WO2013100772A1 - The use of vitamin d analogues in combination with imatinib in therapy of non-small cell lung cancer - Google Patents
The use of vitamin d analogues in combination with imatinib in therapy of non-small cell lung cancer Download PDFInfo
- Publication number
- WO2013100772A1 WO2013100772A1 PCT/PL2012/000134 PL2012000134W WO2013100772A1 WO 2013100772 A1 WO2013100772 A1 WO 2013100772A1 PL 2012000134 W PL2012000134 W PL 2012000134W WO 2013100772 A1 WO2013100772 A1 WO 2013100772A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- imatinib
- vitamin
- analogues
- lung cancer
- combination
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/59—Compounds containing 9, 10- seco- cyclopenta[a]hydrophenanthrene ring systems
- A61K31/593—9,10-Secocholestane derivatives, e.g. cholecalciferol, i.e. vitamin D3
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- the invention relates to the use of vitamin D analogues in combination with tyrosine kinase inhibitor imatinib in therapy of non-small cell lung cancer.
- Fig, l A depicts kinetics of a tumour growth in mice (all groups) with non-small cell lung cancer A54 treated with imatinib (GV) (50 mg/kg daily) in combination with analogues of vitamin D.
- GV imatinib
- PRI-2191 dose 1 ⁇ g/kg day, administered subcutaneousl per half of body weight, multiple administration, 3 times per week (on days 10, 12, 14, 17, 19, 21 , 24, 26, 28).
- tumour growth inhibition was carried out by analysis of percent of inhibition in individual combinations of compounds.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Description
The use of vitamin D analogues in combination with imatinib in therapy of non- small cell lung cancer
Field of the invention The invention relates to the use of vitamin D analogues in combination with tyrosine kinase inhibitor imatinib in therapy of non-small cell lung cancer.
Background of the invention
Lung cancer is the most common malignant lung cancer originating from cell lining bronchial tree. Lung cancer morbidity is still increasing. In Poland, lung cancer is the first cause of morbidity in men and the second in women, and the rate of morbidity increase is one of the highest in the world. Mortality due to lung cancer is very high. About 80% of the patients die within the first year after the diagnosis, and only 10% of patients survive 5 years after lung cancer diagnosis. Lung cancer includes many different histological subtypes of the cancer. From the point of view of treatment strategies, lung cancer is divided into two groups of the cancer: small-cell cancers (anaplastic) consisting mainly of non-differentiated cells and non-small cell cancers originating from epithelial cells, differentiated into glandular cells or cells of squamous epithelium. In treatment of the lung cancer, three therapeutic methods are used - surgical treatment, radiotherapy and chemotherapy.
The selection of the appropriate treatment method depends mainly upon histological type of cancer and a degree of its progression.
In treatment of non-small cell cancers, the most appropriate method is surgical treatment. The best prognosis of survival is with radical procedures - lobectomy, pneumonectomy and radical pneumonectomy. The same type of procedure is also carried out when the secondary treatment with chemotherapy or radiotherapy is planned. Often, in non-operable tumours, the attempts of administration of
chemotherapy are made in order to eliminate the part of the tumour and reducing its size to size that qualifies for radical procedure,
In the advanced non-small cell lung cancer, the first line treatment method is chemotherapy, in many cases combined with radiotherapy. Usually, two drug chemotherapy is used based on platinum compounds (cisplatin or carboplatin) combined with one of the third generation drugs (most often vinorelbine, gemcitabine, docetaxel or pemetrexed). In case when no response is seen to the first line therapy or a case of recurrence of the disease, the second line therapy may be considered (monotherapy with docetaxel, pemetrexed or erlotinibe), and in the case of its failure - the third line therapy (erlotinibe).
In recent years, cancer treatment was revolutionized by molecules directed at growth and signal pathways in cancer cells, such as erlotinibe and bevacizumab used in non-small cell lung cancer.
One of such molecules is also imatinib, a compound named 4-[(4-methyl-l - piperazin- l-ylomethyl)-N[[4-methyl-3-[[4-(3-pyridynyl)-2-pirymidynyl]amino3- phenyl]]benzamide, which, among other compounds exhibiting activity towards tyrosine kinases receptors, has been disclosed in European patent application EP-A- 0 564-409.
Protein kinases which include among others a family of serine-threonine kinases and family of tyrosine kinases, catalyze protein phosphorylation, which is indispensable step of cell differentiation and division. Tyrosine kinases include: receptor kinase of platelet-derived growth factor (PDGF), Abl kinase and c-Kit kinase. Deregulation of activity of tyrosine kinases, for example by a way of mutation or activation by factors bound to them, such as natural endogenous compounds, e.g. PDGF or exogenous compounds, is a common cause of cell growth disturbances, and development of neopastic diseases as a consequence.
Tyrosine kinases play a key role in transduction pathways of intracellular signals, leading to different cell responses, such as proliferation, apoptosis and differentiation. As a consequence, these enzymes became basic targets for development of new drugs intended for blockade of proliferation, metastasis, angiogenesis and supporting of apoptosis of cancer cell.
Imatinib is characterised by especially high selectivity of inhibition of tyrosine kinase which is a result of translocation of fusion protein Bcr-Abl and c-Abl compared to other tyrosine kinases, but preliminary studies show that it inhibits also tyrosine kinase of a receptor of platelet-derived growth factor PDGFR and tyrosine kinase of c- Kit receptor in vitro. As a result, imatimb may potentially show efficacy in treatment of malignant neoplasms, in which activation of these receptors play a significant role.
The receptor of growth factor c-Kit is expressed in solid tumours, including small-cell lung cancer (SCLC), at least 70% of cases of small-cell lung cancer are characterized by expression of tyrosine kinase c-Kit and its ligand - stem cell growth factor (SCF). In a publication G. W. Krystal el al, Clin. Cancer Res., 2000, 8, 11 3319-3326, The selective Tyrosine Kinase Inhibitor STI571 Inhibits Small Cell Lung Cancer Growth, the hypothesis was proved that Imatinib inhibits phosphorylation of tyrosine kinase similar to c-kit protein and its activity in vitro in cells of small cell lung cancer. Due to an important role which receptor of platelet derived growth factor (PDGFR) and its ligand - PDGF play in signalization within cells, this receptor became an attractive therapeutic target in various malignant neoplasms. PDGF acts on cells through two different receptors of tyrosine kinase: a (PDGFR a) and receptor β (PDGFR β). Biological role of PDGF in development of neoplasm includes autocrine stimulation of neoplastic cells to paracrine stimulation of interstitium adjacent to the tumour and vascular system (Mantur M., Roper O.; Pfytkopochodny czynnik wzrostu - budowa. rola i jego receptory. Pol. Merk. Lek., 2008, XXIV, 140, 173).
Imatinib, apart from inhibition of Bcr-Abl in chronic myeloplastie leukemia, shows also strong PDGFR inhibitory properties. It was shown that it is effective in both types of neoplasms in which PDGF plays a role - dermatofibrosareoma protuberans and glioma multiforme. The increased expression of PDGF constitutes a bad prognostic factor in a human small- cell lung cancer. In the examination of a tissue taken from patients with NSCLC, the expression of PDGFR β limited to tumour stroma was found and reactivity of PDGF β was documented in cancer cells in 60% of samples (Bauman J.E. et al.: Antagonism of Platelet-Derived Growth Factor Receptor in Non-Small Cell Lung Cancer: Rationale and Investigations, Clin Cancer Res 2007, 13(15 Suppl), August 1 , 2007). It is known that PDGF β induces the growth of neoplastic tumour and is responsible for metastases through development of lymphatic vessels (Mantur M., Koper O..' Pfytkopochodny czynnik wzrostu - budowa, rola ijego receptory. Pol. Merk. Lek., 2008, XXIV, 140, 173).
In other studies in which expression of PGDF was examined in cell lines of human lung cancer and samples after surgical resection of the tumour, it was observed that PGDFot was expressed both in squamous cells cell lines and adenocarcinoma and seemed to regulate strictly the expression of growth factor of vascular endothelium (VEGF). In a xenogenieally transplanted NSCLC model, tumour growth correlated with PDGFR expression, and PDGF loss caused inhibition of tumour growth, a decrease of VEGF level and density of microvessels in the tumour. Probably it is related to occurrence of paracrine loop concerning, on one hand, PDGF expression in tumour cells, and on the other hand expression of receptor for PDGF (PDGFR) in microenvironment of the neoplasm. This hypothesis was verified in .clinical studies on NSCLC with imatinib combination, carried out by scientists from Washington University.
Another interesting direction of studies on targeted therapy in this type of cancer are strategies concerning special conditions existing in environment of neoplastic tumour localized in the lung tissue, because there are factors which contribute to a weak response of the tumour to chemo- and radiotherapy through limitation of drug availability. These are: high interstitial fluid pressure (IFP) and hypoxia ((Vlahovic G. et at, Treatment with Imatinib in NSCLC is associated with decrease of phosphorylated PDGFR-beta and VEGF expression, decrease in interstitial fluid pressure and improvement of oxygenation. Br. J. Cancer 2006; 95:1013-1019.). In these studies, the concept aiming at a decrease of interstitial fluid pressure (IFP) is verified through targeted action on PDGFR, tested previously in preclinical studies of different tumour models. The increased interstitial fluid pressure is characteristic for solid tumours and it is thought that it disturbs transport of chemotherapeutics through capillaries. Phosphorylated PDGFR, a receptor of tyrosine kinase activity, is considered a strategic target for a decrease of IFP in tumours with its overexpression. At the same time, NSCLC tumours showing often PDGFR expression should be susceptible to the use of therapeutic strategies targeted against this receptor {(Bauman J.E. et ah, Antagonism of platelet-derived growth facto receptor in non small cell lung cancer: rationale and investigations Clin.Cancer Res. 2007;13:4632-4636), Data from preclinical studies show that antagonistic action on receptor of platelet-derived growth factor decreases interstitial fluid pressure, increases concentration of chemotherapeutic within the tumour and disturbs neoplasm growth.
At the same time it was shown that stem cells isolated from NSCLC line show c- kit expression and that signal axis SCF-c-kit plays an important role in self-regeneration and proliferation of stem cells in lung cancers ((Levina V. et al., Elimination of human lung cancer stem ceils through targeting of the stem cell factor -c-kit autocrine signaling loop. Cancer Res. 2010;70:338-346.), Blockade of signal pathway of SCF-c-kit, similarly to PDGF, should improve therapeutic efficacy of NSCLC chemotherapy. The drug which therapeutic targets are both PDGFR and c-kit, is imatinib (Zhang P. et al., Gleevec (STI-571) inhibits lung cancer cell growth (A549) and potentiates the cisplatin effect in vitro. Mol. Cancer 2003;2:1., Wang W.L .et al. Growth inhibition and modulation of kinase pathways of small cell lung cancer cell lines by the novel tyrosine kinase inhibitor STI 571. Oncogene 2000;19:3521-3528., Vlahovic G. et al, Treatment with imatinib in NSCLC is associated with decrease of phosphorylated PDGFR-beta and VEGF expression, decrease in interstitial fluid pressure and improvement of oxygenation. Br.J.Cancer 2006;95: 1013-1019). It has been shown by now that the use of therapy targeted at these factors with the use of tyrosine kinase inhibitors, such as imatinib, causes a decrease of p-PDGFR-beta, VEGF and IFP, moreover, oxidation of NSCLC tumour improves (Vlahovic G. et al, Treatment with Imatinib in NSCLC is associated with decrease of phosphorylated PDGFR-beta and VEGF expression, decrease in interstitial fluid pressure and improvement of oxygenation. Br.J.Cancer 2006;95:1013-1019).
Different attitude to the combination therapy of NSCLC uses the fact that in compartment of tumour vascularisation (the second compartment apart from neoplastic cells at which the therapy with neoplastic substances is targeted), there are two cell types independently: endothelial cells and pericytes. Data from preclinical studies suggests that the action double targeting at pericytes which function is regulated by PDGFR and endothelial cells which physiological and pathological angiogenesis, is dependent on signal pathway VEGF may constitute a more synergic and effective strategy against angiogenesis than therapy targeted only against VEGF. This data are currently verified in clinical trials testing a new maintenance strategy with the use of imatinib and antibody blocking process of angiogenesis of blood vessels of endothelium, bevacizumab, after failure of the first line therapy with bevacizumab.
Thus, current concepts in antineoplastic treatment, including treatment of non- small cell lung cancer, aim at combining different modulators of signal pathways in a cell.
The above reports have inspired present Inventors to carry out studies on the use of combination of modulator of signal pathways imatinib and calcitriol analogues in a model of non-small cell lung cancer.
Calcitriol, hormonally active form of vitamin D, similarly to imatinib, possesses activity regulating cell cycle, however the effects of action of both compounds probably occur through different molecular mechanisms. The results of in vitro and in vivo studies concerning cytostatic activity of calcitriol and its analogues indicate possibility of use of these compounds in treatment of patients with some types of cancer (Ma Y. et al, Vitamin D in combination cancer treatment, Journal of Cancer 2010; 1, 101-107). It i s well-known that in the development of a neopiasm, similarly as in the course of other hyperproliferative diseases, such as psoriasis, a disturbance occurs in a balance between proliferation and apoptosis processes. Induction of differentiation of cells with calcitriol and its analogues may restore this balance and, as a consequence, lead to inhibition of neoplasm growth (Diaz G.D. et al., Apoptosis i induced by the active metabolite of vitamin D3 and its analogue EB1089 in colorectal adenoma and carcinoma cells: possible implications for prevention and therapy. Cancer Res 60:2304-2312, 2000). As it was shown previously in a model of head and neck cancer and human leukemia HL-60, combined use of calcitriol and analogue PRI-2191 with imatinib enhances antiproliferative action of imatinib causing at the same time synergistic effect in action of both drugs (Wietrzyk J. Et al. The effect of combined treatment on head and neck human cancer cell lines with novel analogs of calcitriol and cytostatics. Oncol Res 16:517-525,. 2007, Switalska M. et al, The in-vitro antiproliferative effect of PRI-2191 and imatinib applied in combined treatment with cisplatin, idarubicin, or docetaxel on human leukemia cells. Anticancer Drugs 2011).
Calcitriol and its analogues are good candidates for combined treatment with imatinib due to the fact that calcitriol influences signal pathways activated by protein kinase activated by myogenes (MAPK), especially cascades Raf/MEK/ERK and INK, which are also common elements for signal pathways activated by activation of c- it or Bcr-Abl kinases. It is also known that during differentiation of cells HL-60 calcitriol decreases expression PDGFR (receptor of platelet-derived growth factor), a kinase
being the one of targets for imatinib (Reiterer G, Yen A. Platelet-derived growth factor receptor regulates myeloid and monocytic differentiation of HL-60 cells. Cancer Res. 2007;67:7765-7772).
The results of studies published recently show that antineoplastic activity of calcitriol is related to activation of inhibition of different signal pathways in a cell. It is known that calcitriol, apart from classic genome effects, regulates many cellular signal pathways transferred by protein kinase C, ras, MAPK, protein lipase A and prostaglandines, cyclic AMP and protein kinase A, phosphatydylinositiol 3 and other. Activation of cytoplasmatic signal pathway often causes fast changes in calcium level in the cell and activation or inactivation such proteins as bcl-2 or e-jun. As a consequence, many of these signal pathways influence cell proliferation, differentiation, apoptosis and may work with classical genome pathways.
Also, a significant role of vitamin D receptor (VDR) in proliferation of endothelial cells originating from the tumour and antianglogenic action of calcitriol was shown. Upon the use of VDR gene knockout mice (VDR KO), it was found that in these animals, more impaired vasculature is formed within developing neoplastic tumours (transplantable) compared with wild type mice. Moreover, widening and deformation of blood vessels in VDR KO mice were related to smaller number of pericytes and an increased angiogenic factors such as: HIF-lalfa, VEGF, Angl and PDGF-BB. Significance of VDR in regulation of these factors is not fully understood. In case of different human neoplasms it was shown that calcitriol inhibits expression HIF-1 alfa protein and its target genes: VEGF, endotheline-1 and glucose- 1 transporter. At the same time, cells of vascular endothelium isolated from tumours of mice VDR KO treated with calcitriol did not react with a decrease of VEGF, Angl and PDGF-BB contrary to cells isolated from wild type mice (Chung I. et ah, Role of vitamin D receptor in the antiproliferative effects of calcitriol in tumor-derived endothelial cells and tumor angiogenesis in vivo. Cancer Res 69: 967-975).
Considering the mechanism of action of both imatinib and calcitriol, it was assumed that these compounds may act either on the same signal pathways but on their different steps or on similar processes occurring within the neoplasm and thus may support their mutual antineoplastic action. This hypothesis was confirmed by results of in vitro studies in a model human non- small cell lung cancer A549, where it was shown that selected analogues of vitamin D not only slow down tumour growth themselves while used independently but also enhance antineoplastic effect of imatinib. Thus, antineoplastic activity of imatinib including both inhibition of proliferation and induction of apoptosis may beneficially work with activity of these analogues.
Brief description of drawings
Fig, l A depicts kinetics of a tumour growth in mice (all groups) with non-small cell lung cancer A54 treated with imatinib (GV) (50 mg/kg daily) in combination with analogues of vitamin D.
Fig. 1 B depicts kinetics of tumour growth in mice with non-small cell lung cancer A549 treated with imatinib (GV) (50 mg/kg daily) in combination with vitamin D analogues.
Fig. 2 depicts changes of body mass of NQD/SCID mice with human non-small cell lung cancer A549 during treatment with imatinib and vitamin D analogues.
Fig. 3 depicts total calcium blood level in mice after the end of treatment [mmol/L](day 34).
Disclosure of the invention The invention relates to the use of vitamin D analogues selected from tacalcitol and (lS,3R,5E,7¾22£,24S)-24-cyclopropyI-9,10-secochoIa-5,7,10(19),2
1,3,24-triol (5,6-trans-calcipotriol) in combination with tyrosine kinase inhibitor imatinib in treatment of non-small cell lung cancer.
In in vivo studies in model of human non-small cell lung cancer A549 imatinib decreases tumour mass in statistically significant manner compared to control only in case of combined use with vitamin D analogues.
Synergic action of compounds evaluated on the basis of parameter of tumour growth inhibition (TGI) indicates that combined use vitamin D analogues with imatinib inhibits tumour growth in additive manner, turning into synergism, in the further days of the experiment, especially in case of tacalcitol administration.
The use of combination included in the invention is safe and do not cause risk of hypercalcaemia and hyperphosphataemia what is confirmed by the observed lack of increase of total calcium level in blood serum of mice both after administration of vitamin D analogue and imatinib independently, and after administration of combination of imatinib and each of studied vitamin D analogues.
Based on the results of studies of combination in vivo it may be found that vitamin D analogues enhance beneficial antineoplastic action of imatinib, so that they may be used in combination therapy with the indication for treatment of non-small cell lung cancer.
Components of the combination may be administered to the patient in the form of one or separate pharmaceutical agents containing therapeuticall effective amount of active substance(-s) in combination with known pharmaceutically acceptable carriers and/or excipients. Imatinib may be administered in the form of free base or acid addition salt, especially with methanesulfonic acid. Imatinib addition salt with methanesulfonic acid, known as imatinib mesylate may be used in any crystalline form, for example in crystalline form a or β known from International Patent Publication WO 99/03854.
Tacalcitol may be administered in an anhydrous or solvated form, preferably as monohydrate.
Therapeutically effective doses of components of the combination in treatment of non-small cell lung cancer may be determined by specialist physician based on the results of clinical trials and adjusted according to disease progression, patient's age and weight, risk of disease development in a certain individual and the route of administration.
A daily dose of a substance may be administered to the patient as a single unit dose once daily or divided into several daily doses.
Therapeutically effective daily dose of vitamin D analogue in case of an adult may be from 0.1 to 200 μg of the active substance, preferably from 0.5 to 50 μg. Therapeutically effective daily doses of imatinib in treatment of leukemia in humans are from 100 to 800 mg of the active substance, and the recommended single dose for oral administration is 100 to 400 mg. Thus, imatinib may be administered as a unit dose including 119.5 or 478 mg of imatinib mesylate, respectively.
In case of the present invention, due to synergic action with vitamin D analogue, daily dose of imatinib may be reduced respectively.
In general, therapeutically effective daily dose of imatinib is from 50 to 1000 mg of the active substance corresponding to a free base. This dose may be administered once or twice daily.
As in vivo studies in mouse model of human non-small eell lung cancer A549 show, the route of administration of the components of the combination significantl influences their antineoplastic activity. Clear synergic therapeutic effect of imatinib and vitamin D analogue is observed upon administration of imatinib intraperitoneally in combination with subcutaneous administration of tacalcitol, when 40% inhibition of tumour growth is seen compared with control group.
The obtained results of animal studies confirm efficacy of intraperitoneal chemotherapy combined with subcutaneous. Combination of these two methods of administration of anticancer drugs in humans may allow a decrease of administered doses of both components of the combination and an increase of treatment efficacy.
Thus, in an embodiment according to invention in treatment of non-small cell lung cancer in humans, imatinib may be administered orally or intravenously, and vitamin D analogue by oral or subcutaneous route.
The pharmaceutical agent, apart from the active substance(-s) may contain known pharmaceutically acceptable carriers and/or excipients, suitable for a given pharmaceutical form, not having their own pharmacological action and not reacting adversely with the active substance(-s).
The pharmaceutical agent may be prepared in the form suitable for general administration, for example oral, such as tablets, capsules, coated tablets, enteric-coated tablets, in form suitable for parenteral administration, such as solutions, suspensions or lyophylisate for reconstitution ex tempore or in the form suitable for local administration. Selection and amount of carriers and excipients depends on the form and route of administration of the agent. In order to prepare suitable drug form, techniques well known to those skilled in the art are used with the use of any pharmaeeutically acceptable carriers, solvents, fillers and other excipients.
The pharmaceutical agent for oral administration may be in the form of capsules. In this case the active substance is combined with the carrier and gelatine capsules are filled with the obtained composition. Capsule filling is in the form of oil solution, suspension or emulsion. Suitable carriers include for example castor oil, coconut oil, olive oil, palm oil, corn oil, peanut oil, synthetic and natural triglycerides of fat acids, unsaturated fat acids of medium length chain, modified long-chain fat acids, glycol
esters, polyethylene glycols and the like. Suitable excipients are tensides, for example lecithin, mono- and di-glycerides, fat acid esters of polyoxyethylenesorbitan.
Capsules may be in the form of soft or hard gelatine capsules differing by composition of gelatine mass for its preparation. Composition of gelatine mass in the case of soft capsules include plasticizers, such as glycerol, sorbitol, preservatives, such as benzoic acid and its salts or alkyl hydroxybenzoates, colouring and favouring agents.
The pharmaceutical agent for parenteral administration may be in the form of ready to use suspension, lyophilisate for ex tempore reeonstitution or concentrate for intravenous infusions. Carriers suitable for intravenous administration of the pharmaceutical agent include for example sterilized water solutions, such as normal saline solution, solutions of carbohydrates, e.g. glucose, mannitol, dextrose, lactose and water solutions of buffers, for example phosphate buffer. Moreover, the agent may contain other excipients, traditionally used in order to ensure iso-osmocity, antioxidants, preservatives and the like. The combination according to invention is a potential option for treatment of non-small cell lung cancer.
Studies on biological activity
Further in the description, the following designations are used:
PRI-2191 - tacalcitol
PRI-2205 - 5,6-trans-caleipotriol
GV - imatinib
In the tudies, tacalcitol monohydrate and isomer of 5,6-trans-calcipotriol were used manufactured in the Pharmaceutical Research Institute in Warsaw.
Imatinib was administered in the form of mesylate, crystalline form a, manufactured in the Pharmaceutical Research Institute.
Amounts and concentrations of the active substances used in the form of hydrates and salts correspond to free compounds.
In vivo studies
Influence of vitamin D analogues on antineoplastic activity of Imatinib in the model of human non-small cell lung cancer A549
Methods
• NOD/SCID mice (female) with non-small cell lung cancer, cancer cells implanted subcutaneously in concentration 5x106 cells/0.25 ml/mouse
• The number of mice in groups: control - 8, PRI-2191 - 8, PRI-2205 - 8, Imatinib (GV) - 8, GV + PRI-21 1 ; GV + PRI-2205 - 8.
» Treatment of mice started on day 7 after implantation of cancer cells (7th day of the experiment),
• Imatinib: dose 50 mg/kg/day, administered intraperitoneal]}-, everyday (on days 7-21)
• PRI-2191 : dose 1 μg/kg day, administered subcutaneousl per half of body weight, multiple administration, 3 times per week (on days 10, 12, 14, 17, 19, 21 , 24, 26, 28).
• PRI-2205: dose 10 μg/kg/day administered subcutaneously per half of bod weight, multiple administration, 3 times per week (on days 10, 12, 14, 17, 19, 21, 24, 26, 28).
On day 34 the experiment was ended, blood and tumours were taken for further analysis.
Blood was analyzed for total serum calcium content.
Subcutaneous tumours were measured during experiment, 3 times per week, Tumour volume [mm ] were calculated according to formula (a x b), wherein a = shorter size of tumour in mm, b = longer tumour size in mm.
Analysis of results
Statistical analysis was carried out according to Kruskal-Wallis ANOVA, test for multiple comparisons and comparison of groups according to Mann- Whitney U-test.
Statistical analysis of the obtained results of tumour growth inhibition (TGI) was carried out by analysis of percent of inhibition in individual combinations of compounds.
TGI [%} was calculated according to the formula:
TGI=I00-{(mean tumour volume in treatment group/mean tumour volume in control)xl00
TGI (%) calculated in such way show percentage by which tumours in a given group are smaller than in control.
Hypothetical value TGI was calculated according to the formula:
%H=100[(1OO-TGI GV) x (100-TGI Wit)/100]
wherein:
%H - hypothetical inhibition of tumour growth by combination of compounds [%] ;
TGI GV - inhibition of tumour growth by imatinib only [%];
TGI Wit - inhibition of proliferation by vitamin D analogue only [%]
Depending on values obtained in the experiments, the obtained values were evaluated as a synergic, additive, sub-additive or antagonistic effect.
When a hypothetical value is below experimental value determined for combination of the used drugs, it confirms a synergism of action of drugs. Additive effect occurs when experimental and hypothetical values are the same, sub-additive effect when experimentally determined value is below the hypothetical value, but above a value for imatinib only, and antagonistic effect - if a value determined for imatinib used as a single drug is above the experimental value for combination of both compounds [Peters G.J. et al.: Pharmacol Ther 2000, 87, 227-253].
TGI was also sho wn as a percentage of tumour volume of control group which constitutes the tumour volume in the examined group. Then tumour volume in control was assumed as 100%, and tumour volume in treated group was expressed as (100%-
%TGI)(7 W 1).
Results
• Kinetics of A549 tumour growth
In order to show action of the used combination of vitamin D analogues with imatinib, percentage of tumour growth inhibition TGI [%] determining how much smaller the tumours are in mice in treated groups compared to tumours in mice in control group receiving placebo (solvent) was calculated. The results are shown in diagrams in Fig 1 A (all groups) and IB - for selected groups,
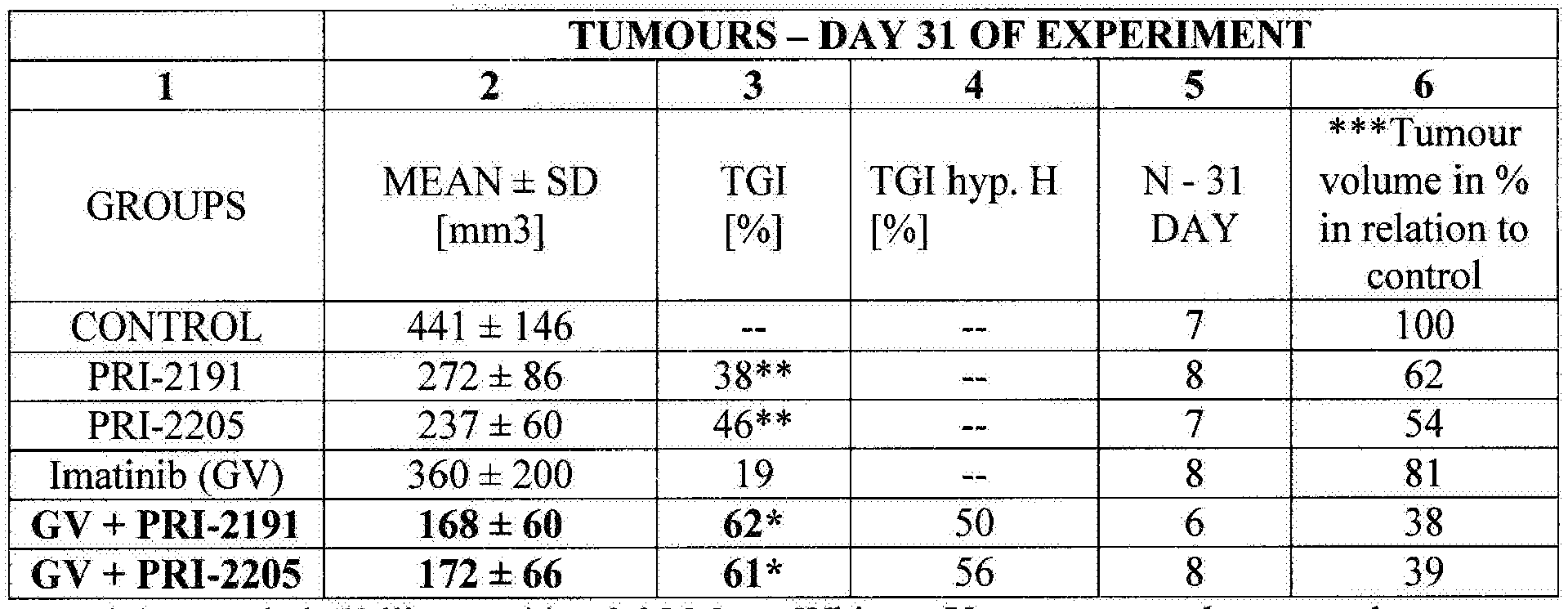
Table 1 shows calculations carried out for the last measurement day of the experiment, where classical parameter TGI is shown in column no. 3. Of all three preparations used independently, imatinib showed the weakest action on A549 tumour growth (1 %). Vitamin D analogues used independently inhibited tumour growth in 38% for PRI-2191 and in 46% for PRI-2205. Combinations of each
analogue with imatinib equally inhibited tumour growth compared to control, imatinib + PRI-2191 by 62%, whereas imatinib + PRI-2205 by 62%. TGI values obtained for combination as well as both vitamin D analogues used independently are statistically significant.
Table 1. The influence of vitamin D analogues on tumour growth in mice with non- small cell lung cancer A549 treated with imatinib - mean tumour volume, standard deviation, % tumour growth inhibition TGI and hypothetical % TGI, N- number of mice in a group.
* p<0.05 Kruskal-Wallis test, **p<0.05 Mann- Whitney U test compared to control
***TGI was shown as percentage of tumour volume in a group.
N- mice number in a group In column 4 of table 1 , hypothetical parameter TGI was shown calculated on the basis of the formula: %H=100-[(100-GI GV)x(100-TGI Wit)/100], By analysing theoretical data (%H) obtained for combination of imatinib and vitamin D analogues and experimental data, it was observed that synergic effect of co-operation of analogues with imatinib, especially analogue PRI-2191 was maintained until the last measurement da of experiment. Hypothetical value TGI (%H) for GV+PRI-2191 combination is by 12% lower than experimental TGI, and for GC+PRI-22Q5 combination is by 5% lower. For better presentation of findings in column 6 of Table 1, tumour volume was presented in particular groups on day 31 of the experiment as a percent of control tumours. Tumour volume in groups treated with imatinib and vitamin D analogues PR1- 2191 and PRI-2205 combination is slightly more than 1/3 of the volume of control tumours (38% for GV+PR1-21 39% for GV+PRI-2205).
• Body wieght changes of mice during treatment and total blood calcium level in mice after the end of treatment.
Another parameter of treatment effectiveness evaluation was observation of mice body weight. Results are shown in Fig. 2.
On the basis of Fig. 2 which illustrates body weight changes of mice during experiment [shown in %], where body weight on a given day was compared to body weight on day "0" it may be concluded that administration of vitami D analogues is safe for mice. The observed decreases of body mass by more tha 5% for both groups receiving the combination of imatmib and vitamin D analogue PRI-2191 or PRI-2205 was probably caused by everyday administration of imatinib (on days from 7 to 21). After day 22 of the experiment the body mass decrease in both groups was about 10%, and after day 28 about 6-8%.
The observed changes of body mass are not caused by oalcaemic toxicity which can be expected in case of vitamin D analogues. It is confirmed by the obtained results of measurement of serum calcium level in mice. On day 34 the animals were sacrificed, blood was drawn and serum calcium level analysis was carried out (Fig. 3). None of both vitamin D analogues used in this experiment did not raise serum calcium level above the level determined in the control mice, considered normal for mice with cancer. Mice receiving vitamin D analogues or imatinib independently showed slightly lower serum calcium level than the control mice. Even in mice in groups receiving combination PRI-2191 or PRI-2205 with imatinib, calcium level was similar to that of control group and comparable to the level in mice receiving each drug independently, what suggests safety of use of combination of imatinib with vitamin D analogues.
Conclusions:
Imatinib in combination with vitamin D analogues decreases in statistically significant manner A549 tumour volume compared to control staring from day 12 to day 31 for combination of GV+PRI-2205 and on days 12, 14, 19-31 for combination GV+PRI-2191 (Mann-Whitney U test). According to analysis carried out with Kruskal- Wallis ANOVA, a more beneficial combination is imatinib with PRI-21 1 (statistically significant differences on days 12, 14, 19-31).
Imatinib only, similarly as vitamin D analogues: PRI2191 or PRI-22G5, decreases tumour volume, but differences between tumour volume in groups treated with each preparations independently are statistically significant only on selected days or periods of the experiment (Fig. 1. Mann-Whitney U test).
Synergic action of the tested combination of compounds was evaluated with consideration of parameters of tumour growth inhibition (TGI). The use of combination of vitamin D analogues with imatinib inhibits tumour growth in additive manner turning into synergism on further days of experiments and lasting to the end of experiment (Table 1, day 31), especially in the case of analogue PRI-2191, what suggests beneficial use of imatinib-tacalcitol. The use of analogues PRI-2191 and PRI-2205 administered to mice independently and in combination with imatinib do not raise serum calcium level above normal, what ensures safety of the combination use.
Based on the results of studies on murine model in vivo it may be concluded that vitamin D analogues enhance beneficial antineoplastic action of imatinib, therefore they may be used in combination therapy of non-small cell lung cancer.
Claims
The use of vitamin D analogues selected from tacalcitol and (1 S,3R,5E JE,22E,24S -24-eyclo^^
1,3,24-triol (5,6-trans-calcipotriol) in combination with tyrosine kinase inhibitor imatinib for treatment of non-small cell lung cancer.
The use according to claim 1 wherein vitamin D analogue is tacalcitol.
The use according to claim 1 or 2 wherein imatinib and vitamin D analogue are used as active substances in one or in two separate unit doses.
The use according to an of claims 1 - 3 wherein imatinib and vitamin D analogue are used as active substances in two separate unit dose forms.
The use according to any of claims 1 - 4 wherein unit dose forms contain therapeutically effective doses of imatinib and vitamin D analogues.
The use according to any of claims 1 - 4 wherein imatinib is in a unit dose form intended for administration by oral or intravenous routes and vitamin D analogue is in a unit dose form intended for oral or subcutaneous route.
The use according to any of claims 1—5 wherein therapeutically effective daily dose of imatinib is 50 to 1000 mg of active substance corresponding to a free base.
The use according to any of claims 1 - 6 wherein therapeutically effective daily dose of vitamin D analogue is 0.1 to 200 μg, preferably 0.5 to 50 μ of the active substance corresponding to a free base.
The use according to claim 9 wherein vitamin D analogue is tacalcitol in the form of monohydrate.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PLP-397662 | 2011-12-30 | ||
| PL397662A PL226174B1 (en) | 2011-12-30 | 2011-12-30 | Combination therapy of non-small-cell lung carcinoma |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013100772A1 true WO2013100772A1 (en) | 2013-07-04 |
Family
ID=47666456
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/PL2012/000134 Ceased WO2013100772A1 (en) | 2011-12-30 | 2012-12-30 | The use of vitamin d analogues in combination with imatinib in therapy of non-small cell lung cancer |
Country Status (2)
| Country | Link |
|---|---|
| PL (1) | PL226174B1 (en) |
| WO (1) | WO2013100772A1 (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0564409A1 (en) | 1992-04-03 | 1993-10-06 | Ciba-Geigy Ag | Pyrimidin derivatives and process for their preparation |
| WO1999003854A1 (en) | 1997-07-18 | 1999-01-28 | Novartis Ag | Crystal modification of a n-phenyl-2-pyrimidineamine derivative, processes for its manufacture and its use |
| WO2004098612A2 (en) * | 2003-05-07 | 2004-11-18 | Ab Science | Calcitriol analogs of uses thereof |
| WO2005016872A1 (en) * | 2003-06-11 | 2005-02-24 | Novacea, Inc. | Treatment of lung cancer with active vitamin d compounds in combination with other treatments |
-
2011
- 2011-12-30 PL PL397662A patent/PL226174B1/en unknown
-
2012
- 2012-12-30 WO PCT/PL2012/000134 patent/WO2013100772A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0564409A1 (en) | 1992-04-03 | 1993-10-06 | Ciba-Geigy Ag | Pyrimidin derivatives and process for their preparation |
| WO1999003854A1 (en) | 1997-07-18 | 1999-01-28 | Novartis Ag | Crystal modification of a n-phenyl-2-pyrimidineamine derivative, processes for its manufacture and its use |
| WO2004098612A2 (en) * | 2003-05-07 | 2004-11-18 | Ab Science | Calcitriol analogs of uses thereof |
| WO2005016872A1 (en) * | 2003-06-11 | 2005-02-24 | Novacea, Inc. | Treatment of lung cancer with active vitamin d compounds in combination with other treatments |
Non-Patent Citations (24)
Also Published As
| Publication number | Publication date |
|---|---|
| PL397662A1 (en) | 2013-07-08 |
| PL226174B1 (en) | 2017-06-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DK2786754T3 (en) | Combination therapy with a mitotic inhibitor | |
| US20230181565A1 (en) | Crenolanib combination therapy | |
| ES2741399T3 (en) | Apilimod for use in the treatment of renal cancer | |
| CA2960989A1 (en) | Human dosing of phosphatase inhibitor | |
| US20160158253A1 (en) | Treatment of pancreatic cancer with a combination of a hypoxia-activated prodrug and a taxane | |
| US10765682B2 (en) | Apilimod for use in the treatment of melanoma | |
| EP2892535B1 (en) | Method of adjuvant cancer treatment | |
| US20200352937A1 (en) | Combination comprising at least one spliceosome modulator and at least one inhibitor chosen from bcl2 inhibitors, bcl2/bclxl inhibitors, and bclxl inhibitors and methods of use | |
| KR20230113594A (en) | Combination therapy to treat PIK3CA mutant cancer | |
| Greystoke et al. | A phase I study of intravenous TZT-1027 administered on day 1 and day 8 of a three-weekly cycle in combination with carboplatin given on day 1 alone in patients with advanced solid tumours | |
| US20150297623A1 (en) | Methods for treatment of primary cancer and cancer metastasis | |
| WO2013100772A1 (en) | The use of vitamin d analogues in combination with imatinib in therapy of non-small cell lung cancer | |
| US20150110864A1 (en) | Novel antitumor agent comprising combination of three agents | |
| US10238679B2 (en) | Antitumor activity of multi-kinase inhibitors in colorectal cancer | |
| JP6243850B2 (en) | Prevention, treatment or alleviation of peripheral neuropathy with anticancer drugs | |
| CA2894153A1 (en) | Combinations of a pi3k/akt inhibitor compound with an her3/egfr inhibitor compound and use thereof in the treatment of a hyperproliferative disorder | |
| WO2025232723A1 (en) | Treatment method with focal adhesion kinase inhibitor | |
| WO2024088193A1 (en) | Combination of aurora a and parp inhibitors for treatment of cancers | |
| US20150374702A1 (en) | Anticancer adjuvant containing pentoxifylline | |
| HK40070843A (en) | Dabrafenib and trametinib in a method of adjuvant cancer treatment | |
| TW202126298A (en) | Combination for cancer treatment and uses of the same | |
| Greystoke et al. | combination with carboplatin given on day 1 alone in patients with advanced solid tumours | |
| HK1240937A1 (en) | Small molecules for the treatment of primary cancer and cancer metastasis | |
| HK1206642B (en) | Method of adjuvant cancer treatment | |
| OA16757A (en) | Compositions and methods for treating cancer using P13K beta inhibitor and MAPK pathway inhibitor, including MEK and RAF inhibitors. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12821331 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12821331 Country of ref document: EP Kind code of ref document: A1 |