WO2016083624A1 - Means for inhibiting the expression of edn1 - Google Patents
Means for inhibiting the expression of edn1 Download PDFInfo
- Publication number
- WO2016083624A1 WO2016083624A1 PCT/EP2015/078108 EP2015078108W WO2016083624A1 WO 2016083624 A1 WO2016083624 A1 WO 2016083624A1 EP 2015078108 W EP2015078108 W EP 2015078108W WO 2016083624 A1 WO2016083624 A1 WO 2016083624A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sirna
- lipoplex
- modified
- nucleotides
- seq
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC(C)OCCOC(CNCCOP([O-])(OCC(COC(*)=O)OC(*)=O)=O)=O Chemical compound CC(C)OCCOC(CNCCOP([O-])(OCC(COC(*)=O)OC(*)=O)=O)=O 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/69—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit
- A61K47/6905—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a colloid or an emulsion
- A61K47/6911—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a colloid or an emulsion the form being a liposome
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/19—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles lyophilised, i.e. freeze-dried, solutions or dispersions
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering nucleic acids [NA]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/50—Methods for regulating/modulating their activity
- C12N2320/51—Methods for regulating/modulating their activity modulating the chemical stability, e.g. nuclease-resistance
Definitions
- the present invention is related to a double-stranded nucleic acid suitable to inhibit the expression of EDNl, compositions, pharmaceutical formulations and uses thereof.
- Endothelin 1 also known as preproendothelin-1 (PPET1), is a protein that in humans is encoded by the EDNl gene. The protein encoded by this gene is proteolytically processed to release a secreted peptide termed endothelin 1. This peptide is a potent vasoconstrictor and is produced by vascular endothelial cells. Endothelin 1 is one of three isoforms of human endothelin (ET-1).
- Preproendothelin is precursor of the peptide ET-1. Endothelial cells convert preproendothelin to proendothelin and subsequently to mature endothelin, which the cells release.
- Endothelin- 1 receptor antagonists are used in the treatment of pulmonary hypertension. Inhibition of these receptors prevents pulmonary vasculature constriction and thus decreases pulmonary vascular resistance.
- the present invention relates to compositions comprising short interfering RNA (siRNA) directed to an expressed RNA transcript of EDNl (sometimes referred to as a "target nucleic acid” herein) and compositions thereof.
- siRNA short interfering RNA
- EDNl expressed RNA transcript of EDNl
- compositions thereof can be used in the treatment of a variety of diseases and disorders where reduced expression of EDNl gene product is desirable.
- Fig. 1 is a summary of siRNA molecules tested in vitro for inhibition of EDNl target gene expression; nucleotides with 2'-0-methyl are marked bold; nucleotides with 2'-F are underlined.
- Fig. 2A is a bar diagram showing the efficacy of different EDNl -specific siRNA molecules on mEDNl mRNA degradation in murine MSI cells; knock-down of mEDNl mRNA is shown in comparison to untreated murine cells or to cells transfected with PTEN (PTENVlO)-specific siRNA and luciferase-specific siRNA being used as negative controls.
- PTEN PTENVlO
- Fig. 2B is a bar diagram showing the efficacy of different EDNl -specific siRNA molecules on PTEN mRNA in murine MSI cells; PTEN mRNA levels are depicted in cells transfected with EDNl siRNAs in comparison to untreated murine cells or cells transfected with luciferase-specific siRNA or PTEN siRNA (PTENVIO) being used as negative controls and positive control, respectively.
- PTENVIO PTEN siRNA
- Fig. 3 is a bar diagram showing the content of endothelin 1 (pg/ml) in supernatant of murine MSI cells 72 hours after transfection with different EDNl-specific siRNA molecules.
- Fig. 4 is a bar diagram showing the content of endothelin 1 (pg/ml) in supernatant of human Human umbilical vein endothelial cells (HuVeCs) 72 hours after transfection with different EDNl-specific siRNA molecules.
- Fig. 5 is a summary outlining the experimental set-up for determining reduction of EDNl expression in mice using different siRNA molecules targeting either EDNl or soluble VEGF receptor 1 (sFltl) and different delivery systems.
- Fig. 6A is a bar diagram showing EDNl mRNA expression in total lysates of lung tissue from mice treated according to the experimental set-up illustrated in Fig. 5; EDNl mRNA expression is normalized relative to PTEN mRNA.
- Fig. 6B is a bar diagram showing relative endothelin 1 protein level in serum from mice treated according to the experimental set-up illustrated in Fig. 5; endothelin 1 protein level is normalized relative to buffer treated group.
- Fig. 8 is a bar diagram showing the result of the in vivo study described in Fig. 7.
- FIG. 9 is a bar diagram showing the result of the in vivo study described in Fig. 7 using mean arterial blood pressure (MABP) as endpoint measurement.
- RVSP right ventricular systolic pressure
- Fig. 10 is a bar diagram showing the result of the in vivo study described in Fig. 7 using right ventricular hypertrophy (expressed as right ventricle weight/body weight) as endpoint measurement.
- Fig. 11 is a bar diagram showing the result of the in vivo study described in Fig. 7 using right ventricular hypertrophy (expressed as right ventricle weight/body weight) as endpoint measurement indicating the values obtained from each animal tested.
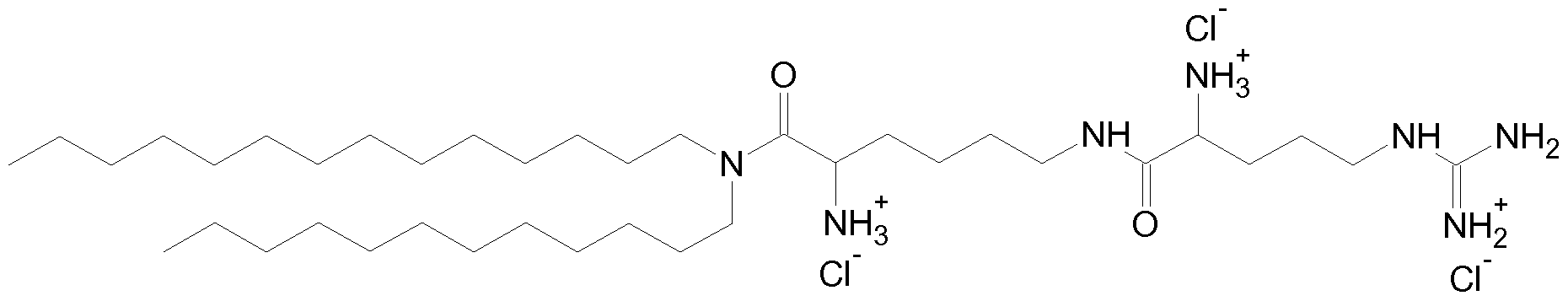
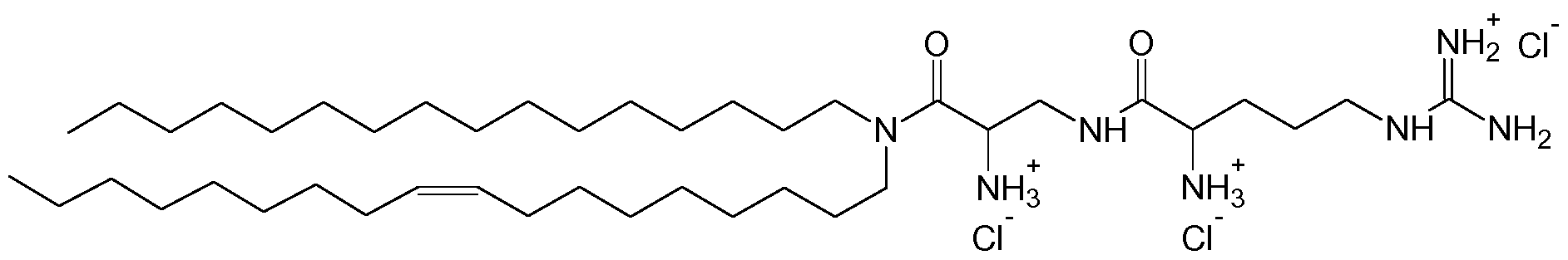
- Fig. 12 Additional EDN1 siRNAs:- Nucleotides with 2' OMe modification are depicted in bold, nucleotides with 2 ' fluoro modification are underlined.
- Fig. 13 Inhibition of EDN1 target gene expression by EDN1 siRNAs in HuVecs (human umbilicord vein endothelial cells). Cells were transfected with 20 nM siRNAs and lmg/ml AtuFECT. Target gene expression (mRNA) was assessed by qRT-PCR two days after transfection. PTEN is a house keeping gene. EDN1 expression levels were normalized to PTEN expression levels. Luc-l-f is a non targeting control siRNA.
- Fig. 14 - is a scheme outlining the experimental set-up of the treatment groups in an intervention study.
- RVSP right ventricular systolic pressure
- Fig. 16 Right ventricular hypertrophy (right ventricle: left ventricle+septum, RV/[LV+S], ratio) in normoxic mice and animals exposed to hypoxia (10% 02, 5 weeks) plus sugen (20mg/kg at day 0, 7 & 14) in the absence and presence of Dacc9/luc (2X/week), Dacc9/EDNl-fl (2X/week), Dacc9/edn (2X/week) or Bosentan (lOOmg/kg/day; p.o.).
- Fig. 18 - EDN1 mR A expression levels in lung tissue were determined after the end of the treatment by qRT-PCR.
- SEQ ID NOs: 1 to 16 are strands of different siRNAs targeting human EDN1.
- SEQ ID NO: 17 is the mRNA sequence encoding human EDN1. This sequence has been taken from GenBank Accession number NM 001955.4. which is incorporated herein by reference in its entirety.
- SEQ ID NOs: 18 and 19 are the two strands of an siRNA targeting luciferase.
- SEQ ID NOs: 20 and 21 are the two strands of an siRNA targeting PTEN.
- SEQ ID NOs: 22 to 45 are further strands of different double stranded siRNAs targeting human EDN1
- EDNl-hmr-2A 5 ' uuucaauuugugcauuccu 3 ' SEQ ID NO:l 5 ' uuucaauuugugcauuccu 3 ' EDNl-hmr-2B 5 ' aggaaugcacaaauugaaa 3 ' SEQ ID NO: 2 5 'aggaaugcacaaauugaaa 3'
- the present invention relates to compositions comprising short interfering RNA
- siRNA directed to an expressed RNA transcript of EDNl (sometimes referred to as a "target nucleic acid” herein).
- the siRNA of the invention are nucleic acid molecules comprising a double stranded or duplex region.
- the present invention further relates to methods of using the siRNA compositions to reduce the expression level of EDNl .
- the terms "silence” or “knock-down” when referring to gene expression means a reduction in gene expression.
- the present invention further relates to processes for making the siRNA.
- the target nucleic acid is an RNA expressed from a mammalian EDNl gene. In one embodiment, the target nucleic acid is an RNA expressed from mouse EDNl . In another embodiment, the target nucleic acid is an RNA expressed from human EDNl . In another embodiment, the target nucleic acid is a human EDNl mRNA. In another embodiment, the target nucleic acid is a human EDNl mRNA. In another embodiment, the target nucleic acid is an mRNA comprising the sequence of SEQ ID NO: 17.
- the siRNA of the present invention are suitable to inhibit the expression of EDNl .
- the siRNA according to the present invention is, thus, suitable to trigger the RNA interference response resulting in the reduction of the EDNl mRNA in a mammalian cell.
- the siRNA according to the present invention are further suitable to decrease the expression of EDNl protein by decreasing gene expression at the level of mRNA.
- siRNA Design An siRNA of the present invention comprises two strands of a nucleic acid, a first, antisense strand and a second, sense strand.
- the nucleic acid normally consists of ribonucleotides or modified ribonucleotides however; the nucleic acid may comprise deoxynucleotides (DNA) as described herein.
- the siRNA further comprises a double- stranded nucleic acid portion or duplex region formed by all or a portion of the antisense strand and all or a portion of the sense strand.
- the portion of the antisense strand forming the duplex region with the sense strand is the antisense strand duplex region or simply, the antisense duplex region, and the portion of the sense strand forming the duplex region with the antisense strand is the sense strand duplex region or simply, the sense duplex region.
- the duplex region is defined as beginning with the first base pair formed between the antisense strand and the sense strand and ending with the last base pair formed between the antisense strand and the sense strand, inclusive.
- the portion of the siRNA on either side of the duplex region is the flanking regions.
- the portion of the antisense strand on either side of the antisense duplex region is the antisense flanking regions.
- the portion of the antisense strand 5' to the antisense duplex region is the antisense 5' flanking region.
- the portion of the antisense strand 5' to the antisense duplex region is the antisense 3' flanking region.
- the portion of the sense strand on either side of the sense duplex region is the sense flanking regions.
- the portion of the sense strand 5 ' to the sense duplex region is the sense 5' flanking region.
- the portion of the sense strand 5' to the sense duplex region is the sense 3' flanking region.
- the antisense duplex region and the sense duplex region may be fully complementary and are at least partially complementary to each other.
- Such complementarity is based on Watson-Crick base pairing (i.e., A:U and G:C base pairing).
- A:U and G:C base pairing i.e., A:U and G:C base pairing.
- the antisense and sense strands must be able to hybridize under physiological conditions.
- the complementarity between the antisense strand and sense strand is perfect (no nucleotide mismatches or additional/deleted nucleotides in either strand).
- the complementarity between the antisense duplex region and sense duplex region is perfect (no nucleotide mismatches or additional/deleted nucleotides in the duplex region of either strand).
- the complementarity between the antisense duplex region and the sense duplex region is not perfect.
- the identity between the antisense duplex region and the complementary sequence of the sense duplex region is selected from the group consisting of at least 75%, 80%>, 85%, 90% and 95%>; wherein a siRNA comprising the antisense duplex region and the sense duplex region is suitable for reducing expression of EDN1
- the siRNA, wherein the identity between the antisense duplex region and complementary sequence of the sense duplex region is selected from the group consisting of at least 75%, 80%>, 85%, 90% and 95% is able to reduce expression of EDN1 by at least 25%, 50% or 75% of a comparative siRNA having a duplex region with perfect identity between the antisense duplex region and the sense duplex region.
- the term "comparative siRNA” is a siRNA that is identical to the siRNA to which it is being compared, except for the specified difference, and which is tested under identical conditions.
- R Ai using siRNA involves the formation of a duplex region between all or a portion of the antisense strand and a portion of the target nucleic acid.
- the portion of the target nucleic acid that forms a duplex region with the antisense strand defined as beginning with the first base pair formed between the antisense strand and the target sequence and ending with the last base pair formed between the antisense strand and the target sequence, inclusive, is the target nucleic acid sequence or simply, target sequence.
- the duplex region formed between the antisense strand and the sense strand may, but need not be the same as the duplex region formed between the antisense strand and the target sequence. That is, the sense strand may have a sequence different from the target sequence however; the antisense strand must be able to form a duplex structure with both the sense strand and the target sequence.
- the complementarity between the antisense strand and the target sequence is perfect (no nucleotide mismatches or additional/deleted nucleotides in either nucleic acid).
- the complementarity between the antisense duplex region (the portion of the antisense strand forming a duplex region with the sense strand) and the target sequence is perfect (no nucleotide mismatches or additional/deleted nucleotides in either nucleic acid).
- the complementarity between the antisense duplex region and the target sequence is not perfect.
- the identity between the antisense duplex region and the complementary sequence of the target sequence is selected from the group consisting of at least 75%, 80%, 85%, 90% or 95%, wherein a siRNA comprising the antisense duplex region is suitable for reducing expression of EDN1.
- the siRNA, wherein the identity between the antisense duplex region and complementary sequence of the target sequence is selected from the group consisting of at least 75%, 80%, 85%, 90% and 95% is able to reduce expression of EDN1 by at least 25%, 50% or 75% of a comparative siRNA with perfect identity to the antisense strand and target sequence.
- the siRNA of the invention comprises a duplex region wherein the antisense duplex region has a number of nucleotides selected from the group consisting of 1, 2, 3, 4 and 5 that are not base-paired to a nucleotide in the sense duplex region, and wherein said siRNA is suitable for reducing expression of EDN1.
- Lack of base- pairing is due to either lack of complementarity between bases (i.e., no Watson-Crick base pairing) or because there is no corresponding nucleotide on either the antisense duplex region or the sense duplex region such that a bulge is created.
- a siRNA comprising an antisense duplex region having a number of nucleotides selected from the group consisting of 1, 2, 3, 4 and 5 that are not base-paired to the sense duplex region, is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense duplex region are base paired with all nucleotides of said sense duplex region.
- the antisense strand has a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that do not base-pair to the sense strand, and wherein a siRNA comprising said antisense strand is suitable for reducing expression of EDN1.
- Lack of complementarity is due to either lack of complementarity between bases or because there is no corresponding nucleotide on either the antisense strand or the sense strand. The lack of a corresponding nucleotide results in either a single-stranded overhang or a bulge (if in the duplex region), in either the antisense strand or the sense strand.
- a siRNA comprising an antisense strand having a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that do not base pair to the sense strand, is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of the sense strand.
- a siRNA comprising an antisense strand having a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that are mismatched to the target sequence, is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of said sense strand. In another embodiment, all of the mismatched nucleotides are outside the duplex region.
- the antisense duplex region has a number of nucleotides selected from 1, 2, 3, 4 or 5 that do not base-pair to the sense duplex region, and wherein a siRNA comprising said antisense duplex region is suitable for reducing expression of EDN1.
- Lack of complementarity is due to either lack of complementarity between bases or because there is no corresponding nucleotide on either the antisense duplex region or the sense duplex region such that a bulge in created in either the antisense duplex region or the sense duplex region.
- a siRNA comprising an antisense duplex region having a number of nucleotides selected from the group consisting of 1, 2, 3, 4 and 5 that do not base pair to the sense duplex region is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense duplex region are complementary to all of the nucleotides of said sense duplex region.
- the antisense strand has a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that do not base-pair to the target sequence, and wherein a siRNA comprising said antisense strand is suitable for reducing expression of EDNl .
- a siRNA comprising an antisense strand having a number of nucleotides selected from 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10 do not base pair to the target sequence, is able to reduce expression of EDNl by at least 25%, 50%>, 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of said target sequence.
- a siRNA comprising an antisense strand having a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that are mismatched to the target sequence, is able to reduce expression of EDNl by at least 25%, 50% or 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of said target sequence.
- the complementarity between an antisense duplex region and both a sense duplex region and a target sequence of an siRNA is such that the antisense duplex region and the sense duplex region or the target sequence hybridize to one another under physiological conditions (37°C in a physiological buffer) and the siRNA is suitable for reducing expression of EDNl .
- the siRNA comprising an antisense duplex region that hybridizes to a sense duplex region and a target sequence under physiological conditions is able to reduce expression of EDNl by at least 25%, 50%, 75% of a comparative siRNA with perfect complementarity between the antisense strand and target sequence.
- the complementarity between an antisense duplex region and a sense duplex region of a siRNA is such that the antisense duplex region and sense duplex region hybridize under the following conditions: 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C, and is suitable for reducing expression of EDNl .
- the siRNA comprising an antisense duplex region and a sense duplex region that hybridize to one another under the conditions 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C, is able to reduce expression of EDNl by at least 25%, 50%, 75% of a comparative siRNA with perfect complementarity between the antisense duplex region and sense duplex region.
- the complementarity between an antisense strand of a siRNA and a target sequence is such that the antisense strand and target sequence hybridize under the following conditions: 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C and wherein the siRNA is suitable for reducing expression of EDNl .
- the siRNA comprising an antisense strand that hybridizes to the target sequence under the following conditions: 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C, is able to reduce expression of EDNl by at least 25%, 50%>, 75% of a comparative siRNA with perfect complementarity between the antisense strand and the target sequence.
- RNA interference is observed using long nucleic acid molecules comprising several dozen or hundreds of base pairs, although shorter RNAi molecules are generally preferred.
- the length of the siRNA duplex region is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 base pairs. In one embodiment, the length of the siRNA duplex region is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 consecutive base pairs.
- the length of the siRNA duplex region is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 base pairs. In another embodiment, the length of the siRNA duplex region is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 consecutive base pairs.
- the length of the antisense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides.
- the length of the antisense stand is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides.
- the length of the sense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides.
- the length of the sense stand is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides.
- the length of the antisense strand and the length of the sense strand are independently selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides.
- the length of the antisense strand and the length of the sense stand are independently selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides.
- the antisense strand and the sense strand are equal in length.
- the antisense strand and the sense stand are equal in length, wherein the length is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides.
- the length of the antisense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NO: 1, 3, 5, 7, 9, 11, 13 or 15, preferably 1, 3, 5, or 7.
- the length of the antisense strand is selected from the group consisting of about 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7.
- the length of the sense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
- the length of the sense strand is selected from the group consisting of about 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides, wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
- the length of the antisense strand and the length of the sense strand are independently selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NO. NOs: 2, 4, 6, 8, 10, 12, 14, 16, 18, 20, 22,
- the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
- the length of the antisense strand and the length of the sense stand are independently selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7, and wherein the sense strand comprises the nucleotide sequence SEQ ID NOs: 2,
- the antisense strand and the sense strand are equal in length, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7, and wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
- the antisense strand and the sense stand are equal in length, wherein the length is selected from the group consisting of 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11 , 13 or 15, preferably SEQ ID NOs: 1, 3,
- the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4,
- the antisense strand and the sense stand are equal in length, wherein the length is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24,
- the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7, and wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
- Certain embodiments provide for antisense and sense strand combinations (identified by SEQ ID NO:): 1 and 2; 3 and 4; 5 and 6; and 7 and 8.
- the siRNA of the present invention may comprise an overhang or be blunt ended.
- An "overhang” as used herein has its normal and customary meaning in the art, i.e., a single stranded portion of a nucleic acid that extends beyond the terminal nucleotide of a complementary strand in a double strand nucleic acid.
- the term "blunt end” includes double stranded nucleic acid whereby both strands terminate at the same position, regardless of whether the terminal nucleotide(s) are base paired.
- the terminal nucleotide of an antisense strand and a sense strand at a blunt end are base paired.
- the terminal nucleotide of a antisense strand and a sense strand at a blunt end are not paired. In another embodiment, the terminal two nucleotides of an antisense strand and a sense strand at a blunt end are base paired. In another embodiment, the terminal two nucleotides of a antisense strand and a sense strand at a blunt end are not paired.
- the siRNA has an overhang at one end and a blunt end at the other. In another embodiment, the siRNA has an overhang at both ends. In another embodiment, the siRNA is blunt ended at both ends. In one embodiment, the siRNA is blunt ended at one end. In another embodiment, the siRNA is blunt ended at the end with the 5 '- end of the antisense strand and the 3 '-end of the sense strand. In another embodiment, the siRNA is blunt ended at the end with the 3 '-end of the antisense strand and the 5 '-end of the sense strand. In another embodiment, the siRNA is blunt ended at both ends.
- the siRNA comprises a overhang at a 3'- or 5 '-end. In one embodiment, the siRNA has a 3 '-overhang on the antisense strand. In another embodiment, the siRNA has a 3 '-overhang on the sense strand. In another embodiment, the siRNA has a 5'- overhang on the antisense strand. In another embodiment, the siRNA has a 5 '-overhang on the sense strand. In another embodiment, the siRNA has an overhang at both the 5 '-end and 3'- end of the antisense stand. In another embodiment, the siRNA has an overhang at both the 5'- end and 3 '-end of the sense stand.
- the siRNA has a 5' overhang on the antisense stand and a 3 ' overhang on the sense strand. In another embodiment, the siRNA has a 3' overhang on the antisense stand and a 5' overhang on the sense strand. In another embodiment, the siRNA has a 3' overhang on the antisense stand and a 3' overhang on the sense strand. In another embodiment, the siRNA has a 5' overhang on the antisense stand and a 5 ' overhang on the sense strand.
- the overhang at the 3 '-end of the antisense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides. In one embodiment, the overhang at the 3 '-end of the sense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides. In one embodiment, the overhang at the 5 '-end of the antisense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides. In one embodiment, the overhang at the 5 '-end of the sense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides.
- the siR A according to the invention are a ribonucleic acid or a modified ribonucleic acid.
- Chemical modifications of the siRNA of the present invention provides a powerful tool in overcoming potential limitations including, but not limited to, in vitro and in vivo stability and bioavailability inherent to native RNA molecules. Chemically-modified siRNA can also minimize the possibility of activating interferon activity in humans. Chemical modification can further enhance the functional delivery of a siRNA to a target cell.
- the modified siRNA of the present invention may comprise one or more chemically modified ribonucleotides of either or both of the antisense strand or the sense strand.
- a ribonucleotide may comprise a chemical modification of the base, sugar or phosphate moieties.
- a secondary aspect relates to modifications to a base moiety.
- One or more nucleotides of a siRNA of the present invention may comprise a modified base.
- a "modified base” means a nucleotide base other than an adenine, guanine, cytosine or uracil at the ⁇ position.
- the siRNA comprises at least one nucleotide comprising a modified base.
- the modified base in on the antisense strand.
- the modified base in on the sense strand.
- the modified base is in the duplex region.
- the modified base is outside the duplex region, i.e., in a single stranded region.
- the modified base is on the antisense strand and is outside the duplex region.
- the modified base is on the sense strand and is outside the duplex region.
- the 3 '-terminal nucleotide of the antisense strand is a nucleotide with a modified base.
- the 3 '-terminal nucleotide of the sense strand is nucleotide with a modified base.
- the 5 '-terminal nucleotide of the antisense strand is nucleotide with a modified base.
- the 5 '-terminal nucleotide of the sense strand is nucleotide with a modified base.
- a siRNA has 1 modified base. In another embodiment, a siRNA has about 2-4 modified bases. In another embodiment, a siRNA has about 4-6 modified bases. In another embodiment, a siRNA has about 6-8 modified bases. In another embodiment, a siRNA has about 8-10 modified bases. In another embodiment, a siRNA has about 10-12 modified bases. In another embodiment, a siRNA has about 12-14 modified bases. In another embodiment, a siRNA has about 14-16 modified bases. In another embodiment, a siRNA has about 16-18 modified bases. In another embodiment, a siRNA has about 18-20 modified bases. In another embodiment, a siRNA has about 20-22 modified bases. In another embodiment, a siRNA has about 22-24 modified bases.
- a siRNA has about 24-26 modified bases. In another embodiment, a siRNA has about 26-28 modified bases. In each case the siRNA comprising said modified bases retains at least 50% of its activity as compared to the same siRNA but without said modified bases.
- the modified base is a purine. In another embodiment, the modified base is a pyrimidine. In another embodiment, at least half of the purines are modified. In another embodiment, at least half of the pyrimidines are modified. In another embodiment, all of the purines are modified. In another embodiment, all of the pyrimidines are modified.
- the siRNA comprises a nucleotide comprising a modified base, wherein the base is selected from the group consisting of 2-aminoadenosine, 2,6- diaminopurine,inosine, pyridin-4-one, pyridin-2-one, phenyl, pseudouracil, 2, 4, 6-trimethoxy benzene, 3-methyl uracil, dihydrouridine, naphthyl, aminophenyl, 5-alkylcytidine (e.g., 5- methylcytidine), 5-alkyluridine (e.g., ribothymidine), 5-halouridine (e.g., 5-bromouridine), 6- azapyrimidine, 6-alkylpyrimidine (e.g.
- 6-methyluridine 6-methyluridine
- propyne quesosine, 2-thiouridine, 4- thiouridine, wybutosine, wybutoxosine, 4-acetylcytidine, 5-(carboxyhydroxymethyl)uridine, 5'-carboxymethylaminomethyl-2-thiouridine, 5-carboxymethylaminomethyluridine, beta-D- galactosylqueosine, 1-methyladenosine, 1-methylinosine, 2,2-dimethylguanosine, 3- methylcytidine, 2-methyladenosine, 2-methylguanosine, N6-methyladenosine, 7- methylguanosine, 5-methoxyaminomethyl-2-thiouridine, 5-methylaminomethyluridine, 5- methylcarbonylmethyluridine, 5-methyloxyuridine, 5-methyl-2-thiouridine, 2-methylthio-N6- isopentenyladenosine, beta-D-mannosylqueo
- a siRNA of the present invention comprises an abasic nucleotide.
- abasic refers to moieties lacking a base or having other chemical groups in place of a base at the ⁇ position, for example a 3',3'-linked or 5',5'-linked deoxyabasic ribose derivative.
- a nucleotide with a modified base does not include abasic nucleotides.
- the siRNA comprises at least one abasic nucleotide.
- the abasic nucleotide is on the antisense strand.
- the abasic nucleotide is on the sense strand.
- the abasic nucleotide is in the duplex region. In another embodiment, the abasic nucleotide is outside the duplex region. In another embodiment, the abasic nucleotide is on the antisense strand and is outside the duplex region. In another embodiment, the abasic nucleotide is on the sense strand and is outside the duplex region. In another embodiment, the 3 '-terminal nucleotide of the antisense strand is an abasic nucleotide. In another embodiment, the 3 '-terminal nucleotide of the sense strand is an abasic nucleotide.
- the 5 '-terminal nucleotide of the antisense strand is an abasic nucleotide.
- the 5 '-terminal nucleotide of the sense strand is an abasic nucleotide.
- a siR A has a number of abasic nucleotides selected from the group consisting of 1, 2, 3, 4, 5 and 6.
- Modifications to sugar moiety Another secondary aspect relates to modifications to a sugar moiety.
- One or more nucleotides of an siRNA of the present invention may comprise a modified ribose moiety.
- Modifications at the 2'-position wherein the 2'-OH is substituted include the non- limiting examples selected from the group consisting of alkyl, substituted alkyl, alkaryl-, aralkyl-, -F, -CI, -Br, -CN, -CF3, -OCF3, -OCN, -O-alkyl, -S-alkyl, HS-alkyl-O, -O-alkenyl, -S-alkenyl, -N-alkenyl, -SO-alkyl, -alkyl-OSH, -alkyl-OH, -O-alkyl-OH, -O-alkyl-SH, -S- alkyl-OH, -S-alkyl-SH, -alkyl-S-alkyl, -alkyl-O-alkyl, -ON02, -N02, -N3, -NH2, alkylamino, dialkyla
- LNA Locked nucleic acids
- Preferred substitutents are 2'-methoxy ethyl, 2'-0-CH3, 2'-0-allyl, 2'-C-allyl, and 2'- fluoro.
- the siRNA comprises 1-5 2'-modified nucleotides. In another embodiment, the siRNA comprises 5-10 2'-modified nucleotides. In another embodiment, the siRNA comprises 15-20 2'-modified nucleotides. In another embodiment, the siRNA comprises 20-25 2'-modified nucleotides. In another embodiment, the siRNA comprises 25- 30 2 '-modified nucleotides.
- the antisense strand comprises 1-2 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 2-4 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 4-6 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 6-8 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 8-10 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 10-12 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 12-14 2'-modified nucleotides.
- the antisense strand comprises about 14-16 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 16-18 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 18-20 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 22-24 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 24-26 2'-modified nucleotides.
- the sense strand comprises 1-2 2'-modified nucleotides. In one embodiment, the sense strand comprises about 2-4 2'-modified nucleotides. In one embodiment, the sense strand comprises about 4-6 2'-modified nucleotides. In one embodiment, the sense strand comprises about 6-8 2'-modified nucleotides. In one embodiment, the sense strand comprises about 8-10 2'-modified nucleotides. In one embodiment, the sense strand comprises about 10-12 2'-modified nucleotides. In one embodiment, the sense strand comprises about 12-14 2'-modified nucleotides. In one embodiment, the sense strand comprises about 14-16 2'-modified nucleotides.
- the sense strand comprises about 16-18 2'-modified nucleotides. In one embodiment, the sense strand comprises about 18-20 2'-modified nucleotides. In one embodiment, the sense strand comprises about 22-24 2'-modified nucleotides. In one embodiment, the sense strand comprises about 24-26 2'-modified nucleotides.
- the siR A comprises 1-5 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 5-10 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 15-20 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 20-25 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 25-30 2'-0-CH3 modified nucleotides.
- the antisense strand comprises 1-2 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 2-4 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 4-6 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 6-8 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 8-10 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 10-12 2'-0- CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 12-14 2'-0-CH3 modified nucleotides.
- the antisense strand comprises about 14- 16 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 16-18 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 18-20 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 22-24 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 24-26 2'-0-CH3 modified nucleotides.
- the sense strand comprises 1-2 2'-0-CH3 modified nucleotides.
- the sense strand comprises about 2-4 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 4-6 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 6-8 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 8-10 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 10-12 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 12-14 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 14-16 2'-0-CH3 modified nucleotides.
- the sense strand comprises about 16-18 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 18-20 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 22-24 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 24-26 2'-0-CH3 modified nucleotides.
- the siR A duplex region comprises 1-5 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 5-10 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 15-20 2'- 0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 20-25 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 25-30 2'-0-CH3 modified nucleotides.
- the antisense duplex region comprises 1-2 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 2-4 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 4-6 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 6-8 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 8-10 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 10-12 2'-0-CH3 modified nucleotides.
- the antisense duplex region comprises about 12-14 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 14-16 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 16-18 2'-0- CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 18-20 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 22-24 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 24-26 2'-0-CH3 modified nucleotides.
- the sense duplex region comprises 1-2 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 2-4 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 4-6 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 6-8 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 8-10 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 10-12 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 12-14 2'-0-CH3 modified nucleotides.
- the sense duplex region comprises about 14-16 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 16-18 2'-0- CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 18-20 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 22-24 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 24-26 2'-0-CH3 modified nucleotides.
- the siRNA comprises an antisense strand 19 nucleotides in length and a sense strand 19 nucleotides in length, wherein said antisense strand comprises 2'-0- CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17 and 19, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16 and 18, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'.
- the siRNA comprises an antisense strand 20 nucleotides in length and a sense strand 20 nucleotides in length, wherein said antisense strand comprises 2'- 0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17 and 19, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18 and 20 wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'.
- the siRNA comprises an antisense strand 21 nucleotides in length and a sense strand 21 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17, 19 and 21, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18 and 20, wherein said antisense strand is numbered from 5'-3' and said sense strand is numbered from 3 '-5'.
- the siRNA comprises an antisense strand 22 nucleotides in length and a sense strand 22 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17, 19 and 21, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18, 20 and 22, wherein said antisense strand is numbered from 5'-3 ' and said sense strand is numbered from 3 '-5'.
- the siR A comprises an antisense strand 23 nucleotides in length and a sense strand 23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17, 19, 21 and 23, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18, 20 and 22 wherein said antisense strand is numbered from 5 '-3 ' and said sense strand is numbered from 3 '-5 ' .
- the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 3, 5, 7, 9, 11, 13, 15 and 17, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 4, 6, 8, 10, 12 ,14 and 16, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5' .
- the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0- CH3 modifications at nucleotides 5, 7, 9, 11, 13 and 15, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 6, 8, 10, 12 and 14, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'.
- the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 7, 9, 11, 13 and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 8, 10 and 12, wherein said antisense strand is numbered from 5'- 3' and said sense strand is numbered from 3 '-5'.
- the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 7, 9 and 11, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 8, 10 and 12, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'.
- the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 7 and 9, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 8 and 10, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'.
- the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0- CH3 modifications at nucleotides 9 and 11, and wherein said sense strand comprises 2'-0- CH3 modifications at nucleotides 8 and 10, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'.
- the antisense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25 2'-deoxy nucleotides.
- the sense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25 2 '-deoxy nucleotides.
- the antisense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11,
- the sense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12,
- the pyrimidine nucleotides in the antisense strand are 2'-0- methyl pyrimidine nucleotides.
- purine nucleotides in the antisense strand are 2'-0- methyl purine nucleotides.
- the pyrimidine nucleotides in the antisense strand are 2'- deoxy pyrimidine nucleotides.
- the purine nucleotides in the antisense strand are 2 '-deoxy purine nucleotides.
- the pyrimidine nucleotides in the antisense strand are 2'- fluoro pyrimidine nucleotides.
- the purine nucleotides in the antisense strand are 2'-fluoro purine nucleotides.
- the pyrimidine nucleotides in the sense strand are 2'-0-methyl pyrimidine nucleotides.
- purine nucleotides in the sense strand are 2'-0-methyl purine nucleotides.
- the pyrimidine nucleotides in the sense strand are 2'-deoxy pyrimidine nucleotides.
- the purine nucleotides in the sense strand are 2'-deoxy purine nucleotides.
- the pyrimidine nucleotides in the sense strand are 2'-fluoro pyrimidine nucleotides.
- the purine nucleotides in the sense strand are 2'-fluoro purine nucleotides.
- the pyrimidine nucleotides in the antisense duplex region are 2'-0-methyl pyrimidine nucleotides.
- purine nucleotides in the antisense duplex region are 2'- O-methyl purine nucleotides.
- the pyrimidine nucleotides in the antisense duplex region are 2'-deoxy pyrimidine nucleotides.
- the purine nucleotides in the antisense duplex region are 2'- deoxy purine nucleotides.
- the pyrimidine nucleotides in the antisense duplex region are
- the purine nucleotides in the antisense duplex region are 2'- fluoro purine nucleotides.
- the pyrimidine nucleotides in the sense duplex region are 2'- O-methyl pyrimidine nucleotides.
- purine nucleotides in the sense duplex region are 2'-0- methyl purine nucleotides.
- the pyrimidine nucleotides in the sense duplex region are 2'- deoxy pyrimidine nucleotides.
- the purine nucleotides in the sense duplex region are 2'-deoxy purine nucleotides.
- the pyrimidine nucleotides in the sense duplex region are 2'- fluoro pyrimidine nucleotides.
- the purine nucleotides in the sense duplex region are 2'-fluoro purine nucleotides.
- the pyrimidine nucleotides in the antisense duplex flanking regions are 2'-0-methyl pyrimidine nucleotides.
- of the purine nucleotides in the antisense duplex flanking regions are 2'-0-methyl purine nucleotides.
- the pyrimidine nucleotides in the antisense duplex flanking regions are 2 '-deoxy pyrimidine nucleotides.
- the purine nucleotides in the antisense duplex flanking regions are 2'-deoxy purine nucleotides.
- the pyrimidine nucleotides in the antisense duplex flanking regions are 2'-fluoro pyrimidine nucleotides.
- the purine nucleotides in the antisense duplex flanking regions are 2'-fluoro purine nucleotides.
- the pyrimidine nucleotides in the sense duplex flanking regions are 2'-0-methyl pyrimidine nucleotides.
- purine nucleotides in the sense duplex flanking regions are 2'-0-methyl purine nucleotides.
- the pyrimidine nucleotides in the sense duplex flanking regions are 2 '-deoxy pyrimidine nucleotides.
- the purine nucleotides in the sense duplex flanking regions are 2 '-deoxy purine nucleotides.
- the pyrimidine nucleotides in the sense duplex flanking regions are 2'-fluoro pyrimidine nucleotides.
- the purine nucleotides in the sense duplex flanking regions are 2'-fluoro purine nucleotides.
- the antisense duplex region comprises a plurality of groups of modified nucleotides, referred to herein as "modified groups", wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a second group of nucleotides, referred to herein as “flanking groups", wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group.
- each modified group in the antisense duplex region is identical, i.e., each modified group consists of an equal number of identically modified nucleotides.
- each flanking group has an equal number of nucleotide. In another embodiment, each flanking group is identical. In another embodiment, the nucleotides of said modified groups in the antisense duplex region comprise a modified base. In another embodiment, the nucleotides of said modified groups comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups comprise a modified 2' position.
- the sense duplex region comprises a plurality of groups of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group.
- each modified group in the sense duplex region is identical.
- each flanking group has an equal number of nucleotides.
- each flanking group is identical.
- the nucleotides of said modified groups in the sense duplex region comprise a modified base.
- the nucleotides of said modified groups comprise a modified phosphate backbone.
- the nucleotides of said modified groups comprise a modified 2' position.
- the antisense duplex region and the sense duplex region each comprise a plurality of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group.
- each modified group in the antisense duplex region and the sense duplex region are identical.
- each flanking group in the antisense duplex region and the sense duplex region each have an equal number of nucleotides.
- each flanking group in the antisense duplex region and in the sense duplex region are identical.
- the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise the same modified groups and the same flanking groups.
- the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise a modified base.
- the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise a modified phosphate backbone.
- the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise a modified 2' position.
- the antisense strand comprises a plurality of groups of modified nucleotides, referred to herein as "modified groups", wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a second group of nucleotides, referred to herein as “flanking groups", wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group.
- each modified group in the antisense strand is identical, i.e., each modified group consists of an equal number of identically modified nucleotides.
- each flanking group has an equal number of nucleotide. In another embodiment, each flanking group is identical. In another embodiment, the nucleotides of said modified groups in the antisense strand comprise a modified base. In another embodiment, the nucleotides of said modified groups comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups comprise a modified 2' position.
- the sense strand comprises a plurality of groups of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group.
- each modified group in the sense strand is identical.
- each flanking group has an equal number of nucleotides.
- each flanking group is identical.
- the nucleotides of said modified groups in the sense strand comprise a modified base.
- the nucleotides of said modified groups comprise a modified phosphate backbone.
- the nucleotides of said modified groups comprise a modified 2' position.
- the antisense strand and the sense strand each comprise a plurality of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group.
- each modified group in the antisense strand and the sense strand are identical.
- each flanking group in the antisense strand and the sense strand each have an equal number of nucleotides.
- each flanking group in the antisense strand and in the sense strand are identical.
- nucleotides of said modified groups in the antisense strand and the sense strand each comprise the same modified groups and the same flanking groups. In another embodiment, the nucleotides of said modified groups in the antisense strand and the sense strand each comprise a modified base. In another embodiment, the nucleotides of said modified groups in the antisense strand and the sense strand each comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups in the antisense strand and the sense strand each comprise a modified 2' position.
- the modified groups and the flanking groups form a regular pattern on the antisense stand. In another aspect, the modified groups and the flanking groups form a regular pattern on the sense strand. In one embodiment, the modified groups and the flanking groups form a regular pattern on the both the antisense strand and the sense strand. In another embodiment, the modified groups and the flanking groups form a regular pattern on the antisense duplex region. In another aspect, the modified groups and the flanking groups form a regular pattern on the sense duplex region. In one embodiment, the modified groups and the flanking groups form a regular pattern on the both the antisense duplex region and the sense duplex region.
- the pattern is a spatial or positional pattern.
- a spatial or positional pattern means that (a) nucleotide(s) are modified depending on their position within the nucleotide sequence of a double-stranded portion. Accordingly, it does not matter whether the nucleotide to be modified is a pyrimidine or a purine.
- a modified nucleotide is dependent upon: (a) its numbered position on a strand of nucleic acid, wherein the nucleotides are numbered from the 5 '-end to the 3 '-end with the 5 '-end nucleotide of the strand being position one (both the antisense strand and sense strand are numbered from their respective 5 '-end nucleotide), or (b) the position of the modified group relative to a flanking group.
- the modification pattern will always be the same, regardless of the sequence which is to be modified.
- the number of modified groups on the antisense strand is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
- the number of modified groups on the sense strand is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
- the number of flanking groups on the antisense strand of nucleic acid is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
- the number of flanking groups on the sense strand of nucleic acid is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
- the number of modified groups and the number of flanking groups on either or both the antisense strand and the sense strand are the same.
- the number of modified groups on the antisense duplex region is selected 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of modified groups on the sense duplex region is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13 or 14. In another embodiment, the number of flanking groups on the antisense duplex region of nucleic acid is 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of flanking groups on the sense duplex region of nucleic acid is selected from 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13 or 14. In one embodiment, the number of modified groups and the number of flanking groups on either or both the antisense duplex region and the sense duplex region are the same.
- the number of modified groups and the number of flanking groups on a strand or on a duplex region are the same. In another embodiment, the number of modified groups and the number of flanking groups on a strand or on a duplex region are the same, wherein the number is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
- the number of nucleotides in a modified group is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of nucleotides in a flanking group is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
- each modified group on both the antisense strand and the sense strand is identical. In one embodiment, each modified group on both the antisense duplex region and the sense duplex region is identical. In another embodiment, each modified group and each flanking group on both the antisense strand and the sense strand are identical. In one embodiment, each modified group and each flanking group on both the antisense duplex region and the sense duplex region are identical.
- each modified group, each modified group position, each flanking group and each flanking group position on both the antisense strand and the sense strand are identical. In one embodiment, each modified group, each modified group position, each flanking group and each flanking group position on both the antisense duplex region and the sense duplex region are identical. In another embodiment, the modified groups on the antisense strand are complementary with the modified groups on the sense strand (the modified groups on the antisense strand and the sense strand are perfectly aligned across from one another). In another embodiment, there are no mismatches in the modified groups such that each modified group on the antisense strand is base paired with each modified group on the sense strand.
- each modified group on the sense strand is shifted by 1 , 2, 3, 4 or 5 nucleotides relative to the modified groups on the antisense strand. For example, if each modified group on the sense strand is shifted by one nucleotide and a modified group started at position one on the antisense strand, a modified group on the sense strand would begin at position two.
- the modified groups of the antisense strand do not overlap the modified groups of the sense strand, i.e., no nucleotide of a modified group on the antisense strand is base paired with a nucleotide of a modified group on the sense strand.
- deoxyribonucleotides at an end of a strand of nucleic acid are not considered when determining a position of a modified group, i.e., the positional numbering begins with the first ribonucleotide or modified ribonucleotide.
- abasic nucleotides at an end of a strand of nucleic acid are not considered when determining a position of a modified group.
- a modified group comprises a 5 '-end nucleotide of either or both of the antisense strand and the sense strand.
- a flanking group comprises the 5 '-end nucleotide of either or both of the antisense strand and the sense strand.
- the 5 '-end nucleotide of either or both of the antisense strand and the sense strand is unmodified.
- a modified group comprises the 5 '-most nucleotide of either or both of the antisense duplex region and sense duplex region.
- a flanking group comprises the 5 '-most nucleotide of either or both of the antisense duplex region or the sense duplex region.
- the 5 '-most nucleotide of either or both of the antisense duplex region or the sense duplex region is unmodified.
- the nucleotide at position 10 of the antisense strand is unmodified.
- the nucleotide at position 10 of the sense strand is modified.
- a modified group comprises the nucleotide at position 10 of the sense strand.
- the modification at the 2' position is selected from the group comprising amino, fluoro, methoxy, alkoxy and Ci-C3-alkyl. In another embodiment, the modification is 2'-0-methyl.
- each modified group consists of one nucleotide and each flanking group consists of one nucleotide. In one embodiment, each modified group on the antisense strand is aligned with a flanking group on the sense strand. In another aspect, each modified group consists of one 2'-0-methyl modified nucleotide and each flanking group consists of one nucleotide. In one embodiment, each flanking group consists of one unmodified nucleotide. In one embodiment, each flanking group consists of one 2'-0-methyl modified nucleotide.
- each modified group on both the antisense strand and the sense strand consists of one 2'-0-methyl modified nucleotide and each flanking group on both the antisense strand and the sense strand consists of one nucleotide, wherein no modified group on one strand is either aligned or both aligned and base paired with another modified group on the other strand and no flanking group on one strand is either aligned or both aligned and base paired with a flanking group on the other strand.
- each modified group on each strand is either aligned or both aligned and based paired with a flanking group on the other strand.
- the flanking group is unmodified.
- the nucleotide of position one on the antisense strand is 2'-0-methyl modified.
- the 5 '-most nucleotide of the antisense duplex region is 2'-0-methyl modified.
- Modifications to phosphate backbone Another secondary aspect relates to modifications to a phosphate backbone.
- All or a portion of the nucleotides of the siRNA of the invention may be linked through phosphodiester bonds, as found in unmodified nucleic acid.
- a siRNA of the present invention may comprise a modified phosphodiester linkage.
- the phosphodiester linkages of either the antisense stand or the sense strand may be modified to independently include at least one heteroatom selected from the group consisting of nitrogen and sulfur.
- a phosphoester group connecting a ribonucleotide to an adjacent ribonucleotide is replaced by a modified group.
- the modified group replacing the phosphoester group is selected from the group consisting of phosphorothioate, methylphosphonate or phosphoramidate group.
- all of the nucleotides of the antisense strand are linked through phosphodiester bonds. In another embodiment, all of the nucleotides of the antisense duplex region are linked through phosphodiester bonds. In another embodiment, all of the nucleotides of the sense strand are linked through phosphodiester bonds. In another embodiment, all of the nucleotides of the sense duplex region are linked through phosphodiester bonds. In another embodiment, the antisense strand comprises a number of modified phosphoester groups selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10. In another embodiment, the antisense duplex region comprises a number of modified phosphoester groups selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10.
- the sense strand comprises a number of modified phosphoester groups selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10.
- the sense duplex region comprises a number of modified phosphoester groups selected from 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10.
- the siR A of the present invention may include nucleic acid molecules comprising one or more modified nucleotides, abasic nucleotides, acyclic or deoxyribonucleotide at the terminal 5 '- or 3 '-end on either or both of the sense or antisense strands.
- the 5'- and 3 '-end nucleotides of both the sense and antisense strands are unmodified.
- the 5 '-end nucleotide of the antisense strand is modified.
- the 5 '-end nucleotide of the sense strand is modified.
- the 3 '-end nucleotide of the antisense strand is modified. In another embodiment, the 3 '-end nucleotide of the sense strand is modified. In another embodiment, the 5 '-end nucleotide of the antisense strand and the 5 '-end nucleotide of the sense strand are modified. In another embodiment, the 3 '-end nucleotide of the antisense strand and the 3 '-end nucleotide of the sense strand are modified. In another embodiment, the 5 '-end nucleotide of the antisense strand and the 3 '-end nucleotide of the sense strand are modified.
- the 3 '-end nucleotide of the antisense strand and the 5 '-end nucleotide of the sense strand are modified.
- the 3 '-end nucleotide of the antisense strand and both the 5'- and 3 '-end nucleotides of the sense strand are modified.
- both the 5'- and 3 '-end nucleotides of the antisense strand are modified.
- both the 5'- and 3 '-end nucleotides of the sense strand are modified.
- the 5 '-end nucleotide of the antisense strand is phosphorylated. In another embodiment, the 5 '-end nucleotide of the sense strand is phosphorylated. In another embodiment, the 5 '-end nucleotides of both the antisense strand and the sense strand are phosphorylated. In another embodiment, the 5 '-end nucleotide of the antisense strand is phosphorylated and the 5 '-end nucleotide of the sense strand has a free hydroxyl group (5' -OH). In another embodiment, the 5 '-end nucleotide of the antisense strand is phosphorylated and the 5 '-end nucleotide of the sense strand is modified.
- Modifications to the 5'- and 3 '-end nucleotides are not limited to the 5' and 3 ' positions on these terminal nucleotides.
- modifications to end nucleotides include, but are not limited to, biotin, inverted (deoxy) abasics, amino, fluoro, chloro, bromo, CN, CF, methoxy, imidazole, caboxylate, thioate, Ci to C 10 lower alkyl, substituted lower alkyl, alkaryl or aralkyl, OCF 3 , OCN, 0-, S-, or N-alkyl; 0-, S-, or N-alkenyl; SO-CH3 ; S0 2 CH 3 ; ON0 2 ; N0 2 , N 3 ; heterozycloalkyl; heterozycloalkaryl; amino alky lamino; polyalkylamino or substituted silyl, as, among others, described, e.g., in PCT patent application WO
- alkyl means Ci-Ci 2 -alkyl and "lower alkyl” means Ci-C 6 -alkyl, including Ci-, C 2 -, C 3 -, C 4 -, C 5 - and C 6 -alkyl.
- the 5 '-end of the antisense strand, the 5 '- end of the sense strand, the 3 '-end of the antisense strand or the 3 '-end of the sense strand is covalently connected to a prodrug moiety.
- the moiety is cleaved in an endosome. In another the moiety is cleaved in the cytoplasm.
- siRNA of the present invention having different kinds of end modification(s) are presented in the following Table.
- the terminal 3' nucleotide or two terminal 3 '-nucleotides on either or both of the antisense strand or sense strand is a 2'-deoxynucleotide.
- the 2'-deoxynucleotide is a 2'-deoxy-pyrimidine.
- the 2'- deoxynucleotide is a 2' deoxy-thymidine.
- shRNA and linked siRNA Another aspect relates to shR A and linked siR A. It is within the present invention that the double-stranded structure is formed by two separate strands, i.e. the antisense strand and the sense strand. However, it is also with in the present invention that the antisense strand and the sense strand are covalently linked to each other. Such linkage may occur between any of the nucleotides forming the antisense strand and sense strand, respectively. Such linkage can be formed by covalent or non-covalent linkages. Covalent linkage may be formed by linking both strands one or several times and at one or several positions, respectively, by a compound preferably selected from the group comprising methylene blue and bifunctinoal groups.
- Such bifunctional groups are preferably selected from the group comprising bis(2-chloroethyl)amine, N-acetly-N'-(p- glyoxylbenzoyl)cystamine, 4-thiouracile and psoralene.
- the antisense strand and the sense strand are linked by a loop structure.
- of the loop structure is comprised of a non-nucleic acid polymer.
- the non-nucleic acid polymer is polyethylene glycol.
- the 5 ' -end of the antisense strand is linked to the 3 ' -terminus of the sense strand.
- the 3 ' -end of the antisense strand is linked to the 5 ' -end of the sense strand.
- the loop consists of a nucleic acid.
- locked nucleic acid LNA
- PNA peptide nucleic acid
- the nucleic acid is ribonucleic acid.
- the 5 '-terminus of the antisense strand is linked to the 3 '-terminus of the sense strand.
- the 3 '-end of the antisense strand is linked to the 5 '-terminus of the sense strand.
- the loop consists of a minimum length of four nucleotides or nucleotide analogues. In one embodiment, the loop consists of a length of nucleotides or nucleotide analogues selected from 4, 5, 6, 7, 8, 9, 10, 1 1, 12, 13, 14 or 15. In one embodiment, the length of the loop is sufficient for linking the two strands covalently in a manner that a back folding can occur through a loop structure or similar structure.
- the ribonucleic acid constructs may be incorporated into suitable vector systems.
- the vector comprises a promoter for the expression of RNAi.
- the respective promoter is pol III and more preferably the promoters are the U6, HI, 7SK promoter as described in Good et al. (1997) Gene Ther, 4, 45-54.
- the nucleic acid according to the present invention comprises a phosphorothioate internucleotide linkage.
- a phosphorothioate internucleotide linkage is within 5 nucleotides from the 3 '-end or the 5 '-end of either or both of the antisense strand and the sense strand.
- the antisense strand can comprise about one to about five phosphorothioate internucleotide linkages.
- an overhang at the 3 '-end of the sense strand is selected from consisting of 1, 2, 3, 4 and 5 nucleotides in length. In one embodiment, an overhang at the 5'- end of the antisense strand is selected from consisting of 1, 2, 3, 4 and 5 nucleotides in length. In one embodiment, an overhang at the 5 '-end of the sense strand is selected from consisting of 1, 2, 3, 4 and 5 nucleotides in length.
- the siRNA molecule is blunt-ended on both ends and has a length selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28 or 29 consecutive nucleotides.
- the siRNA molecule is blunt-ended on one end and the double stranded portion of the siRNA molecule has a length selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28 or 29 consecutive nucleotides.
- the siRNA molecule has overhangs on both ends and the double stranded portion of the siRNA molecule has a length selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28 or 29 consecutive nucleotides.
- the siRNA molecule comprises an overhang, said overhang comprising at least one deoxyribonucleotide. In one embodiment, the siRNA molecule comprises an overhang, said overhang comprising two deoxyribonucleotides.
- the siRNA molecule has overhangs on the 3 '-end of the antisense strand and at the 3 '-end of the sense strand, said overhangs comprising at least one deoxyribonucleotide. In one embodiment, the siRNA molecule has overhangs on the 3 '-end of the antisense strand and at the 3 '-end of the sense strand, said overhangs consisting two deoxyribonucleotides.
- nucleotide sequence As “even numbered” or “odd numbered”, such numbering starts from the 5' end of such nucleotide sequence.
- the nucleotide(s) forming the overhang may be (a) desoxyribonucleotide(s), (a) ribonucleotide(s) or a combination thereof.
- the antisense strand and/or the sense strand comprise a TT dinucleotide at the 3 ' end.
- the nucleic acid of the present invention can be produced using routine methods in the art including chemically synthesis or expressing the nucleic acid either in vitro (e.g., run off transcription) or in vivo.
- the siRNA is produced using solid phase chemical synthesis.
- the nucleic acid is produced using an expression vector.
- the expression vector produced the nucleic acid of the invention in the target cell. Accordingly, such vector can be used for the manufacture of a medicament. Methods for the synthesis of the nucleic acid molecule described herein are known to the ones skilled in the art.
- siRNA can be delivered to cells, both in vitro and in vivo, by a variety of methods known to those of skill in the art, including direct contact with cells ("naked” siRNA) or by in combination with one or more agents that facilitate targeting or delivery into cells.
- agents and methods include lipoplexes, liposomes, iontophoresis, hydrogels, cyclodextrins, nanocapsules, micro- and nanospheres and proteinaceous vectors (e.g., Bioconjugate Chem. (1999) 10: 1068-1074 and WO 00/53722).
- the nucleic acid/vehicle combination may be locally delivered in vivo by direct injection or by use of an infusion pump.
- the siRNA of the invention can be delivered in vivo by various means including intravenous subcutaneous, intramuscular or intradermal injection or inhalation.
- the molecules of the instant invention can be used as pharmaceutical agents.
- pharmaceutical agents prevent, modulate the occurrence, or treat (alleviate a symptom to some extent, preferably all of the symptoms) of a disease state in a subject.
- compositions comprising surface-modified liposomes containing poly (ethylene glycol) lipids (PEG-modified, or long-circulating liposomes or stealth liposomes).
- PEG-modified, or long-circulating liposomes or stealth liposomes offer a method for increasing stability of a liposome or lipoplex solutions by preventing their aggregation and fusion.
- the formulations also have the added benefit in vivo of resisting opsonization and elimination by the mononuclear phagocytic system (MPS or RES), thereby enabling longer blood circulation times and enhanced tissue exposure for the encapsulated drug.
- MPS or RES mononuclear phagocytic system
- liposomes have been shown to accumulate selectively in tumors, presumably by extravasation and capture in the neovascularized target tissues (Lasic et al, Science 1995, 267, 1275-1276; Oku et al, 1995, Biochim. Biophys. Acta, 1238, 86-90).
- the long-circulating liposomes enhance the pharmacokinetics and pharmacodynamics of DNA and RNA, particularly compared to conventional cationic liposomes which are known to accumulate in tissues of the MPS (Liu et al, J. Biol. Chem. 1995,42,24864-24780; Choi et al, International PCT Publication No.
- WO 96/10391 Ansell et al, International PCT Publication No. WO 96/10390; Holland et al, International PCT Publication No. WO 96/10392).
- Long-circulating liposomes also protect the siRNA from nuclease degradation.
- the siRNA of the present invention may be formulated as pharmaceutical compositions.
- the pharmaceutical compositions may be used as medicaments or as diagnostic agents, alone or in combination with other agents.
- one or more siRNAs of the invention can be combined with a delivery vehicle (e.g., liposomes) and excipients, such as carriers, diluents. Other agents such as preservatives and stabilizers can also be added.
- a delivery vehicle e.g., liposomes
- excipients such as carriers, diluents.
- Other agents such as preservatives and stabilizers can also be added.
- Methods for the delivery of nucleic acid molecules are known in the art and described, e.g., in Akhtar et al, 1992, Trends Cell Bio., 2, 139; Delivery Strategies for Antisense Oligonucleotide Therapeutics, ed. Akhtar, 1995, Maurer et al, 1999, Mol. Memb.
- siRNA of the present invention can also be administered in combination with other therapeutic compounds, either administrated separately or simultaneously, e.g., as a combined unit dose.
- the invention includes a pharmaceutical composition comprising one or more siRNA according to the present invention in a physio logically/pharmaceutically acceptable excipient, such as a stabilizer, preservative, diluent, buffer, and the like.
- a physio logically/pharmaceutically acceptable excipient such as a stabilizer, preservative, diluent, buffer, and the like.
- a unit dose contains between about 0.01 mg/kg and about 100 mg/kg body weight of siRNA.
- the dose of siRNA is about 10 mg/kg and about 25 mg/kg body weight.
- the dose of siRNA is about 1 mg/kg and about 10 mg/kg body weight.
- the dose of siRNA is about 0.05 mg/kg and about 5 mg/kg body weight.
- the dose of siRNA is about 0.1 mg/kg and about 5 mg/kg body weight.
- the dose of siRNA is about 0.1 mg/kg and about 1 mg/kg body weight.
- the dose of siRNA is about 0.1 mg/kg and about 0.5 mg/kg body weight.
- the dose of siRNA is about 0.5 mg/kg and about 1 mg/kg body weight.
- the pharmaceutical composition is a sterile injectable aqueous suspension or solution. In one aspect, the pharmaceutical composition is in lyophilized form. In one embodiment, the pharmaceutical composition comprises lyophilized lipoplexes, wherein the lipoplexes comprises a siRNA of the present invention. In another embodiment, the pharmaceutical composition comprises an aqueous suspension of lipoplexes, wherein the lipoplexes comprises a siRNA of the present invention.
- compositions and medicaments of the present invention may be administered to a subject (mammal) in the disclosed methods of treatment.
- the mammal is selected from the group consisting humans, dogs, cats, horses, cattle, pig, goat, sheep, mouse, rat, hamster and guinea pig.
- the mammal is a human.
- the mammal is a non-human mammal.
- the present invention is related to lipoplexes comprising a siRNA according to the present invention.
- lipoplexes consist of siRNA and liposomes.
- Such lipoplexes may be used to deliver the siR A of the invention to a target cell either in vitro or in vivo.
- the lipoplex has a zeta-potential of about 40 to 55 mV, preferably about 45 to 50 mV.
- the size of the lipoplex according to the present invention is about 80 to 200 nm, about 100 to 140 nm or about 110 nm to 130 nm, as determined by dynamic light scattering (QELS) such as, e. g., by using an N5 submicron particle size analyzer from Beckman Coulter according to the manufacturer's recommendation.
- QELS dynamic light scattering
- the liposome as forming part of the lipoplex is a positively charged liposome consisting of:
- the lipoplex and lipid composition forming the liposomes is preferably in a carrier however, the lipoplex can also be present in a lyophilised form.
- the lipid composition contained in a carrier usually forms a dispersion. More preferably, the carrier is an aqueous medium or aqueous solution as also further characterised herein.
- the lipid composition typically forms a liposome in the carrier, whereby such liposome preferably also contains the carrier inside.
- the lipid composition contained in the carrier and the carrier, respectively, preferably has an osmolality of about 50 to 600 mosmole/kg, preferably about 250 - 350 mosmole/kg, and more preferably about 280 to 320 mosmole/kg.
- the liposomes preferably are formed by the first lipid component and optionally also by the first helper lipid, preferably in combination with the first lipid component, preferably exhibit a particle size of about 20 to 200 nm, preferably about 30 to 100 nm, and more preferably about 40 to 80 nm. It is noted that the size of the particles follows a certain statistical distribution.
- a further optional feature of the lipid composition in accordance with the present invention is that the pH of the carrier is preferably from about 4.0 to 6.0. However, also other pH ranges such as from 4.5 to 8.0, preferably from about 5.5 to 7.5 and more preferably about 6.0 to 7.0 are within the present invention.
- the lipid composition of the present invention may comprise one or several of the following sugars: sucrose, trehalose, glucose, galactose, mannose, maltose, lactulose, inulin and raffinose, whereby sucrose, trehalose, inulin and raffinose are particularly preferred.
- the osmolality mostly adjusted by the addition of sugar is about 300 mosmole/kg which corresponds to a sucrose solution of 270 mM or a glucose solution of 280 mM.
- the carrier is isotonic to the body fluid into which such lipid composition is to be administered.
- the term that the osmolality is mostly adjusted by the addition of sugar means that at least about 80 %, preferably at least about 90 % of the osmolality is provided by said sugar or a combination of said sugars.
- the pH of the lipid composition of the present invention is adjusted, this is done by using buffer substances which, as such, are basically known to the one skilled in the art.
- buffer substances which, as such, are basically known to the one skilled in the art.
- basic substances are used which are suitable to compensate for the basic characteristics of the cationic lipids and more specifically of the ammonium group of the cationic head group.
- the particle size of such lipid composition and the liposomes formed by such lipid composition is preferably determined by dynamic light scattering such as by using an N5 submicron particle size analyzer from Beckman Coulter according to the manufacturer's recommendation.
- the lipid composition contains one or several nucleic acid(s), such lipid composition usually forms a lipoplex (liposome-nucleic acid complex).
- the more preferred concentration of the overall lipid content in the lipoplex in preferably isotonic 270 mM sucrose or 280 mM glucose is from about 0.01 to 100 mg/ml, preferably 0.01 to 40 mg/ml and more preferably 0.01 to 25 mg/ml. It is to be acknowledged that this concentration can be increased so as to prepare a reasonable stock, typically by a factor of 2 to 3. It is also within the present invention that based on this, a dilution is prepared, whereby such dilution is typically made such that the osmolality is within the range specified above.
- the dilution is prepared in a carrier which is identical or in terms of function and more specifically osmolality similar to the carrier used in connection with the lipid composition or in which the lipid composition is contained.
- the lipid composition of the present invention whereby the lipid composition also comprises a nucleic acid, preferably a functional nucleic acid such as, but not limited to, a si NA
- the concentration of the functional nucleic acid, preferably of siRNA in the lipid composition is about 0.2 to 0.4 mg/ml, preferably 0.28 mg/ml
- the total lipid concentration is about 1.5 to 2.7 mg/ml, preferably 2.17 mg/ml.
- this mass ratio between the nucleic acid fraction and the lipid fraction is particularly preferred, also with regard to the charge ratio thus realized.
- the mass ratio and the charge ratio, respectively, realized in this particular embodiment is preferably maintained despite such concentration or dilution.
- Such concentration as used in, for example, a pharmaceutical composition can be either obtained by dispersing the lipid in a suitable amount of medium, preferably a physiologically acceptable buffer or any carrier described herein, or can be concentrated by appropriate means.
- appropriate means are, for example, ultra filtration methods including cross-flow ultra- filtration.
- the filter membrane may exhibit a pore width of 1,000 to 300,000 Da molecular weight cut-off (MWCO) or 5 nm to 1 ⁇ . Preferred is a pore width of about 10,000 to 100,000 Da MWCO. It will also be acknowledged by the one skilled in the art that the lipid composition more specifically the lipoplexes in accordance with the present invention may be present in a lyophilized form.
- Such lyophilized form is typically suitable to increase the shelve life of a lipoplex.
- the sugar added, among others, to provide for the appropriate osmolality is used in connection therewith as a cryo -protectant.
- the aforementioned characteristics of osmolality, pH as well as lipoplex concentration refers to the dissolved, suspended or dispersed form of the lipid composition in a carrier, whereby such carrier is in principle any carrier described herein and typically an aqueous carrier such as water or a physiologically acceptable buffer, preferably an isotonic buffer or isotonic solution.
- the delivery system described therein is a composition comprising a lipid composition, wherein the lipid composition consists of
- Y " is an anion, wherein each of Rl and R2 is individually and independently selected from the group consisting of linear C 12-C 18 alkyl and linear C 12-C 18 alkenyl; a sterol compound, wherein the sterol compound is selected from the group consisting of cholesterol and stigmasterol; and a PEGylated lipid, wherein the PEGylated lipid comprises a PEG moiety and wherein the PEGylated lipid is selected from the group consisting of a PEGylated phosphoethanolamine of formula (II)
- each of R3 and R4 is individually and independently linear C13-C17 alkyl, and p is any integer from 15 to 130; a PEGylated ceramide of formula (III)
- R5 is linear C7-C15 alkyl, and q is any integer from 15 to 130;
- each of R6 and R7 is individually and independently linear CI 1-C17 alkyl, and r is any integer from 15 to 130.
- One aspect of the present invention provides a siRNA molecule that reduces the expression of EDNl and that is useful for the treatment of human diseases and pathological conditions.
- the siRNA molecules can be used in combination with other therapeutic agents to enhance the therapeutic effects of a given treatment modality.
- the present invention provides reagents and methods useful for treating diseases and conditions characterized by undesirable or aberrant levels of EDNl activity in a cell.
- Another aspect of the present invention is using the materials and methods for the treatment of conditions which can be ameliorated by decreasing constriction and constriction of vascular endothelial cells in particular.
- Another aspect of the present invention includes treating pulmonary hypertension.
- a still other aspect of the present invention includes treating pre-eclampsia.
- Endothelial MS-1 cells were obtained from ATCC/LGC Promochem and cultivated according to the manufacturer ' s recommendations.
- HUVECs (Lonza) were cultured in EGM- 2 bullet kit medium containing EBM2+ SingleQuots growth supplements.
- Endothelin 1 protein level in supernatant of cell cultures was determined by ELISA 72 hours post infection.
- Atuplex is a lipid composition containing
- DACC9 is a lipid composition containing
- DACC10 is a lipid composition containing
- Normoxic animals were maintained at 21% 0 2 throughout. Hypoxic animals were exposed to 10% 02 in a normobaric chamber and administered the vascular endothelial growth factor (VEGF) receptor antagonist Sugen 5416 (suspension in 0.5%> [w/v] carboxymethylcellulose sodium, 0.9%> [w/v] sodium chloride, 0.4%> [v/v] polysorbate 80, 0.9%) [v/v] benzyl alcohol in deionized water; 20mg/kg, s.c, lx/week for 3 weeks).
- VEGF vascular endothelial growth factor
- siRNA/vehicle treatments were given lx or 2x a week by tail vein injection, Bosentan (suspension in 0.1 % [w/v] carboxymethylcellulose sodium, polysorbate 80, 0.5%> [v/v]) was administered as daily gavage. Haemodynamic measurements were carried out within 72 hours of the last drug intervention.
- RVSP right ventricular systolic pressure
- MABP mean arterial blood pressure
- RVSP right jugular vein was isolated and a Millar micro -manometer tipped catheter (Millar MicroTip 1.4F catheter, Millar Instruments, Houston, Texas, USA) introduced into the superior vena cava and then advanced into the right ventricle. Both MABP and RVSP were recorded onto a pre-calibrated PowerLab system (ADInstruments, Castle Hill, New South Wales, Australia) running Chart 5 software. Following measurement of pulmonary and systemic haemodynamics, animals were sacrificed by anaesthetic overdose and exsanguination, blood collected, the heart removed, and heart chamber weights measured to evaluate right ventricular hypertrophy (right ventricle to body weight ratio; RV/BW).
- RV/BW right ventricular hypertrophy
- the lung was fixed by inflation with 10% formalin in PBS before paraffin embedding and sectioning.
- the remaining lung tissue, heart, liver and kidney were dissected and snap frozen in liquid N2.
- Serum was collected by allowing the blood to stand at room temperature for 45 minutes, then spinning at 4500rpm for 10 minutes. Serum was collected & stored at - 80°C.
- Results are expressed as mean ⁇ sem. Statistical analyses were performed by one-way ANOVA, with Bonferroni post-hoc tests, using GraphPad Prism version 5. P ⁇ 0.05 denotes significance. The n value denotes the number of animals in each group.
- siRNA molecules which are directed to the mRNA encoding EDN1 and the various siRNA molecules directed to Luciferase and PTEN which were used in connection with the experiments and examples described herein, were synthesized by BioSpring (Frankfurt a. M., Germany) and are indicated in Fig. 1, Table 1 and Table 2 in terms of the sequences of both the first strand (being the antisense strand) and the second strand (being the sense strand) forming the double-stranded nucleic acid molecules of the present invention.
- Luciferase-specific siRNA was formed by the following two strands:
- siRNA PTENV10 was formed by the following two strands:
- PTENV10 A 5 ' UaAgUuCuAgCuGuGgUgGgUuA 3' (SEQ ID NO: 20)
- PTENV10B 5' uAaCcCaCcAcAgCuAgAaCuCa 3' (SEQ ID NO: 21)
- A stands for the antisense strand which is also referred to herein as the first strand
- B stands for the sense strand which is also referred to herein as the second strand.
- the antisense strands can be modified at the 2' position (e.g., with a 2'-0-methyl group) on one or more odd numbered nucleotide (or on each odd numbered nucleotide) and one or more even numbered nucleotides remain unmodified (e.g., a OH group is present at the 2' position on each of the unmodified nucleotides, for example each of the unmodified nucleotides is unmodified).
- Sense strands can be modified on one or more even numbered nucleotide (or on each even numbered nucleotide) at the 2' position (e.g., with a 2'-0-methyl group) and one or more odd numbered nucleotides can remain unmodified (e.g., a OH group is present at the 2' position on each of the unmodified nucleotides, for example each of the odd numbered nucleotides is unmodified).
- Alternative embodiments provide for antisense strands (as set forth above) that are modified at the 2' position (e.g., with a 2'-0-methyl group) on one or more even numbered nucleotide (or on each even numbered nucleotide) and sense strands are modified on one or more odd numbered nucleotide (or on each odd numbered nucleotide) at the 2' position (e.g., with a 2'-0-methyl group).
- One or more unmodified nucleotide is present in both the sense and antisense strands in these alternative embodiments (e.g., the unmodified nucleotides have a OH group at the 2' position in each of these alternative embodiments).
- each odd numbered nucleotide is unmodified in the antisense strand and each even numbered nucleotide is unmodified in the sense strand for the alternative embodiments discussed in this paragraph.
- siRNA molecules of Fig. 1 were tested in vitro for inhibition of EDN1 target expression in cell cultures using degradation of mEDNl mRNA and PTEN mRNA as readout. The results are indicated in Fig. 2A and Fig. 2B.
- siRNA consisting of EDNl-hmr-2A and EDNl-hmr-2B was particularly effective, whereby siRNA consisting of EDNl-hm4A and EDNl-hm4B, siRNA consisting of of EDNl-hmr6A and EDNl-hmr-6B, and siRNA consisting of EDNl-hmr-8A and EDNl-hmr-8B also exhibited some efficiency.
- siRNA molecules of Fig. 1 were also tested in vitro for inhibition of EDN1 target expression in cell cultures using endothelin 1 protein concentration in the supernatant of cell cultures of murine MSI cells and human HuVe cells as read-out. The results are indicated in Figs. 3 and 4. The results as to particularly effective siRNA molecules as shown in connection with Fig. 2 was confirmed.
- Example 4 Animal studies - reduction of EDN-1 expression in mice
- mice The purpose of these animal studies was to evaluate different delivery systems for targeting EDN1.
- the experimental set-up and treatment scheme for the mice is outlined in Fig. 5.
- mice were treated with a single dose of EDNl-hmr2 siRNA (consisting of EDNl-hmr 2A and EDNl-hmr-2B) formulated with DACC9, DACC10 or with three doses of EDN1- hmr2 siRNA formulated with Atuplex by bolus application.
- Control lipoplexes contained siRNA for SFltl .
- Target gene expression was analyzed in lung tissues and in serum 48 hours post treatment.
- Example 5 Animal studies - effect of DACC9 formulated siRNA targeting EDN1 in hypoxia-induced pulmonary hypertension The purpose of these animal studies was to show therapeutic effect of DACC9 formulated siRNA targeting EDN1 in hypoxia- induced pulmonary hypertension.
- the siRNA used was EDNl-hmr2.
- the DACC9 formulated siRNA composition is also referred to herein as Dacc9/EDN1.
- RVSP right ventricular systolic pressure
- RVSP right ventricular hypertrophy
- RVH hypoxia induced a significant increase in RVH (normoxia: 0.09 ⁇ 003 g vs. hypoxia: 0.113 ⁇ 003 g).
- Dacc9/EDN1 groups As well as the Bosentan-treated animals (Figs. 10 and 11).
- this index of disease severity was more greatly affected by the low dose formulation compared to the high dose siRNA. Since treatment with Sugen causes a drop in body weight (BW) over the time-course of the study (data not shown), RVH is expressed as RV:BW ratio to correct for this.
- Dacc9/EDN1 is an effective intervention for the prevention of hypoxia-induced PH.
- the efficacy of Dacc9/EDN1 is approximately equivalent to Bosentan.
- Dacc9/EDN1 produced a pulmonary-selective effect since MABP remained unchanged.
- mice Male C57BL/6 mice (Harlan; 8-10 weeks) were randomly assigned to one of 6
- Normoxic animals were maintained at 21% 02 throughout. Hypoxic animals were exposed to 10% 02 in a normobaric chamber and administered the vascular endothelial growth factor (VEGF) receptor antagonist Sugen 5416 (suspension in 0.5%> [w/v] carboxymethylcellulose sodium, 0.9%) [w/v] sodium chloride, 0.4%> [v/v] polysorbate 80, 0.9%> [v/v] benzyl alcohol in deionized water; 20mg/kg, s.c, lx/week for 3 weeks). siRNA (2mg/kg) treatment and vehicle (sucrose) were initiated at day 14 once the PH phenotype had established ('reversal protocol') and administered 2x a week by tail vein injection. Bosentan (suspension in 0.1% [w/v] carboxymethylcellulose sodium, polysorbate 80, 0.5%> [v/v]) was administered as daily gavage. Haemodynamic measurements were carried out within 72 hours of the last drug intervention.
- RVSP right ventricular systolic pressure
- MABP mean arterial blood pressure
- RVSP right jugular vein was isolated and a Millar micro -manometer tipped catheter (Millar MicroTip 1.4F catheter, Millar Instruments, Houston, Texas, USA) introduced into the superior vena cava and then advanced into the right ventricle. Both MABP and RVSP were recorded onto a pre-calibrated PowerLab system (ADInstruments,
- RVH right ventricular hypertrophy
- RV/[LV+S] right ventricular hypertrophy
- the lung was fixed by inflation with 10% formalin in PBS before paraffin embedding and sectioning.
- the remaining lung tissue, liver and kidney were dissected and snap frozen in liquid N2.
- Whole blood samples were collected in EDTA tubes at baseline (tail vein; 3 animals per group) and at day 35 (cardiac puncture; all animals) and plasma obtained by spinning at 4500rpm for 10 min at 4oC. Plasma was separated and stored at -80oC.
- RVSP Hypoxia induced a significant increase in RVSP (normoxia: 22.75 ⁇ 1.5mmHg vs. hypoxia: 39.83 ⁇ 2.2 mmHg) that was significantly reversed in the presence of Dacc9/EDN1 and Bosentan (Figure 15).
- the Dacc9/Luc control had no effect on RVSP, and whilst the modified siRNA Dacc9/EDNl-flu showed a trend towards a reduction in RVSP this did not reach statistical significance.
- RVH right ventricular hypertrophy
- EDNl mRNA expression levels in lung tissue were determined after the end of the treatment by qRT-PCR. It has been showed that EDNl mRNA levels are elevated by hypoxia/Sugen treatment, but reduced in DACC9/EDN1 treatment groups. It has been surprisingly found by the inventors that Dacc9/EDN1 is an effective intervention for the reversal of hypoxia-induced PH. The treatment caused a significant reduction in the development of elevated pulmonary artery pressure (i.e. RVSP) and also abrogated the accompanying RVH. The efficacy of Dacc9/EDN1 is approximately equivalent to Bosentan at the dose employed, with a superior activity against the hypoxia-induced increases in RVSP and RVH. The mRNA knockdown of EDN1 by Dacc9/EDN1 produced a pulmonary-selective effect since MABP remained unchanged.
- RVSP elevated pulmonary artery pressure
- siRNA-lipoplexes for cancer therapy.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Animal Behavior & Ethology (AREA)
- Genetics & Genomics (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Wood Science & Technology (AREA)
- Biochemistry (AREA)
- Organic Chemistry (AREA)
- Zoology (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Plant Pathology (AREA)
- Microbiology (AREA)
- Dermatology (AREA)
- Biophysics (AREA)
- Dispersion Chemistry (AREA)
- Physics & Mathematics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The present invention is related to an siRNA comprising an antisense strand and a sense strand where at least a portion of the antisense strand is complementary to a portion of the EDN-1 gene, compositions and formulations of the siRNA with lipid delivery systems and uses thereof.
Description
MEANS FOR INHIBITING THE EXPRESSION OF EDNl
FIELD OF THE INVENTION
The present invention is related to a double-stranded nucleic acid suitable to inhibit the expression of EDNl, compositions, pharmaceutical formulations and uses thereof.
BACKGROUND OF INVENTION
Endothelin 1, also known as preproendothelin-1 (PPET1), is a protein that in humans is encoded by the EDNl gene. The protein encoded by this gene is proteolytically processed to release a secreted peptide termed endothelin 1. This peptide is a potent vasoconstrictor and is produced by vascular endothelial cells. Endothelin 1 is one of three isoforms of human endothelin (ET-1).
Preproendothelin is precursor of the peptide ET-1. Endothelial cells convert preproendothelin to proendothelin and subsequently to mature endothelin, which the cells release.
Endothelin- 1 receptor antagonists (Bosentan) are used in the treatment of pulmonary hypertension. Inhibition of these receptors prevents pulmonary vasculature constriction and thus decreases pulmonary vascular resistance.
There is an ongoing need in the art for means of silencing or knocking down the expression levels of EDNl in vitro and in vivo, including the use of siRNA for the treatment of lung diseases and pulmonary hypertension. The present invention addresses these unmet needs through discovery of compositions, methods of using and processes of making siRNA directed to ADN1. SUMMARY OF THE INVENTION
The present invention relates to compositions comprising short interfering RNA (siRNA) directed to an expressed RNA transcript of EDNl (sometimes referred to as a "target nucleic acid" herein) and compositions thereof. These siRNA molecules can be used in the treatment of a variety of diseases and disorders where reduced expression of EDNl gene product is desirable.
BRIEF DESCRIPTION OF DRAWINGS
Further features, embodiments and advantages may be taken from the following figures:
Fig. 1 is a summary of siRNA molecules tested in vitro for inhibition of EDNl target gene expression; nucleotides with 2'-0-methyl are marked bold; nucleotides with 2'-F are underlined.
Fig. 2A is a bar diagram showing the efficacy of different EDNl -specific siRNA molecules on mEDNl mRNA degradation in murine MSI cells; knock-down of mEDNl mRNA is shown in comparison to untreated murine cells or to cells transfected with PTEN (PTENVlO)-specific siRNA and luciferase-specific siRNA being used as negative controls.
Fig. 2B is a bar diagram showing the efficacy of different EDNl -specific siRNA molecules on PTEN mRNA in murine MSI cells; PTEN mRNA levels are depicted in cells transfected with EDNl siRNAs in comparison to untreated murine cells or cells transfected with luciferase-specific siRNA or PTEN siRNA (PTENVIO) being used as negative controls and positive control, respectively..
Fig. 3 is a bar diagram showing the content of endothelin 1 (pg/ml) in supernatant of murine MSI cells 72 hours after transfection with different EDNl-specific siRNA molecules.
Fig. 4 is a bar diagram showing the content of endothelin 1 (pg/ml) in supernatant of human Human umbilical vein endothelial cells (HuVeCs) 72 hours after transfection with different EDNl-specific siRNA molecules.
Fig. 5 is a summary outlining the experimental set-up for determining reduction of EDNl expression in mice using different siRNA molecules targeting either EDNl or soluble VEGF receptor 1 (sFltl) and different delivery systems.
Fig. 6A is a bar diagram showing EDNl mRNA expression in total lysates of lung tissue from mice treated according to the experimental set-up illustrated in Fig. 5; EDNl mRNA expression is normalized relative to PTEN mRNA.
Fig. 6B is a bar diagram showing relative endothelin 1 protein level in serum from mice treated according to the experimental set-up illustrated in Fig. 5; endothelin 1 protein level is normalized relative to buffer treated group.
Fig. 7 is a scheme outlining the experimental set-up of an in vivo study in mC57/BL6 mice, 6 - 10 weeks, (n=10) using different formulations including lipoplexes of DACC9 comprising EDNl -targeting siRNA.
Fig. 8 is a bar diagram showing the result of the in vivo study described in Fig. 7.
Depicted is the average right ventricular systolic pressure (RVSP) in the different treatment group at the end of the experiment
Fig. 9 is a bar diagram showing the result of the in vivo study described in Fig. 7 using mean arterial blood pressure (MABP) as endpoint measurement.
Fig. 10 is a bar diagram showing the result of the in vivo study described in Fig. 7 using right ventricular hypertrophy (expressed as right ventricle weight/body weight) as endpoint measurement.
Fig. 11 is a bar diagram showing the result of the in vivo study described in Fig. 7 using right ventricular hypertrophy (expressed as right ventricle weight/body weight) as endpoint measurement indicating the values obtained from each animal tested.
Fig. 12 - Additional EDN1 siRNAs:- Nucleotides with 2' OMe modification are depicted in bold, nucleotides with 2'fluoro modification are underlined.
Fig. 13 Inhibition of EDN1 target gene expression by EDN1 siRNAs in HuVecs (human umbilicord vein endothelial cells). Cells were transfected with 20 nM siRNAs and lmg/ml AtuFECT. Target gene expression (mRNA) was assessed by qRT-PCR two days after transfection. PTEN is a house keeping gene. EDN1 expression levels were normalized to PTEN expression levels. Luc-l-f is a non targeting control siRNA.
Fig. 14 - is a scheme outlining the experimental set-up of the treatment groups in an intervention study.
Fig.15 -Right ventricular systolic pressure (RVSP) in normoxic mice and animals exposed to hypoxia (10% 02, 5 weeks) plus sugen (20mg/kg at day 0, 7 & 14) in the absence and presence of Dacc9/luc (2X/week), DACC9/EDN 1 -fl (2X/week), DACC9/EDN 1
(2X/week) or Bosentan (lOOmg/kg/day; p.o.). Interventions were started at day 14. Data are expressed as mean±sem. # # # P<0.001 vs normoxia; ***P<0.001 vs hypoxia + sugen; n=9- 10.
Fig. 16 - Right ventricular hypertrophy (right ventricle: left ventricle+septum, RV/[LV+S], ratio) in normoxic mice and animals exposed to hypoxia (10% 02, 5 weeks) plus sugen (20mg/kg at day 0, 7 & 14) in the absence and presence of Dacc9/luc (2X/week), Dacc9/EDNl-fl (2X/week), Dacc9/edn (2X/week) or Bosentan (lOOmg/kg/day; p.o.).
Interventions were started at day 14. Data are expressed as mean±sem. # #P<0.01 vs normoxia; *P<0.05 vs hypoxia + sugen; n=10.
Fig. 17 - Mean arterial blood pressure (MABP) in normoxic mice and animals exposed to hypoxia (10%> 02, 5 weeks) plus sugen (20mg/kg at day 0, 7 & 14) in the absence and presence of Dacc9/luc (2/week), Dacc9/flu (2/week), Dacc9/edn (2/week) or Bosentan
(lOOmg/kg/day; p.o.). Interventions were started at day 14. Data are expressed as mean±sem. n=8-10.
Fig. 18 - EDN1 mR A expression levels in lung tissue were determined after the end of the treatment by qRT-PCR.
Fig. 19 - Change in body weight in normoxic mice and in animals exposed to hypoxia (10% 02, 5 weeks) plus Sugen (20mg/kg at day 0, 7 & 14) in the absence and presence of Dacc9/luc (2X/week), Dacc9/EDNlfi (2X/week), Dacc9/EDN1 (2X/week) or Bosentan (lOOmg/kg/day; p.o.). Interventions were started at day 14. Data are expressed as mean±sem. n=10.
BRIEF DESCRIPTION OF THE SEQUENCES
SEQ ID NOs: 1 to 16 are strands of different siRNAs targeting human EDN1.
SEQ ID NO: 17 is the mRNA sequence encoding human EDN1. This sequence has been taken from GenBank Accession number NM 001955.4. which is incorporated herein by reference in its entirety.
SEQ ID NOs: 18 and 19 are the two strands of an siRNA targeting luciferase.
SEQ ID NOs: 20 and 21 are the two strands of an siRNA targeting PTEN.
SEQ ID NOs: 22 to 45 are further strands of different double stranded siRNAs targeting human EDN1
Table 1 - Sequences with modifications. Nucleotides with 2' OMe modification are depicted in bold, nucleotides with 2'fluoro modification are underlined.
SEQ ID No. Unmodified sequence
EDNl-hm-lA 5 ' auauuuaacaugguaaaua 3' SEQ ID NO: 9 5 ' auauuuaacaugguaaaua 3 ' EDNl-hm-lB 5 ' uauuuaccauguuaaauau 3' SEQ ID NO: 10 5 ' uauuuaccauguuaaauau 3'
EDNl-hmr-2A 5 ' uuucaauuugugcauuccu 3' SEQ ID NO:l 5 ' uuucaauuugugcauuccu 3 ' EDNl-hmr-2B 5 ' aggaaugcacaaauugaaa 3 ' SEQ ID NO: 2 5 'aggaaugcacaaauugaaa 3'
EDNl-hm-3A 5 ' aaaauauuuauuuucuaaa 3' SEQ ID NO: 11 5 ' aaaauauuuauuuucuaaa 3 ' EDNl-hm-3B 5 ' uuuagaaaauaaauauuuu 3' SEQ ID NO: 12 5 ' uuuagaaaauaaauauuuu 3'
EDNl-hm-4A 5 ' auacauagaccaauauggc 3' SEQ ID NO: 3 5 ' auacauagaccaauauggc 3 ' EDNl-hm-4B 5 ' gccauauuggucuauguau 3' SEQ ID NO: 4 5 ' gccauauuggucuauguau 3'
EDNl-hmr-5A 5 ' aaauuccagcacuucuugu 3' SEQ ID NO: 13 5 ' aaauuccagcacuucuugu 3 ' EDNl-hmr-5B 5 ' acaagaagugcuggaauuu 3' SEQ ID NO: 14 5 ' acaagaagugcuggaauuu 3 '
Table 2 - Additional examples of sequences with modifications. Nucleotides with 2' OMe modification are depicted in bold, nucleotides with 2'fluoro modification are underlined.
SEQ ID NO: Unmodified sequence
SEQ ID NO. 22 EDN1-hc-9A-fluoro 5 ' aaguaaauucuccaaggcu 3' 5 ' aaguaaauucuccaaggcu 3 ' SEQ ID NO. 23 EDN1-hc-9B-fluoro 5 ' agccuuggagaauuuacuu 3' 5 ' agccuuggagaauuuacuu 3 '
SEQ ID NO. 24 EDN1-hc-10A-fluoro 5 'uauuauaucuacugcugac 3' 5 ' uauuauaucuacugcugac 3' SEQ ID NO. 25 EDN1-hc-10B-fluoro 5 ' gucagcaguagauauaaua 3' 5 ' gucagcaguagauauaaua 3'
SEQ ID NO. 26 EDN1-hc-1 1A-fluoro 5 'uuauccaucagggacgagc 3' 5 ' uuauccaucagggacgagc 3' SEQ ID NO. 27 EDN1-hc-1 1 B-fluoro 5 ' gcucgucccugauggauaa 3' 5 ' gcucgucccugauggauaa 3'
SEQ ID NO. 28 EDN1-hc-12A-fluoro 5 ' cauuagaccagacuucuac 3' 5 ' cauuagaccagacuucuac 3 ' SEQ ID NO. 29 EDN1-hc-12B-fluoro 5 ' guagaagucuggucuaaug 3' 5 ' guagaagucuggucuaaug 3'
SEQ ID NO. 30 EDN1-hc-13A-fluoro 5 ' acacucuuuauccaucagg 3' 5 ' acacucuuuauccaucagg 3 ' SEQ ID NO. 31 EDN1-hc-13B-fluoro 5 ' ccugauggauaaagagugu 3' 5 ' ccugauggauaaagagugu 3 '
SEQ ID NO. 32 EDN1-hc-14A-fluoro 5 ' ugcauguuugucuuuugag 3' 5 ' ugcauguuugucuuuugag 3' SEQ ID NO. 33 EDN1-hc-14B-fluoro 5 ' cucaaaagacaaacaugca 3' 5 ' cucaaaagacaaacaugca 3 '
SEQ ID NO. 34 EDN1-hc-15A-fluoro 5 ' acuucuacgaggcuaggau 3' 5 ' acuucuacgaggcuaggau 3 ' SEQ ID NO. 35 EDN1-hc-15B-fluoro 5 ' auccuagccucguagaagu 3' 5 ' auccuagccucguagaagu 3 '
SEQ ID NO. 36 EDN1-hc-16A-fluoro 5 ' acauagaccaauauggccu 3' 5 ' acauagaccaauauggccu 3 ' SEQ ID NO. 37 EDN1-hc-16B-fluoro 5 ' aggccauauuggucuaugu 3' 5 ' aggccauauuggucuaugu 3 '
SEQ ID NO. 38 EDN1-hc-17A-fluoro 5 ' aaagucauuaccuugacag 3' 5 ' aaagucauuaccuugacag 3 '
SEQ ID NO. 39 EDN1-hc-17B-fluoro 5 ' cugucaagguaaugacuuu 3' 5 ' cugucaagguaaugacuuu 3 '
SEQ ID NO. 40 EDN1-hc-18A-fluoro 5 ' augugcucgguuguggguc 3' 5 ' augugcucgguuguggguc 3 ' SEQ ID NO. 41 EDN1-hc-18B-fluoro 5 ' gacccacaaccgagcacau 3' 5 ' gacccacaaccgagcacau 3'
SEQ ID NO. 42 EDN1-hc-19A-fluoro 5 ' cuguuucucauggucuccg 3' 5 ' cuguuucucauggucuccg 3 ' SEQ ID NO. 43 EDN1-hc-19B-fluoro 5 ' cggagaccaugagaaacag 3' 5 ' cggagaccaugagaaacag 3 '
SEQ ID NO. 44 EDN1-hc-20A-fluoro 5 ucugucaccaaugugcucg 3' 5 ' ucugucaccaaugugcucg 3' SEQ ID NO. 45 EDN1-hc-20B-fluoro 5 ' cgagcacauuggugacaga 3' 5 ' cgagcacauuggugacaga 3 '
Sequence Id no: 17 - mRNA sequence encoding human EDNl
ggagcuguuu acccccacuc uaauaggggu ucaauauaaa aagccggcag agagcugucc
aagucagacg cgccucugca ucugcgccag gcgaacgggu ccugcgccuc cugcaguccc
agcucuccac cgccgcgugc gccugcagac gcuccgcucg cugccuucuc uccuggcagg
cgcugccuuu ucuccccguu aaaagggcac uugggcugaa ggaucgcuuu gagaucugag
gaacccgcag cgcuuugagg gaccugaagc uguuuuucuu cguuuuccuu uggguucagu
uugaacggga gguuuuugau cccuuuuuuu cagaauggau uauuugcuca ugauuuucuc
ucugcuguuu guggcuugcc aaggagcucc agaaacagca gucuuaggcg cugagcucag
cgcggugggu gagaacggcg gggagaaacc cacucccagu ccacccuggc ggcuccgccg
guccaagcgc ugcuccugcu cgucccugau ggauaaagag ugugucuacu ucugccaccu
ggacaucauu ugggucaaca cucccgagca cguuguuccg uauggacuug gaagcccuag
guccaagaga gccuuggaga auuuacuucc cacaaaggca acagaccgug aaaauagaug
ccaaugugcu agccaaaaag acaagaagug cuggaauuuu ugccaagcag gaaaagaacu
cagggcugaa gacauuaugg agaaagacug gaauaaucau aagaaaggaa aagacuguuc
caagcuuggg aaaaagugua uuuaucagca guuagugaga ggaagaaaaa ucagaagaag
uucagaggaa caccuaagac aaaccagguc ggagaccaug agaaacagcg ucaaaucauc
uuuucaugau cccaagcuga aaggcaagcc cuccagagag cguuauguga cccacaaccg
agcacauugg ugacagaccu ucggggccug ucugaagcca uagccuccac ggagagcccu
guggccgacu cugcacucuc cacccuggcu gggaucagag caggagcauc cucugcuggu
uccugacugg caaaggacca gcguccucgu ucaaaacauu ccaagaaagg uuaaggaguu
cccccaacca ucuucacugg cuuccaucag ugguaacugc uuuggucucu ucuuucaucu
ggggaugaca auggaccucu cagcagaaac acacagucac auucgaauuc ggguggcauc
cuccggagag agagagagga aggagauucc acacaggggu ggaguuucug acgaaggucc
uaagggagug uuugugucug acucaggcgc cuggcacauu ucagggagaa acuccaaagu
ccacacaaag auuuucuaag gaaugcacaa auugaaaaca cacucaaaag acaaacaugc
aaguaaagaa aaaaaaaaga aagacuuuug uuuaaauuug uaaaaugcaa aacugaauga
aacuguuacu accauaaauc aggauauguu ucaugaauau gagucuaccu caccuauauu
gcacucuggc agaaguauuu cccacauuua auuauugccu ccccaaacuc uucccacccc
ugcugccccu uccuccaucc cccauacuaa auccuagccu cguagaaguc uggucuaaug
ugucagcagu agauauaaua uuuucauggu aaucuacuag cucugaucca uaagaaaaaa
aagaucauua aaucaggaga uucccugucc uugauuuuug gagacacaau gguauagggu
uguuuaugaa auauauugaa aaguaagugu uuguuacgcu uuaaagcagu aaaauuauuu
uccuuuauau aaccggcuaa ugaaagaggu uggauugaau uuugauguac uuauuuuuuu
auagauauuu auauucaaac aauuuauucc uuauauuuac cauguuaaau aucuguuugg
gcaggccaua uuggucuaug uauuuuuaaa auauguauuu cuaaaugaaa uugagaacau
gcuuuguuuu gccugucaag guaaugacuu uagaaaauaa auauuuuuuu ccuuacugua
DETAILED DESCRIPTION
The problem underlying the present invention is solved by the subject matter of the independent claims. Preferred embodiments may be taken from the attached dependent claims.
The present invention relates to compositions comprising short interfering RNA
(siRNA) directed to an expressed RNA transcript of EDNl (sometimes referred to as a "target nucleic acid" herein). The siRNA of the invention are nucleic acid molecules comprising a double stranded or duplex region. The present invention further relates to methods of using the siRNA compositions to reduce the expression level of EDNl . As used herein, the terms "silence" or "knock-down" when referring to gene expression means a reduction in gene expression. The present invention further relates to processes for making the siRNA.
In one aspect, the target nucleic acid is an RNA expressed from a mammalian EDNl gene. In one embodiment, the target nucleic acid is an RNA expressed from mouse EDNl . In another embodiment, the target nucleic acid is an RNA expressed from human EDNl . In another embodiment, the target nucleic acid is a human EDNl mRNA. In another embodiment, the target nucleic acid is a human EDNl mRNA. In another embodiment, the target nucleic acid is an mRNA comprising the sequence of SEQ ID NO: 17.
The siRNA of the present invention are suitable to inhibit the expression of EDNl . The siRNA according to the present invention is, thus, suitable to trigger the RNA interference response resulting in the reduction of the EDNl mRNA in a mammalian cell. The siRNA according to the present invention are further suitable to decrease the expression of EDNl protein by decreasing gene expression at the level of mRNA.
siRNA Design: An siRNA of the present invention comprises two strands of a nucleic acid, a first, antisense strand and a second, sense strand. The nucleic acid normally consists of ribonucleotides or modified ribonucleotides however; the nucleic acid may comprise deoxynucleotides (DNA) as described herein. The siRNA further comprises a double- stranded nucleic acid portion or duplex region formed by all or a portion of the antisense strand and all or a portion of the sense strand. The portion of the antisense strand forming the duplex region with the sense strand is the antisense strand duplex region or simply, the antisense duplex region, and the portion of the sense strand forming the duplex region with the antisense strand is the sense strand duplex region or simply, the sense duplex region. The duplex region is defined as beginning with the first base pair formed between the antisense strand and the sense strand and ending with the last base pair formed between the antisense
strand and the sense strand, inclusive. The portion of the siRNA on either side of the duplex region is the flanking regions. The portion of the antisense strand on either side of the antisense duplex region is the antisense flanking regions. The portion of the antisense strand 5' to the antisense duplex region is the antisense 5' flanking region. The portion of the antisense strand 5' to the antisense duplex region is the antisense 3' flanking region. The portion of the sense strand on either side of the sense duplex region is the sense flanking regions. The portion of the sense strand 5 ' to the sense duplex region is the sense 5' flanking region. The portion of the sense strand 5' to the sense duplex region is the sense 3' flanking region.
Complementarity: In one aspect, the antisense duplex region and the sense duplex region may be fully complementary and are at least partially complementary to each other. Such complementarity is based on Watson-Crick base pairing (i.e., A:U and G:C base pairing). Depending on the length of a siRNA a perfect match in terms of base complementarity between the antisense and sense duplex regions is not necessarily required however, the antisense and sense strands must be able to hybridize under physiological conditions.
In one embodiment, the complementarity between the antisense strand and sense strand is perfect (no nucleotide mismatches or additional/deleted nucleotides in either strand).
In one embodiment, the complementarity between the antisense duplex region and sense duplex region is perfect (no nucleotide mismatches or additional/deleted nucleotides in the duplex region of either strand).
In another embodiment, the complementarity between the antisense duplex region and the sense duplex region is not perfect. In one embodiment, the identity between the antisense duplex region and the complementary sequence of the sense duplex region is selected from the group consisting of at least 75%, 80%>, 85%, 90% and 95%>; wherein a siRNA comprising the antisense duplex region and the sense duplex region is suitable for reducing expression of EDN1 In another embodiment, the siRNA, wherein the identity between the antisense duplex region and complementary sequence of the sense duplex region is selected from the group consisting of at least 75%, 80%>, 85%, 90% and 95%, is able to reduce expression of EDN1 by at least 25%, 50% or 75% of a comparative siRNA having a duplex region with perfect identity between the antisense duplex region and the sense duplex region. As used herein the term "comparative siRNA" is a siRNA that is identical to the siRNA to which it is being compared, except for the specified difference, and which is tested under identical conditions.
R Ai using siRNA involves the formation of a duplex region between all or a portion of the antisense strand and a portion of the target nucleic acid. The portion of the target nucleic acid that forms a duplex region with the antisense strand, defined as beginning with the first base pair formed between the antisense strand and the target sequence and ending with the last base pair formed between the antisense strand and the target sequence, inclusive, is the target nucleic acid sequence or simply, target sequence. The duplex region formed between the antisense strand and the sense strand may, but need not be the same as the duplex region formed between the antisense strand and the target sequence. That is, the sense strand may have a sequence different from the target sequence however; the antisense strand must be able to form a duplex structure with both the sense strand and the target sequence.
In one embodiment, the complementarity between the antisense strand and the target sequence is perfect (no nucleotide mismatches or additional/deleted nucleotides in either nucleic acid).
In one embodiment, the complementarity between the antisense duplex region (the portion of the antisense strand forming a duplex region with the sense strand) and the target sequence is perfect (no nucleotide mismatches or additional/deleted nucleotides in either nucleic acid).
In another embodiment, the complementarity between the antisense duplex region and the target sequence is not perfect. In one embodiment, the identity between the antisense duplex region and the complementary sequence of the target sequence is selected from the group consisting of at least 75%, 80%, 85%, 90% or 95%, wherein a siRNA comprising the antisense duplex region is suitable for reducing expression of EDN1. In another embodiment, the siRNA, wherein the identity between the antisense duplex region and complementary sequence of the target sequence is selected from the group consisting of at least 75%, 80%, 85%, 90% and 95%, is able to reduce expression of EDN1 by at least 25%, 50% or 75% of a comparative siRNA with perfect identity to the antisense strand and target sequence.
In another embodiment, the siRNA of the invention comprises a duplex region wherein the antisense duplex region has a number of nucleotides selected from the group consisting of 1, 2, 3, 4 and 5 that are not base-paired to a nucleotide in the sense duplex region, and wherein said siRNA is suitable for reducing expression of EDN1. Lack of base- pairing is due to either lack of complementarity between bases (i.e., no Watson-Crick base pairing) or because there is no corresponding nucleotide on either the antisense duplex region or the sense duplex region such that a bulge is created. In one embodiment, a siRNA
comprising an antisense duplex region having a number of nucleotides selected from the group consisting of 1, 2, 3, 4 and 5 that are not base-paired to the sense duplex region, is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense duplex region are base paired with all nucleotides of said sense duplex region.
In another embodiment, the antisense strand has a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that do not base-pair to the sense strand, and wherein a siRNA comprising said antisense strand is suitable for reducing expression of EDN1. Lack of complementarity is due to either lack of complementarity between bases or because there is no corresponding nucleotide on either the antisense strand or the sense strand. The lack of a corresponding nucleotide results in either a single-stranded overhang or a bulge (if in the duplex region), in either the antisense strand or the sense strand. In one embodiment, a siRNA comprising an antisense strand having a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that do not base pair to the sense strand, is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of the sense strand. In one embodiment, a siRNA comprising an antisense strand having a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that are mismatched to the target sequence, is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of said sense strand. In another embodiment, all of the mismatched nucleotides are outside the duplex region.
In another embodiment, the antisense duplex region has a number of nucleotides selected from 1, 2, 3, 4 or 5 that do not base-pair to the sense duplex region, and wherein a siRNA comprising said antisense duplex region is suitable for reducing expression of EDN1. Lack of complementarity is due to either lack of complementarity between bases or because there is no corresponding nucleotide on either the antisense duplex region or the sense duplex region such that a bulge in created in either the antisense duplex region or the sense duplex region. In one embodiment, a siRNA comprising an antisense duplex region having a number of nucleotides selected from the group consisting of 1, 2, 3, 4 and 5 that do not base pair to the sense duplex region, is able to reduce expression of EDN1 by at least 25%, 50%, 75% of a comparative siRNA wherein all nucleotides of said antisense duplex region are complementary to all of the nucleotides of said sense duplex region.
In another embodiment, the antisense strand has a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that do not base-pair to the target sequence, and wherein a siRNA comprising said antisense strand is suitable for reducing expression of EDNl . Lack of complementarity is due to either lack of complementarity between bases or because there is no corresponding nucleotide on either the antisense strand or the target sequence. The lack of a corresponding nucleotide results in a bulge in either the antisense strand or the target sequence. In one embodiment, a siRNA comprising an antisense strand having a number of nucleotides selected from 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10 do not base pair to the target sequence, is able to reduce expression of EDNl by at least 25%, 50%>, 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of said target sequence. In one embodiment, a siRNA comprising an antisense strand having a number of nucleotides selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 that are mismatched to the target sequence, is able to reduce expression of EDNl by at least 25%, 50% or 75% of a comparative siRNA wherein all nucleotides of said antisense strand are complementary to all nucleotides of said target sequence.
In another embodiment, the complementarity between an antisense duplex region and both a sense duplex region and a target sequence of an siRNA is such that the antisense duplex region and the sense duplex region or the target sequence hybridize to one another under physiological conditions (37°C in a physiological buffer) and the siRNA is suitable for reducing expression of EDNl . In one embodiment, the siRNA comprising an antisense duplex region that hybridizes to a sense duplex region and a target sequence under physiological conditions, is able to reduce expression of EDNl by at least 25%, 50%, 75% of a comparative siRNA with perfect complementarity between the antisense strand and target sequence.
In another aspect, the complementarity between an antisense duplex region and a sense duplex region of a siRNA is such that the antisense duplex region and sense duplex region hybridize under the following conditions: 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C, and is suitable for reducing expression of EDNl . In one embodiment, the siRNA comprising an antisense duplex region and a sense duplex region that hybridize to one another under the conditions 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C, is able to reduce expression of EDNl by at least 25%, 50%, 75% of a comparative siRNA with perfect complementarity between the antisense duplex region and sense duplex region.
In another embodiment, the complementarity between an antisense strand of a siRNA and a target sequence is such that the antisense strand and target sequence hybridize under the following conditions: 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C and wherein the siRNA is suitable for reducing expression of EDNl . In one embodiment, the siRNA comprising an antisense strand that hybridizes to the target sequence under the following conditions: 400 mM NaCl, 40 mM PIPES pH 6.4, 1 mM EDTA, 70°C, is able to reduce expression of EDNl by at least 25%, 50%>, 75% of a comparative siRNA with perfect complementarity between the antisense strand and the target sequence.
Length: RNA interference is observed using long nucleic acid molecules comprising several dozen or hundreds of base pairs, although shorter RNAi molecules are generally preferred.
In one embodiment, the length of the siRNA duplex region is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 base pairs. In one embodiment, the length of the siRNA duplex region is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 consecutive base pairs. In another embodiment, the length of the siRNA duplex region is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 base pairs. In another embodiment, the length of the siRNA duplex region is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 consecutive base pairs.
In one embodiment, the length of the antisense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides. In one embodiment, the length of the antisense stand is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides.
In one embodiment, the length of the sense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides. In one embodiment, the length of the sense stand is selected from the group
consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides.
In one embodiment, the length of the antisense strand and the length of the sense strand are independently selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides. In one embodiment, the length of the antisense strand and the length of the sense stand are independently selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides. In one embodiment, the antisense strand and the sense strand are equal in length. In another embodiment, the antisense strand and the sense stand are equal in length, wherein the length is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides.
In one embodiment, the length of the antisense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NO: 1, 3, 5, 7, 9, 11, 13 or 15, preferably 1, 3, 5, or 7.
In one embodiment, the length of the antisense strand is selected from the group consisting of about 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7.
In one embodiment, the length of the sense strand is selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
In one embodiment, the length of the sense strand is selected from the group consisting of about 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides, wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
In one embodiment, the length of the antisense strand and the length of the sense strand are independently selected from the group consisting of about 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to
23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NO. NOs: 2, 4, 6, 8, 10, 12, 14, 16, 18, 20, 22,
24, 26, 28, 30, 32, 34, 36, 38, 40, 42, 44, 46, 48, 50, 52, 54, 56, 58, 60, 62, 64 or 68, and wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
In one embodiment, the length of the antisense strand and the length of the sense stand are independently selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7, and wherein the sense strand comprises the nucleotide sequence SEQ ID NOs: 2,
4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
In one embodiment, the antisense strand and the sense strand are equal in length, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7, and wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
In another embodiment, the antisense strand and the sense stand are equal in length, wherein the length is selected from the group consisting of 16 to 35, 16 to 30, 17 to 35, 17 to 30, 17 to 25, 17 to 24, 18 to 29, 18 to 25, 18 to 24, 18 to 23, 19 to 25, 19 to 24, 19 to 23, 20 to 25, 20 to 24, 21 to 25 and 21 to 24 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11 , 13 or 15, preferably SEQ ID NOs: 1, 3,
5, or 7, and wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4,
6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
In another embodiment, the antisense strand and the sense stand are equal in length, wherein the length is selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24,
25, 26, 27, 28, 29, 30, 31, 32, 33, 34 and 35 nucleotides, wherein the antisense strand comprises the nucleotide sequence of SEQ ID NOs: 1, 3, 5, 7, 9, 11, 13 or 15, preferably SEQ ID NOs: 1, 3, 5, or 7, and wherein the sense strand comprises the nucleotide sequence of SEQ ID NOs: 2, 4, 6, 8, 10, 12, 14 or 16, preferably SEQ ID NOs: 2, 4, 6, or 8.
Certain embodiments provide for antisense and sense strand combinations (identified by SEQ ID NO:): 1 and 2; 3 and 4; 5 and 6; and 7 and 8.
Ends (overhangs and blunt ends): The siRNA of the present invention may comprise an overhang or be blunt ended. An "overhang" as used herein has its normal and customary
meaning in the art, i.e., a single stranded portion of a nucleic acid that extends beyond the terminal nucleotide of a complementary strand in a double strand nucleic acid. The term "blunt end" includes double stranded nucleic acid whereby both strands terminate at the same position, regardless of whether the terminal nucleotide(s) are base paired. In one embodiment, the terminal nucleotide of an antisense strand and a sense strand at a blunt end are base paired. In another embodiment, the terminal nucleotide of a antisense strand and a sense strand at a blunt end are not paired. In another embodiment, the terminal two nucleotides of an antisense strand and a sense strand at a blunt end are base paired. In another embodiment, the terminal two nucleotides of a antisense strand and a sense strand at a blunt end are not paired.
In one embodiment, the siRNA has an overhang at one end and a blunt end at the other. In another embodiment, the siRNA has an overhang at both ends. In another embodiment, the siRNA is blunt ended at both ends. In one embodiment, the siRNA is blunt ended at one end. In another embodiment, the siRNA is blunt ended at the end with the 5 '- end of the antisense strand and the 3 '-end of the sense strand. In another embodiment, the siRNA is blunt ended at the end with the 3 '-end of the antisense strand and the 5 '-end of the sense strand. In another embodiment, the siRNA is blunt ended at both ends.
In another embodiment, the siRNA comprises a overhang at a 3'- or 5 '-end. In one embodiment, the siRNA has a 3 '-overhang on the antisense strand. In another embodiment, the siRNA has a 3 '-overhang on the sense strand. In another embodiment, the siRNA has a 5'- overhang on the antisense strand. In another embodiment, the siRNA has a 5 '-overhang on the sense strand. In another embodiment, the siRNA has an overhang at both the 5 '-end and 3'- end of the antisense stand. In another embodiment, the siRNA has an overhang at both the 5'- end and 3 '-end of the sense stand. In another embodiment, the siRNA has a 5' overhang on the antisense stand and a 3 ' overhang on the sense strand. In another embodiment, the siRNA has a 3' overhang on the antisense stand and a 5' overhang on the sense strand. In another embodiment, the siRNA has a 3' overhang on the antisense stand and a 3' overhang on the sense strand. In another embodiment, the siRNA has a 5' overhang on the antisense stand and a 5 ' overhang on the sense strand.
In one embodiment, the overhang at the 3 '-end of the antisense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides. In one embodiment, the overhang at the 3 '-end of the sense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides. In one embodiment, the overhang at the 5 '-end of the antisense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides. In one
embodiment, the overhang at the 5 '-end of the sense strand has a length selected from the group consisting of 1, 2, 3, 4 and 5 nucleotides.
Modification: Another aspect relates to modifications of the siRNA. The siR A according to the invention are a ribonucleic acid or a modified ribonucleic acid. Chemical modifications of the siRNA of the present invention provides a powerful tool in overcoming potential limitations including, but not limited to, in vitro and in vivo stability and bioavailability inherent to native RNA molecules. Chemically-modified siRNA can also minimize the possibility of activating interferon activity in humans. Chemical modification can further enhance the functional delivery of a siRNA to a target cell. The modified siRNA of the present invention may comprise one or more chemically modified ribonucleotides of either or both of the antisense strand or the sense strand. A ribonucleotide may comprise a chemical modification of the base, sugar or phosphate moieties.
Modifications to base moiety: A secondary aspect relates to modifications to a base moiety. One or more nucleotides of a siRNA of the present invention may comprise a modified base. A "modified base" means a nucleotide base other than an adenine, guanine, cytosine or uracil at the Γ position.
In one aspect, the siRNA comprises at least one nucleotide comprising a modified base. In one embodiment, the modified base in on the antisense strand. In another embodiment, the modified base in on the sense strand. In another embodiment, the modified base is in the duplex region. In another embodiment, the modified base is outside the duplex region, i.e., in a single stranded region. In another embodiment, the modified base is on the antisense strand and is outside the duplex region. In another embodiment, the modified base is on the sense strand and is outside the duplex region. In another embodiment, the 3 '-terminal nucleotide of the antisense strand is a nucleotide with a modified base. In another embodiment, the 3 '-terminal nucleotide of the sense strand is nucleotide with a modified base. In another embodiment, the 5 '-terminal nucleotide of the antisense strand is nucleotide with a modified base. In another embodiment, the 5 '-terminal nucleotide of the sense strand is nucleotide with a modified base.
In one embodiment, a siRNA has 1 modified base. In another embodiment, a siRNA has about 2-4 modified bases. In another embodiment, a siRNA has about 4-6 modified bases. In another embodiment, a siRNA has about 6-8 modified bases. In another embodiment, a siRNA has about 8-10 modified bases. In another embodiment, a siRNA has about 10-12 modified bases. In another embodiment, a siRNA has about 12-14 modified bases. In another
embodiment, a siRNA has about 14-16 modified bases. In another embodiment, a siRNA has about 16-18 modified bases. In another embodiment, a siRNA has about 18-20 modified bases. In another embodiment, a siRNA has about 20-22 modified bases. In another embodiment, a siRNA has about 22-24 modified bases. In another embodiment, a siRNA has about 24-26 modified bases. In another embodiment, a siRNA has about 26-28 modified bases. In each case the siRNA comprising said modified bases retains at least 50% of its activity as compared to the same siRNA but without said modified bases.
In one embodiment, the modified base is a purine. In another embodiment, the modified base is a pyrimidine. In another embodiment, at least half of the purines are modified. In another embodiment, at least half of the pyrimidines are modified. In another embodiment, all of the purines are modified. In another embodiment, all of the pyrimidines are modified.
In another embodiment, the siRNA comprises a nucleotide comprising a modified base, wherein the base is selected from the group consisting of 2-aminoadenosine, 2,6- diaminopurine,inosine, pyridin-4-one, pyridin-2-one, phenyl, pseudouracil, 2, 4, 6-trimethoxy benzene, 3-methyl uracil, dihydrouridine, naphthyl, aminophenyl, 5-alkylcytidine (e.g., 5- methylcytidine), 5-alkyluridine (e.g., ribothymidine), 5-halouridine (e.g., 5-bromouridine), 6- azapyrimidine, 6-alkylpyrimidine (e.g. 6-methyluridine), propyne, quesosine, 2-thiouridine, 4- thiouridine, wybutosine, wybutoxosine, 4-acetylcytidine, 5-(carboxyhydroxymethyl)uridine, 5'-carboxymethylaminomethyl-2-thiouridine, 5-carboxymethylaminomethyluridine, beta-D- galactosylqueosine, 1-methyladenosine, 1-methylinosine, 2,2-dimethylguanosine, 3- methylcytidine, 2-methyladenosine, 2-methylguanosine, N6-methyladenosine, 7- methylguanosine, 5-methoxyaminomethyl-2-thiouridine, 5-methylaminomethyluridine, 5- methylcarbonylmethyluridine, 5-methyloxyuridine, 5-methyl-2-thiouridine, 2-methylthio-N6- isopentenyladenosine, beta-D-mannosylqueosine, uridine-5-oxyacetic acid and 2-thiocytidine.
In another aspect, a siRNA of the present invention comprises an abasic nucleotide. The term "abasic" as used herein, refers to moieties lacking a base or having other chemical groups in place of a base at the Γ position, for example a 3',3'-linked or 5',5'-linked deoxyabasic ribose derivative. As used herein, a nucleotide with a modified base does not include abasic nucleotides. In one aspect, the siRNA comprises at least one abasic nucleotide. In one embodiment, the abasic nucleotide is on the antisense strand. In another embodiment, the abasic nucleotide is on the sense strand. In another embodiment, the abasic nucleotide is in the duplex region. In another embodiment, the abasic nucleotide is outside the duplex
region. In another embodiment, the abasic nucleotide is on the antisense strand and is outside the duplex region. In another embodiment, the abasic nucleotide is on the sense strand and is outside the duplex region. In another embodiment, the 3 '-terminal nucleotide of the antisense strand is an abasic nucleotide. In another embodiment, the 3 '-terminal nucleotide of the sense strand is an abasic nucleotide. In another embodiment, the 5 '-terminal nucleotide of the antisense strand is an abasic nucleotide. In another embodiment, the 5 '-terminal nucleotide of the sense strand is an abasic nucleotide. In another embodiment, a siR A has a number of abasic nucleotides selected from the group consisting of 1, 2, 3, 4, 5 and 6.
Modifications to sugar moiety: Another secondary aspect relates to modifications to a sugar moiety. One or more nucleotides of an siRNA of the present invention may comprise a modified ribose moiety.
Modifications at the 2'-position wherein the 2'-OH is substituted include the non- limiting examples selected from the group consisting of alkyl, substituted alkyl, alkaryl-, aralkyl-, -F, -CI, -Br, -CN, -CF3, -OCF3, -OCN, -O-alkyl, -S-alkyl, HS-alkyl-O, -O-alkenyl, -S-alkenyl, -N-alkenyl, -SO-alkyl, -alkyl-OSH, -alkyl-OH, -O-alkyl-OH, -O-alkyl-SH, -S- alkyl-OH, -S-alkyl-SH, -alkyl-S-alkyl, -alkyl-O-alkyl, -ON02, -N02, -N3, -NH2, alkylamino, dialkylamino-, aminoalkyl-, aminoalkoxy, aminoacid, aminoacyl-, -ONH2, -O-aminoalkyl, - O-aminoacid, -O-aminoacyl, heterocycloalkyl-, heterocycloalkaryl-, aminoalkylamino-, polyalklylamino-, substituted silyl-, methoxyethyl- (MOE), alkenyl and alkynyl. "Locked" nucleic acids (LNA) in which the 2' hydroxyl is connected, e.g., by a methylene bridge, to the 4' carbon of the same ribose sugar is further included as a 2' modification of the present invention. Preferred substitutents are 2'-methoxy ethyl, 2'-0-CH3, 2'-0-allyl, 2'-C-allyl, and 2'- fluoro.
In one embodiment, the siRNA comprises 1-5 2'-modified nucleotides. In another embodiment, the siRNA comprises 5-10 2'-modified nucleotides. In another embodiment, the siRNA comprises 15-20 2'-modified nucleotides. In another embodiment, the siRNA comprises 20-25 2'-modified nucleotides. In another embodiment, the siRNA comprises 25- 30 2 '-modified nucleotides.
In one embodiment, the antisense strand comprises 1-2 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 2-4 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 4-6 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 6-8 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 8-10 2'-modified nucleotides. In one
embodiment, the antisense strand comprises about 10-12 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 12-14 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 14-16 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 16-18 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 18-20 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 22-24 2'-modified nucleotides. In one embodiment, the antisense strand comprises about 24-26 2'-modified nucleotides.
In one embodiment, the sense strand comprises 1-2 2'-modified nucleotides. In one embodiment, the sense strand comprises about 2-4 2'-modified nucleotides. In one embodiment, the sense strand comprises about 4-6 2'-modified nucleotides. In one embodiment, the sense strand comprises about 6-8 2'-modified nucleotides. In one embodiment, the sense strand comprises about 8-10 2'-modified nucleotides. In one embodiment, the sense strand comprises about 10-12 2'-modified nucleotides. In one embodiment, the sense strand comprises about 12-14 2'-modified nucleotides. In one embodiment, the sense strand comprises about 14-16 2'-modified nucleotides. In one embodiment, the sense strand comprises about 16-18 2'-modified nucleotides. In one embodiment, the sense strand comprises about 18-20 2'-modified nucleotides. In one embodiment, the sense strand comprises about 22-24 2'-modified nucleotides. In one embodiment, the sense strand comprises about 24-26 2'-modified nucleotides.
In one embodiment, the siR A comprises 1-5 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 5-10 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 15-20 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 20-25 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA comprises 25-30 2'-0-CH3 modified nucleotides.
In one embodiment, the antisense strand comprises 1-2 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 2-4 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 4-6 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 6-8 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 8-10 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 10-12 2'-0- CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 12-14 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 14- 16 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about
16-18 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 18-20 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 22-24 2'-0-CH3 modified nucleotides. In one embodiment, the antisense strand comprises about 24-26 2'-0-CH3 modified nucleotides.
In one embodiment, the sense strand comprises 1-2 2'-0-CH3 modified nucleotides.
In one embodiment, the sense strand comprises about 2-4 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 4-6 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 6-8 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 8-10 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 10-12 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 12-14 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 14-16 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 16-18 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 18-20 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 22-24 2'-0-CH3 modified nucleotides. In one embodiment, the sense strand comprises about 24-26 2'-0-CH3 modified nucleotides.
In one embodiment, the siR A duplex region comprises 1-5 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 5-10 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 15-20 2'- 0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 20-25 2'-0-CH3 modified nucleotides. In another embodiment, the siRNA duplex region comprises 25-30 2'-0-CH3 modified nucleotides.
In one embodiment, the antisense duplex region comprises 1-2 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 2-4 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 4-6 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 6-8 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 8-10 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 10-12 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 12-14 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 14-16 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 16-18 2'-0- CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about
18-20 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 22-24 2'-0-CH3 modified nucleotides. In one embodiment, the antisense duplex region comprises about 24-26 2'-0-CH3 modified nucleotides.
In one embodiment, the sense duplex region comprises 1-2 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 2-4 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 4-6 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 6-8 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 8-10 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 10-12 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 12-14 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 14-16 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 16-18 2'-0- CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 18-20 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 22-24 2'-0-CH3 modified nucleotides. In another embodiment, the sense duplex region comprises about 24-26 2'-0-CH3 modified nucleotides.
In one embodiment, the siRNA comprises an antisense strand 19 nucleotides in length and a sense strand 19 nucleotides in length, wherein said antisense strand comprises 2'-0- CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17 and 19, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16 and 18, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'. In another embodiment, the siRNA comprises an antisense strand 20 nucleotides in length and a sense strand 20 nucleotides in length, wherein said antisense strand comprises 2'- 0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17 and 19, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18 and 20 wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'. In another embodiment, the siRNA comprises an antisense strand 21 nucleotides in length and a sense strand 21 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17, 19 and 21, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18 and 20, wherein said antisense strand is numbered from 5'-3' and said sense strand is numbered from 3 '-5'. In another embodiment, the siRNA comprises an antisense
strand 22 nucleotides in length and a sense strand 22 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17, 19 and 21, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18, 20 and 22, wherein said antisense strand is numbered from 5'-3 ' and said sense strand is numbered from 3 '-5'. In another embodiment, the siR A comprises an antisense strand 23 nucleotides in length and a sense strand 23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 1, 3, 5, 7, 9, 11, 13, 15, 17, 19, 21 and 23, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 2, 4, 6, 8, 10, 12 ,14, 16, 18, 20 and 22 wherein said antisense strand is numbered from 5 '-3 ' and said sense strand is numbered from 3 '-5 ' .
In another embodiment, the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 3, 5, 7, 9, 11, 13, 15 and 17, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 4, 6, 8, 10, 12 ,14 and 16, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5' . In another embodiment, the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0- CH3 modifications at nucleotides 5, 7, 9, 11, 13 and 15, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 6, 8, 10, 12 and 14, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'. In another embodiment, the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 7, 9, 11, 13 and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 8, 10 and 12, wherein said antisense strand is numbered from 5'- 3' and said sense strand is numbered from 3 '-5'. In another embodiment, the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 7, 9 and 11, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 8, 10 and 12, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'. In another embodiment, the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0-CH3 modifications at nucleotides 7 and 9, and wherein said sense strand comprises 2'-0-CH3 modifications at nucleotides 8 and 10, wherein said
antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'. In another embodiment, the siRNA comprises an antisense strand 18-23 nucleotides in length and a sense strand 18-23 nucleotides in length, wherein said antisense strand comprises 2'-0- CH3 modifications at nucleotides 9 and 11, and wherein said sense strand comprises 2'-0- CH3 modifications at nucleotides 8 and 10, wherein said antisense strand is numbered from 5 '-3' and said sense strand is numbered from 3 '-5'.
In another embodiment, the antisense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25 2'-deoxy nucleotides.
In another embodiment, the sense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25 2 '-deoxy nucleotides.
In another embodiment, the antisense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11,
12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25 2'-fluoro nucleotides.
In another embodiment, the sense strand comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12,
13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25 2'-fluoro nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense strand are 2'-0- methyl pyrimidine nucleotides.
In another embodiment, of the purine nucleotides in the antisense strand are 2'-0- methyl purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense strand are 2'- deoxy pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the antisense strand are 2 '-deoxy purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense strand are 2'- fluoro pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the antisense strand are 2'-fluoro purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense strand are 2'-0-methyl pyrimidine nucleotides.
In another embodiment, of the purine nucleotides in the sense strand are 2'-0-methyl purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense strand are 2'-deoxy pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the sense strand are 2'-deoxy purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense strand are 2'-fluoro pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the sense strand are 2'-fluoro purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense duplex region are 2'-0-methyl pyrimidine nucleotides.
In another embodiment, of the purine nucleotides in the antisense duplex region are 2'- O-methyl purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense duplex region are 2'-deoxy pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the antisense duplex region are 2'- deoxy purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense duplex region are
2'-fluoro pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the antisense duplex region are 2'- fluoro purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense duplex region are 2'- O-methyl pyrimidine nucleotides.
In another embodiment, of the purine nucleotides in the sense duplex region are 2'-0- methyl purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense duplex region are 2'- deoxy pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the sense duplex region are 2'-deoxy purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense duplex region are 2'- fluoro pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the sense duplex region are 2'-fluoro purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense duplex flanking regions are 2'-0-methyl pyrimidine nucleotides.
In another embodiment, of the purine nucleotides in the antisense duplex flanking regions are 2'-0-methyl purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense duplex flanking regions are 2 '-deoxy pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the antisense duplex flanking regions are 2'-deoxy purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the antisense duplex flanking regions are 2'-fluoro pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the antisense duplex flanking regions are 2'-fluoro purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense duplex flanking regions are 2'-0-methyl pyrimidine nucleotides.
In another embodiment, of the purine nucleotides in the sense duplex flanking regions are 2'-0-methyl purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense duplex flanking regions are 2 '-deoxy pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the sense duplex flanking regions are 2 '-deoxy purine nucleotides.
In another embodiment, the pyrimidine nucleotides in the sense duplex flanking regions are 2'-fluoro pyrimidine nucleotides.
In another embodiment, the purine nucleotides in the sense duplex flanking regions are 2'-fluoro purine nucleotides.
Pattern: In one aspect, the antisense duplex region comprises a plurality of groups of modified nucleotides, referred to herein as "modified groups", wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a second group of nucleotides, referred to herein as "flanking groups", wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group. In one embodiment, each modified group in the antisense duplex region is identical, i.e., each modified group consists of an equal number of identically modified nucleotides. In another embodiment, each flanking group has an equal number of nucleotide. In another embodiment, each flanking group is identical. In another embodiment, the nucleotides of said modified groups in the antisense duplex region comprise a modified base. In another
embodiment, the nucleotides of said modified groups comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups comprise a modified 2' position.
In another aspect, the sense duplex region comprises a plurality of groups of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group. In one embodiment, each modified group in the sense duplex region is identical. In another embodiment, each flanking group has an equal number of nucleotides. In another embodiment, each flanking group is identical. In another embodiment, the nucleotides of said modified groups in the sense duplex region comprise a modified base. In another embodiment, the nucleotides of said modified groups comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups comprise a modified 2' position.
In another aspect, the antisense duplex region and the sense duplex region each comprise a plurality of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group. In one embodiment, each modified group in the antisense duplex region and the sense duplex region are identical. In another embodiment, each flanking group in the antisense duplex region and the sense duplex region each have an equal number of nucleotides. In another embodiment, each flanking group in the antisense duplex region and in the sense duplex region are identical. In another embodiment, the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise the same modified groups and the same flanking groups. In another embodiment, the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise a modified base. In another embodiment, the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups in the antisense duplex region and the sense duplex region each comprise a modified 2' position.
In one aspect, the antisense strand comprises a plurality of groups of modified nucleotides, referred to herein as "modified groups", wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a second group of nucleotides, referred to herein as "flanking groups", wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group. In one embodiment, each modified group in the antisense strand is identical, i.e., each modified group consists of an equal number of identically modified nucleotides. In another embodiment, each flanking group has an equal number of nucleotide. In another embodiment, each flanking group is identical. In another embodiment, the nucleotides of said modified groups in the antisense strand comprise a modified base. In another embodiment, the nucleotides of said modified groups comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups comprise a modified 2' position.
In another aspect, the sense strand comprises a plurality of groups of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group. In one embodiment, each modified group in the sense strand is identical. In another embodiment, each flanking group has an equal number of nucleotides. In another embodiment, each flanking group is identical. In another embodiment, the nucleotides of said modified groups in the sense strand comprise a modified base. In another embodiment, the nucleotides of said modified groups comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups comprise a modified 2' position.
In another aspect, the antisense strand and the sense strand each comprise a plurality of modified groups, wherein each modified group consists of one or more identically modified nucleotides, wherein each modified group is flanked on one or both sides by a flanking group, wherein each said flanking group consists of one or more nucleotides that are either unmodified or modified in a manner different from the nucleotides of said modified group. In one embodiment, each modified group in the antisense strand and the sense strand are identical. In another embodiment, each flanking group in the antisense strand and the sense strand each have an equal number of nucleotides. In another embodiment, each flanking group in the antisense strand and in the sense strand are identical. In another
embodiment, the nucleotides of said modified groups in the antisense strand and the sense strand each comprise the same modified groups and the same flanking groups. In another embodiment, the nucleotides of said modified groups in the antisense strand and the sense strand each comprise a modified base. In another embodiment, the nucleotides of said modified groups in the antisense strand and the sense strand each comprise a modified phosphate backbone. In another embodiment, the nucleotides of said modified groups in the antisense strand and the sense strand each comprise a modified 2' position.
In another aspect, the modified groups and the flanking groups form a regular pattern on the antisense stand. In another aspect, the modified groups and the flanking groups form a regular pattern on the sense strand. In one embodiment, the modified groups and the flanking groups form a regular pattern on the both the antisense strand and the sense strand. In another embodiment, the modified groups and the flanking groups form a regular pattern on the antisense duplex region. In another aspect, the modified groups and the flanking groups form a regular pattern on the sense duplex region. In one embodiment, the modified groups and the flanking groups form a regular pattern on the both the antisense duplex region and the sense duplex region.
In another aspect, the pattern is a spatial or positional pattern. A spatial or positional pattern means that (a) nucleotide(s) are modified depending on their position within the nucleotide sequence of a double-stranded portion. Accordingly, it does not matter whether the nucleotide to be modified is a pyrimidine or a purine. Rather the position of a modified nucleotide is dependent upon: (a) its numbered position on a strand of nucleic acid, wherein the nucleotides are numbered from the 5 '-end to the 3 '-end with the 5 '-end nucleotide of the strand being position one (both the antisense strand and sense strand are numbered from their respective 5 '-end nucleotide), or (b) the position of the modified group relative to a flanking group. Thus, according to this embodiment, the modification pattern will always be the same, regardless of the sequence which is to be modified.
In another embodiment, the number of modified groups on the antisense strand is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of modified groups on the sense strand is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of flanking groups on the antisense strand of nucleic acid is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of flanking groups on the sense strand of nucleic acid is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In one embodiment, the number of modified
groups and the number of flanking groups on either or both the antisense strand and the sense strand are the same.
In another embodiment, the number of modified groups on the antisense duplex region is selected 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of modified groups on the sense duplex region is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13 or 14. In another embodiment, the number of flanking groups on the antisense duplex region of nucleic acid is 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of flanking groups on the sense duplex region of nucleic acid is selected from 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13 or 14. In one embodiment, the number of modified groups and the number of flanking groups on either or both the antisense duplex region and the sense duplex region are the same.
In one embodiment, the number of modified groups and the number of flanking groups on a strand or on a duplex region are the same. In another embodiment, the number of modified groups and the number of flanking groups on a strand or on a duplex region are the same, wherein the number is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
In another embodiment, the number of nucleotides in a modified group is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14. In another embodiment, the number of nucleotides in a flanking group is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14.
In one embodiment, each modified group on both the antisense strand and the sense strand is identical. In one embodiment, each modified group on both the antisense duplex region and the sense duplex region is identical. In another embodiment, each modified group and each flanking group on both the antisense strand and the sense strand are identical. In one embodiment, each modified group and each flanking group on both the antisense duplex region and the sense duplex region are identical.
In one embodiment, each modified group, each modified group position, each flanking group and each flanking group position on both the antisense strand and the sense strand are identical. In one embodiment, each modified group, each modified group position, each flanking group and each flanking group position on both the antisense duplex region and the sense duplex region are identical. In another embodiment, the modified groups on the antisense strand are complementary with the modified groups on the sense strand (the modified groups on the antisense strand and the sense strand are perfectly aligned across from one another). In another embodiment, there are no mismatches in the modified groups such that each modified group on the antisense strand is base paired with each modified group on
the sense strand. In another embodiment, each modified group on the sense strand is shifted by 1 , 2, 3, 4 or 5 nucleotides relative to the modified groups on the antisense strand. For example, if each modified group on the sense strand is shifted by one nucleotide and a modified group started at position one on the antisense strand, a modified group on the sense strand would begin at position two. In another embodiment, the modified groups of the antisense strand do not overlap the modified groups of the sense strand, i.e., no nucleotide of a modified group on the antisense strand is base paired with a nucleotide of a modified group on the sense strand.
In one embodiment, deoxyribonucleotides at an end of a strand of nucleic acid are not considered when determining a position of a modified group, i.e., the positional numbering begins with the first ribonucleotide or modified ribonucleotide. In another embodiment, abasic nucleotides at an end of a strand of nucleic acid are not considered when determining a position of a modified group.
In one aspect, a modified group comprises a 5 '-end nucleotide of either or both of the antisense strand and the sense strand. In another embodiment, a flanking group comprises the 5 '-end nucleotide of either or both of the antisense strand and the sense strand. In another embodiment, the 5 '-end nucleotide of either or both of the antisense strand and the sense strand is unmodified. In another embodiment, a modified group comprises the 5 '-most nucleotide of either or both of the antisense duplex region and sense duplex region. In another embodiment, a flanking group comprises the 5 '-most nucleotide of either or both of the antisense duplex region or the sense duplex region. In another embodiment, the 5 '-most nucleotide of either or both of the antisense duplex region or the sense duplex region is unmodified. In another embodiment, the nucleotide at position 10 of the antisense strand is unmodified. In another embodiment, the nucleotide at position 10 of the sense strand is modified. In another embodiment, a modified group comprises the nucleotide at position 10 of the sense strand.
In one embodiment, the modification at the 2' position is selected from the group comprising amino, fluoro, methoxy, alkoxy and Ci-C3-alkyl. In another embodiment, the modification is 2'-0-methyl.
In another aspect, each modified group consists of one nucleotide and each flanking group consists of one nucleotide. In one embodiment, each modified group on the antisense strand is aligned with a flanking group on the sense strand.
In another aspect, each modified group consists of one 2'-0-methyl modified nucleotide and each flanking group consists of one nucleotide. In one embodiment, each flanking group consists of one unmodified nucleotide. In one embodiment, each flanking group consists of one 2'-0-methyl modified nucleotide. In another embodiment, each modified group on both the antisense strand and the sense strand consists of one 2'-0-methyl modified nucleotide and each flanking group on both the antisense strand and the sense strand consists of one nucleotide, wherein no modified group on one strand is either aligned or both aligned and base paired with another modified group on the other strand and no flanking group on one strand is either aligned or both aligned and base paired with a flanking group on the other strand. In another embodiment, excluding any optional overhangs, each modified group on each strand is either aligned or both aligned and based paired with a flanking group on the other strand. In one embodiment, the flanking group is unmodified. In another embodiment, the nucleotide of position one on the antisense strand is 2'-0-methyl modified. In another embodiment, the 5 '-most nucleotide of the antisense duplex region is 2'-0-methyl modified.
Positional modification schemes are described in international patent application WO 2004/015107, incorporated by reference in its entirety.
Modifications to phosphate backbone: Another secondary aspect relates to modifications to a phosphate backbone. All or a portion of the nucleotides of the siRNA of the invention may be linked through phosphodiester bonds, as found in unmodified nucleic acid. A siRNA of the present invention however, may comprise a modified phosphodiester linkage. The phosphodiester linkages of either the antisense stand or the sense strand may be modified to independently include at least one heteroatom selected from the group consisting of nitrogen and sulfur. In one embodiment, a phosphoester group connecting a ribonucleotide to an adjacent ribonucleotide is replaced by a modified group. In one embodiment, the modified group replacing the phosphoester group is selected from the group consisting of phosphorothioate, methylphosphonate or phosphoramidate group.
In one embodiment, all of the nucleotides of the antisense strand are linked through phosphodiester bonds. In another embodiment, all of the nucleotides of the antisense duplex region are linked through phosphodiester bonds. In another embodiment, all of the nucleotides of the sense strand are linked through phosphodiester bonds. In another embodiment, all of the nucleotides of the sense duplex region are linked through phosphodiester bonds. In another embodiment, the antisense strand comprises a number of modified phosphoester
groups selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10. In another embodiment, the antisense duplex region comprises a number of modified phosphoester groups selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10. In another embodiment, the sense strand comprises a number of modified phosphoester groups selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10. In another embodiment, the sense duplex region comprises a number of modified phosphoester groups selected from 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10.
5 ' and 3 ' end modifications: Another secondary aspect relates to 5' and 3 ' modifications. The siR A of the present invention may include nucleic acid molecules comprising one or more modified nucleotides, abasic nucleotides, acyclic or deoxyribonucleotide at the terminal 5 '- or 3 '-end on either or both of the sense or antisense strands. In one embodiment, the 5'- and 3 '-end nucleotides of both the sense and antisense strands are unmodified. In another embodiment, the 5 '-end nucleotide of the antisense strand is modified. In another embodiment, the 5 '-end nucleotide of the sense strand is modified. In another embodiment, the 3 '-end nucleotide of the antisense strand is modified. In another embodiment, the 3 '-end nucleotide of the sense strand is modified. In another embodiment, the 5 '-end nucleotide of the antisense strand and the 5 '-end nucleotide of the sense strand are modified. In another embodiment, the 3 '-end nucleotide of the antisense strand and the 3 '-end nucleotide of the sense strand are modified. In another embodiment, the 5 '-end nucleotide of the antisense strand and the 3 '-end nucleotide of the sense strand are modified. In another embodiment, the 3 '-end nucleotide of the antisense strand and the 5 '-end nucleotide of the sense strand are modified. In another embodiment, the 3 '-end nucleotide of the antisense strand and both the 5'- and 3 '-end nucleotides of the sense strand are modified. In another embodiment, both the 5'- and 3 '-end nucleotides of the antisense strand are modified. In another embodiment, both the 5'- and 3 '-end nucleotides of the sense strand are modified.
In another embodiment, the 5 '-end nucleotide of the antisense strand is phosphorylated. In another embodiment, the 5 '-end nucleotide of the sense strand is phosphorylated. In another embodiment, the 5 '-end nucleotides of both the antisense strand and the sense strand are phosphorylated. In another embodiment, the 5 '-end nucleotide of the antisense strand is phosphorylated and the 5 '-end nucleotide of the sense strand has a free hydroxyl group (5' -OH). In another embodiment, the 5 '-end nucleotide of the antisense strand is phosphorylated and the 5 '-end nucleotide of the sense strand is modified.
Modifications to the 5'- and 3 '-end nucleotides are not limited to the 5' and 3 ' positions on these terminal nucleotides. Examples of modifications to end nucleotides
include, but are not limited to, biotin, inverted (deoxy) abasics, amino, fluoro, chloro, bromo, CN, CF, methoxy, imidazole, caboxylate, thioate, Ci to C10 lower alkyl, substituted lower alkyl, alkaryl or aralkyl, OCF3, OCN, 0-, S-, or N-alkyl; 0-, S-, or N-alkenyl; SO-CH3 ; S02CH3; ON02; N02, N3; heterozycloalkyl; heterozycloalkaryl; amino alky lamino; polyalkylamino or substituted silyl, as, among others, described, e.g., in PCT patent application WO 99/54459, European patents EP 0 586 520 Bl or EP 0 618 925 B l , incorporated by reference in their entireties.. As used herein, "alkyl" means Ci-Ci2-alkyl and "lower alkyl" means Ci-C6-alkyl, including Ci-, C2-, C3-, C4-, C5- and C6-alkyl.
In another aspect, the 5 '-end of the antisense strand, the 5 '- end of the sense strand, the 3 '-end of the antisense strand or the 3 '-end of the sense strand is covalently connected to a prodrug moiety. In one embodiment, the moiety is cleaved in an endosome. In another the moiety is cleaved in the cytoplasm.
Various possible non-limiting embodiments of the siRNA of the present invention having different kinds of end modification(s) are presented in the following Table.
Various embodiments of the interfering ribonucleic acid according to the
present invention
Antisense strand Sense strand
1.) 5 '-end free OH free OH
3 '-end free OH free OH
2.) 5 -end free OH free OH
3 -end end modification end modification
3.) 5 -end free OH free OH
3 -end free OH end modification
4.) 5 -end free OH free OH
3 -end end modification free OH
5.) 5 -end free OH end modification
3 -end free OH free OH
6.) 5 -end free OH end modification
3 -end end modification free OH
7.) 5 -end free OH end modification
3 -end free OH end modification
8.) 5 -end free OH end modification
3 -end end modification end modification
In another embodiment, the terminal 3' nucleotide or two terminal 3 '-nucleotides on either or both of the antisense strand or sense strand is a 2'-deoxynucleotide. In another embodiment, the 2'-deoxynucleotide is a 2'-deoxy-pyrimidine. In another embodiment, the 2'- deoxynucleotide is a 2' deoxy-thymidine.
shRNA and linked siRNA: Another aspect relates to shR A and linked siR A. It is within the present invention that the double-stranded structure is formed by two separate strands, i.e. the antisense strand and the sense strand. However, it is also with in the present invention that the antisense strand and the sense strand are covalently linked to each other. Such linkage may occur between any of the nucleotides forming the antisense strand and sense strand, respectively. Such linkage can be formed by covalent or non-covalent linkages. Covalent linkage may be formed by linking both strands one or several times and at one or several positions, respectively, by a compound preferably selected from the group comprising methylene blue and bifunctinoal groups. Such bifunctional groups are preferably selected from the group comprising bis(2-chloroethyl)amine, N-acetly-N'-(p- glyoxylbenzoyl)cystamine, 4-thiouracile and psoralene.
In one aspect, the antisense strand and the sense strand are linked by a loop structure. In another embodiment, of the loop structure is comprised of a non-nucleic acid polymer. In another embodiment, the non-nucleic acid polymer is polyethylene glycol. In another embodiment, the 5 '-end of the antisense strand is linked to the 3 '-terminus of the sense strand. In another embodiment, the 3 '-end of the antisense strand is linked to the 5 '-end of the sense strand.
In another aspect, the loop consists of a nucleic acid. As used herein, locked nucleic acid (LNA) (Elayadi and Corey (2001) Curr Opin Investig Drugs. 2(4):558-61) and peptide
nucleic acid (PNA) (reviewed in Faseb J. (2000) 14: 1041-1060) are regarded as nucleic acids and may also be used as loop forming polymers. In one embodiment, the nucleic acid is ribonucleic acid. In one embodiment, the 5 '-terminus of the antisense strand is linked to the 3 '-terminus of the sense strand. In another embodiment, the 3 '-end of the antisense strand is linked to the 5 '-terminus of the sense strand. The loop consists of a minimum length of four nucleotides or nucleotide analogues. In one embodiment, the loop consists of a length of nucleotides or nucleotide analogues selected from 4, 5, 6, 7, 8, 9, 10, 1 1, 12, 13, 14 or 15. In one embodiment, the length of the loop is sufficient for linking the two strands covalently in a manner that a back folding can occur through a loop structure or similar structure. The ribonucleic acid constructs may be incorporated into suitable vector systems. Preferably the vector comprises a promoter for the expression of RNAi. Preferably the respective promoter is pol III and more preferably the promoters are the U6, HI, 7SK promoter as described in Good et al. (1997) Gene Ther, 4, 45-54.
In another aspect, the nucleic acid according to the present invention comprises a phosphorothioate internucleotide linkage. In one embodiment, a phosphorothioate internucleotide linkage is within 5 nucleotides from the 3 '-end or the 5 '-end of either or both of the antisense strand and the sense strand. The antisense strand can comprise about one to about five phosphorothioate internucleotide linkages.
Combinations of embodiments:
In one embodiment, an overhang at the 3 '-end of the sense strand is selected from consisting of 1, 2, 3, 4 and 5 nucleotides in length. In one embodiment, an overhang at the 5'- end of the antisense strand is selected from consisting of 1, 2, 3, 4 and 5 nucleotides in length. In one embodiment, an overhang at the 5 '-end of the sense strand is selected from consisting of 1, 2, 3, 4 and 5 nucleotides in length.
In one embodiment, the siRNA molecule is blunt-ended on both ends and has a length selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28 or 29 consecutive nucleotides.
In one embodiment, the siRNA molecule is blunt-ended on one end and the double stranded portion of the siRNA molecule has a length selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28 or 29 consecutive nucleotides.
In one embodiment, the siRNA molecule has overhangs on both ends and the double stranded portion of the siRNA molecule has a length selected from the group consisting of 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28 or 29 consecutive nucleotides.
In one embodiment, the siRNA molecule comprises an overhang, said overhang comprising at least one deoxyribonucleotide. In one embodiment, the siRNA molecule comprises an overhang, said overhang comprising two deoxyribonucleotides.
In one embodiment, the siRNA molecule has overhangs on the 3 '-end of the antisense strand and at the 3 '-end of the sense strand, said overhangs comprising at least one deoxyribonucleotide. In one embodiment, the siRNA molecule has overhangs on the 3 '-end of the antisense strand and at the 3 '-end of the sense strand, said overhangs consisting two deoxyribonucleotides.
To the extent it is referred herein to the position of a nucleotide within a nucleotide sequence as "even numbered" or "odd numbered", such numbering starts from the 5' end of such nucleotide sequence.
The nucleotide(s) forming the overhang may be (a) desoxyribonucleotide(s), (a) ribonucleotide(s) or a combination thereof. In one embodiment, the antisense strand and/or the sense strand comprise a TT dinucleotide at the 3 ' end.
Processes of making: The nucleic acid of the present invention can be produced using routine methods in the art including chemically synthesis or expressing the nucleic acid either in vitro (e.g., run off transcription) or in vivo. In one embodiment, the siRNA is produced using solid phase chemical synthesis. In another embodiment, the nucleic acid is produced using an expression vector. In one embodiment, the expression vector produced the nucleic acid of the invention in the target cell. Accordingly, such vector can be used for the manufacture of a medicament. Methods for the synthesis of the nucleic acid molecule described herein are known to the ones skilled in the art. Such methods are, among others, described in Caruthers et al, 1992, Methods in Enzymology 211, 3-19, Thompson et al, International PCT Publication No. WO 99/54459, Wincott et al, 1995, Nucleic Acids Res. 23, 2677-2684, Wincott et al, 1997, Methods Mol. Bio., 74, 59, Brennan et al, 1998, Biotechnol Bioeng., 61, 33-45, and Brennan, U.S. Pat. No. 6,001,311 (each incorporated herein by reference in their entireties).
Delivery/formulations : siRNA can be delivered to cells, both in vitro and in vivo, by a variety of methods known to those of skill in the art, including direct contact with cells ("naked" siRNA) or by in combination with one or more agents that facilitate targeting or delivery into cells. Such
agents and methods include lipoplexes, liposomes, iontophoresis, hydrogels, cyclodextrins, nanocapsules, micro- and nanospheres and proteinaceous vectors (e.g., Bioconjugate Chem. (1999) 10: 1068-1074 and WO 00/53722). The nucleic acid/vehicle combination may be locally delivered in vivo by direct injection or by use of an infusion pump. The siRNA of the invention can be delivered in vivo by various means including intravenous subcutaneous, intramuscular or intradermal injection or inhalation. The molecules of the instant invention can be used as pharmaceutical agents. Preferably, pharmaceutical agents prevent, modulate the occurrence, or treat (alleviate a symptom to some extent, preferably all of the symptoms) of a disease state in a subject.
There is also provided the use of a composition comprising surface-modified liposomes containing poly (ethylene glycol) lipids (PEG-modified, or long-circulating liposomes or stealth liposomes). These formulations offer a method for increasing stability of a liposome or lipoplex solutions by preventing their aggregation and fusion. The formulations also have the added benefit in vivo of resisting opsonization and elimination by the mononuclear phagocytic system (MPS or RES), thereby enabling longer blood circulation times and enhanced tissue exposure for the encapsulated drug. Such liposomes have been shown to accumulate selectively in tumors, presumably by extravasation and capture in the neovascularized target tissues (Lasic et al, Science 1995, 267, 1275-1276; Oku et al, 1995, Biochim. Biophys. Acta, 1238, 86-90). The long-circulating liposomes enhance the pharmacokinetics and pharmacodynamics of DNA and RNA, particularly compared to conventional cationic liposomes which are known to accumulate in tissues of the MPS (Liu et al, J. Biol. Chem. 1995,42,24864-24780; Choi et al, International PCT Publication No. WO 96/10391; Ansell et al, International PCT Publication No. WO 96/10390; Holland et al, International PCT Publication No. WO 96/10392). Long-circulating liposomes also protect the siRNA from nuclease degradation.
The siRNA of the present invention may be formulated as pharmaceutical compositions. The pharmaceutical compositions may be used as medicaments or as diagnostic agents, alone or in combination with other agents. For example, one or more siRNAs of the invention can be combined with a delivery vehicle (e.g., liposomes) and excipients, such as carriers, diluents. Other agents such as preservatives and stabilizers can also be added. Methods for the delivery of nucleic acid molecules are known in the art and described, e.g., in Akhtar et al, 1992, Trends Cell Bio., 2, 139; Delivery Strategies for Antisense Oligonucleotide Therapeutics, ed. Akhtar, 1995, Maurer et al, 1999, Mol. Memb. Biol, 16,
129-140; Hofiand and Huang, 1999, Handb. Exp. Pharmacol, 137, 165-192; and Lee et al, 2000, ACS Symp. Ser., 752, 184-192, U.S. Pat. No. 6,395,713 and PCT WO 94/02595 (each of which are incorporated herein by reference in their entireties). The siRNA of the present invention can also be administered in combination with other therapeutic compounds, either administrated separately or simultaneously, e.g., as a combined unit dose. In one embodiment, the invention includes a pharmaceutical composition comprising one or more siRNA according to the present invention in a physio logically/pharmaceutically acceptable excipient, such as a stabilizer, preservative, diluent, buffer, and the like.
Dosage levels for the medicament and pharmaceutical compositions of the invention can be determined by those skilled in the art by routine experimentation. In one embodiment, a unit dose contains between about 0.01 mg/kg and about 100 mg/kg body weight of siRNA. In one embodiment, the dose of siRNA is about 10 mg/kg and about 25 mg/kg body weight. In one embodiment, the dose of siRNA is about 1 mg/kg and about 10 mg/kg body weight. In one embodiment, the dose of siRNA is about 0.05 mg/kg and about 5 mg/kg body weight. In another embodiment, the dose of siRNA is about 0.1 mg/kg and about 5 mg/kg body weight. In another embodiment, the dose of siRNA is about 0.1 mg/kg and about 1 mg/kg body weight. In another embodiment, the dose of siRNA is about 0.1 mg/kg and about 0.5 mg/kg body weight. In another embodiment, the dose of siRNA is about 0.5 mg/kg and about 1 mg/kg body weight.
In one aspect, the pharmaceutical composition is a sterile injectable aqueous suspension or solution. In one aspect, the pharmaceutical composition is in lyophilized form. In one embodiment, the pharmaceutical composition comprises lyophilized lipoplexes, wherein the lipoplexes comprises a siRNA of the present invention. In another embodiment, the pharmaceutical composition comprises an aqueous suspension of lipoplexes, wherein the lipoplexes comprises a siRNA of the present invention.
The pharmaceutical compositions and medicaments of the present invention may be administered to a subject (mammal) in the disclosed methods of treatment. In one embodiment, the mammal is selected from the group consisting humans, dogs, cats, horses, cattle, pig, goat, sheep, mouse, rat, hamster and guinea pig. In one embodiment, the mammal is a human. In another embodiment, the mammal is a non-human mammal.
In one embodiment, the present invention is related to lipoplexes comprising a siRNA according to the present invention. Such lipoplexes consist of siRNA and liposomes. Such
lipoplexes may be used to deliver the siR A of the invention to a target cell either in vitro or in vivo.
In one aspect, the lipoplex has a zeta-potential of about 40 to 55 mV, preferably about 45 to 50 mV. The size of the lipoplex according to the present invention is about 80 to 200 nm, about 100 to 140 nm or about 110 nm to 130 nm, as determined by dynamic light scattering (QELS) such as, e. g., by using an N5 submicron particle size analyzer from Beckman Coulter according to the manufacturer's recommendation.
In one embodiment, the liposome as forming part of the lipoplex is a positively charged liposome consisting of:
a) about 50 mol% -arginyl-2,3-diaminopropionic acid-N-palmityl-N-oleyl-amide trihydrochloride, preferably P-(L-arginyl)-2,3-L-diaminopropionic acid-N-palmityl-N-oleyl- amide tri-hydrochloride,
b) about 48 to 49 mol% l,2-diphytanoyl-sn-glycero-3-phosphoethanolamine (DPhyPE), and
c) about 1 to 2 mol% l,2-distearoyl-sn-glycero-3-phosphoethanolamine-polyethylen- glycole, preferably N-(Carbonyl-methoxypolyethyleneglyco 1-2000)- 1 ,2-distearoyl-sn- glycero-3-phosphoethanolamine sodium salt.
The lipoplex and lipid composition forming the liposomes is preferably in a carrier however, the lipoplex can also be present in a lyophilised form. The lipid composition contained in a carrier usually forms a dispersion. More preferably, the carrier is an aqueous medium or aqueous solution as also further characterised herein. The lipid composition typically forms a liposome in the carrier, whereby such liposome preferably also contains the carrier inside.
The lipid composition contained in the carrier and the carrier, respectively, preferably has an osmolality of about 50 to 600 mosmole/kg, preferably about 250 - 350 mosmole/kg, and more preferably about 280 to 320 mosmole/kg.
The liposomes preferably are formed by the first lipid component and optionally also by the first helper lipid, preferably in combination with the first lipid component, preferably exhibit a particle size of about 20 to 200 nm, preferably about 30 to 100 nm, and more preferably about 40 to 80 nm. It is noted that the size of the particles follows a certain statistical distribution.
A further optional feature of the lipid composition in accordance with the present invention is that the pH of the carrier is preferably from about 4.0 to 6.0. However, also other
pH ranges such as from 4.5 to 8.0, preferably from about 5.5 to 7.5 and more preferably about 6.0 to 7.0 are within the present invention.
For realizing these particular features various measures may be taken. For adjusting the osmolarity, for example, a sugar or a combination of sugars is particularly useful. Insofar, the lipid composition of the present invention may comprise one or several of the following sugars: sucrose, trehalose, glucose, galactose, mannose, maltose, lactulose, inulin and raffinose, whereby sucrose, trehalose, inulin and raffinose are particularly preferred. In a particularly preferred embodiment, the osmolality mostly adjusted by the addition of sugar is about 300 mosmole/kg which corresponds to a sucrose solution of 270 mM or a glucose solution of 280 mM. Preferably the carrier is isotonic to the body fluid into which such lipid composition is to be administered. As used herein the term that the osmolality is mostly adjusted by the addition of sugar means that at least about 80 %, preferably at least about 90 % of the osmolality is provided by said sugar or a combination of said sugars.
If the pH of the lipid composition of the present invention is adjusted, this is done by using buffer substances which, as such, are basically known to the one skilled in the art. Preferably, basic substances are used which are suitable to compensate for the basic characteristics of the cationic lipids and more specifically of the ammonium group of the cationic head group. When adding basic substances such as basic amino acids and weak bases, respectively, the above osmolality is to be taken into consideration. The particle size of such lipid composition and the liposomes formed by such lipid composition is preferably determined by dynamic light scattering such as by using an N5 submicron particle size analyzer from Beckman Coulter according to the manufacturer's recommendation.
If the lipid composition contains one or several nucleic acid(s), such lipid composition usually forms a lipoplex (liposome-nucleic acid complex). The more preferred concentration of the overall lipid content in the lipoplex in preferably isotonic 270 mM sucrose or 280 mM glucose is from about 0.01 to 100 mg/ml, preferably 0.01 to 40 mg/ml and more preferably 0.01 to 25 mg/ml. It is to be acknowledged that this concentration can be increased so as to prepare a reasonable stock, typically by a factor of 2 to 3. It is also within the present invention that based on this, a dilution is prepared, whereby such dilution is typically made such that the osmolality is within the range specified above. More preferably, the dilution is prepared in a carrier which is identical or in terms of function and more specifically osmolality similar to the carrier used in connection with the lipid composition or in which the lipid composition is contained. In the embodiment, of the lipid composition of the present
invention whereby the lipid composition also comprises a nucleic acid, preferably a functional nucleic acid such as, but not limited to, a si NA, the concentration of the functional nucleic acid, preferably of siRNA in the lipid composition is about 0.2 to 0.4 mg/ml, preferably 0.28 mg/ml, and the total lipid concentration is about 1.5 to 2.7 mg/ml, preferably 2.17 mg/ml. It is to be acknowledged that this mass ratio between the nucleic acid fraction and the lipid fraction is particularly preferred, also with regard to the charge ratio thus realized. In connection with any further concentration or dilution of the lipid composition of the present invention, it is preferred that the mass ratio and the charge ratio, respectively, realized in this particular embodiment, is preferably maintained despite such concentration or dilution.
Such concentration as used in, for example, a pharmaceutical composition, can be either obtained by dispersing the lipid in a suitable amount of medium, preferably a physiologically acceptable buffer or any carrier described herein, or can be concentrated by appropriate means. Such appropriate means are, for example, ultra filtration methods including cross-flow ultra- filtration. The filter membrane may exhibit a pore width of 1,000 to 300,000 Da molecular weight cut-off (MWCO) or 5 nm to 1 μιη. Preferred is a pore width of about 10,000 to 100,000 Da MWCO. It will also be acknowledged by the one skilled in the art that the lipid composition more specifically the lipoplexes in accordance with the present invention may be present in a lyophilized form. Such lyophilized form is typically suitable to increase the shelve life of a lipoplex. The sugar added, among others, to provide for the appropriate osmolality, is used in connection therewith as a cryo -protectant. In connection therewith it is to be acknowledged that the aforementioned characteristics of osmolality, pH as well as lipoplex concentration refers to the dissolved, suspended or dispersed form of the lipid composition in a carrier, whereby such carrier is in principle any carrier described herein and typically an aqueous carrier such as water or a physiologically acceptable buffer, preferably an isotonic buffer or isotonic solution.
Another delivery system is for example described in European patent application EP 13 005 672.4 the disclosure of which is incorporated herein by reference. The delivery system described therein is a composition comprising a lipid composition, wherein the lipid composition consists of
Y Y" Y
(I) wherein n is any one of 1, 2, 3, and 4, wherein m is any one of 1 , 2 and 3,
Y" is an anion, wherein each of Rl and R2 is individually and independently selected from the group consisting of linear C 12-C 18 alkyl and linear C 12-C 18 alkenyl; a sterol compound, wherein the sterol compound is selected from the group consisting of cholesterol and stigmasterol; and a PEGylated lipid, wherein the PEGylated lipid comprises a PEG moiety and wherein the PEGylated lipid is selected from the group consisting of a PEGylated phosphoethanolamine of formula (II)
(II) wherein each of R3 and R4 is individually and independently linear C13-C17 alkyl, and p is any integer from 15 to 130; a PEGylated ceramide of formula (III)
wherein R5 is linear C7-C15 alkyl, and q is any integer from 15 to 130;
and a PEGylated diacylglycerol of formula (IV)
(IV) wherein each of R6 and R7 is individually and independently linear CI 1-C17 alkyl, and r is any integer from 15 to 130.
Diseases: One aspect of the present invention provides a siRNA molecule that reduces the expression of EDNl and that is useful for the treatment of human diseases and pathological conditions. The siRNA molecules can be used in combination with other therapeutic agents to enhance the therapeutic effects of a given treatment modality. In another aspect, the present invention provides reagents and methods useful for treating diseases and conditions characterized by undesirable or aberrant levels of EDNl activity in a cell.
Another aspect of the present invention is using the materials and methods for the treatment of conditions which can be ameliorated by decreasing constriction and constriction of vascular endothelial cells in particular. Another aspect of the present invention includes
treating pulmonary hypertension. A still other aspect of the present invention includes treating pre-eclampsia.
Example 1: Materials and Methods
If not specifically indicated differently in the further examples, the following materials and methods were used in the practicing of said examples.
Cell Lines
Endothelial MS-1 cells were obtained from ATCC/LGC Promochem and cultivated according to the manufacturer's recommendations. HUVECs (Lonza) were cultured in EGM- 2 bullet kit medium containing EBM2+ SingleQuots growth supplements.
Transfection of cultured cells and quantification of mRNA levels
Transfections of murine MS-1 endothelial cells and human HuVeCs with lipoplexed siRNA were carried out at indicated cell numbers and siRNA concentrations as previously described (Santel et al, 2006). Typically, cells were seeded at 40,000 cells/ wells and transfected 24 hours after seeding- 24 hours or 48 hours post transfection the cells were lysed and total RNA prepared (Invisorb Spin Cell RNA isolation kit, Invitek). Total RNA was subsequently used for mRNA quantification assays using the quantitative realtime RT-PCR approach (TaqMan, ABI). Normalized levels of the indicated mRNA were determined by the 2"l'D DCt^method showing expression levels for both the target and reference genes.
Determining endothelin 1 protein in supernatant of cells cultures
Endothelin 1 protein level in supernatant of cell cultures was determined by ELISA 72 hours post infection.
Lipid compositions
Atuplex is a lipid composition containing
a) 50 mol% P-(L-arginyl)-2,3-L-diaminopropionic acid-N-palmityl-N-oleyl- amide tri-hydrochloride);
b) 49 mol% l,2-diphytanoyl-sn-glycero-3-phosphoethanolamine (DPhyPE); and
c) 1 mol% N-(Carbonyl-methoxypolyethylenegly co 1-2000)- 1 ,2-distearoyl- sn- glycero-3-phosphoethanolamine sodium salt.
DACC9 is a lipid composition containing
70 mol% P-(L-arginyl)-2,3-L-diaminopropionic acid-N-palmityl-N-oleyl- amide tri-hydrochloride of the following formula:
29 mol% cholesterol; and
, whereby the charge ratio [lipids/phosphate oligo] is 8.4.
DACC10 is a lipid composition containing
70 mol% P-(L-arginyl)-2,3-L-diaminopropionic
amide tri-hydrochloride of the following formula:
29 mol% cholesterol; and
1 mol% mPEG-2000-Ceramide-C8 of the following formula:
, whereby the charge ratio [lipids/phosphate oligo] is 8.4.
Animal studies
Hypoxia-induced pulmonary hypertension
Male C57BL/6 mice (Harlan; 8-10 weeks) were randomly assigned to one of 7 groups, n=10 per group, for 21 days as follows (Fig. 7):
- (CI) Normoxia + Sucrose (2x/week, i.v.)
- (C2) Normoxia + Dacc9/EDN1 (2x/week, i.v.)
- (C3) Hypoxia/Sugen + Sucrose (2x/week, i.v.)
- (C4) Hypoxia/Sugen + Dacc9/Luc (2x/week, i.v.)
- (C5) Hypoxia/Sugen + Dacc9/EDN1 High dose (2x/week, i.v.)
- (C6) Hypoxia/Sugen + Dacc9/EDN1 Low dose (lx/week, i.v.)
- (C7) Hypoxia/Sugen + Bosentan (lOOmg/kgday, p.o.)
Normoxic animals were maintained at 21% 02 throughout. Hypoxic animals were exposed to 10% 02 in a normobaric chamber and administered the vascular endothelial growth factor (VEGF) receptor antagonist Sugen 5416 (suspension in 0.5%> [w/v] carboxymethylcellulose sodium, 0.9%> [w/v] sodium chloride, 0.4%> [v/v] polysorbate 80, 0.9%) [v/v] benzyl alcohol in deionized water; 20mg/kg, s.c, lx/week for 3 weeks). siRNA/vehicle treatments were given lx or 2x a week by tail vein injection, Bosentan (suspension in 0.1 % [w/v] carboxymethylcellulose sodium, polysorbate 80, 0.5%> [v/v]) was administered as daily gavage. Haemodynamic measurements were carried out within 72 hours of the last drug intervention.
Hemodynamic measurements
At 21 days, the right ventricular systolic pressure (RVSP) and mean arterial blood pressure (MABP) were recorded. Animals were removed from the chamber individually and anaesthetized immediately to minimize the time spent outside the hypoxic environment prior to haemodynamic measurement. Mice were anaesthetized with 1% isofluorane and placed
supine on a thermostatically controlled heating blanket (37oC). To measure MABP, the left common carotid artery was isolated and a fluid filled catheter introduced into the vessel. To measure RVSP, the right jugular vein was isolated and a Millar micro -manometer tipped catheter (Millar MicroTip 1.4F catheter, Millar Instruments, Houston, Texas, USA) introduced into the superior vena cava and then advanced into the right ventricle. Both MABP and RVSP were recorded onto a pre-calibrated PowerLab system (ADInstruments, Castle Hill, New South Wales, Australia) running Chart 5 software. Following measurement of pulmonary and systemic haemodynamics, animals were sacrificed by anaesthetic overdose and exsanguination, blood collected, the heart removed, and heart chamber weights measured to evaluate right ventricular hypertrophy (right ventricle to body weight ratio; RV/BW). In 6 animals, the lung was fixed by inflation with 10% formalin in PBS before paraffin embedding and sectioning. The remaining lung tissue, heart, liver and kidney were dissected and snap frozen in liquid N2. Serum was collected by allowing the blood to stand at room temperature for 45 minutes, then spinning at 4500rpm for 10 minutes. Serum was collected & stored at - 80°C.
Statistical evaluation
Results are expressed as mean±sem. Statistical analyses were performed by one-way ANOVA, with Bonferroni post-hoc tests, using GraphPad Prism version 5. P<0.05 denotes significance. The n value denotes the number of animals in each group.
Example 2: EDN1 specific siRNA molecules
The siRNA molecules which are directed to the mRNA encoding EDN1 and the various siRNA molecules directed to Luciferase and PTEN which were used in connection with the experiments and examples described herein, were synthesized by BioSpring (Frankfurt a. M., Germany) and are indicated in Fig. 1, Table 1 and Table 2 in terms of the sequences of both the first strand (being the antisense strand) and the second strand (being the sense strand) forming the double-stranded nucleic acid molecules of the present invention.
Luciferase-specific siRNA was formed by the following two strands:
Luc-23-2A (capital nucleotides modified
at the 2' position with an O-methyl UcGaAgUaUuCcGcGuAcGuGaU (SEQ group) ID NO: 18
Luc-23-2B (capital nucleotides modified
at the 2' position with an O-methyl aUcAcGuAcGcGgAaUaCuUcGa (SEQ group) ID NO: 19 siRNA PTENV10 was formed by the following two strands:
PTENV10 A: 5 ' UaAgUuCuAgCuGuGgUgGgUuA 3' (SEQ ID NO: 20) PTENV10B: 5' uAaCcCaCcAcAgCuAgAaCuCa 3' (SEQ ID NO: 21)
"A" stands for the antisense strand which is also referred to herein as the first strand; "B" stands for the sense strand which is also referred to herein as the second strand. Please note that any sequence indicated in the instant application is presented in 5'-> 3' direction, if not explicitly indicated to the contrary.
In certain embodiments, the antisense strands (as set forth in the Tables above) can be modified at the 2' position (e.g., with a 2'-0-methyl group) on one or more odd numbered nucleotide (or on each odd numbered nucleotide) and one or more even numbered nucleotides remain unmodified (e.g., a OH group is present at the 2' position on each of the unmodified nucleotides, for example each of the unmodified nucleotides is unmodified). Sense strands can be modified on one or more even numbered nucleotide (or on each even numbered nucleotide) at the 2' position (e.g., with a 2'-0-methyl group) and one or more odd numbered nucleotides can remain unmodified (e.g., a OH group is present at the 2' position on each of the unmodified nucleotides, for example each of the odd numbered nucleotides is unmodified).
Alternative embodiments provide for antisense strands (as set forth above) that are modified at the 2' position (e.g., with a 2'-0-methyl group) on one or more even numbered nucleotide (or on each even numbered nucleotide) and sense strands are modified on one or more odd numbered nucleotide (or on each odd numbered nucleotide) at the 2' position (e.g.,
with a 2'-0-methyl group). One or more unmodified nucleotide is present in both the sense and antisense strands in these alternative embodiments (e.g., the unmodified nucleotides have a OH group at the 2' position in each of these alternative embodiments). In certain embodiments, each odd numbered nucleotide is unmodified in the antisense strand and each even numbered nucleotide is unmodified in the sense strand for the alternative embodiments discussed in this paragraph.
The siRNA molecules of Fig. 1 were tested in vitro for inhibition of EDN1 target expression in cell cultures using degradation of mEDNl mRNA and PTEN mRNA as readout. The results are indicated in Fig. 2A and Fig. 2B. As may be taken from Fig. 2A siRNA consisting of EDNl-hmr-2A and EDNl-hmr-2B was particularly effective, whereby siRNA consisting of EDNl-hm4A and EDNl-hm4B, siRNA consisting of of EDNl-hmr6A and EDNl-hmr-6B, and siRNA consisting of EDNl-hmr-8A and EDNl-hmr-8B also exhibited some efficiency.
The siRNA molecules of Fig. 1 were also tested in vitro for inhibition of EDN1 target expression in cell cultures using endothelin 1 protein concentration in the supernatant of cell cultures of murine MSI cells and human HuVe cells as read-out. The results are indicated in Figs. 3 and 4. The results as to particularly effective siRNA molecules as shown in connection with Fig. 2 was confirmed. Example 4: Animal studies - reduction of EDN-1 expression in mice
The purpose of these animal studies was to evaluate different delivery systems for targeting EDN1. The experimental set-up and treatment scheme for the mice is outlined in Fig. 5.
Mice were treated with a single dose of EDNl-hmr2 siRNA (consisting of EDNl-hmr 2A and EDNl-hmr-2B) formulated with DACC9, DACC10 or with three doses of EDN1- hmr2 siRNA formulated with Atuplex by bolus application. Control lipoplexes contained siRNA for SFltl . Target gene expression was analyzed in lung tissues and in serum 48 hours post treatment.
The results are shown in Figs. 6 A and 6B.
Example 5: Animal studies - effect of DACC9 formulated siRNA targeting EDN1 in hypoxia-induced pulmonary hypertension
The purpose of these animal studies was to show therapeutic effect of DACC9 formulated siRNA targeting EDN1 in hypoxia- induced pulmonary hypertension. The siRNA used was EDNl-hmr2. The DACC9 formulated siRNA composition is also referred to herein as Dacc9/EDN1.
The treatment scheme is shown in Fig. 7.
The results are shown in Fig. 8 to 11.
Effect of Dacc9/EDN1 on right ventricular systolic pressure (RVSP)
Hypoxia induced a significant increase in RVSP (normoxia: 28.76±1.3mmHg vs. hypoxia: 39.20±2.5 mmHg). The higher RVSP in normoxic mice was reduced in a dose-dependent manner by Dacc9/EDN1 (Fig. 8). The Dacc9/Luc control had no effect on RVSP, nor did the Dacc9/EDN1 high dose formulation alter RVSP in normoxic control animals. There was also a significant reduction in RVSP in the bosentan-treated group (Fig. 8).There was no significant difference in MABP observed in any of the groups (Fig. 9). Effect of Dacc9/EDN1 on right ventricular hypertrophy (RVH)
Hypoxia induced a significant increase in RVH (normoxia: 0.09±003 g vs. hypoxia: 0.113±003 g). There was a trend towards reduced RVH in the high and low dose Dacc9/EDN1 groups, as well as the Bosentan-treated animals (Figs. 10 and 11). However, these differences failed to achieve statistical significance. Interestingly, this index of disease severity was more greatly affected by the low dose formulation compared to the high dose siRNA. Since treatment with Sugen causes a drop in body weight (BW) over the time-course of the study (data not shown), RVH is expressed as RV:BW ratio to correct for this.
The inventors have shown for the first time that Dacc9/EDN1 is an effective intervention for the prevention of hypoxia-induced PH. The efficacy of Dacc9/EDN1 is approximately equivalent to Bosentan. Dacc9/EDN1 produced a pulmonary-selective effect since MABP remained unchanged.
Example 6 - Hypoxia-induced pulmonary hypertension
Male C57BL/6 mice (Harlan; 8-10 weeks) were randomly assigned to one of 6
groups, n=10 per group, for 35 days as described below and depicted in Figure 14:
(CI) Normoxia + Sucrose (2x/week, i.v.)
(C2) Hypoxia/Sugen + Sucrose (2x/week, i.v.)
(C3) Hypoxia/Sugen + Dacc9/Luc (2x/week, i.v.)
(C4) Hypoxia/Sugen + Dacc9/EDNl-flu (2x/week, i.v.)
(C5) Hypoxia/Sugen + Dacc9/EDN1 (2x/week, i.v.)
(C6) Hypoxia/Sugen + Endothelin receptor antagonist (ERA; Bosentan
(lOOmg/kg/day, p.o.)
Normoxic animals were maintained at 21% 02 throughout. Hypoxic animals were exposed to 10% 02 in a normobaric chamber and administered the vascular endothelial growth factor (VEGF) receptor antagonist Sugen 5416 (suspension in 0.5%> [w/v] carboxymethylcellulose sodium, 0.9%) [w/v] sodium chloride, 0.4%> [v/v] polysorbate 80, 0.9%> [v/v] benzyl alcohol in deionized water; 20mg/kg, s.c, lx/week for 3 weeks). siRNA (2mg/kg) treatment and vehicle (sucrose) were initiated at day 14 once the PH phenotype had established ('reversal protocol') and administered 2x a week by tail vein injection. Bosentan (suspension in 0.1% [w/v] carboxymethylcellulose sodium, polysorbate 80, 0.5%> [v/v]) was administered as daily gavage. Haemodynamic measurements were carried out within 72 hours of the last drug intervention.
Hemodynamic measurements
At 35 days, the right ventricular systolic pressure (RVSP) and mean arterial blood pressure (MABP) were recorded. Animals were removed from the chamber individually and anaesthetized immediately to minimize the time spent outside the hypoxic environment prior to haemodynamic measurement. Mice were anaesthetized with 1% isofluorane and placed supine on a thermostatically controlled heating blanket (37oC). To measure MABP, the left common carotid artery was isolated and a fluid filled catheter introduced into the vessel. To measure RVSP, the right jugular vein was isolated and a Millar micro -manometer tipped catheter (Millar MicroTip 1.4F catheter, Millar Instruments, Houston, Texas, USA) introduced into the superior vena cava and then advanced into the right ventricle. Both MABP and RVSP were recorded onto a pre-calibrated PowerLab system (ADInstruments,
Castle Hill, New South Wales, Australia) running Chart 5 software.
Following measurement of pulmonary and systemic haemodynamics, animals were sacrificed by anaesthetic overdose and exsanguination, blood collected, the heart removed, and heart
chamber weights measured to evaluate right ventricular hypertrophy (RVH; right ventricle to left ventricle plus septum ratio; RV/[LV+S]). In 6 animals, the lung was fixed by inflation with 10% formalin in PBS before paraffin embedding and sectioning. The remaining lung tissue, liver and kidney were dissected and snap frozen in liquid N2. Whole blood samples were collected in EDTA tubes at baseline (tail vein; 3 animals per group) and at day 35 (cardiac puncture; all animals) and plasma obtained by spinning at 4500rpm for 10 min at 4oC. Plasma was separated and stored at -80oC.
Effect of Dacc9/EDN1 on body weight and mean arterial blood pressure
(MABP)
Body weight changes as a result of hypoxia/Sugen exposure were relatively consistent across groups (Figure 19) suggesting the treatments were equally well tolerated. There were no significant differences in MABP observed across any of the groups (Figure 17). Effect of Dacc9/EDN1 on right ventricular systolic pressure (RVSP)
Hypoxia induced a significant increase in RVSP (normoxia: 22.75±1.5mmHg vs. hypoxia: 39.83±2.2 mmHg) that was significantly reversed in the presence of Dacc9/EDN1 and Bosentan (Figure 15). The Dacc9/Luc control had no effect on RVSP, and whilst the modified siRNA Dacc9/EDNl-flu showed a trend towards a reduction in RVSP this did not reach statistical significance.
Effect of Dacc9/EDN1 on right ventricular hypertrophy (RVH)
Hypoxia induced a significant increase in RVH (RV/[LV+S]: normoxia: 0.290±0.01 vs. hypoxia: 0.357±0.01). There was a trend towards reduced RVH in the Dacc9/EDNl-flu and Bosentan-treated animals, but these modest changes were mirrored by administration of the DACC9/Luc control (Figure 16). However, these differences failed to achieve statistical significance. Interestingly, only the Dacc9/EDN1 group showed a significant reversal of the hypoxia- induced RVH in this model (Figure 16). Effect of Dacc9/EDN1 on EDNl mRNA level in the lungs
EDNl mRNA expression levels in lung tissue were determined after the end of the treatment by qRT-PCR. It has been showed that EDNl mRNA levels are elevated by hypoxia/Sugen treatment, but reduced in DACC9/EDN1 treatment groups.
It has been surprisingly found by the inventors that Dacc9/EDN1 is an effective intervention for the reversal of hypoxia-induced PH. The treatment caused a significant reduction in the development of elevated pulmonary artery pressure (i.e. RVSP) and also abrogated the accompanying RVH. The efficacy of Dacc9/EDN1 is approximately equivalent to Bosentan at the dose employed, with a superior activity against the hypoxia-induced increases in RVSP and RVH. The mRNA knockdown of EDN1 by Dacc9/EDN1 produced a pulmonary-selective effect since MABP remained unchanged.
The features of the present invention disclosed in the specification, the claims, the sequence listing and/or the drawings may both separately and in any combination thereof be material for realizing the invention in various forms thereof.
REFERENCES
Aird, W.C. 2003. The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome. Blood. 101 :3765-77.
Daly, C, E. Pasnikowski, E. Burova, V. Wong, T.H. Aldrich, J. Griffiths, E. Ioffe, T.J. Daly,
J.P. Fandl, N. Papadopoulos, D.M. McDonald, G. Thurston, G.D. Yancopoulos, and Maniatis, N.A., and S.E. Orfanos. 2008. The endothelium in acute lung injury/acute
respiratory distress syndrome. Curr Opin Crit Care. 14:22-30.
Matute-Bello, G., C.W. Frevert, O. Kajikawa, S.J. Skerrett, R.B. Goodman, D.R. Park, and T.R. Martin. 2001. Septic shock and acute lung injury in rabbits with peritonitis: failure of the neutrophil response to localized infection. Am J Respir Crit Care Med.
163:234-43.
Opitz, B., A. Puschel, B. Schmeck, A.C. Hocke, S. Rosseau, S. Hammerschmidt, R.R.
Schumann, N. Suttorp, and S. Hippenstiel. 2004. Nucleotide-binding oligomerization domain proteins are innate immune receptors for internalized Streptococcus pneumoniae. J Biol Chem. 279:36426-32.
Paton, J.C., J.K. Morona, S. Harrer, D. Hansman, and R. Morona. 1993. Immunization of mice with Salmonella typhimurium C5 aroA expressing a genetically toxoided derivative of the pneumococcal toxin pneumolysin. Microb Pathog. 14:95-102.
Santel, A., M. Aleku, O. Keil, J. Endruschat, V. Esche, B. Durieux, K. Loffler, M. Fechtner,
T. Rohl, G. Fisch, S. Dames, W. Arnold, K. Giese, A. Klippel, and J. Kaufmann.
2006a. RNA interference in the mouse vascular endothelium by systemic
administration of siRNA-lipoplexes for cancer therapy. Gene Ther. 13 : 1360-70. Santel, A., M. Aleku, O. Keil, J. Endruschat, V. Esche, G. Fisch, S. Dames, K. Loffler, M.
Fechtner, W. Arnold, K. Giese, A. Klippel, and J. Kaufmann. 2006b. A novel siRNA- lipoplex technology for RNA interference in the mouse vascular endothelium. Gene
Ther. 13 : 1222-34.
Schmeck, B., J. Zahlten, K. Moog, V. van Laak, S. Huber, A.C. Hocke, B. Opitz, E.
Hoffmann, M. Kracht, J. Zerrahn, S. Hammerschmidt, S. Rosseau, N. Suttorp, and S. Hippenstiel. 2004. Streptococcus pneumoniae-induced p38 MAPK-dependent phosphorylation of RelA at the interleukin-8 promotor. J Biol Chem. 279:53241-7. Ware, L.B. 2006. Pathophysiology of acute lung injury and the acute respiratory distress syndrome. Semin Respir Crit Care Med. 27:337-49.
Claims
1. An siRNA comprising an antisense strand and a sense strand, wherein all or a portion of said antisense strand comprises an antisense duplex region, wherein all or a portion of said sense strand comprises a sense duplex region, wherein said antisense duplex region is at least partially complementary to said sense duplex region, wherein said siRNA comprises a duplex region consisting of said antisense duplex region and said sense duplex region, and wherein: a) said antisense strand comprises a nucleotide sequence of SEQ ID NO: 1, 3, 5 or 7; or b) said antisense strand comprises an antisense duplex region, all or a portion of which, is complementary to a portion of SEQ ID NO: 17.
2. The siRNA of claim 1, wherein said sense strand comprises a nucleotide sequence of SEQ ID NO: 2, 4, 6 or 8.
3. The siRNA of any one of claims 1 to 2, wherein said antisense strand and said sense strand are each from 19 to 25 nucleotides in length.
4. The siRNA of any one of claims 1 to 3, wherein said duplex region is from 19 to 25 nucleotides.
5. The siRNA of claim 4, wherein said duplex region consists of 19 to 25 consecutive nucleotide base pairs.
6. The siRNA of any one of claims 1 to 5, wherein said siRNA:
a) is blunt ended at both ends;
b) has an overhang at one end and a blunt end at the other; or
c) has an overhang at both ends.
7. The siRNA of any one of claims 1 to 6, wherein said siRNA is selected from the group consisting of
5 uuucaauuugugcauuccu 3 ' (SEQ ID NO: 1)
5'aggaaugcacaaauugaaa 3' (SEQ ID NO: 2),
5 'auacauagaccaauauggc 3 ' (SEQ ID NO: 3)
5 'gccauauuggucuauguau 3 ' (SEQ ID NO: 4),
5 uguuucuggagcuccuugg 3 ' (SEQ ID NO: 5)
5 'ccaaggagcuccagaaaca 3 ' (SEQ ID NO: 6), and
5 uggacuuuggaguuucucc 3 ' (SEQ ID NO: 7)
5 'ggagaaacuccaaagucca 3 ' (SEQ ID NO: 8).
8. The siRNA of claim 7, wherein the siRNA is
5 uuucaauuugugcauuccu 3 ' (SEQ ID NO: 1)
5'aggaaugcacaaauugaaa 3' (SEQ ID NO: 2).
9. The siRNA of any one of claims 1 to 8, wherein at least one nucleotide of the antisense strand and/or the sense strand is modified by a first modification.
10. The siRNA of claim 9, wherein at least one nucleotide of the antisense strand is modified by the first modification.
11. The siRNA of claim 9, wherein at least one nucleotide of the antisense strand and of the sense strand is modified by the first modification.
12. The siRNA of any one of claims 9 to 11, wherein at least one nucleotide of the antisense strand and/or the sense strand is modified by a second modification, wherein the at least one nucleotide of the antisense strand and/or the sense strand which is modified by a second modification is different from the at least one nucleotide of the antisense strand and/or the sense strand which is modified by a first modification, and wherein the first modification is different from the second modification.
13. The siRNA of claim 12, wherein at least one nucleotide of the antisense strand is modified by the second modification.
14. The siRNA of claim 12, wherein at least one nucleotide of the antisense strand and of the sense strand is modified by the second modification.
15. The siRNA of any one of claims 9 to 14, wherein one or more alternating nucleotides of the antisense strand and/or of the sense strand are modified by a first modification.
16. The siRNA of claim 15, wherein each of the odd numbered nucleotides of the antisense strand are modified by the first modification.
17. The siRNA of any one of claims 15 to 16, wherein each of the even numbered nucleotides of the antisense strand are modified by a second modification, wherein the second modification is different from the first modification.
18. The siRNA of any one of claims 15 to 17, wherein each of the even numbered nucleotides of the sense strand are modified by the first modification.
19. The siRNA of any one of claims 15 to 18, wherein each of the odd numbered nucleotides of the sense strand are modified by the second modification, wherein the second modification is different from the first modification.
20. The siRNA of any one of claims 15 to 19, wherein the first modification and the second modification are each and individually selected from the group comprising 2'-0- methyl modification and 2'-F modification.
21. The siRNA of any one of claims 9 to 20, wherein the first modification is 2'-0-methyl modification.
22. The siRNA of any one of claims 12 to 21, wherein the second modification is 2'-F modification.
23. The siRNA of any one of claims 1 to 22, wherein the antisense strand and/or the sense strand comprises at least one phosphorothioate bond.
24. The siRNA of any one of claims 1 to 23, wherein the siRNA is selected from the group consisting of
5 'UuUcAaUuUgUgCaUuCcU 3 ' (SEQ ID NO: 1)
5'aGgAaUgCaCaAaUuGaAa 3' (SEQ ID NO: 2),
5 'AuAcAuAgAcCaAuAuGgC 3 ' (SEQ ID NO: 3)
5 gCcAuAuUgGuCuAuGuAu 3 ' (SEQ ID NO: 4),
5 'UgUuUcUgGaGcUcCuUgG 3 ' (SEQ ID NO: 5)
5 'cGaAgGaGcUcCaGaAaCa 3 ' (SEQ ID NO: 6), and
5 'UgGaCuUuGgAgUuUcUcC 3 ' (SEQ ID NO: 7)
5 'gGaGaAaCuCcAaAgUcCa 3 ' (SEQ ID NO: 8), wherein a capital letter for a nucleotide indicates that said nucleotide is 2'-0-methyl modified.
25. The siRNA of claim 1, wherein the siRNA is
5 'UuUcAaUuUgUgCaUuCcU 3 ' (SEQ ID NO: 1)
5'aGgAaUgCaCaAaUuGaAa 3' (SEQ ID NO: 2), wherein a capital letter for a nucleotide indicates that said nucleotide is 2'-0-methyl modified.
26. The siRNA of any one of claims 1 to 25, wherein the siRNA is selected from the group consisting of
5 UuUcAaUuUgUgCaUuCcU 3 ' (SEQ ID NO: 1)
5'aGgAaUgCaCaAaUuGaAa 3' (SEQ ID NO: 2),
5 'AuAcAuAgAcCaAuAuGgC 3 ' (SEQ ID NO: 3)
5 gCcAuAuUgGuCuAuGuAu 3 ' (SEQ ID NO: 4),
5 UgUuUcUgGaGcUcCuUgG 3 ' (SEQ ID NO: 5)
5 'cGaAgGaGcUcCaGaAaCa 3 ' (SEQ ID NO: 6), and
5 UgGaCuUuGgAgUuUcUcC 3 ' (SEQ ID NO: 7)
5 gGaGaAaCuCcAaAgUcCa 3 ' (SEQ ID NO: 8), wherein a capital letter for a nucleotide indicates that said nucleotide is 2'-0-methyl modified and an underlined nucleotide indicates that said nucleotide is 2'-F modified.
27. The siRNA of any one of claims 1 to 26, wherein the siRNA is
5 UuUcAaUuUgUgCaUuCcU 3 ' (SEQ ID NO: 1)
5'aGgAaUgCaCaAaUuGaAa 3' (SEQ ID NO: 2), wherein a capital letter for a nucleotide indicates that said nucleotide is 2'-0-methyl modified and an underlined nucleotide indicates that said nucleotide is 2'-F modified.
28. The siRNA of any one of claims 1 to 27, wherein the siRNA is targeting a nucleic acid coding for endothelin 1 (EDN1).
29. The siRNA of any one of claims 1 to 28, wherein the siRNA is silencing the expression of a nucleic acid coding for endothelin 1 (ENDl) or wherein the siRNA is post- transcriptionally inhibiting the expression of a nucleic acid coding for endothelin 1 (ENDl).
30. A lipoplex comprising an siRNA according to any one of claims 1 to 29 and a lipid composition, wherein the lipid composition consists of a cationic lipid of formula (I)
wherein m is any one of 1, 2 and 3,
Y" is an anion, wherein each of Rl and R2 is individually and independently selected from the group consisting of linear C12-C18 alkyl and linear C12-C18 alkenyl; a sterol compound, wherein the sterol compound is selected from the group consisting of cholesterol and stigmasterol; and a PEGylated lipid, wherein the PEGylated lipid comprises a PEG moiety and wherein the PEGylated lipid is selected from the group consisting of a PEGylated phosphoethanolamine of formula (II)
wherein each of R3 and R4 is individually and independently linear C13-C17 alkyl, and p is any integer from 15 to 130; a PEGylated ceramide of formula (III)
wherein R5 is linear C7-C15 alkyl, and q is any integer from 15 to 130;
and a PEGylated diacylglycerol of formula (IV)
wherein each of R6 and R7 is individually and independently linear CI 1 -CI 7 alkyl, and r is any integer from 15 to 130.
31. The lipoplex of claim 30, wherein Rl and R2 are different from each other.
32. The lipoplex of claim 30, wherein Rl and R2 are the same.
33. The lipoplex of any one of claims 30 to 32, wherein each of Rl and R2 is individually and independently selected from the group consisting of C12 alkyl, C14 alkyl, C16 alkyl, CI 8 alkyl, C12 alkenyl, CI 4 alkenyl, C16 alkenyl and CI 8 alkenyl.
34. The lipoplex of claim 33, wherein each of C12 alkenyl, C14 alkenyl, C16 alkenyl and C18 alkenyl comprises one or two double bonds.
35. The lipoplex of claim 34, wherein CI 8 alkenyl is CI 8 alkenyl with one double bond between C9 and CIO, preferably cis-9-octadecyl].
36. The lipoplex of any one of claims 3 to 35, wherein Rl and R2 are different and Rl is palmityl and R2 is oleyl.
37. The lipoplex of any one of claims 29 to 36, wherein Rl and R2 are different and wherein Rl is lauryl and R2 is myristyl.
38. The lipoplex of any one of claims 30 to 37, wherein the cationic lipid is a compound of formula (la)
Y Y
(la)
39. The lipoplex of any one of claims 30 to 38, wherein Y" is selected from the group comprising halogenids, acetate and trifluoroacetate.
40. The lipoplex of claim 39, wherein Y" is CI".
41. The lipoplex of any one of claims 30 to 40, wherein the cationic lipid is P-arginyl-2,3- diamino propionic acid-N-palmityl-N-oleyl-amide trihydrochloride of formula (lb):
43. The lipoplex of any one of claims 30 to 40 wherein the cationic lipid is ε-arginyl-lysine-N- lauryl-N-myristyl-amide trihydrochloride of formula (Id):
(Id)
44. The lipoplex of any one of claims 30 to 43, wherein the sterol compound is cholesterol.
45. The lipoplex of any one of claims 31 to 43, preferably claim 43, wherein the sterol compound is cholesterol.
46. The lipoplex of any one of claims 30 to 43, wherein the sterol compound is stigmasterin.
47. The lipoplex of any one of claims 41 to 43, preferably claim 43, wherein the sterol compound is stigmasterin.
48. The lipoplex of any one of claims 30 to 47, preferably any one of claims 41 to 43, more preferably any one of claims 45 and 47, wherein the PEG moiety of the PEGylated lipid has a molecular weight from about 800 to about 5000 Da.
49. The lipoplex of claim 48, wherein the molecular weight of the PEG moiety of the PEGylated lipid is about 800 Da.
50. The lipoplex of claim 48, wherein the molecular weight of the PEG moiety of the PEGylated lipid is about 2000 Da.
51. The lipoplex of claim 48, wherein the molecular weight of the PEG moiety of the PEGylated lipid is about 5000 Da.
52. The lipoplex of any one of claims 30 to 51, preferably any one of claims 48 to 51, wherein the PEGylated lipid is a PEGylated phosphoethanolamine of formula (II), wherein each of R3 and R4 is individually and independently linear C13-C17 alkyl, and p is any integer from 18, 19 or 20, or from 44, 45 or 46 or from 113, 114 or 115.
53. The lipoplex of claim 52, wherein R3 and R4 are the same.
54. The lipoplex of claim 52, wherein R3 and R4 are different.
55. The lipoplex of any one of claims 52 and 54, wherein each of R3 and R4 is individually and independently selected from the group consisting of C13 alkyl, CI 5 alkyl and CI 7 alkyl.
56. The lipoplex of any one of claims 30 to 55, preferably one of claims 41 to 43, more preferably any one of claims 43 and 45, wherein the PEGylated phosphoethanolamine of formula (II) is
(Ila)
57. The lipoplex of any one of claims 30 to 55, preferably one of claims 41 to 43, more preferably any one of claims 16 and 18, wherein the PEGylated phosphoethanolamine of formula (II) is
b)
58. The lipoplex of any one of claims 30 to 51, preferably any one of claims 48 to 51, wherein the PEGylated lipid is a PEGylated ceramide of formula (III), wherein R5 is linear C7-C15 alkyl, and q is any integer from 18, 19 or 20, or from 44, 45 or 46 or from 113, 114 or 115.
59. The lipoplex of claim 58, wherein R5 is linear C7 alkyl.
60. The lipoplex of claim 59, wherein R5 is linear CI 5 alkyl.
61. The lipoplex of any one of claims 30 to 51 and 58 to 60, preferably one of claims 41 to 43, more preferably any one of claims 45 and 47, wherein the PEGylated ceramide of formula (III) is
(Ilia)
The lipoplex of any one of claims 30 to 51 and 58 to 60, preferably one of claims 41 to 43, preferably any one of claims 45 and 47, wherein the PEGylated ceramide of formula (III) is
(nib)
63. The lipoplex of any one of claims 30 to 51, preferably any one of claims 48 to 51, wherein the PEGylated lipid is a PEGylated diacylglycerol of formula (IV), wherein each of R6 and R7 is individually and independently linear CI 1 -CI 7 alkyl, and r is any integer from 18, 19 or 20, or from 44, 45 or 46 or from 113, 114 or 115.
64. The lipoplex of claim 63, wherein R6 and R7 are the same.
65. The lipoplex of claim 63, wherein R6 and R7 are different.
66. The lipoplex of any one of claims 63 to 65, wherein each of R6 and R7 is individually and independently selected from the group consisting of linear CI 7 alkyl, linear CI 5 alkyl and linear CI 3 alkyl.
67. The lipoplex of any one of claims 30 to 51 and 63 to 66, preferably one of claims 41 to 43, more preferably any one of claims 45 and 47, wherein the PEGylated diacylglycerol of formula (IV) is
(IVa).
68. The lipoplex of any one of claims 30 to 51 and 63 to 65, preferably one of claims 41 to 43, more preferably any one of claims 45 and 47, wherein the PEGylated diacylglycerol of formula (IV) is
The lipoplex of any one of claims 30 to 51 and 63 to 65, preferably one of claims 41 to 43, preferably any one of claims 45 and 47, wherein the PEGylated diacylglycerol of formula (IV) is
•ginyl-2,3-diamino propionic acid-N-palmityl-N-oleyl-amide trihydrochloride
P-arginyl-2,3-diamino propionic acid-N-lauryl-N-myristyl-amide trihydrochloride
wherein the sterol compound is selected from the group consisting of cholesterol and stigmasterin; and wherein the PEGylated lipid is a PEGylated phosphoethanolamine of formula (II), wherein the PEGylated phosphoethanolamine is selected from the group consisting of l,2-distearoyl-i,n-glycero-3-phosphoethanolamine-N-[methoxy(poly ethylene glycol)-2000]
71. The lipoplex of any one of claims 30 to 69, wherein the cationic lipid of formula (I) is selected from the group consisting of
P-arginyl-2,3-diamino propionic acid-N-palmityl-N-oleyl-amide trihydrochloride
P-arginyl-2,3-diamino propionic acid-N-lauryl-N-myristyl-amide trihydrochloride
and ε-arginyl-lysine-N-lauryl-N-myristyl-amide trihydrochloride
wherein the sterol compound is selected from the group consisting of cholesterol and stigmasterin; and wherein the PEGylated lipid is a PEGylated ceramide of formula (III), wherein the PEGylated ceramide is selected from the group consisting of
N-octanoyl-sphingosine- 1 - {succinyl[methoxy(polyethylene glycol)2000] }
and
72. The lipoplex of any one of claims 30 to 69, wherein
the cationic lipid of formula (I) is selected from the group consisting of
P-arginyl-2,3-diamino propionic acid-N-lauryl-N-myristyl-amide trihydrochloride
wherein the sterol compound is selected from the group consisting of cholesterol and stigmasterin; and wherein the PEGylated lipid is a PEGylated diacylglycerol of formula (IV), wherein the PEGylated diacylglycerol is selected from the group consisting of
1 ,2-Distearoyl-sn-glycerol [methoxy(polyethylene glycol)2000]
1 ,2-Dipalmitoyl-sn-glycerol [methoxy(polyethylene glycol)2000]
73. The lipoplex of any one of claims 30 to 72, preferably of any one of claims 70 to 72, wherein the cationic lipid is P-arginyl-2,3-diamino propionic acid-N-palmityl-N-oleyl-amide trihydrochloride
the sterol compound is cholesterol, and the PEGylated lipid is PEGylated phosphoethanolamine of formula (II) is l,2-distearoyl-i,n-glycero-3-phosphoethanolamine-N-[methoxy(poly ethylene glycol)-2000]
74. The lipoplex of any of clams 30 to 73, preferably any of claims 70 to 73 and more preferably claim 73, wherein, in the lipid composition, the content of the cationic lipid composition is from about 65 mole % to about 75 mole %, the content of the sterol compound is from about 24 mole % to about 34 mole % and the content of the PEGylated lipid is from about 0.5 mole % to about 1.5 mole %, wherein the sum of the content of the cationic lipid, of the sterol compound and of the PEGylated lipid for the lipid composition is 100 mole %.
75. The lipoplex of claim 74, wherein, in the lipid composition, the content of the cationic lipid is about 70 mole %, the content of the sterol compound is about 29 mole % and the content of the PEGylated lipid is about 1 mole %.
76. A lipoplex of any of the preceding claims, wherein the lipid composition is as follows:
29 mole % of cholesterol, and
1 mole % of l,2-distearoyl-i,n-glycero-3-phosphoethanolamine-N-[methoxy(poly ethylene glycol)-2000]
A lipoplex of any of the preceding claims, wherein the lipid composition is as follows
70 mol% -(L-arginyl)-2,3-L-diaminopropionic acid-N-palmityl-N-oleyl- amide tri- hydrochloride of the following formula:
29 mol% cholesterol; and
1 mol% mPEG-2000-Ceramide-C8 of the following formula:
The lipoplex of any one of claims 30 to 77, wherein the composition comprises , preferably the carrier is a pharmaceutically acceptable carrier.
79. The lipoplex of claim 78, wherein the carrier is selected from the group comprising water, an aqueous solution, preferably an isotonic aqueous solution, a salt solution, preferably an isotonic salt solution, a buffer, preferably an isotonic buffer, and a water miscible solvent.
80. The lipoplex of claim 79, wherein the carrier is a water miscible solvent and wherein the water miscible solvent is selected from the group consisting of ethanol and tertiary butanol
81. The lipoplex of any one of claims 78 to 80, wherein the carrier is an aqueous sucrose solution, preferably a 270 mM aqueous sucrose solution.
82. The lipoplex according to any one of the preceding claims, wherein the lipid composition is as follows:
70 mole % of P-arginyl-2,3-diamino propionic acid-N-palmityl-N-oleyl-amide trihydrochloride
29 mole % of cholesterol, and
1 mole % of PEGylated phosphoethanolamine of formula (II) is l,2-distearoyl-i,n-glycero-3-phosphoethanolamine-N-[methoxy(poly ethylene glycol)-2000]
wherein the composition comprises a 270 mM aqueous sucrose solution.
83. The lipoplex according to any one of claims 78 to 82, wherein the lipid composition forms particles in the carrier.
84. The lipoplex of claim 83, wherein the particles have a Z-average size according to DLS measurement of about 30 nm to about 150 nm.
85. The lipoplex of claim 84, wherein the particles have a Z-average size according to DLS measurement of about 50 nm to about 100 nm.
86. The lipoplex of any one of claims 83 to 85, wherein the Z-average size according to DLS measurement of the particles is about 60 - 80 nm as determined by dynamic light scattering.
87. The lipoplex of any one of claims 30 to 86, preferably any one of claims 78 to 86, wherein the composition, determined at a temperature of 20 °C and in a 270 mM sucrose solution, has a zeta potential of about + 25 to about + 80 mV, preferably of about + 30 mV to about + 60 mV, more preferably of about + 46 mV.
88. The lipoplex of any one of claims 30 to 87, wherein the lipoplex comprises an siRNA of any one of claims 1 to 29.
89. A lipoplex comprising a siRNA of any one of claims 1 to 29 and a liposome.
90. The lipoplex according to claim 89, wherein the liposome consists of a) about 50 mol% B-arginyl-2,3-diaminopropionic acid-N-palmityl-N-oleyl- amide trihydrochloride, preferably (P-(L-arginyl)-2,3-L-diaminopropionic acid-N-palmityl-N- oleyl-amide tri-hydrochloride);
b) about 48 to 49 mol% l,2-diphytanoyl-sn-glycero-3-phosphoethanolamine (DPhyPE); and
c) about 1 to 2 mol% l,2-distearoyl-sn-glycero-3-phosphoethanolamine- polyethylen-glycole, preferably N-(Carbonyl-methoxypolyethyleneglycol-2000)- 1 ,2- distearoyl-sn-glycero-3-phosphoethanolamine sodium salt.
91. A vector comprising or encoding a nucleic acid according to any of claims 1 to 8.
92. A cell comprising a nucleic acid according to any one of claims 1 to 29 or vector according to claim 91 provided that if the cell is a human cell, said human cell is an isolated cell.
93. A composition comprising an siRNA of any one of claims 1 to 29 and a physiologically acceptable excipient.
94. A pharmaceutical composition comprising an siRNA of any one of claims 1 to 29 and a pharmaceutically acceptable excipient.
95. The pharmaceutical composition of claim 94, wherein the siRNA is part of or contained in a lipoplex according to any one of claims 88 to 90.
96. A method of treating a human disease or pathological condition comprising the administration to a subject of a therapeutically effective amount of a composition comprising an siRNA molecule according to claims 1 to 29 or of a therapeutically effective amount of a lipoplex of any one of claims 80 to 90.
97. The method of claim 96, wherein the disease or pathological condition is pre-eclamsia.
98. A method for treating a lung disease comprising the administration to a subject of a therapeutically effective amount of a composition comprising an siRNA molecule according to claims 1-29 or of a therapeutically effective amount of a lipoplex of any one of claims 88 to 90.
99. The method of claim 98, wherein the lung disease is pulmonary hypertension.
100. Use of an siRNA of any one of claims 1 to 29 for the manufacture of a medicament.
101. Use of a lipoplex of any one of claims 88 to 90 for the manufacture of a medicament.
102. Use of an siRNA of any one of claim 1 to 29 for the manufacture of a medicament for the treatment of a human disease or a pathological condition.
103. Use of a lipoplex of any one of claims 88 to 90 for the manufacture of a medicament for the treatment of a human disease or a pathological condition.
104. Use of any one of claims 100 to 103, preferably 102 to 103, wherein the disease is pulmonary hypertension.
105. Use of any one of claims 100 to 103, preferably 102 to 103, wherein the disease is preeclampsia.
106. The siRNA of any one of claims 1 to 29, for use in treatment of a subject.
107. The siRNA for use of claim 106, wherein the treatment is treatment of a disease or condition which can be alleviated by inhibition of endothelin 1 (END1).
108. The siRNA for use of any one of claims 106 to 107, wherein the disease or condition is a lung disease, preferably the lung disease if pulmonary hypertension.
109. The siRNA for use any one of claims 106 to 107, wherein the disease is pre-eclampsia.
110. The siRNA for use of any one of claims 106 to 100, wherein the subject is a human being.
111. The lipolex of any one of claims 88 to 90, for use in treatment of a subject.
112. The lipoplex for use of claim 111, wherein the treatment is treatment of a disease or condition which can be alleviated by inhibition of endothelin 1 (END1).
113. The lipoplex for use of any one of claims 111 to 1 12, wherein the disease or condition is a lung disease, preferably the lung disease if pulmonary hypertension.
114. The lipoplex for use any one of claims 111 to 112, wherein the disease is preeclampsia.
115. The lipoplex for use of any one of claims 11 1 to 114, wherein the subject is a human being.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP14004030 | 2014-11-28 | ||
| EP14004030.4 | 2014-11-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016083624A1 true WO2016083624A1 (en) | 2016-06-02 |
Family
ID=52002648
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2015/078108 Ceased WO2016083624A1 (en) | 2014-11-28 | 2015-11-30 | Means for inhibiting the expression of edn1 |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2016083624A1 (en) |
Citations (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1994002595A1 (en) | 1992-07-17 | 1994-02-03 | Ribozyme Pharmaceuticals, Inc. | Method and reagent for treatment of animal diseases |
| WO1996010391A1 (en) | 1994-09-30 | 1996-04-11 | The University Of British Columbia | Polyethylene glycol modified ceramide lipids and liposome uses thereof |
| WO1996010392A1 (en) | 1994-09-30 | 1996-04-11 | The University Of British Columbia | Bilayer stabilizing components and their use in forming programmable fusogenic liposomes |
| WO1996010390A1 (en) | 1994-09-30 | 1996-04-11 | Inex Pharmaceuticals Corp. | Novel compositions for the introduction of polyanionic materials into cells |
| WO1999011778A1 (en) * | 1997-09-02 | 1999-03-11 | University Of Sheffield | Antisense treatment of pulmonary hypertension |
| WO1999054459A2 (en) | 1998-04-20 | 1999-10-28 | Ribozyme Pharmaceuticals, Inc. | Nucleic acid molecules with novel chemical compositions capable of modulating gene expression |
| US6001311A (en) | 1997-02-05 | 1999-12-14 | Protogene Laboratories, Inc. | Apparatus for diverse chemical synthesis using two-dimensional array |
| EP0586520B1 (en) | 1991-05-21 | 2000-04-19 | Isis Pharmaceuticals, Inc. | Backbone modified oligonucleotide analogs |
| WO2000053722A2 (en) | 1999-03-10 | 2000-09-14 | Phogen Limited | Delivery of nucleic acids and proteins to cells |
| WO2000055314A2 (en) * | 1999-03-18 | 2000-09-21 | United Therapeutics Corporation | Inhibitors of endothelin-1 synthesis |
| EP0618925B1 (en) | 1991-12-24 | 2001-08-29 | Isis Pharmaceuticals, Inc. | Antisense oligonucleotides |
| US6395713B1 (en) | 1997-07-23 | 2002-05-28 | Ribozyme Pharmaceuticals, Inc. | Compositions for the delivery of negatively charged molecules |
| WO2004015107A2 (en) | 2002-08-05 | 2004-02-19 | Atugen Ag | Further novel forms of interfering rna molecules |
| WO2004061423A2 (en) * | 2003-01-06 | 2004-07-22 | Wyeth | Compositions and methods for diagnosing and treating colon cancers |
| WO2010094491A1 (en) * | 2009-02-18 | 2010-08-26 | Silence Therapeutics Ag | Means for inhibiting the expression of ang2 |
| WO2015082080A1 (en) * | 2013-12-05 | 2015-06-11 | Silence Therapeutics Gmbh | Means for lung specific delivery |
-
2015
- 2015-11-30 WO PCT/EP2015/078108 patent/WO2016083624A1/en not_active Ceased
Patent Citations (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0586520B1 (en) | 1991-05-21 | 2000-04-19 | Isis Pharmaceuticals, Inc. | Backbone modified oligonucleotide analogs |
| EP0618925B1 (en) | 1991-12-24 | 2001-08-29 | Isis Pharmaceuticals, Inc. | Antisense oligonucleotides |
| WO1994002595A1 (en) | 1992-07-17 | 1994-02-03 | Ribozyme Pharmaceuticals, Inc. | Method and reagent for treatment of animal diseases |
| WO1996010391A1 (en) | 1994-09-30 | 1996-04-11 | The University Of British Columbia | Polyethylene glycol modified ceramide lipids and liposome uses thereof |
| WO1996010392A1 (en) | 1994-09-30 | 1996-04-11 | The University Of British Columbia | Bilayer stabilizing components and their use in forming programmable fusogenic liposomes |
| WO1996010390A1 (en) | 1994-09-30 | 1996-04-11 | Inex Pharmaceuticals Corp. | Novel compositions for the introduction of polyanionic materials into cells |
| US6001311A (en) | 1997-02-05 | 1999-12-14 | Protogene Laboratories, Inc. | Apparatus for diverse chemical synthesis using two-dimensional array |
| US6395713B1 (en) | 1997-07-23 | 2002-05-28 | Ribozyme Pharmaceuticals, Inc. | Compositions for the delivery of negatively charged molecules |
| WO1999011778A1 (en) * | 1997-09-02 | 1999-03-11 | University Of Sheffield | Antisense treatment of pulmonary hypertension |
| WO1999054459A2 (en) | 1998-04-20 | 1999-10-28 | Ribozyme Pharmaceuticals, Inc. | Nucleic acid molecules with novel chemical compositions capable of modulating gene expression |
| WO2000053722A2 (en) | 1999-03-10 | 2000-09-14 | Phogen Limited | Delivery of nucleic acids and proteins to cells |
| WO2000055314A2 (en) * | 1999-03-18 | 2000-09-21 | United Therapeutics Corporation | Inhibitors of endothelin-1 synthesis |
| WO2004015107A2 (en) | 2002-08-05 | 2004-02-19 | Atugen Ag | Further novel forms of interfering rna molecules |
| WO2004061423A2 (en) * | 2003-01-06 | 2004-07-22 | Wyeth | Compositions and methods for diagnosing and treating colon cancers |
| WO2010094491A1 (en) * | 2009-02-18 | 2010-08-26 | Silence Therapeutics Ag | Means for inhibiting the expression of ang2 |
| WO2015082080A1 (en) * | 2013-12-05 | 2015-06-11 | Silence Therapeutics Gmbh | Means for lung specific delivery |
Non-Patent Citations (30)
| Title |
|---|
| AIRD, W.C.: "The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome", BLOOD, vol. 101, 2003, pages 3765 - 77 |
| AKHTAR ET AL., TRENDS CELL BIO., vol. 2, 1992, pages 139 |
| AKHTAR,: "Delivery Strategies for Antisense Oligonucleotide Therapeutics", 1995 |
| BIOCONJUGATE CHEM., vol. 10, 1999, pages 1068 - 1074 |
| BRENNAN ET AL., BIOTECHNOL BIOENG., vol. 61, 1998, pages 33 - 45 |
| CARUTHERS ET AL., METHODS IN ENZYMOLOGY, vol. 211, 1992, pages 3 - 19 |
| ELAYADI; COREY; 2001, CURR OPIN INVESTIG DRUGS, vol. 2, no. 4, pages 558 - 61 |
| FASEB J., vol. 14, 2000, pages 1041 - 1060 |
| GOOD ET AL., GENE THER, vol. 4, 1997, pages 45 - 54 |
| HAO-JIE ZHANG ET AL: "New Therapeutic Schedule for Prostatic Cancer-3 Cells with ET-1 RNAi and Endostar", ASIAN PACIFIC JOURNAL OF CANCER PREVENTION, vol. 15, no. 23, 6 January 2015 (2015-01-06), TH, pages 10079 - 10083, XP055250156, ISSN: 1513-7368, DOI: 10.7314/APJCP.2014.15.23.10079 * |
| HOFLAND; HUANG, HANDB. EXP. PHARMACOL., vol. 137, 1999, pages 165 - 192 |
| JASON GIEN ET AL: "Endothelin-1 impairs angiogenesis in vitro through Rho-kinase activation after chronic intrauterine pulmonary hypertension in fetal sheep", PEDIATRIC RESEARCH, vol. 73, no. 3, 23 January 2013 (2013-01-23), US, pages 252 - 262, XP055250161, ISSN: 0031-3998, DOI: 10.1038/pr.2012.177 * |
| LASIC ET AL., SCIENCE, vol. 267, 1995, pages 1275 - 1276 |
| LEE ET AL., ACS SYMP. SER., vol. 752, 2000, pages 184 - 192 |
| LIU ET AL., J. BIOL. CHEM., vol. 42, 1995, pages 24864 - 24780 |
| MATUTE-BELLO, G.; C.W. FREVERT; O. KAJIKAWA; S.J. SKERRETT; R.B. GOODMAN; D.R. PARK; T.R. MARTIN: "Septic shock and acute lung injury in rabbits with peritonitis: failure of the neutrophil response to localized infection", AM JRESPIR CRIT CARE MED., vol. 163, 2001, pages 234 - 43 |
| MAURER ET AL., MOL. MEMB. BIOL., vol. 16, 1999, pages 129 - 140 |
| NEVEEN SAID ET AL: "Tumor endothelin-1 enhances metastatic colonization of the lung in mouse xenograft models of bladder cancer", JOURNAL OF CLINICAL INVESTIGATION, vol. 121, no. 1, 4 January 2011 (2011-01-04), US, pages 132 - 147, XP055250188, ISSN: 0021-9738, DOI: 10.1172/JCI42912 * |
| OKU ET AL., BIOCHIM. BIOPHYS. ACTA, vol. 1238, 1995, pages 86 - 90 |
| OPITZ, B.; A. PUSCHEL; B. SCHMECK; A.C. HOCKE; S. ROSSEAU; S. HAMMERSCHMIDT; R.R. SCHUMANN; N. SUTTORP; S. HIPPENSTIEL: "Nucleotide-binding oligomerization domain proteins are innate immune receptors for internalized Streptococcus pneumoniae", JBIOL CHEM., vol. 279, 2004, pages 36426 - 32 |
| PATON, J.C.; J.K. MORONA; S. HARRER; D. HANSMAN; R. MORONA: "Immunization of mice with Salmonella typhimurium C5 aroA expressing a genetically toxoided derivative of the pneumococcal toxin pneumolysin", MICROB PATHOG., vol. 14, 1993, pages 95 - 102 |
| S.E. ORFANOS: "The endothelium in acute lung injury/acute respiratory distress syndrome", CURR OPIN CRIT CARE, vol. 14, 2008, pages 22 - 30 |
| SANTEL A ET AL: "A novel siRNA-lipoplex technology for RNA interference in the mouse vascular endothelium", GENE THERAPY, NATURE PUBLISHING GROUP, GB, vol. 13, no. 16, 20 April 2006 (2006-04-20), pages 1222 - 1234, XP002459352, ISSN: 0969-7128, DOI: 10.1038/SJ.GT.3302777 * |
| SANTEL, A.; M. ALEKU; O. KEIL; J. ENDRUSCHAT; V. ESCHE; B. DURIEUX; K. LOFFLER; M. FECHTNER; T. ROHL; G. FISCH: "RNA interference in the mouse vascular endothelium by systemic administration of siRNA-lipoplexes for cancer therapy", GENE THER., vol. 13, 2006, pages 1360 - 70 |
| SANTEL, A.; M. ALEKU; O. KEIL; J. ENDRUSCHAT; V. ESCHE; G. FISCH; S. DAMES; K. LOFFLER; M. FECHTNER; W. ARNOLD: "A novel siRNA-lipoplex technology for RNA interference in the mouse vascular endothelium", GENE THER., vol. 13, 2006, pages 1222 - 34 |
| SCHMECK, B.; J. ZAHLTEN; K. MOOG; V. VAN LAAK; S. HUBER; A.C. HOCKE; B. OPITZ; E. HOFFMANN; M. KRACHT; J. ZERRAHN: "Streptococcus pneumoniae-induced p38 MAPK-dependent phosphorylation of RelA at the interleukin-8 promotor", JBIOL CHEM., vol. 279, 2004, pages 53241 - 7 |
| V FEHRING ET AL: "Delivery of Therapeutic siRNA to the Lung Endothelium via Novel Lipoplex Formulation DACC", MOLECULAR THERAPY, vol. 22, no. 4, 6 January 2014 (2014-01-06), GB, pages 811 - 820, XP055250235, ISSN: 1525-0016, DOI: 10.1038/mt.2013.291 * |
| WARE, L.B.: "Pathophysiology of acute lung injury and the acute respiratory distress syndrome", SEMIN RESPIR CRIT CARE MED., vol. 27, 2006, pages 337 - 49 |
| WINCOTT ET AL., METHODS MOL. BIO., vol. 74, 1997, pages 59 |
| WINCOTT ET AL., NUCLEIC ACIDS RES., vol. 23, 1995, pages 2677 - 2684 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9458461B2 (en) | Means for inhibiting the expression of ANG2 | |
| CA2917320C (en) | Respiratory disease-related gene specific sirna, double-helical oligo rna structure containing sirna, composition containing same for preventing or treating respiratory disease | |
| EP3228326A1 (en) | Nucleic acid linked to a trivalent glycoconjugate | |
| WO2023041079A1 (en) | Lpa inhibitor and use thereof | |
| WO2016083623A1 (en) | Means for the treatment of pre-eclampsia | |
| US20220389430A1 (en) | Chemical modifications of small interfering rna with minimal fluorine content | |
| US20250207137A1 (en) | Amphiregulin gene-specific double-stranded oligonucleotide and composition for preventing and treating fibrosis-related diseases and respiratory diseases, comprising same | |
| CN114901821A (en) | Use of SEPT9 inhibitors for treating hepatitis B virus infection | |
| WO2017135397A1 (en) | Antisense oligonucleotide for suppressing expression of complement b factor | |
| KR102701681B1 (en) | Double Strand Oligonucleotide and Composition for Treating COVID-19 Infection Containing Thereof | |
| WO2010091878A2 (en) | Means for inhibiting the expression of opa1 | |
| JP2025094072A (en) | Use of COPS3 inhibitors to treat hepatitis B virus infection | |
| JP2023538630A (en) | Use of A1CF inhibitors to treat hepatitis B virus infection | |
| KR101286053B1 (en) | The shRNA downregulating TGF-β1 for treatment of tumor | |
| WO2020139866A2 (en) | Compositions and methods for treating cancer | |
| WO2023134705A1 (en) | Rna interference agent for inhibiting angptl3 expression, and use thereof | |
| WO2016083624A1 (en) | Means for inhibiting the expression of edn1 | |
| WO2013123996A1 (en) | Novel sirna inhibitors of human icam-1 | |
| CN116478997B (en) | siRNA for inhibiting FGL1 and its modifications and applications | |
| US20250354156A1 (en) | Compositions and Methods for Treating Cancer | |
| EP4446414A1 (en) | Antisense oligonucleotide complex | |
| WO2025256564A1 (en) | iRNA COMPOSITION AND METHOD OF USING SAME | |
| CN117904110A (en) | SiRNA for inhibiting DPP4, and modification and application thereof | |
| KR20240116658A (en) | Antisense Oligomers Targeting GFRAL and Use Thereof | |
| WO2025252184A1 (en) | iRNA COMPOSITION AND METHOD OF USING SAME |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15802098 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 15802098 Country of ref document: EP Kind code of ref document: A1 |