WO2018222134A1 - Cancer therapy - Google Patents
Cancer therapy Download PDFInfo
- Publication number
- WO2018222134A1 WO2018222134A1 PCT/SG2018/050209 SG2018050209W WO2018222134A1 WO 2018222134 A1 WO2018222134 A1 WO 2018222134A1 SG 2018050209 W SG2018050209 W SG 2018050209W WO 2018222134 A1 WO2018222134 A1 WO 2018222134A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- inhibitor
- therapy
- treatment according
- treatment
- her2
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/455—Nicotinic acids, e.g. niacin; Derivatives thereof, e.g. esters, amides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/20—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7028—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages
- A61K31/7034—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin
- A61K31/704—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin attached to a condensed carbocyclic ring system, e.g. sennosides, thiocolchicosides, escin, daunorubicin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- the present disclosure relates to a method of treating breast cancer which is negative for at least one marker selected from ER, PR, HER2, and a combination of two or more of the same (such as triple negative breast cancer] with a therapy comprising a DHODH inhibitor or a pharmaceutically acceptable salt thereof.
- Breast cancer is a heterogeneous disease exhibiting diverse biological characteristics and clinical responses. There are three molecules that have been identified which promote many breast cancers: estrogen receptor (ER], progesterone receptor (PR], and human epidermal growth factor receptor 2 (HER2]. Triple negative breast cancer is a particularly aggressive form of breast cancer which fails to express all three of these markers. Although triple negative breast cancer represents a relatively small percentage of all breast cancers ( ⁇ 20%], it is typically high grade (poorly differentiated] and rapidly progressive, with a higher risk of relapse and lower survival than other subtypes of breast cancer. Therefore, triple negative breast cancer is associated with a disproportionate number of breast cancer related deaths.
- estrogen receptor ER
- PR progesterone receptor
- HER2 human epidermal growth factor receptor 2
- DHODH inhibitors Dihydroorotate dehydrogenase inhibitors
- Known DHODH inhibitors include leflunomide or teriflunomide.
- the present inventors believe that the DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof is useful in the treatment of breast cancers that are negative for one or more of ER, PR and HER2, in particular triple negative breast cancer.
- the present disclosure provides a method of treating a breast cancer, which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same comprising administering a therapeutically effect amount of a DHODH inhibitor 2-(3,5- difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof.
- the DHODH inhibitor 2- (3,5-difluoro-3'methoxybiphenyl-4- ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof does not rely on targetting the expression of ER, PR and/or HER2, it can effectively treat a breast cancer negative for one or more of these receptor molecules, in particular the so-called triple negative breast cancer.
- the breast cancer is negative for ER. In another embodiment, the breast cancer is negative for PR. In another embodiment, the breast cancer is negative for HER2.
- the breast cancer is negative for ER and PR. In another embodiment, the breast cancer is negative for PR and HER2. In another embodiment, the breast cancer is negative for ER and HER2.
- the breast cancer is negative for ER, PR and HER2, also referred herein as triple negative breast cancer.
- the DHODH inhibitor is employed in a combination therapy with a second therapy, in particular a second cancer therapy.
- the second therapy is an inhibitor of DNA repair.
- the inhibitor is a small molecule therapy.
- inhibitor mechanism is via the base excision repair pathway.
- the inhibitor's target is independently selected from APE1, Pol ⁇ , FEN1, and PARP.
- the inhibitor is selected from TRC102, (2E]-2-[(4,5-Dimethoxy-2- methyl-3,6-dioxo-l,4-cyclohexadien-l-yl]methylene]-undecanoic acid [also known as E3330], NCS- 666715 and NSC-124854, 8-oxoguamine, tanespirmycin, luminespib, alvespimycin, genetespib, retaspimycin, 6-Amino-8- [(6-iodo-l,3-benzodioxol-5-yl]thio]-N-(l-methylethyl]-9H-purine-9- propanamine (PU-H71], 4-[2-carbamoyl-5- [6,6-dimethyl-4-oxo-3-(trifluoromethyl]-5,7- dihydroindazol-l-yl]anilino]cyclohexyl] 2-amin
- the inhibitor is a PARP inhibitor, such as a PARP-1 and/or PARP- 2 inhibitor.
- the PARP inhibitor is independently selected from olaparib, rucaparib, niraparib, iniparib, talazoparib, veliparib, CEP9722, E7016, BGB-290, AZD-2461, 3-aminobenzamide and combinations thereof.
- the inhibitor mechanism is via the mismatch repair pathway.
- the inhibitor mechanism is via the nucleotide excision pathway.
- the inhibitor is independently selected from 7-hydroxystaurosporine [UCN-01], trabectedin, MCI13E, NERI01 and combinations of two or more of the same.
- the inhibitor mechanism is via the double stranded break repair pathway.
- the inhibitor mechanism is via the non-homologous end joining pathway.
- the inhibitor mechanism is via the homologous recombination pathway.
- the second therapy is a topoisomerase inhibitor, such as topoisomerase I and/or II inhibitor.
- the topoisomerase inhibitor is independently selected from irinotecan, topotecan, camptothecin lamellarin D and combinations thereof.
- the topoisomerase inhibitor is independently selected from etoposide
- VP- 16 teniposide, doxorubicin, daunorubicin, mitoxantrone, amsacrine, ellipticines, aurintricarboxylic acid, 3-Hydroxy-2- [(li?]-6-isopropenyl-3-methyl-cyclohex-2-en- l-yl]-5-pentyl- 1,4-benzoquinone (HU-331] and combinations thereof.
- the topoisomerase inhibitor is doxorubicin.
- the DHODH inhibitor provides anticancer efficacy via induction of p53.
- the DHODH inhibitor is administered orally, for example once daily, or bi- daily.
- the DHODH inhibitor is administered at a dose of 50 to 500 mg/kg, for example 50, 60, 70, 80, 90, 100, 150, 200, 250, 300, 350, 400, 450 or 500 mg/kg.
- the DHODH inhibitor is administered at a dose of 100 mg/kg.
- the DHODH inhibitor is administered at a dose of 100 mg/kg, once daily.
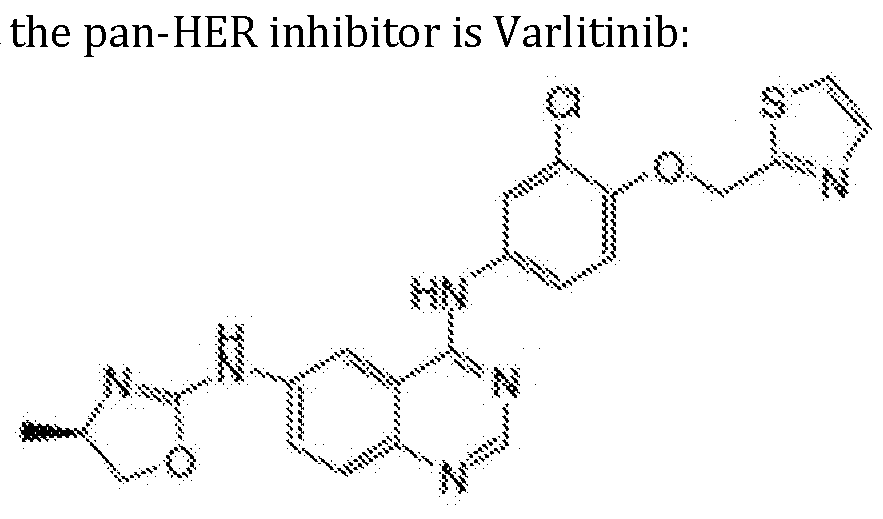
- the breast cancer therapy further comprises a pan-HER inhibitor [R)- N4- [3-Chloro-4- (thiazol2-ylmethoxy]-phenyl]-N6- (4-methyl-4,5,-dihydro-oxazol-2-yl]- quinazoline-4,6-diamine or a pharmaceutically acceptable salt thereof.
- pan-HER inhibitor is administered parenterally.
- pan-HER inhibitor is administered orally, for example is administered bi-daily.
- 2-(3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof for use in the treatment of a breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as triple negative breast cancer.
- pan-HER inhibitor such as (i?]-N4- [3-Chloro-4-(thiazol2-ylmethoxy]-phenyl]-N6-(4-methyl-4,5,-dihydro-oxazol-2-yl]- quinazoline-4,6-diamine or a pharmaceutically acceptable salt thereof
- a DHODH inhibitor 2- (3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof for use in the treatment of a breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as a triple negative negative breast cancer.
- a DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt in the manufacture of a therapy for the treatment of breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as a triple negative breast cancer.
- a DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt in the manufacture of a combination therapy for the treatment of breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as triple negative breast cancer.
- pan-HER inhibitor such as i?]-N4-[3-Chloro-4-(thiazol2- ylmethoxy]-phenyl]-N6-(4-methyl-4,5,-dihydro-oxazol-2-yl]-quinazoline-4,6-diamine or a pharmaceutically acceptable salt thereof
- DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt in the manufacture of a combination therapy for the treatment of a breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same.
- the therapy of the present disclosure continues for 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60 months or more.
- dosing of the second therapy continues for 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60 months or more.
- Varlitinib (i?]-N4-[3-Chloro-4-(thiazol2-ylmethoxy]-phenyl]-N6-(4-methyl-4,5,-dihydro-oxazol-2-yl]- quinazoline-4,6-diamine is also known as Varlitinib.
- Varlitinib is administered in a 28 day cycle.
- DHODH is a key enzyme in the production of uridine, which is a central building block in the cell. Whilst not wishing to be bound by theory it may be that the DHODH inhibitor is able to upregulate p53 based apoptosis.
- the up-regulation of p53 (which may lead to cell cycle arrest and at higher levels of p 53, to apoptosis] is likely to occur via mechanisms sensing the levels of intracellular uridine, and then setting forth a series of reactions leading to stabilisation of p53, and increasing its concentration (also referred to herein as p53 induction].
- DHODH inhibitors such as -(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof may be particularly advantageous for use in the treatment of breast cancers because they kill cancer cells via apoptosis, which does not involve necrotic cell death.
- Necrosis is a form of cell injury which results in premature death of cells in living tissue by autolysis (i.e. destruction of the cell through the action of its own enzymes]. Necrosis is caused by factors external to the cell or tissue, such as infection, toxins, or trauma which result in the unregulated digestion of cell components. In contrast, apoptosis is a naturally occurring programmed and targeted cause of cellular death.
- necrotic cell death does not follow the apoptotic signal transduction pathway, but rather various receptors are activated, and result in the loss of cell membrane integrity and an uncontrolled release of products of cell death into the extracellular space.
- necrosis results in a build-up of decomposing dead tissue and cell debris at or near the site of the cell death.
- a classic example is gangrene. For this reason, it is often necessary to remove necrotic tissue surgically, a procedure known as debridement.
- the ability to treat breast cancers with a DHODH inhibitor that causes apoptotic cell death is likely to lead to less side effects and an overall better therapeutic outcome.
