WO2017054524A1 - Nematicide of trans structure containing lactone ring and preparation method and use thereof - Google Patents
Nematicide of trans structure containing lactone ring and preparation method and use thereof Download PDFInfo
- Publication number
- WO2017054524A1 WO2017054524A1 PCT/CN2016/086427 CN2016086427W WO2017054524A1 WO 2017054524 A1 WO2017054524 A1 WO 2017054524A1 CN 2016086427 W CN2016086427 W CN 2016086427W WO 2017054524 A1 WO2017054524 A1 WO 2017054524A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- low toxicity
- carbon atoms
- hydrogen
- group
- nematicide
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *c1c(*)c(*)c(C(C(C2)C(O)=O)OC2=O)c(*)c1* Chemical compound *c1c(*)c(*)c(C(C(C2)C(O)=O)OC2=O)c(*)c1* 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/08—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings with oxygen as the ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/26—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D307/30—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D307/32—Oxygen atoms
- C07D307/33—Oxygen atoms in position 2, the oxygen atom being in its keto or unsubstituted enol form
Definitions
- the invention relates to the field of agrochemical and medical technology, in particular to a transesterification agent containing a lactone ring having a trans structure, a preparation method thereof and use thereof.
- nematodes live in the soil, some parasitic in plants, spread through soil or seeds, can destroy the roots of plants, or invade the organs of the aboveground parts, affect the growth and development of crops, and indirectly spread the diseases caused by other microorganisms, resulting in agriculture. A big economic loss.
- the existing nematicides are toxic by the penetration of the nematode epidermis.
- nematicides and fungicides there are few nematicidal agents with good effects in the world.
- There are only ten kinds of nematicides and because the existing nematicides have better toxicity to humans and animals, Some varieties have phytotoxicity to crops and affect their use, so new, efficient and environmentally friendly nematicides are urgently needed.
- the object of the present invention is to provide a transesterification agent containing a lactone ring, a preparation method and use thereof, and a lactone ring-containing nematicide agent of the trans structure of the present invention, in view of the problems in the prior art. It has good insecticidal activity against nematodes and is less toxic to humans and animals.
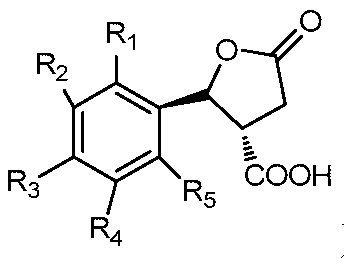
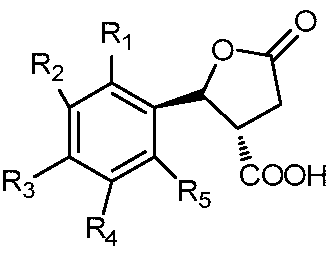
- a trans-structured nematicidal agent containing a lactone ring the structural formula I of which is as follows:
- R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms.
- R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine.
- R 6 is a fluorine atom.
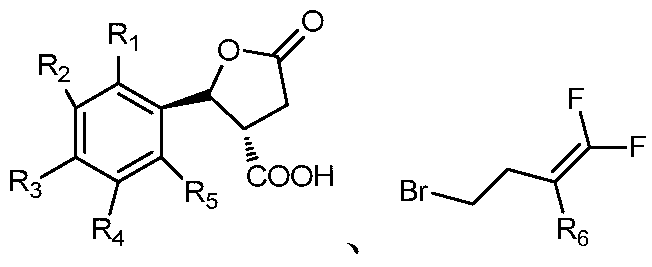
- the invention also provides a preparation method of a lactone ring-containing nematicidal agent having a trans structure, comprising the following steps:
- step 2 will And the acid of the trans structure obtained in step 1. Adding to the solvent, adding an acid binding agent, stirring the reaction at 20 to 30 ° C for 22 to 26 hours, distilling off the solvent under a vacuum of 0.08 to 0.10 kPa, adding methylene chloride and water, stirring uniformly, and standing to separate the layer to remove water. Distilling off methylene chloride at a vacuum of 0.08 to 0.10 kPa to obtain a lactone ring-containing nematicide of the trans structure represented by Formula I; The molar ratio to the acid binding agent is 1:0.8 to 1.2:3 to 5;
- R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms.
- R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine;
- the solvent is one of methanol, ethanol, acetone or N,N-dimethylformamide
- the acid binding agent is one of potassium carbonate, sodium carbonate, pyridine or triethylamine.
- the invention also provides the use of a lactone ring-containing nematicide having a trans structure for controlling agricultural nematode diseases.
- the lactone ring-containing nematicide of the trans structure of the present invention has a good control effect on the eggs of the root knot nematode and the second instar larvae due to the structure containing the polyfluorobutene and the lactone ring, especially good.
- Inhibiting the hatching of root-knot nematode eggs such as cucumber, tomato, tobacco, soybean, etc.; and the lactone ring-containing nematicide of the trans structure of the present invention is less toxic, has less residue in crops, and is suitable for humans and animals. The harm is small, which solves the problem that the existing nematicide is more toxic, and the residue in the crop is more, which improves the safety of agricultural production; the use of pests in a long time does not kill the invention.
- the nematode produces antibody and has good insecticidal effect; the invention also provides a preparation method of the transester structure containing a lactone ring-containing nematicide, the preparation method has few steps, the process is easy to operate, and is suitable for large-scale industry. produce.
- a trans-structured nematicidal agent containing a lactone ring the structural formula I of which is as follows:
- R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms.
- R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine.
- the nematicide containing an anti-lactone ring structure of formula, R 6 is a fluorine atom.
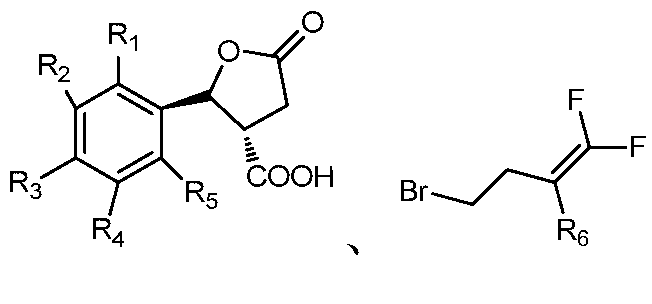
- the invention also provides a preparation method of a lactone ring-containing nematicidal agent having a trans structure, comprising the following steps:
- step 2 will And the acid of the trans structure obtained in step 1. Adding to the solvent, adding an acid binding agent, stirring the reaction at 20 to 30 ° C for 22 to 26 hours, distilling off the solvent under a vacuum of 0.08 to 0.10 kPa, adding methylene chloride and water, stirring uniformly, and standing to separate the layer to remove water. Distilling off methylene chloride at a vacuum of 0.08 to 0.10 kPa to obtain a lactone ring-containing nematicide of the trans structure represented by Formula I; The molar ratio to the acid binding agent is 1:0.8 to 1.2:3 to 5;
- R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms.
- R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine;
- the solvent is one of methanol, ethanol, acetone or N,N-dimethylformamide
- the acid binding agent is one of potassium carbonate, sodium carbonate, pyridine or triethylamine.
- the invention also provides the use of a lactone ring-containing nematicide having a trans structure for controlling agricultural nematode diseases.
