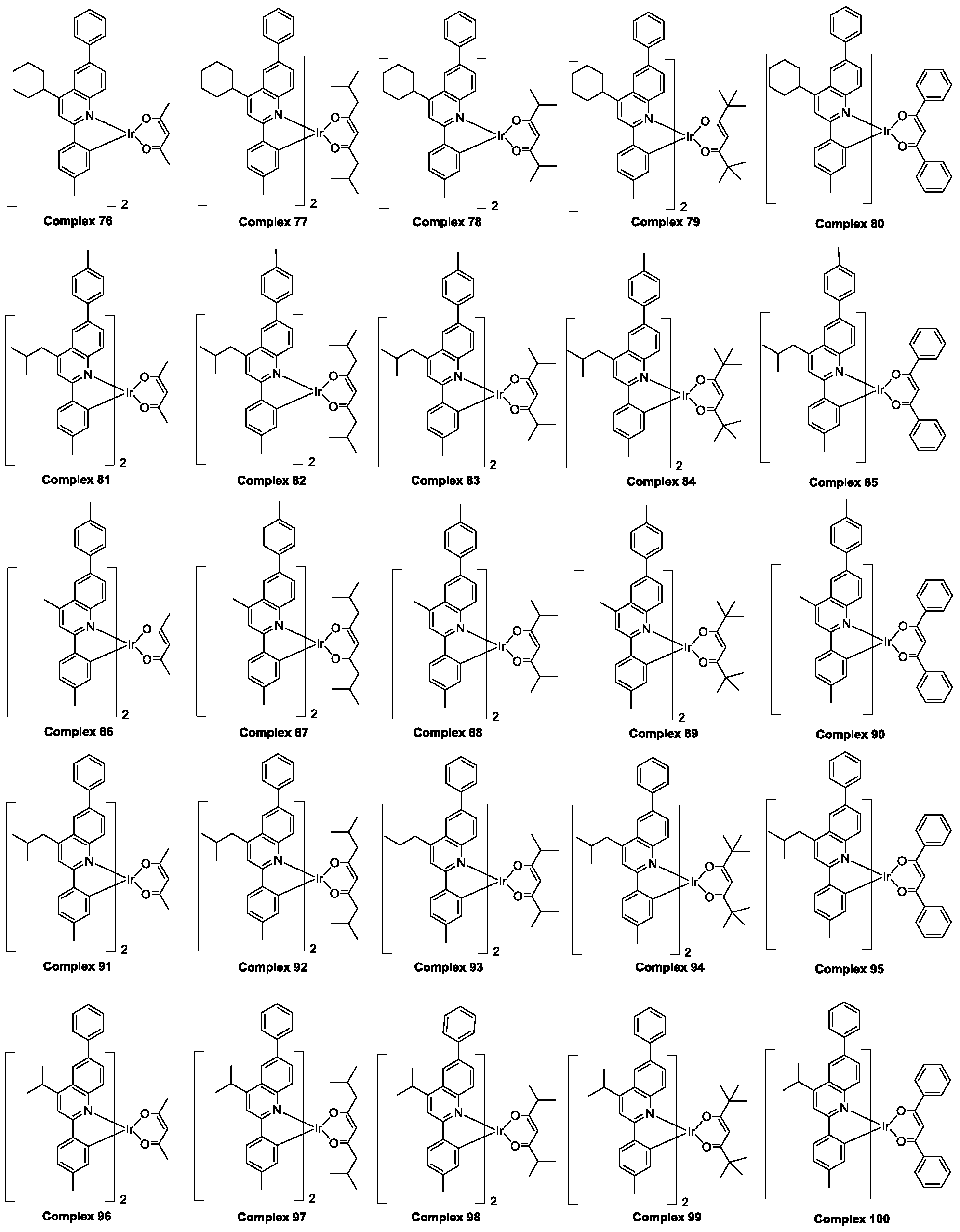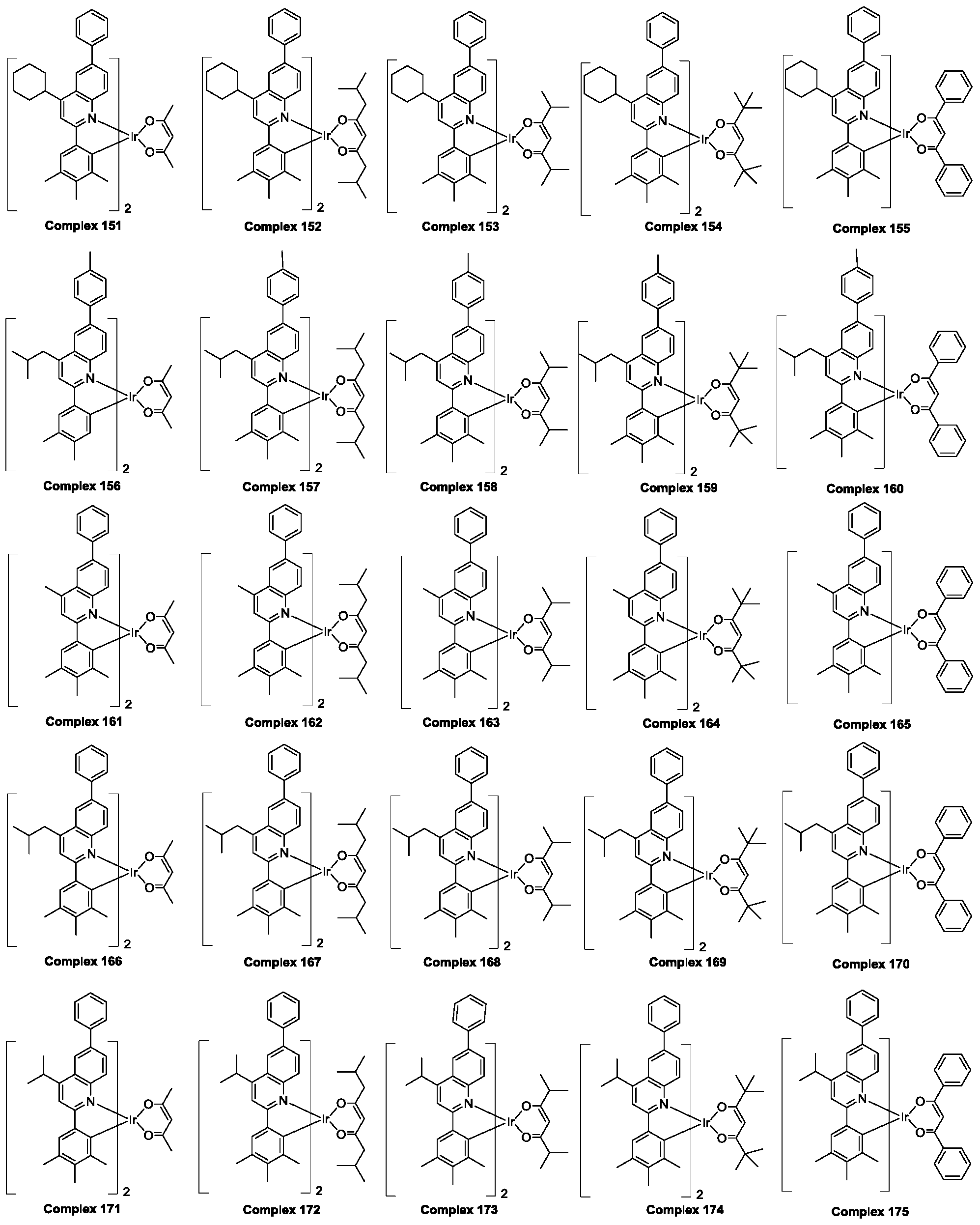WO2019056515A1 - Multi-substituted quinoline-coordinated iridium-hybridized compound, preparation method therefor and application thereof - Google Patents
Multi-substituted quinoline-coordinated iridium-hybridized compound, preparation method therefor and application thereof Download PDFInfo
- Publication number
- WO2019056515A1 WO2019056515A1 PCT/CN2017/109732 CN2017109732W WO2019056515A1 WO 2019056515 A1 WO2019056515 A1 WO 2019056515A1 CN 2017109732 W CN2017109732 W CN 2017109732W WO 2019056515 A1 WO2019056515 A1 WO 2019056515A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- reaction
- methyl
- quinoline
- tolyl
- ethyl acetate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- MTMUQWYGWBBKCQ-UHFFFAOYSA-N CC(C)CC(CC(Nc(cc1)ccc1-c1ccc(C)cc1)=O)=O Chemical compound CC(C)CC(CC(Nc(cc1)ccc1-c1ccc(C)cc1)=O)=O MTMUQWYGWBBKCQ-UHFFFAOYSA-N 0.000 description 1
- PWRUKIPYVGHRFL-UHFFFAOYSA-N CCOC(C(C(C)=O)c1ccccc1)=O Chemical compound CCOC(C(C(C)=O)c1ccccc1)=O PWRUKIPYVGHRFL-UHFFFAOYSA-N 0.000 description 1
- ZMAARKKWBKJGRE-OHSJAEPPSA-N C[C@H]([C@H](C)c1ccc(C)cc1)C(C=C)NC(C(C(C)=O)c1ccccc1)=O Chemical compound C[C@H]([C@H](C)c1ccc(C)cc1)C(C=C)NC(C(C(C)=O)c1ccccc1)=O ZMAARKKWBKJGRE-OHSJAEPPSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/0006—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table compounds of the platinum group
- C07F15/0033—Iridium compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/342—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising iridium
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/18—Metal complexes
- C09K2211/185—Metal complexes of the platinum group, i.e. Os, Ir, Pt, Ru, Rh or Pd
Definitions
- the invention belongs to the technical field of electronic materials, and particularly relates to a polysubstituted quinoline coordinated doping compound and a preparation method thereof, and an application of the compound in an electroluminescent material.
- Organic luminescent materials are relatively inexpensive, and thus organic optoelectronic devices have potential cost advantages over inorganic devices.
- Organic optoelectronic devices include organic light emitting diodes (OLEDs), organic transistors, organic solar cells, and organic photodetectors.
- OLEDs organic light emitting diodes
- organic luminescent materials have performance advantages over traditional materials. For example, the wavelength at which the organic light-emitting layer emits light can usually be easily adjusted with a suitable dopant.
- the energy conversion mode of the organic light-emitting material is to directly convert electrical energy into light energy, and the organic light-emitting material can be closer to the requirement of light adaptability than the inorganic light-emitting material.
- Displays and illuminators based on organic light emitting diode (OLED) technology have a flexible form factor and add a number of artistic elements to the electronic device.
- OLED organic light emitting diode
- the earliest organic electroluminescent device was developed by Eastman Kodak, using an aromatic amine organic small molecule as a hole transport layer and 8-hydroxyquinoline aluminum as a light-emitting layer ("Organic electroluminescent diodes" Tang, Vanslyke et al, Appl Phys Lett, Volume 51, pp. 913-915, 1987).
- Such devices using organic molecules as the core luminescent materials are called organic light-emitting diodes (OLEDs), and can be applied to new fields of display and illumination, and have many advantages and potentials.
- the light-emitting device prepared by the organic material has the advantages of high quantum efficiency, high brightness, high luminous efficiency, etc.; the light-emitting device prepared from the organic light-emitting material has the advantages of lightness, thinness and softness, and can be specially prepared into a flexible device which is another light-emitting material.
- conventional OLEDs can be classified into fluorescent OLEDs and phosphorescent OLEDs. Compared with fluorescent OLEDs (the theoretical luminous efficiency is up to 25%), phosphorescent OLEDs (theoretical luminous efficiency 100%) have become the mainstream of OLED technology research and development due to their higher luminous efficiency.
- the decay of excitons from the singlet excited state to the ground state produces fast luminescence, ie fluorescence.
- the decay of the excitons from the triplet excited state to the ground state produces light, ie phosphorescence.
- phosphorescent metal complexes such as platinum complexes have been shown. They harvest both single and triplet excited states and the potential to achieve 100% internal quantum efficiency. Therefore, the phosphorescent metal complex is a good candidate for the dopant of the organic light-emitting device (OLED) luminescent layer, which is obtained in both academic and industrial fields. A lot of attention. And in the past decade, people have achieved some results on the road to high-margin commercialization. For example, OLED has been applied in advanced displays for smartphones, televisions and digital cameras.
- the ruthenium (III) complex is a widely used phosphorescent material, such as a green light complex: fac-triphenylpyridinium oxime, ie fac-tri(2-phenylpyridinato)iridiums(III)[Ir(ppy) 3 Red light-emitting material: bis(2-(2'-benzothienyl)pyridine-N,C 3' ) (acetylacetone) ruthenium, ie bis[2-(2'-benzothienyl)pyridinato-N, C 3 ' ] (acetylacetonato) iridium (III) [Btp 2 Ir (acac)]; blue luminescent material: bis (4,6-difluorophenylpyridine-N, C2) pyridine formyl hydrazine, ie bis[( 4,6-difluorophenyl)pyridinato-N,C 2 ](picolinato)i
- the actual yield of ligands reported in the literature is about 10%.
- the reaction involves introducing a reaction of alkyl suzuki coupling, which is harsh and costly. Based on the above factors, the material manpower and material cost prepared by the known industrial route are very high, the purification difficulty is extremely large, and a large amount of waste residue and waste liquid are generated.
- the invention discloses a polysubstituted quinoline coordinated doping compound and a preparation method thereof, and the application of the compound in electroluminescent materials and related optoelectronic physical properties.
- the preparation route and method involved therein have the advantages of versatility, high efficiency and suitable mass production cost in preparing the doped complex compound coordinated by the polysubstituted quinoline (quinoline 3, 4, 6 position).
- the heterocyclic compound formed by complexing a polysubstituted quinoline ligand of a polysubstituted phenyl group with Ir can greatly increase the fluorescence quantum efficiency (PLQE) of the luminescent material molecule. Based on the spectral characteristics of these molecules and the function of compound modification, it is clear that such molecules can be used to prepare high quality OLED related devices and applications.
- PQE fluorescence quantum efficiency
- the technical problem to be solved by the present invention is to disclose a polysubstituted quinoline coordinated doped compound and a preparation method thereof, and the use of the compound in electroluminescent materials.
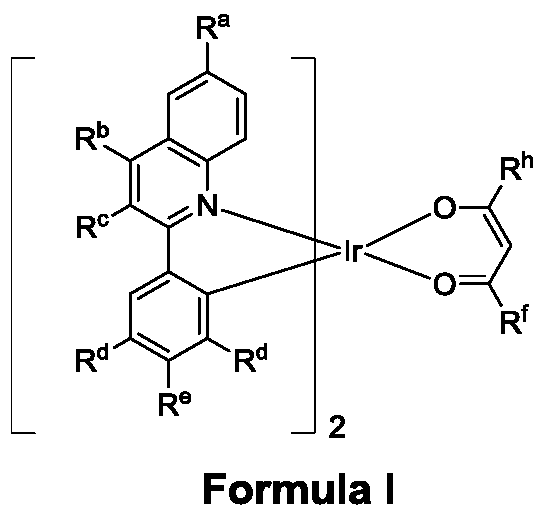
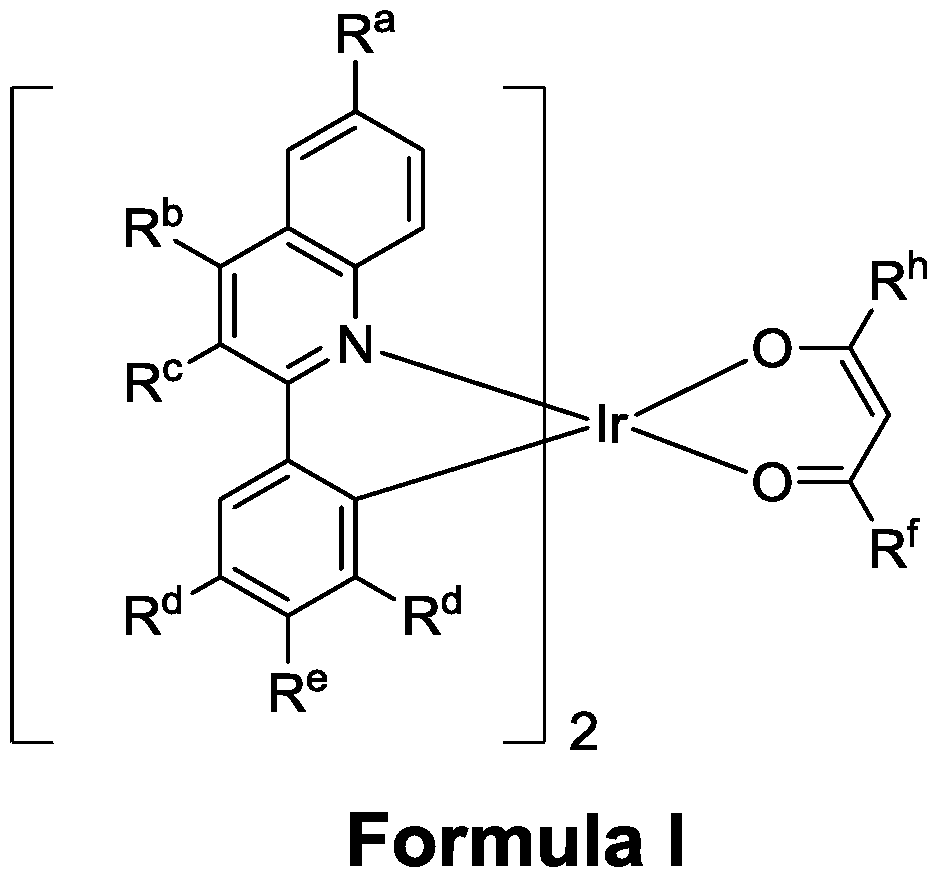
- the technical scheme adopted by the present invention is to provide a polysubstituted quinoline coordinated doping compound having the following molecular structural formula:
- R a , R b , R c R e represents a hydrogen atom, a halogen atom, a (C1-C6) alkyl substituent or an aryl substituent (the related substituent may be substituted, substituted with (C1-C6) alkyl or Aryl substitution).
- R d represents a hydrogen atom or a (C1-C6)alkyl substituent.
- R f , R h represents a (C1-C6)alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with (C1-C6)alkyl or substituted with aryl).
- the (C1-C6)alkyl group means a straight or branched alkyl group having 1 to 6 carbon atoms, and such an alkyl group includes a methyl group, an ethyl group, a n-propyl group, an isopropyl group, an n-butyl group, Isobutyl, tert-butyl, n-pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,2-dimethylpropyl, 2,3-dimethyl Propyl and 1-ethylpropyl, cyclopentyl, cyclohexyl, 2-methyl-3-pentyl, 3,3-dimethyl-2-butyl.
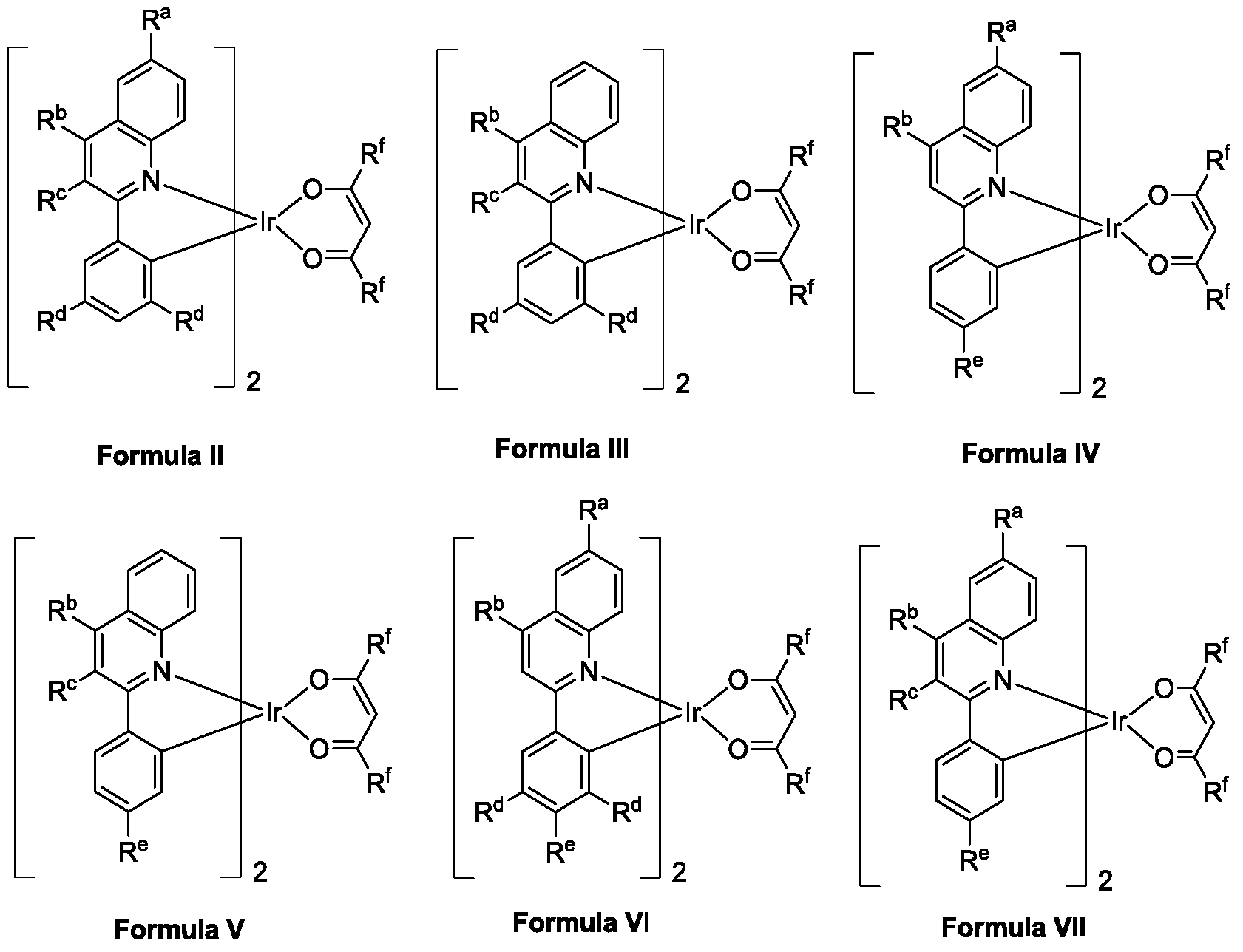
- the molecular structure formula of the polysubstituted quinoline-coordinated hydrazine compound can be divided into the following six structures, which are respectively named as molecular formula II, molecular formula III, molecular formula IV, molecular formula V, molecular formula VI or formula VII:
- R ac , R f is a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R bc , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R ab , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R bc , R e , R f represents a halogen atom or a (C1-C6) alkyl substituent or an aryl substituent;
- R ab , R e , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R ac , R ef represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent.
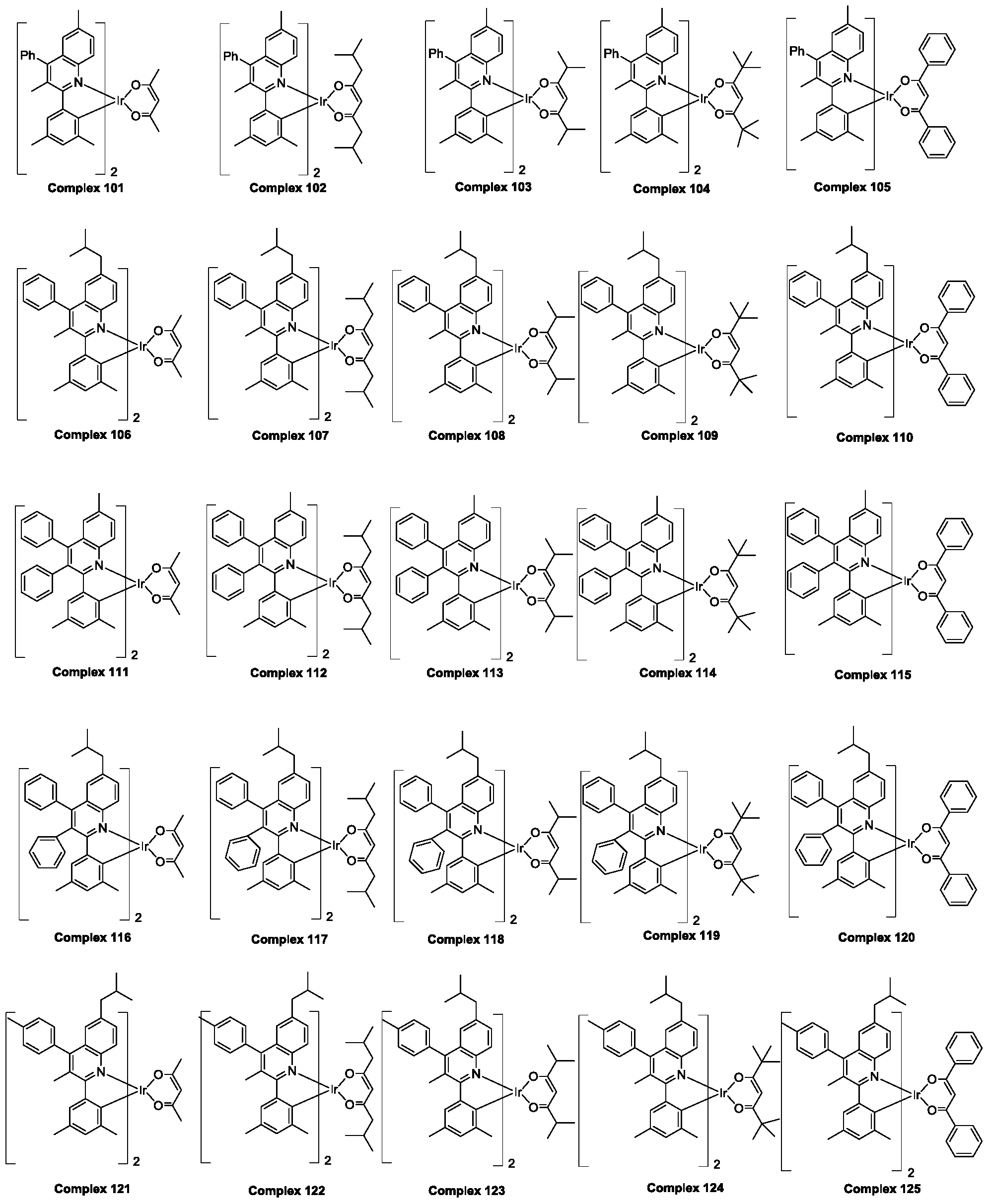
- the polysubstituted quinoline coordinated doped compound is selected from the group consisting of ruthenium complex 1 to ruthenium complex 225 hereinafter.
- the present invention also provides the use of polysubstituted quinoline coordinated doped compounds in electroluminescent materials.
- the invention has the beneficial effects that compared with the prior art, the invention discloses a novel electroluminescent material---the polysubstituted quinoline coordination doped compound compound and the preparation method thereof, and the related photoelectric physics of the compound Properties, and the use of the compound as an electroluminescent material in optoelectronic appliances.
- the preparation route and method involved therein have the advantages of versatility, high efficiency and suitable mass production cost in preparing the doped complex compound coordinated by the polysubstituted quinoline (quinoline 3, 4, 6 position).
- the heterocyclic compound formed by complexing a polysubstituted quinoline ligand of a polysubstituted phenyl group with Ir can greatly increase the fluorescence quantum efficiency (PLQE) of the luminescent material molecule. Based on the spectral characteristics of these molecules and the function of compound modification, it is clear that such molecules can be used to prepare high quality OLED related devices and applications.
- PQE fluorescence quantum efficiency
- Figure 1 is a diagram showing the structure of an OLED using a polysubstituted quinoline coordinated doping compound as a light-emitting material according to the present invention.
- Figure 2 is an emission spectrum of ruthenium complex 7 in CH2Cl2 and PMMA at room temperature.
- FIG. 3 is an illuminating I-V diagram of an OLED device made of ruthenium complex 7 at normal temperature.
- Figure 4 is a graph showing the luminous current efficiency of an OLED device made of yttrium complex 7 at room temperature.
- the polysubstituted quinoline coordinated doping compound of the present invention has a molecular formula of I-VII, and the type I formula is as follows:
- R a , R b , R c R e represents a hydrogen atom, a halogen atom, a (C1-C6) alkyl substituent or an aryl substituent (the related substituent may be substituted, substituted with (C1-C6) alkyl or Aryl substitution).
- R d represents a hydrogen atom or a (C1-C6)alkyl substituent.
- R f , R h represents a (C1-C6)alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with (C1-C6)alkyl or substituted with aryl).
- the (C1-C6)alkyl group means a straight or branched alkyl group having 1 to 6 carbon atoms, and such an alkyl group includes a methyl group, an ethyl group, a n-propyl group, an isopropyl group, an n-butyl group, Isobutyl, tert-butyl, n-pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,2-dimethylpropyl, 2,3-dimethyl Propyl and 1-ethylpropyl, cyclopentyl, cyclohexyl, 2-methyl-3-pentyl, 3,3-dimethyl-2-butyl.
- R f and R h are independent of each other and represent a (C1-C6) alkyl chain. In general, R f and R h are identical to each other;
- R a , R b and R c are independent of each other and represent a hydrogen atom, an alkyl or aryl group of (C1-C6);
- the ligand modification includes 3,5 disubstituted (R d R d ), 4-substituted (R e ), and 3,4,5 trisubstituted (R d R e R d )phenyl in the 2-substituted phenyl group.
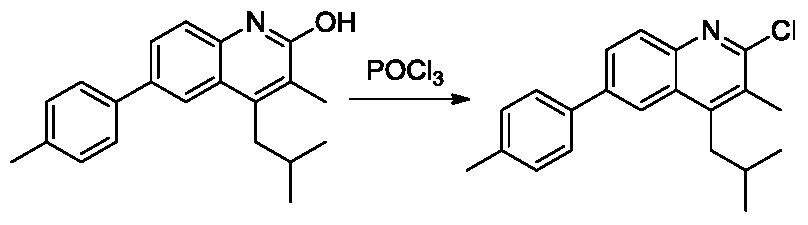
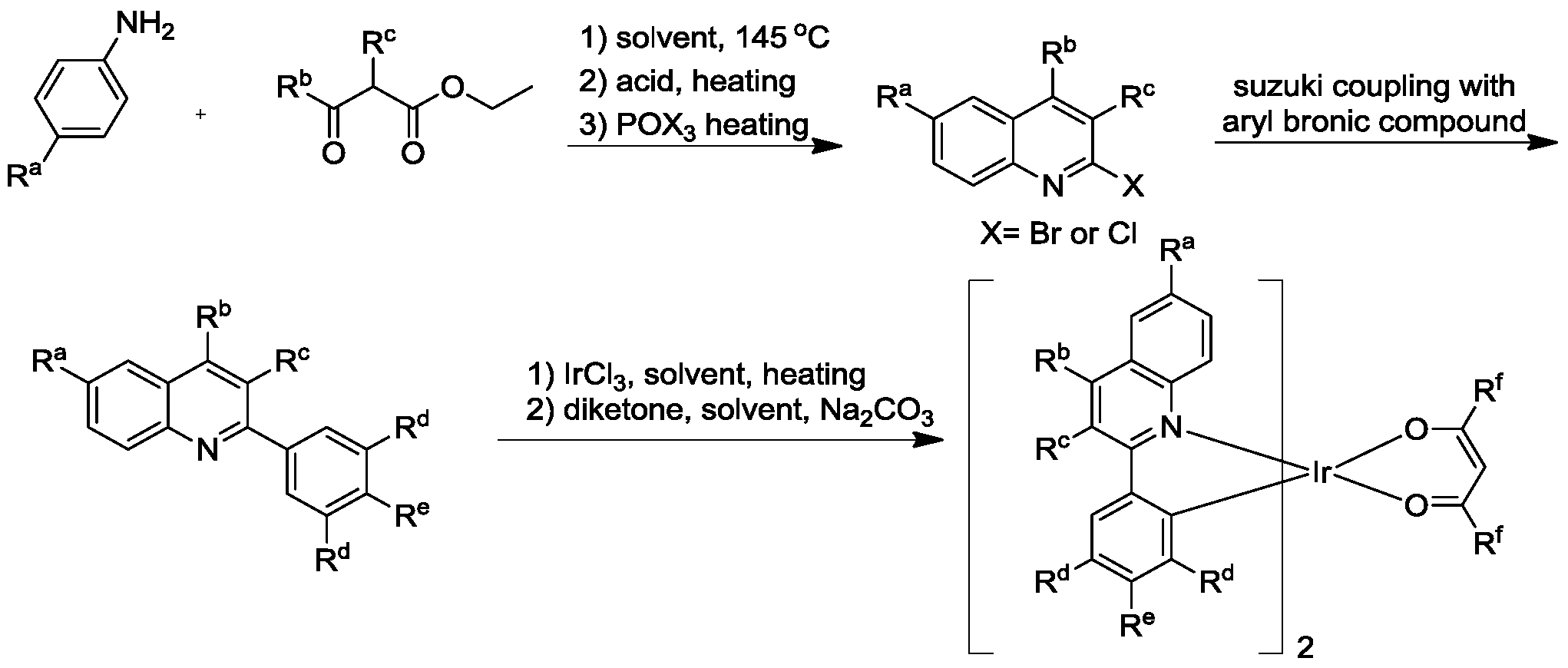
- the specific synthetic route of the ligand is: a polysubstituted quinoline is formed by an amine transesterification reaction, an Skraup ring condensation reaction under acid catalysis, and a hydroxyhalogenation reaction, and a ligand can be obtained by a Suzuki coupling reaction.
- R a , R b , R c represent an H (or oxime) atom or a (C1-C6) alkyl substituent and an aryl substituent (the related substituent may be substituted by a deuterated, containing (C1-C6) alkyl group or Aryl substitution).
- R d represents a H atom or a (C1-C6) alkyl substituent.
- R e represents a H (or hydrazine) atom or a (C1-C6) alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with a (C1-C6) alkyl group or substituted with an aryl group).
- R h represents a (C1-C6)alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with (C1-C6)alkyl or substituted with aryl).
- R f and R h are independent of each other and represent a (C1-C6) alkyl chain. In general, R f and R h are identical to each other.
- R a , R b and R c are independent of each other and represent a hydrogen atom, an alkyl group or an aryl group of (C1-C6).
- the polysubstituted quinoline-coordinated oxime compound further has a structure of the formula 2, the formula 3, the formula 4, the formula 5, the formula 6 or the formula 7, and the structural formula is as follows:
- R a , R c , R d and R e may exist independently or not.
- R ac , R de and R fg are independent of each other and belong to their parent groups (quinoline, phenyl and diketone).
- the substituent on the quinoline, 3,4,6-trisubstituted quinoline represents R a , R b , R c are all present separately, 4,6-disubstituted quinoline represents R b , R c alone exists, 3, 4 - Disubstituted quinolines represent R a , R b is present alone.
- R d and R e may exist independently or not.
- R ac , R f is a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R bc , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R ab , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R bc , R e , R f represents a halogen atom or a (C1-C6) alkyl substituent or an aryl substituent;
- R ab , R e , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
- R ac , R ef represents a halogen atom or a (C1-C6) alkyl substituent or an aryl substituent:
- the polysubstituted quinoline-coordinated hydrazine compound may be of the following structure: an anthracene compound 1-polysubstituted quinoline which is respectively named as a polysubstituted quinoline coordination. Position of the compound compound 225
- dimer 11 (30 mg, 71%).
- dimer 126 (25 mg, 76%).
- 2-(3,5-dimethylphenyl)-6-methyl-4-(p-tolyl)quinoline (64.4 mg, 0.2 mmol)
- ruthenium trichloride trihydrate (14.1 Mg, 0.04 mmol)
- ethylene glycol diethyl ether (3 mL)
- water (1 mL)
- nitrogen was bubbled for three minutes, and the reaction was heated to 120 ° C and stirred for 12 hours.
- the reaction solution was cooled to room temperature, and a solid was precipitated, filtered, and dried in air to give dimer 126 (25 mg, 76%).
- dimer 201 (25 mg, 76%).
- dimer 187 (5) Synthesis of dimer 187.
- 2-(3,5-dimethylphenyl)-4-isobutyl-3,6-dimethylquina to a 15 ml sealed tube
- the ruthenium (65.4 mg, 0.2 mmol), ruthenium trichloride trihydrate (14.1 mg, 0.04 mmol), ethylene glycol diethyl ether (3 mL) and water (1 mL) were placed under nitrogen atmosphere, and the reaction system was heated to reflux for 12 hours.
- the reaction solution was cooled to room temperature, and a solid was precipitated, filtered, and dried in air to give dimer 187 (30 mg, 87%).
- dimer 82 Synthesis of dimer 82.
- 2-(4-methylphenyl)-4-isobutyl-6-(p-tolyl)quinoline 1.1 g, 3 mmol.
- Trihydrate ruthenium trichloride 195 mg, 0.6 mmol
- ethylene glycol ether 15 mL
- water 5 mL
- the reaction system was heated to reflux for 12 hours.
- the reaction solution was cooled to room temperature, and a solid was precipitated, which was filtered, washed with diethyl ether (8 mL ⁇ 3), and dried in air to obtain a dimer 82 (500 mg, 87%).
- dimer 157 (155 mg, 77%).
- 4-isobutyl-6-(p-tolyl)-2-(3,4,5-trimethylphenyl)quinoline 393.3 mg, 1.0 mmol
- trichlorotrichloride Plutonium (65.5 mg, 0.2 mmol)
- ethylene glycol diethyl ether (3 mL)
- water 1 mL
- dimer 6 (180 mg, 89%).
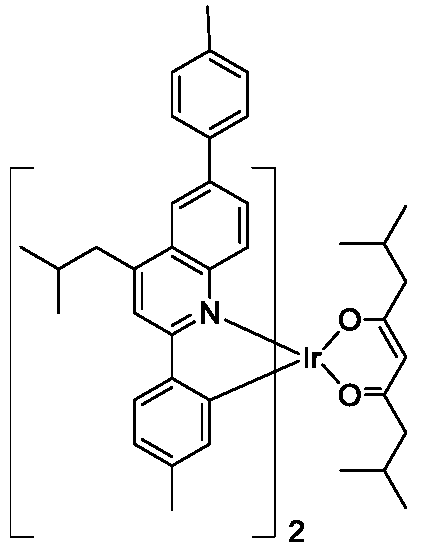
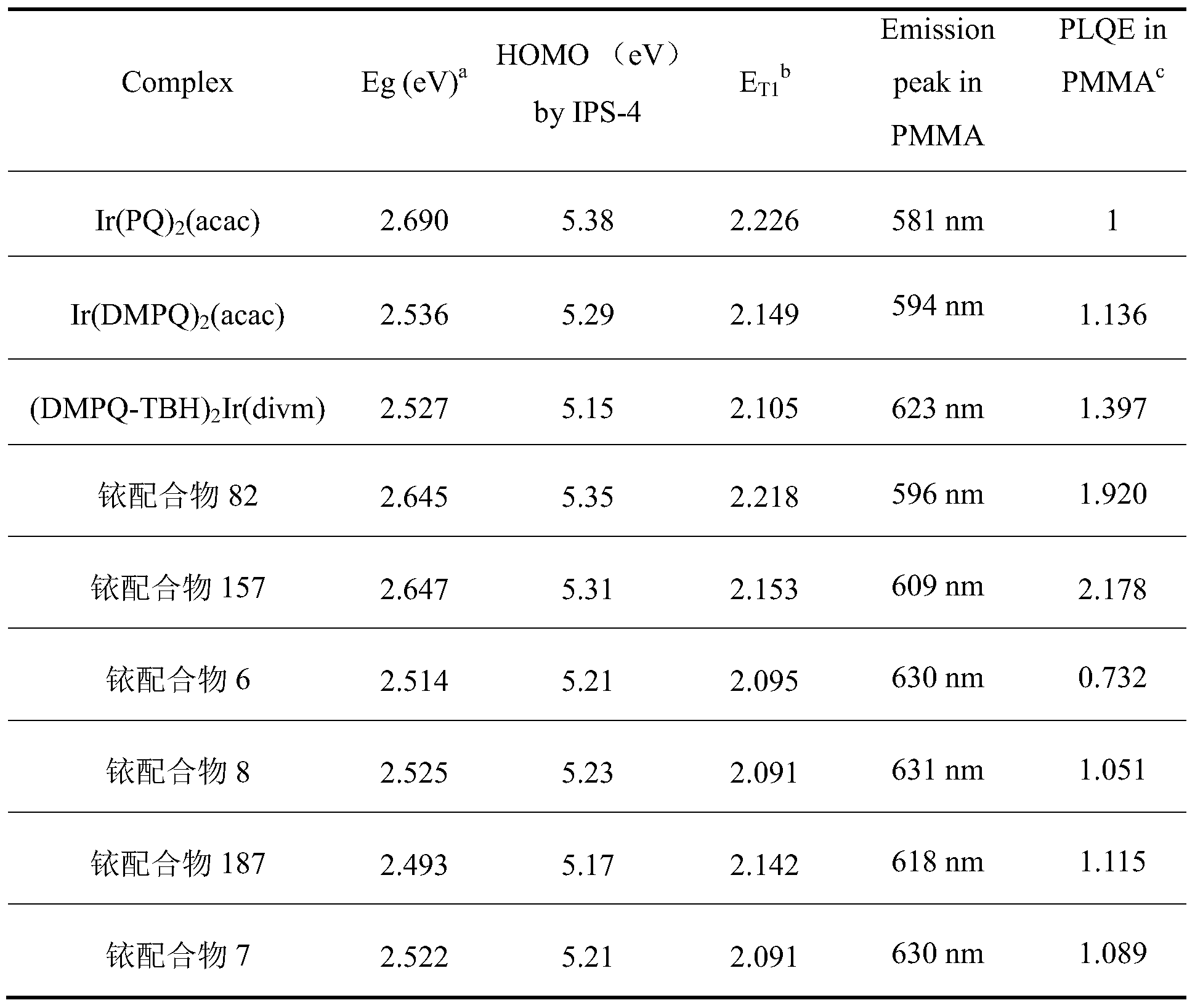
- Example 11 Comparison of optical properties of iridium complexes 6, 7, 8, 11, 82, 126, 157, 187, 201 as phosphorescent materials and conventional phosphorescent red materials
- the ruthenium complexes 6, 7, 8, 11, 82, 126, 157, 187, 201 of the present invention are respectively used as phosphorescent materials and conventional phosphorescent red materials Ir(PQ) 2 (acac), Ir(DMPQ) 2 (acac) and ( DMPQ-TBH) 2 Ir (divm) material optical properties were compared as follows: the bandgap value (Eg) and LUMO value of the material were measured by cyclic voltammetry (CV).
- the whole test process was carried out on the CHI600D electrochemical workstation (Shanghai Chenhua Instrument Co., Ltd.) in the glove box (Lab2000, Etelux), with the Pt column as the working electrode, Ag/AgCl as the reference electrode, and Pt wire as the auxiliary electrode.
- the medium used in the test was 0.1 M tetrabutylammonium hexafluorophosphate (Bu 4 NPF 6 ) in dimethyl diamide (DMF).
- the measured potentials were all within the added ferrocene (Fc). Standard.
- the HOMO values of the materials were directly tested by the Model IPS-4 Ionization Energy Measurement System.
- the fluorescence quantum efficiency (PLQE) of the material is based on the formula (where: ⁇ s is the fluorescence quantum yield of the sample, ⁇ r is the fluorescence quantum yield of the standard, ⁇ is the refractive index of the solution, and A s and A r are the absorption values at the fluorescence excitation wavelength of the sample and the standard, ⁇ s and ⁇ r are the fluorescence integral areas of the samples and standards) calculated using the relative method.
- the material and the known quantum yield standard were configured to the same concentration of polymethyl methacrylate (PMMA) in chloroform solution, spin-coated into a film, and the ultraviolet absorption spectrum was measured under the same measurement conditions (GENESYS).
- Thermo 10S, Thermo and fluorescence spectra (F97pro Fluorescence Spectrophotometer, Prism).
- c PLQE efficiency is one unit of the luminous efficiency of Ir(PQ) 2 (acac) at 298K.
- the following characteristics can be obtained: based on the wavelength and efficiency of Ir(DMPQ) 2 (acac) in PMMA, the Ir complex at the 4, 6 position of quinoline can be used. By shifting the spectrum red by 30 nm, the PLQE in the PMMA film is increased by about 20%. The Ir complex substituted at the 3, 4, and 6 positions of the quinoline can continue to red shift the luminescence spectrum by about 7 nm.
- the ligand in which the substituent is introduced at the para position of the phenyl group substituted at the 2-position of the quinoline can greatly improve the fluorescence quantum efficiency, such as the ruthenium complexes 82 and 157, and the spectrum has a blue shift of 14-27 nm.
- the highest film PLQE is a hetero complex of quinoline 4, 6-substituted and Ir substituted at the 3, 4, and 5 positions of phenyl. From the comparison of Eg (band gap value), it is understood that the above ruthenium complex can be used as a red light material, which is used as a display material to emit light, and the red light saturation is high.
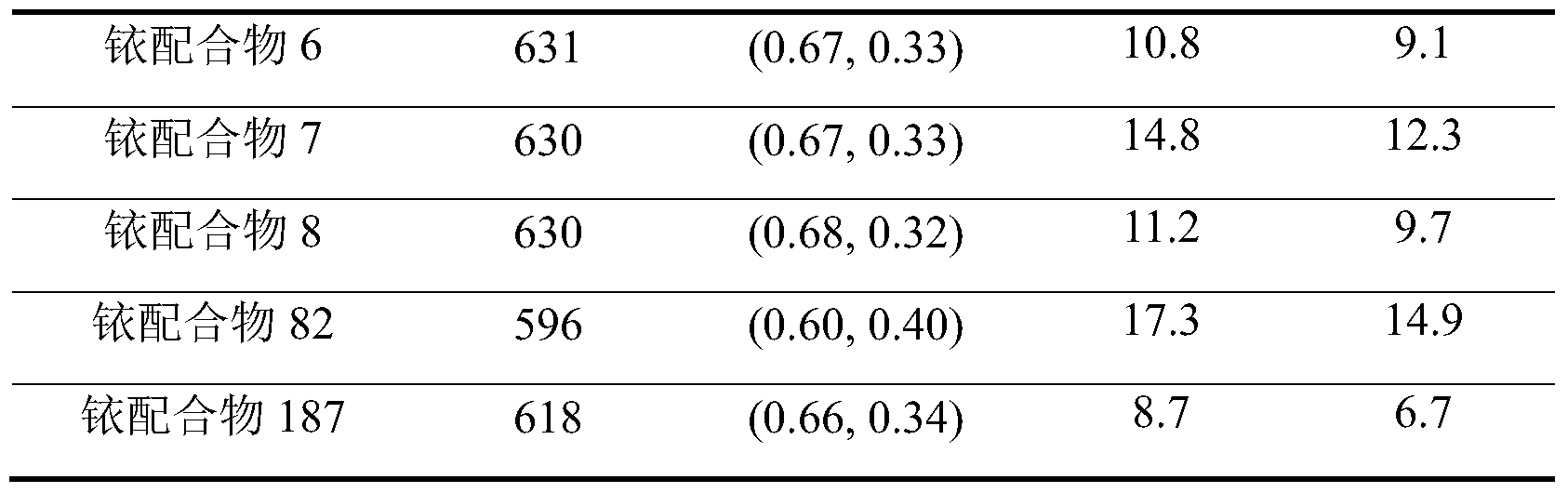
- Example 12 Comparison of ruthenium complex 6,7,8,11,82,126,157,187,201 as a phosphorescent luminescent material and a conventional phosphorescent red light material applied to a device
- the above ruthenium complexes 6, 7, 8, 82, 187 were compared as a phosphorescent luminescent material with conventional phosphorescent red materials Ir(PQ) 2 (acac) and Ir(DMPQ) 2 (acac).
- the structure of the OLED device was designed as follows: ITO/ HAT-CN (10 nm) / TAPC (40 nm) / NPB: red dopant (95: 5, 20 nm) / Balq (10 nm) / Bphen (50 nm) / Liq (1 nm) / Al.
- the crucible containing the OLED organic material and the crucible containing the metal aluminum particles are sequentially placed at the positions of the organic evaporation source and the inorganic evaporation source.
- the cavity is closed, and the initial vacuuming and high vacuum steps are performed, so that the internal vapor deposition degree of the OLED evaporation apparatus reaches 10E-7 Torr.
- the OLED organic evaporation source is turned on, and the OLED organic material is preheated at 100 ° C for a preheating time of 15 minutes to ensure further removal of moisture in the OLED organic material. Then, the organic material that needs to be evaporated is subjected to rapid heating and heat treatment, and the baffle above the evaporation source is opened until the evaporation source of the material has organic material running out, and the crystal oscillator detects the evaporation rate, and then slowly heats up. The temperature rise is 1 ⁇ 5°C until the evaporation rate is stable at 1A/sec. The baffle directly under the mask plate is opened to form the OLED film.
