WO2019175093A1 - Method for treating lung cancer - Google Patents
Method for treating lung cancer Download PDFInfo
- Publication number
- WO2019175093A1 WO2019175093A1 PCT/EP2019/056002 EP2019056002W WO2019175093A1 WO 2019175093 A1 WO2019175093 A1 WO 2019175093A1 EP 2019056002 W EP2019056002 W EP 2019056002W WO 2019175093 A1 WO2019175093 A1 WO 2019175093A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- egfr
- subject
- pharmaceutical composition
- mutation
- tyrosine kinase
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- VIMLDCCJUWCIEI-IBGZPJMESA-N C[C@@H](CCc1cc(C#CN)ccc1)c1c(cc(c(OCCOC)c2)OCCOC)c2ncn1 Chemical compound C[C@@H](CCc1cc(C#CN)ccc1)c1c(cc(c(OCCOC)c2)OCCOC)c2ncn1 VIMLDCCJUWCIEI-IBGZPJMESA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
- C12Q1/6886—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material for cancer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/517—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with carbocyclic ring systems, e.g. quinazoline, perimidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/106—Pharmacogenomics, i.e. genetic variability in individual responses to drugs and drug metabolism
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
Definitions
- the present invention relates to a method of treatment with an EGFR tyrosine kinase inhibitor.
- the present invention further relates to a method for selecting suitable cancer treatment regimens for patients and to a method of treating certain drug- resistant cancers.
- the present invention also relates to a pharmaceutical composition for use in treatment with an EGFR tyrosine kinase inhibitor.
- the present invention relates to a method of treatment with an EGFR tyrosine kinase inhibitor, to selecting suitable cancer treatment regimens for patients, and to a pharmaceutical composition for use in such methods.
- the epidermal growth factor receptor (EGFR) lias been identified as a target for the treatment of a number of cancers, in particular, solid tumors, as it is involved in regulating cellular functions important in the proliferation and survival of cancer cells.
- Increased expression of EGFR has been observed in bladder, breast, glioblastoma, head and neck, lung and stomach cancers.
- Development of cancer may for example be associated with an activating mutation in EGFR, and high expression of EGFR is often related to poor prognosis.
- Activating cancerous mutations in EGFR are often somatic gain-of- function mutations in exons that encode the tyrosine kinase domain of the receptor.
- Examples of such mutations identified in lung adenocarcinomas in non-small cell lung cancer (NSCLC) patients, include multi-nucleotide in-frame deletions in exon 19 (“Exonl9 del” involving elimination of four amino acids, Leu-Axg-Glu-Ala) , and a single nucleotide substitution at nucleotide 2573 (T G) in exon 21, resulting in substitution of arginine for leucine at position 858 (L858R).
- NSCLC non-small cell lung cancer
- activating mutations have been found to increase sensitivity to EGFR tyrosine kinase inhibitors (TKIs). Consequently, first line therapies targeting EGFR are often based on inhibition of the tyrosine kinase activity of the mutant receptor.
- Established first line therapies for patients with an activating mutation in EGFR include use of EGFR TKIs such as gefitinib (IressaTM), erlotinib (TarcevaTM) and afatinib (GilotrifTM).
- EGFR TKIs such as gefitinib (IressaTM), erlotinib (TarcevaTM) and afatinib (GilotrifTM).
- IressaTM gefitinib
- TarcevaTM erlotinib
- GalotrifTM afatinib
- EGFR TKIs such as osimertinib (AZD9291), rociletinib (CO-1686), WZ4002 and EGF816, which inhibit EGFR receptors that possess the activating mutations in the tyrosine kinase domain, such as exon 19del and L858R mutations, as well as the T790M mutation in pre- clinical models (Patent Document 1).
- T790M mutation in EGFR was found in about 50% or more of patients with acquired resistance to EGFR TKI treatment (Non- patent document 1).
- Osimertinib exhibits significant efficacy in patients with T790M positive advanced non-small-cell lung cancer, which progressed during first-line EGFR-TKI therapy (Non-patent document 2).
- the regulatory authority in Japan has issued approval of osimertinib mesylate (TagrissoTM) for the treatment of patients with EGFR T790M mutation-positive inoperable or recurrent non-small cell lung cancer (NSCLC) that is resistant to EGFR TKI therapy.
- T790M mutation in EGFR was found in about 50% or more of patients with acquired resistance to EGFR TKI treatment (Non- patent document 1).
- Osimertinib exhibits significant efficacy in patients with T790M positive advanced non-small-cell lung cancer, which progressed during first-line EGFR-TK
- Patent Document 1 WO 2013/014448;
- Non-Patent Document 1 A. Cortot et al., Eur. Respir. Rev. 2014; 23: 356-366;
- Non-Patent Document 2 T.S. Mok et al., N. Engl. J. Med. 2017; 376: 629-640;
- Non-Patent Document 3 M. Watanabe et al., Clin. Cancer Res. 2015; 21: 3552-
- NSCLC non-small cell lung cancer
- the present inventors conducted detailed analysis on the T790M mutation in EGFR of patients before starting treatment with EGFR-TKIs.
- the inventors have found that certain levels of allele frequency of the EGFR T790M mutation is predictive of the efficacy of the results of treatment with first-line EGFR-TKIs such as gefitinib, erlotinib, and afatinib.
- the inventors have further found that the required levels of allele frequency which are predictive of such efficacy cannot currently be detected with real time PCR technique.
- a pharmaceutical composition comprising an effective amount of an EGFR tyrosine kinase inhibitor, for use in a method of treating a subject suffering from non small cell lung cancer (NSCLC),
- NSCLC non small cell lung cancer
- a pharmaceutical composition comprising an effective amount of a third- generation EGFR-TKI or a pharmaceutically acceptable salt thereof, for use in a method of treating a subject suffering from non-small cell lung cancer (NSCLC),
- NSCLC non-small cell lung cancer
- a pharmaceutical composition comprising an effective amount of a third- generation EGFR-TKI or a pharmaceutically acceptable salt thereof, for use in a method of treating a subject suffering from non- small cell lung cancer (NSCLC),
- NSCLC non- small cell lung cancer
- the method comprises administration of the composition to the subject where the allele frequency of T790M mutation in EGFR is more than about 0.3%.
- composition according to any one of (1-3) to (1-5), wherein the third- generation EGFR-TKI is selected from osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof.
- (2-2) The method according to (2-1), wherein the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotinib, afatinib, osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof, in particular the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotinib, afatinib and osimertinib, and a pharmaceutically acceptable salt thereof.
- TKI or a pharmaceutically acceptable salt thereof is selected to be administered to a subject having T790M mutation in EGFR with allele frequency of more than about 0.3%.
- NSCLC NSCLC
- a sample of the cancer derived from the said subject has been analyzed and the allele frequency of T790M mutation in EGFR has been confirmed
- the method comprises administration of a third-generation EGFR-TKI or a pharmaceutically acceptable salt thereof to the subject where the allele frequency of T790M mutation in EGFR is more than about 0.3%.
- (2-6) The method according to any one of (2-3) to (2-5), wherein the third- generation EGFR-TKI is selected from osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof.
- the present invention provides a pharmaceutical composition comprising an EGFR tyrosine kinase inhibitor, for use in treatment of NSCLC, with the purpose of preventing or delaying disease progression at least partially due to acquired resistance thereto, and is beneficial for a patient suffering from NSCLC by providing a longer progression free survival (PFS).
- PFS progression free survival
- the present invention enables the clinical practitioner to select the appropriate EGFR-TKI treatment for a given patient based upon the allele frequency of T790M mutation in EGFR.
- Figure 1 shows a graph indicating that the cut-off value, for which sum of sensitivity plus specificity was maximal, was 0.34% in light of the results of the study.
- Figure 2 shows a graph indicating that the subgroups of treatment-naive patients who are T790M positive with high allele frequency had a shorter median PFS (7.7 months) than the subgroups of treatment-naive patients who are T790M positive with low allele frequency (17.1 months) and T790M negative (13.5 months).
- Figure 3 shows a graph indicating the PFS of the subgroups of treatment-naive patients who are T790M positive with high allele frequency (median PFS: 7.7 months) compared with the subgroup of treatment-naxve patients who are T790M positive with low allele frequency or negative (13.6 months).
- an epidermal growth factor receptor tyrosine kinase inhibitor may be selected from the compounds that are known in the art, as exemplified by gefitinib, erlotinib, icotinib, neratinib, afatinib, dacomitinib (PF00299804), osimertinib, rociletinib, WZ4002, EGF816, ASP8273, HM61713, PF06747775, and the like.
- gefitinib As an EGFR tyrosine kinase inhibitor, gefitinib or a pharm aceutically acceptable salt thereof may be used in the present invention.
- Gefitinib is an EGFR TKI drug that is approved for use in advanced NSCLC in a number of territories. The gefitinib structure is shown below:
- Gefitinib may also be known by the chemical name N-(3-chloro-4-£luorophenyI)- 7-methoxy-6-(3-morpholinopropoxy)quinazolin-4-amine.
- erlotinib As an EGFR tyrosine kinase inhibitor, erlotinib or a pharmaceutically acceptable salt thereof may be used in the present invention.
- Erlotinib is an EGFR TKI drug used against pancreatic, lung and other cancers.
- the chemical structure of erlotinib is shown below:
- Erlotinib may also be known by the chemical name: N - (3 -ethynylphenyl) -6,7- bis (2 - methoxyethoxy)quinazolin- 4- amine.
- afatinib As an EGFR tyrosine kinase inhibitor, afatinib or a pharmaceutically acceptable salt thereof may be used in the present invention.
- Afatinib is an EGFR TKI drug used against NSCLC in particular. The drug can sometimes be used to treat cancers which are, or have become, resistant to treatment with gefitinib or erlotinib.
- the afatinib structure is shown below:
- Afatinib may also be known by the chemical name (2E)-N-[4-[(3-chloro-4- fluorophenyl)amino] -7-[[(3S) -tetrahydro- 3-furanyljoxy] -6- quinazolinyl] -4- (dim ethylamino) - 2 -bufenamide .
- Osimertinib As an EGFR tyrosine kinase inhibitor, osimertinib (AZD9291) may be used in the present invention. Osimertinib may sometimes be used to treat cancers which are, or have become, resistant to treatment with EGFR inhibitors such as gefitinib, erlotinib, and/or afatinib. Osimertinib has the following chemical structure:
- Osimertinib may be known by the chemical name: N-(2- ⁇ 2- (dimethylamino)ethyl-methyl-amino ⁇ -4-methoxy-5- ⁇ [4-(l-methylindol-3-yl)pyrimidin-2- yl] a mino]phenyl)prop -2 -enami de .
- Osimertinib and pharmaceutically acceptable salts thereof are disclosed in international patent application number PCT/GB2012/051783 (publication number WO2013/014448).
- a pharmaceutically acceptable salt of osimertinib may be any of the salts described herein, for example a mesylate salt.
- CO-1686 As an EGFR tyrosine kinase inhibitor, CO-1686 or a pharmaceutically acceptable salt thereof can be used in the present invention.
- CO- 1686 may be also known as "rociletinib, and the chemical structure of CO-1686 is shown below:
- Rociletinib may be known as CO-1686 or by the chemical name: N-(3- ⁇ [2- ⁇ [4-(4- acetylpip erazin-l-yl) -2 -methoxyphenyl] amino] - 5- (trifluorom ethyl)pyrimidin- 4 - yl]amino ⁇ phenyl)prop-2-enamide.
- a pharmaceutically acceptable salt of rociletinib may, for example, comprise an HBr salt.
- EGF816 As an EGFR tyrosine kinase inhibitor, EGF816 or a pharmaceutically acceptable salt thereof can be used in the present invention.
- the chemical structure of EGF816 is shown below:
- EGF816 may be known by the chemical name: (i££)-N-(7-chloro-l-(l-(4-)
- WZ4002 As an EGFR tyrosine kinase inhibitor, WZ4002 can be used in the present invention. This compound is described in a publication by Zhou et al. (Nature 462,
- an approved EGFR-TKI is used in the present invention.
- WZ4002 Bioorganic & Medicinal Chemistry Letters, Vol. 27, No. 21, 2017, 4832-4837
- rociletinib Liung Cancer (Auckl). 2016; 7: 91-97
- EGF816 WO 2016/185333 Al
- HM61713 J. Thorac. Oncol. 2016, ll(4):e45-7.
- PF06747775 Annals of Oncology (2017) 28 (suppl 5): v460-v496) and osimertinib are known herein as“third-generation EGFR- TKIs”.
- the expression“first generation EGFR TKIs” includes erlotinib, gefitinib, icotinib and any combinations thereof.
- the expression“second generation EGFR TKIs” includes afatinib, dacomitinib and any combinations thereof.
- the EGFR-TKIs may be administered in accordance with dosage amount, administration route, pharmaceutical formulation, and dosage regimen in accordance with the approval by regulatory authority.
- the NSCLC includes advanced NSCLC or metastatic NSCLC.
- Advanced NSCLC refers to locally advanced or metastatic NSCLC.
- advanced NSCLC the NSCLC has spread to nearby tissue or to far away lymph nodes.
- Locally advanced NSCLC is defined as stage IIIB NSCLC not amenable to definitive multi- modality therapy including surgery.
- Metastatic NSCLC refers to stage IV NSCLC.
- metastatic NSCLC the cancer has spread to other organs of the body.
- the NSCLC is inoperable or recurrent NSCLC. The progression of NSCLC may be monitored by methods well known to those in the art.
- the subject may have had treatment with a first- or second-generation EGFR TKI.
- the subject may suffer from NSCLC harboring EGFR activating mutation (e.g. L858R and/or exl9del) and/or an acquired EGFR T790M mutation.
- the NSCLC may be primary or recurrent.
- treatment-naive patients refers to patients suffering from NSCLC, who have not had previously received treatment with EGFR tyrosine kinase inhibitors.
- the subject is a patient suffering from NSCLC, who has not had previously received treatment with EGFR tyrosine kinase inhibitors.
- the subject invention may be applied to patients, who have received a first diagnosis of primary or recurrent NSCLC.
- EGFR mutation status may be determined by tests available in the art, e.g. QIAGEN therascreenTM EGFR test or other tests approved by regulatory authorities.
- the therascreen EGFR RGQ PCR Kit is an FDA- approved, qualitative real-time PCR assay for the detection of specific mutations in the EGFR oncogene.
- Evidence of EGFR mutation can be obtained from existing local data and testing of tumor samples.
- EGFR mutation status may be determined from any available tumor tissue, or from plasma samples.
- allele frequency of mutation in EGFR may be confirmed by the procedure known in the art, including droplet digital PCR (ddPCR) technique. Analysis using ddPCR may be conducted by the procedures known in the art (e.g. M. Watanabe et al., Clin. Cancer Res. 2015; 21: 3552-3560). Allele frequency of the mutation may be calculated in the manner described in the subject specification.
- ddPCR droplet digital PCR
- the allele frequency of T790M mutation in EGFR may be used as a criteria to determine the selection of treatment with EGFR TKIs In one embodiment, the selection is made based on the fact as to whether the tumor sample derived from the subject has T790M mutation in EGFR with allele frequency of more than about 0.30%, about 0.35%, about 0.40%, about 0.45%, or about 0.50%.
- the term“about” when referring to any given numerical value means within [0068]
- the anti-cancer treatment described hereinbefore may be applied as a sole therapy or may involve, in addition to the compound of the invention, conventional surgery or radiotherapy or chemotherapy or im munotherapy. Such chemotherapy could be administered concurrently, simultaneously, sequentially or separately to treatment with the compound of the invention and may include one or more of the following categories of anti-tumour agents :-
- antiproliferative/antineoplastic drugs and combinations thereof, as used in medical oncology such as alkylating agents (for example cis-platin, oxaliplatin, carboplatin, cyclophosphamide, nitrogen mustard, melphalan, chlorambucil, busulphan, temozolamide and nitrosoureas); antimetabolites (for example gemcitabine and antifolates such as fhioropyrimi dines like 5-fluorouracil and tegafur, raltitrexed, methotrexate, cytosine arabinoside, and hydroxyurea); antitumour antibiotics (for example anthracyclines like adriamycin, bleomycin, doxorubicin, daunomycin, epirubicin, idarubicin, mitomycin- C, dactinomycin and mithramycin); antimitotic agents (for example vinca alkaloids like vincristine
- cytostatic agents such as antioestrogens (for example tamoxifen, fulvestrant, toremifene, raloxifene, droloxifene and iodoxyfene), antiandrogens (for example bicalutamide, flutamide, nilutamide and cyproterone acetate), LHRH antagonists or LHRH agonists (for example goserelin, leuprorelin and buserelin), progestagens (for example megestrol acetate), aromatase inhibitors (for example as anastrozole, letrozole, vorazole and exemestane) and inhibitors of 5a-reductase such as finasteride;
- antioestrogens for example tamoxifen, fulvestrant, toremifene, raloxifene, droloxifene and iodoxyfene
- antiandrogens for example b
- anti-invasion agents for example c-Src kinase family inhibitors like 4-(6-chloro- 2,3-methylenedioxyanilino)-7-[2-(4-methylpiperazin-l-yl)ethoxy]-5-tetrahydropyran-4- yloxyquinazoline [AZD0530 (saracatinib); WOOl/94341], N - (2 -chloro- 6 -methylphenyl) - 2 - ⁇ 6-[4-(2-hydroxyethyl)piperazin-l-yl]-2-methylpyrimidin-4-ylamino ⁇ thiazole-6- carboxamide (dasatinib, BMS-354825; J.
- c-Src kinase family inhibitors like 4-(6-chloro- 2,3-methylenedioxyanilino)-7-[2-(4-methylpiperazin-l-yl)ethoxy]-5-tetrahydropyr
- inhibitors of growth factor function include growth factor antibodies and growth factor receptor antibodies (for example the anti-erbB2 antibody trastuzumab [HerceptinTM] , the anti-EGFR antibodies panitumumab or necitumumab, the anti-erbBl antibody cetuximab [Erbitux, C225] and any growth factor or growth factor receptor antibodies disclosed by Stern et al. Critical reviews in oncology/haematology, 2005, Vol.
- growth factor antibodies and growth factor receptor antibodies for example the anti-erbB2 antibody trastuzumab [HerceptinTM] , the anti-EGFR antibodies panitumumab or necitumumab, the anti-erbBl antibody cetuximab [Erbitux, C225] and any growth factor or growth factor receptor antibodies disclosed by Stern et al. Critical reviews in oncology/haematology, 2005, Vol.
- inhibitors of the hepatocyte growth factor family include inhibitors of the insulin growth factor family; inhibitors of the platelet- derived growth factor family such as imatinib and/or nilotinib (AMN107); inhibitors of serine/threonine kinases (for example Ras/Raf signalling inhibitors such as farnesyl transferase inhibitors, for example sorafenib (BAY 43-9006), tipifarnib (R115777) and lonafarnib (SCH66336)), inhibitors of cell signalling through MEK and/or AKT kinases (such as selumetinib), c-kit inhibitors, abl kinase inhibitors, PI3 kinase inhibitors, Plt3 kinase inhibitors, CSF-1R kinase inhibitors, IGF receptor (insulin -like growth factor) kinase inhibitors; aurora kinase inhibitors (for example
- antiangio genic agents such as those which inhibit the effects of vascular endothelial growth factor, [for example the anti -vascular endothelial cell growth factor antibody bevacizumab (AvastinTM) or ramucirinab) and for example, a VEGF receptor tyrosine kinase inhibitor such as vandetanib (ZD6474), vatalanib (PTK787), sunitinib (SU11248), axitinib (AG-013736), pazopanib (GW 786034) and 4- (4 -fluoro- 2 -methylindol- 5 -yloxy) - 6 -methoxy- 7 - (3-pyrrolidin- 1 -ylpropoxy)quinazoline (AZD2171; Example 240 within WO 00/47212), compounds such as those disclosed in W097/22596, W097/30035, W097/32856 and W098/13354 and compounds
- vascular damaging agents such as Combretastatin A4 and compounds disclosed in WO99/02166, WOOO/40529, WOOO/41669, WOO 1/92224, W002/04434 and
- an endothelin receptor antagonist for example zibotentan (ZD4054) or atrasentan
- antisense therapies for example those which are directed to the targets listed above, such as ISIS 2503, an anti-ras antisense;
- (ix) gene therapy approaches including for example approaches to replace aberrant genes such as aberrant p53 or aberrant BRCAl or BRCA2, GDEPT (gene-directed enzyme pro-drug therapy) approaches such as those using cytosine deaminase, thymidine kinase or a bacterial nitroreductase enzyme and approaches to increase patient tolerance to chemotherapy or radiotherapy such as multi-drug resistance gene therapy; and
- GDEPT gene-directed enzyme pro-drug therapy
- (x) immunotherapy approaches including for example ex-vivo and in-vivo approaches to increase the immunogenicity of patient tumour cells, such as transfection with cytokines such as interleukin 2, interleukin 4 or granulocyte-macrophage colony stimulating factor, approaches to decrease T-cell anergy, approaches using transfected immune cells such as cytokine-transfected dendritic cells, approaches using cytokine-transfected tumour cell lines, approaches using anti- idiotypic antibodies, approaches to decrease the function of immune suppressive cells such as regulatory T cells, myeloid-derived suppressor cells or IDO (indoleamine 2,3,-deoxygenase)- expressing dendritic cells, and approaches using cancer vaccines consisting of proteins or peptides derived from tumour-associated antigens such as NY-ESO-1, MAGE-3, WTl or Her2/neu.
- cytokines such as interleukin 2, interleukin 4 or granulocyte-macrophage colony stimulating factor
- the EGFR TKIs or other drugs may he administered in the form of a pharmaceutically acceptable salt.
- pharmaceutically acceptable salt refers to a salt, which is non-toxic and is prepared with a pharm a ceutically acceptable base or acid.
- the pharmaceutically acceptable salt thereof is generally prepared by adding the compound with a suitable organic or inorganic acid.
- the acid- addition salt include acetate, benzenesulfonate, benzoate, citrate, fumarate, glutamate, hydrobromide, hydrochloride, lactate, maleate, mandelate, mesylate, oxalate, palmitate.
- the pharmaceutically acceptable salt thereof is generally prepared by adding the compound with a suitable organic or inorganic base.
- the pharmaceutical composition may be administered as a pharmaceutical formulation.
- the pharmaceutical formulation may be produced by well-known methods, using an additive such as an excipient, a coating agent, a lubricant, a binder, a disintegrator, a stabilizer, a corrigent, a diluent, a solvent, a surfactant or an emulsifier.
- excipients include starches such as potato starch and cornstarch, lactose, erythritol, mannitol, crystalline cellulose, microcrystalline cellulose, and calcium hydrogen phosphate.
- coating agents include ethylcellulose, hydroxypropyleellulose. hydroxypropylmethylcellulose, shellac, talc, camauba wax, paraffin, and Opadry IITM.
- Examples of lubricants include magnesium stearate sodium stearyl fumarate, and stearic acid.
- binders examples include polyvinylpyrrolidone, macrogol, and the same substances exemplified as the excipients.
- disintegrators include the same substances exemplified as the excipients and low-substituted hydroxypropyl cellulose, croscarmellose sodium, sodium carboxymethylstarch, and chemically modified starch or celluloses.
- stabilizers include p araoxyb enzoic acid esters such as methylparaben and propylparaben; alcohols such as chlorobutanol, benzylaleohol, and phenylethylaleohol; benzalkonium chloride; phenols such as phenol and cresol; thimerosal; dehydro acetic acid; and ascorbic acid.
- p araoxyb enzoic acid esters such as methylparaben and propylparaben
- alcohols such as chlorobutanol, benzylaleohol, and phenylethylaleohol
- benzalkonium chloride phenols such as phenol and cresol
- thimerosal thimerosal
- dehydro acetic acid and ascorbic acid.
- Examples of corrigents include sweeteners which are conventionally used.
- diluents include the substances exemplified as excipients.
- solvents include ethanol, phenol, chlorocresol, purified water, and distilled water
- surfactants or emulsifiers examples include polysorbate 80, polyoxyl 40 stearate, lauromacrogol, soybean oil, Maisin 35-1 (Gattefosse, France) and Cremophor
- the dosage amount of the EGFR TKIs or other drugs differs depending on the symptoms, age, body weight, relative health condition, presence of other medication, method of administration, and the like.
- An appropriate route of the administration of the EGFR TKIs or other drugs may be determined by a person skilled in the art. Examples thereof include oral, endorectal, parenteral (intravenous, intramuscular, or subcutaneous), intracisternal, vaginal, intraperitoneal, intravesical, topical (drip infusion, powder, ointment, gel, or cream) application, and inhalation (intraoral or nasal spray).
- the dosage form of the EGFR TKIs or other drugs for the administration includes tablets capsules, granules, powders, pills, aqueous and non-aqueous solutions or suspensions for oral use, and parenteral solutions loaded into containers.
- the EGFR TKI is administered orally in an appropriate formulation including tablets.
- FFPE Formalin- Fixed Paraffin-Embedded
- Genomic DNA was extracted from the FFPE specimens with the QIAmp DNA FFPE tissue kit (Qiagen) in accordance with the manufacturer’s instructions. Quality and quantity of the genomic DNA sample were assessed using NanoDrop 2000 (Life Technologies) and Qubit 2.0 (Life Technologies) platforms.
- Primers and probes were procured from Integrated DNA Technologies K.K. (IDT K.K.). Fluorescent probes targeting wild-type and mutant sequences were respectively conjugated to tetrachlorofluorescein (TET, Aex 522 nm/Aem 539 nm) or 6- carboxyfluorescein (FAM, Aex 494 nm/Aem 522 nm) fluorophores with ZEN/IABkFQ double quencher. The sequences of primers and probes for detection of the T790M mutation are shown in Table 2. Table 2:
- a collection of uniformly sized aqueous droplets was produced by hydrodynamic flow-focusing with a droplet-generating microfluidic chip (Source chip, RainDance Technologies) in accordance with the manufacturer’s instructions.
- a droplet-generating microfluidic chip (Source chip, RainDance Technologies) in accordance with the manufacturer’s instructions.
- One lane of the Source chip was used for positive control.
- the resulting emulsion was collected in a PCR tube strip comprising eight 0.2-mL conical-bottom PCR tubes.
- the PCR tube strip containing a total of 75 pL of droplets and carrier oil, was tightly capped with an 8-Strip Dome Cap, and then placed in a thermal cycler with a hot lid (Proflex PCR system, Life Technologies).
- the emulsion was subjected to thermal cycling, namely starting with a hot start denaturation step of 10 min at 95 °C, followed by 45 cycles of: 95 °C, 15 sec; 58 °C, 15 sec; 60 °C, 45 sec (using a 0.5 °C/min ramp rate). These cycles were followed by a final step at 98 °C for 10 min and then at 12 °C for 10 min.
- the thermal cycling condition is summarized in Table 4.
- the emulsions were processed immediately to measure the endpoint fluorescence signal from each droplet. Namely the thermal-cycled emulsion was transferred into a second microfluidic chip (Sense chip, RainDance Technologies), and then endpoint fluorescence signals were measured in accordance with the manufacturer’s instructions.
- the droplet event data were analyzed with the RainDrop Analyst software (RainDance Technologies) in accordance with the manufacturer’s instructions. Briefly, sample data were loaded with a drop size gating template (RainDance Technologies). Data from the positive control sample were used to create the compensation matrix in the RainDrop Analyst software. The compensation matrix was applied to the data from each sample to eliminate the crosstalk fluorescence signals from the TET and FAM fluorophores. The sizes and locations of the wild-type and the mutant gates were established by manual selection of the area containing wild-type or mutant clusters in the positive control. [0098] For each unknown sample, the number of PCR-positive droplet events was counted within each gate. The number of events within each gate was converted to the number of events per assay using the total number of intact drops.
- raw mutant allele frequency (MAF) was calculated as follows:
- tumor content means ratio of an area of tumor in a section to a total area of a section.
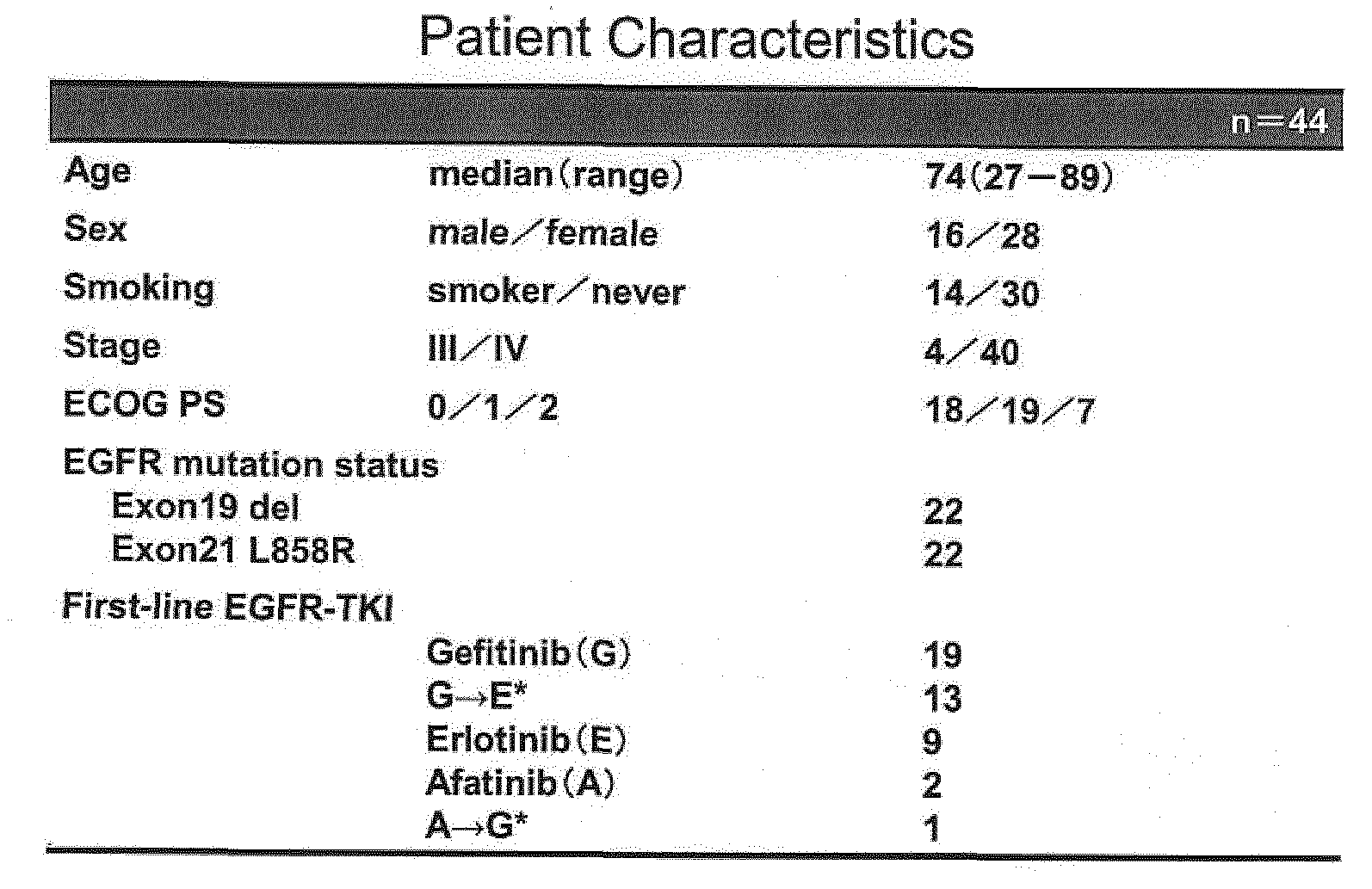
- the patients were stratified with the corrected pretreatment T790M MAF.
- the patients with more than 30 events of the mutation per assay is defined as positive (i.e. cut-off value: >30 events/assay), and overall incidence of T790M mutation in treatment -naive patents is 40.9% (18/44).
- High frequency of the mutant allele is found in 12 patients, and low frequency is found in 6 patients.
- PFS of each subgroup is indicated in Figs. 2 and 3. The results of Fig.
- Fig. 3 indicates the PFS of the subgroups of treatment-naive patients who are T790M positive with high frequency (median PFS: 7.7 months) compared with the subgroup of treatment-naive patients who are T790M positive with low frequency or negative (13.6 months).
- T790M allele mutation frequency in treatment-naive patents is an adverse factor for PFS in the treatment with EGFR- TKIs.
- the T790M allele frequency can be used for determining a selection of an EGFR-TKI for treating NSCLC.
- treatment-naive patients who exhibit more than 0.3% of T790M allele mutation frequency are unlikely to show an optimal response to first- or second- generation EGFR-TKIs, and such patients are more likely to respond more favorably to early administration of third-generation EGFR-TKIs, such as osimertinib.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Analytical Chemistry (AREA)
- Pathology (AREA)
- Genetics & Genomics (AREA)
- Oncology (AREA)
- Hospice & Palliative Care (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
An object of the present invention is to provide further measures to treat patients with NSCLC without disease progression. The present invention provides a pharmaceutical composition comprising an effective amount of an EGFR tyrosine kinase inhibitor, for use in a method of treating a subject suffering from non-small cell lung cancer, wherein the method comprises determining selection of an EGFR tyrosine kinase inhibitor for administration to the subject based on the allele frequency of T790M mutation in EGFR.
Description
Title: Method for treating lung cancer
TECHNICAL FIELD
[0001] The present invention relates to a method of treatment with an EGFR tyrosine kinase inhibitor. The present invention further relates to a method for selecting suitable cancer treatment regimens for patients and to a method of treating certain drug- resistant cancers. The present invention also relates to a pharmaceutical composition for use in treatment with an EGFR tyrosine kinase inhibitor. In particular, the present invention relates to a method of treatment with an EGFR tyrosine kinase inhibitor, to selecting suitable cancer treatment regimens for patients, and to a pharmaceutical composition for use in such methods.
BACKGROUND ART;
[0002] The epidermal growth factor receptor (EGFR) lias been identified as a target for the treatment of a number of cancers, in particular, solid tumors, as it is involved in regulating cellular functions important in the proliferation and survival of cancer cells. Increased expression of EGFR has been observed in bladder, breast, glioblastoma, head and neck, lung and stomach cancers. Development of cancer may for example be associated with an activating mutation in EGFR, and high expression of EGFR is often related to poor prognosis.
[0003] Activating cancerous mutations in EGFR are often somatic gain-of- function mutations in exons that encode the tyrosine kinase domain of the receptor. Examples of such mutations, identified in lung adenocarcinomas in non-small cell lung cancer (NSCLC) patients, include multi-nucleotide in-frame deletions in exon 19 (“Exonl9 del” involving elimination of four amino acids, Leu-Axg-Glu-Ala) , and a single nucleotide substitution at nucleotide 2573 (T G) in exon 21, resulting in substitution of arginine for leucine at position 858 (L858R). These mutations (generally known as“activating mutations”) have been found to increase sensitivity to EGFR tyrosine kinase inhibitors (TKIs). Consequently, first line therapies targeting EGFR are often based on inhibition of the tyrosine kinase activity of the mutant receptor.
[0004] Established first line therapies for patients with an activating mutation in EGFR include use of EGFR TKIs such as gefitinib (Iressa™), erlotinib (Tarceva™) and afatinib (Gilotrif™). However, despite initial responses to these EGFR TKIs, a significant proportion of patients ultimately show disease progression due to acquired resistance, which has been shown in many cases to be associated with an additional mutation in EGFR, known as T790M.
[0005] Acquired resistance has led to the development of third-generation EGFR TKIs, such as osimertinib (AZD9291), rociletinib (CO-1686), WZ4002 and EGF816, which inhibit EGFR receptors that possess the activating mutations in the tyrosine kinase domain, such as exon 19del and L858R mutations, as well as the T790M mutation in pre- clinical models (Patent Document 1).
[0006] T790M mutation in EGFR was found in about 50% or more of patients with acquired resistance to EGFR TKI treatment (Non- patent document 1). Osimertinib exhibits significant efficacy in patients with T790M positive advanced non-small-cell lung cancer, which progressed during first-line EGFR-TKI therapy (Non-patent document 2). The regulatory authority in Japan has issued approval of osimertinib mesylate (Tagrisso™) for the treatment of patients with EGFR T790M mutation-positive inoperable or recurrent non-small cell lung cancer (NSCLC) that is resistant to EGFR TKI therapy.
CITATION LIST
PATENT DOCUMENTS
[0007] Patent Document 1: WO 2013/014448;
NON-PATENT DOCUMENTS
[0008] Non-Patent Document 1: A. Cortot et al., Eur. Respir. Rev. 2014; 23: 356-366;
Non-Patent Document 2: T.S. Mok et al., N. Engl. J. Med. 2017; 376: 629-640; Non-Patent Document 3: M. Watanabe et al., Clin. Cancer Res. 2015; 21: 3552-
3560;
SUMMARY OF INVENTION TECHNICAL PROBLEM
[0009] Many patients suffering from non-small cell lung cancer (NSCLC) whose tumors possess EGFR activating mutations and are treated with first- line EGFR-TKI therapy, nevertheless show disease progression during first-line EGFR-TKI therapy with the development of further mutations in EGFR. Particularly resistance due to T790M mutation in EGFR is often found in patients with disease progression. Whether, and when, any given patient being treated with first -line EGFR-TKI therapy will develop acquired resistance cannot presently be accurately predicted. A better understanding of which patients are likely to develop such resistance would therefore provide the clinical practitioner with an important further measure to treat patients with NSCLC
and to prevent or delay disease progression.
SOLUTION TO PROBLEM
[0010] The present inventors conducted detailed analysis on the T790M mutation in EGFR of patients before starting treatment with EGFR-TKIs. The inventors have found that certain levels of allele frequency of the EGFR T790M mutation is predictive of the efficacy of the results of treatment with first-line EGFR-TKIs such as gefitinib, erlotinib, and afatinib. The inventors have further found that the required levels of allele frequency which are predictive of such efficacy cannot currently be detected with real time PCR technique.
[0011] According to one aspect of the present invention, there is provided the following relating to a pharmaceutical composition:
[0012] (1-1) A pharmaceutical composition comprising an effective amount of an EGFR tyrosine kinase inhibitor, for use in a method of treating a subject suffering from non small cell lung cancer (NSCLC),
wherein the method comprises:
preparing a sample of the cancer derived from the subject;
analyzing the sample to confirm allele frequency of T790M mutation in EGFR; confirming whether the subject has T790M mutation in EGFR with allele frequency of more than about 0.3%; and
determining the selection of an EGFR tyrosine kinase inhibitor for administration to the subject based on the allele frequency.
[0013] (1-2) The pharmaceutical composition according to (1-1), wherein the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotinib, afatinib, osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof, in particular the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotinib, afatinib and osimertinib, and a pharmaceutically acceptable salt thereof.
[0014] (1-3) The pharmaceutical composition according to (1-1) or (1-2), wherein a third- generation EGFR-TKI or a pharmaceutically acceptable salt thereof is selected to be administered to a subject having T790M mutation in EGFR with allele frequency of more than about 0.3%.
[0015] (1-4) A pharmaceutical composition comprising an effective amount of a third- generation EGFR-TKI or a pharmaceutically acceptable salt thereof, for use in a method of treating a subject suffering from non-small cell lung cancer (NSCLC),
wherein the method comprises:
preparing a sample of the cancer derived from the subject;
analyzing the sample to confirm allele frequency of T790M mutation in EGFR;
selecting a subject having T790M mutation in EGFR with allele frequency of more than about 0,3%; and
administering the composition to the selected subject.
[0016] (1-5) A pharmaceutical composition comprising an effective amount of a third- generation EGFR-TKI or a pharmaceutically acceptable salt thereof, for use in a method of treating a subject suffering from non- small cell lung cancer (NSCLC),
wherein a sample of the cancer derived from the said subject has been analyzed and wherein the allele frequency of T790M mutation in EGFR has been confirmed, wherein the method comprises administration of the composition to the subject where the allele frequency of T790M mutation in EGFR is more than about 0.3%.
[0017] (1-6) The pharmaceutical composition according to any one of (1-3) to (1-5), wherein the third- generation EGFR-TKI is selected from osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof.
[0018] (1-7) The pharmaceutical composition according to (1-6), wherein the third - generation EGFR-TKI is osimertinib or a pharmaceutically acceptable salt thereof.
[0019] (1-8) The pharmaceutical composition according to any one of (1-1) to (1-7), wherein the subject is a patient who has not previously received treatment with EGFR tyrosine kinase inhibitors.
[0020] (1-9) The pharmaceutical composition according to any one of (1-1) to (1-7), wherein the subject is a patient, who has previously received treatment with an EGFR tyrosine kinase inhibitor other than osimertinib or a pharmaceutically acceptable salt thereof, wherein the EGFR tyrosine kinase inhibitor previously used is changed to osimertinib or a pharmaceutically acceptable salt thereof.
[0021] (1-10) The pharmaceutical composition according to any one of (1-1) to (1-9), wherein the frequency of T790M mutation in EGFR is confirmed by an assay using droplet digital PCR.
[0022] (1-11) The pharmaceutical composition according to (1-10), wherein T790M mutation in EGFR of a patient is determined as negative in a case that the number of PCR-positive droplet events is 30 or less per assay.
[0023] (1-12) The pharmaceutical composition according to any one of (1-1) to (1-11), wherein the subject has one or more mutations in EGFR other than T790M.
[0024] (1-13) The pharmaceutical composition according to any one of (1-1) to (1-12), wherein the mutation in EGFR is selected from Exon 19 del and Exon21 L858R.
[0025] (1-14) The pharmaceutical composition according to any one of (1-1) to (1-13), which is administered to a subject in combination with one or more other anti-cancer agents.
[0026] (1-15) The pharmaceutical composition according to any one of (1-1) to (1-14), wherein the NSCLC is inoperable or recurrent NSCLC.
[0027] (1-16) The pharmaceutical composition according to any one of (1-1) to (1-15), wherein the EGFR tyrosine kinase inhibitor that is administered to a subject is osimertinib mesylate.
[0028] According to another aspect of the present invention, there is provided the following method:
[0029] (2-1) A method of treating a subject suffering from non-small cell lung cancer
(NSCLC),
which comprises:
preparing a sample of the cancer derived from the subject;
analyzing the sample to confirm allele frequency of T790M mutation in EGFR; confirming whether the subject has T790M mutation in EGFR with allele frequency of more than about 0.3%;
determining selection of an EGFR tyrosine kinase inhibitor for administration to the subject based on the allele frequency, and
administering the selected EGFR tyrosine kinase inhibitor to the subject.
[0030] (2-2) The method according to (2-1), wherein the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotinib, afatinib, osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof, in particular the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotinib, afatinib and osimertinib, and a pharmaceutically acceptable salt thereof.
[0031] (2-3) The method according to (2-1) or (2-2), wherein a third-generation EGFR-
TKI or a pharmaceutically acceptable salt thereof is selected to be administered to a subject having T790M mutation in EGFR with allele frequency of more than about 0.3%.
[0032] (2-4) A method of treating a subject suffering from non-small cell lung cancer
(NSCLC),
wherein the method comprises:
preparing a sample derived from the cancer;
analyzing the sample to confirm allele frequency of T790M mutation in EGFR; selecting a subject having T790M mutation in EGFR with allele frequency of more than about 0.3%; and
administering a third-generation EGFR-TKI or a pharmaceutically acceptable salt thereof to the selected subject.
[0033] (2-5) A method of treating a subject suffering from non- small cell lung cancer
(NSCLC), wherein a sample of the cancer derived from the said subject has been analyzed and the allele frequency of T790M mutation in EGFR has been confirmed, wherein the method comprises administration of a third-generation EGFR-TKI or a pharmaceutically acceptable salt thereof to the subject where the allele frequency of T790M mutation in EGFR is more than about 0.3%.
[0034] (2-6) The method according to any one of (2-3) to (2-5), wherein the third- generation EGFR-TKI is selected from osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof.
[0035] (2-7) The method according to (2-6), wherein the third- generation EGFR-TKI is osimertinib or a pharmaceutically acceptable salt thereof.
[0036] (2-8) The method according to any one of (2-1) to (2-7), wherein the subject is a patient, who has not previously received treatment with EGFR tyrosine kinase inhibitors.
[0037] (2-9) The method according to any one of (2-1) to (2-7), wherein the subject is a patient, who has previously received treatment with an EGFR tyrosine kinase inhibitor other than osimertinib or a pharmaceutically acceptable salt thereof, and wherein the EGFR tyrosine kinase inhibitor previously used is changed to osimertinib or a pharmaceutically acceptable salt thereof.
[0038] (2-10) The method according to any one of (2-1) to (2-9), wherein the frequency of T790M mutation in EGFR is confirmed by an assay using droplet digital PCR.
[0039] (2-11) The method according to (2-10), wherein T790M mutation in EGFR of a patient is determined as negative in a case that the number of PCR-positive droplet events is 30 or less per assay.
[0040] (2-12) The method according to any one of (2-1) to (2-11), wherein the subject has one or more mutations in EGFR other than T790M.
[0041] (2-13) The method according to any one of (2-1) to (2-12), wherein the mutation in EGFR is selected from Exon 19 del and Exon21 L858R.
[0042] (2-14) The method according to any one of (2-1) to (2-13). wherein one or more other anti-cancer agents are administered to a subject in combination with the EGFR tyrosine kinase inhibitor.
[0043] (2-15) The method according to any one of (2-1) to (2-14), wherein the NSCLC is inoperable or recurrent NSCLC.
[0044] (2-16) The method according to any one of (2-1) to (2-15), wherein the EGFR tyrosine kinase inhibitor that is administered to the subject is osimertinib mesylate.
E FFECTS OF T1IE INVENTION
[0045] The present invention provides a pharmaceutical composition comprising an EGFR tyrosine kinase inhibitor, for use in treatment of NSCLC, with the purpose of preventing or delaying disease progression at least partially due to acquired resistance thereto, and is beneficial for a patient suffering from NSCLC by providing a longer progression free survival (PFS). In particular, the present invention enables the clinical practitioner to select the appropriate EGFR-TKI treatment for a given patient based upon the allele frequency of T790M mutation in EGFR.
BR1 K K DESCRIPTION OF THE DRAWINGS
[0046] Figure 1 shows a graph indicating that the cut-off value, for which sum of sensitivity plus specificity was maximal, was 0.34% in light of the results of the study.
Figure 2 shows a graph indicating that the subgroups of treatment-naive patients who are T790M positive with high allele frequency had a shorter median PFS (7.7 months) than the subgroups of treatment-naive patients who are T790M positive with low allele frequency (17.1 months) and T790M negative (13.5 months).
Figure 3 shows a graph indicating the PFS of the subgroups of treatment-naive patients who are T790M positive with high allele frequency (median PFS: 7.7 months) compared with the subgroup of treatment-naxve patients who are T790M positive with low allele frequency or negative (13.6 months).
DESCRIPTION OF EMBODIMENTS
[0047] In the present invention, an epidermal growth factor receptor tyrosine kinase inhibitor (EGFR tyrosine kinase inhibitor or EGFR-TKI) may be selected from the compounds that are known in the art, as exemplified by gefitinib, erlotinib, icotinib, neratinib, afatinib, dacomitinib (PF00299804), osimertinib, rociletinib, WZ4002, EGF816, ASP8273, HM61713, PF06747775, and the like.
[0048] As an EGFR tyrosine kinase inhibitor, gefitinib or a pharm aceutically acceptable salt thereof may be used in the present invention. Gefitinib is an EGFR TKI drug that is approved for use in advanced NSCLC in a number of territories. The gefitinib structure is shown below:
[0049] Gefitinib may also be known by the chemical name N-(3-chloro-4-£luorophenyI)- 7-methoxy-6-(3-morpholinopropoxy)quinazolin-4-amine.
[0050] As an EGFR tyrosine kinase inhibitor, erlotinib or a pharmaceutically acceptable salt thereof may be used in the present invention. Erlotinib is an EGFR TKI drug used against pancreatic, lung and other cancers. The chemical structure of erlotinib is shown below:
[0051] Erlotinib may also be known by the chemical name: N - (3 -ethynylphenyl) -6,7- bis (2 - methoxyethoxy)quinazolin- 4- amine.
[0052] As an EGFR tyrosine kinase inhibitor, afatinib or a pharmaceutically acceptable salt thereof may be used in the present invention. Afatinib is an EGFR TKI drug used against NSCLC in particular. The drug can sometimes be used to treat cancers which are, or have become, resistant to treatment with gefitinib or erlotinib. The afatinib structure is shown below:
[0053] Afatinib may also be known by the chemical name (2E)-N-[4-[(3-chloro-4- fluorophenyl)amino] -7-[[(3S) -tetrahydro- 3-furanyljoxy] -6- quinazolinyl] -4- (dim ethylamino) - 2 -bufenamide .
[0054] As an EGFR tyrosine kinase inhibitor, osimertinib (AZD9291) may be used in the present invention. Osimertinib may sometimes be used to treat cancers which are, or have become, resistant to treatment with EGFR inhibitors such as gefitinib, erlotinib, and/or afatinib. Osimertinib has the following chemical structure:
[0055] Osimertinib may be known by the chemical name: N-(2-{2- (dimethylamino)ethyl-methyl-amino}-4-methoxy-5-{[4-(l-methylindol-3-yl)pyrimidin-2- yl] a mino]phenyl)prop -2 -enami de . Osimertinib and pharmaceutically acceptable salts thereof are disclosed in international patent application number PCT/GB2012/051783 (publication number WO2013/014448). A pharmaceutically acceptable salt of osimertinib may be any of the salts described herein, for example a mesylate salt.
[0056] As an EGFR tyrosine kinase inhibitor, CO-1686 or a pharmaceutically acceptable salt thereof can be used in the present invention. CO- 1686 may be also known as "rociletinib, and the chemical structure of CO-1686 is shown below:
[0057] Rociletinib may be known as CO-1686 or by the chemical name: N-(3-{[2-{[4-(4- acetylpip erazin-l-yl) -2 -methoxyphenyl] amino] - 5- (trifluorom ethyl)pyrimidin- 4 - yl]amino}phenyl)prop-2-enamide. A pharmaceutically acceptable salt of rociletinib may, for example, comprise an HBr salt.
[0058] As an EGFR tyrosine kinase inhibitor, EGF816 or a pharmaceutically acceptable salt thereof can be used in the present invention. The chemical structure of EGF816 is shown below:
[0059] EGF816 may be known by the chemical name: (i££)-N-(7-chloro-l-(l-(4-
(dimethylamino)but-2-enoyl)azepan-3-yl)-lHbenzo[d]imidazol-2-yl)-2- methylisonicotinamide.
[0060] As an EGFR tyrosine kinase inhibitor, WZ4002 can be used in the present invention. This compound is described in a publication by Zhou et al. (Nature 462,
1070-1074; 2009).
[0061] It is preferred that an approved EGFR-TKI is used in the present invention. WZ4002 (Bioorganic & Medicinal Chemistry Letters, Vol. 27, No. 21, 2017, 4832-4837), rociletinib (Lung Cancer (Auckl). 2016; 7: 91-97), EGF816 (WO 2016/185333 Al), HM61713 (J. Thorac. Oncol. 2016, ll(4):e45-7.), PF06747775 (Annals of Oncology (2017) 28 (suppl 5): v460-v496) and osimertinib are known herein as“third-generation EGFR- TKIs”. As used herein, the expression“first generation EGFR TKIs” includes erlotinib, gefitinib, icotinib and any combinations thereof. As used herein, the expression“second generation EGFR TKIs” includes afatinib, dacomitinib and any combinations thereof.
[0062] The EGFR-TKIs may be administered in accordance with dosage amount, administration route, pharmaceutical formulation, and dosage regimen in accordance
with the approval by regulatory authority.
[0063] In the present invention, the NSCLC includes advanced NSCLC or metastatic NSCLC. Advanced NSCLC refers to locally advanced or metastatic NSCLC. In advanced NSCLC, the NSCLC has spread to nearby tissue or to far away lymph nodes. Locally advanced NSCLC is defined as stage IIIB NSCLC not amenable to definitive multi- modality therapy including surgery. Metastatic NSCLC refers to stage IV NSCLC. In metastatic NSCLC, the cancer has spread to other organs of the body. In one embodiment of the subject invention, the NSCLC is inoperable or recurrent NSCLC. The progression of NSCLC may be monitored by methods well known to those in the art.
[0064] In one aspect of the present invention, the subject may have had treatment with a first- or second-generation EGFR TKI. The subject may suffer from NSCLC harboring EGFR activating mutation (e.g. L858R and/or exl9del) and/or an acquired EGFR T790M mutation. In the present invention, the NSCLC may be primary or recurrent.
[0065] In the subject specification, the term“treatment-naive patients” refers to patients suffering from NSCLC, who have not had previously received treatment with EGFR tyrosine kinase inhibitors. In one aspect of the present invention, the subject is a patient suffering from NSCLC, who has not had previously received treatment with EGFR tyrosine kinase inhibitors. The subject invention may be applied to patients, who have received a first diagnosis of primary or recurrent NSCLC.
[0066] Generally, EGFR mutation status may be determined by tests available in the art, e.g. QIAGEN therascreen™ EGFR test or other tests approved by regulatory authorities. The therascreen EGFR RGQ PCR Kit is an FDA- approved, qualitative real-time PCR assay for the detection of specific mutations in the EGFR oncogene. Evidence of EGFR mutation can be obtained from existing local data and testing of tumor samples. EGFR mutation status may be determined from any available tumor tissue, or from plasma samples.
[0067] In the present invention, allele frequency of mutation in EGFR may be confirmed by the procedure known in the art, including droplet digital PCR (ddPCR) technique. Analysis using ddPCR may be conducted by the procedures known in the art (e.g. M. Watanabe et al., Clin. Cancer Res. 2015; 21: 3552-3560). Allele frequency of the mutation may be calculated in the manner described in the subject specification. In one aspect of the subject invention, the allele frequency of T790M mutation in EGFR may be used as a criteria to determine the selection of treatment with EGFR TKIs In one embodiment, the selection is made based on the fact as to whether the tumor sample derived from the subject has T790M mutation in EGFR with allele frequency of more than about 0.30%, about 0.35%, about 0.40%, about 0.45%, or about 0.50%. As used herein, the term“about” when referring to any given numerical value means within
[0068] The anti-cancer treatment described hereinbefore may be applied as a sole therapy or may involve, in addition to the compound of the invention, conventional surgery or radiotherapy or chemotherapy or im munotherapy. Such chemotherapy could be administered concurrently, simultaneously, sequentially or separately to treatment with the compound of the invention and may include one or more of the following categories of anti-tumour agents :-
(i) antiproliferative/antineoplastic drugs and combinations thereof, as used in medical oncology, such as alkylating agents (for example cis-platin, oxaliplatin, carboplatin, cyclophosphamide, nitrogen mustard, melphalan, chlorambucil, busulphan, temozolamide and nitrosoureas); antimetabolites (for example gemcitabine and antifolates such as fhioropyrimi dines like 5-fluorouracil and tegafur, raltitrexed, methotrexate, cytosine arabinoside, and hydroxyurea); antitumour antibiotics (for example anthracyclines like adriamycin, bleomycin, doxorubicin, daunomycin, epirubicin, idarubicin, mitomycin- C, dactinomycin and mithramycin); antimitotic agents (for example vinca alkaloids like vincristine, vinblastine, vindesine and vinorelbine and taxoids like taxol and taxotere and polokinase inhibitors); and topoisomerase inhibitors (for example epipodophyllotoxins like etoposide and teniposide, amsacrine, topotecan and camptotheein);
(ii) cytostatic agents such as antioestrogens (for example tamoxifen, fulvestrant, toremifene, raloxifene, droloxifene and iodoxyfene), antiandrogens (for example bicalutamide, flutamide, nilutamide and cyproterone acetate), LHRH antagonists or LHRH agonists (for example goserelin, leuprorelin and buserelin), progestagens (for example megestrol acetate), aromatase inhibitors (for example as anastrozole, letrozole, vorazole and exemestane) and inhibitors of 5a-reductase such as finasteride;
(iii) anti-invasion agents [for example c-Src kinase family inhibitors like 4-(6-chloro- 2,3-methylenedioxyanilino)-7-[2-(4-methylpiperazin-l-yl)ethoxy]-5-tetrahydropyran-4- yloxyquinazoline [AZD0530 (saracatinib); WOOl/94341], N - (2 -chloro- 6 -methylphenyl) - 2 - {6-[4-(2-hydroxyethyl)piperazin-l-yl]-2-methylpyrimidin-4-ylamino}thiazole-6- carboxamide (dasatinib, BMS-354825; J. Med. Chem., 2004, 47, 6658-6661) and bosutinib (SKI-606), and metalloproteinase inhibitors like marimastat, inhibitors of urokinase plasminogen activator receptor function or antibodies to Heparanase];
(iv) inhibitors of growth factor function: for example such inhibitors include growth factor antibodies and growth factor receptor antibodies (for example the anti-erbB2 antibody trastuzumab [Herceptin™] , the anti-EGFR antibodies panitumumab or necitumumab, the anti-erbBl antibody cetuximab [Erbitux, C225] and any growth factor or growth factor receptor antibodies disclosed by Stern et al. Critical reviews in oncology/haematology, 2005, Vol. 54, ppll-29); inhibitors of the hepatocyte growth factor family; inhibitors of the insulin growth factor family; inhibitors of the platelet- derived
growth factor family such as imatinib and/or nilotinib (AMN107); inhibitors of serine/threonine kinases (for example Ras/Raf signalling inhibitors such as farnesyl transferase inhibitors, for example sorafenib (BAY 43-9006), tipifarnib (R115777) and lonafarnib (SCH66336)), inhibitors of cell signalling through MEK and/or AKT kinases (such as selumetinib), c-kit inhibitors, abl kinase inhibitors, PI3 kinase inhibitors, Plt3 kinase inhibitors, CSF-1R kinase inhibitors, IGF receptor (insulin -like growth factor) kinase inhibitors; aurora kinase inhibitors (for example AZD1152, PH739358, VX-680, MLN8054, R763, MP235, MP529, VX-528 AND AX39439); cyclin dependent kinase inhibitors such as CDK2 and/or CDK4 inhibitors (such as palbociclib); and MET inhibitors such as savolitinib;
(v) antiangio genic agents such as those which inhibit the effects of vascular endothelial growth factor, [for example the anti -vascular endothelial cell growth factor antibody bevacizumab (Avastin™) or ramucirinab) and for example, a VEGF receptor tyrosine kinase inhibitor such as vandetanib (ZD6474), vatalanib (PTK787), sunitinib (SU11248), axitinib (AG-013736), pazopanib (GW 786034) and 4- (4 -fluoro- 2 -methylindol- 5 -yloxy) - 6 -methoxy- 7 - (3-pyrrolidin- 1 -ylpropoxy)quinazoline (AZD2171; Example 240 within WO 00/47212), compounds such as those disclosed in W097/22596, W097/30035, W097/32856 and W098/13354 and compounds that work by other mechanisms (for example linomide, inhibitors of in te grin anb3 function and angiostatin)];
(vi) vascular damaging agents such as Combretastatin A4 and compounds disclosed in WO99/02166, WOOO/40529, WOOO/41669, WOO 1/92224, W002/04434 and
WO02/08213;
(vii) an endothelin receptor antagonist, for example zibotentan (ZD4054) or atrasentan; (viii) antisense therapies, for example those which are directed to the targets listed above, such as ISIS 2503, an anti-ras antisense;
(ix) gene therapy approaches, including for example approaches to replace aberrant genes such as aberrant p53 or aberrant BRCAl or BRCA2, GDEPT (gene-directed enzyme pro-drug therapy) approaches such as those using cytosine deaminase, thymidine kinase or a bacterial nitroreductase enzyme and approaches to increase patient tolerance to chemotherapy or radiotherapy such as multi-drug resistance gene therapy; and
(x) immunotherapy approaches, including for example ex-vivo and in-vivo approaches to increase the immunogenicity of patient tumour cells, such as transfection with cytokines such as interleukin 2, interleukin 4 or granulocyte-macrophage colony stimulating factor, approaches to decrease T-cell anergy, approaches using transfected immune cells such as cytokine-transfected dendritic cells, approaches using cytokine-transfected tumour cell lines, approaches using anti- idiotypic antibodies, approaches to decrease the function of immune suppressive cells such as regulatory T
cells, myeloid-derived suppressor cells or IDO (indoleamine 2,3,-deoxygenase)- expressing dendritic cells, and approaches using cancer vaccines consisting of proteins or peptides derived from tumour-associated antigens such as NY-ESO-1, MAGE-3, WTl or Her2/neu.
[0069] In the present invention, the EGFR TKIs or other drugs may he administered in the form of a pharmaceutically acceptable salt. The term“pharmaceutically acceptable salt” refers to a salt, which is non-toxic and is prepared with a pharm a ceutically acceptable base or acid. When the EGFR TKIs or other drugs are a basic compound, the pharmaceutically acceptable salt thereof is generally prepared by adding the compound with a suitable organic or inorganic acid. Examples of the acid- addition salt include acetate, benzenesulfonate, benzoate, citrate, fumarate, glutamate, hydrobromide, hydrochloride, lactate, maleate, mandelate, mesylate, oxalate, palmitate. phosphate/ diphosphate, salicylate, stearate, sulfate, succinate, tartrate and tosylate.
[0070] Furthermore, when the EGFR TKIs or other drugs are an acidic compound, the pharmaceutically acceptable salt thereof is generally prepared by adding the compound with a suitable organic or inorganic base.
[0071] The pharmaceutical composition may be administered as a pharmaceutical formulation. The pharmaceutical formulation may be produced by well-known methods, using an additive such as an excipient, a coating agent, a lubricant, a binder, a disintegrator, a stabilizer, a corrigent, a diluent, a solvent, a surfactant or an emulsifier.
[0072] Examples of excipients include starches such as potato starch and cornstarch, lactose, erythritol, mannitol, crystalline cellulose, microcrystalline cellulose, and calcium hydrogen phosphate.
[0073] Examples of coating agents include ethylcellulose, hydroxypropyleellulose. hydroxypropylmethylcellulose, shellac, talc, camauba wax, paraffin, and Opadry II™.
[0074] Examples of lubricants include magnesium stearate sodium stearyl fumarate, and stearic acid.
[0075] Examples of binders include polyvinylpyrrolidone, macrogol, and the same substances exemplified as the excipients.
[0076] Examples of disintegrators include the same substances exemplified as the excipients and low-substituted hydroxypropyl cellulose, croscarmellose sodium, sodium carboxymethylstarch, and chemically modified starch or celluloses.
[0077] Examples of stabilizers include p araoxyb enzoic acid esters such as methylparaben and propylparaben; alcohols such as chlorobutanol, benzylaleohol, and phenylethylaleohol; benzalkonium chloride; phenols such as phenol and cresol; thimerosal; dehydro acetic acid; and ascorbic acid.
[0078] Examples of corrigents include sweeteners which are conventionally used.
[0079] Examples of diluents include the substances exemplified as excipients.
[0080] Examples of solvents include ethanol, phenol, chlorocresol, purified water, and distilled water
[0081] Examples of surfactants or emulsifiers include polysorbate 80, polyoxyl 40 stearate, lauromacrogol, soybean oil, Maisin 35-1 (Gattefosse, France) and Cremophor
(BASF, Germany),
[0082] The dosage amount of the EGFR TKIs or other drugs differs depending on the symptoms, age, body weight, relative health condition, presence of other medication, method of administration, and the like.
[0083] An appropriate route of the administration of the EGFR TKIs or other drugs may be determined by a person skilled in the art. Examples thereof include oral, endorectal, parenteral (intravenous, intramuscular, or subcutaneous), intracisternal, vaginal, intraperitoneal, intravesical, topical (drip infusion, powder, ointment, gel, or cream) application, and inhalation (intraoral or nasal spray). The dosage form of the EGFR TKIs or other drugs for the administration includes tablets capsules, granules, powders, pills, aqueous and non-aqueous solutions or suspensions for oral use, and parenteral solutions loaded into containers. In one embodiment, the EGFR TKI is administered orally in an appropriate formulation including tablets.
EXAMPLES
[0084] Herein below, the present invention will be more specifically described using Examples, however, it is not to be construed as being limited thereto. The Study regarding allele frequency of T790M mutation in treatment-naive patients with NSCLC was conducted in the following procedures.
[0085] Test Example 1
[0086] Subjects
[0087] Formalin- Fixed Paraffin-Embedded (FFPE) specimens of patients suffering from NSCLC, who had not received treatment with EGFR-TKIs, were used. Patient characteristics are indicated in Table 1 In this study, switch of EGFR-TKI due to an adverse event is considered as continuation of therapy with EGFR-TKI. In such a case, progression-free survival (PFS) is defined to be a period from start of the treatment of the first-line EGFR-TKI to disease progression during the subsequent EGFR-TKI treatment following the switch.
Table 1
[0088] Sample collection
[0089] Four 10-mpi sections were cut from each FFPE specimens. Genomic DNA was extracted from the FFPE specimens with the QIAmp DNA FFPE tissue kit (Qiagen) in accordance with the manufacturer’s instructions. Quality and quantity of the genomic DNA sample were assessed using NanoDrop 2000 (Life Technologies) and Qubit 2.0 (Life Technologies) platforms.
[0090] Probes and primers for droplet digital PCR (ddPCR)
[0091] Primers and probes were procured from Integrated DNA Technologies K.K. (IDT K.K.). Fluorescent probes targeting wild-type and mutant sequences were respectively conjugated to tetrachlorofluorescein (TET, Aex 522 nm/Aem 539 nm) or 6- carboxyfluorescein (FAM, Aex 494 nm/Aem 522 nm) fluorophores with ZEN/IABkFQ double quencher. The sequences of primers and probes for detection of the T790M mutation are shown in Table 2.
Table 2:
[0092] Droplet Digital PCR
[0093] Analysis by droplet digital PCT technique was conducted in the procedures described in Non-patent document 3 (M. Watanabe et al, Clin. Cancer Res. 2015; 21: 3552-3560), Briefly, the duplex assay is based on the parallel amplification of wild-type and specific mutant sequences. In a pre-PCR environment, 20.0 pL TaqMan Genotyping Master Mix (Life Technologies) was mixed with the assay solution containing 2.0 pL (10 pM) forward and reverse primers, 2.0 pL (10 pM) of FAM and TET labeled- probes, 1.6 pL Droplet Stabilizer (RainDance Technologies), 2.4 pL sterile DNase- and RNase-free water (Life Technologies), and 8 pL genomic DNA from patients to a final reaction volume of 40 pL to prepare a template. The preparation of the template is summarized in Table 3.
Table 3:
[0094] A collection of uniformly sized aqueous droplets was produced by hydrodynamic flow-focusing with a droplet-generating microfluidic chip (Source chip, RainDance
Technologies) in accordance with the manufacturer’s instructions. One lane of the Source chip was used for positive control. The resulting emulsion was collected in a PCR tube strip comprising eight 0.2-mL conical-bottom PCR tubes. The PCR tube strip, containing a total of 75 pL of droplets and carrier oil, was tightly capped with an 8-Strip Dome Cap, and then placed in a thermal cycler with a hot lid (Proflex PCR system, Life Technologies). The emulsion was subjected to thermal cycling, namely starting with a hot start denaturation step of 10 min at 95 °C, followed by 45 cycles of: 95 °C, 15 sec; 58 °C, 15 sec; 60 °C, 45 sec (using a 0.5 °C/min ramp rate). These cycles were followed by a final step at 98 °C for 10 min and then at 12 °C for 10 min. The thermal cycling condition is summarized in Table 4.
[0095] The emulsions were processed immediately to measure the endpoint fluorescence signal from each droplet. Namely the thermal-cycled emulsion was transferred into a second microfluidic chip (Sense chip, RainDance Technologies), and then endpoint fluorescence signals were measured in accordance with the manufacturer’s instructions.
[0096] Data analysis and Determination of cut-off value for positive events
[0097] The droplet event data were analyzed with the RainDrop Analyst software (RainDance Technologies) in accordance with the manufacturer’s instructions. Briefly, sample data were loaded with a drop size gating template (RainDance Technologies). Data from the positive control sample were used to create the compensation matrix in the RainDrop Analyst software. The compensation matrix was applied to the data from each sample to eliminate the crosstalk fluorescence signals from the TET and FAM fluorophores. The sizes and locations of the wild-type and the mutant gates were established by manual selection of the area containing wild-type or mutant clusters in the positive control.
[0098] For each unknown sample, the number of PCR-positive droplet events was counted within each gate. The number of events within each gate was converted to the number of events per assay using the total number of intact drops.
[0099] In this study, another twenty-one FFPE samples were prepared as negative controls to determine cut-off values for positive events. Negative control samples included eleven samples of lung squamous cell carcinoma and nine samples of adenocarcinoma . which had been confirmed as EGFR wild-type and one normal tissue. Assay of ddPCR was also performed on these negative control samples and the number of false-positive droplet events were measured. The raw data for limit of blank (LOB) analysis is shown in Table 5. The LOB was determined by evaluating the 95% and 99% one-tailed upper limit of the model distribution. The number of false events of mutant droplets detected per analysis is 23 for 95% upper limit and 26 for 99% upper limit. Thus, assays were considered "positive" if the number of the measured event was >30 events/assay and considered "negative" if the event within a gated region was <30 events/assay.
[0100] Determination of best cut-off value for allele frequency of T790M in treatment- naive patients
[0101] If an assay result indicates T790M mutation positive in light of the foregoing, raw mutant allele frequency (MAF) was calculated as follows:
[0103] The term“tumor content” means ratio of an area of tumor in a section to a total area of a section.
[0104] To determine the cut-off value of MAF of T790M in treatment- naive patients (also referred to as“pretreatment T790M MAF”) for the“poor responder,” a receiver operating characteristic curve analysis was performed. Poor responders is defined as patients who had disease progression for the first-line EGFR-TKIs within 12 months (i.e. PFS < 12 months). This is based on the median progression free survival of 11.7 months for all patients in this study. The area under the curve for the corrected MAF was 0.889 (95% Cl: 0.724-1) and the cut-off value, for which sum of sensitivity plus specificity was maximal, was 0.34% (91.7% sensitivity and 83.3% specificity, see Fig. 1). Thus, the subgroup with the corrected pretreatment T790M MAF >0.3% was considered to have high frequency of the mutant allele.
[0105] The patients were stratified with the corrected pretreatment T790M MAF. The patients with more than 30 events of the mutation per assay is defined as positive (i.e. cut-off value: >30 events/assay), and overall incidence of T790M mutation in treatment -naive patents is 40.9% (18/44). High frequency of the mutant allele is found in 12 patients, and low frequency is found in 6 patients. PFS of each subgroup is indicated in Figs. 2 and 3. The results of Fig. 2 indicates that the subgroups of treatment-naive patients who are T790M positive with high frequency had a shorter median PFS (7.7 months) than the subgroups of treatment-naive patients who are T790M positive with low frequency (17.1 months) and T790M negative (13.5 months). The results of Fig. 3 indicates the PFS of the subgroups of treatment- naive patients who are T790M positive with high frequency (median PFS: 7.7 months) compared with the subgroup of treatment-naive patients who are T790M positive with low frequency or negative (13.6 months).
[0106] This study indicates that more than 0.3% of T790M allele mutation frequency in treatment-naive patents is an adverse factor for PFS in the treatment with EGFR-
TKIs. Namely, the T790M allele frequency can be used for determining a selection of an EGFR-TKI for treating NSCLC. In particular, treatment-naive patients who exhibit more than 0.3% of T790M allele mutation frequency are unlikely to show an optimal response to first- or second- generation EGFR-TKIs, and such patients are more likely to respond more favorably to early administration of third-generation EGFR-TKIs, such as osimertinib.
Claims
1. A pharmaceutical composition comprising an effective amount of an EGFE tyrosine kinase inhibitor, for use in a method of treating a subject suffering from non small cell lung cancer (NSCLC),
wherein the method comprises:
preparing a sample derived from the cancer;
analyzing the sample to confirm allele frequency of T790M mutation in EGFR; confirming whether the subject has T790M mutation in EGFR with allele frequency of more than about 0.3%; and
determining the selection of an EGFR tyrosine kinase inhibitor for administration to the subject based on the allele frequency.
2. The pharmaceutical composition according to claim 1, wherein the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotmib, afatinib, osimertinib, rociletinib, WZ4002 and EGF816, and a pharmaceutically acceptable salt thereof, in particular the EGFR tyrosine kinase inhibitor is selected from gefitinib, erlotmib, afatinib and osimertinib, and a pharmaceutically acceptable salt thereof.
3. The pharmaceutical composition according to claim 1 or 2, wherein a third- generation EGFR TKI or a pharmaceutically acceptable salt thereof is selected to be administered to a subject having T790M mutation in EGFR with allele frequency of more than about 0.3% in the selection of an EGFR tyrosine kinase inhibitor.
4. A pharmaceutical composition comprising an effective amount of a third- generation EGFR TKI or a pharmaceutically acceptable salt thereof, for use in a method of treating a subject suffering from non-small cell lung cancer (NSCLC),
wherein the method comprises:
preparing a sample of the cancer derived from the subject;
analyzing the sample to confirm allele frequency of T790M mutation in EGFR; selecting a subject having T790M mutation in EGFR with allele frequency of more than about 0.3%; and
administering the composition to the selected subject.
5. A pharmaceutical composition comprising an effective amount of a third- generation EGFR-TKI or a pharmaceutically acceptable salt thereof, for use in a method of treating a subject suffering from non-small cell lung cancer (NSCLC), wherein a sample of the cancer derived from the said subject has been analyzed and wherein the allele frequency of T790M mutation in EGFR has been confirmed,
wherein the method comprises administration of the composition to the subject where the allele frequency of T790M mutation in EGFR is more than about 0.3%.
6. The pharmaceutical composition according to any one of claims 3 to 5, wherein the third-generation EGFR-TKI is selected from osimertinib, rociletinib, WZ4G02 and EGF816, and a pharmaceutically acceptable salt thereof.
7. The pharmaceutical composition according to claim 6, wherein the third- generation EGFR-TKI is osimertinib or a pharmaceutically acceptable salt thereof.
8. The pharmaceutical composition according to any one of claims 1 to 7. wherein the subject is a patient, who has not previously received treatment with EGFR tyrosine kinase inhibitors.
9. The pharmaceutical composition according to any one of claims 1 to 7, wherein the subject is a patient, who previously received treatment with an EGFR tyrosine kinase inhibitor other than osimertinib or a pharmaceutically acceptable salt thereof, and wherein the EGFR tyrosine kinase inhibitor previously used is changed to osimertinib or a pharm aceutically acceptable salt thereof.
10. The pharmaceutical composition according to any one of claims 1 to 9, wherein the frequency of T790M mutation in EGFR is confirmed by an assay using droplet digital PCR.
11. The pharmaceutical composition according to claim 10, wherein T790M mutation in EGFR of a patient is determined as negative in a case that the number of PCR-positive droplet events is 30 or less per assay.
12. The pharmaceutical composition according to any one of claims 1 to 11, wherein the subject has one or more mutations in EGFR other than T790M.
13. The pharmaceutical composition according to any one of claims 1 to 12, wherein the mutation in EGFR is selected from Exon 19 del and Exon21 L858R.
14. The pharmaceutical composition according to any one of claims 1 to 13, which is administered to a subject in combination with one or more other anti-cancer agents.
15. The pharmaceutical composition according to any one of claims 1 to 14, wherein
the NSCLC is inoperable or recurrent NSCLC.
16. The pharmaceutical composition according to any one of claims 1 to 15, wherein the EGFR tyrosine kinase inhibitor that is administered to the subject is osimertinib mesylate.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018043943 | 2018-03-12 | ||
| JP2018-043943 | 2018-03-12 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019175093A1 true WO2019175093A1 (en) | 2019-09-19 |
Family
ID=65911112
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2019/056002 Ceased WO2019175093A1 (en) | 2018-03-12 | 2019-03-11 | Method for treating lung cancer |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2019175093A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114980883A (en) * | 2020-01-20 | 2022-08-30 | 阿斯利康(瑞典)有限公司 | Epidermal growth factor receptor tyrosine kinase inhibitors for the treatment of cancer |
Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997022596A1 (en) | 1995-12-18 | 1997-06-26 | Zeneca Limited | Quinazoline derivatives |
| WO1997030035A1 (en) | 1996-02-13 | 1997-08-21 | Zeneca Limited | Quinazoline derivatives as vegf inhibitors |
| WO1997032856A1 (en) | 1996-03-05 | 1997-09-12 | Zeneca Limited | 4-anilinoquinazoline derivatives |
| WO1998013354A1 (en) | 1996-09-25 | 1998-04-02 | Zeneca Limited | Quinazoline derivatives and pharmaceutical compositions containing them |
| WO1999002166A1 (en) | 1997-07-08 | 1999-01-21 | Angiogene Pharmaceuticals Ltd. | Use of colchinol derivatives as vascular damaging agents |
| WO2000040529A1 (en) | 1999-01-07 | 2000-07-13 | Angiogene Pharmaceuticals Ltd. | Colchinol derivatives as vascular damaging agents |
| WO2000041669A2 (en) | 1999-01-15 | 2000-07-20 | Angiogene Pharmaceuticals Ltd. | Benzimidazole vascular damaging agents |
| WO2000047212A1 (en) | 1999-02-10 | 2000-08-17 | Astrazeneca Ab | Quinazoline derivatives as angiogenesis inhibitors |
| WO2001092224A1 (en) | 2000-05-31 | 2001-12-06 | Astrazeneca Ab | Indole derivatives with vascular damaging activity |
| WO2001094341A1 (en) | 2000-06-06 | 2001-12-13 | Astrazeneca Ab | Quinazoline derivatives for the treatment of tumours |
| WO2002004434A1 (en) | 2000-07-07 | 2002-01-17 | Angiogene Pharmaceuticals Limited | Colchinol derivatives as vascular damaging agents |
| WO2002008213A1 (en) | 2000-07-07 | 2002-01-31 | Angiogene Pharmaceuticals Limited | Colchinol derivatives as angiogenesis inhibitors |
| US20130017540A1 (en) * | 2011-06-07 | 2013-01-17 | Yun Yen | Identification of mutation types associated with acquired resistance and methods for using same |
| WO2013014448A1 (en) | 2011-07-27 | 2013-01-31 | Astrazeneca Ab | 2 - (2, 4, 5 - substituted -anilino) pyrimidine derivatives as egfr modulators useful for treating cancer |
| WO2014201092A1 (en) * | 2013-06-11 | 2014-12-18 | Dana-Farber Cancer Institute, Inc. | Non-invasive blood based monitoring of genomic alterations in cancer |
| WO2016141324A2 (en) * | 2015-03-05 | 2016-09-09 | Trovagene, Inc. | Early assessment of mechanism of action and efficacy of anti-cancer therapies using molecular markers in bodily fluids |
| WO2016185333A1 (en) | 2015-05-15 | 2016-11-24 | Novartis Ag | Methods for treating egfr mutant cancers |
-
2019
- 2019-03-11 WO PCT/EP2019/056002 patent/WO2019175093A1/en not_active Ceased
Patent Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997022596A1 (en) | 1995-12-18 | 1997-06-26 | Zeneca Limited | Quinazoline derivatives |
| WO1997030035A1 (en) | 1996-02-13 | 1997-08-21 | Zeneca Limited | Quinazoline derivatives as vegf inhibitors |
| WO1997032856A1 (en) | 1996-03-05 | 1997-09-12 | Zeneca Limited | 4-anilinoquinazoline derivatives |
| WO1998013354A1 (en) | 1996-09-25 | 1998-04-02 | Zeneca Limited | Quinazoline derivatives and pharmaceutical compositions containing them |
| WO1999002166A1 (en) | 1997-07-08 | 1999-01-21 | Angiogene Pharmaceuticals Ltd. | Use of colchinol derivatives as vascular damaging agents |
| WO2000040529A1 (en) | 1999-01-07 | 2000-07-13 | Angiogene Pharmaceuticals Ltd. | Colchinol derivatives as vascular damaging agents |
| WO2000041669A2 (en) | 1999-01-15 | 2000-07-20 | Angiogene Pharmaceuticals Ltd. | Benzimidazole vascular damaging agents |
| WO2000047212A1 (en) | 1999-02-10 | 2000-08-17 | Astrazeneca Ab | Quinazoline derivatives as angiogenesis inhibitors |
| WO2001092224A1 (en) | 2000-05-31 | 2001-12-06 | Astrazeneca Ab | Indole derivatives with vascular damaging activity |
| WO2001094341A1 (en) | 2000-06-06 | 2001-12-13 | Astrazeneca Ab | Quinazoline derivatives for the treatment of tumours |
| WO2002004434A1 (en) | 2000-07-07 | 2002-01-17 | Angiogene Pharmaceuticals Limited | Colchinol derivatives as vascular damaging agents |
| WO2002008213A1 (en) | 2000-07-07 | 2002-01-31 | Angiogene Pharmaceuticals Limited | Colchinol derivatives as angiogenesis inhibitors |
| US20130017540A1 (en) * | 2011-06-07 | 2013-01-17 | Yun Yen | Identification of mutation types associated with acquired resistance and methods for using same |
| WO2013014448A1 (en) | 2011-07-27 | 2013-01-31 | Astrazeneca Ab | 2 - (2, 4, 5 - substituted -anilino) pyrimidine derivatives as egfr modulators useful for treating cancer |
| WO2014201092A1 (en) * | 2013-06-11 | 2014-12-18 | Dana-Farber Cancer Institute, Inc. | Non-invasive blood based monitoring of genomic alterations in cancer |
| WO2016141324A2 (en) * | 2015-03-05 | 2016-09-09 | Trovagene, Inc. | Early assessment of mechanism of action and efficacy of anti-cancer therapies using molecular markers in bodily fluids |
| WO2016185333A1 (en) | 2015-05-15 | 2016-11-24 | Novartis Ag | Methods for treating egfr mutant cancers |
Non-Patent Citations (10)
| Title |
|---|
| ANNALS OF ONCOLOGY, vol. 28, no. 5, 2017, pages v460 - v496 |
| BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 27, no. 21, 2017, pages 4832 - 4837 |
| CORTOT ET AL., EUR. RESPIR. REV., vol. 23, 2014, pages 356 - 366 |
| J. MED. CHEM., vol. 47, 2004, pages 6658 - 6661 |
| J. THORAC. ONCOL., vol. ll, no. 4, 2016, pages e45 - 7 |
| LUNG CANCER (AUCKL, vol. 7, 2016, pages 91 - 97 |
| M. WATANABE ET AL., CLIN. CANCER RES., vol. 21, 2015, pages 3552 - 3560 |
| STERN ET AL., CRITICAL REVIEWS IN ONCOLOGY/HAEMATOLOGY, vol. 54, 2005, pages ll-29 |
| T.S. MOK ET AL., N. ENGL. J. MED., vol. 376, 2017, pages 629 - 640 |
| ZHOU ET AL., NATURE, vol. 462, 2009, pages 1070 - 1074 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114980883A (en) * | 2020-01-20 | 2022-08-30 | 阿斯利康(瑞典)有限公司 | Epidermal growth factor receptor tyrosine kinase inhibitors for the treatment of cancer |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8877764B2 (en) | Method for treating cancer harboring EGFR mutations | |
| JP2022009090A (en) | Dosing of kras inhibitor for treatment of cancers | |
| ES2966512T3 (en) | Crystalline forms of (S)-1-(4-fluorophenyl)-1-(2-(4-(6-(1-methyl-1H-pyrazol-4-yl)pyrrolo[2,1-f][1, 2,4]triazin-4-yl)piperazinyl)-pyrimidin-5-yl)ethane-1-amine and methods of preparation | |
| JP5778735B2 (en) | Use of pyrimidine derivatives to treat EGFR-dependent diseases or diseases that have acquired resistance to drugs targeting EGFR family members | |
| US20130012465A1 (en) | Bibw 2992 for use in the treatment of triple negative breast cancer | |
| JP6571105B2 (en) | Pharmaceutical formulations of pan-RAF kinase inhibitors, processes for their preparation and methods of use | |
| JPWO2003101491A1 (en) | Prophylactic and / or therapeutic agent for Her2 or / and EGFR expression or activation target | |
| US20100004247A1 (en) | Combination comprising a mek inhibitor and an aurora kinase inhibitor 188 | |
| AU2022358413A1 (en) | Combination therapy using substituted pyrimidin-4(3h)-ones and sotorasib | |
| CN101516376A (en) | Methods for treating cancers carrying EGFR mutations | |
| WO2019175093A1 (en) | Method for treating lung cancer | |
| CN1960733A (en) | Combination products comprising the SRC kinase inhibitor AZD0530 and an antiestrogen or EGFR-TK inhibitor | |
| Sun et al. | Systemic therapy for mutation-driven NSCLC | |
| TW202406552A (en) | Egfr inhibitors for treatment of cancer | |
| CA3230424A1 (en) | Methods of treating cancer | |
| EP4504202B1 (en) | Combination therapies comprising gdc-6036 and gdc-0077 for the treatment of cancer | |
| US20240116880A1 (en) | Certain chemical entities, compositions, and methods | |
| HK40118757A (en) | Combination therapies comprising gdc-6036 and gdc-0077 for the treatment of cancer | |
| JP2025115258A (en) | Method for predicting therapeutic effect or prognosis, therapeutic drug, kit, or biomarker | |
| TW202313056A (en) | PHARMACEUTICAL COMBINATIONS COMPRISING A PI3K <i>α</i> INHIBITOR | |
| WO2023209611A1 (en) | Methods of treating cancer with a b-raf inhibitor, in particular lifirafenib | |
| CN118043049A (en) | Sotolacizumab and EGFR antibodies for the treatment of cancers harboring the KRAS G12C mutation | |
| HK40111140A (en) | Sotorasib and an egfr antibody for treating cancer comprising a kras g12c mutation | |
| WO2025217209A2 (en) | Crystalline forms of hdac inhibitor and uses thereof | |
| KR20250092216A (en) | How to cure cancer with sotorasib |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19713367 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19713367 Country of ref document: EP Kind code of ref document: A1 |