WO2019009674A1 - 아데노신 유도체를 포함하는 당뇨병성 신증 예방 및 치료용 약학적 조성물 - Google Patents
아데노신 유도체를 포함하는 당뇨병성 신증 예방 및 치료용 약학적 조성물 Download PDFInfo
- Publication number
- WO2019009674A1 WO2019009674A1 PCT/KR2018/007717 KR2018007717W WO2019009674A1 WO 2019009674 A1 WO2019009674 A1 WO 2019009674A1 KR 2018007717 W KR2018007717 W KR 2018007717W WO 2019009674 A1 WO2019009674 A1 WO 2019009674A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- diabetic nephropathy
- formula
- substituted
- unsubstituted
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/50—Pyridazines; Hydrogenated pyridazines
- A61K31/5025—Pyridazines; Hydrogenated pyridazines ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
- A61K31/52—Purines, e.g. adenine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
- A61K31/7076—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines containing purines, e.g. adenosine, adenylic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
- A61K47/38—Cellulose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4841—Filling excipients; Inactive ingredients
- A61K9/4866—Organic macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
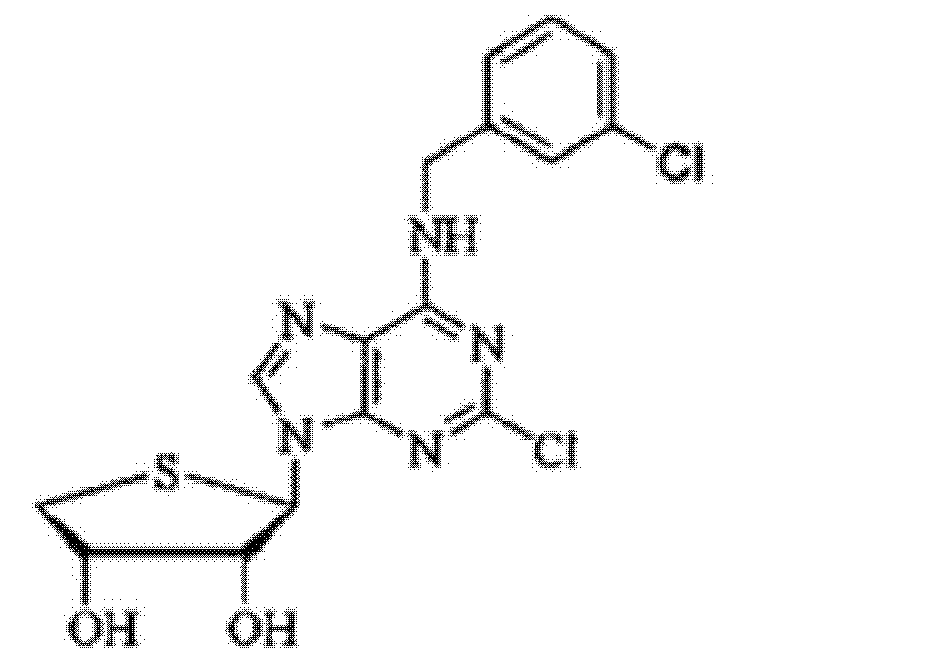
- Y is H or Cl.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nutrition Science (AREA)
- Physiology (AREA)
- Urology & Nephrology (AREA)
- Molecular Biology (AREA)
- Diabetes (AREA)
- Inorganic Chemistry (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
Claims (9)
- 하기 화학식 1로 표시되는 화합물 또는 이의 약학적으로 허용가능한 염을 유효성분으로 포함하는 당뇨병성 신증(diabetic nephropathy) 예방 또는 치료용 약학적 조성물.[화학식 1](상기 식에서,A는 O 또는 S이고,R은 비치환되거나, 독립적으로 또는 선택적으로 1 또는 2 이상의 C6~C10의아릴로 치환된 직쇄 또는 측쇄의 C1~C5의 알킬, 비치환 되거나 독립적으로 또는 선택적으로 할로겐 및 직쇄 또는 측쇄의 C1~C4의 알콕시가 1 또는 2 이상으로 치환된 벤질 또는 히드록시카보닐로 치환된 벤질이며,Y는 H 또는 할로겐 원소이다.)
- 제 1 항에 있어서, 상기 당뇨병성 신증은 제1형 당뇨병 및 제2형 당뇨병 중 하나 이상에 의한 것인 당뇨병성 신증 예방 또는 치료용 약학적 조성물.
- 제 4 항에 있어서, 상기 당뇨병성 신증은 제1형 당뇨병 및 제2형 당뇨병 중 하나 이상에 의한 것인 당뇨병성 신증 예방 또는 치료용 경구 투여제.
- 제 4 항에 있어서, 상기 경구 투여제는 메틸 셀룰로오스(Methyl cellulose, MC), 디메틸설폭사이드(Dimethyl sulfoxide, DMSO), 폴리에틸렌글리콜(Polyethylene glycol, PEG) 및 증류수로 이루어진 군으로부터 선택되는 하나 이상의 부형제를 더 포함하는 당뇨병성 신증 예방 또는 치료용 경구 투여제.
- 제 6 항에 있어서, 상기 경구 투여제는 부형제로서 0.5 wt% 메틸 셀룰로오스를 포함하는 당뇨병성 신증 예방 또는 치료용 경구 투여제.
- 제 4 항에 있어서, 상기 화학식 1로 표시되는 화합물 또는 이의 약학적으로 허용가능한 염은 분말 상태로 캡슐 내에 충진되는 당뇨병성 신증 예방 또는 치료용 경구 투여제.
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA3069118A CA3069118C (en) | 2017-07-07 | 2018-07-06 | Pharmaceutical composition for preventing and treating diabetic nephropaphy including adenosine derivative |
| CN201880045444.3A CN110869030B (zh) | 2017-07-07 | 2018-07-06 | 用于预防及治疗糖尿病性肾病的包含腺苷衍生物的药学组合物 |
| EP18827831.1A EP3650029B1 (en) | 2017-07-07 | 2018-07-06 | Pharmaceutical composition comprising an adenosine derivative for use in the prevention or treatment of diabetic nephropathy |
| EP23178716.9A EP4234021B1 (en) | 2017-07-07 | 2018-07-06 | Pharmaceutical composition comprising an adenosine derivative for use in the prevention or treatment of diabetic nephropathy |
| JP2020500619A JP6965429B2 (ja) | 2017-07-07 | 2018-07-06 | アデノシン誘導体を含む糖尿病性腎症の予防及び治療用薬学的組成物 |
| US16/618,396 US11266644B2 (en) | 2017-07-07 | 2018-07-06 | Pharmaceutical composition for preventing and treating diabetic nephropaphy including adenosine derivative |
| AU2018297009A AU2018297009B2 (en) | 2017-07-07 | 2018-07-06 | Pharmaceutical composition for preventing and treating diabetic nephropathy including adenosine derivative |
| CN202410170774.2A CN118001283A (zh) | 2017-07-07 | 2018-07-06 | 用于预防及治疗糖尿病性肾病的包含腺苷衍生物的药学组合物 |
| JP2021171570A JP2022017355A (ja) | 2017-07-07 | 2021-10-20 | アデノシン誘導体を含む糖尿病性腎症の予防及び治療用薬学的組成物 |
| US17/585,497 US20220143017A1 (en) | 2017-07-07 | 2022-01-26 | Pharmaceutical composition for preventing and treating diabetic nephropaphy including adenosine derivative |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2017-0086703 | 2017-07-07 | ||
| KR1020170086703A KR101820909B1 (ko) | 2017-07-07 | 2017-07-07 | 아데노신 유도체를 포함하는 만성신장질환 예방 및 치료용 약학적 조성물 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/618,396 A-371-Of-International US11266644B2 (en) | 2017-07-07 | 2018-07-06 | Pharmaceutical composition for preventing and treating diabetic nephropaphy including adenosine derivative |
| US17/585,497 Continuation US20220143017A1 (en) | 2017-07-07 | 2022-01-26 | Pharmaceutical composition for preventing and treating diabetic nephropaphy including adenosine derivative |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019009674A1 true WO2019009674A1 (ko) | 2019-01-10 |
Family