- the film When the organic film on the ITO substrate is observed at the computer end, the film is preset. When thick, close the mask baffle and the baffle directly above the evaporation source to close the evaporation source heater of the organic material. The evaporation process of other organic materials and cathode metal materials is as described above.
- the package is UV-cured for photocuring.
- the encapsulated samples were tested for IVL performance and the IVL equipment was tested with the Mc Science M6100.
- the device data comparison data test is shown in the following table:
- the device electroluminescence wavelength is mainly determined by the photoluminescence of the Ir complex itself.
- the efficiency of the device is also consistent with the PLQE trend of the Ir complex itself. . Therefore, the high PLQE polysubstituted quinoline-coordinated doped compound of the present invention can achieve higher device efficiency in other devices than existing red light materials.
- the polysubstituted quinoline-coordinated oxime compound compounds described herein are suitable for use in a variety of optical and optoelectronic devices, such as light absorbing devices such as solar and light sensors, light emitters, or devices having both light absorbing and light emitting capabilities. And markers for biological applications.
- optical and optoelectronic devices such as light absorbing devices such as solar and light sensors, light emitters, or devices having both light absorbing and light emitting capabilities. And markers for biological applications.
- the use of the polysubstituted quinoline-coordinated doped compound of the present invention in an optoelectronic device will be described below by taking an organic light-emitting diode (OLED) as an example.
- OLED organic light-emitting diode
- the OLED 1000 includes an anode 1004 over a substrate 1002 made of a transparent material such as indium tin oxide; a hole transport material layer (HTL) 1006 and an anode 1004. Connected; the light-emitting function layer 1008 is located above the hole transport material layer 1006, the light-emitting function layer 1008 includes the emitter and the body of the light-emitting material; the electron transport material layer (ETL) 1010 and a metal cathode layer 1012 are sequentially disposed on the light-emitting function. Above layer 1008.
- the OLED and similar light emitting devices may comprise a single layer or a laminate.
- any layer of the single or laminated layer may comprise indium tin oxide (ITO), MoO 3 , Ni 2 O 3 , poly(3,4-ethylenedioxythiophene) (PEDOT), polystyrene sulfonate.
- ITO indium tin oxide
- MoO 3 MoO 3
- Ni 2 O 3 Ni 2 O 3
- PEDOT poly(3,4-ethylenedioxythiophene)
- PDOT polystyrene sulfonate
- the luminescent functional layer 1008 may comprise one or more compounds of the polysubstituted quinoline coordinated doping compound of the present invention, and the ruthenium complex 7 is selectively used in the experiment. Comes with a main material.
- the ETL layers 1010 and 1006 may also comprise one or more polysubstituted quinoline coordinated anthracene compound and another implant layer in proximity to the electrode.
- the material of the injection layer may include (electron injection layer) EIL, (hole injection layer) HIL, and CPL (cap layer), which may be in the form of a single layer or dispersed in a transmission material.
- the host material can be any suitable host material known in the art.
- the luminescent color of the OLED is determined by the luminescent energy (optical energy gap) of the luminescent functional layer 1008, and the luminescent energy (optical energy gap) of the luminescent functional layer 1008 is tuned by tuning the luminescent compound and/or the electronic structure of the host material.
- the hole transport material in the HTL layer 1006 and the electron transport material in the ETL layer 1010 can comprise any suitable hole transport body known in the art.
- the ruthenium complex 7 prepared by the experiment was placed in CH2Cl2 and PMMA for spectral test. The results are shown in Fig. 2. It can be seen from the emission spectrum obtained by the measurement of this experiment that it can be excited by different excitation lights. Strong fluorescence is emitted, the two main peaks are at 629nm and 630nm, respectively, so the ruthenium complex 7 can be used to prepare a high saturation deep red light-emitting device.
- Example 15 Electroluminescence spectrum of a device of ruthenium complex 7.
- the electroluminescence spectroscopy test was performed on the device prepared by using the ruthenium complex 7 as a luminescent material, and the result is shown in FIG. 5, indicating that the ruthenium complex 7 is a high-saturation red light, and the material can fully satisfy the red color in the display. Degree requirements.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Crystallography & Structural Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
本发明属于电子材料技术领域,具体涉及多取代喹啉配位的铱杂配化合物及其制备方法,以及该化合物在电致发光材料方面的应用。The invention belongs to the technical field of electronic materials, and particularly relates to a polysubstituted quinoline coordinated doping compound and a preparation method thereof, and an application of the compound in an electroluminescent material.
利用有机发光材料的光电器器件正变得越来越受欢迎,而它们受欢迎的原因有很多:有机发光材料相对便宜,因此有机光电器件相对于无机器件具有潜在的成本优势。此外,有机材料的固有特性,如其柔韧性,可使它们非常适合于一些特殊应用,如柔性衬底上的加工。有机光电器器件包括有机发光二极管(OLED),有机晶体管,有机太阳能电池和有机光电探测器。对于OLED来说,有机发光材料相比传统材料有性能上的优势。例如,有机发光层发光的波长通常可以很容易用适当的掺杂剂进行调整。Photoelectric devices utilizing organic luminescent materials are becoming more and more popular, and they are popular for a number of reasons: organic luminescent materials are relatively inexpensive, and thus organic optoelectronic devices have potential cost advantages over inorganic devices. In addition, the inherent properties of organic materials, such as their flexibility, make them ideal for special applications such as processing on flexible substrates. Organic optoelectronic devices include organic light emitting diodes (OLEDs), organic transistors, organic solar cells, and organic photodetectors. For OLEDs, organic luminescent materials have performance advantages over traditional materials. For example, the wavelength at which the organic light-emitting layer emits light can usually be easily adjusted with a suitable dopant.
有机发光材料的能量转换模式是直接将电能转化为光能,相对于无机发光材料,有机发光材料可以更贴近对光适应性方面的需求。基于有机发光二极管(OLED)技术制造的显示器和发光器具有柔性外形,并且增加了诸多艺术元素于电子器件中。最早的有机电致发光器件由Eastman Kodak发展出来,通过芳香胺有机小分子作为空穴传输层,8-羟基喹啉铝作为发光层(“Organic electroluminescent diodes”Tang,Vanslyke等,Appl Phys Lett,第51卷,第913-915页,1987年)。这类以有机分子作为核心发光材料的器件称之为有机发光二极管(OLED),可以应用于新型显示和照明领域,具有诸多优点和潜力。有机材料制备的发光器件具有高量子效率、高亮度、高发光效率等优点;由有机发光材料制备成的发光设备外形上具有轻、薄、柔等优势,尤其可制备成柔性设备是其它发光材料无法与之相比的优点。根据核心的电致发光材料进行分类,传统的OLED可以分为荧光类OLED和磷光类OLED。与荧光OLED相比(理论发光效率最高25%),磷光OLED(理论发光效率100%)由于具有更高的发光效率而成为OLED技术研究和发展的主流方向。The energy conversion mode of the organic light-emitting material is to directly convert electrical energy into light energy, and the organic light-emitting material can be closer to the requirement of light adaptability than the inorganic light-emitting material. Displays and illuminators based on organic light emitting diode (OLED) technology have a flexible form factor and add a number of artistic elements to the electronic device. The earliest organic electroluminescent device was developed by Eastman Kodak, using an aromatic amine organic small molecule as a hole transport layer and 8-hydroxyquinoline aluminum as a light-emitting layer ("Organic electroluminescent diodes" Tang, Vanslyke et al, Appl Phys Lett, Volume 51, pp. 913-915, 1987). Such devices using organic molecules as the core luminescent materials are called organic light-emitting diodes (OLEDs), and can be applied to new fields of display and illumination, and have many advantages and potentials. The light-emitting device prepared by the organic material has the advantages of high quantum efficiency, high brightness, high luminous efficiency, etc.; the light-emitting device prepared from the organic light-emitting material has the advantages of lightness, thinness and softness, and can be specially prepared into a flexible device which is another light-emitting material. The advantages that cannot be compared. According to the classification of core electroluminescent materials, conventional OLEDs can be classified into fluorescent OLEDs and phosphorescent OLEDs. Compared with fluorescent OLEDs (the theoretical luminous efficiency is up to 25%), phosphorescent OLEDs (theoretical
激子从单重激发态到基态的衰减产生快速发光,即荧光。激子从三重激发态到基态的衰减产生光,即磷光。由于重金属原子的强自旋轨道耦合(ISC)能非常有效地增强单重激发态和三重激发态之间的电子自旋态交叉变化,磷光金属络合物,如铂络合物,已显示了它们收获单重和三重激发态两者以及达到100%内部量子效率的潜力。因此,磷光金属络合物是作为有机发光器件(OLED)发光层掺杂物的很好候选材料,它在学术和工业领域都以得到 非常多的注意。并且在过去的十年中,人们已经在该技术走向高利润商业化的道路上取得了一定成果,例如,OLED已经在智能手机、电视机和数码相机的先进显示器中得到应用。The decay of excitons from the singlet excited state to the ground state produces fast luminescence, ie fluorescence. The decay of the excitons from the triplet excited state to the ground state produces light, ie phosphorescence. Since the strong spin-orbit coupling (ISC) of heavy metal atoms can very effectively enhance the electron spin state cross-change between the singlet excited state and the triplet excited state, phosphorescent metal complexes such as platinum complexes have been shown. They harvest both single and triplet excited states and the potential to achieve 100% internal quantum efficiency. Therefore, the phosphorescent metal complex is a good candidate for the dopant of the organic light-emitting device (OLED) luminescent layer, which is obtained in both academic and industrial fields. A lot of attention. And in the past decade, people have achieved some results on the road to high-margin commercialization. For example, OLED has been applied in advanced displays for smartphones, televisions and digital cameras.
铱(Ⅲ)络合物是一类应用广泛的磷光材料,比如绿光配合物:fac-三苯基吡啶基铱,即fac-tri(2-phenylpyridinato)iridiums(III)[Ir(ppy)3];红光发光材料:双(2-(2'-苯并噻吩基)吡啶-N,C3')(乙酰丙酮)合铱,即bis[2-(2’-benzothienyl)pyridinato-N,C3’](acetylacetonato)iridium(III)[Btp2Ir(acac)];蓝光发光材料:双(4,6-二氟苯基吡啶-N,C2)吡啶甲酰合铱,即bis[(4,6-difluorophenyl)pyridinato-N,C2](picolinato)iridium(III)(FIrpic)(“100%phosphorescence quantum efficiency of Ir(III)complexes in organic semiconductor films Yuichiro Kawamura,Kenichi Goushi”,Jason Brooks等,Appl Phys Lett,86卷,2005年)。尽管上述有机发光材料在制造OLED面板和照明器具方面已经商业化,然而这些材料仍然有很多提升空间,比如降低材料制备工艺成本、提高材料的基本光电性能、提升最终产品应用感受质量、减少器件制程中材料配套成本,以及提高材料在器件集成后整体耐受性和耐候性等方面。The ruthenium (III) complex is a widely used phosphorescent material, such as a green light complex: fac-triphenylpyridinium oxime, ie fac-tri(2-phenylpyridinato)iridiums(III)[Ir(ppy) 3 Red light-emitting material: bis(2-(2'-benzothienyl)pyridine-N,C 3' ) (acetylacetone) ruthenium, ie bis[2-(2'-benzothienyl)pyridinato-N, C 3 ' ] (acetylacetonato) iridium (III) [Btp 2 Ir (acac)]; blue luminescent material: bis (4,6-difluorophenylpyridine-N, C2) pyridine formyl hydrazine, ie bis[( 4,6-difluorophenyl)pyridinato-N,C 2 ](picolinato)iridium(III)(FIrpic)("100% phosphorescence quantum efficiency of Ir(III) complexes in organic semiconductor films Yuichiro Kawamura,Kenichi Goushi", Jason Brooks, etc. , Appl Phys Lett, Vol. 86, 2005). Although the above organic light-emitting materials have been commercialized in the manufacture of OLED panels and lighting fixtures, these materials still have a lot of room for improvement, such as reducing the cost of material preparation processes, improving the basic photoelectric performance of materials, improving the quality of the final product application, and reducing the device process. The cost of matching materials and improving the overall resistance and weather resistance of the materials after device integration.
现有技术中,(US20120181511)公开了5位单取代的喹啉配体及其Ir配合物的合成及其在有机光电器件中的应用;(US2016093808)公开了4或者5位单取代的喹啉配体及其Ir配合物的合成及其在有机光电器件中的应用;(WO2016072780)公开了4,6位取代喹啉配体及其Ir配合物的合成及其在有机光电器件中的应用,该配体合成方法所使用的红光材料的分子结构的合成路径以丙二酸(胡萝卜酸)为起始原料,通过Skraup合成法得到双卤素取代的关键中间体。此中间体通过两步suzuki偶联得到配体,最后通过配位得到最终红光材料。此条路径每一步反应都有非常多的竞争性副反应,需要通过柱层析仔细分离,效率非常低,文献报道的配体实际收率最高在10%左右。并且反应中还涉及到引入烷基suzuki偶联的反应,条件苛刻,成本高。综合以上因素,通过已知的工业路径制备得到的材料人力和材料成本非常高,提纯难度异常大,并且产生大量的废渣和废液。In the prior art, (US20120181511) discloses the synthesis of 5-position monosubstituted quinoline ligands and their Ir complexes and their use in organic optoelectronic devices; (US2016093808) discloses 4- or 5-position monosubstituted quinolines. Synthesis of ligands and their Ir complexes and their use in organic optoelectronic devices; (WO2016072780) discloses the synthesis of 4,6-substituted substituted quinoline ligands and their Ir complexes and their use in organic optoelectronic devices, The synthetic route of the molecular structure of the red light material used in the ligand synthesis method is based on malonic acid (carotic acid), and a key intermediate of dihalogen substitution is obtained by Skraup synthesis. This intermediate was obtained by a two-step suzuki coupling to obtain a final red light material by coordination. Each step of this path has a lot of competitive side reactions, which need to be carefully separated by column chromatography, and the efficiency is very low. The actual yield of ligands reported in the literature is about 10%. Moreover, the reaction involves introducing a reaction of alkyl suzuki coupling, which is harsh and costly. Based on the above factors, the material manpower and material cost prepared by the known industrial route are very high, the purification difficulty is extremely large, and a large amount of waste residue and waste liquid are generated.
本发明公开了一种多取代喹啉配位的铱杂配化合物及其制备方法,以及该化合物在电致发光材料方面的应用和相关光电物理性质。其中涉及的制备路径和方法在制备多取代喹啉(喹啉3,4,6位置)配位的铱杂配化合物上具有通用性、高效性和量产成本适合等优点。2位为多取代苯基的多取代喹啉配体与Ir络合形成的杂配化合物,可以大幅度提高发光材料分子的荧光量子效率(PLQE)。据这些分子的光谱特征和化合物改造功能,很明确的这类分子可以用来制备高质量的OLED相关器件和应用器具。The invention discloses a polysubstituted quinoline coordinated doping compound and a preparation method thereof, and the application of the compound in electroluminescent materials and related optoelectronic physical properties. The preparation route and method involved therein have the advantages of versatility, high efficiency and suitable mass production cost in preparing the doped complex compound coordinated by the polysubstituted quinoline (quinoline 3, 4, 6 position). The heterocyclic compound formed by complexing a polysubstituted quinoline ligand of a polysubstituted phenyl group with Ir can greatly increase the fluorescence quantum efficiency (PLQE) of the luminescent material molecule. Based on the spectral characteristics of these molecules and the function of compound modification, it is clear that such molecules can be used to prepare high quality OLED related devices and applications.
发明内容 Summary of the invention
本发明所要解决的技术问题是:公开了一种多取代喹啉配位的铱杂配化合物及其制备方法,以及该化合物在电致发光材料方面的应用。The technical problem to be solved by the present invention is to disclose a polysubstituted quinoline coordinated doped compound and a preparation method thereof, and the use of the compound in electroluminescent materials.
为了解决上述技术问题,本发明所采用的技术方案是:提供一种多取代喹啉配位的铱杂配化合物,具有以下分子结构式:In order to solve the above technical problems, the technical scheme adopted by the present invention is to provide a polysubstituted quinoline coordinated doping compound having the following molecular structural formula:
Ra,Rb,RcRe代表氢原子,氘原子,(C1-C6)烷基取代基或芳基取代基(相关取代基可以被氘代、含有(C1-C6)烷基取代或者芳基取代)。Rd代表氢原子或(C1-C6)烷基取代基。Rf,Rh代表(C1-C6)烷基取代基和芳基取代基(相关取代基可以被氘代、含有(C1-C6)烷基取代或者芳基取代)。R a , R b , R c R e represents a hydrogen atom, a halogen atom, a (C1-C6) alkyl substituent or an aryl substituent (the related substituent may be substituted, substituted with (C1-C6) alkyl or Aryl substitution). R d represents a hydrogen atom or a (C1-C6)alkyl substituent. R f , R h represents a (C1-C6)alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with (C1-C6)alkyl or substituted with aryl).
所述(C1-C6)烷基指直链或者支链的含有1-6个碳原子的烷基,这类烷基包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、特丁基、正戊基、1-甲基丁基、2-甲基丁基、3-甲基丁基、1,2-二甲基丙基、2,3-二甲基丙基和1-乙基丙基、环戊基、环己基、2-甲基-3-戊基、3,3-二甲基-2-丁基。The (C1-C6)alkyl group means a straight or branched alkyl group having 1 to 6 carbon atoms, and such an alkyl group includes a methyl group, an ethyl group, a n-propyl group, an isopropyl group, an n-butyl group, Isobutyl, tert-butyl, n-pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,2-dimethylpropyl, 2,3-dimethyl Propyl and 1-ethylpropyl, cyclopentyl, cyclohexyl, 2-methyl-3-pentyl, 3,3-dimethyl-2-butyl.
所述多取代喹啉配位的铱杂配化合物的分子结构式可分为以下六种结构,分别命名为分子式Ⅱ、分子式Ⅲ、分子式Ⅳ、分子式Ⅴ、分子式Ⅵ或分子式Ⅶ: The molecular structure formula of the polysubstituted quinoline-coordinated hydrazine compound can be divided into the following six structures, which are respectively named as molecular formula II, molecular formula III, molecular formula IV, molecular formula V, molecular formula VI or formula VII:
分子式Ⅱ:Ra-c、Rf为氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula II: R ac , R f is a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅲ:Rb-c、Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula III: R bc , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅳ:Ra-b、Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula IV: R ab , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅴ:Rb-c,Re,Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基;Molecular formula V: R bc , R e , R f represents a halogen atom or a (C1-C6) alkyl substituent or an aryl substituent;
分子式Ⅵ:Ra-b、Re,Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula VI: R ab , R e , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅶ:Ra-c、Re-f代表氘原子或者(C1-C6)烷基取代基或芳基取代基。 Formula VII: R ac , R ef represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent.
所述多取代喹啉配位的铱杂配化合物是从由下文中铱配合物1至铱配合物225组成的群组中选择。The polysubstituted quinoline coordinated doped compound is selected from the group consisting of
上述多取代喹啉配位的铱杂配化合物的方法,其合成路径如下:The above synthetic method of the polysubstituted quinoline coordinated doping compound is as follows:
此外,本发明还提供了多取代喹啉配位的铱杂配化合物在电致发光材料方面的应用.In addition, the present invention also provides the use of polysubstituted quinoline coordinated doped compounds in electroluminescent materials.
本发明的有益效果是:与现有技术相比,本发明公开了一种新型电致发光材料----多取代喹啉配位的铱杂配化合物及其制备方法,该化合物相关光电物理性质,以及该化合物作为电致发光材料在光电电器中的应用。其中涉及的制备路径和方法在制备多取代喹啉(喹啉3,4,6位置)配位的铱杂配化合物上具有通用性、高效性和量产成本适合等优点。2位为多取代苯基的多取代喹啉配体与Ir络合形成的杂配化合物,可以大幅度提高发光材料分子的荧光量子效率(PLQE)。据这些分子的光谱特征和化合物改造功能,很明确的这类分子可以用来制备高质量的OLED相关器件和应用器具。The invention has the beneficial effects that compared with the prior art, the invention discloses a novel electroluminescent material---the polysubstituted quinoline coordination doped compound compound and the preparation method thereof, and the related photoelectric physics of the compound Properties, and the use of the compound as an electroluminescent material in optoelectronic appliances. The preparation route and method involved therein have the advantages of versatility, high efficiency and suitable mass production cost in preparing the doped complex compound coordinated by the polysubstituted quinoline (quinoline 3, 4, 6 position). The heterocyclic compound formed by complexing a polysubstituted quinoline ligand of a polysubstituted phenyl group with Ir can greatly increase the fluorescence quantum efficiency (PLQE) of the luminescent material molecule. Based on the spectral characteristics of these molecules and the function of compound modification, it is clear that such molecules can be used to prepare high quality OLED related devices and applications.
图1为使用本发明所述多取代喹啉配位的铱杂配化合物作为致电发光材料的OLED结构图BRIEF DESCRIPTION OF THE DRAWINGS Figure 1 is a diagram showing the structure of an OLED using a polysubstituted quinoline coordinated doping compound as a light-emitting material according to the present invention.
图2为室温下铱配合物7在CH2Cl2、PMMA中的发射光谱图Figure 2 is an emission spectrum of ruthenium complex 7 in CH2Cl2 and PMMA at room temperature.
图3为常温下铱配合物7制成的OLED器件的发光I-V图3 is an illuminating I-V diagram of an OLED device made of ruthenium complex 7 at normal temperature.
图4为常温下铱配合物7制成的OLED器件的发光电流效率图Figure 4 is a graph showing the luminous current efficiency of an OLED device made of yttrium complex 7 at room temperature.
图5铱配合物7制成的OLED器件的的电致发光光谱Figure 5: Electroluminescence spectrum of an OLED device made of complex 7
本发明所述多取代喹啉配位的铱杂配化合物,其分子式分为Ⅰ-Ⅶ型,如下所示Ⅰ型分子式为: The polysubstituted quinoline coordinated doping compound of the present invention has a molecular formula of I-VII, and the type I formula is as follows:
Ra,Rb,RcRe代表氢原子,氘原子,(C1-C6)烷基取代基或芳基取代基(相关取代基可以被氘代、含有(C1-C6)烷基取代或者芳基取代)。Rd代表氢原子或(C1-C6)烷基取代基。Rf,Rh代表(C1-C6)烷基取代基和芳基取代基(相关取代基可以被氘代、含有(C1-C6)烷基取代或者芳基取代)。R a , R b , R c R e represents a hydrogen atom, a halogen atom, a (C1-C6) alkyl substituent or an aryl substituent (the related substituent may be substituted, substituted with (C1-C6) alkyl or Aryl substitution). R d represents a hydrogen atom or a (C1-C6)alkyl substituent. R f , R h represents a (C1-C6)alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with (C1-C6)alkyl or substituted with aryl).
所述(C1-C6)烷基指直链或者支链的含有1-6个碳原子的烷基,这类烷基包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、特丁基、正戊基、1-甲基丁基、2-甲基丁基、3-甲基丁基、1,2-二甲基丙基、2,3-二甲基丙基和1-乙基丙基、环戊基、环己基、2-甲基-3-戊基、3,3-二甲基-2-丁基。The (C1-C6)alkyl group means a straight or branched alkyl group having 1 to 6 carbon atoms, and such an alkyl group includes a methyl group, an ethyl group, a n-propyl group, an isopropyl group, an n-butyl group, Isobutyl, tert-butyl, n-pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,2-dimethylpropyl, 2,3-dimethyl Propyl and 1-ethylpropyl, cyclopentyl, cyclohexyl, 2-methyl-3-pentyl, 3,3-dimethyl-2-butyl.
在Ⅰ型分子中,Rf和Rh,是相互独立的,代表(C1-C6)烷基链.一般情况下,Rf和Rh相互一致;In the type I molecule, R f and R h are independent of each other and represent a (C1-C6) alkyl chain. In general, R f and R h are identical to each other;
在Ⅰ型分子中,Ra,Rb和Rc是相互独立的,代表一个氢原子,(C1-C6)的烷基或者芳基;In the type I molecule, R a , R b and R c are independent of each other and represent a hydrogen atom, an alkyl or aryl group of (C1-C6);
上述Ⅰ型分子的合成路经如下图所示:The synthetic route of the above type I molecules is shown in the following figure:
配体修饰包括2位取代的苯基中3,5双取代(RdRd)、4位取代(Re)和3,4,5三取代(RdReRd)苯基。配体具体合成路径为:通过胺酯交换反应、酸催化下的Skraup环缩合反 应和羟基卤化反应生成多取代的喹啉,再通过Suzuki偶联反应即可以得到配体。(如上所示化学方程式)此路径避免了烷基的suzuki偶联的反应和多个反应位点竞争的副反应,可以提高合成效率和降低提纯难度。由喹啉配体、二酮与Ir配位生成Ir的多取代的喹啉配体配位杂配物的方法为常规方法。The ligand modification includes 3,5 disubstituted (R d R d ), 4-substituted (R e ), and 3,4,5 trisubstituted (R d R e R d )phenyl in the 2-substituted phenyl group. The specific synthetic route of the ligand is: a polysubstituted quinoline is formed by an amine transesterification reaction, an Skraup ring condensation reaction under acid catalysis, and a hydroxyhalogenation reaction, and a ligand can be obtained by a Suzuki coupling reaction. (Chemical equation as shown above) This route avoids the suzuki coupling reaction of the alkyl group and the side reaction which competes with a plurality of reaction sites, which can improve the synthesis efficiency and reduce the difficulty of purification. A method of synthesizing a polysubstituted quinoline ligand coordination complex of Ir by a quinoline ligand, a diketone and Ir is a conventional method.
上述Ra,Rb,Rc代表H(或者氘)原子或者(C1-C6)烷基取代基和芳基取代基(相关取代基可以被氘代、含有(C1-C6)烷基取代或者芳基取代)。Rd代表H原子或(C1-C6)烷基取代基。Re代表代表H(或者氘)原子或者(C1-C6)烷基取代基和芳基取代基(相关取代基可以被氘代、含有(C1-C6)烷基取代或者芳基取代).Rf,Rh代表(C1-C6)烷基取代基和芳基取代基(相关取代基可以被氘代、含有(C1-C6)烷基取代或者芳基取代)。The above R a , R b , R c represent an H (or oxime) atom or a (C1-C6) alkyl substituent and an aryl substituent (the related substituent may be substituted by a deuterated, containing (C1-C6) alkyl group or Aryl substitution). R d represents a H atom or a (C1-C6) alkyl substituent. R e represents a H (or hydrazine) atom or a (C1-C6) alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with a (C1-C6) alkyl group or substituted with an aryl group). f , R h represents a (C1-C6)alkyl substituent and an aryl substituent (the related substituent may be deuterated, substituted with (C1-C6)alkyl or substituted with aryl).
在Ⅰ型分子式中,Rf和Rh,是相互独立的,代表(C1-C6)烷基链.一般情况下,Rf和Rh相互一致。In the formula I formula, R f and R h are independent of each other and represent a (C1-C6) alkyl chain. In general, R f and R h are identical to each other.
在Ⅰ型分子中,Ra,Rb和Rc是相互独立的,代表一个氢原子,(C1-C6)的烷基或者芳基。In the type I molecule, R a , R b and R c are independent of each other and represent a hydrogen atom, an alkyl group or an aryl group of (C1-C6).
上述多取代喹啉配位的铱杂配化合物还具有分子式2、分子式3、分子式4、分子式5、分子式6或分子式7的结构,其结构式如下:The polysubstituted quinoline-coordinated oxime compound further has a structure of the formula 2, the formula 3, the formula 4, the formula 5, the formula 6 or the formula 7, and the structural formula is as follows:
每一个Ra,Rc,Rd和Re可以独立存在或者不存在。Ra-c,Rd-e和Rf-g之间相互独立,分别属于其的母体基团(喹啉、苯基和二酮)。喹啉上的取代基,3,4,6–三取代喹啉代表Ra,Rb,Rc都单独存在,4,6–二取代喹啉代表Rb,Rc单独存在,3,4–二取代喹啉代表Ra,Rb单独存在。在喹啉2位的苯基上,Rd和Re可以独立存在或者不存在。Each of R a , R c , R d and R e may exist independently or not. R ac , R de and R fg are independent of each other and belong to their parent groups (quinoline, phenyl and diketone). The substituent on the quinoline, 3,4,6-trisubstituted quinoline represents R a , R b , R c are all present separately, 4,6-disubstituted quinoline represents R b , R c alone exists, 3, 4 - Disubstituted quinolines represent R a , R b is present alone. On the phenyl group at the 2-position of quinoline, R d and R e may exist independently or not.
分子式2-7中取代基的关系,它的群类和定义描述如下:The relationship of the substituents in the formula 2-7, its group and definition are described as follows:
分子式Ⅱ:Ra-c、Rf为氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula II: R ac , R f is a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅲ:Rb-c、Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula III: R bc , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅳ:Ra-b、Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula IV: R ab , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅴ:Rb-c,Re,Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基;Molecular formula V: R bc , R e , R f represents a halogen atom or a (C1-C6) alkyl substituent or an aryl substituent;
分子式Ⅵ:Ra-b、Re,Rf代表氘原子或者(C1-C6)烷基取代基或芳基取代基,Rd代表(C1-C6)烷基取代基;Molecular formula VI: R ab , R e , R f represents a halogen atom or a (C1-C6)alkyl substituent or an aryl substituent, and R d represents a (C1-C6)alkyl substituent;
分子式Ⅶ:Ra-c、Re-f代表氘原子或者(C1-C6)烷基取代基或芳基取代基:Formula VII: R ac , R ef represents a halogen atom or a (C1-C6) alkyl substituent or an aryl substituent:
根据上述对Ra-Rh的定义,所述多取代喹啉配位的铱杂配化合物可以是以下结构:分别命名为多取代喹啉配位的铱杂配化合物1-多取代喹啉配位的铱杂配化合物225 According to the above definition of R a -R h , the polysubstituted quinoline-coordinated hydrazine compound may be of the following structure: an anthracene compound 1-polysubstituted quinoline which is respectively named as a polysubstituted quinoline coordination. Position of the compound compound 225
下面以上述多取代喹啉配位的铱杂配化合物6,7,8,11,82,126,157,187,201为例(以下简称为铱配合物6,7,8,11,82,126,157,187,201),对部分多取代喹啉配位的铱杂配化合物的制备方法及其光学性质以及作为有机发光材料在发光器件方面的应用做具体说明 Hereinafter, the above-mentioned polysubstituted quinoline-coordinated hydrazine compound 6,7,8,11,82,126,157,187,201 is exemplified (hereinafter referred to as ruthenium complex 6,7,8,11,82,126,157,187,201). , preparation method and optical properties of some polysubstituted quinoline coordination doped compounds, and specific application as an organic light-emitting material in light-emitting devices
实施例1铱配合物11的制备Example 1 Preparation of ruthenium complex 11
Ra=对甲苯基,Rb=甲基,Rc=苯基,Rd=甲基,Re=氢,Rf=甲基R a = p-tolyl, R b = methyl, R c = phenyl, R d = methyl, R e = hydrogen, R f = methyl
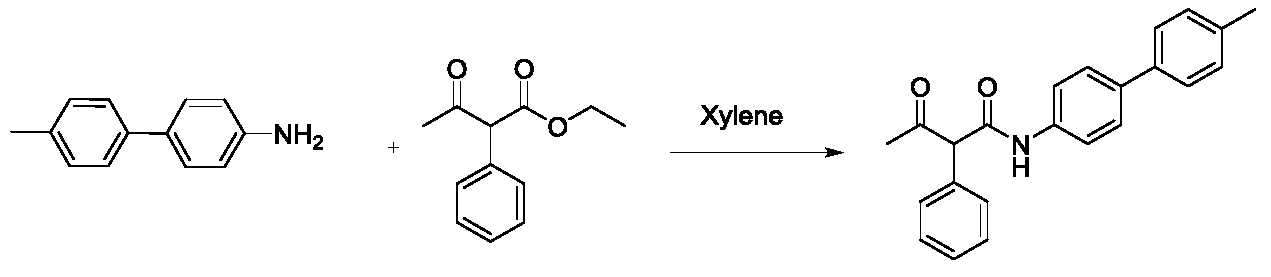
(1)N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代-2-苯基丁酰胺的合成。100mL的封管中加入4'-甲基-[1,1'-联苯]-4-胺(1g,5.5mmol),2-苯基乙酰乙酸乙酯(3.1g,15mmol)和二甲苯(15mL),反应体系N2鼓泡三分后,缓慢升温至145℃,并在145℃条件下搅拌16小时。TLC(石油醚:乙酸乙酯=5:1)显示反应完全,将反应液冷至室温,产物析出,过滤,空气中干燥得淡黄色N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代-2-苯基丁酰胺(500mg,产率26%)。1H NMR(300MHz,DMSO)δ10.40(s,1H),7.66(d,J=8.7Hz,2H),7.60(d,J=8.7Hz,2H),7.53(d,J=8.1Hz,2H),7.38–7.31(m,5H),7.24(d,J=7.8Hz,2H),5.00(s,1H),2.33(s,3H),2.19(s,3H)。(1) Synthesis of N-(4'-methyl-[1,1'-biphenyl]-4-yl)-3-oxo-2-phenylbutanamide. To a 100 mL sealed tube was added 4'-methyl-[1,1'-biphenyl]-4-amine (1 g, 5.5 mmol), ethyl 2-phenylacetoacetate (3.1 g, 15 mmol) and xylene ( 15 mL), after the reaction system N 2 was bubbled for three minutes, the temperature was slowly raised to 145 ° C, and stirred at 145 ° C for 16 hours. TLC (petroleum ether: ethyl acetate = 5:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, product was precipitated, filtered, and dried in air to give a pale yellow N-(4'-methyl-[1,1'- Biphenyl]-4-yl)-3-oxo-2-phenylbutanamide (500 mg, yield 26%). 1 H NMR (300MHz, DMSO) δ10.40 (s, 1H), 7.66 (d, J = 8.7Hz, 2H), 7.60 (d, J = 8.7Hz, 2H), 7.53 (d, J = 8.1Hz, 2H), 7.38 - 7.31 (m, 5H), 7.24 (d, J = 7.8 Hz, 2H), 5.00 (s, 1H), 2.33 (s, 3H), 2.19 (s, 3H).
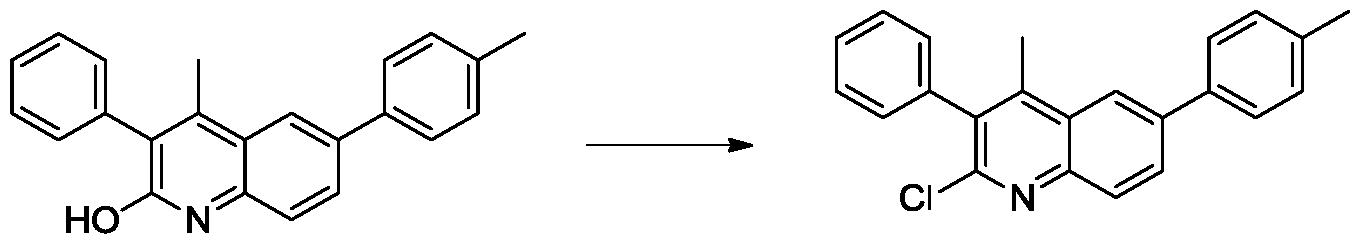
(2)4-甲基-3-苯基-6-(对甲苯基)喹啉-2-醇的合成。100mL三颈烧瓶中加入N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代-2-苯基丁酰胺(500mg,1.45mmol),氮气置换反应瓶氛围,3mL浓硫酸逐滴滴加入反应体系。将反应体系加热至50℃,搅拌12小时。TLC(石油醚:乙酸乙酯=1:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,空气中干燥得到淡黄色4-甲基-3-苯基-6-(对甲苯基)喹啉-2-醇(320mg,产率68%)。1H NMR(500MHz,DMSO)δ11.83(s,1H),7.96(s,1H),7.81(d,J=8.5Hz,1H),7.64(d,J=8.0Hz,2H),7.46-7.43(m,2H),7.42–7.36(m,2H),7.28(d,J=7.5Hz,2H),7.26(d,J=7.5Hz,2H),2.35(s,3H),2.34(s,3H).(2) Synthesis of 4-methyl-3-phenyl-6-(p-tolyl)quinolin-2-ol. N-(4'-methyl-[1,1'-biphenyl]-4-yl)-3-oxo-2-phenylbutanamide (500 mg, 1.45 mmol) was added to a 100 mL three-necked flask. The reaction flask atmosphere was replaced, and 3 mL of concentrated sulfuric acid was added dropwise to the reaction system. The reaction system was heated to 50 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 1:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, and dried in air to give pale yellow 4-methyl-3-phenyl -6-(p-tolyl)quinolin-2-ol (320 mg, yield 68%). 1 H NMR (500MHz, DMSO) δ11.83 (s, 1H), 7.96 (s, 1H), 7.81 (d, J = 8.5Hz, 1H), 7.64 (d, J = 8.0Hz, 2H), 7.46- 7.43 (m, 2H), 7.42 - 7.36 (m, 2H), 7.28 (d, J = 7.5 Hz, 2H), 7.26 (d, J = 7.5 Hz, 2H), 2.35 (s, 3H), 2.34 (s) , 3H).
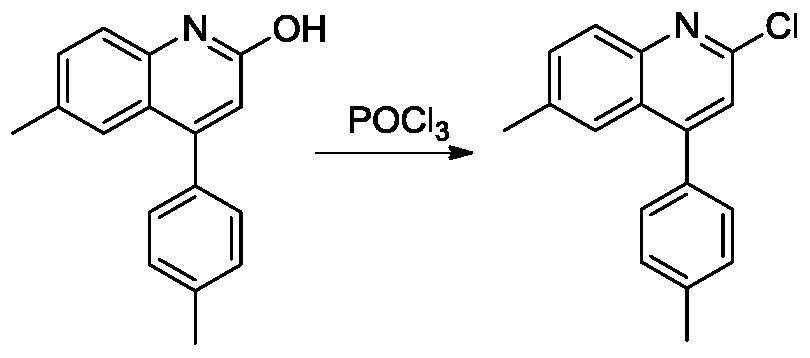
(3)2-氯-4-甲基-3-苯基-6-(对甲苯基)喹啉的合成。50ml二颈圆底烧瓶中加入4-甲基-3-苯基-6-(对甲苯基)喹啉-2-醇(320mg,1mmol),氮气置换反应瓶氛围,3mL三氯氧磷逐滴滴加入反应体系。将反应体系加热至100℃,搅拌18小时。TLC(石油醚:乙酸乙酯=1:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,水洗,空气中干燥得到淡黄色2-氯-4-甲基-3-苯基-6-(对甲苯基)喹啉(300mg,87%)。1H NMR(300MHz,DMSO) δ8.32(s,1H),8.16(d,J=8.7Hz,1H),8.06(d,J=8.7Hz,1H),7.80(d,J=8.1Hz,2H),7.58-7.49(m,3H),7.37-7.34(m,4H),2.49(s,3H)2.39(s,3H).(3) Synthesis of 2-chloro-4-methyl-3-phenyl-6-(p-tolyl)quinoline. 4-ml-3-phenyl-6-(p-tolyl)quinolin-2-ol (320 mg, 1 mmol) was added to a 50 ml two-necked round bottom flask, and the atmosphere of the reaction flask was replaced with nitrogen, and 3 mL of phosphorus oxychloride was dropped. The reaction system was added dropwise. The reaction system was heated to 100 ° C and stirred for 18 hours. TLC (petroleum ether: ethyl acetate = 1:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, washed with water, and dried in air to give pale yellow 2-chloro-4-methyl 3-Benzyl-6-(p-tolyl)quinoline (300 mg, 87%). 1 H NMR (300MHz, DMSO) δ8.32 (s, 1H), 8.16 (d, J = 8.7Hz, 1H), 8.06 (d, J = 8.7Hz, 1H), 7.80 (d, J = 8.1Hz, 2H), 7.58-7.49 (m, 3H), 7.37-7.34 (m, 4H), 2.49 (s, 3H) 2.39 (s, 3H).
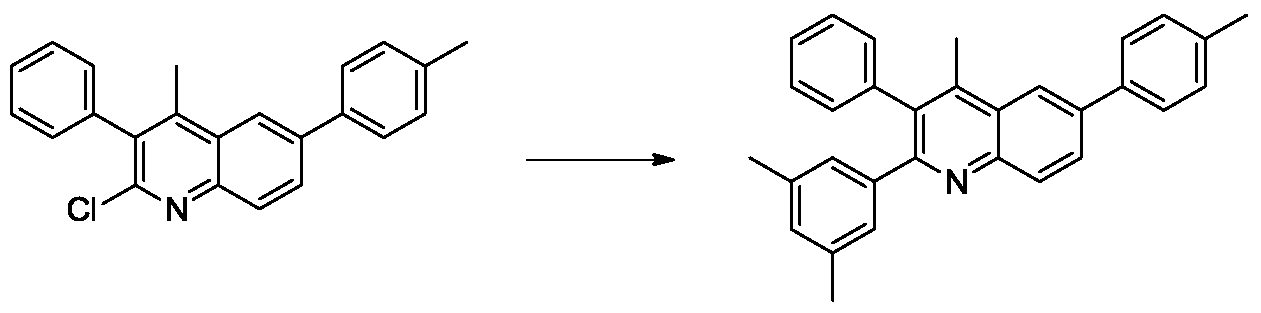
(4)2-(3,5-二甲基苯基)-4-甲基-3-苯基-6-(对-甲苯基)喹啉的合成。向100mL的schlenk管中加入2-氯-4-甲基-3-苯基-6-(对甲苯基)喹啉(300mg,0.87mmol),3,5-二甲苯硼酸(130.7mg,1.76mmol),三水磷酸钾(703mg,2.64mmol),2-双环己基膦-2',6'-二甲氧基联苯(28.9mg,0.07mmol),三(二亚苄基丙酮)二钯(16.1mg,0.0176mmol)和甲苯(10mL),氮气置换反应瓶氛围。将反应体系加热至100℃,搅拌12小时。TLC(石油醚:乙酸乙酯=10:1)显示反应完全,反应液用乙酸乙酯稀释,过滤,滤液旋干过柱(石油醚:乙酸乙酯=50:1)得白色2-(3,5-二甲基苯基)-4-甲基-3-苯基-6-(对-甲苯基)喹啉(300mg,70%)。1H NMR(300MHz,CDCl3)δ8.27-8.21(m,2H),8.00(dd,J=8.7,1.8Hz,1H),7.67(d,J=8.1Hz,2H),7.34–7.28(m,5H),7.14–7.12(m,2H),6.92(s,2H),6.82(s,1H),2.58(s,3H),2.44(s,3H),2.17(s,6H)(4) Synthesis of 2-(3,5-dimethylphenyl)-4-methyl-3-phenyl-6-(p-tolyl)quinoline. To a 100 mL schlenk tube was added 2-chloro-4-methyl-3-phenyl-6-(p-tolyl)quinoline (300 mg, 0.87 mmol), 3,5-dimethylbenzene boronic acid (130.7 mg, 1.76 mmol). Potassium phosphate trihydrate (703 mg, 2.64 mmol), 2-biscyclohexylphosphine-2',6'-dimethoxybiphenyl (28.9 mg, 0.07 mmol), tris(dibenzylideneacetone)dipalladium ( 16.1 mg, 0.0176 mmol) and toluene (10 mL) were replaced with nitrogen in a reaction flask atmosphere. The reaction system was heated to 100 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 10:1) showed that the reaction was completed, the reaction mixture was diluted with ethyl acetate, filtered, and the filtrate was evaporated to dryness ( petroleum ether: ethyl acetate = 50:1) ,5-Dimethylphenyl)-4-methyl-3-phenyl-6-(p-tolyl)quinoline (300 mg, 70%). 1 H NMR (300MHz, CDCl 3 ) δ8.27-8.21 (m, 2H), 8.00 (dd, J = 8.7,1.8Hz, 1H), 7.67 (d, J = 8.1Hz, 2H), 7.34-7.28 ( m,5H), 7.14–7.12 (m, 2H), 6.92 (s, 2H), 6.82 (s, 1H), 2.58 (s, 3H), 2.44 (s, 3H), 2.17 (s, 6H)
(5)二聚体11的合成。向15ml封管中加入2-(3,5-二甲基苯基)-4-甲基-3-苯基-6-(对-甲苯基)喹啉(82.6mg,0.2mmol),三水三氯化铱(14.1mg,0.04mmol),乙二醇乙醚(3mL)和水(1mL),氮气置换反应瓶氛围。将反应体系加热至100℃,搅拌12小时。将反应液冷至室温,固体析出,抽滤,空气中干燥得二聚体11(30mg,71%)。(5) Synthesis of dimer 11. To a 15 ml sealed tube was added 2-(3,5-dimethylphenyl)-4-methyl-3-phenyl-6-(p-tolyl)quinoline (82.6 mg, 0.2 mmol), tris Lanthanum trichloride (14.1 mg, 0.04 mmol), ethylene glycol diethyl ether (3 mL) and water (1 mL) were replaced with nitrogen atmosphere. The reaction system was heated to 100 ° C and stirred for 12 hours. The reaction solution was cooled to room temperature, and a solid was precipitated, suction filtered, and dried in air to give dimer 11 (30 mg, 71%).
(6)铱配合物11的合成:向15ml封管中加入二聚体11(100mg,0.047mmol),戊烷-2,4-二酮(0.1ml),碳酸钠(32mg,10mmol)和乙二醇乙醚(2mL),氮气置换反应瓶氛围,反应体系 加热回流12小时。将反应液冷至室温,固体析出,过滤,固体过柱(石油醚:乙酸乙酯=30:1)得到红色的铱配合物11(80mg,76%)。1H NMR(300MHz,CDCl3)δ8.06(d,J=1.5Hz,2H),7.93(d,J=9.0Hz,2H),7.71–7.68(m,5H),7.68-7.65(m,3H),7.59-7.57(m,5H),7.55-7.50(m,3H),7.28(d,J=9.0Hz,4H),6.39(d,J=4.8Hz,4H),4.32(s,1H),2.60(s,6H),2.41(s,6H),1.93(s,6H),1.42(s,6H),1.33(s,6H).MALDI-TOF MASS 1116.87[M+]Peak at 639nm,FWHM=62nm in DCM;Peak at 639nm,FWHM=58nm in PMMA。(6) Synthesis of ruthenium complex 11: Dimer 11 (100 mg, 0.047 mmol), pentane-2,4-dione (0.1 ml), sodium carbonate (32 mg, 10 mmol) and B were added to a 15 ml sealed tube. The diol ether (2 mL) was replaced with nitrogen in a reaction flask atmosphere, and the reaction system was heated to reflux for 12 hours. The reaction mixture was cooled to room temperature, and the solid was crystallised, filtered, and then evaporated to dryness (EtOAc (EtOAc:EtOAc) 1 H NMR (300MHz, CDCl 3 ) δ8.06 (d, J = 1.5Hz, 2H), 7.93 (d, J = 9.0Hz, 2H), 7.71-7.68 (m, 5H), 7.68-7.65 (m, 3H), 7.59-7.57 (m, 5H), 7.55-7.50 (m, 3H), 7.28 (d, J = 9.0 Hz, 4H), 6.39 (d, J = 4.8 Hz, 4H), 4.32 (s, 1H) ), 2.60 (s, 6H), 2.41 (s, 6H), 1.93 (s, 6H), 1.42 (s, 6H), 1.33 (s, 6H). MALDI-TOF MASS 1116.87 [M + ]Peak at 639nm, FWHM = 62 nm in DCM; Peak at 639 nm, FWHM = 58 nm in PMMA.
实施例2铱配合物126的制备Example 2 Preparation of ruthenium complex 126
Ra=甲基,Rb=对甲苯基,Rc=氢,Rd=甲基,Re=氢,Rf=甲基R a = methyl, R b = p-tolyl, R c = hydrogen, R d = methyl, R e = hydrogen, R f = methyl
(1)3-氧代-N,3-二-对-甲苯基丙酰胺的合成。向100ml封管加入对甲苯胺(2.7g,25mmol),3-氧-3-对甲苯基-丙酸乙酯(10.3g,50mmol)和二甲苯(20mL),反应体系N2鼓泡三分后,缓慢升温至145℃并在145℃条件下搅拌16小时。TLC(石油醚:乙酸乙酯=5:1)显示反应完全,将反应液冷至室温,产物析出,过滤,空气中干燥得白色3-氧代-N,3-二-对-甲苯基丙酰胺(3.4g,60%)。1H NMR(300MHz,CDCl3)δ9.21(s,1H),7.94(d,J=8.1Hz,2H),7.46(d,J=7.5Hz,2H),7.31(d,J=8.1Hz,2H),7.13(d,J=7.5Hz,2H),4.07(s,1H),4.04(br,1H),2.44(s,3H),2.32(s,3H).(1) Synthesis of 3-oxo-N,3-di-p-tolylpropionamide. To a 100 ml sealed tube was added p-toluidine (2.7 g, 25 mmol), 3-oxo-3-p-tolyl-propionic acid ethyl ester (10.3 g, 50 mmol) and xylene (20 mL), and the reaction system N 2 was bubbled three minutes. Thereafter, the temperature was slowly raised to 145 ° C and stirred at 145 ° C for 16 hours. TLC (petroleum ether: ethyl acetate = 5:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, product was precipitated, filtered, and dried in air to give white 3-oxo-N,3-di-p-tolylpropane Amide (3.4 g, 60%). 1 H NMR (300MHz, CDCl 3 ) δ 9.21 (s, 1H), 7.94 (d, J = 8.1 Hz, 2H), 7.46 (d, J = 7.5 Hz, 2H), 7.31 (d, J = 8.1 Hz) , 2H), 7.13 (d, J = 7.5 Hz, 2H), 4.07 (s, 1H), 4.04 (br, 1H), 2.44 (s, 3H), 2.32 (s, 3H).
