WO2016171242A1 - Detection of epha2 - Google Patents
Detection of epha2 Download PDFInfo
- Publication number
- WO2016171242A1 WO2016171242A1 PCT/JP2016/062719 JP2016062719W WO2016171242A1 WO 2016171242 A1 WO2016171242 A1 WO 2016171242A1 JP 2016062719 W JP2016062719 W JP 2016062719W WO 2016171242 A1 WO2016171242 A1 WO 2016171242A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- epha2
- seq
- amino acid
- antigen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/10—Cells modified by introduction of foreign genetic material
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
Definitions
- the present invention introduces a novel antibody, a functional fragment of the antibody, a modified form of the antibody, a nucleotide containing a base sequence encoding the amino acid sequence of the antibody, a vector having the nucleotide inserted therein, the nucleotide or a vector.
- the present invention relates to a method for producing the antibody, a pharmaceutical composition, a diagnostic or test composition, and the like, which comprise a step of culturing the cell.
- EPHA2 is a receptor tyrosine kinase having a single transmembrane structure with a molecular weight of 130 kDa (Non-patent Document 1).
- EPHA2 has an N-terminal extracellular region with a ligand binding domain and two fibronectin type 3 domains, and a C-terminal intracellular region with a tyrosine kinase domain and a sterile- ⁇ -motif (SAM) domain.
- SAM sterile- ⁇ -motif
- Non-patent Document 2 Ephrin-A1 to A5 of GPI-anchored cell surface proteins are known (Non-patent Document 2). Activation of the tyrosine kinase domain of EPHA2 by ligand binding results in autophosphorylation of tyrosine residues present in the intracellular region of EPHA2, and induces intracellular signal transduction. In addition, it has been reported that EPHA2 is taken into cells by endocytosis after binding to a ligand and finally degraded by proteasome (Non-patent Document 3).
- EPHA2 is clinically highly expressed in many cancers, particularly breast cancer, esophageal cancer, prostate cancer, gastric cancer, non-small cell lung cancer, colon cancer, and glioblastoma multiforme (Non-Patent Documents 4 and 5). , 6, 7, 8, 9, 10, 11). Furthermore, in esophageal cancer, a significant correlation has been observed between EPHA2 expression and local lymph node metastasis, the number of lymph node metastases, and the low degree of differentiation of cancer, and the survival rate of EPHA2 positive patients is EPHA2 negative. It has been reported that it is lower than that of patients (Non-patent Document 5).
- the expression level of EPHA2 is higher in patients less than 5 years compared to patients whose disease-free survival is longer than 5 years, and those who relapse have higher expression levels of EPHA2 than those who do not relapse, In patients who have progressed to brain metastasis, it has been reported that the expression level of EPHA2 is higher than those in patients who have no recurrence or in patients whose metastasis is confined to the contralateral lung (Non-patent Document 9). It has been reported that the expression level of EPHA2 also significantly correlates with liver metastasis, lymphatic vessel invasion and clinical stage in colorectal cancer (Non-patent Document 10).
- non-cancerous cells can acquire cancer traits such as anchorage-independent growth ability, tubular morphogenesis on the extracellular matrix, and in vivo tumor growth ability.
- cancer traits such as anchorage-independent growth ability, tubular morphogenesis on the extracellular matrix, and in vivo tumor growth ability.
- Non-Patent Document 4 and cancer cells have been reported to be more invasive to the extracellular matrix (Non-Patent Documents 12 and 13).
- Non-Patent Documents 13 and 14 suppression of EPHA2 expression with siRNA suppresses cancer cell invasion, anchorage-independent growth, in vivo tumor growth
- Non-Patent Documents 13 and 14 ligand Ephrin-A1 EPHA2 is activated using a fusion protein of human and human IgG Fc region, and the degradation of EPHA2 by endocytosis suppresses the invasion, anchorage-independent growth, and tubular morphogenesis of cancer cells.
- EPHA2 has been reported to be expressed not only in cancer cells but also in blood vessels in or around tumors (Non-patent Document 15).
- EPHA2 signal is involved in angiogenesis induced by Ephrin-A1, especially that EPHA2 expressed in vascular endothelial cells is required for luminal formation and survival of vascular endothelial cells.
- a fusion protein of the extracellular region of EPHA2 and the Fc region of human IgG suppresses angiogenesis in vivo and exhibits an antitumor effect (Non-patent document 17).
- EPHA2 may be an excellent therapeutic target for cancer, and some monoclonal antibodies against EPHA2 have actually been acquired and clinical trials are being conducted (Patent Documents 1 to 3).
- the provision of a method capable of detecting the expression of EPHA2 is useful for testing or diagnosis of diseases related to EPHA2, such as cancer, and EPHA2 expression.
- Non-patent Document 18 Many monoclonal antibodies that recognize human EPHA2 are known, but there are few that can be applied to immunohistochemical staining, and monoclonal antibodies that can be immunostained by recognizing modified EPHA2 fixed with formalin. As such, only clones D7 and B2D6 (Non-patent Document 18) are known.
- One object of the present invention is to provide an antibody against EPHA2.
- Another object of the present invention is to provide a diagnostic or test composition containing an anti-EPHA2 antibody.
- the subject of the present invention also includes a nucleotide encoding the amino acid sequence of the antibody, a vector into which the nucleotide has been inserted, a cell into which the nucleotide or vector has been introduced, and a step of culturing the cell. Methods etc. are included.
- Another object of the present invention is to provide a pharmaceutical composition and a treatment method.
- the heavy chain sequence includes a variable region having CDRH1, CDRH2, and CDRH3, the CDRH1 is composed of an amino acid sequence represented by SEQ ID NO: 60, the CDRH2 is composed of an amino acid sequence represented by SEQ ID NO: 61, and the CDRH3 Consists of the amino acid sequence shown in SEQ ID NO: 62;
- the light chain sequence includes a variable region having CDRL1, CDRL2, and CDRL3, wherein the CDRL1 is composed of an amino acid sequence represented by SEQ ID NO: 63, the CDRL2 is composed of an amino acid sequence represented by SEQ ID NO: 64, and the CDRL3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 65; and specifically binding to EPHA2 shown in SEQ ID NO: 2;
- the CDRL1 is composed of an amino acid sequence represented by SEQ ID NO: 69
- the CDRL2 is composed of an amino acid sequence represented by SEQ ID NO: 70
- the CDRL3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 71; and specifically binding to EPHA2 shown in SEQ ID NO: 2;
- An antibody or an antigen-binding fragment of the antibody (5) A heavy chain variable region sequence consisting of amino acid residues 20 to 138 of the amino acid sequence shown in SEQ ID NO: 57 and a light chain variable consisting of amino acid residues 20 to 133 of the amino acid sequence shown in SEQ ID NO: 59
- (6) A heavy chain variable region sequence consisting of amino acid residues 20 to 138 of the amino acid sequence shown in SEQ ID NO: 57 and a light chain variable consisting of amino acid residues 20 to 133 of the amino acid sequence shown in SEQ ID NO: 59
- the test specimen is determined to be positive, and EPHA2 is not detected or measured in the test specimen, or the expression level or expression level of EPHA2 in the test specimen is equal to or lower than a predetermined criterion
- the subject from which the test specimen determined to be positive in the detection or measurement of EPHA2 by the test or diagnostic method for EPHA2-positive disease is an antibody that specifically binds to EPHA2, or an antigen-binding fragment of the antibody
- Gray dotted line and gray solid line indicate the binding of isotype control antibody and anti-FLAG antibody to 293F cells transfected with pFLAG-GW, respectively.
- the black dotted line and the black solid line indicate the binding of the isotype control antibody and the anti-FLAG antibody to 293F cells transfected with the gene shown in FIG.
- Evaluation of binding specificity of mouse anti-human EPHA2 antibody A201 by flow cytometry Gray dotted line and gray solid line indicate binding of isotype control antibody and A201 antibody to 293F cells transfected with pFLAG-GW, respectively.
- the black dotted line and the black solid line indicate the binding of the isotype control antibody and the A201 antibody to 293F cells transfected with the gene shown in FIG. Evaluation of Binding Specificity of Mouse Anti-Human EPHA2 Antibody A205 by Flow Cytometry
- Gray dotted line and gray solid line indicate the binding of isotype control antibody and A205 antibody to 293F cells transfected with pFLAG-GW, respectively.
- the black dotted line and the black solid line indicate the binding of the isotype control antibody and the A205 antibody to 293F cells transfected with the gene shown in FIG. 3, respectively. It is a figure which shows the EPHA2 specific binding property of the mouse
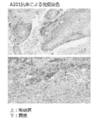
- FIG. 6 is an immunostained image of mouse throat and laryngeal cancer and gastric cancer according to Example 5 with mouse anti-human EPHA2 antibody A205. It is the figure which showed the amino acid sequence (sequence number 51) of the heavy chain of mouse
- FIG. 5 is an immunostained image of mouse anti-human EPHA2 antibodies A201 and A205 of human esophageal cancer according to Example 6, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924). .
- FIG. 5 is an immunostained image of mouse anti-human EPHA2 antibodies A201 and A205 of human esophageal cancer according to Example 6, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924). .
- FIG. 4 is an immunostaining image of mouse anti-EPHA2 antibodies A201 and A205 of human cervical cancer according to Example 6, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924).
- FIG. 9 shows immunostained images of mouse colonic cancer mouse anti-EPHA2 antibodies A201 and A205 according to Example 6, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924).
- FIG. 9 shows immunostained images of mouse colonic cancer mouse anti-EPHA2 antibodies A201 and A205 according to Example 6, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924).
- FIG. 9 shows immunostaining images of mouse gastric cancer mouse anti-EPHA2 antibodies A201 and A205 according to Example 6, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924).
- gene means a nucleotide containing a nucleotide sequence encoding a protein amino acid or a complementary strand thereof, for example, a nucleotide containing a nucleotide sequence encoding a protein amino acid or a complementary strand thereof.
- Certain polynucleotides, oligonucleotides, DNA, mRNA, cDNA, cRNA and the like are included in the meaning of “gene”.
- Such a gene is a single-stranded, double-stranded, or triple-stranded nucleotide, and an assembly of a DNA strand and an RNA strand, and ribonucleotide (RNA) and deoxyribonucleotide (DNA) are mixed on a single nucleotide strand.
- RNA ribonucleotide
- DNA deoxyribonucleotide
- EPHA2 gene include DNA, mRNA, cDNA, cRNA and the like containing a base sequence encoding the amino acid sequence of EPHA2 protein.
- nucleotide and “nucleic acid” are synonymous, and for example, DNA, RNA, probe, oligonucleotide, polynucleotide, primer and the like are also included in the meaning of “nucleotide”.
- a nucleotide is a nucleotide composed of a single strand, a double strand, or three or more strands, and an assembly of a DNA strand and an RNA strand, and ribonucleotide (RNA) and deoxyribonucleotide (DNA) on a single nucleotide strand.
- RNA ribonucleotide
- DNA deoxyribonucleotide
- nucleotide also included within the meaning of “nucleotide” are those that are intermingled and aggregates of two or more strands containing such nucleotide strands.
- polypeptide In the present invention, “polypeptide”, “peptide” and “protein” are synonymous.
- antigen is sometimes used to mean “immunogen”.
- cell includes various cells derived from individual animals, subculture cells, primary culture cells, cell lines, recombinant cells, microorganisms, and the like.
- an antibody that recognizes EPHA2 may be referred to as an “anti-EPHA2 antibody”.
- anti-EPHA2 antibody Such antibodies include chimerized antibodies, humanized antibodies, human antibodies and the like.
- the “functional fragment of an antibody” means an antibody fragment that exhibits at least a part of the function exhibited by the original antibody.
- the “functional fragment of an antibody” include, but are not limited to, Fab, F (ab ′) 2, scFv, Fab ′, single chain immunoglobulin and the like.
- Such functional fragments of antibodies are recombinant proteins produced in appropriate host cells using recombinant genes in addition to those obtained by treating full-length antibody protein molecules with enzymes such as papain and pepsin. May be.
- the “site” to which the antibody binds that is, the “site” recognized by the antibody means a partial peptide or a partial higher order structure on the antigen to which the antibody binds or recognizes. In the present invention, such a site is also referred to as an epitope or an antibody binding site.
- bonds or recognizes the partial peptide or partial higher order structure etc. on EPHA2 protein can be illustrated.
- CDRs complementarity determining regions
- the complementarity-determining region is also called a hypervariable domain, and is located in the variable region of the heavy and light chains of an antibody and has a particularly high primary structure variability. In the primary structure of the polypeptide chain, it is usually separated at three points.
- the complementarity determining region of an antibody the complementarity determining region of an antibody
- the complementarity determining region of the heavy chain is denoted as CDRH1, CDRH2, CDRH3 from the amino terminal side of the heavy chain amino acid sequence
- the complementarity determining region of the light chain is defined as the light chain amino acid.
- CDRL1, CDRL2, and CDRL3 are represented from the amino terminal side of the sequence. These sites are close to each other on the three-dimensional structure and determine the specificity for the antigen to be bound.
- antibody variant has an amino acid sequence in which amino acids are substituted, deleted, added and / or inserted (hereinafter collectively referred to as “mutation”) in the amino acid sequence of the original antibody. And a polypeptide that binds to the EPHA2 protein of the present invention.
- the number of variant amino acids in such antibody variants is 1 to 2, 1 to 3, 1 to 4, 1 to 5, 1 to 6, 1 to 7, 1 to 8, 1 to 9 1 to 10, 1 to 12, 1 to 15, 1 to 20, 1 to 25, 1 to 30, 1 to 40, or 1 to 50.
- Such antibody variants are also included in the “antibody” of the present invention. *
- “several” in “1 to several” refers to 3 to 10.
- Examples of the activity / property exhibited by the antibody of the present invention include biological activity, physicochemical properties, and the like. Specifically, various biological activities, binding activity to antigens and epitopes, stability during production and storage Property, heat stability, etc. can be raised.
- hybridize under stringent conditions means hybridization at 65 ° C. in a solution containing 5 ⁇ SSC, and then in an aqueous solution containing 2 ⁇ SSC-0.1% SDS. 20 minutes at 65 ° C. in an aqueous solution containing 0.5 ⁇ SSC-0.1% SDS for 20 minutes at 65 ° C. and 65 ° C. in an aqueous solution containing 0.2 ⁇ SSC-0.1% SDS Means to hybridize under conditions of washing for 20 minutes or under equivalent conditions.
- SSC is an aqueous solution of 150 mM NaCl-15 mM sodium citrate, and nx SSC means n-fold concentration of SSC.
- cytotoxicity refers to causing a pathological change in a cell in some form, and is not limited to direct trauma, but also includes DNA breakage, base dimer formation, chromosome breakage, It means any structural or functional damage of cells, such as damage to cell division apparatus or reduction of various enzyme activities.
- cytotoxic activity means causing the above cytotoxicity.
- antibody-dependent cytotoxic activity refers to “antibody dependent cellular cytotoxicity (ADCC) activity”, and means an activity in which NK cells damage target cells such as tumor cells via antibodies.
- antibody-dependent cell-mediated phagocytic activity refers to “antibody dependent cell phagocytosis (ADCP) activity”, which is an activity that monocytes and macrophage cells phagocytose target cells such as tumor cells via antibodies. means. Also referred to as “antibody-dependent phagocytic activity”.
- ADCP antibody dependent cell phagocytosis
- complement-dependent cytotoxic activity refers to “complement dependent cytotoxicity (CDC) activity” and means an activity that complements a target cell such as a tumor cell via an antibody.
- cancer and “tumor” are used interchangeably.
- immunohistochemistry means a histological (histochemical) method for detecting an antigen in a tissue specimen, and is synonymous with “immunoantibody method”. “immunostaining”) is also used interchangeably.
- denatured EPHA2 means an EPHA2 molecule in a specimen fixed with formalin. EPHA2 molecules in specimens fixed with formalin and then paraffin-treated and deparaffinized are also referred to as “denatured” EPHA2.
- non-denatured EPHA2 means EPHA2 in a sample not fixed with formalin.
- EPHA2 molecules in specimens not fixed with formalin are also referred to as “non-denaturing” EPHA2.
- the cell membrane fraction of the above-mentioned cells can be prepared and used, and it can be obtained by synthesizing EPHA2 in vitro or producing it in a host cell by genetic manipulation.
- EPHA2 cDNA is incorporated into a vector capable of expression and then synthesized in a solution containing enzymes, substrates and energy substances necessary for transcription and translation, or other prokaryote or eukaryote.
- the protein can be obtained by expressing EPHA2 by transforming a host cell of an organism.
- the nucleotide sequence of human EPHA2 cDNA is registered in GenBank with accession number: NM_004431. Further, the amino acid sequence of human EPHA2 is registered in GenBank with an accession number: NP_004422.
- EPHA2 cDNA is, for example, a polymerase chain reaction (hereinafter referred to as “PCR”) using a cDNA library of an organ expressing EPHA2 mRNA as a template and a primer that specifically amplifies EPHA2 cDNA (Saiki, R K., et al., Science, (1988) 239, 487-49).
- PCR polymerase chain reaction
- a polynucleotide that hybridizes with a polynucleotide comprising a nucleotide sequence complementary to the nucleotide sequence encoding human EPHA2 under stringent conditions and encodes a protein having biological activity equivalent to that of EPHA2, is also included in the EPHA2 cDNA. included.
- a polynucleotide encoding a splicing variant transcribed from the human EPHA2 locus or a protein that hybridizes to this under stringent conditions and also has a biological activity equivalent to that of EPHA2 is also included in the EPHA2 cDNA. included.
- EPHA2 amino acid sequence of human EPHA2
- amino acid sequence obtained by substituting, deleting, or adding 1, 2, or 3, or 4 or 5 amino acids in the amino acid sequence obtained by removing the signal sequence from these sequences a protein having a biological activity equivalent to that of EPHA2 is also included in EPHA2.
- an amino acid sequence encoded by a splicing variant transcribed from the human EPHA2 locus, or an amino acid sequence in which 1, 2 or 3 or 4 or 5 amino acids are substituted, deleted or added in the amino acid sequence EPHA2 is also included in the protein having a biological activity equivalent to that of EPHA2.
- the nucleotide sequence encoding mature human EPHA2 that does not contain a signal peptide is set forth in SEQ ID NO: 1 in the Sequence Listing, and the amino acid sequence of the human EPHA2 is set forth in SEQ ID NO: 2 in the Sequence Listing. Has been. 2.
- Production of anti-EPHA2 antibody The antibody against EPHA2 of the present invention is obtained by immunizing an animal with any polypeptide selected from the amino acid sequence of EPHA2 or EPHA2 using a conventional method, and collecting and purifying the antibody produced in vivo Can be obtained.
- the species of EPHA2 serving as an antigen is not limited to humans, and animals can be immunized with EPHA2 derived from animals other than humans such as mice and rats.
- an antibody applicable to a human disease can be selected by testing the cross-reactivity between the obtained antibody that binds to heterologous EPHA2 and human EPHA2.
- EPHA2 used as an antigen can be obtained by causing a host cell to produce the EPHA2 gene by genetic manipulation. Specifically, a vector capable of expressing the EPHA2 gene is prepared, introduced into a host cell to express the gene, and the expressed EPHA2 may be purified.
- the antibody against EPHA2 of the present invention can also be obtained using a DNA immunization method.
- the DNA immunization method is a technique for inducing immunity to an antigen by introducing an antigen expression plasmid into an individual animal such as a mouse or a rat and expressing the antigen in the individual.
- Gene transfer methods include direct injection of plasmids into muscle, intravenous injection of liposomes and polyethyleneimine and other introduction reagents, viral vector methods, and gold particles with plasmids attached to them by Gene Gun.
- the amount of expression is further improved by treating the muscle with hyaluronidase before intramuscular injection of the plasmid (McMahon JM1, Signori E, Wells KE, Fazio VM, Wells DJ. Gene Ther. 2001 Aug; 8 (16) : 1264-70).
- Hybridomas can be established by fusing antibody-producing cells that produce antibodies against EPHA2 and myeloma cells to obtain monoclonal antibodies. Specific examples of such a method are described in International Publication No. WO 09/48072 (published on April 16, 2009) and WO 10/11711 (published on October 14, 2010).
- Examples of the mouse anti-human EPHA2 antibody thus established include A201 antibody and A205 antibody.
- the amino acid sequence of the A201 antibody heavy chain is shown in SEQ ID NO: 51 of the Sequence Listing, and the nucleotide sequence encoding it is shown in SEQ ID NO: 50 of the Sequence Listing.
- the amino acid sequence of the A201 antibody light chain is shown in SEQ ID NO: 54 of the sequence listing, and the nucleotide sequence encoding it is shown in SEQ ID NO: 53 of the sequence listing.
- the amino acid sequence of the A205 antibody heavy chain is shown in SEQ ID NO: 57 in the sequence listing, and the nucleotide sequence encoding this is shown in SEQ ID NO: 56 in the sequence listing.
- the amino acid sequence of the A205 antibody light chain is shown in SEQ ID NO: 59 in the sequence listing, and the nucleotide sequence encoding it is shown in SEQ ID NO: 58 in the sequence listing.
- the antibody of the present invention may be an antibody that retains all six CDR sequences derived from A201 and has an activity of binding to EPHA2. That is, the heavy chain variable region of the antibody of the present invention is represented by CDRH1 (DTYVH) consisting of the amino acid sequence shown in SEQ ID NO: 60, CDRH2 (RIDPANANTKYDPKFQG) consisting of the amino acid sequence shown in SEQ ID NO: 61, and SEQ ID NO: 62. It possesses CDRH3 (YGKSAWFY) consisting of an amino acid sequence.
- the light chain variable region of the antibody comprises CDRL1 (RASQDIGNYLN) consisting of the amino acid sequence shown in SEQ ID NO: 63, CDRL2 (YTSRLHS) consisting of the amino acid sequence shown in SEQ ID NO: 64, and amino acids shown in SEQ ID NO: 65. It has CDRL3 (QQGHSLPPT) consisting of the sequence.
- CDRL1 RASQDIGNYLN
- YTSRLHS amino acid sequence shown in SEQ ID NO: 64
- QQGHSLPPT amino acid sequences of these CDRs are also shown in FIG.
- the antibody of the present invention may be an antibody that retains all six CDR sequences derived from A205 and has an activity of binding to EPHA2. That is, the heavy chain variable region of the antibody of the present invention is shown in CDRH1 (DYSMN) consisting of the amino acid sequence shown in SEQ ID NO: 66, CDRH2 (WINTYTGEPTYADDFKG) consisting of the amino acid sequence shown in SEQ ID NO: 67, and SEQ ID NO: 68. It possesses CDRH3 (PISLLLRLDY) consisting of an amino acid sequence.
- the light chain variable region of the above-mentioned antibody has CDRL1 (RSSQSLVHSNGNTYLH) consisting of the amino acid sequence shown in SEQ ID NO: 69, CDRL2 (KVSNRFS) consisting of the amino acid sequence shown in SEQ ID NO: 70, and amino acids shown in SEQ ID NO: 71. It possesses CDRL3 (SQSTHVPWT) consisting of the sequence.
- CDRL1 RSSQSLVHSNGNTYLH
- KVSNRFS the amino acid sequence shown in SEQ ID NO: 70
- SEQ ID NO: 71 amino acids shown in SEQ ID NO: 71.
- CDRL3 SQSTHVPWT
- suitable antibodies specifically bind to both undenatured human EPHA2 and denatured human EPHA2 in formalin-fixed specimens. More preferable antibodies include antibodies that specifically bind to both non-denatured human EPHA2 and denatured human EPHA2 in formalin-fixed specimens and that do not specifically bind to other Eph families. However, it is not limited to them.
- the antibodies of the present invention include genetically modified antibodies that have been artificially modified for the purpose of reducing heteroantigenicity against humans, such as chimeric antibodies, or humans. Also included are humanized antibodies. These antibodies can be produced using known methods.
- chimeric antibody examples include antibodies in which the variable region and the constant region of the antibody are different from each other, for example, a chimeric antibody in which the variable region of a mouse or rat-derived antibody is joined to a human-derived constant region (Proc. Natl. Acad). Sci.U.S.A., 81, 6851-6855, (1984)).
- the above-described chimeric antibody sequence against EPHA2 can be artificially modified for the purpose of, for example, reducing the heterologous antigenicity to humans, thereby producing a humanized antibody that is a recombinant antibody.
- the antibody of the present invention includes an antibody obtained by modifying the CDR of the humanized antibody. These antibodies can be produced using known methods.
- humanized antibodies antibodies (see Nature (1986) 321, p.522-525) in which only complementarity determining regions (CDRs) are incorporated into human-derived antibodies (see Nature (1986) 321, p.522-525), some sequences in addition to CDR sequences
- CDRs complementarity determining regions
- Examples of the amino acid residues of the framework include an antibody grafted on a human antibody (International Publication No. WO90 / 07861 pamphlet).
- the present invention also includes antibodies having such modifications, including deletions in which one or two amino acids have been deleted at the heavy chain carboxyl terminus, and such a deletions that have been amidated (eg, at the carboxyl terminus site).
- Heavy chain in which a proline residue is amidated e.g., at the carboxyl terminus site.
- the carboxyl-terminal deletion of the heavy chain of the antibody according to the present invention is not limited to the above type.
- the two heavy chains constituting the antibody according to the present invention may be either one of the full length and the heavy chain selected from the group consisting of the above-mentioned deletion forms, or a combination of any two of them. It may be a thing.
- the amount ratio of each deletion can be influenced by the type and culture conditions of the cultured mammalian cells that produce the antibody according to the present invention, but the main component of the antibody according to the present invention is a carboxyl in both two heavy chains. A case where one terminal amino acid residue is deleted can be mentioned.
- the antibody obtained by the above method can be evaluated for binding to an antigen and a suitable antibody can be selected.
- An example of another index for comparing antibody properties is antibody stability.
- Differential scanning calorimetry (DSC) is a method that can quickly and accurately measure the thermal denaturation midpoint (Tm), which is an indicator of good relative structural stability of proteins. The difference in thermal stability can be compared by measuring the Tm value using DSC and comparing the values. It is known that the storage stability of an antibody shows a certain degree of correlation with the thermal stability of the antibody (Lori Burton, et. Al., Pharmaceutical Development and Technology (2007) 12, p.265-273), and heat.
- a suitable antibody can be selected using stability as an index.
- Other indicators for selecting antibodies include high yields in appropriate host cells and low aggregation in aqueous solutions. For example, since the antibody with the highest yield does not always exhibit the highest thermal stability, it is necessary to select the most suitable antibody based on a comprehensive judgment based on the above-described indicators.
- the antibody of the present invention may be an antibody having a single heavy chain variable region and no light chain sequence.
- Such antibodies are called single domain antibodies (sdAbs) or nanobodies, and are actually observed in camels or llamas and reported to retain antigen-binding ability.
- sdAbs single domain antibodies
- nanobodies are actually observed in camels or llamas and reported to retain antigen-binding ability.
- the above-described antibody can also be interpreted as a kind of antigen-binding fragment of the antibody in the present invention.
- an antibody gene When an antibody gene is once isolated and then introduced into an appropriate host to produce an antibody, a combination of an appropriate host and an expression vector can be used.
- Specific examples of the antibody gene include a combination of a gene encoding the heavy chain sequence of the antibody described herein and a gene encoding the light chain sequence.
- the heavy chain sequence gene and the light chain sequence gene When transforming a host cell, the heavy chain sequence gene and the light chain sequence gene can be inserted into the same expression vector, or can be inserted into separate expression vectors. is there.
- eukaryotic cells When eukaryotic cells are used as hosts, animal cells, plant cells, and eukaryotic microorganisms can be used. Examples of animal cells include (1) mammalian cells such as COS cells (Gluzman, Y. Cell (1981) 23, p.
- ATCC CRL-1650 which are monkey cells, mouse fibroblasts NIH3T3 (ATCC). No. CRL-1658) and Chinese hamster ovary cells (CHO cells, ATCC CCL-61) dihydrofolate reductase-deficient strains (Urlauub, G. and Chasin, LA Proc. Natl. Acad. Sci. U.). S. A. (1980) 77, p. 4126-4220).
- Escherichia coli and Bacillus subtilis can be mentioned, for example.
- An antibody can be obtained by introducing a desired antibody gene into these cells by transformation, and culturing the transformed cells in vitro. In the above culture method, the yield may vary depending on the sequence of the antibody. From the antibodies having equivalent binding activity, those that can be easily produced as pharmaceuticals can be selected using the yield as an index.
- IgG IgG1, IgG2, IgG3, IgG4
- IgM IgA (IgA1, IgA2)
- IgD or IgE preferably IgG or IgM
- IgG1 or IgG2 More preferably, IgG1 or IgG2 can be mentioned.
- the antibody of the present invention may be an antigen-binding fragment of an antibody having an antigen-binding portion of the antibody or a modified product thereof.
- a fragment of the antibody can be obtained by treating the antibody with a proteolytic enzyme such as papain or pepsin, or modifying the antibody gene by a genetic engineering technique and expressing it in an appropriate cultured cell.
- a fragment that retains all or part of the functions of the full-length antibody molecule can be called an antigen-binding fragment of an antibody.
- Antibody functions generally include antigen-binding activity, activity that neutralizes antigen activity, activity that enhances antigen activity, antibody-dependent cytotoxic activity, complement-dependent cytotoxic activity, and complement-dependence Mention may be made of cellular cytotoxic activity.
- the function retained by the antigen-binding fragment of the antibody in the present invention is the binding activity to EPHA2.
- antibody fragments include Fab, F (ab ′) 2, Fv, or single chain Fv (scFv), diabodies (diabodies), linear antibody in which heavy and light chain Fvs are linked by an appropriate linker.
- Fab ' which is a monovalent fragment of the variable region of an antibody obtained by treating F (ab') 2 under reducing conditions, is also included in the antibody fragment.
- the antibody of the present invention may be a multispecific antibody having specificity for at least two different antigens.
- a molecule binds to two types of antigens (ie, bispecific antibodies), but the “multispecific antibody” in the present invention is more than that (for example, three types). It includes an antibody having specificity for the antigens.
- the multispecific antibody of the present invention may be a full-length antibody or a fragment of such an antibody (for example, F (ab ') 2 bispecific antibody).
- Bispecific antibodies can be prepared by combining the heavy and light chains (HL pairs) of two types of antibodies, or by hybridizing hybridomas that produce different monoclonal antibodies to produce a bispecific antibody. It can also be produced by producing cells (Millstein et al., Nature (1983) 305, p. 537-539).
- the antibody of the present invention may be a single chain antibody (also referred to as scFv).
- a single chain antibody is obtained by linking an antibody heavy chain variable region and a light chain variable region with a polypeptide linker (Pluckthun, The Pharmacology of Monoclonal Antibodies, 113 (Rosenberg and Moore, edited by Springer Verlag, New). York, p. 269-315 (1994), Nature Biotechnology (2005), 23, p. 1126-1136)
- a BiscFv fragment produced by linking two scFvs with a polypeptide linker is used as a bispecific antibody. It can also be used.
- the heavy chain variable region and the light chain variable region are linked via a linker that does not form a conjugate, preferably a polypeptide linker (Huston, JS et al., Proc. Natl. Acad. Sci.U.S.A. (1988), 85, p.5879-5883).
- the heavy chain variable region and the light chain variable region in scFv may be derived from the same antibody or different antibodies.
- the polypeptide linker that links the variable regions for example, any single chain peptide consisting of 12 to 19 residues is used.
- the DNA encoding the scFv is the DNA encoding the heavy chain or heavy chain variable region of the antibody, and the DNA encoding the light chain or light chain variable region.
- Amplification is performed by PCR using a coding DNA portion as a template and a primer pair defining both ends thereof, and then the DNA encoding the polypeptide linker portion and both ends thereof are connected to the heavy chain and light chain, respectively. Obtained by combining and amplifying the primer pairs defined in 1.
- an expression vector containing them and a host transformed with the expression vector can be obtained according to conventional methods, and by using the host, ScFv can be obtained according to the method.
- These antibody fragments can be produced by a host after obtaining and expressing the gene in the same manner as described above.
- the antibody of the present invention may be one that has been increased in quantity and has increased affinity for the antigen.
- the antibody that multiplies may be one type of antibody or a plurality of antibodies that recognize multiple epitopes of the same antigen. Examples of the method for increasing the amount of antibody include binding of IgG CH3 domain to two scFvs, binding to streptavidin, and introduction of helix-turn-helix motif.
- the antibody of the present invention may be a polyclonal antibody that is a mixture of a plurality of types of anti-EPHA2 antibodies having different amino acid sequences.
- a polyclonal antibody a mixture of plural kinds of antibodies having different CDRs can be mentioned.
- a polyclonal antibody a mixture of cells producing different antibodies can be cultured, and an antibody purified from the culture can be used (see WO 2004/061104).
- an antibody conjugated with various molecules such as polyethylene glycol (PEG) can also be used.
- PEG polyethylene glycol
- the antibody of the present invention may be one in which these antibody and another drug form a conjugate (Immunoconjugate).
- conjugate examples include those in which the antibody is bound to a radioactive substance or a compound having a pharmacological action (Nature Biotechnology (2005) 23, p. 1137-1146).
- the obtained antibody can be purified to homogeneity. Separation and purification of antibodies may be carried out using separation and purification methods used for ordinary proteins. For example, antibodies can be separated and purified by appropriately selecting and combining column chromatography, filter filtration, ultrafiltration, salting out, dialysis, preparative polyacrylamide gel electrophoresis, isoelectric focusing, etc. (Stratesies) for Protein Purification and Characterization: A Laboratory Course Manual, Daniel R.Marshak et al.eds, Cold Spring Harbor Laboratory Press (1996); Antibodies:. A Laboratory Manual.Ed Harlow and David Lane, Cold Spring Harbor Laboratory ( 988)) it is not intended to be limited thereto.
- chromatography examples include affinity chromatography, ion exchange chromatography, hydrophobic chromatography, gel filtration chromatography, reverse phase chromatography, and adsorption chromatography. These chromatography can be performed using liquid chromatography, such as HPLC and FPLC.
- column used for affinity chromatography include a protein A column and a protein G column.
