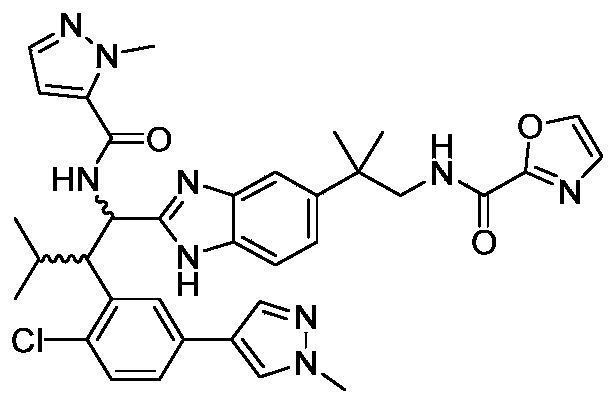
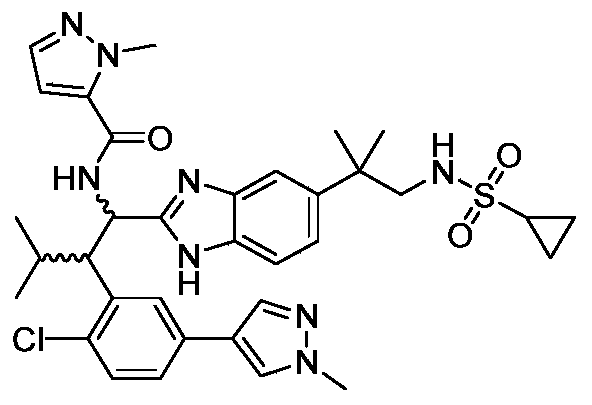
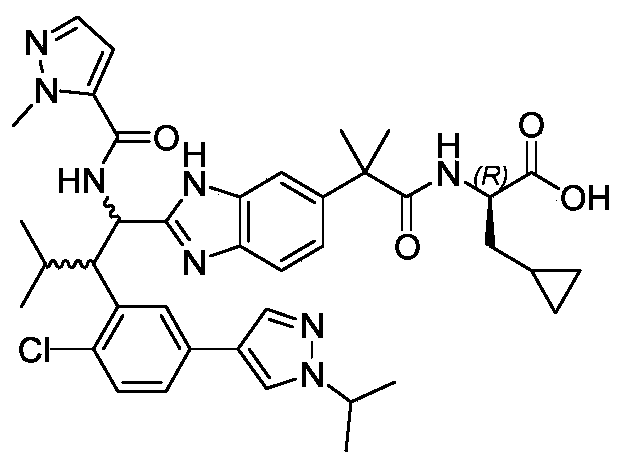
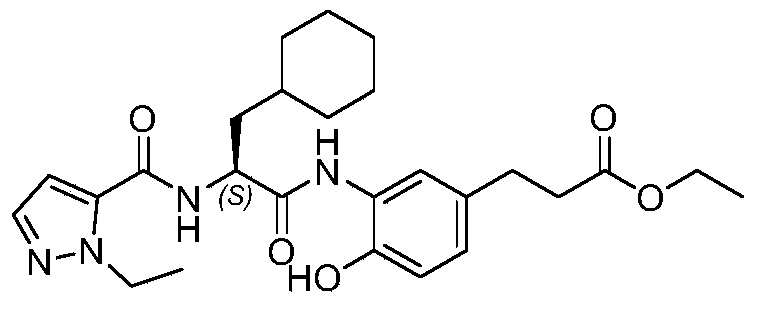
WO2019223718A1 - 一种免疫调节剂 - Google Patents
一种免疫调节剂 Download PDFInfo
- Publication number
- WO2019223718A1 WO2019223718A1 PCT/CN2019/087966 CN2019087966W WO2019223718A1 WO 2019223718 A1 WO2019223718 A1 WO 2019223718A1 CN 2019087966 W CN2019087966 W CN 2019087966W WO 2019223718 A1 WO2019223718 A1 WO 2019223718A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- membered
- alkyl
- compound
- group
- preparation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CCOC(C(C)c1cc([N+]([O-])=O)c(*)cc1)=O Chemical compound CCOC(C(C)c1cc([N+]([O-])=O)c(*)cc1)=O 0.000 description 24
- MTNXAWLFCNWTIX-UHFFFAOYSA-N CC(C)C(C(c1nc(ccc(C(C)C)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cc(cc1)OC)c1Cl Chemical compound CC(C)C(C(c1nc(ccc(C(C)C)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cc(cc1)OC)c1Cl MTNXAWLFCNWTIX-UHFFFAOYSA-N 0.000 description 2
- QJJAEMHKUSPWOT-UHFFFAOYSA-N CC(C)(C)OC(NC(C(c(cccc1)c1Cl)OC1CC1)C(O)=O)=O Chemical compound CC(C)(C)OC(NC(C(c(cccc1)c1Cl)OC1CC1)C(O)=O)=O QJJAEMHKUSPWOT-UHFFFAOYSA-N 0.000 description 1
- HEZXASHYBIATPN-SDPHREGOSA-N CC(C)(C1OC1N[C@H](C1CCC1)C(NC)=O)c(cc1F)cc2c1[nH]c(C(Cc1ccccc1C)CNC(c1ccn[n]1C)=O)n2 Chemical compound CC(C)(C1OC1N[C@H](C1CCC1)C(NC)=O)c(cc1F)cc2c1[nH]c(C(Cc1ccccc1C)CNC(c1ccn[n]1C)=O)n2 HEZXASHYBIATPN-SDPHREGOSA-N 0.000 description 1
- LHCZIRQRPHFGKG-UHFFFAOYSA-N CC(C)(CN)c(cc1)ccc1N Chemical compound CC(C)(CN)c(cc1)ccc1N LHCZIRQRPHFGKG-UHFFFAOYSA-N 0.000 description 1
- TYOCMFPNMFRLIX-UHFFFAOYSA-N CC(C)C(C(C(O)=O)NC(OC(C)(C)C)=O)c(cc(cc1)-c2c[n](C(C)C)nc2)c1Cl Chemical compound CC(C)C(C(C(O)=O)NC(OC(C)(C)C)=O)c(cc(cc1)-c2c[n](C(C)C)nc2)c1Cl TYOCMFPNMFRLIX-UHFFFAOYSA-N 0.000 description 1
- KHJXAYSKLVIXQX-FDALDRLYSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(/N=C(\C)/C(N2CCCC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(/N=C(\C)/C(N2CCCC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl KHJXAYSKLVIXQX-FDALDRLYSA-N 0.000 description 1
- QQSDNBYGJZMDBR-UHFFFAOYSA-O CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)C(N2CCN(C)CC2)=O)=O)cc2)c2[nH]1)NC(C([NH2+]C)=CC=N)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)C(N2CCN(C)CC2)=O)=O)cc2)c2[nH]1)NC(C([NH2+]C)=CC=N)=O)c(cccc1)c1Cl QQSDNBYGJZMDBR-UHFFFAOYSA-O 0.000 description 1
- YAEZCLUWSQTBMO-UHFFFAOYSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)c2c[nH]cn2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)c2c[nH]cn2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl YAEZCLUWSQTBMO-UHFFFAOYSA-N 0.000 description 1
- ODCVPZPHHDJHRZ-UHFFFAOYSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)c2ccn[nH]2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)c2ccn[nH]2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl ODCVPZPHHDJHRZ-UHFFFAOYSA-N 0.000 description 1
- LONIWJLJLOJWHQ-UHFFFAOYSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)c2nnc(C)[nH]2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(NC(C)c2nnc(C)[nH]2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl LONIWJLJLOJWHQ-UHFFFAOYSA-N 0.000 description 1
- SDLACXPDEOWSIG-UHFFFAOYSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(NC(COC2)C2C(NC2CC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(NC(COC2)C2C(NC2CC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl SDLACXPDEOWSIG-UHFFFAOYSA-N 0.000 description 1
- JYHAFGNFVUSLHY-UHFFFAOYSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(NC2(CCC2)C(NC)=O)=O)c(F)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(NC2(CCC2)C(NC)=O)=O)c(F)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl JYHAFGNFVUSLHY-UHFFFAOYSA-N 0.000 description 1
- DIAAZTXYQRFJLP-UHFFFAOYSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(NC2CCOCC2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(NC2CCOCC2)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl DIAAZTXYQRFJLP-UHFFFAOYSA-N 0.000 description 1
- DTQHAVIIHJTWAU-QBYNGQAISA-N CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C(C)C)C(NC)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C(C)C)C(NC)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl DTQHAVIIHJTWAU-QBYNGQAISA-N 0.000 description 1
- GTPFRARPVBBNHW-FNPAZFQPSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C(C)C)C(NC2CC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C(C)C)C(NC2CC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl GTPFRARPVBBNHW-FNPAZFQPSA-N 0.000 description 1
- LOTLKZPZTPMBQE-LWRMFSKNSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(N(C)C)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(N(C)C)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl LOTLKZPZTPMBQE-LWRMFSKNSA-N 0.000 description 1
- UMMFNUJEUQLTJB-PQLMJIFMSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(N2CCOCC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(N2CCOCC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl UMMFNUJEUQLTJB-PQLMJIFMSA-N 0.000 description 1
- RJCDSZPZVIEVJI-AHTGFLRJSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(NC(C)C)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(NC(C)C)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl RJCDSZPZVIEVJI-AHTGFLRJSA-N 0.000 description 1
- YFHGLEJFJOQYLU-NNKJGBLQSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(NC2CCOCC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C)C(NC2CCOCC2)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl YFHGLEJFJOQYLU-NNKJGBLQSA-N 0.000 description 1
- YPOHCYVGHDHZOO-YOXBCDPKSA-N CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C2CCC2)C(NC)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(cc(C(C)(C)C(N[C@H](C2CCC2)C(NC)=O)=O)cc2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl YPOHCYVGHDHZOO-YOXBCDPKSA-N 0.000 description 1
- ZBMIYCPEZJNNLD-UHFFFAOYSA-N CC(C)C(C(c1nc(cc(cc2)-c3c(C)[n](C(C)C(NC)=O)nc3C)c2[nH]1)NC(c1ccn[n]1C)=O)c1ccccc1Cl Chemical compound CC(C)C(C(c1nc(cc(cc2)-c3c(C)[n](C(C)C(NC)=O)nc3C)c2[nH]1)NC(c1ccn[n]1C)=O)c1ccccc1Cl ZBMIYCPEZJNNLD-UHFFFAOYSA-N 0.000 description 1
- AGYMXZHFDAIXJI-UHFFFAOYSA-N CC(C)C(C(c1nc(ccc(C(C)(C)CNOC(c2ccccc2)=O)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl Chemical compound CC(C)C(C(c1nc(ccc(C(C)(C)CNOC(c2ccccc2)=O)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cccc1)c1Cl AGYMXZHFDAIXJI-UHFFFAOYSA-N 0.000 description 1
- PAKIJSQWPPPOLT-UHFFFAOYSA-N CC(C)C(C(c1nc(ccc(C(C)C)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cc(cc1)F)c1Cl Chemical compound CC(C)C(C(c1nc(ccc(C(C)C)c2)c2[nH]1)NC(c1ccn[n]1C)=O)c(cc(cc1)F)c1Cl PAKIJSQWPPPOLT-UHFFFAOYSA-N 0.000 description 1
- ZLNCIFZYTSYIQN-UHFFFAOYSA-N CC(C)C(C(c1nc2cc(C(C)(C)CNC(C3CC3)=O)ccc2[nH]1)NC(c1ccn[n]1C)=O)c1ccccc1 Chemical compound CC(C)C(C(c1nc2cc(C(C)(C)CNC(C3CC3)=O)ccc2[nH]1)NC(c1ccn[n]1C)=O)c1ccccc1 ZLNCIFZYTSYIQN-UHFFFAOYSA-N 0.000 description 1
- DLSVVKQKKKKGRZ-UHFFFAOYSA-N CC(C)C(Cc1nc(cc(C(C)(C)C(N=C)O)cc2)c2[nH]1)c1c(C)cccc1 Chemical compound CC(C)C(Cc1nc(cc(C(C)(C)C(N=C)O)cc2)c2[nH]1)c1c(C)cccc1 DLSVVKQKKKKGRZ-UHFFFAOYSA-N 0.000 description 1
- CPMSRJWTAHFCGD-UHFFFAOYSA-N CC(C)C(Cc1nc(cc(C(C)(C)C(NC2CCOCC2)=O)cc2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl Chemical compound CC(C)C(Cc1nc(cc(C(C)(C)C(NC2CCOCC2)=O)cc2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl CPMSRJWTAHFCGD-UHFFFAOYSA-N 0.000 description 1
- LROXPZZOKFUCAM-UHFFFAOYSA-N CC(C)C(Cc1nc(cc(C(C)(C)C(NCCN2CCOCC2)=O)cc2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl Chemical compound CC(C)C(Cc1nc(cc(C(C)(C)C(NCCN2CCOCC2)=O)cc2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl LROXPZZOKFUCAM-UHFFFAOYSA-N 0.000 description 1
- AKTSECQZMOCUQB-GEPVFLLWSA-N CC(C)C(Cc1nc(cc(C(C)(C)C(N[C@H](C)C(N2CCN(C)CC2)=O)=O)cc2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl Chemical compound CC(C)C(Cc1nc(cc(C(C)(C)C(N[C@H](C)C(N2CCN(C)CC2)=O)=O)cc2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl AKTSECQZMOCUQB-GEPVFLLWSA-N 0.000 description 1
- HNSNZENPWWIHQW-WPNNEGNSSA-N CC(C)C(Cc1nc(ccc(C(C)(C)C(N[C@H](C2CCC2)C(NCC2(CC2)O)=O)=O)c2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c1ccccc1Cl Chemical compound CC(C)C(Cc1nc(ccc(C(C)(C)C(N[C@H](C2CCC2)C(NCC2(CC2)O)=O)=O)c2)c2[n]1/[O]=C(/c1ccn[n]1C)\N)c1ccccc1Cl HNSNZENPWWIHQW-WPNNEGNSSA-N 0.000 description 1
- CSEVTHUOIOZWHR-UFUCKMQHSA-N CC(C)C(Cc1nc2cc(C(C)(C)C(N[C@H](C)C(N3CCOCC3)=O)=O)ccc2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl Chemical compound CC(C)C(Cc1nc2cc(C(C)(C)C(N[C@H](C)C(N3CCOCC3)=O)=O)ccc2[n]1/[O]=C(/c1ccn[n]1C)\N)c(cccc1)c1Cl CSEVTHUOIOZWHR-UFUCKMQHSA-N 0.000 description 1
- ZQUMWQJBSUFHSD-ZCFIWIBFSA-N CC(C)C[C@H](C(NS(C)(=O)=O)=O)N Chemical compound CC(C)C[C@H](C(NS(C)(=O)=O)=O)N ZQUMWQJBSUFHSD-ZCFIWIBFSA-N 0.000 description 1
- FPFXPQBGWQKSKU-LFHRXCRSSA-N CC(C)C[C@H](C(NS(C)(=O)=O)=O)NC(C(C)(C)c(cc1)cc(nc2CC(C(C)C)c(cccc3)c3Cl)c1[n]2/[O]=C(/c1ccn[n]1C)\N)=O Chemical compound CC(C)C[C@H](C(NS(C)(=O)=O)=O)NC(C(C)(C)c(cc1)cc(nc2CC(C(C)C)c(cccc3)c3Cl)c1[n]2/[O]=C(/c1ccn[n]1C)\N)=O FPFXPQBGWQKSKU-LFHRXCRSSA-N 0.000 description 1
- FYKQOGDXFYYUDA-ANWICMFUSA-N CC(C)C[C@H](C(O)=O)NC(C(C)(C)c(cc1)cc(nc2CC(C(C)C)c(cccc3)c3Cl)c1[n]2/[O]=C(/c1ccn[n]1C)\N)=O Chemical compound CC(C)C[C@H](C(O)=O)NC(C(C)(C)c(cc1)cc(nc2CC(C(C)C)c(cccc3)c3Cl)c1[n]2/[O]=C(/c1ccn[n]1C)\N)=O FYKQOGDXFYYUDA-ANWICMFUSA-N 0.000 description 1
- PLEDOXVENPJVFN-SISPBFPDSA-N CCNC(/C(/C(C)C)=N/C(C(C)(C)c(cc1)cc2c1[nH]c(C(C(C(C)C)c(cccc1)c1Cl)NC(c1ccn[n]1C)=O)n2)=O)=O Chemical compound CCNC(/C(/C(C)C)=N/C(C(C)(C)c(cc1)cc2c1[nH]c(C(C(C(C)C)c(cccc1)c1Cl)NC(c1ccn[n]1C)=O)n2)=O)=O PLEDOXVENPJVFN-SISPBFPDSA-N 0.000 description 1
- UEWLHROWUMWFEK-LWRMFSKNSA-N CCNC([C@@H](C)NC(C(C)(C)c(cc1)cc2c1[nH]c(C(C(C(C)C)c(cccc1)c1Cl)NC(c1ccn[n]1C)=O)n2)=O)=O Chemical compound CCNC([C@@H](C)NC(C(C)(C)c(cc1)cc2c1[nH]c(C(C(C(C)C)c(cccc1)c1Cl)NC(c1ccn[n]1C)=O)n2)=O)=O UEWLHROWUMWFEK-LWRMFSKNSA-N 0.000 description 1
- JNFNIFUBIYUSNS-UHFFFAOYSA-N CCOC(C(C(c1ccccc1Cl)OC)[N+]([O-])=O)=O Chemical compound CCOC(C(C(c1ccccc1Cl)OC)[N+]([O-])=O)=O JNFNIFUBIYUSNS-UHFFFAOYSA-N 0.000 description 1
- IANAIFOSQIPCEG-UHFFFAOYSA-N CC[n]1nccc1/C(/N)=[O]/[n]1c(cc(C(C)(C)C(NC)=O)cc2)c2nc1CC(C(C)C)c(cccc1)c1Cl Chemical compound CC[n]1nccc1/C(/N)=[O]/[n]1c(cc(C(C)(C)C(NC)=O)cc2)c2nc1CC(C(C)C)c(cccc1)c1Cl IANAIFOSQIPCEG-UHFFFAOYSA-N 0.000 description 1
- ULFDGKMIFJZDSB-UHFFFAOYSA-N CC[n]1nccc1C(NC(CC1CCCCC1)C(Nc(cc(CC(OCC)=O)cc1)c1N)=O)=O Chemical compound CC[n]1nccc1C(NC(CC1CCCCC1)C(Nc(cc(CC(OCC)=O)cc1)c1N)=O)=O ULFDGKMIFJZDSB-UHFFFAOYSA-N 0.000 description 1
- HEEJTTIXASACLP-UHFFFAOYSA-N CC[n]1nccc1C(NC(CC1CCCCC1)C(Nc(ccc(CC(OCC)=O)c1)c1N)=O)=O Chemical compound CC[n]1nccc1C(NC(CC1CCCCC1)C(Nc(ccc(CC(OCC)=O)c1)c1N)=O)=O HEEJTTIXASACLP-UHFFFAOYSA-N 0.000 description 1
- PLLCIOVRDVKKLN-UHFFFAOYSA-N CC[n]1nccc1C(NC(CC1CCCCC1)c1nc(ccc(-c2c(C)[nH]nc2C)c2)c2[nH]1)=O Chemical compound CC[n]1nccc1C(NC(CC1CCCCC1)c1nc(ccc(-c2c(C)[nH]nc2C)c2)c2[nH]1)=O PLLCIOVRDVKKLN-UHFFFAOYSA-N 0.000 description 1
- MNBUWQQOXNLJMH-QFIPXVFZSA-N CC[n]1nccc1C(N[C@@H](CC1CCCCC1)c1nc(cc(cc2)-c3c(C)[o]nc3C)c2[nH]1)=O Chemical compound CC[n]1nccc1C(N[C@@H](CC1CCCCC1)c1nc(cc(cc2)-c3c(C)[o]nc3C)c2[nH]1)=O MNBUWQQOXNLJMH-QFIPXVFZSA-N 0.000 description 1
- AVFZOVWCLRSYKC-UHFFFAOYSA-N CN1CCCC1 Chemical compound CN1CCCC1 AVFZOVWCLRSYKC-UHFFFAOYSA-N 0.000 description 1
- MFMKTVNIVSWSTK-UHFFFAOYSA-N C[n]1nccc1C(N)=O Chemical compound C[n]1nccc1C(N)=O MFMKTVNIVSWSTK-UHFFFAOYSA-N 0.000 description 1
- LEDHHAQSWZTASO-UHFFFAOYSA-N Cc1n[nH]c(CC(OC)=O)c1-c(cc1N)ccc1N Chemical compound Cc1n[nH]c(CC(OC)=O)c1-c(cc1N)ccc1N LEDHHAQSWZTASO-UHFFFAOYSA-N 0.000 description 1
- UHEXOJKNBPXPQE-CSKARUKUSA-N O=C(CC1CCC1)/N=[SiH]/I Chemical compound O=C(CC1CCC1)/N=[SiH]/I UHEXOJKNBPXPQE-CSKARUKUSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/4184—1,3-Diazoles condensed with carbocyclic rings, e.g. benzimidazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/42—Oxazoles
- A61K31/422—Oxazoles not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/10—Drugs for disorders of the urinary system of the bladder
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D235/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings
- C07D235/02—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings condensed with carbocyclic rings or ring systems
- C07D235/04—Benzimidazoles; Hydrogenated benzimidazoles
- C07D235/06—Benzimidazoles; Hydrogenated benzimidazoles with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
Definitions
- the present invention relates to an immune modulators and their use in the manufacture of a medicament.
- IL-17 (interleukin-17) is a pro-inflammatory cytokine that plays a role in inducing other inflammatory cytokines, chemokines and adhesion factors.
- the IL-17 family consists of cytokines involved in acute and chronic inflammatory responses, including IL-17A (CTLA-8), IL-17B, IL-17C, IL-17D, IL-17E (IL-25), and IL-17F .
- IL-17A is expressed by TH17 cells and is involved in the pathogenesis of inflammation and autoimmune diseases.
- Human IL-17A is a glycoprotein with a molecular weight of about 17,000 Daltons.
- IL-17A transmits signals into cells through the IL-17 receptor complexes (IL-17RA and IL-17RC) (Wright, et al. Journal of immunology, 2008, 181: 2799-2805).
- the main function of IL-17A is through the up-regulation of pro-inflammatory and neutrophil migration cytokines and chemokines (including IL-6, G-CSF, TNF- ⁇ , IL-1, CXCL1, CCL2, CXCL2). Coordinates local tissue inflammation and matrix metalloproteinases to allow activated T cells to penetrate the extracellular matrix.
- IL-17A plays an important role in severe asthma and chronic obstructive pulmonary disease (COPD), and those patients usually do not respond or respond poorly to currently available drugs (Al-Ramli et al. J Allergy Clin Immunol, 2009, 123: 1185-1187). Upregulation of IL-17A levels is implicated in many diseases including rheumatoid arthritis (RA), bone erosion, intraperitoneal abscess, inflammatory bowel disease, allograft rejection, psoriasis, atherosclerosis, asthma and multiple Sclerosis (Gaffen, SL et al. Arthritis Research & Therapy, 2004, 6: 240-247).
- Targeted binding IL-17A and IL-17RA is the treatment of IL-17A-mediated autoimmune effective strategy for inflammatory diseases.
- IL-17A reduced by the treatment of animals neutralizing antibodies Autoimmune disease incidence and severity ( Komiyama Y et al.J.Immunol, 2006,177:. 566-573).
- IL-17A antibody has clinical trials in inflammatory diseases mediated by IL-7A (including asthma, psoriasis, rheumatoid arthritis, ankylosing spondylitis and multiple sclerosis) showed good results on.
- the IL-17A antibody (Cosentyx / secukinumab by Novartis) was approved by the FDA in January 2015 for the treatment of psoriasis.
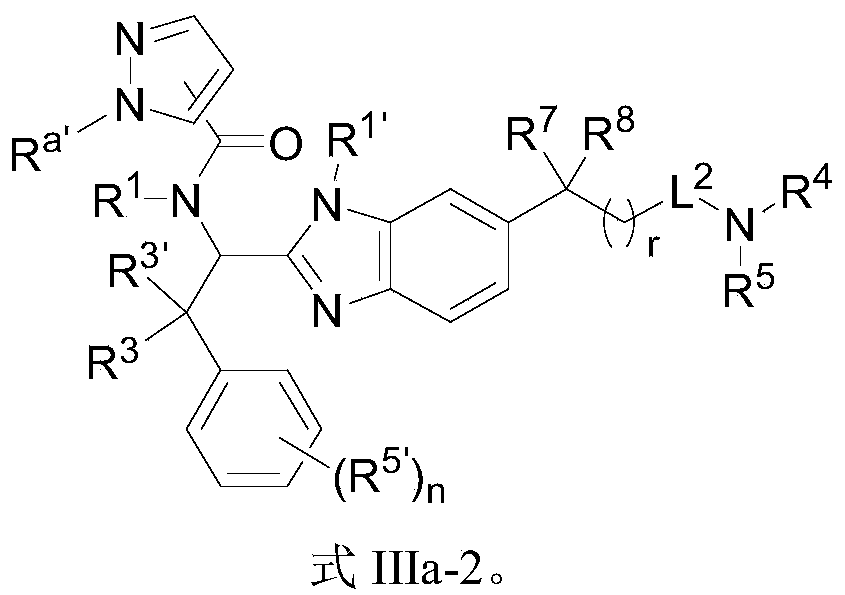
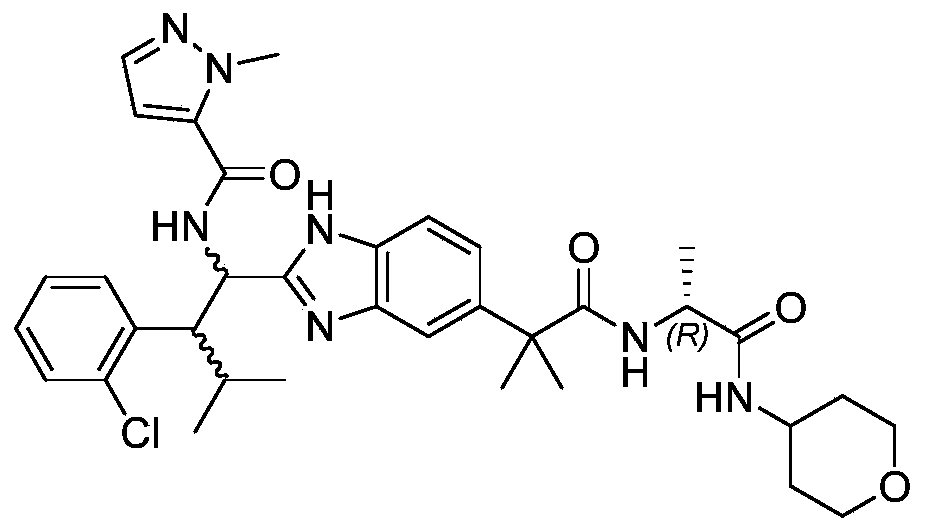
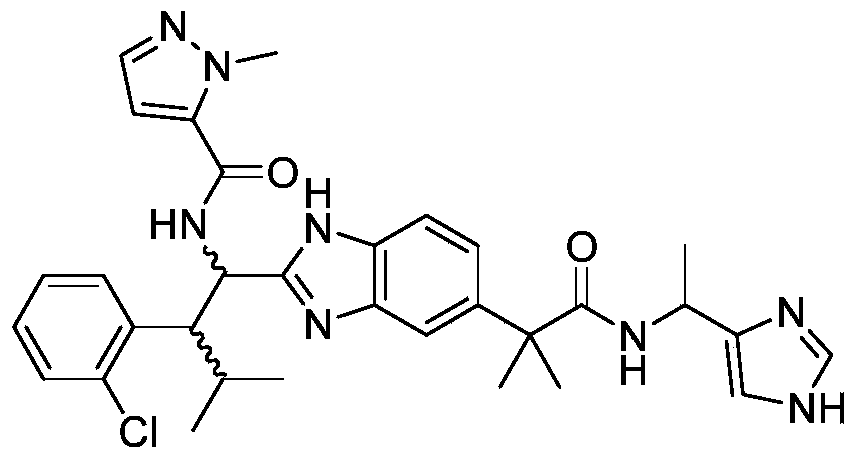
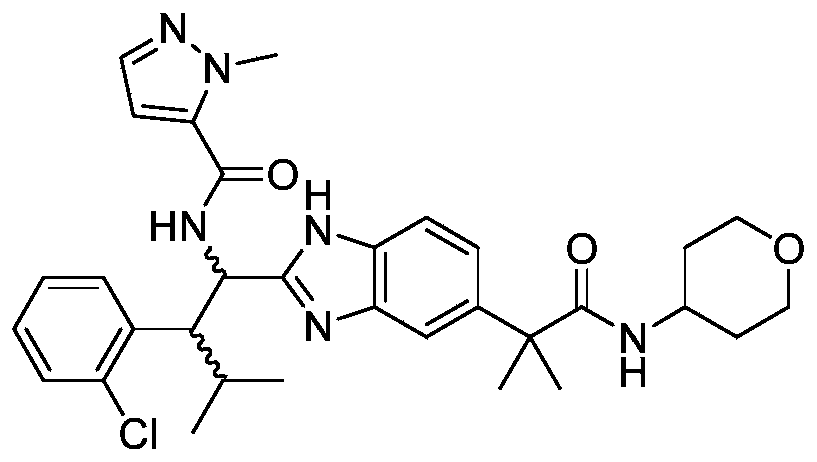
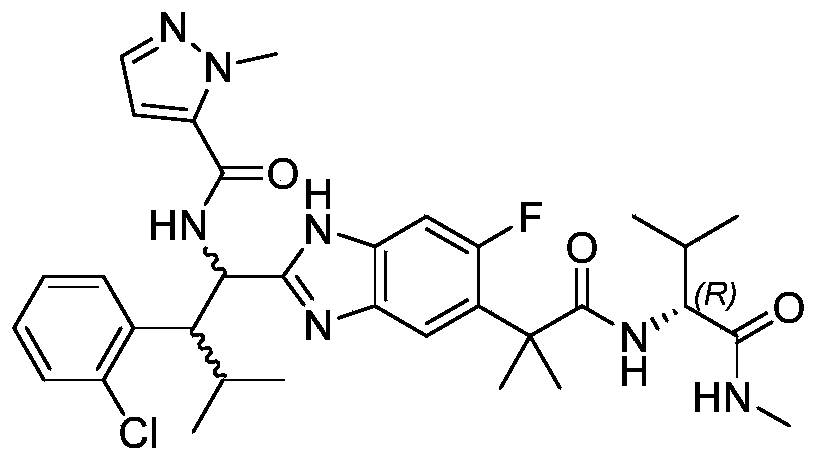
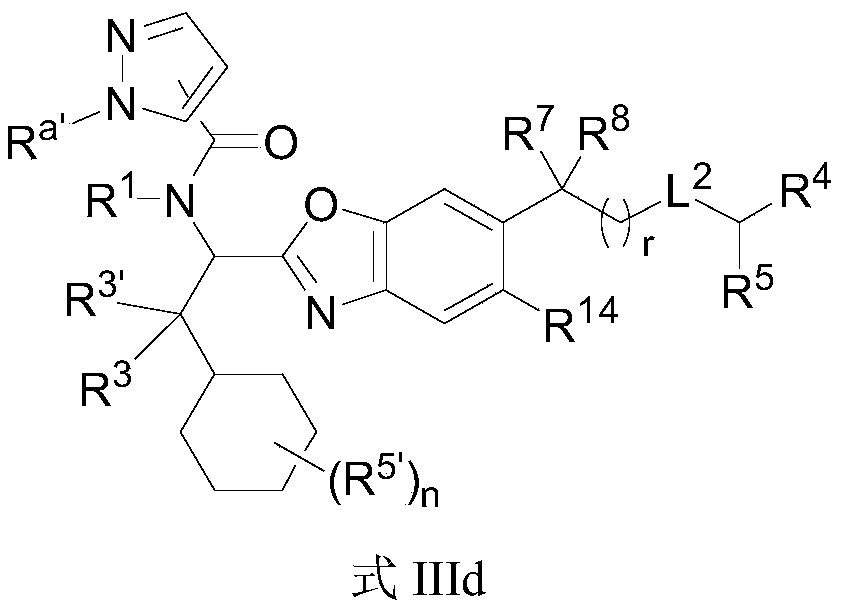
- the present invention provides compounds of Formula I, or a stereoisomer thereof, or a pharmaceutically acceptable salt thereof:
- X is selected from O, S, NR 1 ' ;
- R 1, R 2 are independently selected from hydrogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10 membered cycloalkyl, 3 to 10-membered heterocycloalkyl, 5 ⁇ 10 membered aromatic ring, 5 ⁇ 10 membered aromatic heterocyclic ring, -S (O) 2 R 6 , -S (O) R 6 , -S (O) 2 NR 6 R 7 , -S (O) NR 6 R 7 , -C (O) R 6 , -C (O) OR 6 , -C (O) NR 6 R 7 , -P (O) (OR 7 ) R 6 , -P (O) R 6 R 7 , -P (O) R 6 (NR 6 R 7 ), -P (O) (NR 6 R 7 ) (NR 6 R 7 ), -P (O) (OR 7 ) (NR 6 R 7 ),
- R 6, R 7 each independently selected from hydrogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10 membered cycloalkyl, 3 to 10-membered heterocycloalkyl, 5 ⁇ 10-membered aromatic ring, 5 ⁇ 10-membered aromatic heterocyclic ring; wherein alkyl, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aromatic ring, and aromatic heterocyclic ring are substituted by m R c ;
- R 3 is independently selected from hydrogen, -OR d, -SR d, -NR d R d, halogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 ⁇ 10 membered cycloalkyl, 3 ⁇ 10 membered heterocycloalkyl, 5 ⁇ 10 membered aromatic ring; wherein R d , alkyl, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, and aromatic ring are m R c is substituted; or, R 3 and R 3 ′ are connected to form a 3 to 10-membered cycloalkyl group and a 3 to 10-membered heterocycloalkyl group;
- R d is selected from hydrogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10 membered cycloalkyl, 3-10 membered heterocycloalkyl, 5-10-membered aromatic ring , 5 to 10-membered heteroaromatic ring;
- the B ring is selected from 3 to 10-membered cycloalkyl, 3 to 10-membered heterocycloalkyl, 5 to 10-membered aromatic ring, and 5 to 10-membered heteroaryl ring; among which cycloalkyl, heterocycloalkyl, aromatic ring, aromatic
- the heterocyclic ring is substituted by m R c ;
- R 1 ′ is selected from hydrogen and C 1-10 alkyl
- L 1 is selected from
- r 0, 1;
- R 8 and R 9 each independently selected from hydrogen, halo, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10-membered cycloalkyl, 3 to 10-membered heterocyclic ring Alkyl; wherein alkyl, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl are substituted by m R c ;
- R 8 and R 9 are connected to form a 3 to 10 membered cycloalkyl group, a 3 to 10 membered heterocycloalkyl group, a 5 to 10 membered aromatic ring, and a 5 to 10 membered heteroaryl ring; , Aromatic ring, heteroaryl ring are substituted by m R c ;
- L 2 is selected from -C (O) NR 10- , -NR 10 C (O)-, -C (O)-, -C (O) O-, -S (O) 2 NR 10- , -S ( O) NR 10- , -NR 10 S (O) 2- , -NR 10 S (O)-, -P (O) NR 10- , -NR 10 P (O)-or none;
- R 10 is selected from hydrogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10 membered cycloalkyl, 3 to 10-membered heterocycloalkyl group, wherein the alkyl, alkenyl, , Alkynyl, cycloalkyl, heterocycloalkyl are substituted by m R c ;
- R 4 and R 5 are each independently selected from hydrogen, halo, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10 membered cycloalkyl, 3 to 10-membered heterocycloalkyl group , 5- to 10-membered aromatic ring, 5- to 10-membered aromatic heterocyclic ring, -CN, -NO 2 , -OR a , -OC (O) R a , -OC (O) NR a R b , -OS (O) 2 R a , -SR a , -S (O) 2 R a , -S (O) 2 NR a R b , -OS (O) 2 NR a R b , -C (O) R a , -C ( O) OR a , -C (O) NR a R b , -NR a R b , -
- R 4 and R 5 are connected to form a 3 to 10-membered cycloalkyl group and a 3 to 10-membered heterocycloalkyl group; wherein the cycloalkyl group and the heterocycloalkyl group are substituted with m R c ;
- X 1 is selected from CR 11 or N;
- X 4 is selected from N or CH;
- R 11 is selected from hydrogen, halogen, -CN, -NO 2, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl group, C 1 ⁇ 10 alkyl halo, 3 to 10-membered ring Alkyl, 3- to 10-membered heterocycloalkyl, -OR a , -NR a R b ;
- R 12 is selected from hydrogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10-membered cycloalkyl, 3 to 10-membered heterocycloalkyl, -S (O ) 2 R a , -S (O) 2 NR a R b , -C (O) R a , -C (O) OR a , -C (O) NR a R b ;
- R 13, R 14 are independently selected from hydrogen, halogen, -CN, -NO 2, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl group, C 1 ⁇ 10 alkyl halo, 3 ⁇ 10 membered cycloalkyl, 3 ⁇ 10 membered heterocycloalkyl, -OR a , -NR a R b ; wherein alkyl, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl are m R c replaced;
- n 0,1,2,3,4;
- R a, R b are independently selected from hydrogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl group, -S (O) 2 R d , -S (O) R d, - P (O) 2 R d , -P (O) R d , 3 to 10-membered cycloalkyl, 3 to 10-membered heterocycloalkyl, 5 to 10-membered aromatic ring, 5 to 10-membered heteroaryl ring; Alkyl, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aromatic ring, heteroaryl ring are substituted by m R c ;
- R 1 and R 2 are independently selected from hydrogen, C 1-6 alkyl, 3 to 10-membered cycloalkyl, 3 to 10-membered heterocycloalkyl, 6 to 10-membered aromatic ring, 5 to 10-membered aromatic heterocycle, -S (O) 2 R 6 , -S (O) R 6 , -S (O) 2 NR 6 R 7 , -C (O) R 6 , -C (O) OR 6 , -C (O) NR 6 R 7 ; wherein alkyl, cycloalkyl, heterocycloalkyl, aromatic ring, and aromatic heterocyclic ring are substituted by m R c ;
- R 6 and R 7 are independently selected from hydrogen, C 1-6 alkyl, 3 to 10-membered cycloalkyl, 3 to 10-membered heterocycloalkyl, 6 to 10-membered aromatic ring, and 5 to 10-membered aromatic heterocycle; Wherein alkyl, cycloalkyl, heterocycloalkyl, aromatic ring, and aromatic heterocyclic ring are substituted by m R c ;
- R 3 and R 3 ′ are independently selected from hydrogen, -OR d , halogen, C 1-6 alkyl, 3 to 10-membered cycloalkyl, 3 to 10-membered heterocycloalkyl, and 5 to 10-membered aromatic ring; An alkyl group, a cycloalkyl group, a heterocycloalkyl group, and an aromatic ring are substituted with m R c ; or, R 3 and R 3 ′ are connected to form a 3 to 10-membered cycloalkyl group;
- R d is selected from hydrogen, C 1 ⁇ 10 alkyl group, C 2 ⁇ 10 alkylene group, C 2 ⁇ 10 alkynyl groups, 3 to 10 membered cycloalkyl, 3-10 membered heterocycloalkyl, 5-10-membered aromatic ring , 5 to 10-membered heteroaromatic ring;
- R 8 and R 9 are independently selected from hydrogen, halogen, C 1-6 alkyl, 3 to 10-membered cycloalkyl, and 3 to 10-membered heterocycloalkyl; among them alkyl, cycloalkyl, and heterocycloalkane The group is substituted by m R c ;
- R 8 and R 9 are connected to form a 3 to 10 membered cycloalkyl group, a 3 to 10 membered heterocycloalkyl group, a 6 to 10 membered aromatic ring, and a 5 to 10 membered heteroaryl ring;
- Aromatic ring, heteroaryl ring may be further substituted by m R c ;
- R 10 is selected from hydrogen, C 1-6 alkyl, 3 to 10 membered cycloalkyl, and 3 to 10 membered heterocycloalkyl, wherein alkyl, cycloalkyl, and heterocycloalkyl are substituted by m R c ;
- R 4 and R 5 are independently selected from hydrogen, halogen, C 1-6 alkyl, 3 to 10 membered cycloalkyl, 3 to 10 membered heterocycloalkyl, 6 to 10 membered aromatic ring, and 5 to 10 membered Aromatic heterocycle, -CN, -NO 2 , -OR a , -OC (O) R a , -C (O) R a , -C (O) OR a , -C (O) NR a R b ,- NR a R b , -NR a C (O) R b , -NR a C (O) R b , -NR a C (O) NR a R b , -NR a S (O) 2 R b , -NR a S (O) 2 NR a R b -S (O) 2 R a , -S (O) 2 NR a R b ; wherein al
- R 4 and R 5 are connected to form a 3 to 10-membered cycloalkyl group and a 3 to 10-membered heterocycloalkyl group; wherein the cycloalkyl group and the heterocycloalkyl group are substituted with m R c ;
- R 11 is selected from hydrogen, halogen, and C 1-6 alkyl
- R 12 is selected from hydrogen and C 1-6 alkyl
- R 13 and R 14 are each independently selected from hydrogen, halogen, C 1-6 alkyl, and C 2-6 alkenyl; wherein alkyl and alkenyl are substituted by m R c ;
- n 0, 1, 2, 3;
- R a and R b are independently selected from hydrogen, C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, -S (O) 2 R d , -S (O) R d ,- P (O) 2 R d , -P (O) R d 3 to 10 membered cycloalkyl, 3 to 10 membered heterocycloalkyl, 6 to 10 membered aromatic ring, 5 to 10 membered heteroaromatic ring; Group, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aromatic ring, and heteroaryl ring are substituted with m R c .
- R 1 and R 2 are independently selected from hydrogen, -S (O) 2 R 6 , -S (O) R 6 , -S (O) 2 NR 6 R 7 , -C (O) R 6 , -C ( O) OR 6 , -C (O) NR 6 R 7 ;
- R 6 and R 7 are independently selected from hydrogen, 3 to 10 membered cycloalkyl, 3 to 10 membered heterocycloalkyl, 6 to 10 membered aromatic ring, and 5 to 10 membered aromatic heterocyclic ring;
- An alkyl group, an aromatic ring, and an aromatic heterocyclic ring are substituted by m R c ;
- R 3 and R 3 ′ are independently selected from hydrogen, -OR d , C 1-6 alkyl, 3 to 10-membered cycloalkyl, 3 to 10-membered heterocycloalkyl, and 5 to 10-membered aromatic ring; , Cycloalkyl, heterocycloalkyl, and aromatic ring are substituted with m R c ; or, R 3 and R 3 ′ are connected to form a 3 to 8-membered cycloalkyl group;
- R 8 and R 9 are independently selected from hydrogen, C 1-6 alkyl, 3 to 10-membered cycloalkyl, and 3 to 10-membered heterocycloalkyl;
- R 8 and R 9 are connected to form a 3 to 10 membered cycloalkyl group, a 3 to 10 membered heterocycloalkyl group, a 6 to 10 membered aromatic ring, and a 5 to 10 membered heteroaryl ring;
- R 4 and R 5 are each independently selected from hydrogen, C 1-6 alkyl, 3-10 membered cycloalkyl, 3 to 10 membered heterocycloalkyl, 6 to 10 membered aromatic ring, 5 to 10 membered aromatic heterocyclic ring, -OR a , -C (O) R a , -C (O) OR a , -C (O) NR a R b , -NR a R b , -NR a C (O) R b , -NR a C (O) R b , -NR a C (O) NR a R b , -NR a S (O) 2 R b , -NR a S (O) 2 NR a R b , -S (O) 2 R a , -S (O) 2 NR a R b , wherein the alkylcycloalkyl, heterocycloalkyl, aromatic ring, and aromatic
- R 4 and R 5 are connected to form a 3 to 10-membered cycloalkyl group and a 3 to 10-membered heterocycloalkyl group; wherein the cycloalkyl group and the heterocycloalkyl group may be further substituted with m R c ;
- R 11 is selected from hydrogen and C 1-6 alkyl
- R 12 is selected from hydrogen and C 1-6 alkyl
- R 13 and R 14 are each independently selected from hydrogen, C 1-6 alkyl, and C 2-6 alkenyl; wherein alkyl and alkenyl are substituted by m R c ;
- m 0,1,2.
