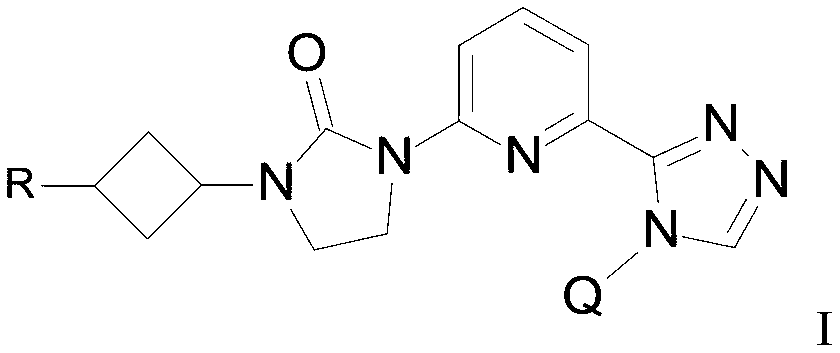
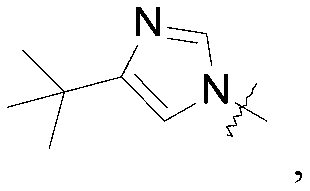
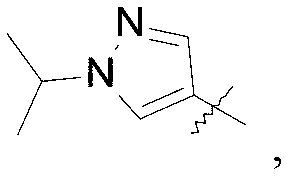
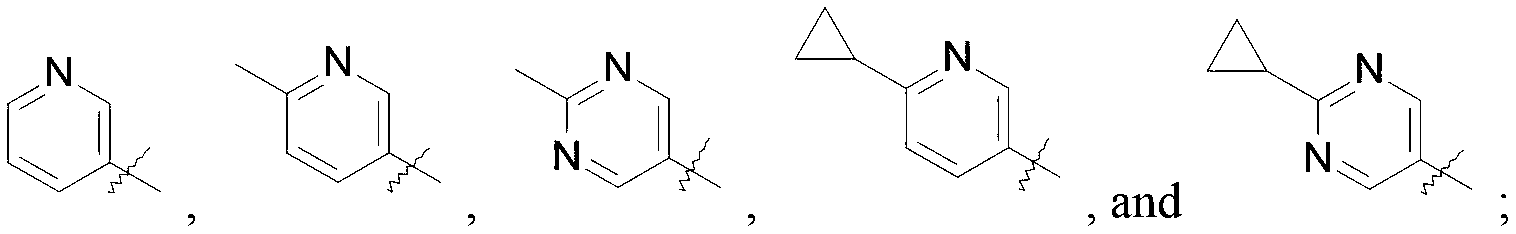
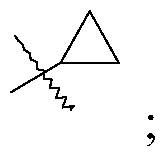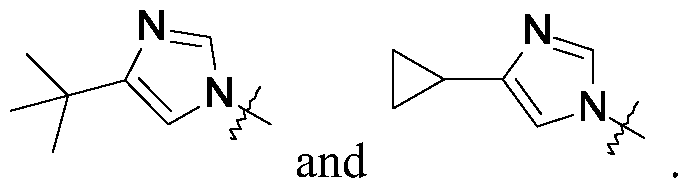WO2019047094A1 - Cyclobutyl-imidazolidinone compounds - Google Patents
Cyclobutyl-imidazolidinone compounds Download PDFInfo
- Publication number
- WO2019047094A1 WO2019047094A1 PCT/CN2017/100858 CN2017100858W WO2019047094A1 WO 2019047094 A1 WO2019047094 A1 WO 2019047094A1 CN 2017100858 W CN2017100858 W CN 2017100858W WO 2019047094 A1 WO2019047094 A1 WO 2019047094A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- mmol
- triazol
- pharmaceutically acceptable
- isopropyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*c1ccc(C)nc1 Chemical compound C*c1ccc(C)nc1 0.000 description 2
- XBHHKVWZNGPYQD-UHFFFAOYSA-N CC(C)[n]1ncc(C)c1 Chemical compound CC(C)[n]1ncc(C)c1 XBHHKVWZNGPYQD-UHFFFAOYSA-N 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-N c1ccncc1 Chemical compound c1ccncc1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
Definitions
- Cyclobutyl-Imidazolidinone compounds of this invention are inhibitors of apoptois signal-regulating kinase 1 (ASK1) .
- ASK1 is a member of the large mitogen-activated protein kinase kinase kinase ( “MAP3K” ) family. ASK1 activation and signaling are associated with broad range of diseases. Compounds that inhibit ASK1 are desired for use in the treatment of ASK1 mediated conditions.
- MA3K mitogen-activated protein kinase kinase kinase
- Nonalcoholic steatohepatitis is a liver disease with an etiological constellation characterized by macrovesicular hepatic steatosis, inflammation hepatocyte ballooning and fibrosis.
- NASH Nonalcoholic steatohepatitis
- U.S. Patent No. 8,742,126 discloses 5- (4-cyclopropyl-1H-imidazol-l-yl) -N- (6- (4-isopropyl-4H-1, 2, 4-triazol-3-y-1) pyridin-2-yl) -2-fluoro-4-methylbenzamide as an ASK1 inhibitor.
- U.S. Patent Application Publication No. US 2015/0342943 discloses a method of preventing and/or treating liver disease using an ASK1 inhibitor.
- the present invention provides a compound of Formula I
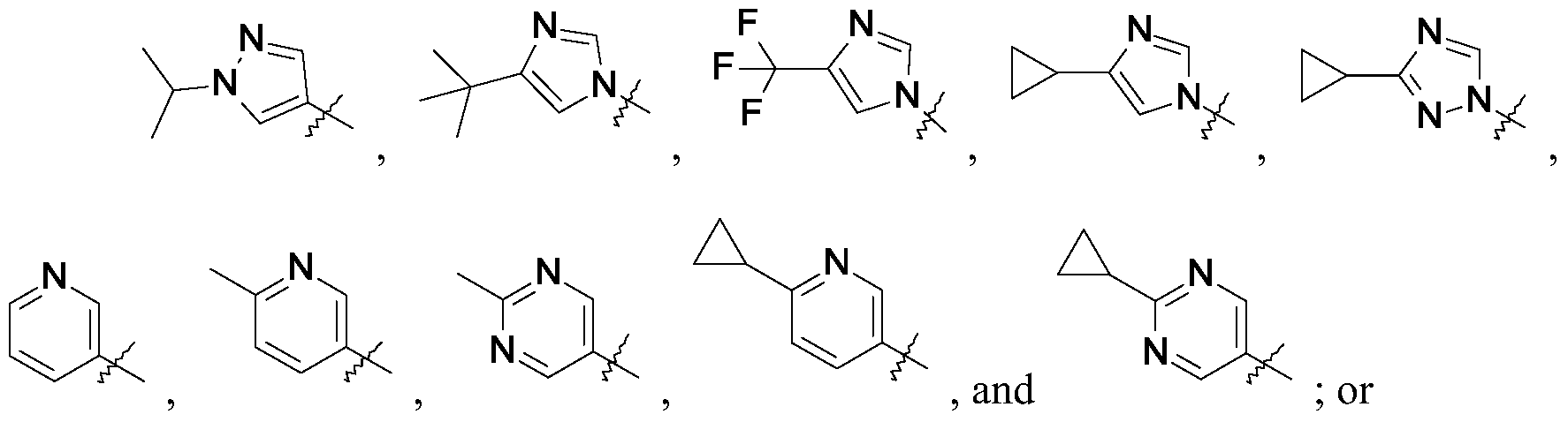
- Q is selected from the group consisting of-C (CH 3 ) 2 and
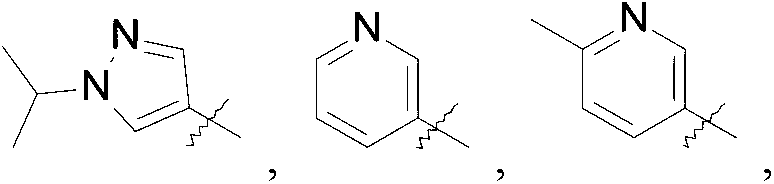
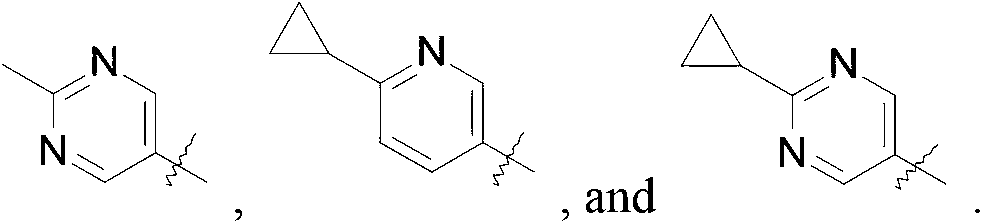
- R is selected from the group consisting of
- R is selected from the group consisting of and Q is -C (CH 3 ) 2 .
- R is selected from the group consisting of and Q is
- R is and Q is -C (CH 3 ) 2 .
- R is selected from the group consisting of
- Q is-C (CH 3 ) 2 .
- R is selected from the group consisting of and Q is
- R is and Q is -C (CH 3 ) 2
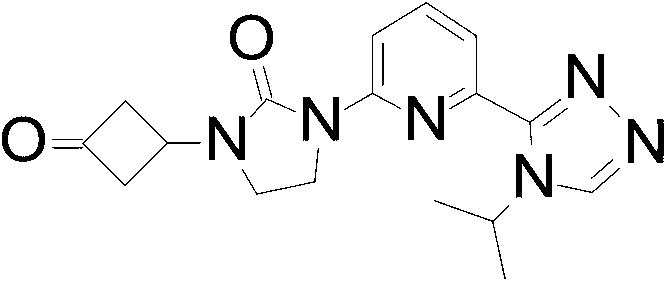
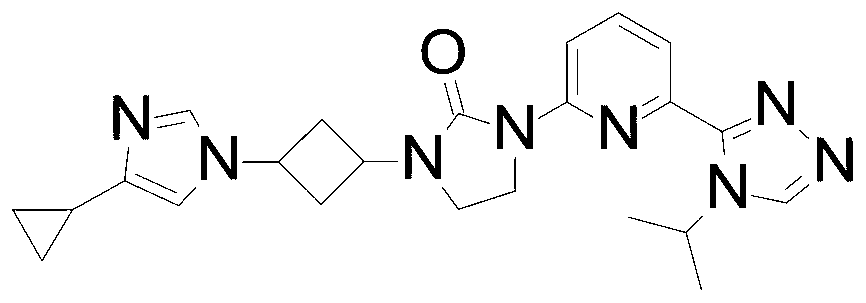
- the compound of Formula I is 1- [3- (4-cyclopropylimidazol-1-yl) cyclobutyl] -3 - [6- (4-isopropyl-1, 2, 4-triazol-3 -yl) -2-pyridyl] imidazolidin-2-one, or a pharmaceutically acceptable salt thereof.
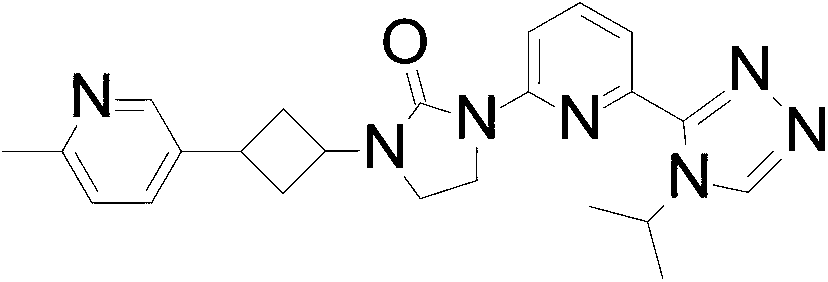
- the compound of Formula I is 1- (6- (4-isopropyl-4H-1, 2, 4-triazol-3-yl) pyridin-2-yl) -3- (3- (6-methylpyridin-3-yl) cyclobutyl) imidazolidin-2-one, or a pharmaceutically acceptable salt thereof.
- the present invention provides a pharmaceutical composition
- a pharmaceutical composition comprising a compound of Formula I, or a pharmaceutically acceptable salt thereof, together with one or more pharmaceutically acceptable carriers, diluents, or excipients.
- the present invention provides a method for treating a condition mediated by ASK1 activity comprising administering to the mammal in need of treatment, an effective amount of a compound of Formula I, or a pharmaceutically acceptable salt thereof.
- the present invention also provides a method for treating liver disease, comprising administering to a mammal in need thereof, an effective amount of a compound of Formula I, or a pharmaceutically acceptable salt thereof.
- the present invention provides a method for treating nonalcoholic steatohepatitis (NASH) , comprising administering to a mammal in need thereof, an effective amount of a compound of Formula I, or a pharmaceutically acceptable salt thereof.
- NASH nonalcoholic steatohepatitis
- the present invention provides a compound of Formula I, or a pharmaceutically acceptable salt thereof, for use in therapy. Further, provided is a compound of the present invention, or a pharmaceutically acceptable salt thereof or pharmaceutical composition, for use in the treatment of liver disease. Further, provided is a compound of the present invention, or a pharmaceutically acceptable salt thereof or pharmaceutical composition, for use in the treatment of NASH.
- a compound of Formula I for use in the manufacture of a medicament for the treatment of liver disease.
- the medicament is for the treatment of NASH.
- a compound of the present invention is preferably formulated as pharmaceutical compositions administered by any route which makes the compound bioavailable. Most preferably, such compositions are for oral administration.
- Such pharmaceutical compositions and processes for preparing same are well known in the art. See, e.g., Remington: The Science and Practice of Pharmacy (L.V. Allen, Editor, 22 nd Edition, Pharmaceutical Press, 2012) .
- “Pharmaceutically-acceptable salt” refers to salts of the compound of the invention considered to be acceptable for clinical and/or veterinary use.
- Pharmaceutically acceptable salts and common methodology for preparing them are well known in the art. See, e.g., P. Stahl, et al., Handbook of Pharmaceutical Salts: Properties, Selection and Use, (VCHA/Wiley-VCH, 2002)
- a human is a preferred mammal.
- “patient” refers to a mammal in need of treatment.
- the term “effective amount” or “therapeutically effective amount” of a compound refers to an amount, or a dosage, which is effective in treating a disorder or a disease, such as NASH, chronic kidney disease, or diabetic nephropathy as described herein.
- the attending diagnostician as one skilled in the art, can readily determine an effective amount by the use of conventional techniques and by observing results obtained under analogous circumstances.
- a number of factors are considered, including but not limited to, the compound to be administered; the co-administration of other agents, if used; the species of mammal; its size, age, and general health; the degree of involvement or the severity of the disorder; the response of the individual patient; the mode of administration; the bioavailability characteristics of the preparation administered; the dose regimen selected; the use of other concomitant medication; and other relevant circumstances.
- the pharmaceutical composition is administered to a patient in amounts effective to treat liver disease, more particularly, NASH.
- An appropriate amount or dose effective to treat a patient can be determined by a health care provider.
- treatment and “treating” as used herein are intended to refer to all processes wherein there may be a slowing, interrupting, arresting, controlling, or stopping of the progression of an existing disorder and/or symptoms thereof, but does not necessarily indicate a total elimination of all symptoms.
- liver disease as used herein embraces liver conditions or symptoms associated with ASK1 mediation, for example, metabolic liver disease, steatosis, liver fibrosis, primary sclerosing cholangitis (PSC) , cirrhosis, liver fibrosis, non-alcoholic fatty liver disease (NAFLD) , nonalcoholic steatohepatitis (NASH) , hepatic ischemia reperfusion injury, and primary biliary cirrhosis (PBC) .
- metabolic liver disease for example, metabolic liver disease, steatosis, liver fibrosis, primary sclerosing cholangitis (PSC) , cirrhosis, liver fibrosis, non-alcoholic fatty liver disease (NAFLD) , nonalcoholic steatohepatitis (NASH) , hepatic ischemia reperfusion injury, and primary biliary cirrhosis (PBC) .
- PSC primary sclerosing cholangit
- compositions are formulated as a tablet or capsule for oral administration.
- the tablet or capsule can include a compound of the present invention in an amount effective to treat liver disease, particularly NASH.
- ACN refers to acetonitrile
- ADP refers to adenosine diphosphate
- AIBN refers to azobisisobutyronitrile
- ATP refers to adenosine triphosphate
- boc refers to tert-butoxycarbonyl
- BSA Bovine Serum Albumin
- DCM refers to dichloromethane
- DMEDA refers to N, N’ -dimethylethylenediamine
- DMEM refers to Dulbecco’s Modified Eagle’s Medium
- DF refers to N, N-dimethylformamide
- DMP refers to 1, 1, 1 -triacetoxy-1, 1 -dihydro-1, 2-benziodoxol-3 (1H) -one, also known as Dess-
- the intermediates described in the following preparations may contain a number of nitrogen, hydroxy, and acid protecting groups such as esters.
- the variable protecting group may be the same or different in each occurrence depending on the particular reaction conditions and the particular transformations to be performed.
- the protection and deprotection conditions are well known to the skilled artisan and are described in the literature. See. e.g., Greene and Wuts, Protective Groups in Organic Synthesis, (T. Greene and P. Wuts, eds., 2d ed. 1991) .
- the compounds of the present invention, or salts thereof may be prepared by a variety of procedures known in the art, some of which are illustrated in the Preparations and Examples below.
- the specific synthetic steps for each of the routes described may be combined in different ways, to prepare compounds of the invention, or salts thereof.
- the products of each step can be recovered by conventional methods well known in the art, including extraction, evaporation, precipitation, chromatography, filtration, trituration, and crystallization.
- the reagents and starting materials are readily available to one of ordinary skill in the art. Others may be made by standard techniques of organic and heterocyclic chemistry which are analogous to the syntheses of known structurally-similar compounds and the procedures described in the Preparations and Examples which follow including any novel procedures.
- Methansulfonyl chloride (0.747 g, 6.45 mmol) is added to a solution of 1- (3-hydroxycyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one (1.227 g, 3.23 mmol) and trimethylamine (0.979 g, 9.68 mmol) in DCM (20 mL) at 0 °C. The mixture is stirred at 0 °C for 15 minutes. The reaction is quenched with aq. NaHCO 3 (30 mL) and the product is extracted with DCM (2 ⁇ 50 mL) .
- DMP (6.9 g, 16.0 mmol) is added to a solution of 1- (3-hydroxycyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one; hydrochloride (3.9 g, 11.0 mmol) in DCM (10 mL) at room temperature. The mixture is stirred at room temperature for 20 hours. The reaction is quenched with a sat. Na 2 SO 3 and NaHCO 3 (30 mL) solution and the product is extracted with DCM (3 ⁇ 50 mL) . The organic extracts are dried over Na 2 SO 4 and concentrated in vacuo.
- the filtrate is purified by silica gel flash chromatography, eluting with a gradient of 0%to 3%MeOH in DCM to give the title compound (1.175 g, 92.81%) as a yellow oil with a cis/trans ratio of about 1 ⁇ 2.
- ES/MS (m/z) 452.8 (M+1) .
- the reaction is quenched with water (80 mL) and the product is extracted with DCM (3 ⁇ 150 mL) .
- the organic extracts are washed with brine (30 mL) , dried over Na 2 SO 4 , and concentrated in vacuo.
- the reaction is quenched with water (20 mL) and the product is extracted with DCM (3 ⁇ 40 mL) .
- the organic extracts are dried over Na 2 SO 4 and concentrated in vacuo.
- the residue is purified by HPLC with the following conditions: column: SunFire C18 5 ⁇ , 30*100 mm, eluting with a gradient of 13-28%ACN (0.1%FA in water (0.1%FA) over 18 minutes; stop at 25 minutes; flow rate: 30 mL/minute, t (R) 9.0 min.
- the material is concentrated, dissolved in water, and lyophilized to give the title compound (87.0 mg, 51.8%) as a white solid with a trans ratio of about 94%.
- the reaction mixture is concentrated in vacuo.
- the residue is purified by HPLC with the following conditions column: SunFire C 18 5 ⁇ , 30*100 mm, eluting with a gradient of 10-25%ACN (0.1%FA) in water (0.1%FA) over 10 minutes; stop at 17 minutes; flow rate: 30 mL/minute; t (R) 18.2 minutes.
- the material is concentrated, dissolved in water, and lyophilized to give the title compound (61.4 mg, 54.7%) as a white solid with a trans ratio of about 94%.
- the purpose of this assay is to determine the effect of ASK1 inhibitors on the production of ADP by ASK1.
- the recombinant human ASK 1 (hASK1) catalytic domain tagged with Glutathione S-transferase is used, and histidine-tagged full-length human MAP kinase kinase 6 (MKK6) and ATP are the substrate and cofactor, respectively.
- the assay is done using an ADP-Glo TM Kinase Assay Kit (Promega, Catalog #V9102) according to the manufacturer’s protocol with the following modifications. Briefly, hASK1 (0.25 nM) and MKK6 (300 nM) in a buffer (10 mM MOPS pH 7.0; 10 mM Mg-Acetate; 1 mM DTT; 0.025%NP-40; 0.05%BSA; 1.5%glycerol) are incubated with ASK1 inhibitors at varying concentrations ranging from 10.00 uM to 0.17 nM for 15 minutes, followed by incubation with ATP (100 uM) for 30 minutes at room temperature.
- ASK1 inhibitors at varying concentrations ranging from 10.00 uM to 0.17 nM for 15 minutes, followed by incubation with ATP (100 uM) for 30 minutes at room temperature.
- ADP-Glo TM Reagent is added to terminate the kinase reaction and deplete the remaining ATP.
- the Kinase Detection Reagent is then added to convert ADP to ATP.
- the newly synthesized ATP is measured using a luciferase/luciferin reaction, and the luminescence determined by Envision (PerkinElmer) .
- the luminescence intensities are analyzed by GeneData, and fit to a 4 parameter dose response-inhibitor logistics curve to determine IC 50 values, using the effects of 5- (4-cyclopropyl-1H-imidazol-1-yl) -2-fluoro-4-methyl-N- ⁇ 6- [4- (propan-2-yl) -4H-1, 2, 4-triazol-3-yl] pyridin-2-yl ⁇ benzamide as a standard and DMSO vehicle for 100%and 0%inhibition, respectively.
- This assay is to determine the effect of ASK1 inhibitors on H 2 O 2 -stimulated ASK1 autophosphorylation at Thr838 in HEK293 cells overexpressing human ASK1.
- HEK293 cells overexpressing human influenza hemagglutinin- (HA-) tagged full length human ASK1 are maintained in DMEM supplemented with 10%FBS at 37 °C and 5%CO 2 .
- the cells are plated in matrigel-coated 96-well plates (25,000 cells/well) and incubated overnight.
- the cells are treated with ASK1 inhibitors at varying concentrations ranging from 10.00 ⁇ M to 0.17 nM for 1 hour, followed by stimulation with 1 mM H 2 O 2 for 10 minutes.
- pASK1 is quantified by using an anti-HA and anti-pASK1 (Thr838) antibody pair customized by Cisbio, on Envision (PerkinElmer) with emission and excitation wavelengths of 620 and 665 nm, respectively.
- the ratios of fluorescence intensities at 665 nm and 620 nm are analyzed by GeneData, and fit to a 4 parameter dose response-inhibitor logistics curve to determine IC 50 values, using the effects of 5- (4-cyclopropyl-1H-imidazol-1-yl) -2-fluoro-4-methyl-N- ⁇ 6- [4- (propan-2-yl) -4H-1, 2, 4-triazol-3-yl] pyridin-2-yl ⁇ benzamide as a standard and DMSO vehicle as 100%and 0%inhibition, respectively.
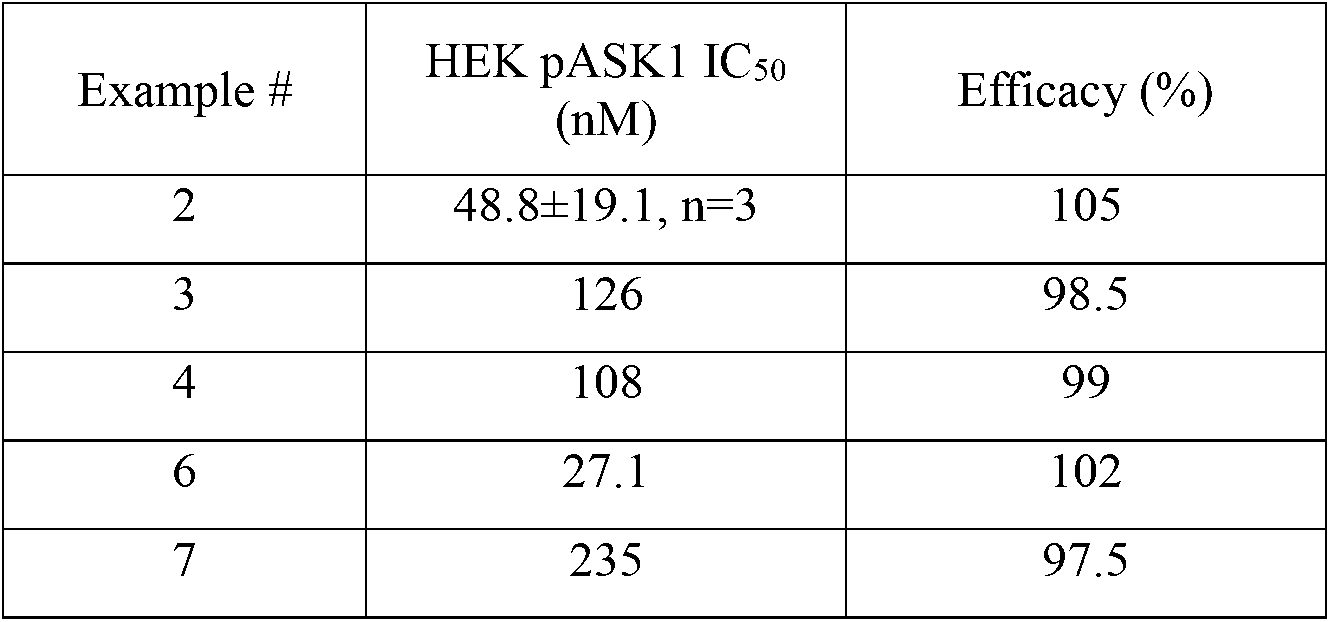
- Example compounds as shown in Table 2 above inhibit H 2 O 2 -stimulated ASK1 autophosphorylation at Thr838 in HEK293 cells.
Landscapes
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Virology (AREA)
- Gastroenterology & Hepatology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
A compound of the formula (I) wherein Q is selected from the group consisting of -C(CH 3) 2and (II); R is selected from the group consisting of (III) and (IV); or a pharmaceutically acceptable salt thereof; compositions, methods to treat liver disease and NASH.
Description
This invention provides cyclobutyl-imidazolidinone compounds or pharmaceutically acceptable salts thereof, and for use of the compounds in therapy. Cyclobutyl-Imidazolidinone compounds of this invention are inhibitors of apoptois signal-regulating kinase 1 (ASK1) .
ASK1 is a member of the large mitogen-activated protein kinase kinase kinase ( “MAP3K” ) family. ASK1 activation and signaling are associated with broad range of diseases. Compounds that inhibit ASK1 are desired for use in the treatment of ASK1 mediated conditions.
Compounds that inhibit ASK1 are desired for use in the treatment of Nonalcoholic steatohepatitis (NASH) . Nonalcoholic steatohepatitis is a liver disease with an etiological constellation characterized by macrovesicular hepatic steatosis, inflammation hepatocyte ballooning and fibrosis. Currently, there is no approved pharmaceutical medicament specifically for the treatment of nonalcoholic steatohepatitis. There is a need for pharmaceutical medicaments to offer additional treatment options for patients suffering from nonalcoholic steatohepatitis.
U.S. Patent No. 8,742,126 discloses 5- (4-cyclopropyl-1H-imidazol-l-yl) -N- (6- (4-isopropyl-4H-1, 2, 4-triazol-3-y-1) pyridin-2-yl) -2-fluoro-4-methylbenzamide as an ASK1 inhibitor.
U.S. Patent Application Publication No. US 2015/0342943 discloses a method of preventing and/or treating liver disease using an ASK1 inhibitor.
There is a need for compounds that have ASK1 inhibitory activity.
The present invention provides a compound of Formula I
wherein
R is selected from the group consisting of
a pharmaceutically acceptable salt thereof.
Q is-C (CH3) 2.
In an embodiment the compound of Formula I is 1- [3- (4-cyclopropylimidazol-1-yl) cyclobutyl] -3 - [6- (4-isopropyl-1, 2, 4-triazol-3 -yl) -2-pyridyl] imidazolidin-2-one, or a pharmaceutically acceptable salt thereof.
In an embodiment the compound of Formula I is 1- (6- (4-isopropyl-4H-1, 2, 4-triazol-3-yl) pyridin-2-yl) -3- (3- (6-methylpyridin-3-yl) cyclobutyl) imidazolidin-2-one, or a pharmaceutically acceptable salt thereof.
The present invention provides a pharmaceutical composition comprising a compound of Formula I, or a pharmaceutically acceptable salt thereof, together with one or more pharmaceutically acceptable carriers, diluents, or excipients.
The present invention provides a method for treating a condition mediated by ASK1 activity comprising administering to the mammal in need of treatment, an effective amount of a compound of Formula I, or a pharmaceutically acceptable salt thereof. The present invention also provides a method for treating liver disease, comprising administering to a mammal in need thereof, an effective amount of a compound of
Formula I, or a pharmaceutically acceptable salt thereof. The present invention provides a method for treating nonalcoholic steatohepatitis (NASH) , comprising administering to a mammal in need thereof, an effective amount of a compound of Formula I, or a pharmaceutically acceptable salt thereof.
In another embodiment, the present invention provides a compound of Formula I, or a pharmaceutically acceptable salt thereof, for use in therapy. Further, provided is a compound of the present invention, or a pharmaceutically acceptable salt thereof or pharmaceutical composition, for use in the treatment of liver disease. Further, provided is a compound of the present invention, or a pharmaceutically acceptable salt thereof or pharmaceutical composition, for use in the treatment of NASH.
In another embodiment, provided is a compound of Formula I, or a pharmaceutically acceptable salt thereof, for use in the manufacture of a medicament for the treatment of liver disease. Preferably, the medicament is for the treatment of NASH.
A compound of the present invention is preferably formulated as pharmaceutical compositions administered by any route which makes the compound bioavailable. Most preferably, such compositions are for oral administration. Such pharmaceutical compositions and processes for preparing same are well known in the art. See, e.g., Remington: The Science and Practice of Pharmacy (L.V. Allen, Editor, 22nd Edition, Pharmaceutical Press, 2012) .
Compounds of the present invention can be provided as a pharmaceutically acceptable salt. “Pharmaceutically-acceptable salt” refers to salts of the compound of the invention considered to be acceptable for clinical and/or veterinary use. Pharmaceutically acceptable salts and common methodology for preparing them are well known in the art. See, e.g., P. Stahl, et al., Handbook of Pharmaceutical Salts: Properties, Selection and Use, (VCHA/Wiley-VCH, 2002)
A human is a preferred mammal. As used herein, “patient” refers to a mammal in need of treatment. As used herein, the term “effective amount” or “therapeutically effective amount” of a compound refers to an amount, or a dosage, which is effective in treating a disorder or a disease, such as NASH, chronic kidney disease, or diabetic nephropathy as described herein. The attending diagnostician, as one skilled in the art, can readily determine an effective amount by the use of conventional techniques and by
observing results obtained under analogous circumstances. In determining the effective amount or dose of a compound, a number of factors are considered, including but not limited to, the compound to be administered; the co-administration of other agents, if used; the species of mammal; its size, age, and general health; the degree of involvement or the severity of the disorder; the response of the individual patient; the mode of administration; the bioavailability characteristics of the preparation administered; the dose regimen selected; the use of other concomitant medication; and other relevant circumstances.
The pharmaceutical composition is administered to a patient in amounts effective to treat liver disease, more particularly, NASH. An appropriate amount or dose effective to treat a patient can be determined by a health care provider.
The terms “treatment” and “treating” as used herein are intended to refer to all processes wherein there may be a slowing, interrupting, arresting, controlling, or stopping of the progression of an existing disorder and/or symptoms thereof, but does not necessarily indicate a total elimination of all symptoms.
The term “liver disease” as used herein embraces liver conditions or symptoms associated with ASK1 mediation, for example, metabolic liver disease, steatosis, liver fibrosis, primary sclerosing cholangitis (PSC) , cirrhosis, liver fibrosis, non-alcoholic fatty liver disease (NAFLD) , nonalcoholic steatohepatitis (NASH) , hepatic ischemia reperfusion injury, and primary biliary cirrhosis (PBC) .
The term “pharmaceutically acceptable carrier, diluent, or excipients” means that the carrier, diluent, and excipients are pharmaceutically compatible with the other ingredients of the composition. In a particular embodiment, the pharmaceutical compositions are formulated as a tablet or capsule for oral administration. The tablet or capsule can include a compound of the present invention in an amount effective to treat liver disease, particularly NASH.
The abbreviations used herein are defined according to Aldrichimica Acta, Vol. 17, No. 1, 1984. Other abbreviations are defined as follows: “ACN” refers to acetonitrile; “ADP” refers to adenosine diphosphate; “AIBN” refers to azobisisobutyronitrile; “ATP” refers to adenosine triphosphate; “boc” refers to tert-butoxycarbonyl; “BSA “refers to Bovine Serum Albumin; ; “DCM” refers to
dichloromethane; “DMEDA” refers to N, N’ -dimethylethylenediamine; “DMEM” refers to Dulbecco’s Modified Eagle’s Medium; “DMF” refers to N, N-dimethylformamide; “DMP” refers to 1, 1, 1 -triacetoxy-1, 1 -dihydro-1, 2-benziodoxol-3 (1H) -one, also known as Dess-Martin periodinane; “DMSO” refers to dimethylsulfoxide; “DTT” refers to dithiothreitol; “EtOH” refers to ethanonl or ethyl alcohol; “FA” refers to formic acid; “FBS” refers to Fetal Bovine Serum; “HEK” refers to human embryonic kidney; “HPLC” refers to high performance liquid chromatography; “IC50” refers to the concentration of an agent that produces 50%of the maximal inhibitory response possible for that agent; IPA” refers to isopropanol or isopropyl alcohol; “MAP” refers to mitogen-activated protein; “MeOH” refers to methanol or methyl alcohol; “MOPS” refers to (3- (N-morpholino) propanesulfonic acid) ; “NBS” refers to N-bromosuccinimide; “NIS” refers to N-iodosuccinimide; “NP-40 refers to Tergitol-type NP-40 which is nonyl phenoxypolyethoxylethanoll; “pASK1” refers to phosphorylated ASK1; “Pd2 (dba) 3” refers to tris (dibenzylideneacetone) dipalladium (0) ; “PE” refers to petroleum ether; “SFC” refers to supercritical fluid chromatography; “TBAF” refers to tetra-n-butylammonium fluoride; “TFA” refers to trifluoroacetic acid; “THF” refers to tetrahydrofuran; “TMSCN” refers to trimethylsilyl cyanide; “t (R) ” refers to retention time; XantPhos refers to 4, 5-bis (diphenylphosphino) -9, 9-dimethylxanthene; .
The intermediates described in the following preparations may contain a number of nitrogen, hydroxy, and acid protecting groups such as esters. The variable protecting group may be the same or different in each occurrence depending on the particular reaction conditions and the particular transformations to be performed. The protection and deprotection conditions are well known to the skilled artisan and are described in the literature. See. e.g., Greene and Wuts, Protective Groups in Organic Synthesis, (T. Greene and P. Wuts, eds., 2d ed. 1991) .
The compounds of the present invention, or salts thereof, may be prepared by a variety of procedures known in the art, some of which are illustrated in the Preparations and Examples below. The specific synthetic steps for each of the routes described may be combined in different ways, to prepare compounds of the invention, or salts thereof. The products of each step can be recovered by conventional methods well known in the art, including extraction, evaporation, precipitation, chromatography, filtration, trituration,
and crystallization. The reagents and starting materials are readily available to one of ordinary skill in the art. Others may be made by standard techniques of organic and heterocyclic chemistry which are analogous to the syntheses of known structurally-similar compounds and the procedures described in the Preparations and Examples which follow including any novel procedures.
Preparations and Examples
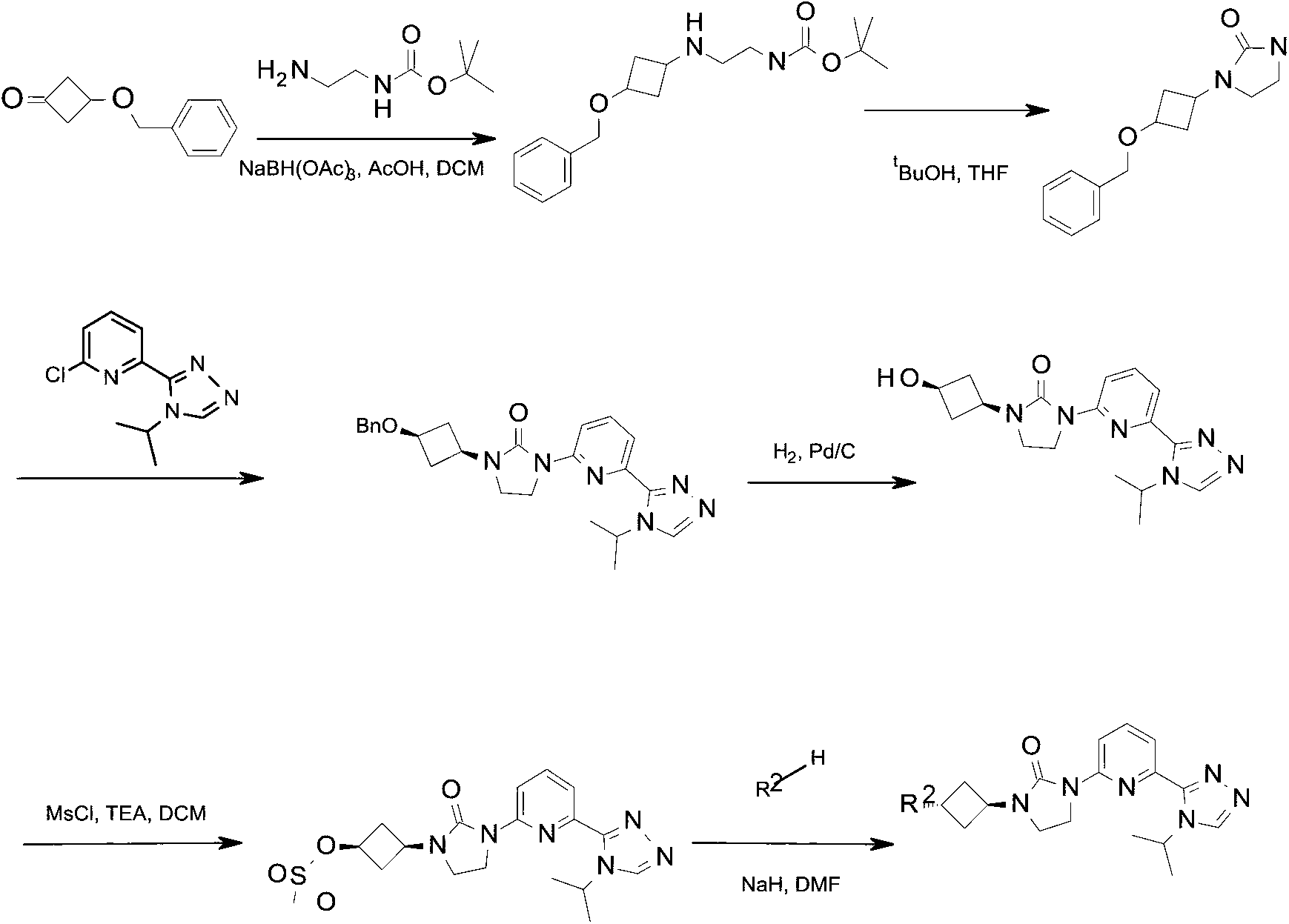
Scheme 1
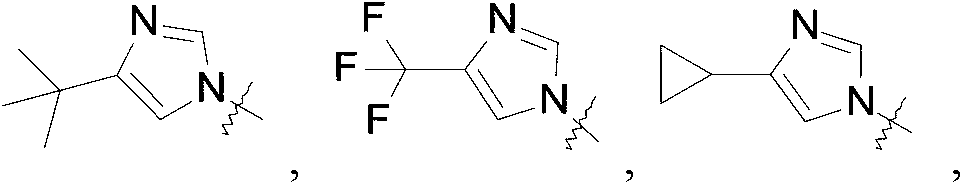
Compounds of this invention may be prepared as generally illustrated by Scheme 1 and using methods substantially as described by the Examples, when R2 is selected from the group consisting of
Preparation 1
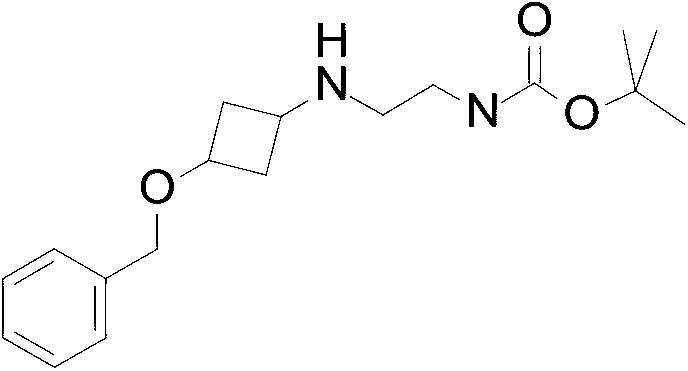
tert-Butyl N- [2- [ (3-benzyloxycyclobutyl) amino] ethyl] carbamate
A solution of 3- (benzyloxy) cyclobutanone (4.0 g, 22.0 mmol) , N-boc-ethylenediamine (7.80 g, 48.7 mmol) , DCM (50 mL) and acetic acid (1.0 mL) are stirred at room temperature for 30 minutes. The residue is purified by silica gel flash chromatography, eluting with a gradient of 0%to 5%MeOH in DCM to give the title compound (2.8 g, 41.0%) as a yellow oil. ES/MS (m/z) : 321.3 (M+1) .
Preparation 2
tert-Butyl N- [2- [ (3-benzyloxycyclobutyl) amino] ethyl] carbamate
Potassium tert-butoxide (3.0 g, 26 mmol) is added a solution of tert-butyl N- [2-[ (3-benzyloxycyclobutyl) amino] ethyl] carbamate (2.8 g, 8.7 mmol) in THF (200 mL) at room temperature under N2. The mixture is stirred at 60 ℃ for 2 hours. The reaction is quenched by addition of water and the product is extracted with DCM (3× 150 mL) . The organic extracts are dried over Na2SO4, filtered, and concentrated in vacuo. The residue is purified by silica gel flash chromatography eluting with a gradient of 0%to 4%MeOH in DCM to give the title compound (1.9 g, 88.0%) as a white solid. ES/MS (m/z) : 247.3 (M+1) .
Preparation 3
N- (3-Benzyloxycyclobutyl) -6- (4-isopropyl-1, 2, 4-triazol-3-yl) pyridin-2-amine
Cs2CO3 (4.0 g, 12.3 mmol) is added to a solution of2-chloro-6- (4-isopropyl-1, 2, 4-triazol-3-yl) pyridine (0.95 g, 4.1 mmol) and tert-butyl N- [2- [ (3-benzyloxycyclobutyl) amino] ethyl] carbamate (1.0 g, 4.1 mmol) in 1, 4-dioxane (20 mL) . The mixture is degassed with a stream of N2 for 5 minutes. XantPhos (0.48 g, 0.81 mmol) and Pd2 (dba) 3 (0.38 g, 0.41 mmol) are added sequentially and the resulting mixture is stirred at 130 ℃ for 4 hours under N2. The mixture is diluted with DCM (2×75mL) and washed sequentially with saturated brine (25 mL) . The organic extracts are dried over Na2SO4, filtered, and evaporated to dryness. The crude material is purified by silica gel flash chromatography, eluting with a gradient of 0%to 50%EtOAc in hexanes to give the title compound (1.69 g, 96.0%) as a white solid. ES/MS (m/z) : 433.3 (M+1) .
Preparation 4
1- (3-Hydroxycyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one; hydrochloride
N- (3-Benzyloxycyclobutyl) -6- (4-isopropyl-1, 2, 4-triazol-3-yl) pyridin-2-amine (9.0 g, 20.81 mmol) , palladium (5 mass%) in Lindlar catalyst (3.0 g, 2.8 mmol) , MeOH (150 mL) and hydrochloric acid (32 mass%) in H2O (1.5 mL) are combined. The mixture is degassed with H2 and stirred under balloon pressure of H2 at room temperature for 12
hours. The mixture is filtered and concentrated to give the title compound (7.5 g, 90.4%) as a yellow solid, which is used without further purification. LC/MS (m/z) : 343.3 (M+1-HCl) .
Preparation 5
[3- [3- [6- (4-Isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -2-oxo-imidazolidin-1-yl] cyclobutyl] methanesulfonate
Methansulfonyl chloride (0.747 g, 6.45 mmol) is added to a solution of 1- (3-hydroxycyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one (1.227 g, 3.23 mmol) and trimethylamine (0.979 g, 9.68 mmol) in DCM (20 mL) at 0 ℃. The mixture is stirred at 0 ℃ for 15 minutes. The reaction is quenched with aq. NaHCO3 (30 mL) and the product is extracted with DCM (2×50 mL) . The organic extracts are dried over Na2SO4 and concentrated in vacuo. The residue is purified by silica gel flash chromatography, eluting with a gradient of 0%to 3%MeOH in DCM to give the title compound (1.224 g, 81.21%) as a white solid with a cis ratio of about 94%. ES/MS (m/z) : 421.2 (M+1) .
Preparation 6
1- [6- (4-Isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -3- (3-oxocyclobutyl) imidazolidin-2-one
DMP (6.9 g, 16.0 mmol) is added to a solution of 1- (3-hydroxycyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one; hydrochloride (3.9 g, 11.0 mmol) in DCM (10 mL) at room temperature. The mixture is stirred at room temperature
for 20 hours. The reaction is quenched with a sat. Na2SO3 and NaHCO3 (30 mL) solution and the product is extracted with DCM (3×50 mL) . The organic extracts are dried over Na2SO4 and concentrated in vacuo. The residue is purified by silica gel flash chromatography, eluting with a gradient of 0%to 2%MeOH in DCM to give the title compound (2.5 g, 64.0%) as a white solid. ES/MS (m/z) : 341.3 (M+1) .
Preparation 7
1- (3-Hydroxycyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one
Sodium borohydride (249.0 mg, 6.45 mmol) is added to a solution of 1- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -3- (3-oxocyclobutyl) imidazolidin-2-one (1.22 g, 3.226 mmol) in MeOH (10 mL) at 0 ℃. The mixture is stirred at 0 ℃ for 30 minutes. The reaction is quenched with aq. NaHCO3 (10 mL) solution and the product is extracted with DCM (3×50 mL) . The organic extracts are dried over Na2SO4 and concentrated in vacuo to give the title compound (1.227 g, 100%) as a white solid with a cis ratio of about 94%. ES/MS (m/z) : 343.2 (M+1) .
Scheme 2
Compounds of this invention may be prepared as generally illustrated by Scheme 2 and using methods substantially described by the Examples, when R3 is selected from the group consisting of
Preparation 8
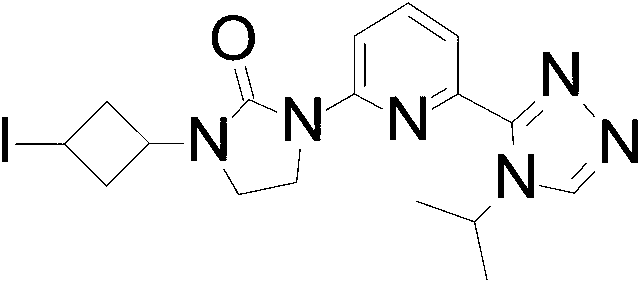
1- (3-Iodocyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one
Sodium iodide (3.986 g, 26.59 mmol) is added to a solution of [3- [3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -2-oxo-imidazolidin-1-yl] cyclobutyl] methanesulfonate (1.177 g, 2.66 mmol) in acetone (10 mL) at room temperature. The mixture is stirred at 80 ℃ for 6 hrs. The reaction is concentrated, the solid is filtered, and the filter is washed with DCM. The filtrate is concentrated in vacuo. The filtrate is
purified by silica gel flash chromatography, eluting with a gradient of 0%to 3%MeOH in DCM to give the title compound (1.175 g, 92.81%) as a yellow oil with a cis/trans ratio of about 1∶2. ES/MS (m/z) : 452.8 (M+1) .
Example 1
1- [3- (4-Cyclopropylimidazol-1-yl) cyclobutyl] -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one
Sodium hydride (60 mass%) in mineral oil (1.5 g, 39.0 mmol) is added to a solution of 4-cyclopropyl-lH-imidazole (2.8 g, 26.0 mmol) in DMF (50 mL) at room temperature. The mixture is stirred at room temperature for 30 minutes. [3- [3- [6- (4-Isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -2-oxo-imidazolidin-1-yl] cyclobutyl] methanesulfonate (5.7 g, 13 mmol) is added to the solution and the mixture is heated to 80 ℃ and stirred for 17 hours. The reaction is quenched with water (80 mL) and the product is extracted with DCM (3× 150 mL) . The organic extracts are washed with brine (30 mL) , dried over Na2SO4, and concentrated in vacuo. The residue is purified by silica gel flash chromatography, eluting with a gradient of 0%to 4%MeOH in DCM followed by Preparative HPLC eluting with an isocratic system of 23 %ACN (0.5%FA) in H2O (0.1%FA) for 41 min; column temperature: room temperature; flow rate: 30 mL/min, t (R) = 27.1 minutes (UV) . The material is concentrated, dissolved in water, and lyophilized to give the title compound impure with the cis isomer in about a 1∶2 ratio of cis and trans (2.7 g, 46.0%) as a white solid. ES/MS (m/z) : 433.3 (M+1) .
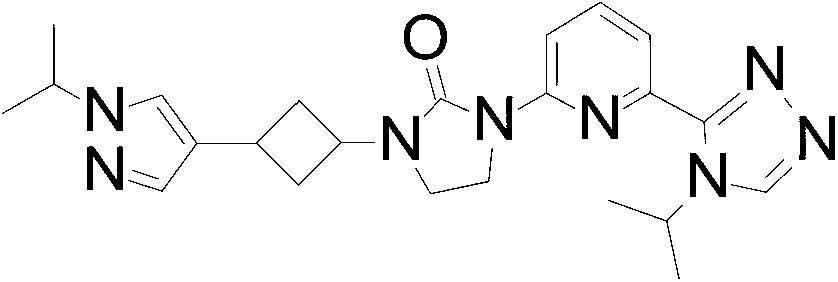
Example 2
1- (3- (4-cyclopropyl-1H-imidazol-1-yl) cyclobutyl) -3- (6- (4-isopropyl-4H-1, 2, 4-triazol-3 -yl) pyridin-2-yl) imidazolidin-2-one
The mixture of 1- [3- (4-cyclopropylimidazol-1-yl) cyclobutyl] -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one (5.60 g, 12.9 mmol) is separated by SFC with the following conditions: column: OJ (250 mm*30 mm, 5 μm) eluting with 80%0.1%NH3 in H2O and 20%EtOH to give the title compound (3.01 g, 53.3%) as a white solid. ES/MS (m/z) : 433.2 (M+1) , LCMS: (Xtimate C18 2.1*30 mm) ; eluting with a gradient of 10%-80%ACN (0.5%TFA) in H2O (0.5%TFA) 4 minute chromatography t (R) = 1.30 min, 1H NMR (400 MHz, CDCl3) δ 8.39 -8.26 (m, 2H) , 7.91 (d, J=7.2 Hz, 1H) , 7.79 (t, J=8.0 Hz, 1H) , 7.43 (s, 1H) , 6.81 (s, 1H) , 5.61-5.47 (m, 1H) , 4.82-4.62 (m, 2H) , 4.07 (t, J=7.6 Hz, 2H) , 3.64 (t, J=8.0 Hz, 2H) , 3.02-2.84 (m, 2H) , 2.74-2.58 (m, 2H) , 1.85-1.80 (m, 1H) , 1.56 (d, J=6.8 Hz, 6H) , 0.88-0.80 (m, 2H) , 0.79-0.71 (m, 2H) .
Example 3
1- (6- (4-Isopropyl-4H-1, 2, 4-triazol-3-yl) pyridin-2-yl) -3 - (3- (4- (trifluoromethyl) -1H-imidazol-1-yl) cyclobutyl) imidazolidin-2-one
Sodium hydride (60 mass%) in mineral oil (41.6 mg, 1.04 mmol) is added to a solution of 4- (trifluoromethyl) -1H-imidazole (96.3 mg, 0.694 mmol) in DMF (5.0 mL) at 0 ℃. The mixture is stirred at room temperature for 30 minutes. [3- [3- [6- (4-Isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -2-oxo-imidazolidin-1-yl] cyclobutyl] methanesulfonate (162.0 mg, 0.347 mmol) is added at room temperature, the mixture is heated to 90 ℃, and stirred for 17 hours. The reaction is quenched with water (20 mL) and the product is
extracted with DCM (3×40 mL) . The organic extracts are dried over Na2SO4 and concentrated in vacuo. The residue is purified by HPLC under the following conditions column: SunFire C18 5 μ, 30*100 mm, eluting with a gradient of 28%-43%of ACN (0.1%FA) in H2O (0.1%FA) over 10 minutes and stop at 17 minutes; flow rate: 30 mL/minutes. t (R) 7.5 minutes. The material is concentrated, dissolved in water, and lyophilized to give the title compound (87.0 g, 51.8%) as a white solid and a trans ratio of about 94%. ES/MS (m/z) : 461.0 (M+1) , 1H NMR (500 MHz, CDCl3) δ 8.36 (s, 1H) , 8.33 (d, J= 8.3 Hz, 1H) , 7.93 (d, J= 7.5 Hz, 1H) , 7.81 (t, J= 8.0 Hz, 1H) , 7.65 (s, 1H) , 7.43 (s, 1H) , 5.58-5.52 (m, 1H) , 4.90-4.85 (m, 1H) , 4.72-4.69 (m, 1H) , 4.09 (t, J= 8.0 Hz, 2H) , 3.65 (t, J= 8.0 Hz, 2H) , 3.11-3.05 (m, 2H) , 2.76-2.70 (m, 2H) , 1.57 (d, J= 7.0 Hz, 6H) .
Example 4
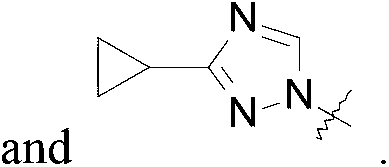
1 - [3- (3-Cyclopropyl-1, 2, 4-triazol-1 -yl) cyclobutyl] -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one
Sodium hydride (60 mass%) in mineral oil (41.6 mg, 1.04 mmol) is added to a solution of 3-cyclopropyl-1H-1, 2, 4-triazole (79.7 mg, 0.694 mmol) in DMF (5.0 mL) at 0 ℃. The mixture is stirred at room temperature for 30 minutes. [3- [3- [6- (4-Isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -2-oxo-imidazolidin-1-yl] cyclobutyl] methanesulfonate (162.0 mg, 0.347 mmol) is added at room temperature, the mixture is heated to 90 ℃, and stirred for 17 hours. The reaction is quenched with water (20 mL) and the product is extracted with DCM (3×40 mL) . The organic extracts are dried over Na2SO4 and concentrated in vacuo. The residue is purified by HPLC with the following conditions: column: SunFire C18 5 μ, 30*100 mm, eluting with a gradient of 13-28%ACN (0.1%FA in water (0.1%FA) over 18 minutes; stop at 25 minutes; flow rate: 30 mL/minute, t (R) 9.0 min. The material is concentrated, dissolved in water, and lyophilized to give the title compound (87.0 mg, 51.8%) as a white solid with a trans ratio of about 94%. ES/MS
(m/z) : 434.0 (M+i) , 1H NMR (500 MHz, CDCl3) δ 8.20-8.19 (m, 1H) , 8.18 (s, 1H) , 7.80 (s, 1H) , 7.76 (d, J= 7.5 Hz, 1H) , 7.64 (t, J= 8.0 Hz, 1H) , 5.45-5.38 (m, 1H) , 4.76-4.67 (m, 2H) , 3.92 (t, J= 8.0 Hz, 2H) , 3.49 (t, J= 8.0 Hz, 2H) , 2.86-2.80 (m, 2H) , 2.76-2.68 (m, 2H) , 1.96-1.90 (m, 1H) , 1.41 (d, J= 7.0 Hz, 6H) , 0.84-0.82 (m, 4H) .
Example 5
1- (3- (4- (tert-Butyl) -1H-imidazol-1-yl) cyclobutyl) -3- (6- (4-isopropyl-4H-1, 2, 4-triazol-3-yl) pyridin-2-yl) imidazolidin-2-one
Sodium hydride (60 mass%) in mineral oil (28.5 mg, 0.713 mmol) is added to a solution of 4-tert-butyl-1H-imidazole (59.1 mg, 0.476 mmol) in DMF (10.0 mL) at 0 ℃. The mixture is stirred at room temperature for 30 minutes. [3- [3- [6- (4-Isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -2-oxo-imidazolidin-1 -yl] cyclobutyl] methanesulfonate (100.0 mg, 0.238 mmol) is added at room temperature, the mixture is heated to 80 ℃, and stirred for 2 hours. The reaction mixture is concentrated in vacuo. The residue is purified by HPLC with the following conditions column: SunFire C 18 5 μ, 30*100 mm, eluting with a gradient of 10-25%ACN (0.1%FA) in water (0.1%FA) over 10 minutes; stop at 17 minutes; flow rate: 30 mL/minute; t (R) 18.2 minutes. The material is concentrated, dissolved in water, and lyophilized to give the title compound (61.4 mg, 54.7%) as a white solid with a trans ratio of about 94%. ES/MS (m/z) : 449.4 (M+1) , 1H NMR (500 MHz, CD3OD) δ 8.87 (s, 1H) , 8.37 (d, J= 7.5 Hz, 1H) , 8.23 (s, 1H) , 7.92 (t, J= 7.5 Hz, 1H) , 7.76 (d, J= 7.5 Hz, 1H) , 7.27 (s, 1H) , 5.62-5.57 (m, 1H) , 4.96-4.93 (m, 1H) , 4.82-4.76 (m, 1H) , 4.18 (t, J= 8.0 Hz, 2H) , 3.79 (t, J= 8.0 Hz, 2H) , 3.07-3.01 (m, 2H) , 2.91-2.77 (m, 2H) , 1.63-1.61 (m, 6H) , 1.35 (s, 9H) .
Example 6
1- [3- (1-Isopropylpyrazol-4-yl) cyclobutyl] -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one
Isopropylmagnesium chloride-lithium chloride complex (1.3 mol/L) in THF (2.6 mL, 3.36 mmol is added to a solution of 4-bromo-1-isopropyl-pyrazole (327.5 mg, 1.680 mmol) in THF (2.0 mL) at 0 ℃ under N2. The mixture is stirred at room temperature for 4 hours. The mixture is added dropwise to a solution of 1- (3-iodocyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one (200.0 mg, 0.42 mmol) , ferric acetylacetonate (156.2 mg, 0.42 mmol) and N, N, N′, N′-tetramethylethylenediamine (0.16 mL, 1.05 mmol) in THF (2.0 mL) at 0 ℃. The mixture is stirred at room temperature for 1 hour. The reaction is quenched with water (20 mL) and the product is extracted with DCM (3×40 mL) . The organic extracts are dried over Na2SO4. concentrated in vacuo, and the residue is purified by HPLC with the following conditions: column: SunFire C18 5 μ, 30*100 mm, eluting with a gradient of 23%-38%ACN (0.1%FA) in water (0.1%FA) over 18 minutes; stop at 25 minutes; flow rate: 30 mL/minute; t (R) 17.2 minutes. The material is concentrated, dissolved in water, and lyophilized to give the title compound (12.0 mg, 6.25%) as a white solid. ES/MS (m/z) : 435.0 (M+1) , 1H NMR (500 MHz, CDCl3) δ 8.36-8.34 (m, 2H) , 7.89 (d, J= 7.5 Hz, 1H) , 7.78 (t, J= 7.5 Hz, 1H) , 7.43 (s, 1H) , 7.35 (s, 1H) , 5.61-5.56 (m, 1H) , 4.86-4.79 (m, 1H) , 4.51-4.44 (m, 1H) , 4.06 (t, J=8.0 Hz, 2H) , 3.66 (t, J= 8.0 Hz, 2H) , 3.49-3.32 (m, 1H) , 2.76-2.67 (m, 2H) , 2.31-2.21 (m, 2H) , 1.57 (d, J= 7.0 Hz, 6H) , 1.52 (d, J= 7.0 Hz, 6H) .
Example 7
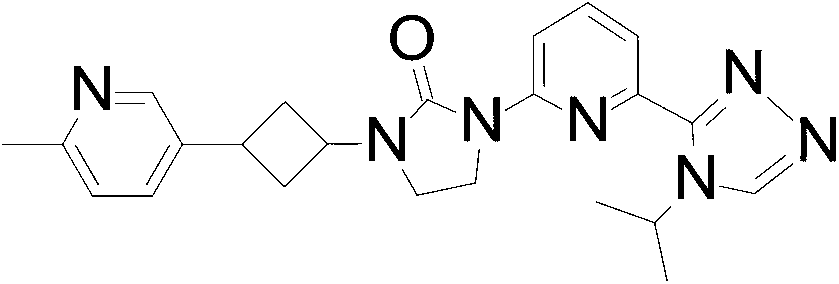
1- [6- (4-Isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -3- [3- (6-methyl-3-pyridyl) cyclobutyl] imidazolidin-2-one
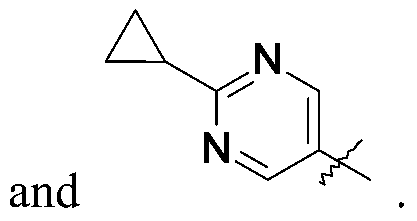
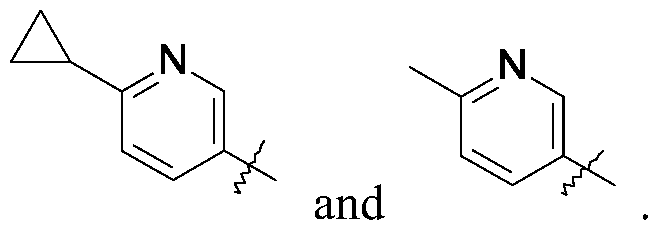
Isopropylmagnesium chloride-lithium chloride complex (1.3 mol/L) in THF (1.9 mL, 2.52 mmol) is added to a solution of 5-bromo-2-methyl-pyridine (223.5 mg, 1.260 mmol) in THF (5.0 mL) at 0 ℃ under N2. The mixture is stirred at room temperature for 4.0 hours. The mixture is added dropwise to the solution of 1- (3-iodocyclobutyl) -3- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] imidazolidin-2-one (200.0 mg, 0.42 mmol) , ferric acetylacetonate (31.2 mg, 0.084 mmol) and N, N, N′, N′-tetramethylethylene diamine (0.16 mL, 1.05 mmol) in THF (5.0 mL) at 0 ℃. The mixture is stirred at room temperature for 1 hour. The reaction is quenched with water (20 mL) and the product is extracted with DCM (3×40 mL) . The organic extracts are dried over Na2SO4, concentrated in vacuo, and purified by silica gel flash chromatography, eluting with a gradient of 0%to 6%MeOH in DCM to give the title compound (60.0 mg, 32.5%) as a yellow oil with a cis and trans ratio is about 2∶1. ES/MS (m/z) : 418.1 (M+1) .
Example 8
1- (6- (4-Isopropyl-4H-1, 2, 4-triazol-3-yl) pyridin-2-yl) -3- (3- (6-methylpyridin-3-yl) cyclobutyl) imidazolidin-2-one
1- [6- (4-isopropyl-1, 2, 4-triazol-3 -yl) -2-pyridyl] -3 - [3 - (6-methyl-3 -pyridyl) cyclobutyl] imidazolidin-2-one (60.0 mg, 0.133 mmol) is separated by SFC: column: AD (250mm*30 mm, 5 μm) , eluting an isocratic system of 60%IPA and 40%in H2O (0.1%NH3) ; flow rate: 50 mL/minute to give the title compound (9.7 mg, 16.9%) as
a white solid. ES/MS (m/z) : 418.1 (M+1) , LCMS: column: (Xtimate C18 2.1*30 mm) ; eluting with a gradient of 10%to 80%IPA in H2O (0.1%NH3) over 4 minutes; t (R) =1.393 minutes, 1H NMR (400 MHz, CDCl3) δ 8.41 (s, 1H) , 8.37 -8.30 (m, 2H) , 7.88 (d, J=7.2 Hz, 1H) , 7.77 (d, J=8.0 Hz, 1H) , 7.59 (dd, J=2.4, 8.0 Hz, 1H) , 7.15 (d, J=8.0 Hz, 1H) , 5.63-5.50 (m, 1H) , 4.82-4.70 (m, 1H) , 4.07 (t, J=7.6 Hz, 2H) , 3.69 (t, J=8.4 Hz, 2H) , 3.56-3.45 (m, 1H) , 2.86-2.74 (m, 2H) , 2.54 (s, 3H) , 2.50-2.41 (m, 2H) , 1.56 (d, J=6.4 Hz, 6H) .
Biological Assays
ASK1 Inhibitor Effect Determined by ASK1 Enzymatic Assay
The purpose of this assay is to determine the effect of ASK1 inhibitors on the production of ADP by ASK1. The recombinant human ASK 1 (hASK1) catalytic domain tagged with Glutathione S-transferase is used, and histidine-tagged full-length human MAP kinase kinase 6 (MKK6) and ATP are the substrate and cofactor, respectively.
The assay is done using an ADP-GloTM Kinase Assay Kit (Promega, Catalog #V9102) according to the manufacturer’s protocol with the following modifications. Briefly, hASK1 (0.25 nM) and MKK6 (300 nM) in a buffer (10 mM MOPS pH 7.0; 10 mM Mg-Acetate; 1 mM DTT; 0.025%NP-40; 0.05%BSA; 1.5%glycerol) are incubated with ASK1 inhibitors at varying concentrations ranging from 10.00 uM to 0.17 nM for 15 minutes, followed by incubation with ATP (100 uM) for 30 minutes at room temperature. ADP-GloTM Reagent is added to terminate the kinase reaction and deplete the remaining ATP. The Kinase Detection Reagent is then added to convert ADP to ATP. The newly synthesized ATP is measured using a luciferase/luciferin reaction, and the luminescence determined by Envision (PerkinElmer) . The luminescence intensities are analyzed by GeneData, and fit to a 4 parameter dose response-inhibitor logistics curve to determine IC50 values, using the effects of 5- (4-cyclopropyl-1H-imidazol-1-yl) -2-fluoro-4-methyl-N- {6- [4- (propan-2-yl) -4H-1, 2, 4-triazol-3-yl] pyridin-2-yl} benzamide as a standard and DMSO vehicle for 100%and 0%inhibition, respectively.
Compounds of the Examples herein are tested essentially as described above and shown in Table 1.
Table 1
| Example# | hASK1 IC50 (nM) | Efficacy (%) |
| 2 | 1.47±0.60, n=14 | 100 |
| 3 | 2.80±0.97, n=6 | 100 |
| 4 | 2.56±0.68, n=4 | 100 |
| 5 | 1.84±0.64, n=4 | 100 |
| 6 | 2.17±0.45, n=2 | 100 |
| 7 | 5.26±0.72, n=4 | 100 |
Mean ± standard deviation
These results indicate that the Examples tested inhibit ASK1 enzymatic activity as shown in Table 1.
ASK1 Inhibitor Effect Determined by ASK1 Autophosphorylation (Thr838) Assay
The purpose of this assay is to determine the effect of ASK1 inhibitors on H2O2-stimulated ASK1 autophosphorylation at Thr838 in HEK293 cells overexpressing human ASK1.
HEK293 cells overexpressing human influenza hemagglutinin- (HA-) tagged full length human ASK1 are maintained in DMEM supplemented with 10%FBS at 37 ℃ and 5%CO2. For the assay, the cells are plated in matrigel-coated 96-well plates (25,000 cells/well) and incubated overnight. The cells are treated with ASK1 inhibitors at varying concentrations ranging from 10.00 μM to 0.17 nM for 1 hour, followed by stimulation with 1 mM H2O2 for 10 minutes. The cells are then lysed with Homogeneous Time-Resolved Fluorescencelysis buffer (Cisbio, Catalog #64KL1FDF) containing phosphatase inhibitors (ThermoFisher, Catalog #78430) . pASK1 is quantified by using an anti-HA and anti-pASK1 (Thr838) antibody pair customized by Cisbio, on Envision (PerkinElmer) with emission and excitation wavelengths of 620 and 665 nm, respectively. The ratios of fluorescence intensities at 665 nm and 620 nm are analyzed by
GeneData, and fit to a 4 parameter dose response-inhibitor logistics curve to determine IC50 values, using the effects of 5- (4-cyclopropyl-1H-imidazol-1-yl) -2-fluoro-4-methyl-N- {6- [4- (propan-2-yl) -4H-1, 2, 4-triazol-3-yl] pyridin-2-yl} benzamide as a standard and DMSO vehicle as 100%and 0%inhibition, respectively.
Compounds of the Examples herein are tested essentially as described and the results are shown in Table 2.
Table 2
Mean ± standard deviation
These results support that the Example compounds as shown in Table 2 above inhibit H2O2-stimulated ASK1 autophosphorylation at Thr838 in HEK293 cells.
Claims (13)
- A compound as claimed by Claim 1 wherein Q is -C (CH3) 2.
- A compound as claimed by Claim 1 wherein the compound is 1- [6- (4-isopropyl-1, 2, 4-triazol-3-yl) -2-pyridyl] -3- [3- (6-methyl-3-pyridyl) cyclobutyl] imidazolidin-2-one, or a pharmaceutically acceptable salt thereof.
- A compound as claimed by Claim 1 wherein the compound is 1- (3- (4-cyclopropyl-1H-imidazol-1-yl) cyclobutyl) -3- (6- (4-isopropyl-4H-1, 2, 4-triazol-3-yl) pyridin-2-yl) imidazolidin-2-one, or a pharmaceutically acceptable salt thereof.
- A compound as claimed by Claim 1 wherein the compound is 1- (3- (4- (tert-butyl) -1H-imidazol-1-yl) cyclobutyl) -3- (6- (4-isopropyl-4H-1, 2, 4-triazol-3-yl) pyridin-2-yl) imidazolidin-2-one, or a pharmaceutically acceptable salt thereof.
- A pharmaceutical composition comprising a compound as claimed by any one of Claims 1 to 9, or a pharmaceutically acceptable salt thereof, and at least one selected from the group consisting of a pharmaceutically acceptable carrier, diluent, and excipient.
- A method for treating nonalcoholic steatohepatitis (NASH) , comprising administering to a mammal in need thereof, an effective amount of a compound as claimed by any one of Claims 1 to 9, or a pharmaceutically acceptable salt thereof.
- A compound or a pharmaceutically acceptable salt thereof, as claimed by any one of Claims 1 to 9 for use in therapy.
- A compound, or a pharmaceutically acceptable salt thereof, as claimed by any one of Claims 1 to 9 for use in the treatment of NASH.
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2017/100858 WO2019047094A1 (en) | 2017-09-07 | 2017-09-07 | Cyclobutyl-imidazolidinone compounds |
| PCT/CN2018/100515 WO2019047686A1 (en) | 2017-09-07 | 2018-08-14 | Cyclobutyl-imidazolidinone compounds |
| ARP180102522A AR112791A1 (en) | 2017-09-07 | 2018-09-05 | CYCLOBUTYL-IMIDAZOLIDINONE COMPOUNDS |
| US16/123,916 US10370352B2 (en) | 2017-09-07 | 2018-09-06 | Cyclobutyl-imidazolidinone compounds |
| TW107131411A TWI692476B (en) | 2017-09-07 | 2018-09-07 | Cyclobutyl-imidazolidinone compounds |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2017/100858 WO2019047094A1 (en) | 2017-09-07 | 2017-09-07 | Cyclobutyl-imidazolidinone compounds |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019047094A1 true WO2019047094A1 (en) | 2019-03-14 |
Family
ID=65633337
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2017/100858 Ceased WO2019047094A1 (en) | 2017-09-07 | 2017-09-07 | Cyclobutyl-imidazolidinone compounds |
| PCT/CN2018/100515 Ceased WO2019047686A1 (en) | 2017-09-07 | 2018-08-14 | Cyclobutyl-imidazolidinone compounds |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2018/100515 Ceased WO2019047686A1 (en) | 2017-09-07 | 2018-08-14 | Cyclobutyl-imidazolidinone compounds |
Country Status (3)
| Country | Link |
|---|---|
| AR (1) | AR112791A1 (en) |
| TW (1) | TWI692476B (en) |
| WO (2) | WO2019047094A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002039987A2 (en) * | 2000-11-14 | 2002-05-23 | Neurosearch A/S | Use of malaria parasite anion channel blockers for treating malaria |
| WO2016049069A1 (en) * | 2014-09-24 | 2016-03-31 | Gilead Sciences, Inc. | Methods of treating liver disease |
| US20170173031A1 (en) * | 2012-12-21 | 2017-06-22 | Gilead Sciences, Inc. | Apoptosis signal-regulating kinase inhibitors |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102985418B (en) * | 2010-07-02 | 2015-09-09 | 吉利德科学股份有限公司 | Apoptosis signal-regulating kinase inhibitors |
| UY34573A (en) * | 2012-01-27 | 2013-06-28 | Gilead Sciences Inc | QUINASE INHIBITOR REGULATING THE APOPTOSIS SIGNAL |
| US20150342943A1 (en) * | 2014-06-03 | 2015-12-03 | Gilead Sciences, Inc. | Methods of treating liver disease |
-
2017
- 2017-09-07 WO PCT/CN2017/100858 patent/WO2019047094A1/en not_active Ceased
-
2018
- 2018-08-14 WO PCT/CN2018/100515 patent/WO2019047686A1/en not_active Ceased
- 2018-09-05 AR ARP180102522A patent/AR112791A1/en unknown
- 2018-09-07 TW TW107131411A patent/TWI692476B/en active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002039987A2 (en) * | 2000-11-14 | 2002-05-23 | Neurosearch A/S | Use of malaria parasite anion channel blockers for treating malaria |
| US20170173031A1 (en) * | 2012-12-21 | 2017-06-22 | Gilead Sciences, Inc. | Apoptosis signal-regulating kinase inhibitors |
| WO2016049069A1 (en) * | 2014-09-24 | 2016-03-31 | Gilead Sciences, Inc. | Methods of treating liver disease |
Also Published As
| Publication number | Publication date |
|---|---|
| TWI692476B (en) | 2020-05-01 |
| WO2019047686A1 (en) | 2019-03-14 |
| TW201920144A (en) | 2019-06-01 |
| AR112791A1 (en) | 2019-12-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3589625B1 (en) | Isoquinolin and naphthydrin compounds | |
| EP2637502B1 (en) | Mutant-selective egfr inhibitors and uses thereof | |
| JP5406725B2 (en) | Compounds useful as protein kinase inhibitors | |
| EP3679029B1 (en) | Imidazolidine compounds | |
| WO2019094920A1 (en) | Azepin-2-one derivatives as rsv inhibitors | |
| WO2003055890A1 (en) | Thienopyrimidine derivative compounds as inhibitors of prolylpeptidase, inducers of apoptosis and cancer treatment agents | |
| CN104125957A (en) | Substituted benzylpyrazoles | |
| AU2014228374A1 (en) | CDC7 inhibitors | |
| EP3596085A1 (en) | Deuterated analogs of mk2 inhibitors and uses thereof | |
| US20060142572A1 (en) | Inhibitors of ERK protein kinase and uses thereof | |
| US9000182B2 (en) | 2H-imidazol-4-amine compounds and their use as BACE inhibitors | |
| US20190241542A1 (en) | Substituted hydantoin and thiohydantoin derivatives as androgen receptor antagonists | |
| JP2008525461A (en) | Selective inhibitor of ERK protein kinase and use thereof | |
| US10370352B2 (en) | Cyclobutyl-imidazolidinone compounds | |
| KR20180083426A (en) | Bis-pyridazine compounds and uses thereof in the treatment of cancer | |
| JP2016539983A (en) | Alkylpyrazolylguanidine F1F0-ATPase inhibitor and therapeutic use thereof | |
| WO2019047094A1 (en) | Cyclobutyl-imidazolidinone compounds | |
| US10550125B2 (en) | Prodrugs of imidazotriazine compounds as CK2 inhibitors | |
| US20120214846A1 (en) | Hexahydrocyclopentyl[f]indazole pyridyl ethanols and derivatives thereof as selective glucocorticoid receptor modulators | |
| WO2019134082A1 (en) | Novel pyrimidine derivatives as mth1 inhibitors | |
| WO2025176178A1 (en) | Novel cdk12/13 covalent inhibitor having a fused ring structure substituent or pharmaceutical composition thereof and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17924284 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 17924284 Country of ref document: EP Kind code of ref document: A1 |