(2)6-甲基-4-(对甲苯基)喹啉-2-醇的合成.向100mL三颈烧瓶中加入3-氧代-N,3-二-对-甲苯基丙酰胺(1g,2.4mmol),氮气置换反应瓶氛围,3mL浓硫酸逐滴滴加入反应体系。将反应体系加热至50℃,搅拌12小时。TLC(石油醚:乙酸乙酯=3:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,空气中干燥得到白色6-甲基-4-(对甲苯基)喹啉-2-醇(800mg,83%)。1H NMR(300MHz,DMSO)δ11.74(s,1H),7.37-7.36(m,5H),7.30(d,J=8.4Hz,1H),7.18(s,1H),6.32(s,1H),2.41(s,3H),2.26(s,3H). (2) Synthesis of 6-methyl-4-(p-tolyl)quinolin-2-ol. To a 100 mL three-necked flask was added 3-oxo-N,3-di-p-tolylpropanamide (1 g , 2.4 mmol), the atmosphere of the reaction flask was replaced with nitrogen, and 3 mL of concentrated sulfuric acid was added dropwise to the reaction system. The reaction system was heated to 50 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 3:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, and dried in air to give white 6-methyl-4-(p-toluene) Quinoline-2-ol (800 mg, 83%). 1 H NMR (300MHz, DMSO) δ11.74 (s, 1H), 7.37-7.36 (m, 5H), 7.30 (d, J = 8.4Hz, 1H), 7.18 (s, 1H), 6.32 (s, 1H ), 2.41 (s, 3H), 2.26 (s, 3H).
(3)2-氯-6-甲基-4-(对甲苯基)喹啉的合成。向50ml二颈圆底烧瓶中加入6-甲基-4-(对甲苯基)喹啉-2-醇(499mg,2mmol),氮气置换反应瓶氛围,3.5mL三氯氧磷逐滴滴加入反应体系。将反应体系加热至100℃,搅拌18小时。TLC(石油醚:乙酸乙酯=6:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,水洗,空气中干燥得到白色的2-氯-6-甲基-4-(对甲苯基)喹啉(500mg,87%)。1H NMR(500MHz,DMSO)7.93(d,J=8.5Hz,1H),7.69(d,J=8.5Hz,1H),7.63(s,1H),7.46(d,J=8.0Hz,2H),7.42–7.40(m,3H),2.44(s,3H),2.43(s,3H).(3) Synthesis of 2-chloro-6-methyl-4-(p-tolyl)quinoline. To a 50 ml two-necked round bottom flask was added 6-methyl-4-(p-tolyl)quinolin-2-ol (499 mg, 2 mmol), the atmosphere of the reaction flask was replaced with nitrogen, and 3.5 mL of phosphorus oxychloride was added dropwise. system. The reaction system was heated to 100 ° C and stirred for 18 hours. TLC (petroleum ether: ethyl acetate = 6:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, washed with water and dried in air to give white 2-chloro-6- Base-4-(p-tolyl)quinoline (500 mg, 87%). 1 H NMR (500MHz, DMSO) 7.93 (d, J = 8.5Hz, 1H), 7.69 (d, J = 8.5Hz, 1H), 7.63 (s, 1H), 7.46 (d, J = 8.0Hz, 2H) , 7.42–7.40 (m, 3H), 2.44 (s, 3H), 2.43 (s, 3H).
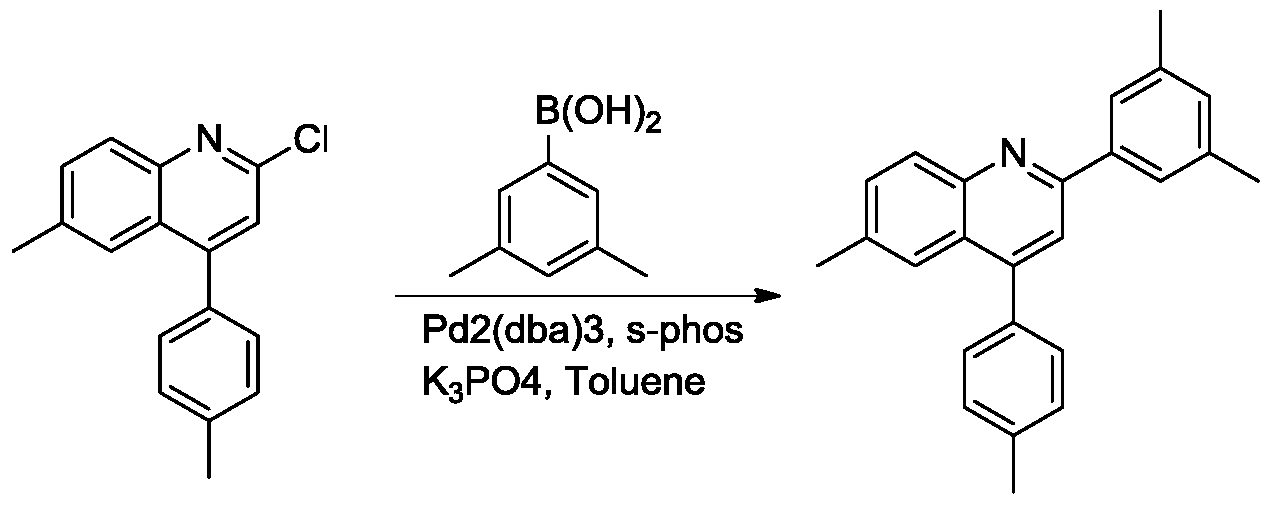
(4)2-(3,5-二甲基苯基)-6-甲基-4-(对甲苯基)喹啉的合成。向100ml圆底烧瓶中加入2-氯-6-甲基-4-(对甲苯基)喹啉(1g,3.74mmmol)3,5-二甲苯硼酸(1.12g,7.48mmol),三水磷酸钾(g,14.96mmol),2-双环己基膦-2',6'-二甲氧基联苯(mg,0.30mmol),三(二亚苄基丙酮)二钯(mg,0.075mmol)和甲苯(10mL),氮气置换反应瓶氛围。将反应体系加热至100℃,搅拌12小时。TLC(石油醚:乙酸乙酯=10:1)显示反应完全,反应液用乙酸乙酯稀释,过滤,滤液旋干过柱(石油醚:乙酸乙酯=50:1)得白色2-(3,5-二甲基苯基)-6-甲基-4-(对甲苯基)喹啉(1.0g,79%)。1H NMR(300MHz,CDCl3)δ8.14(d,J=8.4Hz,1H),7.78(s,2H),7.75(s,1H),7.67(s,1H),7.56(d,J=8.4Hz,1H),7.47(d,J=7.8Hz,2H),7.37(d,J=7.8Hz,2H),7.10(s,1H),2.50(s,3H),2.48(s,3H),2.43(s,6H).(4) Synthesis of 2-(3,5-dimethylphenyl)-6-methyl-4-(p-tolyl)quinoline. To a 100 ml round bottom flask was added 2-chloro-6-methyl-4-(p-tolyl)quinoline (1 g, 3.74 mmol) 3,5-xylene boronic acid (1.12 g, 7.48 mmol), potassium phosphate trihydrate (g, 14.96 mmol), 2-biscyclohexylphosphine-2',6'-dimethoxybiphenyl (mg, 0.30 mmol), tris(dibenzylideneacetone)dipalladium (mg, 0.075 mmol) and toluene (10 mL), the atmosphere of the reaction flask was replaced with nitrogen. The reaction system was heated to 100 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 10:1) showed that the reaction was completed, the reaction mixture was diluted with ethyl acetate, filtered, and the filtrate was evaporated to dryness ( petroleum ether: ethyl acetate = 50:1) , 5-dimethylphenyl)-6-methyl-4-(p-tolyl)quinoline (1.0 g, 79%). 1 H NMR (300MHz, CDCl 3 ) δ8.14 (d, J = 8.4Hz, 1H), 7.78 (s, 2H), 7.75 (s, 1H), 7.67 (s, 1H), 7.56 (d, J = 8.4 Hz, 1H), 7.47 (d, J = 7.8 Hz, 2H), 7.37 (d, J = 7.8 Hz, 2H), 7.10 (s, 1H), 2.50 (s, 3H), 2.48 (s, 3H) , 2.43 (s, 6H).
(5)二聚体126的合成。向15ml封管中加入2-(3,5-二甲基苯基)-6-甲基-4-(对甲苯基)喹啉(64.4mg,0.2mmol),三水三氯化铱(14.1mg,0.04mmol),乙二醇乙醚(3mL)和水(1mL),氮气鼓泡三分钟,将反应体系加热至120℃,搅拌12小时。将反应液冷至室温,固体析出,过滤,空气中干燥得到二聚体126(25mg,76%)。(5) Synthesis of dimer 126. To a 15 ml sealed tube was added 2-(3,5-dimethylphenyl)-6-methyl-4-(p-tolyl)quinoline (64.4 mg, 0.2 mmol), ruthenium trichloride trihydrate (14.1 Mg, 0.04 mmol), ethylene glycol diethyl ether (3 mL) and water (1 mL), nitrogen was bubbled for three minutes, and the reaction was heated to 120 ° C and stirred for 12 hours. The reaction solution was cooled to room temperature, and a solid was precipitated, filtered, and dried in air to give dimer 126 (25 mg, 76%).
(6)铱配合物126的合成。向75ml封管中加入二聚体126(166.2mg,0.1mmol),戊烷-2,4-二酮(40mg,0.4mmol),碳酸钠(53mg,0.5mmol)和乙二醇乙醚(3mL),氮气鼓泡三分钟,反应体系加热回流12小时。将反应液冷至室温,固体析出,过滤,乙醚洗涤,空气中干燥得铱配合物126。1H NMR(300MHz,DMSO)δ7.90(d,J=9.0Hz,4H),7.58(s,2H),7.57–7.52(m,6H),7.41(d,J=7.8Hz,4H),7.14(d,J=9.0Hz,2H),6.53(s,2H),4.37(s,1H),2.52(s,6H),2.34(s,12H),1.37(s,6H),1.34(s,6H).MS(MALDI-TOF):964.4[M]+Emission peak in DCM at 626nm,FWHM=72nm,peak in PMMA at 625nm,FWHM=64nm。(6) Synthesis of ruthenium complex 126. Dimer 126 (166.2 mg, 0.1 mmol), pentane-2,4-dione (40 mg, 0.4 mmol), sodium carbonate (53 mg, 0.5 mmol) and ethyl acetate (3 mL) Nitrogen gas was bubbled for three minutes, and the reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and the solid was precipitated, filtered, washed with diethyl ether, and dried in air to give ruthenium complex 126. 1 H NMR (300MHz, DMSO) δ7.90 (d, J = 9.0Hz, 4H), 7.58 (s, 2H), 7.57-7.52 (m, 6H), 7.41 (d, J = 7.8Hz, 4H), 7.14 (d, J = 9.0 Hz, 2H), 6.53 (s, 2H), 4.37 (s, 1H), 2.52 (s, 6H), 2.34 (s, 12H), 1.37 (s, 6H), 1.34 (s) , 6H). MS (MALDI-TOF): 964.4 [M] + Emission peak in DCM at 626 nm, FWHM = 72 nm, peak in PMMA at 625 nm, FWHM = 64 nm.
实施例3铱配合物201的制备方法Example 3 Preparation Method of Ruthenium Complex 201
Ra=甲基,Rb=对甲苯基,Rc=氢,Rd=氢,Re=甲基,Rf=甲基R a = methyl, R b = p-tolyl, R c = hydrogen, R d = hydrogen, R e = methyl, R f = methyl
(1)二聚体201的合成。向15ml封管中加入6-甲基-2,4-二-对-甲苯基喹啉(61.6mg,0.2mmol),三水三氯化铱(14.1mg,0.04mmol),乙二醇乙醚(3mL)和水(1mL),氮气鼓泡三分钟,将反应体系加热回流12小时。将反应液冷至室温,固体析出,过滤,空气中干燥得到二聚体201(25mg,76%),且直接用于下一步。(1) Synthesis of dimer 201. To a 15 ml sealed tube was added 6-methyl-2,4-di-p-tolylquinoline (61.6 mg, 0.2 mmol), ruthenium trichloride trihydrate (14.1 mg, 0.04 mmol), ethylene glycol ether ( 3 mL) and water (1 mL) were bubbled with nitrogen for three minutes, and the reaction was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and the solid was crystallised, filtered, and dried in vacuo to give dimer 201 (25 mg, 76%).
(2)铱配合物201的合成。向75ml封管中加入二聚体(160.6mg,0.1mmol),戊烷-2,4-二酮(40mg,0.4mmol),碳酸钠(53mg,0.5mmol)和乙二醇乙醚(3mL),氮气鼓泡三分钟,反应 体系加热回流12小时。将反应液冷至室温,固体析出,过滤,乙醚洗涤,空气中干燥得铱配合物201(132mg,71%)。1H NMR(300MHz,DMSO)δ8.32(d,J=8.7Hz,2H),8.11(s,2H),7.90(d,J=8.1Hz,2H),7.65(d,J=7.8Hz,4H),7.61(s,2H),7.49(d,J=8.1Hz,4H),7.37(dd,J=8.7,1.5Hz,2H),6.69(d,J=8.1Hz,2H),6.20(s,2H),4.71(s,1H),2.51(s,6H),2.39(s,6H),1.90(s,6H),1.47(s,6H).MS MALDI-TOF 836.1[M-acac]+Peak in DCM solution at RT at 611nm,FWHM 83nm,Peak in PMMA solution at RT at 609nm,FWHM 79nm.(2) Synthesis of ruthenium complex 201. To a 75 ml sealed tube was added a dimer (160.6 mg, 0.1 mmol), pentane-2,4-dione (40 mg, 0.4 mmol), sodium carbonate (53 mg, 0.5 mmol) and ethyl ether (3 mL). Nitrogen gas was bubbled for three minutes, and the reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and the solid was crystallised, filtered, washed with diethyl ether and dried in vacuo to afford EtOAc (201 mg, 71%). 1 H NMR (300MHz, DMSO) δ8.32 (d, J = 8.7Hz, 2H), 8.11 (s, 2H), 7.90 (d, J = 8.1Hz, 2H), 7.65 (d, J = 7.8Hz, 4H), 7.61 (s, 2H), 7.49 (d, J = 8.1 Hz, 4H), 7.37 (dd, J = 8.7, 1.5 Hz, 2H), 6.69 (d, J = 8.1 Hz, 2H), 6.20 ( s, 2H), 4.71 (s, 1H), 2.51 (s, 6H), 2.39 (s, 6H), 1.90 (s, 6H), 1.47 (s, 6H). MS MALDI-TOF 836.1 [M-acac] + Peak in DCM solution at RT at 611nm, FWHM 83nm, Peak in PMMA solution at RT at 609nm, FWHM 79nm.
实施例4铱配合物187的制备Example 4 Preparation of ruthenium complex 187
Ra=甲基,Rb=异丁基,Rc=甲基,Rd=甲基,Re=氢,Rf=异丁基R a = methyl, R b = isobutyl, R c = methyl, R d = methyl, R e = hydrogen, R f = isobutyl
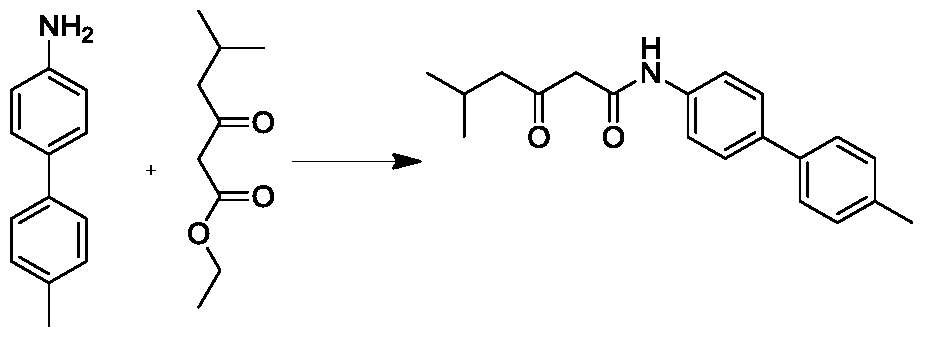
(1)2,5-二甲基-3-氧代-N-(对甲苯基)己酰胺的合成。向100ml封管加入对甲苯胺(107.12mg,1mmol),2,5-二甲基-3-氧代-己酸乙酯(346.4mg,2mmol)和二甲苯(2mL),反应体系N2鼓泡三分后,缓慢升温至145℃,并在145℃条件下搅拌16小时。TLC(石油醚:乙酸乙酯=5:1)显示反应完全,将反应液冷至室温,产物析出,过滤,空气中干燥得白色2,5-二甲基-3-氧代-N-(对甲苯基)己酰胺(120mg,40%)。1H NMR(300MHz,CDCl3)δ8.22(s,1H),7.39(d,J=7.8Hz,2H),7.12(d,J=7.8Hz,2H),3.54(q,J=7.2Hz,1H),2.48(d,J=6.9Hz,2H),2.31(s,3H),2.22–2.14(m,1H),1.49(d,J=6.9Hz,3H),0.94–0.91(m,6H).(1) Synthesis of 2,5-dimethyl-3-oxo-N-(p-tolyl)hexanamide. Sealed tube was added to 100ml toluidine (107.12mg, 1mmol), 2,5- dimethyl-3-oxo - hexanoic acid ethyl ester (346.4mg, 2mmol) and xylene (2mL), the reaction system drum N 2 After soaking for three minutes, the temperature was slowly raised to 145 ° C and stirred at 145 ° C for 16 hours. TLC (petroleum ether: ethyl acetate = 5:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, product was precipitated, filtered, and dried in air to give white 2,5-dimethyl-3-oxo-N- ( p-Tolyl) hexanamide (120 mg, 40%). 1 H NMR (300MHz, CDCl 3 ) δ 8.22 (s, 1H), 7.39 (d, J = 7.8 Hz, 2H), 7.12 (d, J = 7.8 Hz, 2H), 3.54 (q, J = 7.2 Hz) , 1H), 2.48 (d, J = 6.9 Hz, 2H), 2.31 (s, 3H), 2.22 - 2.14 (m, 1H), 1.49 (d, J = 6.9 Hz, 3H), 0.94 - 0.91 (m, 6H).
(2)4-异丁基-3,6-二甲基喹-2-醇的合成。向100mL三颈烧瓶中加入2,5-二甲基-3-氧代-N-(对甲苯基)己酰胺(1g,4mmol),氮气置换反应瓶氛围,3.5mL浓硫酸逐滴滴加入反应体系。将反应体系加热至50℃,搅拌12小时。TLC(石油醚:乙酸乙酯=3:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,空气中干燥得到淡黄色4-异丁基-3,6-二甲基喹-2-醇(830mg,90%)。1H NMR(300MHz,CDCl3)δ11.55(s,1H),7.50(s,1H),7.25-7.17(m,2H),2.78(d,J=7.2Hz,2H),2.36(s,3H),2.11(s,3H),1.98-1.89(m,1H),0.95(d,J=6.6Hz,6H)。 (2) Synthesis of 4-isobutyl-3,6-dimethylquin-2-ol. 2,5-Dimethyl-3-oxo-N-(p-tolyl)hexanamide (1 g, 4 mmol) was added to a 100 mL three-necked flask, and the atmosphere of the reaction flask was replaced with nitrogen, and 3.5 mL of concentrated sulfuric acid was added dropwise to the reaction. system. The reaction system was heated to 50 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 3:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, and dried in air to give pale yellow 4-isobutyl-3,6 - dimethylquin-2-ol (830 mg, 90%). 1 H NMR (300MHz, CDCl 3 ) δ11.55 (s, 1H), 7.50 (s, 1H), 7.25-7.17 (m, 2H), 2.78 (d, J = 7.2Hz, 2H), 2.36 (s, 3H), 2.11 (s, 3H), 1.98-1.89 (m, 1H), 0.95 (d, J = 6.6 Hz, 6H).
(3)2-氯-4-异丁基-3,6-二甲基喹啉的合成。向50ml二颈圆底烧瓶中加入4-异丁基-3,6-二甲基喹-2-醇(800mg,3.5mmol),氮气置换反应瓶氛围,3.5mL三氯氧磷逐滴滴加入反应体系。将反应体系加热至100℃,搅拌18小时。TLC(石油醚:乙酸乙酯=5:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,水洗,空气中干燥得到淡黄色2-氯-4-异丁基-3,6-二甲基喹啉(830mg,93%)。1H NMR(300MHz,DMSO)δ7.89(s,1H),7.79(d,J=8.4Hz,1H),7.56(d,J=8.4Hz,1H),3.05(d,J=7.2Hz,2H),2.53(s,3H),2.49(s,3H),2.02-1.93(m,1H),0.94(d,J=6.6Hz,6H).(3) Synthesis of 2-chloro-4-isobutyl-3,6-dimethylquinoline. To a 50 ml two-necked round bottom flask was added 4-isobutyl-3,6-dimethylquin-2-ol (800 mg, 3.5 mmol), the atmosphere of the reaction flask was replaced with nitrogen, and 3.5 mL of phosphorus oxychloride was added dropwise. reaction system. The reaction system was heated to 100 ° C and stirred for 18 hours. TLC (petroleum ether: ethyl acetate = 5:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, washed with water, and dried in air to give pale yellow 2-chloro-4-iso Butyl-3,6-dimethylquinoline (830 mg, 93%). 1 H NMR (300MHz, DMSO) δ7.89 (s, 1H), 7.79 (d, J = 8.4Hz, 1H), 7.56 (d, J = 8.4Hz, 1H), 3.05 (d, J = 7.2Hz, 2H), 2.53 (s, 3H), 2.49 (s, 3H), 2.02-1.93 (m, 1H), 0.94 (d, J = 6.6 Hz, 6H).
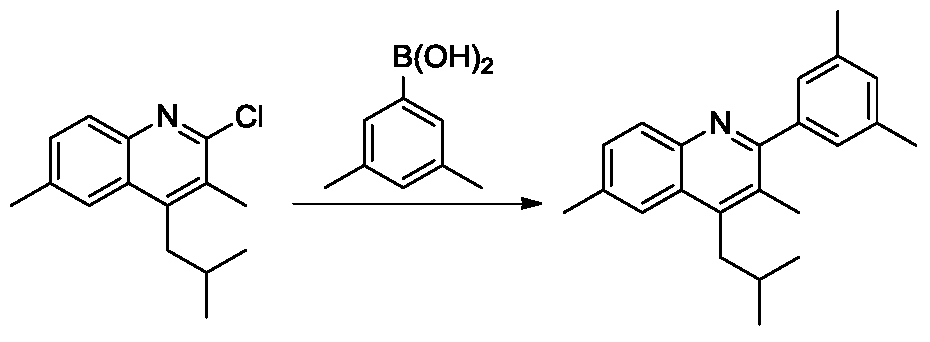
(4)2-(3,5-二甲基苯基)-4-异丁基-3,6-二甲基喹啉的合成。向100mL的schlenk管中加入2-氯-4-异丁基-3,6-二甲基喹啉(800mg,3.23mmol),3,5-二甲苯硼酸(969.6mg,6.46mmol),三水磷酸钾(4.3g,16.15mmol),2-双环己基膦-2',6'-二甲氧基联苯(106.1mg,0.26mmol),三(二亚苄基丙酮)二钯(59mg,0.065mmol)和甲苯(10mL),氮气置换反应瓶氛围。将反应体系加热至100℃,搅拌12小时。TLC(石油醚:乙酸乙酯=10:1)显示反应完全,反应液用乙酸乙酯稀释,过滤,滤液旋干过柱(石油醚:乙酸乙酯=10:1)得白色2-(3,5-二甲基苯基)-4-异丁基-3,6-二甲基喹啉(700mg,55%)。1H NMR(500MHz,CDCl3)δ8.02(s,1H),7.76(s,1H),7.46(d,J=8.0Hz,1H),7.10(s,2H),7.05(s,1H),3.05(d,J=7.5Hz,2H),2.57(s,3H),2.38(s,6H),2.37(s,3H),2.15–2.13(m,1H),1.04(d,J=6.5Hz,6H).(4) Synthesis of 2-(3,5-dimethylphenyl)-4-isobutyl-3,6-dimethylquinoline. To a 100 mL schlenk tube was added 2-chloro-4-isobutyl-3,6-dimethylquinoline (800 mg, 3.23 mmol), 3,5-dimethylbenzene boronic acid (969.6 mg, 6.46 mmol), tris Potassium phosphate (4.3 g, 16.15 mmol), 2-biscyclohexylphosphine-2',6'-dimethoxybiphenyl (106.1 mg, 0.26 mmol), tris(dibenzylideneacetone)dipalladium (59 mg, 0.065 Methyl) and toluene (10 mL) were replaced with nitrogen in a reaction flask atmosphere. The reaction system was heated to 100 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 10:1) showed that the reaction was completed, the reaction mixture was diluted with ethyl acetate, filtered, and the filtrate was evaporated to dryness ( petroleum ether: ethyl acetate = 10:1) , 5-dimethylphenyl)-4-isobutyl-3,6-dimethylquinoline (700 mg, 55%). 1 H NMR (500 MHz, CDCl 3 ) δ 8.02 (s, 1H), 7.76 (s, 1H), 7.46 (d, J = 8.0 Hz, 1H), 7.10 (s, 2H), 7.05 (s, 1H) , 3.05 (d, J = 7.5 Hz, 2H), 2.57 (s, 3H), 2.38 (s, 6H), 2.37 (s, 3H), 2.15 - 2.13 (m, 1H), 1.04 (d, J = 6.5 Hz, 6H).
(5)二聚体187的合成。向15ml封管中加入2-(3,5-二甲基苯基)-4-异丁基-3,6-二甲基喹 啉(65.4mg,0.2mmol),三水三氯化铱(14.1mg,0.04mmol),乙二醇乙醚(3mL)和水(1mL),氮气置换反应瓶氛围,将反应体系加热回流12小时。将反应液冷至室温,固体析出,过滤,空气中干燥得二聚体187(30mg,87%)。(5) Synthesis of dimer 187. Add 2-(3,5-dimethylphenyl)-4-isobutyl-3,6-dimethylquina to a 15 ml sealed tube The ruthenium (65.4 mg, 0.2 mmol), ruthenium trichloride trihydrate (14.1 mg, 0.04 mmol), ethylene glycol diethyl ether (3 mL) and water (1 mL) were placed under nitrogen atmosphere, and the reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and a solid was precipitated, filtered, and dried in air to give dimer 187 (30 mg, 87%).
(6)铱配合物187的合成。向15ml封管中加入二聚体187(250mg),2,8-二甲基壬烷-4,6-二酮(0.1ml),碳酸钠(32mg,10mmol)和乙二醇乙醚(2mL),氮气置换反应瓶氛围,反应体系加热回流12小时。将反应液冷至室温,溶液旋干过柱(石油醚:乙酸乙酯=50:1)得到红色的铱配合物187。1H NMR(500MHz,CDCl3)δ7.74(d,J=8.5Hz,2H),7.62(s,2H),7.56(s,2H),7.02(d,J=8.5Hz,2H),6.52(s,2H),4.08(s,1H),3.16–3.06(m,4H),3.01(s,6H),2.96-2.92(m,2H),2.41(s,6H),2.35(s,6H),2.23–2.20(m,2H),1.57-1.55(m,2H),1.51–1.49(m,2H),1.24(s,6H),1.10(d,J=6.5Hz,6H),1.08(d,J=6.5Hz,6H),0.47(d,J=6.5Hz,6H),0.23(d,J=6.5Hz,6H).MS(MALDI-TOF):1007.9[M-1]+Peak in DCM at room temperature at 617nm,FWHM=68nm.Peak in PMMA at room temperature at 618nm,FWHM=68nm。(6) Synthesis of ruthenium complex 187. Dimer 187 (250 mg), 2,8-dimethyldecane-4,6-dione (0.1 ml), sodium carbonate (32 mg, 10 mmol) and ethylene glycol diethyl ether (2 mL) were added to a 15 ml sealed tube. The atmosphere of the reaction flask was replaced with nitrogen, and the reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and the solution was evaporated to dryness (yield: petroleum ether: ethyl acetate = 50:1). 1 H NMR (500MHz, CDCl 3 ) δ7.74 (d, J = 8.5Hz, 2H), 7.62 (s, 2H), 7.56 (s, 2H), 7.02 (d, J = 8.5Hz, 2H), 6.52 (s, 2H), 4.08 (s, 1H), 3.16–3.06 (m, 4H), 3.01 (s, 6H), 2.96-2.92 (m, 2H), 2.41 (s, 6H), 2.35 (s, 6H) ), 2.23–2.20 (m, 2H), 1.57-1.55 (m, 2H), 1.51–1.49 (m, 2H), 1.24 (s, 6H), 1.10 (d, J = 6.5 Hz, 6H), 1.08 ( d, J = 6.5 Hz, 6H), 0.47 (d, J = 6.5 Hz, 6H), 0.23 (d, J = 6.5 Hz, 6H). MS (MALDI-TOF): 1007.9 [M-1] + Peak in DCM at room temperature at 617 nm, FWHM = 68 nm. Peak in PMMA at room temperature at 618 nm, FWHM = 68 nm.
实施例5铱配合物82的合成Example 5 Synthesis of Complex 82
Ra=对甲苯基,Rb=异丁基,Rc=氢,Rd=氢,Re=甲基,Rf=异丁基R a = p-tolyl, R b = isobutyl, R c = hydrogen, R d = hydrogen, R e = methyl, R f = isobutyl
(1)5-甲基-N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代己酰胺的合成。1000mL的三颈圆底烧瓶中加入4'-甲基-[1,1'-联苯]-4-胺(52.6g,287mmol),5-甲基-3-氧代己酸乙酯(59.3g,344mmol)和二甲苯(450mL),反应体系N2鼓泡三分后,缓慢升温至145℃,并在145℃条件下搅拌3-4小时。TLC(石油醚:乙酸乙酯=5:1)显示反应完全,将反应液冷至室温,产物析出,过滤,空气中干燥得淡黄色5-甲基-N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代己酰胺(46.1g,52%)。1H NMR(300MHz,DMSO)δ10.13(s,1H),7.66-7.58(m,4H),7.53(d,J=8.1Hz,2H),7.25(d,J=8.1Hz,2H),3.53(s,2H),2.46(d,J=6.9Hz,2H),2.33(s,3H),2.11-2.02(m,1H),0.89(d,J=6.6Hz,6H). (1) Synthesis of 5-methyl-N-(4'-methyl-[1,1'-biphenyl]-4-yl)-3-oxohexanamide. Add a 4'-methyl-[1,1'-biphenyl]-4-amine (52.6 g, 287 mmol), ethyl 5-methyl-3-oxohexanoate (59.3) to a 1000 mL 3-neck round bottom flask. g, 344 mmol) and xylene (450 mL), after the reaction system N 2 was bubbled for three minutes, the temperature was slowly raised to 145 ° C, and stirred at 145 ° C for 3-4 hours. TLC (petroleum ether: ethyl acetate = 5:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, product was precipitated, filtered, and dried in air to give pale yellow 5-methyl-N-(4'-methyl-[ 1,1'-Biphenyl]-4-yl)-3-oxohexanamide (46.1 g, 52%). 1 H NMR (300MHz, DMSO) δ10.13 (s, 1H), 7.66-7.58 (m, 4H), 7.53 (d, J = 8.1Hz, 2H), 7.25 (d, J = 8.1Hz, 2H), 3.53 (s, 2H), 2.46 (d, J = 6.9 Hz, 2H), 2.33 (s, 3H), 2.11-2.02 (m, 1H), 0.89 (d, J = 6.6 Hz, 6H).
(2)4-异丁基-6-(对甲苯基)喹啉-2-醇的合成。向250ml三颈圆底烧瓶中加入5-甲基-N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代己酰胺(18.3g,59.1mmol),氮气置换反应氛围,50mL浓硫酸逐滴滴加入反应体系,将反应体系加热至40-50℃,搅拌12小时。TLC(石油醚:乙酸乙酯=3:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,水洗,空气中干燥得白色4-异丁基-6-(对甲苯基)喹啉-2-醇(15.5g,95%)。1H NMR(500MHz,CDCl3)δ9.16(s,0.8H,enol),7.60(d,J=8.5Hz,2H),7.54(d,J=8.5Hz,2H),7.46(d,J=8.0Hz,2H),7.23(d,J=8.0Hz,2H),3.56(s,2H),2.47(d,J=6.8Hz,2H),2.38(s,3H),2.20(th,J=6.8,6.6Hz;1H),2.06(s,0.2H),0.97(d,J=6.6Hz,6H).(2) Synthesis of 4-isobutyl-6-(p-tolyl)quinolin-2-ol. To a 250 ml 3-neck round bottom flask was charged 5-methyl-N-(4'-methyl-[1,1'-biphenyl]-4-yl)-3-oxohexanamide (18.3 g, 59.1 Methyl), the reaction atmosphere was replaced by nitrogen, 50 mL of concentrated sulfuric acid was added dropwise to the reaction system, and the reaction system was heated to 40-50 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 3:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, washed with water and dried in air to give white 4-isobutyl-6- (p-Tolyl)quinolin-2-ol (15.5 g, 95%). 1 H NMR (500MHz, CDCl 3 ) δ9.16 (s, 0.8H, enol), 7.60 (d, J = 8.5Hz, 2H), 7.54 (d, J = 8.5Hz, 2H), 7.46 (d, J = 8.0 Hz, 2H), 7.23 (d, J = 8.0 Hz, 2H), 3.56 (s, 2H), 2.47 (d, J = 6.8 Hz, 2H), 2.38 (s, 3H), 2.20 (th, J) = 6.8, 6.6 Hz; 1H), 2.06 (s, 0.2 H), 0.97 (d, J = 6.6 Hz, 6H).
(3)2-氯-4-异丁基-6-(对甲苯基)喹啉的合成。向500ml三颈圆底烧瓶中加入4-异丁基-6-(对甲苯基)喹啉-2-醇(1g,3.6mmol),氮气置换反应氛围,3mL三氯氧磷逐滴滴加入反应体系,将反应体系缓慢升温至100℃,搅拌18小时。TLC(石油醚:乙酸乙酯=6:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,水洗,空气中干燥得深黄色2-氯-4-异丁基-6-(对甲苯基)喹啉(1g,94%)。1H NMR(400MHz,CDCl3)δ8.09(d,J=2.0,1H),8.06(d,J=5.3Hz,1H),7.94(dd,J=10.2,2.0Hz,1H),7.58(d,J=8.1Hz,2H),7.33(d,J=8.1Hz,2H),7.22(s,1H),2.94(d,J=7.3Hz,2H),2.44(s,3H),2.35–2.05(m,1H),1.02(d,J=6.6Hz,6H).(3) Synthesis of 2-chloro-4-isobutyl-6-(p-tolyl)quinoline. To a 500 ml three-necked round bottom flask was added 4-isobutyl-6-(p-tolyl)quinolin-2-ol (1 g, 3.6 mmol), the reaction atmosphere was replaced with nitrogen, and 3 mL of phosphorus oxychloride was added dropwise. The system was slowly warmed to 100 ° C and stirred for 18 hours. TLC (petroleum ether: ethyl acetate = 6:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, washed with water, and dried in air to obtain a dark yellow 2-chloro-4-iso Butyl-6-(p-tolyl)quinoline (1 g, 94%). 1 H NMR (400 MHz, CDCl 3 ) δ 8.09 (d, J = 2.0, 1H), 8.06 (d, J = 5.3 Hz, 1H), 7.94 (dd, J = 10.2, 2.0 Hz, 1H), 7.58 ( d, J = 8.1 Hz, 2H), 7.33 (d, J = 8.1 Hz, 2H), 7.22 (s, 1H), 2.94 (d, J = 7.3 Hz, 2H), 2.44 (s, 3H), 2.35 - 2.05 (m, 1H), 1.02 (d, J = 6.6 Hz, 6H).
(4)2-(4-甲基苯基)-4-异丁基-6-(对甲苯基)喹啉的合成.向500ml圆底烧瓶中加入2-氯-4-异丁基-6-(对甲苯基)喹啉(4.8g,15.5mmol),对甲基苯硼酸(4.2g g,31mmol),三水磷酸钾(20.6g,77.5mmol),2-双环己基膦-2',6'-二甲氧基联苯(509mg,1.24mmol),三(二亚苄基丙酮)二钯(284mg,0.31mmol)和甲苯(110mL),氮气置换反应氛围,将反应液加热回流12小时。TLC(石油醚:乙酸乙酯=10:1)显示反应完全,反应液用乙酸乙酯稀释,过滤,滤液旋干过柱(石油醚:乙酸乙酯=10:1)得白色2-(4-甲基苯基)-4-异丁基-6-(对甲苯基)喹啉(4g,71%)。(4) Synthesis of 2-(4-methylphenyl)-4-isobutyl-6-(p-tolyl)quinoline. Add 2-chloro-4-isobutyl-6 to a 500 ml round bottom flask -(p-tolyl)quinoline (4.8 g, 15.5 mmol), p-methylphenylboronic acid (4.2 g, 31 mmol), potassium phosphate trihydrate (20.6 g, 77.5 mmol), 2-dicyclohexylphosphine-2', 6'-dimethoxybiphenyl (509 mg, 1.24 mmol), tris(dibenzylideneacetone)dipalladium (284 mg, 0.31 mmol) and toluene (110 mL) were replaced with nitrogen, and the reaction mixture was heated to reflux for 12 hours. . TLC (petroleum ether: ethyl acetate = 10:1) showed that the reaction was completed, the reaction mixture was diluted with ethyl acetate, filtered, and the filtrate was evaporated to dryness ( petroleum ether: ethyl acetate = 10:1) Methylphenyl)-4-isobutyl-6-(p-tolyl)quinoline (4 g, 71%).
(5)二聚体82的合成.向100ml圆底烧瓶中加入2-(4-甲基苯基)-4-异丁基-6-(对甲苯基)喹啉(1.1g,3mmol),三水三氯化铱(195mg,0.6mmol),乙二醇乙醚(15mL)和水(5mL),氮气置换反应瓶氛围。将反应体系加热回流12小时。将反应液冷至室温,固体析出,过滤,乙醚(8mL×3)洗涤,空气中干燥得二聚体82(500mg,87%)。(5) Synthesis of dimer 82. To a 100 ml round bottom flask was charged 2-(4-methylphenyl)-4-isobutyl-6-(p-tolyl)quinoline (1.1 g, 3 mmol). Trihydrate ruthenium trichloride (195 mg, 0.6 mmol), ethylene glycol ether (15 mL) and water (5 mL) were replaced with a nitrogen atmosphere. The reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and a solid was precipitated, which was filtered, washed with diethyl ether (8 mL × 3), and dried in air to obtain a dimer 82 (500 mg, 87%).
(6)铱配合物82的合成。向100ml圆底烧瓶中加入二聚体(650mg,0.34mmol),2,8-二甲基壬烷-4,6-二酮(304mg,1.65mmol),碳酸钠(350mg,3.3mmol)和乙二醇乙醚(20mL),氮气置换反应瓶氛围,反应体系加热回流12小时。将反应液冷至室温,旋干过柱(石油醚:二氯甲烷=1:1)得到红色的铱配合物82(740mg,99%)。1H NMR(400MHz,CDCl3)δ8.49(d,J=9.1Hz,2H),8.05(d,J=2.0Hz,2H),7.84(s,2H),7.73(d,J=8.1Hz,2H),7.58(dd,9.1,2Hz,2H),7.57(dd,8.0Hz,4H),7.29(d,J=8.0Hz,4H),6.77(d,J=8.1Hz,2H),6.45(s,2H),4.57(s,1H),3.25(dd,J=13.5,6.3Hz,2H),2.99(dd,J=13.5,7.7Hz,2H),2.42(s,6H),2.28–2.22(m,2H),1.99(s,6H),1.74(dd,J=12.2,5.4Hz,2H),1.45(dd,J=12.4,8.9Hz,2H),1.33(m,2H),1.17(d,J=6.6Hz,6H),1.10(d,J=6.5Hz,6H),0.45(d,J=6.6Hz,6H),0.05(d,J=6.5Hz,6H).Peak at 596nm,FWHM=71nm in DCM;Peak at 596nm,FWHM=67nm in PMMA(6) Synthesis of ruthenium complex 82. Add a dimer (650 mg, 0.34 mmol), 2,8-dimethylnonane-4,6-dione (304 mg, 1.65 mmol), sodium carbonate (350 mg, 3.3 mmol) and B to a 100 ml round bottom flask. The diol ether (20 mL) was replaced with nitrogen in a reaction flask atmosphere, and the reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and dried over EtOAc (EtOAc:EtOAc: 1 H NMR (400MHz, CDCl 3 ) δ8.49 (d, J = 9.1Hz, 2H), 8.05 (d, J = 2.0Hz, 2H), 7.84 (s, 2H), 7.73 (d, J = 8.1Hz , 2H), 7.58 (dd, 9.1, 2 Hz, 2H), 7.57 (dd, 8.0 Hz, 4H), 7.29 (d, J = 8.0 Hz, 4H), 6.77 (d, J = 8.1 Hz, 2H), 6.45 (s, 2H), 4.57 (s, 1H), 3.25 (dd, J = 13.5, 6.3 Hz, 2H), 2.99 (dd, J = 13.5, 7.7 Hz, 2H), 2.42 (s, 6H), 2.28– 2.22 (m, 2H), 1.99 (s, 6H), 1.74 (dd, J = 12.2, 5.4 Hz, 2H), 1.45 (dd, J = 12.4, 8.9 Hz, 2H), 1.33 (m, 2H), 1.17 (d, J = 6.6 Hz, 6H), 1.10 (d, J = 6.5 Hz, 6H), 0.45 (d, J = 6.6 Hz, 6H), 0.05 (d, J = 6.5 Hz, 6H). Peak at 596 nm , FWHM=71nm in DCM; Peak at 596nm, FWHM=67nm in PMMA
实施例6配合物(DMPQ-TBH)2Ir(divm)的合成Synthesis of Complex 6 (DMPQ-TBH) 2Ir(divm)
Ra=对甲苯基,Rb=异丁基,Rc=氢,Rd=甲基,Re=氢,Rf=异丁基专利WO2016072780报道了该配合物R a = p-tolyl, R b = isobutyl, R c = hydrogen, R d = methyl, R e = hydrogen, R f = isobutyl patent WO2016072780 reports the complex
(1)2-(3,5-甲基苯基)-4-异丁基-6-(对甲苯基)喹啉的合成。向500ml圆底烧瓶中加入2-氯-4-异丁基-6-(对甲苯基)喹啉(22.3g,60mmol),1,3-二甲基苯硼酸(18g,120mmol),三水磷酸钾(80g,300mmol),2-双环己基膦-2',6'-二甲氧基联苯(1.97g,4.8mmol),三(二亚苄基丙酮)二钯(1.1g,1.2mmol)和甲苯(300mL),氮气置换反应氛围,将反应液加热回流12小时。TLC(石油醚:乙酸乙酯=10:1)显示反应完全,反应液用乙酸乙酯稀释,过滤,滤液旋干过柱(石油醚:乙酸乙酯=10:1)得白色2-(3,5-二甲基苯基)-4-异丁基-6-(对甲苯基)喹啉(17.3g,76%)。1H NMR(400MHz,CDCl3)δ8.23(d,J=8.4Hz,1H),8.14(d,J=2.0Hz,1H),7.95(dd,J=8.4,2.0Hz,1H),7.76(s,2H),7.66(s,1H),7.63(d,J=8.0Hz,2H),7.33(d,J=8.0Hz,2H),7.11(s,1H),3.03(d,J=7.2Hz,2H),2.45(s,9H),2.19(ht,J=7.2,6.7Hz,1H),1.58(s,6H),1.04(d,J=6.7Hz,6H).(1) Synthesis of 2-(3,5-methylphenyl)-4-isobutyl-6-(p-tolyl)quinoline. To a 500 ml round bottom flask was added 2-chloro-4-isobutyl-6-(p-tolyl)quinoline (22.3 g, 60 mmol), 1,3-dimethylphenylboronic acid (18 g, 120 mmol), tris Potassium phosphate (80 g, 300 mmol), 2-biscyclohexylphosphine-2',6'-dimethoxybiphenyl (1.97 g, 4.8 mmol), tris(dibenzylideneacetone)dipalladium (1.1 g, 1.2 mmol) And toluene (300 mL), the reaction atmosphere was replaced with nitrogen, and the reaction solution was heated under reflux for 12 hours. TLC (petroleum ether: ethyl acetate = 10:1) showed that the reaction was completed, the reaction mixture was diluted with ethyl acetate, filtered, and the filtrate was evaporated to dryness ( petroleum ether: ethyl acetate = 10:1) ,5-Dimethylphenyl)-4-isobutyl-6-(p-tolyl)quinoline (17.3 g, 76%). 1 H NMR (400 MHz, CDCl 3 ) δ 8.23 (d, J = 8.4 Hz, 1H), 8.14 (d, J = 2.0 Hz, 1H), 7.95 (dd, J = 8.4, 2.0 Hz, 1H), 7.76 (s, 2H), 7.66 (s, 1H), 7.63 (d, J = 8.0 Hz, 2H), 7.33 (d, J = 8.0 Hz, 2H), 7.11 (s, 1H), 3.03 (d, J = 7.2 Hz, 2H), 2.45 (s, 9H), 2.19 (ht, J = 7.2, 6.7 Hz, 1H), 1.58 (s, 6H), 1.04 (d, J = 6.7 Hz, 6H).
(2)二聚体的合成.向100ml圆底烧瓶中加入2-(3,5-二甲基苯基)-4-异丁基-6-(对甲苯基)喹啉(1.9g,4.0mmol),三水三氯化铱(0.353g,1.0mmol),乙二醇乙醚(15mL)和水(5mL),氮气置换反应瓶氛围。将反应体系加热回流12小时。将反应液冷至室温,固体析出,过滤,乙醚(8mL×3)洗涤,空气中干燥得二聚体(2.3g)。.(2) Synthesis of dimer. To a 100 ml round bottom flask was added 2-(3,5-dimethylphenyl)-4-isobutyl-6-(p-tolyl)quinoline (1.9 g, 4.0). Methyl) ruthenium trichloride trihydrate (0.353 g, 1.0 mmol), ethylene glycol diethyl ether (15 mL) and water (5 mL). The reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and a solid was precipitated, filtered, washed with diethyl ether (8 mL × 3), and dried in air to obtain a dimer (2.3 g). .
(3)配合物(DMPQ-TBH)2Ir(divm)的合成。向100ml圆底烧瓶中加入二聚体(2.3g,1.17 mmol),2,8-二甲基壬烷-4,6-二酮(1.2mL,5.85mmol),碳酸钠(1.24g,11.7mmol)和乙二醇乙醚(20mL),氮气置换反应瓶氛围,反应体系加热回流12小时。将反应液冷至室温,旋干过柱(石油醚:二氯甲烷=1:1)得到红色的配合物(DMPQ-TBH)2Ir(divm)(3.0g,85%)。1H NMR(400MHz,CDCl3)δ8.04-7.96(m,4H),7.86(s,2H),7.61(s,2H),7.53(d,J=7.9Hz,4H),7.52(m,2H),7.27(d,J=7.9Hz,4H),6.52(s,2H),4.27(s,1H),3.13-2.95(m,4H),2.40(s,6H),2.38(s,6H),2.33-2.16(m,2H),1.52-1.60(m,2H),1.43-1.52(m,2H),1.24-1.36(m,2H),1.12(d,J=5.1Hz,12H),0.51(d,J=6.5Hz,6H),0.18(d,J=6.4Hz,6H).MS(MALDI-TOF):1133.5[M]+Peak at 615nm,FWHM=58nm in DCM;Peak at 623nm,FWHM=68nm in PMMA(3) Synthesis of complex (DMPQ-TBH) 2 Ir(divm). Add a dimer (2.3 g, 1.17 mmol), 2,8-dimethyldecane-4,6-dione (1.2 mL, 5.85 mmol), sodium carbonate (1.24 g, 11.7 mmol) to a 100 ml round bottom flask. ) and ethylene glycol ether (20 mL), the atmosphere of the reaction flask was replaced with nitrogen, and the reaction system was heated to reflux for 12 hours. The reaction was cooled to room temperature, worked spin column (petroleum ether: dichloromethane = 1: 1) to give a red complex (DMPQ-TBH) 2 Ir ( divm) (3.0g, 85%). 1 H NMR (400MHz, CDCl 3 ) δ8.04-7.96 (m, 4H), 7.86 (s, 2H), 7.61 (s, 2H), 7.53 (d, J = 7.9Hz, 4H), 7.52 (m, 2H), 7.27 (d, J = 7.9 Hz, 4H), 6.52 (s, 2H), 4.27 (s, 1H), 3.13 - 2.95 (m, 4H), 2.40 (s, 6H), 2.38 (s, 6H) ), 2.33-2.16 (m, 2H), 1.52-1.60 (m, 2H), 1.43-1.52 (m, 2H), 1.24-1.36 (m, 2H), 1.12 (d, J = 5.1 Hz, 12H), 0.51 (d, J = 6.5 Hz, 6H), 0.18 (d, J = 6.4 Hz, 6H). MS (MALDI-TOF): 1133.5 [M] + Peak at 615 nm, FWHM = 58 nm in DCM; Peak at 623 nm, FWHM=68nm in PMMA
实施例7铱配合物157的合成Example 7 Synthesis of Ruthenium Complex 157
Ra=对甲苯基,Rb=异丁基,Rc=氢,Rd=甲基,Re=甲基,Rf=异丁基R a = p-tolyl, R b = isobutyl, R c = hydrogen, R d = methyl, R e = methyl, R f = isobutyl
(1)二聚体157的合成。向15ml封管中加入4-异丁基-6-(对-甲苯基)-2-(3,4,5-三甲基苯基)喹啉(393.3mg,1.0mmol),三水三氯化铱(65.5mg,0.2mmol),乙二醇乙醚(3mL)和水(1mL),氮气置换反应瓶氛围,将反应体系加热回流12小时。将反应液冷至室温,固体析出,过滤,空气中干燥得二聚体157(155mg,77%)。(1) Synthesis of dimer 157. To a 15 ml sealed tube was added 4-isobutyl-6-(p-tolyl)-2-(3,4,5-trimethylphenyl)quinoline (393.3 mg, 1.0 mmol), trichlorotrichloride Plutonium (65.5 mg, 0.2 mmol), ethylene glycol diethyl ether (3 mL) and water (1 mL) were replaced with nitrogen, and the reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, and a solid was precipitated, filtered, and dried in air to give dimer 157 (155 mg, 77%).
(2)铱配合物157的合成。向15ml封管中加入二聚体(202.5mg,0.1mmol),2,8-二甲基壬烷-4,6-二酮(0.1ml),碳酸钠(53mg,0.5mmol)和乙二醇乙醚(3mL),氮气置换反应瓶氛围,反应体系加热回流12小时。将反应液冷至室温,旋干,过柱(石油醚:乙酸乙酯=30:1)得到红色的铱配合物157(155mg,67%)。1H NMR(300MHz,CDCl3)δ7.97(d,J=1.8Hz,2H),7.88(s,1H),7.84(d,J=6.6Hz,3H),7.63(s,2H),7.57-7.54(m,3H),7.52-7.51(m,3H),7.48(d,J=2.1Hz,1H),7.28-7.25(m,3H),4.25(s,1H),3.03(d,J=6.9Hz,4H),2.40(s,12H),2.31-2.14(m,4H),2.13(s,0H),2.01(s,6H),1.57-1.55(m,2H),1.50-1.51(m,2H),1.35(s,6H),1.26(s,1H),1.14-1.11(m,12H),0.52(d,J=6.3Hz,6H),0.22(d,J=6.3Hz,6H).MS(MALDI-TOF):1160.6[M]+Peak in DCM at room temperature at 610nm,FWHM=58nm,Peak in PMMA at room temperature at 609nm,FWHM=51nm.(2) Synthesis of ruthenium complex 157. Dimer (202.5 mg, 0.1 mmol), 2,8-dimethyldecane-4,6-dione (0.1 ml), sodium carbonate (53 mg, 0.5 mmol) and ethylene glycol were added to a 15 ml sealed tube. Diethyl ether (3 mL) was placed under nitrogen to replace the atmosphere of the reaction flask, and the reaction was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, dried, and dried (EtOAc mjjjjjjjj 1 H NMR (300MHz, CDCl 3 ) δ7.97 (d, J = 1.8Hz, 2H), 7.88 (s, 1H), 7.84 (d, J = 6.6Hz, 3H), 7.63 (s, 2H), 7.57 -7.54 (m, 3H), 7.52 - 7.51 (m, 3H), 7.48 (d, J = 2.1 Hz, 1H), 7.28-7.25 (m, 3H), 4.25 (s, 1H), 3.03 (d, J) = 6.9 Hz, 4H), 2.40 (s, 12H), 2.31-2.14 (m, 4H), 2.13 (s, 0H), 2.01 (s, 6H), 1.57-1.55 (m, 2H), 1.50-1.51 ( m, 2H), 1.35 (s, 6H), 1.26 (s, 1H), 1.14-1.11 (m, 12H), 0.52 (d, J = 6.3 Hz, 6H), 0.22 (d, J = 6.3 Hz, 6H) MS (MALDI-TOF): 1160.6 [M] + Peak in DCM at room temperature at 610 nm, FWHM = 58 nm, Peak in PMMA at room temperature at 609 nm, FWHM = 51 nm.
实施例8铱配合物6的合成Example 8 Synthesis of ruthenium complex 6
Ra=对甲苯基,Rb=异丁基,Rc=甲基,Rd=甲基,Re=氢,Rf=甲基R a = p-tolyl, R b = isobutyl, R c = methyl, R d = methyl, R e = hydrogen, R f = methyl
(1)2,5-二甲基-N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代己酰胺的合成。向100ml封管中加入4'-甲基-[1,1'-联苯]-4-胺(183mg,1.0mmol),2,5-二甲基-3-氧代己酸乙酯(346.4mg,2mmol)和二甲苯(2mL),反应体系N2鼓泡三分后,缓慢升温至145℃,并在145℃条件下搅拌16小时。TLC(石油醚:乙酸乙酯=5:1)显示反应完全,将反应液冷至室温,产物析出,过滤,空气中干燥得淡黄色2,5-二甲基-N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代己酰胺(198mg,47%)。1H NMR(300MHz,DMSO)δ10.23(s,1H),7.67(d,J=8.7Hz,2H),7.60(d,J=8.7Hz,2H),7.53(d,J=8.1Hz,2H),7.25(d,J=8.1Hz,2H),3.66(q,J=6.9Hz,1H),2.45(t,J=6.6Hz,2H),2.33(s,3H),2.12-1.98(m,1H),1.24(d,J=6.9Hz,3H),0.85(t,J=6.6Hz,6H).(1) Synthesis of 2,5-dimethyl-N-(4'-methyl-[1,1'-biphenyl]-4-yl)-3-oxohexanamide. Add 4'-methyl-[1,1'-biphenyl]-4-amine (183 mg, 1.0 mmol), ethyl 2,5-dimethyl-3-oxohexanoate (346.4) to a 100 ml sealed tube. After the reaction system N 2 was bubbled for three minutes, the mixture was slowly heated to 145 ° C and stirred at 145 ° C for 16 hours. TLC (petroleum ether: ethyl acetate = 5:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, product was precipitated, filtered, and dried in air to give pale yellow 2,5-dimethyl-N-(4'-A Base-[1,1'-biphenyl]-4-yl)-3-oxohexanamide (198 mg, 47%). 1 H NMR (300MHz, DMSO) δ10.23 (s, 1H), 7.67 (d, J = 8.7Hz, 2H), 7.60 (d, J = 8.7Hz, 2H), 7.53 (d, J = 8.1Hz, 2H), 7.25 (d, J = 8.1 Hz, 2H), 3.66 (q, J = 6.9 Hz, 1H), 2.45 (t, J = 6.6 Hz, 2H), 2.33 (s, 3H), 2.12-1.98 ( m, 1H), 1.24 (d, J = 6.9 Hz, 3H), 0.85 (t, J = 6.6 Hz, 6H).
(2)4-异丁基-3-甲基-6-(对甲苯基)喹啉-2-醇的合成。向100mL三颈烧瓶中加入2,5-二甲基-N-(4'-甲基-[1,1'-联苯基]-4-基)-3-氧代己酰胺(1.61g,5mmol),氮气置换反应瓶氛围,9mL浓硫酸逐滴滴加入反应体系。将反应体系加热至50℃,搅拌12小时。TLC(石油醚:乙酸乙酯=3:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,空气中干燥得到黄色4-异丁基-3-甲基-6-(对甲苯基)喹啉-2-醇(1.3g,86%)。1H NMR(300MHz,CDCl3)δ11.69(s,1H),7.86(s,1H),7.71(d,J=8.4Hz,1H),7.57(d,J=8.1Hz,2H),7.36(d,J=8.4Hz,1H),7.25(d,J=8.1Hz,2H),2.89(d,J=7.2Hz,2H),2.35(s,3H),2.15(s,3H),2.01-1.94(m,1H),0.98(d,J=6.6Hz,6H).(2) Synthesis of 4-isobutyl-3-methyl-6-(p-tolyl)quinolin-2-ol. To a 100 mL three-necked flask was added 2,5-dimethyl-N-(4'-methyl-[1,1'-biphenyl]-4-yl)-3-oxohexanamide (1.61 g, 5 mmol), the atmosphere of the reaction flask was replaced with nitrogen, and 9 mL of concentrated sulfuric acid was added dropwise to the reaction system. The reaction system was heated to 50 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 3:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered and dried in air to give yellow 4-isobutyl-3-methyl -6-(p-Tolyl)quinolin-2-ol (1.3 g, 86%). 1 H NMR (300MHz, CDCl 3 ) δ11.69 (s, 1H), 7.86 (s, 1H), 7.71 (d, J = 8.4Hz, 1H), 7.57 (d, J = 8.1Hz, 2H), 7.36 (d, J = 8.4 Hz, 1H), 7.25 (d, J = 8.1 Hz, 2H), 2.89 (d, J = 7.2 Hz, 2H), 2.35 (s, 3H), 2.15 (s, 3H), 2.01 -1.94 (m, 1H), 0.98 (d, J = 6.6 Hz, 6H).
(3)2-氯-4-异丁基-3-甲基-6-(对甲苯基)喹啉的合成。向100ml三颈圆底烧瓶中加入4-异丁基-3-甲基-6-(对甲苯基)喹啉-2-醇(1g,3.3mmol),氮气置换反应瓶氛围,6mL三氯氧磷逐滴滴加入反应体系。将反应体系加热至100℃,搅拌18小时。TLC(石油醚:乙酸乙酯=6:1)显示反应完全,将反应液冷至室温,缓慢倒入冰水中,固体析出,过滤,水洗,空气中干燥得到黄色2-氯-4-异丁基-3-甲基-6-(对甲苯基)喹啉(950mg,90%)。(3) Synthesis of 2-chloro-4-isobutyl-3-methyl-6-(p-tolyl)quinoline. To a 100 ml 3-neck round bottom flask was added 4-isobutyl-3-methyl-6-(p-tolyl)quinolin-2-ol (1 g, 3.3 mmol), and the atmosphere of the reaction flask was replaced with nitrogen. Phosphorus was added dropwise to the reaction system. The reaction system was heated to 100 ° C and stirred for 18 hours. TLC (petroleum ether: ethyl acetate = 6:1) showed the reaction was completed, the reaction mixture was cooled to room temperature, poured slowly into ice water, solids were precipitated, filtered, washed with water, and dried in air to obtain yellow 2-chloro-4-isobutyl 3-methyl-6-(p-tolyl)quinoline (950 mg, 90%).
2-(3,5-二甲基苯基)-4-异丁基-3-甲基-6-(对-甲苯基)喹啉的合成。向100mL的schlenk管中加入2-氯-4-异丁基-3-甲基-6-(对甲苯基)喹啉(900mg,2.8mmol),3,5-二甲苯硼酸(836.5mg,5.6mmol),三水磷酸钾(3.73g,14mmol),2-双环己基膦-2',6'-二甲氧基联苯(91.9mg,0.224mmol),三(二亚苄基丙酮)二钯(51.3mg,0.056mmol)和甲苯(10mL),氮气置换反应瓶氛围。将反应体系加热至100℃,搅拌12小时。TLC(石油醚:乙酸乙酯=10:1)显示反应完全,反应液用乙酸乙酯稀释,过滤,滤液旋干过柱(石油醚:乙酸乙酯=50:1)得白色2-(3,5-二甲基苯基)-4-异丁基-3-甲基-6-(对-甲苯基)喹啉(770mg,70%)。1H NMR(300MHz,CDCl3)δ8.14(s,1H),8.13(s,1H),7.86(d,J=8.1Hz,1H),7.59(d,J=7.8Hz,2H),7.30(d,J=7.8Hz,2H),7.23(s,1H),7.10(s,2H),7.04(s,1H),3.09(d,J=7.2Hz,2H),2.41(s,3H),2.37(s,9H),2.22–2.10(m,1H),1.04(d,J=6.6Hz,6H).Synthesis of 2-(3,5-dimethylphenyl)-4-isobutyl-3-methyl-6-(p-tolyl)quinoline. To a 100 mL schlenk tube was added 2-chloro-4-isobutyl-3-methyl-6-(p-tolyl)quinoline (900 mg, 2.8 mmol), 3,5-xylene boronic acid (836.5 mg, 5.6 Methyl) potassium phosphate trihydrate (3.73 g, 14 mmol), 2-biscyclohexylphosphine-2',6'-dimethoxybiphenyl (91.9 mg, 0.224 mmol), tris(dibenzylideneacetone) palladium (51.3 mg, 0.056 mmol) and toluene (10 mL) were replaced with a nitrogen atmosphere. The reaction system was heated to 100 ° C and stirred for 12 hours. TLC (petroleum ether: ethyl acetate = 10:1) showed that the reaction was completed, the reaction mixture was diluted with ethyl acetate, filtered, and the filtrate was evaporated to dryness ( petroleum ether: ethyl acetate = 50:1) , 5-dimethylphenyl)-4-isobutyl-3-methyl-6-(p-tolyl)quinoline (770 mg, 70%). 1 H NMR (300MHz, CDCl 3 ) δ8.14 (s, 1H), 8.13 (s, 1H), 7.86 (d, J = 8.1Hz, 1H), 7.59 (d, J = 7.8Hz, 2H), 7.30 (d, J = 7.8 Hz, 2H), 7.23 (s, 1H), 7.10 (s, 2H), 7.04 (s, 1H), 3.09 (d, J = 7.2 Hz, 2H), 2.41 (s, 3H) , 2.37 (s, 9H), 2.22–2.10 (m, 1H), 1.04 (d, J = 6.6 Hz, 6H).
(4)二聚体6的合成。向15ml封管中加入2-(3,5-二甲基苯基)-4-异丁基-3-甲基-6-(对-甲苯基)喹啉(393.2mg,1mmol),三水三氯化铱(70.5mg,0.2mmol),乙二醇乙醚(3mL)和水(1mL),氮气置换反应瓶氛围。将反应体系加热至100℃,搅拌12小时。将反应液冷至室温,固体析出,过滤,乙醚洗涤,空气中干燥得二聚体6(180mg,89%)。 (4) Synthesis of dimer 6. To a 15 ml sealed tube was added 2-(3,5-dimethylphenyl)-4-isobutyl-3-methyl-6-(p-tolyl)quinoline (393.2 mg, 1 mmol), tris Ammonium trichloride (70.5 mg, 0.2 mmol), ethylene glycol diethyl ether (3 mL) and water (1 mL) were replaced with nitrogen atmosphere. The reaction system was heated to 100 ° C and stirred for 12 hours. The reaction solution was cooled to room temperature, and a solid was precipitated, filtered, washed with diethyl ether and dried in vacuo to give dimer 6 (180 mg, 89%).
(5)铱配合物6的合成。向15ml封管中加入二聚体6(202.5mg,0.1mmol),戊烷-2,4-二酮(0.1ml),碳酸钠(53mg,0.5mmol)和乙二醇乙醚(3mL),氮气置换反应瓶氛围,反应体系加热回流12小时。将反应液冷至室温,旋干,过柱(石油醚:乙酸乙酯=30:1)得到红色的铱配合物6(150mg,70%)。1H NMR(300MHz,DMSO)δ7.96(s,2H),7.84(d,J=9.0Hz,2H),7.62(s,2H),7.50-7.47(m,6H),7.24(m,J=7.5Hz,4H),6.55(s,2H),4.11(s,1H),3.20–3.08(m,4H),3.04(s,6H),2.36(d,J=8.9Hz,12H),2.18(m,1H),2.00(m,1H),1.31(s,6H),1.17(s,6H),1.09(d,J=6.6Hz,6H),0.97(d,J=6.6Hz,6H).MS(MALDI-TOF):1074.4[M-2]+Peak in DCM at room temperature at 631nm,FWHM=70nm,Peak in PMMA at room temperature at 630nm,FWHM=62nm.(5) Synthesis of ruthenium complex 6. Dimer 6 (202.5 mg, 0.1 mmol), pentane-2,4-dione (0.1 ml), sodium carbonate (53 mg, 0.5 mmol) and ethylene glycol diethyl ether (3 mL), nitrogen The atmosphere of the reaction flask was replaced, and the reaction system was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, dried, and dried (EtOAc:EtOAc:EtOAc 1 H NMR (300MHz, DMSO) δ7.96 (s, 2H), 7.84 (d, J = 9.0Hz, 2H), 7.62 (s, 2H), 7.50-7.47 (m, 6H), 7.24 (m, J = 7.5 Hz, 4H), 6.55 (s, 2H), 4.11 (s, 1H), 3.20 - 3.08 (m, 4H), 3.04 (s, 6H), 2.36 (d, J = 8.9 Hz, 12H), 2.18 (m, 1H), 2.00 (m, 1H), 1.31 (s, 6H), 1.17 (s, 6H), 1.09 (d, J = 6.6 Hz, 6H), 0.97 (d, J = 6.6 Hz, 6H) .MS (MALDI-TOF): 1074.4 [M-2] + Peak in DCM at room temperature at 631 nm, FWHM = 70 nm, Peak in PMMA at room temperature at 630 nm, FWHM = 62 nm.
实施例9铱配合物7的合成Example 9 Synthesis of Ruthenium Complex 7
Ra=对甲苯基,Rb=异丁基,Rc=甲基,Rd=甲基,Re=氢,Rf=异丁基.R a = p-tolyl, R b = isobutyl, R c = methyl, R d = methyl, R e = hydrogen, R f = isobutyl.
向15ml封管中加入上述二聚体(202.5mg,0.1mmol),2,8-二甲基壬烷-4,6-二酮(0.1ml),碳酸钠(53mg,0.5mmol)和乙二醇乙醚(3mL),氮气置换反应瓶氛围,反应体系加热回流12小时。将反应液冷至室温,旋干,过柱(石油醚:乙酸乙酯=15:1)得到红色的铱配合物7(155mg,72%)。1H NMR(300MHz,DMSO)δ7.99(d,J=1.5Hz,2H),7.89(d,J=9.0Hz,2H),7.67(s,2H),7.51-7.50(m,4H),7.50–7.46(m,2H),7.27(d,J=7.8Hz,4H),6.57(s,2H),4.12(s,1H),3.19–2.98(m,4H),3.05(s,6H),2.40(s,6H),2.38(s,6H),2.31–2.24(m,2H),1.62–1.54(m,2H),1.42–1.36(m,2H),1.30(s,6H),1.36–1.20(m,2H),1.18-1.11(m,12H),0.47(d,J=6.6Hz,6H), 0.26(d,J=6.6Hz,6H)。MS(MALDI-TOF):1160.6[M]+Peak in DCM at room temperature at 629nm,FWHM=66nm,Peak in PMMA at room temperature at 630nm,FWHM=60nm。The above dimer (202.5 mg, 0.1 mmol), 2,8-dimethyldecane-4,6-dione (0.1 ml), sodium carbonate (53 mg, 0.5 mmol) and ethylene were added to a 15 ml sealed tube. Alcohol ether (3 mL) was replaced with nitrogen in a reaction flask atmosphere, and the reaction was heated to reflux for 12 hours. The reaction solution was cooled to room temperature, dried and dried (EtOAc mjjjjjjjjjj 1 H NMR (300MHz, DMSO) δ7.99 (d, J = 1.5Hz, 2H), 7.89 (d, J = 9.0Hz, 2H), 7.67 (s, 2H), 7.51-7.50 (m, 4H), 7.50–7.46 (m, 2H), 7.27 (d, J = 7.8 Hz, 4H), 6.57 (s, 2H), 4.12 (s, 1H), 3.19–2.98 (m, 4H), 3.05 (s, 6H) , 2.40 (s, 6H), 2.38 (s, 6H), 2.31 - 2.24 (m, 2H), 1.62 - 1.54 (m, 2H), 1.42 - 1.36 (m, 2H), 1.30 (s, 6H), 1.36 -1.20 (m, 2H), 1.18-1.11 (m, 12H), 0.47 (d, J = 6.6 Hz, 6H), 0.26 (d, J = 6.6 Hz, 6H). MS (MALDI-TOF): 1160.6 [M] + Peak in DCM at room temperature at 629 nm, FWHM = 66 nm, Peak in PMMA at room temperature at 630 nm, FWHM = 60 nm.
实施例10铱配合物8的合成Example 10 Synthesis of ruthenium complex 8
Ra=对甲苯基,Rb=异丁基,Rc=甲基,Rd=甲基,Re=氢,Rf=异丙基.R a = p-tolyl, R b = isobutyl, R c = methyl, R d = methyl, R e = hydrogen, R f = isopropyl.
向15ml封管中加入上述二聚体(202.5mg 0.1mmol),2,6-二甲基庚烷-3,5-二酮(0.1ml),碳酸钠(53mg,0.5mmol)和乙二醇乙醚(3mL),氮气置换反应瓶氛围,反应体系加热回流12小时。将反应液冷至室温,旋干,过柱(石油醚:乙酸乙酯=15:1)得到红色的铱配合物8(153mg,71%)。1H NMR(300MHz,DMSO)δ8.14(s,2H),7.79(d,J=9.3Hz,2H),7.71(s,2H),7.65(s,2H),7.62-7.59(m,4H),7.29(d,J=8.1Hz,4H),6.45(s,2H),4.26(s,1H),3.18(s,4H),3.03(s,6H),2.35(s,6H),2.31(s,6H),2.21-2.11(m,2H),1.77-1.66(m,2H),1.13(s,6H),1.08-1.05(m,12H),0.61(d,J=6.6Hz,6H),0.41(d,J=6.6Hz,6H).MS(MALDI-TOF):1131.9[M-1]+Peak in DCM at room temperature at 630nm,FWHM=63nm,Peak in PMMA at room temperature at 631nm,FWHM=59nm.The above dimer (202.5 mg 0.1 mmol), 2,6-dimethylheptane-3,5-dione (0.1 ml), sodium carbonate (53 mg, 0.5 mmol) and ethylene glycol were added to a 15 ml sealed tube. Diethyl ether (3 mL) was placed under nitrogen to replace the atmosphere of the reaction flask, and the reaction was heated to reflux for 12 hours. The reaction mixture was cooled to room temperature, dried, and then evaporated, mjjjjjjj 1 H NMR (300MHz, DMSO) δ8.14 (s, 2H), 7.79 (d, J = 9.3Hz, 2H), 7.71 (s, 2H), 7.65 (s, 2H), 7.62-7.59 (m, 4H ), 7.29 (d, J = 8.1 Hz, 4H), 6.45 (s, 2H), 4.26 (s, 1H), 3.18 (s, 4H), 3.03 (s, 6H), 2.35 (s, 6H), 2.31 (s, 6H), 2.21-2.11 (m, 2H), 1.77-1.66 (m, 2H), 1.13 (s, 6H), 1.08-1.05 (m, 12H), 0.61 (d, J = 6.6 Hz, 6H) ), 0.41 (d, J = 6.6 Hz, 6H). MS (MALDI-TOF): 1131.9 [M-1] + Peak in DCM at room temperature at 630 nm, FWHM = 63 nm, Peak in PMMA at room temperature at 631 nm, FWHM=59nm.
实施例11铱配合物6,7,8,11,82,126,157,187,201作为磷光发光材料与传统磷光红光材料在光学性质方面的比较Example 11 Comparison of optical properties of iridium complexes 6, 7, 8, 11, 82, 126, 157, 187, 201 as phosphorescent materials and conventional phosphorescent red materials
分别对本发明所述铱配合物6,7,8,11,82,126,157,187,201作为磷光发光材料与传统磷光红光材料Ir(PQ)2(acac)、Ir(DMPQ)2(acac)和(DMPQ-TBH)2Ir(divm)材料光学性质进行比较测试方法如下:材料的带隙值(Eg)和LUMO值采用循环伏安法(CV)测得。整个测试过程在手套箱(Lab2000,Etelux)中的CHI600D电化学工作站(上海辰华仪器公司)上进行,以Pt柱为工作电极、以Ag/AgCl为参比电极,Pt丝为辅助电极构成三电极系统,测试过程采用的介质是0.1M六氟磷酸四丁基胺(Bu4NPF6)的二甲基二酰胺(DMF)溶液,所测电势均以加入的二茂铁(Fc)作为内标。材料的HOMO值通过Model IPS-4Ionization Energy Measurement System直接测试得到。材料的荧光量子效率(PLQE)依据公式(其中:Φs是样品的荧光量子产率,Φr是标样的荧光量子产率,η是溶液的折射率,As和Ar是样品和标样的荧光激发波长处的吸收值,Ιs和Ιr是样品和标样的荧光积分面积)采用相对法计算得到。将材料和已知量子产率的标物配置成相同浓度的聚甲基丙烯酸甲酯(PMMA)的三氯甲烷溶液,旋涂成膜,在相同的测量条件下,测得紫外吸收光谱(GENESYS 10S,Thermo)和荧光光谱(F97pro荧光分光光度计,棱光科技)。材料的光子能量(ET1)由公式E=hν=1240/λ(其中λ为材料PMMA膜的荧光光谱起始位置的切线波长)计算而来。)The ruthenium complexes 6, 7, 8, 11, 82, 126, 157, 187, 201 of the present invention are respectively used as phosphorescent materials and conventional phosphorescent red materials Ir(PQ) 2 (acac), Ir(DMPQ) 2 (acac) and ( DMPQ-TBH) 2 Ir (divm) material optical properties were compared as follows: the bandgap value (Eg) and LUMO value of the material were measured by cyclic voltammetry (CV). The whole test process was carried out on the CHI600D electrochemical workstation (Shanghai Chenhua Instrument Co., Ltd.) in the glove box (Lab2000, Etelux), with the Pt column as the working electrode, Ag/AgCl as the reference electrode, and Pt wire as the auxiliary electrode. In the electrode system, the medium used in the test was 0.1 M tetrabutylammonium hexafluorophosphate (Bu 4 NPF 6 ) in dimethyl diamide (DMF). The measured potentials were all within the added ferrocene (Fc). Standard. The HOMO values of the materials were directly tested by the Model IPS-4 Ionization Energy Measurement System. The fluorescence quantum efficiency (PLQE) of the material is based on the formula (where: Φ s is the fluorescence quantum yield of the sample, Φ r is the fluorescence quantum yield of the standard, η is the refractive index of the solution, and A s and A r are the absorption values at the fluorescence excitation wavelength of the sample and the standard, Ι s and Ι r are the fluorescence integral areas of the samples and standards) calculated using the relative method. The material and the known quantum yield standard were configured to the same concentration of polymethyl methacrylate (PMMA) in chloroform solution, spin-coated into a film, and the ultraviolet absorption spectrum was measured under the same measurement conditions (GENESYS). 10S, Thermo) and fluorescence spectra (F97pro Fluorescence Spectrophotometer, Prism). The photon energy (ET1) of the material is calculated from the formula E = hν = 1240 / λ (where λ is the tangential wavelength of the starting position of the fluorescence spectrum of the PMMA film of the material). )
a通过循环伏安计测得a measured by cyclic voltammeter
b三线态的能量b triplet energy
c PLQE效率以Ir(PQ)2(acac)在298K下的发光效率为基准值一个单位。 c PLQE efficiency is one unit of the luminous efficiency of Ir(PQ) 2 (acac) at 298K.
上表中本发明实施例与以公布的例子对比可以得到以下特征:以Ir(DMPQ)2(acac)在PMMA中的发光波长和效率为基准,喹啉4,6位取代的Ir配合物可以使光谱红移30nm,PMMA薄膜中的PLQE提高约20%。喹啉3,4,6位取代的Ir配合物可以使发光光谱继续红移约7nm。在喹啉2位取代的苯基的对位引入取代基的配体可以大幅度提高荧光量子效率,如铱配合物82、157,光谱有14-27nm的蓝移。薄膜PLQE最高的为喹啉4,6位取代和苯基3、4、5位取代的Ir的杂配合物。通过Eg(带隙值)的比较可知上述铱配合物可以用作红光材料,用作显示材料发光,红光饱和度很高。Comparing the examples of the present invention in the above table with the published examples, the following characteristics can be obtained: based on the wavelength and efficiency of Ir(DMPQ) 2 (acac) in PMMA, the Ir complex at the 4, 6 position of quinoline can be used. By shifting the spectrum red by 30 nm, the PLQE in the PMMA film is increased by about 20%. The Ir complex substituted at the 3, 4, and 6 positions of the quinoline can continue to red shift the luminescence spectrum by about 7 nm. The ligand in which the substituent is introduced at the para position of the phenyl group substituted at the 2-position of the quinoline can greatly improve the fluorescence quantum efficiency, such as the ruthenium complexes 82 and 157, and the spectrum has a blue shift of 14-27 nm. The highest film PLQE is a hetero complex of quinoline 4, 6-substituted and Ir substituted at the 3, 4, and 5 positions of phenyl. From the comparison of Eg (band gap value), it is understood that the above ruthenium complex can be used as a red light material, which is used as a display material to emit light, and the red light saturation is high.
实施例12铱配合物6,7,8,11,82,126,157,187,201作为磷光发光材料与传统磷光红光材料应用于器件后的比较Example 12 Comparison of ruthenium complex 6,7,8,11,82,126,157,187,201 as a phosphorescent luminescent material and a conventional phosphorescent red light material applied to a device
上述铱配合物6,7,8,82,187作为磷光发光材料与传统磷光红光材料Ir(PQ)2(acac)和Ir(DMPQ)2(acac)进行器件比较,设计OLED器件结构如下:ITO/HAT-CN(10nm)/TAPC(40nm)/NPB:red dopant(95:5,20nm)/Balq(10nm)/Bphen(50nm)/Liq(1nm)/Al。The above ruthenium complexes 6, 7, 8, 82, 187 were compared as a phosphorescent luminescent material with conventional phosphorescent red materials Ir(PQ) 2 (acac) and Ir(DMPQ) 2 (acac). The structure of the OLED device was designed as follows: ITO/ HAT-CN (10 nm) / TAPC (40 nm) / NPB: red dopant (95: 5, 20 nm) / Balq (10 nm) / Bphen (50 nm) / Liq (1 nm) / Al.
将装有OLED有机材料的坩埚和装有金属铝粒的坩埚依次放置在有机蒸发源和无机蒸发源位置上。关闭腔体,进行初抽真空和抽高真空步骤,使得OLED蒸镀设备内部蒸镀度达到10E-7Torr。The crucible containing the OLED organic material and the crucible containing the metal aluminum particles are sequentially placed at the positions of the organic evaporation source and the inorganic evaporation source. The cavity is closed, and the initial vacuuming and high vacuum steps are performed, so that the internal vapor deposition degree of the OLED evaporation apparatus reaches 10E-7 Torr.
OLED蒸镀成膜:打开OLED有机蒸发源,对OLED有机材料进行100℃预热,预热时间为15分钟,保证进一步移除OLED有机材料中的水汽。然后对需要蒸镀的有机材料进行快速升温加热处理,并打开蒸发源上方的挡板,直到该材料的蒸发源有有机材料跑出,同时晶振片检测器检测到蒸发速率时,然后进行缓慢升温,升温幅度为1~5℃,直到蒸发速率稳定在1A/秒时,打开掩膜板板正下方的挡板,进行OLED成膜,当电脑端观测到ITO基板上的有机膜达到预设膜厚时,关闭掩膜板挡板和蒸发源正上方挡板,关闭该有机材料的蒸发源加热器。其它有机材料和阴极金属材料的蒸镀工艺如上所述。封装采用UV环氧树脂进行光固化封装。封装后的样品进行IVL性能测试,IVL设备采用Mc Science M6100进行测试。器件数据对比数据测试如下表所示:OLED evaporation film formation: the OLED organic evaporation source is turned on, and the OLED organic material is preheated at 100 ° C for a preheating time of 15 minutes to ensure further removal of moisture in the OLED organic material. Then, the organic material that needs to be evaporated is subjected to rapid heating and heat treatment, and the baffle above the evaporation source is opened until the evaporation source of the material has organic material running out, and the crystal oscillator detects the evaporation rate, and then slowly heats up. The temperature rise is 1~5°C until the evaporation rate is stable at 1A/sec. The baffle directly under the mask plate is opened to form the OLED film. When the organic film on the ITO substrate is observed at the computer end, the film is preset. When thick, close the mask baffle and the baffle directly above the evaporation source to close the evaporation source heater of the organic material. The evaporation process of other organic materials and cathode metal materials is as described above. The package is UV-cured for photocuring. The encapsulated samples were tested for IVL performance and the IVL equipment was tested with the Mc Science M6100. The device data comparison data test is shown in the following table:
如图3,图4所示,通过器件数据对比,器件电致发光波长主要由Ir配合物本身光致发光的决定,在同一条件下,器件的效率高低也与Ir配合物本身的PLQE趋势一致。因此,本发明所公开的高PLQE的多取代喹啉配位的铱杂配化合物可以在其它器件中获得比现有红光材料更高的器件效率。As shown in Fig. 3 and Fig. 4, the device electroluminescence wavelength is mainly determined by the photoluminescence of the Ir complex itself. Under the same conditions, the efficiency of the device is also consistent with the PLQE trend of the Ir complex itself. . Therefore, the high PLQE polysubstituted quinoline-coordinated doped compound of the present invention can achieve higher device efficiency in other devices than existing red light materials.
实施例13本发明所述多取代喹啉配位的铱杂配化合物在OLED方面的应用Example 13 Application of the polysubstituted quinoline coordinated doping compound of the present invention in OLED
本文所述多取代喹啉配位的铱杂配化合物化合物适用于各种光学和光电器件,例如太阳能和光敏感器这样的光吸收器件、光发射器或既有光吸收又有光发射能力的器件以及用于生物应用的标志物。下面以有机发光二极管(OLED)为例对本发明所述多取代喹啉配位的铱杂配化合物在光电器件方面的应用进行描述。The polysubstituted quinoline-coordinated oxime compound compounds described herein are suitable for use in a variety of optical and optoelectronic devices, such as light absorbing devices such as solar and light sensors, light emitters, or devices having both light absorbing and light emitting capabilities. And markers for biological applications. The use of the polysubstituted quinoline-coordinated doped compound of the present invention in an optoelectronic device will be described below by taking an organic light-emitting diode (OLED) as an example.
图1显示了OLED 1000的断面图,如图所示,OLED 1000包含一阳极1004位于基体1002之上,其材质为透明材料,例如氧化铟锡;空穴传输材料层(HTL)1006与阳极1004相连;发光功能层1008位于空穴传输材料层1006之上,所述发光功能层1008包括发射体和主体的发光材料;电子传输材料层(ETL)1010及一金属阴极层1012依次设置于发光功能层1008之上。所述OLED及类似发光器件可包含单层或叠层。从各方面说,该单或叠层的任何层均可包含氧化铟锡(ITO)、MoO3、Ni2O3、聚(3,4-乙烯二氧噻吩)(PEDOT)、聚苯乙烯磺酸钠(PSS)、4,4',4”-((1E,1'E,1”E)-环丙烷-1,2,3-三亚甲基三(氰基-甲基亚基))三(2,3,5,6-四氟苯腈)(NHT-49)、2,2'-(全氟萘烷-2,6-二基)丙二腈(NHT-51)、2,3,5,6-四氟四氰基-对醌二甲烷(F4-TCNQ)、N,N′-二-1-萘基-N,N′-联苯-1,1′-联苯-4,4′二胺(NPD)、1,1-顺((二-4-对甲苯氨基)苯基)环己烷(TAPC),2,6-顺(N-咔唑基)-吡啶(mCpy)、2,8-顺(叠氮磷酸二苯酯)二苯并噻吩(PO15)、LiF、LiQ、Cs2CO3、CaCO3、Al或其组合物。在这一具体实施方式中,发光功能层1008可包含本发明所述多取代喹啉配位的铱杂配化合物中的一或多个化合物,本实验选择使用铱配合物7,可选择性地连带一主体材料。ETL层1010和1006也可包含一或多个多取代喹啉配位的铱杂配化合物以及与电极接近的另一注入层。注入层的材料可包括(电子注
入层)EIL、(空穴注入层)HIL和CPL(盖层),其形式可以是单一层或分散在传输材料中。主体材料可以是本技术中已知的任何合适的主体材料。OLED的发光颜色由发光功能层1008的发光能量(光学能隙)决定,通过对发光化合物进行调谐处理和/或主体材料的电子结构来调谐发光功能层1008的发光能量(光学能隙)。HTL层1006中的空穴传输材料和ETL层1010中的电子传输材料可包含本技术中已知的任何合适的空穴传输体。1 shows a cross-sectional view of an
实施例14铱配合物7在CH2Cl2、PMMA中的光谱测试Example 14 Spectroscopic Testing of Ruthenium Complex 7 in CH 2 Cl 2 , PMMA
将实验所制备的铱配合物7置于CH2Cl2、PMMA中进行光谱测试,结果如图2所示,在本实验的测量所得到的发射光谱中可以看出,在不同激发光的激发下都可以发射出很强的荧光,两个主峰分别在629nm和630nm,因此铱配合物7可以用来制备高饱和度的深红光发光器件The ruthenium complex 7 prepared by the experiment was placed in CH2Cl2 and PMMA for spectral test. The results are shown in Fig. 2. It can be seen from the emission spectrum obtained by the measurement of this experiment that it can be excited by different excitation lights. Strong fluorescence is emitted, the two main peaks are at 629nm and 630nm, respectively, so the ruthenium complex 7 can be used to prepare a high saturation deep red light-emitting device.
实施例15铱配合物7的器件的电致发光光谱。Example 15 Electroluminescence spectrum of a device of ruthenium complex 7.
对铱配合物7作为发光材料制备的器件进行电致发光光谱测试,结果如图5所示,,说明铱配合物7属于高饱和度红光,此材料可以完全满足显示器中的红光的色度要求。The electroluminescence spectroscopy test was performed on the device prepared by using the ruthenium complex 7 as a luminescent material, and the result is shown in FIG. 5, indicating that the ruthenium complex 7 is a high-saturation red light, and the material can fully satisfy the red color in the display. Degree requirements.
以上显示和描述了本发明的基本原理和主要特征和本发明的优点。本行业的技术人员应该了解,本发明不受上述实施例的限制,上述实施例和说明书中描述的只是说明本发明的原理,在不脱离本发明精神和范围的前提下,本发明还会有各种变化和改进,这些变化和改进都落入要求保护的本发明范围内。本发明要求保护范围由所附的权利要求书及其等效物界定。 The basic principles and main features of the present invention and the advantages of the present invention are shown and described above. It should be understood by those skilled in the art that the present invention is not limited by the foregoing embodiments, and that the present invention is only described in the foregoing description and the description of the present invention, without departing from the spirit and scope of the invention. Various changes and modifications are intended to be included within the scope of the invention as claimed. The scope of the invention is defined by the appended claims and their equivalents.
Claims (9)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710858082.7 | 2017-09-21 | ||
| CN201710858082.7A CN107459535A (en) | 2017-09-21 | 2017-09-21 | Polysubstituted quinoline coordinated iridium hybrid compound and preparation method and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019056515A1 true WO2019056515A1 (en) | 2019-03-28 |
Family
ID=60552901
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2017/109732 Ceased WO2019056515A1 (en) | 2017-09-21 | 2017-11-07 | Multi-substituted quinoline-coordinated iridium-hybridized compound, preparation method therefor and application thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN107459535A (en) |
| WO (1) | WO2019056515A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115010765A (en) * | 2021-07-20 | 2022-09-06 | 安徽工业大学 | Red light organic electrophosphorescent material and preparation method and application thereof |
| US20230109178A1 (en) * | 2021-07-23 | 2023-04-06 | Beijing Summer Sprout Technology Co., Ltd. | Luminescent material having multi-substituted phenyl ligand |
| WO2024102884A1 (en) * | 2022-11-10 | 2024-05-16 | Bristol-Myers Squibb Company | Substituted quinolone compounds useful as inhibitors of tlr9 |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019114668A1 (en) * | 2017-12-14 | 2019-06-20 | 广州华睿光电材料有限公司 | Transition metal complex material and application thereof in electronic devices |
| CN108299510B (en) * | 2018-02-22 | 2020-06-05 | 南京工业大学 | A class of iridium complexes containing azaarenes auxiliary ligands and their preparation methods and applications |
| EP3560941B1 (en) * | 2018-04-23 | 2021-09-08 | Samsung Electronics Co., Ltd. | Organometallic compound, organic light-emitting device including the organometallic compound, and diagnostic compound including the organometallic compound |
| KR102675779B1 (en) * | 2018-09-19 | 2024-06-18 | 삼성전자주식회사 | Organometallic compound, organic light emitting device including the same and a composition for diagnosing including the same |
| CN112094301B (en) * | 2020-09-23 | 2022-12-27 | 南京佳诺霖光电科技有限公司 | Isoquinoline metal complex and preparation method and application thereof |
| CN114751940B (en) * | 2022-03-23 | 2023-04-07 | 广州追光科技有限公司 | Binuclear iridium complex of pyrazole auxiliary ligand and application thereof |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102703059A (en) * | 2012-05-31 | 2012-10-03 | 吉林奥来德光电材料股份有限公司 | Phosphorescent luminescent materials and preparation method and application thereof |
| CN103045231A (en) * | 2012-10-18 | 2013-04-17 | 吉林奥来德光电材料股份有限公司 | Aryl-biquinoline iridium-complex organic phosphorescent material, preparation method and applications of aryl-biquinoline iridium-complex organic phosphorescent material |
| CN103254243A (en) * | 2013-05-02 | 2013-08-21 | 太原理工大学 | Polysubstituted phenylquinoline iridium (III) complex, preparation method thereof and application |
| CN103694277A (en) * | 2013-12-12 | 2014-04-02 | 江西冠能光电材料有限公司 | Red-phosphorescence organic light emitting diode (LED) |
| WO2016080749A1 (en) * | 2014-11-18 | 2016-05-26 | Rohm And Haas Electronic Materials Korea Ltd. | A plurality of dopant materials and organic electroluminescent device comprising the same |
| CN106699763A (en) * | 2016-12-16 | 2017-05-24 | 江苏三月光电科技有限公司 | Chemical compound taking quinazolinone derivative as core and application of chemical compound |
| CN107075361A (en) * | 2014-11-04 | 2017-08-18 | 罗门哈斯电子材料韩国有限公司 | Novel combination of host compound and dopant compound and organic electroluminescent device comprising same |
| CN107075362A (en) * | 2014-11-06 | 2017-08-18 | 罗门哈斯电子材料韩国有限公司 | Organic electroluminescent compounds and the organic electroluminescence device for including the compound |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100933226B1 (en) * | 2007-11-20 | 2009-12-22 | 다우어드밴스드디스플레이머티리얼 유한회사 | Novel red phosphorescent compound and organic light emitting device employing it as light emitting material |
-
2017
- 2017-09-21 CN CN201710858082.7A patent/CN107459535A/en active Pending
- 2017-11-07 WO PCT/CN2017/109732 patent/WO2019056515A1/en not_active Ceased
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102703059A (en) * | 2012-05-31 | 2012-10-03 | 吉林奥来德光电材料股份有限公司 | Phosphorescent luminescent materials and preparation method and application thereof |
| CN103045231A (en) * | 2012-10-18 | 2013-04-17 | 吉林奥来德光电材料股份有限公司 | Aryl-biquinoline iridium-complex organic phosphorescent material, preparation method and applications of aryl-biquinoline iridium-complex organic phosphorescent material |
| CN103254243A (en) * | 2013-05-02 | 2013-08-21 | 太原理工大学 | Polysubstituted phenylquinoline iridium (III) complex, preparation method thereof and application |
| CN103694277A (en) * | 2013-12-12 | 2014-04-02 | 江西冠能光电材料有限公司 | Red-phosphorescence organic light emitting diode (LED) |
| CN107075361A (en) * | 2014-11-04 | 2017-08-18 | 罗门哈斯电子材料韩国有限公司 | Novel combination of host compound and dopant compound and organic electroluminescent device comprising same |
| CN107075362A (en) * | 2014-11-06 | 2017-08-18 | 罗门哈斯电子材料韩国有限公司 | Organic electroluminescent compounds and the organic electroluminescence device for including the compound |
| WO2016080749A1 (en) * | 2014-11-18 | 2016-05-26 | Rohm And Haas Electronic Materials Korea Ltd. | A plurality of dopant materials and organic electroluminescent device comprising the same |
| CN106699763A (en) * | 2016-12-16 | 2017-05-24 | 江苏三月光电科技有限公司 | Chemical compound taking quinazolinone derivative as core and application of chemical compound |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115010765A (en) * | 2021-07-20 | 2022-09-06 | 安徽工业大学 | Red light organic electrophosphorescent material and preparation method and application thereof |
| CN115160378A (en) * | 2021-07-20 | 2022-10-11 | 安徽工业大学 | A kind of red light organic electrophosphorescent material, preparation method and application thereof |
| US20230109178A1 (en) * | 2021-07-23 | 2023-04-06 | Beijing Summer Sprout Technology Co., Ltd. | Luminescent material having multi-substituted phenyl ligand |
| WO2024102884A1 (en) * | 2022-11-10 | 2024-05-16 | Bristol-Myers Squibb Company | Substituted quinolone compounds useful as inhibitors of tlr9 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107459535A (en) | 2017-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019056515A1 (en) | Multi-substituted quinoline-coordinated iridium-hybridized compound, preparation method therefor and application thereof | |
| Sasabe et al. | Wide-energy-gap electron-transport materials containing 3, 5-dipyridylphenyl moieties for an ultra high efficiency blue organic light-emitting device | |
| EP2871222B1 (en) | Compound for organic optoelectric device, organic optoelectric device comprising same, and display apparatus comprising organic optoelectric device | |
| TWI530493B (en) | Benzimidazole compounds and organic photoelectric device with the same | |
| WO2006114966A1 (en) | Organic electroluminescent device, display and illuminating device | |
| CN107501330A (en) | Metal complex comprising azabenzimidazoles carbene ligands and its purposes in OLED | |
| WO2006098120A1 (en) | Organic electroluminescent device material and organic electroluminescent device | |
| WO2007097153A1 (en) | Organic electroluminescence element material, organic electroluminescence element, display device and illuminating device | |
| TW201522570A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR102815129B1 (en) | Metal complexes and their applications | |
| TW201726699A (en) | Compound and its electronic components | |
| CN110372756B (en) | A kind of divalent platinum complex and its preparation method and application | |
| WO2006132012A1 (en) | Organic electroluminescence element, lighting apparatus and display device | |
| US8753756B2 (en) | Green phosphorescent iridium complexes, fabrication method thereof and organic light-emitting diodes comprising the same | |
| KR20130122602A (en) | New compounds and organic electronic device using the same | |
| CN106431827A (en) | Novel compound and organic electronic device using the same | |
| WO2006013739A1 (en) | Organic electroluminescent device, illuminating device and display | |
| CN110229192B (en) | Deuterated iridium complex and preparation method and application thereof | |
| CN111961089B (en) | An organic metal complex and its preparation method and use | |
| CN108690091B (en) | Iridium complex and preparation method and application thereof | |
| CN108299510B (en) | A class of iridium complexes containing azaarenes auxiliary ligands and their preparation methods and applications | |
| TWI387634B (en) | Green phosphorescent iridium complexes, fabrication method thereof and organic light-emitting diodes comprising the same | |
| WO2020237885A1 (en) | Dark blue thermal activation delayed fluorescent material and preparation method therefor, and electroluminescent device | |
| CN108409797B (en) | Organic metal complex, preparation method and application thereof | |
| Xie et al. | Synthesis and properties of naphthalimide derivative functionalized N, N′-bicarbazole for complementary white light-emitting diodes with exciplex |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17926246 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 17926246 Country of ref document: EP Kind code of ref document: A1 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 17926246 Country of ref document: EP Kind code of ref document: A1 |