- R a ' is selected from C 1-6 alkyl.
- R a ' is selected from C 1-6 alkyl.
- R 5 ′ is halogen and CN; n is selected from 0, 1, 2; L 2 is -C (O) NR 10- , -NR 10 C (O)-, -C (O)-, -C (O) O- or None.
- R 5 ′ is a halogen and a 5- to 10-membered heteroaromatic ring, wherein the 5- to 10-membered aromatic heterocyclic ring may be further substituted with 0 to 3 C 1 to 10 alkyl groups, 3 to 10-membered heterocyclic alkyl groups, 5- to 10-membered aromatic ring, 5- to 10-membered aromatic heterocyclic substitution;
- n is selected from 0, 1, 2, 3;
- L 2 is -C (O) NR 10- , -NR 10 C (O)-, -C (O)-, -C (O) O-, or none.
- R 5 ′ is halogen and —OR a ; n is selected from 0, 1, 2, 3; L 2 is none.
- R a ' is selected from substituted or unsubstituted C 1-6 alkyl; R 5' is hydrogen and halogen; n is selected from 0, 1, 2; L 2 is -C (O) NR 10- , -NR 10 C (O)-, -C (O)-, -C (O) O- or none.
- R a ' and R c are each selected from C 1-6 alkyl; R 5' is hydrogen and halogen; n is selected from 0, 1, 2; L 2 is -C (O) NR 10- , -NR 10 C (O)-, -C (O)-, -C (O) O- or none.
- R a ' is selected from substituted or unsubstituted C 1-6 alkyl; R 5' is hydrogen and halogen; n is selected from 0, 1, 2; L 2 is -C (O) NR 10- , -NR 10 C (O)-, -C (O)-, -C (O) O- or none.
- R a ' is selected from C 1-6 alkyl.
- the present invention also provides the use of the aforementioned compound, or a stereoisomer thereof, or a pharmaceutically acceptable salt thereof in the manufacture of a medicament for treating an IL-17A-mediated disease.
- the IL-17A-mediated disease defined in the present invention is a disease in which IL-17A plays an important role in the pathogenesis of the disease.
- the main function of IL-17A is to coordinate local tissue inflammation and thus play a role in various diseases.
- IL-17A-mediated diseases include one or more of inflammation, autoimmune diseases, infectious diseases, cancer, and diseases associated with precancerous syndrome.
- Cancer or “malignant tumor” refers to any of a variety of diseases characterized by the uncontrolled proliferation of cells, the ability of affected cells to spread to other sites locally or through the bloodstream and lymphatic system The body (ie metastasis) and any of a number of characteristic structures and / or molecular characteristics.
- Cancer cells refer to cells that undergo early, intermediate, or advanced stages of multi-step tumor progression. Cancers include sarcoma, breast cancer, lung cancer, brain cancer, bone cancer, liver cancer, kidney cancer, colon cancer, and prostate cancer.
- a compound of Formula I is used to treat a cancer selected from the group consisting of colon cancer, brain cancer, breast cancer, fibrosarcoma, and squamous cell carcinoma.
- the cancer is selected from the group consisting of melanoma, breast cancer, colon cancer, lung cancer, and ovarian cancer.
- the cancer treated is metastatic cancer.
- autoimmune diseases are caused by the body's immune response to substances and tissues normally present in the body.
- autoimmune diseases include myocarditis, lupus nephritis, primary biliary cirrhosis, psoriasis, Type 1 diabetes, Graves' disease, celiac disease, Crohn's disease, autoimmune neutropenia, juvenile arthritis, rheumatoid arthritis, fibromyalgia, Guillain-Barre syndrome, multiple sclerosis and autoimmune retinopathy.
- Some embodiments of the present invention relates to the treatment of autoimmune diseases such as psoriasis or multiple sclerosis.
- Inflammatory diseases include a variety of conditions characterized by histopathological inflammation.
- inflammatory diseases include acne vulgaris, asthma, celiac disease, chronic prostatitis, glomerulonephritis, inflammatory bowel disease, pelvic inflammation, reperfusion injury, rheumatoid arthritis, sarcoidosis, vasculitis, House dust mite-induced airway inflammation and interstitial cystitis.
- inflammatory diseases include acne vulgaris, asthma, celiac disease, chronic prostatitis, glomerulonephritis, inflammatory bowel disease, pelvic inflammation, reperfusion injury, rheumatoid arthritis, sarcoidosis, vasculitis, House dust mite-induced airway inflammation and interstitial cystitis.
- inflammatory diseases include acne vulgaris, asthma, celiac disease, chronic prostatitis, glomerulonephritis, inflammatory bowel disease, pelvic inflammation, reperfusion injury, rheumatoid arthritis, sarc
- the invention also provides a medicament, which is a preparation prepared from the aforementioned compound, or a stereoisomer thereof, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable auxiliary material.
- the compounds and derivatives provided in the present invention can be named according to the IUPAC (International Union of Pure and Applied Chemistry) or CAS (Chemical Abstracts Service, Columbus, OH) naming system.
- Substitute refers to the replacement of a hydrogen atom in a molecule with another different atom or molecule.
- the minimum and maximum values of the carbon atom content in a hydrocarbon group are indicated by a prefix, for example, the prefix Ca-b alkyl indicates any alkyl group containing "a" to "b” carbon atoms. Therefore, for example, "C 1-4 alkyl” means an alkyl group containing 1 to 4 carbon atoms.
- Alkyl refers to a saturated hydrocarbon chain having a specified number of member atoms.
- C 1 to C 6 alkyl refers to an alkyl group having 1 to 6 member atoms, such as 1 to 4 member atoms.
- the alkyl group can be straight or branched. Representative branched alkyl groups have one, two, or three branches. An alkyl group may be optionally substituted with one or more substituents as defined herein.
- Alkyl includes methyl, ethyl, propyl (n-propyl and isopropyl), butyl (n-butyl, isobutyl, and tert-butyl), pentyl (n-pentyl, isopentyl, and neopentyl) Base) and hexyl.
- the alkyl group may be a part of other groups such as a C 1 to C 6 alkoxy group.
- Cycloalkyl refers to a saturated or partially saturated cyclic group having 3 to 14 carbon atoms without ring heteroatoms and having a single ring or multiple rings (including fused, bridged, and spiro ring systems).
- cycloalkyl is applicable when the point of attachment is at a non-aromatic carbon atom (e.g. 5, 6, 7, 8, 8- Naphthalene-5-yl).
- cycloalkyl includes cycloalkenyl groups, such as cyclohexenyl.
- Examples of the cycloalkyl group include, for example, adamantyl, cyclopropyl, cyclobutyl, cyclohexyl, cyclopentyl, cyclooctyl, cyclopentenyl, and cyclohexenyl.
- Examples of a cycloalkyl group including a polybicycloalkyl ring system are dicyclohexyl, dicyclopentyl, bicyclooctyl, and the like. Two such bicyclic alkyl polycyclic structures are exemplified and named below: Bicyclohexyl and Dicyclohexyl.
- (Ca-Cb) alkenyl refers to an alkenyl group having a to b carbon atoms and is intended to include, for example, vinyl, propenyl, isopropenyl, 1,3-butadienyl, and the like.
- Alkynyl means a linear monovalent hydrocarbon radical or a branched monovalent hydrocarbon radical containing at least one triple bond.
- alkynyl is intended to include further having one triple bond and one double bond are those hydrocarbyl groups.
- (C2-C6) alkynyl is meant to include ethynyl, propynyl and the like.
- Halogen is fluorine, chlorine, bromine or iodine.
- Haloalkyl means that a hydrogen atom in an alkyl group may be substituted with one or more halogen atoms.
- C 1-4 haloalkyl refers to an alkyl group containing 1 to 4 carbon atoms in which a hydrogen atom is replaced by one or more halogen atoms.
- Heterocycle and “heterocycloalkyl” refer to a saturated ring or a non-aromatic unsaturated ring containing at least one heteroatom; wherein the heteroatom refers to a nitrogen atom, an oxygen atom, and a sulfur atom;
- Aromatic heterocycle means an aromatic unsaturated ring containing at least one heteroatom; wherein heteroatom refers to a nitrogen atom, an oxygen atom, and a sulfur atom;
- Steps include enantiomers and diastereomers
- pharmaceutically acceptable means that a carrier, carrier, diluent, excipient, and / or salt formed is generally chemically or physically compatible with the other ingredients that make up a pharmaceutical dosage form and is physiologically Compatible with the receptor.
- salts and “pharmaceutically acceptable salt” refer to the above-mentioned compounds or their stereoisomers, acidic and / or basic salts formed with inorganic and / or organic acids and bases, and also include zwitterionic salts (internal Salts), and also include quaternary ammonium salts, such as alkylammonium salts. These salts can be obtained directly in the final isolation and purification of the compounds. It can also be obtained by mixing the above-mentioned compound, or a stereoisomer thereof, with an appropriate amount of an acid or a base (e.g., equivalent).
- salts may be precipitated in solution and collected by filtration, or recovered after evaporation of the solvent, or prepared by freeze-drying after reaction in an aqueous medium.
- the salt described in the present invention may be the hydrochloride, sulfate, citrate, benzenesulfonate, hydrobromide, hydrofluorate, phosphate, acetate, propionate, succinate Acid salt, oxalate, malate, succinate, fumarate, maleate, tartrate or trifluoroacetate.
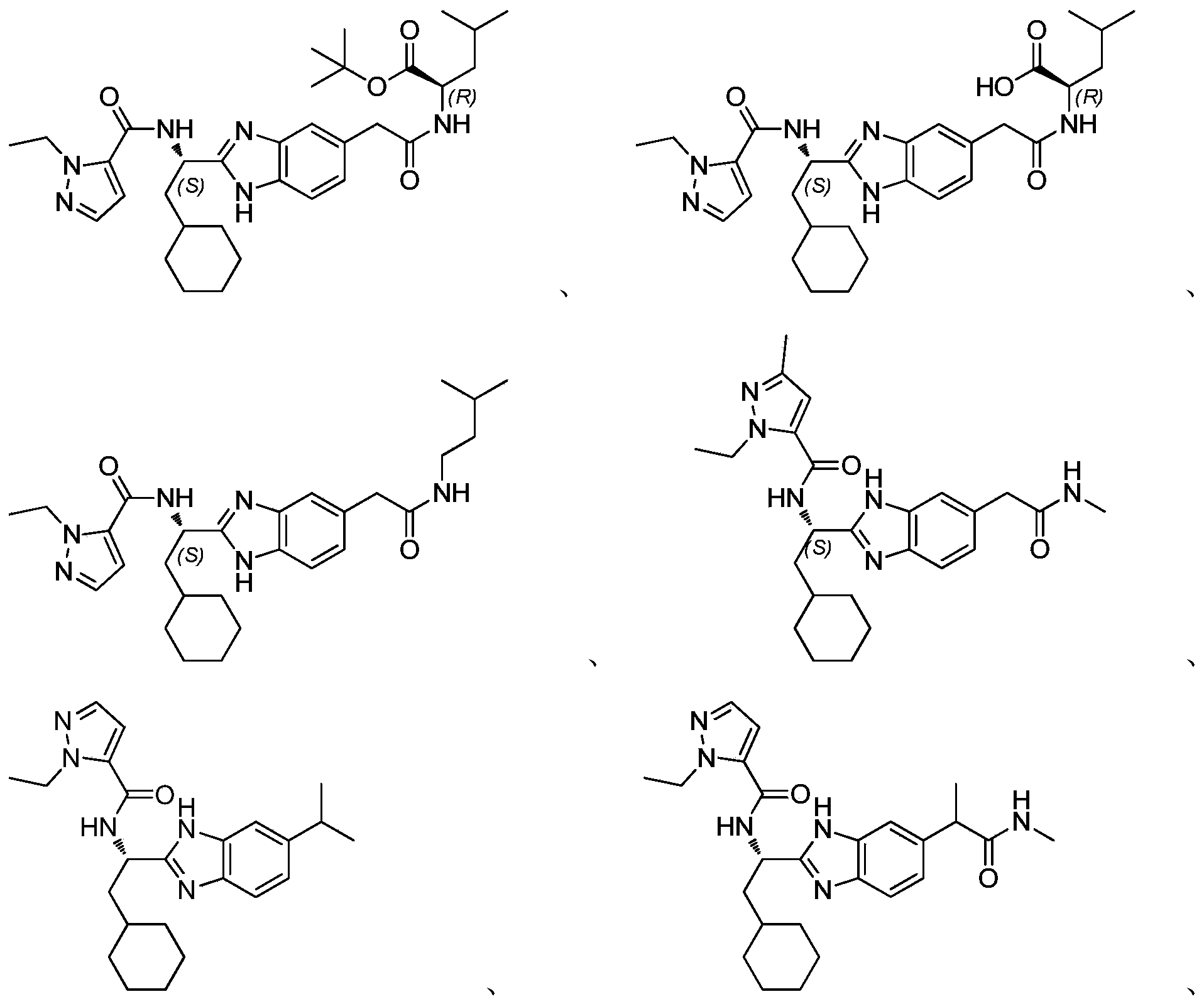
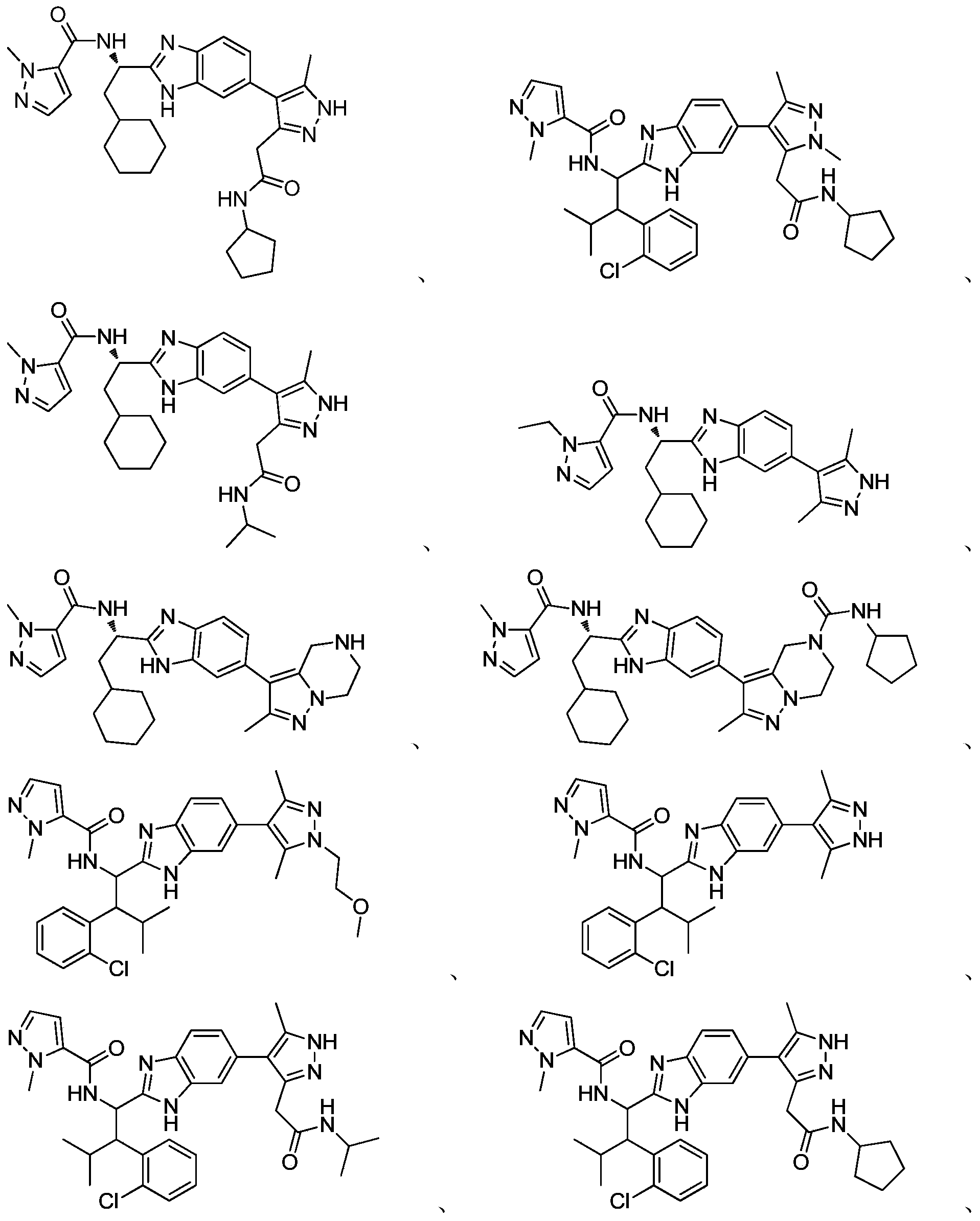
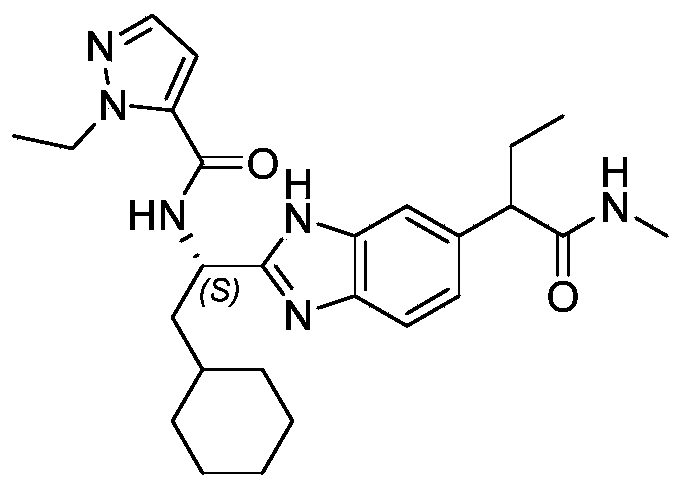
- novel compound of formula I disclosed by the present invention exhibits good IL-17A inhibitory activity, and provides a new medicinal possibility for clinical treatment of diseases related to abnormal IL-17A activity.
- one or more compounds of the invention can be used in combination with each other.
- the compound of the present invention can be used in combination with any other active agent to prepare a medicament or a pharmaceutical composition for regulating cell function or treating a disease. If a group of compounds is used, the compounds can be administered to the subject simultaneously, separately, or sequentially.
- the structure of the compound is determined by nuclear magnetic resonance (NMR) and mass spectrometry (MS).
- NMR shift ( ⁇ ) is given in units of 10-6 (ppm).
- NMR measurements were performed using (Bruker AvanceIII 400 and Bruker Avance 300) nuclear magnetic instruments.
- the solvents used for the determination were deuterated dimethyl sulfoxide (DMSO-d6), deuterated chloroform (CDCl 3 ), deuterated methanol (CD3OD), and internal standard. It is tetramethylsilane (TMS).
- MPLC medium pressure preparative chromatography
- Gilson GX-281 reverse phase preparative chromatography uses a Gilson GX-281 reverse phase preparative chromatography.
- the thin-layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate.
- the specifications for thin-layer chromatography purification products are 0.4mm ⁇ 0.5mm.
- the known starting materials of the present invention can be synthesized by or in accordance with methods known in the art, or can be purchased from companies such as Anaiji Chemical, Chengdu Kelong Chemical, Shaoyuan Chemical Technology, and Bailingwei Technology.
- the solution means an aqueous solution.
- reaction temperature is room temperature.
- M is mole per liter.
- Room temperature is the most suitable reaction temperature and is 20 ° C to 30 ° C.
- DMF refers to N, N-dimethylformamide.
- DMSO refers to dimethyl sulfoxide
- DIPEA refers to diisopropylethylamine.
- Boc refers to the t-butyloxycarbonyl group.
- TFA refers to trifluoroacetic acid.
- DBU 1,8-diazabicycloundec-7-ene.
- HATU 2- (7-Azobenzotriazole) -N, N, N ', N'-tetramethylurea hexafluorophosphate.
- HBTU O-benzotriazole-tetramethylurea hexafluorophosphate.
- EDCI 1- ethyl - (3-dimethylaminopropyl) carbodiimide hydrochloride.
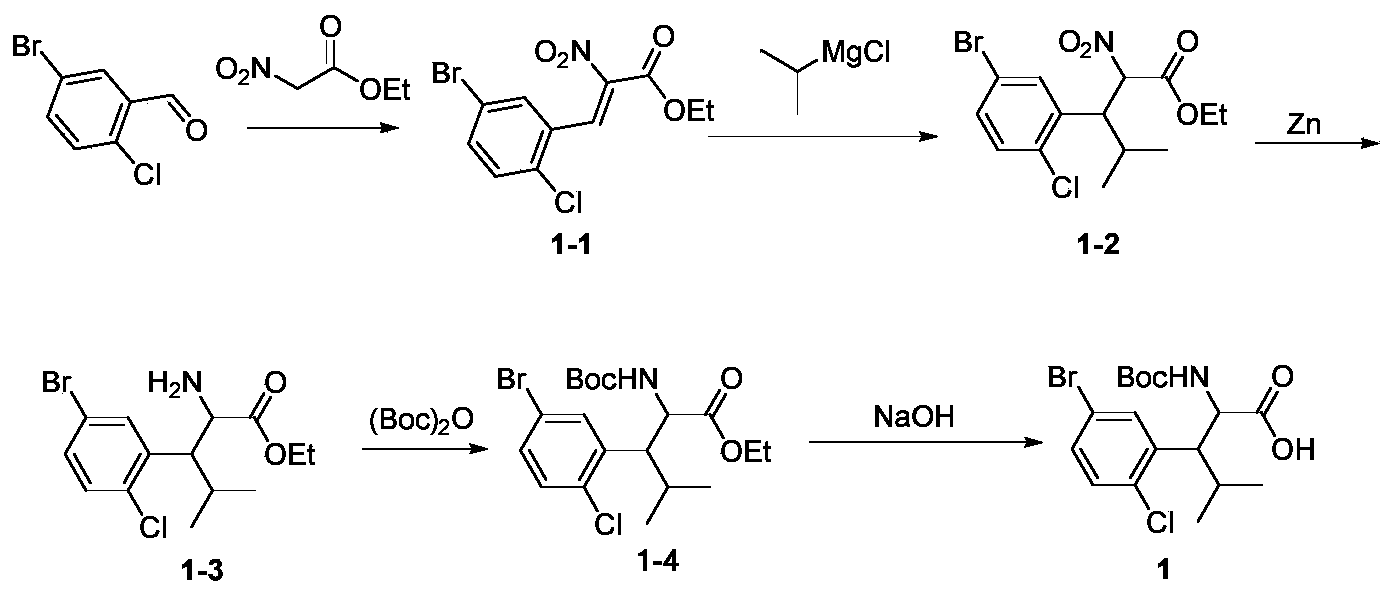
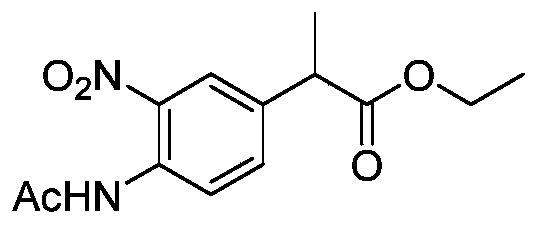
- intermediate 2-1 can be obtained by reacting o-chlorobenzaldehyde with ethyl nitroacetate.
- intermediate 2-1 5 g, 19.56 mmol
- methanol 50 mL
- sodium methoxide 4.23 g, 78.23 mmol
- TLC TLC showed that the starting materials disappeared.
- the solvent was concentrated and removed under reduced pressure. 20 mL of saturated ammonium chloride aqueous solution and ethyl acetate (20 mL ⁇ 2) were added.
- the intermediate 7-1 (1.58 g, 5.48 mmol) was dissolved in 10 mL of glacial acetic acid, zinc powder (1.79 g, 27.41 mmol) was added, and the reaction was stirred at room temperature for 2 hours.
- the reaction was suction filtered, washed with a small amount of ethyl acetate, and the filtrate It was concentrated to dryness under reduced pressure, and the crude product was separated on a silica gel column (petroleum ether / ethyl acetate 2: 1) to obtain intermediate 7-2 (0.83 g, 3.22 mmol, 58.76% yield), a pale yellow solid, MS m / z: 258 [M + 1].
- Intermediate 2-1 can be obtained by reacting Intermediate 2-1 with cyclopropanol in the presence of potassium tert-butoxide, and then reducing it by nitro, protecting the amino group with Boc, and hydrolyzing.
- Dichloromethane-activated resin was added to a solution of FMOC-D-CHA-OH (2.00 g, 5.08 mmol) and DIPEA (1.97 g, 15.24 mmol, 2.66 mL) in DMF (40.00 mL).
- intermediate 7 in Example 7 Referring to the method for preparing intermediate 7 in Example 7, intermediate 2-1 and 4-methylpyrazole were reacted in a potassium carbonate-DMF system, and then reduced with nitrozinc powder, Boc protected amino groups, and finally hydrolyzed by SFC.
- the four single chiral isomers 17-a, 17-b, 17-c, and 17-d of intermediate 17 can be obtained by separation and preparation with a chiral resolution column.
- MS m / z 324 [M-55] + .
- Example 1 Under nitrogen protection, Example 1 Intermediate 1-4 (710mg, 1.63mmol), Intermediate 18-1 (509.69mg, 2.45mmol), Pd (PPh3) 4 (94.31mg, 81.66umol) and Na2CO3 (519.33mg (4.90mmol), suspended in a mixed solvent of 1,4-dioxane (3mL) and H2O (0.3mL), and degassed by ultrasound for 15min. The reaction solution was heated to 80 ° C and reacted overnight, and concentrated to dryness under reduced pressure.
- Pd (PPh3) 4 94.31mg, 81.66umol
- Na2CO3 519.33mg (4.90mmol
- the intermediates 1-4 and 19-1 were coupled and then subjected to alkaline hydrolysis, and finally separated by SFC chiral separation column to prepare four single chiral isomers of intermediate 19, respectively.
- intermediate 1-4 was coupled with 2-methoxypyridine-4-valerylboronic acid and then subjected to alkaline hydrolysis, and finally separated and prepared by SFC chiral separation column to obtain intermediate 21 respectively.
- MS m / z 449 [M + 1] + .
- Example 19 Referring to the preparation method of Example 1, using 2-chloro-4-bromobenzaldehyde as a starting material, after condensation with ethyl nitroacetate, cyclopropyl magnesium bromide for Grignard reaction, nitro reduction, amino Boc protection, Referring to the coupling method of Example 19, coupling with intermediate 19-1, followed by alkaline hydrolysis, and finally separation and preparation by SFC chiral separation column can obtain four single chiral isomers 22-a of intermediate 22, respectively. , 22-b, 22-c, 22-d. MS m / z: 448 [M + 1] + .
- the intermediate 23-1 (2.30 g, 10.30 mmol) was dissolved in EtOH (20 mL). After replacing with nitrogen, 10% Pd / C (0.5 g) was added. The reaction was stirred overnight under a hydrogen atmosphere at atmospheric pressure. Diatomite was filtered with suction, washed with ethanol, and the filtrate was concentrated to dryness under reduced pressure.
- the intermediate 23-2 (1.30 g, 6.73 mmol, 65.31% yield) was isolated and purified on a silica gel column. MS m / z: 194 [M + 1] + .
- the intermediate 23-3 (3.45 g, 12.32 mmol) was dissolved in 20 ml of ethanol, SOCl 2 (4.40 g, 36.96 mmol, 2.68 mL) was added, and the mixture was heated to 50 ° C. and stirred for 1 hour.
- LC-MS showed that the starting materials disappeared and the reaction solution Concentrated to dryness under reduced pressure, added CH 2 Cl 2 (150 mL) and H 2 O (150 mL), adjusted the pH to 8 with saturated NaHCO 3 , and extracted the aqueous phase with CH 2 Cl 2 (2 * 150 mL).
- the intermediate 23-4 (2.89 g, 12.07 mmol) was dissolved in 10 ml of ethanol, and Pd / C (0.5 g) was added under a nitrogen atmosphere. The reaction was hydrogenated at normal pressure overnight. After the raw materials disappeared, the solution was filtered through suction through celite. The filtrate was concentrated to dryness under reduced pressure, and purified by MPLC C18 reverse-phase column to obtain intermediate 23 (2.13 g, 10.26 mmol, 85% yield), MS m / z: 209 [M + 1] + .
- the intermediate 26-1 of step 1 is used as the raw material of step 1 to reduce the p-nitro group, meta-nitrogenate and p-amino group at the same time, and then deacetylate and hydrogenate to obtain the intermediate.
- Example 23 using 2-F-4-nitrophenyl ethyl acetate as a starting material, after benzyl dimethylation with methyl iodide under DMF-cesium carbonate catalysis, the para-nitro group was reduced, Intermediate nitration and para-amino acetylation at the same time, and then deacetylation, hydrogenation reduction to obtain intermediate 27, MS m / z: 241 [M + 1] + .
- Example 23 ethyl 2-fluoro-4-nitrophenylacetate was used as a starting material, and benzyl dimethylation was performed with methyl iodide under DMF-cesium carbonate catalysis, and the para nitro group was reduced.
- the meta (3-position) is nitrated and para-amino acetylated at the same time, then deacetylated and hydrogenated to obtain intermediate 28, MS m / z: 241 [M + 1] + .
- Example 23 ethyl 2-fluoro-4-nitro-5-bromophenylacetate was used as a starting material, and after benzyl dimethylation with methyl iodide under the catalyst of DMF-cesium carbonate, 4- Nitro reduction, meta (3-position) nitration and para-amino acetylation at the same time, then deacetylation, hydrogenation reduction and simultaneous removal of 5-position bromine can obtain intermediate 29, MS m / z: 241 [M + 1] + .
- the intermediate 30-1 (15 g, 67.49 mmol) was dissolved in ethanol (150 mL), NaOH (4.05 g, 101.24 mmol) was added, and the mixture was heated to 80 ° C for 2 hours.
- LC-MS showed that the raw materials had been reacted, and the reaction solution was poured. It was poured into water and extracted with DCM (100 ml * 3). The organic phases were combined, and the organic phases were washed with saturated brine, dried over anhydrous sodium sulfate, filtered, and concentrated to dryness under reduced pressure to obtain a crude intermediate 30-2 (10 g, 49.94 mmol, 74.00). % Yield), used directly in the next step, MS m / z: 181 [M + 1] + .
- intermediate 30-2 is hydrogenated and reduced by Pd / C, and then reverse-phase purified by MPLC to obtain intermediate 30, MS m / z: 151 [M + 1] + .
- the intermediate 34-1 (3.20 g, 13.55 mmol) was dissolved in 30 ml of ethanol, and Pd / C (0.2 g) was added under a nitrogen atmosphere. The hydrogenation reaction was carried out at normal pressure overnight. After the raw materials disappeared, the mixture was suction filtered through celite. The filtrate was concentrated to dryness under reduced pressure, and purified by MPLC C18 reverse-phase column to obtain intermediate 34-2 (1.76 g, 8.53 mmol, 62.95% yield), MS m / z: 207 [M + 1] + .
- the intermediate 34-2 (640.00mg, 3.10mmol) was dissolved in 30ml dissolved in methanol, and PtO2 (60.00mg) was added under a nitrogen atmosphere. The reaction was hydrogenated under normal pressure overnight. After the raw materials disappeared, the solution was filtered through celite and the filtrate was reduced. It was concentrated to dryness under pressure and purified by MPLC C18 reverse-phase column to obtain intermediate 34 (620.00mg, 2.98mmol, 96.13% yield). MS m / z: 209 [M + 1] + .
- Example 23 can be obtained by palladium-carbon reduction method, MS m / z: 204 [M + 1] + .
- step 2 of Example 18 first, after coupling 2-nitro-4-bromoaniline with pinacol borate 37-1, and then referring to the reduction method of nitropalladium carbon in Example 23, intermediate 37 can be obtained. , MS m / z: 261 [M + 1] + .
- step 2 of Example 18 the pinacol borate 38-1 obtained in step 1 was coupled with 38-2 obtained in step 2 to obtain intermediate 38, MS m / z: 246 [M + 1] + .
- the intermediate 40-2 (0.5 g, 1.70 mmol) was dissolved in 10 ml of DCM, and HBTU (647.29 mg, 2.55 mmol), DIEA (1.10 g, 8.50 mmol), and methylamine (105.58 mg, 3.40 mmol) were sequentially added at room temperature. ), The reaction solution was stirred at 50 ° C for 10 hours, concentrated under reduced pressure, and extracted with water and ethyl acetate. The organic phase was dried and concentrated under reduced pressure, and then purified by silica gel column chromatography (DCM / MeOH: 20/1). Intermediate 40-3 (80mg, 260.42umol, 15.32% yield), MS m / z: 308 [M + 1] + .
- step 2 of Example 18 first, after coupling 2-nitro-4-bromoaniline with pinacol borate 40-3, and then referring to the reduction method of nitropalladium carbon in Example 23, intermediate 40 can be obtained. , MS m / z: 288 [M + 1] + .
- the intermediate 41-5 (800 mg, 1.82 mmol) was dissolved in 10 mL of THF-H2O (1: 1), LiOH (438 mg, 18.3 mmol) was added, and the mixture was stirred at room temperature for 3 hours. The pH was adjusted to 5 through 1N HCl. Acetic acid Extraction with ethyl acetate and concentration of the organic phase gave the crude intermediate 41-6 (500 mg, 1.18 mmol, 64.5% yield), which was used in the next step without purification. MS m / z: 424 [M + 1] + .
- the intermediate 41-6 is condensed with D-leucine tert-butyl ester hydrochloride to obtain intermediate 42-1, MS m / z: 593 (M + 1) + .
- the intermediate 41-6 is condensed with isoamylamine to obtain compound 43, MS m / z: 493 (M + 1) + .
- the intermediate 41-3 is sequentially condensed with the o-phenylenediamine 30 of Example 30, and the imidazole ring can be used to obtain compound 45.
- the intermediate 41-3 is condensed with o-phenylenediamine 23 of Example 23, the imidazole ring, the ethyl ester is hydrolyzed, and finally condensed with methylamine to obtain compound 46, MS m / z: 451 [M + 1] + .
- step 4-7 of Example 41 the intermediate 41-3 and the o-phenylenediamine 24 of Example 24 are condensed in sequence, the imidazole ring, the ethyl ester is hydrolyzed, and finally the compound 47 is condensed with methylamine to obtain compound 47, MS m / z: 465 [M + 1] + .
- the intermediate 41-3 is condensed with the ortho-phenylenediamine 25 of Example 25, the imidazole ring, the ethyl ester is hydrolyzed, and finally condensed with methylamine to obtain compound 48, MS m / z: 479 [M + 1] + .
- the intermediate 41-3 is condensed with the intermediate o-phenylenediamine 33, the imidazole ring, the ethyl ester is hydrolyzed, and finally condensed with methylamine to obtain compound 49, MS m. / z: 505 [M + 1] + .
- the intermediate 41-3 is condensed with the ortho-phenylenediamine 31 of Example 31, the imidazole ring, the ethyl ester is hydrolyzed, and finally the compound 50 is obtained by condensation with methylamine. / z: 463 [M + 1] + .
- the intermediate 41-3 is sequentially condensed with the o-phenylenediamine 32 of Example 32, the imidazole ring, the ethyl ester is hydrolyzed, and finally the compound 51 is condensed with methylamine to obtain compound 51, MS m / z: 477 [M + 1] + .
- the intermediate 41-3 is condensed with the intermediate o-phenylenediamine 26, the imidazole ring, the ethyl ester is hydrolyzed, and finally condensed with methylamine to obtain compound 52, MS m. / z: 465 [M + 1] + .
- the intermediate 41-3 is condensed with the intermediate 2-bromo-4,5-o-phenylenediamine ethyl acetate in sequence (from 2-bromo-4-nitrophenyl ethyl acetate) Obtained by referring to the method of steps 2 to 5 of Example 23), the imidazole ring, ethyl ester is hydrolyzed, and finally condensed with methylamine to obtain compound 53, MS m / z: 515 [M + 1] + .
- the intermediate 56-2 can be obtained by hydrolysis of the intermediate 56-1 with lithium hydroxide, MS m / z: 171 [M + 1] + .
- step 2-7 of Example 41 similarly, (S)-(-)-cyclohexylalanine methyl ester is condensed with intermediate 56-2, methyl ester is hydrolyzed, and condensed with intermediate 26, and the imidazole ring is condensed. Ethyl ester is hydrolyzed and finally condensed with methylamine to obtain compound 56, MS m / z: 495 [M + 1] + .
- the intermediate 57-1 (structural isomer mixture) can be obtained by condensing intermediate 15 and intermediate 26, MS m / z: 476 (M + 1) + . In the next step.
- the intermediate 57-1 (structural isomer mixture) is heated to form an imidazole ring in acetic acid to obtain the intermediate 57-2, MS m / z: 458 (M + 1) + .
- the intermediate 57-4 can be obtained by condensing intermediate 57-3 with 1-methyl-1H-pyrazole-5-carboxylic acid. MS m / z: 466 (M + 1) + .
- the intermediate 57-4 was hydrolyzed by LiOH to obtain the intermediate 57-5, MS m / z: 438 (M + 1) + .
- the intermediate 41-3 is condensed with the intermediate 34 of Example 34 in sequence, the imidazole ring, the ethyl ester is hydrolyzed, and the D-leucine tert-butyl ester hydrochloride is condensed.
- Butyl ester can be hydrolyzed by trifluoroacetic acid to obtain compound 60, MS m / z: 551 [M + 1] + .
- the intermediate 41-3 is condensed with the intermediate o-phenylenediamine 26, the imidazole ring, the ethyl ester is hydrolyzed, and the D-leucine tert-butyl ester hydrochloride is condensed in sequence.
- compound 61 can be obtained by hydrolysis of tert-butyl ester with trifluoroacetic acid. MS m / z: 565 [M + 1] + .
- the intermediate 41-3 is condensed with the intermediate o-phenylenediamine 35 of Example 35, and the imidazole ring can be obtained as compound 64.
- the intermediate 65-2 can be obtained by coupling the pinacol borate 38-1 with the bromo compound 65-1 obtained in step 2 of Example 65, MS m / z: 378 [M + 1] + .
- Example 57 Referring to Steps 1-6 of Example 57, the intermediate 15 of Example 15 and the intermediate 65-2 of Example 65 are sequentially condensed, the imidazole ring is removed, the Boc is removed, the 2-methyl-1H-pyrazolyl group is introduced, and then hydrogenated. Cbz protecting group can obtain compound 65, MS m / z: 487 [M + 1] + .
- Example compound 65 (20 mg, 41.10umol) was dissolved in 1 ml of DCM, and a solution of cyclopentyl isocyanate (4.57 mg, 41.10 umol) in 1 ml of dichloromethane was added dropwise. After the drop was completed, the reaction was performed at room temperature for 4 hours, and the crude product was concentrated Compound 66 was obtained through MPLC reversed-phase C18 column purification, MS m / z: 598 [M + 1] + .
- the single stereoisomer 14a of the intermediate 14 of Example 14 is condensed with the intermediate 41-1 of Example 41, the imidazole ring is removed, the Boc is removed, and 1-ethyl-1H-pyridine is introduced.
- Azole-5-acyl, ester is hydrolyzed and finally condensed with methylamine hydrochloride to obtain compound 68, MS m / z: 548 [M + 1] + .
- the single stereoisomer 14d of intermediate 14 of example 14 is condensed with the intermediate 41-1 of example 41, the imidazole ring is removed, the Boc is removed, and 1-ethyl-1H-pyridine is introduced.
- the azole-5-acyl, ester is hydrolyzed and finally condensed with methylamine hydrochloride to obtain compound 69, MS m / z: 548 [M + 1] + .
- the single stereoisomer 14b of intermediate 14 of example 14 is condensed with the intermediate 41-1 of example 41, the imidazole ring is removed, the Boc is removed, and 1-ethyl-1H-pyridine is introduced.
- the azole-5-acyl, ester is hydrolyzed and finally condensed with methylamine hydrochloride to obtain compound 70, MS m / z: 548 [M + 1] + .
- the single stereoisomer 14c of the intermediate 14 of Example 14 is condensed with the intermediate 41-1 of Example 41, the imidazole ring is removed, the Boc is removed, and 1-ethyl-1H-pyridine is introduced.
- the azole-5-acyl, ester is hydrolyzed and finally condensed with methylamine hydrochloride to obtain compound 71, MS m / z: 548 [M + 1] + .
- the intermediate 73-1 (structural isomer mixture) was obtained by condensing o-chloro-D-Boc-phenylalanine with intermediate 34 as the starting material, MS m / z: 490 ( M + 1) + , the two need not be separated and used in the next step.
- the intermediate 73-2 (structural isomer mixture) is heated to form an imidazole ring in acetic acid to obtain the intermediate 73-2, MS m / z: 472 (M + 1) + .
- the intermediate 74-2 (29.0 g, 139.3 mmol) was dissolved in methanol (300 mL), palladium on carbon (2.9 g, palladium content 10%) was added, the reaction system was replaced with hydrogen three times, and stirred under a hydrogen atmosphere (1 atm) for 12 hour. The reaction was filtered through celite and the solvent was evaporated under reduced pressure to obtain intermediate 74-3 (22.4 g, 125.4 mmol, yield 90%). MS m / z: 179 [M + 1] + .
- the intermediate 74-3 (22.4g, 125.4mmol) was dissolved in tetrahydrofuran (300mL), and a 1M borane tetrahydrofuran solution (627.0mL, 627.0mmol) was added dropwise under an ice bath. After the dropwise addition, the reaction was stirred at 60 ° C for 12 hours under nitrogen protection. .
- the intermediate 74-6 (22.3 g, 46.7 mmol) was dissolved in ethanol (400 mL), sodium hydroxide (1.9 g, 46.7 mmol) was added in portions at room temperature, and the temperature was raised to 70 ° C. and the reaction was stirred for 1 hour. It was then extracted with water and ethyl acetate, and the aqueous phase was extracted twice with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure to obtain the crude intermediate 74-7 (15.4 g, 44.8 mmol, Yield: 96%). MS m / z: 344 [M + 1] + .
- the compound 74 was obtained by condensing intermediate 74-10 with acetyl chloride, MS m / z: 535 (M + 1) + .
- Example 74 the single stereoisomer 2-d of the intermediate 2 of Example 2 was condensed with the o-phenylenediamine 74-8 of the intermediate of Example 74, and the ring closure, de-Boc, and 1-methyl- 1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with acetyl chloride to obtain compound 75, MS m / z: 535 (M + 1) + .
- Example 74 the single stereoisomer 2-b of the intermediate 2 of Example 2 was condensed with the o-phenylenediamine 74-8 of the intermediate of Example 74, and the ring closure, de-Boc, and 1-methyl- 1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with acetyl chloride to obtain compound 76, MS m / z: 535 (M + 1) + .
- Example 74 using the single stereoisomer 2-a of the intermediate 2 of Example 2 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, and 1-methyl 1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with acetyl chloride to obtain compound 77, MS m / z: 535 (M + 1) + .
- Example 74 using the single stereoisomer 13-a of the intermediate 13 of Example 13 as a raw material, condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, and 1-formaldehyde 1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with acetyl chloride to obtain compound 78, MS m / z: 547 (M + 1) + .1 HNMR (400MHz, DMSO-d 6 ) ⁇ 12.
- Example 74 using the single stereoisomer 13-d of the intermediate 13 of Example 13 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, and 1-formaldehyde 1H-pyrazole-5-acyl, hydrode Cbz, and finally condensed with acetyl chloride to obtain compound 79, MS m / z: 547 (M + 1) + .
- Example 74 using the single stereoisomer 13-b of the intermediate 13 of Example 13 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, and 1-methyl 1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with acetyl chloride to obtain compound 80, MS m / z: 547 (M + 1) + .
- Example 74 using the single stereoisomer 13-c of the intermediate 13 of Example 13 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, and 1-form 1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with acetyl chloride to obtain compound 81, MS m / z: 547 (M + 1) + .1 HNMR (400MHz, DMSO-d 6 ) ⁇ 12.
- Example 74 using the single chiral isomer 81-1b of the intermediate 82-1 of Example 82 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 82, MS m / z: 539 (M + 1) + .
- Example 74 the single chiral isomer 81-1c of the intermediate 82-1 of Example 82 was used as a raw material to undergo condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1-Methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 83, MS m / z: 539 (M + 1) + .
- Example 74 using the single chiral isomer 81-1a of the intermediate 82-1 of Example 82 as the raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 84, MS m / z: 539 (M + 1) + .
- Example 74 With reference to the method of Example 74, a single chiral isomer 81-1d of the intermediate 82-1 of Example 82 was used as a raw material to undergo condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 85, MS m / z: 539 (M + 1) + .
- Example 74 using the single chiral isomer 13-b of the intermediate 13 of Example 13 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 86, MS m / z: 573 (M + 1) + .
- Example 74 using the single chiral isomer 13-c of the intermediate 13 of Example 13 as the raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- -1H- pyrazol-5-methyl group, the compound hydrodehalogenation Cbz, and finally condensed with cyclopropanecarbonyl chloride to obtain 87, MS m / z:.
- Example 74 using the single chiral isomer 13-a of the intermediate 13 of Example 13 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 88, MS m / z: 573 (M + 1) + .
- Example 74 the single chiral isomer 13-d of the intermediate 13 of Example 13 was used as a raw material to undergo condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 89, MS m / z: 573 (M + 1) + .
- Example 74 using the single chiral isomer 19-b of the intermediate 19 of Example 19 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrode Cbz, and finally condensed with acetyl chloride to obtain compound 90, MS m / z: 643 (M + 1) + .
- Example 74 using the single chiral isomer 19-d of the intermediate 19 of Example 19 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with acetyl chloride to obtain compound 91, MS m / z: 643 (M + 1) + .
- Example 74 the single chiral isomer 19-a of the intermediate 19 of Example 19 was used as a raw material to undergo condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with acetyl chloride to obtain compound 92, MS m / z: 643 (M + 1) + .
- Example 74 using the single chiral isomer 19-c of the intermediate 19 of Example 19 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with acetyl chloride to obtain compound 93, MS m / z: 643 (M + 1) + .
- Example 74 using the single chiral isomer 94-1b of the intermediate 94-1 of step 1 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, above 1 -Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 94, MS m / z: 527 (M + 1) + .
- Example 74 With reference to the method of Example 74, the single chiral isomer 94-1c of the intermediate 94-1 of step 1 of Example 94 was used as a raw material to undergo condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, and de-Boc , On 1-methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 95, MS m / z: 527 (M + 1) + .
- Example 74 using the single chiral isomer 94-1a of the intermediate 94-1 of Step 1 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, above 1 -Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 96, MS m / z: 527 (M + 1) + .
- Example 74 using the single chiral isomer 94-1d of the intermediate 94-1 of step 1 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, above 1 -Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 97, MS m / z: 527 (M + 1) + .
- Example 74 using the single chiral isomer 1-b of Intermediate 1 of Example 1 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrobromic acid-acetic acid deCbz, and finally condensed with acetyl chloride to obtain compound 98, MS m / z: 579 (M + 1) + .
- Example 74 using the single chiral isomer 1-c of Intermediate 1 of Example 1 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrobromic acid-acetic acid deCbz, and finally condensed with acetyl chloride to obtain compound 99, MS m / z: 579 (M + 1) + .
- Example 74 using the single chiral isomer 1-c of Intermediate 1 of Example 1 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Ethyl-1H-pyrazole-5-acyl, hydrobromic acid-acetic acid deCbz, and finally condensed with acetyl chloride to obtain compound 100, MS m / z: 627 (M + 1) + .
- Example 74 using the single chiral isomer 1-a of the intermediate 1 of Example 1 as a raw material, condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrobromic acid-acetic acid de-Cbz, and finally condensed with acetyl chloride to obtain 101, MS m / z: 579 (M + 1) + .
- Example 74 using the single chiral isomer 1-d of Intermediate 1 of Example 1 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrobromic acid-acetic acid deCbz, and finally condensed with acetyl chloride to obtain compound 102, MS m / z: 579 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with acetyl chloride to obtain compound 103, MS m / z: 615 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with 3-tetrahydrofurancarboxylic acid to obtain compound 104, MS m / z: 671 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 105, MS m / z: 641 (M + 1) + .
- Example 74 using the single chiral isomer 18-b of the intermediate 18 of Example 18 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 106, MS m / z: 641 (M + 1) + .
- Example 74 using the single chiral isomer 18-a of the intermediate 18 of Example 18 as the raw material, condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 107, MS m / z: 641 (M + 1) + .
- Example 74 the intermediate 18 of Example 18, a single chiral isomer 18-c, was used as a raw material to undergo condensation with o-phenylenediamine 74-8 of Example 74, ring closure, de-Boc, and 1-formaldehyde.
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclobutanoic acid to obtain compound 109, MS m / z: 655 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopentanoic acid to obtain compound 110, MS m / z: 669 (M + 1) + .
- acetaldehyde was used as a starting material, and it was subjected to condensation reaction with ethyl nitroacetate, Grignard reaction with o-chlorophenyl magnesium bromide, reduction of nitrozinc powder, and Boc-protected amino group. , Alkali hydrolysis, and finally separation and preparation by SFC chiral separation column to obtain four single chiral isomers 111-1a, 111-1b, 111-1c, 111-1d of the intermediate 111-1. MS m / z: 314 [M + 1] + .
- the single chiral isomer 111-1c of the intermediate 111-1 was used as a raw material to undergo condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, and 1-formaldehyde 1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 111, MS m / z: 533 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with n-butyric acid to obtain compound 112, MS m / z: 643 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, condensed with 1-Boc-pyrrolidine-3-acetic acid, and finally de-Boc from trifluoroacetic acid to obtain compound 113, MS m / z: 684 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropaneacetic acid to obtain compound 114, MS m / z: 655 (M + 1) + .
- Example 74 using the single chiral isomer 19-c of the intermediate 19 of Example 19 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropaneacetic acid to obtain compound 115, MS m / z: 669 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with 4-methylvaleric acid to obtain compound 116, MS m / z: 671 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with isobutyric acid to obtain compound 117, MS m / z: 643 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with 3-methylbutyric acid to obtain compound 118, MS m / z: 657 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with N-methylaminosulfonyl chloride to obtain compound 120, MS m / z: 666 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with nicotinic acid to obtain compound 121, MS m / z: 678 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with 1-methylpyrazole-4-carboxylic acid to obtain compound 122, MS m / z: 681 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropylchloroformate to obtain compound 123, MS m / z: 657 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with oxazole-2-carboxylic acid to obtain compound 124, MS m / z: 668 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanesulfonyl chloride to obtain compound 125, MS m / z: 677 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensation with N, N-dimethylformyl chloride to obtain compound 126, MS m / z: 644 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with methanesulfonyl chloride to obtain compound 127, MS m / z: 651 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, to obtain amino intermediate, take this intermediate (100mg, 174.48umol), dissolve in 3ml methanol, add ethyl glyoxylate (17.81mg, 174.48umol ), Stirred at 60 ° C for 1 hour, then added NaBH3CN (32.89mg, 523.44umol) and continued stirring at 60 ° C overnight, quenched with water, extracted with ethyl acetate, concentrated under reduced pressure, and the crude product was separated and purified by silica gel column chromatography to obtain Intermediate 128-1, MS m / z: 659 (M + 1) + .
- Example 74 using the single chiral isomer 131-c of the intermediate of Example 131-131 as the raw material, condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 131, MS m / z: 533 (M + 1) + .
- Example 74 a single chiral isomer 11-c of the intermediate 11 of Example 11 was used as a raw material to undergo condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 132, MS m / z: 547 (M + 1) + .
- Example 74 using the single chiral isomer 11-a of the intermediate 11 of Example 11 as the raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 133, MS m / z: 547 (M + 1) + .
- Example 74 using the single chiral isomer 11-d of the intermediate 11 of Example 11 as the raw material, condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 134, MS m / z: 547 (M + 1) + .
- Example 74 using the single chiral isomer 11-b of the intermediate 11 of Example 11 as the raw material, the condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 135, MS m / z: 547 (M + 1) + .
- Example 74 a single chiral isomer 12-a of the intermediate 12 of Example 12 was used as a raw material to undergo condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 137, MS m / z: 560 (M + 1) + .
- Example 74 using the single chiral isomer 12-d of the intermediate 12 of Example 12 as the raw material, condensation with the o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, dehydrogenated Cbz, and finally condensed with cyclopropanoyl chloride to obtain compound 138, MS m / z: 560 (M + 1) + .
- Example 74 using the single chiral isomer 1-c of Intermediate 1 of Example 1 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrobromide deCbz, and finally condensed with cyclopropanoyl chloride to obtain compound 140, MS m / z: 639 (M + 1) + .
- the compound 140 of Example 140 was coupled with 2-fluoropyridine-5-borate to obtain compound 141, MS m / z: 656 (M + 1) + .
- Example 141 Compound 141 was reacted with N, N-dimethylethylenediamine in tetrahydrofuran in the presence of triethylamine to obtain compound 142, MS m / z: 724 (M + 1) + .
- Example 140 was coupled with 1,3-dimethyl-1H-pyrazole-4-boronic acid pinacol ester to obtain compound 144, MS m / z: 655 ( M + 1) + .
- Example 74 using the single chiral isomer 18-b of the intermediate 18 of Example 18 as a raw material, condensation with o-phenylenediamine 74-8 of the intermediate of Example 74, ring closure, de-Boc, upper 1- Methyl-1H-pyrazole-5-acyl, hydrode-Cbz, and finally condensed with methylsulfonyl chloride to obtain compound 145, MS m / z: 666 (M + 1) + .
- Example 23 Referring to the method of Example 23, the ethyl p-nitrophenylacetate was hydrogenated, the nitro group was meta-nitrogenated, deacetylated, and the hydrogenation reduction step was performed to obtain the intermediate 146-1, MS m / z: 195 (M + 1 ) + .
- Example 74 using the single chiral isomer 2-a of Intermediate 2 of Example 2 as a raw material, condensation with o-phenylenediamine 146-1 of the intermediate of Example 146, ring closure, de-Boc, upper 1- Ethyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condensation with methylamine hydrochloride to obtain compound 147, MS m / z: 507 (M + 1) + .
- Example 74 using the single chiral isomer 2-d of Intermediate 2 of Example 2 as the raw material, condensation with o-phenylenediamine 146-1 of the intermediate of Example 146, ring closure, de-Boc, upper 1- Ethyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condensed with methylamine hydrochloride to obtain compound 149, MS m / z: 507 (M + 1) + .
- Example 74 using the single chiral isomer 3-b of Intermediate 3 of Example 3 as a raw material, condensation with o-phenylenediamine 146-1 of the intermediate of Example 146, ring closure, de-Boc, upper 1- Ethyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condense with methylamine hydrochloride to obtain compound 150, MS m / z: 507 (M + 1) + .
- Example 74 using the single chiral isomer 3-a of Intermediate 3 of Example 3 as a raw material, condensation with o-phenylenediamine 146-1 of the intermediate of Example 146, ring closure, de-Boc, upper 1- Ethyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condensation with methylamine hydrochloride to obtain compound 151, MS m / z: 507 (M + 1) + .
- Example 74 using the single chiral isomer 3-c of the intermediate 3 of Example 3 as a raw material, condensation with the o-phenylenediamine 146-1 of the intermediate of Example 146, ring closure, de-Boc, upper 1- Ethyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condensed with methylamine hydrochloride to obtain compound 152, MS m / z: 507 (M + 1) + .
- Example 74 using the single chiral isomer 3-d of Intermediate 3 of Example 3 as a raw material, condensation with o-phenylenediamine 146-1 of the intermediate of Example 146, ring closure, de-Boc, upper 1- Ethyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condense with methylamine hydrochloride to obtain compound 153, MS m / z: 507 (M + 1) + .
- Example 74 using the single chiral isomer 2-b of Intermediate 2 of Example 2 as a raw material, condensation with o-phenylenediamine 26 of Intermediate of Example 26, ring closure, de-Boc, and 1-ethyl -1H-pyrazole-5-acyl, with reference to step 6-7 of Example 41, alkali hydrolysis, and finally condensation with methylamine hydrochloride to obtain compound 154, MS m / z: 535 (M + 1) + .
- Example 74 using the single chiral isomer 2-a of Intermediate 2 of Example 2 as a raw material, condensation with o-phenylenediamine 26 of Example 26, ring closure, de-Boc, and 1-ethyl -1H-pyrazole-5-acyl, and referring to step 6-7 of Example 41, alkali hydrolysis, and finally condensation with methylamine hydrochloride to obtain compound 155, MS m / z: 535 (M + 1) + .
- Example 74 using the single chiral isomer 2-c of the intermediate 2 of Example 2 as a raw material, condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-ethyl -1H-pyrazole-5-acyl, referring to step 6-7 of Example 41, alkali hydrolysis, and finally condensation with methylamine hydrochloride to obtain compound 156, MS m / z: 535 (M + 1) + .
- Example 74 using the single chiral isomer 2-d of Intermediate 2 of Example 2 as a raw material, condensation with o-phenylenediamine 26 of Intermediate of Example 26, ring closure, de-Boc, and 1-ethyl -1H-pyrazole-5-acyl, and then refer to step 6-7 of Example 41, alkali hydrolysis, and finally condensation with methylamine hydrochloride to obtain compound 157, MS m / z: 535 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as a raw material, condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-ethyl -1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condensation with methylamine hydrochloride to obtain compound 158, MS m / z: 615 (M + 1) + .
- Example 74 using the single chiral isomer 21-c of the intermediate 21 of Example 21 as the raw material, the condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-ethyl -1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, alkali hydrolysis, and finally condensed with methylamine hydrochloride to obtain compound 159, MS m / z: 642 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as the raw material, condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-methyl -1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali to obtain compound 160, MS m / z: 701 (M + 1) + .
- Example 74 using the single chiral isomer 18-d of the intermediate 18 of Example 18 as a raw material, the condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-methyl -1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, after hydrolysis with base, and condense with D-leucine methyl ester hydrochloride, and finally hydrolyze methyl ester with base to obtain compound 161, MS m / z: 701 (M + 1) + .
- Boc-D-leucine (2.49 g, 10 mmol) was dissolved in DMF (25 mL), and HBTU (4.17 g, 11 mmol), DBU (2.58 g, 20 mmol) and methylsulfonamide (1.9 g, 20 mmol) were added under ice bath. ), And the mixture was allowed to rise to room temperature and stirred for 5 hours. It was then extracted with water and ethyl acetate, and the aqueous phase was extracted twice with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure to obtain a crude intermediate 162-1 (1.5 g, 6.5 mmol, Yield 50%). MS m / z: 309 (M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as the raw material, condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-methyl -1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with base, and finally condensed with intermediate 162-2 of step 2 of Example 162 to obtain compound 162, MS m / z: 778 ( M + 1) + .
- Example 74 using the single chiral isomer 18-c of the intermediate 18 of Example 18 as the raw material, condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-methyl -1H-pyrazole-5-acyl, with reference to step 6-7 of Example 41, followed by alkali hydrolysis, and finally condensing with intermediate 164-1 of step 1 of Example 163 to obtain compound 164, MS m / z: 804 ( M + 1) + .
- Boc-D-leucine (2.49 g, 10 mmol) was dissolved in DMF (25 mL), and HBTU (4.17 g, 11 mmol), DBU (2.58 g, 20 mmol), and ammonium chloride (1.07 g, 20 mmol) were added under an ice bath. After the addition, the temperature was raised to room temperature and the reaction was stirred for 5 hours. It was then extracted with water and ethyl acetate, and the aqueous phase was extracted twice with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure to obtain a crude intermediate 165-1 (1.5 g, 6.5 mmol, Yield 50%). MS m / z: 231 (M + 1) + .
- the single chiral isomer 94-1c of the intermediate 94-1 of step 1 of Example 94 was used as a raw material to undergo condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, hydrolyzed by alkali, and then condensed with D-leucine tert-butyl ester hydrochloride, and finally tert-butyl ester was subjected to trifluoro Hydrolysis with acetic acid gives compound 168, MS m / z: 587 (M + 1) + .
- the single chiral isomer 94-1c of the intermediate 94-1 of step 1 of Example 94 was used as a raw material to undergo condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, hydrolyzed by base, and then condensed with intermediate 162-2 of Example 162 to obtain compound 169, MS m / z: 664 (M + 1) + .
- Example 74 using Boc-L-3,3-diphenylalanine as a raw material, condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, and 1-methyl-1H -Pyrazole-5-acyl, refer to step 6-7 of Example 41, hydrolyzed with alkali, and then condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali to obtain compound 170, MS m / z: 621 (M + 1) + .
- Example 74 using the single chiral isomer 171-1c of the intermediate 171-1 of step 171 in Example 171 as a raw material, the condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali Compound 171 was obtained, MS m / z: 641 (M + 1) + .
- the single chiral isomer 171-1b of the intermediate 171-1 of step 171 of Example 171 was used as a raw material to condense with the intermediate o-phenylenediamine 26 of Example 26, the ring was closed, and the Boc was removed.
- 1-methyl-1H-pyrazole-5-acyl refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali Compound 172 was obtained, MS m / z: 641 (M + 1) + .
- the single chiral isomer 173-2a of the intermediate 173-2 of step 173 of Example 173 was used as a raw material to undergo condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali Compound 173 was obtained, MS m / z: 585 (M + 1) + .
- the single chiral isomer 173-2b of the intermediate 173-2 of step 173 of Example 173 was used as a raw material to undergo condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali Compound 174 was obtained, MS m / z: 585 (M + 1) + .
- Example 74 using the single chiral isomer 175-1a of the intermediate 175-1 of step 2 of Example 173 as a raw material, the condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, 1-methyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali Compound 175 was obtained, MS m / z: 619 (M + 1) + .
- Example 74 using the single chiral isomer 175-1b of the intermediate 175-1 in step 2 of Example 173 as a raw material, the condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, upper 1-methyl-1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with D-leucine methyl ester hydrochloride, and finally hydrolyzed methyl ester with alkali Compound 176 was obtained, MS m / z: 619 (M + 1) + .
- Example 74 using the intermediate 19 (mixture of the enantiomers 19-c and 19-d) of Example 19 as the raw material, the condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, and de-Boc, Add 1-methyl-1H-pyrazole-5-acyl, and then refer to step 6-7 of Example 41. After alkaline hydrolysis, then condense with D-leucine methyl ester hydrochloride, and finally hydrolyze methyl ester by alkali.
- Example 74 using the single chiral isomer 19-c of the intermediate 19 of Example 19 as the raw material, the condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-methyl -1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with D-leucine methyl ester hydrochloride, and then hydrolyzed and ammonolyzed to obtain compound 180.
- Example 74 using the single chiral isomer 19-c of the intermediate 19 of Example 19 as the raw material, the condensation with the o-phenylenediamine 26 of the intermediate of Example 26, ring closure, de-Boc, and 1-methyl -1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, and then hydrolyzed with alkali, then condensed with DN-methyl-gamma-hydroxyleucine methyl ester, and finally ester hydrolyzed to obtain the compound 181, MS m / z:.
- the intermediate 1-4 is coupled with 1-cyclopropylpyrazole-4-boronic acid linalyl ester, and then hydrolyzed, and finally separated and prepared by SFC chiral separation column.
- the four single chiral isomers of intermediate 185-1 are 185-1a, 185-1b, 185-1c, 185-1d.
- Example 74 using the single chiral isomer 185-1c of the intermediate 185-1 as a raw material, condensation with the intermediate o-phenylenediamine 26 of Example 26, ring closure, de-Boc, and 1-methyl- 1H-pyrazole-5-acyl, refer to step 6-7 of Example 41, hydrolyzed by alkali, and then condensed with D-leucine methyl ester hydrochloride, and finally ester hydrolyzed to obtain compound 185, MS m / z : 727 (M + 1) + .
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Urology & Nephrology (AREA)
- Diabetes (AREA)
- Immunology (AREA)
- Rheumatology (AREA)
- Pulmonology (AREA)
- Epidemiology (AREA)
- Dermatology (AREA)
- Endocrinology (AREA)
- Heart & Thoracic Surgery (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Physical Education & Sports Medicine (AREA)
- Cardiology (AREA)
- Ophthalmology & Optometry (AREA)
- Vascular Medicine (AREA)
- Emergency Medicine (AREA)
- Pain & Pain Management (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Gastroenterology & Hepatology (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Pyridine Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
Abstract
公开了一种免疫调节剂,具体涉及一类抑制IL-17A的化合物及其作为免疫调节剂在制备药物中的用途。公开了式I所示的化合物、或其立体异构体、或其药学上可接受的盐在制备抑制IL-17A类药物中的用途,为临床上筛选和/或制备与IL-17A活性相关的疾病的药物提供了一种新的选择。
Description
本发明涉及一种免疫调节剂及其在制备药物中的用途。
IL-17(白细胞介素-17)是促炎性细胞因子,在诱导其他炎性细胞因子、趋化因子和粘附因子中发挥作用。IL-17家族由参与急性和慢性炎症反应的细胞因子组成,包括IL-17A(CTLA-8)、IL-17B、IL-17C、IL-17D、IL-17E(IL-25)和IL-17F。IL-17A由TH17细胞表达,其参与炎症和自身免疫性疾病的病理发生。人类IL-17A是分子量约为17000道尔顿的糖蛋白。IL-17A通过IL-17受体复合物(IL-17RA和IL-17RC)将信号传送至细胞内(Wright,et al.Journal of immunology,2008,181:2799-2805)。IL-17A的主要功能是通过促炎和嗜中性粒细胞迁移细胞因子和趋化因子(包括IL-6,G-CSF,TNF-α,IL-1,CXCL1,CCL2,CXCL2)的上调来协调局部组织炎症,以及基质金属蛋白酶来允许活化的T细胞穿透细胞外基质。有研究表明IL-17A在严重哮喘和慢性阻塞性肺疾病(COPD)中发挥重要作用,那些患者通常对目前可用的药物无响应或响应不良(Al-Ramli et al.J Allergy Clin Immunol,2009,123:1185-1187)。IL-17A水平上调涉及许多疾病,包括类风湿性关节炎(RA)、骨侵蚀、腹膜内脓肿、炎性肠病、同种异体移植物排斥反应、牛皮癣、动脉粥样硬化、哮喘和多发性硬化症(Gaffen,SL et al.Arthritis Research&Therapy,2004,6:240-247)。
靶向IL-17A与IL-17RA的结合是治疗IL-17A介导的自身免疫性炎性疾病的有效策略。通过IL-17A中和抗体治疗动物在自身免疫性脑脊髓炎中降低疾病发病率和严重性(Komiyama Y et al.J.Immunol.,2006,177:566-573)。已有IL-17A抗体的临床试验在IL-7A介导的炎性疾病(包括哮喘、牛皮癣、类风湿性关节炎、强直性脊柱炎和多发性硬化症)上显示出良好的结果。IL-17A抗体(Novartis的Cosentyx/secukinumab)在2015年1月已被FDA批准用于牛皮癣的治疗。
尽管存在多种IL-17A抗体,但很少有对具有口服生物利用度的IL-17的小分子特异性抑制剂进行研究。鉴于产生抗体的成本考虑和给药途径的限制,开发IL-17A小分子抑制剂药物具有良好的研发前景。
发明内容
本发明提供了式I所示的化合物、或其立体异构体、或其药学上可接受的盐:
其中,
X选自O、S、NR
1’;
R
1、R
2分别独自选自氢、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环、-S(O)
2R
6、-S(O)R
6、-S(O)
2NR
6R
7、-S(O)NR
6R
7、-C(O)R
6、-C(O)OR
6、-C(O)NR
6R
7、-P(O)(OR
7)R
6、-P(O)R
6R
7、-P(O)R
6(NR
6R
7)、-P(O)(NR
6R
7)(NR
6R
7)、-P(O)(OR
7)(NR
6R
7)、P(O)(OR
7)(OR
7);其中烷基、烯基、炔基、环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
R
6、R
7分别独自选自氢、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环;其中烷基、烯基、炔基、环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
R
3、R
3’分别独自选自氢、-OR
d、-SR
d、-NR
dR
d、卤素、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环;其中R
d、烷基、烯基、炔基、环烷基、杂环烷基、芳环被m个R
c取代;或者,R
3和R
3’连接成3~10元环烷基、3~10元杂环烷基;
R
d选自氢、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;
B环选自3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;其中环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
R
1’选自氢、C
1~10烷基;
r为0、1、2、3;
R
8和R
9分别独自选自氢、卤素、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元的环烷基、3~10元的杂环烷基;其中烷基、烯基、炔基、环烷基、杂环烷基被m个R
c取代;
或者,R
8和R
9相连形成3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;其中环烷基、杂环烷基、芳环、杂芳环被m个R
c取代;
L
2选自-C(O)NR
10-、-NR
10C(O)-、-C(O)-、-C(O)O-、-S(O)
2NR
10-、-S(O)NR
10-、-NR
10S(O)
2-、-NR
10S(O)-、-P(O)NR
10-、-NR
10P(O)-或无;
R
10选自氢、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元环烷基、3~10元杂环烷基,其中烷基、烯基、炔基、环烷基、杂环烷基被m个R
c取代;
R
4和R
5分别独自选自氢、卤素、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环、-CN、-NO
2、-OR
a、-OC(O)R
a、-OC(O)NR
aR
b、-OS(O)
2R
a、-SR
a、-S(O)
2R
a、-S(O)
2NR
aR
b、-OS(O)
2NR
aR
b、-C(O)R
a、-C(O)OR
a、-C(O)NR
aR
b、-NR
aR
b、-NR
aC(O)R
b、-NR
aC(O)OR
b、-NR
aC(O)NR
aR
b、-NR
aS(O)
2R
b、-NR
aS(O)
2NR
aR
b;其中R
a、R
b、烷基、烯基、炔基、环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
或者,R
4和R
5相连形成3~10元环烷基、3~10元杂环烷基;其中环烷基、杂环烷基被m个R
c取代;
X
1选自CR
11或N;
X
2选自NR
12、O、S或-(CR
13=CR
14)-;
X
4选自N或CH;
R
11选自氢、卤素、-CN、-NO
2、C
1~10烷基、C
2~10烯基、C
2~10炔基、C
1~10卤素烷基、3~10元的环烷基、3~10元的杂环烷基、-OR
a、-NR
aR
b;
R
12选自氢、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元的环烷基、3~10元的杂环烷基、-S(O)
2R
a、-S(O)
2NR
aR
b、-C(O)R
a、-C(O)OR
a、-C(O)NR
aR
b;
R
13、R
14分别独自选自氢、卤素、-CN、-NO
2、C
1~10烷基、C
2~10烯基、C
2~10炔基、C
1~10卤素烷基、3~10元的环烷基、3~10元的杂环烷基、-OR
a、-NR
aR
b;其中烷基、烯基、炔基、环烷基、杂环烷基被m个R
c取代;
m为0、1、2、3、4;
R
a、R
b分别独自选自氢、C
1~10烷基、C
2~10烯基、C
2~10炔基、-S(O)
2R
d、-S(O)R
d、-P(O)
2R
d、-P(O)R
d、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;其中,烷基、烯基、炔基、环烷基、杂环烷基、芳环、杂芳环被m个R
c取代;
R
c分别独自选自C
1~10烷基、=S、=O、卤素、-CN、-NO
2、-OR
a、-OC(O)R
a、-OS(O)
2R
a、-SR
a、-S(O)
2R
a、-S(O)
2NR
aR
b、-C(O)R
a、-C(O)OR
a、-C(O)NR
aR
b、-NR
aR
b、-NR
aC(O)R
b、-NR
aS(O)
2R
b、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环,其中, 烷基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环被0~3个C
1~10烷基、卤素、-NR
aR
b、-OR
d、-C(O)NR
aR
b、3~10元杂环烷基、5~10元芳环、5~10元芳杂环取代。
进一步地,
R
1、R
2分别独自选自氢、C
1~6烷基、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环、-S(O)
2R
6、-S(O)R
6、-S(O)
2NR
6R
7、-C(O)R
6、-C(O)OR
6、-C(O)NR
6R
7;其中烷基、环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
R
6、R
7分别独自选自氢、C
1~6烷基、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环;其中烷基、环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
R
3、R
3’分别独自选自氢、-OR
d、卤素、C
1~6烷基、3~10元环烷基、3~10元杂环烷基、5~10元芳环;其中烷基、环烷基、杂环烷基、芳环被m个R
c取代;或者,R
3和R
3’连接成3~10元环烷基;
R
d选自氢、C
1~10烷基、C
2~10烯基、C
2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;
R
8和R
9分别独自选自氢、卤素、C
1~6烷基、3~10元的环烷基、3~10元的杂环烷基;其中烷基,环烷基、杂环烷基被m个R
c取代;
或者,R
8和R
9相连形成3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元杂芳环;其中环烷基、杂环烷基、芳环、杂芳环可进一步被m个R
c取代;
R
10选自氢、C
1~6烷基、3~10元环烷基、3~10元杂环烷基,其中烷基、环烷基、杂环烷基被m个R
c取代;;
R
4和R
5分别独自选自氢、卤素、C
1~6烷基、3~10元的环烷基、3~10元的杂环烷基、6~10元芳环、5~10元芳杂环、-CN、-NO
2、-OR
a、-OC(O)R
a、-C(O)R
a、-C(O)OR
a、-C(O)NR
aR
b、-NR
aR
b、-NR
aC(O)R
b、-NR
aC(O)NR
aR
b、-NR
aS(O)
2R
b、-NR
aS(O)
2NR
aR
b、-S(O)
2R
a、-S(O)
2NR
aR
b;其中烷基、环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
或者,R
4和R
5相连形成3~10元的环烷基、3~10元的杂环烷基;其中环烷基、杂环烷基被m个R
c取代;
R
11选自氢、卤素、C
1~6烷基;
R
12选自氢、C
1~6烷基;
R
13、R
14分别独自选自氢、卤素、C
1~6烷基、C
2~6烯基;其中烷基、烯基被m个R
c取代;
m为0、1、2、3;
R
a、R
b分别独自选自氢、C
1~6烷基、C
2~6烯基、C
2~6炔基、-S(O)
2R
d、-S(O)R
d、-P(O)
2R
d、-P(O)R
d 3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元杂芳环;其中,烷基、烯基、炔基、环烷基、杂环烷基、芳环、杂芳环被m个R
c取代。
进一步地,
R
1、R
2分别独自选自氢、-S(O)
2R
6、-S(O)R
6、-S(O)
2NR
6R
7、-C(O)R
6、-C(O)OR
6、-C(O)NR
6R
7;
R
6、R
7分别独自选自氢、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环;其中环烷基、杂环烷基、芳环、芳杂环被m个R
c取代;
R
3、R
3’分别独自选自氢、-OR
d、C
1~6烷基、3~10元环烷基、3~10元杂环烷基、5~10元芳环;其中烷基、环烷基、杂环烷基、芳环被m个R
c取代;或者,R
3和R
3’连接成3~8元环烷基;
R
8和R
9分别独自选自氢、C
1~6烷基、3~10元的环烷基、3~10元的杂环烷基;
或者,R
8和R
9相连形成3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元杂芳环;
R
4和R
5分别独自选自氢、C
1~6烷基、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环、-OR
a、-C(O)R
a、-C(O)OR
a、-C(O)NR
aR
b、-NR
aR
b、-NR
aC(O)R
b、-NR
aC(O)NR
aR
b、-NR
aS(O)
2R
b、-NR
aS(O)
2NR
aR
b、-S(O)
2R
a、-S(O)
2NR
aR
b、;其中烷基环烷基、杂环烷基、芳环、芳杂环可进一步被m个R
c取代;
或者,R
4和R
5相连形成3~10元环烷基、3~10元杂环烷基;其中环烷基、杂环烷基可进一步被m个R
c取代;
R
11选自氢、C
1~6烷基;
R
12选自氢、C
1~6烷基;
R
13、R
14分别独自选自氢、C
1~6烷基、C
2~6烯基;其中烷基、烯基被m个R
c取代;
m为0、1、2。
进一步地,式I所示化合物如式IIa所示:
其中,
R
a’选自C
1~6的烷基。
进一步地,式IIa所示化合物如式IIa-1或IIa-2所示:
其中,
R
a’选自C
1~6的烷基。
进一步地,式IIa-1所示化合物如式IIIa-1或IIIa-2所示:
进一步地,R
5’为卤素、CN;n选自0、1、2;L
2为-C(O)NR
10-、-NR
10C(O)-、-C(O)-、-C(O)O-或无。
进一步地,式IIIa-1或式IIIa-2所示化合物具体为:
进一步地,R
5’为卤素和5~10元杂芳环,其中,5~10元芳杂环可进一步被0~3个C
1~10烷基取代、3~10元杂环烷基、5~10元芳环、5~10元芳杂环取代;
n选自0、1、2、3;L
2为-C(O)NR
10-、-NR
10C(O)-、-C(O)-、-C(O)O-或无。
进一步地,式IIIa-1或式IIIa-2所示化合物具体为:
进一步地,R
5’为卤素和-OR
a;n选自0、1、2、3;L
2为无。
进一步地,式IIIa-1或式IIIa-2所示化合物具体为:
进一步地,式IIa-1所示化合物如式IIIb-1或IIIb-2所示:
其中,
R
a’选自取代或未取代的C
1~6的烷基;R
5’为氢、卤素;n选自0、1、2;L
2为-C(O)NR
10-、-NR
10C(O)-、-C(O)-、-C(O)O-或无。
进一步地,式IIIb-1或IIIb-2所示化合物具体为:
进一步地,式IIa-1所示化合物如式IIIc所示:
R
a’、R
c分别选自C
1~6的烷基;R
5’为氢、卤素;n选自0、1、2;L
2为-C(O)NR
10-、-NR
10C(O)-、-C(O)-、-C(O)O-或无。
进一步地,式IIIc所示化合物具体为:
进一步地,式IIa-2所示化合物如式IIId所示:
其中,
R
a’选自取代或未取代的C
1~6的烷基;R
5’为氢、卤素;n选自0、1、2;L
2为-C(O)NR
10-、-NR
10C(O)-、-C(O)-、-C(O)O-或无。
进一步地,式IIId所示化合物具体为:
进一步地,式I所示化合物如式IIb所示:
其中,
R
a’选自C
1~6的烷基。
进一步地,式IIb所示化合物具体为:
本发明还提供了前述的化合物、或其立体异构体、或其药学上可接受的盐在制备治疗IL-17A介导的疾病的药物中的用途。
本发明所定义的IL-17A介导的疾病是IL-17A在该疾病的病理发生中起重要作用的疾病。IL-17A的主要功能是协调局部组织炎症,从而在各种疾病中起作用。IL-17A介导的疾病包括炎症、自身免疫性疾病、感染性疾病、癌症、癌前期综合征相关的疾病中的一种或几种。
“癌症”或“恶性肿瘤”是指以不受控制的细胞异常增殖为特征的多种疾病中的任何一种,受影响的细胞在局部或通过血流和淋巴系统扩散到其他部位的能力的身体(即转移) 以及许多特征结构和/或分子特征中的任何一个。“癌细胞”是指经历多步骤肿瘤进展的早期,中期或晚期阶段的细胞。癌症包括肉瘤、乳腺癌、肺癌、脑癌、骨癌、肝癌、肾癌、结肠癌和前列腺癌。在一些实施方案中,式I的化合物用于治疗选自结肠癌、脑癌、乳腺癌、纤维肉瘤和鳞状细胞癌的癌症。在一些实施方案中,癌症选自黑素瘤、乳腺癌、结肠癌、肺癌和卵巢癌。在一些实施方案中,所治疗的癌症是转移性癌症。
自身免疫性疾病是由身体对体内正常存在的物质和组织的免疫反应引起的。自身免疫疾病的例子包括心肌炎、狼疮性肾炎、原发性胆汁性肝硬化、牛皮癣、1型糖尿病、格雷夫氏病、腹腔疾病、克罗恩病、自身免疫性中性白细胞减少症、幼年型关节炎、类风湿性关节炎、纤维肌痛、吉兰巴利综合征、多发性硬化症和自身免疫性视网膜病变。本发明的一些实施方案涉及治疗自身免疫疾病如牛皮癣或多发性硬化症。
炎症疾病包括以组织病理性炎症为特征的多种病症。炎性疾病的例子包括寻常性痤疮、哮喘、腹腔疾病、慢性前列腺炎、肾小球性肾炎、炎症性肠病、盆腔炎、再灌注损伤、类风湿性关节炎、结节病、血管炎、房尘螨引起的气道炎症和间质性膀胱炎。炎性疾病与自身免疫性疾病之间存在显著重叠。本发明的一些实施方案涉及炎性疾病哮喘的治疗。免疫系统通常涉及炎症性疾病,在过敏反应和一些肌病中都有表现,许多免疫系统疾病导致异常炎症。IL-17A介导的疾病也包括自身免疫性炎症性疾病。
本发明还提供了一种药物,它是以前述的化合物、或其立体异构体、或其药学上可接受的盐,加上药学上可接受的辅料制备而成的制剂。
本发明中提供的化合物和衍生物可以根据IUPAC(国际纯粹与应用化学联合会)或CAS(化学文摘服务社,Columbus,OH)命名系统命名。
关于本发明的使用术语的定义:除非另有说明,本文中基团或者术语提供的初始定义适用于整篇说明书的该基团或者术语;对于本文没有具体定义的术语,应该根据公开内容和上下文,给出本领域技术人员能够给予它们的含义。
“取代”是指分子中的氢原子被其它不同的原子或分子所替换。
碳氢基团中碳原子含量的最小值和最大值通过前缀表示,例如,前缀C
a~b烷基表明任何含“a”至“b”个碳原子的烷基。因此,例如,“C
1~4烷基”是指包含1~4个碳原子的烷基。
“烷基”是指具有指定数目的成员原子的饱和烃链。例如,C
1~C
6烷基是指具有1至6个成员原子,例如1至4个成员原子的烷基基团。烷基基团可以是直链或支链的。代表性的支链烷基基团具有一个、两个或三个支链。烷基基团可任选地被一个或多个如本文所 定义的取代基取代。烷基包括甲基、乙基、丙基(正丙基和异丙基)、丁基(正丁基、异丁基和叔丁基)、戊基(正戊基、异戊基和新戊基)和己基。烷基基团也可以是其他基团的一部分,所述其他基团为例如C
1~C
6烷氧基。
“环烷基”是指具有3至14个碳原子且没有环杂原子且具有单个环或多个环(包括稠合、桥连和螺环体系)的饱和或部分饱和的环状基团。对于具有不含环杂原子的芳族和非芳族环的多环体系,当连接点位于非芳族碳原子时,适用术语“环烷基”(例如5,6,7,8,-四氢化萘-5-基)。术语“环烷基”包括环烯基基团,诸如环己烯基。环烷基基团的实例包括例如,金刚烷基、环丙基、环丁基、环己基、环戊基、环辛基、环戊烯基和环己烯基。包括多双环烷基环体系的环烷基基团的实例是双环己基、双环戊基、双环辛基等。下面例举并命名两种此类双环烷基多环结构:
双环己基和
双环己基。
“烯基”是指具有2至10个碳原子和在一些实施方案中2至6个碳原子或2至4个碳原子且具有至少1个乙烯基不饱和位点(>C=C<)的直链或支链烃基基团。例如,(Ca-Cb)烯基是指具有a至b个碳原子的烯基基团并且意在包括例如乙烯基、丙烯基、异丙烯基、1,3-丁二烯基等。
“炔基”是指含有至少一个三键的直链一价烃基或支链一价烃基。术语“炔基”还意在包括具有一个三键和一个双键的那些烃基基团。例如,(C2-C6)炔基意在包括乙炔基、丙炔基等。
“卤素”为氟、氯、溴或碘。
“卤素烷基”指烷基中的氢原子可被一个或多个卤素原子取代。例如C
1~4卤素烷基指氢原子被一个或多个卤素原子取代的包含1~4个碳原子的烷基。
“杂环”、“杂环烷基”指包含至少一个杂原子的饱和环或非芳香性的不饱和环;其中杂原子指氮原子、氧原子、硫原子;
“芳杂环”指包含至少一个杂原子的芳香性不饱和环;其中杂原子指氮原子、氧原子、硫原子;
“立体异构体”包括对映异构体和非对映异构体;
术语“药学上可接受的”是指某载体、运载物、稀释剂、辅料,和/或所形成的盐通常在化学上或物理上与构成某药物剂型的其它成分相兼容,并在生理上与受体相兼容。
术语“盐”和“可药用的盐”是指上述化合物或其立体异构体,与无机和/或有机酸和碱形成的酸式和/或碱式盐,也包括两性离子盐(内盐),还包括季铵盐,例如烷基铵盐。 这些盐可以是在化合物的最后分离和纯化中直接得到。也可以是通过将上述化合物,或其立体异构体,与一定数量的酸或碱适当(例如等当量)进行混合而得到。这些盐可能在溶液中形成沉淀而以过滤方法收集,或在溶剂蒸发后回收而得到,或在水介质中反应后冷冻干燥制得。本发明中所述盐可以是化合物的盐酸盐、硫酸盐、枸橼酸盐、苯磺酸盐、氢溴酸盐、氢氟酸盐、磷酸盐、乙酸盐、丙酸盐、丁二酸盐、草酸盐、苹果酸盐、琥珀酸盐、富马酸盐、马来酸盐、酒石酸盐或三氟乙酸盐。
本发明公开的式I所示的新化合物,表现出了良好的IL-17A抑制活性,为临床治疗与IL-17A活性异常相关的疾病提供了一种新的药用可能。
在某些实施方式中,本发明的一种或多种化合物可以彼此联合使用。也可选择将本发明的化合物与任何其它的活性试剂结合使用,用于制备调控细胞功能或治疗疾病的药物或药物组合物。如果使用的是一组化合物,则可将这些化合物同时、分别或有序地对受试对象进行给药。
显然,根据本发明的上述内容,按照本领域的普通技术知识和惯用手段,在不脱离本发明上述基本技术思想前提下,还可以做出其它多种形式的修改、替换或变更。
以下通过实施例形式的具体实施方式,对本发明的上述内容再作进一步的详细说明。但不应将此理解为本发明上述主题的范围仅限于以下的实例。凡基于本发明上述内容所实现的技术均属于本发明的范围。
化合物的结构是通过核磁共振(NMR)和质谱(MS)来确定的。NMR位移(δ)以10-6(ppm)的单位给出。NMR的测定是用(Bruker AvanceIII 400和Bruker Avance 300)核磁仪,测定溶剂为氘代二甲基亚砜(DMSO-d6),氘代氯仿(CDCl
3),氘代甲醇(CD3OD),内标为四甲基硅烷(TMS)。
LC-MS的测定使用岛津液质联用仪(Shimadzu LC-MS 2020(ESI))。
HPLC的测定使用岛津高压液相色谱仪(Shimadzu LC-20A)。
MPLC(中压制备色谱)使用Gilson GX-281反相制备色谱仪。
薄层层析硅胶板用烟台黄海HSGF254或青岛GF254硅胶板,薄层层析分离纯化产品采用的规格是0.4mm~0.5mm。
柱层析一般使用烟台黄海硅胶200~300目硅胶为载体。
本发明的已知的起始原料可以采用或按照本领域已知的方法来合成,或可购买于安耐吉化学、成都科龙化工、韶远化学科技、百灵威科技等公司。
实施例中无特殊说明,反应在氮气氛围下进行。
实施例中无特殊说明,溶液是指水溶液。
实施例中无特殊说明,反应的温度为室温。
实施例中无特殊说明,M是摩尔每升。
室温为最适宜的反应温度,为20℃~30℃。
DMF:是指N,N-二甲基甲酰胺。
DMSO:是指二甲基亚砜。
DIPEA:是指二异丙基乙基胺。
Boc:是指叔丁基氧羰基。
TFA:是指三氟乙酸。
DBU:1,8-二氮杂二环十一碳-7-烯。
HATU:2-(7-偶氮苯并三氮唑)-N,N,N',N'-四甲基脲六氟磷酸酯。
HBTU:O-苯并三氮唑-四甲基脲六氟磷酸盐。
EDCI:1-乙基-(3-二甲基氨基丙基)碳酰二亚胺盐酸盐。
HOAT:1-羟基-7-偶氮苯并三氮唑。
实施例1中间体1的制备
步骤1中间体1-1的制备
将硝基乙酸乙酯(28.5g,130mmol)和5-溴-2-氯苯甲醛(17.3g,130mmol)溶于无水四氢呋喃(400mL),氮气保护下在0℃缓慢滴加四氯化钛(28.5mL,260.0mmol),滴加完毕后继续在0℃搅拌反应1小时。将N-甲基吗啉(57.8mL,520.0mmol)缓慢滴加入反应液,滴加完毕缓慢升至室温,继续搅拌反应2小时。加入蒸馏水淬灭反应,用乙酸乙酯萃取,合并有机相,无水硫酸钠干燥,过滤,减压蒸除溶剂,经柱层析纯化(洗脱剂:石油醚:乙酸乙酯=20:1)后得中间体1-1(23.6g,71mmol,产率54%)。MS m/z 335(M+1)
+。
步骤2中间体1-2的制备
将无水氯化锌(14.4g,,106.0mmol)溶于无水四氢呋喃(100mL),氮气保护下在0℃缓慢滴加2M的异丙基氯化镁四氢呋喃溶液(53mL,106.0mmol),滴加完毕继续在0℃搅拌反应1小时。将得到的异丙基溴化镁锌在0℃氮气保护下缓慢滴加至中间体1-1(23.6g,70.5mmol)的无水四氢呋喃(100mL)溶液中,滴加完毕后继续在0℃搅拌反应2小时。加入饱和氯化铵溶液淬灭反应,用乙酸乙酯萃取,合并有机相,无水硫酸钠干燥,过滤,减压蒸除溶剂,经柱层析纯化(洗脱剂:石油醚:乙酸乙酯=20:1)后得中间体1-2(18.6g,49.1mmol,产率70%)。MS m/z 379(M+1)
+。
步骤3中间体1-3的制备
将中间体1-2(18.6g,,49.1mmol)溶于冰乙酸(300mL),0℃下分批次加入锌粉(16.0g,245.5mmol),升温至室温搅拌反应12小时。过滤除去锌粉,减压蒸除溶剂,经柱层析纯化(洗脱剂:石油醚:乙酸乙酯=10:1)后得中间体1-3(7.6g,21.8mmol,产率44%)。MS m/z 350(M+1)
+.
步骤4中间体1-4的制备
将中间体1-3(3.8g,10.9mmol)溶于二氯甲烷(60mL),0℃下依次滴加三乙胺(4.6mL,32.7mmol)和二碳酸二叔丁酯(3.8mL,16.4mmol),滴加完毕升至室温搅拌反应1小时。减压蒸除溶剂,经柱层析纯化(洗脱剂:石油醚:乙酸乙酯=20:1)后得中间体1-4(4.5g,产率92%)。MS m/z 350(M-99)
+,394(M-55)
+。
步骤5中间体1的制备
将中间体1-4(4.5g,,10.0mmol)溶于四氢呋喃(15mL)、水(15mL)和甲醇(5mL)的混合溶剂中,0℃分批加入氢氧化钠(2.0g,50.0mmol),加完后升至室温搅拌反应4小时。减压蒸除溶剂,冰浴下滴加1N HCl调至pH~4,析出固体,过滤真空干燥后得粗品,粗品经SFC手性拆分柱分离制备分别得到中间体1的四个单一手性异构体1-a,1-b,1-c,1-d,分别0.8g,产率19%。MS m/z 322(M-99)
+,366(M-55)
+。
实施例2中间体2的制备
参照实施例1制备中间体1的方法,以邻氯苯甲醛为起始原料、经缩合、格氏反应、硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体2的四个单一手性异构体2-a,2-b,2-c,2-d。MS m/z:242[M-99]
+,286[M-55]
+。
实施例3中间体3的制备
参照实施例1制备中间体1的方法,以间氯苯甲醛为起始原料、经缩合、格氏反应、硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体3的四个单一手性异构体3-a,3-b,3-c,3-d。MS m/z:242[M-99]
+,286[M-55]
+。
实施例4中间体4的制备
参照实施例1制备中间体1的方法,以2-氯-5-氟苯甲醛为起始原料、经缩合、格氏反应、硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体4的四个单一手性异构体4-a,4-b,4-c,4-d。MS m/z:257[M-99]
+,304[M-55]
+。
实施例5中间体5的制备
参照实施例1制备中间体1的方法,以2-氯-5-甲氧基苯甲醛为起始原料、经缩合、格氏反应、硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体5的四个单一手性异构体5-a,5-b,5-c,5-d。MS m/z:257[M-99]
+,316[M-55]
+。
实施例6中间体6的制备
参照实施例1制备中间体1的方法,以2-氯-5-氰基苯甲醛为起始原料、经缩合、格氏反应、硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体6的四个单一手性异构体6-a,6-b,6-c,6-d。MS m/z:267[M-99]
+,311[M-55]
+。
实施例7中间体7的制备
步骤1中间体7-1的制备
参考实施1中间体1-2的制备方法,由邻氯苯甲醛与硝基乙酸乙酯反应可得到中间体2-1,室温下,将中间体2-1(5g,19.56mmol)溶于甲醇中(50mL),加入甲醇钠(4.23g,78.23mmol),室温下搅拌反应2小时,TLC显示原料消失,减压浓缩除去溶剂,加入饱和氯化铵水溶液20mL,乙酸乙酯(20mL×2)萃取,合并有机相,经水洗,饱和氯化钠洗,无水硫酸钠干燥,过滤,减压浓缩至干,粗品经硅胶柱分离(石油醚/乙酸乙酯4:1)得到中间体7-1(3g,10.43mmol,53.32%yield),淡黄色液体,Rf=0.5(Hexanes/EtOAc=8:1)。
步骤2中间体7-2的制备
将中间体7-1(1.58g,5.48mmol)溶于10mL冰醋酸中,加入锌粉(1.79g,27.41mmol),反应于室温下搅拌反应2小时,抽滤,少量乙酸乙酯洗涤,滤液减压浓缩至干,粗品经硅胶柱分离(石油醚/乙酸乙酯2:1)得到中间体7-2(0.83g,3.22mmol,58.76%yield),淡黄色固体,MS m/z:258[M+1].
步骤3中间体7-3的制备
将中间体7-2(0.83g,3.22mmol)溶于THF(3mL)和水(1mL)的混合溶液中,分别加入NaHCO
3(541.11mg,6.44mmol)和(Boc)
2O(737.20mg,3.38mmol),室温下搅拌反应12小时,加入乙酸乙酯(15mL)和水(15mL)洗涤,有机相经水洗,饱和氯化钠洗,无水硫酸钠干燥,过滤,减压浓缩至干,粗品经硅胶柱分离(石油醚/乙酸乙酯4:1)得到中间体7-3(1.1g,3.07mmol,95.45%yield),淡黄色液体,MS m/z:302[M-55].
步骤4中间体7的制备
将中间体7-3(1.23g,3.44mmol)溶于甲醇(15mL)、水(15mL)的混合溶液中,加入氢氧化锂一水合物(1.20g,28.53mmol),于室温搅拌反应4小时。减压蒸除溶剂,冰浴下滴加0.5N HCl调至pH~4,乙酸乙酯萃取-四氢呋喃5:1萃取,有机相减压浓缩得淡黄色液体中间体7(1.1g,3.34mmol,97.04%yield)。MS m/z 274[M-55].
实施例8中间体8的制备
参照实施例7制备中间体7的方法,中间体2-1与乙醇钠反应、再经硝基还原、Boc保护氨基、水解即可得到中间体8。MS m/z:288[M-55]
+。
实施例9中间体9的制备
参照实施例7制备中间体7的方法,中间体2-1与异丙醇钠反应、再经硝基还原、Boc保护氨基、水解即可得到中间体9。MS m/z:302[M-55]
+。
实施例10中间体10的制备
参照实施例7制备中间体7的方法,中间体2-1与环丙醇在叔丁醇钾存在下反应、再经硝基还原、Boc保护氨基、水解即可得到中间体10。MS m/z:300[M-55]
+。
实施例11中间体11的制备
参照实施例1制备中间体1的方法,以丙醛为起始原料、经与硝基乙酸乙酯缩合、与邻氯苯基溴化镁进行格氏反应、硝基锌粉还原、Boc保护氨基、碱水解,最后经SFC手 性拆分柱分离制备可分别得到中间体11的四个单一手性异构体11-a,11-b,11-c,11-d。MS m/z:228[M-99]
+,272[M-55]
+。
实施例12中间体12的制备
参照实施例1制备中间体1的方法,以环丙甲醛为起始原料、经与硝基乙酸乙酯缩合、与邻氯苯基溴化镁进行格氏反应、硝基锌粉还原、Boc保护氨基、碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体12的四个单一手性异构体12-a,12-b,12-c,12-d。MS m/z:228[M-99]
+,272[M-55]
+。
实施例13中间体13的制备
参照实施例1制备中间体1的方法,以环丁甲醛为起始原料、经与硝基乙酸乙酯缩合、与邻氯苯基溴化镁进行格氏反应、硝基锌粉还原、Boc保护氨基、碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体13的四个单一手性异构体13-a,13-b,13-c,13-d。MS m/z:254[M-99]
+,298[M-55]
+。
实施例14中间体14的制备
参照实施例1制备中间体1的方法,以环己基甲醛为起始原料、经与硝基乙酸乙酯缩合、与邻氯苯基溴化镁进行格氏反应、硝基锌粉还原、Boc保护氨基、碱水解,最后经 SFC手性拆分柱分离制备可分别得到中间体14的四个单一手性异构体14a,14-b,14-c,14-d。MS m/z:282[M-99]
+,326[M-55]
+。
实施例15中间体15的制备
将二氯甲烷活化过的树脂加至FMOC-D-CHA-OH(2.00g,5.08mmol)及DIPEA(1.97g,15.24mmol,2.66mL)的DMF(40.00mL)溶液中,震荡反应12小时后,抽滤,将DCM/MeOH/DIPEA(85/10/5)加至树脂中,振荡30min,过滤,DCM(50*10ml),DMF(50*10ml),DCM(50*10ml)依次洗涤,将HFIP(20%in DCM)(913.89mg,5.08mmol,40.00mL)加至树脂中,震荡片刻,抽滤,再加入HFIP(20%in DCM)(913.89mg,5.08mmol,40.00mL)至树脂中,震荡抽滤,合并滤液,浓缩得到环己基-D-甘氨酸(870.00mg,5.08mmol,100.00%yield),取其500mg(2.92mmol),加入THF-水(1:1,20ml)及碳酸钠(618.98mg,5.84mmol)和Boc2O(954.84mg,4.38mmol),室温搅拌反应过夜,减压除去大部分有机溶剂,6N HCl调pH~4,乙酸乙酯萃取(3*10ml),合并有机相,减压浓缩,得中间体15的粗品0.78g,MS m/z:272[M+1]
+。
实施例16中间体16的制备
参照实施例7制备中间体7的方法,以环己基甲醛为起始原料、经与硝基乙酸乙酯缩合、与甲醇钠反应、再经硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体16的四个单一手性异构体16-a,16-b,16-c,16-d。MS m/z:302[M+1]
+。
实施例17中间体17的制备
参照实施例7制备中间体7的方法,中间体2-1与4-甲基吡唑在碳酸钾-DMF体系下反应、再经硝基锌粉还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体17的四个单一手性异构体17-a,17-b,17-c,17-d。MS m/z:324[M-55]
+。
实施例18中间体18的制备
步骤1中间体18-1的制备
冰浴和氮气保护下,将4-吡唑硼酸频哪醇酯(5g,25.8mmol)和碳酸铯(16.9g,51.5mmol)加入装有DMF(100mL)的三口瓶中,加入碘甲烷(5.68g,40mmol),升至室温。室温搅拌8小时后过滤,减压蒸除大部分溶剂,然后用水和乙酸乙酯萃取,再将水相用乙酸乙酯萃取两次,合并有机相,减压蒸除溶剂,即得中间体18-1粗品(4.85g,23.3mmol,产率90%),MS m/z:209[M+1]
+。
步骤2中间体18的制备
将氮气保护下,实施例1中间体1-4(710mg,1.63mmol),中间体18-1(509.69mg,2.45mmol),Pd(PPh3)4(94.31mg,81.66umol)以及Na2CO3(519.33mg,4.90mmol)悬浮于1,4-二氧六环(3mL)和H2O(0.3mL)的混合溶剂中,并超声脱气15min,反应液加热至80℃反应过夜,减压浓缩至干,残余物经硅胶柱分离纯化得到偶联产物(0.45g,1.03mmol,63.21%yield),MS m/z:435[M+1]
+。偶联产物再经碱水解,最后经SFC手性拆分柱分离 制备可分别得到中间体18的四个单一手性异构体18-a,18-b,18-c,18-d,MS m/z:422[M+1]
+。
实施例19中间体19的制备
步骤1中间体19-1的制备
冰浴和氮气保护下,将4-吡唑硼酸频哪醇酯(5g,25.8mmol)和碳酸铯(16.9g,51.5mmol)加入至DMF(100mL)中,加入2-碘丙烷(6.1g,36mmol),升至室温。室温搅拌8小时后过滤,减压蒸除大部分溶剂,然后用水和乙酸乙酯萃取,再将水相用乙酸乙酯萃取两次,合并有机相,减压蒸除溶剂,即得中间体19-1粗品(5.5g,23.3mmol,产率90%)。MS m/z:237[M+1]
+。
步骤2中间体19的制备
参照实施例18步骤2的方法,中间体1-4与19-1偶联后再经碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体19的四个单一手性异构体19-a,19-b,19-c,19-d。MS m/z:450[M+1]
+。
实施例20中间体20的制备
参照实施例18步骤1,以4-吡唑硼酸频哪醇酯与N-Boc-4-碘哌啶发生N-取代反应得到中间体20,MS m/z:378[M+1]
+。
实施例21中间体21的制备
参照实施例18步骤2的方法,中间体1-4与2-甲氧基吡啶-4-戊酰硼酸偶联后再碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体21四个单一手性异构体21-a,21-b,21-c,21-d。MS m/z:449[M+1]
+。
实施例22中间体22的制备
参照实施例1制备方法,以2-氯-4-溴苯甲醛为起始原料,经与硝基乙酸乙酯缩合,环丙基溴化镁进行格氏反应,硝基还原,氨基Boc保护,再参照实施例19偶联方法,与中间体19-1偶联,然后再碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体22四个单一手性异构体22-a,22-b,22-c,22-d。MS m/z:448[M+1]
+。
实施例23中间体23的制备
步骤1中间体23-1的制备
氮气保护和0℃下,向对硝基苯乙酸乙酯(156g,745.71mmol)的干燥的DMF(700mL)溶液中加入Cs2CO3(290.82g,894.85mmol),升至室温并搅拌1小时,随后降至0℃并缓慢滴加碘甲烷(116.43g,820.28mmol),滴毕,反应过夜,抽滤,滤液用2L乙酸乙酯稀释,饱和食盐水洗涤(3*1.5L),有机相无水硫酸钠干燥,过滤,浓缩,即可得到中间体23-1(165g,739.16mmol,99.12%yield),MS m/z:224[M+1]
+,粗品直接用于下一步。
步骤2中间体23-2的制备
将中间体23-1(2.30g,10.30mmol)溶于EtOH(20mL)中,氮气置换后,加入10%Pd/C(0.5g),常压氢气氛下搅拌反应过夜,原料消失后,经硅藻土抽滤,乙醇洗涤,滤液减压浓缩至干,硅胶柱分离纯化得到中间体23-2(1.30g,6.73mmol,65.31%yield),MS m/z:194[M+1]
+。
步骤3中间体23-3的制备
将中间体23-2(2.70g,13.97mmol)溶于醋酐(10mL)中,冷却至0℃,并搅拌15min,缓慢滴加HNO3(1.76g,27.94mmol,68%质量分数),滴毕,反应继续搅拌30min,原料消失,将反应液倾入冰水中,乙酸乙酯(2*30mL)萃取,合并有机相,经饱和碳酸钠洗涤,无水硫酸钠干燥,过滤,减压浓缩至干得到中间体23-3粗品(3.45g,12.32mmol,88%yield),MS m/z:281[M+1]
+。
步骤4中间体23-4的制备
将中间体23-3(3.45g,12.32mmol)溶于20ml乙醇中,加入SOCl
2(4.40g,36.96mmol,2.68mL),加热至50℃搅拌1小时,LC-MS显示原料消失,反应液减压浓缩至干,加入CH
2Cl
2(150mL)和H
2O(150mL),用饱和NaHCO
3调pH值~8,水相再经CH
2Cl
2(2*150mL)萃取,合并有机相,无水硫酸钠干燥,过滤,减压浓缩至干,得到中间体23-4粗品(2.89g,12.07mmol,98%yield),MS m/z:239[M+1]
+,产物未经纯化直接用于下一步反应。
步骤5中间体23的制备
将中间体23-4(2.89g,12.07mmol)溶于10ml溶于乙醇中,氮气氛下加入Pd/C(0.5g),常压氢化反应过夜,原料消失后,经硅藻土抽滤,滤液减压浓缩至干,MPLC C18反相柱纯化得到中间体23(2.13g,10.26mmol,85%yield),MS m/z:209[M+1]
+.
实施例24中间体24的制备
参照实施例23步骤1~5的方法,以对硝基苯乙酸乙酯为原料,经与碘乙烷在DMF-碳酸铯催化下发生苄位烷基化,对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体24,MS m/z:223[M+1]
+.
实施例25中间体25的制备
参照实施例23步骤1~5的方法,以对硝基苯乙酸乙酯为原料,经与2-溴丙烷在DMF-碳酸钾催化下发生苄位烷基化,对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体25,MS m/z:237[M+1]
+.
实施例26中间体26的制备
步骤1中间体26-1的制备
氮气保护和0℃下,向对硝基苯乙酸乙酯(350g,1.67mol l)的干燥的DMF(2L)溶液中加入Cs2CO3(2.73kg,8.37mol),升至室温并搅拌1小时,随后缓慢滴加碘甲烷(1.19kg,8.37mol),滴毕,室温反应过夜,抽滤,滤液用10L乙酸乙酯稀释,饱和食盐水洗涤(3*10L),有机相无水硫酸钠干燥,过滤,浓缩,即可得到中间体26-1(320g,1.24mol,74.17%yield),MS m/z:238[M+1]
+,粗品直接用于下一步。
步骤2~5中间体26的制备
参照实施例23步骤2~5的方法,以步骤1中间体26-1原料,经对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体26,MS m/z:223[M+1]
+.
实施例27中间体27的制备
参照实施例23的方法,以2-F-4-硝基苯乙酸乙酯为起始原料,经与碘甲烷在DMF-碳酸铯催化下发生苄位双甲基化,对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体27,MS m/z:241[M+1]
+.
实施例28中间体28的制备
参照实施例23的方法,以2-氟-4-硝基苯乙酸乙酯为起始原料,经与碘甲烷在DMF-碳酸铯催化下发生苄位双甲基化,对位硝基还原,间位(3-位)硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体28,MS m/z:241[M+1]
+.
实施例29中间体29的制备
参照实施例23的方法,以2-氟-4-硝基-5-溴苯乙酸乙酯为起始原料,经与碘甲烷在DMF-碳酸铯催化下发生苄位双甲基化,4-硝基还原,间位(3-位)硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原并同时脱5-位溴即可得到中间体29,MS m/z:241[M+1]
+.
实施例30中间体30的制备
步骤1中间体30-1的制备
将对异丙基苯胺(15.6g,115.38mmol)溶于醋酐(100mL)中,冷却至0℃,并搅拌15min,缓慢滴加HNO
3(21.81g,346.14mmol,68%质量分数),滴毕,反应继续搅拌12h,原料消失,将反应液倾入冰水中,乙酸乙酯(2*300mL)萃取,合并有机相,经饱和碳酸钠洗涤,无水硫酸钠干燥,过滤,减压浓缩至干得到中间体30-1粗品(16g,64.79mmol,56.16%yield),MS m/z:223[M+1]
+。
步骤2中间体30-2的制备
将中间体30-1(15g,67.49mmol)溶于乙醇(150mL)中,加入NaOH(4.05g,101.24mmol),加热至80℃反应2小时,LC-MS显示原料已反应完,反应液倾入水中,DCM(100ml*3)萃取,合并有机相,有机相再经饱和食盐水洗,无水硫酸钠干燥,过滤,减压浓缩至干得到中间体30-2粗品(10g,49.94mmol,74.00%yield),直接用于下一步,MS m/z:181[M+1]
+。
步骤3中间体30的制备
参照步骤23硝基还原方法,中间体30-2经Pd/C氢化还原,再经MPLC反相纯化即可得到中间体30,MS m/z:151[M+1]
+。
实施例31中间体31的制备
参照实施例23步骤1~5的方法,以对硝基苯乙酸乙酯为原料,经与1,2-二溴乙烷在DMF-钠氢条件下发生苄位环丙化,对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体31,MS m/z:221[M+1]
+.
实施例32中间体32的制备
参照实施例23步骤1~5的方法,以对硝基苯乙酸乙酯为原料,经与1,3-二碘丙烷在DMF-碳酸铯催化下发生苄位环丁化,对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体32,MS m/z:235[M+1]
+.
实施例33中间体33的制备
参照实施例23步骤1~5的方法,以对硝基苯乙酸乙酯为原料,经与溴代环戊烷在DMF-碳酸铯催化下发生苄位引入环戊基,对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体33,MS m/z:263[M+1]
+.
实施例34中间体34的制备
步骤1中间体34-1的制备
将2-硝基-4-溴苯胺(5.00g,23.04mmol),丙烯酸乙酯(4.61g,46.08mmol)和DIPEA(8.94g,23.04mmol,12.50mL)依次加至100ml乙腈中,氮气鼓泡下超声脱气5min,随后加入Pd(PPh3)4(2.60g,23.04mmol),氮气保护下加热至90℃反应过夜,反应完毕后,冷却至室温,加水稀释,乙酸乙酯萃取(3x200mL),合并有机相,无水硫酸钠干燥抽滤,滤液浓缩至干,粗品经硅胶柱层析分离纯化得到中间体34-1(2.20g,9.31mmol,40.42%yield),MS m/z:237[M+1]
+.
步骤2中间体34-2的制备
将中间体34-1(3.20g,13.55mmol)溶于30ml溶于乙醇中,氮气氛下加入Pd/C(0.2g),常压氢化反应过夜,原料消失后,经硅藻土抽滤,滤液减压浓缩至干,MPLC C18反相柱纯化得到中间体34-2(1.76g,8.53mmol,62.95%yield),MS m/z:207[M+1]
+.
步骤3中间体34的制备
将中间体34-2(640.00mg,3.10mmol)溶于30ml溶于甲醇中,氮气氛下加入PtO2(60.00mg),常压氢化反应过夜,原料消失后,经硅藻土抽滤,滤液减压浓缩至干,MPLC C18反相柱纯化得到中间体34(620.00mg,2.98mmol,96.13%yield),MS m/z:209[M+1]
+.
实施例35中间体35的制备
参照实施例18步骤2的方法,首先,2-硝基-4-溴苯胺与3,5-二甲基吡唑-4-硼酸频那醇酯偶联后,再参考实施例23硝基钯碳还原方法即可得到中间体35,MS m/z:203[M+1]
+.
实施例36中间体36的制备
参照实施例18步骤2的方法,首先,2-硝基-4-溴苯胺与3,5-二甲基异恶唑-4-硼酸频哪醇酯偶联后,再参考实施例23硝基钯碳还原方法即可得到中间体36,MS m/z:204[M+1]
+.
实施例37中间体37的制备
步骤1中间体37-1的制备
将3,5-二甲基异恶唑-4-硼酸频哪醇酯(1g,4.50mmol)与1-溴-2-甲氧基乙烷(688.41mg,4.95mmol)及碳酸铯(2.93g,9.01mmol)加至10mL的DMF中,室温下搅拌反应24小时,加入水100ml,乙酸乙酯(100ml*3)萃取,合并有机相,饱和食盐水洗,无水硫酸钠干燥,过滤,浓缩,粗品经硅胶柱分离纯化(石油醚/乙酸乙酯3:1~1:1)得到黄绿色中间体37-1,(1.12g,4.0mmol,89%yield),MS m/z:281[M+1]
+.
步骤2中间体37的制备
参照实施例18步骤2的方法,首先,2-硝基-4-溴苯胺与硼酸频哪醇酯37-1偶联后,再参考实施例23硝基钯碳还原方法即可得到中间体37,MS m/z:261[M+1]
+.
实施例38中间体38的制备
步骤1中间体38-1的制备
将4-溴邻苯二胺(5.00g,26.73mmol),联硼酸频那醇酯(20.37g,80.20mmol)和KOAc(10.48g,106.93mmol)依次加至100ml二氧六环中,氮气鼓泡下超声脱气5min,随后加入Pd(dppf)Cl
2(586.25mg,801.98umol),氮气保护下加热至100℃反应20小时,反应完毕后,冷却至室温,经短硅胶柱过滤,滤液减压浓缩,粗品经硅胶柱层析分离纯化(石油醚 /乙酸乙酯2:1)到中间体38-1(86mg,367.36umol,6.87%yield),MS m/z:235[M+1]
+.
步骤2中间体38-2的制备
将2-(4-溴-1,3-二甲基-1H-吡唑-5-基)乙酸甲酯(660mg,2,83mmol)溶于15ml的7M氨水-乙醇溶液中,室温搅拌20小时,减压浓缩至干即可得到中间体38-2(574mg,2,63mmol,93%yield),MS m/z:232[M+1]
+.
步骤3中间体38的制备
参照实施例18步骤2的方法,步骤1所得硼酸频哪醇酯38-1与步骤2所得38-2偶联即可得到中间体38,MS m/z:246[M+1]
+.
实施例39中间体39的制备
参照实施例18步骤2的方法,由实施例38步骤1所得硼酸频哪醇酯38-1与2-(4-溴-1,3-二甲基-1H-吡唑-5-基)乙胺偶联即可得到中间体39,MS m/z:218[M+1]
+.
实施例40中间体40的制备
步骤1中间体40-1的制备
将O-甲磺酸基乳酸乙酯(1.24g,6.30mmol)和碳酸铯(3.08g,9.46mmol)于室温下加至3,5-二甲基吡唑-4-硼酸频那醇酯(0.7g,3.15mmol)的10ml乙腈溶液中,反应于60℃搅拌10小时,减压浓缩,加水和乙酸乙酯萃取,有机相浓缩后经硅胶柱层析分离纯化(石油醚/甲基叔丁基醚1:1)得到中间体40-1(700mg,2.17mmol,68.93%yield),MS m/z:323[M+1]
+.
步骤2中间体40-2的制备
将中间体40-1(0.7g,2.17mmol)溶于THF/EtOH/H2O=4/1/1(10mL)中,加入LiOH一水合物(260.16mg,10.86mmol),于室温搅拌3小时,经1N HCl调pH~5,乙酸乙酯萃取,有机相浓缩,得到中间体40-2的粗品(500mg,1.70mmol,78.24%yield),未经纯化直接用于下一步,MS m/z:295[M+1]
+.
步骤3中间体40-3的制备
将中间体40-2(0.5g,1.70mmol)溶于10ml的DCM中,室温下依次加入HBTU(647.29mg,2.55mmol),DIEA(1.10g,8.50mmol)及甲胺(105.58mg,3.40mmol),反应液于50℃下搅拌反应10小时,减压浓缩,加水和乙酸乙酯萃取,有机相干燥后减压浓缩,硅胶柱层析分离纯化(DCM/MeOH:20/1)即可得到中间体40-3(80mg,260.42umol,15.32%yield),MS m/z:308[M+1]
+.
步骤4中间体40的制备
参照实施例18步骤2的方法,首先,2-硝基-4-溴苯胺与硼酸频哪醇酯40-3偶联后,再参考实施例23硝基钯碳还原方法即可得到中间体40,MS m/z:288[M+1]
+.
实施例41化合物41的制备
步骤1中间体41-1的制备
参照实施例23步骤1~5的方法,以对硝基苯乙酸乙酯为原料,经对位硝基还原,间位硝化并同时对位氨基乙酰化,再脱乙酰基,氢化还原即可得到中间体41-1,MS m/z:195[M+1]
+.
步骤2中间体41-2的制备
将1-乙基-1H-吡唑-5-羧酸(1.40g,9.99mmol溶于5ml DMF,0℃下依次加入HOAt(2.27g,9.99mmol),DIPEA(1.29g,9.99mmol,1.74mL)以及EDCI(2.23g,9.99mmol),5min后,加入(S)-(-)-环己基丙氨酸甲酯(2.22g,11.99mmol),转至室温搅拌2小时,反应完毕,加水和乙酸乙酯萃取,有机相经饱和氯化铵洗涤,无水硫酸钠干燥,过滤,减压浓缩,粗品经硅胶柱层析分析纯化得中间体41-2(1.70g,5.53mmol,55.36%yield),MS m/z:308[M+1]
+.
步骤3中间体41-3的制备
将中间体41-2(1.40g,4.55mmol)和LiOH(218.16mg,9.11mmol)加至10ml的THF-水(1:1)中,室温下搅拌4小时,减压浓缩后,用1N HCl调pH~4,DCM(2×20ml)萃取,合并有机相,无水硫酸钠干燥,过滤,滤液减压浓缩至干得中间体41-3(700.00mg,2.39mmol,52.44%yield),粗品未经纯化直接用于下一步反应,MS m/z:294[M+1]
+.
步骤4中间体41-4结构异构体混合物的制备
冰浴下,将中间体41-3(280.92mg,957.60umol),HATU(1.31g,3.46mmol)溶于5mL的DCM中,搅拌片刻后,依次加入二胺41-1(194mg,1.0mmol)和DIPEA(1.03g,7.98mmol),室温反应3小时,加水淬灭,减压除去大部分有机溶剂,乙酸乙酯(20ml*3)萃取,合并有机相,再分别饱和食盐水洗,无水硫酸钠干燥,减压旋干,粗品经硅胶柱层析纯化分离(石油醚/乙酸乙酯1:1)即可得到中间体41-4的结构异构体混合物(992.00mg,2.11mmol,79.22%yield),MS m/z:470(M+1)
+,二者不用分离并用于下一步。
步骤5中间体41-5的制备
将步骤4所得中间体41-4混合物(992.00mg,2.11mmol)加至AcOH(10mL)中,55℃反应12h,减压浓缩旋干,硅胶柱层析分离纯化(石油醚/乙酸乙酯1:1)得到中间体41-5(800mg,1.82mmol,86.3%yield),MS m/z:452(M+1)
+。
步骤6中间体41-6的制备
将中间体41-5(800mg,1.82mmol)溶于10mL的THF-H2O(1:1)中,加入LiOH(438mg,18.3mmol),于室温搅拌3小时,经1N HCl调pH~5,乙酸乙酯萃取,有机相浓缩,得到中间体41-6的粗品(500mg,1.18mmol,64.5%yield),未经纯化直接用于下一步,MS m/z:424[M+1]
+.
步骤7中间体41制备
冰浴下,中间体41-6的粗品(500mg,1.18mmol)与HOAt(293.42mg,2.16mmol)及EDCI(412.08mg,2.16mmol)的10mL的DCM混合液搅拌片刻后,依次加入甲胺盐酸盐(239mg,3.54mmol)和DIPEA(697.08mg,5.39mmol,939.46uL),转至室温反应3小时,加水淬灭,减压除去大部分有机溶剂,乙酸乙酯(20ml*3)萃取,合并有机相,再分别饱和氯化铵和饱和食盐水洗,无水硫酸钠干燥,减压旋干,粗品经硅胶柱层析纯化分离(石油醚/乙酸乙酯3:1)即可得到化合物41(0.44g,0.1mmol,85%yield),MS m/z:437(M+1)
+.
实施例42化合物42的制备
步骤1中间体42-1的制备
参照实施例41步骤7方法,中间体41-6与D-亮氨酸叔丁酯盐酸盐缩合即可得到中间体42-1,MS m/z:593(M+1)
+.
步骤2化合物42的制备
中间体42-1(600mg,1.01mmol)溶于4ml二氯甲烷,冰浴下加入4mL三氟乙酸,滴毕,冰浴下搅拌3小时,反应完毕,减压浓缩,粗品经MPLC反相C18柱纯化即可得化合物42(26.85mg,47.03umol,4.66%yield),MS m/z:537(M+1)
+.
实施例43化合物43的制备
参照实施例41步骤7方法,中间体41-6与异戊胺缩合即可得到化合物43,MS m/z:493(M+1)
+.
实施例44化合物44的制备
参照实施例41步骤2-7,经1-乙基-3-甲基-1H-吡唑-5-羧酸与(S)-(-)-环己基丙氨酸甲酯缩合,甲酯水解,与邻苯二胺缩合,关咪唑环,乙酯水解,最后与胺缩合即可得到化合物44,MS m/z:451[M+1]
+。
实施例45化合物45的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例30中间体邻苯二胺30缩合,关咪唑环即可得到化合物45,MS m/z:408[M+1]
+。
实施例46化合物46的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例23中间体邻苯二胺23缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物46,MS m/z:451[M+1]
+.
实施例47化合物47的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例24中间体邻苯二胺24缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物47,MS m/z:465[M+1]
+.
实施例48化合物48的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例25中间体邻苯二胺25缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物48,MS m/z:479[M+1]
+.
实施例49化合物49的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例33中间体邻苯二胺33缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物49,MS m/z:505[M+1]
+.
实施例50化合物50的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例31中间体邻苯二胺31缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物50,MS m/z:463[M+1]
+.
实施例51化合物51的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例32中间体邻苯二胺32缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物51,MS m/z:477[M+1]
+.
实施例52化合物52的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例26中间体邻苯二胺26缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物52,MS m/z:465[M+1]
+.
实施例53化合物53的制备
参照实施例41步骤4-7,依次经过中间体41-3与中间体2-溴-4,5-邻苯二胺乙酸乙酯缩合(可由2-溴-4-硝基苯基乙酸乙酯参照实施例23步骤2~5的方法得到),关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物53,MS m/z:515[M+1]
+.
实施例54化合物54的制备
室温下将实施例53化合物53(89mg,0.173mmol),2-苯基乙烯基硼酸(33.5mg,0.227mmol),K
2CO
3(71.6mg,0.519mmol)依次加至1,4-二氧六环(2ml)和水(0.3ml)的混合溶液中,氮气鼓泡下超声10min,随后加入Pd(dppf)Cl
2(25mg,35.00umol),反应于80℃ 下搅拌过夜,反应完毕,冷却至室温,加入水稀释,乙酸乙酯萃取,有机相经水洗,饱和氯化钠洗涤,无水硫酸钠干燥后过滤,滤液减压浓缩至干,粗品经硅胶柱层析分离纯化即可得到中化合物54(20.7mg,38.4umol,22.21%yield),MS m/z:539[M+1]
+.
实施例55化合物55的制备
将化合物54(10.00mg,0.019mmol)溶于5ml溶于甲醇中,氮气氛下加入10%Pd/C(3.00mg),常压氢化反应过夜,原料消失后,经硅藻土抽滤,滤液减压浓缩至干,MPLC C18反相柱纯化得到化合物55(9.85mg,0.018mmol,96%yield),MS m/z:541[M+1]
+.
实施例56化合物56的制备
步骤1中间体56-1得制备
冰浴下,将4-吡唑甲酸乙酯(1.00g,7.14mmol)溶于20ml无水THF中,分批加入NaH(430.00mg,17.92mmol),氮气保护下搅拌1小时,随后加入溴乙基甲醚(1.20g,8.64mmol),允许反应自然升至室温,2小时后,反应完毕,用1N HCl调pH~5,乙酸乙酯萃取,饱和食盐水洗,有机相无水硫酸钠干燥,过滤,减压浓缩,粗品经硅胶柱层析分离纯化即可得到中间体56-1(1.20g,6.05mmol,84.79%yield),MS m/z:199[M+1]
+.
步骤2中间体56-2得制备
参照实施例41步骤6方法,由中间体56-1经氢氧化锂水解即可得到中间体56-2,MS m/z:171[M+1]
+.
步骤3化合物56的制备
参照实施例41步骤2-7,类似地,由(S)-(-)-环己基丙氨酸甲酯与中间体56-2缩合、甲酯水解、与中间体26缩合,关咪唑环,乙酯水解,最后与甲胺缩合即可得到化合物56,MS m/z:495[M+1]
+.
实施例57化合物57的制备
步骤1中间体57-1结构异构体混合物的制备
参照实施例41步骤4方法,由中间体15与中间体26缩合可得到中间体57-1(结构异构体混合物),MS m/z:476(M+1)
+,二者不用分离并用于下一步。
步骤2中间体57-2的制备
参照实施例41步骤5方法,由中间体57-1(结构异构体混合物)在醋酸中加热成咪唑环可得到中间体57-2,MS m/z:458(M+1)
+。
步骤3中间体57-3的制备
中间体57-2(1.2g,2.62mmol)溶于5mL二氯甲烷,冰浴下加入5mL三氟乙酸,滴毕,冰浴下搅拌3小时,反应完毕,减压浓缩,粗品经MPLC反相C18柱纯化即可得化合物42(800mg,2.24mol,85%yield),MS m/z:358(M+1)
+.
步骤4中间体57-4的制备
参照实施例41步骤2方法,由中间体57-3与1-甲基-1H-吡唑-5-羧酸缩合可得到中间体57-4,MS m/z:466(M+1)
+。
步骤5中间体57-5的制备
参照实施例41步骤6方法,由中间体57-4经LiOH水解可得到中间体57-5,MS m/z:438(M+1)
+。
步骤6化合物57的制备
参照实施例41步骤7方法,由中间体57-5与(2R)-2-氨基-N,N-二甲基-丙酰胺缩合可得到化合物57,MS m/z:536(M+1)
+,白色固体,
1H NMR(400MHz,DMSO-d
6)δ7.70–7.57(m,2H),7.51(d,J=2.1Hz,1H),7.35(d,J=8.5Hz,1H),7.04(d,J=2.1Hz,1H),5.44(dd,J=8.8,6.7Hz,1H),4.66(q,J=6.8Hz,1H),4.03(s,3H),2.95(s,3H),2.78(s,3H),2.00(t,J=7.1Hz,2H),1.84–1.56(m,6H),1.52(s,6H),1.32–1.21(m,2H),1.20–0.92(m,9H).
实施例58化合物58的制备
参照实施例57方法,由57-5与(2R)-2-氨基-N-甲基-丙酰胺缩合可得到化合物58,MS m/z:522(M+1)
+,白色固体,
1H NMR(400MHz,DMSO-d
6)δ7.70–7.60(m,2H),7.52(d,J=2.1Hz,1H),7.40(d,J=8.8Hz,1H),7.05(d,J=2.1Hz,1H),5.45(dd,J=9.5,5.9Hz,1H),4.23(q,J=7.1Hz,1H),4.03(s,3H),2.07–1.96(m,2H),1.84–1.72(m,2H),1.66(s,2H),1.61(s,1H),1.53(s,6H),1.35–1.23(m,1H),1.16(dd,J=19.5,9.0Hz,6H),1.02(dt,J=17.4,7.8Hz,3H).
实施例59化合物59的制备
参照实施例57方法,由57-5与(2R)-2-氨基-丙酰胺缩合可得到化合物59,MS m/z:508(M+1)
+,白色固体,
1H NMR(400MHz,DMSO-d
6)δ7.57(d,J=8.1Hz,2H),7.48(d,J=2.0Hz,1H),7.37–7.16(m,1H),7.05(t,J=6.7Hz,1H),5.51–5.34(m,1H),4.20(d,J=7.1Hz,1H),4.02(s,3H),1.97(t,J=7.0Hz,2H),1.75(t,J=14.3Hz,2H),1.64(s,2H),1.53(t,J=16.2Hz,6H),1.37(s,1H),1.31–1.22(m,3H),1.22–0.89(m,7H).
实施例60化合物60的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例34的中间体34缩合,关咪唑环,乙酯水解,与D-亮氨酸叔丁酯盐酸盐缩合,最后叔丁酯经三氟乙酸水解即可得到化合物60,MS m/z:551[M+1]
+.
实施例61化合物61的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例26中间体邻苯二胺26缩合,关咪唑环,乙酯水解,与D-亮氨酸叔丁酯盐酸盐缩合,最后叔丁酯经三氟乙酸水解即可 得到化合物61,MS m/z:565[M+1]
+.
1H NMR(400MHz,DMSO-d
6)δ9.11(d,J=7.2Hz,1H),7.57~7.63(m,2H),7.54(d,J=2.4Hz,1H),7.41(d,J=8Hz,1H),7.33(d,J=8Hz,),7.03(dd,J1=1.6Hz,J2=2Hz,1H),5.45(dd,J
1=7.6Hz,J
2=14.8Hz,1H),4.39~4.54(m,2H),4.23~4.28(m,1H),1.99(t,J=7.2Hz,2H),1.72~1.84(m,2H),1.65~1.71(m,2H),1.57~1.62(m,2H),1.52(d,J=2.8Hz,6H),1.34~1.46(m,3H),1.28(t,J=6.8Hz,3H),1.11~1.21(m,3H),0.92~1.06(m,2H),0.78(t,J=5.6Hz,6H).
实施例62化合物62的制备
首先,参照实施例38方法,由中间体38-1与2-(4-溴-1,3-二甲基-1H-吡唑-5-基)乙酸甲酯偶联,再参照实施例57步骤1-6,偶联产物与实施例15中间体15缩合,关咪唑环,脱Boc,引入2-甲基-1H-吡唑酰基,酯水解,最后与环戊胺缩合即可得到化合物62,MS m/z:557[M+1]
+.
实施例63化合物63的制备
首先,参照实施例38方法,由中间体38-1与2-(4-溴-1,3-二甲基-1H-吡唑-5-基)乙酸甲酯偶联,再参照实施例57步骤1-6,偶联产物与实施例15中间体15缩合,关咪唑环,脱Boc,引入2-甲基-1H-吡唑酰基,酯水解,最后与2-丙胺缩合即可得到化合物63, MS m/z:531[M+1]
+.
实施例64化合物64的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例35中间体邻苯二胺35缩合,关咪唑环可得到化合物64,MS m/z:460[M+1]
+.
实施例65化合物65的制备
步骤1中间体65-1的制备
将2-甲基-6,7-二氢吡唑[1,5-a]并吡嗪-5(4H)-苄氧羰基(110mg,0.405mmol)溶于1mL的DMF,加入NBS(72mg,0.405mmol),室温搅拌3小时,硅胶柱层析分离纯化即可得溴代中间体65-1,MS m/z:350[M+1]
+.
步骤2中间体65-2的制备
参照实施例38方法,由硼酸频哪醇酯38-1与实施例65步骤2所得溴代物65-1偶联即可得到中间体65-2,MS m/z:378[M+1]
+.
步骤3化合物65的制备
参照实施例57步骤1-6,依次经实施例15中间体15与实施例65中间体65-2缩合,关咪唑环,脱Boc,引入2-甲基-1H-吡唑酰基,再氢化脱Cbz保护基即可得到化合物65,MS m/z:487[M+1]
+.
实施例66化合物66的制备
将实施例化合物65(20mg,41.10umol)溶于1ml的DCM中,环戊基异氰酸酯(4.57mg,41.10umol)的1ml二氯甲烷溶液滴加其中,滴毕,室温反应4小时,浓缩,粗品经MPLC反相C18柱分离纯化即可得化合物66,MS m/z:598[M+1]
+.
实施例67化合物67的制备
参照实施例57步骤1-6方法,实施例14中间体14的四个异构体混合物(拆分前)与实施例41中间体41-1缩合,关咪唑环,脱Boc,引入1-乙基-1H-吡唑-5-酰基,酯水解,最后与甲胺盐酸盐缩合即可得到化合物67,MS m/z:548[M+1]
+.
实施例68化合物68的制备
参照实施例57步骤1-6方法,实施例14中间体14的单一立体异构体14a与实施例41中间体41-1缩合,关咪唑环,脱Boc,引入1-乙基-1H-吡唑-5-酰基,酯水解,最后与甲胺盐酸盐缩合即可得到化合物68,MS m/z:548[M+1]
+.
实施例69化合物69的制备
参照实施例57步骤1-6方法,实施例14中间体14的单一立体异构体14d与实施例41中间体41-1缩合,关咪唑环,脱Boc,引入1-乙基-1H-吡唑-5-酰基,酯水解,最后与甲胺盐酸盐缩合即可得到化合物69,MS m/z:548[M+1]
+.
实施例70化合物70的制备
参照实施例57步骤1-6方法,实施例14中间体14的单一立体异构体14b与实施例41中间体41-1缩合,关咪唑环,脱Boc,引入1-乙基-1H-吡唑-5-酰基,酯水解,最后与 甲胺盐酸盐缩合即可得到化合物70,MS m/z:548[M+1]
+.
实施例71化合物71的制备
参照实施例57步骤1-6方法,实施例14中间体14的单一立体异构体14c与实施例41中间体41-1缩合,关咪唑环,脱Boc,引入1-乙基-1H-吡唑-5-酰基,酯水解,最后与甲胺盐酸盐缩合即可得到化合物71,MS m/z:548[M+1]
+.
实施例72化合物72的制备
参照实施例57步骤1-6方法,实施例16的中间体16的四个异构体混合物(拆分前)与实施例26中间体26缩合,关咪唑环,脱Boc,引入1-乙基-1H-吡唑-5-酰基,乙酯水解,最后与(2R)-2-氨基-N-甲基-丙酰胺缩合即可得到化合物72,MS m/z:552[M+1]
+。
实施例73化合物73的制备
步骤1中间体73-1结构异构体混合物的制备
参照实施例41步骤4方法,由起始原料邻氯-D-Boc-苯丙氨酸与中间体34缩合可得到中间体73-1(结构异构体混合物),MS m/z:490(M+1)
+,二者不用分离并用于下一步。
步骤2中间体73-2的制备
参照实施例41步骤5方法,由中间体73-2(结构异构体混合物)在醋酸中加热成咪唑环可得到中间体73-2,MS m/z:472(M+1)
+。
步骤3-6化合物73的制备
参照实施例57步骤3-6,依次经过中间体73-2脱Boc,与1-甲基-1H-吡唑-5-羧酸缩合,乙酯水解,与D-亮氨酸叔丁酯盐酸盐缩合,最后叔丁酯经三氟乙酸水解即可得到化合物73,MS m/z:579[M+1]
+.
实施例74化合物74的制备
步骤1中间体74-1的制备
将2-甲基-2-(4-硝基苯基)丙酸乙酯(40.0g,168.6mmol)溶于乙醇(600mL),室温 下分批加入氢氧化钠(10.1g,252.9mmol),加毕升温至80℃搅拌反应2小时。减压蒸除溶剂,冰浴下滴加1N HCl溶液调节pH~4,乙酸乙酯萃取3次,合并有机相,无水硫酸钠干燥,减压蒸除溶剂后得中间体74-1(33.5g,160.2mmol,产率95%)。MS m/z:210[M+1]
+.
步骤2中间体74-2的制备
将中间体74-1(33.5g,160.2mmol)溶于DMF(500mL),冰浴下加入HBTU(72.9g,192.2mmol),N,N-二异丙基乙胺(45.5g,352.4mmol)和氯化铵(12.9g,240.3mmol),加毕升温至室温搅拌反应2小时。然后用水和乙酸乙酯萃取,再将水相用乙酸乙酯萃取两次,合并有机相,无水硫酸钠干燥,减压蒸除溶剂后得中间体74-2粗品(29.0g,139.3mmol,产率87%)。MS m/z:209[M+1]
+.
步骤3中间体74-3的制备
将中间体74-2(29.0g,139.3mmol)溶于甲醇(300mL),加入钯碳(2.9g,钯含量10%),反应体系用氢气置换三次,并在氢气氛围下(1atm)搅拌12小时。反应物用硅藻土过滤,减压蒸除溶剂,即得中间体74-3(22.4g,125.4mmol,产率90%)。MS m/z:179[M+1]
+.
步骤4中间体74-4的制备
将中间体74-3(22.4g,125.4mmol)溶于四氢呋喃(300mL),冰浴下滴加1M硼烷四氢呋喃溶液(627.0mL,627.0mmol),滴加完毕氮气保护下60℃搅拌反应12小时。加 入饱和氯化铵溶液淬灭反应,乙酸乙酯萃取,合并有机相,无水硫酸钠干燥,减压蒸除溶剂,经MPLC纯化(洗脱剂梯度:乙腈:水=0:100至40:60,20分钟,水中含0.005%甲酸)后得中间体74-4(12.4g,75.2mmol,产率60%)。MS m/z:165[M+1]
+.
步骤5中间体74-5的制备
将中间体74-4(12.4g,75.2mmol)溶于四氢呋喃(500mL),0℃加入三乙胺(30.4g,300.8mmol)和苯甲氧羰酰琥珀酰亚胺(39.4g,157.9mmol)。然后升温至室温搅拌反应1小时。经硅胶柱层析纯化(洗脱剂:石油醚:乙酸乙酯=5:1)后即得中间体74-5(30.6g,70.7mmol,产率94%)。MS m/z:433[M+1]
+.
步骤6中间体74-6的制备
将中间体74-5(30.6g,70.7mmol)溶于醋酸酐(300mL),0℃滴加硝酸(6.3mL,141.4mmol),滴加完毕继续在0℃下搅拌反应5小时。经硅胶柱层析纯化(洗脱剂:石油醚:乙酸乙酯=3:1),后即得中间体74-6(22.3g,46.7mmol,产率66%)。MS m/z:478[M+1]
+.
步骤7中间体74-7的制备
将中间体74-6(22.3g,46.7mmol)溶于乙醇(400mL),室温下分批加入氢氧化钠(1.9g,46.7mmol),加毕升温至70℃搅拌反应1小时。然后用水和乙酸乙酯萃取,再将水相用乙酸乙酯萃取两次,合并有机相,无水硫酸钠干燥,减压蒸除溶剂后得中间体74-7粗品(15.4g,44.8mmol,产率96%)。MS m/z:344[M+1]
+.
步骤8中间体74-8的制备
将中间体74-7粗品(15.4g,44.8mmol)溶于甲醇(600mL),冰浴下加入锌粉(14.6g,224.0mmol)和氯化铵(239.6g,448.0mmol),加毕升至室温搅拌反应1小时。过滤除去锌粉,减压蒸除溶剂,经MPLC纯化(洗脱剂梯度:乙腈:水=0:100至50:50,20分钟,水中含0.005%甲酸)后得中间体74-8(12.5g,40mmol,产率89%),MS m/z:314[M+1]
+.
步骤9中间体74-9的制备
参照实施例57步骤1-4,以实施例2中间体2的单一立体异构体2-c为原料,与实施例74中间体邻苯二胺74-8缩合,关咪唑环,脱Boc,与1-甲基-1H-吡唑-5-羧酸缩合即可得到中间体74-9,MS m/z:627[M+1]
+.
步骤10中间体74-10的制备
将中间体74-9(1.25g,2.0mmol)溶于甲醇(20mL),室温下加入钯碳(140.0mg,钯含量10%)。反应体系用氢气置换三次,并在氢气氛围下(1atm)搅拌12小时。反应物用硅藻土过滤,减压蒸除溶剂,即得中间体74-10粗品(0.935g,1.9mmol,产率96%)。MS m/z:493[M+1]
+.
步骤11化合物74的制备
参照实施例41步骤7方法,由中间体74-10与乙酰氯缩合可得到化合物74,MS m/z:535(M+1)
+.
实施例75化合物75的制备
参照实施例74方法,以实施例2中间体2的单一立体异构体2-d经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物75,MS m/z:535(M+1)
+.
实施例76化合物76的制备
参照实施例74方法,以实施例2中间体2的单一立体异构体2-b经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物76,MS m/z:535(M+1)
+.
实施例77化合物77的制备
参照实施例74方法,以实施例2中间体2的单一立体异构体2-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物77,MS m/z:535(M+1)
+.
实施例78化合物78的制备
参照实施例74方法,以实施例13中间体13的单一立体异构体13-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物78,MS m/z:547(M+1)
+.
1HNMR(400MHz,DMSO-d
6)δ12.67(s,1H),8.79(s,1H),8.42(s,1H),7.58(t,J=4.0Hz,1H),7.52(s,2H),7.42(t,J=9.6,2H),7.36-7.32(m,2H),7.24–7.19(m,2H),6.67(d,J=2.0Hz,1H),5.59(t,J=10.0Hz,1H),4.13(t,J=10.0Hz,1H),3.83(s,3H),2.69-2.63(m,1H),2.36-2.33(m,1H),1.78(s,3H),1.56-1.49(m,2H),1.46-1.39(m,2H),1.30(s,6H),1.25-1.15(m,2H).
实施例79化合物79的制备
参照实施例74方法,以实施例13中间体13的单一立体异构体13-d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物79,MS m/z:547(M+1)
+.
实施例80化合物80的制备
参照实施例74方法,以实施例13中间体13的单一立体异构体13-b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物80,MS m/z:547(M+1)
+.
实施例81化合物81的制备
参照实施例74方法,以实施例13中间体13的单一立体异构体13-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物81,MS m/z:547(M+1)
+.
1HNMR(400MHz,DMSO-d
6)δ12.10(s,1H),8.93(s,1H),7.54(t,J=5.6Hz,1H),7.48(d,J=2.0Hz,1H), 7.45-7.41(m,1H),7.35(d,J=7.6Hz,1H),7.30(d,J=7.6Hz,2H),7.23(t,J=7.2Hz,1H),7.16-7.09(m,2H),7.07(d,J=2.0Hz,1H),5.73(t,J=8.8Hz,1H),4.27(t,J=8.8Hz,1H),4.01(s,3H),3.27-3.25(m,2H),2.97-2.87(m,1H),1.89-1.79(m,2H),1.76(s,3H),1.69(dd,J
1=8.4,J
2=18.0,1H),1.59-1.46(m,3H),1.25(s,6H).
实施例82化合物82的制备
步骤1中间体82-1的制备
参照实施例1制备中间体1的方法,以苯甲醛为起始原料经缩合、格氏反应、硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到分别得到中间体82-1四个单一手性异构体82-1a,82-1b,82-1c,82-1d。MS m/z:320[M+1]
+。
步骤2化合物82的制备
参照实施例74方法,以实施例82中间体82-1的单一手性异构体81-1b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物82,MS m/z:539(M+1)
+.
实施例83化合物83的制备
参照实施例74方法,以实施例82中间体82-1的单一手性异构体81-1c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物83,MS m/z:539(M+1)
+.
实施例84化合物84的制备
参照实施例74方法,以实施例82中间体82-1的单一手性异构体81-1a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物84,MS m/z:539(M+1)
+.
实施例85化合物85的制备
参照实施例74方法,以实施例82中间体82-1的单一手性异构体81-1d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物85,MS m/z:539(M+1)
+.
实施例86化合物86的制备
参照实施例74方法,以实施例13中间体13的单一手性异构体13-b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物86,MS m/z:573(M+1)
+.
实施例87化合物87的制备
参照实施例74方法,以实施例13中间体13的单一手性异构体13-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物87,MS m/z:573(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ12.44(s,1H),8.67(d,J=10.0Hz,1H),7.79(t,J=6.0Hz,1H),7.60-7.48(m,2H),7.44-7.29(m,5H),7.26-7.19(m,2H),6.65(d,J=2.0Hz,1H),5.58(t,J=6.0Hz,1H),4.12(t,J=5.6Hz,1H),3.83(s,3H),2.71-2.63(m,1H),1.65-1.40(m,6H),1.31(s,6H),0.63-0.56(m,5H).
实施例88化合物88的制备
参照实施例74方法,以实施例13中间体13的单一手性异构体13-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物88,MS m/z:573(M+1)
+.
实施例89化合物89的制备
参照实施例74方法,以实施例13中间体13的单一手性异构体13-d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物89,MS m/z:573(M+1)
+.
实施例90化合物90的制备
参照实施例74方法,以实施例19中间体19的单一手性异构体19-b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物90,MS m/z:643(M+1)
+.
实施例91化合物91的制备
参照实施例74方法,以实施例19中间体19的单一手性异构体19-d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物91,MS m/z:643(M+1)
+.
实施例92化合物92的制备
参照实施例74方法,以实施例19中间体19的单一手性异构体19-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物92,MS m/z:643(M+1)
+.
实施例93化合物93的制备
参照实施例74方法,以实施例19中间体19的单一手性异构体19-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物93,MS m/z:643(M+1)
+.
1HNMR(400MHz,CD
3OD)δ=8.42(br,1H),8.05(s,1H),7.86(s,1H),7.61~7.69(m,2H),7.56(d,J=8.4Hz,1H),7.33~7.44(m,4H),6.48(s,1H),6.10(d,J=10.0Hz,1H),4.17~4.24(m,1H),3.87(s,3H),3.47(s,2H),1.92~2.03(m,1H),1.88(s,3H),1.55(d,J=6.4Hz,6H),1.30~1.41(m,6H),1.01(d,J=6.4Hz,3H),0.90(d,J=6.8Hz,3H).
实施例94化合物94的制备
步骤1中间体94-1的制备
参照实施例1制备中间体1的方法,以苯甲醛为起始原料、经缩合、格氏反应、硝基还原、Boc保护氨基、水解,最后经SFC手性拆分柱分离制备可分别得到中间体94-1的四个单一手性异构体94-1a,94-1b,94-1c,94-1d。MS m/z:308[M+1]
+。
步骤2化合物94的制备
参照实施例74方法,以步骤1中间体94-1的单一手性异构体94-1b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物94,MS m/z:527(M+1)
+.
1H NMR(400MHz,MeOD):δ7.54(s,1H),7.48(d,J=8.7Hz,1H),7.37(d,J=7.6Hz,1H),7.27–7.12(m,7H),6.33(d,J=2.1Hz,1H),5.86(d,J=9.8Hz,1H),4.97(d,J=3.2Hz,1H),4.16(t,J=6.1Hz, 1H),3.52–3.44(m,1H),3.42–3.33(m,3H),3.13(dd,J=11.5,6.9Hz,1H),1.78(ddd,J=19.2,12.0,6.5Hz,2H),1.60(dt,J=13.1,6.5Hz,1H),1.36–1.27(m,6H),0.83(d,J=6.7Hz,3H),0.77(d,J=6.7Hz,3H),0.64(dt,J=5.8,3.8Hz,2H),0.56(ddd,J=10.2,6.6,3.8Hz,2H).
实施例95化合物95的制备
参照实施例74方法,以实施例94步骤1中间体94-1的单一手性异构体94-1c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物95,MS m/z:527(M+1)
+.
实施例96化合物96的制备
参照实施例74方法,以步骤1中间体94-1的单一手性异构体94-1a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物96,MS m/z:527(M+1)
+.
实施例97化合物97的制备
参照实施例74方法,以步骤1中间体94-1的单一手性异构体94-1d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物97,MS m/z:527(M+1)
+.
实施例98化合物98的制备
参照实施例74方法,以实施例1中间体1的单一手性异构体1-b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢溴酸-乙酸脱Cbz,最后与乙酰氯缩合可得到化合物98,MS m/z:579(M+1)
+.
实施例99化合物99的制备
参照实施例74方法,以实施例1中间体1的单一手性异构体1-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢溴酸-乙酸脱Cbz,最后与乙酰氯缩合可得到化合物99,MS m/z:579(M+1)
+.
实施例100化合物100的制备
参照实施例74方法,以实施例1中间体1的单一手性异构体1-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,氢溴酸-乙酸脱Cbz,最后与乙酰氯缩合可得到化合物100,MS m/z:627(M+1)
+.
实施例101化合物101的制备
参照实施例74方法,以实施例1中间体1的单一手性异构体1-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢溴酸-乙酸脱Cbz,最后与乙酰氯缩合可得到101,MS m/z:579(M+1)
+.
实施例102化合物102的制备
参照实施例74方法,以实施例1中间体1的单一手性异构体1-d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢溴酸-乙酸脱Cbz,最后与乙酰氯缩合可得到化合物102,MS m/z:579(M+1)
+.
实施例103化合物103的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与乙酰氯缩合可得到化合物103,MS m/z:615(M+1)
+.
实施例104化合物104的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与3-四氢呋喃甲酸缩合可得到化合物104,MS m/z:671(M+1)
+.
实施例105化合物105的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物105,MS m/z:641(M+1)
+.
实施例106化合物106的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物106,MS m/z:641(M+1)
+.
实施例107化合物107的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合可得到化合物107,MS m/z:641(M+1)
+.
实施例108化合物108的制备
参照实施例74方法,以实施例18中间体18单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,与1-Boc-吡咯烷-3-甲酸缩合,最后三氟乙酸脱Boc即可得到化合物108,MS m/z:670(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ0.79(d,J=8.4Hz,3H)0.84(d,J=6.8Hz,3H)1.32(d,J=4.0Hz,6H)1.66~1.76(m,1H)1.83~1.91(m,1H)1.97~2.06(m,1H)3.02~3.07(m,3H)3.17~3.21(m,2H)3.78(s,3H)3.87(s,3H)4.11~4.22(m,2H)6.07(t,J=9.2Hz,1H)6.60(s,1H) 7.32~7.38(m,5H)7.55~7.61(m,3H)7.83(s,1H)7.96(t,J=6.4Hz,1H)8.09(s,1H)8.73~8.96(brs,3H)。
实施例109化合物109的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丁甲酸缩合即可得到化合物109,MS m/z:655(M+1)
+.
实施例110化合物110的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环戊甲酸缩合即可得到化合物110,MS m/z:669(M+1)
+.
实施例111化合物111的制备
步骤1中间体111-1的制备
参照实施例1制备中间体1的方法,以乙醛为起始原料、经与硝基乙酸乙酯缩合、与 邻氯苯基溴化镁进行格氏反应、硝基锌粉还原、Boc保护氨基、碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体111-1的四个单一手性异构体111-1a,111-1b,111-1c,111-1d。MS m/z:314[M+1]
+。
步骤2化合物111的制备
参照实施例74方法,以中间体111-1的单一手性异构体111-1c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物111,MS m/z:533(M+1)
+.
实施例112化合物112的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与正丁酸缩合即可得到化合物112,MS m/z:643(M+1)
+.
实施例113化合物113的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,与1-Boc-吡咯烷-3-乙酸缩合,最后三氟乙酸脱Boc即可得到化合物113,MS m/z:684(M+1)
+.
实施例114化合物114的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙乙酸缩合可得到化合物114,MS m/z:655(M+1)
+.
实施例115化合物115的制备
参照实施例74方法,以实施例19中间体19的单一手性异构体19-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙乙酸缩合可得到化合物115,MS m/z:669(M+1)
+.
1H NMR(400MHz,DMSO):δ=12.64(s,1H),8.36(s,1H),8.16(s,1H),7.83(s,1H),7.78(s,1H),7.58(s,1H),7.46(s,1H),7.40(d,J=2.4Hz,2H),7.29(d,J=2.0Hz,2H),6.55(d,J=2.0Hz,1H),6.02(t,J=10.0Hz,1H),4.55-4.51(m,1H),4.28(d,J=11.2Hz,1H),3.77(s,3H),2.68(t,J=1.6Hz,1H),2.34(t,J=2.0Hz,1H),1.85(s,1H),1.63(m,1H),1.47(d,J=6.4Hz,6H),1.31(s,6H),1.25(s,1H),0.81(t,J=8.0Hz 6H),0.63-0.57(m,4H).
实施例116化合物116的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与4-甲基戊酸缩合可得到化合物116,MS m/z:671(M+1)
+.
实施例117化合物117的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与异丁酸缩合可得到化合物117,MS m/z:643(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ12.57(s,1H),8.88(s,1H),8.08(s,1H),7.81(d,J=0.8Hz,1H),7.56(s,2H),7.52-7.43(m,2H),7.43-7.34(m,2H),7.29(d,J=0.8Hz,1H),7.26-7.23(m,1H),6.53(d,J=2.0Hz,1H),6.00(t,J=10.0Hz,1H),4.26(dd,J
1=4.0Hz,J
2=10.8Hz,1H),3.89(s,3H),3.76(s,3H),2.45-2.31(m,1H),2.05-1.95(m,1H),1.90-1.75(m,1H),1.30(s,6H),0.93(d,J=6.8Hz,6H),0.81(d,J=6.8Hz,6H).
实施例118化合物118的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与3-甲基丁酸缩合可得到化合物118,MS m/z:657(M+1)
+.
实施例119化合物119的制备
参照实施例74方法,以实施例1中间体1的单一手性异构体1-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢溴酸-乙酸脱Cbz,与环丙甲酰氯缩合,再参照实施例18步骤2方法,与实施例20中间体20偶联,最后三氟乙酸脱Boc保护基即可得到化合物119,MS m/z:710(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.92(d,J=6.4Hz,1H),8.18(s,1H),7.89(s,1H),7.79(t,J=5.2Hz,1H),7.59(s,1H),7.46~7.56(m,2H),7.41(m,2H),7.25~7.29(m,2H),6.54(s,1H),6.02(t,J=10.4Hz,1H),4.43(s,1H),4.26(dd,J1=3.2Hz,J2=11.6Hz,1H),3.76(s,3H),2.94(s,2H),2.11~2.23(m,3H),1.94~2.11(m,3H),1.76~1.91(m,2H),1.58~1.67(m,2H),1.31(s,6H),1.24(s,1H),0.81(d,J=5.2Hz,6H),0.57~0.63(m,4H).
实施例120化合物120的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱 Cbz,最后与N-甲基氨基磺酰氯缩合可得到化合物120,MS m/z:666(M+1)
+.
实施例121化合物121的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与烟酸缩合可得到化合物121,MS m/z:678(M+1)
+.
实施例122化合物122的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与1-甲基吡唑-4-甲酸缩合可得到化合物122,MS m/z:681(M+1)
+.
实施例123化合物123的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实 施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙基氯甲酸酯缩合可得到化合物123,MS m/z:657(M+1)
+.
实施例124化合物124的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与恶唑-2-羧酸缩合可得到化合物124,MS m/z:668(M+1)
+.
实施例125化合物125的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙磺酰氯缩合可得到化合物125,MS m/z:677(M+1)
+.
实施例126化合物126的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与N,N-二甲基甲酰氯缩合可得到化合物126,MS m/z:644(M+1)
+.
实施例127化合物127的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与甲磺酰氯缩合可得到化合物127,MS m/z:651(M+1)
+.
实施例128化合物128的制备
步骤1中间体128-1的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,得氨基中间体,取该中间体(100mg,174.48umol),溶于3ml甲醇中,加入乙醛酸乙酯(17.81mg,174.48umol),60℃下搅拌1小时,随后加入NaBH3CN(32.89mg,523.44umol)并继续60℃搅拌过夜,加水淬灭,乙酸乙酯萃取,减压浓缩,粗品经硅胶柱层析分离纯化即可得到中间体128-1,MS m/z:659(M+1)
+.
步骤2化合物128的制备
将中间体128-1(13mg,19.72umol)溶于1ml乙酸,加入氰酸钾(1.92mg,23.66umol),室温搅拌反应过夜,浓缩后由MPLC C18柱分离纯化即可得到化合物128(5.01mg,7.40umol,37.52%yield,96.9%purity),MS m/z:656(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ=12.71(s,1H),8.90(s,1H),8.30(s,1H),8.06(s,1H),7.81(s,1H),7.55(s,2H),7.41-7.33(m,2H),7.31-7.18(m,2H),6.53(d,J=1.6Hz,1H),6.01(t,J=10.4Hz,1H),4.28-4.21(m,1H),3.88(s,3H),3.78(s,3H),3.46(s,2H),1.85(s,1H),1.36(s,4H),1.29(s,2H),0.83-0.77(m,6H).
实施例129化合物129的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,随后参考实施例128步骤1方法,与4-甲基-2-氧代戊酸进行还原胺化即可得到化合物129,MS m/z:687(M+1)
+.
1H NMR(400MHz,MeOD):δ7.98(s,1H),7.86(s,1H),7.71(s,1H),7.63-7.65(m,1H),7.28-7.46(m,5H),6.472-6.476(d,1H,J=1.6Hz),6.094-6.12(d,1H,J=10Hz),4.21-4.27(m,1H),3.96(s,3H),3.86(s,3H),3.71-3.79(m,1H),3.40-3.50(m,1H),1.64-1.80(m,3H),1.57-1.59(d,5H,J=5.2Hz),1.38-1.41(m,3H),0.99-1.01(d,2H,J=7.2Hz),0.89-0.93(m,9H).
实施例130化合物130的制备
步骤1中间体130-1的制备
将化合物129(20mg,29.10umol)溶于10ml甲醇中,加入催化量浓硫酸(285.42ug,2.91umol,1.55e
-1uL),于60℃搅拌12小时,LC-MS跟踪原料消失,反应液浓缩后加入水,乙酸乙酯萃取,浓缩后由硅胶柱层析分离纯化(MeOH:DCM=1:10),粗品再经高效液相制备(water:ACN=1:1)即可得到中间体130-1(20mg,25.67umol,88.20%yield),MS m/z:701(M+1)
+.
步骤2化合物130的制备
参照实施例128步骤2方法,由130-1在乙酸中与氰酸钾反应关环即可得到化合物130,MS m/z:712(M+1)
+.
1H NMR(400MHz,MeOD):δ8.52(s,1H),7.98(s,1H),7.86(s,1H),7.63(s,2H),7.34-7.48(m,3H),6.49(s,1H),6.09-6.12(m,1H),4.63(s,2H),4.19-4.21(m,1H),3.88-3.96(m,6H),3.34-3.40(m,2H),1.96-2.00(m,1H),1.60-1.66(m,1H),1.46-1.50(d,4H,J=16Hz),1.24-1.27(m,2H),1.02-1.03(d,3H,J=6Hz),0.89-0.91(d,3H,J=6.8Hz),0.73-0.75(t,3H,J=6Hz),0.35-0.37(d,3H,J=6.4Hz).
实施例131化合物131的制备
步骤1中间体131-1的制备
参照实施例1制备中间体1的方法,以乙醛为起始原料、经与硝基乙酸乙酯缩合、与邻氯苯基溴化镁进行格氏反应、硝基锌粉还原、Boc保护氨基、碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体131-1的四个单一手性异构体131-1a,131-1b,131-1c,131-1d。MS m/z:214[M-99]
+,258[M-55]
+。
步骤2化合物131的制备
参照实施例74方法,以实施例131中间体131-1的单一手性异构体131-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物131,MS m/z:533(M+1)
+.
1H NMR(400MHz,DMSO-d
6):δ=7.83(s,1H),7.64(s,1H),7.46-7.36(m,6H),7.21(t,J=8.0Hz,2H),6.54(s,1H),6.47(s,1H),5.90(s,1H),5.63(s,1H),4.89(d,J=4.4Hz,1H),4.84(d,J=4.0Hz,1H),4.43(s,2H),3.83(s,2H),3.33(d,J=6.0Hz,2H),1.74(s,1H),1.60(s,1H),1.43(s,1H),1.27(s,6H),1.25(s,1H),0.63-0.58(m,4H).
实施例132化合物132的制备
参照实施例74方法,以实施例11中间体11的单一手性异构体11-c为原料经过与实 施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物132,MS m/z:547(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.87(d,J=8.8Hz,1H),7.78(t,J=5.6Hz,1H),7.49~7.62(m,2H),7.47(d,J=7.6Hz,1H),7.42(d,J=8Hz,1H),7.31~7.37(m,2H),7.20~7.26(m,2H),6.3(d,J=1.6Hz,1H),5.70(s,1H),4.00~4.15(m,1H),3.83(s,3H),3.35(s,2H),1.46~1.66(m,3H),1.31(s,6H),1.24(s,1H),0.56~0.65(m,7H).
实施例133化合物133的制备
参照实施例74方法,以实施例11中间体11的单一手性异构体11-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物133,MS m/z:547(M+1)
+.
1H NMR(400MHz,DMSO-d6)δ9.42(d,J=7.6Hz,1H),7.82(t,J=6Hz,1H),7.57(s,1H),7.55(s,1H),7.53(d,J=2Hz,1H),7.51(s,1H),7.43(d,J=8.8Hz,1H),7.33(t,J=7.2Hz,1H),7.27~7.29(m,1H),7.15~7.21(m,1H),7.12(d,J=2Hz,1H),5.71(s,1H),4.09~4.18(m,1H),3.97(s,3H),3.32(d,J=6Hz,2H),1.96~2.11(m,2H),1.82~1.94(m,1H),1.52~1.58(m,1H),1.27(d,J=2Hz,6H),0.71(t,J=7.6Hz,3H),0.53~0.57(m,4H).
实施例134化合物134的制备
参照实施例74方法,以实施例11中间体11的单一手性异构体11-d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱 Cbz,最后与环丙甲酰氯缩合即可得到化合物134,MS m/z:547(M+1)
+.
实施例135化合物135的制备
参照实施例74方法,以实施例11中间体11的单一手性异构体11-b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物135,MS m/z:547(M+1)
+.
实施例136化合物136的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物136,MS m/z:560(M+1)
+.
1HNMR(400MHz,DMSO-d
6)δ12.21(s,1H),8.99(d,J=8.4Hz,1H),7.76(t,J=5.6Hz,1H),7.56(d,J=6.4Hz,1H),7.49(d,J=1.6Hz,1H),7.44-7.22(m,4H),7.17-7.12(m,2H),7.10(s,1H),5.95(t,J=8.8Hz,1H),4.00(s,3H),3.53-3.50(m,2H),3.32-3.30(m,2H),1.64-1.59(m,1H),1.42-1.31(m,1H),1.26(s,6H),0.62-0.56(m,4H),0.44-0.39(m,2H),0.31-0.23(m,1H),-0.02--0.05(m,1H).
实施例137化合物137的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-a为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物137,MS m/z:560(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ9.42(d,J=7.6Hz,1H),7.82(t,J=6Hz,1H),7.57(s,1H),7.55(s,1H),7.53(d,J=2Hz,1H),7.51(s,1H),7.43(d,J=8.8Hz,1H),7.33(t,J=7.2Hz,1H),7.27~7.29(m,1H),7.15~7.21(m,1H),7.12(d,J=2Hz,1H),5.71(s,1H),4.09~4.18(m,1H),3.97(s,3H),3.32(d,J=6Hz,2H),1.96~2.11(m,2H),1.82~1.94(m,1H),1.52~1.58(m,1H),1.27(d,J=2Hz,6H),0.71(t,J=7.6Hz,3H),0.53~0.57(m,4H).
实施例138化合物138的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-d为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物138,MS m/z:560(M+1)
+.
实施例139化合物139的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-b为原料经过与实 施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与环丙甲酰氯缩合即可得到化合物139,MS m/z:560(M+1)
+.
1HNMR(400MHz,DMSO-d
6)δ12.62(s,1H),8.87(s,1H),7.80(t,J=6.0Hz,1H),7.59-7.47(m,4H),7.40(dd,J
1=1.2Hz,J
2=8.0Hz,1H),7.35-7.19(m,4H),6.65(d,J=2.0Hz,1H),5.86(s,1H),3.84(s,3H),1.66-1.59(m,1H),1.31(s,6H),0.65-0.56(m,4H),0.24-0.17(m,1H),0.14-0.07(m,1H),0.02--0.02(m,1H).
实施例140化合物140的制备
参照实施例74方法,以实施例1中间体1的单一手性异构体1-c为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢溴酸脱Cbz,最后与环丙甲酰氯缩合即可得到化合物140,MS m/z:639(M+1)
+.
实施例141化合物141的制备
参照实施例18步骤2方法,以实施例140化合物140与2-氟吡啶-5-硼酸酯偶联即可得到化合物141,MS m/z:656(M+1)
+.
实施例142化合物142的制备
以实施例141化合物141在三乙胺存在下与N,N-二甲基乙二胺在四氢呋喃中反应即可得到化合物142,MS m/z:724(M+1)
+.
实施例143化合物143的制备
参照实施例18步骤2方法,以实施例140化合物140与1,5-二甲基-1H-吡唑-4-硼酸频哪醇酯偶联即可得到化合物143,MS m/z:655(M+1)
+.
实施例144化合物144的制备
参照实施例18步骤2方法,以实施例140化合物140与1,3-二甲基-1H-吡唑-4-硼酸频那醇酯偶联即可得到化合物144,MS m/z:655(M+1)
+.
实施例145化合物145的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-b为原料经过与实施例74中间体邻苯二胺74-8缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,氢化脱Cbz,最后与甲胺磺酰氯缩合可得到化合物145,MS m/z:666(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ12.47(d,J=10.0Hz,1H),8.84(dd,J=24.4,9.6Hz,1H),8.08(s,1H),7.82(s,1H),7.64–7.57(m,2H),7.48(d,J=8.4Hz,1H),7.39(s,2H),7.30–7.24(m,2H),6.63(d,J=5.2Hz,2H),6.52(s,1H),6.02(t,J=10.4Hz,1H),4.27(s,1H),3.89(s,3H),3.77(s,3H),3.00(d,J=6.8Hz,2H),2.34(d,J=4.8Hz,3H),1.84(s,1H),1.36(s,6H),0.81(d,J=6.4Hz,6H).
实施例146化合物146的制备
步骤1中间体146-1的制备
参照实施例23的方法,对硝基苯乙酸乙酯经氢化,硝化间位引入硝基,脱乙酰基,氢化还原步骤即可得到中间体146-1,MS m/z:195(M+1)
+.
步骤2化合物146的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-b为原料经过与实施 例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物146,MS m/z:507(M+1)
+.
1HNMR(400MHz,MeOD):δ7.51(d,J=2.0,1H),7.37-7.35(m,2H),7.24-7.21(m,2H),7.17-7.13(m,1H),7.09-7.05(m,2H),6.90(d,J=2.0,1H),5.92(d,J=10.0,1H),4.58-4.51(m,2H),4.24-4.20(m,1H),3.54(s,2H),2.69(s,3H),2.45-2.40(m,1H),1.37(t,J=7.2,3H),1.05(d,J=6.8,3H),0.94(d,J=8.4,3H)
实施例147化合物147的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-a为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物147,MS m/z:507(M+1)
+.
实施例148化合物148的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物148,MS m/z:507(M+1)
+.
1HNMR(400MHz,MeOD):δ8.51(s,1H),7.51(d,J=6.8,3H),7.38-7.31(m,3H),7.24-7.19(m,2H),6.47(s,1H),5.99(d,J=9.6,1H),4.40-4.29(m,2H),4.20-4.17(m,1H),3.61(s,2H), 2.72(s,3H),1.91-1.86(m,1H),1.23(t,J=7.2,3H),0.96(d,J=6.8,3H),0.85(d,J=6.8,3H).
实施例149化合物149的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-d为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物149,MS m/z:507(M+1)
+.
实施例150化合物150的制备
参照实施例74方法,以实施例3中间体3的单一手性异构体3-b为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物150,MS m/z:507(M+1)
+.
实施例151化合物151的制备
参照实施例74方法,以实施例3中间体3的单一手性异构体3-a为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物151,MS m/z:507(M+1)
+.
实施例152化合物152的制备
参照实施例74方法,以实施例3中间体3的单一手性异构体3-c为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物152,MS m/z:507(M+1)
+.
实施例153化合物153的制备
参照实施例74方法,以实施例3中间体3的单一手性异构体3-d为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实 施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物153,MS m/z:507(M+1)
+.
实施例154化合物154的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-b为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物154,MS m/z:535(M+1)
+.
1HNMR(400MHz,MeOD):δ7.57-7.48(m,3H),7.37-7.30(m,3H),7.24-7.19(m,3H),6.45(s,1H),5.97(d,J=10.0,1H),4.36-4.29(m,2H),4.22-4.18(m,1H),2.67(d,J=4.0,3H),1.89-1.85(m,1H),1.59(s,6H),1.22(t,J=7.2,3H),0.94(d,J=5.6,3H),0.85(d,J=6.4,3H).
实施例155化合物155的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-a为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物155,MS m/z:535(M+1)
+.
实施例156化合物156的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物156,MS m/z:535(M+1)
+.
1HNMR(400MHz,MeOD):δ7.50(d,J=2.0,1H),7.42-7.35(m,2H),7.27-7.21(m,2H),7.18-7.04(m,3H),6.69(d,J=2.0,1H),5.91(d,J=10.4,1H),4.56-4.49(m,2H),4.26-4.22(m,1H),2.62(s,3H),2.45-2.40(m,1H),1.52(s,6H),1.35(t,J=7.2,3H),1.03(d,J=6.4,3H),0.94(d,J=6.8,3H).
实施例157化合物157的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-d为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物157,MS m/z:535(M+1)
+.
实施例158化合物158的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物158,MS m/z:615(M+1)
+.
实施例159化合物159的制备
参照实施例74方法,以实施例21中间体21的单一手性异构体21-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-乙基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物159,MS m/z:642(M+1)
+.
实施例160化合物160的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物160,MS m/z:701(M+1)
+.
实施例161化合物161的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-d为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物161,MS m/z:701(M+1)
+.
实施例162化合物162的制备
步骤1中间体162-1的制备
将Boc-D-亮氨酸(2.49g,10mmol)溶于DMF(25mL),冰浴下加入HBTU(4.17g,11mmol)、DBU(2.58g,20mmol)和甲基磺酰胺(1.9g,20mmol),加毕升至室温搅拌反应5小时。然后用水和乙酸乙酯萃取,再将水相用乙酸乙酯萃取两次,合并有机相,无水硫酸钠干燥,减压蒸除溶剂后得中间体162-1粗品(1.5g,6.5mmol,产率50%)。MS m/z:309(M+1)
+.
步骤2中间体162-2的制备
将中间体162-1(1.5g,6.5mmol)加入到二氯甲烷(20mL)中,冰浴下加入三氟乙酸(10mL),冰浴搅拌1小时,减压蒸除溶剂,即得中间体9粗品(0.68g,11.9mmol,产率80%),MS m/z:209(M+1)
+.
步骤3化合物162的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与实施例162步骤2中间体162-2缩合即可得到化合物162,MS m/z:778(M+1)
+.
实施例163化合物163的制备
步骤1中间体163-1的制备
参照实施例162步骤1-2方法,由Boc-D-亮氨酸为起始原料,与乙基磺酰氯磺酰化和脱Boc保护基得到中间体163-1,MS m/z:223(M+1)
+.
步骤2化合物163的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与实施例163步骤1中间体163-1缩合即可得到化合物163,MS m/z:792(M+1)
+.
1HNMR(400MHz,DMSO-d
6)δ=12.63(s,1H),8.94(d,J=8.8Hz,1H),8.27(s,4H),8.08(s,1H),7.82(s,1H),7.56(s,2H),7.44-7.34(m,1H),7.28(d,J=2.0Hz,1H),7.17(d,J=8.8Hz,1H),6.53(s,1H),6.02(t,J=10.4Hz,1H),4.31-4.25(m,1H),4.05-3.99(m,1H),3.89(s,3H),3.76(s,3H),2.88-2.76(m,2H),1.85(s,1H),1.52(d,J=6.8Hz,6H),1.47-1.39(m,2H),1.36-1.28(m,1H),0.97(t,J=7.2Hz,3H),0.83-0.78(m,12H).
实施例164化合物164的制备
步骤1中间体164-1的制备
参照实施例162步骤1-2方法,由Boc-D-亮氨酸为起始原料,与环丙基磺酰氯磺酰化和脱Boc保护基得到中间体164-1,MS m/z:235(M+1)
+.
步骤2化合物164的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与实施例163步骤1中间体164-1缩合即可得到化合物164,MS m/z:804(M+1)
+.
实施例165化合物165的制备
步骤1中间体165-1的制备
将Boc-D-亮氨酸(2.49g,10mmol)溶于DMF(25mL),冰浴下加入HBTU(4.17g,11mmol)、DBU(2.58g,20mmol)和氯化铵(1.07g,20mmol),加毕升至室温搅拌反应5小时。然后用水和乙酸乙酯萃取,再将水相用乙酸乙酯萃取两次,合并有机相,无水硫酸钠干燥,减压蒸除溶剂后得中间体165-1粗品(1.5g,6.5mmol,产率50%)。MS m/z:231(M+1)
+.
步骤2中间体165-2的制备
将中间体165-1(1.5g,6.5mmol)加入到DMF(20mL)中,冰浴下加入三聚氯氰(0.7g,3.9mmol),室温搅拌10小时,减压蒸除溶剂,即得中间体165-2粗品(0.64g,3mmol,产率46%),MS m/z:213(M+1)
+.
步骤3中间体165-3的制备
将中间体165-2(0.64g,3mmol)加入到DMF(20mL)中,加入叠氮钠(0.29g,4.5mmol),氯化铵(0.24g,4.5mmol)100℃搅拌10小时,减压蒸除溶剂,即得中间体165-3粗品(0.51g,2mmol,产率66%),MS m/z:256(M+1)
+.
步骤4中间体165-4的制备
将中间体165-3(0.51g,2mmol)加入到二氯甲烷(20mL)中,冰浴下加入三氟乙酸(10mL),冰浴搅拌1小时,减压蒸除溶剂,即得中间体11粗品(0.28g,1.8mmol,产率90%),MS m/z:156(M+1)
+.
步骤5化合物165的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与中间体165-4缩合即可得到化合物165,MS m/z:725(M+1)
+.
1HNMR(400MHz,DMSO-d
6)δ=8.86(d,J=9.2Hz,1H),8.07(s,1H),7.81(s,1H),7.55(s,1H),7.49(t,J=6.4Hz,3H),7.38(s,2H),7.28(d,J=2Hz,1H),7.11(d,J=8.8Hz,1H), 6.51(d,J=1.6Hz,1H),6.00(t,J=10.4Hz,1H),5.24(dd,J
1=7.6Hz,J
2=14.4Hz,1H),4.25(d,J=10.8Hz,1H),3.88(s,3H),3.75(s,3H),1.77~1.86(m,1H),1.70~1.75(m,1H),1.58(t,J=7Hz,1H),1.52(d,J=6.4Hz,6H),1.29~1.39(m,1H),0.81(s,4H),0.78(s,6H),0.77(s,2H)
实施例166化合物166的制备
参照实施例74方法,以实施例4中间体4的单一手性异构体4-c为原料经过与实施例30中间体邻苯二胺30缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基即可得到化合物166,MS m/z:482(M+1)
+.
1H NMR(400MHz,DMSO-d
6):δ12.55(s,1H),8.89-8.94(t,1H),7.08-7.56(m,6H),6.563-6.568(d,J=1.5Hz,1H),5.84-5.89(t,1H),4.23-4.26(m,1H),3.87(s,3H),2.98-3.03(m,1H),1.82-1.83(m,1H),1.25-1.27(d,J=6.8Hz,5H),0.78-0.79(d,J=7.2Hz,5H).
实施例167化合物167的制备
参照实施例74方法,以实施例4中间体4的单一手性异构体4-b为原料经过与实施例30中间体邻苯二胺30缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基即可得到化合物167,MS m/z:482(M+1)
+.
1H NMR(400MHz,DMSO-d
6):δ12.15(s,1H),9.10-9.12(d,J=8.8Hz,1H),8.21(s,1H),6.97-7.47(m,8H),5.81-5.86(t,J=1.5Hz,1H),4.26-4.30(m,1H),4.06(s,3H),3.87(s,1H),2.91-2.94(m,1H),2.40-2.46(m,1H),1.19-1.27(d,J=6.8Hz,6H),0.84-0.91(d,J=7.2Hz,6H).
实施例168化合物168的制备
参照实施例74方法,以实施例94步骤1中间体94-1的单一手性异构体94-1c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸叔丁酯盐酸盐缩合,最后叔丁酯经三氟乙酸水解即可得到化合物168,MS m/z:587(M+1)
+.
实施例169化合物169的制备
参照实施例74方法,以实施例94步骤1中间体94-1的单一手性异构体94-1c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与实施例162中间体162-2缩合即可得到化合物169,MS m/z:664(M+1)
+.
实施例170化合物170的制备
参照实施例74方法,以Boc-L-3,3-二苯基丙氨酸为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经 碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物170,MS m/z:621(M+1)
+.
实施例171化合物171的制备
步骤1中间体171-1的制备
参照实施例1制备中间体1的方法,以1-甲基-1H-吲唑-6-甲醛为起始原料、经与硝基乙酸乙酯缩合、与异丙基氯化镁进行格氏反应、硝基锌粉还原、Boc保护氨基、碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体171-1的四个单一手性异构体171-1a,171-1b,171-1c,171-1d。MS m/z:362[M+1]
+。
步骤2化合物171的制备
参照实施例74方法,以实施例171步骤1中间体171-1的单一手性异构体171-1c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物171,MS m/z:641(M+1)
+.
实施例172化合物172的制备
参照实施例74方法,以实施例171步骤1中间体171-1的单一手性异构体171-1b为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物172,MS m/z:641(M+1)
+.
实施例173化合物173的制备
步骤1中间体173-1的制备
将硝基乙酸乙酯(9.50g,71.34mmol)和环丁酮(5g,71.34mmol)溶于无水四氢呋喃(200mL),氮气保护下在0℃缓慢滴加四氯化钛(27.06g,142.67mmol),滴加完毕后继续在0℃搅拌反应1小时。将N-甲基吗啉(28.82g,285.35mmol)缓慢滴加入反应液,滴加完毕缓慢升至室温,继续搅拌反应2小时。加入蒸馏水淬灭反应,用乙酸乙酯萃取,合并有机相,无水硫酸钠干燥,过滤,减压蒸除溶剂,经柱层析纯化(洗脱剂:石油醚:乙酸乙酯=50:1)后得中间体173-1(10g,54.00mmol,75.70%yield)。
步骤2中间体173-2的制备
参照实施例1制备中间体1的方法,经由中间体173-1与苯基溴化镁进行格氏反应、 硝基还原、Boc保护氨基、乙酯碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体173-2的两个单一手性异构体173-2a和173-2b,MS m/z:306[M+1]
+。
步骤3化合物173的制备
参照实施例74方法,以实施例173步骤2中间体173-2的单一手性异构体173-2a为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物173,MS m/z:585(M+1)
+.
实施例174化合物174的制备
参照实施例74方法,以实施例173步骤2中间体173-2的单一手性异构体173-2b为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物174,MS m/z:585(M+1)
+.
实施例175化合物175的制备
步骤1中间体175-1的制备
参照实施例1制备中间体1的方法,经由中间体173-1与邻氯苯基溴化镁进行格氏反应、硝基还原、Boc保护氨基、乙酯碱水解,最后经SFC手性拆分柱分离制备可分别得到中间体175-1的两个单一手性异构体175-1a和175-1b,MS m/z:340[M+1]
+。
步骤2化合物175的制备
参照实施例74方法,以实施例173步骤2中间体175-1的单一手性异构体175-1a为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物175,MS m/z:619(M+1)
+.
实施例176化合物176的制备
参照实施例74方法,以实施例173步骤2中间体175-1的单一手性异构体175-1b为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物176,MS m/z:619(M+1)
+.
实施例177化合物177的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与实施例162中间体162-2缩合即可得到化合物177,MS m/z:805(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ=11.77(s,1H),8.80(d,J=8.8Hz,1H),8.17(s,1H),7.83(s,1H),7.59-7.54(m,3H),7.42–7.30(m,4H),7.15(d,J=8.4Hz,1H),6.55(s,1H),6.02(t,J=10.0Hz,1H),4.55–4.49(m,1H),4.37-4.31(m,1H),4.22-4.21(m,1H),3.77(s,3H),3.21(d,J=1.6Hz,3H),1.87(s,1H),1.55–1.45(m,12H),1.25(s,1H),0.85–0.76(m,12H).
实施例178化合物178的制备
参照实施例74方法,以实施例19的中间体19(对映体19-c和19-d的混合物)为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物178,MS m/z:729(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ12.50(s,2H),8.81-8.79(d,J=9.2Hz,1H),8.13-8.11(d,J=8.4Hz,1H),7.80(s,1H),7.63–7.45(t,J=29.6Hz,3H),7.37–7.36(t,J=2.8Hz,2H),7.26–7.25(m,2H),7.13-7.11(d,J=8.4Hz,1H),6.50(s,1H),6.01-5.96(t,J=10Hz,1H),4.53-4.46(m,1H),4.23-4.21(d,J=10Hz,2H),4.23(s,3H),1.81(s,1H),1.59-1.25(m,15H),0.80–0.75(m,11H).
实施例179化合物179的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物179,MS m/z:729(M+1)
+.
1HNMR(400MHz,MeOD):δppm 12.20-13.13(m,1.5H),8.86-8.88(d,J=9.2Hz,1H),8.15-8.17(m,1H),7.82(s,1H),7.34-7.57(m,5H),7.22-7.27(m,2H),7.12-7.14(m,1H),6.52-6.53(m,1H),5.93-6.03(m,1H),4.46-4.56(m,1H),4.22-4.26(m,2H),4.22-4.26(m,2H),3.76(s,3H),1.80-1.84(m,1H),1.49-1.52(d,J=9.6Hz,6H),1.44-1.46(d,J=6.4Hz,1H),0.77-0.85(m,11H)。
实施例180化合物180的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,酯水解并氨解即可得到化合物180,MS m/z:728(M+1)
+.
实施例181化合物181的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实 施例41步骤6-7,经碱水解,再与D-N-甲基-伽马-羟基亮氨酸甲酯缩合,最后酯水解即可得到化合物181,MS m/z:745(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.76(d,J=8.4Hz,1H),8.27(s,1H),7.93(t,J=8.0Hz,2H),7.70(t,J=4.0Hz,3H),7.66(s,1H),7.47(dd,J 1=1.6Hz,J 2=8.4Hz,1H),7.41–7.39(m,2H),7.34(d,J=8.4Hz,1H),6.69(d,J=2.0Hz,1H),6.14(t,J=8.8Hz,1H),4.02(t,J=8.0Hz,1H),3.84(s,3H),2.23(dd,J=11.9,9.6Hz,1H),2.11–2.02(m,2H),1.53–1.34(m,18H),0.98(d,J=6.4Hz,3H),0.79(d,J=6.8Hz,3H).
实施例182化合物182的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-环丙基丙氨酸甲酯缩合,最后酯水解即可得到化合物182,MS m/z:727(M+1)
+.
1H NMR(400MHz,MeOD)δ8.06(s,1H),7.87(s,1H),7.68(dd,2H),7.59(dd,1H),7.44-7.42(d,J=2.1Hz,1H),7.37–7.33(m,3H),6.50(s,1H),6.11(d,J=9.5Hz,1H),4.59(dt,J=13.6,6.7Hz,2H),4.43(dd,J=7.4,4.9Hz,1H),4.19(dd,J=8.3,6.4Hz,1H),3.883(s,3H),3.53–3.48(m,1H),1.98(dd,J=12.0,7.5Hz,1H),1.66(d,J=12.0Hz,6H),1.56(d,J=12.0Hz,6H),1.39–1.23(m,3H),1.03(d,J=6.9Hz,3H),0.89(d,J=6.9Hz,3H),0.61–0.48(m,1H),0.30(ddd,J=11.5,7.7,3.9Hz,2H).
实施例183化合物183的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-环丙基甘氨酸甲酯缩合,最后酯水解即可得到化合 物183,MS m/z:713(M+1)
+.
1H NMR(400MHz,MeOD)δ8.06(s,1H),7.87(s,1H),7.65(d,J=2.9Hz,2H),7.57(d,J=8.4Hz,1H),7.43(dd,J=8.3,2.0Hz,1H),7.38–7.31(m,3H),6.51(d,J=2.2Hz,1H),6.12(d,J=9.2Hz,1H),4.59(m,J=11.8,5.9Hz,2H),4.23–4.14(m,2H),3.89(s,3H),3.82(d,J=9.4Hz,1H),1.99(d,J=4.6Hz,2H),1.65(d,J=7.3Hz,6H),1.56(d,J=6.7Hz,6H),1.04(d,J=5.9Hz,3H),0.89(d,J=6.8Hz,3H),0.54(m,J=12.2,5.0Hz,1H),0.46–0.40(m,2H),0.28–0.22(m,1H).
实施例184化合物184的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-脯氨酸甲酯缩合,最后酯水解即可得到化合物184,MS m/z:713(M+1)
+.
1H NMR(400MHz,DMSO-d
6):δ=8.38-8.33(m,1H),8.15(s,1H),7.81(s,1H),7.57-7.50(m,2H),7.37-7.33(m,1H),7.27-7.24(m,1H),6.64-6.61(m,1H),6.07-5.98(m,1H),4.54-4.52(m,1H),4.27-4.19(m,2H),3.79(s,3H),2.70-2.68(m,3H),2.03-2.01(m1H),1.87-1.85(m,1H),1.66-1.59(m,3H),1.53-1.44(m,13H),1.27-1.24(m,1H),0.80(s,6H)。
实施例185化合物185的制备
步骤1中间体185-1的制备
参照实施例18步骤2的方法,中间体1-4与1-环丙基吡唑-4-硼酸嚬哪醇酯偶联后再经水解,最后经SFC手性拆分柱分离制备可分别得到中间体185-1的四个单一手性异构体185-1a,185-1b,185-1c,185-1d。MS m/z:448[M+1]
+。
步骤2化合物185的制备
参照实施例74方法,以中间体185-1的单一手性异构体185-1c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后酯水解即可得到化合物185,MS m/z:727(M+1)
+.
实施例186化合物186的制备
步骤1中间体186-1的制备
参照实施例1制备中间体1的方法,以2-氯-5-溴苯甲醛为起始原料、经与硝基乙酸乙酯缩合、与环丙基氯化镁进行格氏反应、硝基锌粉还原、Boc保护氨基,即可得到中间体186-1,MS m/z:446[M+1]
+。
步骤2中间体186-2的制备
参照实施例18步骤2的方法,中间体186-1与1-异丙基吡唑-4-硼酸嚬哪醇酯偶联后再经水解,最后经SFC手性拆分柱分离制备可分别得到中间体186-1的四个单一手性异构体186-2a,186-2b,186-2c,186-2d。MS m/z:448[M+1]
+。
步骤3化合物186的制备
参照实施例74方法,以中间体186-2的单一手性异构体186-2c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯盐酸盐缩合,最后酯水解即可得到化合物186,MS m/z:727(M+1)
+.
1H NMR(400MHz,Methanol-d
4)δ8.04(s,1H),7.80(m,1H),7.73-7.67(s,2H),7.59(d,J=8.5Hz,1H),7.43–7.33(m,4H),6.63(s,1H),5.95-5.93(m,1H),4.61-4.54(m,1H),4.48-4.45(m,1H),3.87(s,3H),1.68-1.66(m,3H),1.64-1.62(m,3H),1.60-1.58(m,2H),1.55(d,J=6.7Hz,6H),1.51–1.46(m,1H),1.31-1.29(m,1H),0.90–0.83(m,6H),0.38-0.32(m,1H),0.29-0.22(m,1H),0.15–0.07(m,1H),-0.17--0.23(m,1H).
实施例187化合物187的制备
参照实施例74方法,以中间体186-2的单一手性异构体186-2c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-环丙基甘氨酸甲酯缩合,最后酯水解即可得到化合物187,MS m/z:711(M+1)+.
1H NMR(400MHz,DMSO-d
6)δ8.19(s,1H),7.85(s,1H),7.76(s,1H),7.66(d,J=7.9Hz,2H),7.48–7.42(m,1H),7.41–7.31(m,3H),6.63(s,1H),5.96(s,1H),4.58–4.46(m,1H),3.80(s,3H),3.48–3.40(m,1H),1.59–1.54(m,3H),1.54(s,3H),1.47–1.45(m,3H),1.44(s,3H),1.33–1.21(m,1H),1.16–1.04(m,1H),0.56–0.44(m,1H),0.41–0.24(m,3H),0.23–-0.10(m,5H),-0.18–-0.34(m,1H).
实施例188化合物188的制备
参照实施例74方法,以中间体186-2的单一手性异构体186-2c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-环丙基丙氨酸甲酯缩合,最后酯水解即可得到化合物188,MS m/z:725(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.22(s,1H),7.87(s,1H),7.80(s,1H),7.74–7.64(m,2H),7.52–7.33(m,4H),6.66(s,1H),6.03(s,1H),4.52(dt,J=13.3,6.6Hz,1H),4.29(dd,J=9.3,4.7Hz,1H),3.82(s,3H),1.69–1.61(m,1H),1.58(s,3H),1.56(s,3H),1.47(s,3H),1.46(s,3H),1.33–1.22(m,1H),0.62–0.48(m,1H),0.36–0.21(m,3H),0.19–0.10(m,1H),0.09–-0.03(m,3H),-0.04–-0.13(m,1H),-0.16–-0.32(m,1H).
实施例189化合物189的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(S)-3-氨基四氢呋喃-3-甲酸甲酯缩合,最后酯水解即可得到化合物189,MS m/z:729(M+1)
+.
1H NMR(401MHz,MeOD)δ8.04–8.02(m,1H),7.85–7.83(m,1H),7.64–7.60(m,2H),7.55–7.51(m,1H),7.39(s,1H),7.31(s,4H),6.49–6.46(s,1H),6.11–6.06(m,1H),4.60–4.53(m,1H),4.21–4.15(m,2H),3.86(s,3H),2.42–2.19(m,2H),2.03–1.93(m,1H),1.61(s,6H),1.54(d,J=6.8Hz,6H),1.00(s,3H),0.87(d,J=6.6Hz,3H).
实施例190化合物190的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与3-氨基氧杂环丁烷-3-甲酸乙酯缩合,最后酯水解即可得到化合物190,MS m/z:715(M+1)
+.
1H NMR(400MHz,MeOD)δ8.07(s,1H),7.88(s,1H),7.74(d,J=5.8Hz,1H),7.64(d,J=9.0Hz,1H),7.45–7.33(m,3H),6.50–6.48(m,1H),6.10(s,1H),4.30(d,J=6.6Hz,1H),4.12(m,2H),3.86(m,2H),1.67(s,2H),1.56(d,J=6.8Hz,2H),1.26(t,J=7.1Hz,2H),1.22(s,1H),1.00(t,J=7.4Hz,3H),0.94–0.86(m,3H).
实施例191化合物191的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-氨基环丙烷羧酸甲酯缩合,最后酯水解即可得到化合物191,MS m/z:699(M+1)
+.
1H NMR(400MHz,MeOD)δ8.04(s,1H),7.85(s,1H),7.62(s,2H),7.52(d,J=8.4Hz,1H),7.41(dd,J=8.3,2.0Hz,1H),7.31(ddd,J=10.4,9.7,5.0Hz,3H),6.47(s,1H),6.08(d,J=9.5Hz,1H),4.56(m,1H),4.20–4.14(m,1H),3.86(s,3H),2.00–1.91(m,1H),1.59(s,6H),1.53(d,J=6.7Hz,6H),1.45(dd,J=7.8,4.6Hz,2H),1.01(d,J=3.8Hz,2H),1.00–0.96(m,3H),0.87(d,J=6.8Hz,3H).
实施例192化合物192的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与L-亮氨酸甲酯盐酸盐缩合,最后经碱水解甲酯即可得到化合物192,MS m/z:729(M+1)
+.
1H NMR(400MHz,MeOD)δ8.09(s,1H),7.89(s,1H),7.69(d,J=1.3Hz,1H),7.67(d,J=1.3Hz,3H),7.66(d,J=1.2Hz,1H),7.64(d,J=1.6Hz,2H),7.40(d,J=8.2Hz,2H),7.37(d,J=1.7Hz,1H),6.54(s,1H),6.15(d,J=8.9Hz,1H),4.58(dd,J=13.4,6.7Hz,1H),4.48(d,J=4.8Hz,1H),4.18(s,1H),3.91(s,3H),2.04(s,1H),1.66(d,J=15.1Hz,6H),1.56(d,J=6.7Hz,6H),1.32–1.29(m,3H),1.07(d,J=6.1Hz,3H),0.90(d,J=6.5Hz,3H),0.88–0.83(m,6H).
实施例193化合物193的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-脯氨酸甲酯缩合,最后酯水解并氨解即可得到化合物193,MS m/z:712(M+1)
+.
1H NMR(400MHz,MeOD)δ7.82(d,J=5.1Hz,1H),7.53–7.44(m,4H),7.29(s,2H),7.17(d,J=7.9Hz,1H),7.10(d,J=14.2Hz,1H),6.93(s,1H),6.01(d,J=7.5Hz,1H),4.63(s,1H),4.49(s,1H),4.42–4.36(m,1H),4.13(s,3H),2.83(s,2H),2.58–2.41(m,2H),2.10–1.99(m,1H),1.76–1.56(m,5H),1.51(d,J=5.4Hz,6H),1.34–1.30(m,2H),1.24–1.19(m,3H),0.92(s,3H).
实施例194化合物194的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-顺-3-羟基脯氨酸甲酯缩合,最后酯水解即可得到化合物194,MS m/z:729(M+1)
+.
1H NMR(400MHz,Methanol-d
4)δ8.04(s,1H),7.85(s,1H),7.62(s,1H),7.57-7.56(m,1H),7.42-7.40(m,1H),7.36–7.27(m,3H),7.24(d,J=8.6Hz,1H),6.47(s,1H),6.10(d,J=8.5Hz,1H),4.60-4.51(m,1H),4.45-4.42(m,1H),4.20-4016(m,1H),4.10(s,1H),3.94-3.91(m,1H),3.86(s,3H),3.50–3.41(m,1H),3.14–3.05(m,1H),2.83–2.74(m,1H),2.29-2.25(m,1H),1.98-1.95(m,1H),1.72-1.68(m,1H),1.61(s,2H),1.58(s,2H),1.53(d,J=6.7Hz,6H),1.50-1.47(m,1H),1.02(d,J=6.7Hz,2H),0.88(d,J=6.7Hz,4H).
实施例195化合物195的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与氮杂环庚烷-2-甲酸甲酯缩合,最后酯水解即可得到化合物195,MS m/z:729(M+1)
+.
1H NMR(400MHz,MeOD)δ8.06(s,1H),7.87(s,1H),7.61(d,J=29.3Hz,3H),7.51(s,1H),7.43(dd,J=8.3,1.9Hz,1H),7.34(d,J=8.4Hz,2H),7.27(d,J=7.9Hz,1H),7.19(d,J=8.4Hz,1H),6.53(s,1H),6.13(d,J=8.6Hz,1H),4.59(dt,J=13.4,6.8Hz,1H),4.49(s,1H),4.28(s,1H),4.13(s,2H),3.91(d,J=5.4Hz,3H),2.19(d,J=16.9Hz,1H),2.02(s,2H),1.87(s,2H),1.62(d,J=15.2Hz,6H),1.56(d,J=6.7Hz,6H),1.44(s,2H),1.22(d,J=18.8Hz,2H),1.08(s,3H),0.88(d,J=6.7Hz,3H).
实施例196化合物196的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-反-3-羟基脯氨酸甲酯缩合,最后酯水解即可得到化合物196,MS m/z:729(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ8.14(s,1H),7.93(s,1H),7.79-7.73(d,J=4.1Hz,3H),7.58-7.50(m,3H),7.44–7.38(m,3H),7.36–7.29(m,3H),7.27-7.25(m,2H),6.69(t,J=4.1Hz,1H),6.28(d,J=7.4Hz,1H),4.63-4.56(m,2H),4.45–4.33(m,2H),4.14–4.05(m,2H),3.99(d,J=1.9Hz,4H),3.76–3.61(m,3H),2.29(dd,J=13.6,6.9Hz,2H),1.67(d,J=3.2Hz,2H),1.62(d,J=3.9Hz,2H),1.56(dd,J=6.5,4.5Hz,8H),1.44(d,J=18.4Hz,10H),1.32–1.18(m,6H),1.13–1.00(m,7H),0.91(d,J=6.5Hz,3H),0.79(dd,J=16.2,6.7Hz,4H).
实施例197化合物197的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-吗啉-3-甲酸甲酯盐酸盐缩合,最后酯水解即可得到化合物197,MS m/z:729(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ8.14(s,1H),7.92(s,1H),7.80(d,J=11.0Hz,2H),7.72(d,J=6.5Hz,1H),7.67–7.57(m,2H),7.57–7.45(m,2H),7.44–7.28(m,2H),6.71(s,1H),6.33(dd,J=8.4,3.5Hz,1H),4.59(d,J=10.4Hz,2H),4.16(d,J=12.9Hz,2H),3.97(d,J=9.0Hz,3H),3.03–2.91(m,1H),2.76(dd,J=11.4,3.8Hz,1H),2.31–2.08(m,2H),1.90(d,J=4.4Hz,1H),1.80–1.45(m,14H),1.18(d,J=6.5Hz,3H),1.06(d,J=2.7Hz,1H),0.91(d,J=6.6Hz,3H).
实施例198化合物198的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)--环丁胺-2-羧酸甲酯缩合,最后酯水解即可得到化合物198,MS m/z:699(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ8.06(s,1H),7.87(s,1H),7.68–7.54(m,3H),7.54–7.40(m,2H),7.40–7.27(m,4H),6.52(d,J=2.2Hz,1H),6.13(d,J=9.2Hz,1H),4.62–4.53(m,2H),4.13(s,2H),3.90(s,3H),2.10–1.76(m,3H),1.71–1.44(m,16H),1.31(s,1H),1.25-1.21(m,1H),1.06(d,J=6.8Hz,3H),0.90(d,J=6.8Hz,4H).
实施例199化合物199的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与反式-4-氟-L-脯氨酸甲酯缩合,最后酯水解即可得到化合物199,MS m/z:731(M+1)
+.
1H NMR(400MHz,MeOD)δ8.06(s,1H),7.87(s,1H),7.66(d,J=12.4Hz,1H),7.59(d,J=8.1Hz,2H),7.43(dd,J=8.3,1.9Hz,1H),7.38–7.33(m,2H),7.28(d,J=9.4Hz,1H),6.50(s,1H),6.12(d,J=9.4Hz,1H),4.59(dt,J=13.5,6.9Hz,2H),4.18(s,1H),3.89(s,3H),2.55–2.36(m,2H),2.00(d,J=5.1Hz,2H),1.88(s,1H),1.62(d,J=8.6Hz,6H),1.56(d,J=6.7Hz,6H),1.05(d,J=6.0Hz,3H),0.90(d,J=6.7Hz,3H).
实施例200化合物200的制备
参照实施例74方法,以实施例186中间体186-2的单一手性异构体186-2c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-环丙基甘氨酸甲酯缩合即可得到化合物187,MS m/z:725(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.19(d,J=2.0Hz,1H),7.84(d,J=0.8Hz,1H),7.73-7.68(m,1H),7.58(d,J=7.2Hz,1H),7.44–7.41(m,1H),7.38-7.35(m,2H),7.21(d,J=8.4Hz,1H),6.63(d,J=4.0Hz,1H),5.95(s,1H),4.55–4.49(m,1H),3.80(s,H),3.62(s,3H),3.61–3.60(m,2H),1.54–1.45(m,12H),1.16–1.08(m,2H),0.52–0.48(m,1H),0.41–-0.02(m,7H),-0.29(s,1H).
施例201化合物201的制备
参照实施例74方法,以中间体186-2的单一手性异构体186-2c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-环丙基丙氨酸甲酯缩合可得到化合物201,MS m/z:739(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.21(s,1H),7.87(s,1H),7.79(s,1H),7.74(d,J=8.6Hz,1H),7.69(d,J=1.2Hz,1H),7.47(dd,J=8.3,2.1Hz,1H),7.44–7.36(m,3H),6.65(s,1H),6.02(s,1H),4.57–4.47(m,1H),4.35–4.28(m,1H),3.81(s,3H),1.69–1.61(m,1H),1.56(s,3H),1.55(s,3H),1.48–1.46(m,3H),1.46–1.43(m,3H),1.32–1.21(m,1H),0.63–0.52(m,1H),0.36–0.25(m,3H),0.20–0.10(m,1H),0.09–-0.03(m,3H),-0.04–-0.11(m,1H),-0.16–-0.30(m,1H).
实施例202化合物202的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-c为原料经过与实施例23中间体邻苯二胺23缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与甲胺缩合即可得到化合物202,MS m/z:615(M+1)
+.
1H NMR(400MHz,MeOD)δ8.50(s,1H),8.03(s,1H),7.84(s,1H),7.61(s,1H),7.56(s,1H),7.54–7.47(m,1H),7.43–7.38(m,1H),7.38–7.29(m,2H),7.29–7.22(m,1H),6.47(s,1H),6.08(d,J=9.7Hz,1H),4.21–4.13(m,1H),3.84(s,3H),3.78–3.71(m,1H),2.69(s,3H),1.93(d,J=7.1Hz,1H),1.57–1.46(m,9H),0.99(d,J=6.2Hz,3H),0.87(d,J=6.8Hz,3H).
实施例203化合物203的制备
参照实施例74方法,以实施例19的中间体19的单一手性异构体19-d为原料经过与实施例23中间体邻苯二胺23缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与甲胺缩合即可得到化合物203,MS m/z:615(M+1)
+.
1H NMR(400MHz,CD
3OD)δ=7.69(s,1H),7.52-7.44(m,4H),7.29-7.25(m,3H),7.13-7.08(d,J=22.0,1H),6.95(s,1H),6.02-5.99(d,J=8.8,1H),4.50-4.32(m,1H),4.21-4.14(m,1H),4.11(s,3H),3.74-3.68(m,1H),2.7-2.68(d,J=2.8,3H),2.53-2.45(m,1H),1.50-1.44(m,9H),1.21-1.18(t,J=6.8,3H),0.95-0.92(m,3H).
实施例204化合物204的制备
参照实施例74方法,以实施例6的中间体6的单一手性异构体6-c为原料经过与实施例41中间体邻苯二胺41-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与甲胺缩合即可得到化合物204,MS m/z:518(M+1)
+.
实施例205化合物205的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物205,MS m/z:493(M+1)
+.
实施例206化合物206的制备
参照实施例74方法,以实施例2中间体2的(单一手性异构体2-c为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与R-N-甲基-2-氨基-丙酰胺缩合可得到化合物206,MS m/z:564(M+1)
+.
实施例207化合物207的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与R-2-氨基-丙酰胺缩合可得到化合物207,MS m/z:550(M+1)
+.
实施例208化合物208的制备
参照实施例74方法,以实施例5中间体5的单一手性异构体5-c为原料经过与实施例30中间体邻苯二胺30缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基可得到化合物208,MS m/z:494(M+1)
+.
实施例209化合物209的制备
参照实施例74方法,以实施例4中间体4的单一手性异构体4-c为原料经过与实施例30中间体邻苯二胺30缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基可得到化合物209,MS m/z:482(M+1)
+.
实施例210化合物210的制备
参照实施例74方法,以实施例4中间体4的单一手性异构体4-d为原料经过与实施例30中间体邻苯二胺30缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基可得到化合物210,MS m/z:482(M+1)
+.
实施例211化合物211的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-d为原料经过与实施例37中间体邻苯二胺37缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基可得到化合物211,MS m/z:574(M+1)
+.
实施例212化合物212的制备
参照实施例74方法,以实施例2中间体2单一手性异构体2-c为原料经过与实施例37中间体邻苯二胺37缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基可得到化合物212,MS m/z:574(M+1)
+.
实施例213化合物213的制备
参照实施例74方法,以实施例2中间体2(未拆分的四个手性异构体混合物)为原料经过与实施例35中间体邻苯二胺35缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基可得到化合物213,MS m/z:516(M+1)
+.
实施例214化合物214的制备
步骤1中间体214-1的制备
参照实施例38方法,由中间体38-1与2-(4-溴-1,3-二甲基-1H-吡唑-5-基)乙酸甲酯偶联即可得到中间体214-1,MS m/z:261(M+1)
+.
步骤2化合物214的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与中间体邻苯二胺214-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与2-丙胺缩合可得到化合物214,MS m/z:601(M+1)
+.
实施例215化合物215的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-b为原料经过与中间 体邻苯二胺214-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与环戊胺缩合可得到化合物215,MS m/z:627(M+1)
+.
实施例216化合物216的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与中间体邻苯二胺214-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与环戊胺缩合可得到化合物216,MS m/z:627(M+1)
+.
实施例217化合物217的制备
步骤1中间体217-1的制备
参照实施例38方法,由中间体38-1与溴代吡唑物“6-苄基-4-甲基-3-溴-2-甲基-4,5,7,8-四氢-6H-吡唑[1,5-d]二氮杂卓-4,6-二羧酸酯”偶联可得到中间体217-1,MS m/z:450(M+1)
+.
步骤2化合物217的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与中间体邻苯二胺217-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,与环戊胺缩合,最后钯碳氢化脱苄氧羰基保护基可得到化合物217,MS m/z: 682(M+1)
+.
实施例218化合物218制备
步骤1中间体218-1的制备
参照实施例38方法,由中间体38-1与溴代吡唑物“甲基-3-溴-2-甲基-4,5,7,8-四氢吡唑[1,5-d][1,4]氧氮杂环庚烷-4-羧酸酯”偶联可得到中间体218-1,MS m/z:317(M+1)
+.
步骤2化合物218的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与中间体邻苯二胺218-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,与环戊胺缩合可得到化合物218,MS m/z:683(M+1)
+.
实施例219化合物219的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与中间体邻苯二胺217-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再经钯碳氢化脱苄氧羰基保护基可得到化合物219,MS m/z:629(M+1)
+.
实施例220化合物220的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例40中间体邻苯二胺40缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,可得到化合物220,MS m/z:601(M+1)
+.
1H NMR(400MHz,MeOD):δ=7.67-7.61(m,1H),7.53-7.51(m,1H),7.47-7.34(m,3H),7.27-7.21(m,2H),6.49(s,1H),6.02(d,J=9.6Hz,1H),5.03-4.98(m,1H),4.73(s,1H),4.24-4.16(m,1H),3.93(s,3H),2.79(s,3H),2.26(d,J=6.4Hz,4H),2.16(d,J=3.6Hz,2H),1.99-1.93(m,1H),1.76(d,J=7.2Hz,3H),0.99(d,J=6.8Hz,2H),0.89(d,J=6.8Hz,3H)。
实施例221化合物221的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与4-氨基四氢吡喃-4-羧酸甲酯缩合,最后酯水解并氨解即可得到化合物221,MS m/z:632(M+1)
+.
实施例222化合物222的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施 例41步骤6-7,经碱水解,再与3-(R)-氨基哌啶-2-酮缩合即可得到化合物222,MS m/z:602(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.91(s,1H),7.81-7.76(m,2H),7.69-7.67(m,2H),7.62-7.52(m,2H),7.49–7.37(m,4H),7.34-7.30(m,2H),6.67(s,1H),5.96-5.91(m,1H),4.28-4.21(m,1H),3.95(s,3H),3.31–3.28(m,2H),1.94–1.83(m,4H),1.67(d,J=5.2Hz,6H),0.68-.055(m,1H),0.25-0.19(m,2H),-0.02-0.08(m,1H).
实施例223化合物223的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(2R,4S)-反式-4-氟-D-脯氨酰胺缩合即可得到化合物223,MS m/z:620(M+1)
+.
1H NMR(400MHz,MeOD)δ7.57(d,J=8.1Hz,1H),7.41(d,J=2.1Hz,2H),7.39(s,1H),7.36–7.30(m,2H),7.28(s,1H),7.26(s,1H),7.23(d,J=6.9Hz,1H),6.63(s,1H),5.84(s,1H),5.02(s,1H),4.64–4.58(m,2H),3.91(d,J=1.1Hz,3H),3.14–3.51(m,1H),1.66(s,3H),1.60(s,3H),0.91(m,J=13.5,7.5Hz,3H),0.33(m,J=13.1,8.7,4.0Hz,2H),0.24(s,2H),0.07–0.00(m,2H),-0.25(s,2H).
实施例224化合物224的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-氨基环丙烷甲酰胺缩合即可得到化合物224,MS m/z:588(M+1)
+.
1H NMR(400MHz,MeOD)δ7.90(d,J=1.1Hz,1H),7.79(d,J=8.7Hz,1H), 7.69(dd,J=8.8,1.7Hz,1H),7.60–7.54(m,1H),7.48(dd,J=7.9,1.3Hz,1H),7.44(d,J=2.2Hz,1H),7.40(td,J=7.6,1.3Hz,1H),7.32(ddd,J=7.5,4.8,1.7Hz,1H),6.67(t,J=2.5Hz,1H),5.95(s,1H),3.96(s,3H),3.45(s,1H),2.88(s,1H),1.65(s,6H),1.51(q,J=7.9,4.6Hz,2H),1.06(q,J=7.9,4.6Hz,2H),0.97–0.85(m,1H),0.51–0.41(m,1H),0.40–0.30(m,1H),0.25–0.14(m,1H),-0.04(s,1H).
实施例225化合物225的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-环丙基丙氨酰胺缩合即可得到化合物225,MS m/z:616(M+1)
+.
1H NMR(400MHz,MeOD)δ7.85(d,J=1.3Hz,1H),7.81(s,1H),7.78(s,1H),7.66(dd,J=8.8,1.7Hz,1H),7.56(dd,J=6.4,4.9Hz,1H),7.48(dd,J=8.0,1.3Hz,1H),7.45(d,J=2.2Hz,1H),7.40(t,J=7.4Hz,1H),7.36–7.30(m,1H),7.17(dd,J=7.7,4.1Hz,1H),5.94–5.85(m,1H),4.58–4.37(m,1H),3.59–3.45(m,2H),3.27–2.95(m,1H),1.70(d,J=3.2Hz,6H),1.59–1.50(m,2H),0.68–0.60(m,2H),0.51–0.43(m,1H),0.43–0.35(m,3H),0.18(dd,J=9.9,5.4Hz,2H),0.08–0.02(m,2H).
实施例226化合物226的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与氨水发生氨解即可得到化合物226,MS m/z:505(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.84(d,J=1.2Hz,1H),7.80(dd,J=8.4,0.8Hz,1H),7.68-7.65(m,1H),7.60–7.55(m,1H),7.48(dd,J=8.0 1.2Hz,1H),7.44(d,J=2.0Hz,1H),7.42-7.38(m,1H),7.32-7.28(m,1H),6.66(d,J=2.0Hz,1H),5.94(s,1H),3.96(s,3H),3.52–3.45(m,1H),1.68(s,6H),1.63(s,1H),1.31(s,1H),0.48–0.42(m,1H),0.40-0.33(m,1H),0.21-0.16(m,1H),-0.04(s,1H).
实施例227化合物227的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与3-(R)-氨基-2-吡咯烷酮缩合即可得到化合物227,MS m/z:588(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.93(s,1H),7.81(d,J=8.8Hz,1H),7.71(dd,J=8.4,1.6Hz,1H),7.58(d,J=7.6Hz,1H),7.47(dd,J=8.0,1.6Hz,1H),7.43–7.42(m,1H),7.39(dd,J=7.6,1.6Hz,1H),7.34-7.30(m,1H),6.67(d,J=2.0Hz,1H),6.00(s,1H),4.46(t,J=8.8Hz,1H),3.96(s,3H),3.38(dd,J=9.6,2.8Hz,2H),2.47–2.39(m,1H),2.14-2.02(m,1H),1.68(d,J=2.4Hz,6H),1.31(s,1H),0.48–0.35(m,2H),0.22-0.16(m,1H),-0.06(s,1H).
实施例228化合物228的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-氨基-1-环戊甲酰胺缩合即可得到化合物228,MS m/z: 616(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.87(d,J=1.6Hz,1H),7.81(d,J=8.8Hz,1H),7.66-7.63(m,1H),7.59–7.54(m,1H),7.48-7.46(m,1H),7.44-7.42(m,1H),7.40-7.37(m,1H),7.34-7.30(m,1H),6.67(d,J=2.0Hz,1H),6.00(s,1H),3.96(s,3H),3.45(s,1H),2.18-2.13(m 2H),2.00–1.94(m,2H),1.76–1.50(m,12H),1.30(s,1H),0.49-0.42(m,1H),0.38-0.31(m,1H),0.22–0.16(m,1H).
实施例229化合物229的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-2-环丙乙酰胺缩合即可得到化合物229,MS m/z:602(M+1)
+.
1H NMR(400MHz,DMSO-d6)δ7.57–7.46(m,3H),7.43–7.34(m,2H),7.38–7.20(m,2H),7.24–7.11(m,1H),6.65-6.64(m,1H),5.90-5.81(m,1H),3.82(s,3H),3.81–3.70(m,2H),1.56–1.44(m,8H),1.53(d,J=4.8Hz,3H),1.21(d,J=6.6Hz,4H),1.02-0.95(m,1H),0.38-0.28(m,3H),0.23-0.18(m,1H),0.14-0.08(m,2H),0.01--0.09(m,1H)-0.33--0.41(m,1H).
实施例230化合物230的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与3-氨基四氢呋喃-3-甲酰胺缩合即可得到化合物230,MS m/z:618(M+1)
+.
1H NMR(400MHz,Methanol-d
4)δ7.58(s,2H),7.41–7.39(m,2H),7.33–7.30 (m,2H),7.25(dd,J=7.8,1.6Hz,1H),6.63(s,1H),5.84(s,1H),4.21(d,J=9.4Hz,1H),3.90(s,3H),3.86(dd,J=9.5,2.4Hz,1H),3.79–3.69(m,1H),2.44-2.37(m,1H),2.26-2.17(m,1H),1.68-1.61(m,7H),1.31(s,3H),0.91(d,J=9.2Hz,1H),0.34–0.23(m,3H),0.04(dd,J=9.6,4.8Hz,1H).
实施例231化合物231的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-氨基环丁烷甲酰胺缩合即可得到化合物231,MS m/z:602(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.64–7.52(m,3H),7.41-7.39(m,2H),7.35-7.31(m,2H),7.28–7.21(m,1H),6.63(s,1H),5.84(s,1H),3.90(s,3H),2.63–2.56(m,2H),2.20-2.12(m,2H),1.93–1.86(m,3H),1.66(s,7H),1.31-1.24(m,1H),0.435–0.21(m,2H),0.07-0.03(m,1H).
实施例232化合物232的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-氨基环丁烷甲酸甲酯缩合,最后酯水解即可得到化合物232,MS m/z:603(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.69(s,1H),7.59(d,J=8.4Hz,2H),7.43–7.20(m,8H),6.65(s,1H),5.87(s,1H),3.90(s,3H),3.62(s,1H),3.19–3.12(m,1H),3.00(s,1H),2.84(s,1H),2.57–2.35(m,5H),1.94(s,18H),1.78(d,J=3.6Hz,1H), 1.65(s,6H),1.37–1.20(m,4H),0.36–0.18(m,2H).
实施例233化合物233的制备
参照实施例74方法,以实施例12中间体12的单一手性异构体12-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酰胺缩合即可得到化合物233,MS m/z:618(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.85(s,1H),7.79(d,J=8.8Hz,1H),7.65-7.62(m,3H),7.49–7.40(m,3H),7.32-7.23(m,2H),6.65(d,J=2.0Hz,1H),5.93(s,1H),4.49(dd,J=10.4,4.4Hz,1H),3.96(s,3H),3.54–3.45(m,1H),1.69(s,6H),0.92-0.87(m,6H),0.49-0.42(m,1H),0.37(s,1H),0.21-0.15(m,1H).
实施例234化合物234的制备
参照实施例74方法,以实施例9中间体9为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-2-环丁基-N-甲基乙酰胺缩合即可得到化合物234,MS m/z:648(M+1)
+.
实施例235化合物235的制备
参照实施例74方法,以实施例10中间体10为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-2-环丁基-N-甲基乙酰胺缩合即可得到化合物235,MS m/z:648(M+1)
+.
1H NMR(400M,MeOD)δ7.70-7.79(m,2H),7.65-7.69(m,1H),7.50-7.60(m,3H),7.43-7.49(m,1H),7.35-7.42(m,2H),7.18-7.28(m,1H),6.89-7.01(m,2H),6.06-6.10(m,1H),5.71-5.73(m,1H),5.67-5.70(m,1H),4.28-4.35(m,1H),4.00(s,1H),3.90(s,2H),2.71-2.72(m,3H),2.49-2.60(m,1H),1.82-1.98(m,4H),1.74-1.81(m,1H),1.63-1.69(m,6H),0.60-0.67(m,1H),0.45-0.54(m,2H),0.31-0.43(m,2H).
实施例236化合物236的制备
参照实施例74方法,以实施例8中间体8为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-2-甲基-N-甲基乙酰胺缩合即可得到化合物236,MS m/z:594(M+1)
+.
实施例237化合物237的制备
参照实施例74方法,以实施例8中间体8为原料经过与实施例26中间体邻苯二胺 26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-2-异丙基-N-甲基乙酰胺缩合即可得到化合物237,MS m/z:622(M+1)
+.
实施例238化合物238的制备
参照实施例74方法,以实施例7中间体7为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-2-甲基-乙酰胺缩合即可得到化合物238,MS m/z:566(M+1)
+.
1H NMR(400M,CDCl
3)δ8.18(s,1H),7.88-8.06(m,1H),7.58-7.67(m,1H),7.50-7.57(m,1H),7.38-7.48(m,2H),7.30-7.38(m,2H),7.14-7.28(m,4H),6.78-6.90(m,1H),6.34-6.43(m,1H),5.84-6.09(m,3H),5.71-5.78(m,1H),5.54-5.60(m,1H),5.31-5.39(m,1H),4.49-4.57(m,1H),4.00-4.07(m,2H),3.94-3.98(m,1H),3.35-3.42(m,2H),3.24(s,1H),1.58-1.72(m,6H),1.23-1.32(m,3H).
实施例239化合物239的制备
参照实施例74方法,以实施例7中间体7为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-2-甲基-N-甲基乙酰胺缩合即可得到化合物239,MS m/z:580(M+1)
+.
1H NMR(400M,CDCl
3)δ8.14(s,1H),7.61-7.72(m,2H),7.50-7.59(m,2H),7.46-7.49(m,1H),7.41-7.45(m,1H),7.30-7.37(m,3H),7.14-7.26(m,4H),6.81-6.89(m,1H),6.28-6.41(m,2H),6.01-6.10(m,1H),5.82-5.95(m,2H),5.55-5.61(m,1H),5.31-5.41(m,1H),4.39-4.55(m,2H),4.03-4.13(m,3H),3.95-4.02(m,2H),3.36-3.48(m,3H),3.24(s,2H),2.70-2.83(m,5H),1.66(s,9H),1.17-1.36(m,6H).
实施例240化合物240的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与N,N-二甲基乙二胺缩合即可得到化合物240,MS m/z:578(M+1)
+.
实施例241化合物241的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与N-(2-氨基乙基)吗啉缩合即可得到化合物241,MS m/z:620(M+1)
+.
实施例242化合物242的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酸甲酯缩合,最后酯水解即可得到化合物242,MS m/z: 621(M+1)
+.
1HNMR(400MHz,MeOD):δ=8.52(s,1H),7.98(s,1H),7.86(s,1H),7.63(s,2H),7.34-7.48(m,3H),6.49(s,1H),6.09-6.12(m,1H),4.63(s,2H),4.19-4.21(m,1H),3.88-3.96(m,6H),3.34-3.40(m,2H),1.96-2.00(m,1H),1.60-1.66(m,1H),1.46-1.50(d,4H,J=16Hz),1.24-1.27(m,2H),1.02-1.03(d,3H,J=6Hz),0.89-0.91(d,3H,J=6.8Hz),0.73-0.75(t,3H,J=6Hz),0.35-0.37(d,3H,J=6.4Hz)
实施例243化合物243的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与实施例162中间体162-2缩合即可得到化合物243,MS m/z:698(M+1)
+。
实施例244化合物244的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与实施例165中间体165-4缩合即可得到化合物244,MS m/z:645(M+1)
+。
实施例245化合物245的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与实施例164中间体164-1缩合即可得到化合物245,MS m/z:724(M+1)
+。
实施例246化合物246的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与实施例163中间体163-1缩合即可得到化合物246,MS m/z:712(M+1)
+。
实施例247化合物247的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与D-亮氨酰胺缩合即可得到化合物247,MS m/z:620(M+1)
+。
实施例248化合物248的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-环丙基-甘氨酸甲酯缩合,最后经酯水解即可得到化合物248,MS m/z:605(M+1)
+。
实施例249化合物249的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-2-环丙甲基-乙酸乙酯缩合,最后经酯水解即可得到化合物249,MS m/z:619(M+1)
+.
实施例250化合物250的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与D-脯氨酸甲酯缩合,最后经酯水解即可得到化合物250,MS m/z: 605(M+1)
+.
1H NMR(400MHz,MeOD)δ7.58–7.55(m,2H),7.53–7.50(m,1H),7.37(dd,J=14.5,5.8Hz,3H),7.28–7.22(m,2H),6.52(s,1H),6.02(d,J=9.0Hz,1H),4.45(m,1H),4.18(s,1H),3.95(s,3H),2.94–2.88(m,2H),2.13(m,2H),1.96(m,1H),1.82–1.68(m,2H),1.61(d,J=6.8Hz,6H),1.02(d,J=5.7Hz,3H),0.87(d,J=6.8Hz,3H).
实施例251化合物251的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与D-脯氨酸甲酯缩合,再经酯水解和氨解即可得到化合物251,MS m/z:604(M+1)
+.
1H NMR(400MHz,MeOD)δ7.60–7.48(m,3H),7.39(dm,3H),7.24(m,2H),6.51(s,1H),6.02(d,J=9.0Hz,1H),4.44(m,1H),4.18(s,1H),3.94(s,3H),2.92(m,2H),1.96(m,1H),1.80–1.68(m,2H),1.65(s,3H),1.57(s,3H),1.54–1.42(m,2H),0.99(d,J=17.1Hz,3H),0.87(d,J=6.4Hz,3H).
实施例252化合物252的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与1-氨基环丙烷甲酸甲酯缩合,再经酯水解可得到化合物252,MS m/z:591(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.61(s,1H),7.54–7.46(m,2H),7.39–7.34(m,1H),7.34–7.26(m,2H),7.25–7.13(m,2H),6.46(s,1H),5.96(d,J=9.5Hz,1H),4.27–4.12(m,1H),4.09(s,1H),3.90(s,3H),1.95-1.81(m 1H),1.59(s,6H),1.52(s, 2H),1.47-1.39(m,2H),1.00–0.91(m,6H),0.84(d,J=6.8Hz,3H).
实施例253化合物253的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与3-(R)-氨基-2-吡咯烷酮缩合可得到化合物253,MS m/z:590(M+1)
+.
1H NMR(400MHz,MeOD)δ7.65(s,1H),7.57–7.49(m,3H),7.42–7.31(m,4H),7.27–7.21(m,2H),6.48(s,1H),5.99(d,J=9.5Hz,1H),4.45–4.36(m,1H),4.30–4.17(m,1H),3.92(s,3H),2.48–2.35(m,2H),2.08–1.85(m,3H),1.64(d,J=6.5Hz,6H),0.96(t,J=6.5Hz,3H),0.87(d,J=6.8Hz,3H).
实施例254化合物254的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-2-环丙甲基-乙酰胺缩合可得到化合物254,MS m/z:618(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.55-7047(m,2H),7.43-7.38(m,2H),7.34–7.29(m,2H),7.26–7.14(m,2H),6.86(d,J=8.4Hz,1H),6.48-6.47(m,1H),5.88(d,J=11.0Hz,1H),4.31–4.19(m,2H),3.82(s,3H),1.80-1.68(m,1H),1.54(d,J=7.3Hz,6H),1.40-1.33(m,1H),0.80-0.75(m,6H),0.52-0.44(m,1H),0.26-0.22(m,2H),-0.03--0.13(m,3H).
实施例255化合物255的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-2-环戊烷甲酰胺缩合可得到化合物255,MS m/z:618(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.64–7.46(m,3H),7.44-7.39(m,2H),7.37–7.29(m,2H),7.25-7.21(m,1H),7.13(d,J=8.6Hz,1H),6.90(s,1H),6.49-6.44(m,1H),5.88(d,J=9.6Hz,1H),3.82(s,3H),1.98-1.92(m,3H),1.84-1.75(m,4H),1.52(s,6H),1.45-1.41(m,2H),0.78(d,J=6.9Hz,6H).
实施例256化合物256的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后氨解可得到化合物256,MS m/z:507(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ12.53(d,J=14.8Hz,1H),8.76(dd,J=31.2,9.4Hz,1H),7.61–7.56(m,1H),7.47–7.40(m,3H),7.34–7.20(m,2H),7.25–7.16(m,2H),6.87-6.80(m,2H),6.50(d,J=2.4Hz,1H),5.90(t,J=10.0Hz,1H),4.32–4.24(m,1H),4.12(d,J=7.6Hz,1H),3.83(s,3H),3.18(d,J=4.4Hz,1H),1.78(d,J=9.2Hz,1H),1.50(s,6H),0.78(t,J=7.2Hz,6H).
实施例257化合物257的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与甲胺盐酸盐缩合可得到化合物257,MS m/z:521(M+1)
+.
实施例258化合物258的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-2-环丙基-乙酰胺缩合可得到化合物258,MS m/z:604(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.56(d,J=13.5Hz,2H),7.43(dd,J=8.0,2.4Hz,2H),7.38–7.28(m,2H),7.26–7.21(m,1H),7.18(d,J=8.6Hz,1H),6.51(s,1H),5.92(d,J=11.2Hz,1H),4.25(d,J=10.9Hz,1H),3.84(s,3H),3.80–3.73(m,1H),2.68(dd,J=3.7,1.8Hz,1H),2.34(dt,J=3.6,1.8Hz,1H),1.81(s,1H),1.53(s,6H),1.46(d,J=8.5Hz,1H),1.00(dt,J=11.0,4.1Hz,1H),0.81(t,J=11.9Hz,6H),0.44–0.28(m,3H),0.16(d,J=3.7Hz,1H),-0.05(s,1H).
实施例259化合物259的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与1-氨基环丙烷甲酰胺缩合可得到化合物259,MS m/z:590(M+1)
+.
实施例260化合物260的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与3-氨基四氢呋喃-3-甲酰胺缩合可得到化合物260,MS m/z:620(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.78(s,1H),7.75–7.66(m,3H),7.65–7.57(m,3H),7.54–7.43(m,5H),7.38(d,J=8.0Hz,1H),7.34–7.28(m,2H),7.24(dd,J=8.0,1.2Hz,1H),7.16–7.12(m,1H),7.08(d,J=2.0Hz,1H),6.71(s,1H),6.14(d,J=8.0Hz,1H),5.92(d,J=11.4Hz,1H),4.45(dd,J=11.4,5.2Hz,1H),4.27–4.21(m,2H),4.08(s,3H),3.99(s,3H),3.92(qd,J=8.4,4.8Hz,2H),3.85–3.69(m,4H),2.61(dt,J=12.8,6.4Hz,1H),2.40(dq,J=13.2,8.4Hz,2H),2.29-2.24(m,3H),1.63(dd,J=27.46 2.4Hz,11H),1.35–1.31(m,1H),1.13(d,J=6.8Hz,3H),1.02(dd,J=15.6,6.8Hz,5H),0.87(d,J=6.8Hz,4H).
实施例261化合物261的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与4-氨基四氢吡喃-4-羧酸甲酯缩合,最后酯水解并氨解即可 得到化合物261,MS m/z:634(M+1)
+.
实施例262化合物262的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与3-(R)-氨基哌啶-2-酮缩合即可得到化合物262,MS m/z:604(M+1)
+.
1H NMR(400MHz,Methanol-d4)δ7.83(s,1H),7.73-7.69(m,2H),7.65-7.62(m,2H),7.57–7.53(m,2H),7.45(s,2H),7.40-7.37(m,1H),7.34–7.24(m,3H),6.68(s,1H),6.13-6.09(m,1H),5.82-5.87(m,1H),4.43-4.37(m,1H),4.24-4.18(m,3H),4.08(s,2H),3.99(s,3H),3.30-3.27(m,3H),2.20-2.11(m,2H),1.98-1.95(m,3H),1.93–1.80(m,6H),1.67-1.63(m,6H),1.60-1.57(m,5H),1.33(t,J=7.3Hz,3H),1.12(d,J=6.5Hz,3H),1.05-1.01(m,3H),0.88(d,J=6.7Hz,3H).
实施例263化合物263的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-氨基环丁烷甲酰胺缩合即可得到化合物263,MS m/z:604(M+1)
+.
实施例264化合物264的制备
步骤1中间体264-1的制备
将4-溴-2-硝基-苯酚(4.36g,20.00mmol)与4-甲氧基苄溴(8.04g,40.00mmol)溶于DMF(40.00mL)反应液升温至80℃并搅拌2小时,反应完毕,冷却至室温,加水稀释,DCM萃取(2×60ml),合并有机相,水洗(3×60ml),饱和氯化钠水溶液洗涤,无水硫酸钠干燥,过滤,滤液减压浓缩,粗品经硅胶柱层析分离纯化(石油醚/乙酸乙酯30:1)即可得到中间体264-1(3.70g,10.94mmol,54.70%yield),LC-MS不响应。
步骤2中间体264-2的制备
将中间体264-1(6.00g,17.74mmol)、丙烯酸乙酯(5.33g,53.22mmol)溶于乙腈中(50mL),混合物氮气保护下升温至90℃并搅拌12小时,冷却至室温,减压浓缩后加水稀释,DCM萃取(2×60ml),合并有机相,水洗(3×60ml),饱和氯化钠水溶液洗涤,无水硫酸钠干燥,过滤,滤液减压浓缩,粗品经硅胶柱层析分离纯化(石油醚/乙酸乙酯20:1)即可得到中间体264-2(3.50g,9.79mmol,55.21%yield),MS m/z:358(M+1)
+.
步骤3中间体264-3的制备
将中间体264-2(1.50g,4.20mmol)溶于30ml溶于甲醇中,氮气氛下加入PtO2(50mg),常压氢化反应过夜,原料消失后,再加入10%Pd/C(50mg),常压氢化反应过夜,经硅藻土抽滤,滤液减压浓缩至干,粗品经MPLC C18反相柱纯化得到中间体264-3(800.00mg,3.82mmol,91.03%yield),MS m/z:210(M+1)
+.
步骤3中间体264-4的制备
将中间体264-3(300.00mg,1.43mmol)溶于DCM(10mL),依次加入实施例41中间体41-3(419.50mg,1.43mmol),EDCI(273.13mg,1.43mmol)、HOAT(194.48mg,1.43mmol)和DIPEA(553.41mg,4.29mmol),反应液于室温搅拌反应2小时,饱和食盐水洗涤,无水硫酸钠干燥,过滤,减压浓缩,粗品经硅胶柱层析分离纯化(二氯甲烷/甲醇20:1)即可得到中间体264-4(544mg,1.14mmol,80%yield),MS m/z:485(M+1)
+.
步骤5中间体264-5的制备
将中间体264-4(500.00mg,1.03mmol)溶于15mL甲苯中,加入TsOH(268.26mg,5.15mmol),升温至100℃并搅拌15小时,反应完毕,减压浓缩,硅胶柱层析分离纯化(二氯甲烷/甲醇30:1)即可得到中间体264-5(130.00mg,278.63umol,27.05%yield),MS m/z:467(M+1)
+.
步骤6中间体264-6的制备
将中间体264-6(100.00mg,214.33umol)溶于5ml乙醇中,加入NaOH(17.15mg,428.66umol),室温搅拌过夜,反应完毕,用1N HCl调pH值~6,乙酸乙酯萃取,减压浓缩至干,得中间体264-6的粗品(93.00mg,212.08umol,98.95%yield),未经纯化直接用于下一步。MS m/z:439(M+1)
+.
步骤7中间体264-7的制备
将中间体264-6(50.00mg,114.02umol)溶于DCM(5mL),依次加入D-亮氨酸叔丁酯 (23.49mg,125.42umol),EDCI(26.13mg,136.82umol)、HOAT(18.61mg,136.82umol)和DIPEA(73.54mg,570.10umol),反应液于室温搅拌反应2小时,饱和食盐水洗涤,无水硫酸钠干燥,过滤,减压浓缩,粗品经硅胶柱层析分离纯化(二氯甲烷/甲醇30:1)即可得到中间体264-7(40.00mg,65.81umol,57.72%yield),MS m/z:608(M+1)
+.
步骤8化合物264的制备
中间体264-7(40.00mg,65.81umol)溶于2ml二氯甲烷,冰浴下加入2mL三氟乙酸,滴毕,冰浴下搅拌3小时,反应完毕,减压浓缩,粗品经MPLC反相C18柱纯化即可得化合物264(5.10mg,8.80umol,13.37%yield),MS m/z:552(M+1)
+.
实施例265化合物265的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-7-氨基-5-氮杂螺[2.4]庚-4-酮缩合即可得到化合物265,MS m/z:604(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.60-7.55(m,1H),7.53-7.47(m,1H),7.47–7.38(m,3H),7.35–7.29(m,2H),7.25-7.19(m,1H),7.13-7.08(m,1H),6.49-6.47(m,1H),5.88(d,J=11.1Hz,1H),4.51-4.43(m,1H),4.28-4.17(m,1H),3.82(s,3H),3.11-3.05(m,1H),1.78-1.71(m,1H),1.57–1.42(m,6H),0.87–0.68(m,8H),0.52-0.42(m,2H).
实施例266化合物266的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例27中间体邻苯二胺27缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-2-环丙基-乙酰胺缩合可得到化合物266,MS m/z:622(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.54–7.30(m,8H),7.24(d,J=7.6Hz,2H),6.65(s,1H),6.48(s,1H),5.90(d,J=11.2Hz,2H),4.27(t,J=6.8Hz,1H),3.82(s,3H),1.80(s,2H),1.57-1.41(m,8H),1.24(s,3H),0.98(d,J=6.4Hz,2H),0.90–0.72(m,9H),0.39–0.30(m,4H),0.16(s,2H).
实施例267化合物267的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-2-氨基-丙酰胺缩合即可得到化合物267,MS m/z:578(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.69(d,J=8.9Hz,2H),7.55–7.46(m,1H),7.46–7.34(m,4H),7.28(td,J=7.7,1.6Hz,1H),6.61(d,J=1.8Hz,1H),6.02(d,J=9.6Hz,1H),4.26–4.20(m,1H),4.15(dd,J=9.4,5.9Hz,1H),3.86(s,3H),1.92(dd,J=13.1,6.3Hz,1H),1.53(s,6H),1.17(d,J=7.1Hz,3H),0.90(d,J=6.6Hz,3H),0.77(d,J=6.7Hz,3H).
实施例268化合物268的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-3-氨基-3-甲基-2-吡咯烷酮缩合即可得到化合物268,MS m/z:604(M+1)
+.
实施例269化合物269的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-2-氨基-N-甲基丙酰胺缩合即可得到化合物269,MS m/z:592(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.64(d,J=8.9Hz,2H),7.47(d,J=7.5Hz,1H),7.44–7.30(m,4H),7.26(dd,J=12.1,4.6Hz,1H),6.56(s,1H),5.97(d,J=9.7Hz,1H),4.25–4.20(m,1H),4.16(s,1H),3.84(s,3H),1.87(s,1H),1.51(d,J=9.5Hz,6H),1.14(d,J=7.1Hz,3H),0.86(d,J=6.5Hz,3H),0.77(d,J=6.7Hz,3H).
实施例270化合物270的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施 例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-丙酸甲酯缩合,酯水解,最后与3-氨基四氢呋喃缩合即可得到化合物270,MS m/z:648(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.68–7.60(m,2H),7.47(d,J=7.4Hz,1H),7.43–7.30(m,4H),7.29–7.22(m,1H),6.56(s,1H),5.98(d,J=9.5Hz,1H),4.26–4.21(m,1H),4.21–4.10(m,3H),3.84(s,3H),2.06–1.98(m,1H),1.90–1.83(m,1H),1.53(d,J=9.0Hz,6H),1.14(dd,J=7.1,1.4Hz,3H),0.86(d,J=6.6Hz,3H),0.78(d,J=6.8Hz,3H).
实施例271化合物271的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N,N’-二甲基丙酰胺缩合即可得到化合物271,MS m/z:606(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.66(d,J=8.4Hz,2H),7.49(d,J=7.7Hz,1H),7.45–7.32(m,4H),7.28(t,J=6.9Hz,1H),6.58(s,1H),5.98(d,J=9.7Hz,1H),4.73–4.61(m,1H),4.16(dd,J=9.9,6.0Hz,1H),3.85(s,3H),3.00–2.89(m,3H),2.78(dd,J=13.6,6.7Hz,3H),1.94–1.83(m,1H),1.57–1.44(m,6H),1.12(t,J=9.7Hz,3H),0.88(d,J=6.4Hz,3H),0.78(d,J=6.7Hz,3H).
实施例272化合物272的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例 41步骤6-7,经碱水解,与(R)-2-氨基-N-乙基丙酰胺缩合即可得到化合物272,MS m/z:606(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.68(d,J=7.9Hz,2H),7.55–7.46(m,1H),7.46–7.33(m,4H),7.32–7.24(m,1H),6.59(s,1H),6.00(d,J=9.9Hz,1H),4.27–4.12(m,2H),3.85(s,3H),3.08–2.95(m,2H),1.93–1.83(m,1H),1.51(dd,J=28.2,6.2Hz,6H),1.15(d,J=9.7Hz,3H),0.99–0.91(m,3H),0.89(d,J=10.6,4.5Hz,3H),0.78(d,J=6.7Hz,3H).
实施例273化合物273的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-异丙基丙酰胺缩合即可得到化合物273,MS m/z:620(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.77–7.64(m,2H),7.51(d,J=7.2Hz,1H),7.41(dt,J=13.7,6.9Hz,4H),7.30(dd,J=10.7,4.5Hz,1H),6.59(s,1H),6.00(d,J=9.7Hz,1H),4.23–4.11(m,2H),3.85(s,3H),1.91(d,J=6.2Hz,1H),1.62–1.46(m,6H),1.15(d,J=7.1Hz,3H),1.04–0.92(m,6H),0.90(d,J=6.6Hz,3H),0.78(d,J=6.7Hz,3H).
实施例274化合物274的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-环丙基丙酰胺缩合即可得到化合物274,MS m/z:618(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.67(s,2H),7.48(d,J=7.6Hz,1H),7.37(d,J=14.2Hz,4H),7.30–7.23(m,1H),6.57(s,1H),5.98(d,J=9.6Hz,1H),4.20–4.08(m, 2H),1.94–1.80(m,1H),1.47(dd,J=28.3,9.5Hz,6H),1.11(d,J=7.1Hz,3H),0.86(d,J=6.2Hz,3H),0.76(d,J=6.5Hz,3H),0.56(d,J=5.4Hz,2H),0.27(s,2H).
实施例275化合物275的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-环丙甲基丙酰胺缩合即可得到化合物275,MS m/z:632(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.76–7.60(m,2H),7.56–7.46(m,1H),7.46–7.32(m,4H),7.30–7.21(m,1H),6.58(d,J=1.6Hz,1H),6.00(d,J=10.0Hz,1H),4.29–4.22(m,1H),4.21–4.13(m,1H),3.85(s,3H),2.97–2.85(m,2H),1.92–1.81(m,1H),1.51(d,J=28.5,7.6Hz,6H),1.17(d,J=7.1Hz,3H),0.87(d,J=6.6Hz,3H),0.78(d,J=6.7Hz,3H),0.39–0.27(m,2H),0.12–0.02(m,2H).
实施例276化合物276的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-(4-氨基四氢吡喃)丙酰胺缩合即可得到化合物276,MS m/z:662(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.62(d,J=8.6Hz,2H),7.47(d,J=7.9Hz,1H),7.42–7.23(m,5H),6.54(s,1H),5.94(d,J=9.7Hz,1H),4.24–4.20(m,1H),4.18(s,2H),3.83(s,3H),3.33–3.24(m,2H),1.85(s,2H),1.59(s,2H),1.54(t,J=15.7Hz,6H),1.25(d,J=22.6Hz,3H),1.14(d,J=7.2Hz,3H),0.85(d,J=6.3Hz,3H),0.77 (d,J=6.8Hz,3H).
实施例277化合物277的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-3-甲基-2-羰基哌嗪缩合即可得到化合物277,MS m/z:604(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.67(d,J=9.2Hz,1H),7.82(s,1H),7.66(d,J=8.4Hz,1H),7.57–7.32(m,5H),7.24(dd,J=9.2,4.4Hz,1H),7.10(d,J=6.4Hz,1H),6.59(s,1H),5.98(t,J=9.6Hz,1H),4.71(s,1H),4.19(s,1H),3.87(s,3H),2.74(d,J=46.4Hz,4H),1.92(s,1H),1.51(d,J=26.0Hz,6H),1.31–1.16(m,3H),0.82(dd,J=34.4,6.4Hz,6H).
实施例278化合物278的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-N-甲基-3-甲基丁酰胺缩合即可得到化合物278,MS m/z:620(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.65(d,J=9.2Hz,1H),7.84(s,1H),7.64(d,J=9.2Hz,2H),7.47-7.38(m,4H),7.33–7.20(m,2H),6.70(s,2H),6.58(s,1H),5.98(s,1H),4.18(s,2H),4.11–4.04(m,1H),3.85(s,3H),1.86(dd,J=13.6,6.8Hz,3H),1.55(d,J=7.6Hz,5H),0.91–0.61(m,10H),0.01(s,1H).
实施例279化合物279的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-3-甲基丁酰胺缩合即可得到化合物279,MS m/z:606(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.64(d,J=7.6Hz,1H),7.66(d,J=10.0Hz,2H),7.49(d,J=7.2Hz,1H),7.42-7.25(m,5H),7.02(s,1H),6.70–6.55(m,2H),6.00(s,1H),4.20–4.07(m,2H),3.87(s,3H),1.90(dt,J=13.2,6.8Hz,2H),1.55(d,J=4.0Hz,5H),0.89(d,J=6.0Hz,2H),0.79(dd,J=10.4,6.8Hz,5H),0.68(d,J=6.8Hz,3H).
实施例280化合物280的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与2-氨基-2-环丁基-N-甲基-乙酰胺缩合即可得到化合物280,MS m/z:632(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.72(d,J=10.0Hz,1H),7.71(s,1H),7.57(s,4H),7.42(d,J=8.0Hz,2H),7.33(d,J=2.0Hz,2H),7.22(dd,J=14.4,7.2Hz,3H),6.70(s,2H),6.53(s,1H),5.93(s,1H),4.25(t,J=8.4Hz,3H),3.84(s,3H),2.42–2.33(m,3H),1.87–1.44(m,14H),1.26(dd,J=12.4,5.54Hz,3H),0.78(d,J=6.8Hz,5H).
实施例281化合物281的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与环丁基甘胺酰胺缩合即可得到化合物281,MS m/z:618(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.73(d,J=9.2Hz,2H),7.71–7.40(m,7H),7.31(d,J=2.0Hz,2H),7.24–7.03(m,4H),6.96(s,1H),6.51(d,J=14.8Hz,4H),5.90(s,2H),4.31–4.19(m,3H),3.83(s,3H),3.06(s,1H),2.34(s,1H),1.74(d,J=38.4Hz,6H),1.53(s,7H),1.31–1.21(m,3H),0.77(s,6H).
实施例282化合物282的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例27中间体邻苯二胺27缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与1-氨基环丁烷甲酰胺缩合即可得到化合物282,MS m/z:622(M+1)
+.
1HNMR(400MHz,MeOD):δ=7.72-7.64(m,1H),7.52-7.51(m,1H),7.42-7.34(m,3H),7.27-7.23(m,1H),6.48(s,1H),5.98(d,J=9.6Hz,1H),4.60(s,1H),4.25-4.24(m,1H),3.96(s,3H),2.62-2.55(m,2H),2.21-2.14(m,2H),1.95-1.89(m,2H),1.63(s,6H),0.99-0.97(m,3H),0.87(d,J=6.8Hz,3H).
实施例283化合物283的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例27中间体邻苯二胺27缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与1-氨基环丁烷-N-甲基甲酰胺缩合即可得到化合物283,MS m/z:636(M+1)
+.
1HNMR(400MHz,MeOD):δ=7.74-7.62(m,1H),7.52-7.51(m,1H),7.42-7.31(m,3H),7.27-7.23(m,1H),6.48(s,1H),5.98(d,J=9.2Hz,1H),4.60(s,1H),4.25-4.23(m,1H), 3.93(s,3H),2.78(s,3H),2.60-2.53(m,2H),2.21-2.13(m,2H),1.93-1.89(m,2H),1.63(s,6H),0.99-0.98(m,1H),0.87(d,J=6.8Hz,3H)
实施例284化合物284的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与1-(1H-吡唑-3-基)乙胺缩合即可得到化合物284,MS m/z:601(M+1)
+.
实施例285化合物285的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与1-(1H-咪唑)-4-乙胺缩合即可得到化合物285,MS m/z:601(M+1)
+.
实施例286化合物286的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(1R,2R)-2-氨基环丁烷-(N-甲基)-1-甲酰胺缩合缩合即可得到化 合物286,MS m/z:618(M+1)
+.
1H NMR(400MHz,MeOD)δ7.56-7.53(m,2H),7.49(d,J=8.0Hz,1H),7.36(dd,J=8.8,1.6Hz,1H),7.34–7.25(m,3H),7.21–7.15(m,1H),6.53(s,1H),5.96(d,J=8.4Hz,1H),4.06(t,J=7.6Hz,1H),3.86(s,3H),2.59(s,3H),1.98(s,1H),1.51(s,6H),1.19(s,1H),0.97(d,J=6.4Hz,2H),0.91–0.88(m,1H),0.77(t,J=6.8Hz,3H).
实施例287化合物287的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(1S,2R)-2-氨基环丁烷-(N-甲基)-1-甲酰胺缩合缩合即可得到化合物287,MS m/z:618(M+1)
+.
1H NMR(400MHz,MeOD)δ7.64-7.55(m,6H),7.40-7.24(m,6H),6.54(s,1),6.04–6.00(m,1H),4.27–4.18(m,3H),3.95(s,3H),2.68(s,2H),2.51(s,2H),2.00-1.93(m,3H),1.61(d,J=2.4Hz,6H),1.42–1.25(m,12H),1.04(d,J=7.2Hz,3H),,0.94-0.87(m,7H).
实施例288化合物288的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(1S,2R)-2-氨基环戊烷-(N-甲基)-1-甲酰胺缩合缩合即可得到化合物288,MS m/z:632(M+1)
+.
1H NMR(400MHz,MeOD)δ7.71–7.67(m,2H),7.59(d,J=7.6Hz,1H),7.48-7.38(m,4H),7.30–7.27(m,1H),6.66(s,1H),6.11(d,J=8.4Hz,1H),4.29(dd,J=14.0,6.8Hz,1H),4.20(s,1H),3.98(s,3H),3.80–3.70(m,4H),3.25(q,J=7.6Hz, 4H),2.81(d,J=8.0Hz,1H),2.68(s,1H),2.54(s,2H),2.07(s,1H),1.93–1.78(m,3H),1.68–1.51(m,9H),1.09(d,J=5.6Hz,3H),0.87(d,J=6.8Hz,3H),0.12(s,1H).
实施例289化合物289的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-(4-甲基哌嗪)丙酰胺缩合即可得到化合物289,MS m/z:661(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.67(d,J=8.8Hz,2H),7.52(d,J=2.1Hz,2H),7.41(ddd,J=13.3,9.9,8.3Hz,4H),7.30–7.24(m,2H),6.60(d,J=1.6Hz,1H),6.02(d,J=10.0Hz,1H),3.86(s,3H),2.78(s,7H),1.53(d,J=4.8Hz,6H),1.13(d,J=6.8Hz,3H),0.89–0.85(m,3H),0.77(d,J=6.7Hz,3H).
实施例290化合物290的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-四氢吡咯烷丙酰胺缩合即可得到化合物290,MS m/z:632(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.69(d,J=9.0Hz,2H),7.51(dd,J=12.5,4.8Hz,2H),7.43–7.37(m,4H),7.31–7.25(m,2H),6.03(d,J=9.5Hz,1H),3.86(s,3H),1.53(d,J=6.6Hz,6H),1.30–1.21(m,4H),1.16–1.06(m,4H),0.92–0.87(m,4H), 0.77(d,J=6.7Hz,3H).
实施例291化合物291的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-吗啉丙酰胺缩合即可得到化合物291,MS m/z:648(M+1)
+.
实施例292化合物292的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与3-氨基四氢呋喃-4-(N-环丙基)甲酰胺缩合即可得到化合物292,MS m/z:660(M+1)
+.
1H NMR(400MHz,MeOD)δ7.70(dd,J=20.0,8.4Hz,2H),7.61(d,J=13.2Hz,1H),7.54(dd,J=12.4,8.4Hz,1H),7.50–7.36(m,3H),7.34–7.27(m,1H),6.67(s,1H),6.08(d,J=7.6Hz,1H),4.56(d,J=4.4Hz,1H),4.17(s,1H),4.08(s,1H),3.98(s,2H),1.69–1.61(m,4H),1.56(s,1H),1.15–0.98(m,4H),0.87(d,J=6.8Hz,2H),0.76–0.68(m,1H),0.51–0.42(m,1H).
实施例293化合物293的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(3S,4R)-3-氨基呋喃-4-(N-环丙基)甲酰胺缩合即可得到化合物293,MS m/z:658(M+1)
+.
1H NMR(400MHz,MeOD)δ7.79–7.67(m,1H),7.58(d,J=21.2Hz,1H),7.54–7.38(m,2H),7.34–7.23(m,1H),6.64(s,1H),6.09(d,J=8.0Hz,1H),4.28(d,J=7.2Hz,1H),4.19(s,1H),4.09(s,1H),3.98(s,2H),2.51–2.35(m,1H),1.99–1.49(m,10H),1.40–1.26(m,4H),1.18–0.99(m,3H),0.88(d,J=6.8Hz,3H),0.61(d,J=6.8Hz,2H),0.37(dd,J=13.2,9.2Hz,2H).
实施例294化合物294的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与1-(2H-1,2,4-三氮唑-3-基)乙胺缩合即可得到化合物294,MS m/z:602(M+1)
+.
实施例295化合物295的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施 例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与4-氨基四氢吡喃缩合即可得到化合物295,MS m/z:591(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.64(d,J=8.4Hz,1H),7.58(s,1H),7.50(dd,J=12.2,5.3Hz,2H),7.39(dd,J=4.9,2.8Hz,2H),7.28(d,J=8.4Hz,2H),6.58(s,1H),5.97(d,J=9.9Hz,1H),3.85(s,3H),3.36–3.22(m,4H),2.07(s,6H),1.89(s,1H),1.56(d,J=13.0Hz,2H),1.51(s,6H),1.43(s,2H),1.06(t,J=7.0Hz,6H).
实施例296化合物296的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-2-异丙基-N-环丙乙酰胺缩合即可得到化合物296,MS m/z:646(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.92(s,1H),7.65–7.54(m,2H),7.47(d,J=8.5Hz,1H),7.45–7.36(m,2H),7.31(dd,J=7.1,4.9Hz,2H),7.22(t,J=7.7Hz,1H),7.13(t,J=9.3Hz,1H),6.65–6.56(m,1H),5.90(d,J=10.9Hz,1H),3.82(s,3H),1.86–1.73(m,3H),1.52(d,J=12.4Hz,7H),1.41–1.33(m,2H),1.25(dd,J=20.6,6.9Hz,5H),0.94–0.84(m,3H),0.78(d,J=6.0Hz,5H),0.74(d,J=6.8Hz,4H),0.68–0.61(m,3H),0.57(d,J=7.0Hz,2H),0.30(dd,J=11.5,5.0Hz,2H).
实施例297化合物297的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例 41步骤6-7,经碱水解,与3-(ALPHA-氨乙基)-5-甲基-4-H-1,2,4-三唑缩合即可得到化合物297,MS m/z:616(M+1)
+.
实施例298化合物298的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与D-环丁基-N-甲基甘胺酰胺缩合即可得到化合物298,MS m/z:632(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.74–7.66(m,1H),7.64–7.60(m,1H),7.59–7.55(m,1H),7.50–7.45(m,1H),7.45–7.37(m,2H),7.35–7.28(m,2H),7.25–7.19(m,1H),7.19–7.12(m,1H),6.71–6.58(m,2H),6.51–6.45(m,1H),5.94–5.85(m,1H),4.32–4.17(m,3H),4.07–4.04(m,1H),3.87–3.80(m,3H),1.86–1.58(m,8H),1.53(s,7H),1.49–1.42(m,1H),0.81–0.71(m,6H).
实施例299化合物299的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与D-环丁基-N-乙基甘胺酰胺缩合即可得到化合物299,MS m/z:646(M+1)
+.
实施例300化合物300的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-3-甲基-N-乙基丁酰胺缩合即可得到300,MS m/z:634(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.82(s,1H),7.63(s,1H),7.57(d,J=7.9Hz,1H),7.47(d,J=8.5Hz,1H),7.41(t,J=7.8Hz,2H),7.31(d,J=8.3Hz,2H),7.25–7.19(m,1H),7.14(t,J=9.0Hz,1H),6.64–6.50(m,2H),6.48(s,1H),5.89(d,J=11.2Hz,1H),4.28–4.21(m,1H),4.10–4.04(m,2H),3.83(s,3H),1.53(d,J=9.8Hz,5H),0.95(dd,J=13.7,7.0Hz,3H),0.78(d,J=5.4Hz,8H),0.69–0.63(m,3H).
实施例301化合物301的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例27中间体邻苯二胺27缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-2-氨基-3-甲基-N-甲基丁酰胺缩合即可得到301,MS m/z:638(M+1)
+.
1HNMR(400MHz,MeOD):δ=7.74-7.65(m,1H),7.51(d,J=7.2Hz,1H),7.41-7.23(m,4H),6.48(s,1H),5.98(d,J=9.6Hz,1H),4.27-4.20(m,1H),4.16(d,J=7.6Hz,1H),3.93(s,3H),2.71(s,3H),1.96-1.85(m,2H),1.63(s,5H),0.98(d,J=6.8Hz,3H),0.90-0.87(m,6H),0.82(d,J=6.4Hz,3H).
实施例302化合物302的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与4-氨基吡唑缩合即可得到化合物302,MS m/z:573(M+1)
+.
1HNMR(400MHz,MeOD):δ=7.68(s,1H),7.60-7.58(m,1H),7.54-7.49(m,2H),7.39-7.34(m,3H),7.26-7.23(m,1H),6.50(s,1H),6.01(d,J=9.6Hz,1H),5.36(t,d=4.8Hz,1H),4.21-4.19(m,1H),3.93(s,3H),2.21(t,J=7.6Hz,1H),2.06-2.03(m,4H),1.72(s,3H),1.65-1.61(m,1H),1.00-0.99(m,2H),0.94-0.90(m,2H),0.86(d,J=6.8Hz,2H)
实施例303化合物303的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例27中间体邻苯二胺27缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与R-环丁基-N-甲基甘酰胺缩合即可得到303,MS m/z:650(M+1)
+.
1HNMR(400MHz,MeOD):δ=7.73-7.64(m,1H),7.51(d,J=7.6Hz,1H),7.41-7.23(m,4H),6.48(s,1H),5.98(d,J=9.6Hz,1H),4.34(d,d=8.8Hz,1H),4.27-4.22(m,1H),3.93(s,3H),2.70(s,3H),2.52-2.49(m,1H),2.26-2.19(m,1H),1.93-1.71(m,5H),1.63(d,J=8.0Hz,6H),0.98(d,J=6.4Hz,3H),0.87(d,J=6.8Hz,3H).
实施例304化合物304的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-吗啉甘酰胺缩合即可得到化合物304,MS m/z:688(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.82(s,2H),8.34(s,1H),7.63(s,1H),7.56(d,J=7.4Hz,1H),7.47(s,1H),7.44–7.37(m,2H),7.31(d,J=8.5Hz,2H),7.23–7.10(m,4H),6.49(s,1H),5.93–5.86(m,2H),4.68(t,J=8.4Hz,2H),4.24(s,2H),3.82(s,3H),3.54–3.37(m,8H),1.84–1.57(m,9H),1.57–1.47(m,6H),0.80–0.69(m,6H).
实施例305化合物305的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(4-甲基哌嗪)甘酰胺缩合即可得到化合物305,MS m/z:701(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.82(s,2H),8.34(s,1H),7.63(s,1H),7.56(d,J=7.4Hz,1H),7.47(s,1H),7.44–7.37(m,2H),7.31(d,J=8.5Hz,2H),7.23–7.10(m,4H),6.49(s,1H),5.93–5.86(m,2H),4.68(t,J=8.4Hz,2H),4.24(s,2H),3.82(s,3H),3.54–3.37(m,8H),1.84–1.57(m,9H),1.57–1.47(m,6H),0.80–0.69(m,6H).
实施例306化合物306的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基哌啶-4-基)甘酰胺缩合即可得到化合物306,MS m/z:715(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.20(s,1H),7.62(s,2H),7.51–7.38(m,4H),7.31(s,2H),7.23(d,J=7.8Hz,1H),7.15(s,2H),6.49(s,1H),5.96–5.86(m,2H),4.32–4.21(m,3H),3.83(s,3H),2.13(s,3H),1.97–1.87(m,3H),1.85–1.56(m,10H),1.56–1.44(m,6H),0.78(d,J=4.6Hz,5H).
实施例307化合物307的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基四氢吡咯-3R-基)甘酰胺缩合即可得到化合物307,MS m/z:701(M+1)
+.
1H NMR(400MHz,MeOD)δ7.74(s,1H),7.69(d,J=8.8Hz,1H),7.59(d,J=7.2Hz,1H),7.53–7.50(m,1H),7.41(d,J=9.2Hz,2H),7.34(d,J=8.0Hz,1H),7.27(t,J=7.6Hz,1H),7.07(t,J=16.0Hz,1H),6.71(s,1H),6.12(d,J=8.0Hz,1H),4.37(s,1H),4.24-4.18(m,1H),4.14–4.10(m,1H),3.97(s,3H),2.94(s,3H),2.54(d,J=7.6Hz,2H),2.14–1.78(m,8H),1.63(d,J=7.2Hz,7H),1.28(s,1H),1.10(d,J=5.2Hz,3H),0.84(d,J=6.8Hz,3H).
实施例308化合物308的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基哌啶-3S-基)甘酰胺缩合即可得到化合物308,MS m/z:715(M+1)
+.
1HNMR(400MHz,MeOD):δ=8.43(s,1H),7.64(s,1H),7.57-7.51(m,2H),7.39-7.34(m,2H),7.31-7.23(m,2H),6.51(s,1H),6.01(d,J=9.2Hz,1H),4.32-4.19(m,2H),3.93(m,J=2.0Hz,3H),3.22-3.08(m,2H),2.67(d,J=3.6Hz,3H),2.58-2.41(m,2H),2.02-1.95(m,1H),1.94-1.81(m,5H),1.64(d,J=5.6Hz,6H),1.48-1.41(m,2H),1.01-0.98(m,3H),0.86(d,J=6.8Hz,3H)
实施例309化合物309的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基哌啶-3R-基)甘酰胺缩合即可得到化合物309,MS m/z:715(M+1)
+.
实施例310化合物310的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N-(1-甲基四氢吡咯-3S-基)丙酰胺缩合即可得到化合物310,MS m/z:661(M+1)
+.
1H NMR(400M,MeOD)δ7.77-7.82(m,1H),8.31-8.45(m,1H),7.22-7.67(m,6H),6.51(s,1H),5.92-6.06(m,1H),5.51(s,1H),4.12-4.40(m,3H),3.89-3.98(m,2H),3.38-3.57(m,2H),3.11-3.28(m,2H),2.81-2.93(m,2H),2.67-2.80(m,1H),2.32-2.51(m,2H),1.86-2.07(m,2H),1.54-1.72(m,4H),1.23-1.33(m,2H),0.94-1.04(m,2H),0.81-0.92(m,2H).
实施例311化合物311的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-3-甲基-N-(1-甲基四氢吡咯-3S-基)丁酰胺缩合即可得到化合物311,MS m/z:689(M+1)
+.
实施例312化合物312的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例 41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基四氢吡咯-3R-基)甘酰胺缩合即可得到化合物312,MS m/z:701(M+1)
+.
1H NMR(400MHz,MeOD)δ8.43(s,1H),7.64(s,1H),7.54(dd,J=17.6,8.0Hz,2H),7.39–7.34(m,3H),7.31–7.23(m,2H),6.52(s,1H),6.00(d,J=8.8Hz,1H),4.34(s,1H),4.28–4.25(m,1H),4.22-4.17(m,1H),3.94(s,3H),3.51-3.40(s,2H),3.23-3.12(m,3H),2.86(d,J=12.4Hz,3H),2.60–2.52(m,3H),2.00-1.75(m,8H),1.64(d,J=7.6Hz,8H),1.41(d,J=11.2Hz,2H),1.30(d,J=9.3Hz,7H),0.99(s,4H),0.86(d,J=6.8Hz,3H).
实施例313化合物313的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-3-甲基-N-(1-甲基四氢吡咯-3R-基)丁酰胺缩合即可得到化合物313,MS m/z:689(M+1)
+.
1H NMR(400MHz,MeOD)δ8.48(s,1H),7.64(s,1H),7.56(d,J=8.0Hz,1H),7.52(d,J=4.0Hz,1H),7.42–7.32(m,3H),7.30–7.22(m,2H),6.52(s,1H),6.01(d,J=8.0Hz,1H),4.35(s,1H),4.19(s,1H),4.14–4.07(m,1H),3.94(d,J=1.2Hz,3H),3.37(s,1H),3.15–3.02(m,2H),2.78(d,J=17.6Hz,3H),2.47–2.34(m,1H),2.02–1.86(m,3H),1.65(s,6H),1.00(d,J=5.2Hz,3H),0.94–0.82(m,6H),0.82–0.72(m,3H).
实施例314化合物314的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基--N-(1-甲基四氢吡咯-3R-基)丙酰胺缩合即可得到 化合物314,MS m/z:661(M+1)
+.
1H NMR(400M,MeOD)δ7.77-7.82(m,1H),7.69-7.75(m,1H),7.56-7.65(m,2H),7.43-7.49(m,2H),7.36-7.40(m,1H),7.26-7.34(m,1H),6.70-6.77(m,1H),6.11-6.16(m,1H),4.39-4.44(m,1H),4.23-4.32(m,1H),4.14-4.21(m,1H),4.00(s,3H),3.78-3.88(m,1H),3.58-3.67(m,1H),3.12-3.19(m,1H),2.93-3.02(m,3H),2.51-2.62(m,1H),2.31-2.43(m,1H),2.02-2.22(m,3H),1.58-1.71(m,6H),1.28-1.34(m,3H),1.09-1.18(m,3H),0.84-0.91(m,3H).
实施例315化合物315的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例29中间体邻苯二胺29缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与R-2-氨基-3-甲基-N-甲基丁酰胺缩合即可得到化合物315,MS m/z:638(M+1)
+.
1H NMR(400MHz,MeOD)δ7.54(d,J=8.0Hz,1H),7.46(d,J=1.2Hz,1H),7.43–7.37(m,4H),7.28–7.24(m,2H),7.13–7.10(m,1H),6.62(s,1H),6.07(d,J=8.4Hz,1H),4.14-4.10(m,3H),3.97(s,3H),2.71(d,J=3.2Hz,4H),2.03–1.89(m,3H),1.64(d,J=0.8Hz,6H),1.58(d,J=1.6Hz,1H),1.07(d,J=6.4Hz,3H),1.01(t,J=6.4Hz,2H),0.89–0.84(m,6H),0.80(d,J=6.8Hz,3H),0.75(d,J=6.8Hz,1H).
实施例316化合物316的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例29中间体邻苯二胺29缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-环丁基-N-甲基甘酰胺缩合即可得到化合物316,MS m/z:650(M+1)
+.
1H NMR(400MHz,MeOD)δ7.53(d,J=8.0Hz,1H),7.43(d,J=1.2Hz,1H),7.42(d,J=2.0Hz,1H),7.39(s,1H),7.37(s,1H),7.26(s,1H),7.09(dd,J=12.4,1.2Hz,1H), 6.60(s,1H),6.05(d,J=8.0Hz,1H),4.32–4.29(m,1H),4.16-4.11(m,1H),3.97(s,3H),2.70(s,3H),2.57–2.51(m,1H),2.04–1.73(m,8H),1.64(t,J=5.2Hz,7H),1.31(s,4H),1.07(d,J=6.4Hz,3H),0.86(d,J=6.8Hz,3H).
实施例317化合物317的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例28中间体邻苯二胺28缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-环丁基-N-甲基甘酰胺缩合即可得到化合物317,MS m/z:650(M+1)
+.
1H NMR(400MHz,MeOD)δ7.53(d,J=8.4Hz,1H),7.47-7.43(m,2H),7.41(d,J=2.0Hz,1H),7.38–7.32(m,2H),7.26–7.22(m,1H),6.62(s,1H),6.05(d,J=8.0Hz,1H),4.33–4.29(m,1H),4.07(t,J=7.2Hz,1H),3.95(s,3H),2.68(s,3H),2.48(dd,J=15.2,6.8Hz,1H),2.01–1.68(m,8H),1.60(d,J=14.0Hz,6H),1.07(d,J=6.4Hz,3H),0.82(d,J=6.8Hz,3H).
实施例318化合物318的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-(5-甲基-1H-吡唑-3-基)异丁胺缩合即可得到化合物318,MS m/z:643(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.65–7.61(m,1H),7.48–7.35(m,4H),7.35–7.27(m,3H),7.24–7.16(m,2H),7.15–7.08(m,2H),6.56–6.44(m,3H),5.97–5.82(m,3H),5.81–5.64(m,3H),4.78–4.50(m,5H),4.38–4.19(m,4H),3.83(s,3H),2.92–2.88(m,1H),2.15–2.04(m,4H),2.02–1.86(m,3H),1.86–1.74(m,3H),1.60–1.44(m,7H),0.83–0.75(m,5H),0.71–0.59(m,6H).
实施例319化合物319的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与1-(5-甲基-1H-吡唑-3-基)环丁甲胺缩合即可得到化合物319,MS m/z:655(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.96–8.70(m,2H),8.37–8.04(m,3H),7.67–7.50(m,2H),7.50–7.36(m,3H),7.36–7.28(m,2H),7.28–7.19(m,1H),7.19–7.08(m,1H),7.08–6.85(m,2H),6.56–6.47(m,1H),5.95–5.82(m,2H),5.72–5.65(m,1H),5.03–4.74(m,3H),4.38–4.17(m,2H),3.83(s,3H),2.13–2.03(m,3H),1.81–1.59(m,6H),1.56–1.40(m,6H),1.03(d,J=6.5Hz,11H),0.78–0.69(m,4H).
实施例320化合物320的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与N-甲基-(1-四氢呋喃-3R-基)乙酰胺缩合即可得到化合物320,MS m/z:648(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.83(s,1H),7.68(s,1H),7.63–7.54(m,1H),7.47(d,J=8.5Hz,1H),7.41(t,J=7.3Hz,2H),7.31(dd,J=7.1,5.4Hz,2H),7.22(t,J=6.9Hz,1H),7.17–6.93(m,3H),6.47(s,1H),5.88(d,J=11.1Hz,1H),4.28–4.16(m,2H),3.81(s,3H),3.58(dd,J=10.7,5.2Hz,2H),3.35(d,J=7.3Hz,1H),1.75(s,3H),1.52(d,J=8.1Hz,6H),0.77(d,J=6.3Hz,6H).
实施例321化合物321的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基-1-环丙醇)甘酰胺缩合即可得到化合物321,MS m/z:688(M+1)
+.
实施例322化合物322的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基-1-甲氧环丙烷)甘酰胺缩合即可得到化合物322,MS m/z:702(M+1)
+.
实施例323化合物323的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,再与(R)-N-甲基-(1-四氢呋喃-3S-基)乙酰胺缩合即可得到化合物323,MS m/z:648(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ7.83(s,1H),7.68(s,1H),7.63 –7.54(m,1H),7.47(d,J=8.5Hz,1H),7.41(t,J=7.3Hz,2H),7.31(dd,J=7.1,5.4Hz,2H),7.22(t,J=6.9Hz,1H),7.17–6.93(m,3H),6.47(s,1H),5.88(d,J=11.1Hz,1H),4.28–4.16(m,2H),3.81(s,3H),3.58(dd,J=10.7,5.2Hz,2H),3.35(d,J=7.3Hz,1H),1.75(s,3H),1.52(d,J=8.1Hz,6H),0.77(d,J=6.3Hz,6H).
实施例324化合物324的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(1-甲基-1-F环丙烷)甘酰胺缩合即可得到化合物324,MS m/z:690(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.89–8.67(m,3H),8.23(s,1H),8.12(s,2H),7.63(s,1H),7.57(d,J=8.5Hz,1H),7.54–7.37(m,4H),7.31(t,J=7.2Hz,3H),7.27–7.18(m,2H),7.18–7.07(m,2H),6.70(dd,J=22.6,8.2Hz,2H),6.49(s,2H),5.96–5.85(m,2H),4.33(t,J=8.6Hz,1H),4.26(d,J=7.6Hz,2H),3.83(s,3H),1.85–1.59(m,8H),1.52(s,7H),1.24(s,1H),0.89(d,J=19.0Hz,3H),0.78(d,J=6.5Hz,6H),0.62(d,J=8.7Hz,3H).
实施例325化合物325的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-(2-氟乙基)甘酰胺缩合即可得到化合物325,MS m/z:664(M+1)
+.
1H NMR(400MHz,MeOD)δ8.25(s,1H),7.64(s,1H),7.58(s,1H),7.56–7.49(m,1H),7.45–7.29(m,4H),7.25(dd,J=10.7,4.5Hz,1H),6.74(d,J=8.0Hz, 1H),6.52(s,2H),6.01(d,J=9.2Hz,1H),4.50–4.42(m,1H),4.41–4.30(m,2H),4.19(s,1H),3.94(s,3H),3.58(s,1H),3.50(s,1H),3.47–3.41(m,1H),2.64–2.41(m,3H),2.04–1.76(m,7H),1.76–1.59(m,7H),1.53–1.37(m,3H),1.31(s,1H),1.00(d,J=6.3Hz,4H),0.88(t,J=13.7Hz,4H).
实施例326化合物326的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例23中间体邻苯二胺23缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-2-氨基-N,N’-二甲基丙酰胺缩合即可得到化合物326,MS m/z:592(M+1)
+.
实施例327化合物327的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例23中间体邻苯二胺23缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,与(R)-环丁基-N-甲基甘酰胺缩合即可得到化合物327,MS m/z:618(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ8.95(s,1H),8.42(s,1H),8.06–7.76(m,2H),7.65–7.36(m,4H),7.36–7.26(m,2H),7.24-7.19(m,2H),6.54(s,1H),5.90(t,J=7.2Hz,1H),4.35–4.14(m,2H),3.83(d,J=3.2Hz,3H),2.57(d,J=4.4Hz,1H),2.48(d,J=4.4Hz,2H),1.97–1.43(m,8H),1.38-1.35(m,3H),0.77-0.74(d,J=6.1Hz,6H).
实施例328化合物328的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc保护基,与1,6-二氢-1-甲基-6-氧代-2-吡啶羧酸缩合,再参考实施例41步骤6-7,经碱水解,与1-氨基-1-环丁甲酰胺缩合即可得到化合物328,MS m/z:631(M+1)
+.
1H NMR(400MHz,DMSO-d
6)δ9.42(d,J=8.8Hz,1H),7.64(d,J=29.6Hz,3H),7.48(d,,J=8.0Hz,2H),7.40(t,J=7.2Hz,1H),7.28(dd,J=9.2,7.2Hz,3H),6.83(s,1H),6.55(s,1H),6.38(dd,J=9.2,1.0Hz,1H),5.95-5.85(m,1H),5.73(d,J=6.4Hz,1H),4.22(dd,J=11.2,4.4Hz,1H),2.85(s,3H),2.06(d,J=14.4Hz,3H),1.76(d,J=8.0Hz,3H),1.54(s,6H),0.79(t,J=6.4Hz,5H).
实施例329化合物329的制备
参照实施例74方法,以实施例18中间体18的单一手性异构体18-c为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与D-2-氨基-4-甲基戊酰胺缩合即可得到化合物329,MS m/z:700(M+1)
+.
实施例330化合物330的制备
参照实施例74方法,以实施例2中间体2的单一手性异构体2-c为原料经过与实施例146中间体邻苯二胺146-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,碱水解,最后与甲胺盐酸盐缩合可得到化合物330,MS m/z:493(M+1)
+.
实施例331化合物331的制备
参照实施例74方法,以实施例5中间体5的单一手性异构体5-a为原料经过与实施例30中间体邻苯二胺30缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基可得到化合物331,MS m/z:494(M+1)
+.
实施例332化合物332的制备
参照实施例57步骤1-6方法,中间体15与实施例26中间体26缩合,关咪唑环,脱Boc,引入1-乙基-1H-吡唑-5-酰基,乙酯水解,最后与甲胺缩合即可得到化合物332,MS m/z:465[M+1]
+。
实施例333化合物333的制备
参照实施例74方法,以实施例171步骤1中间体171-1(未拆分的四个异构体混合物)为原料经过与实施例26中间体邻苯二胺26缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与R-2-氨基-丙酰胺缩合即可得到化合物333,MS m/z:598(M+1)
+.
实施例334化合物334的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例34的中间体34-2缩合,关咪唑环,乙酯水解,与D-亮氨酸叔丁酯盐酸盐缩合,最后叔丁酯经三氟乙酸水解即可得到化合物334,MS m/z:549[M+1]
+.
实施例335化合物335的制备
参照实施例41步骤4-7,依次经过中间体41-3与实施例36的中间体36缩合,关咪唑环即可得到化合物335,MS m/z:461[M+1]
+.
实施例336中间体336的制备
步骤1中间体336-1的制备
参照实施例38步骤3方法,由中间体38-1与2-(4-溴-1,3-二甲基-1H-吡唑-5-基)乙酸甲酯偶联即可得到中间体336-1,MS m/z:275[M+1]
+
步骤2化合物336的制备
参照实施例74方法,以实施例2中间体2的(对映异构体2-c和2-d的混合物)为原料经过与中间体邻苯二胺336-1缩合,关环,脱Boc,上1-甲基-1H-吡唑-5-酰基,再参考实施例41步骤6-7,经碱水解,最后与环戊胺缩合即可得到化合物336,MS m/z:641(M+1)
+。
为了说明本发明的有益效果,本发明提供以下试验例。
试验例1IL-17酶联免疫吸附测定(ELISA)实验
通过竞争性ELISA对IL-17a抑制剂对受体-配体结合的抑制情况进行了定量检测。将0.2μg/ml IL-17a(Sino Biological lnc.Cat#12047-H07B)以100μl每孔在96孔板中37度孵育30分钟。用PBST(PBS,0.05%Tween-20)洗板4次,每次200μl每孔,加入200μl 5%脱脂牛奶于25度摇床上孵育30分钟。准备100X浓度待测化合物,浓度从0.003μM到30μM。用PBST(PBS,0.05%Tween-20)洗板4次后加入89μl PBST和1μl 100X浓度待测化合物混匀后于25度预孵育10分钟。加入10μl 16nM IL-17R于25度摇床上孵育30分钟。洗板4次后,加入100μl抗Fc标签HRP偶联抗体于25度摇床上孵育30分钟。洗板4次后,加入100μl TMB底物溶液25度避光孵育。加入20%HCl后,采用酶标仪于450nm波长检测光吸收值。
按照上述方法对实施例制备的化合物进行去IL-17A抑制活性检测,试验结果见表1,其中测定各化合物的IC
50按照说明分类,表1中:
“+”表示IC
50测定值大于1uM并小于100μM;
“++”表示IC
50测定值小于1uM大于250nM;
“+++”表示IC
50测定值小于250nM.
表1、化合物对IL-17A的抑制活性
| 实施例化合物 | IL-17A抑制活性 | 实施例化合物 | IL-17A抑制活性 |
| 41 | + | 188 | +++ |
| 42 | + | 189 | + |
| 43 | + | 190 | + |
| 44 | + | 191 | ++ |
| 45 | + | 192 | + |
| 46 | + | 193 | + |
| 47 | + | 194 | + |
| 48 | + | 195 | ++ |
| 49 | + | 196 | + |
| 50 | + | 197 | + |
| 51 | + | 198 | + |
| 52 | + | 199 | ++ |
| 53 | + | 200 | ++ |
| 54 | + | 201 | ++ |
| 55 | + | 202 | + |
| 56 | + | 203 | ++ |
| 57 | ++ | 204 | + |
| 58 | +++ | 205 | ++ |
| 59 | ++ | 206 | + |
| 60 | + | 207 | + |
| 61 | ++ | 208 | + |
| 62 | + | 209 | + |
| 63 | + | 210 | + |
| 64 | + | 211 | + |
| 65 | + | 212 | + |
| 66 | + | 213 | + |
| 67 | + | 214 | + |
| 68 | + | 215 | ++ |
| 69 | ++ | 216 | + |
| 70 | + | 217 | + |
| 71 | ++ | 218 | + |
| 72 | +++ | 219 | + |
| 73 | +++ | 220 | + |
| 74 | +++ | 221 | + |
| 75 | + | 222 | ++ |
| 76 | ++ | 223 | ++ |
| 77 | ++ | 224 | ++ |
| 78 | +++ | 225 | +++ |
| 79 | +++ | 226 | ++ |
| 80 | +++ | 227 | ++ |
| 81 | +++ | 228 | ++ |
| 82 | + | 229 | +++ |
| 83 | ++ | 230 | + |
| 84 | ++ | 231 | ++ |
| 85 | + | 232 | + |
| 86 | + | 233 | +++ |
| 87 | + | 234 | +++ |
| 88 | + | 235 | +++ |
| 89 | + | 236 | ++ |
| 90 | + | 237 | +++ |
| 91 | + | 238 | ++ |
| 92 | + | 239 | + |
| 93 | + | 240 | ++ |
| 94 | ++ | 241 | + |
| 95 | +++ | 242 | ++ |
| 96 | + | 243 | ++ |
| 97 | + | 244 | +++ |
| 98 | + | 245 | +++ |
| 99 | + | 246 | ++ |
| 100 | + | 247 | +++ |
| 101 | + | 248 | ++ |
| 102 | + | 249 | ++ |
| 103 | + | 250 | + |
| 104 | + | 251 | ++ |
| 105 | + | 252 | ++ |
| 106 | ++ | 253 | +++ |
| 107 | + | 254 | +++ |
| 108 | + | 255 | +++ |
| 109 | + | 256 | ++ |
| 110 | + | 257 | +++ |
| 111 | + | 258 | +++ |
| 112 | + | 259 | + |
| 113 | + | 260 | + |
| 114 | + | 261 | + |
| 115 | + | 262 | ++ |
| 116 | + | 263 | +++ |
| 117 | + | 264 | + |
| 118 | + | 265 | +++ |
| 119 | + | 266 | +++ |
| 120 | + | 267 | +++ |
| 121 | + | 268 | +++ |
| 122 | + | 269 | +++ |
| 123 | + | 270 | +++ |
| 124 | + | 271 | +++ |
| 125 | + | 272 | +++ |
| 126 | + | 273 | +++ |
| 127 | + | 274 | +++ |
| 128 | ++ | 275 | +++ |
| 129 | + | 276 | +++ |
| 130 | + | 277 | ++ |
| 131 | + | 278 | +++ |
| 132 | ++ | 279 | +++ |
| 133 | ++ | 280 | +++ |
| 134 | + | 281 | +++ |
| 135 | + | 282 | +++ |
| 136 | ++ | 283 | + |
| 137 | + | 284 | +++ |
| 138 | + | 285 | +++ |
| 139 | ++ | 286 | +++ |
| 140 | + | 287 | +++ |
| 141 | ++ | 288 | +++ |
| 142 | + | 289 | +++ |
| 143 | + | 290 | +++ |
| 144 | + | 291 | +++ |
| 145 | + | 292 | +++ |
| 146 | + | 293 | ++ |
| 147 | + | 294 | + |
| 148 | ++ | 295 | ++ |
| 149 | ++ | 296 | +++ |
| 150 | + | 297 | +++ |
| 151 | + | 298 | +++ |
| 152 | + | 299 | +++ |
| 153 | + | 300 | +++ |
| 154 | + | 301 | +++ |
| 155 | + | 302 | ++ |
| 156 | + | 303 | +++ |
| 157 | + | 304 | +++ |
| 158 | + | 305 | +++ |
| 159 | + | 306 | +++ |
| 160 | + | 307 | +++ |
| 161 | + | 308 | +++ |
| 162 | ++ | 309 | ++ |
| 163 | ++ | 310 | ++ |
| 164 | + | 311 | ++ |
| 165 | ++ | 312 | ++ |
| 166 | + | 313 | ++ |
| 167 | + | 314 | ++ |
| 168 | ++ | 315 | + |
| 169 | + | 316 | +++ |
| 170 | ++ | 317 | +++ |
| 171 | + | 318 | ++ |
| 172 | + | 319 | ++ |
| 173 | + | 320 | +++ |
| 174 | + | 321 | +++ |
| 175 | + | 322 | +++ |
| 176 | + | 323 | +++ |
| 177 | ++ | 324 | +++ |
| 178 | + | 325 | +++ |
| 179 | ++ | 326 | ++ |
| 180 | ++ | 327 | +++ |
| 181 | + | 328 | + |
| 182 | ++ | 329 | + |
| 183 | ++ | 330 | + |
| 184 | + | 331 | + |
| 185 | ++ | 332 | + |
| 186 | ++ | 333 | +++ |
| 187 | ++ | 334 | + |
| 335 | + | 336 | + |
试验表明,本发明实施例的化合物具有良好的IL-17A抑制活性,可以有效用于与IL-17A活性异常疾病的治疗。
综上所述,本发明公开的式I所示的新化合物,表现出了良好的IL-17A抑制活性,为临床治疗与IL-17A活性异常相关的疾病提供了一种新的药用可能。
Claims (23)
- 式I所示的化合物、或其立体异构体、或其药学上可接受的盐:其中,X选自O、S、NR 1’;R 1、R 2分别独自选自氢、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环、-S(O) 2R 6、-S(O)R 6、-S(O) 2NR 6R 7、-S(O)NR 6R 7、-C(O)R 6、-C(O)OR 6、-C(O)NR 6R 7、-P(O)(OR 7)R 6、-P(O)R 6R 7、-P(O)R 6(NR 6R 7)、-P(O)(NR 6R 7)(NR 6R 7)、-P(O)(OR 7)(NR 6R 7)、P(O)(OR 7)(OR 7);其中烷基、烯基、炔基、环烷基、杂环烷基、芳环、芳杂环被m个R c取代;R 6、R 7分别独自选自氢、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环;其中烷基、烯基、炔基、环烷基、杂环烷基、芳环、芳杂环被m个R c取代;R 3、R 3’分别独自选自氢、-OR d、-SR d、-NR dR d、卤素、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环;其中R d、烷基、烯基、炔基、环烷基、杂环烷基、芳环被m个R c取代;或者,R 3和R 3’连接成3~10元环烷基、3~10元杂环烷基;R d选自氢、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;B环选自3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;其中环烷基、杂环烷基、芳环、芳杂环被m个R c取代;R 1’选自氢、C 1~10烷基;r为0、1、2、3;R 8和R 9分别独自选自氢、卤素、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元的环烷基、3~10元的杂环烷基;其中烷基、烯基、炔基、环烷基、杂环烷基被m个R c取代;或者,R 8和R 9相连形成3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;其中环烷基、杂环烷基、芳环、杂芳环被m个R c取代;L 2选自-C(O)NR 10-、-NR 10C(O)-、-C(O)-、-C(O)O-、-S(O) 2NR 10-、-S(O)NR 10-、-NR 10S(O) 2-、-NR 10S(O)-、-P(O)NR 10-、-NR 10P(O)-或无;R 10选自氢、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元环烷基、3~10元杂环烷基,其中烷基、烯基、炔基、环烷基、杂环烷基被m个R c取代;R 4和R 5分别独自选自氢、卤素、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环、-CN、-NO 2、-OR a、-OC(O)R a、-OC(O)NR aR b、-OS(O) 2R a、-SR a、-S(O) 2R a、-S(O) 2NR aR b、-OS(O) 2NR aR b、-C(O)R a、-C(O)OR a、-C(O)NR aR b、-NR aR b、-NR aC(O)R b、-NR aC(O)OR b、-NR aC(O)NR aR b、-NR aS(O) 2R b、-NR aS(O) 2NR aR b;其中R a、R b、烷基、烯基、炔基、环烷基、杂环烷基、芳环、芳杂环被m个R c取代;或者,R 4和R 5相连形成3~10元环烷基、3~10元杂环烷基;其中环烷基、杂环烷基被m个R c取代;X 1选自CR 11或N;X 2选自NR 12、O、S或-(CR 13=CR 14)-;X 4选自N或CH;R 11选自氢、卤素、-CN、-NO 2、C 1~10烷基、C 2~10烯基、C 2~10炔基、C 1~10卤素烷基、3~10元的环烷基、3~10元的杂环烷基、-OR a、-NR aR b;R 12选自氢、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元的环烷基、3~10元的杂环烷基、-S(O) 2R a、-S(O) 2NR aR b、-C(O)R a、-C(O)OR a、-C(O)NR aR b;R 13、R 14分别独自选自氢、卤素、-CN、-NO 2、C 1~10烷基、C 2~10烯基、C 2~10炔基、C 1~10卤素烷基、3~10元的环烷基、3~10元的杂环烷基、-OR a、-NR aR b;其中烷基、烯基、炔基、环烷基、杂环烷基被m个R c取代;m为0、1、2、3、4;R a、R b分别独自选自氢、C 1~10烷基、C 2~10烯基、C 2~10炔基、-S(O) 2R d、-S(O)R d、-P(O) 2R d、-P(O)R d、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;其中,烷基、烯基、炔基、环烷基、杂环烷基、芳环、杂芳环被m个R c取代;R c分别独自选自C 1~10烷基、=S、=O、卤素、-CN、-NO 2、-OR a、-OC(O)R a、-OS(O) 2R a、-SR a、-S(O) 2R a、-S(O) 2NR aR b、-C(O)R a、-C(O)OR a、-C(O)NR aR b、-NR aR b、-NR aC(O)R b、-NR aS(O) 2R b、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环,其中,烷基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元芳杂环被0~3个C 1~10烷基、卤素、-NR aR b、-OR d、-C(O)NR aR b、3~10元杂环烷基、5~10元芳环、5~10元芳杂环取代。
- 根据权利要求1所述的化合物、或其立体异构体、或其药学上可接受的盐,其特征在于:R 1、R 2分别独自选自氢、C 1~6烷基、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环、-S(O) 2R 6、-S(O)R 6、-S(O) 2NR 6R 7、-C(O)R 6、-C(O)OR 6、-C(O)NR 6R 7;其中烷基、环烷基、杂环烷基、芳环、芳杂环被m个R c取代;R 6、R 7分别独自选自氢、C 1~6烷基、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环;其中烷基、环烷基、杂环烷基、芳环、芳杂环被m个R c取代;R 3、R 3’分别独自选自氢、-OR d、卤素、C 1~6烷基、3~10元环烷基、3~10元杂环烷基、5~10元芳环;其中烷基、环烷基、杂环烷基、芳环被m个R c取代;或者,R 3和R 3’连接成3~10元环烷基;R d选自氢、C 1~10烷基、C 2~10烯基、C 2~10炔基、3~10元环烷基、3~10元杂环烷基、5~10元芳环、5~10元杂芳环;R 8和R 9分别独自选自氢、卤素、C 1~6烷基、3~10元的环烷基、3~10元的杂环烷基;其中烷基,环烷基、杂环烷基被m个R c取代;或者,R 8和R 9相连形成3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元杂芳环;其中环烷基、杂环烷基、芳环、杂芳环可进一步被m个R c取代;R 10选自氢、C 1~6烷基、3~10元环烷基、3~10元杂环烷基,其中烷基、环烷基、杂环烷基被m个R c取代;;R 4和R 5分别独自选自氢、卤素、C 1~6烷基、3~10元的环烷基、3~10元的杂环烷基、6~10元芳环、5~10元芳杂环、-CN、-NO 2、-OR a、-OC(O)R a、-C(O)R a、-C(O)OR a、-C(O)NR aR b、-NR aR b、-NR aC(O)R b、-NR aC(O)NR aR b、-NR aS(O) 2R b、-NR aS(O) 2NR aR b、-S(O) 2R a、-S(O) 2NR aR b;其中烷基、环烷基、杂环烷基、芳环、芳杂环被m个R c取代;或者,R 4和R 5相连形成3~10元的环烷基、3~10元的杂环烷基;其中环烷基、杂环烷基被m个R c取代;R 11选自氢、卤素、C 1~6烷基;R 12选自氢、C 1~6烷基;R 13、R 14分别独自选自氢、卤素、C 1~6烷基、C 2~6烯基;其中烷基、烯基被m个R c取代;m为0、1、2、3;R a、R b分别独自选自氢、C 1~6烷基、C 2~6烯基、C 2~6炔基、-S(O) 2R d、-S(O)R d、-P(O) 2R d、-P(O)R d3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元杂芳环;其中,烷基、烯基、炔基、环烷基、杂环烷基、芳环、杂芳环被m个R c取代。
- 根据权利要求2所述的化合物、或其立体异构体、或其药学上可接受的盐,其特征在于:R 1、R 2分别独自选自氢、-S(O) 2R 6、-S(O)R 6、-S(O) 2NR 6R 7、-C(O)R 6、-C(O)OR 6、-C(O)NR 6R 7;R 6、R 7分别独自选自氢、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环;其中环烷基、杂环烷基、芳环、芳杂环被m个R c取代;R 3、R 3’分别独自选自氢、-OR d、C 1~6烷基、3~10元环烷基、3~10元杂环烷基、5~10元芳环;其中烷基、环烷基、杂环烷基、芳环被m个R c取代;或者,R 3和R 3’连接成3~8元环烷基;R 8和R 9分别独自选自氢、C 1~6烷基、3~10元的环烷基、3~10元的杂环烷基;或者,R 8和R 9相连形成3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元杂芳环;R 4和R 5分别独自选自氢、C 1~6烷基、3~10元环烷基、3~10元杂环烷基、6~10元芳环、5~10元芳杂环、-OR a、-C(O)R a、-C(O)OR a、-C(O)NR aR b、-NR aR b、-NR aC(O)R b、-NR aC(O)NR aR b、-NR aS(O) 2R b、-NR aS(O) 2NR aR b、-S(O) 2R a、-S(O) 2NR aR b、;其中烷基环烷基、杂环烷基、芳环、芳杂环可进一步被m个R c取代;或者,R 4和R 5相连形成3~10元环烷基、3~10元杂环烷基;其中环烷基、杂环烷基可进一步被m个R c取代;R 11选自氢、C 1~6烷基;R 12选自氢、C 1~6烷基;R 13、R 14分别独自选自氢、C 1~6烷基、C 2~6烯基;其中烷基、烯基被m个R c取代;m为0、1、2。
- 根据权利要求6所述的化合物、或其立体异构体、或其药学上可接受的盐,其特征在于:R 5’为卤素、CN;n选自0、1、2;L 2为-C(O)NR 10-、-NR 10C(O)-、-C(O)-、-C(O)O-或无。
- 根据权利要求6所述的化合物、或其立体异构体、或其药学上可接受的盐,其特征在于:R 5’为卤素和5~10元杂芳环,其中,5~10元芳杂环可进一步被0~3个C 1~10烷基取代、3~10元杂环烷基、5~10元芳环、5~10元芳杂环取代;n选自0、1、2、3;L 2为-C(O)NR 10-、-NR 10C(O)-、-C(O)-、-C(O)O-或无。
- 根据权利要求6所述的化合物、或其立体异构体、或其药学上可接受的盐,其特征在于:R 5’为卤素和-OR a;n选自0、1、2、3;L 2为无。
- 权利要求1-20任一项所述的化合物、或其立体异构体、或其药学上可接受的盐在制备治疗IL-17A介导的疾病的药物中的用途。
- 根据权利要求21所述的用途,其特征在于:所述IL-17A介导的疾病是与炎症、自身免疫性疾病、感染性疾病、癌症、癌前期综合征相关的疾病中的一种或几种。
- 一种药物,其特征在于:它是以权利要求1~20任一项所述的化合物、或其立体异构体、或其药学上可接受的盐,加上药学上可接受的辅料制备而成的制剂。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810493931.8 | 2018-05-22 | ||
| CN201810493931 | 2018-05-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019223718A1 true WO2019223718A1 (zh) | 2019-11-28 |
Family
ID=68615925
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2019/087966 Ceased WO2019223718A1 (zh) | 2018-05-22 | 2019-05-22 | 一种免疫调节剂 |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN110511213B (zh) |
| WO (1) | WO2019223718A1 (zh) |
Cited By (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020127685A1 (en) | 2018-12-19 | 2020-06-25 | Leo Pharma A/S | Amino-acid anilides as small molecule modulators of il-17 |
| WO2020182666A1 (en) | 2019-03-08 | 2020-09-17 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021098844A1 (zh) * | 2019-11-20 | 2021-05-27 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| WO2021170627A1 (en) | 2020-02-25 | 2021-09-02 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| WO2021170631A1 (en) | 2020-02-25 | 2021-09-02 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| WO2021204800A1 (en) | 2020-04-07 | 2021-10-14 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| WO2021204801A1 (en) | 2020-04-07 | 2021-10-14 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| WO2021250194A1 (en) | 2020-06-12 | 2021-12-16 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021255174A1 (en) | 2020-06-18 | 2021-12-23 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021255086A1 (en) | 2020-06-18 | 2021-12-23 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021255085A1 (en) | 2020-06-18 | 2021-12-23 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2022007462A1 (zh) * | 2020-07-04 | 2022-01-13 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| CN113943278A (zh) * | 2020-07-16 | 2022-01-18 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| EP3943495A1 (en) | 2020-07-24 | 2022-01-26 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2022096411A1 (en) | 2020-11-09 | 2022-05-12 | UCB Biopharma SRL | Dicyclopropylmethyl derivatives as il-17 modulators |
| WO2022096412A1 (en) | 2020-11-09 | 2022-05-12 | UCB Biopharma SRL | Dicyclopropylmethyl derivatives as il-17 modulators |
| WO2022128584A1 (en) | 2020-12-14 | 2022-06-23 | UCB Biopharma SRL | Imidazopyridazine derivatives as il-17 modulators |
| WO2023275301A1 (en) | 2021-07-01 | 2023-01-05 | UCB Biopharma SRL | Imidazotriazine derivatives as il-17 modulators |
| WO2023283453A1 (en) | 2021-07-09 | 2023-01-12 | Dice Alpha, Inc. | Phenyl acetamide based il-17a modulators and uses thereof |
| KR20230021083A (ko) * | 2020-07-04 | 2023-02-13 | 히트젠 주식회사 | 면역 조절제 |
| WO2023025783A1 (en) | 2021-08-23 | 2023-03-02 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2023049888A1 (en) * | 2021-09-27 | 2023-03-30 | Janssen Pharmaceutica Nv | Benzimidazoles as modulators of il-17 |
| JP2023523770A (ja) * | 2020-04-30 | 2023-06-07 | ヤンセン ファーマシューティカ エヌ.ベー. | Il-17のモジュレーターとしてのイミダゾピリミジン |
| WO2023111181A1 (en) | 2021-12-16 | 2023-06-22 | Leo Pharma A/S | Small molecule modulators of il-17 |
| US11691979B2 (en) | 2020-04-30 | 2023-07-04 | Janssen Pharmaceutica Nv | Imidazopyridazines as modulators of IL-17 |
| WO2023166172A1 (en) | 2022-03-04 | 2023-09-07 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2024017880A1 (en) | 2022-07-22 | 2024-01-25 | UCB Biopharma SRL | Imidazotriazine derivatives as il-17 modulators |
| WO2024115662A1 (en) | 2022-12-02 | 2024-06-06 | Leo Pharma A/S | Small molecule modulators of il-17 |
| US12252491B2 (en) | 2019-01-07 | 2025-03-18 | Eli Lilly And Company | IL-17A inhibitors |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021027724A1 (zh) * | 2019-08-09 | 2021-02-18 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| CN112341446B (zh) * | 2019-08-09 | 2022-06-17 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| CN112341440B (zh) * | 2019-08-09 | 2022-05-31 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| CN112341429B (zh) * | 2019-08-09 | 2021-11-23 | 成都先导药物开发股份有限公司 | 一种免疫调节剂的中间体化合物 |
| WO2021027721A1 (zh) * | 2019-08-09 | 2021-02-18 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| CN112824399B (zh) * | 2019-11-20 | 2022-04-12 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| CN113683598B (zh) * | 2020-05-18 | 2022-10-14 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| WO2023078319A1 (zh) * | 2021-11-04 | 2023-05-11 | 海思科医药集团股份有限公司 | 一种可抑制il-17a的杂环化合物及其用途 |
| WO2023202664A1 (en) * | 2022-04-21 | 2023-10-26 | Beigene, Ltd. | Small molecule il-17a modulators |
| CN116425648B (zh) * | 2023-02-27 | 2025-12-05 | 南京望知星医药科技有限公司 | 一种吲哚布芬杂质的合成方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005035551A2 (en) * | 2003-10-08 | 2005-04-21 | Incyte Corporation | Inhibitors of proteins that bind phosphorylated molecules |
| CN1860103A (zh) * | 2003-09-22 | 2006-11-08 | S*Bio私人有限公司 | 苯并咪唑衍生物:制备方法及医药应用 |
| CN103476771A (zh) * | 2011-01-28 | 2013-12-25 | 4Sc探索有限责任公司 | 用于治疗自身免疫炎症的IL17和IFN-γ抑制 |
| CN104271565A (zh) * | 2012-04-13 | 2015-01-07 | 田边三菱制药株式会社 | 酰胺吡啶衍生物及其用途 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2337599A (en) * | 1998-01-23 | 1999-08-09 | Microcide Pharmaceuticals, Inc. | Efflux pump inhibitors |
| CN101006063B (zh) * | 2004-06-15 | 2013-07-17 | 布里斯托尔-迈尔斯斯奎布公司 | 用作丝氨酸蛋白酶抑制剂的五元杂环类化合物 |
| CN104069102A (zh) * | 2013-03-27 | 2014-10-01 | 郑荣远 | 2-(2-苯并呋喃基)-2-咪唑啉在制备调节白介素-17的药物中应用 |
-
2019
- 2019-05-22 WO PCT/CN2019/087966 patent/WO2019223718A1/zh not_active Ceased
- 2019-05-22 CN CN201910429679.9A patent/CN110511213B/zh active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1860103A (zh) * | 2003-09-22 | 2006-11-08 | S*Bio私人有限公司 | 苯并咪唑衍生物:制备方法及医药应用 |
| WO2005035551A2 (en) * | 2003-10-08 | 2005-04-21 | Incyte Corporation | Inhibitors of proteins that bind phosphorylated molecules |
| CN103476771A (zh) * | 2011-01-28 | 2013-12-25 | 4Sc探索有限责任公司 | 用于治疗自身免疫炎症的IL17和IFN-γ抑制 |
| CN104271565A (zh) * | 2012-04-13 | 2015-01-07 | 田边三菱制药株式会社 | 酰胺吡啶衍生物及其用途 |
Non-Patent Citations (1)
| Title |
|---|
| DATABASE REGISTRY 25 February 2014 (2014-02-25), retrieved from STN Database accession no. 1555879-28-1 * |
Cited By (41)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11377425B1 (en) | 2018-12-19 | 2022-07-05 | Leo Pharma A/S | Small molecule modulators of IL-17 |
| WO2020127685A1 (en) | 2018-12-19 | 2020-06-25 | Leo Pharma A/S | Amino-acid anilides as small molecule modulators of il-17 |
| US12252491B2 (en) | 2019-01-07 | 2025-03-18 | Eli Lilly And Company | IL-17A inhibitors |
| WO2020182666A1 (en) | 2019-03-08 | 2020-09-17 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021098844A1 (zh) * | 2019-11-20 | 2021-05-27 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| WO2021170627A1 (en) | 2020-02-25 | 2021-09-02 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| WO2021170631A1 (en) | 2020-02-25 | 2021-09-02 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| JP7682920B2 (ja) | 2020-04-07 | 2025-05-26 | ユーシービー バイオファルマ エスアールエル | Il-17モジュレータとしてのジフルオロシクロヘキシル誘導体 |
| WO2021204800A1 (en) | 2020-04-07 | 2021-10-14 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| WO2021204801A1 (en) | 2020-04-07 | 2021-10-14 | UCB Biopharma SRL | Difluorocyclohexyl derivatives as il-17 modulators |
| JP2023521107A (ja) * | 2020-04-07 | 2023-05-23 | ユーシービー バイオファルマ エスアールエル | Il-17モジュレータとしてのジフルオロシクロヘキシル誘導体 |
| US11702422B2 (en) | 2020-04-30 | 2023-07-18 | Janssen Pharmaceutica Nv | Imidazopyridazines as modulators of IL-17 |
| JP7661361B2 (ja) | 2020-04-30 | 2025-04-14 | ヤンセン ファーマシューティカ エヌ.ベー. | Il-17のモジュレーターとしてのイミダゾピリミジン |
| US12024524B2 (en) | 2020-04-30 | 2024-07-02 | Janssen Pharmaceutica Nv | Imidazopyridazines as modulators of IL-17 |
| US11691979B2 (en) | 2020-04-30 | 2023-07-04 | Janssen Pharmaceutica Nv | Imidazopyridazines as modulators of IL-17 |
| JP2023523770A (ja) * | 2020-04-30 | 2023-06-07 | ヤンセン ファーマシューティカ エヌ.ベー. | Il-17のモジュレーターとしてのイミダゾピリミジン |
| WO2021250194A1 (en) | 2020-06-12 | 2021-12-16 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021255085A1 (en) | 2020-06-18 | 2021-12-23 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021255086A1 (en) | 2020-06-18 | 2021-12-23 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2021255174A1 (en) | 2020-06-18 | 2021-12-23 | Leo Pharma A/S | Small molecule modulators of il-17 |
| KR20230020513A (ko) * | 2020-07-04 | 2023-02-10 | 히트젠 주식회사 | 면역 조절제 |
| KR102817731B1 (ko) | 2020-07-04 | 2025-06-10 | 히트젠 주식회사 | 면역 조절제 |
| WO2022007462A1 (zh) * | 2020-07-04 | 2022-01-13 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| AU2021304045B2 (en) * | 2020-07-04 | 2024-05-30 | Hitgen Inc. | Immunomodulator |
| KR20230021083A (ko) * | 2020-07-04 | 2023-02-13 | 히트젠 주식회사 | 면역 조절제 |
| KR102817726B1 (ko) | 2020-07-04 | 2025-06-10 | 히트젠 주식회사 | 면역 조절제 |
| CN113943278A (zh) * | 2020-07-16 | 2022-01-18 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| CN113943278B (zh) * | 2020-07-16 | 2023-08-29 | 成都先导药物开发股份有限公司 | 一种免疫调节剂 |
| EP3943495A1 (en) | 2020-07-24 | 2022-01-26 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2022096411A1 (en) | 2020-11-09 | 2022-05-12 | UCB Biopharma SRL | Dicyclopropylmethyl derivatives as il-17 modulators |
| WO2022096412A1 (en) | 2020-11-09 | 2022-05-12 | UCB Biopharma SRL | Dicyclopropylmethyl derivatives as il-17 modulators |
| WO2022128584A1 (en) | 2020-12-14 | 2022-06-23 | UCB Biopharma SRL | Imidazopyridazine derivatives as il-17 modulators |
| WO2023275301A1 (en) | 2021-07-01 | 2023-01-05 | UCB Biopharma SRL | Imidazotriazine derivatives as il-17 modulators |
| WO2023283453A1 (en) | 2021-07-09 | 2023-01-12 | Dice Alpha, Inc. | Phenyl acetamide based il-17a modulators and uses thereof |
| WO2023025783A1 (en) | 2021-08-23 | 2023-03-02 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2023049888A1 (en) * | 2021-09-27 | 2023-03-30 | Janssen Pharmaceutica Nv | Benzimidazoles as modulators of il-17 |
| WO2023111181A1 (en) | 2021-12-16 | 2023-06-22 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2023166172A1 (en) | 2022-03-04 | 2023-09-07 | Leo Pharma A/S | Small molecule modulators of il-17 |
| WO2024017880A1 (en) | 2022-07-22 | 2024-01-25 | UCB Biopharma SRL | Imidazotriazine derivatives as il-17 modulators |
| US12065429B2 (en) | 2022-12-02 | 2024-08-20 | Leo Pharma A/S | Small molecule modulators of IL-17 |
| WO2024115662A1 (en) | 2022-12-02 | 2024-06-06 | Leo Pharma A/S | Small molecule modulators of il-17 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN110511213A (zh) | 2019-11-29 |
| CN110511213B (zh) | 2021-10-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN110511213B (zh) | 一种免疫调节剂 | |
| CN109311851B (zh) | 二氢嘧啶基苯并氮杂䓬甲酰胺化合物 | |
| CN112341435B (zh) | 一种免疫调节剂 | |
| DE69509173T2 (de) | 2-(2-Amino-1,6-dihydro-6-oxo-purin-9-yl)methoxy-1,3-propandiol-Derivate | |
| KR102764121B1 (ko) | 항-염증제, 면역조절제 및 항-증식제로서의 신규한 칼슘 염 다형체 | |
| AU2002316235A2 (en) | HIV protease inhibitors, compositions containing the same, their pharmaceutical uses and materials for their synthesis | |
| JP6821701B2 (ja) | N−[2−(3−ヒドロキシ−3−メチルブチル)−6−(2−ヒドロキシプロパン−2−イル)−2h−インダゾール−5−イル]−6−(トリフルオロメチル)ピリジン−2−カルボキサミドの結晶形態 | |
| WO2021027724A1 (zh) | 一种免疫调节剂 | |
| CN107001378A (zh) | 吡咯并嘧啶化合物 | |
| CN112824399B (zh) | 一种免疫调节剂 | |
| CN113943278B (zh) | 一种免疫调节剂 | |
| CA3197619A1 (en) | Benzylamine or benzyl alcohol derivative and use thereof | |
| JP6827942B2 (ja) | トリプトリドのc14ヒドロキシルエステル化アミノ酸誘導体、ならびにその製造方法および使用 | |
| CN104974221B (zh) | 二肽及三肽类蛋白酶体抑制剂及其制法和药物用途 | |
| TWI537251B (zh) | Fpr1拮抗劑的衍生物及其用途 | |
| CN114957270B (zh) | 一种s(+)-普拉洛芬衍生物及其制备方法与用途 | |
| EP1966167B1 (fr) | Derives diaryltriazolmethylamine, leur preparation et leur application en therapeutique. | |
| CN118541366A (zh) | 二氢噁唑衍生物化合物 | |
| JP2006502975A (ja) | 化学物質または熱刺激または侵害受容体炎症媒介物に対する応答を阻害することのできる化合物、該化合物を得る方法、および該化合物を含む組成物 | |
| CN108484640B (zh) | 一种抗肿瘤的细胞凋亡蛋白抑制剂 | |
| US20230017312A1 (en) | Centrally active p38alpha inhibiting compounds | |
| WO2015114660A1 (en) | Novel heterobicyclic compounds as kappa opioid agonists | |
| TWI892182B (zh) | 吡唑化合物、製備方法、其醫藥組合物及其用途 | |
| TWI614021B (zh) | Fpr1拮抗劑的衍生物及其用途(一) | |
| CN113365980B (zh) | 氘原子取代的吲哚甲酰胺类衍生物的晶型及其制备方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19808056 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19808056 Country of ref document: EP Kind code of ref document: A1 |