WO2016207345A1 - 3-amino-pyrazin-2-yl carboxamide and 2-amino-pyridin-3-yl carboxamide derivatives as polo-like kinase 1 (plk-1) inhibitors for the treatment of cancer - Google Patents
3-amino-pyrazin-2-yl carboxamide and 2-amino-pyridin-3-yl carboxamide derivatives as polo-like kinase 1 (plk-1) inhibitors for the treatment of cancer Download PDFInfo
- Publication number
- WO2016207345A1 WO2016207345A1 PCT/EP2016/064659 EP2016064659W WO2016207345A1 WO 2016207345 A1 WO2016207345 A1 WO 2016207345A1 EP 2016064659 W EP2016064659 W EP 2016064659W WO 2016207345 A1 WO2016207345 A1 WO 2016207345A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- arom
- heterocycle
- compound
- optionally substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
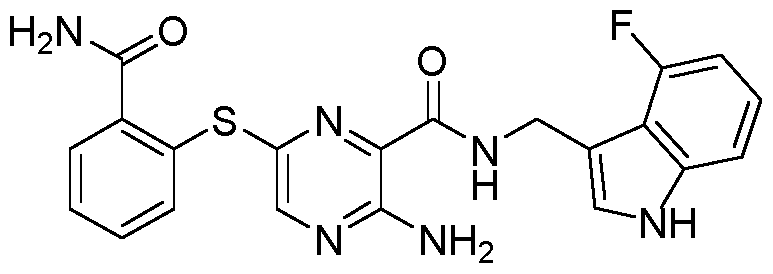
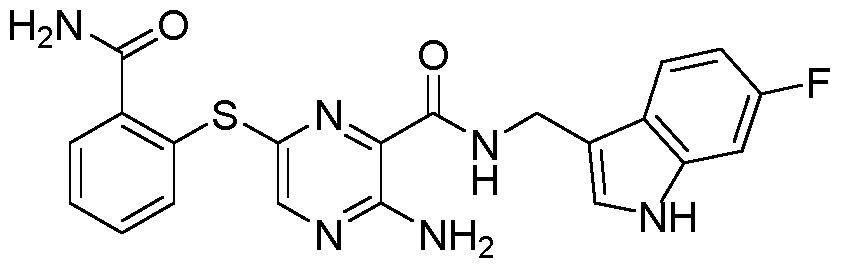
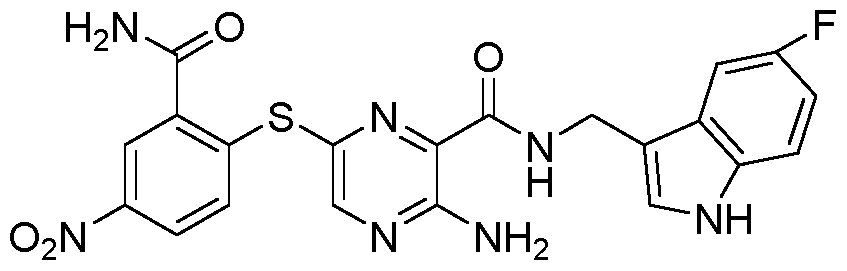
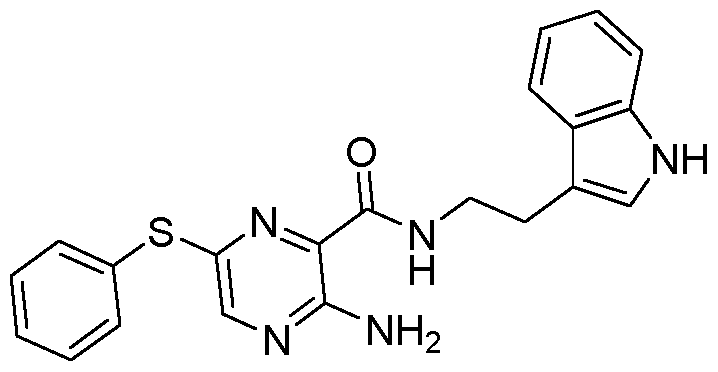
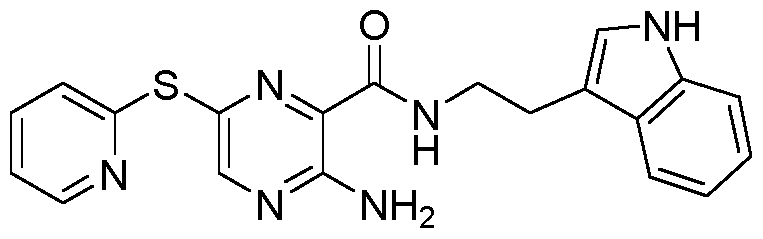
- 0 CC=C(CNC(c1nc(Sc2ccccc2C(N)=O)cnc1N)=O)c(c(*)ccc1)c1F Chemical compound CC=C(CNC(c1nc(Sc2ccccc2C(N)=O)cnc1N)=O)c(c(*)ccc1)c1F 0.000 description 1
- IWMCZGCZISEFRS-UHFFFAOYSA-N NC(c(cccc1)c1Sc(nc1C(NCc(cc(c(F)c2)F)c2O)=O)cnc1N)=O Chemical compound NC(c(cccc1)c1Sc(nc1C(NCc(cc(c(F)c2)F)c2O)=O)cnc1N)=O IWMCZGCZISEFRS-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/10—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D241/14—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D241/24—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D241/26—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals with nitrogen atoms directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
Definitions
- the present invention relates to novel amino-pyridine and amino-pyrazine derivatives, their methods of preparation, pharmaceutical compositions containing the same and their use as a drug, in particular as PLK-1 inhibitors, especially for treating cancer.
- Protein Kinases form a class of enzymes which transferring phosphate groups to substrates, other protein kinases or even themselves put them at the core of most signalling pathways. They are numerous as wild type isoforms as well as disease-causing mutated variants. Their dysregulated expression either through mutation, overexpression or amplification is often a potent driver to carcinogenesis and, as such, qualifies them as highly relevant targets to fight against neoplasia.
- Polo-like Kinase 1 is a still undrugged serine/threonine protein kinase whose many functions qualify it as highly relevant to fight tumorigenesis.
- Polo-Like Kinases Although four variants of Polo-Like Kinases are known in humans (PLK1-4), only PLK1 inhibition by a small molecule is documented to be sufficient to trigger a full G2/M cell cycle block, induction of apoptosis into cancerous cell line and tumor shrinkage in xenografted tumor models.
- PLK1 has been described in several neoplastic conditions such as breast cancer, colorectal cancer, endometrial carcinoma, non small cell lung cancer, pancreatic cancer, ovarian cancer and head-and-neck squamous cell carcinomas .
- PLK1 has also been linked to cancer-specific functions associated with PI3K and MAPK pathways as well as with activation of oncogenic chimeric transcription factors (PAX3-FOX01) .
- the present invention relates thus to a compound of following Formula (I) :
- A is N or C-H
- Y is 0, S or one of the following groups : sulfoxide (S(0)), sulfone (S(0) 2 ) or N(R 3 ) with R 3 being H, or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl;
- Ri is H or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl;
- Ri and R 3 may optionally be together included into any optionally substituted 4 to 7-membered heterocycle or 8 to 10- membered heterobicycle;
- R 2 is any of the optionally substituted following groups: alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl, heteroaryl-alkyl , heterocycle-alkyl or heteroaryl, preferably alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl .
- pharmaceutically acceptable means that which is used for preparing a pharmaceutical composition that is generally safe, nontoxic and neither biologically nor otherwise undesirable and that is acceptable for veterinary use as well as for human pharmaceutical use.
- salts that are pharmaceutically acceptable, as defined herein, and that have the desired pharmacological activity of the parent compound.
- Such salts include :
- inorganic acids such as hydrochloric acid, hydrobromic acid, sulphuric acid, nitric acid, phosphoric acid and similar; or formed with organic acids such as acetic acid, benzenesulfonic acid, benzoic acid, camphorsulfonic acid, citric acid, ethanesulfonic acid, fumaric acid, glucoheptonic acid, gluconic acid, glutamic acid, glycolic acid, hydroxynaphthoic acid, 2-hydroxyethanesulfonic acid, lactic acid, maleic acid, malic acid, mandelic acid, methanesulfonic acid, muconic acid, 2-naphthalenesulfonic acid, propionic acid, salicylic acid, succinic acid, dibenzoyl-L-tartaric acid, tartaric acid, p- toluenesulfonic acid, trimethylacetic acid, trifluoroacetic acid and similar; and
- salts formed when an acid proton present in the parent compound is either replaced by a metal ion, for example an alkaline metal ion (Na + , K + or Li + for example) , an alkaline-earth metal ion (such as Ca 2+ or Mg 2+ ) or an aluminium ion; or coordinates with an organic or inorganic base.
- a metal ion for example an alkaline metal ion (Na + , K + or Li + for example) , an alkaline-earth metal ion (such as Ca 2+ or Mg 2+ ) or an aluminium ion; or coordinates with an organic or inorganic base.
- Acceptable organic bases include diethanolamine, ethanolamine, N-methylglucamine, triethanolamine, tromethamine and similar.
- Acceptable inorganic bases include aluminium hydroxide, calcium hydroxide, potassium hydroxide, sodium carbonate and sodium hydroxide.
- halogen means fluorine (F) , bromine (Br) , chlorine (CI) , or iodine (I) .
- alkyl refers to a straight or branched saturated hydrocarbon chain containing preferably from 1 to 6 carbon atoms (i.e. (Ci-C 6 ) alkyl ) , including, but not limited to, methyl, ethyl, n-propyl, iso- propyl, n-butyl, iso-butyl, sec-butyl, tert-butyl, n-pentyl, n- hexyl, and the like.
- alkenyl refers to a straight or branched unsaturated hydrocarbon chain containing preferably from 2 to 6 carbon atoms, and comprising at least one double bond, notably one double bond, including, but not limited to, ethenyl, propenyl, butenyl, pentenyl, hexenyl and the like.
- alkynyl refers to a straight or branched unsaturated hydrocarbon chain, containing preferably from 2 to 6 carbon atoms, and comprising at least one triple bond, notably one triple bond, including, but not limited to, ethynyl, propynyl, butynyl, pentynyl, hexynyl and the like.
- cycloalkyl relates to a saturated hydrocarbon monocycle or bicycle containing preferably from 3 to 10 carbon atoms, advantageously to a saturated hydrocarbon monocycle containing from 3 to 8, notably 3, 5 or 6 carbon atoms including, but not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, decaline, norbornane, bicyclo [ 3.2.1 ] octane and the like.
- the two cycles can be fused, bridged or have a spiro configuration.
- the term "cycloalkyl" refers to cyclopropyl, cyclopentyl or cyclohexyl.
- aryl refers to an aromatic hydrocarbon group comprising preferably 6 to 10 carbon atoms and comprising one or more fused rings, such as, for example, a phenyl or naphtyl group.
- fused rings such as, for example, a phenyl or naphtyl group.
- it will be a phenyl group.
- aralkyl or "aryl-alkyl” , as used in the present invention, refers to an aryl group as defined above bound to the molecule via a alkyl group as defined above, preferably via a (Ci-C 6 ) alkyl group, in particular via a (Ci-C 2 ) alkyl group.
- heterocycle refers to a saturated, unsaturated or aromatic hydrocarbon monocycle or bicycle, in which one or more, in particular one to three, preferably one or two, carbon atoms have been replaced with a heteroatom selected from 0, N and S, preferably selected from 0 and N.
- each cycle is a 3- to 7-membered, preferably 5- to 6-membered, cycle.
- the two cycles can be fused, bridged or have a spiro configuration.
- heterocycle could be used then to refer to a heterocycle comprising 2 cycles.
- the term "4 to 7-membered heterocycle”, as used in the present invention, refers to a heterocycle as defined above comprising one cycle having 4 to 7 members, such as a pyrrole or a pyrazole.
- the term “8 to 10-membered heterobicycle”, as used in the present invention refers to a heterocycle as defined above comprising two cycles having 8 to 10 members, such as a benzimidazole or a benzotriazole .
- heteroaryl refers to an aromatic heterocycle as defined above.
- the heteroaryl is an aromatic hydrocarbon monocycle or bicycle (i.e. comprising two fused rings), each cycle having 5 or 6 members, notably 6 members, and 1 to 4, notably 1 or 2, carbon atoms having each been replaced with a nitrogen, a oxygen or a sulfur atom, preferably a nitrogen or oxygen atom, notably a nitrogen atom.
- a heteroaryl can be notably thiophene, furan, pyrrole, imidazole, pyrazole, oxazole, isoxazole, thiazole, isothiazole, triazoles ( 1 , 2 , 3-triazole and 1 , 2 , 4-triazole ) , benzofuran, indole, azaindole, benzothiophene , benzimidazole, benzotriazole , benzoxazole, benzisoxazole , benzothiazole, benzisothiazole, pyridine, pyrimidine, pyridazine, pyrazine, triazine, quinoline, isoquinoline, quinoxaline, quinazoline, etc.
- it will be a pyridine, an indole, a furan, a thiophene, an azaindole notably a 7-azaindole or an indazole, a benzimidazole, or a benzothiophene.
- heterocycle-alkyl refers to a heterocycle group as defined above bound to the molecule via an alkyl group as defined above, preferably via a ( Ci- C 6 ) alkyl group, in particular via a ( Ci-C 2 ) alkyl group.
- heteroaryl-alkyl refers to a heteroaryl group as defined above bound to the molecule via an alkyl group as defined above, preferably via a ( Ci- C 6 ) alkyl group, in particular via a ( Ci- C 2 ) alkyl group.
- acyl refers to a group of formula -C (0) R, wherein R is an alkyl group, preferably a ( Ci- C 6 ) alkyl group.
- Y, R x and R 3 may optionally be together included into any optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle” means that when Y is NR 3 , R x and R 3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle.
- the heterocycle or heterobicycle thus comprises at least one nitrogen atom which carries the group R 3 .
- Y is S, 0 or a sulfone group (-S(0) 2 -) . More particularly, Y is S.
- Y is N(R 3 ) and Ri and R 3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle, preferably into a 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, in particular a 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, more particularly a heterobicycle selected from the group consisting of benzotriazole and benzimidazole .
- R x is an optionally substituted aryl or heteroaryl .
- Ri is an optionally substituted phenyl or pyridyl.
- Ri is an optionally substituted phenyl or an unsubstituted pyridyl.
- Ri is an aryl or heteroaryl, in particular a pyridyl or phenyl, notably a phenyl, optionally substituted with one or two substituent ( s ) selected from the group consisting of: halogen, nitro (N0 2 ) , R 4 , -0R 5 , -NR 7 R 8 , -C ( 0 ) R 9 , -C(O)ORi 0 , C(0) RiiRi 2 , -0C(0) Ri 3 , -NR i C (0) R15, and -NRi 6 C (0 ) Ri 7 Ri 8 ; R 4 to R 23 being, independently of one another, a hydrogen atom or one of the following groups: (Ci-C 6 ) alkyl , cycloalkyl, aryl, aralkyl, heterocycle, and heterocycle-alkyl, notably (Ci-C 6 ) alkyl , cycloalkyl, heterocycle,
- Ri is an aryl or heteroaryl, in particular a pyridyl or phenyl, notably a phenyl, optionally substituted with one or two substituent ( s ) selected from the group consisting of: halogen, nitro (N0 2 ) , R 4 , -OR 5 , -NR 7 R 8 , - C ( 0 ) RnRi 2 , -NR 14 C ( 0 ) R i 5 , and -NRi 6 C ( 0 ) Ri 7 Ri 8 ; R 4 to R 23 being, independently of one another, a hydrogen atom or one of the following groups: ( Ci-C 6 ) alkyl , cycloalkyl, aryl, aralkyl, heterocycle, and heterocycle-alkyl, notably ( Ci-C 6 ) alkyl, cycloalkyl, heterocycle, and heterocycle- alkyl, said group being optionally substituted with one or more, preferably one to
- the aryl of the aryl and aralkyl groups is advantageously a phenyl or naphtyl, in aprticular a phenyl.
- the cycloalkyl comprises advantageously 3 to 6 carbon atoms, such as a cyclopropyl, a cyclopentyl or a cyclohexyl, in particular a cyclopropyl or a cyclohexyl .
- the heterocycle of the heterocycle and heterocycle-alkyl groups is advantageously a saturated heterocycle. It will advantageously contain at least one, notable one to three, such as one or two, nitrogen atoms and optionally one or two, notably one oxygen atoms. It can be a saturated 4- to 7-, such as 5- or 6-membered heterocycle comprising one cycle and comprising one to three, such as one or two nitrogen atoms and optionally one or two, notably one oxygen atoms. It can be a pyrrolidine, piperidine, piperazine or morpholine cycle, such as a pyrrolidine, piperidine, or piperazine cycle, in particular a pyrrolidine or piperidine cycle .
- R 2 is a group selected from (d-C 6 ) alkyl, aryl- (d-C 6 ) alkyl, aryl, heteroaryl, heterocycle- (Ci-C 6 ) alkyl or heteroaryl- (d-C 6 ) alkyl , in particular aryl- (d ⁇ d) alkyl, aryl, heteroaryl, heterocycle- (d ⁇ d) alkyl or heteroaryl- (d ⁇ d) alkyl, such as aryl- (d _ d) alkyl, aryl, heteroaryl, heterocycle- (d _ d) alkyl or heteroaryl- (d _ d) alkyl, more particularly aryl- (d ⁇ d) alkyl or heteroaryl- (d ⁇ d) alkyl , such as aryl- (d _ d) alkyl or heteroaryl- (d _ d) alkyl , wherein the aryl (in the
- R 2 is a group selected from aryl- (d-C 6 ) alkyl , aryl, heteroaryl, heterocycle- (Ci-C 6 ) alkyl or heteroaryl- (d-C 6 ) alkyl , such as aryl- (Ci-C 2 ) alkyl , aryl, heteroaryl, heterocycle- (Ci-C 2 ) alkyl or heteroaryl- (d- d) alkyl, more particularly aryl- (d _ d) alkyl or heteroaryl- (d _ d) alkyl, such as aryl- (d _ d) alkyl or heteroaryl- (d _ d) alkyl , wherein the aryl (in the aryl and aryl-alkyl groups) is a phenyl or a naphtyl; and the heterocycle and heteroaryl (in the heterocycle, heterocycle-alkyl, heteroaryl, heteroaryl, heteroaryl
- R 27 to R 46 being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C 6 ) alkyl , aralkyl, and heterocycle-alkyl , said groups being optionally substituted with one or more, preferably one to five, notably one to two, halogen, (d-C 6 ) alkyl , NR 47 R 48 or OR 49 , R 47 to R 49 being an hydrogen atom or
- R 2 is a group selected from aryl- (d-C 6 ) alkyl , aryl, heteroaryl, heterocycle- (Ci-C 6 ) alkyl or heteroaryl- (d-C 6 ) alkyl , such as aryl- (Ci-C 2 ) alkyl , aryl, heteroaryl, heterocycle- (Ci-C 2 ) alkyl or heteroaryl- (d _ d) alkyl, more particularly aryl- (d _ d) alkyl or heteroaryl- (d _ d) alkyl, such as aryl- (d _ d) alkyl or heteroaryl- (d _ d) alkyl , wherein the aryl (in the aryl and aryl-alkyl groups) is a phenyl or a naphtyl; and the heterocycle and heteroaryl (in the heterocycle, heterocycle-alkyl, heteroaryl, heteroaryl, heteroary
- the present invention relates to a compound of formula (I) wherein:
- ⁇ A is N or C-H, preferably N;
- Y is S, 0 or a sulfone group (-S(0) 2 -), preferably S;
- Ri is a phenyl, optionally substituted with one or two, substituent ( s ) , preferably selected from the group consisting of: halogen, nitro (N0 2 ) , R , -0R 5 , -NR 7 R 8 , -C (0) RnRi 2 , -NR i4 C (0) R i5 , and -NRi 6 C (0) Ri 7 Ri 8 ; R to Ri 8 being, independently of one another, an hydrogen atom or one of the following groups: a (Ci-C 6 ) alkyl , cycloalkyl, heterocycle, and heterocycle-alkyl, said group being optionally substituted with one to three, halogen, (d-C 6 ) alkyl , NR 2 R 2 5 or OR 26 , 24 to R 26 being an hydrogen atom or a (Ci-C 6 ) alkyl .
- substituent ( s ) preferably selected from the group consisting of: halogen,
- the present invention relates to a compound of formula (I) wherein:
- A is N or C-H, preferably N
- Y is S, 0 or a sulfone group (-S(0) 2 -), preferably S;
- ⁇ Ri is a phenyl, optionally substituted with a -C(0)NRnRi 2 group, R 11 and R i2 being an hydrogen atom or a (Ci-C 6 ) alkyl , preferably an hydrogen atom.
- the present invention relates to a compound of formula (I) wherein:
- A is N or CH, preferably N;
- Ri and R 3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle, preferably into a 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, in particular a a 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, more particularly a heterobicycle selected from the group consisting of benzotriazole and benzimidazole.
- the present invention relates to a compound of formula (I) wherein:
- Ri is phenyl optionally substituted by one of the following groups: hydroxy, hydroxy-alkyl more particularly hydroxymethyl , In the above definitions of Ri :
- the alkyl group is preferably a (Ci-C 6 )alkyl group, in particular a (C 1 -C3) alkyl group;
- the aryl group in the aryl and aralkyl groups is preferably a phenyl group
- heteroaryl in the heteroaryl and heteroaryl-alkyl groups is preferably a pyridyl group
- the cycloalkyl group is preferably a (C 3 -C 6 ) cycloalkyl group, in particular a cyclopropyl, cyclopentyl or a cyclohexyl, such as a cyclopropyl or a cyclohexyl; and
- the heterocycle group in the heterocycle and heterocycle-alkyl groups is preferably a saturated heterocycle containing at least one, notable one to three, such as one or two, nitrogen atoms and optionally one or two, notably one oxygen atoms. It can be a saturated 4- to 7-, such as 5- or 6-membered heterocycle comprising one cycle and comprising one to three, such as one or two nitrogen atoms and optionally one or two, notably one oxygen atoms. It can be a pyrrolidine, piperidine, piperazine or morpholine cycle, such as a pyrrolidine, piperidine, or piperazine cycle, in particular a pyrrolidine or piperidine cycle.
- R 2 In the above definitions of R 2 :
- the alkyl group is preferably a (Ci-C 6 )alkyl group, in particular a (Ci-C 4 ) alkyl group, more particularly a (Ci ⁇ C 2 ) alkyl group;
- the aryl group in the aryl and aralkyl groups is preferably a phenyl or a naphtyl
- the heteroaryl group in the heteroaryl and heteroaryl-alkyl groups is preferably a heteroaryl comprising one or two fused cycles comprising one to three, notably one or two, heteroatoms selected from 0, S and N, such as a thiophene, a furane, a pyrrole, a pyrazole, an imidazole, an oxazole, an isoxazole, a thiazole, an isothiazole, a triazole, a pyridine, an indole, an azaindole (such as an indazole or a 7-azainole) , an indoline, a benzofurane or a benzothiophene, in particular a thiophene, a furane, a pyridine, an indole, an indazole, a 7-azainole, an indoline or a benzothiophene, for example an indole
- the heterocycle group in the heterocycle and heterocycle-alkyl groups is preferably a saturated 4- to 7-, notably 5- or 6- membered heterocycle comprising one to three, notably one or two heteroatoms chosen from 0, N and S, notably chosen from N and 0, such as a pyrrolidine, piperidine, piperazine or morpholine cycle, in particular a piperazine or morpholine cycle .
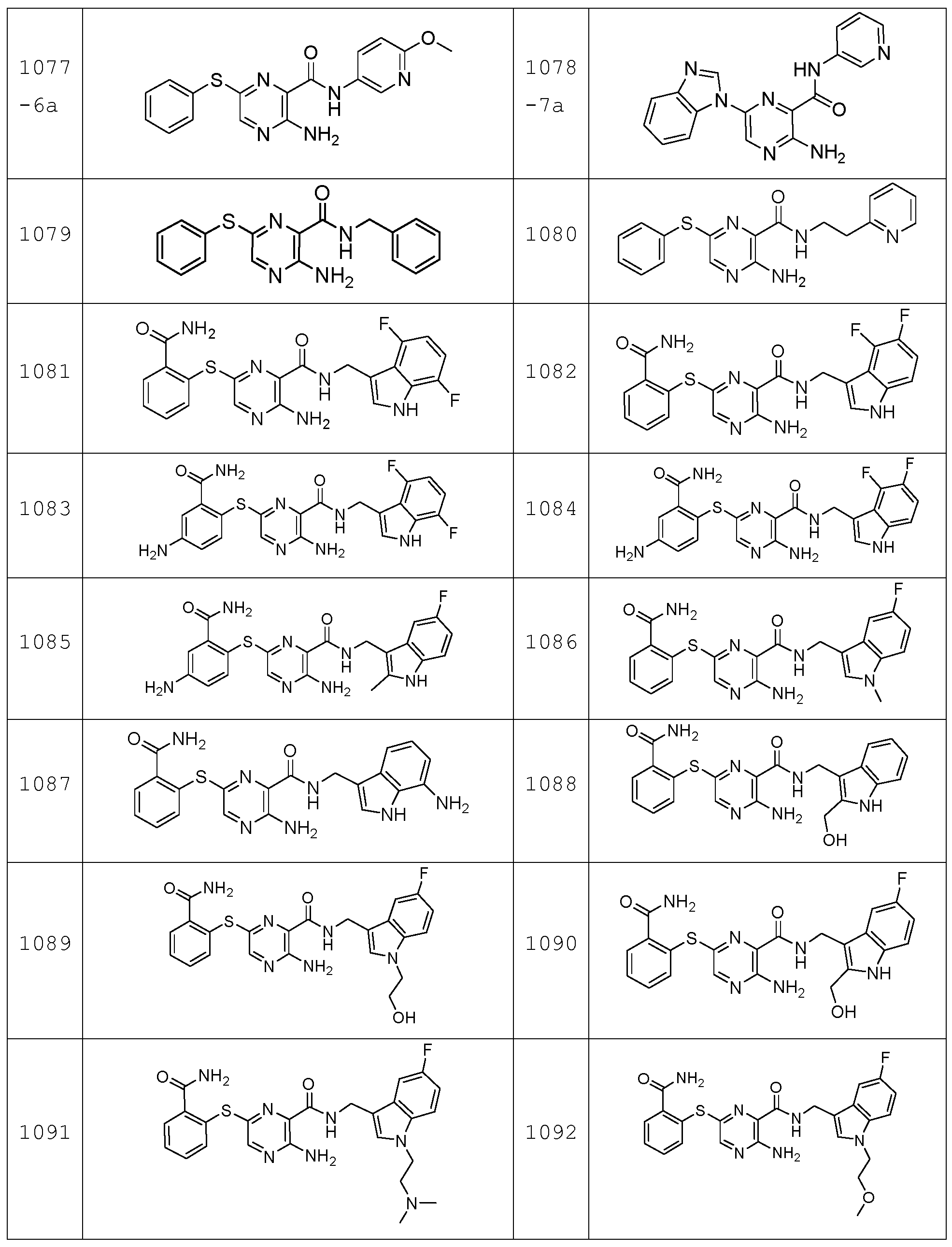
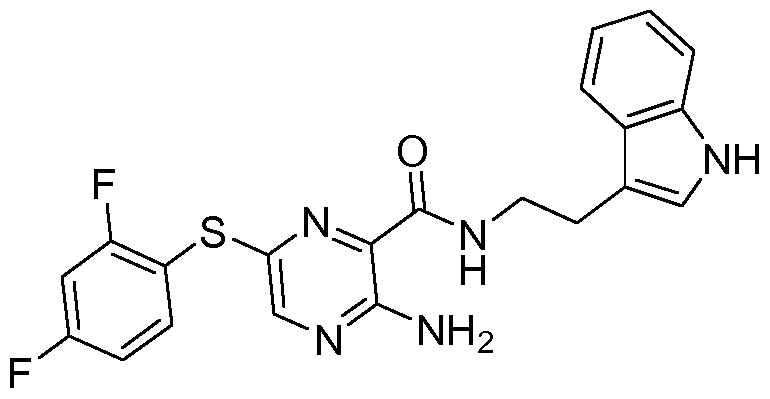
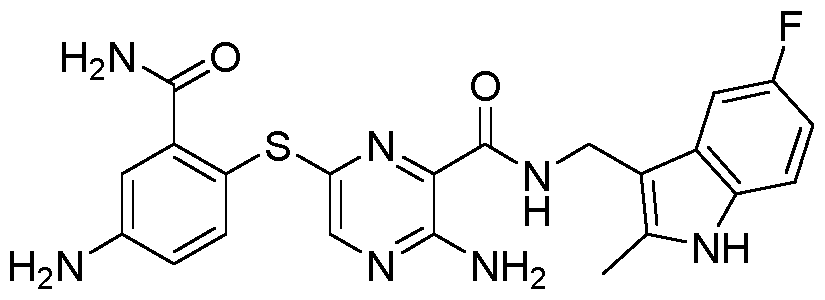
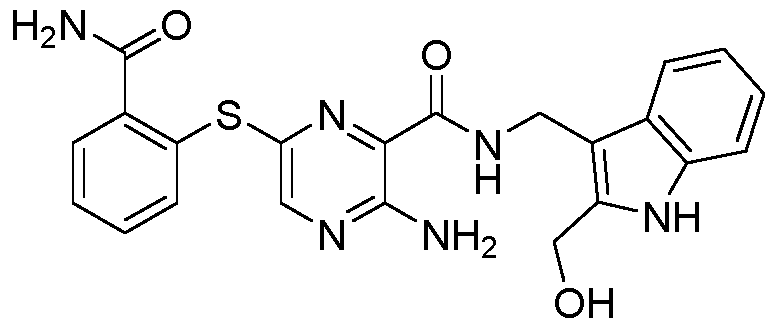
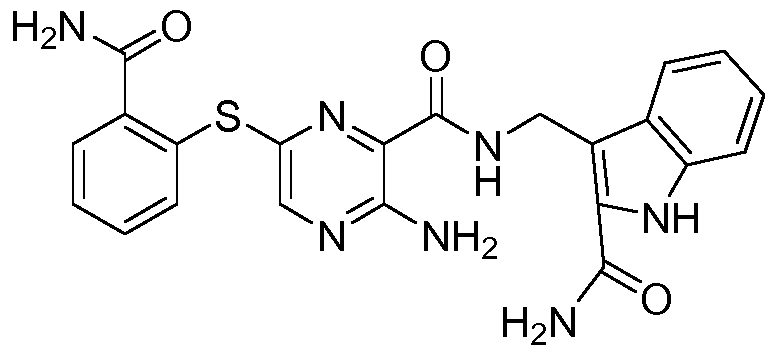
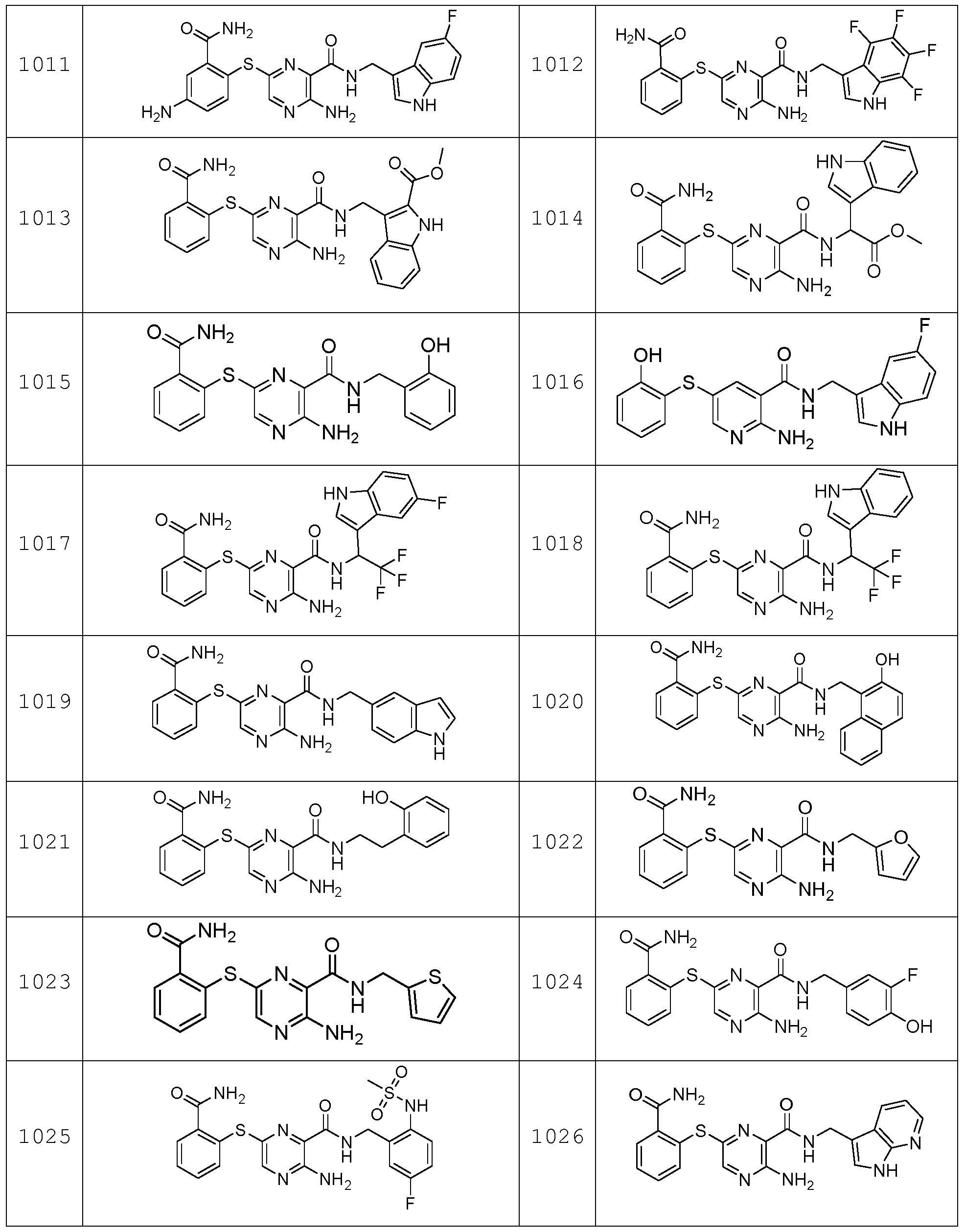
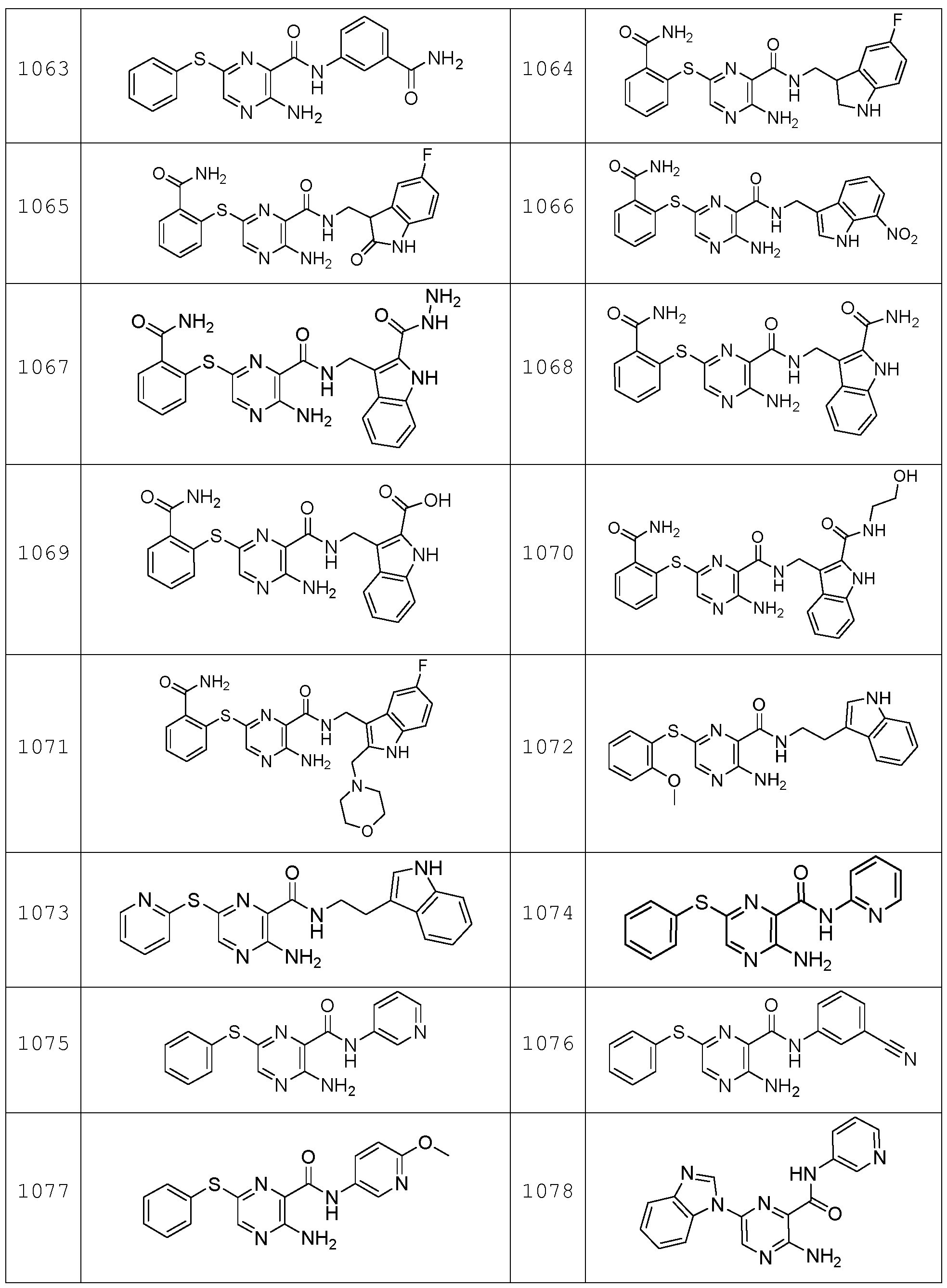
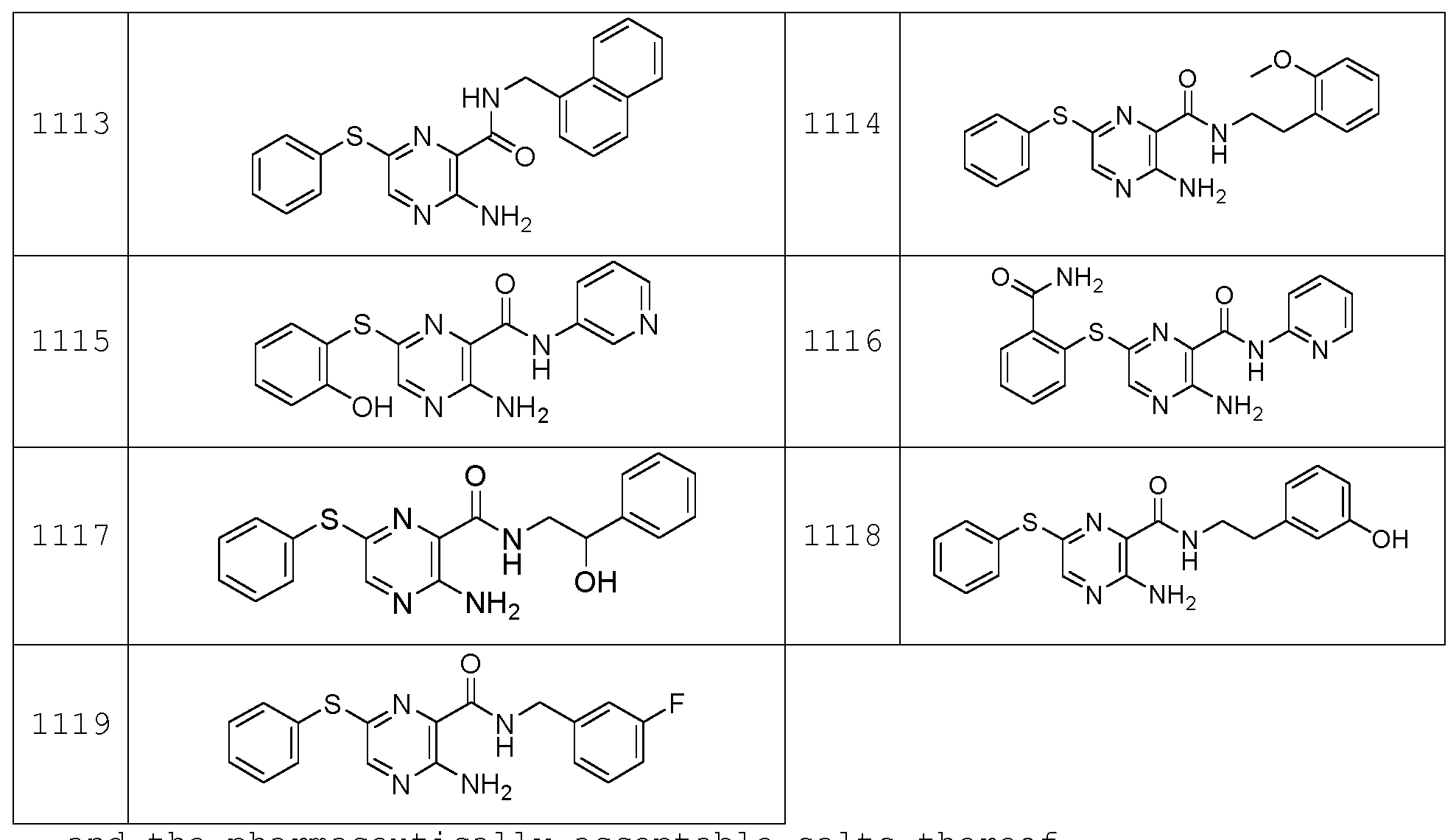
- the compound according to the present invention can be in particular selected from the compounds 1001 to 1119 of the examples below and the pharmaceutically acceptable salts thereof.
- the present invention also relates to a compound of formula (I) as defined above for use as a drug, in particular as PLK-1 inhibitors, notably for treating cancer.
- the invention also relates to the use of a compound of the invention for manufacturing a drug, notably useful as PLK-1 inhibitors, in particular intended for treating cancer.
- the invention further relates to a method for treating cancer, comprising the administration of an effective quantity of at least one compound of formula (I) as defined above to a patient in need thereof.
- the cancer can be more particularly in this case a colon cancer, breast cancer, kidney cancer, liver cancer, pancreas cancer, prostate cancer, lung cancer, ovarian cancer, head and neck cancer, glioblastoma, neuroblastoma, lymphoma, leukaemia, inflammatory myofibroblastic tumour, myelodysplastic syndrome, or myelofibrosis .
- the present invention also relates to a pharmaceutical composition
- a pharmaceutical composition comprising at least one compound according to the invention and at least one pharmaceutically acceptable carrier.
- the compound of the invention can be administered by oral, sublingual, parenteral, subcutaneous, intramuscular, intravenous, transdermal, local or rectal route, preferably by oral, intravenous or subcutaneous route.
- the active ingredient can be administered in unit dosage forms, in mixture with traditional pharmaceutical carriers, to animals or humans .
- Suitable unit dosage forms comprise forms for oral route such as tablets, gelatine capsules, powders, granules and oral solutions or suspensions; sublingual and oral administration forms; parenteral, subcutaneous, intramuscular, intravenous, intranasal or intraocular administration forms; and forms for rectal administration.
- the main active ingredient is mixed with a pharmaceutical vehicle such as gelatin, starch, lactose, magnesium stearate, talc, gum arabic and the like.
- a pharmaceutical vehicle such as gelatin, starch, lactose, magnesium stearate, talc, gum arabic and the like.
- the tablets may be coated with sucrose or with other suitable materials, or they may be treated in such a way that they have a prolonged or delayed activity and they continuously release a predetermined amount of active principle.
- a preparation in gelatin capsules can be obtained by mixing the active ingredient with a diluent and pouring the mixture obtained into soft or hard gelatin capsules.
- a preparation in the form of a syrup or an elixir may contain the active ingredient together with a sweetener, an antiseptic, a taste enhancer or a suitable coloring agent.
- the water-dispersible powders or granules may contain the active ingredient mixed with dispersing agents, wetting agents, or suspending agents, and with flavor correctors or sweeteners.
- aqueous suspensions, isotonic saline solutions or sterile and injectable solutions which contain pharmacologically compatible dispersing agents and/or wetting agents can be used.
- the active principle may also be formulated in the form of microcapsules, optionally with one or more carrier additives.
- the compounds of the invention can be used in a pharmaceutical composition at a dose ranging from 0.01 mg to 1000 mg a day, administered in only one dose once a day or in several doses along the day, for example twice a day.
- the daily administered dose is advantageously comprises between 5 mg and 500 mg, and more advantageously between 10 mg and 200 mg. However, it can be necessary to use doses out of these ranges, which could be noticed by the person skilled in the art.
- compositions according to the present invention can further comprise at least another active principle, such as an anticancer agent.
- the present invention also relates to a pharmaceutical composition
- a pharmaceutical composition comprising:
- the present invention also relates to a pharmaceutical composition as defined previously for use as a drug, in particular as PLK-1 inhibitors, notably for treating cancer.
- the present invention also relates to a method for treating cancer comprising the administration to a person in need thereof of an effective amount of a pharmaceutical composition according to the invention.
- the present invention relates also to the processes to prepare the compound according to the present invention.
- the present invention concerns thus a first process to prepare a compound according to the present invention comprising the following steps:
- step (bl) optionally salifying the compound of formula (I) obtained in step (al) to give a pharmaceutically acceptable salt of a compound of formula (I) .
- activated form of COOH is meant a COOH group in which the OH moiety has been replaced with an activated leaving group (LG) enabling coupling of the activated COOH group with an amino group by formation of an amide bond and release of the compound LG-H.
- Activated forms may be activated esters, activated amides, anhydrides or acyl halides such as acyl chlorides.
- Activated esters include derivatives formed by reaction of the COOH group with N-hydroxybenzotriazole or N-hydroxysuccinimide . It can be in particular COC1.
- step (al) can be performed by a peptide coupling .
- the peptide coupling will be advantageously carried out in the presence of a coupling agent, such as diisopropylcarbodiimide (DIC) , dicyclohexylcarbodiimide (DCC) , 1- (3-dimethylaminopropyl ) - 3-ethylcarbodiimide hydrochloride (EDC) , carbonyldiimidazole (CDI), hexafluorophosphate 2- ( lH-benzotriazole-l-yl ) -1 , 1 , 3 , 3- tetramethyluronium (HBTU) , tetrafluoroborate 2- ( lH-benzotriazole- 1-yl) -1, 1, 3, 3-tetramethyluronium (TBTU) , hexafluorophosphate 0- (7-azobenzotriazol-l-yl) -1,1,3, 3-tetramethyluronium (HATU) , (benzotriazol-l
- This reaction can be carried out for example in dimethylformamide (DMF) as solvent.
- DMF dimethylformamide
- reaction can be performed in the presence of a base in conditions well-known to the one skilled in the art.
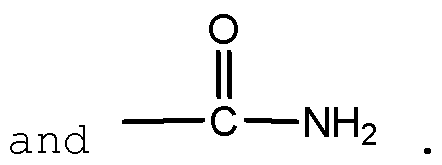
- X 3 CO- (Ci-C 6 ) alkyl, such as COMe or COEt, notably COMe, and A, Y and Ri are as defined previously.
- the hydrolysis can be performed in acidic conditions or basic conditions (saponification) . It can be performed notably according to Heterocycles , 2005, vol. 65(10), 2321-2327 or US5252538.
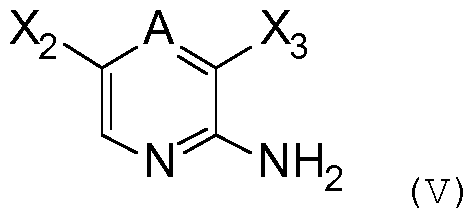
- the compound of formula (IV) can be prepared by coupling of a compound of formula RiYH with Y and Ri as defined above and a compound of following formula
- the salification step can be carried out by methods well known to the one skilled in the art, in particular by reaction of the compound of formula (I) obtained in step (a) with a pharmaceutically acceptable acid (organic or inorganic acid) , base (organic or inorganic base) or solvent, as defined previously .
- a pharmaceutically acceptable acid organic or inorganic acid
- base organic or inorganic base
- solvent solvent
- a and R 2 are as defined previously, and X 2 represents a halogen atom, such as CI, Br or I,
- step (b2) optionally salifying the compound of formula (I) obtained in step (a2) to give a pharmaceutically acceptable salt of a compound of formula (I) .
- Step (a2) The coupling reaction of step (a2) can be performed notably in the presence of Cul .
- the compound of formula (III) can be prepared by coupling of a compound of formula R 2 NH 2 and a compound of following formula (VI) :
- Such a coupling reaction can be carried out in the conditions detailed in step (al) above.
- Step (b2) cf. step (bl)
- Protection / deprotection steps or functionalization steps can be carried out in the processes described above, such steps and their reaction conditions being well known to the one skilled in the art.
- the compound obtained can be separated from the reaction medium by methods well known to the person skilled in the art, such as by extraction, evaporation of the solvent or by precipitation or crystallisation (followed by filtration) .
- the compound can also be purified if necessary by methods well known to the person skilled in the art, such as by recrystallisation, by distillation, by chromatography on a column of silica gel or by high performance liquid chromatography (HPLC) .
- methods well known to the person skilled in the art such as by recrystallisation, by distillation, by chromatography on a column of silica gel or by high performance liquid chromatography (HPLC) .
- the main final product may either be an ester (-3a-z-) or an acid (-4a-z-) derivative.
- esterification of acids to the corresponding methyl or ethyl esters were conducted using standard well-known procedures (ex.: Heterocycles, 2005, vol. 65(10), 2321-2327; US5252538) .
- reaction mixture is agitated for a two hours period while the temperature is progressively allowed to rise to ambient and then to 50°C.
- Reaction crude is then poured into 25 mL of dichloromethane and successively washed with NaHC0 3 saturated water, water and brine. The combined organic layers are dried over Na 2 S0 4 , evaporated to dryness and the crude purified by preparative reverse-phase HPLC (yield: 74%) .
- Molecules are designated by an incremented four digits serial number followed, if available, by a 1 digit & 1-2 letters code referencing the synthetic methodology used for its synthesis .
- Substrate coated 96- ell microplates [Packard #6005182] are generated by addition of 100 ⁇ per well of a lmg/ml phosphate buffer saline (PBS) solution of dephosphorylated casein [Sigma # C-4032] for two hours under mild agitation. Casein solution is then replaced with blocking solution comprising 5% bovine serum albumin (BSA) [Sigma #B-7906] in PBS buffer and placed two hours under mild agitation.
- PBS lmg/ml phosphate buffer saline
- BSA bovine serum albumin
- Compound is added to the desired final concentration (typical range between 30 ⁇ and 10 nM) and the enzymatic reaction is carried out for 30 minutes at 30°C under the following condition: Tris 20mM pH 7.5; MgCl 2 lOmM; dithiothreitol (DTT) ImM ; ⁇ -glycerophosphate 40mM; sodium orthovanadate ImM; glycerol 2%; adenosine 5 ' -triphosphate (ATP) [Sigma #A-2383] 20 ⁇ and 0.5 ⁇ 1 of purified PLK1 in a final volume of ⁇ per well.
- DTT dithiothreitol
- ⁇ -glycerophosphate 40mM sodium orthovanadate ImM
- glycerol 2% adenosine 5 ' -triphosphate (ATP) [Sigma #A-2383] 20 ⁇ and 0.5 ⁇ 1 of purified PLK1 in a final volume of ⁇ per well.
- the level of PLKl-dependent casein phosphorylation is detected by sequential addition of ⁇ of anti-Phospho-Threonine antibody [Cell Signaling #9381] and Horseradish Peroxidase (HRP) conjugated anti-rabbit antibody [Amersham #NA934V] intertwined with washing steps in PBS tween-20 0.1%. Both antibodies are diluted to 1/1000 in 5% BSA PBS solution and left for incubation at room temperature for 1 hour. The reaction is finally quantified by addition of chemiluminescent HRP substrate SuperSignal ELISA Pico according to manufacturer's instruction [Pierce #37070] and detected with Envision plate reader [Perkin Elmer] .
- A549 human lung adenocarcinoma epithelial cell line
- HCT116 human colon cancer cell lines
- Namalwa human Burkitt lymphoma cell lines
- Adherent cells (either from A549 or HCT116) and nonadherent cells (Namalwa cell lines) are transferred into 96-well plates (10 000 cells/ml for A549, 15 000 cells/ml for HCT116 and 300,000 cells/ml for Namalwa) at day 0 for A549 and HCT116, and day 1 for Namalwa, at a concentration compatible with logarithmic growth for the 72 hours required for the evaluation of the compounds. All of the cells are treated with the assayed molecules at day 1 and then placed in an incubator at 37 °C under an atmosphere of 5% C0 2 . Cell viability is evaluated at day 4 by assaying released ATP, which is characteristic of viable cells.
- ATPLite induces a luminescent signal read by TopCount microplate reader.
- IC50s are determined by nonlinear regression on the basis of a sigmoidal dose/response relationship model, wherein the Hill coefficient is left variable, carried out on the GraphPad (Prism) software package according to the algorithm provided . d) Results
- Potencies are designated by IC 50 binned into four activity classes as follows:
- D IC 50 > 10 ⁇
- C 10 ⁇ > IC 50 > ⁇
- B ⁇ > IC 50 > ⁇
- A IC 50 ⁇ ⁇ ; not conducted.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
The present invention concerns 2-amino-pyridin-3-yl carboxamide and 3-amino-pyrazin-2-yl carboxamide derivatives of formula (I): (I) or a pharmaceutically acceptable salt thereof, wherein: • A is N or C-H, • Y is 0, S or one of the following groups : sulfoxide (-S(O)-), sulfone (- S(0)2-) or N (R3) with R3being H, or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl, • R1 is H or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl, or when Y is NR3, R1 and R3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle, and • R2 is any of the optionally substituted following groups: alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl, heteroaryl-alkyl or heteroaryl; for use as polo-like kinase 2 inhibitors for the treatment of cancer; as well as pharmaceutical compositions containing such a compound and processes to prepare such a compound.
Description
3-AMINO-PYRAZIN-2-YL CARBOXAMIDE AND 2-AMINO-PYRIDIN-3-YL CARBOXAMIDE DERIVATIVES AS POLO-LIKE KINASE 1 (PLK-1) INHIBITORS FOR THE TREATMENT OF CANCER
The present invention relates to novel amino-pyridine and amino-pyrazine derivatives, their methods of preparation, pharmaceutical compositions containing the same and their use as a drug, in particular as PLK-1 inhibitors, especially for treating cancer.
Protein Kinases form a class of enzymes which transferring phosphate groups to substrates, other protein kinases or even themselves put them at the core of most signalling pathways. They are numerous as wild type isoforms as well as disease-causing mutated variants. Their dysregulated expression either through mutation, overexpression or amplification is often a potent driver to carcinogenesis and, as such, qualifies them as highly relevant targets to fight against neoplasia.
Despite the enormous investment of the pharmaceutical industry into this class of targets since 1990, only a small fraction of the kinome is successfully addressed by a marketed inhibitor yet- leaving many protein kinases undrugged.
Polo-like Kinase 1 (PLK1) is a still undrugged serine/threonine protein kinase whose many functions qualify it as highly relevant to fight tumorigenesis.
Among the many functions of PLK1 which continue to be documented at a steady pace, its known key role as regulator of mitosis is particularly attractive to justify the pursuit of a drug development program against it.
Although four variants of Polo-Like Kinases are known in humans (PLK1-4), only PLK1 inhibition by a small molecule is documented to be sufficient to trigger a full G2/M cell cycle block, induction of apoptosis into cancerous cell line and tumor shrinkage in xenografted tumor models.
Moreover, overexpression of PLK1 has been described in several neoplastic conditions such as breast cancer, colorectal cancer, endometrial carcinoma, non small cell lung cancer,
pancreatic cancer, ovarian cancer and head-and-neck squamous cell carcinomas .
Beyond its role in mitotic control, relevant but not exclusive to highly proliferating cancer cells, PLK1 has also been linked to cancer-specific functions associated with PI3K and MAPK pathways as well as with activation of oncogenic chimeric transcription factors (PAX3-FOX01) .
There is thus a need for new inhibitors of PLK-1.
The present invention relates thus to a compound of following Formula (I) :
or a pharmaceutically acceptable salt thereof, wherein:
• A is N or C-H;
• Y is 0, S or one of the following groups : sulfoxide (S(0)), sulfone (S(0)2) or N(R3) with R3 being H, or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl;
• Ri is H or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl;
• Y, Ri and R3 may optionally be together included into any optionally substituted 4 to 7-membered heterocycle or 8 to 10- membered heterobicycle; and
• R2 is any of the optionally substituted following groups: alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl, heteroaryl-alkyl , heterocycle-alkyl or heteroaryl, preferably alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl .
In the present invention, "pharmaceutically acceptable"
means that which is used for preparing a pharmaceutical composition that is generally safe, nontoxic and neither biologically nor otherwise undesirable and that is acceptable for veterinary use as well as for human pharmaceutical use.
In the present invention, "pharmaceutically acceptable salts" of a compound means salts that are pharmaceutically acceptable, as defined herein, and that have the desired pharmacological activity of the parent compound. Such salts include :
1) hydrates and solvates,
2) acid addition salts formed with inorganic acids such as hydrochloric acid, hydrobromic acid, sulphuric acid, nitric acid, phosphoric acid and similar; or formed with organic acids such as acetic acid, benzenesulfonic acid, benzoic acid, camphorsulfonic acid, citric acid, ethanesulfonic acid, fumaric acid, glucoheptonic acid, gluconic acid, glutamic acid, glycolic acid, hydroxynaphthoic acid, 2-hydroxyethanesulfonic acid, lactic acid, maleic acid, malic acid, mandelic acid, methanesulfonic acid, muconic acid, 2-naphthalenesulfonic acid, propionic acid, salicylic acid, succinic acid, dibenzoyl-L-tartaric acid, tartaric acid, p- toluenesulfonic acid, trimethylacetic acid, trifluoroacetic acid and similar; and
3) salts formed when an acid proton present in the parent compound is either replaced by a metal ion, for example an alkaline metal ion (Na+, K+ or Li+ for example) , an alkaline-earth metal ion (such as Ca2+ or Mg2+) or an aluminium ion; or coordinates with an organic or inorganic base. Acceptable organic bases include diethanolamine, ethanolamine, N-methylglucamine, triethanolamine, tromethamine and similar. Acceptable inorganic bases include aluminium hydroxide, calcium hydroxide, potassium hydroxide, sodium carbonate and
sodium hydroxide.
The term "halogen", as used in the present invention, means fluorine (F) , bromine (Br) , chlorine (CI) , or iodine (I) .
The term "alkyl", as used in the present invention, refers to a straight or branched saturated hydrocarbon chain containing preferably from 1 to 6 carbon atoms (i.e. (Ci-C6) alkyl ) , including, but not limited to, methyl, ethyl, n-propyl, iso- propyl, n-butyl, iso-butyl, sec-butyl, tert-butyl, n-pentyl, n- hexyl, and the like.
The term "alkenyl", as used in the present invention, refers to a straight or branched unsaturated hydrocarbon chain containing preferably from 2 to 6 carbon atoms, and comprising at least one double bond, notably one double bond, including, but not limited to, ethenyl, propenyl, butenyl, pentenyl, hexenyl and the like.
The term "alkynyl", as used in the present invention, refers to a straight or branched unsaturated hydrocarbon chain, containing preferably from 2 to 6 carbon atoms, and comprising at least one triple bond, notably one triple bond, including, but not limited to, ethynyl, propynyl, butynyl, pentynyl, hexynyl and the like.
The term "cycloalkyl", as used in the present invention, relates to a saturated hydrocarbon monocycle or bicycle containing preferably from 3 to 10 carbon atoms, advantageously to a saturated hydrocarbon monocycle containing from 3 to 8, notably 3, 5 or 6 carbon atoms including, but not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, decaline, norbornane, bicyclo [ 3.2.1 ] octane and the like. In the case of a bicycle, the two cycles can be fused, bridged or have a spiro configuration. Advantageously, the term "cycloalkyl", as used in the present invention, refers to cyclopropyl, cyclopentyl or cyclohexyl.
The term "aryl", as used in the present invention, refers to an aromatic hydrocarbon group comprising preferably 6 to 10
carbon atoms and comprising one or more fused rings, such as, for example, a phenyl or naphtyl group. Advantageously, it will be a phenyl group.
The term "aralkyl" or "aryl-alkyl" , as used in the present invention, refers to an aryl group as defined above bound to the molecule via a alkyl group as defined above, preferably via a (Ci-C6) alkyl group, in particular via a (Ci-C2) alkyl group.
The term "heterocycle", as used in the present invention, refers to a saturated, unsaturated or aromatic hydrocarbon monocycle or bicycle, in which one or more, in particular one to three, preferably one or two, carbon atoms have been replaced with a heteroatom selected from 0, N and S, preferably selected from 0 and N. Advantageously each cycle is a 3- to 7-membered, preferably 5- to 6-membered, cycle. In the case of a bicycle, the two cycles can be fused, bridged or have a spiro configuration. The term "heterobicycle" could be used then to refer to a heterocycle comprising 2 cycles. It includes notably the following cycles: aziridine, azetidine, oxetane, thiooxetane, pyrrolidine, pyrroline, tetrahydrofurane, dihydrofurane , tetrahydrothiophene, dihydrothiophene, piperidine, dihydropyridine, tetrahydropyridine, pyrane, dihydropyrane, tetrahydropyrane, thiopyrane, dihydrothiopyrane , tetrahydrothiopyrane, morpholine, thiomorpholine, piperazine, azepane, diazepane, imidadole, imidazoline, indoline, pyrrole, pyrazole, pyridine, pyrazine, pyridazine piperidazine, and pyrimidine .
Accordingly, the term "4 to 7-membered heterocycle", as used in the present invention, refers to a heterocycle as defined above comprising one cycle having 4 to 7 members, such as a pyrrole or a pyrazole. Similarly, the term "8 to 10-membered heterobicycle", as used in the present invention, refers to a heterocycle as defined above comprising two cycles having 8 to 10 members, such as a benzimidazole or a benzotriazole .
The term "heteroaryl" as used in the present invention
refers to an aromatic heterocycle as defined above. According to a particular embodiment, the heteroaryl is an aromatic hydrocarbon monocycle or bicycle (i.e. comprising two fused rings), each cycle having 5 or 6 members, notably 6 members, and 1 to 4, notably 1 or 2, carbon atoms having each been replaced with a nitrogen, a oxygen or a sulfur atom, preferably a nitrogen or oxygen atom, notably a nitrogen atom. A heteroaryl can be notably thiophene, furan, pyrrole, imidazole, pyrazole, oxazole, isoxazole, thiazole, isothiazole, triazoles ( 1 , 2 , 3-triazole and 1 , 2 , 4-triazole ) , benzofuran, indole, azaindole, benzothiophene , benzimidazole, benzotriazole , benzoxazole, benzisoxazole , benzothiazole, benzisothiazole, pyridine, pyrimidine, pyridazine, pyrazine, triazine, quinoline, isoquinoline, quinoxaline, quinazoline, etc. Preferably, it will be a pyridine, an indole, a furan, a thiophene, an azaindole notably a 7-azaindole or an indazole, a benzimidazole, or a benzothiophene.
The term "heterocycle-alkyl" , as used in the present invention, refers to a heterocycle group as defined above bound to the molecule via an alkyl group as defined above, preferably via a ( Ci- C6 ) alkyl group, in particular via a ( Ci-C2 ) alkyl group.
Similarly, the term "heteroaryl-alkyl" refers to a heteroaryl group as defined above bound to the molecule via an alkyl group as defined above, preferably via a ( Ci- C6 ) alkyl group, in particular via a ( Ci- C2 ) alkyl group.
The term "acyl" as used in the present invention refers to a group of formula -C (0) R, wherein R is an alkyl group, preferably a ( Ci- C6 ) alkyl group.
In the context of the invention, an "optionally substituted" group refers to a group which can be substituted with one or more, notably one to ten, in particular one to five, substituent ( s ) , preferably selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0) , R4, -0R5, -SR6, -NR7R8, -C(0)R9, - C(0) ORio , - C(0) NR R12 , -0C(0)R13, -NR14C (0 ) R15 , NRi6C (0) Ri7Ri8, -C (0 ) NRi9NR20R2i , and -NR22S02R23 ; R4 to R23 being,
independently of one another, a hydrogen atom or one of the following groups: (Ci-C6) alkyl, cycloalkyl, aryl, aralkyl, heterocycle, and heterocycle-alkyl , said groups being optionally substituted with one or more, preferably one to five, notably one or two, substituents selected from halogen, (d-C6) alkyl , NR24R25 and OR26, R24 to R26 being a hydrogen atom or a (Ci-C6) alkyl .
In the context of the invention, the expression "Y, Rx and R3 may optionally be together included into any optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle" means that when Y is NR3, Rx and R3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle. The heterocycle or heterobicycle thus comprises at least one nitrogen atom which carries the group R3.
In a particular embodiment of the invention, Y is S, 0 or a sulfone group (-S(0)2-) . More particularly, Y is S.
In another particular embodiment of the invention, Y is N(R3) and Ri and R3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle, preferably into a 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, in particular a 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, more particularly a heterobicycle selected from the group consisting of benzotriazole and benzimidazole .
In a particular embodiment of the present invention, Rx is an optionally substituted aryl or heteroaryl . More particularly, Ri is an optionally substituted phenyl or pyridyl. Advantageously, Ri is an optionally substituted phenyl or an unsubstituted pyridyl.
Preferably Ri is an aryl or heteroaryl, in particular a pyridyl or phenyl, notably a phenyl, optionally substituted with one or more, notably one to ten, in particular one to five, more
particularly one or two, substituent ( s ) , preferably selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo ( =0 ) , R4, -0R5 , -SR6, -NR7R8, -C ( 0 ) R9, -C(O)OR10, -C ( 0 ) NRnR12 , - 0C(0) Ri3 , -NR14C (0) Ri5, -NRi6C (0) Ri7Ri8, -C (0 ) Ri9 R20R2i , and R22SO2R23 ; R4 to R23 being, independently of one another, a hydrogen atom or one of the following groups: (Ci-C6) alkyl , cycloalkyl, aryl, aralkyl, heterocycle, and heterocycle-alkyl , notably (Ci-C6) alkyl, cycloalkyl, heterocycle, and heterocycle- alkyl, said groups being optionally substituted with one or more, preferably one to five, notably one to three, halogen, (Ci~ C6) alkyl, NR24R25 or OR26r R24 to R26 being a hydrogen atom or a (Ci~ C6) alkyl .
Preferably, Ri is an aryl or heteroaryl, in particular a pyridyl or phenyl, notably a phenyl, optionally substituted with one or two substituent ( s ) selected from the group consisting of: halogen, cyano (CN) , nitro ( N02 ) , oxo ( =0 ) , R4, -0R5 , -SR6, -NR7R8, -C(0)R9, -C(0)ORio, -C (0) R11R12 , -0C(0) Ri3 , -NR14C (0) Ri5, and - Ri6C ( 0 ) Ri7Ri8; R4 to R23 being, independently of one another, a hydrogen atom or one of the following groups: (Ci-C6) alkyl , cycloalkyl, aryl, aralkyl, heterocycle, and heterocycle-alkyl, notably (Ci-C6) alkyl, cycloalkyl, heterocycle, and heterocycle- alkyl, said group being optionally substituted with one or more, preferably one to five, notably one to three, halogen, (Ci~ C6) alkyl, NR24R25 or OR26r R24 to R26 being a hydrogen atom or a (Ci~ C6) alkyl.
Preferably, Ri is an aryl or heteroaryl, in particular a pyridyl or phenyl, notably a phenyl, optionally substituted with one or two substituent ( s ) selected from the group consisting of: halogen, nitro (N02 ) , R4, -0R5 , -NR7R8, -C ( 0 ) R9, -C(O)ORi0, C(0) RiiRi2 , -0C(0) Ri3 , -NRi C (0) R15, and -NRi6C (0 ) Ri7Ri8 ; R4 to R23 being, independently of one another, a hydrogen atom or one of the following groups: (Ci-C6) alkyl , cycloalkyl, aryl, aralkyl, heterocycle, and heterocycle-alkyl, notably (Ci-C6) alkyl , cycloalkyl, heterocycle, and heterocycle-alkyl, said group being
optionally substituted with one or more, preferably one to five, notably one to three, halogen, (d-C6) alkyl, NR24R25 or OR26 , R24 to R26 being a hydrogen atom or a ( Ci-C6 ) alkyl .
Preferably, Ri is an aryl or heteroaryl, in particular a pyridyl or phenyl, notably a phenyl, optionally substituted with one or two substituent ( s ) selected from the group consisting of: halogen, nitro (N02 ) , R4 , -OR5 , -NR7R8 , - C ( 0 ) RnRi2 , -NR14C ( 0 ) Ri 5 , and -NRi6C ( 0 ) Ri7Ri8 ; R4 to R23 being, independently of one another, a hydrogen atom or one of the following groups: ( Ci-C6 ) alkyl , cycloalkyl, aryl, aralkyl, heterocycle, and heterocycle-alkyl, notably ( Ci-C6 ) alkyl, cycloalkyl, heterocycle, and heterocycle- alkyl, said group being optionally substituted with one or more, preferably one to five, notably one to three, ( Ci- C6 ) alkyl , NR24R25 or OR26 , R24 to R26 being a hydrogen atom or a ( Ci- C6 ) alkyl .
In the above mentioned embodiments for Ri , the aryl of the aryl and aralkyl groups is advantageously a phenyl or naphtyl, in aprticular a phenyl.
In the above mentioned embodiments for Rlr the cycloalkyl comprises advantageously 3 to 6 carbon atoms, such as a cyclopropyl, a cyclopentyl or a cyclohexyl, in particular a cyclopropyl or a cyclohexyl .
In the above mentioned embodiments for Rlr the heterocycle of the heterocycle and heterocycle-alkyl groups is advantageously a saturated heterocycle. It will advantageously contain at least one, notable one to three, such as one or two, nitrogen atoms and optionally one or two, notably one oxygen atoms. It can be a saturated 4- to 7-, such as 5- or 6-membered heterocycle comprising one cycle and comprising one to three, such as one or two nitrogen atoms and optionally one or two, notably one oxygen atoms. It can be a pyrrolidine, piperidine, piperazine or morpholine cycle, such as a pyrrolidine, piperidine, or piperazine cycle, in particular a pyrrolidine or piperidine cycle .
In a particular embodiment of the invention, R2 is a group selected from (d-C6) alkyl, aralkyl, aryl, heteroaryl, heterocycle-alkyl or heteroaryl-alkyl , in particular aralkyl, aryl, heteroaryl, heterocycle-alkyl or heteroaryl-alkyl, more particularly aralkyl or heteroaryl-alkyl, preferably aralkyl or heteroaryl-alkyl in which the alkyl group is a (Ci-C2) alkyl group; said group being optionally substituted with one or more, notably one to ten, in particular one to five, more particularly one to four, substituent ( s ) , preferably selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0), R27, - OR28, -SR29, -NR30R31, -C(0)R32, -C(0)OR33, -C (0) NR34R35, -OC(0)R36, - NR37C(0)R38, -NR39C (0) NR40R41, -C (0) R42 R43R44, and - R45S02R46; R27 to R46 being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C6) alkyl , cycloalkyl, aryl, aralkyl, heterocycle and heterocycle-alkyl, said groups being optionally substituted with one or more, preferably one to five, notably one to two, halogen, (d-C6) alkyl , NR47R48 or OR 9, R 7 to R 9 being an hydrogen atom or a (Ci-C6) alkyl .
In a particular embodiment of the invention, R2 is a group selected from (d-C6) alkyl, aralkyl, aryl, heteroaryl, heterocycle-alkyl or heteroaryl-alkyl, in particular aralkyl, aryl, heteroaryl, heterocycle-alkyl or heteroaryl-alkyl, more particularly aralkyl or heteroaryl-alkyl, preferably aralkyl or heteroaryl-alkyl in which the alkyl group is a (Ci-C2) alkyl group; said group being optionally substituted with one to four substituent ( s ) selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0), R27, -OR28, -NR30R3i, -C(0)R32, - C(0)OR33, -C (0)NR34R35, -OC(0)R36, -NR37C (0 ) R38 , -C (0) NR42NR43R44, and - NR 5S02R 6; R27 to R 6 being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C6) alkyl , aralkyl, and heterocycle-alkyl, said groups being optionally substituted with one or more, preferably one to five, notably one to two, halogen, (d-C6) alkyl , NR47R48 or OR 9, R 7 to R 9 being an hydrogen atom or a (d~d) alkyl .
In a particular embodiment of the invention, R2 is a group selected from (d-C6) alkyl, aralkyl, aryl, heteroaryl, heterocycle-alkyl or heteroaryl-alkyl , in particular aralkyl, aryl, heteroaryl, heterocycle-alkyl or heteroaryl-alkyl, more particularly aralkyl or heteroaryl-alkyl, preferably aralkyl or heteroaryl-alkyl in which the alkyl group is a (Ci-C2) alkyl group; said groups being optionally substituted with one to four substituent ( s ) selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0), R27, -OR28, -NR30R3i, -C(0)R32, - C(0)OR33, -C (0) NR34R35, -C (0) NR42NR43R44, and - R45S02R46; R27 to R46 being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C6) alkyl , and heterocycle-alkyl, said group being optionally substituted with one or more, preferably one to five, notably one to two, (Ci-C6) alkyl , NR47R48 or OR49, R7 to R9 being an hydrogen atom or a (Ci-C6) alkyl .
In a particular embodiment of the invention, R2 is a group selected from (d-C6) alkyl, aryl- (d-C6) alkyl, aryl, heteroaryl, heterocycle- (Ci-C6) alkyl or heteroaryl- (d-C6) alkyl , in particular aryl- (d~d) alkyl, aryl, heteroaryl, heterocycle- (d~d) alkyl or heteroaryl- (d~d) alkyl, such as aryl- (d_d) alkyl, aryl, heteroaryl, heterocycle- (d_d) alkyl or heteroaryl- (d_d) alkyl, more particularly aryl- (d~d) alkyl or heteroaryl- (d~d) alkyl , such as aryl- (d_d) alkyl or heteroaryl- (d_d) alkyl , wherein the aryl (in the aryl and aryl-alkyl groups) is a phenyl or a naphtyl; and the heterocycle and heteroaryl (in the heterocycle, heterocycle-alkyl, heteroaryl and heteroaryl-alkyl) are a heterocycle or heteroaryl comprising one or two fused cycles comprising one to three, notably one or two, heteroatoms selected from 0, S and N, such as a thiophene, a furane, a pyrrole, a pyrazole, an imidazole, an oxazole, an isoxazole, a thiazole, an isothiazole, a triazole, a pyridine, an indole, an azaindole (such as an indazole or a 7-azainole) , an indoline, a benzofurane or a benzothiophene, in particular a thiophene, a furane, a pyridine, an indole, an indazole, a 7-azainole, an indoline or a
benzothiophene, for example an indole,
said group being optionally substituted with one or more, notably one to ten, in particular one to five, more particularly one to four, substituent ( s ) , preferably selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0), R27, - OR28, -SR29, -NR30R31, -C(0)R32, -C(0)OR33, -C (0) NR34R35, -OC(0)R36, - NR37C(0)R38, -NR39C (0) NR40R41, -C (0) R42 R43R44, and - R45S02R46; R27 to R46 being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C6) alkyl , cycloalkyl, aryl, aralkyl, heterocycle and heterocycle-alkyl , said groups being optionally substituted with one or more, preferably one to five, notably one to two, halogen, (d-C6) alkyl , NR47R48 or OR9, R7 to R9 being an hydrogen atom or a (Ci-C6) alkyl .
In a particular embodiment of the invention, R2 is a group selected from aryl- (d-C6) alkyl , aryl, heteroaryl, heterocycle- (Ci-C6) alkyl or heteroaryl- (d-C6) alkyl , such as aryl- (Ci-C2) alkyl , aryl, heteroaryl, heterocycle- (Ci-C2) alkyl or heteroaryl- (d- d) alkyl, more particularly aryl- (d_d) alkyl or heteroaryl- (d_ d) alkyl, such as aryl- (d_d) alkyl or heteroaryl- (d_d) alkyl , wherein the aryl (in the aryl and aryl-alkyl groups) is a phenyl or a naphtyl; and the heterocycle and heteroaryl (in the heterocycle, heterocycle-alkyl, heteroaryl and heteroaryl-alkyl ) are a heterocycle or heteroaryl comprising one or two fused cycles comprising one to three, notably one or two, heteroatoms selected from 0, S and N, such as a thiophene, a furane, a pyrrole, a pyrazole, an imidazole, an oxazole, an isoxazole, a thiazole, an isothiazole, a triazole, a pyridine, an indole, an azaindole (such as an indazole or a 7-azainole) , an indoline, a benzofurane or a benzothiophene, in particular a thiophene, a furane, a pyridine, an indole, an indazole, a 7-azainole, an indoline or a benzothiophene, for example an indole,
said group being optionally substituted with one to four substituent ( s ) selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0), R27, -OR28, -NR30R3i, -C(0)R32, -
C(0)OR33, -C (ONR34R35, -OC(0)R36, -NR37C (0) R38, -C (0) NR42NR43R44, and - R45SO2R46 R27 to R46 being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C6) alkyl , aralkyl, and heterocycle-alkyl , said groups being optionally substituted with one or more, preferably one to five, notably one to two, halogen, (d-C6) alkyl , NR47R48 or OR49, R47 to R49 being an hydrogen atom or a (Ci-C6) alkyl , wherein the heterocycle of the heterocycle-alkyl can be a saturated 4- to 7-, notably 5- or 6- membered heterocycle comprising one to three, notably one or two heteroatoms chosen from 0, N and S, notably chosen from N and 0, such as a pyrrolidine, piperidine, piperazine or morpholine cycle, in particular a piperazine or morpholine cycle.
In a particular embodiment of the invention, R2 is a group selected from aryl- (d-C6) alkyl , aryl, heteroaryl, heterocycle- (Ci-C6) alkyl or heteroaryl- (d-C6) alkyl , such as aryl- (Ci-C2) alkyl , aryl, heteroaryl, heterocycle- (Ci-C2) alkyl or heteroaryl- (d_ d) alkyl, more particularly aryl- (d_d) alkyl or heteroaryl- (d_ d) alkyl, such as aryl- (d_d) alkyl or heteroaryl- (d_d) alkyl , wherein the aryl (in the aryl and aryl-alkyl groups) is a phenyl or a naphtyl; and the heterocycle and heteroaryl (in the heterocycle, heterocycle-alkyl, heteroaryl and heteroaryl-alkyl ) are a heterocycle or heteroaryl comprising one or two fused cycles comprising one to three, notably one or two, heteroatoms selected from 0, S and N, such as a thiophene, a furane, a pyrrole, a pyrazole, an imidazole, an oxazole, an isoxazole, a thiazole, an isothiazole, a triazole, a pyridine, an indole, an azaindole (such as an indazole or a 7-azainole) , an indoline, a benzofurane or a benzothiophene, in particular a thiophene, a furane, a pyridine, an indole, an indazole, a 7-azainole, an indoline or a benzothiophene, for example an indole,
said groups being optionally substituted with one to four substituent ( s ) selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0), R27, -OR28, -NR30R3i, -C(0)R32, - C(0)OR33, -C (0)NR34R35, -C(0)NR42NR43R44, and - R45S02R46; R27 to R46
being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C6) alkyl , and heterocycle-alkyl, said group being optionally substituted with one or more, preferably one to five, notably one to two, (Ci-C6) alkyl , NR47R48 or OR49, R47 to R9 being an hydrogen atom or a (Ci-C6) alkyl , wherein the heterocycle of the heterocycle-alkyl can be a saturated 4- to 7-, notably 5- or 6-membered heterocycle comprising one to three, notably one or two heteroatoms chosen from 0, N and S, notably chosen from N and 0, such as a pyrrolidine, piperidine, piperazine or morpholine cycle, in particular a piperazine or morpholine cycle.
In a particular embodiment, the present invention relates to a compound of formula (I) wherein:
· A is N or C-H, preferably N;
• Y is S, 0 or a sulfone group (-S(0)2-), preferably S;
• Ri is a phenyl, optionally substituted with one or two, substituent ( s ) , preferably selected from the group consisting of: halogen, nitro (N02) , R , -0R5, -NR7R8, -C (0) RnRi2, -NRi4C (0) Ri5, and -NRi6C (0) Ri7Ri8; R to Ri8 being, independently of one another, an hydrogen atom or one of the following groups: a (Ci-C6) alkyl , cycloalkyl, heterocycle, and heterocycle-alkyl, said group being optionally substituted with one to three, halogen, (d-C6) alkyl , NR2 R25 or OR26, 24 to R26 being an hydrogen atom or a (Ci-C6) alkyl .
In a particular embodiment, the present invention relates to a compound of formula (I) wherein:
• A is N or C-H, preferably N;
• Y is S, 0 or a sulfone group (-S(0)2-), preferably S;
· Ri is a phenyl, optionally substituted with a -C(0)NRnRi2 group, R11 and Ri2 being an hydrogen atom or a (Ci-C6) alkyl , preferably an hydrogen atom.
In a particular embodiment, the present invention relates to
a compound of formula (I) wherein:
• A is N or CH, preferably N;
• Y is N (R3) ; and
• Ri and R3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle, preferably into a 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, in particular a a 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, more particularly a heterobicycle selected from the group consisting of benzotriazole and benzimidazole.
In a particular embodiment, the present invention relates to a compound of formula (I) wherein:
• A is N;
• Y is 0;
• Ri is phenyl optionally substituted by one of the following groups: hydroxy, hydroxy-alkyl more particularly hydroxymethyl ,
In the above definitions of Ri :
the alkyl group is preferably a (Ci-C6)alkyl group, in particular a (C1-C3) alkyl group;
the aryl group in the aryl and aralkyl groups is preferably a phenyl group;
- the heteroaryl in the heteroaryl and heteroaryl-alkyl groups is preferably a pyridyl group
the cycloalkyl group is preferably a (C3-C6) cycloalkyl group, in particular a cyclopropyl, cyclopentyl or a cyclohexyl, such as a cyclopropyl or a cyclohexyl; and
- the heterocycle group in the heterocycle and heterocycle-alkyl groups is preferably a saturated heterocycle containing at least one, notable one to three, such as one or two, nitrogen
atoms and optionally one or two, notably one oxygen atoms. It can be a saturated 4- to 7-, such as 5- or 6-membered heterocycle comprising one cycle and comprising one to three, such as one or two nitrogen atoms and optionally one or two, notably one oxygen atoms. It can be a pyrrolidine, piperidine, piperazine or morpholine cycle, such as a pyrrolidine, piperidine, or piperazine cycle, in particular a pyrrolidine or piperidine cycle. In the above definitions of R2 :
the alkyl group is preferably a (Ci-C6)alkyl group, in particular a (Ci-C4) alkyl group, more particularly a (Ci~ C2) alkyl group;
the aryl group in the aryl and aralkyl groups is preferably a phenyl or a naphtyl;
the heteroaryl group in the heteroaryl and heteroaryl-alkyl groups is preferably a heteroaryl comprising one or two fused cycles comprising one to three, notably one or two, heteroatoms selected from 0, S and N, such as a thiophene, a furane, a pyrrole, a pyrazole, an imidazole, an oxazole, an isoxazole, a thiazole, an isothiazole, a triazole, a pyridine, an indole, an azaindole (such as an indazole or a 7-azainole) , an indoline, a benzofurane or a benzothiophene, in particular a thiophene, a furane, a pyridine, an indole, an indazole, a 7-azainole, an indoline or a benzothiophene, for example an indole ;
the heterocycle group in the heterocycle and heterocycle-alkyl groups is preferably a saturated 4- to 7-, notably 5- or 6- membered heterocycle comprising one to three, notably one or two heteroatoms chosen from 0, N and S, notably chosen from N and 0, such as a pyrrolidine, piperidine, piperazine or morpholine cycle, in particular a piperazine or morpholine cycle .
The compound according to the present invention can be in particular selected from the compounds 1001 to 1119 of the examples below and the pharmaceutically acceptable salts thereof.
The present invention also relates to a compound of formula (I) as defined above for use as a drug, in particular as PLK-1 inhibitors, notably for treating cancer.
The invention also relates to the use of a compound of the invention for manufacturing a drug, notably useful as PLK-1 inhibitors, in particular intended for treating cancer.
The invention further relates to a method for treating cancer, comprising the administration of an effective quantity of at least one compound of formula (I) as defined above to a patient in need thereof.
The cancer can be more particularly in this case a colon cancer, breast cancer, kidney cancer, liver cancer, pancreas cancer, prostate cancer, lung cancer, ovarian cancer, head and neck cancer, glioblastoma, neuroblastoma, lymphoma, leukaemia, inflammatory myofibroblastic tumour, myelodysplastic syndrome, or myelofibrosis .
The present invention also relates to a pharmaceutical composition comprising at least one compound according to the invention and at least one pharmaceutically acceptable carrier.
The compound of the invention can be administered by oral, sublingual, parenteral, subcutaneous, intramuscular, intravenous, transdermal, local or rectal route, preferably by oral, intravenous or subcutaneous route.
In the pharmaceutical compositions of the present invention for oral, sublingual, parenteral, subcutaneous, intramuscular, intravenous, transdermal, local or rectal administration, the active ingredient can be administered in unit dosage forms, in mixture with traditional pharmaceutical carriers, to animals or humans .
Suitable unit dosage forms comprise forms for oral route such as tablets, gelatine capsules, powders, granules and oral solutions or suspensions; sublingual and oral administration forms; parenteral, subcutaneous, intramuscular, intravenous, intranasal or intraocular administration forms; and forms for rectal administration.
When a solid composition is prepared in the form of tablets, the main active ingredient is mixed with a pharmaceutical vehicle such as gelatin, starch, lactose, magnesium stearate, talc, gum arabic and the like. The tablets may be coated with sucrose or with other suitable materials, or they may be treated in such a way that they have a prolonged or delayed activity and they continuously release a predetermined amount of active principle.
A preparation in gelatin capsules can be obtained by mixing the active ingredient with a diluent and pouring the mixture obtained into soft or hard gelatin capsules.
A preparation in the form of a syrup or an elixir may contain the active ingredient together with a sweetener, an antiseptic, a taste enhancer or a suitable coloring agent.
The water-dispersible powders or granules may contain the active ingredient mixed with dispersing agents, wetting agents, or suspending agents, and with flavor correctors or sweeteners.
For parenteral administration, aqueous suspensions, isotonic saline solutions or sterile and injectable solutions which contain pharmacologically compatible dispersing agents and/or wetting agents can be used.
The active principle may also be formulated in the form of microcapsules, optionally with one or more carrier additives.
The compounds of the invention can be used in a pharmaceutical composition at a dose ranging from 0.01 mg to 1000 mg a day, administered in only one dose once a day or in several doses along the day, for example twice a day. The daily administered dose is advantageously comprises between 5 mg and 500 mg, and more advantageously between 10 mg and 200 mg.
However, it can be necessary to use doses out of these ranges, which could be noticed by the person skilled in the art.
The pharmaceutical compositions according to the present invention can further comprise at least another active principle, such as an anticancer agent.
The present invention also relates to a pharmaceutical composition comprising:
(i) at least one compound of formula ( I ) as defined previously, and
(ii) at least another active principle, such as an anticancer agent,
as a combination product for a simultaneous, separate or sequential use.
The present invention also relates to a pharmaceutical composition as defined previously for use as a drug, in particular as PLK-1 inhibitors, notably for treating cancer.
The present invention also relates to a method for treating cancer comprising the administration to a person in need thereof of an effective amount of a pharmaceutical composition according to the invention.
The present invention relates also to the processes to prepare the compound according to the present invention.
The present invention concerns thus a first process to prepare a compound according to the present invention comprising the following steps:
in which A, Y and Ri are as defined previously, and Xi represents COOH or an activated form thereof,
with a compound of formula R2NH2 in which R2 is as defined previously,
to give a compound of formula (I) as defined above, and (bl) optionally salifying the compound of formula (I) obtained in step (al) to give a pharmaceutically acceptable salt of a compound of formula (I) .
Step (al) :
By "activated form" of COOH is meant a COOH group in which the OH moiety has been replaced with an activated leaving group (LG) enabling coupling of the activated COOH group with an amino group by formation of an amide bond and release of the compound LG-H. Activated forms may be activated esters, activated amides, anhydrides or acyl halides such as acyl chlorides. Activated esters include derivatives formed by reaction of the COOH group with N-hydroxybenzotriazole or N-hydroxysuccinimide . It can be in particular COC1.
The reaction between the compound of formula (II) , optionally in an activated form, and the nucleophile of formula R2NH2 can be performed by methods well-known to the one skilled in the art. When X1 = COOH, step (al) can be performed by a peptide coupling .
The peptide coupling will be advantageously carried out in the presence of a coupling agent, such as diisopropylcarbodiimide (DIC) , dicyclohexylcarbodiimide (DCC) , 1- (3-dimethylaminopropyl ) - 3-ethylcarbodiimide hydrochloride (EDC) , carbonyldiimidazole (CDI), hexafluorophosphate 2- ( lH-benzotriazole-l-yl ) -1 , 1 , 3 , 3- tetramethyluronium (HBTU) , tetrafluoroborate 2- ( lH-benzotriazole- 1-yl) -1, 1, 3, 3-tetramethyluronium (TBTU) , hexafluorophosphate 0- (7-azobenzotriazol-l-yl) -1,1,3, 3-tetramethyluronium (HATU) , (benzotriazol-l-yloxy) tripyrrolodinophosphonium
hexafluorophosphate (PyBOP) or propylphosphonic anhydride; optionally associated with an additive or a base, such as N- hydroxy-succinimide (NHS), N-hydroxy-benzotriazole (HOBt) , 3,4- dihydro-3-hydroxy-4-oxo-l , 2 , 3-benzotriazole (HOOBt) , l-hydroxy-7- azabenzotriazole (HAt) , N-hydroxysylfosuccinimide (sulfo NHS) , dimethylaminopyridine (DMAP) , diisopropylethylamine (DIPEA) or N- methylmorpholine (NMM) . It can be carried out in particular in the presence of HATU, HBTU or PyBOP, optionally in the presence of DIPEA.
This reaction can be carried out for example in dimethylformamide (DMF) as solvent.
When Xi is an activated COOH group, as a COC1 group, the reaction can be performed in the presence of a base in conditions well-known to the one skilled in the art.
In this case, the compound of formula (II) can be prepared from the carboxylic acid derivative (i.e. compound of formula (II) with Xi = COOH) by methods well-known in the art. The compound of formula (II) with Xi = COOH can be prepared by hydrolysis of the ester function of the compound of following formula (IV) :
in which X3 = CO- (Ci-C6) alkyl, such as COMe or COEt, notably COMe, and A, Y and Ri are as defined previously.
The hydrolysis can be performed in acidic conditions or basic conditions (saponification) . It can be performed notably according to Heterocycles , 2005, vol. 65(10), 2321-2327 or US5252538.
The compound of formula (IV) can be prepared by coupling of a compound of formula RiYH with Y and Ri as defined above and a
compound of following formula
in which X3 is as defined above and A and X2 are as defined above.
Such a coupling reaction can be carried out in the conditions detailed in step (a2) below.
Step (bl) :
The salification step can be carried out by methods well known to the one skilled in the art, in particular by reaction of the compound of formula (I) obtained in step (a) with a pharmaceutically acceptable acid (organic or inorganic acid) , base (organic or inorganic base) or solvent, as defined previously . The present invention concerns also a second process to prepare a compound according to the present invention comprising the following steps:
in which A and R2 are as defined previously, and X2 represents a halogen atom, such as CI, Br or I,
with a compound of formula RiYH in which Rx and Y are as defined previously,
to give a compound of formula (I) as defined above, and
(b2) optionally salifying the compound of formula (I) obtained in step (a2) to give a pharmaceutically acceptable salt of a compound of formula (I) .
Step (a2)
The coupling reaction of step (a2) can be performed notably in the presence of Cul .
The compound of formula (III) can be prepared by coupling of a compound of formula R2NH2 and a compound of following formula (VI) :
in which A and X2 are as defined above.
Such a coupling reaction can be carried out in the conditions detailed in step (al) above.
Step (b2) : cf. step (bl)
Protection / deprotection steps or functionalization steps can be carried out in the processes described above, such steps and their reaction conditions being well known to the one skilled in the art.
The compound obtained can be separated from the reaction medium by methods well known to the person skilled in the art, such as by extraction, evaporation of the solvent or by precipitation or crystallisation (followed by filtration) .
The compound can also be purified if necessary by methods well known to the person skilled in the art, such as by recrystallisation, by distillation, by chromatography on a column of silica gel or by high performance liquid chromatography (HPLC) .
The present invention is illustrated by the following non- limitative examples.
EXAMPLES :
1. Synthesis of the compounds according to the invention :
A = CH, N Nu = RrY A = CH, N CH, N
X = CI, Br, I X = CI, Br, I Nu = R Y
Production of claimed compounds arises from a synthesis sequence starting from a reactant -la-f- to a compound -6a-z- or -7a-z-, conducted in either of the two depicted orders, optionally followed by post-synthetic modifications.
Name Substitution Method of Synthesis
Pattern
-la- (A = CH, X = WO2006/88840; WO2006/91428; WO2006/88837;
CI) WO2011/117254; J. Med. Chem., 2006, vol. 49(3), p. 1125-1139; WO2014/23814.
-lb- (A = CH, X = WO2006/88837.
tsr)
-lc- (A = CH, X = WO2011/117254.
I)
-ld- (A = N, X = US5866568; US5668137; J. Am. Chem. Soc ., 1949,
CI) 71, 2798.
-le- (A = N, X = WO2007/61360; Heterocycles 2005, vol 65 (10) ,
Br) p.2321-2327; WO2004/92177; WO2014/23814; J. Med.
Chem., 2012, vol. 22(6), p. 2266-2270; J. Med.
Chem., 2012, vol. 22(8), p. 2784-2788; J. Org.
Chem., 1964, 29, 2491; J. Med. Chem. 1965, 8,
638.
-lf- (A = N, X = I) WO2012/101239; US2013/85144.
Product Substitution Reactant Method of Synthesis
Pattern
-2a- (A = CH, X = -la- WO2004/52890; US3950160.
CI)
-2b- (A = CH, X = -lb- US6369056; US2006/30583; US6399593;
Br) US3950160.
-2c- (A = N, X = -le- WO2011/143419; Biorg. Med. Chem.
Br) Lett., 2012, 22(8), 2784.
-2d- (A = N, X = I) -lf- J. Am. Chem. Soc, 1949, 71, 2798;
WO2006/15124; EP2157090.
Intermediate -4a- Depending on the methods used, the main final product may either be an ester (-3a-z-) or an acid (-4a-z-) derivative. In the former cases, supplementary hydrolysis of esters to the corresponding acids were conducted using procedures such as the ones used below. If necessary, esterification of acids to the corresponding methyl or ethyl esters were conducted using standard well-known procedures (ex.: Heterocycles, 2005, vol. 65(10), 2321-2327; US5252538) .
Method Ai
(From Buchwald S.L., Org. Lett., 2002, 4(20), 3517-2520) . Example -4a- To a mixture under argon of 0.5 mmol of intermediate -If-,
0.025 mmol of Cul and 1 mmol of potassium carbonate in a small thick glass reactor (Ace® Tube) , are successively added 1 mL of isopropanol, 1 mmol of ethylene glycol and 0.75 mmol of thiophenol. The reactor is sealed with a Teflon stopper, placed under magnetic stirring and heated two hours at 80°C. The reaction mixture is cooled to room temperature and the thus obtained precipitate isolated through filtration, washed and recrystallized from hot methanol (yield : 88%) .
LCMS (IE, m/z) : (M+l) 248; 1H NMR: δΗ ppm 400 MHz, DMSO: 7.19 - 7.14 (3H, m, CHarom.), 7.30-7.26 (2H, m, CHarom.), 7.97 (1H, s,
CHar0m. ) ·
Substitution Pattern
Product X A R Reactant Method of Synthesis
-5a- I N -2d- Method Ci
Method Ci
Example -5a-
To an ice-cooled stirred solution of 0.1 mmole of -2d- and 0.13 mmole of HATU (hexafluorophosphate of 0- ( 7-Azabenzotriazol- 1-yl) -N, N, N ' , ' -tetramethyluronium) in 1 mL of dry DMF (dimethylformamide) , are successively added at a 20 min interval, 0.5 mmole of DIPEA (N, -diisoprolylethylamine) and 0.13 mmole of ( lH-indol-3-yl ) methanamine . The reaction mixture is agitated for a two hours period while the temperature is progressively allowed to rise to ambient and then to 50°C. Reaction crude is then poured into 25 mL of dichloromethane and successively washed with NaHC03 saturated water, water and brine. The combined organic layers are dried over Na2S04, evaporated to dryness and the crude purified by preparative reverse-phase HPLC (yield: 74%) .
LCMS (IE, m/z) : (M+l) 394; 1H NMR: δΗ ppm 400 MHz, DMSO: 10.94 (1H, si, NH) , 8.71 (1H, t, NH) , 8.36 (1H, s, CHarom. ) , 7.67 (2H , si, NH2),7.65 (1H, d, CHarom.) , 7.37 (1H, dt, CHarom. ) , 7.29 (1H, d, CHarom.) , 7.01 (1H, td, CHarom.) , 6.79 (1H, td, CHarom. ) , 4.59 (2H, d, CH2) .
Method C2
Example -5b-
To a stirred solution of 3.5 g (13.21 mmol) of -2d- in 30 mL of anhydrous DMF, are successively added 1.87 g (19.81 mmol) of pyridine-3-ylamine, 10.31 g (19.81 mmol) of benzotriazol-l-yloxy- tripyrrolidinophosphonium hexafluorophosphate and 4.4 mL (26.41 mmol) of diisopropylethylamine .
The reaction mixture is maintained under stirring for two days at 50°C, concentrated until precipitation and filtered. The thus obtained solid is washed with cold methanol to yield 3.1g of -5b- as a yellow solid (yield: 69%) .
LCMS (IE, m/z) : (M+l) 342.15; RMN 1H : δΗ ppm 400 MHz, DMSO: 10.47 (1H, si, NH), 8.95 (1H, d, CHarom.), 8.49 (1H, s, CHarom.), 8.33 (1H, dd, CHarom.), 8.17 (1H, ddd, CHarom.), 7.67 (2H, si, NH2) , 7.40 (1H, dd, CHarom. ) . Method C3
Example -5c-
To a stirred solution of 18.8 mmole of the acid -2d- (5 g) into 250 mL of dry DMF, are successively added 24 mmole (12.7g) of benzotriazol-l-yloxy-tripyrrolidinophosphonium hexafluorophosphate and 75 mmole (12g) of 2- (lH-Indol-3-yl) - ethylamine. The reaction mixture is agitated for 24 hours at 70°C, evaporated to dryness and redisolved into 380 mL of dichloromethane and 150 mL of a 5% solution of citric acid. The collected organic layer is washed twice with aqueous sodium hydroxide (1M, 75 mL) , brine, water and dried over magnesium sulfate. The evaporated crude is then purified by chromatography to yield 1.5 g of -5c- as a yellow solid (yield: 20%) .
LCMS (IE, m/z) : (M+l) 408; 1H NMR: δΗ ppm 400 MHz, DMSO: 10.82 (1H, s, NH) , 8.73 (1H, t, NH) , 8.40 (1H, s, CHarom. ) , 7.62 (2H, mlarge, NH2) , 7.60 (1H, d, CHarom.), 7.32 (1H, d, CHarom.), 7.18 (1H, m, CHarom.), 7.09 (1H, td, CHarom) , 6.89 (1H, t, CHarom.), 3.57 (2H, q, CH2) , 2.94 (2H, t, CH2) .
Compounds -6a- to -6c- from Intermediate -4a-
Example -6a-
Same as method C2 above except that stirring was maintained for 4 days .
93 mg (yield: 32.5%)
LCMS (IE, m/z) : (M+l) 354; 1H NMR δΗ ppm 400 MHz, DMSO : 10.28 (1H, s, NH), 8.47 (1H, d, CHarom.), 8.27 (1H, s, CHarom.), 8.03 (1H, dd, CHarom.), 7.77 (2H, s, NH2) , 7.35 (5H, m, CHarom.), 6.84 (1H, d, CHarom.) , 3.85 (3H, s, CH3) .
Method C4
Example -6b-
To Heck tube containing an ice-cold stirred solution of 0.7 mmole of the acid -4a- and 1.05 mmole of hexafluorophosphate of 0- (Benzotriazol-l-yl) -Ν,Ν,Ν' , ' -tetramethyluronium (HBTU) into 1 mL of dry DMF, are successively added 1.05 mmole of diisopropylethylamine and 1.05 mmole of 3-aminopyridine. The tube is sealed and the reaction mixture is agitated for 48 hours while heated to 50°C.
The evaporated crude is then purified by flash chromatography to yield -6a- as a yellow solid (yield: 23%) .
LCMS (IE, m/z) : (M+l) 324; 1H NMR δΗ ppm 400 MHz, DMSO: 10.39 (1H, s, CHarom.), 8.92 (1H, m, CHarom.), 8.35 (1H, slarge, NH) , 8.28 (1H, m, CHarom.), 8.16 (1H, m, CHarom.), 7.77 (2H, slarge, NH2) , 7.42 (5H, m, CHarom.), 7.33 (1H, m, CHarom.) .
Example -6c-
Same as above (yield: 24%) .
LCMS (IE, m/z) : (M+l) 348; 1H NMR δΗ ppm 400 MHz, DMSO : 10.51 (1H, s, CHarom.), 8.27 (2H, m, CHarom.), 8.05 (1H, m, NH) , 7.78 (2H, slarge, NH) , 7.59 (2H, m, CHarom.), 7.34 (5H, m, CHarom.) .
Compounds -7a- to -7d- from Intermediates -5a- to -5c-
Method A2
Example -7a-
To a mixture under argon of 0.5 mmol of intermediate -5b-, 0.1 mmol of Cul and 1 mmol of potassium phosphate in a small thick glass reactor (Ace® Tube) , are successively added 2 mL of
dioxane, 0.1 mmol of (1R, 2S) -cyclohexane-1, 2-diamine and 0.75 mmol of benzimidazole . The reactor is sealed with a Teflon stopper, placed under magnetic stirring and heated 120 hours at 80°C. The reaction mixture is cooled to room temperature, evaporated to dryness and the thus obtained crude purified by flash chromatography (yield: 54%) .
LCMS (IE, m/z) : (M+l) 332; 1H NMR δΗ ppm 400 MHz, DMSO : 10.57 (1H, s, NH), 9.02 (1H, m, CHarom.), 8.86 (1H, s, CHarom.), 8.20 (1H, m, CHarom.), 7.95 (1H, m, CHarom.), 7.81 (3H, m, NH2+ CHarom.), 7.52 (1H, m, CHarom.), 7.36 (3H, m, CHarom.) .
Method A3
Example -7b-
To a mixture under argon of 0.25 mmol of intermediate -5b- and 0.37 mmol of Cul in a small thick glass reactor (Ace® Tube), are successively added 0.8 mL of dry DMF and 0.39 mmol of phenyl sulfinate. The reactor is sealed with a Teflon stopper, placed under magnetic stirring and heated 3 hours at 130°C. The reaction mixture is cooled to room temperature, evaporated to dryness and the thus obtained crude purified by flash chromatography (yield: 37%) .
LCMS (IE, m/z) : (M+l) 422; 1H NMR δΗ ppm 400 MHz, DMSO: 10.86 (1H, s, CHarom.), 8.79 (1H, s, CHarom.), 8.61 (1H, s, NH) , 8.56 (1H, m, CHarom.), 8.33 (1H, s, NH) , 8.05 (2H, m, CHarom.), 7.68 (1H, m, CHarom.), 7.59 (3H, m, NH2+ CHarom.), 7.35 (1H, m, CHarom.), 7.18 (1H, m, CHarom.), 7.07 (1H, t, CHarom.), 6.94 (1H, t, CHarom.), 3.59 (2H, t, CH2) , 2.95 (2H, t, CH2) .
Method Ai
Example -7c-
Same as example -4a- above, applied to intermediate -5a-, with the reaction time extended to 120 hours (yield : 32%) .
LCMS (IE, m/z) : (M+l) 419; 1H NMR δΗ ppm 400 MHz, DMSO:; 10.98 (1H, s, NH) , 8.76 (1H, t, NH) , 8.28 (1H, s, CHarom.), 7.93 (3H, m,
NH2+ CHarom.), 7.70 (1H, d, CHarom.), 7.57 (2H, m, NH2) , 7.39 (1H, d, CHarom.), 7.32 (1H, s, CHarom.), 7.26 (2H, m, CHarom.), 7.12 (1H, t, CHarom.), 7.01 (2H, m, CHarom.), 4.63 (2H, d, CH2) .
Example -7d-
Same as example -4a- above, applied to intermediate -5c-, with the reaction time extended to 48 hours (yield: 53%) .
LCMS (IE, m/z) : (M+l) 408; 1H NMR δΗ ppm 400 MHz, DMSO : 10.85
(1H, s, NH), 8.55 (1H, slarge, NH) , 8.25 (1H, s, CHarom.), 7.80 (2H, s, NH2), 7.61 (1H, d, CHarom.), 6.90 - 7.45 (9H, m, CHarom.), 3.55 (2H, m, CH2) , 2.95 (2H, m, CH2) .
Complete list of synthesized molecules:
Note: Molecules are designated by an incremented four digits serial number followed, if available, by a 1 digit & 1-2 letters code referencing the synthetic methodology used for its synthesis .
Compound characterisation:
LCMS (IE, m/z) : (M+l) 437.46; XH NMR: δΗ ppm 400 MHz, DMSO; 11.30 (1H, si, NH),8.56 (1H, t, NH) , 8.25 (1H, s, CH arom.), 7.67-7.52 (3H, si, NH2, CH arom.), 7.37-7.49 (2H, m, CH arom.), 7.37-7.29 (4H, m, CH arom.), 6.99-7.19 (2H, m, CH arom.), 6.74 (1H, td, CH arom. ) , 4.67 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 437.46; XH NMR: δΗ ppm 400 MHz, DMSO; 11.43 (1H, si, NH) , 8.79 (1H, t, NH) , 8.23 (1H, s, CH arom. ) , 7.67-7.52 (3H , si, NH2, CH arom.), 7.54-7.89 (3H, m, CH arom.), 7.19 (1H, s, CH arom.), 6.88-6.97 (3H, m, CH arom.), 4.59 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 437.46; 1H NMR: δΗ ppm 400 MHz, DMSO; 11.01 (1H, si, NH) , 8.78 (1H, t, NH) , 8.23 (1H, s, CH arom.), 7.90 (3H , si, NH2, CH arom.), 7.63-7.67 (1H, m, CH arom.), 7.49-7.54 (2H, m, CH arom.), 7.20-7.27 (3H, m, CHarom) , 7.10-7.13 (1H, m, CH arom.), 6.94-6.96 (1H, m, CH arom.), 6.82-6.86 (1H, m, CH arom.), 4.59 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 481.51; XH NMR: δΗ ppm 400 MHz, DMSO; 8.85 (1H, t, NH) , 8.24 (1H, s, CH arom.), 7.90 (3H, si, NH2, CH arom.), 7.46-7.54 (4H, m, CH arom.), 7.38 (1H, s, CH arom.), 7.22-7.27 (2H, m, CH arom.), 6.92-6.96 (2H, m, CH arom.), 4.87 (1H, t, CH2) , 4.54 (2H, d, CH2) , 4.16 (2H, t, CH2) , 3.67 (2H, m, CH2) .
LCMS (IE, m/z) : (M+l) 470.47; XH NMR: δΗ ppm 400 MHz, DMSO; 11.32 (1H, si, NH) , 8.66 (1H, t, NH) , 7.88 (1H, s, CH arom.), 7.69 (1H , s, CH arom.), 7.47 (2H, si, NH2 ), 7.17-7.32 (2H, m, CH arom.), 7.11-7.20 (3H, m, CH arom.), 6.62 (1H, m, CH arom.), 6.52 (1H, m, CH arom.), 5.59 (2H, m, CH arom.), 4.67 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 432.09; XH NMR: δΗ ppm 400 MHz, DMSO; 10.05 (1H, si, OH), 8.85 (1H, t, NH) , 8.50 (1H, s, CH arom.), 7.85 (1H , s, CH arom.), 7.80 (2H , si, NH2) , 7.50-7.60 (2H, m, CH arom.), 7.25-7.30 (2H, m, CH arom.), 6.90-7.10 (2H, m, CH arom.), 6.85 (1H, m, CH arom.), 4.30 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 434.49; 1H NMR: δΗ ppm 400 MHz, DMSO; 10.45 (1H, si, NH) , 8.50 (1H, t, NH) , 8.20 (1H, s, CH arom.), 7.47 (3H, m, NH2, CH arom.), 7.40-7.60 (2H, m, CH arom.), 7.15-7.25 (3H, m,
CH arom.), 6.95 (1H, m, CH arom.), 6.80 (1H, m, CH arom.), 6.65 (1H, m, CH arom.), 6.25 (1H, d, CH arom.), 5.0 (2H , si, NH2) ,
LCMS (IE, m/z) : (M+l) 436.52; XH NMR: δΗ ppm 400 MHz, DMSO; 9.15 (1H, t, NH) , 8.25 (1H, s, CH arom.), 7.80-8.10 (5H, m, NH2, CH arom.), 7.45-7.58 (3H, m, CH arom.), 7.30-7.45 (2H, m, CH arom.), 7.20-7.30 (2H, m, CH arom.), 6.80-7.10 (1H, m, CH arom.), 4.70 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 437.46; XH NMR: δΗ ppm 400 MHz, DMSO; 11.04 (1H, si, NH) , 8.85 (1H, t, NH) , 8.28 (1H, s, CH arom.), 7.53-7.90 (3H, m, NH2, CH arom.), 7.19-7.47 (7H, m, CH arom.), 6.88-6.96 (2H, m, CH arom.), 4.53 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 452.48; XH NMR: δΗ ppm 400 MHz, DMSO; 11.04 (1H, si, NH) , 8.85 (1H, t, NH) , 7.85 (1H, s, CH arom.), 7.53-7.65 (1H, s, CH arom. ) , 7.53-7.65 (6H, m, NH2, CH arom.), 7.19-7.20 (1H, m, CH arom.), 6.88-6.96 (1H, m, CH arom.), 6.60 (1H, m, CHarom) , 6.40 (1H, m, CH arom.), 5.45 (2H, NH2, CH arom.), 4.40 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 491.53; XH NMR: δΗ ppm 400 MHz, DMSO; 9.40 (1H, si, NH) , 9.20 (1H, t, NH) , 8.25 (1H, s, CH arom. ) , 7.65-8.00 (3H, m, NH2, CH arom.), 7.45-7.60 (2H , m, CH arom. ) , 7.20-7.40 (3H, m, CH arom.), 7.15 (2H, m, CH arom.), 6.95 (1H, m, CH arom.), 4.45 (2H, d, CH2) , 3.00 (3H, s, CH3) .
LCMS (IE, m/z) : (M+l) 482.46; 1H NMR: δΗ ppm 400 MHz, DMSO; 11.04 (1H, si, NH) , 8.85 (1H, t, NH) , 7.40 (3H, m, CH arom.), 8.30 (1H, s, NH) , 8.00 (1H, d, CH arom.), 7.70 (1H, s, NH) , 7.55 (1H, d, CH arom.), 7.26-7.40 (3H, m, CH arom.), 7.20 (2H, m, CH arom.), 6.90 (1H, d, CH arom.), 6.80 (1H, m, CH arom.), 4.55 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 434.49; 1H NMR: δΗ ppm 400 MHz, DMSO; 10.45 (1H, si, NH) , 8.40 (1H, t, NH) , 8.20 (1H, s, CH arom.), 7.80-7.90 (5H, NH2, CH arom.), 7.40-7.47 (2H, m, CH arom.), 7.18-7.25 (2H, m, CH arom.), 6.90 (1H, d, CH arom.), 6.65 (1H, s, CH arom.), 6.45 (1H, d, CH arom.), 4.50 (2H, m, NH2) , 4.40 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 449.63; XH NMR: δΗ ppm 400 MHz, DMSO; 11.00 (1H, si, NH) , 8.75 (1H, t, NH) , 7.85 (1H, s, CH arom.), 7.55 (1H, s, CH arom.), 7.40-7.50 (3H, m, NH2, CH arom.), 7.40-7.45 (3H, m, CH arom.), 7.25 (1H, m, CH arom.), 6.90 (1H, t, CH arom.), 6.70 (1H, s, CH arom.), 6.5 (1H, d, CH arom.), 6.00 (1H, d, CH arom.), 4.50 (2H, d, CH2) , 2.60 (2H, m, CH2) , 2.40 (1H, m, CH2) , 2.20 (3H, m, CH3) , 2.00 (2H, m, CH2) , 1.85 (2H, m, CH2) , 1.00 (2H, m, CH2) .
LCMS (IE, m/z) : (M+l) 520.55; 1H NMR: δΗ ppm 400 MHz, DMSO; 11.00 (1H, si, NH) , 10.40 (1H, si, NH) , 8.85 (1H, t, NH) , 8.10 (1H, s,
CH arom.), 7.60-7.80 (3H, m, NH2, CH arom.), 7.40-7.50 (4H, m CH arom.), 7.30-7.35 (2H, m, CH arom.), 7.10 (1H, d, CH arom.), 6.80 (1H, t, CH arom.), 4.50 (2H, d, CH2) , 1.55 (4H, m, CH2) , 1.85 (1H, m, CH) .
LCMS (IE, m/z) : (M+l) 577.65; XH NMR: δΗ ppm 400 MHz, DMSO; 11.00 (1H, si, NH) , 8.85 (1H, t, NH) , 8.50 (1H, s, NH) , 8.10 (1H, s, CH arom.), 7.50-7.80 (3H, m, NH2, CH arom.), 7.20-7.40 (6H, m CH arom.), 7.20 (1H, m, CH arom.), 6.80 (1H, t, CH arom.), 6.20 (1H,
d, NH) , 4.50 (2H, d, CH2) , 3.45 (1H, m, CH) , 1.10-1.80 (10H, m,
LCMS (IE, m/z) : (M+l) 390; XH NMR: δΗ ppm 400 MHz, DMSO; 10.82 (1H, si, NH) , 8.60 (1H, t, NH) , 8.23 (1H, s, NH) , 7.81 (2H, s, NH2) , 7.60 (1H, d, CH arom.), 7.33 (7H, m, CH arom.), 7.15 (1H, s, CH arom.), 7.07 (1H, t, CH arom.), 6.97 (1H, t, CH arom.), 3.54 (2H, q, CH2 ), 2.93 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 374.15; 1H NMR: δΗ ppm 400 MHz, DMSO; 10.80 (1H, si, NH) , 8.33 (1H, t, NH) , 8.15 (1H, s, NH) , 7.56 (1H, d, CH arom.), 7.46 (2H, s , NH2) , 7.37 (3H, m, CH arom.), 7.10 (5H, m, CH arom.), 6.95 (1H, t, CH arom.), 3.51 (2H, q, CH2) , 2.90 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 411.14; XH NMR: δΗ ppm 400 MHz, DMSO; 8.48 (1H, t, NH) , 8.45 (1H, s, CH arom.), 7.77 (2H, s, NH2) , 7.37 (5H, m, CH arom.), 6.80 (2H, m, CH arom.), 6.68 (1H, m, CH arom.), 3.70 (6H, s, OCH3) , 3.47 (2H, m, CH2) , 2.77 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 367.12; XH NMR: δΗ ppm 400 MHz, DMSO; 9.18 (1H, s, 8.46 (1H, t, NH) , 8.23 (1H, s, CH arom.), 7.80 (2H, s, CH arom.), 7.34 (5H, m, CH arom.), 6.98 (2H, d, CH arom.), 6.65 (2H, d, CH arom.), 3.42 (2H, m, CH2) , 2.69 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 408.12; XH NMR: δΗ ppm 400 MHz, DMSO; 10.67 (1H, s, NH) , 8.55 (1H, s, NH) , 8.15 (1H, s, CH arom.), 7.80 (2H, s, NH2) , 7.50 (1H, d, CH arom.), 7.17 (2H, m, CH arom.), 7.01 (6H, m, CH arom.), 3.49 (2H, m, CH2) , 2.93 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 426.11; 1H NMR: δΗ ppm 400 MHz, DMSO; 10.82 (1H, s, NH) , 8.45 (1H, t, NH) , 8.20 (1H, s, CH arom.), 7,77 (2H, s, NH2) , 7.58 (1H, d, CH arom.), 7.46 (1H, m, CH arom.), 7.36 (2H, m, CH arom.), 7.15 (1H, s, CH arom.), 7.09 (2H, m, CH arom.), 6.95 (1H, t, CH arom.), 3.53 (2H, q, CH2) , 2.91 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 550.62; XH NMR: δΗ ppm 400 MHz, DMSO; 8.85 (1H, t, NH) , 8.25 (1H, s, CH arom.), 7.70-7.80 (3H, m, NH2, CH arom.), 7.40-7.60 (5H, s, CH arom.), 7.19-7.20 (2H, m, CH arom.), 6.88-6.96 (2H, m, CH arom.), 4.50 (2H, t, CH2) , 4.25 (2H, t, CH2) , 3.50 (4H, m, CH2) , 2.60 (2H, t, CH2) , 2.40 (4H, m, CH2) .
LCMS (IE, m/z) : (M+l) 451.49; XH NMR: δΗ ppm 400 MHz, DMSO; 11.00 (1H, si, NH) , 8.85 (1H, t, NH) , 8.00 (1H, s, CH arom.), 7.60-7.80
(3H, m, NH2, CH arom.), 7.50 (2H, m, CH arom.), 7.40 (lH,d, CH arom.), 7.20-7.40 (3H, m CH arom.), 6.80 (1H, d, CH arom.), 6.70
(1H, t, CH arom.), 4.50 (2H, d, CH2) , 2.40 (3H, s, CH3) .
LCMS (IE, m/z) : (M+l) 466.50; 1H NMR: δΗ ppm 400 MHz, DMSO; 10.95 (1H, si, NH) , 8.81 (1H, t, NH) , 7.84 (1H, s, CH arom.), 7.32-7.48 (5H, m, NH2, CH arom.), 7.21 (1H, s, CH arom.), 7.13-7.19 (1H, dd, CH arom.), 7.10-7.13 (1H, d, CH arom.), 6.78-6.82 (1H, m CH arom.), 6.62 (1H, s, CH arom.), 6.51 (1H, d, CH arom.), 5.58 (1H, s, NH2) , 4.49 (2H, d, CH2) , 2.46 (3H, s, CH3) .
LCMS (IE, m/z) : (M+l) 449.50; XH NMR: δΗ ppm 400 MHz, DMSO; 10.99 (1H, si, NH) , 8.69 (1H, t, NH) , 8.22 (1H, s, CH arom.),7.89 (3H, m, NH2, CH arom. ) , 7.63-7.65 (1H, d, CH arom. ) , 7.49-7.52 (2H, m, CH arom.), 7.28-7.30 (1H, d, CH arom.), 7.08-7.30 (2H, m CH arom.), 7.01-7.05 (1H, t, CH arom.), 6.92-6.95 (2H, m, CH arom.), 5.16-5.18 (1H, t, OH), 4.72 (2H, d, CH2) , 4.61 (2H, d, CH3) .
LCMS (IE, m/z) : (M+l) 451.49; XH NMR: δΗ ppm 400 MHz, DMSO; 8.89 (1H, t, NH) , 8.22 (1H, s, CH arom.), 7.91 (3H, m, NH2, CH arom.),
7.40-7.49 (3H, m, CH arom.), 7.37-7.39 (1H, m, CH arom.), 7.33 (1H, s, CH arom.), 7.18-7.24 (2H, m CH arom.), 6.93-7.00 (2H, m,
CH arom.), 4.52 (2H, d, CH2) , 3.74 (3H, s, CH3) .
LCMS (IE, m/z) : (M+l) 340.37; XH NMR: δΗ ppm 400 MHz, DMSO; 10.30 (1H, s, NH) , 11.18 (1H, s, 8.88 (1H, s, CH arom.), 8.34 (1H, d, CH arom.), 8.17 (1H, m, CH arom.), 8.02 (1H, s, CH arom.), 7.61 (2H, s, NH2) , 7.41 (1H, m, CH arom.), 7.24 (2H, m, CH arom.), 6.95 (1H, dd, CH arom.), 6.84 (1H, dt, CH arom.) .
LCMS (IE, m/z) : (M+l) 332; XH NMR: δΗ ppm 400 MHz, DMSO; 10.28 (1H, s, NH) , 8.77 (1H, s, CH arom.), 8.73 (1H, s, CH arom.), 8.26 (1H, s, CH arom.), 8.17 (2H, m, CH arom.), 8.01 (1H, d, CH arom.), 7.73 (2H, d, CH arom.), 7.41 (2H, s, NH2) , 7.39 (1H, d,
CH arom.), 7.24 (1H, m, CH arom.), 7.13 (1H, t, CH arom.) .
LCMS (IE, m/z) : (M+l) 439.48; XH NMR: δΗ ppm 400 MHz, MeOH; 8.22 (1H, s, NH) , 7.59-7.60 (1H, d, CH arom.), 7.29-7.35 (3H, m, CH arom.), 6.77-6.78 (1H, dd, CH arom.), 6.72-6.75 (1H, m, CH arom.), 6.60-6.63 (1H, d, CH arom.), 3.49-3.62 (5H, m, CH2, CH) .
LCMS (IE, m/z) : (M+l) 477.14; XH NMR: δΗ ppm 400 MHz, DMSO; 11.40 (1H, si, NH) , 10.15 (1H, t, NH) , 9.25 (1H, t, CHarom) , 8.20 (1H, s, CHarom) , 7.80-7.89 (4H, m, NH2, CHarom) , 7.45-7.60 (2H, m, CHarom) , 7.35 (1H, d, CHarom) , 7.15-7.25 (3H, m CHarom) , 6.90-7.15 (2H, m, CHarom) , 5.80 (2H, d, CH2) , 5.60 (2H, si, NH2) .
LCMS (IE, m/z) : (M+l) 462.13; XH NMR: δΗ ppm 400 MHz, DMSO; 11.41 (1H, si, NH) , 9.22 (1H, t, NH),8.26 (1H, s, NH) , 8.23 (1H, s, CH arom.), 7.56 (1H, s, NH) , 7.52-7.54 (1H, d, CH arom. ) , 7.40 (1H, s, NH) , 7.36-7.38 (3H, m, CH arom.), 7.25 (1H, d, CH arom.), 7.18-7.20 (3H, m, CH arom.), 6.99-7.07 (2H, m, CH arom.), 4.82 (2H, d, CH2) .
LCMS (IE, m/z) : (M+l) 506.15; 1H NMR: δΗ ppm 400 MHz, DMSO; 11.47 (1H, si, NH) , 9.26 (1H, t, NH) , 9.03 (1H, t, NH) , 8.24 (1H, s, CH arom.), 7.56-7.93 (4H, m, NH, CH arom.), 7.54 (2H, m, CH arom.), 7.39 (1H, d, CH arom.), 7.19-7.21 (3H, m, CH arom.), 7.01-7.06 (2H, m, CH arom.), 4.82 (3H, m, OH, CH2) , 3.61 (2H, m, CH2) , 3.46 (2H, m, CH2) .
LCMS (IE, m/z) : (M+l) 420.14; 1H NMR: δΗ ppm 400 MHz, DMSO; 10.80 (1H, s, NH) , 8.55 (1H, s, NH) , 8.11 (1H, s, CH arom.), 7, .74 (2H, s, NH2) , 7.58 (1H, d, CH arom.), 7.32 (1H, d, CH arom.), 7.27 (1H, t, CH arom.), 7.14 (1H, s, CH arom.), 7.06 (2H, m, CH arom.), 6.99 (2H, m, CH arom.), 6.90 (1H, t, CH arom.), 3.80 (3H, s, OCH3), 3.53 (2H, q, CH2 ) , 2.92 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 391.46; XH NMR: δΗ ppm 400 MHz, DMSO; 8.73
(1H, t, NH) , 8.39 (1H, s, CH arom.), 8.00 (2H, s, NH2) , 7.68 (1H, dt, CH arora. ) , 7.58 (1H, d, CH arom.), 7.31 (1H, d, CH arom.), 7.11 (4H, m, CH arom. ) , 6.93 (1H, t, CH arom.), 3.54 (2H, q, CH2) , 2.92 (2H, t, CH2) .
LCMS (IE, m/z) : (M+l) 324.08; 1H NMR: δΗ ppm 400 MHz, DMSO; 9.95 (1H, s, NH) , 8.37 (1H, s, CH arom.), 8.28 (1H, m, CH arom.), 8.16 (1H, dd, CH arom.), 7.89 (1H, m, CH arom.), 7.82 (2H, flattened s, NH2) , 7.85 (5H, m, CH arom.), 7.20 (1H, m, CH arom.) .
LCMS (IE, m/z) : (M+l) 324.08; XH NMR: δΗ ppm 400 MHz, DMSO; 11.39 (1H, s, NH) , 8.93 (1H, s, CH arom.), 8.35 (1H, m, CH arom.), 8.28 (1H, s, CH arom.), 8.15 (1H, d, CH arom.), 7.77 (2H, flattened s, NH2) , 7.41 (5H, m, CH arom.), 7.32 (1H, m, CH arom.) .
LCMS (IE, m/z) : (M+l) 348; XH NMR: δΗ ppm 400 MHz, DMSO; 10.50 (1H, s, NH) , 8.28 (1H, s, CH arom.), 8.27 (1H, m, CH arom.), 8.05
(1H, m, CH arom.), 7.78 (2H, s, NH2) , 7.59 (2H, m, CH arom.),
7.41 (4H, m, CH arom.), 7.32 (1H, m, CH arom.) .
2. Biological Testing of the compounds a) Plkl (Elisa Technology)
Substrate coated 96- ell microplates [Packard #6005182] are
generated by addition of 100 μΐ per well of a lmg/ml phosphate buffer saline (PBS) solution of dephosphorylated casein [Sigma # C-4032] for two hours under mild agitation. Casein solution is then replaced with blocking solution comprising 5% bovine serum albumin (BSA) [Sigma #B-7906] in PBS buffer and placed two hours under mild agitation.
Compound is added to the desired final concentration (typical range between 30 μΜ and 10 nM) and the enzymatic reaction is carried out for 30 minutes at 30°C under the following condition: Tris 20mM pH 7.5; MgCl2 lOmM; dithiothreitol (DTT) ImM ; β-glycerophosphate 40mM; sodium orthovanadate ImM; glycerol 2%; adenosine 5 ' -triphosphate (ATP) [Sigma #A-2383] 20μΜ and 0.5μ1 of purified PLK1 in a final volume of ΙΟΟμΙ per well. The level of PLKl-dependent casein phosphorylation is detected by sequential addition of ΙΟΟμΙ of anti-Phospho-Threonine antibody [Cell Signaling #9381] and Horseradish Peroxidase (HRP) conjugated anti-rabbit antibody [Amersham #NA934V] intertwined with washing steps in PBS tween-20 0.1%. Both antibodies are diluted to 1/1000 in 5% BSA PBS solution and left for incubation at room temperature for 1 hour. The reaction is finally quantified by addition of chemiluminescent HRP substrate SuperSignal ELISA Pico according to manufacturer's instruction [Pierce #37070] and detected with Envision plate reader [Perkin Elmer] . b) Plkl (KinaseGlo)
Compound is added to the desired final concentration (typical range between 30 μΜ and 10 nM) and the enzymatic reaction is carried out in 96-well plates for 120 minutes at 30°C under the following condition : MOPS 8mM pH7.1; EDTA 0.2mM; MnCl2 5mM; β-glycerophosphate 25mM; DTT ImM; sodium orthovanadate ImM; Triton X-100 0.01%; Casein 1.5mg/ml; ATP 10μΜ and 5μ1 of purified PLK1 in a final volume of 50μ1 per well. Upon completion, the enzymatic reaction is stopped by adding 50μ1 of Kinase-Glo
reagent (Promega) . The luminescent detection is performed according to manufacturer's instruction with Envision plate reader [Perkin Elmer] . c) Cellular Proliferation Assays
The antiproliferative activities on A549 (human lung adenocarcinoma epithelial cell line) , HCT116 (human colon cancer cell lines) and Namalwa (human Burkitt lymphoma cell lines) cell lines of the compounds were measured by the ATPlite technique (Perkin Elmer) .
Adherent cells (either from A549 or HCT116) and nonadherent cells (Namalwa cell lines) are transferred into 96-well plates (10 000 cells/ml for A549, 15 000 cells/ml for HCT116 and 300,000 cells/ml for Namalwa) at day 0 for A549 and HCT116, and day 1 for Namalwa, at a concentration compatible with logarithmic growth for the 72 hours required for the evaluation of the compounds. All of the cells are treated with the assayed molecules at day 1 and then placed in an incubator at 37 °C under an atmosphere of 5% C02. Cell viability is evaluated at day 4 by assaying released ATP, which is characteristic of viable cells. The ATPLite technique induces a luminescent signal read by TopCount microplate reader. IC50s are determined by nonlinear regression on the basis of a sigmoidal dose/response relationship model, wherein the Hill coefficient is left variable, carried out on the GraphPad (Prism) software package according to the algorithm provided . d) Results
Enzymatic inhibition Cellular proliferation
Molecule
assays assays
PLK-1 Elisa PLK-1 KGlo A549 HCT116 Namalwa
1001 B - C c -
1002 B - C c -
1003 A - c c C
1004 A - B B B
1005 A - C C C
1006 A - B A A
1007 A - B B B
1008 A - - B B
1009 A - B B -
1010 B - B B B
1011 A - B B B
1012 A - B B B
1013 A - - - B
1014 B - - - -
1015 A - B C B
1016 A - C C -
1017 B - C - -
1018 A - C D -
1019 A - C C -
1020 A - B B B
1021 A - C B C
1022 B - C - -
1023 A - - - C
1024 B - - B -
1025 A - c C c
1026 B - c C c
1027 B - - C c
1028 C - - B -
1029 B - - C c
1030 B - - - -
1031 B - D C c
1032 - B B - c
1033 - C - - -
1034 - D - - -
1035 - D - - -
1036 - C - - -
1037-7d- - C - - -
1038 - C - - -
1039-7b- - D - - -
1040 - C - - -
1041 B - c c c
1042 A - c B B
1043 A - B B -
1044 B - - D -
1045-7C- A A B B B
1046 B - - - -
1047 A - C - -
1048 A - A B B
1049 A - B B B
1050 A - B B B
1051 B - C C B
1052 A - C B B
1053 A - C B -
1054 B - - - -
1055 B - - - C
1056 A - C - C
1057 B - - - -
1058 B - - - -
1059 A - c C c
1060 - D - - -
1061 - C - - -
1062 A - c c c
1063 - C B - -
1064 B - C c -
1065 A - C B B
1066 C - - - -
1067 A - c - -
1068 B - - - -
1069 B - - - -
1070 B - - - -
1071 B - - - -
1072 - C - - -
1073 - D - - -
1074 - D - - -
1075-6b- - C - - -
1076-6C- - D - - -
1077-6a- - C - - -
1078-7a- - D - - -
1079 - D - - -
1080 - D - - -
1081 A - C C D
1082 A - A A A
1083 A - C B B
1084 A - C A A
1085 A - B B B
1086 A - B B B
1087 A - - C C
1088 A - B B B
1089 A - B B B
1090 A - B B B
1091 B - B B B
1092 A - B B B
1093 A - B B C
1094 A - A A A
1095 C - - C C
1096 B - - - -
1097 B - - - -
1098 A - C C c
1099 A - C c c
1100 A - B B B
1101 B - - - -
1102 A - C c
1103 A - C D C
1104 B - C D -
1105 B - - C -
1106 A - c C c
1107 C - - C -
1108 A - B B B
1109 A - B B B
1110 A - B B B
1111 - C - - -
1112 - C - - -
1113 - c - - -
1114 - c - - -
1115 - B - - -
1116 - B - - -
1117 - B - - -
1118 - C - - -
1119 - C - - -
Potencies are designated by IC50 binned into four activity classes as follows:
D: IC50 > 10μΜ; C: 10μΜ > IC50 > ΙμΜ; B: ΙμΜ > IC50 > ΙΟΟηΜ; A: IC50 < ΙΟΟηΜ; not conducted.
Claims
1. A compound of followin formula (I) :
or a pharmaceutically acceptable salt thereof, wherein:
• A is N or C-H,
• Y is 0, S or one of the following groups : sulfoxide (-S(O)-), sulfone (-S(0)2~) or N(R3) with R3 being H, or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl,
• Ri is H or an optionally substituted alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl or heteroaryl,
or when Y is NR3, Rx and R3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10- membered heterobicycle , and
• R2 is any of the optionally substituted following groups: alkyl, acyl, alkenyl, alkynyl, cycloalkyl, aralkyl, aryl, heteroaryl-alkyl or heteroaryl.
2. The compound according to claim 1, wherein Y is S, 0 or a sulfone group (-S(0)2 ~) .
3. The compound according to any one of claims 1 and 2, wherein Rx is an optionally substituted aryl or heteroaryl.
4. The compound according to any one of claims 1 to 3, wherein Rx is a pyridyl or phenyl, optionally substituted with one or more, notably one to ten, in particular one to five, more particularly one or two, substituent ( s ) , preferably selected from
the group consisting of: halogen, cyano (CN) , nitro (N02) , oxo (=0), R4, -0R5, -SR6, -NR7R8, -C (0) R9, -C (O) OR10, -C (0 ) NRnR12 , - 0C(0)Ri3, -NR14C (0) Ri5, -NRi6C (0) Ri7Ri8, -C (0 ) Ri9 R20R2i , and R22SO2R23; 4 to R23 being, independently of one another, an hydrogen atom or one of the following groups : alkyl preferably a (Ci-C6) alkyl , cycloalkyl, aryl, aralkyl, heterocycle, heterocycle- alkyl, and heteroaryl-alkyl , said group being optionally substituted with one or more, preferably one to five, notably one to three, halogen, (d-C6) alkyl , NR24R25 or OR26r 24 to R26 being an hydrogen atom or a (Ci-C6) alkyl .
5. The compound according to any one of claims 1 to 4, wherein Rx is a pyridyl or phenyl, optionally substituted with one or two substituent ( s ) selected from the group consisting of: halogen, nitro (N02) , R4, -0R5, -NR7R8, -C (0) RnRi2, -NR14C (0) Ri5, and -NRi6C (0) Ri7Ri8; R4, R5, R7, R8, Ru, R12 and Ri4 to Ri8 being, independently of one another, a hydrogen atom or one of the following groups: (Ci-C6) alkyl, cycloalkyl, heterocycle, and heterocycle-alkyl , said group being optionally substituted with one or more, preferably one to five, notably one to three, (Ci~ C6) alkyl, NR24R25 or OR26r 24 to R26 being a hydrogen atom or a (Ci~ C6) alkyl .
6. The compound according to claim 1, wherein Y is N (R3) and Ri and R3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle, preferably into a 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, more preferably into a heterobicycle selected from the group consisting of benzotriazole and benzimidazole .
7. The compound according to any one of claims 1 to 6, wherein R2 is a group selected from (Ci-C6) alkyl, aralkyl, aryl, heteroaryl, heterocycle-alkyl and heteroaryl-alkyl; said group
being optionally substituted with one or more, notably one to four, substituent ( s ) selected from the group consisting of: halogen, cyano (CN) , nitro (N02) , OXO (—0) , R-27, — OR-28, — SR-29, — NR30R31, -C(0) R32, -C(0) OR33, -C(0) NR34R35, -OC (0) R36, -NR37C (0) R38, - NR39C (0) NR40R41, -C (0) R42 R43R44, - R45SO2R46; R27 to R46 being, independently of one another, an hydrogen atom or one of the following groups: (Ci-C6) alkyl, cycloalkyl, aryl, aralkyl, heterocycle, heterocycle-alkyl , and heteroaryl-alkyl , said group being optionally substituted with one or more, preferably one to five, notably one to two, halogen, (d-C6) alkyl, NR47R48 or OR49, R47 to R49 being an hydrogen atom or a (Ci-C6) alkyl .
8. The compound according to any one of claims 1 to 7, wherein :
· A is N or C-H, preferably N;
• Y is S, 0 or a sulfone group (-S(0)2-), preferably S; and
• Ri is a phenyl, optionally substituted with one or two, substituent ( s ) , preferably selected from the group consisting of: halogen, nitro (N02) R4, -0R5, -NR7R8, -C (0) NRuR12, -NR14C (0) Ri5, and -NRi6C (0) Ri7Ri8; R4 to Ri8 being, independently of one another, an hydrogen atom or one of the following groups: a (Ci-C6) alkyl , cycloalkyl, heterocycle, and heterocycle-alkyl, said group being optionally substituted with one to three, halogen, (Ci~ C6) alkyl , R24R25 or OR26r R24 to R26 being an hydrogen atom or a (Ci~ C6) alkyl.
9. The compound according to any one of claims 1 to 7, wherein :
• A is N or CH, preferably N;
· Y is N (R3) ; and
• Ri and R3 are together included into an optionally substituted 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle, preferably into a 4 to 7-membered heterocycle or 8 to 10-membered heterobicycle comprising 1 to 3 nitrogen atoms, in particular a
heterobicycle selected from the group consisting of benzotriazole and benzimidazole.
10. The compound of formula (I) according to any one of claims 1 to 8 wherein:
• A is N;
• Y is 0;
• Ri is phenyl optionally substituted by one of the following groups: hydroxy, hydroxy-alkyl more particularly hydroxymethyl , and
O
II
C— NH2 ;
and
• R2 is as defined in claim 1.
11. The compound according to any one of claims 1 to 10, wherein it is selected from the following compounds:
an t e p armaceut ca y accepta e sa ts t ereo .
12. A pharmaceutical composition comprising at least one compound as defined in any one of claims 1 to 11 and at least one pharmaceutically acceptable carrier.
13. The pharmaceutical composition according to claim 12, wherein it further comprises another active principle, such as an anticancer agent.
14. A compound according to any one of claims 1 to 11 or a pharmaceutical composition according to claim 12 or 13, for use as a drug, in particular as PLK-1 inhibitors.
15. A compound according to any one of claims 1 to 11 or a pharmaceutical composition according to claim 12 or 13, for use for treating cancer.
16. A process to prepare a compound according to any one of claims 1 to 11, comprising the following steps:
(al) reacting a compound of followin formula (II) :
in which A, Y and Ri are as defined in claim 1, and Xi represents COOH or an activated form thereof,
with a compound of formula R2NH2 in which R2 is as defined in claim 1,
to give a compound of formula (I) as define in claim 1, and (bl) optionally salifying the compound of formula (I) obtained in step (al) to give a pharmaceutically acceptable salt of a compound of formula (I) .
17. A process to prepare a compound according to any one of claims 1 to 11, comprising the following steps:
in which A and Ri are as defined in claim 1, and X2 represents a halogen atom, such as CI , Br or I,
with a compound of formula RiYH in which Rx and Y are as defined in claim 1,
to give a compound of formula (I) as defined in claim 1, and
(b2) optionally salifying the compound of formula (I) obtained in step (a2) to give a pharmaceutically acceptable salt of a compound of formula (I) .
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201562183993P | 2015-06-24 | 2015-06-24 | |
| US62/183,993 | 2015-06-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016207345A1 true WO2016207345A1 (en) | 2016-12-29 |
Family
ID=56194501
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2016/064659 Ceased WO2016207345A1 (en) | 2015-06-24 | 2016-06-24 | 3-amino-pyrazin-2-yl carboxamide and 2-amino-pyridin-3-yl carboxamide derivatives as polo-like kinase 1 (plk-1) inhibitors for the treatment of cancer |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2016207345A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11613548B2 (en) | 2021-02-19 | 2023-03-28 | Sudo Biosciences Limited | Substituted pyridines, pyridazines, pyrimidines, and 1,2,4-triazines as TYK2 inhibitors |
| US12084458B2 (en) | 2021-02-19 | 2024-09-10 | Sudo Biosciences Limited | Substituted pyridines, pyridazines, and pyrimidines as TYK2 inhibitors |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3360517A (en) * | 1962-10-30 | 1967-12-26 | Merck & Co Inc | 3-amino-6-substituted-pyrazinoyl-guanidines |
| US5252538A (en) | 1984-05-21 | 1993-10-12 | American Cyanamid Company | (2-imidazolin-2-yl) fused heteropyridine compounds, intermediates for the preparation of and use of said compounds as herbicidal agents |
| WO2005079802A1 (en) * | 2004-02-12 | 2005-09-01 | Merck & Co., Inc. | Bipyridyl amides as modulators of metabotropic glutamate receptor-5 |
| EP1782811A1 (en) * | 2004-08-09 | 2007-05-09 | Eisai R&D Management Co., Ltd. | Novel antimalaria agent containing heterocyclic compound |
| EP2147914A1 (en) * | 2007-04-24 | 2010-01-27 | Shionogi&Co., Ltd. | Aminodihydrothiazine derivatives substituted with cyclic groups |
| EP2157090A1 (en) * | 2007-06-21 | 2010-02-24 | Taisho Pharmaceutical Co. Ltd. | Pyrazinamide compound |
| WO2011117254A1 (en) * | 2010-03-22 | 2011-09-29 | Prosidion Limited | Piperidine amides as modulators of the ghrelin receptor |
| WO2011143419A1 (en) * | 2010-05-12 | 2011-11-17 | Vertex Pharmaceuticals Incorporated | Pyrazines useful as inhibitors of atr kinase |
| WO2015011084A1 (en) * | 2013-07-23 | 2015-01-29 | Bayer Pharma Aktiengesellschaft | Substituted dihydropyrido[3,4-b]pyrazinones as dual inhibitors of bet proteins and polo-like kinases |
-
2016
- 2016-06-24 WO PCT/EP2016/064659 patent/WO2016207345A1/en not_active Ceased
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3360517A (en) * | 1962-10-30 | 1967-12-26 | Merck & Co Inc | 3-amino-6-substituted-pyrazinoyl-guanidines |
| US5252538A (en) | 1984-05-21 | 1993-10-12 | American Cyanamid Company | (2-imidazolin-2-yl) fused heteropyridine compounds, intermediates for the preparation of and use of said compounds as herbicidal agents |
| WO2005079802A1 (en) * | 2004-02-12 | 2005-09-01 | Merck & Co., Inc. | Bipyridyl amides as modulators of metabotropic glutamate receptor-5 |
| EP1782811A1 (en) * | 2004-08-09 | 2007-05-09 | Eisai R&D Management Co., Ltd. | Novel antimalaria agent containing heterocyclic compound |
| EP2147914A1 (en) * | 2007-04-24 | 2010-01-27 | Shionogi&Co., Ltd. | Aminodihydrothiazine derivatives substituted with cyclic groups |
| EP2157090A1 (en) * | 2007-06-21 | 2010-02-24 | Taisho Pharmaceutical Co. Ltd. | Pyrazinamide compound |
| WO2011117254A1 (en) * | 2010-03-22 | 2011-09-29 | Prosidion Limited | Piperidine amides as modulators of the ghrelin receptor |
| WO2011143419A1 (en) * | 2010-05-12 | 2011-11-17 | Vertex Pharmaceuticals Incorporated | Pyrazines useful as inhibitors of atr kinase |
| WO2015011084A1 (en) * | 2013-07-23 | 2015-01-29 | Bayer Pharma Aktiengesellschaft | Substituted dihydropyrido[3,4-b]pyrazinones as dual inhibitors of bet proteins and polo-like kinases |
Non-Patent Citations (3)
| Title |
|---|
| BUCHWALD S.L., ORG. LETT., vol. 4, no. 20, 2002, pages 3517 - 2520 |
| HETEROCYCLES, vol. 65, no. 10, 2005, pages 2321 - 2327 |
| SCOTT, JAMES S. ET AL: "Achieving improved permeability by hydrogen bond donor modulation in a series of MGAT2 inhibitors", MEDCHEMCOMM, vol. 4, no. 9, 2013, pages 1305 - 1311, XP002760150, ISSN: 2040-2503, DOI: 10.1039/c3md00156c * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11613548B2 (en) | 2021-02-19 | 2023-03-28 | Sudo Biosciences Limited | Substituted pyridines, pyridazines, pyrimidines, and 1,2,4-triazines as TYK2 inhibitors |
| US12084458B2 (en) | 2021-02-19 | 2024-09-10 | Sudo Biosciences Limited | Substituted pyridines, pyridazines, and pyrimidines as TYK2 inhibitors |
| US12103937B2 (en) | 2021-02-19 | 2024-10-01 | Sudo Biosciences Limited | Substituted pyridines and pyridazines as TYK2 inhibitors |
| US12122785B2 (en) | 2021-02-19 | 2024-10-22 | Sudo Biosciences Limited | Substituted pyridines, pyridazines, pyrimidines, and 1,2,4-triazines as TYK2 inhibitors |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2017279878B9 (en) | N-(substituted-phenyl)-sulfonamide derivatives as kinase inhibitors | |
| NO323952B1 (en) | Substituted indoles for modulating NFκB activity, method of preparation, drug comprising them, their use and a method for the preparation of a drug. | |
| CA2694284A1 (en) | Heterocyclic compounds useful as raf kinase inhibitors | |
| WO2006034317A2 (en) | Chemical compounds | |
| WO2017091661A1 (en) | Bicyclic bet bromodomain inhibitors and uses thereof | |
| WO2011082098A1 (en) | Lysine and arginine methyltransferase inhibitors for treating cancer | |
| KR20170118688A (en) | Methods and compositions for inhibition of bromodomain and extraterminal proteins | |
| CN113454081A (en) | Imidazopyridinyl compounds and their use for the treatment of proliferative diseases | |
| CN104497014A (en) | Chiral fluoroquinolone C-3 fused heterocycle alpha, beta-unsaturated ketone derivative as well as preparation method and application thereof | |
| ES2347185T3 (en) | NEW DERIVATIVES OF IMIDAZOLES, ITS PREPARATION AND ITS USE AS A MEDICINAL PRODUCT. | |
| US20080242685A1 (en) | Chemical Compounds | |
| CN104926788A (en) | Substituted piperidin derivative, and pharmaceutical composition containing substituted piperidin derivative and application thereof in antitumor | |
| KR101552361B1 (en) | Novel imidazolone derivatives, their pharmaceutical preparations as pharmaceutical compositions, and their use as protein kinase inhibitors, in particular as CDC7 inhibitors | |
| TW202003496A (en) | Novel phosphoinositide 3-kinase inhibitor and preparation method and use thereof | |
| CA2567569A1 (en) | Selective inhibitors against cdk4 and cdk6 having aminothiazole skeleton | |
| ZA200609759B (en) | Novel indazole carboxamides and their use | |
| JP2011518168A (en) | Inhibitors of phosphatidylinositol 3-kinase | |
| JP2009513677A (en) | Compound | |
| WO2009107767A1 (en) | Novel bicyclic pyrimidine derivative having antagonistic activity on h4 receptor | |
| WO2016207345A1 (en) | 3-amino-pyrazin-2-yl carboxamide and 2-amino-pyridin-3-yl carboxamide derivatives as polo-like kinase 1 (plk-1) inhibitors for the treatment of cancer | |
| JP2019001715A (en) | Tricyclic compound | |
| WO2019091277A1 (en) | 2-(1h-pyrazol-3-yl) phenol compound and use thereof | |
| CN107903208A (en) | A kind of biaryl pyridines deubiquitination enzyme inhibitor, its preparation method and application | |
| WO2007076286A2 (en) | Chemical compounds | |
| BR112020021922A2 (en) | 1h-indazole-3-carboxamide compound, use of 1h-indazole-3-carboxamide compounds, method of treating a pathological condition arising from uncontrolled activation and / or overexpression of gsk-3b, and pharmaceutical composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16731613 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 16731613 Country of ref document: EP Kind code of ref document: A1 |