ID=61071116
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2018/007717 Ceased WO2019009674A1 (ko) | 2017-07-07 | 2018-07-06 | 아데노신 유도체를 포함하는 당뇨병성 신증 예방 및 치료용 약학적 조성물 |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US11266644B2 (ko) |
| EP (2) | EP3650029B1 (ko) |
| JP (2) | JP6965429B2 (ko) |
| KR (1) | KR101820909B1 (ko) |
| CN (2) | CN110869030B (ko) |
| AU (1) | AU2018297009B2 (ko) |
| CA (1) | CA3069118C (ko) |
| WO (1) | WO2019009674A1 (ko) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101805400B1 (ko) * | 2017-03-21 | 2017-12-07 | 퓨쳐메디신 주식회사 | 아데노신 유도체를 포함하는 녹내장 예방 및 치료용 약학적 조성물 |
| KR101820909B1 (ko) * | 2017-07-07 | 2018-01-23 | 퓨쳐메디신 주식회사 | 아데노신 유도체를 포함하는 만성신장질환 예방 및 치료용 약학적 조성물 |
| KR102667692B1 (ko) * | 2020-12-11 | 2024-05-21 | 서울대학교산학협력단 | 1'-호모아데노신 유도체 및 이를 유효성분으로 포함하는 저아디포넥틴혈증 관련 질환의 예방 또는 치료용 약학 조성물 |
| KR20240013997A (ko) * | 2022-07-22 | 2024-01-31 | 서울대학교산학협력단 | 아데노신 수용체에 작용하는 리간드 내지 이를 포함하는 비만 예방, 개선 또는 치료용 조성물 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006031505A1 (en) * | 2004-09-09 | 2006-03-23 | Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Purine derivatives as a3 and a1 adenosine receptor agonists |
| US20100137577A1 (en) * | 2007-03-07 | 2010-06-03 | Lak Shin Jeong | Adenosine derivatives, method for the synthesis thereof, and the pharmaceutical compositions for the prevention and treatment of the inflammatory diseases containing the same as an active ingredient |
| KR101323413B1 (ko) * | 2012-07-26 | 2013-10-29 | 이화여자대학교 산학협력단 | (2r,3r,4s)-2-(2-클로로-6-(3-아이도벤질아미노)-9h-퓨린-9-일)-테트라하이드로싸이오펜-3,4-다이올 및 이의 유도체를 포함하는 만성신장질환 예방 또는 치료용 약학적 조성물 |
| KR101396092B1 (ko) | 2007-03-07 | 2014-05-15 | 미합중국 정부, 디파트먼트 오브 헬쓰 앤드 휴먼 써비스 내셔널 인스티투트 오브 헬쓰 | 아데노신 유도체, 이의 제조방법 및 이를 유효성분으로 함유하는 염증성 질환의 예방 및 치료용 약학적 조성물 |
| KR101820909B1 (ko) * | 2017-07-07 | 2018-01-23 | 퓨쳐메디신 주식회사 | 아데노신 유도체를 포함하는 만성신장질환 예방 및 치료용 약학적 조성물 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004038006A2 (en) * | 2002-10-25 | 2004-05-06 | The Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Purine nucleosides |
| HUP0203976A3 (en) * | 2002-11-15 | 2004-08-30 | Sanofi Aventis | Adenozine a3 receptors, process for their preparation and pharmaceutical compositions containing them |
| KR20080076382A (ko) * | 2007-02-15 | 2008-08-20 | (주)아모레퍼시픽 | 실로스타졸의 제어방출 제제 및 그 제조방법 |
| KR101207755B1 (ko) * | 2010-02-02 | 2012-12-03 | 이화여자대학교 산학협력단 | 염증성 질환의 예방 및 치료용 약제학적 조성물 |
| KR20120082045A (ko) * | 2011-01-13 | 2012-07-23 | 정영조 | 영구자석에 의한 회전력 발생장치 |
| EP2912033A4 (en) * | 2012-10-24 | 2016-09-14 | Univ Illinois | COMPOSITIONS AND METHODS FOR TREATING ESTROGEN-MEDIATED ILLNESSES |
| US20170216244A1 (en) * | 2014-03-06 | 2017-08-03 | Yale University | Methods for treating diabetic nephropathy |
-
2017
- 2017-07-07 KR KR1020170086703A patent/KR101820909B1/ko active Active
-
2018
- 2018-07-06 EP EP18827831.1A patent/EP3650029B1/en active Active
- 2018-07-06 AU AU2018297009A patent/AU2018297009B2/en active Active
- 2018-07-06 JP JP2020500619A patent/JP6965429B2/ja active Active
- 2018-07-06 CA CA3069118A patent/CA3069118C/en active Active
- 2018-07-06 WO PCT/KR2018/007717 patent/WO2019009674A1/ko not_active Ceased
- 2018-07-06 EP EP23178716.9A patent/EP4234021B1/en active Active
- 2018-07-06 CN CN201880045444.3A patent/CN110869030B/zh active Active
- 2018-07-06 CN CN202410170774.2A patent/CN118001283A/zh not_active Withdrawn
- 2018-07-06 US US16/618,396 patent/US11266644B2/en active Active
-
2021
- 2021-10-20 JP JP2021171570A patent/JP2022017355A/ja not_active Ceased
-
2022
- 2022-01-26 US US17/585,497 patent/US20220143017A1/en not_active Abandoned
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006031505A1 (en) * | 2004-09-09 | 2006-03-23 | Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Purine derivatives as a3 and a1 adenosine receptor agonists |
| US20100137577A1 (en) * | 2007-03-07 | 2010-06-03 | Lak Shin Jeong | Adenosine derivatives, method for the synthesis thereof, and the pharmaceutical compositions for the prevention and treatment of the inflammatory diseases containing the same as an active ingredient |
| KR101396092B1 (ko) | 2007-03-07 | 2014-05-15 | 미합중국 정부, 디파트먼트 오브 헬쓰 앤드 휴먼 써비스 내셔널 인스티투트 오브 헬쓰 | 아데노신 유도체, 이의 제조방법 및 이를 유효성분으로 함유하는 염증성 질환의 예방 및 치료용 약학적 조성물 |
| KR101323413B1 (ko) * | 2012-07-26 | 2013-10-29 | 이화여자대학교 산학협력단 | (2r,3r,4s)-2-(2-클로로-6-(3-아이도벤질아미노)-9h-퓨린-9-일)-테트라하이드로싸이오펜-3,4-다이올 및 이의 유도체를 포함하는 만성신장질환 예방 또는 치료용 약학적 조성물 |
| KR101820909B1 (ko) * | 2017-07-07 | 2018-01-23 | 퓨쳐메디신 주식회사 | 아데노신 유도체를 포함하는 만성신장질환 예방 및 치료용 약학적 조성물 |
Non-Patent Citations (2)
| Title |
|---|
| LEE, JIYOUN ET AL.: "The Selective A3 AR Antagonist LJ-1888 Ameliorates UUO-lnduced Tubulointerstrtial Fibrosis", THE AMERICAN JOURNAL OF PATHOLOGY, vol. 183, no. 5, 2013, pages 1488 - 1497, XP055565178 * |
| ROBERTO, PECOITS-FILHO ET AL.: "Interactions between Kidney Disease and Diabetes: Dangerous Liaisons", DIABETOLOGY & METABOLIC SYNDROME, vol. 8, no. 50, 2016, pages 1 - 21, XP055565182 * |
Also Published As
| Publication number | Publication date |
|---|---|
| US20200289511A1 (en) | 2020-09-17 |
| CA3069118A1 (en) | 2019-01-10 |
| AU2018297009A1 (en) | 2020-03-05 |
| CN110869030A (zh) | 2020-03-06 |
| CN110869030B (zh) | 2024-03-01 |
| CA3069118C (en) | 2022-05-31 |
| JP6965429B2 (ja) | 2021-11-10 |
| EP3650029A1 (en) | 2020-05-13 |
| JP2020526526A (ja) | 2020-08-31 |
| CN118001283A (zh) | 2024-05-10 |
| US11266644B2 (en) | 2022-03-08 |
| KR101820909B1 (ko) | 2018-01-23 |
| JP2022017355A (ja) | 2022-01-25 |
| EP4234021A3 (en) | 2023-09-27 |
| EP3650029B1 (en) | 2023-06-28 |
| EP4234021A2 (en) | 2023-08-30 |
| US20220143017A1 (en) | 2022-05-12 |
| EP4234021B1 (en) | 2025-03-12 |
| EP3650029A4 (en) | 2021-05-05 |
| AU2018297009B2 (en) | 2021-10-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019009674A1 (ko) | 아데노신 유도체를 포함하는 당뇨병성 신증 예방 및 치료용 약학적 조성물 | |
| WO2011118976A2 (en) | Pharmaceutical composition for the prevention or the treatment of non-alcoholic fatty liver disease and the method for prevention or treatment of non-alcoholic fatty liver disease using the same | |
| WO2019088444A1 (en) | Therapeutic agent for liver diseases | |
| KR20170123658A (ko) | 장 수용체를 활성화시키는 파네소이드 x 수용체 작용제를 사용한 성인 잠복성 자가면역 당뇨병의 치료 | |
| WO2018030879A1 (ko) | 아모디아퀸 및 항당뇨 약물을 유효성분으로 함유하는 당뇨병의 예방 또는 치료용 약학적 조성물 | |
| US20110092517A1 (en) | Use of PPAR Delta Ligands for the Treatment or Prevention of Inflammation or Energy Metabolism/Production Related Diseases | |
| WO2020130502A1 (ko) | 엠파글리플로진 및 시타글립틴을 포함하는 약학적 조성물 | |
| US20140221390A1 (en) | Prophylactic and/or therapeutic agent for non-alcoholic steatohepatitis | |
| WO2021206316A1 (ko) | 조영제 유발 급성 신부전 치료제 | |
| WO2022124795A1 (ko) | 트라메티닙 및 선택적으로 파르네소이드 x 수용체 작용제를 포함하는 대사성 또는 담즙정체성 간질환의 예방 또는 치료용 약학적 조성물 | |
| WO2020246737A1 (ko) | 콰시노이드를 포함하는 거세 저항성 전립선암 치료용 조성물 | |
| WO2019107964A1 (ko) | 아데노신 유도체를 포함하는 망막 질환 또는 시신경 질환 예방 및 치료용 약학적 조성물 | |
| WO2015111971A1 (ko) | Gpr119 리간드를 유효성분으로 포함하는 비알콜성 지방간 질환의 예방 또는 치료용 약학적 조성물 | |
| WO2023286963A1 (ko) | 아데노신 유도체를 포함하는 담관염(cholangitis) 또는 담관염에 의한 간 질환의 예방 또는 치료용 약학적 조성물 | |
| WO2022114881A1 (ko) | 벤즈브로마론을 포함하는 상처 또는 흉터의 예방 또는 치료용 약학 조성물 | |
| WO2023014117A1 (ko) | 안구건조증 치료를 위한 레코플라본 함유 점안 조성물 및 이의 제조방법 | |
| WO2023140582A1 (ko) | 이소옥사졸린 유도체를 포함하는 주사용 제제 | |
| WO2022203432A1 (ko) | 특정 약동학적 매개변수를 나타내는 디메틸푸마레이트를 유효성분으로 함유한 약학적 조성물 | |
| WO2016126026A2 (ko) | 당뇨병 치료 조성물 및 이의 용도 | |
| WO2023048526A1 (ko) | 염증복합체 억제제를 유효성분으로 함유하는, 비알코올성 지방간염, 간 섬유화의 예방, 개선 또는 치료용 약학적 조성물 | |
| WO2024054097A1 (ko) | 이나보글리플로진의 대사체를 포함하는 약학적 조성물 및 이의 용도 | |
| WO2024085698A1 (ko) | 글리코겐 가인산분해효소(pygl) 저해제를 유효성분으로 포함하는 염증성 장 질환의 예방 또는 치료용 약학 조성물 | |
| WO2018012769A1 (ko) | 자가포식 향상물질 및 그 용도 | |
| WO2020242225A1 (ko) | IRE1α 키나아제 활성화제를 유효성분으로 포함하는 단백질 형태 이상 질환의 예방 또는 치료용 조성물 | |
| WO2011074903A2 (ko) | A3 아데노신 수용체 효능제를 포함하는 약제학적 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18827831 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 3069118 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2020500619 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2018827831 Country of ref document: EP Effective date: 20200207 |
|
| ENP | Entry into the national phase |
Ref document number: 2018297009 Country of ref document: AU Date of ref document: 20180706 Kind code of ref document: A |