- a column using a protein A column Hyper D, POROS, Sepharose F.R. F. (Pharmacia) and the like. It is also possible to purify an antibody using a carrier on which an antigen is immobilized, utilizing the binding property to the antigen.
- composition comprising an anti-EPHA2 antibody or a functional fragment thereof or a modified form thereof.
- the pharmaceutical composition of the present invention can be used in various diseases (hereinafter referred to as “induced or exacerbated” by EPHA2 signal abnormality or enhancement by EPHA2 or its ligand overexpression or EPHA2 mutation or gene amplification, or by switching of EPHA2 isoforms). , Referred to as “diseases associated with EPHA2”), especially for the treatment or prevention of various cancers.
- SNP single nucleotide substitution
- cancer types include lung cancer such as breast cancer, endometrial cancer, ovarian cancer, non-small cell lung cancer, gastric cancer, prostate cancer, kidney cancer, liver cancer, pancreatic cancer, colon cancer, esophageal cancer, bladder cancer, uterus.
- lung cancer such as breast cancer, endometrial cancer, ovarian cancer, non-small cell lung cancer, gastric cancer, prostate cancer, kidney cancer, liver cancer, pancreatic cancer, colon cancer, esophageal cancer, bladder cancer, uterus.
- Cervical cancer, hematological cancer, lymphoma, malignant melanoma and the like can be mentioned, and those cancers expressing the EPHA2 protein can be mentioned preferably.
- the onset of such a disease preferably the onset of such a disease in an individual expressing the EPHA2 protein, the suppression or inhibition of progression or progression, and the individual suffering from such a disease
- examples include, but are not limited to, alleviation of one or more symptoms present, suppression or remission of progression or progression, treatment or prevention of secondary diseases, and the like.
- the pharmaceutical composition of the present invention comprises a therapeutically or prophylactically effective amount of an anti-EPHA2 antibody or a functional fragment of the antibody and a pharmaceutically acceptable diluent, carrier, solubilizer, emulsifier, preservative and / or adjuvant. Can be contained.
- “Therapeutically or prophylactically effective amount” means an amount that exhibits a therapeutic or prophylactic effect for a specific disease, administration form, and administration route, and is synonymous with “pharmacologically effective amount”.
- the pharmaceutical composition of the present invention has pH, osmotic pressure, viscosity, transparency, color, isotonicity, sterility, stability of the composition or antibody contained therein, solubility, sustained release, absorbability, penetration.
- Substances for changing, maintaining, and maintaining properties, dosage forms, strength, properties, shapes, etc. can be included.
- the substance for the preparation is not particularly limited as long as it is a pharmacologically acceptable substance.
- non-toxicity or low toxicity is a property that a substance for preparation preferably comprises.
- Substances for formulation include, for example, amino acids, antibacterial agents, antioxidants, buffers, fillers, chelating agents, complexing agents, bulking agents, monosaccharides, disaccharides, carbohydrates, coloring agents, flavoring agents, diluents , Emulsifier, hydrophilic polymer, preservative, solvent, sugar alcohol, suspending agent, surfactant, stabilization enhancer, elasticity enhancer, transport agent, diluent, excipient, and / or pharmaceutical adjuvant
- the amount of these substances to be added is 0.001 to 1000 times, preferably 0.01 to 100 times, more preferably the weight of the anti-EPHA2 antibody or functional fragment or modified product thereof. Is 0.1 to 10 times.
- a pharmaceutical composition containing an anti-EPHA2 antibody or a functional fragment thereof or a modified product thereof in an liposome, or an antibody modified product (US Pat. No. 6,214,388, etc.) in which an antibody and a liposome are bound is also present. Included in the pharmaceutical composition of the invention.
- the excipient or carrier is usually liquid or solid, and is not particularly limited as long as it is a substance used for water for injection, physiological saline, artificial cerebrospinal fluid, and other preparations for oral administration or parenteral administration.
- physiological saline include neutral ones and those containing serum albumin.
- the buffer examples include Tris buffer prepared so that the final pH of the pharmaceutical composition is 7.0 to 8.5, acetate buffer prepared so as to be 4.0 to 5.5, and 5. Examples thereof include a citrate buffer prepared to be 0 to 8.0, a histidine buffer prepared to be 5.0 to 8.0, and the like.
- the pharmaceutical composition of the present invention is a solid, liquid, suspension or the like. Freeze-dried preparations can be mentioned. An excipient such as sucrose can be used to mold the lyophilized preparation.
- the administration route of the pharmaceutical composition of the present invention may be any of enteral administration, topical administration and parenteral administration.
- enteral administration intravenous administration, intraarterial administration, intramuscular administration, intradermal administration, subcutaneous administration, intraperitoneal administration Administration, transdermal administration, intraosseous administration, intraarticular administration and the like can be mentioned.
- composition of such a pharmaceutical composition can be determined according to the administration method, the EPHA2 protein binding affinity of the antibody, and the like.
- the dose of the anti-EPHA2 antibody of the present invention is not limited as long as it is a pharmacologically effective amount, and the species of the individual, the type of disease, the symptom, sex, age, prevalence, EPHA2 protein binding affinity of the antibody or Although it can be appropriately determined depending on the biological activity and other factors, it is usually 0.01 to 1000 mg / kg, preferably 0.1 to 100 mg / kg once every 1 to 180 days, or 1 It can be administered twice or more times a day.
- the form of the pharmaceutical composition includes injections (including lyophilized preparations and infusions), suppositories, nasal absorption preparations, transdermal absorption preparations, sublingual preparations, capsules, tablets, ointments, granules, aerosols. Examples thereof include pills, pills, powders, suspensions, emulsions, eye drops, and implantable preparations.
- a pharmaceutical composition comprising an anti-EPHA2 antibody or a functional fragment thereof or a modified form thereof as an active ingredient can be administered simultaneously with or separately from other drugs.
- a pharmaceutical composition containing an anti-EPHA2 antibody or a functional fragment of the antibody as an active ingredient is administered, or after administering such a pharmaceutical composition, another pharmaceutical is administered, or You may administer the said pharmaceutical composition and another pharmaceutical simultaneously.
- other medicaments include various anticancer agents such as chemotherapeutic agents and radiotherapy. These are collectively referred to as “the combined use of the antibody of the present invention with another drug”, and a pharmaceutical composition containing an additional drug in addition to the antibody of the present invention, a functional fragment thereof or a modified form thereof is also included in the present invention.
- the present invention relates to a method for treating or preventing a disease associated with EPHA2, such as cancer, the use of the antibody of the present invention for preparing a pharmaceutical composition for treating or preventing the disease, and the present invention for treating or preventing the disease.
- a disease associated with EPHA2 such as cancer
- the use of the antibody of the present invention for preparing a pharmaceutical composition for treating or preventing the disease and the present invention for treating or preventing the disease.
- a therapeutic or prophylactic kit containing the antibody of the present invention is also included in the present invention.
- Diagnostic Composition A diagnostic or diagnostic composition (hereinafter collectively referred to as “diagnostic composition”) comprising the anti-EPHA2 antibody of the present invention or a functional fragment thereof or a modified form thereof is provided.
- the diagnostic composition of the present invention is useful for testing or diagnosis of diseases related to EPHA2, such as cancer, and EPHA2 expression.
- the examination or diagnosis in the present invention includes, for example, determination or measurement of morbidity risk, determination of the presence or absence of morbidity, measurement of the degree of progression or deterioration, measurement or determination of the effect of drug treatment with a pharmaceutical composition such as an anti-EPHA2 antibody.
- measurement or determination of the effect of treatment other than drug treatment measurement of recurrence risk, determination of the presence or absence of recurrence, and the like are included, but the test or diagnosis is not limited thereto.
- the diagnostic composition of the present invention is useful for identification of an individual to which the antibody of the present invention or a functional fragment thereof or a modified product thereof, a composition containing them, or a pharmaceutical composition containing them is administered.
- Such a diagnostic composition may contain a pH buffer, an osmotic pressure regulator, salts, a stabilizer, a preservative, a developer, a sensitizer, an aggregation inhibitor, and the like.
- the present invention relates to a method for testing or diagnosing a disease associated with EPHA2, such as cancer, use of the antibody of the present invention for preparing a diagnostic composition for the disease, use of the antibody of the present invention for testing or diagnosing the disease , Also provide.
- a test or diagnostic kit containing the antibody of the present invention is also included in the present invention.
- a sandwich ELISA is preferable as a test or diagnostic method including the antibody of the present invention, but a normal ELISA method, RIA method, ELISPOT (Enzyme-Linked ImmunoSpot) method, dot blot method, octalony method, CIE (Counterimmunoelectrophoresis) method, CLIA Detection methods using antibodies such as (Chemiluminescent immunoassay) and FCM (Flow Cytometry) can be used.
- labeling method for antibodies in addition to biotin, labeling methods that can be used for biochemical analysis such as labels for fluorophores such as HRP, alkaline phosphatase, FITC and ALEXA, and radioisotopes can be used.
- TMB (3,3 ′, 5,5′-tetramethylbenzidine), BCIP (5-bromo-4-chloro-3-indoyl phosphate), ⁇ -NPP ( ⁇ -nitrophenyl phosphate), OPD Fluorescent substrates such as (O-Phenylenediamine), ABTS (3-Ethylbenzothiazine-6-sulfonic acid), SuperSignal ELISA Pico Chemiluminescent Substrate TM (Sermo Fisher Scientific) Substrate In addition, it is possible to use chemiluminescent substrate.
- BCIP 5-bromo-4-chloro-3-indoyl phosphate
- ⁇ -NPP ⁇ -nitrophenyl phosphate
- OPD Fluorescent substrates such as (O-Phenylenediamine), ABTS (3-Ethylbenzothiazine-6-sulfonic acid), SuperSignal ELISA Pico Chemiluminescent Subst
- test samples derived from living organisms include, but are not limited to, blood, joint fluid, ascites, lymph, cerebrospinal fluid, tissue homogenate supernatant, tissue section, and the like.
- the test or diagnostic sandwich ELISA kit containing the antibody of the present invention may contain an EPHA2 protein standard solution, a coloring reagent, a dilution buffer, a solid phase antibody, a detection antibody, a washing solution, and the like.
- an absorption method, a fluorescence method, a luminescence method, an RI (Radioisotope) method or the like is preferably applied.
- An RI liquid scintillation counter or the like is preferably used.
- a soluble protein is prepared from cells, tissues or organs or a part thereof in a sample according to a conventional method, and the soluble protein is reacted with a labeled antibody in the soluble protein. It can also be used in Western blotting or dot blotting to confirm the presence or absence of EPHA2.
- the present invention provides antibodies useful for immunohistochemistry (IHC) analysis, functional fragments thereof and modifications thereof, and compositions containing them. Such a composition is also included in the “diagnostic composition” of the present invention.
- IHC immunohistochemistry
- Immunohistochemistry is not particularly limited as long as it is a technique in which a tissue section is reacted with an antibody (primary antibody) that binds to an antigen to detect the primary antibody bound to the antigen.
- Tissue sections are preferably paraffin-embedded after formalin fixation. After embedding in paraffin, the sliced tissue section is deparaffinized and then subjected to antigen activation treatment and nonspecific reaction suppression treatment.
- the antigen activation treatment method include heat treatment, enzyme treatment with protease, etc., and heat treatment is preferred.
- As conditions for the heat treatment a temperature range of 90 to 110 ° C., a pH of 8 to 10 and a treatment time of 20 to 60 minutes are usually preferable.
- Tris-EDTA buffer for example, 10 mM Tris buffer containing 1 mM EDTA
- a method of inactivating an endogenous enzyme having a catalytic activity similar to or similar to the enzyme used for color development is usually used.
- H 2 O 2 As the H 2 O 2 solvent, water, methanol or the like can be used, and the concentration of H 2 O 2 is 0.1 to 3%, preferably 0.3 to 3%.
- Sodium azide can be added to the H 2 O 2 solution.
- a method of blocking with serum or casein can also be used as a nonspecific reaction suppression treatment.
- Serum and casein can treat tissues before the primary antibody reaction, but can also be included in a solvent that dilutes the primary antibody.
- the reaction conditions for the primary antibody are not particularly limited, but the temperature is 4 to 50 ° C, preferably 20 to 37 ° C, more preferably 24 ° C.
- the reaction time is 5 minutes to 1 day, preferably 10 minutes to 4 hours, more preferably 30 minutes to 1 hour.
- an antibody that can be visualized and binds to the primary antibody can be preferably used.
- the reaction is performed three or more times using an antibody (tertiary antibody) that binds to the secondary antibody itself.
- the secondary or tertiary antibody can be visualized by binding an enzyme such as peroxidase or alkaline phosphatase to these antibodies, or by adding biotin or the like to these antibodies and binding to the aforementioned enzymes such as streptavidin.
- a method of reacting a chromogenic substrate corresponding to those enzymes can be preferably used.
- Examples of a method for binding an enzyme to a secondary antibody or a tertiary antibody include a method using a reagent in which a large number of the enzyme and secondary antibody are bound to a dextrin polymer or an amino acid polymer (polymer method).
- polymer method a method of reacting a biotinylated secondary antibody and peroxidase-labeled streptavidin (LSAB method), DAB or the like can be used as a chromogenic substrate.
- a secondary antibody labeled with a fluorescent dye or the like can also be used. When treated with a fluorescently labeled secondary antibody, positive cells are detected using a fluorescence microscope after the treatment.
- the isolated cells are applied to glass or separated by a centrifugal separator, divided into cell components and liquid components, and immunostaining is performed on the cell components. That is, cell components can be applied on a slide glass and fixed with an ethanol solution or a 10% formalin solution, and then immunostaining similar to a tissue section can be performed.
- the excised tissue is rapidly frozen with liquid nitrogen after embedding with an OCT compound and sliced with a cryostat to prepare a slide specimen. After fixing this specimen with 10% formalin or ethanol solution, immunostaining similar to the tissue section can be performed.
- the operation related to immunohistochemistry can be performed automatically by programming the reaction solution, reaction conditions, number of washings, etc., and incorporating it into the immune device.
- diagnostic imaging label the antibody with a pharmaceutically acceptable radionuclide or illuminant, administer the antibody to a subject, take an image using diagnostic imaging techniques such as PET / CT, and then the presence of EPHA2 Can be determined or inspected.
- the antibody, functional fragment thereof or modified product thereof contained in the diagnostic composition of the present invention is preferably an antibody that binds to or recognizes EPHA2, that is, an antibody having EPHA2 selectivity, a functional fragment thereof, or a modified product thereof.
- antibodies having human EPHA2 selectivity include heavy chain CDRH1 to CDRH3 of the mouse A201 antibody (amino acid sequence shown in SEQ ID NOs: 60 to 62 (FIG. 11)), and light chain CDRL1 to CDRRL3 ( An antibody comprising a light chain comprising the amino acid sequence shown in SEQ ID NO: 63 to 65 (FIG. 11), a heavy chain variable region of the mouse A201 antibody (amino acid sequence shown in amino acid numbers 20 to 137 of SEQ ID NO: 51) and light Examples include an antibody containing a chain variable region (amino acid sequence shown at amino acid numbers 20 to 128 of SEQ ID NO: 54), an antibody containing the heavy and light chains of mouse A201 antibody, and the like. Examples of such antibodies include mouse A201 antibody and chimeric A201 antibody, but are not limited thereto.
- Antibodies having human EPHA2 selectivity include heavy chain CDRH1 to CDRH3 (amino acid sequence shown in SEQ ID NOs: 66 to 68 (FIG. 12)) of mouse A205 antibody, and light chain CDRL1 to CDRRL3 ( An antibody comprising a light chain comprising the amino acid sequence shown in SEQ ID NOs: 69 to 71 (FIG. 12), a heavy chain variable region of the mouse A205 antibody (amino acid sequence shown in amino acids 20 to 138 of SEQ ID NO: 57), and Examples include an antibody containing a light chain variable region (amino acid sequence shown in amino acid numbers 20 to 133 of SEQ ID NO: 59), an antibody containing the heavy chain and light chain of mouse A205 antibody, and the like. Examples of such antibodies include, but are not limited to, mouse A205 antibody and chimeric A205 antibody.
- the diagnostic composition is for EPHA2 detection or measurement.
- the present invention provides a method for detecting or measuring human EPHA2 in a test sample.
- the diagnostic composition of the present invention can be used for these detection or measurement methods. Such measurement methods and diagnostic compositions are also included in the present invention for diagnosis or testing of human EPHA2-positive cancer.
- a method for identifying an individual to which the pharmaceutical composition of the present invention is administered is also encompassed by the present invention.
- human EPHA2 in the sample derived from the individual is measured, and human EPHA2 is detected in the sample, or compared with the amount of human EPHA2 detected in the sample derived from a healthy individual. If many human EPHA2s are detected, the individual can be determined as positive.
- the diagnostic composition of the present invention can be used.
- the individual is suffering from or at risk of having cancer.
- the pharmaceutical composition of the present invention can be administered to an individual who has been determined to be positive by such an identification method.
- Reagent The antibody of the present invention or a functional fragment thereof or a modified form thereof is also useful as a reagent. Such reagents are used in the above-described examination or diagnostic, research and other applications.
- Example 1 Production of Mouse Anti-Human EPHA2 Antibody 1 -1 Immunization Fcgr2b KO (BALB / c) Homo mouse female (Immuno-Biological Laboratories) was used for immunization. A mixture of Recombinant Human EPHA2 (R & D SYSTEM; 3035-A2) and Freund's Complete Adjuvant (Wako Pure Chemical Industries) was collected subcutaneously and used for hybridoma production.
- Example 2 Evaluation of antigen-binding ability of mouse anti-human EPHA2 antibody 2) -1 Construction of vector expressing human EPHA2 and protein having sequence similarity to human EPHA2 2) -1-1 Construction of expression vector pFLAG- Reading frame of Gateway Vector Conversion System (Life Technologies, Thermo Fisher Scientific) on blind-ended HindIII and BglII sites of myc-CMV19 (Sigma). This vector was named “pFLAG-GW”.
- the gene encoding EPHA2 was transferred from this entry vector to pFLAG-GW by LR recombination reaction.
- the resulting vector was named pFLAG-EPHA2.
- the amino acid sequence of SEQ ID NO: 1 is shown in SEQ ID NO: 2.
- PTPRS-2 A DNA fragment (SEQ ID NO: 39) encoding human PTPRS isoform 2 (hereinafter referred to as PTPRS-2) without a signal sequence was used as a primer set 5'-GGGACAAGTTTGTACAAAAAAAGCAGCCTTCGAGT 3 ′ (PTPRS-2 / 4-Fw: SEQ ID NO: 41) 5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTTAGGTTGCATAGTGTCAAAG-3 ′ (PTPRS-2 / 4-Re: SEQ ID NO: 42) And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction.
- PTPRF-2-Fw SEQ ID NO: 47
- PTPRF-2-Re SEQ ID NO: 48
- This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction.
- the gene encoding PTPRF-2 was transferred from this entry vector to pFLAG-GW by LR recombination reaction.
- the resulting vector was named pFLAG-PTPRF-2.
- the amino acid sequence of SEQ ID NO: 45 is shown in SEQ ID NO: 46.
- Sections are prepared from these blocks and using an automated immunostaining device Ventana Discovery ULTRA and DAB map kit (above, Roche Diagnostics) or Autostainer Link 48 and EnVisionFLEX Mini Kit (DAKO) using the instrument's instructions.
- IHC screening of the antibody contained in the culture supernatant derived from was performed. First, among the 67 hybridoma-derived culture supernatants obtained in Example 1) -2, 12 antibodies that react strongly with EPHA2-expressing 293 ⁇ cells and do not react with non-transfected 293 ⁇ cells were obtained. Selected. Further, 6 cells that react with MDA-MB-231 cells were selected, and 3 cells that strongly reacted with endogenous EPHA2 were selected.
- Example 2 Three antibodies selected in 2-1 were purified from the culture supernatant of hybridomas producing these antibodies, Example 2) -2-1 In the same manner as human esophageal cancer (CR2), stomach cancer (CQ2), colon cancer (CDA3), cervical cancer (CZA2), lung cancer (CCA4), pharyngeal laryngeal cancer (CH3), malignant melanoma (LM481) and commercially available paraffin sections of normal tissues (MAN, MBN, MCN) (above, SuperBioChip Laboratories) were stained. Among the three, A201 and A205 that responded strongly to tumor cells and had low background staining were selected.
- A201 and A205 which are mouse anti-human EPHA2 antibodies, were determined by the Mouse monoclonal isotyping test kit (AbD Serotec). As a result, it was shown that the isotype of A201 is IgG1, and the isotype of A205 is IgG2b.
- pFLAG-PTPRS-4, pFLAG-PTPRF-2 and pFLAG-GW as controls were transfected into 293F cells using 293fectin (Life Technologies, Thermo Fisher Scientific), 37 ° C., 125 rpm, 8% CO 2, respectively. Cultured overnight under 2 conditions. The next day, these 293F cells were collected, washed, and then used for flow cytometry analysis.
- each cell prepared by the method shown in Example 2) -4 was treated with A201, A205, a mouse IgG control antibody (Becton Dickinson), and Anti-FLAG M2 antibody (Sigma-Aldrich) were added and incubated at 4 ° C. for 30 minutes. These cells were washed twice with PBS containing 5% FBS, Anti-Mouse IgG FITC conjugate (MP Bio) diluted 500 times with PBS containing 5% FBS was added, and incubated at 4 ° C. for 20 minutes.
- A205 binds to EPHA2, while for molecules with sequence similarity to EPHA2 (EPHA3, EPHA5, EPHA6, EPHA7, EPHAB1, EPHAB3, EPHB4, PTPRS-2, PTPRS-4, and PTPRF-2) It was shown not to bind ( Figure 3). A205 also bound to cells transfected with the negative control pFLAG-GW, which was presumed to be due to A205 binding to EPHA2 that is endogenously expressed in 293F cells. .
- mG1VR1 SEQ ID NO: 49
- mG1VR1 was designed from the sequence of the constant region of mouse heavy chain (IgG1) in the database.
- cDNA encoding the heavy chain fragment of A201 was amplified by 5'-RACE PCR. This PCR was performed using a touch-down PCR program according to the manual of SMARTER RACE cDNA Amplification Kit using KOD-Plus- (TOYOBO) as Polymerase.
- a cDNA encoding a heavy chain fragment amplified by 5'-RACE PCR is purified using MinElute PCR Purification Kit (QIAGEN), then cloned using Zero Blunt TOPO PCR Cloning Kit (Invitrogen), and cloned cDNA was sequenced.
- 5'-CATCCCCAGGGTCACCCATGGAGTTTAGTTTG-3 '(mG1VR1: SEQ ID NO: 49) and NUP (Nested Universal Primer A: attached to SMARTER RACE cDNA Amplification Kit) were used.
- the sequencing reaction was performed with GeneAmp 9700 (Applied Biosystems), and the DNA sequencing was performed with ABI PRISM 3700 DNA Analyzer (Applied Biosystems) or Applied Biosystems 3730xxl Analyzer (Applied Biosystems).
- the nucleotide sequence of the cDNA encoding the determined heavy chain of A201 is shown in SEQ ID NO: 50, and the amino acid sequence is shown in SEQ ID NO: 51.
- the amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 51 corresponds to the signal sequence of the A201 heavy chain, and the amino acid sequence consisting of the 20th to 137th amino acid residues corresponds to the heavy chain variable region of A201.
- SEQ ID NO: 50 and SEQ ID NO: 51 are also shown in FIG.
- cDNA encoding the light chain fragment of A201 was amplified by 5'-RACE PCR. This PCR was performed using a touch-down PCR program according to the manual of SMARTER RACE cDNA Amplification Kit using KOD-Plus- (TOYOBO) as Polymerase.
- a cDNA encoding a light chain fragment amplified by 5'-RACE PCR is purified using MinElute PCR Purification Kit (QIAGEN), then cloned using Zero Blunt TOPO PCR Cloning Kit (Invitrogen), and cloned cDNA was sequenced.
- 5'-AGTCCAACTGTTCAGGACGCCCATTTGTCG-3 '(mKVR2: SEQ ID NO: 52) and NUP (Nested Universal Primer A: attached to SMARTER RACE cDNA Amplification Kit) were used.
- the sequencing reaction was performed with GeneAmp 9700 (Applied Biosystems), and the DNA sequencing was performed with ABI PRISM 3700 DNA Analyzer (Applied Biosystems) or Applied Biosystems 3730xxl Analyzer (Applied Biosystems).
- the determined nucleotide sequence of cDNA encoding the light chain of A201 is shown in SEQ ID NO: 53 of the Sequence Listing, and the amino acid sequence is shown in SEQ ID NO: 54.
- the amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 54 corresponds to the signal sequence of the A201 light chain, and the amino acid sequence consisting of the 20th to 128th amino acid residues corresponds to the light chain variable region of A201.
- SEQ ID NO: 53 and SEQ ID NO: 54 are also shown in FIG.
- mG2bVR2 was designed from the sequence of the constant region of the mouse heavy chain (IgG2b) in the database.
- 5′-TGCACACTGCTGGACAGGGGATCCAGAGTTC-3 ′ (mG2bVR2: SEQ ID NO: 55) and NUP (Nested Universal Primer A: attached to SMARTER RACE cDNA Amplification Kit) were used.
- the nucleotide sequence of the cDNA encoding the determined heavy chain of A205 is shown in SEQ ID NO: 56 in the sequence listing, and the amino acid sequence is shown in SEQ ID NO: 57.
- the amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 57 corresponds to the signal sequence of the A205 heavy chain, and the amino acid sequence consisting of the 20th to 138th amino acid residues corresponds to the heavy chain variable region of A205.
- SEQ ID NO: 56 and SEQ ID NO: 57 are also shown in FIG.
- Example 3 3) -2-4 Amplification of cDNA encoding light chain fragment of A205 by 5'-RACE PCR and determination of nucleotide sequence Example 3) cDNA synthesized in 2-2 (5'-RACE-Ready cDNA) Using the template, the cDNA encoding the light chain fragment of antibody A205 was amplified and sequenced in the same manner as in Example 3) -1-4.
- the nucleotide sequence of the determined cDNA encoding the light chain of A205 is shown in SEQ ID NO: 58 in the sequence listing, and the amino acid sequence is shown in SEQ ID NO: 59.
- the amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 59 corresponds to the signal sequence of the A205 light chain, and the amino acid sequence consisting of the 20th to 133rd amino acid residues corresponds to the light chain variable region of A205.
- SEQ ID NO: 58 and SEQ ID NO: 59 are also shown in FIG.
- Example 4 Production of mouse anti-human EPHA2 antibody
- Mouse anti-human EPHA2 antibodies A201 and A205 were purified from the hybridoma culture supernatant.
- A201 and A205-producing hybridomas were grown to a sufficient amount with ClonCell-HY Selection Medium E, and then Ultra Low IgG FBS (Life Technologies, ThermoFisher Scifi) was added to 20% of the world's TiMeMeSriMiMeFriSriMeMriFiSriMiSriFiSriMiSriMiSriMiSriFiSriMiSriFlM. The medium was changed to Fisher Scientific) and cultured for 5 days. The main culture supernatant was collected and sterilized through a 0.45 ⁇ m filter.
- the antibody was purified from the above hybridoma supernatant by Protein G affinity chromatography (under 4-6 ° C.) in a one-step process.
- the buffer substitution step after protein G affinity chromatography purification was performed at 4 to 6 ° C.
- the culture supernatant of the hybridoma was applied to a column packed with Protein G (GE Healthcare Bioscience) equilibrated with 0.2 M sodium phosphate buffer (pH 7.0). After all of the culture supernatant liquid entered the column, the column was washed with a sodium phosphate buffer having a column volume of 2 times or more.
- FIG. 4 shows the specificity of the mouse anti-EPHA2 antibody in immunohistochemical staining.
- the Eph family molecules shown in FIG. 4 were transiently expressed in 293F cells, and the pellets of these cells were embedded in paraffin after formalin fixation.
- Sections prepared from these blocks were subjected to antigen activation treatment with Tris-EDTA solution (DAKO) at 97 ° C for 40 minutes, endogenous peroxidase (HRP) blocking reagent (DAKO) 5 minutes, nonspecific protein adsorption blocking reagent (DAKO) After reacting in order of 30 minutes, mouse anti-EPHA2 antibody A201 or A205 prepared to a concentration of 2.5 ⁇ g / mL with an antibody diluent (DAKO) was reacted for 1 hour. Immunohistochemical staining was performed by a conventional method using an HRP-labeled universal secondary antibody (DAKO) and DAB chromogenic substrate (DAKO).
- FIG. 5 shows the results of immunohistochemical staining with mouse anti-EPHA2 antibody A201.
- a commercially available human micropharyngeal cancer and gastric cancer tissue microarray (SuperBioChips Laboratories) was subjected to antigen activation treatment with a Tris-EDTA solution (DAKO) at 97 ° C. for 40 minutes, and an endogenous peroxidase (HRP) blocking reagent (DAKO) After reacting in the order of 5 minutes and non-specific protein adsorption blocking reagent (DAKO) for 30 minutes, mouse anti-EPHA2 antibody A201 prepared to a concentration of 5 ⁇ g / mL with an antibody diluent (DAKO) was reacted for 1 hour.
- DAKO endogenous peroxidase
- FIG. 5 is a photomicrograph showing specific staining of part of the cell membrane and cytoplasm of tumor cells of laryngeal squamous cell carcinoma and gastric adenocarcinoma by mouse anti-EPHA2 antibody A201. There is no staining in the tissues and cells of the tumor stroma. Tumor tissue and stromal tissue can be distinguished by morphological differences.
- FIG. 6 shows the results of immunohistochemical staining with mouse anti-EPHA2 antibody A205.
- a commercially available human micropharyngeal cancer and gastric cancer tissue microarray (SuperBioChips Laboratories) was subjected to an antigen activation treatment with a Tris-EDTA solution (DAKO) at 97 ° C. for 40 minutes, and an endogenous HRP blocking reagent (DAKO) 5 minutes.
- DAKO Tris-EDTA solution
- DAKO endogenous HRP blocking reagent
- mouse anti-EPHA2 antibody A205 prepared to a concentration of 15 ⁇ g / mL with an antibody diluent (DAKO) was reacted for 1 hour.
- FIG. 6 is a photomicrograph showing specific staining of the cell membrane and part of the cytoplasm of tumor cells of laryngeal squamous cell carcinoma and gastric adenocarcinoma by the mouse anti-EPHA2 antibody A205. There is no staining in the tissues and cells of the tumor stroma. Tumor tissue and stromal tissue can be distinguished by morphological differences.
- mice anti-EPHA2 antibodies A201 and A205 The same site is stained with mouse anti-EPHA2 antibodies A201 and A205, but A201 is more sensitive than A205, and more tumor cells are stained.
- FIGS. 13 and 14 show the staining conditions and staining properties of the anti-EPHA2 antibody in immunohistochemical staining.
- EPHA2 non-expressing 293 ⁇ cells, EPHA2 overexpressing 293 ⁇ cells, and EPHA2 endogenously expressed MDA-MB-231 cell pellets were embedded in paraffin after formalin fixation. Sections prepared from these blocks were subjected to antigen activation treatment with Tris-EDTA solution (DAKO or Roche Diagnostics) at 97 ° C. for 36 to 40 minutes, and endogenous peroxidase (HRP) blocking, non-blocking was performed by a conventional method.
- Tris-EDTA solution DAKO or Roche Diagnostics
- concentration optimized by preliminary examination was made to react for 1 hour.
- immunohistochemical staining was carried out by the LSAB method or the polymer method using a kit of Roche Diagnostics or DAKO. By microscopic observation, a positive positive reaction was observed in the cell membrane or cytoplasm, and a positive positive reaction was observed when a weak positive reaction was observed.
- Both A201 and A205 antibodies show strong positive reaction to both EPHA2 overexpressing 293 ⁇ cells and EPHA2 endogenously expressed MDA-MB-231 cells, and recognize EPHA2 with higher sensitivity than various commercially available EPHA2 antibodies compared. Was confirmed (FIG. 13).
- FIG. 14 is a photomicrograph of cells. In all antibodies, no positive reaction is observed in EPHA2 non-expressing 293 ⁇ cells, whereas in EPHA2 overexpressing 293 ⁇ cells, a strong positive reaction is observed in the cell membrane and cytoplasm.
- the mouse anti-EPHA2 antibody (D4) manufactured by Millipore high non-specific staining was observed throughout the cells.
- 15 to 18 show the results of immunohistochemical staining with mouse anti-EPHA2 antibodies A201 and A205, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924).
- a commercially available human tumor tissue microarray (SuperBioChips Laboratories) was subjected to antigen activation treatment with a Tris-EDTA solution (DAKO) at 97 ° C. for 40 minutes, blocked in the same manner as described above, and then diluted with an antibody diluent (DAKO). Each anti-EPHA2 antibody diluted in 1) was reacted for 1 hour.
- Immunohistochemical staining was performed by a conventional method using an HRP-labeled universal secondary antibody (DAKO) or an HRP-labeled anti-goat IgG antibody (Nichirei Bioscience) or a DAB chromogenic substrate (DAKO).
- DAKO an anti-mouse linker antibody
- 15 to 18 are micrographs of tumor tissues, which are stained images of human esophageal cancer (FIG. 15), cervical cancer (FIG. 16), colon cancer (FIG. 17), and stomach cancer (FIG. 18).
- A201 antibody and A205 antibody showed specific staining in the cell membrane and part of cytoplasm of tumor cells, and A201 was more sensitive than A205, and more tumor cells were stained. Yes. There is little or no specific staining with the AF3035 and sc-924 antibodies.
- the A201 antibody, A205 antibody, and AF3035 antibody show no staining in the tissues and cells of the tumor stroma, but the sc-924 antibody also shows a cross reaction in the non-tumor cells of the tumor stroma. Tumor tissue and stromal tissue can be distinguished by morphological differences.
- the use of the antibody provided by the present invention enables examination or diagnosis of various cancers.
- SEQ ID NO: 1 Nucleotide sequence encoding human EPHA2 SEQ ID NO: 2: Amino acid sequence of human EPHA2 SEQ ID NO: 3: PCR primer EPHA2-Fw SEQ ID NO: 4: PCR primer EPHA2-Re SEQ ID NO: 5: nucleotide sequence encoding human EPHA3 SEQ ID NO: 6: amino acid sequence of human EPHA3 SEQ ID NO: 7: PCR primer EPHA3-Fw SEQ ID NO: 8: PCR primer EPHA3-Re SEQ ID NO: 9: nucleotide sequence encoding human EPHA4 SEQ ID NO: 10: amino acid sequence of human EPHA4 SEQ ID NO: 11: PCR primer EPHA4-Fw SEQ ID NO: 12: PCR primer EPHA4-Re SEQ ID NO: 13: nucleotide sequence encoding human EPHA5 SEQ ID NO: 14: amino acid sequence of human EPHA5 SEQ ID NO: 15: PCR primer EPHA5-Fw SEQ ID NO: 16
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Biomedical Technology (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Microbiology (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Cell Biology (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- General Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Analytical Chemistry (AREA)
- Food Science & Technology (AREA)
- Pathology (AREA)
- Mycology (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Peptides Or Proteins (AREA)
Abstract
Description
本発明は、新規な抗体、該抗体の機能断片、該抗体の修飾体、該抗体の有するアミノ酸配列をコードする塩基配列を含むヌクレオチド、該ヌクレオチドが挿入されたベクター、該ヌクレオチドまたはベクターが導入された細胞、該細胞を培養する工程を含む該抗体の製造方法、医薬組成物、診断用または検査用組成物等に関する。 The present invention introduces a novel antibody, a functional fragment of the antibody, a modified form of the antibody, a nucleotide containing a base sequence encoding the amino acid sequence of the antibody, a vector having the nucleotide inserted therein, the nucleotide or a vector. The present invention relates to a method for producing the antibody, a pharmaceutical composition, a diagnostic or test composition, and the like, which comprise a step of culturing the cell.
EPHA2は、分子量130kDaの1回膜貫通構造を有するレセプター型チロシンキナーゼである(非特許文献1)。EPHA2のN末端側の細胞外領域には、リガンド結合ドメインと2ヶ所のフィブロネクチンタイプ3ドメインが存在し、C末端側の細胞内領域にはチロシンキナーゼドメインとsterile-α-motif(SAM)ドメインが存在している。 EPHA2 is a receptor tyrosine kinase having a single transmembrane structure with a molecular weight of 130 kDa (Non-patent Document 1). EPHA2 has an N-terminal extracellular region with a ligand binding domain and two fibronectin type 3 domains, and a C-terminal intracellular region with a tyrosine kinase domain and a sterile-α-motif (SAM) domain. Existing.
EPHA2のリガンドとして、GPIアンカー型細胞表面蛋白質のEphrin-A1~A5が知られている(非特許文献2)。リガンド結合によるEPHA2のチロシンキナーゼドメインの活性化により、EPHA2の細胞内領域に存在するチロシン残基が自己リン酸化され、細胞内シグナルの伝達が誘導される。また、リガンドと結合した後にEPHA2はエンドサイトーシスにより細胞内に取り込まれ、最終的にプロテアソームで分解されることが報告されている(非特許文献3)。 As a ligand for EPHA2, Ephrin-A1 to A5 of GPI-anchored cell surface proteins are known (Non-patent Document 2). Activation of the tyrosine kinase domain of EPHA2 by ligand binding results in autophosphorylation of tyrosine residues present in the intracellular region of EPHA2, and induces intracellular signal transduction. In addition, it has been reported that EPHA2 is taken into cells by endocytosis after binding to a ligand and finally degraded by proteasome (Non-patent Document 3).
EPHA2は臨床的に多くの癌、特に乳癌、食道癌、前立腺癌、胃癌、非小細胞肺癌、大腸癌、多形神経膠芽腫での高発現が報告されている(非特許文献4、5、6、7、8、9、10、11)。さらに、食道癌では、EPHA2発現と局所リンパ節転移、リンパ節転移の数、及び癌の分化度の低さとの間に有意な相関が観察されており、EPHA2陽性の患者の生存率はEPHA2陰性の患者に比べて低いことが報告されている(非特許文献5)。非小細胞肺癌では、無病生存期間が5年より長い患者に比べ5年未満の患者ではEPHA2の発現レベルが高いこと、再発した患者は再発のない患者に比べてEPHA2の発現レベルが高いこと、脳転移へ進行した患者では、再発のない患者や転移が対側の肺に限局している患者に比べてEPHA2の発現レベルが高いことが報告されている(非特許文献9)。大腸癌においてもEPHA2の発現レベルが、肝転移、リンパ管浸潤及び臨床ステージと有意に相関することが報告されている(非特許文献10)。
EPHA2 is clinically highly expressed in many cancers, particularly breast cancer, esophageal cancer, prostate cancer, gastric cancer, non-small cell lung cancer, colon cancer, and glioblastoma multiforme (Non-Patent
また、EPHA2遺伝子を細胞に導入することによって、非癌細胞が足場非依存的増殖能や細胞外基質上での管状形態形成能、in vivoでの腫瘍増殖能といった癌形質を獲得するようになること(非特許文献4)、癌細胞の細胞外基質に対する浸潤性が亢進すること(非特許文献12、13)が報告されている。逆にEPHA2の発現をsiRNAで抑制すると癌細胞の浸潤性や足場非依存的増殖、in vivoでの腫瘍増殖が抑制されること(非特許文献13、14)、および、リガンドであるEphrin-A1とヒトIgGのFc領域との融合蛋白質を使用してEPHA2を活性化し、エンドサイトーシスによるEPHA2の分解を誘導することで癌細胞の浸潤性や足場非依存的増殖、管状形態形成能が抑制されること(非特許文献4、11、12)が報告されている。
Also, by introducing the EPHA2 gene into cells, non-cancerous cells can acquire cancer traits such as anchorage-independent growth ability, tubular morphogenesis on the extracellular matrix, and in vivo tumor growth ability. (Non-Patent Document 4) and cancer cells have been reported to be more invasive to the extracellular matrix (Non-Patent Documents 12 and 13). Conversely, suppression of EPHA2 expression with siRNA suppresses cancer cell invasion, anchorage-independent growth, in vivo tumor growth (Non-Patent Documents 13 and 14), and ligand Ephrin-A1 EPHA2 is activated using a fusion protein of human and human IgG Fc region, and the degradation of EPHA2 by endocytosis suppresses the invasion, anchorage-independent growth, and tubular morphogenesis of cancer cells (Non-Patent
一方でEPHA2は、癌細胞のみならず腫瘍内または腫瘍周囲の血管にも発現していることが報告されている(非特許文献15)。マウスにおいてはEphrin-A1によって誘導される血管新生にEPHA2シグナルが関与していること、特に血管内皮細胞に発現するEPHA2が血管内皮細胞の管腔形成や生存に必要とされることが報告されており(非特許文献16)、EPHA2の細胞外領域とヒトIgGのFc領域との融合蛋白質がin vivoで血管新生を抑制し、抗腫瘍効果を示すことも報告されている(非特許文献17)。 On the other hand, EPHA2 has been reported to be expressed not only in cancer cells but also in blood vessels in or around tumors (Non-patent Document 15). In mice, it has been reported that EPHA2 signal is involved in angiogenesis induced by Ephrin-A1, especially that EPHA2 expressed in vascular endothelial cells is required for luminal formation and survival of vascular endothelial cells. It has also been reported that a fusion protein of the extracellular region of EPHA2 and the Fc region of human IgG suppresses angiogenesis in vivo and exhibits an antitumor effect (Non-patent document 17). .
以上より、EPHA2が癌に対する優れた治療標的となる可能性が示唆されており、実際にEPHA2に対するモノクローナル抗体が取得され、臨床試験が実施されているものも存在する(特許文献1乃至3)。
From the above, it is suggested that EPHA2 may be an excellent therapeutic target for cancer, and some monoclonal antibodies against EPHA2 have actually been acquired and clinical trials are being conducted (
これらこのことから、EPHA2の発現を検出できる方法の提供は、癌などEPHA2に関わる疾患、EPHA2発現の検査または診断に有用である。 From these facts, the provision of a method capable of detecting the expression of EPHA2 is useful for testing or diagnosis of diseases related to EPHA2, such as cancer, and EPHA2 expression.
ヒトEPHA2を認識するモノクローナル抗体は多数知られているが、免疫組織染色への適用が可能とされるものは少なく、さらにホルマリンで固定した変性型EPHA2認識による免疫組織染色が可能とされるモノクローナル抗体としては、僅かにクローンD7やB2D6(非特許文献18)が知られているに過ぎなかった。 Many monoclonal antibodies that recognize human EPHA2 are known, but there are few that can be applied to immunohistochemical staining, and monoclonal antibodies that can be immunostained by recognizing modified EPHA2 fixed with formalin. As such, only clones D7 and B2D6 (Non-patent Document 18) are known.
本発明の一つの課題は、EPHA2に対する抗体を提供することである。 One object of the present invention is to provide an antibody against EPHA2.
本発明の他の一つの課題は抗EPHA2抗体を含有する診断または検査用組成物等を提供することである。 Another object of the present invention is to provide a diagnostic or test composition containing an anti-EPHA2 antibody.
また、本発明の課題には、該抗体が有するアミノ酸配列をコードするヌクレオチド、該ヌクレオチドが挿入されたベクター、該ヌクレオチドまたはベクターが導入された細胞、該細胞を培養する工程を含む該抗体の製造方法等が含まれる。 The subject of the present invention also includes a nucleotide encoding the amino acid sequence of the antibody, a vector into which the nucleotide has been inserted, a cell into which the nucleotide or vector has been introduced, and a step of culturing the cell. Methods etc. are included.
さらに、本発明の他の一つの課題は、医薬組成物および治療方法を提供することである。 Furthermore, another object of the present invention is to provide a pharmaceutical composition and a treatment method.
発明者は上記課題を解決するために鋭意、検討を行い、新規な抗EPHA2抗体を創出し、該抗体を用いてEPHA2を検出できることを見出し、本発明を完成させた。すなわち、本発明は、以下の発明を包含する。
(1)重鎖配列が、CDRH1、CDRH2、CDRH3を有する可変領域を含み、前記CDRH1は配列番号60に示されるアミノ酸配列からなり、前記CDRH2は配列番号61に示されるアミノ酸配列からなり、前記CDRH3は配列番号62に示されるアミノ酸配列からなること;
軽鎖配列が、CDRL1、CDRL2、CDRL3を有する可変領域を含み、前記CDRL1は配列番号63に示されるアミノ酸配列からなり、前記CDRL2は配列番号64に示されるアミノ酸配列からなり、前記CDRL3は配列番号65に示されるアミノ酸配列からなること;および
配列番号2に示されるEPHA2に特異的に結合すること;
を特徴とする抗体または該抗体の抗原結合性断片、
(2)配列番号51に示されるアミノ酸配列の20乃至137番目のアミノ酸残基からなる重鎖可変領域配列および配列番号54に示されるアミノ酸配列の20乃至128番目のアミノ酸残基からなる軽鎖可変領域配列を含むことを特徴とする(1)に記載の抗体または該抗体の抗原結合性断片、
(3)配列番号51に示されるアミノ酸配列の20乃至137番目のアミノ酸残基からなる重鎖可変領域配列および配列番号54に示されるアミノ酸配列の20乃至128番目のアミノ酸残基からなる軽鎖可変領域配列を含み、重鎖定常領域のアイソタイプがIgG1であることを特徴とする(1)または(2)に記載の抗体または該抗体の抗原結合性断片、
(4)重鎖配列が、CDRH1、CDRH2、CDRH 3を有する可変領域を含み、前記CDRH1は配列番号66に示されるアミノ酸配列からなり、前記CDRH2は配列番号67に示されるアミノ酸配列からなり、前記CDRH3は配列番号68に示されるアミノ酸配列からなること;
軽鎖配列が、CDRL1、CDRL2、CDRL3を有する可変領域を含み、前記CDRL1は配列番号69に示されるアミノ酸配列からなり、前記CDRL2は配列番号70に示されるアミノ酸配列からなり、前記CDRL3は配列番号71に示されるアミノ酸配列からなること;および
配列番号2に示されるEPHA2に特異的に結合すること;
を特徴とする抗体または該抗体の抗原結合性断片、
(5)配列番号57に示されるアミノ酸配列の20乃至138番目のアミノ酸残基からなる重鎖可変領域配列および配列番号59に示されるアミノ酸配列の20乃至133番目のアミノ酸残基からなる軽鎖可変領域配列を含むことを特徴とする(4)に記載の抗体または該抗体の抗原結合性断片、
(6)配列番号57に示されるアミノ酸配列の20乃至138番目のアミノ酸残基からなる重鎖可変領域配列および配列番号59に示されるアミノ酸配列の20乃至133番目のアミノ酸残基からなる軽鎖可変領域配列を含み、重鎖定常領域のアイソタイプがIgG2bであることを特徴とする(4)または(5)に記載の抗体または該抗体の抗原結合性断片、
(7)Fab、F(ab’)2、Fab’およびFvからなる群から選択される、(1)、(2)、(4)または(5)のいずれか一つに記載の抗体の抗原結合性断片、
(8)scFvであることを特徴とする、(1)、(2)、(4)または(5)のいずれか一つに記載の抗体、
(9)(1)乃至(8)のいずれか一つに記載の抗体または該抗体の抗原結合性断片を含む組成物、
(10)(1)乃至(8)のいずれか一つに記載の抗体または該抗体の抗原結合性断片を含み、パラフィン包埋処理した後脱パラフィン処理した組織標本(以下、単に「標本」という。)中のEPHA2の検出または測定方法に使用される、(9)に記載の組成物、
(11)(1)乃至(8)のいずれか一つに記載の抗体または該抗体の抗原結合性断片と被検標本を接触させる工程を含む、標本中のEPHA2の検出または測定方法に使用される、請求項(9)または(10)に記載の組成物、
(12)EPHA2の検出または測定方法が、被検標本においてEPHA2が検出もしくは測定されたか、または被検標本におけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより高い場合、該被検標本を陽性と判定し、被検標本においてEPHA2が検出もしくは測定されなかったか、または被検標本におけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより低い場合、該被検標本を陰性と判定する工程を含む、(11)に記載の組成物、
(13)EPHA2陽性疾患の検査または診断方法に使用される、(9)乃至(12)のいずれか一つに記載の組成物、
(14)EPHA2陽性疾患の検査または診断方法が、EPHA2の検出または測定において、陽性と判定された被検標本が由来する被験者は、EPHA2に特異的に結合する抗体もしくは該抗体の抗原結合性断片を投与する工程を含むEPHA2陽性疾患の治療または予防方法に適していると判定し、陰性と判定された被検標本が由来する被験者は、EPHA2に特異的に結合する抗体もしくは該抗体の抗原結合性断片を投与する工程を含むEPHA2陽性疾患の治療または予防方法には適していないと判定することを含む、(13)に記載の組成物、
(15)EPHA2陽性疾患がEPHA2陽性癌である、(13)または(14)に記載の組成物、
(16)下記(i)乃至(iii)のいずれか一つに記載の被験者に投与される、EPHA2に特異的に結合する抗体もしくは該抗体の抗原結合性断片を含む医薬組成物:
(i)(9)乃至(11)または(13)のいずれか一つに記載の組成物を用いてEPHA2が検出または測定された被検標本の由来する被験者;
(ii)(12)に記載の組成物を用いたEPHA2の検出または測定において陽性と判定された被検標本の由来する被験者;
(iii)(14)または(15)に記載の組成物を用いて、EPHA2に特異的に結合する抗体もしくは該抗体の抗原結合性断片を投与する工程を含むEPHA2陽性疾患の治療または予防に適していると判定された被験者、
(17)(1)乃至(8)のいずれか一つに記載の抗体または該抗体の抗原結合性断片をコードするポリヌクレオチド、
(18)(17)に記載のポリヌクレオチドを含むベクター、
(19)(17)に記載のポリヌクレオチドまたは(18)に記載のベクターを含む細胞、
(20)下記の工程(i)および(ii)を含む、(1)乃至(8)のいずれか一つに記載のモノクローナル抗体または該抗体の抗原結合性断片の製造方法:
(i)(19)に記載の細胞を培養する工程;
(ii)工程(i)の培養物からモノクローナル抗体または該抗体の抗原結合性断片を回収する工程、
(21)抗体がモノクローナル抗体である、(1)乃至(8)のいずれか一つに記載の抗体又はその抗原結合性断片、
(22)下記の工程(i)、または(i)および(ii)を含む、EPHA2の検出または測定方法:
(i)(1)乃至(8)のいずれか一つに記載の抗体または該抗体の抗原結合性断片又は(9)に記載の組成物と被検標本を接触させる工程;
(ii)該被検標本においてEPHA2が検出もしくは測定されたか、または該被検標本におけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより高い場合、該被検標本を陽性と判定し、被検標本においてEPHA2が検出もしくは測定されなかったか、また該被検標本におけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより低い場合、該被検標本を陰性と判定する工程、
(23)下記の工程(i)、または(i)および(ii)を含む、EPHA2に特異的に結合する抗体もしくは該抗体の抗原結合性断片を含む医薬組成物を投与する個体の同定方法:
(i)(1)乃至(8)のいずれか一つに記載の抗体または該抗体の抗原結合性断片又は(9)に記載の組成物と個体由来のサンプルを接触させる工程;
(ii)該個体由来のサンプルにおいてEPHA2が検出もしくは測定されたか、または該個体由来のサンプルにおけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより高い場合、該個体を陽性と判定し、被検標本においてEPHA2が検出もしくは測定されなかったか、また該個体由来のサンプルにおいてEPHA2が検出もしくは測定されなかったか、また該個体由来のサンプルにおけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより低い場合、該個体を陰性と判定する工程、
(24)EPHA2陽性疾患の検査または診断方法に使用される、(22)または(23)に記載の方法、
(25)EPHA2陽性疾患を罹患しているもしくはそのリスクがある個体の同定方法に使用される、(23)に記載の方法、
(26)(16)の(i)乃至(iii)のいずれか一つに記載の被験者に対し、EPHA2に特異的に結合する抗体もしくは該抗体の抗原結合性断片を含む医薬組成物を投与することを含む、EPHA2陽性疾患の治療方法、
(27)(1)乃至(8)のいずれか一つに記載の抗体または該抗体の抗原結合性断片を含む、EPHA2陽性疾患を検査または診断するためのキット、
および、
(28)EPHA2陽性疾患がEPHA2陽性癌である、(24)乃至(26)に記載の方法または(27)に記載のキット、
等に関する。
The inventor diligently studied to solve the above problems, found out that a novel anti-EPHA2 antibody was created, and EPHA2 could be detected using the antibody, thereby completing the present invention. That is, the present invention includes the following inventions.
(1) The heavy chain sequence includes a variable region having CDRH1, CDRH2, and CDRH3, the CDRH1 is composed of an amino acid sequence represented by SEQ ID NO: 60, the CDRH2 is composed of an amino acid sequence represented by SEQ ID NO: 61, and the CDRH3 Consists of the amino acid sequence shown in SEQ ID NO: 62;
The light chain sequence includes a variable region having CDRL1, CDRL2, and CDRL3, wherein the CDRL1 is composed of an amino acid sequence represented by SEQ ID NO: 63, the CDRL2 is composed of an amino acid sequence represented by SEQ ID NO: 64, and the CDRL3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 65; and specifically binding to EPHA2 shown in SEQ ID NO: 2;
An antibody or an antigen-binding fragment of the antibody,
(2) A heavy chain variable region sequence consisting of
(3) A heavy chain variable region sequence consisting of
(4) The heavy chain sequence includes a variable region having CDRH1, CDRH2, CDRH 3, wherein CDRH1 consists of an amino acid sequence shown in SEQ ID NO: 66, CDRH2 consists of an amino acid sequence shown in SEQ ID NO: 67, CDRH3 consists of the amino acid sequence shown in SEQ ID NO: 68;
The light chain sequence includes a variable region having CDRL1, CDRL2, and CDRL3. The CDRL1 is composed of an amino acid sequence represented by SEQ ID NO: 69, the CDRL2 is composed of an amino acid sequence represented by SEQ ID NO: 70, and the CDRL3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 71; and specifically binding to EPHA2 shown in SEQ ID NO: 2;
An antibody or an antigen-binding fragment of the antibody,
(5) A heavy chain variable region sequence consisting of
(6) A heavy chain variable region sequence consisting of
(7) The antigen of the antibody according to any one of (1), (2), (4) or (5) selected from the group consisting of Fab, F (ab ′) 2, Fab ′ and Fv Binding fragment,
(8) The antibody according to any one of (1), (2), (4) or (5), which is scFv,
(9) A composition comprising the antibody according to any one of (1) to (8) or an antigen-binding fragment of the antibody,
(10) A tissue specimen comprising the antibody according to any one of (1) to (8) or an antigen-binding fragment of the antibody, which has been subjected to paraffin embedding and then deparaffinized (hereinafter simply referred to as “specimen”) .) The composition according to (9), which is used for a method of detecting or measuring EPHA2 in
(11) Used in a method for detecting or measuring EPHA2 in a specimen, comprising a step of contacting the test specimen with the antibody or antigen-binding fragment of the antibody according to any one of (1) to (8). The composition according to claim (9) or (10),
(12) When EPHA2 is detected or measured in the test specimen, EPHA2 is detected or measured in the test specimen, or the expression level or expression level of EPHA2 in the test specimen is equal to or higher than a predetermined standard The test specimen is determined to be positive, and EPHA2 is not detected or measured in the test specimen, or the expression level or expression level of EPHA2 in the test specimen is equal to or lower than a predetermined criterion The composition according to (11), comprising a step of determining that the test specimen is negative,
(13) The composition according to any one of (9) to (12), which is used in a test or diagnostic method for an EPHA2-positive disease,
(14) The subject from which the test specimen determined to be positive in the detection or measurement of EPHA2 by the test or diagnostic method for EPHA2-positive disease is an antibody that specifically binds to EPHA2, or an antigen-binding fragment of the antibody The subject from which the test specimen determined to be suitable for the treatment or prevention method of EPHA2-positive disease including the step of administering an antibody and specifically determined to be negative is an antibody that specifically binds to EPHA2, or antigen binding of the antibody The composition according to (13), comprising determining that it is not suitable for a method of treating or preventing an EPHA2-positive disease comprising a step of administering a sex fragment
(15) The composition according to (13) or (14), wherein the EPHA2-positive disease is EPHA2-positive cancer,
(16) A pharmaceutical composition comprising an antibody that specifically binds to EPHA2 or an antigen-binding fragment of the antibody, which is administered to the subject according to any one of (i) to (iii) below:
(I) A subject derived from a test specimen in which EPHA2 is detected or measured using the composition according to any one of (9) to (11) or (13);
(Ii) A subject derived from a test specimen determined to be positive in the detection or measurement of EPHA2 using the composition according to (12);
(Iii) Suitable for treatment or prevention of an EPHA2-positive disease comprising a step of administering an antibody that specifically binds to EPHA2 or an antigen-binding fragment thereof using the composition according to (14) or (15) Subjects determined to be,
(17) A polynucleotide encoding the antibody according to any one of (1) to (8) or an antigen-binding fragment of the antibody,
(18) A vector comprising the polynucleotide according to (17),
(19) A cell comprising the polynucleotide according to (17) or the vector according to (18),
(20) The method for producing the monoclonal antibody or antigen-binding fragment of the antibody according to any one of (1) to (8), comprising the following steps (i) and (ii):
(I) culturing the cell according to (19);
(Ii) recovering a monoclonal antibody or an antigen-binding fragment of the antibody from the culture of step (i),
(21) The antibody or antigen-binding fragment thereof according to any one of (1) to (8), wherein the antibody is a monoclonal antibody;
(22) EPHA2 detection or measurement method comprising the following steps (i) or (i) and (ii):
(I) contacting the test specimen with the antibody according to any one of (1) to (8) or the antigen-binding fragment of the antibody or the composition according to (9);
(Ii) if EPHA2 is detected or measured in the test specimen, or if the expression level or expression level of EPHA2 in the test specimen is equal to or higher than a predetermined criterion, If it is judged positive and EPHA2 is not detected or measured in the test specimen, and the expression level or expression level of EPHA2 in the test specimen is equal to or lower than a predetermined criterion, the test specimen Determining the sample as negative,
(23) A method for identifying an individual to be administered with a pharmaceutical composition comprising an antibody that specifically binds to EPHA2 or an antigen-binding fragment of the antibody, comprising the following steps (i) or (i) and (ii):
(I) contacting the antibody according to any one of (1) to (8) or an antigen-binding fragment of the antibody or the composition according to (9) with a sample derived from an individual;
(Ii) if EPHA2 is detected or measured in a sample from the individual, or if the expression level or level of EPHA2 in the sample from the individual is equal to or higher than a predetermined criterion, Whether or not EPHA2 was detected or measured in the test specimen, EPHA2 was not detected or measured in the sample derived from the individual, and the expression level or expression level of EPHA2 in the sample derived from the individual was determined in advance Determining the individual as negative if equal to or less than the criteria determined in
(24) The method according to (22) or (23), which is used for a test or diagnostic method for EPHA2-positive disease,
(25) The method according to (23), which is used for a method for identifying an individual suffering from or at risk of having an EPHA2-positive disease,
(26) A pharmaceutical composition comprising an antibody that specifically binds to EPHA2 or an antigen-binding fragment of the antibody is administered to the subject according to any one of (i) to (iii) of (16) A method of treating an EPHA2-positive disease, comprising:
(27) A kit for examining or diagnosing an EPHA2-positive disease comprising the antibody according to any one of (1) to (8) or an antigen-binding fragment of the antibody,
and,
(28) The method according to (24) to (26) or the kit according to (27), wherein the EPHA2-positive disease is EPHA2-positive cancer,
Etc.
1.定義
本発明において、「遺伝子」とは、蛋白質のアミノ酸をコードする塩基配列が含まれるヌクレオチドまたはその相補鎖を意味し、例えば、蛋白質のアミノ酸をコードする塩基配列が含まれるヌクレオチドまたはその相補鎖であるポリヌクレオチド、オリゴヌクレオチド、DNA、mRNA、cDNA、cRNA等は「遺伝子」の意味に含まれる。かかる遺伝子は一本鎖、二本鎖または三本鎖以上のヌクレオチドであり、DNA鎖とRNA鎖の会合体、一本のヌクレオチド鎖上にリボヌクレオチド(RNA)とデオキシリボヌクレオチド(DNA)が混在するものおよびそのようなヌクレオチド鎖を含む二本鎖または三本鎖以上のヌクレオチドも「遺伝子」の意味に含まれる。本発明の「EPHA2遺伝子」としては、例えば、EPHA2蛋白質のアミノ酸配列をコードする塩基配列が含まれるDNA、mRNA、cDNA、cRNA等をあげることができる。
1. Definitions In the present invention, “gene” means a nucleotide containing a nucleotide sequence encoding a protein amino acid or a complementary strand thereof, for example, a nucleotide containing a nucleotide sequence encoding a protein amino acid or a complementary strand thereof. Certain polynucleotides, oligonucleotides, DNA, mRNA, cDNA, cRNA and the like are included in the meaning of “gene”. Such a gene is a single-stranded, double-stranded, or triple-stranded nucleotide, and an assembly of a DNA strand and an RNA strand, and ribonucleotide (RNA) and deoxyribonucleotide (DNA) are mixed on a single nucleotide strand. Also included within the meaning of “gene” are double-stranded or triple-stranded nucleotides including those and such nucleotide chains. Examples of the “EPHA2 gene” of the present invention include DNA, mRNA, cDNA, cRNA and the like containing a base sequence encoding the amino acid sequence of EPHA2 protein.
本発明において、「ヌクレオチド」と「核酸」は同義であり、例えば、DNA、RNA、プローブ、オリゴヌクレオチド、ポリヌクレオチド、プライマー等も「ヌクレオチド」の意味に含まれる。かかるヌクレオチドは一本鎖、二本鎖または三本以上の鎖からなるヌクレオチドであり、DNA鎖とRNA鎖の会合体、一本のヌクレオチド鎖上にリボヌクレオチド(RNA)とデオキシリボヌクレオチド(DNA)が混在するものおよびそのようなヌクレオチド鎖を含む二本鎖または三本以上の鎖の会合体も「ヌクレオチド」の意味に含まれる。 In the present invention, “nucleotide” and “nucleic acid” are synonymous, and for example, DNA, RNA, probe, oligonucleotide, polynucleotide, primer and the like are also included in the meaning of “nucleotide”. Such a nucleotide is a nucleotide composed of a single strand, a double strand, or three or more strands, and an assembly of a DNA strand and an RNA strand, and ribonucleotide (RNA) and deoxyribonucleotide (DNA) on a single nucleotide strand. Also included within the meaning of “nucleotide” are those that are intermingled and aggregates of two or more strands containing such nucleotide strands.
本発明において、「ポリペプチド」、「ペプチド」および「蛋白質」は同義である。 In the present invention, “polypeptide”, “peptide” and “protein” are synonymous.
本発明において、「抗原」を「免疫原」の意味に用いることがある。 In the present invention, “antigen” is sometimes used to mean “immunogen”.
本発明において、「細胞」には、動物個体に由来する各種細胞、継代培養細胞、初代培養細胞、細胞株、組換え細胞および微生物等も含まれる。 In the present invention, “cell” includes various cells derived from individual animals, subculture cells, primary culture cells, cell lines, recombinant cells, microorganisms, and the like.
本発明においては、EPHA2を認識する抗体を、それぞれ「抗EPHA2抗体」と表記することがある。かかる抗体には、キメラ化抗体、ヒト化抗体、ヒト抗体等が含まれる。 In the present invention, an antibody that recognizes EPHA2 may be referred to as an “anti-EPHA2 antibody”. Such antibodies include chimerized antibodies, humanized antibodies, human antibodies and the like.
本発明における「抗体の機能断片」とは、元の抗体が奏する機能の少なくとも一部を奏する抗体断片を意味する。「抗体の機能断片」としては、例えば、Fab、F(ab’)2、scFv、Fab’、一本鎖免疫グロブリン等をあげることができるが、それらに限定されるものではない。かかる抗体の機能断片は、抗体蛋白質の全長分子をパパイン、ペプシン等の酵素で処理することによって得られたものに加え、組換え遺伝子を用いて適当な宿主細胞において産生された組換え蛋白質であってもよい。 In the present invention, the “functional fragment of an antibody” means an antibody fragment that exhibits at least a part of the function exhibited by the original antibody. Examples of the “functional fragment of an antibody” include, but are not limited to, Fab, F (ab ′) 2, scFv, Fab ′, single chain immunoglobulin and the like. Such functional fragments of antibodies are recombinant proteins produced in appropriate host cells using recombinant genes in addition to those obtained by treating full-length antibody protein molecules with enzymes such as papain and pepsin. May be.
本発明において、抗体が結合する「部位」、すなわち抗体が認識する「部位」とは、抗体が結合または認識する抗原上の部分ペプチドまたは部分高次構造を意味する。本発明においては、かかる部位のことをエピトープ、抗体の結合部位とも呼ぶ。本発明の抗EPHA2抗体が結合または認識するEPHA2蛋白質上の部位としては、EPHA2蛋白質上の部分ペプチドまたは部分高次構造等を例示することができる。 In the present invention, the “site” to which the antibody binds, that is, the “site” recognized by the antibody means a partial peptide or a partial higher order structure on the antigen to which the antibody binds or recognizes. In the present invention, such a site is also referred to as an epitope or an antibody binding site. As a site | part on EPHA2 protein which the anti- EPHA2 antibody of this invention couple | bonds or recognizes, the partial peptide or partial higher order structure etc. on EPHA2 protein can be illustrated.
抗体分子の重鎖および軽鎖にはそれぞれ3箇所の相補性決定領域(CDR:Complementarity determining region)があることが知られている。相補性決定領域は、超可変領域(hypervariable domain)とも呼ばれ、抗体の重鎖および軽鎖の可変領域内にあって、一次構造の変異性が特に高い部位であり、重鎖および軽鎖のポリペプチド鎖の一次構造上において、通常、それぞれ3ヶ所に分離している。本発明においては、抗体の相補性決定領域について、重鎖の相補性決定領域を重鎖アミノ酸配列のアミノ末端側からCDRH1、CDRH2、CDRH3と表記し、軽鎖の相補性決定領域を軽鎖アミノ酸配列のアミノ末端側からCDRL1、CDRL2、CDRL3と表記する。これらの部位は立体構造の上で相互に近接し、結合する抗原に対する特異性を決定している。 It is known that there are three complementarity determining regions (CDRs) in each of the heavy and light chains of an antibody molecule. The complementarity-determining region is also called a hypervariable domain, and is located in the variable region of the heavy and light chains of an antibody and has a particularly high primary structure variability. In the primary structure of the polypeptide chain, it is usually separated at three points. In the present invention, for the complementarity determining region of an antibody, the complementarity determining region of the heavy chain is denoted as CDRH1, CDRH2, CDRH3 from the amino terminal side of the heavy chain amino acid sequence, and the complementarity determining region of the light chain is defined as the light chain amino acid. CDRL1, CDRL2, and CDRL3 are represented from the amino terminal side of the sequence. These sites are close to each other on the three-dimensional structure and determine the specificity for the antigen to be bound.
本発明において、「抗体変異体」とは、元の抗体が有するアミノ酸配列においてアミノ酸が置換、欠失、付加および/または挿入(以下、「変異」と総称する)してなるアミノ酸配列を有し、且つ本発明のEPHA2蛋白質に結合するポリペプチドを意味する。かかる抗体変異体における変異アミノ酸の数は、1乃至2個、1乃至3個、1乃至4個、1乃至5個、1乃至6個、1乃至7個、1乃至8個、1乃至9個、1乃至10個、1乃至12個、1乃至15個、1乃至20個、1乃至25個、1乃至30個、1乃至40個または1乃至50個である。かかる抗体変異体も本発明の「抗体」に包含される。 In the present invention, “antibody variant” has an amino acid sequence in which amino acids are substituted, deleted, added and / or inserted (hereinafter collectively referred to as “mutation”) in the amino acid sequence of the original antibody. And a polypeptide that binds to the EPHA2 protein of the present invention. The number of variant amino acids in such antibody variants is 1 to 2, 1 to 3, 1 to 4, 1 to 5, 1 to 6, 1 to 7, 1 to 8, 1 to 9 1 to 10, 1 to 12, 1 to 15, 1 to 20, 1 to 25, 1 to 30, 1 to 40, or 1 to 50. Such antibody variants are also included in the “antibody” of the present invention. *
本発明において、「1乃至数個」における「数個」とは、3乃至10個を指す。 In the present invention, “several” in “1 to several” refers to 3 to 10.
本発明の抗体が奏する活性・性質としては、例えば、生物的活性、理化学的性質等を挙げることができ、具体的には、各種生物活性、抗原やエピトープに対する結合活性、製造や保存時における安定性、熱安定性等をあげることができる。 Examples of the activity / property exhibited by the antibody of the present invention include biological activity, physicochemical properties, and the like. Specifically, various biological activities, binding activity to antigens and epitopes, stability during production and storage Property, heat stability, etc. can be raised.
本発明において、「ストリンジェントな条件下でハイブリダイズする」とは、5×SSCを含む溶液中で65℃にてハイブリダイゼーションを行い、ついで2×SSC-0.1%SDSを含む水溶液中で65℃にて20分間、0.5×SSC-0.1%SDSを含む水溶液中で65℃にて20分間、ならびに、0.2×SSC-0.1%SDSを含む水溶液中で65℃にて20分間、それぞれ洗浄する条件またはそれと同等の条件でハイブリダイズすることを意味する。SSCとは150mMNaCl-15mMクエン酸ナトリウムの水溶液であり、n×SSCはn倍濃度のSSCを意味する。 In the present invention, “hybridize under stringent conditions” means hybridization at 65 ° C. in a solution containing 5 × SSC, and then in an aqueous solution containing 2 × SSC-0.1% SDS. 20 minutes at 65 ° C. in an aqueous solution containing 0.5 × SSC-0.1% SDS for 20 minutes at 65 ° C. and 65 ° C. in an aqueous solution containing 0.2 × SSC-0.1% SDS Means to hybridize under conditions of washing for 20 minutes or under equivalent conditions. SSC is an aqueous solution of 150 mM NaCl-15 mM sodium citrate, and nx SSC means n-fold concentration of SSC.
本発明において「細胞傷害」とは、何らかの形で、細胞に病理的な変化をもたらすことを指し、直接的な外傷にとどまらず、DNAの切断や塩基の二量体の形成、染色体の切断、細胞分裂装置の損傷、各種酵素活性の低下などあらゆる細胞の構造や機能上の損傷を意味する。 In the present invention, “cytotoxicity” refers to causing a pathological change in a cell in some form, and is not limited to direct trauma, but also includes DNA breakage, base dimer formation, chromosome breakage, It means any structural or functional damage of cells, such as damage to cell division apparatus or reduction of various enzyme activities.
本発明において「細胞傷害活性」とは上記細胞傷害を引き起こすことを意味する。本発明において「抗体依存性細胞傷害活性」とは、「antibody dependent cellular cytotoxicity(ADCC)活性」を指し、NK細胞が抗体を介して腫瘍細胞等の標的細胞を傷害する作用活性を意味する。 In the present invention, “cytotoxic activity” means causing the above cytotoxicity. In the present invention, “antibody-dependent cytotoxic activity” refers to “antibody dependent cellular cytotoxicity (ADCC) activity”, and means an activity in which NK cells damage target cells such as tumor cells via antibodies.
本発明において「抗体依存性細胞媒介食活性」とは、「antibody dependent cell phagocytosis(ADCP)活性」を指し、単球やマクロファージ細胞が抗体を介して腫瘍細胞等の標的細胞を貪食する作用活性を意味する。「抗体依存的貪食作用活性」ともいう。 In the present invention, “antibody-dependent cell-mediated phagocytic activity” refers to “antibody dependent cell phagocytosis (ADCP) activity”, which is an activity that monocytes and macrophage cells phagocytose target cells such as tumor cells via antibodies. means. Also referred to as “antibody-dependent phagocytic activity”.
本発明において「補体依存性細胞傷害作用活性」とは、「complement dependent cytotoxicity(CDC)活性」を指し、補体が抗体を介して腫瘍細胞等の標的細胞を傷害する作用活性を意味する。 In the present invention, “complement-dependent cytotoxic activity” refers to “complement dependent cytotoxicity (CDC) activity” and means an activity that complements a target cell such as a tumor cell via an antibody.
本発明において「癌」と「腫瘍」は同じ意味で用いられる。 In the present invention, “cancer” and “tumor” are used interchangeably.
本発明において「免疫組織化学(immunohistochemistry:IHC)」とは組織標本中の抗原を検出する組織学的(組織化学的)手法を意味し、「免疫抗体法」と同義であり、「免疫染色(immunostaining)」も同じ意味に用いられる。 In the present invention, “immunohistochemistry (IHC)” means a histological (histochemical) method for detecting an antigen in a tissue specimen, and is synonymous with “immunoantibody method”. “immunostaining”) is also used interchangeably.
本発明において「変性型」EPHA2とは、ホルマリンで固定した標本中のEPHA2分子を意味する。ホルマリンで固定した後にパラフィン処理および脱パラフィン処理した標本中のEPHA2分子も「変性型」EPHA2という。 In the present invention, “denatured” EPHA2 means an EPHA2 molecule in a specimen fixed with formalin. EPHA2 molecules in specimens fixed with formalin and then paraffin-treated and deparaffinized are also referred to as “denatured” EPHA2.
本発明において「非変性型」EPHA2とは、ホルマリンで固定していないサンプル中のEPHA2を意味する。ホルマリンで固定していない標本中のEPHA2分子も「非変性型」EPHA2という。 In the present invention, “non-denatured” EPHA2 means EPHA2 in a sample not fixed with formalin. EPHA2 molecules in specimens not fixed with formalin are also referred to as “non-denaturing” EPHA2.
2.EPHA2
本発明で用いるEPHA2は、ヒト、ヒト以外の哺乳動物(例えば、モルモット、ラット、マウス、ウサギ、ブタ、ヒツジ、ウシ、サルなど)あるいはニワトリのT細胞あるいは肥満細胞から直接精製して使用するか、あるいは上記の細胞の細胞膜画分を調製して使用することができ、また、EPHA2をin vitroにて合成する、あるいは遺伝子操作により宿主細胞に産生させることによって得ることができる。遺伝子操作では、具体的には、EPHA2 cDNAを発現可能なベクターに組み込んだ後、転写と翻訳に必要な酵素、基質およびエネルギー物質を含む溶液中で合成する、あるいは他の原核生物、または真核生物の宿主細胞を形質転換させることによってEPHA2を発現させることにより、該蛋白質を得ることが出来る。
2. EPHA2
Is EPHA2 used in the present invention directly purified from human, non-human mammals (eg, guinea pig, rat, mouse, rabbit, pig, sheep, cow, monkey, etc.) or chicken T cells or mast cells? Alternatively, the cell membrane fraction of the above-mentioned cells can be prepared and used, and it can be obtained by synthesizing EPHA2 in vitro or producing it in a host cell by genetic manipulation. Specifically, in genetic manipulation, EPHA2 cDNA is incorporated into a vector capable of expression and then synthesized in a solution containing enzymes, substrates and energy substances necessary for transcription and translation, or other prokaryote or eukaryote. The protein can be obtained by expressing EPHA2 by transforming a host cell of an organism.
ヒトEPHA2のcDNAのヌクレオチド配列は、GenBankにアクセッション番号:NM_004431で登録されている。また、ヒトEPHA2のアミノ酸配列は、GenBankにアクセッション番号:NP_004422で登録されている。 The nucleotide sequence of human EPHA2 cDNA is registered in GenBank with accession number: NM_004431. Further, the amino acid sequence of human EPHA2 is registered in GenBank with an accession number: NP_004422.
EPHA2のcDNAは例えば、EPHA2のmRNAを発現している臓器のcDNAライブラリーを鋳型として、EPHA2のcDNAを特異的に増幅するプライマーを用いてポリメラーゼ連鎖反応(以下「PCR」という)(Saiki,R. K.,et al.,Science,(1988)239,487-49)を行なう、いわゆるPCR法により取得することができる。 EPHA2 cDNA is, for example, a polymerase chain reaction (hereinafter referred to as “PCR”) using a cDNA library of an organ expressing EPHA2 mRNA as a template and a primer that specifically amplifies EPHA2 cDNA (Saiki, R K., et al., Science, (1988) 239, 487-49).
なお、ヒトEPHA2をコードするヌクレオチド配列と相補的なヌクレオチド配列からなるポリヌクレオチドとストリンジェントな条件でハイブリダイズし、かつ、EPHA2と同等の生物活性を有する蛋白質をコードするポリヌクレオチドもEPHA2のcDNAに含まれる。さらに、ヒトEPHA2遺伝子座から転写されるスプライシングバリアントまたはこれにストリンジェントな条件でハイブリダイズするポリヌクレオチドであって、かつ、EPHA2と同等の生物活性を有する蛋白質をコードするポリヌクレオチドもEPHA2のcDNAに含まれる。 In addition, a polynucleotide that hybridizes with a polynucleotide comprising a nucleotide sequence complementary to the nucleotide sequence encoding human EPHA2 under stringent conditions and encodes a protein having biological activity equivalent to that of EPHA2, is also included in the EPHA2 cDNA. included. Furthermore, a polynucleotide encoding a splicing variant transcribed from the human EPHA2 locus or a protein that hybridizes to this under stringent conditions and also has a biological activity equivalent to that of EPHA2 is also included in the EPHA2 cDNA. included.
また、ヒトEPHA2のアミノ酸配列、またはこれらの配列からシグナル配列が除かれたアミノ酸配列において、1個、2もしくは3個または4もしくは5個のアミノ酸が置換、欠失、または付加されたアミノ酸配列からなり、EPHA2と同等の生物活性を有する蛋白質もEPHA2に含まれる。さらに、ヒトEPHA2遺伝子座から転写されるスプライシングバリアントにコードされるアミノ酸配列または該アミノ酸配列において、1個、2もしくは3個または4もしくは5個のアミノ酸が置換、欠失、または付加されたアミノ酸配列からなり、かつ、EPHA2と同等の生物活性を有する蛋白質もEPHA2に含まれる。 In addition, from the amino acid sequence of human EPHA2, or the amino acid sequence obtained by substituting, deleting, or adding 1, 2, or 3, or 4 or 5 amino acids in the amino acid sequence obtained by removing the signal sequence from these sequences Thus, a protein having a biological activity equivalent to that of EPHA2 is also included in EPHA2. Furthermore, an amino acid sequence encoded by a splicing variant transcribed from the human EPHA2 locus, or an amino acid sequence in which 1, 2 or 3 or 4 or 5 amino acids are substituted, deleted or added in the amino acid sequence EPHA2 is also included in the protein having a biological activity equivalent to that of EPHA2.
本明細書で使用された、シグナルペプチドを含まない成熟型ヒトEPHA2をコードするヌクレオチド配列は配列表の配列番号1に記載されており、該ヒトEPHA2のアミノ酸配列は配列表の配列番号2に記載されている。
2.抗EPHA2抗体の製造
本発明のEPHA2に対する抗体は、常法を用いて、EPHA2またはEPHA2のアミノ酸配列から選択される任意のポリペプチドを動物に免疫し、生体内に産生される抗体を採取、精製することによって得ることができる。抗原となるEPHA2の生物種はヒトに限定されず、マウス、ラット等のヒト以外の動物に由来するEPHA2を動物に免疫することもできる。この場合には、取得された異種EPHA2に結合する抗体とヒトEPHA2との交差性を試験することによって、ヒトの疾患に適用可能な抗体を選別できる。なお、抗原となるEPHA2はEPHA2遺伝子を遺伝子操作により宿主細胞に産生させることによって得ることができる。具体的には、EPHA2遺伝子を発現可能なベクターを作製し、これを宿主細胞に導入して該遺伝子を発現させ、発現したEPHA2を精製すればよい。
As used herein, the nucleotide sequence encoding mature human EPHA2 that does not contain a signal peptide is set forth in SEQ ID NO: 1 in the Sequence Listing, and the amino acid sequence of the human EPHA2 is set forth in SEQ ID NO: 2 in the Sequence Listing. Has been.
2. Production of anti-EPHA2 antibody The antibody against EPHA2 of the present invention is obtained by immunizing an animal with any polypeptide selected from the amino acid sequence of EPHA2 or EPHA2 using a conventional method, and collecting and purifying the antibody produced in vivo Can be obtained. The species of EPHA2 serving as an antigen is not limited to humans, and animals can be immunized with EPHA2 derived from animals other than humans such as mice and rats. In this case, an antibody applicable to a human disease can be selected by testing the cross-reactivity between the obtained antibody that binds to heterologous EPHA2 and human EPHA2. In addition, EPHA2 used as an antigen can be obtained by causing a host cell to produce the EPHA2 gene by genetic manipulation. Specifically, a vector capable of expressing the EPHA2 gene is prepared, introduced into a host cell to express the gene, and the expressed EPHA2 may be purified.
本発明のEPHA2に対する抗体は、DNA免疫法を使用して得ることもできる。DNA免疫法とは、抗原発現プラスミドをマウスやラットなどの動物個体に遺伝子導入し、抗原を個体内で発現させることによって、抗原に対する免疫を誘導する手法である。遺伝子導入の手法には、直接プラスミドを筋肉に注射する方法や、リポソームやポリエチレンイミンなどの導入試薬を静脈注射する方法、ウイルスベクターを用いる手法、プラスミドを付着させた金粒子をGene Gunにより射ち込む手法、急速に大量のプラスミド溶液を静脈注射するHydrodynamic法などが存在する。発現プラスミドの筋注による遺伝子導入法に関して、発現量を向上させる手法として、プラスミドを筋注後、同部位にエレクトロポレーションを加えるin vivo electroporationという技術が知られている(Aihara H, Miyazaki J. Nat Biotechnol. 1998 Sep;16(9):867-70またはMir LM, Bureau MF, Gehl J, Rangara R, Rouy D, Caillaud JM, Delaere P, Branellec D, Schwartz B, Scherman D. Proc Natl Acad Sci U S A. 1999 Apr 13;96(8):4262-7.)。本手法はプラスミドの筋注前にヒアルロニダーゼで筋肉を処理することにより、さらに発現量が向上する(McMahon JM1, Signori E, Wells KE, Fazio VM, Wells DJ. Gene Ther. 2001 Aug;8(16):1264-70)。 The antibody against EPHA2 of the present invention can also be obtained using a DNA immunization method. The DNA immunization method is a technique for inducing immunity to an antigen by introducing an antigen expression plasmid into an individual animal such as a mouse or a rat and expressing the antigen in the individual. Gene transfer methods include direct injection of plasmids into muscle, intravenous injection of liposomes and polyethyleneimine and other introduction reagents, viral vector methods, and gold particles with plasmids attached to them by Gene Gun. There are techniques, such as the Hydrodynamic method in which a large amount of plasmid solution is intravenously injected. As a technique for improving gene expression by intramuscular injection of an expression plasmid, a technique called in vivo electroporation is known (Aihara H, Miyazaki J. et al.). Nat Biotechnol.1998 Sep; 16 (9): 867-70 or Mir LM, Bureau MF, Gehl J, Rangara R, Roue D, Cailaud JM, DelaureD, BranellS.Branellec. S A. 1999 Apr 13; 96 (8): 4262-7.). In this method, the amount of expression is further improved by treating the muscle with hyaluronidase before intramuscular injection of the plasmid (McMahon JM1, Signori E, Wells KE, Fazio VM, Wells DJ. Gene Ther. 2001 Aug; 8 (16) : 1264-70).
また、公知の方法(例えば、Kohler and Milstein,Nature (1975)256,p.495-497、Kennet,R.ed.,Monoclonal Antibodies,p.365-367,Plenum Press,N.Y.(1980))に従って、EPHA2に対する抗体を産生する抗体産生細胞とミエローマ細胞とを融合させることによりハイブリドーマを樹立し、モノクローナル抗体を得ることもできる。このような方法の具体的な例は、国際公開第WO09/48072号パンフレット(2009年4月16日公開)および第WO10/117011号(2010年10月14日公開)パンフレットに記載されている。 Also, known methods (for example, Kohler and Milstein, Nature (1975) 256, p. 495-497, Kennet, R. ed., Monoclonal Antibodies, p. 365-367, Plenum Press, NY (1980). ), Hybridomas can be established by fusing antibody-producing cells that produce antibodies against EPHA2 and myeloma cells to obtain monoclonal antibodies. Specific examples of such a method are described in International Publication No. WO 09/48072 (published on April 16, 2009) and WO 10/11711 (published on October 14, 2010).
このようにして樹立されたマウス抗ヒトEPHA2抗体の実例としては、A201抗体およびA205抗体を挙げることができる。A201抗体重鎖のアミノ酸配列は配列表の配列番号51に、これをコードするヌクレオチド配列は配列表の配列番号50に、それぞれ示されている。A201抗体軽鎖のアミノ酸配列は配列表の配列番号54に、これをコードするヌクレオチド配列は配列表の配列番号53に、それぞれ示されている。また、A205抗体重鎖のアミノ酸配列は配列表の配列番号57に、これをコードするヌクレオチド配列は配列表の配列番号56に、それぞれ示されている。A205抗体軽鎖のアミノ酸配列は配列表の配列番号59に、これをコードするヌクレオチド配列は配列表の配列番号58に、それぞれ示されている。 Examples of the mouse anti-human EPHA2 antibody thus established include A201 antibody and A205 antibody. The amino acid sequence of the A201 antibody heavy chain is shown in SEQ ID NO: 51 of the Sequence Listing, and the nucleotide sequence encoding it is shown in SEQ ID NO: 50 of the Sequence Listing. The amino acid sequence of the A201 antibody light chain is shown in SEQ ID NO: 54 of the sequence listing, and the nucleotide sequence encoding it is shown in SEQ ID NO: 53 of the sequence listing. The amino acid sequence of the A205 antibody heavy chain is shown in SEQ ID NO: 57 in the sequence listing, and the nucleotide sequence encoding this is shown in SEQ ID NO: 56 in the sequence listing. The amino acid sequence of the A205 antibody light chain is shown in SEQ ID NO: 59 in the sequence listing, and the nucleotide sequence encoding it is shown in SEQ ID NO: 58 in the sequence listing.
本発明の抗体は、A201に由来する6種全てのCDR配列を保持し、EPHA2に結合する活性を有している抗体であってよい。すなわち、本発明の抗体の重鎖可変領域は、配列番号60に示されるアミノ酸配列からなるCDRH1(DTYVH)、配列番号61に示されるアミノ酸配列からなるCDRH2(RIDPANANTKYDPKFQG)、および配列番号62に示されるアミノ酸配列からなるCDRH3(YGKSAWFAY)を保有している。また、前記の抗体の軽鎖可変領域は、配列番号63に示されるアミノ酸配列からなるCDRL1(RASQDIGNYLN)、配列番号64に示されるアミノ酸配列からなるCDRL2(YTSRLHS)、および配列番号65に示されるアミノ酸配列からなるCDRL3(QQGHSLPPT)を保有している。これらのCDRのアミノ酸配列は、図11にも記載されている。 The antibody of the present invention may be an antibody that retains all six CDR sequences derived from A201 and has an activity of binding to EPHA2. That is, the heavy chain variable region of the antibody of the present invention is represented by CDRH1 (DTYVH) consisting of the amino acid sequence shown in SEQ ID NO: 60, CDRH2 (RIDPANANTKYDPKFQG) consisting of the amino acid sequence shown in SEQ ID NO: 61, and SEQ ID NO: 62. It possesses CDRH3 (YGKSAWFY) consisting of an amino acid sequence. The light chain variable region of the antibody comprises CDRL1 (RASQDIGNYLN) consisting of the amino acid sequence shown in SEQ ID NO: 63, CDRL2 (YTSRLHS) consisting of the amino acid sequence shown in SEQ ID NO: 64, and amino acids shown in SEQ ID NO: 65. It has CDRL3 (QQGHSLPPT) consisting of the sequence. The amino acid sequences of these CDRs are also shown in FIG.
本発明の抗体は、A205に由来する6種全てのCDR配列を保持し、EPHA2に結合する活性を有している抗体であってよい。すなわち、本発明の抗体の重鎖可変領域は、配列番号66に示されるアミノ酸配列からなるCDRH1(DYSMN)、配列番号67に示されるアミノ酸配列からなるCDRH2(WINTYTGEPTYADDFKG)、および配列番号68に示されるアミノ酸配列からなるCDRH3(PISLLRLLDY)を保有している。また、前記の抗体の軽鎖可変領域は、配列番号69に示されるアミノ酸配列からなるCDRL1(RSSQSLVHSNGNTYLH)、配列番号70に示されるアミノ酸配列からなるCDRL2(KVSNRFS)、および配列番号71に示されるアミノ酸配列からなるCDRL3(SQSTHVPWT)を保有している。これらのCDRのアミノ酸配列は、図12にも記載されている。
本発明の抗体はEPHA2蛋白質を特異的に認識する。言い換えれば、本発明の好適な抗体はEPHA2蛋白質に特異的に結合する。
ある態様において、好適な抗体は、非変性型のヒトEPHA2、及び、ホルマリンで固定した標本中の変性型ヒトEPHA2の両方に特異的に結合する。また、より好適な抗体は、非変性型のヒトEPHA2、及び、ホルマリンで固定した標本中の変性型ヒトEPHA2の両方に特異的に結合し、かつ他のEphファミリーに特異的に結合しない抗体等を挙げることができるが、それらに限定されるものではない。
The antibody of the present invention may be an antibody that retains all six CDR sequences derived from A205 and has an activity of binding to EPHA2. That is, the heavy chain variable region of the antibody of the present invention is shown in CDRH1 (DYSMN) consisting of the amino acid sequence shown in SEQ ID NO: 66, CDRH2 (WINTYTGEPTYADDFKG) consisting of the amino acid sequence shown in SEQ ID NO: 67, and SEQ ID NO: 68. It possesses CDRH3 (PISLLLRLDY) consisting of an amino acid sequence. In addition, the light chain variable region of the above-mentioned antibody has CDRL1 (RSSQSLVHSNGNTYLH) consisting of the amino acid sequence shown in SEQ ID NO: 69, CDRL2 (KVSNRFS) consisting of the amino acid sequence shown in SEQ ID NO: 70, and amino acids shown in SEQ ID NO: 71. It possesses CDRL3 (SQSTHVPWT) consisting of the sequence. The amino acid sequences of these CDRs are also shown in FIG.
The antibody of the present invention specifically recognizes EPHA2 protein. In other words, preferred antibodies of the present invention specifically bind to the EPHA2 protein.
In certain embodiments, suitable antibodies specifically bind to both undenatured human EPHA2 and denatured human EPHA2 in formalin-fixed specimens. More preferable antibodies include antibodies that specifically bind to both non-denatured human EPHA2 and denatured human EPHA2 in formalin-fixed specimens and that do not specifically bind to other Eph families. However, it is not limited to them.
本発明の抗体には、上記のEPHA2に対するモノクローナル抗体に加え、ヒトに対する異種抗原性を低下させること等を目的として人為的に改変した遺伝子組換え型抗体、例えば、キメラ(Chimeric)抗体、またはヒト化(Humanized)抗体も含まれる。これらの抗体は、既知の方法を用いて製造することができる。 In addition to the above monoclonal antibody against EPHA2, the antibodies of the present invention include genetically modified antibodies that have been artificially modified for the purpose of reducing heteroantigenicity against humans, such as chimeric antibodies, or humans. Also included are humanized antibodies. These antibodies can be produced using known methods.
キメラ抗体としては、抗体の可変領域と定常領域が互いに異種である抗体、例えばマウスまたはラット由来抗体の可変領域をヒト由来の定常領域に接合したキメラ抗体を挙げることができる(Proc.Natl.Acad.Sci.U.S.A.,81,6851-6855,(1984)参照)。 Examples of the chimeric antibody include antibodies in which the variable region and the constant region of the antibody are different from each other, for example, a chimeric antibody in which the variable region of a mouse or rat-derived antibody is joined to a human-derived constant region (Proc. Natl. Acad). Sci.U.S.A., 81, 6851-6855, (1984)).
上記のEPHA2に対するキメラ抗体の配列を、ヒトに対する異種抗原性を低下させること等を目的として人為的に改変して、遺伝子組換え型抗体であるヒト化(Humanized)抗体を作製することができる。本発明の抗体には、前記ヒト化抗体のCDRを改変した抗体が含まれる。これらの抗体は、既知の方法を用いて製造することができる。 The above-described chimeric antibody sequence against EPHA2 can be artificially modified for the purpose of, for example, reducing the heterologous antigenicity to humans, thereby producing a humanized antibody that is a recombinant antibody. The antibody of the present invention includes an antibody obtained by modifying the CDR of the humanized antibody. These antibodies can be produced using known methods.
ヒト化抗体としては、相補性決定領域(CDR;complementarity determining region)のみをヒト由来の抗体に組み込んだ抗体(Nature(1986)321,p.522-525参照)、CDRの配列に加え一部のフレームワークのアミノ酸残基もヒト抗体に移植した抗体(国際公開第WO90/07861号パンフレット)を挙げることができる。 As humanized antibodies, antibodies (see Nature (1986) 321, p.522-525) in which only complementarity determining regions (CDRs) are incorporated into human-derived antibodies (see Nature (1986) 321, p.522-525), some sequences in addition to CDR sequences Examples of the amino acid residues of the framework include an antibody grafted on a human antibody (International Publication No. WO90 / 07861 pamphlet).
なお、哺乳類培養細胞で生産される抗体の重鎖のカルボキシル末端のリシン残基が欠失することが知られており(Journal of Chromatography A,705:129-134(1995))、また、同じく重鎖カルボキシル末端のグリシン、リシンの2アミノ酸残基が欠失し、新たにカルボキシル末端に位置するプロリン残基がアミド化されることが知られている(Analytical Biochemistry,360:75-83(2007))。しかし、これらの重鎖配列の欠失および修飾は、抗体の抗原結合能およびエフェクター機能(補体の活性化や抗体依存性細胞傷害作用など)には影響を及ぼさない。従って、本発明には当該修飾を受けた抗体も含まれ、重鎖カルボキシル末端において1または2つのアミノ酸が欠失した欠失体、およびアミド化された当該欠失体(例えば、カルボキシル末端部位のプロリン残基がアミド化された重鎖)等を挙げることができる。但し、抗原結合能およびエフェクター機能が保たれている限り、本発明に係る抗体の重鎖のカルボキシル末端の欠失体は上記の種類に限定されない。本発明に係る抗体を構成する2本の重鎖は、完全長および上記の欠失体からなる群から選択される重鎖のいずれか一種であっても良いし、いずれか二種を組み合わせたものであっても良い。各欠失体の量比は本発明に係る抗体を産生する哺乳類培養細胞の種類および培養条件に影響を受け得るが、本発明に係る抗体の主成分としては2本の重鎖の双方でカルボキシル末端の1つのアミノ酸残基が欠失している場合を挙げることができる。 It is known that the lysine residue at the carboxyl terminus of the heavy chain of an antibody produced by cultured mammalian cells is deleted (Journal of Chromatography A, 705: 129-134 (1995)). It is known that two amino acid residues of glycine and lysine at the chain carboxyl terminal are deleted, and a proline residue located at the carboxyl terminal is newly amidated (Analytical Biochemistry, 360: 75-83 (2007)). ). However, deletion and modification of these heavy chain sequences do not affect the antigen-binding ability and effector functions (such as complement activation and antibody-dependent cytotoxicity) of the antibody. Accordingly, the present invention also includes antibodies having such modifications, including deletions in which one or two amino acids have been deleted at the heavy chain carboxyl terminus, and such a deletions that have been amidated (eg, at the carboxyl terminus site). Heavy chain in which a proline residue is amidated). However, as long as the antigen binding ability and the effector function are maintained, the carboxyl-terminal deletion of the heavy chain of the antibody according to the present invention is not limited to the above type. The two heavy chains constituting the antibody according to the present invention may be either one of the full length and the heavy chain selected from the group consisting of the above-mentioned deletion forms, or a combination of any two of them. It may be a thing. The amount ratio of each deletion can be influenced by the type and culture conditions of the cultured mammalian cells that produce the antibody according to the present invention, but the main component of the antibody according to the present invention is a carboxyl in both two heavy chains. A case where one terminal amino acid residue is deleted can be mentioned.
以上の方法によって得られた抗体は、抗原に対する結合性を評価し、好適な抗体を選抜することができる。抗体の性質を比較する際の別の指標の一例としては、抗体の安定性を挙げることができる。示差走査カロリメトリー(DSC)は、蛋白の相対的構造安定性の良い指標となる熱変性中点(Tm)を素早く、また正確に測定することができる方法である。DSCを用いてTm値を測定し、その値を比較することによって、熱安定性の違いを比較することができる。抗体の保存安定性は、抗体の熱安定性とある程度の相関を示すことが知られており(Lori Burton,et. al.,Pharmaceutical Development and Technology(2007)12,p.265-273)、熱安定性を指標に、好適な抗体を選抜することができる。抗体を選抜するための他の指標としては、適切な宿主細胞における収量が高いこと、および水溶液中での凝集性が低いことを挙げることができる。例えば収量の最も高い抗体が最も高い熱安定性を示すとは限らないので、以上に述べた指標に基づいて総合的に判断して、最も適した抗体を選抜する必要がある。 The antibody obtained by the above method can be evaluated for binding to an antigen and a suitable antibody can be selected. An example of another index for comparing antibody properties is antibody stability. Differential scanning calorimetry (DSC) is a method that can quickly and accurately measure the thermal denaturation midpoint (Tm), which is an indicator of good relative structural stability of proteins. The difference in thermal stability can be compared by measuring the Tm value using DSC and comparing the values. It is known that the storage stability of an antibody shows a certain degree of correlation with the thermal stability of the antibody (Lori Burton, et. Al., Pharmaceutical Development and Technology (2007) 12, p.265-273), and heat. A suitable antibody can be selected using stability as an index. Other indicators for selecting antibodies include high yields in appropriate host cells and low aggregation in aqueous solutions. For example, since the antibody with the highest yield does not always exhibit the highest thermal stability, it is necessary to select the most suitable antibody based on a comprehensive judgment based on the above-described indicators.
また、抗体の重鎖および軽鎖の全長配列を適切なリンカーを用いて連結し、一本鎖イムノグロブリン(single chain immunoglobulin)を取得する方法も知られている(Lee,H-S,et.al.,Molecular Immunology(1999)36,p.61-71;Shirrmann,T.et.al.,mAbs(2010),2,(1)p.1-4)。このような一本鎖イムノグロブリンは二量体化することによって、本来は四量体である抗体と類似した構造と活性を保持することが可能である。また、本発明の抗体は、単一の重鎖可変領域を有し、軽鎖配列を有さない抗体であっても良い。このような抗体は、単一ドメイン抗体(single domain antibody:sdAb)またはナノボディ(nanobody)と呼ばれており、実際にラクダまたはラマで観察され、抗原結合能が保持されていることが報告されている(Muyldemans S.et.al.,Protein Eng.(1994)7(9),1129-35,Hamers-Casterman C.et.al.,Nature(1993)363(6428)446-8)。上記の抗体は、本発明における抗体の抗原結合性断片の一種と解釈することも可能である。 Also known is a method of obtaining single chain immunoglobulins by linking the full length sequences of antibody heavy and light chains using an appropriate linker (Lee, HS, et. al., Molecular Immunology (1999) 36, p. 61-71; Shirrmann, T. et. al., mAbs (2010), 2, (1) p. By dimerizing such a single-chain immunoglobulin, it is possible to retain a structure and activity similar to those of an antibody that is originally a tetramer. The antibody of the present invention may be an antibody having a single heavy chain variable region and no light chain sequence. Such antibodies are called single domain antibodies (sdAbs) or nanobodies, and are actually observed in camels or llamas and reported to retain antigen-binding ability. (Muyldemans S. et.al., Protein Eng. (1994) 7 (9), 1129-35, Hamers-Casterman C. et.al., Nature (1993) 363 (6428) 446-8). The above-described antibody can also be interpreted as a kind of antigen-binding fragment of the antibody in the present invention.
抗体遺伝子を一旦単離した後、適当な宿主に導入して抗体を作製する場合には、適当な宿主と発現ベクターの組み合わせを使用することができる。抗体遺伝子の具体例としては、本明細書に記載された抗体の重鎖配列をコードする遺伝子、および軽鎖配列をコードする遺伝子を組み合わせたものを挙げることができる。宿主細胞を形質転換する際には、重鎖配列遺伝子と軽鎖配列遺伝子は、同一の発現ベクターに挿入されていることが可能であり、又別々の発現ベクターに挿入されていることも可能である。真核細胞を宿主として使用する場合、動物細胞、植物細胞、真核微生物を用いることができる。動物細胞としては、(1)哺乳類細胞、例えば、サルの細胞であるCOS細胞(Gluzman,Y.Cell(1981)23,p.175-182、ATCC CRL-1650)、マウス線維芽細胞NIH3T3(ATCC No.CRL-1658)やチャイニーズ・ハムスター卵巣細胞(CHO細胞、ATCC CCL-61)のジヒドロ葉酸還元酵素欠損株(Urlaub,G.and Chasin,L.A.Proc.Natl.Acad.Sci.U.S.A.(1980)77,p.4126-4220)を挙げることができる。また、原核細胞を使用する場合は、例えば、大腸菌、枯草菌を挙げることができる。これらの細胞に目的とする抗体遺伝子を形質転換により導入し、形質転換された細胞をin vitroで培養することにより抗体が得られる。以上の培養法においては抗体の配列によって収量が異なる場合があり、同等な結合活性を持つ抗体の中から収量を指標に医薬としての生産が容易なものを選別することが可能である。 When an antibody gene is once isolated and then introduced into an appropriate host to produce an antibody, a combination of an appropriate host and an expression vector can be used. Specific examples of the antibody gene include a combination of a gene encoding the heavy chain sequence of the antibody described herein and a gene encoding the light chain sequence. When transforming a host cell, the heavy chain sequence gene and the light chain sequence gene can be inserted into the same expression vector, or can be inserted into separate expression vectors. is there. When eukaryotic cells are used as hosts, animal cells, plant cells, and eukaryotic microorganisms can be used. Examples of animal cells include (1) mammalian cells such as COS cells (Gluzman, Y. Cell (1981) 23, p. 175-182, ATCC CRL-1650) which are monkey cells, mouse fibroblasts NIH3T3 (ATCC). No. CRL-1658) and Chinese hamster ovary cells (CHO cells, ATCC CCL-61) dihydrofolate reductase-deficient strains (Urlauub, G. and Chasin, LA Proc. Natl. Acad. Sci. U.). S. A. (1980) 77, p. 4126-4220). Moreover, when using a prokaryotic cell, Escherichia coli and Bacillus subtilis can be mentioned, for example. An antibody can be obtained by introducing a desired antibody gene into these cells by transformation, and culturing the transformed cells in vitro. In the above culture method, the yield may vary depending on the sequence of the antibody. From the antibodies having equivalent binding activity, those that can be easily produced as pharmaceuticals can be selected using the yield as an index.
本発明の抗体のアイソタイプとしての制限はなく、例えばIgG(IgG1,IgG2,IgG3,IgG4)、IgM、IgA(IgA1,IgA2)、IgDあるいはIgE等を挙げることができるが、好ましくはIgGまたはIgM、さらに好ましくはIgG1またはIgG2を挙げることができる。 There is no limitation as to the isotype of the antibody of the present invention, and examples include IgG (IgG1, IgG2, IgG3, IgG4), IgM, IgA (IgA1, IgA2), IgD or IgE, preferably IgG or IgM, More preferably, IgG1 or IgG2 can be mentioned.
また本発明の抗体は、抗体の抗原結合部を有する抗体の抗原結合性断片またはその修飾物であってもよい。抗体をパパイン、ペプシン等の蛋白質分解酵素で処理するか、あるいは抗体遺伝子を遺伝子工学的手法によって改変し適当な培養細胞において発現させることによって、該抗体の断片を得ることができる。このような抗体断片のうちで、抗体全長分子の持つ機能の全てまたは一部を保持している断片を抗体の抗原結合性断片と呼ぶことができる。抗体の機能としては、一般的には抗原結合活性、抗原の活性を中和する活性、抗原の活性を増強する活性、抗体依存性細胞傷害活性、補体依存性細胞傷害活性および補体依存性細胞性細胞傷害活性を挙げることができる。本発明における抗体の抗原結合性断片が保持する機能は、EPHA2に対する結合活性である。 The antibody of the present invention may be an antigen-binding fragment of an antibody having an antigen-binding portion of the antibody or a modified product thereof. A fragment of the antibody can be obtained by treating the antibody with a proteolytic enzyme such as papain or pepsin, or modifying the antibody gene by a genetic engineering technique and expressing it in an appropriate cultured cell. Among such antibody fragments, a fragment that retains all or part of the functions of the full-length antibody molecule can be called an antigen-binding fragment of an antibody. Antibody functions generally include antigen-binding activity, activity that neutralizes antigen activity, activity that enhances antigen activity, antibody-dependent cytotoxic activity, complement-dependent cytotoxic activity, and complement-dependence Mention may be made of cellular cytotoxic activity. The function retained by the antigen-binding fragment of the antibody in the present invention is the binding activity to EPHA2.
例えば、抗体の断片としては、Fab、F(ab’)2、Fv、または重鎖および軽鎖のFvを適当なリンカーで連結させたシングルチェインFv(scFv)、diabody(diabodies)、線状抗体、および抗体断片より形成された多特異性抗体などを挙げることができる。また、F(ab’)2を還元条件下で処理した抗体の可変領域の一価の断片であるFab’も抗体の断片に含まれる。 For example, antibody fragments include Fab, F (ab ′) 2, Fv, or single chain Fv (scFv), diabodies (diabodies), linear antibody in which heavy and light chain Fvs are linked by an appropriate linker. And multispecific antibodies formed from antibody fragments. Fab ', which is a monovalent fragment of the variable region of an antibody obtained by treating F (ab') 2 under reducing conditions, is also included in the antibody fragment.
さらに、本発明の抗体は少なくとも2種類の異なる抗原に対して特異性を有する多特異性抗体であってもよい。通常このような分子は2種類の抗原に結合するものであるが(即ち、二重特異性抗体(bispecific antibody))、本発明における「多特異性抗体」は、それ以上(例えば、3種類)の抗原に対して特異性を有する抗体を包含するものである。 Furthermore, the antibody of the present invention may be a multispecific antibody having specificity for at least two different antigens. Usually, such a molecule binds to two types of antigens (ie, bispecific antibodies), but the “multispecific antibody” in the present invention is more than that (for example, three types). It includes an antibody having specificity for the antigens.
本発明の多特異性抗体は、全長からなる抗体、またはそのような抗体の断片(例えば、F(ab’)2二重特異性抗体)でもよい。二重特異性抗体は2種類の抗体の重鎖と軽鎖(HL対)を結合させて作製することもできるし、異なるモノクローナル抗体を産生するハイブリドーマを融合させて、二重特異性抗体産生融合細胞を作製することによっても、作製することができる(Millstein et al.,Nature(1983)305,p.537-539)。 The multispecific antibody of the present invention may be a full-length antibody or a fragment of such an antibody (for example, F (ab ') 2 bispecific antibody). Bispecific antibodies can be prepared by combining the heavy and light chains (HL pairs) of two types of antibodies, or by hybridizing hybridomas that produce different monoclonal antibodies to produce a bispecific antibody. It can also be produced by producing cells (Millstein et al., Nature (1983) 305, p. 537-539).
本発明の抗体は一本鎖抗体(scFvとも記載する)でもよい。一本鎖抗体は、抗体の重鎖可変領域と軽鎖可変領域とをポリペプチドのリンカーで連結することにより得られる(Pluckthun,The Pharmacology of Monoclonal Antibodies,113(RosenbergおよびMoore編、Springer Verlag,New York,p.269-315(1994)、Nature Biotechnology(2005),23,p.1126-1136)。また、2つのscFvをポリペプチドリンカーで結合させて作製されるBiscFv断片を二重特異性抗体として使用することもできる。 The antibody of the present invention may be a single chain antibody (also referred to as scFv). A single chain antibody is obtained by linking an antibody heavy chain variable region and a light chain variable region with a polypeptide linker (Pluckthun, The Pharmacology of Monoclonal Antibodies, 113 (Rosenberg and Moore, edited by Springer Verlag, New). York, p. 269-315 (1994), Nature Biotechnology (2005), 23, p. 1126-1136) In addition, a BiscFv fragment produced by linking two scFvs with a polypeptide linker is used as a bispecific antibody. It can also be used.
一本鎖抗体を作製する方法は当技術分野において周知である(例えば、米国特許第4,946,778号、米国特許第5,260,203号、米国特許第5,091,513号、米国特許第5,455,030号等を参照)。このscFvにおいて、重鎖可変領域と軽鎖可変領域は、コンジュゲートを作らないようなリンカー、好ましくはポリペプチドリンカーを介して連結される(Huston,J.S.et al.,Proc.Natl.Acad.Sci.U.S.A.(1988),85,p.5879-5883)。scFvにおける重鎖可変領域および軽鎖可変領域は、同一の抗体に由来してもよく、別々の抗体に由来してもよい。可変領域を連結するポリペプチドリンカーとしては、例えば12~19残基からなる任意の一本鎖ペプチドが用いられる。 Methods for making single chain antibodies are well known in the art (eg, US Pat. No. 4,946,778, US Pat. No. 5,260,203, US Pat. No. 5,091,513, US (See Japanese Patent No. 5,455,030). In this scFv, the heavy chain variable region and the light chain variable region are linked via a linker that does not form a conjugate, preferably a polypeptide linker (Huston, JS et al., Proc. Natl. Acad. Sci.U.S.A. (1988), 85, p.5879-5883). The heavy chain variable region and the light chain variable region in scFv may be derived from the same antibody or different antibodies. As the polypeptide linker that links the variable regions, for example, any single chain peptide consisting of 12 to 19 residues is used.
scFvをコードするDNAは、前記抗体の重鎖または重鎖可変領域をコードするDNA、および軽鎖または軽鎖可変領域をコードするDNAのうち、それらの配列のうちの全部または所望のアミノ酸配列をコードするDNA部分を鋳型とし、その両端を規定するプライマー対を用いてPCR法により増幅し、次いで、さらにポリペプチドリンカー部分をコードするDNA、およびその両端が各々重鎖、軽鎖と連結されるように規定するプライマー対を組み合わせて増幅することにより得られる。 The DNA encoding the scFv is the DNA encoding the heavy chain or heavy chain variable region of the antibody, and the DNA encoding the light chain or light chain variable region. Amplification is performed by PCR using a coding DNA portion as a template and a primer pair defining both ends thereof, and then the DNA encoding the polypeptide linker portion and both ends thereof are connected to the heavy chain and light chain, respectively. Obtained by combining and amplifying the primer pairs defined in 1.
また、一旦scFvをコードするDNAが作製されると、それらを含有する発現ベクター、および該発現ベクターにより形質転換された宿主を常法に従って得ることができ、また、その宿主を用いることにより、常法に従ってscFvを得ることができる。これらの抗体断片は、前記と同様にして遺伝子を取得し発現させ、宿主により産生させることができる。 In addition, once a DNA encoding scFv is prepared, an expression vector containing them and a host transformed with the expression vector can be obtained according to conventional methods, and by using the host, ScFv can be obtained according to the method. These antibody fragments can be produced by a host after obtaining and expressing the gene in the same manner as described above.
本発明の抗体は、多量化して抗原に対する親和性を高めたものであってもよい。多量化する抗体としては、1種類の抗体であっても、同一の抗原の複数のエピトープを認識する複数の抗体であってもよい。抗体を多量化する方法としては、IgG CH3ドメインと2つのscFvとの結合、ストレプトアビジンとの結合、へリックスーターン-へリックスモチーフの導入等を挙げることができる。 The antibody of the present invention may be one that has been increased in quantity and has increased affinity for the antigen. The antibody that multiplies may be one type of antibody or a plurality of antibodies that recognize multiple epitopes of the same antigen. Examples of the method for increasing the amount of antibody include binding of IgG CH3 domain to two scFvs, binding to streptavidin, and introduction of helix-turn-helix motif.
本発明の抗体は、アミノ酸配列が異なる複数種類の抗EPHA2抗体の混合物である、ポリクローナル抗体であってもよい。ポリクローナル抗体の一例としては、CDRが異なる複数種類の抗体の混合物を挙げることができる。そのようなポリクローナル抗体としては、異なる抗体を産生する細胞の混合物を培養し、該培養物から精製された抗体を用いることが出来る(WO2004/061104号参照)。 The antibody of the present invention may be a polyclonal antibody that is a mixture of a plurality of types of anti-EPHA2 antibodies having different amino acid sequences. As an example of the polyclonal antibody, a mixture of plural kinds of antibodies having different CDRs can be mentioned. As such a polyclonal antibody, a mixture of cells producing different antibodies can be cultured, and an antibody purified from the culture can be used (see WO 2004/061104).
抗体の修飾物として、ポリエチレングリコール(PEG)等の各種分子と結合した抗体を使用することもできる。 As the modified antibody, an antibody conjugated with various molecules such as polyethylene glycol (PEG) can also be used.
本発明の抗体は、更にこれらの抗体と他の薬剤がコンジュゲートを形成しているもの(Immunoconjugate)でもよい。このような抗体の例としては、該抗体が放射性物質や薬理作用を有する化合物と結合している物を挙げることができる(Nature Biotechnology(2005)23,p.1137-1146)。 The antibody of the present invention may be one in which these antibody and another drug form a conjugate (Immunoconjugate). Examples of such an antibody include those in which the antibody is bound to a radioactive substance or a compound having a pharmacological action (Nature Biotechnology (2005) 23, p. 1137-1146).
得られた抗体は、均一にまで精製することができる。抗体の分離、精製は通常の蛋白質で使用されている分離、精製方法を使用すればよい。例えばカラムクロマトグラフィー、フィルター濾過、限外濾過、塩析、透析、調製用ポリアクリルアミドゲル電気泳動、等電点電気泳動等を適宜選択、組み合わせれば、抗体を分離、精製することができる(Strategies for Protein Purification and Characterization:A Laboratory Course Manual,Daniel R.Marshak et al.eds.,Cold Spring Harbor Laboratory Press(1996);Antibodies:A Laboratory Manual.Ed Harlow and David Lane,Cold Spring Harbor Laboratory(1988))が、これらに限定されるものではない。 The obtained antibody can be purified to homogeneity. Separation and purification of antibodies may be carried out using separation and purification methods used for ordinary proteins. For example, antibodies can be separated and purified by appropriately selecting and combining column chromatography, filter filtration, ultrafiltration, salting out, dialysis, preparative polyacrylamide gel electrophoresis, isoelectric focusing, etc. (Stratesies) for Protein Purification and Characterization: A Laboratory Course Manual, Daniel R.Marshak et al.eds, Cold Spring Harbor Laboratory Press (1996); Antibodies:. A Laboratory Manual.Ed Harlow and David Lane, Cold Spring Harbor Laboratory ( 988)) it is not intended to be limited thereto.
クロマトグラフィーとしては、アフィニティークロマトグラフィー、イオン交換クロマトグラフィー、疎水性クロマトグラフィー、ゲル濾過クロマトグラフィー、逆相クロマトグラフィー、吸着クロマトグラフィー等を挙げることができる。これらのクロマトグラフィーは、HPLCやFPLC等の液体クロマトグラフィーを用いて行うことができる。アフィニティークロマトグラフィーに用いるカラムとしては、プロテインAカラム、プロテインGカラムを挙げることができる。例えばプロテインAカラムを用いたカラムとして、Hyper D,POROS,Sepharose F.F.(ファルマシア)等を挙げることができる。また、抗原を固定化した担体を用いて、抗原への結合性を利用して抗体を精製することも可能である。 Examples of chromatography include affinity chromatography, ion exchange chromatography, hydrophobic chromatography, gel filtration chromatography, reverse phase chromatography, and adsorption chromatography. These chromatography can be performed using liquid chromatography, such as HPLC and FPLC. Examples of the column used for affinity chromatography include a protein A column and a protein G column. For example, as a column using a protein A column, Hyper D, POROS, Sepharose F.R. F. (Pharmacia) and the like. It is also possible to purify an antibody using a carrier on which an antigen is immobilized, utilizing the binding property to the antigen.
3.医薬組成物
本発明は抗EPHA2抗体もしくはその機能断片またはその修飾体を含む医薬組成物を提供する。
3. Pharmaceutical Composition The present invention provides a pharmaceutical composition comprising an anti-EPHA2 antibody or a functional fragment thereof or a modified form thereof.
本発明の医薬組成物はEPHA2もしくはそのリガンドの過剰発現またはEPHA2の変異もしくは遺伝子増幅によるEPHA2シグナル異常または亢進、または、EPHA2のアイソフォームのスイッチングにより惹起されるかまたは増悪化される各種疾患(以下、「EPHA2に関わる疾患」という)、とりわけ各種癌の治療または予防に有用である。 The pharmaceutical composition of the present invention can be used in various diseases (hereinafter referred to as “induced or exacerbated” by EPHA2 signal abnormality or enhancement by EPHA2 or its ligand overexpression or EPHA2 mutation or gene amplification, or by switching of EPHA2 isoforms). , Referred to as “diseases associated with EPHA2”), especially for the treatment or prevention of various cancers.
かかる治療または予防の対象となる癌の惹起または増悪化の原因としては、EPHA2遺伝子のイントロン内の一塩基置換(SNP)、EPHA2の高発現、EPHA2を恒常的に活性化するミスセンス変異、EPHA2遺伝子の増幅または過剰発現、EPHA2アイソフォームのスイッチング等を例示することができる。 Causes of the induction or exacerbation of cancer that is the subject of treatment or prevention include single nucleotide substitution (SNP) in the intron of the EPHA2 gene, high expression of EPHA2, a missense mutation that constantly activates EPHA2, and the EPHA2 gene Amplification or overexpression, switching of EPHA2 isoform, etc. can be exemplified.
かかる癌種としては、例えば、乳癌、子宮内膜癌、卵巣癌、非小細胞肺癌などの肺癌、胃癌、前立腺癌、腎癌、肝臓癌、膵臓癌、大腸癌、食道癌、膀胱癌、子宮頚癌、血液癌、リンパ腫、悪性黒色腫等をあげることができ、好適にはEPHA2蛋白質を発現しているそれらの癌をあげることができる。 Examples of such cancer types include lung cancer such as breast cancer, endometrial cancer, ovarian cancer, non-small cell lung cancer, gastric cancer, prostate cancer, kidney cancer, liver cancer, pancreatic cancer, colon cancer, esophageal cancer, bladder cancer, uterus. Cervical cancer, hematological cancer, lymphoma, malignant melanoma and the like can be mentioned, and those cancers expressing the EPHA2 protein can be mentioned preferably.
本発明において、疾患の治療または予防には、かかる疾患、好適にはEPHA2蛋白質を発現している個体におけるかかる疾患の発症の予防、増悪化または進行の抑制または阻害、かかる疾患に罹患した個体が呈する一つまたは二つ以上の症状の軽減、増悪化もしくは進行の抑制または寛解、二次性疾患の治療または予防等が含まれるが、それらに限定されるものではない。 In the present invention, for the treatment or prevention of a disease, the onset of such a disease, preferably the onset of such a disease in an individual expressing the EPHA2 protein, the suppression or inhibition of progression or progression, and the individual suffering from such a disease Examples include, but are not limited to, alleviation of one or more symptoms present, suppression or remission of progression or progression, treatment or prevention of secondary diseases, and the like.
本発明の医薬組成物には、治療または予防に有効な量の抗EPHA2抗体または該抗体の機能断片と薬学上許容される希釈剤、担体、可溶化剤、乳化剤、保存剤および/または補助剤を含有せしめることができる。 The pharmaceutical composition of the present invention comprises a therapeutically or prophylactically effective amount of an anti-EPHA2 antibody or a functional fragment of the antibody and a pharmaceutically acceptable diluent, carrier, solubilizer, emulsifier, preservative and / or adjuvant. Can be contained.
「治療または予防に有効な量」とは、特定の疾患、投与形態および投与径路につき治療または予防効果を奏する量を意味し、「薬理学的に有効な量」と同義である。 “Therapeutically or prophylactically effective amount” means an amount that exhibits a therapeutic or prophylactic effect for a specific disease, administration form, and administration route, and is synonymous with “pharmacologically effective amount”.
本発明の医薬組成物には、pH、浸透圧、粘度、透明度、色、等張性、無菌性、該組成物またはそれに含まれる抗体の安定性、溶解性、徐放性、吸収性、浸透性、剤型、強度、性状、形状等を変化させたり、維持したり、保持したりするための物質(以下、「製剤用の物質」という)を含有せしめることができる。製剤用の物質としては、薬理学的に許容される物質であれば特に限定されるものではない。例えば、非毒性または低毒性であることは、製剤用の物質が好適に具備する性質である。 The pharmaceutical composition of the present invention has pH, osmotic pressure, viscosity, transparency, color, isotonicity, sterility, stability of the composition or antibody contained therein, solubility, sustained release, absorbability, penetration. Substances for changing, maintaining, and maintaining properties, dosage forms, strength, properties, shapes, etc. (hereinafter referred to as “substances for formulation”) can be included. The substance for the preparation is not particularly limited as long as it is a pharmacologically acceptable substance. For example, non-toxicity or low toxicity is a property that a substance for preparation preferably comprises.
製剤用の物質として、例えば、アミノ酸類、抗菌剤、抗酸化剤、緩衝剤、充填剤、キレート剤、錯化剤、増量剤、単糖類、二糖類、炭水化物、着色剤、香味剤、希釈剤、乳化剤、親水ポリマー、の防腐剤、溶媒、糖アルコール、懸濁剤、界面活性剤、安定化増強剤、弾性増強剤、輸送剤、希釈剤、賦形剤、および/または薬学上の補助剤 等を挙げることができ、それらの物質の添加量は、抗EPHA2抗体もしくはその機能断片またはその修飾体の重量に対して0.001乃至1000倍、好適には0.01乃至100倍、より好適には0.1乃至10倍である。 Substances for formulation include, for example, amino acids, antibacterial agents, antioxidants, buffers, fillers, chelating agents, complexing agents, bulking agents, monosaccharides, disaccharides, carbohydrates, coloring agents, flavoring agents, diluents , Emulsifier, hydrophilic polymer, preservative, solvent, sugar alcohol, suspending agent, surfactant, stabilization enhancer, elasticity enhancer, transport agent, diluent, excipient, and / or pharmaceutical adjuvant The amount of these substances to be added is 0.001 to 1000 times, preferably 0.01 to 100 times, more preferably the weight of the anti-EPHA2 antibody or functional fragment or modified product thereof. Is 0.1 to 10 times.
抗EPHA2抗体もしくはその機能断片またはその修飾体をリポソーム中に含有せしめたイムノリポソーム、抗体とリポソームとが結合してなる抗体修飾体(米国特許第6214388号等)を含有する医薬組成物も、本発明の医薬組成物に含まれる。 A pharmaceutical composition containing an anti-EPHA2 antibody or a functional fragment thereof or a modified product thereof in an liposome, or an antibody modified product (US Pat. No. 6,214,388, etc.) in which an antibody and a liposome are bound is also present. Included in the pharmaceutical composition of the invention.
賦形剤や担体は、通常液体または固体であり、注射用の水、生理食塩水、人工脳脊髄液、その他の、経口投与または非経口投与用の製剤に用いられる物質であれば特に限定されない。生理食塩水としては、中性のもの、血清アルブミンを含むもの等をあげることができる。 The excipient or carrier is usually liquid or solid, and is not particularly limited as long as it is a substance used for water for injection, physiological saline, artificial cerebrospinal fluid, and other preparations for oral administration or parenteral administration. . Examples of the physiological saline include neutral ones and those containing serum albumin.
緩衝剤としては、医薬組成物の最終pHが7.0乃至8.5になるように調製されたTrisバッファー、同じく4.0乃至5.5になるように調製された酢酸バッファー、同じく5.0乃至8.0になるように調製されたクエン酸バッファー、同じく5.0乃至8.0になるように調製されたヒスチジンバッファー等を例示することができる。 Examples of the buffer include Tris buffer prepared so that the final pH of the pharmaceutical composition is 7.0 to 8.5, acetate buffer prepared so as to be 4.0 to 5.5, and 5. Examples thereof include a citrate buffer prepared to be 0 to 8.0, a histidine buffer prepared to be 5.0 to 8.0, and the like.
本発明の医薬組成物は、固体、液体、懸濁液等である。凍結乾燥製剤をあげることができる。凍結乾燥製剤を成型するには、スクロース等の賦形剤を用いることができる。 The pharmaceutical composition of the present invention is a solid, liquid, suspension or the like. Freeze-dried preparations can be mentioned. An excipient such as sucrose can be used to mold the lyophilized preparation.
本発明の医薬組成物の投与径路としては、経腸投与、局所投与および非経口投与のいずれでもよく、例えば、静脈内投与、動脈内投与、筋肉内投与、皮内投与、皮下投与、腹腔内投与、経皮投与、骨内投与、関節内投与等をあげることができる。 The administration route of the pharmaceutical composition of the present invention may be any of enteral administration, topical administration and parenteral administration. For example, intravenous administration, intraarterial administration, intramuscular administration, intradermal administration, subcutaneous administration, intraperitoneal administration Administration, transdermal administration, intraosseous administration, intraarticular administration and the like can be mentioned.
かかる医薬組成物の組成は、投与方法、抗体のEPHA2蛋白質結合親和性等に応じて決定することができる。本発明の抗EPHA2抗体もしくはその機能断片またはその修飾体のEPHA2蛋白質に対する親和性が高いほど(KD値が低いほど)、少ない投与量でその薬効を発揮し得る。 The composition of such a pharmaceutical composition can be determined according to the administration method, the EPHA2 protein binding affinity of the antibody, and the like. The higher the affinity of the anti-EPHA2 antibody of the present invention or the functional fragment thereof or the modified product thereof for the EPHA2 protein (the lower the KD value), the more effective the drug can be exerted.
本発明の抗EPHA2抗体の投与量は、薬理学的に有効な量であれば限定されず、個体の種、疾患の種類、症状、性別、年齢、持病、該抗体のEPHA2蛋白質結合親和性またはその生物活性、その他の要素に応じて適宜決定することができるが、通常、0.01乃至1000mg/kg、好適には0.1乃至100mg/kgを、1乃至180日間に1回、または1日2回もしくは3回以上投与することができる。 The dose of the anti-EPHA2 antibody of the present invention is not limited as long as it is a pharmacologically effective amount, and the species of the individual, the type of disease, the symptom, sex, age, prevalence, EPHA2 protein binding affinity of the antibody or Although it can be appropriately determined depending on the biological activity and other factors, it is usually 0.01 to 1000 mg / kg, preferably 0.1 to 100 mg / kg once every 1 to 180 days, or 1 It can be administered twice or more times a day.
医薬組成物の形態としては、注射剤(凍結乾燥製剤、点滴剤を含む)、坐剤、経鼻型吸収製剤、経皮型吸収製剤、舌下剤、カプセル、錠剤、軟膏剤、顆粒剤、エアーゾル剤、丸剤、散剤、懸濁剤、乳剤、点眼剤、生体埋め込み型製剤等を例示することができる。 The form of the pharmaceutical composition includes injections (including lyophilized preparations and infusions), suppositories, nasal absorption preparations, transdermal absorption preparations, sublingual preparations, capsules, tablets, ointments, granules, aerosols. Examples thereof include pills, pills, powders, suspensions, emulsions, eye drops, and implantable preparations.
抗EPHA2抗体もしくはその機能断片またはその修飾体を有効成分として含む医薬組成物は他の医薬と同時にあるいは個々に投与することができる。例えば、他の医薬を投与した後に抗EPHA2抗体または該抗体の機能断片を有効成分として含む医薬組成物を投与するか、かかる医薬組成物を投与した後に、他の医薬を投与するか、または、当該医薬組成物と他の医薬とを同時に投与してもよい。他の医薬としては、化学療法剤、放射線療法など各種抗癌剤等をあげることができる。それらをまとめて本発明の抗体と「他の薬剤との併用」と呼び、本発明の抗体、その機能断片またはその修飾体に加えてさらなる薬剤を含む医薬組成物も本発明に含まれる。 A pharmaceutical composition comprising an anti-EPHA2 antibody or a functional fragment thereof or a modified form thereof as an active ingredient can be administered simultaneously with or separately from other drugs. For example, after administering another pharmaceutical, a pharmaceutical composition containing an anti-EPHA2 antibody or a functional fragment of the antibody as an active ingredient is administered, or after administering such a pharmaceutical composition, another pharmaceutical is administered, or You may administer the said pharmaceutical composition and another pharmaceutical simultaneously. Examples of other medicaments include various anticancer agents such as chemotherapeutic agents and radiotherapy. These are collectively referred to as “the combined use of the antibody of the present invention with another drug”, and a pharmaceutical composition containing an additional drug in addition to the antibody of the present invention, a functional fragment thereof or a modified form thereof is also included in the present invention.
本発明は癌などEPHA2に関わる疾患の治療方法または予防方法、該疾患の治療用または予防用医薬組成物を調製するための本発明の抗体の使用、該疾患の治療または予防のための本発明の抗体の使用、をも提供する。本発明の抗体を含む治療用または予防用キットも本発明に含まれる。 The present invention relates to a method for treating or preventing a disease associated with EPHA2, such as cancer, the use of the antibody of the present invention for preparing a pharmaceutical composition for treating or preventing the disease, and the present invention for treating or preventing the disease. Of the use of antibodies. A therapeutic or prophylactic kit containing the antibody of the present invention is also included in the present invention.
4.診断用組成物
本発明の抗EPHA2抗体もしくはその機能断片またはその修飾体を含む検査用または診断用組成物(以下、まとめて「診断用組成物」という)を提供する。
4). Diagnostic Composition A diagnostic or diagnostic composition (hereinafter collectively referred to as “diagnostic composition”) comprising the anti-EPHA2 antibody of the present invention or a functional fragment thereof or a modified form thereof is provided.
本発明の診断用組成物は、癌などEPHA2に関わる疾患、EPHA2発現の検査または診断に有用である。本発明において検査または診断には、例えば、罹患リスクの判定または測定、罹患の有無の判定、進行や増悪化の程度の測定、抗EPHA2抗体等の医薬組成物による薬物治療の効果の測定または判定、薬物治療以外の治療の効果の測定または判定、再発リスクの測定、再発の有無の判定等が含まれるが、検査または診断であればそれらに限定されるものではない。 The diagnostic composition of the present invention is useful for testing or diagnosis of diseases related to EPHA2, such as cancer, and EPHA2 expression. The examination or diagnosis in the present invention includes, for example, determination or measurement of morbidity risk, determination of the presence or absence of morbidity, measurement of the degree of progression or deterioration, measurement or determination of the effect of drug treatment with a pharmaceutical composition such as an anti-EPHA2 antibody. In addition, measurement or determination of the effect of treatment other than drug treatment, measurement of recurrence risk, determination of the presence or absence of recurrence, and the like are included, but the test or diagnosis is not limited thereto.
本発明の診断用組成物は、本発明の抗体もしくはその機能断片またはその修飾体、それらを含む組成物、それらを含む医薬組成物を投与する個体の同定に有用である。 The diagnostic composition of the present invention is useful for identification of an individual to which the antibody of the present invention or a functional fragment thereof or a modified product thereof, a composition containing them, or a pharmaceutical composition containing them is administered.
かかる診断用組成物には、pH緩衝剤、浸透圧調節剤、塩類、安定化剤、防腐剤、顕色剤、増感剤、凝集防止剤等を含有せしめることができる。 Such a diagnostic composition may contain a pH buffer, an osmotic pressure regulator, salts, a stabilizer, a preservative, a developer, a sensitizer, an aggregation inhibitor, and the like.
本発明は癌などEPHA2に関わる疾患の検査方法または診断方法、該疾患の診断用組成物を調製するための本発明の抗体の使用、該疾患の検査または診断のための本発明の抗体の使用、をも提供する。本発明の抗体を含む検査または診断用キットも本発明に含まれる。 The present invention relates to a method for testing or diagnosing a disease associated with EPHA2, such as cancer, use of the antibody of the present invention for preparing a diagnostic composition for the disease, use of the antibody of the present invention for testing or diagnosing the disease , Also provide. A test or diagnostic kit containing the antibody of the present invention is also included in the present invention.
本発明の抗体を含む検査または診断の方法としてはサンドウィッチELISAが望ましいが、通常のELISA法やRIA法、ELISPOT(Enzyme-Linked ImmunoSpot)法、ドットブロット法、オクタロニー法、CIE(Counterimmunoelectrophoresis)法、CLIA(Chemiluminescent immuno assay)、FCM(Flow Cytometry)などの抗体を利用した検出方法が利用可能である。抗体の標識法としてはビオチンのほか、HRP、アルカリフォスファターゼ、FITCやALEXAなどの発蛍光団、放射性同位元素などのラベルなど生化学的解析に実施可能な標識法が利用できる。酵素標識を利用した検出にはTMB(3,3’,5,5’-tetramethylbenzidine)、BCIP(5-bromo-4-chloro-3-indolyl phosphate)、ρ-NPP(ρ-nitrophenyl phosphate)、OPD(o-Phenylenediamine)、ABTS(3-Ethylbenzothiazoline-6-sulfonic acid)、SuperSignal ELISA Pico Chemiluminescent Substrate(サーモフィッシャーサイエンティフィック社)などの発色基質やQuantaBluTM Fluorogenic Peroxidase Substrate(サーモフィッシャーサイエンティフィック社)蛍光基質のほか、化学発光基質を用いることができる。本測定には、ヒトまたは非ヒト動物由来の試料に加え、組換え蛋白質等の人工的な処理を加えた試料をも供することができる。生物個体由来の被検試料としては、例えば、血液、関節液、腹水、リンパ液、脳脊髄液、組織ホモジネート上清、組織切片等をあげることができるが、それらに限定されるものではない。 A sandwich ELISA is preferable as a test or diagnostic method including the antibody of the present invention, but a normal ELISA method, RIA method, ELISPOT (Enzyme-Linked ImmunoSpot) method, dot blot method, octalony method, CIE (Counterimmunoelectrophoresis) method, CLIA Detection methods using antibodies such as (Chemiluminescent immunoassay) and FCM (Flow Cytometry) can be used. As a labeling method for antibodies, in addition to biotin, labeling methods that can be used for biochemical analysis such as labels for fluorophores such as HRP, alkaline phosphatase, FITC and ALEXA, and radioisotopes can be used. For detection using an enzyme label, TMB (3,3 ′, 5,5′-tetramethylbenzidine), BCIP (5-bromo-4-chloro-3-indoyl phosphate), ρ-NPP (ρ-nitrophenyl phosphate), OPD Fluorescent substrates such as (O-Phenylenediamine), ABTS (3-Ethylbenzothiazine-6-sulfonic acid), SuperSignal ELISA Pico Chemiluminescent Substrate TM (Sermo Fisher Scientific) Substrate In addition, it is possible to use chemiluminescent substrate. For this measurement, in addition to a sample derived from a human or non-human animal, a sample to which an artificial treatment such as a recombinant protein has been added can also be used. Examples of test samples derived from living organisms include, but are not limited to, blood, joint fluid, ascites, lymph, cerebrospinal fluid, tissue homogenate supernatant, tissue section, and the like.
本発明の抗体を含む検査または診断用のサンドウィッチELISAキットには、EPHA2蛋白質標準液、発色試薬、希釈用緩衝液、固相用抗体、検出用抗体、ならびに洗浄液等が含まれてよい。抗原に結合した抗体量を測定する方法としては、吸光法、蛍光法、発光法、RI(Radioisotope)法等が好適に適用され、測定には、吸光プレートリーダー、蛍光プレートリーダー、発光プレートリーダー、RI液体シンチレーションカウンター等が好適に使用される。 The test or diagnostic sandwich ELISA kit containing the antibody of the present invention may contain an EPHA2 protein standard solution, a coloring reagent, a dilution buffer, a solid phase antibody, a detection antibody, a washing solution, and the like. As a method for measuring the amount of antibody bound to the antigen, an absorption method, a fluorescence method, a luminescence method, an RI (Radioisotope) method or the like is preferably applied. An RI liquid scintillation counter or the like is preferably used.
前記の免疫組織学的試験だけでなく、試料中の細胞、組織または臓器もしくはその一部から、常法に従って可溶性蛋白を調製し、当該可溶性蛋白に標識化抗体を反応させることにより可溶性蛋白中のEPHA2の存否を確認するウェスタンブロッティング法やドットブロット法においても用いることができる。 In addition to the immunohistological test described above, a soluble protein is prepared from cells, tissues or organs or a part thereof in a sample according to a conventional method, and the soluble protein is reacted with a labeled antibody in the soluble protein. It can also be used in Western blotting or dot blotting to confirm the presence or absence of EPHA2.
本発明は免疫組織化学(immunohistochemistry:IHC)の分析に有用な抗体、その機能断片およびその修飾体、ならびにそれらを含む組成物を提供する。かかる組成物も本発明の「診断用組成物」に包含される。 The present invention provides antibodies useful for immunohistochemistry (IHC) analysis, functional fragments thereof and modifications thereof, and compositions containing them. Such a composition is also included in the “diagnostic composition” of the present invention.
免疫組織化学は、組織切片を抗原に結合する抗体(一次抗体)を反応させ、抗原に結合した一次抗体を検出する手法であれば特に限定されない。
組織切片は、好適にはホルマリン固定後にパラフィン包埋処理する。パラフィン包埋後、薄切した組織切片を脱パラフィン処理した後、抗原賦活処理および非特異的反応抑制処理を行う。抗原賦活処理の方法としては、加熱処理、プロテアーゼ等による酵素処理を例示することができ、好適には加熱処理である。加熱処理の条件としては、通常、温度90乃至110℃、pH8乃至10、処理時間20乃至60分の範囲が好適である。pHの調整にはTris-EDTA緩衝液(例えば、1mMのEDTAを含有する10mMトリス緩衝液)等を使用することができる。非特異的反応抑制処理としては、発色に使用する酵素と同様または類似の触媒活性を有する内因性の酵素を不活性化する方法が通常用いられる。ペルオキシダーゼ反応により発色させる場合、予め組織中に存在する内因性のペルオキシダーゼをH2O2等で阻害することが好ましい。H2O2の溶媒は水、メタノール等を使用することができ、H2O2の濃度は0.1乃至3%、好適には0.3乃至3%である。H2O2溶液にはアジ化ナトリウムを添加することができる。また、血清やカゼインによりブロッキングする方法も非特異的反応抑制処理として使用することができる。血清やカゼインは、一次抗体反応の前に組織を処理することができるが、1次抗体を希釈する溶媒に含有せしめることもできる。
一次抗体の反応条件は特に限定されないが、温度4乃至50℃、好適には20乃至37℃、より好適には24℃である。反応時間は5分乃至一昼夜、好適には10分乃至4時間、より好適には30分乃至1時間である。
Immunohistochemistry is not particularly limited as long as it is a technique in which a tissue section is reacted with an antibody (primary antibody) that binds to an antigen to detect the primary antibody bound to the antigen.
Tissue sections are preferably paraffin-embedded after formalin fixation. After embedding in paraffin, the sliced tissue section is deparaffinized and then subjected to antigen activation treatment and nonspecific reaction suppression treatment. Examples of the antigen activation treatment method include heat treatment, enzyme treatment with protease, etc., and heat treatment is preferred. As conditions for the heat treatment, a temperature range of 90 to 110 ° C., a pH of 8 to 10 and a treatment time of 20 to 60 minutes are usually preferable. Tris-EDTA buffer (for example, 10 mM Tris buffer containing 1 mM EDTA) or the like can be used for pH adjustment. As the nonspecific reaction suppression treatment, a method of inactivating an endogenous enzyme having a catalytic activity similar to or similar to the enzyme used for color development is usually used. When color is developed by a peroxidase reaction, it is preferable to inhibit endogenous peroxidase existing in the tissue in advance with H 2 O 2 or the like. As the H 2 O 2 solvent, water, methanol or the like can be used, and the concentration of H 2 O 2 is 0.1 to 3%, preferably 0.3 to 3%. Sodium azide can be added to the H 2 O 2 solution. A method of blocking with serum or casein can also be used as a nonspecific reaction suppression treatment. Serum and casein can treat tissues before the primary antibody reaction, but can also be included in a solvent that dilutes the primary antibody.
The reaction conditions for the primary antibody are not particularly limited, but the temperature is 4 to 50 ° C, preferably 20 to 37 ° C, more preferably 24 ° C. The reaction time is 5 minutes to 1 day, preferably 10 minutes to 4 hours, more preferably 30 minutes to 1 hour.
一次抗体の検出には、好適には、可視化することができ且つ一次抗体に結合する抗体(二次抗体)を用いることができる。二次抗体自体に結合する抗体(三次抗体)を用いて3回以上の反応を行う場合もある。二次抗体または三次抗体の可視化法としては、ペルオキシダーゼやアルカリフォスファターゼ等の酵素をそれらの抗体に結合させるか、またはそれらの抗体にビオチン等を付加して前記酵素を結合させたストレプトアビジン等と結合させ、それらの酵素に対応する発色基質を反応させる方法を好適に使用することができる。酵素を二次抗体または三次抗体に結合させる方法としては、デキストリンポリマーやアミノ酸ポリマーに前記酵素と二次抗体を多数結合させた試薬を用いる方法(ポリマー法)を例示することができる。ビオチン化二次抗体およびペルオキシダーゼ標識したストレプトアビジンを反応させる方法(LSAB法)では、発色基質としてDAB等を用いることができる。また、蛍光色素などで標識された二次抗体を使用することもできる。蛍光標識二次抗体で処理した場合、処理後に蛍光顕微鏡を用いて陽性細胞を検出する。 For detection of the primary antibody, an antibody that can be visualized and binds to the primary antibody (secondary antibody) can be preferably used. In some cases, the reaction is performed three or more times using an antibody (tertiary antibody) that binds to the secondary antibody itself. The secondary or tertiary antibody can be visualized by binding an enzyme such as peroxidase or alkaline phosphatase to these antibodies, or by adding biotin or the like to these antibodies and binding to the aforementioned enzymes such as streptavidin. And a method of reacting a chromogenic substrate corresponding to those enzymes can be preferably used. Examples of a method for binding an enzyme to a secondary antibody or a tertiary antibody include a method using a reagent in which a large number of the enzyme and secondary antibody are bound to a dextrin polymer or an amino acid polymer (polymer method). In a method of reacting a biotinylated secondary antibody and peroxidase-labeled streptavidin (LSAB method), DAB or the like can be used as a chromogenic substrate. A secondary antibody labeled with a fluorescent dye or the like can also be used. When treated with a fluorescently labeled secondary antibody, positive cells are detected using a fluorescence microscope after the treatment.
スメア法では、摘出細胞をガラスに塗布または遠心分離機で分離し、細胞成分と液性成分に分け、細胞成分について免疫染色を行う。すなわち、細胞成分をスライドガラス上に塗布し、エタノール液または10%ホルマリン液などで固定した後、組織切片と同様の免疫染色を行うことができる。 In the smear method, the isolated cells are applied to glass or separated by a centrifugal separator, divided into cell components and liquid components, and immunostaining is performed on the cell components. That is, cell components can be applied on a slide glass and fixed with an ethanol solution or a 10% formalin solution, and then immunostaining similar to a tissue section can be performed.
凍結包埋法では、摘出組織をOCTコンパウンド等での包埋後に液体窒素等で急速凍結し、クリオスタットで薄切することでスライド標本を作製する。この標本を10%ホルマリンやエタノール液などで固定した後、組織切片と同様の免疫染色を行うことができる。 In the freezing embedding method, the excised tissue is rapidly frozen with liquid nitrogen after embedding with an OCT compound and sliced with a cryostat to prepare a slide specimen. After fixing this specimen with 10% formalin or ethanol solution, immunostaining similar to the tissue section can be performed.
免疫組織化学に係る操作は、反応液、反応条件、洗浄回数等をプログラムして免疫装置に組み込み、自動化して行なうことができる。 The operation related to immunohistochemistry can be performed automatically by programming the reaction solution, reaction conditions, number of washings, etc., and incorporating it into the immune device.
画像診断の場合は、薬学的に許容可能な放射性核種や発光体を抗体にラベル氏、被験者に該抗体を投与し、PET/CTなどの画像診断技術を使用して画像を取り、EPHA2の存在を判定または検査することができる。 In the case of diagnostic imaging, label the antibody with a pharmaceutically acceptable radionuclide or illuminant, administer the antibody to a subject, take an image using diagnostic imaging techniques such as PET / CT, and then the presence of EPHA2 Can be determined or inspected.
本発明の診断用組成物に含まれる抗体、その機能断片またはその修飾体は、好適にはEPHA2に結合又は認識する抗体、すなわちEPHA2選択性を有する抗体、その機能断片またはその修飾体である。 The antibody, functional fragment thereof or modified product thereof contained in the diagnostic composition of the present invention is preferably an antibody that binds to or recognizes EPHA2, that is, an antibody having EPHA2 selectivity, a functional fragment thereof, or a modified product thereof.
ヒトEPHA2選択性を有する抗体としては、マウスA201抗体の重鎖のCDRH1乃至CDRH3(配列番号60乃至62に示されるアミノ酸配列(図11))を含む重鎖、ならびに、軽鎖のCDRL1乃至CDRL3(配列番号63乃至65に示されるアミノ酸配列(図11))を含む軽鎖、を含む抗体、マウスA201抗体の重鎖可変領域(配列番号51のアミノ酸番号20乃至137に示されるアミノ酸配列)および軽鎖可変領域(配列番号54のアミノ酸番号20乃至128番目に示されるアミノ酸配列)を含む抗体、マウスA201抗体の重鎖および軽鎖を含む抗体等を例示することができる。かかる抗体としては、マウスA201抗体、キメラA201抗体等をあげることができるが、それらに限定されるものではない。
Examples of antibodies having human EPHA2 selectivity include heavy chain CDRH1 to CDRH3 of the mouse A201 antibody (amino acid sequence shown in SEQ ID NOs: 60 to 62 (FIG. 11)), and light chain CDRL1 to CDRRL3 ( An antibody comprising a light chain comprising the amino acid sequence shown in SEQ ID NO: 63 to 65 (FIG. 11), a heavy chain variable region of the mouse A201 antibody (amino acid sequence shown in
ヒトEPHA2選択性を有する抗体としては、マウスA205抗体の重鎖のCDRH1乃至CDRH3(配列番号66乃至68に示されるアミノ酸配列(図12))を含む重鎖、ならびに、軽鎖のCDRL1乃至CDRL3(配列番号69乃至71に示されるアミノ酸配列(図12))を含む軽鎖、を含む抗体、マウスA205抗体の重鎖可変領域(配列番号57のアミノ酸番号20乃至138番に示されるアミノ酸配列)および軽鎖可変領域(配列番号59のアミノ酸番号20乃至133番に示されるアミノ酸配列)を含む抗体、マウスA205抗体の重鎖および軽鎖を含む抗体等を例示することができる。かかる抗体としては、マウスA205抗体、キメラA205抗体等をあげることができるが、それらに限定されるものではない。
Antibodies having human EPHA2 selectivity include heavy chain CDRH1 to CDRH3 (amino acid sequence shown in SEQ ID NOs: 66 to 68 (FIG. 12)) of mouse A205 antibody, and light chain CDRL1 to CDRRL3 ( An antibody comprising a light chain comprising the amino acid sequence shown in SEQ ID NOs: 69 to 71 (FIG. 12), a heavy chain variable region of the mouse A205 antibody (amino acid sequence shown in
本発明の一つの好適な態様において、診断用組成物は、EPHA2検出用または測定用である。 In one preferred embodiment of the present invention, the diagnostic composition is for EPHA2 detection or measurement.
本発明は被検サンプル中のヒトEPHA2を検出または測定する方法を提供する。 The present invention provides a method for detecting or measuring human EPHA2 in a test sample.
これらの検出または測定方法には、本発明の診断用組成物を使用することができる。かかる測定方法および診断用組成物は、ヒトEPHA2陽性癌の診断用または検査用としても本発明に含まれる。 The diagnostic composition of the present invention can be used for these detection or measurement methods. Such measurement methods and diagnostic compositions are also included in the present invention for diagnosis or testing of human EPHA2-positive cancer.
本発明の医薬組成物が投与される個体を同定する方法も本発明に包含される。かかる同定方法においては、該個体由来サンプル中のヒトEPHA2を測定し、該サンプル中に、ヒトEPHA2が検出されたか、または、健常個体由来サンプル中に検出されたヒトEPHA2の量と比較してより多くのヒトEPHA2が検出された場合に、該個体を陽性と判定することができる。 A method for identifying an individual to which the pharmaceutical composition of the present invention is administered is also encompassed by the present invention. In such an identification method, human EPHA2 in the sample derived from the individual is measured, and human EPHA2 is detected in the sample, or compared with the amount of human EPHA2 detected in the sample derived from a healthy individual. If many human EPHA2s are detected, the individual can be determined as positive.
当該方法には、本発明の診断用組成物を使用することができる。 In the method, the diagnostic composition of the present invention can be used.
また、かかる同定方法の好適な一態様において、該個体は癌に罹患しているかまたはそのリスクがある。 Also, in a preferred embodiment of such an identification method, the individual is suffering from or at risk of having cancer.
さらに、本発明の医薬組成物は、その一態様において、かかる同定方法において陽性と判定された個体に投与され得る。 Furthermore, in one aspect, the pharmaceutical composition of the present invention can be administered to an individual who has been determined to be positive by such an identification method.
5.試薬
本発明の抗体もしくはその機能断片またはその修飾体は、試薬としても有用である。かかる試薬は、上述の検査または診断用、研究用およびその他の用途で使用される。
5. Reagent The antibody of the present invention or a functional fragment thereof or a modified form thereof is also useful as a reagent. Such reagents are used in the above-described examination or diagnostic, research and other applications.
以下、本発明を実施例により具体的に説明するが、本発明はこれらに限定されるものではない。なお、下記実施例において遺伝子操作に関する各操作は特に明示がない限り、「モレキュラークローニング(Molecular Cloning)」(Sambrook,J.,Fritsch,E.F.およびManiatis,T.著,Cold Spring Harbor Laboratory Pressより1989年に発刊)に記載の方法により行うか、または、市販の試薬やキットを用いる場合には市販品の指示書に従って使用した。 Hereinafter, the present invention will be specifically described by way of examples, but the present invention is not limited to these examples. In the following examples, unless otherwise specified, each operation relating to gene manipulation is “Molecular Cloning” (Sambrook, J., Fritsch, EF, and Maniatis, T., Cold Spring Harbor Laboratory Press. (Published in 1989) or when using commercially available reagents and kits, they were used according to the instructions for commercially available products.
(実施例1)マウス抗ヒトEPHA2抗体の作製
1)-1 免疫
免疫にはFcgr2b KO(BALB/c)Homoマウスの雌(免疫生物研究所社)を使用した。Recombinant Human EPHA2(R&D SYSTEM社;3035-A2)とFreund‘s Complete Adjuvant (和光純薬社) を混合したものを皮下に投与したマウスの脾臓を採取しハイブリドーマ作製に用いた。
Example 1 Production of Mouse Anti-Human EPHA2 Antibody 1) -1 Immunization Fcgr2b KO (BALB / c) Homo mouse female (Immuno-Biological Laboratories) was used for immunization. A mixture of Recombinant Human EPHA2 (R & D SYSTEM; 3035-A2) and Freund's Complete Adjuvant (Wako Pure Chemical Industries) was collected subcutaneously and used for hybridoma production.
1)-2 ハイブリドーマ作製
脾臓細胞とマウスミエローマSP2/0-Ag14細胞(ATCC:CRL-1581)とをPEG4000(免疫生物研究所社)またはHybrimune HybriProduction System(Cyto Pulse Sciences社)を用いて細胞融合し、ClonaCell-HY Selection Medium D(StemCell Technologies社)に希釈して培養した。出現したハイブリドーマコロニーを回収することでモノクローナルハイブリドーマを作製した。回収された各ハイブリドーマコロニーを培養し、得られたハイブリドーマ培養上清を用いて抗EPHA2抗体産生ハイブリドーマのスクリーニングを行った。
1) -2 Hybridoma preparation Spleen cells and mouse myeloma SP2 / 0-Ag14 cells (ATCC: CRL-1581) were cell-fused using PEG4000 (Immuno-Biological Laboratories) or Hybridene Hybrid Production System (Cyto Pulse Sciences). The cells were diluted with ClonaCell-HY Selection Medium D (StemCell Technologies) and cultured. Monoclonal hybridomas were prepared by collecting the emerged hybridoma colonies. Each collected hybridoma colony was cultured, and the resulting hybridoma culture supernatant was used to screen for anti-EPHA2 antibody-producing hybridomas.
(実施例2)マウス抗ヒトEPHA2抗体の抗原結合能の評価
2)-1 ヒトEPHA2及びヒトEPHA2に配列類似性のあるタンパク質を発現するベクターの構築
2)-1-1 発現ベクターの構築
pFLAG-myc-CMV19(Sigma社)の平滑末端化されたHindIIIとBglIIサイトにGateway Vector Conversion System(Life Technologies社、Thermo Fisher Scientific社)のReading Frame Bを結合した。このベクターを「pFLAG-GW」と名づけた。
(Example 2) Evaluation of antigen-binding ability of mouse anti-human EPHA2 antibody 2) -1 Construction of vector expressing human EPHA2 and protein having sequence similarity to human EPHA2 2) -1-1 Construction of expression vector pFLAG- Reading frame of Gateway Vector Conversion System (Life Technologies, Thermo Fisher Scientific) on blind-ended HindIII and BglII sites of myc-CMV19 (Sigma). This vector was named “pFLAG-GW”.
2)-1-2 EPHA2を発現するベクターの構築
シグナル配列のないヒトEPHA2をコードするDNA断片(配列番号1)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCAGCGCAGGGCAAGGAAG-3’(EPHA2-Fw:配列番号3)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTCAGATGGGGATCCCCAC-3’(EPHA2-Re:配列番号4)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221(Life Technologies社、Thermo Fisher Scientific社)にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHA2をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHA2と名づけた。なお、配列番号1のアミノ酸配列を配列番号2に記す。
2) -1-2 Construction of a vector expressing EPHA2 A DNA fragment (SEQ ID NO: 1) encoding human EPHA2 without a signal sequence was used as a primer set 5'-GGGGGACAAGTTTGTACAAAAAAAGCAGGCTCAGCGCAGGGGCCAAGGAAG-3 '(EPHA2-Fw: SEQ ID NO: 3)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTCAGATGGGGATCCCCCAC-3 ′ (EPHA2-Re: SEQ ID NO: 4)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 (Life Technologies, Thermo Fisher Scientific) by BP recombination reaction. The gene encoding EPHA2 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHA2. The amino acid sequence of SEQ ID NO: 1 is shown in SEQ ID NO: 2.
2)-1-3 EPHA3を発現するベクターの構築
シグナル配列のないヒトEPHA3をコードするDNA断片(配列番号5)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCAGAACTGATTCCGCAGCCT-3’(EPHA3-Fw:配列番号7)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTTACACGGGAACTGGGCC-3’(EPHA3-Re:配列番号8)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHA3をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHA3と名づけた。なお、配列番号5のアミノ酸配列を配列番号6に記す。
2) -1-3 Construction of a vector expressing EPHA3 A DNA fragment (SEQ ID NO: 5) encoding human EPHA3 without a signal sequence was used as a primer set 5′-GGGGGACAAGTTTGTACAAAAAAAGCAGGCTCAGAACTGATCTCCAGCACT-3 ′ (EPHA3-Fw: SEQ ID NO: 7)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTTACACGGGAACTGGGGCC-3 ′ (EPHA3-Re: SEQ ID NO: 8)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, the gene encoding EPHA3 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHA3. The amino acid sequence of SEQ ID NO: 5 is shown in SEQ ID NO: 6.
2)-1-4 EPHA4を発現するベクターの構築
シグナル配列のないヒトEPHA4をコードするDNA断片(配列番号9)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCAGTCACGGGTTCCAGGGTA-3’(EPHA4-Fw:配列番号11)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTCAGACGGGAACCATTCTG-3’(EPHA4-Re:配列番号12)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHA4をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHA4と名づけた。なお、配列番号9のアミノ酸配列を配列番号10に記す。
2) -1-4 Construction of a vector expressing EPHA4 A DNA fragment (SEQ ID NO: 9) encoding human EPHA4 without a signal sequence was used as a primer set 5′-GGGGGACAAGTTTGTACAAAAAAAGCAGGCTTCAGTCACGGGTTCGCGGTA-3 ′ (EPHA4-Fw: SEQ ID NO: 11)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTCAGACGGGAACCATTTCTG-3 ′ (EPHA4-Re: SEQ ID NO: 12)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, a gene encoding EPHA4 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHA4. The amino acid sequence of SEQ ID NO: 9 is shown in SEQ ID NO: 10.
2)-1-5 EPHA5を発現するベクターの構築
シグナル配列のないヒトEPHA5をコードするDNA断片(配列番号13)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTTCAGCCCCAGCAACGAAGTGAATTTATTGG-3’(EPHA5-Fw:配列番号15)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTTACAATGGCACCATTCCGTTTACCAG-3’(EPHA5-Re:配列番号16)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHA5をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHA5と名づけた。なお、配列番号13のアミノ酸配列を配列番号14に記す。
2) -1-5 Construction of a vector that expresses EPHA5 A DNA fragment (SEQ ID NO: 13) encoding human EPHA5 without a signal sequence was used as a primer set 5'-GGGGGACAAGTTTGTACAAAAAAAGCAGCCTTCAGCCCCAGCAACGAAGGTGAATTATTTGG-3 '(EPHA5-Fw: SEQ ID NO: 15)
5'-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTCATACAGGCACCATTCCGTTTACCAG-3 '(EPHA5-Re: SEQ ID NO: 16)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, a gene encoding EPHA5 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHA5. The amino acid sequence of SEQ ID NO: 13 is shown in SEQ ID NO: 14.
2)-1-6 EPHA6を発現するベクターの構築
ヒトEPHA6のDNA断片(配列番号17)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCATGGCCAGGCGACTGCAG-3’(EPHA6-Fw:配列番号19)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTCATACATGAAATCCCTTC-3’(EPHA6-Re:配列番号20)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。次にGeneArt(登録商標) Site-directed Mutagenesis System(Life Technologies社、Thermo Fisher Scientific社)とプライマーセット
5’-TCCAACAACCAAGTTGTGTTGCTTGATAC-3’(EPHA6-Mut-Fw:配列番号21)
5’-CAACACAACTTGGTTGTTGGATGAGCCTGCTTTTTTG-3’(EPHA6-Mut-Re:配列番号22)
を用いて、このベクターから配列番号17に示される核酸配列の1乃至24番目までの塩基が削除された。そして、このエントリーベクターからLR組換え反応によってEPHA6をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHA6と名づけた。なお、配列番号17のアミノ酸配列を配列番号18に記す。
2) -1-6 Construction of a vector expressing EPHA6 The DNA fragment of human EPHA6 (SEQ ID NO: 17) was used as a primer set 5′-GGGGGACAAGTTTGTACAAAAAAAGCAGCCTCATGGCCAGGCGAACTGCAG-3 ′ (EPHA6-Fw: SEQ ID NO: 19)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTCATACATAGAATCCCTTC-3 ′ (EPHA6-Re: SEQ ID NO: 20)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Next, GeneArt (registered trademark) Site-directed Mutagenesis System (Life Technologies, Thermo Fisher Scientific) and primer set 5′-TCCAACAACACAGTGTGTCTCTGATAC-3 ′ (No.)
5′-CAACACACTACTTGGTTGTTGGATGAGCCCTGCTTTTTG-3 ′ (EPHA6-Mut-Re: SEQ ID NO: 22)
The
2)-1-7 EPHA7を発現するベクターの構築
シグナル配列のないヒトEPHA7をコードするDNA断片(配列番号23)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCACAGGCTGCGAAGGAAGTAC-3’(EPHA7-Fw:配列番号25)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTCACACTTGAATGCCAGTTC-3’(EPHA7-Re:配列番号26)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHA7をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHA7と名づけた。なお、配列番号23のアミノ酸配列を配列番号24に記す。
2) -1-7 Construction of a vector expressing EPHA7 A DNA fragment (SEQ ID NO: 23) encoding human EPHA7 without a signal sequence was used as a primer set 5′-GGGGGACAAGTTTGTACAAAAAACAGCGCTCACAGGCTGGCGAAGGAAGTAC-3 ′ (EPHA7-Fw: SEQ ID NO: 25)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTCACCACTGAATGCCAGTTC-3 ′ (EPHA7-Re: SEQ ID NO: 26)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, the gene encoding EPHA7 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHA7. The amino acid sequence of SEQ ID NO: 23 is shown in SEQ ID NO: 24.
2)-1-8 EPHB1を発現するベクターの構築
シグナル配列のないヒトEPHB1をコードするDNA断片(配列番号27)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCAATGGAAGAAACGTTAATGGAC-3’(EPHB1-Fw:配列番号29)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTCATGCCATTGCCGTTGG-3’(EPHB1-Re:配列番号30)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHB1をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHB1と名づけた。なお、配列番号27のアミノ酸配列を配列番号28に記す。
2) -1-8 Construction of a vector expressing EPHB1 A DNA fragment (SEQ ID NO: 27) encoding human EPHB1 without a signal sequence was used as a primer set 5′-GGGGGACAAGTTTGTACAAAAAAAGCAGCCTCAATGGGAAGAAACGTTAATGGAC-3 ′ (EPHB1-Fw: SEQ ID NO: 29)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTCATGCCATGCCGTTGG-3 ′ (EPHB1-Re: SEQ ID NO: 30)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, the gene encoding EPHB1 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHB1. The amino acid sequence of SEQ ID NO: 27 is shown in SEQ ID NO: 28.
2)-1-9 EPHB3を発現するベクターの構築
シグナル配列のないヒトEPHB3をコードするDNA断片(配列番号31)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCACTGGAAGAGACCCTCATG-3’(EPHB3-Fw:配列番号33)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTCAGACCTGCACAGGCAG-3’(EPHB3-Re:配列番号34)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHB3をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHB3と名づけた。なお、配列番号31のアミノ酸配列を配列番号32に記す。
2) -1-9 Construction of a vector that expresses EPHB3 A DNA fragment (SEQ ID NO: 31) encoding human EPHB3 without a signal sequence was used as a primer set 5′-GGGGGACAAGTTTGTACAAAAAAAGGCGCTCACTGGGAAGAGACCCTCATG-3 ′ (EPHB3-Fw: SEQ ID NO: 33)
5′-GGGGACCACTTTGTTACAAGAAAGCTGGGTCTCAGACCTGCACAGGGCAG-3 ′ (EPHB3-Re: SEQ ID NO: 34)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, a gene encoding EPHB3 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHB3. The amino acid sequence of SEQ ID NO: 31 is shown in SEQ ID NO: 32.
2)-1-10 EPHB4を発現するベクターの構築
シグナル配列のないヒトEPHB4をコードするDNA断片(配列番号35)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTCATTGGAAGAGACCCTGCTG-3’(EPHB4-Fw:配列番号37)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTCAGTACTGCGGGGCCG-3’(EPHB4-Re:配列番号38)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってEPHB4をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-EPHB4と名づけた。なお、配列番号35のアミノ酸配列を配列番号36に記す。
2) -1-10 Construction of a vector expressing EPHB4 A DNA fragment (SEQ ID NO: 35) encoding human EPHB4 without a signal sequence was used as a primer set 5'-GGGGGACAAGTTTGTACAAAAAAAGCAGCCTCATTGGAAGAGACCCTGCTG-3 '(EPHB4-Fw: SEQ ID NO: 37)
5'-GGGGACCACTTTGTTACAAGAAAGCTGGGTCTCAGTACTGCGGGGCCG-3 '(EPHB4-Re: SEQ ID NO: 38)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, a gene encoding EPHB4 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-EPHB4. The amino acid sequence of SEQ ID NO: 35 is shown in SEQ ID NO: 36.
2)-1-11 PTPRS isoform 2を発現するベクターの構築
シグナル配列のないヒトPTPRS isoform 2 (以下、PTPRS-2と記す)をコードするDNA断片(配列番号39)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTTCGAAGAGCCCCCCAGGTTTATC-3’(PTPRS-2/4-Fw:配列番号41)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTTAGGTTGCATAGTGGTCAAAG-3’(PTPRS-2/4-Re:配列番号42)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってPTPRS-2をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-PTPRS-2と名づけた。なお、配列番号39のアミノ酸配列を配列番号40に記す。
2) -1-11 Construction of a vector that expresses PTPRS isoform 2 A DNA fragment (SEQ ID NO: 39) encoding human PTPRS isoform 2 (hereinafter referred to as PTPRS-2) without a signal sequence was used as a primer set 5'-GGGACAAGTTTGTACAAAAAAAGCAGCCTTCGAGT 3 ′ (PTPRS-2 / 4-Fw: SEQ ID NO: 41)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTTAGGTTGCATAGTGTCAAAG-3 ′ (PTPRS-2 / 4-Re: SEQ ID NO: 42)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, the gene encoding PTPRS-2 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-PTPRS-2. The amino acid sequence of SEQ ID NO: 39 is shown in SEQ ID NO: 40.
2)-1-12 PTPRS isoform 4を発現するベクターの構築
シグナル配列のないヒトPTPRS isoform 4 (以下、PTPRS-4と記す)をコードするDNA断片(配列番号43)を配列番号41及び42に示されたプライマーセットを使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってPTPRS-4をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-PTPRS-4と名づけた。なお、配列番号43のアミノ酸配列を配列番号44に記す。
2) -1-12 Construction of a vector expressing PPTRS isoform 4 A DNA fragment (SEQ ID NO: 43) encoding human PTPRS isoform 4 (hereinafter referred to as PTPRS-4) without a signal sequence is shown in SEQ ID NOs: 41 and 42. Amplified by PCR using the prepared primer set. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. Then, the gene encoding PTPRS-4 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-PTPRS-4. The amino acid sequence of SEQ ID NO: 43 is shown in SEQ ID NO: 44.
2)-1-13 PTPRF isoform 2を発現するベクターの構築
シグナル配列のないヒトPTPRF isoform 2 (以下、PTPRF-2と記す)をコードするDNA断片(配列番号45)をプライマーセット
5’-GGGGACAAGTTTGTACAAAAAAGCAGGCTTCGACAGCAAACCTGTCTTCATTAAAG-3’(PTPRF-2-Fw:配列番号47)
5’-GGGGACCACTTTGTACAAGAAAGCTGGGTCTTACGTTGCATAGTGGTCAAAGCTG-3’(PTPRF-2-Re:配列番号48)
を使用しPCRで増幅した。このPCR断片をBP組換え反応によりエントリーベクターpDONR221にトランスファーした。そして、このエントリーベクターからLR組換え反応によってPTPRF-2をコードする遺伝子をpFLAG-GWにトランスファーした。この結果できたベクターをpFLAG-PTPRF-2と名づけた。なお、配列番号45のアミノ酸配列を配列番号46に記す。
2) -1-13 Construction of a vector expressing PTPRF isoform 2 A DNA fragment (SEQ ID NO: 45) encoding human PTPRF isoform 2 (hereinafter referred to as PTPRF-2) without a signal sequence was used as a primer set 5′-GGGGGACAAGTTTGTACAAAAAATAGCAGCCTACCTGCTAGCAAACTCTGTCT 3 ′ (PTPRF-2-Fw: SEQ ID NO: 47)
5′-GGGGACCACTTTTGTACAAGAAAGCTGGGTCTTACGTTGCATAGTGGTCAAAGCTG-3 ′ (PTPRF-2-Re: SEQ ID NO: 48)
And was amplified by PCR. This PCR fragment was transferred to the entry vector pDONR221 by BP recombination reaction. The gene encoding PTPRF-2 was transferred from this entry vector to pFLAG-GW by LR recombination reaction. The resulting vector was named pFLAG-PTPRF-2. The amino acid sequence of SEQ ID NO: 45 is shown in SEQ ID NO: 46.
2)-2 IHCスクリーニング
2)-2-1 培養上清を用いたIHCスクリーニング
293α細胞(インテグリンαv及びインテグリンβ3を発現するHEK293由来の安定発現細胞株)に、Lipofectamine 2000(Life Technologies社、Thermo Fisher Scientific社)を用いて、pFLAG-EPHA2をトランスフェクションした(以下、EPHA2発現293α細胞)。EPHA2発現293α細胞のパラフィンブロックを作製するために、この細胞のペレットをホルマリンで固定し、パラフィンで包埋した。トランスフェクションしていない293α細胞および内在性EPHA2発現MDA-MB-231細胞(ATCC:No.HTB-26)についても同様にホルマリン固定後にパラフィンで包埋した。これらのブロックから切片を調製し、自動免疫染色装置Ventana Discovery ULTRAおよびDAB map kit(以上、Roche Diagnostics社)またはAutostainer Link48およびEnVisionFLEX Mini Kit(DAKO社)を用いて機器の使用説明書にしたがって、ハイブリドーマ由来の培養上清に含まれる抗体のIHCスクリーニングを実施した。まず、実施例1)-2において取得された67個のハイブリドーマ由来の培養上清の中から、EPHA2発現293α細胞に強く反応し、且つトランスフェクションしていない293α細胞に反応しない12個の抗体を選抜した。さらにMDA-MB-231細胞に反応する6個を選抜し、それらの中から内在性EPHA2に強く反応する3個を選抜した。
2) -2 IHC screening 2) -2-1 IHC screening using culture supernatants 293α cells (a stable expression cell line derived from HEK293 expressing integrin αv and integrin β3) were added to Lipofectamine 2000 (Life Technologies, Thermo Fisher). (Scientific) was used to transfect pFLAG-EPHA2 (hereinafter referred to as EPHA2-expressing 293α cells). To produce a paraffin block of EPHA2-expressing 293α cells, the cell pellet was fixed in formalin and embedded in paraffin. Untransfected 293α cells and endogenous EPHA2-expressing MDA-MB-231 cells (ATCC: No. HTB-26) were similarly embedded in paraffin after fixation with formalin. Sections are prepared from these blocks and using an automated immunostaining device Ventana Discovery ULTRA and DAB map kit (above, Roche Diagnostics) or Autostainer Link 48 and EnVisionFLEX Mini Kit (DAKO) using the instrument's instructions. IHC screening of the antibody contained in the culture supernatant derived from was performed. First, among the 67 hybridoma-derived culture supernatants obtained in Example 1) -2, 12 antibodies that react strongly with EPHA2-expressing 293α cells and do not react with non-transfected 293α cells were obtained. Selected. Further, 6 cells that react with MDA-MB-231 cells were selected, and 3 cells that strongly reacted with endogenous EPHA2 were selected.
2)-2-2 精製抗体を用いたIHCスクリーニング
実施例2)-2-1で選抜された3種の抗体を、これらの抗体を産生するハイブリドーマの培養上清より精製し、実施例2)-2-1と同様の方法でヒトの食道癌(CR2)、胃癌(CQ2)、大腸癌(CDA3)、子宮頸癌(CZA2)、肺癌(CCA4)、咽喉頭癌(CH3)、悪性黒色腫(LM481)、及び正常組織(MAN、MBN、MCN)の市販パラフィン切片(以上、SuperBioChips Laboratories社)を染色した。3個の中から、腫瘍細胞に強く反応し、バックグラウンド染色の低いA201およびA205を選抜した。
2) -2-2 IHC Screening Using Purified Antibodies Example 2) Three antibodies selected in 2-1 were purified from the culture supernatant of hybridomas producing these antibodies, Example 2) -2-1 In the same manner as human esophageal cancer (CR2), stomach cancer (CQ2), colon cancer (CDA3), cervical cancer (CZA2), lung cancer (CCA4), pharyngeal laryngeal cancer (CH3), malignant melanoma (LM481) and commercially available paraffin sections of normal tissues (MAN, MBN, MCN) (above, SuperBioChip Laboratories) were stained. Among the three, A201 and A205 that responded strongly to tumor cells and had low background staining were selected.
2)-3 抗体のアイソタイプ決定
マウス抗ヒトEPHA2抗体であるA201およびA205のアイソタイプは、Mouse monoclonal isotyping test kit(AbD Serotec社)により決定された。その結果、A201のアイソタイプはIgG1、A205のアイソタイプはIgG2bであることが示された。
2) -3 Isotype determination of antibodies The isotypes of A201 and A205, which are mouse anti-human EPHA2 antibodies, were determined by the Mouse monoclonal isotyping test kit (AbD Serotec). As a result, it was shown that the isotype of A201 is IgG1, and the isotype of A205 is IgG2b.
2)-4 フローサイトメトリー解析用抗原遺伝子発現細胞の調製
Free style 293F細胞(Life Technologies社、Thermo Fisher Scientific社。以下、293F細胞)を1×106細胞/mLに調製し、このうち、8mLをErlenmeyerFlask with Vent Cap(125mL)に播種した。pFLAG-EPHA2、ならびにEPHA2と配列類似性を持つ蛋白質をコードするpFLAG-EPHA3、pFLAG-EPHA5、pFLAG-EPHA6、pFLAG-EPHA7、pFLAG-EPHB1、pFLAG-EPHB3、pFLAG-EPHB4、pFLAG-PTPRS-2、pFLAG-PTPRS-4、およびpFLAG-PTPRF-2とコントロールとしてpFLAG-GWをそれぞれ293F細胞に293 fectin(Life Technologies社、Thermo Fisher Scientific社)を用いてトランスフェクションし、37℃、125rpm、8% CO2の条件下で一晩培養した。翌日、これら293F細胞を回収し、細胞を洗浄した後、この細胞をフローサイトメトリー解析に使用した。
2) -4 Preparation of antigen gene expressing cells for flow cytometry analysis Free style 293F cells (Life Technologies, Thermo Fisher Scientific, 293F cells) were prepared at 1 × 10 6 cells / mL, of which 8 mL Was seeded in Erlenmeyer Flask with Vent Cap (125 mL). pFLAG-EPHA2, and pFLAG-EPHA3, pFLAG-EPHA5, pFLAG-EPHA6, pFLAG-EPHA7, pFLAG-EPHB1, pFLAG-EPHAB3, pFLAG-EPHB4, pFLAG-PTPRS-2, which encode proteins having sequence similarity to EPHA2. pFLAG-PTPRS-4, pFLAG-PTPRF-2 and pFLAG-GW as controls were transfected into 293F cells using 293fectin (Life Technologies, Thermo Fisher Scientific), 37 ° C., 125 rpm, 8
2)-5 フローサイトメトリーによるマウス抗ヒトEPHA2抗体の結合能評価
A201とA205のEPHA2に対する結合特異性を評価するため、実施例2)-4で示す方法により調製したそれぞれの細胞に、A201、A205、マウスIgGコントロール抗体(Becton Dickinson社)、及びAnti-FLAG M2抗体(Sigma-Aldrich社)を加え、4℃で30分間インキュベートした。これらの細胞を5% FBS含有PBSで2回洗浄した後、5% FBS含有PBSで500倍に希釈したAnti-Mouse IgG FITC conjugate(MP Bio社)を加え、4℃で20分間インキュベートした。そして、これらの細胞を5% FBS含有PBSで2回洗浄した後、2μg/mLの7-Aminoactinomycin D(Life Technologies社、Thermo Fisher Scientific社)を含む5% FBS含有PBSに再懸濁し、フローサイトメーター(FACS CantoII、BD Biosciences社)で解析した。データ解析はFlowjoで行った。7-Aminoactinomycin D陽性の死細胞をゲートで除外した後、生細胞のFITC蛍光強度のヒストグラムを作成した。
2) -5 Evaluation of binding ability of mouse anti-human EPHA2 antibody by flow cytometry In order to evaluate the binding specificity of A201 and A205 to EPHA2, each cell prepared by the method shown in Example 2) -4 was treated with A201, A205, a mouse IgG control antibody (Becton Dickinson), and Anti-FLAG M2 antibody (Sigma-Aldrich) were added and incubated at 4 ° C. for 30 minutes. These cells were washed twice with PBS containing 5% FBS, Anti-Mouse IgG FITC conjugate (MP Bio) diluted 500 times with PBS containing 5% FBS was added, and incubated at 4 ° C. for 20 minutes. These cells were washed twice with PBS containing 5% FBS, then resuspended in PBS containing 5% FBS containing 2 μg / mL 7-Aminoactinomycin D (Life Technologies, Thermo Fisher Scientific), and flow site. Analysis was performed with a meter (FACS Canto II, BD Biosciences). Data analysis was performed with Flowjo. 7-Aminoactinomycin D-positive dead cells were excluded at the gate, and a histogram of FITC fluorescence intensity of live cells was prepared.
トランスフェクションした遺伝子が293F細胞で発現しているかを確認するため、FLAGタグへのAnti-FLAG M2抗体の結合を調べた。その結果、トランスフェクションされた全ての遺伝子(EPHA2,EPHA3、EPHA5、EPHA6、EPHA7、EPHB1、EPHB3、EPHB4、PTPRS-2、PTPRS-4、およびPTPRF-2)の発現が確認された(図1)。A201は、EPHA2に結合を示した。EPHB1にもわずかに結合するものの、それ以外のEPHA2と配列類似性を持つ分子(EPHA3、EPHA5、EPHA6、EPHA7、EPHB3、EPHB4、PTPRS-2、PTPRS-4、およびPTPRF-2)にはほどんど結合しないことが示された(図2)。A205は、EPHA2と結合する一方で、EPHA2と配列類似性を持つ分子(EPHA3、EPHA5、EPHA6、EPHA7、EPHB1、EPHB3、EPHB4、PTPRS-2、PTPRS-4、およびPTPRF-2)に対しては結合しないことが示された(図3)。なお、A205は陰性対照であるpFLAG-GWをトランスフェクションした細胞に対しても結合したが、これは内在的に293F細胞に発現するEPHA2に対してもA205が結合しているためと推測された。 In order to confirm whether the transfected gene was expressed in 293F cells, the binding of the Anti-FLAG M2 antibody to the FLAG tag was examined. As a result, the expression of all the transfected genes (EPHA2, EPHA3, EPHA5, EPHA6, EPHA7, EPHB1, EPHB3, EPHB4, PTPRS-2, PTPRS-4, and PTPRF-2) was confirmed (FIG. 1). . A201 showed binding to EPHA2. Although it binds slightly to EPHB1, other molecules with sequence similarities to EPHA2 (EPHA3, EPHA5, EPHA6, EPHA7, EPHB3, EPHB4, PTPRS-2, PTPRS-4, and PTPRF-2) are rare. It was shown not to bind (FIG. 2). A205 binds to EPHA2, while for molecules with sequence similarity to EPHA2 (EPHA3, EPHA5, EPHA6, EPHA7, EPHAB1, EPHAB3, EPHB4, PTPRS-2, PTPRS-4, and PTPRF-2) It was shown not to bind (Figure 3). A205 also bound to cells transfected with the negative control pFLAG-GW, which was presumed to be due to A205 binding to EPHA2 that is endogenously expressed in 293F cells. .
(実施例3)マウス抗ヒトEPHA2抗体をコードするcDNAのヌクレオチド配列の決定 (Example 3) Determination of nucleotide sequence of cDNA encoding mouse anti-human EPHA2 antibody
3)-1 マウス抗ヒトEPHA2抗体A201の重鎖及び軽鎖断片をコードするcDNAのヌクレオチド配列の決定 3) -1 Determination of nucleotide sequence of cDNA encoding heavy chain and light chain fragment of mouse anti-human EPHA2 antibody A201
3)-1-1 A201産生ハイブリドーマからのtotal RNAの調製
A201産生ハイブリドーマよりTRIzol Reagent(Ambion社)を用いてtotal RNAを調製した。
3) -1-1 Preparation of total RNA from A201-producing hybridoma Total RNA was prepared from A201-producing hybridoma using TRIzol Reagent (Ambion).
3)-1-2 cDNA(5’-RACE-Ready cDNA)の合成
実施例3)-1-1で調製したtotal RNAの1μgとSMARTer RACE cDNA Amplification Kit(Clontech社)を用いてcDNA(5’-RACE-Ready cDNA)を合成した。
3) -1-2 Synthesis of cDNA (5′-RACE-Ready cDNA) cDNA (5′-1 cDNA) using 1 μg of total RNA prepared in Example 3) -1-1 and SMARTER RACE cDNA Amplification Kit (Clontech) -RACE-Ready cDNA) was synthesized.
3)-1-3 5’-RACE PCRによるA201の重鎖断片をコードするcDNAの増幅とヌクレオチド配列の決定
A201の重鎖断片をコードするcDNAをPCRで増幅するためのプライマーとして、UPM(Universal Primer A Mix:SMARTer RACE cDNA Amplification Kitに付属)、及び
5’-CATCCCAGGGTCACCATGGAGTTAGTTTGG-3’(mG1VR1:配列番号49)を用いた。mG1VR1はデータベースのマウス重鎖(IgG1)の定常領域の配列から設計した。
3) -1-3 Amplification of cDNA encoding heavy chain fragment of A201 by 5′-RACE PCR and determination of nucleotide sequence As a primer for PCR amplification of cDNA encoding heavy chain fragment of A201, UPM (Universal Primer A Mix: attached to SMARTER RACE cDNA Amplification Kit) and 5′-CATCCCCAGGGTCACCATGGAGTTTAGTTGG-3 ′ (mG1VR1: SEQ ID NO: 49) were used. mG1VR1 was designed from the sequence of the constant region of mouse heavy chain (IgG1) in the database.
これらプライマーと、鋳型として実施例3)-1-2で合成したcDNA(5’-RACE-Ready cDNA)を用い、5’-RACE PCRによりA201の重鎖断片をコードするcDNAを増幅した。このPCRは、PolymeraseとしてKOD-Plus-(TOYOBO社)を用い、SMARTer RACE cDNA Amplification Kitのマニュアルに従い、タッチダウンPCRプログラムで実施した。 Using these primers and the cDNA synthesized in Example 3) -1-2 (5'-RACE-Ready cDNA) as a template, cDNA encoding the heavy chain fragment of A201 was amplified by 5'-RACE PCR. This PCR was performed using a touch-down PCR program according to the manual of SMARTER RACE cDNA Amplification Kit using KOD-Plus- (TOYOBO) as Polymerase.
5’-RACE PCRで増幅した重鎖断片をコードするcDNAをMinElute PCR Purification Kit(QIAGEN社)を用いて精製後、Zero Blunt TOPO PCR Cloning Kit(Invitrogen社)を用いてクローニングし、クローニングされたcDNAの配列決定を行った。 A cDNA encoding a heavy chain fragment amplified by 5'-RACE PCR is purified using MinElute PCR Purification Kit (QIAGEN), then cloned using Zero Blunt TOPO PCR Cloning Kit (Invitrogen), and cloned cDNA Was sequenced.
シークエンシングプライマーとして、
5’-CATCCCAGGGTCACCATGGAGTTAGTTTGG-3’(mG1VR1:配列番号49)、及びNUP(Nested Universal Primer A:SMARTer RACE cDNA Amplification Kitに付属)を用いた。
As a sequencing primer,
5'-CATCCCCAGGGTCACCCATGGAGTTTAGTTTG-3 '(mG1VR1: SEQ ID NO: 49) and NUP (Nested Universal Primer A: attached to SMARTER RACE cDNA Amplification Kit) were used.
シークエンス反応は、GeneAmp 9700(Applied Biosystems社)で行い、DNAの配列決定はABI PRISM 3700 DNA Analyzer(Applied Biosystems社)あるいはApplied Biosystems 3730xl Analyzer(Applied Biosystems社)で行った。 The sequencing reaction was performed with GeneAmp 9700 (Applied Biosystems), and the DNA sequencing was performed with ABI PRISM 3700 DNA Analyzer (Applied Biosystems) or Applied Biosystems 3730xxl Analyzer (Applied Biosystems).
決定されたA201の重鎖をコードするcDNAのヌクレオチド配列を配列番号50に示し、アミノ酸配列を配列番号51に示した。配列番号51の1乃至19番目のアミノ酸残基からなるアミノ酸配列はA201重鎖のシグナル配列に相当し、20乃至137番目のアミノ酸残基からなるアミノ酸配列はA201の重鎖可変領域に相当する。配列番号50および配列番号51は図7にも示されている。 The nucleotide sequence of the cDNA encoding the determined heavy chain of A201 is shown in SEQ ID NO: 50, and the amino acid sequence is shown in SEQ ID NO: 51. The amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 51 corresponds to the signal sequence of the A201 heavy chain, and the amino acid sequence consisting of the 20th to 137th amino acid residues corresponds to the heavy chain variable region of A201. SEQ ID NO: 50 and SEQ ID NO: 51 are also shown in FIG.
3)-1-4 5’-RACE PCRによるA201の軽鎖断片をコードするcDNAの増幅とヌクレオチド配列の決定
A201の軽鎖断片をコードするcDNAをPCRで増幅するためのプライマーとして、UPM(Universal Primer A Mix:SMARTer RACE cDNA Amplification Kitに付属)、及び
5’-AGTCCAACTGTTCAGGACGCCATTTTGTCG-3’(mKVR2:配列番号52)を用いた。なお、mKVR2はデータベースのマウス軽鎖の定常領域の配列から設計した。
3) -1-4 Amplification of cDNA encoding the light chain fragment of A201 by 5'-RACE PCR and determination of nucleotide sequence As a primer for PCR amplification of the cDNA encoding the light chain fragment of A201, UPM (Universal) Primer A Mix: attached to SMARTER RACE cDNA Amplification Kit) and 5′-AGTCCAACTGTTCAGGAGCCCATTTTGTCG-3 ′ (mKVR2: SEQ ID NO: 52) were used. In addition, mKVR2 was designed from the sequence of the constant region of the mouse light chain in the database.
これらのプライマーと、鋳型として実施例3)-1-2で合成したcDNA(5’-RACE-Ready cDNA)を用い、5’-RACE PCRによりA201の軽鎖断片をコードするcDNAを増幅した。このPCRは、PolymeraseとしてKOD-Plus-(TOYOBO社)を用い、SMARTer RACE cDNA Amplification Kitのマニュアルに従い、タッチダウンPCRプログラムで実施した。 Using these primers and the cDNA synthesized in Example 3) -1-2 as a template (5'-RACE-Ready cDNA), cDNA encoding the light chain fragment of A201 was amplified by 5'-RACE PCR. This PCR was performed using a touch-down PCR program according to the manual of SMARTER RACE cDNA Amplification Kit using KOD-Plus- (TOYOBO) as Polymerase.
5’-RACE PCRで増幅した軽鎖断片をコードするcDNAをMinElute PCR Purification Kit(QIAGEN社)を用いて精製後、Zero Blunt TOPO PCR Cloning Kit(Invitrogen社)を用いてクローニングし、クローニングされたcDNAの配列決定を行った。 A cDNA encoding a light chain fragment amplified by 5'-RACE PCR is purified using MinElute PCR Purification Kit (QIAGEN), then cloned using Zero Blunt TOPO PCR Cloning Kit (Invitrogen), and cloned cDNA Was sequenced.
シークエンシングプライマーとして、
5’-AGTCCAACTGTTCAGGACGCCATTTTGTCG-3’(mKVR2:配列番号52)、及びNUP(Nested Universal Primer A:SMARTer RACE cDNA Amplification Kitに付属)を用いた。
As a sequencing primer,
5'-AGTCCAACTGTTCAGGACGCCCATTTGTCG-3 '(mKVR2: SEQ ID NO: 52) and NUP (Nested Universal Primer A: attached to SMARTER RACE cDNA Amplification Kit) were used.
シークエンス反応は、GeneAmp 9700(Applied Biosystems社)で行い、DNAの配列決定はABI PRISM 3700 DNA Analyzer(Applied Biosystems社)あるいはApplied Biosystems 3730xl Analyzer(Applied Biosystems社)で行った。 The sequencing reaction was performed with GeneAmp 9700 (Applied Biosystems), and the DNA sequencing was performed with ABI PRISM 3700 DNA Analyzer (Applied Biosystems) or Applied Biosystems 3730xxl Analyzer (Applied Biosystems).
決定されたA201の軽鎖をコードするcDNAのヌクレオチド配列を配列表の配列番号53に示し、アミノ酸配列を配列番号54に示した。配列番号54の1乃至19番目のアミノ酸残基からなるアミノ酸配列はA201軽鎖のシグナル配列に相当し、20乃至128番目のアミノ酸残基からなるアミノ酸配列はA201の軽鎖可変領域に相当する。配列番号53および配列番号54は図8にも示されている。 The determined nucleotide sequence of cDNA encoding the light chain of A201 is shown in SEQ ID NO: 53 of the Sequence Listing, and the amino acid sequence is shown in SEQ ID NO: 54. The amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 54 corresponds to the signal sequence of the A201 light chain, and the amino acid sequence consisting of the 20th to 128th amino acid residues corresponds to the light chain variable region of A201. SEQ ID NO: 53 and SEQ ID NO: 54 are also shown in FIG.
3)-2 マウス抗ヒトEPHA2抗体A205の重鎖及び軽鎖断片をコードするcDNAのヌクレオチド配列の決定
3)-2-1 A205産生ハイブリドーマからのtotal RNAの調製
A205産生ハイブリドーマより実施例3)-1-1と同様の方法でtotal RNAを調製した。
3) -2 Determination of nucleotide sequence of cDNA encoding heavy and light chain fragments of mouse anti-human EPHA2 antibody A205 3) -2-1 Preparation of total RNA from A205 producing hybridoma Example 3) from A205 producing hybridoma Total RNA was prepared in the same manner as in 1-1.
3)-2-2 cDNA(5’-RACE-Ready cDNA)の合成
cDNA(5’-RACE-Ready cDNA)は、実施例3)-2-1で調製したtotal RNAの1μgを用い、実施例3)-1-2と同様の方法で合成した。
3) -2-2 Synthesis of cDNA (5′-RACE-Ready cDNA) The cDNA (5′-RACE-Ready cDNA) was prepared using 1 μg of the total RNA prepared in Example 3) -2-1. 3) Synthesis was performed in the same manner as in 1-2.
3)-2-3 5’-RACE PCRによるA205の重鎖断片をコードするcDNAの増幅とヌクレオチド配列の決定
実施例3)-2-2で合成したcDNA(5’-RACE-Ready cDNA)を鋳型として用いて、実施例3)-1-3と同様の方法でA205の重鎖断片をコードするcDNAを増幅して配列を決定した。
3) -2-3 Amplification of cDNA encoding heavy chain fragment of A205 by 5′-RACE PCR and determination of nucleotide sequence Example 3) cDNA synthesized in 2-2 (5′-RACE-Ready cDNA) Using the template, the cDNA encoding the heavy chain fragment of A205 was amplified and sequenced in the same manner as in Example 3) -1-3.
ただし、A205の重鎖断片のcDNAをPCRで増幅するためのプライマーとして、UPM(Universal Primer A Mix:SMARTer RACE cDNA Amplification Kitに付属)、及び
5’-TGCACACTGCTGGACAGGGATCCAGAGTTC-3’(mG2bVR2:配列番号55)を用いた。なお、mG2bVR2はデータベースのマウス重鎖(IgG2b)の定常領域の配列から設計した。
However, as primers for amplifying the cDNA of the heavy chain fragment of A205 by PCR, UPM (attached to Universal Primer A Mix: SMARTER RACE cDNA Amplification Kit), and 5′-TGCACCACTGCTGGACAGGGATCCAGAGTTC-3 ′ (mG2bVR) Was used. In addition, mG2bVR2 was designed from the sequence of the constant region of the mouse heavy chain (IgG2b) in the database.
シークエンシングプライマーとして、
5’-TGCACACTGCTGGACAGGGATCCAGAGTTC-3’(mG2bVR2:配列番号55)、及びNUP(Nested Universal Primer A:SMARTer RACE cDNA Amplification Kitに付属)を用いた。
As a sequencing primer,
5′-TGCACACTGCTGGACAGGGGATCCAGAGTTC-3 ′ (mG2bVR2: SEQ ID NO: 55) and NUP (Nested Universal Primer A: attached to SMARTER RACE cDNA Amplification Kit) were used.
決定されたA205の重鎖をコードするcDNAのヌクレオチド配列を配列表の配列番号56に示し、アミノ酸配列を配列番号57に示した。配列番号57の1乃至19番目のアミノ酸残基からなるアミノ酸配列はA205重鎖のシグナル配列に相当し、20乃至138番目のアミノ酸残基からなるアミノ酸配列はA205の重鎖可変領域に相当する。配列番号56および配列番号57は図9にも示されている。 The nucleotide sequence of the cDNA encoding the determined heavy chain of A205 is shown in SEQ ID NO: 56 in the sequence listing, and the amino acid sequence is shown in SEQ ID NO: 57. The amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 57 corresponds to the signal sequence of the A205 heavy chain, and the amino acid sequence consisting of the 20th to 138th amino acid residues corresponds to the heavy chain variable region of A205. SEQ ID NO: 56 and SEQ ID NO: 57 are also shown in FIG.
3)-2-4 5’-RACE PCRによるA205の軽鎖断片をコードするcDNAの増幅とヌクレオチド配列の決定
実施例3)-2-2で合成したcDNA(5’-RACE-Ready cDNA)を鋳型として用いて、実施例3)-1-4と同様の方法で抗体A205の軽鎖断片をコードするcDNAを増幅して配列を決定した。
3) -2-4 Amplification of cDNA encoding light chain fragment of A205 by 5'-RACE PCR and determination of nucleotide sequence Example 3) cDNA synthesized in 2-2 (5'-RACE-Ready cDNA) Using the template, the cDNA encoding the light chain fragment of antibody A205 was amplified and sequenced in the same manner as in Example 3) -1-4.
決定されたA205の軽鎖をコードするcDNAのヌクレオチド配列を配列表の配列番号58に示し、アミノ酸配列を配列番号59に示した。配列番号59の1乃至19番目のアミノ酸残基からなるアミノ酸配列はA205軽鎖のシグナル配列に相当し、20乃至133番目のアミノ酸残基からなるアミノ酸配列はA205の軽鎖可変領域に相当する。配列番号58および配列番号59は図10にも示されている。 The nucleotide sequence of the determined cDNA encoding the light chain of A205 is shown in SEQ ID NO: 58 in the sequence listing, and the amino acid sequence is shown in SEQ ID NO: 59. The amino acid sequence consisting of the 1st to 19th amino acid residues of SEQ ID NO: 59 corresponds to the signal sequence of the A205 light chain, and the amino acid sequence consisting of the 20th to 133rd amino acid residues corresponds to the light chain variable region of A205. SEQ ID NO: 58 and SEQ ID NO: 59 are also shown in FIG.
(実施例4)マウス抗ヒトEPHA2抗体の生産
マウス抗ヒトEPHA2抗体A201およびA205は、ハイブリドーマ培養上清から精製した。
(Example 4) Production of mouse anti-human EPHA2 antibody Mouse anti-human EPHA2 antibodies A201 and A205 were purified from the hybridoma culture supernatant.
まず、A201およびA205産生ハイブリドーマをClonaCell-HY Selection Medium Eで十分量まで増殖させた後、Ultra Low IgG FBS(Life Technologies社、Thermo Fisher Scientific社)を20%添加したHybridoma SFM(Life Technologies社、Thermo Fisher Scientific社)に培地交換し、5日間培養した。本培養上清を回収し0.45μmのフィルターを通して滅菌した。 First, A201 and A205-producing hybridomas were grown to a sufficient amount with ClonCell-HY Selection Medium E, and then Ultra Low IgG FBS (Life Technologies, ThermoFisher Scifi) was added to 20% of the world's TiMeMeSriMiMeFriSriMeMriFiSriMiSriFiSriMiSriMiSriMiSriFiSriMiSriFlM. The medium was changed to Fisher Scientific) and cultured for 5 days. The main culture supernatant was collected and sterilized through a 0.45 μm filter.
抗体は、上記のハイブリドーマ上清からProteinGアフィニティークロマトグラフィー(4~6℃下)1段階工程で精製した。ProteinGアフィニティークロマトグラフィー精製後のバッファー置換工程は4~6℃下で実施した。最初に、0.2M リン酸ナトリウムバッファー(pH7.0)で平衡化したProteinG(GE Healthcare Bioscience社)が充填されたカラムにハイブリドーマの培養上清をアプライした。培養上清液がカラムに全て入ったのち、カラム容量2倍以上のリン酸ナトリウムバッファーでカラムを洗浄した。次に0.1 Mグリシン/塩酸水溶液(pH2.7)で溶出し、抗体の含まれる画分を集めた。集めた画分に1M Tris-HCl(pH9.0)を加えてpH7.0~7.5に調製した後に、Amicon Ultra4(分画分子量UF50K、Merck Millipore社,4~6℃下)にて0.02% NaN3/PBSへのバッファー置換を行うとともに濃縮を行い、抗体濃度を0.3mg/mL以上に調製し、精製サンプルとした。 The antibody was purified from the above hybridoma supernatant by Protein G affinity chromatography (under 4-6 ° C.) in a one-step process. The buffer substitution step after protein G affinity chromatography purification was performed at 4 to 6 ° C. First, the culture supernatant of the hybridoma was applied to a column packed with Protein G (GE Healthcare Bioscience) equilibrated with 0.2 M sodium phosphate buffer (pH 7.0). After all of the culture supernatant liquid entered the column, the column was washed with a sodium phosphate buffer having a column volume of 2 times or more. Next, elution was performed with 0.1 M glycine / hydrochloric acid aqueous solution (pH 2.7), and fractions containing the antibody were collected. 1 M Tris-HCl (pH 9.0) was added to the collected fractions to adjust the pH to 7.0 to 7.5, and then 0% was obtained using Amicon Ultra 4 (fractionated molecular weight UF50K, Merck Millipore, 4 to 6 ° C.). 0.02% NaN 3 / PBS was substituted and concentrated, and the antibody concentration was adjusted to 0.3 mg / mL or more to prepare a purified sample.
(実施例5)マウス抗ヒトEPHA2抗体による免疫組織染色
図4に免疫組織化学染色におけるマウス抗EPHA2抗体の特異度を示す。図4に示すEphファミリー分子を293F細胞に一過性発現させ、それらの細胞のペレットをホルマリン固定後にパラフィンで包埋した。それらのブロックから調製した切片にTris-EDTA溶液(DAKO社)で97℃、40分間抗原賦活化処理を施し、内因性ペルオキシダーゼ(HRP)ブロッキング試薬(DAKO社)5分、非特異タンパク吸着ブロッキング試薬(DAKO社)30分の順に反応させた後、抗体希釈液(DAKO社)で2.5μg/mLの濃度に調製したマウス抗EPHA2抗体A201またはA205を1時間反応させた。HRP標識ユニバーサル二次抗体(DAKO社)、DAB発色基質(DAKO社)を用いて常法にて免疫組織化学染色を実施した。顕微鏡観察により、両抗体はEPHA2発現細胞に強く結合し、EPHA2を認識することが確認された。なお、A201はEPHA4発現細胞にもわずかに結合したが、強制発現細胞に対する極めて弱い交差反応であり、ヒト組織では問題にならない程度と判断された。
(Example 5) Immunohistochemical staining with mouse anti-human EPHA2 antibody FIG. 4 shows the specificity of the mouse anti-EPHA2 antibody in immunohistochemical staining. The Eph family molecules shown in FIG. 4 were transiently expressed in 293F cells, and the pellets of these cells were embedded in paraffin after formalin fixation. Sections prepared from these blocks were subjected to antigen activation treatment with Tris-EDTA solution (DAKO) at 97 ° C for 40 minutes, endogenous peroxidase (HRP) blocking reagent (DAKO) 5 minutes, nonspecific protein adsorption blocking reagent (DAKO) After reacting in order of 30 minutes, mouse anti-EPHA2 antibody A201 or A205 prepared to a concentration of 2.5 μg / mL with an antibody diluent (DAKO) was reacted for 1 hour. Immunohistochemical staining was performed by a conventional method using an HRP-labeled universal secondary antibody (DAKO) and DAB chromogenic substrate (DAKO). Microscopic observation confirmed that both antibodies strongly bound to EPHA2-expressing cells and recognized EPHA2. A201 was also slightly bound to EPHA4-expressing cells, but it was considered to be a level that was not a problem in human tissues because it was a very weak cross reaction with forced expression cells.
図5にマウス抗EPHA2抗体A201による免疫組織化学染色の結果を示す。市販のヒト咽喉頭癌および胃癌の組織マイクロアレイ(SuperBioChips Laboratories社)にTris-EDTA溶液(DAKO社)で97℃、40分間抗原賦活化処理を施し、内因性ペルオキシダーゼ(HRP)ブロッキング試薬(DAKO社)5分、非特異タンパク吸着ブロッキング試薬(DAKO社)30分の順に反応させた後、抗体希釈液(DAKO社)で5μg/mLの濃度に調製したマウス抗EPHA2抗体A201を1時間反応させた。HRP標識ユニバーサル二次抗体(DAKO社)、DAB発色基質(DAKO社)を用いて常法にて免疫組織化学染色を実施した。抗原賦活化処理以外の反応は室温にて実施した。図5は顕微鏡写真であり、マウス抗EPHA2抗体A201により、喉頭部扁平上皮癌および胃腺癌の腫瘍細胞の細胞膜および細胞質の一部に特異的な染色が認められる。腫瘍間質の組織および細胞には染色は認められない。腫瘍組織と間質組織は形態学的な相違により識別できる。 FIG. 5 shows the results of immunohistochemical staining with mouse anti-EPHA2 antibody A201. A commercially available human micropharyngeal cancer and gastric cancer tissue microarray (SuperBioChips Laboratories) was subjected to antigen activation treatment with a Tris-EDTA solution (DAKO) at 97 ° C. for 40 minutes, and an endogenous peroxidase (HRP) blocking reagent (DAKO) After reacting in the order of 5 minutes and non-specific protein adsorption blocking reagent (DAKO) for 30 minutes, mouse anti-EPHA2 antibody A201 prepared to a concentration of 5 μg / mL with an antibody diluent (DAKO) was reacted for 1 hour. Immunohistochemical staining was performed by a conventional method using an HRP-labeled universal secondary antibody (DAKO) and DAB chromogenic substrate (DAKO). Reactions other than the antigen activation treatment were performed at room temperature. FIG. 5 is a photomicrograph showing specific staining of part of the cell membrane and cytoplasm of tumor cells of laryngeal squamous cell carcinoma and gastric adenocarcinoma by mouse anti-EPHA2 antibody A201. There is no staining in the tissues and cells of the tumor stroma. Tumor tissue and stromal tissue can be distinguished by morphological differences.
図6にマウス抗EPHA2抗体A205による免疫組織化学染色の結果を示す。市販のヒト咽喉頭癌および胃癌の組織マイクロアレイ(SuperBioChips Laboratories社)にTris-EDTA溶液(DAKO社)で97℃、40分間抗原賦活化処理を施し、内因性HRPブロッキング試薬(DAKO社)5分、非特異タンパク吸着ブロッキング試薬(DAKO社)30分の順に反応させた後、抗体希釈液(DAKO社)で15μg/mLの濃度に調製したマウス抗EPHA2抗体A205を1時間反応させた。抗マウスリンカー抗体(DAKO社)、HRP標識ユニバーサル二次抗体(DAKO社)、DAB発色基質(DAKO社)を用いて常法にて免疫組織化学染色を実施した。抗原賦活化処理以外の反応は室温にて実施した。図6は顕微鏡写真であり、マウス抗EPHA2抗体A205により、喉頭部扁平上皮癌および胃腺癌の腫瘍細胞の細胞膜および細胞質の一部に特異的な染色が認められる。腫瘍間質の組織および細胞には染色は認められない。腫瘍組織と間質組織は形態学的な相違により識別できる。 FIG. 6 shows the results of immunohistochemical staining with mouse anti-EPHA2 antibody A205. A commercially available human micropharyngeal cancer and gastric cancer tissue microarray (SuperBioChips Laboratories) was subjected to an antigen activation treatment with a Tris-EDTA solution (DAKO) at 97 ° C. for 40 minutes, and an endogenous HRP blocking reagent (DAKO) 5 minutes. After reacting in order of non-specific protein adsorption blocking reagent (DAKO) for 30 minutes, mouse anti-EPHA2 antibody A205 prepared to a concentration of 15 μg / mL with an antibody diluent (DAKO) was reacted for 1 hour. Immunohistochemical staining was performed by an ordinary method using an anti-mouse linker antibody (DAKO), an HRP-labeled universal secondary antibody (DAKO), and a DAB chromogenic substrate (DAKO). Reactions other than the antigen activation treatment were performed at room temperature. FIG. 6 is a photomicrograph showing specific staining of the cell membrane and part of the cytoplasm of tumor cells of laryngeal squamous cell carcinoma and gastric adenocarcinoma by the mouse anti-EPHA2 antibody A205. There is no staining in the tissues and cells of the tumor stroma. Tumor tissue and stromal tissue can be distinguished by morphological differences.
マウス抗EPHA2抗体A201とA205によって同じ部位が染色されているが、A205よりもA201のほうが感度が高く、より多くの腫瘍細胞が染色されている。 The same site is stained with mouse anti-EPHA2 antibodies A201 and A205, but A201 is more sensitive than A205, and more tumor cells are stained.
(実施例6)マウス抗ヒトEPHA2抗体による免疫組織染色
図13および14に免疫組織化学染色における抗EPHA2抗体の染色条件および染色性を示す。EPHA2非発現293α細胞、EPHA2過剰発現293α細胞、およびEPHA2内因性発現MDA-MB-231細胞のペレットをホルマリン固定後にパラフィンで包埋した。それらのブロックから調製した切片にTris-EDTA溶液(DAKO社乃至はRoche Diagnostics社)で97℃、36分間乃至40分間抗原賦活化処理を施し、常法にて内因性ペルオキシダーゼ(HRP)ブロッキング、非特異タンパク吸着ブロッキングを行った後、予備検討により最適化した濃度(図13)に調製した各抗EPHA2抗体を1時間反応させた。検出にはRoche Diagnostics社乃至はDAKO社のキットを用い、LSAB法乃至はポリマー法で免疫組織化学染色を実施した。顕微鏡観察により、細胞膜または細胞質に明確な陽性反応が認められる場合をプラス、弱い陽性反応が認められる場合はプラスマイナスとした。A201およびA205の両抗体は、EPHA2過剰発現293α細胞およびEPHA2内因性発現MDA-MB-231細胞の両方に強い陽性反応を示し、比較した市販の各種EPHA2抗体よりも高い感度でEPHA2を認識することが確認された(図13)。なお、検討した市販の各種EPHA2抗体の中では、R&D Systems社のヤギ抗EPHA2ポリクローナル抗体(AF3035)が最も感度および特異性に優れると判断された。図14は細胞の顕微鏡写真であり、全ての抗体でEPHA2非発現293α細胞には陽性反応は認められない一方、EPHA2過剰発現293α細胞では細胞膜および細胞質に強い陽性反応が認められる。MDA-MB-231細胞については、マウス抗EPHA2抗体A201およびA205、ならびにR&D Systems社ヤギ抗EPHA2ポリクローナル抗体(AF3035)により細胞膜に強い陽性反応、Santa Cruz社ウサギ抗EPHA2ポリクローナル抗体(sc-924)およびCell Signaling Technology社ウサギ抗EPHA2モノクローナル抗体(6997)により細胞膜に弱い陽性反応が認められる。なお、Millipore社のマウス抗EPHA2抗体(D4)では、細胞全体に高い非特異染色が認められた。
(Example 6) Immunohistochemical staining with mouse anti-human EPHA2 antibody FIGS. 13 and 14 show the staining conditions and staining properties of the anti-EPHA2 antibody in immunohistochemical staining. EPHA2 non-expressing 293α cells, EPHA2 overexpressing 293α cells, and EPHA2 endogenously expressed MDA-MB-231 cell pellets were embedded in paraffin after formalin fixation. Sections prepared from these blocks were subjected to antigen activation treatment with Tris-EDTA solution (DAKO or Roche Diagnostics) at 97 ° C. for 36 to 40 minutes, and endogenous peroxidase (HRP) blocking, non-blocking was performed by a conventional method. After carrying out specific protein adsorption blocking, each anti-EPHA2 antibody prepared to the density | concentration optimized by preliminary examination (FIG. 13) was made to react for 1 hour. For detection, immunohistochemical staining was carried out by the LSAB method or the polymer method using a kit of Roche Diagnostics or DAKO. By microscopic observation, a positive positive reaction was observed in the cell membrane or cytoplasm, and a positive positive reaction was observed when a weak positive reaction was observed. Both A201 and A205 antibodies show strong positive reaction to both EPHA2 overexpressing 293α cells and EPHA2 endogenously expressed MDA-MB-231 cells, and recognize EPHA2 with higher sensitivity than various commercially available EPHA2 antibodies compared. Was confirmed (FIG. 13). Of the various commercially available EPHA2 antibodies examined, the goat anti-EPHA2 polyclonal antibody (AF3035) from R & D Systems was judged to have the highest sensitivity and specificity. FIG. 14 is a photomicrograph of cells. In all antibodies, no positive reaction is observed in EPHA2 non-expressing 293α cells, whereas in EPHA2 overexpressing 293α cells, a strong positive reaction is observed in the cell membrane and cytoplasm. For MDA-MB-231 cells, the mouse anti-EPHA2 antibodies A201 and A205, and R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035) strongly reacted with the cell membrane, Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924) and A weak positive reaction is observed in the cell membrane by the Cell Signaling Technology rabbit anti-EPHA2 monoclonal antibody (6997). In addition, in the mouse anti-EPHA2 antibody (D4) manufactured by Millipore, high non-specific staining was observed throughout the cells.
図15~18にマウス抗EPHA2抗体A201およびA205、R&D Systems社ヤギ抗EPHA2ポリクローナル抗体(AF3035)、ならびにSanta Cruz社ウサギ抗EPHA2ポリクローナル抗体(sc-924)による免疫組織化学染色の結果を示す。市販のヒト腫瘍組織マイクロアレイ(SuperBioChips Laboratories社)にTris-EDTA溶液(DAKO社)で97℃、40分間抗原賦活化処理を施し、前述と同様の方法でブロッキングを行った後、抗体希釈液(DAKO社)で希釈した各抗EPHA2抗体を1時間反応させた。HRP標識ユニバーサル二次抗体(DAKO社)乃至はHRP標識抗ヤギIgG抗体(ニチレイバイオサイエンス社)、DAB発色基質(DAKO社)を用いて常法にて免疫組織化学染色を実施した。A205抗体についてはHRP標識ユニバーサル二次抗体の反応前に抗マウスリンカー抗体(DAKO社)を反応させた。図15~18は腫瘍組織の顕微鏡写真であり、ヒトの食道癌(図15)、子宮頸癌(図16)、大腸癌(図17)、胃癌(図18)の染色像である。いずれの癌においても、A201抗体およびA205抗体によって、腫瘍細胞の細胞膜および細胞質の一部に特異的な染色が認められ、A205よりもA201のほうが感度が高く、より多くの腫瘍細胞が染色されている。AF3035抗体およびsc-924抗体では特異的な染色は認められないか、ごくわずかである。A201抗体、A205抗体、およびAF3035抗体では腫瘍間質の組織および細胞には染色は認められないが、sc-924抗体では腫瘍間質の非腫瘍細胞にも交差反応が認められる。腫瘍組織と間質組織は形態学的な相違により識別できる。 15 to 18 show the results of immunohistochemical staining with mouse anti-EPHA2 antibodies A201 and A205, R & D Systems goat anti-EPHA2 polyclonal antibody (AF3035), and Santa Cruz rabbit anti-EPHA2 polyclonal antibody (sc-924). A commercially available human tumor tissue microarray (SuperBioChips Laboratories) was subjected to antigen activation treatment with a Tris-EDTA solution (DAKO) at 97 ° C. for 40 minutes, blocked in the same manner as described above, and then diluted with an antibody diluent (DAKO). Each anti-EPHA2 antibody diluted in 1) was reacted for 1 hour. Immunohistochemical staining was performed by a conventional method using an HRP-labeled universal secondary antibody (DAKO) or an HRP-labeled anti-goat IgG antibody (Nichirei Bioscience) or a DAB chromogenic substrate (DAKO). For the A205 antibody, an anti-mouse linker antibody (DAKO) was reacted before the reaction of the HRP-labeled universal secondary antibody. 15 to 18 are micrographs of tumor tissues, which are stained images of human esophageal cancer (FIG. 15), cervical cancer (FIG. 16), colon cancer (FIG. 17), and stomach cancer (FIG. 18). In any cancer, A201 antibody and A205 antibody showed specific staining in the cell membrane and part of cytoplasm of tumor cells, and A201 was more sensitive than A205, and more tumor cells were stained. Yes. There is little or no specific staining with the AF3035 and sc-924 antibodies. The A201 antibody, A205 antibody, and AF3035 antibody show no staining in the tissues and cells of the tumor stroma, but the sc-924 antibody also shows a cross reaction in the non-tumor cells of the tumor stroma. Tumor tissue and stromal tissue can be distinguished by morphological differences.
本発明の提供する抗体を用いることにより各種癌の検査または診断が可能となる。 The use of the antibody provided by the present invention enables examination or diagnosis of various cancers.
配列番号1:ヒトEPHA2をコードするヌクレオチド配列
配列番号2:ヒトEPHA2のアミノ酸配列
配列番号3:PCRプライマー EPHA2-Fw
配列番号4:PCRプライマー EPHA2-Re
配列番号5:ヒトEPHA3をコードするヌクレオチド配列
配列番号6:ヒトEPHA3のアミノ酸配列
配列番号7:PCRプライマー EPHA3-Fw
配列番号8:PCRプライマー EPHA3-Re
配列番号9:ヒトEPHA4をコードするヌクレオチド配列
配列番号10:ヒトEPHA4のアミノ酸配列
配列番号11:PCRプライマー EPHA4-Fw
配列番号12:PCRプライマー EPHA4-Re
配列番号13:ヒトEPHA5をコードするヌクレオチド配列
配列番号14:ヒトEPHA5のアミノ酸配列
配列番号15:PCRプライマー EPHA5-Fw
配列番号16:PCRプライマー EPHA5-Re
配列番号17:ヒトEPHA6をコードするヌクレオチド配列
配列番号18:ヒトEPHA6のアミノ酸配列
配列番号19:PCRプライマー EPHA6-Fw
配列番号20:PCRプライマー EPHA6-Re
配列番号21:プライマー EPHA6-Mut-Fw
配列番号22:プライマー EPHA6-Mut-Re
配列番号23:ヒトEPHA7をコードするヌクレオチド配列
配列番号24:ヒトEPHA7のアミノ酸配列
配列番号25:PCRプライマー EPHA7-Fw
配列番号26:PCRプライマー EPHA7-Re
配列番号27:ヒトEPHB1をコードするヌクレオチド配列
配列番号28:ヒトEPHB1のアミノ酸配列
配列番号29:PCRプライマー EPHB1-Fw
配列番号30:PCRプライマー EPHB1-Re
配列番号31:ヒトEPHB3をコードするヌクレオチド配列
配列番号32:ヒトEPHB3のアミノ酸配列
配列番号33:PCRプライマー EPHB3-Fw
配列番号34:PCRプライマー EPHB3-Re
配列番号35:ヒトEPHB4をコードするヌクレオチド配列
配列番号36:ヒトEPHB4のアミノ酸配列
配列番号37:PCRプライマー EPHB4-Fw
配列番号38:PCRプライマー EPHB4-Re
配列番号39:ヒトPTPRS-2をコードするヌクレオチド配列
配列番号40:ヒトPTPRS-2のアミノ酸配列
配列番号41:PCRプライマー PTPRS-2/4-Fw
配列番号42:PCRプライマー PTPRS-2/4-Re
配列番号43:ヒトPTPRS-4をコードするヌクレオチド配列
配列番号44:ヒトPTPRS-4のアミノ酸配列
配列番号45:ヒトPTPRF-2をコードするヌクレオチド配列
配列番号46:ヒトPTPRF-2のアミノ酸配列
配列番号47:PCRプライマー PTPRF-2-Fw
配列番号48:PCRプライマー PTPRF-2-Re
配列番号49:PCRプライマー mG1VR1(A201重鎖可変領域用)
配列番号50:A201抗体重鎖をコードするヌクレオチド配列
配列番号51:A201抗体重鎖のアミノ酸配列
配列番号52:PCRプライマー mKVR2(A201、A205軽鎖可変領域用)
配列番号53:A201抗体軽鎖をコードするヌクレオチド配列
配列番号54:A201抗体軽鎖のアミノ酸配列
配列番号55:PCRプライマー mG2bVR2(A205重鎖可変領域用)
配列番号56:A205抗体重鎖をコードするヌクレオチド配列
配列番号57:A205抗体重鎖のアミノ酸配列
配列番号58:A205抗体軽鎖をコードするヌクレオチド配列
配列番号59:A205抗体軽鎖のアミノ酸配列
配列番号60:A201抗体のCDRH1のアミノ酸配列
配列番号61:A201抗体のCDRH2のアミノ酸配列
配列番号62:A201抗体のCDRH3のアミノ酸配列
配列番号63:A201抗体のCDRL1のアミノ酸配列
配列番号64:A201抗体のCDRL2のアミノ酸配列
配列番号65:A201抗体のCDRL3のアミノ酸配列
配列番号66:A205抗体のCDRH1のアミノ酸配列
配列番号67:A205抗体のCDRH2のアミノ酸配列
配列番号68:A205抗体のCDRH3のアミノ酸配列
配列番号69:A205抗体のCDRL1のアミノ酸配列
配列番号70:A205抗体のCDRL2のアミノ酸配列
配列番号71:A205抗体のCDRL3のアミノ酸配列
SEQ ID NO: 1: Nucleotide sequence encoding human EPHA2 SEQ ID NO: 2: Amino acid sequence of human EPHA2 SEQ ID NO: 3: PCR primer EPHA2-Fw
SEQ ID NO: 4: PCR primer EPHA2-Re
SEQ ID NO: 5: nucleotide sequence encoding human EPHA3 SEQ ID NO: 6: amino acid sequence of human EPHA3 SEQ ID NO: 7: PCR primer EPHA3-Fw
SEQ ID NO: 8: PCR primer EPHA3-Re
SEQ ID NO: 9: nucleotide sequence encoding human EPHA4 SEQ ID NO: 10: amino acid sequence of human EPHA4 SEQ ID NO: 11: PCR primer EPHA4-Fw
SEQ ID NO: 12: PCR primer EPHA4-Re
SEQ ID NO: 13: nucleotide sequence encoding human EPHA5 SEQ ID NO: 14: amino acid sequence of human EPHA5 SEQ ID NO: 15: PCR primer EPHA5-Fw
SEQ ID NO: 16: PCR primer EPHA5-Re
SEQ ID NO: 17: nucleotide sequence encoding human EPHA6 SEQ ID NO: 18: amino acid sequence of human EPHA6 SEQ ID NO: 19: PCR primer EPHA6-Fw
SEQ ID NO: 20: PCR primer EPHA6-Re
SEQ ID NO: 21: primer EPHA6-Mut-Fw
SEQ ID NO: 22: Primer EPHA6-Mut-Re
SEQ ID NO: 23: nucleotide sequence encoding human EPHA7 SEQ ID NO: 24: amino acid sequence of human EPHA7 SEQ ID NO: 25: PCR primer EPHA7-Fw
SEQ ID NO: 26: PCR primer EPHA7-Re
SEQ ID NO: 27: nucleotide sequence encoding human EPHB1 SEQ ID NO: 28: amino acid sequence of human EPHB1 SEQ ID NO: 29: PCR primer EPHB1-Fw
SEQ ID NO: 30: PCR primer EPHB1-Re
SEQ ID NO: 31: nucleotide sequence encoding human EPHB3 SEQ ID NO: 32: amino acid sequence of human EPHB3 SEQ ID NO: 33: PCR primer EPHB3-Fw
SEQ ID NO: 34: PCR primer EPHB3-Re
SEQ ID NO: 35: nucleotide sequence encoding human EPHB4 SEQ ID NO: 36: amino acid sequence of human EPHB4 SEQ ID NO: 37: PCR primer EPHB4-Fw
SEQ ID NO: 38: PCR primer EPHB4-Re
SEQ ID NO: 39: nucleotide sequence encoding human PTPRS-2 SEQ ID NO: 40: amino acid sequence of human PTPRS-2 SEQ ID NO: 41: PCR primer PTPRS-2 / 4-Fw
SEQ ID NO: 42: PCR primer PTPRS-2 / 4-Re
SEQ ID NO: 43: nucleotide sequence encoding human PTPRS-4 SEQ ID NO: 44: amino acid sequence of human PTPRS-4 SEQ ID NO: 45: nucleotide sequence encoding human PTPRF-2 SEQ ID NO: 46: amino acid sequence of human PTPRF-2 47: PCR primer PTPRF-2-Fw
SEQ ID NO: 48: PCR primer PTPRF-2-Re
SEQ ID NO: 49: PCR primer mG1VR1 (for A201 heavy chain variable region)
SEQ ID NO: 50: nucleotide sequence encoding A201 antibody heavy chain SEQ ID NO: 51: amino acid sequence of A201 antibody heavy chain SEQ ID NO: 52: PCR primer mKVR2 (for A201, A205 light chain variable region)
SEQ ID NO: 53: nucleotide sequence encoding A201 antibody light chain SEQ ID NO: 54: amino acid sequence of A201 antibody light chain SEQ ID NO: 55: PCR primer mG2bVR2 (for A205 heavy chain variable region)
SEQ ID NO: 56: nucleotide sequence encoding A205 antibody heavy chain SEQ ID NO: 57: amino acid sequence of A205 antibody heavy chain SEQ ID NO: 58: nucleotide sequence encoding A205 antibody light chain SEQ ID NO: 59: amino acid sequence sequence number of A205 antibody light chain 60: A201 antibody CDRH1 amino acid sequence SEQ ID NO: 61: A201 antibody CDRH2 amino acid sequence SEQ ID NO: 62: A201 antibody CDRH3 amino acid sequence SEQ ID NO: 63: A201 antibody CDRRL1 amino acid sequence SEQ ID NO: 64: A201 antibody CDRRL2 Amino acid sequence of SEQ ID NO: 65: amino acid sequence of CDR1 of antibody A201 SEQ ID NO: 66: amino acid sequence of CDRH1 of antibody A205 SEQ ID NO: 67: amino acid sequence of CDRH2 of antibody A205 SEQ ID NO: 68: amino acid of CDRH3 of antibody A205 SEQ SEQ ID NO 69: A 205 antibodies of CDRL1 amino acid sequence SEQ ID NO 70: A 205 antibodies of CDRL2 amino acid sequence SEQ ID NO 71: A 205 CDRL3 amino acid sequence of the antibody
Claims (28)
軽鎖配列が、CDRL1、CDRL2、CDRL3を有する可変領域を含み、前記CDRL1は配列番号63に示されるアミノ酸配列からなり、前記CDRL2は配列番号64に示されるアミノ酸配列からなり、前記CDRL3は配列番号65に示されるアミノ酸配列からなること;および
配列番号2に示されるEPHA2に特異的に結合すること;
を特徴とする抗体または該抗体の抗原結合性断片。 The heavy chain sequence includes a variable region having CDRH1, CDRH2, CDRH3, the CDRH1 is composed of the amino acid sequence represented by SEQ ID NO: 60, the CDRH2 is composed of the amino acid sequence represented by SEQ ID NO: 61, and the CDRH3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 62;
The light chain sequence includes a variable region having CDRL1, CDRL2, and CDRL3, wherein the CDRL1 is composed of an amino acid sequence represented by SEQ ID NO: 63, the CDRL2 is composed of an amino acid sequence represented by SEQ ID NO: 64, and the CDRL3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 65; and specifically binding to EPHA2 shown in SEQ ID NO: 2;
Or an antigen-binding fragment of the antibody.
軽鎖配列が、CDRL1、CDRL2、CDRL3を有する可変領域を含み、前記CDRL1は配列番号69に示されるアミノ酸配列からなり、前記CDRL2は配列番号70に示されるアミノ酸配列からなり、前記CDRL3は配列番号71に示されるアミノ酸配列からなること;および
配列番号2に示されるEPHA2に特異的に結合すること;
を特徴とする抗体または該抗体の抗原結合性断片。 The heavy chain sequence includes a variable region having CDRH1, CDRH2, CDRH3, the CDRH1 is composed of an amino acid sequence represented by SEQ ID NO: 66, the CDRH2 is composed of an amino acid sequence represented by SEQ ID NO: 67, and the CDRH3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 68;
The light chain sequence includes a variable region having CDRL1, CDRL2, and CDRL3. The CDRL1 is composed of an amino acid sequence represented by SEQ ID NO: 69, the CDRL2 is composed of an amino acid sequence represented by SEQ ID NO: 70, and the CDRL3 is represented by SEQ ID NO: Consisting of the amino acid sequence shown in 71; and specifically binding to EPHA2 shown in SEQ ID NO: 2;
Or an antigen-binding fragment of the antibody.
(i)請求項9乃至11または13のいずれか一つに記載の組成物を用いてEPHA2が検出または測定された被検標本の由来する被験者;
(ii)請求項12に記載の組成物を用いたEPHA2の検出または測定において陽性と判定された被検標本の由来する被験者;
(iii)請求項14または15に記載の組成物を用いて、EPHA2に特異的に結合する抗体もしくは該抗体の抗原結合性断片を投与する工程を含むEPHA2陽性疾患の治療または予防に適していると判定された被験者。 A pharmaceutical composition comprising an antibody that specifically binds to EPHA2 or an antigen-binding fragment of the antibody, which is administered to the subject according to any one of (i) to (iii) below:
(I) a subject derived from a test specimen in which EPHA2 is detected or measured using the composition according to any one of claims 9 to 11 or 13;
(Ii) a subject derived from a test specimen determined to be positive in the detection or measurement of EPHA2 using the composition according to claim 12;
(Iii) The composition according to claim 14 or 15 is suitable for treating or preventing an EPHA2-positive disease comprising a step of administering an antibody that specifically binds to EPHA2 or an antigen-binding fragment of the antibody. Subject determined to be.
(i)請求項19に記載の細胞を培養する工程;
(ii)工程(i)の培養物からモノクローナル抗体または該抗体の抗原結合性断片を回収する工程。 The method for producing a monoclonal antibody or an antigen-binding fragment of the antibody according to any one of claims 1 to 8, comprising the following steps (i) and (ii):
(I) a step of culturing the cell according to claim 19;
(Ii) A step of recovering a monoclonal antibody or an antigen-binding fragment of the antibody from the culture of step (i).
(i)請求項1乃至8のいずれか一つに記載の抗体または該抗体の抗原結合性断片又は請求項9に記載の組成物と被検標本を接触させる工程;
(ii)該被検標本においてEPHA2が検出もしくは測定されたか、または該被検標本におけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより高い場合、該被検標本を陽性と判定し、被検標本においてEPHA2が検出もしくは測定されなかったか、また該被検標本におけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより低い場合、該被検標本を陰性と判定する工程。 EPHA2 detection or measurement method comprising the following steps (i) or (i) and (ii):
(I) contacting the test specimen with the antibody according to any one of claims 1 to 8 or the antigen-binding fragment of the antibody or the composition according to claim 9;
(Ii) if EPHA2 is detected or measured in the test specimen, or if the expression level or expression level of EPHA2 in the test specimen is equal to or higher than a predetermined criterion, If it is judged positive and EPHA2 is not detected or measured in the test specimen, and the expression level or expression level of EPHA2 in the test specimen is equal to or lower than a predetermined criterion, the test specimen The step of judging a specimen as negative.
(i)請求項1乃至8のいずれか一つに記載の抗体または該抗体の抗原結合性断片又は請求項9に記載の組成物と個体由来のサンプルを接触させる工程;
(ii)該個体由来のサンプルにおいてEPHA2が検出もしくは測定されたか、または該個体由来のサンプルにおけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより高い場合、該個体を陽性と判定し、該個体由来のサンプルにおいてEPHA2が検出もしくは測定されなかったか、また該個体由来のサンプルにおけるEPHA2の発現レベルもしくは発現量が事前に決定された基準と同等かまたはそれより低い場合、該個体を陰性と判定する工程。 A method for identifying an individual to be administered a pharmaceutical composition comprising an antibody that specifically binds to EPHA2 or an antigen-binding fragment of the antibody, comprising the following steps (i) or (i) and (ii):
(I) a step of bringing the antibody according to any one of claims 1 to 8 or an antigen-binding fragment of the antibody or the composition according to claim 9 into contact with a sample derived from an individual;
(Ii) if EPHA2 is detected or measured in a sample from the individual, or if the expression level or level of EPHA2 in the sample from the individual is equal to or higher than a predetermined criterion, If it is determined positive and EPHA2 is not detected or measured in the sample derived from the individual, and the expression level or expression level of EPHA2 in the sample derived from the individual is equal to or lower than a predetermined criterion, Determining the individual as negative.
The method according to claims 24 to 26 or the kit according to claim 27, wherein the EPHA2-positive disease is EPHA2-positive cancer.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015089156 | 2015-04-24 | ||
| JP2015-089156 | 2015-04-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016171242A1 true WO2016171242A1 (en) | 2016-10-27 |
Family
ID=57143160
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/062719 Ceased WO2016171242A1 (en) | 2015-04-24 | 2016-04-22 | Detection of epha2 |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2016171242A1 (en) |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110198956A (en) * | 2017-01-23 | 2019-09-03 | 学校法人东洋大学 | Anti-EPHA 2 antibody and the EPHA2 immune detection for using the anti-EPHA 2 antibody |
| WO2021250418A1 (en) * | 2020-06-12 | 2021-12-16 | Bicycletx Limited | Treatment of diseases characterized by overexpression of erythropoietin-producing hepatocellular receptor a2 (epha2) |
| US11623012B2 (en) | 2017-12-19 | 2023-04-11 | Bicyclerd Limited | Bicyclic peptide ligands specific for EphA2 |
| US11814447B2 (en) | 2018-06-22 | 2023-11-14 | Bicyclerd Limited | Peptide ligands for binding to EphA2 |
| US11833211B2 (en) | 2017-12-19 | 2023-12-05 | Bicycletx Limited | Methods of suppression and treatment of disease comprising administering bicycle peptide ligands specific for EphA2 |
| US11912792B2 (en) | 2018-06-22 | 2024-02-27 | Bicycletx Limited | Bicyclic peptide ligands specific for nectin-4 |
| US11970553B2 (en) | 2019-07-30 | 2024-04-30 | Bicycletx Limited | Heterotandem bicyclic peptide complex |
| US12049520B2 (en) | 2017-08-04 | 2024-07-30 | Bicycletx Limited | Bicyclic peptide ligands specific for CD137 |
| US12318454B2 (en) | 2014-10-29 | 2025-06-03 | Bicyclerd Limited | Bicyclic peptide ligands specific for MT1-MMP |
| US12350343B2 (en) | 2018-12-13 | 2025-07-08 | Bicycletx Limited | Bicyclic peptide ligands specific for MT1-MMP |
| US12377155B2 (en) | 2018-12-13 | 2025-08-05 | Bicyclerd Limited | Bicyclic peptide ligands specific for PSMA |
| US12378288B2 (en) | 2018-02-23 | 2025-08-05 | Bicycletx Limited | Multimeric bicyclic peptide ligands |
| US12492224B2 (en) | 2018-12-21 | 2025-12-09 | Bicycletx Limited | Bicyclic peptide ligands specific for PD-L1 |
| US12491253B2 (en) | 2018-12-13 | 2025-12-09 | Bicyclerd Limited | Bicyclic peptide ligands specific for MT1-MMP |
| US12516084B2 (en) | 2019-01-15 | 2026-01-06 | Bicycletx Limited | Bicyclic peptide ligands specific for CD38 |
| US12540161B2 (en) | 2020-08-03 | 2026-02-03 | Bicycletx Limited | Linkers |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008500021A (en) * | 2003-11-20 | 2008-01-10 | メディミューン,インコーポレーテッド | EphA2 agonistic monoclonal antibody and method of use thereof |
| WO2012008397A1 (en) * | 2010-07-12 | 2012-01-19 | 第一三共株式会社 | Anti-epha2 antibody and use thereof |
-
2016
- 2016-04-22 WO PCT/JP2016/062719 patent/WO2016171242A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008500021A (en) * | 2003-11-20 | 2008-01-10 | メディミューン,インコーポレーテッド | EphA2 agonistic monoclonal antibody and method of use thereof |
| WO2012008397A1 (en) * | 2010-07-12 | 2012-01-19 | 第一三共株式会社 | Anti-epha2 antibody and use thereof |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12318454B2 (en) | 2014-10-29 | 2025-06-03 | Bicyclerd Limited | Bicyclic peptide ligands specific for MT1-MMP |
| EP3572426A4 (en) * | 2017-01-23 | 2020-12-23 | Toyo University | ANTI-EPHA2 ANTIBODIES AND IMMUNOLOGICAL DETECTION OF EPHA2 THEREFORE |
| CN110198956A (en) * | 2017-01-23 | 2019-09-03 | 学校法人东洋大学 | Anti-EPHA 2 antibody and the EPHA2 immune detection for using the anti-EPHA 2 antibody |
| US12049520B2 (en) | 2017-08-04 | 2024-07-30 | Bicycletx Limited | Bicyclic peptide ligands specific for CD137 |
| US11623012B2 (en) | 2017-12-19 | 2023-04-11 | Bicyclerd Limited | Bicyclic peptide ligands specific for EphA2 |
| US11833211B2 (en) | 2017-12-19 | 2023-12-05 | Bicycletx Limited | Methods of suppression and treatment of disease comprising administering bicycle peptide ligands specific for EphA2 |
| US12378288B2 (en) | 2018-02-23 | 2025-08-05 | Bicycletx Limited | Multimeric bicyclic peptide ligands |
| US11814447B2 (en) | 2018-06-22 | 2023-11-14 | Bicyclerd Limited | Peptide ligands for binding to EphA2 |
| US11912792B2 (en) | 2018-06-22 | 2024-02-27 | Bicycletx Limited | Bicyclic peptide ligands specific for nectin-4 |
| US12459974B2 (en) | 2018-06-22 | 2025-11-04 | Bicycletx Limited | Bicyclic peptide ligands specific for Nectin-4 |
| US12350343B2 (en) | 2018-12-13 | 2025-07-08 | Bicycletx Limited | Bicyclic peptide ligands specific for MT1-MMP |
| US12377155B2 (en) | 2018-12-13 | 2025-08-05 | Bicyclerd Limited | Bicyclic peptide ligands specific for PSMA |
| US12491253B2 (en) | 2018-12-13 | 2025-12-09 | Bicyclerd Limited | Bicyclic peptide ligands specific for MT1-MMP |
| US12492224B2 (en) | 2018-12-21 | 2025-12-09 | Bicycletx Limited | Bicyclic peptide ligands specific for PD-L1 |
| US12516084B2 (en) | 2019-01-15 | 2026-01-06 | Bicycletx Limited | Bicyclic peptide ligands specific for CD38 |
| US11970553B2 (en) | 2019-07-30 | 2024-04-30 | Bicycletx Limited | Heterotandem bicyclic peptide complex |
| US12435107B2 (en) | 2019-07-30 | 2025-10-07 | Bicycletx Limited | Heterotandem bicyclic peptide complex |
| CN115698720A (en) * | 2020-06-12 | 2023-02-03 | 拜斯科技术开发有限公司 | Treatment of diseases characterized by overexpression of erythropoietin-producing hepatocyte receptor A2 (EPHA2) |
| WO2021250418A1 (en) * | 2020-06-12 | 2021-12-16 | Bicycletx Limited | Treatment of diseases characterized by overexpression of erythropoietin-producing hepatocellular receptor a2 (epha2) |
| US12540161B2 (en) | 2020-08-03 | 2026-02-03 | Bicycletx Limited | Linkers |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2016171242A1 (en) | Detection of epha2 | |
| CA3027417C (en) | Anti-human trop-2 antibody having an antitumor activity in vivo | |
| CA2669731C (en) | Anti-human dlk-1 antibody showing anti-tumor activity in vivo | |
| CN119350498A (en) | Antibody and use thereof | |
| JP2021524242A (en) | Anti-ROR1 antibody and its uses | |
| KR102029248B1 (en) | Cancer-cell-specific antibody, anticancer agent, and cancer testing method | |
| CN110139874B (en) | Anti-CEACAM6 antibodies and methods of use thereof | |
| WO2023219147A1 (en) | Novel anti-ccr8 antibodies for detecting ccr8 | |
| Romani et al. | Evaluation of a novel human IgG1 anti-claudin3 antibody that specifically recognizes its aberrantly localized antigen in ovarian cancer cells and that is suitable for selective drug delivery | |
| JP2024020436A (en) | HER2 targeting agent | |
| CA2948743C (en) | Antibody binding to carbonic anhydrase and use thereof | |
| JPWO2016171107A1 (en) | Detection of FGFR2 | |
| WO2022175474A1 (en) | Vegfa-binding molecules | |
| JP7082112B2 (en) | Anti-GPR20 antibody | |
| JP2022504472A (en) | HER2-binding tetrameric polypeptide | |
| WO2023190465A1 (en) | Human anti-sema7a antibody | |
| HK40013436A (en) | Anti-gpr20 antibody | |
| HK40123863A (en) | Anti-cldn18.2 antibodies and diagnostic uses thereof | |
| HK1198042A (en) | Anti-human trop-2 antibody exhibiting antitumor activity in vivo | |
| NZ623464B2 (en) | Anti-human trop-2 antibody having an antitumor activity in vivo | |
| NZ716839B2 (en) | Anti-human trop-2 antibody having an antitumor activity in vivo | |
| NZ716839A (en) | Anti-human trop-2 antibody having an antitumor activity in vivo |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16783263 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 16783263 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |