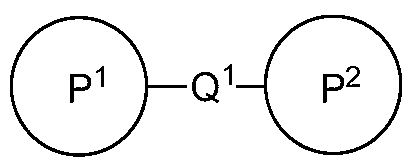WO2015106294A1 - Bivalent bcr-abl tyrosine kinase ligands, and methods of using same - Google Patents
Bivalent bcr-abl tyrosine kinase ligands, and methods of using same Download PDFInfo
- Publication number
- WO2015106294A1 WO2015106294A1 PCT/US2015/011273 US2015011273W WO2015106294A1 WO 2015106294 A1 WO2015106294 A1 WO 2015106294A1 US 2015011273 W US2015011273 W US 2015011273W WO 2015106294 A1 WO2015106294 A1 WO 2015106294A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- alkyl
- carbon atoms
- substituted
- amino
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- LBNKFOTZHYYHIY-UHFFFAOYSA-N CC(C)(C)OC(N1CCN(CCOCc2c[n](CCOCCOCCOCCOCCOCCOCCOCCO)nn2)CC1)=O Chemical compound CC(C)(C)OC(N1CCN(CCOCc2c[n](CCOCCOCCOCCOCCOCCOCCOCCO)nn2)CC1)=O LBNKFOTZHYYHIY-UHFFFAOYSA-N 0.000 description 1
- WVXNSAVVKYZVOE-UHFFFAOYSA-N CC(C)(C)c(cc1NC(Nc(ccc(Oc2cc(C(NC)=O)ncc2)c2)c2F)=O)n[n]1-c1cc2cccnc2cc1 Chemical compound CC(C)(C)c(cc1NC(Nc(ccc(Oc2cc(C(NC)=O)ncc2)c2)c2F)=O)n[n]1-c1cc2cccnc2cc1 WVXNSAVVKYZVOE-UHFFFAOYSA-N 0.000 description 1
- QXRYVDVCRNBTKW-LNKXUWQBSA-N CC(C)Cc1ccc([N](C)(C)O[N](C)(C)C2[C@@H](CC3)C[C@@H]3C2)[o]1 Chemical compound CC(C)Cc1ccc([N](C)(C)O[N](C)(C)C2[C@@H](CC3)C[C@@H]3C2)[o]1 QXRYVDVCRNBTKW-LNKXUWQBSA-N 0.000 description 1
- ICUCTLZTAZTJNL-JNIOUFIGSA-N CCCc([nH]1)ccc1[N](C)(C)O[N](C)(C)C1[C@@H](C2)CCC[C@H]2C1 Chemical compound CCCc([nH]1)ccc1[N](C)(C)O[N](C)(C)C1[C@@H](C2)CCC[C@H]2C1 ICUCTLZTAZTJNL-JNIOUFIGSA-N 0.000 description 1
- BXBGPRNISBNEGW-UHFFFAOYSA-N Cc(cccc1Cl)c1NC(c1cnc(Nc2nc(C)nc(N3CCN(CCOCc4c[n](CCOCCOCCOCCOCCOCCOCCOCCOCCOCCN)nn4)CC3)c2)[s]1)=O Chemical compound Cc(cccc1Cl)c1NC(c1cnc(Nc2nc(C)nc(N3CCN(CCOCc4c[n](CCOCCOCCOCCOCCOCCOCCOCCOCCOCCN)nn4)CC3)c2)[s]1)=O BXBGPRNISBNEGW-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0834—Compounds having one or more O-Si linkage
- C07F7/0838—Compounds with one or more Si-O-Si sequences
Definitions
- Bcr gene at human chromosome 22
- Abelson (Abl) tyrosine kinase gene at human chromosome 9 has been implicated as a cause of chronic myelogenous leukemia.
- Several inhibitors of Bcr-Abl tyrosine kinase have been developed for the treatment of chronic myelogenous leukemia, for example imatinib, nilotinib, dasatinib, bosutinib, ponatinib, and bafetinib.
- the kinase domain of Bcr-Abl tyrosine kinase contains at least two binding pockets, i.e., a myristoyl binding pocket and an ATP binding pocket, and inhibitors of Bcr-Abl tyrosine kinase generally bind to the ATP binding pocket.
- Bcr-Abl tyrosine kinase inhibitors in treating cancer (e.g., chronic myelogenous leukemia) has been limited by the emergence of resistant Bcr-Abl tyrosine kinase mutants.
- a protein e.g., Bcr-Abl tyrosine kinase
- Q 1 is a connecting moiety covalently bound to P 1 and P 2 , wherein Q 1 is selected from the group consisting of -Si(R 7 )(R 8 )-, -Si(R 7 )(R 8 )-0-, -0-Si(R 7 )(R 8 )-, -NR'-, -N(R' )C(0)-, ⁇ C(0)N(R' )-, -N( R " )SO >-. -SO N; R " ;-.
- a method of treating a disease associated with Bcr-Abl tyrosine kinase in a patient in need thereof comprises administering to the patient the bivalent compound as described above.
- FIG. 1 shows a screenshot of a protein X-ray crystal structure in which the structures of imatinib and dasatinib are overlaid, according to an embodiment.
- FIG. 2 shows a non-limiting set of pharmacophores (i.e., ligands) with preferred attachment points for connecting the pharmacophores to connecting moieties indicated by arrows, according to an embodiment.
- pharmacophores i.e., ligands
- Described herein are compounds capable of modulating one or more biomolecules and, in some cases, modulating two or more binding sites on a protein (e.g., Bcr- Abl tyrosine kinase).
- a protein e.g., Bcr- Abl tyrosine kinase
- the bivalent compound may be capable of interacting with a relatively large target site as compared to the individual ligands that form the bivalent compound.
- a target may comprise, in some embodiments, two protein binding sites separated by a distance such that a bivalent compound, but not an individual ligand moiety, may be capable of binding to both binding sites essentially simultaneously.
- contemplated bivlalent compounds may bind to a target with greater affinity as compared to an individual ligand moiety binding affinity alone.
- a bivalent compound that, e.g., may be capable of modulating proximal binding sites on the same target.
- the proximal binding sites may be the myristoyl binding pocket and ATP binding pocket of Bcr-Abl tyrosine kinase.
- bivalent compounds may bind to a first Bcr-Abl tyrosine kinase binding site and a second Bcr-Abl tyrosine kinase binding site.
- the bivalent compound may be used for a variety of purposes.
- the bivalent compound may be used to perturb a biological system.
- the bivalent compound may bind to or modulate a target biomolecule, such as a protein (e.g., Bcr-Abl tyrosine kinase).
- a contemplated bivalent compound may be used as a pharmaceutical.
- a bivalent compound may bind to a target biomolecule with a dissociation constant of less than 1 mM, in some embodiments less than 500 microM, in some embodiments less than 300 microM, in some embodiments less than 100 microM, in some embodiments less than 10 microM, in some embodiments less than 1 microM, in some embodiments less than 100 nM, in some embodiments less than 10 nM, and in some embodiments less than 1 nM, in some embodiments less than 1 pM, in some embodiments less than 1 fM, in some embodiments less than 1 aM, and in some embodiments less than 1 zM.
- bivalent compounds of formula I are provided:
- P 1 is a first ligand capable of modulating a first Bcr-Abl tyrosine kinase binding site
- P 2 is a second ligand capable of modulating a second Bcr-Abl tyrosine kinase binding site
- Q 1 is a connecting moiety covalently bound to P 1 and P 2 that comprises between 3 and 30 atoms, where the atoms are connected to form a cyclic or acyclic, substituted or
- unsubstituted, branched or unbranched aliphatic moiety cyclic or acyclic, substituted or unsubstituted, branched or unbranched heteroaliphatic moiety; substituted or unsubstituted phenyl or naphthyl moiety; substituted or unsubstituted heteroaryl moiety; or a combination thereof.
- the ligand may be a pharmacophore.
- a pharmacophore is typically an arrangement of the substituents of a moiety that confers biochemical or pharmacological effects (e.g., by targeting a Bcr-Abl tyrosine kinase).
- identification of a pharmacophore may be facilitated by knowing the structure of the ligand in association with a target biomolecule.
- pharmacophores may be moieties derived from molecules previously known to bind to target biomolecules (e.g., proteins), fragments identified, for example, through NMR or crystallographic screening efforts, molecules that have been discovered to bind to target proteins after performing high- throughput screening of natural products libraries, previously synthesized commercial or non-commercial combinatorial compound libraries, or molecules that are discovered to bind to target proteins by screening of newly synthesized combinatorial libraries. Since most pre-existing combinatorial libraries are limited in the structural space and diversity that they encompass, newly synthesized combinatorial libraries may include molecules that are based on a variety of scaffolds.
- a bivalent compound may include a first pharmacophore that may bind to the myristoyl binding pocket of Bcr-Abl tyrosine kinase and a second pharmacophore that may bind to the ATP binding pocket of Bcr-Abl tyrosine kinase.
- a ligand e.g., a pharmacophore
- an attachment point on a pharmacophore may be chosen so as to preserve at least some ability of the pharmacophore to bind to a binding site.
- preferred attachment points may be identified using X-ray crystallography. The following description of a non-limiting exemplary method illustrates how a preferred attachment point may be identified. For example, as shown in FIG.
- STI2 small molecule 1 10 (dark gray) labeled "STI2" in the PDB file [also known as Imatinib or Gleevec or STI-571] may be identified.
- the Imatinib pyridyl ring (indicated by black circle 120) contains a nitrogen atom in the 3 position, which constitutes an adenine ring mimetic.
- the corresponding adenine ring mimetic in the new pharmacophore 140 should be aligned to this element.
- the final conformation and orientation of the newly aligned pharmacophore 140 in the site may be determined using a variety of approaches known to computational chemists, but can be done as simply as performing an energy minimization using a molecular mechanics forcefield.
- the alphanumeric identifiers in FIG. 1 correspond to amino acid residues in the 3K5V structure, where the letter of the identifier is the one-letter amino acid symbol and the number of the identifier is the position of the amino acid residue in the primary sequence of the protein.
- Attachment points such as the one labelled 150 on the aligned pharmacophore permit access to solvent exposed amino acid residues on the outer lip of the
- ATP-binding pocket such as T338 and L389 in the 3K5V structure and are considered preferred attachment points for linkers. It should be apparent to those skilled in the art that overlays of the imatinib pharmacophore with other alternate pharmacophores can be used to identify potential attachment points.
- FIG. 2 provides a non-limiting set of pharmacophores (i.e., ligands) showing preferred attachment points (indicated by arrows) for connecting the pharmacophore to a connecting moiety.
- ligands i.e., ligands
- preferred attachment points indicated by arrows
- the ligands disclosed herein can be attached at any open site to a connector moiety as described herein. Such embodiments described below include specific references to each attachment site.
- Exemplary binding site ligands include Bcr-Abl tyrosine kinase ATP binding site ligands represented by the formula: w herein:
- R is:
- R 6 is alkyl, alkenyl, aikynyl, cycloalkyl, cycloalkylalkyl, cycloalkenyl, cycloalkenyl alkyl, aryi, aralkyl, heterocyclo, or heterocyeioalkyl, each of which is unsubstituted or substituted with U 1 , L ' and one or more groups U J ;
- R 2 is:
- R 3 is:
- (1) are each independently hydrogen or R 6 ;
- R ' and R 8 may together be alkylene, alkenylene or heteroalkyl, completing a 3- to 8-membered saturated or unsaturated ring with the nitrogen atom to which they are attached, which ring is unsubsiituted or substituted with U ⁇ U 2 and U 3 ; or
- any two of R 9 , R 10 , and R n may together be alkylene or alkenylene completing a 3- to 8-membered saturated or unsaturated ring together with the nitrogen atoms to which they are attached, which ring is unsubstituted or substituted with U 1 , U z and U J ;
- R i3 is:
- R 14 is:
- R i 5 is:
- ⁇ ', U 2 and U 3 are each independently:
- LI 6 is (i) alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, cycloalkenyl, eycloalkenylaikyl, aryl, aralkyi, alkylaryl, cycloalkylaryl, heterocyclo, or heterocycloalkyl; (ii) a group (i) which is itself substituted by one or more of the same or different groups (i); or (iii) a group (i) or (ii) which is substituted by one or more of the following groups (2) to (16) of the definition of U 1 , U 2 and U 3 ;
- any two of U 1 , U z , and U 3 may together be alkylene or alkenylene completing a 3- to 8-membered saturated or unsaturated ring together with the atoms to which they are attached; or
- any two of U 1 , U", and U 3 may together be— O— (CH2 V----G— , where r is 1 to 5, completing a 4- to 8-membered saturated or unsaturated ring together with the atoms to which they are attached;
- (1) are each independently hydrogen or U 6 ;
- ⁇ ' and U 8 , or U°and U 10 may together be alkylene or alkenylene, completing a 3- to 8-membered saturated or unsaturated ring together with the atoms to which they are attached, which ring is un substituted or substituted with U 1 , U a d LP; or
- U ? or U 8 together with U 9 , may be alkylene or alkenylene completing a. 3- to 8-membered saturated or unsaturated ring together with the nitrogen atoms to which they are attached, which ring is unsubstituted or substituted with U 1 , U 2 and U ;
- U 11 and U 12 are each independently:
- q 1 or 2;
- a Bcr-Abl tyrosine kinase ligand may be represented by:
- a Bcr-Abl tyrosine kinase ATP binding site ligand may be represented by imatinib, nilotinib, bosutinib, ponatinib, DCC-2036, GNF5, or bafetinib.
- Exemplary Bcr-Abl tyrosine kinase myristoyl binding site ligands include those represented by the formula:
- R ! is selected from the group consisting of -U ' R 6 R', -UOR' and -U 3 R' , wherein U 3 is a bond or Ci _ 4 alkylene, R 6 is hydrogen or C h alky 1 and R 7 is selected from the group consisting of Ce-ioaryl and Cs-eheteroaryl; wherein any aiyl or heteroaryl is optionally substituted with i to 3 radicals independently selected from the group consisting of halo, amin halo - substituted Cj_ 4 alkyl,
- R is selected f rom the group consisting ofhydrogen, halo, amino, alo- substituted Ci.. 4 aikyl, Ci. 4 aikoxy and halo-substituted Cj . 4 alkoxy;
- R 3 is selected from the group consisting of Cs-gheterocycloalkyl-Co ⁇ alkyl, CV ioheteroaryl-Co -4 alkyl and Ce-i oary l-Co ⁇ alkyl; wherein any alkyl group is optionally substituted with 1 to 3 radicals selected from the group consisting of hydroxy, halo and amino; and any aryi, heteroaryl or heterocycloalkyl is optionally substituted with 1 to 3 radicals independently selected from the group consisting of halo, nitro, C h alky 1, halo-substituted hydroxy - Chalky], halo-substituted phenyl, Cs-sheterocycloalkyl, -
- R 9 is hydroxy, C6-ioaryl--Co- 4 aikyi, C6-ioaryl-Co- 4 aikyloxy, CYioheteroaryl-Co- 4 aikyl, Cs-sheterocycloalkyl-Co ⁇ alkyl or Cs-scycloalkyl; wherein said aryl, heteroaryl, cycloalkyl, heterocycloalkyl or alkyl of R 9 is further optionally substituted by up to 2 radicals selected from the group consisting of halo, hydroxy, cyano, amino, nitro, C i. 4 alkyl, hydroxy-Ct.
- R i0 is Cs-eheteroaryl and R u is hydroxy-Ci -4 alkyl; wherein R 3 is substituted with Q 1 ; and the N-oxide derivatives, prodrug derivatives, protected derivatives, individual isomers and mixture of isomers thereof; and the pharmaceutically acceptable salts and solvates of such compounds.
- a Bcr-Abl tyrosine kinase ligand may be represented by:
- Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
- R 1 is pyrazolyl; wherein said pyrazolyl is unsubstituted or substituted with 1 to 2 R° groups;
- R;2 is pyrroiidinyl: wherein said pyrrolidinyl is substituted with one R' group;
- R J is selected from hydrogen and halo
- R 4 is selected from » SF 5 and -Y 2 -CF 2 -Y 3 ;
- R 6 at each occurrence is independently selected from the group consisting of hydrogen, hydroxy, methyl, methoxy, cyano, trifiuoromethyl, hydroxy-methyl, halo, amino, fluoro-ethyl, ethyl and cyclopropyl;
- R' is selected from hydroxy, methyl, halo, methoxy, hydroxy-methyl, amino, methyl- amino, amino-methyl, trifiuoromethyl, 2-hydroxypropan-2-yl, methyl-carbonyl-amino, dimethyl-amino, 2-amino-3-methylbiitanoyl)oxy, carboxy, methoxy-carbonyl, phosphonooxy, cyano, and amino-carbonyi;
- Y is selected from CH and N;
- Y 1 is selected from CH and N;
- Y 2 is selected from CF 2 , O and S(0)o -2 ;
- Y ' is selected from hydrogen, chioro, fiuoro, methyl, difluoromethyl, and
- -Abl tyrosine kinase ligand may be represented by:
- Exemplary Bcr-Abl tyrosine kinase myristoyl binding site ligands include those
- Y at each occurrence is independently selected from N and CH;
- Y 1 is selected from and CR 5 ; wherein R "1 is selected from hydrogen, methoxy and imidazolyl; wherein said imidazolyl is unsubstituted or substituted with methyl;
- R 1 is selected from pyrazoiyl, thiazoiyl, pyrrolyl, imidazolyl, isoxazolyl, furanyl and thienyl; wherein said thiazoiyl, pyrrolyl, imidazolyl, isoxazolyl, furanyl, or thienyl of R 1 is unsubstituted or substituted with 1 to 3 R 6 groups;
- R 1 is selected from pyrrolidinyl, piperidinyl, azetidinyl, morpholino, piperazinyl, 2-oxa- 6-azaspiro[3.4j-ocianyl, 3-azabicyc3o[3.1.0]hexan-3-yl, pyrrolo[3,4-c]pyrazo3-5(lH,4H,6H)-yL hexahydropyrrolo[3,4-c]pyrrolyl, 6-oxo-2,7-diazaspiro[4.4]-nonanyl, lH-pyrrolo[3,4- cjpyridinyl, 1 ,4-oxazepan-4-yl, 2-oxooxazolidinyl, j ,4-diazepanyl, tetrahydro-2H-pyranyl, 3,6- dihydro-2H-pyranyl, 3,8-dioxa-10-azabicyclo[4.3.1]decan
- R 3 is selected from hydrogen and halo;
- R 4 is selected from -SF 5 and -Y 2 -CF 2 -Y 3 ;
- R 33 is selected from hydrogen and C h alky 1;
- R 3D is selected from and tetrahydro-2H-pyran-4-yI; wherein said aikyl of R 5 ° is unsubstituted or substituted with 1 to 3 groups independently selected from hydroxy and dimethyl-amino;
- R° at each occurrence is independently selected from hydrogen, hydroxy, methyl, hydroxy, methoxy, cyano, trifluoromethyl, hydroxy-methyl, halo, amino, fluoro-ethyl, ethyl, cyclopropyl and dimethyl-amino-carbonyl;
- R ' at each occurrence is independently selected from hydroxy, methyl, halo, methoxy, hydroxy-methyl, amino, methyl-amino, amino -methyl, trifluoromethyl, 2-hydroxypropan-2-yl, meihy l-carbonyl-amino, dimethyl-amino, 2-amino-3 -methylbutanoy l)oxy, carboxy, methoxy- carbonyl, phosphonooxy, cyano, and amino-carbonyl; or two R' groups combine with the atom to which they are attached to form a ring selected from cyclopropyl, azetidin-3-yl and 3- azabicycio[3.1.Ojhexan-3-yl;
- Y 2 is selected from CF 2 , O and S(0)o- 2 ;
- Y 3 is selected from hydrogen, halo, methyl, difluoromethyl and trifluoromethyl; or pharmaceutically acceptable salts thereof
- Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
- X is cycloaikyl of 3 to 7 carbon atoms, which may be optionally substituted with one or more aikyl of 1 to 6 carbon atom groups; or is a pyridinyl, pyrimidinyl, or phenyl ring; wherein the pyridinyl, pyrimidinyl, or phenyl ring optionally mono- di-, or tri-substituted with a substituent selected from the group consisting of halogen, aikyl of 1 -6 carbon atoms, alkenyl of 2-6 carbon atoms, alkynyl of 2-6 carbon atoms, azido, hydroxyalkyl of 1-6 carbon atoms, halomethyl, aikoxymethyl of 2-7 carbon atoms, alkanoyloxymethyl of 2-7 carbon atoms, alkoxy of 1-6 carbon atoms, alky lth io of 1 -6 carbon atoms, hydroxy, trifluoromethyl, cyan
- Y is -NH-, -0-, -S-, or - R-;
- R is Ci-ealkyl
- R ⁇ R , R ⁇ and R 4 are each, independently, hydrogen, halogen, alkyl of 1-6 carbon atoms, alkenyl of 2-6 carbon atoms, alkynyl of 2-6 carbon atoms, alkenyloxy of 2-6 carbon atoms, alkynyloxy of 2-6 carbon atoms, hydroxymethy], alomethyl, alkanoyloxy of 1-6 carbon atoms, alkenoyloxy of 3-8 carbon atoms, alkynovloxy of 3-8 carbon atoms, alkanoyloxymethyl of 2-7 carbon atoms, alkenoyloxymethyl of 4-9 carbon atoms, alkynoyloxymethyl of 4-9 carbon atoms, alkoxymethyl of 2-7 carbon atoms, alkoxy of 1-6 carbon atoms, alkoxy of 1-6 carbon atoms, alkylthio of 1-6 carbon atoms, alkylsulphinyl of 1 -6 carbon atoms, alkylsulphonyl of
- R 5 is alkyl of 1-6 carbon atoms, alkyl optionally substituted with one or more halogen atoms, phenyl, or phenyl optionally substituted with one or more halogen, alkoxy of 1-6 carbon atoms, trifluoromethy], amino, nitro, cyano, or alkyl of 1 -6 carbon atoms groups;
- R 6 is hydrogen, alkyl of 1-6 carbon atoms, or alkenyl of 2-6 carbon atoms
- R ! is chloro or bromo
- R* is hydrogen, alkyl of 1-6 carbon atoms, aminoaikyl of 1-6 carbon atoms, N- alky!ammoalkyl of 2-9 carbon atoms, ⁇ , ⁇ -dialkylaminoalkyl of 3-12 carbon atoms, - cycloalkylaminoalkyl of 4-12 carbon atoms, N-cycloaikyl-N-alkylaminoalkyl of 5- 18 carbon atoms, ⁇ , ⁇ -dicycloalkylaminoalkyl of 7-18 carbon atoms, morpholino-N-alkyl wherein the alkyl group is 1-6 carbon atoms, piperidino-N-aikyl wherein the alkyl group is 1-6 carbon atoms, N-alkyl-piperidino-N-alkyl wherein either alkyl group is 1 -6 carbon atoms, azacycloalkyl- -alkyl of 3-11 carbon atoms, hydroxyalkyl of 1-6 carbon
- n is an integer from 1-4, q is an integer from 1 -3, and p is an integer from 0-3;
- any two of the substituents R ! , R 2 , R:', or R 4 that are located on contiguous carbon atoms can together be the divalent radical -0-C(R 8 ) 2 -0-;
- -Abl tyrosine kinase ligand may be represented by:
- Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
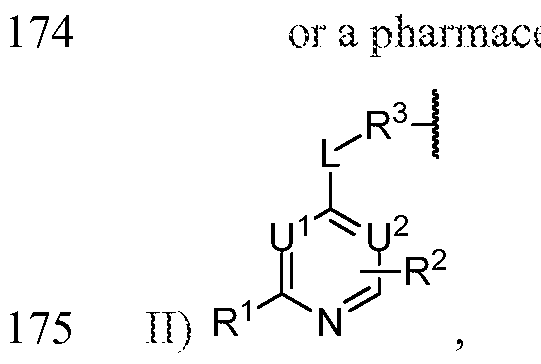
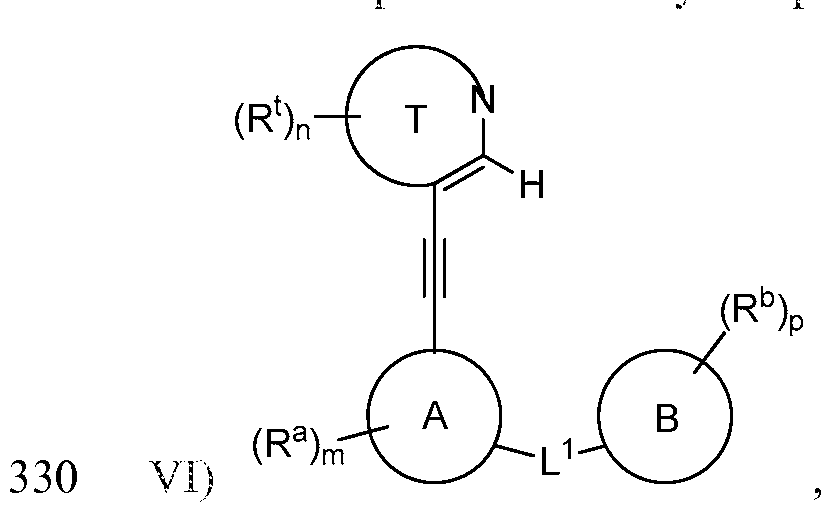
- Ring T is a 5-membered heteroaryl ring containing 1-2 nitrogens with the remaining ring atoms being carbon, substituted on at least two ring atoms with R l groups, at least two of which being located on adjacent ring atoms, and, together with the atoms to which they are attached, forming a saturated, partially saturated or unsaturated 5- or 6- membered ring (Ring E), containing 0-3 heteroatoms selected from O, N, and S and being optionally substituted with 1-4 R e groups;
- Ring A is a 5- or 6-membered aryl or heteroaryl ring
- Ring B is a 5- or 6-membered aryl or heteroaryl ring;
- L 1 is selected from N ⁇ QO), C ⁇ NR 1 , ⁇ (0)0, NR 1 C(0)NR 1 , and OC(0)NR 1 ;
- R 1 , R 2 , and R 3 are independently selected from H, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl;
- R 2 and R 3 taken together with the atom to which they are attached, form a 5- or 6- membered saturated, partially saturated or unsaturated ring, which can be optionally substituted and which contains 0-2 heteroatoms selected from N, O and S(0) r -;
- each occurrence of R 4 is independently selected from alkyl, haloalkyl, trifluoromethyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl; each of the alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl moieties is optionally substituted;
- n 0, 1 , 2, 3 or 4;
- n 2 or 3;
- p 0, 1, 2, 3, 4 or 5;
- r 0, 1 or 2;
- a Bcr-Abl tyrosine kinase ligand may be represented by: or a pharmaceutically acceptable salt thereof.
- Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
- R 1 represents hydrogen, lower alkyl, lower alkoxy-lower alkyl, or benzyl
- R z represents phenyl substituted by one or two substituents selected from the group consisting of C h alky!, trifluoro-C t -salkyL hydroxy-Ci-ealkyl, amino-Ct-ealkyl, Ci-ealkyl- ammo-Ci-ealkyl, N-cyclo exyl-N-Ci-ealkylamino-Ct-galkyl, Ci- 6 alkoxy-C(0)-piperidino-Ci- 6 alkyl, N-Ci-ealkyl-piperazino-Ci-ealkyl,
- Ci-ealkoxy hydroxy, Ci-ealkoxy, trifluoro-CVeaikoxy, IH-imidazolyi-Ci-ealkoxy, Ci- 6 alkyl-C(0)-, benzoyloxy, carboxy, carbamoyl, C h lky 1-carbamoyi, amino, Ci_ 6 alkyl-C(0)-amino, benzoylarnino, amino mono- or disubstituted by Ci-ealkyl, by hydroxy-Ci-ealkyl or by Q.
- R 3 represents hydrogen, d-ealkyl, or halo
- a Bcr-Abl tyrosine kinase ligand may be represented by: , or a pharmaceutically acceptable salt thereof.
- Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
- R 1 is 4-pyrazinyl, 1 -methyl- lH-pyrrolyl, amino- or amino-Cj.ealkyl-substituted phenyl wherein the amino group in each case is free, alkylated or acylated, lH-indolyl or 1H- imidazolyl bonded at a five-membered ring carbon atom, or unsubstituted or C...galkyl- substituted pyridyl bonded at a ring carbon atom and unsubstituted or substituted at the nitrogen atom by oxygen,
- R 2 and R 3 are each independently of the other hydrogen or C h alk ], one or two of the radicals R 4 , R 5 , R 6 , R 7 and R 8 are each nitro, fluoro-substituted Cj -ealkoxy or a radical represented by:
- X is oxo, thio, imino, N-Cj-6alkyl-imino, hydroximino or O-Cj-ealkyl-hydroximino
- Y is oxygen or the group NH
- n 0 or 1
- R 10 is phenyl that is unsubstituted or substituted by halogen, cyano, Ci- 6 alkoxy, carboxy, Ci- 6 alkyl, or by 4-methyl-piperazinylmethyl; C5_7alkyl; thienyl; 2-naphthyl;
- R 4 , R 5 , R 6 , R 7 and R 8 are each independently hydrogen, Cj- 6 alkyi that is unsubstituted or substituted by free or alkylated amino, piperazinyl, piperidinyl, pyrrolidinyl or by morpholinyl, or Q.ealkyl-QO)-, trifluoromethyl, free, etherified or esterifed hydroxy, free, alkylated or acylated amino or free or esterified carboxy;
- -Abl tyrosine kinase ligand may be represented by: , or a pharmaceutically acceptable salt thereof.
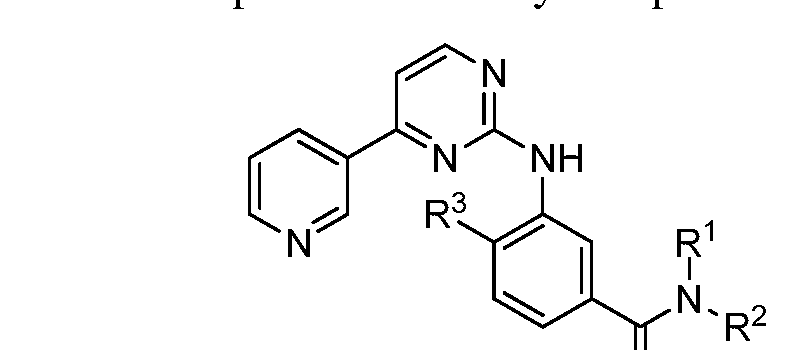
- Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
- E 1 is phenyl substituted with 1 , 2, or 3 groups independently selected from fluoro and methyl;
- A is selected from the group consisting of imidazolyl and pyrazolvl, wherein A is substituted at any suitable position with a 10-membered bicyclic heteroaryl ring optionally substituted with 1 , 2, or 3 R 1 groups;
- R 1 is independently selected from the group consisting of d-ealkyl, branched Cj. alkyf, Cs-scarbocyclyL halogen, fluoroCi-ealkyl wherein the alky! moiety can be partially or fully fluorinated, cyano, hydroxy 1, methoxy, oxo, (R 2 ) ? C(0)-, - (R 2 )C(0)R ⁇ i R ' bXSO *-. - (R 2 )S0 2 R 2 , -(CH 2 )qN(R 2 ) 2 , -0(CH 2 ) q N(R 2 ) 2 , -Oi CI l .
- R 2 is H, Cj-ealkyl, branched Cs-yalkyl, Cj-gcarbocyclyl, or phenyl;
- t is 1 , 2, or 3;
- a Bcr-Abl tyrosine kinase ligand may be represented by:
- Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the
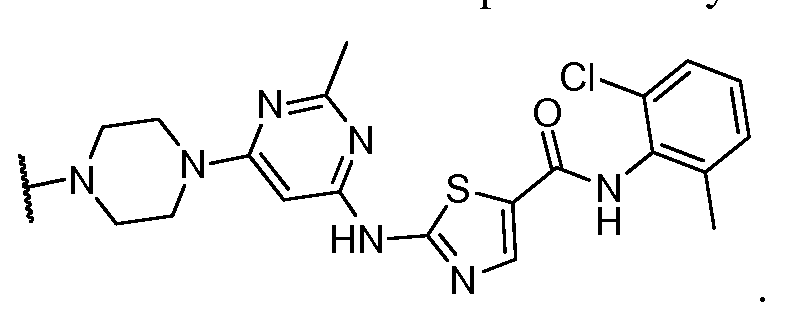
- Ring A is selected from the group consisting of phenylene, pyridinylene, pyrazinylene, pyrimidinylene, pyridazinylene, and triazinylene;
- Ring B is selected from the group consisting of pyridyi, pyrimidinyl, pyrazinyl, pyridazinyl, and 1 ,2-dihy dropyridaziny 1, each optionally substituted with 1 , 2, or 3 groups independently selected from C h alky!, halo, and amino;
- R ! is wherein R J is a saturated, nitrogen-containing heterocyclic group substituted by oxo, a saturated, nitrogen-containing heterocyclic group, aminoalkyl, monoalkylaminoalkyl, dialkylaminoaikyl, Ci-ealkoxy, d-ealkyl, alkyoxycarbonyl, halo, haloalkyl, hydroxyalkyl, amino, monoalkylamino, dialkylamino, carbamoyl,
- R is selected from Cuealkyl, halogen, haloalkyl, trifiuoromethyl, hydroxyalkyl, d_ ealkoxy, alkoxyalkyl, alkoxycarbonyl, acyl, amino, monoalkylamino, dialkylamino, nitro, carbamoyl, monoalkylcarbamoyl, dialkyicarbamoyl, and cyano;
- R 3 is selected from hydrogen, C 3 - ⁇ alkoxy, and halo
- R 4 is selected from hydrogen, Q-ealkyl, and halo
- n 1, 2, 3, or 4;
- a Bcr-Abl tyrosine kinase ligand may be represented by: , or a pharmaceutically acceptable salt thereof.
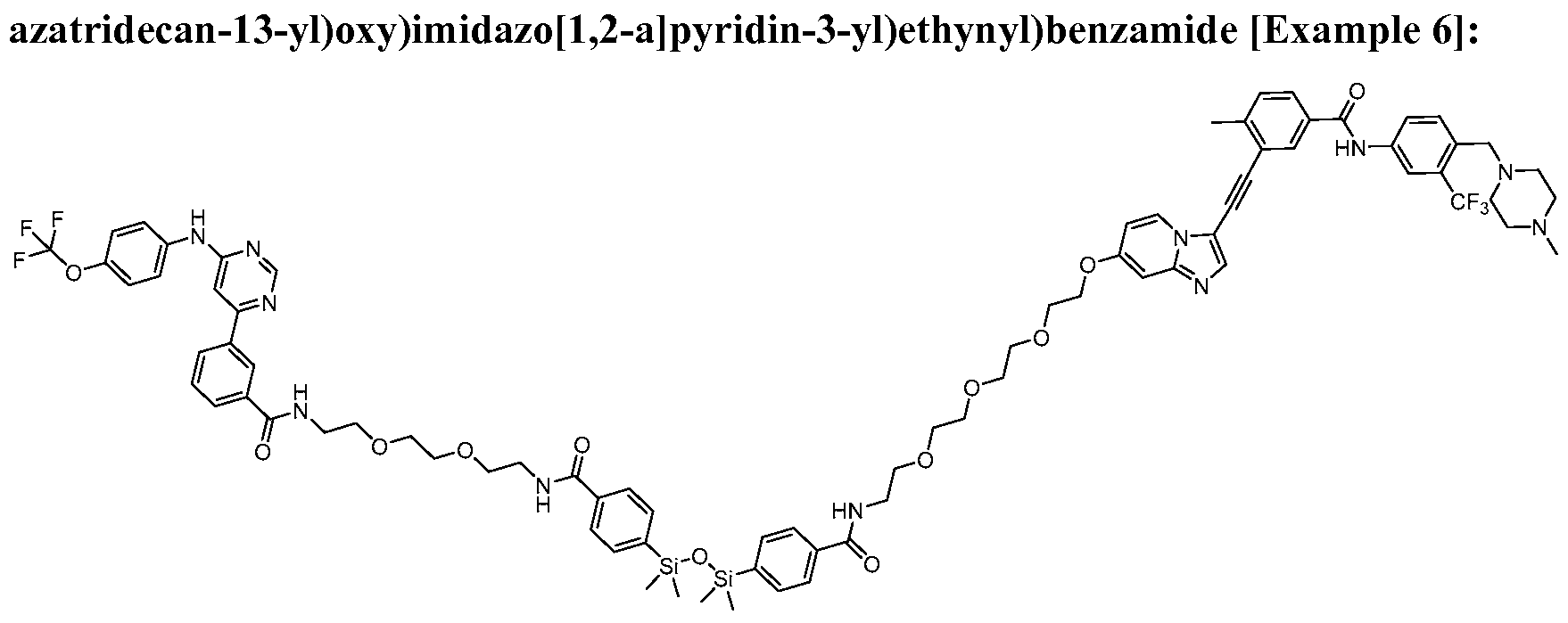
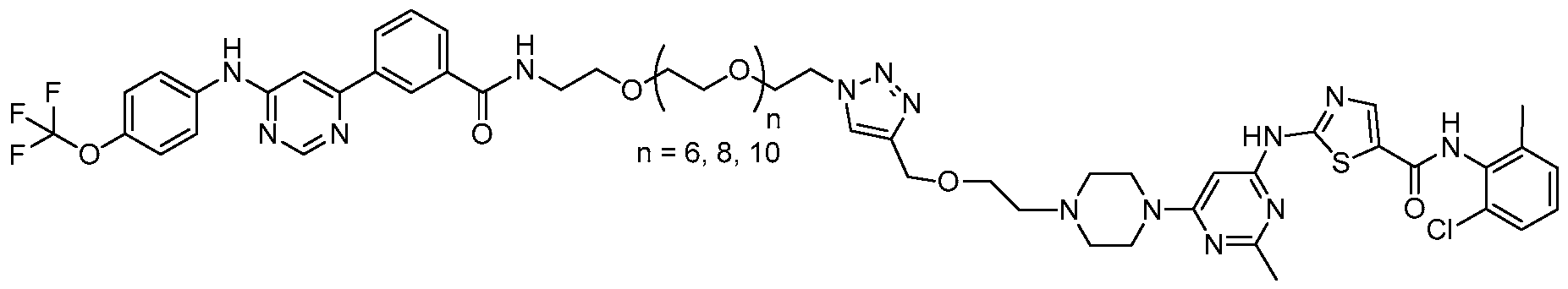
- a contemplated bivalent compound may be selected from the group consisting of:
- certain compounds contemplated herein comprise a first ligand and a second ligand covalently joined by a connector moiety.
- such connector moieties do not have significant binding or other affinity towards an intended target.
- a connector may contribute to the affinity of a ligand moiety to a target.
- the connector moiety may be varied to control the spacing between two ligands. For example, in some cases, it may be desirable to adjust the spacing between two ligands so as, for instance, to achieve optimal binding of the bivalent compound to a target. In some cases, the connector moiety may be used to adjust the orientation of the ligands. In certain embodiments, the spacing and/or orientation the connector moiety relative to the ligand moiety can affect the binding affinity of the ligand moiety (e.g., a pharmacophore) to a target. In some cases, connector moieties with restricted degrees of freedom are preferred to reduce the entropic losses incurred upon the binding of a bivalent compound to its target biomolecule. In some embodiments, connector moieties with restricted degrees of freedom are preferred to promote cellular permeability of the bivalent compound.
- the ligand moiety e.g., a pharmacophore
- the connector moiety may be used for modular assembly of ligands.
- a connector moiety may comprise a functional group formed from reaction of a first and second molecule.
- a series of ligand moieties may be provided, where each ligand moiety comprises a common functional group that can participate in a reaction with a compatible functional group on a connector moiety.
- the connector moiety may comprise a spacer having a first functional group that forms a bond with a first ligand moiety and a second functional group that forms a bond with a second ligand moiety.
- Contemplated connector moieties may be any acceptable (e.g., pharmaceutically and/or chemically acceptable) bivalent linker.
- such connecter moieties may comprise 3 to 30 atoms, 3 to 20 atoms, 3 to 15 atoms, 3 to 10 atoms, 5 to 15 atoms, 10 to 20 atoms, 15 to 25 atoms, 20 to 30 atoms, or 10 to 30 atoms.
- the atoms may be connected in any suitable arrangement.
- the atoms may be connected to form a cyclic or acyclic, substituted or unsubstituted, branched or unbranched aliphatic moiety; cyclic or acyclic, substituted or unsubstituted, branched or unbranched heteroaliphatic moiety; substituted or unsubstituted phenyl or naphthyl moiety; substituted or unsubstituted heteroaryl moiety; or a combination thereof.
- a connector moiety may include a substituted or unsubstituted Ci-Cio alkylene, substituted or unsubstituted cycloalkylene, acyl, sulfone, phosphate, ester, carbamate, or amide.
- contemplat oieties may include polymeric connectors, such a polyethylene glycol (e.g., , where n is 1, 2, 3, 4, 5, 6, 7, 8, 9,
- X is O, S, NH, or -C(O)-) or other
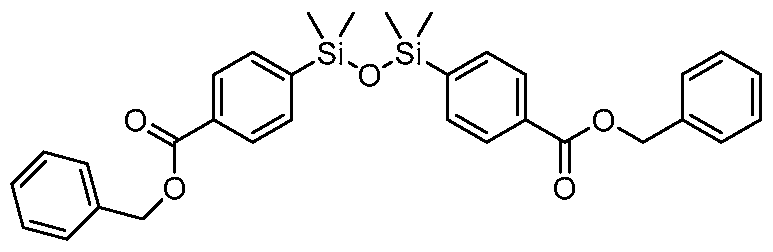
- the connector moiety may be substituted with -SiR 2 - and -SiR 2 -0-, where -SiR 2 - and -SiR 2 -0- are covalently bonded to form -SiR. 2 -0-SiR 2 -.
- a connector may be from about 7 atoms to about 13 atoms in length, or about 8 atoms to about 12 atoms, or about 9 atoms to about 1 1 atoms in length. For purposes of counting connector length when a ring is present in the connector group, the ring is counted as three atoms from one end to the other.
- R 7 and R 8 are selected, independently for each occurrence, from the group consisting of -OH, Ci- 6 alkyl, -0-Ci_ 6 alkyl, C 2 _ 6 alkenyl, C3_ 6 cycloalkyl, -Ci_ 6 alkyl- NR a R b , phenyl and heteroaryl; wherein Ci_ 6 alkyl, C 2 _ 6 alkenyl, C3_ 6 cycloalkyl, phenyl, and heteroaryl may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, hydroxyl, Ci- 6 alkyl, and phenyl; or
- R 7 and R 8 together with the silicon to which they are attached, form a 4-7 membered heterocyclic ring, optionally containing one, two, three, or four heteroatoms independently selected from O, S, or N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, and hydroxyl; R' is, independently for each occurrence, selected from the group consisting of hydrogen and Ci- 4 alkyl; wherein may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; and
- s is an integer from 1-15.
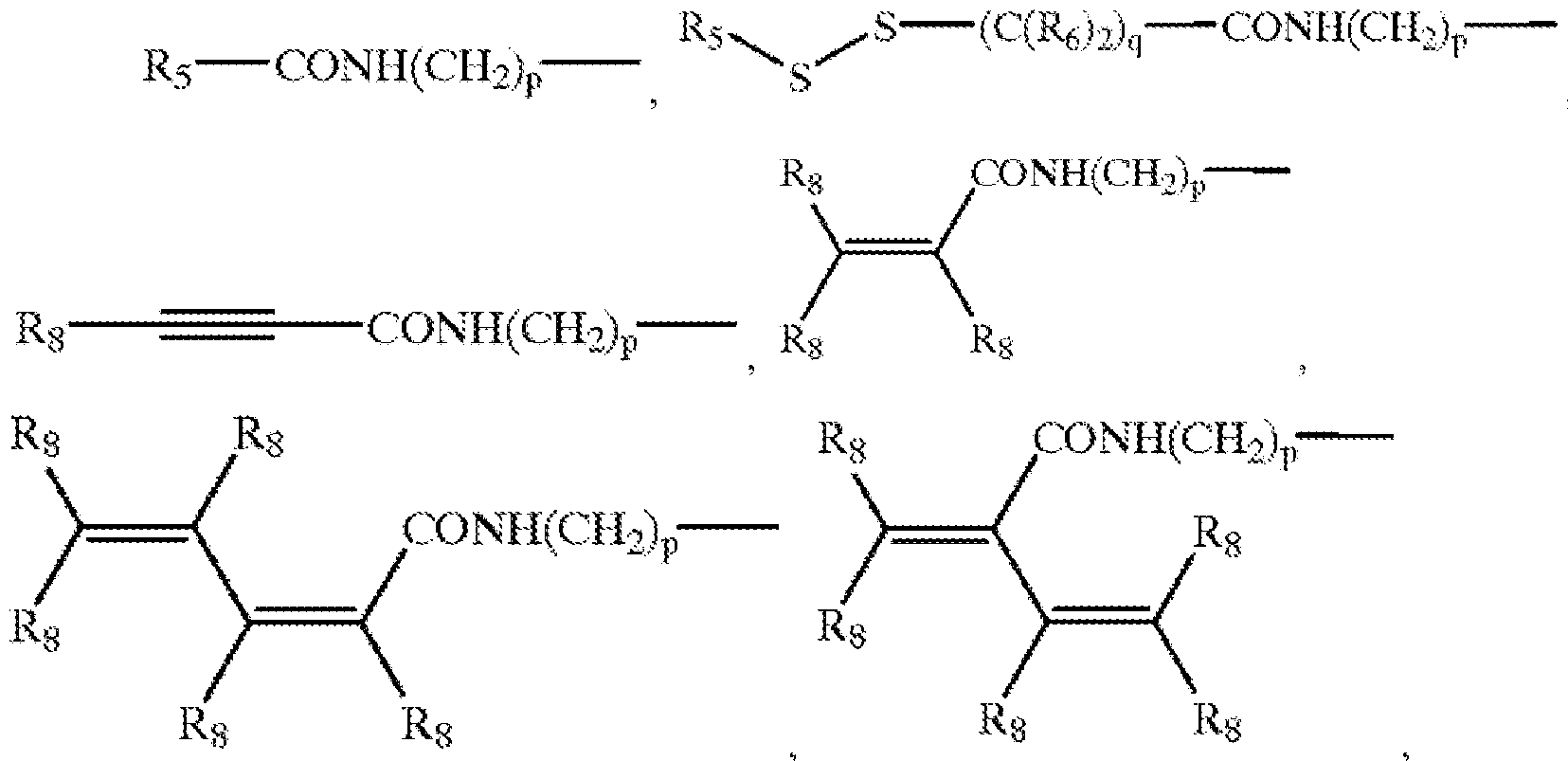
- a connector moiety may comprise:
- R 8 R 7 , R 8 R 7 , R 8 R 7 , and R 8 R 7 wherein thee first group and the Si of the second group are connected by an oxygen atom;
- R w is, independently for each occurrence, absent or selected from the group consisting of -Ci-4alkylene-, -0-Ci-4alkylene-, -N(R )-, -N(R )-Ci-4alkylene-, -0-, - C(0)Ci_ 4 alkylene-, -C(0)-0-Ci_ 4 alkylene-, -C 2 - 6 alkenylene-, -C 2 - 6 alkynylene-, -C3-
- Ci- 4 alkylene, C 2 - 6 alkenylene, C 2 - 6 alkynylene, C3_ 6 cycloalkylene, phenylene, and heteroarylene may be optionally substituted by one, two, three, or more substituents independently selected from the group consisting of Ci- 4 alkoxy, -C(0)Ci_ 4 alkyl, -C(0)-0-Ci_ 4 alkyl, -C(O)- NR a R b , halogen, cyano, hydroxyl, and phenyl; or R w and R 7 , together with the silicon to which they are attached, form a 3-8 membered heterocyclic ring, wherein the 3-8 membered ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, hydroxyl, and Ci- 6 al
- W 3 is, independently for each occurrence, (a) absent; or (b) selected from the group consisting of -Ci- 4 alkylene-, -0-Ci- 4 alkylene-, -C(0)-Ci_ 4 alkylene-, -N(R )-Ci_
- R 7 and R 8 are selected, independently for each occurrence, from the group consisting of -OH, Ci- 6 alkyl, -0-Ci_ 6 alkyl, C 2 - 6 alkenyl, C3_ 6 cycloalkyl, -Ci_ 6 alkyl- NR a R b , phenyl and heteroaryl; wherein Ci- 6 alkyl, C 2 - 6 alkenyl, C3_ 6 cycloalkyl, phenyl, and heteroaryl may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, hydroxyl, Ci- 6 alkyl, and phenyl; or
- R 7 and R 8 together with the silicon to which they are attached, form a 4-7 membered heterocyclic ring, optionally containing one, two, three, or four heteroatoms independently selected from O, S, or N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, and hydroxyl;
- R', R a , and R b are selected, independently for each occurrence, from the group consisting of hydrogen and C 1-4 alkyl; wherein Ci-4alkyl may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; or
- R a and R b together with the nitrogen to which they are attached, may form a 4-7 membered heterocyclic ring, optionally containing an additional heteroatom selected from O, S, and N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo and hydroxyl;
- BB independently for each occurrence, is a 4-7-membered cycloalkyl, heterocyclic, phenyl, naphthyl, or heteroaryl moiety, wherein the cycloalkyl, heterocyclic, phenyl, naphthyl, or heteroaryl moiety is optionally substituted with one, two, three, or more groups represented by R BB ; wherein R 7 , independently for each occurrence, may be optionally bonded to BB; and
- each R BB is selected, independently for each occurrence, from the group consisting of hydrogen, halogen, nitro, cyano, hydroxyl, amino, thio, -COOH, - CONHR', substituted or unsubstituted aliphatic, and substituted or unsubstituted heteroaliphatic; or two R together with the atoms to which they are attached form a fused 5- or 6-membered cycloalkyl or heterocyclic bicyclic ring system.
- a connector moiety may comprise:
- a first group selected from the group consisting of:
- R 7 and R 8 are as defined above.
- R 7 and R 8 are Ci_ 6 alkyl.
- the connector moiety is selected from the group consisting of:
- a contemplated connector moiety may be an optionally substituted unsaturated heteroaliphatie moiety, wherein the optionaliy substituted unsaturated heteroaliphatie moiety comprises at least one functional group, e.g., one, two. three, or four groups, selected from -SiR 2 -, -SiR 2 -0-, -NR-, -N(R)C(0)-, -CfO)N(R)-, -N(R)S0 2 -, -S0 2 N(R)- , -0-, ⁇ -( ⁇ ; ( ⁇ ; ⁇ -.
- the functional group may be positioned at any suitable position within the optionally substituted unsaturated heteroaliphatie moiety.
- the optionally substituted unsaturated heteroaliphatie moiety may comprise 3 to 30 atoms, 3 to 20 atoms, 3 to 15 atoms, 3 to 10 atoms, 5 to 15 atoms, 10 to 20 atoms, 15 to 25 atoms, 20 to 30 atoms, or 10 to 30 atoms.
- the optionally substituted " unsaturated heteroaliphatie moiety comprises at least one unsaturated group, e.g., an alkenylene group or an alkynylene group.
- the optionally substituted unsaturated heteroaliphatie moiety may comprise I to 10, 1 to 3, 2 to 4, 3 to 5, 4 to 6, 5 to 7, 6 to 8, 7 to 9, or 8 to 10 unsaturated groups.
- the optionally substituted unsaturated heteroaliphatie moiety may contain one, two, three, four, five, six, seven, eight, nine, or ten unsaturated groups.
- the optionally substituted unsaturated heteroaliphatie moiety comprises a pl rality of unsaturated groups
- the unsaturated groups may be alkenylene groups, alkynylene groups, or a mixture thereof.
- An unsaturated group may be positioned at any suitable position within the optionally substituted unsaturated heteroaliphatie moiety.
- an unsaturated group may be positioned adjacent to a ligand moiety (i.e., directly connected by a covalent bond) and/or a functional group.
- an unsaturated group and a ligand moiety may be separated by one or more atoms.
- the optionaliy substituted unsaturated heteroaliphatie moiety is directly connected to a first ligand moiety by a ftrst unsaturated group and directly connected to a second ligand moiety by a second unsaturated group.
- a connector may have the following structure: , where:
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20;
- R 1 and R 2 are, independently for each occurrence, selected from the group consisting of H, Ci- 6 alkyl, Ci- 6 heteroalkyl, phenyl, or heteroaryl, wherein alkyl, heteroalkyl, phenyl, and heteroaryl are optionally substituted with -OH, -NH 2 , -SH, -COOH, -C(0)NH 2 , halo, phenyl, and heteroaryl; or
- a connector may comprise a phenyl, naphthyl, or mono or bicyclic heteroaryl ring, each optionally substituted.
- a connector may comprise one or more of the following aryl structures:
- a connector may comprise a triazole ring having the following structure:
- a bivalent compound comprising a triazole-containing connector may have the following general structure:
- Such triazole-joined compounds may be formed, e.g., as a result of a "click" type reaction (i.e., an azide-alkyne cycloaddition).
- a first segment of a connector having a terminal alkyne and a second segment of a connector having a terminal azide may be joined by a "click" reaction to form a single connector joined by a triazole, as shown above.
- the first connector and the second connector each are less than or equal to 20 atoms in length, or in some embodiments each are less than or equal to 12 atoms in length.
- a triazole-containing connector may be represented by -
- the triazole-containing connector may be represented by:
- the optionally substituted unsaturated lieteroaliphati moiety may be: j Wherein .
- R Li is, independently for each occurrence, selected from the groups consisting of hydrogen or any suitable substituent
- W 2 is selected from the group consisting of -SiR 2 -, -SiR 2 -0-, -0-SiR 2 -, -NR-, ⁇
- a connecter moiety may maximally span from about 5A to about 50A, in some embodiments about 5A to about 25 A, in some embodiments about 20A to about 50A, and in some embodiments about 6A to about 15A in length. In another embodiment, a connecter moiety may maximally span less than about 30A, in some embodiments less than about 20A, and in some embodiments less than about IOA.
- a connecter moiety may maximally span from about 1 A to about 30A, in some embodiments about 1 A to about 20A, in some embodiments about 1 A to about IOA, in some embodiments about 1 A to about 5 A., in some embodiments about 20A to about 30A, in some embodiments about 15A to about 25A, in some embodiments about IOA to about 2()A, and in some embodiments about 5A to about 15.4, in length.
- a connector moiety may maximally span about 1 A, about 3 A, about SA, about 7 A, about 9A, about 1 lA, about 13 A, about 15 , about 17A, about 19A, , about 21 A, about 23 A, about 25 A, about 27A, or about 29A.
- a connector i.e., Q 1
- Q 1 is selected from the group consisting of:
- W 1 is, independently for each occurrence, NR.', O, or S;
- R' is, independently for each occurrence, selected from the group consisting of hydrogen and C 1-4 alkyl; wherein C 1-4 alkyl may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; and
- s is an integer from 1-10.
- a connector is selected from the group consisting of: -S-(C 2 -6alkylene-0) s -C 2 -6alkylene-S-; -0-(CH 2 -CH 2 -0) s -; and -O-Ci-isalkylene- 0-; wherein s is an integer from 1-10.
- the connector is -0-(CH 2 -CH 2 -0) s -.
- the connector is selected from the group consisting of: -S-(CH 2 -CH 2 -CH 2 -0) s - CH 2 -CH 2 -CH 2 -S-; -S-(CH 2 -CH 2 -0) s -CH 2 -CH 2 -S-; and -0-(CH 2 ) u -0-; wherein u is an integer from 1-15.
- u is 4, 5, 6, 7, 8, 9, 10, 1 1, 12, 13, 14, or 15.
- s is 2, 3, 4, 5, 6, 7, or 8.
- a connector i.e., Q 1
- Q 1 is selected from the group consisting of:
- W 1 is, independently for each occurrence, N, O, or S; s is an integer from 1-10; and t is an integer from 3-10.
- a connector is selected from the group consisting of: -S-(C3_ 6 alkylene-0) s -Ci_ 6 alkylene-S-; -S-(CH 2 -CH 2 -0) t -Ci_ 6 alkylene-S-; -0-(CH 2 -CH 2 -0) s -; and -0-Ci_i 5 alkylene-0-; wherein s is an integer from 1-10, and t is an integer from 3-10.
- the connector is -0-(CH 2 -CH 2 -0) s -.
- the connector is selected from the group consisting of: -S-(CH 2 -CH 2 -CH 2 -0) s -CH 2 -CH 2 -CH 2 -S-; -S-(CH 2 -CH 2 -0) t -CH 2 -CH 2 - S-; and -0-(CH 2 ) u -0-; wherein u is an integer from 1-15.
- u is 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15.
- s is 2, 3, 4, 5, 6, 7, or 8.
- the synthetic route in Scheme Xa illustrates a general method for preparing ligand-connector derivatives. The method involves attaching the desired substituents to the carbonyl substituent.
- the desired connector can be installed by reacting the ligand 1 with the appropriate nucleophile 2 to provide 3 (ligand-connector derivative).
- Scheme Xa provides for a connector (Q 1 ).
- the desired connector attached at the carbonyl substituent can be installed by reacting carboxylic acid 1 with common coupling reagents such as l-ethyl-3-(3- dimethylaminopropyl)carbodiimide (EDC) and hydroxybenzotriazole (HOBt) and then further reacting the resultant activated ester with the appropriate nucleophile, for example, amine 2, to provide 3 (ligand-connector derivative).
- EDC l-ethyl-3-(3- dimethylaminopropyl)carbodiimide
- HOBt hydroxybenzotriazole
- the connector may be selected from the group consisting of:
- n may be 0, 1, 2, 3,4 or i
- Q 1 may generally be represented for example, by: where n may be 0, 1, 2, 3, 4, 5, or 6.
- Any free amino group seen in the connector examples of Table A above may be functionalized further to include additional functional groups.
- the attachment point on the ligand may be further elaborated to incorporate not only a connector moiety, but also a second ligand, as e.g., represented by:
- the Q x -P 2 moiety may be formed from direct attachment of Q x -P 2 to the carbonyl, or the Q x -P 2 moiety may be formed from the further functionalization of any free amino group seen in the -NH-R examples (i.e., Q 1 examples) of Table A above to include the second ligand moiety (P 2 ).
- Q x -P 2 may be represented by the structure:
- n 0, 1, 2, 3, 4, or 5, e.g. n is 1 to 5.
- Q x -P 2 may be represented by the structure:
- n 0, 1, 2, 3, 4 or 5, e.g. n is 1 to 5.
- the synthetic route in Scheme Xb illustrates another general method for preparing ligand-connector derivatives (i.e., P ⁇ Q 1 derivatives). The method involves attaching the desired substituents to an -OH group (e.g., a phenol group) of the ligand.
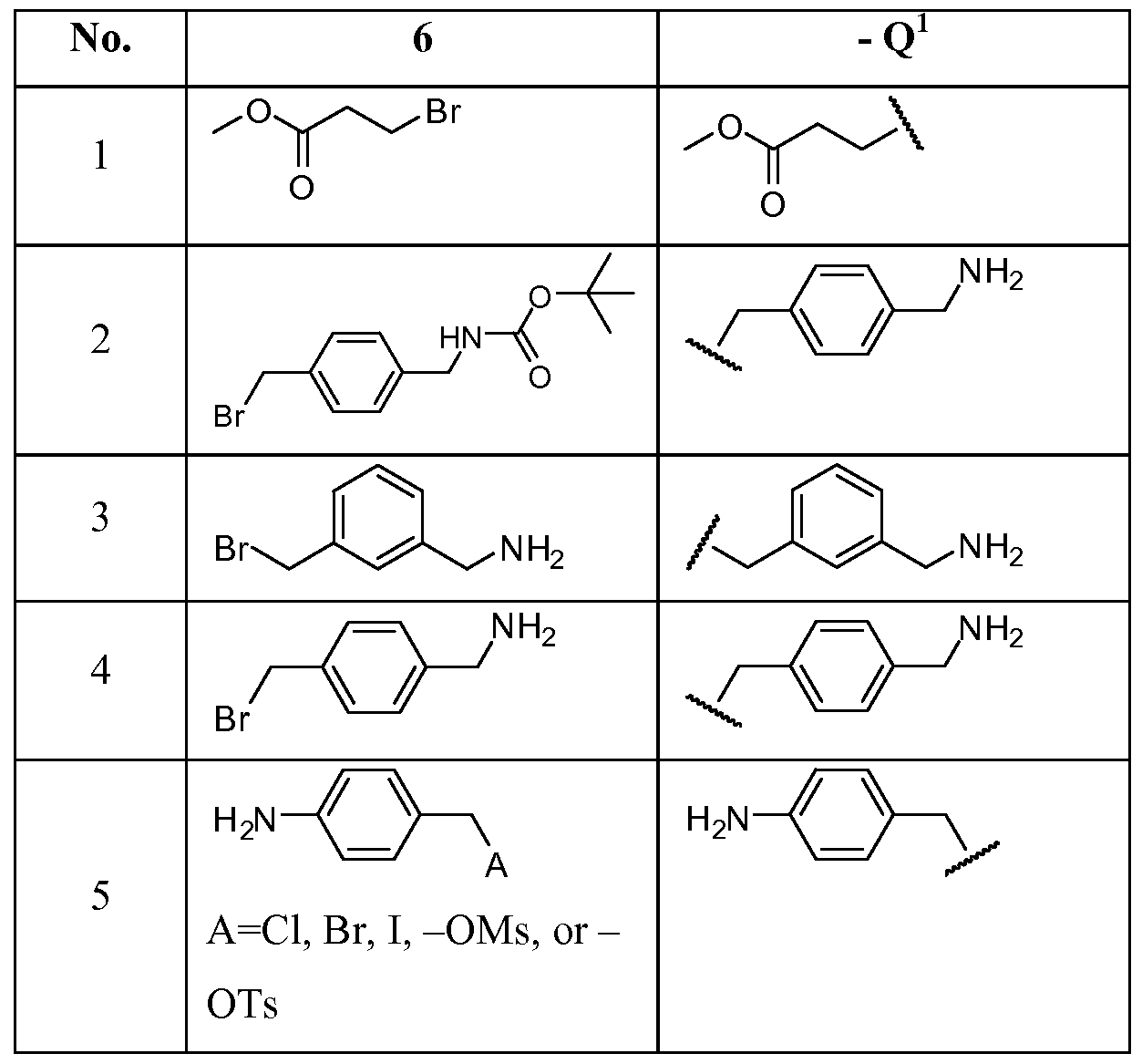
- an -OH group e.g., a phenol group
- X Br, CI, I, O s or OTs
- the connector (Q 1 ) may be selected from the group consisting of:
- n 1, 2, 3, 4 or 5.
- Any free amino group seen in the Q 1 examples of Table B above may be functionalized further to include additional functional groups, e.g., a benzoyl moiety.
- the P 1 may be further elaborated to incorporate not only the connector moiety (Q 1 ), but also a second ligand (P 2 ), as represented by:
- X Br, CI, I, OMs or OTs
- the Q x -P 2 moiety may be formed from direct attachment of Q x -P 2 to the phenyl ether, or the Q x -P 2 moiety may be formed from the further functionalization of any free amino group seen in the Q 1 examples of Table B above to include the second ligand (P 2 ).
- the synthetic route in Scheme Xc illustrates a general method for preparing ligand-connector derivatives. The method involves attaching the desired carbonyl substituents to the free amine to form an amide, urea, or carbamate. For example, the carbonyl group can be installed by reacting amine 12 (see Scheme Xd) with carboxylic acid 13 to provide 14
- -C(0)R i.e., Q 1
- Q 1 Q 1
- contemplated bivalent compounds may be administered to a patient in need thereof.
- a method of administering a patient may be administered to a patient in need thereof.
- the target biomolecule may be a protein. In other embodiments, the target biomolecule may be nucleic acid.
- a method of modulating two or more target biomolecule binding sites is provided, e.g., of Bcr-Abl tyrosine kinase.
- a compound may be used to inhibit or facilitate protein-protein interactions.
- a compound may be capable of activating or inactivating a signaling pathway.
- a compound may bind to a target protein and affect the conformation of the target protein such that the target protein is more biologically active as compared to when the compound does not bind the target protein.
- the compound may bind to one region (e.g., domain) of a target molecule.
- the compound may bind to two regions of a target molecule.
- the compound may bind to a first region of a first target molecule and a second region of a second target molecule.
- P 1 and P 2 of Formula I may each be capable of binding to a Bcr-Abl tyrosine kinase binding site.
- one of P 1 may be capable of binding to the myristoyl binding pocket of Bcr-Abl tyrosine kinase and and P 2 may be capable of binding to the ATP binding pocket of Bcr-Abl tyrosine kinase.
- a contemplated bivalent compound may be capable of binding to a first binding site and a second binding site, wherein the protein domain is within, e.g., about 40 A, or about 50 A, of the binding site.
- the compounds contemplated herein may be used in a method for treating diseases or conditions for which a Bcr-Abl tyrosine kinase inhibitor is indicated, for example, a compound may be used for treating treating cancer, such as chronic myelogenous leukemia.
- a method of treating a disease associated with a Bcr-Abl tyrosine kinase in a patient in need thereof is provided herein.
- a compound in the manufacture of a medicament for the treatment of diseases or conditions for which a Bcr-Abl tyrosine kinase inhibitor inhibitor is indicated is indicated.
- a compound or a pharmaceutically acceptable salt thereof in the manufacture of a medicament for the treatment of cancer such as chronic myelogenous leukemia.
- cancers e.g., cancers such as including hematological, epithelial including lung, breast and colon carcinomas, mesenchymal, hepatic, renal and neurological tumors
- contemplated herein is a method of treating squamous cell carcinoma, midline carcinoma or leukemia such as acute myeloid leukemia in a patient in need thereof comprising administering a contemplated bivalent compound.
- a ligand moiety (e.g., a pharmacophore) may have a molecular weight between 50 Da and 2000 Da, in some embodiments between 50 Da and 1500 Da, in some embodiments, between 50 Da and 1000 Da, and in some embodiments, between 50 Da and 500 Da. In certain embodiments, a ligand moiety may have a molecular weight of less than 2000 Da, in some embodiments, less than 1000 Da, and in some embodiments less than 500 Da.
- the compound utilized by one or more of the foregoing methods is one of the generic, subgeneric, or specific compounds described herein.
- compositions may be administered to patients (animals and humans) in need of such treatment in dosages that will provide optimal pharmaceutical efficacy. It will be appreciated that the dose required for use in any particular application will vary from patient to patient, not only with the particular compound or composition selected, but also with the route of administration, the nature of the condition being treated, the age and condition of the patient, concurrent medication or special diets then being followed by the patient, and other factors which those skilled in the art will recognize, with the appropriate dosage ultimately being at the discretion of the attendant physician.
- a compound may be administered orally, subcutaneously, topically, parenterally, by inhalation spray or rectally in dosage unit formulations containing conventional non-toxic pharmaceutically acceptable carriers, adjuvants, and vehicles.
- Parenteral administration may include subcutaneous injections, intravenous or intramuscular injections, or infusion techniques.
- Treatment can be continued for as long or as short a period as desired.
- the compositions may be administered on a regimen of, for example, one to four or more times per day.
- a suitable treatment period can be, for example, at least about one week, at least about two weeks, at least about one month, at least about six months, at least about 1 year, or indefinitely.
- a treatment period can terminate when a desired result, for example a partial or total alleviation of symptoms, is achieved.
- compositions comprising bivalent compounds as disclosed herein formulated together with a pharmaceutically acceptable carrier provided.
- the present disclosure provides pharmaceutical compositions bivalent compounds as disclosed herein formulated together with one or more pharmaceutically acceptable carriers.
- These formulations include those suitable for oral, rectal, topical, buccal, parenteral (e.g., subcutaneous, intramuscular, intradermal, or intravenous) rectal, vaginal, or aerosol administration, although the most suitable form of administration in any given case will depend on the degree and severity of the condition being treated and on the nature of the particular compound being used.
- disclosed compositions may be formulated as a unit dose, and/or may be formulated for oral or subcutaneous administration.
- Exemplary pharmaceutical compositions may be used in the form of a pharmaceutical preparation, for example, in solid, semisolid, or liquid form, which contains one or more of the compounds, as an active ingredient, in admixture with an organic or inorganic carrier or excipient suitable for external, enteral, or parenteral applications.
- the active ingredient may be compounded, for example, with the usual non-toxic, pharmaceutically acceptable carriers for tablets, pellets, capsules, suppositories, solutions, emulsions, suspensions, and any other form suitable for use.
- the active object compound is included in the pharmaceutical composition in an amount sufficient to produce the desired effect upon the process or condition of the disease.
- the principal active ingredient may be mixed with a pharmaceutical carrier, e.g., conventional tableting ingredients such as corn starch, lactose, sucrose, sorbitol, talc, stearic acid, magnesium stearate, dicalcium phosphate or gums, and other pharmaceutical diluents, e.g., water, to form a solid
- a pharmaceutical carrier e.g., conventional tableting ingredients such as corn starch, lactose, sucrose, sorbitol, talc, stearic acid, magnesium stearate, dicalcium phosphate or gums, and other pharmaceutical diluents, e.g., water
- preformulation composition containing a homogeneous mixture of a compound, or a non-toxic pharmaceutically acceptable salt thereof.
- preformulation compositions as homogeneous, it is meant that the active ingredient is dispersed evenly throughout the composition so that the composition may be readily subdivided into equally effective unit dosage forms such as tablets, pills and capsules.
- solid dosage forms for oral administration capsules, tablets, pills, dragees, powders, granules and the like
- the subject composition is mixed with one or more
- pharmaceutically acceptable carriers such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose and/or acacia; (3) humectants, such as glycerol; (4)
- disintegrating agents such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds; (7) wetting agents, such as, for example, acetyl alcohol and glycerol monostearate; (8) absorbents, such as kaolin and bentonite clay; (9) lubricants, such a talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, and mixtures thereof; and (10) coloring agents.
- solution retarding agents such as paraffin
- absorption accelerators such as quaternary ammonium compounds
- wetting agents such as, for example, acetyl alcohol and glycerol monostearate
- absorbents such as kaolin and bentonite clay
- lubricants such a talc,
- compositions may also comprise buffering agents.
- Solid compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
- a tablet may be made by compression or molding, optionally with one or more accessory ingredients.
- Compressed tablets may be prepared using binder (for example, gelatin or hydroxypropylmethyl cellulose), lubricant, inert diluent, preservative, disintegrant (for example, sodium starch glycolate or cross-linked sodium carboxymethyl cellulose), surface- active or dispersing agent.
- Molded tablets may be made by molding in a suitable machine a mixture of the subject composition moistened with an inert liquid diluent. Tablets, and other solid dosage forms, such as dragees, capsules, pills and granules, may optionally be scored or prepared with coatings and shells, such as enteric coatings and other coatings well known in the pharmaceutical-formulating art.
- compositions for inhalation or insufflation include solutions and suspensions in pharmaceutically acceptable, aqueous or organic solvents, or mixtures thereof, and powders.
- Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, microemulsions, solutions, suspensions, syrups and elixirs.
- the liquid dosage forms may contain inert diluents commonly used in the art, such as, for example, water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, cyclodextrins and mixtures thereof.
- inert diluents commonly used in the art, such as, for example, water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate
- Suspensions in addition to the subject composition, may contain suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
- suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
- Formulations for rectal or vaginal administration may be presented as a suppository, which may be prepared by mixing a subject composition with one or more suitable non-irritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the body cavity and release the active agent.
- suitable non-irritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the body cavity and release the active agent.
- Dosage forms for transdermal administration of a subject composition includes powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants.
- the active component may be mixed under sterile conditions with a pharmaceutically acceptable carrier, and with any preservatives, buffers, or propellants which may be required.
- the ointments, pastes, creams and gels may contain, in addition to a subject composition, excipients, such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
- excipients such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
- Powders and sprays may contain, in addition to a subject composition, excipients such as lactose, talc, silicic acid, aluminum hydroxide, calcium silicates and polyamide powder, or mixtures of these substances.
- Sprays may additionally contain customary propellants, such as chlorofluorohydrocarbons and volatile unsubstituted hydrocarbons, such as butane and propane.
- compositions and compounds may alternatively be administered by aerosol.
- an aqueous aerosol is made by formulating an aqueous solution or suspension of a subject composition together with conventional pharmaceutically acceptable carriers and stabilizers.
- the carriers and stabilizers vary with the requirements of the particular subject composition, but typically include non-ionic surfactants (T weens, Pluronics, or polyethylene glycol), innocuous proteins like serum albumin, sorbitan esters, oleic acid, lecithin, amino acids such as glycine, buffers, salts, sugars, or sugar alcohols. Aerosols generally are prepared from isotonic solutions.
- compositions suitable for parenteral administration comprise a subject composition in combination with one or more pharmaceutically-acceptable sterile isotonic aqueous or non-aqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents.
- aqueous and non-aqueous carriers examples include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate and cyclodextrins.
- polyols such as glycerol, propylene glycol, polyethylene glycol, and the like
- vegetable oils such as olive oil
- injectable organic esters such as ethyl oleate and cyclodextrins.
- Proper fluidity may be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants
- enteral pharmaceutical formulations including a disclosed pharmaceutical composition comprising bivalent compounds, an enteric material; and a pharmaceutically acceptable carrier or excipient thereof are provided.
- Enteric materials refer to polymers that are substantially insoluble in the acidic environment of the stomach, and that are predominantly soluble in intestinal fluids at specific pHs.
- the small intestine is the part of the gastrointestinal tract (gut) between the stomach and the large intestine, and includes the duodenum, jejunum, and ileum.
- the pH of the duodenum is about 5.5
- the pH of the jejunum is about 6.5
- the pH of the distal ileum is about 7.5.
- enteric materials are not soluble, for example, until a pH of about 5.0, of about 5.2, of about 5.4, of about 5.6, of about 5.8, of about 6.0, of about 6.2, of about 6.4, of about 6.6, of about 6.8, of about 7.0, of about 7.2, of about 7.4, of about 7.6, of about 7.8, of about 8.0, of about 8.2, of about 8.4, of about 8.6, of about 8.8, of about 9.0, of about 9.2, of about 9.4, of about 9.6, of about 9.8, or of about 10.0.
- Exemplary enteric materials include cellulose acetate phthalate (CAP), hydroxypropyl methylcellulose phthalate (HPMCP), polyvinyl acetate phthalate (PVAP), hydroxypropyl methylcellulose acetate succinate (HPMCAS), cellulose acetate trimellitate, hydroxypropyl methylcellulose succinate, cellulose acetate succinate, cellulose acetate hexahydrophthalate, cellulose propionate phthalate, cellulose acetate maleat, cellulose acetate butyrate, cellulose acetate propionate, copolymer of methylmethacrylic acid and methyl methacrylate, copolymer of methyl acrylate, methylmethacrylate and methacrylic acid, copolymer of methylvinyl ether and maleic anhydride (Gantrez ES series), ethyl methyacrylate-methylmethacrylate- chlorotrimethylammonium ethyl acrylate copolymer, natural resins such
- kits are provided containing one or more compositions.
- Such kits include a suitable dosage form such as those described above and instructions describing the method of using such dosage form to treat a disease or condition.
- the instructions would direct the consumer or medical personnel to administer the dosage form according to administration modes known to those skilled in the art.
- kits could advantageously be packaged and sold in single or multiple kit units.
- An example of such a kit is a so-called blister pack.
- Blister packs are well known in the packaging industry and are being widely used for the packaging of pharmaceutical unit dosage forms (tablets, capsules, and the like). Blister packs generally consist of a sheet of relatively stiff material covered with a foil of a preferably transparent plastic material. During the packaging process recesses are formed in the plastic foil.
- the recesses have the size and shape of the tablets or capsules to be packed.
- the tablets or capsules are placed in the recesses and the sheet of relatively stiff material is sealed against the plastic foil at the face of the foil which is opposite from the direction in which the recesses were formed.
- the tablets or capsules are sealed in the recesses between the plastic foil and the sheet.
- the strength of the sheet is such that the tablets or capsules can be removed from the blister pack by manually applying pressure on the recesses whereby an opening is formed in the sheet at the place of the recess. The tablet or capsule can then be removed via said opening.
- a memory aid on the kit, e.g., in the form of numbers next to the tablets or capsules whereby the numbers correspond with the days of the regimen which the tablets or capsules so specified should be ingested.
- a memory aid is a calendar printed on the card, e.g., as follows "First Week, Monday, Tuesday, . . . etc. . . . Second Week, Monday, Tuesday, . . . " etc.
- a “daily dose” can be a single tablet or capsule or several pills or capsules to be taken on a given day.
- a daily dose of a first compound can consist of one tablet or capsule while a daily dose of the second compound can consist of several tablets or capsules and vice versa.
- the memory aid should reflect this.
- compositions that include a second active agent, or administering a second active agent.
- the compounds, as described herein may be substituted with any number of substituents or functional moieties.
- substituted whether preceded by the term “optionally” or not, and substituents contained in formulas, refer to the replacement of hydrogen radicals in a given structure with the radical of a specified substituent.
- the substituent when more than one position in any given structure may be substituted with more than one substituent selected from a specified group, the substituent may be either the same or different at every position.
- the term "substituted" is contemplated to include all permissible substituents of organic and inorganic compounds.
- the permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, aromatic and non-aromatic substituents of organic compounds.
- heteroatoms such as nitrogen may have hydrogen substituents and/or any permissible substituents of organic compounds described herein which satisfy the valencies of the heteroatoms.
- substituents include acyl; aliphatic; heteroaliphatic; phenyl; naphthyl; heteroaryl; arylalkyl; heteroarylalkyl; alkoxy; cycloalkoxy;
- the compounds described herein are not intended to be limited in any manner by the permissible substituents of organic compounds. In some embodiments, combinations of substituents and variables described herein may be preferably those that result in the formation of stable compounds.
- stable refers to compounds which possess stability sufficient to allow manufacture and which maintain the integrity of the compound for a sufficient period of time to be detected and preferably for a sufficient period of time to be useful for the purposes detailed herein.
- acyl refers to a moiety that includes a carbonyl group.
- an acyl group may have a general formula selected from - C(0)R x ; -C0 2 (Rx); -C(0)N(R x ) 2 ; -OC(0)R x ; -OC0 2 R x ; and -OC(0)N(R x ) 2 ; wherein each occurrence of R x independently includes, but is not limited to, hydrogen, aliphatic,
- heteroaliphatic, phenyl, naphthyl, heteroaryl, arylalkyl, or heteroarylalkyl wherein any of the aliphatic, heteroaliphatic, arylalkyl, or heteroarylalkyl substituents described above and herein may be substituted or unsubstituted, branched or unbranched, cyclic or acyclic, and wherein any of the phenyl, naphthyl, or heteroaryl substituents described above and herein may be substituted or unsubstituted.
- aliphatic includes both saturated and unsaturated, straight chain (i.e., unbranched), branched, acyclic, cyclic, or polycyclic aliphatic
- aliphatic is intended herein to include, but is not limited to, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, and cycloalkynyl moieties.
- heteroaliphatic refers to aliphatic moieties that contain one or more oxygen, sulfur, nitrogen, phosphorus, or silicon atoms, e.g., in place of carbon atoms.
- Heteroaliphatic moieties may be branched, unbranched, cyclic or acyclic and include saturated and unsaturated heterocycles such as morpholino, pyrrolidinyl, etc.
- aryl refers to stable mono- or polycyclic, heterocyclic, polycyclic, and polyheterocyclic unsaturated moieties having preferably 3-14 carbon atoms, each of which may be substituted or unsubstituted.
- Substituents include, but are not limited to, any of the previously mentioned substituents, i.e., the substituents recited for aliphatic moieties, or for other moieties as disclosed herein, resulting in the formation of a stable compound.
- aryl or aromatic refers to a mono- or bicyclic carbocyclic ring system having one or two aromatic rings selected from phenyl, naphthyl, tetrahydronaphthyl, indanyl, and indenyl.
- heteroaryl refers to a cyclic aromatic radical having from five to ten ring atoms of which one ring atom is selected from the group consisting of S, O, and N; zero, one, or two ring atoms are additional heteroatoms independently selected from the group consisting of S, O, and N; and the remaining ring atoms are carbon, the radical being joined to the rest of the molecule via any of the ring atoms.
- Heteroaryl moieties may be selected from: pyridyl, pyrazinyl, pyrimidinyl, pyrrolyl, pyrazolyl, imidazolyl, thiazolyl, oxazolyl, isooxazolyl, thiadiazolyl,oxadiazolyl, thiophenyl, furanyl, quinolinyl, isoquinolinyl, and the like.
- aryl, aromatic, heteroaryl, and heteroaromatic groups described herein can be unsubstituted or substituted, wherein substitution includes replacement of one, two, three, or more of the hydrogen atoms thereon independently with a group selected from: Ci_ 6 alkyl; phenyl; heteroaryl; benzyl; heteroarylalkyl; Ci_ 6 alkoxy; Ci_ 6 cycloalkoxy; Ci_ 6 heterocyclylalkoxy; heterocyclyloxyalkyl; C2- 6 alkenyloxy; C 2 _
- heterocyclic refers to an aromatic or non-aromatic, partially unsaturated or fully saturated, 3- to 10-membered ring system, which includes single rings of 3 to 8 atoms in size and bi- and tri-cyclic ring systems which may include aromatic five- or six-membered aryl or aromatic heterocyclic groups fused to a non-aromatic ring.
- heterocyclic rings include those having from one to three heteroatoms independently selected from the group consisting of oxygen, sulfur, and nitrogen, in which the nitrogen and sulfur heteroatoms may optionally be oxidized and the nitrogen heteroatom may optionally be quaternized.
- the term heterocyclic refers to a non-aromatic 5-, 6-, or 7-membered ring or a polycyclic group wherein at least one ring atom is a heteroatom selected from the group consisting of O, S, and N (wherein the nitrogen and sulfur heteroatoms may be optionally oxidized), including, but not limited to, a bi- or tri-cyclic group, comprising fused six-membered rings having between one and three heteroatoms independently selected from the group consisting of the oxygen, sulfur, and nitrogen, wherein (i) each 5-membered ring has 0 to 2 double bonds, each 6-membered ring has 0 to 2 double bonds, and each 7-membered ring has 0 to 3 double bonds, (ii) the nitrogen and
- alkenyl refers to an unsaturated straight or branched hydrocarbon having at least one carbon-carbon double bond, such as a straight or branched group of 2-6 or 3-4 carbon atoms, referred to herein for example as C2- 6 alkenyl, and C 3- 4 alkenyl, respectively.
- alkenyl groups include, but are not limited to, vinyl, allyl, butenyl, pentenyl, etc.
- alkenyloxy refers to a straight or branched alkenyl group attached to an oxygen (alkenyl-O).
- alkenoxy groups include, but are not limited to, groups with an alkenyl group of 3-6 carbon atoms referred to herein as C3_ 6 alkenyloxy.
- alkenyloxy groups include, but are not limited to allyloxy, butenyloxy, etc.
- alkoxy refers to a straight or branched alkyl group attached to an oxygen (alkyl-O-).
- exemplary alkoxy groups include, but are not limited to, groups with an alkyl group of 1-6 or 2-6 carbon atoms, referred to herein as Ci- 6 alkoxy, and C 2 - C6alkoxy, respectively.
- exemplary alkoxy groups include, but are not limited to methoxy, ethoxy, isopropoxy, etc.
- alkoxycarbonyl refers to a straight or branched alkyl group attached to oxygen, attached to a carbonyl group (alkyl-O-C(O)-).
- exemplary alkoxycarbonyl groups include, but are not limited to, alkoxycarbonyl groups of 1-6 carbon atoms, referred to herein as Ci_ 6 alkoxycarbonyl.
- exemplary alkoxycarbonyl groups include, but are not limited to, methoxycarbonyl, ethoxycarbonyl, t-butoxycarbonyl, etc.
- alkynyloxy refers to a straight or branched alkynyl group attached to an oxygen (alkynyl-O)).
- exemplary alkynyloxy groups include, but are not limited to, propynyloxy.
- alkyl refers to a saturated straight or branched hydrocarbon, for example, such as a straight or branched group of 1-6, 1-4, or 1-3 carbon atoms, referred to herein as Ci- 6 alkyl, Ci_ 4 alkyl, and Ci_ 3 alkyl, respectively.
- Exemplary alkyl groups include, but are not limited to, methyl, ethyl, propyl, isopropyl, 2-methyl- 1 -propyl, 2- methyl-2-propyl, 2-methyl-l -butyl, 3 -methyl- 1 -butyl, 3-methyl-2-butyl, 2,2-dimethyl-l -propyl, 2-methyl- 1-pentyl, 3 -methyl- 1-pentyl, 4-methyl-l-pentyl, 2-methyl-2-pentyl, 3-methyl-2- pentyl, 4-methyl-2-pentyl, 2,2-dimethyl-l -butyl, 3,3-dimethyl-l-butyl, 2-ethyl-l -butyl, butyl, isobutyl, t-butyl, pentyl, isopentyl, neopentyl, hexyl, etc.
- alkylene refers to a bivalent saturated straight or branched hydrocarbon, for example, such as a straight or branched group of 1-6, 1-4, or 1-3 carbon atoms, referred to herein as -Ci_ 6 alkylene-, -Ci_ 4 alkylene-, and -Ci_ 3 alkylene-, respectively, where the alkylene has two open valences.
- Exemplary alkyl groups include, but are not limited to, methylene, ethylene, propylene, isopropylene, 2-methyl-l -propylene, 2- methyl-2-propylene, 2-methyl-l -butylene, 3 -methyl- 1-butylene, 3-methyl-2-butylene, 2,2- dimethyl-1 -propylene, 2-methyl-l -pentylene, 3 -methyl- 1-pentylene, 4-methyl-l-pentylene, 2- methyl-2-pentylene, 3-methyl-2-pentylene, 4-methyl-2-pentylene, 2,2-dimethyl- 1-butylene, 3, 3 -dimethyl- 1-butylene, 2-ethyl- 1-butylene, butylene, isobutylene, t-butylene, pentylene, isopentylene, neopentylene, hexylene, etc.
- alkylcarbonyl refers to a straight or branched alkyl group attached to a carbonyl group (alkyl-C(O)-).
- exemplary alkylcarbonyl groups include, but are not limited to, alkylcarbonyl groups of 1-6 atoms, referred to herein as Ci_
- alkylcarbonyl groups include, but are not limited to, acetyl, propanoyl, isopropanoyl, butanoyl, etc.
- alkynyl refers to an unsaturated straight or branched hydrocarbon having at least one carbon-carbon triple bond, such as a straight or branched group of 2-6, or 3-6 carbon atoms, referred to herein as C 2 - 6 alkynyl, and C3_ 6 alkynyl, respectively.
- alkynyl groups include, but are not limited to, ethynyl, propynyl, butynyl, pentynyl, hexynyl, methylpropynyl, etc.
- carbonyl refers to the radical -C(O)-.
- carboxylic acid refers to a group of formula -CO 2 H.
- cyano refers to the radical -CN.
- cycloalkoxy refers to a cycloalkyl group attached to an oxygen (cycloalkyl-O-).
- cycloalkyl refers to a monocyclic saturated or partially unsaturated hydrocarbon group of for example 3-6, or 4-6 carbons, referred to herein, e.g., as C3_ 6 cycloalkyl or C4_ 6 cycloalkyl and derived from a cycloalkane.
- exemplary cycloalkyl groups include, but are not limited to, cyclohexyl, cyclohexenyl, cyclopentyl, cyclobutyl or, cyclopropyl.
- halo or halogen as used herein refer to F, CI, Br, or I.
- heterocyclylalkoxy refers to a heterocyclyl- alkyl-O- group.
- heterocyclyloxyalkyl refers to a heterocyclyl-O-alkyl- group.
- heterocyclyloxy refers to a heterocyclyl-O- group.
- heteroaryloxy refers to a heteroaryl-O- group.
- hydroxy and “hydroxyl” as used herein refers to the radical -OH.
- connection refers to an atom or a collection of atoms optionally used to link interconnecting moieties, such as a disclosed connecting moiety (i.e., linker) and a pharmacophore.
- Contemplated connectors are generally hydrolytically stable.
- Treating includes any effect, e.g., lessening, reducing, modulating, or eliminating, that results in the improvement of the condition, disease, disorder and the like.
- “Pharmaceutically or pharmacologically acceptable” include molecular entities and compositions that do not produce an adverse, allergic, or other untoward reaction when administered to an animal, or a human, as appropriate.
- preparations should meet sterility, pyrogenicity, general safety and purity standards as required by FDA Office of Biologies standards.
- pharmaceutically acceptable carrier or “pharmaceutically acceptable excipient” as used herein refers to any and all solvents, dispersion media, coatings, isotonic and absorption delaying agents, and the like, that are compatible with pharmaceutical
- compositions may also contain other active compounds providing supplemental, additional, or enhanced therapeutic functions.
- composition refers to a composition comprising at least one compound as disclosed herein formulated together with one or more pharmaceutically acceptable carriers.
- “Individual,” “patient,” or “subject” are used interchangeably and include any animal, including mammals, preferably mice, rats, other rodents, rabbits, dogs, cats, swine, cattle, sheep, horses, or primates, and most preferably humans.
- the compounds can be administered to a mammal, such as a human, but can also be administered to other mammals such as an animal in need of veterinary treatment, e.g., domestic animals (e.g., dogs, cats, and the like), farm animals (e.g., cows, sheep, pigs, horses, and the like) and laboratory animals (e.g., rats, mice, guinea pigs, and the like).
- the mammal treated is desirably a mammal in which treatment of obesity, or weight loss is desired.
- “Modulation” includes antagonism (e.g., inhibition), agonism, partial antagonism and/or partial agonism.
- the term "therapeutically effective amount” means the amount of the subject compound that will elicit the biological or medical response of a tissue, system, animal, or human that is being sought by the researcher, veterinarian, medical doctor, or other clinician.
- the compounds are administered in therapeutically effective amounts to treat a disease.
- a therapeutically effective amount of a compound is the quantity required to achieve a desired therapeutic and/or prophylactic effect, such as an amount which results in weight loss.
- salt(s) refers to salts of acidic or basic groups that may be present in compounds used in the present compositions.
- Compounds included in the present compositions that are basic in nature are capable of forming a wide variety of salts with various inorganic and organic acids.
- the acids that may be used to prepare pharmaceutically acceptable acid addition salts of such basic compounds are those that form non-toxic acid addition salts, i.e., salts containing pharmacologically acceptable anions, including but not limited to malate, oxalate, chloride, bromide, iodide, nitrate, sulfate, bisulfate, phosphate, acid phosphate, isonicotinate, acetate, lactate, salicylate, citrate, tartrate, oleate, tannate, pantothenate, bitartrate, ascorbate, succinate, maleate, gentisinate, fumarate, gluconate, glucaronate, saccharate, formate, benzoate, glutamate, methanesulfonate, ethanesulfonate, benzenesulfonate, >-toluenesulfonate and pamoate (i.e., l, l'-methylene-
- Compounds included in the present compositions that are acidic in nature are capable of forming base salts with various pharmacologically acceptable cations.
- Examples of such salts include alkali metal or alkaline earth metal salts and, particularly, calcium, magnesium, sodium, lithium, zinc, potassium, and iron salts.
- Compounds included in the present compositions that include a basic or acidic moiety may also form pharmaceutically acceptable salts with various amino acids.
- the compounds of the disclosure may contain both acidic and basic groups; for example, one amino and one carboxylic acid group. In such a case, the compound can exist as an acid addition salt, a zwitterion, or a base salt.
- the compounds of the disclosure may contain one or more chiral centers and/or double bonds and, therefore, exist as stereoisomers, such as geometric isomers, enantiomers or diastereomers.
- stereoisomers when used herein comprises all geometric isomers, enantiomers or diastereomers. These compounds may be designated by the symbols "R” or "5,” depending on the configuration of substituents around the stereogenic carbon atom.
- Stereoisomers include enantiomers and diastereomers. Mixtures of enantiomers or diastereomers may be designated "( ⁇ )" in nomenclature, but the skilled artisan will recognize that a structure may denote a chiral center implicitly. [00152]
- the compounds of the disclosure may contain one or more chiral centers and/or double bonds and, therefore, exist as geometric isomers, enantiomers or diastereomers.
- the enantiomers and diastereomers may be designated by the symbols "(+),” "(-).” “R” or “5,” depending on the configuration of substituents around the stereogenic carbon atom, but the skilled artisan will recognize that a structure may denote a chiral center implicitly.
- Geometric isomers resulting from the arrangement of substituents around a carbon-carbon double bond or arrangement of substituents around a cycloalkyl or heterocyclic ring, can also exist in the compounds.
- the symbol denotes a bond that may be a single, double or triple bond as described herein.
- Substituents around a carbon-carbon double bond are designated as being in the "Z” or "is” configuration wherein the terms "Z" and "is” are used in accordance with
- trans represents substituents on the same side of the plane of the ring and the term “trans” represents substituents on opposite sides of the plane of the ring. Mixtures of compounds wherein the substituents are disposed on both the same and opposite sides of plane of the ring are designated "cis/trans.”
- stereoisomers when used herein comprises all geometric isomers, enantiomers or diastereomers. Various stereoisomers of these compounds and mixtures thereof are encompassed by this disclosure.
- Individual enantiomers and diastereomers of the compounds can be prepared synthetically from commercially available starting materials that contain asymmetric or stereogenic centers, or by preparation of racemic mixtures followed by resolution methods well known to those of ordinary skill in the art. These methods of resolution are exemplified by (1) attachment of a mixture of enantiomers to a chiral auxiliary, separation of the resulting mixture of diastereomers by recrystallization or chromatography and liberation of the optically pure product from the auxiliary, (2) salt formation employing an optically active resolving agent, (3) direct separation of the mixture of optical enantiomers on chiral liquid chromatographic columns or (4) kinetic resolution using stereoselective chemical or enzymatic reagents.
- Racemic mixtures can also be resolved into their component enantiomers by well known methods, such as chiral-phase gas chromatography or crystallizing the compound in a chiral solvent.
- Stereoselective syntheses a chemical or enzymatic reaction in which a single reactant forms an unequal mixture of stereoisomers during the creation of a new stereocenter or during the transformation of a pre-existing one, are well known in the art.
- Stereoselective syntheses encompass both enantio- and diastereoselective transformations. For examples, see Carreira and Kvaerno, Classics in Stereoselective Synthesis, Wiley-VCH: Weinheim, 2009.
- the compounds disclosed herein can exist in solvated as well as unsolvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like.
- the compound is amorphous.
- the compound is a polymorph.
- the compound is in a crystalline form.
- isotopically labeled compounds which are identical to those recited herein, except that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature.
- isotopes that can be incorporated into the compounds include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine and chlorine, such as 10 B, 2 H, 3 H, 13 C, 14 C, 15 N, 18 0, 17 0, 31 P, 32 P, 35 S, 18 F, and 36 C1, respectively.
- a compound may have one or more H atom replaced with deuterium.
- isotopically-labeled disclosed compounds are useful in compound and/or substrate tissue distribution assays. Tritiated (i.e., 3 H) and carbon-14 (i.e., 14 C) isotopes are particularly preferred for their ease of preparation and detectability. Further, substitution with heavier isotopes such as deuterium (i.e., 2 H) may afford certain therapeutic advantages resulting from greater metabolic stability (e.g., increased in vivo half-life or reduced dosage requirements) and hence may be preferred in some circumstances.
- Isotopically labeled compounds can generally be prepared by following procedures analogous to those disclosed in the Examples herein by substituting an isotopically labeled reagent for a non-isotopically labeled reagent.
- prodrug refers to compounds that are transformed in vivo to yield a disclosed compound or a pharmaceutically acceptable salt, hydrate or solvate of the compound. The transformation may occur by various mechanisms (such as by esterase, amidase, phosphatase, oxidative and or reductive metabolism) in various locations (such as in the intestinal lumen or upon transit of the intestine, blood, or liver). Prodrugs are well known in the art (for example, see Rautio, Kumpulainen, et al, Nature Reviews Drug Discovery 2008, 7, 255).
- a prodrug can comprise an ester formed by the replacement of the hydrogen atom of the acid group with a group such as (Ci_8)alkyl, (C2-i2)alkanoyloxymethyl, l-(alkanoyloxy)ethyl having from 4 to 9 carbon atoms, 1 -methyl- l-(alkanoyloxy)-ethyl having from 5 to 10 carbon atoms, alkoxycarbonyloxymethyl having from 3 to 6 carbon atoms, l-(alkoxycarbonyloxy)ethyl having from 4 to 7 carbon atoms, 1 -methyl- l-(alkoxycarbonyloxy)ethyl having from 5 to 8 carbon atoms, N- (alkoxycarbonyl)aminomethyl having from 3 to 9 carbon atoms,
- di-N,N-(Ci-C2)alkylamino(C2-C 3 )alkyl such as ⁇ -dimethylaminoethyl
- carbamoyl-(Ci-C2)alkyl N,N-di(Ci-C2)alkylcarbamoyl-(Ci-C2)alkyl
- a prodrug can be formed by the replacement of the hydrogen atom of the alcohol group with a group such as (Ci_ 6)alkanoyloxymethyl, l-((Ci-6)alkanoyloxy)ethyl, 1 -methyl- l-((Ci-6)alkanoyloxy)ethyl (Ci- 6 )alkoxycarbonyloxymethyl, N-(Ci_6)alkoxycarbonylaminomethyl, succinoyl, (Ci_6)alkanoyl, a- amino(Ci_4)alkanoyl, arylacyl and a-aminoacyl, or ⁇ -aminoacyl-a-aminoacyl, where each a- aminoacyl group is independently selected from the naturally occurring L-amino acids, P(0)(OH) 2 , -P(0)(0(Ci-C6)alkyl) 2 or glycosyl (
- a prodrug can be formed, for example, by creation of an amide or carbamate, an N-acyloxyalkyl derivative, an
- oxodioxolenyl (oxodioxolenyl)methyl derivative, an N-Mannich base, imine, or enamine.
- a secondary amine can be metabolically cleaved to generate a bioactive primary amine, or a tertiary amine can be metabolically cleaved to generate a bioactive primary or secondary amine.
- a starting material or intermediate used in the synthesis of a contemplated compound may have an enantiomeric excess greater than 0, e.g., greater than about 95%, greater than about 98%, greater than about 99%, or essentially 100%.
- a starting material or intermediate may be essentially stereoisomerically pure.
- partial or complete loss of chiral integrity may occur during the synthesis of the contemplated compound thereby reducing or eliminating the enantiomeric excess.
- a stereoisomerically pure starting material or intermediate is used in a synthesis of a contemplated compound
- partial or complete loss of chiral integrity results in a stereoisomeric mixture.
- a stereoisomeric mixture may be partially or essentially completely resolved by subjecting the stereoisomeric mixture to a chiral purification technique (e.g., chiral HPLC purification).
- reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 3% methanol in DCM to obtain 1.20 g, 73% yield of the title compound as light yellow sticky solid.
- reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-3% methanol in DCM to give 320 mg, 75% yield of the title compound as colorless thick oil.
- reaction mixture was partitioned between ethyl acetate (10 mL) and water (5 mL) and separated.
- the aqueous layer was extracted with ethyl acetate (3 X 10 mL) and the combined organic layers were dried over anhydrous Na 2 S0 4 , filtered and concentrated in vacuo resulting in a crude compound which was purified by preparative HPLC to give 20 mg, 12% yield of the title compound as an off white solid.
- reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-3% methanol in DCM to obtain 1.0 g, 70% yield of the title compound as light yellow sticky solid.
- reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in the crude compound which was purified by chromatography on silica gel eluting with 5-10 % methanolic ammonia in DCM and repurified by preparative TLC (10% MeOH in DCM) to give 25 mg, 7% yield, of the title compound as light brown solid.
- K562 purchased from ATCC: CCL-243 or BaF3 cells expressing either wild type BCR-Abl or mutant BCR-Abl (T3151) (obtained from Dr James Griffin Lab, Dana-Faber cancer Institute, Boston MA) were plated into 96 well plates (10,000 cells/well). Serial dilutions of compound starting at 10 ⁇ were added to the cells and the plates incubated at 37 °C for 72 hours at which point CellTitreGlo Reagent (Promega) was added to each plate and luminescence measured. Cell viability was determined relative to DMSO treated cells and GI50 values obtained after plotting dose-dependent effects using PRISM.
- Group A GI50 range of 0.1-30 nM
- Group B GI50 range of 30-100 nM
- Group C GI50 range of 100 nM - 30 ⁇ .
- Example 9 ABL1 (v/t) Omnia kinase assay
- kinase domain from wild type human Abll kinases, Ala46-Val515, were purchased from Cayman Chemical (Ann Arbor, MI), and were purified in the absence of detergent to allow GNF5 and analogues to bind in the myristoyl binding site.
- Abl kinase activity was measured in a semi-continuous fluorescence-based kinase assay (Omnia) utilizing a synthetic peptide Y6 which was purchased from Life Technologies (Carlsbad, CA).
- Assay was run in the presence of 20 mM HEPES pH 8.0, 50 mM NaCl, 10 mM MgC12, .05% BSA, 10 ⁇ sodium vanadate, 200 ⁇ DTT, 2% DMSO, and 10 ⁇ Y6 peptide at protein concentrations of 25 nM for wild type Abll and 6 nM for the T3151 mutant Abll .
- the assay was performed at the ATP Km, which was determined to be 40 ⁇ for the wild type Abll and 10 ⁇ for the T3151 mutant Abl 1.
- Abl 1 inhibitors were titrated in 100% DMSO and then diluted ten-fold into reaction buffer, which was followed by a subsequent ten-fold dilution into the assay plate.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Described herein are compounds capable of modulating one or more biomolecules substantially simultaneously, e.g., modulating two or more binding sites on a protein, e.g., Bcr-AbI tyrosine kinase, as well as methods for the treatment of diseases related to said modulation of a protein including various cancers, and further including drug resistant cancers.
Description
BIVALENT BCR-ABL TYROSINE KINASE LIGANDS, AND METHODS OF USING
SAME
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to and the benefit of U.S. Provisional Patent
Application No. 61/926,466, filed January 13, 2014, entitled, "Bcr-Abl Tyrosine Kinase Ligands Capable of Dimerizing in an Aqueous Solution, and Methods of Using Same," and U.S. Provisional Patent Application No. 61/926,470, filed January 13, 2014, entitled, "Bivalent Bcr-Abl Tyrosine Kinase Ligands, and Methods of Using Same," the entirety of each of which is incorporated by reference herein.
BACKGROUND
[0002] Bcr-Abl tyrosine kinase arises as a result of fusion between break point cluster
(Bcr) gene at human chromosome 22 and Abelson (Abl) tyrosine kinase gene at human chromosome 9 and has been implicated as a cause of chronic myelogenous leukemia. Several inhibitors of Bcr-Abl tyrosine kinase have been developed for the treatment of chronic myelogenous leukemia, for example imatinib, nilotinib, dasatinib, bosutinib, ponatinib, and bafetinib. Structurally, the kinase domain of Bcr-Abl tyrosine kinase contains at least two binding pockets, i.e., a myristoyl binding pocket and an ATP binding pocket, and inhibitors of Bcr-Abl tyrosine kinase generally bind to the ATP binding pocket. The success of Bcr-Abl tyrosine kinase inhibitors in treating cancer (e.g., chronic myelogenous leukemia) has been limited by the emergence of resistant Bcr-Abl tyrosine kinase mutants. Further, recent clinical compounds, such as ponatinib, which target the mutant and wild-type forms of Bcr-Abl, also show more severe side-effects at efficacious doses, suggesting off-target activities which may be hard to remove with traditional medicinal chemistry approaches. Accordingly, there is a need to develop new Bcr-Abl tyrosine kinase inhibitors which can successfully target both wild-type and mutant forms of Bcr-Abl, and preferably prevent the emergence of resistance mutations.
[0003] Current standard drug design and drug therapy approaches typically focus on modulating one protein binding site with limited selectivity and do not address the urgent need
to find drugs that are capable of modulating tandem binding sites substantially simultaneously in order to further improve on specificity and potency. Although antibodies and other biological therapeutic agents may have sufficient specificity to distinguish among closely related protein surfaces, factors such as their high molecular weight prevent oral administration and cellular uptake of the antibodies. Conversely, orally active pharmaceuticals are generally too small to effectively overcome mutational resistance mechanisms while retaining specificity.
SUMMARY
[0004] Described herein, for example, are compounds capable of modulating one or more biomolecules substantially simultaneously, e.g., modulate two or more binding domains on a protein (e.g., Bcr-Abl tyrosine kinase).
or a pharmaceutically acceptable salt, stereoisomer, metabolite, or hydrate thereof is provided; wherein:
Q1 is a connecting moiety covalently bound to P1 and P2, wherein Q1 is selected from the group consisting of -Si(R7)(R8)-, -Si(R7)(R8)-0-, -0-Si(R7)(R8)-, -NR'-, -N(R' )C(0)-,■■ C(0)N(R' )-, -N( R" )SO >-. -SO N; R " ;-. -0-, -C(O)-, - GO O K -C(OK -S-, -SO-, -S02-, - C(=S)-, -C(== R')-5 aliphatic, heteroaliphatic, phenyl, naphthyl, heterocyclyl; heteroaryl, or a covalently bonded combination thereof; wherein P1 and P2 are as defined below.
[0006] In another aspect, a method of treating a disease associated with Bcr-Abl tyrosine kinase in a patient in need thereof is provided. The method comprises administering to the patient the bivalent compound as described above.
BRIEF DESCRIPTION OF THE DRAWINGS
[0007] FIG. 1 shows a screenshot of a protein X-ray crystal structure in which the structures of imatinib and dasatinib are overlaid, according to an embodiment.
[0008] FIG. 2 shows a non-limiting set of pharmacophores (i.e., ligands) with preferred attachment points for connecting the pharmacophores to connecting moieties indicated by arrows, according to an embodiment.
DETAILED DESCRIPTION
[0009] Described herein are compounds capable of modulating one or more biomolecules and, in some cases, modulating two or more binding sites on a protein (e.g., Bcr- Abl tyrosine kinase).
[0010] Advantageously, the bivalent compound may be capable of interacting with a relatively large target site as compared to the individual ligands that form the bivalent compound. For example, a target may comprise, in some embodiments, two protein binding sites separated by a distance such that a bivalent compound, but not an individual ligand moiety, may be capable of binding to both binding sites essentially simultaneously. In some embodiments, contemplated bivlalent compounds may bind to a target with greater affinity as compared to an individual ligand moiety binding affinity alone. Also contemplated herein, in some embodiments, is a bivalent compound that, e.g., may be capable of modulating proximal binding sites on the same target. For example, in one embodiment, the proximal binding sites may be the myristoyl binding pocket and ATP binding pocket of Bcr-Abl tyrosine kinase.
[0011] In an exemplary embodiment, disclosed bivalent compounds may bind to a first Bcr-Abl tyrosine kinase binding site and a second Bcr-Abl tyrosine kinase binding site. The bivalent compound may be used for a variety of purposes. For example, in some instances, the bivalent compound may be used to perturb a biological system. As described in more detail below, in some embodiments, the bivalent compound may bind to or modulate a target biomolecule, such as a protein (e.g., Bcr-Abl tyrosine kinase). In certain embodiments, a contemplated bivalent compound may be used as a pharmaceutical.
[0012] In some cases, a bivalent compound may bind to a target biomolecule with a dissociation constant of less than 1 mM, in some embodiments less than 500 microM, in some embodiments less than 300 microM, in some embodiments less than 100 microM, in some embodiments less than 10 microM, in some embodiments less than 1 microM, in some embodiments less than 100 nM, in some embodiments less than 10 nM, and in some embodiments less than 1 nM, in some embodiments less than 1 pM, in some embodiments less than 1 fM, in some embodiments less than 1 aM, and in some embodiments less than 1 zM.
Bivalent Compounds
and pharmaceutically acceptable salts, stereoisomers, metabolites, and hydrates thereof, wherein:
P1 is a first ligand capable of modulating a first Bcr-Abl tyrosine kinase binding site; P2 is a second ligand capable of modulating a second Bcr-Abl tyrosine kinase binding site; and
Q1 is a connecting moiety covalently bound to P1 and P2 that comprises between 3 and 30 atoms, where the atoms are connected to form a cyclic or acyclic, substituted or
unsubstituted, branched or unbranched aliphatic moiety; cyclic or acyclic, substituted or unsubstituted, branched or unbranched heteroaliphatic moiety; substituted or unsubstituted phenyl or naphthyl moiety; substituted or unsubstituted heteroaryl moiety; or a combination thereof.
[0014] In some embodiments, the ligand may be a pharmacophore. A pharmacophore is typically an arrangement of the substituents of a moiety that confers biochemical or pharmacological effects (e.g., by targeting a Bcr-Abl tyrosine kinase). In some embodiments, identification of a pharmacophore may be facilitated by knowing the structure of the ligand in association with a target biomolecule. In some cases, pharmacophores may be moieties derived from molecules previously known to bind to target biomolecules (e.g., proteins), fragments identified, for example, through NMR or crystallographic screening efforts, molecules that have been discovered to bind to target proteins after performing high- throughput screening of natural products libraries, previously synthesized commercial or non-commercial combinatorial compound libraries, or molecules that are discovered to bind to target proteins by screening of newly synthesized combinatorial libraries. Since most pre-existing combinatorial libraries are limited in the structural space and diversity that they encompass, newly synthesized combinatorial libraries may include molecules that are based on a variety of scaffolds.
[0015] In one embodiment, a bivalent compound may include a first pharmacophore that may bind to the myristoyl binding pocket of Bcr-Abl tyrosine kinase and a second pharmacophore that may bind to the ATP binding pocket of Bcr-Abl tyrosine kinase. A person skilled in the art may appreciate that additional pharmacophores may be discovered in the future and that the pharmacophores illustrated herein are not intended to limit in any way the claims.
[0016] In some embodiments, a ligand (e.g., a pharmacophore) may have one or more preferred attachment points for connecting the pharmacophore to the linker (e.g., with or without a connector moiety). In certain embodiments, an attachment point on a pharmacophore may be chosen so as to preserve at least some ability of the pharmacophore to bind to a binding site. In one embodiment, preferred attachment points may be identified using X-ray crystallography. The following description of a non-limiting exemplary method illustrates how a preferred attachment point may be identified. For example, as shown in FIG. 1, using the 3K5V structure 100 from the protein databank (PDB), a small molecule 1 10 (dark gray) labeled "STI2" in the PDB file [also known as Imatinib or Gleevec or STI-571] may be identified. The Imatinib pyridyl ring (indicated by black circle 120) contains a nitrogen atom in the 3 position, which constitutes an adenine ring mimetic. The corresponding adenine ring mimetic in the new pharmacophore 140 (light gray) should be aligned to this element. The final conformation and orientation of the newly aligned pharmacophore 140 in the site may be determined using a variety of approaches known to computational chemists, but can be done as simply as performing an energy minimization using a molecular mechanics forcefield. It should be noted that the alphanumeric identifiers in FIG. 1 (e.g., F401, T334, M337, etc.) correspond to amino acid residues in the 3K5V structure, where the letter of the identifier is the one-letter amino acid symbol and the number of the identifier is the position of the amino acid residue in the primary sequence of the protein. Attachment points such as the one labelled 150 on the aligned pharmacophore permit access to solvent exposed amino acid residues on the outer lip of the
ATP-binding pocket such as T338 and L389 in the 3K5V structure and are considered preferred attachment points for linkers. It should be apparent to those skilled in the art that overlays of the imatinib pharmacophore with other alternate pharmacophores can be used to identify potential attachment points.
[0017] FIG. 2 provides a non-limiting set of pharmacophores (i.e., ligands) showing preferred attachment points (indicated by arrows) for connecting the pharmacophore to a connecting moiety. It will be appreciated that the ligands disclosed herein can be attached at any open site to a connector moiety as described herein. Such embodiments described below include specific references to each attachment site.
[0018] Exemplary binding site ligands include Bcr-Abl tyrosine kinase ATP binding site ligands represented by the formula:
w herein:
Q is:
(1) a 5-membered heteroaryi ring;
(2) a 6-membered heteroaryi ring; or (3) an aryi ring; optionally subsiituted with one or more groups R1;
U is:
(1) a single bond;
(2) R150=CH ; or
(3)— (CH2)m— , where m is 1 to 2;
T1 and T" are each hydrogen, or together form =0 or =S;
R: is:
(1) hydrogen or R6, where R6is alkyl, alkenyl, aikynyl, cycloalkyl, cycloalkylalkyl, cycloalkenyl, cycloalkenyl alkyl, aryi, aralkyl, heterocyclo, or heterocyeioalkyl, each of which is unsubstituted or substituted with U1, L' and one or more groups UJ;
(2)— OH or—OR6;
(3) --SHor--SR6:
(4) C(0)2H, C(0)qR6, or O C(0)qR6;
(5)— S03H or— S(0)qR6;
(6) halo;
(7) cyano;
(8 ) nitro;
(9)— U4— NR7R8;
( 10) t.'' MR") I " \R! °R! ; ;
(11) -ΙΓ - (Ri2) U5 R6;
(12)— P(0)(OR6)2;
R2 is:
(1) R6, substituted with Q1;
(2)— U4— R6, substituted with Q1; or
(3)— U13— R7R8, substituted with Q1;
R3is:
(1) hydrogen or R6;
(2) -U4 R6; or
(3)— Ui— NR7R8;
R4andR5:
(1) are each independently hydrogen or R6;
(2) i.; \iR") I.' \R:,!R:::
(3)— N(R9)U4R6; or
(4) together with the nitrogen atom to which they are attached complete a.3- to
8-membered saturated or unsaturated heterocyclic ring which is unsubstituted or substituted with U1, U2 and !r , which heterocyclic ring may optionally have fused to it a benzene ring itself unsubstituted or substituted with U\ U and Ό '; R7,R8, R9, R10, Rn, andR12:
(1 ) are each independe tly hydrogen or R6;
(2) R ' and R8 may together be alkylene, alkenylene or heteroalkyl, completing a 3- to 8-membered saturated or unsaturated ring with the nitrogen atom to which they are attached, which ring is unsubsiituted or substituted with U\ U2 and U3; or
(3) any two of R9, R10, and Rn may together be alkylene or alkenylene completing a 3- to 8-membered saturated or unsaturated ring together with the nitrogen atoms to which they are attached, which ring is unsubstituted or substituted with U1, Uz and UJ;
Ri3is:
(l)cyano;
(2) nitro;
(3)— N¾;
(4) NBOaikyl;
(5)— OFT;
(6) \UOaryi:
(7) -- HCOOalkyl;
(8) - HCOOaryl;
(9)— NHS02alkyl;
(10)— NHS02aiyl;
(1 1) aryl;
(12) heteroaryl;
(13) Oalkyl; or
(14)— Oaryl;
R14 is:
(1 ) NO2;
(2)— COOalkyl; or
(3)— COOaryl;
Ri 5 is:
( 1) hydrogen;
(2) alkyl;
(3) aryl;
(4) aryialkyl: or
(5) cycloalkyl;
ϋ', U2 and U3 are each independently:
(1) hydrogen or U6, where LI6 is (i) alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, cycloalkenyl, eycloalkenylaikyl, aryl, aralkyi, alkylaryl, cycloalkylaryl, heterocyclo, or heterocycloalkyl; (ii) a group (i) which is itself substituted by one or more of the same or different groups (i); or (iii) a group (i) or (ii) which is substituted by one or more of the following groups (2) to (16) of the definition of U1, U2 and U3;
(2)— OH or— OU6;
(3) --SH or --SU6;
(4) Ci O y i . C(0)qU*, or O ( ί ί) )Μί
(5)— S03H,— S(0) U6; or S(0)qN(U9)U6;
(6) h lo;
(7) cyano;
(8 ) nitro;
(9)— U4— N(U7)U8;
(10)— U— N(UV- U5— N(U7)U8;
( 1 1) -ΙΓ -N(U10) U5 U6;
(12)— U4— N(U10)— U5— H;
(13) oxo;
(14)— O— C(O)— U6;
(15) any two of U1, Uz, and U3 may together be alkylene or alkenylene completing a 3- to 8-membered saturated or unsaturated ring together with the atoms to which they are attached; or
(16) any two of U1 , U", and U3 may together be— O— (CH2 V----G— , where r is 1 to 5, completing a 4- to 8-membered saturated or unsaturated ring together with the atoms to which they are attached;
1. 1 arid U5 are each independently:
(1) a single bond;
(2) -U! ! S(0)<f U12 ;
(3)— U11— C(O)— U12— ;
(4) ....... Un--C(S)--U12--;
(5) {. " O -U 12 ;
(6)— U11— S— U12— ;
(7) I . " O Ci O ) I . 1 ' : or
(g) (·; () > () I . 1 2 --;
U7, U8, U9 and U10:
(1) are each independently hydrogen or U6;
(2) Ό ' and U8, or U°and U10, may together be alkylene or alkenylene, completing a 3- to 8-membered saturated or unsaturated ring together with the atoms to which they are attached, which ring is un substituted or substituted with U1, U a d LP; or
(3) U? or U8, together with U9, may be alkylene or alkenylene completing a. 3- to 8-membered saturated or unsaturated ring together with the nitrogen atoms to which they are attached, which ring is unsubstituted or substituted with U1, U2 and U ;
U 11 and U 12 are each independently:
(! ) a single bond;
(2) alkylene;
(3) alkenylene; or
(4) alkynylene; and
(ί ) a single bond;
(2)— \ju— S(0)q—Ui2—;
s 3 ) Ci () > l ' :
(4) -U! ! C(S) -U12 ;
(5)— U11— O— U12— ;
(6) .--Un---S--U12--;
(7) U11 0 C(O) U12 ;
(8)— U11— C(O)— O— U12— ;
(9)— C(NR13)— ;
( 10) --C(CHR14)--; o
( 1 1) C(C(Ri4)2) ; and
q is 1 or 2;
or a pharmaceutically acceptable salt thereof,
[0019] For example, a Bcr-Abl tyrosine kinase ligand may be represented by:
[0020] A person of skill in the art will appreciate that certain substituents may, in some embodiments, result in compounds that may have some instability and hence would be less preferred,
[0021] In certain embodiments, a Bcr-Abl tyrosine kinase ATP binding site ligand may be represented by imatinib, nilotinib, bosutinib, ponatinib, DCC-2036, GNF5, or bafetinib.
[0022] Exemplary Bcr-Abl tyrosine kinase myristoyl binding site ligands include those represented by the formula:
wherein:
U1 and U2 are independently selected from the group consisting of -N= and -CR4=, wherein R4 is hydrogen or Ci^alkyl;
L Is selected from the group consisting of a bond, -O- and -NR"1 -, wherein R5 is hydrogen or Ci alkyl;
R! is selected from the group consisting of -U ' R6R', -UOR' and -U3R' , wherein U3 is a bond or Ci _4alkylene, R6 is hydrogen or Chalky 1 and R7 is selected from the group consisting of Ce-ioaryl and Cs-eheteroaryl; wherein any aiyl or heteroaryl is optionally substituted with i to 3 radicals independently selected from the group consisting of halo, amin halo - substituted Cj_4alkyl,
R is selected f
rom the group consisting ofhydrogen, halo, amino, alo- substituted Ci..4aikyl, Ci.4aikoxy and halo-substituted Cj .4alkoxy;
R3 is selected from the group consisting of Cs-gheterocycloalkyl-Co^alkyl, CV ioheteroaryl-Co-4alkyl and Ce-i oary l-Co^alkyl; wherein any alkyl group is optionally substituted with 1 to 3 radicals selected from the group consisting of hydroxy, halo and amino; and any aryi, heteroaryl or heterocycloalkyl is optionally substituted with 1 to 3 radicals independently selected from the group consisting of halo, nitro, Chalky 1, halo-substituted
hydroxy - Chalky],
halo-substituted phenyl, Cs-sheterocycloalkyl, -
U3C(0) R8R8, -U3C(0)NR8R9, -U3C(0)R9, -U3S(0)NR8R8, -U3NR8R9, -U3 R8R8, - U3S(0)2NR8R8, -U3S(0)2R8, -U3S(0)2R9,
-U3S R8R8, -U3ONR8R8, -U3C(0)R8, -U3NR8C(0)R8, -U3NR8S(0)2R8, -U3S{0)2NRsR9, - U3NR8S(0)2R9, -U3NR8C(0)R9, -U3 R8C(0) R8R9, -U3NR8C(0) R8R8, -U C(0)OR8, =NOR8, -U3 R8OR8, -U3NR8(CH2)i.4NR8R8, -U C(0) R8(CH2)i.4NR8R8, -U3C(0)NR8(CH2)i- 4R9, · ;. · !( ·ί () ;Ν Κ ' ( ( ·ϋ . !; ,Οκ9. -U30(CH2)i-4 R8R8, -U3C(0)NR8(CH2),„4GRs and■■
U3NR8(CI¾)i-4R9; wherein phenyl can be further substituted by a radical selected from -NR8RS or -C(0) RbR8; UJ is as described above; R8 is hydrogen, Chalky!, hydroxy-Ci ^alkyl or C2.. 6aikenyl; and R9 is hydroxy, C6-ioaryl--Co-4aikyi, C6-ioaryl-Co-4aikyloxy, CYioheteroaryl-Co- 4aikyl, Cs-sheterocycloalkyl-Co^alkyl or Cs-scycloalkyl; wherein said aryl, heteroaryl, cycloalkyl, heterocycloalkyl or alkyl of R9 is further optionally substituted by up to 2 radicals selected from the group consisting of halo, hydroxy, cyano, amino, nitro, C i.4alkyl, hydroxy-Ct. fialkyi, halo-substituted Chalky 1, Ci-4alkoxy, halo-substituted Ci-4alkoxy, halo-alkyl- substituted-phenyl, benzoxy, Cs.gheteroaryL Cs-gheterocycloalkyl, -C(0)NR°R°, -S(0)2NR8R8, - NR8R8, -C(O)Ri0 and
-NRuRn, wherein Ri0 is Cs-eheteroaryl and Ru is hydroxy-Ci-4alkyl; wherein R3 is substituted with Q1; and
the N-oxide derivatives, prodrug derivatives, protected derivatives, individual isomers and mixture of isomers thereof; and the pharmaceutically acceptable salts and solvates of such compounds.
For example, a Bcr-Abl tyrosine kinase ligand may be represented by:
wherein:
R1 is pyrazolyl; wherein said pyrazolyl is unsubstituted or substituted with 1 to 2 R° groups;
R;2 is pyrroiidinyl: wherein said pyrrolidinyl is substituted with one R' group;
RJ is selected from hydrogen and halo;
R4 is selected from »SF5 and -Y2-CF2-Y3;
R6 at each occurrence is independently selected from the group consisting of hydrogen, hydroxy, methyl, methoxy, cyano, trifiuoromethyl, hydroxy-methyl, halo, amino, fluoro-ethyl, ethyl and cyclopropyl;
R' is selected from hydroxy, methyl, halo, methoxy, hydroxy-methyl, amino, methyl- amino, amino-methyl, trifiuoromethyl, 2-hydroxypropan-2-yl, methyl-carbonyl-amino, dimethyl-amino, 2-amino-3-methylbiitanoyl)oxy, carboxy, methoxy-carbonyl, phosphonooxy, cyano, and amino-carbonyi;
Y is selected from CH and N;
Y1 is selected from CH and N;
Y2 is selected from CF2, O and S(0)o-2; and
Y ' is selected from hydrogen, chioro, fiuoro, methyl, difluoromethyl, and
trifiuoromethyl; or pharmaceutically acceptable salts thereof;
[0025] -Abl tyrosine kinase ligand may be represented by:
Exemplary Bcr-Abl tyrosine kinase myristoyl binding site ligands include those
wherein:
Y at each occurrence is independently selected from N and CH;
Y1 is selected from and CR5; wherein R"1 is selected from hydrogen, methoxy and imidazolyl; wherein said imidazolyl is unsubstituted or substituted with methyl;
R1 is selected from pyrazoiyl, thiazoiyl, pyrrolyl, imidazolyl, isoxazolyl, furanyl and thienyl; wherein said thiazoiyl, pyrrolyl, imidazolyl, isoxazolyl, furanyl, or thienyl of R1 is unsubstituted or substituted with 1 to 3 R6 groups;
R1 is selected from pyrrolidinyl, piperidinyl, azetidinyl, morpholino, piperazinyl, 2-oxa- 6-azaspiro[3.4j-ocianyl, 3-azabicyc3o[3.1.0]hexan-3-yl, pyrrolo[3,4-c]pyrazo3-5(lH,4H,6H)-yL hexahydropyrrolo[3,4-c]pyrrolyl, 6-oxo-2,7-diazaspiro[4.4]-nonanyl, lH-pyrrolo[3,4- cjpyridinyl, 1 ,4-oxazepan-4-yl, 2-oxooxazolidinyl, j ,4-diazepanyl, tetrahydro-2H-pyranyl, 3,6- dihydro-2H-pyranyl, 3,8-dioxa-10-azabicyclo[4.3.1]decanyl, -OR50 and - R"3R5b; wherein said piperidinyl, azetidinyl, morpholino, piperazinyl, l,4-oxazepan-4-yl, pyrrolo[3,4-c]pyrazol- 5(lH,4H,6H)-yl, 2-oxa-6-azaspiro[3.4]-octanyl, 3-azabicyclo[3, 1.0]hexan-3-yl,
hexahydropyrrolo[3,4-c]pyrrolyl, 6-oxo-2,7-diazaspiro[4.4]-nooanyl, lH-pyrrolo[3,4- cjpyridinyl 2-oxooxazolidinyl, 1 ,4-diazepanyl, tetrahydro-2H-pyranyl, 3,6-dihydro-2H- pyranyl, or 3,8-dioxa-10-azabicyclo[4,3.1]decanyi of R? is unsubstituted or substituted with 1 to 3 R ? groups; wherein said pyrrolidinyl of Rz is unsubstituted or substituted with 2 or 3 R ' groups;
R3 is selected from hydrogen and halo;
R4 is selected from -SF5 and -Y2-CF2-Y3;
R33 is selected from hydrogen and Chalky 1;
R3D is selected from
and tetrahydro-2H-pyran-4-yI; wherein said aikyl of R5° is unsubstituted or substituted with 1 to 3 groups independently selected from hydroxy and dimethyl-amino;
R° at each occurrence is independently selected from hydrogen, hydroxy, methyl, hydroxy, methoxy, cyano, trifluoromethyl, hydroxy-methyl, halo, amino, fluoro-ethyl, ethyl, cyclopropyl and dimethyl-amino-carbonyl;
R ' at each occurrence is independently selected from hydroxy, methyl, halo, methoxy, hydroxy-methyl, amino, methyl-amino, amino -methyl, trifluoromethyl, 2-hydroxypropan-2-yl, meihy l-carbonyl-amino, dimethyl-amino, 2-amino-3 -methylbutanoy l)oxy, carboxy, methoxy- carbonyl, phosphonooxy, cyano, and amino-carbonyl; or two R' groups combine with the atom to which they are attached to form a ring selected from cyclopropyl, azetidin-3-yl and 3- azabicycio[3.1.Ojhexan-3-yl;
Y2 is selected from CF2, O and S(0)o-2; and
Y3 is selected from hydrogen, halo, methyl, difluoromethyl and trifluoromethyl; or pharmaceutically acceptable salts thereof
[0027] Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
wherein:
X is cycloaikyl of 3 to 7 carbon atoms, which may be optionally substituted with one or more aikyl of 1 to 6 carbon atom groups; or is a pyridinyl, pyrimidinyl, or phenyl ring; wherein the pyridinyl, pyrimidinyl, or phenyl ring optionally mono- di-, or tri-substituted with a substituent selected from the group consisting of halogen, aikyl of 1 -6 carbon atoms, alkenyl of 2-6 carbon atoms, alkynyl of 2-6 carbon atoms, azido, hydroxyalkyl of 1-6 carbon atoms, halomethyl, aikoxymethyl of 2-7 carbon atoms, alkanoyloxymethyl of 2-7 carbon atoms, alkoxy of 1-6 carbon atoms, alky lth io of 1 -6 carbon atoms, hydroxy, trifluoromethyl, cyano, nitro, carboxy, carboaikoxy of 2-7 carbon atoms, carboalkyl of 2-7 carbon atoms, phenoxy, phenyl, thiophenoxy, benzoyl, benzyl, amino, alkylamino of 1-6 carbon atoms, dialkylamino of
2 to 12 carbon atoms, phenylamino, benzylamino, alkanoylamino of 1-6 carbon atoms, alkenoyfamino of 3-8 carbon atoms, alkynoylamino of 3-8 carbon atoms, and benzoylamino; n is 0 or 1 ;
Y is -NH-, -0-, -S-, or - R-;
R is Ci-ealkyl;
R\ R , R\ and R4 are each, independently, hydrogen, halogen, alkyl of 1-6 carbon atoms, alkenyl of 2-6 carbon atoms, alkynyl of 2-6 carbon atoms, alkenyloxy of 2-6 carbon atoms, alkynyloxy of 2-6 carbon atoms, hydroxymethy], alomethyl, alkanoyloxy of 1-6 carbon atoms, alkenoyloxy of 3-8 carbon atoms, alkynovloxy of 3-8 carbon atoms, alkanoyloxymethyl of 2-7 carbon atoms, alkenoyloxymethyl of 4-9 carbon atoms, alkynoyloxymethyl of 4-9 carbon atoms, alkoxymethyl of 2-7 carbon atoms, alkoxy of 1-6 carbon atoms, alkylthio of 1-6 carbon atoms, alkylsulphinyl of 1 -6 carbon atoms, alkylsulphonyl of 1-6 carbon atoms, alkylsulfonamido of 1 -6 carbon atoms, alkenylsulfonamido of 2-6 carbon atoms,
alkynylsulfonamido of 2-6 carbon atoms, hydroxy, trifiuoromethyl, cyano, nitro, carboxy, carboaikoxy of 2-7 carbon atoms, carboalkyl of 2-7 carbon atoms, phenoxy, phenyl, thiopbenoxy, benzyl, amino, hydroxy amino, aSkoxyamino of 1 -4 carbon atoms, alkylamino of 1-6 carbon atoms, dialkylamino of 2 to 12 carbon atoms, aminoalkyl of 1 -4 carbon atoms, N- alkylaminoalkyl of 2-7 carbon atoms, N, -dialkylaminoalkyl of 3-14 carbon atoms, phenylamino, benzylamino,
R5 is alkyl of 1-6 carbon atoms, alkyl optionally substituted with one or more halogen atoms, phenyl, or phenyl optionally substituted with one or more halogen, alkoxy of 1-6 carbon atoms, trifluoromethy], amino, nitro, cyano, or alkyl of 1 -6 carbon atoms groups;
R6 is hydrogen, alkyl of 1-6 carbon atoms, or alkenyl of 2-6 carbon atoms;
R! is chloro or bromo;
R* is hydrogen, alkyl of 1-6 carbon atoms, aminoaikyl of 1-6 carbon atoms, N- alky!ammoalkyl of 2-9 carbon atoms, Ν,Ν-dialkylaminoalkyl of 3-12 carbon atoms, - cycloalkylaminoalkyl of 4-12 carbon atoms, N-cycloaikyl-N-alkylaminoalkyl of 5- 18 carbon atoms, Ν,Ν-dicycloalkylaminoalkyl of 7-18 carbon atoms, morpholino-N-alkyl wherein the alkyl group is 1-6 carbon atoms, piperidino-N-aikyl wherein the alkyl group is 1-6 carbon atoms, N-alkyl-piperidino-N-alkyl wherein either alkyl group is 1 -6 carbon atoms, azacycloalkyl- -alkyl of 3-11 carbon atoms, hydroxyalkyl of 1-6 carbon atoms, alkoxyalkyl of
2-8 carbon atoms, carboxy, carboalkoxy of 1 -6 carbon atoms, phenyl, carboalkyl of 2-7 carbon atoms, chloro, fluoro, or bromo; Z is amino, hydroxy, alkoxy of 1 -6 carbon atoms, alkylamino wherein the aikyl moiety is of 1-6 carbon atoms, dialkylamino wherein each of the alkyl moieties is of 1-6 carbon atoms, morphoiino, piperazino, N-alkylpiperazino wherein the alkyl moiety is of 1-6 carbon atoms, or pyrrolidine;
m is an integer from 1-4, q is an integer from 1 -3, and p is an integer from 0-3;
any two of the substituents R!, R2, R:', or R4 that are located on contiguous carbon atoms can together be the divalent radical -0-C(R8)2-0-;
or a pharmaceutically acceptable salt thereof.
-Abl tyrosine kinase ligand may be represented by:
[0029] Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
wherein:
Ring T is a 5-membered heteroaryl ring containing 1-2 nitrogens with the remaining ring atoms being carbon, substituted on at least two ring atoms with Rl groups, at least two of which being located on adjacent ring atoms, and, together with the atoms to which they are attached, forming a saturated, partially saturated or unsaturated 5- or 6- membered ring (Ring E), containing 0-3 heteroatoms selected from O, N, and S and being optionally substituted with 1-4 Re groups;
Ring A is a 5- or 6-membered aryl or heteroaryl ring;
Ring B is a 5- or 6-membered aryl or heteroaryl ring;
L1 is selected from N^QO), C^NR1, Ν^Ο(0)0, NR1C(0)NR1, and OC(0)NR1; each occurrence of Ra, Rb and Rl is independently selected from the group consisting of -(CH2),„-(Rmg C), wherein Ring C is a 5- or 6-membered heterocyclic or heteroaryl ring comprising 1 -3 heteroatoms independently selected from O, N and S(0)f; halo; -CN; - O2; -R4; -OR2; -NR2R3; -C(0)YR2; -OC(0)YR2; -NR2C(0)YR2; -SC(0)YR2; -NR2C(=S)YR2; -
OC(=S)YR2; -C(=S)YR2; -YC(=NR3)YR2; -YP(=0)(YR4)(YR4); -Si(R2)3; -NR2S02R2; -S(0)r- R2; -S02NR2R3; and -NR2S02NR2R3, wherein each Y is independently a bond, -0-, -S- or - NR3-;
Re, at each occurrence, is independently selected from the group consisting of halo, =0, -CN, -NO2, -R4, -OR2, -NR2R3, -C(0)YR2, -OC(0)YR2, -NR2C(0)YR2, -SC(0)YR2, -
NR2C(=S)YR2, -OC(=S)YR2, -C(=S)YR2, -YC(=NR3)YR2, -YP(=0)(YR4)(YR4), -Si(R2)3, - NR2S02R2, -S(0)rR2, -S02NR2R3 and -NR2S02NR2R3, wherein each Y is independently a bond, -0-, -S- or -NR3-;
R1, R2, and R3 are independently selected from H, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl;
alternatively, R2 and R3, taken together with the atom to which they are attached, form a 5- or 6- membered saturated, partially saturated or unsaturated ring, which can be optionally substituted and which contains 0-2 heteroatoms selected from N, O and S(0)r-;
each occurrence of R4 is independently selected from alkyl, haloalkyl, trifluoromethyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl; each of the alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl moieties is optionally substituted;
m is 0, 1 , 2, 3 or 4;
n is 2 or 3;
p is 0, 1, 2, 3, 4 or 5; and
r is 0, 1 or 2;
or a pharmaceutically acceptable salt,
[0030] For example, a Bcr-Abl tyrosine kinase ligand may be represented by:
or a pharmaceutically acceptable salt thereof.
[0031] Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
wherein:
R1 represents hydrogen, lower alkyl, lower alkoxy-lower alkyl, or benzyl;
Rz represents phenyl substituted by one or two substituents selected from the group consisting of Chalky!, trifluoro-Ct-salkyL hydroxy-Ci-ealkyl, amino-Ct-ealkyl, Ci-ealkyl- ammo-Ci-ealkyl,
N-cyclo exyl-N-Ci-ealkylamino-Ct-galkyl, Ci- 6alkoxy-C(0)-piperidino-Ci-6alkyl, N-Ci-ealkyl-piperazino-Ci-ealkyl,
6alkyi, hydroxy, Ci-ealkoxy, trifluoro-CVeaikoxy, IH-imidazolyi-Ci-ealkoxy, Ci-6alkyl-C(0)-, benzoyloxy, carboxy, carbamoyl, Ch lky 1-carbamoyi, amino, Ci_6alkyl-C(0)-amino, benzoylarnino, amino mono- or disubstituted by Ci-ealkyl, by hydroxy-Ci-ealkyl or by Q. ealkoxy-Ci-ealkyl, lH-imidazolyl,
carboxy- lH-imidazolyl, Ci-ealkyl- estercarboxy- lH-imidazoiyl, pyrrolidine, piperidino, piperazino, N-(Ci-6aIkyi)piperazinG, morpholino, sulfarnoyl, Ci-ealkyisulfonyl, phenyl, pyridyl, halogenyl, or benzoyl; and
R3 represents hydrogen, d-ealkyl, or halo;
or a -oxide or pharmaceutically acceptable salts thereof.
[0032] For example, a Bcr-Abl tyrosine kinase ligand may be represented by:
, or a pharmaceutically acceptable salt thereof.
0033 Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the formula:
wherein:
R1 is 4-pyrazinyl, 1 -methyl- lH-pyrrolyl, amino- or amino-Cj.ealkyl-substituted phenyl wherein the amino group in each case is free, alkylated or acylated, lH-indolyl or 1H- imidazolyl bonded at a five-membered ring carbon atom, or unsubstituted or C...galkyl- substituted pyridyl bonded at a ring carbon atom and unsubstituted or substituted at the nitrogen atom by oxygen,
R2 and R3 are each independently of the other hydrogen or Chalk ], one or two of the radicals R4, R5, R6, R7 and R8 are each nitro, fluoro-substituted Cj -ealkoxy or a radical represented by:
10
-N(R9)-C(=X)-(Y)n-R'
wherein:
hydrogen or Cj-salkyl,
X is oxo, thio, imino, N-Cj-6alkyl-imino, hydroximino or O-Cj-ealkyl-hydroximino, Y is oxygen or the group NH,
n is 0 or 1 and
R10 is phenyl that is unsubstituted or substituted by halogen, cyano, Ci-6alkoxy, carboxy, Ci-6alkyl, or by 4-methyl-piperazinylmethyl; C5_7alkyl; thienyl; 2-naphthyl;
cyclohexyl; an aliphatic radical having at least 5 carbon atoms; or an aromatic, aromatic- aliphatic, cycloaliphatic, cycloaliphatic-aliphatic, heterocyclic or hetero-cyclicaliphatic radical; and the remaining radicals R4, R5, R6, R7 and R8 are each independently hydrogen, Cj- 6alkyi that is unsubstituted or substituted by free or alkylated amino, piperazinyl, piperidinyl,
pyrrolidinyl or by morpholinyl, or Q.ealkyl-QO)-, trifluoromethyl, free, etherified or esterifed hydroxy, free, alkylated or acylated amino or free or esterified carboxy;
or a pharmaceutically acceptable salt thereof.
wherein:
E1 is phenyl substituted with 1 , 2, or 3 groups independently selected from fluoro and methyl;
A is selected from the group consisting of imidazolyl and pyrazolvl, wherein A is substituted at any suitable position with a 10-membered bicyclic heteroaryl ring optionally substituted with 1 , 2, or 3 R1 groups;
R1 is independently selected from the group consisting of d-ealkyl, branched Cj. alkyf, Cs-scarbocyclyL halogen, fluoroCi-ealkyl wherein the alky! moiety can be partially or fully fluorinated, cyano, hydroxy 1, methoxy, oxo, (R2)? C(0)-, - (R2)C(0)R\ i R ' bXSO *-. - (R2)S02R2, -(CH2)qN(R2)2, -0(CH2)qN(R2)2, -Oi CI l . )„()-< Ί (.aikv!. -\i R * }« 1 I■!,,(>-( Ί <.alkyi. -N{R )(CH2)qN(R )2, -0{CH2)qR2, -N(R )(CH2)qR2, -C(0)R2, and nitro
R2 is H, Cj-ealkyl, branched Cs-yalkyl, Cj-gcarbocyclyl, or phenyl;
t is 1 , 2, or 3;
or a pharmaceutically acceptable salt thereof.
For example, a Bcr-Abl tyrosine kinase ligand may be represented by:
or a pharmaceutically acceptable salt thereof. Exemplary Bcr-Abl tyrosine kinase ligands include those represented by the
wherein:
Ring A is selected from the group consisting of phenylene, pyridinylene, pyrazinylene, pyrimidinylene, pyridazinylene, and triazinylene;
Ring B is selected from the group consisting of pyridyi, pyrimidinyl, pyrazinyl, pyridazinyl, and 1 ,2-dihy dropyridaziny 1, each optionally substituted with 1 , 2, or 3 groups independently selected from Chalky!, halo, and amino;
R! is
wherein RJ is a saturated, nitrogen-containing heterocyclic group substituted by oxo, a saturated, nitrogen-containing heterocyclic group, aminoalkyl, monoalkylaminoalkyl, dialkylaminoaikyl, Ci-ealkoxy, d-ealkyl, alkyoxycarbonyl, halo, haloalkyl, hydroxyalkyl, amino, monoalkylamino, dialkylamino, carbamoyl,
monoalkylcarbamoyl, and dialkyicarbamoyl;
R is selected from Cuealkyl, halogen, haloalkyl, trifiuoromethyl, hydroxyalkyl, d_ ealkoxy, alkoxyalkyl, alkoxycarbonyl, acyl, amino, monoalkylamino, dialkylamino, nitro, carbamoyl, monoalkylcarbamoyl, dialkyicarbamoyl, and cyano;
R3 is selected from hydrogen, C 3 -^alkoxy, and halo;
R4 is selected from hydrogen, Q-ealkyl, and halo; and
n is 1, 2, 3, or 4;
or a pharmaceutically acceptable salt thereof.
[0038] For example, a Bcr-Abl tyrosine kinase ligand may be represented by:
, or a pharmaceutically acceptable salt thereof.
[0039] In certain embodiments, a contemplated bivalent compound may be selected from the group consisting of:
wherein m and n are independently 2, 4, 6, 8, or 10; and pharmaceutically acceptable salts thereof. Connectors (Q1)
[0040] As discussed above, certain compounds contemplated herein comprise a first ligand and a second ligand covalently joined by a connector moiety. In some instances, such connector moieties do not have significant binding or other affinity towards an intended target. However, in certain embodiments, a connector may contribute to the affinity of a ligand moiety to a target.
[0041] In some instances, the connector moiety may be varied to control the spacing between two ligands. For example, in some cases, it may be desirable to adjust the spacing between two ligands so as, for instance, to achieve optimal binding of the bivalent compound to a target. In some cases, the connector moiety may be used to adjust the orientation of the
ligands. In certain embodiments, the spacing and/or orientation the connector moiety relative to the ligand moiety can affect the binding affinity of the ligand moiety (e.g., a pharmacophore) to a target. In some cases, connector moieties with restricted degrees of freedom are preferred to reduce the entropic losses incurred upon the binding of a bivalent compound to its target biomolecule. In some embodiments, connector moieties with restricted degrees of freedom are preferred to promote cellular permeability of the bivalent compound.
[0042] In some embodiments, the connector moiety may be used for modular assembly of ligands. For example, in some instances, a connector moiety may comprise a functional group formed from reaction of a first and second molecule. In some cases, a series of ligand moieties may be provided, where each ligand moiety comprises a common functional group that can participate in a reaction with a compatible functional group on a connector moiety. In some embodiments, the connector moiety may comprise a spacer having a first functional group that forms a bond with a first ligand moiety and a second functional group that forms a bond with a second ligand moiety.
[0043] Contemplated connector moieties may be any acceptable (e.g., pharmaceutically and/or chemically acceptable) bivalent linker. For instance, such connecter moieties may comprise 3 to 30 atoms, 3 to 20 atoms, 3 to 15 atoms, 3 to 10 atoms, 5 to 15 atoms, 10 to 20 atoms, 15 to 25 atoms, 20 to 30 atoms, or 10 to 30 atoms. The atoms may be connected in any suitable arrangement. For example, the atoms may be connected to form a cyclic or acyclic, substituted or unsubstituted, branched or unbranched aliphatic moiety; cyclic or acyclic, substituted or unsubstituted, branched or unbranched heteroaliphatic moiety; substituted or unsubstituted phenyl or naphthyl moiety; substituted or unsubstituted heteroaryl moiety; or a combination thereof. In some instances, a connector moiety may include a substituted or unsubstituted Ci-Cio alkylene, substituted or unsubstituted cycloalkylene, acyl, sulfone, phosphate, ester, carbamate, or amide.
[0044] In some instances, contemplat oieties may include polymeric connectors, such a polyethylene glycol (e.g.,
, where n is 1, 2, 3, 4, 5, 6, 7, 8, 9,
10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20, and X is O, S, NH, or -C(O)-) or other
pharmaceutically acceptable polymers. For example, contemplated connecter moieties may be a covalent bond or a bivalent CMO, Ci-30, Ci-2o, CMO, CKMO, or C20-40, saturated or unsaturated, branched or unbranched, hydrocarbon chain, wherein one, two, or three or four methylene units of the hydrocarbon chain are optionally and independently replaced by -SiR2-, --SiR?-Q~, -O-
SiR2~, cyclopropylene, -NR-, -N(R)C(0)-, -C(O)NfR)-, -N(R)S02-, -S02N(R.)-, -0-, ~C(0)~, - OC(0)-, -0(0)0-, -S-, -SO-, -S02-, -C(=S)-, -C(=NR>, phenyl, or a mono or bicyclic heterocyclic or heteroaryl ring, where R is independently hydrogen or any suitable substiiuent. In certain embodiments, the connector moiety may be substituted with -SiR2- and -SiR2-0-, where -SiR2- and -SiR2-0- are covalently bonded to form -SiR.2-0-SiR2-. In some
embodiments, a connector may be from about 7 atoms to about 13 atoms in length, or about 8 atoms to about 12 atoms, or about 9 atoms to about 1 1 atoms in length. For purposes of counting connector length when a ring is present in the connector group, the ring is counted as three atoms from one end to the other.
[0045] For example, in some embodiments, a connector moiety may be selected from the group consisting of Ci-2oalkylene, wherein one, two, three, or four methylene units of the hydrocarbon chain are optionally and independently replaced by -Si(R')(R8)~, ~Si(R'')(R8)-0-, - 0-Si(R7)(R8)-, cyclopropylene, -NR'-, - (R')C(Q)-, -C(0)N(R')-, -1SI(R')S02-, -S02N(R , - 0-, -C(O k -OC(O)-, -C(0)0-, -S-, -SO-, -SO,-, -C(=S)-, -C(=NR')-, C2-6alkenylene, C2- ealkynylene, phenyl, naphthyl, or a mono or bicyclic heterocyclic or heteroaryl ring; -NR'-Ci_ 15alkyl-NR'-C(0)-; -NR'-(CH2-CH2-0)s-Ci_6alkyl-NR'-C(0)-; -(0-CH2-CH2)s-NR'-C(0)-; - (0-CH2-CH2)s; -S-Co-6alkyl-; -NR'-Ci_6alkyl-; -N(Ci_3alkyl)-Ci_6alkyl-NH-C(0)-; -NH-Ci_ 6alkyl-N(Ci_3alkyl)-C(0)-; -S02-NR'-Co-6alkyl-; -SO2-heterocyclyl-C0-6alkyl-; -heterocyclyl- C(O)-; -heterocyclyl-C0-6alkyl-NR'-C(O)-; -NR'-Co-6alkylene-heterocyclene-C(0)-; -0-Ci_ 6alkylene-C(0)-; -0-C1-15alkylene- R'-C(0)-; -0-C1-15alkylene-C(0)-NR'-; and -O-Ci- 6alkylene-, wherein Ci-6alkylene is optionally substituted by -OH;
wherein:
R7 and R8 are selected, independently for each occurrence, from the group consisting of -OH, Ci-6alkyl, -0-Ci_6alkyl, C2_6alkenyl, C3_6cycloalkyl, -Ci_6alkyl- NRaRb, phenyl and heteroaryl; wherein Ci_6alkyl, C2_6alkenyl, C3_6cycloalkyl, phenyl, and heteroaryl may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, hydroxyl, Ci-6alkyl, and phenyl; or
R7 and R8, together with the silicon to which they are attached, form a 4-7 membered heterocyclic ring, optionally containing one, two, three, or four heteroatoms independently selected from O, S, or N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, and hydroxyl;
R' is, independently for each occurrence, selected from the group consisting of hydrogen and Ci-4alkyl; wherein
may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; and
s is an integer from 1-15.
In certain embodiments, a connector moiety may comprise:
a first group selected from the group consisting of:
; and a second group selected from the group consisting of:
R8 R7 , R8 R7 , R8 R7 , and R8 R7 ; wherein thee first group and the Si of the second group are connected by an oxygen atom;
wherein:
Rw is, independently for each occurrence, absent or selected from the group consisting of -Ci-4alkylene-, -0-Ci-4alkylene-, -N(R )-, -N(R )-Ci-4alkylene-, -0-, - C(0)Ci_4alkylene-, -C(0)-0-Ci_4alkylene-, -C2-6alkenylene-, -C2-6alkynylene-, -C3-
6cycloalkylene-, -phenylene-, and -heteroarylene-; wherein Ci-4alkylene, C2-6alkenylene, C2-6alkynylene, C3_6cycloalkylene, phenylene, and heteroarylene may be optionally substituted by one, two, three, or more substituents independently selected from the group consisting of
Ci-4alkoxy, -C(0)Ci_4alkyl, -C(0)-0-Ci_4alkyl, -C(O)- NRaRb, halogen, cyano, hydroxyl, and phenyl; or Rw and R7, together with the silicon to which they are attached, form a 3-8 membered heterocyclic ring, wherein the 3-8 membered ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, hydroxyl, and Ci-6alkyl;
W3 is, independently for each occurrence, (a) absent; or (b) selected from the group consisting of -Ci-4alkylene-, -0-Ci-4alkylene-, -C(0)-Ci_4alkylene-, -N(R )-Ci_
4alkylene-, -C(0)-0-Ci_4alkylene-, -C2-6alkenylene-, -C2-6alkynylene-, -C3- 6cycloalkylene-, -phenylene-, and -heteroarylene-; wherein Ci-4alkylene, C2-6alkenylene, C2-6alkynylene, C3_6cycloalkylene, phenylene, and heteroarylene are optionally substituted by one, two, three, or more substituents independently selected from the
group consisting of Ci-4alkyl, Ci-4alkoxy, -C(0)Ci_6alkyl, -C(0)-0-Ci_4alkyl, halogen, hydroxyl, nitro, and cyano;
R7 and R8 are selected, independently for each occurrence, from the group consisting of -OH, Ci-6alkyl, -0-Ci_6alkyl, C2-6alkenyl, C3_6cycloalkyl, -Ci_6alkyl- NRaRb, phenyl and heteroaryl; wherein Ci-6alkyl, C2-6alkenyl, C3_6cycloalkyl, phenyl, and heteroaryl may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, hydroxyl, Ci-6alkyl, and phenyl; or
R7 and R8, together with the silicon to which they are attached, form a 4-7 membered heterocyclic ring, optionally containing one, two, three, or four heteroatoms independently selected from O, S, or N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, and hydroxyl;
R', Ra, and Rb are selected, independently for each occurrence, from the group consisting of hydrogen and C1-4alkyl; wherein Ci-4alkyl may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; or
Ra and Rb, together with the nitrogen to which they are attached, may form a 4-7 membered heterocyclic ring, optionally containing an additional heteroatom selected from O, S, and N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo and hydroxyl;
BB, independently for each occurrence, is a 4-7-membered cycloalkyl, heterocyclic, phenyl, naphthyl, or heteroaryl moiety, wherein the cycloalkyl, heterocyclic, phenyl, naphthyl, or heteroaryl moiety is optionally substituted with one, two, three, or more groups represented by RBB; wherein R7, independently for each occurrence, may be optionally bonded to BB; and
each RBB is selected, independently for each occurrence, from the group consisting of hydrogen, halogen, nitro, cyano, hydroxyl, amino, thio, -COOH, - CONHR', substituted or unsubstituted aliphatic, and substituted or unsubstituted
heteroaliphatic; or two R together with the atoms to which they are attached form a fused 5- or 6-membered cycloalkyl or heterocyclic bicyclic ring system.
[0047] For example, in some embodiments, a connector moiety may comprise:
a first group selected from the group consisting of:
a second group selected from the group consisting
group and the Si of the second group are connected by an oxygen atom, and wherein R7 and R8 are as defined above.
[0048] In certain embodiments, R7 and R8 are Ci_6alkyl.
[0049] In certain embodiments, the connector moiety is selected from the group consisting of:
[0050] In some embodiments, a contemplated connector moiety may be an optionally substituted unsaturated heteroaliphatie moiety, wherein the optionaliy substituted unsaturated heteroaliphatie moiety comprises at least one functional group, e.g., one, two. three, or four groups, selected from -SiR2-, -SiR2-0-, -NR-, -N(R)C(0)-, -CfO)N(R)-, -N(R)S02-, -S02N(R)- , -0-, ·-(·; (} ;·-. --OC(O)-, -C(0)0-, -S-, -SO-, -S02-, -C(=S>, and -C(=NR)-, where R is independently hydrogen or any suitable subsiituent. The functional group may be positioned at any suitable position within the optionally substituted unsaturated heteroaliphatie moiety. In some embodiments, the optionally substituted unsaturated heteroaliphatie moiety may comprise 3 to 30 atoms, 3 to 20 atoms, 3 to 15 atoms, 3 to 10 atoms, 5 to 15 atoms, 10 to 20 atoms, 15 to 25 atoms, 20 to 30 atoms, or 10 to 30 atoms.
[0051] The optionally substituted "unsaturated heteroaliphatie moiety comprises at least one unsaturated group, e.g., an alkenylene group or an alkynylene group. For example, in some embodiments, the optionally substituted unsaturated heteroaliphatie moiety may comprise I to 10, 1 to 3, 2 to 4, 3 to 5, 4 to 6, 5 to 7, 6 to 8, 7 to 9, or 8 to 10 unsaturated groups. For instance, the optionally substituted unsaturated heteroaliphatie moiety may contain one, two, three, four, five, six, seven, eight, nine, or ten unsaturated groups. In instances where the optionally substituted unsaturated heteroaliphatie moiety comprises a pl rality of unsaturated groups, the unsaturated groups may be alkenylene groups, alkynylene groups, or a mixture thereof.
[0052] An unsaturated group may be positioned at any suitable position within the optionally substituted unsaturated heteroaliphatie moiety. For example, in some embodiments, an unsaturated group may be positioned adjacent to a ligand moiety (i.e., directly connected by a covalent bond) and/or a functional group. In other embodiments, an unsaturated group and a ligand moiety may be separated by one or more atoms. In one embodiment, the optionaliy substituted unsaturated heteroaliphatie moiety is directly connected to a first ligand moiety by a
ftrst unsaturated group and directly connected to a second ligand moiety by a second unsaturated group.
n is 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20;
R1 and R2 are, independently for each occurrence, selected from the group consisting of H, Ci-6alkyl, Ci-6heteroalkyl, phenyl, or heteroaryl, wherein alkyl, heteroalkyl, phenyl, and heteroaryl are optionally substituted with -OH, -NH2, -SH, -COOH, -C(0)NH2, halo, phenyl, and heteroaryl; or
R1 and R2, or R2 and R2, together with the atoms to which they are attached, form a heterocyclic structure optionally substituted with -OH, -NH2, -SH, -COOH, -C(0)NH2, halo, phenyl, and heteroaryl.
[0054] In some embodiments, a connector may comprise a phenyl, naphthyl, or mono or bicyclic heteroaryl ring, each optionally substituted. For example, a connector may comprise one or more of the following aryl structures:
, where R1 and R2 are the remainder of the connector. A person of skill in the art would recognize that some substitutions may be chemically less stable and hence less preferred.
[0055] In another embodiment, a connector may comprise a triazole ring having the following structure:
, where R and R are the remainder of the connector. For example, a bivalent compound comprising a triazole-containing connector may have the following general structure:
Ligand— Connector. Connector— Linker
N^. ,NH
N . Such triazole-joined compounds may be formed, e.g., as a result of a "click" type reaction (i.e., an azide-alkyne cycloaddition). For example, a
first segment of a connector having a terminal alkyne and a second segment of a connector having a terminal azide may be joined by a "click" reaction to form a single connector joined by a triazole, as shown above. In some embodiments, the first connector and the second connector each are less than or equal to 20 atoms in length, or in some embodiments each are less than or equal to 12 atoms in length.
[0056] In certain embodiments, a triazole-containing connector may be represented by -
W1-(CH2-CH2-0)t-Ci-6alkylene-triazolylene-Ci-6alkylene-0-Ci-6alkylene-. For example, the triazole-containing connector may be represented by:
[0057] In certain embodiments, the optionally substituted unsaturated lieteroaliphati moiety may be:
j Wherein.
RLi is, independently for each occurrence, selected from the groups consisting of hydrogen or any suitable substituent; and
W2 is selected from the group consisting of -SiR2-, -SiR2-0-, -0-SiR2-, -NR-,■■
N(R)C(0)-, ··( '; ( } s\{ R -N(R)S02-, -SO.,N( R ···. -0-, -QO K -0C(0V, -00)0-, -S-, -SO-, - SO2-, -C(=S)-, and -C(=NR)-, wherein R is hydrogen or any suitable substituent.
[0058] In another embodiment, a connecter moiety may maximally span from about 5A to about 50A, in some embodiments about 5A to about 25 A, in some embodiments about 20A to about 50A, and in some embodiments about 6A to about 15A in length. In another embodiment, a connecter moiety may maximally span less than about 30A, in some embodiments less than about 20A, and in some embodiments less than about IOA. In another embodiment, a connecter moiety may maximally span from about 1 A to about 30A, in some embodiments about 1 A to about 20A, in some embodiments about 1 A to about IOA, in some embodiments about 1 A to about 5 A., in some embodiments about 20A to about 30A, in some embodiments about 15A to about 25A, in some embodiments about IOA to about 2()A, and in some embodiments about 5A to about 15.4, in length. For example, a connector moiety may
maximally span about 1 A, about 3 A, about SA, about 7 A, about 9A, about 1 lA, about 13 A, about 15 , about 17A, about 19A, , about 21 A, about 23 A, about 25 A, about 27A, or about 29A.
[0059] In certain embodiments, a connector (i.e., Q1) is selected from the group consisting of:
-W1-(C2-6alkylene-0)s-C2-6alkylene-W1-; -W1-(CH2-CH2-0)s-; and -W^Cusalkylene-
W1-;
wherein:
W1 is, independently for each occurrence, NR.', O, or S;
R' is, independently for each occurrence, selected from the group consisting of hydrogen and C1-4alkyl; wherein C1-4alkyl may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; and
s is an integer from 1-10.
[0060] For example, in some embodiments, a connector is selected from the group consisting of: -S-(C2-6alkylene-0)s-C2-6alkylene-S-; -0-(CH2-CH2-0)s-; and -O-Ci-isalkylene- 0-; wherein s is an integer from 1-10.
[0061] In certain embodiments, the connector is -0-(CH2-CH2-0)s-. In certain other embodiments, the connector is selected from the group consisting of: -S-(CH2-CH2-CH2-0)s- CH2-CH2-CH2-S-; -S-(CH2-CH2-0)s-CH2-CH2-S-; and -0-(CH2)u-0-; wherein u is an integer from 1-15. For example, in some embodiments, u is 4, 5, 6, 7, 8, 9, 10, 1 1, 12, 13, 14, or 15. In some embodiments, s is 2, 3, 4, 5, 6, 7, or 8.
[0062] In certain embodiments, a connector (i.e., Q1) is selected from the group consisting of:
[0063]
-W1-(C3-6alkylene-0)s-Ci_6alkylene-W1-;
-W1- (CH2-CH2-0)s-; and
wherein W1 is, independently for each occurrence, N, O, or S; s is an integer from 1-10; and t is an integer from 3-10. For example, in some embodiments, a connector is selected from the group consisting of: -S-(C3_6alkylene-0)s-Ci_ 6alkylene-S-; -S-(CH2-CH2-0)t-Ci_6alkylene-S-; -0-(CH2-CH2-0)s-; and -0-Ci_i5alkylene-0-; wherein s is an integer from 1-10, and t is an integer from 3-10. In certain embodiments, the
connector is -0-(CH2-CH2-0)s-. In certain other embodiments, the connector is selected from the group consisting of: -S-(CH2-CH2-CH2-0)s-CH2-CH2-CH2-S-; -S-(CH2-CH2-0)t-CH2-CH2- S-; and -0-(CH2)u-0-; wherein u is an integer from 1-15. For example, in some embodiments, u is 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15. In some embodiments, s is 2, 3, 4, 5, 6, 7, or 8.
[0064] The synthetic route in Scheme Xa illustrates a general method for preparing ligand-connector derivatives. The method involves attaching the desired substituents to the carbonyl substituent.
SCHEME Xa
Q H2N-Connector o
Ligand^ - ► Ligand^
OH HN-Connector
1 3
[0065] The desired connector can be installed by reacting the ligand 1 with the appropriate nucleophile 2 to provide 3 (ligand-connector derivative). For example, Scheme Xa provides for a connector (Q1). The desired connector attached at the carbonyl substituent can be installed by reacting carboxylic acid 1 with common coupling reagents such as l-ethyl-3-(3- dimethylaminopropyl)carbodiimide (EDC) and hydroxybenzotriazole (HOBt) and then further reacting the resultant activated ester with the appropriate nucleophile, for example, amine 2, to provide 3 (ligand-connector derivative).
[0066] For example, the connector may be selected from the group consisting of:
VY"
O^ /\ /\ L /\ and \ M^OH
v ^-o^/ \^OH \ J " t where n may be 0, 1, 2, 3,4 or i
In some embodiments, Q1 may generally be represented for example, by:
where n may be 0, 1, 2, 3, 4, 5, or 6.
Additional examples for 2 and -NH-R (e.g., Q1) can be found in Table A, seen
Table A
2 -NH-R
No.
(e.g., -Q1)
[0069] Any free amino group seen in the connector examples of Table A above may be functionalized further to include additional functional groups.
[0070] In another embodiment, the attachment point on the ligand may be further elaborated to incorporate not only a connector moiety, but also a second ligand, as e.g., represented by:
SCHEME Xa'
Q H2N-Connector-Ligand2 o
Ligand^ ► Ligand^
OH HN-Connector-Ligand
1 5
[0071] The Qx-P2 moiety may be formed from direct attachment of Qx-P2 to the carbonyl, or the Qx-P2 moiety may be formed from the further functionalization of any free amino group seen in the -NH-R examples (i.e., Q1 examples) of Table A above to include the second ligand moiety (P2).
[0072] Other examples of Qx-P2 may be represented by the structure:
H n , wherein n is 0, 1, 2, 3, 4, or 5, e.g. n is 1 to 5.
Ligand-electrophile N ■or ^r~^ -NH, electrophile-Ligand2 H
Ligand— NL
■QT ^ N-Ligand^
n H
wherein n is 0, 1, 2, 3, 4 or 5, e.g. n is 1 to 5.
[0074] The synthetic route in Scheme Xb illustrates another general method for preparing ligand-connector derivatives (i.e., P^Q1 derivatives). The method involves attaching the desired substituents to an -OH group (e.g., a phenol group) of the ligand.
SCHEME Xb
X-Connector
Ligand— OH ► Ligand— O-Connector
X = Br, CI, I, O s or OTs
1 7
H2N ' NH2 ■ Ή
^ 0 Nh2 , wherein n is 1, 2, 3, 4 or 5.
[0076] Additional examples for 6 and the connector (Q1) can be found in Table B, seen below:
Table B
[0077] Any free amino group seen in the Q1 examples of Table B above may be functionalized further to include additional functional groups, e.g., a benzoyl moiety.
[0078] In another embodiment, the P1 may be further elaborated to incorporate not only the connector moiety (Q1), but also a second ligand (P2), as represented by:
X-Connector-Ligand2
Ligand— OH - ► Ligand— O-Connector-Ligand2
X = Br, CI, I, OMs or OTs
1 9
[0079] The Qx-P2 moiety may be formed from direct attachment of Qx-P2 to the phenyl ether, or the Qx-P2 moiety may be formed from the further functionalization of any free amino group seen in the Q1 examples of Table B above to include the second ligand (P2).
[0080] The synthetic route in Scheme Xc illustrates a general method for preparing ligand-connector derivatives. The method involves attaching the desired carbonyl substituents to the free amine to form an amide, urea, or carbamate. For example, the carbonyl group can be installed by reacting amine 12 (see Scheme Xd) with carboxylic acid 13 to provide 14
(ligand-connector derivative).
SCHEME Xc
O
^—Connector Q
HO 13 H I I
Ligand— NH2 ■ ► Ligand— N— Connector
12 14
[0082] Additional examples for 13 and -C(0)R (i.e., -Q1) can be found in Table C, seen below:
Table C
Additional examples of connecting moieties can be found in Table D, seen
Table D
Example No. HW-R-WH -W-R-W- (e.g. 14, 14' or 17)
1
2
Methods
[0084] In some embodiments, contemplated bivalent compounds may be administered to a patient in need thereof. In some embodiments, a method of administering a
pharmaceutically effective amount of a compound to a patient in need thereof is provided. In some instances, a method of modulating two or more target biomolecule domains is provided. In some embodiments, the target biomolecule may be a protein. In other embodiments, the target biomolecule may be nucleic acid.
[0085] In some instances, a method of modulating two or more target biomolecule binding sites is provided, e.g., of Bcr-Abl tyrosine kinase. In some embodiments, a compound may be used to inhibit or facilitate protein-protein interactions. For example, in some cases, a compound may be capable of activating or inactivating a signaling pathway. Without wishing to be bound by any theory, a compound may bind to a target protein and affect the conformation of the target protein such that the target protein is more biologically active as
compared to when the compound does not bind the target protein. In some embodiments, the compound may bind to one region (e.g., domain) of a target molecule. In some embodiments, the compound may bind to two regions of a target molecule. In some embodiments, the compound may bind to a first region of a first target molecule and a second region of a second target molecule.
[0086] For example, in some embodiments, P1 and P2 of Formula I may each be capable of binding to a Bcr-Abl tyrosine kinase binding site. For example one of P1 may be capable of binding to the myristoyl binding pocket of Bcr-Abl tyrosine kinase and and P2 may be capable of binding to the ATP binding pocket of Bcr-Abl tyrosine kinase.
[0087] In one embodiment, a contemplated bivalent compound may be capable of binding to a first binding site and a second binding site, wherein the protein domain is within, e.g., about 40 A, or about 50 A, of the binding site.
[0088] In an embodiment, the compounds contemplated herein may be used in a method for treating diseases or conditions for which a Bcr-Abl tyrosine kinase inhibitor is indicated, for example, a compound may be used for treating treating cancer, such as chronic myelogenous leukemia. For example, provided herein is a method of treating a disease associated with a Bcr-Abl tyrosine kinase in a patient in need thereof.
[0089] Also provided herein, for example, is a use of a compound in the manufacture of a medicament for the treatment of diseases or conditions for which a Bcr-Abl tyrosine kinase inhibitor inhibitor is indicated. In a further embodiment, provided herein is a use of a compound or a pharmaceutically acceptable salt thereof in the manufacture of a medicament for the treatment of cancer, such as chronic myelogenous leukemia.
[0090] Contemplated herein are methods of treating cancers, e.g., cancers such as including hematological, epithelial including lung, breast and colon carcinomas, mesenchymal, hepatic, renal and neurological tumors, comprising administering a disclosed compound to a patient in need thereof. For example, contemplated herein is a method of treating squamous cell carcinoma, midline carcinoma or leukemia such as acute myeloid leukemia in a patient in need thereof comprising administering a contemplated bivalent compound.
[0091] In some embodiments, a ligand moiety (e.g., a pharmacophore) may have a molecular weight between 50 Da and 2000 Da, in some embodiments between 50 Da and 1500 Da, in some embodiments, between 50 Da and 1000 Da, and in some embodiments, between 50 Da and 500 Da. In certain embodiments, a ligand moiety may have a molecular weight of less
than 2000 Da, in some embodiments, less than 1000 Da, and in some embodiments less than 500 Da.
[0092] In certain embodiments, the compound utilized by one or more of the foregoing methods is one of the generic, subgeneric, or specific compounds described herein.
[0093] Disclosed compositions may be administered to patients (animals and humans) in need of such treatment in dosages that will provide optimal pharmaceutical efficacy. It will be appreciated that the dose required for use in any particular application will vary from patient to patient, not only with the particular compound or composition selected, but also with the route of administration, the nature of the condition being treated, the age and condition of the patient, concurrent medication or special diets then being followed by the patient, and other factors which those skilled in the art will recognize, with the appropriate dosage ultimately being at the discretion of the attendant physician. For treating clinical conditions and diseases noted above, a compound may be administered orally, subcutaneously, topically, parenterally, by inhalation spray or rectally in dosage unit formulations containing conventional non-toxic pharmaceutically acceptable carriers, adjuvants, and vehicles. Parenteral administration may include subcutaneous injections, intravenous or intramuscular injections, or infusion techniques.
[0094] Treatment can be continued for as long or as short a period as desired. The compositions may be administered on a regimen of, for example, one to four or more times per day. A suitable treatment period can be, for example, at least about one week, at least about two weeks, at least about one month, at least about six months, at least about 1 year, or indefinitely. A treatment period can terminate when a desired result, for example a partial or total alleviation of symptoms, is achieved.
[0095] In another aspect, pharmaceutical compositions comprising bivalent compounds as disclosed herein formulated together with a pharmaceutically acceptable carrier provided. In particular, the present disclosure provides pharmaceutical compositions bivalent compounds as disclosed herein formulated together with one or more pharmaceutically acceptable carriers. These formulations include those suitable for oral, rectal, topical, buccal, parenteral (e.g., subcutaneous, intramuscular, intradermal, or intravenous) rectal, vaginal, or aerosol administration, although the most suitable form of administration in any given case will depend on the degree and severity of the condition being treated and on the nature of the particular compound being used. For example, disclosed compositions may be formulated as a unit dose, and/or may be formulated for oral or subcutaneous administration.
[0096] Exemplary pharmaceutical compositions may be used in the form of a pharmaceutical preparation, for example, in solid, semisolid, or liquid form, which contains one or more of the compounds, as an active ingredient, in admixture with an organic or inorganic carrier or excipient suitable for external, enteral, or parenteral applications. The active ingredient may be compounded, for example, with the usual non-toxic, pharmaceutically acceptable carriers for tablets, pellets, capsules, suppositories, solutions, emulsions, suspensions, and any other form suitable for use. The active object compound is included in the pharmaceutical composition in an amount sufficient to produce the desired effect upon the process or condition of the disease.
[0097] For preparing solid compositions such as tablets, the principal active ingredient may be mixed with a pharmaceutical carrier, e.g., conventional tableting ingredients such as corn starch, lactose, sucrose, sorbitol, talc, stearic acid, magnesium stearate, dicalcium phosphate or gums, and other pharmaceutical diluents, e.g., water, to form a solid
preformulation composition containing a homogeneous mixture of a compound, or a non-toxic pharmaceutically acceptable salt thereof. When referring to these preformulation compositions as homogeneous, it is meant that the active ingredient is dispersed evenly throughout the composition so that the composition may be readily subdivided into equally effective unit dosage forms such as tablets, pills and capsules.
[0098] In solid dosage forms for oral administration (capsules, tablets, pills, dragees, powders, granules and the like), the subject composition is mixed with one or more
pharmaceutically acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose and/or acacia; (3) humectants, such as glycerol; (4)
disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds; (7) wetting agents, such as, for example, acetyl alcohol and glycerol monostearate; (8) absorbents, such as kaolin and bentonite clay; (9) lubricants, such a talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, and mixtures thereof; and (10) coloring agents. In the case of capsules, tablets and pills, the compositions may also comprise buffering agents. Solid compositions of a similar type may also be employed as fillers in soft and hard-filled
gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
[0099] A tablet may be made by compression or molding, optionally with one or more accessory ingredients. Compressed tablets may be prepared using binder (for example, gelatin or hydroxypropylmethyl cellulose), lubricant, inert diluent, preservative, disintegrant (for example, sodium starch glycolate or cross-linked sodium carboxymethyl cellulose), surface- active or dispersing agent. Molded tablets may be made by molding in a suitable machine a mixture of the subject composition moistened with an inert liquid diluent. Tablets, and other solid dosage forms, such as dragees, capsules, pills and granules, may optionally be scored or prepared with coatings and shells, such as enteric coatings and other coatings well known in the pharmaceutical-formulating art.
[00100] Compositions for inhalation or insufflation include solutions and suspensions in pharmaceutically acceptable, aqueous or organic solvents, or mixtures thereof, and powders. Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, microemulsions, solutions, suspensions, syrups and elixirs. In addition to the subject composition, the liquid dosage forms may contain inert diluents commonly used in the art, such as, for example, water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, cyclodextrins and mixtures thereof.
[00101] Suspensions, in addition to the subject composition, may contain suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
[00102] Formulations for rectal or vaginal administration may be presented as a suppository, which may be prepared by mixing a subject composition with one or more suitable non-irritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the body cavity and release the active agent.
[00103] Dosage forms for transdermal administration of a subject composition includes powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants. The
active component may be mixed under sterile conditions with a pharmaceutically acceptable carrier, and with any preservatives, buffers, or propellants which may be required.
[00104] The ointments, pastes, creams and gels may contain, in addition to a subject composition, excipients, such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
[00105] Powders and sprays may contain, in addition to a subject composition, excipients such as lactose, talc, silicic acid, aluminum hydroxide, calcium silicates and polyamide powder, or mixtures of these substances. Sprays may additionally contain customary propellants, such as chlorofluorohydrocarbons and volatile unsubstituted hydrocarbons, such as butane and propane.
[00106] Compositions and compounds may alternatively be administered by aerosol.
This is accomplished by preparing an aqueous aerosol, liposomal preparation or solid particles containing the compound. A non-aqueous (e.g., fluorocarbon propellant) suspension could be used. Sonic nebulizers may be used because they minimize exposing the agent to shear, which may result in degradation of the compounds contained in the subject compositions. Ordinarily, an aqueous aerosol is made by formulating an aqueous solution or suspension of a subject composition together with conventional pharmaceutically acceptable carriers and stabilizers. The carriers and stabilizers vary with the requirements of the particular subject composition, but typically include non-ionic surfactants (T weens, Pluronics, or polyethylene glycol), innocuous proteins like serum albumin, sorbitan esters, oleic acid, lecithin, amino acids such as glycine, buffers, salts, sugars, or sugar alcohols. Aerosols generally are prepared from isotonic solutions.
[00107] Pharmaceutical compositions suitable for parenteral administration comprise a subject composition in combination with one or more pharmaceutically-acceptable sterile isotonic aqueous or non-aqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents.
[00108] Examples of suitable aqueous and non-aqueous carriers which may be employed in the pharmaceutical compositions include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as
olive oil, and injectable organic esters, such as ethyl oleate and cyclodextrins. Proper fluidity may be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants
[00109] In another aspect, enteral pharmaceutical formulations including a disclosed pharmaceutical composition comprising bivalent compounds, an enteric material; and a pharmaceutically acceptable carrier or excipient thereof are provided. Enteric materials refer to polymers that are substantially insoluble in the acidic environment of the stomach, and that are predominantly soluble in intestinal fluids at specific pHs. The small intestine is the part of the gastrointestinal tract (gut) between the stomach and the large intestine, and includes the duodenum, jejunum, and ileum. The pH of the duodenum is about 5.5, the pH of the jejunum is about 6.5 and the pH of the distal ileum is about 7.5. Accordingly, enteric materials are not soluble, for example, until a pH of about 5.0, of about 5.2, of about 5.4, of about 5.6, of about 5.8, of about 6.0, of about 6.2, of about 6.4, of about 6.6, of about 6.8, of about 7.0, of about 7.2, of about 7.4, of about 7.6, of about 7.8, of about 8.0, of about 8.2, of about 8.4, of about 8.6, of about 8.8, of about 9.0, of about 9.2, of about 9.4, of about 9.6, of about 9.8, or of about 10.0. Exemplary enteric materials include cellulose acetate phthalate (CAP), hydroxypropyl methylcellulose phthalate (HPMCP), polyvinyl acetate phthalate (PVAP), hydroxypropyl methylcellulose acetate succinate (HPMCAS), cellulose acetate trimellitate, hydroxypropyl methylcellulose succinate, cellulose acetate succinate, cellulose acetate hexahydrophthalate, cellulose propionate phthalate, cellulose acetate maleat, cellulose acetate butyrate, cellulose acetate propionate, copolymer of methylmethacrylic acid and methyl methacrylate, copolymer of methyl acrylate, methylmethacrylate and methacrylic acid, copolymer of methylvinyl ether and maleic anhydride (Gantrez ES series), ethyl methyacrylate-methylmethacrylate- chlorotrimethylammonium ethyl acrylate copolymer, natural resins such as zein, shellac and copal collophorium, and several commercially available enteric dispersion systems (e. g. , Eudragit L30D55, Eudragit FS30D, Eudragit L100, Eudragit S 100, Kollicoat EMM30D, Estacryl 30D, Coateric, and Aquateric). The solubility of each of the above materials is either known or is readily determinable in vitro. The foregoing is a list of possible materials, but one of skill in the art with the benefit of the disclosure would recognize that it is not comprehensive and that there are other enteric materials that may be used.
[00110] Advantageously, kits are provided containing one or more compositions. Such kits include a suitable dosage form such as those described above and instructions describing
the method of using such dosage form to treat a disease or condition. The instructions would direct the consumer or medical personnel to administer the dosage form according to administration modes known to those skilled in the art. Such kits could advantageously be packaged and sold in single or multiple kit units. An example of such a kit is a so-called blister pack. Blister packs are well known in the packaging industry and are being widely used for the packaging of pharmaceutical unit dosage forms (tablets, capsules, and the like). Blister packs generally consist of a sheet of relatively stiff material covered with a foil of a preferably transparent plastic material. During the packaging process recesses are formed in the plastic foil. The recesses have the size and shape of the tablets or capsules to be packed. Next, the tablets or capsules are placed in the recesses and the sheet of relatively stiff material is sealed against the plastic foil at the face of the foil which is opposite from the direction in which the recesses were formed. As a result, the tablets or capsules are sealed in the recesses between the plastic foil and the sheet. Preferably the strength of the sheet is such that the tablets or capsules can be removed from the blister pack by manually applying pressure on the recesses whereby an opening is formed in the sheet at the place of the recess. The tablet or capsule can then be removed via said opening.
[00111] It may be desirable to provide a memory aid on the kit, e.g., in the form of numbers next to the tablets or capsules whereby the numbers correspond with the days of the regimen which the tablets or capsules so specified should be ingested. Another example of such a memory aid is a calendar printed on the card, e.g., as follows "First Week, Monday, Tuesday, . . . etc. . . . Second Week, Monday, Tuesday, . . . " etc. Other variations of memory aids will be readily apparent. A "daily dose" can be a single tablet or capsule or several pills or capsules to be taken on a given day. Also, a daily dose of a first compound can consist of one tablet or capsule while a daily dose of the second compound can consist of several tablets or capsules and vice versa. The memory aid should reflect this.
[00112] Also contemplated herein are methods and compositions that include a second active agent, or administering a second active agent.
[00113] Certain terms employed in the specification, examples, and appended claims are collected here. These definitions should be read in light of the entirety of the disclosure and understood as by a person of skill in the art. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by a person of ordinary skill in the art.
DEFINITIONS
[00114] In some embodiments, the compounds, as described herein, may be substituted with any number of substituents or functional moieties. In general, the term "substituted" whether preceded by the term "optionally" or not, and substituents contained in formulas, refer to the replacement of hydrogen radicals in a given structure with the radical of a specified substituent.
[00115] In some instances, when more than one position in any given structure may be substituted with more than one substituent selected from a specified group, the substituent may be either the same or different at every position.
[00116] As used herein, the term "substituted" is contemplated to include all permissible substituents of organic and inorganic compounds. In a broad aspect, the permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, aromatic and non-aromatic substituents of organic compounds. In some embodiments, heteroatoms such as nitrogen may have hydrogen substituents and/or any permissible substituents of organic compounds described herein which satisfy the valencies of the heteroatoms. Non-limiting examples of substituents include acyl; aliphatic; heteroaliphatic; phenyl; naphthyl; heteroaryl; arylalkyl; heteroarylalkyl; alkoxy; cycloalkoxy;
heterocyclylalkoxy; heterocyclyloxy; heterocyclyloxyalkyl; alkenyloxy; alkynyloxy; phenoxy; heteroalkoxy; heteroaryloxy; alkylthio; phenylthio; heteroalkylthio; heteroarylthio; oxo; -F; - CI; -Br; -I; -OH; -N02; -CN; -SCN; -SRX; -CF3; -CH2CF3; -CHC12; -CH2OH; -CH2CH2OH; - CH2NH2; -CH2S02CH3; -ORx, -C(0)Rx; -C02(Rx); -C(0)N(Rx)2; -OC(0)Rx; -OC02Rx; - OC(0)N(Rx)2; -N(RX)2; -SORx; -S(0)2Rx; -NRxC(0)Rx; or -C(RX)3; wherein each occurrence of Rx independently is hydrogen, aliphatic, heteroaliphatic, phenyl, naphthyl, heteroaryl, arylalkyl, or heteroarylalkyl, wherein any of the aliphatic, heteroaliphatic, arylalkyl, or heteroarylalkyl substituents described above and herein may be substituted or unsubstituted, branched or unbranched, cyclic or acyclic, and wherein any of the phenyl, naphthyl, or heteroaryl substituents described above and herein may be substituted or unsubstituted. Furthermore, the compounds described herein are not intended to be limited in any manner by the permissible substituents of organic compounds. In some embodiments, combinations of substituents and variables described herein may be preferably those that result in the formation of stable compounds. The term "stable," as used herein, refers to compounds which possess stability sufficient to allow manufacture and which maintain the integrity of the compound for a
sufficient period of time to be detected and preferably for a sufficient period of time to be useful for the purposes detailed herein.
[00117] The term "acyl," as used herein, refers to a moiety that includes a carbonyl group. In some embodiments, an acyl group may have a general formula selected from - C(0)Rx; -C02(Rx); -C(0)N(Rx)2; -OC(0)Rx; -OC02Rx; and -OC(0)N(Rx)2; wherein each occurrence of Rx independently includes, but is not limited to, hydrogen, aliphatic,
heteroaliphatic, phenyl, naphthyl, heteroaryl, arylalkyl, or heteroarylalkyl, wherein any of the aliphatic, heteroaliphatic, arylalkyl, or heteroarylalkyl substituents described above and herein may be substituted or unsubstituted, branched or unbranched, cyclic or acyclic, and wherein any of the phenyl, naphthyl, or heteroaryl substituents described above and herein may be substituted or unsubstituted.
[00118] The term "aliphatic," as used herein, includes both saturated and unsaturated, straight chain (i.e., unbranched), branched, acyclic, cyclic, or polycyclic aliphatic
hydrocarbons, which are optionally substituted with one or more functional groups. As will be appreciated by one of ordinary skill in the art, "aliphatic" is intended herein to include, but is not limited to, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, and cycloalkynyl moieties. The term "heteroaliphatic," as used herein, refers to aliphatic moieties that contain one or more oxygen, sulfur, nitrogen, phosphorus, or silicon atoms, e.g., in place of carbon atoms.
Heteroaliphatic moieties may be branched, unbranched, cyclic or acyclic and include saturated and unsaturated heterocycles such as morpholino, pyrrolidinyl, etc.
[00119] In general, the terms "aryl," "aromatic," "heteroaryl," and "heteroaromtic" as used herein, refer to stable mono- or polycyclic, heterocyclic, polycyclic, and polyheterocyclic unsaturated moieties having preferably 3-14 carbon atoms, each of which may be substituted or unsubstituted. Substituents include, but are not limited to, any of the previously mentioned substituents, i.e., the substituents recited for aliphatic moieties, or for other moieties as disclosed herein, resulting in the formation of a stable compound. In certain embodiments, aryl or aromatic refers to a mono- or bicyclic carbocyclic ring system having one or two aromatic rings selected from phenyl, naphthyl, tetrahydronaphthyl, indanyl, and indenyl. In certain embodiments, the term heteroaryl, as used herein, refers to a cyclic aromatic radical having from five to ten ring atoms of which one ring atom is selected from the group consisting of S, O, and N; zero, one, or two ring atoms are additional heteroatoms independently selected from the group consisting of S, O, and N; and the remaining ring atoms are carbon, the radical being joined to the rest of the molecule via any of the ring atoms. Heteroaryl moieties may be
selected from: pyridyl, pyrazinyl, pyrimidinyl, pyrrolyl, pyrazolyl, imidazolyl, thiazolyl, oxazolyl, isooxazolyl, thiadiazolyl,oxadiazolyl, thiophenyl, furanyl, quinolinyl, isoquinolinyl, and the like.
[00120] It will be appreciated that aryl, aromatic, heteroaryl, and heteroaromatic groups described herein can be unsubstituted or substituted, wherein substitution includes replacement of one, two, three, or more of the hydrogen atoms thereon independently with a group selected from: Ci_6alkyl; phenyl; heteroaryl; benzyl; heteroarylalkyl; Ci_6alkoxy; Ci_6cycloalkoxy; Ci_ 6heterocyclylalkoxy;
heterocyclyloxyalkyl; C2-6alkenyloxy; C2_
6alkynyloxy; phenoxy; heteroalkoxy; heteroaryloxy; Ci_6alkylthio; phenylthio; heteroalkylthio; heteroarylthio; oxo; -F; -CI; -Br; -I; -OH; -N02; -CN; -CF3; -CH2CF3; -CHC12; -CH2OH; -
CH2CH2OH; -CH2NH2; -CH2S02CH3; -C(0)Rx; -C02(Rx); -CON(Rx)2; -OC(0)Rx; -OC02Rx; - OCON(Rx)2; -N(RX)2; - S(0)2Rx; -NRx(CO)Rx, wherein each occurrence of Rx is selected from hydrogen, Ci-6alkyl; aliphatic, heteroaliphatic, phenyl, or heteroaryl. Additional examples of generally applicable substituents are illustrated by the specific embodiments shown in the Examples that are described herein.
[00121] The term "heterocyclic," as used herein, refers to an aromatic or non-aromatic, partially unsaturated or fully saturated, 3- to 10-membered ring system, which includes single rings of 3 to 8 atoms in size and bi- and tri-cyclic ring systems which may include aromatic five- or six-membered aryl or aromatic heterocyclic groups fused to a non-aromatic ring.
These heterocyclic rings include those having from one to three heteroatoms independently selected from the group consisting of oxygen, sulfur, and nitrogen, in which the nitrogen and sulfur heteroatoms may optionally be oxidized and the nitrogen heteroatom may optionally be quaternized. In certain embodiments, the term heterocyclic refers to a non-aromatic 5-, 6-, or 7-membered ring or a polycyclic group wherein at least one ring atom is a heteroatom selected from the group consisting of O, S, and N (wherein the nitrogen and sulfur heteroatoms may be optionally oxidized), including, but not limited to, a bi- or tri-cyclic group, comprising fused six-membered rings having between one and three heteroatoms independently selected from the group consisting of the oxygen, sulfur, and nitrogen, wherein (i) each 5-membered ring has 0 to 2 double bonds, each 6-membered ring has 0 to 2 double bonds, and each 7-membered ring has 0 to 3 double bonds, (ii) the nitrogen and sulfur heteroatoms may be optionally oxidized, (iii) the nitrogen heteroatom may optionally be quaternized, and (iv) any of the above heterocyclic rings may be fused to an aryl or heteroaryl ring.
[00122] The term "alkenyl" as used herein refers to an unsaturated straight or branched hydrocarbon having at least one carbon-carbon double bond, such as a straight or branched group of 2-6 or 3-4 carbon atoms, referred to herein for example as C2-6alkenyl, and C3- 4alkenyl, respectively. Exemplary alkenyl groups include, but are not limited to, vinyl, allyl, butenyl, pentenyl, etc.
[00123] The term "alkenyloxy" used herein refers to a straight or branched alkenyl group attached to an oxygen (alkenyl-O). Exemplary alkenoxy groups include, but are not limited to, groups with an alkenyl group of 3-6 carbon atoms referred to herein as C3_6alkenyloxy.
Exemplary "alkenyloxy" groups include, but are not limited to allyloxy, butenyloxy, etc.
[00124] The term "alkoxy" as used herein refers to a straight or branched alkyl group attached to an oxygen (alkyl-O-). Exemplary alkoxy groups include, but are not limited to, groups with an alkyl group of 1-6 or 2-6 carbon atoms, referred to herein as Ci-6alkoxy, and C2- C6alkoxy, respectively. Exemplary alkoxy groups include, but are not limited to methoxy, ethoxy, isopropoxy, etc.
[00125] The term "alkoxycarbonyl" as used herein refers to a straight or branched alkyl group attached to oxygen, attached to a carbonyl group (alkyl-O-C(O)-). Exemplary alkoxycarbonyl groups include, but are not limited to, alkoxycarbonyl groups of 1-6 carbon atoms, referred to herein as Ci_6alkoxycarbonyl. Exemplary alkoxycarbonyl groups include, but are not limited to, methoxycarbonyl, ethoxycarbonyl, t-butoxycarbonyl, etc.
[00126] The term "alkynyloxy" used herein refers to a straight or branched alkynyl group attached to an oxygen (alkynyl-O)). Exemplary alkynyloxy groups include, but are not limited to, propynyloxy.
[00127] The term "alkyl" as used herein refers to a saturated straight or branched hydrocarbon, for example, such as a straight or branched group of 1-6, 1-4, or 1-3 carbon atoms, referred to herein as Ci-6alkyl, Ci_4alkyl, and Ci_3alkyl, respectively. Exemplary alkyl groups include, but are not limited to, methyl, ethyl, propyl, isopropyl, 2-methyl- 1 -propyl, 2- methyl-2-propyl, 2-methyl-l -butyl, 3 -methyl- 1 -butyl, 3-methyl-2-butyl, 2,2-dimethyl-l -propyl, 2-methyl- 1-pentyl, 3 -methyl- 1-pentyl, 4-methyl-l-pentyl, 2-methyl-2-pentyl, 3-methyl-2- pentyl, 4-methyl-2-pentyl, 2,2-dimethyl-l -butyl, 3,3-dimethyl-l-butyl, 2-ethyl-l -butyl, butyl, isobutyl, t-butyl, pentyl, isopentyl, neopentyl, hexyl, etc.
[00128] The term "alkylene" as used herein refers to a bivalent saturated straight or branched hydrocarbon, for example, such as a straight or branched group of 1-6, 1-4, or 1-3 carbon atoms, referred to herein as -Ci_6alkylene-, -Ci_4alkylene-, and -Ci_3alkylene-,
respectively, where the alkylene has two open valences. Exemplary alkyl groups include, but are not limited to, methylene, ethylene, propylene, isopropylene, 2-methyl-l -propylene, 2- methyl-2-propylene, 2-methyl-l -butylene, 3 -methyl- 1-butylene, 3-methyl-2-butylene, 2,2- dimethyl-1 -propylene, 2-methyl-l -pentylene, 3 -methyl- 1-pentylene, 4-methyl-l-pentylene, 2- methyl-2-pentylene, 3-methyl-2-pentylene, 4-methyl-2-pentylene, 2,2-dimethyl- 1-butylene, 3, 3 -dimethyl- 1-butylene, 2-ethyl- 1-butylene, butylene, isobutylene, t-butylene, pentylene, isopentylene, neopentylene, hexylene, etc.
[00129] The term "alkylcarbonyl" as used herein refers to a straight or branched alkyl group attached to a carbonyl group (alkyl-C(O)-). Exemplary alkylcarbonyl groups include, but are not limited to, alkylcarbonyl groups of 1-6 atoms, referred to herein as Ci_
6alkylcarbonyl groups. Exemplary alkylcarbonyl groups include, but are not limited to, acetyl, propanoyl, isopropanoyl, butanoyl, etc.
[00130] The term "alkynyl" as used herein refers to an unsaturated straight or branched hydrocarbon having at least one carbon-carbon triple bond, such as a straight or branched group of 2-6, or 3-6 carbon atoms, referred to herein as C2-6alkynyl, and C3_6alkynyl, respectively.
Exemplary alkynyl groups include, but are not limited to, ethynyl, propynyl, butynyl, pentynyl, hexynyl, methylpropynyl, etc.
[00131] The term "carbonyl" as used herein refers to the radical -C(O)-.
[00132] The term "carboxylic acid" as used herein refers to a group of formula -CO2H.
[00133] The term "cyano" as used herein refers to the radical -CN.
[00134] The term "cycloalkoxy" as used herein refers to a cycloalkyl group attached to an oxygen (cycloalkyl-O-).
[00135] The term "cycloalkyl" as used herein refers to a monocyclic saturated or partially unsaturated hydrocarbon group of for example 3-6, or 4-6 carbons, referred to herein, e.g., as C3_6cycloalkyl or C4_6cycloalkyl and derived from a cycloalkane. Exemplary cycloalkyl groups include, but are not limited to, cyclohexyl, cyclohexenyl, cyclopentyl, cyclobutyl or, cyclopropyl.
[00136] The terms "halo" or "halogen" as used herein refer to F, CI, Br, or I.
[00137] The term "heterocyclylalkoxy" as used herein refers to a heterocyclyl- alkyl-O- group.
[00138] The term "heterocyclyloxyalkyl" refers to a heterocyclyl-O-alkyl- group.
[00139] The term "heterocyclyloxy" refers to a heterocyclyl-O- group.
[00140] The term "heteroaryloxy" refers to a heteroaryl-O- group.
[00141] The terms "hydroxy" and "hydroxyl" as used herein refers to the radical -OH.
[00142] The term "oxo" as used herein refers to the radical =0.
[00143] The term "connector" as used herein to refers to an atom or a collection of atoms optionally used to link interconnecting moieties, such as a disclosed connecting moiety (i.e., linker) and a pharmacophore. Contemplated connectors are generally hydrolytically stable.
[00144] "Treating" includes any effect, e.g., lessening, reducing, modulating, or eliminating, that results in the improvement of the condition, disease, disorder and the like.
[00145] "Pharmaceutically or pharmacologically acceptable" include molecular entities and compositions that do not produce an adverse, allergic, or other untoward reaction when administered to an animal, or a human, as appropriate. For human administration, preparations should meet sterility, pyrogenicity, general safety and purity standards as required by FDA Office of Biologies standards.
[00146] The term "pharmaceutically acceptable carrier" or "pharmaceutically acceptable excipient" as used herein refers to any and all solvents, dispersion media, coatings, isotonic and absorption delaying agents, and the like, that are compatible with pharmaceutical
administration. The use of such media and agents for pharmaceutically active substances is well known in the art. The compositions may also contain other active compounds providing supplemental, additional, or enhanced therapeutic functions.
[00147] The term "pharmaceutical composition" as used herein refers to a composition comprising at least one compound as disclosed herein formulated together with one or more pharmaceutically acceptable carriers.
[00148] "Individual," "patient," or "subject" are used interchangeably and include any animal, including mammals, preferably mice, rats, other rodents, rabbits, dogs, cats, swine, cattle, sheep, horses, or primates, and most preferably humans. The compounds can be administered to a mammal, such as a human, but can also be administered to other mammals such as an animal in need of veterinary treatment, e.g., domestic animals (e.g., dogs, cats, and the like), farm animals (e.g., cows, sheep, pigs, horses, and the like) and laboratory animals (e.g., rats, mice, guinea pigs, and the like). The mammal treated is desirably a mammal in which treatment of obesity, or weight loss is desired. "Modulation" includes antagonism (e.g., inhibition), agonism, partial antagonism and/or partial agonism.
[00149] In the present specification, the term "therapeutically effective amount" means the amount of the subject compound that will elicit the biological or medical response of a tissue, system, animal, or human that is being sought by the researcher, veterinarian, medical
doctor, or other clinician. The compounds are administered in therapeutically effective amounts to treat a disease. Alternatively, a therapeutically effective amount of a compound is the quantity required to achieve a desired therapeutic and/or prophylactic effect, such as an amount which results in weight loss.
[00150] The term "pharmaceutically acceptable salt(s)" as used herein refers to salts of acidic or basic groups that may be present in compounds used in the present compositions. Compounds included in the present compositions that are basic in nature are capable of forming a wide variety of salts with various inorganic and organic acids. The acids that may be used to prepare pharmaceutically acceptable acid addition salts of such basic compounds are those that form non-toxic acid addition salts, i.e., salts containing pharmacologically acceptable anions, including but not limited to malate, oxalate, chloride, bromide, iodide, nitrate, sulfate, bisulfate, phosphate, acid phosphate, isonicotinate, acetate, lactate, salicylate, citrate, tartrate, oleate, tannate, pantothenate, bitartrate, ascorbate, succinate, maleate, gentisinate, fumarate, gluconate, glucaronate, saccharate, formate, benzoate, glutamate, methanesulfonate, ethanesulfonate, benzenesulfonate, >-toluenesulfonate and pamoate (i.e., l, l'-methylene-bis-(2-hydroxy-3- naphthoate)) salts. Compounds included in the present compositions that are acidic in nature are capable of forming base salts with various pharmacologically acceptable cations. Examples of such salts include alkali metal or alkaline earth metal salts and, particularly, calcium, magnesium, sodium, lithium, zinc, potassium, and iron salts. Compounds included in the present compositions that include a basic or acidic moiety may also form pharmaceutically acceptable salts with various amino acids. The compounds of the disclosure may contain both acidic and basic groups; for example, one amino and one carboxylic acid group. In such a case, the compound can exist as an acid addition salt, a zwitterion, or a base salt.
[00151] The compounds of the disclosure may contain one or more chiral centers and/or double bonds and, therefore, exist as stereoisomers, such as geometric isomers, enantiomers or diastereomers. The term "stereoisomers" when used herein comprises all geometric isomers, enantiomers or diastereomers. These compounds may be designated by the symbols "R" or "5," depending on the configuration of substituents around the stereogenic carbon atom.
Various stereoisomers of these compounds and mixtures thereof are encompassed by this disclosure. Stereoisomers include enantiomers and diastereomers. Mixtures of enantiomers or diastereomers may be designated "(±)" in nomenclature, but the skilled artisan will recognize that a structure may denote a chiral center implicitly.
[00152] The compounds of the disclosure may contain one or more chiral centers and/or double bonds and, therefore, exist as geometric isomers, enantiomers or diastereomers. The enantiomers and diastereomers may be designated by the symbols "(+)," "(-)." "R" or "5," depending on the configuration of substituents around the stereogenic carbon atom, but the skilled artisan will recognize that a structure may denote a chiral center implicitly. Geometric isomers, resulting from the arrangement of substituents around a carbon-carbon double bond or arrangement of substituents around a cycloalkyl or heterocyclic ring, can also exist in the compounds. The symbol = denotes a bond that may be a single, double or triple bond as described herein. Substituents around a carbon-carbon double bond are designated as being in the "Z" or "is" configuration wherein the terms "Z" and "is" are used in accordance with
IUPAC standards. Unless otherwise specified, structures depicting double bonds encompass both the "is " and "Z" isomers. Substituents around a carbon-carbon double bond alternatively can be referred to as "cis" or "trans," where "cis" represents substituents on the same side of the double bond and "trans" represents substituents on opposite sides of the double bond. The arrangement of substituents around a carbocyclic ring can also be designated as "cis" or
"trans." The term "cis" represents substituents on the same side of the plane of the ring and the term "trans" represents substituents on opposite sides of the plane of the ring. Mixtures of compounds wherein the substituents are disposed on both the same and opposite sides of plane of the ring are designated "cis/trans."
[00153] The term "stereoisomers" when used herein comprises all geometric isomers, enantiomers or diastereomers. Various stereoisomers of these compounds and mixtures thereof are encompassed by this disclosure.
[00154] Individual enantiomers and diastereomers of the compounds can be prepared synthetically from commercially available starting materials that contain asymmetric or stereogenic centers, or by preparation of racemic mixtures followed by resolution methods well known to those of ordinary skill in the art. These methods of resolution are exemplified by (1) attachment of a mixture of enantiomers to a chiral auxiliary, separation of the resulting mixture of diastereomers by recrystallization or chromatography and liberation of the optically pure product from the auxiliary, (2) salt formation employing an optically active resolving agent, (3) direct separation of the mixture of optical enantiomers on chiral liquid chromatographic columns or (4) kinetic resolution using stereoselective chemical or enzymatic reagents.
Racemic mixtures can also be resolved into their component enantiomers by well known methods, such as chiral-phase gas chromatography or crystallizing the compound in a chiral
solvent. Stereoselective syntheses, a chemical or enzymatic reaction in which a single reactant forms an unequal mixture of stereoisomers during the creation of a new stereocenter or during the transformation of a pre-existing one, are well known in the art. Stereoselective syntheses encompass both enantio- and diastereoselective transformations. For examples, see Carreira and Kvaerno, Classics in Stereoselective Synthesis, Wiley-VCH: Weinheim, 2009.
[00155] The compounds disclosed herein can exist in solvated as well as unsolvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like. In one embodiment, the compound is amorphous. In one embodiment, the compound is a polymorph. In another embodiment, the compound is in a crystalline form.
[00156] Also embraced are isotopically labeled compounds which are identical to those recited herein, except that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature.
Examples of isotopes that can be incorporated into the compounds include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine and chlorine, such as 10B, 2H, 3H, 13C, 14C, 15N, 180, 170, 31P, 32P, 35S, 18F, and 36C1, respectively. For example, a compound may have one or more H atom replaced with deuterium.
[00157] Certain isotopically-labeled disclosed compounds (e.g., those labeled with 3H and 14C) are useful in compound and/or substrate tissue distribution assays. Tritiated (i.e., 3H) and carbon-14 (i.e., 14C) isotopes are particularly preferred for their ease of preparation and detectability. Further, substitution with heavier isotopes such as deuterium (i.e., 2H) may afford certain therapeutic advantages resulting from greater metabolic stability (e.g., increased in vivo half-life or reduced dosage requirements) and hence may be preferred in some circumstances. Isotopically labeled compounds can generally be prepared by following procedures analogous to those disclosed in the Examples herein by substituting an isotopically labeled reagent for a non-isotopically labeled reagent.
[00158] The term "prodrug" refers to compounds that are transformed in vivo to yield a disclosed compound or a pharmaceutically acceptable salt, hydrate or solvate of the compound. The transformation may occur by various mechanisms (such as by esterase, amidase, phosphatase, oxidative and or reductive metabolism) in various locations (such as in the intestinal lumen or upon transit of the intestine, blood, or liver). Prodrugs are well known in the art (for example, see Rautio, Kumpulainen, et al, Nature Reviews Drug Discovery 2008, 7, 255). For example, if a compound or a pharmaceutically acceptable salt, hydrate, or solvate of the compound contains a carboxylic acid functional group, a prodrug can comprise an ester
formed by the replacement of the hydrogen atom of the acid group with a group such as (Ci_8)alkyl, (C2-i2)alkanoyloxymethyl, l-(alkanoyloxy)ethyl having from 4 to 9 carbon atoms, 1 -methyl- l-(alkanoyloxy)-ethyl having from 5 to 10 carbon atoms, alkoxycarbonyloxymethyl having from 3 to 6 carbon atoms, l-(alkoxycarbonyloxy)ethyl having from 4 to 7 carbon atoms, 1 -methyl- l-(alkoxycarbonyloxy)ethyl having from 5 to 8 carbon atoms, N- (alkoxycarbonyl)aminomethyl having from 3 to 9 carbon atoms,
l-(N-(alkoxycarbonyl)amino)ethyl having from 4 to 10 carbon atoms, 3-phthalidyl,
4-crotonolactonyl, gamma-butyrolacton-4-yl, di-N,N-(Ci-C2)alkylamino(C2-C3)alkyl (such as β-dimethylaminoethyl), carbamoyl-(Ci-C2)alkyl, N,N-di(Ci-C2)alkylcarbamoyl-(Ci-C2)alkyl and piperidino-, pyrrolidino- or morpholino(C2-C3)alkyl.
[00159] Similarly, if a compound contains an alcohol functional group, a prodrug can be formed by the replacement of the hydrogen atom of the alcohol group with a group such as (Ci_ 6)alkanoyloxymethyl, l-((Ci-6)alkanoyloxy)ethyl, 1 -methyl- l-((Ci-6)alkanoyloxy)ethyl (Ci- 6)alkoxycarbonyloxymethyl, N-(Ci_6)alkoxycarbonylaminomethyl, succinoyl, (Ci_6)alkanoyl, a- amino(Ci_4)alkanoyl, arylacyl and a-aminoacyl, or α-aminoacyl-a-aminoacyl, where each a- aminoacyl group is independently selected from the naturally occurring L-amino acids, P(0)(OH)2, -P(0)(0(Ci-C6)alkyl)2 or glycosyl (the radical resulting from the removal of a hydroxyl group of the hemiacetal form of a carbohydrate).
[00160] If a compound incorporates an amine functional group, a prodrug can be formed, for example, by creation of an amide or carbamate, an N-acyloxyalkyl derivative, an
(oxodioxolenyl)methyl derivative, an N-Mannich base, imine, or enamine. In addition, a secondary amine can be metabolically cleaved to generate a bioactive primary amine, or a tertiary amine can be metabolically cleaved to generate a bioactive primary or secondary amine. For examples, see Simplicio, et ah, Molecules 2008, 13, 519 and references therein.
INCORPORATION BY REFERENCE
[00161] All publications and patents mentioned herein, including those items listed below, are hereby incorporated by reference in their entirety for all purposes as if each individual publication or patent was specifically and individually incorporated by reference. In case of conflict, the present application, including any definitions herein, will control.
EXAMPLES
[00162] The compounds described herein can be prepared in a number of ways based on the teachings contained herein and synthetic procedures known in the art. Where a particular stereochemistry is indicated for a compound, one of ordinary skill in the art would recognize that other stereoisomers of the compound may also be formed. In some cases, a starting material or intermediate used in the synthesis of a contemplated compound may have an enantiomeric excess greater than 0, e.g., greater than about 95%, greater than about 98%, greater than about 99%, or essentially 100%. For example, in some cases, a starting material or intermediate may be essentially stereoisomerically pure. However, partial or complete loss of chiral integrity may occur during the synthesis of the contemplated compound thereby reducing or eliminating the enantiomeric excess. For example, where a stereoisomerically pure starting material or intermediate is used in a synthesis of a contemplated compound, partial or complete loss of chiral integrity results in a stereoisomeric mixture. A stereoisomeric mixture may be partially or essentially completely resolved by subjecting the stereoisomeric mixture to a chiral purification technique (e.g., chiral HPLC purification).
EXAMPLE:
[00163] Examples 1-6: Synthesis of bivalent compounds.
[00164] Bivalent compounds were synthesized according to the procedures described below. Reagents were either purchased commercially or prepared according to previously described methods.
[00165] Synthetic scheme: Example 1:
[00166] 7V-(2-Chloro-6-methylphenyl)-2-((2-methyl-6-(4-(2-((l-(l-oxo-l-(3-(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)phenyl)-5,8,ll,14,l'7,20,23-heptaoxa-2- azapentacosan-25-yl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l-yl)pyrimidin-4- yl)amino)thiazole-5-carboxamide [Example 1] :
[00167] A solution of 3-(6-((4-(trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)benzoic acid (39 mg, 0.104 mmol) in DMF (2 mL) was charged with 2-((6-(4-(2-((l-(23-amino- 3,6,9, 12, 15, 18,21-heptaoxatricosyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2- methylpyrimidin-4-yl)amino)-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide (80 mg, 0.087 mmol), HATU (66 mg, 0.174 mmol) and DIPEA (22 mg, 0.174 mmol). The resulting solution was stirred at room temperature for 1 h and the reaction mixture was partitioned between ethyl acetate (10 mL) and water (5 mL) and separated. The aqueous layer was extracted with ethyl acetate (3 X 10 mL) and the combined organic layers were dried over anhydrous Na2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by preparative HPLC to afford 30 mg, 27% yield of the title compound as an orange solid. 'H NMR (400 MHz, DMSO-i¾): δ = 10.03 (s, 1H), 9.89 (s, 1H), 8.75 (m, 2H), 8.54 (s, 1H), 8.31 (s, 1H), 8.24 - 8.16 (m, 2H), 8.06 (s, 1H), 7.98 (d, J= 7.7 Hz, 1H), 7.86 (d, J = 8.6 Hz, 2H), 7.63 (t, J= 7.7 Hz, 1H), 7.40 - 7.25 (m, 6H), 6.04 (s, 1H), 4.50 (d, J= 5.9 Hz, 4H), 3.80 (t, J= 5.2 Hz, 2H), 3.61 - 3.41 (m, 40H), 2.39 (s, 3H), 2.23 (s, 3H); MS (ES+): m/z = 639.15, 640.65 [M/2] +; LCMS: tR = 2.76 min.
[00168] 2-((6-(4-(2-((l-(23-Amino-3,6,9,12,15,18,21-heptaoxatricosyl)-lH-l,2,3- triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2-methylpyrimidin-4-yl)amino)-7V-(2-chloro-6- methylphenyl)thiazole-5-carboxamide (9) :
[00169] A solution of 2-((6-(4-(2-((l-(23-azido-3,6,9, 12, 15, 18,21-heptaoxatricosyl)-lH- l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2-methylpyrimidin-4-yl)amino)-N-(2-chloro- 6-methylphenyl)thiazole-5-carboxamide (200 mg, 0.212 mmol) in THF:H20 (4: 1 mL) was charged with triphenylphosphine (TPP) (166 mg, 0.635 mmol) and stirred at room temperature for 12 h. The reaction mixture was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 3% methanol in DCM to give 80 mg, 41% yield of the title compound as an off white solid. LCMS didn't show ionize mass peak.
[00170] 2-((6-(4-(2-((l-(23-Azido-3,6,9,12,15,18,21-heptaoxatricosyl)-lH-l,2,3- triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2-methylpyrimidin-4-yl)amino)-7V-(2-chloro-6- methylphenyl)thiazole-5-carboxamide (8) :
[00171] A solution of l-(2-((l-(23-Azido-3,6,9,12,15,18,21-heptaoxatricosyl)-lH-l,2,3- triazol-4-yl)methoxy)ethyl)piperazine (400 mg, 0.680 mmol) in DMF (10 mL) was charged with potassium carbonate (282 mg, 2.04 mmol) and 2-((6-chloro-2-methylpyrimidin-4- yl)amino)-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide (161 mg, 0.408 mmol). The resulting solution was heated at 80°C for 16 h and allowed cool to room temperature and concentrated in vacuo. The residue obtained was diluted with water (10 mL) and extracted with ethyl acetate (3 X 20 mL) and the combined organic layers were dried over anhydrous Na2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 2% methanol in DCM to give 200 mg , 31% yield of the title compound as a yellow solid. MS (ES+): m/z = 946.05, 947.05, 948.15 [Μ+Η] +; LCMS: fR = 2.47 min.
[00172] l-(2-((l-(23-Azido-3,6,9,12,15,18,21-heptaoxatricosyl)-lH-l,2,3-triazol-4- yl)methoxy)ethyl)piperazine (7):
[00173] A solution of tert-butyl 4-(2-((l-(23-azido-3,6,9,12,15,18,21-heptaoxatricosyl)- lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (900 mg, 1.31 mmol) in dioxane (5 mL) was charged with dioxane: HCl (4M, 10 mL) at 0°C and stirred at room temperature for 2 h. The reaction mixture was triturated with diethyl ether (10 mL) and concentrated in vacuo resulting in 800 mg of crude compound as an off white solid. The crude compound was used in the next step without further purification. MS (ES+): m/z = 589.00, 590.00 [M+H]+.
[00174] teri-Butyl 4-(2-((l-(23-azido-3,6,9,12,15,18,21-heptaoxatricosyl)-lH-l,2,3- triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (6) :
[00175] A solution of tert-butyl 4-(2-((l-(23-(tosyloxy)-3,6,9,12,15,18,21- heptaoxatricosyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (1.20 g, 1.47
mmol) in EtOH (20 mL) was charged with NaN3 (573 mg, 8.81 mmol) and heated at 80°C for 4 h. The reaction mixture was cooled to room temperature and evaporated in vacuo. The residue obtained was diluted with DCM (20 mL) and filtered. The filtrate was concentrated in vacuo resulting in 900 mg of crude compound as a light yellow sticky solid. The crude compound was used in the next step without further purification. XH NMR (400 MHz, DMSO-i¾): δ = 8.05 (s, 1H), 5.76 (s, 2H), 4.50 - 4.40 (m, 4H), 3.81 (t, J= 5.2 Hz, 2H), 3.57 - 3.35 (m, 30H), 3.20 - 3.10 (m, 4H), 2.40-2.30 (m, 4H), 1.38 (s, 9H).
[00176] teri-Butyl 4-(2-((l-(23-(tosyloxy)-3,6,9,12,15,18,21-heptaoxatricosyl)-lH- l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (5):
[00177] A solution of tert-butyl 4-(2-((l-(23-hydroxy-3,6,9,12, 15, 18,21- heptaoxatricosyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (1.30 g, 1.96 mmol) in DCM (30 mL) was charged with Ag20 (1.36 g, 5.88 mmol), KI (163 mg, 0.980 mmol), tosyl chloride (820 mg, 4.32 mmol) and stirred at room temperature for 1 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 3% methanol in DCM to obtain 1.20 g, 73% yield of the title compound as light yellow sticky solid. XH NMR (400 MHz, DMSO-i/6): δ = 8.10 (s, 1H), 7.78 (t, J= 8.4 Hz, 2H), 7.48 (d, J = 7.9 Hz, 2H), 4.50 (d, J= A J Hz, 4H), 4.1 1 (dt, J= 5.9, 1.7 Hz, 2H), 3.80 (t, J= 5.1 Hz, 3H), 3.68 - 3.41 (m, 32H), 3.35 - 3.23 (m, 5H), 2.42 (s, 3H), 1.38 (s, 9H).
[00178] tert-Butyl 4-(2-((l-(23-hydroxy-3,6,9,12,15,18,21-heptaoxatricosyl)-lH-l,2,3- triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (4):
[00179] A solution of tert-butyl 4-(2-(prop-2-yn- 1 -yloxy)ethyl)piperazine- 1 -carboxylate (978 mg, 3.72 mmol) in ?-BuOH:H20 (20:20 mL) under argon atmosphere was charged with 23-azido-3, 6,9, 12, 15, 18,21-heptaoxatricosan-l-ol (1.62 g, 4.09 mmol), Cul (353 mg, 1.86 mmol) and stirred at room temperature for 2h. The reaction mixture was filtered through a pad of celite. The filtrate was concentrated in vacuo and the residue obtained was diluted with water (20 mL) and extracted with DCM (3 X 50 mL). The combined organic layers were dried
over anhydrous a2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-2% methanol in DCMto give 1.30 g, 48% yield of the title compound as colorless thick oil. XH NMR (400 MHz, DMSO-i¾): δ = 8.10 (s, 1H), 4.61 - 4.47 (m, 4H), 3.85 - 3.78(m, 2H), 3.58 - 3.37 (m, 30H), 3.33 - 3.30 (m, 3H), 3.25 -3.18 (m, 4H), 2.38 - 2.25 (m, 4H), 1.38 (s, 9H).
[00180] 23-Azido-3,6,9,12,15,18,21-heptaoxatricosan-l-ol (3):
[00181] A solution of 23-hydroxy-3,6,9, 12, 15, 18,21-heptaoxatricosyl 4- methylbenzenesulfonate (2.80 g, 5.34 mmol) in EtOH (50 mL) was charged with a 3 (2.08 g, 32.06 mmol) and heated at 80°C for 3 h. The reaction mixture was cooled to room temperature and evaporated in vacuo. The residue obtained was diluted with DCM (50 mL) and filtered.
The filtrate was concentrated in vacuo resulting in 2.80 g of the crude compound as colorless oil. The crude compound was used in the next step without further purification. XH NMR (400
MHz, CDC13): δ = 3.75 - 3.57 (m, 30H), 3.38 (t, J= 5.1 Hz, 2H).
[00182] 23-Hydroxy-3,6,9,12,15,18,21-heptaoxatricosyl 4-methylbenzenesulfonate
(2):
[00183] A solution of octaethylene glycol (3.10 g, 8.38 mmol) in DCM (50 mL) were charged with Ag20 (2.90 g, 12.56 mmol), KI (275 mg, 1.66 mmol) and stirred at room temperature for 15 min. This solution was charged with tosyl chloride (1.75 g, 9.22 mmol) and stirred at room temperature for 2h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 2% methanol in DCM to give 2.80 g, 63% yield of the title compound as a colorless oil. XH NMR (400 MHz, CDC13): δ = 7.79 (d, J= 8.2 Hz, 2H), 7.34 (d, J= 8.0 Hz, 2H), 4.19 - 4.12 (m, 4H), 3.76 - 3.55 (m, 28H), 2.44 (s, 3H).
-Butyl 4-(2-(prop-2-yn-l-yloxy)ethyl)piperazine-l-carboxylate (C):
[00186] A solution of tert-butyl 4-(2-hydroxyethyl)piperazine-l-carboxylate (5.0 g, 21.74 mmol) in THF (100 mL) at 0 °C was charged with NaH (1.56 g, 65.21 mmol) and stirred at same temperature for 10 min. The solution was charged with 3-bromoprop-l-yne (5.17 g, 43.48 mmol) and refluxed for 3h. The reaction mixture was cooled to room temperature and concentrated in vacuo. The residue obtained was diluted with water (50 mL) and extracted with ethyl acetate (2 X 100 mL) and the combined organic layers were dried over anhydrous a2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 2% methanol in DCM to give 2.50 g, 41% yield of the title compound as a yellow solid. XH NMR (400 MHz, DMSO-i¾): δ = 4.12 (d, J= 2.2 Hz, 2H), 3.54 (td, J= 5.8, 1.4 Hz, 2H), 3.35 - 3.24 (m, 5H), 2.52 - 2.45 (m, 2H), 2.40 - 2.34 (m, 4H), 1.39 (s, 9H).
[00188] A solution of tert-butyl piperazine-l-carboxylate (10 g, 53.76 mmol) in ACN (100 mL) was charged with potassium carbonate (37.09 g, 268.8 mmol) and 2-bromoethanol (8.06 g, 64.51 mmol). The resulting solution was heated at 80°C for 16 h. The reaction mixture was cooled to room temperature and concentrated in vacuo. The residue obtained was diluted with water (100 mL) and extracted with ethyl acetate (3 X 100 mL) and the combined organic layers were dried over anhydrous a2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 2% methanol in DCM to afford 6.50 g, 53% yield of the title compound as a yellow solid. XH NMR (400 MHz, CDC13): δ = 3.62 (t, J= 5.4 Hz, 2H), 3.50 - 3.43 (m, 4H), 2.55 (t, J= 5.4 Hz, 2H), 2.51 - 2.45 (m, 4H), 1.41 (s, 9H).
[00189] Synthetic scheme; Example 2
References:
a) JOC, 2011 , 76, 6832-6836; b) JOC, 2010, 75, 7002-7005; c) JACS, 2012, 134, 9285-9286
[00190] 7V-(2-Chloro-6-methylphenyl)-2-((2-methyl-6-(4-(2-((l-(l-oxo-l-(3-(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)phenyl)-5,8,ll5l4,17,20,23,26,29- nonaoxa-2-azahentriacontan-31-yl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l- yl)pyrimidin-4-yl)amino)thiazole-5-carboxamide [Example 2] :
[00191] A solution of 3-(6-((4-(trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)benzoic acid (18 mg, 0.049 mmol) in DMF (2 mL) was charged with (2-((6-(4-(2-((l-(29-amino- 3,6,9, 12, 15, 18,21, 24,27-nonaoxanonacosyl)-2,3-dihydro-lH-l,2,3-triazol-4- yl)methoxy)ethyl)piperazin- 1 -yl)-2-methylpyrimidin-4-yl)amino)-N-(2,6- dichlorophenyl)thiazole-5-carboxamide (50 mg, 0.049 mmol), HATU (28 mg, 0.074 mmol) and DIPEA (15 mg, 0.124 mmol). The resulting solution was stirred at room temperature for 1 h and the reaction mixture was partitioned between ethyl acetate (10 mL) and water (5 mL) and separated. The aqueous layer was extracted with ethyl acetate (3 X 10 mL) and the combined organic layers were dried over anhydrous Na2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by preparative HPLC to afford 35 mg, 53% yield of the title compound as a light brown sticky solid. XH NMR (400 MHz, DMSO-i¾): δ = 10.00 (s, 1H), 9.88 (s, 1H), 8.80 - 8.69 (m, 2H), 8.54 (t, J= 1.8 Hz, 1H), 8.30 (s, 1H), 8.24 - 8.16 (m, 2H), 8.05 (s, lH), 7.99 (d, J= 8.0 Hz, 1H), 7.90 - 7.82 (m, 2H), 7.63 (t, J= 7.7 Hz, 1H), 7.42 - 7.32 (m, 4H), 7.32 - 7.21 (m, 2H), 6.04 (s, 1H), 4.51 (s, 1H), 3.81 (t, J= 5.2 Hz, 2H), 3.62 - 3.41 (m, 44H), 2.47 (s, 3H), 2.40 (s, 3H); MS (ES+): m/z = 684.45, 685.65 [M/2+H]+; LCMS: ¾ = 2.27 min.
[00192] 2-((6-(4-(2-((l-(29-Amino-3,6,9,12,15,18,21,24,27-nonaoxanonacosyl)-lH- l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2-methylpyrimidin-4-yl)amino)-iV-(2-
[00193] A solution of 2-((6-(4-(2-((l-(29-azido-3,6,9, 12, 15, 18,21,24,27- nonaoxanonacosyl)-2,3-dihydro-lH- 1,2,3 -triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2- methylpyrimidin-4-yl)amino)-N-(2,6-dichlorophenyl)thiazole-5-carboxamide (100 mg, 0.095 mmol) in THF:H20 (6:3 mL) mixture was charged with TPP (149 mg, 0.570 mmol) and stirred at room temperature for 12 h. The reaction mixture was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 3% methanol in DCM to obtain 50 mg, 51% yield of the title compound as an off white solid. XH NMR (400
MHz, DMSO-i¾): δ = 9.90 (bs, 1H), 8.22 (s, 1H), 8.15 (s, 1H), 7.42-7.23 (m, 3H), 6.13 (s, 1H), 6.95 (s, 2H), 5.79 (s, lH), 4.57 - 4.45 (m, 4H), 3.81 - 3.78 (m, 3H), 3.56 - 3.42 (m, 35H), 2.80 (t, J= 5.5 Hz, 3H), 2.41 (s, 3H), 2.40 (s, 3H).
[00194] 2-((6-(4-(2-((l-(29-Azido-3,6,9,12,15,18,21,24,27-nonaoxanonacosyl)-lH- l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2-methylpyrimidin-4-yl)amino)-iV-(2-
[00195] A solution of l-(2-((l-(29-azido-3,6,9, 12, 15,18,21,24,27-nonaoxanonacosyl)-
2,3-dihydro-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine (200 mg, 0.295 mmol) in DMF (4 mL) was charged with potassium carbonate (245 mg, 1.77 mmol) and 2-((6-chloro-2- methylpyrimidin-4-yl)amino)-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide (70 mg, 0.17 mmol). The resulting solution was heated at 80°C for 16 h. The reaction mixture was cooled to room temperature and concentrated in vacuo. The residue obtained was diluted with water (5 mL) and extracted with ethyl acetate (3 X 10 mL). The combined organic layers were dried over anhydrous Na2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by silica gel column chromatography eluting with 1-2% methanol in CHCI3 to afford 100 mg, 32% yield of the title compound as an off white solid. XH NMR (400 MHz, DMSO-i¾: δ = 1 1.50 (s, lH), 9.85 (s, lH), 8.24 (s, lH), 8.15 (s,lH), 7.44 - 7.25 (m,2H), 6.13 (s, lH), 5.80 (s, lH), 4.56 - 4.48 (m, 4H), 3.81 (t, J= 5.2 Hz, 2H), 3.60 - 3.44 (m, 35H), 3.30 - 3.20 (m, 3H), 2.47 (s, 3H), 2.40 (s, 3H)
[00197] A solution of tert-butyl 4-(2-((l-(29-azido-3,6,9, 12, 15, 18,21,24,27- nonaoxanonacosyl)-2,3 -dihydro- 1H- 1 ,2,3 -triazol-4-yl)methoxy)ethyl)piperazine- 1 -carboxylate (230 mg, 0.296 mmol) in dioxane (2 mL) was charged with 4M HQ in dioxane (3 mL) at 0 °C and stirred at room temperature for 2 h. The reaction mixture was triturated with diethyl ether (5 mL) and concentrated in vacuo resulting in 200 mg of crude compound as an off white solid. The crude compound was used in the next step without further purification. XH NMR (400
MHz, DMSO-i/6): δ = 9.38 (s, 1H), 8.16 (s, 1H), 3.85 (s, 1H), 3.82 (t, J= 5.3 Hz, 2H), 3.82 - 3.75 (m, 4H), 3.60 - 3.31 (m, 48H).
[00198] teri-Butyl 4-(2-((l-(29-azido-3,6,9,12,15,18,21,24,27-nonaoxanonacosyl)-lH- l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (6):
[00199] A solution of tert-butyl 4-(2-((l-(29-(tosyloxy)-3,6,9, 12,15, 18,21,24,27- nonaoxanonacosyl)-2,3 -dihydro- 1Η- 1 ,2,3 -triazol-4-yl)methoxy)ethyl)piperazine- 1 -carboxylate (320 mg, 0.353 mmol) in EtOH (10 mL) was charged with a 3 (137 mg, 2.11 mmol) and heated at 80 °C for 15 h. The reaction mixture was cooled to room temperature and evaporated in vacuo. The residue obtained was diluted with DCM (20 mL) and filtered. The filtrate was concentrated in vacuo resulting in a crude compound 230 mg as a light yellow sticky solid. The crude compound was used in the next step without further purification. XH NMR (400 MHz, DMSO-i¾): δ = 8.10(s, 1H), 4.50 (d, J= 5.5 Hz, 4H), 3.81 (t, J= 5.2 Hz, 2H), 3.63 - 3.35 (m, 39H), 3.30 - 3.25(m, 4H), 2.50 - 2.25 (m, 4H), 1.38 (s, 9H).
[00200] teri-Butyl 4-(2-((l-(29-(tosyloxy)-3,6,9,12,15,18,21,24,27-nonaoxanonacosyl)-
2,3-dihydro-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (5):
[00201] A solution of tert-butyl 4-(2-((l-(29-hydroxy-3,6,9, 12, 15, 18,21,24,27- nonaoxanonacosyl)- 1H- 1 ,2,3-triazol-4-yl)methoxy)ethyl)piperazine- 1 -carboxylate (350 mg, 0.466 mmol) in DCM (10 mL) were charged with Ag20 (323 mg, 1.40 mmol), KI (31 mg, 0.186 mmol), tosyl chloride (195 mg, 1.03 mmol) and heated at 40°C for 12 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-3% methanol in DCM to give 320 mg, 75% yield of the title compound as colorless thick oil. XH NMR (400 MHz, DMSO-i¾): δ = 8.00 (s, 1H), 7.78 (d, J= 8.2 Hz, 2H), 7.48 (dd, J= 7.9, 5.3 Hz, 2H), 7.20 - 7.10(m, lH),4.51 - 4.45 (m, 5H), 4.14 - 4.07 (m, 2H), 3.81 (t, J= 5.2 Hz, 2H), 3.60 - 3.40 (m, 38H), 3.33 - 3.25 (m, 4H), 2.45 - 2.40 (m, 4H), 1.39 (s, 9H).
[00202] teri-Butyl 4-(2-((l-(29-hydroxy-3,6,9,12,15,18,21,24,27-nonaoxanonacosyl)- lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (4):
[00203] A solution of tert-butyl 4-(2-(prop-2-yn- 1 -yloxy)ethyl)piperazine- 1 -carboxylate
(200 mg, 0.760 mmol) in 1 : 1 ?-BuOH:H20 (14 mL) under argon atmosphere was charged with 29-azido-3,6,9, 12, 15, 18,21,24,27-nonaoxanonacosan-l-ol (404 mg, 0.836 mmol), Cul (28 mg, 0.147 mmol) and stirred at room temperature for 2 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo and the residue obtained was diluted with water (5 mL) and extracted with DCM (3 X 20 mL). The combined organic layers were dried over anhydrous a2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-2% methanol in DCM to give 350 mg, 61% yield of the title compound as colorless thick oil. XH NMR (400 MHz, DMSO-i¾): δ = 8.01 (s, 1H), 4.59 - 4.47 (m, 4H), 3.81 (t, J= 5.2 Hz, 2H), 3.58 - 3.37 (m, 38H), 3.27 (t, J= 4.9 Hz, 2H), 2.50 - 2.40 (m, 4H), 2.33 (t, J= 4.9 Hz, 4H), 1.39 (s, 9H).
[00204] 29-Azido-3,6,9,12,15,18,21,24,27-nonaoxanonacosan-l-ol (3): [00205] A solution of 29-hydroxy-3,6,9, 12,15, 18,21,24,27-nonaoxanonacosyl benzenesulfonate (1.10 g, 1.79 mmol) in EtOH (20 mL) was charged with a 3 (700 mg, 10.78 mmol) and heated at 80 °C for 3 h. The reaction mixture was cooled to room
temperature and evaporated in vacuo. The residue obtained was diluted with DCM (20 mL) and filtered. The filtrate was concentrated in vacuo resulting in 780 mg of a crude compound as an off white solid. The crude compound was used in the next step without further purification. 'H NMR (400 MHz, CDC13): δ = 3.76 - 3.58 (m, 40H), 3.39 (t, J= 5.1 Hz, 1H).
[00206] 29-Hydroxy-3,6,9,12,15,18,21,24,27-nonaoxanonacosyl 4- methylbenzenesulfonate (2): [00207] A solution of decaethylene glycol (1.0 g, 2.18 mmol) in DCM (10 mL) were charged with Ag20 (759 mg, 3.27 mmol), KI (72 mg, 0.436 mmol), tosyl chloride (456 mg, 2.40 mmol) and stirred at room temperature for 3 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 3% methanol in DCM to give 1.10 g, 83% yield of the title compound as an off white solid. XH NMR (400 MHz, CDC13): δ = 7.40 - 7.70 (m, 4H), 3.74 - 3.60 (m, 24H), 3.40 - 3.30 (m, 16H), 2.34 (s, 3H).
[00208] Synthetic scheme; Example 3
[00209] 7V-(2-Chloro-6-methylphenyl)-2-((2-methyl-6-(4-(2-((l-(l-oxo-l-(3-(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)phenyl)-5,8,ll, 14,17,20,23,26,29,32,35- undecaoxa-2-azaheptatriacontan-37-yl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l- yl)pyrimidin-4-yl)amino)thiazole-5-carboxamide [Example 3] :
[00210] A solution of 3-(6-((4-(trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)benzoic acid (47 mg, 0.125 mmol) in DMF (2 mL) was charged with 2-((6-(4-(2-((l-(35-amino- 3 ,6,9, 12, 15, 18,21 ,24,27,30,33 -undecaoxapentatriacontyl)- 1H- 1 ,2,3-triazol-4- yl)methoxy)ethyl)piperazin-l-yl)-2-methylpyrimidin-4-yl)amino)-N-(2-chloro-6- methylphenyl)thiazole-5-carboxamide (125 mg, 0.1 14 mmol), HATU (65 mg, 0.171 mmol) and DIPEA (36 mg, 0.285 mmol) and stirred at room temperature for lh. The reaction mixture was partitioned between ethyl acetate (10 mL) and water (5 mL) and separated. The aqueous layer was extracted with ethyl acetate (3 X 10 mL) and the combined organic layers were dried over anhydrous Na2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by preparative HPLC to give 20 mg, 12% yield of the title compound as an off white solid. ¾ NMR (400 MHz, DMSO-i¾): δ = 1 1.47 (bs, 1H), 9.94 (s, 1H), 9.87 (s, 1H), 8.76 (s, 1H), 8.74 - 8.64 (m, 1H), 8.53 (s, 1H), 8.24 - 8.14 (m, 3H), 8.05 (s, 1H), 7.99 (d, J = 6.8 Hz, 1H), 7.86 (d, J= 8.4 Hz, 1H ), 7.63 (t, J= 7.7 Hz, 1H), 7.43 - 7.21 (m, 5H), 6.04 (s, 1H), 4.52 (s, 4H), 3.81 (t, J= 5.0 Hz, 2H), 3.62 - 3.44 (m, 56H), 2.40 (s, 3H), 2.23 (s, 3H); MS (ES+): m/z = 728.15, 730.45 [M/2+H] +; LCMS: tR = 2.32 min.
[00211] 2-((6-(4-(2-((l-(35-Amino-3,6,9,12,15,18,21,24,27,30,33- undecaoxapentatriacontyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2-
[00212] A solution of 2-((6-(4-(2-((l-(35-azido-3,6,9, 12,15, 18,21,24,27,30,33- undecaoxapentatriacontyl)- 1H- 1 ,2,3-triazol-4-yl)methoxy)ethyl)piperazin- 1 -yl)-2- methylpyrimidin-4-yl)amino)-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide (240 mg, 0.213 mmol) in THF:H20 3:2 (5 mL) mixture was charged with TPP (224 mg, 0.855 mmol) and stirred at room temperature for 12 h. The reaction mixture was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 3% methanol in DCM to give 125 mg, 53% yield of the title compound as an off white solid. XH NMR (400 MHz, DMSO-i¾): δ = 8.28 (d, J= 10.5 Hz, 1H), 8.07 (s, 1H), 7.40 (d, J= 7.5 Hz, 1H), 7.33 - 7.21 (m, 2H), 6.04 (s, 1H), 4.57 (dd, 2H), 4.22 (d, J= 6.3 Hz, 2H), 4.09 (s, 2H), 3.90 - 3.85 (m, 4H), 3.76 - 3.60 (m, 4H), 3.61 - 3.52 (m, 48H), 3.17 (t, J= 3.7 Hz, 2H), 2.65 (dd, J= 1 1.5, 5.7 Hz, 2H), 2.24 (s, 6H).
[00213] 2-((6-(4-(2-((l-(35-Azido-3,6,9,12,15,18,21,24,27,30,33- undecaoxapentatriacontyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazin-l-yl)-2-
[00214] A solution of l-(2-((l-(35-azido-3,6,9, 12,15,18,21,24,27,30,33- undecaoxapentatriacontyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine (450 mg, 0.589 mmol) in DMF (5 mL) was charged with potassium carbonate (487 mg, 3.53 mmol) and 2-((6- chloro-2-methylpyrimidin-4-yl)amino)-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide (116 mg, 0.294 mmol). The resulting solution was heated at 80°C for 16 h then cooled to room temperature and concentrated in vacuo. The residue obtained was diluted with water (5 mL) and extracted with ethyl acetate (3 X 10 mL) and the combined organic layers were dried over anhydrous a2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-2% methanol in DCM to give 240 mg, 36% yield of the title compound as a yellow solid. XH NMR (400 MHz, DMSO-i¾): δ = 8.12 (s, 1H), 7.29 (t, J= 5.6 Hz, 1H), 5.76 (s, 2H), 4.52 (d, J= 6.2 Hz, 4H), 3.82 (t, J= 5.3 Hz, 3H), 3.63 - 3.24 (m, 31H), 2.54 (s, 9H), 2.44 (d, J= 29.0 Hz, 22H), 2.24 (s, 3H).
[00216] A solution of tert-butyl 4-(2-((l-(35-azido-3,6,9, 12, 15, 18,21,24,27,30,33- undecaoxapentatriacontyl)- 1H- 1 ,2,3-triazol-4-yl)methoxy)ethyl)piperazine- 1 -carboxylate (800 mg, 0.925 mmol) in dioxane (5 mL) was cooled to 0 °C and charged with 4M HQ in dioxane (3 mL) then stirred at room temperature for 2 h. The reaction mixture was triturated with diethyl ether (5 mL) and concentrated in vacuo resulting in 700 mg of crude compound as an off white solid. The crude compound was used in the next step without further purification. XH NMR (400 MHz, DMSO-i¾): δ = 8.15 (s, 1H), 4.60 (s, 2H), 4.52 (t, J= 5.2 Hz, 2H), 3.83 (q, J = 5.5 Hz, 5H), 3.63 - 3.46 (m, 46H), 3.50 - 3.40 (m, 8H).
[00217] teri-Butyl 4-(2-((l-(35-azido-3,6,9,12,15,18,21,24,27,30,33- undecaoxapentatriacontyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate
[00218] A solution of tert-butyl 4-(2-((l-(35-(tosyloxy)-3,6,9, 12, 15,18,21,24,27,30,33- undecaoxapentatriacontyl)-lH-l, 2, 3-triazol-4-yl)methoxy)ethyl)piperazine-l -carboxylate (1.0 g, 1.01 mmol) in EtOH (10 mL) was charged with a 3 (392 mg, 6.04 mmol) and heated at 80 °C for 4 h. The reaction mixture was cooled to room temperature and evaporated in vacuo. The crude was diluted with DCM (20 mL) and filtered. The filtrate was concentrated in vacuo resulting in 800 mg of crude compound as a light yellow sticky solid. The crude compound was used in the next step without further purification. XH NMR (400 MHz, DMSO-i¾): δ = 8.10(s, 1H), 4.50 - 4.45 (m, 4H), 3.81 (t, J= 5.2 Hz, 3H), 3.63 - 3.46 (m, 41H), 3.39 (t, J= 4.9 Hz, 3H), 3.27 - 3.20 (m, 4H), 2.47 - 2.43 (m, 6H), 1.38 (s, 9H).
[00219] teri-Butyl 4-(2-((l-(35-(tosyloxy)-3,6,9,12,15,18,21,24,27,30,33- undecaoxapentatriacontyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate
[00220] A solution of tert-butyl 4-(2-((l-(35-hydroxy-3,6,9, 12, 15, 18,21,24,27,30,33- undecaoxapentatriacontyl)- 1H- 1 ,2,3-triazol-4-yl)methoxy)ethyl)piperazine- 1 -carboxylate (1.20 g, 1.43 mmol) in DCM (30 mL) was charged with Ag20 (990 mg, 4.29 mmol), KI (94 mg, 0.566 mmol), tosyl chloride (597 mg, 3.14 mmol) and heated at 40 °C for 12 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-3% methanol in DCM to obtain 1.0 g, 70% yield of the title compound as light yellow sticky solid. XH NMR (400 MHz, CDC13): δ = 7.80 (d, J= 8.0 Hz, 2H), 7.55 (s, lH), 7.34 (d, J= 8.0 Hz, 2H), 4.65 (s, 2H), 4.54 (t, J= 5.1 Hz, 2H), 4.16 (t, J= 4.9 Hz, 2H), 3.87 (t, J= 5.1 Hz, 2H), 3.64 - 3.43 (m, 40H), 3.40 - 3.35 (m, 4H), 2.60 - 2.58 (m, 2H), 2.44 - 2.35 (m, 4H), 1.45 (s, 9H).
[00221] teri-Butyl 4-(2-((l-(35-hydroxy-3,6,9,12,15,18,21,24,27,30,33- undecaoxapentatriacontyl)-lH-l,2,3-triazol-4-yl)methoxy)ethyl)piperazine-l-carboxylate (4):
[00222] A solution of tert-butyl 4-(2-(prop-2-yn- 1 -yloxy)ethyl)piperazine- 1 -carboxylate
(500 mg, 1.90 mmol) in ?-BuOH : H20 (1: 1) (40 mL) under argon atmosphere was charged with 35-azido-3,6,9,12,15,18,21,24,27,30,33-undecaoxapentatriacontan-l-ol (1.17 g, 2.05 mmol), Cul (180 mg, 0.947 mmol) and stirred at room temperature for 2 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo. The crude residue was diluted with water (20 mL) and extracted with DCM (3 X 50 mL). The combined organic layers were dried over anhydrous Na2S04, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-2% methanol in DCM to give 1.20 g, 70% yield of the title compound as colorless thick oil. XH NMR (400 MHz, DMSO-i¾): 5 = 8.10 (s, 1H), 5.75 (s, 1H), 4.53 - 4.48 (m, 5H), 3.81 (t, J= 5.2 Hz, 2H), 3.54 - 3.37 (m, 44H), 3.29 - 3.20 (m, 6H), 2.48 - 2.40 (m, 4H), 1.39 (s, 9H).
[00223] 35-Azido-3,6,9,12,15,18,21,24,27,30,33-undecaoxapentatriacontan-l-ol (3): [00224] A solution of 35-hydroxy-3, 6,9,12, 15, 18,21,24,27,30,33- undecaoxapentatriacontyl 4-methylbenzenesulfonate (1.90 g, 2.71 mmol) in EtOH (20 mL) was charged with a 3 (882 mg, 13.57 mmol) and heated at 80 °C for 3 h. The reaction mixture was cooled to room temperature and evaporated in vacuo then diluted with DCM (20 mL) and filtered. The filtrate was concentrated in vacuo resulting in 1.37 g of crude compound as colorless oil. The crude compound was used in the next step without further purification. XH NMR (400 MHz, CDC13): δ = 3.76 - 3.56 (m, 48H), 3.39 (t, J= 5.0 Hz, 1H).
[00225] 35-Hydroxy-3,6,9,12,15,18,21,24,27,30,33-undecaoxapentatriacontyl 4- methylbenzenesulfonate (2): [00226] A solution of dodcaethylene glycol (3.50 g, 6.41 mmol) in DCM (35 mL) was charged with Ag20 (2.21 g, 9.60 mmol), KI (212 mg, 1.27 mmol), tosyl chloride (1.34 g, 7.05 mmol) and stirred at room temperature for 3h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel eluting with 1-3% methanol in DCM to give 1.97 g, 44% yield of the title compound as colorless oil. XH NMR (400 MHz, CDC13): δ = 7.83 - 7.76 (m, 2H), 7.34 (d, J= 8.0 Hz, 2H), 4.16 (td, J= 4.8, 2.3 Hz, 2H), 3.77 - 3.56 (m, 46H), 2.45 (s, 3H).
Synthetic scheme for Example 4;
[00227] 4-Methyl-7V-(4-((4-methylpiperazin-l-yl)methyl)-3- (trifluoromethyl)phenyl)-3-((7-((l-oxo-l-(4-(l,l,3,3-tetramethyl-3-(4-((2-(2-(2-(3-(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4- yl)benzamido)ethoxy)ethoxy)ethyl)carbamoyl)phenyl)disiloxanyl)phenyl)- 5,8,ll914,17,20,23,26,29-nonaoxa-2-azahentriacontan-31-yl)oxy)imidazo[l,2-a]pyridin-3- yl)ethynyl)benzamide [Example 4] :
[00228] A solution of 4,4'-(l,l,3,3-tetramethyldisiloxane-l,3-diyl)dibenzoic acid (75 mg,
0.20 mmol) in DCM (7.5 mL) was charged with potassium carbonate (70 mg, 0.50 mmol), HBTU (166 mg, 0.44 mmol) and stirred at room temperature for 30 min. The resulting solution was charged with 3-((7-((29-amino-3,6,9, 12, 15, 18,21,24,27- nonaoxanonacosyl)oxy)imidazo[l,2-a]pyridin-3-yl)ethynyl)-4-methyl-N-(4-((4- methylpiperazin-l-yl)methyl)-3-(trifluoromethyl)phenyl)benzamide (197 mg, 0.20 mmol) and N-(2-(2-(2-aminoethoxy)ethoxy)ethyl)-3-(6-((4-(trifluoromethoxy)phenyl)amino)pyrimidin-4- yl)benzamide (101 mg, 0.20 mmol) and stirred at room temperature for 16 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in the crude compound which was purified by chromatography on silica gel eluting with 5-10 % methanolic ammonia in DCM and repurified by preparative TLC (10% MeOH in DCM) to give 25 mg, 7% yield, of the title compound as light brown solid. ¾ NMR (400 MHz, DMSO- d6): δ = 10.54 (s, 1H), 9.94 (s, 1H), 8.76 (s, 1H), 8.70 (t, J= 5.48 Hz, 1H), 8.47 - 8.54 (m, 3H), 8.44 (d, J= 7.45 Hz, 2H), 8.23 (dd, J= 1.75, 7.02 Hz, 2H), 8.18 (d, J= 7.89 Hz, 1H), 8.08 (d, J = 8.77 Hz, 1H), 7.98 (d, J= 7.89 Hz, 1H), 7.89 - 7.93 (m, 2H), 7.85 (d, J= 9.21 Hz, 2H), 7.79 - 7.83 (m, 3H), 7.71 (d, J= 8.77 Hz, 1H), 7.62 (s, 1H), 7.59 (d, J= 7.02 Hz, 4H), 7.53 (d, J = 8.33 Hz, 1H), 7.32 - 7.38 (m, 3H), 7.15 (d, J= 2.19 Hz, 1H), 6.88 (dd, J= 2.19, 7.45 Hz, 1H), 4.19 - 4.25 (m, 2H), 3.76 - 3.81 (m, 2H), 3.37 - 3.62 (m, 50H), 2.59 (s, 3H), 2.22 - 2.46 (m, 1 1H), 0.31 (s, 12H); MS (ES+): m/z = 916.45 [M/2+H]+; LCMS: fR = 2.77 min.
'-(l,l,3,3-Tetramethyldisiloxane-l,3-diyl)dibenzoic acid (5):
[00230] A solution of dibenzyl 4,4'-(l, l,3,3-tetramethyldisiloxane-l,3-diyl)dibenzoate (11 g, 19.8 mmol) in ethyl acetate was charged with 10% Pd/C (2.20 g, 50% moist) in a 2 litre autoclave and stirred under hydrogen atmosphere at room temperature for 2 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in the crude compound which was stirred in diethyl ether, filtered and dried to give 6.70 g of the title compound as a white solid. The crude compound was used in the next step without further purification. XH NMR (400 MHz, CDC13): δ = 7.80 (d, J= 7.83 Hz, 4H), 7.36 (d, J = Ί.34 Hz, 4H), 0.40 (s, 12H).
[00232] A solution of benzyl 4-iodobenzoate (50 g, 147.8 mmol) in toluene (1500 mL) was charged with DIPEA (79 mL), 1, 1,3,3-tetramethyl disiloxane (9.9 g, 73.7 mmol) and purged with argon for 30 min. The resulting solution was charged with bis(tri-tert butyl phosphine)Pd (0) (3.8 g, 73.7 mmol) and stirred at room temperature for 14 h. The reaction mixture was filtered through a pad of celite, the filtrate was concentrated in vacuo and the residue was dissolved in DCM (500 mL) and washed with citric acid solution (250 mL) followed by saturated sodium bicarbonate solution (250 mL). The combined organic layers were dried over anhydrous Na2S04, filtered and concentrated in vacuo resulting in the crude compound which was purified by chromatography on silica gel eluting with 5-10 % ethyl acetate in w-hexane to give 13 g, 16% yield of the title compound as a white solid. XH NMR (400 MHz, CDC13): δ = 8.03 (d, J= 7.91 Hz, 4H), 7.59 (d, J= 7.91 Hz, 4H), 7.32 - 7.48 (m, 10H), 5.37 (s, 4H), 0.31 - 0.35 (m, 12H); MS (ES+): m/z = 572.20 [M+H]+; LCMS: tR = 3.46 min.
[00234] A solution of 4-iodobenzoic acid (50 g, 201.5 mmol) in DMF (500 mL) was charged with potassium carbonate (30.7 g, 222.4 mmol) and stirred at room temperature for 1 h. The resulting solution was charged with benzyl bromide (24 mL, 201.5 mmol) and stirred at room temperature for 12 h. The reaction mixture was diluted with ice water and the solid
precipitated out was filtered and washed with w-hexane to give 58 g of the title compound as a white solid. The crude compound was used in the next step without further purification. XH NMR (400 MHz, CDC13): 5 = 7.71 - 7.89 (m, 4H), 7.33 - 7.52 (m, 5H), 5.35 (br. s, 2H).
[00236] 4,4'-(l,l,3,3-Tetramethyldisiloxane-l,3-diyl)bis(7V-(2-(2-(2-(3-(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4- yl)benzamido)ethoxy)ethoxy)ethyl)benzamide) [Example 5] :
[00237] A solution of 4,4'-(l,l,3,3-tetramethyldisiloxane-l,3-diyl)dibenzoic acid (150 mg, 0.40 mmol) in DCM (15 mL) was charged with potassium carbonate (138 mg, 1.00 mmol), HBTU (333 mg, 0.88 mmol) and stirred at room temperature for 30 min. The resulting solution was charged with N-(2-(2-(2-aminoethoxy)ethoxy)ethyl)-3-(6-((4-
(trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)benzamide (202 mg, 0.40 mmol) and 3-((7-(2- (2-(2-(2-aminoethoxy)ethoxy)ethoxy)ethoxy)imidazo[l,2-a]pyridin-3-yl)ethynyl)-4-methyl-N-
(4-((4-methylpiperazin-l-yl)methyl)-3-(trifluoromethyl)phenyl)benzamide (289 mg, 0.40 mmol) and stirred at room temperature for 16 h. The reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo resulting in the crude compound which was purified by chromatography on silica gel eluting with 5-10% methanolic ammonia in DCM and repuriifed by preparative TLC (10% MeOH in DCM) to give 75 mg, 19% yield, of the Example 5 as a brown solid and 60 mg, 9% yield, of Example 6 as an off-white solid.
[00238] Example 5 : XH NMR (400 MHz, DMSO-i¾): δ = 9.92 (s, 2H), 8.76 (s, 2H),
8.70 (t, J= 5.38 Hz, 2H), 8.46 - 8.54 (m, 4H), 8.18 (d, J= 7.49 Hz, 2H), 7.98 (d, J= 7.49 Hz, 2H), 7.83 (dd, J= 8.43, 15.92 Hz, 8H), 7.55 - 7.66 (m, 6H), 7.30 - 7.40 (m, 6H), 3.50 - 3.62 (m, 16H), 3.42 (qd, J= 5.71, 17.49 Hz, 8H), 0.30 (s, 12H) ; MS (ES+): m/z = 675.32 [M/2+H]+; LCMS: ¾ = 3.69 min.
[00239] 4-Methyl-7V-(4-((4-methylpiperazin-l-yl)methyl)-3- (trifluoromethyl)phenyl)-3-((7-((l-oxo-l-(4-(l,l,3,3-tetramethyl-3-(4-((2-(2-(2-(3-(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4- yl)benzamido)ethoxy)ethoxy)ethyl)carbamoyl)phenyl)disiloxanyl)phenyl)-5,8,ll-trioxa-2-
[00240] Example 6: XH NMR (400 MHz, DMSO-i¾): δ = 10.53 (s, 1H), 9.92 (s, 1H),
8.76 (s, 1H), 8.70 (br. s, 1H), 8.51 (d, J= 8.43 Hz, 3H), 8.43 (d, J= 7.02 Hz, 1H), 8.22 (d, J= 7.49 Hz, 2H), 8.18 (d, J= 7.49 Hz, 1 H), 8.07 (d, J= 8.43 Hz, 1H), 7.98 (d, J= 7.49 Hz, 1H), 7.91 (br. s, 2H), 7.77 - 7.88 (m, 6H), 7.71 (d, J= 8.43 Hz, 1H), 7.56 - 7.65 (m, 5H), 7.52 (d, J = 7.96 Hz, 1H), 7.31 - 7.38 (m, 3H), 7.14 (br. s, 1H), 6.87 (d, J= 6.55 Hz, 1H), 4.20 (br. s, 2H), 3.75 - 3.79 (m, 2H), 3.39 - 3.64 (m, 26H), 2.59 (s, 3H), 2.31 - 2.45 (m, 8H), 2.18 (br. s, 3H), 0.30 (s, 12H); MS (ES+): m/z = 783.84 [M/2+H]+; LCMS: fR = 3.10 min.
[00241] Example 7; Synthesis of Bivalent Compounds
[00242] Bivalent compounds are synthesized according to the procedures described below. Reagents are either purchased commercially or prepared according to previously described methods.
Scheme 1.
[00243] Example 8; Cell Proliferation Assays
[00244] K562 (purchased from ATCC: CCL-243) or BaF3 cells expressing either wild type BCR-Abl or mutant BCR-Abl (T3151) (obtained from Dr James Griffin Lab, Dana-Faber cancer Institute, Boston MA) were plated into 96 well plates (10,000 cells/well). Serial dilutions of compound starting at 10μΜ were added to the cells and the plates incubated at 37 °C for 72 hours at which point CellTitreGlo Reagent (Promega) was added to each plate and luminescence measured. Cell viability was determined relative to DMSO treated cells and GI50
values obtained after plotting dose-dependent effects using PRISM. Group A = GI50 range of 0.1-30 nM; Group B = GI50 range of 30-100 nM; Group C = GI50 range of 100 nM - 30 μΜ.
[00245] Table 1. K562 Data.
[00249] Kinase domain from wild type human Abll kinases, Ala46-Val515, were purchased from Cayman Chemical (Ann Arbor, MI), and were purified in the absence of detergent to allow GNF5 and analogues to bind in the myristoyl binding site. Abl kinase activity was measured in a semi-continuous fluorescence-based kinase assay (Omnia) utilizing a synthetic peptide Y6 which was purchased from Life Technologies (Carlsbad, CA). Assay was run in the presence of 20 mM HEPES pH 8.0, 50 mM NaCl, 10 mM MgC12, .05% BSA, 10 μΜ sodium vanadate, 200 μΜ DTT, 2% DMSO, and 10 μΜ Y6 peptide at protein concentrations of 25 nM for wild type Abll and 6 nM for the T3151 mutant Abll . The assay was performed at the ATP Km, which was determined to be 40 μΜ for the wild type Abll and 10 μΜ for the T3151 mutant Abl 1. Abl 1 inhibitors were titrated in 100% DMSO and then
diluted ten-fold into reaction buffer, which was followed by a subsequent ten-fold dilution into the assay plate. Inhibitors were incubated with the Abll kinases for 60 minutes before initiating reaction with the addition of aY6/ATP cocktail. The reaction was read for 1-3 hours at one read per minute on a Spectramax Paradigm plate reader (Molecular Devices), at excitation and emission wavelengths of 360 nm and 465 nm, respectively. Slopes (reaction rates) were calculated from the linear phase of progress curves generated from the Softmax 6.3 program (Molecular Devices). Slopes were then plotted as a function of inhibitor concentration to determine the IC50 values of the inhibitors tested. IC50S were generated through non-linear fitting of data with Prism (GraphPad). Group A = GI50 range of 0.1-30 nM; Group B = GI50 range of 30-100 nM; Group C = GI50 range of 100 nM - 30 μΜ.
[00250] Table 4. Omnia Wild-Type Data.
EQUIVALENTS
[00251] While specific embodiments have been discussed, the above specification is illustrative and not restrictive. Many variations will become apparent to those skilled in the art upon review of this specification. The full scope of the embodiments should be determined by reference to the claims, along with their full scope of equivalents, and the specification, along with such variations.
[00252] Unless otherwise indicated, all numbers expressing quantities of ingredients, reaction conditions, and so forth used in the specification and claims are to be understood as being modified in all instances by the term "about." Accordingly, unless indicated to the contrary, the numerical parameters set forth in this specification and attached claims are approximations that may vary depending upon the desired properties sought to be obtained.
Claims
What is claimed is;
or a pharmaceutically acceptable salt, stereoisomer, metabolite, or hydrate thereof;
wherein:
Q1 is a connecting moiety covalently bound to P1 and P2, wherein Q1 is selected from the group consisting of:
-W1-(C3-6alkylene-0)s-Ci_6alkylene-W1-; -W^CHz-CHz-OyCi-galkylene-W1-; -W1- (CH2-CH2-0)s-;
an optionally substituted unsaturated heteroaliphatic moiety; -Si(R7)(R8)-; -Si(R7)(R8)-0-; -0-Si(R7)(R8)-; -MR'-; -N(R')C(0)-; -C(0)N( ')-; - N(R')S02-; -S02 (R')-; -0-; -C(0)-; -OC(O)-; -C(0)0-; -S-; -SO-; -S02-; -C(-S ; -C(=NR ; aliphatic, heteroaliphatic, phenyl, naphthyl, heterocyclyl; heteroaryl, or a covalently bonded combination thereof;
wherein:
W1 is, independently for each occurrence, N, O, or S;
s is an integer from 1-10;
t is an integer from 3-10;
R7 and R8 are selected, independently for each occurrence, from the group consisting of -OH, Ci_6alkyl, -0-Ci_6alkyl, C2-6alkenyl, C3-6cycloalkyl, -Ci_6alkyl-NRaRb, phenyl and heteroaryl; wherein Ci_6alkyl, C2_6alkenyl, C3_6cycloalkyl, phenyl, and heteroaryl may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, hydroxyl, Ci-6alkyl, and phenyl; or
R7 and R8, together with the silicon to which they are attached, form a 4-7 membered heterocyclic ring, optionally containing one, two, three, or four heteroatoms independently selected from O, S, or N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, and hydroxyl; and
R' is, independently for each occurrence, selected from the group consisting of hydrogen and Ci-4alkyl; wherein
may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; and
wherein:
Q is:
(1 ) a 5-membered heteroaryl ring;
(2) a 6-membered hcteroaryl ring; or (3) an aryl ring; optionally substituted with one or more groups R! ;
U is:
(1) a single bond;
(2) l ?( :::::C! i I ΟΓ
(3) (' ( '! !.> ),„ , where m is 1 to 2:
T* and T2 are each hydrogen, or together form =0 or— S;
R1 is:
(1) hydrogen or R6, where R6 is aikyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, cycloalkenyl, cycioalkenylalkyi, aryl, aralkyl, heterocyclo, or heterocycloalkyl, each of which is unsubstituted or substituted with U1, Uz and one or more groups LI3;
(2) OH or OR6;
(3)— SH or— SR6;
(4)— C(0)2H,— C(0)qR6, or— -O— C(0)qR6;
(5) ----SO3H or --S(0)qR6;
(6) halo;
(7) cyano;
(8) nitro;
(9) U4 - R7RS;
(10)— U4— (R9)— U5— R K'RU;
(1 1 )— U4— N(R12)— U5— R6;
(12) --P(0)(OR6)2;
R2 is:
(1) R6, substituted with Q1;
(2)— U4— R6, substituted with Q1; or
(3)— U13— R7R8, substituted with Q1 ;
R3 is:
(1 ) hydrogen or R6;
(2) --U4--R6: or
(3 ) U13 NR7R8;
R4 and R5:
(1) are each independently hydrogen or R6;
(2) I..'1 M R" ) N R ; °R! 1 ;
(3) -N(R9)U4R6; or
(4) together with the nitrogen atom to which they are attached complete a 3- to 8-membered saturated or unsaturated heterocyclic ring which is unsubstituted or substituted with U1 , U2 and U3, which heterocyclic ring may optionally have fused to it a benzene ring itself unsubstituted or substituted with U1, U and UJ; R . R8, R9, R i0, R! and R12:
(1) are each independently hydrogen or R°;
(2) R'' and Rs may together be aikylene, alkenyiene or heteroalkyl, completing a 3- to 8-membered saturated or unsaturated ring with the nitrogen atom to which they are attached, which ring is unsubstituted or substituted with U1, U"' and U3; or
(3) any two of R9, R10, and R" may together be aikylene or alkenyiene completing a 3- to 8-membered saturated or unsaturated ring together with the nitrogen atoms to which they are attached, which ring is unsubstituted or substituted with U1, U2 and U3;
R i ! is:
(l ) cyano;
(2) nitro;
(3 ) NH2;
(4)— - HOalkyl;
( ? ) Oi l :
(6) - HOaryl;
(7)— NHCOOalkyl;
94 (8)— NHCOOaryl;
95 (9)— - HS02alkyl;
96 (10) N i iSO .aryl:
97 (l l) aryl;
98 (12) heteroaryl;
99 (13)— Oalkyl; or
100 (14) Oaryi:
101 R14 is:
102 (1)— N02;
103 (2) --COOalkyl; or
104 (3) COOaryl;
105 Ri5 is:
106 (! ) hydrogen;
107 (2) alkyi;
108 (3) aryl;
109 (4) ary!alkyl; or
1 10 (5) cycloalkyi;
1 11 U\ U2 and U3 are each independently:
1 12 (1 ) hydrogen or U6, where U6 is (i) alkyi, alkenyl, alkynyl, cycloalkyi,
1 13 cycloalkyialkyl, eycioalkenyl, eycioalkenylaikyi, aryl, aralkyl, aikyiaryl,
1 14 cycloalkylaryl, heterocycio, or heterocycloaikyl; (ii) a group (i) which is itself
1 15 substituted by one or more of the same or different groups (i); or (iii) a group (i)
1 16 or (ii) which is substituted by one or more of the following groups (2) to (16) of
1 17 the definition of U1 , U2 and U ;
1 18 (2) OH or OU6;
1 19 (3)— SH or— SU6;
120 (4)— C(0)qH,— C(0)qTJ6, or—O— C(0)qU6;
121 (5 ) S03H, S(0)qU6; or S(0)qN(U9)U6;
122 (6) halo;
123 (7) cyano;
124 (8) nitro;
125 (9) U -NU7U8;
126 (10)— U4— -N(U9)— U5— U7U8;
127 (1 1)— U4— (U10)— U5— U6;
128 (12)— —NiU10)— Us— H;
129 (13) oxo;
130 ( 14) O C(O) -U6;
131 (15) any two of U1, U2, and U3 may together be alkylene or alkersylene
132 completing a 3- to 8 -membered saturated or unsaturated ring together with the
133 atoms to which they are attached; or
134 (16) any two of U1, U2, and U3 may together be— O— (CH2)r— O— , where r is
135 1 to 5, completing a 4- to 8-membered saturated or unsaturated ring together
136 with the atoms to which they are attached:
137 U4 and U"1 are each independently :
138 (1 ) a single bond;
139 (2)— U11— S(0)q— U12— ;
140 (3 ) Un C(O) U12 ;
141 (4)— U11— C(S)— U12— ;
142 ( 5 ; I . " O I 12 :
143 (6)— U1 1— S— U1 2— ;
144 (7) U11 0 C(O) -U!2 ; or
145 (8)— U11— C(0)— O— Ui2— ;
146 U7, U8, U9 and U50i
147 (1) are each independently hydrogen or U6;
148 (2) U' and U8, or U°and IP0, may together be alkylene or alkenylene, completing
149 a 3- to 8-membered saturated or unsaturated ring together with the atoms to
150 which they are attached, which ring is unsubstituted or substituted with U1, U2
151 and U"'; or
152 (3) U' or U8, together with U9, may be alkylene or alkenylene completing a 3- to
153 8-membered saturated or unsaturated ring together with the nitrogen atoms to
154 which they are attached, which ring is unsubstituted or substituted with U\ U2
155 and lJ3;
156 Ul ! and ϋί2 are each independently;
157 (1) a single bond;
158 (2) alkylene;
159 (3) alkenylene; or
(4) alkyiiyierse; and
U° is:
(1) a single bond;
(2) !. : ; ; S(0)q U12 ;
(3)— U11— C(O)— Ui2— ;
(4) ....... Ui i--C(S)---Ui2--;
(5) Un O U12 ;
(6)— U11— S— U12— ;
(7)— U11— G— C(O)— Un— ;
(g) [ ; ; Ci () ; () 2_;
(9) CCNR13) ;
(10)— C(CHR14)— ; or
(1 1) ( '{ ('{ R ! ' ;.> ) : and
q is 1 or 2;
eutically acceptable salt thereof;
176 wherein:
177 LI1 and U2 are independently selected from the group consisting of -N= and -CR4=,
179 L is selected from the group consisting of a bond, -O- and - R5 -, wherein R3 is
180 hydrogen or Q ^alkyl;
181 R is selected from the group consisting of -U NR 'R', -U OR' and -U'R' , wherein U" is
183 of Ce-ioa yl and Cs-eheteroaryl; wherein any aryl or heteroaryi is optionally substituted with 1 to
184 3 radicals independently selected from the group consisting of halo, amino, Q^alkyi, halo-
186 R2 is selected from the group consisting ofhydrogen, halo, amino, Chalky], halo-
188 R3 is selected from the group consisting of Ca-sheterocycloalkyl-CiMalkyl, C$.
189 loheteroaryl-Cc alkyl and
wherein any alkyl group is optionally substituted
190 with 1 to 3 radicals selected from the group consisting of hydroxy, halo and amino; and any
191 aryl, heteroaiyl or heterocycioalkyl is optionally substituted with 1 to 3 radicals independently
193 Ci-ealkyl, Ci-4alkoxy, halo-substituted Ci-4alkoxy, phenyl, C3-gheterocyclQalkyl, -
194 U3C(0)NR8R8, -U3C(0)NR8R9, ~U C(0)R9, -U S(0) R8R8, -U3NR8R9, -U3NR8R8, -
195 U3S(Q)2NR8R8, -U S(G)2R8, -U3S(0)2R9,
196 -U SNR8R8, -U3OMR8R8, -U C(0)R8, -lJ3 R8CiO)R8, -U R8S{0)2R8, -U S(0)2 R8R9, -
197 U3NR8S(0)2R9, -U3 R8C(0)R9, -U3NR8C(0) R8R9, -U3NR8C(0)NR8R8, -U3C(0)OR8,
198 NOR '.■■{ . '' WVOlV. -U NR8(CH2)i .4NR8R8, -U3C(0) R8(CH2)1.4N 8 8, -U3C(0) R8(CH2)i .
199 4R9, -U C(0)NR8(CH2),_4OR9, -U30(CH2)i_4NR8R8, -U C(0) R8(CH2)i.4OR8 and■■
200 U3NR8(CH 2)i -4R9; wherein phenyl can be further substituted by a radical selected from - R8R8
201 or -C(0)NR8R8; U3 is as described above; R8 is hydrogen, Ci-ealkyl, hydroxy-Q-ealkyl or C2.
202 ealkenyl; and R9 is hydroxy, C&.1 oaryl-Co alkyi, C&.1 oaiy l-Co4alk lox , Cs-ioheteroaryl-Co-
203 4 alky 1, C3-gheterocycloalkyl-Co-4alkyl or C3-8cycloalkyl; wherein said aryl, heteroaryl,
204 cycloalkyl, heterocycioalkyl or alkyi of R9 is further optionally substituted by up to 2 radicals
205 selected from the group consisting of halo, hydroxy, cyano, amino, nitro, Ci..4alkyl, hydroxy-Cj .
206 eal yl, halo-substituted Chalky 1, Cj- alkoxy, halo-substituted Cj. alkoxy, halo-alky!--
207 substituted-phenyl, benzoxy, C^heteroaryl, C3-8heterocycloalkyl, -C(0) R8R8, -S(0)2NR8R8, -
208 NR8R8, -C(0)R10 and
209 - Rl lRn, wherein R10 is C ieteroaryl and R5 ! is hydroxy- Ci..4alkyl; wherein IV is substituted
210 with Q1; and
211 the N-oxide derivatives, prodnig derivatives, protected derivatives, individual isomers
212 and mixture of isomers thereof; and the pharmaceutically acceptable salts and solvates of such
213 compounds;
215 wherein:
6
216 R is pyrazolyl; wherein said pyrazolyf is unsubstituted or substituted with 1 to 2 R'
217 groups;
218 R? is pyrroiidinyl; wherein said pyrrolidinyl is substituted with one R' group;
219 RJ is selected from hydrogen and halo;
220 R4 is selected from -SF< and -Y2-CF?~Y3;
221 R° at each occurrence is independently selected from the group consisting of hydrogen,
222 hydroxy, methyl, methoxy, cyano, trifluoromethyi, hydroxy-methyl, halo, amino, fluoro- ethyl,
223 ethyl and cyclopropyl;
224 R'' is selected from hydroxy, methyl, halo, methoxy, hydroxy-methyl, amino, methyl-
225 amino, amino-methyl, trifluoromethyi, 2-hydroxypropan-2-yI, memyl-carbonyl-amino,
226 dimethyl-amino, 2-amino-3-methylbutanoyl)oxy, carboxy, methoxy-carbonyl, phosphonooxy,
227 cyano, and amino-carbonyl;
228 V is selected from CH and N;
229 Y1 is selected from CH and ;
230 Y2 is selected from CF2, O and S(0)o-2; and
231 Y3 is selected from hydrogen, chloro, fluoro, methyl, difluoromethyl, and
232 trifliioromethyl; or pharmaceutically acceptable salts thereof;
234 wherein:
235 Y at each occurrence is independently selected from N and CH;
236 Y1 is selected from and CR5; wherein R5 is selected from hydrogen, methoxy and
237 imidazolyl; wherein said imidazolyl is unsubstituted or substituted with methyl;
238 R1 is selected from pyrazolyi, thiazolyl, pyrrolyl, imidazolyl, isoxazolyl, furanyl and
239 thienyl; wherein said thiazolyl, pyrrolyl, imidazolyl, isoxazolyl, furanyl, or thienyl of R1 is
240 unsubstituted or substituted with 1 to 3 R6 groups;
241 R2 is selected from pyrrolidinyl, piperidinyl, azetidinyl, morpholino, piperazinyl, 2-oxa-
242 6-azaspiro[3.4]-octanyl, 3-azabicyclo[3.1 .0]hexan-3-yl, pyrrolo[3,4-c]pyrazol-5(lH,4H,6H)-yl,
243 hexahydropyrrolo[3,4~c]pyrrolyl, 6-oxo-2,7-diazaspiro[4.4]-nonanyl, 1 H-pyrroSo[3,4-
244 c]pyridinyl, l,4-oxazepan-4-yl, 2-oxooxazoiidinyl, 1,4-diazepanyi, tetrahydro-2H-pyranyl, 3,6-
245 dihydro-2H-pyranyl, 3,8-dioxa-10-azabicyclo[4.3.1 ]decanyl, -OR5b and - R^R50; wherein said
246 piperidinyl, azetidinyl, morpholino, piperazinyl, 1 ,4-oxazepan-4-yl, pyrrolo[3,4-c]pyrazol-
247 5(lH,4H,6H)-yl, 2-oxa-6-azaspiro[3.4]-octanyI, 3 -azabicyclo[3.1.0]hexan-3-yl,
248 hexahydropyrrolo[3,4-c]pyrroiyl, 6-oxo-2,7-diazaspiro[4.4]-nonanyl, lH-pyrrolo[3,4-
249 cjpyridinyl, 2-oxooxazolidinyl, 1 ,4-diazepanyl, tetrahydro-2H-pyranyl, 3,6-dihydro-2H-
250 pyranyl, or 3,8-dioxa-10-azabicyclo[4.3.1]decanyl of R2 is un substituted or substituted with 1
251 to 3 R' groups; wherein said pyrrolidinyl of R2 is unsubstituted or substituted with 2 or 3 R.''
252 groups;
253 R5 is selected from hydrogen and halo;
254 R4 is selected from -SF5 and -Y2-CF2-Y3;
255 R5a is selected from hydrogen and Ci-4alkyl;
257 unsubstituted or substituted with 1 to 3 groups independently selected from hydroxy and
258 dim.ethyl-am.ino;
259 R6 at each occurrence is independently selected from hydrogen, hydroxy, methyl,
260 hydroxy, methoxy, cyano, trifluoromethyl, hydroxy-methyl, halo, amino, fluoro-eihyl, ethyl,
261 cyciopropyl and dimethyl-amino-carbonyl;
262 R.? at each occurrence is independently selected from hydroxy, methyl, halo, methoxy,
263 hydroxy-methyl, amino, methyl-amino, ammo-methyl, trifluoromethyl, 2-hydroxypropan-2-yl,
264 methyl-carbonyl-amino, dimethyl-amino, 2-amino-3-methylbutanoyl)oxy, carboxy, methoxy -
265 carbonyl, phosphonooxy, cyano, and amino-carbonyl ; or two R? groups combine with the atom
266 to which they are attached to form a ring selected from cyciopropyl, azetidin-3-yl and 3-
267 azabicyclo[3.1.Ojhexan-3-yi;
268 Y2 is selected from CF2, O and S(0)o-2; and
269 Y"' is selected from hydrogen, halo, methyl, difruoromefhyl and trifluoromethyl; or
270 pharmaceutically acceptable salts thereof;
272 wherein:
273 X is cycloalkyl of 3 to 7 carbon atoms, which may be optionally substituted with one or
274 more alkyl of 1 to 6 carbon atom groups; or is a pyridmyl, pyrimidinyl, or phenyl ring; wherein
275 the pyridinyl, pyrimidinyl, or phenyl ring optionally mono- di-, or tri-substituted with a
276 substitueni selected from the group consisting of halogen, alkyl of 1-6 carbon atoms, alkenyl of
277 2-6 carbon atoms, alkynyl of 2-6 carbon atoms, azido, hydroxyalkyl of 1-6 carbon atoms,
278 halomethyl, alkoxymethyi of 2-7 carbon atoms, alkanoyloxymethyi of 2-7 carbon atoms,
279 alkoxy of 1-6 carbon atoms, aikylthio of 1-6 carbon atoms, hydroxy, trifluoromethyl, cyano,
280 nitro, carboxy, carboalkoxy of 2-7 carbon atoms, carboalkyl of 2-7 carbon atoms, phenoxy,
281 phenyl, thiophenoxy, benzoyl, benzyl, amino, alkylammo of 1-6 carbon atoms, dialkylamino of
282 2 to 12 carbon atoms, phenylamino, benzylamino, alka.noyla.mino of 1-6 carbon atoms,
283 alkenoylamino of 3-8 carbon atoms, alkynoylamino of 3-8 carbon atoms, and benzoylammo;
284 n is 0 or 1 ;
285 Y is -NH-, -0-, -S-, or -NR-;
286 R is Ci.6alkyi;
287 R1, R\ R3, and R4 are each, independently, hydrogen, halogen, alkyl of 1 -6 carbon
288 atoms, alkenyf of 2-6 carbon atoms, alkynyl of 2-6 carbon atoms, aikenyfoxy of 2-6 carbon
289 atoms, alkynyloxy of 2-6 carbon atoms, hydroxymethyl, halomethyl, alkanoyloxy of 1-6 carbon
290 atoms, alkenoyloxy of 3-8 carbon atoms, alkynoyloxy of 3-8 carbon atoms, alkanoyloxymethyl
291 of 2-7 carbon atoms, alkenoyloxyniethyl of 4-9 carbon atoms, alkynoyloxymethyl of 4-9
292 carbon atoms, alkoxymethyf of 2-7 carbon atoms, alkoxy of 1-6 carbon atoms, alkylthio of 1-6
293 carbon atoms, alkylsulphinyl of 1-6 carbon atoms, aikylsulphonyl of 1-6 carbon atoms,
294 alkylsulfonamido of 1-6 carbon atoms, alkenylsulfonamido of 2-6 carbon atoms,
295 alkynylsulfonamido of 2-6 carbon atoms, hydroxy, trifluorometliyl, cyano, nitro, carboxy,
296 carboalkoxy of 2-7 carbon atoms, carboalkyl of 2-7 carbon atoms, phenoxy, phenyl,
297 thiophenoxy, benzyl, amino, hydroxyamino, alkoxy amino of 1-4 carbon atoms, alkylamino of
298 .1 -6 carbon atoms, dialkylamino of 2 to 12 carbon atoms, aminoalkyl of 1 -4 carbon atoms, N-
299 alkyfaminoalkyl of 2-7 carbon atoms, N,N-dia3kylaminoalky3 of 3-14 carbon atoms,
300 phenylamino, benzylamino,
309 R5 is alkyl of 1-6 carbon atoms, alkyl optionally substituted with one or more halogen
310 atoms, phenyl, or phenyl optionally substituted with one or more halogen, alkoxy of 1-6 carbon
311 atoms, trifiuoromethyl, amino, itro, cyano, or alkyl of 1 -6 carbon atoms groups;
312 R6 is hydrogen, alkyl of 1-6 carbon atoms, or alkenyl of 2-6 carbon atoms;
313 R'' is chloro or bromo;
314 R* is hydrogen, alkyl of 1 -6 carbon atoms, aminoaikyl of 1 -6 carbon atoms, N-
315 alkylaminoalkyl of 2-9 carbon atoms, Ν,Ν-dialkylaminoalkyl of 3-12 carbon atoms, -
316 cycioalkylaminoalkyl of 4-12 carbon atoms, N-cycloalkyl-N-alkylaminoalkyl of 5-18 carbon
317 atoms, Ν,Ν-dicycloalkylaminoalkyl of 7-18 carbon atoms, ιηο ηοΙϊηο-Ν-βυί)*! wherein the
318 alkyl group is 1-6 carbon atoms, piperidino-N-alkyl wherein the alkyl group is 1-6 carbon
319 atoms, N-afkyl-piperidino-N-alkyl wherein either alkyl group is 1 -6 carbon atoms,
320 azacycloalkyl-isl -alkyl of 3- 1 1 carbon atoms, hydroxyalkyl of 1-6 carbon atoms, alkoxyalkyl of
321 2-8 carbon atoms, carboxy, carboalkoxy of 1 -6 carbon atoms, phenyl, carboalk l of 2-7 carbon
322 atoms, chloro, fluoro, or bromo; Z is amino, hydroxy, alkoxy of 1 -6 carbon atoms, alkylamino
323 wherein the aikyl moiety is of 1-6 carbon atoms, dialkylamino wherein each of the alkyl
324 moieties is of 1-6 carbon atoms, morpholino, piperazino, N-alkylpiperazino wherein the alkyl
325 moiety is of 1-6 carbon atoms, or pyrrolidine;
326 m is an integer from 1-4, q is an integer from 1 -3, and p is an integer from 0-3;
327 any two of the substituents Rl, R7', R3, or R4 that are located on contiguous carbon
328 atoms can together be the divalent radical -0-C(R8)2-0-;
329 or a pharmaceutically acceptable salt thereof;
331 wherein:
332 Ring T is a 5-membered heteroaryl ring containing 1-2 nitrogens with the remaining
333 ring atoms being carbon, substituted on at least two ring atoms with Rl groups, at least two of
334 which being located on adjacent ring atoms, and, together with the atoms to which they are
335 attached, forming a saturated, partially saturated or unsaturated 5- or 6- membered ring (Ring
336 E), containing 0-3 heteroatoms selected from O, N, and S and being optionally substituted with
337 1-4 Re groups;
338 Ring A is a 5- or 6-membered aryl or heteroaryl ring;
339 Ring B is a 5- or 6-membered aryl or heteroaryl ring;
340 L1 is selected from ^Ο(Ο), C(0) R1, Ν^Ο(0)0, R^C R1, and OC(0)NR1;
341 each occurrence of Ra, Rb and Rl is independently selected from the group consisting of
342 -(CH2)m-(Ring C), wherein Ring C is a 5- or 6-membered heterocyclic or heteroaryl ring
343 comprising 1-3 heteroatoms independently selected from O, N and S(0)r; halo; -CN; - O2; -R4;
344 -OR2; -NR2R3; -C(0)YR2; -OC(0)YR2; -NR2C(0)YR2; -SC(0)YR2; -NR2C(=S)YR2; -
345 OC(=S)YR2; -C(=S)YR2; -YC(=NR3)YR2; -YP(=0)(YR4)(YR4); -Si(R2)3; -NR2S02R2; -S(0)r-
346 R2; -S02NR2R3; and -NR2S02NR2R3, wherein each Y is independently a bond, -0-, -S- or -
347 NR3-;
348 Re, at each occurrence, is independently selected from the group consisting of halo, =0,
349 -CN, -N02, -R4, -OR2, -NR2R3, -C(0)YR2, -OC(0)YR2, -NR2C(0)YR2, -SC(0)YR2, -
350 NR2C(=S)YR2, -OC(=S)YR2, -C(=S)YR2, -YC(=NR3)YR2, -YP(=0)(YR4)(YR4), -Si(R2)3, -
351 NR2S02R2, -S(0)rR2, -S02NR2R3 and -NR2S02NR2R3, wherein each Y is independently a
352 bond, -0-, -S- or -NR3-;
353 R2, and R3 are independently selected from H, alkyl, alkenyl, alkynyl, cycloalkyl,
354 cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl;
355 alternatively, R2 and R3, taken together with the atom to which they are attached, form a
356 5- or 6- membered saturated, partially saturated or unsaturated ring, which can be optionally
357 substituted and which contains 0-2 heteroatorns selected from N, O and S(0)r-;
358 each occurrence of R4 is independently selected from alkyl, haloalkyl, trifluoromethyl,
359 alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl, heterocyclic and heteroaryl;
360 each of the alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkynyl, aryl,
361 heterocyclic and heteroaryl moieties is optionally substituted;
362 m is 0, 1 , 2, 3 or 4;
363 n is 2 or 3;
364 p is 0, 1, 2, 3, 4 or 5; and
365 r is 0, 1 or 2;
366 or a pharmaceutically acceptable salt;
368 wherein:
369 1 represents hydrogen, Chalk !, Ci-ealkoxy-Ci-ealkyl, or benzyl;
370 R2 represents phenyl substituted by one or two substituents selected from the group
371 consisting of Chalky!, trifluoro-C j -ealkyl, hydroxy -d-eaikyl, amino-Cj-ealkyl, C^alky!-
372 amino-Ci-eaikyl, (Cx-ealkyl^N-Cx-ealkyl, N-cyclohexyl-N-Cx-ealkylamino-Ci-ealkyl, C5_
375 benzoyloxy, carboxy, carbamoyl, Ci-ealkyl-carbamoyl, amino, C!-6aIkyi-C(0)-amino,
376 benzoylamino, amino mono- or disubstituted by Ci-ealkyl, by hydroxy-Ci-ea kyl or by C-.-
377 ealkoxy-Ci-eaikyl, ].H-imidazoly3, Ci-ealkyl-lIT-imidazolyl, carboxy- IH-imidazolyL Ci-ealkyl-
378 estercarboxy- 1 H-imidazolyl, pyrrolidin ), piperidino, piperazino, N-(Ci.6alkyl)piperazino,
379 morphoiino, sulfamoyl, Ci-ealkylsulfonyl, phenyl, pyridyl, halogenyl, or benzoyl; and
380 RJ represents hydrogen, Chalk ! or halo;
-oxide or pharmaceutically acceptable salts thereof;
383 wherein:
384 R1 is 4-pyrazinyl, 1 -methyl- lH-pyrrolyl, amino- or amino-Ci ealkyl-substituted phenyl
385 wherein the amino group in each case is free, alkylated or acylated, lH-indolyl or 1H-
386 imidazolyl bonded at a five-membered ring carbon atom, or unsubstituted or C-.-ealkyl-
387 substituted pyridyl bonded at a ring carbon atom and unsubstituted or substituted at the nitrogen
388 atom by oxygen,
389 R2 and R3 are each independently of the other hydrogen or Chalky 1, one or two of the
390 radicals R4, R5, R6, R7 and R8 are each nitro, fluoro-substituted G-ealkoxy or a radical
391 represented by:
392 -N(R9)-C(=X)-(Y)„-R10
393 wherein:
394 R9 is hydrogen or Chalky!,
395 X is oxo, thio, imino, N-Ci-ealkyl-imino, hydroximino or O-Ci-ealkyf-hydroximino,
396 Y is oxygen or the group NH,
397 n is 0 or 1 and
398 R10 is phenyl that is unsubstituted or substituted by halogen, cyano, Ci-6alkoxy,
399 carboxy, Ci-6alkyl, or by 4-methyl-piperazinylmethyl; C5_7alkyl; thienyl; 2-naphthyl;
400 cyclohexyl; an aliphatic radical having at least 5 carbon atoms; or an aromatic, aromatic -
401 aliphatic, cycloaliphatic, cycloaliphatic-aliphatic, heterocyclic or hetero-cyclicaliphatic radical;
402 and the remaining radicals R4, R5, R6, R7 and R8 are each independently hydrogen, G .
403 6alkyl that is unsubstituted or substituted by free or alkylated amino, piperazinyl, piperidinyl,
404 pyrrolidinyl or by morpholinyl, or Chalky l-C(Q)-, trifluoromethyl, free, etherified or esterifed
405 hydroxy, free, alkylated or acylated amino or free or esterified carboxy;
406 or a pharmaceutically acceptable salt thereof;
408 wherein:
409 E1 is phenyl substituted with 1, 2, or 3 groups independently selected from fluoro and
410 methyl;
411 A is selected from the group consisting of imidazolyl and pyrazolyl, wherein A is
412 substituted at any suitable position with a 10-membered bicyclic heteroaryl ring optionally
413 substituted with 1, 2, or 3 R! groups;
414 R1 is independently selected from the group consisting of Ci-eaikyl, branched CV/alkyl,
415 Cvgcarbocyclyl, halogen, fluoroCj-ealkyl wherein the alkyl moiety can be partially or fully
416 fluorinated, cyano, hydroxyl, methoxy, oxo, (R2)2NC{0 , -N(R2)C(0)R', (R2)2NS02-, -
417 N(R jS02R2, -(CH2)qN(R2)2, -0(CH2)qN(R2)2, -0(CH2)qO-Ci..6alkTl, -N(R2)(CH2)qO-Ci-6alkyi,
418 - (R2)(CH2)q (R2)2, -0(CH2)qR2, -N(R )(CH2)qR2, -C(0)R2, and nitro
419 R2 is H, Ci-ealkyl, branched Ch lky!, Cs-scarbocyclyl, or phenyl
420 t is 1 , 2, or 3 ;
421 or a pharmaceutically acceptable salt thereof; and
423 wherein:
424 Ring A is selected from the group consisting of phenylene, pyridinylene, pyrazinylene,
425 pyrirnidinylene, pyridazinylene, and triazinylene;
426 Ring B is selected from the group consisting of pyridyl, pyrimidinyl, pyrazinyl,
427 pyridazinyl, and 1,2 -dihydropyridazinyL each optionally substituted with 1, 2, or 3 groups
428 independently selected from Chalky!, halo, and amino;
429 Rl is -(CH2)„-R3, wherein R5 is a saturated, nitrogen-containing heterocyclic group
430 substituted by oxo, a saturated, nitrogen-containing heterocyclic group, aminoalkyl,
431 monoalkylaminoalkyl, dialkylaminoalkyi, Ci-ealkoxy, Ci-ealkyl, alkyoxycarbonyl, halo,
432 haloaikyl, hydroxyalkyl, amino, monoaikylamino, dialkylamino, carbamoyl,
433 monoalkylcarbamoyl, and dialkylcarbamoyl;
434 2 is selected from Ci-ealkyl, halogen, haloaikyl, trifluoromethyl, hydroxyalkyl, Cj .
435 ealkoxy, alkoxyalkyl, aikoxvcarbonvl, acyl, amino, monoaikylamino, diaikylamino, nitro,
436 carbamoyl, monoalkylcarbamoyl, dialkylcarbamoyl, and cyano;
437 R is selected from hydrogen, C. -ealkoxy, and halo;
438 R4 is selected from hydrogen, Ci_6alkyi, and halo; and
439 n is 1 , 2, 3, or 4;
440 or a pharmaceutically acceptable salt thereof.
1 2. The bivalent compound of claim 1, wherein Q1 is selected from the group consisting of:
2 -W1-(C3-6alkylene-0)s-Ci_6alkylene-W1-; -W^CHz-CHz-OyCi-galkylene-W1-; -W1-
4 heteroaliphatic moiety;
5 wherein:
6 W1 is, independently for each occurrence, N, O, or S;
7 s is an integer from 1 -10;
8 t is an integer from 3- 10.
1 3. The bivalent compound of claim 1, wherein Q1 is -S-(C3_6alkylene-0)s-Ci_6alkylene-S-;
2 -S-(CH2-CH2-0)t-Ci-6alkylene-S-; -0-(CH2-CH2-0)s-; and -0-C1-15alkylene-0-; wherein s is an
3 integer from 1- 10, and t is an integer from 3- 10.
1 4. The bivalent compound of claim 1, wherein Q1 is -0-(CH2-CH2-0)s-.
1 5. The bivalent compound of claim 1, wherein Q1 is selected from the group consisting of:
2 -S-(CH2-CH2-CH2-0)s-CH2-CH2-CH2-S-; -S-(CH2-CH2-0)t-CH2-CH2-S-; and -O-
3 (CH2)u-0-; wherein u is an integer from 1 -15.
1 6. The bivalent compound of claim 5, wherein u is 4, 5, 6, 7, 8, 9, 10, 1 1 , 12, 13, 14, or 15.
1 7. The bivalent compound of any one of claims 1-6, wherein s is 2, 3, 4, 5, 6, 7, or 8.
1 8. The bivalent compound of any one of claims 1-6, wherein Q1 has a length of less than
2 about 30 A;
1 9. The bivalent compound of claim 8, wherein Q1 has a length of less than about 20 A.
10. The bivalent compound of claim 8, wherein Q1 has a length of less than about 10 A. 1 1. The bivalent compound of claim I, wherein Q1 is selected from the group consisting of:
C|.2oalkylene, wherein one, two, or three or four methylene units of the hydrocarbon chain are optionally and independently replaced by -Si(R'')(Rs)-, -Si(R7)(R8)--Q--, -0-Si(R'')(R8)- , cyclopropylene, -NR'-, -N(R')C(0)-, -C(0)N(R')-, -N(R')S02-, - SU .Xi RV. -0-, -C(O)-, - OC(0)-, -C(0)0-, -S-, -SO-, -S02-, -C(=S)-, -C(=NR , C2-6alkenyleiie, C^ealkynylene, phenyl, naphthyl, or a mono or bicyclic heterocyclic or heteroaryl ring; -NR'-Ci-isalkyl-NR'- C(O)-; -NR'-(CH2-CH2-0)s-C1_6alkyl-NR'-C(0)-; -(0-CH2-CH2)s-NR'-C(0)-; -(0-CH2-CH2)s; -S-Co-6alkyl-; -NR'-Ci_6alkyl-; -N(Ci_3alkyl)-Ci_6alkyl-NH-C(0)-; -NH-Ci_6alkyl-N(Ci_3alkyl)- C(O)-; -S02-NR'-Co-6alkyl-; -SO2-heterocyclyl-C0-6alkyl-; -heterocyclyl-C(O)-; -heterocyclyl- C0-6alkyl-NR'-C(O)-; -NR'-C0-6alkylene-heterocyclene-C(O)-; -0-Ci_6alkylene-C(0)-; -0-Ci_ 15alkylene-NR'-C(0)-; -0-Ci_i5alkylene-C(0)-NR'-; and -0-Ci_6alkylene-, wherein Ci_ 6alkylene is optionally substituted by -OH;
wherein:
R7 and R8 are selected, independently for each occurrence, from the group consisting of -OH, Ci-6alkyl, -0-Ci_6alkyl, C2_6alkenyl, C3_6cycloalkyl, -Ci_6alkyl- NRaRb, phenyl and heteroaryl; wherein Ci-6alkyl, C2_6alkenyl, C3_6cycloalkyl, phenyl, and heteroaryl may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, hydroxyl, Ci-6alkyl, and phenyl; or
R7 and R8, together with the silicon to which they are attached, form a 4-7 membered heterocyclic ring, optionally containing one, two, three, or four heteroatoms independently selected from O, S, or N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, and hydroxyl;
R' is, independently for each occurrence, selected from the group consisting of hydrogen and C1-4alkyl; wherein
may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; and
s is an integer from 1-15.
12. The bivalent compound of claim 11, wherein Q1 comprises:
a second group selected from the group consisting of:
R8 R7 , R8 R7 s
, and R8 R7 wherein thee first group and the Si of the second group are connected by an oxygen atom;
wherein:
Rw is, independently for each occurrence, absent or selected from the group consisting of
-0-Ci-4alkylene-, -N(R )-, -N(R )-Ci_4alkylene-, -0-, - C(0)Ci_4alkylene-, -C(0)-0-Ci_4alkylene-, -C2-6alkenylene-, -C2-6alkynylene-, -C3_ 6cycloalkylene-, -phenylene-, and -heteroarylene-; wherein
C2-6alkenylene, C2-6alkynylene, C3_6cycloalkylene, phenylene, and heteroarylene may be optionally substituted by one, two, three, or more substituents independently selected from the group consisting of d_4alkyl, Ci_4alkoxy, -C(0)Ci_4alkyl, -C(0)-0-Ci_4alkyl, -C(O)- NRaRb, halogen, cyano, hydroxyl, and phenyl; or Rw and R7, together with the silicon to which they are attached, form a 3-8 membered heterocyclic ring, wherein the 3-8 membered ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, hydroxyl, and Ci-6alkyl;
W3 is, independently for each occurrence, (a) absent; or (b) selected from the group consisting of -C^alkylene-, -0-Ci-4alkylene-, -C(0)-Ci_4alkylene-, -N(R )-Ci_ 4alkylene-, -C(0)-0-Ci_4alkylene-, -C2-6alkenylene-, -C2-6alkynylene-, -C3_
6cycloalkylene-, -phenylene-, and -heteroarylene-; wherein
C2-6alkenylene, C2-6alkynylene, C3-6cycloalkylene, phenylene, and heteroarylene are optionally substituted by one, two, three, or more substituents independently selected from the group consisting of C^alkyl, d^alkoxy, -C(0)Ci_6alkyl, -C(0)-0-Ci_4alkyl, halogen, hydroxyl, nitro, and cyano;
R7 and R8 are selected, independently for each occurrence, from the group consisting of -OH, Ci-6alkyl, -0-Ci_6alkyl, C2-6alkenyl, C3_6cycloalkyl, -Ci_6alkyl- NRaRb, phenyl and heteroaryl; wherein Ci-6alkyl, C2-6alkenyl, C3_6cycloalkyl, phenyl, and heteroaryl may be optionally substituted by one or more substituents independently
selected from the group consisting of halogen, cyano, hydroxyl, Ci-6alkyl, and phenyl; or
R7 and R8, together with the silicon to which they are attached, form a 4-7 membered heterocyclic ring, optionally containing one, two, three, or four heteroatoms independently selected from O, S, or N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo, and hydroxyl;
R', Ra, and Rb are selected, independently for each occurrence, from the group consisting of hydrogen and Ci-4alkyl; wherein Ci-4alkyl may be optionally substituted by one or more substituents selected from the group consisting of halogen, cyano, oxo, and hydroxyl; or
Ra and Rb, together with the nitrogen to which they are attached, may form a 4-7 membered heterocyclic ring, optionally containing an additional heteroatom selected from O, S, and N; wherein the 4-7 membered heterocyclic ring may be optionally substituted by one or more substituents independently selected from the group consisting of halogen, cyano, oxo and hydroxyl;
BB, independently for each occurrence, is a 4-7-membered cycloalkyl, heterocyclic, phenyl, naphthyl, or heteroaryl moiety, wherein the cycloalkyl, heterocyclic, phenyl, naphthyl, or heteroaryl moiety is optionally substituted with one, two, three, or more groups represented by RBB; wherein R7, independently for each occurrence, may be optionally bonded to BB; and
each RBB is selected, independently for each occurrence, from the group consisting of hydrogen, halogen, nitro, cyano, hydroxyl, amino, thio, -COOH, - CONHR', substituted or unsubstituted aliphatic, and substituted or unsubstituted heteroaliphatic; or two RBB together with the atoms to which they are attached form a fused 5- or 6-membered cycloalkyl or heterocyclic bicyclic ring system.
13. The bivalent compound of claim 12, wherein Q1 comprises:
a first group selected from the group consisting of:
a second group selected from the group consisting of:
14. The bivalent compound of claim 12 or 13, wherein R7 and R8 are Ci-6alkyl.
15. The bivalent compound of any one of claims 1 1-14, wherein Q1 is selected from the group consisting of:
The bivalent compound of claim 1, wherein Q1 comprises a trizolyl group.
17. The bivalent compound of claim 1, wherein Q1 is -W1-(CH2-CH2-0)t-Ci-6alkylene- triazolylene-Ci-6alkylene-0-Ci_6alkylene-.
18. The bivalent compound of claim 17, wherein Q1 has the following structure:
19. The bivalent compound of any one of claims 1-18, wherein P1 and P2 are different. 20. The bivalent compound of any one of claims 1-19, wherein P1 is:
21. The bivalent compound of any one of claims 1-19, wherein P1 is:
22. The bivalent compound of any one of claims 1-19, wherein P1 is selected from the group consisting of imatinib, nilotinib, bosutinib, ponatinib, DCC-2036, GNF5, and bafetinib. 23. The bivalent compound of any one of claims 1-19, wherein P2 is selected from the group consisting of imatinib, nilotinib, bosutinib, ponatinib, DCC-2036, GNF5, and bafetinib. 24. A bivalent compound selected from the group consisting of:
, wherein m and n are independently 2, 4, 6, 8, or 10;
N-(2-Chloro-6-methylphenyl)-2-((2-methyl-6-(4-(2-(( 1 -( 1 -oxo- 1 -(3 -(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)phenyl)-5,8, 11 , 14, 17,20,23 -heptaoxa-2- azapentacosan-25-yl)- 1H- 1 ,2,3 -triazol-4-yl)methoxy)ethyl)piperazin- 1 -yl)pyrimidin-4- yl)amino)thiazole-5-carboxamide;
N-(2-Chloro-6-methylphenyl)-2-((2-methyl-6-(4-(2-(( 1 -( 1 -oxo- 1 -(3 -(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)phenyl)-5,8, l 1,14, 17,20,23, 26,29-nonaoxa-2- azahentriacontan-31 -yl)- 1H- 1 ,2,3 -triazol-4-yl)methoxy)ethyl)piperazin- 1 -yl)pyrimidin-4- yl)amino)thiazole-5-carboxamide;
N-(2-Chloro-6-methylphenyl)-2-((2-methyl-6-(4-(2-(( 1 -( 1 -oxo- 1 -(3 -(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)phenyl)-5, 8, 11, 14, 17,20,23,26,29,32,35- undecaoxa-2-azaheptatriacontan-37-yl)- 1H- 1 ,2,3 -triazol-4-yl)methoxy)ethyl)piperazin- 1 - yl)pyrimidin-4-yl)amino)thiazole-5-carboxamide;
4-Methyl-N-(4-((4-methylpiperazin-l-yl)methyl)-3-(trifluoromethyl)phenyl)-3-((7-((l- oxo- 1 -(4-( 1 , 1 ,3 ,3 -tetramethyl-3 -(4-((2-(2-(2-(3 -(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4- yl)benzamido)ethoxy)ethoxy)ethyl)carbamoyl)phenyl)disiloxanyl)phenyl)- 5,8, 1 1, 14, 17,20,23,26,29-nonaoxa-2-azahentriacontan-31-yl)oxy)imidazo[l,2-a]pyridin-3- yl)ethynyl)benzamide;
4,4'-(l, l,3,3-Tetramethyldisiloxane-l,3-diyl)bis(N-(2-(2-(2-(3-(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4-yl)benzamido)ethoxy)ethoxy)ethyl)benzamide);
4-Methyl-N-(4-((4-methylpiperazin-l-yl)methyl)-3-(trifluoromethyl)phenyl)-3-((7-((l- oxo- 1 -(4-( 1 , 1 ,3 ,3 -tetramethyl-3 -(4-((2-(2-(2-(3 -(6-((4- (trifluoromethoxy)phenyl)amino)pyrimidin-4- yl)benzamido)ethoxy)ethoxy)ethyl)carbamoyl)phenyl)disiloxanyl)phenyl)-5,8, 11 -trioxa-2-
azatridecan-13-yl)oxy)imidazo[l,2-a]pyridin-3-yl)ethynyl)benzamide; and pharmaceutically acceptable salts thereof.
25. A method of treating a disease associated with Bcr-Abl tyrosine kinase in a patient in need thereof comprising:
administering to the patient the bivalent compound of any one of claims 1 -24.
26. The method of claim 25, wherein the disease is chronic myelogenous leukemia.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201461926470P | 2014-01-13 | 2014-01-13 | |
| US201461926466P | 2014-01-13 | 2014-01-13 | |
| US61/926,466 | 2014-01-13 | ||
| US61/926,470 | 2014-01-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015106294A1 true WO2015106294A1 (en) | 2015-07-16 |
Family
ID=53524433
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2015/011273 Ceased WO2015106294A1 (en) | 2014-01-13 | 2015-01-13 | Bivalent bcr-abl tyrosine kinase ligands, and methods of using same |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2015106294A1 (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10966966B2 (en) | 2019-08-12 | 2021-04-06 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11185535B2 (en) | 2019-12-30 | 2021-11-30 | Deciphera Pharmaceuticals, Llc | Amorphous kinase inhibitor formulations and methods of use thereof |
| US11266635B2 (en) | 2019-08-12 | 2022-03-08 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11395818B2 (en) | 2019-12-30 | 2022-07-26 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea |
| WO2023114759A3 (en) * | 2021-12-13 | 2023-07-27 | The Regents Of The University Of California | Abl inhibitors and uses thereof |
| US11779572B1 (en) | 2022-09-02 | 2023-10-10 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| WO2023196991A1 (en) * | 2022-04-08 | 2023-10-12 | The Brigham And Women's Hospital, Inc. | Bifunctional chimeric molecules for labeling of kinases with target binding moieties and methods of use thereof |
| US11986463B2 (en) | 2018-01-31 | 2024-05-21 | Deciphera Pharmaceuticals, Llc | Combination therapy for the treatment of gastrointestinal stromal tumor |
| US12102620B2 (en) | 2018-01-31 | 2024-10-01 | Deciphera Pharmaceuticals, Llc | Combination therapy for the treatment of mastocytosis |
| US12509456B2 (en) | 2019-03-11 | 2025-12-30 | Deciphera Pharmaceuticals, Llc | Solid state forms of ripretinib |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5130461A (en) * | 1990-06-06 | 1992-07-14 | Shin-Etsu Chemical Company Limited | 1,3-bis(p-hydroxybenzyl)-1,1,3,3-tetramethyldisiloxane and method for making |
| US20110021524A1 (en) * | 2008-01-14 | 2011-01-27 | Irm Llc | Compositions and methods for treating cancers |
| US20120094998A1 (en) * | 2009-04-17 | 2012-04-19 | Nektar Therapeutics | Oligomer-Protein Tyrosine Kinase Inhibitor Conjugates |
| WO2013033268A2 (en) * | 2011-08-29 | 2013-03-07 | Coferon, Inc. | Bivalent bromodomain ligands, and methods of using same |
| WO2013127361A1 (en) * | 2012-03-01 | 2013-09-06 | The Hong Kong Polytechnic University | Alkyne-, azide- and triazole-containing flavonoids as modulators for multidrug resistance in cancers |
-
2015
- 2015-01-13 WO PCT/US2015/011273 patent/WO2015106294A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5130461A (en) * | 1990-06-06 | 1992-07-14 | Shin-Etsu Chemical Company Limited | 1,3-bis(p-hydroxybenzyl)-1,1,3,3-tetramethyldisiloxane and method for making |
| US20110021524A1 (en) * | 2008-01-14 | 2011-01-27 | Irm Llc | Compositions and methods for treating cancers |
| US20120094998A1 (en) * | 2009-04-17 | 2012-04-19 | Nektar Therapeutics | Oligomer-Protein Tyrosine Kinase Inhibitor Conjugates |
| WO2013033268A2 (en) * | 2011-08-29 | 2013-03-07 | Coferon, Inc. | Bivalent bromodomain ligands, and methods of using same |
| WO2013127361A1 (en) * | 2012-03-01 | 2013-09-06 | The Hong Kong Polytechnic University | Alkyne-, azide- and triazole-containing flavonoids as modulators for multidrug resistance in cancers |
Cited By (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11986463B2 (en) | 2018-01-31 | 2024-05-21 | Deciphera Pharmaceuticals, Llc | Combination therapy for the treatment of gastrointestinal stromal tumor |
| US12102620B2 (en) | 2018-01-31 | 2024-10-01 | Deciphera Pharmaceuticals, Llc | Combination therapy for the treatment of mastocytosis |
| US12509456B2 (en) | 2019-03-11 | 2025-12-30 | Deciphera Pharmaceuticals, Llc | Solid state forms of ripretinib |
| US12023327B2 (en) | 2019-08-12 | 2024-07-02 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US12023326B2 (en) | 2019-08-12 | 2024-07-02 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11426390B2 (en) | 2019-08-12 | 2022-08-30 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11433056B1 (en) | 2019-08-12 | 2022-09-06 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11529336B2 (en) | 2019-08-12 | 2022-12-20 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11534432B2 (en) | 2019-08-12 | 2022-12-27 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US12318373B2 (en) | 2019-08-12 | 2025-06-03 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11576904B2 (en) | 2019-08-12 | 2023-02-14 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US12295944B2 (en) | 2019-08-12 | 2025-05-13 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11266635B2 (en) | 2019-08-12 | 2022-03-08 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US12059410B2 (en) | 2019-08-12 | 2024-08-13 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US12059411B2 (en) | 2019-08-12 | 2024-08-13 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US10966966B2 (en) | 2019-08-12 | 2021-04-06 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US12023325B2 (en) | 2019-08-12 | 2024-07-02 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11344536B1 (en) | 2019-08-12 | 2022-05-31 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11969414B2 (en) | 2019-08-12 | 2024-04-30 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11813251B2 (en) | 2019-08-12 | 2023-11-14 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
| US11844788B1 (en) | 2019-12-30 | 2023-12-19 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11911370B1 (en) | 2019-12-30 | 2024-02-27 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11850241B1 (en) | 2019-12-30 | 2023-12-26 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US12023328B2 (en) | 2019-12-30 | 2024-07-02 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11918564B1 (en) | 2019-12-30 | 2024-03-05 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11793795B2 (en) | 2019-12-30 | 2023-10-24 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea |
| US11969415B1 (en) | 2019-12-30 | 2024-04-30 | Deciphera Pharmaceuticals, Llc | (methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11185535B2 (en) | 2019-12-30 | 2021-11-30 | Deciphera Pharmaceuticals, Llc | Amorphous kinase inhibitor formulations and methods of use thereof |
| US12318374B2 (en) | 2019-12-30 | 2025-06-03 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11850240B1 (en) | 2019-12-30 | 2023-12-26 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11395818B2 (en) | 2019-12-30 | 2022-07-26 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea |
| US11801237B2 (en) | 2019-12-30 | 2023-10-31 | Deciphera Pharmaceuticals, Llc | Amorphous kinase inhibitor formulations and methods of use thereof |
| US11903933B2 (en) | 2019-12-30 | 2024-02-20 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11576903B2 (en) | 2019-12-30 | 2023-02-14 | Deciphera Pharmaceuticals, Llc | Amorphous kinase inhibitor formulations and methods of use thereof |
| US12064422B2 (en) | 2019-12-30 | 2024-08-20 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11896585B2 (en) | 2019-12-30 | 2024-02-13 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea |
| US12213967B2 (en) | 2019-12-30 | 2025-02-04 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US12213968B2 (en) | 2019-12-30 | 2025-02-04 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US12226406B2 (en) | 2019-12-30 | 2025-02-18 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea |
| US11612591B2 (en) | 2019-12-30 | 2023-03-28 | Deciphera Pharmaceuticals, Llc | Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea |
| WO2023114759A3 (en) * | 2021-12-13 | 2023-07-27 | The Regents Of The University Of California | Abl inhibitors and uses thereof |
| WO2023196991A1 (en) * | 2022-04-08 | 2023-10-12 | The Brigham And Women's Hospital, Inc. | Bifunctional chimeric molecules for labeling of kinases with target binding moieties and methods of use thereof |
| US11779572B1 (en) | 2022-09-02 | 2023-10-10 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2015106294A1 (en) | Bivalent bcr-abl tyrosine kinase ligands, and methods of using same | |
| US12473272B2 (en) | JAK1 selective inhibitors | |
| CN111801327B (en) | Benzamide compounds | |
| US20240300942A1 (en) | Small molecules for treatement of cancer | |
| WO2015106292A1 (en) | Bcr-abl tyrosine-kinase ligands capable of dimerizing in an aqueous solution, and methods of using same | |
| EP3191472B1 (en) | Compounds and compositions as raf kinase inhibitors | |
| WO2021051034A1 (en) | Ras protein degraders, pharmaceutical compositions thereof, and their therapeutic applications | |
| CN115175901A (en) | Compounds and methods for targeted degradation of androgen receptor | |
| US20140243321A1 (en) | Bioorthogonal monomers capable of dimerizing and targeting bromodomains, and methods of using same | |
| US20140243322A1 (en) | Bivalent bromodomain ligands, and methods of using same | |
| RU2692479C2 (en) | (5,6-dihydro)pyrimido[4,5-e]indolysines | |
| CN112574255A (en) | Organic arsine-based CDK inhibitor and preparation method and application thereof | |
| KR20160140739A (en) | Macrocylic pyrimidine derivatives | |
| CA3083228A1 (en) | Small molecule degraders that recruit dcaft15 | |
| WO2023108108A1 (en) | Raf kinase inhibitors and methods of use thereof | |
| WO2022170164A1 (en) | Sulfonamides with egfr inhibition activities and their use thereof | |
| AU2016293841B2 (en) | Fused quinoline compunds as pi3k/mTor inhibitors | |
| UA73728C2 (en) | Derivatives of n-triazolylmethylpiperazine, methods for the preparation thereof, medicament and intermediary compound | |
| US20240190842A1 (en) | Substituted quinolinone-8-carbonitrile derivatives having androgen degradation activity and uses thereof | |
| CN114126616A (en) | Nanoparticle formulations of BCL-2 inhibitors | |
| JP2025533088A (en) | HSD17B13 inhibitors and/or degraders | |
| JP2025513725A (en) | TEAD INHIBITORS AND METHODS OF USE | |
| JP7186874B2 (en) | Pyrazolyl compound and method of use thereof | |
| WO2022268065A1 (en) | Compounds as erk inhibitors | |
| EP4495111A1 (en) | Novel compound and use thereof for inhibiting checkpoint kinase 2 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15735416 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 15735416 Country of ref document: EP Kind code of ref document: A1 |