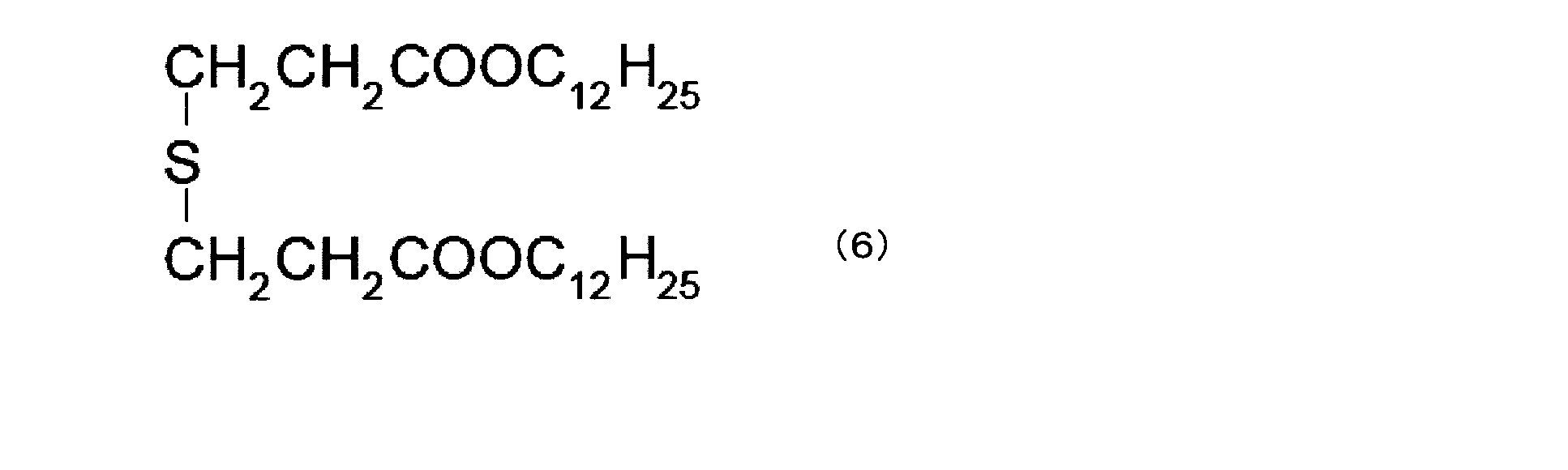WO2014141984A1 - Solvent-soluble elastic resin composition - Google Patents
Solvent-soluble elastic resin composition Download PDFInfo
- Publication number
- WO2014141984A1 WO2014141984A1 PCT/JP2014/055722 JP2014055722W WO2014141984A1 WO 2014141984 A1 WO2014141984 A1 WO 2014141984A1 JP 2014055722 W JP2014055722 W JP 2014055722W WO 2014141984 A1 WO2014141984 A1 WO 2014141984A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- resin composition
- solvent
- less
- carbon atoms
- mol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J167/00—Adhesives based on polyesters obtained by reactions forming a carboxylic ester link in the main chain; Adhesives based on derivatives of such polymers
- C09J167/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
- C09J167/025—Polyesters derived from dicarboxylic acids and dihydroxy compounds containing polyether sequences
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/13—Phenols; Phenolates
- C08K5/134—Phenols containing ester groups
- C08K5/1345—Carboxylic esters of phenolcarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/51—Phosphorus bound to oxygen
- C08K5/53—Phosphorus bound to oxygen bound to oxygen and to carbon only
- C08K5/5317—Phosphonic compounds, e.g. R—P(:O)(OR')2
- C08K5/5333—Esters of phosphonic acids
Definitions
- the present invention relates to an elastic resin composition that is soluble in a solvent and can retain adhesive properties in a wide temperature range.
- the material used for sealing electrical and electronic parts must have electrical insulation from the outside, and it is necessary to follow the shape of the electrical and electronic parts reliably and not to cause peeling. Is done.
- adhesive characteristics in a wide temperature range are required to achieve the purpose of use.
- Patent Document 1 discloses an elastic resin composition composed of a polyether segment and a crystalline polyester segment. By copolymerizing a polyether glycol having a high molecular weight, conditions such as electrical insulation are disclosed. It is disclosed that high elasticity can be maintained while satisfying the above. With this resin composition, a good molded product can be obtained, and the polyester resin composition can be applied to general electric and electronic parts.
- An object of the present invention is to solve the above-mentioned problems, and is an elastic resin composition (hereinafter simply referred to as “resin composition”) that has good solvent solubility and can maintain adhesive properties in a wide temperature range. .) To provide.
- the present invention is an elastic resin composition having solvent solubility shown below and capable of maintaining adhesive properties in a wide temperature range.
- the storage elastic modulus (E'20) at 20 ° C. when measured at a frequency of 10 Hz is 800 to 2000 MPa
- the storage elastic modulus (E'80) at 80 ° C. is 0.5 to 2.5 MPa
- the ratio of the storage elastic modulus at 80 ° C. to the storage elastic modulus at 20 ° C. (E′80 / E′20) is in the range of 0.025% to 0.25%.
- an antioxidant (c) containing phenolic O atoms in the molecule is preferable to contain 0.05 to 5 parts by weight of an antioxidant (c) containing phenolic O atoms in the molecule with respect to 100 parts by weight of the solvent-soluble elastic resin composition.
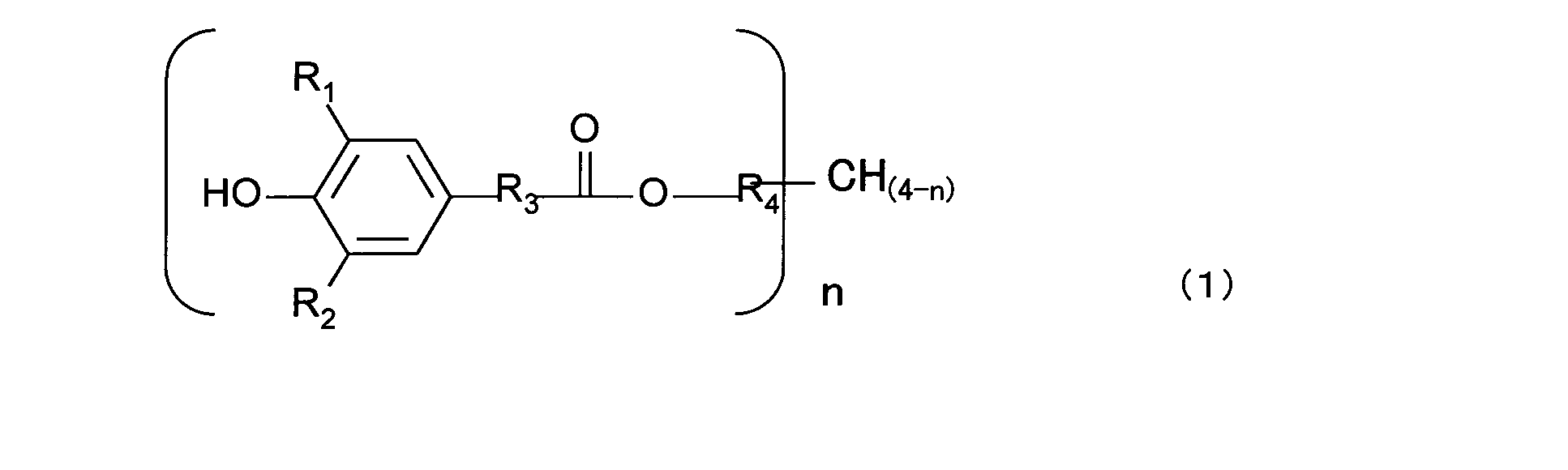
- the antioxidant (c) containing the phenolic O atom in the molecule is a compound (c1) represented by the general formula (1) and / or a compound (c2) represented by the general formula (2).
- General formula (1); (R 1 and R 2 are each independently a linear or branched alkyl group having 1 to 4 carbon atoms or hydrogen.
- R 3 is an alkylene group having 1 to 4 carbon atoms.
- R 4 Is a linear or branched alkyl group having 5 to 10 carbon atoms, n represents an integer of 1 to 4.
- R 5 and R 6 are each independently a linear or branched alkyl group having 1 to 4 carbon atoms, or hydrogen.
- R 7 is an alkylene group having 1 to 4 carbon atoms.
- R 8 And R 9 are each independently an alkyl group having 5 to 10 carbon atoms which may be linear or branched.)
- the solvent-soluble elastic resin composition (hereinafter also simply referred to as “resin composition”) according to the present invention has good solvent solubility, excellent solution stability, and a wide range from room temperature (about 20 ° C.) to 80 ° C. Maintains adhesiveness and adhesive heat resistance in the temperature range.
- the crystalline polyether segment (a) used in the present invention is not particularly limited, but is preferably mainly crystalline polyalkylene glycol. It is preferable in terms of heat resistance because it is crystalline. Specific examples include polyethylene glycol (hereinafter also referred to as PEG), polytrimethylene glycol (hereinafter also referred to as PPG), polytetramethylene glycol (hereinafter also referred to as PTMG), and the like. It is not limited. Among these, PTMG is preferable from the viewpoint of heat resistance.
- the number average molecular weight of the crystalline polyether segment (a) is preferably 230 or more, more preferably 400 or more, and further preferably 800 or more. When the number average molecular weight of the crystalline polyether segment (a) is less than 230, there is a tendency that the resin composition cannot be given flexibility and problems such as inflexibility cannot be exhibited.
- the number average molecular weight of the crystalline polyether segment (a) is preferably 5000 or less, more preferably 4000 or less, and further preferably 3000 or less. When the number average molecular weight exceeds 5,000, the compatibility with other copolymerization components is poor, and problems such as inability to copolymerize tend to occur.
- the term “crystallinity” means that the temperature is raised from ⁇ 100 ° C. to 300 ° C. at 20 ° C./min using a differential scanning calorimeter (DSC) and shows a clear melting peak in the temperature raising process. .
- the amorphous polyester segment (b) is preferably a polyester obtained by polymerizing an acid component and a glycol component, and is preferably amorphous in terms of solvent solubility.
- Amorphous means that the temperature is raised from ⁇ 100 ° C. to 300 ° C. at 20 ° C./min using a differential scanning calorimeter (DSC) and does not show a clear melting peak in the temperature raising process.
- DSC differential scanning calorimeter
- the acid component constituting the amorphous polyester segment (b) used in the present invention is not particularly limited, but an aromatic dicarboxylic acid is preferable.
- the aromatic dicarboxylic acid preferably has 8 to 14 carbon atoms, more preferably 9 to 13 carbon atoms.
- the aromatic dicarboxylic acid is preferably contained in an amount of 50 mol% or more when the total acid component of the resin composition is 100 mol% in order to improve the elasticity of the resin composition, more preferably 60 mol% or more. It is more preferably at least mol%, particularly preferably at least 95 mol%, and may be 100 mol%.
- aromatic dicarboxylic acid examples include, but are not particularly limited to, one or more acids selected from the group consisting of terephthalic acid, isophthalic acid, and naphthalenedicarboxylic acid, which have good reactivity with the glycol component, and polymerization. In terms of productivity and productivity.
- the total of terephthalic acid, isophthalic acid and naphthalenedicarboxylic acid is preferably 50 mol% or more, more preferably 60 mol% or more, and more preferably 80 mol% or more in the total acid component of the resin composition.
- the total acid component may be composed of one or more acids selected from the group consisting of terephthalic acid, isophthalic acid and naphthalenedicarboxylic acid.
- naphthalenedicarboxylic acid any of 1,4-naphthalenedicarboxylic acid, 1,8-naphthalenedicarboxylic acid, 2,3-naphthalenedicarboxylic acid, 2,6-naphthalenedicarboxylic acid, and 2,7-naphthalenedicarboxylic acid may be used. Of these, 2,6-naphthalenedicarboxylic acid is preferred.
- amorphous polyester segment (b) include diphenyl dicarboxylic acid, aromatic dicarboxylic acid such as 5-sodium sulfisophthalic acid, cyclohexane dicarboxylic acid, and alicyclic dicarboxylic acid such as tetrahydrophthalic anhydride.
- dicarboxylic acids such as aliphatic dicarboxylic acids such as succinic acid, glutaric acid, adipic acid, azelaic acid, sebacic acid, dodecanedioic acid, dimer acid and hydrogenated dimer acid.
- the copolymerization ratio of these dicarboxylic acid components is preferably less than 50 mol%, more preferably less than 40 mol%, still more preferably less than 20 mol%, and more preferably 5 mol% when the total acid component of the resin composition is 100 mol%. Less than is particularly preferable, and it may be 0 mol%.
- the other acid component constituting the amorphous polyester segment (b) it is possible to use a tri- or higher functional polycarboxylic acid such as trimellitic acid or pyromellitic acid.
- the copolymerization ratio of the trifunctional or higher polycarboxylic acid is preferably 10 mol% or less, and more preferably 5 mol% or less from the viewpoint of preventing gelation of the resin composition.
- the glycol component constituting the amorphous polyester segment (b) used in the present invention is not particularly limited, but is preferably an aliphatic glycol and / or an alicyclic glycol, and in particular, a branched aliphatic glycol and / or a branched fat.
- a cyclic glycol is more preferred.
- the number of carbon atoms of the branched aliphatic glycol and / or the branched alicyclic glycol is 4 or more, more preferably 5 or more, still more preferably 6 or more, preferably 20 or less, more preferably It is 15 or less, more preferably 10 or less, and particularly preferably 8 or less.
- the glycol component is preferably 20 mol% or more, more preferably 25 mol% or more, still more preferably 30 mol% or more, and more preferably 40 mol% or more when the total glycol component of the resin composition is 100 mol%. Particularly preferred. Moreover, it is preferable that it is 99 mol% or less, 97 mol% or less is more preferable, and 95 mol% or less is further more preferable. If it is less than 20 mol%, the solvent solubility may be insufficient, and if it exceeds 99 mol%, the amount of crystalline polyether segment (a) will be insufficient, so the storage elastic modulus at 20 ° C. (E'20) exceeds 2000 MPa, and the storage elasticity may be too high, resulting in a decrease in adhesion.
- the branched aliphatic glycol is an aliphatic glycol having a side chain group having 1 or more carbon atoms in a linear hydrocarbon
- the branched alicyclic glycol is a cyclic carbon of an alicyclic hydrocarbon. Is an alicyclic glycol having a side chain group having 1 or more carbon atoms.
- examples of the branched aliphatic glycol include neopentyl glycol, 2-methyl-1,3-propanediol, 1,2-butanediol, 1,3-butanediol, 2,3 -Butanediol, 3-methyl-1,5-pentanediol, 2,2,4-trimethyl-1,3-pentanediol, 2-ethyl-1,3-hexanediol, 2,2-dimethyl-3-hydroxy Propyl-2 ′, 2′-dimethyl-3-hydroxypropanoate, 2-normalbutyl-2-ethyl-1,3-propanediol, 3-ethyl-1,5-pentanediol, 3-propyl-1,5 -Pentanediol, 2,2-diethyl-1,3-propanediol, 2-butyl-2-ethyl-1,3-propanediol, 2-butyl
- branched alicyclic glycols examples include 1,3-bis (Hydroxymethyl) cyclohexane, 1,4-bis (hydroxymethyl) cyclohexane, 1,4-bis (hydroxyethyl) cyclohexane, 1,4-bis (hydroxypropyl) cyclohexane, 1,4-bis (hydroxymethoxy) cyclohexane, 1,4-bis (hydroxyethoxy) cyclohexane, 2,2-bis (4-hydroxymethoxycyclohexyl) propane, 2,2-bis (4-hydroxyethoxycyclohexyl) propane, bis (4-hydroxycyclohexyl) methane, 2, 2-bis (4-hydroxycyclohex Sil) propane, 3 (4), 8 (9) -tricyclo [5.2.1.0 2 , 6 ] decandimethanol, hydrogenated bisphenol A, ethylene oxide adduct of hydrogenated bisphenol A, hydrogenated bisphenol A
- These propylene oxide adducts can be used
- the content of the short-chain glycol having 3 or less carbon atoms is preferably 70 mol% or less, more preferably 60 mol% or less, and even more preferably 50 mol% or less, when the total glycol component of the resin composition is 100 mol%. 40 mol% or less is particularly preferable. If the amount is too large, the compatibility with the crystalline polyether segment (a) tends to be poor, and the polymerizability tends to deteriorate.
- Specific examples of the short-chain glycol having 3 or less carbon atoms include, but are not limited to, ethylene glycol, 1,2-propylene glycol, or 1,3-propylene glycol.
- linear glycol components having 4 or more carbon atoms can be used.
- the blending amount of the linear glycol is preferably 20 mol% or less, more preferably 10 mol% or less, further preferably 5 mol% or less, and may be 0 mol%.
- a tri- or higher functional polyol such as glycerin, trimethylolpropane, pentaerythritol, etc.
- the content is made 10 mol% or less from the viewpoint of preventing gelation of the resin composition.
- it is more preferably 5 mol% or less. If the amount is too large, the resin composition may be gelled.
- the solvent-soluble elastic resin composition according to the present invention contains the crystalline polyether segment (a) and the amorphous polyester segment (b), and has a storage elastic modulus at 20 ° C. (measured at a frequency of 10 Hz).
- E'20 is 800 to 2000 MPa
- storage elastic modulus at 80 ° C. is 0.5 to 2.5 MPa
- storage elastic modulus at 80 ° C. relative to storage elastic modulus at 20 ° C. (E'80 / E'20) is a resin composition in the range of 0.025% to 0.25%.
- the resin composition of the present invention needs to contain a crystalline polyether segment (a) and an amorphous polyester segment (b).
- the crystalline polyether segment (a) and the amorphous polyester segment (b) Is preferably copolymerized.
- the resin composition is preferably amorphous.
- Amorphous means that the temperature is raised from ⁇ 100 ° C. to 300 ° C. at 20 ° C./min using a differential scanning calorimeter (DSC) and does not show a clear melting peak in the temperature raising process.
- the content of the crystalline polyether segment (a) in the resin composition of the present invention is preferably 1 mol% or more, preferably 3 mol% or more when the total glycol component of the resin composition is 100 mol%. More preferably, it is 5 mol% or more. Moreover, 80 mol% or less is preferable, 75 mol% or less is more preferable, 70 mol% or less is more preferable, 60 mol% or less is especially preferable.
- the storage elastic modulus (E′20) at 20 ° C. exceeds 2000 MPa, and sufficient storage elasticity may not be obtained.
- the storage elastic modulus (E′80) at 80 ° C. becomes less than 0.5 MPa, and sufficient storage elasticity may not be obtained.
- the resin composition of the present invention needs to have a storage elastic modulus (E′20) at 20 ° C. of 800 to 2000 MPa.
- E′20 storage elastic modulus
- it is 900 MPa or more, More preferably, it is 1000 MPa or more, More preferably, it is 1100 MPa or more.
- 1900 MPa or less is preferable, 1800 MPa or less is more preferable, and 1700 MPa or less is more preferable.
- the resin composition of the present invention is required to have a storage elastic modulus (E′80) at 80 ° C. of 0.5 to 2.5 MPa.
- E′80 storage elastic modulus
- it is 0.8 MPa or more, More preferably, it is 1.0 MPa or more, More preferably, it is 1.5 MPa or more.
- 2.4 MPa or less is preferable, 2.2 MPa or less is more preferable, and 2.0 MPa or less is more preferable.
- the ratio of the storage elastic modulus at 80 ° C. to the storage elastic modulus at 20 ° C. is in the range of 0.025% to 0.25%. is necessary. Preferably it is 0.03% or more, More preferably, it is 0.05% or more, More preferably, it is 0.1% or more. Moreover, less than 0.25% is preferable, 0.24% or less is more preferable, and 0.23% or less is more preferable.
- the storage elastic modulus of the resin composition of the present invention is measured by the following method. That is, the resin composition is placed on a heat press adjusted to about 200 ° C. via a polyimide film. Hold for about 20 seconds at a pressure of about 2 N / mm 2 to obtain a sheet sample of about 1 mm thickness. This is cut into a length of about 15 mm (excluding the grip allowance) and a width of about 4 mm, set in a dynamic viscoelasticity measuring device “DVA-200” manufactured by IT Measurement Control Co., Ltd., and measured in a tensile mode. .
- the measurement condition is that the frequency is fixed at 10 Hz, and scanning is performed from ⁇ 100 ° C. until the measurement becomes impossible (maximum 250 ° C.) at a heating rate of 4 ° C./min.
- the lower limit of the number average molecular weight of the resin composition of the present invention is not particularly limited, but is preferably 3,000 or more, more preferably 5,000 or more, and further preferably 7,000 or more.
- the upper limit of the number average molecular weight is not particularly limited, but is preferably 50,000 or less, more preferably 40,000 or less, and still more preferably 30,000 or less. If the number average molecular weight is less than 3,000, elasticity may be insufficient, and if the number average molecular weight exceeds 50,000, the solvent solubility of the resin composition may be lowered.
- the lower limit of the reduced viscosity of the resin composition of the present invention is preferably 0.6 dl / g or more, more preferably 0.7 dl / g or more, and further preferably 0.8 dl / g or more.
- the upper limit of the reduced viscosity is not particularly limited, but is preferably 1.5 dl / g or less, more preferably 1.4 dl / g or less, and further preferably 1.3 dl / g or less. If the reduced viscosity is less than 0.6 dl / g, the elasticity may be insufficient, and if the reduced viscosity exceeds 1.5 dl / g, the solvent solubility of the resin composition may be lowered.
- the resin composition of the present invention exhibits good solubility in a solvent.
- the solvent include aromatic hydrocarbons, aliphatic hydrocarbons, alicyclic hydrocarbons, ketone solvents, ether solvents, and aprotic polar solvents.
- acetone methyl ethyl ketone (hereinafter, also referred to as “MEK”), toluene, xylene, hexane, heptane, cyclohexane, methylcyclohexane, tetrahydrofuran, N-methylpyrrolidone, dimethylformamide, dimethylacetamide, dimethylsulfoxide, Solvesso 100 , Solvesso 150, Solvesso 200 and the like, but are not limited thereto.
- solvents can be used alone or in combination of two or more.
- the solubility in these solvents is preferably 20% by weight or more at 25 ° C., more preferably 25% by weight or more, and further preferably 30% by weight or more. Since the solvent solubility is excellent, the processability when an adhesive composition is obtained is improved.
- the resin composition of the present invention is stable enough to maintain fluidity even after being stored at 25 ° C. for 8 hours after being dissolved in the solvent so as to be 30% by weight.
- the resin composition has good adhesive properties at room temperature (about 20 ° C.) and 80 ° C.
- the carboxyl group concentration of the resin composition of the present invention is preferably 5 to 60 equivalent / t, more preferably 10 to 55 equivalent / t, and further preferably 15 to 50 equivalent / t. When it is less than 5 equivalents / t, the adhesiveness is lowered, and when it exceeds 60 equivalents / t, the heat resistance may be lowered.
- a known method can be used. For example, an esterification reaction of a crystalline polyether segment (a) and an amorphous polyester segment (b) at 150 to 250 ° C. Thereafter, a polycondensation reaction is carried out at 230 to 300 ° C. under reduced pressure to obtain a solvent-soluble elastic resin composition.
- a metal catalyst Ti atom is preferable from a polymeric viewpoint, and specifically, tetrabutyl titanate is especially preferable from a polymeric viewpoint.
- the amount of the catalyst is not particularly limited, but is preferably 200 ppm to 1000 ppm. If it exceeds 1000 ppm, the environmental resistance may deteriorate.
- the resin composition of the present invention may be copolymerized with a tri- or higher functional polycarboxylic acid such as trimellitic anhydride or trimethylolpropane or a polyol, if necessary.
- a tri- or higher functional polycarboxylic acid such as trimellitic anhydride or trimethylolpropane or a polyol, if necessary.
- Examples of the method for determining the composition and composition ratio of the resin composition include 1 H-NMR and 13 C-NMR in which a polyester resin is dissolved in a solvent such as deuterated chloroform, and gas chromatography that is measured after methanolysis of the polyester resin. (Hereinafter sometimes abbreviated as methanolysis-GC method).
- the composition and composition ratio are determined by 1 H-NMR.
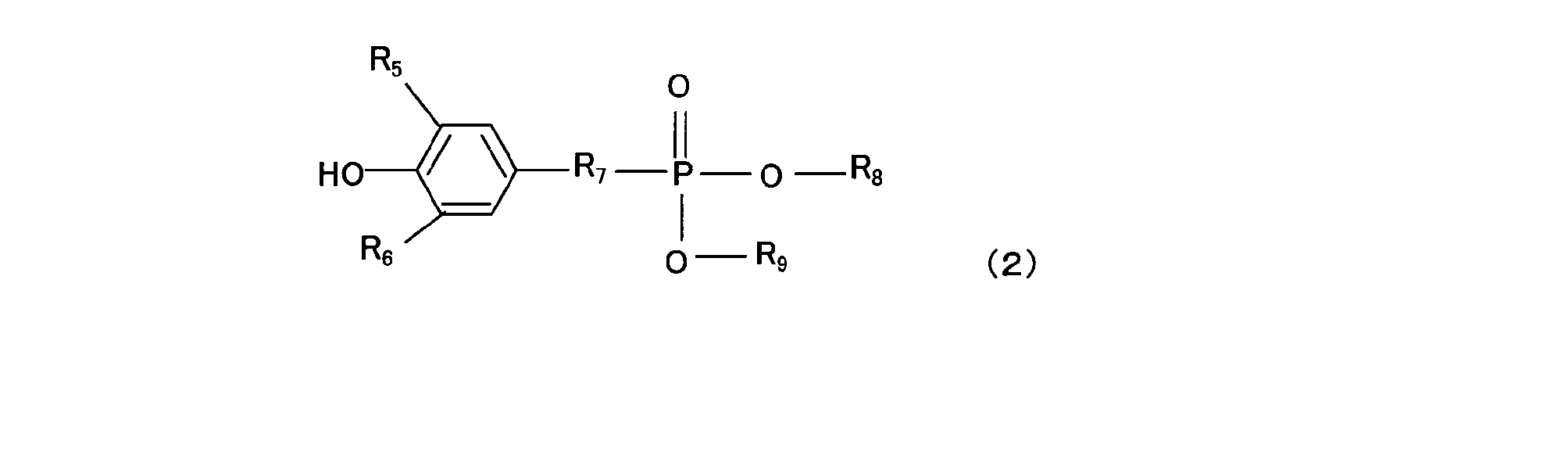
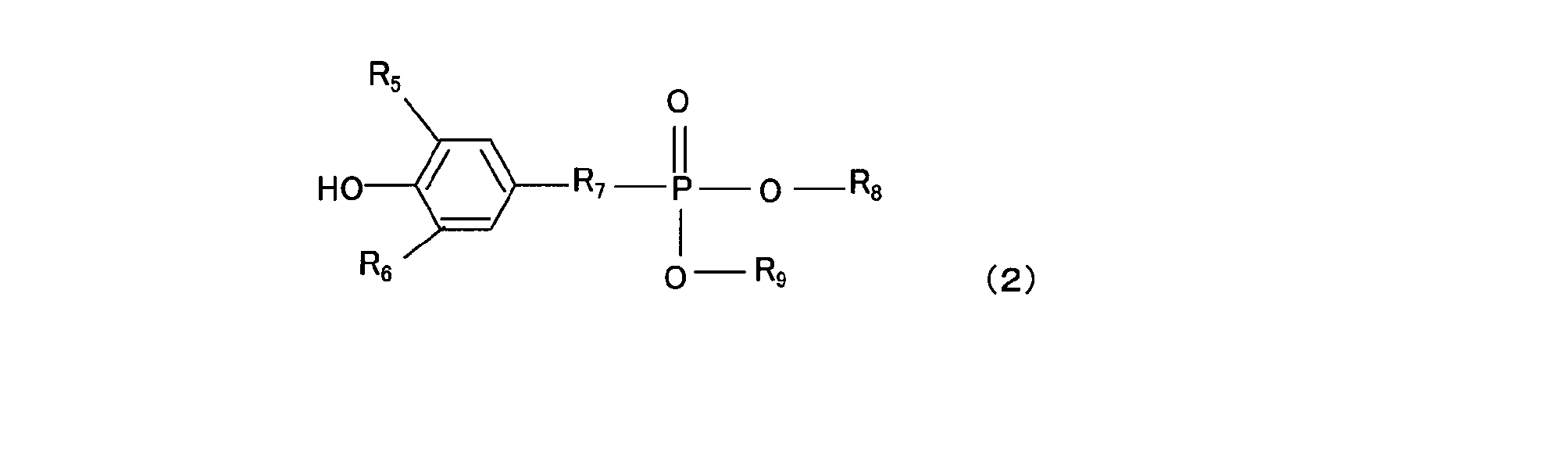
- the antioxidant (c) (hereinafter also simply referred to as “antioxidant (c)”) containing a phenolic O atom in the molecule used in the resin composition of the present invention is an antioxidant having phenol as a basic skeleton. Preferably there is. Specific examples include hindered phenol compounds represented by the following general formula (1) and the following general formula (2).
- the antioxidant (c) has a function of preventing the crystalline polyether segment (a) from being decomposed by being depolymerized by the catalyst Ti atom and decreasing in elasticity. Therefore, it is particularly effective to use it during the polymerization of the resin composition. Furthermore, it also has an effect of preventing the adhesive force from being reduced due to a decrease in molecular weight when the resin composition is used for a long time in a high temperature environment.
- the content of the antioxidant (c) is preferably 0.05 to 5 parts by weight, more preferably 0.08 to 4.5 parts by weight, even more preferably 100 parts by weight of the resin composition. 0.1 to 4 parts by weight. If it is less than 0.05 part by weight, the antioxidant effect cannot be sufficiently exhibited, and if it exceeds 5 parts by weight, it may bleed out from the resin and adversely affect the adhesion.
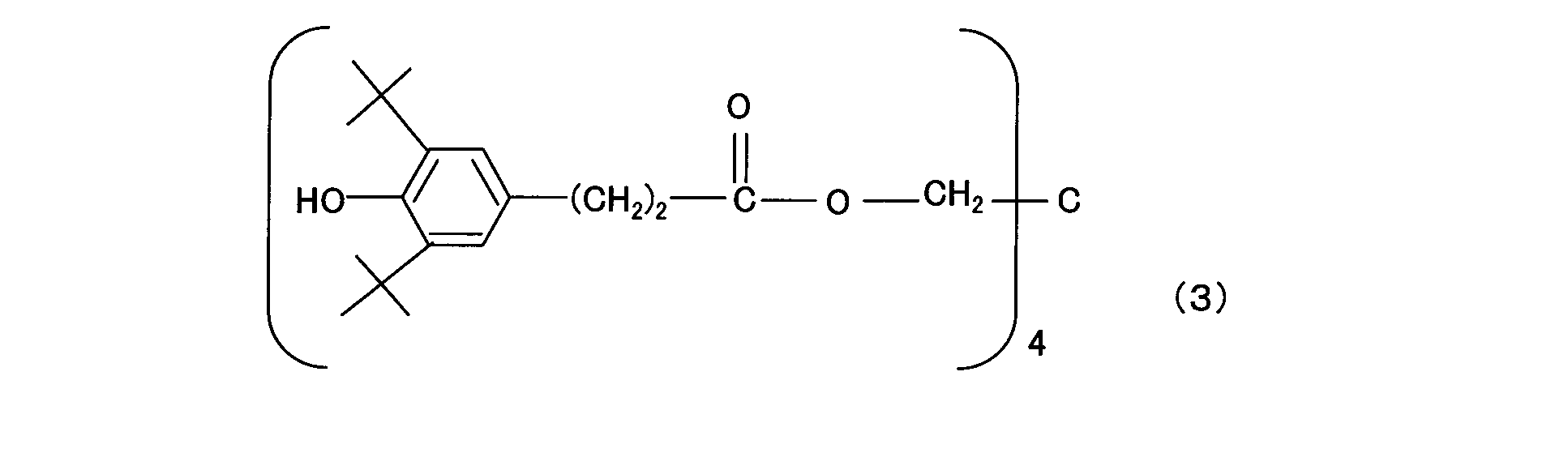
- the component (c1) is preferably a hindered phenol compound represented by the general formula (1).
- General formula (1); R 1 and R 2 are each independently an alkyl group which may be linear or branched, or hydrogen, and the alkyl group preferably has 1 to 4 carbon atoms, more preferably 2 to 3 carbon atoms.
- R 3 is an alkylene group, preferably having 1 to 4 carbon atoms, more preferably 2 to 3 carbon atoms.
- R 4 is an alkyl group which may be linear or branched, and preferably has 5 to 10 carbon atoms, more preferably 6 to 9 carbon atoms, and even more preferably 7 to 8 carbon atoms.
- n is an integer of 1 to 4.
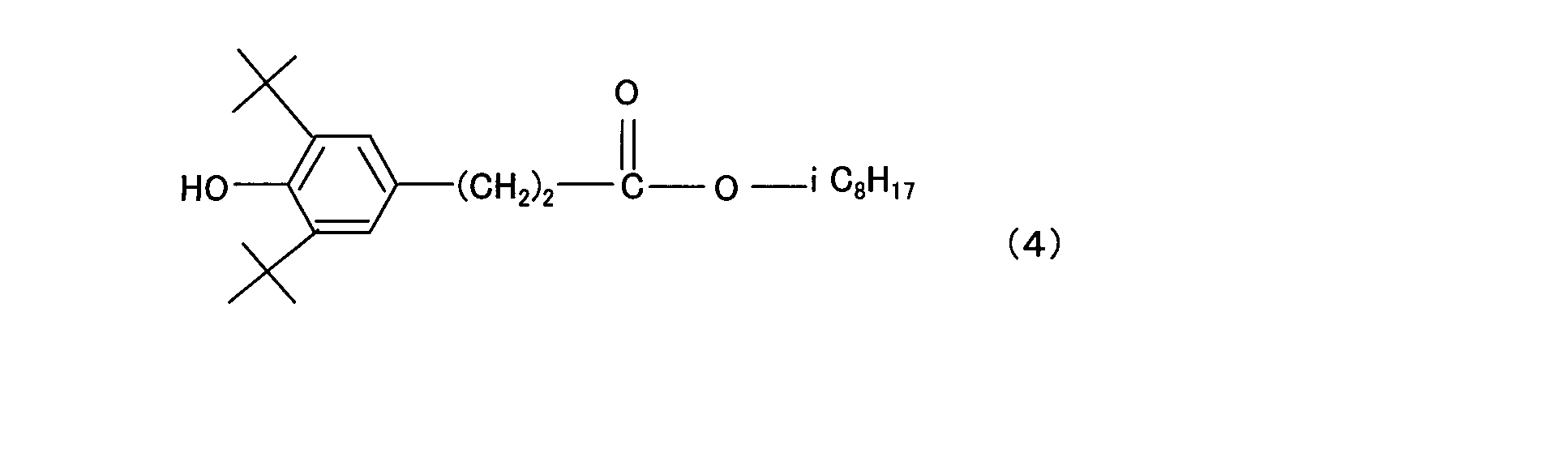
- the component (c1) is thiodiethylene bis [3- (3,5-di-tert-butyl 4-hydroxyphenyl) propionate], octadecyl [3- (3,5-di-tert-butyl 4-hydroxy Phenyl) propionate], N, N′-hexane-1,6-diylbis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionamide], 1,3,5-trimethyl-2, 4,6-tris (3,5-di-tert-butyl-4-hydroxy) benzene, 1,6-hexanediol-bis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate ], Triethylene glycol-bis-3- (3-tert-butyl-4-hydroxy-5-methylphenyl) propionate, 3,3′-thiobi Propionic acid dioctadecyl ester, 2,5,7,2,5
- the component (c2) is preferably a hindered phenol compound represented by the general formula (2).
- General formula (2); R 5 and R 6 are each independently an alkyl group which may have a straight chain or a branch, or hydrogen, and the alkyl group preferably has 1 to 4 carbon atoms, more preferably 2 to 3 carbon atoms.
- R 7 is an alkylene group, preferably having 1 to 4 carbon atoms, more preferably 2 to 3 carbon atoms.
- R 8 and R 9 are each independently an alkyl group which may be linear or branched, and preferably has 5 to 10 carbon atoms, more preferably 6 to 9 carbon atoms, still more preferably 7 to 8 carbon atoms. is there.
- the component (c2) is tributyl phosphate, tris (2,4-di-butylphenyl) phosphite, distearyl pentaerythritol diphosphite, cyclic neopentanetetraylbis (2,6-di-).
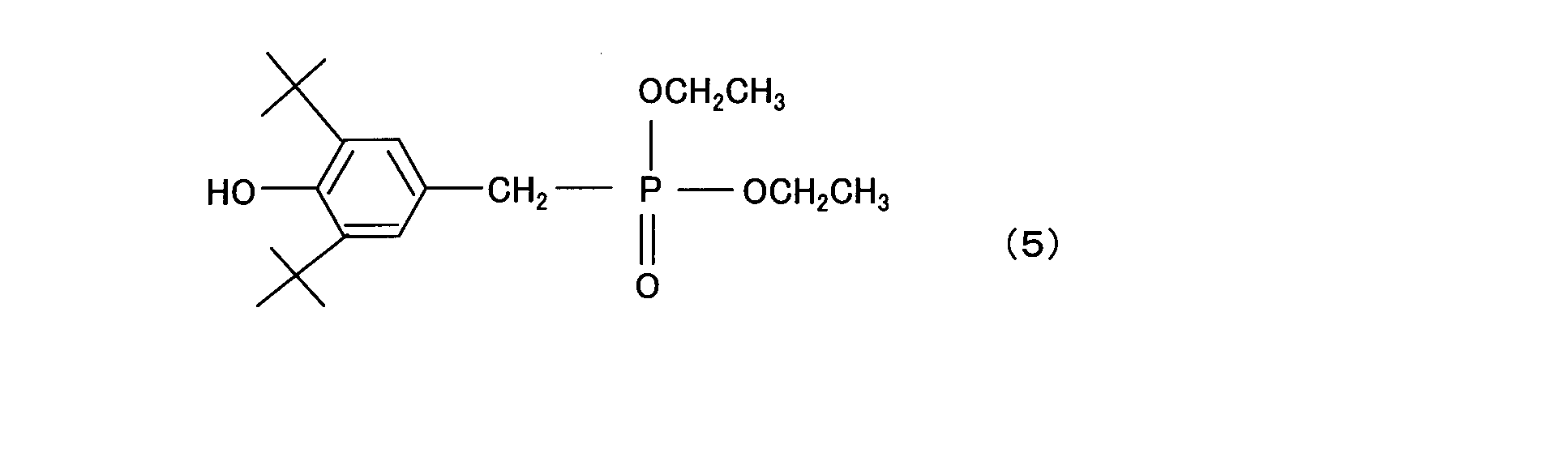
- tert-butyl-4-methylphenyl) phosphite trisnonylphenyl phosphite, tris (2,4-di-tert-butylphenyl) phosphite, distearyl pentaerythritol diphosphite, bis (2,4-di-) tert-butylphenyl) pentaerythritol phosphite, bis (2,6-di-tert-butyl-4-methyl-phenyl) pentaerythritol phosphite, 2,2-methylene-bis (4,6-di-tert-butyl) Phenyl) octyl phosphite, tetrakis ( 2,4-di-tert-butylphenyl) -4,4′-biphenylene-diphosphonite, tetrakis (2,4-di-tert-butylpheny
- antioxidants (c1) Only one or more of the antioxidants (c1) can be used, and only one or more of the antioxidants (c2) can be used. Furthermore, antioxidant (c1) and antioxidant (c2) can also be used together.
- the adhesive composition of this invention is a composition which contains the said resin composition and has a resin composition as a main component.
- the main component means that the resin composition contains 50% by weight or more in the adhesive composition, preferably 60% by weight or more, and more preferably 70% by weight or more. If the amount is too small, good adhesiveness and adhesive heat resistance may not be exhibited. As other components, the components described later can be blended.
- components other than the resin composition can be blended for the purpose of improving adhesion, flexibility, durability and the like within a range not impairing the effects of the present invention.
- Specific examples include, but are not limited to, polyesters, polyamides, polyolefins, epoxies, polycarbonates, acrylics, ethylene vinyl acetate, phenols and other resins, isocyanate compounds and curing agents such as melamine, fillers such as talc and mica, carbon black, Examples thereof include pigments such as titanium oxide, antimony trioxide, and brominated polystyrene.
- the content of these components is preferably 50% by weight or less in the adhesive composition, more preferably 40% by weight or less, and still more preferably 30% by weight or less. If the content exceeds 50% by weight, adhesion to electric / electronic parts and adhesion heat resistance may be reduced.
- compositions and composition ratio of the polyester were determined by 1 H-NMR measurement (proton nuclear magnetic resonance spectroscopy) at a resonance frequency of 400 MHz.
- the measuring apparatus was an NMR apparatus 400-MR manufactured by VARIAN, and deuterated chloroform was used as a solvent.
- ⁇ Storage modulus> A sample of the resin composition was placed on a heat press adjusted to 200 ° C. via a polyimide film (“Kapton (registered trademark)” manufactured by Toray DuPont) and held at a pressure of 2 N / mm 2 for 20 seconds to hold 1 mm. A sheet sample of thickness was obtained. This was cut into a length of 15 mm (excluding the gripping allowance) and a width of 4 mm, and a sample was set in a dynamic viscoelasticity measuring device “DVA-200” manufactured by IT Measurement Control Co., Ltd., and measured in a tensile mode. The measurement condition was a scan from ⁇ 100 ° C. until measurement became impossible (maximum 250 ° C.) at a heating rate of 4 ° C./min with the frequency fixed at 10 Hz.
- DVA-200 dynamic viscoelasticity measuring device
- ⁇ Adhesion test> The sample whose solution stability test was “ ⁇ ” was applied to a PET film, and then dried at 120 ° C. for 7 minutes to prepare a coating film having a thickness of 60 ⁇ m. The coating film was adhered to a tin-plated copper foil whose surface was washed well at 160 ° C. and 3 MPa to prepare an adhesion sample A. At room temperature (about 25 ° C.), the adhesive sample A was peeled off at a rate of 100 mm / min, and the adhesive strength was measured.
- Adhesion strength 25 N / 25 mm or more Adhesion strength 20 N / 25 mm or more and less than 25 N / 25 mm ⁇ : Adhesion strength 15 N / 25 mm or more and less than 20 N / 25 mm X: Adhesion strength 15 N / 25 mm or less
- Adhesion heat resistance test The adhesive sample A was kept at 80 ° C. for 5 minutes, peeled T-shaped at a speed of 100 mm / min in an environment of 80 ° C., and the adhesive strength was measured.
- X Adhesion strength 15 N / 25 mm or less
- TPA terephthalic acid
- IPA isophthalic acid
- SA sebacic acid
- NDCA 2,6-naphthalenedicarboxylic acid
- EG ethylene glycol
- NPG neopentyl glycol
- BD 1,4-butanediol
- 2MG 2-methyl 1,3-propanediol
- PTMG1000 polytetramethylene ether glycol 1000
- PEG2000 polyethylene glycol 2000
- PTXG1000 poly (tetramethylene ether glycol-neopentyl glycol) 1000
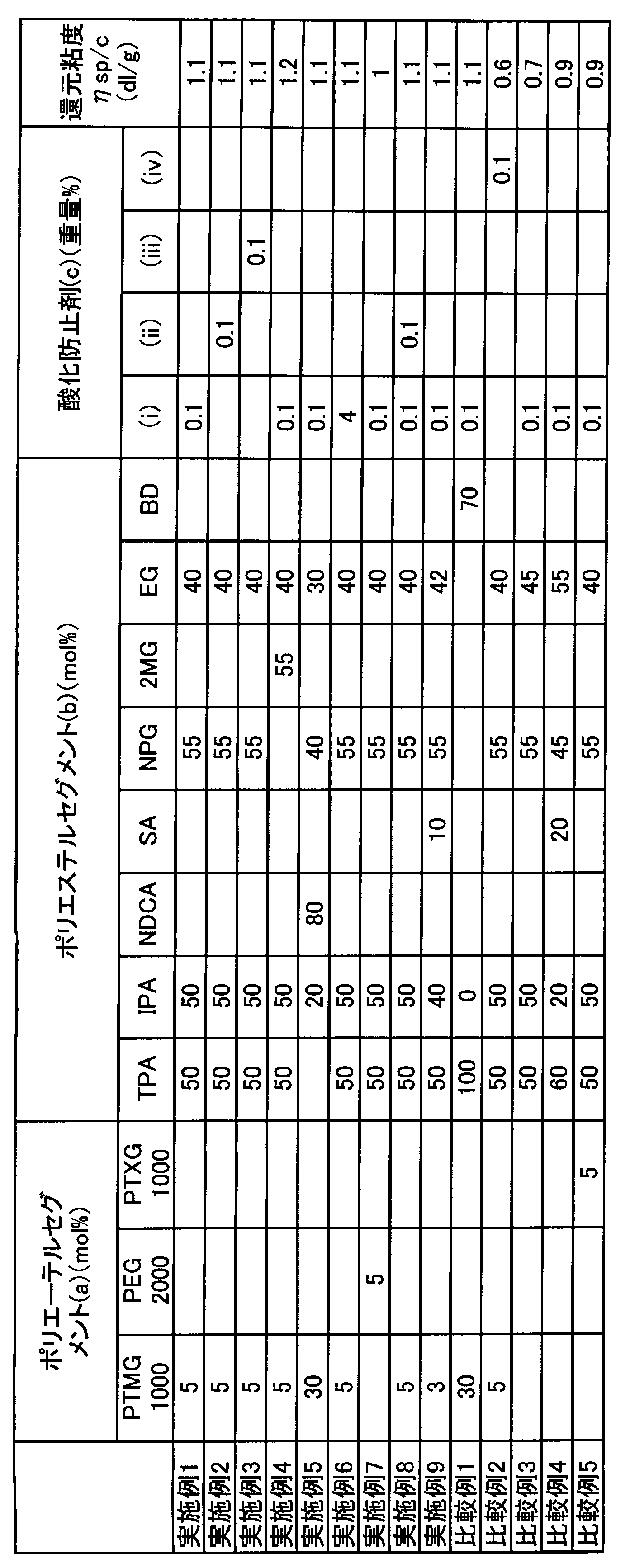
- Example 1 The resin composition of Example 1 was subjected to ⁇ solvent solubility test>, ⁇ solution stability test>, ⁇ adhesion test>, and ⁇ adhesion heat resistance test>.
- Comparative Examples 1-5 The same test as in Example 1 was performed using Comparative Examples 1-5. The evaluation results are shown in Table 2.
- Examples 1 to 9 satisfied the claims, and all the results of ⁇ solvent solubility test>, ⁇ solution stability test>, ⁇ adhesion test>, and ⁇ adhesion heat resistance test> were satisfactory.
- the component (b) is a crystalline polyester
- the comparative example 1 does not dissolve in the solvent and the ⁇ solvent solubility test> is poor.
- the comparative example 2 does not use the antioxidant which contains a phenolic O atom in a molecule
- Comparative Example 3 does not contain the component (a), and the ⁇ adhesion test> is poor.
- Comparative Example 4 does not contain the component (a), and since the component (b) is a crystalline polyester, it does not dissolve in the solvent, and the ⁇ solvent solubility test> is poor.
- Comparative Example 5 since the component (a) is an amorphous polyether, it was dissolved before reaching 80 ° C. during storage elastic modulus measurement, and the storage elastic modulus at 80 ° C. could not be measured. Moreover, ⁇ adhesion heat resistance test> is poor.
- the resin composition of the present invention has solvent solubility, excellent handling properties, can maintain adhesive properties in a wide temperature range, and is excellent in durability against high temperature environmental loads and high temperature high humidity environmental loads. Accordingly, it is suitable for various applications such as automobiles, communications, computers, home appliances, flexible flat cables, electronic components, switches having a printed circuit board, and sensors.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Adhesives Or Adhesive Processes (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
Abstract
Description
本発明は、溶剤可溶性を有し幅広い温度範囲での接着特性を保持できる弾性樹脂組成物に関する。
The present invention relates to an elastic resin composition that is soluble in a solvent and can retain adhesive properties in a wide temperature range.
電気電子部品の封止に用いる材料は、その使用目的を達成する為に、外部との電気絶縁性が必須とされ、電気電子部品の形状に確実に追随し剥離部が発生しない事が必要とされる。加えて、自動車のステアリング部位に組み込まれた回路部分など、高熱環境下で繰り返し屈曲を受ける場合には、その使用目的を達成する為に、幅広い温度範囲での接着特性が必要とされる。 In order to achieve the purpose of use, the material used for sealing electrical and electronic parts must have electrical insulation from the outside, and it is necessary to follow the shape of the electrical and electronic parts reliably and not to cause peeling. Is done. In addition, when subjected to repeated bending in a high heat environment such as a circuit portion incorporated in a steering part of an automobile, adhesive characteristics in a wide temperature range are required to achieve the purpose of use.
電気絶縁性・耐水性が共に高いポリエステルはこの用途に非常に有用な材料と考えられるが、一般に接着剤として用いられる溶剤可溶性の非晶性樹脂は弾性が低く、繰り返し屈曲に耐えられない傾向にあった。これに対し、特許文献1には、ポリエーテルセグメントと結晶性ポリエステルセグメントからなる弾性樹脂組成物が開示されており、高い分子量を持つポリエーテルグリコールを共重合する事で、電気絶縁性などの条件を満たしつつ高い弾性の保持が可能であることが開示されている。この樹脂組成物により、良好な成型品が得られるようになり、一般電気電子部品へのポリエステル系樹脂組成物の適用が可能となった。 Polyester with high electrical insulation and water resistance is considered to be a very useful material for this application, but solvent-soluble amorphous resins generally used as adhesives have low elasticity and tend not to withstand repeated bending. there were. On the other hand, Patent Document 1 discloses an elastic resin composition composed of a polyether segment and a crystalline polyester segment. By copolymerizing a polyether glycol having a high molecular weight, conditions such as electrical insulation are disclosed. It is disclosed that high elasticity can be maintained while satisfying the above. With this resin composition, a good molded product can be obtained, and the polyester resin composition can be applied to general electric and electronic parts.
しかしながら、特許文献1に開示されている樹脂組成物は、結晶性であるために溶剤溶解後の安定性が悪く、加工方法が限定的であった。本発明の課題は、上記の問題を解決することであり、良好な溶剤可溶性を有し、かつ幅広い温度範囲での接着特性を保持できる弾性樹脂組成物(以下、単に「樹脂組成物」ともいう。)を提供することにある。
However, since the resin composition disclosed in Patent Document 1 is crystalline, the stability after dissolving the solvent is poor, and the processing method is limited. An object of the present invention is to solve the above-mentioned problems, and is an elastic resin composition (hereinafter simply referred to as “resin composition”) that has good solvent solubility and can maintain adhesive properties in a wide temperature range. .) To provide.
上記目的を達成する為、本発明者等は鋭意検討し、以下の発明を提案するに至った。即ち本発明は、以下に示す溶剤可溶性を有し、幅広い温度範囲での接着特性を保持できる弾性樹脂組成物である。 In order to achieve the above object, the present inventors have intensively studied and have come up with the following inventions. That is, the present invention is an elastic resin composition having solvent solubility shown below and capable of maintaining adhesive properties in a wide temperature range.
結晶性ポリエーテルセグメント(a)と非晶性ポリエステルセグメント(b)を含有する溶剤可溶性弾性樹脂組成物であって、
周波数10Hzで測定した際の
20℃での貯蔵弾性率(E’20)が800~2000MPaであり、
80℃での貯蔵弾性率(E’80)が0.5~2.5MPaであり、
かつ20℃での貯蔵弾性率に対する80℃での貯蔵弾性率の割合(E’80/E’20)が0.025%~0.25%の範囲にあることを特徴とする溶剤可溶性弾性樹脂組成物。
A solvent-soluble elastic resin composition containing a crystalline polyether segment (a) and an amorphous polyester segment (b),
The storage elastic modulus (E'20) at 20 ° C. when measured at a frequency of 10 Hz is 800 to 2000 MPa,
The storage elastic modulus (E'80) at 80 ° C. is 0.5 to 2.5 MPa,
And the ratio of the storage elastic modulus at 80 ° C. to the storage elastic modulus at 20 ° C. (E′80 / E′20) is in the range of 0.025% to 0.25%. Composition.
さらに、前記溶剤可溶性弾性樹脂組成物100重量部に対して、フェノール性O原子を分子中に含む酸化防止剤(c)を0.05~5重量部含有することが好ましい。 Furthermore, it is preferable to contain 0.05 to 5 parts by weight of an antioxidant (c) containing phenolic O atoms in the molecule with respect to 100 parts by weight of the solvent-soluble elastic resin composition.
また、前記フェノール性O原子を分子中に含む酸化防止剤(c)が、一般式(1)で表される化合物(c1)および/または一般式(2)で表される化合物(c2)であることが好ましい。
一般式(1);
一般式(2);
General formula (1);
General formula (2);
前記のいずれかに記載の溶剤可溶性弾性樹脂組成物を含有する接着剤組成物。 An adhesive composition containing the solvent-soluble elastic resin composition according to any one of the above.
本発明に係る溶剤可溶性弾性樹脂組成物(以下、単に「樹脂組成物」ともいう。)は良好な溶剤可溶性を有し、優れた溶液安定性かつ室温(約20℃)から80℃までの幅広い温度範囲での接着性、接着耐熱性を保持できる。 The solvent-soluble elastic resin composition (hereinafter also simply referred to as “resin composition”) according to the present invention has good solvent solubility, excellent solution stability, and a wide range from room temperature (about 20 ° C.) to 80 ° C. Maintains adhesiveness and adhesive heat resistance in the temperature range.
以下、本発明を詳細に説明する。 Hereinafter, the present invention will be described in detail.
<結晶性ポリエーテルセグメント(a)>
本発明に用いる結晶性ポリエーテルセグメント(a)は、特に限定されないが、主として結晶性のポリアルキレングリコールであることが好ましい。結晶性であることで耐熱性の面で好ましい。具体的にはポリエチレングリコール(以下、PEGともいう。)、ポリトリメチレングリコール(以下、PPGともいう。)、ポリテトラメチレングリコール(以下、PTMGともいう。)等を挙げることができるが、これらに限定されない。この中でも、耐熱性の観点からPTMGが好ましい。
<Crystalline polyether segment (a)>
The crystalline polyether segment (a) used in the present invention is not particularly limited, but is preferably mainly crystalline polyalkylene glycol. It is preferable in terms of heat resistance because it is crystalline. Specific examples include polyethylene glycol (hereinafter also referred to as PEG), polytrimethylene glycol (hereinafter also referred to as PPG), polytetramethylene glycol (hereinafter also referred to as PTMG), and the like. It is not limited. Among these, PTMG is preferable from the viewpoint of heat resistance.
結晶性ポリエーテルセグメント(a)の数平均分子量は230以上である事が好ましく、400以上であることがより好ましく、800以上であることがさらに好ましい。結晶性ポリエーテルセグメント(a)の数平均分子量が230未満であると、樹脂組成物に柔軟性を付与出来ず、耐屈曲性が発現できないなどの問題を生じる傾向にある。また結晶性ポリエーテルセグメント(a)の数平均分子量は5000以下であることが好ましく、4000以下であることがより好ましく、3000以下であることがさらに好ましい。数平均分子量が5000を超えると他の共重合成分との相溶性が悪く共重合できない等の問題を生じる傾向にある。 The number average molecular weight of the crystalline polyether segment (a) is preferably 230 or more, more preferably 400 or more, and further preferably 800 or more. When the number average molecular weight of the crystalline polyether segment (a) is less than 230, there is a tendency that the resin composition cannot be given flexibility and problems such as inflexibility cannot be exhibited. The number average molecular weight of the crystalline polyether segment (a) is preferably 5000 or less, more preferably 4000 or less, and further preferably 3000 or less. When the number average molecular weight exceeds 5,000, the compatibility with other copolymerization components is poor, and problems such as inability to copolymerize tend to occur.
本発明で結晶性とは、示差走査型熱量計(DSC)を用いて、-100℃~300℃まで20℃/分で昇温し、該昇温過程に明確な融解ピークを示すものをいう。 In the present invention, the term “crystallinity” means that the temperature is raised from −100 ° C. to 300 ° C. at 20 ° C./min using a differential scanning calorimeter (DSC) and shows a clear melting peak in the temperature raising process. .
<非晶性ポリエステルセグメント(b)>
非晶性ポリエステルセグメント(b)は酸成分とグリコール成分を重合させたポリエステルであることが好ましく、非晶性であることで溶剤溶解性の面で好ましい。非晶性とは、示差走査型熱量計(DSC)を用いて、-100℃~300℃まで20℃/分で昇温し、該昇温過程に明確な融解ピークを示さないものをいう。
<Amorphous polyester segment (b)>
The amorphous polyester segment (b) is preferably a polyester obtained by polymerizing an acid component and a glycol component, and is preferably amorphous in terms of solvent solubility. Amorphous means that the temperature is raised from −100 ° C. to 300 ° C. at 20 ° C./min using a differential scanning calorimeter (DSC) and does not show a clear melting peak in the temperature raising process.
本発明に用いる非晶性ポリエステルセグメント(b)を構成する酸成分は特に限定されないが、芳香族ジカルボン酸が好ましい。芳香族ジカルボン酸の炭素数は8~14が好ましく、9~13がより好ましい。芳香族ジカルボン酸は、樹脂組成物の全酸成分を100モル%としたとき、50モル%以上含むことが樹脂組成物の弾性を向上させるため好ましく、60モル%以上であるとより好ましく、80モル%以上がさらに好ましく、95モル%以上が特に好ましく、100モル%であっても差し支えない。芳香族ジカルボン酸の具体例としては、特に限定されないが、テレフタル酸、イソフタル酸およびナフタレンジカルボン酸からなる群より選ばれる1以上の酸であることがグリコール成分との反応性が良好であり、重合性および生産性の点で望ましい。テレフタル酸、イソフタル酸およびナフタレンジカルボン酸の合計が、樹脂組成物の全酸成分中50モル%以上であることが好ましく、60モル%以上であることがより好ましく、80モル%以上であることがより好ましく、95モル%以上であることが更に好ましく、全酸成分がテレフタル酸、イソフタル酸およびナフタレンジカルボン酸からなる群より選ばれる1以上の酸で構成されていても差し支えない。ナフタレンジカルボン酸であれば、1,4-ナフタレンジカルボン酸、1,8-ナフタレンジカルボン酸、2,3-ナフタレンジカルボン酸、2,6-ナフタレンジカルボン酸、2,7-ナフタレンジカルボン酸のいずれでも良いが、中でも2,6-ナフタレンジカルボン酸が好ましい。 The acid component constituting the amorphous polyester segment (b) used in the present invention is not particularly limited, but an aromatic dicarboxylic acid is preferable. The aromatic dicarboxylic acid preferably has 8 to 14 carbon atoms, more preferably 9 to 13 carbon atoms. The aromatic dicarboxylic acid is preferably contained in an amount of 50 mol% or more when the total acid component of the resin composition is 100 mol% in order to improve the elasticity of the resin composition, more preferably 60 mol% or more. It is more preferably at least mol%, particularly preferably at least 95 mol%, and may be 100 mol%. Specific examples of the aromatic dicarboxylic acid include, but are not particularly limited to, one or more acids selected from the group consisting of terephthalic acid, isophthalic acid, and naphthalenedicarboxylic acid, which have good reactivity with the glycol component, and polymerization. In terms of productivity and productivity. The total of terephthalic acid, isophthalic acid and naphthalenedicarboxylic acid is preferably 50 mol% or more, more preferably 60 mol% or more, and more preferably 80 mol% or more in the total acid component of the resin composition. More preferably, it is 95 mol% or more, and the total acid component may be composed of one or more acids selected from the group consisting of terephthalic acid, isophthalic acid and naphthalenedicarboxylic acid. As long as it is naphthalenedicarboxylic acid, any of 1,4-naphthalenedicarboxylic acid, 1,8-naphthalenedicarboxylic acid, 2,3-naphthalenedicarboxylic acid, 2,6-naphthalenedicarboxylic acid, and 2,7-naphthalenedicarboxylic acid may be used. Of these, 2,6-naphthalenedicarboxylic acid is preferred.
非晶性ポリエステルセグメント(b)を構成するその他の酸成分としては、ジフェニルジカルボン酸、5-ナトリウムスルイソフタル酸などの芳香族ジカルボン酸、シクロヘキサンジカルボン酸、テトラヒドロ無水フタル酸などの脂環族ジカルボン酸、コハク酸、グルタル酸、アジピン酸、アゼライン酸、セバシン酸、ドデカン二酸、ダイマー酸、水添ダイマー酸などの脂肪族ジカルボン酸などのジカルボン酸が挙げられる。 これらジカルボン酸成分の共重合比率は樹脂組成物の全酸成分を100モル%としたとき、50モル%未満が好ましく、40モル%未満がより好ましく、20モル%未満がさらに好ましく、5モル%未満が特に好ましく、0モル%であっても差し支えない。また、非晶性ポリエステルセグメント(b)を構成するその他の酸成分として、トリメリット酸、ピロメリット酸等の三官能以上のポリカルボン酸を用いることも可能である。3官能以上のポリカルボン酸の共重合比率は、樹脂組成物のゲル化防止の観点から10モル%以下とすることが好ましく、5モル%以下とすることがより好ましい。 Other acid components constituting the amorphous polyester segment (b) include diphenyl dicarboxylic acid, aromatic dicarboxylic acid such as 5-sodium sulfisophthalic acid, cyclohexane dicarboxylic acid, and alicyclic dicarboxylic acid such as tetrahydrophthalic anhydride. And dicarboxylic acids such as aliphatic dicarboxylic acids such as succinic acid, glutaric acid, adipic acid, azelaic acid, sebacic acid, dodecanedioic acid, dimer acid and hydrogenated dimer acid. The copolymerization ratio of these dicarboxylic acid components is preferably less than 50 mol%, more preferably less than 40 mol%, still more preferably less than 20 mol%, and more preferably 5 mol% when the total acid component of the resin composition is 100 mol%. Less than is particularly preferable, and it may be 0 mol%. Further, as the other acid component constituting the amorphous polyester segment (b), it is possible to use a tri- or higher functional polycarboxylic acid such as trimellitic acid or pyromellitic acid. The copolymerization ratio of the trifunctional or higher polycarboxylic acid is preferably 10 mol% or less, and more preferably 5 mol% or less from the viewpoint of preventing gelation of the resin composition.
本発明に用いる非晶性ポリエステルセグメント(b)を構成するグリコール成分は、特に限定されないが、脂肪族グリコールおよび/または脂環族グリコールが好ましく、なかでも分岐型脂肪族グリコールおよび/または分岐型脂環族グリコールがより好ましい。分岐型脂肪族グリコールおよび/または分岐型脂環族グリコールの好ましい炭素数は4以上であり、より好ましくは5以上であり、さらに好ましくは6以上であり、好ましくは20以下であり、より好ましくは15以下であり、さらに好ましくは10以下であり、特に好ましくは8以下である。前記グリコール成分は、樹脂組成物の全グリコール成分を100モル%としたとき、20モル%以上含むことが好ましく、25モル%以上がより好ましく、30モル%以上がさらに好ましく、40モル%以上が特に好ましい。また、99モル%以下であることが好ましく、97モル%以下がより好ましく、95モル%以下がさらに好ましい。20モル%未満であると、溶剤溶解性が不十分となることがあり、99モル%を超えると、結晶性ポリエーテルセグメント(a)量が不十分となることから20℃での貯蔵弾性率(E’20)が2000MPa超となり、貯蔵弾性が高すぎて接着性が低下することがある。 The glycol component constituting the amorphous polyester segment (b) used in the present invention is not particularly limited, but is preferably an aliphatic glycol and / or an alicyclic glycol, and in particular, a branched aliphatic glycol and / or a branched fat. A cyclic glycol is more preferred. The number of carbon atoms of the branched aliphatic glycol and / or the branched alicyclic glycol is 4 or more, more preferably 5 or more, still more preferably 6 or more, preferably 20 or less, more preferably It is 15 or less, more preferably 10 or less, and particularly preferably 8 or less. The glycol component is preferably 20 mol% or more, more preferably 25 mol% or more, still more preferably 30 mol% or more, and more preferably 40 mol% or more when the total glycol component of the resin composition is 100 mol%. Particularly preferred. Moreover, it is preferable that it is 99 mol% or less, 97 mol% or less is more preferable, and 95 mol% or less is further more preferable. If it is less than 20 mol%, the solvent solubility may be insufficient, and if it exceeds 99 mol%, the amount of crystalline polyether segment (a) will be insufficient, so the storage elastic modulus at 20 ° C. (E'20) exceeds 2000 MPa, and the storage elasticity may be too high, resulting in a decrease in adhesion.
ここで、分岐型脂肪族グリコールとは、直鎖状炭化水素に炭素数1以上の側鎖基を有する脂肪族グリコールであり、分岐型脂環族グリコールとは、脂環式炭化水素の環状炭素に炭素数1以上の側鎖基を有する脂環族グリコールである。具体的には、特に限定されないが、分岐型脂肪族グリコールとしては、ネオペンチルグリコール、2-メチル-1,3-プロパンジオール、1,2-ブタンジオール、1,3-ブタンジオール、2,3-ブタンジオール、3-メチル-1,5-ペンタンジオール、2,2,4-トリメチル-1,3-ペンタンジオール、2-エチル-1,3-ヘキサンジオール、2,2-ジメチル-3-ヒドロキシプロピル-2’,2’-ジメチル-3-ヒドロキシプロパネート、2-ノルマルブチル-2-エチル-1,3-プロパンジオール、3-エチル-1,5-ペンタンジオール、3-プロピル-1,5-ペンタンジオール、2,2-ジエチル-1,3-プロパンジオール、2-ブチル-2-エチル-1,3-プロパンジオール、3-オクチル-1,5-ペンタンジオール、3-フェニル-1,5-ペンタンジオール、2,5-ジメチル-3-ナトリウムスルホ-2,5-ヘキサンジオール等が挙げられ、分岐型脂環族グリコールとしては、1,3-ビス(ヒドロキシメチル)シクロヘキサン、1,4-ビス(ヒドロキシメチル)シクロヘキサン、1,4-ビス(ヒドロキシエチル)シクロヘキサン、1,4-ビス(ヒドロキシプロピル)シクロヘキサン、1,4-ビス(ヒドロキシメトキシ)シクロヘキサン、1,4-ビス(ヒドロキシエトキシ)シクロヘキサン、2,2-ビス(4-ヒドロキシメトキシシクロヘキシル)プロパン、2,2-ビス(4-ヒドロキシエトキシシクロヘキシル)プロパン、ビス(4-ヒドロキシシクロヘキシル)メタン、2,2-ビス(4-ヒドロキシシクロヘキシル)プロパン、3(4),8(9)-トリシクロ[5.2.1.02,6]デカンジメタノール、水素化ビスフェノールA、水素化ビスフェノールAのエチレンオキサイド付加物、水素化ビスフェノールAのプロピレンオキサイド付加物等が挙げられ、これらを1種または2種以上併用して使用することができる。この中で、ネオペンチルグリコールが組成物の溶剤溶解性を向上させる点で最も好ましい。 Here, the branched aliphatic glycol is an aliphatic glycol having a side chain group having 1 or more carbon atoms in a linear hydrocarbon, and the branched alicyclic glycol is a cyclic carbon of an alicyclic hydrocarbon. Is an alicyclic glycol having a side chain group having 1 or more carbon atoms. Specifically, although not particularly limited, examples of the branched aliphatic glycol include neopentyl glycol, 2-methyl-1,3-propanediol, 1,2-butanediol, 1,3-butanediol, 2,3 -Butanediol, 3-methyl-1,5-pentanediol, 2,2,4-trimethyl-1,3-pentanediol, 2-ethyl-1,3-hexanediol, 2,2-dimethyl-3-hydroxy Propyl-2 ′, 2′-dimethyl-3-hydroxypropanoate, 2-normalbutyl-2-ethyl-1,3-propanediol, 3-ethyl-1,5-pentanediol, 3-propyl-1,5 -Pentanediol, 2,2-diethyl-1,3-propanediol, 2-butyl-2-ethyl-1,3-propanediol, 3-octyl-1,5- Nthanediol, 3-phenyl-1,5-pentanediol, 2,5-dimethyl-3-sodium sulfo-2,5-hexanediol and the like. Examples of branched alicyclic glycols include 1,3-bis (Hydroxymethyl) cyclohexane, 1,4-bis (hydroxymethyl) cyclohexane, 1,4-bis (hydroxyethyl) cyclohexane, 1,4-bis (hydroxypropyl) cyclohexane, 1,4-bis (hydroxymethoxy) cyclohexane, 1,4-bis (hydroxyethoxy) cyclohexane, 2,2-bis (4-hydroxymethoxycyclohexyl) propane, 2,2-bis (4-hydroxyethoxycyclohexyl) propane, bis (4-hydroxycyclohexyl) methane, 2, 2-bis (4-hydroxycyclohex Sil) propane, 3 (4), 8 (9) -tricyclo [5.2.1.0 2 , 6 ] decandimethanol, hydrogenated bisphenol A, ethylene oxide adduct of hydrogenated bisphenol A, hydrogenated bisphenol A These propylene oxide adducts can be used, and these can be used alone or in combination of two or more. Among these, neopentyl glycol is most preferable in terms of improving the solvent solubility of the composition.
炭素数3以下の短鎖グリコールの含有量は、樹脂組成物の全グリコール成分を100モル%としたとき、70モル%以下が好ましく、60モル%以下がより好ましく、50モル%以下がさらに好ましく、40モル%以下が特に好ましい。多すぎると結晶性ポリエーテルセグメント(a)との相溶性が悪くなり、重合性が悪化する傾向にある。炭素数3以下の短鎖グリコールの具体例としては、特に限定されないが、エチレングリコール、1,2-プロピレングリコール、または1,3-プロピレングリコールが挙げられる。 The content of the short-chain glycol having 3 or less carbon atoms is preferably 70 mol% or less, more preferably 60 mol% or less, and even more preferably 50 mol% or less, when the total glycol component of the resin composition is 100 mol%. 40 mol% or less is particularly preferable. If the amount is too large, the compatibility with the crystalline polyether segment (a) tends to be poor, and the polymerizability tends to deteriorate. Specific examples of the short-chain glycol having 3 or less carbon atoms include, but are not limited to, ethylene glycol, 1,2-propylene glycol, or 1,3-propylene glycol.
また、炭素数4以上の直鎖型グリコール成分も使用することができる。直鎖型グリコールの配合量は20モル%以下が好ましく、10モル%以下がより好ましく、5モル%以下がさらに好ましく、0モル%であっても差し支えない。直鎖型グリコールが多すぎると、ポリエステルセグメント(b)が結晶性となる傾向にあり、溶剤溶解性が低下することがある。 Also, linear glycol components having 4 or more carbon atoms can be used. The blending amount of the linear glycol is preferably 20 mol% or less, more preferably 10 mol% or less, further preferably 5 mol% or less, and may be 0 mol%. When there are too many linear glycols, there exists a tendency for a polyester segment (b) to become crystallinity, and solvent solubility may fall.
また、グリコール成分の一部として、グリセリン、トリメチロールプロパン、ペンタエリスルトール等の三官能以上のポリオールを用いても良く、樹脂組成物のゲル化防止の観点から10モル%以下とすることが好ましく、5モル%以下とすることがより好ましい。多すぎると樹脂組成物がゲル化することがある。 Further, as a part of the glycol component, a tri- or higher functional polyol such as glycerin, trimethylolpropane, pentaerythritol, etc. may be used, and the content is made 10 mol% or less from the viewpoint of preventing gelation of the resin composition. Preferably, it is more preferably 5 mol% or less. If the amount is too large, the resin composition may be gelled.
<溶剤可溶性弾性樹脂組成物>
本発明に係る溶剤可溶性弾性樹脂組成物は、前記結晶性ポリエーテルセグメント(a)と前記非晶性ポリエステルセグメント(b)を含有し、周波数10Hzで測定した際の20℃での貯蔵弾性率(E’20)が800~2000MPaであり、80℃での貯蔵弾性率(E’80)が0.5~2.5MPaであり、かつ20℃での貯蔵弾性率に対する80℃での貯蔵弾性率の割合(E’80/E’20)が0.025%~0.25%の範囲にある樹脂組成物である。
<Solvent-soluble elastic resin composition>
The solvent-soluble elastic resin composition according to the present invention contains the crystalline polyether segment (a) and the amorphous polyester segment (b), and has a storage elastic modulus at 20 ° C. (measured at a frequency of 10 Hz). E'20) is 800 to 2000 MPa, storage elastic modulus at 80 ° C. (E′80) is 0.5 to 2.5 MPa, and storage elastic modulus at 80 ° C. relative to storage elastic modulus at 20 ° C. (E'80 / E'20) is a resin composition in the range of 0.025% to 0.25%.
本発明の樹脂組成物は、結晶性ポリエーテルセグメント(a)と非晶性ポリエステルセグメント(b)を含有する必要があり、結晶性ポリエーテルセグメント(a)と前記非晶性ポリエステルセグメント(b)が共重合していることが好ましい。また、樹脂組成物は、非晶性であることが好ましい。樹脂組成物を非晶性とすることで溶剤溶解後の安定性を維持しつつ、幅広い温度での接着特性を維持することができる。非晶性とは、示差走査型熱量計(DSC)を用いて、-100℃~300℃まで20℃/分で昇温し、該昇温過程に明確な融解ピークを示さないものをいう。 The resin composition of the present invention needs to contain a crystalline polyether segment (a) and an amorphous polyester segment (b). The crystalline polyether segment (a) and the amorphous polyester segment (b) Is preferably copolymerized. Further, the resin composition is preferably amorphous. By making the resin composition amorphous, it is possible to maintain adhesive properties over a wide range of temperatures while maintaining stability after dissolving the solvent. Amorphous means that the temperature is raised from −100 ° C. to 300 ° C. at 20 ° C./min using a differential scanning calorimeter (DSC) and does not show a clear melting peak in the temperature raising process.
本発明の樹脂組成物中の結晶性ポリエーテルセグメント(a)の含有量としては、樹脂組成物のグリコール成分全体を100モル%としたとき1モル%以上であることが好ましく、3モル%以上であることがより好ましく、5モル%以上であることがさらに好ましい。また、80モル%以下が好ましく、75モル%以下がより好ましく、70モル%以下がさらに好ましく、60モル%以下が特に好ましい。結晶性ポリエーテルセグメント(a)の含有量が1モル%未満であると、20℃での貯蔵弾性率(E’20)が2000MPa超となり、十分な貯蔵弾性が得られないことがある。また、80モル%を超えると80℃での貯蔵弾性率(E’80)が0.5MPa未満となり、十分な貯蔵弾性が得られないことがある。 The content of the crystalline polyether segment (a) in the resin composition of the present invention is preferably 1 mol% or more, preferably 3 mol% or more when the total glycol component of the resin composition is 100 mol%. More preferably, it is 5 mol% or more. Moreover, 80 mol% or less is preferable, 75 mol% or less is more preferable, 70 mol% or less is more preferable, 60 mol% or less is especially preferable. When the content of the crystalline polyether segment (a) is less than 1 mol%, the storage elastic modulus (E′20) at 20 ° C. exceeds 2000 MPa, and sufficient storage elasticity may not be obtained. On the other hand, if it exceeds 80 mol%, the storage elastic modulus (E′80) at 80 ° C. becomes less than 0.5 MPa, and sufficient storage elasticity may not be obtained.
本発明の樹脂組成物は、20℃での貯蔵弾性率(E’20)が800~2000MPaであることが必要である。好ましくは900MPa以上であり、より好ましくは1000MPa以上であり、さらに好ましくは1100MPa以上である。また、1900MPa以下が好ましく、1800MPa以下がより好ましく、1700MPa以下がさらに好ましい。前記範囲を満足することで20℃における優れた接着性を発現することができる。 The resin composition of the present invention needs to have a storage elastic modulus (E′20) at 20 ° C. of 800 to 2000 MPa. Preferably it is 900 MPa or more, More preferably, it is 1000 MPa or more, More preferably, it is 1100 MPa or more. Moreover, 1900 MPa or less is preferable, 1800 MPa or less is more preferable, and 1700 MPa or less is more preferable. By satisfying the above range, excellent adhesiveness at 20 ° C. can be exhibited.
本発明の樹脂組成物は、80℃での貯蔵弾性率(E’80)が0.5~2.5MPaであることが必要である。好ましくは0.8MPa以上であり、より好ましくは1.0MPa以上であり、さらに好ましくは1.5MPa以上である。また、2.4MPa以下が好ましく、2.2MPa以下がより好ましく、2.0MPa以下さらに好ましい。前記範囲を満足することで80℃における優れた接着耐熱性を発現することができる。 The resin composition of the present invention is required to have a storage elastic modulus (E′80) at 80 ° C. of 0.5 to 2.5 MPa. Preferably it is 0.8 MPa or more, More preferably, it is 1.0 MPa or more, More preferably, it is 1.5 MPa or more. Moreover, 2.4 MPa or less is preferable, 2.2 MPa or less is more preferable, and 2.0 MPa or less is more preferable. By satisfying the above range, excellent adhesive heat resistance at 80 ° C. can be exhibited.
本発明の樹脂組成物は、20℃での貯蔵弾性率に対する80℃での貯蔵弾性率の割合(E’80/E’20)が0.025%~0.25%の範囲にあることが必要である。好ましくは0.03%以上であり、より好ましくは0.05%以上であり、さらに好ましくは0.1%以上である。また、0.25%未満が好ましく、0.24%以下がより好ましく、0.23%以下がさらに好ましい。前記範囲を満足することで20℃から80℃までの幅広い温度範囲で、優れた接着性や接着耐熱性を安定して発現することができる。 In the resin composition of the present invention, the ratio of the storage elastic modulus at 80 ° C. to the storage elastic modulus at 20 ° C. (E′80 / E′20) is in the range of 0.025% to 0.25%. is necessary. Preferably it is 0.03% or more, More preferably, it is 0.05% or more, More preferably, it is 0.1% or more. Moreover, less than 0.25% is preferable, 0.24% or less is more preferable, and 0.23% or less is more preferable. By satisfying the above range, excellent adhesiveness and adhesive heat resistance can be stably expressed in a wide temperature range from 20 ° C to 80 ° C.
本発明の樹脂組成物の貯蔵弾性率の測定は下記の方法による。すなわち、樹脂組成物を約200℃に調整したヒートプレス上にポリイミドフィルムを介して載せる。約2N/mm2の圧力で約20秒保持して、約1mm厚みのシートサンプルを得る。これを、長さ約15mm(掴み代除く)、幅約4mmに裁断し、アイティー計測制御株式会社製、動的粘弾性測定装置「DVA-200」にセットして、引張モードにて測定する。測定条件は、周波数を10Hzに固定して、4℃/分の昇温速度で-100℃から測定不能となるまで(最大250℃)スキャンする。 The storage elastic modulus of the resin composition of the present invention is measured by the following method. That is, the resin composition is placed on a heat press adjusted to about 200 ° C. via a polyimide film. Hold for about 20 seconds at a pressure of about 2 N / mm 2 to obtain a sheet sample of about 1 mm thickness. This is cut into a length of about 15 mm (excluding the grip allowance) and a width of about 4 mm, set in a dynamic viscoelasticity measuring device “DVA-200” manufactured by IT Measurement Control Co., Ltd., and measured in a tensile mode. . The measurement condition is that the frequency is fixed at 10 Hz, and scanning is performed from −100 ° C. until the measurement becomes impossible (maximum 250 ° C.) at a heating rate of 4 ° C./min.
前記配合量および貯蔵弾性率を満足することで、良好な溶剤可溶性および優れた溶液安定性を示し、かつ室温(約20℃)から80℃までの幅広い温度範囲で基材への追従性が良好な事により、接着性、接着耐熱性を保持することができる。 Satisfactory solvent solubility and excellent solution stability by satisfying the above blending amount and storage elastic modulus, and good followability to a substrate in a wide temperature range from room temperature (about 20 ° C.) to 80 ° C. It is possible to maintain adhesiveness and adhesive heat resistance.
本発明の樹脂組成物の数平均分子量の下限は特に限定されないが、3,000以上であることが好ましく、より好ましくは5,000以上、さらに好ましくは7,000以上である。また、数平均分子量の上限は特に限定されないが、好ましくは50,000以下、より好ましくは40,000以下、さらに好ましくは30,000以下である。数平均分子量が3,000未満であると弾性が不足することがあり、数平均分子量が50,000を超えると樹脂組成物の溶剤溶解性が低下することがある。 The lower limit of the number average molecular weight of the resin composition of the present invention is not particularly limited, but is preferably 3,000 or more, more preferably 5,000 or more, and further preferably 7,000 or more. The upper limit of the number average molecular weight is not particularly limited, but is preferably 50,000 or less, more preferably 40,000 or less, and still more preferably 30,000 or less. If the number average molecular weight is less than 3,000, elasticity may be insufficient, and if the number average molecular weight exceeds 50,000, the solvent solubility of the resin composition may be lowered.
本発明の樹脂組成物の還元粘度の下限は、0.6dl/g以上であることが好ましく、より好ましくは0.7dl/g以上であり、さらに好ましくは0.8dl/g以上である。また、還元粘度の上限は特に限定されないが、好ましくは1.5dl/g以下であり、より好ましくは1.4dl/g以下であり、さらに好ましくは1.3dl/g以下である。還元粘度が0.6dl/g未満であると弾性が不足することがあり、還元粘度が1.5dl/gを超えると樹脂組成物の溶剤溶解性が低下することがある。 The lower limit of the reduced viscosity of the resin composition of the present invention is preferably 0.6 dl / g or more, more preferably 0.7 dl / g or more, and further preferably 0.8 dl / g or more. The upper limit of the reduced viscosity is not particularly limited, but is preferably 1.5 dl / g or less, more preferably 1.4 dl / g or less, and further preferably 1.3 dl / g or less. If the reduced viscosity is less than 0.6 dl / g, the elasticity may be insufficient, and if the reduced viscosity exceeds 1.5 dl / g, the solvent solubility of the resin composition may be lowered.
本発明の樹脂組成物は、溶剤に良好な溶解性を示す。溶剤とは、芳香族炭化水素、脂肪族炭化水素、脂環族炭化水素、ケトン系溶剤、エーテル系溶剤または非プロトン性極性溶媒が挙げられる。具体的には、アセトン、メチルエチルケトン(以下、「MEK」ともいう。)、トルエン、キシレン、ヘキサン、ヘプタン、シクロヘキサン、メチルシクロヘキサン、テトラヒドロフラン、N-メチルピロリドン、ジメチルホルムアミド、ジメチルアセトアミド、ジメチルスルホキシド、ソルベッソ100、ソルベッソ150、ソルベッソ200などが挙げられるが、これらに限定されない。これらの溶剤を単独でまたは2種以上併用することができる。これら溶剤への溶解性は25℃で20重量%以上であることが好ましく、25重量%以上であることがより好ましく、30重量%以上であることがさらに好ましい。溶剤溶解性が優れるため、接着剤組成物とした際の加工性が良好となる。 The resin composition of the present invention exhibits good solubility in a solvent. Examples of the solvent include aromatic hydrocarbons, aliphatic hydrocarbons, alicyclic hydrocarbons, ketone solvents, ether solvents, and aprotic polar solvents. Specifically, acetone, methyl ethyl ketone (hereinafter, also referred to as “MEK”), toluene, xylene, hexane, heptane, cyclohexane, methylcyclohexane, tetrahydrofuran, N-methylpyrrolidone, dimethylformamide, dimethylacetamide, dimethylsulfoxide, Solvesso 100 , Solvesso 150, Solvesso 200 and the like, but are not limited thereto. These solvents can be used alone or in combination of two or more. The solubility in these solvents is preferably 20% by weight or more at 25 ° C., more preferably 25% by weight or more, and further preferably 30% by weight or more. Since the solvent solubility is excellent, the processability when an adhesive composition is obtained is improved.
本発明の樹脂組成物は、前記溶媒に30重量%となるように溶解後、25℃、8時間、保存しておいても、流動性を維持することができるほど安定であり、保存後の樹脂組成物は、室温(約20℃)および80℃での接着特性ともに良好である。 The resin composition of the present invention is stable enough to maintain fluidity even after being stored at 25 ° C. for 8 hours after being dissolved in the solvent so as to be 30% by weight. The resin composition has good adhesive properties at room temperature (about 20 ° C.) and 80 ° C.
本発明の樹脂組成物のカルボキシル基濃度は5~60当量/tであることが好ましく、10~55当量/tであることがより好ましく、15~50当量/tであることがさらに好ましい。5当量/t未満であると、接着性が低下し、60当量/tを超えると耐熱性が低下することがある。 The carboxyl group concentration of the resin composition of the present invention is preferably 5 to 60 equivalent / t, more preferably 10 to 55 equivalent / t, and further preferably 15 to 50 equivalent / t. When it is less than 5 equivalents / t, the adhesiveness is lowered, and when it exceeds 60 equivalents / t, the heat resistance may be lowered.
本発明の樹脂組成物の製造方法としては、公知の方法をとることができるが、例えば、結晶性ポリエーテルセグメント(a)と非晶性ポリエステルセグメント(b)を150~250℃でエステル化反応させた後、減圧しながら230~300℃で重縮合反応させることにより、溶剤可溶性弾性樹脂組成物を得ることができる。金属触媒としては、特に限定されないが、重合性の観点からTi原子が好ましく、具体的には、テトラブチルチタネートが重合性の観点から特に好ましい。触媒量は、特に限定されないが、好ましくは200ppm~1000ppmであり、1000ppmを超えると耐環境性が悪化することがある。 As a method for producing the resin composition of the present invention, a known method can be used. For example, an esterification reaction of a crystalline polyether segment (a) and an amorphous polyester segment (b) at 150 to 250 ° C. Thereafter, a polycondensation reaction is carried out at 230 to 300 ° C. under reduced pressure to obtain a solvent-soluble elastic resin composition. Although it does not specifically limit as a metal catalyst, Ti atom is preferable from a polymeric viewpoint, and specifically, tetrabutyl titanate is especially preferable from a polymeric viewpoint. The amount of the catalyst is not particularly limited, but is preferably 200 ppm to 1000 ppm. If it exceeds 1000 ppm, the environmental resistance may deteriorate.
本発明の樹脂組成物は、必要に応じて無水トリメリット酸、トリメチロールプロパン等の三官能以上のポリカルボン酸やポリオールを共重合しても差し支えない。 The resin composition of the present invention may be copolymerized with a tri- or higher functional polycarboxylic acid such as trimellitic anhydride or trimethylolpropane or a polyol, if necessary.
樹脂組成物の組成及び組成比を決定する方法としては、例えばポリエステル樹脂を重クロロホルム等の溶媒に溶解して測定する1H-NMRや13C-NMR、ポリエステル樹脂のメタノリシス後に測定するガスクロマトグラフィーによる定量(以下、メタノリシス-GC法と略記する場合がある)等が挙げられる。本発明においては、1H-NMRで組成及び組成比を決定することとする。 Examples of the method for determining the composition and composition ratio of the resin composition include 1 H-NMR and 13 C-NMR in which a polyester resin is dissolved in a solvent such as deuterated chloroform, and gas chromatography that is measured after methanolysis of the polyester resin. (Hereinafter sometimes abbreviated as methanolysis-GC method). In the present invention, the composition and composition ratio are determined by 1 H-NMR.
<フェノール性O原子を分子中に含む酸化防止剤(c)>
本発明の樹脂組成物に用いるフェノール性O原子を分子中に含む酸化防止剤(c)(以下、単に「酸化防止剤(c)」ともいう)は、フェノールを基本骨格に有する酸化防止剤であることが好ましい。限定的ではないが、具体的には、下記一般式(1)や下記一般式(2)に示されるヒンダードフェノール系化合物が挙げられる。酸化防止剤(c)は、結晶性ポリエーテルセグメント(a)が触媒Ti原子による解重合を受けて分解し、弾性が下がるのを防ぐ働きを持つ。そのため、特に樹脂組成物の重合中に使用することが有効である。さらに樹脂組成物を長時間高温環境下で使用し続けた際の、分子量低下によって接着力が低下するのを防ぐ効果も有する。
<Antioxidant (c) containing phenolic O atom in molecule>
The antioxidant (c) (hereinafter also simply referred to as “antioxidant (c)”) containing a phenolic O atom in the molecule used in the resin composition of the present invention is an antioxidant having phenol as a basic skeleton. Preferably there is. Specific examples include hindered phenol compounds represented by the following general formula (1) and the following general formula (2). The antioxidant (c) has a function of preventing the crystalline polyether segment (a) from being decomposed by being depolymerized by the catalyst Ti atom and decreasing in elasticity. Therefore, it is particularly effective to use it during the polymerization of the resin composition. Furthermore, it also has an effect of preventing the adhesive force from being reduced due to a decrease in molecular weight when the resin composition is used for a long time in a high temperature environment.
前記酸化防止剤(c)の含有量は、樹脂組成物100重量部に対して、0.05~5重量部が好ましく、より好ましくは0.08~4.5重量部であり、さらに好ましくは0.1~4重量部である。0.05重量部未満であると、酸化防止効果を十分に発揮することができず、5重量部を超えると樹脂からブリードアウトし、接着性に悪影響を与えることがある。 The content of the antioxidant (c) is preferably 0.05 to 5 parts by weight, more preferably 0.08 to 4.5 parts by weight, even more preferably 100 parts by weight of the resin composition. 0.1 to 4 parts by weight. If it is less than 0.05 part by weight, the antioxidant effect cannot be sufficiently exhibited, and if it exceeds 5 parts by weight, it may bleed out from the resin and adversely affect the adhesion.
<(c1)成分>
(c1)成分は一般式(1)に示されるヒンダードフェノール系化合物が好ましい。
一般式(1);
The component (c1) is preferably a hindered phenol compound represented by the general formula (1).
General formula (1);
(c1)成分は具体的には、チオジエチレンビス[3-(3,5-ジ-tert-ブチル4-ヒドロキシフェニル)プロピオネート]、オクタデシル[3-(3,5-ジ-tertブチル4-ヒドロキシフェニル)プロピオネート]、N,N’-ヘキサン-1,6-ジイルビス[3-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオンアミド]、1,3,5-トリメチル-2,4,6-トリス(3,5-ジ-tert-ブチル-4-ヒドロキシ)ベンゼン、1,6-ヘキサンジオール-ビス[3-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオネート]、トリエチレングリコール-ビス-3-(3-tert-ブチル-4-ヒドロキシ-5-メチルフェニル)プロピオネート、3,3’-チオビスプロピオン酸ジオクタデシルエステル、2,5,7,8-テトラメチル-(4’,8’,12’-トリメチルトリデシル)クロマン-6-オール、ステアリル-β-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオネート、3,9-ビス[1,1-ジメチル-2-[β-(3-tert-ブチル-4-ヒドロキシ-5-メチルフィエニル)プロピオニルオキシ]エチル ]2,4,8,10-テトラオキサスピロ(5,5)-ウンデカンを挙げることができるが、これらに限定されない。 Specifically, the component (c1) is thiodiethylene bis [3- (3,5-di-tert-butyl 4-hydroxyphenyl) propionate], octadecyl [3- (3,5-di-tert-butyl 4-hydroxy Phenyl) propionate], N, N′-hexane-1,6-diylbis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionamide], 1,3,5-trimethyl-2, 4,6-tris (3,5-di-tert-butyl-4-hydroxy) benzene, 1,6-hexanediol-bis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate ], Triethylene glycol-bis-3- (3-tert-butyl-4-hydroxy-5-methylphenyl) propionate, 3,3′-thiobi Propionic acid dioctadecyl ester, 2,5,7,8-tetramethyl- (4 ′, 8 ′, 12′-trimethyltridecyl) chroman-6-ol, stearyl-β- (3,5-di-tert- Butyl-4-hydroxyphenyl) propionate, 3,9-bis [1,1-dimethyl-2- [β- (3-tert-butyl-4-hydroxy-5-methylfienyl) propionyloxy] ethyl] 2, Examples include, but are not limited to, 4,8,10-tetraoxaspiro (5,5) -undecane.
<(c2)成分>
(c2)成分は一般式(2)に示されるヒンダードフェノール系化合物が好ましい。
一般式(2);
The component (c2) is preferably a hindered phenol compound represented by the general formula (2).
General formula (2);
(c2)成分は具体的には、トリブチルフォスフェート、トリス(2,4-ジ-ブチルフェニル)フォスファイト、ジステアリルペンタエリスリトールジフォスファイト、サイクリックネオペンタンテトライルビス(2,6-ジ-tert-ブチル-4-メチルフェニル)フォスファイト、トリスノニルフェニルホスファイト、トリス(2,4-ジ-tert-ブチルフェニル)ホスファイト、ジステアリルペンタエリスリトールジフォスファイト、ビス(2,4-ジ-tert-ブチルフェニル)ペンタエリスリトールホスファイト、ビス(2,6-ジ-tert-ブチル-4-メチル-フェニル)ペンタエリスリトールホスファイト、2,2-メチレン-ビス(4,6-ジ-tert-ブチルフェニル)オクチルホスファイト、テトラキス(2,4-ジ-tert-ブチルフェニル)-4,4’-ビフェニレン-ジホスホナイト、テトラキス(2,4-ジ-tert-ブチルフェニル)-4,4’-ビフェニレン-ジフォスフォナイト等が挙げられるが、これらに限ることはない。 Specifically, the component (c2) is tributyl phosphate, tris (2,4-di-butylphenyl) phosphite, distearyl pentaerythritol diphosphite, cyclic neopentanetetraylbis (2,6-di-). tert-butyl-4-methylphenyl) phosphite, trisnonylphenyl phosphite, tris (2,4-di-tert-butylphenyl) phosphite, distearyl pentaerythritol diphosphite, bis (2,4-di-) tert-butylphenyl) pentaerythritol phosphite, bis (2,6-di-tert-butyl-4-methyl-phenyl) pentaerythritol phosphite, 2,2-methylene-bis (4,6-di-tert-butyl) Phenyl) octyl phosphite, tetrakis ( 2,4-di-tert-butylphenyl) -4,4′-biphenylene-diphosphonite, tetrakis (2,4-di-tert-butylphenyl) -4,4′-biphenylene-diphosphonite, etc. However, it is not limited to these.
前記酸化防止剤(c1)のみを1種または2種以上使用することができるし、また前記酸化防止剤(c2)のみを1種または2種以上使用することができる。さらに、酸化防止剤(c1)と酸化防止剤(c2)を併用することもできる。 Only one or more of the antioxidants (c1) can be used, and only one or more of the antioxidants (c2) can be used. Furthermore, antioxidant (c1) and antioxidant (c2) can also be used together.
<接着剤組成物>
本発明の接着剤組成物は、前記樹脂組成物を含有し、樹脂組成物を主成分とする組成物である。主成分とは、接着剤組成物中、樹脂組成物が50重量%以上含有することを指し、好ましくは60重量%以上であり、より好ましくは70重量%以上である。少なすぎると良好な接着性、接着耐熱性を発揮することができないことがある。その他の成分として、後述の成分を配合することができる。
<Adhesive composition>
The adhesive composition of this invention is a composition which contains the said resin composition and has a resin composition as a main component. The main component means that the resin composition contains 50% by weight or more in the adhesive composition, preferably 60% by weight or more, and more preferably 70% by weight or more. If the amount is too small, good adhesiveness and adhesive heat resistance may not be exhibited. As other components, the components described later can be blended.
<その他の構成成分>
本発明の接着剤組成物には、本発明の効果を損なわない範囲で、密着性、柔軟性、耐久性等を改良する目的で樹脂組成物以外の成分を配合することができる。特に限定されないが、具体例として、ポリエステル、ポリアミド、ポリオレフィン、エポキシ、ポリカーボネート、アクリル、エチレンビニルアセテート、フェノール等の樹脂、イソシアネート化合物やメラミン等の硬化剤、タルクや雲母等の充填材、カーボンブラック、酸化チタン等の顔料、三酸化アンチモン、臭素化ポリスチレン等が挙げられる。これらの成分の含有量は、接着剤組成物中50重量%以下が好ましく、より好ましくは40重量%以下であり、さらに好ましくは30重量%以下である。含有量が50重量%を超えると電気電子部品に対する接着性、接着耐熱性が低下するおそれがある。
<Other components>
In the adhesive composition of the present invention, components other than the resin composition can be blended for the purpose of improving adhesion, flexibility, durability and the like within a range not impairing the effects of the present invention. Specific examples include, but are not limited to, polyesters, polyamides, polyolefins, epoxies, polycarbonates, acrylics, ethylene vinyl acetate, phenols and other resins, isocyanate compounds and curing agents such as melamine, fillers such as talc and mica, carbon black, Examples thereof include pigments such as titanium oxide, antimony trioxide, and brominated polystyrene. The content of these components is preferably 50% by weight or less in the adhesive composition, more preferably 40% by weight or less, and still more preferably 30% by weight or less. If the content exceeds 50% by weight, adhesion to electric / electronic parts and adhesion heat resistance may be reduced.
本発明をさらに詳細に説明するために以下に実施例、比較例を挙げるが、本発明は実施例によってなんら限定されるものではない。尚、実施例、比較例に記載された各測定値は次の方法によって測定したものである。 In order to describe the present invention in more detail, examples and comparative examples are given below, but the present invention is not limited to the examples. In addition, each measured value described in the Example and the comparative example was measured by the following method.
<ポリエステル組成>
ポリエステルの組成及び組成比は、共鳴周波数400MHzの1H-NMR測定(プロトン型核磁気共鳴分光測定)にて行った。測定装置はVARIAN社製NMR装置400-MRを用い、溶媒には重クロロホルムを用いた。
<Polyester composition>
The composition and composition ratio of the polyester were determined by 1 H-NMR measurement (proton nuclear magnetic resonance spectroscopy) at a resonance frequency of 400 MHz. The measuring apparatus was an NMR apparatus 400-MR manufactured by VARIAN, and deuterated chloroform was used as a solvent.
<貯蔵弾性率>
樹脂組成物のサンプルを、200℃に調整したヒートプレス上にポリイミドフィルム(東レデュポン社製「カプトン(登録商標)」)を介して載せ、2N/mm2の圧力で20秒保持して、1mm厚みのシートサンプルを得た。これを、長さ15mm(掴み代除く)、幅4mmに裁断し、アイティー計測制御株式会社製、動的粘弾性測定装置「DVA-200」にサンプルセットして、引張モードにて測定した。測定条件は、周波数を10Hzに固定して、4℃/分の昇温速度で-100℃から測定不能となるまで(最大250℃)のスキャンとした。
<Storage modulus>
A sample of the resin composition was placed on a heat press adjusted to 200 ° C. via a polyimide film (“Kapton (registered trademark)” manufactured by Toray DuPont) and held at a pressure of 2 N / mm 2 for 20 seconds to hold 1 mm. A sheet sample of thickness was obtained. This was cut into a length of 15 mm (excluding the gripping allowance) and a width of 4 mm, and a sample was set in a dynamic viscoelasticity measuring device “DVA-200” manufactured by IT Measurement Control Co., Ltd., and measured in a tensile mode. The measurement condition was a scan from −100 ° C. until measurement became impossible (maximum 250 ° C.) at a heating rate of 4 ° C./min with the frequency fixed at 10 Hz.
<溶剤可溶性試験>
樹脂組成物を、MEK/トルエン=1/4(重量比)の溶液に溶解(固形分濃度=30重量%)し、目視にて完全に溶解した場合「○」、不溶解物が残留していれば「×」とした。
<Solvent solubility test>
When the resin composition is dissolved in a solution of MEK / toluene = 1/4 (weight ratio) (solid content concentration = 30% by weight) and completely dissolved visually, “◯”, insoluble matter remains. “×”.
<溶液安定性試験>
前記樹脂組成物の溶液を室温下(約25℃)で1日静置した。1日静置後静かに傾け、目視にて流動性が見られれば「○」、流動性が見られなければ「×」とした。
<Solution stability test>
The solution of the resin composition was allowed to stand at room temperature (about 25 ° C.) for 1 day. After standing still for one day, it was gently tilted, and “○” was indicated when fluidity was visually observed, and “X” when fluidity was not observed.
<接着性試験>
前記溶液安定性試験が「○」であったサンプルをPETフィルムに塗布後、120℃7分間乾燥し、厚み60μmの塗膜を作製した。表面をよく洗浄したスズメッキ銅箔に、160℃、3MPaで前記塗膜を接着し、接着サンプルAを作製した。室温下(約25℃)、接着サンプルAを100mm/minの速度でT字剥離し、接着強度を測定した。
評価基準 ◎:接着強度25N/25mm以上
○:接着強度20N/25mm以上25N/25mm未満
△:接着強度15N/25mm以上20N/25mm未満
×:接着強度15N/25mm未満
<Adhesion test>
The sample whose solution stability test was “◯” was applied to a PET film, and then dried at 120 ° C. for 7 minutes to prepare a coating film having a thickness of 60 μm. The coating film was adhered to a tin-plated copper foil whose surface was washed well at 160 ° C. and 3 MPa to prepare an adhesion sample A. At room temperature (about 25 ° C.), the adhesive sample A was peeled off at a rate of 100 mm / min, and the adhesive strength was measured.
Evaluation criteria A: Adhesion strength 25 N / 25 mm or more B: Adhesion strength 20 N / 25 mm or more and less than 25 N / 25 mm Δ: Adhesion strength 15 N / 25 mm or more and less than 20 N / 25 mm X: Adhesion strength 15 N / 25 mm or less
<接着耐熱性試験>
前記接着サンプルAを、80℃で5分間保温し、80℃の環境下、100mm/minの速度でT字剥離し、接着強度を測定した。
評価基準 ◎:接着強度25N/25mm以上
○:接着強度20N/25mm以上25N/25mm未満
△:接着強度15N/25mm以上20N/25mm未満
×:接着強度15N/25mm未満
<Adhesion heat resistance test>
The adhesive sample A was kept at 80 ° C. for 5 minutes, peeled T-shaped at a speed of 100 mm / min in an environment of 80 ° C., and the adhesive strength was measured.
Evaluation criteria A: Adhesion strength 25 N / 25 mm or more B: Adhesion strength 20 N / 25 mm or more and less than 25 N / 25 mm Δ: Adhesion strength 15 N / 25 mm or more and less than 20 N / 25 mm X: Adhesion strength 15 N / 25 mm or less
<樹脂組成物の製造例>
撹拌機、温度計、溜出用冷却器を装備した反応缶内にテレフタル酸194重量部、イソフタル酸194重量部、エチレングリコール161重量部、ネオペンチルグリコール151重量部、テトラブチルチタネート(Ti原子として270ppm)を加え、170~220℃で3時間エステル化反応を行った。エステル化反応終了後、数平均分子量1000のポリテトラメチレングリコール「PTMG1000」(三菱化学社製)を300重量部とヒンダードフェノール系酸化防止剤「イルガノックス(登録商標)1010」(チバガイギー社製)を0.6重量部投入し、255℃まで昇温する一方、系内をゆっくり減圧にしてゆき、60分かけて255℃で665Paとした。そしてさらに133Pa以下で60分間重縮合反応を行い、実施例1の樹脂組成物を得た。この樹脂組成物の組成及び物性値を表1、2に示した。また、実施例2~10、比較例1~5の樹脂組成物を、実施例1と同様な方法により合成した。それぞれの組成及び物性値を表1、2に示した。
<Production Example of Resin Composition>
In a reaction vessel equipped with a stirrer, a thermometer, and a condenser for distillation, 194 parts by weight of terephthalic acid, 194 parts by weight of isophthalic acid, 161 parts by weight of ethylene glycol, 151 parts by weight of neopentyl glycol, tetrabutyl titanate (as Ti atoms) 270 ppm) and an esterification reaction was carried out at 170-220 ° C. for 3 hours. After completion of the esterification reaction, 300 parts by weight of polytetramethylene glycol “PTMG1000” (manufactured by Mitsubishi Chemical Corporation) having a number average molecular weight of 1000 and hindered phenolic antioxidant “Irganox (registered trademark) 1010” (manufactured by Ciba Geigy) Was added to 0.6 parts by weight, and the temperature was raised to 255 ° C., while the pressure in the system was slowly reduced to 665 Pa at 255 ° C. over 60 minutes. Further, a polycondensation reaction was performed at 133 Pa or less for 60 minutes to obtain a resin composition of Example 1. The composition and physical property values of this resin composition are shown in Tables 1 and 2. In addition, the resin compositions of Examples 2 to 10 and Comparative Examples 1 to 5 were synthesized by the same method as in Example 1. The respective compositions and physical property values are shown in Tables 1 and 2.
表中の略号は以下の通りである。
TPA:テレフタル酸、IPA:イソフタル酸、SA:セバシン酸、NDCA:2,6-ナフタレンジカルボン酸、EG:エチレングリコール、NPG:ネオペンチルグリコール、BD:1,4-ブタンジオール、2MG:2-メチル-1,3-プロパンジオール、PTMG1000:ポリテトラメチレンエーテルグリコール1000、PEG2000:ポリエチレングリコール2000、PTXG1000:ポリ(テトラメチレンエーテルグリコール-ネオペンチルグリコール)1000
Abbreviations in the table are as follows.
TPA: terephthalic acid, IPA: isophthalic acid, SA: sebacic acid, NDCA: 2,6-naphthalenedicarboxylic acid, EG: ethylene glycol, NPG: neopentyl glycol, BD: 1,4-butanediol, 2MG: 2-methyl 1,3-propanediol, PTMG1000: polytetramethylene ether glycol 1000, PEG2000: polyethylene glycol 2000, PTXG1000: poly (tetramethylene ether glycol-neopentyl glycol) 1000
酸化防止剤(c)は、以下のもの用いた。
酸化防止剤(i):式(3)で表されるイルガノックス(登録商標)1010、チバガイギー社製。
式(3)
式(4)
式(5)
式(6)
Antioxidant (i): Irganox (registered trademark) 1010 represented by formula (3), manufactured by Ciba Geigy Corporation.
Formula (3)
Formula (4)
Formula (5)
Formula (6)
<樹脂組成物の評価>
実施例1
実施例1の樹脂組成物について、<溶剤可溶性試験>、<溶液安定性試験>、<接着性試験>、<接着耐熱性試験>を行った。
<Evaluation of resin composition>
Example 1
The resin composition of Example 1 was subjected to <solvent solubility test>, <solution stability test>, <adhesion test>, and <adhesion heat resistance test>.
実施例2~9
実施例2~9の樹脂組成物を用いて、実施例1と同様の試験を実施した。評価結果を表2に示した。
Examples 2 to 9
Using the resin compositions of Examples 2 to 9, the same test as in Example 1 was performed. The evaluation results are shown in Table 2.
比較例1~5
比較例1~5を用い、実施例1と同様の試験を実施した。評価結果を表2に示した。
Comparative Examples 1-5
The same test as in Example 1 was performed using Comparative Examples 1-5. The evaluation results are shown in Table 2.
実施例1~9は特許請求の範囲を満たし、<溶剤可溶性試験>、<溶液安定性試験>、<接着試験>、<接着耐熱性試験>の結果全てが良好となった。これに対し、比較例1は、(b)成分が結晶性ポリエステルであるため溶剤に溶解せず、<溶剤溶解性試験>が不良である。比較例2は、(c)成分にフェノール性O原子を分子中に含む酸化防止剤を使用していないため、重合性が不良となり、<接着耐熱性試験>が不良である。比較例3は(a)成分が含まれておらず、<接着試験>が不良である。比較例4は(a)成分が含まれておらず、さらに(b)成分が結晶性ポリエステルであるため溶剤に溶解せず、<溶剤溶解性試験>が不良である。比較例5は、(a)成分が非晶性ポリエーテルであるため、貯蔵弾性率測定時に、80℃到達前に溶解してしまい、80℃での貯蔵弾性率を測定できなかった。また、<接着耐熱性試験>が不良である。 Examples 1 to 9 satisfied the claims, and all the results of <solvent solubility test>, <solution stability test>, <adhesion test>, and <adhesion heat resistance test> were satisfactory. On the other hand, since the component (b) is a crystalline polyester, the comparative example 1 does not dissolve in the solvent and the <solvent solubility test> is poor. Since the comparative example 2 does not use the antioxidant which contains a phenolic O atom in a molecule | numerator in (c) component, superposition | polymerization becomes bad and <adhesion heat resistance test> is bad. Comparative Example 3 does not contain the component (a), and the <adhesion test> is poor. Comparative Example 4 does not contain the component (a), and since the component (b) is a crystalline polyester, it does not dissolve in the solvent, and the <solvent solubility test> is poor. In Comparative Example 5, since the component (a) is an amorphous polyether, it was dissolved before reaching 80 ° C. during storage elastic modulus measurement, and the storage elastic modulus at 80 ° C. could not be measured. Moreover, <adhesion heat resistance test> is poor.
本発明の樹脂組成物は、溶剤可溶性を有しハンドリング性に優れ、幅広い温度範囲での接着特性を保持でき、高温環境負荷や高温高湿環境負荷に対する耐久性に優れる。よって、自動車、通信、コンピュータ、家電用途各種のフレクシブルフラットケーブルや電子部品、プリント基板を有するスイッチ、センサー等の用途に好適である。
The resin composition of the present invention has solvent solubility, excellent handling properties, can maintain adhesive properties in a wide temperature range, and is excellent in durability against high temperature environmental loads and high temperature high humidity environmental loads. Accordingly, it is suitable for various applications such as automobiles, communications, computers, home appliances, flexible flat cables, electronic components, switches having a printed circuit board, and sensors.
Claims (4)
周波数10Hzで測定した際の
20℃での貯蔵弾性率(E’20)が800~2000MPaであり、
80℃での貯蔵弾性率(E’80)が0.5~2.5MPaであり、
かつ20℃での貯蔵弾性率に対する80℃での貯蔵弾性率の割合(E’80/E’20)が0.025%~0.25%の範囲にあることを特徴とする溶剤可溶性弾性樹脂組成物。 A solvent-soluble elastic resin composition containing a crystalline polyether segment (a) and an amorphous polyester segment (b),
The storage elastic modulus (E'20) at 20 ° C. when measured at a frequency of 10 Hz is 800 to 2000 MPa,
The storage elastic modulus (E'80) at 80 ° C. is 0.5 to 2.5 MPa,
And the ratio of the storage elastic modulus at 80 ° C. to the storage elastic modulus at 20 ° C. (E′80 / E′20) is in the range of 0.025% to 0.25%. Composition.
一般式(1);
一般式(2);
General formula (1);
General formula (2);
An adhesive composition containing the solvent-soluble elastic resin composition according to any one of claims 1 to 3.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201480003256.6A CN104822770A (en) | 2013-03-11 | 2014-03-06 | Solvent-soluble elastic resin composition |
| JP2014530988A JPWO2014141984A1 (en) | 2013-03-11 | 2014-03-06 | Solvent-soluble elastic resin composition |
| KR1020157011930A KR20150126814A (en) | 2013-03-11 | 2014-03-06 | Solvent-soluble elastic resin composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013047963 | 2013-03-11 | ||
| JP2013-047963 | 2013-03-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014141984A1 true WO2014141984A1 (en) | 2014-09-18 |
Family
ID=51536648
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/055722 Ceased WO2014141984A1 (en) | 2013-03-11 | 2014-03-06 | Solvent-soluble elastic resin composition |
Country Status (5)
| Country | Link |
|---|---|
| JP (1) | JPWO2014141984A1 (en) |
| KR (1) | KR20150126814A (en) |
| CN (1) | CN104822770A (en) |
| TW (1) | TW201439201A (en) |
| WO (1) | WO2014141984A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104449519A (en) * | 2014-12-31 | 2015-03-25 | 明冠新材料股份有限公司 | Glue for combining solar cell backboard film and PE film and preparing method of glue |
| CN104531031A (en) * | 2014-12-31 | 2015-04-22 | 明冠新材料股份有限公司 | Glue for combining back plate film and EVA film of solar cell and preparation method of glue |
| KR20210084462A (en) | 2018-10-25 | 2021-07-07 | 유니띠까 가부시키가이샤 | resin composition |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH09263749A (en) * | 1995-03-15 | 1997-10-07 | Nitto Denko Corp | Adhesive composition and its adhesive sheets |
| JP2003040990A (en) * | 2001-07-31 | 2003-02-13 | Dainippon Ink & Chem Inc | Method for producing polyhydroxycarboxylic acid copolymer |
| JP2008231358A (en) * | 2007-03-23 | 2008-10-02 | Dic Corp | Image display module for electronic terminal and pressure sensitive adhesive sheet |
| JP2010083962A (en) * | 2008-09-30 | 2010-04-15 | Dic Corp | Heat-adhesive sheet for key sheet and key sheet |
| JP2013072009A (en) * | 2011-09-28 | 2013-04-22 | Toray Ind Inc | Hot-melt adhesive film, and hot-melt adhesive film laminate |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0517675A (en) * | 1991-07-15 | 1993-01-26 | Monsant Kasei Kk | Resin composition which is impact-resistant at low temperature |
| JP4409023B2 (en) * | 2000-01-17 | 2010-02-03 | ユニチカ株式会社 | Copolymerized polyester resin for adhesive and adhesive using the same |
| JP2004161890A (en) * | 2002-11-13 | 2004-06-10 | Nippon Ester Co Ltd | Polyester resin for adhesive, adhesive, and laminated body |
| JP2006037013A (en) * | 2004-07-29 | 2006-02-09 | Nippon Ester Co Ltd | Method for producing polyester resin, polyester resin for adhesive given by using the same, adhesive, and laminated product |
| FR2958649B1 (en) * | 2010-04-07 | 2012-05-04 | Arkema France | BLOCK COPOLYMER FROM RENEWABLE MATERIALS AND PROCESS FOR PRODUCING SUCH A BLOCK COPOLYMER |
| CN102476440A (en) * | 2010-11-23 | 2012-05-30 | 合肥杰事杰新材料股份有限公司 | Extrusion equipment and extrusion processing method for thermoplastic polyester elastomer |
-
2014
- 2014-03-06 WO PCT/JP2014/055722 patent/WO2014141984A1/en not_active Ceased
- 2014-03-06 CN CN201480003256.6A patent/CN104822770A/en active Pending
- 2014-03-06 JP JP2014530988A patent/JPWO2014141984A1/en not_active Withdrawn
- 2014-03-06 KR KR1020157011930A patent/KR20150126814A/en not_active Withdrawn
- 2014-03-07 TW TW103107934A patent/TW201439201A/en unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH09263749A (en) * | 1995-03-15 | 1997-10-07 | Nitto Denko Corp | Adhesive composition and its adhesive sheets |
| JP2003040990A (en) * | 2001-07-31 | 2003-02-13 | Dainippon Ink & Chem Inc | Method for producing polyhydroxycarboxylic acid copolymer |
| JP2008231358A (en) * | 2007-03-23 | 2008-10-02 | Dic Corp | Image display module for electronic terminal and pressure sensitive adhesive sheet |
| JP2010083962A (en) * | 2008-09-30 | 2010-04-15 | Dic Corp | Heat-adhesive sheet for key sheet and key sheet |
| JP2013072009A (en) * | 2011-09-28 | 2013-04-22 | Toray Ind Inc | Hot-melt adhesive film, and hot-melt adhesive film laminate |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104449519A (en) * | 2014-12-31 | 2015-03-25 | 明冠新材料股份有限公司 | Glue for combining solar cell backboard film and PE film and preparing method of glue |
| CN104531031A (en) * | 2014-12-31 | 2015-04-22 | 明冠新材料股份有限公司 | Glue for combining back plate film and EVA film of solar cell and preparation method of glue |
| KR20210084462A (en) | 2018-10-25 | 2021-07-07 | 유니띠까 가부시키가이샤 | resin composition |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2014141984A1 (en) | 2017-02-16 |
| CN104822770A (en) | 2015-08-05 |
| KR20150126814A (en) | 2015-11-13 |
| TW201439201A (en) | 2014-10-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5187468B1 (en) | Adhesive composition, laminate and polyester polyol | |
| JP4978753B2 (en) | RESIN COMPOSITION FOR ADHESIVE, ADHESIVE CONTAINING THE SAME, ADHESIVE SHEET AND A PRINTED WIRING BOARD CONTAINING THE SAME AS ADHESIVE LAYER | |
| CN106753015B (en) | Bottom coating suitable for FFC insulating tape and FFC insulating tape | |
| JP7211403B2 (en) | Adhesive composition and adhesive | |
| WO2014141984A1 (en) | Solvent-soluble elastic resin composition | |
| WO2023063386A1 (en) | Crosslinked polyester resin, adhesive composition, and adhesive sheet | |
| JP7334827B2 (en) | Adhesive composition and adhesive | |
| JP7002122B2 (en) | Laminate | |
| CN114555749B (en) | Adhesive composition for flexible printed circuit board, adhesive for flexible printed circuit board, and flexible printed circuit board | |
| TW201437253A (en) | Curable resin and method for producing same | |
| JP7279843B2 (en) | Polyester resin, adhesive composition and adhesive | |
| JP6003090B2 (en) | Biomass plastic paint | |
| TW201938632A (en) | Saturated copolyester resin and coating composition comprising same | |
| JP2018044094A (en) | Resin composition, and coated film and laminate using the same | |
| JP2009263657A (en) | Non-aqueous coating agent composition for plastic | |
| JP6592629B1 (en) | Resin composition | |
| JP2013177496A (en) | Biomass plastic coating | |
| JP5293754B2 (en) | Polyester resin for molding, resin composition and molded product using the same | |
| JP5794425B2 (en) | Resin composition for sealing electric and electronic parts, sealed electric and electronic parts and method for producing the same | |
| WO2022158384A1 (en) | Resin composition and electrical/electronic part encapsulation body | |
| WO2022158385A1 (en) | Resin composition and encapsulated electrical/electronic component | |
| JP2012172102A (en) | Biomass plastic coating | |
| JP2011241265A (en) | Thermoplastic polyester resin composition and molded article comprising the same | |
| JP2010043286A (en) | Polyester resin for molding, resin composition and molded article using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2014530988 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14763718 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20157011930 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14763718 Country of ref document: EP Kind code of ref document: A1 |