3-carbonic-acid derivatives for the protection of cultured plants |
| EP0286816A1 (en) | 1987-03-13 | 1988-10-19 | Nihon Tokushu Noyaku Seizo K.K. | 2,5-Dihydropyrroles |
| EP0297378A2 (en) | 1987-06-29 | 1989-01-04 | Nihon Tokushu Noyaku Seizo K.K. | 2,5-Dihydropyrroles |
| EP0333131A1 (en) | 1988-03-17 | 1989-09-20 | Hoechst Aktiengesellschaft | Plant protective agent based on pyrazolecarboxylic acid derivatives |
| EP0334133A1 (en) | 1988-03-25 | 1989-09-27 | Bayer Ag | Pyrazolyl pyrrolinones |
| EP0339390A1 (en) | 1988-04-27 | 1989-11-02 | Nihon Tokushu Noyaku Seizo K.K. | Herbicidal 2,5-dihydropyrroles |
| EP0346620A1 (en) | 1988-05-20 | 1989-12-20 | Hoechst Aktiengesellschaft | Plant protection agents containing 1,2,4-triazole derivatives, and the 1,2,4-triazole derivatives |
| EP0365484A1 (en) | 1988-10-20 | 1990-04-25 | Ciba-Geigy Ag | Sulfamoyl phenyl ureas |
| WO1991008202A1 (en) | 1989-11-25 | 1991-06-13 | Hoechst Aktiengesellschaft | Isoxazolines, method of preparation thereof, and their use as plant-protection agents |
| WO1991007874A1 (en) | 1989-11-30 | 1991-06-13 | Hoechst Aktiengesellschaft | Pyrazolines for the protection of crops against herbicides |
| EP0492366A2 (en) | 1990-12-21 | 1992-07-01 | Hoechst Schering AgrEvo GmbH | New 5-chloroquinolin-8-oxyalkanecarbonic acid derivatives, process for their preparation and their use as antidotes for herbicides |
| EP0582198A2 (en) | 1992-08-01 | 1994-02-09 | Hoechst Schering AgrEvo GmbH | Substituted (hetero-)aryle compounds, process for their preparation, those containing compositions and their use as safeners |
| WO1995007897A1 (en) | 1993-09-16 | 1995-03-23 | Hoechst Schering Agrevo Gmbh | Substituted isoxazolines, process for producing them, agents containing them and their use as safeners |
| WO1997045016A1 (en) | 1996-05-29 | 1997-12-04 | Hoechst Schering Agrevo Gmbh | Novel n-acyl sulphonamides, novel mixtures of herbicides and antidotes and their use |
| WO1998013361A1 (en) | 1996-09-26 | 1998-04-02 | Novartis Ag | Herbicidal composition |
| WO1998027049A1 (en) | 1996-12-19 | 1998-06-25 | Hoechst Schering Agrevo Gmbh | Novel 2-fluoroacrylic acid derivatives, novel mixtures of herbicides and antidotes and the use thereof |
| WO1998038856A1 (en) | 1997-03-04 | 1998-09-11 | Zeneca Limited | Compositions for safening rice against acetochlor |
| WO1999000020A1 (en) | 1997-06-27 | 1999-01-07 | Hoechst Schering Agrevo Gmbh | 3-(5-tetrazolyl carbonyl)-2-quinolones and products for protecting useful plants containing the same |
| WO1999016744A1 (en) | 1997-09-29 | 1999-04-08 | Aventis Cropscience Gmbh | Acylsulfamoyl benzoic acid amides, plant protection agents containing said acylsulfamoyl benzoic acid amides, and method for producing the same |
| WO2002034048A1 (en) | 2000-10-23 | 2002-05-02 | Syngenta Participations Ag | Agrochemical compositions with quinoline safeners |
| EP1426365A1 (en) | 2001-08-17 | 2004-06-09 | Sankyo Agro Company, Limited | 3-phenoxy-4-pyridazinol derivative and herbicide composition containing the same |
| WO2004084631A1 (en) | 2003-03-26 | 2004-10-07 | Bayer Cropscience Gmbh | Use of aromatic hydroxy compounds as safeners |
| WO2005015994A1 (en) | 2003-08-05 | 2005-02-24 | Bayer Cropscience Gmbh | Use of aromatic compounds as safeners |
| WO2005016001A1 (en) | 2003-08-05 | 2005-02-24 | Bayer Cropscience Gmbh | Safener based on aromatic-aliphatic carboxylic acid derivatives |
| WO2005112630A1 (en) | 2004-05-12 | 2005-12-01 | Bayer Cropscience Gmbh | Quinoxalin-2-one derivatives crop protection agents comprising the same and method for production and use therof |
| WO2007023719A1 (en) | 2005-08-22 | 2007-03-01 | Kumiai Chemical Industry Co., Ltd. | Agent for reducing chemical injury and herbicide composition with reduced chemical injury |
| WO2007023764A1 (en) | 2005-08-26 | 2007-03-01 | Kumiai Chemical Industry Co., Ltd. | Agent for reduction of harmful effect of herbicide and herbicide composition having reduced harmful effect |
| WO2008055840A1 (en) | 2006-11-09 | 2008-05-15 | F. Hoffmann-La Roche Ag | Thiazole and oxazole-substituted arylamides |
| WO2008131860A2 (en) | 2007-04-30 | 2008-11-06 | Bayer Cropscience Ag | Pyridonecarboxamides crop protection agents containing the same method for production and use thereof |
| WO2008131861A1 (en) | 2007-04-30 | 2008-11-06 | Bayer Cropscience Ag | Use of pyridine-2-oxy-3-carbonamides as safeners |
| US20090143448A1 (en) | 2007-11-30 | 2009-06-04 | Paul Hebeisen | New aminothiazoles as fbpase inhibitors for diabetes |
| CN101838227A (en) | 2010-04-30 | 2010-09-22 | 孙德群 | Safener of benzamide herbicide |
| US20120122067A1 (en) | 2010-11-15 | 2012-05-17 | Age Of Learning, Inc. | Online educational system with mentor guided learning |
| WO2012093809A2 (en) | 2011-01-03 | 2012-07-12 | Hanmi Pharm Co., Ltd. | New bicyclic compound for modulating g protein-coupled receptors |
| WO2014131843A1 (en) | 2013-02-28 | 2014-09-04 | Syngenta Participations Ag | Plant growth regulating compounds |
| WO2015018434A1 (en) | 2013-08-05 | 2015-02-12 | Syngenta Participations Ag | Pyrazolyl pyrrolinones and their use as herbicides |
| WO2015018431A1 (en) | 2013-08-05 | 2015-02-12 | Syngenta Participations Ag | Pyrazolyl pyrrolinones and their use as herbicides |
| WO2015128321A1 (en) | 2014-02-26 | 2015-09-03 | Syngenta Participations Ag | Plant growth regulating compounds |
| WO2016071363A1 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071361A2 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071359A1 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071362A2 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
| WO2016071360A1 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal pyrrolone derivatives |
| WO2016071364A2 (en) | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Herbicidal compounds |
Non-Patent Citations (22)
| Title |
|---|
| "Heterocyclic Chemistry", 2000, BLACKWELL PUBLISHING, pages: 418 - 420 |
| "Name Reactions in Heterocyclic Chemistry", 2005, WILEY, pages: 275283 |
| "Perry's Chemical Engineer's Handbook", 1973, MCGRAW-HILL, pages: 8 - 57 |
| "The Chemistry of Heterocycles", 2003, pages: 149 - 155 |
| BIOORG. & MEDCHEM. LET., vol. 14, 2004, pages 5521 - 5525 |
| BRAUN ET AL., EMBO J., vol. 11, 1992, pages 3219 - 3227 |
| CALDWELL N.J.H.V. OLPHEN: "Handbook of Insecticide Dust Diluents and Carriers", 1963, J. WILEY & SONS |
| CESKA A SLOVENSKA FARMACIE, vol. 56, 2007, pages 27 |
| CHEMICAL ABSTRACTS, Columbus, Ohio, US; abstract no. 133993-74-5 |
| GRUPPE DER STOFFE: "The Pesticide Manual", 2012, THE BRITISH CROP PROTECTION COUNCIL UND THE ROYAL SOC. OF CHEMISTRY |
| J. CHEM. SOC., PERKIN TRANS., vol. 1, 1982, pages 215 |
| J. ORG. CHEM., vol. 63, 1998, pages 2646 |
| J.D. FREYERS.A. EVANS: "Weed Control Handbook", 1968, BLACKWELL SCIENTIFIC PUBLICATIONS, pages: 101 - 103 |
| J.E. BROWNING: "Agglomeration", 1967, CHEMICAL AND ENGINEERING, pages: 147 |
| KLINGMAN: "Weed Control as a Science", 1961, JOHN WILEY AND SONS, INC., pages: 81 - 96 |
| NEOPLASMA, vol. 47, 2000, pages 313 |
| RIDGEWOOD N.J.SISLEYWOOD: "Encyclopedia of Surface Active Agents", 1964, CHEM. PUBL. CO. INC. |
| RUSSIAN J. ORG. CHEM., vol. 43, 2007, pages 801 |
| SCHÖNFELDT: "Grenzflächenaktive Äthylenoxidaddukte", 1976, WISS. VERLAGSGESELLSCHAFT |
| TETRAHEDRON, vol. 68, 2012, pages 5863 |
| WEED RESEARCH, vol. 26, 1986, pages 441 - 445 |
| ZUR HERSTELLUNG VON TELLERFLIESSBETTEXTRUDERSPRÜHGRANULATEN: "Spray-Drying Handbook", 1979, G. GOODWIN LTD. |
Also Published As
| Publication number | Publication date |
|---|---|
| AR115627A1 (en) | 2021-02-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2020002089A1 (en) | Substituted 2-heteroaryloxypyridines and salts thereof and their use as herbicidal agents | |
| EP3728235A1 (en) | Substituted thiophenyl uracils, salts thereof and the use thereof as herbicidal agents | |
| EP3558976A1 (en) | Substituted azolylpyrrolones and azolylhydantoines and salts thereof and use thereof as herbicidal active substances | |
| EP4200282A1 (en) | Substituted n-phenyluracils and salts thereof, and use thereof as herbicidal active substances | |
| WO2023161172A1 (en) | Substituted n-benzoic acid uracils and salts thereof, and use thereof as herbicidal active substances | |
| WO2020193474A1 (en) | Substituted 2-heteroarylaminobenzenes and the salts thereof and their use as herbicidal agents | |
| WO2019121544A1 (en) | Substituted thiophenyl uracils, salts thereof and the use thereof as herbicidal agents | |
| WO2019025156A1 (en) | SUBSTITUTED PYRROLIDINONE AND THEIR SALTS AND THEIR USE AS HERBICIDAL ACTIVE SUBSTANCES | |
| EP3655393A1 (en) | Substituted 5-(het-)arylpyrazolamides and salts thereof and their use as herbicidal active substances | |
| WO2018184890A1 (en) | 4-substituted n-amino and n-hydroxy 2-oxo-pyrrolidie-3-carboxamide derivatives, salts thereof and use thereof as herbicidal agents | |
| WO2019121547A1 (en) | Substituted thiophenyl uracils, salts thereof and the use thereof as herbicidal agents | |
| EP4097087B1 (en) | [(1,4,5-trisubstituted-1h-pyrazol-3-yl)sulfanyl]acetic acid derivatives, their salts and use of said compounds as herbicidal agents | |
| WO2019081477A1 (en) | SUBSTITUTED PYRAZOLE AND THEIR SALTS AND THEIR USE AS HERBICIDAL ACTIVE SUBSTANCES | |
| WO2018114596A1 (en) | Substituted heteroaryl pyrrolones and salts thereof and use thereof as herbicidal active substances | |
| WO2018178008A1 (en) | Substituted isoxazoline-5-ones and salts thereof and use thereof as herbicidal agents | |
| WO2018177837A1 (en) | 4-cyclopentyl- and 4-cyclopropyl-2-oxopyrrolidine-3-carboxamide derivatives and related compounds as herbicidal plant protection agents | |
| WO2018178010A1 (en) | N-cyclopropyl-2-oxo-4-phenyl-piperidine-3-carboxamide derivatives and related compounds as herbicidal plant protection agents | |
| EP3558986A1 (en) | Substituted 1,2,4-thiadiazolyl pyrrolones and 1,2,4-thiadiazolyl hydantoines and salts thereof and use thereof as herbicides | |
| EP4230621A1 (en) | Substituted n-benzoic acid uracils, their salts and use of said compounds as herbicidal agents | |
| EP4230620A1 (en) | Substituted n-amino-n-benzoic acid uracils, their salts and use of said compounds as herbicidal agents | |
| WO2020002087A1 (en) | Substituted 3-heteroaryloxypyridines and salts thereof and their use as herbicidal agents | |
| WO2020064260A1 (en) | Substituted 5-(sulfanyl)-3,4-dihydro-2h-pyrrol-4-carboxamides and salts thereof and their use as herbicidal active substances | |
| WO2020002085A1 (en) | Substituted 4-heteroaryloxypyridines and salts thereof and their use as herbicidal agents | |
| WO2019179928A1 (en) | Substituted succinimide-3-carboxamides, salts thereof and the use thereof as herbicidal agents | |
| EP3728214A1 (en) | Substituted n-heterocyclyl- and n-heteroaryl-tetrahydropyrimidinones and the salts thereof, and the use of same as herbicidal active substances |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19734314 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19734314 Country of ref document: EP Kind code of ref document: A1 |