- the DHODH inhibitor herein may be beneficial in the therapy according to the present disclosure because the biological effects are stronger than those of "first generation" DHODH inhibitors, such as leflunomide and teriflunomide.
- the DHODH inhibitor of the present disclosure may have less toxicity (off target effects] than the so-called "first generations” inhibitors, such as leflunomide and teriflunomide.
- the DHODH is employed in a combination therapy with a biological therapeutic agent, such as an antibody or binding fragment thereof.
- a biological therapeutic agent such as an antibody or binding fragment thereof.
- the biological therapeutic agent is administered parenterally.
- DHODH inhibitor as a combination therapy, for example wherein the second therapy is an inhibitor of the DNA repair and/or a pan-HER inhibitor may be particular beneficial to minimises the cancers ability to resist treatment, in particular by "attacking" the cancer cells by two or more mechanisms with the combination therapy.
- an inhibitor of DNA repair for example, the sensitivity of a tumor cell to DNA damage caused by anti-cancer agents and thus DNA damage-induced cell death is enhanced.
- ABT-737 a PARP-1 and PARP-2 inhibitor
- Topoisomerase inhibitors which may be employed in a method of the present disclosure include type I topoisomerase inhibitors, type II topoisomerase inhibitors and type II
- Type I inhibitors include topotecan, irinotecan, indotecan and indimitecan.
- Type II inhibitors include genistein and ICRF 193 which has the following structure:
- Type II poisons include amsacrine, etoposide, etoposide phosphate, teniposide and doxorubicin and fluoroquinolones.
- the DHODH inhibitor of the present disclosure is employed in a combination therapy comprising chemotherapy, in particular a described herein.
- a method of treating a patient comprising administering a therapeutically effective amount of an inhibitor of a pan-HER inhibitor and a therapeutically effective amount of DHODH inhibitor.
- the pan-HER inhibitor is an organic chemistry molecule, for example with a molecular weight of 500 or less.
- pan-HER inhibitor has a molecular formula of formula (I):
- pan-HER inhibitor is an inhibitor of one or more HER receptors independently selected from HER1, HER2, HER3, HER4.
- Varlitinib is employed as a free base. Varlitinib at an appropriate dose is capable of inhibiting HERl, HER2 and HER4 directly and thought to be capable of inhibiting HER3 indirectly.
- the compound of formula (I] (including formula (la] and Varlitinib] at least inhibits the activity of HERl and HER2, HERl and HER4 or HER2 and HER4.
- the compound of formula (I] (including formula (la] and Varlitinib] at least inhibits the activity of HERl, HER2 and HER4, for example directly inhibits the activity of HERl, HER2 and HER4.
- the compound of formula (I] (including formula (la] and Varlitinib] inhibits the activity of HERl, HER2, HER3 and HER4, for example directly inhibits the activity of HERl, HER2, and HER4, and indirectly inhibits the activity of HER3
- each dose of the compound of formula (I], (including formula (la] and Varlitinib] is in the range 100 to 900mg, for example each dose is in the range of 300 to 500mg, such as 400mg, for example administered once or twice daily, such as twice daily.
- patients may benefit from having the initial dose reduced to 300mg or 200mg bi-daily.
- Other patients may benefit from receiving the compound of formula (I], such as Varlitinib, for example, in a regime which is non-continuous, such as taking medication on alternate days instead of each day or taking medication for four sequential days followed by one, two or three days without medication.
- a regime which is non-continuous such as taking medication on alternate days instead of each day or taking medication for four sequential days followed by one, two or three days without medication.
- the compound of formula (I], (including formula (la] and Varlitinib] is administered orally.
- the pan-HER inhibitor is a combination of HER inhibitors.
- pan-HER inhibitor such as Varlitinib is administered twice daily, for example a dose in the range disclosed herein.
- the DHODH inhibitor is 2- (3, 5-difluoro-3'-methoxybiphenyl-4-ylamino] nicotinic acid
- ASLAN003 (referred to herein as ASLAN003] or a pharmaceutically acceptable salt thereof, which has the structure:
- the DHODH inhibitor is administered daily, for example once daily.
- the DHODH inhibitor is administered orally.
- the DHODH inhibitor and the second therapy (for example a pan-HER inhibitor such as particular Varlitinib or chemotherapy] are administered sequentially in a treatment regimen, for example are administered on the same day.
- the DHODH inhibitor and the second therapy are administered simultaneously, at approximately the same time.
- the DHODH inhibitor is administered in regimen that is daily or weekly for a continuous period of time for example 1 to 60 months or more.
- the second therapy (for example the pan-HER inhibitor such as Varlitinib or chemotherapy] may be administered intermittently during this period, and for example may be administered in a one or more 28 days cycles.
- the administration protocol is likely to be very different to that of small molecule inhibitors.
- An antibody or binding fragment may be administered (in particular in combination with cytotoxic chemotherapy] in a regime as follows:
- the second therapy (for example a pan-HER inhibitor or chemotherapy] is administered in regimen that is daily or weekly for a continuous period of time, for example 1 to 60 months or more, and the DHODH inhibitor is administered intermittently during this period.
- Administered intermittently as employed herein refers to a period wherein the therapy is administered and then stopped with the option of starting the therapy again at some point in the future, for example in accordance with a treatment plan.
- the DHODH inhibitor is administered in regimen that is daily or weekly for a continuous period of time, for example 1 to 60 months or more
- the second therapy for example a pan-HER inhibitor or chemotherapy] is administered in regimen that is daily or weekly for a continuous period of time, for example 1 to 60 months or more.
- the DHODH inhibitor is administered in regimen that is daily or weekly continuously over for example 1 to 60 months or more
- the second therapy for example a pan- HER inhibitor or chemotherapy] is administered conjunctly with DHODH inhibitor in regimen that is daily or weekly for intermittent periods over, for example 1 to 60 months or more.
- the DHODH inhibitor is administered in regimen that is daily or weekly for intermittent periods over for example 1 to 60 months or more
- the second therapy for example a pan-HER inhibitor or chemotherapy] is administered conjunctly with DHODH inhibitor in regimen that is daily or weekly for intermittent periods over, for example 1 to 60 months or more.
- the DHODH inhibitor and the second therapy are co-formulated.
- the DHODH inhibitor is administered orally.
- the second therapy is administered orally and/or administered parenterally, such as intravenously.
- the DHODH inhibitor and the second therapy are both administered orally.
- the DHODH inhibitor is administered orally and the second therapy is administered parenterally.
- the patient is a human.
- the therapy of the present disclosure is for example beneficial in that it provides augmented therapeutic activity in comparison to therapies currently employed for breast cancer, such as triple negative breast cancer.
- Augmented activity may be any beneficial therapeutic effect of employing the therapy of the present disclosure, for example an increase in anti-tumor activity and/or a reduced propensity for the cancer to become resistant
- Other benefits may be therapeutic effect in patients who have failed one or more lines of therapy.
- the patient population has a cancer that is resistant or refractory to known therapies, such as cytotoxic chemotherapy.
- Combination therapy refers to two or more modes of therapy being employed over the same treatment period, i.e. the opposite of sequential therapy.
- Two or more modes of therapy as employed herein refers to at least two therapies which have different modes of action and/or different activities and/or different routes of administration.
- the DHODH inhibitor and the second therapy have to administered in a time frame, where the pharmacological effects of a both therapies overlap, i.e. the treatment regimens for the said therapies partly coincide in time.
- the pharmacological effects of a both therapies overlap, i.e. the treatment regimens for the said therapies partly coincide in time.
- Combination therapy refers to where a medicament according to the present disclosure is administered in a treatment regimen along with at least one further therapeutic agent.
- the regime may be separate formulations administered at the same time or different times or co-formulations of the two or more therapeutic agents.
- the "first" medicament employed in the combination therapy according to the present disclosure may be administered; prior to the further therapeutic agent or agents, concomitant with the further therapeutic agent or agents, or after the further therapeutic agent or agents.
- therapeutic agent or agents such as an anti-cancer therapy are employed in combination with the therapy of the present disclosure.
- the therapeutic agent is a chemotherapeutic agent
- Chemotherapeutic agent as employed herein is intended to refer to specific antineoplastic chemical agents or drugs that are destructive to malignant cells and tissues, including alkylating agents, antimetabolites, anthracyclines, plant alkaloids, topoisomerase inhibitors, and other antitumour agents.
- Specific examples of chemotherapy include doxorubicin, 5-fluorouracil (5-FU], paclitaxel (for example abraxane or docetaxel], capecitabine, irinotecan, and platins, such as cisplatin and oxaliplatin or a combination thereof.
- a suitable dose may be chosen by the practitioner based on the nature of the cancer being treated and the patient
- Co-administered as employed herein refers to administration of the DHODH inhibitor and the second therapy at the same time or approximately the same time (including where the actives are administered by the same or different routes].
- Inhibitor refers to the reduction of a relevant biological activity, for example by 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95 or 100%, such as when measured in a relevant in vitro assay.
- Direct inhibition is where the inhibitor binds directly to or physically blocks a binding interaction to inhibit a biological activity, or when the inhibitor inhibits the activation through phosphorylation of the target molecule.
- Indirect inhibition refers to where the biological activity in question is inhibited as a result of directly inhibiting a target that is other than the entity that is indirectly inhibited.
- Dihydroorotate dehydrogenase is the enzyme that catalyzes the fourth step in the pyrimidine biosynthetic pathway namely the conversion of dihydroorotate to orotate concomitantly with an electron transfer to ubiquinone (cofactor Q] via a flavin mononucleotide intermediate (Loffler Mol Cell Biochem, 1997].
- DHODH Dihydroorotate dehydrogenase
- mitochondrial cytochrome bcl a component of the electron transport chain complex III
- DHODH dihydroorotate dehydrogenase
- the p53 activation has been shown to be triggered by the impairment of the de novo pyrimidine 15 biosynthesis due to the suppression of DHODH.
- a DHODH inhibitor as employed herein refers to a compound which inhibits the activity of dihydroorotate dehydrogenase, in particular in vivo.
- ASLAN003 is disclosed in WO2008/077639, incorporated herein by reference.
- a biological therapeutic is one based on a protein, for example an antibody or binding fragment thereof, including fusion proteins and biological molecules conjugated to a polymer, toxin or similar payload.
- a “drug” as employed herein refers to a chemical entity, organic chemistry molecule with pharmacological activity.
- An example of a biological therapeutic conjugated to a payload is trastuzumab emtansine.
- Examples of pharmaceutically acceptable salts include but are not limited to acid addition salts of strong mineral acids such as HC1 and HBr salts and addition salts of strong organic acids, such as a methansulfonic acid salt, tosylates, furoates and the like, including di, tri salts thereof, such as ditosylates.
- the combination therapy according to the present disclosure further comprises a RON inhibitor, for example as disclosed WO2008/058229, incorporated herein by reference.
- the combination therapy of the present disclosure comprises a checkpoint inhibitor, such as a CTLA4 inhibitor, a PD-1 inhibitor or a PD-Ll inhibitor, in particular an antibody or binding fragment thereof.
- a checkpoint inhibitor such as a CTLA4 inhibitor, a PD-1 inhibitor or a PD-Ll inhibitor, in particular an antibody or binding fragment thereof.
- the DHODH therapy of the present disclosure may be employed in combination with a further cancer therapy, for example chemotherapy.
- Chemotherapeutic agent and chemotherapy or cytotoxic agent are employed interchangeably herein unless the context indicates otherwise.
- Chemotherapy as employed herein is intended to refer to specific antineoplastic chemical agents or drugs that are "selectively" destructive to malignant cells and tissues, for example alkylating agents, antimetabolites including thymidylate synthase inhibitors, anthracyclines, anti- microtubule agents including plant alkaloids, topoisomerase inhibitors, parp inhibitors and other antitumour agents. Selectively in this context is used loosely because of course many of these agents have serious side effects.
- the preferred dose may be chosen by the practitioner, based on the nature of the cancer being treated.
- alkylating agents which may be employed in the method of the present disclosure include an alkylating agent nitrogen mustards, nitrosoureas, tetrazines, aziridines, platins and derivatives, and non-classical alkylating agents.
- platinum containing chemotherapeutic agent also referred to as platins
- platins such as cisplatin, carboplatin, oxaliplatin, satraplatin, picoplatin, nedaplatin, triplatin and lipoplatin (a 25 liposomal version of cisplatin]
- cisplatin carboplatin and oxaliplatin.
- the dose for cisplatin ranges from about 20 to about 270 mg/m 2 depending on the exact cancer. Often the dose is in the range about 70 to about 100mg/m 2 .
- Nitrogen mustards include mechlorethamine, cyclophosphamide, melphalan, chlorambucil, ifosfamide and busulfan.
- Nitrosoureas include N-Nitroso-N-methylurea (MNU], carmustine (BCNU], lomustine (CCNU] and semustine (MeCCNU], fotemustine and streptozotocin.
- Tetrazines include dacarbazine, mitozolomide and temozolomide.
- Aziridines include thiotepa, mytomycin and diaziquone (AZQ].
- antimetabolites examples include anti-folates (for example methotrexate and pemetrexed], purine analogues (for example thiopurines, such as azathiopurine, mercaptopurine, thiopurine, fludarabine (including the phosphate form], pentostatin and cladribine], pyrimidine analogues (for example fluoropyrimidines, such as 5-fluorouracil and prodrugs thereof such as capecitabine [Xeloda®]], floxuridine, gemcitabine, cytarabine, decitabine, raltitrexed(tomudex] hydrochloride, cladribine and 6-azauracil.
- anti-folates for example methotrexate and pemetrexed
- purine analogues for example thiopurines, such as azathiopurine, mercaptopurine, thiopurine, fludarabine (including the phosphate form],
- anthracyclines examples include daunorubicin (Daunomycin], daunorubicin (liposomal], doxorubicin (Adriamycin], doxorubicin (liposomal], epimbicin, idambicin, valmbicin currenlty used only to treat bladder cancer and mitoxantrone an anthracycline analog, in particular doxorubicin.
- anti-microtubule agents examples include vinca alkaloids and taxanes.
- Vinca alkaloids include completely natural chemicals for example vincristine and vinblastine and also semi-synthetic vinca alkaloids, for example vinorelbine, vindesine, and vinflunine
- Taxanes include paclitaxel, docetaxel, abraxane, carbazitaxel and derivatives of thereof.
- Derivatives of taxanes as employed herein includes reformulations of taxanes like taxol, for example in a micelluar formulations, derivatives also include chemical derivatives wherein synthetic chemistry is employed to modify a starting material which is a taxane.
- chemotherapeutic agents employed is, for example a platin and 5-FU or a prodrug thereof, for example cisplatin or oxaplatin and capecitabine or gemcitabine, such as FOLFOX.
- the chemotherapy comprises a combination of chemotherapy agents, in particular cytotoxic chemotherapeutic agents.
- the chemotherapy combination comprises a platin, such as cisplatin and fluorouracil or capecitabine.
- the chemotherapy combination is capecitabine and oxaliplatin (Xelox].
- the chemotherapy is a combination of folinic acid and 5-FU, optionally in combination with oxaliplatin.
- the chemotherapy is a combination of folinic acid, 5-FU and irinotecan (FOLFIRI], optionally in combination with oxaliplatin (FOLFIRINOX].
- the regimen consists of: irinotecan (180 mg/m 2 IV over 90 minutes] concurrently with folinic acid (400 mg/m 2 [or 2 x 250 mg/m 2 ] IV over 120 minutes]; followed by fluorouracil (400-500 mg/m 2 IV bolus] then fluorouracil (2400-3000 mg/m 2 intravenous infusion over 46 hours]. This cycle is typically repeated every two weeks.
- the dosages shown above may vary from cycle to cycle.
- the chemotherapy combination employs a microtubule inhibitor, for example vincristine sulphate, epothilone A, N- [2-[(4-Hydroxyphenyl]amino]-3-pyridinyl]- 4methoxybenzenesulfonamide (ABT-751], a taxol derived chemotherapeutic agent, for example paclitaxel, abraxane, or docetaxel or a combination thereof.
- a microtubule inhibitor for example vincristine sulphate, epothilone A, N- [2-[(4-Hydroxyphenyl]amino]-3-pyridinyl]- 4methoxybenzenesulfonamide (ABT-751]
- ABT-751 N- [2-[(4-Hydroxyphenyl]amino]-3-pyridinyl]- 4methoxybenzenesulfonamide
- the chemotherapy combination employs an mTor inhibitor.
- mTor inhibitors include: everolimus (RAD001], WYE-354, KU-0063794, papamycin (Sirolimus], Temsirolimus, DeforolimusfMK-8669), AZD8055 and BEZ235 (NVP-BEZ235).
- the chemotherapy combination employs a MEK inhibitor.
- MEK inhibitors include: AS703026, CI-1040 (PD184352), AZD6244 (Selumetinib], PD318088, PD0325901, AZD8330, PD98059, U0126-EtOH, BIX 02189 or BIX 02188.
- the chemotherapy combination employs an AKT inhibitor.
- AKT inhibitors include: MK-2206 and AT7867.
- the combination employs an aurora kinase inhibitor.
- aurora kinase inhibitors include: Aurora A Inhibitor I, VX-680, AZD1152-HQPA (Barasertib], SNS314 Mesylate, PHA-680632, ZM-447439, CCT129202 and Hesperadin.
- the chemotherapy combination employs a p38 inhibitor, for example as disclosed in WO2010/038086, such as iV- [4-( ⁇ 4-[3-(3-tert-Butyl-l-p-tolyl-lH-pyrazol- 5yl]ureido]naphthalen-l-yloxy ⁇ methyl]pyridin-2-yl]-2-methoxyacetamide.
- a p38 inhibitor for example as disclosed in WO2010/038086, such as iV- [4-( ⁇ 4-[3-(3-tert-Butyl-l-p-tolyl-lH-pyrazol- 5yl]ureido]naphthalen-l-yloxy ⁇ methyl]pyridin-2-yl]-2-methoxyacetamide.
- the combination employs a Bcl-2 inhibitor.
- Bcl-2 inhibitors include: obatoclax mesylate, ABT-737, ABT-263 (navitoclax] and TW-37.
- the chemotherapy combination comprises an antimetabolite such as capecitabine (xeloda], fludarabine phosphate, fludarabine (fludara], decitabine, raltitrexed (tomudex], gemcitabine hydrochloride and cladribine.
- an antimetabolite such as capecitabine (xeloda], fludarabine phosphate, fludarabine (fludara], decitabine, raltitrexed (tomudex], gemcitabine hydrochloride and cladribine.
- the chemotherapy combination comprises ganciclovir, which may assist in controlling immune responses and/or tumour vasculation.
- one or more therapies employed in the method herein are metronomic, that is a continuous or frequent treatment with low doses of anticancer drugs, often given concomitant with other methods of therapy.
- the therapy of the present disclosure is employed after chemotherapy. In one embodiment the therapy of the present disclosure is employed before chemotherapy.
- the dose of chemotherapy employed in the therapy of the present disclosure is lower than the dose of chemotherapy employed in "monotherapy" (where monotherapy may include the dose of chemotherapy employed when combinations of chemotherapy agents are employed].
- the medicament is administered in combination with therapy complimentary to the cancer therapy, for example a treatment for cachexia, such as cancer cachexia, for example S-pindolol, S-mepindolol or S-bopindolol.
- a treatment for cachexia such as cancer cachexia, for example S-pindolol, S-mepindolol or S-bopindolol.
- Suitable doses may be in the range of 2.5mg to lOOmg, such as 2.5mg to 50mg per day provided a single dose or multiple doses given as multiple doses administered during the day.
- Treatment as employed herein refers to where the patient has a disease or disorder, for example cancer and the medicament according to the present disclosure is administered to stabilise the disease, delay the disease, amelorate the disease, send the disease into remission, maintain the disease in remission or cure the disease.
- Treating as employed herein includes administration of a medicament according to the present disclosure for treatment or prophylaxis.
- the present disclosure is explained in the context of a method of treating a patients.
- the disclosure extends to use of the combination therapy as described herein for use in treatment, in particular for the treatment of cancer, such as a cancer described herein.
- Also provided is use of the combination of compounds as described herein for the manufacture of a medicament for the treatment of cancer, in particular a cancer described herein.
- the combination therapy according to the present disclosure is employed as cancer adjuvant therapy, for example after surgery to remove some or all of the cancerous cells.
- a therapeutically effective dose (such as a daily dose] of a DHODH inhibitor is in the range lOmgto lOOOmg, for example 50 to 500mg, such as 100, 150, 200, 250, 300, 350, 00, 50, 500mg, in particular administered once a day.
- the DHODH inhibitor for example 2-(3,5-difluoro-3'methoxybiphenyl-
- 4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof is administered bi-daily, such as at a dose in the range lOOmgto 900mgon each occasion, in particular 300mg, or 400mgor 500mg each dose.
- active ingredients employed in the therapy of the present disclosure will be provided in the form of a pharmaceutical formulation comprising one or more excipients, diluents or carriers.
- the DHODH inhibitor for example 2-(3,5-difluoro-3'methoxybiphenyl- 4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof is administered as pharmaceutical formulation comprising one or more pharmaceutically acceptable excipients.
- Embodiments of the invention comprising certain features/elements are also intended to extend to alternative embodiments "consisting" or “consisting essentially” of the relevant elements/features.
- ASLAN003 is a small molecule inhibitor of Dihydroorotate Dehydrogenase (DHODH].
- DHODH Dihydroorotate Dehydrogenase
- ASLAN003 would reduce triple negative breast tumor growth, particularly when combined with doxorubicin. This would potentially give rise to a new therapeutic regimen and/or allow for reduction of doxorubicin doses to reduce risks of side effects attributed to doxorubicin, such as cardiotoxicity.
- TNBC PDX lines with/without mutations in PTEN and/or TP53
- ASLAN003 +/- doxocirubicin for in 4 treatment groups for each line (12mice/group]: (A] vehicle control; (B] ASLAN003 alone; (C] doxorubicin alone; and (D] ASLAN003 + doxorubicin. Mice were randomized to drug treatment groups when tumors reached approximately 100 mm 3 (rolling enrollment]. Table 1. Huntsman Cancer Institute Human Breast Cancer POX models used in this study
- ASLAN003 Prepared fresh every day: 10 mg/mL in 30% PEG300+30% Captisol, sonicated for 1.5 hrs before use.
- Doxorubicin 10X stock (Selleck Chemical Cat# S1208] : 20mg/ml in H2O. Stored at -80C in 0.5mL aliquots. Working solution prepared fresh by diluting 1 : 10 in H2O (for high dose] or 1 : 100 in H 2 0 (for low dose].
- Frozen tissue fragments ( ⁇ 4x2x2 mm] were thawed by placing the vial in a 37 ° C water bath and removed before the medium completely melted.
- mice were randomized into 1 of 4 treatment groups:
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
A method of treating breast cancer which is negative for at least one marker selected from ER, PR, HER2, and a combination of two or more of the same (such as triple negative breast cancer) with a therapy comprising a DHODH inhibitor or a pharmaceutically acceptable salt thereof.
Description
CANCER THERAPY
The present disclosure relates to a method of treating breast cancer which is negative for at least one marker selected from ER, PR, HER2, and a combination of two or more of the same (such as triple negative breast cancer] with a therapy comprising a DHODH inhibitor or a pharmaceutically acceptable salt thereof.
BACKGROUND
Breast cancer is a heterogeneous disease exhibiting diverse biological characteristics and clinical responses. There are three molecules that have been identified which promote many breast cancers: estrogen receptor (ER], progesterone receptor (PR], and human epidermal growth factor receptor 2 (HER2]. Triple negative breast cancer is a particularly aggressive form of breast cancer which fails to express all three of these markers. Although triple negative breast cancer represents a relatively small percentage of all breast cancers (~20%], it is typically high grade (poorly differentiated] and rapidly progressive, with a higher risk of relapse and lower survival than other subtypes of breast cancer. Therefore, triple negative breast cancer is associated with a disproportionate number of breast cancer related deaths.
Current clinical approaches for breast cancer typically include agents that target at least one of the three molecules identified, for example endocrine therapies and monoclonal antibodies, such as trastuzumab (Herceptin] which target HER2. However, because triple negative breast cancer is defined as absence of all of these targets, conventional cytotoxic chemotherapy is currently the main form of therapy for patients with triple negative breast cancer. Unfortunately, conventional systemic treatments are limited by poor therapeutic response, high toxicity, and the development of resistance. Thus, there is a pressing need for novel targeted approaches for treating breast cancers that are negative for one or more of ER, PR and HER2, in particular triple negative breast cancer.
Dihydroorotate dehydrogenase inhibitors (DHODH inhibitors] are thought to be useful in the treatment of rheumatoid arthritis. Known DHODH inhibitors include leflunomide or teriflunomide. The present inventors believe that the DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof is useful in the treatment of breast cancers that are negative for one or more of ER, PR and HER2, in particular triple negative breast cancer. SUMMARY OF THE DISCLOSURE
Thus in a first aspect the present disclosure provides a method of treating a breast cancer, which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same comprising administering a therapeutically effect amount of a DHODH inhibitor 2-(3,5- difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof.
Advantageously, because the DHODH inhibitor 2- (3,5-difluoro-3'methoxybiphenyl-4- ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof does not rely on targetting the
expression of ER, PR and/or HER2, it can effectively treat a breast cancer negative for one or more of these receptor molecules, in particular the so-called triple negative breast cancer.
In one embodiment, the breast cancer is negative for ER. In another embodiment, the breast cancer is negative for PR. In another embodiment, the breast cancer is negative for HER2.
In one embodiment, the breast cancer is negative for ER and PR. In another embodiment, the breast cancer is negative for PR and HER2. In another embodiment, the breast cancer is negative for ER and HER2.
In one embodiment, the breast cancer is negative for ER, PR and HER2, also referred herein as triple negative breast cancer.
Thus in one aspect there is provided a new patient population according to the present disclosure.
In one embodiment, the DHODH inhibitor is employed in a combination therapy with a second therapy, in particular a second cancer therapy.
In one embodiment, the second therapy is an inhibitor of DNA repair.
In one embodiment, the inhibitor is a small molecule therapy.
In one embodiment, wherein the inhibitor mechanism is via the base excision repair pathway.
In one embodiment, the inhibitor's target is independently selected from APE1, Pol β, FEN1, and PARP.
In one embodiment, the inhibitor is selected from TRC102, (2E]-2-[(4,5-Dimethoxy-2- methyl-3,6-dioxo-l,4-cyclohexadien-l-yl]methylene]-undecanoic acid [also known as E3330], NCS- 666715 and NSC-124854, 8-oxoguamine, tanespirmycin, luminespib, alvespimycin, genetespib, retaspimycin, 6-Amino-8- [(6-iodo-l,3-benzodioxol-5-yl]thio]-N-(l-methylethyl]-9H-purine-9- propanamine (PU-H71], 4-[2-carbamoyl-5- [6,6-dimethyl-4-oxo-3-(trifluoromethyl]-5,7- dihydroindazol-l-yl]anilino]cyclohexyl] 2-aminoacetate(SNX-5422], luminespib (resorcyinylic], 2- (2-ethyl-3,5-dihydroxy-6-(3-methoxy-4-(2-morpholinoethoxy]benzoyl]phenyl]-N,N-bis(2- methoxyethyl]acetamide (KW-2478], AT13387, 5,6-bis((E]-benzylideneamino]-2-thioxo-2,3- dihydropyrimidin-4(lH]-one (SCR7] and combinations of two or more of the same.
In one embodiment, the inhibitor is a PARP inhibitor, such as a PARP-1 and/or PARP- 2 inhibitor.
In one embodiment, the PARP inhibitor is independently selected from olaparib, rucaparib, niraparib, iniparib, talazoparib, veliparib, CEP9722, E7016, BGB-290, AZD-2461, 3-aminobenzamide and combinations thereof.
In one embodiment, the inhibitor mechanism is via the mismatch repair pathway.
In one embodiment, the inhibitor mechanism is via the nucleotide excision pathway.
In one embodiment, the inhibitor is independently selected from 7-hydroxystaurosporine [UCN-01], trabectedin, MCI13E, NERI01 and combinations of two or more of the same.
In one embodiment, the inhibitor mechanism is via the double stranded break repair pathway.
In one embodiment, the inhibitor mechanism is via the non-homologous end joining pathway.
In one embodiment, the inhibitor mechanism is via the homologous recombination pathway.
In one embodiment, the second therapy is a topoisomerase inhibitor, such as topoisomerase I and/or II inhibitor.
In one embodiment, the topoisomerase inhibitor is independently selected from irinotecan, topotecan, camptothecin lamellarin D and combinations thereof.
In one embodiment, the topoisomerase inhibitor is independently selected from etoposide
(VP- 16], teniposide, doxorubicin, daunorubicin, mitoxantrone, amsacrine, ellipticines, aurintricarboxylic acid, 3-Hydroxy-2- [(li?]-6-isopropenyl-3-methyl-cyclohex-2-en- l-yl]-5-pentyl- 1,4-benzoquinone (HU-331] and combinations thereof.
In one embodiment, the topoisomerase inhibitor is doxorubicin.
In one embodiment, the DHODH inhibitor provides anticancer efficacy via induction of p53.
In one embodiment, the DHODH inhibitor is administered orally, for example once daily, or bi- daily.
In one embodiment, the DHODH inhibitor is administered at a dose of 50 to 500 mg/kg, for example 50, 60, 70, 80, 90, 100, 150, 200, 250, 300, 350, 400, 450 or 500 mg/kg.
In one embodiment, the DHODH inhibitor is administered at a dose of 100 mg/kg.
In one embodiment, the DHODH inhibitor is administered at a dose of 100 mg/kg, once daily.
In one embodiment the breast cancer therapy further comprises a pan-HER inhibitor [R)- N4- [3-Chloro-4- (thiazol2-ylmethoxy]-phenyl]-N6- (4-methyl-4,5,-dihydro-oxazol-2-yl]- quinazoline-4,6-diamine or a pharmaceutically acceptable salt thereof.
In one embodiment the pan-HER inhibitor is administered parenterally.
In one embodiment the pan-HER inhibitor is administered orally, for example is administered bi-daily.
Also provided is 2-(3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof for use in the treatment of a breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as triple negative breast cancer.
In a further aspect there is provided 2- (3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof as part of a combination therapy for use in the treatment of a breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same.
The disclosure also extends to a combination therapy comprising pan-HER inhibitor (such as (i?]-N4- [3-Chloro-4-(thiazol2-ylmethoxy]-phenyl]-N6-(4-methyl-4,5,-dihydro-oxazol-2-yl]-
quinazoline-4,6-diamine or a pharmaceutically acceptable salt thereof) and a DHODH inhibitor 2- (3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof for use in the treatment of a breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as a triple negative negative breast cancer.
In a further aspect there is provided use of a DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt in the manufacture of a therapy for the treatment of breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as a triple negative breast cancer.
In a further aspect there is provided use of a DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt in the manufacture of a combination therapy for the treatment of breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same, such as triple negative breast cancer.
Also provided is use of a pan-HER inhibitor (such as i?]-N4-[3-Chloro-4-(thiazol2- ylmethoxy]-phenyl]-N6-(4-methyl-4,5,-dihydro-oxazol-2-yl]-quinazoline-4,6-diamine or a pharmaceutically acceptable salt thereof) and a DHODH inhibitor 2-(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt in the manufacture of a combination therapy for the treatment of a breast cancer, in particular a breast cancer disclosed herein which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same.
In one embodiment the therapy of the present disclosure continues for 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60 months or more.
In one embodiment dosing of the second therapy (such as Varlitinib] continues for 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60 months or more.
(i?]-N4-[3-Chloro-4-(thiazol2-ylmethoxy]-phenyl]-N6-(4-methyl-4,5,-dihydro-oxazol-2-yl]- quinazoline-4,6-diamine is also known as Varlitinib. In one embodiment Varlitinib is administered in a 28 day cycle.
DETAILED DISCLOSURE
DHODH is a key enzyme in the production of uridine, which is a central building block in the cell. Whilst not wishing to be bound by theory it may be that the DHODH inhibitor is able to upregulate p53 based apoptosis. The up-regulation of p53 (which may lead to cell cycle arrest and at higher levels of p 53, to apoptosis] is likely to occur via mechanisms sensing the levels of intracellular uridine, and then setting forth a series of reactions leading to stabilisation of p53, and increasing its concentration (also referred to herein as p53 induction].
The present inventors have established that DHODH inhibitors, such as -(3,5-difluoro- 3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof may be particularly advantageous for use in the treatment of breast cancers because they kill cancer cells via apoptosis, which does not involve necrotic cell death.
Necrosis is a form of cell injury which results in premature death of cells in living tissue by autolysis (i.e. destruction of the cell through the action of its own enzymes]. Necrosis is caused by factors external to the cell or tissue, such as infection, toxins, or trauma which result in the unregulated digestion of cell components. In contrast, apoptosis is a naturally occurring programmed and targeted cause of cellular death.
Apoptosis often provides beneficial effects to the organism, whereas necrosis is almost always damaging to the surrounding tissue. Furthermore, necrotic cell death does not follow the apoptotic signal transduction pathway, but rather various receptors are activated, and result in the loss of cell membrane integrity and an uncontrolled release of products of cell death into the extracellular space.
This initiates in the surrounding tissue an inflammatory response which attracts leukocytes and nearby phagocytes which eliminate the dead cells by phagocytosis. However, pathogen damaging substances released by leukocytes create collateral damage to surrounding tissues. This excess collateral damage inhibits the healing process. Thus, untreated necrosis results in a build-up of decomposing dead tissue and cell debris at or near the site of the cell death. A classic example is gangrene. For this reason, it is often necessary to remove necrotic tissue surgically, a procedure known as debridement.
Thus, the ability to treat breast cancers with a DHODH inhibitor that causes apoptotic cell death is likely to lead to less side effects and an overall better therapeutic outcome.
Advantageously, the DHODH inhibitor herein may be beneficial in the therapy according to the present disclosure because the biological effects are stronger than those of "first generation" DHODH inhibitors, such as leflunomide and teriflunomide.
In addition, the DHODH inhibitor of the present disclosure may have less toxicity (off target effects] than the so-called "first generations" inhibitors, such as leflunomide and teriflunomide.
Furthermore, after administration of a therapeutic dose full expose exposure is achieved with 24 hours, with a half-life of about 18 hours.
In one embodiment the DHODH is employed in a combination therapy with a biological therapeutic agent, such as an antibody or binding fragment thereof. In one embodiment the biological therapeutic agent is administered parenterally.
Employing the DHODH inhibitor as a combination therapy, for example wherein the second therapy is an inhibitor of the DNA repair and/or a pan-HER inhibitor may be particular beneficial to minimises the cancers ability to resist treatment, in particular by "attacking" the cancer cells by two or more mechanisms with the combination therapy. By employing an inhibitor of DNA repair
for example, the sensitivity of a tumor cell to DNA damage caused by anti-cancer agents and thus DNA damage-induced cell death is enhanced.
In one embodiment the PARP inhibitor is one or more compounds independently selected from of the followin :
MP- 124 a PARP-
ABT-737 a PARP-1 and PARP-2 inhibitor,
or a pharmaceutically acceptable salt or solvate of any one of the same.
Topoisomerase inhibitors, which may be employed in a method of the present disclosure include type I topoisomerase inhibitors, type II topoisomerase inhibitors and type II
topoisomerase poisons. Type I inhibitors include topotecan, irinotecan, indotecan and indimitecan. Type II inhibitors include genistein and ICRF 193 which has the following structure:
Type II poisons include amsacrine, etoposide, etoposide phosphate, teniposide and doxorubicin and fluoroquinolones.
In one embodiment the DHODH inhibitor of the present disclosure is employed in a combination therapy comprising chemotherapy, in particular a described herein.
In one embodiment there is provided a method of treating a patient comprising administering a therapeutically effective amount of an inhibitor of a pan-HER inhibitor and a therapeutically effective amount of DHODH inhibitor.
In one embodiment the pan-HER inhibitor is an organic chemistry molecule, for example with a molecular weight of 500 or less.
In one embodiment the pan-HER inhibitor has a molecular formula of formula (I):
wherein the substituents are disclosed in WO2005/016346 fully incorporated herein by reference In one embodiment the pan-HER inhibitor is an inhibitor of one or more HER receptors independently selected from HER1, HER2, HER3, HER4.
In one embod (la):
In one embodiment
In one embodiment Varlitinib is employed as a free base.
Varlitinib at an appropriate dose is capable of inhibiting HERl, HER2 and HER4 directly and thought to be capable of inhibiting HER3 indirectly.
In one embodiment the compound of formula (I] (including formula (la] and Varlitinib] at least inhibits the activity of HERl and HER2, HERl and HER4 or HER2 and HER4.
In one embodiment the compound of formula (I] (including formula (la] and Varlitinib] at least inhibits the activity of HERl, HER2 and HER4, for example directly inhibits the activity of HERl, HER2 and HER4.
In one embodiment the compound of formula (I] (including formula (la] and Varlitinib] inhibits the activity of HERl, HER2, HER3 and HER4, for example directly inhibits the activity of HERl, HER2, and HER4, and indirectly inhibits the activity of HER3
In one embodiment each dose of the compound of formula (I], (including formula (la] and Varlitinib] is in the range 100 to 900mg, for example each dose is in the range of 300 to 500mg, such as 400mg, for example administered once or twice daily, such as twice daily.
In some instances, patients may benefit from having the initial dose reduced to 300mg or 200mg bi-daily.
Other patients may benefit from receiving the compound of formula (I], such as Varlitinib, for example, in a regime which is non-continuous, such as taking medication on alternate days instead of each day or taking medication for four sequential days followed by one, two or three days without medication.
In one embodiment the compound of formula (I], (including formula (la] and Varlitinib] is administered orally.
In one embodiment the pan-HER inhibitor is a combination of HER inhibitors.
In one embodiment the pan-HER inhibitor such as Varlitinib is administered twice daily, for example a dose in the range disclosed herein.
The DHODH inhibitor is 2- (3, 5-difluoro-3'-methoxybiphenyl-4-ylamino] nicotinic acid
(referred to herein as ASLAN003] or a pharmaceutically acceptable salt thereof, which has the structure:
In one embodiment the DHODH inhibitor is administered daily, for example once daily.
In one embodiment the DHODH inhibitor is administered orally.
In one embodiment the DHODH inhibitor and the second therapy (for example a pan-HER inhibitor such as particular Varlitinib or chemotherapy] are administered sequentially in a treatment regimen, for example are administered on the same day.
In one embodiment the DHODH inhibitor and the second therapy (for example a HER inhibitor, such as the pan HER inhibitor or chemotherapy] are administered simultaneously, at approximately the same time.
In one embodiment the DHODH inhibitor is administered in regimen that is daily or weekly for a continuous period of time for example 1 to 60 months or more.
The second therapy (for example the pan-HER inhibitor such as Varlitinib or chemotherapy] may be administered intermittently during this period, and for example may be administered in a one or more 28 days cycles.
Where the second therapy (for example the pan-HER inhibitor] comprises an antibody molecule then the administration protocol is likely to be very different to that of small molecule inhibitors. An antibody or binding fragment may be administered (in particular in combination with cytotoxic chemotherapy] in a regime as follows:
1] First administration 4mg/Kg over 90 minute; and
2] Weekly administrations of 2mg/Kg over 30 minutes for the next 12 weeks; &
3] One week post 2] initiation of 6mg/Kg over 30-90 minutes every three weeks.
In one embodiment the second therapy (for example a pan-HER inhibitor or chemotherapy] is administered in regimen that is daily or weekly for a continuous period of time, for example 1 to 60 months or more, and the DHODH inhibitor is administered intermittently during this period.
Administered intermittently as employed herein refers to a period wherein the therapy is administered and then stopped with the option of starting the therapy again at some point in the future, for example in accordance with a treatment plan.
In one embodiment the DHODH inhibitor is administered in regimen that is daily or weekly for a continuous period of time, for example 1 to 60 months or more, and the second therapy, (for example a pan-HER inhibitor or chemotherapy] is administered in regimen that is daily or weekly for a continuous period of time, for example 1 to 60 months or more.
In one embodiment the DHODH inhibitor is administered in regimen that is daily or weekly continuously over for example 1 to 60 months or more, and the second therapy (for example a pan- HER inhibitor or chemotherapy] is administered conjunctly with DHODH inhibitor in regimen that is daily or weekly for intermittent periods over, for example 1 to 60 months or more.
In one embodiment the DHODH inhibitor is administered in regimen that is daily or weekly for intermittent periods over for example 1 to 60 months or more, and the second therapy (for example a pan-HER inhibitor or chemotherapy] is administered conjunctly with DHODH inhibitor in regimen that is daily or weekly for intermittent periods over, for example 1 to 60 months or more.
In one embodiment the DHODH inhibitor and the second therapy (for example a pan-HER inhibitor] are co-formulated.
In one embodiment the DHODH inhibitor is administered orally.
In one embodiment the second therapy is administered orally and/or administered parenterally, such as intravenously.
In one embodiment the DHODH inhibitor and the second therapy are both administered orally.
In one embodiment the DHODH inhibitor is administered orally and the second therapy is administered parenterally.
In one embodiment the patient is a human.
In one embodiment the therapy of the present disclosure is for example beneficial in that it provides augmented therapeutic activity in comparison to therapies currently employed for breast cancer, such as triple negative breast cancer.
Augmented activity may be any beneficial therapeutic effect of employing the therapy of the present disclosure, for example an increase in anti-tumor activity and/or a reduced propensity for the cancer to become resistant Other benefits may be therapeutic effect in patients who have failed one or more lines of therapy. Thus, in one embodiment the patient population has a cancer that is resistant or refractory to known therapies, such as cytotoxic chemotherapy.
Unless the context indicates otherwise refractory and resistant are used to interchangeably herein to refer to where the cancer does not respond to therapy or responds poorly to therapy.
Combination therapy as employed herein refers to two or more modes of therapy being employed over the same treatment period, i.e. the opposite of sequential therapy.
Two or more modes of therapy as employed herein refers to at least two therapies which have different modes of action and/or different activities and/or different routes of administration.
To obtain the benefits of a combination therapy of the present disclosure the DHODH inhibitor and the second therapy have to administered in a time frame, where the pharmacological effects of a both therapies overlap, i.e. the treatment regimens for the said therapies partly coincide in time. A skilled person will understand in practice what this means.
Combination therapy as employed herein refers to where a medicament according to the present disclosure is administered in a treatment regimen along with at least one further therapeutic agent. The regime may be separate formulations administered at the same time or different times or co-formulations of the two or more therapeutic agents. The "first" medicament employed in the combination therapy according to the present disclosure may be administered; prior to the further therapeutic agent or agents, concomitant with the further therapeutic agent or agents, or after the further therapeutic agent or agents.
In one embodiment further therapeutic agent or agents, such as an anti-cancer therapy are employed in combination with the therapy of the present disclosure.
In one embodiment the therapeutic agent is a chemotherapeutic agent Chemotherapeutic agent as employed herein is intended to refer to specific antineoplastic chemical agents or drugs that are destructive to malignant cells and tissues, including alkylating agents, antimetabolites, anthracyclines, plant alkaloids, topoisomerase inhibitors, and other antitumour agents. Specific examples of chemotherapy include doxorubicin, 5-fluorouracil (5-FU], paclitaxel (for example
abraxane or docetaxel], capecitabine, irinotecan, and platins, such as cisplatin and oxaliplatin or a combination thereof. A suitable dose may be chosen by the practitioner based on the nature of the cancer being treated and the patient
Co-administered as employed herein refers to administration of the DHODH inhibitor and the second therapy at the same time or approximately the same time (including where the actives are administered by the same or different routes].
Inhibitor as employed refers to the reduction of a relevant biological activity, for example by 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95 or 100%, such as when measured in a relevant in vitro assay.
Direct inhibition is where the inhibitor binds directly to or physically blocks a binding interaction to inhibit a biological activity, or when the inhibitor inhibits the activation through phosphorylation of the target molecule.
Indirect inhibition as employed herein refers to where the biological activity in question is inhibited as a result of directly inhibiting a target that is other than the entity that is indirectly inhibited.
Dihydroorotate dehydrogenase (DHODH] is the enzyme that catalyzes the fourth step in the pyrimidine biosynthetic pathway namely the conversion of dihydroorotate to orotate concomitantly with an electron transfer to ubiquinone (cofactor Q] via a flavin mononucleotide intermediate (Loffler Mol Cell Biochem, 1997]. In contrast to parasites [Plasmodium falciparum) (McRobert et al Mol Biochem Parasitol 2002] and bacteria [E.coli) which exclusively have this de novo pathway as the source of pyrimidines, mammal cells have an additional salvage pathway.
During homeostatic proliferation, the salvage pathway, which is independent of DHODH, seems sufficient for the cellular supply with pyrimidine bases. However in cells with a high turnover and particularly T and B lymphocytes the de novo pathway is required to proliferate. In these cells, DHODH inhibition stops the cell cycle progression by suppressing DNA synthesis and ultimately cell proliferation (Breedveld F.C. Ann Rheum Dis 2000].
There are some suggestions that inhibition of mitochondrial cytochrome bcl, a component of the electron transport chain complex III, leads to activation of tumor suppressor p53, followed by apoptosis induction. The mitochondrial respiratory chain is coupled to the de novo pyrimidine biosynthesis pathway via the mitochondrial enzyme dihydroorotate dehydrogenase (DHODH].
The p53 activation has been shown to be triggered by the impairment of the de novo pyrimidine 15 biosynthesis due to the suppression of DHODH.
A DHODH inhibitor as employed herein refers to a compound which inhibits the activity of dihydroorotate dehydrogenase, in particular in vivo. ASLAN003 is disclosed in WO2008/077639, incorporated herein by reference.
A biological therapeutic is one based on a protein, for example an antibody or binding fragment thereof, including fusion proteins and biological molecules conjugated to a polymer, toxin or similar payload.
A "drug" as employed herein refers to a chemical entity, organic chemistry molecule with pharmacological activity.
An example of a biological therapeutic conjugated to a payload is trastuzumab emtansine.
Examples of pharmaceutically acceptable salts include but are not limited to acid addition salts of strong mineral acids such as HC1 and HBr salts and addition salts of strong organic acids, such as a methansulfonic acid salt, tosylates, furoates and the like, including di, tri salts thereof, such as ditosylates.
In one embodiment the combination therapy according to the present disclosure further comprises a RON inhibitor, for example as disclosed WO2008/058229, incorporated herein by reference.
In one embodiment the combination therapy of the present disclosure comprises a checkpoint inhibitor, such as a CTLA4 inhibitor, a PD-1 inhibitor or a PD-Ll inhibitor, in particular an antibody or binding fragment thereof.
In one embodiment the combination therapy of the present disclosure further comprises a chemotherapeutic agent
Chemotherapeutic Agents
The DHODH therapy of the present disclosure (including a combination therapy disclosed herein] may be employed in combination with a further cancer therapy, for example chemotherapy.
Chemotherapeutic agent and chemotherapy or cytotoxic agent are employed interchangeably herein unless the context indicates otherwise.
Chemotherapy as employed herein is intended to refer to specific antineoplastic chemical agents or drugs that are "selectively" destructive to malignant cells and tissues, for example alkylating agents, antimetabolites including thymidylate synthase inhibitors, anthracyclines, anti- microtubule agents including plant alkaloids, topoisomerase inhibitors, parp inhibitors and other antitumour agents. Selectively in this context is used loosely because of course many of these agents have serious side effects.
The preferred dose may be chosen by the practitioner, based on the nature of the cancer being treated.
Examples of alkylating agents, which may be employed in the method of the present disclosure include an alkylating agent nitrogen mustards, nitrosoureas, tetrazines, aziridines, platins and derivatives, and non-classical alkylating agents.
Examples of a platinum containing chemotherapeutic agent (also referred to as platins], such as cisplatin, carboplatin, oxaliplatin, satraplatin, picoplatin, nedaplatin, triplatin and lipoplatin (a 25 liposomal version of cisplatin], in particular cisplatin, carboplatin and oxaliplatin.
The dose for cisplatin ranges from about 20 to about 270 mg/m2 depending on the exact cancer. Often the dose is in the range about 70 to about 100mg/m2.
Nitrogen mustards include mechlorethamine, cyclophosphamide, melphalan, chlorambucil, ifosfamide and busulfan.
Nitrosoureas include N-Nitroso-N-methylurea (MNU], carmustine (BCNU], lomustine (CCNU] and semustine (MeCCNU], fotemustine and streptozotocin. Tetrazines include dacarbazine, mitozolomide and temozolomide.
Aziridines include thiotepa, mytomycin and diaziquone (AZQ].
Examples of antimetabolites, which may be employed in the therapy of the present disclosure, include anti-folates (for example methotrexate and pemetrexed], purine analogues (for example thiopurines, such as azathiopurine, mercaptopurine, thiopurine, fludarabine (including the phosphate form], pentostatin and cladribine], pyrimidine analogues (for example fluoropyrimidines, such as 5-fluorouracil and prodrugs thereof such as capecitabine [Xeloda®]], floxuridine, gemcitabine, cytarabine, decitabine, raltitrexed(tomudex] hydrochloride, cladribine and 6-azauracil.
Examples of anthracyclines, which may be employed in the method of the present disclosure, include daunorubicin (Daunomycin], daunorubicin (liposomal], doxorubicin (Adriamycin], doxorubicin (liposomal], epimbicin, idambicin, valmbicin currenlty used only to treat bladder cancer and mitoxantrone an anthracycline analog, in particular doxorubicin.
Examples of anti-microtubule agents, which may be employed in the therapy of the present disclosure, include include vinca alkaloids and taxanes.
Vinca alkaloids include completely natural chemicals for example vincristine and vinblastine and also semi-synthetic vinca alkaloids, for example vinorelbine, vindesine, and vinflunine
Taxanes include paclitaxel, docetaxel, abraxane, carbazitaxel and derivatives of thereof. Derivatives of taxanes as employed herein includes reformulations of taxanes like taxol, for example in a micelluar formulations, derivatives also include chemical derivatives wherein synthetic chemistry is employed to modify a starting material which is a taxane.
Topoisomerase inhibitors are described above.
In one embodiment a combination of chemotherapeutic agents employed is, for example a platin and 5-FU or a prodrug thereof, for example cisplatin or oxaplatin and capecitabine or gemcitabine, such as FOLFOX.
In one embodiment the chemotherapy comprises a combination of chemotherapy agents, in particular cytotoxic chemotherapeutic agents.
In one embodiment the chemotherapy combination comprises a platin, such as cisplatin and fluorouracil or capecitabine.
In one embodiment the chemotherapy combination is capecitabine and oxaliplatin (Xelox]. In one embodiment the chemotherapy is a combination of folinic acid and 5-FU, optionally in combination with oxaliplatin.
In one embodiment the chemotherapy is a combination of folinic acid, 5-FU and irinotecan (FOLFIRI], optionally in combination with oxaliplatin (FOLFIRINOX]. The regimen consists of: irinotecan (180 mg/m2 IV over 90 minutes] concurrently with folinic acid (400 mg/m2 [or 2 x 250 mg/m2] IV over 120 minutes]; followed by fluorouracil (400-500 mg/m2 IV bolus] then fluorouracil (2400-3000 mg/m2 intravenous infusion over 46 hours]. This cycle is typically repeated every two weeks. The dosages shown above may vary from cycle to cycle.
In one embodiment the chemotherapy combination employs a microtubule inhibitor, for example vincristine sulphate, epothilone A, N- [2-[(4-Hydroxyphenyl]amino]-3-pyridinyl]-
4methoxybenzenesulfonamide (ABT-751], a taxol derived chemotherapeutic agent, for example paclitaxel, abraxane, or docetaxel or a combination thereof.
In one embodiment the chemotherapy combination employs an mTor inhibitor. Examples of mTor inhibitors include: everolimus (RAD001], WYE-354, KU-0063794, papamycin (Sirolimus], Temsirolimus, DeforolimusfMK-8669), AZD8055 and BEZ235 (NVP-BEZ235).
In one embodiment the chemotherapy combination employs a MEK inhibitor. Examples of MEK inhibitors include: AS703026, CI-1040 (PD184352), AZD6244 (Selumetinib], PD318088, PD0325901, AZD8330, PD98059, U0126-EtOH, BIX 02189 or BIX 02188.
In one embodiment the chemotherapy combination employs an AKT inhibitor. Examples of AKT inhibitors include: MK-2206 and AT7867.
In one embodiment the combination employs an aurora kinase inhibitor. Examples of aurora kinase inhibitors include: Aurora A Inhibitor I, VX-680, AZD1152-HQPA (Barasertib], SNS314 Mesylate, PHA-680632, ZM-447439, CCT129202 and Hesperadin.
In one embodiment the chemotherapy combination employs a p38 inhibitor, for example as disclosed in WO2010/038086, such as iV- [4-({4-[3-(3-tert-Butyl-l-p-tolyl-lH-pyrazol- 5yl]ureido]naphthalen-l-yloxy}methyl]pyridin-2-yl]-2-methoxyacetamide.
In one embodimentthe combination employs a Bcl-2 inhibitor. Examples of Bcl-2 inhibitors include: obatoclax mesylate, ABT-737, ABT-263 (navitoclax] and TW-37.
In one embodiment the chemotherapy combination comprises an antimetabolite such as capecitabine (xeloda], fludarabine phosphate, fludarabine (fludara], decitabine, raltitrexed (tomudex], gemcitabine hydrochloride and cladribine.
In one embodiment the chemotherapy combination comprises ganciclovir, which may assist in controlling immune responses and/or tumour vasculation.
In one embodiment one or more therapies employed in the method herein are metronomic, that is a continuous or frequent treatment with low doses of anticancer drugs, often given concomitant with other methods of therapy.
In one embodiment, there is provided the use of multiple cycles of treatment (such as chemotherapy] for example 2, 3, 4, 5, 6, 7, 8 cycles.
In one embodiment the therapy of the present disclosure is employed after chemotherapy. In one embodiment the therapy of the present disclosure is employed before chemotherapy.
In one embodiment the dose of chemotherapy employed in the therapy of the present disclosure is lower than the dose of chemotherapy employed in "monotherapy" (where monotherapy may include the dose of chemotherapy employed when combinations of chemotherapy agents are employed].
In one embodiment the medicament is administered in combination with therapy complimentary to the cancer therapy, for example a treatment for cachexia, such as cancer cachexia, for example S-pindolol, S-mepindolol or S-bopindolol. Suitable doses may be in the range of 2.5mg to lOOmg, such as 2.5mg to 50mg per day provided a single dose or multiple doses given as multiple doses administered during the day.
Treatment
Treatment as employed herein refers to where the patient has a disease or disorder, for example cancer and the medicament according to the present disclosure is administered to stabilise the disease, delay the disease, amelorate the disease, send the disease into remission, maintain the disease in remission or cure the disease. Treating as employed herein includes administration of a medicament according to the present disclosure for treatment or prophylaxis. The present disclosure is explained in the context of a method of treating a patients. However, the disclosure extends to use of the combination therapy as described herein for use in treatment, in particular for the treatment of cancer, such as a cancer described herein. Also provided is use of the combination of compounds as described herein for the manufacture of a medicament for the treatment of cancer, in particular a cancer described herein.
In one embodiment the combination therapy according to the present disclosure is employed as cancer adjuvant therapy, for example after surgery to remove some or all of the cancerous cells.
In one embodiment the combination according to the present disclosure is employed as neoadjuvant therapy, for example before surgery to remove some or all of the cancerous cells. In one embodiment a therapeutically effective dose (such as a daily dose] of a DHODH inhibitor is in the range lOmgto lOOOmg, for example 50 to 500mg, such as 100, 150, 200, 250, 300, 350, 00, 50, 500mg, in particular administered once a day.
In one embodiment the DHODH inhibitor, for example 2-(3,5-difluoro-3'methoxybiphenyl-
4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof is administered bi-daily, such as at a dose in the range lOOmgto 900mgon each occasion, in particular 300mg, or 400mgor 500mg each dose.
Generally active ingredients employed in the therapy of the present disclosure will be provided in the form of a pharmaceutical formulation comprising one or more excipients, diluents or carriers.
In one embodiment the DHODH inhibitor, for example 2-(3,5-difluoro-3'methoxybiphenyl- 4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof is administered as pharmaceutical formulation comprising one or more pharmaceutically acceptable excipients.
In the context of this specification "comprising" is to be interpreted as "including".
Embodiments of the invention comprising certain features/elements are also intended to extend to alternative embodiments "consisting" or "consisting essentially" of the relevant elements/features.
Where technically appropriate, embodiments of the invention may be combined.
Technical references such as patents and applications are incorporated herein by reference.
Any embodiments specifically and explicitly recited herein may form the basis of a disclaimer either alone or in combination with one or more further embodiments.
Headings herein are employed to divide the document into sections and are not intended to be used to construe the meaning of the disclosure provided herein.
BRIEF DESCRIPTION OF FIGURES
Figure 1. HCI-001 treatment results. (A] Average tumor volumes for all 4 groups vs Vehicle,
(B] Vehicle vs ASLAN003, (C] Vehicle vs Doxorubicin, (D] Vehicle vs combination of ASLAN003 + Doxorubicin
Figure 2. HCI-002 treatment results. (A] Average tumor volumes for all 4 groups vs Vehicle,
(B] Vehicle vs ASLAN003, (C] Vehicle vs Doxorubicin, (D] Vehicle vs combination of ASLAN003 + Doxorubicin
Figure 3. HCI-009 treatment results. (A] Average tumor volumes for all 4 groups vs Vehicle,
(B] Vehicle vs ASLAN003, (C] Vehicle vs Doxorubicin, (D] Vehicle vs combination of ASLAN003 + Doxorubicin
Figure 4. HCI-004 treatment results. (A] Average tumor volumes for all 4 groups vs Vehicle,
(B] Vehicle vs ASLAN003, (C] Vehicle vs Doxorubicin, (D] Vehicle vs combination of ASLAN003 + Doxorubicin
EXAMPLES
Example 1
ASLAN003 is a small molecule inhibitor of Dihydroorotate Dehydrogenase (DHODH]. In studies conducted to date, ASLAN003 has generated promising emerging preclinical data in oncology indications, where the mechanism is linked with induction of p53 and apoptosis.
We tested ASLAN003 alone (lOOmg/Kg daily, oral gavage], doxorubicin alone (1 or 10 mg/kg once per week, intravenous], and the combination of ASLAN003 and doxorubicin treatments for their ability to affect orthotopic growth of tumors in the mammary fat pad, using four human breast cancer PDX lines in NRG mice. The PDX lines were all triple negative breast cancers and were selected to have varying TP53 and PTEN mutation status. 2. Objectives and Study Design
We hypothesized that ASLAN003 would reduce triple negative breast tumor growth, particularly when combined with doxorubicin. This would potentially give rise to a new therapeutic regimen and/or allow for reduction of doxorubicin doses to reduce risks of side effects attributed to doxorubicin, such as cardiotoxicity.
To test this hypothesis, we conducted experiments using 4 triple negative breast cancer
(TNBC) PDX lines with/without mutations in PTEN and/or TP53 (Table 1). We tested ASLAN003 +/- doxocirubicin for in 4 treatment groups for each line (12mice/group]: (A] vehicle control; (B] ASLAN003 alone; (C] doxorubicin alone; and (D] ASLAN003 + doxorubicin. Mice were randomized to drug treatment groups when tumors reached approximately 100 mm3 (rolling enrollment].
Table 1. Huntsman Cancer Institute Human Breast Cancer POX models used in this study
3. Methods
Drugs:
1. ASLAN003 : Prepared fresh every day: 10 mg/mL in 30% PEG300+30% Captisol, sonicated for 1.5 hrs before use.
2. Doxorubicin 10X stock (Selleck Chemical Cat# S1208] : 20mg/ml in H2O. Stored at -80C in 0.5mL aliquots. Working solution prepared fresh by diluting 1 : 10 in H2O (for high dose] or 1 : 100 in H20 (for low dose].
PDX tumor transplantation:
1. Frozen tissue fragments (~4x2x2 mm] were thawed by placing the vial in a 37 ° C water bath and removed before the medium completely melted.
2. The outside of the vial was sprayed with 70% ethanol, then the vial was moved into a biosafety hood and the contents transferred into a 50-mL conical tube.
3. 10 mL of HBEC medium was added, then aspirated to remove the DMSO-containing freezing medium.
4. Step 3 was repeated three times.
5. Fresh HBEC medium was added, the lid was closed, and the tube placed on ice until the implantation.
6. 1 piece of tumor fragment was transplanted into the right mammary fat pad of each 3-4wk old NRG mouse (total 48mice/each PDX line]. The extra fat pad was cleared around the nipple and lymph node.
Treatment: When the tumor volume reached 100-200 mm3, mice were randomized into 1 of 4 treatment groups:
Experiment ! fHCI-0011:
(A] Vehicle (30% PEG300+30% Captisol] : 10 uL/g; oral gavage; continuously QD for 17 days
(B] ASLAN003 : 100 mg/kg; oral gavage; continuously QD for 17 days
(C] Doxorubicin: 10 mg/kg IV once a week over the 17 day treatment period (2 doses]
(D] ASLAN003+Doxorubicin: The same doses/routes as B and C for 17 days treatment
Experiment 2 fHCI-0021:
(A] Vehicle (30% PEG300+30% Captisol] : 10 uL/g; oral gavage; continuously QD for 17 days
(B] ASLAN003 : 100 mg/kg; oral gavage; continuously QD for 17 days
(C] Doxorubicin: 10 mg/kg IV once a week over the 17 day treatment period (2 doses]
(D] ASLAN003+Doxorubicin: The same doses/routes as B and C for 17 days treatment
Experiment 3 fHCI-0091:
(A] Vehicle (30% PEG300+30% Captisol) : 10 uL/g; oral gavage; continuously QD for 17 days
(B] ASLAN003 : 100 mg/kg; oral gavage; continuously QD for 17 days
(C] Doxorubicin: 10 mg/kg IV once a week over the 17 day treatment period (2 doses]
(D] ASLAN003+Doxorubicin: The same doses/routes as B and C for 17 days treatment
Experiment 4 fHCI-0041:
(A] Vehicle (30% PEG300+30% Captisol] : 10 uL/g; oral gavage; continuously QD for 29 days
(B] ASLAN003 : 100 mg/kg; oral gavage; continuously QD for 29 days
(C] Doxorubicin: 1 mg/kg IV once a week over the 29 day treatment period (4 doses]
(D] ASLAN003+Doxorubicin: The same doses/routes as B and C for 29 days treatment
Tumor monitoring: Tumors were measured for length and width every other day and recorded. Tumor volumes were calculated with the formula V = ((WA2] L]/2. 4. Results
Experiment 1: HCIOOl (PTEN and TP53 mutant)
We did not observe any evidence of tumor shrinkage in any of the treatment groups but tumor growth in all treatment groups was significantly slower than the vehicle control group (Figure 1]. No difference was observed between single treatments and the combination group. Some toxicity (weight loss] was observed in doxorubicin only and combination groups but not in ASLAN003 only, so this was attributed to doxorubicin.
Experiment 2: HCI002 (PTEN and TP53 WT)
This tumor appeared to be more responsive to doxorubicin than ASLAN003 when used as single agents. Combination treatment was similar to doxorubicin alone (Figure 2]. Experiment 3: HCI009 (PTEN mutant; TP53 WT)
Tumors behaved similarly to HCI-001 : either ASLAN003 or doxorubicin was effective as a single agent, with no benefit of combination treatment (no tumor shrinkage] (Figure 3].
Experiment 4: HCI004 (PTEN WT; TP53 mutant)
For this experiment we reduced the dose of doxorubicin 10-fold, from 10 mg/kg to 1 mg/kg, in order to determine if an additive effect with ASLAN003 would be more apparent with lower dose doxorubicin. However, tumors still behaved similarly to HCI-001 (Experiment 1] and HCI-009 (Experiment 3] : either ASLAN003 or doxorubicin was effective as a single agent, with no benefit of combination treatment (no tumor shrinkage] (Figure 4]. 5. Conclusions
1. ASLAN003 QD dosing on a continuous basis prevented tumor growth (sustained stable disease] in all 4 TNBC PDX models tested, for the duration of treatment
Claims
1. A method of treating a breast cancer, which is negative for a marker selected from ER, PR, HER2 and a combination of two or more of the same (such as ER & PR, ER & HER2, PR & HER2 or ER, PR & HER2 (triple negative]] said treatment comprising administering a therapeutically effect amount of a DHODH inhibitor 2-(3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof.
2. A method of treatment according to claim 1, wherein the DHODH inhibitor is employed in a combination therapy with a second therapy.
3. A method of treatment according to claim 2 wherein the second therapy is an inhibitor of DNA repair.
4. A method of treatment according to claim 2 or 3, wherein the inhibitor is a small molecule therapy.
5. A method of treatment according to claim 3 or 4, wherein the inhibitor mechanism is via the base excision repair pathway.
6. A method of treatment according to claim 5, wherein the inhibitor's target is independently selected from APE1, Pol β, FEN1, and PARP.
7. A method of treatment according to claim 5 or 6, wherein the inhibitor is selected from TRC102, (2E]-2-[(4,5-Dimethoxy-2-methyl-3,6-dioxo-l,4-cyclohexadien-l-yl]methylene]- undecanoic acid [also known as E3330], NCS-666715 and NSC-124854, 8-oxoguamine, tanespirmycin, luminespib, alvespimycin, genetespib, retaspimycin, 6-Amino-8-[(6-iodo-l,3- benzodioxol-5-yl]thio]-N-(l-methylethyl]-9H-purine-9-propanamine (PU-H71], 4-[2- carbamoyl-5- [6,6-dimethyl-4-oxo-3-(trifluoromethyl]-5,7-dihydroindazol-l- yl]anilino]cyclohexyl] 2-aminoacetate(SNX-5422], luminespib (resorcyinylic], 2-(2-ethyl-3,5- dihydroxy-6-(3-methoxy-4-(2-morpholinoethoxy]benzoyl]phenyl]-N,N-bis(2- methoxyethyl]acetamide (KW-2478], AT13387, 5,6-bis((E]-benzylideneamino]-2-thioxo-2,3- dihydropyrimidin-4(lH]-one (SCR7] and combinations of two or more of the same.
8. A method of treatment according to any one of claims 5 to 7, wherein the inhibitor is a PARP inhibitor, such as a PARP-1 and/or PARP-2 inhibitor.
9. A method of treatment according to claim 8, wherein the PARP inhibitor is independently selected from olaparib, rucaparib, niraparib, iniparib, talazoparib, veliparib, CEP9722, E7016, BGB-290, AZD-2461, 3-aminobenzamide and combinations thereof.
10. A method of treatment according to any one of claims 3 to 9 wherein the inhibitor mechanism is via the mismatch repair pathway.
11. A method of treatment according to any one of claims 3 to 10, wherein the inhibitor mechanism is via the nucleotide excision pathway.
12. A method of treatment according to claim 11, wherein the inhibitor is independently selected from 7-hydroxystaurosporine [UCN-01], trabectedin, MCI13E, NERI01 and combinations of two or more of the same.
13. A method of treatment according to any one of claims 3 to 12, wherein the inhibitor mechanism is via the double stranded break repair pathway.
14. A method of treatment according to claim 13, wherein the inhibitor mechanism is via the nonhomologous end joining pathway.
15. A method of treatment according to claim 13 or 14, wherein the inhibitor mechanism is via the homologous recombination pathway.
16. A method of treatment according to any one of claims 2 to 15, wherein the second therapy is a topoisomerase inhibitor, such as topoisomerase I and/or II inhibitor.
17. A method of treatment according to claim 16, wherein the topoisomerase inhibitor is independently selected from irinotecan, topotecan, camptothecin lamellarin D and combinations thereof.
18. A method of treatment according to claim 16 or 17, wherein the topoisomerase inhibitor is independently selected from etoposide (VP- 16], teniposide, doxorubicin, daunorubicin, mitoxantrone, amsacrine, ellipticines, aurintricarboxylic acid, 3-Hydroxy-2-[(li?]-6- isopropenyl-3-methyl-cyclohex-2-en-l-yl]-5-pentyl-l,4-benzoquinone (HU-331] and combinations thereof.
19. A method according to any one of claims 1 to 18, wherein the treatment further comprises doxorubicin.
20. A method according to any one of claims 1 to 19, wherein the DHODH inhibitor provides anticancer efficacy via induction of p53.
21. A method according to any one of claims 1 to 20, wherein the DHODH inhibitor is administered orally, for example once daily.
22. A 2-(3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt thereof for use in the treatment of a breast cancer, which in negative for a marker selected from ER, PR, HER 2 and a combination of two or more of the same (such as ER & PR, ER & HER2, PR & HER2 or triple negative].
23. Use of a DHODH inhibitor 2-(3,5-difluoro-3'methoxybiphenyl-4-ylamino]nicotinic acid or a pharmaceutically acceptable salt in the manufacture of a therapy for the treatment of a breast cancer, which in negative for a marker selected from ER, PR, HER 2 and a combination of two or more of the same (such as ER & PR, ER & HER2, PR & HER2 or triple negative].
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SG10201704545Q | 2017-06-02 | ||
| SG10201704545Q | 2017-06-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018222134A1 true WO2018222134A1 (en) | 2018-12-06 |
Family
ID=64455499
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/SG2018/050209 Ceased WO2018222134A1 (en) | 2017-06-02 | 2018-04-30 | Cancer therapy |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2018222134A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20210009522A1 (en) * | 2017-11-09 | 2021-01-14 | The Henry M. Jackson Foundation For The Advancement Of Military Medicine, Inc. | Compounds for inhibiting ly6k and methods of using same |
| US11311548B2 (en) | 2017-03-02 | 2022-04-26 | Aslan Pharmaceuticals Pte. Ltd. | Cancer therapy |
| US11382903B2 (en) | 2017-03-02 | 2022-07-12 | Aslan Pharmaceuticals Pte. Ltd. | Cancer therapy |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005016346A1 (en) | 2003-08-14 | 2005-02-24 | Array Biopharma Inc. | Quinazoline analogs as receptor tyrosine kinase inhibitors |
| WO2008058229A1 (en) | 2006-11-08 | 2008-05-15 | Bristol-Myers Squibb Company | Pyridinone compounds |
| WO2008077639A1 (en) | 2006-12-22 | 2008-07-03 | Laboratorios Almirall, S.A. | Amino nicotinic and isonicotinic acid derivatives as dhodh inhibitors |
| WO2010038086A2 (en) | 2008-10-02 | 2010-04-08 | Respivert Limited | Novel compounds |
| WO2018136010A1 (en) * | 2017-01-20 | 2018-07-26 | Aslan Pharmaceuticals Pte Ltd | Combination therapy |
-
2018
- 2018-04-30 WO PCT/SG2018/050209 patent/WO2018222134A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005016346A1 (en) | 2003-08-14 | 2005-02-24 | Array Biopharma Inc. | Quinazoline analogs as receptor tyrosine kinase inhibitors |
| WO2008058229A1 (en) | 2006-11-08 | 2008-05-15 | Bristol-Myers Squibb Company | Pyridinone compounds |
| WO2008077639A1 (en) | 2006-12-22 | 2008-07-03 | Laboratorios Almirall, S.A. | Amino nicotinic and isonicotinic acid derivatives as dhodh inhibitors |
| WO2010038086A2 (en) | 2008-10-02 | 2010-04-08 | Respivert Limited | Novel compounds |
| WO2018136010A1 (en) * | 2017-01-20 | 2018-07-26 | Aslan Pharmaceuticals Pte Ltd | Combination therapy |
Non-Patent Citations (6)
| Title |
|---|
| A.K. MOHAMAD FAIRUS ET AL: "Dihydroorotate dehydrogenase (DHODH) inhibitors affect ATP depletion, endogenous ROS and mediate S-phase arrest in breast cancer cells", BIOCHIMIE, vol. 135, 1 April 2017 (2017-04-01), FR, pages 154 - 163, XP055500522, ISSN: 0300-9084, DOI: 10.1016/j.biochi.2017.02.003 * |
| BREEDVELD F.C., ANN RHEUM DIS, 2000 |
| JIANG, M. ET AL.: "Abstract P2-09-21: Teriflunamide, an immunomodulatory drug, exerts anticancer activity in triple-negative breast cancer (TNBC) cells via modulation of multiple cell signal pathways", CANCER RESEARCH, vol. 73, no. 24, 1 December 2013 (2013-12-01), XP002783960, Retrieved from the Internet <URL:http://cancerres.aacrjournals.org/content/73/24_Supplement/P2-09-21> [retrieved on 20180817] * |
| KRISTIN K. BROWN ET AL: "Adaptive Reprogramming of De Novo Pyrimidine Synthesis Is a Metabolic Vulnerability in Triple-Negative Breast Cancer", CANCER DISCOVERY, vol. 7, no. 4, 1 April 2017 (2017-04-01), US, pages 391 - 399, XP055500540, ISSN: 2159-8274, DOI: 10.1158/2159-8290.CD-16-0611 * |
| LOFFLER, MOL CELL BIOCHEM, 1997 |
| MCROBERT ET AL., MOL BIOCHEM PARASITOL, 2002 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11311548B2 (en) | 2017-03-02 | 2022-04-26 | Aslan Pharmaceuticals Pte. Ltd. | Cancer therapy |
| US11382903B2 (en) | 2017-03-02 | 2022-07-12 | Aslan Pharmaceuticals Pte. Ltd. | Cancer therapy |
| US20210009522A1 (en) * | 2017-11-09 | 2021-01-14 | The Henry M. Jackson Foundation For The Advancement Of Military Medicine, Inc. | Compounds for inhibiting ly6k and methods of using same |
| AU2018364990B2 (en) * | 2017-11-09 | 2023-08-24 | The Henry M. Jackson Foundation For The Advancement Of Military Medicine, Inc. | Compounds for inhibiting Ly6K and methods of using same |
| US12134601B2 (en) * | 2017-11-09 | 2024-11-05 | The Henry M. Jackson Foundation For The Advancement Of Military Medicine, Inc. | Compounds for inhibiting LY6K and methods of using same |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2757373C2 (en) | Combination therapy with antitumor alkaloid | |
| US20220088029A1 (en) | Combination therapy of tetracyclic quinolone analogs for treating cancer | |
| US20180353510A1 (en) | Treatment of Biliary Duct Cancer | |
| US11311548B2 (en) | Cancer therapy | |
| US11382903B2 (en) | Cancer therapy | |
| CN107427522A (en) | Alpimod for the treatment of melanoma | |
| US11622965B2 (en) | Methods for treating lymphoid malignancies | |
| US20190134034A1 (en) | Method of Treating Liver Cancer | |
| CN108524511B (en) | Cancer therapy | |
| WO2018222134A1 (en) | Cancer therapy | |
| CA2985379C (en) | Micronized pharmaceutical compositions for treatment of angiogenisis conditions | |
| WO2018222135A1 (en) | Cancer therapy | |
| US12377089B2 (en) | Quinoline derivative used for soft tissue sarcoma combination therapy | |
| US20230038138A1 (en) | Combination therapy for treating cancer | |
| HK1261004A1 (en) | Cancer therapy | |
| HK40065487A (en) | Quinoline derivative used for soft tissue sarcoma combination therapy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18730883 Country of ref document: EP Kind code of ref document: A1 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 18730883 Country of ref document: EP Kind code of ref document: A1 |