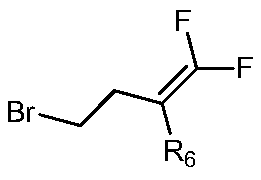
- Raw materials in the preparation method of the invention Can be purchased on the market or prepared by the following steps: And anhydrous zinc chloride is added to dichloromethane in turn, triethylamine is added dropwise at 0 to 5 ° C, the reaction solution is obtained after the dropwise addition, and the reaction solution is stirred at 20 to 30 ° C for 10 to 20 hours. Add hydrochloric acid to adjust the pH of the reaction solution to 2, extract with ethyl acetate, collect the organic phase, and recrystallize by adding toluene. among them The molar ratio of anhydrous zinc chloride to triethylamine is from 1:0.8 to 1.2:1 to 3:3 to 5; the reaction formula is:
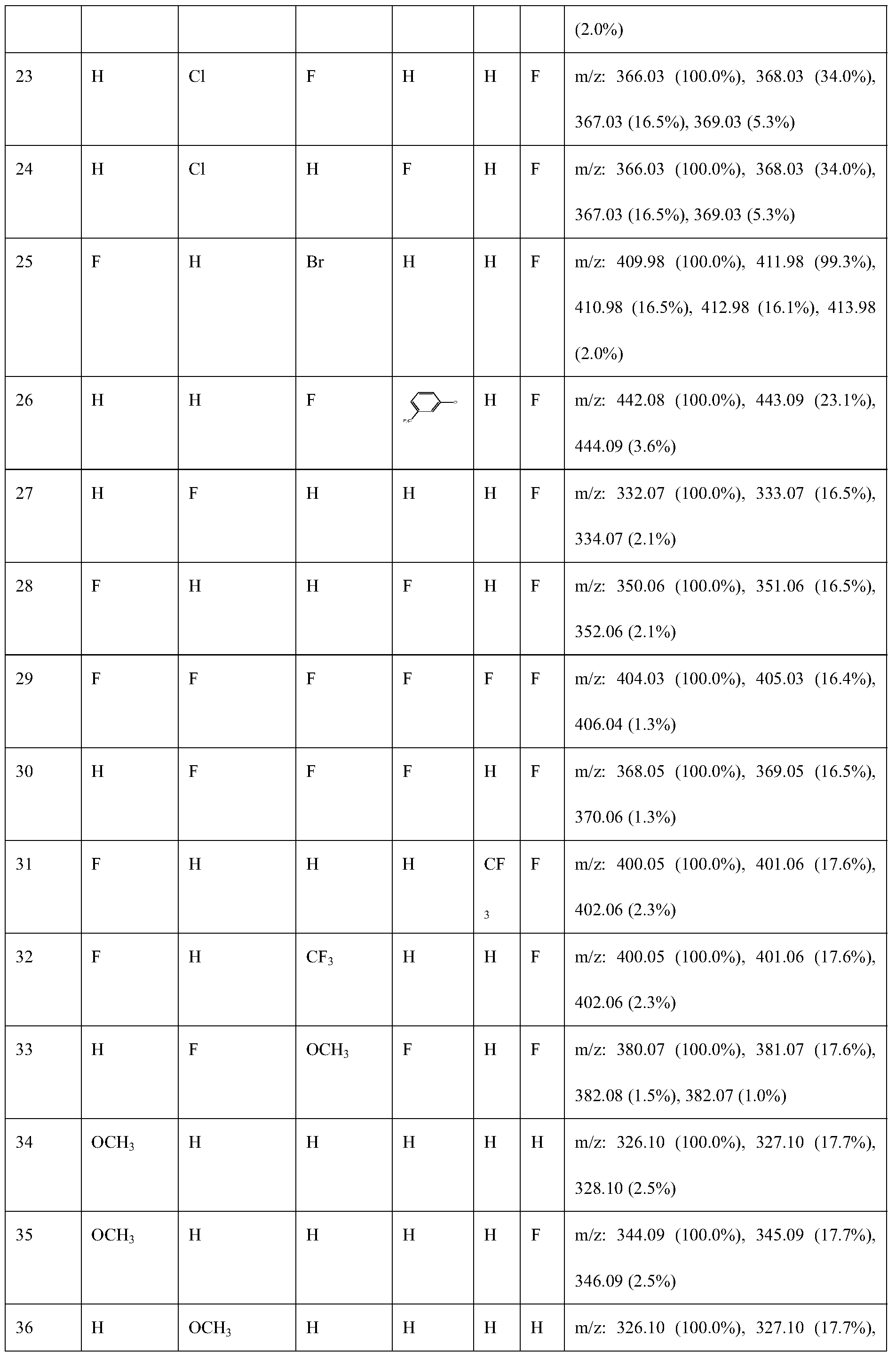
- Table 1 Structural measurement analysis table of a trans-seed nematicide-containing compound of trans structure
- a lactone ring-containing nematicide of the trans structure of the present invention can be obtained according to the preparation method of the present invention.
- the following are specific examples of the compounds in Table 1:
- the preparation of the compound of No. 2 in Table 1 comprises the following steps:
- the preparation of the compound of No. 26 in Table 1 comprises the following steps:
- the preparation of the compound of No. 41 in Table 1 includes the following steps:
- step 2 The 5-oxo-2-(2-(trifluoromethoxy)phenyl)tetrahydrofuran-3-carboxylic acid obtained in step 1 was added to a 50% sulfuric acid solution, stirred at 70 ° C for 5 h, filtered, and the filter cake was added. The organic phase is extracted with ethyl acetate and water, and the organic phase is dried over magnesium sulfate. The solvent is evaporated, and the remaining organics are recrystallized from toluene, and dried to give 5-oxy-2-(2-(trifluoro) Oxy)phenyl)tetrahydrofuran-3-carboxylic acid.
- the preparation of the compound of No. 46 in Table 1 includes the following steps:
- the preparation of the compound of No. 48 in Table 1 comprises the following steps:
- the lactone ring-containing nematicidal compound of the trans structure of the present invention has a good control effect on the second instar larvae and eggs of the nematode, and the hatching inhibition rate of the nematode eggs is higher than that. Good effect on second instar larvae.
- the cockroach toxicity test and the soil microbial toxicity test of the compounds 1 to 60 in Table 1 of the present invention are carried out according to the steps of the cockroach toxicity test and the soil microbial toxicity test in the chemical pesticide environmental safety evaluation test standard, wherein the commonly used pesticide amount in the soil microbial test is simulated. At 40 ppm, the results of the two tests are as follows:
- Compound 29 >10, low toxicity Less than 50%, low toxicity Compound 30 >10, low toxicity Less than 50%, low toxicity Compound 31 >10, low toxicity Less than 50%, low toxicity Compound 32 >10, low toxicity Less than 50%, low toxicity Compound 33 >10, low toxicity Less than 50%, low toxicity Compound 34 >10, low toxicity Less than 50%, low toxicity Compound 35 >10, low toxicity Less than 50%, low toxicity Compound 36 >10, low toxicity Less than 50%, low toxicity Compound 37 >10, low toxicity Less than 50%, low toxicity Compound 38 >10, low toxicity Less than 50%, low toxicity Compound 39 >10, low toxicity Less than 50%, low toxicity Compound 40 >10, low toxicity Less than 50%, low toxicity Compound 41 >10, low toxicity Less than 50%, low toxicity Compound 42 >10, low toxicity Less than 50%, low toxicity Compound 43 >10, low toxicity Less than 50%, low
- the transestericide-containing nematicidal compound of the present invention has low toxicity to organisms in the soil environment, is safe to use, and is an environmentally friendly compound.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
本发明涉及农化与医药技术领域,具体涉及一种反式结构的含内酯环的杀线虫剂及其制备方法和用途。The invention relates to the field of agrochemical and medical technology, in particular to a transesterification agent containing a lactone ring having a trans structure, a preparation method thereof and use thereof.
线虫大多生活在土壤中,有的寄生在植物体内,通过土壤或种子传播,能破坏植物的根系,或侵入地上部分的器官,影响农作物的生长发育,并且间接传播其他微生物引起的病害,造成农业上很大的经济损失。现有的杀线虫剂是通过线虫表皮透入起毒杀作用。Most of the nematodes live in the soil, some parasitic in plants, spread through soil or seeds, can destroy the roots of plants, or invade the organs of the aboveground parts, affect the growth and development of crops, and indirectly spread the diseases caused by other microorganisms, resulting in agriculture. A big economic loss. The existing nematicides are toxic by the penetration of the nematode epidermis.
和杀线虫剂、杀菌剂相比,目前世界上专用的效果比较好的杀线虫剂很少,仅有十余种,并且由于现有的效果较好的杀线虫剂对人畜的毒性较高,有些品种对作物有药害,影响其使用,所以新型、高效、对环境友好的杀线虫剂亟待开发。Compared with nematicides and fungicides, there are few nematicidal agents with good effects in the world. There are only ten kinds of nematicides, and because the existing nematicides have better toxicity to humans and animals, Some varieties have phytotoxicity to crops and affect their use, so new, efficient and environmentally friendly nematicides are urgently needed.
发明内容Summary of the invention
本发明的目的是针对现有技术中存在的问题,提供一种反式结构的含内酯环的杀线虫剂及其制备方法和用途,本发明反式结构的含内酯环的杀线虫剂对线虫有良好的杀虫活性,并且对人畜毒性小。The object of the present invention is to provide a transesterification agent containing a lactone ring, a preparation method and use thereof, and a lactone ring-containing nematicide agent of the trans structure of the present invention, in view of the problems in the prior art. It has good insecticidal activity against nematodes and is less toxic to humans and animals.
本发明为实现以上目的,所采取的技术方案是:In order to achieve the above objects, the technical solution adopted by the present invention is:
一种反式结构的含内酯环的杀线虫剂,其结构通式I如下所示:A trans-structured nematicidal agent containing a lactone ring, the structural formula I of which is as follows:
结构通式I, Structural formula I,
其中,R1、R2、R3、R4、R5选自氢、氰基、氟、氯、溴、含1~4个碳原子的烷基、含1~4个碳原子的烷氧基、烷氧苯基、含1~4个碳原子并且碳原子上氢原子被一个或多个氯原子取代的烷氧基、含1~4个碳原子并且碳原子上氢原子被一个或多个氟原子取代的烷氧基、硝基或胺基;Wherein R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms. Alkoxyphenyl, an alkoxy group having from 1 to 4 carbon atoms and having a hydrogen atom on the carbon atom substituted by one or more chlorine atoms, having from 1 to 4 carbon atoms and having one or more hydrogen atoms on the carbon atom a fluorine atom-substituted alkoxy group, a nitro group or an amine group;
R6选自氢、氟、氯。R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine.
优选的,一种反式结构的含内酯环的杀线虫剂,R6为氟原子。Preferably, a lactone ring-containing nematicide of trans structure, R 6 is a fluorine atom.
优选的,一种反式结构的含内酯环的杀线虫剂,R1=CF3时,R2=R3=R4=R5=H。 Preferably, a lactone ring-containing nematicide of trans structure, when R 1 =CF 3 , R 2 =R 3 =R 4 =R 5 =H.
优选的,一种反式结构的含内酯环的杀线虫剂,R1=OCF3时,R2=R3=R4=R5=H。Preferably, a lactone ring-containing nematicide of trans structure, when R 1 =OCF 3 , R 2 =R 3 =R 4 =R 5 =H.
优选的,一种反式结构的含内酯环的杀线虫剂,R1=R2=R5=H,R3=F时,R4=-O-C6H5。Preferably, a lactone ring-containing nematicide having a trans structure, R 1 = R 2 = R 5 = H, and when R 3 = F, R 4 = -OC 6 H 5 .
优选的,一种反式结构的含内酯环的杀线虫剂,R2=R4=CF3时,R1=R3=R5=H。本发明还提供了反式结构的含内酯环的杀线虫剂的制备方法,包括以下步骤:Preferably, a lactone ring-containing nematicide of trans structure, when R 2 =R 4 =CF 3 , R 1 =R 3 =R 5 =H. The invention also provides a preparation method of a lactone ring-containing nematicidal agent having a trans structure, comprising the following steps:
①将具有顺反式外消旋的酸加入到体积分数为40~70%的硫酸溶液,50~70℃搅拌3~5小时,过滤,将滤饼加入乙酸乙酯和水中,萃取收集有机相将有机相经硫酸镁干燥,蒸掉溶剂,用甲苯重结晶,干燥得到反式结构的酸 1 will have a cis-trans racemic acid Adding to a sulfuric acid solution with a volume fraction of 40 to 70%, stirring at 50 to 70 ° C for 3 to 5 hours, filtering, adding the filter cake to ethyl acetate and water, extracting and collecting the organic phase, drying the organic phase with magnesium sulfate, and evaporating the solvent Recrystallized from toluene and dried to give the acid of the trans structure
②将和步骤①所得的反式结构的酸加入溶剂中,加入缚酸剂,在20~30℃下搅拌反应22~26小时,在真空度0.08~0.10kPa下,蒸馏除去溶剂,加二氯甲烷和水搅拌均匀,静置分层除去水,在真空度0.08~0.10kPa下,蒸馏除去二氯甲烷得到通式I所示的反式结构的含内酯环的杀线虫剂;其中和缚酸剂的摩尔比为1∶0.8~1.2∶3~5;2 will And the acid of the trans structure obtained in step 1. Adding to the solvent, adding an acid binding agent, stirring the reaction at 20 to 30 ° C for 22 to 26 hours, distilling off the solvent under a vacuum of 0.08 to 0.10 kPa, adding methylene chloride and water, stirring uniformly, and standing to separate the layer to remove water. Distilling off methylene chloride at a vacuum of 0.08 to 0.10 kPa to obtain a lactone ring-containing nematicide of the trans structure represented by Formula I; The molar ratio to the acid binding agent is 1:0.8 to 1.2:3 to 5;
其中,R1、R2、R3、R4、R5选自氢、氰基、氟、氯、溴、含1~4个碳原子的烷基、含1~4个碳原子的烷氧基、烷氧苯基、含1~4个碳原子并且碳原子上氢原子被一个或多个氯原子取代的烷氧基、含1~4个碳原子并且碳原子上氢原子被一个或多个氟原子取代的烷氧基、硝基或胺基;R6选自氢、氟、氯;Wherein R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms. Alkoxyphenyl, an alkoxy group having from 1 to 4 carbon atoms and having a hydrogen atom on the carbon atom substituted by one or more chlorine atoms, having from 1 to 4 carbon atoms and having one or more hydrogen atoms on the carbon atom a fluorine atom-substituted alkoxy group, a nitro group or an amine group; and R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine;
所述的溶剂为甲醇、乙醇、丙酮或N,N-二甲基甲酰胺中的一种; The solvent is one of methanol, ethanol, acetone or N,N-dimethylformamide;
所述的缚酸剂为碳酸钾、碳酸钠、吡啶或三乙胺中的一种。The acid binding agent is one of potassium carbonate, sodium carbonate, pyridine or triethylamine.
本发明还提供了反式结构的含内酯环的杀线虫剂的用途,用于防治农业线虫类病害。The invention also provides the use of a lactone ring-containing nematicide having a trans structure for controlling agricultural nematode diseases.
本发明的优点在于:The advantages of the invention are:
本发明的反式结构的含内酯环的杀线虫剂由于含有多氟丁烯和内酯环的结构,因而对根结线虫的卵与二龄幼虫有很好的防治效果,尤其能很好地抑制黄瓜、番茄、烟草、大豆等根结线虫卵的孵化;并且本发明的反式结构的含内酯环的杀线虫剂的毒性较低,在农作物中的残留少,对人、畜的危害性小,很好解决了现有的杀线虫剂毒性较大,在作物中的残留较多的问题,提高了农业生产的安全性;在较长时间内使用害虫不会对本发明的杀线虫剂产生抗体,具有很好的杀虫效果;本发明还提供了该反式结构的含内酯环的杀线虫剂的制备方法,该制备方法步骤少,工艺易于操作,适合大规模的工业生产。The lactone ring-containing nematicide of the trans structure of the present invention has a good control effect on the eggs of the root knot nematode and the second instar larvae due to the structure containing the polyfluorobutene and the lactone ring, especially good. Inhibiting the hatching of root-knot nematode eggs such as cucumber, tomato, tobacco, soybean, etc.; and the lactone ring-containing nematicide of the trans structure of the present invention is less toxic, has less residue in crops, and is suitable for humans and animals. The harm is small, which solves the problem that the existing nematicide is more toxic, and the residue in the crop is more, which improves the safety of agricultural production; the use of pests in a long time does not kill the invention. The nematode produces antibody and has good insecticidal effect; the invention also provides a preparation method of the transester structure containing a lactone ring-containing nematicide, the preparation method has few steps, the process is easy to operate, and is suitable for large-scale industry. produce.
一种反式结构的含内酯环的杀线虫剂,其结构通式I如下所示:A trans-structured nematicidal agent containing a lactone ring, the structural formula I of which is as follows:
结构通式I, Structural formula I,
其中,R1、R2、R3、R4、R5选自氢、氰基、氟、氯、溴、含1~4个碳原子的烷基、含1~4个碳原子的烷氧基、烷氧苯基、含1~4个碳原子并且碳原子上氢原子被一个或多个氯原子取代的烷氧基、含1~4个碳原子并且碳原子上氢原子被一个或多个氟原子取代的烷氧基、硝基或胺基;Wherein R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms. Alkoxyphenyl, an alkoxy group having from 1 to 4 carbon atoms and having a hydrogen atom on the carbon atom substituted by one or more chlorine atoms, having from 1 to 4 carbon atoms and having one or more hydrogen atoms on the carbon atom a fluorine atom-substituted alkoxy group, a nitro group or an amine group;
R6选自氢、氟、氯。R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine.
优选的,一种反式结构的含内酯环的杀线虫剂,R6为氟原子。Preferably, the nematicide containing an anti-lactone ring structure of formula, R 6 is a fluorine atom.
优选的,一种反式结构的含内酯环的杀线虫剂,R1=CF3时,R2=R3=R4=R5=H。Preferably, a lactone ring-containing nematicide of trans structure, when R 1 =CF 3 , R 2 =R 3 =R 4 =R 5 =H.
优选的,一种反式结构的含内酯环的杀线虫剂,R1=OCF3时,R2=R3=R4=R5=H。Preferably, a lactone ring-containing nematicide of trans structure, when R 1 =OCF 3 , R 2 =R 3 =R 4 =R 5 =H.
优选的,一种反式结构的含内酯环的杀线虫剂,R1=R2=R5=H,R3=F时,R4=-O-C6H5。Preferably, a lactone ring-containing nematicide having a trans structure, R 1 = R 2 = R 5 = H, and when R 3 = F, R 4 = -OC 6 H 5 .
优选的,一种反式结构的含内酯环的杀线虫剂,R2=R4=CF3时,R1=R3=R5=H。 本发明还提供了反式结构的含内酯环的杀线虫剂的制备方法,包括以下步骤:Preferably, a lactone ring-containing nematicide of trans structure, when R 2 =R 4 =CF 3 , R 1 =R 3 =R 5 =H. The invention also provides a preparation method of a lactone ring-containing nematicidal agent having a trans structure, comprising the following steps:
①将具有顺反式外消旋的酸加入到体积分数为40~70%的硫酸溶液,50~70℃搅拌3~5小时,过滤,将滤饼加入乙酸乙酯和水中,萃取收集有机相将有机相经硫酸镁干燥,蒸掉溶剂,用甲苯重结晶,干燥得到反式结构的酸 1 will have a cis-trans racemic acid Adding to a sulfuric acid solution with a volume fraction of 40 to 70%, stirring at 50 to 70 ° C for 3 to 5 hours, filtering, adding the filter cake to ethyl acetate and water, extracting and collecting the organic phase, drying the organic phase with magnesium sulfate, and evaporating the solvent Recrystallized from toluene and dried to give the acid of the trans structure
②将和步骤①所得的反式结构的酸加入溶剂中,加入缚酸剂,在20~30℃下搅拌反应22~26小时,在真空度0.08~0.10kPa下,蒸馏除去溶剂,加二氯甲烷和水搅拌均匀,静置分层除去水,在真空度0.08~0.10kPa下,蒸馏除去二氯甲烷得到通式I所示的反式结构的含内酯环的杀线虫剂;其中和缚酸剂的摩尔比为1∶0.8~1.2∶3~5;2 will And the acid of the trans structure obtained in step 1. Adding to the solvent, adding an acid binding agent, stirring the reaction at 20 to 30 ° C for 22 to 26 hours, distilling off the solvent under a vacuum of 0.08 to 0.10 kPa, adding methylene chloride and water, stirring uniformly, and standing to separate the layer to remove water. Distilling off methylene chloride at a vacuum of 0.08 to 0.10 kPa to obtain a lactone ring-containing nematicide of the trans structure represented by Formula I; The molar ratio to the acid binding agent is 1:0.8 to 1.2:3 to 5;
其中,R1、R2、R3、R4、R5选自氢、氰基、氟、氯、溴、含1~4个碳原子的烷基、含1~4个碳原子的烷氧基、烷氧苯基、含1~4个碳原子并且碳原子上氢原子被一个或多个氯原子取代的烷氧基、含1~4个碳原子并且碳原子上氢原子被一个或多个氟原子取代的烷氧基、硝基或胺基;R6选自氢、氟、氯;Wherein R 1 , R 2 , R 3 , R 4 and R 5 are selected from the group consisting of hydrogen, cyano, fluorine, chlorine, bromine, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms. Alkoxyphenyl, an alkoxy group having from 1 to 4 carbon atoms and having a hydrogen atom on the carbon atom substituted by one or more chlorine atoms, having from 1 to 4 carbon atoms and having one or more hydrogen atoms on the carbon atom a fluorine atom-substituted alkoxy group, a nitro group or an amine group; and R 6 is selected from the group consisting of hydrogen, fluorine, and chlorine;
其反应通式为:Its reaction formula is:
所述的溶剂为甲醇、乙醇、丙酮或N,N-二甲基甲酰胺中的一种; The solvent is one of methanol, ethanol, acetone or N,N-dimethylformamide;
所述的缚酸剂为碳酸钾、碳酸钠、吡啶或三乙胺中的一种。The acid binding agent is one of potassium carbonate, sodium carbonate, pyridine or triethylamine.
本发明还提供了反式结构的含内酯环的杀线虫剂的用途,用于防治农业线虫类病害。The invention also provides the use of a lactone ring-containing nematicide having a trans structure for controlling agricultural nematode diseases.
本发明制备方法中的原料可在市场上购买,或采用以下步骤制备:将和无水氯化锌依次加入到二氯甲烷中,在0~5℃下,滴加三乙胺,滴加完毕后得到反应液,将反应液置于20~30℃下搅拌10~20小时,向其中加入盐酸调节反应液的pH至2,加入乙酸乙酯萃取,收集有机相,加入甲苯重结晶得到其中无水氯化锌和三乙胺的摩尔比为1∶0.8~1.2∶1~3∶3~5;其反应通式为:Raw materials in the preparation method of the invention Can be purchased on the market or prepared by the following steps: And anhydrous zinc chloride is added to dichloromethane in turn, triethylamine is added dropwise at 0 to 5 ° C, the reaction solution is obtained after the dropwise addition, and the reaction solution is stirred at 20 to 30 ° C for 10 to 20 hours. Add hydrochloric acid to adjust the pH of the reaction solution to 2, extract with ethyl acetate, collect the organic phase, and recrystallize by adding toluene. among them The molar ratio of anhydrous zinc chloride to triethylamine is from 1:0.8 to 1.2:1 to 3:3 to 5; the reaction formula is:
表1一种反式结构的含内酯环的杀线虫剂的化合物的结构测量分析表Table 1 Structural measurement analysis table of a trans-seed nematicide-containing compound of trans structure
本发明的一种反式结构的含内酯环的杀线虫剂均可按照本发明的制备方法得到,下面为表1中化合物的具体实施例:A lactone ring-containing nematicide of the trans structure of the present invention can be obtained according to the preparation method of the present invention. The following are specific examples of the compounds in Table 1:
实施例1Example 1
制备表1中序号2的化合物包括下述步骤:The preparation of the compound of No. 2 in Table 1 comprises the following steps:
①将1mol邻甲基苯甲醛、0.8mol琥珀酐和1mol无水氯化锌加入300ml二氯甲烷中,在0℃下滴加3mol三乙胺,滴加完毕得到反应液,将反应液置于20℃下搅拌10h,向其中加入盐酸调节反应液的pH至2,加入200ml乙酸乙酯萃取, 收集有机相,向有机相中加入200ml甲苯重结晶得到5-氧代-2-(2-甲基苯基)四氢呋喃-3-羧酸;1 1 mol of o-methylbenzaldehyde, 0.8 mol of succinic anhydride and 1 mol of anhydrous zinc chloride were added to 300 ml of dichloromethane, and 3 mol of triethylamine was added dropwise at 0 ° C, and the reaction liquid was obtained by dropwise addition, and the reaction liquid was placed. After stirring at 20 ° C for 10 h, hydrochloric acid was added thereto to adjust the pH of the reaction mixture to 2, and extracted with 200 ml of ethyl acetate. The organic phase was collected, and 200 ml of toluene was added to the organic phase to recrystallize to give 5-oxo-2-(2-methylphenyl)tetrahydrofuran-3-carboxylic acid;
②将步骤①所得5-氧代-2-(2-甲基苯基)四氢呋喃-3-羧酸加入40%的硫酸溶液中,50℃搅拌3h,过滤,将滤饼加入乙酸乙酯和水中,萃取收集有机相将有机相经硫酸镁干燥,蒸掉溶剂,剩余的有机物用甲苯重结晶,干燥得到反式结构的5-氧代-2-(2-甲基苯基)四氢呋喃-3-羧酸。2 The 5-oxo-2-(2-methylphenyl)tetrahydrofuran-3-carboxylic acid obtained in the step 1 was added to a 40% sulfuric acid solution, stirred at 50 ° C for 3 h, filtered, and the filter cake was added to ethyl acetate and water. The organic phase is extracted and dried over magnesium sulfate, the solvent is evaporated, and the remaining organics are recrystallized from toluene and dried to give 5-methoxy-2-(2-methylphenyl)tetrahydrofuran-3- carboxylic acid.
③将步骤②所得0.5mol反式结构的5-氧代-2-(2-甲基苯基)四氢呋喃-3-羧酸和0.4mol 4-溴-1,1-二氟-1-丁烯加入500ml甲醇中,加入1.5mol碳酸钾,在20℃下搅拌反应22小时,在真空度0.08kPa下,蒸馏除去甲醇,将剩余物加入100ml二氯甲烷和50ml水搅拌均匀,静置分层,收集二氯甲烷相,将收集的二氯甲烷相在真空度0.08kPa下蒸馏除去二氯甲烷得到产品,即表1中序号2的化合物。3 0.5 mol of trans-structured 5-oxo-2-(2-methylphenyl)tetrahydrofuran-3-carboxylic acid obtained in step 2 and 0.4 mol of 4-bromo-1,1-difluoro-1-butene After adding 500 ml of methanol, 1.5 mol of potassium carbonate was added, and the reaction was stirred at 20 ° C for 22 hours. At a vacuum of 0.08 kPa, methanol was distilled off, and the residue was added to 100 ml of dichloromethane and 50 ml of water, and the mixture was uniformly stirred. The methylene chloride phase was collected, and the collected dichloromethane phase was distilled off at a vacuum of 0.08 kPa to obtain a product, that is, a compound of the formula 2 in Table 1.
元素分析结果:C,61.93;H,5.20;F,12.25;O,20.62。Elemental analysis results: C, 61.93; H, 5.20; F, 12.25; O, 20.62.
核磁分析结果:δ2.24,2H;δ2.34,3H;δ2.52-2.77,2H;δ3.31,1H;δ4.12,2H;δ4.28,1H;δ6.21,1H;δ7.19-7.26,3H;δ7.39,1H。Nuclear magnetic analysis results: δ2.24, 2H; δ 2.34, 3H; δ 2.52-2.77, 2H; δ3.31, 1H; δ 4.12, 2H; δ 4.28, 1H; δ 6.21, 1H; δ 7. 19-7.26, 3H; δ 7.39, 1H.
实施例2Example 2
制备表1中序号26的化合物包括下述步骤:The preparation of the compound of No. 26 in Table 1 comprises the following steps:
①将1mol4-氟-5-苯氧基苯甲醛、1.2mol琥珀酐和3mol无水氯化锌加入400ml二氯甲烷中,在5℃下滴加5mol三乙胺,滴加完毕得到反应液,将反应液置于30℃下搅拌20h,向其中加入盐酸调节反应液的pH至2,加入250ml乙酸乙酯萃取,收集有机相,向有机相中加入250ml甲苯重结晶得到5-氧代-2-(2-(4-氟-5-苯氧基)苯基)四氢呋喃-3-羧酸;1 1 mol of 4-fluoro-5-phenoxybenzaldehyde, 1.2 mol of succinic anhydride and 3 mol of anhydrous zinc chloride were added to 400 ml of dichloromethane, and 5 mol of triethylamine was added dropwise at 5 ° C, and the reaction liquid was obtained by dropwise addition. The reaction solution was stirred at 30 ° C for 20 h, hydrochloric acid was added thereto to adjust the pH of the reaction mixture to 2, and extracted with 250 ml of ethyl acetate. The organic phase was collected, and the organic phase was recrystallized by adding 250 ml of toluene to give 5-oxo-2. -(2-(4-fluoro-5-phenoxy)phenyl)tetrahydrofuran-3-carboxylic acid;
②将步骤①所得5-氧代-2-(2-(4-氟-5-苯氧基)苯基)四氢呋喃-3-羧酸加入70%的硫酸溶液中,40℃搅拌5h,过滤,将滤饼加入乙酸乙酯和水中,萃取收集有机相将有机相经硫酸镁干燥,蒸掉溶剂,剩余的有机物用甲苯重结晶,干燥得到反式结构的5-氧代-2-(2-(4-氟-5-苯氧基)苯基)四氢呋喃-3-羧酸。2 The 5-oxo-2-(2-(4-fluoro-5-phenoxy)phenyl)tetrahydrofuran-3-carboxylic acid obtained in the step 1 was added to a 70% sulfuric acid solution, stirred at 40 ° C for 5 h, and filtered. The filter cake was added to ethyl acetate and water, and the organic phase was extracted and dried. The organic phase was dried over magnesium sulfate, and the solvent was evaporated, and the remaining organics were recrystallized from toluene and dried to give 5-oxy-2-(2-) (4-Fluoro-5-phenoxy)phenyl)tetrahydrofuran-3-carboxylic acid.
③将步骤②所得0.5mol反式结构的5-氧代-2-(2-(4-氟-5-苯氧基)苯基)四氢呋喃-3-羧酸和0.6mol 4-溴-1,1-二氟-1-丁烯加入400ml乙醇中,加入2.5mol碳酸钠,在30℃下搅拌反应26小时,在真空度0.10kPa下,蒸馏除去乙醇,将剩余物加入120ml二氯甲烷和80ml水搅拌均匀,静置分层,收集二氯甲烷相, 将收集的二氯甲烷相在真空度0.12kPa下蒸馏除去二氯甲烷得到产品,即表1中序号26的化合物。3 0.5 mol of the 5-methoxy-2-(2-(4-fluoro-5-phenoxy)phenyl)tetrahydrofuran-3-carboxylic acid obtained in the step 2 and 0.6 mol of 4-bromo-1, 1-Difluoro-1-butene was added to 400 ml of ethanol, 2.5 mol of sodium carbonate was added, and the reaction was stirred at 30 ° C for 26 hours. At a vacuum of 0.10 kPa, ethanol was distilled off, and the residue was added to 120 ml of dichloromethane and 80 ml. The water was stirred well, and the layers were allowed to stand, and the dichloromethane phase was collected. The collected dichloromethane phase was distilled off at a vacuum of 0.12 kPa to give the product, that is, the compound of No. 26 in Table 1.
元素分析结果:C,59.44;H,3.80;F,17.91;O,18.85。Elemental analysis results: C, 59.44; H, 3.80; F, 17.91; O, 18.85.
核磁分析结果:δ2.24,2H;δ2.52-2.77,2H;δ3.31,1H;δ4.12,2H;δ6.21,1H;δ7.06-7.14,5H;δ7.27,1H;δ7.41,2H。Nuclear magnetic analysis results: δ2.24, 2H; δ 2.52-2.77, 2H; δ 3.31, 1H; δ 4.12, 2H; δ 6.21, 1H; δ7.06-7.14, 5H; δ 7.27, 1H; δ 7.41, 2H.
实施例3Example 3
制备表1中序号41的化合物包括下述步骤:The preparation of the compound of No. 41 in Table 1 includes the following steps:
①将1mol邻三氟甲基苯甲醛、1mol琥珀酐和2mol无水氯化锌加入300ml二氯甲烷中,在5℃下滴加4mol三乙胺,滴加完毕得到反应液,将反应液置于25℃下搅拌15h,向其中加入盐酸调节反应液的pH至2,加入220ml乙酸乙酯萃取,收集有机相,向有机相中加入220ml甲苯重结晶得到5-氧代-2-(2-(三氟甲氧基)苯基)四氢呋喃-3-羧酸;1 1 mol of o-trifluoromethylbenzaldehyde, 1 mol of succinic anhydride and 2 mol of anhydrous zinc chloride were added to 300 ml of dichloromethane, 4 mol of triethylamine was added dropwise at 5 ° C, and the reaction liquid was obtained after the dropwise addition, and the reaction liquid was placed. After stirring at 25 ° C for 15 h, hydrochloric acid was added thereto to adjust the pH of the reaction mixture to 2, and extracted with 220 ml of ethyl acetate. The organic phase was collected, and the organic phase was recrystallized by adding 220 ml of toluene to give 5-oxo-2-(2- (trifluoromethoxy)phenyl)tetrahydrofuran-3-carboxylic acid;
②将步骤①所得5-氧代-2-(2-(三氟甲氧基)苯基)四氢呋喃-3-羧酸加入50%的硫酸溶液中,70℃搅拌5h,过滤,将滤饼加入乙酸乙酯和水中,萃取收集有机相将有机相经硫酸镁干燥,蒸掉溶剂,剩余的有机物用甲苯重结晶,干燥得到反式结构的5-氧代-2-(2-(三氟甲氧基)苯基)四氢呋喃-3-羧酸。2 The 5-oxo-2-(2-(trifluoromethoxy)phenyl)tetrahydrofuran-3-carboxylic acid obtained in step 1 was added to a 50% sulfuric acid solution, stirred at 70 ° C for 5 h, filtered, and the filter cake was added. The organic phase is extracted with ethyl acetate and water, and the organic phase is dried over magnesium sulfate. The solvent is evaporated, and the remaining organics are recrystallized from toluene, and dried to give 5-oxy-2-(2-(trifluoro) Oxy)phenyl)tetrahydrofuran-3-carboxylic acid.
③将步骤②所得0.5mol反式结构的5-氧代-2-(2-(三氟甲氧基)苯基)四氢呋喃-3-羧酸和0.5mol 4-溴-1,1,2-三氟-1-丁烯加入380ml丙酮中,加入2.0mol吡啶,在25℃下搅拌反应24小时,在真空度0.10kPa下,蒸馏除去丙酮,将剩余物加入120ml二氯甲烷和100ml水搅拌均匀,静置分层,收集二氯甲烷相,将收集的二氯甲烷相在真空度0.10kPa下蒸馏除去二氯甲烷得到产品,即表1中序号41的化合物。3 0.5 mol of trans-structured 5-oxo-2-(2-(trifluoromethoxy)phenyl)tetrahydrofuran-3-carboxylic acid obtained in step 2 and 0.5 mol of 4-bromo-1,1,2- Trifluoro-1-butene was added to 380 ml of acetone, 2.0 mol of pyridine was added, and the reaction was stirred at 25 ° C for 24 hours. The acetone was distilled off under a vacuum of 0.10 kPa, and the residue was added to 120 ml of dichloromethane and 100 ml of water and stirred well. The mixture was allowed to stand for stratification, the dichloromethane phase was collected, and the collected dichloromethane phase was distilled off at a vacuum of 0.10 kPa to obtain a product, that is, a compound of the formula 41 in Table 1.
元素分析结果:C,48.25;H,3.04;F,28.62;O,20.09。Elemental analysis results: C, 48.25; H, 3.04; F, 28.62; O, 20.09.
核磁分析结果:δ2.24,2H;δ2.52-2.77,2H;δ3.31,1H;δ4.12,2H;δ6.21,1H;δ6.92-6.96,3H;δ7.25,1H。Nuclear magnetic analysis results: δ2.24, 2H; δ 2.52-2.77, 2H; δ 3.31, 1H; δ 4.12, 2H; δ 6.21, 1H; δ 6.92-6.96, 3H; δ 7.25, 1H.
实施例4Example 4
制备表1中序号46的化合物包括下述步骤:The preparation of the compound of No. 46 in Table 1 includes the following steps:
①将1mol邻三氟甲基苯甲醛、1.1mol琥珀酐和2.5mol无水氯化锌加入300ml二氯甲烷中,在3℃下滴加3.5mol三乙胺,滴加完毕得到反应液,将反应液置于 22℃下搅拌12h,向其中加入盐酸调节反应液的pH至2,加入250ml乙酸乙酯萃取,收集有机相,向有机相中加入250ml甲苯重结晶得到5-氧代-2-(2-(三氟甲基)苯基)四氢呋喃-3-羧酸;1 1 mol of o-trifluoromethylbenzaldehyde, 1.1 mol of succinic anhydride and 2.5 mol of anhydrous zinc chloride were added to 300 ml of dichloromethane, and 3.5 mol of triethylamine was added dropwise at 3 ° C, and the reaction liquid was obtained by dropwise addition. Reaction solution After stirring at 22 ° C for 12 h, hydrochloric acid was added thereto to adjust the pH of the reaction mixture to 2, and extracted with 250 ml of ethyl acetate. The organic phase was collected, and the organic phase was recrystallized by adding 250 ml of toluene to give 5-oxo-2-(2-( Trifluoromethyl)phenyl)tetrahydrofuran-3-carboxylic acid;
②将步骤①所得5-氧代-2-(2-(三氟甲基)苯基)四氢呋喃-3-羧酸加入40%的硫酸溶液中,70℃搅拌4h,过滤,将滤饼加入乙酸乙酯和水中,萃取收集有机相将有机相经硫酸镁干燥,蒸掉溶剂,剩余的有机物用甲苯重结晶,干燥得到反式结构的5-氧代-2-(2-(三氟甲基)苯基)四氢呋喃-3-羧酸。2 The 5-oxo-2-(2-(trifluoromethyl)phenyl)tetrahydrofuran-3-carboxylic acid obtained in the step 1 was added to a 40% sulfuric acid solution, stirred at 70 ° C for 4 h, filtered, and the filter cake was added to acetic acid. The organic phase is extracted with ethyl acetate and water. The organic phase is dried over magnesium sulfate, and the solvent is evaporated. The remaining organics are recrystallized from toluene and dried to give 5- thio-2-(2-(trifluoromethyl) )phenyl)tetrahydrofuran-3-carboxylic acid.
③将步骤②所得0.5mol反式结构的5-氧代-2-(2-(三氟甲基)苯基)四氢呋喃-3-羧酸和0.5mol 4-溴-1,1,2-三氟-1-丁烯加入380ml丙酮中,加入2.0mol三乙胺,在25℃下搅拌反应22小时,在真空度0.10kPa下,蒸馏除去丙酮,将剩余物加入120ml二氯甲烷和100ml水搅拌均匀,静置分层,收集二氯甲烷相,将收集的二氯甲烷相在真空度0.10kPa下蒸馏除去二氯甲烷得到产品,即表1中序号46的化合物。3 0.5 mol of trans-structured 5-oxo-2-(2-(trifluoromethyl)phenyl)tetrahydrofuran-3-carboxylic acid obtained in step 2 and 0.5 mol of 4-bromo-1,1,2-tri Fluor-1-butene was added to 380 ml of acetone, 2.0 mol of triethylamine was added, and the reaction was stirred at 25 ° C for 22 hours. At a vacuum of 0.10 kPa, acetone was distilled off, and the residue was added to 120 ml of dichloromethane and 100 ml of water and stirred. The product was homogeneous, and the mixture was allowed to stand for separation. The methylene chloride phase was collected, and the collected methylene chloride phase was distilled off at a vacuum of 0.10 kPa to obtain a product, that is, the compound of No. 46 in Table 1.
元素分析结果:C,50.28;H,3.16;F,29.82;O,16.73。Elemental analysis results: C, 50.28; H, 3.16; F, 29.82; O, 16.73.
核磁分析结果:δ2.24,2H;δ2.52-2.77,2H;δ3.31,1H;δ4.12,2H;δ6.21,1H;δ7.29-7.38,3H;δ7.55,1H。Nuclear magnetic analysis results: δ2.24, 2H; δ 2.52-2.77, 2H; δ 3.31, 1H; δ 4.12, 2H; δ 6.21, 1H; δ 7.29-7.38, 3H; δ 7.55, 1H.
实施例5Example 5
制备表1中序号48的化合物包括下述步骤:The preparation of the compound of No. 48 in Table 1 comprises the following steps:
①将1mol 3,5-双三氟甲基苯甲醛、0.9mol琥珀酐和2.5mol无水氯化锌加入300ml二氯甲烷中,在4℃下滴加3.5mol三乙胺,滴加完毕得到反应液,将反应液置于22℃下搅拌16h,向其中加入盐酸调节反应液的pH至2,加入250ml乙酸乙酯萃取,收集有机相,向有机相中加入250ml甲苯重结晶得到5-氧代-2-(2-(3,5-双三氟甲基)苯基)四氢呋喃-3-羧酸;1 1 mol of 3,5-bistrifluoromethylbenzaldehyde, 0.9 mol of succinic anhydride and 2.5 mol of anhydrous zinc chloride were added to 300 ml of dichloromethane, and 3.5 mol of triethylamine was added dropwise at 4 ° C, and the addition was completed. The reaction solution was stirred at 22 ° C for 16 h, hydrochloric acid was added thereto to adjust the pH of the reaction mixture to 2, and extracted with 250 ml of ethyl acetate. The organic phase was collected, and the organic phase was recrystallized by adding 250 ml of toluene to obtain 5-oxo. 2-(2-(3,5-bistrifluoromethyl)phenyl)tetrahydrofuran-3-carboxylic acid;
②将步骤①所得5-氧代-2-(2-(3,5-双三氟甲基)苯基)四氢呋喃-3-羧酸加入70%的硫酸溶液中,70℃搅拌5h,过滤,将滤饼加入乙酸乙酯和水中,萃取收集有机相将有机相经硫酸镁干燥,蒸掉溶剂,剩余的有机物用甲苯重结晶,干燥得到反式结构的5-氧代-2-(2-(3,5-双三氟甲基)苯基)四氢呋喃-3-羧酸。2 The 5-oxo-2-(2-(3,5-bistrifluoromethyl)phenyl)tetrahydrofuran-3-carboxylic acid obtained in the step 1 was added to a 70% sulfuric acid solution, stirred at 70 ° C for 5 h, and filtered. The filter cake was added to ethyl acetate and water, and the organic phase was extracted and dried. The organic phase was dried over magnesium sulfate, and the solvent was evaporated, and the remaining organics were recrystallized from toluene and dried to give 5-oxy-2-(2-) (3,5-Bistrifluoromethyl)phenyl)tetrahydrofuran-3-carboxylic acid.
③将步骤②所得0.5mol反式结构的5-氧代-2-(2-(3,5-双三氟甲基)苯基)四氢呋喃-3-羧酸和0.5mol 4-溴-1,1,2-三氟-1-丁烯加入380ml丙酮中,加入 2.0mol三乙胺,在25℃下搅拌反应22小时,在真空度0.10kPa下,蒸馏除去丙酮,将剩余物加入120ml二氯甲烷和100ml水搅拌均匀,静置分层,收集二氯甲烷相,将收集的二氯甲烷相在真空度0.10kPa下蒸馏除去二氯甲烷得到产品,即表1中序号48的化合物。3 0.5 mol of trans-structured 5-oxo-2-(2-(3,5-bistrifluoromethyl)phenyl)tetrahydrofuran-3-carboxylic acid and 0.5 mol of 4-bromo-1 obtained in step 2. 1,2-Trifluoro-1-butene was added to 380 ml of acetone and added 2.0 mol of triethylamine, the reaction was stirred at 25 ° C for 22 hours, the acetone was distilled off under a vacuum of 0.10 kPa, and the residue was added to 120 ml of dichloromethane and 100 ml of water, stirred uniformly, and allowed to stand for separation. The collected dichloromethane phase was distilled off at a vacuum of 0.10 kPa to give the product, that is, the compound of No. 48 in Table 1.
元素分析结果:C,45.35;H,2.46;F,37.98;O,14.21。Elemental analysis results: C, 45.35; H, 2.46; F, 37.98; O, 14.21.
核磁分析结果:δ2.24,2H;δ2.52-2.77,2H;δ3.31,1H;δ4.12,2H;δ6.21,1H;δ7.62,2H;δ7.94,1H。Nuclear magnetic analysis results: δ2.24, 2H; δ 2.52-2.77, 2H; δ 3.31, 1H; δ 4.12, 2H; δ 6.21, 1H; δ 7.62, 2H; δ 7.94, 1H.
杀线虫试验Nematicidal test
对表1中的化合物采用浸虫法进行了抑制植物病原线虫试验,以测定以上化合物对二龄幼虫的活性,同时参照NY/T 1154.5-2006(第5部分:杀卵活性试验浸渍法)测试以上化合物对线虫卵孵化的抑制活性,结果如表2所示:The compounds in Table 1 were tested by the immersion method for the inhibition of phytopathogenic nematodes to determine the activity of the above compounds against the second instar larvae, and tested in accordance with NY/T 1154.5-2006 (Part 5: Oviposition activity test impregnation method) The inhibitory activities of the above compounds on the hatching of nematode eggs are shown in Table 2:
表2含反式内酯环的杀线虫剂的化合物抑制植物病原线虫试验结果Table 2: Inhibition of phytopathogenic nematodes by compounds of nematicides containing trans lactone ring
由表1中的数据可知,本发明反式结构的含内酯环的杀线虫剂的化合物对线虫二龄幼虫及卵均有很好的防治效果,并且对线虫卵的孵化抑制率要比对二龄幼虫的效果好。It can be seen from the data in Table 1 that the lactone ring-containing nematicidal compound of the trans structure of the present invention has a good control effect on the second instar larvae and eggs of the nematode, and the hatching inhibition rate of the nematode eggs is higher than that. Good effect on second instar larvae.
毒性试验Toxicity test
根据化学农药环境安全评价试验准则中的蚯蚓毒性试验和土壤微生物毒性试验的步骤对本发明表1中的化合物1~60进行蚯蚓毒性试验和土壤微生物毒性试验,其中土壤微生物试验中模拟的农药常用量为40ppm,两个试验的结果如下:The cockroach toxicity test and the soil microbial toxicity test of the compounds 1 to 60 in Table 1 of the present invention are carried out according to the steps of the cockroach toxicity test and the soil microbial toxicity test in the chemical pesticide environmental safety evaluation test standard, wherein the commonly used pesticide amount in the soil microbial test is simulated. At 40 ppm, the results of the two tests are as follows:
表2反式内酯环的杀线虫剂的化合物的毒性实验结果Table 2 Results of toxicity test of nematicidal compound of trans lactone ring
由表2中的数据可知,该本发明反式结构含内酯环的杀线虫剂的化合物对土壤环境中的生物的毒性低,使用安全,属于环境友好型化合物。 As can be seen from the data in Table 2, the transestericide-containing nematicidal compound of the present invention has low toxicity to organisms in the soil environment, is safe to use, and is an environmentally friendly compound.
Claims (8)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201520768728 | 2015-09-30 | ||
| CN201520768728.9 | 2015-09-30 | ||
| CN201510653910.4 | 2015-10-10 | ||
| CN201510653910.4A CN106554335B (en) | 2015-09-30 | 2015-10-10 | A kind of nematicide containing lactonic ring of transconfiguration and its production and use |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017054524A1 true WO2017054524A1 (en) | 2017-04-06 |
Family
ID=58417786
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2016/086427 Ceased WO2017054524A1 (en) | 2015-09-30 | 2016-06-20 | Nematicide of trans structure containing lactone ring and preparation method and use thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN106554335B (en) |
| WO (1) | WO2017054524A1 (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2025117659A1 (en) | 2023-11-29 | 2025-06-05 | Basf Corporation | Insecticide delivery by nanocarriers |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107372506A (en) * | 2017-07-26 | 2017-11-24 | 山东省联合农药工业有限公司 | A kind of composition pesticide and its formulation and purposes containing new lactone cyclization compound |
| CN108484538B (en) * | 2018-06-08 | 2020-06-12 | 山东省联合农药工业有限公司 | Synthesis method of nematicide containing lactone ring |
| CN113912495A (en) * | 2020-07-09 | 2022-01-11 | 山东省联合农药工业有限公司 | Fluorine-containing butylene ester derivative and preparation method and application thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3513172A (en) * | 1965-09-27 | 1970-05-19 | Stauffer Chemical Co | 3-phenyl-5-(3,4,4-trifluoro-3-butenylthio)1,2,4-thiadiazole |
| CN86104207A (en) * | 1985-06-20 | 1987-04-01 | Fmc有限公司 | The pesticidal use of many halogen alkene derivatives |
| CN1143958A (en) * | 1994-03-10 | 1997-02-26 | 曾尼卡有限公司 | Pesticidally active (4,4-difluorobut-3-enylthio)-substituted heterocyclic or carbocyclic compounds |
| CN102696609A (en) * | 2012-01-10 | 2012-10-03 | 山东中农联合生物科技有限公司 | Trifluoro butene pesticide containing pyridine |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3914251A (en) * | 1971-12-08 | 1975-10-21 | Stauffer Chemical Co | Certain trifluorobutenyl compounds and their utility as nematocides |
| DE19531276A1 (en) * | 1995-08-25 | 1997-02-27 | Bayer Ag | New benzoic acid-(4,4-di:fluoro-but-3-ene-yl)ester derivs. |
| DE19531300A1 (en) * | 1995-08-25 | 1997-02-27 | Bayer Ag | Fluorobutenic acid amides |
| JP2001019685A (en) * | 1999-07-06 | 2001-01-23 | Nippon Bayer Agrochem Co Ltd | Nematocidal trifluorobutene |
| JP2003192675A (en) * | 2001-12-13 | 2003-07-09 | Bayer Ag | Nematicidal trifluorobutenyl imidazole thioether derivative |
| CN102675213A (en) * | 2012-05-28 | 2012-09-19 | 扬州鸿诚化工技术服务有限公司 | Pyridazinone-containing trifluorobutenyl derivatives and preparation method and applications thereof |
| CN104177315B (en) * | 2014-08-06 | 2016-08-24 | 大连大学 | A kind of method synthesizing β-hydrocarbon oxygen acyl group-gamma-alkyl-gamma-butyrolacton |
-
2015
- 2015-10-10 CN CN201510653910.4A patent/CN106554335B/en active Active
-
2016
- 2016-06-20 WO PCT/CN2016/086427 patent/WO2017054524A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3513172A (en) * | 1965-09-27 | 1970-05-19 | Stauffer Chemical Co | 3-phenyl-5-(3,4,4-trifluoro-3-butenylthio)1,2,4-thiadiazole |
| CN86104207A (en) * | 1985-06-20 | 1987-04-01 | Fmc有限公司 | The pesticidal use of many halogen alkene derivatives |
| CN1143958A (en) * | 1994-03-10 | 1997-02-26 | 曾尼卡有限公司 | Pesticidally active (4,4-difluorobut-3-enylthio)-substituted heterocyclic or carbocyclic compounds |
| CN102696609A (en) * | 2012-01-10 | 2012-10-03 | 山东中农联合生物科技有限公司 | Trifluoro butene pesticide containing pyridine |
| CN102696593A (en) * | 2012-01-10 | 2012-10-03 | 山东省联合农药工业有限公司 | Benzene-ring-containing trifluorobutene insecticides |
Non-Patent Citations (2)
| Title |
|---|
| MEOU, A. ET AL.: "Efficient MnIII-mediated synthesis of functionalized trans-3,44-disubstituted- y -butyrolactones", TETRAHEDRON LETTERS, vol. 43, no. 30, 31 December 2002 (2002-12-31), pages 5301 - 5304, XP004368379 * |
| SHEN, DELONG ET AL.: "Progress in Nematicidal Trifluorobutenes Researches", ZHEJIANG CHEMICAL INDUSTRY, vol. 36, no. 12, 31 December 2005 (2005-12-31), pages 22 - 24 , and 33 * |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2025117659A1 (en) | 2023-11-29 | 2025-06-05 | Basf Corporation | Insecticide delivery by nanocarriers |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
Also Published As
| Publication number | Publication date |
|---|---|
| CN106554335B (en) | 2017-09-19 |
| CN106554335A (en) | 2017-04-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2017054524A1 (en) | Nematicide of trans structure containing lactone ring and preparation method and use thereof | |
| WO2017054523A1 (en) | Nematicide containing lactone ring and preparation method and use thereof | |
| CN100448876C (en) | Heterocyclic carboxamide derivatives | |
| WO2017067500A1 (en) | Biphenyl compound and application thereof | |
| WO2015169096A1 (en) | Polysubstituted pyridylpyrazole amide and preparation method and use thereof | |
| CN109320471B (en) | 3- (2, 6-difluorophenyl) -1,2, 4-oxadiazole compound and application thereof | |
| WO2015051572A1 (en) | Class of substituted phenyl pyrazole amide derivatives and preparation method and use thereof | |
| CN102757400B (en) | 2, 5-substituent group oxazole derivative and applications thereof | |
| CN108341808B (en) | A kind of oxadiazole dipyrazole compound and use thereof | |
| CN106632044A (en) | Amide compound containing methylpyrazole and preparation method the application thereof | |
| CN113788801A (en) | Compound containing halogenated thiazole structure, preparation method and application thereof | |
| CN109232550B (en) | A kind of compound containing 3-chloro-5-trifluoromethylpyridyl-1,3,4-oxadiazol-2-one and application thereof | |
| WO2017148226A1 (en) | Fluorobenzoxazole compound and use thereof | |
| CN109320506B (en) | Difluorophenyl oxadiazole insecticide and acaricide | |
| CN111978306A (en) | Furanol pyrazole formamide derivative and preparation method and application thereof | |
| CN117534624A (en) | A kind of uracil phenyl ring ester compound and its preparation method and pesticide composition | |
| CN108570028A (en) | 3- (2- aroyl acetyls base) -4- hydroxyl -6- Methylpyrane -2- ketone derivatives and its application | |
| Zhao et al. | Design, synthesis, and bioactivity study of α-thiocyanato-substituted spirocyclic tetramic acid derivatives | |
| KR910005710B1 (en) | New thiadiazines, preparation method thereof, and insecticidal mites containing the compound | |
| CN105294596B (en) | The dioxide derivative of 4 hydroxyl, 3 benzoyl, 2 alkyl, 1,2 benzothiazine 1,1 | |
| CN102190598A (en) | Cyclopropane oxime ester derivatives and preparation method and application thereof | |
| CN110776463A (en) | Preparation and application of pyrazole oxime derivative containing 3-trifluoromethylpyrazole | |
| CN104557691B (en) | A kind of 3 amine acyl bishydrazide derivatives and its preparation method and application | |
| CN103420975A (en) | Fluorine-contained o-amino thiobenzamide type compound and application thereof | |
| CN101948447B (en) | 4-methylbenzothiazole derivative and preparation method and applications thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16850149 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 32PN | Ep: public notification in the ep bulletin as address of the adressee cannot be established |
Free format text: NOTING OF LOSS OF RIGHTS PURSUANT TO RULE 112(1) EPC (EPO FORM 1205A DATED 23.08.2018) |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 16850149 Country of ref document: EP Kind code of ref document: A1 |