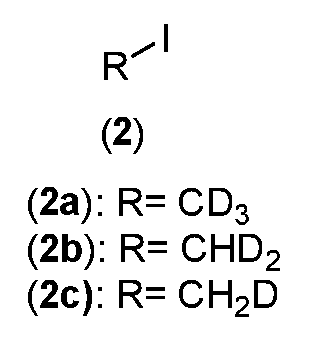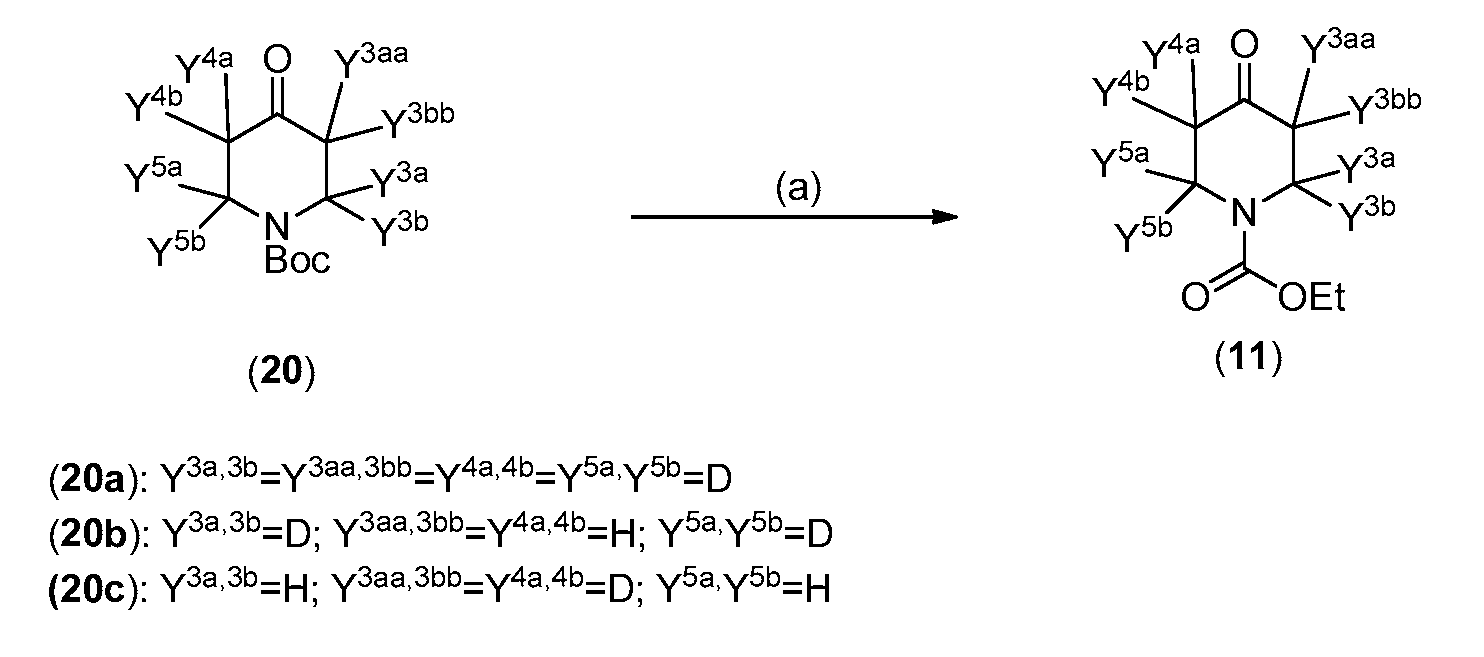WO2018106916A1 - Deuterated quinoxaline compounds - Google Patents
Deuterated quinoxaline compounds Download PDFInfo
- Publication number
- WO2018106916A1 WO2018106916A1 PCT/US2017/065111 US2017065111W WO2018106916A1 WO 2018106916 A1 WO2018106916 A1 WO 2018106916A1 US 2017065111 W US2017065111 W US 2017065111W WO 2018106916 A1 WO2018106916 A1 WO 2018106916A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- hydrogen
- same
- deuterium
- composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C[C@]1(C(*)(*)N(C2(C(*)(*)C(*)(C(*)(*)c(c(*)c3*)c(*)c(*)c3F)*=C)*=*2)C(*)(*)C(*)(*)[C@@]11C)c(c(*)c(*)c(*)c2N(*)C3(C)*)c2N1C3(*)*=C Chemical compound C[C@]1(C(*)(*)N(C2(C(*)(*)C(*)(C(*)(*)c(c(*)c3*)c(*)c(*)c3F)*=C)*=*2)C(*)(*)C(*)(*)[C@@]11C)c(c(*)c(*)c(*)c2N(*)C3(C)*)c2N1C3(*)*=C 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains three hetero rings
- C07D471/14—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B59/00—Introduction of isotopes of elements into organic compounds ; Labelled organic compounds per se
- C07B59/002—Heterocyclic compounds
Definitions
- ADME absorption, distribution, metabolism and/or excretion
- ADME limitation that affects many medicines is the formation of toxic or biologically reactive metabolites.
- some patients receiving the drug may experience toxicities, or the safe dosing of such drugs may be limited such that patients receive a suboptimal amount of the active agent.
- modifying dosing intervals or formulation approaches can help to reduce clinical adverse effects, but often the formation of such undesirable metabolites is intrinsic to the metabolism of the compound.
- a metabolic inhibitor will be co-administered with a drug that is cleared too rapidly.
- a drug that is cleared too rapidly.
- the FDA recommends that these drugs be co- dosed with ritonavir, an inhibitor of cytochrome P450 enzyme 3A4 (CYP3A4), the enzyme typically responsible for their metabolism (see Kempf, D.J. et al., Antimicrobial agents and chemotherapy, 1997, 41(3): 654-60).
- CYP3A4 cytochrome P450 enzyme 3A4
- Ritonavir causes adverse effects and adds to the pill burden for HIV patients who must already take a combination of different drugs.
- the CYP2D6 inhibitor quinidine has been added to dextromethorphan for the purpose of reducing rapid CYP2D6 metabolism of dextromethorphan in a treatment of pseudobulbar affect.
- Quinidine has unwanted side effects that greatly limit its use in potential combination therapy (see Wang, L et al., Clinical Pharmacology and Therapeutics, 1994, 56(6 Pt 1): 659-67; and FDA label for quinidine at www.accessdata.fda.gov).
- a potentially attractive strategy for improving a drug's metabolic properties is deuterium modification.
- Deuterium is a safe, stable, non-radioactive isotope of hydrogen. Compared to hydrogen, deuterium forms stronger bonds with carbon. In select cases, the increased bond strength imparted by deuterium can positively impact the ADME properties of a drug, creating the potential for improved drug efficacy, safety, and/or tolerability.
- the size and shape of deuterium are essentially identical to those of hydrogen, replacement of hydrogen by deuterium would not be expected to affect the biochemical potency and selectivity of the drug as compared to the original chemical entity that contains only hydrogen.
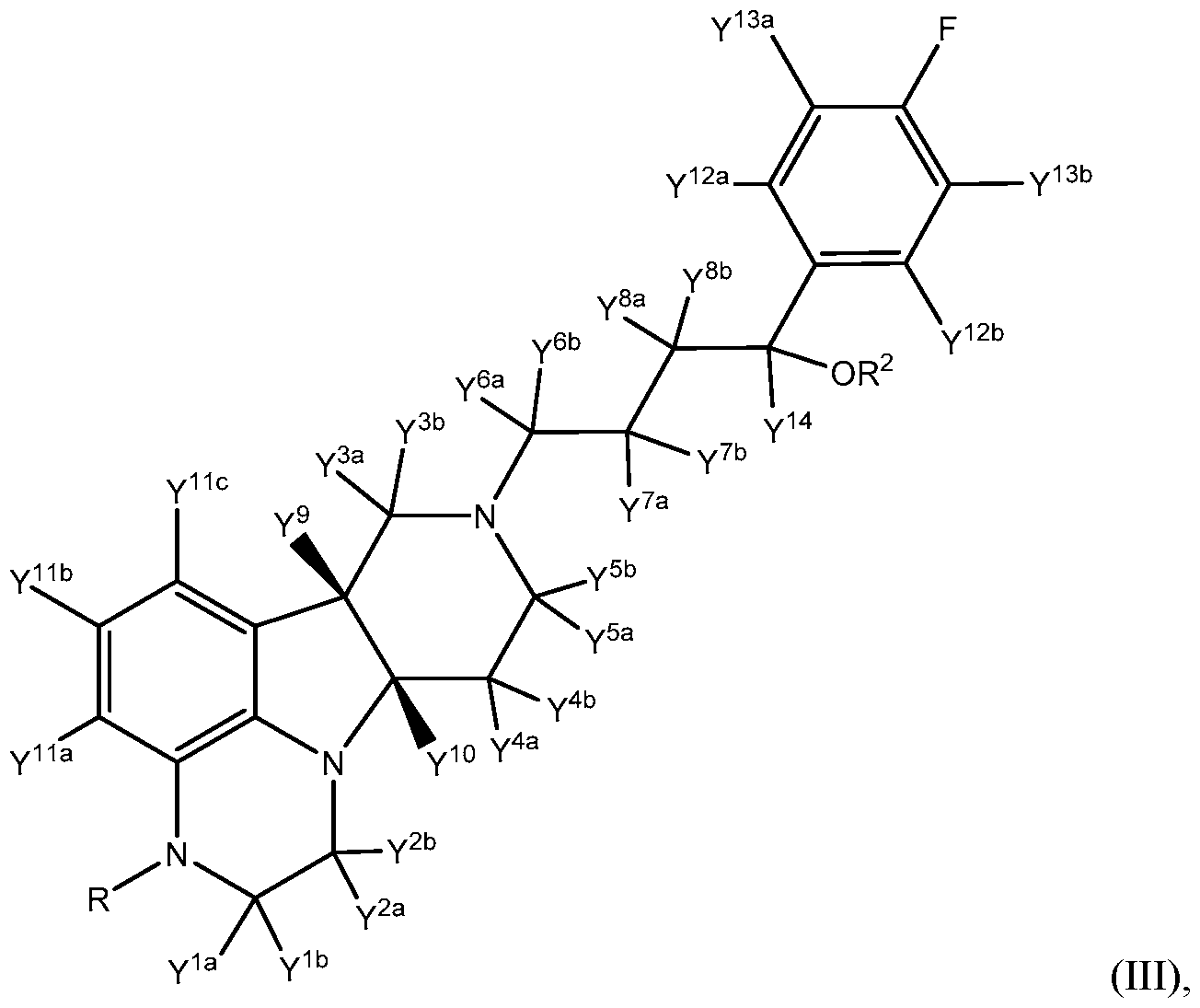
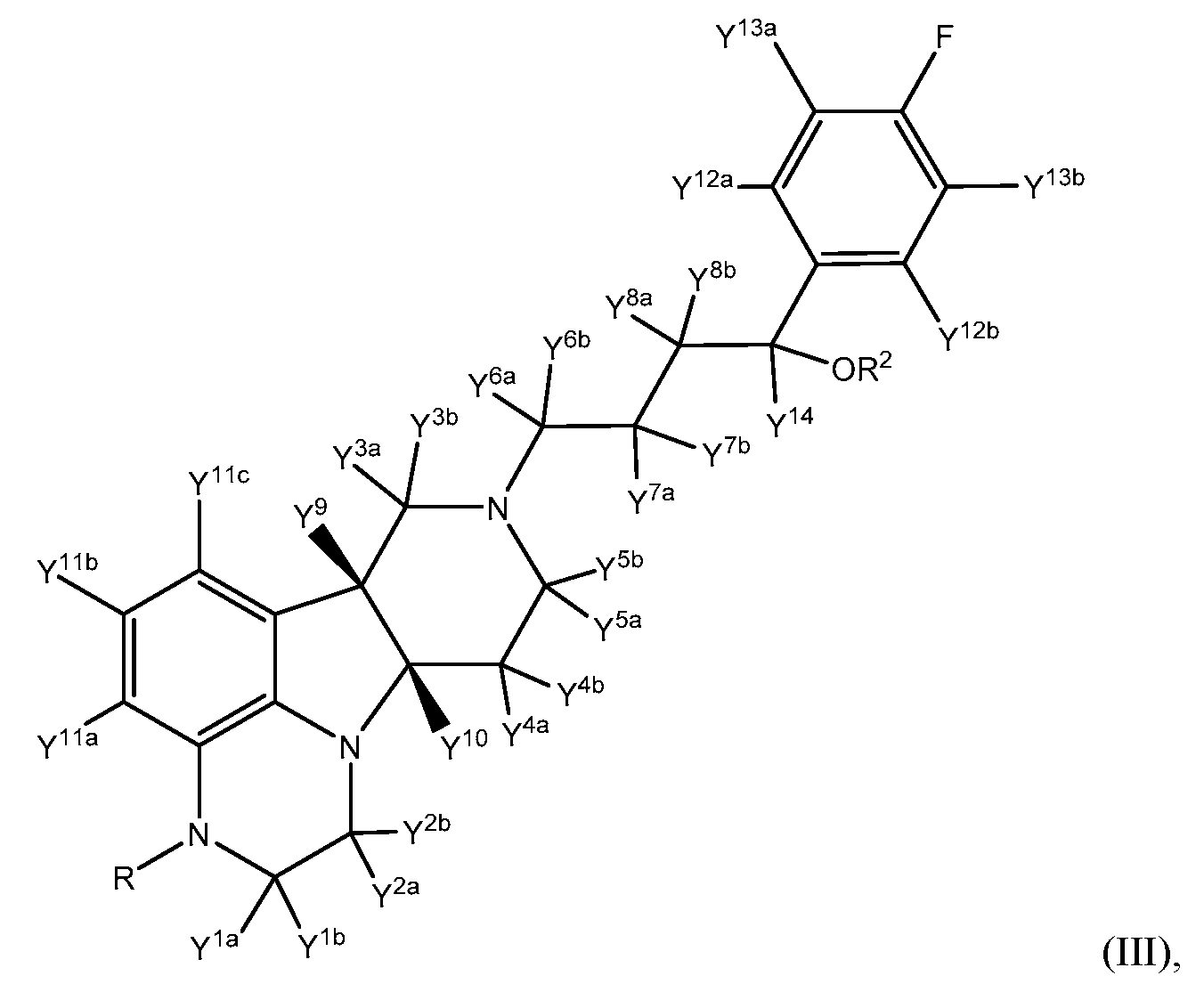
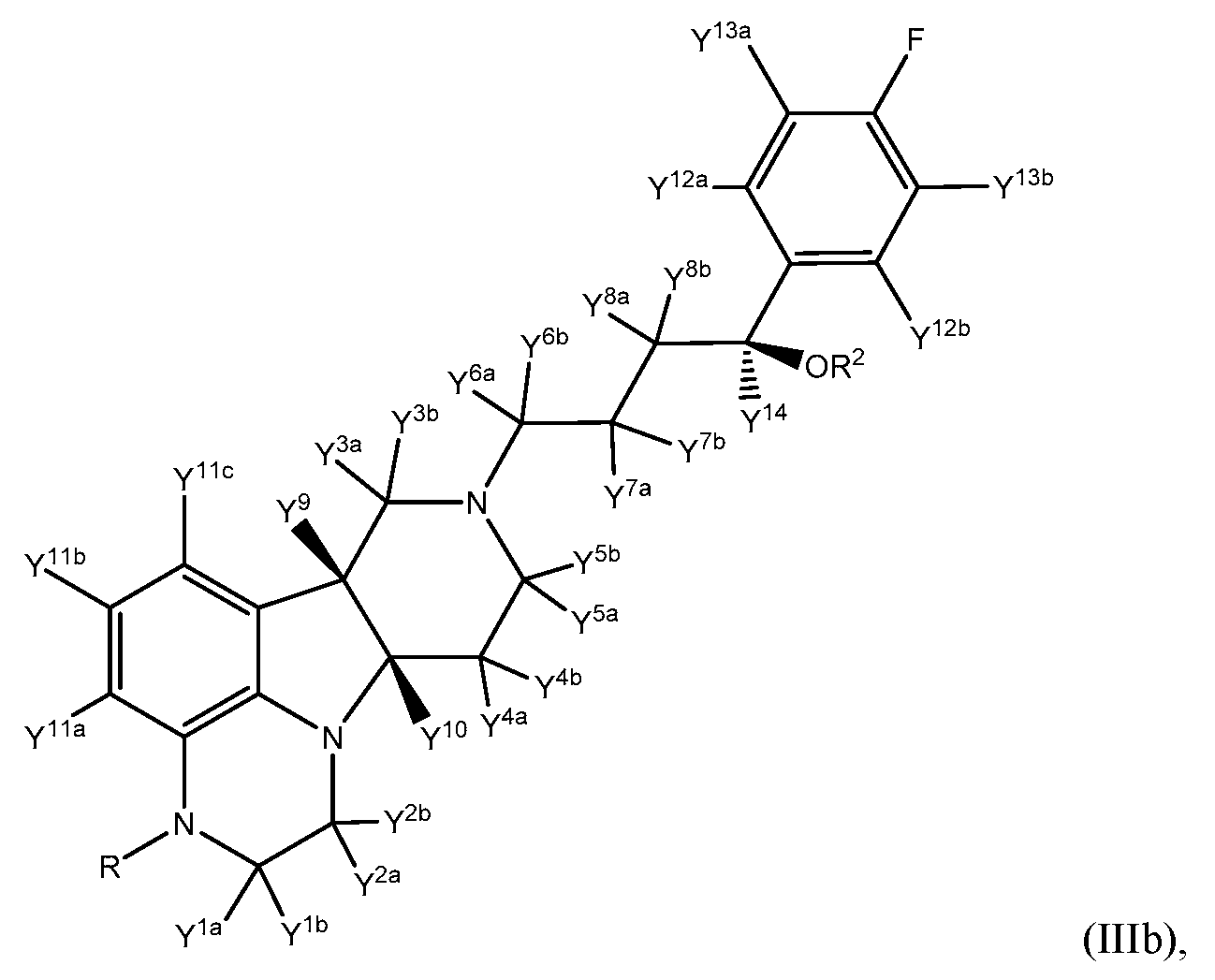
- This invention relates to novel 2,3,6b,7,8,9,10,10a-octahydro-lH- pyrido[3',4':4,5]pyrrolo[l,2,3-de]quinoxalines, and pharmaceutically acceptable salts thereof. Certain aspects of the present invention are directed to compounds of Formula III:
- R is CH3, CH 2 D, CHD 2 or CD3;
- R 2 is hydrogen, deuterium, or C(0)-C 1-21 alkyl;
- Y 8a , Y ®> , and Y 14 are each deuterium;
- Y 12a and Y 12b are each hydrogen;
- Y 13a and Y 13b are each hydrogen; and
- Y 1a , Y 1b , Y 2a , Y 2b , Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , Y 5b , Y 6a , Y 6b , Y 7a , Y 711 , Y 9 , and Y 10 are independently selected from hydrogen and deuterium.
- compositions comprising a compound of this invention, including pharmaceutical compositions comprising a compound of this invention and a pharmaceutically acceptable carrier. Certain aspects of the present invention also provide the use of such compounds and compositions in methods of treating diseases and conditions that are beneficially treated by administering a serotonergic, dopaminergic, and glutamatergic modulator. Some exemplary embodiments include a method of treating a disease or condition selected from schizophrenia, depression, bipolar disorder, agitation, insomnia, dementia, and Alzheimer's Disease, the method comprising the step of administering to a subject in need thereof a therapeutically effective amount of a compound or pharmaceutical composition of the present invention.
- ITI-007 (CAS Registry numbers 1187020-80-9, 313368-91-1) also known as rn-722 and l-(4-fluorophenyl)-4-[(6bR,10aS)-3-methyl-2,3,6b,7,8,9,10,10a- octahydro-lH-pyrido[3',4':4,5]pyrrolo[l,2,3-de]quinoxalin-8-yl]butan-l-one tosylate, modulates the serotonergic, dopaminergic, and glutamatergic systems.
- ITI-007 is currently in phase III clinical trials for the treatment of
- phase II clinical trials for the treatment of insomnia phase Ib/II clinical trials for the treatment of dementia, including Alzheimer's disease; and is currently being tested for safety and efficacy in patients with an acute exacerbation of the aforementioned diseases.
- ITI-007 is metabolized into two active metabolites, IC200131 and IC200161.
- IC200131 (CAS Registry number 1469343-42-7) is also known as (6bR,10aS)- -(4-fluorophenyl)- 2,3,6b,9,10,10a-hexahydro-3-methyl-lH-Pyrido[3',4':4,5]pyrrolo[l,2,3- de]quinoxaline-8(7H)-butanol.
- IC200131 is reported to be a serotonin-2A (5-HT2A) receptor antagonist and inhibitor of serotonin reuptake with roughly equal potency at each of these two targets.
- IC200131 is reported to have a longer plasma half-life than IT1-007 or IC200161.
- IC200131 is reported to be able to undergo back- conversion into IT1-007. The metabolism of ⁇ -007 is also discussed in
- treat means decrease, suppress, attenuate, diminish, arrest, or stabilize the development or progression of a disease (e.g., a disease or disorder delineated herein), lessen the severity of the disease or improve the symptoms associated with the disease.
- a disease e.g., a disease or disorder delineated herein
- Disease means any condition or disorder that damages or interferes with the normal function of a cell, tissue, or organ.
- the term "subject” includes humans and non-human mammals.
- Non-limiting examples of non-human mammals include mice, rats, guinea pigs, rabbits, dogs, cats, monkeys, apes, pigs, cows, sheep, horses, etc.
- any atom not specifically designated as a particular isotope is meant to represent any stable isotope of that atom.
- the position is understood to have hydrogen at its natural abundance isotopic composition.
- the position has at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% hydrogen.
- a position when a position is designated specifically as ⁇ " or “hydrogen”, the position incorporates ⁇ 20% deuterium, ⁇ 10% deuterium, ⁇ 5% deuterium, ⁇ 4% deuterium, ⁇ 3% deuterium, ⁇ 2% deuterium, or ⁇ 1% deuterium. Also unless otherwise stated, when a position is designated specifically as "D" or
- deuterium the position is understood to have deuterium at an abundance that is at least 3340 times greater than the natural abundance of deuterium, which is 0.015% (i.e., at least 50.1% incorporation of deuterium).
- isotopic enrichment factor means the ratio between the isotopic abundance and the natural abundance of a specified isotope.
- a compound of this invention has an isotopic enrichment factor for each designated deuterium atom of at least 3500 (52.5% deuterium incorporation at each designated deuterium atom), at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75% deuterium), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5% deuterium incorporation).
- each designated deuterium atom has deuterium incorporation of at least 52.5%.
- each designated deuterium atom has deuterium incorporation of at least 60%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 67.5%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 75%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 82.5%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 90%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 95%.
- each designated deuterium atom has deuterium incorporation of at least 97.5%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 99%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 99.5%.
- isotopologue refers to a species in which the chemical structure differs from a specific compound of this invention only in the isotopic composition thereof.
- a compound represented by a particular chemical structure containing indicated deuterium atoms will also contain lesser amounts of isotopologues having hydrogen atoms at one or more of the designated deuterium positions in that structure.
- the relative amount of such isotopologues in a compound of this invention will depend upon a number of factors including the isotopic purity of deuterated reagents used to make the compound and the efficiency of incorporation of deuterium in the various synthesis steps used to prepare the compound.
- the invention also provides salts of the compounds of the invention.
- a salt of a compound of this invention is formed between an acid and a basic group of the compound, such as an amino functional group, or a base and an acidic group of the compound, such as a carboxyl functional group.
- the compound is a pharmaceutically acceptable acid addition salt.
- pharmaceutically acceptable refers to a component that is, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and other mammals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit risk ratio.
- pharmaceutically acceptable salt means any non-toxic salt that, upon administration to a recipient, is capable of providing, either directly or indirectly, a compound of this invention.
- pharmaceutically acceptable counterion is an ionic portion of a salt that is not toxic when released from the salt upon administration to a recipient.
- Acids commonly employed to form pharmaceutically acceptable salts include inorganic acids such as hydrogen bisulfide, hydrochloric acid, hydrobromic acid, hydroiodic acid, sulfuric acid and phosphoric acid, as well as organic acids such as para-toluenesulfonic acid, salicylic acid, tartaric acid, bitartaric acid, ascorbic acid, maleic acid, besylic acid, fumaric acid, gluconic acid, glucuronic acid, formic acid, glutamic acid, methanesulfonic acid, ethanesulfonic acid, benzenesulfonic acid, lactic acid, oxalic acid, para-bromophenylsulfonic acid, carbonic acid, succinic acid, citric acid, benzoic acid and acetic acid, as well as related inorganic and organic acids.
- inorganic acids such as hydrogen bisulfide, hydrochloric acid, hydrobromic acid, hydroiodic acid, sulfuric acid and phosphoric acid
- Such pharmaceutically acceptable salts thus include sulfate, pyrosulfate, bisulfate, sulfite, bisulfite, phosphate, monohydrogenphosphate, dihydrogenphosphate, metaphosphate, pyrophosphate, chloride, bromide, iodide, acetate, propionate, decanoate, caprylate, acrylate, formate, isobutyrate, caprate, heptanoate, propiolate, oxalate, malonate, succinate, suberate, sebacate, fumarate, maleate, butyne-l,4-dioate, hexyne-l,6-dioate, benzoate, chlorobenzoate, methylbenzoate, dinitrobenzoate, hydroxybenzoate, methoxybenzoate, phthalate, terephthalate, sulfonate, xylene sulfonate, phenylacetate, phenylprop
- pharmaceutically acceptable acid addition salts include those formed with mineral acids such as hydrochloric acid and hydrobromic acid, and especially those formed with organic acids such as maleic acid.
- the acids commonly employed to form pharmaceutically acceptable salts include the above- listed inorganic acids, wherein at least one hydrogen is replaced with deuterium.
- the compounds of the present invention may contain an asymmetric carbon atom, for example, as the result of deuterium substitution or otherwise.
- compounds of this invention can exist as either individual enantiomers, or mixtures of the two enantiomers.
- a compound of the present invention may exist as either a racemic mixture or a scalemic mixture, or as individual respective stereoisomers that are substantially free from another possible stereoisomer.
- substantially free of other stereoisomers as used herein means less than 25% of other stereoisomers, preferably less than 10% of other stereoisomers, more preferably less than 5% of other stereoisomers and most preferably less than 2% of other stereoisomers are present.
- stable compounds refers to compounds which possess stability sufficient to allow for their manufacture and which maintain the integrity of the compound for a sufficient period of time to be useful for the purposes detailed herein (e.g., formulation into therapeutic products, intermediates for use in production of therapeutic compounds, isolatable or storable intermediate compounds, treating a disease or condition responsive to therapeutic agents).
- Substituted with deuterium refers to the replacement of one or more hydrogen atoms with a corresponding number of deuterium atoms.
- variable may be referred to generally (e.g.,"each R") or may be referred to specifically (e.g., R 1 , R 2 , R 3 , etc.). Unless otherwise indicated, when a variable is referred to generally, it is meant to include all specific embodiments of that particular variable.
- the present invention in one embodiment provides a compound of Formula III:
- R is CH 3 , CH 2 D, CHD 2 or CD 3 ;
- R 2 is hydrogen, deuterium, or C(0)-d- 21 alkyl
- Y 8a , Y 8b , and Y 14 are each deuterium
- Y 1a Y 1b Y 2a Y 2b Y 3a Y 3b Y 4a Y 4b Y 5a Y 5 ' 5 Y 6a Y 6b Y 7a Y 7 * 5 Y 9 Y 10 Y 11a Y 11b , Y 11c , Y 12a , Y 12b , Y 13a , and Y 13b are independently selected from hydrogen and deuterium.
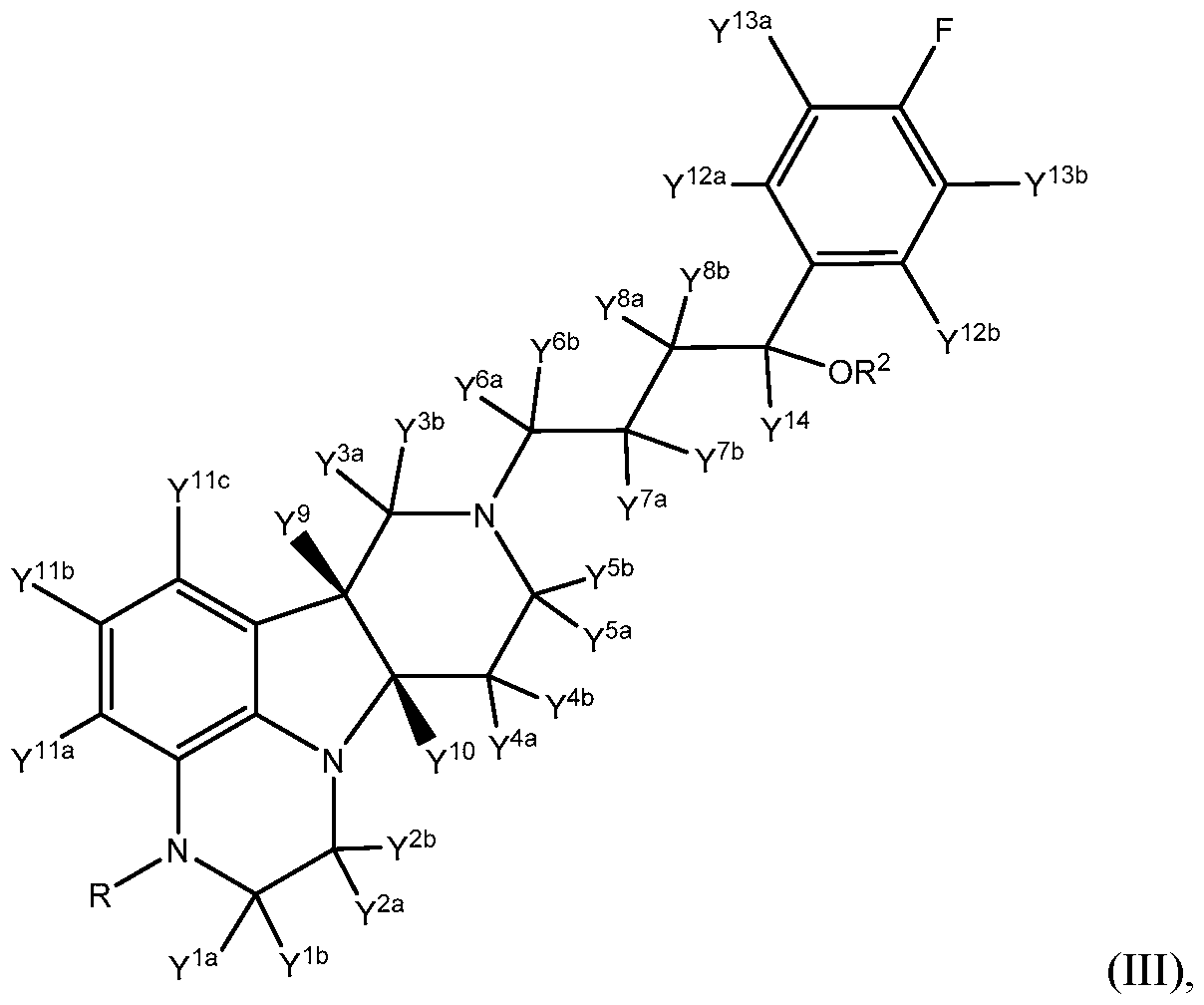
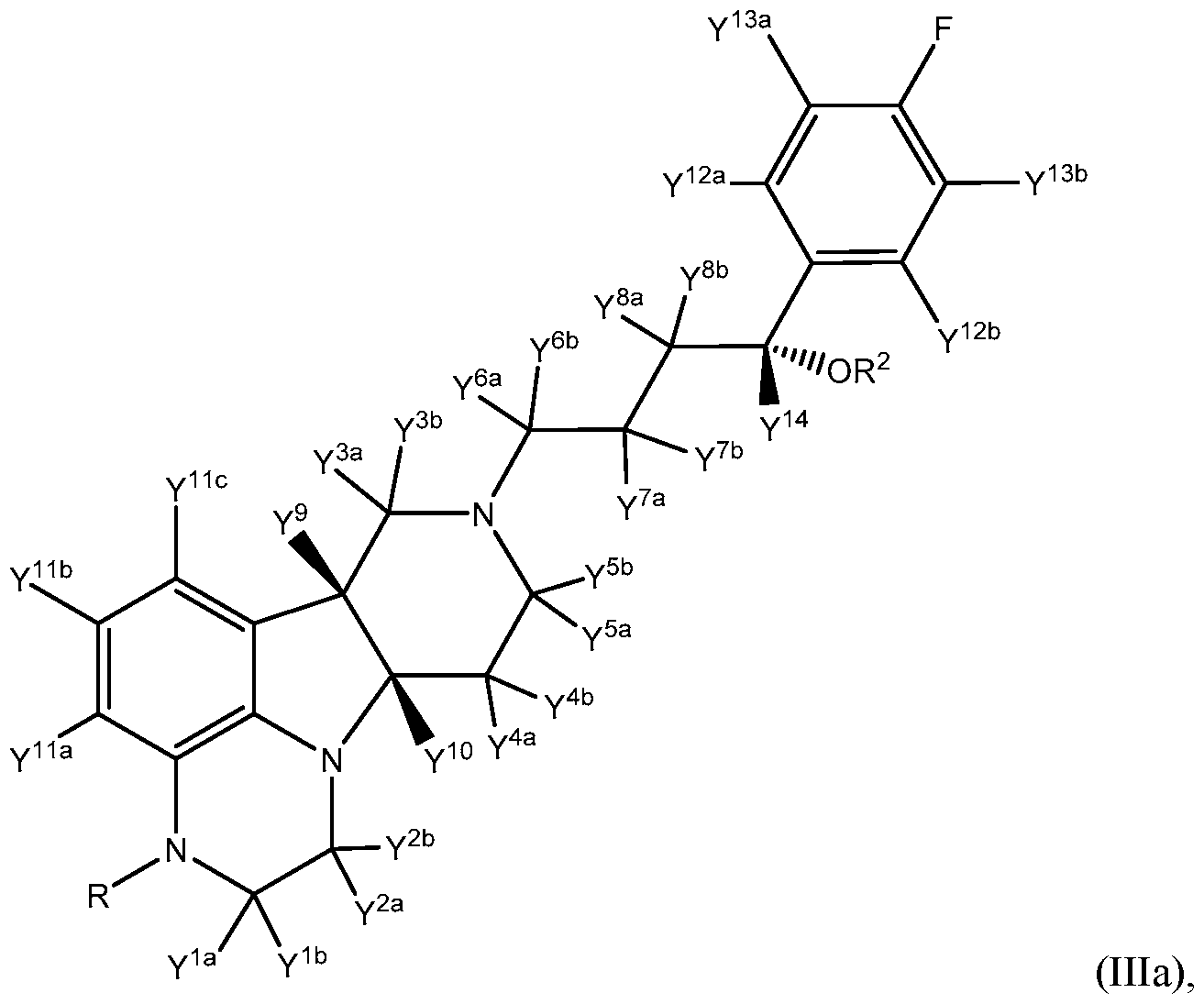
- the compound of formula III is a compound of Formula Ilia:
- R, R 2 , Y 1a , Y 1b , Y 2a , Y 2b , Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , Y a , Y 6a , Y 6b , Y 7a , Y 7b , Y 8a , Y 8b , Y 9 , Y 10 , Y 11a , Y 11b , Y 11c , Y 12a , Y 12b , Y 13a , Y 13b , and Y 14 are as defined herein.
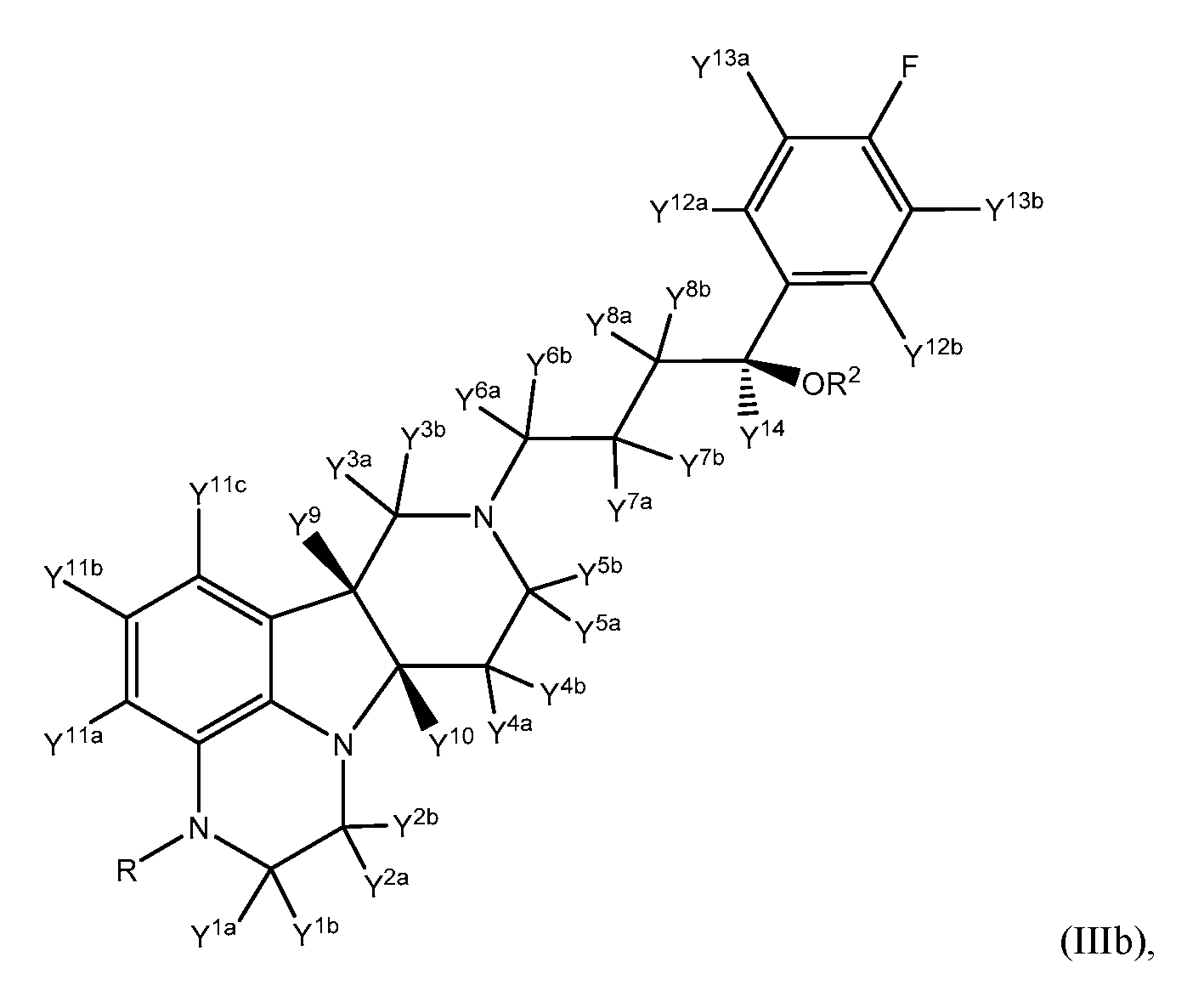
- the compound of formula III is a compound of Formula Illb:
- R, R 2 , Y 1a , Y 1b , Y 2a , Y 2b , Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , Y 5b , Y 6a , Y 6b , Y 7a , Y 7b , Y 8a , Y 8b , Y 9 , Y 10 , Y 11a , Y 11b , Y 11c , Y 12a , Y 12b , Y 13a , Y 13b , and Y 14 are as defined herein.
- R is CH 3 .
- R is CD3. 11 [43] In some embodiments of any one of Formulae III, Ilia and Illb, Y 1a and Y 1b are the same. In some aspects of these embodiments, Y 1a and Y 1b are each hydrogen.
- Y 1a and Y 1b are each deuterium.
- Y 2a and Y 2b are the same. In some aspects of these embodiments, Y 2a and Y 2b are each hydrogen.
- Y 2a and Y 2b are each deuterium.
- Y 1a , Y 1b , Y 2a and Y 2b are the same.
- Y 1a , Y 1b , Y 2a and Y 2b are each hydrogen.
- Y 1a , Y 1b , Y 2a and Y 2b are each deuterium.
- Y 1a and Y 1b are each hydrogen; and Y 2a and Y 2b are each deuterium. In other embodiments, Y 1a and Y 1b are each deuterium; and Y 2a and Y 2b are each hydrogen.
- Ilia and Illb are the same. In some aspects of these embodiments, Y 3a and Y 3b are each hydrogen.
- Y 3a and Y 3b are each deuterium.
- Y 4a and Y 4b are the same. In some aspects of these embodiments, Y 4a and Y 4b are each hydrogen.
- Y 4a and Y 4b are each deuterium.
- Ilia and Illb are the same. In some aspects of these embodiments, Y 5a and Y* are each hydrogen.
- Y 5a and Y 5b are each deuterium.
- Y 4b , Y 5a and Y 5b are the same. In some aspects of these embodiments, Y 3a , Y 3b , Y 4a ,
- Y 4b , Y 5a and Y 5b are each hydrogen. In other aspects of these embodiments, Y 3a , Y 3b ,
- Y 4a , Y 4b , Y 5a and Y 5b are each deuterium.
- Ilia and Illb, Y 3a , Y 3b , Y 5a and ⁇ ⁇ are each hydrogen; and Y 4a and Y 4b are each deuterium.
- Y 3a , Y 3b , Y 5a and Y 5b are each deuterium; and Y 4a and Y 4b are each hydrogen.
- Ilia and Illb, Y 6a and Y 6b are the same. In some aspects of these embodiments, Y 6a and Y* are each hydrogen. In other aspects of these embodiments, Y 6a and Y 6b are each deuterium. [53] In some embodiments of any one of Formulae III, Ilia and Illb, Y 7a and Y 711 are the same. In some aspects of these embodiments, Y 7a and Y 715 are each hydrogen. In other aspects of these embodiments, Y 7a and Y 711 are each deuterium.
- Ilia and Illb, Y 6a , Y*, Y 7a , and Y 711 are the same.
- Y 6a , Y a , Y 7a , and Y 711 are each hydrogen.
- Y 6a , Y 6b , Y 7a , and Y 715 are each deuterium.
- Y 7a and Y 711 are each hydrogen; and Y 6a and Y 6b are each deuterium. In other embodiments, Y 7a and Y 711 are each deuterium; and Y 6a and Y 6b are each hydrogen.
- Y 11a , Y 11b , Y 11 c Y i2a yi2b ; yi3a md yi3b each hydrogen.
- Y 11a , Y 11b , Y 11c , Y 12a , Y 12b , Y 13a , and Y 13b are each deuterium.
- R 2 is hydrogen
- R 2 is deuterium
- R 2 is C(0)-C 1- 21 alkyl. In some embodiments, R 2 is C(0)-Cs- 1 9 alkyl. In some embodiments, R 2 is C(0)-C9-n alkyl. In some embodiments, R 2 is C(0)-C 1 5 alkyl. In some embodiments, R 2 is palmitoyl.
- Y 9 is hydrogen
- Y 9 is deuterium.
- Y 10 is hydrogen
- Y 10 is deuterium.
- Ilia and Illb, Y 9 and Y 10 are each hydrogen.
- Ilia and Illb, Y 9 and Y 10 are each deuterium.
- Ilia and Illb, Y 11a , Y 11b and Y 11c are each hydrogen.
- Ilia and Illb, Y 11a , Y 11b and Y 11c are each deuterium.
- Ilia and Illb, Y 12a and Y 12b are each hydrogen.
- Ilia and Illb, Y 12a and Y 12b are each deuterium.
- Ilia and Illb, Y 13a and Y 13b are each hydrogen.
- Ilia and Illb, Y 13a and Y 13b are each deuterium.
- R is CD 3 ;
- R 2 is hydrogen; Y 1a and Y 1b are the same; Y 2a and Y 2b are the same; Y 3a and Y 3b are the same; Y 4a and Y 4b are the same; Y 5a and Y 5b are the same; Y 6a and Y 6b are the same; Y 7a and Y 711 are the same; Y 9 is hydrogen;
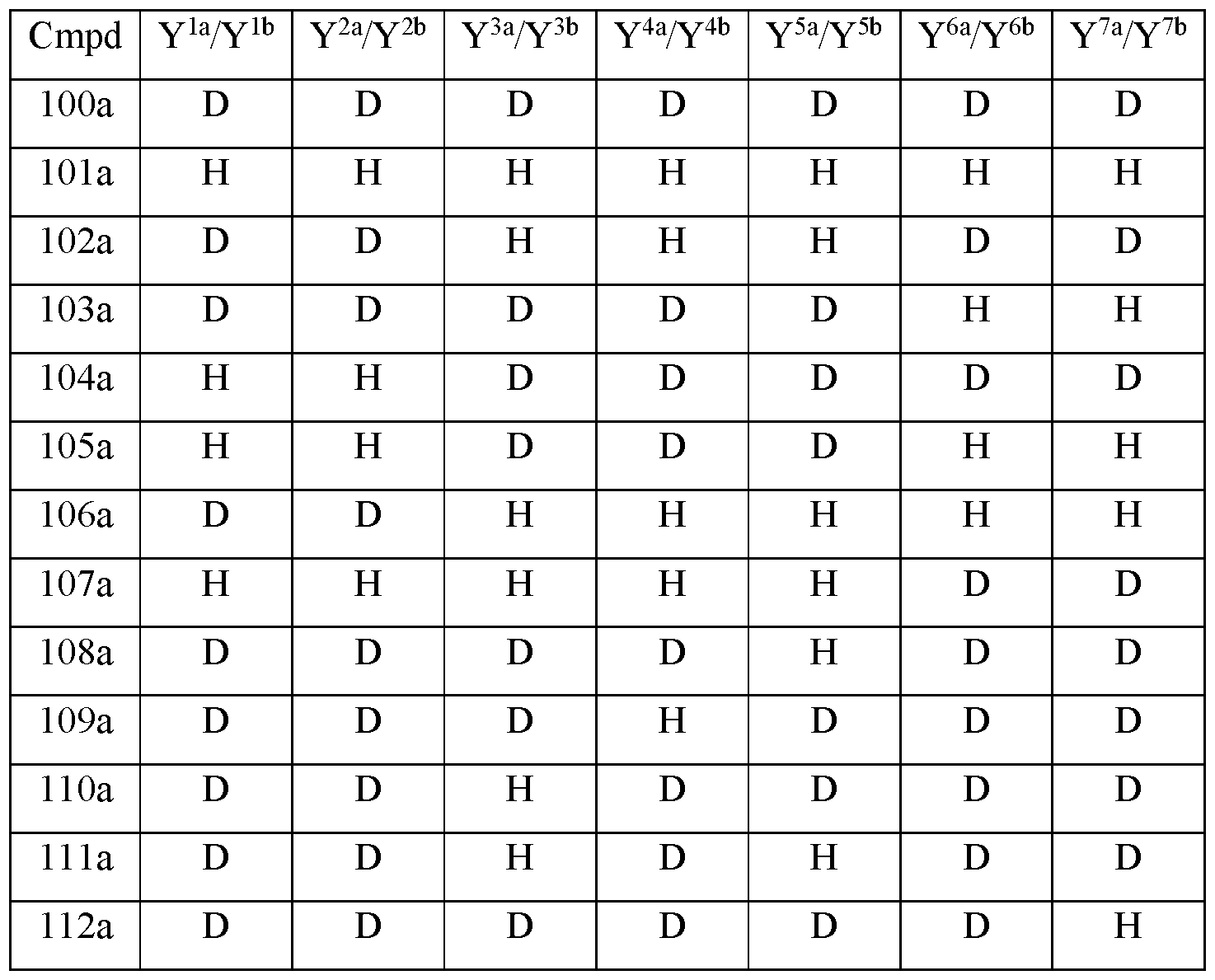
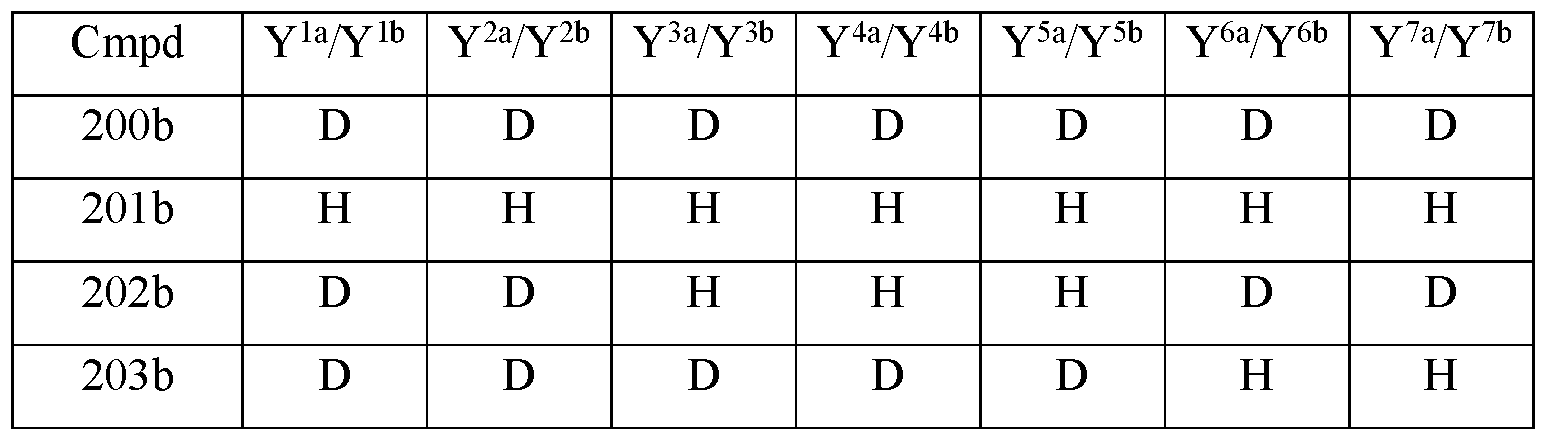
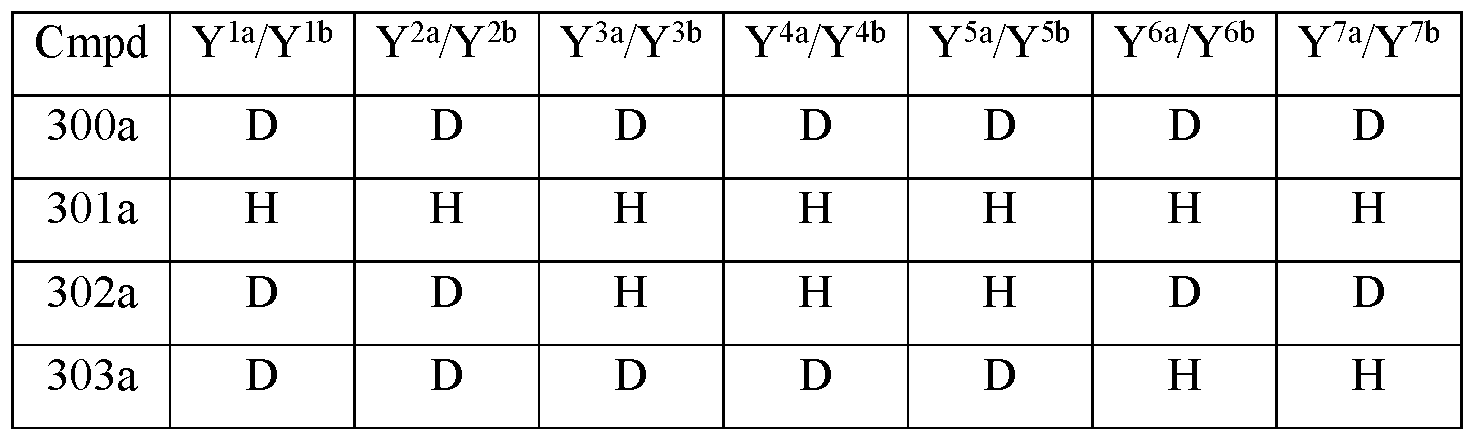
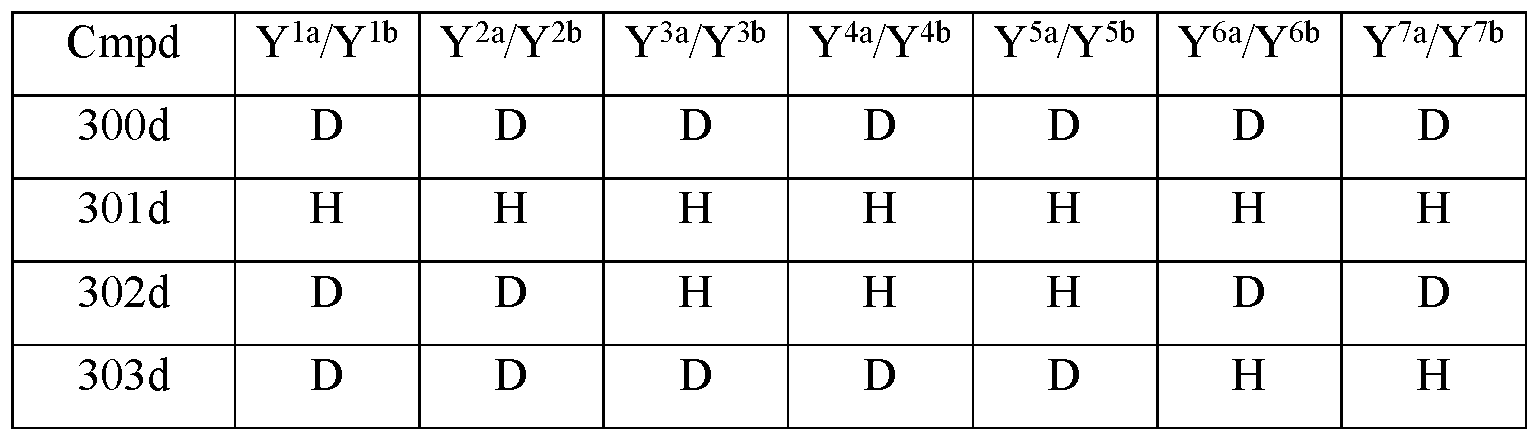
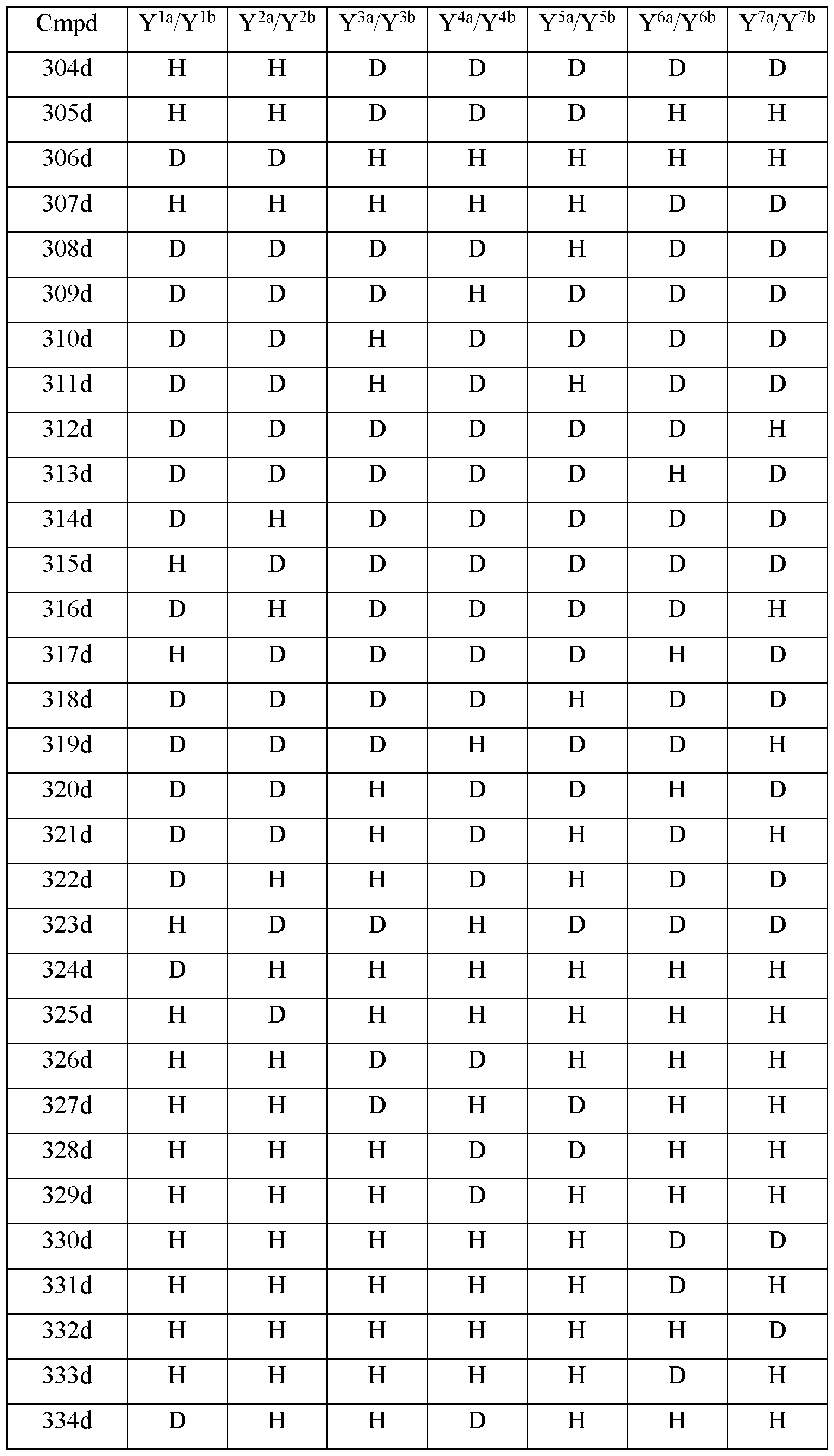
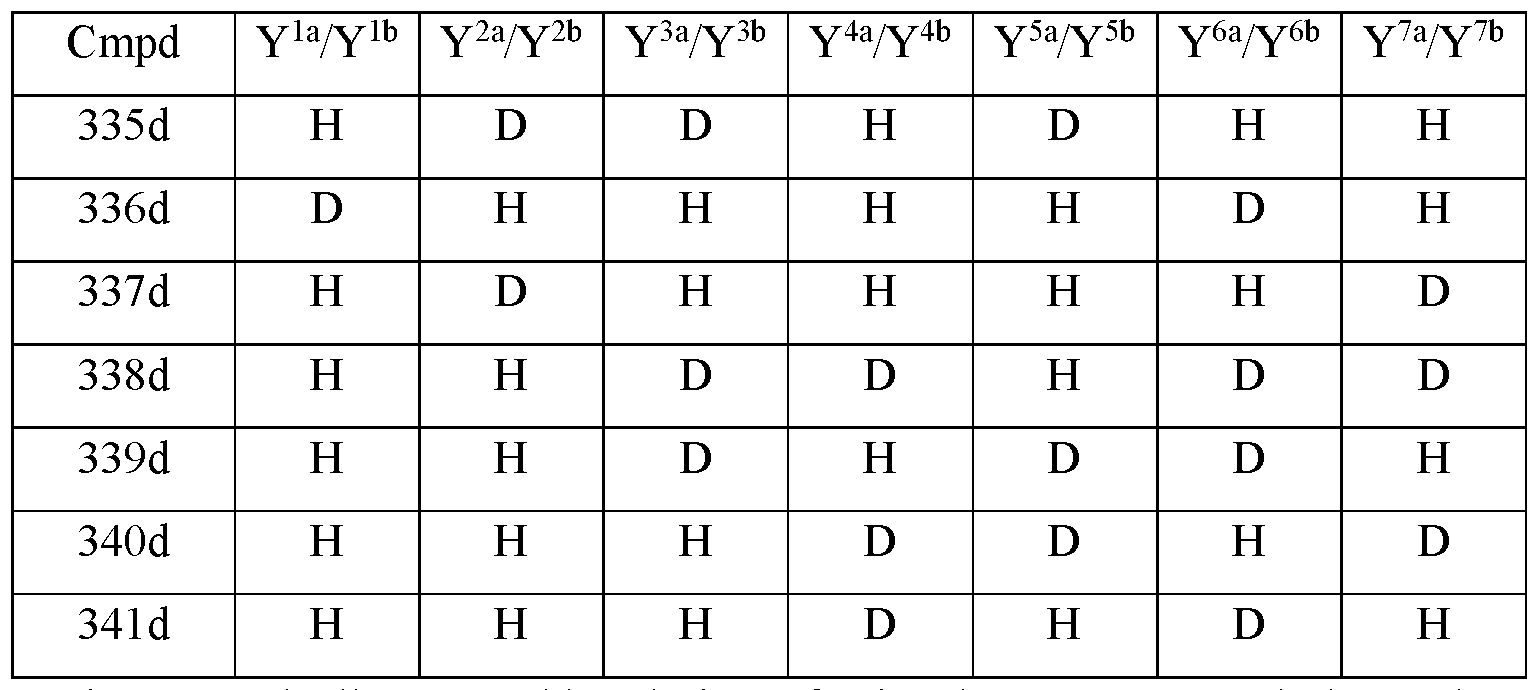
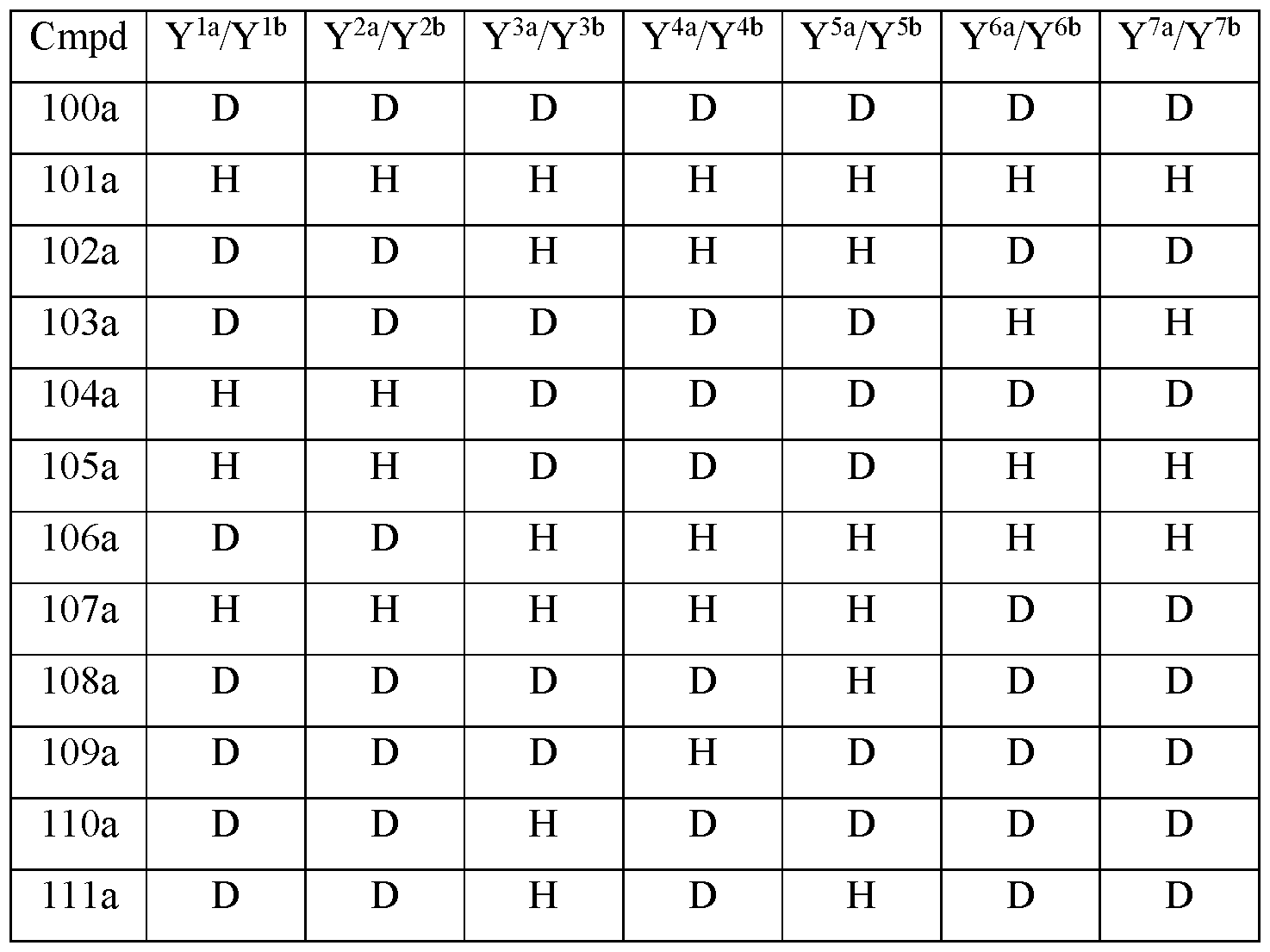
- Y 10 is hydrogen; Y 11a , Y 11b and Y 11c are each hydrogen; Y 12a and Y 12b are each hydrogen; Y 13a and Y 13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table la (below):
- R is CH3;
- R 2 is hydrogen; Y 1a and Y 1b are the same; Y 2a and Y 2b are the same; Y 3a and Y 3b are the same; Y 4a and Y 4b are the same; Y 5a and Y 5b are the same; Y 6a and Y 6b are the same; Y 7a and Y 711 are the same; Y 9 is hydrogen;
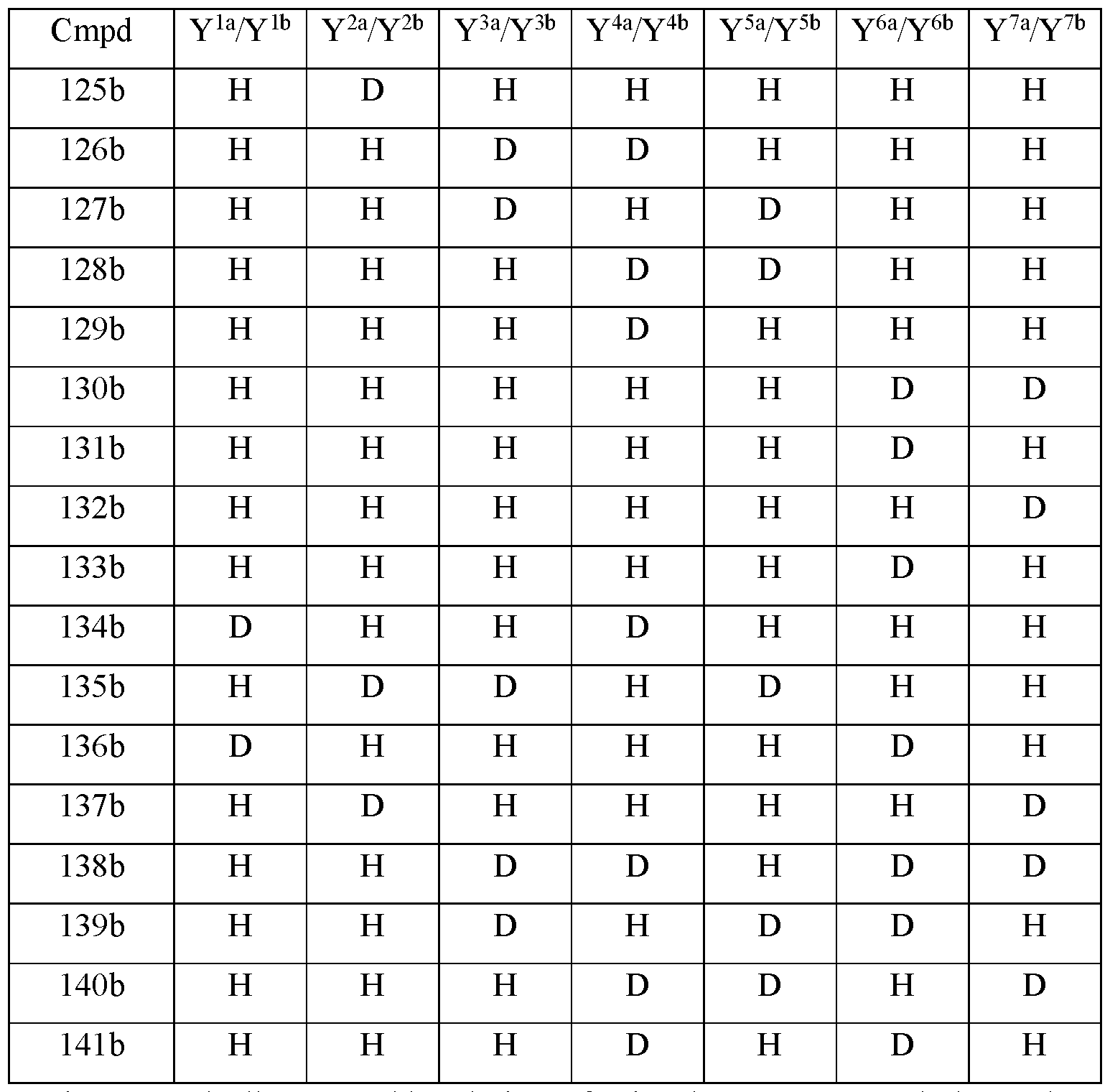
- Y 10 is hydrogen; Y 11a , Y 11b and Y 11c are each hydrogen; Y 12a and Y 12b are each hydrogen; Y 13a and Y 13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table lb (below):
- R is CD 3 ;
- R 2 is palmitoyl;
- Y 1a and Y 1b are the same;
- Y 2a and Y 2b are the same;
- Y 3a and Y 3b are the same;
- Y 4a and Y 4b are the same;
- Y 5a and Y 5b are the same;
- Y 6a and Y 6b are the same;
- Y 7a and Y 711 are the same;
- Y 9 is hydrogen;
- Y 10 is hydrogen;
- Y 11a , Y 11b and Y 11c are each hydrogen;
- Y 12a and Y 12b are each hydrogen;
- Y 13a and Y 13b are each hydrogen; and
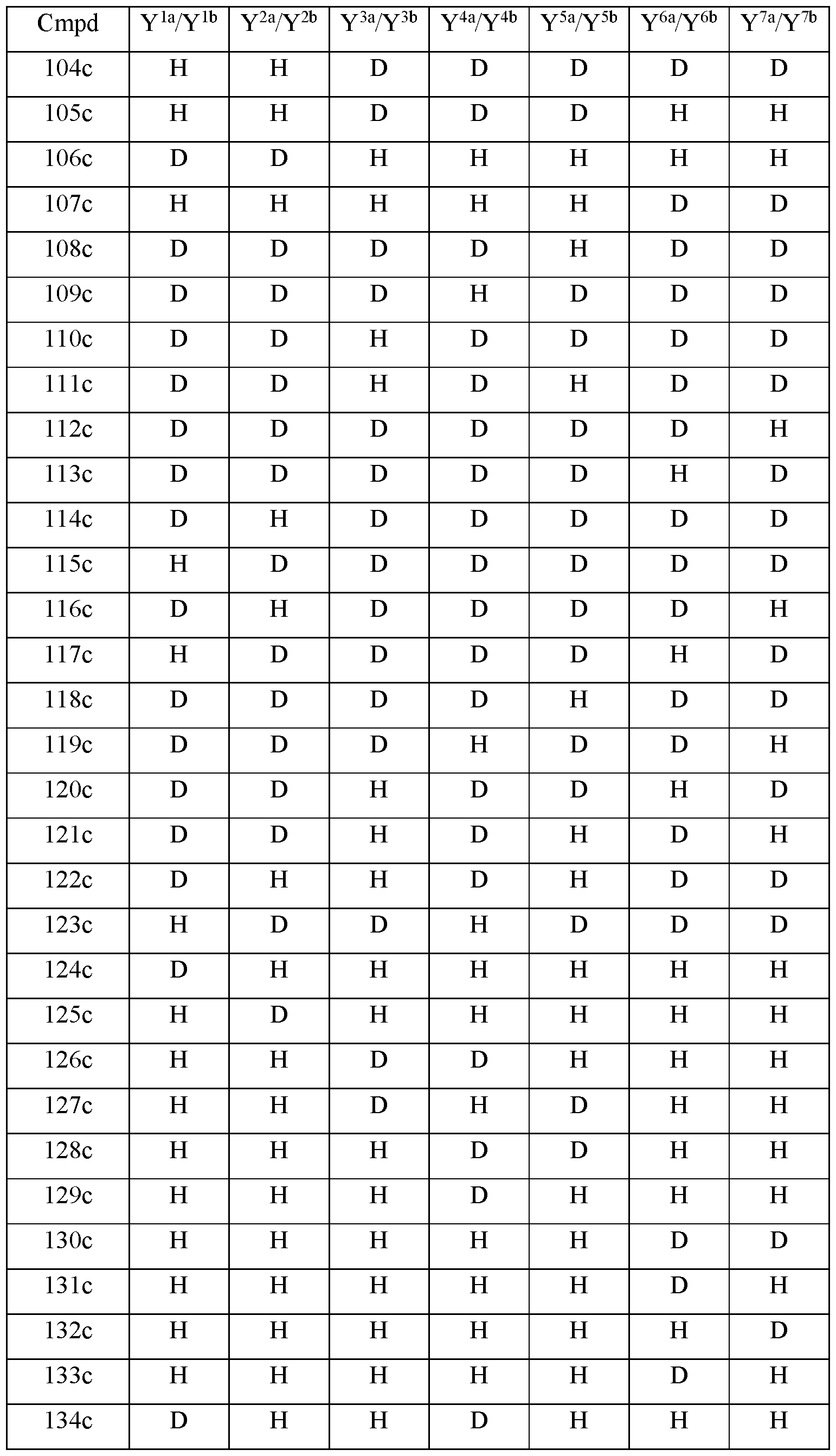
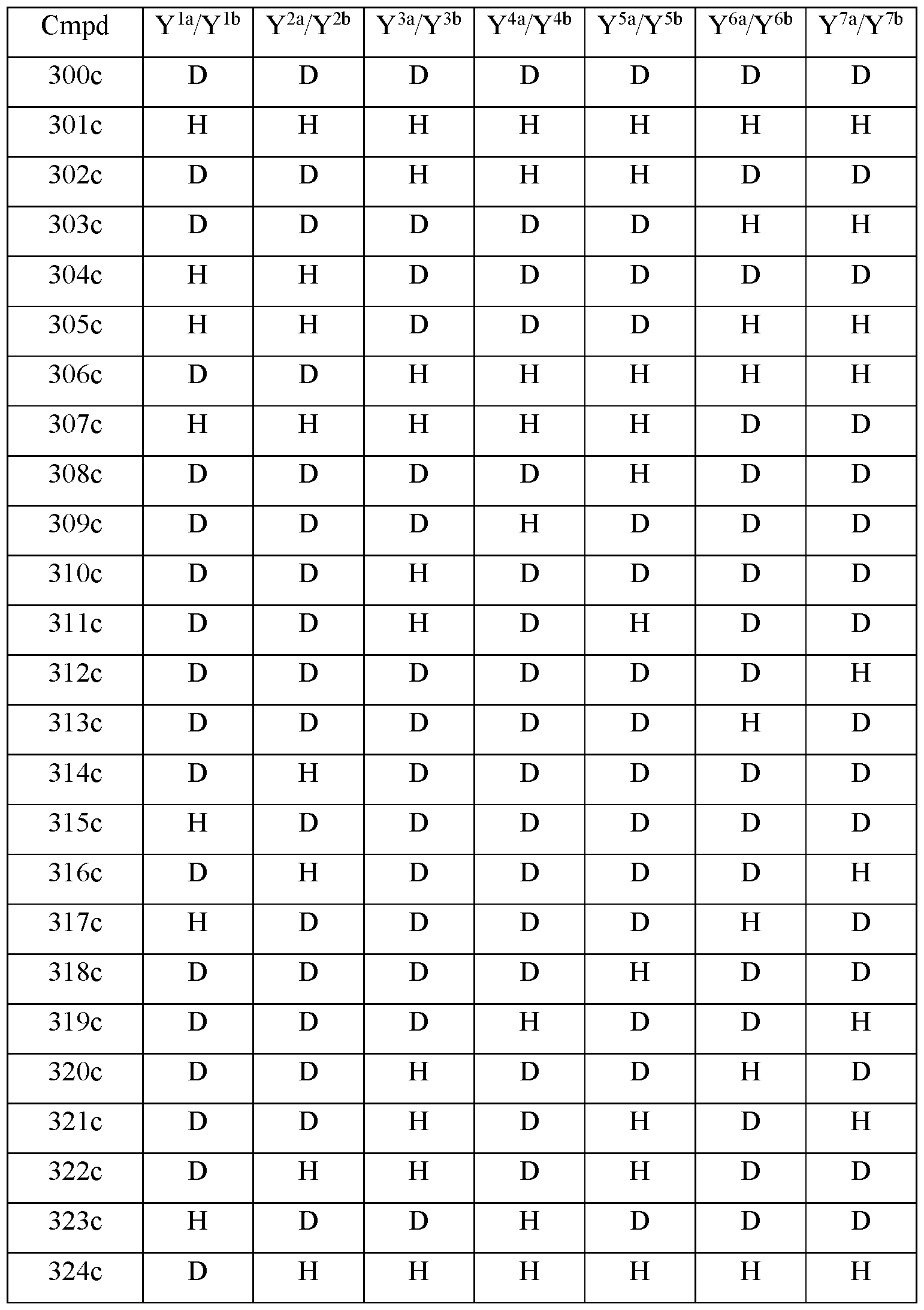
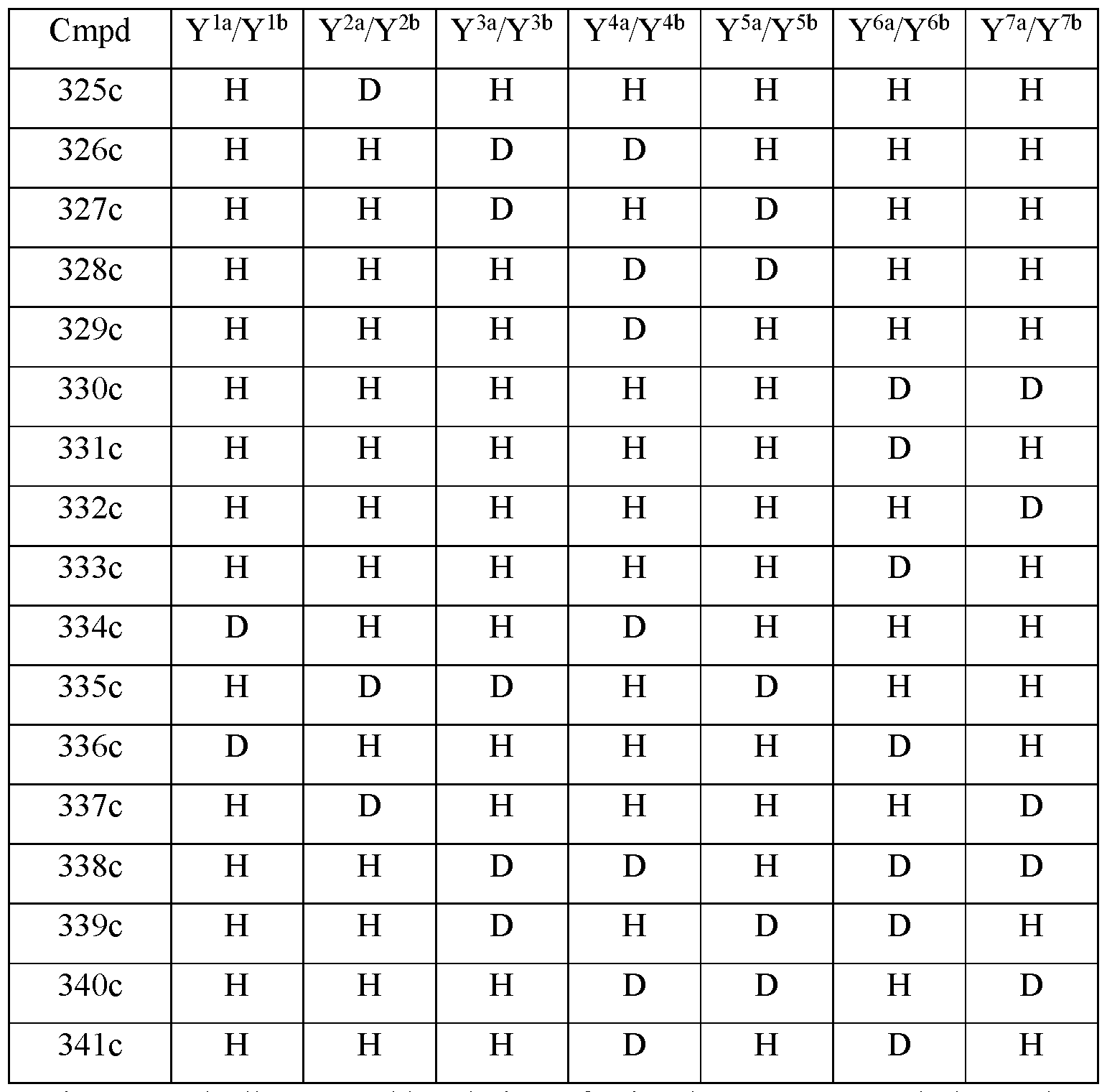
- the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table lc (below):
- R is CH 3 ;
- R 2 is palmitoyl;
- Y 1a and Y 1b are the same;
- Y 2a and Y 2b are the same;
- Y 3a and Y 3b are the same;
- Y 4a and Y 4b are the same;
- Y 5a and Y 5b are the same;
- Y 6a and Y 6b are the same;
- Y 7a and Y 711 are the same;
- Y 9 is hydrogen;
- Y 10 is hydrogen;
- Y 11a , Y 11b and Y 11c are each hydrogen;
- Y 12a and Y 12b are each hydrogen;
- Y 13a and Y 13b are each hydrogen; and
- the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table Id (below):
- R is CD3;
- R 2 is hydrogen;
- Y 1a and Y 1b are the same;
- Y 2a and Y 2b are the same;
- Y 3a and Y 3b are the same;
- Y 4a and Y 4b are the same;
- Y 5a and Y 5b are the same;
- Y 6a and Y 6b are the same;
- Y 7a and Y 711 are the same;
- Y 9 is hydrogen;
- Y 10 is hydrogen;
- Y 11a , Y 11b and Y 11c are each hydrogen;
- Y 12a and Y 12b are each hydrogen;
- Y 13a and Y 13b are each hydrogen; and
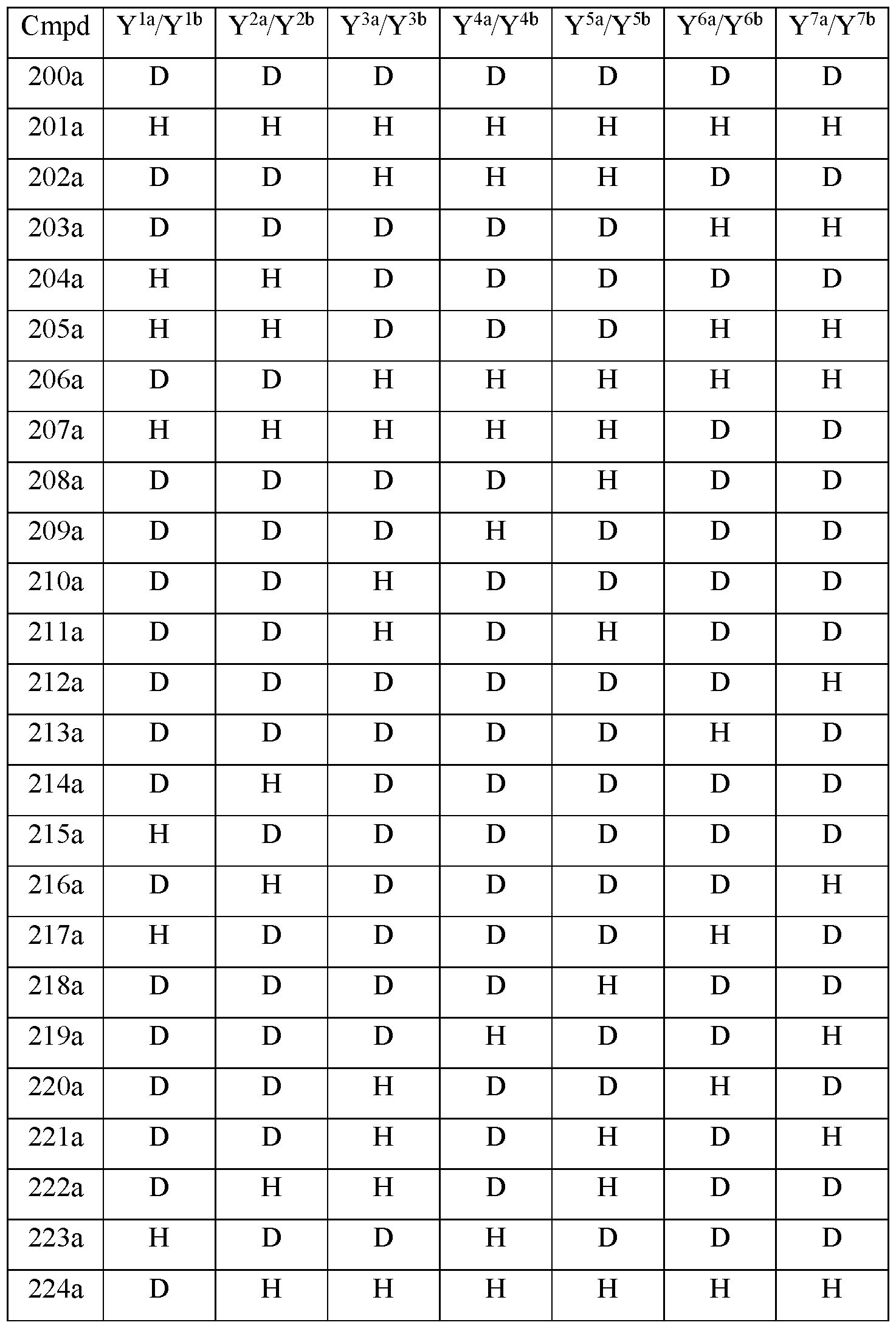
- the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2a (below):
- R is CH 3 ;
- R 2 is hydrogen; Y 1a and Y 1b are the same; Y 2a and Y 2b are the same; Y 3a and Y 3b are the same; Y 4a and Y 4b are the same; Y 5a and Y 5b are the same; Y 6a and Y 6b are the same; Y 7a and Y 711 are the same; Y 9 is hydrogen;
- Y 10 is hydrogen; Y 11a , Y 11b and Y 11c are each hydrogen; Y 12a and Y 12b are each hydrogen; Y 13a and Y 13b are each hydrogen; and the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2b (below):
- R is CD 3 ;
- R 2 is palmitoyl;
- Y 1a and Y 1b are the same;
- Y 2a and Y 2b are the same;
- Y 3a and Y 3b are the same;
- Y 4a and Y 4b are the same;
- Y 5a and Y 5b are the same;
- Y 6a and Y 6b are the same;
- Y 7a and Y 711 are the same;
- Y 9 is hydrogen;
- Y 10 is hydrogen;
- Y 11a , Y 11b and Y 11c are each hydrogen;
- Y 12a and Y 12b are each hydrogen;
- Y 13a and Y 13b are each hydrogen; and
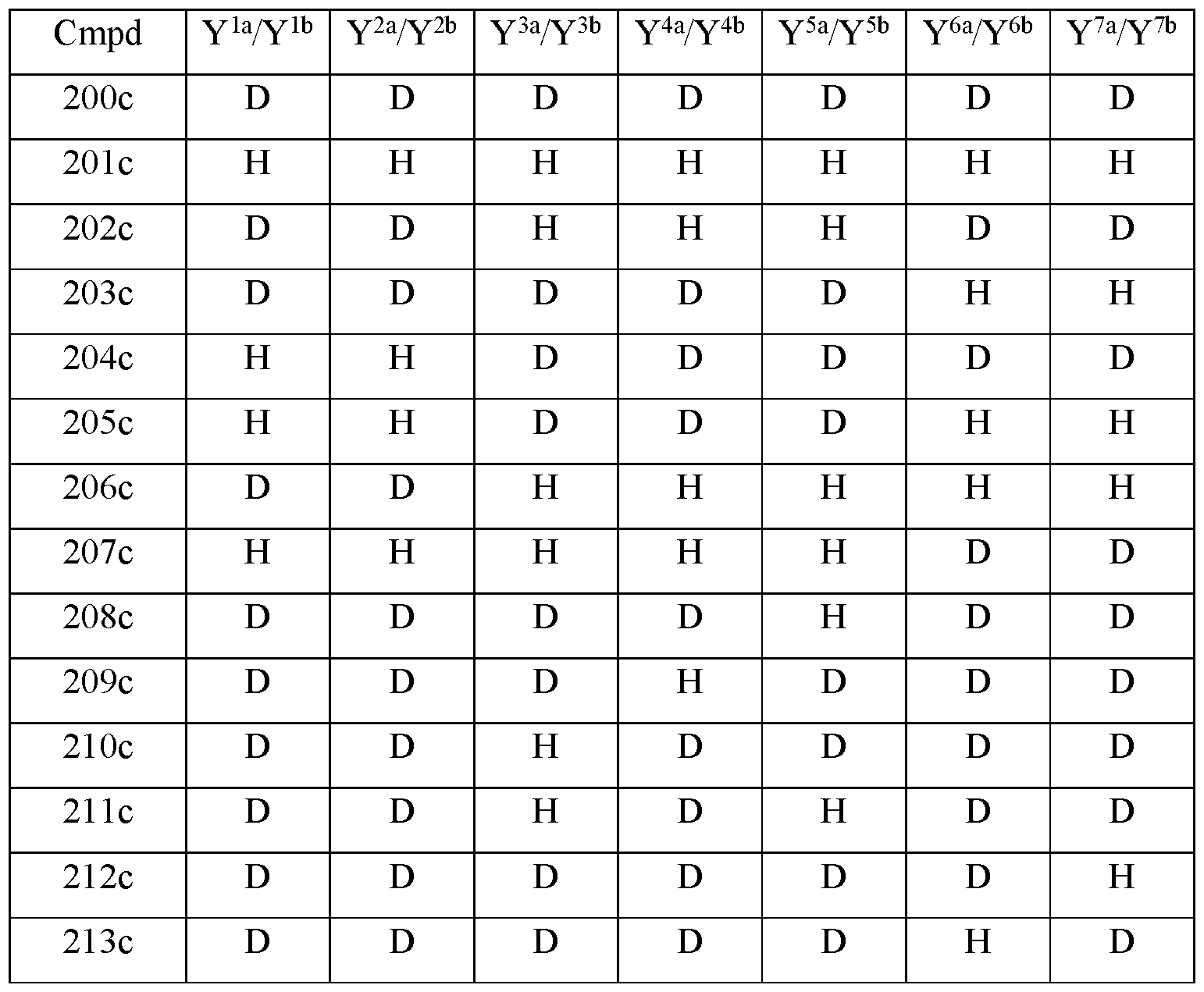
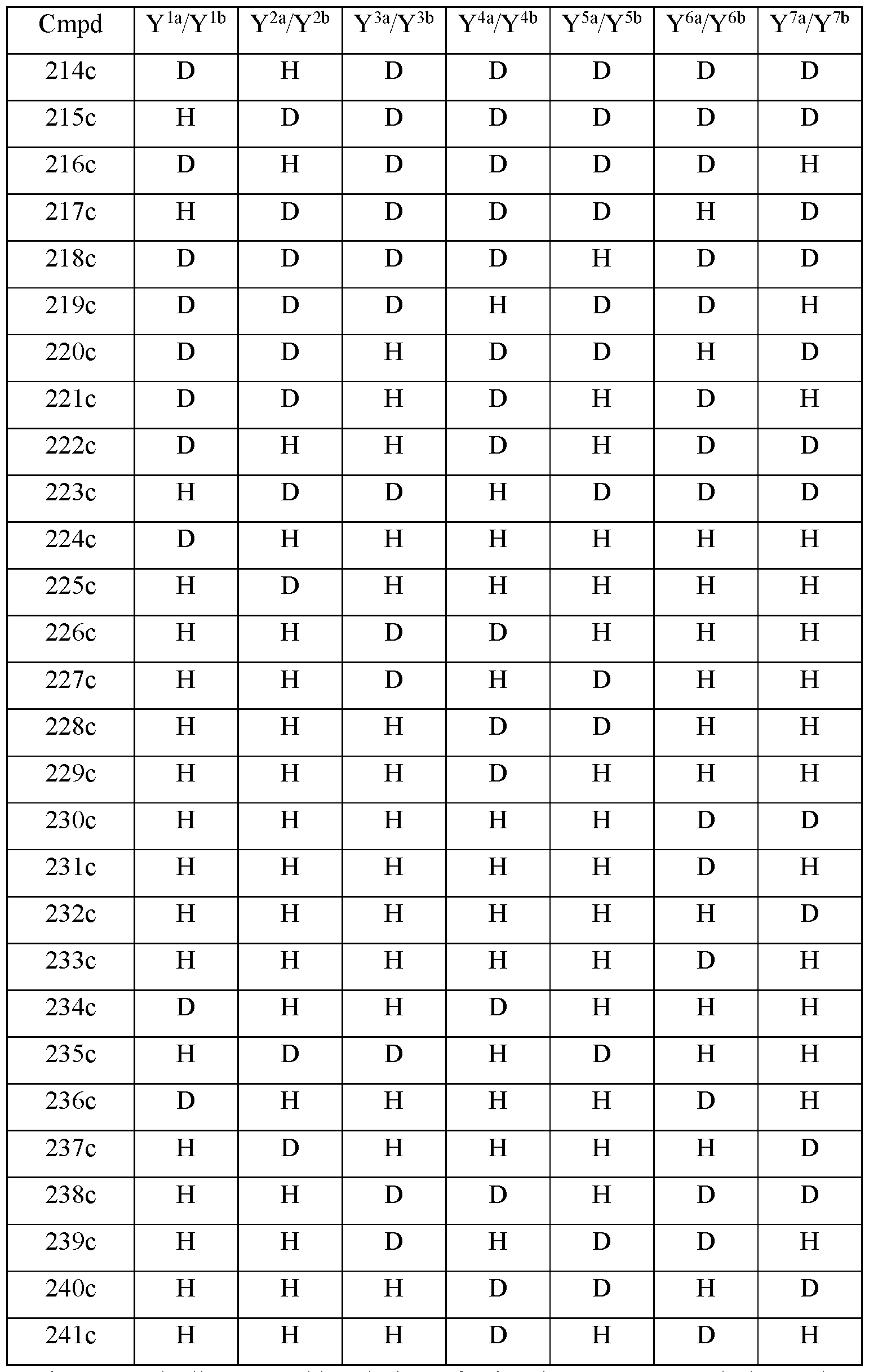
- the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2c (below):
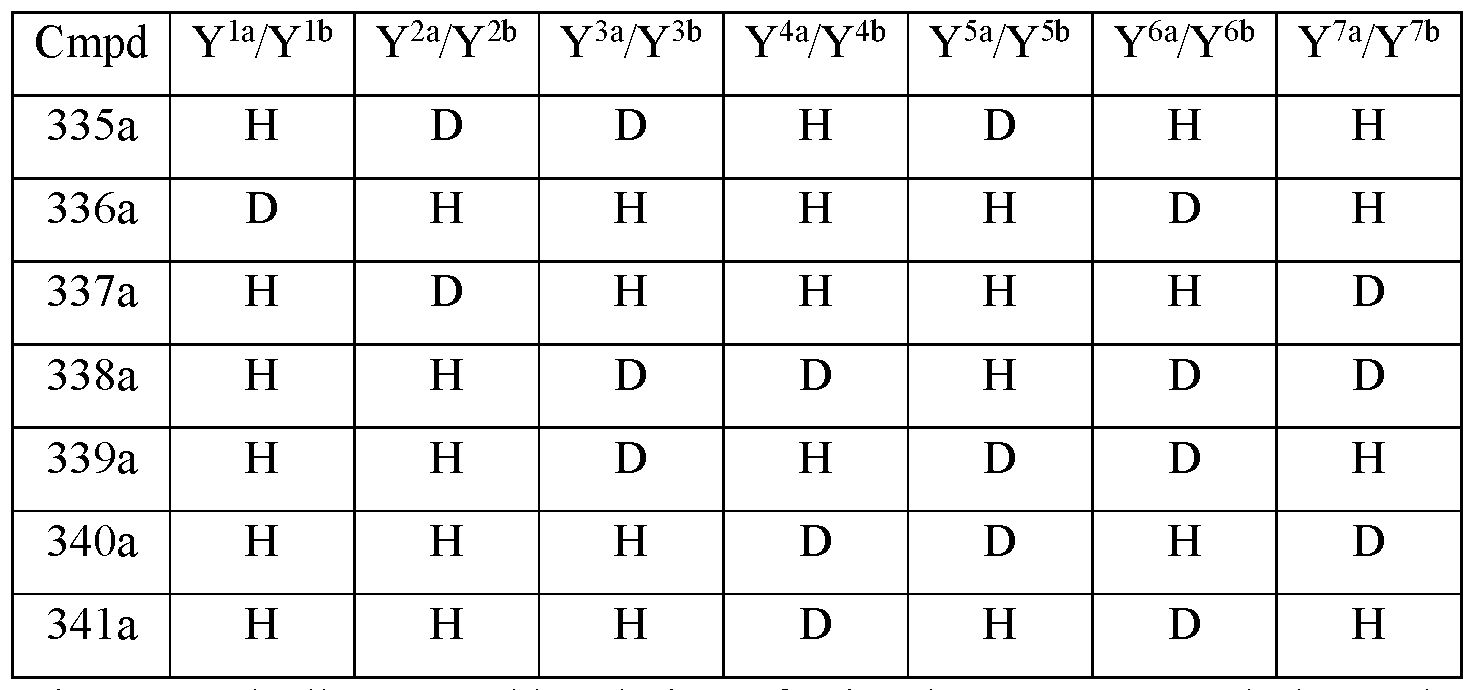
- R is CH3; R 2 is palmitoyl; Y 1a and Y 1b are the same; Y 2a and Y 2b are the same; Y 3a and Y 3b are the same; Y 4a and Y 4b are the same; Y 5a and Y 5b are the same; Y 6a and Y 6b are the same; Y 7a and Y 711 are the same; Y 9 is hydrogen; Y 10 is hydrogen; Y 11a , Y 11b and Y 11c are each hydrogen; Y 12a and Y 12b are each hydrogen; Y 13a and Y 13b are each hydrogen; and the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2d (below):
- R is CD 3 ;
- R 2 is hydrogen; Y 1a and Y 1b are the same; Y 2a and Y 2b are the same; Y 3a and Y 3b are the same; Y 4a and Y 4b are the same; Y 5a and Y 5b are the same; Y 6a and Y 6b are the same; Y 7a and Y 711 are the same; Y 9 is hydrogen;
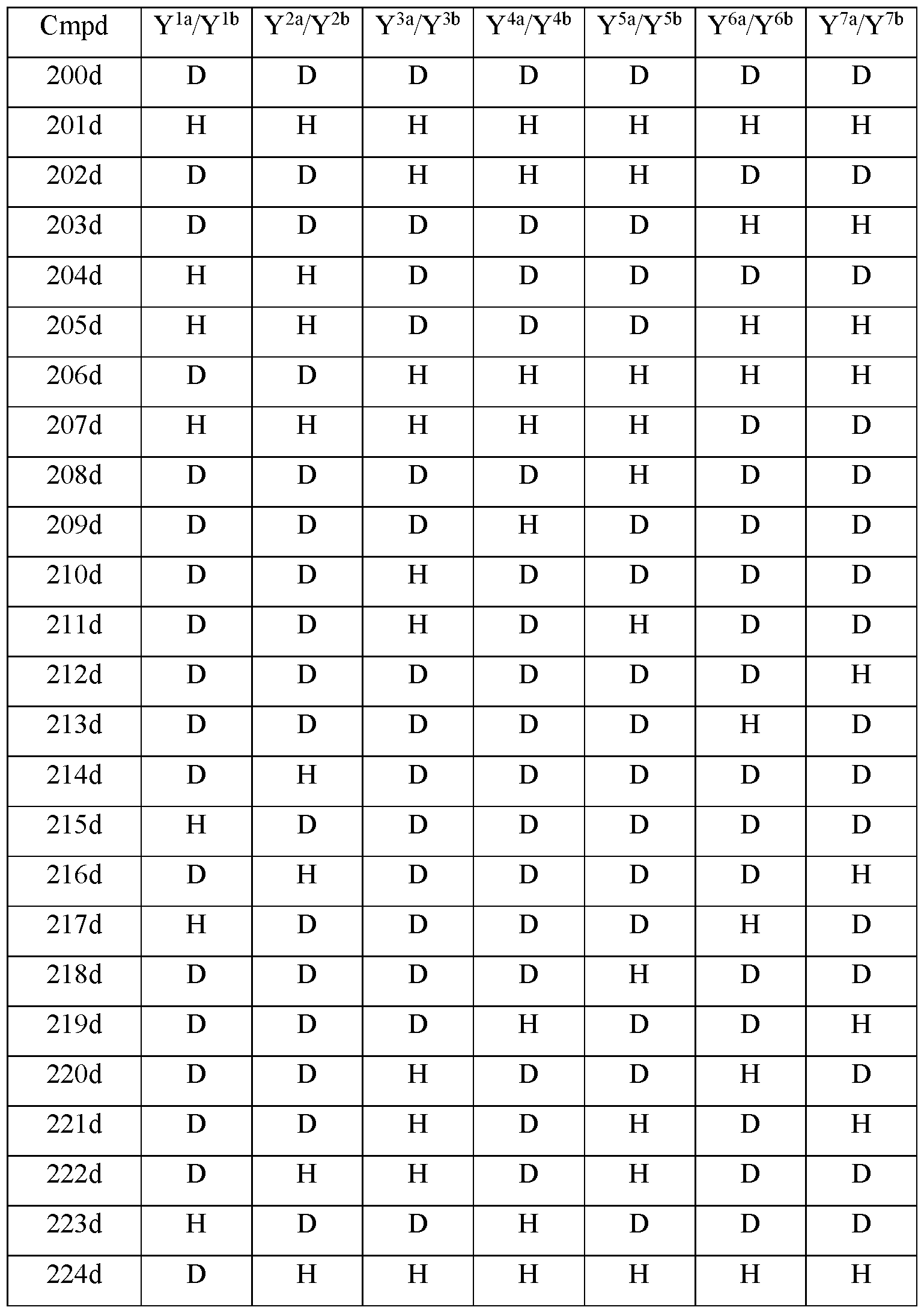
- Y 10 is hydrogen; Y 11a , Y 11b and Y 11c are each hydrogen; Y 12a and Y 12b are each hydrogen; Y 13a and Y 13b are each hydrogen; and the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3a (below):
- R is CH 3 ;
- R 2 is hydrogen; Y 1a and Y 1b are the same; Y 2a and Y 2b are the same; Y 3a and Y 3b are the same; Y 4a and Y 4b are the same; Y 5a and Y 5b are the same; Y 6a and Y 6b are the same; Y 7a and Y 711 are the same; Y 9 is hydrogen;
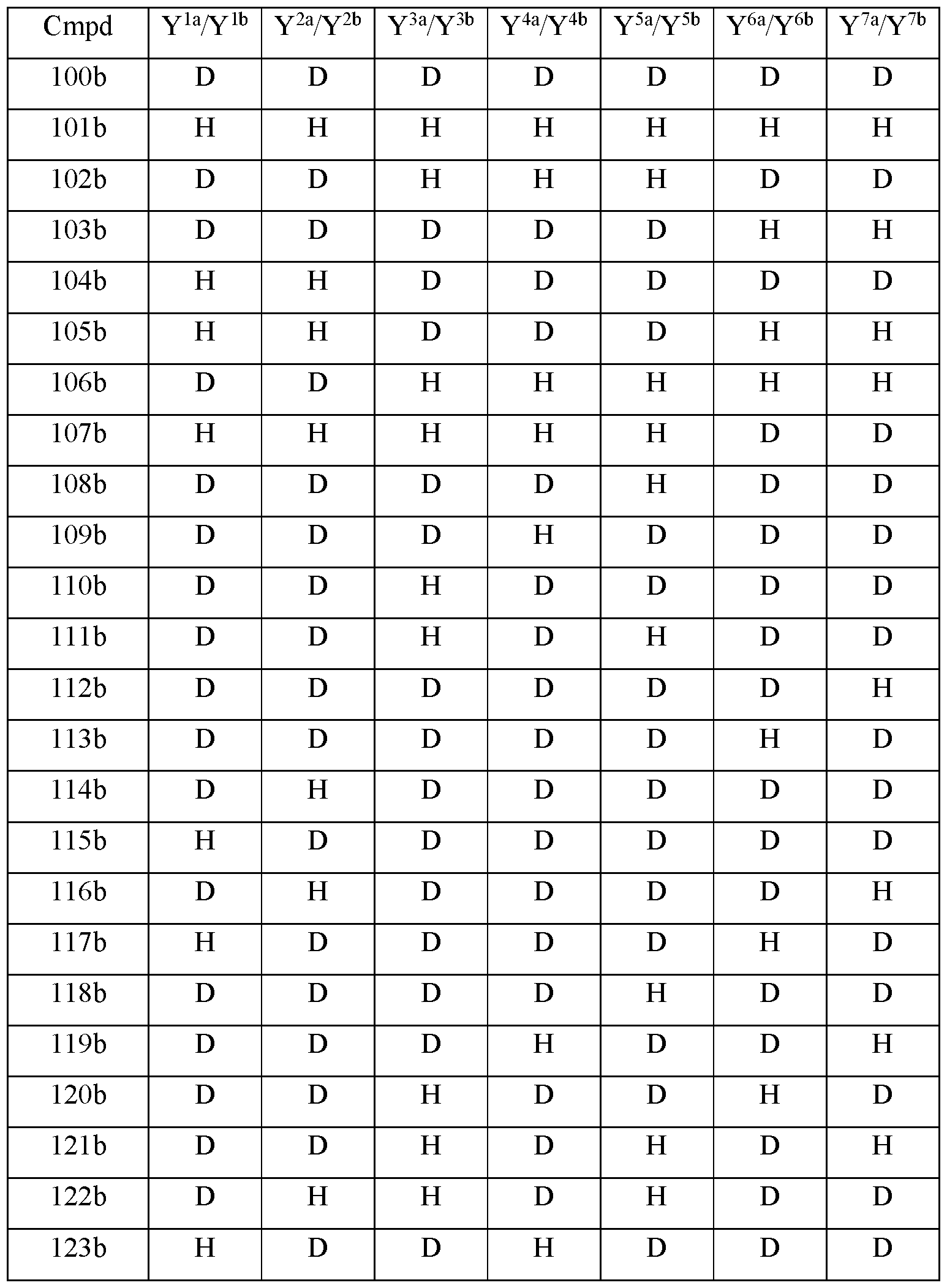
- Y 10 is hydrogen; Y 11a , Y 11b and Y 11c are each hydrogen; Y 12a and Y 12b are each hydrogen; Y 13a and Y 13b are each hydrogen; and the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3b (below):
- R is CD3; R 2 is palmitoyl; Y 1a and Y 1b are the same; Y 2a and Y 2b are the same; Y 3a and Y 3b are the same; Y 4a and Y 4b are the same; Y 5a and Y 5b are the same; Y 6a and Y 6b are the same; Y 7a and Y 711 are the same; Y 9 is hydrogen; Y 10 is hydrogen; Y 11a , Y 11b and Y 11c are each hydrogen; Y 12a and Y 12b are each hydrogen; Y 13a and Y 13b are each hydrogen; and the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3c (below):
- R is CH 3 ;
- R 2 is palmitoyl;
- Y 1a and Y 1b are the same;
- Y 2a and Y 2b are the same;
- Y 3a and Y 3b are the same;
- Y 4a and Y 4b are the same;
- Y 5a and Y 5b are the same;
- Y 6a and Y 6b are the same;
- Y 7a and Y 711 are the same;
- Y 9 is hydrogen;
- Y 10 is hydrogen;
- Y 11a , Y 11b and Y 11c are each hydrogen;
- Y 12a and Y 12b are each hydrogen;
- Y 13a and Y 13b are each hydrogen; and
- the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3d (below):
- the level of deuterium incorporation at each Y 1a or Y 1b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 2a or Y 2b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 3a or Y 3b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 4a or Y 4b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 5a or Y 5b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 6a or Y 6b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 7a or Y 711 is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 8a or Y 8b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at Y 9 is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at Y 10 is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 11a , Y 11b , or Y 11c is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 12a or Y 12b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- the level of deuterium incorporation at each Y 13a or Y 13b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
- deuterium incorporation at each designated deuterium atom is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, at least 97%, or at least 99%.
- Y 13b is hydrogen.
- Y 6a , Y a , Y 7a , Y 7b , Y 8a , Y 8b , and Y 14 are as described hereinabove.
- any atom not designated as deuterium in any of the embodiments set forth above is present at its natural isotopic abundance.
- the synthesis of compounds of Formula III may be readily achieved by synthetic chemists of ordinary skill by reference to the Exemplary Synthesis and Examples disclosed herein. Relevant procedures analogous to those of use for the preparation of compounds of Formula III and intermediates thereof are disclosed, for instance in PCT application No. PCT/US2008/003340 (published as
- Such methods can be carried out utilizing corresponding deuterated and optionally, other isotope-containing reagents and/or intermediates to synthesize the compounds delineated herein, or invoking standard synthetic protocols known in the art for introducing isotopic atoms to a chemical structure.
- R, R 2 , Y 1a , Y 1b , Y 2a , Y 2b , Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , Y a , Y 6a , Y 6b , Y 7a , Y 7b , Y 8a , Y 8b , Y 9 , Y 10 and Y 14 are as described hereinabove.
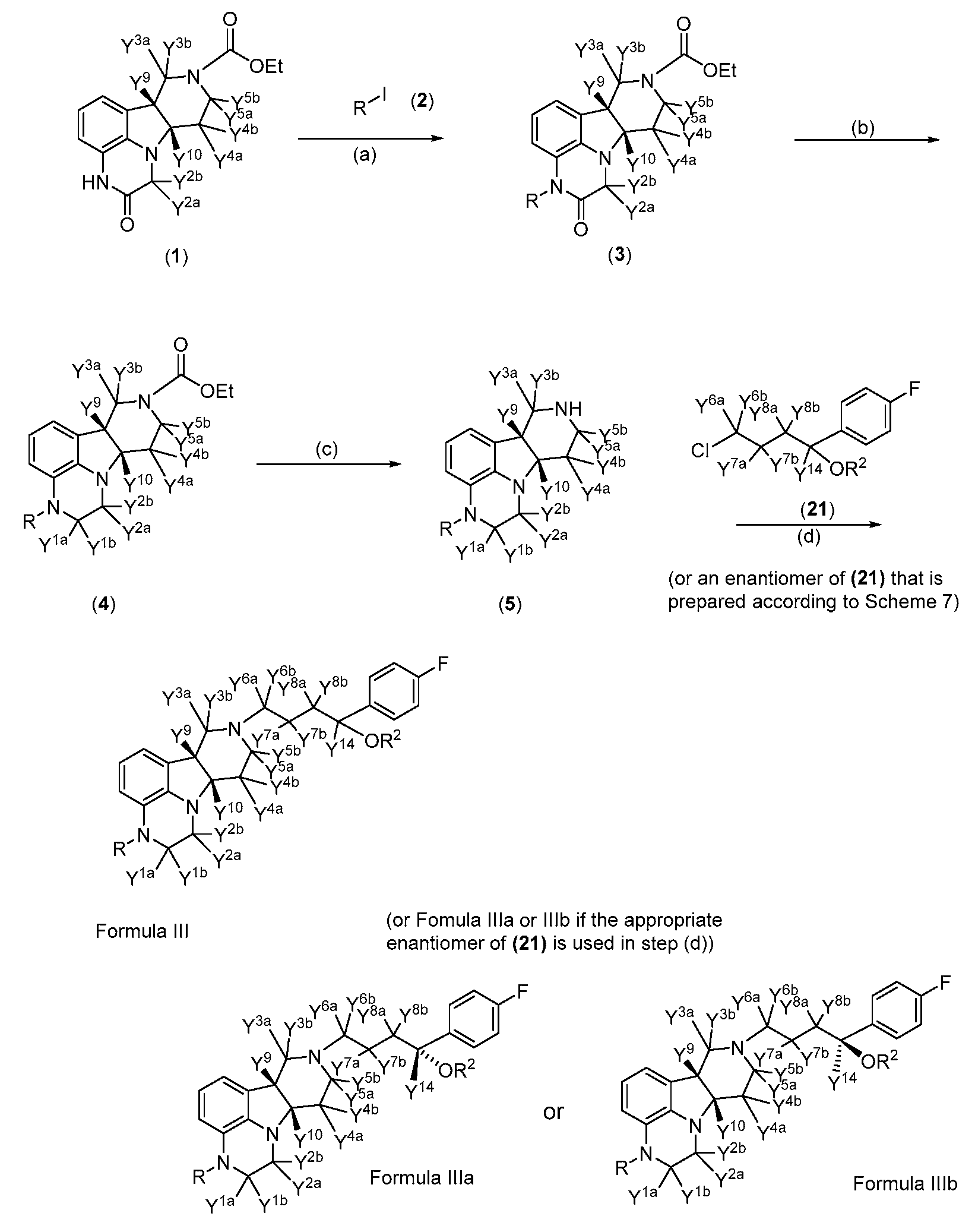
- a compound of Formula III, Ilia and/or Illb may be prepared as shown in Scheme 1 (which illustrates an exemplary embodiment wherein Y 11a , Y 11b , Y 11c , Y 12a , Y 12b , Y 13a , and Y 13b are each hydrogen).
- Scheme 1 which illustrates an exemplary embodiment wherein Y 11a , Y 11b , Y 11c , Y 12a , Y 12b , Y 13a , and Y 13b are each hydrogen.
- appropriately deuterated c/s-indoline intermediate (1) is treated with appropriately deuterated alkyl halide intermediate (2), in the presence of a base such as potassium carbonate or sodium hydride to produce appropriately deuterated alkylated c/s-indoline intermediate (3) which is reduced with an appropriately deuterated reducing agent such as Borane-d3, to produce appropriately deuterated ethyl ester protected c/s-indoline intermediate (4).
- Intermediate (4) is then treated with a base such as potassium hydroxide to remove the ethoxycarbonyl protection, furnishing appropriately deuterated free amino c/s-indoline intermediate (5).
- Intermediate (5) is alkylated with appropriately deuterated intermediate (21), prepared as shown in Scheme 5 below, under basic conditions to produce an appropriately deuterated compound of Formula III.
- appropriately deuterated intermediate (21) is first resolved into its enantiomers, as shown in Scheme 5, and alkylation of intermediate (5) with the desired enantiomer of (21) provides the corresponding optical isomer of Formula III, that is, a compound of Formula Ilia or Formula Illb.
- compounds of Formula III, Ilia and/or Illb can be prepared with greater than 90% or greater than 95% deuterium incorporation at each position designated as D (see below for details).
- deuterated intermediate (1) for use in the preparation of compounds of Formula III (or Ilia or Illb) according to Scheme 1 may be prepared from corresponding deuterated reagents exemplified in Scheme 2.
- Y 2a , Y 2b , Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , Y 5b , Y 9 and Y 10 are as described hereinabove.
- Reagents and conditions (a) NaN0 2 , HOAc; (b) Zn, HOAc; (c) HOAc, HC1; (d) NaBD 3 CN, d-TFA, NH 4 OH, NaOH or NaBH 3 CN, TFA, NH4OH, NaOH; (e) chiral resolution
- compounds of Formula III can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99.5 %) deuterium incorporation at some or all of Y 2a , Y 2b , Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , Y 5b , Y 9 and Y 10 in Formula III (or Ilia or Illb).
- deuterated intermediate (8) for use in the preparation of intermediate (1), and hence of compounds of Formula III (or Ilia or Illb) according to Scheme 1, may be prepared from corresponding deuterated reagents exemplified in Scheme 3.
- Reagents and conditions (a) N 2 0 5 , Bismuth triflate; (b) EtN(Pr-i) 2 , KF,18-Crown-6; (c) H 2 , Pd.
- Intermediate 16 is commercially available. Using such commercially available reagents and deuterated reagents that can be readily prepared by known methods, compounds of Formula III (or Ilia or Illb) can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%) deuterium incorporation at Y 2a and/or Y 2b in Formula III (or Ilia or Illb).
- compounds of Formula III can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%) deuterium incorporation at Y 2a and/or Y 2b .
- Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , Y 5b are as described hereinabove; and Y 3aa and Y 3bb are selected from hydrogen and deuterium.
- Reagents and conditions (a) 1) HQ or TFA, 2) Ethyl chloroformate, Et3N.
- deuterated intermediates (20) are commercially available: N- Boc-4-Piperidone-2,2,3,3,5,5,6,6-ds (98 atom % D) (20a), [2,6- 2 H 4 ]-N-Boc-4- Piperidinone (98 atom % D) (20b), N-Boc-4-Piperidone-3,3,5,5-d 4 (98 atom % D) (20c).
- appropriately deuterated (20a), (20b), and (20c) may be prepared as described in WO 2010/108103.
- compounds of Formula III can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%)deuterium incorporation at some or all of Y 3a , Y 3b , Y 4a , Y 4b , Y 5a , and or Y a in Formula III (or Ilia or Illb).
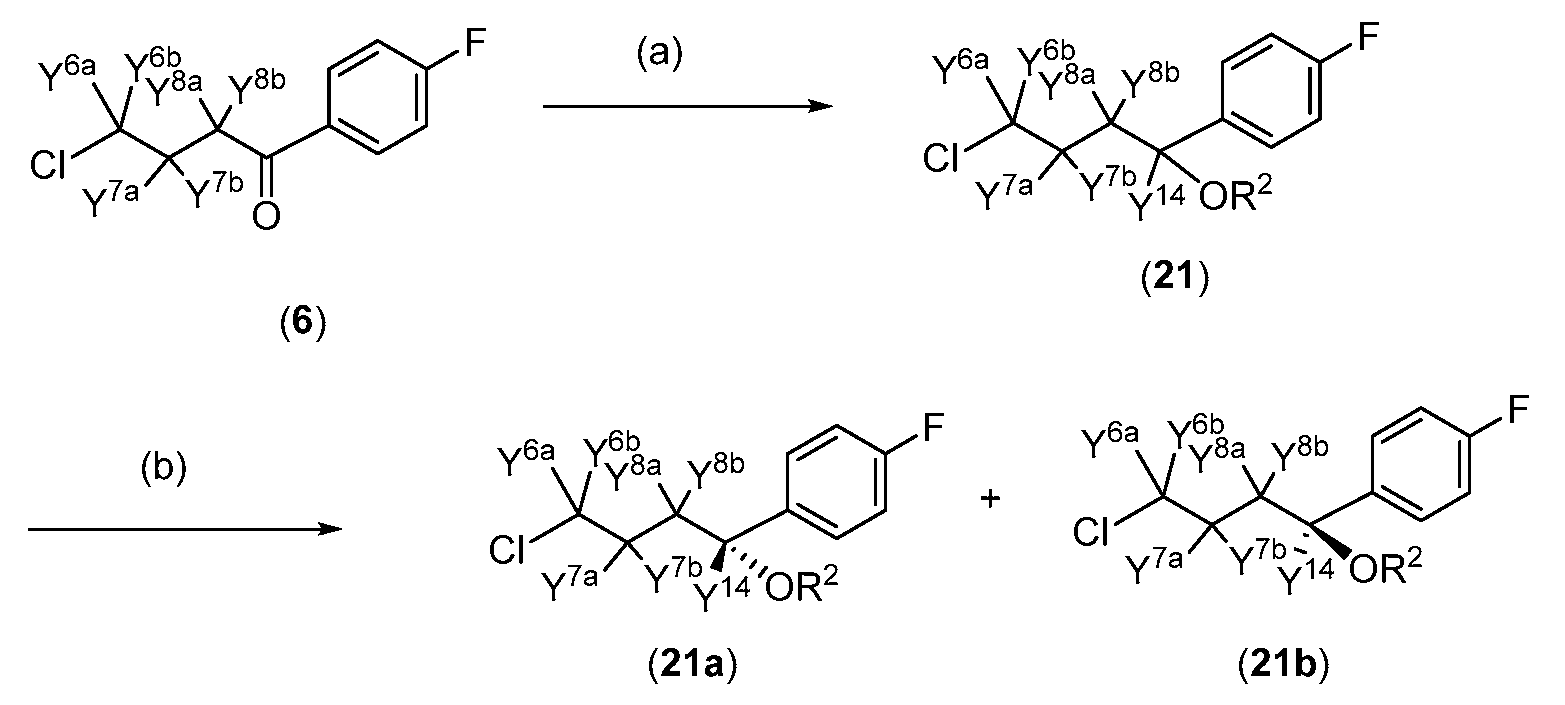
- deuterated intermediate (21) for use in the preparation of compounds of Formula III (or Ilia or Illb) according to Scheme 1, may be prepared from corresponding deuterated reagents exemplified in Scheme 5.
- Reagents and conditions (a) reduction with BD3 or other D source, (b) (optional) chiral resolution
- deuterated intermediate (6) for use in the preparation of intermediate (21), in turn used in the preparation of compounds of Formula III (or Ilia or Illb) according to Scheme 1, may be prepared from corresponding deuterated reagents exemplified in Scheme 6.
- compounds of Formula III can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%) deuterium incorporation at some or all of Y 6a , Y 6b , Y 7a , Y 7 *, Y 8a Y* Y 9 , Y 10 and/or ⁇ 14
- the invention also provides pharmaceutical compositions comprising an effective amount of a compound of Formula III (e.g., including any of the formulae herein, such as Formula Ilia or Illb), or a pharmaceutically acceptable salt of said compound; and a pharmaceutically acceptable carrier.
- a compound of Formula III e.g., including any of the formulae herein, such as Formula Ilia or Illb
- a pharmaceutically acceptable carrier e.g., including any of the formulae herein, such as Formula Ilia or Illb
- the carrier(s) are "acceptable" in the sense of being compatible with the other ingredients of the formulation and, in the case of a pharmaceutically acceptable carrier, not deleterious to the recipient thereof in an amount used in the medicament.
- Pharmaceutically acceptable carriers, adjuvants and vehicles that may be used in the pharmaceutical compositions of this invention include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene- polyoxypropylene-block polymers, polyethylene glycol and wool fat.
- ion exchangers alumina, aluminum stearate, lecithin
- serum proteins such as human serum albumin
- buffer substances such as phosphate
- the solubility and bioavailability of the compounds of the present invention in pharmaceutical compositions may be enhanced by methods well-known in the art.
- One method includes the use of lipid excipients in the formulation. See “Oral Lipid-Based Formulations: Enhancing the Bioavailability of Poorly Water- Soluble Drugs (Drugs and the Pharmaceutical Sciences),” David J. Hauss, ed. Informa Healthcare, 2007; and “Role of Lipid Excipients in Modifying Oral and Parenteral Drug Delivery: Basic Principles and Biological Examples," Kishor M. Wasan, ed. Wiley-Interscience, 2006.
- Another known method of enhancing bioavailability is the use of an amorphous form of a compound of this invention optionally formulated with a poloxamer, such as LUTROLTM and PLURONICTM (BASF Corporation), or block copolymers of ethylene oxide and propylene oxide. See United States patent 7,014,866; and United States patent publications 20060094744 and 20060079502.
- compositions of the invention include those suitable for oral, rectal, nasal, topical (including buccal and sublingual), vaginal or parenteral (including subcutaneous, intramuscular, intravenous and intradermal) administration.
- the compound of the formulae herein is administered transdermally (e.g., using a transdermal patch or iontophoretic techniques).
- Other formulations may conveniently be presented in unit dosage form, e.g., tablets, sustained release capsules, and in liposomes, and may be prepared by any methods well known in the art of pharmacy. See, for example, Remington: The Science and Practice of Pharmacy, Lippincott Williams & Wilkins, Baltimore, MD (20th ed. 2000).
- Such preparative methods include the step of bringing into association with the molecule to be administered ingredients such as the carrier that constitutes one or more accessory ingredients.
- the compositions are prepared by uniformly and intimately bringing into association the active ingredients with liquid carriers, liposomes or finely divided solid carriers, or both, and then, if necessary, shaping the product.
- the compound is administered orally.
- compositions of the present invention suitable for oral administration may be presented as discrete units such as capsules, sachets, or tablets each containing a predetermined amount of the active ingredient; a powder or granules; a solution or a suspension in an aqueous liquid or a non-aqueous liquid; an oil-in-water liquid emulsion; a water-in-oil liquid emulsion; packed in liposomes; or as a bolus, etc.
- Soft gelatin capsules can be useful for containing such suspensions, which may beneficially increase the rate of compound absorption.
- carriers that are commonly used include lactose and corn starch.
- Lubricating agents such as magnesium stearate, are also typically added.
- useful diluents include lactose and dried cornstarch.
- aqueous suspensions are administered orally, the active ingredient is combined with emulsifying and suspending agents. If desired, certain sweetening and/or flavoring and/or coloring agents may be added.
- compositions suitable for oral administration include lozenges comprising the ingredients in a flavored basis, usually sucrose and acacia or tragacanth; and pastilles comprising the active ingredient in an inert basis such as gelatin and glycerin, or sucrose and acacia.
- compositions suitable for parenteral administration include aqueous and nonaqueous sterile injection solutions which may contain anti-oxidants, buffers, bacteriostats and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which may include suspending agents and thickening agents.
- the formulations may be presented in unit-dose or multi-dose containers, for example, sealed ampules and vials, and may be stored in a freeze dried (lyophilized) condition requiring only the addition of the sterile liquid carrier, for example water for injections, immediately prior to use.
- Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules and tablets.
- Such injection solutions may be in the form, for example, of a sterile injectable aqueous or oleaginous suspension.
- This suspension may be formulated according to techniques known in the art using suitable dispersing or wetting agents (such as, for example, Tween 80) and suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally-acceptable diluent or solvent, for example, as a solution in 1,3- butanediol.
- the acceptable vehicles and solvents that may be employed are mannitol, water, Ringer's solution and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- Fatty acids, such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions.
- These oil solutions or suspensions may also contain a long-chain alcohol diluent or dispersant.
- R 2 is C(0)-C 1-21 alkyl. In some embodiments, R 2 is C(0)-C5-!9 alkyl. In some embodiments, R 2 is C(0)-C9-n alkyl. In some embodiments, R 2 is C(0)-Cis alkyl. In some embodiments, R 2 is palmitoyl.
- compositions of this invention may be administered in the form of suppositories for rectal administration.
- These compositions can be prepared by mixing a compound of this invention with a suitable non-irritating excipient which is solid at room temperature but liquid at the rectal temperature and therefore will melt in the rectum to release the active components.
- suitable non-irritating excipient include, but are not limited to, cocoa butter, beeswax and polyethylene glycols.
- compositions of this invention may be administered by nasal aerosol or inhalation.
- Such compositions are prepared according to techniques well-known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art. See, e.g.: Rabinowitz JD and Zaffaroni AC, US Patent 6,803,031, assigned to Alexza Molecular Delivery Corporation.
- Topical administration of the pharmaceutical compositions of this invention is especially useful when the desired treatment involves areas or organs readily accessible by topical application.
- the pharmaceutical composition should be formulated with a suitable ointment containing the active components suspended or dissolved in a carrier.
- Carriers for topical administration of the compounds of this invention include, but are not limited to, mineral oil, liquid petroleum, white petroleum, propylene glycol, polyoxyethylene polyoxypropylene compound, emulsifying wax, and water.
- the pharmaceutical composition can be formulated with a suitable lotion or cream containing the active compound suspended or dissolved in a carrier.
- Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2-octyldodecanol, benzyl alcohol, and water.
- the pharmaceutical compositions of this invention may also be topically applied to the lower intestinal tract by rectal suppository formulation or in a suitable enema formulation. Topically-transdermal patches and iontophoretic administration are also included in this invention.
- Application of the subject therapeutics may be local, so as to be administered at the site of interest.
- Various techniques can be used for providing the subject compositions at the site of interest, such as injection, use of catheters, trocars, projectiles, pluronic gel, stents, sustained drug release polymers or other device which provides for internal access.
- the compounds of this invention may be incorporated into compositions for coating an implantable medical device, such as prostheses, artificial valves, vascular grafts, stents, or catheters.
- an implantable medical device such as prostheses, artificial valves, vascular grafts, stents, or catheters.
- Suitable coatings and the general preparation of coated implantable devices are known in the art and are exemplified in US Patents 6,099,562; 5,886,026; and 5,304,121.
- the coatings are typically biocompatible polymeric materials such as a hydrogel polymer, polymethyldisiloxane, polycaprolactone, polyethylene glycol, polylactic acid, ethylene vinyl acetate, and mixtures thereof.
- the coatings may optionally be further covered by a suitable topcoat of fluorosilicone, polysaccharides, polyethylene glycol, phospholipids or combinations thereof to impart controlled release characteristics in the composition.
- Coatings for invasive devices are to be included within the definition of pharmaceutically acceptable carrier, adjuvant or vehicle, as those terms are used herein.
- the invention provides a method of coating an implantable medical device comprising the step of contacting said device with the coating composition described above. It will be obvious to those skilled in the art that the coating of the device will occur prior to implantation into a mammal.
- the invention provides a method of impregnating an implantable drug release device comprising the step of contacting said drug release device with a compound or composition of this invention.
- Implantable drug release devices include, but are not limited to, biodegradable polymer capsules or bullets, non-degradable, diffusible polymer capsules and biodegradable polymer wafers.
- the invention provides an implantable medical device coated with a compound or a composition comprising a compound of this invention, such that said compound is therapeutically active.
- the invention provides an implantable drug release device impregnated with or containing a compound or a composition comprising a compound of this invention, such that said compound is released from said device and is therapeutically active.
- composition of this invention may be painted onto the organ, or a composition of this invention may be applied in any other convenient way.
- a composition of this invention further comprises one or more additional therapeutic agents.
- the additional therapeutic agent may be selected from any compound or therapeutic agent known to have or that demonstrates advantageous properties when administered with a compound having the same mechanism of action as ITI-007.
- Such agents include those indicated as being useful in combination with ITI-007, including but not limited to, those described in PCT application No. PCT/US2008/009357 (published as WO2009/017836).
- the additional therapeutic agent is selected from selective serotonin re-uptake inhibitor (SSRI) (e.g. citalopram (Celexa, Cipramil, Cipram, Dalsan, Recital, Emocal, Sepram, Seropram, Citox, Cital); dapoxetine (Priligy); escitalopram (Lexapro, Cipralex, Seroplex, Esertia); fluoxetine (Depex, Prozac, Fontex, Seromex, Seronil, Sarafem, Lactose, Motivest, Flutop, Fluctin (EUR), Fluox (NZ), Depress (UZB), Lovan (AUS), Prodep (IND)); fluvoxamine (Luvox, Fevarin, Faverin, Dumyrox, Favoxil, Movox); indalpine (Upstene); paroxetine (Paxil, Seroxat, Sereupin, Aropax, Derox
- SSRI selective
- cholinesterase inhibitor e.g., acetylcholinesterase inhibitor
- Tacrine rivastigmine
- donepezil Aricept
- galantamine Razadyne, formerly called Reminyl
- NMD A N-Methyl D- Asparate receptor antagonist
- memantine combination of agents useful for the prophylaxis or treatment of Alzheimer's disease (donepezil and memantine), anti-depressant, anti-psychotic, other hypnotic agents, agents use to treat Parkinson's disease or mood disorders, compounds that modulate GABA activity (e.g.
- doxepin doxepin
- alprazolam bromazepam, clobazam
- clonazepam clorazepate
- diazepam flunitrazepam
- fiurazepam lorazepam
- midazolam nitrazepam
- oxazepam temazapam
- triazolam indiplon, zopiclone, eszopiclone, zaleplon
- Zolpidem gabaxadol
- vigabatrin tiagabine
- EVT 201 Euvotec Pharmaceuticals
- estazolam a GABA-B agonist
- 5-HT modulator e.g.
- a 5-HT 2 A agonist e.g. ketanserin, risperidone, eplivanserin, volinanserin (Sanofi-Aventis, France), pruvanserin, MDL 100907 (SanofiAventis, France), HY 10275 (Eli Lilly), APD 125 (Arena),
- a 5-HT 2 A inverse agonist etc.
- a melatonin agonist e.g. melatonin, ramelteon (ROZEREM®, Takeda Pharmaceuticals, Japan)
- VEC-162 Vehicle-to-live
- PD-6735 Phase II Discovery
- agomelatine an ion channel modulator
- SARI antagonist/reuptake inhibitor
- Org 50081 Organon -Netherlands
- ritanserin nefazodone
- serzone or trazodone
- an orexin receptor antagonist e.g. orexin, a 1,3-biarylurea, SB-334867-a (GlaxoSmithKline, UK
- H3 agonist or antagonist an H3 agonist or antagonist, a noradrenergic agonist or antagonist, a galanin agonist, a CRH antagonist, human growth hormone, a growth hormone agonist, estrogen, an estrogen agonist, a neurokinin- 1 drug (e.g. Casopitant), an anti-depressant (e.g.
- amitriptyline amoxapine, bupropion, citalopram, clomipramine, desipramine, doxepin, duloxetine, escitaloprame, fluoxetine, fluvoxamine, imipramine, isocarboxazid, maprotiline, mirtazapine, nefazodone, nortriptyline, paroxetine, phenlzine sulfate, protiptyline, sertraline, tranylcypromine, trazodone, trimipramine, and velafaxine), and an antipsychotic agent (e.g.
- chlorpromazine haloperidol, droperidol, fluphenazine, loxapine, mesoridazine molidone, pe ⁇ henazine, pimozide, prochlorperazine promazine, thioridazine, thiothixene, trifluoperazine, clozapine, aripiparazole, olanzapine, quetiapine, risperidone, ziprasidone and paliperidone), an atypical antipsychotic agent (e.g.
- 5HTLA agonist e.g. repinotan, sarizotan, eptapirone, buspirone or MN-305
- anti-Parkinson's agent e.g.
- L- dopa co-careldopa, duodopa, stalova, Symmetrel, benzotropine, biperiden, bromocryiptine, entacapone, pergolide, pramipexole, procyclidine, ropinirole, selegiline or tolcapone), levodopa and levodopa adjuncts (carbidopa, COMT inhibitors, MAO-B inhibitors), dopamine agonists, anticholinergics (e.g. levodopa), atypical stimulant (e.g. modafinil, adrafinil, or armodafinil), antidepressant (e.g.
- SSRJs selective serotonin reuptake inhibitors
- SNRJs serotonin-norepinephrine reuptake inhibitors
- tricyclic antidepressants antihistamine agents
- the invention provides separate dosage forms of a compound of this invention and one or more of any of the above-described additional therapeutic agents, wherein the compound and additional therapeutic agent are associated with one another.
- association with one another means that the separate dosage forms are packaged together or otherwise attached to one another such that it is readily apparent that the separate dosage forms are intended to be sold and administered together (within less than 24 hours of one another, consecutively or simultaneously).
- the compound of the present invention is present in an effective amount.
- the term is a pharmaceutical composition of the invention.
- an “effective amount” refers to an amount which, when administered in a proper dosing regimen, is sufficient to treat the target disorder.
- an effective amount of a compound of this invention can be 20 mg per dose. In one embodiment, an effective amount of a compound of this invention can be 30 mg per dose. In one embodiment, an effective amount of a compound of this invention can be 40 mg per dose. In one embodiment, an effective amount of a compound of this invention can be 60 mg per dose. In one embodiment, an effective amount of a compound of this invention can range from about 20 mg to about 60 mg per dose. In one embodiment, an effective amount of a compound of this invention can range from about 4 mg to about 120 mg per dose. In one embodiment, an effective amount of a compound of this invention can range from about 2 mg to about 300 mg per dose.
- an effective amount of a compound of this invention can range from about 0.2 mg to about 600 mg per dose. In one embodiment, the dose is administered twice daily. In one embodiment, the dose is administered once daily. In some embodiments, a dose of the injectable compositions of the present invention may be administered once a week, once every two weeks, or once a month.
- Effective doses will also vary, as recognized by those skilled in the art, depending on the diseases treated, the severity of the disease, the route of administration, the sex, age and general health condition of the subject, excipient usage, the possibility of co-usage with other therapeutic treatments such as use of other agents and the judgment of the treating physician. For example, guidance for selecting an effective dose can be determined by reference to the prescribing information for ITI-007.
- an effective amount of the additional therapeutic agent is between about 20% and 100% of the dosage normally utilized in a monotherapy regime using just that agent. Preferably, an effective amount is between about 70% and 100% of the normal monotherapeutic dose.
- the normal monotherapeutic dosages of these additional therapeutic agents are well known in the art. See, e.g., Wells et al., eds., Pharmacotherapy Handbook, 2nd Edition, Appleton and Lange, Stamford, Conn. (2000); PDR Pharmacopoeia, Tarascon Pocket Pharmacopoeia 2000, Deluxe Edition, Tarascon Publishing, Loma Linda, Calif. (2000), each of which references are incorporated herein by reference in their entirety.
- Some of the additional therapeutic agents referenced above may act synergistically with the compounds of this invention. When this occurs, it will allow the effective dosage of the additional therapeutic agent and/or the compound of this invention to be reduced from that required in a monotherapy. This has the advantage of minimizing toxic side effects of either the additional therapeutic agent of a compound of this invention, synergistically improving efficacy, improving ease of administration or use and/or reduced overall expense of compound preparation or formulation.
- the invention provides a method of modulating the activity of serotonin, glutamate, and dopamine receptors in a cell (in vivo or in vitro), comprising contacting a cell with one or more compounds of Formula III herein (e.g., Formula Ilia or Illb), or a pharmaceutically acceptable salt thereof.
- the cell is contacted in vitro.
- the cell is contacted in vivo.
- the cell is contacted ex vivo.
- the invention provides a method of modulating the activity of a 5-hydroxytryptamine (5-HT) receptor.
- the method comprises contacting a 5-hydroxytryptamine (5-HT) receptor in a cell, in vivo or in vitro, such that activity of the 5-hydroxytryptamine (5-HT) receptor is modulated (e.g., increased or decreased).
- the invention provides a method of modulating the activity of 5-HT 2 A receptor.
- the invention provides a method of antagonizing the activity of 5-HT 2 A receptor.
- the invention provides a method of modulating the activity of a serotonin transporter (SERT).
- SERT serotonin transporter
- the method comprises contacting a serotonin transporter in a cell, in vivo or in vitro, such that activity of the serotonin transporter is modulated (e.g., increased or decreased).
- the invention provides a method of inhibiting a serotonin transporter.
- the invention provides a method of modulating the activity of a dopamine (e.g. D 2 ) receptor.
- the method comprises contacting a dopamine receptor in a cell, in vivo or in vitro, such that activity of the dopamine receptor is modulated (e.g., increased or decreased).
- the invention provides a method of antagonizing a postsynaptic D2 receptor.
- the invention provides a method of partially agonizing a presynaptic striatal D2 receptor.
- the invention provides a method of modulating the activity of GluN2B-type N-methyl-D-aspartate (NMD A) receptor.
- the method comprises contacting a GluN2B-type N-methyl-D-aspartate receptor in a cell, in vivo or in vitro, such that activity of the GluN2B-type N-methyl-D-aspartate receptor is modulated (e.g., increased or decreased).
- the invention provides a method of indirectly modulating glutamatergic
- N-methyl-D- aspartate (NMD A) receptor GluN2B-type N-methyl-D- aspartate
- the invention provides a method of treating a disease that is beneficially treated by ITI-007 in a subject in need thereof, comprising the step of administering to the subject an effective amount of a compound or a composition of this invention.
- the subject is a patient in need of such treatment.
- diseases are well known in the art and are disclosed in, but not limited to the following patents and published applications: PCT application No. PCT/US2008/003340 (published as WO2008112280); PCT application No. PCT/US2009/001608 (published as WO2009114181); PCT application No. PCT/US2000/016373 (published as WO2000077010); PCT application No.
- PCT/US2014/029914 published asWO2014145192
- PCT application No. PCT/US2013/036515 published as WO2013155506
- PCT application No. PCT/US2013/036512 published as WO2013155504;
- Such diseases include, but are not limited to, addictive behavior (e.g. behavior associated with and or caused by physical and/or psychological dependence on narcotics, opiates, analgesics, painkillers, amphetamines, cocaine, heroin, opium, marijuana, alcohol, smoking, nicotine, gambling or eating), affective disorders, aggressive and assaultive behavior, aggressive behavior, agitation, agitation in Alzheimer's disease, agitation in autism and/or related autistic disorders, agitation in dementia (e.g. agitation in Alzheimer's disease), Alzheimer's disease, amyotrophic lateral sclerosis, anger, anorexia, anxiety, assaultive behavior, attention deficit disorder, attention deficit hyperactivity disorder, behavioral disturbances in patients with dementia (e.g.
- addictive behavior e.g. behavior associated with and or caused by physical and/or psychological dependence on narcotics, opiates, analgesics, painkillers, amphetamines, cocaine, heroin, opium, marijuana, alcohol, smoking, nicotine, gambling or
- agitation and irritability e.g. agitation irritation
- bipolar depression e.g. depression
- bipolar disorder e.g. depression
- brain spinal cord trauma e.g. bulemia
- cancer e.g., cancer
- central nervous system disorders e.g., central nervous system disorders
- cephalic pain e.g., cognition associated with schizophrenia, conditions associated with cephalic pain, corticobasal degenerations, dementia, dementia with Lewy bodies, dementing illnesses (e.g. senile dementia, Alzheimer's disease, Pick' s disease, frontotemporal dementia,
- Parkinson's disease e.g. schizophrenia
- depression e.g. refractory depression, co-morbid depression, MDD
- depression in a patient suffering from psychosis e.g. schizophrenia
- psychosis e.g. schizophrenia
- Down syndrome dyskinesia, elderly depression, excessive sleepiness, fatigue in conditions such as Parkinson's disease, frontotemporal dementia, gastrointestinal disorders, gastrointestinal disorders (e.g. dysfunction of the gastrointestinal tract motility), Huntington's disease, impulse control disorder, impulse control disorder or intermittent explosive disorder in patients suffering from psychosis (e.g. schizophrenia), intermittent explosive disorder, irritation, major depressive disorder, migraine, mild cognition impairment, mood disorders associated with Parkinson's disease, mood disorders associated with psychosis (e.g.
- schizophrenia multiple sclerosis, neurodegenerative diseases, obesity, obsessive-compulsive disorder, other psychiatric or neurological diseases, paraminenculear palsy, Parkinson's disease, physical or emotional outbursts, Pick's disease, post-traumatic stress disorder, prion disease, psychosis, schizophrenia, senile dementia, sexual disorders, sleep disorder (e.g. maintenance insomnia, frequent awakenings, waking up feeling unrefreshed, insomnia, narcolepsy, sleep apnea), sleep disorders (particularly sleep disorders associated with schizophrenia), sleep disorders associated with Parkinson's disease, sleep disorders associated with psychosis (e.g. schizophrenia), sleep disorders in patients suffering from dementia (e.g. Alzheimer's disease), social phobias, traumatic conditions, vascular dementia, Wemicke- Korsakoffs syndrome..
- dementia e.g. Alzheimer's disease
- social phobias traumatic conditions
- vascular dementia Wemicke- Korsakoffs syndrome.
- the method of this invention is used to treat a disease or condition selected from schizophrenia, schizophrenia having an acute exacerbation of psychosis, acute exacerbation of psychosis, depression, bipolar disorder, agitation, insomnia, dementia, and Alzheimer's Disease, the method comprising the step of administering to a subject in need thereof a therapeutically effective amount of a compound or a composition of the present invention.
- the method of this invention is used to treat a disease or condition selected from schizophrenia, schizophrenia having an acute exacerbation of psychosis, acute exacerbation of psychosis, and Alzheimer's Disease in a subject in need thereof.
- Identifying a subject in need of such treatment can be in the judgment of a subject or a health care professional and can be subjective (e.g. opinion) or objective (e.g. measurable by a test or diagnostic method).
- the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once a week.
- the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once every two weeks.
- the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once every three weeks. [181] In some embodiments, the method comprises administering to a subject an injectable composition comprising a compound disclosed herein twice a month.
- the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once a month.
- R 2 is C(0)-C 1-21 alkyl.
- R 2 is C(0)-Cs-i9 alkyl.
- R 2 is C(O)- C9-n alkyl.
- R 2 is C(0)-C 1 5 alkyl.
- R 2 is palmitoyl.
- any of the above methods of treatment comprises the further step of co-administering to the subject in need thereof one or more additional therapeutic agents.
- additional therapeutic agent may be made from any additional therapeutic agent known to be useful for co-administration with ITI-007.
- additional therapeutic agent is also dependent upon the particular disease or condition to be treated. Examples of additional therapeutic agents that may be employed in the methods of this invention are those set forth above for use in combination compositions comprising a compound of this invention and an additional therapeutic agent.
- co-administered means that the additional therapeutic agent may be administered together with a compound of this invention as part of a single dosage form (such as a composition of this invention comprising a compound of the invention and an additional therapeutic agent as described above) or as separate, multiple dosage forms. Alternatively, the additional agent may be administered prior to, consecutively with, or following the administration of a compound of this invention. In such combination therapy treatment, both the compounds of this invention and the additional therapeutic agent(s) are administered by conventional methods.
- composition of this invention comprising both a compound of the invention and an additional therapeutic agent, to a subject does not preclude the separate administration of that same therapeutic agent, any other additional therapeutic agent or any compound of this invention to said subject at another time during a course of treatment.
- the effective amount of the compound of this invention is less than its effective amount would be where the additional therapeutic agent is not administered. In another embodiment, the effective amount of the additional therapeutic agent is less than its effective amount would be where the compound of this invention is not administered. In this way, undesired side effects associated with high doses of either agent may be minimized. Other potential advantages (including without limitation improved dosing regimens and/or reduced drug cost) will be apparent to those of skill in the art.
- the invention provides the use of a compound of Formula III alone or together with one or more of the above-described additional therapeutic agents in the manufacture of a medicament, either as a single composition or as separate dosage forms, for treatment in a subject of a disease, disorder or symptom set forth above.
- Another aspect of the invention is a compound of Formula III for use in the treatment in a subject of a disease, disorder or symptom thereof delineated herein.
- Microsomal Assay Human liver microsomes (20 mg/mL) are obtained from Xenotech, LLC (Lenexa, KS). ⁇ -nicotinamide adenine dinucleotide phosphate, reduced form (NADPH), magnesium chloride (MgCh), and dimethyl sulfoxide (DMSO) are purchased from Sigma- Aldrich.
- 7.5 mM stock solutions of test compounds are prepared in DMSO.
- the 7.5 mM stock solutions are diluted to 12.5- 50 ⁇ in acetonitrile (ACN).
- ACN acetonitrile
- the 20 mg/mL human liver microsomes are diluted to 0.625 mg/mL in 0.1 M potassium phosphate buffer, pH 7.4, containing 3 mM MgCh.
- the diluted microsomes are added to wells of a 96-well deep-well polypropylene plate in triplicate.
- a 10 ⁇ , aliquot of the 12.5-50 ⁇ test compound is added to the microsomes and the mixture is pre- warmed for 10 minutes. Reactions are initiated by addition of pre- warmed NADPH solution.
- the final reaction volume is 0.5 mL and contains 0.5 mg/mL human liver microsomes, 0.25-1.0 ⁇ test compound, and 2 mM NADPH in 0.1 M potassium phosphate buffer, pH 7.4, and 3 mM MgCl 2 .
- the reaction mixtures are incubated at 37 °C, and 50 ⁇ , aliquots are removed at 0, 5, 10, 20, and 30 minutes and added to shallow- well 96- well plates which contain 50 ⁇ , of ice-cold ACN with internal standard to stop the reactions.
- the plates are stored at 4 °C for 20 minutes after which 100 ⁇ , of water is added to the wells of the plate before centrifugation to pellet precipitated proteins.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
This invention relates to novel 2,3,6b,7,8,9,10,10a-octahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxalines, and pharmaceutically acceptable salts thereof. This invention also provides compositions comprising a compound of this invention and the use of such compositions in methods of treating diseases and conditions that are beneficially treated by administering a serotonergic, dopaminergic, and glutamatergic modulator.
Description
DEUTERATED QUINOXALINE COMPOUNDS
CROSS-REFERENCE TO RELATED APPLICATIONS
[1] This application claims priority to U.S. Provisional Application No.
62/431,282, filed on December 7, 2016. The contents of the foregoing are hereby incorporated by reference.
BACKGROUND OF THE INVENTION
[2] Many current medicines suffer from poor absorption, distribution, metabolism and/or excretion (ADME) properties that prevent their wider use or limit their use in certain indications. Poor ADME properties are also a major reason for the failure of drug candidates in clinical trials. While formulation technologies and prodrug strategies can be employed in some cases to improve certain ADME properties, these approaches often fail to address the underlying ADME problems that exist for many drugs and drug candidates. One such problem is rapid metabolism that causes a number of drugs, which otherwise would be highly effective in treating a disease, to be cleared too rapidly from the body. A possible solution to rapid drug clearance is frequent or high dosing to attain a sufficiently high plasma level of drug. This, however, introduces a number of potential treatment problems such as poor patient compliance with the dosing regimen, side effects that become more acute with higher doses, and increased cost of treatment. A rapidly metabolized drug may also expose patients to undesirable toxic or reactive metabolites.
[3] Another ADME limitation that affects many medicines is the formation of toxic or biologically reactive metabolites. As a result, some patients receiving the drug may experience toxicities, or the safe dosing of such drugs may be limited such that patients receive a suboptimal amount of the active agent. In certain cases, modifying dosing intervals or formulation approaches can help to reduce clinical adverse effects, but often the formation of such undesirable metabolites is intrinsic to the metabolism of the compound.
[4] In some select cases, a metabolic inhibitor will be co-administered with a drug that is cleared too rapidly. Such is the case with the protease inhibitor class of drugs that are used to treat HIV infection. The FDA recommends that these drugs be co- dosed with ritonavir, an inhibitor of cytochrome P450 enzyme 3A4 (CYP3A4), the enzyme typically responsible for their metabolism (see Kempf, D.J. et al.,
Antimicrobial agents and chemotherapy, 1997, 41(3): 654-60). Ritonavir, however, causes adverse effects and adds to the pill burden for HIV patients who must already take a combination of different drugs. Similarly, the CYP2D6 inhibitor quinidine has been added to dextromethorphan for the purpose of reducing rapid CYP2D6 metabolism of dextromethorphan in a treatment of pseudobulbar affect. Quinidine, however, has unwanted side effects that greatly limit its use in potential combination therapy (see Wang, L et al., Clinical Pharmacology and Therapeutics, 1994, 56(6 Pt 1): 659-67; and FDA label for quinidine at www.accessdata.fda.gov).
[5] In general, combining drugs with cytochrome P450 inhibitors is not a satisfactory strategy for decreasing drug clearance. The inhibition of a CYP enzyme's activity can affect the metabolism and clearance of other drugs metabolized by that same enzyme. CYP inhibition can cause other drugs to accumulate in the body to toxic levels.
[6] A potentially attractive strategy for improving a drug's metabolic properties is deuterium modification. In this approach, one attempts to slow the CYP-mediated metabolism of a drug or to reduce the formation of undesirable metabolites by replacing one or more hydrogen atoms with deuterium atoms. Deuterium is a safe, stable, non-radioactive isotope of hydrogen. Compared to hydrogen, deuterium forms stronger bonds with carbon. In select cases, the increased bond strength imparted by deuterium can positively impact the ADME properties of a drug, creating the potential for improved drug efficacy, safety, and/or tolerability. At the same time, because the size and shape of deuterium are essentially identical to those of hydrogen, replacement of hydrogen by deuterium would not be expected to affect the biochemical potency and selectivity of the drug as compared to the original chemical entity that contains only hydrogen.
[7] Over the past 35 years, the effects of deuterium substitution on the rate of metabolism have been reported for a very small percentage of approved drugs (see, e.g., Blake, MI et al, J Pharm Sci, 1975, 64:367-91; Foster, AB, Adv Drug Res 1985, 14: 1-40 ("Foster"); Kushner, DJ et al, Can J Physiol Pharmacol 1999, 79-88; Fisher, MB et al, Curr Opin Drug Discov Devel, 2006, 9: 101-09 ("Fisher")). The results have been variable and unpredictable. For some compounds deuteration caused decreased metabolic clearance in vivo. For others, there was no change in
metabolism. Still others demonstrated increased metabolic clearance. The variability in deuterium effects has also led experts to question or dismiss deuterium
modification as a viable drug design strategy for inhibiting adverse metabolism (see Foster at p. 35 and Fisher at p. 101).
[8] The effects of deuterium modification on a drug's metabolic properties are not predictable even when deuterium atoms are incorporated at known sites of metabolism. Only by actually preparing and testing a deuterated drug can one determine if and how the rate of metabolism will differ from that of its non-deuterated counterpart. See, for example, Fukuto et al. (J. Med. Chem. 1991, 34, 2871-76). Many drugs have multiple sites where metabolism is possible. The site(s) where deuterium substitution is required and the extent of deuteration necessary to see an effect on metabolism, if any, will be different for each drug.
SUMMARY OF THE INVENTION
[9] This invention relates to novel 2,3,6b,7,8,9,10,10a-octahydro-lH- pyrido[3',4':4,5]pyrrolo[l,2,3-de]quinoxalines, and pharmaceutically acceptable salts thereof. Certain aspects of the present invention are directed to compounds of Formula III:
or a pharmaceutically acceptable salt thereof, wherein R is CH3, CH2D, CHD2 or CD3; R2 is hydrogen, deuterium, or C(0)-C1-21 alkyl; Y8a, Y®>, and Y14 are each deuterium; Y11a Y11b m^ Y11c ^ each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b
are each hydrogen; and Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y6a, Y6b, Y7a, Y711, Y9, and Y10 are independently selected from hydrogen and deuterium.
[10] Certain aspects of the present invention also provide compositions comprising a compound of this invention, including pharmaceutical compositions comprising a compound of this invention and a pharmaceutically acceptable carrier. Certain aspects of the present invention also provide the use of such compounds and compositions in methods of treating diseases and conditions that are beneficially treated by administering a serotonergic, dopaminergic, and glutamatergic modulator. Some exemplary embodiments include a method of treating a disease or condition selected from schizophrenia, depression, bipolar disorder, agitation, insomnia, dementia, and Alzheimer's Disease, the method comprising the step of administering to a subject in need thereof a therapeutically effective amount of a compound or pharmaceutical composition of the present invention.
DETAILED DESCRIPTION OF THE INVENTION
[11] ITI-007 (CAS Registry numbers 1187020-80-9, 313368-91-1) also known as rn-722 and l-(4-fluorophenyl)-4-[(6bR,10aS)-3-methyl-2,3,6b,7,8,9,10,10a- octahydro-lH-pyrido[3',4':4,5]pyrrolo[l,2,3-de]quinoxalin-8-yl]butan-l-one tosylate, modulates the serotonergic, dopaminergic, and glutamatergic systems.
[12] ITI-007 is currently in phase III clinical trials for the treatment of
schizophrenia, depression, bipolar disorder, and agitation; phase II clinical trials for the treatment of insomnia, phase Ib/II clinical trials for the treatment of dementia, including Alzheimer's disease; and is currently being tested for safety and efficacy in patients with an acute exacerbation of the aforementioned diseases.
[13] Despite the beneficial activities of ITI-007, there is a continuing need for new compounds to treat the aforementioned diseases and conditions. According to Abstracts of the 4th Biennial Schizophrenia International Research Conference / Schizophrenia Research 153, Supplement 1 (2014) S I -S384, ITI-007 is metabolized into two active metabolites, IC200131 and IC200161. IC200131 (CAS Registry number 1469343-42-7) is also known as (6bR,10aS)- -(4-fluorophenyl)- 2,3,6b,9,10,10a-hexahydro-3-methyl-lH-Pyrido[3',4':4,5]pyrrolo[l,2,3- de]quinoxaline-8(7H)-butanol. IC200131 is reported to be a serotonin-2A (5-HT2A) receptor antagonist and inhibitor of serotonin reuptake with roughly equal potency at each of these two targets. IC200131 is reported to have a longer plasma half-life than
IT1-007 or IC200161. In addition, IC200131 is reported to be able to undergo back- conversion into IT1-007. The metabolism of ΓΠ-007 is also discussed in
Psychopharmacology (2015) 232:2863-2872.
Definitions
[14] The term "treat" means decrease, suppress, attenuate, diminish, arrest, or stabilize the development or progression of a disease (e.g., a disease or disorder delineated herein), lessen the severity of the disease or improve the symptoms associated with the disease.
[15] "Disease" means any condition or disorder that damages or interferes with the normal function of a cell, tissue, or organ.
[16] As used herein, the term "subject" includes humans and non-human mammals. Non-limiting examples of non-human mammals include mice, rats, guinea pigs, rabbits, dogs, cats, monkeys, apes, pigs, cows, sheep, horses, etc.
[17] It will be recognized that some variation of natural isotopic abundance occurs in a synthesized compound depending upon the origin of chemical materials used in the synthesis. Thus, a preparation of ITI-007 will inherently contain small amounts of deuterated isotopologues. The concentration of naturally abundant stable hydrogen and carbon isotopes, notwithstanding this variation, is small and immaterial as compared to the degree of stable isotopic substitution of compounds of this invention. See, for instance, Wada, E et al., Seikagaku, 1994, 66:15; Gannes, LZ et al., Comp Biochem Physiol Mol lntegr Physiol, 1998, 119:725.
[18] In the compounds of this invention any atom not specifically designated as a particular isotope is meant to represent any stable isotope of that atom. Unless otherwise stated, when a position is designated specifically as "H" or '¾ydrogen", the position is understood to have hydrogen at its natural abundance isotopic composition. However, in certain embodiments where stated, when a position is designated specifically as "H" or "hydrogen", the position has at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% hydrogen. In some embodiments where stated, when a position is designated specifically as Ή" or "hydrogen", the position incorporates <20% deuterium, <10% deuterium, <5% deuterium, <4% deuterium, <3% deuterium, <2% deuterium, or <1% deuterium. Also unless otherwise stated, when a position is designated specifically as "D" or
"deuterium", the position is understood to have deuterium at an abundance that is at
least 3340 times greater than the natural abundance of deuterium, which is 0.015% (i.e., at least 50.1% incorporation of deuterium).
[19] The term "isotopic enrichment factor" as used herein means the ratio between the isotopic abundance and the natural abundance of a specified isotope.
[20] In other embodiments, a compound of this invention has an isotopic enrichment factor for each designated deuterium atom of at least 3500 (52.5% deuterium incorporation at each designated deuterium atom), at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75% deuterium), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5% deuterium incorporation).
[21] In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 52.5%. In some
embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 60%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 67.5%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 75%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 82.5%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 90%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 95%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 97.5%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 99%. In some embodiments, in a compound of this invention, each designated deuterium atom has deuterium incorporation of at least 99.5%.
[22] The term "isotopologue" refers to a species in which the chemical structure differs from a specific compound of this invention only in the isotopic composition thereof.
[23] The term "compound," when referring to a compound of this invention, refers to a collection of molecules having an identical chemical structure, except that there
may be isotopic variation among the constituent atoms of the molecules. Thus, it will be clear to those of skill in the art that a compound represented by a particular chemical structure containing indicated deuterium atoms, will also contain lesser amounts of isotopologues having hydrogen atoms at one or more of the designated deuterium positions in that structure. The relative amount of such isotopologues in a compound of this invention will depend upon a number of factors including the isotopic purity of deuterated reagents used to make the compound and the efficiency of incorporation of deuterium in the various synthesis steps used to prepare the compound.
[24] The invention also provides salts of the compounds of the invention.
[25] A salt of a compound of this invention is formed between an acid and a basic group of the compound, such as an amino functional group, or a base and an acidic group of the compound, such as a carboxyl functional group. According to another embodiment, the compound is a pharmaceutically acceptable acid addition salt.
[26] The term "pharmaceutically acceptable," as used herein, refers to a component that is, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and other mammals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit risk ratio. A "pharmaceutically acceptable salt" means any non-toxic salt that, upon administration to a recipient, is capable of providing, either directly or indirectly, a compound of this invention. A "pharmaceutically acceptable counterion" is an ionic portion of a salt that is not toxic when released from the salt upon administration to a recipient.
[27] Acids commonly employed to form pharmaceutically acceptable salts include inorganic acids such as hydrogen bisulfide, hydrochloric acid, hydrobromic acid, hydroiodic acid, sulfuric acid and phosphoric acid, as well as organic acids such as para-toluenesulfonic acid, salicylic acid, tartaric acid, bitartaric acid, ascorbic acid, maleic acid, besylic acid, fumaric acid, gluconic acid, glucuronic acid, formic acid, glutamic acid, methanesulfonic acid, ethanesulfonic acid, benzenesulfonic acid, lactic acid, oxalic acid, para-bromophenylsulfonic acid, carbonic acid, succinic acid, citric acid, benzoic acid and acetic acid, as well as related inorganic and organic acids. Such pharmaceutically acceptable salts thus include sulfate, pyrosulfate, bisulfate, sulfite, bisulfite, phosphate, monohydrogenphosphate, dihydrogenphosphate, metaphosphate, pyrophosphate, chloride, bromide, iodide, acetate, propionate, decanoate, caprylate, acrylate, formate, isobutyrate, caprate, heptanoate, propiolate,
oxalate, malonate, succinate, suberate, sebacate, fumarate, maleate, butyne-l,4-dioate, hexyne-l,6-dioate, benzoate, chlorobenzoate, methylbenzoate, dinitrobenzoate, hydroxybenzoate, methoxybenzoate, phthalate, terephthalate, sulfonate, xylene sulfonate, phenylacetate, phenylpropionate, phenylbutyrate, citrate, lactate, β- hydroxybutyrate, glycolate, maleate, tartrate, methanesulfonate, propanesulfonate, naphthalene- 1 -sulfonate, naphthalene-2- sulfonate, mandelate and other salts. In one embodiment, pharmaceutically acceptable acid addition salts include those formed with mineral acids such as hydrochloric acid and hydrobromic acid, and especially those formed with organic acids such as maleic acid. In one embodiment, the acids commonly employed to form pharmaceutically acceptable salts include the above- listed inorganic acids, wherein at least one hydrogen is replaced with deuterium.
[28] The compounds of the present invention may contain an asymmetric carbon atom, for example, as the result of deuterium substitution or otherwise. As such, compounds of this invention can exist as either individual enantiomers, or mixtures of the two enantiomers. Accordingly, a compound of the present invention may exist as either a racemic mixture or a scalemic mixture, or as individual respective stereoisomers that are substantially free from another possible stereoisomer. The term "substantially free of other stereoisomers" as used herein means less than 25% of other stereoisomers, preferably less than 10% of other stereoisomers, more preferably less than 5% of other stereoisomers and most preferably less than 2% of other stereoisomers are present. Methods of obtaining or synthesizing an individual enantiomer for a given compound are known in the art and may be applied as practicable to final compounds or to starting material or intermediates.
[29] Unless otherwise indicated, when a disclosed compound is named or depicted by a structure without specifying the stereochemistry and has one or more chiral centers, it is understood to represent all possible stereoisomers of the compound.
[30] The term "stable compounds," as used herein, refers to compounds which possess stability sufficient to allow for their manufacture and which maintain the integrity of the compound for a sufficient period of time to be useful for the purposes detailed herein (e.g., formulation into therapeutic products, intermediates for use in production of therapeutic compounds, isolatable or storable intermediate compounds, treating a disease or condition responsive to therapeutic agents).
[31] "D" and "d" both refer to deuterium. "Stereoisomer" refers to both enantiomers and diastereomers. "Tert" and "t-" each refer to tertiary. "US" refers to
the United States of America.
[32] "Substituted with deuterium" refers to the replacement of one or more hydrogen atoms with a corresponding number of deuterium atoms.
[33] Throughout this specification, a variable may be referred to generally (e.g.,"each R") or may be referred to specifically (e.g., R1, R2, R3, etc.). Unless otherwise indicated, when a variable is referred to generally, it is meant to include all specific embodiments of that particular variable.
Therapeutic Compounds
[34] The present invention in one embodiment provides a compound of Formula III:
or a pharmaceutically acceptable salt thereof,
wherein R is CH3, CH2D, CHD2 or CD3;
R2 is hydrogen, deuterium, or C(0)-d-21 alkyl;
Y8a, Y8b, and Y14 are each deuterium;
and Y1a Y1b Y2a Y2b Y3a Y3b Y4a Y4b Y5a Y5'5 Y6a Y6b Y7a Y7*5 Y9 Y10 Y11a Y11b, Y11c, Y12a, Y12b, Y13a, and Y13b are independently selected from hydrogen and deuterium.
[35] In some embodiments the compound of formula III is a compound of Formula Ilia:
or a pharmaceutically acceptable salt thereof,
wherein R, R2, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Ya, Y6a, Y6b, Y7a, Y7b, Y8a, Y8b, Y9, Y10, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, Y13b, and Y14 are as defined herein.
[36] In some embodiments the compound of formula III is a compound of Formula Illb:
or a pharmaceutically acceptable salt thereof,
wherein R, R2, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y6a, Y6b, Y7a, Y7b, Y8a, Y8b, Y9, Y10, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, Y13b, and Y14 are as defined herein.
[37] In some embodiments of any one of Formulae III, IIIa and IIIb, R, R2, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y6a, Y6b, Y7a, Y7b, Y9, Y10, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, and Y13b together comprise at least two atoms of deuterium.
[38] In some embodiments of any one of Formulae III, IIIa and IIIb, R, R2, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y6a, Y6b, Y7a, Y7b, Y9, Y10, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, and Y13b together comprise at least four atoms of deuterium.
[39] In some embodiments of any one of Formulae III, IIIa and IIIb, R, R2, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y6a, Y6b, Y7a, Y7b, Y9, Y10, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, and Y13b together comprise at least five atoms of deuterium.
[40] In some embodiments of any one of Formulae III, IIIa and IIIb, at least one of R, R2, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y6a, Y6b, Y7a, Y7b, Y9, Y10, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, and Y13b comprises hydrogen. In some embodiments of any one of Formulae III, IIIa and IIIb, at least one of R, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y6a, Y6b, Y7a, Y7b, Y9, Y10, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, and Y13b comprises hydrogen.
[41] In some embodiments of any one of Formulae III, IIIa and IIIb, R is CH3.
[42] In some embodiments of any one of Formulae III, IIIa and IIIb, R is CD3. 11
[43] In some embodiments of any one of Formulae III, Ilia and Illb, Y1a and Y1b are the same. In some aspects of these embodiments, Y1a and Y1b are each hydrogen.
In other aspects of these embodiments, Y1a and Y1b are each deuterium.
[44] In some embodiments of any one of Formulae III, Ilia and Illb, Y2a and Y2b are the same. In some aspects of these embodiments, Y2a and Y2b are each hydrogen.
In other aspects of these embodiments, Y2a and Y2b are each deuterium.
[45] In some embodiments of any one of Formulae III, Ilia and Illb, Y1a, Y1b, Y2a and Y2b are the same. In some aspects of these embodiments, Y1a, Y1b, Y2a and Y2b are each hydrogen. In other aspects of these embodiments, Y1a, Y1b, Y2a and Y2b are each deuterium.
[46] In some embodiments of any one of Formulae III, Ilia and Illb, Y1a and Y1b are each hydrogen; and Y2a and Y2b are each deuterium. In other embodiments, Y1a and Y1b are each deuterium; and Y2a and Y2b are each hydrogen.
[47] In some embodiments of any one of Formulae III, Ilia and Illb, Y3a and Y3b are the same. In some aspects of these embodiments, Y3a and Y3b are each hydrogen.
In other aspects of these embodiments, Y3a and Y3b are each deuterium.
[48] In some embodiments of any one of Formulae III, Ilia and Illb, Y4a and Y4b are the same. In some aspects of these embodiments, Y4a and Y4b are each hydrogen.
In other aspects of these embodiments, Y4a and Y4b are each deuterium.
[49] In some embodiments of any one of Formulae III, Ilia and Illb, Y5a and Y5b are the same. In some aspects of these embodiments, Y5a and Y* are each hydrogen.
In other aspects of these embodiments, Y5a and Y5b are each deuterium.
[50] In some embodiments of any one of Formulae III, Ilia and Illb, Y3a, Y3b, Y4a,
Y4b, Y5a and Y5b are the same. In some aspects of these embodiments, Y3a, Y3b, Y4a,
Y4b, Y5a and Y5b are each hydrogen. In other aspects of these embodiments, Y3a, Y3b,
Y4a, Y4b, Y5a and Y5b are each deuterium.
[51] In some embodiments of any one of Formulae III, Ilia and Illb, Y3a, Y3b, Y5a and ΥΆ are each hydrogen; and Y4a and Y4b are each deuterium. In other
embodiments, Y3a, Y3b, Y5a and Y5b are each deuterium; and Y4a and Y4b are each hydrogen.
[52] In some embodiments of any one of Formulae III, Ilia and Illb, Y6a and Y6b are the same. In some aspects of these embodiments, Y6a and Y* are each hydrogen. In other aspects of these embodiments, Y6a and Y6b are each deuterium.
[53] In some embodiments of any one of Formulae III, Ilia and Illb, Y7a and Y711 are the same. In some aspects of these embodiments, Y7a and Y715 are each hydrogen. In other aspects of these embodiments, Y7a and Y711 are each deuterium.
[54] In some embodiments of any one of Formulae III, Ilia and Illb, Y6a, Y*, Y7a, and Y711 are the same. In some aspects of these embodiments, Y6a, Ya, Y7a, and Y711 are each hydrogen. In other aspects of these embodiments, Y6a, Y6b, Y7a, and Y715 are each deuterium.
[55] In some embodiments of any one of Formulae III, Ilia and Illb, Y7a and Y711 are each hydrogen; and Y6a and Y6b are each deuterium. In other embodiments, Y7a and Y711 are each deuterium; and Y6a and Y6b are each hydrogen.
[56] In some embodiments of any one of Formulae III, Ilia and Illb, Y11a, Y11b, Y11c Yi2a yi2b; yi3a md yi3b each hydrogen. In other embodiments, Y11a, Y11b, Y11c, Y12a, Y12b, Y13a, and Y13b are each deuterium.
[57] In some embodiments of any one of Formulae III, Ilia and Illb, R2 is hydrogen.
[58] In some embodiments of any one of Formulae III, Ilia and Illb, R2 is deuterium.
[59] In some embodiments of any one of Formulae III, Ilia and Illb, R2 is C(0)-C1- 21 alkyl. In some embodiments, R2 is C(0)-Cs-19 alkyl. In some embodiments, R2 is C(0)-C9-n alkyl. In some embodiments, R2 is C(0)-C15 alkyl. In some embodiments, R2 is palmitoyl.
[60] In some embodiments of any one of Formulae III, Ilia and Illb, Y9 is hydrogen.
[61] In some embodiments of any one of Formulae III, Ilia and Illb, Y9 is deuterium.
[62] In some embodiments of any one of Formulae III, Ilia and Illb, Y10 is hydrogen.
[63] In some embodiments of any one of Formulae III, Ilia and Illb, Y10 is deuterium.
[64] In some embodiments of any one of Formulae III, Ilia and Illb, Y9 and Y10 are each hydrogen.
[65] In some embodiments of any one of Formulae III, Ilia and Illb, Y9 and Y10 are each deuterium.
[66] In some embodiments of any one of Formulae III, Ilia and Illb, Y11a, Y11b and Y11c are each hydrogen.
[67] In some embodiments of any one of Formulae III, Ilia and Illb, Y11a, Y11b and Y11c are each deuterium.
[68] In some embodiments of any one of Formulae III, Ilia and Illb, Y12a and Y12b are each hydrogen.
[69] In some embodiments of any one of Formulae III, Ilia and Illb, Y12a and Y12b are each deuterium.
[70] In some embodiments of any one of Formulae III, Ilia and Illb, Y13a and Y13b are each hydrogen.
[71] In some embodiments of any one of Formulae III, Ilia and Illb, Y13a and Y13b are each deuterium.
[72] In one embodiment, R is CD3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table la (below):
Table la: Exemplary Embodiments of Formula III
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[73] In one embodiment, R is CH3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table lb (below):
Table lb: Exemplary Embodiments of Formula III
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[74] In one embodiment, R is CD3; R2 is palmitoyl; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table lc (below):
Table lc: Exemplary Embodiments of Formula III
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[75] In one embodiment, R is CH3; R2 is palmitoyl; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table Id (below):
Table Id: Exemplary Embodiments of Formula III
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[76] In one embodiment, R is CD3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2a (below):
Table 2a: Exemplary Embodiments of Formula Ilia
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[77] In one embodiment, R is CH3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2b (below):
Table 2b: Exemplary Embodiments of Formula Ilia
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[78] In one embodiment, R is CD3; R2 is palmitoyl; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2c (below):
Table 2c: Exemplary Embodiments of Formula Ilia
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[79] In one embodiment, R is CH3; R2 is palmitoyl; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Ilia is selected from any one of the compounds (Cmpd) set forth in Table 2d (below):
Table 2d: Exemplary Embodiments of Formula Ilia
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[80] In one embodiment, R is CD3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3a (below):
Table 3a: Exemplary Embodiments of Formula Illb
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[81] In one embodiment, R is CH3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3b (below):
Table 3b: Exemplary Embodiments of Formula Illb
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[82] In one embodiment, R is CD3; R2 is palmitoyl; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3c (below):
Table 3c: Exemplary Embodiments of Formula Illb
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[83] In one embodiment, R is CH3; R2 is palmitoyl; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula Illb is selected from any one of the compounds (Cmpd) set forth in Table 3d (below):
Table 3d: Exemplary Embodiments of Formula Illb
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[84] In some embodiments, when Y1a or Y1b is deuterium, the level of deuterium incorporation at each Y1a or Y1b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[85] In some embodiments of a compound of this invention, when Y2a or Y2b is deuterium, the level of deuterium incorporation at each Y2a or Y2b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[86] In some embodiments of a compound of this invention, when Y3a or Y3b is deuterium, the level of deuterium incorporation at each Y3a or Y3b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[87] In some embodiments, when Y4a or Y4b is deuterium, the level of deuterium incorporation at each Y4a or Y4b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[88] In some embodiments, when Y5a or Y5b is deuterium, the level of deuterium incorporation at each Y5a or Y5b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[89] In some embodiments, when Y6a or Y6b is deuterium, the level of deuterium incorporation at each Y6a or Y6b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[90] In some embodiments, when Y7a or Y711 is deuterium, the level of deuterium incorporation at each Y7a or Y711 is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[91] In some embodiments, when Y8a or Y8b is deuterium, the level of deuterium incorporation at each Y8a or Y8b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[92] In some embodiments, when Y9 is deuterium, the level of deuterium incorporation at Y9 is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[93] In some embodiments, when Y10 is deuterium, the level of deuterium incorporation at Y10 is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[94] In some embodiments, when Y11a, Y11b, or Y11c is deuterium, the level of deuterium incorporation at each Y11a, Y11b, or Y11c is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[95] In some embodiments, when Y12a or Y12b is deuterium, the level of deuterium incorporation at each Y12a or Y12b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[96] In some embodiments, when Y13a or Y13b is deuterium, the level of deuterium incorporation at each Y13a or Y13b is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, is at least 97%, or at least 99%.
[97] In some embodiments, deuterium incorporation at each designated deuterium atom is at least 52.5%, at least 75%, at least 82.5%, at least 90%, at least 95%, at least 97%, or at least 99%.
[98] In some embodiments, at least one of Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, y5a y5b y6a y6b y7a y7b y8a y8b γ9 γ10 yUa yllb yllc yl2a yl2b yl3a gj^j
Y13b is hydrogen.
[99] In one embodiment, the following intermediates are novel and fall within the scope of this invention:
wherein Y6a, Ya, Y7a, Y7b, Y8a, Y8b, and Y14 are as described hereinabove.
[100] In another set of embodiments, any atom not designated as deuterium in any of the embodiments set forth above is present at its natural isotopic abundance.
[101] The synthesis of compounds of Formula III may be readily achieved by synthetic chemists of ordinary skill by reference to the Exemplary Synthesis and Examples disclosed herein. Relevant procedures analogous to those of use for the preparation of compounds of Formula III and intermediates thereof are disclosed, for instance in PCT application No. PCT/US2008/003340 (published as
WO2008112280); PCT application No. PCT/US2009/001608 (published as
WO2009114181); PCT patent application No. PCT/US2000/016373 (published as WO2000/077010); U.S. Patent Application Publication No. 2006/0178362; U.S. Patent No. 6,552,017; U.S. Patent No. 7,183,282; and Journal of Medicinal Chemistry (2014), 57(6), 2670-2682.
[102] Such methods can be carried out utilizing corresponding deuterated and optionally, other isotope-containing reagents and/or intermediates to synthesize the compounds delineated herein, or invoking standard synthetic protocols known in the art for introducing isotopic atoms to a chemical structure.
Exemplary Synthesis
[103] The synthesis of compounds of Formula III may be readily achieved by synthetic chemists of ordinary skill by reference to the Exemplary Synthesis. Relevant procedures analogous to those of use for the preparation of compounds of Formula III, and intermediates thereof are disclosed, for instance in PCT application No.
PCT/US2008/003340 (published as WO2008112280); PCT application No.
PCT/US2009/001608 (published as WO2009114181); PCT application No.
PCT/US2000/016373 (published as WO2000077010); US Patent Application No. 20060178362; US patent No. 6,552,017; US patent No. 7,183,282; and Journal of Medicinal Chemistry (2014), 57(6), 2670-2682. Such methods can be carried out utilizing corresponding deuterated and optionally, other isotope-containing reagents and/or intermediates to synthesize the compounds delineated herein, or invoking standard synthetic protocols known in the art for introducing isotopic atoms to a chemical structure.
[104] A convenient method for synthesizing compounds of Formula III is depicted in Scheme la below:
[105] Scheme 1: General Synthesis of Compounds of Formula HI (wherein Y11a. yUb yllc yl2a y12b yl3a md yl3b m each hydrogen)
wherein R, R2, Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Ya, Y6a, Y6b, Y7a, Y7b, Y8a, Y8b, Y9, Y10 and Y14 are as described hereinabove. Reagents and conditions: (a) K2C03 or NaH; (b) reduction with BD3 or other D source, or with BH3 or other H source; (c) KOH; (d) Et3N, KI.
[106] A compound of Formula III, Ilia and/or Illb may be prepared as shown in Scheme 1 (which illustrates an exemplary embodiment wherein Y11a, Y11b, Y11c, Y12a,
Y12b, Y13a, and Y13b are each hydrogen). In a manner analogous to a procedure described in US6552017, and by Li, P. et al., Journal of Medicinal Chemistry (2014), 57(6), 2670-2682, appropriately deuterated c/s-indoline intermediate (1) is treated with appropriately deuterated alkyl halide intermediate (2), in the presence of a base such as potassium carbonate or sodium hydride to produce appropriately deuterated alkylated c/s-indoline intermediate (3) which is reduced with an appropriately deuterated reducing agent such as Borane-d3, to produce appropriately deuterated ethyl ester protected c/s-indoline intermediate (4). Intermediate (4) is then treated with a base such as potassium hydroxide to remove the ethoxycarbonyl protection, furnishing appropriately deuterated free amino c/s-indoline intermediate (5).
Intermediate (5) is alkylated with appropriately deuterated intermediate (21), prepared as shown in Scheme 5 below, under basic conditions to produce an appropriately deuterated compound of Formula III. Alternatively, appropriately deuterated intermediate (21) is first resolved into its enantiomers, as shown in Scheme 5, and alkylation of intermediate (5) with the desired enantiomer of (21) provides the corresponding optical isomer of Formula III, that is, a compound of Formula Ilia or Formula Illb.
[107] Using commercially available reagents and deuterated reagents that can be readily prepared by known methods, compounds of Formula III, Ilia and/or Illb can be prepared with greater than 90% or greater than 95% deuterium incorporation at each position designated as D (see below for details).
[108] Appropriately deuterated intermediate (2), for use in the preparation of compounds of Formula III (or Ilia or Illb) according to Scheme la or lb, are commercially available, and are exemplified below.
Intermediate (2)
[109] The following are commercially available deuterated alkyl iodide
intermediates (2): Iodomethane-d3 (99.5 atom %D) (2a), Iodomethane-d2 (98 atom %D) (2b), Iodomethane-d (98 atom %D) (2c).
[110] Appropriately deuterated intermediate (1), for use in the preparation of
compounds of Formula III (or Ilia or Illb) according to Scheme 1 may be prepared from corresponding deuterated reagents exemplified in Scheme 2.
[Ill] Scheme 2: Preparation of Intermediate (1)
wherein Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y9 and Y10 are as described hereinabove.
Reagents and conditions: (a) NaN02, HOAc; (b) Zn, HOAc; (c) HOAc, HC1; (d) NaBD3CN, d-TFA, NH4OH, NaOH or NaBH3CN, TFA, NH4OH, NaOH; (e) chiral resolution
[112] In a manner analogous to a procedure described in US 6552017, and Li, P. et al., Journal of Medicinal Chemistry (2014), 57(6), 2670-2682, appropriately deuterated quinoxaline intermediate (8) is treated with sodium nitrite and acetic acid to furnish appropriately deuterated nitroso-quinoxaline intermediate (9), which is reduced with zinc in acetic acid to produce appropriately deuterated aryl hydrazine intermediate (10). Under acidic conditions at elevated temperature, Fischer indole cyclization of intermediate (10), and suitably protected and appropriately deuterated piperidone intermediate (11) produces appropriately deuterated tetracyclic indole
intermediate (12). Finally, cis-reduction of intermediate (12) using NaBD3CN or NaBH3CN in d-TFA or TFA under ambient reaction conditions, produces appropriately deuterated cis-indoline intermediate (1) and its enantiomer (la) having opposite stereochemistry at Y9 and Y10. Chiral resolution affords (1), which can be used in the next step in the preparation of compounds of Formula III (or Ilia or Illb).
[113] Using such commercially available reagents and deuterated reagents that can be readily prepared by known methods, compounds of Formula III (or Ilia or Illb) can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99.5 %) deuterium incorporation at some or all of Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Y5b, Y9 and Y10 in Formula III (or Ilia or Illb).
[114] Appropriately deuterated intermediate (8), for use in the preparation of intermediate (1), and hence of compounds of Formula III (or Ilia or Illb) according to Scheme 1, may be prepared from corresponding deuterated reagents exemplified in Scheme 3.
Scheme 3: Preparation of Intermediate (8)
Reagents and conditions: (a) N205, Bismuth triflate; (b) EtN(Pr-i)2, KF,18-Crown-6; (c) H2, Pd.
[115] In manner analogous to a procedure described by Qian, H. et al., Letters in
Organic Chemistry, 11(7), 509-512; 2014, bismuth triflate catalyzed nitration of appropriately deuterated aryl fluoride intermediate (16) affords appropriately deuterated fluoro-nitro aryl intermediate (17). Subsequent treatment of intermediate (17) with appropriately deuterated glycinate such as methyl ester intermediate (18) furnishes appropriately deuterated nitrophenyl ester intermediate (19), which upon reduction, and ring closure produces appropriately deuterated quinoxalinone intermediate (8) by analogy to a procedure described in WO 2003078398.
[116] Intermediate 16 is commercially available. Using such commercially available reagents and deuterated reagents that can be readily prepared by known methods, compounds of Formula III (or Ilia or Illb) can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%) deuterium incorporation at Y2a and/or Y2b in Formula III (or Ilia or Illb).
[117] Appropriately deuterated intermediate (18) exemplified below, for use in the preparation of intermediate (8) that may be ultimately converted in compounds of Formula III (or Ilia or Illb) according to the above schemes may be prepared from corresponding deuterated reagents.
Intermediate (18)
[118] Appropriately deuterated glycine ester intermediate (18a) is prepared according to a procedure described by Springer, J. et al., Journal of Labelled
Compounds and Radiopharmaceuticals, 50(2), 115-122; 2007.
[119] Using such commercially available reagents and deuterated reagents that can be readily prepared by known methods, compounds of Formula III can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%) deuterium incorporation at Y2a and/or Y2b.
[120] Appropriately deuterated intermediate (11), for use in the preparation of intermediate (1) and hence of compounds of Formula III (or Ilia or Illb) according to Scheme 1 may be prepared from corresponding deuterated reagents exemplified in Scheme 4.
[121] Scheme 4: Preparation of Intermediate (11)
wherein Y3a, Y3b, Y4a, Y4b, Y5a, Y5b are as described hereinabove; and Y3aa and Y3bb are selected from hydrogen and deuterium.
Reagents and conditions: (a) 1) HQ or TFA, 2) Ethyl chloroformate, Et3N.
[122] Using a procedure known to anyone skilled in the art, or by analogy to a procedure described in WO2008137436, appropriately deuterated Boc-protected intermediate (20) is treated with acid such as HC1 or TFA to remove the protecting group. In a manner analogous to a procedure described in WO 2009/037001, subsequent treatment with ethyl chloroformate in the presence of a base such as Et3N produces appropriately deuterated intermediate (11).
[123] The following deuterated intermediates (20) are commercially available: N- Boc-4-Piperidone-2,2,3,3,5,5,6,6-ds (98 atom % D) (20a), [2,6-2H4]-N-Boc-4- Piperidinone (98 atom % D) (20b), N-Boc-4-Piperidone-3,3,5,5-d4 (98 atom % D) (20c). Alternatively, appropriately deuterated (20a), (20b), and (20c) may be prepared as described in WO 2010/108103.
[124] Using such commercially available reagents and deuterated reagents that can be readily prepared by known methods, compounds of Formula III can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%)deuterium incorporation at some or all of Y3a, Y3b, Y4a, Y4b, Y5a, and or Ya in Formula III (or Ilia or Illb).
[125] Appropriately deuterated intermediate (21), for use in the preparation of compounds of Formula III (or Ilia or Illb) according to Scheme 1, may be prepared from corresponding deuterated reagents exemplified in Scheme 5.
[126] Scheme 5: Preparation of Intermediate (21)
Reagents and conditions: (a) reduction with BD3 or other D source, (b) (optional) chiral resolution
[127] As shown in Scheme 5, appropriately deuterated ketone (6) (which may be prepared as shown in Scheme 6 herein) is reduced with a source of deuterium such as BD3 to form the corresponding appropriately deuterated secondary alcohol (21). (21) is optionally subjected to a chiral resolution step to provide enantiomers (21a) and (21b). Either (21) or a desired enantiomer ((21a) or (21b)) may be then used as described in Scheme 1 above, to afford a compound of Formula III or the
corresponding enantiomer (Formula Ilia or Formula Illb).
[128] Appropriately deuterated intermediate (6), for use in the preparation of intermediate (21), in turn used in the preparation of compounds of Formula III (or Ilia or Illb) according to Scheme 1, may be prepared from corresponding deuterated reagents exemplified in Scheme 6.
[129] Scheme 6: Preparation of Intermediate (6)
wherein Y6a, Y* Y7a, Υ , Y8a, Y8b are as described hereinabove.
Reagents and conditions: (a) SOCI2, Zn(¾ (b) AICI3
[130] In a manner analogous to a procedure described in CN1654461, ring-opening of appropriately deuterated γ-butyrolactone intermediate (13) with SOCh in the presence of ZnCl2 produces appropriately deuterated chlorobutyric chloride intermediate (14) which is subsequently submitted to Friedel-Crafts acylation using appropriately deuterated aryl fluoride intermediate (15) in the presence of AlCh to furnish appropriately deuterated aryl chloro-butanone intermediate (6).
[131] The following intermediates (13) are commercially available: γ- Butyrolactone-d6 (98 atom %D) (13a). Appropriately deuterated intermediates (13c), (13e), and (13f) are prepared in accordance with a method described in WO
2014031840.
[132] Synthetic chemists of ordinary skills will appreciate that preparation of additional partially and appropriately deuterated analogs may be readily achieved using relevant and analogous methods known in the art that successfully accomplishes the synthesis of intermediates (13) and (14) for use in preparation of intermediate (6).
[133] Using such commercially available reagents and deuterated reagents that can be readily prepared by known methods, compounds of Formula III (or Ilia or Illb) can be prepared with greater than 90% or greater than 95% (e.g., up to 98% or 99%) deuterium incorporation at some or all of Y6a, Y6b, Y7a, Y7*, Y8a Y* Y9, Y10 and/or γ14
[134] The specific approaches and compounds shown above are not intended to be
limiting. The chemical structures in the schemes herein depict variables that are hereby defined commensurately with chemical group definitions (moieties, atoms, etc.) of the corresponding position in the compound formulae herein, whether identified by the same variable name (i.e., R1, R2, R3, etc.) or not. The suitability of a chemical group in a compound structure for use in the synthesis of another compound is within the knowledge of one of ordinary skill in the art.
[135] Additional methods of synthesizing compounds of Formula I and their synthetic precursors, including those within routes not explicitly shown in schemes herein, are within the means of chemists of ordinary skill in the art. Synthetic chemistry transformations and protecting group methodologies (protection and deprotection) useful in synthesizing the applicable compounds are known in the art and include, for example, those described in Larock R, Comprehensive Organic Transformations, VCH Publishers (1989); Greene, TW et al., Protective Groups in Organic Synthesis, 3rd Ed., John Wiley and Sons (1999); Fieser, L et al., Fieser and Fieser 's Reagents for Organic Synthesis, John Wiley and Sons (1994); and Paquette, L, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995) and subsequent editions thereof.
[136] Combinations of substituents and variables envisioned by this invention are only those that result in the formation of stable compounds.
Compositions
[137] The invention also provides pharmaceutical compositions comprising an effective amount of a compound of Formula III (e.g., including any of the formulae herein, such as Formula Ilia or Illb), or a pharmaceutically acceptable salt of said compound; and a pharmaceutically acceptable carrier. The carrier(s) are "acceptable" in the sense of being compatible with the other ingredients of the formulation and, in the case of a pharmaceutically acceptable carrier, not deleterious to the recipient thereof in an amount used in the medicament.
[138] Pharmaceutically acceptable carriers, adjuvants and vehicles that may be used in the pharmaceutical compositions of this invention include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium
hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene- polyoxypropylene-block polymers, polyethylene glycol and wool fat.
[139] If required, the solubility and bioavailability of the compounds of the present invention in pharmaceutical compositions may be enhanced by methods well-known in the art. One method includes the use of lipid excipients in the formulation. See "Oral Lipid-Based Formulations: Enhancing the Bioavailability of Poorly Water- Soluble Drugs (Drugs and the Pharmaceutical Sciences)," David J. Hauss, ed. Informa Healthcare, 2007; and "Role of Lipid Excipients in Modifying Oral and Parenteral Drug Delivery: Basic Principles and Biological Examples," Kishor M. Wasan, ed. Wiley-Interscience, 2006.
[140] Another known method of enhancing bioavailability is the use of an amorphous form of a compound of this invention optionally formulated with a poloxamer, such as LUTROL™ and PLURONIC™ (BASF Corporation), or block copolymers of ethylene oxide and propylene oxide. See United States patent 7,014,866; and United States patent publications 20060094744 and 20060079502.
[141] The pharmaceutical compositions of the invention include those suitable for oral, rectal, nasal, topical (including buccal and sublingual), vaginal or parenteral (including subcutaneous, intramuscular, intravenous and intradermal) administration. In certain embodiments, the compound of the formulae herein is administered transdermally (e.g., using a transdermal patch or iontophoretic techniques). Other formulations may conveniently be presented in unit dosage form, e.g., tablets, sustained release capsules, and in liposomes, and may be prepared by any methods well known in the art of pharmacy. See, for example, Remington: The Science and Practice of Pharmacy, Lippincott Williams & Wilkins, Baltimore, MD (20th ed. 2000).
[142] Such preparative methods include the step of bringing into association with the molecule to be administered ingredients such as the carrier that constitutes one or more accessory ingredients. In general, the compositions are prepared by uniformly and intimately bringing into association the active ingredients with liquid carriers, liposomes or finely divided solid carriers, or both, and then, if necessary, shaping the product.
[143] In certain embodiments, the compound is administered orally. Compositions of the present invention suitable for oral administration may be presented as discrete units such as capsules, sachets, or tablets each containing a predetermined amount of the active ingredient; a powder or granules; a solution or a suspension in an aqueous liquid or a non-aqueous liquid; an oil-in-water liquid emulsion; a water-in-oil liquid emulsion; packed in liposomes; or as a bolus, etc. Soft gelatin capsules can be useful for containing such suspensions, which may beneficially increase the rate of compound absorption.
[144] In the case of tablets for oral use, carriers that are commonly used include lactose and corn starch. Lubricating agents, such as magnesium stearate, are also typically added. For oral administration in a capsule form, useful diluents include lactose and dried cornstarch. When aqueous suspensions are administered orally, the active ingredient is combined with emulsifying and suspending agents. If desired, certain sweetening and/or flavoring and/or coloring agents may be added.
[145] Compositions suitable for oral administration include lozenges comprising the ingredients in a flavored basis, usually sucrose and acacia or tragacanth; and pastilles comprising the active ingredient in an inert basis such as gelatin and glycerin, or sucrose and acacia.
[146] Compositions suitable for parenteral administration include aqueous and nonaqueous sterile injection solutions which may contain anti-oxidants, buffers, bacteriostats and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which may include suspending agents and thickening agents. The formulations may be presented in unit-dose or multi-dose containers, for example, sealed ampules and vials, and may be stored in a freeze dried (lyophilized) condition requiring only the addition of the sterile liquid carrier, for example water for injections, immediately prior to use. Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules and tablets.
[147] Such injection solutions may be in the form, for example, of a sterile injectable aqueous or oleaginous suspension. This suspension may be formulated according to techniques known in the art using suitable dispersing or wetting agents (such as, for example, Tween 80) and suspending agents. The sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally-acceptable diluent or solvent, for example, as a solution in 1,3-
butanediol. Among the acceptable vehicles and solvents that may be employed are mannitol, water, Ringer's solution and isotonic sodium chloride solution. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending medium. For this purpose, any bland fixed oil may be employed including synthetic mono- or diglycerides. Fatty acids, such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions. These oil solutions or suspensions may also contain a long-chain alcohol diluent or dispersant.
[148] Exemplary embodiments of such injectable compositions include
embodiments wherein R2 is C(0)-C1-21 alkyl. In some embodiments, R2 is C(0)-C5-!9 alkyl. In some embodiments, R2 is C(0)-C9-n alkyl. In some embodiments, R2 is C(0)-Cis alkyl. In some embodiments, R2 is palmitoyl.
[149] The pharmaceutical compositions of this invention may be administered in the form of suppositories for rectal administration. These compositions can be prepared by mixing a compound of this invention with a suitable non-irritating excipient which is solid at room temperature but liquid at the rectal temperature and therefore will melt in the rectum to release the active components. Such materials include, but are not limited to, cocoa butter, beeswax and polyethylene glycols.
[150] The pharmaceutical compositions of this invention may be administered by nasal aerosol or inhalation. Such compositions are prepared according to techniques well-known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art. See, e.g.: Rabinowitz JD and Zaffaroni AC, US Patent 6,803,031, assigned to Alexza Molecular Delivery Corporation.
[151] Topical administration of the pharmaceutical compositions of this invention is especially useful when the desired treatment involves areas or organs readily accessible by topical application. For topical application topically to the skin, the pharmaceutical composition should be formulated with a suitable ointment containing the active components suspended or dissolved in a carrier. Carriers for topical administration of the compounds of this invention include, but are not limited to, mineral oil, liquid petroleum, white petroleum, propylene glycol, polyoxyethylene polyoxypropylene compound, emulsifying wax, and water. Alternatively, the pharmaceutical composition can be formulated with a suitable lotion or cream
containing the active compound suspended or dissolved in a carrier. Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2-octyldodecanol, benzyl alcohol, and water. The pharmaceutical compositions of this invention may also be topically applied to the lower intestinal tract by rectal suppository formulation or in a suitable enema formulation. Topically-transdermal patches and iontophoretic administration are also included in this invention.
[152] Application of the subject therapeutics may be local, so as to be administered at the site of interest. Various techniques can be used for providing the subject compositions at the site of interest, such as injection, use of catheters, trocars, projectiles, pluronic gel, stents, sustained drug release polymers or other device which provides for internal access.
[153] Thus, according to yet another embodiment, the compounds of this invention may be incorporated into compositions for coating an implantable medical device, such as prostheses, artificial valves, vascular grafts, stents, or catheters. Suitable coatings and the general preparation of coated implantable devices are known in the art and are exemplified in US Patents 6,099,562; 5,886,026; and 5,304,121. The coatings are typically biocompatible polymeric materials such as a hydrogel polymer, polymethyldisiloxane, polycaprolactone, polyethylene glycol, polylactic acid, ethylene vinyl acetate, and mixtures thereof. The coatings may optionally be further covered by a suitable topcoat of fluorosilicone, polysaccharides, polyethylene glycol, phospholipids or combinations thereof to impart controlled release characteristics in the composition. Coatings for invasive devices are to be included within the definition of pharmaceutically acceptable carrier, adjuvant or vehicle, as those terms are used herein.
[154] According to another embodiment, the invention provides a method of coating an implantable medical device comprising the step of contacting said device with the coating composition described above. It will be obvious to those skilled in the art that the coating of the device will occur prior to implantation into a mammal.
[155] According to another embodiment, the invention provides a method of impregnating an implantable drug release device comprising the step of contacting said drug release device with a compound or composition of this invention.
Implantable drug release devices include, but are not limited to, biodegradable
polymer capsules or bullets, non-degradable, diffusible polymer capsules and biodegradable polymer wafers.
[156] According to another embodiment, the invention provides an implantable medical device coated with a compound or a composition comprising a compound of this invention, such that said compound is therapeutically active.
[157] According to another embodiment, the invention provides an implantable drug release device impregnated with or containing a compound or a composition comprising a compound of this invention, such that said compound is released from said device and is therapeutically active.
[158] Where an organ or tissue is accessible because of removal from the subject, such organ or tissue may be bathed in a medium containing a composition of this invention, a composition of this invention may be painted onto the organ, or a composition of this invention may be applied in any other convenient way.
[159] In another embodiment, a composition of this invention further comprises one or more additional therapeutic agents. The additional therapeutic agent may be selected from any compound or therapeutic agent known to have or that demonstrates advantageous properties when administered with a compound having the same mechanism of action as ITI-007. Such agents include those indicated as being useful in combination with ITI-007, including but not limited to, those described in PCT application No. PCT/US2008/009357 (published as WO2009/017836).
[160] Preferably, the additional therapeutic agent is selected from selective serotonin re-uptake inhibitor (SSRI) (e.g. citalopram (Celexa, Cipramil, Cipram, Dalsan, Recital, Emocal, Sepram, Seropram, Citox, Cital); dapoxetine (Priligy); escitalopram (Lexapro, Cipralex, Seroplex, Esertia); fluoxetine (Depex, Prozac, Fontex, Seromex, Seronil, Sarafem, Lactose, Motivest, Flutop, Fluctin (EUR), Fluox (NZ), Depress (UZB), Lovan (AUS), Prodep (IND)); fluvoxamine (Luvox, Fevarin, Faverin, Dumyrox, Favoxil, Movox); indalpine (Upstene); paroxetine (Paxil, Seroxat, Sereupin, Aropax, Deroxat, Divarius, Rexetin, Xetanor, Paroxat, Loxamine,
Deparoc); sertraline (Zoloft, Lustral, Serlain, Asentra); vilazodone (Viibryd); or zimelidine (Zelmid, Normud)), therapeutic agents useful for the prophylaxis or treatment of dementia, particularly Alzheimer's disease: cholinesterase inhibitor (e.g., acetylcholinesterase inhibitor), such as Tacrine, rivastigmine (Exelon), donepezil (Aricept), and galantamine (Razadyne, formerly called Reminyl)), or an N-Methyl D- Asparate (NMD A) receptor antagonist, such as memantine, combination of agents
useful for the prophylaxis or treatment of Alzheimer's disease (donepezil and memantine), anti-depressant, anti-psychotic, other hypnotic agents, agents use to treat Parkinson's disease or mood disorders, compounds that modulate GABA activity (e.g. enhances the activity and facilitates GABA transmission) (e.g. doxepin, alprazolam, bromazepam, clobazam, clonazepam, clorazepate, diazepam, flunitrazepam, fiurazepam, lorazepam, midazolam, nitrazepam, oxazepam, temazapam, triazolam, indiplon, zopiclone, eszopiclone, zaleplon, Zolpidem, gabaxadol, vigabatrin, tiagabine, EVT 201 (Evotec Pharmaceuticals) and estazolam), a GABA-B agonist, a 5-HT modulator (e.g. a 5-HT2A agonist, a 5-HT^A antagonist (e.g. ketanserin, risperidone, eplivanserin, volinanserin (Sanofi-Aventis, France), pruvanserin, MDL 100907 (SanofiAventis, France), HY 10275 (Eli Lilly), APD 125 (Arena
Pharmaceuticals, San Diego, CA), or AVE8488 (Sanofi-Aventis, France)), a 5-HT2A inverse agonist, etc.), a melatonin agonist (e.g. melatonin, ramelteon (ROZEREM®, Takeda Pharmaceuticals, Japan), VEC-162 (Vanda Pharmaceuticals, Rockville, MD), PD-6735 (Phase II Discovery) or agomelatine), an ion channel modulator (e.g.
blocker) (e.g. lamotrigine, gabapentin and pregabalin), a serotonin-2
antagonist/reuptake inhibitor (SARI) (e.g. Org 50081 (Organon -Netherlands), ritanserin, nefazodone, serzone or trazodone), an orexin receptor antagonist (e.g. orexin, a 1,3-biarylurea, SB-334867-a (GlaxoSmithKline, UK), GW649868
(GlaxoSmithKline) and a benzamide derivative), an H3 agonist or antagonist, a noradrenergic agonist or antagonist, a galanin agonist, a CRH antagonist, human growth hormone, a growth hormone agonist, estrogen, an estrogen agonist, a neurokinin- 1 drug (e.g. Casopitant), an anti-depressant (e.g. amitriptyline, amoxapine, bupropion, citalopram, clomipramine, desipramine, doxepin, duloxetine, escitaloprame, fluoxetine, fluvoxamine, imipramine, isocarboxazid, maprotiline, mirtazapine, nefazodone, nortriptyline, paroxetine, phenlzine sulfate, protiptyline, sertraline, tranylcypromine, trazodone, trimipramine, and velafaxine), and an antipsychotic agent (e.g. chlorpromazine, haloperidol, droperidol, fluphenazine, loxapine, mesoridazine molidone, peφhenazine, pimozide, prochlorperazine promazine, thioridazine, thiothixene, trifluoperazine, clozapine, aripiparazole, olanzapine, quetiapine, risperidone, ziprasidone and paliperidone), an atypical antipsychotic agent (e.g. clozapine, aripiparazole, olanzapine, quetiapine, risperidone, ziprasidone, or paliperidone), 5HTLA agonist (e.g. repinotan, sarizotan, eptapirone, buspirone or MN-305), anti-Parkinson's agent (e.g. L- dopa, co-careldopa, duodopa,
stalova, Symmetrel, benzotropine, biperiden, bromocryiptine, entacapone, pergolide, pramipexole, procyclidine, ropinirole, selegiline or tolcapone), levodopa and levodopa adjuncts (carbidopa, COMT inhibitors, MAO-B inhibitors), dopamine agonists, anticholinergics (e.g. levodopa), atypical stimulant (e.g. modafinil, adrafinil, or armodafinil), antidepressant (e.g. selective serotonin reuptake inhibitors (SSRJs), serotonin-norepinephrine reuptake inhibitors (SNRJs), or tricyclic antidepressants), antihistamine agents (e.g. latrepirdine).
[161] In another embodiment, the invention provides separate dosage forms of a compound of this invention and one or more of any of the above-described additional therapeutic agents, wherein the compound and additional therapeutic agent are associated with one another. The term "associated with one another" as used herein means that the separate dosage forms are packaged together or otherwise attached to one another such that it is readily apparent that the separate dosage forms are intended to be sold and administered together (within less than 24 hours of one another, consecutively or simultaneously).
[162] In the pharmaceutical compositions of the invention, the compound of the present invention is present in an effective amount. As used herein, the term
"effective amount" refers to an amount which, when administered in a proper dosing regimen, is sufficient to treat the target disorder.
[163] The interrelationship of dosages for animals and humans (based on milligrams per meter squared of body surface) is described in Freireich et al., Cancer Chemother. Rep, 1966, 50: 219. Body surface area may be approximately determined from height and weight of the subject. See, e.g., Scientific Tables, Geigy Pharmaceuticals, Ardsley, N.Y., 1970, 537.
[164] In one embodiment, an effective amount of a compound of this invention can be 20 mg per dose. In one embodiment, an effective amount of a compound of this invention can be 30 mg per dose. In one embodiment, an effective amount of a compound of this invention can be 40 mg per dose. In one embodiment, an effective amount of a compound of this invention can be 60 mg per dose. In one embodiment, an effective amount of a compound of this invention can range from about 20 mg to about 60 mg per dose. In one embodiment, an effective amount of a compound of this invention can range from about 4 mg to about 120 mg per dose. In one embodiment, an effective amount of a compound of this invention can range from about 2 mg to about 300 mg per dose. In one embodiment, an effective amount of a compound of
this invention can range from about 0.2 mg to about 600 mg per dose. In one embodiment, the dose is administered twice daily. In one embodiment, the dose is administered once daily. In some embodiments, a dose of the injectable compositions of the present invention may be administered once a week, once every two weeks, or once a month.
[165] Effective doses will also vary, as recognized by those skilled in the art, depending on the diseases treated, the severity of the disease, the route of administration, the sex, age and general health condition of the subject, excipient usage, the possibility of co-usage with other therapeutic treatments such as use of other agents and the judgment of the treating physician. For example, guidance for selecting an effective dose can be determined by reference to the prescribing information for ITI-007.
[166] For pharmaceutical compositions that comprise one or more additional therapeutic agents, an effective amount of the additional therapeutic agent is between about 20% and 100% of the dosage normally utilized in a monotherapy regime using just that agent. Preferably, an effective amount is between about 70% and 100% of the normal monotherapeutic dose. The normal monotherapeutic dosages of these additional therapeutic agents are well known in the art. See, e.g., Wells et al., eds., Pharmacotherapy Handbook, 2nd Edition, Appleton and Lange, Stamford, Conn. (2000); PDR Pharmacopoeia, Tarascon Pocket Pharmacopoeia 2000, Deluxe Edition, Tarascon Publishing, Loma Linda, Calif. (2000), each of which references are incorporated herein by reference in their entirety.
[167] Some of the additional therapeutic agents referenced above may act synergistically with the compounds of this invention. When this occurs, it will allow the effective dosage of the additional therapeutic agent and/or the compound of this invention to be reduced from that required in a monotherapy. This has the advantage of minimizing toxic side effects of either the additional therapeutic agent of a compound of this invention, synergistically improving efficacy, improving ease of administration or use and/or reduced overall expense of compound preparation or formulation.
Methods of Treatment
[168] In another embodiment, the invention provides a method of modulating the activity of serotonin, glutamate, and dopamine receptors in a cell (in vivo or in vitro),
comprising contacting a cell with one or more compounds of Formula III herein (e.g., Formula Ilia or Illb), or a pharmaceutically acceptable salt thereof. In some embodiments, the cell is contacted in vitro. In some embodiments, the cell is contacted in vivo. In some embodiments, the cell is contacted ex vivo.
[169] In one embodiment, the invention provides a method of modulating the activity of a 5-hydroxytryptamine (5-HT) receptor. The method comprises contacting a 5-hydroxytryptamine (5-HT) receptor in a cell, in vivo or in vitro, such that activity of the 5-hydroxytryptamine (5-HT) receptor is modulated (e.g., increased or decreased). In an aspect of this embodiment, the invention provides a method of modulating the activity of 5-HT2A receptor. In a particular aspect of this embodiment, the invention provides a method of antagonizing the activity of 5-HT2A receptor.
[170] In one embodiment, the invention provides a method of modulating the activity of a serotonin transporter (SERT). The method comprises contacting a serotonin transporter in a cell, in vivo or in vitro, such that activity of the serotonin transporter is modulated (e.g., increased or decreased). In an aspect of this embodiment, the invention provides a method of inhibiting a serotonin transporter.
[171] In one embodiment, the invention provides a method of modulating the activity of a dopamine (e.g. D2) receptor. The method comprises contacting a dopamine receptor in a cell, in vivo or in vitro, such that activity of the dopamine receptor is modulated (e.g., increased or decreased). In an aspect of this embodiment, the invention provides a method of antagonizing a postsynaptic D2 receptor. In an aspect of this embodiment, the invention provides a method of partially agonizing a presynaptic striatal D2 receptor.
[172] In one embodiment, the invention provides a method of modulating the activity of GluN2B-type N-methyl-D-aspartate (NMD A) receptor. The method comprises contacting a GluN2B-type N-methyl-D-aspartate receptor in a cell, in vivo or in vitro, such that activity of the GluN2B-type N-methyl-D-aspartate receptor is modulated (e.g., increased or decreased). In an aspect of this embodiment, the invention provides a method of indirectly modulating glutamatergic
neurotransmission by increasing phosphorylation of a GluN2B-type N-methyl-D- aspartate (NMD A) receptor.
[173] According to another embodiment, the invention provides a method of treating a disease that is beneficially treated by ITI-007 in a subject in need thereof, comprising the step of administering to the subject an effective amount of a
compound or a composition of this invention. In one embodiment the subject is a patient in need of such treatment. Such diseases are well known in the art and are disclosed in, but not limited to the following patents and published applications: PCT application No. PCT/US2008/003340 (published as WO2008112280); PCT application No. PCT/US2009/001608 (published as WO2009114181); PCT application No. PCT/US2000/016373 (published as WO2000077010); PCT application No. PCT/US2014/029914 (published asWO2014145192); PCT application No. PCT/US2013/036515 (published as WO2013155506); PCT application No. PCT/US2013/036512 (published as WO2013155504);
Psychopharmacology (2015), 232(3), 605-621.
[174] Such diseases include, but are not limited to, addictive behavior (e.g. behavior associated with and or caused by physical and/or psychological dependence on narcotics, opiates, analgesics, painkillers, amphetamines, cocaine, heroin, opium, marijuana, alcohol, smoking, nicotine, gambling or eating), affective disorders, aggressive and assaultive behavior, aggressive behavior, agitation, agitation in Alzheimer's disease, agitation in autism and/or related autistic disorders, agitation in dementia (e.g. agitation in Alzheimer's disease), Alzheimer's disease, amyotrophic lateral sclerosis, anger, anorexia, anxiety, assaultive behavior, attention deficit disorder, attention deficit hyperactivity disorder, behavioral disturbances in patients with dementia (e.g. agitation and irritability), behavioral or mood disorders (e.g. agitation irritation), bipolar depression, bipolar disorder, brain spinal cord trauma, bulemia, cancer, central nervous system disorders, cephalic pain, cognition associated with schizophrenia, conditions associated with cephalic pain, corticobasal degenerations, dementia, dementia with Lewy bodies, dementing illnesses (e.g. senile dementia, Alzheimer's disease, Pick' s disease, frontotemporal dementia,
parasupranculear palsy, dementia with Lewy bodies, vascular dementia), depression (e.g. refractory depression, co-morbid depression, MDD), depression in a patient suffering from psychosis (e.g. schizophrenia), Down syndrome, dyskinesia, elderly depression, excessive sleepiness, fatigue in conditions such as Parkinson's disease, frontotemporal dementia, gastrointestinal disorders, gastrointestinal disorders (e.g. dysfunction of the gastrointestinal tract motility), Huntington's disease, impulse control disorder, impulse control disorder or intermittent explosive disorder in patients suffering from psychosis (e.g. schizophrenia), intermittent explosive disorder, irritation, major depressive disorder, migraine, mild cognition impairment, mood
disorders associated with Parkinson's disease, mood disorders associated with psychosis (e.g. schizophrenia), multiple sclerosis, neurodegenerative diseases, obesity, obsessive-compulsive disorder, other psychiatric or neurological diseases, parasupranculear palsy, Parkinson's disease, physical or emotional outbursts, Pick's disease, post-traumatic stress disorder, prion disease, psychosis, schizophrenia, senile dementia, sexual disorders, sleep disorder (e.g. maintenance insomnia, frequent awakenings, waking up feeling unrefreshed, insomnia, narcolepsy, sleep apnea), sleep disorders (particularly sleep disorders associated with schizophrenia), sleep disorders associated with Parkinson's disease, sleep disorders associated with psychosis (e.g. schizophrenia), sleep disorders in patients suffering from dementia (e.g. Alzheimer's disease), social phobias, traumatic conditions, vascular dementia, Wemicke- Korsakoffs syndrome..
[175] In some embodiments, the method of this invention is used to treat a disease or condition selected from schizophrenia, schizophrenia having an acute exacerbation of psychosis, acute exacerbation of psychosis, depression, bipolar disorder, agitation, insomnia, dementia, and Alzheimer's Disease, the method comprising the step of administering to a subject in need thereof a therapeutically effective amount of a compound or a composition of the present invention.
[176] In one particular embodiment, the method of this invention is used to treat a disease or condition selected from schizophrenia, schizophrenia having an acute exacerbation of psychosis, acute exacerbation of psychosis, and Alzheimer's Disease in a subject in need thereof.
[177] Identifying a subject in need of such treatment can be in the judgment of a subject or a health care professional and can be subjective (e.g. opinion) or objective (e.g. measurable by a test or diagnostic method).
[178] In some embodiments, the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once a week.
[179] In some embodiments, the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once every two weeks.
[180] In some embodiments, the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once every three weeks.
[181] In some embodiments, the method comprises administering to a subject an injectable composition comprising a compound disclosed herein twice a month.
[182] In some embodiments, the method comprises administering to a subject an injectable composition comprising a compound disclosed herein once a month.
[183] In exemplary embodiments of methods comprising administering to a subject such injectable compositions once a week; once every two weeks; once every three weeks; twice a month; or once a month, R2 is C(0)-C1-21 alkyl. In some exemplary embodiments, R2 is C(0)-Cs-i9 alkyl. In some exemplary embodiments, R2 is C(O)- C9-n alkyl. In some exemplary embodiments, R2 is C(0)-C15 alkyl. In some embodiments, R2 is palmitoyl.
[184] In another embodiment, any of the above methods of treatment comprises the further step of co-administering to the subject in need thereof one or more additional therapeutic agents. The choice of additional therapeutic agent may be made from any additional therapeutic agent known to be useful for co-administration with ITI-007. The choice of additional therapeutic agent is also dependent upon the particular disease or condition to be treated. Examples of additional therapeutic agents that may be employed in the methods of this invention are those set forth above for use in combination compositions comprising a compound of this invention and an additional therapeutic agent.
[185] The term "co-administered" as used herein means that the additional therapeutic agent may be administered together with a compound of this invention as part of a single dosage form (such as a composition of this invention comprising a compound of the invention and an additional therapeutic agent as described above) or as separate, multiple dosage forms. Alternatively, the additional agent may be administered prior to, consecutively with, or following the administration of a compound of this invention. In such combination therapy treatment, both the compounds of this invention and the additional therapeutic agent(s) are administered by conventional methods. The administration of a composition of this invention, comprising both a compound of the invention and an additional therapeutic agent, to a subject does not preclude the separate administration of that same therapeutic agent, any other additional therapeutic agent or any compound of this invention to said subject at another time during a course of treatment.
[186] Effective amounts of these additional therapeutic agents are well known to those skilled in the art and guidance for dosing may be found in patents and published
patent applications referenced herein, as well as in Wells et al., eds., Pharmacotherapy Handbook, 2nd Edition, Appleton and Lange, Stamford, Conn. (2000); PDR
Pharmacopoeia, Tarascon Pocket Pharmacopoeia 2000, Deluxe Edition, Tarascon Publishing, Loma Linda, Calif. (2000), and other medical texts. However, it is well within the skilled artisan's purview to determine the additional therapeutic agent's optimal effective-amount range.
[187] In one embodiment of the invention, where an additional therapeutic agent is administered to a subject, the effective amount of the compound of this invention is less than its effective amount would be where the additional therapeutic agent is not administered. In another embodiment, the effective amount of the additional therapeutic agent is less than its effective amount would be where the compound of this invention is not administered. In this way, undesired side effects associated with high doses of either agent may be minimized. Other potential advantages (including without limitation improved dosing regimens and/or reduced drug cost) will be apparent to those of skill in the art.
[188] In yet another aspect, the invention provides the use of a compound of Formula III alone or together with one or more of the above-described additional therapeutic agents in the manufacture of a medicament, either as a single composition or as separate dosage forms, for treatment in a subject of a disease, disorder or symptom set forth above. Another aspect of the invention is a compound of Formula III for use in the treatment in a subject of a disease, disorder or symptom thereof delineated herein.
Example X. Evaluation of Metabolic Stability
[189] Microsomal Assay: Human liver microsomes (20 mg/mL) are obtained from Xenotech, LLC (Lenexa, KS). β-nicotinamide adenine dinucleotide phosphate, reduced form (NADPH), magnesium chloride (MgCh), and dimethyl sulfoxide (DMSO) are purchased from Sigma- Aldrich.
[190] Determination of Metabolic Stability: 7.5 mM stock solutions of test compounds are prepared in DMSO. The 7.5 mM stock solutions are diluted to 12.5- 50 μΜ in acetonitrile (ACN). The 20 mg/mL human liver microsomes are diluted to 0.625 mg/mL in 0.1 M potassium phosphate buffer, pH 7.4, containing 3 mM MgCh. The diluted microsomes are added to wells of a 96-well deep-well polypropylene plate in triplicate. A 10 μί, aliquot of the 12.5-50 μΜ test compound is added to the
microsomes and the mixture is pre- warmed for 10 minutes. Reactions are initiated by addition of pre- warmed NADPH solution. The final reaction volume is 0.5 mL and contains 0.5 mg/mL human liver microsomes, 0.25-1.0 μΜ test compound, and 2 mM NADPH in 0.1 M potassium phosphate buffer, pH 7.4, and 3 mM MgCl2. The reaction mixtures are incubated at 37 °C, and 50 μί, aliquots are removed at 0, 5, 10, 20, and 30 minutes and added to shallow- well 96- well plates which contain 50 μί, of ice-cold ACN with internal standard to stop the reactions. The plates are stored at 4 °C for 20 minutes after which 100 μί, of water is added to the wells of the plate before centrifugation to pellet precipitated proteins. Supernatants are transferred to another 96-well plate and analyzed for amounts of parent remaining by LC-MS/MS using an Applied Bio-systems API 4000 mass spectrometer. The same procedure is followed for the non-deuterated counterpart of the compound of Formula III and the positive control, 7-ethoxycoumarin (1 μΜ). Testing is done in triplicate.
[191] Data analysis: The in vitro t s for test compounds are calculated from the slopes of the linear regression of % parent remaining (In) vs incubation time relationship,
in vitro t ½ = 0.693/k
k = -[slope of linear regression of % parent remaining (In) vs incubation time]
[192] Data analysis is performed using Microsoft Excel Software.
[193] The relevant teachings of all patents, published applications and references cited herein are incorporated by reference in their entirety.
[194] Without further description, it is believed that one of ordinary skill in the art can, using the preceding description and the illustrative examples, make and utilize the compounds of the present invention and practice the claimed methods. It should be understood that the foregoing discussion and examples merely present a detailed description of certain preferred embodiments. It will be apparent to those of ordinary skill in the art that various modifications and equivalents can be made without departing from the spirit and scope of the invention.
Claims
1. A compound of Formula III :
or a pharmaceutically acceptable salt thereof,
wherein R is CHs, CH2D, CHD2 or CDs;
R2 is hydrogen, deuterium, or C(0)-C1-21 alkyl;
Y8a, Y8b, and Y14 are each deuterium;
Y11a Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen;
and Y1a, Y1b, Y2a, Y2b, Y3a, Y3b, Y4a, Y4b, Y5a, Ya, Y6a, Y6b, Y7a, Y7b, Y9, and Y10 are independently selected from hydrogen and deuterium.
2. The compound of claim 1, wherein the compound is a compound of Formula
Ilia:
or a pharmaceutically acceptable salt thereof.
3. The compound of claim 1, wherein the compound is a compound of Formula Illb:
harmaceutically acceptable salt thereof.
4. The compound of any one of claims 1, 2 or 3, wherein R is CH3.
5. The compound of any one of claims 1, 2 or 3, wherein R is CD3.
6. The compound of any one of the preceding claims, wherein Y1a and Y1b are the same.
7. The compound of any one of the preceding claims, wherein Y2a and Y2b are the same.
8. The compound of any one of the preceding claims, wherein Y3a and Y3b are the same.
9. The compound of any one of the preceding claims, wherein Y4a and Y4b are the same.
10. The compound of any one of the preceding claims, wherein Y5a and Y5b are the same.
11. The compound of any one of the preceding claims, wherein Y6a and Y6b are the same.
12. The compound of claim 11, wherein Y6a and Y6b are each hydrogen.
13. The compound of claim 11, wherein Y6a and Y6b are each deuterium.
14. The compound of any one of the preceding claims, wherein Y7a and Y711 are the same.
15. The compound of claim 14, wherein Y7a and Y711 are each hydrogen.
16. The compound of claim 14, wherein Y7a and Y711 are each deuterium.
17. The compound of any one of the preceding claims, wherein Y9 and Y10 are
each hydrogen.
18. The compound of any one of the preceding claims, wherein Y9 and Y10 are each deuterium.
19. The compound of any one of the preceding claims, wherein R2 is hydrogen.
20. The compound of any one of claims 1 to 18, wherein R2 is deuterium.
21. The compound of any one of claims 1 to 18, wherein R2 is C(0)-C1-21 alkyl.
22. The compound of any one of claims 1 to 18, wherein R2 is C(0)-Cs-i9 alkyl.
23. The compound of any one of claims 1 to 18, wherein R2 is palmitoyl.
24. The compound of claim 1, wherein R is CD3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y715 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table la (below):
Table la: Exemplary Embodiments of Formula III
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
25. The compound of claim 1, wherein R is CH3; R2 is hydrogen; Y1a and Y1b are the same; Y2a and Y2b are the same; Y3a and Y3b are the same; Y4a and Y4b are the same; Y5a and Y5b are the same; Y6a and Y6b are the same; Y7a and Y711 are the same; Y9 is hydrogen; Y10 is hydrogen; Y11a, Y11b and Y11c are each hydrogen; Y12a and Y12b are each hydrogen; Y13a and Y13b are each hydrogen; and the compound of Formula III is selected from any one of the compounds (Cmpd) set forth in Table lb (below):
Table lb: Exemplary Embodiments of Formula III
, or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
26. The compound of any of the claims 1-25, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
27. A pharmaceutical composition comprising the compound of claim 1 or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier.
28. The composition of claim 27, wherein the composition is an injectable composition.
29. The composition of claim 28, wherein R2 is C(0)-C1-21 alkyl.
30. The composition of claim 28, wherein R2 is C(0)-Cs-i9 alkyl.
31. The composition of claim 28, wherein R2 is palmitoyl.
32. A method of modulating activity of a 5-hydroxytryptamine (5-HT) receptor in a cell, comprising contacting the cell with the compound of claim 1 or the composition of claim 27.
33. The method of claim 32, comprising antagonizing the activity of the 5-HT2A receptor.
34. A method of modulating activity of a serotonin transporter (SERT) in a cell, comprising contacting the cell with the compound of claim 1 or the composition of claim 27.
35. A method of modulating activity of a dopamine (D2) receptor in a cell, comprising contacting the cell with the compound of claim 1 or the composition of claim 27.
36. The method of claim 35, wherein the method of modulating a dopamine (D2) receptor comprises antagonizing a postsynaptic dopamine (D2) receptor.
37. The method of claim 35, comprising agonizing a presynaptic striatal dopamine (D2) receptor.
38. A method of modulating activity of a GluN2B-type N-methyl-D-aspartate (NMD A) receptor in a cell, comprising contacting the cell with the compound of claim 1 or the composition of claim 27.
39. The method of claim 38, comprising indirectly modulating glutamatergic neurotransmission by increasing phosphorylation of the GluN2B-type N-methyl-D- aspartate (NMD A) receptor.
40. A method of treating a disease or condition selected from schizophrenia, schizophrenia having an acute exacerbation of psychosis, acute exacerbation of psychosis, depression, bipolar disorder, agitation, insomnia, dementia, and
Alzheimer's Disease, comprising the step of administering to a subject in need thereof
a therapeutically effective amount of the compound of claim 1 or the composition of claim 27.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201662431282P | 2016-12-07 | 2016-12-07 | |
| US62/431,282 | 2016-12-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018106916A1 true WO2018106916A1 (en) | 2018-06-14 |
Family
ID=62492325
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2017/065111 Ceased WO2018106916A1 (en) | 2016-12-07 | 2017-12-07 | Deuterated quinoxaline compounds |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2018106916A1 (en) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3407889A4 (en) * | 2016-03-25 | 2019-03-13 | Intra-Cellular Therapies, Inc. | ORGANIC COMPOUNDS |
| US10597394B2 (en) | 2014-04-04 | 2020-03-24 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US10716786B2 (en) | 2017-03-24 | 2020-07-21 | Intra-Cellular Therapies, Inc. | Transmucosal and subcutaneous compositions |
| US10844061B2 (en) | 2013-03-15 | 2020-11-24 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-(6BR,10AS)-3-methyl-2,3,6B,9,10,10A-hexahydro-1h, 7H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8-yl)-1-(4-fluorophenyl)butan-1-one and methods of treating conditions of the central nervous system |
| US10960010B2 (en) | 2013-12-03 | 2021-03-30 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions for sustained or delayed release |
| US11052084B2 (en) | 2018-08-31 | 2021-07-06 | Intra-Cellular Therapies, Inc. | Pharmaceutical capsule compositions comprising lumateperone mono-tosylate |
| US11331316B2 (en) | 2016-10-12 | 2022-05-17 | Intra-Cellular Therapies, Inc. | Amorphous solid dispersions |
| US11957791B2 (en) | 2018-08-31 | 2024-04-16 | Intra-Cellular Therapies, Inc. | Methods |
| US12122792B2 (en) | 2019-12-11 | 2024-10-22 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-((6bR,10aS)-3-methyl2,3,6b,9,10,10a-hexahydro-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin8(7H)-yl)-1-(4-((6bR,10aS)-3-methyl-2,3,6b,9,10,10a-hexahydro-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl)phenyl)butan-1-one for treating conditions of the central nervous system and cardiac disorders |
| US12144808B2 (en) | 2018-08-29 | 2024-11-19 | Intra-Cellular Therapies, Inc. | Compositions and methods |
| US12195464B2 (en) | 2018-06-06 | 2025-01-14 | Intra-Cellular Therapies, Inc. | Lumateperone bis-tosylate salts and crystals and methods for manufacture thereof |
| WO2025128666A1 (en) * | 2023-12-11 | 2025-06-19 | Bioxcel Therapeutics, Inc. | Combination of latrepirdine and a pharmacokinetic stabilizer |
| US12384783B2 (en) | 2017-09-26 | 2025-08-12 | Intra-Cellular Therapies, Inc. | Salts and crystals |
| US12414948B2 (en) | 2022-05-18 | 2025-09-16 | Intra-Cellular Therapies, Inc. | Methods |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5902815A (en) * | 1996-09-03 | 1999-05-11 | Washington University | Use of 5HT-2A serotonin agonists to prevent adverse effects of NMDA receptor hypofunction |
| US20040034015A1 (en) * | 1999-06-15 | 2004-02-19 | Robichaud Albert J. | Substituted heterocycle fused gamma-carbolines |
| US20130202692A1 (en) * | 2010-04-22 | 2013-08-08 | Sharon Mates | Organic compounds |
| WO2015154025A1 (en) * | 2014-04-04 | 2015-10-08 | Intra-Cellular Therapies, Inc. | Organic compounds |
| WO2015154030A1 (en) * | 2014-04-04 | 2015-10-08 | Intra-Cellular Therapies, Inc. | Organic compounds |
-
2017
- 2017-12-07 WO PCT/US2017/065111 patent/WO2018106916A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5902815A (en) * | 1996-09-03 | 1999-05-11 | Washington University | Use of 5HT-2A serotonin agonists to prevent adverse effects of NMDA receptor hypofunction |
| US20040034015A1 (en) * | 1999-06-15 | 2004-02-19 | Robichaud Albert J. | Substituted heterocycle fused gamma-carbolines |
| US20130202692A1 (en) * | 2010-04-22 | 2013-08-08 | Sharon Mates | Organic compounds |
| WO2015154025A1 (en) * | 2014-04-04 | 2015-10-08 | Intra-Cellular Therapies, Inc. | Organic compounds |
| WO2015154030A1 (en) * | 2014-04-04 | 2015-10-08 | Intra-Cellular Therapies, Inc. | Organic compounds |
Cited By (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10844061B2 (en) | 2013-03-15 | 2020-11-24 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-(6BR,10AS)-3-methyl-2,3,6B,9,10,10A-hexahydro-1h, 7H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8-yl)-1-(4-fluorophenyl)butan-1-one and methods of treating conditions of the central nervous system |
| US12240850B2 (en) | 2013-03-15 | 2025-03-04 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-((6bR, 10aS)-3-methyl-2,3,6b,9,10, 10a-hexahydro-1H,7H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8-yl)-1-(4-fluorophenyl)butan-1-ol for treating central nervous system disorders |
| US11680065B2 (en) | 2013-03-15 | 2023-06-20 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-(6Br,10aS)-3-methyl-2, 3, 6b, 9, 10, 10a-hexahydro-1H, 7H-pyrido[3′, 4′, 5] pyrolo[1,2,3-de] quinoxalin-8YL)-1-(4-fluorophenyl)-butane-1-one and methods of treating conditions of the central nervous system |
| US12264160B2 (en) | 2013-03-15 | 2025-04-01 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-((6bR, 10aS)-3-methyl-2,3,6b,9,10,10a-hexahydro-1H,7H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8-yl)-1-(4-fluorophenyl)butan-1-one for treating central nervous system disorders |
| US11026951B2 (en) | 2013-12-03 | 2021-06-08 | Intra-Cellular Therapies, Inc. | Methods of treating bipolar disorder |
| US10960010B2 (en) | 2013-12-03 | 2021-03-30 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions for sustained or delayed release |
| US10960009B2 (en) | 2013-12-03 | 2021-03-30 | Intra-Cellular Therapies, Inc. | Methods of treating schizophrenia and depression |
| US10899762B2 (en) | 2014-04-04 | 2021-01-26 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US10597394B2 (en) | 2014-04-04 | 2020-03-24 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US11560382B2 (en) | 2014-04-04 | 2023-01-24 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US12269825B2 (en) | 2014-04-04 | 2025-04-08 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US11096944B2 (en) | 2016-03-25 | 2021-08-24 | Intra-Cellular Therapies, Inc. | Organic compounds |
| EP3888656A1 (en) * | 2016-03-25 | 2021-10-06 | Intra-Cellular Therapies, Inc. | Deuterated heterocyclic gamma-carboline compounds and their use in the treatment or prophylaxis of a central nervous system disorder |
| EP3407889A4 (en) * | 2016-03-25 | 2019-03-13 | Intra-Cellular Therapies, Inc. | ORGANIC COMPOUNDS |
| US10688097B2 (en) | 2016-03-25 | 2020-06-23 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US11723909B2 (en) | 2016-03-25 | 2023-08-15 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US12194044B2 (en) | 2016-03-25 | 2025-01-14 | 1Ntra-Cellular Therap1Es, 1Nc. | Organic compounds |
| US11331316B2 (en) | 2016-10-12 | 2022-05-17 | Intra-Cellular Therapies, Inc. | Amorphous solid dispersions |
| US11826367B2 (en) | 2016-10-12 | 2023-11-28 | Intra-Cellular Therapies, Inc. | Amorphous solid dispersions |
| US11052083B2 (en) | 2017-03-24 | 2021-07-06 | Intra-Cellular Therapies, Inc. | Transmucosal methods for treating psychiatric and neurological conditions |
| US12336989B2 (en) | 2017-03-24 | 2025-06-24 | Intra-Cellular Therapies, Inc. | Transmucosal methods for treating psychiatric and neurological conditions |
| US10716786B2 (en) | 2017-03-24 | 2020-07-21 | Intra-Cellular Therapies, Inc. | Transmucosal and subcutaneous compositions |
| US12268686B2 (en) | 2017-03-24 | 2025-04-08 | Intra-Cellular Therapies, Inc. | Transmucosal methods for treating psychiatric and neurological conditions |
| US11806347B2 (en) | 2017-03-24 | 2023-11-07 | Intra-Cellular Therapies, Inc. | Transmucosal methods for treating psychiatric and neurological conditions |
| US12384783B2 (en) | 2017-09-26 | 2025-08-12 | Intra-Cellular Therapies, Inc. | Salts and crystals |
| US12195464B2 (en) | 2018-06-06 | 2025-01-14 | Intra-Cellular Therapies, Inc. | Lumateperone bis-tosylate salts and crystals and methods for manufacture thereof |
| US12144808B2 (en) | 2018-08-29 | 2024-11-19 | Intra-Cellular Therapies, Inc. | Compositions and methods |
| US11806348B2 (en) | 2018-08-31 | 2023-11-07 | Intra-Cellular Therapies, Inc. | Methods of treatment using pharmaceutical capsule compositions comprising lumateperone mono-tosylate |
| US11052084B2 (en) | 2018-08-31 | 2021-07-06 | Intra-Cellular Therapies, Inc. | Pharmaceutical capsule compositions comprising lumateperone mono-tosylate |
| US11957791B2 (en) | 2018-08-31 | 2024-04-16 | Intra-Cellular Therapies, Inc. | Methods |
| US12128043B2 (en) | 2018-08-31 | 2024-10-29 | Intra-Cellular Therapies, Inc. | Pharmaceutical capsules comprising lumateperone mono-tosylate |
| US12070459B2 (en) | 2018-08-31 | 2024-08-27 | Intra-Cellular Therapies, Inc. | Pharmaceutical capsule compositions comprising lumateperone mono-tosylate |
| US12465570B2 (en) | 2018-08-31 | 2025-11-11 | Intra-Cellular Therapies, Inc. | Methods |
| US12533355B2 (en) | 2018-08-31 | 2026-01-27 | Intra-Cellular Therapies, Inc. | Methods of treatment comprising administering lumateperone mono-tosylate capsules |
| US12122792B2 (en) | 2019-12-11 | 2024-10-22 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-((6bR,10aS)-3-methyl2,3,6b,9,10,10a-hexahydro-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin8(7H)-yl)-1-(4-((6bR,10aS)-3-methyl-2,3,6b,9,10,10a-hexahydro-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl)phenyl)butan-1-one for treating conditions of the central nervous system and cardiac disorders |
| US12410195B2 (en) | 2019-12-11 | 2025-09-09 | Intra-Cellular Therapies, Inc. | Pharmaceutical compositions comprising 4-((6bR,10aS)-3-methyl-2,3,6b,9,10,10a-hexahydro-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl)-1-(4-((6bR,10aS)-3-methyl-2,3,6b,9,10,10a-hexahydro-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl)phenyl)butan-1-one for treating conditions of the central nervous system and cardiac disorders |
| US12414948B2 (en) | 2022-05-18 | 2025-09-16 | Intra-Cellular Therapies, Inc. | Methods |
| WO2025128666A1 (en) * | 2023-12-11 | 2025-06-19 | Bioxcel Therapeutics, Inc. | Combination of latrepirdine and a pharmacokinetic stabilizer |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2018106916A1 (en) | Deuterated quinoxaline compounds | |
| WO2017117514A1 (en) | Deuterated iti-007 | |
| AU2013296627B2 (en) | Deuterated ibrutinib | |
| AU2014235462B2 (en) | Deuterated palbociclib | |
| EP2935251A1 (en) | Deuterated alk inhibitors | |
| CA2897814C (en) | Deuterated momelotinib | |
| WO2012151361A1 (en) | Carbamoylpyridone derivatives | |
| EP2872159A2 (en) | Deuterated carfilzomib | |
| WO2017136375A1 (en) | Deuterated tozadenant | |
| WO2018005328A1 (en) | Deuterated bictegravir | |
| US9676790B2 (en) | Substituted thienotriazolodiazapines | |
| AU2014237569B2 (en) | Inhibitors of the enzyme UDP-glucose: N-acyl-sphingosine glucosyltransferase | |
| WO2012129381A1 (en) | Deuterated preladenant | |
| CA2904148A1 (en) | Deuterated pacritinib | |
| WO2010068480A1 (en) | Deuterated derivatives of dimeboline | |
| WO2015009889A1 (en) | Deuterated intedanib derivatives and their use for the treatment of proliferative disorders | |
| WO2014081816A1 (en) | Fluoro-derivatives of pyrazole-substituted amino-heteroaryl compounds | |
| WO2020092604A1 (en) | Deuterated pitolisant | |
| WO2018013686A1 (en) | Deuterated idalopirdine | |
| WO2014150044A1 (en) | Amine reuptake inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17877517 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 17877517 Country of ref document: EP Kind code of ref document: A1 |