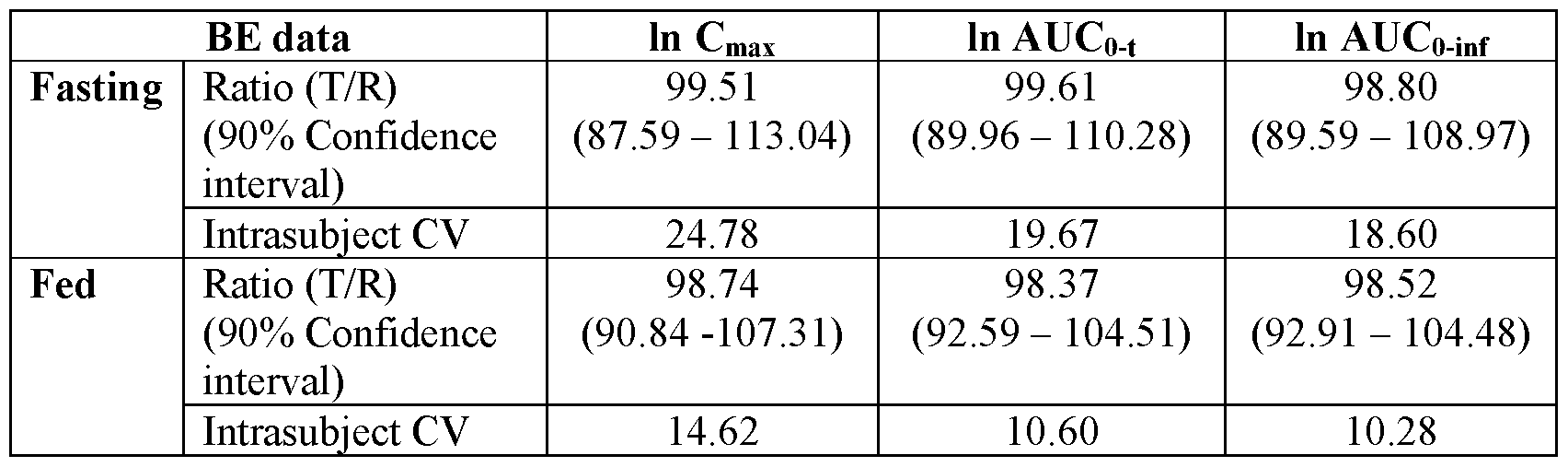WO2015019237A1 - Pharmaceutical composition of vilazodone - Google Patents
Pharmaceutical composition of vilazodone Download PDFInfo
- Publication number
- WO2015019237A1 WO2015019237A1 PCT/IB2014/063484 IB2014063484W WO2015019237A1 WO 2015019237 A1 WO2015019237 A1 WO 2015019237A1 IB 2014063484 W IB2014063484 W IB 2014063484W WO 2015019237 A1 WO2015019237 A1 WO 2015019237A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pharmaceutical composition
- vilazodone
- pharmaceutically acceptable
- acceptable excipients
- particles
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
Definitions
- a first aspect of the present invention provides a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients.
- Bioequivalence is defined to mean the term used by the drug approval agencies, such as the US Food and Drug Administration: "the absence of a significant difference in the rate and extent to which the active ingredient or active moiety in pharmaceutical equivalents or pharmaceutical alternatives becomes available at the site of drug action when administered at the same molar dose under similar conditions in an appropriately designed study.” This is typically understood to mean that the reference drug is within +25% and -20% of the reference drug product for AUC and Cmax, for example as explained in the US FDA's various bioequivalence guidance documents for oral tablets and capsules, which are incorporated herein by reference.
- step (d) compressing the blend of step (c) to form tablets
- Suitable plasticizers are selected from the group consisting of triethyl citrate, dibutyl sebacate, acetylated triacetin, tributyl citrate, glycerol tributyrate, monoglyceride, rapeseed oil, olive oil, sesame oil, acetyl tributyl citrate, acetyl triethyl citrate, glycerin sorbitol, diethyl oxalate, diethyl phthalate, diethyl malate, diethyl fumarate, dibutyl succinate, diethyl malonate, dioctyl phthalate, or mixtures thereof.
- Microcrystalline cellulose and colloidal silicon dioxide were sifted and mixed together.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The present invention relates to pharmaceutical compositions comprising micronized particles of vilazodone and processes for their preparation. It further relates to a method of treating and/or preventing central nervous system disorders by administering said pharmaceutical compositions.
Description
PHARMACEUTICAL COMPOSITION OF VILAZODONE
Field of the Invention
The present invention relates to pharmaceutical compositions comprising micronized particles of vilazodone and processes for their preparation. It further relates to a method of treating and/or preventing central nervous system disorders by administering said pharmaceutical compositions.
Background of the Invention
Vilazodone hydrochloride, disclosed in U.S. Patent No. 5,532,241 is chemically designated as 2-benzofurancarboxamide, 5-[4-[4-(5-cyano-lH-indol-3-yl)butyl]-l- piperazinyl]-, hydrochloride (1 : 1). Vilazodone hydrochloride is a selective serotonin reuptake inhibitor and a 5HTIA receptor partial agonist. It is indicated for the treatment of major depressive disorder.
A review of the art shows that U.S. Patent No. 7,834,020 discloses fifteen crystalline modifications of vilazodone hydrochloride designated as Form I through Form XV. It also discloses conventional forms of administration such as tablets and capsules. PCT Publication No. WO 2012/131706 discloses the amorphous form of vilazodone hydrochloride, and PCT Publication No. WO 2013/078361 discloses several other polymorphic forms of vilazodone hydrochloride.
As the aqueous solubility of any drug substance plays an important role in pharmaceutical compositions, it always remains a challenge to formulators to increase the aqueous solubility in order to further enhance dissolution and absorption characteristics. Vilazodone hydrochloride, being poorly soluble in an aqueous medium, exhibits poor bioavailability following oral administration.
Thus there remains a need in the art for an improved formulation of vilazodone which can be formulated in pharmaceutical compositions that have enhanced dissolution and absorption characteristics.
Particle size plays an important role in establishing drug solubility. As the particle size is reduced, the surface area of the individual particles of the drug increases, thus a greater amount of a drug can be solubilized for obtaining a therapeutic effect, which leads to higher therapeutic efficacy. The present invention demonstrates that by processing vilazodone to bring its particle size within a specified narrow range, pharmaceutical
compositions may be prepared which exhibit both a desired in-vitro dissolution profile and in-vivo bioavailability. Further, it has been found that there is no polymorphic conversion observed while micronizing the vilazodone particles. In addition to this, polymorphic stability has been surprisingly observed even after applying great force during micronization.
Summary of the Invention
The present invention includes pharmaceutical compositions comprising micronized particles of vilazodone with enhanced dissolution and absorption
characteristics. It also includes processes for the preparation of said pharmaceutical compositions. It further includes a method of treating and/or preventing central nervous system disorders, in particular major depressive disorder, by administering said pharmaceutical compositions.
Detailed Description of the Invention
A first aspect of the present invention provides a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients.
According to one embodiment of the present invention, there is provided a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the particles have a D50 range of about 0.1 μιη to about 20 μιη.
According to another embodiment of the present invention, there is provided a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the particles have a D90 range of about 0.5 μιη to about 40 μιη.
According to another embodiment of the present invention, there is provided a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the particles have a Di0 range of about 0.05 μιη to about 10 μιη.
According to another embodiment of the present invention, there is provided a pharmaceutical composition comprising micronized particles of vilazodone and one or
more pharmaceutically acceptable excipients, wherein more than 40% w/w of the vilazodone dissolves within 30 minutes in 0. IN HC1, in USP type II apparatus.
According to another embodiment of the present invention, there is provided a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the composition is bioequivalent to the reference listed drug Viibryd® tablets when bioavailability studies are conducted in humans. Bioequivalence is defined to mean the term used by the drug approval agencies, such as the US Food and Drug Administration: "the absence of a significant difference in the rate and extent to which the active ingredient or active moiety in pharmaceutical equivalents or pharmaceutical alternatives becomes available at the site of drug action when administered at the same molar dose under similar conditions in an appropriately designed study." This is typically understood to mean that the reference drug is within +25% and -20% of the reference drug product for AUC and Cmax, for example as explained in the US FDA's various bioequivalence guidance documents for oral tablets and capsules, which are incorporated herein by reference.
According to another embodiment of the present invention, there is provided a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the composition is stable.
A second aspect of the present invention provides a process for the preparation of a pharmaceutical composition of vilazodone, wherein the process comprises the steps of:
(a) blending micronized vilazodone with one or more pharmaceutically
acceptable excipients;
(b) directly compressing the blend of step (a) to form tablets; and
(c) film coating the tablets obtained from step (b).
A third aspect of the present invention provides a process for the preparation of a pharmaceutical composition of vilazodone, wherein the process comprises the steps of:
(a) blending micronized vilazodone with one or more pharmaceutically
acceptable excipients;
(b) granulating the blend of step (a);
(c) blending the granules of step (b) with one or more pharmaceutically acceptable excipients;
(d) compressing the blend of step (c) to form tablets; and
(e) film coating the tablets obtained from step (d).
A fourth aspect of the present invention provides a method of treating and/or preventing a central nervous system disorder by administering a pharmaceutical composition comprising micronized particles of vilazodone and one or more
pharmaceutically acceptable excipients.
According to one embodiment of the present invention, there is provided a method of treating and/or preventing a central nervous system disorder by administering a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the central nervous system disorder is selected the group consisting of from major depressive disorder, anxiety disorders, bipolar disorders, mania, dementia, eating disorders, sleeping disorders, psychiatric disorders, cerebral infarct, tension, and combinations thereof.
According to another embodiment of the present invention, there is provided a method of treating and/or preventing a central nervous system disorder by administering a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the central nervous system disorder is major depressive disorder.
According to another embodiment of the present invention, there is provided a method of treating and/or preventing central nervous system disorders by administering a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients, wherein the method comprises coadministration of additional drugs acting on the central nervous system.
The term "vilazodone", as used herein, refers to 2-benzofurancarboxamide, 5-[4- [4-(5-cyano-lH-indol-3-yl)butyl]-l-piperazinyl]. It further includes salts, polymorphs, hydrates, solvates, prodrugs, chelates, and complexes thereof. The preferred salt of vilazodone is vilazodone hydrochloride. The pharmaceutical composition of the present invention comprises vilazodone in an amount of from about 1% w/w to about 30% w/w, in particular from about 5% w/w to about 20% w/w of the total composition. Vilazodone
hydrochloride may be used in the pharmaceutical composition in strengths of 10 mg, 20 mg, and 40 mg of vilazodone. The vilazodone hydrochloride used in the pharmaceutical composition may encompass any known polymorphs at the time of the invention. It may also encompass the amorphous from of vilazodone hydrochloride. In particular, the pharmaceutical composition of the present invention comprises vilazodone hydrochloride form IV.
The term "micronized", as used herein, means vilazodone particles with reduced particle size. The reduced particle sizes have been defined by particular D50, D90, and D-, 0 ranges.
The term "D50", as used herein, refers to a particle diameter at which 50% of the particles have diameters which are greater or smaller than the given value. The present invention includes vilazodone particles having D50 range from about 0.1 μπι to about 20 μπι, in particular, from about Ι μπι to about 10 μπι.
The term "D90", as used herein, refers to a particle diameter at which 90% of the particles have diameters which are smaller than the given value. The present invention includes vilazodone particles having D90 range from about 0.5 μπι to about 40 μπι, in particular, about 5 μπι to about 20 μπι.
The term "Dio", as used herein, refers to a particle diameter at which 10% of the particles have diameters which are smaller than the given value. The present invention includes vilazodone particles having Di0 range from about 0.05 μπι to about 10 μπι, in particular, from about 0.1 μπι to about 5 μπι.
The size reduction or micronization of vilazodone may be carried out by using any of the conventional mills, such as air jet mills, ball mills, colloid mills, grinding mills, roller mills, or impact mills. Preferably, the particle size is reduced by a ball mill.
Further, the particle size may be determined by any known technique including sieve analyzers; electrical conductance instruments such as coulter counters; and laser diffraction particle size analyzers such as a Malvern® instrument. Preferably, the particle size is determined by a Malvern® instrument.
The term "stable", as used herein, refers to chemical as well as physical stability. The chemical stability means maintaining the original formulation specifications, and physical stability means there is no change in the polymorphic form as determined by X- ray powder diffraction after exposure to a relative humidity of 75% at 40°C or a relative
humidity of 60% at 25°C, for a period of at least about three months to the extent necessary for sale and use of the composition.
Further, in the present invention, unmicronized and micronized vilazodone hydrochloride particles exhibit polymorphic stability, as there is no conversion of polymorphic forms during micronization.
The term "about", as used herein, refers to any value which lies within the range defined by a variation of up to ±10% of the value.
The term "pharmaceutical composition", as used herein, means a composition which is selected from the group consisting of tablets, capsules, pellets, pills, caplets, and granules; preferably tablets.
The term "pharmaceutically acceptable excipients", as used herein, includes excipients that may be added intragranularly and/or extragranularly in the pharmaceutical compositions. The pharmaceutically acceptable excipients are selected from the group consisting of fillers, binders, lubricants, disintegrants, surfactants, coloring agents, plasticizers, opacifiers, mixtures thereof.
Suitable fillers are selected from the group consisting of lactose monohydrate, lactose anhydrous, pregelatinized starch, calcium carbonate, calcium phosphate dibasic, calcium phosphate tribasic, calcium sulphate, kaolin, starch, or mixtures thereof.
Suitable binders are selected from the group consisting of methyl cellulose, hydroxypropyl cellulose, hydroxypropyl methylcellulose, polyvinylpyrrolidone, gelatin, gum arabic, ethyl cellulose, polyvinyl alcohol, pullulan, pregelatinized starch, agar, tragacanth, sodium alginate, propylene glycol, or mixtures thereof.
Suitable lubricants are selected from the group consisting of colloidal silicon dioxide, stearic acid, magnesium stearate, calcium stearate, zinc stearate, sodium stearyl fumarate, talc, hydrogenated castor oil, sucrose esters of fatty acids, microcrystalline wax, yellow beeswax, white beeswax, or mixtures thereof.
Suitable disintegrants are selected from the group consisting of sodium starch glycolate, croscarmellose, pregelatinized starch, microcrystalline cellulose, calcium carboxymethyl cellulose, low substituted hydroxypropyl cellulose, magnesium silicate, aluminum silicate, or mixtures thereof.
Suitable surfactants are selected form the group consisting of polyoxyethyl- sorbitan-fatty acid esters such as polysorbates; ethers of sugars such as ethers of lactose, sucrose, xylose, mannitol, or xylitol; ethoxylated fatty alcohols; fatty acids and their salts; ethoxylated fatty acids; polyglyceryl esters; copolymers of propylene oxide and of ethylene oxide; phospholipids or lecithins; amino acid fatty chain acrylates; triglycerides of plant or synthetic origin and their ethoxylated derivatives such as polyoxyl 35 castor oil and polyoxyl 40 hydrogenated castor oil; acetylated monoglycerides; sodium lauryl sulphate and its derivatives; taurocholic acid; or mixtures thereof.
Coloring agents may be any FDA approved color for oral use.
Suitable plasticizers are selected from the group consisting of triethyl citrate, dibutyl sebacate, acetylated triacetin, tributyl citrate, glycerol tributyrate, monoglyceride, rapeseed oil, olive oil, sesame oil, acetyl tributyl citrate, acetyl triethyl citrate, glycerin sorbitol, diethyl oxalate, diethyl phthalate, diethyl malate, diethyl fumarate, dibutyl succinate, diethyl malonate, dioctyl phthalate, or mixtures thereof.
Suitable opacifiers are selected from the group consisting of titanium dioxide, manganese dioxide, iron oxide, silicon dioxide, or mixtures thereof.
The present invention provides a process for the preparation of a pharmaceutical composition, wherein the process comprises the steps of:
(a) blending micronized vilazodone with one or more pharmaceutically
acceptable excipients;
(b) directly compressing the blend of step (a) to form tablets; and
(c) film coating the tablets obtained from step (b).
The present invention also provides a process for the preparation of a
pharmaceutical composition, wherein the process comprises the steps of:
(a) blending micronized vilazodone with one or more pharmaceutically
acceptable excipients;
(b) granulating the blend of step (a);
(c) blending the granules of step (b) with one or more pharmaceutically
acceptable excipients;
(d) compressing the blend of step (c) to form tablets; and
(e) film coating the tablets obtained from step (d).
The granulation may be carried out by using either a wet granulation or dry granulation process. Wet granulation may include rapid mixer granulation or fluid bed granulation. Dry granulation may involve the formation of slugs or the use of a chilsonator.
Examples of suitable solvents used for wet granulation include water, ethanol, methanol, isopropyl alcohol, dichloromethane, acetone, or mixtures thereof.
The pharmaceutical composition of the present invention is further coated with one or more functional or non-functional coatings. Preferably, the pharmaceutical composition is coated with one or more non-functional coatings. The coating layers may comprise one or more film-forming polymers and coating additives.
Examples of film-forming polymers include ethylcellulose, hydroxypropyl methylcellulose, hydroxypropyl cellulose, methylcellulose, carboxymethyl cellulose, hydroxymethyl cellulose, hydroxyethyl cellulose, cellulose acetate, hydroxypropyl methylcellulose phthalate, cellulose acetate phthalate, cellulose acetate trimellitate, waxes, and methacrylic acid polymers such as Eudragit® and the like. Alternatively,
commercially available coating compositions comprising film-forming polymers marketed under various trade names, such as Opadry®, may also be used.
Coating additives may be selected from the group consisting of binders, plasticizers, coloring agents, and lubricants.
Specific examples of granulating fluid/solvents for coating include acetone, ethanol, isopropyl alcohol, methylene chloride, or combinations thereof.
Coating may be performed by applying the coating composition as a
solution/suspension/blend using any conventional coating technique known in the art, such as spray coating in a conventional coating pan or fluidized bed processor, dip coating, or compression coating.
The invention may be further illustrated by the following examples, which are for illustrative purposes only and should not be construed as limiting the scope of the invention in any way.
EXAMPLES
Example 1
Vilazodone hydrochloride was micronized using an air-jet mill to give a powder having a D50 of 3.55 μιη, a D90 of 10.03 μιη, and a Di0 of 0.68 μιη.
Tablets comprising micronized vilazodone hydrochloride
1. Microcrystalline cellulose and colloidal silicon dioxide were sifted and mixed together.
Micronized vilazodone hydrochloride was sifted and mixed with lactose monohydrate.
3. The mixtures from steps 1 and 2 were mixed together.
4. The mixture of step 3 was lubricated with sifted magnesium stearate.
5. The final blend of step 4 was compressed.
6. Opadry® pink was dispersed in water and stirred for 45 minutes.
7. The core tablets of step 5 were coated with the coating dispersion of step 6.
Example 2
Unmicronized vilazodone hydrochloride particles having a D50 of 52.08 μιη, a D90 of 97.66 μιη, and a D10 of 23.28 μm were used.
Tablets comprising unmicronized vilazodone hydrochloride
Procedure:
1. Microcrystalline cellulose and colloidal silicon dioxide were sifted and mixed together.
2. Unmicronized vilazodone hydrochloride was sifted and mixed with lactose monohydrate.
3. The mixtures from steps 1 and 2 were mixed together.
4. The mixture of step 3 was lubricated with sifted magnesium stearate.
5. The final blend of step 4 was compressed.
6. Opadry® pink was dispersed in water and stirred for 45 minutes.
7. The core tablets of step 5 were coated with the coating dispersion of step 6.
In-vitro Studies
The tablets of vilazodone hydrochloride prepared according to Examples 1 and 2 were subjected to dissolution studies in 1000 mL of 0. IN HC1 using a USP apparatus II with a paddle speed at 60 rpm. Table 1 provides a comparative dissolution profile of tablets comprising micronized vilazodone hydrochloride (Example 1) and unmicronized vilazodone hydrochloride (Example 2).
Table 1: Percentage (%) of In-Vitro Drug Release in USP Type II Apparatus (Media: 0.1N HC1, 1000 mL at 60rpm)
From the above table, it is clear that the pharmaceutical composition prepared using the micronized vilazodone hydrochloride particles provides increased drug release compared to unmicronized vilazodone hydrochloride particles.
ϊη-vivo Studies
In-vivo performance of vilazodone hydrochloride tablets prepared as per the composition of Example 1 were evaluated with respect to the Viibryd® tablets in 24 healthy human volunteers under fasting and fed conditions with a wash out period of at least 14 days. The study protocol followed was an open label, balanced, randomized single-dose, crossover bioavailability study. Blood samples were collected at appropriate time intervals over a period of 144 hours and vilazodone content was quantified in plasma using validated chromatographic procedures. The 90% confidence intervals for the ratios of the log transformed mean values for Cmax, AUCo-t, and AUCo-inf for the test and reference product (T/R ratio) were calculated. The results of the study are provided in Table 2.
Table 2: Comparative Pharmacokinetic Data
Claims
1. A pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients.
2. The pharmaceutical composition according to claim 1, wherein the particles have a D50 range of about 0.1 μιη to about 20 μιη.
3. The pharmaceutical composition according to claim 1, wherein the particles have a D90 range of about 0.5 μιη to about 40 μιη.
4. The pharmaceutical composition according to claim 1, wherein the particles have a Dio range of about 0.05 μιη to about 10 μιη.
5. The pharmaceutical composition according to claim 1, wherein the
pharmaceutically acceptable excipients are selected from the group consisting of fillers, binders, lubricants, disintegrants, surfactants, coloring agents, plasticizers, opacifiers, and mixtures thereof.
6. The pharmaceutical composition according to any of the preceding claims, wherein the composition is selected from the group consisting of tablets, capsules, pellets, pills, caplets, and granules.
7. The pharmaceutical composition of claim 6, wherein the composition is a tablet.
8. The pharmaceutical composition of claim 7, wherein the tablet is further coated with one or more non-functional coatings.
9. A process for the preparation of a pharmaceutical composition of vilazodone, wherein the process comprises the steps of:
(a) blending micronized vilazodone with one or more pharmaceutically
acceptable excipients;
(b) directly compressing the blend of step (a) to form tablets; and
(c) film coating the tablets obtained from step (b).
10. A process for the preparation of a pharmaceutical composition of vilazodone, wherein the process comprises the steps of:
(a) blending micronized vilazodone with one or more pharmaceutically
acceptable excipients;
(b) granulating the blend of step (a);
(c) blending the granules of step (b) with one or more pharmaceutically
acceptable excipients;
(d) compressing the blend of step (c) to form tablets; and
(e) film coating the tablets obtained from step (d).
11. A method of treating and/or preventing a central nervous system disorder by administering a pharmaceutical composition comprising micronized particles of vilazodone and one or more pharmaceutically acceptable excipients.
12. The method according to claim 11, wherein the central nervous system disorder is selected from the group consisting of major depressive disorder, anxiety disorders, bipolar disorders, mania, dementia, eating disorders, sleeping disorders, psychiatric disorders, cerebral infarct, tension, and any combination thereof.
13. The method according to claim 12, wherein the central nervous system disorder is major depressive disorder.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN2349DE2013 | 2013-08-06 | ||
| IN2349/DEL/2013 | 2013-08-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015019237A1 true WO2015019237A1 (en) | 2015-02-12 |
Family
ID=51359416
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2014/063484 Ceased WO2015019237A1 (en) | 2013-08-06 | 2014-07-28 | Pharmaceutical composition of vilazodone |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2015019237A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106580914A (en) * | 2017-02-27 | 2017-04-26 | 佛山市弘泰药物研发有限公司 | Vilazodone hydrochloride softgel and preparation method thereof |
| EP3360543A1 (en) | 2017-02-13 | 2018-08-15 | Sanovel Ilac Sanayi ve Ticaret A.S. | Pharmaceutical compositions of vilazodone hydrochloride |
| EP3360542A1 (en) * | 2017-02-13 | 2018-08-15 | Sanovel Ilac Sanayi ve Ticaret A.S. | Tablet forms of vilazodone hydrochloride |
| US10688090B2 (en) | 2016-11-02 | 2020-06-23 | Sunshine Lake Pharma Co., Ltd. | Vilazodone inclusion complexes, compositions and preparation thereof |
| EP4268802A4 (en) * | 2020-12-23 | 2024-11-06 | Shanghai Yonsun Biotechnology Co., Ltd. | PHARMACEUTICAL VILAZODONE COMPOSITION, MANUFACTURING PROCESS THEREOF AND USE THEREOF |
| US12171761B2 (en) | 2016-11-02 | 2024-12-24 | Sunshine Lake Pharma Co., Ltd. | Vilazodone inclusion complexes, compositions and preparation thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5532241A (en) | 1993-09-30 | 1996-07-02 | Merck Patent Gesellschaft Mit Beschrankter Haftung | Piperidines and piperazines |
| US7834020B2 (en) | 2001-06-19 | 2010-11-16 | Merck Patent Gesellschaft | Polymorphic forms of 1-′4-(5-cyanoindol-3-yl)butyl-4-(2-carbamoylbenzofuran-5-yl) piperazine hydrochloride |
| WO2012131706A1 (en) | 2011-03-20 | 2012-10-04 | Cadila Healthcare Limited | Amorphous form of vilazodone hydrochloride and process for its preparation |
| WO2013078361A1 (en) | 2011-11-23 | 2013-05-30 | Assia Chemical Industries Ltd. | Solid state forms of vilazodone and vilazodone hydrochloride |
-
2014
- 2014-07-28 WO PCT/IB2014/063484 patent/WO2015019237A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5532241A (en) | 1993-09-30 | 1996-07-02 | Merck Patent Gesellschaft Mit Beschrankter Haftung | Piperidines and piperazines |
| US7834020B2 (en) | 2001-06-19 | 2010-11-16 | Merck Patent Gesellschaft | Polymorphic forms of 1-′4-(5-cyanoindol-3-yl)butyl-4-(2-carbamoylbenzofuran-5-yl) piperazine hydrochloride |
| WO2012131706A1 (en) | 2011-03-20 | 2012-10-04 | Cadila Healthcare Limited | Amorphous form of vilazodone hydrochloride and process for its preparation |
| WO2013078361A1 (en) | 2011-11-23 | 2013-05-30 | Assia Chemical Industries Ltd. | Solid state forms of vilazodone and vilazodone hydrochloride |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10688090B2 (en) | 2016-11-02 | 2020-06-23 | Sunshine Lake Pharma Co., Ltd. | Vilazodone inclusion complexes, compositions and preparation thereof |
| US11517569B2 (en) | 2016-11-02 | 2022-12-06 | Sunshine Lake Pharma Co., Ltd. | Vilazodone inclusion complexes, compositions and preparation thereof |
| US12171761B2 (en) | 2016-11-02 | 2024-12-24 | Sunshine Lake Pharma Co., Ltd. | Vilazodone inclusion complexes, compositions and preparation thereof |
| EP3360543A1 (en) | 2017-02-13 | 2018-08-15 | Sanovel Ilac Sanayi ve Ticaret A.S. | Pharmaceutical compositions of vilazodone hydrochloride |
| EP3360542A1 (en) * | 2017-02-13 | 2018-08-15 | Sanovel Ilac Sanayi ve Ticaret A.S. | Tablet forms of vilazodone hydrochloride |
| CN106580914A (en) * | 2017-02-27 | 2017-04-26 | 佛山市弘泰药物研发有限公司 | Vilazodone hydrochloride softgel and preparation method thereof |
| EP4268802A4 (en) * | 2020-12-23 | 2024-11-06 | Shanghai Yonsun Biotechnology Co., Ltd. | PHARMACEUTICAL VILAZODONE COMPOSITION, MANUFACTURING PROCESS THEREOF AND USE THEREOF |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| HU186078B (en) | Process for producing pharmaceutical compositions for peroral application containing dipyridamol | |
| KR20160101720A (en) | Pharmaceutical compositions comprising azd9291 | |
| WO2015019237A1 (en) | Pharmaceutical composition of vilazodone | |
| JP2010519201A (en) | Controlled release formulation containing cilostazol and method for producing the same | |
| WO2012156981A1 (en) | Pharmaceutical compositions of lurasidone | |
| WO2013022924A1 (en) | Pharmaceutical formulations | |
| JP2022518703A (en) | Emission-regulated tablet formulation containing a phosphodiesterase inhibitor | |
| JP2015500853A (en) | Immediate release multi-unit pellet system | |
| JP2025120468A (en) | Istradefylline preparations | |
| WO2010111264A2 (en) | Rasagiline formulations | |
| EP2384746A2 (en) | Dual release oral tablet compositions of dexlansoprazole | |
| WO2005097070A1 (en) | Solid pharmaceutical preparation with improved stability and process for producing the same | |
| JP2005533774A (en) | Divalproexodium release retardant | |
| WO2015019256A1 (en) | Pharmaceutical composition of vilazodone and processes of preparation thereof | |
| WO2016139683A2 (en) | Pharmaceutical compositions of lurasidone and process for preparing the same | |
| EP1901714A2 (en) | Pharmaceutical dosage forms comprising escitalopram in form of granules | |
| US20110217369A1 (en) | Fenofibrate compositions | |
| CN113795243A (en) | Pharmaceutical composition containing acetaminophen and ibuprofen | |
| US20100172982A1 (en) | Sustained release formulations of divalproex sodium | |
| WO2011000518A1 (en) | Pharmaceutical composition comprising desloratadine | |
| AU2018419112B2 (en) | Instant release pharmaceutical preparation of anticoagulant and preparation method therefor | |
| US20120121700A1 (en) | Pharmaceutical formulations comprising valganciclovir | |
| DE202021100039U1 (en) | Ethyl cellulose-coated particles containing a salt of tapentadol and phosphoric acid | |
| WO2015198225A1 (en) | Oral pharmaceutical composition of tofacitinib | |
| WO2012050539A1 (en) | Pharmaceutical composition comprising eplerenone |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14752952 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14752952 Country of ref document: EP Kind code of ref document: A1 |