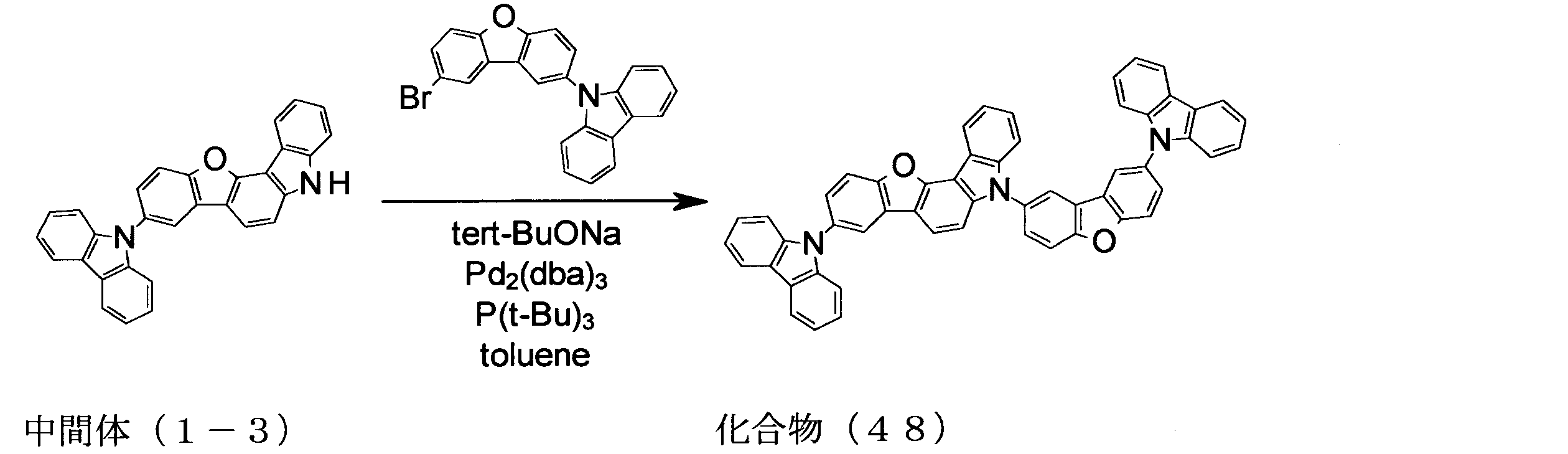
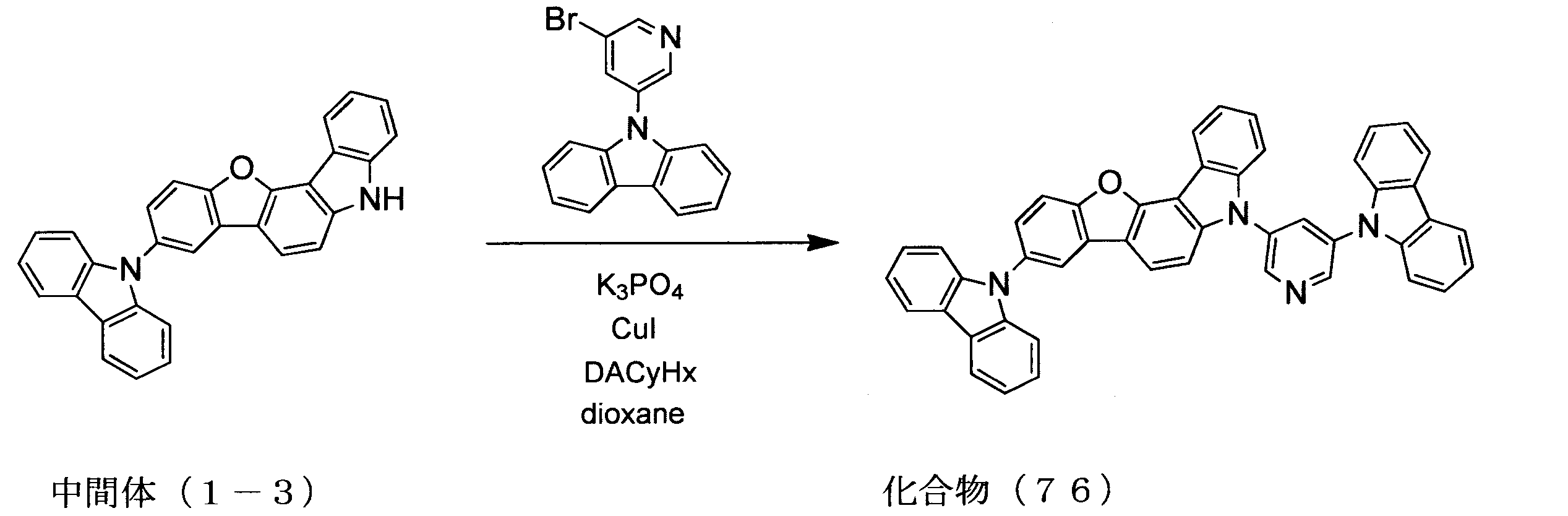
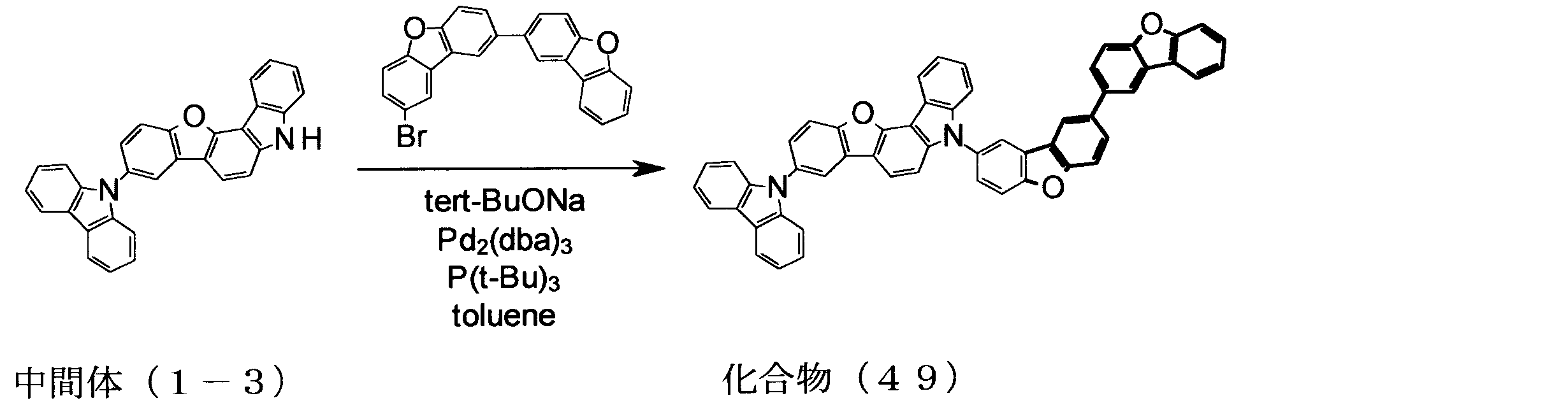
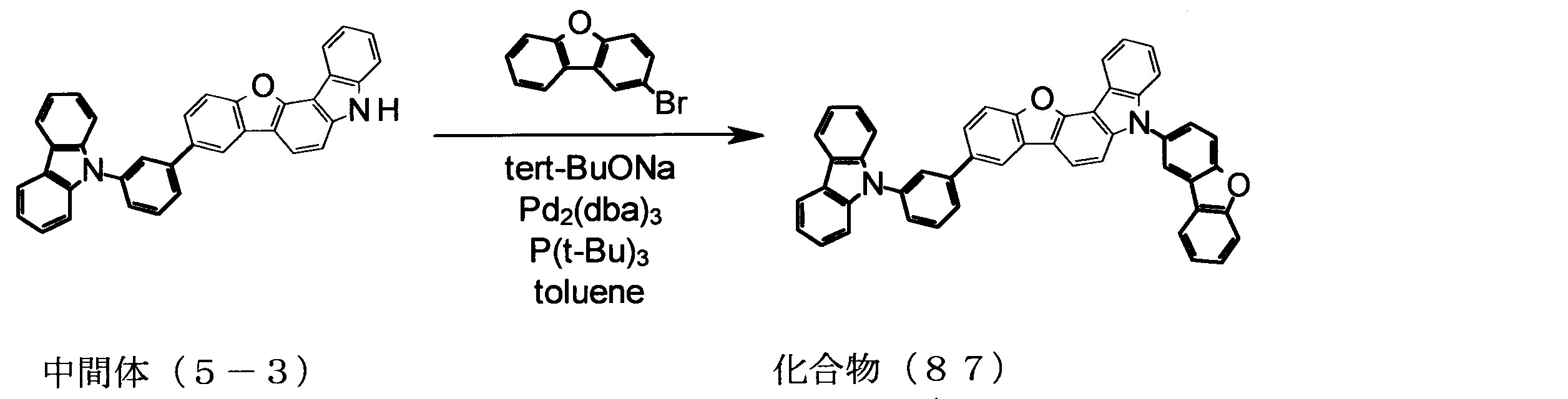
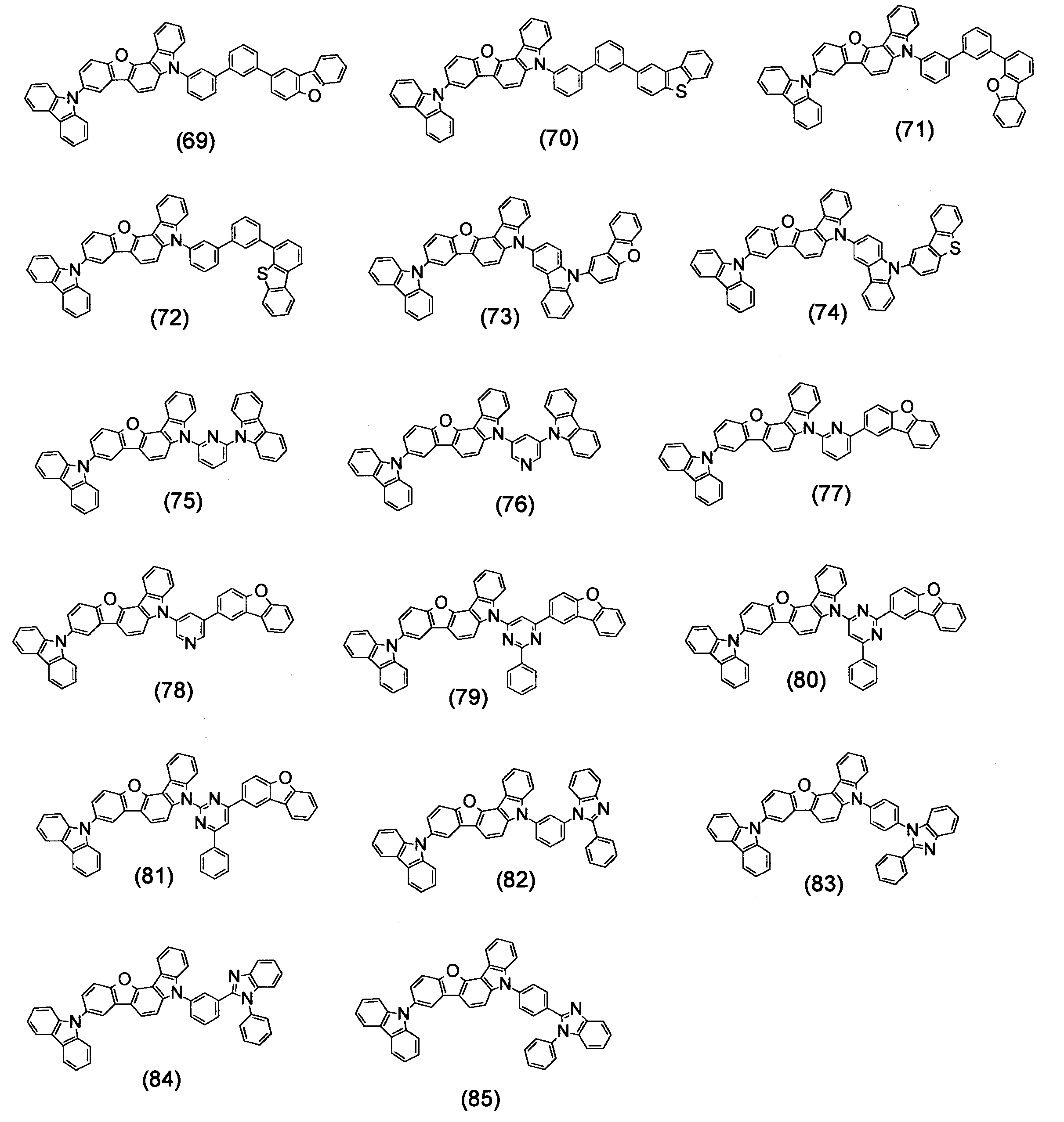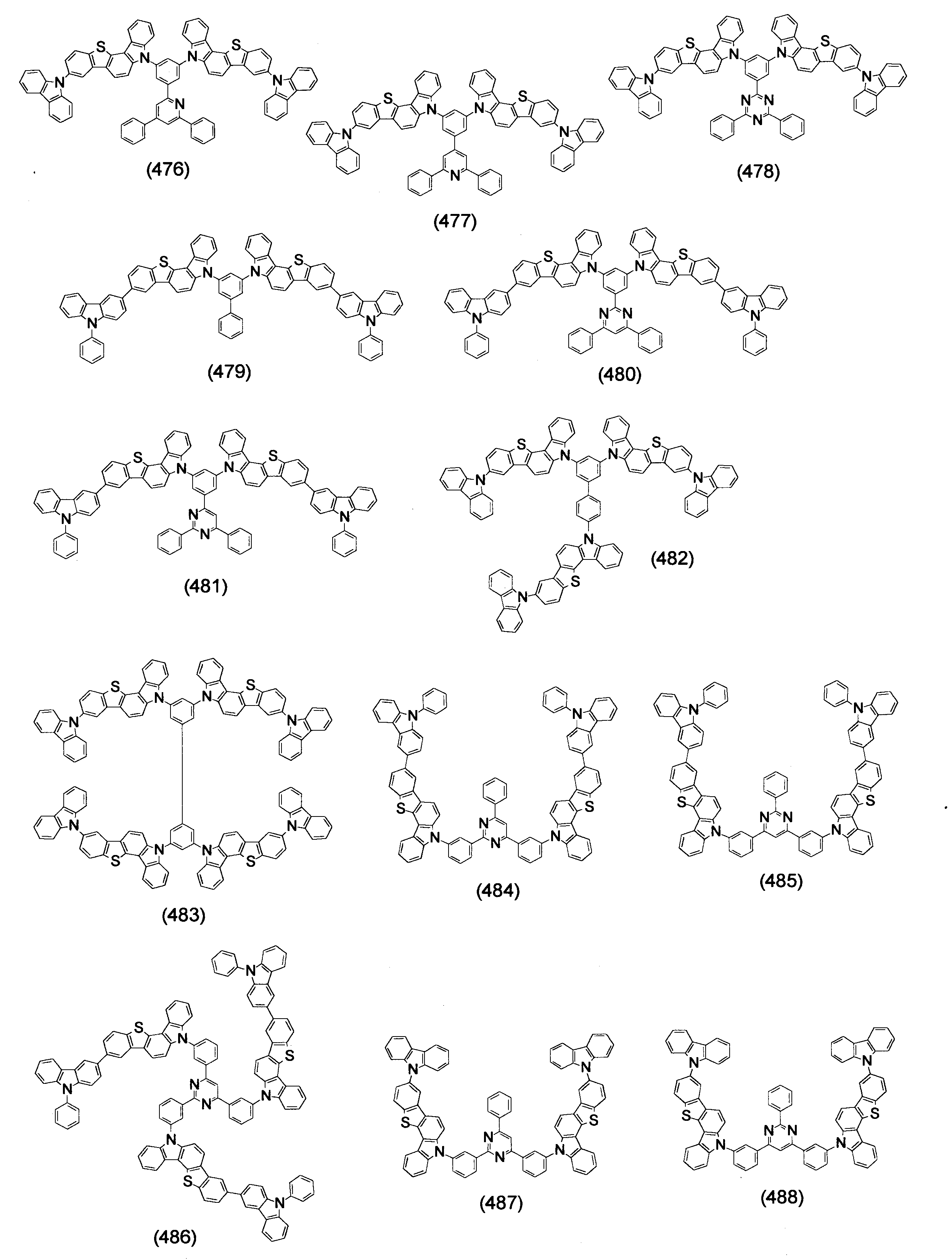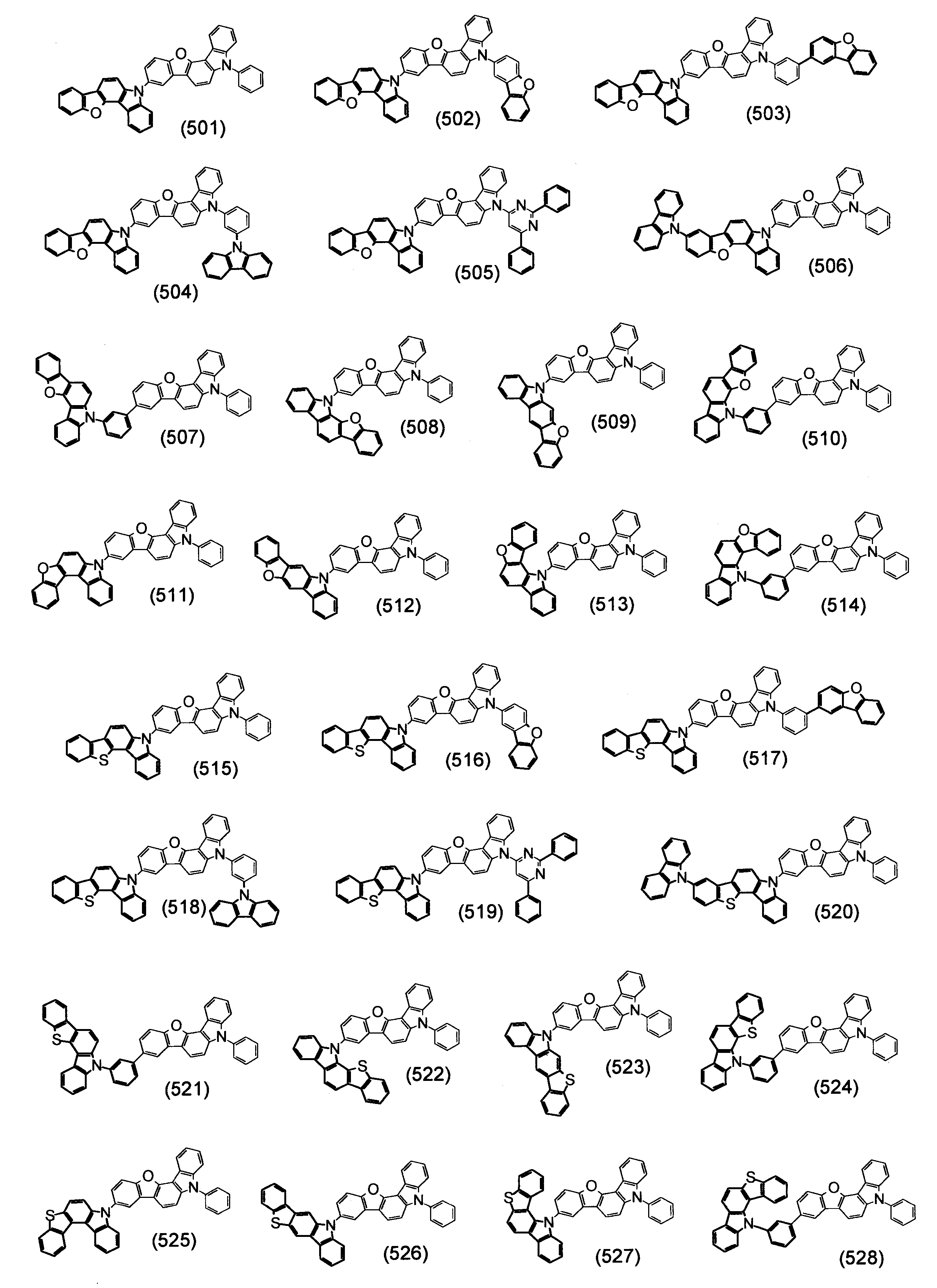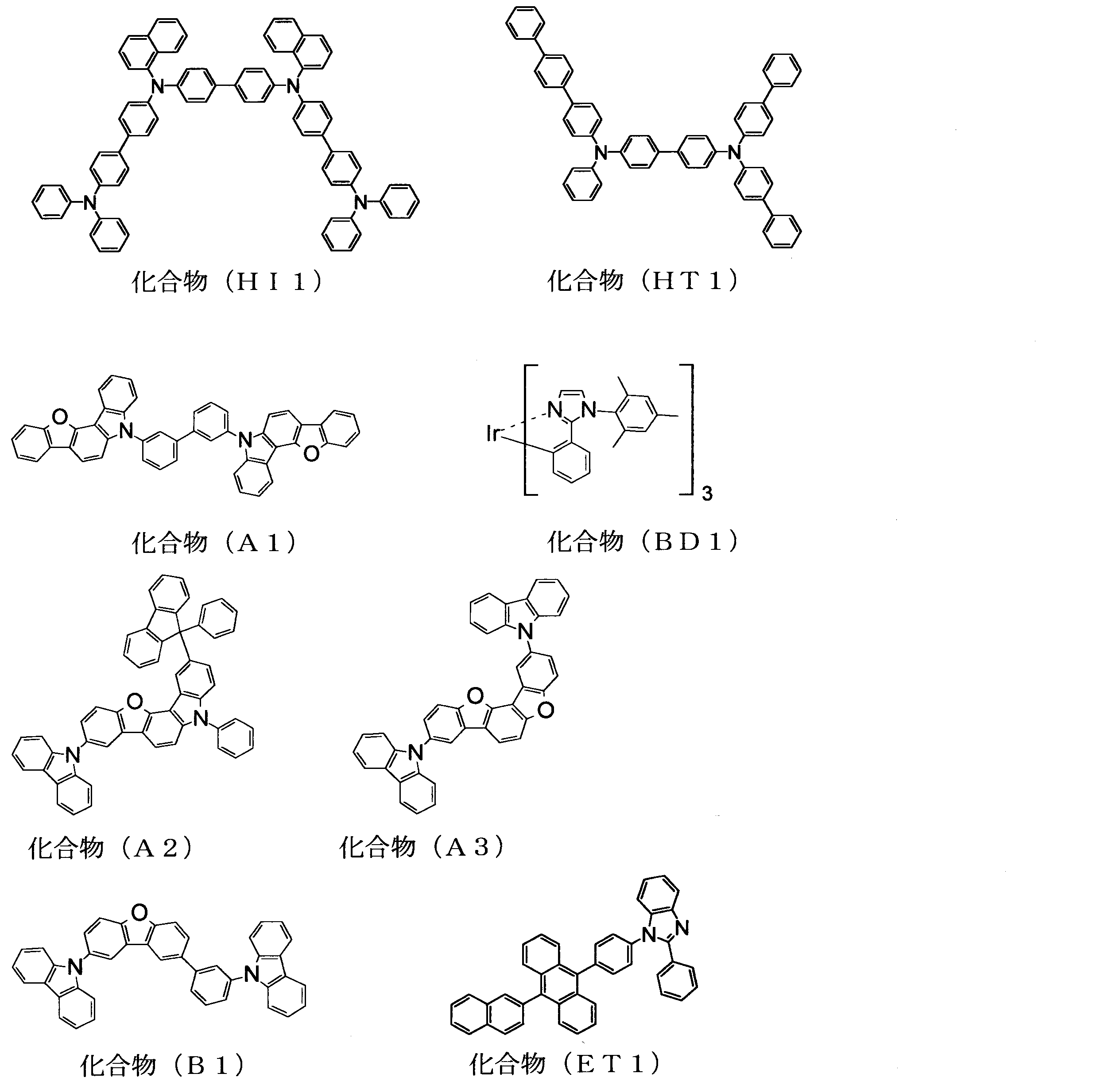WO2013179645A1 - Organic-electroluminescent-element material, and organic electroluminescent element using same - Google Patents
Organic-electroluminescent-element material, and organic electroluminescent element using same Download PDFInfo
- Publication number
- WO2013179645A1 WO2013179645A1 PCT/JP2013/003353 JP2013003353W WO2013179645A1 WO 2013179645 A1 WO2013179645 A1 WO 2013179645A1 JP 2013003353 W JP2013003353 W JP 2013003353W WO 2013179645 A1 WO2013179645 A1 WO 2013179645A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituent
- carbon atoms
- ring
- organic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C(c1ccccc1)=*N Chemical compound *C(c1ccccc1)=*N 0.000 description 17
- SRDPSXHOZNJYMR-UHFFFAOYSA-N c(cc12)ccc1[nH]c(cc1)c2c2c1c1cc(-[n]3c4ccccc4c4ccccc34)ccc1[o]2 Chemical compound c(cc12)ccc1[nH]c(cc1)c2c2c1c1cc(-[n]3c4ccccc4c4ccccc34)ccc1[o]2 SRDPSXHOZNJYMR-UHFFFAOYSA-N 0.000 description 3
- DEWXEFFEEWANBT-UHFFFAOYSA-N c(cc1)ccc1-[n]1c(ccc(-c2ccc3[o]c(c4c(cc5)[nH]c6ccccc46)c5c3c2)c2)c2c2c1cccc2 Chemical compound c(cc1)ccc1-[n]1c(ccc(-c2ccc3[o]c(c4c(cc5)[nH]c6ccccc46)c5c3c2)c2)c2c2c1cccc2 DEWXEFFEEWANBT-UHFFFAOYSA-N 0.000 description 2
- IZSFTGLPJGJPOU-UHFFFAOYSA-N c(cc12)ccc1[nH]c(cc1)c2c2c1c1cc(-c(cc3)cc4c3[o]c(cc3)c4cc3-[n]3c4ccccc4c4c3cccc4)ccc1[o]2 Chemical compound c(cc12)ccc1[nH]c(cc1)c2c2c1c1cc(-c(cc3)cc4c3[o]c(cc3)c4cc3-[n]3c4ccccc4c4c3cccc4)ccc1[o]2 IZSFTGLPJGJPOU-UHFFFAOYSA-N 0.000 description 2
- DRSLRZFMVQKPOI-UHFFFAOYSA-N c(cc12)ccc1[nH]c(cc1)c2c2c1c1cc(-c3cccc(-[n]4c(cccc5)c5c5ccccc45)c3)ccc1[o]2 Chemical compound c(cc12)ccc1[nH]c(cc1)c2c2c1c1cc(-c3cccc(-[n]4c(cccc5)c5c5ccccc45)c3)ccc1[o]2 DRSLRZFMVQKPOI-UHFFFAOYSA-N 0.000 description 2
- DNAWRPMGUMOFCM-ITWBFMHDSA-N C(CC1)CCC1N1C(CC2S[C@@H](CCCC3)C3C2C2)C2C(C2)C1CCC2c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3ccccc3)c4c2c1 Chemical compound C(CC1)CCC1N1C(CC2S[C@@H](CCCC3)C3C2C2)C2C(C2)C1CCC2c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3ccccc3)c4c2c1 DNAWRPMGUMOFCM-ITWBFMHDSA-N 0.000 description 1
- KBDQFOZVAWOOJQ-UHFFFAOYSA-N C[N]1(C2C3OC(CCCC4)C4C3CCC2C(C2)C1CC2c1ccc2[o]c(c(c3c4C=CCC3)c(cc3)[n]4-c4ccccc4)c3c2c1)C1CCCCC1 Chemical compound C[N]1(C2C3OC(CCCC4)C4C3CCC2C(C2)C1CC2c1ccc2[o]c(c(c3c4C=CCC3)c(cc3)[n]4-c4ccccc4)c3c2c1)C1CCCCC1 KBDQFOZVAWOOJQ-UHFFFAOYSA-N 0.000 description 1
- HEDCISGRKNYEFA-UHFFFAOYSA-N Clc(cc(cc1)-[n]2c(ccc(-c(cc3)cc(c4ccccc44)c3[n]4-c3ccccc3)c3)c3c3ccccc23)c1Oc1cccc2c1c1ccccc1[n]2-c1ccccc1 Chemical compound Clc(cc(cc1)-[n]2c(ccc(-c(cc3)cc(c4ccccc44)c3[n]4-c3ccccc3)c3)c3c3ccccc23)c1Oc1cccc2c1c1ccccc1[n]2-c1ccccc1 HEDCISGRKNYEFA-UHFFFAOYSA-N 0.000 description 1
- LWOVHKYUXSSKCV-UHFFFAOYSA-N OC(CCCC1)C1C(CCC1C(C2)C3CCC2c2ccc4[o]c5c(c6ccccc6[n]6-c7cc(N8C(CCCC9)C9C9C8CCCC9)ccc7)c6ccc5c4c2)CC1N3C1CCCCC1 Chemical compound OC(CCCC1)C1C(CCC1C(C2)C3CCC2c2ccc4[o]c5c(c6ccccc6[n]6-c7cc(N8C(CCCC9)C9C9C8CCCC9)ccc7)c6ccc5c4c2)CC1N3C1CCCCC1 LWOVHKYUXSSKCV-UHFFFAOYSA-N 0.000 description 1
- XECWTHNHGINCCX-UHFFFAOYSA-N c(cc1)cc(c2c3[o]c(cccc4)c4c3ccc22)c1[n]2-c1cccc(-c2cc(-[n]3c4ccc(c(cccc5)c5[o]5)c5c4c4c3cccc4)ccc2)c1 Chemical compound c(cc1)cc(c2c3[o]c(cccc4)c4c3ccc22)c1[n]2-c1cccc(-c2cc(-[n]3c4ccc(c(cccc5)c5[o]5)c5c4c4c3cccc4)ccc2)c1 XECWTHNHGINCCX-UHFFFAOYSA-N 0.000 description 1
- UNUZDDUWJNUCMF-UHFFFAOYSA-N c(cc1)cc(c2c3cccc2)c1[n]3-c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3cc(-c5cncc(-[n](c6c7cccc6)c(cc6)c7c7c6c6cc(-[n]8c9ccccc9c9c8cccc9)ccc6[o]7)c5)cnc3)c4c2c1 Chemical compound c(cc1)cc(c2c3cccc2)c1[n]3-c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3cc(-c5cncc(-[n](c6c7cccc6)c(cc6)c7c7c6c6cc(-[n]8c9ccccc9c9c8cccc9)ccc6[o]7)c5)cnc3)c4c2c1 UNUZDDUWJNUCMF-UHFFFAOYSA-N 0.000 description 1
- BMBPOZIXIUELOE-UHFFFAOYSA-N c(cc1)cc(c2c3cccc2)c1[n]3-c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3nc(-c5cccc(-[n](c6ccccc66)c(cc7)c6c6c7c(cc(cc7)-[n]8c9ccccc9c9c8cccc9)c7[o]6)n5)ccc3)c4c2c1 Chemical compound c(cc1)cc(c2c3cccc2)c1[n]3-c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3nc(-c5cccc(-[n](c6ccccc66)c(cc7)c6c6c7c(cc(cc7)-[n]8c9ccccc9c9c8cccc9)c7[o]6)n5)ccc3)c4c2c1 BMBPOZIXIUELOE-UHFFFAOYSA-N 0.000 description 1
- QAMSEMJAFIJBOI-UHFFFAOYSA-N c(cc1)cc(c2c3cccc2)c1[n]3-c1ccc2[s]c(c(c3c4cccc3)c(cc3)[n]4-c4cc(-c5ccc6[o]c(cccc7)c7c6c5)cnc4)c3c2c1 Chemical compound c(cc1)cc(c2c3cccc2)c1[n]3-c1ccc2[s]c(c(c3c4cccc3)c(cc3)[n]4-c4cc(-c5ccc6[o]c(cccc7)c7c6c5)cnc4)c3c2c1 QAMSEMJAFIJBOI-UHFFFAOYSA-N 0.000 description 1
- BHDKGJHLCKRHAA-UHFFFAOYSA-N c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc(c2c3)c1[o]c2ccc3-c1cccc(-[n]2c(cccc3)c3c3c2cccc3)c1 Chemical compound c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc(c2c3)c1[o]c2ccc3-c1cccc(-[n]2c(cccc3)c3c3c2cccc3)c1 BHDKGJHLCKRHAA-UHFFFAOYSA-N 0.000 description 1
- HKGWNPLRLTXSLU-UHFFFAOYSA-N c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c(cccc1)c1[n]2-c1cc(-[n]2c(cccc3)c3c3c2cccc3)cnc1 Chemical compound c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c(cccc1)c1[n]2-c1cc(-[n]2c(cccc3)c3c3c2cccc3)cnc1 HKGWNPLRLTXSLU-UHFFFAOYSA-N 0.000 description 1
- HLHJKHXLHARXBJ-UHFFFAOYSA-N c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-c2cc(-c3c4[s]c(cccc5)c5c4ccc3)ccc2)c1 Chemical compound c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-c2cc(-c3c4[s]c(cccc5)c5c4ccc3)ccc2)c1 HLHJKHXLHARXBJ-UHFFFAOYSA-N 0.000 description 1
- RAWGLPRRNSVDLO-UHFFFAOYSA-N c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-c2cccc(-c(cc3)cc4c3[o]c3c4cccc3)c2)c1 Chemical compound c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-c2cccc(-c(cc3)cc4c3[o]c3c4cccc3)c2)c1 RAWGLPRRNSVDLO-UHFFFAOYSA-N 0.000 description 1
- QSPJJNFQQUEPKW-UHFFFAOYSA-N c(cc1)cc(c2ccccc22)c1[n]2-c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3cc(-c5cc(-[n](c6c7cccc6)c(cc6)c7c7c6c(cc(cc6)-[n]8c9ccccc9c9ccccc89)c6[o]7)ccn5)ncc3)c4c2c1 Chemical compound c(cc1)cc(c2ccccc22)c1[n]2-c1ccc2[o]c(c(c3ccccc33)c(cc4)[n]3-c3cc(-c5cc(-[n](c6c7cccc6)c(cc6)c7c7c6c(cc(cc6)-[n]8c9ccccc9c9ccccc89)c6[o]7)ccn5)ncc3)c4c2c1 QSPJJNFQQUEPKW-UHFFFAOYSA-N 0.000 description 1
- IBLGZEQALXKIIF-UHFFFAOYSA-N c(cc1)ccc1-[n](c(cccc1)c1c1c2)c1ccc2-c(cc1c2c3cccc2)ccc1[n]3-c(cc1)cc2c1[o]c1c2ccc2c1c1ccccc1[n]2-c1ccccc1 Chemical compound c(cc1)ccc1-[n](c(cccc1)c1c1c2)c1ccc2-c(cc1c2c3cccc2)ccc1[n]3-c(cc1)cc2c1[o]c1c2ccc2c1c1ccccc1[n]2-c1ccccc1 IBLGZEQALXKIIF-UHFFFAOYSA-N 0.000 description 1
- GKTLHQFSIDFAJH-UHFFFAOYSA-N c(cc1)ccc1-[n](c(cccc1)c1c1c2)c1ccc2-c1ccc2[nH]c(cccc3)c3c2c1 Chemical compound c(cc1)ccc1-[n](c(cccc1)c1c1c2)c1ccc2-c1ccc2[nH]c(cccc3)c3c2c1 GKTLHQFSIDFAJH-UHFFFAOYSA-N 0.000 description 1
- RCZFKEATVLLOQL-UHFFFAOYSA-N c(cc1)ccc1-[n]1c(-c(cc2)ccc2-[n]2c3ccc(c(cc(cc4)-[n]5c6ccccc6c6c5cccc6)c4[s]4)c4c3c3c2cccc3)nc2c1cccc2 Chemical compound c(cc1)ccc1-[n]1c(-c(cc2)ccc2-[n]2c3ccc(c(cc(cc4)-[n]5c6ccccc6c6c5cccc6)c4[s]4)c4c3c3c2cccc3)nc2c1cccc2 RCZFKEATVLLOQL-UHFFFAOYSA-N 0.000 description 1
- MLENWNSSDYIQSZ-UHFFFAOYSA-N c(cc1)ccc1-[n]1c(-c2cc(-[n](c3c4cccc3)c(cc3)c4c4c3c(cc(cc3)-[n]5c6ccccc6c6c5cccc6)c3[s]4)ccc2)nc2c1cccc2 Chemical compound c(cc1)ccc1-[n]1c(-c2cc(-[n](c3c4cccc3)c(cc3)c4c4c3c(cc(cc3)-[n]5c6ccccc6c6c5cccc6)c3[s]4)ccc2)nc2c1cccc2 MLENWNSSDYIQSZ-UHFFFAOYSA-N 0.000 description 1
- CVOMHZLJPJYUGQ-UHFFFAOYSA-N c(cc1)ccc1-c1nc(-c2cc(-[n](c3c4cccc3)c(cc3)c4c4c3c3cc(-[n]5c6ccccc6c6c5cccc6)ccc3[o]4)ccc2)nc(-c2cc(-[n](c3c4cccc3)c(cc3)c4c4c3c(cc(cc3)-[n]5c6ccccc6c6ccccc56)c3[o]4)ccc2)n1 Chemical compound c(cc1)ccc1-c1nc(-c2cc(-[n](c3c4cccc3)c(cc3)c4c4c3c3cc(-[n]5c6ccccc6c6c5cccc6)ccc3[o]4)ccc2)nc(-c2cc(-[n](c3c4cccc3)c(cc3)c4c4c3c(cc(cc3)-[n]5c6ccccc6c6ccccc56)c3[o]4)ccc2)n1 CVOMHZLJPJYUGQ-UHFFFAOYSA-N 0.000 description 1
- PWGCYDSZVQSUKM-UHFFFAOYSA-N c(cc1)ccc1-c1nc(-c2cc(-[n]3c4ccc(c5cc(-c6cc(-[n]7c8ccccc8c8c7cccc8)ccc6)ccc5[o]5)c5c4c4c3cccc4)ccc2)nc(-c2cccc(-[n]3c4ccc(c5cc(-c6cccc(-[n]7c(cccc8)c8c8ccccc78)c6)ccc5[o]5)c5c4c4ccccc34)c2)n1 Chemical compound c(cc1)ccc1-c1nc(-c2cc(-[n]3c4ccc(c5cc(-c6cc(-[n]7c8ccccc8c8c7cccc8)ccc6)ccc5[o]5)c5c4c4c3cccc4)ccc2)nc(-c2cccc(-[n]3c4ccc(c5cc(-c6cccc(-[n]7c(cccc8)c8c8ccccc78)c6)ccc5[o]5)c5c4c4ccccc34)c2)n1 PWGCYDSZVQSUKM-UHFFFAOYSA-N 0.000 description 1
- YBIPOYJJSFYAEX-UHFFFAOYSA-N c(cc1)ccc1-c1nc(-c2cccc(-[n](c3c4cccc3)c(cc3)c4c4c3c(cc(cc3)-[n]5c6ccccc6c6c5cccc6)c3[o]4)c2)nc(-c2cc(-[n](c3ccccc33)c(cc4)c3c3c4c(cc(cc4)-[n]5c6ccccc6c6ccccc56)c4[o]3)ccc2)c1 Chemical compound c(cc1)ccc1-c1nc(-c2cccc(-[n](c3c4cccc3)c(cc3)c4c4c3c(cc(cc3)-[n]5c6ccccc6c6c5cccc6)c3[o]4)c2)nc(-c2cc(-[n](c3ccccc33)c(cc4)c3c3c4c(cc(cc4)-[n]5c6ccccc6c6ccccc56)c4[o]3)ccc2)c1 YBIPOYJJSFYAEX-UHFFFAOYSA-N 0.000 description 1
- OSNKQBFOAFYRNE-UHFFFAOYSA-N c(cc1)ccc1-c1nc(-c2cccc(-[n](c3c4cccc3)c(cc3)c4c4c3c3cc(-c(cc5)cc(c6ccccc66)c5[n]6-c5ccccc5)ccc3[o]4)c2)nc(-c2cc(-[n](c3ccccc33)c(cc4)c3c3c4c4cc(-c(cc5)cc(c6ccccc66)c5[n]6-c5ccccc5)ccc4[o]3)ccc2)n1 Chemical compound c(cc1)ccc1-c1nc(-c2cccc(-[n](c3c4cccc3)c(cc3)c4c4c3c3cc(-c(cc5)cc(c6ccccc66)c5[n]6-c5ccccc5)ccc3[o]4)c2)nc(-c2cc(-[n](c3ccccc33)c(cc4)c3c3c4c4cc(-c(cc5)cc(c6ccccc66)c5[n]6-c5ccccc5)ccc4[o]3)ccc2)n1 OSNKQBFOAFYRNE-UHFFFAOYSA-N 0.000 description 1
- PHBJYIUTTPNUBD-UHFFFAOYSA-N c(cc1)ccc1-c1nc(cccc2)c2[n]1-c(cc1)ccc1-c1c(cccc2)c2c(-c2ccc(cccc3)c3c2)c2c1cccc2 Chemical compound c(cc1)ccc1-c1nc(cccc2)c2[n]1-c(cc1)ccc1-c1c(cccc2)c2c(-c2ccc(cccc3)c3c2)c2c1cccc2 PHBJYIUTTPNUBD-UHFFFAOYSA-N 0.000 description 1
- AWFIGJMYCBEFAK-UHFFFAOYSA-N c(cc1)ccc1-c1nc(cccc2)c2[n]1-c1cc(-[n](c2c3cccc2)c(cc2)c3c3c2c(cc(cc2)-[n]4c5ccccc5c5ccccc45)c2[s]3)ccc1 Chemical compound c(cc1)ccc1-c1nc(cccc2)c2[n]1-c1cc(-[n](c2c3cccc2)c(cc2)c3c3c2c(cc(cc2)-[n]4c5ccccc5c5ccccc45)c2[s]3)ccc1 AWFIGJMYCBEFAK-UHFFFAOYSA-N 0.000 description 1
- PMBIBMLVAFPBRF-UHFFFAOYSA-N c(cc1c2c3cccc2)ccc1[n]3-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1ccccc21 Chemical compound c(cc1c2c3cccc2)ccc1[n]3-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c(cc1)cc(c2ccccc22)c1[n]2-c(cc1)cc2c1[s]c1ccccc21 PMBIBMLVAFPBRF-UHFFFAOYSA-N 0.000 description 1
- FMSUXKJEHWJLCO-UHFFFAOYSA-N c(cc1c2ccccc22)ccc1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c(cccc1)c1[n]2-c1cccc(-c2ccc3[o]c(cccc4)c4c3c2)n1 Chemical compound c(cc1c2ccccc22)ccc1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c(cccc1)c1[n]2-c1cccc(-c2ccc3[o]c(cccc4)c4c3c2)n1 FMSUXKJEHWJLCO-UHFFFAOYSA-N 0.000 description 1
- AWKMIUWTYUHGPL-UHFFFAOYSA-N c(cc1c2ccccc22)ccc1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-[n]2c3ccccc3c3c2cccc3)n1 Chemical compound c(cc1c2ccccc22)ccc1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-[n]2c3ccccc3c3c2cccc3)n1 AWKMIUWTYUHGPL-UHFFFAOYSA-N 0.000 description 1
- QFMIIEJRIOLIMY-UHFFFAOYSA-N c(cc1c2ccccc22)ccc1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-c2cc(-c3c4[o]c5ccccc5c4ccc3)ccc2)c1 Chemical compound c(cc1c2ccccc22)ccc1[n]2-c(cc1)cc2c1[s]c1c2ccc2c1c1ccccc1[n]2-c1cccc(-c2cc(-c3c4[o]c5ccccc5c4ccc3)ccc2)c1 QFMIIEJRIOLIMY-UHFFFAOYSA-N 0.000 description 1
- FJSNERGLWYYLLA-UHFFFAOYSA-N c(cc1c2ccccc22)ccc1[n]2-c1ccc2[o]c(c(c3c4)c(cc5)[o]c3ccc4-[n]3c4ccccc4c4c3cccc4)c5c2c1 Chemical compound c(cc1c2ccccc22)ccc1[n]2-c1ccc2[o]c(c(c3c4)c(cc5)[o]c3ccc4-[n]3c4ccccc4c4c3cccc4)c5c2c1 FJSNERGLWYYLLA-UHFFFAOYSA-N 0.000 description 1
- IRTYOMSQCFQCHN-UHFFFAOYSA-N c(cc1c2ccccc22)ccc1[n]2-c1ccc2[s]c(c(c3ccccc33)c(cc4)[n]3-c3cccc(-c5cccc(-c(cc6)cc7c6[s]c6ccccc76)c5)c3)c4c2c1 Chemical compound c(cc1c2ccccc22)ccc1[n]2-c1ccc2[s]c(c(c3ccccc33)c(cc4)[n]3-c3cccc(-c5cccc(-c(cc6)cc7c6[s]c6ccccc76)c5)c3)c4c2c1 IRTYOMSQCFQCHN-UHFFFAOYSA-N 0.000 description 1
- UXFQLKOXLVKNEZ-UHFFFAOYSA-N c1ccc(C2(c3ccccc3-c3ccccc23)c(cc2c3c4ccc5c3[o]c(cc3)c5cc3-[n]3c5ccccc5c5ccccc35)ccc2[n]4-c2ccccc2)cc1 Chemical compound c1ccc(C2(c3ccccc3-c3ccccc23)c(cc2c3c4ccc5c3[o]c(cc3)c5cc3-[n]3c5ccccc5c5ccccc35)ccc2[n]4-c2ccccc2)cc1 UXFQLKOXLVKNEZ-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/04—Ortho-condensed systems
- C07D491/044—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring
- C07D491/048—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring the oxygen-containing ring being five-membered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1088—Heterocyclic compounds characterised by ligands containing oxygen as the only heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1092—Heterocyclic compounds characterised by ligands containing sulfur as the only heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
- H10K50/12—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers comprising dopants
Definitions
- the present invention relates to a material for an organic electroluminescence element and an organic electroluminescence element using the same.
- Organic electroluminescence (EL) elements include a fluorescent type and a phosphorescent type, and an optimum element design has been studied according to each light emission mechanism. With respect to phosphorescent organic EL elements, it is known from their light emission characteristics that high-performance elements cannot be obtained by simple diversion of fluorescent element technology. The reason is generally considered as follows. First, since phosphorescence emission is emission using triplet excitons, the energy gap of the compound used for the light emitting layer must be large. This is because the value of the energy gap (hereinafter also referred to as singlet energy) of a compound usually refers to the triplet energy of the compound (in the present invention, the energy difference between the lowest excited triplet state and the ground state). This is because it is larger than the value of).
- a host material having a triplet energy larger than the triplet energy of the phosphorescent dopant material must first be used for the light emitting layer. I must. Furthermore, an electron transport layer and a hole transport layer adjacent to the light emitting layer are provided, and a compound having a triplet energy higher than that of the phosphorescent dopant material must be used for the electron transport layer and the hole transport layer.
- a compound having a larger energy gap than the compound used for the fluorescent organic EL element is used for the phosphorescent organic EL element. The drive voltage of the entire element increases.
- hydrocarbon compounds having high oxidation resistance and reduction resistance useful for fluorescent elements have a large energy gap due to the large spread of ⁇ electron clouds. Therefore, in a phosphorescent organic EL element, it is difficult to select such a hydrocarbon compound, and an organic compound containing a heteroatom such as oxygen or nitrogen is selected. As a result, the phosphorescent organic EL element is There is a problem that the lifetime is shorter than that of a fluorescent organic EL element.
- the exciton relaxation rate of the triplet exciton of the phosphorescent dopant material is much longer than that of the singlet exciton also greatly affects the device performance. That is, since light emitted from singlet excitons has a high relaxation rate that leads to light emission, it is difficult for excitons to diffuse into the peripheral layer of the light emitting layer (for example, a hole transport layer or an electron transport layer). Light emission is expected. On the other hand, light emission from triplet excitons is spin-forbidden and has a slow relaxation rate, so that excitons are likely to diffuse into the peripheral layer, and thermal energy deactivation occurs from other than specific phosphorescent compounds. End up. That is, control of the recombination region of electrons and holes is more important than the fluorescent organic EL element.
- the triplet energy of the host material used for the light-emitting layer needs to be approximately 3.0 eV or more.
- Patent Document 1 discloses a material of a phosphorescent organic EL element, for example, in Patent Document 1, two benzene rings are bonded to a central benzene ring so as to form a condensed ring, and another condensed group is connected to the terminal.
- a polycyclic compound having a structure in which rings are bonded is disclosed.
- Patent Document 2 discloses a polycyclic compound having a ⁇ -conjugated heteroacene skeleton bridged by a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom.
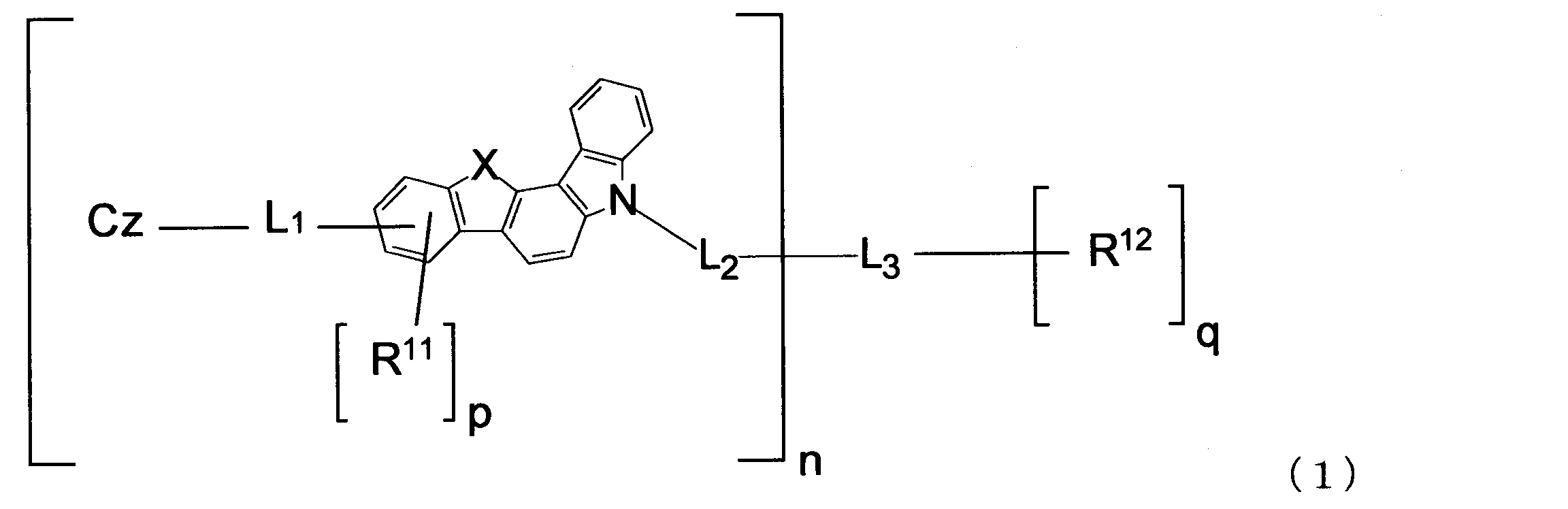
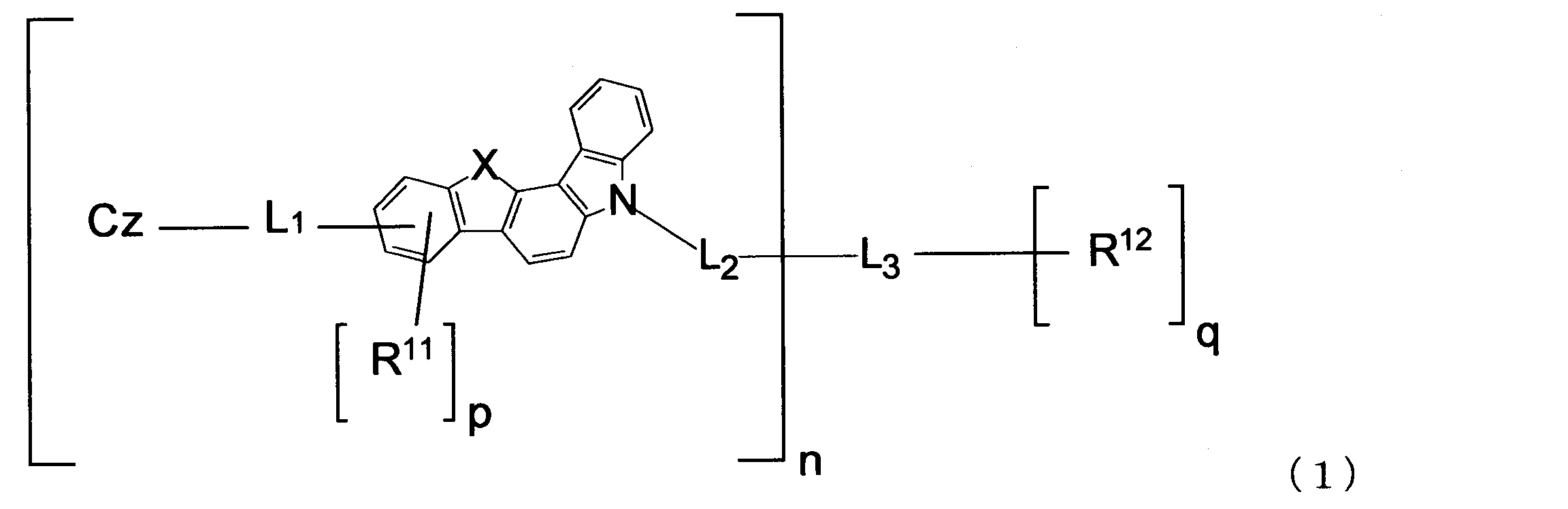
- An object of the present invention is to provide a material for an organic electroluminescence element capable of extending the life and lowering the driving voltage of the organic electroluminescence element.
- n is an integer of 1 to 4.
- X is an oxygen (O) atom or a sulfur (S) atom.
- n is 2 or more, the plurality of X may be the same or different.
- L 1 and L 2 are each a single bond, an arylene group having 6 to 18 ring carbon atoms which may have a substituent R, or an arylene group having 5 to 18 ring atoms which may have a substituent R.
- n is 2 or more, the plurality of L 1 and the plurality of L 2 may be the same or different.
- L 3 has an n + q valent saturated aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have a substituent R, an n + q valent silicon-containing group which may have a substituent R, and a substituent R. And an n + q valent aromatic hydrocarbon group having 6 to 18 ring carbon atoms, or an n + q valent unsaturated heterocyclic group having 5 to 18 ring atoms which may have a substituent R.
- R 11 and R 12 are each an alkyl group having 1 to 20 carbon atoms that may have a substituent R, a cycloalkyl group having 3 to 18 ring carbon atoms that may have a substituent R, and a substituent.
- p is an integer of 0 to 3, and when n is 2 or more, the plurality of p may be the same or different.
- q is an integer of 0 to 3.
- the substituent R is an alkyl group having 1 to 20 carbon atoms, a cycloalkyl group having 3 to 18 ring carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms, or a ring forming carbon.
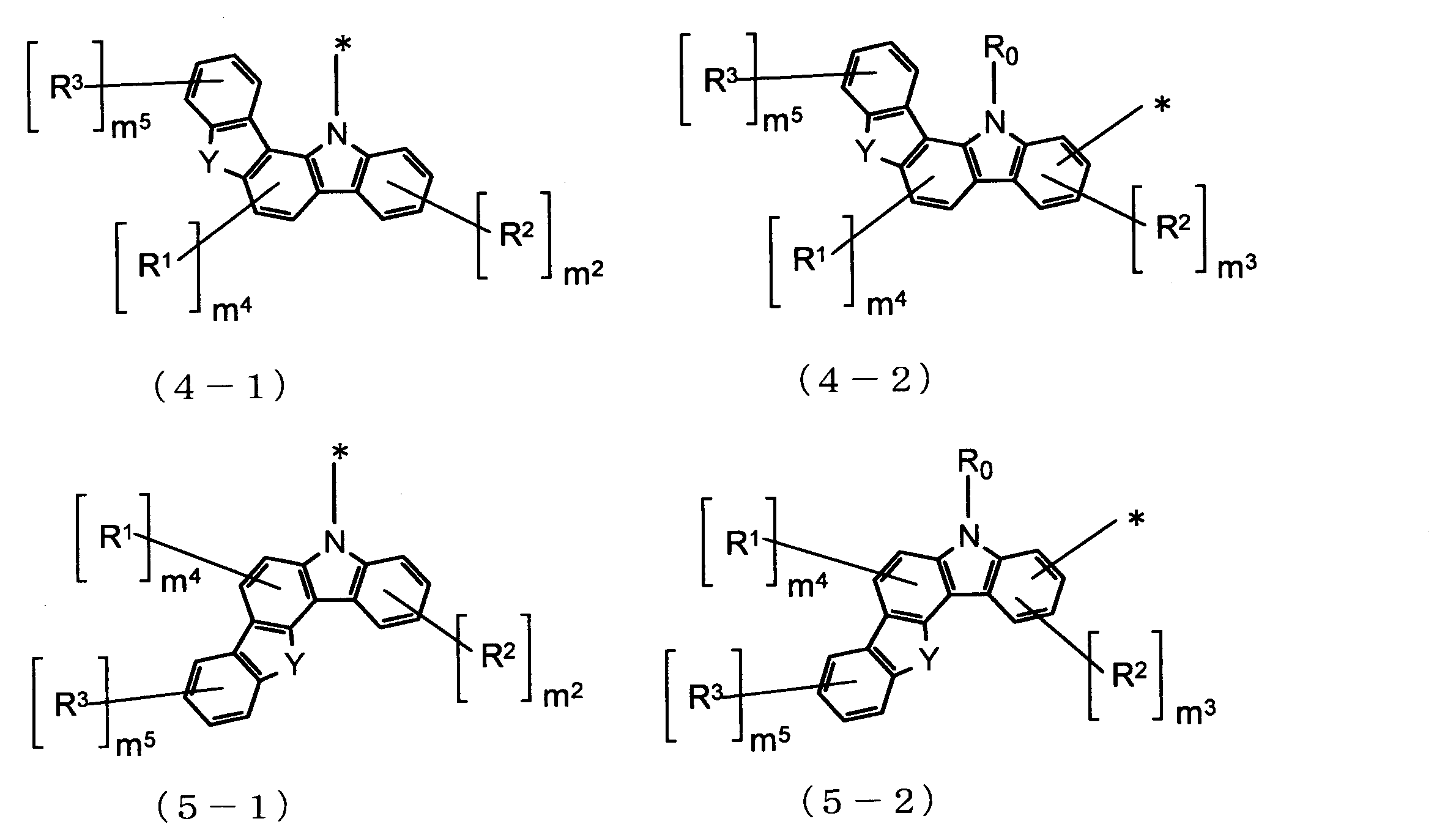
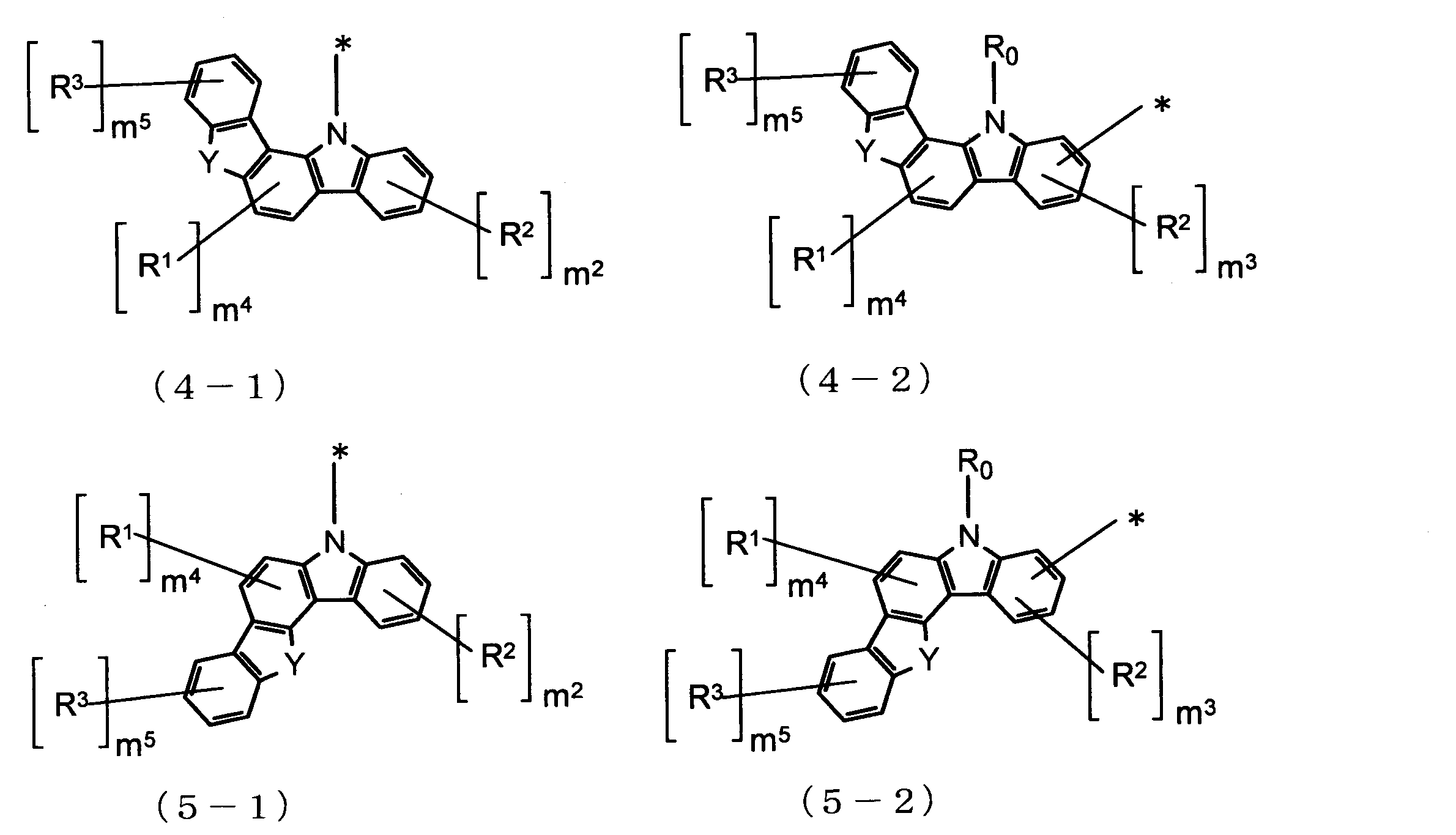
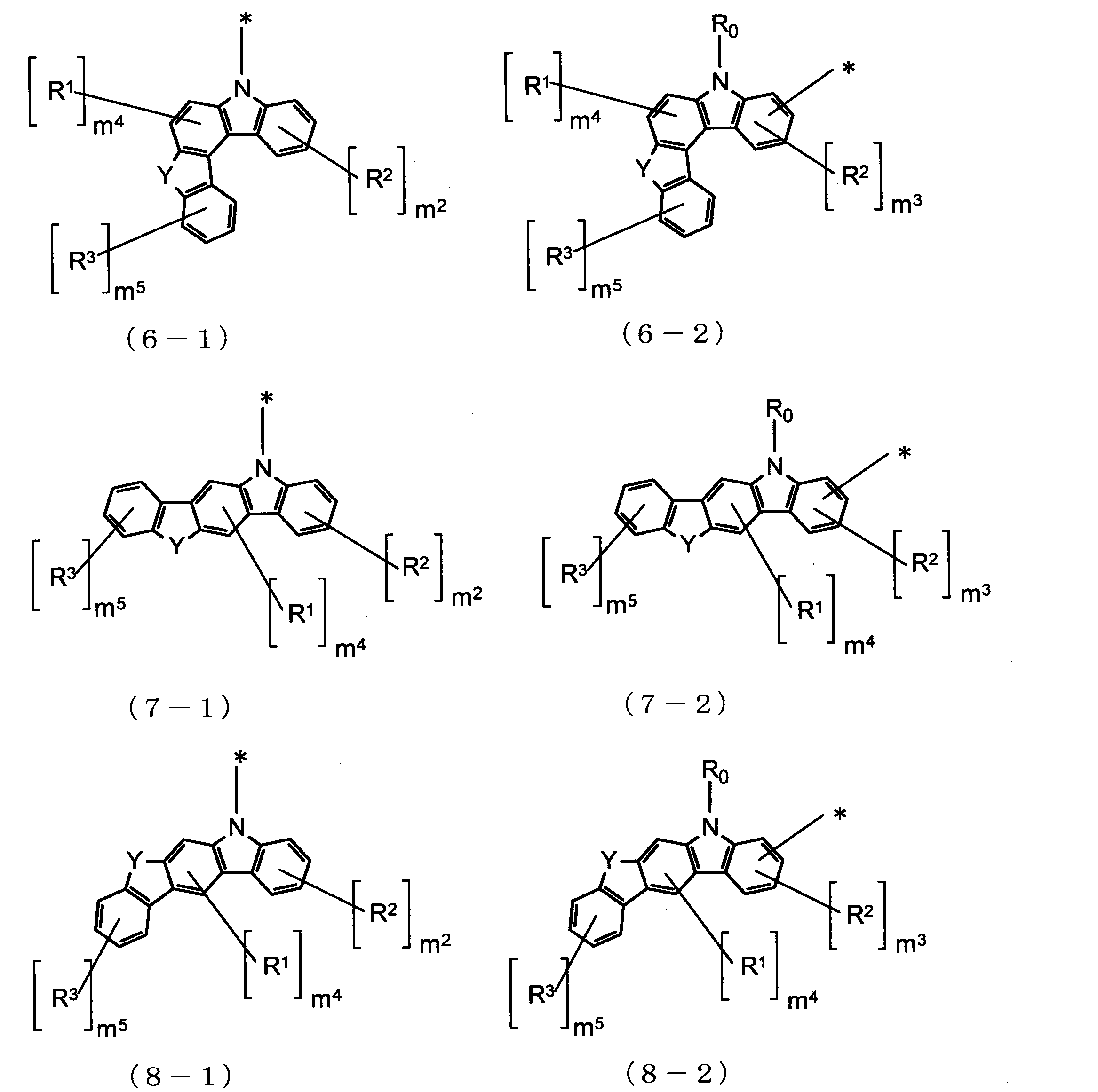
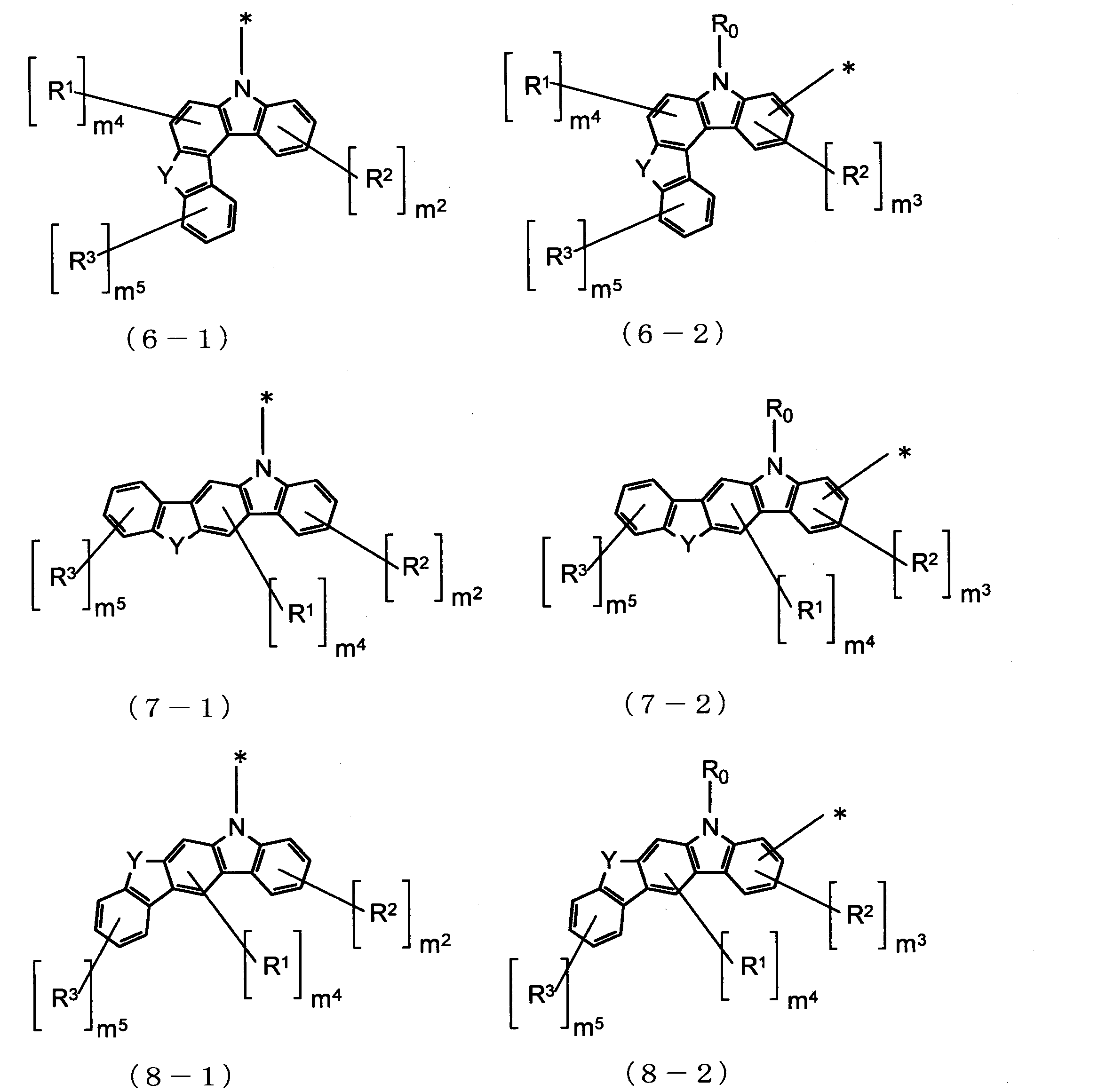
- Cz is represented by the following formulas (2-1), (2-2), (3-1), (3-2), (4-1), (4-2), (5-1), (5- 2) one group selected from the group consisting of (6-1), (6-2), (7-1), (7-2), (8-1), and (8-2) , N is 2 or more, the plurality of Cz may be the same or different.
- Y is an oxygen (O) atom or a sulfur (S) atom, and when n is 2 or more, the plurality of Y may be the same or different, m 1 and m 2 are each independently an integer of 0 to 4, m 3 is an integer from 0 to 3, m 4 is an integer from 0 to 2, m 5 is an integer from 0 to 4, R 0 , R 1 , R 2 and R 3 are each independently an alkyl group having 1 to 20 carbon atoms which may have a substituent R, or a ring having 3 to 3 carbon atoms which may have a substituent R.
- One or more organic thin film layers including a light emitting layer are provided between the cathode and the anode, and at least one of the organic thin film layers contains the material for an organic electroluminescent element according to any one of 1 to 5 Organic electroluminescence device. 7).
- the organic electroluminescence device wherein the organic electroluminescence device has an electron transport zone between the cathode and the light emitting layer, and the electron transport zone contains the material for an organic electroluminescence device.
- the light emitting layer contains a phosphorescent material, and the phosphorescent material is an orthometalated complex of a metal atom selected from iridium (Ir), osmium (Os), and platinum (Pt).
- Ir iridium
- Os osmium
- Pt platinum
- the present invention it is possible to provide a material for an organic electroluminescence element capable of extending the life of the organic electroluminescence element and reducing the driving voltage.
- the material for organic EL elements of the present invention is represented by the following formula (1).
- n is an integer of 1 to 4 (1, 2, 3 or 4). n is 1 or 2, for example.
- X is an oxygen (O) atom or a sulfur (S) atom.
- n is 2 or more, the plurality of X may be the same or different.
- L 1 and L 2 are each a single bond, an arylene group having 6 to 18 ring carbon atoms which may have a substituent R, or an arylene group having 5 to 18 ring atoms which may have a substituent R. When it is a heteroarylene group and n is 2 or more, the plurality of L 1 and the plurality of L 2 may be the same or different.
- ring-forming carbon means a carbon atom constituting a saturated ring, unsaturated ring or aromatic ring
- ring-forming atom means an atom constituting a saturated ring, unsaturated ring or aromatic ring.
- the hydrogen atom includes isotopes having different numbers of neutrons, that is, light hydrogen (protium), deuterium (deuterium), and tritium (tritium).
- L 3 has an n + q valent saturated aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have a substituent R, an n + q valent silicon-containing group which may have a substituent R, and a substituent R. And an n + q valent aromatic hydrocarbon group having 6 to 18 ring carbon atoms, or an n + q valent unsaturated heterocyclic group having 5 to 18 ring atoms which may have a substituent R.
- R 11 and R 12 are each an alkyl group having 1 to 20 carbon atoms that may have a substituent R, a cycloalkyl group having 3 to 18 ring carbon atoms that may have a substituent R, and a substituent.
- p is an integer of 0 to 3 (0, 1, 2, or 3). When n is 2 or more, the plurality of p may be the same or different. p is, for example, 0. q is an integer of 0 to 3 (0, 1, 2, or 3), for example, 0.
- the substituent R is an alkyl group having 1 to 20 carbon atoms, a cycloalkyl group having 3 to 18 ring carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms, or a ring forming carbon.
- An aryloxy group having 6 to 18 atoms, a silyl group, a fluoro group, a cyano group, an aryl group having 6 to 18 ring carbon atoms, or a heteroaryl group having 5 to 18 ring atoms, and when there are a plurality of R may be the same or different.
- Cz is represented by the following formulas (2-1), (2-2), (3-1), (3-2), (4-1), (4-2), (5-1), (5- 2) one group selected from the group consisting of (6-1), (6-2), (7-1), (7-2), (8-1), and (8-2) , N is 2 or more, the plurality of Cz may be the same or different.
- Y is an oxygen (O) atom or sulfur (S) atom.
- n is 2 or more, the plurality of Y may be the same or different.
- n 1 and m 2 are each independently an integer of 0 to 4 (0, 1, 2, 3, or 4). m 1 and m 2 are, for example, 0 or 1, and (m 1 + m 2 ) is preferably 2 or less.
- m 3 is an integer of 0 to 3 (0, 1, 2, or 3). m 3 is 0, for example.
- m 4 is an integer of 0 to 2 (0, 1 or 2).
- m 4 is 0, for example.
- m 5 is an integer of 0 to 4 (0, 1, 2, 3 or 4). m 5 is, for example, 0.
- R 0 , R 1 , R 2 and R 3 are each independently an alkyl group having 1 to 20 carbon atoms which may have a substituent R, or a ring having 3 to 3 carbon atoms which may have a substituent R.
- the organic EL device material of the present invention is particularly preferable as a material for a light emitting layer of an organic EL device that emits phosphorescence or a layer adjacent to the light emitting layer, for example, a hole barrier layer or an electron barrier layer.
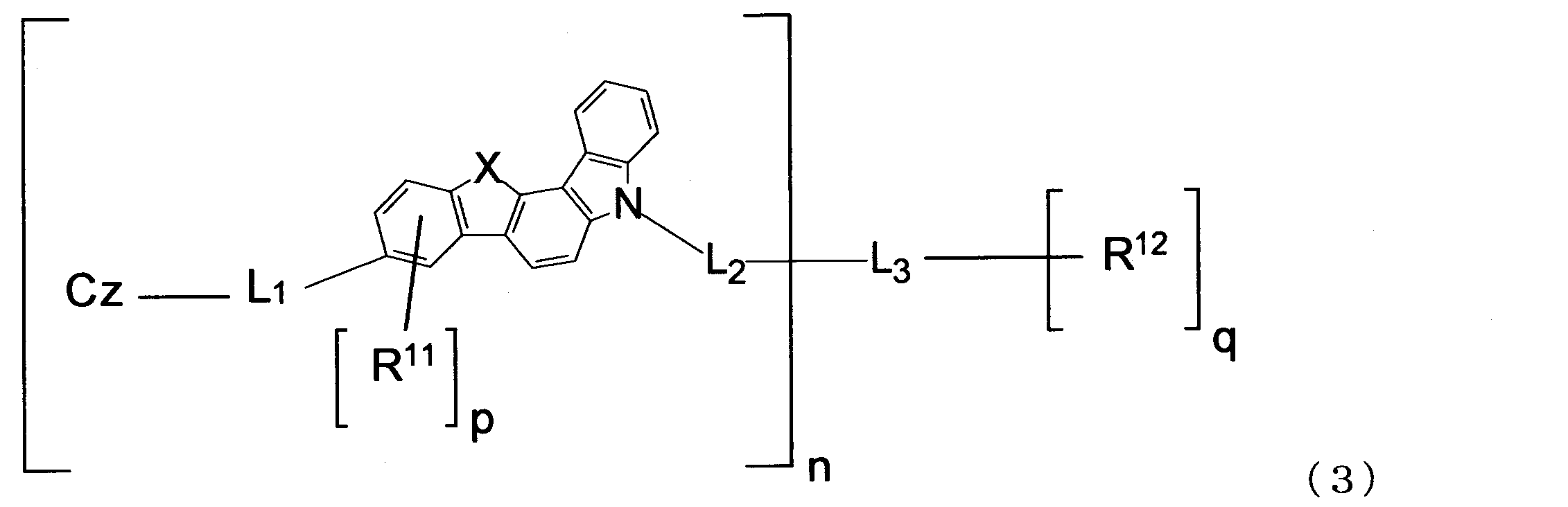
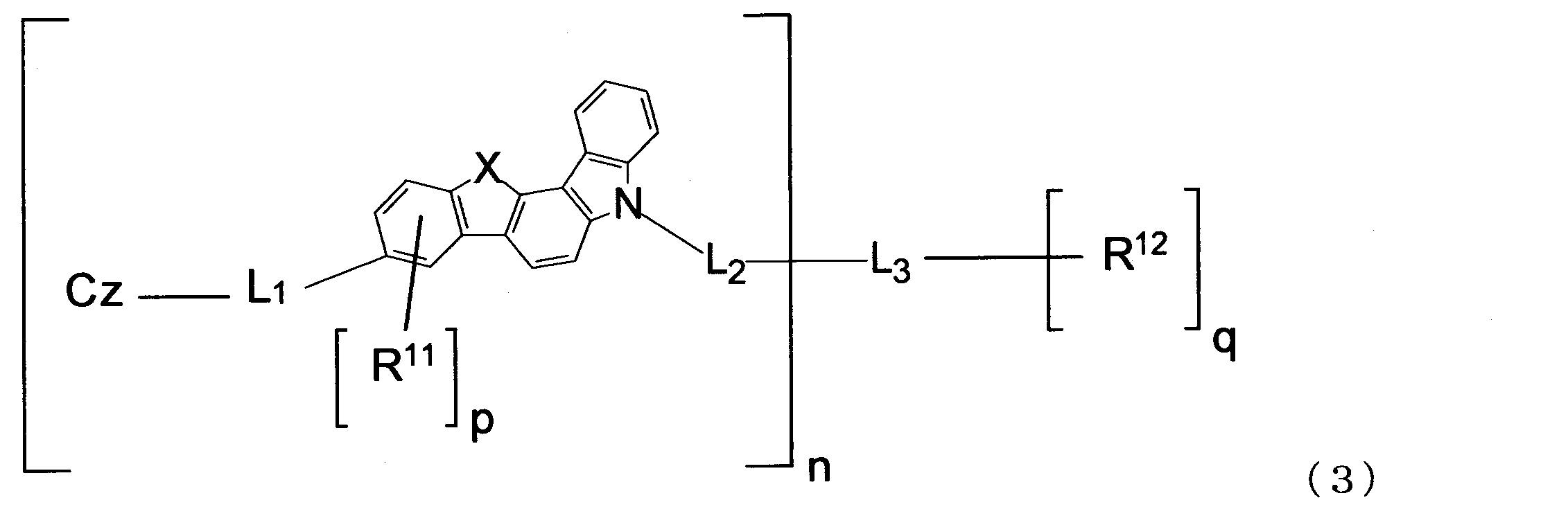
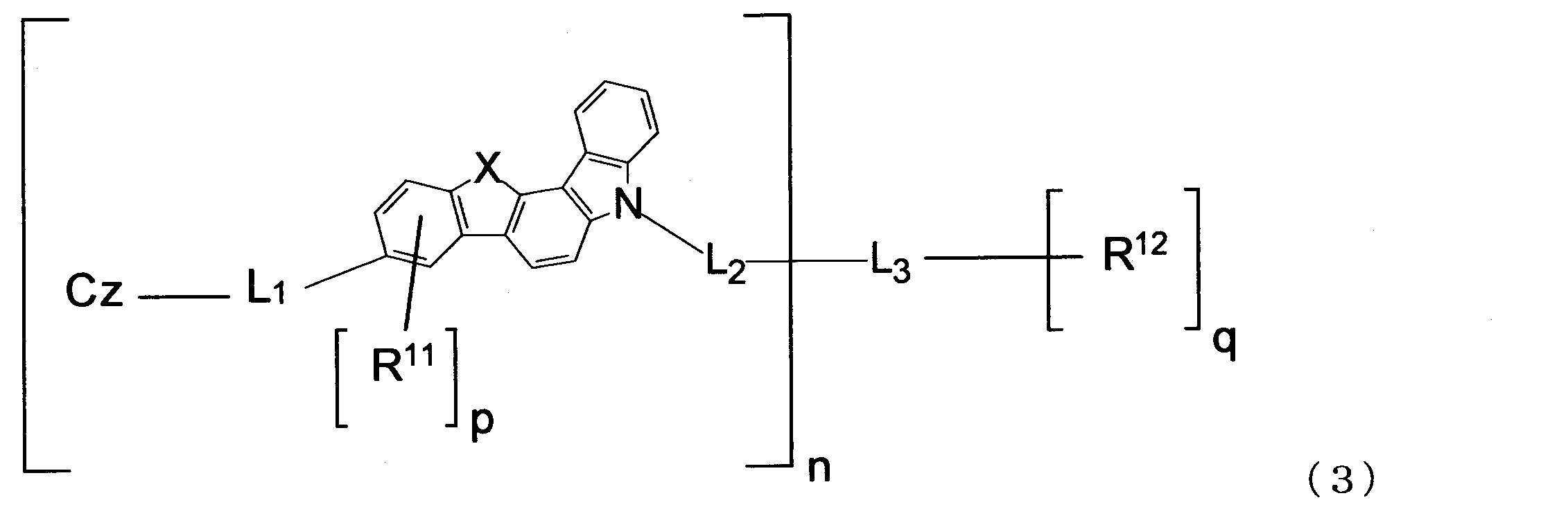
- the material for an organic EL device of the present invention is preferably represented by the following formula (3).
- n, X, L 1 , L 2 , L 3 , R 11 , R 12 , p, q and Cz are the same as those in the formula (1).
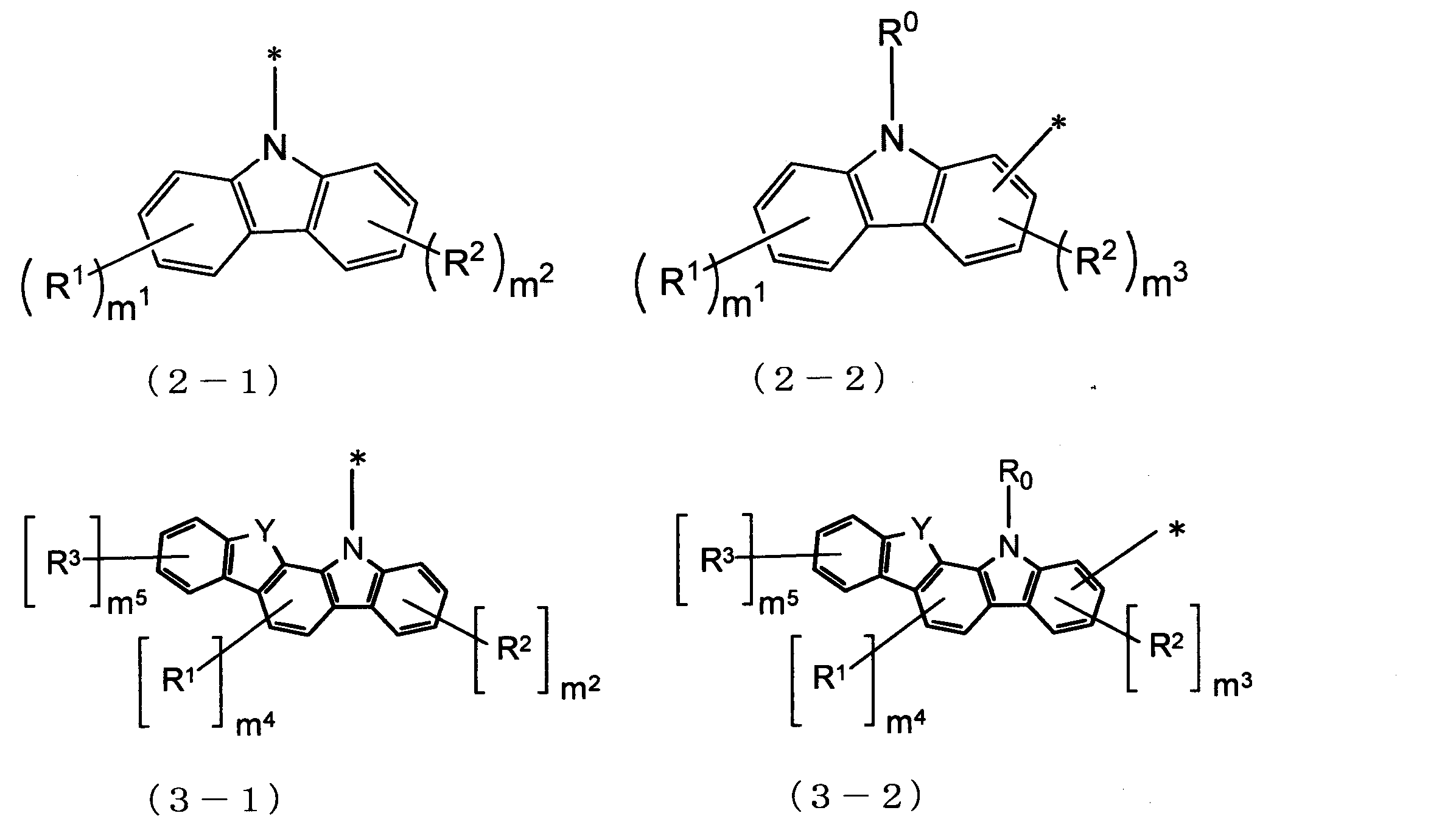
- Cz in the formula (1) has a carbazole skeleton or a ring structure having a carbazole skeleton as a partial structure. Since both the ring structure represented by Cz in the formula (4) and D surrounded by the broken line in the formula (4) have a carbazole skeleton as a partial structure, good affinity between these ring structures And a good film having a high glass transition point and a high carrier transport ability can be formed.
- the organic EL device material of the present invention has no substituent on the benzene rings A and B in the ring structure represented by D surrounded by the broken line in the above formula (4). Thereby, a good aggregation state or molecular orientation state is formed between the molecules of the material or between the material molecule and the dopant molecule, and a good film having a high glass transition point and a high carrier transporting ability is formed. be able to. This is thought to be because the aspect ratio tends to be high as the molecular shape.
- the material for an organic EL element of the present invention is particularly suitable as a host material, an electron barrier layer, and a hole barrier layer of a light emitting layer of the organic EL element.
- Cz in Formula (1) is a group represented by Formula (2-1), that is, when the carbazole skeleton of Cz is bonded to L 1 at the 9-position, the host material of the light-emitting layer, or It is further suitable as a hole blocking layer.
- Cz in formula (1) is a group represented by formula (2-2), that is, when the carbazole skeleton of Cz is bonded to L 1 at positions other than 9-position, hole transportability is improved. Therefore, it is more suitable as a material for the electron barrier layer.
- alkyl group having 1 to 20 carbon atoms include linear or branched alkyl groups, and specifically include methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec- Examples include butyl group, tert-butyl group, n-pentyl group, n-hexyl group, n-heptyl group, n-octyl group and the like, preferably methyl group, ethyl group, propyl group, isopropyl group, n-butyl Group, isobutyl group, sec-butyl group and tert-butyl group, preferably methyl group, ethyl group, propyl group, isopropyl group, n-butyl group, sec-butyl group and tert-butyl group.
- Examples of the cycloalkyl group having 3 to 18 ring carbon atoms include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a 1-adamantyl group, a 2-adamantyl group, a 1-norbornyl group, and a 2-norbornyl group. Preferred are a cyclopentyl group, a cyclohexyl group, a 1-adamantyl group, and a 2-adamantyl group.
- the alkoxy group having 1 to 20 carbon atoms is represented as —OY 1 and examples of Y 1 include the above alkyl examples.
- the alkoxy group is, for example, a methoxy group or an ethoxy group.
- the alkoxy group may be substituted with a fluorine atom, and in this case, a trifluoromethoxy group or the like is preferable.
- a cycloalkoxy group having 3 to 20 ring carbon atoms is represented by —OY 2, and examples of Y 2 include the above-described cycloalkyl groups.
- the cycloalkoxy group is, for example, a cyclopentyloxy group or a cyclohexyloxy group.
- the aryl group having 6 to 18 ring carbon atoms is preferably an aryl group having 6 to 12 ring carbon atoms.
- Specific examples of the monovalent aryl group include phenyl group, biphenyl group, terphenyl group, tolyl group, xylyl group, naphthyl group, anthryl group, phenanthryl group, naphthacenyl group, pyrenyl group, chrysenyl group, triphenylenyl group, fluorenyl group.
- Biphenylyl group, terphenyl group, fluoranthenyl group and the like preferably phenyl group, biphenyl group, terphenyl group, tolyl group, xylyl group, naphthyl group, phenanthryl group and triphenylenyl group.
- n + q valent aromatic hydrocarbon group having 6 to 18 ring carbon atoms examples include the n + q valent groups of the aforementioned groups.
- arylene group having 6 to 18 ring carbon atoms examples include the divalent groups described above.
- An aryloxy group having 6 to 18 ring carbon atoms is represented by —OY 3, and examples of Y 3 include the above aryl groups.
- the aryloxy group is, for example, a phenoxy group.
- the heteroaryl group having 5 to 18 ring atoms is preferably a heteroaryl group having 5 to 10 ring atoms.
- Specific examples of the monovalent heteroaryl group include pyrrolyl group, pyrazinyl group, pyridinyl group, pyrimidinyl group, triazinyl group, indolyl group, isoindolyl group, imidazolyl group, furyl group, benzofuranyl group, isobenzofuranyl group, dibenzofuranyl group.
- n + q valent unsaturated heterocyclic group having 5 to 18 ring atoms examples include the n + q valent group described above.
- heteroarylene group having 5 to 18 ring atoms include the divalent groups described above.
- Examples of the n + q-valent silicon-containing group include a silyl group.
- Examples of the silicon-containing group substituted by R include an alkylsilyl group having 1 to 20 carbon atoms (preferably 1 to 10 carbon atoms, more preferably 1 to 6 carbon atoms), and 6 to 18 ring carbon atoms (preferably Is an arylsilyl group having 6 to 12 ring carbon atoms.
- Specific examples of the alkylsilyl group include a trimethylsilyl group, a triethylsilyl group, a t-butyldimethylsilyl group, a vinyldimethylsilyl group, and a propyldimethylsilyl group.
- arylsilyl group examples include a triphenylsilyl group, a phenyldimethylsilyl group, a t-butyldiphenylsilyl group, a tolylsilylsilyl group, a trixylsilyl group, a trinaphthylsilyl group, and the like.
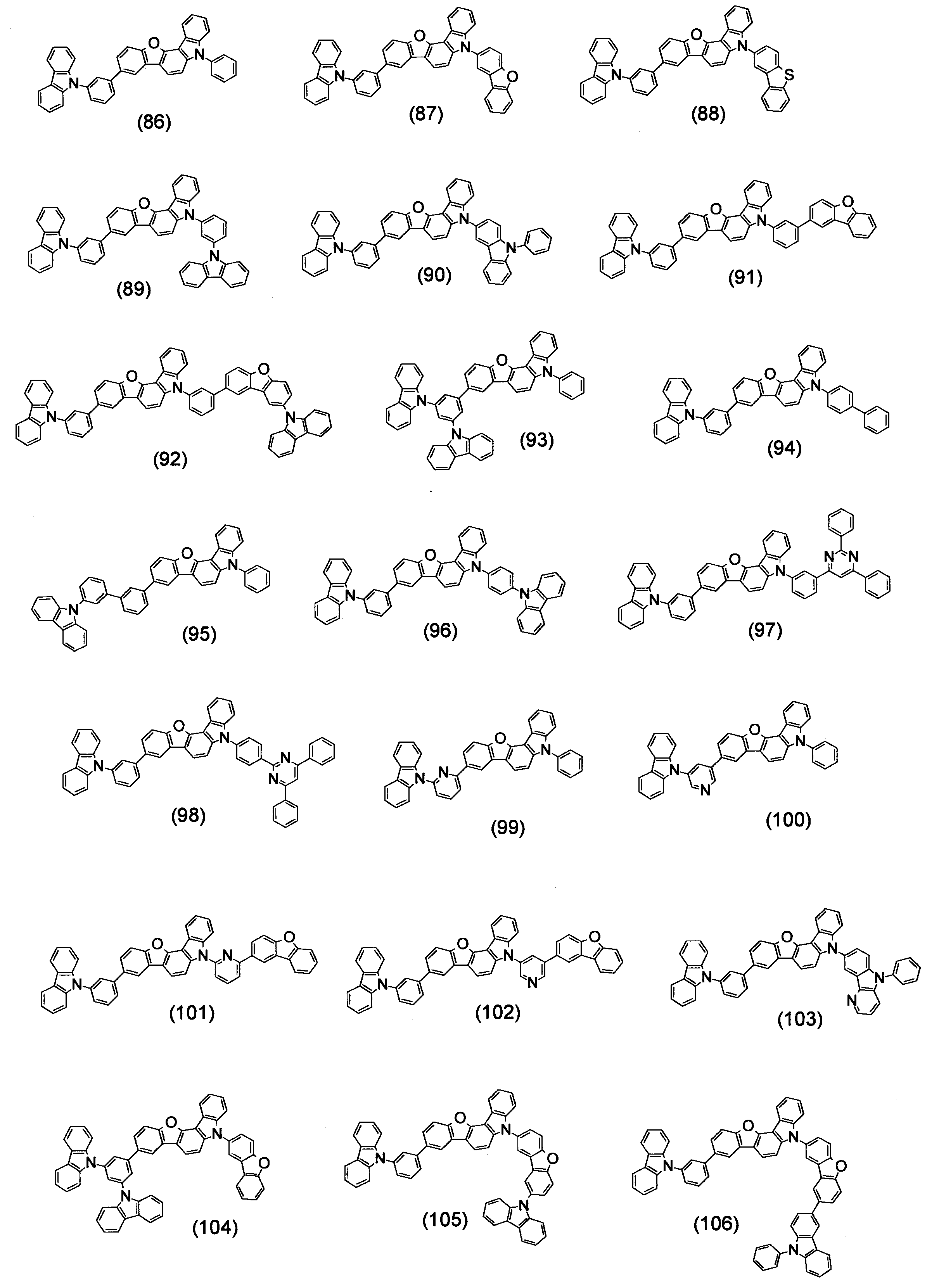
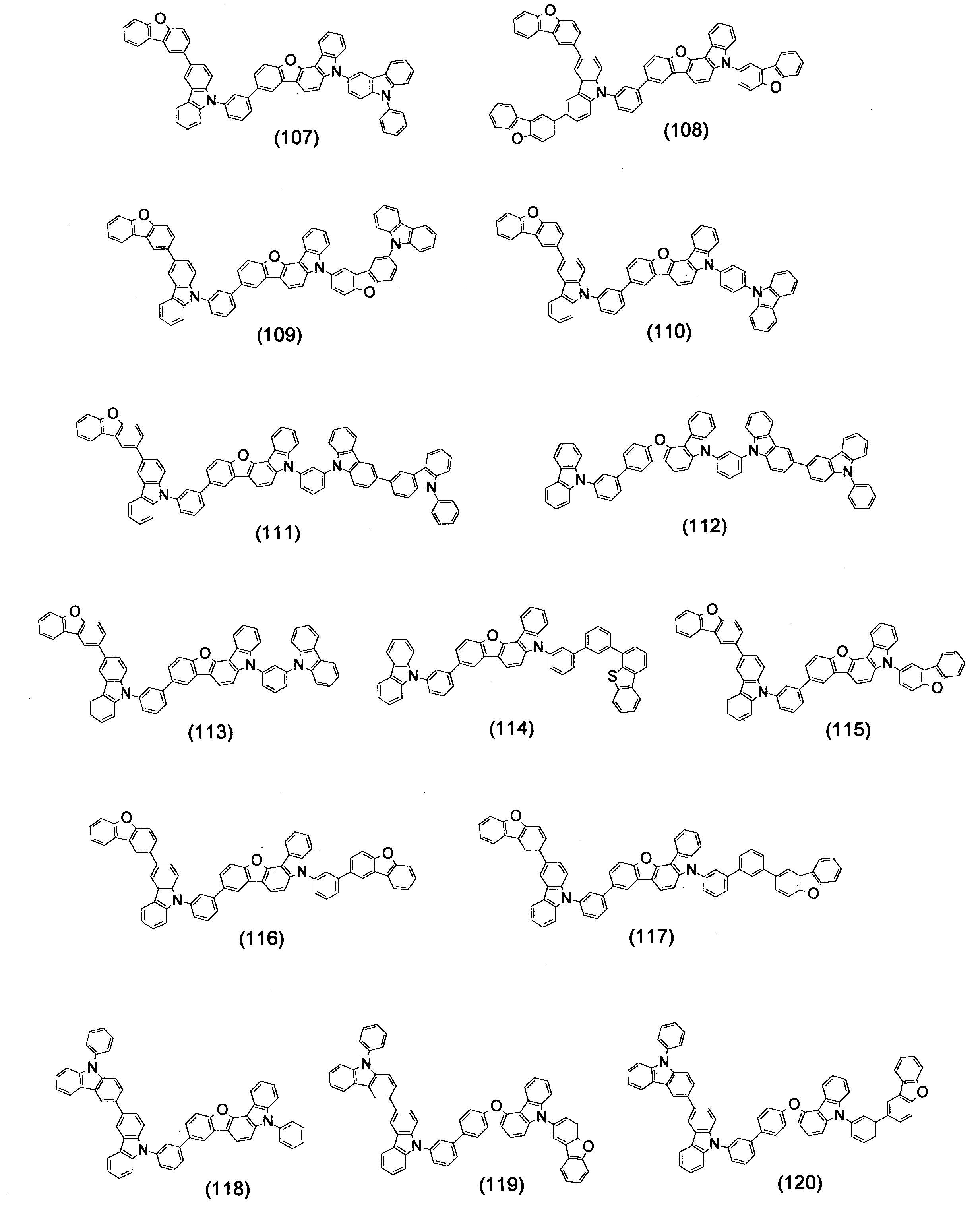
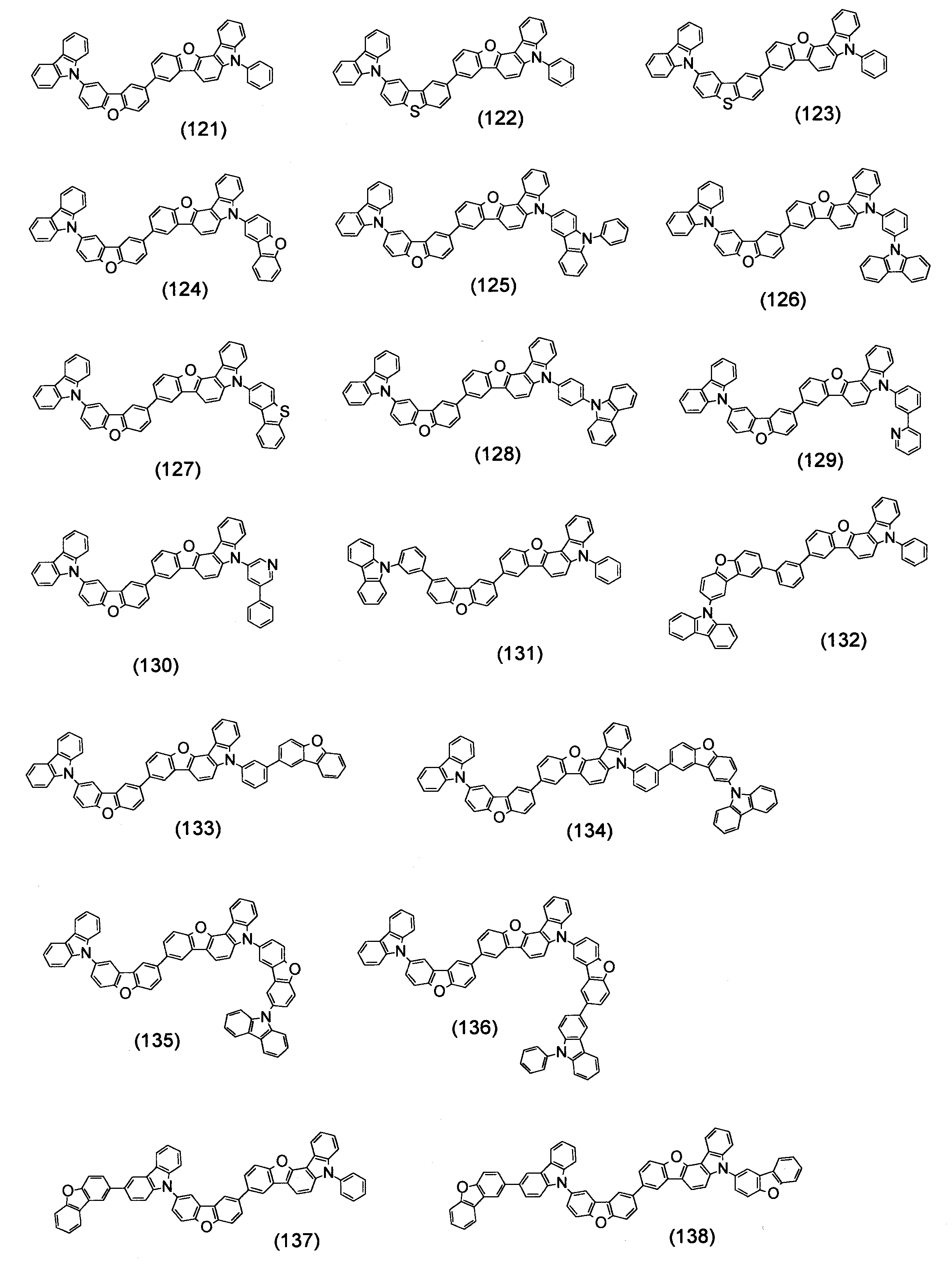
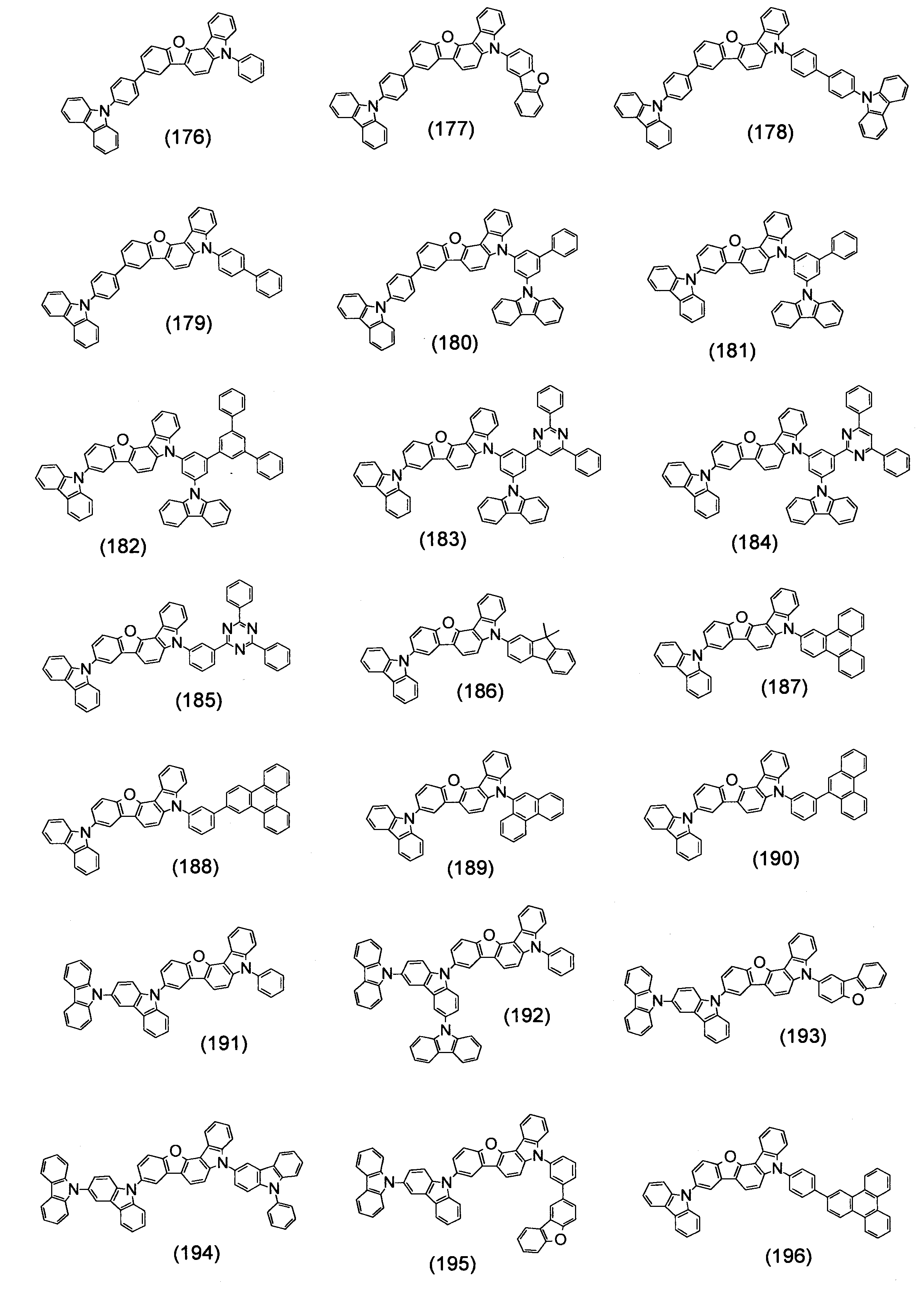
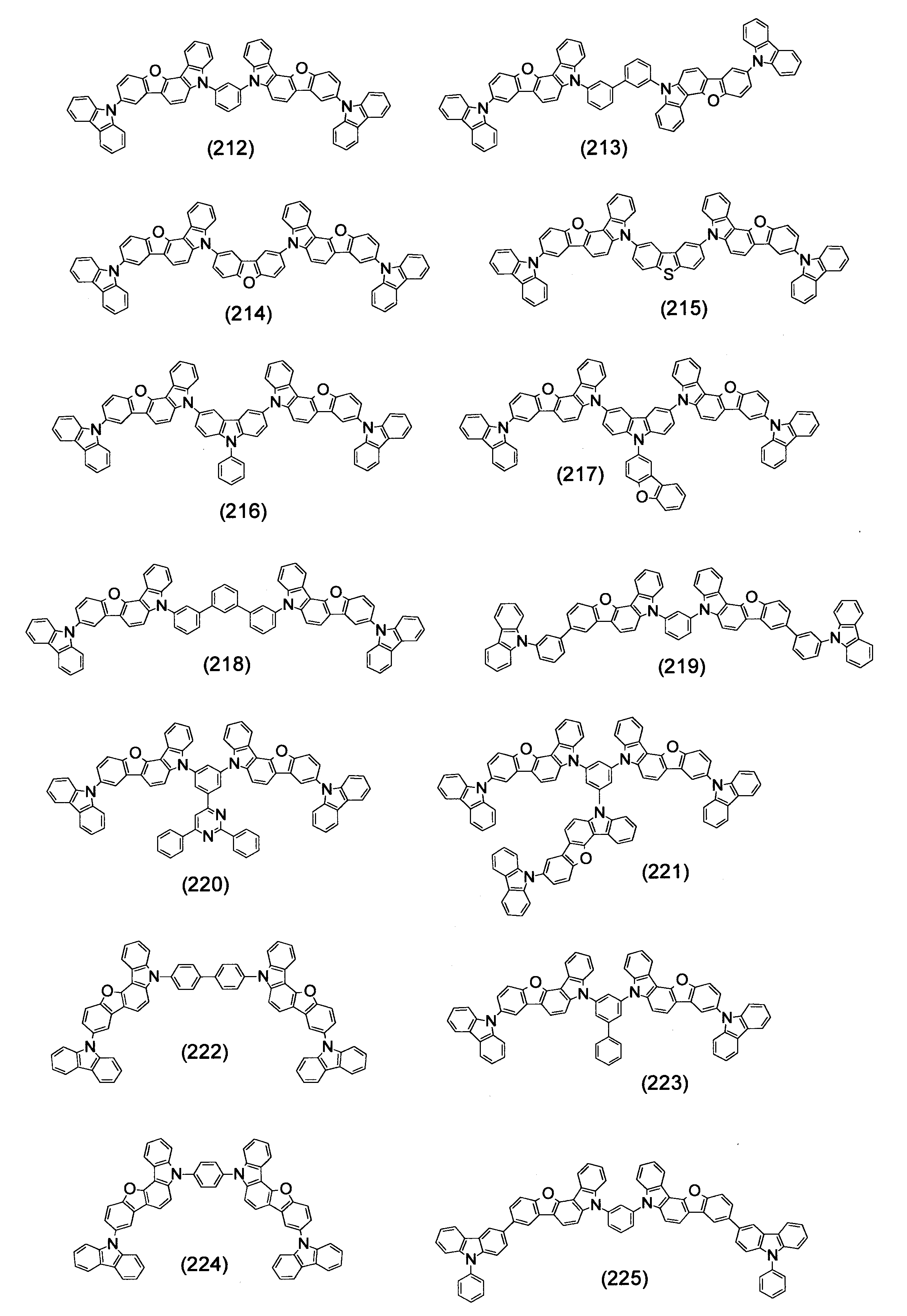
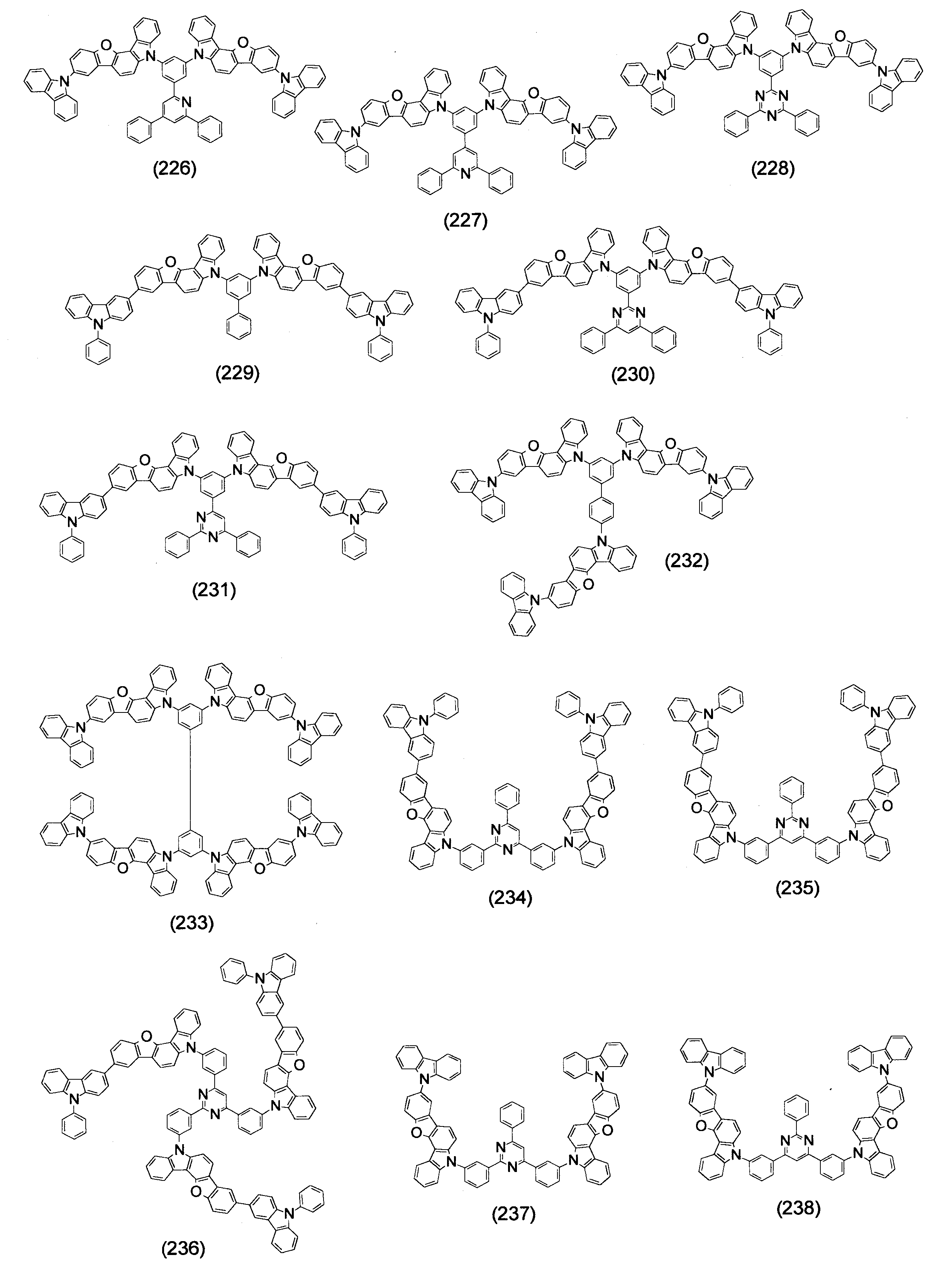
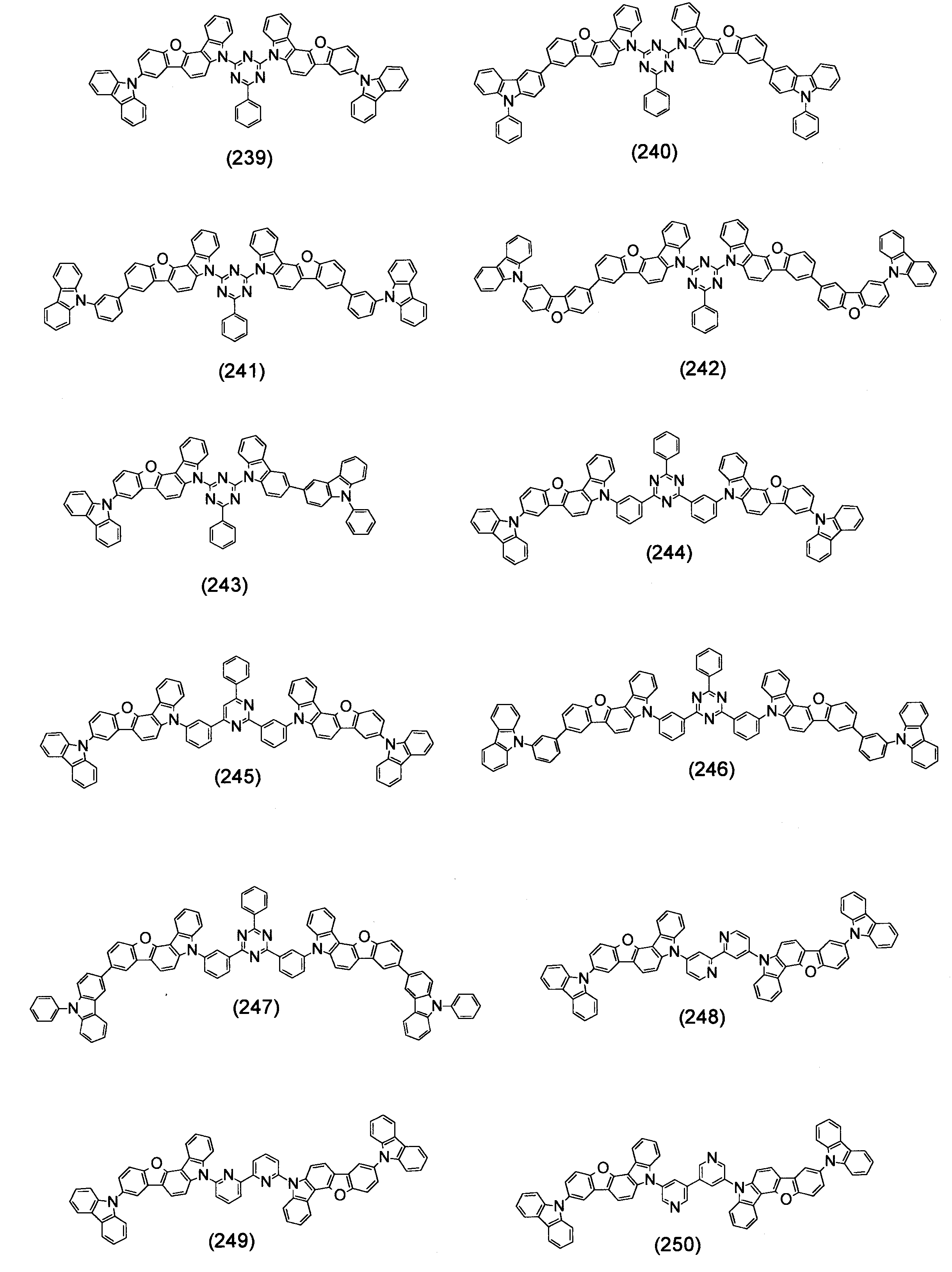
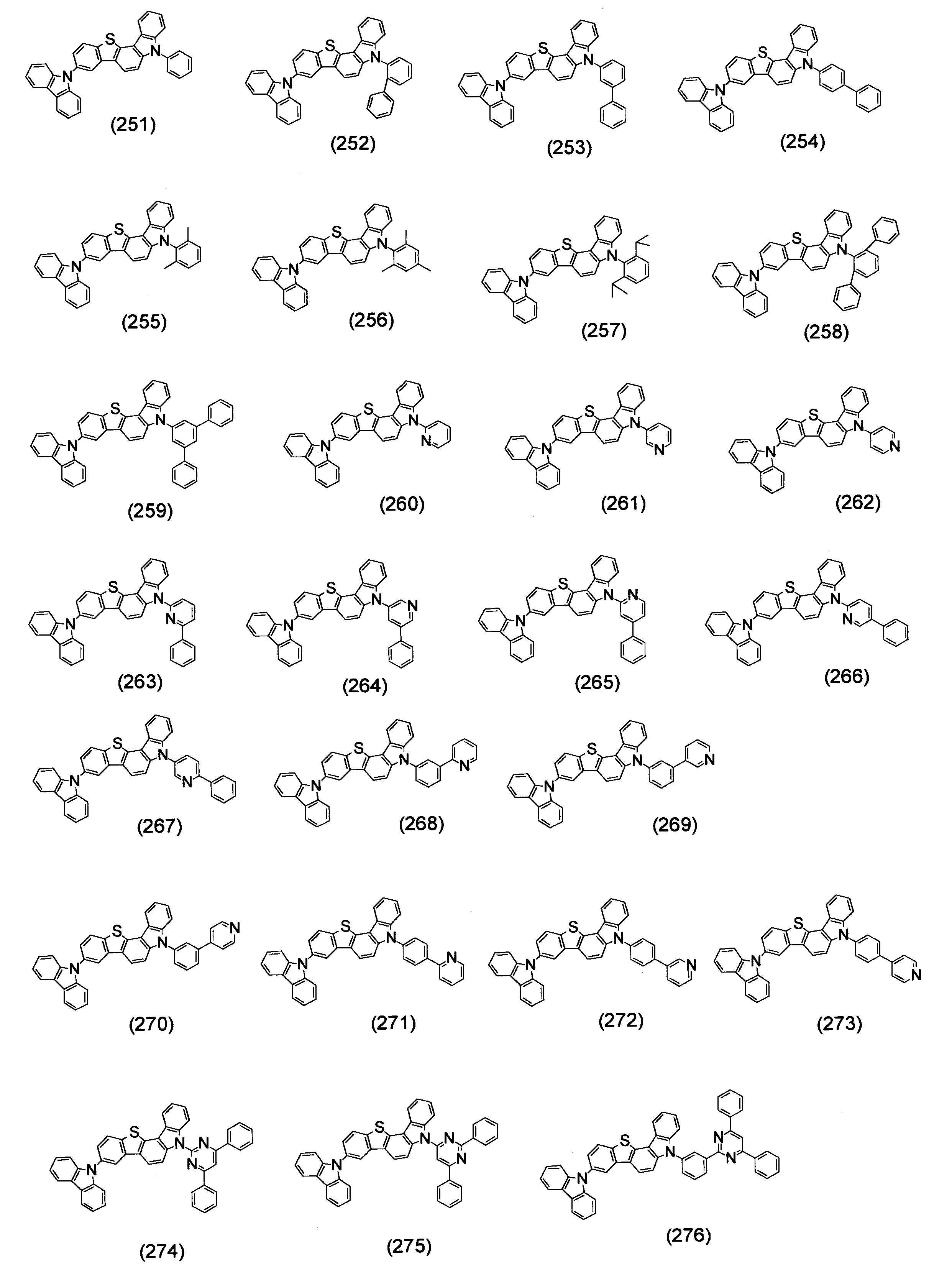
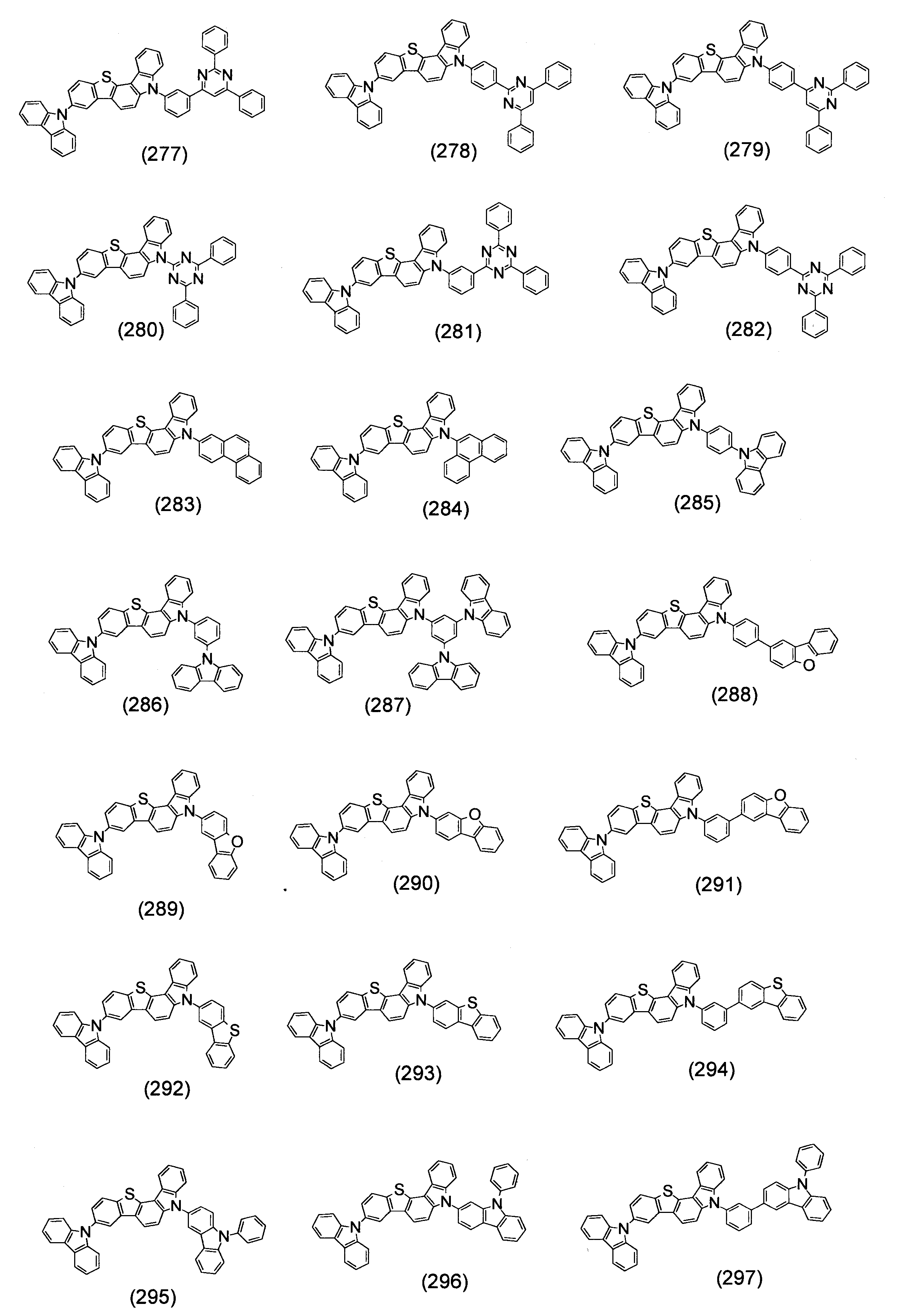
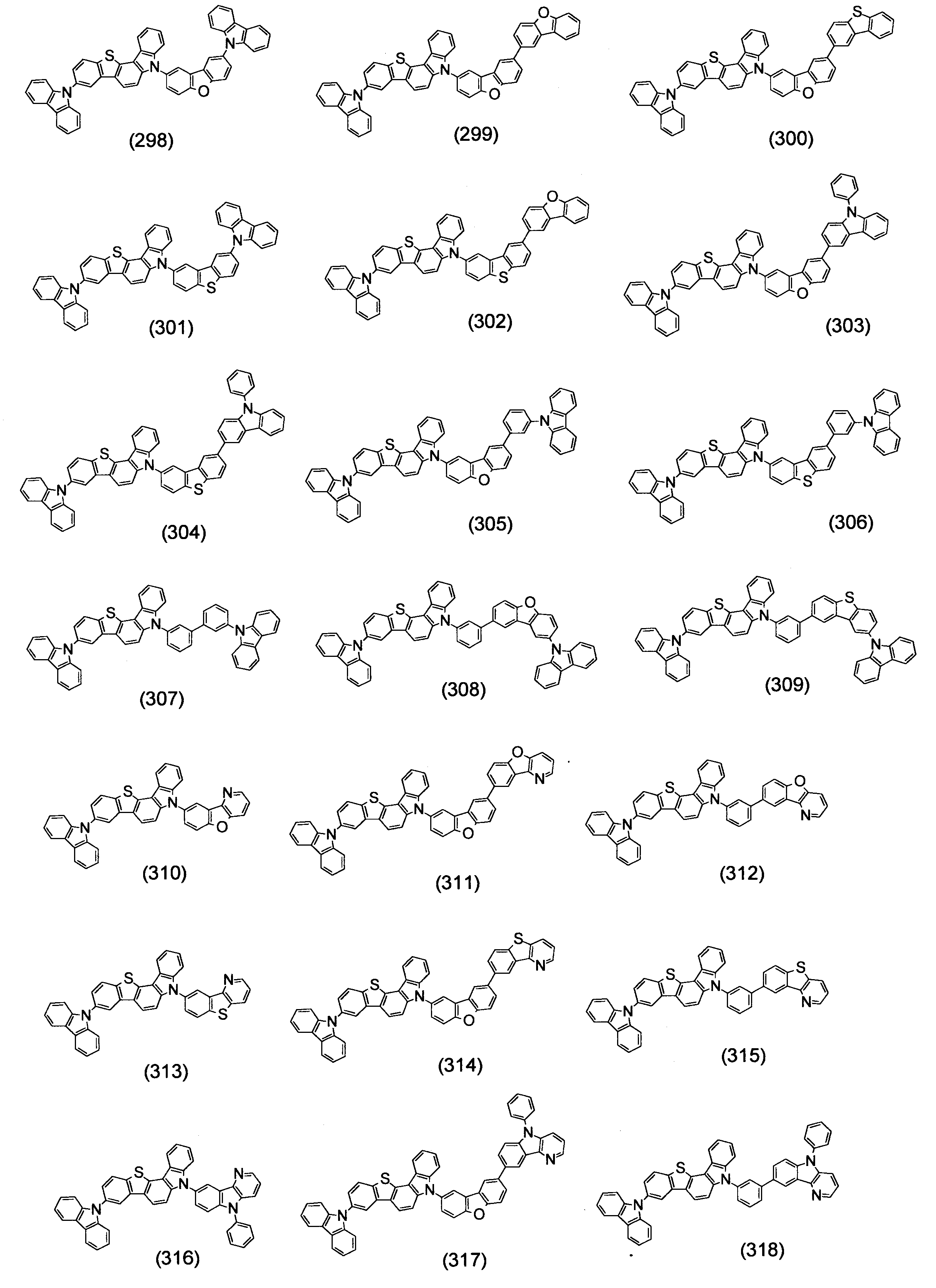
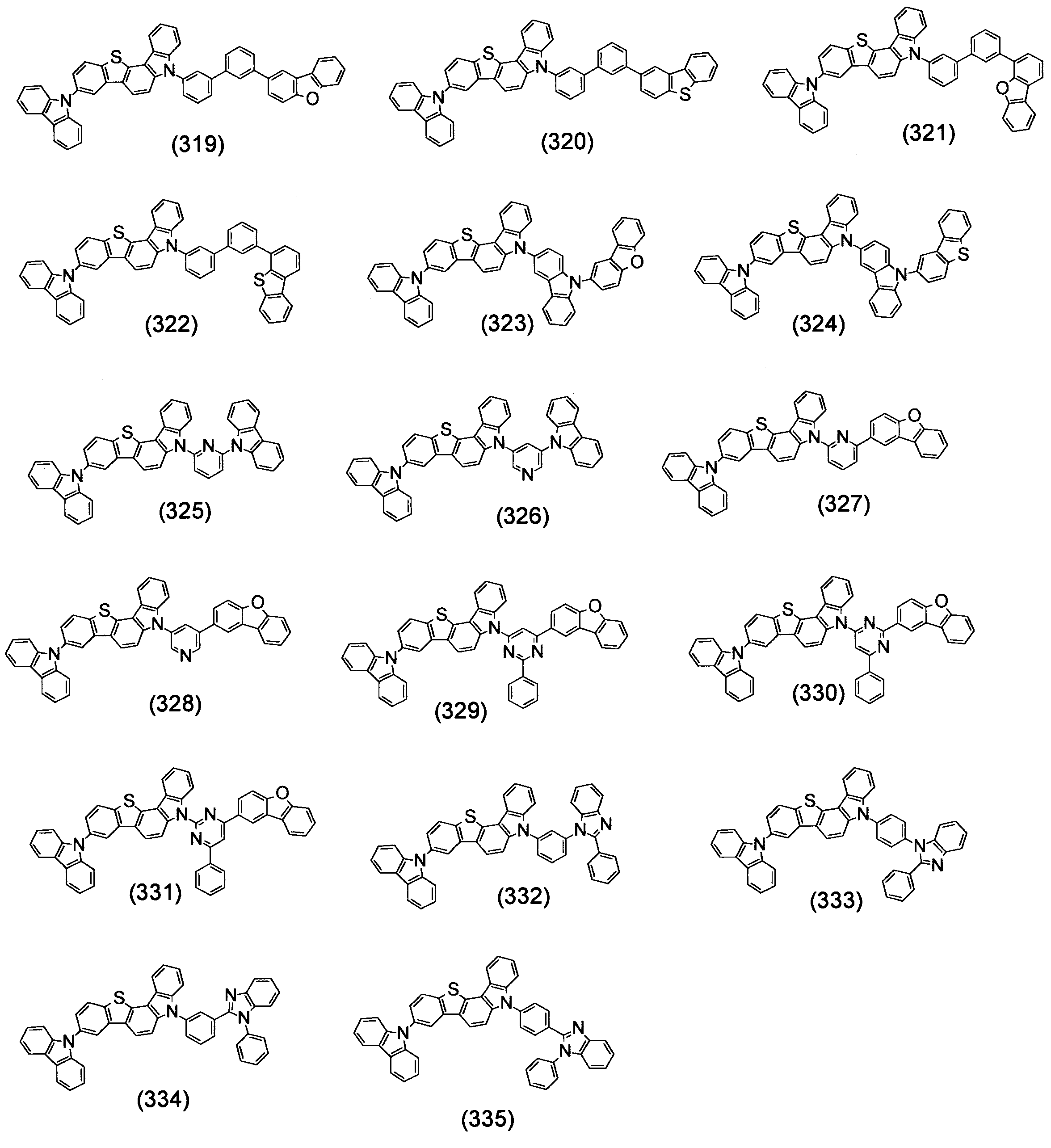
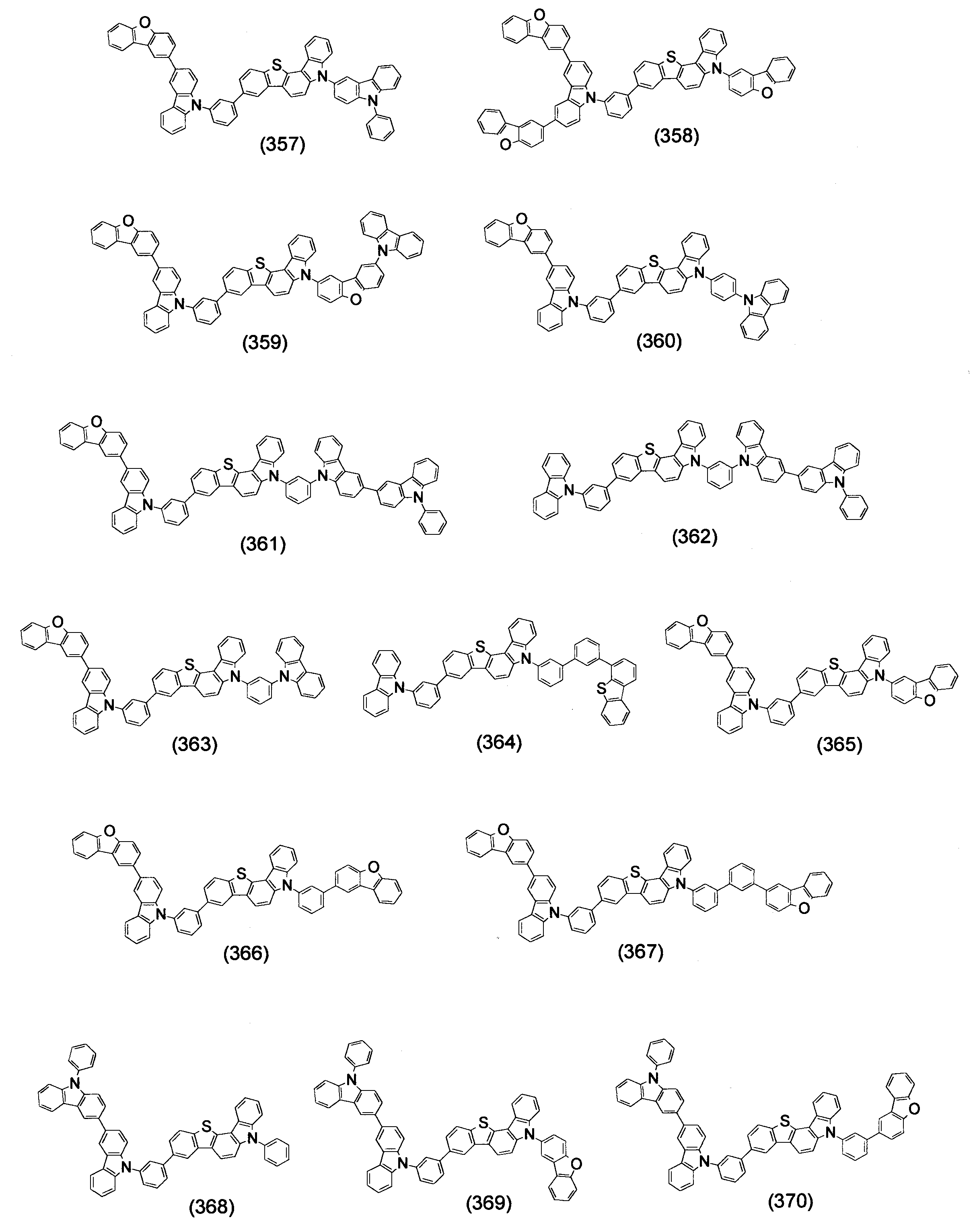
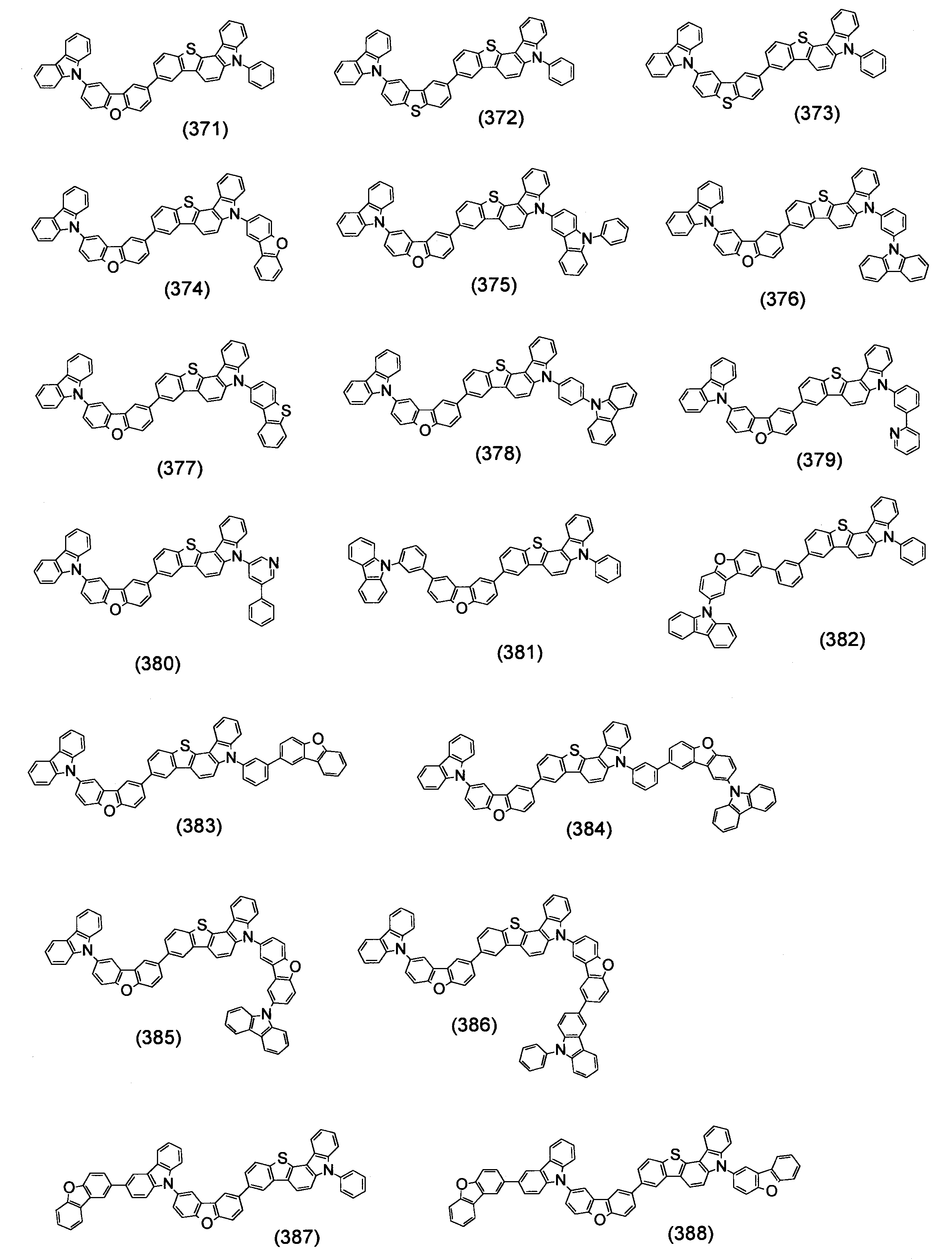
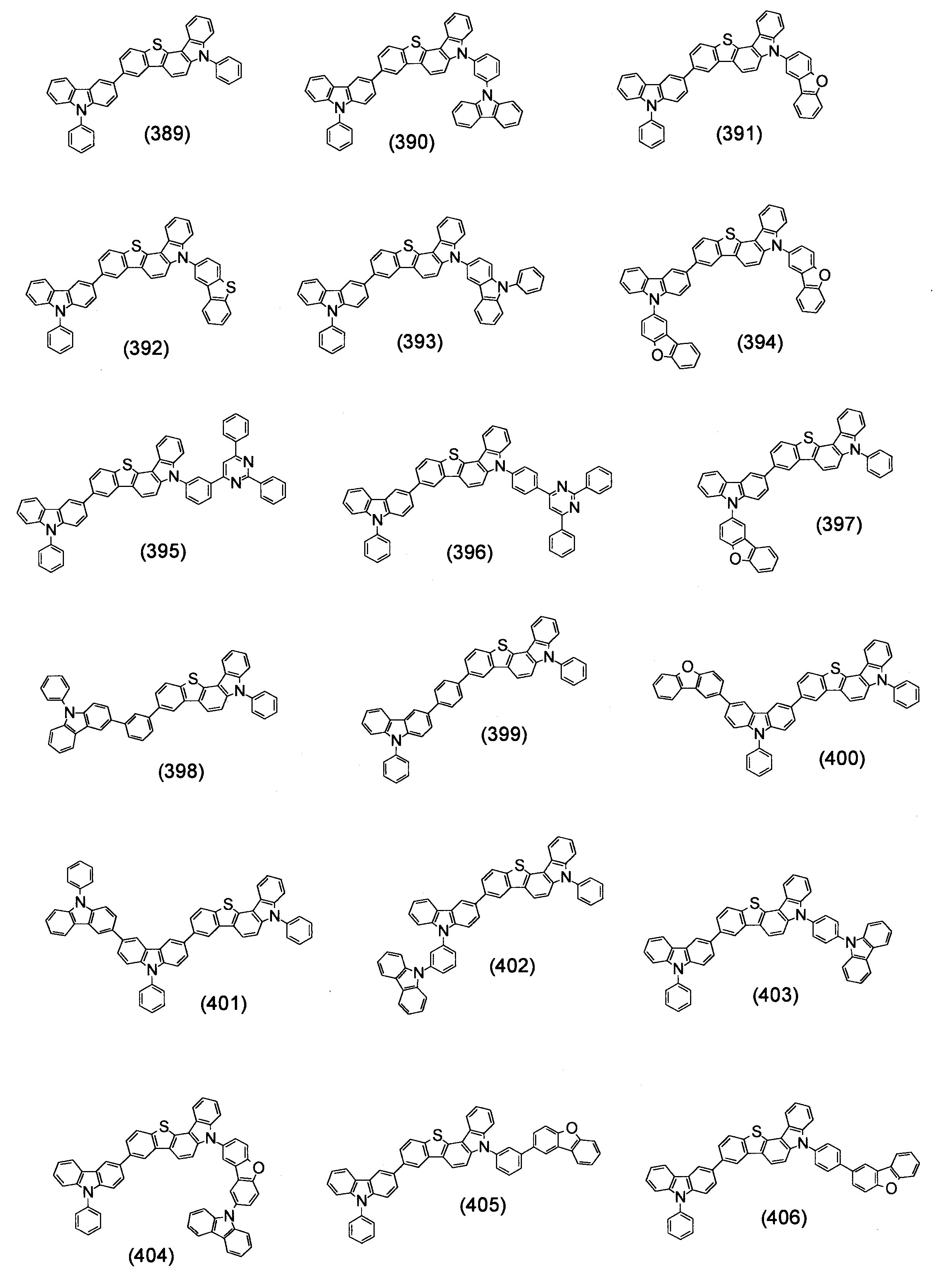
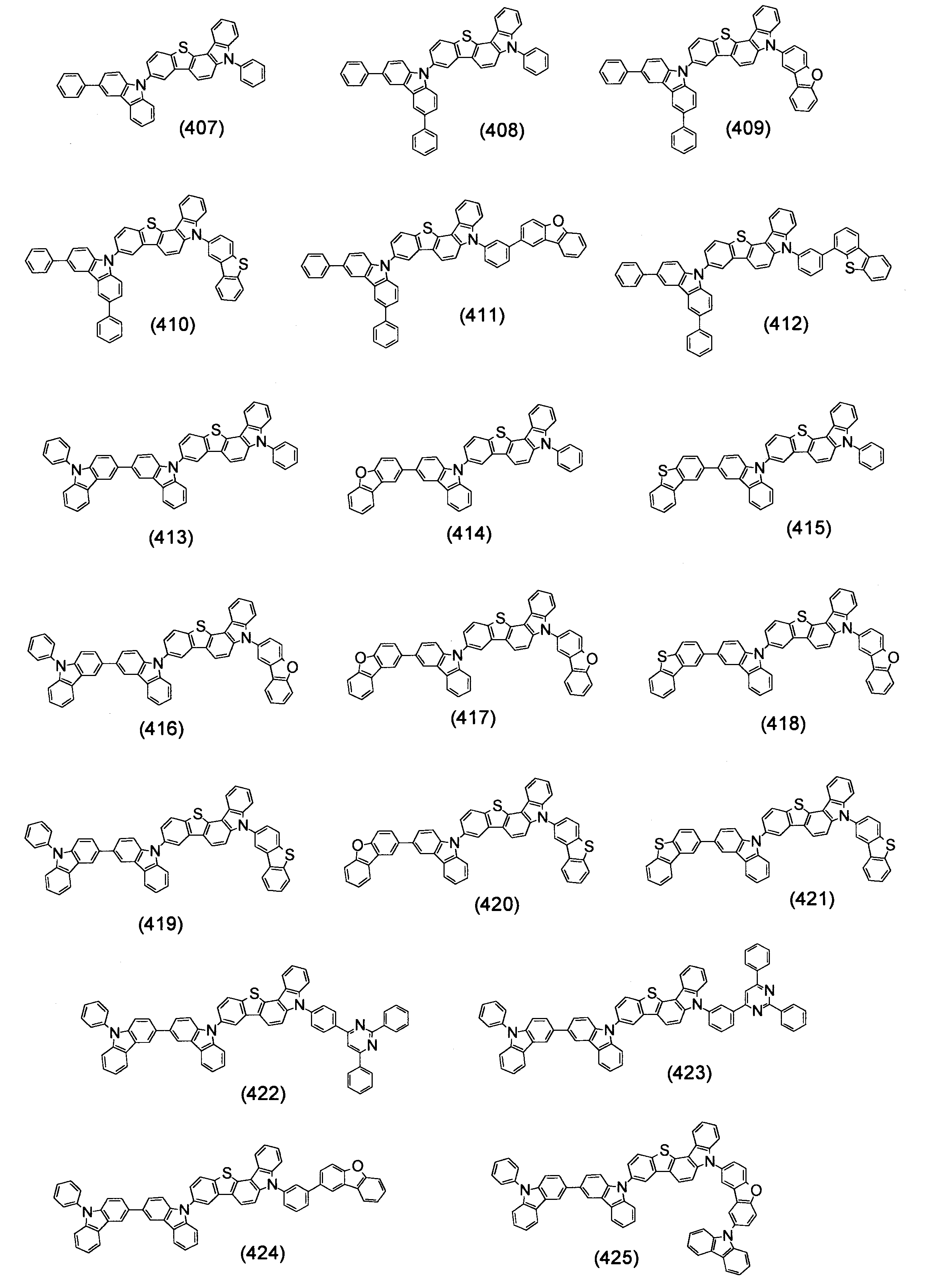
- Specific examples of the compound represented by the above formula (1) are shown below.
- the compounds of the present invention can be synthesized by the methods described in the synthesis examples of the examples or methods known to those skilled in the art.
- the organic EL element of the present invention will be described.
- the organic EL device of the present invention has one or more organic thin film layers including a light emitting layer between an anode and a cathode. And at least 1 layer of an organic thin film layer contains the organic EL element material of this invention.
- FIG. 1 is a schematic view showing a layer structure of an embodiment of the organic EL device of the present invention.
- the organic EL element 1 has a configuration in which an anode 20, a hole transport zone 30, a phosphorescent light emitting layer 40, an electron transport zone 50, and a cathode 60 are laminated on a substrate 10 in this order.
- the hole transport zone 30 means a hole transport layer or a hole injection layer.
- the electron transport zone 50 means an electron transport layer, an electron injection layer, or the like. These need not be formed, but preferably one or more layers are formed.
- the organic thin film layer is each organic layer provided in the hole transport zone 30, each phosphor layer and the organic layer provided in the electron transport zone 50.
- At least one layer contains the organic EL element material of the present invention. Thereby, the drive voltage of an organic EL element can be lowered.
- the content of this material with respect to the organic thin film layer containing the organic EL device material of the present invention is preferably 1 to 100% by weight.
- the phosphorescent light emitting layer 40 preferably contains the material for the organic EL device of the present invention, and particularly preferably used as a host material for the light emitting layer. Since the triplet energy of the material of the present invention is sufficiently large, even when a blue phosphorescent dopant material is used, the triplet energy of the phosphorescent dopant material can be efficiently confined in the light emitting layer. In addition, it can be used not only for the blue light emitting layer but also for a light emitting layer of longer wavelength light (such as green to red).
- the phosphorescent light emitting layer contains a phosphorescent material (phosphorescent dopant).
- phosphorescent dopants include metal complex compounds, preferably selected from iridium (Ir), platinum (Pt), osmium (Os), gold (Au), copper (Cu), rhenium (Re), and ruthenium (Ru).
- the phosphorescent dopant is preferably a compound containing a metal atom selected from Ir, Os and Pt in that the phosphorescent quantum yield is high and the external quantum efficiency of the light-emitting element can be further improved, and an iridium complex, It is more preferable that it is a metal complex such as an osmium complex and a platinum complex, among which an iridium complex and a platinum complex are more preferable, and an orthometalated iridium complex is most preferable.
- the dopant may be a single type or a mixture of two or more types.
- the addition concentration of the phosphorescent dopant in the phosphorescent light emitting layer is not particularly limited, but is preferably 0.1 to 30% by weight (wt%), more preferably 0.1 to 20% by weight (wt%).
- the material of the present invention in a layer adjacent to the phosphorescent light emitting layer 40.
- a layer containing the material of the present invention an anode side adjacent layer
- the layer functions as an electron barrier layer. It functions as an exciton blocking layer.
- the layer when a layer (cathode side adjacent layer) containing the material of the present invention is formed between the phosphorescent light emitting layer 40 and the electron transport zone 50, the layer functions as a hole blocking layer or as an exciton blocking layer. It has a function.
- the barrier layer is a layer having a function of a carrier movement barrier or an exciton diffusion barrier.
- the organic layer for preventing electrons from leaking from the light-emitting layer to the hole transport zone is mainly defined as an electron barrier layer, and the organic layer for preventing holes from leaking from the light-emitting layer to the electron transport zone is defined as a hole barrier. Sometimes defined as a layer.
- an exciton blocking layer is an organic layer for preventing triplet excitons generated in the light emitting layer from diffusing into a peripheral layer having triplet energy lower than that of the light emitting layer. It may be defined as Further, the material of the present invention can be used for a layer adjacent to the phosphorescent light emitting layer 40 and further used for another organic thin film layer bonded to the adjacent layer.
- FIG. 2 is a schematic view showing the layer structure of another embodiment of the organic EL device of the present invention.
- the organic EL element 2 is an example of a hybrid type organic EL element in which a phosphorescent light emitting layer and a fluorescent light emitting layer are laminated.
- the organic EL element 2 has the same configuration as the organic EL element 1 except that a space layer 42 and a fluorescent light emitting layer 44 are formed between the phosphorescent light emitting layer 40 and the electron transport zone 50.
- the excitons formed in the phosphorescent light emitting layer 40 are not diffused into the fluorescent light emitting layer 44, so that a space layer 42 is provided between the fluorescent light emitting layer 44 and the phosphorescent light emitting layer 40. May be provided. Since the material of the present invention has a large triplet energy, it can function as a space layer.
- a white light emitting organic EL element can be obtained by setting the phosphorescent light emitting layer to emit yellow light and the fluorescent light emitting layer to blue light emitting layer.
- the phosphorescent light-emitting layer and the fluorescent light-emitting layer are formed one by one.
- the present invention is not limited to this, and two or more layers may be formed, and can be appropriately set according to the application such as lighting and display device.
- a full color light emitting device is formed using a white light emitting element and a color filter
- a plurality of wavelength regions such as red, green, blue (RGB), red, green, blue, yellow (RGBY) are used from the viewpoint of color rendering. In some cases, it may be preferable to include luminescence.
- the organic EL element of the present invention can employ various known configurations. Further, light emission of the light emitting layer can be taken out from the anode side, the cathode side, or both sides.
- the organic EL device of the present invention preferably has at least one of an electron donating dopant and an organometallic complex in an interface region between the cathode and the organic thin film layer. According to such a configuration, it is possible to improve the light emission luminance and extend the life of the organic EL element.
- the electron donating dopant include at least one selected from alkali metals, alkali metal compounds, alkaline earth metals, alkaline earth metal compounds, rare earth metals, rare earth metal compounds, and the like.
- the organometallic complex include at least one selected from an organometallic complex containing an alkali metal, an organometallic complex containing an alkaline earth metal, an organometallic complex containing a rare earth metal, and the like.
- alkali metal examples include lithium (Li) (work function: 2.93 eV), sodium (Na) (work function: 2.36 eV), potassium (K) (work function: 2.28 eV), rubidium (Rb) (work Function: 2.16 eV), cesium (Cs) (work function: 1.95 eV) and the like, and those having a work function of 2.9 eV or less are particularly preferable.
- K, Rb, and Cs are preferred, Rb and Cs are more preferred, and Cs is most preferred.
- alkaline earth metal examples include calcium (Ca) (work function: 2.9 eV), strontium (Sr) (work function: 2.0 eV to 2.5 eV), barium (Ba) (work function: 2.52 eV).
- a work function of 2.9 eV or less is particularly preferable.
- the rare earth metal examples include scandium (Sc), yttrium (Y), cerium (Ce), terbium (Tb), ytterbium (Yb) and the like, and those having a work function of 2.9 eV or less are particularly preferable.
- preferred metals are particularly high in reducing ability, and by adding a relatively small amount to the electron injection region, it is possible to improve the light emission luminance and extend the life of the organic EL element.
- alkali metal compound examples include lithium oxide (Li 2 O), cesium oxide (Cs 2 O), alkali oxides such as potassium oxide (K 2 O), lithium fluoride (LiF), sodium fluoride (NaF), fluorine.
- alkali halides such as cesium fluoride (CsF) and potassium fluoride (KF), and lithium fluoride (LiF), lithium oxide (Li 2 O), and sodium fluoride (NaF) are preferable.
- alkaline earth metal compound examples include barium oxide (BaO), strontium oxide (SrO), calcium oxide (CaO), and barium strontium oxide (Ba x Sr 1-x O) (0 ⁇ x ⁇ 1), Examples thereof include barium calcium oxide (Ba x Ca 1-x O) (0 ⁇ x ⁇ 1), and BaO, SrO, and CaO are preferable.
- the rare earth metal compound ytterbium fluoride (YbF 3), scandium fluoride (ScF 3), scandium oxide (ScO 3), yttrium oxide (Y 2 O 3), cerium oxide (Ce 2 O 3), gadolinium fluoride (GdF 3), include such terbium fluoride (TbF 3) is, YbF 3, ScF 3, TbF 3 are preferable.
- the organometallic complex is not particularly limited as long as it contains at least one of an alkali metal ion, an alkaline earth metal ion, and a rare earth metal ion as a metal ion as described above.
- the ligands include quinolinol, benzoquinolinol, acridinol, phenanthridinol, hydroxyphenyloxazole, hydroxyphenylthiazole, hydroxydiaryloxadiazole, hydroxydiarylthiadiazole, hydroxyphenylpyridine, hydroxyphenylbenzimidazole, hydroxybenzotriazole, Hydroxyfulborane, bipyridyl, phenanthroline, phthalocyanine, porphyrin, cyclopentadiene, ⁇ -diketones, azomethines, and derivatives thereof are preferred, but are not limited thereto.
- the electron donating dopant and the organometallic complex it is preferable to form a layer or an island in the interface region.
- a forming method while depositing at least one of an electron donating dopant and an organometallic complex by a resistance heating vapor deposition method, an organic material as a light emitting material or an electron injection material for forming an interface region is simultaneously deposited, and an electron is deposited in the organic material.
- a method of dispersing at least one of the donor dopant and the organometallic complex is preferable.
- the dispersion concentration is usually organic substance: electron donating dopant and / or organometallic complex in a molar ratio of 100: 1 to 1: 100, preferably 5: 1 to 1: 5.
- At least one of the electron donating dopant and the organometallic complex is formed in a layered form
- at least one of the electron donating dopant and the organometallic complex is formed.
- These are vapor-deposited by a resistance heating vapor deposition method alone, preferably with a layer thickness of 0.1 nm to 15 nm.
- an electron donating dopant and an organometallic complex is formed in an island shape
- a light emitting material or an electron injecting material which is an organic layer at the interface is formed in an island shape, and then the electron donating dopant and the organometallic complex are formed. At least one of them is vapor-deposited by a resistance heating vapor deposition method, preferably with an island thickness of 0.05 nm to 1 nm.
- the configuration other than the layer using the organic EL element material of the present invention described above is not particularly limited, and a known material or the like can be used.
- a known material or the like can be used.
- the layer of the element of Embodiment 1 is demonstrated easily, the material applied to the organic EL element of this invention is not limited to the following.
- a glass plate, a polymer plate or the like can be used as the substrate.
- the glass plate include soda lime glass, barium / strontium-containing glass, lead glass, aluminosilicate glass, borosilicate glass, barium borosilicate glass, and quartz.
- the polymer plate include polycarbonate, acrylic, polyethylene terephthalate, polyether sulfone, and polysulfone.
- the anode is made of, for example, a conductive material, and a conductive material having a work function larger than 4 eV is suitable.
- the conductive material include carbon, aluminum, vanadium, iron, cobalt, nickel, tungsten, silver, gold, platinum, palladium, and their alloys, ITO substrate, tin oxide used for NESA substrate, indium oxide, and the like.
- examples thereof include metal oxides and organic conductive resins such as polythiophene and polypyrrole.
- the anode may be formed with a layer structure of two or more layers if necessary.
- the cathode is made of, for example, a conductive material, and a conductive material having a work function smaller than 4 eV is suitable.
- the conductive material include, but are not limited to, magnesium, calcium, tin, lead, titanium, yttrium, lithium, ruthenium, manganese, aluminum, lithium fluoride, and alloys thereof.
- the alloy include magnesium / silver, magnesium / indium, lithium / aluminum, and the like, but are not limited thereto.
- the ratio of the alloy is controlled by the temperature of the vapor deposition source, the atmosphere, the degree of vacuum, etc., and is selected to an appropriate ratio.
- the cathode may be formed with a layer structure of two or more layers, and the cathode can be produced by forming a thin film from the conductive material by a method such as vapor deposition or sputtering.
- the transmittance of the cathode for light emission is preferably greater than 10%.
- the sheet resistance as the cathode is preferably several hundred ⁇ / ⁇ or less, and the film thickness is usually 10 nm to 1 ⁇ m, preferably 50 to 200 nm.
- the phosphorescent light emitting layer is formed of a material other than the organic EL element layer material of the present invention
- a known material can be used as the material of the phosphorescent light emitting layer.
- International Publication No. 2005/079118 Japanese Patent Application No. 2005-517938
- the organic EL device of the present invention may have a fluorescent light emitting layer like the device shown in FIG.
- a known material can be used for the fluorescent light emitting layer.
- the light emitting layer may be a double host (also referred to as a host / cohost). Specifically, the carrier balance in the light emitting layer may be adjusted by combining an electron transporting host and a hole transporting host in the light emitting layer. Moreover, it is good also as a double dopant.
- each dopant emits light by adding two or more dopant materials having a high quantum yield. For example, a yellow light emitting layer may be realized by co-evaporating a host, a red dopant, and a green dopant.
- the light emitting layer may be a single layer or a laminated structure. When the light emitting layer is stacked, the recombination region can be concentrated on the light emitting layer interface by accumulating electrons and holes at the light emitting layer interface. This improves the quantum efficiency.
- the hole injection / transport layer is a layer that assists hole injection into the light emitting layer and transports it to the light emitting region, and has a high hole mobility and a small ionization energy of usually 5.6 eV or less.
- As the material for the hole injection / transport layer a material that transports holes to the light emitting layer with lower electric field strength is preferable. Further, when an electric field is applied with a hole mobility of, for example, 10 4 to 10 6 V / cm, At least 10 ⁇ 4 cm 2 / V ⁇ sec is preferable.
- the material for the hole injection / transport layer include triazole derivatives (see US Pat. No. 3,112,197) and oxadiazole derivatives (US Pat. No. 3,189,447). ), Imidazole derivatives (see Japanese Patent Publication No. 37-16096, etc.), polyarylalkane derivatives (US Pat. Nos. 3,615,402, 3,820,989, 3, No. 542,544, JP-B-45-555, JP-A-51-10983, JP-A-51-93224, JP-A-55-17105, JP-A-56-4148, JP-A-55-108667.
- a cross-linkable material can be used as the material of the hole injection / transport layer.
- a cross-linkable hole injection / transport layer for example, Chem. Mater. 2008, 20, 413-422, Chem. Mater. 2011, 23 (3), 658-681, International Publication No. 2008/108430, International Publication No. 2009/102027, International Publication No. 2009/123269, International Publication No. 2010/016555, International Publication No. 2010/018813.
- a layer obtained by insolubilizing a cross-linking material such as heat or light.
- the electron injection / transport layer is a layer that assists the injection of electrons into the light emitting layer and transports it to the light emitting region, and has a high electron mobility.
- an electrode for example, a cathode
- the electron injecting / transporting layer is appropriately selected with a film thickness of several nm to several ⁇ m.
- the electron mobility is preferably at least 10 ⁇ 5 cm 2 / Vs or more when an electric field of V / cm is applied.
- an aromatic heterocyclic compound containing one or more heteroatoms in the molecule is preferably used, and a nitrogen-containing ring derivative is particularly preferable.
- the nitrogen-containing ring derivative is preferably an aromatic ring having a nitrogen-containing 6-membered ring or 5-membered ring skeleton, or a condensed aromatic ring compound having a nitrogen-containing 6-membered ring or 5-membered ring skeleton, such as a pyridine ring. , Pyrimidine ring, triazine ring, benzimidazole ring, phenanthroline ring, quinazoline ring and the like.
- an organic layer having semiconductivity may be formed by doping (n) with a donor material and doping (p) with an acceptor material.
- N doping is to dope an electron transporting material with a metal such as Li or Cs
- P doping is F4TCNQ (2,3,5,6-tetrafluoro) to a hole transporting material. -7,7,8,8-tetracyanoquinodimethane) or the like (see, for example, Japanese Patent No. 3695714).
- each layer of the organic EL device of the present invention a known method such as a dry film forming method such as vacuum deposition, sputtering, plasma, or ion plating, or a wet film forming method such as spin coating, dipping, or flow coating is applied. be able to.
- the thickness of each layer is not particularly limited, but must be set to an appropriate thickness. If the film thickness is too thick, a large applied voltage is required to obtain a constant light output, resulting in poor efficiency. If the film thickness is too thin, pinholes and the like are generated, and sufficient light emission luminance cannot be obtained even when an electric field is applied.
- the normal film thickness is suitably in the range of 5 nm to 10 ⁇ m, but more preferably in the range of 10 nm to 0.2 ⁇ m.
- triplet energy (E T ) The measurement was performed using a commercially available apparatus F-4500 (manufactured by Hitachi).
- the conversion formula of triplet energy (E T ) is as follows.
- E T (eV) 1239.85 / ⁇ ph
- ⁇ ph unit: nm
- the wavelength value of the intersection of the tangent and the horizontal axis The wavelength value of the intersection of the tangent and the horizontal axis.
- the phosphorescence measurement sample placed in the quartz cell was cooled to 77 (K), and the phosphorescence measurement sample was irradiated with excitation light, and the phosphorescence intensity was measured while changing the wavelength.
- the vertical axis represents phosphorescence intensity and the horizontal axis represents wavelength.
- a tangent line was drawn with respect to the rising edge of the phosphorescence spectrum on the short wavelength side, and the wavelength value ⁇ ph (nm) at the intersection of the tangent line and the horizontal axis was obtained.
- the tangent to the rising edge on the short wavelength side of the phosphorescence spectrum is drawn as follows. When moving on the spectrum curve from the short wavelength side of the phosphorescence spectrum to the maximum value on the shortest wavelength side among the maximum values of the spectrum, tangents at each point on the curve are considered toward the long wavelength side. The slope of this tangent increases as the curve rises (that is, as the vertical axis increases). The tangent drawn at the point where the slope value takes the maximum value is taken as the tangent to the rising edge of the phosphorescence spectrum on the short wavelength side.
- the maximum point having a peak intensity of 10% or less of the maximum peak intensity of the spectrum is not included in the above-mentioned maximum value on the shortest wavelength side, and has the maximum slope value closest to the maximum value on the shortest wavelength side.
- the tangent drawn at the point where the value is taken is taken as the tangent to the rising edge of the phosphorescence spectrum on the short wavelength side.
- the evaluation method of an organic EL element is as follows.
- (1) External quantum efficiency (%) The external quantum efficiency at a luminance of 1000 cd / m 2 under a dry nitrogen gas atmosphere at 23 ° C. was measured using a luminance meter (Spectral Luminance Radiometer CS-1000 manufactured by Minolta).
- the compound was identified by FD-MS (field desorption mass spectrometry) and 1 H-NMR (proton nuclear magnetic resonance method). The yield was 73.9 g, and the yield was 69%.
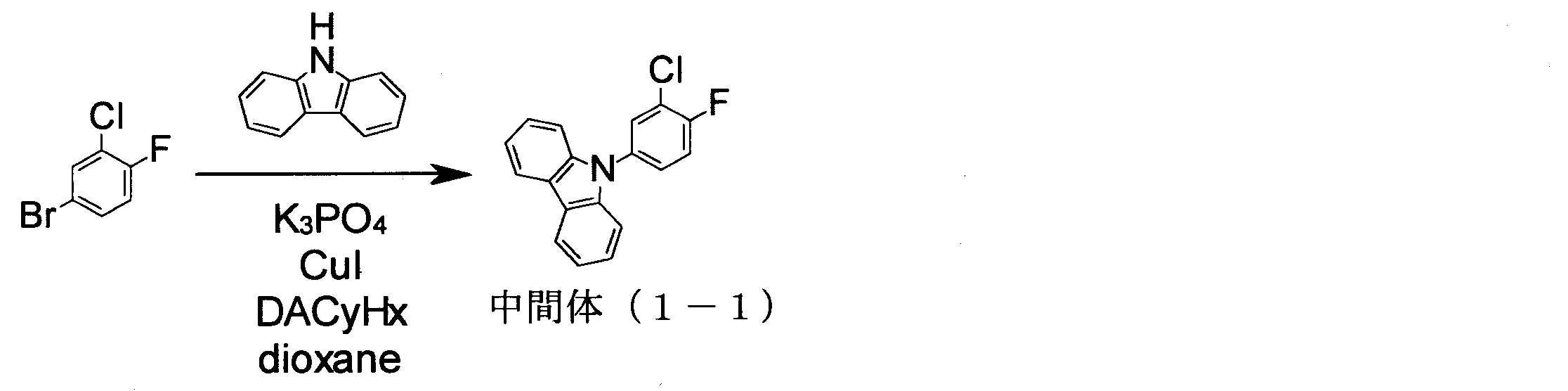
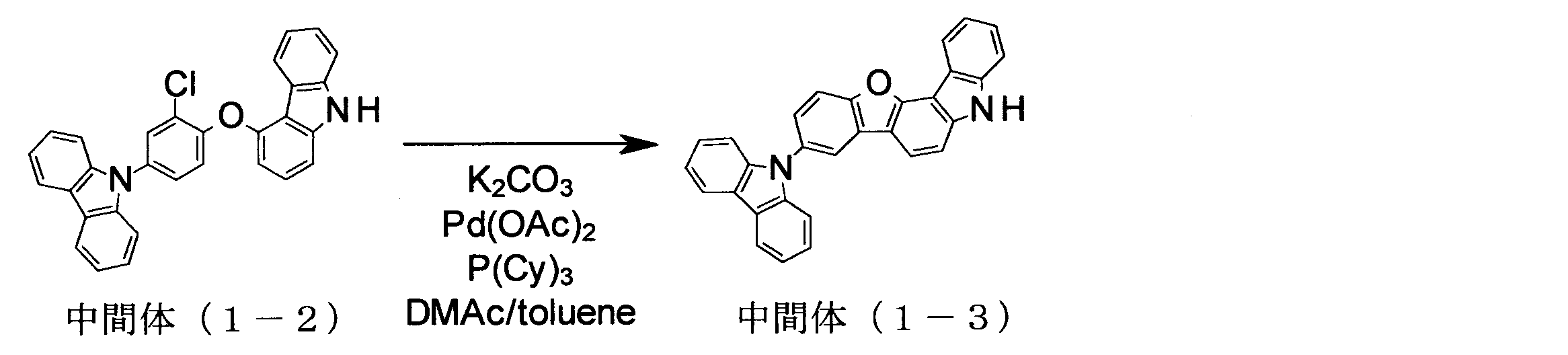
- intermediate (1-1) 70.98 g, 240 mmol
- 4-hydroxycarbazole 48.37 g, 264 mmol
- K 2 CO 3 66.34 g, 480 mmol
- NMP N-methylpyrrolidone
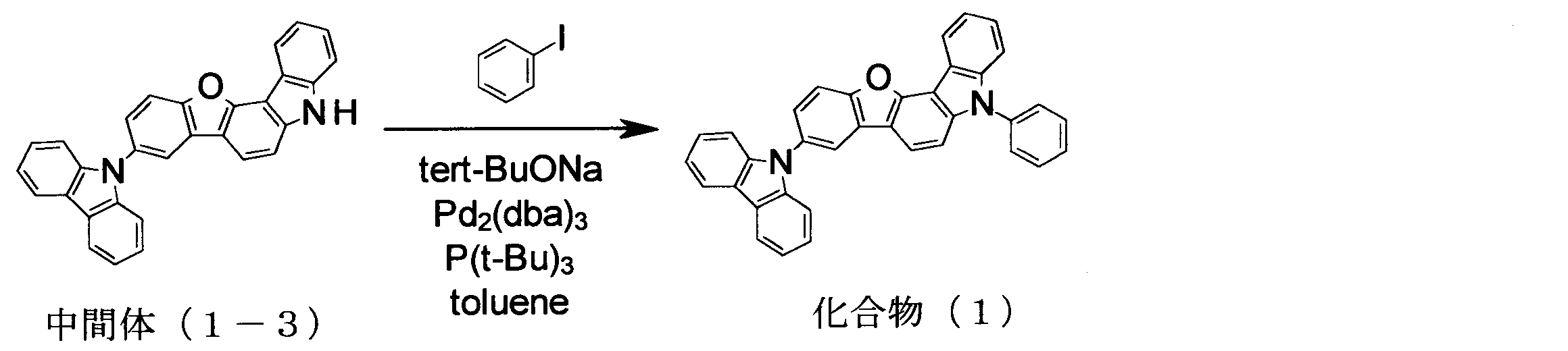
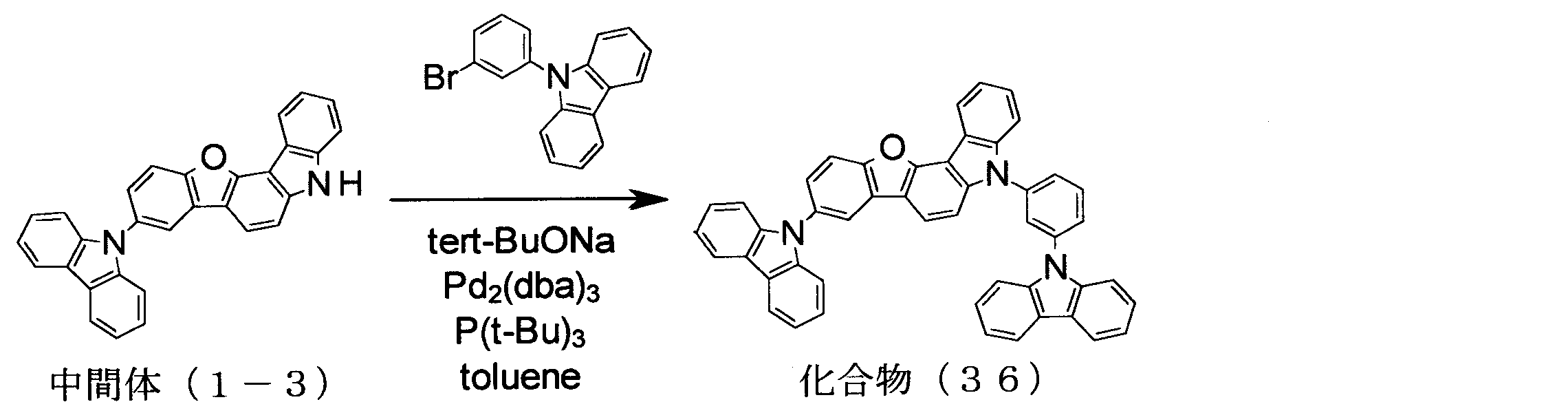
- intermediate (5-1) 136.0 g, 365 mmol
- iodobenzene 148.9 g, 730 mmol
- K 3 PO 4 116.2 g, 547.5 mmol
- CuI 3.48 g, 18. 25 mmol
- trans-1,2-cyclohexanediamine 6.6 ml, 54.8 mmol
- 1,4-dioxane 365 ml
- the compound was identified by FD-MS and 1 H-NMR. The yield was 127.7 g, and the yield was 78%.
- the compound was identified by FD-MS and 1 H-NMR. The yield was 7.5 g, and the yield was 97%.
- Example 1 A glass substrate with a 130 nm-thick ITO electrode line (manufactured by Geomatec) was ultrasonically cleaned in isopropyl alcohol for 5 minutes, and then UV ozone cleaning was performed for 30 minutes.
- the glass substrate with the ITO electrode line after the cleaning is mounted on the substrate holder of the vacuum deposition apparatus, and the compound (HI1) is first thickened so as to cover the ITO electrode line on the surface on which the ITO electrode line is formed.
- the compound (HT1) was deposited by resistance heating at a thickness of 60 nm at a thickness of 20 nm, and thin films were sequentially formed. The film formation rate was 1 ⁇ / s. These thin films function as a hole injection layer and a hole transport layer, respectively.
- the compound (1) and the compound (BD1) were simultaneously deposited by resistance heating to form a thin film having a thickness of 50 nm.
- the compound (BD1) was deposited so as to have a mass ratio of 20% with respect to the total mass of the compound (A1) and the compound (BD1).
- the film formation rates were 1.2 ⁇ / s and 0.3 ⁇ / s, respectively.
- This thin film functions as a phosphorescent light emitting layer, in which the compound (1) functions as a host and the compound (BD1) functions as a light emitting dopant.
- Table 1 shows triplet energy (eV) and glass transition point (° C.) of the compound (BD1).
- a compound (B1) was deposited by resistance heating vapor deposition on this phosphorescent light emitting layer to form a thin film having a thickness of 10 nm (hole blocking layer).
- the film formation rate was 1.2 liter / s.
- a thin film having a thickness of 10 nm was formed on the hole barrier layer by resistance heating evaporation of the compound (ET1).
- the film formation rate was 1 ⁇ / s.
- This film functions as an electron injection layer.
- LiF having a film thickness of 1.0 nm was deposited on the electron injection layer at a film formation rate of 0.1 ⁇ / s.
- metallic aluminum was vapor-deposited on the LiF film at a deposition rate of 8.0 ⁇ / s to form a metal cathode with a film thickness of 80 nm to obtain an organic EL element.
- the voltage and half-life were calculated
- Examples 2 to 14 and Comparative Examples 1 to 2 An organic EL device was prepared and evaluated in the same manner as in Example 1 except that the compound shown in Table 1 was used as the host of the phosphorescent light emitting layer instead of the compound (1).
- Table 1 shows the triplet energy and glass transition point of the compound used, and Table 2 shows the results.
- the “half life (relative%)” is a relative ratio when the half life of the element of Comparative Example 1 is 100%.
- Example 15 A glass substrate with a 130 nm-thick ITO electrode line (manufactured by Geomatec) was ultrasonically cleaned in isopropyl alcohol for 5 minutes, and then UV ozone cleaning was performed for 30 minutes.
- the glass substrate with the ITO electrode line after the cleaning is mounted on the substrate holder of the vacuum deposition apparatus, and the compound (HI1) is first thickened so as to cover the ITO electrode line on the surface on which the ITO electrode line is formed.
- the compound (HT1) was then vapor deposited by resistance heating at a thickness of 50 nm, and thin films were sequentially formed.
- the film formation rate was 1 ⁇ / s.
- These thin films function as a hole injection layer and a hole transport layer, respectively.
- the compound (39) was deposited by resistance heating vapor deposition on the hole transport layer to form a thin film (electronic barrier layer) having a thickness of 10 nm.
- the film formation rate was 1 ⁇ / s.
- the compound (B1) and the compound (BD1) were simultaneously deposited by resistance heating to form a thin film having a thickness of 50 nm.
- the compound (BD1) was deposited so as to have a mass ratio of 20% with respect to the total mass of the compound (B1) and the compound (BD1).
- the film formation rates were 1.2 ⁇ / s and 0.3 ⁇ / s, respectively.
- This thin film functions as a phosphorescent light emitting layer, and in this thin film, the compound (B1) functions as a host and the compound (BD1) functions as a light emitting dopant.
- a compound (B1) was deposited by resistance heating vapor deposition on this phosphorescent light emitting layer to form a thin film having a thickness of 10 nm (hole blocking layer).
- the film formation rate was 1.2 liter / s.
- a thin film having a thickness of 10 nm was formed on the hole barrier layer by resistance heating evaporation of the compound (ET1).
- the film formation rate was 1 ⁇ / s.
- This film functions as an electron injection layer.
- LiF having a film thickness of 1.0 nm was deposited on the electron injection layer at a film formation rate of 0.1 ⁇ / s.
- metallic aluminum was vapor-deposited on the LiF film at a deposition rate of 8.0 ⁇ / s to form a metal cathode with a film thickness of 80 nm to obtain an organic EL element.
- the voltage, external quantum efficiency, and half life were determined by the above methods. The results are shown in Table 3.
- Examples 16 to 18 and Comparative Examples 3 to 4 An organic EL device was prepared and evaluated in the same manner as in Example 15 except that the compound shown in Table 3 was used as the electron barrier layer instead of the compound (39). The results are shown in Table 3.
- Example 19 Organic EL in the same manner as in Example 1 except that compound (B1) was used instead of compound (1) as the host of the phosphorescent layer, and compound (1) was used instead of compound (B1) as the hole barrier layer. A device was fabricated and evaluated. The results are shown in Table 4. Examples 20 to 27 and Comparative Example 5 An organic EL device was prepared and evaluated in the same manner as in Example 11 except that the compounds listed in Table 4 were used as the hole blocking layer instead of the compound (1). The results are shown in Tables 4 and 5.
- Table 4 shows that when the compound of the present invention is used as a hole blocking layer, an organic EL device having a longer life than the compound of the comparative example can be provided.
- Table 5 shows that when the compound of the present invention is used as a hole blocking layer, an organic EL device having a lower voltage and higher efficiency than the compound of the comparative example can be provided.
- the compound of the present invention can be used as a material for an organic EL device.
- an organic EL element material of the present invention is used, an organic EL element having a long life and low power consumption for low voltage driving can be obtained.
- the organic EL element of the present invention is extremely useful as a display, a light source and the like for various electronic devices.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Electroluminescent Light Sources (AREA)
Description
本発明は、有機エレクトロルミネッセンス素子用材料及びそれを用いた有機エレクトロルミネッセンス素子に関する。 The present invention relates to a material for an organic electroluminescence element and an organic electroluminescence element using the same.
有機エレクトロルミネッセンス(EL)素子には、蛍光型及び燐光型があり、それぞれの発光メカニズムに応じ、最適な素子設計が検討されている。燐光型の有機EL素子については、その発光特性から、蛍光素子技術の単純な転用では高性能な素子が得られないことが知られている。その理由は、一般的に以下のように考えられている。
まず、燐光発光は、三重項励起子を利用した発光であるため、発光層に用いる化合物のエネルギーギャップが大きくなくてはならない。何故なら、ある化合物のエネルギーギャップ(以下、一重項エネルギーともいう。)の値は、通常、その化合物の三重項エネルギー(本発明では、最低励起三重項状態と基底状態とのエネルギー差をいう。)の値よりも大きいからである。
Organic electroluminescence (EL) elements include a fluorescent type and a phosphorescent type, and an optimum element design has been studied according to each light emission mechanism. With respect to phosphorescent organic EL elements, it is known from their light emission characteristics that high-performance elements cannot be obtained by simple diversion of fluorescent element technology. The reason is generally considered as follows.
First, since phosphorescence emission is emission using triplet excitons, the energy gap of the compound used for the light emitting layer must be large. This is because the value of the energy gap (hereinafter also referred to as singlet energy) of a compound usually refers to the triplet energy of the compound (in the present invention, the energy difference between the lowest excited triplet state and the ground state). This is because it is larger than the value of).
従って、燐光発光性ドーパント材料の三重項エネルギーを効率的に発光層内に閉じ込めるためには、まず、燐光発光性ドーパント材料の三重項エネルギーよりも大きい三重項エネルギーのホスト材料を発光層に用いなければならない。さらに、発光層に隣接する電子輸送層、及び正孔輸送層を設け、電子輸送層、及び正孔輸送層に燐光発光性ドーパント材料の三重項エネルギーよりも大きい化合物を用いなければならない。
このように、従来の有機EL素子の素子設計思想に基づく場合、蛍光型の有機EL素子に用いる化合物と比べて大きなエネルギーギャップを有する化合物を燐光型の有機EL素子に用いることにつながり、有機EL素子全体の駆動電圧が上昇する。
Therefore, in order to efficiently confine the triplet energy of the phosphorescent dopant material in the light emitting layer, a host material having a triplet energy larger than the triplet energy of the phosphorescent dopant material must first be used for the light emitting layer. I must. Furthermore, an electron transport layer and a hole transport layer adjacent to the light emitting layer are provided, and a compound having a triplet energy higher than that of the phosphorescent dopant material must be used for the electron transport layer and the hole transport layer.
Thus, when based on the element design concept of the conventional organic EL element, a compound having a larger energy gap than the compound used for the fluorescent organic EL element is used for the phosphorescent organic EL element. The drive voltage of the entire element increases.
また、蛍光素子で有用であった酸化耐性や還元耐性の高い炭化水素系の化合物はπ電子雲の広がりが大きいため、エネルギーギャップが小さい。そのため、燐光型の有機EL素子では、このような炭化水素系の化合物が選択され難く、酸素や窒素等のヘテロ原子を含んだ有機化合物が選択され、その結果、燐光型の有機EL素子は、蛍光型の有機EL素子と比較して寿命が短いという問題を有する。 In addition, hydrocarbon compounds having high oxidation resistance and reduction resistance useful for fluorescent elements have a large energy gap due to the large spread of π electron clouds. Therefore, in a phosphorescent organic EL element, it is difficult to select such a hydrocarbon compound, and an organic compound containing a heteroatom such as oxygen or nitrogen is selected. As a result, the phosphorescent organic EL element is There is a problem that the lifetime is shorter than that of a fluorescent organic EL element.
さらに、燐光発光性ドーパント材料の三重項励起子の励起子緩和速度が一重項励起子と比較して非常に長いことも素子性能に大きな影響を与える。即ち、一重項励起子からの発光は、発光に繋がる緩和速度が速いため、発光層の周辺層(例えば、正孔輸送層や電子輸送層)への励起子の拡散が起きにくく、効率的な発光が期待される。一方、三重項励起子からの発光は、スピン禁制であり緩和速度が遅いため、周辺層への励起子の拡散が起きやすく、特定の燐光発光性化合物以外からは熱的なエネルギー失活が起きてしまう。つまり、電子、及び正孔の再結合領域のコントロールが蛍光型の有機EL素子よりも重要である。 Furthermore, the fact that the exciton relaxation rate of the triplet exciton of the phosphorescent dopant material is much longer than that of the singlet exciton also greatly affects the device performance. That is, since light emitted from singlet excitons has a high relaxation rate that leads to light emission, it is difficult for excitons to diffuse into the peripheral layer of the light emitting layer (for example, a hole transport layer or an electron transport layer). Light emission is expected. On the other hand, light emission from triplet excitons is spin-forbidden and has a slow relaxation rate, so that excitons are likely to diffuse into the peripheral layer, and thermal energy deactivation occurs from other than specific phosphorescent compounds. End up. That is, control of the recombination region of electrons and holes is more important than the fluorescent organic EL element.
以上のような理由から燐光型の有機EL素子の高性能化には、蛍光型の有機EL素子と異なる材料選択、及び素子設計が必要になっている。
特に、青色発光する燐光型の有機EL素子の場合、緑~赤色発光する燐光型の有機EL素子と比べて、発光層やその周辺層に三重項エネルギーが大きい化合物を使用する必要がある。具体的に、効率の損失無く青色の燐光発光を得るためには、発光層に使用するホスト材料の三重項エネルギーは概ね3.0eV以上が必要である。このような高い三重項エネルギーを有しながら、その他、有機EL材料として求められる性能を満たす化合物を得るためには、複素環化合物等の三重項エネルギーの高い分子パーツを単純に組み合わせるのではなく、π電子の電子状態を考慮した新たな思想による分子設計が必要になる。
For the above reasons, in order to improve the performance of phosphorescent organic EL elements, material selection and element design different from those of fluorescent organic EL elements are required.
In particular, in the case of a phosphorescent organic EL element that emits blue light, it is necessary to use a compound having a large triplet energy in the light emitting layer and its peripheral layer as compared with a phosphorescent organic EL element that emits green to red light. Specifically, in order to obtain blue phosphorescence without loss of efficiency, the triplet energy of the host material used for the light-emitting layer needs to be approximately 3.0 eV or more. In order to obtain a compound satisfying the performance required as an organic EL material while having such a high triplet energy, not simply combining molecular parts having a high triplet energy such as a heterocyclic compound, Molecular design based on a new concept that considers the electronic state of π electrons is required.
このような状況下、燐光型の有機EL素子の材料として、例えば、特許文献1には、中心ベンゼン環に2つのベンゼン環が縮合環を形成するように結合し、さらにその末端に他の縮合環が結合した構造を有する多環系化合物が開示されている。
また、特許文献2には、炭素原子、窒素原子、酸素原子又は硫黄原子で架橋したπ共役ヘテロアセン骨格を有する多環系化合物が開示されている。
Under such circumstances, as a material of a phosphorescent organic EL element, for example, in Patent Document 1, two benzene rings are bonded to a central benzene ring so as to form a condensed ring, and another condensed group is connected to the terminal. A polycyclic compound having a structure in which rings are bonded is disclosed.
Patent Document 2 discloses a polycyclic compound having a π-conjugated heteroacene skeleton bridged by a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom.
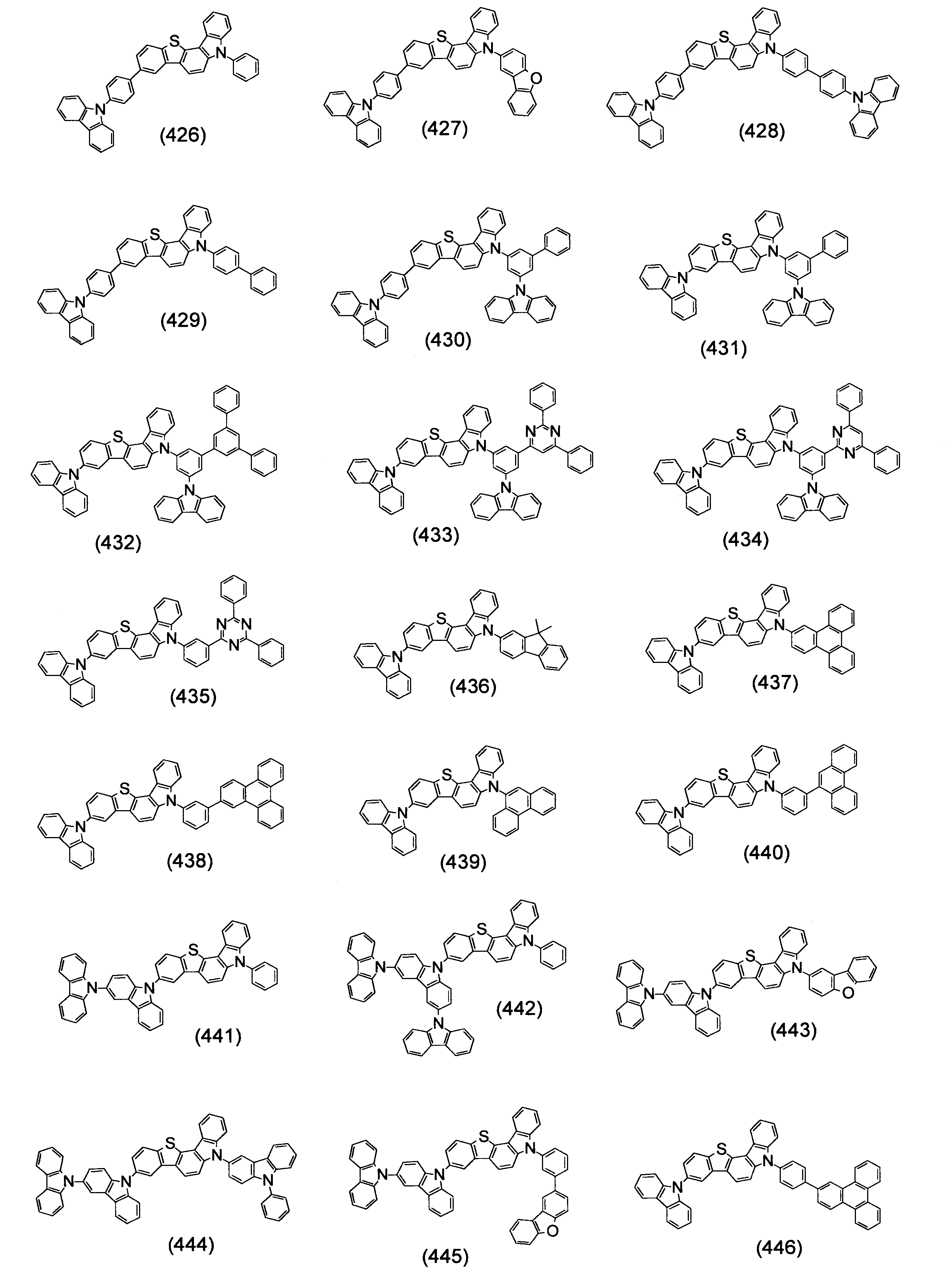
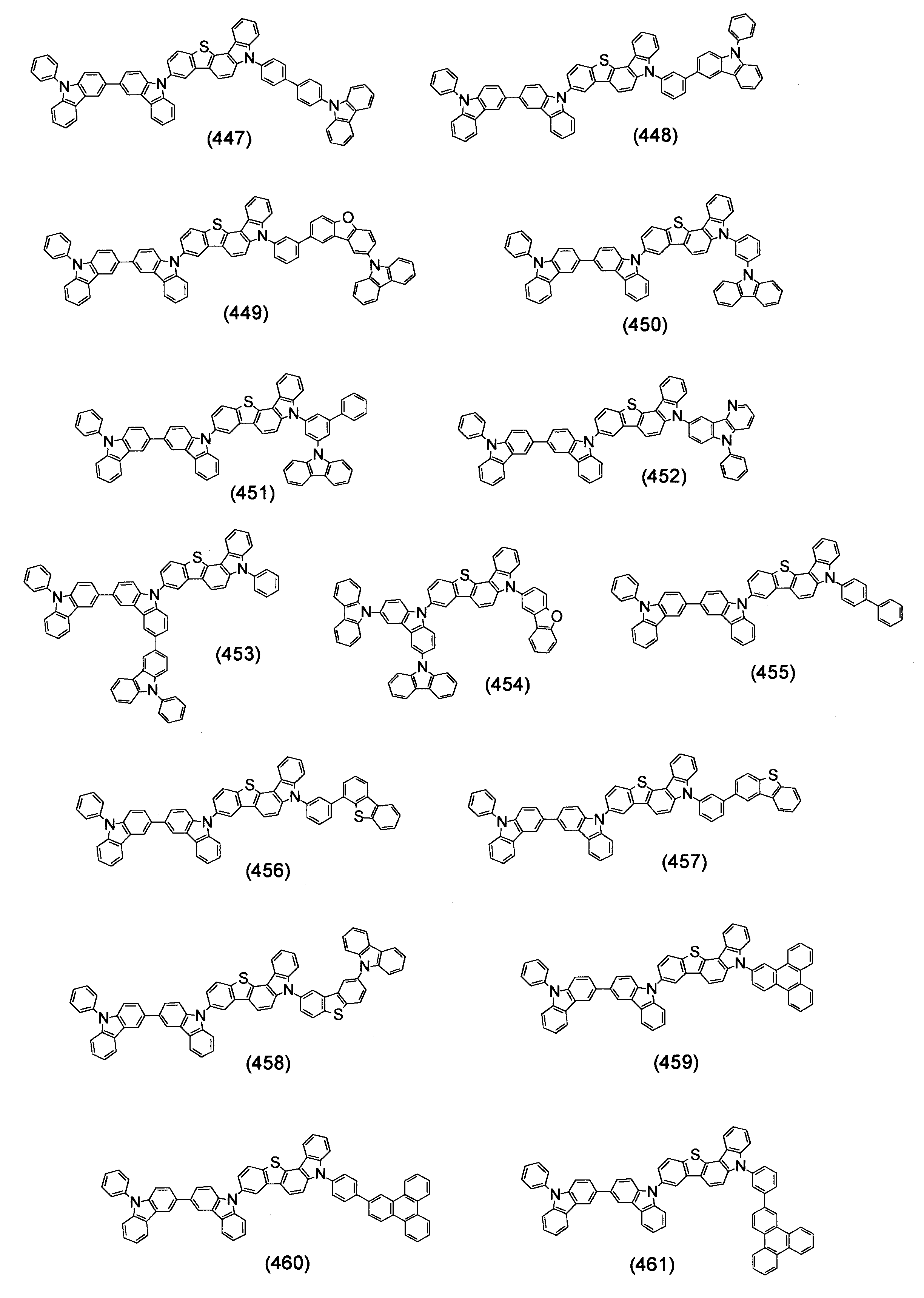
本発明の目的は、有機エレクトロルミネッセンス素子の長寿命化、低駆動電圧化が可能な有機エレクトロルミネッセンス素子用材料を提供することである。 An object of the present invention is to provide a material for an organic electroluminescence element capable of extending the life and lowering the driving voltage of the organic electroluminescence element.
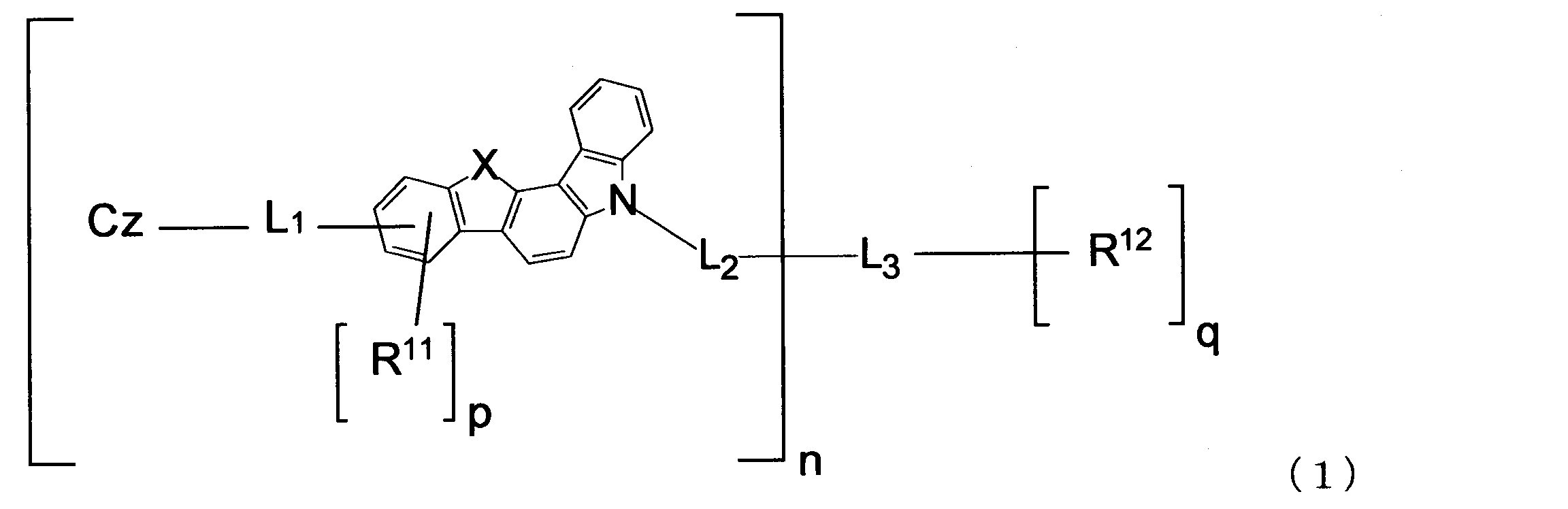
本発明によれば、以下の有機エレクトロルミネッセンス素子用材料等が提供される。
1.下記式(1)で表される有機エレクトロルミネッセンス素子用材料。
Xは、酸素(O)原子又は硫黄(S)原子であり、nが2以上の場合、複数のXはそれぞれ同一でも異なっていてもよい。
L1及びL2は、それぞれ、単結合、置換基Rを有してもよい環形成炭素数6~18のアリーレン基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリーレン基であり、nが2以上の場合、複数のL1及び複数のL2はそれぞれ同一でも異なっていてもよい。
L3は、置換基Rを有してもよい炭素数1~20のn+q価の飽和脂肪族炭化水素基、置換基Rを有してもよいn+q価のケイ素含有基、置換基Rを有してもよい環形成炭素数6~18のn+q価の芳香族炭化水素基、又は置換基Rを有してもよい環形成原子数5~18のn+q価の不飽和複素環基である。
R11及びR12は、それぞれ、置換基Rを有してもよい炭素数1~20のアルキル基、置換基Rを有してもよい環形成炭素数3~18のシクロアルキル基、置換基Rを有してもよい炭素数1~20のアルコキシ基、置換基Rを有してもよい環形成炭素数3~20のシクロアルコキシ基、置換基Rを有してもよい環形成炭素数6~18のアリールオキシ基、置換基Rを有してもよいシリル基、フルオロ基、シアノ基、置換基Rを有してもよい環形成炭素数6~18のアリール基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリール基であり、R11が複数存在する場合、それぞれ同一でも異なっていてもよく、R12が複数存在する場合、それぞれ同一でも異なっていてもよい。
pは、0~3の整数であり、nが2以上の場合、複数のpはそれぞれ同一でも異なっていてもよい。
qは、0~3の整数である。
置換基Rは、炭素数1~20のアルキル基、環形成炭素数3~18のシクロアルキル基、炭素数1~20のアルコキシ基、環形成炭素数3~20のシクロアルコキシ基、環形成炭素数6~18のアリールオキシ基、シリル基、フルオロ基、シアノ基、環形成炭素数6~18のアリール基、又は環形成原子数5~18のヘテロアリール基であり、Rが複数存在する場合、それぞれ同一でも異なっていてもよい。
Czは、下記式(2-1)、(2-2)、(3-1)、(3-2)、(4-1)、(4-2)、(5-1)、(5-2)、(6-1)、(6-2)、(7-1)、(7-2)、(8-1)、及び(8-2)からなる群より選ばれる一つの基であり、nが2以上の場合、複数のCzはそれぞれ同一でも異なっていてもよい。
Yは、酸素(O)原子又は硫黄(S)原子であり、nが2以上の場合、複数のYはそれぞれ同一でも異なっていてもよく、
m1及びm2は、それぞれ独立に、0~4の整数であり、
m3は、0~3の整数であり、
m4は、0~2の整数であり、
m5は、0~4の整数であり、
R0、R1、R2及びR3は、それぞれ独立に、置換基Rを有してもよい炭素数1~20のアルキル基、置換基Rを有してもよい環形成炭素数3~18のシクロアルキル基、置換基Rを有してもよい炭素数1~20のアルコキシ基、置換基Rを有してもよい環形成炭素数3~20のシクロアルコキシ基、置換基Rを有してもよい環形成炭素数6~18のアリールオキシ基、置換基Rを有してもよいシリル基、フルオロ基、シアノ基、置換基Rを有してもよい環形成炭素数6~18のアリール基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリール基であり、R0が複数存在する場合、それぞれ同一でも異なっていてもよく、R1が複数存在する場合、それぞれ同一でも異なっていてもよく、R2が複数存在する場合、それぞれ同一でも異なっていてもよく、R3が複数存在する場合、それぞれ同一でも異なっていてもよい。))
2.下記式(3)で表される1に記載の有機エレクトロルミネッセンス素子用材料。
3.前記Czが、式(2-1)で表される基である1又は2に記載の有機エレクトロルミネッセンス素子用材料。
4.前記Czが、式(2-2)で表される基である1又は2に記載の有機エレクトロルミネッセンス素子用材料。
5.前記nが、1又は2である1~4のいずれかに記載の有機エレクトロルミネッセンス素子用材料。
6.陰極と陽極の間に、発光層を含む1層以上の有機薄膜層を有し、前記有機薄膜層の少なくとも1層が、1~5のいずれかに記載の有機エレクトロルミネッセンス素子用材料を含有する有機エレクトロルミネッセンス素子。
7.前記発光層が、前記有機エレクトロルミネッセンス素子用材料を含有する6に記載の有機エレクトロルミネッセンス素子。
8.前記陽極と前記発光層の間に正孔輸送帯域を有し、前記正孔輸送帯域が前記有機エレクトロルミネッセンス素子用材料を含有する6に記載の有機エレクトロルミネッセンス素子。
9.前記陰極と前記発光層の間に電子輸送帯域を有し、前記電子輸送帯域が前記有機エレクトロルミネッセンス素子用材料を含有する6に記載の有機エレクトロルミネッセンス素子。
10.前記発光層が、燐光発光材料を含有し、前記燐光発光材料がイリジウム(Ir)、オスミウム(Os)及び白金(Pt)から選択される金属原子のオルトメタル化錯体である6~9のいずれかに記載の有機エレクトロルミネッセンス素子。
According to the present invention, the following materials for organic electroluminescence elements are provided.
1. The material for organic electroluminescent elements represented by following formula (1).
X is an oxygen (O) atom or a sulfur (S) atom. When n is 2 or more, the plurality of X may be the same or different.
L 1 and L 2 are each a single bond, an arylene group having 6 to 18 ring carbon atoms which may have a substituent R, or an arylene group having 5 to 18 ring atoms which may have a substituent R. When it is a heteroarylene group and n is 2 or more, the plurality of L 1 and the plurality of L 2 may be the same or different.
L 3 has an n + q valent saturated aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have a substituent R, an n + q valent silicon-containing group which may have a substituent R, and a substituent R. And an n + q valent aromatic hydrocarbon group having 6 to 18 ring carbon atoms, or an n + q valent unsaturated heterocyclic group having 5 to 18 ring atoms which may have a substituent R.
R 11 and R 12 are each an alkyl group having 1 to 20 carbon atoms that may have a substituent R, a cycloalkyl group having 3 to 18 ring carbon atoms that may have a substituent R, and a substituent. An alkoxy group having 1 to 20 carbon atoms which may have R, a cycloalkoxy group having 3 to 20 ring carbon atoms which may have a substituent R, and a ring forming carbon number which may have a substituent R 6-18 aryloxy group, silyl group optionally having substituent R, fluoro group, cyano group, aryl group having 6-18 ring forming carbon atoms optionally having substituent R, or substituent R A heteroaryl group having 5 to 18 ring atoms that may have a ring structure, and when there are a plurality of R 11 s , they may be the same or different, and when there are a plurality of R 12 s , they may be the same or different from each other. May be.
p is an integer of 0 to 3, and when n is 2 or more, the plurality of p may be the same or different.
q is an integer of 0 to 3.
The substituent R is an alkyl group having 1 to 20 carbon atoms, a cycloalkyl group having 3 to 18 ring carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms, or a ring forming carbon. An aryloxy group having 6 to 18 atoms, a silyl group, a fluoro group, a cyano group, an aryl group having 6 to 18 ring carbon atoms, or a heteroaryl group having 5 to 18 ring atoms, and when there are a plurality of R These may be the same or different.
Cz is represented by the following formulas (2-1), (2-2), (3-1), (3-2), (4-1), (4-2), (5-1), (5- 2) one group selected from the group consisting of (6-1), (6-2), (7-1), (7-2), (8-1), and (8-2) , N is 2 or more, the plurality of Cz may be the same or different.
Y is an oxygen (O) atom or a sulfur (S) atom, and when n is 2 or more, the plurality of Y may be the same or different,
m 1 and m 2 are each independently an integer of 0 to 4,
m 3 is an integer from 0 to 3,
m 4 is an integer from 0 to 2,
m 5 is an integer from 0 to 4,
R 0 , R 1 , R 2 and R 3 are each independently an alkyl group having 1 to 20 carbon atoms which may have a substituent R, or a ring having 3 to 3 carbon atoms which may have a substituent R. An cycloalkyl group having 18 carbon atoms which may have a substituent R, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms which may have a substituent R, and a substituent R; An aryloxy group having 6 to 18 ring carbon atoms, a silyl group optionally having substituent R, a fluoro group, a cyano group, and 6 to 18 ring carbon atoms optionally having substituent R Or a heteroaryl group having 5 to 18 ring atoms that may have a substituent R, and when a plurality of R 0 are present, they may be the same or different, and a plurality of R 1 are present. If you, or different and each identical, if R 2 there are a plurality, Re may be the same or different, respectively, when the R 3 there are a plurality may each be the same or different. ))
2. The material for organic electroluminescence elements according to 1, which is represented by the following formula (3).
3. 3. The material for an organic electroluminescent element according to 1 or 2, wherein Cz is a group represented by the formula (2-1).
4). 3. The material for an organic electroluminescence element according to 1 or 2, wherein Cz is a group represented by the formula (2-2).
5. 5. The material for an organic electroluminescent element according to any one of 1 to 4, wherein n is 1 or 2.
6). One or more organic thin film layers including a light emitting layer are provided between the cathode and the anode, and at least one of the organic thin film layers contains the material for an organic electroluminescent element according to any one of 1 to 5 Organic electroluminescence device.
7). 7. The organic electroluminescence device according to 6, wherein the light emitting layer contains the material for an organic electroluminescence device.
8). 7. The organic electroluminescence device according to 6, wherein the organic electroluminescence device has a hole transport zone between the anode and the light emitting layer, and the hole transport zone contains the material for an organic electroluminescence device.
9. 7. The organic electroluminescence device according to 6, wherein the organic electroluminescence device has an electron transport zone between the cathode and the light emitting layer, and the electron transport zone contains the material for an organic electroluminescence device.
10. Any of 6 to 9, wherein the light emitting layer contains a phosphorescent material, and the phosphorescent material is an orthometalated complex of a metal atom selected from iridium (Ir), osmium (Os), and platinum (Pt). The organic electroluminescent element of description.
本発明によれば、有機エレクトロルミネッセンス素子の長寿命化、低駆動電圧化が可能な有機エレクトロルミネッセンス素子用材料を提供できる。 According to the present invention, it is possible to provide a material for an organic electroluminescence element capable of extending the life of the organic electroluminescence element and reducing the driving voltage.
本発明の有機EL素子用材料は、下記式(1)で表される。
式(1)中、nは、1~4の整数(1、2、3又は4)である。nは、例えば1又は2である。
Xは、酸素(O)原子又は硫黄(S)原子であり、nが2以上の場合、複数のXはそれぞれ同一でも異なっていてもよい。
L1及びL2は、それぞれ、単結合、置換基Rを有してもよい環形成炭素数6~18のアリーレン基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリーレン基であり、nが2以上の場合、複数のL1及び複数のL2はそれぞれ同一でも異なっていてもよい。ここで、「環形成炭素」とは飽和環、不飽和環、又は芳香環を構成する炭素原子を意味し、「環形成原子」とは飽和環、不飽和環、又は芳香環を構成する原子を意味する。
また、本発明において、水素原子とは、中性子数が異なる同位体、即ち、軽水素(protium)、重水素(deuterium)、三重水素(tritium)、を包含する。
In the formula (1), n is an integer of 1 to 4 (1, 2, 3 or 4). n is 1 or 2, for example.
X is an oxygen (O) atom or a sulfur (S) atom. When n is 2 or more, the plurality of X may be the same or different.
L 1 and L 2 are each a single bond, an arylene group having 6 to 18 ring carbon atoms which may have a substituent R, or an arylene group having 5 to 18 ring atoms which may have a substituent R. When it is a heteroarylene group and n is 2 or more, the plurality of L 1 and the plurality of L 2 may be the same or different. Here, “ring-forming carbon” means a carbon atom constituting a saturated ring, unsaturated ring or aromatic ring, and “ring-forming atom” means an atom constituting a saturated ring, unsaturated ring or aromatic ring. Means.
In the present invention, the hydrogen atom includes isotopes having different numbers of neutrons, that is, light hydrogen (protium), deuterium (deuterium), and tritium (tritium).
L3は、置換基Rを有してもよい炭素数1~20のn+q価の飽和脂肪族炭化水素基、置換基Rを有してもよいn+q価のケイ素含有基、置換基Rを有してもよい環形成炭素数6~18のn+q価の芳香族炭化水素基、又は置換基Rを有してもよい環形成原子数5~18のn+q価の不飽和複素環基である。
R11及びR12は、それぞれ、置換基Rを有してもよい炭素数1~20のアルキル基、置換基Rを有してもよい環形成炭素数3~18のシクロアルキル基、置換基Rを有してもよい炭素数1~20のアルコキシ基、置換基Rを有してもよい環形成炭素数3~20のシクロアルコキシ基、置換基Rを有してもよい環形成炭素数6~18のアリールオキシ基、置換基Rを有してもよいシリル基、フルオロ基、シアノ基、置換基Rを有してもよい環形成炭素数6~18のアリール基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリール基であり、R11が複数存在する場合、それぞれ同一でも異なっていてもよく、R12が複数存在する場合、それぞれ同一でも異なっていてもよい。
L 3 has an n + q valent saturated aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have a substituent R, an n + q valent silicon-containing group which may have a substituent R, and a substituent R. And an n + q valent aromatic hydrocarbon group having 6 to 18 ring carbon atoms, or an n + q valent unsaturated heterocyclic group having 5 to 18 ring atoms which may have a substituent R.
R 11 and R 12 are each an alkyl group having 1 to 20 carbon atoms that may have a substituent R, a cycloalkyl group having 3 to 18 ring carbon atoms that may have a substituent R, and a substituent. An alkoxy group having 1 to 20 carbon atoms which may have R, a cycloalkoxy group having 3 to 20 ring carbon atoms which may have a substituent R, and a ring forming carbon number which may have a substituent R 6-18 aryloxy group, silyl group optionally having substituent R, fluoro group, cyano group, aryl group having 6-18 ring forming carbon atoms optionally having substituent R, or substituent R A heteroaryl group having 5 to 18 ring atoms that may have a ring structure, and when there are a plurality of R 11 s , they may be the same or different, and when there are a plurality of R 12 s , they may be the same or different from each other. May be.
pは、0~3の整数(0、1、2又は3)であり、nが2以上の場合、複数のpはそれぞれ同一でも異なっていてもよい。pは、例えば0である。
qは、0~3の整数(0、1、2又は3)であり、例えば0である。
置換基Rは、炭素数1~20のアルキル基、環形成炭素数3~18のシクロアルキル基、炭素数1~20のアルコキシ基、環形成炭素数3~20のシクロアルコキシ基、環形成炭素数6~18のアリールオキシ基、シリル基、フルオロ基、シアノ基、環形成炭素数6~18のアリール基、又は環形成原子数5~18のヘテロアリール基であり、Rが複数存在する場合、それぞれ同一でも異なっていてもよい。
p is an integer of 0 to 3 (0, 1, 2, or 3). When n is 2 or more, the plurality of p may be the same or different. p is, for example, 0.
q is an integer of 0 to 3 (0, 1, 2, or 3), for example, 0.
The substituent R is an alkyl group having 1 to 20 carbon atoms, a cycloalkyl group having 3 to 18 ring carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms, or a ring forming carbon. An aryloxy group having 6 to 18 atoms, a silyl group, a fluoro group, a cyano group, an aryl group having 6 to 18 ring carbon atoms, or a heteroaryl group having 5 to 18 ring atoms, and when there are a plurality of R These may be the same or different.
Czは、下記式(2-1)、(2-2)、(3-1)、(3-2)、(4-1)、(4-2)、(5-1)、(5-2)、(6-1)、(6-2)、(7-1)、(7-2)、(8-1)、及び(8-2)からなる群より選ばれる一つの基であり、nが2以上の場合、複数のCzはそれぞれ同一でも異なっていてもよい。 Cz is represented by the following formulas (2-1), (2-2), (3-1), (3-2), (4-1), (4-2), (5-1), (5- 2) one group selected from the group consisting of (6-1), (6-2), (7-1), (7-2), (8-1), and (8-2) , N is 2 or more, the plurality of Cz may be the same or different.
式(2-1)、(2-2)、(3-1)、(3-2)、(4-1)、(4-2)、(5-1)、(5-2)、(6-1)、(6-2)、(7-1)、(7-2)、(8-1)、(8-2)中、*は、L1との結合位置を表す。
Yは、酸素(O)原子又は硫黄(S)原子であり、nが2以上の場合、複数のYはそれぞれ同一でも異なっていてもよい。
Formulas (2-1), (2-2), (3-1), (3-2), (4-1), (4-2), (5-1), (5-2), ( In (6-1), (6-2), (7-1), (7-2), (8-1), and (8-2), * represents a bonding position with L 1 .
Y is an oxygen (O) atom or sulfur (S) atom. When n is 2 or more, the plurality of Y may be the same or different.
m1及びm2は、それぞれ独立に、0~4の整数(0、1、2、3又は4)である。m1及びm2は、例えば0又は1であり、(m1+m2)は2以下が好ましい。
m3は、0~3の整数(0、1、2又は3)である。m3は、例えば0である。
m4は、0~2の整数(0、1又は2)である。m4は、例えば0である。
m5は、0~4(0、1、2、3又は4)の整数である。m5は、例えば0である。
m 1 and m 2 are each independently an integer of 0 to 4 (0, 1, 2, 3, or 4). m 1 and m 2 are, for example, 0 or 1, and (m 1 + m 2 ) is preferably 2 or less.
m 3 is an integer of 0 to 3 (0, 1, 2, or 3). m 3 is 0, for example.
m 4 is an integer of 0 to 2 (0, 1 or 2). m 4 is 0, for example.
m 5 is an integer of 0 to 4 (0, 1, 2, 3 or 4). m 5 is, for example, 0.
R0、R1、R2及びR3は、それぞれ独立に、置換基Rを有してもよい炭素数1~20のアルキル基、置換基Rを有してもよい環形成炭素数3~18のシクロアルキル基、置換基Rを有してもよい炭素数1~20のアルコキシ基、置換基Rを有してもよい環形成炭素数3~20のシクロアルコキシ基、置換基Rを有してもよい環形成炭素数6~18のアリールオキシ基、置換基Rを有してもよいシリル基、フルオロ基、シアノ基、置換基Rを有してもよい環形成炭素数6~18のアリール基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリール基であり、R0が複数存在する場合、それぞれ同一でも異なっていてもよく、R1が複数存在する場合、それぞれ同一でも異なっていてもよく、R2が複数存在する場合、それぞれ同一でも異なっていてもよく、R3が複数存在する場合、それぞれ同一でも異なっていてもよい。 R 0 , R 1 , R 2 and R 3 are each independently an alkyl group having 1 to 20 carbon atoms which may have a substituent R, or a ring having 3 to 3 carbon atoms which may have a substituent R. An cycloalkyl group having 18 carbon atoms which may have a substituent R, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms which may have a substituent R, and a substituent R; An aryloxy group having 6 to 18 ring carbon atoms, a silyl group optionally having substituent R, a fluoro group, a cyano group, and 6 to 18 ring carbon atoms optionally having substituent R Or a heteroaryl group having 5 to 18 ring atoms that may have a substituent R, and when a plurality of R 0 are present, they may be the same or different, and a plurality of R 1 are present. If you, or different and each identical, if R 2 there are a plurality, Re may be the same or different, respectively, when the R 3 there are a plurality may each be the same or different.
本発明の有機EL素子用材料は、燐光発光する有機EL素子の発光層や、発光層に隣接する層、例えば、正孔障壁層や電子障壁層の材料等として特に好ましい。 The organic EL device material of the present invention is particularly preferable as a material for a light emitting layer of an organic EL device that emits phosphorescence or a layer adjacent to the light emitting layer, for example, a hole barrier layer or an electron barrier layer.
本発明の有機EL素子用材料は、好ましくは下記式(3)で表される。
本発明の有機EL素子用材料は、式(1)のCzが、カルバゾール骨格、又はカルバゾール骨格を部分構造として有する環構造を有する。式(4)中のCzと、式(4)中の破線で囲って示すDで表される環構造が、共にカルバゾール骨格を部分構造として有することにより、これらの環構造の間に良好な親和性をもたらし、高いガラス転移点及び高いキャリア輸送能を有する良好な膜を形成することができる。
また、本発明の有機EL素子用材料は、上記式(4)中の破線で囲って示すDで表される環構造において、ベンゼン環A、Bに置換基を有さない。
これにより、本材料の分子同士、又は本材料分子とドーパント分子との間で、良好な凝集状態又は分子配向状態が形成され、高いガラス転移点及び高いキャリア輸送能を有する良好な膜を形成することができる。これは分子形状としてアスペクト比が高くなりやすくなるためだと考えられる。
本発明の有機EL素子用材料を用いることにより、得られる有機EL素子が長寿命化する。本発明の有機EL素子用材料は、有機EL素子の発光層のホスト材料、電子障壁層、正孔障壁層の材料として特に好適である。
Moreover, the organic EL device material of the present invention has no substituent on the benzene rings A and B in the ring structure represented by D surrounded by the broken line in the above formula (4).
Thereby, a good aggregation state or molecular orientation state is formed between the molecules of the material or between the material molecule and the dopant molecule, and a good film having a high glass transition point and a high carrier transporting ability is formed. be able to. This is thought to be because the aspect ratio tends to be high as the molecular shape.
By using the organic EL device material of the present invention, the obtained organic EL device has a long life. The material for an organic EL element of the present invention is particularly suitable as a host material, an electron barrier layer, and a hole barrier layer of a light emitting layer of the organic EL element.
式(1)のCzが、式(2-1)で表される基である場合、即ち、Czのカルバゾール骨格が9位でL1と結合している場合は、発光層のホスト材料、又は正孔障壁層としてさらに好適である。
式(1)のCzが、式(2-2)で表される基である場合、即ち、Czのカルバゾール骨格が9位以外でL1と結合している場合は、正孔輸送性が向上するため、電子障壁層の材料としてさらに好適である。
When Cz in Formula (1) is a group represented by Formula (2-1), that is, when the carbazole skeleton of Cz is bonded to L 1 at the 9-position, the host material of the light-emitting layer, or It is further suitable as a hole blocking layer.
When Cz in formula (1) is a group represented by formula (2-2), that is, when the carbazole skeleton of Cz is bonded to L 1 at positions other than 9-position, hole transportability is improved. Therefore, it is more suitable as a material for the electron barrier layer.
以下、上述した式(1)の各基の例について説明する。
炭素数1~20のアルキル基としては、直鎖状もしくは分岐状のアルキル基があり、具体的には、メチル基、エチル基、プロピル基、イソプロピル基、n-ブチル基、イソブチル基、sec-ブチル基、tert-ブチル基、n-ペンチル基、n-ヘキシル基、n-ヘプチル基、n-オクチル基等が挙げられ、好ましくは、メチル基、エチル基、プロピル基、イソプロピル基、n-ブチル基、イソブチル基、sec-ブチル基、tert-ブチル基が挙げられ、好ましくはメチル基、エチル基、プロピル基、イソプロピル基、n-ブチル基、sec-ブチル基、tert-ブチル基である。
炭素数1~20のn+q価の飽和脂肪族炭化水素基としては、上述した基のn+q価の基が挙げられる。
Hereinafter, examples of each group of the above-described formula (1) will be described.
Examples of the alkyl group having 1 to 20 carbon atoms include linear or branched alkyl groups, and specifically include methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec- Examples include butyl group, tert-butyl group, n-pentyl group, n-hexyl group, n-heptyl group, n-octyl group and the like, preferably methyl group, ethyl group, propyl group, isopropyl group, n-butyl Group, isobutyl group, sec-butyl group and tert-butyl group, preferably methyl group, ethyl group, propyl group, isopropyl group, n-butyl group, sec-butyl group and tert-butyl group.
Examples of the n + q-valent saturated aliphatic hydrocarbon group having 1 to 20 carbon atoms include the n + q-valent groups of the above-described groups.
環形成炭素数3~18のシクロアルキル基としては、シクロプロピル基、シクロブチル基、シクロペンチル基、シクロヘキシル基、1-アダマンチル基、2-アダマンチル基、1-ノルボルニル基、2-ノルボルニル基等が挙げられ、好ましくはシクロペンチル基、シクロヘキシル基、1-アダマンチル基、2-アダマンチル基である。
炭素数1~20のアルコキシ基は、-OY1と表され、Y1の例として上記のアルキルの例が挙げられる。アルコキシ基は、例えばメトキシ基、エトキシ基である。アルコキシ基はフッ素原子で置換されていてもよく、この場合、トリフルオロメトキシ基等が好ましい。
Examples of the cycloalkyl group having 3 to 18 ring carbon atoms include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a 1-adamantyl group, a 2-adamantyl group, a 1-norbornyl group, and a 2-norbornyl group. Preferred are a cyclopentyl group, a cyclohexyl group, a 1-adamantyl group, and a 2-adamantyl group.
The alkoxy group having 1 to 20 carbon atoms is represented as —OY 1 and examples of Y 1 include the above alkyl examples. The alkoxy group is, for example, a methoxy group or an ethoxy group. The alkoxy group may be substituted with a fluorine atom, and in this case, a trifluoromethoxy group or the like is preferable.
環形成炭素数3~20のシクロアルコキシ基は、-OY2と表され、Y2の例として上記のシクロアルキル基の例が挙げられる。シクロアルコキシ基は、例えばシクロペンチルオキシ基、シクロヘキシルオキシ基である。 A cycloalkoxy group having 3 to 20 ring carbon atoms is represented by —OY 2, and examples of Y 2 include the above-described cycloalkyl groups. The cycloalkoxy group is, for example, a cyclopentyloxy group or a cyclohexyloxy group.
環形成炭素数6~18のアリール基は、好ましくは環形成炭素数6~12のアリール基である。
1価のアリール基の具体例としては、フェニル基、ビフェニル基、ターフェニル基、トリル基、キシリル基、ナフチル基、アントリル基、フェナントリル基、ナフタセニル基、ピレニル基、クリセニル基、トリフェニレニル基、フルオレニル基、ビフェニルイル基、ターフェニル基、フルオランテニル基等が挙げられ、好ましくはフェニル基、ビフェニル基、ターフェニル基、トリル基、キシリル基、ナフチル基、フェナントリル基、トリフェニレニル基である。
The aryl group having 6 to 18 ring carbon atoms is preferably an aryl group having 6 to 12 ring carbon atoms.
Specific examples of the monovalent aryl group include phenyl group, biphenyl group, terphenyl group, tolyl group, xylyl group, naphthyl group, anthryl group, phenanthryl group, naphthacenyl group, pyrenyl group, chrysenyl group, triphenylenyl group, fluorenyl group. , Biphenylyl group, terphenyl group, fluoranthenyl group and the like, preferably phenyl group, biphenyl group, terphenyl group, tolyl group, xylyl group, naphthyl group, phenanthryl group and triphenylenyl group.
環形成炭素数6~18のn+q価の芳香族炭化水素基としては、上述した基のn+q価の基が挙げられる。
環形成炭素数6~18のアリーレン基としては、上述した基の2価の基が挙げられる。
環形成炭素数6~18のアリールオキシ基は、-OY3と表され、Y3の例として上記のアリール基の例が挙げられる。アリールオキシ基は、例えばフェノキシ基である。
Examples of the n + q valent aromatic hydrocarbon group having 6 to 18 ring carbon atoms include the n + q valent groups of the aforementioned groups.
Examples of the arylene group having 6 to 18 ring carbon atoms include the divalent groups described above.
An aryloxy group having 6 to 18 ring carbon atoms is represented by —OY 3, and examples of Y 3 include the above aryl groups. The aryloxy group is, for example, a phenoxy group.
環形成原子数5~18のヘテロアリール基は、好ましくは環形成原子数5~10のヘテロアリール基である。
1価のヘテロアリール基の具体例としては、ピロリル基、ピラジニル基、ピリジニル基、ピリミジニル基、トリアジニル基、インドリル基、イソインドリル基、イミダゾリル基、フリル基、ベンゾフラニル基、イソベンゾフラニル基、ジベンゾフラニル基、ジベンゾチオフェニル基、アザジベンゾフラニル基、アザジベンゾチオフェニル基、ジアザジベンゾフラニル基、ジアザジベンゾチオフェニル基、キノリル基、イソキノリル基、キノキサリニル基、カルバゾリル基、フェナントリジニル基、アクリジニル基、フェナントロリニル基、フェナジニル基、フェノチアジニル基、フェノキサジニル基、オキサゾリル基、オキサジアゾリル基、フラザニル基、チエニル基、ベンゾチオフェニル基、ジヒドロアクリジニル基、アザカルバゾリル基、ジアザカルバゾリル基、キナゾリニル基等が挙げられ、好ましくは、ピリジニル基、ピリミジニル基、トリアジニル基、ジベンゾフラニル基、ジベンゾチオフェニル基、アザジベンゾフラニル基、アザジベンゾチオフェニル基、ジアザジベンゾフラニル基、ジアザジベンゾチオフェニル基、カルバゾリル基、アザカルバゾリル基、ジアザカルバゾリル基である。
The heteroaryl group having 5 to 18 ring atoms is preferably a heteroaryl group having 5 to 10 ring atoms.
Specific examples of the monovalent heteroaryl group include pyrrolyl group, pyrazinyl group, pyridinyl group, pyrimidinyl group, triazinyl group, indolyl group, isoindolyl group, imidazolyl group, furyl group, benzofuranyl group, isobenzofuranyl group, dibenzofuranyl group. Nyl group, dibenzothiophenyl group, azadibenzofuranyl group, azadibenzothiophenyl group, diazadibenzofuranyl group, diazadibenzothiophenyl group, quinolyl group, isoquinolyl group, quinoxalinyl group, carbazolyl group, phenanthridinyl Group, acridinyl group, phenanthrolinyl group, phenazinyl group, phenothiazinyl group, phenoxazinyl group, oxazolyl group, oxadiazolyl group, furanyl group, thienyl group, benzothiophenyl group, dihydroacridinyl group, azacarbazolyl group And diazacarbazolyl group, quinazolinyl group, etc., preferably pyridinyl group, pyrimidinyl group, triazinyl group, dibenzofuranyl group, dibenzothiophenyl group, azadibenzofuranyl group, azadibenzothiophenyl group, diaza A dibenzofuranyl group, a diazadibenzothiophenyl group, a carbazolyl group, an azacarbazolyl group, and a diazacarbazolyl group;
環形成原子数5~18のn+q価の不飽和複素環基としては、上述した基のn+q価の基が挙げられる。
環形成原子数5~18のヘテロアリーレン基としては、上述した基の2価の基が挙げられる。
Examples of the n + q valent unsaturated heterocyclic group having 5 to 18 ring atoms include the n + q valent group described above.
Examples of the heteroarylene group having 5 to 18 ring atoms include the divalent groups described above.
n+q価のケイ素含有基としては、例えば、シリル基が挙げられる。
Rで置換されたケイ素含有基としては、例えば、炭素数1~20(好ましくは炭素数1~10、より好ましくは炭素数1~6)のアルキルシリル基、環形成炭素数6~18(好ましくは環形成炭素数6~12)のアリールシリル基が挙げられる。
アルキルシリル基の具体例としては、トリメチルシリル基、トリエチルシリル基、t-ブチルジメチルシリル基、ビニルジメチルシリル基、プロピルジメチルシリル基等が挙げられる。
アリールシリル基の具体例としては、トリフェニルシリル基、フェニルジメチルシリル基、t-ブチルジフェニルシリル基、トリトリルシリル基、トリキシリルシリル基、トリナフチルシリル基等が挙げられる。
上記式(1)で表される化合物の具体例を以下に示す。
Examples of the n + q-valent silicon-containing group include a silyl group.
Examples of the silicon-containing group substituted by R include an alkylsilyl group having 1 to 20 carbon atoms (preferably 1 to 10 carbon atoms, more preferably 1 to 6 carbon atoms), and 6 to 18 ring carbon atoms (preferably Is an arylsilyl group having 6 to 12 ring carbon atoms.
Specific examples of the alkylsilyl group include a trimethylsilyl group, a triethylsilyl group, a t-butyldimethylsilyl group, a vinyldimethylsilyl group, and a propyldimethylsilyl group.
Specific examples of the arylsilyl group include a triphenylsilyl group, a phenyldimethylsilyl group, a t-butyldiphenylsilyl group, a tolylsilylsilyl group, a trixylsilyl group, a trinaphthylsilyl group, and the like.
Specific examples of the compound represented by the above formula (1) are shown below.
本発明の化合物は実施例の合成例に記載の方法、又は当業者に既知の方法により合成できる。
続いて、本発明の有機EL素子について説明する。
本発明の有機EL素子は、陽極と陰極の間に、発光層を含む1層以上の有機薄膜層を有する。そして、有機薄膜層の少なくとも1層が、本発明の有機EL素子用材料を含有する。
The compounds of the present invention can be synthesized by the methods described in the synthesis examples of the examples or methods known to those skilled in the art.
Next, the organic EL element of the present invention will be described.
The organic EL device of the present invention has one or more organic thin film layers including a light emitting layer between an anode and a cathode. And at least 1 layer of an organic thin film layer contains the organic EL element material of this invention.
図1は、本発明の有機EL素子の一実施形態の層構成を示す概略図である。
有機EL素子1は、基板10上に、陽極20、正孔輸送帯域30、燐光発光層40、電子輸送帯域50及び陰極60を、この順で積層した構成を有する。正孔輸送帯域30は、正孔輸送層又は正孔注入層等を意味する。同様に、電子輸送帯域50は、電子輸送層又は電子注入層等を意味する。これらは形成しなくともよいが、好ましくは1層以上形成する。この素子において有機薄膜層は、正孔輸送帯域30に設けられる各有機層、燐光発光層40及び電子輸送帯域50に設けられる各有機層である。これら有機薄膜層のうち、少なくとも1層が本発明の有機EL素子用材料を含有する。これにより、有機EL素子の駆動電圧を低くできる。
尚、本発明の有機EL素子用材料を含有する有機薄膜層に対するこの材料の含有量は、好ましくは1~100重量%である。
FIG. 1 is a schematic view showing a layer structure of an embodiment of the organic EL device of the present invention.
The organic EL element 1 has a configuration in which an anode 20, a hole transport zone 30, a phosphorescent light emitting layer 40, an electron transport zone 50, and a cathode 60 are laminated on a substrate 10 in this order. The hole transport zone 30 means a hole transport layer or a hole injection layer. Similarly, the electron transport zone 50 means an electron transport layer, an electron injection layer, or the like. These need not be formed, but preferably one or more layers are formed. In this element, the organic thin film layer is each organic layer provided in the hole transport zone 30, each phosphor layer and the organic layer provided in the electron transport zone 50. Among these organic thin film layers, at least one layer contains the organic EL element material of the present invention. Thereby, the drive voltage of an organic EL element can be lowered.
The content of this material with respect to the organic thin film layer containing the organic EL device material of the present invention is preferably 1 to 100% by weight.
本発明の有機EL素子においては、燐光発光層40が本発明の有機EL素子用材料を含有することが好ましく、特に、発光層のホスト材料として使用することが好ましい。本発明の材料は、三重項エネルギーが十分に大きいため、青色の燐光発光性ドーパント材料を使用しても、燐光発光性ドーパント材料の三重項エネルギーを効率的に発光層内に閉じ込めることができる。尚、青色発光層に限らず、より長波長の光(緑~赤色等)の発光層にも使用できる。 In the organic EL device of the present invention, the phosphorescent light emitting layer 40 preferably contains the material for the organic EL device of the present invention, and particularly preferably used as a host material for the light emitting layer. Since the triplet energy of the material of the present invention is sufficiently large, even when a blue phosphorescent dopant material is used, the triplet energy of the phosphorescent dopant material can be efficiently confined in the light emitting layer. In addition, it can be used not only for the blue light emitting layer but also for a light emitting layer of longer wavelength light (such as green to red).
燐光発光層は、燐光発光性材料(燐光ドーパント)を含有する。燐光ドーパントとしては、金属錯体化合物が挙げられ、好ましくはイリジウム(Ir)、白金(Pt)、オスミウム(Os)、金(Au)、銅(Cu)、レニウム(Re)及びルテニウム(Ru)から選択される金属原子と、配位子とを有する化合物である。配位子は、オルトメタル結合を有すると好ましい。
燐光量子収率が高く、発光素子の外部量子効率をより向上させることができるという点で、燐光ドーパントは、Ir、Os及びPtから選ばれる金属原子を含有する化合物であると好ましく、イリジウム錯体、オスミウム錯体、白金錯体等の金属錯体であるとさらに好ましく、中でもイリジウム錯体及び白金錯体がより好ましく、オルトメタル化イリジウム錯体が最も好ましい。ドーパントは、1種単独でも、2種以上の混合物でもよい。
The phosphorescent light emitting layer contains a phosphorescent material (phosphorescent dopant). Examples of phosphorescent dopants include metal complex compounds, preferably selected from iridium (Ir), platinum (Pt), osmium (Os), gold (Au), copper (Cu), rhenium (Re), and ruthenium (Ru). A compound having a metal atom and a ligand. The ligand preferably has an ortho metal bond.
The phosphorescent dopant is preferably a compound containing a metal atom selected from Ir, Os and Pt in that the phosphorescent quantum yield is high and the external quantum efficiency of the light-emitting element can be further improved, and an iridium complex, It is more preferable that it is a metal complex such as an osmium complex and a platinum complex, among which an iridium complex and a platinum complex are more preferable, and an orthometalated iridium complex is most preferable. The dopant may be a single type or a mixture of two or more types.
燐光発光層における燐光ドーパントの添加濃度は特に限定されるものではないが、好ましくは0.1~30重量%(wt%)、より好ましくは0.1~20重量%(wt%)である The addition concentration of the phosphorescent dopant in the phosphorescent light emitting layer is not particularly limited, but is preferably 0.1 to 30% by weight (wt%), more preferably 0.1 to 20% by weight (wt%).
また、燐光発光層40に隣接する層に本発明の材料を使用することも好ましい。例えば、図1の素子の正孔輸送帯域30と燐光発光層40の間に、本発明の材料を含有する層(陽極側隣接層)を形成した場合、該層は電子障壁層としての機能や励起子阻止層としての機能を有する。
一方、燐光発光層40と電子輸送帯域50の間に本発明の材料を含有する層(陰極側隣接層)を形成した場合、該層は正孔障壁層としての機能や励起子阻止層としての機能を有する。
It is also preferable to use the material of the present invention in a layer adjacent to the phosphorescent light emitting layer 40. For example, when a layer containing the material of the present invention (an anode side adjacent layer) is formed between the hole transport zone 30 and the phosphorescent light emitting layer 40 of the device of FIG. 1, the layer functions as an electron barrier layer. It functions as an exciton blocking layer.
On the other hand, when a layer (cathode side adjacent layer) containing the material of the present invention is formed between the phosphorescent light emitting layer 40 and the electron transport zone 50, the layer functions as a hole blocking layer or as an exciton blocking layer. It has a function.
尚、障壁層(阻止層)とは、キャリアの移動障壁、又は励起子の拡散障壁の機能を有する層である。発光層から正孔輸送帯域へ電子が漏れることを防ぐための有機層を主に電子障壁層と定義し、発光層から電子輸送帯域へ正孔が漏れることを防ぐための有機層を正孔障壁層と定義することがある。また、発光層で生成された三重項励起子が、三重項エネルギーが発光層よりも低い準位を有する周辺層へ拡散することを防止するための有機層を励起子阻止層(トリプレット障壁層)と定義することがある。
また本発明の材料を燐光発光層40に隣接する層に用い,かつさらにその隣接する層に接合する他の有機薄膜層に用いることもできる。
The barrier layer (blocking layer) is a layer having a function of a carrier movement barrier or an exciton diffusion barrier. The organic layer for preventing electrons from leaking from the light-emitting layer to the hole transport zone is mainly defined as an electron barrier layer, and the organic layer for preventing holes from leaking from the light-emitting layer to the electron transport zone is defined as a hole barrier. Sometimes defined as a layer. In addition, an exciton blocking layer (triplet barrier layer) is an organic layer for preventing triplet excitons generated in the light emitting layer from diffusing into a peripheral layer having triplet energy lower than that of the light emitting layer. It may be defined as
Further, the material of the present invention can be used for a layer adjacent to the phosphorescent light emitting layer 40 and further used for another organic thin film layer bonded to the adjacent layer.
さらに、発光層を2層以上形成する場合、発光層間に形成するスペース層としても好適である。
図2は、本発明の有機EL素子の他の実施形態の層構成を示す概略図である。
有機EL素子2は、燐光発光層と蛍光発光層を積層したハイブリッド型の有機EL素子の例である。
有機EL素子2は、燐光発光層40と電子輸送帯域50の間にスペース層42と蛍光発光層44を形成した他は、上記有機EL素子1と同様な構成を有する。燐光発光層40及び蛍光発光層44を積層した構成では、燐光発光層40で形成された励起子を蛍光発光層44に拡散させないため、蛍光発光層44と燐光発光層40の間にスペース層42を設けることがある。本発明の材料は、三重項エネルギーが大きいため、スペース層として機能できる。
Furthermore, when two or more light emitting layers are formed, it is also suitable as a space layer formed between the light emitting layers.
FIG. 2 is a schematic view showing the layer structure of another embodiment of the organic EL device of the present invention.
The organic EL element 2 is an example of a hybrid type organic EL element in which a phosphorescent light emitting layer and a fluorescent light emitting layer are laminated.
The organic EL element 2 has the same configuration as the organic EL element 1 except that a space layer 42 and a fluorescent light emitting layer 44 are formed between the phosphorescent light emitting layer 40 and the electron transport zone 50. In the configuration in which the phosphorescent light emitting layer 40 and the fluorescent light emitting layer 44 are laminated, the excitons formed in the phosphorescent light emitting layer 40 are not diffused into the fluorescent light emitting layer 44, so that a space layer 42 is provided between the fluorescent light emitting layer 44 and the phosphorescent light emitting layer 40. May be provided. Since the material of the present invention has a large triplet energy, it can function as a space layer.
有機EL素子2において、例えば、燐光発光層を黄色発光とし、蛍光発光層を青色発光層とすることにより、白色発光の有機EL素子が得られる。尚、本実施形態では燐光発光層及び蛍光発光層を1層ずつとしているが、これに限らず、それぞれ2層以上形成してもよく、照明や表示装置等、用途に合わせて適宜設定できる。例えば、白色発光素子とカラーフィルタを利用してフルカラー発光装置とする場合、演色性の観点から、赤、緑、青(RGB)、赤、緑、青、黄(RGBY)等、複数の波長領域の発光を含んでいることが好ましい場合がある。 In the organic EL element 2, for example, a white light emitting organic EL element can be obtained by setting the phosphorescent light emitting layer to emit yellow light and the fluorescent light emitting layer to blue light emitting layer. In this embodiment, the phosphorescent light-emitting layer and the fluorescent light-emitting layer are formed one by one. However, the present invention is not limited to this, and two or more layers may be formed, and can be appropriately set according to the application such as lighting and display device. For example, when a full color light emitting device is formed using a white light emitting element and a color filter, a plurality of wavelength regions such as red, green, blue (RGB), red, green, blue, yellow (RGBY) are used from the viewpoint of color rendering. In some cases, it may be preferable to include luminescence.
上述した実施形態の他に、本発明の有機EL素子は、公知の様々な構成を採用できる。また、発光層の発光は、陽極側、陰極側、あるいは両側から取り出すことができる。 In addition to the above-described embodiments, the organic EL element of the present invention can employ various known configurations. Further, light emission of the light emitting layer can be taken out from the anode side, the cathode side, or both sides.
本発明の有機EL素子は、陰極と有機薄膜層との界面領域に電子供与性ドーパント及び有機金属錯体の少なくともいずれかを有することも好ましい。
このような構成によれば、有機EL素子における発光輝度の向上や長寿命化が図られる。
電子供与性ドーパントとしては、アルカリ金属、アルカリ金属化合物、アルカリ土類金属、アルカリ土類金属化合物、希土類金属、及び希土類金属化合物等から選ばれた少なくとも1種類が挙げられる。
有機金属錯体としては、アルカリ金属を含む有機金属錯体、アルカリ土類金属を含む有機金属錯体、及び希土類金属を含む有機金属錯体等から選ばれた少なくとも1種類が挙げられる。
The organic EL device of the present invention preferably has at least one of an electron donating dopant and an organometallic complex in an interface region between the cathode and the organic thin film layer.
According to such a configuration, it is possible to improve the light emission luminance and extend the life of the organic EL element.
Examples of the electron donating dopant include at least one selected from alkali metals, alkali metal compounds, alkaline earth metals, alkaline earth metal compounds, rare earth metals, rare earth metal compounds, and the like.
Examples of the organometallic complex include at least one selected from an organometallic complex containing an alkali metal, an organometallic complex containing an alkaline earth metal, an organometallic complex containing a rare earth metal, and the like.
アルカリ金属としては、リチウム(Li)(仕事関数:2.93eV)、ナトリウム(Na)(仕事関数:2.36eV)、カリウム(K)(仕事関数:2.28eV)、ルビジウム(Rb)(仕事関数:2.16eV)、セシウム(Cs)(仕事関数:1.95eV)等が挙げられ、仕事関数が2.9eV以下のものが特に好ましい。これらのうち好ましくはK、Rb、Cs、さらに好ましくはRb又はCsであり、最も好ましくはCsである。
アルカリ土類金属としては、カルシウム(Ca)(仕事関数:2.9eV)、ストロンチウム(Sr)(仕事関数:2.0eV以上2.5eV以下)、バリウム(Ba)(仕事関数:2.52eV)等が挙げられ、仕事関数が2.9eV以下のものが特に好ましい。
希土類金属としては、スカンジウム(Sc)、イットリウム(Y)、セリウム(Ce)、テルビウム(Tb)、イッテルビウム(Yb)等が挙げられ、仕事関数が2.9eV以下のものが特に好ましい。
以上の金属のうち好ましい金属は、特に還元能力が高く、電子注入域への比較的少量の添加により、有機EL素子における発光輝度の向上や長寿命化が可能である。
Examples of the alkali metal include lithium (Li) (work function: 2.93 eV), sodium (Na) (work function: 2.36 eV), potassium (K) (work function: 2.28 eV), rubidium (Rb) (work Function: 2.16 eV), cesium (Cs) (work function: 1.95 eV) and the like, and those having a work function of 2.9 eV or less are particularly preferable. Of these, K, Rb, and Cs are preferred, Rb and Cs are more preferred, and Cs is most preferred.
Examples of the alkaline earth metal include calcium (Ca) (work function: 2.9 eV), strontium (Sr) (work function: 2.0 eV to 2.5 eV), barium (Ba) (work function: 2.52 eV). A work function of 2.9 eV or less is particularly preferable.
Examples of the rare earth metal include scandium (Sc), yttrium (Y), cerium (Ce), terbium (Tb), ytterbium (Yb) and the like, and those having a work function of 2.9 eV or less are particularly preferable.
Among the above metals, preferred metals are particularly high in reducing ability, and by adding a relatively small amount to the electron injection region, it is possible to improve the light emission luminance and extend the life of the organic EL element.
アルカリ金属化合物としては、酸化リチウム(Li2O)、酸化セシウム(Cs2O)、酸化カリウム(K2O)等のアルカリ酸化物、フッ化リチウム(LiF)、フッ化ナトリウム(NaF)、フッ化セシウム(CsF)、フッ化カリウム(KF)等のアルカリハロゲン化物等が挙げられ、フッ化リチウム(LiF)、酸化リチウム(Li2O)、フッ化ナトリウム(NaF)が好ましい。
アルカリ土類金属化合物としては、酸化バリウム(BaO)、酸化ストロンチウム(SrO)、酸化カルシウム(CaO)及びこれらを混合したストロンチウム酸バリウム(BaxSr1-xO)(0<x<1)、カルシウム酸バリウム(BaxCa1-xO)(0<x<1)等が挙げられ、BaO、SrO、CaOが好ましい。
希土類金属化合物としては、フッ化イッテルビウム(YbF3)、フッ化スカンジウム(ScF3)、酸化スカンジウム(ScO3)、酸化イットリウム(Y2O3)、酸化セリウム(Ce2O3)、フッ化ガドリニウム(GdF3)、フッ化テルビウム(TbF3)等が挙げられ、YbF3、ScF3、TbF3が好ましい。
Examples of the alkali metal compound include lithium oxide (Li 2 O), cesium oxide (Cs 2 O), alkali oxides such as potassium oxide (K 2 O), lithium fluoride (LiF), sodium fluoride (NaF), fluorine. Examples thereof include alkali halides such as cesium fluoride (CsF) and potassium fluoride (KF), and lithium fluoride (LiF), lithium oxide (Li 2 O), and sodium fluoride (NaF) are preferable.
Examples of the alkaline earth metal compound include barium oxide (BaO), strontium oxide (SrO), calcium oxide (CaO), and barium strontium oxide (Ba x Sr 1-x O) (0 <x <1), Examples thereof include barium calcium oxide (Ba x Ca 1-x O) (0 <x <1), and BaO, SrO, and CaO are preferable.
The rare earth metal compound, ytterbium fluoride (YbF 3), scandium fluoride (ScF 3), scandium oxide (ScO 3), yttrium oxide (Y 2 O 3), cerium oxide (Ce 2 O 3), gadolinium fluoride (GdF 3), include such terbium fluoride (TbF 3) is, YbF 3, ScF 3, TbF 3 are preferable.
有機金属錯体としては、上記の通り、それぞれ金属イオンとしてアルカリ金属イオン、アルカリ土類金属イオン、希土類金属イオンの少なくとも1つ含有するものであれば特に限定はない。また、配位子にはキノリノール、ベンゾキノリノール、アクリジノール、フェナントリジノール、ヒドロキシフェニルオキサゾール、ヒドロキシフェニルチアゾール、ヒドロキシジアリールオキサジアゾール、ヒドロキシジアリールチアジアゾール、ヒドロキシフェニルピリジン、ヒドロキシフェニルベンゾイミダゾール、ヒドロキシベンゾトリアゾール、ヒドロキシフルボラン、ビピリジル、フェナントロリン、フタロシアニン、ポルフィリン、シクロペンタジエン、β-ジケトン類、アゾメチン類、及びそれらの誘導体等が好ましいが、これらに限定されるものではない。 The organometallic complex is not particularly limited as long as it contains at least one of an alkali metal ion, an alkaline earth metal ion, and a rare earth metal ion as a metal ion as described above. The ligands include quinolinol, benzoquinolinol, acridinol, phenanthridinol, hydroxyphenyloxazole, hydroxyphenylthiazole, hydroxydiaryloxadiazole, hydroxydiarylthiadiazole, hydroxyphenylpyridine, hydroxyphenylbenzimidazole, hydroxybenzotriazole, Hydroxyfulborane, bipyridyl, phenanthroline, phthalocyanine, porphyrin, cyclopentadiene, β-diketones, azomethines, and derivatives thereof are preferred, but are not limited thereto.
電子供与性ドーパント及び有機金属錯体の添加形態としては、界面領域に層状又は島状に形成することが好ましい。形成方法としては、抵抗加熱蒸着法により電子供与性ドーパント及び有機金属錯体の少なくともいずれかを蒸着しながら、界面領域を形成する発光材料や電子注入材料である有機物を同時に蒸着させ、有機物中に電子供与性ドーパント及び有機金属錯体の少なくともいずれかを分散する方法が好ましい。分散濃度は通常、モル比で有機物:電子供与性ドーパント及び/又は有機金属錯体=100:1~1:100であり、好ましくは5:1~1:5である。 As the addition form of the electron donating dopant and the organometallic complex, it is preferable to form a layer or an island in the interface region. As a forming method, while depositing at least one of an electron donating dopant and an organometallic complex by a resistance heating vapor deposition method, an organic material as a light emitting material or an electron injection material for forming an interface region is simultaneously deposited, and an electron is deposited in the organic material. A method of dispersing at least one of the donor dopant and the organometallic complex is preferable. The dispersion concentration is usually organic substance: electron donating dopant and / or organometallic complex in a molar ratio of 100: 1 to 1: 100, preferably 5: 1 to 1: 5.
電子供与性ドーパント及び有機金属錯体の少なくともいずれかを層状に形成する場合は、界面の有機層である発光材料や電子注入材料を層状に形成した後に、電子供与性ドーパント及び有機金属錯体の少なくともいずれかを単独で抵抗加熱蒸着法により蒸着し、好ましくは層の厚み0.1nm以上15nm以下で形成する。 In the case where at least one of the electron donating dopant and the organometallic complex is formed in a layered form, after forming the light emitting material or the electron injecting material that is the organic layer at the interface in a layered form, at least one of the electron donating dopant and the organometallic complex is formed. These are vapor-deposited by a resistance heating vapor deposition method alone, preferably with a layer thickness of 0.1 nm to 15 nm.
電子供与性ドーパント及び有機金属錯体の少なくともいずれかを島状に形成する場合は、界面の有機層である発光材料や電子注入材料を島状に形成した後に、電子供与性ドーパント及び有機金属錯体の少なくともいずれかを単独で抵抗加熱蒸着法により蒸着し、好ましくは島の厚み0.05nm以上1nm以下で形成する。 In the case where at least one of an electron donating dopant and an organometallic complex is formed in an island shape, a light emitting material or an electron injecting material which is an organic layer at the interface is formed in an island shape, and then the electron donating dopant and the organometallic complex are formed. At least one of them is vapor-deposited by a resistance heating vapor deposition method, preferably with an island thickness of 0.05 nm to 1 nm.
また、本発明の有機EL素子における、主成分と、電子供与性ドーパント及び有機金属錯体の少なくともいずれかの割合としては、モル比で、主成分:電子供与性ドーパント及び/又は有機金属錯体=5:1~1:5であると好ましく、2:1~1:2であるとさらに好ましい。 In the organic EL device of the present invention, the ratio of at least one of the main component and the electron donating dopant and the organometallic complex is, as a molar ratio, the main component: the electron donating dopant and / or the organometallic complex = 5. It is preferably 1 to 1: 5, and more preferably 2: 1 to 1: 2.
本発明の有機EL素子では、上述した本発明の有機EL素子用材料を使用した層以外の構成については、特に限定されず、公知の材料等を使用できる。以下、実施形態1の素子の層について簡単に説明するが、本発明の有機EL素子に適用される材料は以下に限定されない。 In the organic EL element of the present invention, the configuration other than the layer using the organic EL element material of the present invention described above is not particularly limited, and a known material or the like can be used. Hereinafter, although the layer of the element of Embodiment 1 is demonstrated easily, the material applied to the organic EL element of this invention is not limited to the following.
[基板]
基板としてはガラス板、ポリマー板等を用いることができる。
ガラス板としては、特にソーダ石灰ガラス、バリウム・ストロンチウム含有ガラス、鉛ガラス、アルミノケイ酸ガラス、ホウケイ酸ガラス、バリウムホウケイ酸ガラス、石英等が挙げられる。また、ポリマー板としては、ポリカーボネート、アクリル、ポリエチレンテレフタレート、ポリエーテルサルフォン、ポリサルフォン等を挙げることができる。
[substrate]
As the substrate, a glass plate, a polymer plate or the like can be used.
Examples of the glass plate include soda lime glass, barium / strontium-containing glass, lead glass, aluminosilicate glass, borosilicate glass, barium borosilicate glass, and quartz. Examples of the polymer plate include polycarbonate, acrylic, polyethylene terephthalate, polyether sulfone, and polysulfone.
[陽極]
陽極は例えば導電性材料からなり、4eVより大きな仕事関数を有する導電性材料が適している。
上記導電性材料としては、炭素、アルミニウム、バナジウム、鉄、コバルト、ニッケル、タングステン、銀、金、白金、パラジウム等及びそれらの合金、ITO基板、NESA基板に使用される酸化スズ、酸化インジウム等の酸化金属、さらにはポリチオフェンやポリピロール等の有機導電性樹脂が挙げられる。
陽極は、必要があれば2層以上の層構成により形成されていてもよい。
[anode]
The anode is made of, for example, a conductive material, and a conductive material having a work function larger than 4 eV is suitable.
Examples of the conductive material include carbon, aluminum, vanadium, iron, cobalt, nickel, tungsten, silver, gold, platinum, palladium, and their alloys, ITO substrate, tin oxide used for NESA substrate, indium oxide, and the like. Examples thereof include metal oxides and organic conductive resins such as polythiophene and polypyrrole.
The anode may be formed with a layer structure of two or more layers if necessary.
[陰極]
陰極は例えば導電性材料からなり、4eVより小さな仕事関数を有する導電性材料が適している。
上記導電性材料としては、マグネシウム、カルシウム、錫、鉛、チタニウム、イットリウム、リチウム、ルテニウム、マンガン、アルミニウム、フッ化リチウム等及びこれらの合金が挙げられるが、これらに限定されるものではない。
また、上記合金としては、マグネシウム/銀、マグネシウム/インジウム、リチウム/アルミニウム等が代表例として挙げられるが、これらに限定されるものではない。合金の比率は、蒸着源の温度、雰囲気、真空度等により制御され、適切な比率に選択される。
陰極は、必要があれば2層以上の層構成により形成されていてもよく、陰極は上記導電性材料を蒸着やスパッタリング等の方法により薄膜を形成させることにより、作製することができる。
[cathode]
The cathode is made of, for example, a conductive material, and a conductive material having a work function smaller than 4 eV is suitable.
Examples of the conductive material include, but are not limited to, magnesium, calcium, tin, lead, titanium, yttrium, lithium, ruthenium, manganese, aluminum, lithium fluoride, and alloys thereof.
Examples of the alloy include magnesium / silver, magnesium / indium, lithium / aluminum, and the like, but are not limited thereto. The ratio of the alloy is controlled by the temperature of the vapor deposition source, the atmosphere, the degree of vacuum, etc., and is selected to an appropriate ratio.
If necessary, the cathode may be formed with a layer structure of two or more layers, and the cathode can be produced by forming a thin film from the conductive material by a method such as vapor deposition or sputtering.
発光層からの発光を陰極から取り出す場合、陰極の発光に対する透過率は10%より大きくすることが好ましい。
また、陰極としてのシート抵抗は数百Ω/□以下が好ましく、膜厚は通常10nm~1μmであり、好ましくは50~200nmである。
When light emitted from the light emitting layer is taken out from the cathode, the transmittance of the cathode for light emission is preferably greater than 10%.
The sheet resistance as the cathode is preferably several hundred Ω / □ or less, and the film thickness is usually 10 nm to 1 μm, preferably 50 to 200 nm.
[発光層]
本発明の有機EL素子層材料以外の材料で燐光発光層を形成する場合、燐光発光層の材料として公知の材料が使用できる。具体的には、国際公開第2005/079118号(特願2005-517938号明細書)等を参照すればよい。
本発明の有機EL素子は、図2に示す素子のように蛍光発光層を有していてもよい。蛍光発光層としては、公知の材料が使用できる。
[Light emitting layer]
When the phosphorescent light emitting layer is formed of a material other than the organic EL element layer material of the present invention, a known material can be used as the material of the phosphorescent light emitting layer. Specifically, International Publication No. 2005/079118 (Japanese Patent Application No. 2005-517938) may be referred to.
The organic EL device of the present invention may have a fluorescent light emitting layer like the device shown in FIG. A known material can be used for the fluorescent light emitting layer.
発光層は、ダブルホスト(ホスト・コホストともいう)としてもよい。具体的に、発光層において電子輸送性のホストと正孔輸送性のホストを組み合わせることで、発光層内のキャリアバランスを調整してもよい。
また、ダブルドーパントとしてもよい。発光層において、量子収率の高いドーパント材料を2種類以上入れることによって、それぞれのドーパントが発光する。例えば、ホストと赤色ドーパント、緑色のドーパントを共蒸着することによって、黄色の発光層を実現することがある。
発光層は単層でもよく、また、積層構造でもよい。発光層を積層させると、発光層界面に電子と正孔を蓄積させることによって再結合領域を発光層界面に集中させることができる。これによって、量子効率を向上させる。
The light emitting layer may be a double host (also referred to as a host / cohost). Specifically, the carrier balance in the light emitting layer may be adjusted by combining an electron transporting host and a hole transporting host in the light emitting layer.
Moreover, it is good also as a double dopant. In the light emitting layer, each dopant emits light by adding two or more dopant materials having a high quantum yield. For example, a yellow light emitting layer may be realized by co-evaporating a host, a red dopant, and a green dopant.
The light emitting layer may be a single layer or a laminated structure. When the light emitting layer is stacked, the recombination region can be concentrated on the light emitting layer interface by accumulating electrons and holes at the light emitting layer interface. This improves the quantum efficiency.
[正孔注入層及び正孔輸送層]
正孔注入・輸送層は、発光層への正孔注入を助け、発光領域まで輸送する層であって、正孔移動度が大きく、イオン化エネルギーが通常5.6eV以下と小さい層である。
正孔注入・輸送層の材料としては、より低い電界強度で正孔を発光層に輸送する材料が好ましく、さらに正孔の移動度が、例えば104~106V/cmの電界印加時に、少なくとも10-4cm2/V・秒であれば好ましい。
[Hole injection layer and hole transport layer]
The hole injection / transport layer is a layer that assists hole injection into the light emitting layer and transports it to the light emitting region, and has a high hole mobility and a small ionization energy of usually 5.6 eV or less.
As the material for the hole injection / transport layer, a material that transports holes to the light emitting layer with lower electric field strength is preferable. Further, when an electric field is applied with a hole mobility of, for example, 10 4 to 10 6 V / cm, At least 10 −4 cm 2 / V · sec is preferable.
正孔注入・輸送層の材料としては、具体的には、トリアゾール誘導体(米国特許第3,112,197号明細書等参照)、オキサジアゾール誘導体(米国特許第3,189,447号明細書等参照)、イミダゾール誘導体(特公昭37-16096号公報等参照)、ポリアリールアルカン誘導体(米国特許第3,615,402号明細書、同第3,820,989号明細書、同第3,542,544号明細書、特公昭45-555号公報、同51-10983号公報、特開昭51-93224号公報、同55-17105号公報、同56-4148号公報、同55-108667号公報、同55-156953号公報、同 56-36656号公報等参照)、ピラゾリン誘導体及びピラゾロン誘導体(米国特許第3,180,729号明細書、同第4,278,746号明細書、特開昭55-88064号公報、同55-88065号公報、同49-105537号公報、同55-51086号公報、同56-80051号公報、同56-88141号公報、同57-45545号公報、同54-112637号公報、同55-74546号公報等参照)、フェニレンジアミン誘導体(米国特許第3,615,404号明細書、特公昭51-10105号公報、同46-3712号公報、同47-25336号公報、同54-119925号公報等参照)、アリールアミン誘導体(米国特許第3,567,450号明細書、同第3,240,597号明細書、同第3,658,520号明細書、同第4,232,103号明細書、同第4,175,961号明細書、同第4,012,376号明細書、特公昭49-35702号公報、同39-27577号公報、特開昭55-144250号公報、同56-119132号公報、同56-22437号公報、西独特許第1,110,518号明細書等参照)、アミノ置換カルコン誘導体(米国特許第3,526,501号明細書等参照)、オキサゾール誘導体(米国特許第3,257,203号明細書等に開示のもの)、スチリルアントラセン誘導体(特開昭56-46234号公報等参照)、フルオレノン誘導体(特開昭54-110837号公報等参照)、ヒドラゾン誘導体(米国特許第3,717,462号明細書、特開昭54-59143号公報、同55-52063号公報、同55-52064号公報、同55-46760号公報、同57-11350号公報、同57-148749号公報、特開平2-311591号公報等参照)、スチルベン誘導体(特開昭61-210363号公報、同61-228451号公報、同61-14642号公報、同61-72255号公報、同62-47646号公報、同62-36674号公報、同62-10652号公報、同62-30255号公報、同60-93455号公報、同60-94462号公報、同60-174749号公報、同60-175052号公報等参照)、シラザン誘導体(米国特許第4,950,950号明細書)、ポリシラン系(特開平2-204996号公報)、アニリン系共重合体(特開平2-282263号公報)等を挙げることができる。
また、p型Si、p型SiC等の無機化合物も正孔注入材料として使用することができる。
Specific examples of the material for the hole injection / transport layer include triazole derivatives (see US Pat. No. 3,112,197) and oxadiazole derivatives (US Pat. No. 3,189,447). ), Imidazole derivatives (see Japanese Patent Publication No. 37-16096, etc.), polyarylalkane derivatives (US Pat. Nos. 3,615,402, 3,820,989, 3, No. 542,544, JP-B-45-555, JP-A-51-10983, JP-A-51-93224, JP-A-55-17105, JP-A-56-4148, JP-A-55-108667. Gazette, 55-156953 gazette, 56-36656 gazette, etc.), pyrazoline derivatives and pyrazolone derivatives (US Pat. No. 3,180,729, 4,278,746, JP-A-55-88064, 55-88065, 49-105537, 55-51086, 56-80051, 56 -88141, 57-45545, 54-11126, 55-74546, etc.), phenylenediamine derivatives (US Pat. No. 3,615,404, JP-B 51-10105) No. 46-3712, No. 47-25336, No. 54-1119925, etc.), arylamine derivatives (US Pat. No. 3,567,450, No. 3,240,597) No. 3,658,520, No. 4,232,103, No. 4,175,961, No. 4,012, No. 76, JP-B-49-35702, JP-A-39-27577, JP-A-55-144250, JP-A-56-119132, JP-A-56-22437, West German Patent No. 1,110, 518), amino-substituted chalcone derivatives (see US Pat. No. 3,526,501 etc.), oxazole derivatives (disclosed in US Pat. No. 3,257,203 etc.), styryl Anthracene derivatives (see JP-A-56-46234, etc.), fluorenone derivatives (see JP-A-54-110837, etc.), hydrazone derivatives (US Pat. No. 3,717,462, JP-A-54- 59143, 55-52063, 55-52064, 55-46760, 57-11350, 57-148749, JP-A-2-311591, etc.), stilbene derivatives (JP-A 61-210363, 61-228451, 61-14642, 61-72255, 62-47646, 62-36674, 62-10652, 62-30255, 60-93455, 60-94462, 60-174749, 60-175052, etc.), silazane derivatives (US Pat. No. 4,950,950), polysilanes (JP-A-2-204996), aniline copolymers (JP-A-2-282263) And the like.
In addition, inorganic compounds such as p-type Si and p-type SiC can also be used as the hole injection material.
正孔注入・輸送層の材料には架橋型材料を用いることができ、架橋型の正孔注入輸送層としては、例えば、Chem.Mater.2008,20,413-422、Chem.Mater.2011,23(3),658-681、国際公開第2008/108430号、国際公開第2009/102027号、国際公開第2009/123269号、国際公開第2010/016555号、国際公開第2010/018813号等の架橋材を、熱、光等により不溶化した層が挙げられる。 As the material of the hole injection / transport layer, a cross-linkable material can be used. As the cross-linkable hole injection / transport layer, for example, Chem. Mater. 2008, 20, 413-422, Chem. Mater. 2011, 23 (3), 658-681, International Publication No. 2008/108430, International Publication No. 2009/102027, International Publication No. 2009/123269, International Publication No. 2010/016555, International Publication No. 2010/018813. And a layer obtained by insolubilizing a cross-linking material such as heat or light.
[電子注入層及び電子輸送層]
電子注入・輸送層は、発光層への電子の注入を助け、発光領域まで輸送する層であって、電子移動度が大きい層である。
有機EL素子は発光した光が電極(例えば陰極)により反射するため、直接陽極から取り出される発光と、電極による反射を経由して取り出される発光とが干渉することが知られている。この干渉効果を効率的に利用するため、電子注入・輸送層は数nm~数μmの膜厚で適宜選ばれるが、特に膜厚が厚いとき、電圧上昇を避けるために、104~106V/cmの電界印加時に電子移動度が少なくとも10-5cm2/Vs以上であることが好ましい。
[Electron injection layer and electron transport layer]
The electron injection / transport layer is a layer that assists the injection of electrons into the light emitting layer and transports it to the light emitting region, and has a high electron mobility.
In the organic EL element, since emitted light is reflected by an electrode (for example, a cathode), it is known that light emitted directly from the anode interferes with light emitted via reflection by the electrode. In order to efficiently use this interference effect, the electron injecting / transporting layer is appropriately selected with a film thickness of several nm to several μm. However, particularly when the film thickness is large, in order to avoid a voltage increase, 10 4 to 10 6. The electron mobility is preferably at least 10 −5 cm 2 / Vs or more when an electric field of V / cm is applied.
電子注入・輸送層に用いる電子輸送性材料としては、分子内にヘテロ原子を1個以上含有する芳香族ヘテロ環化合物が好ましく用いられ、特に含窒素環誘導体が好ましい。また、含窒素環誘導体としては、含窒素6員環もしくは5員環骨格を有する芳香族環、又は含窒素6員環もしくは5員環骨格を有する縮合芳香族環化合物が好ましく、例えば、ピリジン環、ピリミジン環、トリアジン環、ベンズイミダゾール環、フェナントロリン環、キナゾリン環等を骨格に含む化合物が挙げられる。 As the electron transporting material used for the electron injection / transport layer, an aromatic heterocyclic compound containing one or more heteroatoms in the molecule is preferably used, and a nitrogen-containing ring derivative is particularly preferable. The nitrogen-containing ring derivative is preferably an aromatic ring having a nitrogen-containing 6-membered ring or 5-membered ring skeleton, or a condensed aromatic ring compound having a nitrogen-containing 6-membered ring or 5-membered ring skeleton, such as a pyridine ring. , Pyrimidine ring, triazine ring, benzimidazole ring, phenanthroline ring, quinazoline ring and the like.
その他、ドナー性材料のドーピング(n)、アクセプター材料のドーピング(p)により、半導体性を備えた有機層を形成してもよい。Nドーピングの代表例は、電子輸送性材料にLiやCs等の金属をドーピングさせるものであり、Pドーピングの代表例は、正孔輸送性材料にF4TCNQ(2,3,5,6-テトラフルオロ-7,7,8,8-テトラシアノキノジメタン)等のアクセプター材をドープするものである(例えば、特許第3695714号明細書参照)。 In addition, an organic layer having semiconductivity may be formed by doping (n) with a donor material and doping (p) with an acceptor material. A typical example of N doping is to dope an electron transporting material with a metal such as Li or Cs, and a typical example of P doping is F4TCNQ (2,3,5,6-tetrafluoro) to a hole transporting material. -7,7,8,8-tetracyanoquinodimethane) or the like (see, for example, Japanese Patent No. 3695714).
本発明の有機EL素子の各層の形成は、真空蒸着、スパッタリング、プラズマ、イオンプレーティング等の乾式成膜法やスピンコーティング、ディッピング、フローコーティング等の湿式成膜法等の公知の方法を適用することができる。
各層の膜厚は特に限定されるものではないが、適切な膜厚に設定する必要がある。膜厚が厚すぎると、一定の光出力を得るために大きな印加電圧が必要になり効率が悪くなる。膜厚が薄すぎるとピンホール等が発生して、電界を印加しても充分な発光輝度が得られない。通常の膜厚は5nm~10μmの範囲が適しているが、10nm~0.2μmの範囲がさらに好ましい。
For the formation of each layer of the organic EL device of the present invention, a known method such as a dry film forming method such as vacuum deposition, sputtering, plasma, or ion plating, or a wet film forming method such as spin coating, dipping, or flow coating is applied. be able to.
The thickness of each layer is not particularly limited, but must be set to an appropriate thickness. If the film thickness is too thick, a large applied voltage is required to obtain a constant light output, resulting in poor efficiency. If the film thickness is too thin, pinholes and the like are generated, and sufficient light emission luminance cannot be obtained even when an electric field is applied. The normal film thickness is suitably in the range of 5 nm to 10 μm, but more preferably in the range of 10 nm to 0.2 μm.
次に、合成例及び実施例を用いて本発明をさらに詳細に説明する。ただし、本発明は以下の合成例、実施例に限定されない。 Next, the present invention will be described in further detail using synthesis examples and examples. However, the present invention is not limited to the following synthesis examples and examples.
尚、化合物の評価方法は下記の通りである。
(1)三重項エネルギー(ET)
市販の装置F-4500(日立社製)を用いて測定した。三重項エネルギー(ET)の換算式は以下のとおりである。
ET(eV)=1239.85/λph
式中、「λph」(単位:nm)は、縦軸に燐光強度、横軸に波長をとって、燐光スペクトルを表したときに、燐光スペクトルの短波長側の立ち上がりに対して接線を引き、その接線と横軸の交点の波長値を意味する。
In addition, the evaluation method of a compound is as follows.
(1) Triplet energy (E T )
The measurement was performed using a commercially available apparatus F-4500 (manufactured by Hitachi). The conversion formula of triplet energy (E T ) is as follows.
E T (eV) = 1239.85 / λ ph
In the formula, “λ ph ” (unit: nm) draws a tangent line to the rising edge of the phosphorescence spectrum on the short wavelength side when the phosphorescence spectrum is represented with the phosphorescence intensity on the vertical axis and the wavelength on the horizontal axis. , The wavelength value of the intersection of the tangent and the horizontal axis.
各化合物をEPA溶媒(ジエチルエーテル:イソペンタン:エタノール=5:5:5(容積比)、各溶媒は分光用グレード)に溶解し(試料10μmol/リットル)、燐光測定用試料とした。石英セルへ入れた燐光測定用試料を77(K)に冷却し、励起光を燐光測定用試料に照射し、波長を変えながら燐光強度を測定した。燐光スペクトルは、縦軸を燐光強度、横軸を波長とした。
この燐光スペクトルの短波長側の立ち上がりに対して接線を引き、その接線と横軸との交点の波長値λph(nm)を求めた。
Each compound was dissolved in an EPA solvent (diethyl ether: isopentane: ethanol = 5: 5: 5 (volume ratio), each solvent was a spectroscopic grade) (sample 10 μmol / liter) to prepare a sample for phosphorescence measurement. The phosphorescence measurement sample placed in the quartz cell was cooled to 77 (K), and the phosphorescence measurement sample was irradiated with excitation light, and the phosphorescence intensity was measured while changing the wavelength. In the phosphorescence spectrum, the vertical axis represents phosphorescence intensity and the horizontal axis represents wavelength.
A tangent line was drawn with respect to the rising edge of the phosphorescence spectrum on the short wavelength side, and the wavelength value λ ph (nm) at the intersection of the tangent line and the horizontal axis was obtained.
燐光スペクトルの短波長側の立ち上がりに対する接線は以下のように引く。燐光スペクトルの短波長側から、スペクトルの極大値のうち、最も短波長側の極大値までスペクトル曲線上を移動する際に、長波長側に向けて曲線上の各点における接線を考える。この接線は、曲線が立ち上がるにつれ(つまり縦軸が増加するにつれ)傾きが増加する。この傾きの値が極大値をとる点において引いた接線を、当該燐光スペクトルの短波長側の立ち上がりに対する接線とする。
なお、スペクトルの最大ピーク強度の10%以下のピーク強度をもつ極大点は、上述の最も短波長側の極大値には含めず、最も短波長側の極大値に最も近い、傾きの値が極大値をとる点において引いた接線を当該燐光スペクトルの短波長側の立ち上がりに対する接線とする。
The tangent to the rising edge on the short wavelength side of the phosphorescence spectrum is drawn as follows. When moving on the spectrum curve from the short wavelength side of the phosphorescence spectrum to the maximum value on the shortest wavelength side among the maximum values of the spectrum, tangents at each point on the curve are considered toward the long wavelength side. The slope of this tangent increases as the curve rises (that is, as the vertical axis increases). The tangent drawn at the point where the slope value takes the maximum value is taken as the tangent to the rising edge of the phosphorescence spectrum on the short wavelength side.
In addition, the maximum point having a peak intensity of 10% or less of the maximum peak intensity of the spectrum is not included in the above-mentioned maximum value on the shortest wavelength side, and has the maximum slope value closest to the maximum value on the shortest wavelength side. The tangent drawn at the point where the value is taken is taken as the tangent to the rising edge of the phosphorescence spectrum on the short wavelength side.
(2)ガラス転移点(℃)
ガラス転移点は、3mg程度の試料を用い、Perkin Elmer社製DSC8500を用い、以下の(i)~(vi)までの2サイクルの昇降温プロセスを行い、(vi)の昇温時のDSC曲線のベースラインが段状に変化している変曲点の立ち上がり温度によって定義する。
(2) Glass transition point (° C)
The glass transition point is about 3 mg of sample, DSC8500 manufactured by Perkin Elmer is used, and the following two steps of (i) to (vi) are performed for the temperature rising and falling processes, and the DSC curve at the time of temperature increase in (vi) This is defined by the rising temperature of the inflection point where the baseline of the curve changes stepwise.
(i)30℃で1分間保持する。
(ii)30℃から該試料の熱分解温度未満の一定温度まで昇温速度10℃/minで加熱する。
(iii)該一定温度にて3分間保持する。
(iv)該一定温度から0℃まで200℃/minで冷却する。
(v)0℃で10分間保持する。
(vi)0℃から200℃まで昇温速度10℃/minで加熱する。
(I) Hold at 30 ° C. for 1 minute.
(Ii) Heat from 30 ° C. to a constant temperature below the thermal decomposition temperature of the sample at a heating rate of 10 ° C./min.
(Iii) Hold at the constant temperature for 3 minutes.
(Iv) Cool from the constant temperature to 0 ° C. at 200 ° C./min.
(V) Hold at 0 ° C. for 10 minutes.
(Vi) Heat from 0 ° C. to 200 ° C. at a heating rate of 10 ° C./min.
また、有機EL素子の評価方法は下記の通りである。
(1)外部量子効率(%)
23℃、乾燥窒素ガス雰囲気下で、輝度1000cd/m2時の外部量子効率を輝度計(ミノルタ社製分光輝度放射計CS-1000)を用いて測定した。
Moreover, the evaluation method of an organic EL element is as follows.
(1) External quantum efficiency (%)
The external quantum efficiency at a luminance of 1000 cd / m 2 under a dry nitrogen gas atmosphere at 23 ° C. was measured using a luminance meter (Spectral Luminance Radiometer CS-1000 manufactured by Minolta).
(2)半減寿命(時間)
初期輝度1000cd/m2で連続通電試験(直流)を行い、初期輝度が半減するまでの時間を測定した。
(2) Half life (hours)
A continuous energization test (DC) was performed at an initial luminance of 1000 cd / m 2 and the time until the initial luminance was reduced by half was measured.
(3)電圧(V)
23℃、乾燥窒素ガス雰囲気下で、KEITHLY 236 SOURCE MEASURE UNITを用いて、電気配線された素子に電圧を印加して発光させ、素子以外の配線抵抗にかかる電圧を差し引いて素子印加電圧を測定した。
電圧の印加・測定と同時に輝度計(ミノルタ社製分光輝度放射計CS-1000)を用いて輝度測定も行い、これらの測定結果から素子輝度が1000cd/m2時の電圧を読み取った。
(3) Voltage (V)
Using a KEITLY 236 SOURCE MEASURE UNIT under a dry nitrogen gas atmosphere at 23 ° C., voltage was applied to the electrically wired element to emit light, and the voltage applied to the wiring resistance other than the element was subtracted to measure the element applied voltage. .
At the same time as the voltage application / measurement, the luminance was also measured using a luminance meter (Spectral Luminance Radiometer CS-1000 manufactured by Minolta Co., Ltd.), and the voltage when the element luminance was 1000 cd / m 2 was read from these measurement results.
合成例1[化合物(1)の合成]
(1)中間体(1-1)の合成
(1) Synthesis of intermediate (1-1)
三口フラスコに4-ブロモ-2-クロロ-1-フルオロベンゼン(75.4g,360mmol)、カルバゾール(72.23g,432mmol)、K3PO4(152.83g,720mmol)、CuI(13.71g,72mmol)、trans-1,2-シクロヘキサンジアミン(DACyHx)(26.0ml,216mmol)、1,4-ジオキサン(360ml)を入れ、窒素雰囲気下で24時間還流させた。反応終了後、室温まで冷却しセライトを用いてろ過した。ろ液を濃縮後、シリカゲルショートカラムを通し、濃縮した。得られた粗体をシリカゲルカラムクロマトグラフィー(展開溶媒 ヘキサン:トルエン=1:1)で精製し、中間体(1-1)を得た。化合物の同定はFD-MS(電界脱離質量分析法)及び1H-NMR(プロトン核磁気共鳴法)にて行った。収量は73.9g、収率は69%であった。 In a three-neck flask, 4-bromo-2-chloro-1-fluorobenzene (75.4 g, 360 mmol), carbazole (72.23 g, 432 mmol), K 3 PO 4 (152.83 g, 720 mmol), CuI (13.71 g, 72 mmol), trans-1,2-cyclohexanediamine (DACyHx) (26.0 ml, 216 mmol) and 1,4-dioxane (360 ml) were added and refluxed for 24 hours under a nitrogen atmosphere. After completion of the reaction, the reaction mixture was cooled to room temperature and filtered using celite. The filtrate was concentrated and then concentrated through a silica gel short column. The resulting crude product was purified by silica gel column chromatography (developing solvent hexane: toluene = 1: 1) to obtain an intermediate (1-1). The compound was identified by FD-MS (field desorption mass spectrometry) and 1 H-NMR (proton nuclear magnetic resonance method). The yield was 73.9 g, and the yield was 69%.
(2)中間体(1-2)の合成
三口フラスコに中間体(1-1)(70.98g,240mmol)、4-ヒドロキシカルバゾール(48.37g,264mmol)、K2CO3(66.34g,480mmol)、N-メチルピロリドン(NMP)(120ml)を入れ、窒素雰囲気下160℃で9時間撹拌した。反応終了後、室温まで冷却し、次いでトルエン(1L)を加え、セライトを用いてろ過した。ろ液を分液ロートに移して、水で数回洗浄した。有機層を無水硫酸マグネシウムで乾燥、ろ過、濃縮し、得られた粗体をシリカゲルカラムクロマトグラフィー(展開溶媒 ヘキサン:ジクロロメタン=1:1)で精製し、その後ヘキサンから再結晶して中間体(1-2)を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は47.5g、収率は43%であった。 In a three-necked flask, intermediate (1-1) (70.98 g, 240 mmol), 4-hydroxycarbazole (48.37 g, 264 mmol), K 2 CO 3 (66.34 g, 480 mmol), N-methylpyrrolidone (NMP) ( 120 ml) and stirred at 160 ° C. for 9 hours under a nitrogen atmosphere. After completion of the reaction, the reaction mixture was cooled to room temperature, and then toluene (1 L) was added, followed by filtration using celite. The filtrate was transferred to a separatory funnel and washed several times with water. The organic layer was dried over anhydrous magnesium sulfate, filtered and concentrated, and the resulting crude product was purified by silica gel column chromatography (developing solvent hexane: dichloromethane = 1: 1), then recrystallized from hexane to give an intermediate (1 -2) was obtained. The compound was identified by FD-MS and 1 H-NMR. The yield was 47.5 g, and the yield was 43%.
(3)中間体(1-3)の合成
ディーン・スターク・トラップ(Dean-Stark trap)を取り付けた三口フラスコに中間体(1-2)(47.3g,103mmol)、K2CO3(28.47g,206mmol)、Pd(OAc)2(2.31g,10.3mmol)、トリシクロヘキシルホスフィン(P(Cy)3)の20wt%トルエン溶液(29ml)、N,N-ジメチルアセトアミド(DMAc)(206ml)、トルエン(103ml)を入れ、窒素雰囲気下でディーン・スターク・トラップを用いて脱水しながら8時間還流した。反応終了後、室温まで冷却し、次いでトルエン(1L)を加え、セライトを用いてろ過した。ろ液を分液ロートに移して、水で数回洗浄した。有機層を無水硫酸マグネシウムで乾燥、ろ過した後、シリカゲルショートカラムを通して原点不純物成分を除去した。濃縮後、トルエンから2回再結晶して中間体(1-3)を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は25.95g、収率は60%であった。 A three-necked flask equipped with a Dean-Stark trap was charged with intermediate (1-2) (47.3 g, 103 mmol), K 2 CO 3 (28.47 g, 206 mmol), Pd (OAc) 2 ( 2.31 g, 10.3 mmol), 20 wt% toluene solution (29 ml) of tricyclohexylphosphine (P (Cy) 3 ), N, N-dimethylacetamide (DMAc) (206 ml), toluene (103 ml), nitrogen atmosphere Under reflux using a Dean Stark trap, the mixture was refluxed for 8 hours. After completion of the reaction, the reaction mixture was cooled to room temperature, and then toluene (1 L) was added, followed by filtration using celite. The filtrate was transferred to a separatory funnel and washed several times with water. The organic layer was dried over anhydrous magnesium sulfate and filtered, and then the origin impurity component was removed through a silica gel short column. After concentration, recrystallization from toluene twice gave intermediate (1-3). The compound was identified by FD-MS and 1 H-NMR. The yield was 25.95 g, and the yield was 60%.
(4)化合物(1)の合成
三口フラスコに中間体(1-3)(3.38g,8mmol)、ヨードベンゼン(1.80g,8.8mmol)、tert-ブトキシナトリウム(tert-BuONa)(1.53g,16mmol)、トリス(ジベンジリデンアセトン)ジパラジウム(0)(Pd2(dba)3)(147mg,0.16mmol)、トリ-tert-ブチルホスフィン(P(t-Bu)3)(0.64mmol)、トルエン(40ml)を入れ、窒素雰囲気下で8時間還流させた。反応終了後、室温まで冷却しセライトを用いてろ過した。ろ液を濃縮後、シリカゲルショートカラムを通し、濃縮した。得られた粗体をシリカゲルカラムクロマトグラフィー(展開溶媒 ヘキサン:トルエン=6:4)で精製し、化合物(1)を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は3.79g、収率は95%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.26-7.34(m,2H),7.36-7.72(m,14H),7.95(t,2H),8.14(d,1H),8.19(d,2H),8.59(d,1H)
FD-MS測定データ 理論値:498 実測値:498
A three-necked flask was charged with intermediate (1-3) (3.38 g, 8 mmol), iodobenzene (1.80 g, 8.8 mmol), tert-butoxy sodium (tert-BuONa) (1.53 g, 16 mmol), Benzylideneacetone) dipalladium (0) (Pd 2 (dba) 3 ) (147 mg, 0.16 mmol), tri-tert-butylphosphine (P (t-Bu) 3 ) (0.64 mmol), toluene (40 ml) And refluxed for 8 hours under a nitrogen atmosphere. After completion of the reaction, the reaction mixture was cooled to room temperature and filtered using celite. The filtrate was concentrated and then concentrated through a silica gel short column. The resulting crude product was purified by silica gel column chromatography (developing solvent hexane: toluene = 6: 4) to obtain compound (1). The compound was identified by FD-MS and 1 H-NMR. The yield was 3.79 g, and the yield was 95%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.26-7.34 (m, 2H), 7.36-7.72 (m, 14H), 7.95 (t, 2H), 8.14 (d, 1H), 8.19 (d, 2H), 8.59 (d, 1H)
FD-MS measurement data Theoretical value: 498 Actual value: 498
合成例2[化合物(36)の合成]
ヨードベンゼンの代わりに9-(3-ブロモフェニル)カルバゾールを用いたほかは合成例1(4)と同様に、化合物(36)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は5.26g、収率は99%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.24-7.34(m,4H),7.36-7.67(m,13H),7.73-7.82(m,2H),7.86-8.00(m,4H),8.10-8.22(m,5H),8.60(d,1H)
FD-MS測定データ 理論値:663 実測値:663
Compound (36) was synthesized in the same manner as in Synthesis Example 1 (4) except that 9- (3-bromophenyl) carbazole was used in place of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 5.26 g, and the yield was 99%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.24-7.34 (m, 4H), 7.36-7.67 (m, 13H), 7.73-7.82 (m , 2H), 7.86-8.00 (m, 4H), 8.10-8.22 (m, 5H), 8.60 (d, 1H)
FD-MS measurement data Theoretical value: 663 Actual value: 663
合成例3[化合物(39)の合成]
ヨードベンゼンの代わりに2-ブロモジベンゾフランを用いたほかは合成例1(4)と同様に、化合物(39)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は4.61g、収率は98%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.25-7.34(m,2H),7.36-7.73(m,13H),7.83(d,1H),7.91-8.03(m,3H),8.12-8.23(m,4H),8.62(d,1H)
FD-MS測定データ 理論値:588 実測値:588
Compound (39) was synthesized in the same manner as in Synthesis Example 1 (4) except that 2-bromodibenzofuran was used in place of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 4.61 g and the yield was 98%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.25-7.34 (m, 2H), 7.36-7.73 (m, 13H), 7.83 (d, 1H), 7.91-8.03 (m, 3H), 8.12-8.23 (m, 4H), 8.62 (d, 1H)
FD-MS measurement data Theoretical value: 588 Actual value: 588
合成例4[化合物(45)の合成]
ヨードベンゼンの代わりに3-ブロモ-9-フェニルカルバゾールを用いたほかは合成例1(4)と同様に、化合物(45)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は5.10g、収率は96%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.25-7.34(m,3H),7.36-7.72(m,18H),7.88-8.00(m,2H),8.10-8.15(m,2H),8.18(d,2H),8.36(d,1H),8.62(d,1H)
FD-MS測定データ 理論値:663 実測値:663
Compound (45) was synthesized in the same manner as in Synthesis Example 1 (4) except that 3-bromo-9-phenylcarbazole was used instead of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 5.10 g, and the yield was 96%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.25-7.34 (m, 3H), 7.36-7.72 (m, 18H), 7.88-8.00 (m , 2H), 8.10-8.15 (m, 2H), 8.18 (d, 2H), 8.36 (d, 1H), 8.62 (d, 1H)
FD-MS measurement data Theoretical value: 663 Actual value: 663
合成例5[化合物(86)の合成]
(1)中間体(5-1)の合成
(1) Synthesis of intermediate (5-1)
三口フラスコに4-ブロモ-2-クロロ-1-フルオロベンゼン(144.9g,692mmol)、4-ヒドロキシカルバゾール(126.7g,692mmol)、K2CO3(191.3g,1.38mol)、NMP(138ml)を入れ、窒素雰囲気下160℃で36時間撹拌した。反応終了後、室温まで冷却し、次いでジクロロメタン(1L)を加え、セライトを用いてろ過した。ろ液を分液ロートに移動して、ブラインで数回洗浄した。有機層を無水硫酸マグネシウムで乾燥、ろ過、シリカゲルショートカラムを通し、濃縮した。これをトルエンから再結晶して中間体(5-1)を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は116g、収率は45%であった。 A three-necked flask was charged with 4-bromo-2-chloro-1-fluorobenzene (144.9 g, 692 mmol), 4-hydroxycarbazole (126.7 g, 692 mmol), K 2 CO 3 (191.3 g, 1.38 mol), NMP. (138 ml) was added and stirred at 160 ° C. for 36 hours under a nitrogen atmosphere. After completion of the reaction, the mixture was cooled to room temperature, then dichloromethane (1 L) was added, and the mixture was filtered through celite. The filtrate was transferred to a separatory funnel and washed several times with brine. The organic layer was dried over anhydrous magnesium sulfate, filtered, passed through a silica gel short column and concentrated. This was recrystallized from toluene to obtain an intermediate (5-1). The compound was identified by FD-MS and 1 H-NMR. The yield was 116 g, and the yield was 45%.
(2)中間体(5-2)の合成
三口フラスコに中間体(5-1)(37.26g,100mmol)、3-(9-カルバゾリル)フェニルボロン酸(110g,31.58mmol)、Na2CO3の2M水溶液(100ml)、テトラキス(トリフェニルホスフィン)パラジウム(0)(Pd(PPh3)4)(2.31g,2mmol)、1,2-ジメトキシエタン(DME)(200ml)を入れ、窒素雰囲気下で10時間還流した。反応終了後、室温まで冷却し、トルエン200mlを加え、セライトを用いてろ過した。ろ液を分液ロートに移動して、水で2回洗浄した。有機層を無水硫酸マグネシウムで乾燥、ろ過し、シリカゲルショートカラムを通し、濃縮した。これをトルエン:ヘキサン混合溶媒から再結晶して中間体(5-2)を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は39.8g、収率は74%であった。 A three-necked flask was charged with intermediate (5-1) (37.26 g, 100 mmol), 3- (9-carbazolyl) phenylboronic acid (110 g, 31.58 mmol), 2M aqueous solution of Na 2 CO 3 (100 ml), tetrakis (tri Phenylphosphine) palladium (0) (Pd (PPh 3 ) 4 ) (2.31 g, 2 mmol) and 1,2-dimethoxyethane (DME) (200 ml) were added, and the mixture was refluxed for 10 hours under a nitrogen atmosphere. After completion of the reaction, the mixture was cooled to room temperature, 200 ml of toluene was added, and the mixture was filtered using celite. The filtrate was transferred to a separatory funnel and washed twice with water. The organic layer was dried over anhydrous magnesium sulfate, filtered, passed through a silica gel short column, and concentrated. This was recrystallized from a toluene: hexane mixed solvent to obtain an intermediate (5-2). The compound was identified by FD-MS and 1 H-NMR. The yield was 39.8 g and the yield was 74%.
(3)中間体(5-3)の合成
中間体(1-2)の代わりに中間体(5-2)を用いたほかは合成例1(3)と同様に、中間体(5-3)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は21.71g、収率は59%であった。 Intermediate (5-3) was synthesized in the same manner as Synthesis Example 1 (3), except that intermediate (5-2) was used instead of intermediate (1-2). The compound was identified by FD-MS and 1 H-NMR. The yield was 21.71 g, and the yield was 59%.
(4)化合物(86)の合成
中間体(1-3)の代わりに中間体(5-3)を用いたほかは合成例1(4)と同様に、化合物(86)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は5.51g、収率は96%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.29(t,2H),7.34-7.68(m,14H),7.69-7.76(m,2H),7.77-7.86(m,2H),7.92-7.97(m,2H),8.17(d,2H),8.23(d,1H),8.52(d,1H)
FD-MS測定データ 理論値:574 実測値:574
Compound (86) was synthesized in the same manner as in Synthesis Example 1 (4) except that Intermediate (5-3) was used instead of Intermediate (1-3). The compound was identified by FD-MS and 1 H-NMR. The yield was 5.51 g and the yield was 96%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.29 (t, 2H), 7.34-7.68 (m, 14H), 7.69-7.76 (m, 2H), 7.77-7.86 (m, 2H), 7.92-7.97 (m, 2H), 8.17 (d, 2H), 8.23 (d, 1H), 8.52 (d, 1H)
FD-MS measurement data Theoretical value: 574 Actual value: 574
合成例6[化合物(121)の合成]
中間体(5-2)の原料として用いた3-(9-カルバゾリル)フェニルボロン酸の代わりに2-(9-カルバゾリル)ジベンゾフラン-8-ボロン酸を用いたほかは合成例5[化合物(86)の合成]と同様に、化合物(121)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は4.08g、収率は85%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.27-7.34(m,2H),7.37-7.57(m,9H),7.59-7.69(m,5H),7.73-7.78(m,2H),7.79-7.83(m,2H),7.90(dd,1H),8.00(d,1H),8.15-8.32(m,5H),8.53(d,1H)
FD-MS測定データ 理論値:664 実測値:664
Synthetic Example 5 [compound (86) except that 2- (9-carbazolyl) dibenzofuran-8-boronic acid was used in place of 3- (9-carbazolyl) phenylboronic acid used as a starting material for intermediate (5-2). Compound (121) was synthesized in the same manner as in the above. The compound was identified by FD-MS and 1 H-NMR. The yield was 4.08 g and the yield was 85%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.27-7.34 (m, 2H), 7.37-7.57 (m, 9H), 7.59-7.69 (m 5H), 7.73-7.78 (m, 2H), 7.79-7.83 (m, 2H), 7.90 (dd, 1H), 8.00 (d, 1H), 8. 15-8.32 (m, 5H), 8.53 (d, 1H)
FD-MS measurement data Theoretical value: 664 Actual value: 664
合成例7[化合物(139)の合成]
(1)中間体(6-1)の合成
(1) Synthesis of intermediate (6-1)
原料として3-(9-カルバゾリル)フェニルボロン酸の代わりに9-フェニルカルバゾール-3-ボロン酸を用いたほかは合成例5(2)と同様に、中間体(6-1)を合成した。
(2)中間体(6-2)の合成
(2) Synthesis of intermediate (6-2)
中間体(1-2)の代わりに中間体(6-1)を用いたほかは合成例1(3)中間体(1-3)と同様に、中間体(6-2)を合成した。
(3)化合物(139)の合成
(3) Synthesis of compound (139)
中間体(1-3)の代わりに中間体(6-2)を用いたほかは合成例1(4)と同様に、化合物(139)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は4.10g、収率は76%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.28-7.37(m,1H),7.40-7.58(m,9H),7.59-7.72(m,8H),7.78-7.88(m,3H),8.07(d,1H),8.25(d,1H),8.33(t,1H),8.50(t,1H),8.58(d,1H)
FD-MS測定データ 理論値:574 実測値:574
Compound (139) was synthesized in the same manner as in Synthesis Example 1 (4), except that Intermediate (6-2) was used instead of Intermediate (1-3). The compound was identified by FD-MS and 1 H-NMR. The yield was 4.10 g, and the yield was 76%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.28-7.37 (m, 1H), 7.40-7.58 (m, 9H), 7.59-7.72 (m , 8H), 7.78-7.88 (m, 3H), 8.07 (d, 1H), 8.25 (d, 1H), 8.33 (t, 1H), 8.50 (t, 1H), 8.58 (d, 1H)
FD-MS measurement data Theoretical value: 574 Actual value: 574
合成例8[化合物(48)の合成]
ヨードベンゼンの代わりに2-ブロモ-8-(9-カルバゾリル)ジベンゾフランを用いたほかは合成例1(4)と同様に、化合物(48)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は1.36g、収率は65%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.24-7.34(m,4H),7.36-7.55(m,12H),7.60(dd,1H),7.72(dd,1H),7.77(dd,1H),7.87-7.98(m,4H),8.10-8.23(m,7H),8.60(d,1H)
FD-MS測定データ 理論値:753 実測値:753
Compound (48) was synthesized in the same manner as in Synthesis Example 1 (4) except that 2-bromo-8- (9-carbazolyl) dibenzofuran was used instead of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 1.36 g, and the yield was 65%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.24-7.34 (m, 4H), 7.36-7.55 (m, 12H), 7.60 (dd, 1H), 7.72 (dd, 1H), 7.77 (dd, 1H), 7.87-7.98 (m, 4H), 8.10-8.23 (m, 7H), 8.60 (d, 1H)
FD-MS measurement data Theoretical value: 753 Actual value: 753
合成例9[化合物(76)の合成]
三口フラスコに中間体(1-3)(2.11g,5mmol)、3-ブロモ-5-(9-カルバゾリル)ピリジン(1.78g,5.5mmol)、K3PO4(2.12g,10mmol)、CuI(95mg,0.5mmol)、trans-1,2-シクロヘキサンジアミン(DACyHx)(0.18ml,1.5mmol)、1,4-ジオキサン(5ml)を入れ、窒素雰囲気下で24時間還流させた。反応終了後、室温まで冷却しセライトを用いてろ過した。ろ液を濃縮後、シリカゲルショートカラムを通し、濃縮した。得られた粗体を酢酸エチルから再結晶して目的物を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は1.72g、収率は52%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.26-7.66(m,17H),7.95-8.05(m,2H),8.13-8.22(m,5H),8.29(t,1H),8.63(d,1H),9.07(t,2H)
FD-MS測定データ 理論値:664 実測値:664
In a three-necked flask, intermediate (1-3) (2.11 g, 5 mmol), 3-bromo-5- (9-carbazolyl) pyridine (1.78 g, 5.5 mmol), K 3 PO 4 (2.12 g, 10 mmol) ), CuI (95 mg, 0.5 mmol), trans-1,2-cyclohexanediamine (DACyHx) (0.18 ml, 1.5 mmol), 1,4-dioxane (5 ml), and refluxed for 24 hours under a nitrogen atmosphere I let you. After completion of the reaction, the reaction mixture was cooled to room temperature and filtered using celite. The filtrate was concentrated and then concentrated through a silica gel short column. The obtained crude product was recrystallized from ethyl acetate to obtain the desired product. The compound was identified by FD-MS and 1 H-NMR. The yield was 1.72 g and the yield was 52%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.26-7.66 (m, 17H), 7.95-8.05 (m, 2H), 8.13-8.22 (m , 5H), 8.29 (t, 1H), 8.63 (d, 1H), 9.07 (t, 2H)
FD-MS measurement data Theoretical value: 664 Actual value: 664
合成例10[化合物(41)の合成]
ヨードベンゼンの代わりに2-(3-ブロモフェニル)ジベンゾフランを用いたほかは合成例1(4)と同様に、化合物(41)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は0.65g、収率は35%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.25-7.67(m,16H),7.73-7.82(m,2H),7.83-7.90(m,1H),7.92-8.04(m,4H),8.13-8.21(m,3H),8.27(d,1H),8.62(d,1H)
FD-MS測定データ 理論値:664 実測値:664
Compound (41) was synthesized in the same manner as in Synthesis Example 1 (4) except that 2- (3-bromophenyl) dibenzofuran was used instead of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 0.65 g, and the yield was 35%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.25-7.67 (m, 16H), 7.73-7.82 (m, 2H), 7.83-7.90 (m , 1H), 7.92-8.04 (m, 4H), 8.13-8.21 (m, 3H), 8.27 (d, 1H), 8.62 (d, 1H)
FD-MS measurement data Theoretical value: 664 Actual value: 664
合成例11[化合物(49)の合成]
ヨードベンゼンの代わりに2-(2-ブロモジベンゾフラン-8-イル)ジベンゾフランを用いたほかは合成例1(4)と同様に、化合物(49)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は1.82g、収率は70%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.25-7.68(m,15H),7.70-7.80(m,3H),7.84-7.91(m,2H),7.93-8.05(m,3H),8.13-8.21(m,3H),8.23-8.32(m,3H),8.62(d,1H)
FD-MS測定データ 理論値:754 実測値:754
Compound (49) was synthesized in the same manner as in Synthesis Example 1 (4) except that 2- (2-bromodibenzofuran-8-yl) dibenzofuran was used instead of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 1.82 g, and the yield was 70%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.25-7.68 (m, 15H), 7.70-7.80 (m, 3H), 7.84-7.91 (m , 2H), 7.93-8.05 (m, 3H), 8.13-8.21 (m, 3H), 8.23-8.32 (m, 3H), 8.62 (d, 1H) )
FD-MS measurement data Theoretical value: 754 Actual value: 754
合成例12[化合物(87)の合成]
中間体(1-3)の代わりに中間体(5-3)、ヨードベンゼンの代わりに2-ブロモジベンゾフランを用いたほかは合成例1(4)と同様に、化合物(87)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は2.50g、収率は83%であった。
1H-NMR(400MHz,CDCl3):δ=7.28-7.57(13H,m),7.62-7.72(4H,m),7.78-7.82(3H,m),7.92-7.94(3H,m),8.14-8.20(4H,m),8.58-8.60(1H,m)
FD-MS測定データ 理論値: 実測値:
Compound (87) was synthesized in the same manner as in Synthesis Example 1 (4) except that Intermediate (5-3) was used instead of Intermediate (1-3) and 2-bromodibenzofuran was used instead of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 2.50 g, and the yield was 83%.
1 H-NMR (400 MHz, CDCl 3 ): δ = 7.28-7.57 (13H, m), 7.62-7.72 (4H, m), 7.78-7.82 (3H, m ), 7.92-7.94 (3H, m), 8.14-8.20 (4H, m), 8.58-8.60 (1H, m)
FD-MS measurement data Theoretical value: Measured value:
合成例13[化合物(141)の合成]
ヨードベンゼンの代わりに2-ブロモジベンゾフランを用いたほかは合成例1(4)と同様に、化合物(141)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は2.22g、収率は79%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.28-7.36(m,1H),7.37-7.58(m,10H),7.60-7.73(m,6H),7.77-7.88(m,4H),8.00(d,1H),8.07(d,1H),8.19-8.27(m,2H),8.33(d,1H),8.50(d,1H),8.59(d,1H)
FD-MS測定データ 理論値:664 実測値:664
Compound (141) was synthesized in the same manner as in Synthesis Example 1 (4) except that 2-bromodibenzofuran was used instead of iodobenzene. The compound was identified by FD-MS and 1 H-NMR. The yield was 2.22 g and the yield was 79%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.28-7.36 (m, 1H), 7.37-7.58 (m, 10H), 7.60-7.73 (m 6H), 7.77-7.88 (m, 4H), 8.00 (d, 1H), 8.07 (d, 1H), 8.19-8.27 (m, 2H), 8. 33 (d, 1H), 8.50 (d, 1H), 8.59 (d, 1H)
FD-MS measurement data Theoretical value: 664 Actual value: 664
合成例14[化合物(163)の合成]
(1)中間体(7-1)の合成
(1) Synthesis of intermediate (7-1)
三口フラスコに中間体(5-1)(136.0g,365mmol)、ヨードベンゼン(148.9g,730mmol)、K3PO4(116.2g,547.5mmol)、CuI(3.48g,18.25mmol)、trans-1,2-シクロヘキサンジアミン(6.6ml,54.8mmol)、1,4-ジオキサン(365ml)を入れ、窒素雰囲気下で24時間還流させた。反応終了後、室温まで冷却しセライトを用いてろ過した。ろ液を濃縮後、シリカゲルショートカラムを通し、濃縮した。得られた粗体をシリカゲルカラムクロマトグラフィー(展開溶媒 ヘキサン:トルエン=9:1)で精製して中間体(7-1)を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は127.7g、収率は78%であった。 In a three-necked flask, intermediate (5-1) (136.0 g, 365 mmol), iodobenzene (148.9 g, 730 mmol), K 3 PO 4 (116.2 g, 547.5 mmol), CuI (3.48 g, 18. 25 mmol), trans-1,2-cyclohexanediamine (6.6 ml, 54.8 mmol) and 1,4-dioxane (365 ml) were added, and the mixture was refluxed for 24 hours under a nitrogen atmosphere. After completion of the reaction, the reaction mixture was cooled to room temperature and filtered using celite. The filtrate was concentrated and then concentrated through a silica gel short column. The resulting crude product was purified by silica gel column chromatography (developing solvent hexane: toluene = 9: 1) to obtain an intermediate (7-1). The compound was identified by FD-MS and 1 H-NMR. The yield was 127.7 g, and the yield was 78%.
(2)中間体(7-2)の合成
三口フラスコに中間体(7-1)(4.49g,10mmol)、3-(9-フェニルカルバゾール-3-イル)カルバゾール(4.49g,11mmol)、K3PO4(4.25g,20mmol)、CuI(0.95g,5mmol)、trans-1,2-シクロヘキサンジアミン(1.8ml,15mmol)、1,4-ジオキサン(20ml)を入れ、窒素雰囲気下で24時間還流させた。反応終了後、室温まで冷却しセライトを用いてろ過した。ろ液を濃縮後、シリカゲルショートカラムを通し、濃縮した。得られた粗体をシリカゲルカラムクロマトグラフィー(展開溶媒 ヘキサン:トルエン=7:3)で精製して中間体(7-2)を得た。化合物の同定はFD-MS及び1H-NMRにて行った。収量は7.5g、収率は97%であった。 Intermediate (7-1) (4.49 g, 10 mmol), 3- (9-phenylcarbazol-3-yl) carbazole (4.49 g, 11 mmol), K 3 PO 4 (4.25 g, 20 mmol) was added to a three-necked flask. , CuI (0.95 g, 5 mmol), trans-1,2-cyclohexanediamine (1.8 ml, 15 mmol) and 1,4-dioxane (20 ml) were added and refluxed for 24 hours under a nitrogen atmosphere. After completion of the reaction, the reaction mixture was cooled to room temperature and filtered using celite. The filtrate was concentrated and then concentrated through a silica gel short column. The obtained crude product was purified by silica gel column chromatography (developing solvent hexane: toluene = 7: 3) to obtain an intermediate (7-2). The compound was identified by FD-MS and 1 H-NMR. The yield was 7.5 g, and the yield was 97%.
(3)化合物(163)の合成
中間体(1-2)の代わりに中間体(7-2)を用いたほかは合成例1(3)と同様に、化合物(163)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は1.05g、収率は45%であった。
1H-NMR(400MHz,CDCl3):δ=7.31-7.55(m,13H),7.61-7.81(m,10H),7.80(dt,2H),7.94(dd,2H),8.16(d,1H),8.26(dd,2H),8.49(dd,2H),8.64(dt,1H)
FD-MS測定データ 理論値:739 実測値:739
Compound (163) was synthesized in the same manner as in Synthesis Example 1 (3) except that Intermediate (7-2) was used instead of Intermediate (1-2). The compound was identified by FD-MS and 1 H-NMR. The yield was 1.05 g and the yield was 45%.
1 H-NMR (400 MHz, CDCl 3 ): δ = 7.31-7.55 (m, 13H), 7.61-7.81 (m, 10H), 7.80 (dt, 2H), 7. 94 (dd, 2H), 8.16 (d, 1H), 8.26 (dd, 2H), 8.49 (dd, 2H), 8.64 (dt, 1H)
FD-MS measurement data Theoretical value: 739 Actual value: 739
合成例15[化合物(501)の合成]
(1)中間体(8-1)の合成
(1) Synthesis of intermediate (8-1)
3-(9-フェニルカルバゾール-3-イル)カルバゾールの代わりに5H-ベンゾフロ[3,2-c]カルバゾールを用いたほかは合成例14(2)と同様に、中間体(8-1)を合成した。 Intermediate (8-1) was prepared in the same manner as in Synthesis Example 14 (2) except that 5H-benzofuro [3,2-c] carbazole was used instead of 3- (9-phenylcarbazol-3-yl) carbazole. Synthesized.
(2)化合物(501)の合成
中間体(1-2)の代わりに中間体(8-1)を用いたほかは合成例1(3)と同様に、化合物(501)を合成した。化合物の同定はFD-MS及び1H-NMRにて行った。収量は2.16g、収率は62%であった。
1H-NMR(400MHz,CD2Cl2):δ=7.35-7.57(m,11H),7.58-7.70(m,5H),7.77(d,1H),7.78-8.02(m,4H),8.18(d,1H),8.53-8.62(m,2H)
FD-MS測定データ 理論値:588 実測値:588
Compound (501) was synthesized in the same manner as in Synthesis Example 1 (3) except that Intermediate (8-1) was used instead of Intermediate (1-2). The compound was identified by FD-MS and 1 H-NMR. The yield was 2.16 g, and the yield was 62%.
1 H-NMR (400 MHz, CD 2 Cl 2 ): δ = 7.35-7.57 (m, 11H), 7.58-7.70 (m, 5H), 7.77 (d, 1H), 7.78-8.02 (m, 4H), 8.18 (d, 1H), 8.53-8.62 (m, 2H)
FD-MS measurement data Theoretical value: 588 Actual value: 588
実施例1
膜厚130nmのITO電極ライン付きガラス基板(ジオマテック社製)を、イソプロピルアルコール中で5分間、超音波洗浄した後、UVオゾン洗浄を30分間行なった。
洗浄後のITO電極ライン付きガラス基板を真空蒸着装置の基板ホルダーに装着し、まずITO電極ラインが形成されている側の面上に、ITO電極ラインを覆うようにして化合物(HI1)を厚さ20nmで、次いで化合物(HT1)を厚さ60nmで抵抗加熱蒸着し、順次薄膜を成膜した。成膜レートは1Å/sとした。これらの薄膜は、それぞれ正孔注入層及び正孔輸送層として機能する。
Example 1
A glass substrate with a 130 nm-thick ITO electrode line (manufactured by Geomatec) was ultrasonically cleaned in isopropyl alcohol for 5 minutes, and then UV ozone cleaning was performed for 30 minutes.
The glass substrate with the ITO electrode line after the cleaning is mounted on the substrate holder of the vacuum deposition apparatus, and the compound (HI1) is first thickened so as to cover the ITO electrode line on the surface on which the ITO electrode line is formed. The compound (HT1) was deposited by resistance heating at a thickness of 60 nm at a thickness of 20 nm, and thin films were sequentially formed. The film formation rate was 1 Å / s. These thin films function as a hole injection layer and a hole transport layer, respectively.
次に、正孔輸送層上に、化合物(1)と化合物(BD1)を同時に抵抗加熱蒸着して膜厚50nmの薄膜を成膜した。このとき、化合物(BD1)を、化合物(A1)と化合物(BD1)の総質量に対し質量比で20%になるように蒸着した。成膜レートはそれぞれ1.2Å/s、0.3Å/sとした。この薄膜は燐光発光層として機能し、この薄膜において化合物(1)はホスト、化合物(BD1)は発光ドーパントとして機能する。尚、化合物(BD1)の三重項エネルギー(eV)とガラス転移点(℃)を表1に示す。
次に、この燐光発光層上に、化合物(B1)を抵抗加熱蒸着して膜厚10nmの薄膜を成膜した(正孔障壁層)。成膜レートは1.2Å/sとした。
Next, on the hole transport layer, the compound (1) and the compound (BD1) were simultaneously deposited by resistance heating to form a thin film having a thickness of 50 nm. At this time, the compound (BD1) was deposited so as to have a mass ratio of 20% with respect to the total mass of the compound (A1) and the compound (BD1). The film formation rates were 1.2 Å / s and 0.3 Å / s, respectively. This thin film functions as a phosphorescent light emitting layer, in which the compound (1) functions as a host and the compound (BD1) functions as a light emitting dopant. Table 1 shows triplet energy (eV) and glass transition point (° C.) of the compound (BD1).
Next, a compound (B1) was deposited by resistance heating vapor deposition on this phosphorescent light emitting layer to form a thin film having a thickness of 10 nm (hole blocking layer). The film formation rate was 1.2 liter / s.
次に、この正孔障壁層上に、化合物(ET1)を抵抗加熱蒸着して膜厚10nmの薄膜を成膜した。成膜レートは1Å/sとした。この膜は電子注入層として機能する。
次に、この電子注入層上に膜厚1.0nmのLiFを成膜レート0.1Å/sで蒸着した。
次に、このLiF膜上に金属アルミニウムを成膜レート8.0Å/sにて蒸着し、膜厚80nmの金属陰極を形成して有機EL素子を得た。
また、上記の方法により電圧及び半減寿命を求めた。結果を表2に示す。
Next, a thin film having a thickness of 10 nm was formed on the hole barrier layer by resistance heating evaporation of the compound (ET1). The film formation rate was 1 Å / s. This film functions as an electron injection layer.
Next, LiF having a film thickness of 1.0 nm was deposited on the electron injection layer at a film formation rate of 0.1 Å / s.
Next, metallic aluminum was vapor-deposited on the LiF film at a deposition rate of 8.0 Å / s to form a metal cathode with a film thickness of 80 nm to obtain an organic EL element.
Moreover, the voltage and half-life were calculated | required by said method. The results are shown in Table 2.
実施例2~14及び比較例1~2
化合物(1)の代わりに表1に記載の化合物を燐光発光層のホストとして用いた以外は実施例1と同様にして有機EL素子を作製し、評価した。使用した化合物の三重項エネルギー及びガラス転移点を表1に、結果を表2に示す。尚、「半減寿命(相対%)」とは、比較例1の素子の半減寿命を100%とした場合の相対割合である。
Examples 2 to 14 and Comparative Examples 1 to 2
An organic EL device was prepared and evaluated in the same manner as in Example 1 except that the compound shown in Table 1 was used as the host of the phosphorescent light emitting layer instead of the compound (1). Table 1 shows the triplet energy and glass transition point of the compound used, and Table 2 shows the results. The “half life (relative%)” is a relative ratio when the half life of the element of Comparative Example 1 is 100%.
表2より、本発明の化合物を燐光発光層のホストとして用いた場合、長寿命であり、かつ、比較例の化合物よりも低電圧で駆動する有機EL素子を提供できることがわかる。 From Table 2, it can be seen that when the compound of the present invention is used as a host of the phosphorescent light emitting layer, an organic EL device having a long life and being driven at a lower voltage than the compound of the comparative example can be provided.
実施例15
膜厚130nmのITO電極ライン付きガラス基板(ジオマテック社製)を、イソプロピルアルコール中で5分間、超音波洗浄した後、UVオゾン洗浄を30分間行なった。
洗浄後のITO電極ライン付きガラス基板を真空蒸着装置の基板ホルダーに装着し、まずITO電極ラインが形成されている側の面上に、ITO電極ラインを覆うようにして化合物(HI1)を厚さ20nmで、次いで化合物(HT1)を厚さ50nmで抵抗加熱蒸着し、順次薄膜を成膜した。成膜レートは1Å/sとした。これらの薄膜は、それぞれ正孔注入層及び正孔輸送層として機能する。
次に、正孔輸送層上に、化合物(39)を抵抗加熱蒸着して膜厚10nmの薄膜(電子障壁層)を成膜した。成膜レートは1Å/sとした。
Example 15
A glass substrate with a 130 nm-thick ITO electrode line (manufactured by Geomatec) was ultrasonically cleaned in isopropyl alcohol for 5 minutes, and then UV ozone cleaning was performed for 30 minutes.
The glass substrate with the ITO electrode line after the cleaning is mounted on the substrate holder of the vacuum deposition apparatus, and the compound (HI1) is first thickened so as to cover the ITO electrode line on the surface on which the ITO electrode line is formed. At 20 nm, the compound (HT1) was then vapor deposited by resistance heating at a thickness of 50 nm, and thin films were sequentially formed. The film formation rate was 1 Å / s. These thin films function as a hole injection layer and a hole transport layer, respectively.
Next, the compound (39) was deposited by resistance heating vapor deposition on the hole transport layer to form a thin film (electronic barrier layer) having a thickness of 10 nm. The film formation rate was 1 Å / s.
次に、この電子障壁層上に、化合物(B1)と化合物(BD1)を同時に抵抗加熱蒸着して膜厚50nmの薄膜を成膜した。このとき、化合物(BD1)を、化合物(B1)と化合物(BD1)の総質量に対し質量比で20%になるように蒸着した。成膜レートはそれぞれ1.2Å/s、0.3Å/sとした。この薄膜は燐光発光層として機能し、この薄膜において化合物(B1)はホスト、化合物(BD1)は発光ドーパントとして機能する。
次に、この燐光発光層上に、化合物(B1)を抵抗加熱蒸着して膜厚10nmの薄膜を成膜した(正孔障壁層)。成膜レートは1.2Å/sとした。
Next, on this electron barrier layer, the compound (B1) and the compound (BD1) were simultaneously deposited by resistance heating to form a thin film having a thickness of 50 nm. At this time, the compound (BD1) was deposited so as to have a mass ratio of 20% with respect to the total mass of the compound (B1) and the compound (BD1). The film formation rates were 1.2 Å / s and 0.3 Å / s, respectively. This thin film functions as a phosphorescent light emitting layer, and in this thin film, the compound (B1) functions as a host and the compound (BD1) functions as a light emitting dopant.
Next, a compound (B1) was deposited by resistance heating vapor deposition on this phosphorescent light emitting layer to form a thin film having a thickness of 10 nm (hole blocking layer). The film formation rate was 1.2 liter / s.
次に、この正孔障壁層上に、化合物(ET1)を抵抗加熱蒸着して膜厚10nmの薄膜を成膜した。成膜レートは1Å/sとした。この膜は電子注入層として機能する。
次に、この電子注入層上に膜厚1.0nmのLiFを成膜レート0.1Å/sで蒸着した。
次に、このLiF膜上に金属アルミニウムを成膜レート8.0Å/sにて蒸着し、膜厚80nmの金属陰極を形成して有機EL素子を得た。
また、上記の方法により電圧、外部量子効率、及び半減寿命を求めた。結果を表3に示す。
Next, a thin film having a thickness of 10 nm was formed on the hole barrier layer by resistance heating evaporation of the compound (ET1). The film formation rate was 1 Å / s. This film functions as an electron injection layer.
Next, LiF having a film thickness of 1.0 nm was deposited on the electron injection layer at a film formation rate of 0.1 Å / s.
Next, metallic aluminum was vapor-deposited on the LiF film at a deposition rate of 8.0 Å / s to form a metal cathode with a film thickness of 80 nm to obtain an organic EL element.
Further, the voltage, external quantum efficiency, and half life were determined by the above methods. The results are shown in Table 3.
実施例16~18及び比較例3~4
化合物(39)の代わりに表3に記載の化合物を電子障壁層として用いた以外は実施例15と同様にして有機EL素子を作製し、評価した。結果を表3に示す。
Examples 16 to 18 and Comparative Examples 3 to 4
An organic EL device was prepared and evaluated in the same manner as in Example 15 except that the compound shown in Table 3 was used as the electron barrier layer instead of the compound (39). The results are shown in Table 3.
表3より、本発明の化合物を電子障壁層として用いた場合、比較例の化合物よりも長寿命であり、低電圧かつ高効率で駆動する有機EL素子を提供できることがわかる。 From Table 3, it can be seen that when the compound of the present invention is used as an electron barrier layer, it is possible to provide an organic EL device which has a longer life than the compound of the comparative example and can be driven at a low voltage and high efficiency.
実施例19
燐光発光層のホストとして化合物(1)の代わりに化合物(B1)を用い、正孔障壁層として化合物(B1)の代わりに化合物(1)を用いた以外は実施例1と同様にして有機EL素子を作製し、評価した。結果を表4に示す。
実施例20~27及び比較例5
化合物(1)の代わりに表4に記載の化合物を正孔障壁層として用いた以外は実施例11と同様にして有機EL素子を作製し、評価した。結果を表4及び表5に示す。
Example 19
Organic EL in the same manner as in Example 1 except that compound (B1) was used instead of compound (1) as the host of the phosphorescent layer, and compound (1) was used instead of compound (B1) as the hole barrier layer. A device was fabricated and evaluated. The results are shown in Table 4.
Examples 20 to 27 and Comparative Example 5
An organic EL device was prepared and evaluated in the same manner as in Example 11 except that the compounds listed in Table 4 were used as the hole blocking layer instead of the compound (1). The results are shown in Tables 4 and 5.
表4より、本発明の化合物を正孔障壁層として用いた場合、比較例の化合物よりも長寿命な有機EL素子を提供できることがわかる。 Table 4 shows that when the compound of the present invention is used as a hole blocking layer, an organic EL device having a longer life than the compound of the comparative example can be provided.
表5より、本発明の化合物を正孔障壁層として用いた場合、比較例の化合物よりも低電圧かつ高効率な有機EL素子を提供できることがわかる。 Table 5 shows that when the compound of the present invention is used as a hole blocking layer, an organic EL device having a lower voltage and higher efficiency than the compound of the comparative example can be provided.
本発明の化合物は、有機EL素子用材料として使用できる。本発明の有機EL素子用材料を利用すると、寿命が長く、且つ、低電圧駆動の消費電力が低い有機EL素子が得られる。本発明の有機EL素子は、各種電子機器のディスプレイ、光源等として極めて有用である。 The compound of the present invention can be used as a material for an organic EL device. When the organic EL element material of the present invention is used, an organic EL element having a long life and low power consumption for low voltage driving can be obtained. The organic EL element of the present invention is extremely useful as a display, a light source and the like for various electronic devices.
上記に本発明の実施形態及び/又は実施例を幾つか詳細に説明したが、当業者は、本発明の新規な教示及び効果から実質的に離れることなく、これら例示である実施形態及び/又は実施例に多くの変更を加えることが容易である。従って、これらの多くの変更は本発明の範囲に含まれる。
この明細書に記載の文献及び本願のパリ優先の基礎となる日本出願明細書の内容を全てここに援用する。
Although several embodiments and / or examples of the present invention have been described in detail above, those skilled in the art will appreciate that these exemplary embodiments and / or embodiments are substantially without departing from the novel teachings and advantages of the present invention. It is easy to make many changes to the embodiment. Accordingly, many of these modifications are within the scope of the present invention.
The contents of the documents described in this specification and the specification of the Japanese application that is the basis of Paris priority of the present application are all incorporated herein.
1、2 有機EL素子
10 基板
20 陽極
30 正孔輸送帯域
40 燐光発光層
42 スペース層
44 蛍光発光層
50 電子輸送帯域
60 陰極
DESCRIPTION OF SYMBOLS 1, 2 Organic EL element 10 Substrate 20 Anode 30 Hole transport zone 40 Phosphorescence layer 42 Space layer 44 Fluorescence layer 50 Electron transport zone 60 Cathode
Claims (10)
Xは、酸素(O)原子又は硫黄(S)原子であり、nが2以上の場合、複数のXはそれぞれ同一でも異なっていてもよい。
L1及びL2は、それぞれ、単結合、置換基Rを有してもよい環形成炭素数6~18のアリーレン基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリーレン基であり、nが2以上の場合、複数のL1及び複数のL2はそれぞれ同一でも異なっていてもよい。
L3は、置換基Rを有してもよい炭素数1~20のn+q価の飽和脂肪族炭化水素基、置換基Rを有してもよいn+q価のケイ素含有基、置換基Rを有してもよい環形成炭素数6~18のn+q価の芳香族炭化水素基、又は置換基Rを有してもよい環形成原子数5~18のn+q価の不飽和複素環基である。
R11及びR12は、それぞれ、置換基Rを有してもよい炭素数1~20のアルキル基、置換基Rを有してもよい環形成炭素数3~18のシクロアルキル基、置換基Rを有してもよい炭素数1~20のアルコキシ基、置換基Rを有してもよい環形成炭素数3~20のシクロアルコキシ基、置換基Rを有してもよい環形成炭素数6~18のアリールオキシ基、置換基Rを有してもよいシリル基、フルオロ基、シアノ基、置換基Rを有してもよい環形成炭素数6~18のアリール基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリール基であり、R11が複数存在する場合、それぞれ同一でも異なっていてもよく、R12が複数存在する場合、それぞれ同一でも異なっていてもよい。
pは、0~3の整数であり、nが2以上の場合、複数のpはそれぞれ同一でも異なっていてもよい。
qは、0~3の整数である。
置換基Rは、炭素数1~20のアルキル基、環形成炭素数3~18のシクロアルキル基、炭素数1~20のアルコキシ基、環形成炭素数3~20のシクロアルコキシ基、環形成炭素数6~18のアリールオキシ基、シリル基、フルオロ基、シアノ基、環形成炭素数6~18のアリール基、又は環形成原子数5~18のヘテロアリール基であり、Rが複数存在する場合、それぞれ同一でも異なっていてもよい。
Czは、下記式(2-1)、(2-2)、(3-1)、(3-2)、(4-1)、(4-2)、(5-1)、(5-2)、(6-1)、(6-2)、(7-1)、(7-2)、(8-1)、及び(8-2)からなる群より選ばれる一つの基であり、nが2以上の場合、複数のCzはそれぞれ同一でも異なっていてもよい。
Yは、酸素(O)原子又は硫黄(S)原子であり、nが2以上の場合、複数のYはそれぞれ同一でも異なっていてもよく、
m1及びm2は、それぞれ独立に、0~4の整数であり、
m3は、0~3の整数であり、
m4は、0~2の整数であり、
m5は、0~4の整数であり、
R0、R1、R2及びR3は、それぞれ独立に、置換基Rを有してもよい炭素数1~20のアルキル基、置換基Rを有してもよい環形成炭素数3~18のシクロアルキル基、置換基Rを有してもよい炭素数1~20のアルコキシ基、置換基Rを有してもよい環形成炭素数3~20のシクロアルコキシ基、置換基Rを有してもよい環形成炭素数6~18のアリールオキシ基、置換基Rを有してもよいシリル基、フルオロ基、シアノ基、置換基Rを有してもよい環形成炭素数6~18のアリール基、又は置換基Rを有してもよい環形成原子数5~18のヘテロアリール基であり、R0が複数存在する場合、それぞれ同一でも異なっていてもよく、R1が複数存在する場合、それぞれ同一でも異なっていてもよく、R2が複数存在する場合、それぞれ同一でも異なっていてもよく、R3が複数存在する場合、それぞれ同一でも異なっていてもよい。)) The material for organic electroluminescent elements represented by following formula (1).
X is an oxygen (O) atom or a sulfur (S) atom. When n is 2 or more, the plurality of X may be the same or different.
L 1 and L 2 are each a single bond, an arylene group having 6 to 18 ring carbon atoms which may have a substituent R, or an arylene group having 5 to 18 ring atoms which may have a substituent R. When it is a heteroarylene group and n is 2 or more, the plurality of L 1 and the plurality of L 2 may be the same or different.
L 3 has an n + q valent saturated aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have a substituent R, an n + q valent silicon-containing group which may have a substituent R, and a substituent R. And an n + q valent aromatic hydrocarbon group having 6 to 18 ring carbon atoms, or an n + q valent unsaturated heterocyclic group having 5 to 18 ring atoms which may have a substituent R.
R 11 and R 12 are each an alkyl group having 1 to 20 carbon atoms that may have a substituent R, a cycloalkyl group having 3 to 18 ring carbon atoms that may have a substituent R, and a substituent. An alkoxy group having 1 to 20 carbon atoms which may have R, a cycloalkoxy group having 3 to 20 ring carbon atoms which may have a substituent R, and a ring forming carbon number which may have a substituent R 6-18 aryloxy group, silyl group optionally having substituent R, fluoro group, cyano group, aryl group having 6-18 ring forming carbon atoms optionally having substituent R, or substituent R A heteroaryl group having 5 to 18 ring atoms that may have a ring structure, and when there are a plurality of R 11 s , they may be the same or different, and when there are a plurality of R 12 s , they may be the same or different from each other. May be.
p is an integer of 0 to 3, and when n is 2 or more, the plurality of p may be the same or different.
q is an integer of 0 to 3.
The substituent R is an alkyl group having 1 to 20 carbon atoms, a cycloalkyl group having 3 to 18 ring carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms, or a ring forming carbon. An aryloxy group having 6 to 18 atoms, a silyl group, a fluoro group, a cyano group, an aryl group having 6 to 18 ring carbon atoms, or a heteroaryl group having 5 to 18 ring atoms, and when there are a plurality of R These may be the same or different.
Cz is represented by the following formulas (2-1), (2-2), (3-1), (3-2), (4-1), (4-2), (5-1), (5- 2) one group selected from the group consisting of (6-1), (6-2), (7-1), (7-2), (8-1), and (8-2) , N is 2 or more, the plurality of Cz may be the same or different.
Y is an oxygen (O) atom or a sulfur (S) atom, and when n is 2 or more, the plurality of Y may be the same or different,
m 1 and m 2 are each independently an integer of 0 to 4,
m 3 is an integer from 0 to 3,
m 4 is an integer from 0 to 2,
m 5 is an integer from 0 to 4,
R 0 , R 1 , R 2 and R 3 are each independently an alkyl group having 1 to 20 carbon atoms which may have a substituent R, or a ring having 3 to 3 carbon atoms which may have a substituent R. An cycloalkyl group having 18 carbon atoms which may have a substituent R, an alkoxy group having 1 to 20 carbon atoms, a cycloalkoxy group having 3 to 20 ring carbon atoms which may have a substituent R, and a substituent R; An aryloxy group having 6 to 18 ring carbon atoms, a silyl group optionally having substituent R, a fluoro group, a cyano group, and 6 to 18 ring carbon atoms optionally having substituent R Or a heteroaryl group having 5 to 18 ring atoms that may have a substituent R, and when a plurality of R 0 are present, they may be the same or different, and a plurality of R 1 are present. If you, or different and each identical, if R 2 there are a plurality, Re may be the same or different, respectively, when the R 3 there are a plurality may each be the same or different. ))
The light-emitting layer contains a phosphorescent material, and the phosphorescent material is an orthometalated complex of a metal atom selected from iridium (Ir), osmium (Os), and platinum (Pt). The organic electroluminescent element in any one.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-123591 | 2012-05-30 | ||
| JP2012123591 | 2012-05-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013179645A1 true WO2013179645A1 (en) | 2013-12-05 |
Family
ID=49672862
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/003353 Ceased WO2013179645A1 (en) | 2012-05-30 | 2013-05-28 | Organic-electroluminescent-element material, and organic electroluminescent element using same |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW201412746A (en) |
| WO (1) | WO2013179645A1 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015004896A1 (en) * | 2013-07-11 | 2015-01-15 | 出光興産株式会社 | Compound and organic electroluminescent element using same |
| WO2016133097A1 (en) * | 2015-02-16 | 2016-08-25 | 出光興産株式会社 | Compound, organic electroluminescent element material, organic electroluminescent element, and electronic device |
| JP2017522732A (en) * | 2014-07-17 | 2017-08-10 | ローム・アンド・ハース・エレクトロニック・マテリアルズ・コリア・リミテッド | Electron transport material and organic electroluminescent device containing the same |
| KR20170136823A (en) * | 2016-06-02 | 2017-12-12 | 삼성전자주식회사 | Condensed cyclic compound and organic light emitting device including the same |
| WO2018230860A1 (en) * | 2017-06-14 | 2018-12-20 | 주식회사 엘지화학 | Novel compound and organic light emitting device comprising same |
| CN112898305A (en) * | 2019-12-03 | 2021-06-04 | 乐金显示有限公司 | Organic compound, organic light emitting diode comprising the same, and organic light emitting device comprising the same |
| JPWO2021157642A1 (en) * | 2020-02-04 | 2021-08-12 | ||
| WO2022134074A1 (en) * | 2020-12-25 | 2022-06-30 | 京东方科技集团股份有限公司 | Organic electroluminescent device, display panel, display device and light-emitting device |
| WO2022264857A1 (en) * | 2021-06-15 | 2022-12-22 | 株式会社Kyulux | Organic light emitting element and method for producing same |
| JPWO2022270592A1 (en) * | 2021-06-23 | 2022-12-29 | ||
| WO2023120062A1 (en) * | 2021-12-23 | 2023-06-29 | 株式会社Kyulux | Electronic barrier material and organic semiconductor element |
| US12133459B2 (en) | 2019-01-15 | 2024-10-29 | Lg Chem, Ltd. | Heterocyclic compound and organic light emitting device comprising same |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009148062A1 (en) * | 2008-06-05 | 2009-12-10 | 出光興産株式会社 | Polycyclic compound and organic electroluminescent device using the same |
| WO2009148015A1 (en) * | 2008-06-05 | 2009-12-10 | 出光興産株式会社 | Halogen compound, polycyclic compound, and organic electroluminescence element comprising the polycyclic compound |
| WO2011125020A1 (en) * | 2010-04-06 | 2011-10-13 | Basf Se | Substituted carbazole derivatives and use thereof in organic electronics |
| JP2012028548A (en) * | 2010-07-23 | 2012-02-09 | Konica Minolta Holdings Inc | Material for organic electroluminescence element, and organic electroluminescence element, and display device and illuminating device using organic electroluminescence element |
| EP2450975A2 (en) * | 2010-11-04 | 2012-05-09 | Cheil Industries Inc. | Compound for organic optoelectronic device and organic light emitting diode including the same |
| KR20120078301A (en) * | 2010-12-31 | 2012-07-10 | 제일모직주식회사 | Compound for organic photoelectric device and organic photoelectric device including the same |
| WO2013012297A1 (en) * | 2011-07-21 | 2013-01-24 | Rohm And Haas Electronic Materials Korea Ltd. | Novel organic electroluminescence compounds and organic electroluminescence device using the same |
-
2013
- 2013-05-28 WO PCT/JP2013/003353 patent/WO2013179645A1/en not_active Ceased
- 2013-05-30 TW TW102119097A patent/TW201412746A/en unknown
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009148062A1 (en) * | 2008-06-05 | 2009-12-10 | 出光興産株式会社 | Polycyclic compound and organic electroluminescent device using the same |
| WO2009148015A1 (en) * | 2008-06-05 | 2009-12-10 | 出光興産株式会社 | Halogen compound, polycyclic compound, and organic electroluminescence element comprising the polycyclic compound |
| WO2011125020A1 (en) * | 2010-04-06 | 2011-10-13 | Basf Se | Substituted carbazole derivatives and use thereof in organic electronics |
| JP2012028548A (en) * | 2010-07-23 | 2012-02-09 | Konica Minolta Holdings Inc | Material for organic electroluminescence element, and organic electroluminescence element, and display device and illuminating device using organic electroluminescence element |
| EP2450975A2 (en) * | 2010-11-04 | 2012-05-09 | Cheil Industries Inc. | Compound for organic optoelectronic device and organic light emitting diode including the same |
| KR20120078301A (en) * | 2010-12-31 | 2012-07-10 | 제일모직주식회사 | Compound for organic photoelectric device and organic photoelectric device including the same |
| WO2013012297A1 (en) * | 2011-07-21 | 2013-01-24 | Rohm And Haas Electronic Materials Korea Ltd. | Novel organic electroluminescence compounds and organic electroluminescence device using the same |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10505123B2 (en) | 2013-07-11 | 2019-12-10 | Idemitsu Kosan Co., Ltd. | Compound and organic electroluminescent device using same |
| WO2015004896A1 (en) * | 2013-07-11 | 2015-01-15 | 出光興産株式会社 | Compound and organic electroluminescent element using same |
| JP2017522732A (en) * | 2014-07-17 | 2017-08-10 | ローム・アンド・ハース・エレクトロニック・マテリアルズ・コリア・リミテッド | Electron transport material and organic electroluminescent device containing the same |
| CN107207522A (en) * | 2015-02-16 | 2017-09-26 | 出光兴产株式会社 | Compound, material for organic electroluminescence element, organic electroluminescence element and electronic device |
| WO2016133097A1 (en) * | 2015-02-16 | 2016-08-25 | 出光興産株式会社 | Compound, organic electroluminescent element material, organic electroluminescent element, and electronic device |
| KR20170136823A (en) * | 2016-06-02 | 2017-12-12 | 삼성전자주식회사 | Condensed cyclic compound and organic light emitting device including the same |
| KR102587955B1 (en) * | 2016-06-02 | 2023-10-16 | 삼성전자주식회사 | Condensed cyclic compound and organic light emitting device including the same |
| WO2018230860A1 (en) * | 2017-06-14 | 2018-12-20 | 주식회사 엘지화학 | Novel compound and organic light emitting device comprising same |
| KR20180136377A (en) * | 2017-06-14 | 2018-12-24 | 주식회사 엘지화학 | Novel compound and organic light emitting device comprising the same |
| CN110088110A (en) * | 2017-06-14 | 2019-08-02 | 株式会社Lg化学 | Novel compound and organic light-emitting element including the same |
| KR102050572B1 (en) * | 2017-06-14 | 2019-12-02 | 주식회사 엘지화학 | Novel compound and organic light emitting device comprising the same |
| CN110088110B (en) * | 2017-06-14 | 2022-09-13 | 株式会社Lg化学 | Novel compound and organic light-emitting element comprising same |
| US12133459B2 (en) | 2019-01-15 | 2024-10-29 | Lg Chem, Ltd. | Heterocyclic compound and organic light emitting device comprising same |
| CN112898305A (en) * | 2019-12-03 | 2021-06-04 | 乐金显示有限公司 | Organic compound, organic light emitting diode comprising the same, and organic light emitting device comprising the same |
| WO2021157642A1 (en) * | 2020-02-04 | 2021-08-12 | 株式会社Kyulux | Host material, composition, and organic electroluminescent element |
| JPWO2021157642A1 (en) * | 2020-02-04 | 2021-08-12 | ||
| JP7594794B2 (en) | 2020-02-04 | 2024-12-05 | 株式会社Kyulux | Host material, composition and organic light-emitting device |
| US12163074B2 (en) | 2020-02-04 | 2024-12-10 | Kyulux, Inc. | Host material, composition, and organic electroluminescent element |
| CN114981274A (en) * | 2020-12-25 | 2022-08-30 | 京东方科技集团股份有限公司 | Organic electroluminescent device, display panel, display apparatus, and light-emitting apparatus |
| WO2022134074A1 (en) * | 2020-12-25 | 2022-06-30 | 京东方科技集团股份有限公司 | Organic electroluminescent device, display panel, display device and light-emitting device |
| US12356855B2 (en) | 2020-12-25 | 2025-07-08 | Boe Technology Group Co., Ltd. | Organic electroluminescence device, display panel, display device, and light emitting device |
| WO2022264857A1 (en) * | 2021-06-15 | 2022-12-22 | 株式会社Kyulux | Organic light emitting element and method for producing same |
| JPWO2022270592A1 (en) * | 2021-06-23 | 2022-12-29 | ||
| WO2022270592A1 (en) * | 2021-06-23 | 2022-12-29 | 株式会社Kyulux | Electron barrier material, organic semiconductor element and compound |
| WO2023120062A1 (en) * | 2021-12-23 | 2023-06-29 | 株式会社Kyulux | Electronic barrier material and organic semiconductor element |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201412746A (en) | 2014-04-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6212391B2 (en) | Organic electroluminescence device | |
| JP6148982B2 (en) | Nitrogen-containing heteroaromatic ring compounds | |
| JP6012611B2 (en) | Material for organic electroluminescence device and organic electroluminescence device using the same | |
| JP6196554B2 (en) | Material for organic electroluminescence device and organic electroluminescence device using the same | |
| WO2013179645A1 (en) | Organic-electroluminescent-element material, and organic electroluminescent element using same | |
| JP6220341B2 (en) | Ladder compound and organic electroluminescence device using the same | |
| JPWO2013105206A1 (en) | Material for organic electroluminescence element and element using the same | |
| WO2013069242A1 (en) | Material for organic electroluminescent elements, and organic electroluminescent element using same | |
| WO2013175746A1 (en) | Organic electroluminescent element | |
| WO2013102992A1 (en) | Material for organic electroluminescence element and element using same | |
| JP2013108015A (en) | Material for organic electroluminescent element | |
| JPWO2013108589A1 (en) | NOVEL COMPOUND, MATERIAL FOR ORGANIC ELECTROLUMINESCENT DEVICE AND ORGANIC ELECTROLUMINESCENT DEVICE | |
| JP6031302B2 (en) | Heteroaromatic compound and organic electroluminescence device using the same | |
| US9496508B2 (en) | Material for organic electroluminescent element and organic electroluminescent element using same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13797678 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13797678 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |