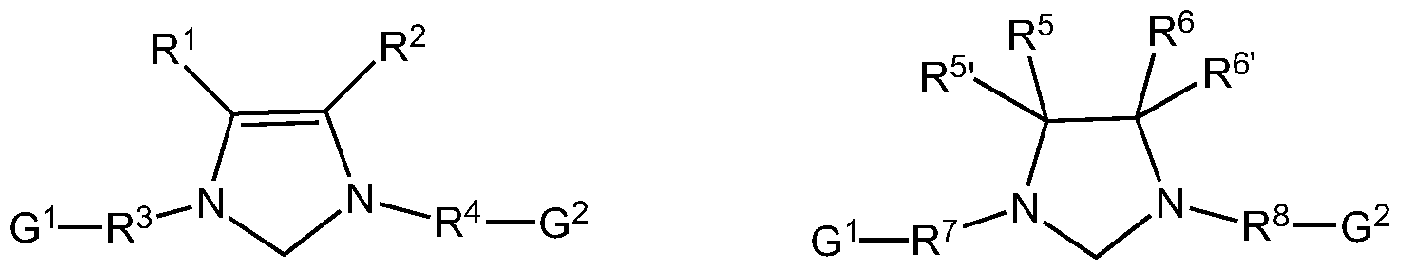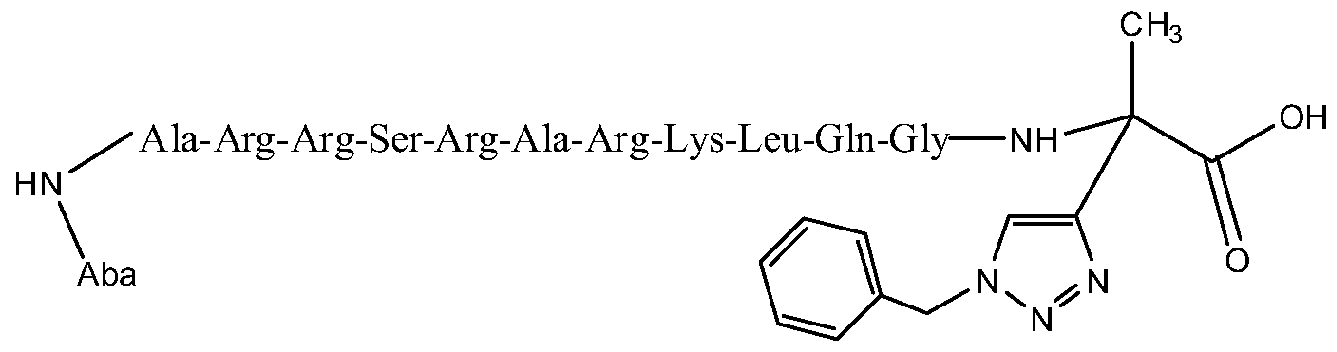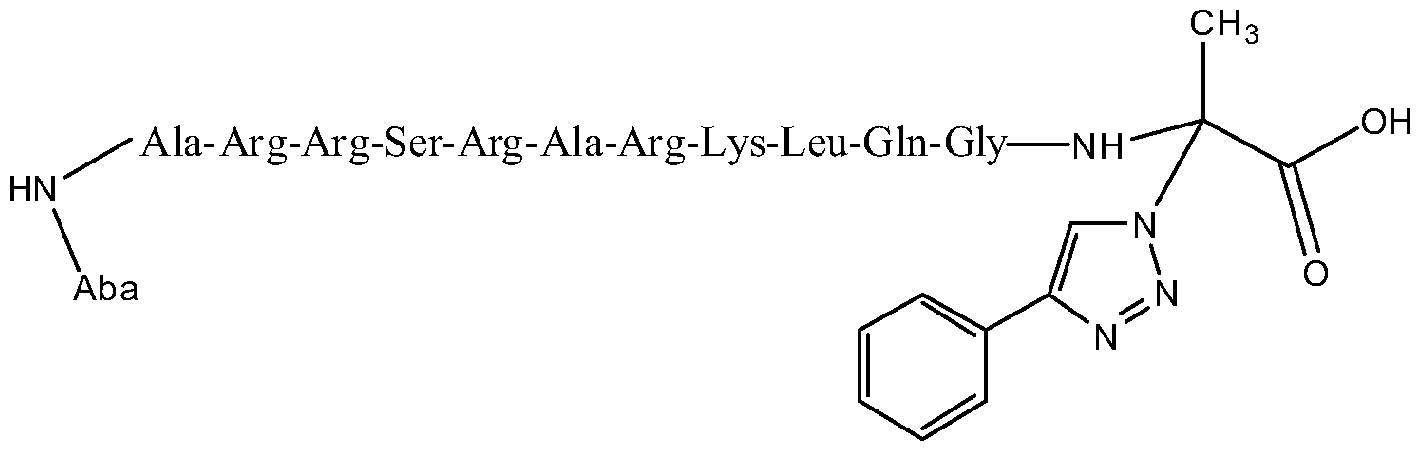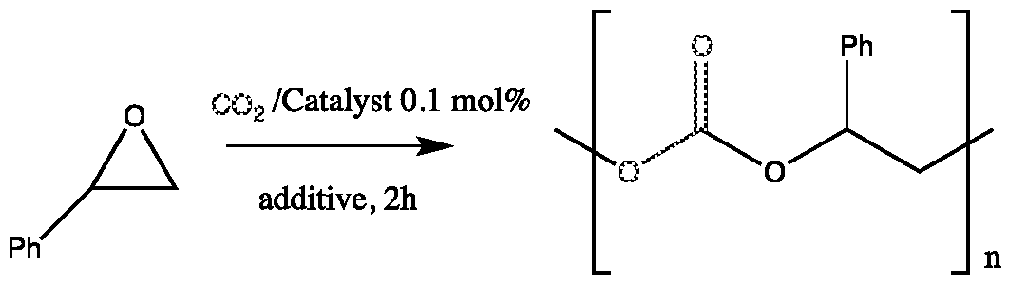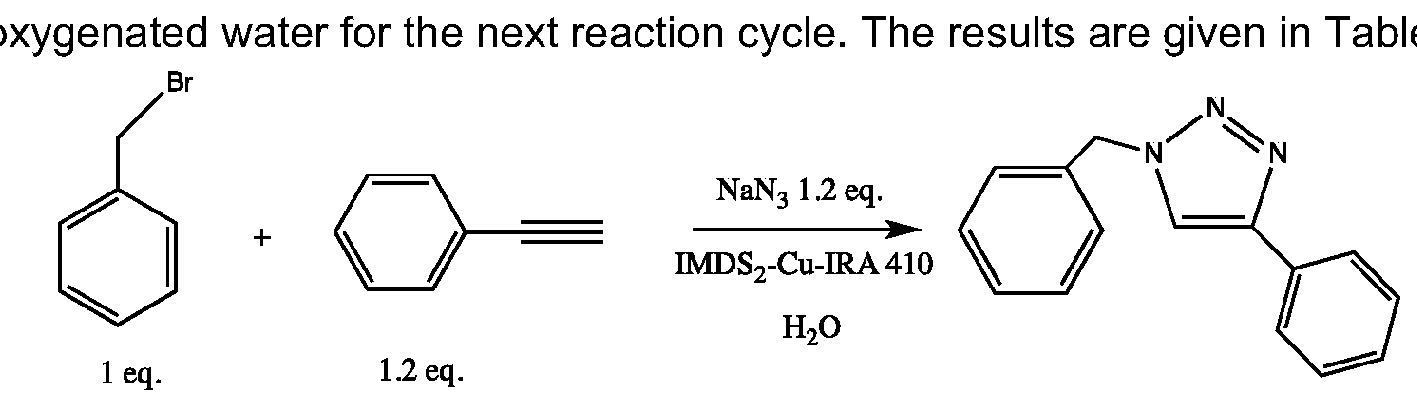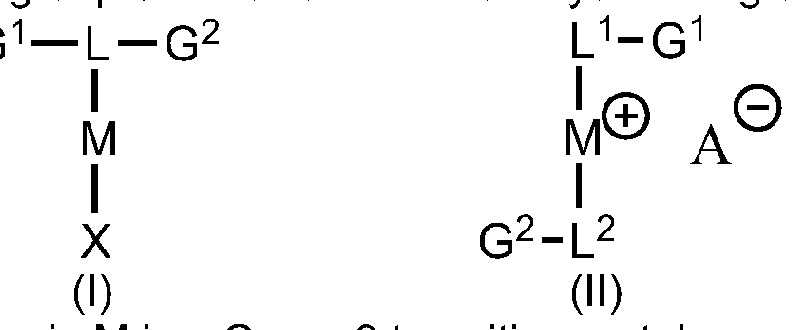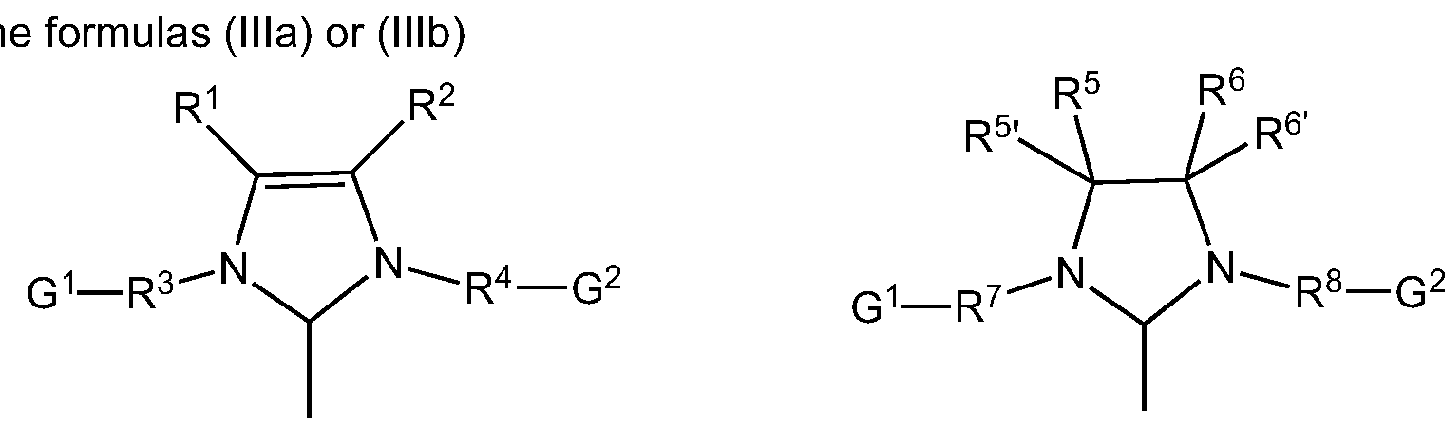WO2014172885A1 - Group 9 transition metal catalysts and process for use of same - Google Patents
Group 9 transition metal catalysts and process for use of same Download PDFInfo
- Publication number
- WO2014172885A1 WO2014172885A1 PCT/CN2013/074755 CN2013074755W WO2014172885A1 WO 2014172885 A1 WO2014172885 A1 WO 2014172885A1 CN 2013074755 W CN2013074755 W CN 2013074755W WO 2014172885 A1 WO2014172885 A1 WO 2014172885A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- alkyl
- catalyst
- substituted
- aryl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2265—Carbenes or carbynes, i.e.(image)
- B01J31/2269—Heterocyclic carbenes
- B01J31/2273—Heterocyclic carbenes with only nitrogen as heteroatomic ring members, e.g. 1,3-diarylimidazoline-2-ylidenes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/02—Catalysts comprising hydrides, coordination complexes or organic compounds containing organic compounds or metal hydrides
- B01J31/06—Catalysts comprising hydrides, coordination complexes or organic compounds containing organic compounds or metal hydrides containing polymers
- B01J31/08—Ion-exchange resins
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/04—1,2,3-Triazoles; Hydrogenated 1,2,3-triazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/04—1,2,3-Triazoles; Hydrogenated 1,2,3-triazoles
- C07D249/06—1,2,3-Triazoles; Hydrogenated 1,2,3-triazoles with aryl radicals directly attached to ring atoms
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2231/00—Catalytic reactions performed with catalysts classified in B01J31/00
- B01J2231/10—Polymerisation reactions involving at least dual use catalysts, e.g. for both oligomerisation and polymerisation
- B01J2231/14—Other (co) polymerisation, e.g. of lactides or epoxides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2231/00—Catalytic reactions performed with catalysts classified in B01J31/00
- B01J2231/30—Addition reactions at carbon centres, i.e. to either C-C or C-X multiple bonds
- B01J2231/32—Addition reactions to C=C or C-C triple bonds
- B01J2231/321—Hydroformylation, metalformylation, carbonylation or hydroaminomethylation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2231/00—Catalytic reactions performed with catalysts classified in B01J31/00
- B01J2231/30—Addition reactions at carbon centres, i.e. to either C-C or C-X multiple bonds
- B01J2231/32—Addition reactions to C=C or C-C triple bonds
- B01J2231/324—Cyclisations via conversion of C-C multiple to single or less multiple bonds, e.g. cycloadditions
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/10—Complexes comprising metals of Group I (IA or IB) as the central metal
- B01J2531/16—Copper
Definitions
- This invention relates generally to organometallic catalysts suitable for the CLICK reaction, and reactions involving carbon dioxide as substrate. More particularly catalyst compounds suitable azide-alkyne cyclo-addition, their synthesis and processes for the use thereof. As well catalyst compounds suitable for conversion of carbon dioxide generating cyclic or poly carbonates and carboxylic acids.
- the invention has utility in the fields of catalysis, organic synthesis, polymer chemistry, and industrial and fine chemicals chemistry.
- the reaction is not significantly affected by the steric and electronic properties of the groups attached to the azide and alkyne reactive centers.
- azides carrying a primary, secondary, or tertiary group; electron-deficient or electron-rich group; and aliphatic, aromatic, or hetero-aromatic substituent usually react well with variously substituted terminal alkynes.
- reaction is unaffected by water and by most organic and inorganic functional groups; thus, all but eliminating the need for protecting-group chemistry.
- un-catalyzed thermal cycloaddition of azides to alkynes usually requires prolonged heating and results in mixtures of the 1 ,4- and 1 ,5-disubstituted regioisomers.
- copper catalyzed Azide Alkyne Cyclization(AAC) produces only 1 ,4-disubstituted-1 ,2,3-triazoles at room temperature in excellent yields.
- the rate of the metal-catalyzed process is approximately 107 times that of the un-catalyzed version, 5 making the reaction conveniently fast in the temperature range of 0 to 25 °C. Furthermore, ligand-accelerated-catalysis effects 18 are also significant, resulting in further increases in the reaction rate.
- the 1 ,2,3-triazole unit that results from the reaction has several advantageous properties: (i) a high chemical stability (in general, being inert to severe hydrolytic, oxidizing, and reducing conditions, even at high temperature), (ii) a strong dipole moment (5.2-5.6 D), (iii) an aromatic character, and (iv) a good hydrogen-bond-accepting ability. 19,20 Thus, it can interact productively in several ways with biological molecules, and serve as a replacement for the amide linkage in some circumstances. A number of different copper(l) sources can be utilized in the reaction.
- Copper(l) salts (CuCI, Cul, CuBr, CuOAc) and coordination complexes (such as [Cu(CH 3 CN) 4 ]PF 6 , 16 (EtO) 3 P « Cul, 21 [Cu Pha ⁇ Br 22 ' 23 ) can be used directly.
- the latter have been particularly effective in organic solvents, in which cuprous salts have limited solubility.
- Cu(l) is thermodynamically unstable and can be relatively easily oxidized to catalytically inactive Cu(ll).
- the standard potential of the Cu2+/Cu+ couple is 159 mV, but can vary widely with the solvent and the ligand environment of the metal.
- Cu(ll) itself, is an oxidant and can mediate the oxidative alkyne coupling reactions mentioned above, thereby impairing the efficiency of the cycloaddition and resulting in the formation of undesired byproducts. Therefore, when a copper(l) catalyst is used directly, whether by itself or in conjunction with amine ligands, exclusion of oxygen may be required to prevent these complications.
- a sacrificial reducing agent for example ascorbate
- a copper(ll) salt e.g. copper(ll) sulfate pentahydrate or copper(ll) acetate. 17
- Catalytic amounts of Cu(l) can also be introduced in the reaction through com-proportionation of Cu(ll) and Cu(0), thus further simplifying the experimental procedure.
- Copper nanoclusters which are easily obtained and are air-stable, 24 and copper/cuprous oxide nanoparticles 25 have also shown good catalytic activity, although this procedure requires longer reaction times.
- TBTA ligand tris[(1-benzyl-1 H-1 ,2,3-triazol-4-yl)methyl]amine
- the catalysts of this invention can also be applied to synthesize cyclic- and poly-carbonate products. Synthetic procedures which utilize C0 2 as a carbon feedstock are currently generating great interest within the scientific community 28 as these may offer viable alternative routes toward various organic structures that are currently derived from fossil fuel based resources 29 .
- the catalysts of this invention can also be applied to synthesize carboxylic acids using C0 2 .
- Carboxylic acids are one of the most important types of compounds in medicinal chemistry and also in fine-chemicals synthesis 35, 36 .
- the direct carboxylation of carbon nucleophiles using C0 2 as the electrophile is the most attractive and straightforward method 35,36 .
- the formation of a stable C— C bond is desired for C0 2 fixation and remains the most challenging aspect thus far.
- this type of reaction is facilitated by the insertion of C0 2 into a metal-carbon bond 35"37 . Widespread use of these methods is limited by the synthesis organometallic reagents as precursors and the restricted substrate scope.
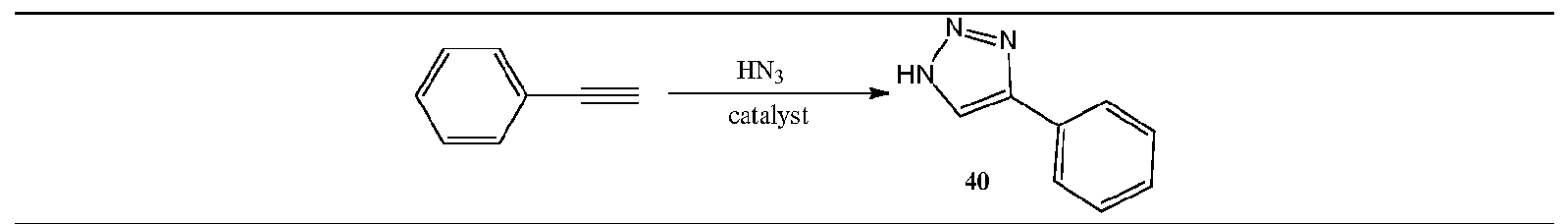
- the present invention introduces the use of water-soluble complexes for the Azide Alkyne Cycloaddition preferably in aqueous media, without the need of external bases or extra ligands and using low catalyst loadings, in which the synthesis of triazoles using either internal or terminal alkynes, as well as acetylene gas with organic azides is aimed.
- the present invention is not limited to organic azides, since it is possible to use /n-s/ft/generated HN 3 (from inorganic azides) for the synthesis of 4- and 4,5-substituted triazoles under aqueous conditions by using these catalysts. Furthermore, the efficiency of these catalysts is also proven by its excellent performance in peptidomimetic chemistry.
- the present invention also relates to an easy applicable catalyst synthesis and the application in different click-processes.
- the organometallic catalyst compound of the present invention can be prepared by contacting a Group 9metal precursor compound with atwo-electron donating ligand bearing two ionic groups or with a two-electron donating ligand bearing one ionic group.
- M is a Group 9 transition metal
- X is preferably an anionic ligand.
- L 1 and L 2 represent identical or different ligands, preferably electron donors.
- G 1 and G 2 represent an ionic group.
- a " is an anion, preferable a non-coordinating
- the invention provides novel organometallic compounds according to the above structure.
- the invention provides a method for performing a catalytic cycloaddition reaction comprising contacting at least one alkyneand an azide compound with the "click' -catalyst of the invention.
- a substituent encompasses a single substituent as well as two or more substituents, and the like.
- substituted means that a hydrogen atom or an atom is replaced by a specified group or an atom, and the valence of the atom indicated is not exceeded and the substitution leads to a stable compound.
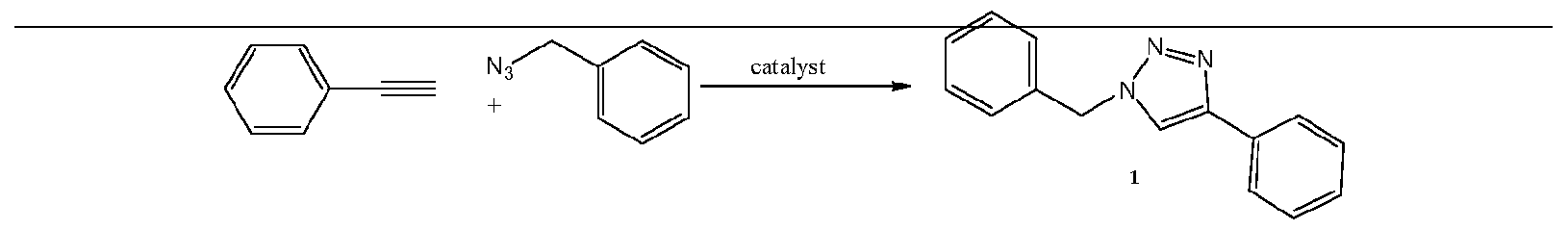
- Figure 1 depicts the comparison of bis(NHC)-Cu catalysts for the click reaction between phenylacetylene and benzylazide.
- alkyl refers to a linear, branched, or cyclic saturated hydrocarbon group typically although not necessarily containing 1 to about 24 carbon atoms, preferably 1 to about 12 carbon atoms, such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, t-butyl, octyl, decyl, and the like, as well as cycloalkyl groups such as cyclopentyl, cyclohexyl and the like. Generally, although again not necessarily, alkyl groups herein contain 1 to about 12 carbon atoms.
- C i-C 6 -alkyl
- cycloalkyl intends a cyclic alkyl group, typically having 3 to 8 carbon atoms.
- substituted alkyl refers to alkyl substituted with one or more substituent groups
- heteroatom-containing alkyl and “heteroalkyl” refer to alkyl in which at least one carbon atom is replaced with a heteroatom. If not otherwise indicated, the term “alkyl” include linear, branched, cyclic, unsubstituted, substituted, and/or heteroatom-containing alkyl.
- alkylene refers to a difunctional linear, branched, or cyclic alkyl group, where "alkyl” is as defined above.
- alkenyl refers to a linear, branched, or cyclic hydrocarbon group of 2 to about 24 carbon atoms containing at least one double bond, such as ethenyl, n-propenyl, isopropenyl, n-butenyl, isobutenyl, octenyl, decenyl, tetradecenyl, hexadecenyl, eicosenyl, and the like.
- Preferred alkenyl groups herein contain 2 to about 12 carbon atoms.
- cycloalkenyl intends a cyclic alkenyl group, preferably having 5 to 8 carbon atoms.
- substituted alkenyl refers to alkenyl substituted with one or more substituent groups
- heteroatom-containing alkenyl and “heteroalkenyl” refer to alkenyl in which at least one carbon atom is replaced with a heteroatom. If not otherwise indicated, the term “alkenyl” include linear, branched, cyclic, unsubstituted, substituted, and/or heteroatom-containing alkenyl.
- alkenylene refers to a difunctional linear, branched, or cyclic alkenyl group, where "alkenyl” is as defined above.
- alkynyl refers to a linear or branched hydrocarbon group of 2 to about 24 carbon atoms containing at least one triple bond, such as ethynyl, n-propynyl, and the like. Preferred alkynyl groups herein contain 2 to about 12 carbon atoms.
- substituted alkynyl refers to alkynyl substituted with one or more substituent groups, and the terms “heteroatom-containing alkynyl” and “heteroalkynyl” refer to alkynyl in which at least one carbon atom is replaced with a heteroatom. Ifnot otherwise indicated, the term “alkynyl” include linear, branched, unsubstituted, substituted, and/or heteroatom-containing alkynyl respectively.
- alkoxy intends an alkyl group bound through a single, terminal ether linkage; that is, an "alkoxy” group may be represented as -O-alkyl where alkyl is as defined above.
- alkenyloxy refers to an alkenyl group bound through a single, terminal ether linkage
- alkynyloxy refers to an alkynyl group bound through a single, terminal ether linkage.
- aryl refers to an aromatic substituent containing a single aromatic ring or multiple aromatic rings that are fused together, directly linked, or indirectly linked (such that the different aromatic rings are bound to a common group such as a methylene or ethylene moiety).
- Preferred aryl groups contain 5 to 24 carbon atoms, and particularly preferred aryl groups contain 5 to 14 carbon atoms.
- Exemplary aryl groups contain one aromatic ring or two fused or linked aromatic rings, e.g., phenyl, naphthyl, biphenyl, diphenylether, diphenylamine, benzophenone, and the like.
- Substituted aryl refers to an aryl moiety substituted with one or more substituent groups
- heteroatom-containing aryl and “heteroaryl” refer to aryl substituents in which at least one carbon atom is replaced with a heteroatom, as will be described in further detail infra.
- aryloxy refers to an aryl group bound through a single, terminal ether linkage, wherein "aryl” is as defined above.
- An "aryloxy” group may be represented as -O-aryl where aryl is as defined above.
- Preferred aryloxy groups contain 5 to 24 carbon atoms, and particularly preferred aryloxy groups contain 5 to 14 carbon atoms.
- aryloxy groups include, without limitation, phenoxy, o-halo-phenoxy, m-halo-phenoxy, p-halo-phenoxy, o-methoxyphenoxy, m-methoxy-phenoxy, p-methoxy-phenoxy, 2,4-dimethoxy-phenoxy, 3,4,5-trimethoxy-phenoxy, and the like.
- alkaryl refers to an aryl group with an alkyl substituent
- aralkyl refers to an alkyl group with an aryl substituent, wherein “aryl” and “alkyl” are as defined above.
- Preferred alkaryl and aralkyl groups contain 6 to 24 carbon atoms.
- Alkaryl groups include, but not limit to, for example, p-methylphenyl, 2,4-dimethylphenyl, p-cyclohexylphenyl, 2, 7 -dimethylnaphthyl, 7 -cyclooctyl naphthyl, 3-ethyl-cyclopenta-1 ,4-diene, and the like.
- aralkyl groups include, without limitation, benzyl, 2-phenyl-ethyl, 3-phenyl-propyl, 4-phenyl-butyl, 5-phenyl-pentyl, 4-phenylcyclohexyl, 4-benzylcyclohexyl, 4-phenylcyclohexylmethyl, 4-benzylcyclohexylmethyl, and the like.
- alkaryloxy and aralkyloxy refer to substituents of the formula -OR wherein R is alkaryl or aralkyl, respectively, as just defined.
- acyl refers to substituents having the formula -(CO)-alkyl, -(CO)-aryl, or -(CO)-aralkyl
- acyloxy refers to substituents having the formula -0(CO)-alkyl, -0(CO)aryl, or -0(CO)-aralkyl, wherein "alkyl,” “aryl, and “aralkyl” are as defined above.
- cyclic and ring refer to alicyclic or aromatic groups that may or may not be substituted and/or heteroatom containing, and that may be monocyclic, bicyclic, or polycyclic.
- alicyclic is used in the conventional sense to refer to an aliphatic cyclic moiety, as opposed to an aromatic cyclic moiety, and may be monocyclic, bicyclic, or polycyclic.
- halo and halogen are used in the conventional sense to refer to a chloro, bromo, fluoro, or iodo substituent.
- Hydrocarbyl refers to univalent hydrocarbyl radicals containing 1 to about 30 carbon atoms, preferably 1 to about 24 carbon atoms, most preferably 1 to about 12 carbon atoms, including linear, branched, cyclic, saturated, and unsaturated species, such as alkyl groups, alkenyl groups, aryl groups, and the like.
- hydrocarbylene intends a divalent hydrocarbyl moiety containing 1 to about 30 carbon atoms, preferably 1 to about 24 carbon atoms, most preferably 1 to about 12 carbon atoms, including linear, branched, cyclic, saturated and unsaturated species.
- Substituted hydrocarbyl refers to hydrocarbyl substituted with one or more substituent groups
- heteroatom-containing hydrocarbyl and “heterohydrocarbyl” refer to hydrocarbyl in which at least one carbon atom is replaced with a heteroatom
- substituted hydrocarbylene refers to hydrocarbylene substituted with one or more substituent groups
- heteroatomcontaining hydrocarbylene and “heterohydrocarbylene” refer to hydrocarbylene in which at least one carbon atom is replaced with a heteroatom.
- hydrocarbyl and hydrocarbylene are to be interpreted as including substituted and/or heteroatom-containing hydrocarbyl and hydrocarbylene moieties, respectively.
- heteroatom-containing refers to a hydrocarbon molecule or a hydrocarbyl molecular fragment in which one or more carbon atoms is replaced with an atom other than carbon, e.g., nitrogen, oxygen, sulfur, phosphorus or silicon, typically nitrogen, oxygen or sulfur.
- heteroalkyl refers to an alkyl substituent that is heteroatom-containing
- heterocyclic refers to a cyclic substituent that is heteroatom-containing
- heteroaryl and “heteroaromatic” respectively refer to "aryl” and "aromatic” substituents that are heteroatom-containing, and the like.
- heterocyclic group or compound may or may not be aromatic, and further that “heterocycles” may be monocyclic, bicyclic, or polycyclic as described above with respect to the term "aryl.”
- heteroalkyl groups include alkoxyalkyl, alkylsulfanyl-substituted alkyl, N-alkylated amino alkyl, and the like.
- heteroaryl substituents include pyrrolyl, pyrrolidinyl, pyridinyl, quinolinyl, indolyl, pyrimidinyl, imidazolyl, 1 ,2,4-triazolyl, tetrazolyl, etc., and examples of heteroatom containing alicyclic groups are pyrrolidino, morpholino, piperazino, piperidino, etc.
- substituted as in “substituted hydrocarbyl”, “substituted alkyl”, “substituted aryl”, and the like, as alluded to in some of the aforementioned definitions, is meant that in the hydrocarbyl, alkyl, aryl, or other moiety, at least one hydrogen atom bound to a carbon (or other) atom is replaced with one or more non-hydrogen substituents.
- substituents include, without limitation: functional groups such as halo, hydroxyl, sulfhydryl, C 1 -C24 alkoxy, C 2 -C 2 4 alkenyloxy, C 2 -C 2 4 alkynyloxy, C 5 -C 24 aryloxy, C 6 -C 24 aralkyloxy, C 6 -C 24 alkaryloxy, acyl (including C 2 C 24 alkylcarbonyl (-CO-alkyl) and C 6 -C 24 arylcarbonyl (-CO-aryl)), acyloxy (-O-acyl, including C 2 C 2 alkylcarbonyloxy (-O-CO-alkyl) and C 6 -C 24 arylcarbonyloxy (-O-CO-aryl)), C 2 C 24 alkoxycarbonyl (-(CO)-O-alkyl), C 6 -C 2 aryloxycarbonyl (-(CO)-aryl), halocarbony
- the aforementioned functional groups may, if a particular group permits, be further substituted with one or more additional functional groups or with one or more hydrocarbyl moieties such as those specifically enumerated above.
- the above-mentioned hydrocarbyl moieties may be further substituted with one or more functional groups or additional hydrocarbyl moieties such as those specifically enumerated.
- the present invention comprises the use of water-soluble group 9transition metal complexes for the AAC preferably in aqueous media, without the need of external bases or extra ligands and using low catalyst loadings, in which the synthesis of triazoles using either internal or terminal alkynes, as well as acetylene gas with organic azides is aimed.
- the present invention is not limited to organic azides, since it is possible to use in-situ generated HN 3 (from inorganic azides) for the synthesis of 4- and 4,5-substituted triazoles under aqueous conditions by using these catalysts. Furthermore, the efficiency of these catalysts is also proven by its excellent performance in peptidomimetic chemistry.
- this invention comprises an azide and an alkyne, in which the catalysts have either of the following structures (I) or (II)
- X is an anionic ligand.
- X is for example, hydrogen, halogen, pseudohalogen, straight-chain or branched C1-C30 alkyl, C 6 -C 24 aryl, CrC 20 alkoxy, C 6 -C 2 aryloxy, C 3 -C 20 alkyl diketonate, C 6 -C 24 aryl diketonate, Ci-C 2 o carboxylate, Ci-C 2 o alkylsulfonate, C6-C 24 aryl sulfonate, Ci-C 2 o alkyl thiol, C6-C 24 aryl thiol, Ci-C 2 o alkylsulfonyl or Ci-C 2 o alkylsulfinyl-radical.
- X may further be substituted by one or more additional residues, for example by halogen, preferably fluorine, CrC 20 alkyl, Ci-C 20 -alkoxy or C 6 -C 24 aryl, where these groups may optionally be in turn be substituted by one or more substituents from the group comprising halogen, preferable fluorine, C 1 -C5 alkyl, C 1 -C5 alkoxy, and phenyl.
- halogen preferably fluorine, CrC 20 alkyl, Ci-C 20 -alkoxy or C 6 -C 24 aryl, where these groups may optionally be in turn be substituted by one or more substituents from the group comprising halogen, preferable fluorine, C 1 -C5 alkyl, C 1 -C5 alkoxy, and phenyl.
- X denote a halogen, in particular, fluorine, chlorine, bromine or iodine, benzoate, C 1 -C5 carboxylate, C 1 -C5 alkyl, phenoxy, C 1 -C5 alkoxy, C 1 -C5 alkyl thiol, C6-C24 arylthiol, C6-C 24 aryl or C 1 -C5 alkyl sulfonate.
- halogen in particular, fluorine, chlorine, bromine or iodine
- X is fluorine, chlorine, bromine or iodine, PhO (phenoxy), C 6 F 5 0 (pentafluorophenoxy).
- G 1 and G 2 are identical or different functional groups, preferably represent ionic groups.
- G is an ionic group or contains an ionic group
- the ionic group can be cationic or anionic.
- Preferable groups are e.g. quaternary ammonium, carboxylate, sulphonate (-SO 3 " ), -OSO 3 " , -PO 3 " , OPO 3 " .
- the ionic group bears a counter ionto neutralize the charge, in case the ionic group is a cation the counter ion will be anionic, in case the ionic group is an anion the counter ionwill be cationic.
- L represents an electron donor
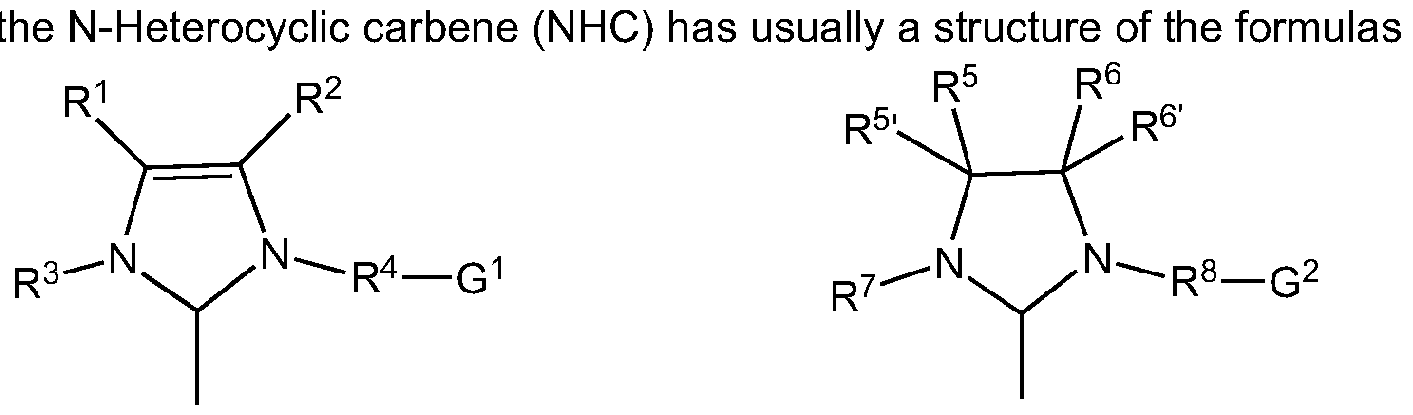
- L represents a N-Heterocyclic carbene ("NHC”), substituted NHC, or cyclic alkyl amino carbene (CAAC) or substituted CAAC.
- NHC N-Heterocyclic carbene
- CAAC cyclic alkyl amino carbene
- R 1 - R 8 , R 5 , R 6 are identical or different and are hydrogen, halogen, hydroxyl, aldehyde, keto, thiol, CF 3 , nitro, nitroso, cyano, thiocyano, isocyanates, carbodiimide, carbamate, thiocarbamate, dithiocarbamate, amino, amido, imino, ammonium, silyl, acyl, acyloxy or represents alkyl, cycloalkyl, alkenyl, cycloalkenyl, substituted alkenyl, heteroalkenyl, heteroatom-containing alkynyl, alkenylene, alkynyl, substituted alkynyl, aryl, substituted aryl, heteroaryl, carboxylate, alkoxy, alkenyloxy, alkynyloxy, aryloxy, alkaryl, aralkyl, alkaryloxy, aralkyloxy, alkoxycarbon
- one or more of the radicals R 1 - R 8 , R 5 , R 6 independently of one another can be substituted by one or more substituents, preferably straight or branched C 1 -C 10 alkyl, C 3 -C 8 cycloalkyl, C 1 -C 10 alkoxy or C 6 -C 2 4 aryl, where these aforementioned substituents may in turn be substituted by one or more radicals, preferably selected from the group comprising halogen, especially chlorine or bromine, C 1 -C5 alkyl, C 1 -C5 alkoxy and phenyl.
- R 1 , R 2 , R 5 , R 5 , R 6 and R 6 are independently of one another denote hydrogen, C 6 -C 2 4-aryl, particularly preferably phenyl, straight or branched C1-C10 alkyl, particularly preferably propyl or butyl, or together with the inclusion of the carbon atoms to which they are attached form a cycloalkyi or aryl radical, where all the abovementioned radicals are optionally substituted may be substituted by one or more further radicals selected from the group comprising straight or branched Ci- C10 alkyl, C1-C10 alkoxy, C 6 -C 2 4 aryl, and a functional group selected from the group consisting of hydroxy, thiol, thioether, ketone, aldehyde, ester, ether, amine, imine, amide, nitro, carboxylic acid, disulf
- the catalysts of the general formula (I) having one N-Heterocyclic carbene (NHC) as ligand L where the radicals R 3 , R 4 , R 7 and R 8 are identical or different and are straight or branched C C 0 alkyl, particularly preferably / ' -propyl or neopentyl, C 3 -C 10 cycloalkyi, preferably adamantyl, C6-C24 aryl, particularly preferably phenyl.
- N-Heterocyclic carbene N-Heterocyclic carbene
- the above-mentioned residues are substituted as the meanings of R 3 , R 4 , R 7 and R 8 by one or more further radicals selected from the group comprising straight or branched C1-C5 alkyl, especially methyl, C1-C5 alkoxy, aryl and a functional group selected from the group consisting of hydroxy, thiol, thioether, ketone, aldehyde, ester, ether, amine imine, amide, nitro, carboxylic acid, disulfide, carbonate, isocyanate, carbodiimide, carboalkoxy, carbamate, and halogen.
- one or more further radicals selected from the group comprising straight or branched C1-C5 alkyl, especially methyl, C1-C5 alkoxy, aryl and a functional group selected from the group consisting of hydroxy, thiol, thioether, ketone, aldehyde, ester, ether, amine imine,
- R 3 , R 4 , R 7 and R 8 are aryl, in each case two directly adjacent radicals from the group of R 3 , R 4 , R 7 and R 8 , including the ring carbon atoms to which they are attached by a cyclic bridging group, generating one or more cyclic structures, including aromatic structures.
- C1-C6 alkyl is, but not limited to, for example methyl, ethyl, n-propyl, /so-propyl, n-butyl, sec- butyl, feri-butyl, n-pentyl, 1 -methylbutyl, 2-methylbutyl, 3-methylbutyl, neo-pentyl, 1 -ethyl-propyl and n-hexyl.
- C 3 -C 8 cycloalkyi includes, but not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl and cyclooctyl.
- C 6 -C 2 4 aryl includes an aromatic radical having 6 to 24 skeletal carbon atoms.
- Preferred mono-, bi-or tricyclic carbocyclic aromatic radicals have 6 to 10 skeletal carbon atoms, for example but not limited to, phenyl, biphenyl, naphthyl, phenanthrenyl or anthracenyl.
- radicals R 3 , R 4 , R 7 and R 8 can be identical or different and denote / ' -propyl, neopentyl, adamantyl, mesityl or 2,6-diisopropylphenyl.
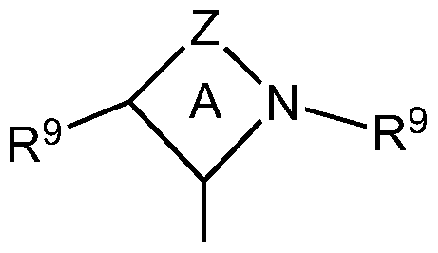
- N-Heterocyclic carbenes have the following structure (IVa) - (IVh).
- one of the N groups bound to the carbene in Formulae (Ilia) or (1Mb) is replaced with another heteroatom, preferably S, O or P, preferably an S heteroatom.
- Other useful N-heterocyclic carbenes include the compounds described in Hermann, W A. Chem. Eur. J 1996, 2, 772 and 1627; Enders, D. et al., Angew. Chem. Int. Ed. 1995, 34, 1021 ; Alder R. W., Angew. Chem. Int. Ed. 1996, 35, 1 121 ; and Bertrand, G. et al., Chem. Rev. 2000, 100, 39.
- CAACs cyclic alkyl amino carbenes
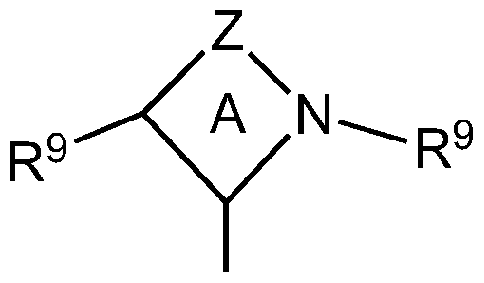
- ring A is a 4-, 5-, 6-, or 7-membered ring
- Z is a linking group comprising from one to four linked vertex atoms selected from the group comprising C, O, N, B, Al, P, S and Si with available valences optionally occupied by hydrogen, oxo or R- substituents
- R is independently selected from the group comprising Ci to C 12 hydrocarbyl groups, substituted Ci to C 12 hydrocarbyl groups, and halides
- each R 9 is independently a hydrocarbyl group or substituted hydrocarbyl group having 1 to 40 carbon atoms, preferably methyl, ethyl, propyl, butyl (including isobutyl and n-butyl), pentyl, cyclopentyl, hexyl, cyclohexyl, octyl, cyclooctyl, nonyl, decyl, cyclodecyl, dodecyl,
- CAACs include the compounds described in U.S. 7,312,331 and Bertrand et al, Angew. Chem. Int. Ed. 2005, 44, 7236-7239.
- L 1 and L 2 represent two electron donor ligands.
- L 1 and L 2 are the same or different and represent a N-Heterocyclic carbene ("NHC”), substituted NHC, or cyclic alkyl amino carbene (CAAC) or substituted CAAC.
- NHS N-Heterocyclic carbene
- CAAC cyclic alkyl amino carbene
- R - R , R and R have the same meanings as in the general formula (I).
- a " is an anion, preferable a weak-coordinating anion or a non-coordinating anion.
- suitable weak-coordinating and non-coordinating anions include, but are not limited to, [BF 4 ] ⁇ , [PF 6 ] ⁇ , and [CIO 4 ] " , Trifluoromethanesulfonate, B[3,5-(CF 3 ) 2 C 6 H 3 ] 4 ] ⁇ , tetrakis(pentafluorophenyl)borate, carborane anion.
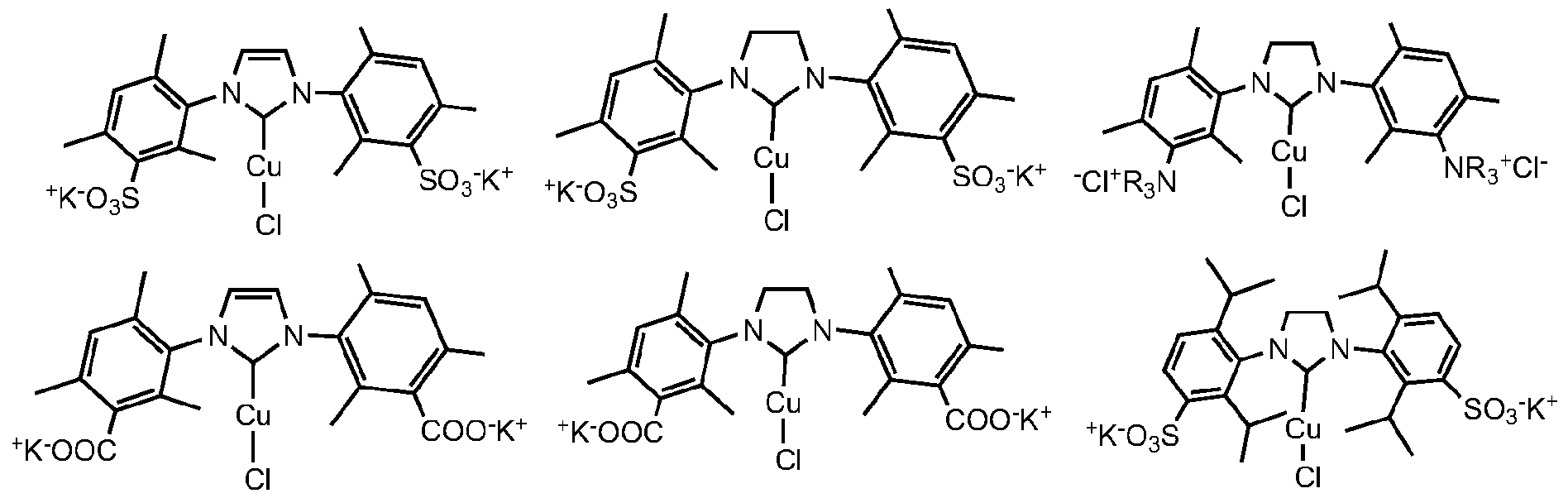
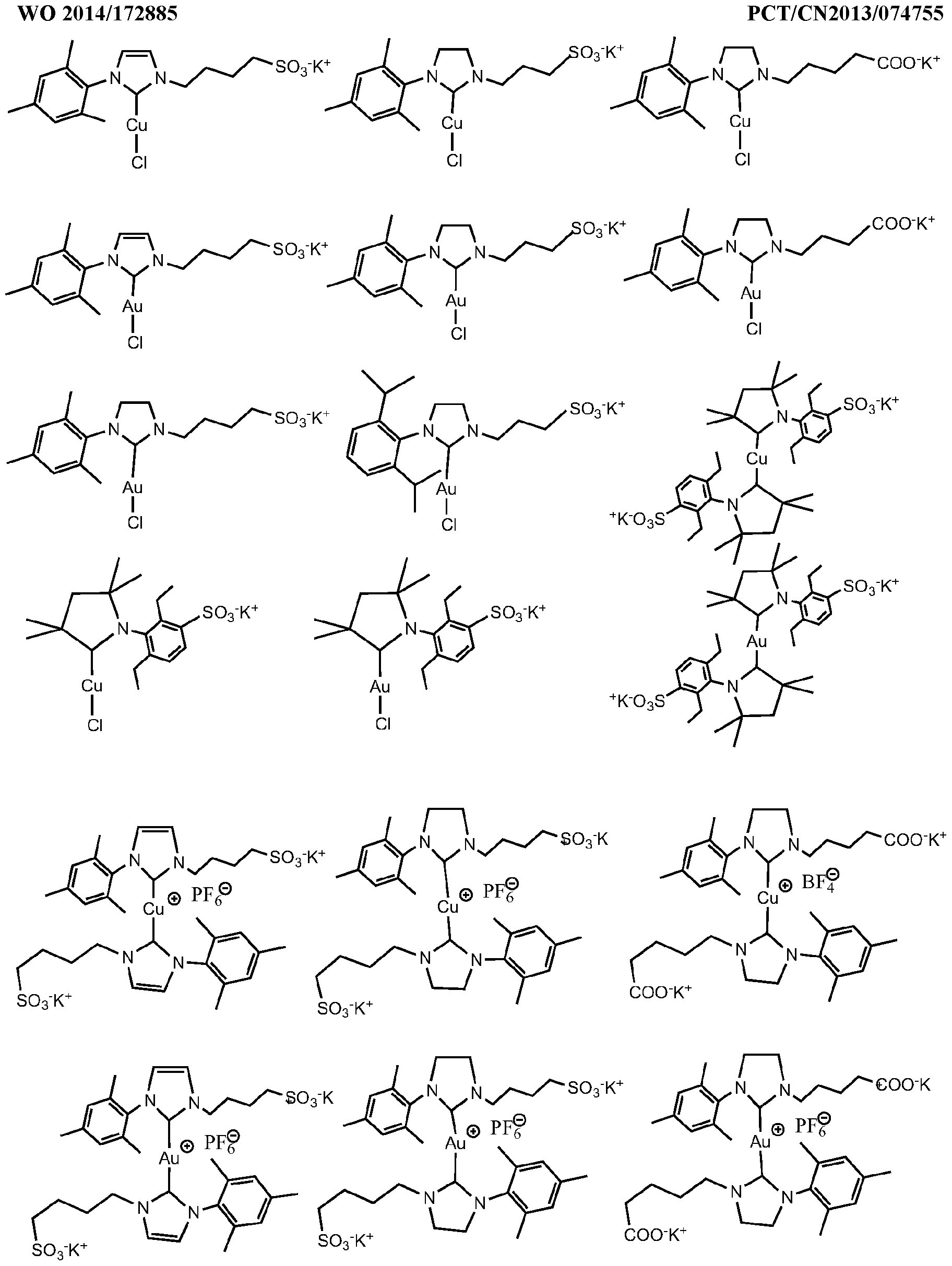
- the invention describes the synthesis of catalysts by using different metal(l) sources depending of the type of catalysts being synthesized.
- Type (I) catalysts based on Au(l) but not limited to can use the salts AuCI, AuBr, Aul.
- Type (II) catalysts require the use of the complexes such as but not limited to [Cu(CH 3 CN) 4 ]PF 6 , [Cu(CH 3 CN) 4 ]BF 4 , [Au(CH 3 CN) 4 ]PF 6 , [Au(CH 3 CN) 4 ]BF 4
- the catalyst compounds described in this invention may be synthesized by any methods known to those skilled in the art.
- R 10 - R 12 are identical or different and are hydrogen, or represents alkyl, cycloalkyl, alkenyl, cycloalkenyl, substituted alkenyl, heteroalkenyl, heteroatom-containing alkynyl, alkenylene, alkynyl, substituted alkynyl, aryl, substituted aryl, heteroaryl, carboxylate, alkoxy, alkenyloxy, alkynyloxy, aryloxy, alkaryl, aralkyl, alkaryloxy, aralkyloxy, alkoxycarbonyl, alkylammonium, alkylamino-, alkylthio-, arylthio, alkylsulfonyl, alkylsulfinyl, dialkylamino, alkylsilyl or alkoxysilyl, where these radicals may each optionally all be substituted by one or more aforementioned groups defined for
- one or more of the radicals R 10 - R 12 independently of one another can be substituted by one or more substituents, preferably straight or branched C-
- R 11 and R 12 independently of one another can be a metal, preferable a transition metal.
- multimetal entities can be prepared. Using this concept "a la carte” multimetal 1 ,2,3-triazole derivatives can be prepared.
- R 12 H .
- R 11 and R 12 are different from hydrogen.
- R 11 and R 12 are H.
- R 10 H
- Reactants containing the alkyne functionality can be aliphatic or aromatic holding various electronic and steric properties.
- Both terminal and internal alkynes can be used in the practice of this invention.
- Both types of alkyne can contain functional groups. Examples of these functional groups include but are not limited to: carbon-carbon double bonds, ether groups, ester groups, ketyl groups, hydroxyl groups, chlorine groups, fluorine groups, trihydrocarbylsilyl groups, nitrogen atoms (e.g., as amino groups) and the like.
- terminal alkynes typically have three to about twenty carbon atoms, and preferably about five to about twenty carbon atoms.
- the invention is also applicable to molecules holding multiple terminal alkyne groups in each endof the same molecule (e.g. but not limited to dendrimeric compounds) or holding multiple internal alkyne groups.
- Reactants containing the azide functionality include, but are not limited to: alkyl azides, ether azides, aryl azides and aralkyl azides.
- One or more functional groups can be included, e.g. cyano groups, nitro groups, amino groups, alkyl groups, halide groups, ester groups, ether groups, carboxylic groups, alkylsilyl groups, hydroxyl groups, alkenyl groups may be present in an aryl azide, in an aralkyl azide or in an alkyl azide.
- the alkyl portion of the alkyl azides can be branched, straight chain or cyclic group. Typically the alkyl azides have one to about twenty carbon atoms. In case of alkyl azides lower than 4 carbon atoms, a reaction procedure is followed, in which alkylazidesare generated/n-s/ft/ during the catalysis.
- the invention is also applicable to molecules holding multiple azide groups in each end of the same molecule, in which the final multi-azide is stable enough to perform the reactions under normal ambient conditions.
- the inorganic azide HN 3 this is generated in-situ by reaction of NaN 3 trimethylsilylazide or CaN 3 with a hydrogen source,
- peptides containing 2 to 20 aminoacid residues in which there is at least one azide group and/or one terminal alkyne group include, but are not limited to peptides containing 2 to 20 aminoacid residues in which there is at least one azide group and/or one terminal alkyne group.
- the peptides susceptible for chemical transformation by the catalysts of this invention may hold every aminoacid present in the proteins, including those susceptible to complexation and/or sensitive to oxidation.
- the catalysts for the chemical ligation of peptides can be used with chemically protected peptides or unprotected peptides preferably in an aqueous media.
- Solvent is not usually necessary in the processes of this invention, in which a neat process will be followed.
- non-neat processes generally it is recommended and preferred to conduct the processes of this invention in water, alcohol-water mixtures, DMSO-water mixtures, DMF-water mixtures or solvents different than water, preferably polar solvents like DMSO, acetone, acetonitrile, DMF or lower alcohols like methanol, ethanol or propanol.
- concentrations of the organic azide and the alkyne are each, independently, typically about 1 molar or higher, however, lower concentrations can be employed.
- concentrations of the organic azide and the alkyne are each, independently, typically about 1 molar or higher, however, lower concentrations can be employed.
- one mole of organic azide and one mole of alkyne are consumed for each mole of 1 ,2,3-triazole produced.
- the amounts of organic azide and alkyne are preferably such that the alkyne is in slight excess relative to the organic azide.
- acetylene gas one atmosphere of acetylene gas is used by bubbling it into the reaction vessel.
- the inorganic azide HN 3 preferable, but not limited to, 1.5 mol HN 3 per mol of alkyne is used.
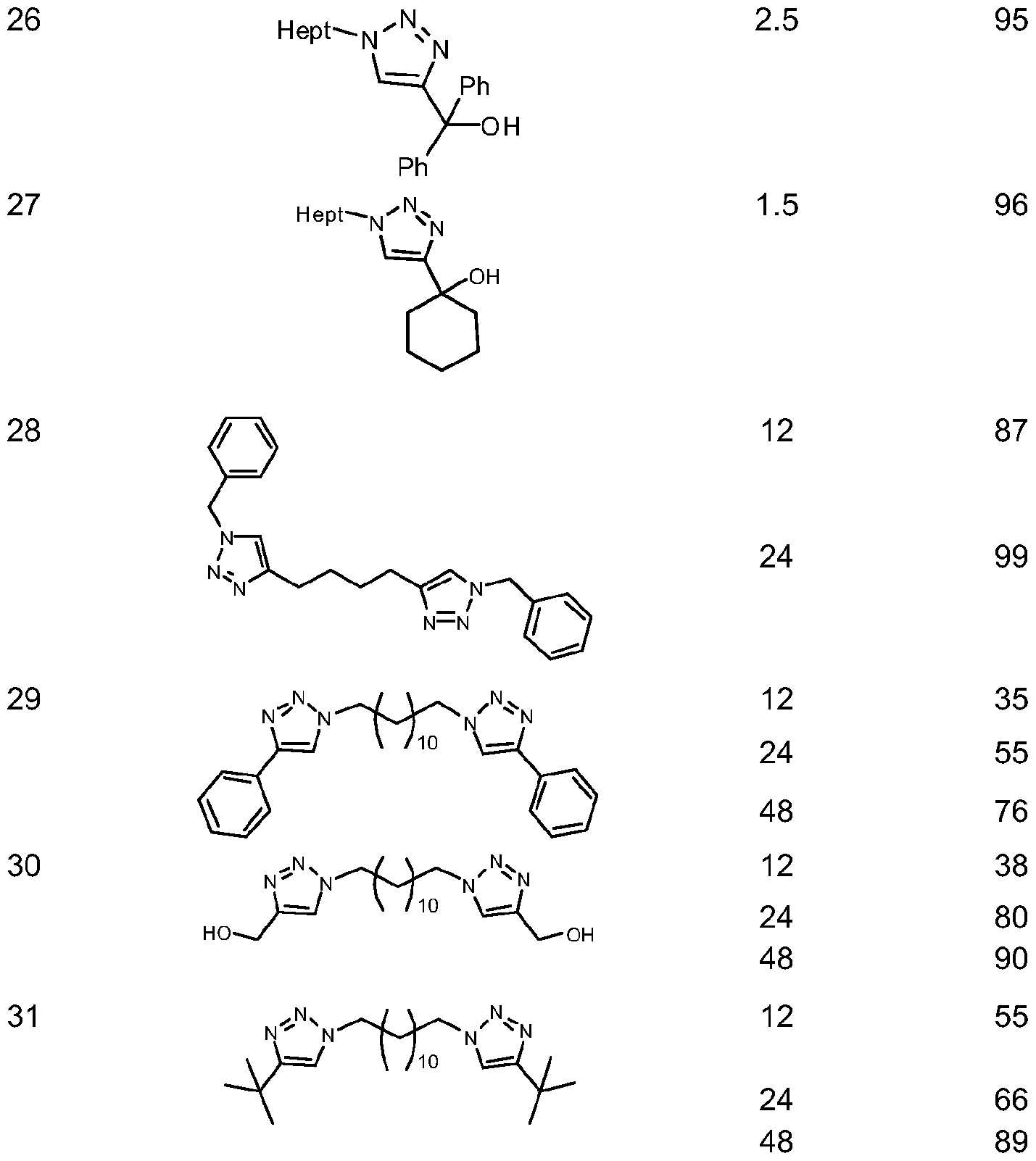
- Catalytic amounts of the catalysts of this invention are used. More particularly, the amount of catalyst is usually in the range of about 0.001 mol% to about 10mol% relative to the terminal alkyne. Preferably, about 0.5mol% to about 5mol% of the catalyst relative to the organic azide is employed. When the alkyne is a terminal alkyne, more preferred amounts of the catalyst are in the range of about 0.1 to 2mol% relative to the organic azide. When the alkyne is an internal alkyne, more preferred amounts of the catalyst are in the range of 5 to 10mol% relative to the organic azide. When peptide synthesis is conducted, it is preferred to use 5 equivalents of catalyst relative to the number of molecules of peptide.
- Temperatures during the processes of the invention generally range from 0°C to 140°C and preferably from about room temperature to 1 10°C.
- the preferred working temperature is about room temperature, depending on the melting point of the alkyne, in which case the temperature will subject to that value.
- temperatures from 50°C to about 80°C are preferred.
- the temperature range goes from 100 to 1 10°C.
- the working temperature is preferably room temperature.
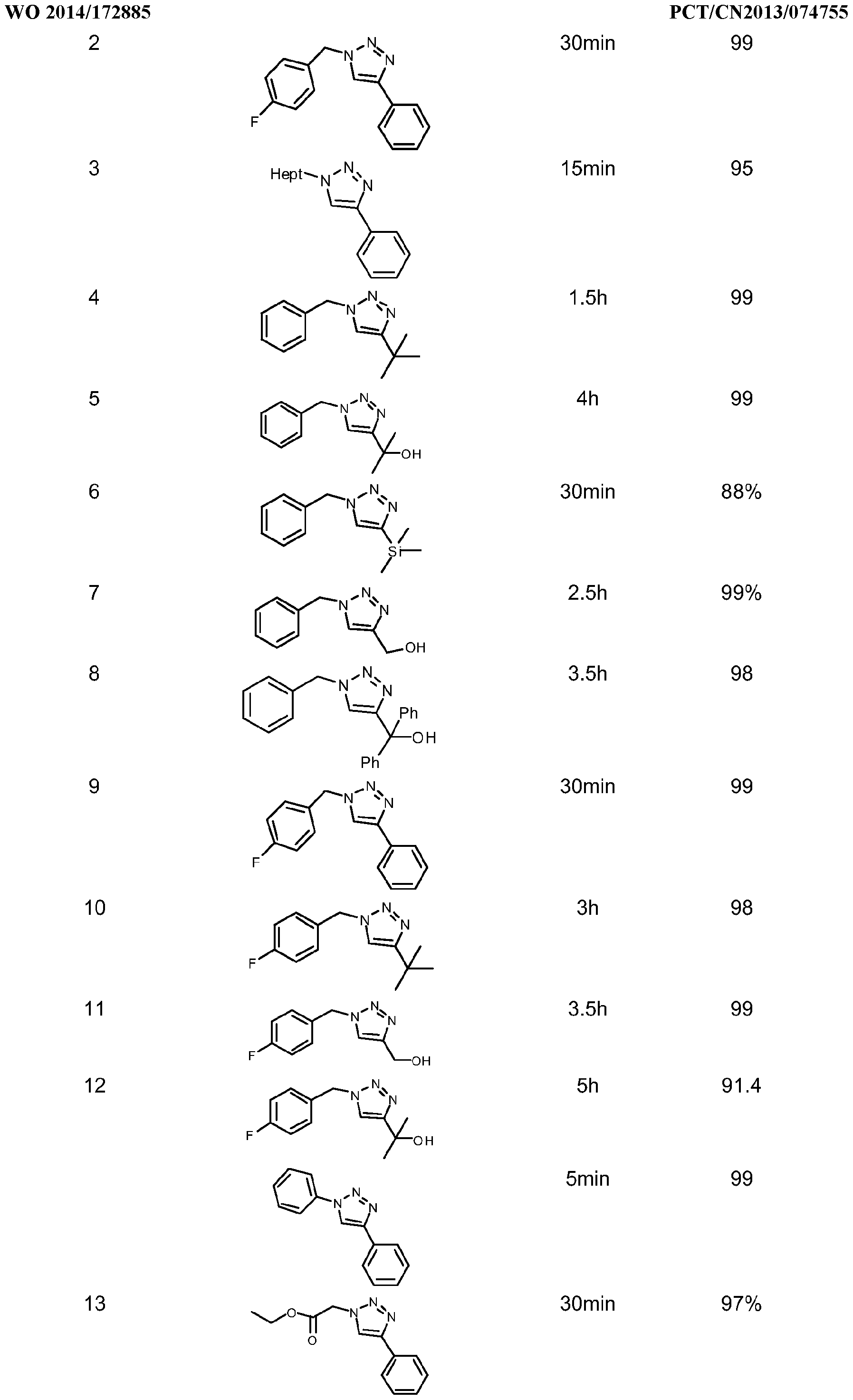
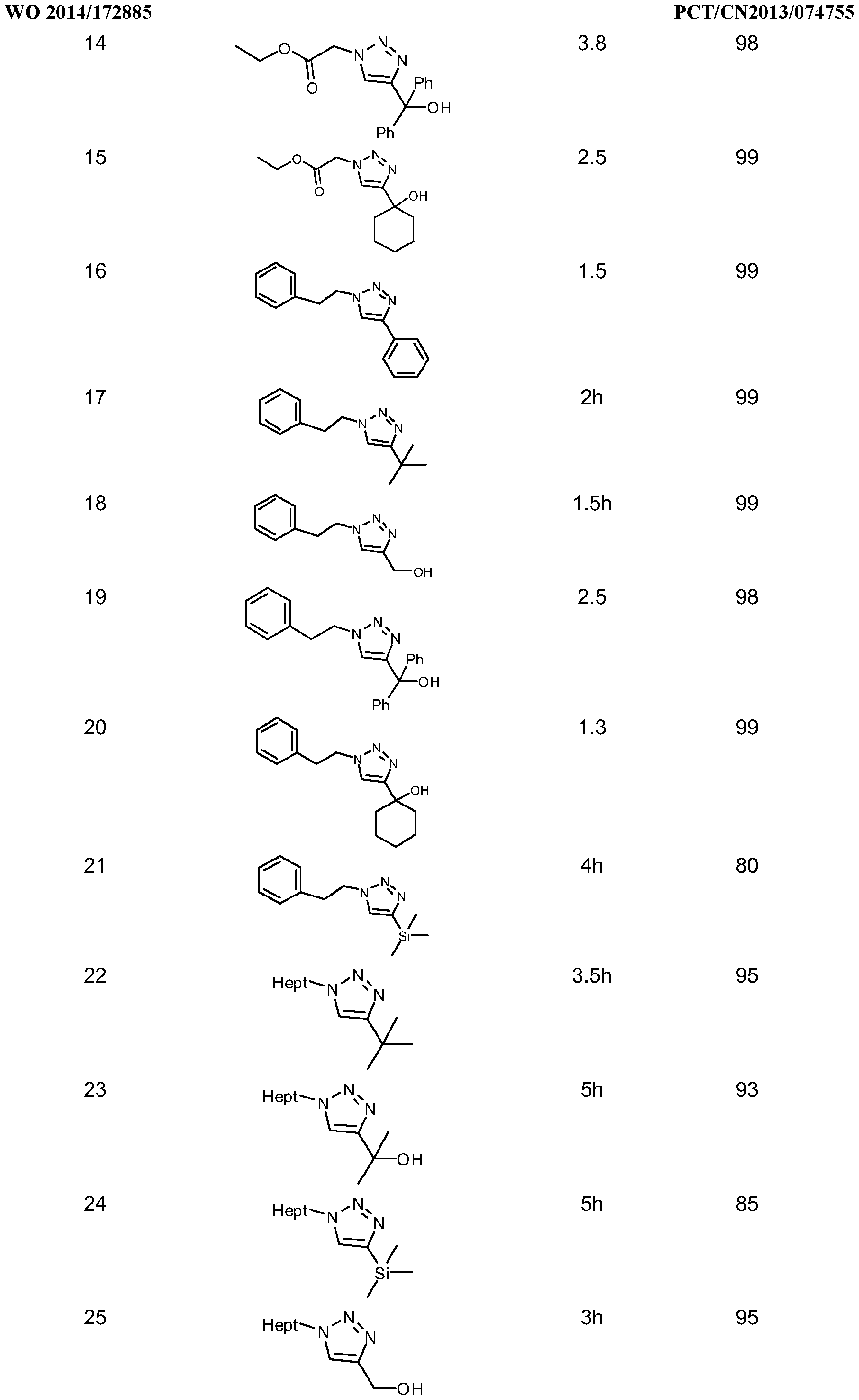
- the processes of this invention comprises a wide range of reaction times, depending on the reaction conditions and the nature of the reactants involved in the catalysis, the reaction times arein the order of minutes (5 to 20minut.es) to hours (2 to 18hours) when acetylene gas, terminal alkynes with organic and/or inorganic azides are used. Internal alkynes react more slowly than do terminal alkynes and/or acetylene; therefore the reaction times are longer. Addition of a higher catalyst loading and/or increasing the reaction temperature may shorten the reaction time. When using acetylene gas and/or terminal alkynes, water may be used to shorten the reaction time. When the 1 ,2,3-triazole products are oils or low melting point solidslonger reaction times are required. Peptide synthesis requires longer reaction times, which are in the range of 12 to 48h, and 18h is the preferable reaction time.
- the 1 ,2,3-triazole products are generally solids, which can be isolated by standard techniques such as precipitation or centrifugation and decantation, depending of the solvent used, it may be necessary to extract with an organic solvent and purify by column chromatography. For oily products, solvent extraction and/or column chromatography are the typical purification methods. Peptides are mainly purified by HPLC techniques.
- an organic azide (1 .0 mmol), an alkyne (1 .05mmol) and catalyst Bis[1-(4-sodiumsulfonatebutyl)-3-(2,4,6-trimethylphenyl)-4,5-dihydroimidazolyl-3-ylidine] copper (I) hexafluorophosphate (IMDS 2 -Cu) (0.5mol% unless specified), are added.
- IMDS 2 -Cu 0.5mol% unless specified
- the invention comprises reactions in aqueous media for which the stability of the azide is too low to be isolated and used as pure.
- a procedure in which the azide was formed in-situ from alkyl and/or aryl halide is used.
- Water and room temperature are the preferred conditions, however, mixtures of water and a polar organic solvent like water/Dimethylsulfoxide, water/acetonitrile, water/alcohol (preferably lower alkyl alcohols: methanol, ethanol, isopropanol or fert-butanol) are also possible to use.
- the reactions may be carried out at a temperature range of 20-75°C.
- the order ofpreference for halide substituents in the alkyl or aryl group are: l>Br>CI.
- alkyl halide (1 mmol) and NaN 3 (1 .1 mmol) and 1 ml_ of water (unless otherwise noted) were added.
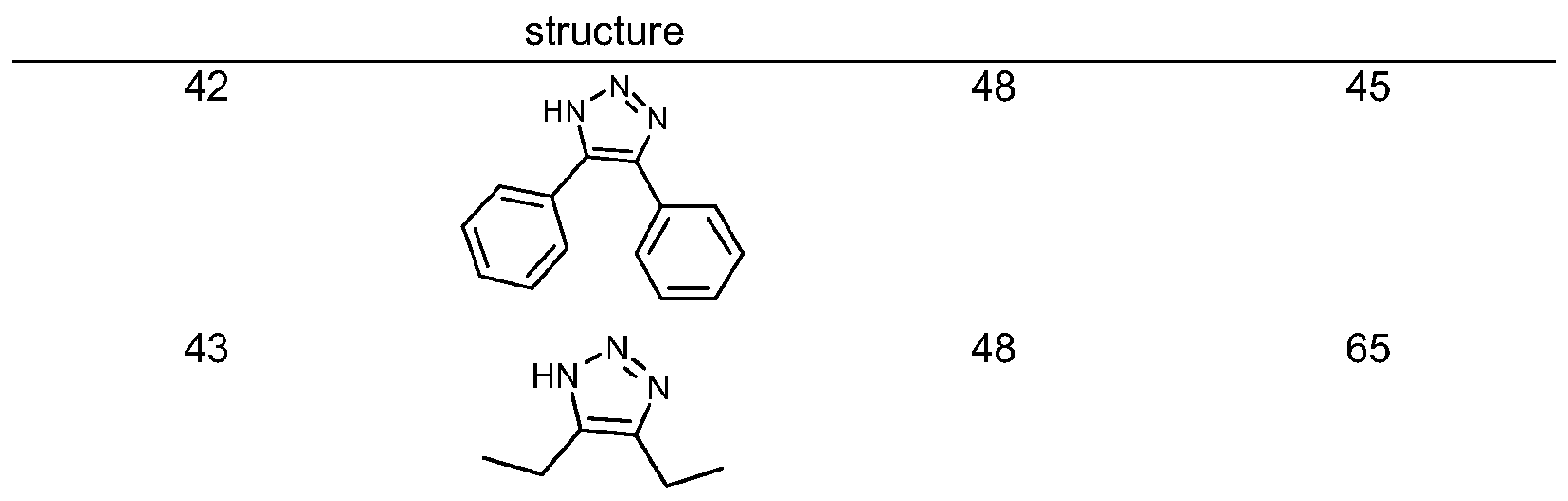
- the reaction was allowed to proceed at room temperature (unless otherwise noted) and monitored by 1 H NMR analysis of aliquots. After total consumption of the starting azide, the solid product was collected by filtration and washed with water and pentane. In case of oily products, these were extracted with dichloromethane and washed with aqueous NH 4 CI. Examples of the triazoles synthesized by this procedure are given in table 4along with the yields and reaction times.
- the invention comprises the synthesis of triazoles in which no substituent is present in the 4,5-positions, by using CaC 2 and acetylene gas as the alkyne source.
- CaC 2 as the source of acetylene:
- the reaction was stirred at room temperature and monitored by TLC. After the reaction was completed, the mixture was acidified at pH ⁇ 5 with HCI 0.1 N and the organic material was washed with brine and extracted with diethyl ether. In case the product is not pure enough, column chromatography may be needed.
- the synthesis of the peptides was followed under standard conditions using microscale techniques. It is possible the use of pH buffers to the solutions of peptides in order to maintain its properties, however highly acidic media may decrease the activity of the catalyst. Co-solvents are also susceptible to be used when the solubility of the peptides is a major issue. For that case MeOH, DMSO, NMP and/or HFIP (Hexafluoroisopropanol)may be used. For the case of poor soluble peptides, the first option for co-solvent should be HFIP. There is no preference on the order of addition for the substrates and/or catalyst. The catalysis is carried out preferably under ambient conditions of temperature and with long reaction times.
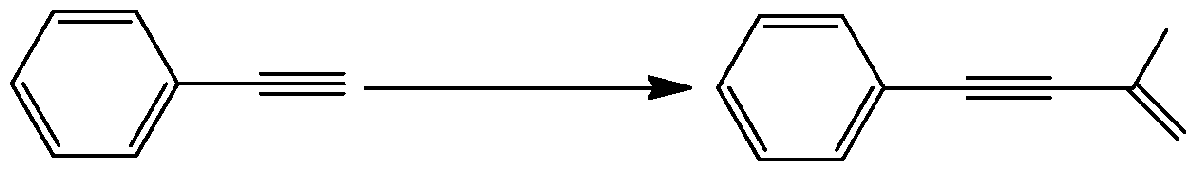
- Neat DMAP 10 120 92 General Procedure for Carboxylation of the Terminal Alkynesis exemplified using phenylacetylene, scheme 6.
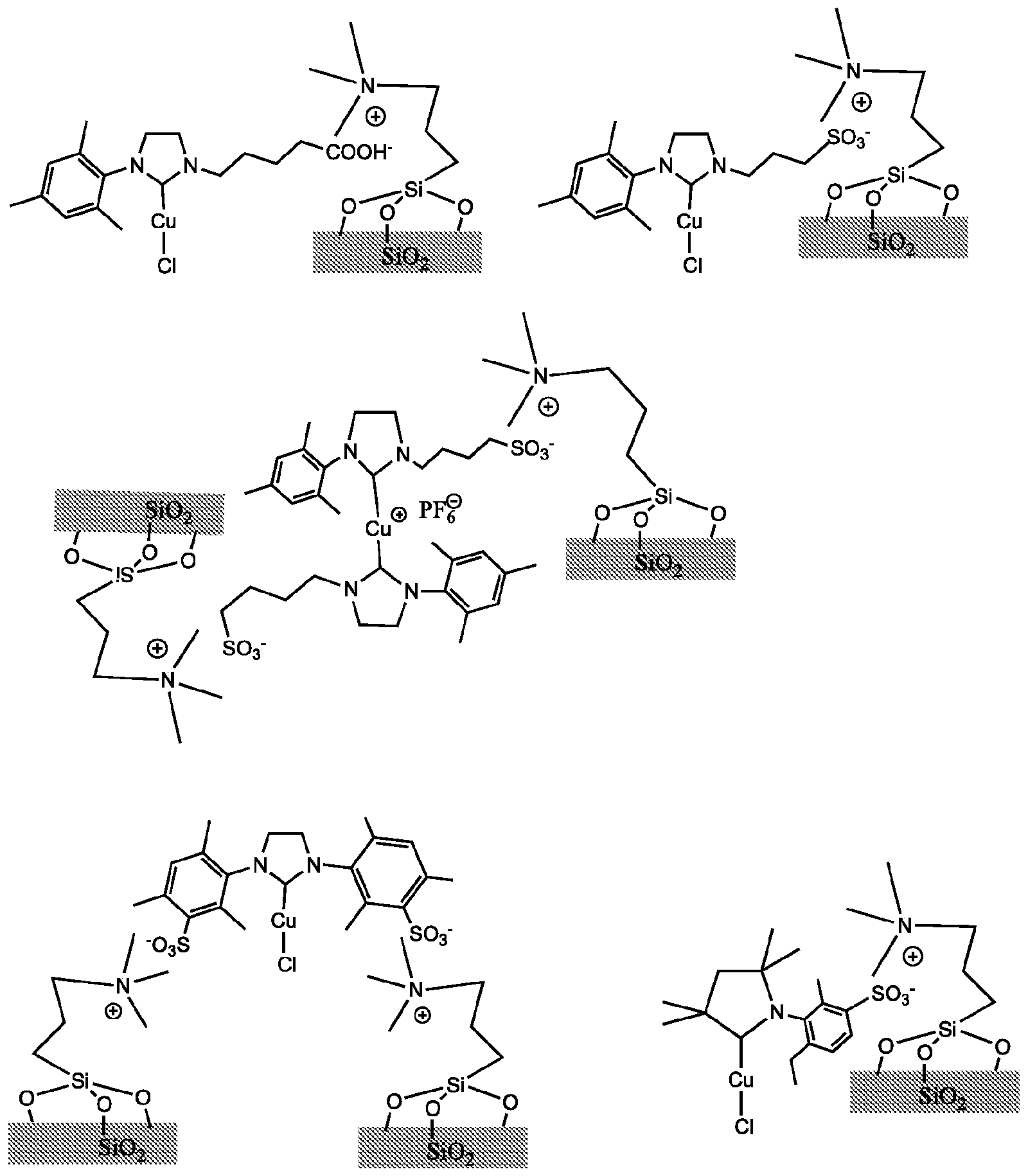
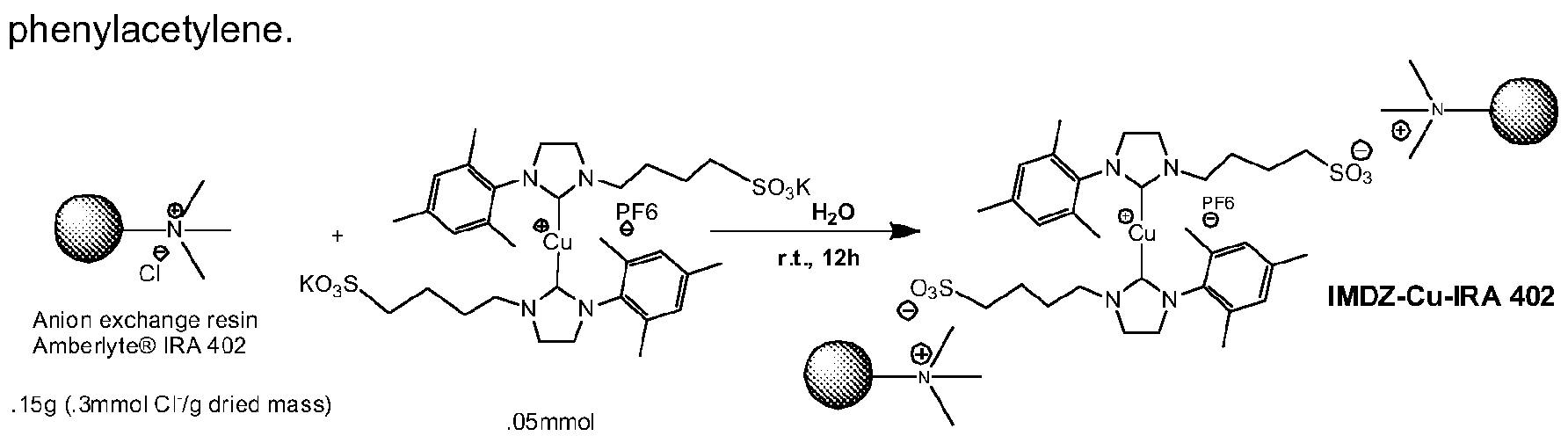
- the catalysts of this invention can also be immobilized in an efficient and straightforward manner, scheme 7.
- the catalysts can easily be recovered and reused for the next run as exemplified with
- Immobilization of the catalysts of this invention is not limited to resins bearing ionic groups.
- the catalysts of this invention can be immobilized on any support (organic, inorganic, ionic liquids, etc.) bearing ionic groups, e.g. inorganic supports which have been modified so that they possess ionic groups, organic support bearing ionic groups, metal organic frameworks with ionic groups and so on. Some examples, but not intended to limit are giving above mentioned.
- 29.111ustrative examples of new conversions include: (a) C. Das Neves Gomes, O. Jacquet, C. V Amsterdam, P. Thuery, M. Ephritikhine, T. Cantat, Angew. Chem., Int. Ed. 2012, 51 , 187. (b) O. Jacquet, C. Das Neves Gomes, M. Ephritikhine, T. Cantat, J. Am. Chem. Soc. 2012, 134, 2934. (c) D. Yu, Y. Zhang, Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 20184.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Catalysts (AREA)
Abstract
This invention relates to group 9 transition metal catalysts and process for use of the catalysts in alkyne-azide cycloaddition (Click reaction), in reaction utilizing CO2 as a carbon feedstock to react with epoxides to produce cyclic or polycarbonate products and in reactions utilizing CO2 as a carbon feedstock to react with terminal alkynes to produce carboxylic acids. The catalyst compounds of the invention are represented by the formula (I) wherein M is a Group 9 metal; X is an anionic ligand; L is a two electron donor ligand bearing two ionic groups (G); and by the formula (II) wherein M is a group 9 metal; L1 and L2 are two-electron donor ligands each bearing one ionic group (G) and A - is anion. The present invention also relates to an easy applicable catalyst synthesis and the application in different click-processes, different reactions involving CO2 and their different application fields. The invention has utility in the fields of catalysis, organic synthesis, polymer chemistry, and industrial and fine chemicals chemistry.
Description
GROUP 9 TRANSITION METAL CATALYSTS AND PROCESS FOR USE OF SAME
FIELD OF THE INVENTION
This invention relates generally to organometallic catalysts suitable for the CLICK reaction, and reactions involving carbon dioxide as substrate. More particularly catalyst compounds suitable azide-alkyne cyclo-addition, their synthesis and processes for the use thereof. As well catalyst compounds suitable for conversion of carbon dioxide generating cyclic or poly carbonates and carboxylic acids. The invention has utility in the fields of catalysis, organic synthesis, polymer chemistry, and industrial and fine chemicals chemistry.
BACKGROUND OF THE INVENTION
The dipolar cycloaddition of terminal alkynes with organic azides, known as the Huisgen reaction is undoubtedly the most direct synthetic method for the generation of triazoles. The development of the Copper(l)-catalyzed reaction between organic azides and terminal alkynes, which regioselectively provides 1 ,4-disubstituted 1 ,2,3-triazoles under mild conditions1, represented a definitive advance in triazole chemistry and has become the most practical and useful "click" reaction2. The reaction is effective in the preparation of a wide variety of triazole-containing molecules3. 1 ,2,3-triazoles have found widespread applications in pharmaceuticals and agrochemicals4. The discovery of metal-catalyzed 1 ,3-dipolar cycloaddition of azides and terminal alkynes has further triggered the use of 1 ,2,3-triazoles in bioconjugation1, drug discovery5, materials science6 and combinatorial chemistry7. In addition, a number of compounds containing 1 ,2,3-triazoles have shown a broad spectrum of biological activities; such compounds possess antibacterial8, herbicidal, fungicidal9, antiallergic10 and anti-HIV properties11. Recently, 1 ,2,3-triazoles have also been used as catalysts and ligands in transition metal-based catalyst systems. The rapidly growing demand for these heterocycles necessitates the development of effective methods for the preparation of a range of 1 ,2,3-triazole derivatives.
The fundamental thermal reaction, involving terminal or internal alkynes, has been known for over a century (to the best of our knowledge, the first 1 ,2,3-triazole was synthesized by A. Michael from phenyl azide and diethyl acetylenedicarboxylate in 1893), 12 and has been most thoroughly investigated by Rolf Huisgen and co-workers in the 1950s to the 1970s.13,14 The process is strongly thermodynamically favored ( Ho = -45 to -55 kcal/mol) due to the high potential-energy content of the two reaction components, but has a relatively high kinetic-energy barrier (ca. 26 kcal/mol for methyl azide and propyne)15 that renders the reaction very slow at room temperature for unactivated reactants. However, the reaction of azides with terminal alkynes dramatically accelerates by using metal-catalysts e.g. copper(l) catalysts16,17 and exhibits several features that make the catalyzed reaction unique among other block-ligation reactions.
1. The reaction is not significantly affected by the steric and electronic properties of the groups attached to the azide and alkyne reactive centers. For example, azides carrying a primary, secondary, or tertiary group; electron-deficient or electron-rich group; and aliphatic, aromatic, or hetero-aromatic substituent usually react well with variously substituted terminal alkynes.
2. The reaction is unaffected by water and by most organic and inorganic functional groups; thus, all but eliminating the need for protecting-group chemistry. The un-catalyzed thermal cycloaddition of azides to alkynes usually requires prolonged heating and results in mixtures of the 1 ,4- and 1 ,5-disubstituted regioisomers. In contrast, copper catalyzed Azide Alkyne Cyclization(AAC) produces only 1 ,4-disubstituted-1 ,2,3-triazoles at room temperature in excellent yields.
3. The rate of the metal-catalyzed process is approximately 107 times that of the un-catalyzed version,5 making the reaction conveniently fast in the temperature range of 0 to 25 °C. Furthermore, ligand-accelerated-catalysis effects18 are also significant, resulting in further increases in the reaction rate.
4. The 1 ,2,3-triazole unit that results from the reaction has several advantageous properties: (i) a high chemical stability (in general, being inert to severe hydrolytic, oxidizing, and reducing conditions, even at high temperature), (ii) a strong dipole moment (5.2-5.6 D), (iii) an aromatic character, and (iv) a good hydrogen-bond-accepting ability.19,20 Thus, it can interact productively in several ways with biological molecules, and serve as a replacement for the amide linkage in some circumstances.
A number of different copper(l) sources can be utilized in the reaction. Copper(l) salts (CuCI, Cul, CuBr, CuOAc) and coordination complexes (such as [Cu(CH3CN)4]PF6,16 (EtO)3P«Cul,21 [Cu Pha^Br22'23) can be used directly. The latter have been particularly effective in organic solvents, in which cuprous salts have limited solubility. However, Cu(l) is thermodynamically unstable and can be relatively easily oxidized to catalytically inactive Cu(ll). The standard potential of the Cu2+/Cu+ couple is 159 mV, but can vary widely with the solvent and the ligand environment of the metal. Cu(ll), itself, is an oxidant and can mediate the oxidative alkyne coupling reactions mentioned above, thereby impairing the efficiency of the cycloaddition and resulting in the formation of undesired byproducts. Therefore, when a copper(l) catalyst is used directly, whether by itself or in conjunction with amine ligands, exclusion of oxygen may be required to prevent these complications. As an alternative to oxygen-free conditions, a sacrificial reducing agent, for example ascorbate, can be used in combination with a copper(ll) salt, e.g. copper(ll) sulfate pentahydrate or copper(ll) acetate.17 Catalytic amounts of Cu(l) can also be introduced in the reaction through com-proportionation of Cu(ll) and Cu(0), thus further simplifying the experimental procedure.
Copper nanoclusters, which are easily obtained and are air-stable,24 and copper/cuprous oxide nanoparticles25 have also shown good catalytic activity, although this procedure requires longer reaction times.
The ligand tris[(1-benzyl-1 H-1 ,2,3-triazol-4-yl)methyl]amine (TBTA), discovered soon after the disclosure of CuAAC,26 significantly accelerates the CuAAC reaction and stabilizes the +1 oxidation state of the copper catalyst. However this catalyst system is highly susceptible to oxidation in air, electrolytic manipulations were necessary to achieve good performance for bio-conjugation tasks.27
Although several catalyst systems have been used for the CuAAC, in most cases they need the presence of co-catalysts such as auxiliary ligands, bases -in most cases amines-, and reducing or oxidizing agents depending on the Cu source used, in order to enhance their catalytic activity. In most of the cases high Cu loading must be used to ensure high performance of the catalysis. The typical protocol for the synthesis of peptides using the CuS04 source and a reducing agent has shown low efficiency and the systems composed by a Cu(l) source and an auxiliary ligand are air sensitive and/or require complex manipulations.
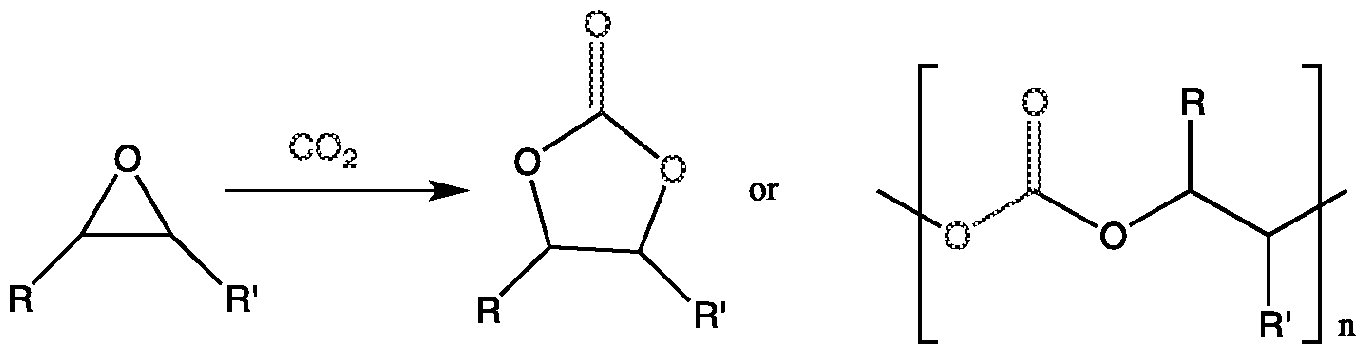
The catalysts of this invention can also be applied to synthesize cyclic- and poly-carbonate products. Synthetic procedures which utilize C02 as a carbon feedstock are currently generating great interest within the scientific community28 as these may offer viable alternative routes toward various organic structures that are currently derived from fossil fuel based resources29.
The catalysis of the atom-efficient (cyclo) addition of C02 to epoxides which yields useful cyclic or polycarbonate products30, scheme 1 , is gaining momentum, as a variety of industrial targets incorporating these carbonates have been developed.28a"d
Scheme 1. Synthesis of Organic Carbonates from Epoxides and C02
Many different binary/bifunctional catalyst systems for this reaction have been reported combining a Lewis acid and a nucleophile which is required for the ring opening of the epoxide.31
To date, most of the reported catalyst systems require relatively high catalyst loadings, thus limiting their potential for industrial application.
Previously, Minakata reported on a facile route toward more functional iodinated (unsaturated) cyclic carbonates elegantly making use of trapping a carbonic acid derivative by a hypoiodite source under mild conditions.32 Unfortunately, this procedure does not allow for the introduction of other functional groups and cannot address the synthesis of sterically encumbered disubstituted carbonates (Scheme 1 , R and R') and oxetanes. Thus, the development of a catalyst system able to combine unusual reactivity (high TOF) with a broad substrate scope and high functional group tolerance to date still represents a challenging task.
Recently, Darensbourg reported an aluminum based catalyst33 and also North ef al. reported a bimetallic Al catalysts which is active under ambient conditions, although due to the catalyst loadings required high TOFs were not achieved and only terminal epoxides proved to be suitable substrates.34
The development of innovative catalysts is still highly desirable especially keeping the industrial applicability in mind.
The catalysts of this invention can also be applied to synthesize carboxylic acids using C02. Carboxylic acids are one of the most important types of compounds in medicinal chemistry and also in fine-chemicals synthesis35, 36. Although there are many well-established protocols for the preparation of carboxylic acids, the direct carboxylation of carbon nucleophiles using C02 as the electrophile is the most attractive and straightforward method35,36. The formation of a stable C— C bond is desired for C02 fixation and remains the most challenging aspect thus far. Typically, this type of reaction is facilitated by the insertion of C02 into a metal-carbon bond35"37. Widespread use of these methods is limited by the synthesis organometallic reagents as precursors and the restricted substrate scope.
R - M + CO2 * R-COOM (M=Li, Mg, Cu, Mn, Sn, Zn, Al, B)
In the past decades, several interesting systems have been reported for metal mediated reductive carboxylation of alkynes38, allenes39, and alkyenes40 with C02 to form carboxylic acids or esters. However, most of those systems need either a stoichiometric amount of transition metals as reactants or an excess amount of organometallic reagents for transmetallation processes. An alternative possibility to achieve the catalytic synthesis of carboxylic acid from C02 is by direct C— H bond activation and carboxylation. Very recently, Zhang reported a copper- and copper-A/-heterocyclic carbene cocatalyzed transformation of C02 to carboxylic acid through C— H bond activation and carboxylation of terminal alkynes41. They proposed that the copper center activates the terminal alkyne with a base to form the copper acetylide intermediate, whereas the free carbene activates C02 to form NHC carboxylate. The presence of the free carbenes makes this catalytic system very sensitive towards impurities, functional groups and humidity.
Therefore, the development of efficient catalyst systems for C02 utilization under mild conditions is highly desired, especially for real world applications.
SUMMARY OF THE INVENTION
The present invention introduces the use of water-soluble complexes for the Azide Alkyne Cycloaddition preferably in aqueous media, without the need of external bases or extra ligands and using low catalyst loadings, in which the synthesis of triazoles using either internal or terminal alkynes, as well as acetylene gas with organic azides is aimed. The present invention is not limited to organic azides, since it is possible to use /n-s/ft/generated HN3(from inorganic azides) for the synthesis of 4- and 4,5-substituted triazoles under aqueous conditions by using these catalysts. Furthermore, the efficiency of these catalysts is also proven by its excellent performance in peptidomimetic chemistry. The present invention also relates to an easy applicable catalyst synthesis and the application in different click-processes.
The organometallic catalyst compound of the present invention can be prepared by contacting a Group 9metal precursor compound with atwo-electron donating ligand bearing two ionic groups or with a two-electron donating ligand bearing one ionic group.
G1 _ |_ _ G2 |_1 _ G1
M M® A®
X G2 -L2
Formule (I) Formule (II)
Wherein,
M is a Group 9 transition metal
X is preferably an anionic ligand.
L represents an electron donor
L1 and L2 represent identical or different ligands, preferably electron donors.
G1 and G2 represent an ionic group.
A" is an anion, preferable a non-coordinating
In another aspect, the invention provides novel organometallic compounds according to the above structure. In an extra aspect, the invention provides a method for performing a catalytic cycloaddition reaction comprising contacting at least one alkyneand an azide compound with the "click' -catalyst of the invention. Reference to "a substituent" encompasses a single substituent as well as two or more substituents, and the like.
In the context of this invention, all the above and below mentioned, general or preferred ranges of definitions, parameters or elucidations among one another, or also between the respective ranges and preferred ranges can be combined in any manner.
In the context of this invention, related to the different types of click catalysts, the term "substituted" means that a hydrogen atom or an atom is replaced by a specified group or an atom, and the valence of the atom indicated is not exceeded and the substitution leads to a stable compound.
BRIEF DESCRIPTION OF THE DRAWINGS
Figure 1 depicts the comparison of bis(NHC)-Cu catalysts for the click reaction between phenylacetylene and benzylazide.
DETAILED DESCRIPTION
Terminology and Definitions
1. Unless otherwise mentioned, the invention is not limited to specific reactants, substituents, catalysts, reaction conditions, or the like, as such may vary. It is also to be understood that the terminology used herein is for the purpose of describing particular embodiments only and is not intended to be limiting.
2. In this specification and in the claims that follow, reference will be made to a number of terms, which shall be defined to have the following meanings:
3. The term "alkyl" as used herein refers to a linear, branched, or cyclic saturated hydrocarbon group typically although not necessarily containing 1 to about 24 carbon atoms, preferably 1 to about 12 carbon atoms, such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, t-butyl, octyl, decyl, and the like, as well as cycloalkyl groups such as cyclopentyl, cyclohexyl and the like. Generally, although again not necessarily, alkyl groups herein contain 1 to about 12 carbon atoms. The term "C"i-C6-alkyl" intends an alkyl group of 1 to 6 carbon atoms, and the specific term "cycloalkyl" intends a cyclic alkyl group, typically having 3 to 8 carbon atoms.
4. The term "substituted alkyl" refers to alkyl substituted with one or more substituent groups, and the terms "heteroatom-containing alkyl" and "heteroalkyl" refer to alkyl in which at least one carbon atom is replaced with a heteroatom. If not otherwise indicated, the term "alkyl" include linear, branched, cyclic, unsubstituted, substituted, and/or heteroatom-containing alkyl.
5. The term "alkylene" as used herein refers to a difunctional linear, branched, or cyclic alkyl group, where "alkyl" is as defined above.
6. The term "alkenyl" as used herein refers to a linear, branched, or cyclic hydrocarbon group of 2 to about 24 carbon atoms containing at least one double bond, such as ethenyl, n-propenyl, isopropenyl, n-butenyl, isobutenyl, octenyl, decenyl, tetradecenyl, hexadecenyl, eicosenyl, and the like. Preferred alkenyl groups herein contain 2 to about 12 carbon atoms. The term "cycloalkenyl" intends a cyclic alkenyl group, preferably having 5 to 8 carbon atoms. The term "substituted alkenyl" refers to alkenyl substituted with one or more substituent groups, and the terms "heteroatom-containing alkenyl" and "heteroalkenyl" refer to alkenyl in which at least one carbon
atom is replaced with a heteroatom. If not otherwise indicated, the term "alkenyl" include linear, branched, cyclic, unsubstituted, substituted, and/or heteroatom-containing alkenyl.
The term "alkenylene" as used herein refers to a difunctional linear, branched, or cyclic alkenyl group, where "alkenyl" is as defined above.
The term "alkynyl" as used herein refers to a linear or branched hydrocarbon group of 2 to about 24 carbon atoms containing at least one triple bond, such as ethynyl, n-propynyl, and the like. Preferred alkynyl groups herein contain 2 to about 12 carbon atoms. The term "substituted alkynyl" refers to alkynyl substituted with one or more substituent groups, and the terms "heteroatom-containing alkynyl" and "heteroalkynyl" refer to alkynyl in which at least one carbon atom is replaced with a heteroatom. Ifnot otherwise indicated, the term "alkynyl" include linear, branched, unsubstituted, substituted, and/or heteroatom-containing alkynyl respectively.
The term "alkoxy" as used herein intends an alkyl group bound through a single, terminal ether linkage; that is, an "alkoxy" group may be represented as -O-alkyl where alkyl is as defined above. Analogously, "alkenyloxy" refers to an alkenyl group bound through a single, terminal ether linkage, and "alkynyloxy" refers to an alkynyl group bound through a single, terminal ether linkage.
The term "aryl" as used herein, and unless otherwise specified, refers to an aromatic substituent containing a single aromatic ring or multiple aromatic rings that are fused together, directly linked, or indirectly linked (such that the different aromatic rings are bound to a common group such as a methylene or ethylene moiety). Preferred aryl groups contain 5 to 24 carbon atoms, and particularly preferred aryl groups contain 5 to 14 carbon atoms. Exemplary aryl groups contain one aromatic ring or two fused or linked aromatic rings, e.g., phenyl, naphthyl, biphenyl, diphenylether, diphenylamine, benzophenone, and the like. "Substituted aryl" refers to an aryl moiety substituted with one or more substituent groups, and the terms "heteroatom-containing aryl" and "heteroaryl" refer to aryl substituents in which at least one carbon atom is replaced with a heteroatom, as will be described in further detail infra.
The term "aryloxy" as used herein refers to an aryl group bound through a single, terminal ether linkage, wherein "aryl" is as defined above. An "aryloxy" group may be represented as -O-aryl where aryl is as defined above. Preferred aryloxy groups contain 5 to 24 carbon atoms, and particularly preferred aryloxy groups contain 5 to 14 carbon atoms. Examples of aryloxy groups include, without limitation, phenoxy, o-halo-phenoxy, m-halo-phenoxy, p-halo-phenoxy, o-methoxyphenoxy, m-methoxy-phenoxy, p-methoxy-phenoxy, 2,4-dimethoxy-phenoxy, 3,4,5-trimethoxy-phenoxy, and the like.
The term "alkaryl" refers to an aryl group with an alkyl substituent, and the term "aralkyl" refers to an alkyl group with an aryl substituent, wherein "aryl" and "alkyl" are as defined above. Preferred alkaryl and aralkyl groups contain 6 to 24 carbon atoms. Alkaryl groups include, but not limit to, for example, p-methylphenyl, 2,4-dimethylphenyl, p-cyclohexylphenyl, 2, 7 -dimethylnaphthyl, 7 -cyclooctyl naphthyl, 3-ethyl-cyclopenta-1 ,4-diene, and the like. Examples of aralkyl groups include, without limitation, benzyl, 2-phenyl-ethyl, 3-phenyl-propyl, 4-phenyl-butyl, 5-phenyl-pentyl, 4-phenylcyclohexyl, 4-benzylcyclohexyl, 4-phenylcyclohexylmethyl, 4-benzylcyclohexylmethyl, and the like. The terms "alkaryloxy" and "aralkyloxy" refer to substituents of the formula -OR wherein R is alkaryl or aralkyl, respectively, as just defined.
The term "acyl" refers to substituents having the formula -(CO)-alkyl, -(CO)-aryl, or -(CO)-aralkyl, and the term "acyloxy" refers to substituents having the formula -0(CO)-alkyl, -0(CO)aryl, or -0(CO)-aralkyl, wherein "alkyl," "aryl, and "aralkyl" are as defined above.
The terms "cyclic" and "ring" refer to alicyclic or aromatic groups that may or may not be substituted and/or heteroatom containing, and that may be monocyclic, bicyclic, or polycyclic. The term
"alicyclic" is used in the conventional sense to refer to an aliphatic cyclic moiety, as opposed to an aromatic cyclic moiety, and may be monocyclic, bicyclic, or polycyclic.
The terms "halo" and "halogen" are used in the conventional sense to refer to a chloro, bromo, fluoro, or iodo substituent.
"Hydrocarbyl" refers to univalent hydrocarbyl radicals containing 1 to about 30 carbon atoms, preferably 1 to about 24 carbon atoms, most preferably 1 to about 12 carbon atoms, including linear, branched, cyclic, saturated, and unsaturated species, such as alkyl groups, alkenyl groups, aryl groups, and the like. The term "hydrocarbylene" intends a divalent hydrocarbyl moiety containing 1 to about 30 carbon atoms, preferably 1 to about 24 carbon atoms, most preferably 1 to about 12 carbon atoms, including linear, branched, cyclic, saturated and unsaturated species. "Substituted hydrocarbyl" refers to hydrocarbyl substituted with one or more substituent groups, and the terms "heteroatom-containing hydrocarbyl" and "heterohydrocarbyl" refer to hydrocarbyl in which at least one carbon atom is replaced with a heteroatom. Similarly, "substituted hydrocarbylene" refers to hydrocarbylene substituted with one or more substituent groups, and the terms "heteroatomcontaining hydrocarbylene" and "heterohydrocarbylene" refer to hydrocarbylene in which at least one carbon atom is replaced with a heteroatom. Unless otherwise indicated, the term "hydrocarbyl" and "hydrocarbylene" are to be interpreted as including substituted and/or heteroatom-containing hydrocarbyl and hydrocarbylene moieties, respectively.
The term "heteroatom-containing" as in a "heteroatom-containing hydrocarbyl group" refers to a hydrocarbon molecule or a hydrocarbyl molecular fragment in which one or more carbon atoms is replaced with an atom other than carbon, e.g., nitrogen, oxygen, sulfur, phosphorus or silicon, typically nitrogen, oxygen or sulfur. Similarly, the term "heteroalkyl" refers to an alkyl substituent that is heteroatom-containing, the term "heterocyclic" refers to a cyclic substituent that is heteroatom-containing, the terms "heteroaryl" and "heteroaromatic" respectively refer to "aryl" and "aromatic" substituents that are heteroatom-containing, and the like. It should be noted that a "heterocyclic" group or compound may or may not be aromatic, and further that "heterocycles" may be monocyclic, bicyclic, or polycyclic as described above with respect to the term "aryl." Examples of heteroalkyl groups include alkoxyalkyl, alkylsulfanyl-substituted alkyl, N-alkylated amino alkyl, and the like. Examples of heteroaryl substituents include pyrrolyl, pyrrolidinyl, pyridinyl, quinolinyl, indolyl, pyrimidinyl, imidazolyl, 1 ,2,4-triazolyl, tetrazolyl, etc., and examples of heteroatom containing alicyclic groups are pyrrolidino, morpholino, piperazino, piperidino, etc.
By "substituted" as in "substituted hydrocarbyl", "substituted alkyl", "substituted aryl", and the like, as alluded to in some of the aforementioned definitions, is meant that in the hydrocarbyl, alkyl, aryl, or other moiety, at least one hydrogen atom bound to a carbon (or other) atom is replaced with one or more non-hydrogen substituents. Examples of such substituents include, without limitation: functional groups such as halo, hydroxyl, sulfhydryl, C1-C24 alkoxy, C2-C24 alkenyloxy, C2-C24 alkynyloxy, C5-C24 aryloxy, C6-C24 aralkyloxy, C6-C24 alkaryloxy, acyl (including C2C24 alkylcarbonyl (-CO-alkyl) and C6-C24 arylcarbonyl (-CO-aryl)), acyloxy (-O-acyl, including C2C2 alkylcarbonyloxy (-O-CO-alkyl) and C6-C24 arylcarbonyloxy (-O-CO-aryl)), C2C24 alkoxycarbonyl (-(CO)-O-alkyl), C6-C2 aryloxycarbonyl (-(CO)-O-aryl), halocarbonyl (-CO)X where X is halo), C2-C2 alkylcarbonato (-O-(CO)-O-alkyl), C6-C24 arylcarbonato (-O-(CO)-O-aryl), carboxy (-COOH), carboxylato (-COO"), carbamoyl (-(CO)-NH2), mono-(C C24 alkyl)substituted carbamoyl (-(CO)-NH(C C24 alkyl)), di-(Ci-C24 alkyl)-substituted carbamoyl (-(CO)N(Ci-C24 alkyl)2), mono-(C5-C24 aryl)-substituted carbamoyl (-(CO)-NH-aryl), di-(C5-C2 aryl)substituted carbamoyl (-(CO)-N(C5-C24 aryl)2), N((C C24 alkyl) (Cs-C24 aryl))-substituted carbamoyl, thiocarbamoyl ( -(CS)-NH2), mono-(Ci-C24 alkyl)-substituted thiocarbamoyl (-(CS)NH( Ci-C2 alkyl)), di-(Ci-C2 alkyl)-substituted thiocarbamoyl (-(CS)-N(Ci-C24 alkyl)2), mono-(C5-C24 aryl)-substituted thiocarbamoyl (-(CS)-NH-aryl), di-(C5-C24 aryl)-substituted thiocarbamoyl ((CS)-N(C5-C24 aryl)2), N-(Ci-C2 alkyl) N-(C5-C24 aryl)-substituted thiocarbamoyl, carbamido (NH-(CO)-NH2), cyano (-C=N), cyanato (-0-C=N), thiocyanato (-S-C=N),
formyl (-(CO)-H), thioformyl (-(CS)-H), amino (-NH2), mono-(Ci-C24 alkyl)-substituted amino, di-(Ci-C24 alkyl)substituted amino, mono-(C5-C24 aryl)-substituted amino, di-(C5-C24 aryl)-substituted amino, C2-C24 alkylamido (-NH-(CO)-alkyl), C6-C24 arylamido (-NH-(CO)-aryl), imino (-C =NH where R = hydrogen, Ci-C24 alkyl, C5-C24 aryl, C6-C24 alkaryl, C6-C2 aralkyl, etc.), C2-C20 alkylimino (-CR=N(alkyl), where R =hydrogen, Ci-C24 alkyl, Cs-C24 aryl, C6-C24 alkaryl, C6-C24 aralkyl, etc.), arylimino (-CR=N(aryl), where R =hydrogen, Ci-C2o alkyl, Cs-C24 aryl, C6-C24 alkaryl, C6-C24 aralkyl, etc.), nitro (-N02), nitroso (-NO), sulfo (-S02-OH), sulfonato (-S02-0"), C C24 alkylsulfanyl (-S-alkyl; also termed "alkylthio"), Cs-C24 arylsulfanyl (-S-aryl; also termed "arylthio"), Ci-C24 alkylsulfinyl (-(SO)-alkyl), C5-C24 arylsulfinyl (-(SO)-aryl), C^C24 alkylsulfonyl (-S02-alkyl), C5-C24 arylsulfonyl (-S02-aryl), boryl (-BH2), borono (-B(OH)2), boronato (-B(OR)2 where R is alkyl or other hydrocarbyl), phosphono (-P(0)(OH)2), phosphonato (-P(0)(0")2), phosphinato (-P(0)(0")), phosphor (-P02), and phosphino (-PH2); and the hydrocarbyl moieties Ci-C2 alkyl (preferably Ci-Ci2 alkyl, more preferably CI-CB alkyl), C2-C24 alkenyl (preferably C2-Ci2 alkenyl, more preferably C2-C6 alkenyl), C2-C24 alkynyl (preferably C2-C12 alkynyl, more preferably C2-C6 alkynyl), C5-C24 aryl (preferably C5-C24 aryl), C6-C24 alkaryl (preferably C6-Ci6 alkaryl), and C6-C24 aralkyl (preferably C6-Ci6 aralkyl).
By "functionalized" as in "functionalized hydrocarbyl", "functionalized alkyl", "functionalized alkyne", "functionalized cyclic alkyne", and the like, is meant that in the hydrocarbyl, alkyl, alkyne, cyclic alkyne, or other moiety, at least one hydrogen atom bound to a carbon (or other) atom is replaced with one or more functional groups such as those described hereinabove.
In addition, the aforementioned functional groups may, if a particular group permits, be further substituted with one or more additional functional groups or with one or more hydrocarbyl moieties such as those specifically enumerated above. Analogously, the above-mentioned hydrocarbyl moieties may be further substituted with one or more functional groups or additional hydrocarbyl moieties such as those specifically enumerated.
The present invention comprises the use of water-soluble group 9transition metal complexes for the AAC preferably in aqueous media, without the need of external bases or extra ligands and using low catalyst loadings, in which the synthesis of triazoles using either internal or terminal alkynes, as well as acetylene gas with organic azides is aimed. The present invention is not limited to organic azides, since it is possible to use in-situ generated HN3 (from inorganic azides) for the synthesis of 4- and 4,5-substituted triazoles under aqueous conditions by using these catalysts. Furthermore, the efficiency of these catalysts is also proven by its excellent performance in peptidomimetic chemistry. In the embodiment, this invention comprises an azide and an alkyne, in which the catalysts have either of the following structures (I) or (II)
G1 - L - G2 L1 - G1
M M® A®
X G2 - L2
For the inventive catalyst with the general formula (I), the following description is given:
is a Group 9 transition metal, preferably Cu, Au
X is an anionic ligand.
In the general formula (I) X is for example, hydrogen, halogen, pseudohalogen, straight-chain or branched C1-C30 alkyl, C6-C24 aryl, CrC20 alkoxy, C6-C2 aryloxy, C3-C20 alkyl diketonate, C6-C24 aryl diketonate, Ci-C2o carboxylate, Ci-C2o alkylsulfonate, C6-C24 aryl sulfonate, Ci-C2o alkyl thiol, C6-C24 aryl thiol, Ci-C2o alkylsulfonyl or Ci-C2o alkylsulfinyl-radical.
The abovementioned X may further be substituted by one or more additional residues, for example by halogen, preferably fluorine, CrC20 alkyl, Ci-C20-alkoxy or C6-C24 aryl, where these groups may
optionally be in turn be substituted by one or more substituents from the group comprising halogen, preferable fluorine, C1-C5 alkyl, C1-C5 alkoxy, and phenyl.
In a preferred embodiment, X denote a halogen, in particular, fluorine, chlorine, bromine or iodine, benzoate, C1-C5 carboxylate, C1-C5 alkyl, phenoxy, C1-C5 alkoxy, C1-C5 alkyl thiol, C6-C24 arylthiol, C6-C24aryl or C1-C5 alkyl sulfonate.
In a particularly preferred embodiment, X is fluorine, chlorine, bromine or iodine, PhO (phenoxy), C6F50 (pentafluorophenoxy).
G1 and G2 are identical or different functional groups, preferably represent ionic groups.
Most preferable G is an ionic group or contains an ionic group, the ionic group can be cationic or anionic. Preferable groups, but not limited to, are e.g. quaternary ammonium, carboxylate, sulphonate (-SO3 "), -OSO3 ", -PO3 ", OPO3 ". The ionic group bears a counter ionto neutralize the charge, in case the ionic group is a cation the counter ion will be anionic, in case the ionic group is an anion the counter ionwill be cationic.
L represents an electron donor.
L represents a N-Heterocyclic carbene ("NHC"), substituted NHC, or cyclic alkyl amino carbene (CAAC) or substituted CAAC.
Preferably, t a) or (lllb)
(Ilia) (lllb)
by which
R1- R8, R5 , R6 are identical or different and are hydrogen, halogen, hydroxyl, aldehyde, keto, thiol, CF3, nitro, nitroso, cyano, thiocyano, isocyanates, carbodiimide, carbamate, thiocarbamate, dithiocarbamate, amino, amido, imino, ammonium, silyl, acyl, acyloxy or represents alkyl, cycloalkyl, alkenyl, cycloalkenyl, substituted alkenyl, heteroalkenyl, heteroatom-containing alkynyl, alkenylene, alkynyl, substituted alkynyl, aryl, substituted aryl, heteroaryl, carboxylate, alkoxy, alkenyloxy, alkynyloxy, aryloxy, alkaryl, aralkyl, alkaryloxy, aralkyloxy, alkoxycarbonyl, alkylammonium, alkylamino-, alkylthio-, arylthio, alkylsulfonyl, alkylsulfinyl, dialkylamino, alkylsilyl or alkoxysilyl, where these radicals may each optionally all be substituted by one or more aforementioned groups defined for R1-R8,
Optionally, one or more of the radicals R1 - R8, R5 , R6 independently of one another can be substituted by one or more substituents, preferably straight or branched C1-C10 alkyl, C3-C8 cycloalkyl, C1-C10 alkoxy or C6-C24 aryl, where these aforementioned substituents may in turn be substituted by one or more radicals, preferably selected from the group comprising halogen, especially chlorine or bromine, C1-C5 alkyl, C1-C5 alkoxy and phenyl.
Just for clarification, the depicted structures of the N-Heterocyclic carbene in the general formulas (Ilia) and (lllb) are equal with the N-Heterocyclic carbenes described in the literature, where frequently the structures (Ilia') and (lllb') are used, which highlighting the carbene character of N-Heterocyclic carbene. This also applies to the corresponding preferred, structures shown below (IVa) - (IVh)
(Ilia') (1Mb')
In a preferred embodiment of the catalysts of the general formulas (Ilia) and (1Mb) R1, R2, R5, R5 , R6 and R6 are independently of one another denote hydrogen, C6-C24-aryl, particularly preferably phenyl, straight or branched C1-C10 alkyl, particularly preferably propyl or butyl, or together with the inclusion of the carbon atoms to which they are attached form a cycloalkyi or aryl radical, where all the abovementioned radicals are optionally substituted may be substituted by one or more further radicals selected from the group comprising straight or branched Ci- C10 alkyl, C1-C10 alkoxy, C6-C24 aryl, and a functional group selected from the group consisting of hydroxy, thiol, thioether, ketone, aldehyde, ester, ether, amine, imine, amide, nitro, carboxylic acid, disulfide, carbonate, isocyanate, carbodiimide, carboalkoxy, carbamate, and halogen.
In a particularly preferred embodiment, the catalysts of the general formula (I) having one N-Heterocyclic carbene (NHC) as ligand L, where the radicals R3, R4, R7 and R8 are identical or different and are straight or branched C C 0 alkyl, particularly preferably /'-propyl or neopentyl, C3-C10 cycloalkyi, preferably adamantyl, C6-C24 aryl, particularly preferably phenyl.
If necessary, the above-mentioned residues are substituted as the meanings of R3, R4, R7 and R8 by one or more further radicals selected from the group comprising straight or branched C1-C5 alkyl, especially methyl, C1-C5 alkoxy, aryl and a functional group selected from the group consisting of hydroxy, thiol, thioether, ketone, aldehyde, ester, ether, amine imine, amide, nitro, carboxylic acid, disulfide, carbonate, isocyanate, carbodiimide, carboalkoxy, carbamate, and halogen.
Alternatively, in case R3, R4, R7 and R8are aryl, in each case two directly adjacent radicals from the group of R3, R4, R7 and R8, including the ring carbon atoms to which they are attached by a cyclic bridging group, generating one or more cyclic structures, including aromatic structures.
C1-C6 alkyl is, but not limited to, for example methyl, ethyl, n-propyl, /so-propyl, n-butyl, sec- butyl, feri-butyl, n-pentyl, 1 -methylbutyl, 2-methylbutyl, 3-methylbutyl, neo-pentyl, 1 -ethyl-propyl and n-hexyl.
C3-C8 cycloalkyi includes, but not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl and cyclooctyl.
C6-C24 aryl includes an aromatic radical having 6 to 24 skeletal carbon atoms. Preferred mono-, bi-or tricyclic carbocyclic aromatic radicals have 6 to 10 skeletal carbon atoms, for example but not limited to, phenyl, biphenyl, naphthyl, phenanthrenyl or anthracenyl.
In particular, the radicals R3, R4, R7 and R8can be identical or different and denote /'-propyl, neopentyl, adamantyl, mesityl or 2,6-diisopropylphenyl.
Particularly preferred N-Heterocyclic carbenes (NHC) have the following structure (IVa) - (IVh).
IVg IVh
In other useful embodiments, one of the N groups bound to the carbene in Formulae (Ilia) or (1Mb) is replaced with another heteroatom, preferably S, O or P, preferably an S heteroatom. Other useful N-heterocyclic carbenes include the compounds described in Hermann, W A. Chem. Eur. J 1996, 2, 772 and 1627; Enders, D. et al., Angew. Chem. Int. Ed. 1995, 34, 1021 ; Alder R. W., Angew. Chem. Int. Ed. 1996, 35, 1 121 ; and Bertrand, G. et al., Chem. Rev. 2000, 100, 39.
For purposes of this invention and claims thereto, "cyclic alkyl amino carbenes" (CAACs) are represented by the Formula (V):
(V)
Wherein the ring A is a 4-, 5-, 6-, or 7-membered ring, and Z is a linking group comprising from one to four linked vertex atoms selected from the group comprising C, O, N, B, Al, P, S and Si with available valences optionally occupied by hydrogen, oxo or R- substituents, wherein R is independently selected from the group comprising Ci to C12 hydrocarbyl groups, substituted Ci to C12 hydrocarbyl groups, and halides, and each R9 is independently a hydrocarbyl group or substituted hydrocarbyl group having 1 to 40 carbon atoms, preferably methyl, ethyl, propyl, butyl (including isobutyl and n-butyl), pentyl, cyclopentyl, hexyl, cyclohexyl, octyl, cyclooctyl, nonyl, decyl, cyclodecyl, dodecyl, cyclododecyl, mesityl, adamantyl, phenyl, benzyl, toluyl, chlorophenyl, phenol, or substituted phenol. Particularly preferably phenyl,
alkylsulfonate, particularly preferably methanesulphonate, C1-C10 arylsulphonate, particularly preferably p-toluenesulfonate.
Some particula
Other useful CAACs include the compounds described in U.S. 7,312,331 and Bertrand et al, Angew. Chem. Int. Ed. 2005, 44, 7236-7239.
24. For the inventive catalyst with the general formula (II), the following description is given:
L -G1
M© ΑΘ
I
25. G2-L2
(II)
wherein M, G1 and G2 have the same meaning as in general formula (I).
L1 and L2 representtwo electron donor ligands.
26. L1 and L2 are the same or different and represent a N-Heterocyclic carbene ("NHC"), substituted NHC, or cyclic alkyl amino carbene (CAAC) or substituted CAAC.
27. Preferably, t (Via) or (VIb)
(Via) (VIb)
wherein R - R , R and R have the same meanings as in the general formula (I).
28. A" is an anion, preferable a weak-coordinating anion or a non-coordinating anion. Examples of suitable weak-coordinating and non-coordinating anions include, but are not limited to, [BF4]~, [PF6]~, and [CIO4]", Trifluoromethanesulfonate, B[3,5-(CF3)2C6H3]4]~, tetrakis(pentafluorophenyl)borate, carborane anion.
29. As examples of the catalysts of the invention, the following structures may be mentioned:
Synthesis of Catalyst Compounds
30. Metal source
The invention describes the synthesis of catalysts by using different metal(l) sources depending of the type of catalysts being synthesized. Type (I) catalysts based on Cu(l) but not limited to can use the salts CuCI, CuBr and Cul, in case of X=Br it is also possible to use the dimer CuBrSMe2. Type (I) catalysts based on Au(l) but not limited to can use the salts AuCI, AuBr, Aul. Type (II) catalysts require the use of the complexes such as but not limited to [Cu(CH3CN)4]PF6, [Cu(CH3CN)4]BF4, [Au(CH3CN)4]PF6 , [Au(CH3CN)4]BF4
31. Procedure for the synthesis of catalysts
The catalyst compounds described in this invention may be synthesized by any methods known to those skilled in the art.
All reagent were used as purchased, Cu(l) and Au(l) sources were stored under argon in a glove box. Solvents were dried under standard procedures. In case of catalysts type (I), the synthesis procedure is exemplified by using the ligand 1 ,3-bis(3-sodiumsulfonate-2,4,6-trimethylphenyl)
-4,5-dihydroimidazolium chloride (sSIMes-HCI) which was synthesized according to the literature procedures9. For catalysts of type (II), the procedure of synthesis is exemplified by the ligand 1-(4-sulfonatebutyl)-3-(2,4,6-trimethylphenyl)-4,5-dihydroimidazolium (IMDS), which was synthesized according to literature procedures9.
Typical procedure for the synthesis of the catalysts with structure (I).
1 mmol of 1 ,3-bis(3-sodiumsulfonate-2,4,6-trimethylphenyl)-4,5-dihydroimidazolium chloride (sSIMes-HCI) (550mg); CuCI (1 mmol, 99mg) and potassium tert-butoxide (1 12.2mg, 1 mmol)are transferred in an oven dried vial in a glove box. The mixture was refluxed in 40ml of methanol for 12h. After the reaction time, the reaction mixture was filtrated through a pad of celite and the solvent concentrated under reduced pressure. The product 1 ,3-bis(3-sodiumsulfonate-2,4,6-trimethylphenyl) -4,5-dihydroimidazolyl-3-ylidene) Copper(l) chloride sSIMes-CuCI is obtained upon precipitation with ether in 86% yield.
1 H-NMR (500MHz, DMSO-d6): 6.93 (2H, ArH), 3.90 (s, 4H, CH2, N-(CH2J2-N), 2.52 (s, 6H, CH3-Ar), 2.50 (s, 6H, CH3-Ar), 2.22 (s, 6H, CH3Ar); ESI-MS (positive mode, MeOH): calc. 71 1 .23 (C32H48CuN406S2+), found 71 1 .21 Typical procedure for the synthesis of the catalysts with structure (II).
324.4mg of IMDS (1 mmol), 186.4mg of [Cu(CH3CN)4] (0.5mmol) in 9ml of dry THF are transferred in an oven-dried vial. To the reaction mixture is added 1 ml solution of potassium f-butoxide 1 M. The reaction mixture is stirred at room temperature for 16h. Thereafter the reaction mixture is filtrated through a pad of celite and concentrated under reduced pressure. The product(IMDS2-Cu) was obtained as a clear brown solid upon precipitation with ether.
1 H-NMR (500MHz, DMSO-d6): 6.91 (m, 2H, Ar), 3.78 (m, 2H, N-CH2), 3.76 (m, 2H, CH2-N), 3.53 (t, 2H, NCH2-CH3)S03K), 2.50 (t, 2H, N(CH2)3-CH2-S03K), 2.24 (s, 3H, CH3 Mes), 2.12 (s, 6H, CH3 Mes) 1.71 (m, 2H, NCH2-CH2-(CH2)2S03K), 1.61 (m, 2H, N (CH2)2-CH2-CH2S03K). 13C-NMR (125 MHz, DMSO-d6): 199.4, 137.05, 136, 135.5, 129, 51.54, 51 .46, 51 .02, 48.06, 20.56, 17.66; ESI-MS: calc. 281 .01 (C2i H26CICuN206N206S22-); found 281 .0. Conditions for the synthesis of triazoles by Alkyne Azide Cycloaddition using catalyst of the invention. The general reaction scheme for the synthesis of triazoles is depicted in scheme 2.
Scheme 2.
R10 - R12 are identical or different and are hydrogen, or represents alkyl, cycloalkyl, alkenyl, cycloalkenyl, substituted alkenyl, heteroalkenyl, heteroatom-containing alkynyl, alkenylene, alkynyl, substituted alkynyl, aryl, substituted aryl, heteroaryl, carboxylate, alkoxy, alkenyloxy, alkynyloxy, aryloxy, alkaryl, aralkyl, alkaryloxy, aralkyloxy, alkoxycarbonyl, alkylammonium, alkylamino-, alkylthio-, arylthio, alkylsulfonyl, alkylsulfinyl, dialkylamino, alkylsilyl or alkoxysilyl, where these radicals may each optionally all be substituted by one or more aforementioned groups defined for
R1-R8,
Optionally, one or more of the radicals R10 - R12 independently of one another can be substituted by one or more substituents, preferably straight or branched C-|-C10 alkyl, C3-C8 cycloalkyl, C1-C10 alkoxy
or C6-C24 aryl, where these aforementioned substituents may in turn be substituted by one or more radicals.
Alternatively, R11 and R12 independently of one another can be a metal, preferable a transition metal. In this manner multimetal entities can be prepared. Using this concept "a la carte" multimetal 1 ,2,3-triazole derivatives can be prepared.
When terminal alkynes are used, then R12=H .
When internal alkynes are used both R11 and R12 are different from hydrogen.
When acetylene gas is used both R11 and R12 are H.
When the inorganic azide HN3 is used, R10=H Reactants containing the alkyne functionality can be aliphatic or aromatic holding various electronic and steric properties. Both terminal and internal alkynes can be used in the practice of this invention. Both types of alkyne can contain functional groups. Examples of these functional groups include but are not limited to: carbon-carbon double bonds, ether groups, ester groups, ketyl groups, hydroxyl groups, chlorine groups, fluorine groups, trihydrocarbylsilyl groups, nitrogen atoms (e.g., as amino groups) and the like. Typically, for practical purposes of this invention, terminal alkynes have three to about twenty carbon atoms, and preferably about five to about twenty carbon atoms. The invention is also applicable to molecules holding multiple terminal alkyne groups in each endof the same molecule (e.g. but not limited to dendrimeric compounds) or holding multiple internal alkyne groups. Reactants containing the azide functionality include, but are not limited to: alkyl azides, ether azides, aryl azides and aralkyl azides. One or more functional groups can be included, e.g. cyano groups, nitro groups, amino groups, alkyl groups, halide groups, ester groups, ether groups, carboxylic groups, alkylsilyl groups, hydroxyl groups, alkenyl groups may be present in an aryl azide, in an aralkyl azide or in an alkyl azide. The alkyl portion of the alkyl azides can be branched, straight chain or cyclic group. Typically the alkyl azides have one to about twenty carbon atoms. In case of alkyl azides lower than 4 carbon atoms, a reaction procedure is followed, in which alkylazidesare generated/n-s/ft/ during the catalysis. The invention is also applicable to molecules holding multiple azide groups in each end of the same molecule, in which the final multi-azide is stable enough to perform the reactions under normal ambient conditions. In case of the inorganic azide HN3, this is generated in-situ by reaction of NaN3 trimethylsilylazide or CaN3with a hydrogen source,
Reactants containing biologically active macromolecules.
These include, but are not limited to peptides containing 2 to 20 aminoacid residues in which there is at least one azide group and/or one terminal alkyne group. The peptides susceptible for chemical transformation by the catalysts of this invention may hold every aminoacid present in the proteins, including those susceptible to complexation and/or sensitive to oxidation. The catalysts for the chemical ligation of peptides can be used with chemically protected peptides or unprotected peptides preferably in an aqueous media.
Solvent is not usually necessary in the processes of this invention, in which a neat process will be followed. For non-neat processes, generally it is recommended and preferred to conduct the processes of this invention in water, alcohol-water mixtures, DMSO-water mixtures, DMF-water mixtures or solvents different than water, preferably polar solvents like DMSO, acetone, acetonitrile, DMF or lower alcohols like methanol, ethanol or propanol.
When a solvent is employed, concentrations of the organic azide and the alkyne are each, independently, typically about 1 molar or higher, however, lower concentrations can be employed.
During the typical procedure of this invention, one mole of organic azide and one mole of alkyne are consumed for each mole of 1 ,2,3-triazole produced. Thus, it is recommended and preferred to use approximately equimolar amounts of organic azide and alkyne. The amounts of organic azide and alkyne are preferably such that the alkyne is in slight excess relative to the organic azide. In the specific case of acetylene gas, one atmosphere of acetylene gas is used by bubbling it into the reaction vessel. In case of the inorganic azide HN3, preferable, but not limited to, 1.5 mol HN3 per mol of alkyne is used.
Catalytic amounts of the catalysts of this invention are used. More particularly, the amount of catalyst is usually in the range of about 0.001 mol% to about 10mol% relative to the terminal alkyne. Preferably, about 0.5mol% to about 5mol% of the catalyst relative to the organic azide is employed. When the alkyne is a terminal alkyne, more preferred amounts of the catalyst are in the range of about 0.1 to 2mol% relative to the organic azide. When the alkyne is an internal alkyne, more preferred amounts of the catalyst are in the range of 5 to 10mol% relative to the organic azide. When peptide synthesis is conducted, it is preferred to use 5 equivalents of catalyst relative to the number of molecules of peptide.
Temperatures during the processes of the invention generally range from 0°C to 140°C and preferably from about room temperature to 1 10°C. When using acetylene gas as well as terminal alkynes with organic azides, the preferred working temperature is about room temperature, depending on the melting point of the alkyne, in which case the temperature will subject to that value. For internal alkynes and organic azides, temperatures from 50°C to about 80°C are preferred. For the inorganic azide HN3 and internal alkynes, the temperature range goes from 100 to 1 10°C. For aminoacids and peptides, the working temperature is preferably room temperature.
The processes of this invention comprises a wide range of reaction times, depending on the reaction conditions and the nature of the reactants involved in the catalysis, the reaction times arein the order of minutes (5 to 20minut.es) to hours (2 to 18hours) when acetylene gas, terminal alkynes with organic and/or inorganic azides are used. Internal alkynes react more slowly than do terminal alkynes and/or acetylene; therefore the reaction times are longer. Addition of a higher catalyst loading and/or increasing the reaction temperature may shorten the reaction time. When using acetylene gas and/or terminal alkynes, water may be used to shorten the reaction time. When the 1 ,2,3-triazole products are oils or low melting point solidslonger reaction times are required. Peptide synthesis requires longer reaction times, which are in the range of 12 to 48h, and 18h is the preferable reaction time.
The 1 ,2,3-triazole products are generally solids, which can be isolated by standard techniques such as precipitation or centrifugation and decantation, depending of the solvent used, it may be necessary to extract with an organic solvent and purify by column chromatography. For oily products, solvent extraction and/or column chromatography are the typical purification methods. Peptides are mainly purified by HPLC techniques.
The following examples are presented for purposes of illustration and are not intended to impose limitations on the scope of this invention.
Examples of synthesis of 1 ,2,3 triazoles by water-soluble N-heterocyclic carbene Cu(l) complexes. Catalytic studies on the different synthesized catalysts were performed by reactions under neat conditions and in solution. A comparison between Bis[1-(4-sodiumsulfonatebutyl)-3-(2,4,6-trimethylphenyl)-4,5-dihydroimidazolyl-3-ylidine] copper (I) hexafluorophosphate (IMDS2-CU) and the unsaturated Bis[1-(4-sodiumsulfonatebutyl) -3-(2,4,6-trimethylphenyl)-imidazolyl-3-ylidine] copper (I) hexafluorophosphate (IMS2-CU) under neat conditions is made and shown in graph 1 , (obtained by NMR analysis of the reaction mixture in CDCI3.) From graph 1 it is clear that the saturated Bis(NHC)-Cu catalysts are more reactive than the unsaturated homologs, about 3.5 times faster for the saturated catalyst (IMDS2-CU).
1 ,3-bis(3-sodiumsulfonate-2,4,6-trimethylphenyl)-4,5-dihydro imidazolyl-3-ylidine copper chloride (sSIMes-HCI) also showed good activity. Its activity is enhanced by the use of water due to its better affinity for it.
Synthesis of 1-benzyl-4-phenyl-1 ,2,3-triazole.
In a vial fitted with a screw cap, benzyl azide (1 mmol), phenylacetylene (1.05mmol) and catalyst (0.5mol%) were loaded. The reaction was allowed to proceed at room temperature and monitored by 1H NM analysis of aliquots. After total consumption of benzyl azide, the solid product is collected by filtration and washed with water and pentane. The results are given in table 1.
Run Catalyst Catalyst load Time (min) Yield (%)b
(mol%)a
1 IMDS2-Cu 0.5 10 99
2 IMS2-Cu 0.5 40 98
3 sSIMes-CuCI 0.5 35 98
4 IMDS2-Cu 0.1 60 95
a Mol% based on copper. Isolated yields are the average of at least two runs
General procedure for the synthesis of 1 ,4- and 1 ,4,5-substituted 1 ,2,3-triazoles under neat conditions.
In a vial fitted with a screw cap, an organic azide (1 .0 mmol), an alkyne (1 .05mmol) and catalyst Bis[1-(4-sodiumsulfonatebutyl)-3-(2,4,6-trimethylphenyl)-4,5-dihydroimidazolyl-3-ylidine] copper (I) hexafluorophosphate (IMDS2-Cu) (0.5mol% unless specified), are added. In case of molecules with two azide groups, a slight excess of terminal alkyne was used, say 1.1 mmol of alkyne per 0.5mmol of azide. When molecules holding two terminal alkyne functionalities, 1.05mmol of alkyne with 2mmol of azide were used. The reaction was allowed to proceed at room temperature (unless otherwise mentioned) and monitored by 1H NMR analysis of aliquots. After total consumption of the azide, the product as collected by filtration and washed with water and pentane. In case of oily products, these were extracted with dichloromethane and washed with aqueous NH4CI.Table 2 shows the results for all the triazoles produced. Reported conversions were determined by 1H NMR for at least two runs.
Table 2.
Triazole Triazole product structure Time Yield (%)
20
General procedure to synthesis an oligomer/polymer by reaction of an organic diazide and a terminal dialkyne by Alkyne Azide cycloaddition reaction under neat conditions.
In a vial fitted with a screw cap, 1 mmol ofdodecyl-1 ,12-diazide (1 .0 mmol), 1 mmol of 1 ,7-octadiyne and O.OI mmol of Bis[1 -(4-sodiumsulfonatebutyl)-3-(2,4,6-trimethylphenyl)
-4,5-dihydroimidazolyl-3-ylidine] copper (I) hexafluorophosphate (IMDS2-Cu) (1 mol%), are added. The reaction was allowed to proceed at room temperature and monitored by 1 H NMR analysis of aliquots. After total consumption of the azide, the product as collected by filtration and washed with water and pentane.Table 3 shows the evolution of the yield for the triazole oligomer poly[(1 -dodecyl-4-butyl)triazole)] produced by this reaction. The yield was determined by NMR analysis. Reported conversions are reported for at least two runs.
Table 3.
The invention comprises reactions in aqueous media for which the stability of the azide is too low to be isolated and used as pure. In this case a procedure in which the azide was formed in-situ from
alkyl and/or aryl halide is used. Water and room temperature are the preferred conditions, however, mixtures of water and a polar organic solvent like water/Dimethylsulfoxide, water/acetonitrile, water/alcohol (preferably lower alkyl alcohols: methanol, ethanol, isopropanol or fert-butanol) are also possible to use. The reactions may be carried out at a temperature range of 20-75°C. The order ofpreference for halide substituents in the alkyl or aryl group are: l>Br>CI.
. Synthesis of 1 ,4-substituted triazoles with in-situ formed azides.
In a vial fitted with a screw cap, alkyl halide (1 mmol) and NaN3 (1 .1 mmol) and 1 ml_ of water (unless otherwise noted) were added. The reaction was allowed to proceed at room temperature (unless otherwise noted) and monitored by 1H NMR analysis of aliquots. After total consumption of the starting azide, the solid product was collected by filtration and washed with water and pentane. In case of oily products, these were extracted with dichloromethane and washed with aqueous NH4CI. Examples of the triazoles synthesized by this procedure are given in table 4along with the yields and reaction times.
Table 4.
Triazole Structure of the triazole product Reaction time Temperature Yield
aFrom benzyl halide, b from alkyl iodide, c from allyl bromide, solvent: DMSO/water 1 :1 , d dibromoethane, solvent DMSO/water 1 : 1 . . Procedure for the synthesis of 1 -substituted triazoles under aqueous media using acetylene gas.
The invention comprises the synthesis of triazoles in which no substituent is present in the 4,5-positions, by using CaC2 and acetylene gas as the alkyne source. The following procedure is followed for CaC2 as the source of acetylene: To a vial fitted with screw cap with 1 mmol of azide, 1.5mmol of CaC2 and 1 mol% of catalyst relative to the azide is added 1 ml_ of deoxygenated water. The reaction was stirred at room temperature and monitored by TLC. After the reaction was completed, the mixture was acidified at pH~5 with HCI 0.1 N and the organic material was washed
with brine and extracted with diethyl ether. In case the product is not pure enough, column chromatography may be needed.
When using acetylene gas directly in the catalytic reaction, the procedure is as follows: To a vial fitted with screw cap with 1 mmol of azide and 0.5mol% of catalyst relative to the azide is added 1 ml_ of deoxygenated water, the vial is purged with acetylene gas and the reaction is stirred under 1 atm acetylene pressure at room temperature and monitored by TLC. After the reaction was completed, organic material was washed with brine and extracted with diethyl ether. The reaction is also applicable to azides prepared in-situ during the catalysis. Table 5 shows the results obtained with benzyl azide both in pure form and/n-s/it/formed in the reaction mixture. Solvents like DMSO, acetonitrile or mixtures of acetonitrile/water and DMSO/water may be used, providing excellent yields.
Table 5.
Triazole Triazole product Source of Solvent Time (h) Yield
product structure alkyne (%)
a From in-situ azide (benzyl bromide and sodium azide)
Synthesis of 1 ,4,5-substituted triazoles.
In a vial fitted with a screw cap, internal alkyne (1 .05mmol) and organic azide (1 mmol) and catalyst (5mol%)are added. The reaction was allowed to proceed at 80°C and monitored by 1H NMR analysis of aliquots. After the reaction time, the solid product was collected by filtration and washed with water and pentane.lf needed, the product is purified by column chromatography (ether:hexane to ether). Table 6 shows the results for the cyclization of 3-hexyne and diphenylacetylene with benzylazide.
Table 6.
Product triazole Triazole structure Reaction time (h) Yield (%)
40 48 38
41 48 72
Synthesis of 4-substituted triazoles with the inorganic hydrogen azide HN3.
Several reaction conditions were tested for this class of triazoles. The use of water was not satisfactory, generating very low yields. The use of acetic acid (AcOH) helped to increase the yield, however the yields were not satisfactory. The best results were achieved by using of DMSO in combination with a proton donor. Table 7 shows the results achieved by applying different conditions for the cyclization of phenylacetylene with HN3. From table 7, the procedure followed by using DMSO as the solvent, NaN3 and AcOH as the source of the hydrogen azide HN3 was chosen as the standard procedure.
Source of N3 " Co-catalyst Solvent Temp (°C) Yield (%)
(2eq) (2eq)
1 NaN3 H20 R.T. N. R.
2 NaN3 H20 100 N. R.
3 NaN3 HCI aq. H20 R.T. N. R.
4 NaN3 HCI aq. H20 100 N. R.
5 NaN3 K2C03 H20 100 N. R.
6 NaN3 NaHC03 H20 100 N. R.
7 NaN3 AcOH H20 100 <5
8 NaN3 AcOH + H20 100 <5
DIPEA
9 NaN3 AcOH H20 100 15
10 NaN3 AcOH + H20 100 24
NaOAc
1 1 NaN3 AcOH AcOH/H20 100 34
(ex)+NaOAc
(.1 eq)
12 TMSN3 H20 100 <5
13 TMSN3 AcOH H20 100 <15
14 NaN3 AcOH (ex) DMSO 1 10 96
15 NaN3 AcOH (2eq) DMSO 1 10 95
16 TMSN3 DMSO/H20 1 10 65
9: 1
17 TMSN3 DMSO/MeO 1 10 94
H 9:1 Procedure for the synthesis of 4-phenyl-1 ,2,3-triazole.
To vial fitted with a screw cap, add 1 mmol of the phenylacetylene and 2 mmol of NaN3 in a mixture of 2 mmol of AcOH and 1 mmol of dry DMSO. The reaction mixture is heated to 1 10°C for 12h. After the reaction time, water is added to the reaction mixture, extracted with ethyl acetate and the organic phase washed with brine, to give the product as a white solid, (table 7)
Procedure for the synthesis of 4,5-substituted 1 ,2,3-triazoles.
To vial fitted with a screw cap, add 1 mmol of the internal alkyne, 2 mmol of NaN3, 2 mmol of AcOH, catalyst (5mol%) and 1 mmol of dry DMSO. The reaction mixture is heated to 1 10°C for 12h. After the reaction time, water is added to the reaction mixture, extracted with ethyl acetate and the organic
phase washed with brine. The product is purified by column chromatography. Table 8 shows the results for the cyclization of 3-hexyne and diphenylacetylene.
Table 8.
Triazole product Triazole product Time (h) Yield
The synthesis of the peptides was followed under standard conditions using microscale techniques. It is possible the use of pH buffers to the solutions of peptides in order to maintain its properties, however highly acidic media may decrease the activity of the catalyst. Co-solvents are also susceptible to be used when the solubility of the peptides is a major issue. For that case MeOH, DMSO, NMP and/or HFIP (Hexafluoroisopropanol)may be used. For the case of poor soluble peptides, the first option for co-solvent should be HFIP. There is no preference on the order of addition for the substrates and/or catalyst. The catalysis is carried out preferably under ambient conditions of temperature and with long reaction times.
Procedure for the synthesis of peptides with 12 amino acid residues holding a terminal alkyne functionality and benzyl azide. To a solution of peptide 0.2M (0.5ml_) was added a solution of benzyl azide (5 eq) and catalyst (1.1 eq) in a mixture of HFIP and deoxygenated water (1 :4, 3ml_). The resulting solution was stirred at room temperature for 18h. After the reaction time, the mixture was analyzed by MALDI-MS mass spectrometry and the product separated by HPLC. In scheme 3 it is shown the structure of the peptides which were synthesized by this procedure.
Scheme 3.
Procedure for the synthesis of peptides with 12 amino acid residues holding an azide functionality and phenyl acetylene. To a solution of peptide 0.2M (0.5ml_) was added a solution of phenyl acetylene (5 eq) and catalyst (1.1 eq) in a mixture of HFIP and deoxygenated water (1 :4, 3ml_). The resulting solution was stirred at room temperature for 18h. After the reaction time, the mixture was analyzed by MALDI-MS mass spectrometry and the product separated by HPLC. In scheme 4 it is shown the structure of the peptides, which were synthesized by this procedure.
Scheme 4.
Procedure for the synthesis of polycarboxylate is exemplified using 2-Phenyloxirane, scheme 5.
Scheme 5.
A Teflon-lined stainless steel pressure vial was charged with phenyloxirane (10mmol, 1.14ml_), Bis[1-(4-sodiumsulfonatebutyl)-3-(2,4,6-trimethylphenyl)-4,5-imidazolyl-3-ylidine] copper (I) hexafluorophosphate (IMDS2-Cu) ( 9.25mg, 0.1 mol%) and DMAP (2.44mg, 0.2% mol). The reaction vessel was purged with CO2 and then taken to a 8 bar presure of CO2 for 2h at 100°C. The yield was determined by analysis of the 1H NMR spectrum, if necessary, separation was carried out by column cromatography. white solid.
1H NMR( 400 MHz, CDCI3): δ 7.47-7.35 (m, 5H), 5.68 (t, J= 8.0 Hz, 1 H), 4.80 (t, J = 8.4 Hz, 1 H), 4.35 (t, J = 8.4 Hz, 1 H). 13C NMR (100MHz, CDCI3): δ 154.8 (C=0), 135.8, 129.7, 129.2, 125.8, 77.9, 71 .1. IR: 1775 cm"1 (vCO).
The result are given in the table 9
Run Solvent Additive Pressure Temp. (°C) Yield-%
(0.2mol%) (bar)
1 Neat DMAP 1 80 <5
2 Neat — 10 100 10
3 Neat DMAP 10 100 12
4 Neat DMAP 10 120 92 General Procedure for Carboxylation of the Terminal Alkynesis exemplified using phenylacetylene, scheme 6.
Scheme 6.
In a 10ml_ Schlenk flask Bis[1-(4-sodiumsulfonatebutyl)-3-(2,4,6-trimethylphenyl) -4,5-imidazolyl-3-ylidine] copper (I) hexafluorophosphate (IMDS2-Cu) (1 mol%), 391 mg of Cs2C03 (1.2 eq) and 1.1 mL of Phenyl acetylene (1 mmol) were introduced. 1 ml_ of dry DMSO added to the reaction mixture and the flask was purged with CO2 and kept at CO2 atmosphere (balloon) for 16h at room temperature (24°C). After completion of the reaction, the reaction mixture was acidified to pH =
1 with concentrated HCI and extracted with diethyl ether, the combined oarganic layers were dried with anhydrous MgS04 and filtered and the solution concentrated in vacuum, affording pure product (phenyl propiolic acid) as white needles.
1H NMR δ = 11.18 ppm (br, s, COOH), 7.53-7.57 (m, 2H, ArH), 7.39-7.44 (m, 1 H, ArH), 7.30-7.35 (m, 2H, ArH). 13C NMR (100 MHz, CDCI3): δ = 154.86 (^ COOH), 133.01 , 131.43, 129.49, 119.45, 84.74, 82.27.
The results are given in table 10.
Run Solvent Additive (1 .2 eq.) Pressure (bar) Temp. (°C) Yield-%
1 DMF K2CO3 1 R.T. 45
2 DMF Cs2C03 1 R.T. 50
3 DMSO K2CO3 1 R.T. 25
4 DMSO Cs2C03 1 R.T. 95
62. The catalysts of this invention can also be immobilized in an efficient and straightforward manner, scheme 7. The catalysts can easily be recovered and reused for the next run as exemplified with
Scheme 7.
Preparation of the ionic Immobilized Bis[1-(4-sodiumsulfonatebutyl) -3-(2,4,6-trimethylphenyl) -4,5-imidazolyl-3-ylidine] copper (I) hexafluorophosphate on Amberlyte® IRA-402 resin beads. In a schlenk vial 0.15g (0.3meq/g dried mass) of the anion resin Amberlyte® IRA-402 are placed and treated at 60°C under vacuum for 2h. Afterwards, 47mg of Bis[1-(4-sodiumsulfonatebutyl)-3-(2,4,6-trimethylphenyl)-4,5-imidazolyl-3-ylidine] copper (I) hexafluorophosphate (.05mmol) and 2ml_ of deoxygenated water are added to the vial and the mixture stirred for 12h at room temperature. After the reaction time, the water phase is removed from the vial and the catalyst-impregnated beads washed with 5ml_ of deoxygenated water. The resin becomes brownish after the treatment.
Preparation of the ionic Immobilized 1 ,3-bis(3-sodiumsulfonate-2,4,6-trimethylphenyl) -4,5-dihydroimidazolyl-3-ylidene) Copper(l) iodide (SIMes-Cul) on Amberlyte® IRA-402 resin beads, s
sSIMes-CuI-IRA 402
Scheme 8.
In a Schlenk vial 0.15g (0.3meq/g dried mass) of the anion resin Amberlyte® IRA-402 are placed and treated at 60°C under vacuum for 2h. Afterwards, 35mg of 1 ,3-bis(3-sodiumsulfonate- 2,4,6-trimethylphenyl)-4,5-dihydroimidazolyl-3-ylidene) Copper(l) iodide (SIMes-Cul) (0.05mmol) and 2mL of deoxygenated water are added to the vial and the mixture stirred for 12h at room temperature. After the reaction time, the water phase is removed from the vial and the catalyst-impregnated beads are washed with 5ml_ of deoxygenated water. The resin becomes off-white colored after the treatment.
Procedure for the heterogeneous click reactions in aqueous media by immobilized IMDS2-Cu (MDS2-Cu-IRA 402).
In a vial containing the heterogeneous catalysts MDS2-Cu-IRA 402 (0.02eq Cu (I)), 78mg of NaN3 (1.2mmol), 130 μΙ_ of phenylacetylene (1.2mmol) and 122 μΙ_ of benzyl bromide (1 mmol) and 1 .5ml_ of deoxygenated water are added. The reaction mixture was stirred for 16h at room temperature. After the reaction time, the product was extracted with dichloromethane and the organic phase was dried under MgS04 and evaporated under vacuum. The aqueous phase was removed and replaced for fresh deo e 11.
Run Yield-%
1 100
2 95
3 93
4 93
5 92
Application of ultrasound accelerates the reactions, improving the yield and reducing the reaction time.
Immobilization of the catalysts of this invention is not limited to resins bearing ionic groups. The catalysts of this invention can be immobilized on any support (organic, inorganic, ionic liquids, etc.) bearing ionic groups, e.g. inorganic supports which have been modified so that they possess ionic groups, organic support bearing ionic groups, metal organic frameworks with ionic groups and so on. Some examples, but not intended to limit are giving above mentioned.
Except as may be expressly otherwise indicated, the article "a" "an" if and as used herein is not intended to limit, and should not be construed as limiting, the description or a claimto a single element to which the article refers. Rather, the article "a" "an" if and as used herein is intended to cover on or more such elements, unless the text expressly indicates otherwise.
Components referred to by chemical name or formula anywhere in thespecification or claims hereof, whether referredto in singular or plural, are identified as they exist prior to coming into contact with another substance referred to by chemical name or chemical type (e.g., another component, a solvent, or etc.). It matter not what chemical changes, transformations and/or reactions are the natural result of bringing the specified componentstogether under the conditions called for pursuant
to this disclosure. Thus the components are identified as ingredients to be brought together in connection with performing a desired operation or in forming a desired composition.
4. Each and every patent, patent application and printed publication referred to above is incorporated herein by reference in to the fullest extent permitted as a matter of law.
5. This invention is susceptible to considerable variation in its practice. Therefore, the foregoing description is not intended to limit, and should not be construed as limiting, the intention to the particular exemplifications presented herein above.
References
1. (a) Torri0e, C. W.; Christensen, C; Meldal, M. J. Org. Chem. 2002, 67, 3057. (b) Rostovtsev, V. V.; Green, L.
G. ; Fokin, V. V.; Sharpless, K. B. Angew. Chem. Int. Ed. 2002, 41, 2596.
2. Kolb, H. C; Finn, M. G.; Sharpless, K. B. Angew. Chem. Int. Ed. 2001, 40, 2004.
3. Bock, V. D.; Hiemstra, H.; van Maarseveen, J. H. Eur. J. Org. Chem. 2006, 51.
4. Katritzky, A. R.; Zhang, Y.; Singh, S. K. Heterocycles 2003, 60, 1225.
5. Kolb, H. C; Sharpless, K. B. Drug Discovery Today 2003, 8, 1 128; and references cited therein.
6. (a) Wu, P.; Feldman, A. K.; Nugent, A. K.; Hawker, C. J.; Scheel, A.; Voit, B.; Pyun, J.; Frechet, J. M. J.; Sharpless, K. B.; Fokin, V. V. Angew. Chem. Int. Ed. 2004, 43, 3928. (b) Helms, B.; Mynar, J. L.; Hawker, C. J.; Frechet, J. M. J. J. Am. Chem. Soc. 2004, 126, 15020. (c) Scheel, A.; Komber, H.; Voit, B. Macromol. Rapid Commun. 2004, 25,1 175. (d) Parent, M.; Mongin, O.; Kamada, K.; Katan, C; Blanchard-Desce, M. Chem. Commun. 2005, 2029. (e) van Steenis, D. J. V. C; David, O. R. P.; van Strijdonck, G. P. F.; van Maarseveen, J.
H. ; Reek, J. N. H. Chem.Commun. 2005, 4333. (f) Lutz J.-F. Angew. Chem. Int. Ed. 2007, 46, 1018- 1025. (g) Moses, IE.; Moorhouse, A.D. Chem.Soc.Rev.2007, 36, 1249.
7. (a) Lober, S.; Rodriguez-Loaiza, P.; Gmeiner, P. Org. Lett. 2003, 5, 1753. (b) Harju, K.; Vahermo, M.; Mutikainen, I.; Yli-Kauhaluoma, J. J. Comb. Chem. 2003, 5, 826. (c) Khanetskyy, B.; Dallinger, D.; Kappe, C. O. J. Comb. Chem. 2004, 6, 884. (d) Tornoe, C. W.; Sanderson, S. J.; Mottram, J. C; Coombs, G. H.; Meldal, M. J. Comb. Chem. 2004, (5, 312.
8. Genin, M. J.; Allwine, D. A.; Anderson, D. J.; Barbachyn, M. R.; Emmert, D. E.; Garmon, S. A.; Graber, D. R.; Grega, K. C; Hester, J. B.; Hutchinson, D. K.; Morris, J.; Reischer, R. D.; Stper, D.; Yagi, B. H. J. Med. Chem. 2000, 43, 953.
9. (a)Wamhoff, H. In Comprehensive Heterocyclic Chemistry; Katritzky, A. R.; Rees, C. W., Eds.; Pergamon: Oxford, 1984, 669-732. (b) C. Marshall, Mark F. W., Janet M. S. S. Synthesis 2006, 6, 1040- 1044. (c) L. R. Moore, Sheritta M. C, Matthew S. A., Hans-Jorg S., Scott T. G., Robin D. R., Marion C. K., Kevin H. S. Organometallics 2006, 25, 5151 -5158. (d) M. A. N. Virboul, Martin L., Maxime. A. S., Anthony L. S., Gerard V. K., Robertas J. M. K. G. Chem. Eur. J. 2009, 15, 9981-9986.
10. Buckle, D. R.; Rockell, C. J. M.; Smith, H.; Spicer, B. A. J. Med. Chem. 1986, 29, 2262.
1 1. Alvarez, R.; Velazquez, S.; San-Felix, A.; Aquaro, S.; De Clercq, E.; Perno, C. F.; Karlsson, A.; Balzarini, J.; Camarasa, M. J. J. Med. Chem. 1994, 37, 4194.
12. Michael, A. J. Prakt. Chem./Chem.-Ztg. 1893, 48, 94.
13. Huisgen, R. Pure Appl. Chem. 1989, 61, 613.
14. Huisgen, R. In 1,3-Dipolar Cycloaddition Chemistry; Padwa, A., Ed.; Wiley: New York, 1984; Vol. 1 , pp 1-176.
15. Himo, F.; Lovell, T.; Hilgraf, R.; Rostovtsev, V. V.; Noodleman, L.; Sharpless, K. B.; Fokin, V. V. /. Am. Chem. Soc. 2005, 727, 210.
16. Rostovtsev, V. V.; Green, L. G.; Fokin, V. V.; Sharpless, K. B. Angew. Chem., Int. Ed. 2002, 41, 2596.
17. Tornoe, C. W.; Christensen, C; Meldal, M. J. Org. Chem. 2002, 67, 3057.
18. Berrisford, D. J.; Bolm, C; Sharpless, K. B. Angew. Chem., Int. Ed. 1995, 34, 1059.
19. Tome, A. C. In Science of Synthesis; Storr, R. C, Gilchrist, T. L., Eds.; Thieme: New York, 2004; Vol. 13, pp 415-601.
20. Krivopalov, V. P.; Shkurko, O. P. Russ. Chem. Rev. 2005, 74, 339.
21. Perez-Balderas, F.; Ortega-Munoz, M.; Morales- Sanfrutos, J.; Hernandez-Mateo, F.; Calvo-Flores, F. G.; Calvo-Asin, J. A.; Isac-Garcia, J.; Santoyo-Gonzalez, F. Org. Lett. 2003, 5, 1951.
22. Malkoch, M.; Schleicher, K.; Drockenmuller, E.; Hawker, C. J.;Russell, T. P.; Wu, P.; Fokin, V. V. Macromolecules 2005, 38, 3663.
23. Wu, P.; Feldman, A. K.; Nugent, A. K.; Hawker, C. J.; Scheel, A.; Voit, B.; Pyun, J.; Frechet, J. M. J.; Sharpless, K. B.; Fokin, V. V. Angew. Chem., Int. Ed. 2004, 43, 3928.
24. Pachon, L. D.; van Maarseveen, J. H.; Rothenberg, G. Adv. Synth.Catal. 2005, 347, 81 1.
25. Molteni, G.; Bianchi, C. L.; Marinoni, G.; Santo, N.; Ponti, A. New J. Chem. 2006, 30, 1 137.
26. Chan, T. R.; Hilgraf, R.; Sharpless, K. B.; Fokin, V. V. Org. Lett. 2004, 6, 2853.
27. Ahlquist, M.; Fokin, V. V. J. Am. Chem. Soc. 2006, submitted.
28. (a) M. Aresta, A. Dibenedetto, Dalton Trans. 2007, 2975. (b) Carbon Dioxide as Chemical Feedstock; M. Aresta, Ed.;Wiley-VCH: Weinheim, 2010. (c) T. Sakakura, J.-C. Choi, H. Yasuda, Chem. Rev. 2007, 107, 2365. (d) T. Sakakura, . ohno, Chem. Commun. 2009, 1312. (e) M. Mikkelsen, M. Jorgensen, F.C. Krebs, Energy Environ. Sci. 2010, 3, 43. (f) R. Martin, A.W. Kleij, ChemSusChem 201 1 , 4, 1259.
29.111ustrative examples of new conversions include: (a) C. Das Neves Gomes, O. Jacquet, C. Villiers, P. Thuery, M. Ephritikhine, T. Cantat, Angew. Chem., Int. Ed. 2012, 51 , 187. (b) O. Jacquet, C. Das Neves Gomes, M. Ephritikhine, T. Cantat, J. Am. Chem. Soc. 2012, 134, 2934. (c) D. Yu, Y. Zhang, Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 20184.
30. (a) Decortes, A.; Castilla, A. M.; Kleij, A. W. Angew.Chem., Int. Ed. 2010, 49, 9822. (b) North, M.; Pasquale, R.; Young, C. Green Chem. 2010, 12, 1514. (c) Kember, M. R.; Knight, P. D.; Reung, P. T. R.; Williams, C. K. Angew. Chem., Int. Ed. 2009, 48, 931. (d) Darensbourg, D. J. Chem. Rev. 2007, 107, 2388. (e) Bruckmeier, C; Rieger, B.; Herrmann, W. A.; Kiihn, F. E. Angew. Chem., Int. Ed. 201 1 , 50, 8510.
31. (a) Paddock, R. L.; Nguyen, S. T. J. Am. Chem. Soc. 2001 , 123, 1 1498. (b) Decortes, A.; Kleij, A. W. ChemCatChem 201 1, 3, 831. (c) North, M.; Pasquale, R. Angew. Chem., Int. Ed. 2009, 48, 2946. (d) Buchard, A.; Kember, M. R.; Sandeman, K. G.; Williams, C. K. Chem. Commun. 201 1, 47, 212. (e) Lu, X.-B.; Darensbourg, D. J. Chem. Soc. Rev. 2012, 41, 1462. (f) Sujith, S.; Min, J. K.; Seong, J. E.; Na, S. J.; Lee, B. Y. Angew. Chem., Int. Ed. 2008, 47, 7306. (g) Cohen, C. T.; Chu, T.; Coates, G. W. J. Am. Chem. Soc. 2005, 127, 10869.
32. Minakata, S.; Sasaki, I.; Ide, T. Angew.Chem., Int. Ed. 2010, 49, 1309.
33. Tian, D.; Liu, B.; Gan, Q.; Li, FL; Darensbourg, D. J. ACS Catal. 2012, 2, 2029.
34. Clegg, W.; Harrington, R. W.; North, M.; Pasquale, R. Chem. Eur. J. 2010, 16, 6828
35. Correa A, Martin R, Angew Chem, Int Ed 2009, 48:6201-6204.
36. Bew SP in "Comprehensive Organic Functional Groups Transformation Π", eds AR Katritzky and RJK Taylor (Elsevier, Oxford) 2005, p 19.
37. (a) Ebert GW, et al. ,J Org Chem 2005, 70:4314-4317. (b) Olah GA, et al., J Am Chem Soc 2002, 124, 1 1379-1 1391. (c) Shi M, Nicholas KM., J Am Chem Soc. 1997, 1 19, 5057-5058. (d) Yeung CS, Dong VM, J Am Chem Soc. 2008, 130, 7826-7827. (e) Ochiai Hidenori, Jang Minsul, Hirano Koji, Yorimitsu Hideki, Oshima Koichiro, Org Lett. 2008, 10, 2681-2683. (f) Kobayashi Koji, Kondo Yoshinori, Org Lett 2009, 1 1, 2035-2037. (g) Ukai K, Aoki M, Takaya J, Iwasawa N, J Am Chem Soc 2006, 128, 8706-8707. (h) Ohishi T, Nishiura M, Hou Z, Angew Chem, Int Ed 2008, 47, 5792-5795. (i) Takaya Jun, Tadami Satoshi, Ukai Kazutoshi, Iwasawa Nobuharu, Org Lett 2008, 10, 2697-2700.
38. (a) Aoki M, Kaneko M, Izumi S, Ukai K, Iwasawa N. Chem. Commun 2004, 2568-2569. (b) Louie J, Gibby JE, Farnworth MV, Tekavec TN. J Am Chem Soc 2002, 124, 15188-15189.
39. (a)Takimoto M, Nakamura Y, Kimura K, Mori M. J Am Chem Soc 2004, 126, 5956-5957. (b) Williams CM, Johnson JB, Rovis T. J Am Chem Soc 2008, 130, 14936-14937.
40. Takaya J, Iwasawa N. J Am Chem Soc 2008, 130, 15254-15255.
41. Yu D., Zhang Y. PNAS, 2010, 107, 20184-20189.
Claims
1. A group 9 transition metal catalyst having structure (I) or (II):
Wherein M is a Group 9 transition metal,
L represents an electron donor ligand,
X is an anionic ligand,
G1 and G2 are individually functional groups, G1 and G2 are ionic groups,
L1 and L2 are individually electron donor ligands,
A is an anion, selected from the group consisting of weak-coordinating anions and
non-coordinating anions.
2. A catalyst as in claim 1 wherein M is Cu or Au.
3. A catalyst as in claim 1 wherein L, L1, or L2 is individually selected from the group consisting of N-Heterocyclic carbene, substituted N-Heterocyclic carbene, cyclic alkyl amino carbene, and substituted cyclic alkyl amino carbene.
4. A catalyst as in claim 3 wherein L, L1, or L2 is individually a N-Heterocyclic carbene having a general structure of th
(Ilia) (lllb)
wherein R1 - R8, R5 , R6 are individually selected from the group consisting of hydrogen, straight or branched C1-C30 alkyl, C3-C20 cycloalkyl, C2-C20 alkenyl, C2-C20 alkynyl, C6-C24 aryl, C1-C20 carboxylate, C1-C20 alkoxy, C2-C2o alkenyloxy, C2-C2o, alkynyloxy, C6-C2o aryloxy, C2-C2o alkoxycarbonyl, C1-C20 alkylthio, C6-C2o arylthio, C1-C20 alkylsulfonyl, C1-C20 alkyl sulfonate, C6-C2o aryl sulfonate, and C1-C20 alkyl sulfinyl, and one or more of the radicals R7 - R14, R1 , R12 are individually substituted by one or more substituents selected from straight or branched C1-C10 alkyl, C3-C8 cycloalkyl, C1-C10 alkoxy, and C6-C24 aryl, where these aforementioned substituents may in turn be substituted by one or more radicals selected from the group comprising halogen, especially chlorine or bromine, C1-C5 alkyl, C1-C5 alkoxy and phenyl.
5. A catalyst as in claim 3 wherein L, L1, or L2 is individually cyclic alkyl amino carbene having a general structure by the Formula (V):
(V)
Wherein the ring A is a 4-, 5-, 6-, or 7-membered ring, and Z is a linking group comprising from one to four linked vertex atoms selected from the group consisting of C, O, N, B, Al, P, S and Si with available valences optionally occupied by hydrogen, oxo or R-substituents, wherein R is independently selected
from the group comprising Ci to C-|2 hydrocarbyl groups, substituted Ci to C-|2 hydrocarbyl groups, and halides, and each 9 is independently a hydrocarbyl group or substituted hydrocarbyl group having 1 to 40 carbon atoms, C1-C10 alkylsulfonate, Ci-Cio arylsulphonate, an aryl group, a substituted aryl group, alkaryl and substituted alkaryl, aralkyl and substituted aralkyl, aryloxy and substituted aryloxy, R9 is selected from methyl, ethyl, propyl, isobutyl, n-butyl, pentyl, cyclopentyl, hexyl, cyclohexyl, octyl, cyclooctyl, nonyl, decyl, cyclodecyl, dodecyl, cyclododecyl, mesityl, adamantyl, phenyl, benzyl, toluyl, chlorophenyl, methanesulphonate, p-toluenesulfonate, phenol, and substituted phenol.
6. A catalyst as in claim 1 wherein X is selected from the group consisting of hydrogen, halogen, pseudohalogen, straight-chain or branched Ci-C3o alkyl, C6-C24 aryl, C1-C20 alkylthiol, C6-C2 arylthiol, C -C2o alkoxy, C6-C24 aryloxy, C2-C24 alkoxycarbonyl, C6-C20 aryloxycarbonyl, C2-C20 acyl, C2-C20 acyloxy, C3-C20 alkyl diketonate, C6-C24 aryl diketonate, C C2o carboxylate, Ci-C20 alkylsulfonato, C5-C2o arylsulfonato, Ci-C20 alkylsulfanyl, Cs-C2o arylsulfanyl, C1-C20 alkylsulfinyl, and Cs-C2o arylsulfinyl, any of which, with the exception of hydrogen and halide, are optionally further substituted with one or more groups selected from halide, Ci-C6 alkyl, Ci-C6 alkoxy, and C5-C20 aryl.
7. A catalyst as in claim 1 wherein X is selected from the group consisting of fluorine, chlorine, bromine, iodine, benzoate, C1-C5 carboxylate, C1-C5 alkyl, phenoxy, C1-C5 alkoxy, C1-C5 alkyl thiol, pentafluorophenoxy, and C C5 alkyl sulfonate.
8. A catalyst as in claim 1 wherein G1 and G2 are independently ionic groups or contain an ionic group selected from cationic or anionic groups bearing an anion or cation respectively as counterion.
9. A catalyst as in claim 8 wherein G1 and G2 are individually selected from the group consisting of quaternary ammonium, carboxylate, sulphonate (-SO3 "), -OSO3 ", -PO3 ", and OPO3 ".
10. A catalyst as in claim 8 or 9 wherein the counterion is selected from a resin, an organic support, an inorganic support or a metal organic framework bearing an anion or cation.
11 . A process which comprises contacting, in a reaction zone, at least one organic and/or inorganic azide, at least one acetylenic compound and at least one catalyst as claimed in any claim of 1 to 10.
12. A process as in claim 1 1 wherein acetylenic compound has at least one alkyne group, the alkyne group is selected from terminal or internal, the acetylenic compound has one or more terminal and/or one or more internal alkyne functionalities, the acetylenic compound can be liquid, solid, or gas.
13. A process as in claim 1 1 wherein wherein the reaction of an unprotected peptide holding one or more azide group(s) produces a new macromolecule holding one or more triazole moieties in the peptide product or the reaction of an unprotected peptide with one or more terminal alkyne groups produces a new macromolecule holding one or more triazole moieties in the peptide product.
14. A process as in claim 1 1 wherein the reaction media is in neat condition or selected from water, organic polar solvents, and mixtures of organic polar solvents and water; temperatures during the process generally range from 0°C to 140°C, reaction times generally range from 5 to 20 minutes or from 2 hours to 18 hours.
15. A process as in claim 11 wherein the amount of catalyst is in the range of 0.001 mol% to 10 mol% relative to the terminal alkyne.
16. A process as in claim 1 1 wherein when the acetylenic compound is acetylene gas, the acetylene gas is used at normal ambient conditions; when the acetylenic compound has at least one internal alkyne group, the acetylenic compound is used at temperatures between 20 and 140°C, preferably between 20 and 80°C.
17. A process as in claim 1 1 wherein either said organic azide is added in the reaction mixture in which the alkyne and the catalyst are already present, or said organic azide is formed in a reaction mixture to which the alkyne and the catalyst are being or will be added.
18. A process as in claim 1 1 wherein either said organic azide is formed in a reaction mixture from an organic halide and a metal azide.
19. A process as in claim 18 wherein water is present in said reaction mixture during said process.
20. A process which comprises utilizing C02 as a carbon feedstock to react with epoxides and in the presence of catalyst as claimed in any claim of 1 to 10 to produce cyclic or polycarbonate products.
21 . A process which comprises utilizing C02 as a carbon feedstock to react with terminal alkynes and in the presence of catalyst as claimed in any claim of 1 to 10 to produce carboxylic acids.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201380077684.9A CN105531028B (en) | 2013-04-26 | 2013-04-26 | Group 9 transition metal catalysts and methods of using the same |
| PCT/CN2013/074755 WO2014172885A1 (en) | 2013-04-26 | 2013-04-26 | Group 9 transition metal catalysts and process for use of same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2013/074755 WO2014172885A1 (en) | 2013-04-26 | 2013-04-26 | Group 9 transition metal catalysts and process for use of same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014172885A1 true WO2014172885A1 (en) | 2014-10-30 |
Family
ID=51791010
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2013/074755 Ceased WO2014172885A1 (en) | 2013-04-26 | 2013-04-26 | Group 9 transition metal catalysts and process for use of same |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN105531028B (en) |
| WO (1) | WO2014172885A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105061340A (en) * | 2015-08-31 | 2015-11-18 | 河南师范大学 | Method for preparing 1,2,3-triazoie compound through active-carbon-carried nano-copper catalyst in catalyzed synthesis mode |
| CN105153051A (en) * | 2015-08-31 | 2015-12-16 | 河南师范大学 | Method for catalytically synthesizing 1, 2, 3-triazole compounds by aid of carboxymethyl cellulose/nanometer copper |
| US10758623B2 (en) | 2013-12-09 | 2020-09-01 | Durect Corporation | Pharmaceutically active agent complexes, polymer complexes, and compositions and methods involving the same |
| CN113559924A (en) * | 2021-07-28 | 2021-10-29 | 绍兴七轩新材料科技有限公司 | An ionic liquid catalyst and its preparation method and application |
| CN117720855A (en) * | 2023-11-20 | 2024-03-19 | 河南大自然家居有限公司 | Anticorrosive coating for wooden floors and preparation method thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009152219A1 (en) * | 2008-06-11 | 2009-12-17 | Henkel Corporation | (n-heterocyclic carbene) copper salt complex as latent click catalyst |
| WO2011094580A2 (en) * | 2010-01-28 | 2011-08-04 | Alnylam Pharmaceuticals, Inc. | Chelated copper for use in the preparation of conjugated oligonucleotides |
| WO2012025195A1 (en) * | 2010-08-25 | 2012-03-01 | Universität Heidelberg | Molecular catalysts based on dinuclear or polynuclear copper complexes |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101402644B (en) * | 2008-10-31 | 2010-12-01 | 浙江大学 | Preparation method of metal nitrogen heterocyclic carbene complex |
| CN101824052A (en) * | 2010-05-19 | 2010-09-08 | 中国科学院上海有机化学研究所 | Method for catalytically synthesizing phosphine compounds by using N-heterocyclic carbene metal complexes |
-
2013
- 2013-04-26 CN CN201380077684.9A patent/CN105531028B/en active Active
- 2013-04-26 WO PCT/CN2013/074755 patent/WO2014172885A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009152219A1 (en) * | 2008-06-11 | 2009-12-17 | Henkel Corporation | (n-heterocyclic carbene) copper salt complex as latent click catalyst |
| WO2011094580A2 (en) * | 2010-01-28 | 2011-08-04 | Alnylam Pharmaceuticals, Inc. | Chelated copper for use in the preparation of conjugated oligonucleotides |
| WO2012025195A1 (en) * | 2010-08-25 | 2012-03-01 | Universität Heidelberg | Molecular catalysts based on dinuclear or polynuclear copper complexes |
Non-Patent Citations (2)
| Title |
|---|
| LI, PINHUA ET AL.: "SiO2-NHC-Cu(I): an efficient and reusable catalyst for [3+2] cycloaddition of organic azides and terminal alkynes under solvent-free reaction conditions at room temperature", TETRAHEDRON, vol. 64, 17 September 2008 (2008-09-17), pages 10825 - 10830 * |
| WAN, LI ET AL.: "Multicomponent Synthesis of 1,2,3-Triazoles in Water Catalyzed by Silica-Immobilized NHC-Cu(I).", CATAL. LETT., vol. 142, 28 July 2012 (2012-07-28), pages 1134 - 1140 * |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10758623B2 (en) | 2013-12-09 | 2020-09-01 | Durect Corporation | Pharmaceutically active agent complexes, polymer complexes, and compositions and methods involving the same |
| US11529420B2 (en) | 2013-12-09 | 2022-12-20 | Durect Corporation | Pharmaceutically active agent complexes, polymer complexes, and compositions and methods involving the same |
| CN105061340A (en) * | 2015-08-31 | 2015-11-18 | 河南师范大学 | Method for preparing 1,2,3-triazoie compound through active-carbon-carried nano-copper catalyst in catalyzed synthesis mode |
| CN105153051A (en) * | 2015-08-31 | 2015-12-16 | 河南师范大学 | Method for catalytically synthesizing 1, 2, 3-triazole compounds by aid of carboxymethyl cellulose/nanometer copper |
| CN105061340B (en) * | 2015-08-31 | 2018-03-20 | 河南师范大学 | The method that activated carbon supported Nanometer Copper catalyst synthesizes 1,2,3 triazole compounds |
| CN105153051B (en) * | 2015-08-31 | 2018-06-22 | 河南师范大学 | The method that carboxymethyl cellulose/Nanometer Copper catalyzes and synthesizes 1,2,3- triazole compounds |
| CN113559924A (en) * | 2021-07-28 | 2021-10-29 | 绍兴七轩新材料科技有限公司 | An ionic liquid catalyst and its preparation method and application |
| CN113559924B (en) * | 2021-07-28 | 2023-10-03 | 绍兴七轩新材料科技有限公司 | An ionic liquid catalyst and its preparation method and application |
| CN117720855A (en) * | 2023-11-20 | 2024-03-19 | 河南大自然家居有限公司 | Anticorrosive coating for wooden floors and preparation method thereof |
| CN117720855B (en) * | 2023-11-20 | 2025-08-01 | 河南大自然家居有限公司 | Anticorrosive paint for wood floor and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN105531028B (en) | 2019-06-25 |
| CN105531028A (en) | 2016-04-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Firouzabadi et al. | The facile and efficient Michael addition of indoles and pyrrole to α, β-unsaturated electron-deficient compounds catalyzed by aluminium dodecyl sulfate trihydrate [Al (DS) 3]· 3H 2 O in water | |
| CN102083798B (en) | With the ruthenium olefin metathesis catalyst with the N-heterocyclic carbene ligand replacing skeleton | |
| Wang et al. | Colorimetric fluoride sensors based on deprotonation of pyrrole–hemiquinone compounds | |
| EP3164409B1 (en) | Group 8 transition metal catalysts and method for making same and process for use of same in metathesis reaction | |
| Samantaray et al. | Silver N-heterocyclic carbene complexes as initiators for bulk ring-opening polymerization (ROP) of l-lactides | |
| WO2014172885A1 (en) | Group 9 transition metal catalysts and process for use of same | |
| Belmonte et al. | Self-assembly of Zn (salphen) complexes: steric regulation, stability studies and crystallographic analysis revealing an unexpected dimeric 3, 3′-t-Bu-substituted Zn (salphen) complex | |
| JP2013099739A (en) | Catalyst complex with carbene ligand | |
| Mendoza-Espinosa et al. | Copper (II) complexes supported by click generated mixed NN, NO, and NS 1, 2, 3-triazole based ligands and their catalytic activity in azide–alkyne cycloaddition | |
| CN105131042B (en) | Chelating type N-heterocyclic carbine palladium compound containing heteroaryl and preparation method thereof | |
| Manna et al. | Influence of the cavity dimension on encapsulation of halides within the capsular assembly and side-cleft recognition of a sulfate–water cluster assisted by polyammonium tripodal receptors | |
| Li et al. | Synthesis, structural characterization and reactivity of aluminium complexes supported by benzotriazole phenoxide ligands: air-stable alumoxane as an efficient catalyst for ring-opening polymerization of L-lactide | |
| Mahkam et al. | Synthesis and characterization of new 5-substituted 1H-tetrazoles in water: a greener approach | |
| CN112812045A (en) | Onium salt organic catalyst and preparation method and application thereof | |
| Yang et al. | Al and Zn complexes bearing N, N, N-tridentate quinolinyl anilido-imine ligands: synthesis, characterization and catalysis in l-lactide polymerization | |
| Xu et al. | A receptor incorporating OH, NH and CH binding motifs for a fluoride selective chemosensor | |
| Kim et al. | Endo-and/or exocyclic silver (i) and mercury (ii) complexes of an NO 2 S 2-macrocycle: effect of ligand ratio and anion | |
| Anitha et al. | Ruthenium (II) 8-quinolinolates: Synthesis, characterization, crystal structure and catalysis in the synthesis of 2-oxazolines | |
| Garnes-Portoles et al. | Synthesis of dehydromuscone by an alkene metathesis macrocyclization reaction at 0.2 M concentration | |
| de la Reberdière et al. | Synthesis of a new family of protected 1, 4, 7, 10-tetraazacyclododecane-1, 4, 7-triacetic acid derivatives with thioctic acid pending arms | |
| Zhou et al. | Synthesis, structure and catalytic activity of alkali metal-free bent-sandwiched lanthanide amido complexes with calix [4]-pyrrolyl ligands | |
| JP2021513572A (en) | Reaction of olefin derivatives in the presence of metathesis catalyst | |
| Walstrom et al. | Unexpected selectivity in electrophilic attack on (PNP) RuN | |
| EP3092263A1 (en) | Polyethylene glycol substituted acyl borates | |
| Rubio et al. | Heterotopic silver–NHC complexes: from coordination polymers to supramolecular assemblies |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201380077684.9 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13882922 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13882922 Country of ref document: EP Kind code of ref document: A1 |