WO2019230207A1 - Method for producing oil-in-water type emulsion composition - Google Patents
Method for producing oil-in-water type emulsion composition Download PDFInfo
- Publication number
- WO2019230207A1 WO2019230207A1 PCT/JP2019/015691 JP2019015691W WO2019230207A1 WO 2019230207 A1 WO2019230207 A1 WO 2019230207A1 JP 2019015691 W JP2019015691 W JP 2019015691W WO 2019230207 A1 WO2019230207 A1 WO 2019230207A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- oil
- emulsion
- fatty acid
- water
- emulsified
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/192—Carboxylic acids, e.g. valproic acid having aromatic groups, e.g. sulindac, 2-aryl-propionic acids, ethacrynic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
- A61K31/222—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin with compounds having aromatic groups, e.g. dipivefrine, ibopamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/14—Esters of carboxylic acids, e.g. fatty acid monoglycerides, medium-chain triglycerides, parabens or PEG fatty acid esters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/24—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing atoms other than carbon, hydrogen, oxygen, halogen, nitrogen or sulfur, e.g. cyclomethicone or phospholipids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/04—Drugs for skeletal disorders for non-specific disorders of the connective tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the present disclosure relates to a method for producing an oil-in-water emulsion composition.
- fat emulsions containing medicinal ingredients, phospholipids, triglycerides, and surfactants are known as pharmaceuticals.
- the fat emulsion is administered to the patient by direct injection, for example, in the medical field, or mixed with an infusion solution such as physiological saline and administered to the patient by instillation.
- Chinese Patent Application No. 1070656575 includes flurbiprofen axetil, phospholipid, triglyceride, and surfactant, and a solution composition that spontaneously emulsifies upon mixing with an infusion such as physiological saline. Things are disclosed.
- an oil-in-water emulsion composition containing flurbiprofen axetil is placed in a container and heat-treated, for example, the emulsion particles in the emulsion composition aggregate to produce coarse particles on the order of ⁇ m.
- the emulsion particles in the emulsion composition aggregate to produce coarse particles on the order of ⁇ m.
- a problem may occur in that blood vessels are blocked after administration. Therefore, it is desirable that there are few coarse particles.
- a problem to be solved by an embodiment of the present invention is an oil-in-water emulsion composition comprising at least one compound of flurbiprofen axetil and flurbiprofen, which is produced through a heat treatment step.
- the present invention also provides a method for producing an oil-in-water emulsion composition with few coarse particles.
- Means for solving the above problems include the following aspects. ⁇ 1> With respect to an emulsified product containing phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen, and water, the pressure is 20 MPa or more and 350 MPa or less.
- Step A to obtain an emulsion by applying an emulsification treatment A to-be-heated product containing at least the emulsion obtained in step A, comprising at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester, and sorbitan fatty acid
- the step of subjecting the object to be heated in which the total content of the ester and the polyoxyethylene sorbitan fatty acid ester is 0.6% by mass or more and 1.8% by mass or less based on the total mass of the object to be heated.
- B and The manufacturing method of the oil-in-water emulsion composition which has NO.
- ⁇ 2> The process for producing an oil-in-water emulsion composition according to ⁇ 1>, wherein in step B, the heat-treated object is subjected to heat treatment at a temperature of 90 ° C. or higher and 140 ° C. or lower.
- ⁇ 3> The oil-in-water emulsification according to ⁇ 1> or ⁇ 2>, wherein at least one surfactant selected from the group consisting of sorbitan fatty acid esters and polyoxyethylene sorbitan fatty acid esters is a polyoxyethylene sorbitan fatty acid ester.
- a method for producing the composition is a method for producing the composition.
- step A selected from the group consisting of phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen, sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester Any one of ⁇ 1> to ⁇ 3>, wherein an emulsified product is obtained by subjecting an emulsified product containing at least one surfactant and water to an emulsified treatment at a pressure of 20 MPa to 350 MPa.
- the manufacturing method of the oil-in-water type emulsion composition as described in one.
- Step A includes phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen, and water, and sorbitan fatty acid ester and polyoxyethylene. Any one of ⁇ 1> to ⁇ 3> to obtain an emulsion by performing an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less on an article to be emulsified containing no surfactant selected from sorbitan fatty acid esters The manufacturing method of the oil-in-water type emulsion composition of description.
- step A an oil-in-water mold according to any one of ⁇ 1> to ⁇ 5>, wherein an emulsion is obtained by performing an emulsification treatment with a pressure of 80 MPa or more and 350 MPa or less on the article to be emulsified
- a method for producing an emulsified composition ⁇ 7> The method for producing an oil-in-water emulsion composition according to any one of ⁇ 1> to ⁇ 6>, wherein the pH of the object to be heated in step B is 6.0 or more and 10.0 or less.
- ⁇ 8> The method for producing an oil-in-water emulsion composition according to any one of ⁇ 1> to ⁇ 7>, wherein the pH of the object to be heated in step B is 7.0 or more and 9.0 or less.
- ⁇ 9> The method for producing an oil-in-water emulsified composition according to any one of ⁇ 1> to ⁇ 8>, in which the product to be emulsified contains a polyhydric alcohol.
- an oil-in-water emulsion composition comprising at least one compound of flurbiprofen axetil and flurbiprofen, even when manufactured through a heat treatment step, A method for producing an oil-in-water emulsion composition with few coarse particles is provided.
- a numerical range indicated by using “to” means a range including the numerical values described before and after “to” as the minimum value and the maximum value, respectively.
- an upper limit value or a lower limit value described in a numerical range may be replaced with an upper limit value or a lower limit value in another numerical range.
- the upper limit value or the lower limit value described in a certain numerical range may be replaced with the values shown in the examples.
- a combination of two or more preferred embodiments is a more preferred embodiment.
- the amount of each component means the total amount of a plurality of types of substances unless there is a specific case when there are a plurality of types of substances corresponding to each component.
- the “small amount of coarse particles” oil-in-water emulsion composition means that the oil-in-water emulsion composition has a diameter of 5 ⁇ m to 50 ⁇ m with respect to the total volume of the emulsion particles contained in the oil-in-water emulsion composition. It means an oil-in-water emulsion composition in which the volume ratio of particles is 0.05% by volume or less.
- “the volume ratio (unit: volume%) of particles having a diameter of 5 ⁇ m to 50 ⁇ m in the oil-in-water emulsion composition to the total volume of the emulsion particles contained in the oil-in-water emulsion composition” is expressed as “PFAT5 Sometimes referred to as “value”.
- the PFAT5 value is preferably 0.03% by volume or less, more preferably 0.01% by volume or less, and particularly preferably no particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less, that is, 0% by volume. .
- the “PFAT5 value” is a value obtained by the following method.
- the oil-in-water emulsion composition is diluted 10 times with water to prepare a sample solution for evaluation. 1 mL of the prepared sample solution for evaluation is injected into a number counting particle size distribution device, and the total volume of particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less in 1 mL of the sample solution for evaluation is automatically calculated by the function of the device.
- the volume ratio of particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid that is, PFAT5 value) (unit) : Volume%).
- the total volume of the emulsified particles contained in the evaluation sample liquid is approximately the same as the volume of the triglyceride contained in the evaluation sample liquid.
- the volume of the triglyceride contained in the sample liquid for evaluation is calculated based on the following formula (2).
- the number-counting particle size distribution device for example, AccuSizer 780AS (product name) manufactured by Nihon Integris Co., Ltd. can be preferably used.
- the number counting type particle size distribution apparatus is not limited to this.
- PFAT5 value [total volume of particles having a diameter of 5 ⁇ m to 50 ⁇ m in 1 mL of sample liquid for evaluation] ⁇ [volume of triglyceride in 1 mL of sample liquid for evaluation] ⁇ 100 (1)
- process is not limited to an independent process, and is included in the term if the intended purpose of the process is achieved even if it cannot be clearly distinguished from other processes.
- the method for producing an oil-in-water emulsion composition of the present disclosure (hereinafter, also simply referred to as “manufacturing method”) is at least one selected from phospholipids, triglycerides, flurbiprofen axetil and flurbiprofen.
- Step A and Step of obtaining an emulsion by subjecting an emulsified product containing the above compound (hereinafter also referred to as “specific compound”) and water to an emulsified treatment with a pressure of 20 MPa to 350 MPa.
- the total content of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester is To be heated product is 0.6 wt% to 1.8 wt% or less based on the total weight of the heat treatment product, and a step B of performing heat treatment.
- the manufacturing method of this indication may have other processes as needed.
- an emulsified composition after the emulsified composition is contained in a container, a heat treatment for sterilization is performed.
- the oil-in-water emulsion composition containing at least one of flurbiprofen axetil and flurbiprofen in a container and heat-treated, the oil-in-water emulsion composition is coarse. It has been found that particles may form. When there are many coarse particles in the oil-in-water emulsion composition, for example, when the oil-in-water emulsion composition is administered by intravenous injection, it may lead to occlusion of blood vessels.
- the volume ratio of coarse particles is the sum of the emulsion particles contained in the oil-in-water emulsion composition. It becomes 0.05 volume% or less with respect to the volume, and there are few coarse particles.
- the object to be heated to be subjected to the heat treatment includes an emulsion obtained through an emulsification treatment in which a high pressure of 20 MPa or more is applied. Therefore, the emulsified particles contained in the object to be heated are It is fine and stable. In addition, it is considered that the generation of coarse particles generated during the heat treatment is caused by the fact that the emulsified particles existing in the vicinity of the gas-liquid interface in the container adhere to the inner surface of the container.
- the to-be-heated material to be subjected to the heat treatment contains the specific surfactant with a content in a specific range, the emulsion particles are prevented from adhering to the inner surface of the container during the heat treatment.
- the emulsified particles in the heat-treated product to be subjected to the heat treatment are fine and stable, and are difficult to adhere to the inner surface of the container, so that there are few coarse particles in water. It is presumed that an oil-type emulsion composition can be produced.
- an oil-in-water emulsion composition is added to a fat emulsion containing flurbiprofen axetil and lightly stirred. It is not obtained by the emulsification treatment in which a high pressure of 20 MPa or more is applied.
- the fat emulsion described in Chinese Patent Application No. 1070656575 contains flurbiprofen axetil, but the oil-in-water emulsion composition containing flurbiprofen axetil is contained in a container and heat-treated. In addition, no attention is paid to the problem that coarse particles are generated.
- Chinese Patent Application No. 1070656575 does not describe a technique for suppressing the generation of coarse particles that may occur during the heat treatment of an oil-in-water emulsion composition.
- the object to be heated to be subjected to the heat treatment includes an emulsion obtained through a high-pressure emulsification treatment having a pressure of 20 MPa or more, and thus is included in the object to be heated.
- the emulsified particles are stable.
- the to-be-heated material used for a heat processing contains the specific surfactant by the content rate of a specific range, and the emulsified particle contained in a to-be-heated material becomes still more stable.
- the specific compound included in the emulsified particles is less likely to come into contact with water. Therefore, the oil-in-water emulsion composition obtained by the production method of the present disclosure is a decomposition product of the specific compound generated by heat treatment. It is estimated that the stability of the specific compound during storage is excellent.
- the oil-in-water emulsion composition obtained by the production method of the present disclosure has few fatty acids generated by the decomposition of phospholipids and triglycerides, and is excellent in stability of phospholipids and triglycerides during storage.
- the heat-treated product to be subjected to the heat treatment is obtained through a high-pressure emulsification treatment with a pressure of 20 MPa or more. Therefore, the emulsified particles contained in the heat-treated material are: It will be stable.
- the to-be-heated material used for a heat processing contains the specific surfactant by the content rate of a specific range, and the emulsified particle contained in a to-be-heated material becomes still more stable.
- the phospholipids and triglycerides that form the emulsified particles are also stabilized. Therefore, the oil-in-water emulsion composition obtained by the production method of the present disclosure is derived from the phospholipids and triglycerides generated by the heat treatment. It is estimated that fatty acids, that is, fatty acids produced by the degradation of phospholipids and triglycerides are small, and that the stability of phospholipids and triglycerides during storage is excellent.
- Step A is for an emulsified product containing phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen (that is, a specific compound), and water.
- This is a step of obtaining an emulsion by performing an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less.
- the emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less in Step A means an emulsification treatment in which a pressure of 20 MPa or more and 350 MPa or less is added to the product to be emulsified.
- step A an oil-in-water emulsion in which emulsified particles containing the specific compound and triglyceride are dispersed in water can be obtained.
- the emulsified particles in the emulsion obtained in step A are fine and stable.
- the emulsified processed product containing phospholipid, triglyceride, specific compound and water may be a mixture containing phospholipid, triglyceride, specific compound and water, and this mixture is a crude emulsion obtained by emulsifying the mixture. It may be. That is, step A may include step A-1 for preparing a mixture containing phospholipid, triglyceride, specific compound, and water, and the mixture prepared in step A-1 is subjected to an emulsification treatment. Step A-2 may be provided to give a crude emulsion.
- a method for preparing a mixture containing phospholipid, triglyceride, specific compound, and water is not particularly limited.
- Each component contained in a mixture should just be mixed, and it is preferable that it is mixed uniformly.
- Each component contained in the mixture may be mixed at one time, or may be mixed while adding another component dividedly into one component.
- the method for mixing each component contained in the mixture is not particularly limited, and examples thereof include a method of mixing by stirring.
- limiting in particular as a stirring means A common stirring instrument or stirring apparatus can be used.
- the stirring time is not particularly limited, and can be appropriately set according to the type of stirring tool or stirring device, the composition (ie, type and amount) of the components to be stirred, and the like.
- the temperature at the time of mixing each component contained in a mixture is not restrict
- a general heating apparatus can be used.
- the method for the emulsification treatment in step A-2 is not particularly limited.
- limiting in particular as a means of the emulsification process for obtaining a rough emulsion For example, the means using a general emulsification apparatus is mentioned.
- the emulsifier include a homogenizer, a homomixer, and an ultrasonic homogenizer.
- the emulsification time is not particularly limited, and can be appropriately set according to the type of the emulsifying device, the composition of the mixture, and the like.
- the rotation speed can be set to 1,000 rpm (revolutions per minute; hereinafter the same) to 100,000 rpm, and the emulsification time can be set to 1 to 30 minutes.
- the frequency can be set to 15 kHz to 40 kHz, and the energy density of the dispersed portion can be set to 100 W / cm 2 or more.
- the processed product to be emulsified may contain components other than phospholipids, triglycerides, specific compounds, and water as necessary.
- the processed product to be emulsified may contain, for example, at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester (that is, a specific surfactant).
- the to-be-emulsified processed material may contain the other additive (for example, polyhydric alcohol) mentioned later.
- step A an emulsified product is subjected to an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less with respect to an emulsified treatment containing phospholipid, triglyceride, specific compound, specific surfactant, and water. It may be obtained, and emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less is performed on an article to be emulsified containing phospholipid, triglyceride, specific compound, and water and not containing a specific surfactant. From the above, an emulsion may be obtained.
- Step A if an emulsified treatment containing a phospholipid, a triglyceride, a specific compound, a specific surfactant, and water is subjected to an emulsification treatment at a pressure of 20 MPa to 350 MPa, it is specified. There is a tendency to obtain an oil-in-water emulsion composition in which the decomposition product of the compound is less.
- Step A when an emulsified treatment containing a phospholipid, a triglyceride, a specific compound, and water and containing no specific surfactant is subjected to an emulsification treatment at a pressure of 20 MPa to 350 MPa.
- the ratio of the phospholipid content to the triglyceride content in the processed product to be emulsified is not particularly limited.
- the oil-in-water type is better. From the standpoint of obtaining an emulsion to be retained in the film, it is preferably 0.03 or more and 2.0 or less, more preferably 0.06 or more and 1.0 or less, and more preferably 0.12 or more and 0.5 on a mass basis. More preferably, it is as follows.
- the content of triglyceride in the processed product is preferably 2% by mass or more, and preferably 5% by mass with respect to the total mass of the processed product. The above is more preferable, and 10 mass% or more is still more preferable.
- the content of triglyceride in the product to be emulsified is preferably 40% by mass or less, and preferably 30% by mass with respect to the total mass of the product to be emulsified from the viewpoint of obtaining an emulsion containing finer emulsified particles. % Or less is more preferable, and 20% by mass or less is still more preferable.
- the content of the specific compound in the processed product to be emulsified is, for example, from 0.01% by mass to 2.0% by mass with respect to the total mass of the processed product from the viewpoint of the dose of the specific compound per one time. The following is preferred.
- the content of water in the emulsion to be processed is preferably 30% by mass or more, more preferably, based on the total mass of the emulsion to be processed. It is 40 mass% or more, More preferably, it is 50 mass% or more.
- the content rate of water in the to-be-emulsified processed material is, for example, from the same viewpoint as described above, preferably 99% by mass or less, more preferably 98% by mass or less, with respect to the total mass of the emulsified processed product. More preferably, it is 97 mass% or less.
- the content of the specific surfactant in the to-be-emulsified processed product is preferably 0.6% by mass or more and 2.25 with respect to the total mass of the to-be-emulsified processed product. % By mass or less, more preferably 0.8% by mass or more and 2.2% by mass or less, still more preferably 1.0% by mass or more and 1.9% by mass or less, and particularly preferably 1.25% by mass. The content is 1.9% by mass or less.
- the content of the specific surfactant in the emulsion to be emulsified is within the above range, an oil-in-water emulsion composition with few coarse particles can be obtained even after the heat treatment step (ie, step B). Tend.
- the content of the polyhydric alcohol in the product to be emulsified is, for example, from the viewpoint of obtaining an emulsion containing finer emulsified particles, to the total mass of the product to be emulsified.
- 0.5 mass% or more is preferable, 1.0 mass% or more is more preferable, and 2.0 mass% or more is still more preferable.
- the content of the polyhydric alcohol in the emulsified treatment is, for example, from the viewpoint of further reducing the possibility of tissue damage due to osmotic pressure difference when administered by injection, with respect to the total mass of the emulsified treatment, 10 mass% or less is preferable, 5 mass% or less is more preferable, and 3 mass% or less is still more preferable.
- step A there is no particular limitation on the means for performing an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less on the product to be emulsified.
- a high-pressure homogenizer is suitable. Dispersion by a high-pressure homogenizer is dispersion due to a large shear force generated when a liquid passes through a very narrow gap at high speed.
- the high-pressure homogenizer include a chamber-type high-pressure homogenizer having a chamber for fixing the flow path of the processing liquid and a homogeneous valve-type high-pressure homogenizer having a homogeneous valve.
- the chamber type high-pressure homogenizer examples include microfluidizer [Paurec Co., Ltd.], nanomizer [Nanomizer Co., Ltd.], and optimizer [Sugino Machine Co., Ltd.].
- the homogeneous valve type high-pressure homogenizer includes Gorin type homogenizer [SMT Co., Ltd.], Lanier type homogenizer [SMT Co., Ltd.], high-pressure homogenizer (Niro Soabi), homogenizer [Sanwa Machinery Co., Ltd.], high-pressure homogenizer [ Izumi Food Machinery Co., Ltd.], ultra-high pressure homogenizer (IKA Co., Ltd.) and the like.
- step A an emulsification treatment is performed on the object to be emulsified with a pressure of 20 MPa to 350 MPa, preferably 80 MPa to 350 MPa.
- a pressure of 20 MPa to 350 MPa preferably 80 MPa to 350 MPa.
- an oil-in-water emulsion composition with few coarse particles is obtained even after the heat treatment step (that is, step B).
- the upper limit value of 350 MPa is a limit value of a commercially available apparatus.
- the lower limit of the pressure is preferably 40 MPa or more, more preferably 60 MPa or more, further preferably 75 MPa or more, and particularly preferably 80 MPa or more.
- the upper limit of the pressure is preferably 245 MPa or less.
- the pressure may be 40 MPa or more and 245 MPa or less, 60 MPa or more and 245 MPa or less, 75 MPa or more and 245 MPa or less, or 80 MPa or more and 245 MPa or less.
- the number of times of emulsification treatment may be one, but from the viewpoint of more uniformly emulsifying and dispersing, it is preferably performed twice or more, more preferably 5 to 30 times.
- the temperature during the emulsification treatment is not particularly limited and can be set, for example, in the range of 5 ° C to 80 ° C.
- Phospholipid The product to be emulsified contains phospholipid. Phospholipids mainly function as emulsifiers. Examples of the phospholipid include lecithin, which is a phospholipid derived from a natural product. Lecithin is phosphatidylcholine itself or a mixture containing at least phosphatidylcholine.
- a mixture containing at least phosphatidylcholine generally includes, in addition to phosphatidylcholine, phosphatidylserine, phosphatidylethanolamine, phosphatidylinositol, N-acylphosphatidylethanolamine, phosphatidylglycerol, phosphatidic acid, lysophosphatidylcholine, lysophosphatidic acid, sphingomyelin, It is a mixture that may contain sphingoethanolamine and the like.
- the lecithin in the present disclosure is preferably lecithin containing phosphatidylcholine and having a phosphatidylcholine content of 80% by mass or more.
- lecithin examples include egg yolk lecithin, soybean lecithin, cottonseed lecithin, rapeseed lecithin, corn lecithin, hydrogenated egg yolk lecithin, hydrogenated soybean lecithin and the like.
- yolk lecithin means lecithin derived from egg yolk
- sey lecithin means lecithin derived from soybean
- cotton seed lecithin means lecithin derived from cotton seed.
- Rapeseed lecithin means rapeseed-derived lecithin
- corn lecithin means corn-derived lecithin.
- the phospholipid is not limited to a phospholipid derived from a natural product, and may be a chemically synthesized phospholipid.
- chemically synthesized phospholipids include phosphatidylcholines (eg, dipalmitoylphosphatidylcholine, dimyristoylphosphatidylcholine, distearoylphosphatidylcholine, and dioleoylphosphatidylcholine), phosphatidylglycerols (eg, dipalmitoylphosphatidylglycerol, dimyristoylphosphatidylglycerol, distearoyl Phosphatidylglycerol, and dioleoylphosphatidylglycerol), phosphatidylethanolamine (for example, dipalmitoyl phosphatidylethanolamine, dimyristoyl phosphatidylethanolamine, distearoyl phosphatidylethanolamine, and diole
- lecithin is preferable, at least one selected from egg yolk lecithin and soybean lecithin is more preferable, and egg yolk lecithin is still more preferable.
- egg yolk lecithin purified egg yolk lecithin obtained by purifying egg yolk lecithin or highly purified egg yolk lecithin is preferable.
- a commercially available product can be used as the phospholipid.
- Examples of commercially available phospholipids include egg yolk lecithin PL-100M [trade name; purified egg yolk lecithin, Kewpie Co., Ltd.], egg yolk lecithin PC-98N [trade name; highly purified egg yolk lecithin, Kewpie Corporation], egg yolk Examples include lecithin Lipoid E80 [trade name; purified egg yolk lecithin, H. Holstein Co., Ltd.].
- the processed product to be emulsified may contain only one type of phospholipid or two or more types.
- Triglyceride The emulsified product contains triglyceride.
- Triglycerides are acylglycerols in which three molecules of fatty acid are ester-bonded to one molecule of glycerin.
- the triglyceride is not particularly limited, and may be a medium chain fatty acid triglyceride or a long chain fatty acid triglyceride.
- the triglyceride is preferably a long-chain fatty acid triglyceride.
- the “medium chain fatty acid triglyceride” means a triglyceride having an average number of carbon atoms in the fatty acid chain of 6 or more and 12 or less.
- the average carbon number of the fatty acid chain is the number of carbon atoms of fatty acids (ie, constituent fatty acids) constituting triglycerides [for example, 8 if caprylic acid (IUPAC system name: octanoic acid), capric acid (IUPAC system name: decanoic acid) Is 10 and lauric acid (IUPAC system name: dodecanoic acid is 12)] is weighted average by the composition ratio of the constituent fatty acids.
- the constituent fatty acid may be a saturated fatty acid or an unsaturated fatty acid, and is preferably a saturated fatty acid.
- the medium-chain fatty acid triglyceride may be derived from a natural product or a synthetic fatty acid triglyceride.
- the “long chain fatty acid triglyceride” means a triglyceride having an average fatty acid chain carbon number of more than 12.
- a long-chain fatty acid triglyceride having an average fatty acid chain carbon number of 14 to 24 is preferable.
- the fatty acid constituting the long-chain fatty acid triglyceride may be a saturated fatty acid or an unsaturated fatty acid.
- the long-chain fatty acid triglyceride may be a vegetable oil corresponding to a natural long-chain fatty acid triglyceride, an animal oil corresponding to a natural long-chain fatty acid triglyceride, or a synthetic fatty acid triglyceride.
- vegetable oil is more preferable as the long-chain fatty acid triglyceride from the viewpoint that the melting point is low and the emulsion stability of the oil-in-water emulsion composition is high.
- Vegetable oils as long-chain fatty acid triglycerides are oils derived from plant seeds or nuts.
- soybean oil examples include soybean oil, cottonseed oil, rapeseed oil, sesame oil, safflower oil, corn oil, peanut oil, olive oil, coconut oil, perilla Oil, castor oil, rose oil and the like.
- vegetable oil for example, at least one selected from the group consisting of soybean oil, sesame oil, and olive oil is preferable from the viewpoint of use results for injection applications, and soybean oil is more preferable from the viewpoint of availability.
- Animal oils as long chain fatty acid triglycerides are fats and oils derived from animals or fish, and specific examples include beef tallow, pork tallow, whale oil, fish oil and the like.
- a commercially available product can be used as the triglyceride.
- Examples of commercially available products of medium-chain fatty acid triglycerides include “Miglyol (registered trademark) 812” (component name: tri (caprylic acid / capric acid) glyceryl) and “Miglyol (registered trademark) 810” (component name: “Coconard (registered trademark) RK” (component name: glyceryl tricaprylate), Kao Co., Ltd., “Coconard (registered trademark) MT” (component name: tri (caprylic acid), such as tri (caprylic acid / capric acid) glyceryl) / Glyceryl caprate), “Coconard (registered trademark) MT-N” (component name: tri (caprylic acid / capric acid) glyceryl), “Coconard (registered trademark) ML” (component name: tri (caprylic acid /
- Examples of commercial products of long-chain fatty acid triglycerides include Kaneda Corporation's “Japanese Pharmacopoeia Soybean Oil (trade name)”, “Japanese Pharmacopoeia Sesame Oil (trade name)”, “Japanese Pharmacopoeia Castor Oil (trade name)” ", Japanese Pharmacopoeia Refined Soybean Oil (trade name)", “Japanese Pharmacopoeia Refined Olive Oil (trade name)”, “Japanese Pharmacopoeia Olive Oil (trade name)”, “Japanese Pharmacopoeia Rapeseed Oil (trade name)””Super Refined Soybean (trade name)", Super Refined Live (trade name) ",” Super Refined Same (trade name) "of Croda,” Olive Oil “(trade name) of Sigma-Aldrich, etc. Is mentioned.
- the processed product to be emulsified may contain only one type of triglyceride or two or more types.
- the product to be emulsified contains at least one compound selected from flurbiprofen axetil and flurbiprofen (that is, a specific compound).
- Flurbiprofen axetil is a non-steroidal analgesic agent that has an analgesic action against post-surgical pain and cancer pain, and is marketed by API Chem under the product name “Flrubiprofen axetil”. Flurbiprofen is for osteoarthritis, shoulder periarthritis, tendinitis, tendonitis, peritonitis, humerus condylaritis (eg tennis elbow), muscle pain, and post-traumatic swelling and pain It is a non-steroidal analgesic / anti-inflammatory agent having analgesic action and anti-inflammatory action, and is marketed by Tokyo Chemical Industry Co., Ltd. under the trade name “Flurbiprofen”.
- the emulsified product may contain only one of flurbiprofen axetil or flurbiprofen, or may contain both flurbiprofen axetil and flurbiprofen.
- the emulsified product contains water.
- Water functions as a dispersion medium.
- Water is not particularly limited as long as it can be used for pharmaceuticals. Examples of water include purified water, sterilized purified water, and water for injection.
- the product to be emulsified may contain at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester (that is, a specific surfactant).
- the specific surfactant is preferably, for example, a polyoxyethylene sorbitan fatty acid ester from the viewpoint of stabilizing the emulsified particles. If the product to be emulsified contains polyoxyethylene sorbitan fatty acid ester, it is considered that a form in which the polyoxyethylene chain is exposed is formed on at least a part of the surface of the emulsified particles. It is considered that the presence of this polyoxyethylene chain causes steric repulsion between adjacent emulsified particles and stabilizes the emulsified particles.
- polyoxyethylene sorbitan fatty acid ester there is no restriction
- the polyoxyethylene sorbitan fatty acid ester may be a compound having one ester bond in one molecule (that is, a mono-fatty acid ester), or a compound having two or more ester bonds in one molecule (that is, a di-fatty acid ester). Fatty acid ester, trifatty acid ester, etc.), or a mixture of two or more compounds having different numbers of ester bonds in one molecule.
- the average number of moles of oxyethylene groups added in the polyoxyethylene sorbitan fatty acid ester is not particularly limited, but is preferably 5 to 60, for example, from the viewpoint of obtaining an oil-in-water emulsion composition with fewer coarse particles. 40 is more preferable, and 15 to 25 is even more preferable.
- the carbon number of the fatty acid in the polyoxyethylene sorbitan fatty acid ester is not particularly limited, and is preferably 8 or more, and more preferably 12 or more.
- polyoxyethylene sorbitan fatty acid esters include polysorbate 20 (also known as polyoxyethylene sorbitan monolaurate), polysorbate 40 (also known as polyoxyethylene sorbitan palmitate), polysorbate 60 (also known as polyoxymonostearate). And polysorbates such as polysorbate 65 (also known as polyoxyethylene sorbitan tristearate) and polysorbate 80 (also known as polyoxyethylene sorbitan oleate).
- polyoxyethylene sorbitan fatty acid esters include, for example, polysorbate 80 (also known as polyoxyethylene sorbitan oleate) and polysorbate 20 (also known as polyoxyethylene sorbitan monolaurate) from the viewpoint of actual use in injection applications. ) Is preferred.
- Polysorbate is a polyoxyethylene ether of sorbitan fatty acid ester obtained by condensing about 20 molecules of ethylene oxide to sorbitan fatty acid ester.
- a commercially available product can be used as the polyoxyethylene sorbitan fatty acid ester.
- Examples of commercially available products of polyoxyethylene sorbitan fatty acid ester include “NIKKOL (registered trademark) TO-10MV” of Nikko Chemicals Co., Ltd., which is polysorbate 80 (also known as polyoxyethylene sorbitan oleate), NOF Corporation ) “Polysorbate 80”, “Polysorbate 80 GS”, “Polysorbate 80 HX2”, “Nonion OT-221”, “MONTANOX (registered trademark) 80” of SEPPIC, “Reodol (registered trademark) TW of Kao Corporation -O120V "," Solgen (registered trademark) TW-80V “of Daiichi Kogyo Seiyaku Co., Ltd.,” Krillet 4HP "of Croda Japan Co., Ltd., polysorbate 20 (also known as polyoxyethylene sorbitan monolaurate), “NIKKOL (registere
- sorbitan fatty acid ester There is no restriction
- sorbitan fatty acid esters include sorbitan sesquioleate, sorbitan monocaprylate, sorbitan monolaurate, sorbitan monostearate, sorbitan sesquistearate, sorbitan tristearate, sorbitan isostearate, sorbitan sesquiisostearate, sorbitan oleate And sorbitan trioleate.
- a commercial item can be used as sorbitan fatty acid ester.
- sorbitan fatty acid esters include “NIKKOL (registered trademark) SO-15MV” (component name: sorbitan sesquioleate) from Nikko Chemicals Co., Ltd., “Leodol (registered trademark) AO” from Kao Corporation.
- Sorbitan Sesquioleate Sorbitan Sesquioleate
- Sorgen registered trademark 30V
- Ionet registered trademark
- S ” component name: sorbitan fatty acid ester
- Cadenax registered trademark
- SO-80C Cadenax (registered trademark) SO-80C
- the emulsified product may contain only one type of specific surfactant or two or more types.
- the emulsified product preferably contains a polyhydric alcohol.
- the processing object contains a polyhydric alcohol, the stability of the emulsified particles tends to be improved.
- a polyhydric alcohol There is no restriction
- the polyhydric alcohol include glycerin, concentrated glycerin, propylene glycol, 1,3-butylene glycol, polyethylene glycol [eg, polyethylene glycol 300 (so-called macrogol 300), polyethylene glycol 400 (so-called macrogol 400), Polyethylene glycol 600 (so-called macrogol 600) and polyethylene glycol 1000 (so-called macrogol 1000)], dipropylene glycol, diethylene glycol and the like.
- concentrated glycerin is preferable from the viewpoint of obtaining finer emulsified particles.
- the emulsified processed product contains a polyhydric alcohol, it may contain only one kind of polyhydric alcohol or two or more kinds.
- the processed product to be emulsified may further contain other additives as necessary within the range not impairing the effects of the production method of the present disclosure, in addition to the components described above.
- other additives include pharmaceutically acceptable additives.
- an additive suitable for intravenous injection when the oil-in-water emulsion composition obtained by the production method of the present disclosure is administered by intravenous injection, it is preferable to further use an additive suitable for intravenous injection.
- antioxidants eg, ascorbic acid, D- ⁇ -tocopherol, butylhydroxyanisole (BHA), and dibutylhydroxytoluene (BHT)
- stabilizers eg, sodium citrate
- preservatives for example, methyl paraoxybenzoate and propyl paraoxybenzoate.
- antioxidants eg, ascorbic acid, D- ⁇ -tocopherol, butylhydroxyanisole (BHA), and dibutylhydroxytoluene (BHT)
- preservatives for example, methyl paraoxybenzoate and propyl paraoxybenzoate.
- other additives are not limited to these.
- the emulsified product may contain only one type of other additive or two or more types.
- Step B is a heat-treated product containing at least the emulsion obtained in Step A, and is at least one surfactant selected from the group consisting of sorbitan fatty acid esters and polyoxyethylene sorbitan fatty acid esters (ie, Specific surfactant), and the total content of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester is 0.6% by mass or more and 1.8% by mass or less with respect to the total mass of the object to be heated.
- This is a step of performing heat treatment on the object to be heated.
- the process B is a so-called sterilization process by heating.
- the to-be-heated product in Step B may be the emulsion obtained in Step A using a specific surfactant, and the emulsion obtained in Step A without using the specific surfactant, It may be an emulsion obtained by mixing with a specific surfactant.
- the content of the specific surfactant in the object to be heated is 0.6% by mass or more and 1.8% by mass or less, preferably 0.8% by mass or more and 1% by mass with respect to the total mass of the object to be heated. It is 0.75 mass% or less, More preferably, it is 1.0 mass% or more and 1.5 mass% or less, More preferably, it is 1.25 mass% or more and 1.5 mass% or less. There exists a tendency for the oil-in-water-type emulsion composition with few coarse particles to be obtained as the content rate of the specific surfactant in a to-be-heated material is in said range.
- step B there is no particular limitation on the method for performing the heat treatment on the object to be heated.
- an autoclave is suitable as the heating means.
- Step B it is preferable to heat-treat the object to be heated at a temperature of 90 ° C. or higher and 140 ° C. or lower, more preferably 100 ° C. or higher and 135 ° C. or lower, and further preferably 105 ° C. or higher and 130 ° C. or lower.
- the temperature of the heat treatment is 90 ° C. or higher, the object to be heated can be sterilized more effectively.
- the temperature of the heat treatment is 140 ° C. or less, decomposition of the components contained in the heat-treated object due to heat is less likely to occur.
- “Temperature” here means the ambient temperature.
- the heating time is not particularly limited and can be appropriately set according to the heating temperature and the like.
- the pH of the object to be heated in step B is preferably 6.0 or more and 10.0 or less, more preferably 7.0 or more and 10.0 or less, and even more preferably 7.0 or more and 9.0 or less.
- the pH of the material to be heated in Step B is 6.0 or more, the emulsion stability of the oil-in-water emulsion composition is improved, and the generation of coarse particles after the heat treatment tends to be suppressed.
- disassembly of a phospholipid and a triglyceride to be suppressed.
- generation of the hydrolyzate of the specific compound after heat processing to be suppressed as pH of the to-be-heated material in process B is 10.0 or less.
- the step B is a step of adjusting the pH of the object to be heated to 6.0 or more and 10.0 or less (hereinafter referred to as “pH adjustment”). It is also preferable to include a “step”.
- step B includes a pH adjustment step, in the pH adjustment step, it is more preferable to adjust the pH of the object to be heated to 7.0 or higher and 10.0 or lower, and to 7.0 or higher and 9.0 or lower. Is more preferable.
- the pH of the object to be heated can be adjusted using, for example, a pH adjuster.
- a pH adjuster Well-known pH adjusters, such as a sodium hydroxide solution and hydrochloric acid, can be used.
- the pH of the object to be heated is measured by setting the temperature of the object to be heated to 25 ° C.
- a general method can be used as a pH measurement method.
- the pH of the object to be heated can be measured with a pH meter.
- a pH meter (model number: F-73) manufactured by HORIBA, Ltd. can be preferably used.
- the pH meter is not limited to this.
- the heat treatment is performed on the object to be heated contained in the container.
- the container for storing the object to be heated is preferably a container used for a distribution product. Prior to storing the object to be heated in the container, the object to be heated may be filtered using a filter.
- the pore size of the filter is not particularly limited, and is preferably 50 ⁇ m or less, more preferably 20 ⁇ m or less, still more preferably 10 ⁇ m or less, particularly preferably 5 ⁇ m or less, and most preferably 0.8 ⁇ m or less.
- the container containing the object to be heated is preferably sealed.
- the accommodation of the object to be heated and the sealing of the container containing the object to be heated are preferably performed in an inert atmosphere such as a nitrogen atmosphere.
- the type of the container that accommodates the object to be heated is not particularly limited, and examples include ampules, vials, syringes, and bags.
- a vial is preferable from the viewpoint of handling at medical sites, and an ampoule is preferable from the viewpoint of high sealing performance of the container.
- a syringe or a bag is preferable.
- the material of the container is not particularly limited, and may be glass or resin.
- the container is preferably a glass container from the viewpoint of oxygen permeability of the container.
- an oil-in-water emulsion composition containing phospholipid, triglyceride, specific compound, specific surfactant, and water can be obtained. Since the oil-in-water emulsion composition obtained by the production method of the present disclosure has few coarse particles, it is suitable, for example, for injection (particularly for intravenous injection). When a large amount of coarse particles are present in the oil-in-water emulsion composition, when administered by intravenous injection, blood vessels may be blocked after administration. On the other hand, since the oil-in-water emulsion composition obtained by the production method of the present disclosure has few coarse particles, the blood vessel is less likely to be blocked due to the coarse particles.
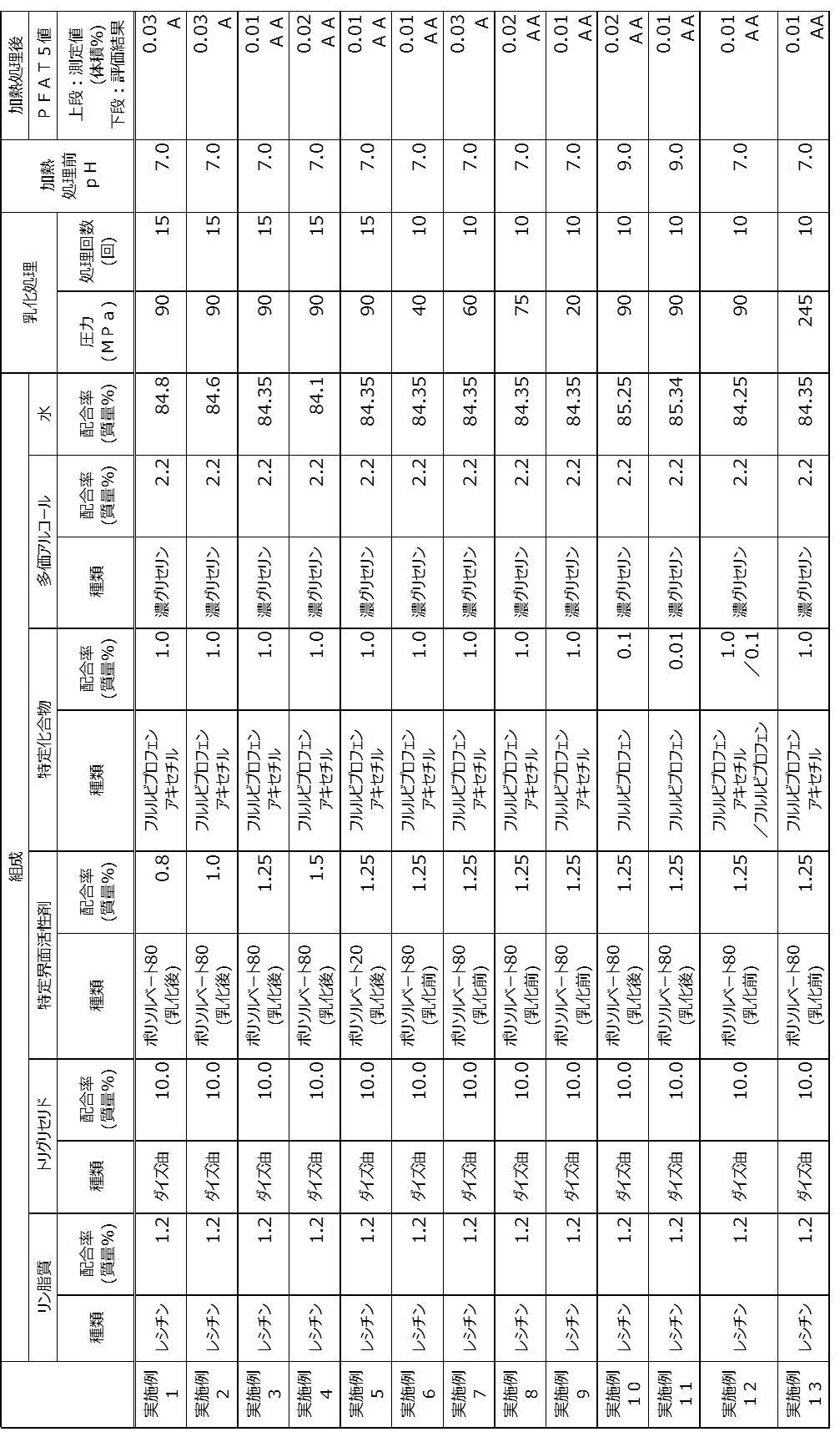
- ⁇ Phospholipid> ⁇ Purified egg yolk lecithin [Brand name: Egg yolk lecithin PL-100M, Kewpie Co., Ltd., indicated as “lecithin” in the table] ⁇ Triglyceride> -Long-chain fatty acid triglycerides- ⁇ Soybean oil [Brand name: Japanese Pharmacopoeia Soybean oil, Kaneda Corporation] ⁇ Specific surfactant> -Polyoxyethylene sorbitan fatty acid ester- Polysorbate 80 [trade name: NIKKOL (registered trademark) TO-10MV, also known as: polyoxyethylene sorbitan oleate, Nikko Chemicals Co., Ltd.] Polysorbate 20 [trade name: NIKKOL (registered trademark) TL-10, also known as: polyoxyethylene sorbitan monolaurate, Nikko Chemicals Co., Ltd.] -Sorbitan fatty acid ester- ⁇ Sorbitan sesquiole
- Flurbiprofen axetil (trade name: Flurbiprofen axtil, diastereomeric mixture, API Chem) Flurbiprofen [trade name: Flurbiprofen, Tokyo Chemical Industry Co., Ltd.]
- Example 11 [Production of oil-in-water emulsion composition] ⁇ Examples 1 to 5, Example 10, and Example 11> Among the components listed in Table 1, components other than the specific surfactant were stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, an emulsion X was obtained by subjecting the obtained crude emulsion to an emulsification treatment by the number of treatments described in Table 1 at a pressure described in Table 1 using a high-pressure homogenizer.
- a homogenizer product name: ULTRA-TURRAX (registered trademark), IKA Corporation
- an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 1.
- the pH of the emulsion Y is adjusted as shown in Table 1 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted.
- the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 ⁇ m.
- Example 12 and Example 13 Each component described in Table 1 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments shown in Table 1 at a pressure shown in Table 1 using a high-pressure homogenizer to obtain an emulsion.
- a homogenizer product name: ULTRA-TURRAX (registered trademark), IKA Corporation
- the pH of the emulsion was adjusted as described in Table 1 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. .
- the pH-adjusted emulsion was filtered using a filter having a pore size of 0.8 ⁇ m.
- 2 mL of the emulsion after filtration was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal were attached. Used to seal the container.
- the emulsion contained in the container is subjected to a heat treatment at 121 ° C.
- Table 1 shows the blending ratio (unit: mass%) of each component used in the preparation of the oil-in-water emulsion composition.
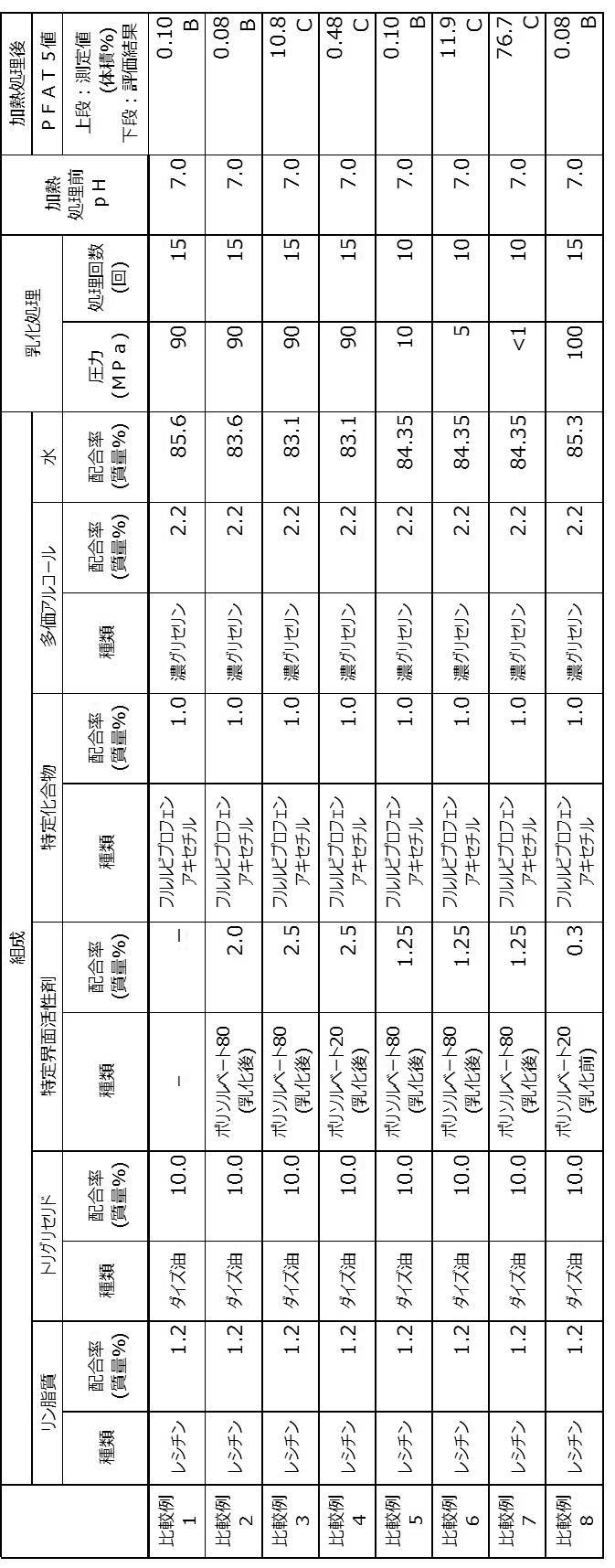
- the pH of the emulsion was adjusted as shown in Table 2 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. .
- the pH-adjusted emulsion was filtered using a filter having a pore size of 0.8 ⁇ m.
- 2 mL of the emulsion after filtration was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal were attached. Used to seal the container.
- the emulsion contained in the container is subjected to a heat treatment at 121 ° C.
- an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 2.
- the pH of the emulsion Y is adjusted as shown in Table 2 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted.
- the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 ⁇ m.
- the pH of the emulsion was adjusted as shown in Table 2 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. .
- a sodium hydroxide solution and hydrochloric acid which are pH adjusting agents
- water water
- 2 mL of the emulsion after pH adjustment was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container.
- the emulsion contained in the container is subjected to a heat treatment at 121 ° C.
- the volume ratio of particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid (that is, PFAT5 value) (unit) : Volume%) was calculated. The calculated value was rounded off to the third decimal place.
- PFAT5 value unit
- volume% the volume ratio of particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid.
- PFAT5 value unit
- Tables 1 and 2 show measured values and evaluation results of PFAT5 values. If the evaluation result was “AA” or “A”, it was judged as acceptable.
- PFAT5 value [total volume of particles with a diameter of 5 ⁇ m to 50 ⁇ m in 1 mL of sample liquid for evaluation (unit: mL)] ⁇ [volume of triglyceride in 1 mL of sample liquid for evaluation (unit: mL) ] X 100 ... (1)
- the PFAT5 value was calculated as follows. [Total volume of particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less in 1 mL of the sample solution for evaluation (unit: mL)] was 3.361 ⁇ 10 ⁇ 6 mL.
- PFAT5 value is less than 0.03% by volume.
- Tables 1 and 2 “-” means that the corresponding component is not blended.
- the descriptions “before emulsification” and “after emulsification” in the column of the type of specific surfactant indicate the timing of blending the specific surfactant, and “before emulsification” It means that an emulsification treatment has been performed on the product to be emulsified with an activator, and “after emulsification” means that a specific surfactant is blended after the emulsification treatment.
- the water described in Table 1 and Table 2 includes water derived from sodium hydroxide solution and hydrochloric acid which are pH adjusters.
- the oil-in-water emulsion compositions obtained by the production methods of Examples 1 to 13 are oil-in-water emulsions obtained by the production methods of Comparative Examples 1 to 8. It became clear that there were few coarse particles compared with the composition. From these results, it was clarified that the production methods of Examples 1 to 13 are production methods capable of producing an oil-in-water emulsion composition with few coarse particles even after the heat treatment step. .
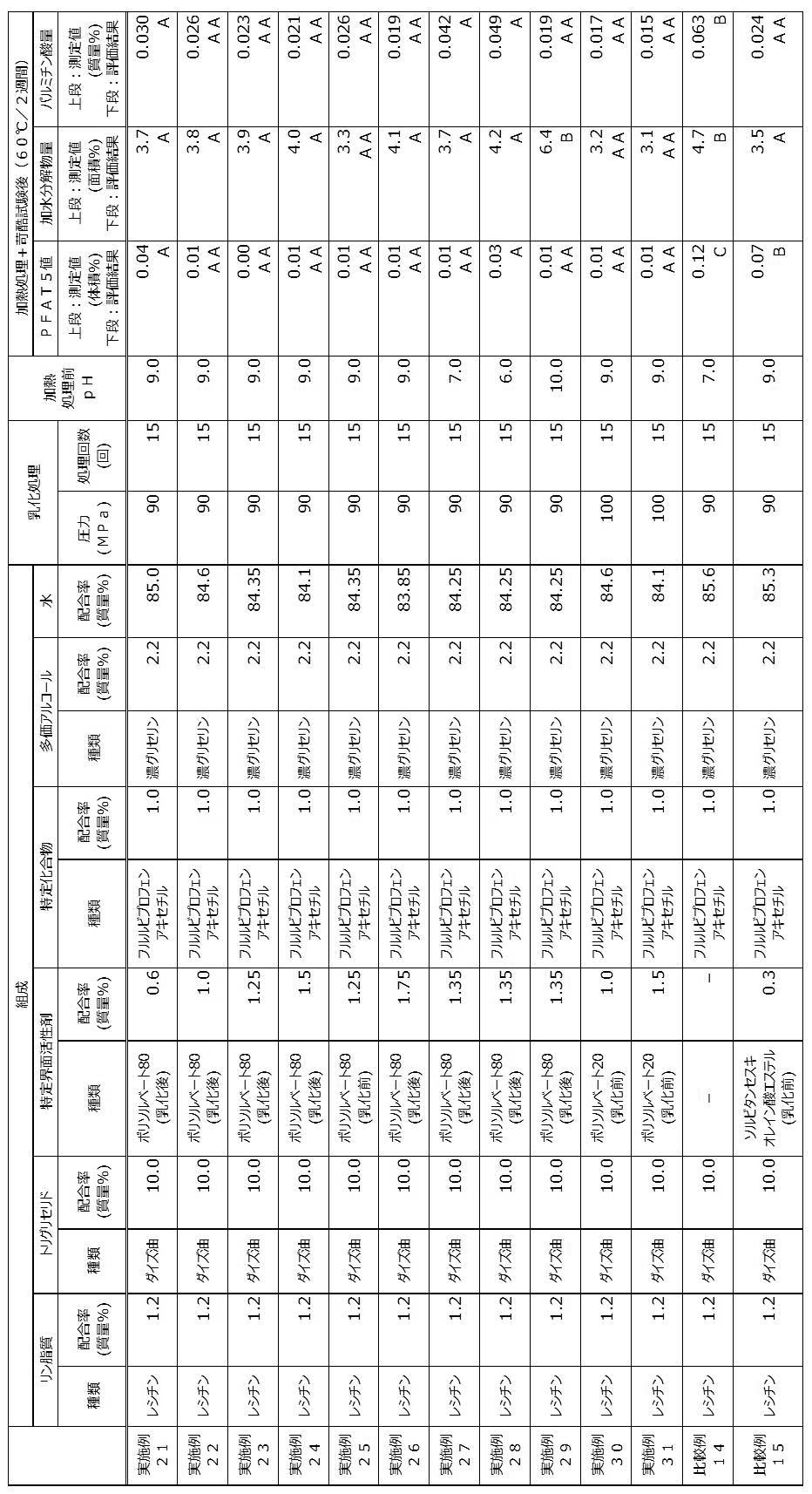
- the pH of the emulsion was adjusted as shown in Table 3 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. .
- the emulsion after pH adjustment was filtered using a filter having a pore diameter of 0.8 ⁇ m as necessary. In Example 14, filtration was performed, and in Examples 15 to 19, filtration was not performed.
- 2 mL of the emulsion after pH adjustment (for Example 14, which was filtered after pH adjustment) was placed in a glass container [trade name: CS-2, type of container] under a nitrogen atmosphere.
- Example 20 Among the components listed in Table 3, components other than the specific surfactant were stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, an emulsion X was obtained by subjecting the obtained crude emulsion to an emulsification treatment by the number of treatments described in Table 3 at a pressure described in Table 3 using a high-pressure homogenizer.
- a homogenizer product name: ULTRA-TURRAX (registered trademark), IKA Corporation
- an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 3.
- the pH of the emulsion Y is adjusted as shown in Table 3 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted.
- the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 ⁇ m.
- the pH of the emulsion was adjusted as shown in Table 3 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Subsequently, the emulsion after pH adjustment was filtered using a filter having a pore diameter of 0.8 ⁇ m as necessary. In Comparative Example 9, Comparative Example 10 and Comparative Example 12, filtration was performed, and in Comparative Example 11 and Comparative Example 13, filtration was not performed.
- a sodium hydroxide solution and hydrochloric acid which are pH adjusting agents
- the volume ratio of particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid (that is, PFAT5 value) (unit) : Volume%) was calculated. The calculated value was rounded off to the third decimal place. Based on the calculated PFAT5 value, the effect of suppressing the formation of coarse particles was evaluated according to the same evaluation criteria as described above. Table 3 shows the measured values and evaluation results of the PFAT5 value. If the evaluation result was “AA” or “A”, it was judged as acceptable.
- the peaks with a peak area of 0.01 area% or more were picked up and picked up.
- the ratio of the peak area of the hydrolyzate of flurbiprofen axetil detected at a retention time of 4.3 minutes with respect to all the peak areas hereinafter referred to as “the peak area ratio of the hydrolyzate”.
- AA The peak area ratio of the hydrolyzate is less than 0.5 area%.
- A The peak area ratio of the hydrolyzate is 0.5 area% or more and less than 1.0 area%.
- B The peak area ratio of the hydrolyzate is 1.0 area% or more and less than 2.0 area%.
- C The peak area ratio of the hydrolyzate is 2.0 area% or more.
- the oil-in-water emulsion compositions obtained by the production methods of Examples 14 to 20 are the same as the oil-in-water emulsion compositions obtained by the production methods of Comparative Examples 9 to 13. In comparison, there were few coarse particles. From these results, it was clarified that the production methods of Examples 14 to 20 were production methods capable of producing an oil-in-water emulsion composition with few coarse particles even after the heat treatment step. .
- oil-in-water emulsion compositions obtained by the production methods of Examples 14 to 20 had few hydrolysates of flurbiprofen axetyl, which is a specific compound. From these results, it was revealed that the production methods of Examples 14 to 20 are production methods in which the specific compound is difficult to hydrolyze and the specific compound is excellent in stability.
- an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 4.
- the pH of the emulsion Y is adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted.
- the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 ⁇ m.
- Example 25 ⁇ Example 25, Example 30 and Example 31>
- Each component described in Table 4 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments shown in Table 4 at a pressure shown in Table 4 using a high-pressure homogenizer to obtain an emulsion.
- a homogenizer product name: ULTRA-TURRAX (registered trademark), IKA Corporation
- the pH of the emulsion was adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Subsequently, the emulsion after pH adjustment was filtered using a filter having a pore diameter of 0.8 ⁇ m as necessary. In Example 25, filtration was performed, and in Examples 30 and 31, filtration was not performed.
- a sodium hydroxide solution and hydrochloric acid which are pH adjusting agents
- the pH of the emulsion was adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. .
- the pH-adjusted emulsion was filtered using a filter having a pore size of 0.8 ⁇ m.
- 2 mL of the emulsion after filtration was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal were attached. Used to seal the container.
- the emulsion contained in the container is subjected to a heat treatment at 121 ° C.
- the pH of the emulsion was adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. .
- a sodium hydroxide solution and hydrochloric acid which are pH adjusting agents
- water water
- 2 mL of the emulsion after pH adjustment was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container.
- the emulsion contained in the container is subjected to a heat treatment at 121 ° C.
- PFAT5 value The oil-in-water emulsion composition after the severe test was diluted 10 times with water to prepare a sample solution for evaluation. 1 mL of the prepared sample solution for evaluation is injected into a number-counting particle size distribution apparatus [Product name: AccuSizer 780AS, Nihon Entegris Co., Ltd.] It was automatically calculated according to the function of the apparatus. Then, in the same manner as described above, the volume ratio of particles having a diameter of 5 ⁇ m or more and 50 ⁇ m or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid (that is, PFAT5 value) (unit) : Volume%) was calculated. The calculated value was rounded off to the third decimal place.
- Table 4 shows the measured values and evaluation results of the PFAT5 value. If the evaluation result was “AA” or “A”, it was judged as acceptable.
- AA The amount of flurbiprofen axetil hydrolyzate is less than 3.5 area%.
- A The amount of hydrolyzate of flurbiprofen axetil is 3.5 area% or more and less than 4.5 area%.
- B The amount of hydrolyzate of flurbiprofen axetil is 4.5 area% or more.
- Table 4 shows measured values and evaluation results of the amount of palmitic acid. If the evaluation result was “AA” or “A”, it was judged as acceptable.
- AA The amount of palmitic acid is less than 0.03% by mass.
- A The amount of palmitic acid is 0.03% by mass or more and less than 0.05% by mass.
- B The amount of palmitic acid is 0.05% by mass or more.
- the oil-in-water emulsion compositions produced by the production methods of Examples 21 to 31 and then stored in a harsh environment were obtained by the production methods of Comparative Examples 14 and 15. There were few coarse particles compared with the obtained oil-in-water emulsion composition. From these results, it became clear that according to the production methods of Examples 21 to 31, an oil-in-water emulsion composition with few coarse particles can be produced even after the heat treatment step. Further, it was revealed that the oil-in-water emulsion compositions obtained by the production methods of Examples 21 to 31 are difficult to produce coarse particles even when stored in a harsh environment.
- the oil-in-water emulsion composition produced by the production method of Examples 21 to 31 and stored in a harsh environment had a small amount of the hydrolyzate of flurbiprofen axetyl, which is a specific compound. . From these results, it was revealed that the production methods of Examples 21 to 31 are production methods in which the specific compound is difficult to hydrolyze in the heat treatment step and is excellent in stability of the specific compound. In addition, the oil-in-water emulsion compositions obtained by the production methods of Examples 21 to 31 are resistant to hydrolysis of the specific compound even when stored in a harsh environment, and are excellent in stability of the specific compound. Became clear.
- the oil-in-water emulsion composition produced by the production method of Examples 21 to 31 and stored in a harsh environment had a small amount of palmitic acid, which is a fatty acid produced by the decomposition of phospholipids and triglycerides. . From these results, it is clear that the production methods of Examples 21 to 31 are production methods in which phospholipids and triglycerides are hardly decomposed in the heat treatment step and phospholipids and triglycerides are excellent in stability. It became. In addition, the oil-in-water emulsion compositions obtained by the production methods of Examples 21 to 31 hardly generate fatty acids derived from phospholipids and triglycerides even when stored in a harsh environment. It was revealed that the triglyceride was excellent in stability.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Physical Education & Sports Medicine (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Neurology (AREA)
- Pain & Pain Management (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Rheumatology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Neurosurgery (AREA)
- Immunology (AREA)
- Biochemistry (AREA)
- Dispersion Chemistry (AREA)
- Dermatology (AREA)
- Biophysics (AREA)
- Emergency Medicine (AREA)
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
本開示は、水中油型乳化組成物の製造方法に関する。 The present disclosure relates to a method for producing an oil-in-water emulsion composition.
従来、医薬品として、薬効成分、リン脂質、トリグリセリド、及び界面活性剤を含む脂肪乳剤が知られている。脂肪乳剤は、例えば、医療現場において、直接注射することにより患者に投与されるか、或いは、生理食塩水等の輸液と混合し、点滴注射により患者に投与される。
例えば、中国特許出願公開第104706575号明細書には、フルルビプロフェンアキセチル、リン脂質、トリグリセリド、及び界面活性剤を含み、生理食塩水等の輸液との混合時に自発的に乳化する溶液組成物が開示されている。
Conventionally, fat emulsions containing medicinal ingredients, phospholipids, triglycerides, and surfactants are known as pharmaceuticals. The fat emulsion is administered to the patient by direct injection, for example, in the medical field, or mixed with an infusion solution such as physiological saline and administered to the patient by instillation.
For example, Chinese Patent Application No. 1070656575 includes flurbiprofen axetil, phospholipid, triglyceride, and surfactant, and a solution composition that spontaneously emulsifies upon mixing with an infusion such as physiological saline. Things are disclosed.
ところで、フルルビプロフェンアキセチルを含む水中油型の乳化組成物を容器に収容して加熱処理すると、例えば、乳化組成物中の乳化粒子が凝集することで、μmオーダーの粗大粒子が生成する場合がある。例えば、静脈内注射によって投与される水中油型乳化組成物では、水中油型乳化組成物中に粗大粒子が多く存在すると、投与後に血管が閉塞するという問題が生じ得る。そのため、粗大粒子は少ないことが望ましい。 By the way, when an oil-in-water emulsion composition containing flurbiprofen axetil is placed in a container and heat-treated, for example, the emulsion particles in the emulsion composition aggregate to produce coarse particles on the order of μm. There is a case. For example, in an oil-in-water emulsion composition administered by intravenous injection, if a large amount of coarse particles are present in the oil-in-water emulsion composition, a problem may occur in that blood vessels are blocked after administration. Therefore, it is desirable that there are few coarse particles.
上述の点に関し、中国特許出願公開第104706575号明細書には、フルルビプロフェンアキセチルを含む脂肪乳剤が記載されているものの、フルルビプロフェンアキセチルを含む乳化組成物において生じ得る、加熱処理に起因する粗大粒子の生成に関する記載はない。また、中国特許出願公開第104706575号明細書では、加熱処理に起因する粗大粒子の生成を抑制することについて何ら着目していない。 In regard to the above points, while Chinese Patent Application No. 1070656575 describes a fat emulsion containing flurbiprofen axetil, the heating that can occur in an emulsified composition comprising flurbiprofen axetil. There is no description about the production | generation of the coarse particle resulting from a process. In addition, in the specification of Chinese Patent Application No. 10706575, no attention is paid to suppressing the generation of coarse particles due to heat treatment.
本発明の一実施形態が解決しようとする課題は、フルルビプロフェンアキセチル及びフルルビプロフェンの少なくとも一方の化合物を含む水中油型乳化組成物であって、加熱処理の工程を経て製造された場合でも、粗大粒子が少ない水中油型乳化組成物の製造方法を提供することである。 A problem to be solved by an embodiment of the present invention is an oil-in-water emulsion composition comprising at least one compound of flurbiprofen axetil and flurbiprofen, which is produced through a heat treatment step. The present invention also provides a method for producing an oil-in-water emulsion composition with few coarse particles.
上記課題を解決するための手段には、以下の態様が含まれる。
<1> リン脂質と、トリグリセリドと、フルルビプロフェンアキセチル及びフルルビプロフェンから選ばれる少なくとも1種の化合物と、水と、を含む被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得る工程Aと、
工程Aにて得られた乳化物を少なくとも含む被加熱処理物であって、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤を含み、かつ、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルの合計含有率が、被加熱処理物の全質量に対して0.6質量%以上1.8質量%以下である被加熱処理物に対し、加熱処理を施す工程Bと、
を有する水中油型乳化組成物の製造方法。
<2> 工程Bでは、被加熱処理物に対し、90℃以上140℃以下の温度で加熱処理を施す<1>に記載の水中油型乳化組成物の製造方法。
<3> ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤が、ポリオキシエチレンソルビタン脂肪酸エステルである<1>又は<2>に記載の水中油型乳化組成物の製造方法。
<4> 工程Aでは、リン脂質と、トリグリセリドと、フルルビプロフェンアキセチル及びフルルビプロフェンから選ばれる少なくとも1種の化合物と、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤と、水と、を含む被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得る<1>~<3>のいずれか1つに記載の水中油型乳化組成物の製造方法。
<5> 工程Aでは、リン脂質と、トリグリセリドと、フルルビプロフェンアキセチル及びフルルビプロフェンから選ばれる少なくとも1種の化合物と、水と、を含み、かつ、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルから選ばれる界面活性剤を含まない被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得る<1>~<3>のいずれか1つに記載の水中油型乳化組成物の製造方法。
<6> 工程Aでは、被乳化処理物に対し、80MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得る<1>~<5>のいずれか1つに記載の水中油型乳化組成物の製造方法。
<7> 工程Bにおける被加熱処理物のpHが、6.0以上10.0以下である<1>~<6>のいずれか1つに記載の水中油型乳化組成物の製造方法。
<8> 工程Bにおける被加熱処理物のpHが、7.0以上9.0以下である<1>~<7>のいずれか1つに記載の水中油型乳化組成物の製造方法。
<9> 被乳化処理物が、多価アルコールを含む<1>~<8>のいずれか1つに記載の水中油型乳化組成物の製造方法。
Means for solving the above problems include the following aspects.
<1> With respect to an emulsified product containing phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen, and water, the pressure is 20 MPa or more and 350 MPa or less. Step A to obtain an emulsion by applying an emulsification treatment;
A to-be-heated product containing at least the emulsion obtained in step A, comprising at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester, and sorbitan fatty acid The step of subjecting the object to be heated, in which the total content of the ester and the polyoxyethylene sorbitan fatty acid ester is 0.6% by mass or more and 1.8% by mass or less based on the total mass of the object to be heated. B and
The manufacturing method of the oil-in-water emulsion composition which has NO.
<2> The process for producing an oil-in-water emulsion composition according to <1>, wherein in step B, the heat-treated object is subjected to heat treatment at a temperature of 90 ° C. or higher and 140 ° C. or lower.
<3> The oil-in-water emulsification according to <1> or <2>, wherein at least one surfactant selected from the group consisting of sorbitan fatty acid esters and polyoxyethylene sorbitan fatty acid esters is a polyoxyethylene sorbitan fatty acid ester. A method for producing the composition.
<4> In step A, selected from the group consisting of phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen, sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester Any one of <1> to <3>, wherein an emulsified product is obtained by subjecting an emulsified product containing at least one surfactant and water to an emulsified treatment at a pressure of 20 MPa to 350 MPa. The manufacturing method of the oil-in-water type emulsion composition as described in one.
<5> Step A includes phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen, and water, and sorbitan fatty acid ester and polyoxyethylene. Any one of <1> to <3> to obtain an emulsion by performing an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less on an article to be emulsified containing no surfactant selected from sorbitan fatty acid esters The manufacturing method of the oil-in-water type emulsion composition of description.
<6> In the step A, an oil-in-water mold according to any one of <1> to <5>, wherein an emulsion is obtained by performing an emulsification treatment with a pressure of 80 MPa or more and 350 MPa or less on the article to be emulsified A method for producing an emulsified composition.
<7> The method for producing an oil-in-water emulsion composition according to any one of <1> to <6>, wherein the pH of the object to be heated in step B is 6.0 or more and 10.0 or less.
<8> The method for producing an oil-in-water emulsion composition according to any one of <1> to <7>, wherein the pH of the object to be heated in step B is 7.0 or more and 9.0 or less.
<9> The method for producing an oil-in-water emulsified composition according to any one of <1> to <8>, in which the product to be emulsified contains a polyhydric alcohol.
本発明の一実施形態によれば、フルルビプロフェンアキセチル及びフルルビプロフェンの少なくとも一方の化合物を含む水中油型乳化組成物であって、加熱処理の工程を経て製造された場合でも、粗大粒子が少ない水中油型乳化組成物の製造方法が提供される。 According to one embodiment of the present invention, an oil-in-water emulsion composition comprising at least one compound of flurbiprofen axetil and flurbiprofen, even when manufactured through a heat treatment step, A method for producing an oil-in-water emulsion composition with few coarse particles is provided.
以下、本発明を適用した水中油型乳化組成物の製造方法の実施形態の一例について説明する。但し、本発明は、以下の実施形態に何ら限定されるものではなく、本発明の目的の範囲内において、適宜、変更を加えて実施することができる。 Hereinafter, an example of an embodiment of a method for producing an oil-in-water emulsion composition to which the present invention is applied will be described. However, the present invention is not limited to the following embodiments, and can be implemented with appropriate modifications within the scope of the object of the present invention.
本開示において「~」を用いて示された数値範囲は、「~」の前後に記載される数値をそれぞれ最小値及び最大値として含む範囲を意味する。
本開示に段階的に記載されている数値範囲において、ある数値範囲で記載された上限値又は下限値は、他の段階的な記載の数値範囲の上限値又は下限値に置き換えてもよい。また、本開示に記載されている数値範囲において、ある数値範囲で記載された上限値又は下限値は、実施例に示されている値に置き換えてもよい。
本開示において、2以上の好ましい態様の組み合わせは、より好ましい態様である。
In the present disclosure, a numerical range indicated by using “to” means a range including the numerical values described before and after “to” as the minimum value and the maximum value, respectively.
In a numerical range described in stages in the present disclosure, an upper limit value or a lower limit value described in a numerical range may be replaced with an upper limit value or a lower limit value in another numerical range. Further, in the numerical ranges described in the present disclosure, the upper limit value or the lower limit value described in a certain numerical range may be replaced with the values shown in the examples.
In the present disclosure, a combination of two or more preferred embodiments is a more preferred embodiment.
本開示において、各成分の量は、各成分に該当する物質が複数種存在する場合には、特に断らない限り、複数種の物質の合計量を意味する。 In the present disclosure, the amount of each component means the total amount of a plurality of types of substances unless there is a specific case when there are a plurality of types of substances corresponding to each component.
本開示において、「粗大粒子が少ない」水中油型乳化組成物とは、水中油型乳化組成物中に含まれる乳化粒子の合計体積に対する、水中油型乳化組成物中の直径5μm以上50μm以下の粒子の体積割合が、0.05体積%以下である水中油型乳化組成物を意味する。
本開示では、「水中油型乳化組成物中に含まれる乳化粒子の合計体積に対する、水中油型乳化組成物中の直径5μm以上50μm以下の粒子の体積割合(単位:体積%)」を「PFAT5値」と称する場合がある。
PFAT5値は、好ましくは0.03体積%以下であり、より好ましくは0.01体積%以下であり、特に好ましくは、直径5μm以上50μm以下の粒子を含まないこと、即ち、0体積%である。
In the present disclosure, the “small amount of coarse particles” oil-in-water emulsion composition means that the oil-in-water emulsion composition has a diameter of 5 μm to 50 μm with respect to the total volume of the emulsion particles contained in the oil-in-water emulsion composition. It means an oil-in-water emulsion composition in which the volume ratio of particles is 0.05% by volume or less.
In the present disclosure, “the volume ratio (unit: volume%) of particles having a diameter of 5 μm to 50 μm in the oil-in-water emulsion composition to the total volume of the emulsion particles contained in the oil-in-water emulsion composition” is expressed as “PFAT5 Sometimes referred to as “value”.
The PFAT5 value is preferably 0.03% by volume or less, more preferably 0.01% by volume or less, and particularly preferably no particles having a diameter of 5 μm or more and 50 μm or less, that is, 0% by volume. .
本開示において、「PFAT5値」は、下記の方法により求めた値である。
水中油型乳化組成物を、水を用いて10倍希釈し、評価用サンプル液を調製する。調製した評価用サンプル液1mLを、個数カウント式粒度分布装置に注入し、評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積を、装置の機能により自動にて算出する。そして、下記の式(1)に基づき、評価用サンプル液中に含まれる乳化粒子の合計体積に対する、評価用サンプル液中の直径5μm以上50μm以下の粒子の体積割合(即ち、PFAT5値)(単位:体積%)を算出する。
下記の式(1)では、評価用サンプル液中に含まれる乳化粒子の合計体積が、評価用サンプル液中に含まれるトリグリセリドの体積と同程度であると仮定している。なお、評価用サンプル液中に含まれるトリグリセリドの体積は、下記の式(2)に基づき、算出する。
個数カウント式粒度分布装置としては、例えば、日本インテグリス(株)のAccuSizer 780AS(製品名)を好適に用いることができる。但し、個数カウント式粒度分布装置は、これに限定されない。
In the present disclosure, the “PFAT5 value” is a value obtained by the following method.
The oil-in-water emulsion composition is diluted 10 times with water to prepare a sample solution for evaluation. 1 mL of the prepared sample solution for evaluation is injected into a number counting particle size distribution device, and the total volume of particles having a diameter of 5 μm or more and 50 μm or less in 1 mL of the sample solution for evaluation is automatically calculated by the function of the device. Based on the following formula (1), the volume ratio of particles having a diameter of 5 μm or more and 50 μm or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid (that is, PFAT5 value) (unit) : Volume%).
In the following formula (1), it is assumed that the total volume of the emulsified particles contained in the evaluation sample liquid is approximately the same as the volume of the triglyceride contained in the evaluation sample liquid. In addition, the volume of the triglyceride contained in the sample liquid for evaluation is calculated based on the following formula (2).
As the number-counting particle size distribution device, for example, AccuSizer 780AS (product name) manufactured by Nihon Integris Co., Ltd. can be preferably used. However, the number counting type particle size distribution apparatus is not limited to this.
PFAT5値(単位:体積%) = [評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積] ÷ [評価用サンプル液1mLに占めるトリグリセリドの体積] × 100・・・(1) PFAT5 value (unit: volume%) = [total volume of particles having a diameter of 5 μm to 50 μm in 1 mL of sample liquid for evaluation] ÷ [volume of triglyceride in 1 mL of sample liquid for evaluation] × 100 (1)
評価用サンプル液1mLに占めるトリグリセリドの体積(単位:mL) = 水中油型乳化組成物中のトリグリセリドの含有量(単位:g) ÷ トリグリセリドの密度(単位:g/mL) ÷ 10(希釈倍率)・・・(2) Triglyceride volume in 1 mL of sample solution for evaluation (unit: mL) = triglyceride content in oil-in-water emulsion composition (unit: g) ÷ triglyceride density (unit: g / mL) ÷ 10 (dilution ratio) ... (2)
本開示において、「工程」の用語は、独立した工程だけではなく、他の工程と明確に区別できない場合であっても、その工程の所期の目的が達成されれば本用語に含まれる。 In the present disclosure, the term “process” is not limited to an independent process, and is included in the term if the intended purpose of the process is achieved even if it cannot be clearly distinguished from other processes.
[水中油型乳化組成物の製造方法]
本開示の水中油型乳化組成物の製造方法(以下、単に「製造方法」ともいう。)は、リン脂質と、トリグリセリドと、フルルビプロフェンアキセチル及びフルルビプロフェンから選ばれる少なくとも1種の化合物(以下、「特定化合物」ともいう。)と、水と、を含む被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得る工程Aと、工程Aにて得られた乳化物を少なくとも含む被加熱処理物であって、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤(以下、「特定界面活性剤)ともいう。)を含み、かつ、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルの合計含有率が、被加熱処理物の全質量に対して0.6質量%以上1.8質量%以下である被加熱処理物に対し、加熱処理を施す工程Bと、を有する。
本開示の製造方法は、必要に応じて、他の工程を有していてもよい。
[Method for producing oil-in-water emulsion composition]
The method for producing an oil-in-water emulsion composition of the present disclosure (hereinafter, also simply referred to as “manufacturing method”) is at least one selected from phospholipids, triglycerides, flurbiprofen axetil and flurbiprofen. Step A and Step of obtaining an emulsion by subjecting an emulsified product containing the above compound (hereinafter also referred to as “specific compound”) and water to an emulsified treatment with a pressure of 20 MPa to 350 MPa. A to-be-heated product containing at least the emulsion obtained in A, and comprising at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester (hereinafter referred to as “specific surfactant”) And the total content of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester is To be heated product is 0.6 wt% to 1.8 wt% or less based on the total weight of the heat treatment product, and a step B of performing heat treatment.
The manufacturing method of this indication may have other processes as needed.
一般に、乳化組成物の製造工程では、乳化組成物を容器に収容した後、滅菌のための加熱処理が行われる。
本発明者らは、フルルビプロフェンアキセチル及びフルルビプロフェンの少なくとも一方の化合物を含む水中油型乳化組成物を容器に収容して加熱処理すると、水中油型乳化組成物中に、粗大粒子が生成する場合があることを見出した。水中油型乳化組成物中に粗大粒子が多く存在すると、例えば、水中油型乳化組成物を静脈内注射によって投与した場合に、血管の閉塞を招来し得る。
Generally, in the manufacturing process of an emulsified composition, after the emulsified composition is contained in a container, a heat treatment for sterilization is performed.
When the inventors of the present invention put an oil-in-water emulsion composition containing at least one of flurbiprofen axetil and flurbiprofen in a container and heat-treated, the oil-in-water emulsion composition is coarse. It has been found that particles may form. When there are many coarse particles in the oil-in-water emulsion composition, for example, when the oil-in-water emulsion composition is administered by intravenous injection, it may lead to occlusion of blood vessels.
これに対し、本開示の製造方法では、リン脂質、トリグリセリド、特定化合物、及び水を含む被乳化処理物に対し、20MPa以上の高い圧力をかける乳化処理を施すことにより得られた乳化物を少なくとも含む被加熱処理物であって、特定の範囲の含有率の特定界面活性剤を含む被加熱処理物に対し、加熱処理を施すことで、特定化合物を含む被加熱処理物の加熱処理に起因する粗大粒子の生成を抑制することができる。そのため、本開示の製造方法により得られる水中油型乳化組成物では、粗大粒子(即ち、直径5μm以上50μm以下の粒子)の体積割合が、水中油型乳化組成物中に含まれる乳化粒子の合計体積に対して0.05体積%以下となり、粗大粒子が少ない。
本開示の製造方法がこのような効果を奏する理由については明らかではないが、本発明者らは、以下のように推測している。
On the other hand, in the production method of the present disclosure, at least an emulsion obtained by subjecting an emulsion to be processed containing phospholipid, triglyceride, specific compound, and water to an emulsion that applies a high pressure of 20 MPa or more. Due to the heat treatment of the heat-treated object including the specific compound by performing the heat treatment on the heat-treated object including the specific surfactant having the content in the specific range. Generation of coarse particles can be suppressed. Therefore, in the oil-in-water emulsion composition obtained by the production method of the present disclosure, the volume ratio of coarse particles (that is, particles having a diameter of 5 μm or more and 50 μm or less) is the sum of the emulsion particles contained in the oil-in-water emulsion composition. It becomes 0.05 volume% or less with respect to the volume, and there are few coarse particles.
Although the reason why the manufacturing method of the present disclosure has such an effect is not clear, the present inventors presume as follows.
本開示の製造方法において、加熱処理に供される被加熱処理物は、20MPa以上の高い圧力をかける乳化処理を経て得られる乳化物を含むため、被加熱処理物中に含まれる乳化粒子は、微細で、かつ、安定なものとなる。
また、加熱処理の際に生じる粗大粒子の生成は、容器内において気液界面付近に存在する乳化粒子が、容器の内部表面に付着することに起因すると考えられるが、本開示の製造方法では、加熱処理に供される被加熱処理物が、特定界面活性剤を特定の範囲の含有率で含むことにより、加熱処理の際に、乳化粒子が容器の内部表面に付着することが抑制される。
すなわち、本開示の製造方法では、加熱処理に供される被加熱処理物中の乳化粒子が、微細かつ安定であること、及び、容器の内部表面に付着し難いことにより、粗大粒子が少ない水中油型乳化組成物を製造できると推測される。
In the production method of the present disclosure, the object to be heated to be subjected to the heat treatment includes an emulsion obtained through an emulsification treatment in which a high pressure of 20 MPa or more is applied. Therefore, the emulsified particles contained in the object to be heated are It is fine and stable.
In addition, it is considered that the generation of coarse particles generated during the heat treatment is caused by the fact that the emulsified particles existing in the vicinity of the gas-liquid interface in the container adhere to the inner surface of the container. When the to-be-heated material to be subjected to the heat treatment contains the specific surfactant with a content in a specific range, the emulsion particles are prevented from adhering to the inner surface of the container during the heat treatment.
That is, in the production method of the present disclosure, the emulsified particles in the heat-treated product to be subjected to the heat treatment are fine and stable, and are difficult to adhere to the inner surface of the container, so that there are few coarse particles in water. It is presumed that an oil-type emulsion composition can be produced.
本開示の製造方法に対し、中国特許出願公開第104706575号明細書では、水中油型乳化組成物を、フルルビプロフェンアキセチルを含む脂肪乳剤に対し、水を添加して、軽く撹拌することにより得ており、20MPa以上の高い圧力をかける乳化処理によっては得ていない。中国特許出願公開第104706575号明細書に記載の脂肪乳剤は、フルルビプロフェンアキセチルを含むものの、フルルビプロフェンアキセチルを含む水中油型乳化組成物を容器に収容して加熱処理した場合に、粗大粒子が生成するという問題について、何ら着目していない。ましてや、中国特許出願公開第104706575号明細書には、水中油型乳化組成物の加熱処理の際に生じ得る、粗大粒子の生成を抑制するための技術についての記載はない。 In the manufacturing method of the present disclosure, in Chinese Patent Application No. 1070656575, an oil-in-water emulsion composition is added to a fat emulsion containing flurbiprofen axetil and lightly stirred. It is not obtained by the emulsification treatment in which a high pressure of 20 MPa or more is applied. The fat emulsion described in Chinese Patent Application No. 1070656575 contains flurbiprofen axetil, but the oil-in-water emulsion composition containing flurbiprofen axetil is contained in a container and heat-treated. In addition, no attention is paid to the problem that coarse particles are generated. In addition, Chinese Patent Application No. 1070656575 does not describe a technique for suppressing the generation of coarse particles that may occur during the heat treatment of an oil-in-water emulsion composition.
一般に、滅菌等の加熱処理では、原料の分解が生じる場合がある。
本開示の製造方法によって得られる水中油型乳化組成物は、特定化合物の分解物(詳細には、加水分解物)が少なく、また、保存中における特定化合物の安定性に優れる。
既述のとおり、本開示の製造方法において、加熱処理に供される被加熱処理物は、圧力が20MPa以上の高圧乳化処理を経て得られる乳化物を含むため、被加熱処理物中に含まれる乳化粒子は、安定なものとなる。また、加熱処理に供される被加熱処理物が、特定界面活性剤を特定の範囲の含有率で含むことで、被加熱処理物中に含まれる乳化粒子は更に安定なものとなる。乳化粒子が安定化すると、乳化粒子に内包された特定化合物がより水に接触し難くなるため、本開示の製造方法によって得られる水中油型乳化組成物は、加熱処理によって生じる特定化合物の分解物が少なく、また、保存中における特定化合物の安定性に優れると推測される。
In general, in heat treatment such as sterilization, decomposition of raw materials may occur.
The oil-in-water emulsion composition obtained by the production method of the present disclosure has few decomposition products (specifically, hydrolyzates) of the specific compound and is excellent in stability of the specific compound during storage.
As described above, in the manufacturing method of the present disclosure, the object to be heated to be subjected to the heat treatment includes an emulsion obtained through a high-pressure emulsification treatment having a pressure of 20 MPa or more, and thus is included in the object to be heated. The emulsified particles are stable. Moreover, the to-be-heated material used for a heat processing contains the specific surfactant by the content rate of a specific range, and the emulsified particle contained in a to-be-heated material becomes still more stable. When the emulsified particles are stabilized, the specific compound included in the emulsified particles is less likely to come into contact with water. Therefore, the oil-in-water emulsion composition obtained by the production method of the present disclosure is a decomposition product of the specific compound generated by heat treatment. It is estimated that the stability of the specific compound during storage is excellent.
本開示の製造方法によって得られる水中油型乳化組成物は、リン脂質及びトリグリセリドの分解によって生じる脂肪酸が少なく、また、保存中におけるリン脂質及びトリグリセリドの安定性に優れる。
既述のとおり、本開示の製造方法において、加熱処理に供される被加熱処理物は、圧力が20MPa以上の高圧乳化処理を経て得られるため、被加熱処理物中に含まれる乳化粒子は、安定なものとなる。また、加熱処理に供される被加熱処理物が、特定界面活性剤を特定の範囲の含有率で含むことで、被加熱処理物中に含まれる乳化粒子は更に安定なものとなる。乳化粒子が安定化すると、乳化粒子を形成するリン脂質及びトリグリセリドも安定化されるため、本開示の製造方法によって得られる水中油型乳化組成物は、加熱処理によって生じるリン脂質及びトリグリセリドに由来の脂肪酸、即ち、リン脂質及びトリグリセリドの分解によって生成する脂肪酸が少なく、また、保存中におけるリン脂質及びトリグリセリドの安定性に優れると推測される。
The oil-in-water emulsion composition obtained by the production method of the present disclosure has few fatty acids generated by the decomposition of phospholipids and triglycerides, and is excellent in stability of phospholipids and triglycerides during storage.
As described above, in the production method of the present disclosure, the heat-treated product to be subjected to the heat treatment is obtained through a high-pressure emulsification treatment with a pressure of 20 MPa or more. Therefore, the emulsified particles contained in the heat-treated material are: It will be stable. Moreover, the to-be-heated material used for a heat processing contains the specific surfactant by the content rate of a specific range, and the emulsified particle contained in a to-be-heated material becomes still more stable. When the emulsified particles are stabilized, the phospholipids and triglycerides that form the emulsified particles are also stabilized. Therefore, the oil-in-water emulsion composition obtained by the production method of the present disclosure is derived from the phospholipids and triglycerides generated by the heat treatment. It is estimated that fatty acids, that is, fatty acids produced by the degradation of phospholipids and triglycerides are small, and that the stability of phospholipids and triglycerides during storage is excellent.
なお、上記の推測は、本開示の製造方法を限定的に解釈するものではなく、一例として説明するものである。 Note that the above estimation does not limit the manufacturing method of the present disclosure, and is described as an example.
以下、本開示の製造方法における各工程について詳細に説明する。 Hereinafter, each step in the manufacturing method of the present disclosure will be described in detail.
〔工程A〕
工程Aは、リン脂質と、トリグリセリドと、フルルビプロフェンアキセチル及びフルルビプロフェンから選ばれる少なくとも1種の化合物(即ち、特定化合物)と、水と、を含む被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得る工程である。
工程Aにおける20MPa以上350MPa以下の圧力による乳化処理は、20MPa以上350MPa以下の圧力を、被乳化処理物に対して付加する乳化処理を意味する。
工程Aでは、特定化合物及びトリグリセリドを含む乳化粒子が水中に分散された水中油型の乳化物を得ることができる。工程Aにおいて得られる乳化物中の乳化粒子は、微細で、かつ、安定なものとなる。
[Process A]
Step A is for an emulsified product containing phospholipid, triglyceride, at least one compound selected from flurbiprofen axetil and flurbiprofen (that is, a specific compound), and water. This is a step of obtaining an emulsion by performing an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less.
The emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less in Step A means an emulsification treatment in which a pressure of 20 MPa or more and 350 MPa or less is added to the product to be emulsified.
In step A, an oil-in-water emulsion in which emulsified particles containing the specific compound and triglyceride are dispersed in water can be obtained. The emulsified particles in the emulsion obtained in step A are fine and stable.
リン脂質、トリグリセリド、特定化合物、及び水を含む被乳化処理物は、リン脂質、トリグリセリド、特定化合物、及び水を含む混合物であってもよく、この混合物に対し、乳化処理を施した粗乳化物であってもよい。
すなわち、工程Aは、リン脂質、トリグリセリド、特定化合物、及び水を含む混合物を調製する工程A-1を有していてもよく、工程A-1にて調製された混合物に対し、乳化処理を施して粗乳化物を得る工程A-2を有していてもよい。
The emulsified processed product containing phospholipid, triglyceride, specific compound and water may be a mixture containing phospholipid, triglyceride, specific compound and water, and this mixture is a crude emulsion obtained by emulsifying the mixture. It may be.
That is, step A may include step A-1 for preparing a mixture containing phospholipid, triglyceride, specific compound, and water, and the mixture prepared in step A-1 is subjected to an emulsification treatment. Step A-2 may be provided to give a crude emulsion.
工程A-1において、リン脂質、トリグリセリド、特定化合物、及び水を含む混合物を調製する方法は、特に制限されない。
混合物中に含まれる各成分は、単に混合されていればよく、均一に混合されていることが好ましい。
混合物中に含まれる各成分は、一度に混合してもよく、或いは、1つの成分に別の成分を分割して添加しながら混合してもよい。
混合物中に含まれる各成分を混合する方法としては、特に制限されず、例えば、撹拌により混合する方法が挙げられる。
撹拌手段としては、特に制限はなく、一般的な撹拌器具又は撹拌装置を使用できる。
撹拌時間は、特に制限されず、撹拌器具又は撹拌装置の種類、撹拌する成分の組成(即ち、種類及び量)等に応じて、適宜設定できる。
混合物中に含まれる各成分を混合する際の温度は、特に制限されず、例えば、5℃以上90℃未満の範囲に設定できる。
温度を調整する手段としては、特に制限はなく、一般的な加熱装置を使用できる。
In Step A-1, a method for preparing a mixture containing phospholipid, triglyceride, specific compound, and water is not particularly limited.
Each component contained in a mixture should just be mixed, and it is preferable that it is mixed uniformly.
Each component contained in the mixture may be mixed at one time, or may be mixed while adding another component dividedly into one component.
The method for mixing each component contained in the mixture is not particularly limited, and examples thereof include a method of mixing by stirring.
There is no restriction | limiting in particular as a stirring means, A common stirring instrument or stirring apparatus can be used.
The stirring time is not particularly limited, and can be appropriately set according to the type of stirring tool or stirring device, the composition (ie, type and amount) of the components to be stirred, and the like.
The temperature at the time of mixing each component contained in a mixture is not restrict | limited in particular, For example, it can set to the range of 5 to 90 degreeC.
There is no restriction | limiting in particular as a means to adjust temperature, A general heating apparatus can be used.
工程A-2における乳化処理の方法としては、特に制限はない。
粗乳化物を得るための乳化処理の手段としては、特に制限はなく、例えば、一般的な乳化装置を用いる手段が挙げられる。
乳化装置としては、ホモジナイザー、ホモミキサー、超音波ホモジナイザー等が挙げられる。
乳化時間は、特に制限されず、乳化装置の種類、混合物の組成等に応じて、適宜設定できる。
乳化処理の手段として、ホモジナイザーを用いる場合には、例えば、回転速度を1,000rpm(revolutions per minute;以下、同じ。)~100,000rpm、乳化時間を1分間~30分間に設定できる。
乳化手段として、超音波ホモジナイザーを用いる場合には、例えば、周波数を15kHz~40kHz、分散部のエネルギー密度を100W/cm2以上に設定できる。
The method for the emulsification treatment in step A-2 is not particularly limited.
There is no restriction | limiting in particular as a means of the emulsification process for obtaining a rough emulsion, For example, the means using a general emulsification apparatus is mentioned.
Examples of the emulsifier include a homogenizer, a homomixer, and an ultrasonic homogenizer.
The emulsification time is not particularly limited, and can be appropriately set according to the type of the emulsifying device, the composition of the mixture, and the like.
When a homogenizer is used as a means for the emulsification treatment, for example, the rotation speed can be set to 1,000 rpm (revolutions per minute; hereinafter the same) to 100,000 rpm, and the emulsification time can be set to 1 to 30 minutes.
When an ultrasonic homogenizer is used as the emulsifying means, for example, the frequency can be set to 15 kHz to 40 kHz, and the energy density of the dispersed portion can be set to 100 W / cm 2 or more.
被乳化処理物は、必要に応じて、リン脂質、トリグリセリド、特定化合物、及び水以外の成分を含んでいてもよい。被乳化処理物は、例えば、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤(即ち、特定界面活性剤)を含んでいてもよい。また、被乳化処理物は、後述する他の添加剤(例えば、多価アルコール)を含んでいてもよい。 The processed product to be emulsified may contain components other than phospholipids, triglycerides, specific compounds, and water as necessary. The processed product to be emulsified may contain, for example, at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester (that is, a specific surfactant). Moreover, the to-be-emulsified processed material may contain the other additive (for example, polyhydric alcohol) mentioned later.
工程Aでは、リン脂質と、トリグリセリドと、特定化合物と、特定界面活性剤と、水と、を含む被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得てもよく、リン脂質と、トリグリセリドと、特定化合物と、水と、を含み、かつ、特定界面活性剤を含まない被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すことにより、乳化物を得てもよい。 In step A, an emulsified product is subjected to an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less with respect to an emulsified treatment containing phospholipid, triglyceride, specific compound, specific surfactant, and water. It may be obtained, and emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less is performed on an article to be emulsified containing phospholipid, triglyceride, specific compound, and water and not containing a specific surfactant. From the above, an emulsion may be obtained.
工程Aにおいて、リン脂質と、トリグリセリドと、特定化合物と、特定界面活性剤と、水と、を含む被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施した場合には、特定化合物の分解物がより少ない水中油型乳化組成物が得られる傾向がある。
工程Aにおいて、リン脂質と、トリグリセリドと、特定化合物と、水と、を含み、かつ、特定界面活性剤を含まない被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施した場合には、リン脂質及びトリグリセリドの分解によって生じる脂肪酸がより少ない水中油型乳化組成物が得られる傾向がある。また、特定界面活性剤を含む被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施すと、泡立ちが生じやすいが、被乳化処理物が特定界面活性剤を含まない場合には、泡立ちが生じ難く、製造適性により優れる傾向がある。
In Step A, if an emulsified treatment containing a phospholipid, a triglyceride, a specific compound, a specific surfactant, and water is subjected to an emulsification treatment at a pressure of 20 MPa to 350 MPa, it is specified. There is a tendency to obtain an oil-in-water emulsion composition in which the decomposition product of the compound is less.
In Step A, when an emulsified treatment containing a phospholipid, a triglyceride, a specific compound, and water and containing no specific surfactant is subjected to an emulsification treatment at a pressure of 20 MPa to 350 MPa. Tends to provide an oil-in-water emulsion composition with fewer fatty acids resulting from the degradation of phospholipids and triglycerides. Further, when the emulsified treatment containing the specific surfactant is subjected to an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less, foaming is likely to occur, but when the emulsified treatment does not contain the specific surfactant, Foaming does not easily occur and tends to be more excellent in production suitability.
被乳化処理物中における、トリグリセリドの含有量に対するリン脂質の含有量の割合(即ち、リン脂質の含有量/トリグリセリドの含有量)は、特に制限されず、例えば、水中油型の形態をより良好に保持する乳化物を得る観点から、質量基準で、0.03以上2.0以下であることが好ましく、0.06以上1.0以下であることがより好ましく、0.12以上0.5以下であることが更に好ましい。 The ratio of the phospholipid content to the triglyceride content in the processed product to be emulsified (that is, the phospholipid content / the triglyceride content) is not particularly limited. For example, the oil-in-water type is better. From the standpoint of obtaining an emulsion to be retained in the film, it is preferably 0.03 or more and 2.0 or less, more preferably 0.06 or more and 1.0 or less, and more preferably 0.12 or more and 0.5 on a mass basis. More preferably, it is as follows.
被乳化処理物中におけるトリグリセリドの含有率としては、特に制限はない。
被乳化処理物中におけるトリグリセリドの含有率は、例えば、トリグリセリドに溶解する特定化合物の含有量を増加させる観点から、被乳化処理物の全質量に対して、2質量%以上が好ましく、5質量%以上がより好ましく、10質量%以上が更に好ましい。
また、被乳化処理物中におけるトリグリセリドの含有率は、例えば、より微細な乳化粒子を含む乳化物を得る観点から、被乳化処理物の全質量に対して、40質量%以下が好ましく、30質量%以下がより好ましく、20質量%以下が更に好ましい。
There is no restriction | limiting in particular as content rate of the triglyceride in a to-be-emulsified processed material.
From the viewpoint of increasing the content of the specific compound dissolved in the triglyceride, the content of triglyceride in the processed product is preferably 2% by mass or more, and preferably 5% by mass with respect to the total mass of the processed product. The above is more preferable, and 10 mass% or more is still more preferable.
Further, the content of triglyceride in the product to be emulsified is preferably 40% by mass or less, and preferably 30% by mass with respect to the total mass of the product to be emulsified from the viewpoint of obtaining an emulsion containing finer emulsified particles. % Or less is more preferable, and 20% by mass or less is still more preferable.
被乳化処理物中における特定化合物の含有率としては、特に制限はない。
被乳化処理物中における特定化合物の含有率は、例えば、1回あたりの特定化合物の投与量の観点から、被乳化処理物の全質量に対して、0.01質量%以上2.0質量%以下が好ましい。
There is no restriction | limiting in particular as content rate of the specific compound in an to-be-emulsified processed material.
The content of the specific compound in the processed product to be emulsified is, for example, from 0.01% by mass to 2.0% by mass with respect to the total mass of the processed product from the viewpoint of the dose of the specific compound per one time. The following is preferred.
被乳化処理物中における水の含有率としては、特に制限はない。
被乳化処理物中における水の含有率は、例えば、水中油型の形態の乳化物を得る観点から、被乳化処理物の全質量に対して、好ましくは30質量%以上であり、より好ましくは40質量%以上であり、更に好ましくは50質量%以上である。
また、被乳化処理物中における水の含有率は、例えば、上記と同様の観点から、被乳化処理物の全質量に対して、好ましくは99質量%以下であり、より好ましくは98質量%以下であり、更に好ましくは97質量%以下である。
There is no restriction | limiting in particular as content rate of the water in a to-be-emulsified processed material.
For example, from the viewpoint of obtaining an oil-in-water emulsion, the content of water in the emulsion to be processed is preferably 30% by mass or more, more preferably, based on the total mass of the emulsion to be processed. It is 40 mass% or more, More preferably, it is 50 mass% or more.
Moreover, the content rate of water in the to-be-emulsified processed material is, for example, from the same viewpoint as described above, preferably 99% by mass or less, more preferably 98% by mass or less, with respect to the total mass of the emulsified processed product. More preferably, it is 97 mass% or less.
被乳化処理物が特定界面活性剤を含む場合、被乳化処理物中における特定界面活性剤の含有率は、被乳化処理物の全質量に対して、好ましくは0.6質量%以上2.25質量%以下であり、より好ましくは0.8質量%以上2.2質量%以下であり、更に好ましくは1.0質量%以上1.9質量%以下であり、特に好ましくは1.25質量%以上1.9質量%以下である。
被乳化処理物中における特定界面活性剤の含有率が上記の範囲内であると、加熱処理の工程(即ち、工程B)を経た場合でも、粗大粒子が少ない水中油型乳化組成物が得られる傾向がある。
When the to-be-emulsified processed product contains a specific surfactant, the content of the specific surfactant in the to-be-emulsified processed product is preferably 0.6% by mass or more and 2.25 with respect to the total mass of the to-be-emulsified processed product. % By mass or less, more preferably 0.8% by mass or more and 2.2% by mass or less, still more preferably 1.0% by mass or more and 1.9% by mass or less, and particularly preferably 1.25% by mass. The content is 1.9% by mass or less.
When the content of the specific surfactant in the emulsion to be emulsified is within the above range, an oil-in-water emulsion composition with few coarse particles can be obtained even after the heat treatment step (ie, step B). Tend.
被乳化処理物が多価アルコールを含む場合、被乳化処理物中における多価アルコールの含有率は、例えば、より微細な乳化粒子を含む乳化物を得る観点から、被乳化処理物の全質量に対して、0.5質量%以上が好ましく、1.0質量%以上がより好ましく、2.0質量%以上が更に好ましい。
また、被乳化処理物中における多価アルコールの含有率は、例えば、注射により投与する際の浸透圧差による組織障害の可能性をより低減させる観点から、被乳化処理物の全質量に対して、10質量%以下が好ましく、5質量%以下がより好ましく、3質量%以下が更に好ましい。
When the product to be emulsified contains polyhydric alcohol, the content of the polyhydric alcohol in the product to be emulsified is, for example, from the viewpoint of obtaining an emulsion containing finer emulsified particles, to the total mass of the product to be emulsified. On the other hand, 0.5 mass% or more is preferable, 1.0 mass% or more is more preferable, and 2.0 mass% or more is still more preferable.
In addition, the content of the polyhydric alcohol in the emulsified treatment is, for example, from the viewpoint of further reducing the possibility of tissue damage due to osmotic pressure difference when administered by injection, with respect to the total mass of the emulsified treatment, 10 mass% or less is preferable, 5 mass% or less is more preferable, and 3 mass% or less is still more preferable.
工程Aにおいて、被乳化処理物に対し、20MPa以上350MPa以下の圧力による乳化処理を施す手段としては、特に制限はない。
乳化処理の手段としては、例えば、高圧ホモジナイザーが好適である。
高圧ホモジナイザーによる分散は、液体が非常に狭い隙間を高速で通過する際に発生する大きなせん断力による分散である。
高圧ホモジナイザーとしては、例えば、処理液の流路を固定するチャンバーを有するチャンバー型高圧ホモジナイザー、及び均質バルブを有する均質バルブ型高圧ホモジナイザーが挙げられる。
チャンバー型高圧ホモジナイザーとしては、マイクロフルイダイザー〔パウレック(株)〕、ナノマイザー〔ナノマイザー(株)〕、アルティマイザー〔スギノマシン(株)〕等が挙げられる。
均質バルブ型高圧ホモジナイザーとしては、ゴーリンタイプホモジナイザー〔(株)エスエムテー〕、ラニエタイプホモジナイザー〔(株)エスエムテー〕、高圧ホモジナイザー(ニロ・ソアビ社)、ホモゲナイザー〔三和機械(株)〕、高圧ホモゲナイザー〔イズミフードマシナリー(株)〕、超高圧ホモジナイザー(IKA社)等が挙げられる。
In step A, there is no particular limitation on the means for performing an emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less on the product to be emulsified.
As a means for the emulsification treatment, for example, a high-pressure homogenizer is suitable.
Dispersion by a high-pressure homogenizer is dispersion due to a large shear force generated when a liquid passes through a very narrow gap at high speed.
Examples of the high-pressure homogenizer include a chamber-type high-pressure homogenizer having a chamber for fixing the flow path of the processing liquid and a homogeneous valve-type high-pressure homogenizer having a homogeneous valve.
Examples of the chamber type high-pressure homogenizer include microfluidizer [Paurec Co., Ltd.], nanomizer [Nanomizer Co., Ltd.], and optimizer [Sugino Machine Co., Ltd.].
The homogeneous valve type high-pressure homogenizer includes Gorin type homogenizer [SMT Co., Ltd.], Lanier type homogenizer [SMT Co., Ltd.], high-pressure homogenizer (Niro Soabi), homogenizer [Sanwa Machinery Co., Ltd.], high-pressure homogenizer [ Izumi Food Machinery Co., Ltd.], ultra-high pressure homogenizer (IKA Co., Ltd.) and the like.
工程Aでは、被乳化処理物に対し、20MPa以上350MPa以下、好ましくは80MPa以上350MPa以下の圧力による乳化処理を施す。
工程Aにおいて、被乳化処理物に対し、20MPa以上の圧力による乳化処理を施すと、加熱処理の工程(即ち、工程B)を経た場合でも、粗大粒子が少ない水中油型乳化組成物が得られる傾向がある。
なお、上限値の350MPaは、市販の装置の限界値である。
圧力の下限は、40MPa以上であることが好ましく、60MPa以上であることがより好ましく、75MPa以上であることが更に好ましく、80MPa以上であることが特に好ましい。
圧力の上限は、245MPa以下であることが好ましい。
圧力は、40MPa以上245MPa以下であってもよく、60MPa以上245MPa以下であってもよく、75MPa以上245MPa以下であってもよく、80MPa以上245MPa以下であってもよい。
In step A, an emulsification treatment is performed on the object to be emulsified with a pressure of 20 MPa to 350 MPa, preferably 80 MPa to 350 MPa.
In step A, when an emulsification treatment is performed on the product to be emulsified with a pressure of 20 MPa or more, an oil-in-water emulsion composition with few coarse particles is obtained even after the heat treatment step (that is, step B). Tend.
The upper limit value of 350 MPa is a limit value of a commercially available apparatus.
The lower limit of the pressure is preferably 40 MPa or more, more preferably 60 MPa or more, further preferably 75 MPa or more, and particularly preferably 80 MPa or more.
The upper limit of the pressure is preferably 245 MPa or less.
The pressure may be 40 MPa or more and 245 MPa or less, 60 MPa or more and 245 MPa or less, 75 MPa or more and 245 MPa or less, or 80 MPa or more and 245 MPa or less.
乳化処理を行う回数(所謂、処理回数)は1回でもよいが、より均一に乳化分散させる観点から、2回以上行うことが好ましく、5回~30回行うことがより好ましい。 The number of times of emulsification treatment (so-called number of treatments) may be one, but from the viewpoint of more uniformly emulsifying and dispersing, it is preferably performed twice or more, more preferably 5 to 30 times.
乳化処理の際の温度は、特に制限されず、例えば、5℃~80℃の範囲に設定できる。 The temperature during the emulsification treatment is not particularly limited and can be set, for example, in the range of 5 ° C to 80 ° C.
以下、工程Aにおける被乳化処理物の各成分について詳細に説明する。 Hereinafter, each component of the to-be-emulsified processed product in step A will be described in detail.
(リン脂質)
被乳化処理物は、リン脂質を含む。
リン脂質は、主に乳化剤として機能する。
リン脂質としては、例えば、天然物由来のリン脂質であるレシチンが挙げられる。
レシチンとは、ホスファチジルコリン自体、又は、少なくともホスファチジルコリンを含む混合物である。
少なくともホスファチジルコリンを含む混合物とは、一般的に、ホスファチジルコリンの他に、ホスファチジルセリン、ホスファチジルエタノールアミン、ホスファチジルイノシトール、N-アシルホスファチジルエタノールアミン、ホスファチジルグリセロール、ホスファチジン酸、リゾホスファチジルコリン、リゾホスファチジン酸、スフィンゴミエリン、スフィンゴエタノールアミン等を含み得る混合物である。
本開示におけるレシチンとしては、ホスファチジルコリンを含み、かつ、ホスファチジルコリンの含有率が80質量%以上のレシチンが好ましい。
(Phospholipid)
The product to be emulsified contains phospholipid.
Phospholipids mainly function as emulsifiers.
Examples of the phospholipid include lecithin, which is a phospholipid derived from a natural product.
Lecithin is phosphatidylcholine itself or a mixture containing at least phosphatidylcholine.
A mixture containing at least phosphatidylcholine generally includes, in addition to phosphatidylcholine, phosphatidylserine, phosphatidylethanolamine, phosphatidylinositol, N-acylphosphatidylethanolamine, phosphatidylglycerol, phosphatidic acid, lysophosphatidylcholine, lysophosphatidic acid, sphingomyelin, It is a mixture that may contain sphingoethanolamine and the like.
The lecithin in the present disclosure is preferably lecithin containing phosphatidylcholine and having a phosphatidylcholine content of 80% by mass or more.
レシチンの例としては、卵黄レシチン、大豆レシチン、綿実レシチン、菜種レシチン、トウモロコシレシチン、水添卵黄レシチン、水添大豆レシチン等が挙げられる。
本開示において、「卵黄レシチン」とは、卵黄由来のレシチンを意味し、「大豆レシチン」とは、大豆由来のレシチンを意味し、「綿実レシチン」とは、綿実由来のレシチンを意味し、「菜種レシチン」とは、菜種由来のレシチンを意味し、「トウモロコシレシチン」とは、トウモロコシ由来のレシチンを意味する。
Examples of lecithin include egg yolk lecithin, soybean lecithin, cottonseed lecithin, rapeseed lecithin, corn lecithin, hydrogenated egg yolk lecithin, hydrogenated soybean lecithin and the like.
In the present disclosure, “yolk lecithin” means lecithin derived from egg yolk, “soy lecithin” means lecithin derived from soybean, and “cotton seed lecithin” means lecithin derived from cotton seed. “Rapeseed lecithin” means rapeseed-derived lecithin, and “corn lecithin” means corn-derived lecithin.
リン脂質は、天然物由来のリン脂質に限定されず、化学合成したリン脂質でもよい。
化学合成したリン脂質の例としては、ホスファチジルコリン(例えば、ジパルミトイルホスファチジルコリン、ジミリストイルホスファチジルコリン、ジステアロイルホスファチジルコリン、及びジオレオイルホスファチジルコリン)、ホスファチジルグリセロール(例えば、ジパルミトイルホスファチジルグリセロール、ジミリストイルホスファチジルグリセロール、ジステアロイルホスファチジルグリセロール、及びジオレオイルホスファチジルグリセロール)、ホスファチジルエタノールアミン(例えば、ジパルミトイルホスファチジルエタノールアミン、ジミリストイルホスファチジルエタノールアミン、ジステアロイルホスファチジルエタノールアミン、及びジオレオイルホスファチジルエタノールアミン)等が挙げられる。
The phospholipid is not limited to a phospholipid derived from a natural product, and may be a chemically synthesized phospholipid.
Examples of chemically synthesized phospholipids include phosphatidylcholines (eg, dipalmitoylphosphatidylcholine, dimyristoylphosphatidylcholine, distearoylphosphatidylcholine, and dioleoylphosphatidylcholine), phosphatidylglycerols (eg, dipalmitoylphosphatidylglycerol, dimyristoylphosphatidylglycerol, distearoyl Phosphatidylglycerol, and dioleoylphosphatidylglycerol), phosphatidylethanolamine (for example, dipalmitoyl phosphatidylethanolamine, dimyristoyl phosphatidylethanolamine, distearoyl phosphatidylethanolamine, and dioleoylphosphatidylethanolamine).
リン脂質としては、例えば、レシチンが好ましく、卵黄レシチン及び大豆レシチンから選ばれる少なくとも1種がより好ましく、卵黄レシチンが更に好ましい。
卵黄レシチンとしては、卵黄レシチンを精製して得られる精製卵黄レシチン又は高度精製卵黄レシチンが好ましい。
As the phospholipid, for example, lecithin is preferable, at least one selected from egg yolk lecithin and soybean lecithin is more preferable, and egg yolk lecithin is still more preferable.
As egg yolk lecithin, purified egg yolk lecithin obtained by purifying egg yolk lecithin or highly purified egg yolk lecithin is preferable.
リン脂質としては、市販品を用いることができる。
リン脂質の市販品の例としては、卵黄レシチンPL-100M〔商品名;精製卵黄レシチン、キューピー(株)〕、卵黄レシチンPC-98N〔商品名;高度精製卵黄レシチン、キューピー(株)〕、卵黄レシチンLipoid E80〔商品名;精製卵黄レシチン、エイチ・ホルスタイン(株)〕等が挙げられる。
A commercially available product can be used as the phospholipid.
Examples of commercially available phospholipids include egg yolk lecithin PL-100M [trade name; purified egg yolk lecithin, Kewpie Co., Ltd.], egg yolk lecithin PC-98N [trade name; highly purified egg yolk lecithin, Kewpie Corporation], egg yolk Examples include lecithin Lipoid E80 [trade name; purified egg yolk lecithin, H. Holstein Co., Ltd.].
被乳化処理物は、リン脂質を1種のみ含んでいてもよく、2種以上含んでいてもよい。 The processed product to be emulsified may contain only one type of phospholipid or two or more types.
(トリグリセリド)
被乳化処理物は、トリグリセリドを含む。
トリグリセリドは、1分子のグリセリンに3分子の脂肪酸がエステル結合したアシルグリセロールである。
(Triglyceride)
The emulsified product contains triglyceride.
Triglycerides are acylglycerols in which three molecules of fatty acid are ester-bonded to one molecule of glycerin.
トリグリセリドとしては、特に制限はなく、中鎖脂肪酸トリグリセリドであってもよく、長鎖脂肪酸トリグリセリドであってもよい。
例えば、入手容易性の観点から、トリグリセリドとしては、長鎖脂肪酸トリグリセリドが好ましい。
The triglyceride is not particularly limited, and may be a medium chain fatty acid triglyceride or a long chain fatty acid triglyceride.
For example, from the viewpoint of easy availability, the triglyceride is preferably a long-chain fatty acid triglyceride.
本開示において、「中鎖脂肪酸トリグリセリド」とは、脂肪酸鎖の平均炭素数が6以上12以下であるトリグリセリドを意味する。
脂肪酸鎖の平均炭素数は、トリグリセリドを構成する脂肪酸(即ち、構成脂肪酸)の炭素数〔例えば、カプリル酸(IUPAC系統名:オクタン酸)であれば8、カプリン酸(IUPAC系統名:デカン酸)であれば10、ラウリン酸(IUPAC系統名:ドデカン酸)であれば12)〕を構成脂肪酸の組成比によって加重平均したものである。構成脂肪酸は、飽和脂肪酸であってもよく、不飽和脂肪酸であってもよく、好ましくは飽和脂肪酸である。中鎖脂肪酸トリグリセリドは、天然物由来であってもよく、合成脂肪酸のトリグリセリドであってもよい。
In the present disclosure, the “medium chain fatty acid triglyceride” means a triglyceride having an average number of carbon atoms in the fatty acid chain of 6 or more and 12 or less.
The average carbon number of the fatty acid chain is the number of carbon atoms of fatty acids (ie, constituent fatty acids) constituting triglycerides [for example, 8 if caprylic acid (IUPAC system name: octanoic acid), capric acid (IUPAC system name: decanoic acid) Is 10 and lauric acid (IUPAC system name: dodecanoic acid is 12)] is weighted average by the composition ratio of the constituent fatty acids. The constituent fatty acid may be a saturated fatty acid or an unsaturated fatty acid, and is preferably a saturated fatty acid. The medium-chain fatty acid triglyceride may be derived from a natural product or a synthetic fatty acid triglyceride.
本開示において、「長鎖脂肪酸トリグリセリド」とは、脂肪酸鎖の平均炭素数が12を超えるトリグリセリドを意味する。
長鎖脂肪酸トリグリセリドとしては、脂肪酸鎖の平均炭素数が14以上24以下の長鎖脂肪酸トリグリセリドが好ましい。
長鎖脂肪酸トリグリセリドの構成脂肪酸は、飽和脂肪酸であってもよく、不飽和脂肪酸であってもよい。
長鎖脂肪酸トリグリセリドは、天然の長鎖脂肪酸トリグリセリドに相当する植物油であってもよく、天然の長鎖脂肪酸トリグリセリドに相当する動物油であってもよく、合成脂肪酸のトリグリセリドであってもよい。
例えば、融点が低く水中油型乳化組成物の乳化安定性が高いという観点から、長鎖脂肪酸トリグリセリドとしては、植物油がより好ましい。
長鎖脂肪酸トリグリセリドとしての植物油は、植物の種子又は堅果由来の油分であり、具体例としては、ダイズ油、綿実油、菜種油、ゴマ油、サフラワー油、コーン油、落花生油、オリーブ油、ヤシ油、シソ油、ヒマシ油、ローズ油等が挙げられる。
これらの中でも、植物油としては、例えば、注射用途への使用実績の観点から、ダイズ油、ゴマ油、及びオリーブ油からなる群より選ばれる少なくとも1種が好ましく、入手容易性の観点から、ダイズ油がより好ましい。
長鎖脂肪酸トリグリセリドとしての動物油は、動物又は魚由来の油脂分であり、具体例としては、牛脂、豚脂、鯨油、魚油等が挙げられる。
In the present disclosure, the “long chain fatty acid triglyceride” means a triglyceride having an average fatty acid chain carbon number of more than 12.
As the long-chain fatty acid triglyceride, a long-chain fatty acid triglyceride having an average fatty acid chain carbon number of 14 to 24 is preferable.
The fatty acid constituting the long-chain fatty acid triglyceride may be a saturated fatty acid or an unsaturated fatty acid.
The long-chain fatty acid triglyceride may be a vegetable oil corresponding to a natural long-chain fatty acid triglyceride, an animal oil corresponding to a natural long-chain fatty acid triglyceride, or a synthetic fatty acid triglyceride.
For example, vegetable oil is more preferable as the long-chain fatty acid triglyceride from the viewpoint that the melting point is low and the emulsion stability of the oil-in-water emulsion composition is high.
Vegetable oils as long-chain fatty acid triglycerides are oils derived from plant seeds or nuts. Specific examples include soybean oil, cottonseed oil, rapeseed oil, sesame oil, safflower oil, corn oil, peanut oil, olive oil, coconut oil, perilla Oil, castor oil, rose oil and the like.
Among these, as the vegetable oil, for example, at least one selected from the group consisting of soybean oil, sesame oil, and olive oil is preferable from the viewpoint of use results for injection applications, and soybean oil is more preferable from the viewpoint of availability. preferable.
Animal oils as long chain fatty acid triglycerides are fats and oils derived from animals or fish, and specific examples include beef tallow, pork tallow, whale oil, fish oil and the like.
トリグリセリドとしては、市販品を用いることができる。
中鎖脂肪酸トリグリセリドの市販品の例としては、Sasol社の「Miglyol(登録商標) 812」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)、「Miglyol(登録商標) 810」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)等、花王(株)の「ココナード(登録商標) RK」(成分名:トリカプリル酸グリセリル)、「ココナード(登録商標) MT」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)、「ココナード(登録商標) MT-N」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)、「ココナード(登録商標) ML」(成分名:トリ(カプリル酸/カプリン酸/ラウリン酸)グリセリル)等、高級アルコール工業(株)の「TCG-M」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)、日清オイリオ(株)の「O.D.O」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)、クローダジャパン(株)の「クロダモル(登録商標) GTCC」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)、日油(株)の「パセナート(登録商標) 810」(成分名:トリ(カプリル酸/カプリン酸)グリセリル)などが挙げられる。
長鎖脂肪酸トリグリセリドの市販品の例としては、カネダ(株)の「日本薬局方 ダイズ油(商品名)」、「日本薬局方 ゴマ油(商品名)」、「日本薬局方 ヒマシ油(商品名)」、「日本薬局方 精製ダイズ油(商品名)」、「日本薬局方 精製オリブ油(商品名)」、「日本薬局方 オリブ油(商品名)」、「日本薬局方 ナタネ油(商品名)」等、Croda社の「Super Refined Soybean(商品名)」、Super Refined Olive(商品名)」、「Super Refined Sesame(商品名)」等、Sigma-Aldrich社の「Olive Oil(商品名)」などが挙げられる。
A commercially available product can be used as the triglyceride.
Examples of commercially available products of medium-chain fatty acid triglycerides include “Miglyol (registered trademark) 812” (component name: tri (caprylic acid / capric acid) glyceryl) and “Miglyol (registered trademark) 810” (component name: “Coconard (registered trademark) RK” (component name: glyceryl tricaprylate), Kao Co., Ltd., “Coconard (registered trademark) MT” (component name: tri (caprylic acid), such as tri (caprylic acid / capric acid) glyceryl) / Glyceryl caprate), “Coconard (registered trademark) MT-N” (component name: tri (caprylic acid / capric acid) glyceryl), “Coconard (registered trademark) ML” (component name: tri (caprylic acid / caprin) Acid / lauric acid) glyceryl), etc., “TCG-M” (component name: tri (caprylic acid / capric acid) (Glyceryl phosphate), “O.D.O” (component name: tri (caprylic acid / capric acid) glyceryl) of Nisshin Oilio Co., Ltd., “Clodamole (registered trademark) GTCC” of Croda Japan Co., Ltd. ( Ingredient name: tri (caprylic acid / capric acid) glyceryl), NOF Corporation "Pasenate (registered trademark) 810" (component name: tri (caprylic acid / capric acid) glyceryl), and the like.
Examples of commercial products of long-chain fatty acid triglycerides include Kaneda Corporation's “Japanese Pharmacopoeia Soybean Oil (trade name)”, “Japanese Pharmacopoeia Sesame Oil (trade name)”, “Japanese Pharmacopoeia Castor Oil (trade name)” ", Japanese Pharmacopoeia Refined Soybean Oil (trade name)", "Japanese Pharmacopoeia Refined Olive Oil (trade name)", "Japanese Pharmacopoeia Olive Oil (trade name)", "Japanese Pharmacopoeia Rapeseed Oil (trade name)""Super Refined Soybean (trade name)", Super Refined Live (trade name) "," Super Refined Same (trade name) "of Croda," Olive Oil "(trade name) of Sigma-Aldrich, etc. Is mentioned.
被乳化処理物は、トリグリセリドを1種のみ含んでいてもよく、2種以上含んでいてもよい。 The processed product to be emulsified may contain only one type of triglyceride or two or more types.
(特定化合物)
被乳化処理物は、フルルビプロフェンアキセチル及びフルルビプロフェンから選ばれる少なくとも1種の化合物(即ち、特定化合物)を含む。
(Specific compounds)
The product to be emulsified contains at least one compound selected from flurbiprofen axetil and flurbiprofen (that is, a specific compound).
フルルビプロフェンアキセチルは、手術後の疼痛及び癌性の疼痛に対し、鎮痛作用を有する非ステロイド性鎮痛剤であり、API Chem社から「Flurbiprofen axetil」の商品名にて販売されている。
フルルビプロフェンは、変形性関節症、肩関節周囲炎、腱炎、腱鞘炎、腱周囲炎、上腕骨上顆炎(例えば、テニス肘)、筋肉痛、並びに、外傷後の腫脹及び疼痛に対し、鎮痛作用及び消炎作用を有する非ステロイド性鎮痛消炎剤であり、東京化成工業(株)から「Flurbiprofen」の商品名にて販売されている。
Flurbiprofen axetil is a non-steroidal analgesic agent that has an analgesic action against post-surgical pain and cancer pain, and is marketed by API Chem under the product name “Flrubiprofen axetil”.
Flurbiprofen is for osteoarthritis, shoulder periarthritis, tendinitis, tendonitis, peritonitis, humerus condylaritis (eg tennis elbow), muscle pain, and post-traumatic swelling and pain It is a non-steroidal analgesic / anti-inflammatory agent having analgesic action and anti-inflammatory action, and is marketed by Tokyo Chemical Industry Co., Ltd. under the trade name “Flurbiprofen”.
被乳化処理物は、フルルビプロフェンアキセチル又はフルルビプロフェンのいずれか一方のみを含んでいてもよく、フルルビプロフェンアキセチル及びフルルビプロフェンの両方を含んでいてもよい。 The emulsified product may contain only one of flurbiprofen axetil or flurbiprofen, or may contain both flurbiprofen axetil and flurbiprofen.
(水)
被乳化処理物は、水を含む。
水は、分散媒として機能する。
水としては、医薬品に使用可能な水であれば、特に制限されない。
水の例としては、精製水、滅菌精製水、注射用水等が挙げられる。
(water)
The emulsified product contains water.
Water functions as a dispersion medium.
Water is not particularly limited as long as it can be used for pharmaceuticals.
Examples of water include purified water, sterilized purified water, and water for injection.
(特定界面活性剤)
被乳化処理物は、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤(即ち、特定界面活性剤)を含んでいてもよい。
特定界面活性剤は、例えば、乳化粒子の安定化の観点から、ポリオキシエチレンソルビタン脂肪酸エステルであることが好ましい。
被乳化処理物が、ポリオキシエチレンソルビタン脂肪酸エステルを含むと、乳化粒子の表面の少なくとも一部において、ポリオキシエチレン鎖が露出する形態が形成されると考えられる。このポリオキシエチレン鎖の存在により、隣接する乳化粒子同士において立体反発が生じ、乳化粒子が安定化されると考えられる。
(Specific surfactant)
The product to be emulsified may contain at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester (that is, a specific surfactant).
The specific surfactant is preferably, for example, a polyoxyethylene sorbitan fatty acid ester from the viewpoint of stabilizing the emulsified particles.
If the product to be emulsified contains polyoxyethylene sorbitan fatty acid ester, it is considered that a form in which the polyoxyethylene chain is exposed is formed on at least a part of the surface of the emulsified particles. It is considered that the presence of this polyoxyethylene chain causes steric repulsion between adjacent emulsified particles and stabilizes the emulsified particles.
ポリオキシエチレンソルビタン脂肪酸エステルとしては、特に制限はない。
ポリオキシエチレンソルビタン脂肪酸エステルは、1分子中のエステル結合が1個である化合物(即ち、モノ脂肪酸エステル)であってもよく、1分子中のエステル結合が2個以上である化合物(即ち、ジ脂肪酸エステル、トリ脂肪酸エステル等)であってもよく、1分子中のエステル結合の数が異なる2種以上の化合物の混合物であってもよい。
There is no restriction | limiting in particular as polyoxyethylene sorbitan fatty acid ester.
The polyoxyethylene sorbitan fatty acid ester may be a compound having one ester bond in one molecule (that is, a mono-fatty acid ester), or a compound having two or more ester bonds in one molecule (that is, a di-fatty acid ester). Fatty acid ester, trifatty acid ester, etc.), or a mixture of two or more compounds having different numbers of ester bonds in one molecule.
ポリオキシエチレンソルビタン脂肪酸エステルにおけるオキシエチレン基の平均付加モル数としては、特に制限はないが、例えば、粗大粒子がより少ない水中油型乳化組成物を得る観点から、5~60が好ましく、10~40がより好ましく、15~25が更に好ましい。 The average number of moles of oxyethylene groups added in the polyoxyethylene sorbitan fatty acid ester is not particularly limited, but is preferably 5 to 60, for example, from the viewpoint of obtaining an oil-in-water emulsion composition with fewer coarse particles. 40 is more preferable, and 15 to 25 is even more preferable.
ポリオキシエチレンソルビタン脂肪酸エステルにおける脂肪酸の炭素数は、特に制限はなく、例えば、8以上が好ましく、12以上がより好ましい。 The carbon number of the fatty acid in the polyoxyethylene sorbitan fatty acid ester is not particularly limited, and is preferably 8 or more, and more preferably 12 or more.
ポリオキシエチレンソルビタン脂肪酸エステルとしては、具体的には、ポリソルベート20(別名:モノラウリン酸ポリオキシエチレンソルビタン)、ポリソルベート40(別名:パルミチン酸ポリオキシエチレンソルビタン)、ポリソルベート60(別名:モノステアリン酸ポリオキシエチレンソルビタン)、ポリソルベート65(別名:トリステアリン酸ポリオキシエチレンソルビタン)、ポリソルベート80(別名:オレイン酸ポリオキシエチレンソルビタン)等のポリソルベートが挙げられる。
これらの中でも、ポリオキシエチレンソルビタン脂肪酸エステルとしては、例えば、注射用途への使用実績の観点からは、ポリソルベート80(別名:オレイン酸ポリオキシエチレンソルビタン)及びポリソルベート20(別名:モノラウリン酸ポリオキシエチレンソルビタン)から選ばれる少なくとも1種が好ましい。
ポリソルベートは、ソルビタン脂肪酸エステルにエチレンオキシドが約20分子縮合したソルビタン脂肪酸エステルのポリオキシエチレンエーテルである。
Specific examples of polyoxyethylene sorbitan fatty acid esters include polysorbate 20 (also known as polyoxyethylene sorbitan monolaurate), polysorbate 40 (also known as polyoxyethylene sorbitan palmitate), polysorbate 60 (also known as polyoxymonostearate). And polysorbates such as polysorbate 65 (also known as polyoxyethylene sorbitan tristearate) and polysorbate 80 (also known as polyoxyethylene sorbitan oleate).
Among these, polyoxyethylene sorbitan fatty acid esters include, for example, polysorbate 80 (also known as polyoxyethylene sorbitan oleate) and polysorbate 20 (also known as polyoxyethylene sorbitan monolaurate) from the viewpoint of actual use in injection applications. ) Is preferred.
Polysorbate is a polyoxyethylene ether of sorbitan fatty acid ester obtained by condensing about 20 molecules of ethylene oxide to sorbitan fatty acid ester.
ポリオキシエチレンソルビタン脂肪酸エステルとしては、市販品を用いることができる。
ポリオキシエチレンソルビタン脂肪酸エステルの市販品の例としては、ポリソルベート80(別名:オレイン酸ポリオキシエチレンソルビタン)である、日光ケミカルズ(株)の「NIKKOL(登録商標) TO-10MV」、日油(株)の「ポリソルベート80」、「ポリソルベート80 GS」、「ポリソルベート80 HX2」、「ノニオンOT-221」、SEPPIC社の「MONTANOX(登録商標) 80」、花王(株)の「レオドール(登録商標) TW-O120V」、第一工業製薬(株)の「ソルゲン(登録商標) TW-80V」、クローダジャパン(株)の「クリレット 4HP」等、ポリソルベート20(別名:モノラウリン酸ポリオキシエチレンソルビタン)である、日光ケミカルズ(株)の「NIKKOL(登録商標) TL-10」、花王(株)の「レオドール(登録商標) TW-L120V」、日油(株)の「ノニオンLT-221」、クローダジャパン(株)の「クリレット 1HP」等が挙げられる。
A commercially available product can be used as the polyoxyethylene sorbitan fatty acid ester.
Examples of commercially available products of polyoxyethylene sorbitan fatty acid ester include “NIKKOL (registered trademark) TO-10MV” of Nikko Chemicals Co., Ltd., which is polysorbate 80 (also known as polyoxyethylene sorbitan oleate), NOF Corporation ) “Polysorbate 80”, “Polysorbate 80 GS”, “Polysorbate 80 HX2”, “Nonion OT-221”, “MONTANOX (registered trademark) 80” of SEPPIC, “Reodol (registered trademark) TW of Kao Corporation -O120V "," Solgen (registered trademark) TW-80V "of Daiichi Kogyo Seiyaku Co., Ltd.," Krillet 4HP "of Croda Japan Co., Ltd., polysorbate 20 (also known as polyoxyethylene sorbitan monolaurate), “NIKKOL (registered trader) of Nikko Chemicals Co., Ltd. Standard) TL-10 ", Kao's" Leodol (registered trademark) TW-L120V ", NOF's" Nonion LT-221 ", Croda Japan's" Krillet 1HP ", etc. .
ソルビタン脂肪酸エステルとしては、特に制限はない。
ソルビタン脂肪酸エステルにおける脂肪酸の炭素数は、特に制限はなく、例えば、8以上が好ましく、12以上がより好ましい。
ソルビタン脂肪酸エステルの例としては、ソルビタンセスキオレイン酸エステル、モノカプリル酸ソルビタン、モノラウリン酸ソルビタン、モノステアリン酸ソルビタン、セスキステアリン酸ソルビタン、トリステアリン酸ソルビタン、イソステアリン酸ソルビタン、セスキイソステアリン酸ソルビタン、オレイン酸ソルビタン、トリオレイン酸ソルビタン等が挙げられる。
There is no restriction | limiting in particular as sorbitan fatty acid ester.
There is no restriction | limiting in particular in carbon number of the fatty acid in sorbitan fatty acid ester, For example, 8 or more are preferable and 12 or more are more preferable.
Examples of sorbitan fatty acid esters include sorbitan sesquioleate, sorbitan monocaprylate, sorbitan monolaurate, sorbitan monostearate, sorbitan sesquistearate, sorbitan tristearate, sorbitan isostearate, sorbitan sesquiisostearate, sorbitan oleate And sorbitan trioleate.
ソルビタン脂肪酸エステルとしては、市販品を用いることができる。
ソルビタン脂肪酸エステルの市販品の例としては、日光ケミカルズ(株)の「NIKKOL(登録商標) SO-15MV」(成分名:ソルビタンセスキオレイン酸エステル)、花王(株)の「レオドール(登録商標) AO-15V」(成分名:ソルビタンセスキオレイン酸エステル)、東京化成工業(株)の「Span 83」(成分名:ソルビタンセスキオレイン酸エステル)、「Span80」(成分名:ソルビタンモノオレイン酸エステル)等、日油(株)の「ノニオンOP-83R NOFA-BLESO」(成分名:ソルビタンセスキオレイン酸エステル)、「NOFABLE(登録商標) 80-582S」(成分名:ソルビタンセスキオレイン酸エステル)等、日清オイリオグループ(株)の「コスモール(登録商標) 82」(成分名:ソルビタンセスキオレイン酸エステル)、第一工業製薬(株)の「ソルゲン(登録商標) 30V」(成分名:ソルビタンセスキオレイン酸エステル)、三洋化成工業(株)の「イオネット(登録商標) S」(成分名:ソルビタン脂肪酸エステル)、ライオン・スペシャリティ・ケミカルズ(株)の「カデナックス(登録商標) SO-80C」(成分名:ソルビタン脂肪酸エステル)などが挙げられる。
A commercial item can be used as sorbitan fatty acid ester.
Examples of commercially available sorbitan fatty acid esters include “NIKKOL (registered trademark) SO-15MV” (component name: sorbitan sesquioleate) from Nikko Chemicals Co., Ltd., “Leodol (registered trademark) AO” from Kao Corporation. -15V "(component name: sorbitan sesquioleate)," Span 83 "(component name: sorbitan sesquioleate) from Tokyo Chemical Industry Co., Ltd.," Span 80 "(component name: sorbitan monooleate) “Nonion OP-83R NOFA-BLESO” (component name: sorbitan sesquioleate), “NOFABLE (registered trademark) 80-582S” (component name: sorbitan sesquioleate), etc. “Cosmall (registered trademark) 82” by Kiyo Oilio Group Co., Ltd. Name: Sorbitan Sesquioleate), “Sorgen (registered trademark) 30V” from Daiichi Kogyo Seiyaku Co., Ltd. (Component name: Sorbitan Sesquioleate), “Ionet” (registered trademark) from Sanyo Chemical Industries, Ltd. S ”(component name: sorbitan fatty acid ester),“ Cadenax (registered trademark) SO-80C ”(component name: sorbitan fatty acid ester) of Lion Specialty Chemicals Co., Ltd., and the like.
被乳化処理物は、特定界面活性剤を含む場合、特定界面活性剤を1種のみ含んでいてもよく、2種以上含んでいてもよい。 When the emulsified product contains a specific surfactant, it may contain only one type of specific surfactant or two or more types.
(多価アルコール)
被乳化処理物は、多価アルコールを含むことが好ましい。
被乳化処理物が多価アルコールを含むと、乳化粒子の安定性が向上する傾向がある。
多価アルコールとしては、特に制限はない。
多価アルコールの例としては、グリセリン、濃グリセリン、プロピレングリコール、1,3-ブチレングリコール、ポリエチレングリコール〔例えば、ポリエチレングリコール300(所謂、マクロゴール300)、ポリエチレングリコール400(所謂、マクロゴール400)、ポリエチレングリコール600(所謂、マクロゴール600)、及びポリエチレングリコール1000(所謂、マクロゴール1000)〕、ジプロピレングリコール、ジエチレングリコール等が挙げられる。
これらの中でも、多価アルコールとしては、例えば、より微細な乳化粒子を得る観点から、濃グリセリンが好ましい。
(Polyhydric alcohol)
The emulsified product preferably contains a polyhydric alcohol.
When the processing object contains a polyhydric alcohol, the stability of the emulsified particles tends to be improved.
There is no restriction | limiting in particular as a polyhydric alcohol.
Examples of the polyhydric alcohol include glycerin, concentrated glycerin, propylene glycol, 1,3-butylene glycol, polyethylene glycol [eg, polyethylene glycol 300 (so-called macrogol 300), polyethylene glycol 400 (so-called macrogol 400), Polyethylene glycol 600 (so-called macrogol 600) and polyethylene glycol 1000 (so-called macrogol 1000)], dipropylene glycol, diethylene glycol and the like.
Among these, as the polyhydric alcohol, for example, concentrated glycerin is preferable from the viewpoint of obtaining finer emulsified particles.
被乳化処理物は、多価アルコールを含む場合、多価アルコールを1種のみ含んでいてもよく、2種以上含んでいてもよい。 When the emulsified processed product contains a polyhydric alcohol, it may contain only one kind of polyhydric alcohol or two or more kinds.
(その他の添加剤)
被乳化処理物は、既述の成分以外に、本開示の製造方法の効果を損なわない範囲において、必要に応じて、その他の添加剤を更に含んでいてもよい。
その他の添加剤としては、例えば、医薬的に許容される添加剤が挙げられる。
例えば、本開示の製造方法によって得られる水中油型乳化組成物を静脈内注射により投与する場合には、静脈内注射に適した添加剤を更に用いることが好ましい。
その他の添加剤の例としては、酸化防止剤〔例えば、アスコルビン酸、D-α-トコフェロール、ブチルヒドロキシアニソール(BHA)、及びジブチルヒドロキシトルエン(BHT)〕、安定化剤(例えば、クエン酸ナトリウム)、防腐剤(例えば、パラオキシ安息香酸メチル及びパラオキシ安息香酸プロピル)等が挙げられる。
但し、その他の添加剤は、これらに限定されない。
(Other additives)
The processed product to be emulsified may further contain other additives as necessary within the range not impairing the effects of the production method of the present disclosure, in addition to the components described above.
Examples of other additives include pharmaceutically acceptable additives.
For example, when the oil-in-water emulsion composition obtained by the production method of the present disclosure is administered by intravenous injection, it is preferable to further use an additive suitable for intravenous injection.
Examples of other additives include antioxidants (eg, ascorbic acid, D-α-tocopherol, butylhydroxyanisole (BHA), and dibutylhydroxytoluene (BHT)), stabilizers (eg, sodium citrate) And preservatives (for example, methyl paraoxybenzoate and propyl paraoxybenzoate).
However, other additives are not limited to these.
被乳化処理物は、その他の添加剤を含む場合、その他の添加剤を1種のみ含んでいてもよく、2種以上含んでいてもよい。 When the emulsified product contains other additives, it may contain only one type of other additive or two or more types.
〔工程B〕
工程Bは、工程Aにて得られた乳化物を少なくとも含む被加熱処理物であって、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤(即ち、特定界面活性剤)を含み、かつ、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルの合計含有率が、被加熱処理物の全質量に対して0.6質量%以上1.8質量%以下である被加熱処理物に対し、加熱処理を施す工程である。
工程Bは、所謂、加熱による滅菌工程である。
[Process B]
Step B is a heat-treated product containing at least the emulsion obtained in Step A, and is at least one surfactant selected from the group consisting of sorbitan fatty acid esters and polyoxyethylene sorbitan fatty acid esters (ie, Specific surfactant), and the total content of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester is 0.6% by mass or more and 1.8% by mass or less with respect to the total mass of the object to be heated. This is a step of performing heat treatment on the object to be heated.
The process B is a so-called sterilization process by heating.
工程Bにおける被加熱処理物は、特定界面活性剤を用いて、工程Aにより得られた乳化物であってもよく、特定界面活性剤を用いずに、工程Aにより得られた乳化物と、特定界面活性剤とを混合して得られた乳化物であってもよい。 The to-be-heated product in Step B may be the emulsion obtained in Step A using a specific surfactant, and the emulsion obtained in Step A without using the specific surfactant, It may be an emulsion obtained by mixing with a specific surfactant.
被加熱処理物中における特定界面活性剤の含有率は、被加熱処理物の全質量に対して、0.6質量%以上1.8質量%以下であり、好ましくは0.8質量%以上1.75質量%以下であり、より好ましくは1.0質量%以上1.5質量%以下であり、更に好ましくは1.25質量%以上1.5質量%以下である。
被加熱処理物中における特定界面活性剤の含有率が上記の範囲内であると、粗大粒子が少ない水中油型乳化組成物が得られる傾向がある。
The content of the specific surfactant in the object to be heated is 0.6% by mass or more and 1.8% by mass or less, preferably 0.8% by mass or more and 1% by mass with respect to the total mass of the object to be heated. It is 0.75 mass% or less, More preferably, it is 1.0 mass% or more and 1.5 mass% or less, More preferably, it is 1.25 mass% or more and 1.5 mass% or less.
There exists a tendency for the oil-in-water-type emulsion composition with few coarse particles to be obtained as the content rate of the specific surfactant in a to-be-heated material is in said range.
工程Bにおいて、被加熱処理物に対し、加熱処理を施す方法としては、特に制限はない。
加熱手段としては、例えば、オートクレーブが好適である。
工程Bでは、被加熱処理物に対し、好ましくは90℃以上140℃以下、より好ましくは100℃以上135℃以下、更に好ましくは105℃以上130℃以下の温度で加熱処理を施すことが好ましい。
加熱処理の温度が90℃以上であると、被加熱処理物をより効果的に滅菌できる。
加熱処理の温度が140℃以下であると、被加熱処理物に含まれる成分の熱による分解がより生じ難い。
ここでいう「温度」は、雰囲気温度を意味する。
加熱時間は、特に制限されず、加熱温度等に応じて、適宜設定できる。
加熱手段として、オートクレーブを用いる場合には、例えば、加熱温度を90℃~140℃に設定し、1分間~60分間加熱することが好ましい。
In step B, there is no particular limitation on the method for performing the heat treatment on the object to be heated.
For example, an autoclave is suitable as the heating means.
In Step B, it is preferable to heat-treat the object to be heated at a temperature of 90 ° C. or higher and 140 ° C. or lower, more preferably 100 ° C. or higher and 135 ° C. or lower, and further preferably 105 ° C. or higher and 130 ° C. or lower.
When the temperature of the heat treatment is 90 ° C. or higher, the object to be heated can be sterilized more effectively.
When the temperature of the heat treatment is 140 ° C. or less, decomposition of the components contained in the heat-treated object due to heat is less likely to occur.
“Temperature” here means the ambient temperature.
The heating time is not particularly limited and can be appropriately set according to the heating temperature and the like.
When using an autoclave as the heating means, for example, it is preferable to set the heating temperature to 90 ° C. to 140 ° C. and heat for 1 minute to 60 minutes.
工程Bにおける被加熱処理物のpHは、6.0以上10.0以下が好ましく、7.0以上10.0以下がより好ましく、7.0以上9.0以下が更に好ましい。
工程Bにおける被加熱処理物のpHが6.0以上であると、水中油型乳化組成物の乳化安定性が向上し、加熱処理後の粗大粒子の生成が抑制される傾向がある。また、リン脂質及びトリグリセリドの分解によって生じる加熱処理後の脂肪酸の生成が抑制される傾向がある。
工程Bにおける被加熱処理物のpHが10.0以下であると、加熱処理後の特定化合物の加水分解物の生成が抑制される傾向がある。
The pH of the object to be heated in step B is preferably 6.0 or more and 10.0 or less, more preferably 7.0 or more and 10.0 or less, and even more preferably 7.0 or more and 9.0 or less.
When the pH of the material to be heated in Step B is 6.0 or more, the emulsion stability of the oil-in-water emulsion composition is improved, and the generation of coarse particles after the heat treatment tends to be suppressed. Moreover, there exists a tendency for the production | generation of the fatty acid after the heat processing produced by decomposition | disassembly of a phospholipid and a triglyceride to be suppressed.
There exists a tendency for the production | generation of the hydrolyzate of the specific compound after heat processing to be suppressed as pH of the to-be-heated material in process B is 10.0 or less.
被加熱処理物のpHが6.0以上10.0以下ではない場合には、工程Bは、被加熱処理物のpHを6.0以上10.0以下に調整する工程(以下、「pH調整工程」ともいう。)を含むことが好ましい。
工程BがpH調整工程を含む場合、pH調整工程では、被加熱処理物のpHを7.0以上10.0以下に調整することがより好ましく、7.0以上9.0以下に調整することが更に好ましい。
被加熱処理物のpHは、例えば、pH調整剤を用いて調整できる。
pH調整剤としては、特に制限はなく、水酸化ナトリウム溶液、塩酸等の公知のpH調整剤を用いることができる。
When the pH of the object to be heated is not 6.0 or more and 10.0 or less, the step B is a step of adjusting the pH of the object to be heated to 6.0 or more and 10.0 or less (hereinafter referred to as “pH adjustment”). It is also preferable to include a “step”.
When step B includes a pH adjustment step, in the pH adjustment step, it is more preferable to adjust the pH of the object to be heated to 7.0 or higher and 10.0 or lower, and to 7.0 or higher and 9.0 or lower. Is more preferable.
The pH of the object to be heated can be adjusted using, for example, a pH adjuster.
There is no restriction | limiting in particular as a pH adjuster, Well-known pH adjusters, such as a sodium hydroxide solution and hydrochloric acid, can be used.
本開示において、被加熱処理物のpHは、被加熱処理物の温度を25℃にして測定する。pHの測定には、pH測定法として一般的な方法を用いることができる。例えば、被加熱処理物のpHは、pHメータにより測定できる。pHメータとしては、(株)堀場製作所のpHメータ(型番:F-73)を好適に用いることができる。但し、pHメータは、これに限定されない。 In the present disclosure, the pH of the object to be heated is measured by setting the temperature of the object to be heated to 25 ° C. For the measurement of pH, a general method can be used as a pH measurement method. For example, the pH of the object to be heated can be measured with a pH meter. As the pH meter, a pH meter (model number: F-73) manufactured by HORIBA, Ltd. can be preferably used. However, the pH meter is not limited to this.
加熱処理は、容器に収容した被加熱処理物に対し、施される。被加熱処理物を収容する容器は、流通製品に用いられる容器であることが好ましい。
被加熱処理物を容器に収容する前に、被加熱処理物に対し、フィルターを用いた濾過を行ってもよい。
フィルターの孔径は、特に制限されず、例えば、50μm以下が好ましく、20μm以下がより好ましく、10μm以下が更に好ましく、5μm以下が特に好ましく、0.8μm以下が最も好ましい。
被加熱処理物を収容した容器は、密閉されることが好ましい。
被加熱処理物の容器への収容及び被加熱処理物を収容した容器の密閉は、窒素雰囲気等の不活性雰囲気下にて行われることが好ましい。
被加熱処理物を収容する容器の種類は、特に制限されず、アンプル、バイアル、シリンジ、バッグ等が挙げられる。
これらの中でも、被加熱処理物を収容する容器の種類としては、例えば、医療現場における取り扱い性の観点からは、バイアルが好ましく、容器の密閉性が高いとの観点からは、アンプルが好ましく、直ぐに投与できるとの観点からは、シリンジ又はバッグが好ましい。
容器の材質は、特に制限されず、ガラスであってもよく、樹脂であってもよい。
容器は、例えば、容器の酸素透過性の観点から、ガラス製容器であることが好ましい。
The heat treatment is performed on the object to be heated contained in the container. The container for storing the object to be heated is preferably a container used for a distribution product.
Prior to storing the object to be heated in the container, the object to be heated may be filtered using a filter.
The pore size of the filter is not particularly limited, and is preferably 50 μm or less, more preferably 20 μm or less, still more preferably 10 μm or less, particularly preferably 5 μm or less, and most preferably 0.8 μm or less.
The container containing the object to be heated is preferably sealed.
The accommodation of the object to be heated and the sealing of the container containing the object to be heated are preferably performed in an inert atmosphere such as a nitrogen atmosphere.
The type of the container that accommodates the object to be heated is not particularly limited, and examples include ampules, vials, syringes, and bags.
Among these, as the type of container for storing the object to be heated, for example, a vial is preferable from the viewpoint of handling at medical sites, and an ampoule is preferable from the viewpoint of high sealing performance of the container. From the viewpoint that administration is possible, a syringe or a bag is preferable.
The material of the container is not particularly limited, and may be glass or resin.
For example, the container is preferably a glass container from the viewpoint of oxygen permeability of the container.
[水中油型乳化組成物]
本開示の製造方法によれば、リン脂質、トリグリセリド、特定化合物、特定界面活性剤、及び水を含む水中油型乳化組成物を得ることができる。
本開示の製造方法によって得られる水中油型乳化組成物は、粗大粒子が少ないため、例えば、注射用(特に、静脈内注射用)として好適である。
水中油型乳化組成物中に粗大粒子が多く存在すると、静脈内注射により投与した場合に、投与後に血管が閉塞する可能性がある。これに対し、本開示の製造方法によって得られる水中油型乳化組成物は、粗大粒子が少ないため、粗大粒子に起因する血管の閉塞が起こり難い。
[Oil-in-water emulsion composition]
According to the production method of the present disclosure, an oil-in-water emulsion composition containing phospholipid, triglyceride, specific compound, specific surfactant, and water can be obtained.
Since the oil-in-water emulsion composition obtained by the production method of the present disclosure has few coarse particles, it is suitable, for example, for injection (particularly for intravenous injection).
When a large amount of coarse particles are present in the oil-in-water emulsion composition, when administered by intravenous injection, blood vessels may be blocked after administration. On the other hand, since the oil-in-water emulsion composition obtained by the production method of the present disclosure has few coarse particles, the blood vessel is less likely to be blocked due to the coarse particles.
以下、本発明を実施例により更に具体的に説明するが、本発明はその主旨を超えない限り、以下の実施例に限定されるものではない。 Hereinafter, the present invention will be described more specifically with reference to examples. However, the present invention is not limited to the following examples as long as the gist thereof is not exceeded.
本実施例において使用した各成分の詳細を以下に示す。 Details of each component used in this example are shown below.
<リン脂質>
・精製卵黄レシチン〔商品名:卵黄レシチンPL-100M、キューピー(株)、表中では「レシチン」と表記〕
<トリグリセリド>
-長鎖脂肪酸トリグリセリド-
・ダイズ油〔商品名:日本薬局方 ダイズ油、カネダ(株)〕
<特定界面活性剤>
-ポリオキシエチレンソルビタン脂肪酸エステル-
・ポリソルベート80〔商品名:NIKKOL(登録商標) TO-10MV、別名:オレイン酸ポリオキシエチレンソルビタン、日光ケミカルズ(株)〕
・ポリソルベート20〔商品名:NIKKOL(登録商標) TL-10、別名:モノラウリン酸ポリオキシエチレンソルビタン、日光ケミカルズ(株)〕
-ソルビタン脂肪酸エステル-
・ソルビタンセスキオレイン酸エステル〔商品名:NIKKOL(登録商標) SO-15MV、日光ケミカルズ(株)〕
<特定界面活性剤以外の界面活性剤>
・ポリオキシエチレン硬化ヒマシ油60〔商品名:NIKKOL(登録商標) HCO-60、日光ケミカルズ(株)〕
<Phospholipid>
・ Purified egg yolk lecithin [Brand name: Egg yolk lecithin PL-100M, Kewpie Co., Ltd., indicated as “lecithin” in the table]
<Triglyceride>
-Long-chain fatty acid triglycerides-
・ Soybean oil [Brand name: Japanese Pharmacopoeia Soybean oil, Kaneda Corporation]
<Specific surfactant>
-Polyoxyethylene sorbitan fatty acid ester-
Polysorbate 80 [trade name: NIKKOL (registered trademark) TO-10MV, also known as: polyoxyethylene sorbitan oleate, Nikko Chemicals Co., Ltd.]
Polysorbate 20 [trade name: NIKKOL (registered trademark) TL-10, also known as: polyoxyethylene sorbitan monolaurate, Nikko Chemicals Co., Ltd.]
-Sorbitan fatty acid ester-
・ Sorbitan sesquioleate [trade name: NIKKOL (registered trademark) SO-15MV, Nikko Chemicals Co., Ltd.]
<Surfactants other than specific surfactants>
・ Polyoxyethylene hydrogenated castor oil 60 [trade name: NIKKOL (registered trademark) HCO-60, Nikko Chemicals Co., Ltd.]
<特定化合物>
・フルルビプロフェンアキセチル〔商品名:Flurbiprofen axetil、ジアステレオマー混合物、API Chem社〕
・フルルビプロフェン〔商品名:Flurbiprofen、東京化成工業(株)〕
<水>
・水〔商品名:注射用水(1000mLプラスチックボトル広口開栓型)、光製薬(株)〕
<Specific compounds>
Flurbiprofen axetil (trade name: Flurbiprofen axtil, diastereomeric mixture, API Chem)
Flurbiprofen [trade name: Flurbiprofen, Tokyo Chemical Industry Co., Ltd.]
<Water>
・ Water [Product name: Water for injection (1000 mL plastic bottle wide-opening type), Hikari Pharmaceutical Co., Ltd.]
<多価アルコール>
・濃グリセリン〔商品名:日本薬局方 濃グリセリン、阪本薬品工業(株)〕
<その他の添加剤>
・オレイン酸〔商品名:オレイン酸、富士フイルム和光純薬(株)〕
・水酸化ナトリウム溶液〔商品名:0.1mol/L 水酸化ナトリウム溶液、富士フイルム和光純薬(株)〕
・塩酸〔商品名:0.1mol/L 塩酸、富士フイルム和光純薬(株)〕
<Polyhydric alcohol>
・ Concentrated glycerin [Brand name: Japanese Pharmacopoeia Concentrated glycerin, Sakamoto Pharmaceutical Co., Ltd.]
<Other additives>
・ Oleic acid [Brand name: Oleic acid, FUJIFILM Wako Pure Chemical Industries, Ltd.]
・ Sodium hydroxide solution [trade name: 0.1 mol / L sodium hydroxide solution, FUJIFILM Wako Pure Chemical Industries, Ltd.]
Hydrochloric acid [trade name: 0.1 mol / L hydrochloric acid, FUJIFILM Wako Pure Chemical Industries, Ltd.]
[水中油型乳化組成物の製造]
<実施例1~実施例5、実施例10及び実施例11>
表1に記載の各成分のうち、特定界面活性剤以外の成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表1に記載の圧力で表1に記載の処理回数、乳化処理を施すことにより、乳化物Xを得た。次いで、得られた乳化物Xと表1に記載の特定界面活性剤とを撹拌混合することにより、乳化物Yを得た。次いで、得られた乳化物Yに対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物YのpHを、表1に記載のとおりに調整した。次いで、pH調整後の乳化物Yに対し、孔径0.8μmのフィルター用いた濾過を行った。次いで、濾過後の乳化物Y 2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物Yに対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表1に示す。
[Production of oil-in-water emulsion composition]
<Examples 1 to 5, Example 10, and Example 11>
Among the components listed in Table 1, components other than the specific surfactant were stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, an emulsion X was obtained by subjecting the obtained crude emulsion to an emulsification treatment by the number of treatments described in Table 1 at a pressure described in Table 1 using a high-pressure homogenizer. Next, an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 1. Next, the pH of the emulsion Y is adjusted as shown in Table 1 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted. Next, the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 μm. Next, 2 mL of the emulsion Y after filtration was stored in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container. Next, the emulsion Y contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [Product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition Got.
Table 1 shows the blending ratio (unit: mass%) of each component used in the preparation of the oil-in-water emulsion composition.
<実施例6~実施例9、実施例12及び実施例13>
表1に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表1に記載の圧力で表1に記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表1に記載のとおりに調整した。次いで、pH調整後の乳化物に対し、孔径0.8μmのフィルター用いた濾過を行った。次いで、濾過後の乳化物2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表1に示す。
<Examples 6 to 9, Example 12 and Example 13>
Each component described in Table 1 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments shown in Table 1 at a pressure shown in Table 1 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as described in Table 1 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Next, the pH-adjusted emulsion was filtered using a filter having a pore size of 0.8 μm. Next, 2 mL of the emulsion after filtration was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal were attached. Used to seal the container. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 1 shows the blending ratio (unit: mass%) of each component used in the preparation of the oil-in-water emulsion composition.
<比較例1>
表2に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表2に記載の圧力で表2に記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表2に記載のとおりに調整した。次いで、pH調整後の乳化物に対し、孔径0.8μmのフィルター用いた濾過を行った。次いで、濾過後の乳化物2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表2に示す。
<Comparative Example 1>
Each component described in Table 2 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments described in Table 2 at a pressure described in Table 2 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as shown in Table 2 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Next, the pH-adjusted emulsion was filtered using a filter having a pore size of 0.8 μm. Next, 2 mL of the emulsion after filtration was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal were attached. Used to seal the container. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 2 shows the blending ratio (unit: mass%) of each component used for preparing the oil-in-water emulsion composition.
<比較例2~比較例7>
表2に記載の各成分のうち、特定界面活性剤以外の成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物に対し、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表2に記載の圧力で表2に記載の処理回数、乳化処理を施すことにより、乳化物Xを得た。次いで、得られた乳化物Xと表2に記載の特定界面活性剤とを撹拌混合することにより、乳化物Yを得た。次いで、得られた乳化物Yに対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物YのpHを、表2に記載のとおりに調整した。次いで、pH調整後の乳化物Yに対し、孔径0.8μmのフィルター用いた濾過を行った。次いで、濾過後の乳化物Y 2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物Yに対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表2に示す。
<Comparative Example 2 to Comparative Example 7>
Among the components listed in Table 2, components other than the specific surfactant were stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Next, the obtained mixture was stirred at 10,000 rpm for a predetermined time using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, an emulsion X was obtained by subjecting the obtained crude emulsion to an emulsification treatment with the number of treatments shown in Table 2 at a pressure shown in Table 2 using a high-pressure homogenizer. Next, an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 2. Next, the pH of the emulsion Y is adjusted as shown in Table 2 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted. Next, the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 μm. Next, 2 mL of the emulsion Y after filtration was stored in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container. Next, the emulsion Y contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [Product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition Got.
Table 2 shows the blending ratio (unit: mass%) of each component used for preparing the oil-in-water emulsion composition.
<比較例8>
表2に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表2に記載の圧力で表2に記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表2に記載のとおりに調整した。次いで、pH調整後の乳化物2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表2に示す。
<Comparative Example 8>
Each component described in Table 2 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments described in Table 2 at a pressure described in Table 2 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as shown in Table 2 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Next, 2 mL of the emulsion after pH adjustment was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 2 shows the blending ratio (unit: mass%) of each component used for preparing the oil-in-water emulsion composition.
[測定及び評価]
1.PFAT5値
実施例1~実施例13及び比較例1~比較例8の各製造方法により得られた水中油型乳化組成物を、水を用いて10倍希釈し、評価用サンプル液を調製した。調製した評価用サンプル液1mLを、個数カウント式粒度分布装置〔製品名:AccuSizer 780AS、日本インテグリス(株)〕に注入し、評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積を、装置の機能により自動にて算出した。そして、下記の式(1)に基づき、評価用サンプル液中に含まれる乳化粒子の合計体積に対する、評価用サンプル液中の直径5μm以上50μm以下の粒子の体積割合(即ち、PFAT5値)(単位:体積%)を算出した。なお、算出した値は、小数点以下3桁目を四捨五入した。
下記の式(1)では、評価用サンプル液中に含まれる乳化粒子の合計体積が、評価用サンプル液中に含まれるトリグリセリドの体積と同程度であると仮定している。なお、評価用サンプル液中に含まれるトリグリセリドの体積(単位:mL)は、下記の式(2)に基づき、算出した。
算出したPFAT5値に基づき、下記の評価基準に従い、粗大粒子の生成抑制効果を評価した。PFAT5値の測定値及び評価結果を表1及び表2に示す。
評価結果が「AA」又は「A」であれば、許容内と判断した。
[Measurement and evaluation]
1. PFAT5 value The oil-in-water emulsion compositions obtained by the production methods of Examples 1 to 13 and Comparative Examples 1 to 8 were diluted 10 times with water to prepare sample solutions for evaluation. 1 mL of the prepared sample solution for evaluation is injected into a number counting type particle size distribution apparatus [product name: AccuSizer 780AS, Nihon Entegris Co., Ltd.], and the total volume of particles having a diameter of 5 μm to 50 μm in 1 mL of the sample solution for evaluation is determined. It was automatically calculated according to the function of the apparatus. Based on the following formula (1), the volume ratio of particles having a diameter of 5 μm or more and 50 μm or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid (that is, PFAT5 value) (unit) : Volume%) was calculated. The calculated value was rounded off to the third decimal place.
In the following formula (1), it is assumed that the total volume of the emulsified particles contained in the evaluation sample liquid is approximately the same as the volume of the triglyceride contained in the evaluation sample liquid. In addition, the volume (unit: mL) of the triglyceride contained in the sample solution for evaluation was calculated based on the following formula (2).
Based on the calculated PFAT5 value, the effect of suppressing the formation of coarse particles was evaluated according to the following evaluation criteria. Tables 1 and 2 show measured values and evaluation results of PFAT5 values.
If the evaluation result was “AA” or “A”, it was judged as acceptable.
PFAT5値(単位:体積%) = [評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積(単位:mL)] ÷ [評価用サンプル液1mLに占めるトリグリセリドの体積(単位:mL)] × 100・・・(1) PFAT5 value (unit: volume%) = [total volume of particles with a diameter of 5 μm to 50 μm in 1 mL of sample liquid for evaluation (unit: mL)] ÷ [volume of triglyceride in 1 mL of sample liquid for evaluation (unit: mL) ] X 100 ... (1)
評価用サンプル液1mLに占めるトリグリセリドの体積(単位:mL) = 水中油型乳化組成物中のトリグリセリドの含有量(単位:g) ÷ トリグリセリドの密度(単位:g/mL) ÷ 10(希釈倍率)・・・(2) Triglyceride volume in 1 mL of sample solution for evaluation (unit: mL) = triglyceride content in oil-in-water emulsion composition (unit: g) ÷ triglyceride density (unit: g / mL) ÷ 10 (dilution ratio) ... (2)
例えば、実施例1の場合、PFAT5値は、以下のようにして算出した。
なお、[評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積(単位:mL)]は、3.361×10-6mLであった。
評価用サンプル液1mLに占めるトリグリセリドの体積(単位:mL) = 容器詰水中油型乳化組成物中のトリグリセリドの含有量(単位:g) ÷ トリグリセリドの密度(単位:g/mL) ÷ 10(希釈倍率) = 0.1÷0.92÷10=1.087×10-2mL
PFAT5値(単位:体積%) = [評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積(単位:mL)] ÷ [評価用サンプル液1mLに占めるトリグリセリドの体積(単位:mL)] × 100 =3.361×10-6÷1.087×10-2×100≒0.031体積%
For example, in the case of Example 1, the PFAT5 value was calculated as follows.
[Total volume of particles having a diameter of 5 μm or more and 50 μm or less in 1 mL of the sample solution for evaluation (unit: mL)] was 3.361 × 10 −6 mL.
Triglyceride volume in 1 mL of sample solution for evaluation (unit: mL) = content of triglyceride in container-packed oil-in-water emulsion composition (unit: g) ÷ density of triglyceride (unit: g / mL) ÷ 10 (dilution Magnification) = 0.1 ÷ 0.92 ÷ 10 = 1.087 × 10 −2 mL
PFAT5 value (unit: volume%) = [total volume of particles having a diameter of 5 μm to 50 μm in 1 mL of evaluation sample liquid (unit: mL)] ÷ [volume of triglyceride in 1 mL of evaluation sample liquid (unit: mL) ] × 100 = 3.361 × 10 −6 ÷ 1.087 × 10 −2 × 100≈0.031% by volume
-評価基準-
AA:PFAT5値が0.03体積%未満である。
A:PFAT5値が0.03体積%以上0.05体積%以下である。
B:PFAT5値が0.05体積%を超えて0.11体積%未満である。
C:PFAT5値が0.11体積%以上である。
-Evaluation criteria-
AA: PFAT5 value is less than 0.03% by volume.
A: The PFAT5 value is 0.03% by volume or more and 0.05% by volume or less.
B: The PFAT5 value is more than 0.05% by volume and less than 0.11% by volume.
C: The PFAT5 value is 0.11% by volume or more.
表1及び表2中、「-」は、該当する成分を配合していないことを意味する。
表1及び表2において、特定界面活性剤の種類の欄における「乳化前」及び「乳化後」との記載は、特定界面活性剤の配合のタイミングを示し、「乳化前」とは、特定界面活性剤を配合した被乳化処理物に対し、乳化処理を行ったことを意味し、「乳化後」とは、乳化処理を行った後に、特定界面活性剤を配合したことを意味する。
表1及び表2に記載の水には、pH調整剤である水酸化ナトリウム溶液及び塩酸に由来する水が含まれる。
In Tables 1 and 2, “-” means that the corresponding component is not blended.
In Tables 1 and 2, the descriptions “before emulsification” and “after emulsification” in the column of the type of specific surfactant indicate the timing of blending the specific surfactant, and “before emulsification” It means that an emulsification treatment has been performed on the product to be emulsified with an activator, and “after emulsification” means that a specific surfactant is blended after the emulsification treatment.
The water described in Table 1 and Table 2 includes water derived from sodium hydroxide solution and hydrochloric acid which are pH adjusters.
表1及び表2に示すように、実施例1~実施例13の製造方法により得られた水中油型乳化組成物は、比較例1~比較例8の製造方法により得られた水中油型乳化組成物と比較して、粗大粒子が少ないことが明らかとなった。
このような結果から、実施例1~実施例13の製造方法は、加熱処理の工程を経た場合でも、粗大粒子が少ない水中油型乳化組成物を製造できる製造方法であることが明らかとなった。
As shown in Tables 1 and 2, the oil-in-water emulsion compositions obtained by the production methods of Examples 1 to 13 are oil-in-water emulsions obtained by the production methods of Comparative Examples 1 to 8. It became clear that there were few coarse particles compared with the composition.
From these results, it was clarified that the production methods of Examples 1 to 13 are production methods capable of producing an oil-in-water emulsion composition with few coarse particles even after the heat treatment step. .
[水中油型乳化組成物の製造]
<実施例14~実施例19>
表3に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表3に記載の圧力で表3に記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表3に記載のとおりに調整した。次いで、pH調整後の乳化物に対し、必要に応じて、孔径0.8μmのフィルター用いた濾過を行った。なお、実施例14では、濾過を行い、実施例15~実施例19では、濾過を行わなかった。次いで、pH調整後の乳化物(pH調整後に濾過を行った実施例14については、濾過後の乳化物)2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表3に示す。
[Production of oil-in-water emulsion composition]
<Examples 14 to 19>
Each component described in Table 3 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments described in Table 3 at a pressure described in Table 3 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as shown in Table 3 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Subsequently, the emulsion after pH adjustment was filtered using a filter having a pore diameter of 0.8 μm as necessary. In Example 14, filtration was performed, and in Examples 15 to 19, filtration was not performed. Next, 2 mL of the emulsion after pH adjustment (for Example 14, which was filtered after pH adjustment) was placed in a glass container [trade name: CS-2, type of container] under a nitrogen atmosphere. : Vial, Fuji Glass Co., Ltd.], and the container was sealed using a rubber stopper and an aluminum seal. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 3 shows the blending ratio (unit: mass%) of each component used in the preparation of the oil-in-water emulsion composition.
<実施例20>
表3に記載の各成分のうち、特定界面活性剤以外の成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表3に記載の圧力で表3に記載の処理回数、乳化処理を施すことにより、乳化物Xを得た。次いで、得られた乳化物Xと表3に記載の特定界面活性剤とを撹拌混合することにより、乳化物Yを得た。次いで、得られた乳化物Yに対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物YのpHを、表3に記載のとおりに調整した。次いで、pH調整後の乳化物Yに対し、孔径0.8μmのフィルター用いた濾過を行った。次いで、濾過後の乳化物Y 2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物Yに対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表3に示す。
<Example 20>
Among the components listed in Table 3, components other than the specific surfactant were stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, an emulsion X was obtained by subjecting the obtained crude emulsion to an emulsification treatment by the number of treatments described in Table 3 at a pressure described in Table 3 using a high-pressure homogenizer. Next, an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 3. Next, the pH of the emulsion Y is adjusted as shown in Table 3 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted. Next, the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 μm. Next, 2 mL of the emulsion Y after filtration was stored in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container. Next, the emulsion Y contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [Product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition Got.
Table 3 shows the blending ratio (unit: mass%) of each component used in the preparation of the oil-in-water emulsion composition.
<比較例9~比較例13>
表3に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表3に記載の圧力で表3に記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表3に記載のとおりに調整した。次いで、pH調整後の乳化物に対し、必要に応じて、孔径0.8μmのフィルター用いた濾過を行った。なお、比較例9、比較例10及び比較例12では、濾過を行い、比較例11及び比較例13では、濾過を行わなかった。次いで、pH調整後の乳化物(pH調整後に濾過を行った比較例9、比較例10及び比較例12については、濾過後の乳化物)2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表3に示す。
<Comparative Example 9 to Comparative Example 13>
Each component described in Table 3 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments described in Table 3 at a pressure described in Table 3 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as shown in Table 3 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Subsequently, the emulsion after pH adjustment was filtered using a filter having a pore diameter of 0.8 μm as necessary. In Comparative Example 9, Comparative Example 10 and Comparative Example 12, filtration was performed, and in Comparative Example 11 and Comparative Example 13, filtration was not performed. Next, 2 mL of the emulsion after pH adjustment (for the Comparative Example 9, Comparative Example 10 and Comparative Example 12 after filtration after pH adjustment) 2 mL of a glass container [trade name : CS-2, container type: vial, Fuji Glass Co., Ltd.], and the container was sealed using a rubber stopper and an aluminum seal. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 3 shows the blending ratio (unit: mass%) of each component used in the preparation of the oil-in-water emulsion composition.
[測定及び評価]
1.PFAT5値
実施例14~実施例20及び比較例9~比較例13の各製造方法により得られた水中油型乳化組成物を、水を用いて10倍希釈し、評価用サンプル液を調製した。調製した評価用サンプル液1mLを、個数カウント式粒度分布装置〔製品名:AccuSizer 780AS、日本インテグリス(株)〕に注入し、評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積を、装置の機能により自動にて算出した。そして、既述の方法と同様にして、評価用サンプル液中に含まれる乳化粒子の合計体積に対する、評価用サンプル液中の直径5μm以上50μm以下の粒子の体積割合(即ち、PFAT5値)(単位:体積%)を算出した。なお、算出した値は、小数点以下3桁目を四捨五入した。
算出したPFAT5値に基づき、上記と同様の評価基準に従い、粗大粒子の生成抑制効果を評価した。PFAT5値の測定値及び評価結果を表3に示す。
評価結果が「AA」又は「A」であれば、許容内と判断した。
[Measurement and evaluation]
1. PFAT5 value The oil-in-water emulsion compositions obtained by the production methods of Examples 14 to 20 and Comparative Examples 9 to 13 were diluted 10-fold with water to prepare sample solutions for evaluation. 1 mL of the prepared sample solution for evaluation is injected into a number-counting particle size distribution apparatus [Product name: AccuSizer 780AS, Nihon Entegris Co., Ltd.] It was automatically calculated according to the function of the apparatus. Then, in the same manner as described above, the volume ratio of particles having a diameter of 5 μm or more and 50 μm or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid (that is, PFAT5 value) (unit) : Volume%) was calculated. The calculated value was rounded off to the third decimal place.
Based on the calculated PFAT5 value, the effect of suppressing the formation of coarse particles was evaluated according to the same evaluation criteria as described above. Table 3 shows the measured values and evaluation results of the PFAT5 value.
If the evaluation result was “AA” or “A”, it was judged as acceptable.
2.特定化合物の加水分解物量
実施例14~実施例20及び比較例9~比較例13の各製造方法により得られた水中油型乳化組成物を、エタノールを用いて100倍希釈し、評価用サンプル液を調製した。調製した評価用サンプル液を用いて、HPLC(高速液体クロマトグラフィー)により、特定化合物であるフルルビプロフェンアキセチルの加水分解物量を求めた。
具体的には、下記のHPLC測定条件において、保持時間4.3分に検出されるフルルビプロフェンアキセチルの加水分解物のピーク、保持時間8.7分及び保持時間8.9分に検出されるフルルビプロフェンアキセチル(所謂、ジアステレオマー混合物)のピーク、並びに、これらのピーク以外に検出される全てのピークのうち、ピーク面積が0.01面積%以上のピークを拾い、拾った全てのピーク面積に対する、保持時間4.3分に検出されるフルルビプロフェンアキセチルの加水分解物のピーク面積の割合(以下、「加水分解物のピーク面積割合」という。単位:面積%)を自動算出した。なお、算出した値は、小数点以下3桁目を四捨五入した。
算出したフルルビプロフェンアキセチルの加水分解物のピーク面積割合を、フルルビプロフェンアキセチルの加水分解物量とし、下記の評価基準に従って、特定化合物の安定性を評価した。
フルルビプロフェンアキセチルの加水分解物量の測定値及び評価結果を表3に示す。
評価結果が「AA」又は「A」であれば、許容内と判断した。
2. Hydrolyzate Amount of Specific Compound The oil-in-water emulsion composition obtained by each of the production methods of Examples 14 to 20 and Comparative Examples 9 to 13 was diluted 100 times with ethanol, and an evaluation sample solution Was prepared. Using the prepared sample liquid for evaluation, the amount of hydrolyzate of flurbiprofen axetyl, which is a specific compound, was determined by HPLC (high performance liquid chromatography).
Specifically, under the following HPLC measurement conditions, the peak of flurbiprofen axetyl hydrolyzate detected at a retention time of 4.3 minutes, detection at a retention time of 8.7 minutes and a retention time of 8.9 minutes Among the peaks of flurbiprofen axetil (so-called diastereomeric mixture) to be detected and all peaks detected other than these peaks, the peaks with a peak area of 0.01 area% or more were picked up and picked up. The ratio of the peak area of the hydrolyzate of flurbiprofen axetil detected at a retention time of 4.3 minutes with respect to all the peak areas (hereinafter referred to as “the peak area ratio of the hydrolyzate”. Unit: area% ) Was automatically calculated. The calculated value was rounded off to the third decimal place.
The calculated peak area ratio of the flurbiprofen axetil hydrolyzate was defined as the amount of flurbiprofen axetil hydrolyzate, and the stability of the specific compound was evaluated according to the following evaluation criteria.
Table 3 shows measured values and evaluation results of the amount of flurbiprofen axetyl hydrolyzate.
If the evaluation result was “AA” or “A”, it was judged as acceptable.
(HPLC測定条件)
検出器:UV検出器(検出波長:254nm)
カラム:TSKgel ODS-100Z〔製品名、平均粒子径:5μm、カラムサイズ:4.6mm(内径)×150mm(長さ)、東ソー(株)〕
カラム温度:40℃
溶離液A:0.1質量%リン酸水溶液
溶離液B:0.1質量%リン酸アセトニトリル溶液
グラジェント条件(溶離液Bの体積分率、時間):(60%、0.1分)→(80%、10分)→(100%、10.1分~15.0分)→(60%、15.1分)→30分で停止
流速:1mL/分
注入量:10μL
(HPLC measurement conditions)
Detector: UV detector (detection wavelength: 254 nm)
Column: TSKgel ODS-100Z [Product name, average particle size: 5 μm, column size: 4.6 mm (inner diameter) × 150 mm (length), Tosoh Corporation]
Column temperature: 40 ° C
Eluent A: 0.1% by mass phosphoric acid aqueous solution Eluent B: 0.1% by mass phosphoric acid acetonitrile solution Gradient conditions (volume fraction of eluent B, time): (60%, 0.1 min) → (80%, 10 minutes) → (100%, 10.1 to 15.0 minutes) → (60%, 15.1 minutes) → Stop at 30 minutes Flow rate: 1 mL / min Injection volume: 10 μL
-評価基準-
AA:加水分解物のピーク面積割合が0.5面積%未満である。
A:加水分解物のピーク面積割合が0.5面積%以上1.0面積%未満である。
B:加水分解物のピーク面積割合が1.0面積%以上2.0面積%未満である。
C:加水分解物のピーク面積割合が2.0面積%以上である。
-Evaluation criteria-
AA: The peak area ratio of the hydrolyzate is less than 0.5 area%.
A: The peak area ratio of the hydrolyzate is 0.5 area% or more and less than 1.0 area%.
B: The peak area ratio of the hydrolyzate is 1.0 area% or more and less than 2.0 area%.
C: The peak area ratio of the hydrolyzate is 2.0 area% or more.
表3中、「-」は、該当する成分を配合していないことを意味する。
表3において、特定界面活性剤の種類の欄における「乳化前」及び「乳化後」との記載は、特定界面活性剤の配合のタイミングを示し、「乳化前」とは、特定界面活性剤を配合した被乳化処理物に対し、乳化処理を行ったことを意味し、「乳化後」とは、乳化処理を行った後に、特定界面活性剤を配合したことを意味する。また、その他の添加剤の種類の欄における「乳化前」及び「乳化後」との記載についても同様である。
表3に記載の水には、pH調整剤である水酸化ナトリウム溶液及び塩酸に由来する水が含まれる。
In Table 3, “-” means that the corresponding component is not blended.
In Table 3, the descriptions “before emulsification” and “after emulsification” in the column of the type of specific surfactant indicate the timing of blending the specific surfactant, and “before emulsification” It means that an emulsification treatment was performed on the blended product to be emulsified, and “after emulsification” means that a specific surfactant was blended after the emulsification treatment. The same applies to the descriptions of “before emulsification” and “after emulsification” in the column of other additive types.
The water described in Table 3 includes water derived from sodium hydroxide solution and hydrochloric acid which are pH adjusters.
表3に示すように、実施例14~実施例20の製造方法により得られた水中油型乳化組成物は、比較例9~比較例13の製造方法により得られた水中油型乳化組成物と比較して、粗大粒子が少なかった。
このような結果から、実施例14~実施例20の製造方法は、加熱処理の工程を経た場合でも、粗大粒子が少ない水中油型乳化組成物を製造できる製造方法であることが明らかとなった。
As shown in Table 3, the oil-in-water emulsion compositions obtained by the production methods of Examples 14 to 20 are the same as the oil-in-water emulsion compositions obtained by the production methods of Comparative Examples 9 to 13. In comparison, there were few coarse particles.
From these results, it was clarified that the production methods of Examples 14 to 20 were production methods capable of producing an oil-in-water emulsion composition with few coarse particles even after the heat treatment step. .
また、実施例14~実施例20の製造方法により得られた水中油型乳化組成物は、特定化合物であるフルルビプロフェンアキセチルの加水分解物が少なかった。
このような結果から、実施例14~実施例20の製造方法は、特定化合物が加水分解し難く、特定化合物の安定性に優れる製造方法であることが明らかとなった。
In addition, the oil-in-water emulsion compositions obtained by the production methods of Examples 14 to 20 had few hydrolysates of flurbiprofen axetyl, which is a specific compound.
From these results, it was revealed that the production methods of Examples 14 to 20 are production methods in which the specific compound is difficult to hydrolyze and the specific compound is excellent in stability.
[水中油型乳化組成物の製造]
<実施例21~実施例24及び実施例26~実施例29>
表4に記載の各成分のうち、特定界面活性剤以外の成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表4に記載の圧力で表4記載の処理回数、乳化処理を施すことにより、乳化物Xを得た。次いで、得られた乳化物Xと表4に記載の特定界面活性剤とを撹拌混合することにより、乳化物Yを得た。次いで、得られた乳化物Yに対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物YのpHを、表4に記載のとおりに調整した。次いで、pH調整後の乳化物Yに対し、孔径0.8μmのフィルター用いた濾過を行った。次いで、濾過後の乳化物Y 2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物Yに対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表4に示す。
[Production of oil-in-water emulsion composition]
<Examples 21 to 24 and Examples 26 to 29>
Among the components listed in Table 4, the components other than the specific surfactant were stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, an emulsion X was obtained by subjecting the obtained crude emulsion to an emulsification treatment at the pressure shown in Table 4 and the number of treatments shown in Table 4, using a high-pressure homogenizer. Next, an emulsion Y was obtained by stirring and mixing the obtained emulsion X and the specific surfactant described in Table 4. Next, the pH of the emulsion Y is adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion Y. It was adjusted. Next, the emulsion Y after pH adjustment was filtered using a filter having a pore diameter of 0.8 μm. Next, 2 mL of the emulsion Y after filtration was stored in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container. Next, the emulsion Y contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [Product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition Got.
Table 4 shows the blending ratio (unit: mass%) of each component used for preparing the oil-in-water emulsion composition.
<実施例25、実施例30及び実施例31>
表4に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表4に記載の圧力で表4記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表4に記載のとおりに調整した。次いで、pH調整後の乳化物に対し、必要に応じて、孔径0.8μmのフィルター用いた濾過を行った。なお、実施例25では、濾過を行い、実施例30及び実施例31では、濾過を行わなかった。次いで、pH調整後の乳化物(pH調整後に濾過を行った実施例25については、濾過後の乳化物)2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表4に示す。
<Example 25, Example 30 and Example 31>
Each component described in Table 4 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments shown in Table 4 at a pressure shown in Table 4 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Subsequently, the emulsion after pH adjustment was filtered using a filter having a pore diameter of 0.8 μm as necessary. In Example 25, filtration was performed, and in Examples 30 and 31, filtration was not performed. Next, 2 mL of the emulsion after pH adjustment (for Example 25 after filtration after pH adjustment, the emulsion after filtration) was placed in a glass container [trade name: CS-2, type of container under a nitrogen atmosphere] : Vial, Fuji Glass Co., Ltd.], and the container was sealed using a rubber stopper and an aluminum seal. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 4 shows the blending ratio (unit: mass%) of each component used for preparing the oil-in-water emulsion composition.
<比較例14>
表4に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表4に記載の圧力で表4記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表4に記載のとおりに調整した。次いで、pH調整後の乳化物に対し、孔径0.8μmのフィルター用いた濾過を行った。次いで、濾過後の乳化物2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表4に示す。
<Comparative example 14>
Each component described in Table 4 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments shown in Table 4 at a pressure shown in Table 4 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Next, the pH-adjusted emulsion was filtered using a filter having a pore size of 0.8 μm. Next, 2 mL of the emulsion after filtration was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal were attached. Used to seal the container. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 4 shows the blending ratio (unit: mass%) of each component used for preparing the oil-in-water emulsion composition.
<比較例15>
表4に記載の各成分を室温(即ち、25℃)にて撹拌混合し、混合物を得た。次いで、得られた混合物を、ホモジナイザー〔製品名:ULTRA-TURRAX(登録商標)、IKA社〕を用いて、10,000rpmにて所定時間撹拌し、粗乳化物を得た。次いで、得られた粗乳化物に対し、高圧ホモジナイザーを用いて、表4に記載の圧力で表4に記載の処理回数、乳化処理を施すことにより、乳化物を得た。次いで、得られた乳化物に対し、pH調整剤である水酸化ナトリウム溶液及び塩酸の少なくとも一方と、水と、を添加することにより、乳化物のpHを、表4に記載のとおりに調整した。次いで、pH調整後の乳化物2mLを、窒素雰囲気下、ガラス製の容器〔商品名:CS-2、容器の種類:バイアル、不二硝子(株)〕に収容した後、ゴム栓及びアルミシールを用いて、容器を密閉した。次いで、容器に収容された乳化物に対し、オートクレーブ〔製品名:オートクレーブSP200、ヤマト科学(株)〕を用いて、121℃で15分間の加熱処理を施すことにより、水中油型乳化組成物を得た。
水中油型乳化組成物の作製に使用した各成分の配合率(単位:質量%)については、表4に示す。
<Comparative Example 15>
Each component described in Table 4 was stirred and mixed at room temperature (ie, 25 ° C.) to obtain a mixture. Subsequently, the obtained mixture was stirred for a predetermined time at 10,000 rpm using a homogenizer [product name: ULTRA-TURRAX (registered trademark), IKA Corporation] to obtain a crude emulsion. Next, the obtained crude emulsion was subjected to an emulsification treatment with the number of treatments described in Table 4 at a pressure described in Table 4 using a high-pressure homogenizer to obtain an emulsion. Next, the pH of the emulsion was adjusted as shown in Table 4 by adding at least one of a sodium hydroxide solution and hydrochloric acid, which are pH adjusting agents, and water to the obtained emulsion. . Next, 2 mL of the emulsion after pH adjustment was placed in a glass container [trade name: CS-2, container type: vial, Fuji Glass Co., Ltd.] under a nitrogen atmosphere, and then a rubber stopper and an aluminum seal Was used to seal the container. Next, the emulsion contained in the container is subjected to a heat treatment at 121 ° C. for 15 minutes using an autoclave [product name: Autoclave SP200, Yamato Scientific Co., Ltd.], whereby an oil-in-water emulsion composition is obtained. Obtained.
Table 4 shows the blending ratio (unit: mass%) of each component used for preparing the oil-in-water emulsion composition.
[測定及び評価]
実施例21~実施例31、比較例14及び比較例15の製造方法により得られた水中油型乳化組成物に対し、雰囲気温度60℃の環境下にて2週間保存する苛酷試験を行った。この苛酷試験後の水中油型乳化組成物を用いて、下記の測定及び評価を行った。
[Measurement and evaluation]
The oil-in-water emulsion compositions obtained by the production methods of Examples 21 to 31, Comparative Example 14 and Comparative Example 15 were subjected to a severe test in which they were stored for 2 weeks in an environment at an ambient temperature of 60 ° C. The following measurement and evaluation were performed using this oil-in-water emulsion composition after the severe test.
1.PFAT5値
苛酷試験後の水中油型乳化組成物を、水を用いて10倍希釈し、評価用サンプル液を調製した。調製した評価用サンプル液1mLを、個数カウント式粒度分布装置〔製品名:AccuSizer 780AS、日本インテグリス(株)〕に注入し、評価用サンプル液1mLに占める直径5μm以上50μm以下の粒子の総体積を、装置の機能により自動にて算出した。そして、既述の方法と同様にして、評価用サンプル液中に含まれる乳化粒子の合計体積に対する、評価用サンプル液中の直径5μm以上50μm以下の粒子の体積割合(即ち、PFAT5値)(単位:体積%)を算出した。なお、算出した値は、小数点以下3桁目を四捨五入した。
算出したPFAT5値に基づき、上記と同様の評価基準に従い、粗大粒子の生成抑制効果を評価した。PFAT5値の測定値及び評価結果を表4に示す。
評価結果が「AA」又は「A」であれば、許容内と判断した。
1. PFAT5 value The oil-in-water emulsion composition after the severe test was diluted 10 times with water to prepare a sample solution for evaluation. 1 mL of the prepared sample solution for evaluation is injected into a number-counting particle size distribution apparatus [Product name: AccuSizer 780AS, Nihon Entegris Co., Ltd.] It was automatically calculated according to the function of the apparatus. Then, in the same manner as described above, the volume ratio of particles having a diameter of 5 μm or more and 50 μm or less in the evaluation sample liquid to the total volume of the emulsified particles contained in the evaluation sample liquid (that is, PFAT5 value) (unit) : Volume%) was calculated. The calculated value was rounded off to the third decimal place.
Based on the calculated PFAT5 value, the effect of suppressing the formation of coarse particles was evaluated according to the same evaluation criteria as described above. Table 4 shows the measured values and evaluation results of the PFAT5 value.
If the evaluation result was “AA” or “A”, it was judged as acceptable.
2.特定化合物の加水分解物量
苛酷試験後の水中油型乳化組成物を、エタノールを用いて100倍希釈し、評価用サンプル液を調製した。調製した評価用サンプル液を用いて、既述の方法と同様にして、フルルビプロフェンアキセチルの加水分解物のピーク面積割合を算出した。なお、算出した値は、小数点以下2桁目を四捨五入した。
算出したフルルビプロフェンアキセチルの加水分解物のピーク面積割合を、フルルビプロフェンアキセチルの加水分解物量とし、下記の評価基準に従って、特定化合物の安定性を評価した。
フルルビプロフェンアキセチルの加水分解物量の測定値及び評価結果を表4に示す。
評価結果が「AA」又は「A」であれば、許容内と判断した。
2. Hydrolyzate Amount of Specific Compound An oil-in-water emulsion composition after a severe test was diluted 100 times with ethanol to prepare a sample solution for evaluation. Using the prepared sample liquid for evaluation, the peak area ratio of the hydrolyzate of flurbiprofen axetil was calculated in the same manner as described above. The calculated value was rounded off to the second decimal place.
The calculated peak area ratio of the flurbiprofen axetil hydrolyzate was defined as the amount of flurbiprofen axetil hydrolyzate, and the stability of the specific compound was evaluated according to the following evaluation criteria.
Table 4 shows measured values and evaluation results of the amount of flurbiprofen axetyl hydrolyzate.
If the evaluation result was “AA” or “A”, it was judged as acceptable.
-評価基準-
AA:フルルビプロフェンアキセチルの加水分解物量が3.5面積%未満である。
A:フルルビプロフェンアキセチルの加水分解物量が3.5面積%以上4.5面積%未満である。
B:フルルビプロフェンアキセチルの加水分解物量が4.5面積%以上である。
-Evaluation criteria-
AA: The amount of flurbiprofen axetil hydrolyzate is less than 3.5 area%.
A: The amount of hydrolyzate of flurbiprofen axetil is 3.5 area% or more and less than 4.5 area%.
B: The amount of hydrolyzate of flurbiprofen axetil is 4.5 area% or more.
3.脂肪酸量
苛酷試験後の水中油型乳化組成物を、エタノールを用いて100倍希釈し、評価用サンプル液を調製した。調製した評価用サンプル液を用いて、LC-MS(液体クロマトグラフィー-質量分析法)により、脂肪酸であるパルミチン酸量を求めた。
具体的には、下記のLC-MS測定条件において、保持時間14.8分に検出されるm/z(所謂、質量電荷比)=225.20のピーク面積から、検量線を用いて、パルミチン酸量を求めた。なお、算出した値は、小数点以下4桁目を四捨五入した。
得られたパルミチン酸量に基づき、下記の評価基準に従って、リン脂質及びトリグリセリドの安定性を評価した。
パルミチン酸量の測定値及び評価結果を表4に示す。
評価結果が「AA」又は「A」であれば、許容内と判断した。
3. Fatty acid amount The oil-in-water emulsion composition after the severe test was diluted 100 times with ethanol to prepare a sample solution for evaluation. Using the prepared sample solution for evaluation, the amount of palmitic acid, which is a fatty acid, was determined by LC-MS (liquid chromatography-mass spectrometry).
Specifically, from the peak area of m / z (so-called mass-to-charge ratio) = 225.20 detected at a retention time of 14.8 minutes under the following LC-MS measurement conditions, using a calibration curve, palmitin The amount of acid was determined. The calculated value was rounded off to the fourth decimal place.
Based on the amount of palmitic acid obtained, the stability of phospholipids and triglycerides was evaluated according to the following evaluation criteria.
Table 4 shows measured values and evaluation results of the amount of palmitic acid.
If the evaluation result was “AA” or “A”, it was judged as acceptable.
(LC-MS測定条件)
イオン化法:ESI(ElectroSpray Ionization)及びAPCI(Atomic Pressure Chemical Ionization)
検出法:m/z=225.20(Nega、モード:SIM(Selected Ion Monitoring)
カラム:Capcellpak(登録商標) C18〔製品名、平均粒子径:3μm、カラムサイズ:2.0mm(内径)×150mm(長さ)、(株)資生堂〕
カラム温度:40℃
溶離液A:10mM酢酸アンモニウム水溶液
溶離液B:10mM酢酸アンモニウムメタノール溶液
グラジェント条件(溶離液Bの体積分率、時間):(75%、0.1分)→(91%、16分)→(100%、16.1分~21.0分)→(75%、21.1分)→36分で停止
流速:0.2mL/分
注入量:4μL
(LC-MS measurement conditions)
Ionization methods: ESI (ElectroSpray Ionization) and APCI (Atomic Pressure Chemical Ionization)
Detection method: m / z = 225.20 (Nega, mode: SIM (Selected Ion Monitoring)
Column: Capcellpak (registered trademark) C18 [Product name, average particle size: 3 μm, column size: 2.0 mm (inner diameter) × 150 mm (length), Shiseido Co., Ltd.]
Column temperature: 40 ° C
Eluent A: 10 mM ammonium acetate aqueous solution Eluent B: 10 mM ammonium acetate methanol solution Gradient conditions (volume fraction of eluent B, time): (75%, 0.1 min) → (91%, 16 min) → (100%, 16.1 to 21.0 minutes) → (75%, 21.1 minutes) → Stop at 36 minutes Flow rate: 0.2 mL / min Injection volume: 4 μL
-評価基準-
AA:パルミチン酸量が0.03質量%未満である。
A:パルミチン酸量が0.03質量%以上0.05質量%未満である。
B:パルミチン酸量が0.05質量%以上である。
-Evaluation criteria-
AA: The amount of palmitic acid is less than 0.03% by mass.
A: The amount of palmitic acid is 0.03% by mass or more and less than 0.05% by mass.
B: The amount of palmitic acid is 0.05% by mass or more.
表4中、「-」は、該当する成分を配合していないことを意味する。
表4において、特定界面活性剤の種類の欄における「乳化前」及び「乳化後」との記載は、特定界面活性剤の配合のタイミングを示し、「乳化前」とは、特定界面活性剤を配合した被乳化処理物に対し、乳化処理を行ったことを意味し、「乳化後」とは、乳化処理を行った後に、特定界面活性剤を配合したことを意味する。
表4に記載の水には、pH調整剤である水酸化ナトリウム溶液及び塩酸に由来する水が含まれる。
In Table 4, “-” means that the corresponding component is not blended.
In Table 4, the descriptions “before emulsification” and “after emulsification” in the column of the type of specific surfactant indicate the timing of blending the specific surfactant, and “before emulsification” It means that an emulsification treatment was performed on the blended product to be emulsified, and “after emulsification” means that a specific surfactant was blended after the emulsification treatment.
The water described in Table 4 includes water derived from sodium hydroxide solution and hydrochloric acid which are pH adjusters.
表4に示すように、実施例21~実施例31の製造方法により製造した後、苛酷な環境下にて保存した水中油型乳化組成物は、比較例14及び比較例15の製造方法により得られた水中油型乳化組成物と比較して、粗大粒子が少なかった。
このような結果から、実施例21~実施例31の製造方法によれば、加熱処理の工程を経た場合でも、粗大粒子が少ない水中油型乳化組成物を製造できることが明らかとなった。また、実施例21~実施例31の製造方法により得られる水中油型乳化組成物は、苛酷な環境下にて保存した場合でも、粗大粒子が生成し難いことが明らかとなった。
As shown in Table 4, the oil-in-water emulsion compositions produced by the production methods of Examples 21 to 31 and then stored in a harsh environment were obtained by the production methods of Comparative Examples 14 and 15. There were few coarse particles compared with the obtained oil-in-water emulsion composition.
From these results, it became clear that according to the production methods of Examples 21 to 31, an oil-in-water emulsion composition with few coarse particles can be produced even after the heat treatment step. Further, it was revealed that the oil-in-water emulsion compositions obtained by the production methods of Examples 21 to 31 are difficult to produce coarse particles even when stored in a harsh environment.
また、実施例21~実施例31の製造方法により製造した後、苛酷な環境下にて保存した水中油型乳化組成物は、特定化合物であるフルルビプロフェンアキセチルの加水分解物が少なかった。
このような結果から、実施例21~実施例31の製造方法は、加熱処理の工程において、特定化合物が加水分解し難く、特定化合物の安定性に優れる製造方法であることが明らかとなった。また、実施例21~実施例31の製造方法により得られる水中油型乳化組成物は、苛酷な環境下にて保存した場合でも、特定化合物が加水分解し難く、特定化合物の安定性に優れることが明らかとなった。
In addition, the oil-in-water emulsion composition produced by the production method of Examples 21 to 31 and stored in a harsh environment had a small amount of the hydrolyzate of flurbiprofen axetyl, which is a specific compound. .
From these results, it was revealed that the production methods of Examples 21 to 31 are production methods in which the specific compound is difficult to hydrolyze in the heat treatment step and is excellent in stability of the specific compound. In addition, the oil-in-water emulsion compositions obtained by the production methods of Examples 21 to 31 are resistant to hydrolysis of the specific compound even when stored in a harsh environment, and are excellent in stability of the specific compound. Became clear.
また、実施例21~実施例31の製造方法により製造した後、苛酷な環境下にて保存した水中油型乳化組成物は、リン脂質及びトリグリセリドの分解により生成する脂肪酸であるパルミチン酸が少なかった。
このような結果から、実施例21~実施例31の製造方法は、加熱処理の工程において、リン脂質及びトリグリセリドの分解が生じ難く、リン脂質及びトリグリセリドの安定性に優れる製造方法であることが明らかとなった。また、実施例21~実施例31の製造方法により得られる水中油型乳化組成物は、苛酷な環境下にて保存した場合でも、リン脂質及びトリグリセリドに由来する脂肪酸が生成し難く、リン脂質及びトリグリセリドの安定性に優れることが明らかとなった。
In addition, the oil-in-water emulsion composition produced by the production method of Examples 21 to 31 and stored in a harsh environment had a small amount of palmitic acid, which is a fatty acid produced by the decomposition of phospholipids and triglycerides. .
From these results, it is clear that the production methods of Examples 21 to 31 are production methods in which phospholipids and triglycerides are hardly decomposed in the heat treatment step and phospholipids and triglycerides are excellent in stability. It became. In addition, the oil-in-water emulsion compositions obtained by the production methods of Examples 21 to 31 hardly generate fatty acids derived from phospholipids and triglycerides even when stored in a harsh environment. It was revealed that the triglyceride was excellent in stability.
2018年5月31日に出願された日本国特許出願2018-105305号の開示は、その全体が参照により本明細書に取り込まれる。
本明細書に記載された全ての文献、特許出願、及び技術規格は、個々の文献、特許出願、及び技術規格が参照により取り込まれることが具体的に、かつ、個々に記された場合と同程度に、本明細書中に参照により取り込まれる。
The disclosure of Japanese Patent Application No. 2018-105305 filed on May 31, 2018 is incorporated herein by reference in its entirety.
All documents, patent applications, and technical standards described in this specification are specifically and individually incorporated by reference as if individual documents, patent applications, and technical standards were incorporated by reference. To the extent it is incorporated herein by reference.
Claims (9)
工程Aにて得られた乳化物を少なくとも含む被加熱処理物であって、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルからなる群より選ばれる少なくとも1種の界面活性剤を含み、かつ、ソルビタン脂肪酸エステル及びポリオキシエチレンソルビタン脂肪酸エステルの合計含有率が、被加熱処理物の全質量に対して0.6質量%以上1.8質量%以下である被加熱処理物に対し、加熱処理を施す工程Bと、
を有する水中油型乳化組成物の製造方法。 An emulsification treatment with a pressure of 20 MPa or more and 350 MPa or less is performed on an article to be emulsified containing phospholipid, triglyceride, at least one compound selected from flurbiprofen axetyl and flurbiprofen, and water. Step A to obtain an emulsion by applying,
A to-be-heated product containing at least the emulsion obtained in step A, comprising at least one surfactant selected from the group consisting of sorbitan fatty acid ester and polyoxyethylene sorbitan fatty acid ester, and sorbitan fatty acid The step of subjecting the object to be heated, in which the total content of the ester and the polyoxyethylene sorbitan fatty acid ester is 0.6% by mass or more and 1.8% by mass or less based on the total mass of the object to be heated. B and
The manufacturing method of the oil-in-water emulsion composition which has NO.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018-105305 | 2018-05-31 | ||
| JP2018105305 | 2018-05-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019230207A1 true WO2019230207A1 (en) | 2019-12-05 |
Family
ID=68696945
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/015691 Ceased WO2019230207A1 (en) | 2018-05-31 | 2019-04-10 | Method for producing oil-in-water type emulsion composition |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2019230207A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000212067A (en) * | 1999-01-19 | 2000-08-02 | Towa Yakuhin Kk | Emulsion preparation including medicinal substance |
| CN102697724A (en) * | 2012-06-07 | 2012-10-03 | 沈阳药科大学 | Clopidogrel and salt submicron emulsion injection thereof as well as preparation method of same |
| CN104706575A (en) * | 2013-12-16 | 2015-06-17 | 天津迈迪瑞康生物医药科技有限公司 | Flurbiprofen axetil fat emulsion concentrate and preparation method and use thereof |
-
2019
- 2019-04-10 WO PCT/JP2019/015691 patent/WO2019230207A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000212067A (en) * | 1999-01-19 | 2000-08-02 | Towa Yakuhin Kk | Emulsion preparation including medicinal substance |
| CN102697724A (en) * | 2012-06-07 | 2012-10-03 | 沈阳药科大学 | Clopidogrel and salt submicron emulsion injection thereof as well as preparation method of same |
| CN104706575A (en) * | 2013-12-16 | 2015-06-17 | 天津迈迪瑞康生物医药科技有限公司 | Flurbiprofen axetil fat emulsion concentrate and preparation method and use thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6741655B2 (en) | Emulsion formulation of aprepitant | |
| JP6814148B2 (en) | Emulsion for parenteral administration | |
| EP2491919B2 (en) | Pharmaceutical solution of taxanes comprising ph regulator and preparation method thereof | |
| KR101680163B1 (en) | Fat emulsion containing prostaglandin | |
| JPWO2018211847A1 (en) | Self-emulsifying composition, method for producing the same, nanoemulsion, and method for producing the same | |
| WO2021180848A1 (en) | Sunflower phospholipid composition containing phosphatidylcholine | |
| JP2019210222A (en) | Oil-in-water type emulsion composition and method for producing the same | |
| WO2019230207A1 (en) | Method for producing oil-in-water type emulsion composition | |
| WO2019195218A1 (en) | Pharmaceutical compositions of meloxicam | |
| JP5591603B2 (en) | Propofol-containing oil-in-water emulsion composition | |
| KR20190110563A (en) | Fat emulsions and preparation methods thereof, methods for improving the stability of fat emulsions, and stability improvers of fat emulsions | |
| JP4894119B2 (en) | Fatty acid-containing liposome dispersion | |
| JP5759804B2 (en) | Container preparation | |
| JP6076520B2 (en) | Fat emulsion containing prostaglandins | |
| WO2019230964A1 (en) | Packed oil-in-water type emulsion composition | |
| JP7353453B1 (en) | Emulsion and its manufacturing method | |
| WO2011047639A1 (en) | Pharmaceutical solution of taxanes comprising chelating agent and preparation method thereof | |
| JP2013001700A (en) | Propofol-containing oil-in-water type emulsion composition | |
| JPH01113315A (en) | Fat emulsion containing vitamin k2 | |
| CN108888594B (en) | Nano fat emulsion for intravenous injection | |
| WO2005065676A1 (en) | Fat emulsion containing paclitaxel or docetaxel | |
| JP2012214430A (en) | Fat emulsion containing prostaglandin | |
| JP2012012331A (en) | Propofol-containing oil-in-water type emulsion composition | |
| JP2012012335A (en) | Propofol-containing oil-in-water type emulsion composition | |
| EP3579829A1 (en) | Long-term efficacy of liver disease treatment with epa and dha |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19810392 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19810392 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |