WO2019152688A1 - Spiro-lactam nmda receptor modulators and uses thereof - Google Patents
Spiro-lactam nmda receptor modulators and uses thereof Download PDFInfo
- Publication number
- WO2019152688A1 WO2019152688A1 PCT/US2019/016114 US2019016114W WO2019152688A1 WO 2019152688 A1 WO2019152688 A1 WO 2019152688A1 US 2019016114 W US2019016114 W US 2019016114W WO 2019152688 A1 WO2019152688 A1 WO 2019152688A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- alkyl
- group
- independently selected
- phenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC(CCCCCN1*)CCC1=O Chemical compound CC(CCCCCN1*)CCC1=O 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/54—Spiro-condensed
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
Definitions
- NMDA N-methyl-d-aspartate
- the NMDA receptor controls the flow of both divalent and monovalent ions into the postsynaptic neural cell through a receptor associated channel (Foster et al,
- NMDA receptor has been implicated during development in specifying neuronal architecture and synaptic connectivity, and may be involved in experience-dependent synaptic
- NMDA receptors are also thought to be involved in long term potentiation and central nervous system disorders.
- the NMDA receptor plays a major role in the synaptic plasticity that underlies many higher cognitive functions, such as memory acquisition, retention and learning, as well as in certain cognitive pathways and in the perception of pain (Collingridge et al. , The NMDA Receptor, Oxford University Press, 1994). In addition, certain properties of NMDA receptors suggest that they may be involved in the information-processing in the brain that underlies consciousness itself.
- the NMDA receptor has drawn particular interest since it appears to be involved in a broad spectrum of CNS disorders. For instance, during brain ischemia caused by stroke or traumatic injury, excessive amounts of the excitatory amino acid glutamate are released from damaged or oxygen deprived neurons. This excess glutamate binds to the NMDA receptors which opens their ligand-gated ion channels; in turn the calcium influx produces a high level of intracellular calcium which activates a biochemical cascade resulting in protein degradation and cell death. This phenomenon, known as excitotoxicity, is also thought to be responsible for the neurological damage associated with other disorders ranging from hypoglycemia and cardiac arrest to epilepsy.
- the NMDA receptor is believed to consist of several protein chains embedded in the postsynaptic membrane.
- the first two types of subunits discovered so far form a large extracellular region, which probably contains most of the allosteric binding sites, several transmembrane regions looped and folded so as to form a pore or channel, which is permeable to Ca ++ , and a carboxyl terminal region.
- the opening and closing of the channel is regulated by the binding of various ligands to domains (allosteric sites) of the protein residing on the extracellular surface.
- the binding of the ligands is thought to affect a conformational change in the overall structure of the protein which is ultimately reflected in the channel opening, partially opening, partially closing, or closing.
- the present disclosure includes compounds that can be NMDA modulators. More specifically, the present disclosure provides a compound represented by Formula I:
- R 5 is independently, for each occurrence, one or more optional substituents selected from the group consisting of Ci-C 6 alkyl, -S(0) w -Ci_C 3 alkyl, -NR a R b , and halogen, wherein Ci- C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s ;
- R 6 is one or two optional substituents bound to one or more available ring carbons, and is independently, for each occurrence, selected from the group consisting of Ci-Cealkyl, - S(0) w -Ci-C 3 alkyl, -NR a R b , and halogen, wherein Ci-C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s ;
- w 0, 1, or 2;
- R 3 is selected from the group consisting of H, phenyl, Ci-Cealkyl, -C(0)R 31 , and - C(0)0R 32 , wherein Ci-C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s ;
- R 31 and R 32 are each independently selected from the group consisting of H, Ci-Cealkyl, -C3-C6cycloalkyl, and phenyl, wherein Ci-C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s , and phenyl is optionally substituted with one, two, or three substitutents each independently selected from R T ;
- R a and R b are independently, for each occurrence, selected from the group consisting of H, phenyl, Ci-C 4 alkyl, -Ci-C4alkylene-phenyl, or R a and R b taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted with one, two, or three substitutents each independently selected from hydroxyl, - Ci-C3alkoxy, -Ci-C3alkyl, and halogen, and Ci-C3alkyl is optionally substituted with one, two or three halogens;
- R s is independently, for each occurrence, selected from the group consisting of - C(0)NR a R b , -NR a R b , hydroxyl, S(0) w -Ci-C 3 alkyl, SH, phenyl and halogen; and
- R T is independently, for each occurrence, selected from the group consisting of -Ci-Cealkyl, hydroxyl, Ci-C 6 alkoxy, and halogen.
- R 3 is selected from the group consisting of H, -C( O )-C
- R a and R b are each independently for each occurrence selected from the group consisting of H, phenyl, Ci-C 4 alkyl, -Ci-C 4 alkylene -phenyl, or R a and R b taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted by one two or three substituents each independently selected from Ci- C 3 alkoxy, Ci-C 3 alkyl, and halogen, wherein Ci-C 3 alkyl is optionally substituted with one, two or three substituents independently selected from F, Cl, and Br.
- compositions comprising a disclosed compound, and a pharmaceutically acceptable excipient.
- Such compositions can be suitable for administration to a patient orally, parenterally, topically, intravaginally, intrarectally, sublingually, ocularly, transdermally, or nasally.
- Such methods may comprise administering to a patient a therapeutically effective amount of a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof, or a pharmaceutical composition including a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof.
- a method of this disclosure includes treating depression. In some embodiments, a method of this disclosure includes treating schizophrenia. In certain embodiments, a method of this disclosure includes treating Alzheimer’s disease. In various embodiments, a method of this disclosure includes treating attention deficit disorder. In some embodiments, a method of this disclosure includes treating anxiety. In certain embodiments, a method of this disclosure includes treating a migraine. In various embodiments, a method of this disclosure includes treating neuropathic pain. In some embodiments, a method of this disclosure includes treating traumatic brain injury. In certain embodiments, a method of this disclosure includes treating a neurodevelopment disorder related to a synaptic dysfunction. In various embodiments, a method of this disclosure includes treating a cognitive impairment disorder.
- Such methods may comprise administering to a patient a therapeutically effective amount of a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof, or a pharmaceutical composition including a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof.
- This disclosure is generally directed to compounds that are capable of modulating NMDA receptors, for example, NMDA receptor antagonists, agonists, or partial agonists, and compositions and/or methods of using the disclosed compounds.
- compounds described herein bind to NMDA receptors expressing certain NR2 subtypes.
- the compounds described herein bind to one NR2 subtype and not another. It should be appreciated that the disclosed compounds may modulate other protein targets and/or specific NMDA receptor subtype.
- alkyl refers to a saturated straight-chain or branched hydrocarbon, such as a straight-chain or branched group of 1-6, 1-4, or 1-3 carbon atoms, referred to herein as -Ce alkyl, C 1 -C 4 alkyl, and C 1 -C 3 alkyl, respectively.
- -Ce alkyl C 1 -C 4 alkyl
- C 1 -C 3 alkyl C 1 -C 3 alkyl
- Examples of a C 1 -C 6 alkyl group include, but are not limited to, methyl, ethyl, propyl, butyl, pentyl, hexyl, isopropyl, isobutyl, sec-butyl, ieri-butyl, isopentyl, and neopentyl.
- “C 1 -C 4 alkyl” refers to a straight-chain or branched saturated hydrocarbon containing 1-4 carbon atoms.
- Examples of a C 1 -C 4 alkyl group include, but are not limited to, methyl, ethyl, propyl, butyl, isopropyl, isobutyl, sec-butyl and ieri-butyl.
- Exemplary alkyl groups include, but are not limited to, methyl, ethyl, propyl, isopropyl, 2-methyl- 1 -propyl, 2- methyl-2-propyl, 2-methyl- l-butyl, 3 -methyl- 1 -butyl, 3-methyl-2-butyl, 2,2-dimethyl- l-propyl, 2-methyl- 1 -pentyl, 3-methyl- 1 -pentyl, 4-methyl- 1 -pentyl, 2-methyl-2-pentyl, 3-methyl-2- pentyl, 4-methyl-2-pentyl, 2,2-dimethyl- 1 -butyl, 3,3-dimethyl-l-butyl, 2-ethyl- 1 -butyl, butyl, isobutyl, t-butyl, pentyl, isopentyl, neopentyl, and hexyl.
- alkoxy refers to an alkyl group attached to an oxygen atom (alkyl-O-).
- Alkoxy groups can have 1-6 or 2-6 carbon atoms and are referred to herein as Ci- C 6 alkoxy and C 2 -Cr, alkoxy, respectively.
- Exemplary alkoxy groups include, but are not limited to, methoxy, ethoxy, propyloxy, isopropoxy, and tert-butoxy.
- carbocyclic ring refers to a hydrocarbon ring system in which all the ring atoms are carbon.
- exemplary carbocyclic rings including cycloalkyls and phenyl.
- cycloalkyl refers to a monocyclic saturated or partially unsaturated hydrocarbon ring (carbocyclic) system, for example, where each ring is either completely saturated or contains one or more units of unsaturation, but where no ring is aromatic.
- a cycloalkyl can have 3-6 or 4-6 carbon atoms in its ring system, referred to herein as C3-C6 cycloalkyl or C 4 -C 6 cycloalkyl, respectively.
- Exemplary cycloalkyl groups include, but are not limited to, cyclohexyl, cyclohexenyl, cyclopentyl, cyclopentenyl, cyclobutyl, and cyclopropyl.
- halo and“halogen,” as used herein, refer to fluoro (F), chloro (Cl), bromo (Br), and/or iodo (I).
- heteroatom refers to an atom of any element other than carbon or hydrogen and includes, for example, nitrogen (N), oxygen (O), silicon (Si), sulfur (S), phosphorus (P), and selenium (Se).
- heterocycloalkyl or“heterocyclic ring” as used herein, is art-recognized and refer to saturated or partially unsaturated 3- to 8-membered ring systems, for example, 3- to 7- or 3- to 6-membered ring systems, whose ring system include one, two or three heteroatoms, such as nitrogen, oxygen, and/or sulfur.
- a heterocycloalkyl can be fused to one or more phenyl, partially unsaturated, or saturated rings. Examples of heterocycloalkyls include, but are not limited to, pyrrolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, and piperazinyl.
- amino acid includes any one of the following alpha amino acids: isoleucine, alanine, leucine, asparagine, lysine, aspartate, methionine, cysteine, phenylalanine, glutamate, threonine, glutamine, tryptophan, glycine, valine, proline, arginine, serine, histidine, and tyrosine.
- An amino acid also can include other art-recognized amino acids such as beta amino acids.
- a compound can refer to a pharmaceutically acceptable salt, or a hydrate, or an N-oxide of a stereoisomer of the compound and/or an isotopically-labelled compound.
- moiety refers to a portion of a compound or molecule.
- the compounds of the disclosure can contain one or more chiral centers and/or double bonds and therefore, can exist as stereoisomers, such as geometric isomers, and enantiomers or diastereomers.
- stereoisomers when used herein, consists of all geometric isomers, enantiomers and/or diastereomers of the compound.
- the compound depicted without such chirality at that and other chiral centers of the compound are within the scope of the present disclosure, i.e., the compound depicted in two-dimensions with“flat” or“straight” bonds rather than in three dimensions, for example, with solid or dashed wedge bonds.
- Stereospecific compounds may be designated by the symbols“R” or“S,” depending on the configuration of substituents around the stereogenic carbon atom.
- the present disclosure encompasses all the various stereoisomers of these compounds and mixtures thereof.
- Mixtures of enantiomers or diastereomers can be designated“( ⁇ )” in nomenclature, but a skilled artisan will recognize that a structure can denote a chiral center implicitly. It is understood that graphical depictions of chemical structures, e.g., generic chemical structures, encompass all stereoisomeric forms of the specified compounds, unless indicated otherwise.
- Individual enantiomers and diastereomers of compounds of the present disclosure can be prepared synthetically from commercially available starting materials that contain asymmetric or stereogenic centers, or by preparation of racemic mixtures followed by resolution methods well known to those of ordinary skill in the art. These methods of resolution are exemplified by (1) attachment of a mixture of enantiomers to a chiral auxiliary, separation of the resulting mixture of diastereomers by recrystallization or chromatography and liberation of the optically pure product from the auxiliary, (2) salt formation employing an optically active resolving agent, (3) direct separation of the mixture of optical enantiomers on chiral liquid chromatographic columns, or (4) kinetic resolution using stereoselective chemical or enzymatic reagents.
- Racemic mixtures also can be resolved into their component enantiomers by well-known methods, such as chiral-phase gas chromatography or crystallizing the compound in a chiral solvent.
- Stereoselective syntheses a chemical or enzymatic reaction in which a single reactant forms an unequal mixture of stereoisomers during the creation of a new stereocenter or during the transformation of a pre-existing one, are well known in the art.
- Stereoselective syntheses encompass both enantio- and diastereoselective transformations.
- Geometric isomers resulting from the arrangement of substituents around a carbon- carbon double bond or arrangement of substituents around a cycloalkyl or heterocycloalkyl, can also exist in the compounds of the present disclosure.
- the symbol . denotes a bond that may be a single, double or triple bond as described herein.
- Substituents around a carbon-carbon double bond are designated as being in the“Z’ or“E” configuration, where the terms“Z’ and “£” are used in accordance with IUPAC standards. Unless otherwise specified, structures depicting double bonds encompass both the“E” and“Z” isomers.
- Substituents around a carbon-carbon double bond alternatively can be referred to as “cis” or“trans,” where“cis” represents substituents on the same side of the double bond and “trans” represents substituents on opposite sides of the double bond.
- the arrangement of substituents around a carbocyclic ring can also be designated as“cis” or“trans.”
- the term “cis” represents substituents on the same side of the plane of the ring and the term“trans” represents substituents on opposite sides of the plane of the ring.
- Mixtures of compounds wherein the substituents are disposed on both the same and opposite sides of plane of the ring are designated“cis/trans.”
- the disclosure also embraces isotopically-labeled compounds which are identical to those compounds recited herein, except that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature.
- isotopes that can be incorporated into compounds described herein include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, fluorine and chlorine, such as 2 H (“D”), 3 H, 13 C, 14 C, 15 N, 18 0, 17 0, 31 P, 32 P, 35 S, 18 F, and 36 Cl, respectively.
- a compound described herein can have one or more H atoms replaced with deuterium.
- Certain isotopically-labeled compounds can be useful in compound and/or substrate tissue distribution assays.
- Tritiated (i.e., 3 H) and carbon- 14 (i.e., 14 C) isotopes can be particularly preferred for their ease of preparation and
- Isotopically-labeled compounds can generally be prepared by following procedures analogous to those disclosed herein, for example, in the Examples section, by substituting an isotopically- labeled reagent for a non-isotopically-labeled reagent.
- phrases“pharmaceutically acceptable” and“pharmacologically acceptable,” as used herein, refer to compounds, molecular entities, compositions, materials, and/or dosage forms that do not produce an adverse, allergic or other untoward reaction when administered to an animal, or a human, as appropriate.
- preparations should meet sterility, pyrogenicity, general safety and purity standards as required by FDA Office of Biologies standards.
- phrases“pharmaceutically acceptable carrier” and“pharmaceutically acceptable excipient,” as used herein, refer to any and all solvents, dispersion media, coatings, isotonic and absorption delaying agents, and the like, that are compatible with pharmaceutical
- compositions can include phosphate buffered saline solution, water, emulsions (e.g., such as an oil/water or water/oil emulsions), and various types of wetting agents.
- emulsions e.g., such as an oil/water or water/oil emulsions
- the compositions also can include stabilizers and preservatives.
- composition refers to a composition comprising at least one compound as disclosed herein formulated together with one or more pharmaceutically acceptable carriers.
- the pharmaceutical compositions can also contain other active compounds providing supplemental, additional, or enhanced therapeutic functions.
- the compounds described in the disclosure can be administered to a mammal, such as a human, but can also be administered to other mammals such as an animal in need of veterinary treatment, for example, domestic animals (e.g., dogs, cats, and the like), farm animals (e.g., cows, sheep, pigs, horses, and the like) and laboratory animals (e.g., rats, mice, guinea pigs, and the like).
- the mammal treated in the methods described in the disclosure is preferably a mammal in which treatment, for example, of pain or depression, is desired.
- treating includes any effect, for example, lessening, reducing, modulating, ameliorating, or eliminating, that results in the improvement of the condition, disease, disorder, and the like, including one or more symptoms thereof. Treating can be curing, improving, or at least partially ameliorating the disorder.
- disorder refers to and is used interchangeably with, the terms“disease,” “condition,” or“illness,” unless otherwise indicated.
- modulation refers to and includes antagonism (e.g., inhibition), agonism, partial antagonism, and/or partial agonism.
- therapeutically effective amount refers to the amount of a compound (e.g., a disclosed compound) that will elicit the biological or medical response of a tissue, system, animal or human that is being sought by the researcher, veterinarian, medical doctor or other clinician.
- the compounds described in the disclosure can be administered in therapeutically effective amounts to treat a disease.
- a therapeutically effective amount of a compound can be the quantity required to achieve a desired therapeutic and/or prophylactic effect, such as an amount which results in lessening of a symptom of a disease such as depression.
- salt refers to any salt of an acidic or a basic group that may be present in a compound of the present disclosure, which salt is compatible with pharmaceutical administration ⁇
- salts of the compounds of the present disclosure may be derived from inorganic or organic acids and bases.
- salts include, but are not limited to: acetate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, citrate, camphorate, camphorsulfonate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, fumarate,
- flucoheptanoate glycerophosphate, hemisulfate, heptanoate, hexanoate, hydrochloride, hydrobromide, hydroiodide, 2-hydroxy ethanesulfonate, lactate, maleate, methanesulfonate, 2- naphthalenesulfonate, nicotinate, oxalate, palmoate, pectinate, persulfate, phenylpropionate, picrate, pivalate, propionate, succinate, tartrate, thiocyanate, tosylate, undecanoate, and the like.
- salts include anions of the compounds of the present disclosure compounded with a suitable cation such as Na + , NH 4 + , and NW 4 + (where W can be a Ci_ 4 alkyl group), and the like.
- a suitable cation such as Na + , NH 4 + , and NW 4 + (where W can be a Ci_ 4 alkyl group), and the like.
- salts of the compounds of the present disclosure can be pharmaceutically acceptable.
- salts of acids and bases that are non-pharmaceutically acceptable may also find use, for example, in the preparation or purification of a
- compositions that are basic in nature are capable of forming a wide variety of salts with various inorganic and organic acids.
- the acids that can be used to prepare pharmaceutically acceptable acid addition salts of such basic compounds are those that form non-toxic acid addition salts, i.e., salts containing pharmacologically acceptable anions, including but not limited to, malate, oxalate, chloride, bromide, iodide, nitrate, sulfate, bisulfate, phosphate, acid phosphate, isonicotinate, acetate, lactate, salicylate, citrate, tartrate, oleate, tannate, pantothenate, bitartrate, ascorbate, succinate, maleate, gentisinate, fumarate, gluconate, glucaronate, saccharate, formate, benzoate, glutamate, methanesulfonate, ethanesulfonate, henzenesul I
- compositions that are acidic in nature are capable of forming base salts with various pharmacologically acceptable cations.
- examples of such salts include alkali metal or alkaline earth metal salts and, particularly, calcium, magnesium, sodium, lithium, zinc, potassium, and iron salts.
- compositions that include a basic or acidic moiety can also form pharmaceutically acceptable salts with various amino acids.
- the compounds of the disclosure can contain both acidic and basic groups; for example, one amino and one carboxylic acid group. In such a case, the compound can exist as an acid addition salt, a zwitterion, or a base salt.
- the compounds disclosed herein can exist in a solvated form as well as an unsolvated form with pharmaceutically acceptable solvents such as water, ethanol, and the like, and it is intended that the disclosure embrace both solvated and unsolvated forms
- compositions and kits are described as having, including, or comprising specific components, or where processes and methods are described as having, including, or comprising specific steps, it is contemplated that, additionally, there are compositions and kits of the present disclosure that consist essentially of, or consist of, the recited components, and that there are processes and methods according to the present disclosure that consist essentially of, or consist of, the recited processing steps.
- an element or component is said to be included in and/or selected from a list of recited elements or components, it should be understood that the element or component can be any one of the recited elements or components, or the element or component can be selected from a group consisting of two or more of the recited elements or components.
- compositions of the present disclosure and/or in methods of the present disclosure, unless otherwise understood from the context.
- embodiments have been described and depicted in a way that enables a clear and concise application to be written and drawn, but it is intended and will be appreciated that embodiments can be variously combined or separated without parting from the present teachings and disclosure(s).
- all features described and depicted herein can be applicable to all aspects of the disclosure(s) described and depicted herein.
- the term“about” refers to a ⁇ 10% variation from the nominal value unless otherwise indicated or inferred from the context.
- molecular weight is provided and not an absolute value, for example, of a polymer, then the molecular weight should be understood to be an average molecule weight, unless otherwise stated or understood from the context.
- substituents are disclosed in groups or in ranges. It is specifically intended that the description include each and every individual subcombination of the members of such groups and ranges.
- the term“Ci -6 alkyl” is specifically intended to individually disclose Ci, C 2 , C 3 , C 4 , C 5 , C 6 , Ci-C 6 , C 1 -C 5 , C 1 -C 4 , Ci- C 3 , Ci-C 2 , C 2 -C 6 , C 2 -C 5 , C 2 -C 4 , C 2 -C 3 , C 3 -C 6 , C 3 -C 5 , C 3 -C 4 , C 4 -C 6 , C 4 -C 5 , and C 5 -C 6 alkyl.
- an integer in the range of 0 to 40 is specifically intended to individually disclose 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25,
- variable is defined as found elsewhere in the disclosure unless understood to be different from the context.
- definition of each variable and/or substituent for example, C i -C 6 alkyl, R 2 , R b , w and the like, when it occurs more than once in any structure or compound, can be independent of its definition elsewhere in the same structure or compound.
- Definitions of the variables and/or substituents in formulae and/or compounds herein encompass multiple chemical groups.
- the present disclosure includes embodiments where, for example, i) the definition of a variable and/or substituent is a single chemical group selected from those chemical groups set forth herein, ii) the definition is a collection of two or more of the chemical groups selected from those set forth herein, and iii) the compound is defined by a combination of variables and/or substituents in which the variables and/or substituents are defined by (i) or (ii).
- Disclosed compounds include a compound represented by Formula I:
- q 1, 2, 3, or 4;
- R 5 is independently, for each occurrence, one or more optional substituents selected from the group consisting of Ci-Cealkyl, -S(0) w -Ci_C 3 alkyl, -NR a R b , and halogen, wherein Ci- C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s ; R 6 is one or two optional substituents bound to one or more available ring carbons, and is independently, for each occurrence, selected from the group consisting of Ci-C 6 alkyl, - S(0) w -Ci-C 3 alkyl, -NR a R b , and halogen, wherein Ci-C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s ;
- w 0, 1, or 2;
- R 3 is selected from the group consisting of H, phenyl, Ci-Cealkyl, -C(0)R 31 , and - C(0)0R 32 , wherein Ci-C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s ;
- R 31 and R 32 are each independently selected from the group consisting of H, Ci-Cealkyl, -C3-C6cycloalkyl, and phenyl, wherein Ci-C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s , and phenyl is optionally substituted with one, two, or three substitutents each independently selected from R T ;
- R a and R b are independently, for each occurrence, selected from the group consisting of H, phenyl, Ci-C 4 alkyl, -Ci-C 4 alkylene-phenyl, or R a and R b taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted with one, two, or three substitutents each independently selected from hydroxyl, - Ci-C3alkoxy, -Ci-C3alkyl, and halogen, and Ci-C3alkyl is optionally substituted with one, two or three halogens;
- R s is independently, for each occurrence, selected from the group consisting of - C(0)NR a R b , -NR a R b , hydroxyl, S(0) w -Ci-C 3 alkyl, SH, phenyl and halogen; and
- R T is independently, for each occurrence, selected from the group consisting of -Ci- C 6 alkyl, hydroxyl, Ci-C 6 alkoxy, and halogen.
- q can be 1, 2 or 3. In certain embodiments, q can be 2. In certain embodiments, q can be 3. In certain embodiments, q can be 1.
- R 5 can be absent. In some embodiments, R 5 can be present one, two, three, or four times, and can be selected from the group consisting of Ci-Cealkyl, - S(0) w -Ci-C 3 alkyl, -NR a R b , and halogen, wherein Ci-C 6 alkyl is optionally substituted with one, two, or three substitutents each independently selected from R s .
- R 6 can be absent.
- R 3 can be Ci-Cealkyl, wherein Ci-C 6 alkyl is optionally substituted by one, two, or three substituents each independently selected from -C(0)-NR a R b , and hydroxyl; and R a and R b are each independently selected for each occurrence from the group consisting of H, Ci-C 4 alkyl, and -Ci-C 4 alkylene -phenyl, wherein phenyl is optionally substituted by one two or three substituents each independently selected from Ci-C alkoxy, Ci- C 3 alkyl, and halogen, and C ] -C 3 alkyl is optionally substituted with one, two, or three halogens independently selected from F, Cl, and Br.
- R 3 can be -C(0)-R 31 , wherein R 31 is Ci-C 6 alkyl, and Ci- C 6 alkyl is optionally substituted by one, two, or three substituents each independently selected from -C(0)-NR a R b , -NR a R b , and hydroxyl; and R a and R b are each independently selected for each occurrence from the group consisting of H and Ci-C alkyl.
- R 3 can be selected from the group consisting of:
- R a and R b can be each independently selected for each occurrence from the group consisting of H and -Ci-C alkyl.
- R 3 can be selected from the group consisting of:
- R a and R b can be each independently selected for each occurrence from the group consisting of H and -Ci-C alkyl, and R 8 can be one, two or three optional substituents each independently selected from the group consisting of hydroxyl, Br, Cl, F, methoxy, and -CF 3
- R a and R b can be H.
- R a can be H or methyl
- R b can be selected from the group consisting of methyl, ethyl, isobutyl, and -CFF-phenyl, wherein phenyl can be optionally substituted by one, two, or three substituents each independently selected from F, methoxy, and CF 3 .
- Disclosed compounds also include a compound represented by Formula II:
- R 3 is selected from the group consisting of H, -C(0)-Ci-C 6 alkyl, and -Ci-C 6 alkyl-C(0)- NR a R b ; wherein Ci-C 6 alkyl is optionally substituted by one, two, or three substituents each independently selected from halogen, hydroxyl, cyano, -CO2H, phenyl, -C(0)-NR a R b , and NR a R b ; and
- R a and R b are each independently for each occurrence selected from the group consisting of H, phenyl, Ci-C 4 alkyl, -Ci-C 4 alkylene -phenyl, or R a and R b taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted by one two or three substituents each independently selected from Ci- C 3 alkoxy, Ci-C 3 alkyl, and halogen, wherein Ci-C 3 alkyl is optionally substituted with one, two or three substituents independently selected from F, Cl, and Br.
- R 3 can be selected from the group consisting of:
- R a and R b can be each independently selected for each occurrence from the group consisting of H and -Ci-C 4 alkyl.
- R 3 can be selected from the group consisting of:
- R a and R b can be each independently selected for each occurrence from the group consisting of H and -Ci-C 4 alkyl, and R 8 can be one, two, or three optional substituents each independently selected from the group consisting of hydroxyl, Br, Cl, F, methoxy, and -CF 3.
- R a and R b can be H.
- R a can be H or methyl
- R b can be selected from the group consisting of methyl, ethyl, isobutyl, and -CH 2 -phenyl, wherein phenyl can be optionally substituted by one, two, or three substituents each independently selected from F, methoxy, and -CF 3 .
- R 3 independently can be an amino acid or a derivative of an amino acid, for example, an alpha“amino amide” represented by l3 ⁇ 4N-CF[(amino acid side chain)-C(0)NH 2 .
- the nitrogen atom of the amino group of the amino acid or the amino acid derivative is a ring nitrogen in a chemical formula described herein.
- the carboxylic acid of the amino acid or the amide group of an amino amide (amino acid derivative) is not within the ring structure, i.e., not a ring atom.
- the carboxylic acid group of the amino acid or the amino acid derivative forms an amide bond with a ring nitrogen in a chemical formula disclosed herein, thereby providing an amino amide, where the amino group of the amino amide is not within the ring structure, i.e., not a ring atom.
- R 3 independently can be an alpha amino acid, an alpha amino acid derivative, and/or another amino acid or amino acid derivative such as a beta amino acid or a beta amino acid derivative, for example, a beta amino amide.
- a disclosed compound is selected from the compounds delineated in the Examples or in Table 1 herein, and includes a pharmaceutically acceptable salt and/or a stereoisomer thereof.
- a disclosed compound is selected from the group consisting of:
- the compounds of the present disclosure and formulations thereof may have a plurality of chiral centers.
- Each chiral center may be independently R, S, or any mixture of R and S.
- a chiral center may have an R:S ratio of between about 100:0 and about 50:50 (“racemate”), between about 100:0 and about 75:25, between about 100:0 and about 85:15, between about 100:0 and about 90:10, between about 100:0 and about 95:5, between about 100:0 and about 98:2, between about 100:0 and about 99:1, between about 0:100 and 50:50, between about 0: 100 and about 25:75, between about 0:100 and about 15:85, between about 0:100 and about 10:90, between about 0:100 and about 5:95, between about 0:100 and about 2:98, between about 0:100 and about 1:99, between about 75:25 and 25:75, and about 50:50.
- Formulations of the disclosed compounds comprising a greater ratio of one or more isomers may possess enhanced therapeutic characteristic relative to racemic formulations of a disclosed compounds or mixture of compounds.
- chemical formulas contain the descriptor“-(R)-” or“-(S)-” that is further attached to solid wedge or dashed wedge. This descriptor is intended to show a methine carbon (CH) that is attached to three other substituents and has either the indicated R or S configuration.
- Disclosed compounds may provide for efficient cation channel opening at the NMDA receptor, e.g. may bind or associate with the glutamate site or glycine site or other modulatory site of the NMDA receptor to assist in opening the cation channel.
- the disclosed compounds may be used to regulate (turn on or turn off) the NMDA receptor through action as an agonist or antagonist.
- the compounds described herein may bind to a specific NMDA receptor subtypes.
- a disclosed compound may bind to one NMDA subtype and not another.
- a disclosed compound may bind to one, or more than one NMDA subtype, and/or may have substantially less (or substantial no) binding activity to certain other NMDA subtypes.
- a disclosed compound binds to NR2A with substantially no binding to NR2D.
- a disclosed compound binds to NR2B and NR2D with substantially lower binding to NR2A and NR2C.
- the compounds as described herein may bind to NMDA receptors.
- a disclosed compound may bind to the NMDA receptor resulting in agonist- like activity (facilitation) over a certain dosing range and/or may bind to the NMDA receptor resulting in antagonist- like activity (inhibition) over a certain dosing range.
- a disclosed compound may possess a potency that is lO-fold or greater than the activity of existing NMDA receptor modulators.
- the disclosed compounds may exhibit a high therapeutic index.
- the therapeutic index refers to the ratio of the dose that produces a toxicity in 50% of the population (i.e., TD 50 ) to the minimum effective dose for 50% of the population (i.e., ED 50 ).
- the therapeutic index (TD5o):(ED5o).
- a disclosed compound may have a therapeutic index of at least about 10:1, at least about 50:1, at least about 100:1, at least about 200:1, at least about 500:1, or at least about 1000:1.
- a pharmaceutical formulation or a pharmaceutical composition including a disclosed compound and a pharmaceutically acceptable excipient are provided.
- a pharmaceutical composition includes a racemic mixture or a varied stereoisomeric mixture of one or more of the disclosed compounds.
- a formulation can be prepared in any of a variety of forms for use such as for administering an active agent to a patient, who may be in need thereof, as are known in the pharmaceutical arts.
- the pharmaceutical compositions of the present disclosure can be formulated for administration in solid or liquid form, including those adapted for the following: (1) oral administration, for example, drenches (aqueous or non-aqueous solutions or suspensions), tablets (e.g., those targeted for buccal, sublingual, and/or systemic absorption), boluses, powders, granules, and pastes for application to the tongue; (2) parenteral administration, for example, drenches (aqueous or non-aqueous solutions or suspensions), tablets (e.g., those targeted for buccal, sublingual, and/or systemic absorption), boluses, powders, granules, and pastes for application to the tongue; (2) parenteral
- administration by, for example, subcutaneous, intramuscular, intraperitoneal, intravenous or epidural injection as, for example, a sterile solution or suspension, or sustained-release formulation; (3) topical administration, for example, as a cream, ointment, or a controlled- release patch or spray applied to the skin; (4) intravaginal or intrarectal administration, for example, as a pessary, cream or foam; (5) sublingual administration; (6) ocular administration; (7) transdermal administration; or (8) nasal administration.
- compositions of the disclosure can be suitable for delivery to the eye, i.e., ocularly.
- Related methods can include administering a therapeutically effective amount of a disclosed compound or a pharmaceutical composition including a disclosed compound to a patient in need thereof, for example, to an eye of the patient, where
- administering can be topically, subconjunctivally, subtenonly, intravitreally, retrobulbarly, peribulbarly, intracomerally, and/or systemically.
- Amounts of a disclosed compound as described herein in a formulation may vary according to factors such as the disease state, age, sex, and weight of the individual. Dosage regimens may be adjusted to provide the optimum therapeutic response. For example, a single bolus may be administered, several divided doses may be administered over time or the dose may be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation. It is especially advantageous to formulate parenteral compositions in dosage unit form for ease of administration and uniformity of dosage.
- Dosage unit form as used herein refers to physically discrete units suited as unitary dosages for the mammalian subjects to be treated; each unit containing a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier.
- compositions typically must be sterile and stable under the conditions of manufacture and storage.
- the composition can be formulated as a solution, microemulsion, liposome, or other ordered structure suitable to high drug concentration.
- the carrier can be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyethylene glycol, and the like), and suitable mixtures thereof.
- the proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants.
- isotonic agents for example, sugars, polyalcohols such as mannitol, sorbitol, or sodium chloride in the composition.
- Prolonged absorption of the injectable compositions can be brought about by including in the composition an agent which delays absorption, for example, monostearate salts and gelatin.
- the compounds can be administered in a time release formulation, for example in a composition which includes a slow release polymer.
- the compounds can be prepared with carriers that will protect the compound against rapid release, such as a controlled release formulation, including implants and microencapsulated delivery systems.
- Biodegradable, biocompatible polymers can be used, such as ethylene vinyl acetate, poly anhydrides, polyglycolic acid, collagen, polyorthoesters, polylactic acid and polylactic, polyglycolic copolymers (PLG). Many methods for the preparation of such formulations are generally known to those skilled in the art.
- Sterile injectable solutions can be prepared by incorporating the compound in the required amount in an appropriate solvent with one or a combination of ingredients enumerated above, as required, followed by filtered sterilization.
- dispersions are prepared by incorporating the active compound into a sterile vehicle which contains a basic dispersion medium and the required other ingredients from those enumerated above.
- the preferred methods of preparation are vacuum drying and freeze-drying which yields a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
- a compound can be formulated with one or more additional compounds that enhance the solubility of the compound.
- Methods of the disclosure for treating a condition in a patient in need thereof generally include administering a therapeutically effective amount of a compound described herein or a composition including such a compound.
- the condition may be a mental condition.
- a mental illness may be treated.
- a nervous system condition may be treated.
- a condition that affects the central nervous system, the peripheral nervous system, and/or the eye may be treated.
- neurodegenerative diseases may be treated.
- the methods include administering a compound to treat patients suffering from autism, anxiety, depression, bipolar disorder, attention deficit disorder, attention deficit hyperactivity disorder (ADHD), schizophrenia, a psychotic disorder, a psychotic symptom, social withdrawal, obsessive-compulsive disorder (OCD), phobia, post-traumatic stress syndrome, a behavior disorder, an impulse control disorder, a substance abuse disorder (e.g., a withdrawal symptom, opiate addiction, nicotine addiction, and ethanol addition), a sleep disorder, a memory disorder (e.g., a deficit, loss, or reduced ability to make new memories), a learning disorder, urinary incontinence, multiple system atrophy, progressive supra-nuclear palsy, Friedrich's ataxia, Down’s syndrome, fragile X syndrome, tuberous sclerosis, olivio- ponto-cerebellar atrophy, cerebral palsy, drug-induced optic neuritis, ischemic retinopathy, diabetic retinopathy, glaucoma, dementia,
- ADHD attention
- the present disclosure provides methods of treating a cognitive impairment disorder, for example, a dysfunction in learning and/or memory such as that seen in age-related cognitive decline, Lewy body dementia, AIDS dementia, HIV dementia, vascular dementia, mild cognitive impairment in Huntington’s disease, Huntington’s disease dementia, mild cognitive impairment in Parkinson’s disease, Parkinson’s disease dementia, mild cognitive impairment in Alzheimer’s disease, Alzheimer’s dementia, frontotemporal dementia, cognitive impairment associated with schizophrenia (CIAS), and cognitive impairment associated with seizures, stroke, cerebral ischemia, hypoglycemia, cardiac arrest, migraine, multiple sclerosis, traumatic brain injury, and/or Down’s syndrome.
- a cognitive impairment disorder for example, a dysfunction in learning and/or memory such as that seen in age-related cognitive decline, Lewy body dementia, AIDS dementia, HIV dementia, vascular dementia, mild cognitive impairment in Huntington’s disease, Huntington’s disease dementia, mild cognitive impairment in Parkinson’s disease, Parkinson’s disease dementia, mild cognitive impairment in Alzheimer’s disease, Alzheimer’s dementia, frontotemporal
- methods for treating schizophrenia are provided.
- paranoid type schizophrenia disorganized type schizophrenia (i.e., hebephrenic schizophrenia), catatonic type schizophrenia, undifferentiated type schizophrenia, residual type schizophrenia, post-schizophrenic depression, and simple schizophrenia
- Psychitic disorders such as schizoaffective disorders, delusional disorders, brief psychotic disorders, shared psychotic disorders, and psychotic disorders with delusions or hallucinations may also be treated using the compositions disclosed herein.
- Paranoid schizophrenia may be characterized where delusions or auditory hallucinations are present, but thought disorder, disorganized behavior, or affective flattening are not.
- Delusions may be persecutory and/or grandiose, but in addition to these, other themes such as ashamedy, religiosity, or somatization may also be present.
- Disorganized type schizophrenia may be characterized where thought disorder and flat affect are present together.
- Catatonic type schizophrenia may be characterized where the patient may be almost immobile or exhibit agitated, purposeless movement. Symptoms can include catatonic stupor and waxy flexibility.
- Undifferentiated type schizophrenia may be characterized where psychotic symptoms are present but the criteria for paranoid, disorganized, or catatonic types have not been met.
- Residual type schizophrenia may be characterized where positive symptoms are present at a low intensity only.
- Post-schizophrenic depression may be characterized where a depressive episode arises in the aftermath of a schizophrenic illness where some low-level schizophrenic symptoms may still be present.
- Simple schizophrenia may be characterized by insidious and progressive development of prominent negative symptoms with no history of psychotic episodes.
- methods are provided for treating psychotic symptoms that may be present in other mental disorders, including, but not limited to, bipolar disorder, borderline personality disorder, drug intoxication, and drug-induced psychosis.
- methods for treating delusions e.g., "non-bizarre" that may be present in, for example, delusional disorder are provided.
- methods for treating social withdrawal in conditions including, but not limited to, social anxiety disorder, avoidant personality disorder, and schizotypal personality disorder are provided.
- the disclosure provides methods for treating a
- the neurodevelopmental disorder related to synaptic dysfunction can be Rett syndrome also known as cerebroatrophic hyperammonemia, MECP2 duplication syndrome (e.g., a MECP2 disorder), CDKL5 syndrome, fragile X syndrome (e.g., a FMR1 disorder), tuberous sclerosis (e.g., a TSC1 disorder and/or a TSC2 disorder), neurofibromatosis (e.g., a NF1 disorder), Angelman syndrome (e.g., a UBE3A disorder), the PTEN hamartoma tumor syndrome, Phelan-McDermid syndrome (e.g., a SHANK3 disorder), or infantile spasms.
- Rett syndrome also known as cerebroatrophic hyperammonemia, MECP2 duplication syndrome (e.g., a MECP2 disorder), CDKL5 syndrome, fragile X syndrome (e.g., a FMR1 disorder), tuberous sclerosis (e.g., a T
- the neurodevelopmental disorder can be caused by mutations in the neuroligin (e.g., a NLGN3 disorder and/or a NLGN2 disorder) and/or the neurexin (e.g., a NRXN1 disorder).
- the neuroligin e.g., a NLGN3 disorder and/or a NLGN2 disorder
- the neurexin e.g., a NRXN1 disorder
- neuropathic pain can be acute or chronic.
- the neuropathic pain can be associated with a condition such as herpes, HIV, traumatic nerve injury, stroke, post- ischemia, chronic back pain, post-herpetic neuralgia, fibromyalgia, reflex sympathetic dystrophy, complex regional pain syndrome, spinal cord injury, sciatica, phantom limb pain, diabetic neuropathy such as diabetic peripheral neuropathy (“DPN”), and cancer chemotherapeutic-induced neuropathic pain.
- methods for enhancing pain relief and for providing analgesia to a patient are also provided.
- Further methods include a method of treating autism and/or an autism spectrum disorder in a patient need thereof, comprising administering an effective amount of a compound to the patient.
- a method for reducing the symptoms of autism in a patient in need thereof comprises administering an effective amount of a disclosed compound to the patient.
- the compound may decrease the incidence of one or more symptoms of autism such as eye contact avoidance, failure to socialize, attention deficit, poor mood, hyperactivity, abnormal sound sensitivity, inappropriate speech, disrupted sleep, and perseveration. Such decreased incidence may be measured relative to the incidence in the untreated individual or an untreated individual(s).
- Also provided herein is a method of modulating an autism target gene expression in a cell comprising contacting a cell with an effective amount of a compound described herein.
- the autism gene expression may be for example, selected from ABAT, APOE, CHRNA4, GABRA5,GFAP, GRIN2A, PDYN, and PENK.
- a method of modulating synaptic plasticity in a patient suffering from a synaptic plasticity related disorder comprising administering to the patient an effective amount of a compound.
- a method of treating Alzheimer’s disease, or e.g., treatment of memory loss that e.g., accompanies early stage Alzheimer’s disease, in a patient in need thereof comprising administering a compound.
- a method of modulating an Alzheimer’s amyloid protein e.g., beta amyloid peptide, e.g. the isoform Abi_ 42 ), in-vitro or in-vivo (e.g. in a cell) comprising contacting the protein with an effective amount of a compound is disclosed.
- a compound may block the ability of such amyloid protein to inhibit long-term potentiation in hippocampal slices as well as apoptotic neuronal cell death.
- a disclosed compound may provide neuroprotective properties to a Alzheimer’s patient in need thereof, for example, may provide a therapeutic effect on later stage Alzheimer’s -associated neuronal cell death.
- the disclosed methods include treating a psychosis or a pseudobulbar affect (“PBA”) that is induced by another condition such as a stroke, amyotrophic lateral sclerosis (ALS or Lou Gehrig’s disease), multiple sclerosis, traumatic brain injury, Alzheimer’s disease, dementia, and/or Parkinson’s disease.
- PBA pseudobulbar affect
- Such methods include administration of a therapeutically effective amount of a disclosed compound to a patient in need thereof.
- a method of treating depression includes administering a therapeutically effective amount of a compound described herein.
- the treatment may relieve depression or a symptom of depression without affecting behavior or motor coordination and without inducing or promoting seizure activity.
- Exemplary depression conditions that are expected to be treated according to this aspect include, but are not limited to, major depressive disorder, dysthymic disorder, psychotic depression, postpartum depression, premenstrual syndrome, premenstrual dysphoric disorder, seasonal affective disorder (SAD), bipolar disorder (or manic depressive disorder), mood disorder, and depressions caused by chronic medical conditions such as cancer or chronic pain, chemotherapy, chronic stress, and post traumatic stress disorders.
- SAD seasonal affective disorder
- bipolar disorder or manic depressive disorder
- mood disorder and depressions caused by chronic medical conditions such as cancer or chronic pain, chemotherapy, chronic stress, and post traumatic stress disorders.
- patients suffering from any form of depression often experience anxiety.
- Various symptoms associated with anxiety include fear, panic, heart palpitations, shortness of breath, fatigue, nausea
- a method of treating depression in a treatment resistant patient comprising a) optionally identifying the patient as treatment resistant and b) administering an effective dose of a compound to said patient.
- a compound described herein may be used for acute care of a patient.
- a compound may be administered to a patient to treat a particular episode (e.g., a severe episode) of a condition disclosed herein.
- combination therapies comprising a compound of the disclosure in combination with one or more other active agents.
- a compound may be combined with one or more antidepressants, such as tricyclic antidepressants, MAO-I's, SSRI's, and double and triple uptake inhibitors and/or anxiolytic drugs.
- Exemplary drugs that may be used in combination with a compound include Anafranil, Adapin, Aventyl, Elavil, Norpramin, Pamelor, Pertofrane, Sinequan, Surmontil, Tofranil, Vivactil, Parnate, Nardil, Marplan, Celexa, Lexapro, Luvox, Paxil, Prozac, Zoloft, Wellbutrin, Effexor, Remeron, Cymbalta, Desyrel (trazodone), and Ludiomill.
- a compound may be combined with an antipsychotic medication.
- Non-limiting examples of antipsychotics include butyrophenones, phenothiazines, thioxanthenes, clozapine, olanzapine, risperidone, quetiapine, ziprasidone, amisulpride, asenapine, paliperidone, iloperidone, zotepine, sertindole, lurasidone, and aripiprazole. It should be understood that combinations of a compound and one or more of the above therapeutics may be used for treatment of any suitable condition and are not limited to use as antidepressants or antipsychotics.
- Ac is acetyl (-C(0)CH 3 ), AIDS is acquired immune deficiency syndrome, Boc and BOC are tert- butoxycarbonyl, Boo 2 0 is di-ieri-butyl dicarbonate, Bn is benzyl, Cbz is carboxybenzyl, DCM is dichloromethane, DCE is l,2-dichloroethane, DEA is diethylamine, DIPEA is NN- diisopropylethylamine, DMF is /V,/V-dirnethylformamide, DMSO is dimethyl sulfoxide, EDC is l-ethyl-3-(3-dimethylaminopropyl)carbodiimide, ESI is electrospray ionization, EtOAc is ethyl acetate, h is hour, HATU is 2-(7-aza- 1 //-benzotri azo
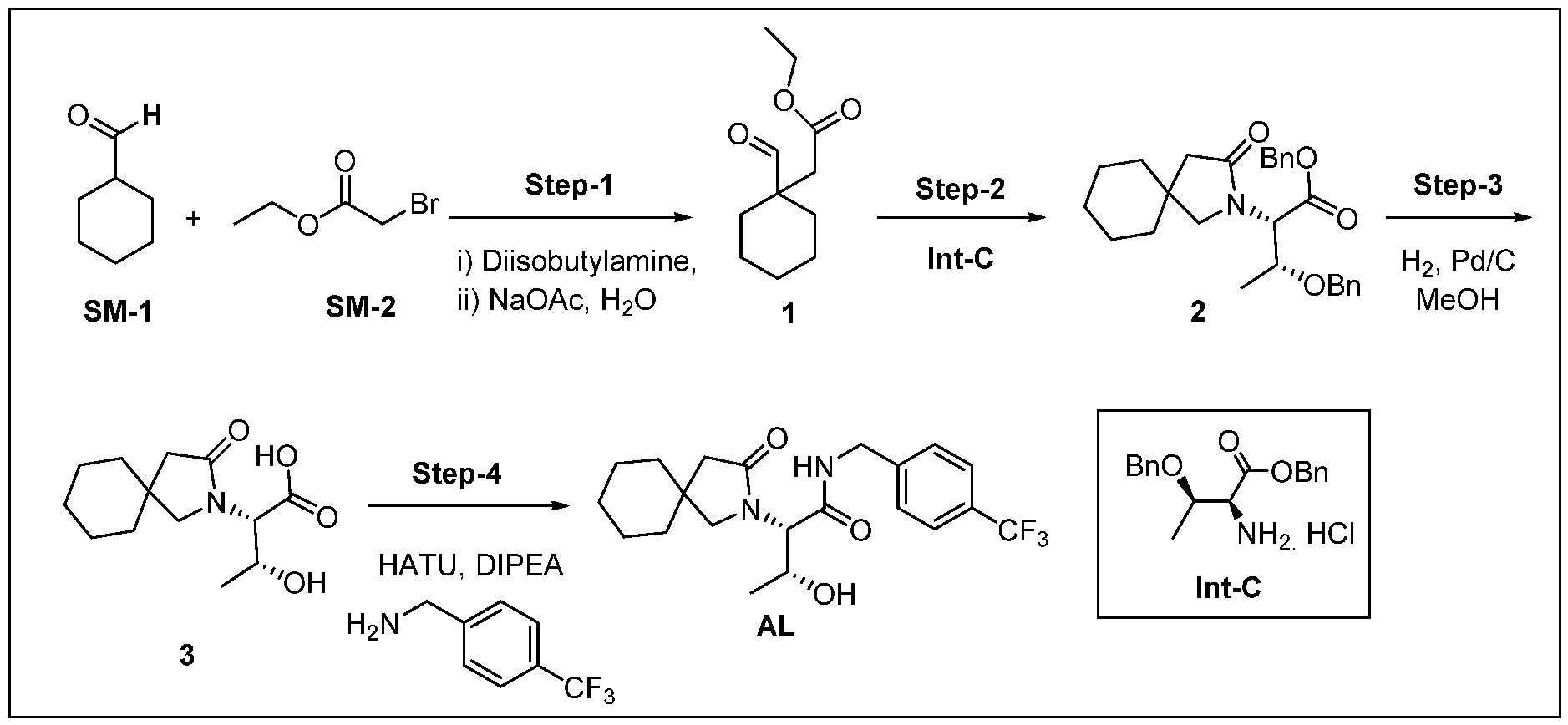
- reaction mixture was quenched with water (30 mL) and extracted with CH2CI2 (3 x 20 mL). The combined organic layer was washed with brine (20 mL), dried over Na 2 S0 4 and concentrated under reduced pressure. The crude was purified by column chromatography using 5% methanol/ CH2CI2 to afford compound 4 (0.40g, 57%) as an off white solid.
- Chiral HPLC >99%; Column: YMC CHIRALART CELLULOSE-SC (250*4.6 mm, 5u); Mobile Phase: A: 77-hexane + 0.1% TLA; Mobile Phase: B: Iso-propyl alcohol; Plow rate : 1.0 mL/min; Retention time: 7.35 min.
- reaction mixture was quenched with water (30 mL) and extracted with DCM (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over Na SCL and concentrated under reduced pressure.
- the crude was purified by column chromatography using 5% methanol/DCM to afford racemic mixture of AC (0.51 g) as an off white solid.
- the racemic mixture was purified by preparative HPLC followed by chiral HPLC to afford AC (0.17 g) as an off white solid.
- reaction mixture was quenched with water (30 mL) and extracted with EtOAc (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over NanSQ* and concentrated under reduced pressure. The crude product was purified by column chromatography eluting with EtOAc/n-hexane to afford compound 1 (7.50 g, 60%) as a colourless oil.
- the crude material was purified by column chromatography by eluting with 1 % MeOH/ CH2CI2 to afford AM (700 mg, 53%) as an off white sticky solid. This material was further purified by preparative HPLC purification to obtain AM (200 mg) as an off white hygroscopic solid.
- the DPM disintegrations per minute
- the baseline value was determined from the best fit curve of the DPM values modeled using the GraphPad program and the log(agonist) vs. response(three parameters) algorithm was then subtracted from all points in the dataset.
- the % maximal [ 3 H]MK-80l binding was then calculated relative to that of 1 mM glycine: all baseline subtracted DPM values were divided by the average value for 1 mM glycine.
- the EC and % maximal activity were then obtained from the best fit curve of the % maximal [ 3 H]MK-80l binding data modelled using the GraphPad program and the log(agonist) vs. response( three parameters) algorithm.
- Microsomal stability of disclosed compounds was investigated. The following table indicates the percent of compound remaining after 60 minutes.
- Plasma stability of disclosed compounds was investigated. The following table indicates the percent of compound remaining after 60 minutes.
- Sprague Dawley rats were dosed intravenously using a normal saline formulation containing 2 mg/kg of the compounds identified in the below table.
- the table below summarizes the results of the IV pharmacokinetics.
- Sprague Dawley rats were dosed per os (oral gavage) using a normal saline formulation containing 10 mg/kg of the compounds identified in the table below.
- Plasma, brain, and CSF samples were analyzed at various time points over a 24 hour period.
- the table below summarizes the results of the oral pharmacokinetics, where the first three values (T max , C max and AUCi ast ) are plasma values. N/D indicates that the measurement was not done.
- the Porsolt forced swim test adapted for use in rats was performed as described by Burgdorf et ak, (The long-lasting antidepressant effects of rapastinel (GLYX-13) are associated with a metaplasticity process in the medial prefrontal cortex and hippocampus. Neuroscience 308:202-211, 2015). Animals were placed in a 46 cm tall x 20 cm in diameter clear glass tube filled to 30 cm with tap water (23 ⁇ 1 °C) for 15 min on the first day (habituation) and 5 min on the subsequent test day. Animals were tested 1 h or 1 week post-dosing with the test compounds or vehicle control (0.5% sodium carboxymethyl cellulose in 0.9% sterile saline).
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Veterinary Medicine (AREA)
- Biomedical Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Psychiatry (AREA)
- Pain & Pain Management (AREA)
- Hospice & Palliative Care (AREA)
- Rheumatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Disclosed are compounds having potency in the modulation of NMDA receptor activity. Such compounds can be used in the treatment of conditions such as depression and related disorders as well as other disorders.
Description
SPIRO-LACTAM NMDA RECEPTOR MODULATORS AND USES THEREOF
CROSS REFERENCE TO RELATED APPLICATIONS
This application claims priority to and the benefit of U.S. Provisional Patent
Application No. 62/718,070, filed on August 13, 2018, and U.S. Provisional Patent Application No. 62/624,217, filed on January 31, 2018; the contents of each of which are hereby incorporated by reference herein in their entirety.
BACKGROUND
An N-methyl-d-aspartate (“NMDA”) receptor is a postsynaptic, ionotropic receptor that is responsive to, inter alia, the excitatory amino acids glutamate and glycine and the synthetic compound NMDA. The NMDA receptor controls the flow of both divalent and monovalent ions into the postsynaptic neural cell through a receptor associated channel (Foster et al,
Nature 1987, 329:395-396; Mayer et al, Trends in Pharmacol. Sci. 1990, 11:254-260). The NMDA receptor has been implicated during development in specifying neuronal architecture and synaptic connectivity, and may be involved in experience-dependent synaptic
modifications. In addition, NMDA receptors are also thought to be involved in long term potentiation and central nervous system disorders.
The NMDA receptor plays a major role in the synaptic plasticity that underlies many higher cognitive functions, such as memory acquisition, retention and learning, as well as in certain cognitive pathways and in the perception of pain (Collingridge et al. , The NMDA Receptor, Oxford University Press, 1994). In addition, certain properties of NMDA receptors suggest that they may be involved in the information-processing in the brain that underlies consciousness itself.
The NMDA receptor has drawn particular interest since it appears to be involved in a broad spectrum of CNS disorders. For instance, during brain ischemia caused by stroke or traumatic injury, excessive amounts of the excitatory amino acid glutamate are released from damaged or oxygen deprived neurons. This excess glutamate binds to the NMDA receptors which opens their ligand-gated ion channels; in turn the calcium influx produces a high level of intracellular calcium which activates a biochemical cascade resulting in protein degradation and cell death. This phenomenon, known as excitotoxicity, is also thought to be responsible for the neurological damage associated with other disorders ranging from hypoglycemia and
cardiac arrest to epilepsy. In addition, there are preliminary reports indicating similar involvement in the chronic neurodegeneration of Huntington's, Parkinson's and Parkinson’s related conditions such as dyskinesia and L-dopa induced dyskinesia and Alzheimer's diseases. Activation of the NMDA receptor has been shown to be responsible for post-stroke convulsions, and, in certain models of epilepsy, activation of the NMDA receptor has been shown to be necessary for the generation of seizures. Neuropsychiatric involvement of the NMDA receptor has also been recognized since blockage of the NMDA receptor Ca++ channel by the animal anesthetic PCP (phencyclidine) produces a psychotic state in humans similar to schizophrenia (reviewed in Johnson, K. and Jones, S., 1990). Further, NMDA receptors have also been implicated in certain types of spatial learning.
The NMDA receptor is believed to consist of several protein chains embedded in the postsynaptic membrane. The first two types of subunits discovered so far form a large extracellular region, which probably contains most of the allosteric binding sites, several transmembrane regions looped and folded so as to form a pore or channel, which is permeable to Ca++, and a carboxyl terminal region. The opening and closing of the channel is regulated by the binding of various ligands to domains (allosteric sites) of the protein residing on the extracellular surface. The binding of the ligands is thought to affect a conformational change in the overall structure of the protein which is ultimately reflected in the channel opening, partially opening, partially closing, or closing.
A need continues to exist in the art for novel and more specific and/or potent compounds that are capable of modulating NMDA receptors, and provide pharmaceutical benefits. In addition, a need continues to exist in the medical arts for orally deliverable forms of such compounds.
SUMMARY
The present disclosure includes compounds that can be NMDA modulators. More specifically, the present disclosure provides a compound represented by Formula I:
or a pharmaceutically acceptable salt and/or a stereoisomer thereof, wherein:
q is 1, 2, 3, or 4;
R5 is independently, for each occurrence, one or more optional substituents selected from the group consisting of Ci-C6alkyl, -S(0)w-Ci_C3alkyl, -NRaRb, and halogen, wherein Ci- C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
R6 is one or two optional substituents bound to one or more available ring carbons, and is independently, for each occurrence, selected from the group consisting of Ci-Cealkyl, - S(0)w-Ci-C3alkyl, -NRaRb, and halogen, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
w is 0, 1, or 2;
R3 is selected from the group consisting of H, phenyl, Ci-Cealkyl, -C(0)R31, and - C(0)0R32, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
R31 and R32 are each independently selected from the group consisting of H, Ci-Cealkyl, -C3-C6cycloalkyl, and phenyl, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs, and phenyl is optionally substituted with one, two, or three substitutents each independently selected from RT;
Ra and Rb are independently, for each occurrence, selected from the group consisting of H, phenyl, Ci-C4alkyl, -Ci-C4alkylene-phenyl, or Ra and Rb taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted with one, two, or three substitutents each independently selected from hydroxyl, - Ci-C3alkoxy, -Ci-C3alkyl, and halogen, and Ci-C3alkyl is optionally substituted with one, two or three halogens;
Rs is independently, for each occurrence, selected from the group consisting of - C(0)NRaRb, -NRaRb, hydroxyl, S(0)w-Ci-C3alkyl, SH, phenyl and halogen; and
RT is independently, for each occurrence, selected from the group consisting of -Ci-Cealkyl, hydroxyl, Ci-C6alkoxy, and halogen.
Also disclosed herein is a compound that can be represented by Formula II:
or a pharmaceutically acceptable salt and/or a stereoisomer thereof, wherein:
R3 is selected from the group consisting of H, -C( O )-C| -Chalky 1, and -Ci-C6alkyl-C(0)- NRaRb; wherein C i -Chalky 1 is optionally substituted by one, two, or three substituents each independently selected from halogen, hydroxyl, cyano, -C02H, phenyl, -C(0)-NRaRb, and NRaRb; and
Ra and Rb are each independently for each occurrence selected from the group consisting of H, phenyl, Ci-C4alkyl, -Ci-C4alkylene -phenyl, or Ra and Rb taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted by one two or three substituents each independently selected from Ci- C3alkoxy, Ci-C3alkyl, and halogen, wherein Ci-C3alkyl is optionally substituted with one, two or three substituents independently selected from F, Cl, and Br.
Also provided herein are pharmaceutically acceptable compositions comprising a disclosed compound, and a pharmaceutically acceptable excipient. Such compositions can be suitable for administration to a patient orally, parenterally, topically, intravaginally, intrarectally, sublingually, ocularly, transdermally, or nasally.
In one aspect, a method of treating a condition selected from the group consisting of autism, anxiety, depression, bipolar disorder, attention deficit disorder, attention deficit hyperactivity disorder (ADHD), schizophrenia, a psychotic disorder, a psychotic symptom, social withdrawal, obsessive-compulsive disorder, phobia, post-traumatic stress disorder or syndrome, a behavior disorder, an impulse control disorder, a substance abuse disorder, a sleep disorder, a cognitive impairment disorder such as a memory disorder or a learning disorder, urinary incontinence, multiple system atrophy, progressive supra-nuclear palsy, Friedrich's ataxia, Down’s syndrome, fragile X syndrome, tuberous sclerosis, olivio-ponto-cerebellar atrophy, Rett syndrome, cerebral palsy, drug-induced optic neuritis, ischemic retinopathy, diabetic retinopathy, glaucoma, dementia, AIDS dementia, Alzheimer’s disease, Huntington’s chorea, spasticity, myoclonus, muscle spasm, Tourette's syndrome, epilepsy, cerebral ischemia, stroke, a brain tumor, traumatic brain injury, cardiac arrest, myelopathy, spinal cord injury, peripheral neuropathy, fibromyalgia, acute neuropathic pain, and chronic neuropathic pain, in a patient in need thereof is provided. Such methods may comprise administering to a patient a therapeutically effective amount of a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof, or a pharmaceutical composition including a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof.
In various embodiments, a method of this disclosure includes treating depression. In
some embodiments, a method of this disclosure includes treating schizophrenia. In certain embodiments, a method of this disclosure includes treating Alzheimer’s disease. In various embodiments, a method of this disclosure includes treating attention deficit disorder. In some embodiments, a method of this disclosure includes treating anxiety. In certain embodiments, a method of this disclosure includes treating a migraine. In various embodiments, a method of this disclosure includes treating neuropathic pain. In some embodiments, a method of this disclosure includes treating traumatic brain injury. In certain embodiments, a method of this disclosure includes treating a neurodevelopment disorder related to a synaptic dysfunction. In various embodiments, a method of this disclosure includes treating a cognitive impairment disorder. Such methods may comprise administering to a patient a therapeutically effective amount of a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof, or a pharmaceutical composition including a disclosed compound, or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof.
DETAILED DESCRIPTION
This disclosure is generally directed to compounds that are capable of modulating NMDA receptors, for example, NMDA receptor antagonists, agonists, or partial agonists, and compositions and/or methods of using the disclosed compounds. In some embodiments, compounds described herein bind to NMDA receptors expressing certain NR2 subtypes. In some embodiments, the compounds described herein bind to one NR2 subtype and not another. It should be appreciated that the disclosed compounds may modulate other protein targets and/or specific NMDA receptor subtype.
The term“alkyl,” as used herein, refers to a saturated straight-chain or branched hydrocarbon, such as a straight-chain or branched group of 1-6, 1-4, or 1-3 carbon atoms, referred to herein as -Ce alkyl, C1-C4 alkyl, and C1-C3 alkyl, respectively. For example,“Ci- C6 alkyl” refers to a straight-chain or branched saturated hydrocarbon containing 1-6 carbon atoms. Examples of a C 1 -C6 alkyl group include, but are not limited to, methyl, ethyl, propyl, butyl, pentyl, hexyl, isopropyl, isobutyl, sec-butyl, ieri-butyl, isopentyl, and neopentyl. In another example,“C1-C4 alkyl” refers to a straight-chain or branched saturated hydrocarbon containing 1-4 carbon atoms. Examples of a C1-C4 alkyl group include, but are not limited to, methyl, ethyl, propyl, butyl, isopropyl, isobutyl, sec-butyl and ieri-butyl. Exemplary alkyl groups include, but are not limited to, methyl, ethyl, propyl, isopropyl, 2-methyl- 1 -propyl, 2- methyl-2-propyl, 2-methyl- l-butyl, 3 -methyl- 1 -butyl, 3-methyl-2-butyl, 2,2-dimethyl- l-propyl, 2-methyl- 1 -pentyl, 3-methyl- 1 -pentyl, 4-methyl- 1 -pentyl, 2-methyl-2-pentyl, 3-methyl-2-
pentyl, 4-methyl-2-pentyl, 2,2-dimethyl- 1 -butyl, 3,3-dimethyl-l-butyl, 2-ethyl- 1 -butyl, butyl, isobutyl, t-butyl, pentyl, isopentyl, neopentyl, and hexyl.
The term“alkoxy,” as used herein, refers to an alkyl group attached to an oxygen atom (alkyl-O-). Alkoxy groups can have 1-6 or 2-6 carbon atoms and are referred to herein as Ci- C6 alkoxy and C2-Cr, alkoxy, respectively. Exemplary alkoxy groups include, but are not limited to, methoxy, ethoxy, propyloxy, isopropoxy, and tert-butoxy.
The term“carbonyl,” as used herein, refers to the radical -C(O)- or C=0.
The phrase,“carbocyclic ring,” as used herein, refers to a hydrocarbon ring system in which all the ring atoms are carbon. Exemplary carbocyclic rings including cycloalkyls and phenyl.
The term“cycloalkyl,” as used herein, refers to a monocyclic saturated or partially unsaturated hydrocarbon ring (carbocyclic) system, for example, where each ring is either completely saturated or contains one or more units of unsaturation, but where no ring is aromatic. A cycloalkyl can have 3-6 or 4-6 carbon atoms in its ring system, referred to herein as C3-C6 cycloalkyl or C4-C6 cycloalkyl, respectively. Exemplary cycloalkyl groups include, but are not limited to, cyclohexyl, cyclohexenyl, cyclopentyl, cyclopentenyl, cyclobutyl, and cyclopropyl.
The terms“halo” and“halogen,” as used herein, refer to fluoro (F), chloro (Cl), bromo (Br), and/or iodo (I).
The term“heteroatom,” as used herein, refers to an atom of any element other than carbon or hydrogen and includes, for example, nitrogen (N), oxygen (O), silicon (Si), sulfur (S), phosphorus (P), and selenium (Se).
The term“heterocycloalkyl” or“heterocyclic ring” as used herein, is art-recognized and refer to saturated or partially unsaturated 3- to 8-membered ring systems, for example, 3- to 7- or 3- to 6-membered ring systems, whose ring system include one, two or three heteroatoms, such as nitrogen, oxygen, and/or sulfur. A heterocycloalkyl can be fused to one or more phenyl, partially unsaturated, or saturated rings. Examples of heterocycloalkyls include, but are not limited to, pyrrolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, and piperazinyl.
The terms“hydroxy” and“hydroxyl,” as used herein, refer to the radical -OH.
The term“oxo,” as used herein, refers to the radical =0 (double bonded oxygen).
The term“amino acid,” as used herein, includes any one of the following alpha amino
acids: isoleucine, alanine, leucine, asparagine, lysine, aspartate, methionine, cysteine, phenylalanine, glutamate, threonine, glutamine, tryptophan, glycine, valine, proline, arginine, serine, histidine, and tyrosine. An amino acid also can include other art-recognized amino acids such as beta amino acids.
The term“compound,” as used herein, refers to the compound itself and its
pharmaceutically acceptable salts, hydrates, and N-oxides including its various stereoisomers and its isotopically-labelled forms, unless otherwise understood from the context of the description or expressly limited to one particular form of the compound, i.e., the compound itself, a specific stereoisomer and/or isotopically-labelled compound, or a pharmaceutically acceptable salt, a hydrate, or an N-oxide thereof. It should be understood that a compound can refer to a pharmaceutically acceptable salt, or a hydrate, or an N-oxide of a stereoisomer of the compound and/or an isotopically-labelled compound.
The term“moiety,” as used herein, refers to a portion of a compound or molecule.
The compounds of the disclosure can contain one or more chiral centers and/or double bonds and therefore, can exist as stereoisomers, such as geometric isomers, and enantiomers or diastereomers. The term“stereoisomers,” when used herein, consists of all geometric isomers, enantiomers and/or diastereomers of the compound. For example, when a compound is shown with specific chiral center(s), the compound depicted without such chirality at that and other chiral centers of the compound are within the scope of the present disclosure, i.e., the compound depicted in two-dimensions with“flat” or“straight” bonds rather than in three dimensions, for example, with solid or dashed wedge bonds. Stereospecific compounds may be designated by the symbols“R” or“S,” depending on the configuration of substituents around the stereogenic carbon atom. The present disclosure encompasses all the various stereoisomers of these compounds and mixtures thereof. Mixtures of enantiomers or diastereomers can be designated“(±)” in nomenclature, but a skilled artisan will recognize that a structure can denote a chiral center implicitly. It is understood that graphical depictions of chemical structures, e.g., generic chemical structures, encompass all stereoisomeric forms of the specified compounds, unless indicated otherwise.
Individual enantiomers and diastereomers of compounds of the present disclosure can be prepared synthetically from commercially available starting materials that contain asymmetric or stereogenic centers, or by preparation of racemic mixtures followed by resolution methods well known to those of ordinary skill in the art. These methods of resolution are exemplified by (1) attachment of a mixture of enantiomers to a chiral auxiliary,
separation of the resulting mixture of diastereomers by recrystallization or chromatography and liberation of the optically pure product from the auxiliary, (2) salt formation employing an optically active resolving agent, (3) direct separation of the mixture of optical enantiomers on chiral liquid chromatographic columns, or (4) kinetic resolution using stereoselective chemical or enzymatic reagents. Racemic mixtures also can be resolved into their component enantiomers by well-known methods, such as chiral-phase gas chromatography or crystallizing the compound in a chiral solvent. Stereoselective syntheses, a chemical or enzymatic reaction in which a single reactant forms an unequal mixture of stereoisomers during the creation of a new stereocenter or during the transformation of a pre-existing one, are well known in the art. Stereoselective syntheses encompass both enantio- and diastereoselective transformations.
See, for example, Carreira and Kvaemo, Classics in Stereoselective Synthesis, Wiley-VCH: Weinheim, 2009.
Geometric isomers, resulting from the arrangement of substituents around a carbon- carbon double bond or arrangement of substituents around a cycloalkyl or heterocycloalkyl, can also exist in the compounds of the present disclosure. The symbol . denotes a bond that may be a single, double or triple bond as described herein. Substituents around a carbon-carbon double bond are designated as being in the“Z’ or“E” configuration, where the terms“Z’ and “£” are used in accordance with IUPAC standards. Unless otherwise specified, structures depicting double bonds encompass both the“E” and“Z” isomers.
Substituents around a carbon-carbon double bond alternatively can be referred to as “cis” or“trans,” where“cis” represents substituents on the same side of the double bond and “trans” represents substituents on opposite sides of the double bond. The arrangement of substituents around a carbocyclic ring can also be designated as“cis” or“trans.” The term “cis” represents substituents on the same side of the plane of the ring and the term“trans” represents substituents on opposite sides of the plane of the ring. Mixtures of compounds wherein the substituents are disposed on both the same and opposite sides of plane of the ring are designated“cis/trans.”
The disclosure also embraces isotopically-labeled compounds which are identical to those compounds recited herein, except that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature. Examples of isotopes that can be incorporated into compounds described herein include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, fluorine and chlorine, such as 2H (“D”), 3H, 13C, 14C, 15N, 180, 170, 31P, 32P, 35S, 18F, and 36Cl, respectively. For
example, a compound described herein can have one or more H atoms replaced with deuterium.
Certain isotopically-labeled compounds (e.g., those labeled with 3H and 14C) can be useful in compound and/or substrate tissue distribution assays. Tritiated (i.e., 3H) and carbon- 14 (i.e., 14C) isotopes can be particularly preferred for their ease of preparation and
detectability. Further, substitution with heavier isotopes such as deuterium (i.e., 2H) can afford certain therapeutic advantages resulting from greater metabolic stability (e.g., increased in vivo half-life or reduced dosage requirements) and hence can be preferred in some circumstances. Isotopically-labeled compounds can generally be prepared by following procedures analogous to those disclosed herein, for example, in the Examples section, by substituting an isotopically- labeled reagent for a non-isotopically-labeled reagent.
The phrases“pharmaceutically acceptable” and“pharmacologically acceptable,” as used herein, refer to compounds, molecular entities, compositions, materials, and/or dosage forms that do not produce an adverse, allergic or other untoward reaction when administered to an animal, or a human, as appropriate. For human administration, preparations should meet sterility, pyrogenicity, general safety and purity standards as required by FDA Office of Biologies standards.
The phrases“pharmaceutically acceptable carrier” and“pharmaceutically acceptable excipient,” as used herein, refer to any and all solvents, dispersion media, coatings, isotonic and absorption delaying agents, and the like, that are compatible with pharmaceutical
administration· Pharmaceutical acceptable carriers can include phosphate buffered saline solution, water, emulsions (e.g., such as an oil/water or water/oil emulsions), and various types of wetting agents. The compositions also can include stabilizers and preservatives.
The phrase“pharmaceutical composition,” as used herein, refers to a composition comprising at least one compound as disclosed herein formulated together with one or more pharmaceutically acceptable carriers. The pharmaceutical compositions can also contain other active compounds providing supplemental, additional, or enhanced therapeutic functions.
The terms“individual,”“patient,” and“subject,” as used herein, are used
interchangeably and include any animal, including mammals, preferably mice, rats, other rodents, rabbits, dogs, cats, swine, cattle, sheep, horses, or primates, and more preferably, humans. The compounds described in the disclosure can be administered to a mammal, such as a human, but can also be administered to other mammals such as an animal in need of veterinary treatment, for example, domestic animals (e.g., dogs, cats, and the like), farm
animals (e.g., cows, sheep, pigs, horses, and the like) and laboratory animals (e.g., rats, mice, guinea pigs, and the like). The mammal treated in the methods described in the disclosure is preferably a mammal in which treatment, for example, of pain or depression, is desired.
The term“treating,” as used herein, includes any effect, for example, lessening, reducing, modulating, ameliorating, or eliminating, that results in the improvement of the condition, disease, disorder, and the like, including one or more symptoms thereof. Treating can be curing, improving, or at least partially ameliorating the disorder.
The term“disorder” refers to and is used interchangeably with, the terms“disease,” “condition,” or“illness,” unless otherwise indicated.
The term“modulation,” as used herein, refers to and includes antagonism (e.g., inhibition), agonism, partial antagonism, and/or partial agonism.
The phrase“therapeutically effective amount,” as used herein, refers to the amount of a compound (e.g., a disclosed compound) that will elicit the biological or medical response of a tissue, system, animal or human that is being sought by the researcher, veterinarian, medical doctor or other clinician. The compounds described in the disclosure can be administered in therapeutically effective amounts to treat a disease. A therapeutically effective amount of a compound can be the quantity required to achieve a desired therapeutic and/or prophylactic effect, such as an amount which results in lessening of a symptom of a disease such as depression.
As used herein, the term“pharmaceutically acceptable salt” refers to any salt of an acidic or a basic group that may be present in a compound of the present disclosure, which salt is compatible with pharmaceutical administration· As is known to those of skill in the art, “salts” of the compounds of the present disclosure may be derived from inorganic or organic acids and bases.
Examples of salts include, but are not limited to: acetate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, citrate, camphorate, camphorsulfonate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, fumarate,
flucoheptanoate, glycerophosphate, hemisulfate, heptanoate, hexanoate, hydrochloride, hydrobromide, hydroiodide, 2-hydroxy ethanesulfonate, lactate, maleate, methanesulfonate, 2- naphthalenesulfonate, nicotinate, oxalate, palmoate, pectinate, persulfate, phenylpropionate, picrate, pivalate, propionate, succinate, tartrate, thiocyanate, tosylate, undecanoate, and the like. Other examples of salts include anions of the compounds of the present disclosure compounded
with a suitable cation such as Na+, NH4 +, and NW4 + (where W can be a Ci_4 alkyl group), and the like. For therapeutic use, salts of the compounds of the present disclosure can be pharmaceutically acceptable. However, salts of acids and bases that are non-pharmaceutically acceptable may also find use, for example, in the preparation or purification of a
pharmaceutically acceptable compound.
Compounds included in the present compositions that are basic in nature are capable of forming a wide variety of salts with various inorganic and organic acids. The acids that can be used to prepare pharmaceutically acceptable acid addition salts of such basic compounds are those that form non-toxic acid addition salts, i.e., salts containing pharmacologically acceptable anions, including but not limited to, malate, oxalate, chloride, bromide, iodide, nitrate, sulfate, bisulfate, phosphate, acid phosphate, isonicotinate, acetate, lactate, salicylate, citrate, tartrate, oleate, tannate, pantothenate, bitartrate, ascorbate, succinate, maleate, gentisinate, fumarate, gluconate, glucaronate, saccharate, formate, benzoate, glutamate, methanesulfonate, ethanesulfonate, henzenesul I'onate, -tol uenesul I'onate and pamoate (i.e., l,r-methylene-bis-(2- hydroxy-3-naphthoate)) salts.
Compounds included in the present compositions that are acidic in nature are capable of forming base salts with various pharmacologically acceptable cations. Examples of such salts include alkali metal or alkaline earth metal salts and, particularly, calcium, magnesium, sodium, lithium, zinc, potassium, and iron salts.
Compounds included in the present compositions that include a basic or acidic moiety can also form pharmaceutically acceptable salts with various amino acids. The compounds of the disclosure can contain both acidic and basic groups; for example, one amino and one carboxylic acid group. In such a case, the compound can exist as an acid addition salt, a zwitterion, or a base salt.
The compounds disclosed herein can exist in a solvated form as well as an unsolvated form with pharmaceutically acceptable solvents such as water, ethanol, and the like, and it is intended that the disclosure embrace both solvated and unsolvated forms
Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this disclosure pertains.
Throughout the description, where compositions and kits are described as having, including, or comprising specific components, or where processes and methods are described as
having, including, or comprising specific steps, it is contemplated that, additionally, there are compositions and kits of the present disclosure that consist essentially of, or consist of, the recited components, and that there are processes and methods according to the present disclosure that consist essentially of, or consist of, the recited processing steps.
In the application, where an element or component is said to be included in and/or selected from a list of recited elements or components, it should be understood that the element or component can be any one of the recited elements or components, or the element or component can be selected from a group consisting of two or more of the recited elements or components.
Further, it should be understood that elements and/or features of a composition or a method described herein can be combined in a variety of ways without departing from the spirit and scope of the present disclosure, whether explicit or implicit herein. For example, where reference is made to a particular compound, that compound can be used in various
embodiments of compositions of the present disclosure and/or in methods of the present disclosure, unless otherwise understood from the context. In other words, within this application, embodiments have been described and depicted in a way that enables a clear and concise application to be written and drawn, but it is intended and will be appreciated that embodiments can be variously combined or separated without parting from the present teachings and disclosure(s). For example, it will be appreciated that all features described and depicted herein can be applicable to all aspects of the disclosure(s) described and depicted herein.
The articles“a” and“an” are used in this disclosure to refer to one or more than one (i.e., to at least one) of the grammatical object of the article, unless the context is inappropriate. By way of example,“an element” means one element or more than one element.
The term“and/or” is used in this disclosure to mean either“and” or“or” unless indicated otherwise.
It should be understood that the expression“at least one of’ includes individually each of the recited objects after the expression and the various combinations of two or more of the recited objects unless otherwise understood from the context and use. The expression“and/or” in connection with three or more recited objects should be understood to have the same meaning unless otherwise understood from the context.
The use of the term“include,”“includes,”“including,”“have,”“has,”“having,”
“contain,”“contains,” or“containing,” including grammatical equivalents thereof, should be understood generally as open-ended and non-limiting, for example, not excluding additional unrecited elements or steps, unless otherwise specifically stated or understood from the context.
Where the use of the term“about” is before a quantitative value, the present disclosure also include the specific quantitative value itself, unless specifically stated otherwise. As used herein, the term“about” refers to a ±10% variation from the nominal value unless otherwise indicated or inferred from the context.
Where a percentage is provided with respect to an amount of a component or material in a composition, the percentage should be understood to be a percentage based on weight, unless otherwise stated or understood from the context.
Where a molecular weight is provided and not an absolute value, for example, of a polymer, then the molecular weight should be understood to be an average molecule weight, unless otherwise stated or understood from the context.
It should be understood that the order of steps or order for performing certain actions is immaterial so long as the present disclosure remain operable. Moreover, two or more steps or actions can be conducted simultaneously.
At various places in the present specification, substituents are disclosed in groups or in ranges. It is specifically intended that the description include each and every individual subcombination of the members of such groups and ranges. For example, the term“Ci-6 alkyl” is specifically intended to individually disclose Ci, C2, C3, C4, C5, C6, Ci-C6, C1-C5, C1-C4, Ci- C3, Ci-C2, C2-C6, C2-C5, C2-C4, C2-C3, C3-C6, C3-C5, C3-C4, C4-C6, C4-C5, and C5-C6 alkyl. By way of other examples, an integer in the range of 0 to 40 is specifically intended to individually disclose 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25,
26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, and 40, and an integer in the range of 1 to 20 is specifically intended to individually disclose 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, and 20. Additional examples include that the phrase“optionally substituted with 1-5 substituents” is specifically intended to individually disclose a chemical group that can include 0, 1, 2, 3, 4, 5, 0-5, 0-4, 0-3, 0-2, 0-1, 1-5, 1-4, 1-3, 1-2, 2-5, 2-4, 2-3, 3-5, 3-4, and 4-5 substituents.
The use of any and all examples, or exemplary language herein, for example,“such as” or“including,” is intended merely to illustrate better the present disclosure and does not pose a limitation on the scope of the disclosure unless claimed. No language in the specification
should be construed as indicating any non-claimed element as essential to the practice of the present disclosure.
Further, if a variable is not accompanied by a definition, then the variable is defined as found elsewhere in the disclosure unless understood to be different from the context. In addition, the definition of each variable and/or substituent, for example, C i -C6 alkyl, R2, Rb, w and the like, when it occurs more than once in any structure or compound, can be independent of its definition elsewhere in the same structure or compound.
Definitions of the variables and/or substituents in formulae and/or compounds herein encompass multiple chemical groups. The present disclosure includes embodiments where, for example, i) the definition of a variable and/or substituent is a single chemical group selected from those chemical groups set forth herein, ii) the definition is a collection of two or more of the chemical groups selected from those set forth herein, and iii) the compound is defined by a combination of variables and/or substituents in which the variables and/or substituents are defined by (i) or (ii).
Various aspects of the disclosure are set forth herein under headings and/or in sections for clarity; however, it is understood that all aspects, embodiments, or features of the disclosure described in one particular section are not to be limited to that particular section but rather can apply to any aspect, embodiment, or feature of the present disclosure.
Compounds
Disclosed compounds include a compound represented by Formula I:
or a pharmaceutically acceptable salt and/or a stereoisomer thereof, wherein:
q is 1, 2, 3, or 4;
R5 is independently, for each occurrence, one or more optional substituents selected from the group consisting of Ci-Cealkyl, -S(0)w-Ci_C3alkyl, -NRaRb, and halogen, wherein Ci- C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
R6 is one or two optional substituents bound to one or more available ring carbons, and is independently, for each occurrence, selected from the group consisting of Ci-C6alkyl, - S(0)w-Ci-C3alkyl, -NRaRb, and halogen, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
w is 0, 1, or 2;
R3 is selected from the group consisting of H, phenyl, Ci-Cealkyl, -C(0)R31, and - C(0)0R32, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
R31 and R32 are each independently selected from the group consisting of H, Ci-Cealkyl, -C3-C6cycloalkyl, and phenyl, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs, and phenyl is optionally substituted with one, two, or three substitutents each independently selected from RT;
Ra and Rb are independently, for each occurrence, selected from the group consisting of H, phenyl, Ci-C4alkyl, -Ci-C4alkylene-phenyl, or Ra and Rb taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted with one, two, or three substitutents each independently selected from hydroxyl, - Ci-C3alkoxy, -Ci-C3alkyl, and halogen, and Ci-C3alkyl is optionally substituted with one, two or three halogens;
Rs is independently, for each occurrence, selected from the group consisting of - C(0)NRaRb, -NRaRb, hydroxyl, S(0)w-Ci-C3alkyl, SH, phenyl and halogen; and
RT is independently, for each occurrence, selected from the group consisting of -Ci- C6alkyl, hydroxyl, Ci-C6alkoxy, and halogen.
In certain embodiments, q can be 1, 2 or 3. In certain embodiments, q can be 2. In certain embodiments, q can be 3. In certain embodiments, q can be 1.
In certain embodiments, R5 can be absent. In some embodiments, R5 can be present one, two, three, or four times, and can be selected from the group consisting of Ci-Cealkyl, - S(0)w-Ci-C3alkyl, -NRaRb, and halogen, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs.
In certain embodiments, R6 can be absent.
In certain embodiments, R3 can be Ci-Cealkyl, wherein Ci-C6alkyl is optionally substituted by one, two, or three substituents each independently selected from -C(0)-NRaRb, and hydroxyl; and Ra and Rb are each independently selected for each occurrence from the group consisting of H, Ci-C4alkyl, and -Ci-C4alkylene -phenyl, wherein phenyl is optionally
substituted by one two or three substituents each independently selected from Ci-C alkoxy, Ci- C3alkyl, and halogen, and C]-C3alkyl is optionally substituted with one, two, or three halogens independently selected from F, Cl, and Br.
In certain embodiments, R3 can be -C(0)-R31, wherein R31 is Ci-C6alkyl, and Ci- C6alkyl is optionally substituted by one, two, or three substituents each independently selected from -C(0)-NRaRb, -NRaRb, and hydroxyl; and Ra and Rb are each independently selected for each occurrence from the group consisting of H and Ci-C alkyl.
In certain embodiments, R3 can be selected from the group consisting of:
wherein Ra and Rb can be each independently selected for each occurrence from the group consisting of H and -Ci-C alkyl.
In certain embodiments, R3 can be selected from the group consisting of:
wherein Ra and Rb can be each independently selected for each occurrence from the group consisting of H and -Ci-C alkyl, and R8 can be one, two or three optional substituents each independently selected from the group consisting of hydroxyl, Br, Cl, F, methoxy, and -CF3
In certain embodiments, Ra and Rb can be H.
In certain embodiments, Ra can be H or methyl, and Rb can be selected from the group consisting of methyl, ethyl, isobutyl, and -CFF-phenyl, wherein phenyl can be optionally substituted by one, two, or three substituents each independently selected from F, methoxy, and CF3.
Disclosed compounds also include a compound represented by Formula II:
or a pharmaceutically acceptable salt and/or a stereoisomer thereof, wherein:
R3 is selected from the group consisting of H, -C(0)-Ci-C6alkyl, and -Ci-C6alkyl-C(0)- NRaRb; wherein Ci-C6alkyl is optionally substituted by one, two, or three substituents each independently selected from halogen, hydroxyl, cyano, -CO2H, phenyl, -C(0)-NRaRb, and NRaRb; and
Ra and Rb are each independently for each occurrence selected from the group consisting of H, phenyl, Ci-C4alkyl, -Ci-C4alkylene -phenyl, or Ra and Rb taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted by one two or three substituents each independently selected from Ci- C3alkoxy, Ci-C3alkyl, and halogen, wherein Ci-C3alkyl is optionally substituted with one, two or three substituents independently selected from F, Cl, and Br.
In some embodiments, R3 can be selected from the group consisting of:
wherein Ra and Rb can be each independently selected for each occurrence from the group consisting of H and -Ci-C4alkyl.
In some embodiments, R3 can be selected from the group consisting of:
wherein Ra and Rb can be each independently selected for each occurrence from the group consisting of H and -Ci-C4alkyl, and R8 can be one, two, or three optional substituents each independently selected from the group consisting of hydroxyl, Br, Cl, F, methoxy, and -CF3.
In some embodiments, Ra and Rb can be H.
In some embodiments, Ra can be H or methyl, and Rb can be selected from the group consisting of methyl, ethyl, isobutyl, and -CH2-phenyl, wherein phenyl can be optionally substituted by one, two, or three substituents each independently selected from F, methoxy, and -CF3.
In certain embodiments, R3 independently can be an amino acid or a derivative of an amino acid, for example, an alpha“amino amide” represented by l¾N-CF[(amino acid side chain)-C(0)NH2. In certain embodiments, the nitrogen atom of the amino group of the amino acid or the amino acid derivative is a ring nitrogen in a chemical formula described herein. In such embodiments, the carboxylic acid of the amino acid or the amide group of an amino amide (amino acid derivative) is not within the ring structure, i.e., not a ring atom. In certain embodiments, the carboxylic acid group of the amino acid or the amino acid derivative forms an amide bond with a ring nitrogen in a chemical formula disclosed herein, thereby providing an amino amide, where the amino group of the amino amide is not within the ring structure, i.e., not a ring atom. In certain embodiments, R3 independently can be an alpha amino acid, an alpha amino acid derivative, and/or another amino acid or amino acid derivative such as a beta amino acid or a beta amino acid derivative, for example, a beta amino amide. In some embodiments, a disclosed compound is selected from the compounds delineated in the Examples or in Table 1 herein, and includes a pharmaceutically acceptable salt and/or a stereoisomer thereof.
In particular embodiments, a disclosed compound is selected from the group consisting of:
or a pharmaceutically acceptable salt, a stereoisomer, and/or an N-oxide thereof.
The compounds of the present disclosure and formulations thereof may have a plurality
of chiral centers. Each chiral center may be independently R, S, or any mixture of R and S. For example, in some embodiments, a chiral center may have an R:S ratio of between about 100:0 and about 50:50 (“racemate”), between about 100:0 and about 75:25, between about 100:0 and about 85:15, between about 100:0 and about 90:10, between about 100:0 and about 95:5, between about 100:0 and about 98:2, between about 100:0 and about 99:1, between about 0:100 and 50:50, between about 0: 100 and about 25:75, between about 0:100 and about 15:85, between about 0:100 and about 10:90, between about 0:100 and about 5:95, between about 0:100 and about 2:98, between about 0:100 and about 1:99, between about 75:25 and 25:75, and about 50:50. Formulations of the disclosed compounds comprising a greater ratio of one or more isomers (i.e., R and/or S ) may possess enhanced therapeutic characteristic relative to racemic formulations of a disclosed compounds or mixture of compounds. In some instances, chemical formulas contain the descriptor“-(R)-” or“-(S)-” that is further attached to solid wedge or dashed wedge. This descriptor is intended to show a methine carbon (CH) that is attached to three other substituents and has either the indicated R or S configuration.
Disclosed compounds may provide for efficient cation channel opening at the NMDA receptor, e.g. may bind or associate with the glutamate site or glycine site or other modulatory site of the NMDA receptor to assist in opening the cation channel. The disclosed compounds may be used to regulate (turn on or turn off) the NMDA receptor through action as an agonist or antagonist.
The compounds described herein, in some embodiments, may bind to a specific NMDA receptor subtypes. For example, a disclosed compound may bind to one NMDA subtype and not another. In other embodiments, a disclosed compound may bind to one, or more than one NMDA subtype, and/or may have substantially less (or substantial no) binding activity to certain other NMDA subtypes. For example, in some embodiments, a disclosed compound binds to NR2A with substantially no binding to NR2D. In some embodiments, a disclosed compound binds to NR2B and NR2D with substantially lower binding to NR2A and NR2C.
The compounds as described herein may bind to NMDA receptors. A disclosed compound may bind to the NMDA receptor resulting in agonist- like activity (facilitation) over a certain dosing range and/or may bind to the NMDA receptor resulting in antagonist- like activity (inhibition) over a certain dosing range. In some embodiments, a disclosed compound may possess a potency that is lO-fold or greater than the activity of existing NMDA receptor modulators.
The disclosed compounds may exhibit a high therapeutic index. The therapeutic index,
as used herein, refers to the ratio of the dose that produces a toxicity in 50% of the population (i.e., TD50) to the minimum effective dose for 50% of the population (i.e., ED50). Thus, the therapeutic index = (TD5o):(ED5o). In some embodiments, a disclosed compound may have a therapeutic index of at least about 10:1, at least about 50:1, at least about 100:1, at least about 200:1, at least about 500:1, or at least about 1000:1.
Compositions
In other aspects of this disclosure, a pharmaceutical formulation or a pharmaceutical composition including a disclosed compound and a pharmaceutically acceptable excipient are provided. In some embodiments, a pharmaceutical composition includes a racemic mixture or a varied stereoisomeric mixture of one or more of the disclosed compounds.
A formulation can be prepared in any of a variety of forms for use such as for administering an active agent to a patient, who may be in need thereof, as are known in the pharmaceutical arts. For example, the pharmaceutical compositions of the present disclosure can be formulated for administration in solid or liquid form, including those adapted for the following: (1) oral administration, for example, drenches (aqueous or non-aqueous solutions or suspensions), tablets (e.g., those targeted for buccal, sublingual, and/or systemic absorption), boluses, powders, granules, and pastes for application to the tongue; (2) parenteral
administration by, for example, subcutaneous, intramuscular, intraperitoneal, intravenous or epidural injection as, for example, a sterile solution or suspension, or sustained-release formulation; (3) topical administration, for example, as a cream, ointment, or a controlled- release patch or spray applied to the skin; (4) intravaginal or intrarectal administration, for example, as a pessary, cream or foam; (5) sublingual administration; (6) ocular administration; (7) transdermal administration; or (8) nasal administration.
For example, pharmaceutical compositions of the disclosure can be suitable for delivery to the eye, i.e., ocularly. Related methods can include administering a therapeutically effective amount of a disclosed compound or a pharmaceutical composition including a disclosed compound to a patient in need thereof, for example, to an eye of the patient, where
administering can be topically, subconjunctivally, subtenonly, intravitreally, retrobulbarly, peribulbarly, intracomerally, and/or systemically.
Amounts of a disclosed compound as described herein in a formulation may vary according to factors such as the disease state, age, sex, and weight of the individual. Dosage regimens may be adjusted to provide the optimum therapeutic response. For example, a single
bolus may be administered, several divided doses may be administered over time or the dose may be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation. It is especially advantageous to formulate parenteral compositions in dosage unit form for ease of administration and uniformity of dosage. Dosage unit form as used herein refers to physically discrete units suited as unitary dosages for the mammalian subjects to be treated; each unit containing a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier.
The specification for the dosage unit forms are dictated by and directly dependent on (a) the unique characteristics of the compound selected and the particular therapeutic effect to be achieved, and (b) the limitations inherent in the art of compounding such an active compound for the treatment of sensitivity in individuals.
Therapeutic compositions typically must be sterile and stable under the conditions of manufacture and storage. The composition can be formulated as a solution, microemulsion, liposome, or other ordered structure suitable to high drug concentration. The carrier can be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyethylene glycol, and the like), and suitable mixtures thereof. The proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants. In many cases, it will be preferable to include isotonic agents, for example, sugars, polyalcohols such as mannitol, sorbitol, or sodium chloride in the composition.
Prolonged absorption of the injectable compositions can be brought about by including in the composition an agent which delays absorption, for example, monostearate salts and gelatin.
The compounds can be administered in a time release formulation, for example in a composition which includes a slow release polymer. The compounds can be prepared with carriers that will protect the compound against rapid release, such as a controlled release formulation, including implants and microencapsulated delivery systems. Biodegradable, biocompatible polymers can be used, such as ethylene vinyl acetate, poly anhydrides, polyglycolic acid, collagen, polyorthoesters, polylactic acid and polylactic, polyglycolic copolymers (PLG). Many methods for the preparation of such formulations are generally known to those skilled in the art.
Sterile injectable solutions can be prepared by incorporating the compound in the required amount in an appropriate solvent with one or a combination of ingredients enumerated above, as required, followed by filtered sterilization. Generally, dispersions are prepared by
incorporating the active compound into a sterile vehicle which contains a basic dispersion medium and the required other ingredients from those enumerated above. In the case of sterile powders for the preparation of sterile injectable solutions, the preferred methods of preparation are vacuum drying and freeze-drying which yields a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
In some embodiments, a compound can be formulated with one or more additional compounds that enhance the solubility of the compound.
Methods
Methods of the disclosure for treating a condition in a patient in need thereof generally include administering a therapeutically effective amount of a compound described herein or a composition including such a compound. In some embodiments, the condition may be a mental condition. For example, a mental illness may be treated. In some embodiments, a nervous system condition may be treated. For example, a condition that affects the central nervous system, the peripheral nervous system, and/or the eye may be treated. In some embodiments, neurodegenerative diseases may be treated.
In some embodiments, the methods include administering a compound to treat patients suffering from autism, anxiety, depression, bipolar disorder, attention deficit disorder, attention deficit hyperactivity disorder (ADHD), schizophrenia, a psychotic disorder, a psychotic symptom, social withdrawal, obsessive-compulsive disorder (OCD), phobia, post-traumatic stress syndrome, a behavior disorder, an impulse control disorder, a substance abuse disorder (e.g., a withdrawal symptom, opiate addiction, nicotine addiction, and ethanol addition), a sleep disorder, a memory disorder (e.g., a deficit, loss, or reduced ability to make new memories), a learning disorder, urinary incontinence, multiple system atrophy, progressive supra-nuclear palsy, Friedrich's ataxia, Down’s syndrome, fragile X syndrome, tuberous sclerosis, olivio- ponto-cerebellar atrophy, cerebral palsy, drug-induced optic neuritis, ischemic retinopathy, diabetic retinopathy, glaucoma, dementia, AIDS dementia, Alzheimer’s disease, Huntington’s chorea, spasticity, myoclonus, muscle spasm, infantile spasm, Tourette's syndrome, epilepsy, cerebral ischemia, stroke, a brain tumor, traumatic brain injury, cardiac arrest, myelopathy, spinal cord injury, peripheral neuropathy, acute neuropathic pain, and chronic neuropathic pain.
In some embodiments, the present disclosure provides methods of treating a cognitive impairment disorder, for example, a dysfunction in learning and/or memory such as that seen in age-related cognitive decline, Lewy body dementia, AIDS dementia, HIV dementia, vascular
dementia, mild cognitive impairment in Huntington’s disease, Huntington’s disease dementia, mild cognitive impairment in Parkinson’s disease, Parkinson’s disease dementia, mild cognitive impairment in Alzheimer’s disease, Alzheimer’s dementia, frontotemporal dementia, cognitive impairment associated with schizophrenia (CIAS), and cognitive impairment associated with seizures, stroke, cerebral ischemia, hypoglycemia, cardiac arrest, migraine, multiple sclerosis, traumatic brain injury, and/or Down’s syndrome.
In certain embodiments, methods for treating schizophrenia are provided. For example, paranoid type schizophrenia, disorganized type schizophrenia (i.e., hebephrenic schizophrenia), catatonic type schizophrenia, undifferentiated type schizophrenia, residual type schizophrenia, post-schizophrenic depression, and simple schizophrenia may be treated using the methods and compositions disclosed herein. Psychotic disorders such as schizoaffective disorders, delusional disorders, brief psychotic disorders, shared psychotic disorders, and psychotic disorders with delusions or hallucinations may also be treated using the compositions disclosed herein.
Paranoid schizophrenia may be characterized where delusions or auditory hallucinations are present, but thought disorder, disorganized behavior, or affective flattening are not.
Delusions may be persecutory and/or grandiose, but in addition to these, other themes such as jealousy, religiosity, or somatization may also be present. Disorganized type schizophrenia may be characterized where thought disorder and flat affect are present together. Catatonic type schizophrenia may be characterized where the patient may be almost immobile or exhibit agitated, purposeless movement. Symptoms can include catatonic stupor and waxy flexibility. Undifferentiated type schizophrenia may be characterized where psychotic symptoms are present but the criteria for paranoid, disorganized, or catatonic types have not been met.
Residual type schizophrenia may be characterized where positive symptoms are present at a low intensity only. Post-schizophrenic depression may be characterized where a depressive episode arises in the aftermath of a schizophrenic illness where some low-level schizophrenic symptoms may still be present. Simple schizophrenia may be characterized by insidious and progressive development of prominent negative symptoms with no history of psychotic episodes.
In some embodiments, methods are provided for treating psychotic symptoms that may be present in other mental disorders, including, but not limited to, bipolar disorder, borderline personality disorder, drug intoxication, and drug-induced psychosis. In other embodiments, methods for treating delusions (e.g., "non-bizarre") that may be present in, for example,
delusional disorder are provided.
In various embodiments, methods for treating social withdrawal in conditions including, but not limited to, social anxiety disorder, avoidant personality disorder, and schizotypal personality disorder are provided.
In some embodiments, the disclosure provides methods for treating a
neurodevelopmental disorder related to synaptic dysfunction in a patient in need thereof, where the methods generally include administering to the patient a therapeutically effective amount of a disclosed compound, or a pharmaceutical composition including a disclosed compound. In certain embodiments, the neurodevelopmental disorder related to synaptic dysfunction can be Rett syndrome also known as cerebroatrophic hyperammonemia, MECP2 duplication syndrome (e.g., a MECP2 disorder), CDKL5 syndrome, fragile X syndrome (e.g., a FMR1 disorder), tuberous sclerosis (e.g., a TSC1 disorder and/or a TSC2 disorder), neurofibromatosis (e.g., a NF1 disorder), Angelman syndrome (e.g., a UBE3A disorder), the PTEN hamartoma tumor syndrome, Phelan-McDermid syndrome (e.g., a SHANK3 disorder), or infantile spasms. In particular embodiments, the neurodevelopmental disorder can be caused by mutations in the neuroligin (e.g., a NLGN3 disorder and/or a NLGN2 disorder) and/or the neurexin (e.g., a NRXN1 disorder).
In some embodiments, methods are provided for treating neuropathic pain. The neuropathic pain can be acute or chronic. In some cases, the neuropathic pain can be associated with a condition such as herpes, HIV, traumatic nerve injury, stroke, post- ischemia, chronic back pain, post-herpetic neuralgia, fibromyalgia, reflex sympathetic dystrophy, complex regional pain syndrome, spinal cord injury, sciatica, phantom limb pain, diabetic neuropathy such as diabetic peripheral neuropathy (“DPN”), and cancer chemotherapeutic-induced neuropathic pain. In certain embodiments, methods for enhancing pain relief and for providing analgesia to a patient are also provided.
Further methods include a method of treating autism and/or an autism spectrum disorder in a patient need thereof, comprising administering an effective amount of a compound to the patient. In other embodiments, a method for reducing the symptoms of autism in a patient in need thereof comprises administering an effective amount of a disclosed compound to the patient. For example, upon administration, the compound may decrease the incidence of one or more symptoms of autism such as eye contact avoidance, failure to socialize, attention deficit, poor mood, hyperactivity, abnormal sound sensitivity, inappropriate speech, disrupted sleep, and perseveration. Such decreased incidence may be measured relative to the incidence in the
untreated individual or an untreated individual(s).
Also provided herein is a method of modulating an autism target gene expression in a cell comprising contacting a cell with an effective amount of a compound described herein.
The autism gene expression may be for example, selected from ABAT, APOE, CHRNA4, GABRA5,GFAP, GRIN2A, PDYN, and PENK. In some embodiments, a method of modulating synaptic plasticity in a patient suffering from a synaptic plasticity related disorder is provided, comprising administering to the patient an effective amount of a compound.
In other embodiments, a method of treating Alzheimer’s disease, or e.g., treatment of memory loss that e.g., accompanies early stage Alzheimer’s disease, in a patient in need thereof is provided, comprising administering a compound. Also provided herein is a method of modulating an Alzheimer’s amyloid protein (e.g., beta amyloid peptide, e.g. the isoform Abi_ 42), in-vitro or in-vivo (e.g. in a cell) comprising contacting the protein with an effective amount of a compound is disclosed. For example, in some embodiments, a compound may block the ability of such amyloid protein to inhibit long-term potentiation in hippocampal slices as well as apoptotic neuronal cell death. In some embodiments, a disclosed compound may provide neuroprotective properties to a Alzheimer’s patient in need thereof, for example, may provide a therapeutic effect on later stage Alzheimer’s -associated neuronal cell death.
In certain embodiments, the disclosed methods include treating a psychosis or a pseudobulbar affect (“PBA”) that is induced by another condition such as a stroke, amyotrophic lateral sclerosis (ALS or Lou Gehrig’s disease), multiple sclerosis, traumatic brain injury, Alzheimer’s disease, dementia, and/or Parkinson’s disease. Such methods, as with other methods of the disclosure, include administration of a therapeutically effective amount of a disclosed compound to a patient in need thereof.
In certain embodiments, a method of treating depression includes administering a therapeutically effective amount of a compound described herein. In some embodiments, the treatment may relieve depression or a symptom of depression without affecting behavior or motor coordination and without inducing or promoting seizure activity. Exemplary depression conditions that are expected to be treated according to this aspect include, but are not limited to, major depressive disorder, dysthymic disorder, psychotic depression, postpartum depression, premenstrual syndrome, premenstrual dysphoric disorder, seasonal affective disorder (SAD), bipolar disorder (or manic depressive disorder), mood disorder, and depressions caused by chronic medical conditions such as cancer or chronic pain, chemotherapy, chronic stress, and post traumatic stress disorders. In addition, patients suffering from any form of depression
often experience anxiety. Various symptoms associated with anxiety include fear, panic, heart palpitations, shortness of breath, fatigue, nausea, and headaches among others. Anxiety or any of the symptoms thereof may be treated by administering a compound as described herein.
Also provided herein are methods of treating a condition in treatment-resistant patients, e.g., patients suffering from a mental or central nervous system condition that does not, and/or has not, responded to adequate courses of at least one, or at least two, other compounds or therapeutics. For example, provided herein is a method of treating depression in a treatment resistant patient, comprising a) optionally identifying the patient as treatment resistant and b) administering an effective dose of a compound to said patient.
In some embodiments, a compound described herein may be used for acute care of a patient. For example, a compound may be administered to a patient to treat a particular episode (e.g., a severe episode) of a condition disclosed herein.
Also provided herein are combination therapies comprising a compound of the disclosure in combination with one or more other active agents. For example, a compound may be combined with one or more antidepressants, such as tricyclic antidepressants, MAO-I's, SSRI's, and double and triple uptake inhibitors and/or anxiolytic drugs. Exemplary drugs that may be used in combination with a compound include Anafranil, Adapin, Aventyl, Elavil, Norpramin, Pamelor, Pertofrane, Sinequan, Surmontil, Tofranil, Vivactil, Parnate, Nardil, Marplan, Celexa, Lexapro, Luvox, Paxil, Prozac, Zoloft, Wellbutrin, Effexor, Remeron, Cymbalta, Desyrel (trazodone), and Ludiomill. In another example, a compound may be combined with an antipsychotic medication. Non-limiting examples of antipsychotics include butyrophenones, phenothiazines, thioxanthenes, clozapine, olanzapine, risperidone, quetiapine, ziprasidone, amisulpride, asenapine, paliperidone, iloperidone, zotepine, sertindole, lurasidone, and aripiprazole. It should be understood that combinations of a compound and one or more of the above therapeutics may be used for treatment of any suitable condition and are not limited to use as antidepressants or antipsychotics.
EXAMPLES
The following examples are provided for illustrative purposes only, and are not intended to limit the scope of the disclosure.
The following abbreviations may be used herein and have the indicated definitions: Ac is acetyl (-C(0)CH3), AIDS is acquired immune deficiency syndrome, Boc and BOC are tert- butoxycarbonyl, Boo20 is di-ieri-butyl dicarbonate, Bn is benzyl, Cbz is carboxybenzyl, DCM
is dichloromethane, DCE is l,2-dichloroethane, DEA is diethylamine, DIPEA is NN- diisopropylethylamine, DMF is /V,/V-dirnethylformamide, DMSO is dimethyl sulfoxide, EDC is l-ethyl-3-(3-dimethylaminopropyl)carbodiimide, ESI is electrospray ionization, EtOAc is ethyl acetate, h is hour, HATU is 2-(7-aza- 1 //-benzotri azole- 1 -yl)- 1 , 1 ,3,3-tetramethyluronium hexafluorophosphate, HIV is human immunodeficiency virus, HOBT is hydroxybenzotriazole, HPLC is high performance liquid chromatography, LCMS is liquid chromatography/mass spectrometry, LiHMDS is lithium hexamethyldisilazane, NMDAR is N-methyl-d-aspartate receptor, NMR is nuclear magnetic resonance, Pd/C is palladium on carbon, RT is room temperature (e.g., from about 20 °C to about 25 °C), T3P is propylphosphonic anhydride, TEA is triethylamine, TLC is thin layer chromatography, TFA is trifluoroacetic acid, THF is tetrahydrofuran, TMS is trimethylsilyl, and Ts is tosyl or para-lo] uenesul fonyl .
A. SYNTHESIS OF COMPOUNDS
Synthesis of A A & AB:
Synthesis of ethyl 2-(l-formylcyclohexyl)acetate (1):
To a stirred solution of compound SM-1 (50.0 g, 446.4 mmol) in toluene (400 mL) was added diisobutylamine (84.1 mL, 481.6 mmol) and the reaction mixture was heated at 110°C in a Dean-Stark apparatus for 2 h. The reaction mixture was concentrated under reduced pressure. The residue in acetonitrile (450 mL) was added compound SM-2 (87.9 g, 526.7 mmol) at room temperature and the reaction mixture was heated at 110°C for 14 h. A solution of acetic acid (45 mL, 0.60 mmol) and sodium acetate (21.9 g, 267.5 mmol) in water (240 ml) was added at room temperature, and reaction mixture was further heated to reflux for 4 h. After consumption
of the starting material (by TLC), reaction mixture was concentrated under reduced pressure. The reaction mixture was diluted with water and extracted with diethyl ether (3 x 400 mL). The combined organic layer was washed with brine (300 mL), dried over Na SCL and concentrated under reduced pressure. The crude was purified by flash column chromatography using eluent with 5% EtOAc/hexane to afford compound 1 (35.0 g, 39%) as a brown oil.
LCMS (ESI): m/z 199 [M++l]
Synthesis of benzyl 3-(benzyloxy)-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)propanoate (2):
To a stirred solution of compound 1 (2.20 g, 11.10 mmol) in dichloroethane (50 mL) was added Int-D (3.16 g, 11.10 mmol), acetic acid (3.8 mL) and stirred for 2 h. NaCNBfL (2.16 g, 33.30 mmol) was added and stirred at room temperature for 16 h. After consumption of the starting material (by TLC), reaction mixture was quenched with saturated NaHCCL solution (30 mL) and extracted with DCM (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over Na SCL and concentrated under reduced pressure. The crude was purified by column chromatography by using 50% EtOAc/hexane to afford compound 2 (1.0 g, 45%) an off white solid.
LCMS (ESI): m/z 422.05 [M++l]
Synthesis of 3-(benzyloxy)-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)propanoic acid (3):
To a stirred solution of compound 2 (1.0 g, 2.37 mmol) in EtOAc (20 mL), 10% Pd/C (50% wet, 0.20 g) was added at room temperature and stirred under hydrogen atmosphere (balloon) for 16 h. After consumption of the starting material (by TLC), the reaction mixture was filtered through a pad of celite and washed with EtOAc (40 mL). The filtrate was concentrated under reduced pressure and washed with pentane to afford compound 3 (0.70 g, crude) as an off white solid.
LCMS (ESI): m/z 331.95 [M++l]
Synthesis of 3-(benzyloxy)-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)propanamide (4):
To a stirred solution of compound 3 (0.7 g, 2.11 mmol) in CH2CI2 (30 mL), EDCI.HC1 (0.61 g, 3.16 mmol), HOBt (0.43g, 3.16 mmol), DIPEA (0.87 gm, 6.33 mmol), NH4Cl (0.34 g, 6.33 mmol), were added at 0 °C and reaction mixture stirred at room temperature for 16 h.
After consumption of the starting material (by TLC), reaction mixture was quenched with water (30 mL) and extracted with CH2CI2 (3 x 20 mL). The combined organic layer was washed with brine (20 mL), dried over Na2S04 and concentrated under reduced pressure. The crude was purified by column chromatography using 5% methanol/ CH2CI2 to afford compound 4 (0.40g, 57%) as an off white solid.
LCMS (ESI): m/z 331.05 [M++l]
Synthesis of 3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)propanamide (AA & AB):
To a stirred solution of compound 4 (0.40 g, 1.21 mmol) in MeOH (20 mL), 10% Pd/C (50% wet, 0.20 g) was added at room temperature and stirred under hydrogen atmosphere (balloon) for 16 h. After consumption of the starting material (by TLC), the reaction mixture was filtered through a pad of celite and washed with MeOH (30 mL). The filtrate was concentrated under reduced pressure to afford mixture of compounds AA & AB (0.300 g,) as an off white solid. The mixture was purified by preparative HPLC followed by chiral preparative HPLC to afford AA (0.12 g) and AB (0.11 g) as off white solids.
AA
NMR (400 MHz, DMSO-ifc) d 7.31 (s, 1H), 7.05 (s, 1H), 4.87 (br.s, 1H), 4.46 - 4.43 (m, 1H), 3.74 - 3.70 (m, 1H), 3.61 - 3.56 (m, 1H), 3.28 (d, 7 = 10 Hz„ 1H), 3.17 (d, J = 9.6 Hz„ 1H), 2.16-2.07 (m, 2H), 1.5-1.3 (m, 10H). LCMS (ESI): m/z 241.0 [M++l] HPLC: 98.91%. Chiral HPLC: >99%; Column: YMC CHIRALART CELLULOSE-SC (250*4.6 mm, 5u); Mobile Phase: A: 77-hexane + 0.1% TLA; Mobile Phase: B: Iso-propyl alcohol; Plow rate : 1.0 mL/min; Retention time: 7.35 min.
AB
MHz, DMSO-76): d 7.32 (s, 1H), 7.05 (s, 1H), 5.32 (br.s, 1H), 4.46 - 4.43 (m, 1H), 3.74 - 3.70 (m, 1H), 3.61 - 3.56 (m, 1H), 3.28 (d, J = 9.2 Hz„ 1H), 3.16 (d, J = 9.6 Hz„ 1H), 2.16-2.07 (m„ 2H), 1.43 - 1.31 (m, 10H). LCMS (ESI): m/z 241.0 [M++l], HPLC:
99.68%. Chiral HPLC: 99.78%; Column: YMC CHIRALART CELLULOSE-SC (250*4.6 mm, 5u); Mobile Phase: A: /7-hexane + 0.1% TLA; Mobile Phase: B: Iso-propyl alcohol; Plow rate : 1.0 mL/min; Retention time : 9.64 min.
Intermediates:
Synthesis of (S)-2-((tert-butoxy carbonyl) amino)-3-hydroxypropanoic acid (A):
To a stirring solution of L-serine (76 g, 723 mmol) in 1, 4 dioxane and H20 (6500 mL, 3.5:3) were added NaOH (61 g, 1.51 mmol), Boc20 (190 mL, 868 mmol) at 0 °C and the reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), the reaction mixture was acidified with aqueous 2N HC1 (pH~4) and extracted with EtOAc (5 x 500 mL). The combined organic layer was dried over anhydrous Na2S04 and concentrated under reduced pressure to afford Int-A (100 g, 67.5%) as a yellow syrup.
Synthesis of (S)-3-(benzyloxy)-2-((fert-butoxycarbonyl) amino) propanoic acid (B):
To a stirring solution of Int-A (50 g, 245 mmol) in DMF (650 mL) was added NaH (60%, 23 g, 563 mmol) at -15 °C and stirred for 2 h. After adding benzyl bromide (32.8 mL, 269 mmol) slowly, the reaction mixture was warmed to room temperature and stirred for 12 h. After consumption of the starting material (by TLC), the reaction mixture was poured into chilled water (200 mL) and extracted with diethyl ether (2x 250 mL). The aqueous layer was acidified with citric acid (pH~4) and extracted with EtOAc (2 x 500 mL). The combined organic layers were washed with water (3 x 250 mL). The organic extracts were dried over anhydrous Na SCL, filtered and concentrated under reduced pressure to afford Int-B (54 g, 75%) as a brown syrup.
7.26 (m, 5H), 5.43 (d, J = 7.6 Hz, 1H), 4.70-4.46 (m, 1H), 4.45 (s, 2H), 4.13-3.91 (m, 1H), 3.73-3.70 (m, 1H), 1.44 (s, 9H).
Synthesis of (S)-benzyl 3-(benzyloxy)-2-((fert-butoxy carbonyl) amino) propanoate (C):
To a stirring solution of Int-B (36 g, 122 mmol) in DMF (250 mL) was added Na CCL (20 g, 183 mmol) at 0 °C and added benzyl bromide (18 mL, 146 mmol) slowly and the reaction mixture was warmed to room temperature and stirred for 12 h. After consumption of the starting material (by TLC), the reaction mixture was poured into chilled water (200 mL) and extracted with diethyl ether (2 x 250 mL). The combined organic layers were washed with water (3 x 250 mL), dried over anhydrous Na2S04, filtered and concentrated under reduced pressure to afford Int-C (42 g, 91%) as a brown syrup was used directly for next step without any purification.
Synthesis of (S)-benzyl 2-amino-3-(benzyloxy) propanoate hydrochloride (D):
To a stirring solution of Int-C (10 g, 25.9 mmol) in HC1 in diethyl ether (50 mL) was added at 0 °C and stirred at room temperature for 12 h. The obtained precipitate was filtered and triturated with diethyl ether (2 x 100 mL). The filtered compound was dried under vacuum to afford Int-D (5 g, 60%) as a white solid.
4.57-4.44 (m, 3H), 3.91-3.81 (m, 2H).
The experimental procedure for the synthesis of compound 1 is captured under the synthesis of AA and AB (as compound 1).
Synthesis of dibenzyl 2-(3-oxo-2-azaspiro[4.5]decan-2-yl)pentanedioate (2):
To a stirred solution of compound 1 (5.50 g, 27.7 mmol) in dichloroethane (100 mL) was added Int-C (9.07 g, 27.7 mmol), acetic acid (0.5 mL) and stirred for 2 h. NaCNBPL (5.50 g, 83.2 mmol) was added to the reaction mixture and stirred at room temperature for 14 h. After consumption of the starting material (by TLC), reaction mixture was quenched with saturated NaHCCL solution (75 mL) and extracted with DCM (3 x 100 mL). The combined organic layer was washed with brine (100 mL), dried over Na2S04 and concentrated under reduced pressure. The crude was purified by flash column chromatography using 50% EtO Ac/hexane to afford compound 2 (2.50 g, 43%) as an off white solid.
LCMS (ESI): m/z 464.3 [M++l]
Synthesis of 2-(3-oxo-2-azaspiro[4.5]decan-2-yl)pentanedioic acid (3):
To a stirred solution of compound 2 (1.90 g, 4.10 mmol) in EtOAc (50 mL), 10% Pd/C (50% wet, 1.5 g) was added at room temperature and stirred under hydrogen atmosphere (balloon) for 12 h. After consumption of the starting material (by TLC), the reaction mixture was filtered through a pad of celite and washed with EtOAc (70 mL). The filtrate was concentrated under reduced pressure to afford compound 3 (1.10 g, crude) as an off white solid.
LCMS (ESI): m/z 284 [M++l]
Synthesis of 2-(3-oxo-2-azaspiro[4.5]decan-2-yl)pentanediamide (AC):
To a stirred solution of compound 3 (0.8 g, 2.82 mmol) in DCM (30 mL), EDCI.HC1 (1.62 g, 8.47 mmol), HOBt (1.14 g, 8.47 mmol), DIPEA (2.94 mL, 16.9 mmol), NH4Cl (0.91 g, 16.9 mmol), were added at 0 °C and reaction mixture stirred at room temperature for 16 h.
After consumption of the starting material (by TLC), reaction mixture was quenched with water (30 mL) and extracted with DCM (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over Na SCL and concentrated under reduced pressure. The crude was purified by column chromatography using 5% methanol/DCM to afford racemic mixture of AC (0.51 g) as an off white solid. The racemic mixture was purified by preparative HPLC followed by chiral HPLC to afford AC (0.17 g) as an off white solid.
7.32 (s, 1H), 7.27 (s, 1H), 7.04 (s, 1H), 6.75 (s, 1H), 4.39 - 4.36 (m, 1H), 3.23 (d, J = 9.6 Hz„ 1H), 3.05 (d, J = 9.6 Hz„ 1H), 2.14-2.16 (m, 2H),
1.96 - 1.93 (m, 3H), 1.7-1.8 (m, 1H), 1.42 - 1.38 (m, 10H). LCMS (ESI): m/z 282.0 [M++l] HPLC: 97.44%. Chiral HPLC: 97.74%; Column: YMC CHIRALART CELLULOSE-SC (250*4.6 mm, 5pm); Mobile Phase: A: 77-hexane + 0.1% Iso-propyl amine; Mobile Phase: B: IPA; Plow rate : 1.0 mL/min; Retention time : 20.70 min.
Intermediates:
Synthesis of (feri-butoxycarbonyl)- -glutamic acid (A):
To a stirring solution of L-glutamic acid (100 g, 680 mmol) in 1, 4 dioxane and ¾0 (700 mL, 4:3) were added NaOH (81.6 g, 2.04 mol), Boo20 (178 g, 816 mmol) at 0 °C and the reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), the reaction mixture was acidified with 2 N HC1 (pH~4) and extracted with EtOAc (5 x 500 mL). The combined organic extracts were dried over anhydrous Na SCL and concentrated under reduced pressure to afford Int-A (75 g, 44.6%) as a yellow syrup.
lH NMR (400MHz, CDCl3) d 5.34-5.29 (m, 1H), 4.43-4.17 (m, 1H), 2.51 (td, J = 2.4, 7.1 Hz, 2H), 2.28-2.18 (m, 1H), 2.07-2.01 (m, 1H), 1.45 (s, 9H). LCMS (ESI): (m/z) 246.0 [M-l]\
Synthesis of dibenzyl (tert-butoxycarbonyl)- -glutamate (B):
To a stirring solution of Int-A (75.0 g, 303 mmol) in DMP (500 mL) was added K2CO3 (125 g, 910 mmol) at 0 °C and added benzyl bromide (114 g, 668 mmol) slowly and the reaction mixture was stirred at room temperature for 12 h. After consumption of the starting material (by TLC), the reaction mixture was poured into chilled water (200 mL) and extracted with diethyl ether (3 x 250 mL). The combined organic layers were washed with water (3 x 250
mL). The organic layers were dried over anhydrous Na2S04, filtered and concentrated under reduced pressure to afford Int-B (60 g, 46%) as a brown syrup was used directly for next step without any purification.
7.30 (m, 10H), 5.16 (s, 2H), 5.10 (s, 2H), 4.70 ( d, J = 4.0 Hz, 1H), 4.38 ( d, J = 4.6 Hz, 1H), 2.49-2.35 (m, 2H), 2.21 (dd, J = 6.2, 12.9 Hz, 1H), 2.02-1.93 (m, 1H), 1.42 (s, 9H).
Synthesis of dibenzyl -glutamate hydrochloride (C):
To a stirring solution of Int-B (60.0 g, 140 mmol) in HC1 in ether (250 mL) was added at 0 °C and stirred at room temperature for 12 h. The obtained precipitate was filtered and triturated with diethyl ether (3 xlOO mL) and hexane (3 x 200 mL). The filtered compound was dried under vaccum to afford Int-C (30 g, 58.8%) as a white solid.
8.66 (s, 2H), 7.41-7.30 (m, 10H), 5.26 (s, 2H), 5.15 (s, 2H), 4.16 (t, J = 6.6 Hz, 1H), 2.64- 2.48 (m, 2H), 2.26 - 2.16 (m, 2H).
Synthesis of AD:
Synthesis of tert- butyl (2-oxo-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)ethyl)carbamate (1)
To a stirred solution of SM (2.0 g, 13.05 mmol) in DMF (30 mL), ( tert - butoxycarbonyl)glycine (2.28 g, 13.05 mmol) and DIPEA (7.73 mL, 45.6 mmol) were added at 0 °C and the reaction mixture stirred for 10 min. HATU (17.3 g, 45.6 mmol) was added to the reaction mixture and stirred at room temperature for 5 h. After consumption of the starting material (by TLC), reaction mixture was quenched with water (30 mL) and extracted with EtOAc (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over Na2S04 and concentrated under reduced pressure. The crude was purified by column chromatography using 50% EtOAc/n-hexane to afford compound 1 (2.3 g, 57%) as a yellow solid.
LCMS (ESI): m/z 211.05 [M++l-Boc]
Synthesis of 2-glycyl-2-azaspiro[4.5]decan-3-one hydrochloride (AD):
To a stirred solution of 4M HC1 in methanol (20 mL), compound 1 (2.10 g, 6.77 mmol) was added at 0 °C and the reaction mixture was stirred at room temperature for 4 h. After consumption of the starting material (by TLC), the reaction mixture was concentrated under reduced pressure. The residue was purified by preparative HPLC to afford compound as TFA salt treated with 4M HC1 in MeOH (5mL) and lyophilized to afford AD (0.24 g, 15%) as a yellow solid.
NMR (400 MHz, DMSO-cfe) d 8.26 (s, 3H), 4.13 (s, 2H), 3.49 (s, 2H), 1.44 - 1.37 (m, 12H). LCMS (ESI): m/z 211.05 [M++l] HPLC: 95.08%.
Synthesis of AE:
Synthesis of benzyl (S)-4-((fer/-butoxycarbonyl)amino)-5-oxo-5-(3-oxo-2- azaspiro[4.5]decan-2-yl)pentanoate (1):
To a stirred solution of SM (4.0 g, 26.1 mmol) in DMF (30 mL), (S)-5-(benzyloxy)-2- ( ( /<? - h u to x y c arbo n y 1 ) a m i n o ) - 5 - o x ope n ta no i c acid (Int-B) (8.80 g, 26.1 mmol), DIPEA (15.53 mL, 91.3 mmol), were added at 0 °C and reaction mixture stirred for 10 min. HATU (34.7 g, 91.3 mmol) was added to the reaction mixture and stirred at room temperature for 5 h. After consumption of the starting material (by TLC), reaction mixture was quenched with water (30 mL) and extracted with EtOAc (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over NanSQ* and concentrated under reduced pressure. The crude product
was purified by column chromatography eluting with EtOAc/n-hexane to afford compound 1 (7.50 g, 60%) as a colourless oil.
LCMS (ESI): m/z 373.2 [M++l-Boc]
Synthesis of (S)-4-((fert-butoxycarbonyl)amino)-5-oxo-5-(3-oxo-2-azaspiro[4.5]decan-2- yl)pentanoic acid (2):
To a stirred solution of compound 1 (3.0 g, 6.35 mmol) in EtOAc (15 mL), 10% Pd/C (50% wet, 0.30 g) was added and stirred under hydrogen atmosphere (balloon) for 12 h. After consumption of the starting material (by TLC), the reaction mixture was filtered through a pad of celite and washed with EtOAc (40 mL). The filtrate was concentrated under reduced pressure to afford compound 2 (2.30 g, crude) as a colourless oil.
LCMS (ESI): m/z 283.10 [M++l-Boc] .
Synthesis of tert- butyl (S)-(5-amino-l,5-dioxo-l-(3-oxo-2-azaspiro[4.5]decan-2-yl)pentan- 2-yl)carbamate (3):
To a stirred solution of compound 2 (2 g, 5.18 mmol) in DMF (20 mL), NH4Cl (0.84 g, 15.6 mmol), DIPEA (3.64 mL, 20.9 mmol), were added at 0 °C and reaction mixture was stirred for 10 min. HATU (6.95 g, 18.3 mmol) was added to the reaction mixture and stirred at room temperature for 12 h. After consumption of the starting material (by TLC), reaction mixture was quenched with water (30 mL) and extracted with EtOAc (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over Na2S04 and concentrated under reduced pressure. The crude was purified by column chromatography using 50% EtOAc/n-hexane to afford compound 3 (1.50 g, 75%) as a colourless solid.
LCMS (ESI): m/z 282.10 [M++l-Boc]
Synthesis of (S)-4-amino-5-oxo-5-(3-oxo-2-azaspiro[4.5]decan-2-yl)pentanamide hydrochloride (AE):
To a stirred solution of compound 3 (1.50 g, 3.93 mmol) in dioxane (10 mL), 4M HC1 in 1,4 dioxane (15 mL) was added at 0 °C and stirred at room temperature for 5 h. After consumption of the starting material (by TLC), the solid obtained was filtered and washed with diethyl ether, dried to afford AE (0.75 g, 60.48%) as a colourless solid.
8.48 (bs, 2H), 7.47 (bs, 1H), 6.93 (bs, 1H), 6.5 (bs, 1H), 4.79-4.77 (m, 1H), 3.57-3.55 (m, 1H), 3.48 - 3.45 (m, 1H), 2.54 - 2.53 (m, 2H), 2.32 - 2.21 (m, 2H), 1.98 - 1.94 (m, 2H), 1.5-1.3 (m, 10H). LCMS (ESI): m/z 282.10 [M++l]
HPLC: 96.35%.
Synthesis of 2-amino-5-(benzyloxy)-5-oxopentanoic acid (A):
To a stirred solution of L-glutamic acid (10 g, 68 mmol) in toluene (100 mL) was added benzyl alcohol (10.6 mL, 102.0 mmol) and the reaction mixture was heated at 45 °C for 2 h. Methane sulphonic acid (5.3 mL, 81.63 mmol) was added dropwise at 45 °C to the reaction mixture and stirred at RT for 3 h. After consumption of the starting material (by TLC), the reaction mixture was quenched with water and extracted with EtOAc (3 x 100 mL). The combined organic extracts were dried over anhydrous Na SCL and concentrated under reduced pressure to afford Int A (8.0 g, 49.63%) as a yellow syrup.
LCMS (ESI): (m/z) 238 [M+l]+.
Synthesis of 5-(benzyloxy)-2-((h?r/-butoxycarbonyl)amino)-5-oxopentanoic acid (B):
To a stirred solution of Int- A (8 g, 33.75 mmol) in 1, 4 dioxane and hLO (80 mL, 1:1) were added NaOH (2.7 g, 67.51 mmol) and Boo20 (8.10 g, 37.13 mmol) at 0 °C. The reaction mixture was stirred at RT for 16 h. After consumption of the starting material (by TLC), the reaction mixture was acidified with 2 N HC1 (pH~4) and extracted with EtOAc (3 x 100 mL). The combined organic extracts were dried over anhydrous Na2S04 and concentrated under reduced pressure to afford Int-B (9.5 g, 83.55%) as a yellow syrup.
LCMS (ESI): (m/z) 238.05 [M-Boc]+.
Synthesis of AF:
Synthesis of methyl 2-(3-oxo-2-azaspiro[4.5]decan-2-yl)acetate (1):
To a stirred solution of SM (0.5 g, 3.26 mmol) in THF (20 mL), potassium tert- butoxide (0.55 g, 4.89 mmol) was added portion wise at 0 °C and stirred reaction mixture at same temperature for 20 min. Methyl 2-bromoacetate, 2 (1.49 g, 9.79 mmol) was added and the reaction mixture stirred at RT for 2 h. After consumption of the starting material (by TLC), reaction mixture was diluted with water (20 mL) and extracted with DCM (3 x 30 mL). The combined organic layer was washed with brine (30 mL), dried over Na SCL and concentrated under reduced pressure to afford compound 1 (0.7 g, crude) as a colorless liquid.
LCMS (ESI): m/z 226 [M++l]
Synthesis of 2-(3-oxo-2-azaspiro[4.5]decan-2-yl)acetamide (AF):
To a stirred solution of compound 1 (1.0 g, 4.43 mmol) in MeOH (20 mL), methanolic ammonia (20 mL) was added in sealed tube at 0 °C and stirred reaction mixture at RT for 16 h. After consumption of the starting material (by TLC), the reaction mixture was concentrated under reduced pressure. The crude was purified by column chromatography eluting with 3-5% MeOH/DCM to afford AF (0.43 g, 46%) as a white solid.
MHz, DMSO-ifc): d 7.34 (brs, 1H), 7.04 (brs, 1H), 3.73 (s, 2H), 3.14 (s, 2H), 2.11 (s, 2H), 1.42 - 1.35 (m, 10H). LCMS (ESI): m/z 211.0 [M++l] HPLC: 99.81%.
Synthesis of AG:
The experimental procedure for the synthesis of compound 1 is captured under the synthesis of AA and AB (as compound 1).
Synthesis of benzyl (2S,3/?)-3-(benzyloxy)-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)butanoate (2):
To a solution of compound 1 (3.4 g, 17.1 mmol) in l,2-DCE (70 mL) were added Int-C (6.32 g, 18.8 mmol) and catalytic amount of acetic acid (0.5 mL) at 0 °C, stirred at room temperature for 1.5 h. Sodium triacetoxyhydroborate (7.3 g, 34.3 mmol) was added and continued stirring for 16 h. After consumption of the starting material (by TLC), quenched with water (20 mL) and extracted with CH2CI2 (3 x 20 mL). Combined organic layer dried over
Na2S04 and concentrated under reduced pressure to obtain crude compound which was purified
by column chromatography by eluting with 20% EtOAc/n-hexane to afford compound 2 (2.2 g, 29%) as colourless liquid.
NMR (400 MHz, DMSO-d6) d 7.36 - 7.24 (m, 8H), 7.23 - 7.18 (m, 2H), 5.21 - 5.07 (m, 2H), 4.80 (d, 7 = 4.1 Hz, 1H), 4.57 (d, 7 = 11.9 Hz, 1H), 4.30 (d, 7 = 11.9 Hz, 1H), 4.28 -
4.22 (m, 1H), 3.38 - 3.33 (m, 1H), 3.30 - 3.25 (m, 1H), 2.23 - 2.16 (m, 1H), 2.11 - 2.05 (m,
1H), 1.45 - 1.28 (m, 10H), 1.12 (d, 7 = 6.3 Hz, 3H). LCMS (ESI): m/z 436.4 [M++l]
Synthesis of (2S,3/?)-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)butanoic acid (3):
To a stirred solution of compound 2 (2.2 g, 5.05 mmol) in MeOH (40 mL) was added 10% Pd/C (50% wet, 2.2 g) and stirred under ¾ atmosphere (balloon pressure) for 16 h. After consumption of the starting material (by TLC), the reaction mixture was filtered through a pad of celite and the pad was washed with methanol. Obtained filtrate was concentrated under reduced pressure to afford compound 3 (1.1 g, 85%) as an off white solid.
NMR (400 MHz, DMSO-76) d 4.38 (br d, 7 = 4.1 Hz, 1H), 4.31 - 4.22 (m, 1H), 4.05 - 3.97 (m, 1H), 3.46 - 3.31 (m, 2H), 2.21 - 2.04 (m, 2H), 1.54 - 1.32 (m, 10H), 1.04 (d, 7 = 6.3 Hz, 3H). LCMS (ESI): m/z 256.3 [M++l]
Synthesis of (2S,3/?)-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)butanamide (AG):
To a stirred solution of compound 3 (0.6 g, 2.35 mmol) in CH2CI2 (25 mL) were added NH4CI (0.37 g, 7.05 mmol), HATU (1.34 g, 3.52 mmol) and DIPEA (1.3 mL, 7.05 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), quenched with water (5 mL) and extracted with 10% MeOH/ CH2CI2 (3 x 15 mL). Combined organic layer was dried over Na2S04 and concentrated to obtain crude compound which was purified by column
chromatography by eluting 3% EtOAc/n-hexane to afford AG (270 mg, 45%) as a white solid.
7.41 (s, 1H), 7.00 (s, 1H), 4.76 (d, 7 = 5.1 Hz, 1H),
4.23 (d, 7 = 7.0 Hz, 1H), 4.12 - 3.99 (m, 1H), 3.44 (d, 7 = 9.8 Hz, 1H), 3.31 (d, 7 = 9.7 Hz, 1H), 2.15 - 2.06 (m, 2H), 1.48 - 1.33 (m, 10H), 1.06 (d, 7 = 6.1 Hz, 3H). LCMS (ESI): m/z 255.2 [M++l] HPLC: 97.48%. Chiral HPLC: 87.31%; Column: CHIRALPAK IA (250*4.6 mm *5.0 pm); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: DCM : MeOH (50 : 50) A : B :: 75 : 25; Plow rate : 1.0 mL/min; Retention time : 5.636 min.
Intermediates:
Synthesis of (2S)-2-((fert-butoxycarbonyl)amino)-3-hydroxybutanoic acid (A) :
To a solution of SM-3 (100 g, 456 mmol) in 1, 4 dioxane (450 mL_ and ¾0 (400 mL) were added NaOH (36.5 g, 913 mmol) and B0C2O (119 g, 547 mmol) at 0 °C. The reaction
mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), the reaction mixture was acidified with aqueous 2N HC1 (pH~4) and extracted with EtOAc (5 x 500 mL). The combined organic extracts were dried over anhydrous Na2S04 and concentrated under reduced pressure to afford Int-A (55 g, 55%) as a yellow syrup.
6.29 (d, J = 8.8 Hz, 1H), 4.04-3.98 (m, 1H), 3.87- 3.84 (m, 1H), 1.30 (s, 9H), 1.05 (d, 7 = 6.4 Hz, 3H).
Synthesis of benzyl (2,S )-3-(benzyloxy)-2-((/e;7-butoxycarbonyl)amino)butanoate (B):
To a solution of Int-A (50 g, 156 mmol) in DMF (200 mL) was added NaH (60%) (7.52 g, 313 mmol) at -15 °C and stirred for 2 h. Benzyl bromide (32.0 g, 188 mmol) was added slowly to the resulting reaction mixture. The reaction mixture temperature was warmed to room temperature and stirred for 12 h. After consumption of the starting material (by TLC), the reaction mixture was poured into chilled water (200 mL) and extracted with diethyl ether (4 x 250 mL). The aqueous layer was acidified with citric acid (pH~4) and extracted with EtOAc (4 x 500 mL). The combined organic layers were washed with water (3 x 250 mL). The organic extracts were dried over anhydrous Na2S04, filtered and concentrated under reduced pressure to afford Int-B (20 g, 31.2%) as a brown syrup.
LCMS (ESI) : m/z 300.15 [M+-Boc]
Synthesis of benzyl (2S)-2-amino-3-(benzyloxy)butanoate hydrochloride (C):
To a solution of Int-B (20.0 g, 48.8 mmol) in HC1 in ether (20 mL) was added at 0 °C and stirred at room temperature for 12 h. The obtained precipitate was filtered and triturated with diethyl ether (2 xlOO mL) and hexane (2 x 200 mL). The filtered compound was dried under vacuum to afford Int-C (14.0 g, 96.4%) as a white solid.
8.68 (s, 3H), 7.50-7.25 (m, 10H), 5.24-5.13 (m, 2H), 4.53 (d, 7 = 11.6 Hz, 1H), 4.35 (d, 7 = 11.6 Hz, 1H), 4.23 (d, 7 = 3.2 Hz, 1H), 4.11-4.07 (m, 1H), 1.27 (d, 7 = 6.4 Hz, 3H).
Synthesis of AH:
The experimental procedure for the synthesis of compound 3 has been captured under the synthesis of AG (as compound 3).
Synthesis of (2S,3/?)-3-hydroxy-/V-methyl-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)butanamide (AH):
To a stirred solution of compound 3 (1 g, 3.92 mmol) in CH2CI2 (25 mL) were added methanamine hydrochloride (0.53 g, 7.84 mmol), HATU (2.2 g, 5.88 mmol) and DIPEA (2.7 mL, 15.6 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), quenched with water (5 mL) and extracted with 10% MeOH/ CH2CI2 (3 x 30 mL). Combined organic layer was dried over Na2S04 and concentrated to obtain crude compound which was purified by column chromatography by eluting with 2% MeOH/ CH2CI2 to afford AH (300 mg, 28%) as an off white hygroscopic solid.
4.21 (d, J = 7.7 Hz, 1H), 4.09 - 3.92 (m, 1H), 3.43 (d, J = 9.8 Hz, 1H), 3.32 (d, J = 9.8 Hz, 1H), 2.55 (d, J = 4.6 Hz, 3H), 2.10 (d, J = 2.8 Hz, 2H), 1.50 - 1.31 (m, 10H), 1.02 (d, J = 6.3 Hz,
3H). LCMS (ESI): m/z 269.2 [M++l] HPLC: 99.27%. Chiral HPLC: >99.00%; Column: CHIRALPAK IC (250*4.6 mm*5.0 pm); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: EtOH : MeOH (50 : 50); A : B :: 70 : 30; Flow rate : 1.0 mL/min; Retention time: 6.959 min.
Synthesis of AI:
The experimental procedure for the synthesis of compound 3 has been captured under the synthesis of AG (as compound 3).
Synthesis of (2S,3/?)-3-hydroxy-/V^V-dimethyl-2-(3-oxo-2-azaspiro[4.5]decan-2- yl)butanamide (AI):
To a stirred solution of compound 3 (1 g, 3.92 mmol) in CH2CI2 (25 mL) were added dimethylamine hydrochloride (0.63 g, 7.84 mmol), HATU (2.2 g, 5.88 mmol) and DIPEA (2.7 mL, 15.6 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), quenched with water (5 mL) and extracted with 10% MeOH/ CH2CI2 (3 x 30 mL). Combined organic layer was dried over Na2S04 and concentrated to obtain crude compound which was purified by column chromatography by eluting with 2% MeOH/ CH2CI2 to afford AI (170 mg, 15%) as a white solid.
4.01 - 3.93 (m, 1H), 3.26 (d, J = 9.8 Hz, 1H), 3.10 (d, J = 9.8 Hz, 1H), 3.02 (s, 3H), 2.81 (s, 3H), 2.20 - 2.06 (m, 2H), 1.53 - 1.30 (m, 10H), 1.02 (d, J = 6.1 Hz, 3H). LCMS (ESI): m/z 283.3 [M++l] HPLC: 99.06%. Chiral HPLC: >99.00%; Column: CHIRALPAK IC (250*4.6 mm *5.0 qm); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: EtOH : MeOH (50 : 50); A : B :: 70 : 30; Flow rate : 1.0 mL/min; Retention time : 8.358 min.
Synthesis of A.T:
The experimental procedure for the synthesis of compound 3 has been captured under the synthesis of AG (as compound 3).
Synthesis of (2S,3/?)-/V-benzyl-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)butanamide (AG):
To a stirred solution of compound 3 (0.9 g, 3.53 mmol) in CH2CI2 (20 mL) were added phenylmethanamine (0.58 mL, 5.29 mmol), HATU (2.02 g, 5.29 mmol) and DIPEA (1.85 mL, 10.59 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), diluted with CH2CI2 (50 mL) and washed with water (3 x 50 mL). Combined organic layer was dried over Na2S04 and concentrated to obtain crude compound which was purified by column chromatography by eluting 3 % MeOH/ CH2CI2 to afford AJ (300 mg, 24%) as a white solid.
NMR (400 MHz, DMSO-ri6) d 8.57 (t, J = 5.9 Hz, 1H), 7.35 - 7.27 (m, 2H), 7.25 - 7.18 (m, 3H), 4.81 (br d, J = 4.6 Hz, 1H), 4.32 (d, J = 7.7 Hz, 1H), 4.29 - 4.16 (m, 2H), 4.11 -
3.99 (m, 1H), 3.44 (d, J = 9.8 Hz, 1H), 3.35 - 3.32 (m, 1H), 2.21 - 1.99 (m, 2H), 1.55 - 1.31 (m, 10H), 1.04 (d, J = 6.3 Hz, 3H). LCMS (ESI): m/z 345.5 [M++l] HPLC: 96.10%. Chiral HPLC: 99.18%; Column: CHIRALPAK IC (250*4.6 mm*5.0 pm); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: EtOH : MeOH (50 : 50); A : B :: 70 : 30; Flow rate : 1.0 mL/min; Retention time: 6.410 min.
Synthesis of AK:
The experimental procedure for the synthesis compound 3 is captured under the synthesis of AG (as compound 3).
Synthesis of (2S,3/?)-/V-(4-fluorobenzyl)-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2- yl)butanamide (AK):
To a stirred solution of compound 3 (0.9 g, 3.52 mmol) in CH2CI2 (20 mL) were added (4-fh orophenyl)methanamine (0.52 mL, 4.58 mmol), HATU (2.68 g, 7.05 mmol) and DIPEA (1.8 mL, 10.5 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), quenched with water (100 mL) and extracted with CH2CI2 (2 x 100 mL). Combined organic layer was with brine, dried over Na2S04 and concentrated. The crude material was purified by column chromatography by eluting 1% MeOH/ CH2CI2 to afford AK (200 mg, 16%) as a off white solid.
7.07 (m, 2H), 4.81 (d, J = 5.4 Hz, 1H), 4.29 (d, 7 = 7.7 Hz, 1H), 4.21 (dd, J = 3.0, 5.8 Hz, 2H), 4.09 - 3.98 (m, 1H), 3.42 (d, J = 9.7 Hz, 1H), 3.34 - 3.23 (m, 1H), 2.19 - 1.99 (m, 2H), 1.51 - 1.29 (m, 10H), 1.02 (d, J = 6.3 Hz, 3H). LCMS (ESI): m/z 363.3 [M++l] HPLC: 99.63%. Chiral HPLC: 98.02%; Column : CHIRALPAK IC (250*4.6 mm*5.0 pm); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: EtOH : MeOH (50 : 50); A : B :: 70 : 30; Flow rate : 1.0 mL/min; Retention time : 5.071 min.
Synthesis of AL:
The experimental procedure for the synthesis of compound 3 has been captured under the synthesis of AG (as compound 3).
Synthesis of (2S,3/?)-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)-N-(4-(trifluoro- methyl) benzyl) butanamide (AL):
To a stirred solution of compound 3 (0.9 g, 3.53 mmol) in CH2CI2 (20 mL) were added (4-(trifluoromethyl)phenyl)methanamine (0.75 mL, 5.29 mmol), HATU (2.02 g, 5.29 mmol) and DIPEA (1.85 mL, 10.59 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), diluted with CH2CI2 (50 mL) and washed with water (3 x 50 mL). Combined organic layer was dried over Na2S04 and concentrated to obtain crude compound which was purified by column chromatography by eluting 2% MeOH/ CH2CI2 to afford AL (300 mg, 24%) as sticky syrup. This material was further purified by preparative HPLC purification to obtain AL (180 mg) as an off white hygroscopic solid.
8.67 (t, J = 6.0 Hz, 1H), 7.66 (d, J = 8.2 Hz, 2H), 7.45 (d, J = 8.0 Hz, 2H), 4.83 (d, J = 5.3 Hz, 1H), 4.39 - 4.26 (m, 3H), 4.12 - 4.01 (m, 1H), 3.42 - 3.37 (m, 1H), 3.34 - 3.29 (m, 1H), 2.18 - 2.05 (m, 2H), 1.50 - 1.28 (m, 10H), 1.04 (d, J = 6.1 Hz, 3H). LCMS (ESI): m/z 413.2 [M++l] .HPLC: 99.82%. Chiral HPLC: 99.78%; Column:
CHIRALPAK IC (250*4.6 mm*5.0 pm); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: EtOH : MeOH (50 : 50); A : B :: 70 : 30; Flow rate : 1.0 mL/min; Retention time : 4.560 min.
Synthesis of AM:
The experimental procedure for the synthesis of compound 3 has been captured under the synthesis of AG (as compound 3).
Synthesis of (2S,3/?)-3-hydroxy-/V-(4-methoxybenzyl)-2-(3-oxo-2-azaspiro[4.5]decan-2- yl)butanamide (AM):
To a stirred solution of compound 3 (0.9 g, 3.53 mmol) in CH2CI2 (20 mL) were added (4-methoxyphenyl)methanamine (0.59 mL, 4.58 mmol), HATU (2.68 g, 7.05 mmol) and DIPEA (1.80 mL, 10.58 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), quenched with water (100 mL) and extracted with CH2CI2 (2 x 100 mL). Combined organic layer was washed with brine, dried over Na2S04 and concentrated. The crude material was purified by column chromatography by eluting with 1 % MeOH/ CH2CI2 to afford AM (700 mg, 53%) as an off white sticky solid. This material was further purified by preparative HPLC purification to obtain AM (200 mg) as an off white hygroscopic solid.
6.85 (d, J = 8.8 Hz, 2H), 4.78 (d, J = 5.3 Hz, 1H), 4.28 (d, J = 7.7 Hz, 1H), 4.23 - 4.10 (m, 2H), 4.08 - 3.98 (m, 1H), 3.72 (s, 3H), 3.43 (d, J = 9.8 Hz, 1H), 3.31 (d, J = 9.8 Hz, 1H), 2.15 - 2.04 (m, 2H), 1.51 - 1.30 (m, 10H), 1.02 (d, J = 6.1 Hz, 3H). LCMS (ESI): m/z 375.2 [M++l]
HPLC: 99.82%. Chiral HPLC: 98.55%; Column: CHIRALPAK IC (250*4.6 mm*5.0 pm); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: EtOH : MeOH (50 : 50); A : B :: 75 : 25; Flow rate : 1.0 mL/min; Retention time : 6.937 min.
Synthesis of BM:
The experimental procedure for the synthesis compound 2 is captured under the synthesis of AA and AB (as compound 2).
Synthesis of (S)-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)propanoic acid (3):
To a stirring solution of compound 2 (4.9 g, 11.63 mmol) in methanol (100 mL) was added 10% Pd/C (50% wet, 5 g) was added at room temperature and stirred under hydrogen atmosphere (balloon pressure) for 16 h. After consumption of the starting material (by TLC), the reaction mixture was filtered through a pad of celite and washed with 50% MeOH/ CH2CI2 (2 x 80 mL). The combined filtrate was concentrated under reduced pressure to afford compound 3 (2.3 g, crude) as a colourless liquid.
NMR (400 MHz, DMSO-d6) d 12.75 (br s, 1H), 4.97 (br s, 1H), 4.52 (t, 7 = 6.1 Hz, 1H), 3.74 (d, J = 6.0 Hz, 2H), 3.29 - 3.19 (m, 2H), 2.12 (d, J = 6.3 Hz, 2H), 1.54 - 1.31 (m, 10H). LCMS (ESI): m/z 242.2 [M++l]
Synthesis of (S)-3-hydroxy-/V-methyl-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)propanamide (BM):
To a stirring solution of compound 3 (1 g, 4.14 mmol) in CH2CI2 (20 mL) were added methanamine hydrochloride (0.42 g, 6.22 mmol), HATU (2.36 g, 6.22 mmol) and DIPEA (3.6 mL, 20.7 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), reaction mixture was diluted with water (50 mL) and extracted with CH2CI2 (3 x 90 mL). The combined organic layer was dried over Na2S04 and concentrated under reduced pressure. The crude compound
was purified by flash column chromatography by eluting 2% MeOH/ CH2CI2 followed by trituration with Et20 (2 x 20 mL). Obtained product was purified by chiral preparative HPLC to afford BM (98 mg, 9%) as a white solid.
NMR (400 MHz, DMSO-d6) d 7.81 (br d , J = 4.3 Hz, 1H), 4.87 (t, J = 5.4 Hz, 1H), 4.45 (dd, J = 5.4, 8.7 Hz, 1H), 3.74 - 3.66 (m, 1H), 3.62 - 3.55 (m, 1H), 3.29 (d, J = 9.5 Hz,
1H), 3.18 (d, / = 9.5 Hz, 1H), 2.55 (d, / = 4.6 Hz, 3H), 2.11 (d, / = 1.4 Hz, 2H), 1.51 - 1.32 (m, 10H). LCMS (ESI): m/z 255.2 [M++l] HPLC: 99.58%. Chiral HPLC: 99.05%; Column: CHIRALPAK IC (250*4.6 hihi*5mhi); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: DCM : MeOH (80 : 20); A : B :: 65 : 35; Flow rate : 1.0 mL/min; Retention time : 12.784 min.
Synthesis of BN:
The experimental procedure for the synthesis compound 3 is captured under the synthesis of BM (as compound 3).
Synthesis of (S)-/V-benzyl-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)propanamide (BN):
To a stirring solution of compound 3 (1 g, 4.14 mmol) in CH2CI2 (20 mL) were added phenylmethanamine (0.72 mL, 6.22 mmol), HATU (2.36 g, 6.22 mmol) and DIPEA (2.2 mL, 12.44 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 h. After consumption of the starting material (by TLC), reaction mixture was diluted with water (50 mL) and extracted with CH2CI2 (3 x 80 mL). The combined organic layer was dried over Na2S04 and concentrated to obtain crude compound which was purified
by flash column chromatography by eluting 1% MeOH/ CH2CI2 followed by chiral preparative HPLC purification to afford BN (180 mg, 13%) as a white solid.
8.44 (t, J = 5.8 Hz, 1H), 7.35 - 7.26 (m, 2H), 7.25 - 7.18 (m, 3H), 4.91 (t, J = 5.3 Hz, 1H), 4.55 (dd, J = 5.6, 8.5 Hz, 1H), 4.38 - 4.15 (m, 2H), 3.78 - 3.72 (m, 1H), 3.66 - 3.60 (m, 1H), 3.29 (d, J = 9.5 Hz, 1H), 3.19 (d, J = 9.5 Hz, 1H), 2.12 (s, 2H), 1.47 - 1.31 (m, 10H). LCMS (ESI): m/z 331.1 [M++l] HPLC: 99.11%. Chiral HPLC: 99.76%; Column: CHIRALPAK IC (250*4.6 hihi*5mhi); Mobile Phase: A: 0.1% DEA in n- Hexane Mobile Phase: B: EtOH : MeOH (50 : 50); A : B :: 70 : 30; Flow rate : 1.0 mL/min; Retention time: 9.477 min.
Synthesis of BO:
The experimental procedure for the synthesis compound 3 is captured under the synthesis of BM (as compound 3).
Synthesis of (S)-/V-(4-fluorobenzyl)-3-hydroxy-2-(3-oxo-2-azaspiro[4.5]decan-2- yl)propanamide (BO):
To a stirring solution of compound 3 (0.2 g, 0.829 mmol) in EtOAc (10 mL) were added (4-fluorophenyl)methanamine (0.14 mL, 1.24 mmol), T3P (50% in EtOAc, 1.6 mL, 2.48 mmol) and Et3N (0.6 mL, 4.14 mmol) at 0 °C under nitrogen atmosphere. The reaction mixture was allowed to room temperature and stirred for 16 h. After consumption of the starting material (by TLC), quenched with water (30 mL) and extracted with EtOAc (3 x 40 mL). The combined organic layer was with brine, dried over Na2S04 and concentrated under reduced pressure. The crude material was purified by column chromatography by eluting 1% MeOH/ CH2CI2 to afford BO (110 mg, 38%) as an off white solid.
2H), 7.16 - 7.06 (m, 2H), 4.91 (t, J = 5.2 Hz, 1H), 4.53 (dd, J = 5.6, 8.3 Hz, 1H), 4.33 - 4.13 (m,
2H), 3.77 - 3.71 (m, 1H), 3.67 - 3.56 (m, 1H), 3.28 (d, J = 9.4 Hz, 1H), 3.18 (d, J = 9.4 Hz,
1H), 2.12 (s, 2H), 1.46 - 1.30 (m, 10H). LCMS (ESI): tn/z 349.2 [M++l] HPLC: 99.07%. Chiral HPLC: 95.67%; Column: CHIRALPAK IC (250*4.6 hihi*5mhi); Mobile Phase: A: 0.1% DEA in n-Hexane; Mobile Phase: B: EtOH : MeOH (50 : 50) A : B :: 70 : 30; Flow rate: 1.0 mL/min; Retention time : 7.215 min.
Following the above procedures, the following compounds and stereoisomers thereof were or are prepared. It will be appreciated by a person of skill in the art that for the structures shown, additional stereoisomers such as diasteromers and/or enantiomers may be present and are included in the present disclosure.
Table 1
B. NMDAR AGONIST ASSAYS
Assays were conducted as described by Moskal et al.,“GLYX-13: a monoclonal antibody-derived peptide that acts as an N-methyl-D-aspartate receptor modulator,”
Neuropharmacology, 49, 1077-87, 2005. These studies were designed to determine if the test compounds act to facilitate NMDAR activation in NMDAR2A or NMDAR2B expressing HEK cell membranes as measured by increases in [3H]MK-80l binding.
In the assay, 300 pg of NMDAR expressing HEK cell membrane extract protein was preincubated for 15 minutes at 25° C in the presence of saturating concentrations of glutamate (50 pM) and varying concentrations of test compound (lxl0 15M - lxlO M), or 1 mM glycine. Following the addition of 0.3 pCi of [3H]MK-80l (22.5 Ci/mmol), reactions were again incubated for 15 minutes at 25 C (nonequilibrium conditions). Bound and free [3H]MK-80l were separated via rapid filtration using a Brandel apparatus.
In analyzing the data, the DPM (disintegrations per minute) of [3H]MK-80l remaining on the filter were measured for each concentration of test compound or for 1 mM glycine. The DPM values for each concentration of a ligand (N=2) were averaged. The baseline value was determined from the best fit curve of the DPM values modeled using the GraphPad program and the log(agonist) vs. response(three parameters) algorithm was then subtracted from all points in the dataset. The % maximal [3H]MK-80l binding was then calculated relative to that of 1 mM glycine: all baseline subtracted DPM values were divided by the average value for 1 mM glycine. The EC and % maximal activity were then obtained from the best fit curve of the % maximal [3H]MK-80l binding data modelled using the GraphPad program and the log(agonist) vs. response( three parameters) algorithm.
The tables below summarize the results for the wild type NMDAR agonists NMDAR2A and NMDAR2B, and whether the compound is not an agonist (-), is an agonist (+), or is a strong agonist (++), where column A is based on the % maximal [3H]MK-80l binding relative
to 1 mM glycine (- = 0; < 100% = +; and > 100% = ++); and column B is based on log EC50 values (0 = -; > lxlO-9 M (e.g., -8) = +; and < lxlO-9 M (e.g., -10) = ++).
C. MICROSOMAL STABILITY
Microsomal stability of disclosed compounds was investigated. The following table indicates the percent of compound remaining after 60 minutes.
Plasma stability of disclosed compounds was investigated. The following table indicates the percent of compound remaining after 60 minutes.
E. PHARMACOKINETICS ASSAYS
Sprague Dawley rats were dosed intravenously using a normal saline formulation containing 2 mg/kg of the compounds identified in the below table. The table below summarizes the results of the IV pharmacokinetics.
In another experiment, Sprague Dawley rats were dosed per os (oral gavage) using a normal saline formulation containing 10 mg/kg of the compounds identified in the table below. Plasma, brain, and CSF samples were analyzed at various time points over a 24 hour period. The table below summarizes the results of the oral pharmacokinetics, where the first three
values (Tmax, Cmax and AUCiast) are plasma values. N/D indicates that the measurement was not done.
F. PORSOLT ASSAY
A non-clinical in vivo pharmacology study (Porsolt assay) was performed to measure antidepressant-like effects. The study allowed for the evaluation of the effects of each compound on the Porsolt forced swim test as assessed by the rats’ response (reduced floating time) during a 5-minute swimming test.
Male 2-3 month old Sprague Dawley rats were used (Harlan, Indianapolis, IN). Rats were housed in Lucite cages with aspen wood chip bedding, maintained on a 12: 12 light:dark cycle (lights on at 5 AM), and given ad libitum access to Purina lab chow (USA) and tap water throughout the study.
The Porsolt forced swim test adapted for use in rats was performed as described by Burgdorf et ak, (The long-lasting antidepressant effects of rapastinel (GLYX-13) are associated with a metaplasticity process in the medial prefrontal cortex and hippocampus. Neuroscience 308:202-211, 2015). Animals were placed in a 46 cm tall x 20 cm in diameter clear glass tube filled to 30 cm with tap water (23 ± 1 °C) for 15 min on the first day (habituation) and 5 min on the subsequent test day. Animals were tested 1 h or 1 week post-dosing with the test compounds or vehicle control (0.5% sodium carboxymethyl cellulose in 0.9% sterile saline). Animals received a 15 min habituation session 1 day before the first 5 min test. Water was changed after every other animal. Animals were videotaped, and floating time as defined as the minimal amount of effort required to keep the animals head above water was scored offline by a blinded experimenter with high inter-rater reliability (Pearson’s r > .9). N/D indicates that the measurement was not done.
EQUIVALENTS
Those skilled in the art will recognize, or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments of the disclosure described herein. Such equivalents are intended to be encompassed by the following claims.
INCORPORATION BY REFERENCE
The entire contents of all patents, published patent applications, websites, and other references cited herein are hereby expressly incorporated herein in their entireties by reference.
Claims
1. A compound represented by:
or a pharmaceutically acceptable salt and/or a stereoisomer thereof, wherein:
q is 1, 2, 3, or 4;
R5 is independently, for each occurrence, one or more optional substituents selected from the group consisting of Ci-C6alkyl, -S(0)w-Ci_C3alkyl, -NRaRb, and halogen, wherein Ci- C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
R6 is one or two optional substituents bound to one or more available ring carbons, and is independently, for each occurrence, selected from the group consisting of Ci-Cealkyl, - S(0)w-Ci-C3alkyl, -NRaRb, and halogen, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
w is 0, 1, or 2;
R3 is selected from the group consisting of H, phenyl, Ci-Cealkyl, -C(0)R31, and - C(0)0R32, wherein C|-Q, alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs;
R31 and R32 are each independently selected from the group consisting of H, Ci-Cealkyl, -C3-C6cycloalkyl, and phenyl, wherein Ci-C6alkyl is optionally substituted with one, two, or three substitutents each independently selected from Rs, and phenyl is optionally substituted with one, two, or three substitutents each independently selected from RT;
Ra and Rb are independently, for each occurrence, selected from the group consisting of H, phenyl, Ci-C4alkyl, -Ci-C4alkylene-phenyl, or Ra and Rb taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted with one, two, or three substitutents each independently selected from hydroxyl, - Ci-C3alkoxy, -Ci-C3alkyl, and halogen, and Ci-C3alkyl is optionally substituted with one, two or three halogens;
Rs is independently, for each occurrence, selected from the group consisting of - C(0)NRaRb, -NRaRb, hydroxyl, S(0)w-Ci-C3alkyl, SH, phenyl and halogen; and
RT is independently, for each occurrence, selected from the group consisting of -Ci- C6alkyl, hydroxyl, Ci-C6alkoxy, and halogen.
2. The compound of claim 1, wherein q is 1, 2 or 3.
3. The compound of claim 1, wherein q is 1.
4. The compound of one of claims 1-3, wherein R5 is absent.
5. The compound of any one of claims 1-4, wherein R6 is absent.
6. The compound of any one of claims 1-5, wherein R3 is Ci-C6alkyl, wherein Ci-C6alkyl is optionally substituted by one, two, or three substituents each independently selected from - C(0)-NRaRb, and hydroxyl; and Ra and Rb are each independently selected for each occurrence from the group consisting of H, Ci-C4alkyl, and -Ci-C4alkylene-phenyl, wherein phenyl is optionally substituted by one two or three substituents each independently selected from Ci- C3alkoxy, Ci-C3alkyl, and halogen, and Ci-C3alkyl is optionally substituted with one, two, or three halogens independently selected from F, Cl, and Br.
7. The compound of any one of claims 1-5, wherein R3 is -C(0)-R31, wherein R31 is Ci- C6alkyl, and Ci-C6alkyl is optionally substituted by one, two, or three substituents each independently selected from -C(0)-NRaRb, -NRaRb, and hydroxyl; and Ra and Rb are each independently selected for each occurrence from the group consisting of H and Ci-C4alkyl.
8. The compound of any one of claims 1-5, wherein R3 is selected from the group consisting of:
wherein Ra and Rb are each independently selected for each occurrence from the group consisting of H and -Ci-C4alkyl.
9. The compound of any one of claims 1-5, wherein R3 is selected from the group consisting of:
wherein Ra and Rb are each independently selected for each occurrence from the group consisting of H and -Ci-C4alkyl, and R8 is one, two, or three optional substituents each independently selected from the group consisting of hydroxyl, Br, Cl, F, methoxy, and -CF3.
10. The compound of any one of claims 1-9, wherein Ra and Rb are H.
11. The compound of any of claims 1-6, wherein Ra is H or methyl and Rb is selected from the group consisting of methyl, ethyl, isobutyl, and -CFF-phenyl, wherein phenyl is optionally substituted by one two or three substituents each independently selected from F, methoxy, and CF3.
12. A compound represented by:
or a pharmaceutically acceptable salt and/or a stereoisomer thereof, wherein:
R3 is selected from the group consisting of H, -C(0)-Ci-C6alkyl, and -Ci-Cealkyl-C(O)- NRaRb; wherein Ci-C6alkyl is optionally substituted by one, two, or three substituents each independently selected from halogen, hydroxyl, cyano, -CCFH, phenyl, -C(0)-NRaRb, and NRaRb; and
Ra and Rb are each independently for each occurrence selected from the group consisting of H, phenyl, Ci-C4alkyl, -Ci-C4alkylene -phenyl, or Ra and Rb taken together with the nitrogen to which they are attached form a 4-6 membered heterocyclic ring, wherein phenyl is optionally substituted by one two or three substituents each independently selected from Ci- C3alkoxy, Ci-C3alkyl, and halogen, wherein Ci-C3alkyl is optionally substituted with one, two or three substituents independently selected from F, Cl, and Br.
13. The compound of claim 12, wherein R3 is -C(0)-Ci-C6alkyl, wherein Ci-C6alkyl is optionally substituted by one, two, or three substituents each independently selected from -
C(0)-NRaRb, -NRaRb, and hydroxyl; and Ra and Rb are each independently, for each occurrence, selected from the group consisting of H and Ci-C4alkyl.
14. The compound of claim 12 or 13, wherein R3 is selected from the group consisting of:
wherein Ra and Rb are each independently selected for each occurrence from the group consisting of H and -Ci-C4alkyl.
15. The compound of claim 12 or 13, wherein R3 is selected from the group consisting of:
wherein Ra and Rb are each independently selected for each occurrence from the group consisting of H and -Ci-C4alkyl, and R8 is one, two, or three optional substituents each independently selected from the group consisting of hydroxyl, Br, Cl, F, methoxy, and -CF3.
16. The compound of any one of claims 12-15, wherein Ra and Rb are H.
17. The compound of any one of claims 12-15, wherein Ra is H or methyl and Rb is selected from the group consisting of methyl, ethyl, isobutyl, and -CH2-phenyl, wherein phenyl is optionally substituted by one, two, or three substituents each independently selected from F, methoxy, and -CF3.
18. A compound selected from the group consisting of:
19. A compound selected from the compounds delineated in Table 1 of the Examples, or a pharmaceutically acceptable salt or a stereoisomer thereof.
20. A pharmaceutical composition comprising a compound of any one of claims 1-19, and a pharmaceutically acceptable excipient.
21. The pharmaceutical composition of claim 20, suitable for oral administration, parenteral administration, topical administration, intravaginal administration, intrarectal administration, sublingual administration, ocular administration, transdermal administration, or nasal administration.
22. A method of treating depression, Alzheimer’s disease, attention deficit disorder, schizophrenia, or anxiety, in a patient in need thereof, the method comprising administering to the patient a therapeutically effective amount of the compound of any one of claims 1-19 or the pharmaceutical composition of claim 20 or 21.
23. A method of treating acute neuropathic pain or chronic neuropathic pain, in a patient in need thereof, the method comprising administering to the patient a therapeutically effective amount of the compound of any one of claims 1-19 or the pharmaceutical composition of claim 20 or 21.
24. The method of claim 23, wherein the neuropathic pain is chronic or acute.
25. The method of claim 23, wherein the neuropathic pain is selected from the group consisting of herpes, HIV, traumatic nerve injury, stroke, post-ischemia, chronic back pain, post-herpetic neuralgia, fibromyalgia, reflex sympathetic dystrophy, complex regional pain syndrome, spinal cord injury, sciatica, phantom limb pain, diabetic neuropathy, and cancer chemotherapeutic-induced neuropathic pain.
26. A method of treating a neurodevelopmental disorder related to synaptic dysfunction in a patient in need thereof, the method comprising administering to the patient a therapeutically effective amount of the compound of any one of claims 1-19, or the pharmaceutical composition of claim 20 or 21.
27. A method of treating a cognitive impairment disorder in a patient in need thereof, the method comprising administering to the patient a therapeutically effective amount of the compound of any one of claims 1-19, or the pharmaceutical composition of claim 20 or 21.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201862624217P | 2018-01-31 | 2018-01-31 | |
| US62/624,217 | 2018-01-31 | ||
| US201862718070P | 2018-08-13 | 2018-08-13 | |
| US62/718,070 | 2018-08-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019152688A1 true WO2019152688A1 (en) | 2019-08-08 |
Family
ID=65494519
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2019/016114 Ceased WO2019152688A1 (en) | 2018-01-31 | 2019-01-31 | Spiro-lactam nmda receptor modulators and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2019152688A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113956183A (en) * | 2021-10-28 | 2022-01-21 | 成都市科隆化学品有限公司 | Boc-Ser (Bzl) -OH and preparation method thereof |
| CN114685277A (en) * | 2020-12-31 | 2022-07-01 | 苏州瑞博生物技术股份有限公司 | Compound and method for purifying hydroxypropionate compound |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1563252A (en) * | 1975-12-19 | 1980-03-26 | Warner Lambert Co | Spirolactams |
| US20020173537A1 (en) * | 1997-11-18 | 2002-11-21 | Feurerstein Thomas J. | 2-pyrrolidinone derivatives substituted at position 4 for reducing the extracellular glutamate level |
| EP1529528A1 (en) * | 2003-11-07 | 2005-05-11 | Universitätsklinikum Freiburg | 2-pyrrolidone derivatives and their use in protecting mammalian cells against oxidation stress |
| US20080039456A1 (en) * | 2005-01-06 | 2008-02-14 | Gelder Joel M V | Rigidified Compounds for Modulating Heparanase Activity |
| WO2014120784A1 (en) * | 2013-01-29 | 2014-08-07 | Naurex, Inc. | Spiro-lactam nmda receptor modulators and uses thereof |
| WO2014120783A1 (en) * | 2013-01-29 | 2014-08-07 | Naurex, Inc. | Spiro-lactam nmda receptor modulators and uses thereof |
| WO2014120800A1 (en) * | 2013-01-29 | 2014-08-07 | Naurex, Inc. | Spiro-lactam nmda receptor modulators and uses thereof |
| US20160016903A1 (en) * | 2014-07-15 | 2016-01-21 | Gruenenthal Gmbh | Substituted Azaspiro(4.5)Decane Derivatives |
| WO2016008582A1 (en) * | 2014-07-15 | 2016-01-21 | Grünenthal GmbH | Substituted azaspiro(4.5)decane derivatives |
-
2019
- 2019-01-31 WO PCT/US2019/016114 patent/WO2019152688A1/en not_active Ceased
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1563252A (en) * | 1975-12-19 | 1980-03-26 | Warner Lambert Co | Spirolactams |
| US20020173537A1 (en) * | 1997-11-18 | 2002-11-21 | Feurerstein Thomas J. | 2-pyrrolidinone derivatives substituted at position 4 for reducing the extracellular glutamate level |
| EP1529528A1 (en) * | 2003-11-07 | 2005-05-11 | Universitätsklinikum Freiburg | 2-pyrrolidone derivatives and their use in protecting mammalian cells against oxidation stress |
| US20080039456A1 (en) * | 2005-01-06 | 2008-02-14 | Gelder Joel M V | Rigidified Compounds for Modulating Heparanase Activity |
| WO2014120784A1 (en) * | 2013-01-29 | 2014-08-07 | Naurex, Inc. | Spiro-lactam nmda receptor modulators and uses thereof |
| WO2014120783A1 (en) * | 2013-01-29 | 2014-08-07 | Naurex, Inc. | Spiro-lactam nmda receptor modulators and uses thereof |
| WO2014120800A1 (en) * | 2013-01-29 | 2014-08-07 | Naurex, Inc. | Spiro-lactam nmda receptor modulators and uses thereof |
| US20160016903A1 (en) * | 2014-07-15 | 2016-01-21 | Gruenenthal Gmbh | Substituted Azaspiro(4.5)Decane Derivatives |
| WO2016008582A1 (en) * | 2014-07-15 | 2016-01-21 | Grünenthal GmbH | Substituted azaspiro(4.5)decane derivatives |
Non-Patent Citations (8)
| Title |
|---|
| CARREIRA; KVAERNO: "Classics in Stereoselective Synthesis", 2009, WILEY-VCH |
| COLLINGRIDGE ET AL.: "The NMDA Receptor", 1994, OXFORD UNIVERSITY PRESS |
| FOSTER ET AL., NATURE, vol. 329, 1987, pages 395 - 396 |
| KAVINA M A ET AL: "Synthesis of substituted 2-(2-oxopyrrolidin-1-yl)acetamides", RUSSIAN JOURNAL OF ORGANIC CHEMISTRY, M A I K NAUKA - INTERPERIODICA, RU, vol. 53, no. 6, 26 July 2017 (2017-07-26), pages 873 - 878, XP036283778, ISSN: 1070-4280, [retrieved on 20170726], DOI: 10.1134/S1070428017060100 * |
| MAHBOOBE AMIRANI POOR ET AL: "Gabapentin-base synthesis and theoretical studies of biologically active compounds: N-cyclohexyl-3-oxo-2-(3-oxo-2-azaspiro[4.5] decan-2-yl)-3-arylpropanamides and N-(tert-butyl)-2-(3-oxo-2-azaspiro[4.5]decan-2-yl)-2-arylacetamide derivatives", JOURNAL OF MOLECULAR STRUCTURE., vol. 1152, 1 January 2018 (2018-01-01), NL, pages 44 - 52, XP055571419, ISSN: 0022-2860, DOI: 10.1016/j.molstruc.2017.09.061 * |
| MAYER ET AL., TRENDS IN PHARMACOL. SCI., vol. 11, 1990, pages 254 - 260 |
| MOSKAL ET AL.: "GLYX-13: a monoclonal antibody-derived peptide that acts as an N-methyl-D-aspartate receptor modulato", NEUROPHARMACOLOGY, vol. 49, 2005, pages 1077 - 87, XP026192890, DOI: doi:10.1016/j.neuropharm.2005.06.006 |
| NEUROSCIENCE, vol. 308, 2015, pages 202 - 211 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114685277A (en) * | 2020-12-31 | 2022-07-01 | 苏州瑞博生物技术股份有限公司 | Compound and method for purifying hydroxypropionate compound |
| CN113956183A (en) * | 2021-10-28 | 2022-01-21 | 成都市科隆化学品有限公司 | Boc-Ser (Bzl) -OH and preparation method thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12084444B2 (en) | Spiro-lactam NMDA modulators and methods of using same | |
| US11530223B2 (en) | Spiro-lactam NMDA receptor modulators and uses thereof | |
| US11578072B2 (en) | Spiro-lactam NMDA receptor modulators and uses thereof | |
| US11376250B2 (en) | Spiro-lactam NMDA receptor modulators and uses thereof | |
| EP3746442A1 (en) | Spiro-lactam nmda receptor modulators and uses thereof | |
| WO2019152685A1 (en) | Spiro-lactam nmda receptor modulators and methods of using same | |
| WO2019152688A1 (en) | Spiro-lactam nmda receptor modulators and uses thereof | |
| WO2019152681A1 (en) | Spiro-lactam and bis-spiro-lactam nmda receptor modulators and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19706079 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19706079 Country of ref document: EP Kind code of ref document: A1 |