WO2014122671A2 - Solid oral compositions of saxagliptin - Google Patents
Solid oral compositions of saxagliptin Download PDFInfo
- Publication number
- WO2014122671A2 WO2014122671A2 PCT/IN2014/000081 IN2014000081W WO2014122671A2 WO 2014122671 A2 WO2014122671 A2 WO 2014122671A2 IN 2014000081 W IN2014000081 W IN 2014000081W WO 2014122671 A2 WO2014122671 A2 WO 2014122671A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- saxagliptin
- coating
- tablets
- tablet
- coating layer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
- A61K9/2086—Layered tablets, e.g. bilayer tablets; Tablets of the type inert core-active coat
- A61K9/209—Layered tablets, e.g. bilayer tablets; Tablets of the type inert core-active coat containing drug in at least two layers or in the core and in at least one outer layer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/2833—Organic macromolecular compounds
- A61K9/286—Polysaccharides, e.g. gums; Cyclodextrin
- A61K9/2866—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
- compositions comprising saxagliptin or a pharmaceutically acceptable salt thereof.
- Saxagliptin is an orally-active inhibitor of the DPP4 enzyme.
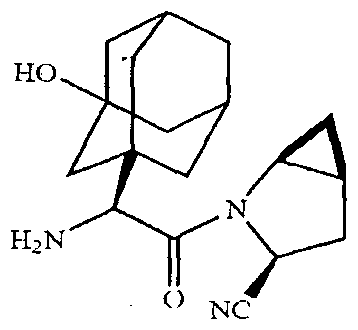
- the chemical name of saxagliptin is (lS,3S,5S)-2-[(2S)-2-Amino-2-(3-hydroxyadamantan-l-yl)acetyl]-2- azabicyclo [3.1.0]hexane-3-carbonitrile, disclosed in U.S. Patent No. 6,395,767. It's structural formula as follows:
- Saxagliptin is commercially available from Bristol Myers Squibb as ONGLYZA* 1 in the form of saxagliptin hydrochloride as oral tablets containing equivalent to 2.5mg or 5.0mg of saxagliptin base.
- ONGLYZA ® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus in multiple clinical settings.
- U.S. Patent No. 7,951,400 disclose coated tablet comprising an inert tablet core coated with three polyvinyl alcohol based coating layers i.e. inner seal coating layer, second coating layer comprising saxagliptin and finally outer protective coating layer.
- inner seal coating layer i.e. inner seal coating layer
- second coating layer comprising saxagliptin
- outer protective coating layer There is a need to develop alternative compositions of saxagliptin using simplified process. Accordingly, inventors of the present invention have developed novel compositions of saxagliptin and process for preparing the same.
- the present invention relates to film coated tablet comprising a core comprising saxagliptin and one more pharmaceutically acceptable excipients.
- One embodiment of the present invention describes a solid oral tablet composition
- a solid oral tablet composition comprising (a) a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients; (b) an inner seal coating layer on the said tablet core; (c) a drug layer comprising saxagliptin and one or more pharmaceutically acceptable excipients on the said seal coating; (d) an outer seal coating layer on the said drug layer; and (e) an outer film coating layer on the said seal coating.
- Another embodiment of the present invention describes process for preparing solid oral tablet comprising: (a) tablet core comprising 5% to 20% saxagliptin based on weight of the saxagliptin in the total composition, prepared by direct compression; (b) coating the tablet core of step (a) with a seal coating layer; (c) coating the tablets of step (b) with drug layer comprising 80% to 95% saxagliptin based on weight of the saxagliptin in the total composition; (d) coating the tablets of step (c) with a seal coating layer, and finally (e) coating the tablets of step (d) with a film coating layer.
- solid oral tablet comprising a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients and atleast one coating layer comprising stearic acid, microcrystalline cellulose and hydroxypropyl methylcellulose.
- the composition of the present invention is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus in multiple clinical settings.
- active agent or “drug” used interchangeably, is defined to mean active drug (e.g. saxagliptin), that induce a desired pharmacological or physiological effect.
- saxagliptin as used herein according to the present invention includes saxagliptin in the form of free base, in the form of a pharmaceutically acceptable salt thereof, or solid dispersion of saxagliptin salt, Preferably saxagliptin hydrochloride.
- excipient means a pharmacologically inactive component such as a diluent, disintegrant, carrier, etc of a pharmaceutical product.
- excipients that are useful in preparing a pharmaceutical composition are generally safe and non-toxic.
- the singular forms "a”, “an”, and “the” include plural references unless the context clearly dictates otherwise.
- a reference to “a method” or “a process” includes one or more methods, one or more processes and/or steps of the type described herein and/or which will become apparent to those persons skilled in the art upon reading this disclosure and so forth.
- composition or “solid oral composition” as used herein synonymously include solid dosage forms such as tablets, capsules, granules, mini-tablets and the like meant for oral administration.
- the present invention relates to pharmaceutical compositions of saxagliptin. More particularly, the present invention relates to solid oral compositions of saxagliptin and process for their preparation.
- One embodiment of the present invention describes a solid oral tablet composition
- a solid oral tablet composition comprising (a) a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients; (b) an inner seal coating layer on the said tablet core; (c) a drug layer comprising saxagliptin and one or more pharmaceutically acceptable excipients on the said seal coating; (d) an outer seal coating layer on the said drug layer; and (e) an outer film coating layer on the said seal coating.
- pharmaceutically acceptable excipients selected from diluents, disintegrants, glidants, lubricants and the like, and combinations thereof.
- diluents include but are not limited to lactose, microcrystalline cellulose, starch, corn starch, pregelatinized starch, maize starch, potato starch, powdered celluloses, sorbitol, xylitol, dibasic calcium phosphate, calcium phosphate, calcium carbonate, magnesium carbonate and the like, and combinations thereof.
- Various disintegrants include but are not limited to croscarmellose sodium, polacrilin potassium, sodium starch glycolate, crospovidone, pregelatinized starch, and the like, and combinations thereof.
- Various glidants include but are not limited to colloidal silicon dioxide, magnesium silicate, magnesium trisilicate, talc, and other fonns of silicon dioxide, such as aggregated silicates and hydrated silica and the like, and combinations thereof.
- Various lubricants include but are not limited to magnesium stearate, stearic acid, talc, fumaric acid, palmitic acid, sodium stearyl fumarate, carnauba wax, hydrogenated vegetable oils, mineral oil, polyethylene glycols and the like, and combinations thereof.
- Another embodiment of the present invention describes process for preparing solid oral tablet comprising: (a) tablet core comprising 5% to 20% saxagliptin based on weight of the saxagliptin in the total composition, prepared by direct compression; (b) coating the tablet core of step (a) with a seal coating layer; (c) coating the tablets of step (b) with drug layer comprising 80% to 95% saxagliptin based on weight of the saxagliptin in the total composition; (d) coating the tablets of step (c) with a seal coating layer, and finally (e) coating the tablets of step (d) with a film coating layer.
- the seal coating used according to the present invention is a hydroxypropyl methylcellulose based coating system.
- Solid dispersion of saxagliptin hydrochloride of the present invention is prepared as per the disclosure of unpublished provisional application No. IN 5537/CHE/2012 assigned to Hetero Research Foundation.
- Solid dispersion of saxagliptin hydrochloride may be prepared by known process such as hot melt extrusion, fluid bed granulation, spray drying, melt agglomeration, co- precipitation, freeze drying, solvent evaporation, nitrogen stream, amorphous precipitation in crystalline matrix, supercritical fluid, eutectics or solid solution process.
- Tablet core of the present invention may be prepared either by direct compression or granulation process.
- Direct compression process involves sifting and blending the saxagliptin and one or more excipients, followed by lubrication, and finally compressing into tablets.
- Granulation process involves sifting and blending saxagliptin and one or more excipients to form a dry mix, and then slugging/ compacting/ wet granulating the dry mix to form the desired granules, followed by lubrication and finally compressing into tablets.
- Tablet core of the present invention further coated with a film coating composition.
- Film coating composition of the present invention comprises stearic acid in an amount of 1% to 35% by weight, preferably 5% to 30% by weight; macrocrystalline cellulose in an amount of 1 % to 20% by weight, preferably 5% to 15% by weight of the film coating composition; and hydroxypropyl methylcellulose.
- Film coating composition used according to the present invention is available as SEPIFILMTM which is being marketed by SEPPIC.
- solid oral tablet comprising a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients and atleast one coating layer comprising stearic acid, microcrystalline cellulose and hydroxypropyl methylcellulose.
- the composition of the present invention is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus in multiple clinical settings.
- Table 1 Tablet composition of SaxagHptin hydrochloride:
- saxagliptin hydrochloride is equivalent to 5 mg of saxagliptin.
- 'SEPIFILM LP 007 contains hypromellose (60% - 70%), microcrystalline cellulose (5%-15%), stearic acid (20% - 30%). Opadry clear is a hypromellose based coating system.
- magnesium stearate was sifted through mesh #60,
- step 3 blend of step 1, was lubricated with magnesium stearate of step 2, and compressed into tablets,
- step 3 tablets of step 3 , were seal coated with Opadry clear,
- step 6 tablets of step 6, were over film coated with SEPIFILMTM LP 007.
- Table 2 Tablet compositions of Saxagliptin hydrochloride:
- 'SEPIFILM LP 007 contains hypromellose (60% - 70%), microcrystalline cellulose (5%- 15%), stearic acid (20% - 30%).
- $SEPIFILM LP 010 contains Hypromellose (75% - 85%), microcrystalline cellulose (5% - 15%), stearic acid (5% - 15%).
- magnesium stearate was sifted through mesh #60,
- step 3 blend of step 1 , was lubricated with magnesium stearate of step 2, and compressed into tablets, and finally
- step 4 tablets of step 3, were coated with SEPIFILMTM LP.
- Table 3 Tablet composition of Saxagliptin hydrochloride: ngredients Example 3 (mg/tab)
- saxagliptin hydrochloride is equivalent to 5 mg of saxagliptin.
- *SEPIFILM LP 007 contains hypromellose (60% - 70%), microcrystalline cellulose (5%-15%), stearic acid (20% - 30%).
- magnesium stearate was sifted through mesh #60,
- step 1 blend of step 1 ,. was lubricated with magnesium stearate of step 2, and compressed into tablets,
- step 3 tablets of step 3 , were seal coated with SEPIFILMTM LP 007,
- Table 4 Tablet compositions of Saxagliptin hydrochloride solid dispersion:
- 22.32 mg of solid dispersion of saxagliptin hydrochloride contains 5.58 mg of saxagliptin hydrochloride or 5 mg of saxagliptin.
- SEPIFILM LP contains hypromellose, microcrystalline cellulose, stearic acid.
- Saxagliptin hydrochloride Solid dispersion
- lactose monohydrate lactose monohydrate
- microcrystalline cellulose microcrystalline cellulose
- croscarmellose sodium were sifted and blended through mesh #40
- magnesium stearate was sifted through mesh #60,
- step 3 blend of step 1, was lubricated with magnesium stearate of step 2, and compressed into tablets, and finally
- step 4 tablets of step 3, were coated with SEPIFILMTM LP.
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Diabetes (AREA)
- Epidemiology (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The present invention relates to pharmaceutical compositions of saxagliptin. Particularly, the present invention relates to tablet compositions of saxagliptin and process for their preparation.
Description
SOLID ORAL COMPOSITIONS OF SAXAGLIPTIN
PRIORITY This patent application claims priority to Indian patent application number
545/CHE/2013, filed on February 08, 2013, the contents of which are incorporated by reference herein in their entirety.
FIELD OF THE DISCLOSURE
The present disclosure relates to pharmaceutical compositions comprising saxagliptin or a pharmaceutically acceptable salt thereof.' ·
BACKGROUND
Saxagliptin is an orally-active inhibitor of the DPP4 enzyme. The chemical name of saxagliptin is (lS,3S,5S)-2-[(2S)-2-Amino-2-(3-hydroxyadamantan-l-yl)acetyl]-2- azabicyclo [3.1.0]hexane-3-carbonitrile, disclosed in U.S. Patent No. 6,395,767. It's structural formula as follows:
Saxagliptin is commercially available from Bristol Myers Squibb as ONGLYZA*1 in the form of saxagliptin hydrochloride as oral tablets containing equivalent to 2.5mg or 5.0mg of saxagliptin base. ONGLYZA® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus in multiple clinical settings.
U.S. Patent No. 7,951,400 disclose coated tablet comprising an inert tablet core coated with three polyvinyl alcohol based coating layers i.e. inner seal coating layer, second coating layer comprising saxagliptin and finally outer protective coating layer.
There is a need to develop alternative compositions of saxagliptin using simplified process. Accordingly, inventors of the present invention have developed novel compositions of saxagliptin and process for preparing the same.
SUMMARY
The present invention relates to film coated tablet comprising a core comprising saxagliptin and one more pharmaceutically acceptable excipients.
One embodiment of the present invention describes a solid oral tablet composition comprising (a) a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients; (b) an inner seal coating layer on the said tablet core; (c) a drug layer comprising saxagliptin and one or more pharmaceutically acceptable excipients on the said seal coating; (d) an outer seal coating layer on the said drug layer; and (e) an outer film coating layer on the said seal coating.
Another embodiment of the present invention describes process for preparing solid oral tablet comprising: (a) tablet core comprising 5% to 20% saxagliptin based on weight of the saxagliptin in the total composition, prepared by direct compression; (b) coating the tablet core of step (a) with a seal coating layer; (c) coating the tablets of step (b) with drug layer comprising 80% to 95% saxagliptin based on weight of the saxagliptin in the total composition; (d) coating the tablets of step (c) with a seal coating layer, and finally (e) coating the tablets of step (d) with a film coating layer.
Another embodiment of the present invention describes solid oral tablet comprising a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients and atleast one coating layer comprising stearic acid, microcrystalline cellulose and hydroxypropyl methylcellulose. The composition of the present invention is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus in multiple clinical settings.
DETAILED DESCRIPTION
The term "active agent" or "drug" used interchangeably, is defined to mean active drug (e.g. saxagliptin), that induce a desired pharmacological or physiological effect.
The term "saxagliptin" as used herein according to the present invention includes saxagliptin in the form of free base, in the form of a pharmaceutically acceptable salt thereof, or solid dispersion of saxagliptin salt, Preferably saxagliptin hydrochloride.
The term "excipient" means a pharmacologically inactive component such as a diluent, disintegrant, carrier, etc of a pharmaceutical product. The excipients that are useful in preparing a pharmaceutical composition are generally safe and non-toxic. As used in this specification and the appended claims, the singular forms "a", "an", and "the" include plural references unless the context clearly dictates otherwise. Thus for example, a reference to "a method" or "a process" includes one or more methods, one or more processes and/or steps of the type described herein and/or which will become apparent to those persons skilled in the art upon reading this disclosure and so forth. The term "composition" or "solid oral composition" as used herein synonymously include solid dosage forms such as tablets, capsules, granules, mini-tablets and the like meant for oral administration.
The present invention relates to pharmaceutical compositions of saxagliptin. More particularly, the present invention relates to solid oral compositions of saxagliptin and process for their preparation.
One embodiment of the present invention describes a solid oral tablet composition comprising (a) a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients; (b) an inner seal coating layer on the said tablet core; (c) a drug layer comprising saxagliptin and one or more pharmaceutically acceptable excipients on the said seal coating; (d) an outer seal coating layer on the said drug layer; and (e) an outer film coating layer on the said seal coating.
According to the present invention pharmaceutically acceptable excipients selected from diluents, disintegrants, glidants, lubricants and the like, and combinations thereof.
Various diluents include but are not limited to lactose, microcrystalline cellulose, starch, corn starch, pregelatinized starch, maize starch, potato starch, powdered celluloses, sorbitol, xylitol, dibasic calcium phosphate, calcium phosphate, calcium carbonate, magnesium carbonate and the like, and combinations thereof.
Various disintegrants include but are not limited to croscarmellose sodium, polacrilin potassium, sodium starch glycolate, crospovidone, pregelatinized starch, and the like, and combinations thereof. Various glidants include but are not limited to colloidal silicon dioxide, magnesium silicate, magnesium trisilicate, talc, and other fonns of silicon dioxide, such as aggregated silicates and hydrated silica and the like, and combinations thereof.
Various lubricants include but are not limited to magnesium stearate, stearic acid, talc, fumaric acid, palmitic acid, sodium stearyl fumarate, carnauba wax, hydrogenated vegetable oils, mineral oil, polyethylene glycols and the like, and combinations thereof.
Another embodiment of the present invention describes process for preparing solid oral tablet comprising: (a) tablet core comprising 5% to 20% saxagliptin based on weight of the saxagliptin in the total composition, prepared by direct compression; (b) coating the tablet core of step (a) with a seal coating layer; (c) coating the tablets of step (b) with drug layer comprising 80% to 95% saxagliptin based on weight of the saxagliptin in the total composition; (d) coating the tablets of step (c) with a seal coating layer, and finally (e) coating the tablets of step (d) with a film coating layer.
The seal coating used according to the present invention is a hydroxypropyl methylcellulose based coating system. Solid dispersion of saxagliptin hydrochloride of the present invention is prepared as per the disclosure of unpublished provisional application No. IN 5537/CHE/2012 assigned to Hetero Research Foundation.
Solid dispersion of saxagliptin hydrochloride may be prepared by known process such as hot melt extrusion, fluid bed granulation, spray drying, melt agglomeration, co- precipitation, freeze drying, solvent evaporation, nitrogen stream, amorphous precipitation in crystalline matrix, supercritical fluid, eutectics or solid solution process. Tablet core of the present invention may be prepared either by direct compression or granulation process.
Direct compression process involves sifting and blending the saxagliptin and one or more excipients, followed by lubrication, and finally compressing into tablets. Granulation process involves sifting and blending saxagliptin and one or more excipients to form a dry mix, and then slugging/ compacting/ wet granulating the dry mix to form the desired granules, followed by lubrication and finally compressing into tablets.
Tablet core of the present invention further coated with a film coating composition. Film coating composition of the present invention comprises stearic acid in an amount of 1% to 35% by weight, preferably 5% to 30% by weight; macrocrystalline cellulose in an amount of 1 % to 20% by weight, preferably 5% to 15% by weight of the film coating composition; and hydroxypropyl methylcellulose.
Film coating composition used according to the present invention is available as SEPIFILM™ which is being marketed by SEPPIC.
Another embodiment of the present invention describes solid oral tablet comprising a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients and atleast one coating layer comprising stearic acid, microcrystalline cellulose and hydroxypropyl methylcellulose. The composition of the present invention is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus in multiple clinical settings.
EXAMPLES
The following examples further describe and demonstrate particular embodiments within the scope of the present invention. The examples are given solely for illustration and are not to be construed as limitations as many variations are possible without departing from spirit and scope of the invention.
Table 1 : Tablet composition of SaxagHptin hydrochloride:
Ingredients Example 1 (mg/tab)
Saxagliptin hydrochloride** 0.28
Lactose monohydrate 120.82
Microcrystalline cellulose 83.16
Croscarmellose sodium 9.00
Magnesium stearate 3.00
Inner seal coating
Opadry clear 4.00
Purified water q.s.
Drug coating
Saxagliptin hydrochloride** 5.30
Purified water q.s.
Outer seal coating.
Opadry clear 4.00
Purified water q.s.
Over film coating
SEPIFILM™ LP 007* 7.00
Purified water q.s.
5.58 mg of saxagliptin hydrochloride is equivalent to 5 mg of saxagliptin.
'SEPIFILM LP 007 contains hypromellose (60% - 70%), microcrystalline cellulose (5%-15%), stearic acid (20% - 30%).
Opadry clear is a hypromellose based coating system.
Preparation method:
1. Saxagliptin hydrochloride, lactose monohydrate, microcrystalline cellulose and croscarmeHose sodium were sifted and blended through mesh #40,
2. magnesium stearate was sifted through mesh #60,
3. blend of step 1, was lubricated with magnesium stearate of step 2, and compressed into tablets,
4. tablets of step 3 , were seal coated with Opadry clear,
5. seal coated tablets of step 4, were coated using drug, suspension,
6. tablets of step 5, were seal coated with Opadry clear, and finally
7. tablets of step 6, were over film coated with SEPIFILM™ LP 007.
Table 2: Tablet compositions of Saxagliptin hydrochloride:
Ingredients Example 2 (mg/tab) Example 3 (mg/tab)
Saxagliptin hydrochloride 5.58 5.58
Lactose monohydrate 199.26 199.26
Microcrystalline cellulose 83.16 83.16
CroscarmeHose sodium 9.00 9.00
Magnesium stearate 3.00 3.00
Film coating.
SEPIFILM™ LP 007* 9.00 -
SEPIFILM™ LP 010s - 9.00
Purified water q.s. q.s. "5.58 mg of saxagliptin hydrochloride is equivalent to 5 mg of saxagliptin.
'SEPIFILM LP 007 contains hypromellose (60% - 70%), microcrystalline cellulose (5%- 15%), stearic acid (20% - 30%).
$SEPIFILM LP 010 contains Hypromellose (75% - 85%), microcrystalline cellulose (5% - 15%), stearic acid (5% - 15%).
Preparation method:
1. Saxagliptin hydrochloride, lactose monohydrate, macrocrystalline cellulose and croscarmellose sodium were sifted and blended through mesh #40,
2. magnesium stearate was sifted through mesh #60,
3. blend of step 1 , was lubricated with magnesium stearate of step 2, and compressed into tablets, and finally
4. tablets of step 3, were coated with SEPIFILM™ LP.
Table 3 : Tablet composition of Saxagliptin hydrochloride: ngredients Example 3 (mg/tab)
Saxagliptin hydrochloride" 0.28
Lactose monohydrate 120.82
Microcrystalline cellulose 83.16
Croscarmellose sodium 9.00
Magnesium stearate 3.00
Inner seal coating
SEPIFILM™ LP 007* 4.00
Purified water q.s.
Drug coating
Saxagliptin hydrochloride" 5.30
Purified water q.s.
Over film coating
SEPIFILM™ LP 007* 7.00
Purified water q.s.
5.58 mg of saxagliptin hydrochloride is equivalent to 5 mg of saxagliptin.
*SEPIFILM LP 007 contains hypromellose (60% - 70%), microcrystalline cellulose (5%-15%), stearic acid (20% - 30%).
Preparation method:
1. Saxagliptin hydrochloride, lactose monohydrate, microcrystalline cellulose and croscarmellose sodium were sifted and blended through mesh #40,
2. magnesium stearate was sifted through mesh #60,
3. blend of step 1 ,. was lubricated with magnesium stearate of step 2, and compressed into tablets,
4. tablets of step 3 , were seal coated with SEPIFILM™ LP 007,
5. seal coated tablets of step 4, were coated using drug suspension, and finally
6. drug coated tablets of step 5, were over film coated with SEPIFILM™ LP 007.
Example 5:
Table 4: Tablet compositions of Saxagliptin hydrochloride solid dispersion:
Ingredients mg/tab
Saxagliptin HC1 (Solid dispersion)* 22.32
Lactose monohydrate 199.26
Microcrystalline cellulose 66.42
Croscarmellose sodium 9.00
Magnesium stearate 3.00
Film coating:
SEPIFILM™ LP* 9.00
Purified water q.s.
22.32 mg of solid dispersion of saxagliptin hydrochloride contains 5.58 mg of saxagliptin hydrochloride or 5 mg of saxagliptin.
SEPIFILM LP contains hypromellose, microcrystalline cellulose, stearic acid.
Preparation method:
1. Saxagliptin hydrochloride (Solid dispersion), lactose monohydrate, microcrystalline cellulose and croscarmellose sodium were sifted and blended through mesh #40,
2. magnesium stearate was sifted through mesh #60,
3. blend of step 1, was lubricated with magnesium stearate of step 2, and compressed into tablets, and finally
4. tablets of step 3, were coated with SEPIFILM™ LP.
Claims
1. A solid oral tablet composition comprising:
(a) a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients;
(b) an inner seal coating layer on the said tablet core;
(c) a drug layer comprising saxagliptin and one or more pharmaceutically acceptable excipients on the said seal coating;
(d) an outer seal coating, layer on the said drug layer; and
(e) an outer film coating layer on the said seal coating.
2. The composition according, to claim 1, wherein said, pharmaceutically acceptable excipient is selected from a diluent, a binder, a disiritegrant, a glidant, a lubricant and combinations thereof.
3. The composition of claim 1, wherein said core is prepared either by direct compression or granulation process.
4. A process for preparing solid oral tablet comprising:
(a) a tablet core comprising 5% to 20% saxagliptin based on weight of the saxagliptin in the total composition, prepared by direct compression;
(b) coating the tablet core of step (a) with a seal coating layer;
(c) coating the tablets of step (b) with drug, layer comprising 80% to 95% saxagliptin based on weight of the saxagliptin in the total composition;
(d) coating the tablets of step (c) with a seal coating layer, and finally
(e) coating the tablets of step (d) with a film coating layer.
5. The direct compression method according to the claim 4, comprises steps of
(a) sifting, and blending saxagliptin and one or more pharmaceutically acceptable excipients, (b) lubricating the blend of step (a) with a lubricant,
(c) compressing the lubricated blend of step (b) into tablets.
6. The film coating layer according to claim 1 and 4, comprises stearic acid, microcrystalline cellulose and hydroxypropyl methylcellulose.
7. A solid oral tablet comprising:
(a) a tablet core comprising saxagliptin as an active agent and one or more pharmaceutically acceptable excipients;
(b) atleast one coating layer comprising stearic acid, microcrystalline cellulose and hydroxypropyl methylcellulose.
8. The tablet according to any of the preceding claims, wherein said active agent saxagliptin is in the form of saxagliptin hydrochloride.
9. The composition according to any of the preceding claims is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus in multiple clinical settings.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN545CH2013 | 2013-02-08 | ||
| IN545/CHE/2013 | 2013-02-08 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2014122671A2 true WO2014122671A2 (en) | 2014-08-14 |
| WO2014122671A3 WO2014122671A3 (en) | 2014-12-24 |
Family
ID=51300224
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2014/000081 Ceased WO2014122671A2 (en) | 2013-02-08 | 2014-02-06 | Solid oral compositions of saxagliptin |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2014122671A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015071887A1 (en) * | 2013-11-18 | 2015-05-21 | Ranbaxy Laboratories Limited | Oral pharmaceutical compositions of saxagliptin |
| WO2015071889A1 (en) * | 2013-11-18 | 2015-05-21 | Ranbaxy Laboratories Limited | Oral compositions of saxagliptin |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4796776A (en) | 1987-02-19 | 1989-01-10 | Northland Aluminum Products, Inc. | Pressure cooker for microwave ovens |
| US4853509A (en) | 1987-11-18 | 1989-08-01 | Hario Kabushiki Kaisha | Rice cooker for microwave ranges |
| US5229563A (en) | 1990-01-11 | 1993-07-20 | Matsushita Electric Industrial Co., Ltd. | Pressure cooker |
| US5310981A (en) | 1993-07-23 | 1994-05-10 | Ensar Corporation | Microwave cooker |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7985422B2 (en) * | 2002-08-05 | 2011-07-26 | Torrent Pharmaceuticals Limited | Dosage form |
| US20070172525A1 (en) * | 2007-03-15 | 2007-07-26 | Ramesh Sesha | Anti-diabetic combinations |
| US8551524B2 (en) * | 2008-03-14 | 2013-10-08 | Iycus, Llc | Anti-diabetic combinations |
| PE20140960A1 (en) * | 2008-04-03 | 2014-08-15 | Boehringer Ingelheim Int | FORMULATIONS INVOLVING A DPP4 INHIBITOR |
| AU2011295837B2 (en) * | 2010-09-03 | 2015-06-18 | Astrazeneca Uk Limited | Drug formulations using water soluble antioxidants |
-
2014
- 2014-02-06 WO PCT/IN2014/000081 patent/WO2014122671A2/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4796776A (en) | 1987-02-19 | 1989-01-10 | Northland Aluminum Products, Inc. | Pressure cooker for microwave ovens |
| US4853509A (en) | 1987-11-18 | 1989-08-01 | Hario Kabushiki Kaisha | Rice cooker for microwave ranges |
| US5229563A (en) | 1990-01-11 | 1993-07-20 | Matsushita Electric Industrial Co., Ltd. | Pressure cooker |
| US5310981A (en) | 1993-07-23 | 1994-05-10 | Ensar Corporation | Microwave cooker |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015071887A1 (en) * | 2013-11-18 | 2015-05-21 | Ranbaxy Laboratories Limited | Oral pharmaceutical compositions of saxagliptin |
| WO2015071889A1 (en) * | 2013-11-18 | 2015-05-21 | Ranbaxy Laboratories Limited | Oral compositions of saxagliptin |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2014122671A3 (en) | 2014-12-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5285105B2 (en) | Pharmaceutical composition | |
| US9770437B2 (en) | Compositions of eltrombopag | |
| US20110136883A1 (en) | Granulation of active pharmaceutical ingredients | |
| CN113939289A (en) | Oral solid tablet containing Bruton's tyrosine kinase inhibitor and preparation method thereof | |
| US9345712B2 (en) | Solid oral compositions of tolvaptan | |
| WO2019073331A2 (en) | Pharmaceutical compositions of apremilast | |
| JP2013502452A (en) | Otyronium direct compression tablets | |
| EP2620140A1 (en) | Crizotinib containing compositions | |
| JP2018177657A (en) | Levetiracetam-containing pharmaceutical composition and method for producing the same | |
| AU2016231883B2 (en) | Pharmaceutical compositions of dimethyl fumarate | |
| JP6461142B2 (en) | Anti-tuberculosis stable pharmaceutical composition in the form of a coated tablet containing isoniazid granules and rifapentine granules, and a process for producing the same | |
| EP2340834A1 (en) | Enhanced Solubility of Ziprasidone | |
| WO2014122671A2 (en) | Solid oral compositions of saxagliptin | |
| JP2010202579A (en) | Acarbose-containing disintegrating preparation in oral cavity | |
| WO2014125504A2 (en) | Pharmaceutical compositions of febuxostat | |
| JP2014118380A (en) | Benzimidazole-7-carboxylic acid derivative comprising tablet composition | |
| CN112057427B (en) | Oral solid tablet containing bruton's tyrosine kinase inhibitor and preparation method thereof | |
| WO2008053295A2 (en) | Pharmaceutical compositions of benzoquinolizine-2-carboxylic acid | |
| JP2017081859A (en) | Methods for producing telmisartan-containing tablets | |
| JP5910311B2 (en) | Pharmaceutical tablet and method for producing the same | |
| JP2007063217A (en) | Tablet containing pranlukast hydrate and method for producing the same | |
| JP6336651B1 (en) | Tablets containing esomeprazole salts with improved chemical stability | |
| EP3576735A1 (en) | Pharmaceutical composition of everolimus | |
| WO2016080357A1 (en) | Method for producing orally disintegrating tablets | |
| WO2015037017A2 (en) | Pharmaceutical compositions of roflumilast and process for preparation thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WA | Withdrawal of international application | ||
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14749121 Country of ref document: EP Kind code of ref document: A2 |