WO2013087631A2 - Oxidation dye composition comprising a particular coupler in a medium rich in fatty substances, and processes and device suitable therefor - Google Patents
Oxidation dye composition comprising a particular coupler in a medium rich in fatty substances, and processes and device suitable therefor Download PDFInfo
- Publication number
- WO2013087631A2 WO2013087631A2 PCT/EP2012/075094 EP2012075094W WO2013087631A2 WO 2013087631 A2 WO2013087631 A2 WO 2013087631A2 EP 2012075094 W EP2012075094 W EP 2012075094W WO 2013087631 A2 WO2013087631 A2 WO 2013087631A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- composition
- dihydro
- chosen
- perimidine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*C(C)(*)[N+](*)C(C)(C)CO Chemical compound C*C(C)(*)[N+](*)C(C)(C)CO 0.000 description 1
- IJKZYGAHGUPSMK-UHFFFAOYSA-N CC(OC1)=CC(O)=C1C(N1)(Nc2c3c1cccc3ccc2)[Ne] Chemical compound CC(OC1)=CC(O)=C1C(N1)(Nc2c3c1cccc3ccc2)[Ne] IJKZYGAHGUPSMK-UHFFFAOYSA-N 0.000 description 1
- DWZZNCGUVUHLRM-UHFFFAOYSA-N COc1cccc(C(N2)Nc3c4c2cccc4ccc3)c1O Chemical compound COc1cccc(C(N2)Nc3c4c2cccc4ccc3)c1O DWZZNCGUVUHLRM-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/41—Amines
- A61K8/411—Aromatic amines, i.e. where the amino group is directly linked to the aromatic nucleus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/46—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing sulfur
- A61K8/466—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing sulfur containing sulfonic acid derivatives; Salts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4906—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom
- A61K8/4913—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom having five membered rings, e.g. pyrrolidone carboxylic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4906—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom
- A61K8/4926—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom having six membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/494—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4973—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom
- A61K8/498—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom having 6-membered rings or their condensed derivatives, e.g. coumarin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/69—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing fluorine
- A61K8/70—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing fluorine containing perfluoro groups, e.g. perfluoroethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/10—Preparations for permanently dyeing the hair
Definitions
- Oxidation dye composition comprising a particular coupler in a medium rich in fatty substances, and processes and device suitable therefor
- the present invention relates to a composition for the oxidation dyeing o f keratin fibres, in particular o f human keratin fibres such as the hair, comprising at least 25 % by weight, relative to the total weight of the composition, o f one or more fatty substances, one or more surfactants, one or more basifying agents, one or more oxidation bases and one or more particular couplers .
- the present invention also relates to dyeing processes using this composition in the presence o f at least one chemical oxidizing agent, and to a multi-compartment device suitable for using this composition.
- the present invention relates to the field o f the dyeing o f keratinous fibres and more particularly to the field of hair dyeing.
- the first type o f dyeing is "permanent" or oxidation dyeing, which uses dye compositions containing oxidation dye precursors, generally known as oxidation bases. These oxidation bases are co lourless or weakly coloured compounds, which, when combined with oxidizing products, may give rise to coloured compounds via a process of oxidative condensation.
- oxidation bases may be varied by combining them with couplers or co louration modifiers, the latter being chosen especially from aromatic meta- diamines, meta-aminopheno ls, meta-dipheno ls and certain heterocyclic compounds such as indo le compounds.
- couplers or co louration modifiers the latter being chosen especially from aromatic meta- diamines, meta-aminopheno ls, meta-dipheno ls and certain heterocyclic compounds such as indo le compounds.
- couplers or co louration modifiers the latter being chosen especially from aromatic meta- diamines, meta-aminopheno ls, meta-dipheno ls and certain heterocyclic compounds such as indo le compounds.
- the second type o f dyeing is " semi-permanent" dyeing or direct dyeing, which consists in applying direct dyes to the keratin fibres, the said dyes being coloured and colouring mo lecules that have affinity for the fibres, and then leaving them to take, to allow the mo lecules to penetrate by diffusion to the interior o f the fibre, and then rinsing the fibres .
- the direct dyes generally used are chosen from nitrobenzene, anthraquinone, nitropyridine, azo , xanthene, acridine, azine and triarylmethane direct dyes .
- one subj ect o f which is especially a composition for the oxidation dyeing keratin fibres, in particular o f human keratin fibres such as the hair, comprising :
- p denotes 0 or 1
- Ri, R 2 , R 3 , R 4 , R 5 , R 6 , R7 and Rs denote, independently of each other, a hydrogen atom; an amino group; a halogen atom; a hydroxyl group; a Ci-Cs alkoxy group; a C 2 -C 5 acylamino group; a group CHO, a linear, branched or cyclic Ci-Cis alkyl or C 2 -Cis alkylene group, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen, an aryl group, the alkyl, alkylene or aryl groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic acid, Ci-Cis alkyl ester, carbamate, carboxamide, amino, alkylamino, dialkylamino, carboxyl, alkoxy and halogen atoms, C 3 -C 6 cycloal
- the groups R 7 and Rs may form, with the atom that bears them, a ring optionally comprising one or more heteroatoms, this ring possibly being substituted with a Ci-C 6 alkyl group or a hydroxyl group, or possibly being fused with one or more other rings optionally bearing one or more heteroatoms, one or more carbon atoms of the ring(s) possibly being replaced with one or more carbonyl groups.
- R 7 may also denote a radical derived from a perimidinyl group of formula (I), thus leading to a dimer; the two perimidinyl groups are then preferably linked via the carbon atoms in position 2,
- R 7 may denote a linear or branched C1-C10 alkylene chain, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen and/or optionally comprising a ring, or alternatively a saturated or unsaturated ring, optionally comprising one or more heteroatoms; a chain comprising several and preferably two saturated or unsaturated rings linked together via a covalent bond or via a heteroatom, the chains or the ring possibly being substituted with a hydroxyl group or with a perimidinyl radical o f formula (I) preferably via the carbon atom in position 2,
- R 4 when R 4 is in position 7 and R 5 is in position 6, then they can form, together with the carbon atoms that bear them, a 5 - to 8- membered ring optionally comprising one or more heteroatoms .
- the present invention also relates to dyeing processes using this composition in the presence o f at least one chemical oxidizing agent, and to a multi-compartment device suitable for using this composition.
- the use o f the composition according to the invention in the presence o f at least one chemical oxidizing agent leads to strong co lorations with little selectivity. Furthermore, the co lorations obtained remain stable, in particular with respect to light.
- the human keratinous fibres treated by the method according to the invention are preferably the hair.
- composition according to the invention comprises one or more fatty substances .
- fatty substance means an organic compound that is inso luble in water at ordinary room temperature (25 °C) and at atmospheric pressure (760 mmHg) (solubility o f less than 5 %, preferably 1 % and even more preferentially 0. 1 %) . They exhibit, in their structure, at least one hydrocarbon chain comprising at least 6 carbon atoms or a sequence o f at least two siloxane groups.
- the fatty substances are generally so luble in organic so lvents under the same temperature and pressure conditions, for instance chloroform, ethano l, benzene, liquid petrolatum or decamethylcyclopentasiloxane. These fatty substances are neither polyoxyethylenated nor polyglycerolated.
- the fatty substances are chosen from C 6 - C i 6 hydrocarbons, hydrocarbons containing more than 16 carbon atoms, non-silicone oils o f animal origin, plant oils o f triglyceride type, synthetic triglycerides, fluoro oils, fatty alcoho ls, esters of fatty acids and/or of fatty alcoho ls other than triglycerides, and plant waxes, non- silicone waxes and silicones .
- the fatty alcoho ls, fatty esters and fatty acids more particularly contain one or more linear or branched, saturated or unsaturated hydrocarbon-based groups comprising 6 to 30 carbon atoms, which are optionally substituted, in particular with one or more (in particular 1 to 4) hydroxyl groups. If they are unsaturated, these compounds may comprise one to three conjugated or unconjugated carbon-carbon double bonds .
- C 6 - C i 6 hydrocarbons they are linear or branched, and possibly cyclic, and are preferably alkanes. Examp les that may be mentioned include hexane, dodecane and isoparaffins such as isohexadecane and isodecane.
- the triglyceride oils o f p lant or synthetic origin are preferably chosen from liquid fatty acid triglycerides containing from 6 to 30 carbon atoms, for instance heptanoic or octanoic acid triglycerides, or alternatively, for examp le, sunflower oil, corn oil, soybean oil, marrow oil, grapeseed oil, sesame seed oil, hazelnut oil, apricot oil, macadamia oil, arara oil, castor oil, avocado oil, caprylic/capric acid triglycerides, for instance those so ld by the company Stearineries Dubois or those so ld under the names Miglyo l® 8 1 0, 8 12 and 8 1 8 by the company Dynamit Nobel, jojoba oil and shea butter oil.
- liquid fatty acid triglycerides containing from 6 to 30 carbon atoms for instance heptanoic or octanoic acid triglycerides, or
- the linear or branched hydrocarbons o f mineral or synthetic origin containing more than 16 carbon atoms are preferably chosen from liquid paraffins, petroleum j elly, liquid petrolatum, polydecenes, and hydrogenated polyisobutene such as Parleam® .
- the fluoro oils may be chosen from perfluoromethylcyclopentane and perfluoro- 1 ,3 -dimethylcyclohexane, so ld under the names Flutec ® PC I and Flutec ® PC3 by the company BNFL Fluorochemicals; perfluoro- 1 ,2-dimethylcyclobutane; perfluoroalkanes such as dodecafluoropentane and tetradecafluorohexane, so ld under the names PF 5050 ® and PF 5060 ® by the company 3M, or bromoperfluorooctyl so ld under the name Foralkyl ® by the company Atochem; nonafluoromethoxybutane and nonafluoroethoxyisobutane; perfluoromorpholine derivatives such as 4-trifluoromethyl perfluoromorpholine sold under the name PF 5052 ® by the company 3M .
- the fatty alcoho ls that may be used in the composition according to the invention are saturated or unsaturated, and linear or branched, and comprise from 6 to 30 carbon atoms and more particularly from 8 to 30 carbon atoms.
- Examples that may be mentioned include cetyl alcoho l, stearyl alcoho l and the mixture thereo f (cetylstearyl alcoho l), octyldodecanol, 2-butyloctanol, 2- hexyldecanol, 2-undecylpentadecanol, oleyl alcoho l and lino leyl alcoho l.
- the wax(es) that may be used in the composition are chosen especially from carnauba wax, candelilla wax, esparto grass wax, paraffin wax, ozokerite, plant waxes, for instance olive wax, rice wax, hydrogenated jojoba wax or the absolute waxes o f flowers such as the essential wax o f blackcurrant blo ssom sold by the company Bertin (France), animal waxes, for instance beeswaxes, or modified beeswaxes (cerabellina); other waxes or waxy starting materials that may be used according to the invention are especially marine waxes such as the product sold by the company Sophim under the reference M82, and polyethylene waxes or polyo lefin waxes in general.
- the fatty acids that may be used in the composition may be saturated or unsaturated and contain from 6 to 30 carbon atoms and in particular from 9 to 30 carbon atoms. They are more particularly chosen from myristic acid, palmitic acid, stearic acid, behenic acid, oleic acid, linoleic acid, linolenic acid and isostearic acid.
- esters o f a fatty acid and/or of a fatty alcoho l, which are advantageously different from the triglycerides mentioned above, mention may be made especially o f esters o f saturated or unsaturated, linear or branched C i -C 26 aliphatic mono- or polyacids and o f saturated or unsaturated, linear or branched C i -C 26 aliphatic mono- or polyalcoho ls, the total carbon number o f the esters more particularly being greater than or equal to 1 0.
- esters of C 4 -C 22 dicarboxylic or tricarboxylic acids and of C i -C 22 alcoho ls and esters of monocarboxylic, dicarboxylic or tricarboxylic acids and o f C 2 -C 26 dihydroxy, trihydroxy, tetrahydroxy or pentahydroxy alcoho ls may also be used.
- esters mentioned above it is preferred to use ethyl, isopropyl, myristyl, cetyl or stearyl palmitate, 2-ethylhexyl palmitate,
- 2-octyldecyl palmitate alkyl myristates such as isopropyl, butyl, cetyl or 2-octyldodecyl myristate, hexyl stearate, butyl stearate, isobutyl stearate; dio ctyl malate, hexyl laurate, 2-hexyldecyl laurate, isononyl isononanoate or cetyl octanoate.
- the composition may also comprise, as fatty ester, sugar esters and diesters of C6 - C30 and preferably C 12 - C22 fatty acids .
- sugar means oxygen-bearing hydrocarbon-based compounds bearing several alcoho l functions, with or without aldehyde or ketone functions, and which comprise at least 4 carbon atoms. These sugars can be monosaccharides, oligosaccharides or polysaccharides .
- sugars for example, o f sucrose (or saccharose), glucose, galactose, ribose, fucose, maltose, fructose, mannose, arabino se, xylose, lactose and their derivatives, in particular alkyl derivatives, such as methyl derivatives, for example methylglucose.
- esters o f sugars and o f fatty acids can be chosen in particular from the group consisting of the esters or mixtures of esters of sugars described above and of saturated or unsaturated and linear or branched C6 - C30 and preferably C 12 - C22 fatty acids. If they are unsaturated, these compounds may comprise one to three conjugated or unconjugated carbon-carbon double bonds .
- esters according to this alternative form can also be chosen from mono-, di-, tri- and tetraesters and polyesters, and mixtures thereof. These esters can, for example, be o leates, laurates, palmitates, myristates, behenates, cocoates, stearates, lino leates, lino lenates, caprates, arachidonates or their mixtures, such as, in particular, oleate/palmitate, oleate/stearate or palmitate/stearate mixed esters .
- Examples o f esters or mixtures o f esters of sugar and o f fatty acid that may also be mentioned include :
- Ryoto Sugar Esters for example referenced B370 and corresponding to sucrose behenate formed of 20% monoester and 80% diester, triester and polyester;
- the silicones that can be used in the composition of the present invention are vo latile or non- vo latile, cyclic, linear or branched silicones, which are unmodified or mo dified with organic groups, having a viscosity from 5 x 10 "6 to 2.5 m 2 /s at 25 °C, and preferably l x l O "5 to 1 m 2 /s .
- the silicones which can be used in accordance with the invention can be provided in the form of oils, waxes, resins or gums .
- the silicone is chosen from polydialkylsiloxanes, in particular polydimethylsiloxanes (PDMSs), and organomodified polysiloxanes comprising at least one functional group chosen from poly(oxyalkylene) groups, amino groups and alkoxy groups.
- PDMSs polydimethylsiloxanes
- organomodified polysiloxanes comprising at least one functional group chosen from poly(oxyalkylene) groups, amino groups and alkoxy groups.
- Organopolysiloxanes are defined in greater detail in Walter Noll's Chemistry and Technology of Silicones (1968), Academic Press. They may be volatile or non-volatile.
- the silicones are more particularly chosen from those with a boiling point of between 60°C and 260°C, and even more particularly from:
- cyclic polydialkylsiloxanes containing from 3 to 7 and preferably from 4 to 5 silicon atoms. They are, for example, octamethylcyclotetrasiloxane, sold in particular under the name Volatile Silicone® 7207 by Union Carbide or Silbione® 70045 V2 by Rhodia, decamethylcyclopentasiloxane, sold under the name Volatile Silicone® 7158 by Union Carbide and Silbione® 70045 V5 by Rhodia, and mixtures thereof.
- linear volatile polydialkylsiloxanes containing 2 to 9 silicon atoms and having a viscosity of less than or equal to 5 x 10 "6 m 2 /s at 25°C.
- An example is decamethyltetrasiloxane sold in particular under the name SH 200 by the company Toray Silicone. Silicones belonging to this category are also described in the article published in Cosmetics and Toiletries, Vol. 91, Jan. 76, pp. 27-32, Todd & Byers, Volatile Silicone Fluids for Cosmetics .
- Use is preferably made of non-volatile polydialkylsiloxanes, polydialkylsiloxane gums and resins, polyorganosiloxanes modified with the organofunctional groups above, and mixtures thereof.
- silicones are more particularly chosen from polydialkylsiloxanes, among which mention may be made mainly of polydimethylsiloxanes having trimethylsilyl end groups.
- the viscosity of the silicones is measured at 25°C according to ASTM standard 445 Appendix C.
- oils of the 200 series from Dow Corning such as DC200 having a viscosity of 60000 mm 2 /s;
- CTFA dimethiconol
- the silicone gums which can be used in accordance with the invention are in particular polydialkylsiloxanes and preferably polydimethylsiloxanes having high number-average molecular weights of between 200000 and 1 000000, used alone or as a mixture in a solvent.
- This solvent can be chosen from volatile silicones, polydimethylsiloxane (PDMS) oils, polyphenylmethylsiloxane (PPMS) oils, isoparaffins, polyisobutylenes, methylene chloride, pentane, dodecane, tridecane or their mixtures .
- Products which can be used more particularly in accordance with the invention are mixtures, such as :
- CTFA dimethicono l
- CFA cyclic polydimethylsiloxane
- this product is an SF 30 gum corresponding to a dimethicone, having a number-average mo lecular weight o f 500 000, dissolved in the oil SF 1202 Silicone Fluid corresponding to decamethy Icy clop entasiloxane;
- the product SF 1236 is the mixture of a gum SE 30 defined above having a viscosity o f 20 m 2 /s and of an oil
- This product preferably comprises 15 % of gum SE 30 and 85 % of an oil SF 96.
- organopolysiloxane resins that can be used in accordance with the invention are crosslinked siloxane systems containing the fo llowing units :
- R represents an alkyl containing 1 to 16 carbon atoms.
- R denotes a C i -C 4 lower alkyl group, more particularly methyl.
- Silicone Fluid S S 4230 and S S 4267 by General Electric which are silicones of dimethyl/trimethylsiloxane structure.
- organomodified silicones that can be used in accordance with the invention are silicones as defined above and comprising in their structure one or more organofunctional groups attached via a hydrocarbon-based group.
- the organomodified silicones can be polydiarylsiloxanes, in particular polydiphenylsiloxanes, and polyalkylarylsiloxanes functionalized by the abovementioned organofunctional groups.
- the polyalkylarylsiloxanes are chosen particularly from linear and/or branched polydimethyl/methylphenylsiloxanes and polydimethyl/diphenylsiloxanes with a viscosity of from lxlO "5 to 5xl0 "2 m 2 /s at 25°C.
- oils of the SF series from General Electric such as SF 1023, SF 1154, SF 1250 and SF 1265.
- organomodified silicones of polyorganosiloxanes comprising:
- polyethyleneoxy and/or polypropyleneoxy groups optionally comprising C6-C 2 4 alkyl groups, such as the products named dimethicone copolyol sold by Dow Corning under the name DC 1248 or the oils Silwet ® L 722, L 7500, L 77 and L 711 by Union Carbide, and the (Ci 2 )alkyl methicone copolyol sold by Dow Corning under the name Q25200;
- substituted or unsubstituted amino groups such as the products sold under the names GP 4 Silicone Fluid and GP 7100 by Genesee or the products sold under the names Q2 8220 and Dow Corning 929 or 939 by Dow Corning.
- the substituted amino groups are in particular C 1 - C 4 aminoalkyl groups;
- the fatty substances are chosen from compounds that are liquid or pasty at room temperature and at atmospheric pressure.
- the fatty substance is a compound which is liquid at the temperature of 25 °C and at atmospheric pressure.
- the fatty substances are advantageously chosen from C 6 - C i 6 hydrocarbons, hydrocarbons containing more than 16 carbon atoms, non-silicone oils o f animal origin, plant oils o f triglyceride type, synthetic triglycerides, fluoro oils, fatty alcoho ls, esters of fatty acids and/or of fatty alcoho ls other than triglycerides, and plant waxes, non- silicone waxes and silicones, and a mixture thereof.
- the fatty substance(s) are chosen from liquid petrolatum, polydecenes, fatty alcoho ls and liquid esters o f a fatty acid and/or of a fatty alcohol, or mixtures thereof.
- the fatty substances are chosen from liquid petrolatum and octyldodecanol.
- the composition according to the invention has a fatty substance content of at least 25 % by weight. More particularly, the fatty substance concentration ranges from 25 % to 80% by weight, preferably from 30% to 70% by weight and even more advantageously from 30% to 60% by weight relative to the total weight o f the composition.
- composition according to the invention also comprises one or more surfactants .
- the surfactant(s) are chosen from anionic, amphoteric, zwitterionic, cationic and nonionic surfactants, and preferentially nonionic surfactants .
- anionic surfactant means a surfactant comprising, as ionic or ionizable groups, only anionic groups.
- alkyl sulfates alkyl ether sulfates, alkylamido ether sulfates, alkylaryl polyether sulfates, monoglyceride sulfates, alkylsulfonates, alkylamidesulfonates, alkylarylsulfonates, a-o lefin sulfonates, paraffin sulfonates, alkyl sulfo succinates, alkyl ether sulfo succinates, alkylamide sulfo succinates, alkyl sulfoacetates, acylsarcosinates, acylglutamates, alkyl sulfosuccinamates, acylisethionates and N- acyltaurates, polyglycoside polycarboxylic acid and alkyl monoester
- These compounds can be oxyethylenated and then preferably comprise from 1 to 50 ethylene oxide units.
- the salts o f C 6 -C 2 4 alkyl monoesters o f polyglycoside- polycarboxylic acids can be chosen from C 6 -C 2 4 alkyl polyglycoside- citrates, C 6 -C 2 4 alkyl polyglycoside-tartrates and C 6 -C 2 4 alkyl poly glycoside-sulfo succinates .
- the anionic surfactant(s) When the anionic surfactant(s) are in salt form, it (they) may be chosen from alkali metal salts such as the sodium or potassium salt and preferably the sodium salt, ammonium salts, amine salts and in particular amino alcoho l salts, or alkaline earth metal salts such as the magnesium salts .
- alkali metal salts such as the sodium or potassium salt and preferably the sodium salt, ammonium salts, amine salts and in particular amino alcoho l salts, or alkaline earth metal salts such as the magnesium salts .
- alkali metal salts such as the sodium or potassium salt and preferably the sodium salt, ammonium salts, amine salts and in particular amino alcoho l salts, or alkaline earth metal salts such as the magnesium salts .
- amino alcohol salts that may especially be mentioned include monoethanolamine, diethanolamine and triethanolamine salts, monoisopropanolamine, diisopropano
- Use is preferably made of alkali metal or alkaline earth metal salts and in particular of sodium or magnesium salts.
- Use is preferably made, among the anionic surfactants mentioned, of (C 6 -C 2 4)alkyl sulfates, (C 6 -C 2 4)alkyl ether sulfates comprising from 2 to 50 ethylene oxide units, in particular in the form of alkali metal, ammonium, aminoalcohol and alkaline earth metal salts, or a mixture of these compounds.
- (Ci 2 -C 2 o)alkyl sulfates it is preferable to use (Ci 2 -C 2 o)alkyl sulfates, (Ci 2 -C 2 o)alkyl ether sulfates comprising from 2 to 20 ethylene oxide units, in particular in the form of alkali metal, ammonium, aminoalcohol and alkaline earth metal salts, or a mixture of these compounds.
- sodium lauryl ether sulfate comprising 2.2 mol of ethylene oxide.
- amphoteric or zwitterionic surfactant(s), which are preferably (a) non-silicone surfactant(s), which can be used in the present invention can in particular be derivatives of secondary or tertiary, optionally quaternized aliphatic amines, in which derivatives the aliphatic group is a linear or branched chain comprising from 8 to 22 carbon atoms, said amine derivatives comprising at least one anionic group, such as, for example, a carboxylate, sulfonate, sulfate, phosphate or phosphonate group.
- Ra represents a C 1 0 - C30 alkyl or alkenyl group derived from an acid RaCOOH preferably present in hydrolysed coconut oil, a heptyl, nonyl group or undecyl group;
- Rb represents a ⁇ -hydroxyethyl group
- Rc represents a carboxymethyl group
- M + represents a cationic counterion derived from an alkali metal or alkaline-earth metal, such as sodium, an ammonium ion or an ion derived from an organic amine, and
- X " represents an organic or inorganic anionic counterion, such as that chosen from halides, acetates, phosphates, nitrates, (C i - C 4 )alkyl sulfates, (C i -C 4 )alkyl- or (C i -C 4 )alkylarylsulfonates, in particular methyl sulfate and ethyl sulfate; or alternatively M + and X " are absent;
- B represents the group -CH 2 -CH 2 -0-X'
- X * represents the group -CH 2 -C(0)OH, -CH 2 -C(0)OZ ' , -CH 2 - CH 2 -C(0)OH or -CH 2 -CH 2 -C(0)OZ ' , or a hydrogen atom;
- Y * represents the group -C(0)OH, -C(0)OZ ' , -CH 2 -CH(OH)- S O 3 H or the group -CH 2 -CH(OH)-S0 3 -Z ' ;
- Z' represents a cationic counterion derived from an alkali metal or alkaline-earth metal, such as sodium, an ammonium ion or an ion derived from an organic amine;
- Ra' represents a C 1 0 - C30 alkyl or alkenyl group of an acid Ra' - C(0)OH which is preferably present in coconut oil or in hydrolysed linseed oil, or an alkyl group, especially a C 1 7 group , and its iso form, or an unsaturated C 1 7 group .
- amphoteric or zwitterionic surfactants use is preferably made o f (C 8 - C2o)alkylbetaines such as cocobetaine, and (C 8 - C2o)alkylamido(C 3 -C 8 )alkylbetaines such as cocamidopropylbetaine, and mixtures thereof. More preferably, the amphoteric or zwitterionic surfactant(s) are chosen from cocamidopropyl betaine and coco betaine.
- the cationic surfactant(s) which can be used in the composition according to the invention comprise, for example, salts of primary, secondary or tertiary fatty amines, which are optionally polyoxyalkylenated, quaternary ammonium salts and mixtures thereof.
- quaternary ammonium salts examples include :
- Rs to Rn which may be identical or different, represent a linear or branched aliphatic group comprising from 1 to 30 carbon atoms, or an aromatic group such as aryl or alkylaryl, it being understood that at least one o f the groups Rs to Rn comprises from 8 to 30 carbon atoms and preferably from 12 to 24 carbon atoms; and
- X " represents an organic or inorganic anionic counterion, such as that chosen from halides, acetates, phosphates, nitrates, (C i - C 4 )alkyl sulfates, (C i -C 4 )alkyl or (C i -C 4 )alkylaryl sulfonates, in particular methyl sulfate and ethyl sulfate .
- the aliphatic groups o f Rs to Rn may also comprise heteroatoms especially such as oxygen, nitrogen, sulfur and halogens .
- the aliphatic groups of Rs to Rn are chosen, for example, from C1-C30 alkyl, C1-C30 alkoxy, polyoxy(C2-Ce)alkylene, C1-C30 alkylamide, (Ci2-C22)alkylamido(C2-C6)alkyl, (Ci2-C22)alkylacetate, C1-C30 hydroxyalkyl, X " is an anionic counterion chosen from halides, phosphates, acetates, lactates, (Ci-C4)alkyl sulfates, and (Ci-C4)alkyl- or (Ci-C4)alkylarylsulfonates.
- quaternary ammonium salts of formula (A3) preference is given firstly to tetraalkylammonium chlorides, for instance dialkyldimethylammonium or alkyltrimethylammonium chlorides in which the alkyl group contains approximately from 12 to 22 carbon atoms, in particular behenyltrimethylammonium chloride, distearyldimethylammonium chloride, cetyltrimethylammonium chloride, benzyldimethylstearylammonium chloride, or else, secondly, distearoylethylhydroxyethylmethylammonium met ho sulfate, dipalmitoylethylhydroxyethylammonium methosulfate or distearoylethylhydroxyethylammonium methosulfate, or else, lastly, palmitylamidopropyltrimethylammonium chloride or stearamidopropyldimethyl(myristyl acetate)ammonium
- R12 represents an alkenyl or alkyl group comprising from 8 to 30 carbon atoms, for example tallow fatty acid derivatives
- R represents a hydrogen atom, a C1-C4 alkyl group or an alkenyl or alkyl group comprising from 8 to 30 carbon atoms;
- Ri4 represents a C1-C4 alkyl group
- Ri5 represents a hydrogen atom or a C1-C4 alkyl group
- X " represents an organic or inorganic anionic counterion, such as that chosen from halides, phosphates, acetates, lactates, (Ci- C 4 )alkyl sulfates, (Ci-C 4 )alkyl or (Ci-C 4 )alkylaryl sulfonates.
- Ri2 and R13 preferably denote a mixture of alkenyl or alkyl groups comprising from 12 to 21 carbon atoms, for example tallow fatty acid derivatives, Ri 4 denotes a methyl group, and R15 denotes a hydrogen atom.
- a product is sold, for example, under the name Rewoquat® W 75 by the company Rewo;
- Ri6 denotes an alkyl group comprising approximately from 16 to 30 carbon atoms, which is optionally hydroxylated and/or interrupted by one or more oxygen atoms;
- Ri7 is chosen from hydrogen, an alkyl group comprising from 1 to 4 carbon atoms or a group -(CH 2 )3-N + (Ri6a)(Ri7a)(Ri8a), X " ;
- Ri6a, Ri7a, Ri8a, Ri8, Ri9, R 2 o and R 2 1 which may be identical or different, are chosen from hydrogen and an alkyl group comprising from 1 to 4 carbon atoms;
- X " which may be identical or different, represents an organic or inorganic anionic counterion, such as that chosen from halides, acetates, phosphates, nitrates, (Ci-C 4 )alkyl sulfates, (Ci-C 4 )alkyl or (Ci-C 4 )alkylaryl sulfonates, in particular methyl sulfate and ethyl sulfate.
- organic or inorganic anionic counterion such as that chosen from halides, acetates, phosphates, nitrates, (Ci-C 4 )alkyl sulfates, (Ci-C 4 )alkyl or (Ci-C 4 )alkylaryl sulfonates, in particular methyl sulfate and ethyl sulfate.
- Such compounds are, for example, Finquat CT-P, provided by Finetex (Quaternium 89), or Finquat CT, provided by Finetex (Quaternium 75);
- R22 is chosen from Ci-C 6 alkyl groups and Ci-C 6 hydroxyalkyl or dihydroxyalkyl groups,
- R23 is chosen from:
- R25 is chosen from:
- R24, R26 and R28 which may be identical or different, are chosen from linear or branched, saturated or unsaturated C7-C 21 hydrocarbon-based groups;
- r, s and t which may be identical or different, are integers ranging from 2 to 6,
- y is an integer ranging from 1 to 10,
- x and z which may be identical or different, are integers ranging from 0 to 10,
- X " represents an organic or inorganic anionic counterion, with the proviso that the sum x + y + z is from 1 to 15, that when x is 0, then R23 denotes R27 and that when z is 0, then R25 denotes R29.
- the alkyl groups R22 may be linear or branched, and more particularly linear.
- R22 denotes a methyl, ethyl, hydroxyethyl or dihydroxypropyl group, and more particularly a methyl or ethyl group .
- the sum x + y + z ranges from 1 to 1 0.
- R23 is a hydrocarbon-based group R27 , it may be long and may contain from 12 to 22 carbon atoms, or may be short and may contain from 1 to 3 carbon atoms .
- R25 is a hydrocarbon-based group R29 , it preferably contains 1 to 3 carbon atoms .
- R24 , R26 and R28 which may be identical or different, are chosen from linear or branched, saturated or unsaturated C 1 1 -C 2 1 hydrocarbon-based groups, and more particularly from linear or branched, saturated or unsaturated C n -C 2 1 alkyl and alkenyl groups .
- x and z which may be identical or different, are preferably 0 or 1 .
- y is advantageously equal to 1 .
- r, s and t which may be identical or different, equal 2 or 3 , and even more particularly are equal to 2.
- the anionic counterion X " is preferably a halide, such as chloride, bromide or iodide; a (C i - C 4 )alkyl sulfate or a (C i - C 4 )alkylsulfonate or (C i -C 4 )alkylarylsulfonate.
- a halide such as chloride, bromide or iodide
- a (C i - C 4 )alkyl sulfate or a (C i - C 4 )alkylsulfonate or (C i -C 4 )alkylarylsulfonate it is possible to use methanesulfonate, phosphate, nitrate, tosylate, an anion derived from an organic acid, such as acetate or lactate, or any other anion that is compatible with the ammonium containing an ester function.
- the anionic counterion X " is even more particularly chloride, methyl sulfate or ethyl sulfate.
- R22 denotes a methyl or ethyl group
- - R25 is chosen from:
- R24 , R26 and R28 which may be identical or different, are chosen from linear or branched, saturated or unsaturated C 1 3 -C 1 7 hydrocarbon-based groups, and more particularly from linear or branched, saturated or unsaturated C 1 3 -C 1 7 alkyl and alkenyl groups .
- hydrocarbon radicals are linear.
- examples that may be mentioned include salts, especially the chloride or methyl sulfate, of diacyloxyethyldimethyl ammonium,
- the acyl groups preferably contain 14 to 1 8 carbon atoms and are obtained more particularly from a plant oil such as palm oil or sunflower oil. When the compound contains several acyl groups, these groups may be identical or different.
- This esterification is fo llowed by a quaternization by means of an alkylating agent such as an alkyl halide, preferably methyl or ethyl halide, a dialkyl sulfate, preferably dimethyl or diethyl sulfate, methyl methanesulfonate, methyl /?ara-toluenesulfonate, glycol chlorohydrin or glycerol chlorohydrin.
- an alkylating agent such as an alkyl halide, preferably methyl or ethyl halide, a dialkyl sulfate, preferably dimethyl or diethyl sulfate, methyl methanesulfonate, methyl /?ara-toluenesulfonate, glycol chlorohydrin or glycerol chlorohydrin.
- Such compounds are sold, for example, under the names Dehyquart® by the company Henkel, Stepanquat® by the company Stepan, Noxamium® by the company Ceca or Rewoquat® WE 18 by the company Rewo-Witco.
- composition according to the invention may contain, for example, a mixture of quaternary ammonium salts of monoesters, diesters and triesters with a weight majority of diester salts.
- ammonium salts containing at least one ester function that are described in patents US-A-4 874 554 and US-A-4 137 180.
- Use may be made of behenoylhydroxypropyltrimethylammonium chloride sold by KAO under the name Quatarmin BTC 131.
- the ammonium salts containing at least one ester function contain two ester functions.
- cationic surfactants that may be present in the composition according to the invention, it is more particularly preferred to choose cetyltrimethylammonium, behenyltrimethylammonium and dipalmitoylethylhydroxyethylmethyl- ammonium salts, and mixtures thereof, and more particularly behenyltrimethylammonium chloride, cetyltrimethylammonium chloride, and dipalmitoylethylhydroxyethylammonium methosulfate, and mixtures thereof.
- nonionic surfactants that can be used in the composition used according to the invention are described, for example, in the Handbook of Surfactants by M.R. Porter, published by Blackie & Son (Glasgow and London), 1991, pp. 116-178.
- They are especially chosen from alcohols, a-diols and (Ci-C2o)alkylphenols, these compounds being polyethoxylated, polypropoxylated or polyglycerolated, and containing at least one fatty chain comprising, for example, from 8 to 18 carbon atoms, it being possible for the number of ethylene oxide and/or propylene oxide groups to especially range from 2 to 50, and for the number o f glycerol groups to especially range from 2 to 30.
- the nonionic surfactants are chosen more particularly from mono- or polyoxyalkylenated or mono- or polyglycero lated nonionic surfactants.
- the oxyalkylene units are more particularly oxyethylene or oxypropylene units, or their combination, preferably oxyethylene units.
- Examples o f oxyalkylenated nonionic surfactants that may be mentioned include :
- esters of saturated or unsaturated and linear or branched C 8 - C30 acids and of polyethylene glycols are esters of saturated or unsaturated and linear or branched C 8 - C30 acids and of polyethylene glycols;
- the surfactants contain a number o f mo les o f ethylene oxide and/or of propylene oxide of between 1 and 100, preferably between 2 and 50 and preferably between 2 and 30.
- the nonionic surfactants do not comprise any oxypropylene units.
- the oxyalkylenated nonionic surfactants are chosen from oxyethylenated C8-C30 alcohols comprising from 1 to 100 mol of ethylene oxide; and polyoxyethylenated esters of saturated or unsaturated and linear or branched C8-C30 acids and of sorbitol comprising from 1 to 100 mol of ethylene oxide.
- monoglycerolated or polyglycerolated nonionic surfactants monoglycerolated or polyglycerolated nonionic surfactants.
- monoglycerolated or polyglycerolated nonionic surfactants monoglycerolated or polyglycerolated C8-C40 alcohols are preferably used.
- the monoglycerolated or polyglycerolated C8-C40 alcohols correspond to the formula (A7) below:
- R 2 9 represents a linear or branched C8-C40 and preferably C 8 - C30 alkyl or alkenyl radical
- m represents a number ranging from 1 to 30 and preferably from 1 to 10.
- lauryl alcohol containing 4 mol of glycerol (INCI name: Polyglyceryl-4 Lauryl Ether), lauryl alcohol comprising 1.5 mol of glycerol, oleyl alcohol comprising 4 mol of glycerol (INCI name: Polyglyceryl-4 Oleyl Ether), oleyl alcohol comprising 2 mol of glycerol (INCI name: Polyglyceryl-2 Oleyl Ether), cetearyl alcohol comprising 2 mol of glycerol, cetearyl alcohol comprising 6 mol of glycerol, oleocetyl alcohol comprising 6 mol of glycerol, and octadecanol comprising 6 mol of glycerol.
- the alcohol of formula (A8) may represent a mixture of alcohols in the same way that the value of m represents a statistical value, which means that, in a commercial product, several species of polyglycerolated fatty alcohols may coexist in the form of a mixture.
- Use is more preferably made, among the mono- or polyglycerolated alcohols, of the Cs/Cio alcohol comprising 1 mol of glycerol, the C 10 /C 12 alcohol comprising 1 mol of glycerol and the C 12 alcohol comprising 1.5 mol of glycerol.
- the surfactant used in the composition o f the invention is chosen from nonionic monooxyalkylenated or polyoxyalkylenated and monoglycerolated or polyglycero lated surfactants, more particularly monooxyethylenated or polyoxyethylenated, or monooxypropylenated or po lyoxypropylenated, nonionic surfactants, or a combination thereo f, more particularly monooxyethylenated or polyoxyethylenated.
- the surfactant(s) are chosen from nonionic surfactants or from anionic surfactants . More particularly, the surfactant(s) present in the composition are chosen from nonionic surfactants .
- the nonionic surfactants are chosen from polyoxyethylenated sorbito l esters and polyoxyethylenated fatty alcoho ls, and mixtures thereof.
- the amount of surfactant(s) in the composition preferably ranges from 0. 1 % to 50% by weight and better still from 0.5 % to 20% by weight relative to the total weight of the composition.
- composition according to the invention comprises one or more basifying agents .
- the basifying agent(s) may be mineral or organic or hybrid.
- the term "mineral compound” means any compound bearing in its structure one or more elements from co lumns 1 to 13 of the Periodic Table o f the Elements other than hydrogen.
- the mineral basifying agent contains one or more elements from columns 1 and 2 of the Perio dic Table of the Elements other than hydrogen.
- the mineral basifying agent has the fo llowing structure :
- Z 2 denotes a metal from columns 1 to 13 and preferably 1 or 2 of the Periodic Table of the Elements, such as sodium or potassium;
- Zi x denotes an anion chosen from the ions C0 3 2 , OH “ , HC0 3 2” , Si0 3 2” , HP0 4 2” , P0 4 3” and B 4 0 7 2" , and preferably from the ions C0 3 2 , OH " and Si0 3 2" ;
- x denotes 1, 2 or 3;
- y denotes 1, 2, 3 or 4;
- n and n denote, independently of each other, 1, 2, 3 or 4;
- the mineral basifying agent(s) are preferably chosen from aqueous ammonia, alkali carbonates or bicarbonates such as sodium carbonate, potassium carbonate, sodium bicarbonate, potassium bicarbonate, sodium hydroxide (soda), potassium hydroxide (potash), sodium metasilicate and potassium metasilicate, and a mixture thereof.
- the basifying agent is a mineral base chosen from alkali metal carbonates.
- the organic basifying agent(s) are preferably chosen from organic amines with a pK b at 25°C of less than 12, preferably less than 10 and even more advantageously less than 6. It should be noted that it is the pK b corresponding to the functional group of highest basicity.
- the organic amines do not comprise any alkyl or alkenyl fatty chains comprising more than ten carbon atoms.
- the organic basifying agent(s) are chosen, for example, from alkanolamines, oxyethylenated and/or oxypropylenated ethylenediamines, amino acids and the compounds of formula (III) below: Rx Rz
- W is a divalent Ci-C 6 alkylene residue optionally substituted with a hydroxyl group or a Ci-C 6 alkyl radical; and/or optionally interrupted with one or more heteroatoms such as O, or NRu; Rx, Ry, Rz, Rt and Ru, which may be identical or different, represent a hydrogen atom or a Ci-C 6 alkyl, Ci-C 6 hydroxyalkyl or Ci- C 6 aminoalkyl radical.
- Examples of amines of formula (II) that may be mentioned include 1 ,3-diaminopropane, 1 ,3-diamino-2-propanol, spermine and spermidine.
- alkanolamine means an organic amine comprising a primary, secondary or tertiary amine function, and one or more linear or branched Ci-Cs alkyl groups bearing one or more hydroxyl radicals.
- Organic amines chosen from alkanolamines, such as mono-, di- or trialkanolamines, comprising from one to three identical or different C 1 -C4 hydroxyalkyl radicals, are suitable in particular for the implementation of the invention.
- MAA monoethanolamine
- diethanolamine triethanolamine
- monoisopropanolamine diisopropanolamine
- N,N- dimethylethanolamine 2-amino-2-methyl- 1 -propanol
- triisopropanolamine 2 -amino -2 -methyl- 1 ,3 -propanediol
- 2- propanediol 3-dimethylamino- 1 ,2-propanediol
- tris(hydroxymethyl)amino methane tris(hydroxymethyl)amino methane.
- amino acids which can be used are of natural or synthetic origin, in their L, D or racemic form, and comprise at least one acid functional group chosen more particularly from carboxylic acid, sulfonic acid, phosphonic acid or phosphoric acid functional groups.
- the amino acids can be in the neutral or ionic form.
- amino acids which can be used in the present invention, of aspartic acid, glutamic acid, alanine, arginine, ornithine, citrulline, asparagine, carnitine, cysteine, glutamine, glycine, histidine, lysine, iso leucine, leucine, methionine, N-phenylalanine, proline, serine, taurine, threonine, tryptophan, tyrosine and valine.
- amino acids which can be used in the present invention, of aspartic acid, glutamic acid, alanine, arginine, ornithine, citrulline, asparagine, carnitine, cysteine, glutamine, glycine, histidine, lysine, iso leucine, leucine, methionine, N-phenylalanine, proline, serine, taurine, threonine, tryptophan, tyrosine and valine.
- the amino acids are basic amino acids comprising an additional amine functional group optionally included in a ring or in a ureido functional group .
- Such basic amino acids are preferably chosen from those corresponding to formula (IV) below :
- R represents a group chosen from:
- the compounds corresponding to formula (IV) are histidine, lysine, arginine, ornithine and citrulline.
- the organic amine may also be chosen from organic amines o f heterocyclic type. Besides histidine that has already been mentioned in the amino acids, mention may in particular be made o f pyridine, piperidine, imidazole, triazo le, tetrazole and benzimidazole.
- the organic amine may also be chosen from amino acid dipeptides.
- amino acid dipeptides that may be used in the present invention, mention may be made especially o f carnosine, anserine and balenine.
- the organic amine may also be chosen from compounds comprising a guanidine function.
- amines o f this type that can be used in the present invention, besides arginine, which has already been mentioned as an amino acid, mention may be made especially o f creatine, creatinine, 1 , 1 -dimethylguanidine, 1 , 1 -diethylguanidine, glycocyamine, metformin, agmatine, N-amidinoalanine, 3 - guanidinopropionic acid, 4-guanidinobutyric acid and 2- ([ amino (imino)methyl] amino) ethane- 1 - sulfonic acid.
- Hybrid compounds that may be mentioned include the salts o f the amines mentioned previously with acids such as carbonic acid or hydrochloric acid.
- Guanidine carbonate or monoethanolamine hydrochloride may be used in particular.
- the basifying agent(s) present in the composition of the invention are chosen from alkanolamines, amino acids in neutral or ionic form, in particular basic amino acids, and preferably corresponding to those of formula (IV), and compounds comprising a guanidine function, and mixtures thereof.
- the basifying agent(s) are chosen from alkano lamines such as monoethanolamine (MEA) .
- the composition according to the invention has a content of organic basifying agent(s) ranging from 0.01 % to 30% by weight and preferably from 0. 1 % to 20% by weight relative to the weight of the said composition.
- the composition according to the invention or else the process according to the invention does not use aqueous ammonia, or a salt thereo f, as basifying agent.
- composition or the process according to the invention does use aqueous ammonia, or a salt thereo f, as basifying agent, its content should not exceed 0.03 %> by weight (expressed as NH 3 ) and preferably should not exceed 0.01 % by weight, relative to the weight of the composition of the invention.
- the amount of basifying agent(s) other than the aqueous ammonia is greater than that of the aqueous ammonia (expressed as NH 3 ) .
- composition according to the invention comprises one or more oxidation bases .
- the oxidation bases may be chosen from heterocyclic bases and benzenic bases, and mixtures thereof.
- the oxidation bases are chosen from para- phenylene diamines, bis (phenyl) alky lene diamines, para-aminophenols, ortho-aminopheno ls and heterocyclic bases, and the addition salts thereo f.
- para-phenylenediamines examples that may be mentioned include para-phenylenediamine, para-tolylenediamine, 2- chloro-para-phenylenediamine, 2, 3 -dimethyl-para-pheny lene diamine, 2, 6 -dimethyl-para-pheny lene diamine, 2,6-diethyl-para- phenylenediamine, 2,5 -dimethyl-para-phenylenediamine, N,N- dimethyl-para-phenylenediamine, ⁇ , ⁇ -diethyl-para-pheny lene diamine, ⁇ , ⁇ -dipropyl-para-pheny lene diamine, 4 -amino -N,N- diethyl- 3 - methylaniline, N,N-bis(P -hydroxy ethyl) -para-phenylenediamine, 4- N,N-bis( -hydroxy ethyl) amino -2 -met hylaniline,
- para-phenylenediamine para-tolylenediamine, 2-isopropyl-para- phenylenediamine, 2- -hydroxyethyl-para-phenylenediamine, 2- ⁇ - hydroxyethyloxy-para-phenylenediamine, 2, 6 -dimethyl-par a- phenylenediamine, 2,6-diethyl-para-phenylenediamine, 2, 3 -dimethyl- par a-phenylenediamine, N, N-bis( - hydroxy ethyl) -par a- phenylenediamine, 2-chloro-para-phenylenediamine and 2- ⁇ - acetylaminoethyloxy-para-phenylenediamine, and the addition salts thereo f with an acid, are particularly preferred.
- bis(phenyl)alkylenediamines examples that may be mentioned include N,N'-bis( -hydroxyethyl)-N,N'-bis(4'- aminophenyl)- l ,3 -diaminopropanol, N, N'-bis( -hydroxy ethyl)-N, N'- bis(4'-aminophenyl)ethylenediamine, N,N'-bis(4- aminophenyl)tetramethylenediamine, N, N'-bis( -hydroxy ethyl) -N,N '- bis(4- amino phenyl)tetramethylenediamine, N,N'-bis(4- methylaminophenyl)tetramethylenediamine, N,N'-bis(ethyl)-N,N'- bis(4'-amino-3 '-methylphenyl)ethylenediamine and l , 8-bis(2,5 - dia
- para-aminophenols examples that may be mentioned include para-aminophenol, 4-amino-3 -methylpheno l, 4- amino-3 -fluoropheno l, 4-amino-3 -chlorophenol, 4-amino-3 - hydroxymethylpheno l, 4-amino-2-methylpheno l, 4-amino-2- hydroxymethylpheno l, 4-amino-2-methoxymethylpheno l, 4-amino-2- amino methylpheno l, 4-amino-2-( -hydroxyethylaminomethyl)pheno l and 4-amino-2-fluorophenol, and the addition salts thereof with an acid.
- heterocyclic bases by way o f example, o f pyridine derivatives, pyrimidine derivatives and pyrazole derivatives.
- pyridine derivatives that may be mentioned are the compounds described, for examp le, in patents GB 1 026 978 and GB 1 153 196, for instance 2,5 -diaminopyridine, 2-(4- methoxyphenyl)amino-3 -aminopyridine and 3 ,4-diaminopyridine, and the addition salts thereof.
- Other pyridine oxidation bases that are useful in the present invention are the 3-aminopyrazolo[ 1 ,5-a]pyridine oxidation bases or the addition salts thereof described, for example, in patent application FR 2 801 308.
- Examples that may be mentioned include pyrazolo[ 1 ,5- a]pyrid-3-ylamine, 2-acetylaminopyrazolo[ 1 ,5-a]pyrid-3-ylamine, 2- morpholin-4-ylpyrazolo[ 1 ,5-a]pyrid-3-ylamine, 3-aminopyrazolo[ 1 ,5- a]pyridine-2-carboxylic acid, 2-methoxypyrazolo[ 1 ,5-a]pyrid-3- ylamine, (3-aminopyrazolo[ 1 ,5-a]pyrid-7-yl)methanol, 2-(3- aminopyrazolo[ 1 ,5-a]pyrid-5-yl)ethanol, 2-(3-aminopyrazolo[ 1 ,5- a]pyrid-7-yl)ethanol, (3-aminopyrazolo[ 1 ,5-a]pyrid-2-yl)methanol, 3,6
- pyrimidine derivatives that may be mentioned are the compounds described, for example, in patents DE 2359399, JP 88- 169571, JP 05-63124 and EP 0 770 375 or patent application WO 96/15765, such as 2,4,5,6-tetraaminopyrimidine, 4-hydroxy-2,5,6- triaminopyrimidine, 2-hydroxy-4,5,6-triaminopyrimidine, 2,4- dihydroxy-5,6-diaminopyrimidine, 2,5,6-triaminopyrimidine and addition salts thereof, and tautomeric forms thereof, when a tautomeric equilibrium exists.
- pyrazole derivatives that may be mentioned are the compounds described in patents DE 3843892 and DE 4133957 and patent applications WO 94/08969, WO 94/08970, FR-A-2733 749 and DE 19543988, such as 4,5-diamino- 1 -methylpyrazole, 4,5-diamino- 1 - ( -hydroxyethyl)pyrazole, 3,4-diaminopyrazole, 4,5-diamino- 1 -(4 '- chlorobenzyl)pyrazole, 4,5-diamino- 1 ,3-dimethylpyrazole, 4,5- diamino -3 -methyl- 1 -phenylpyrazole, 4,5-diamino - 1 -methyl- 3- phenylpyrazole, 4 -amino - 1 ,3-dimethyl-5-hydrazinopyrazole, 1 -benzyl- 4,5-d
- Use will preferably be made of a 4,5-diaminopyrazole and even more preferentially of 4,5-diamino- 1 -( -hydroxyethyl)pyrazole and/or a salt thereof.
- pyrazole derivatives of diamino-N,N-dihydropyrazolopyrazolones and in particular those described in application FR-A-2886 136, such as the following compounds and the addition salts thereof: 2,3-diamino-6,7-dihydro- lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 -one, 2-amino-3-ethylamino-6,7- dihydro- lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 - one, 2-amino-3- isopropylamino-6,7-dihydro-lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 -one, 2- amino-3-(pyrrolidin-l-yl)-6,7-dihydro-lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1-one,
- heterocyclic bases use will preferably be made of 4,5- diamino- 1 - ( ⁇ -hydro xyethyl)pyrazole and/or 2,3-diamino-6,7-dihydro- lH,5H-pyrazolo[l,2-a]pyrazol-l-one and/or a salt thereof.
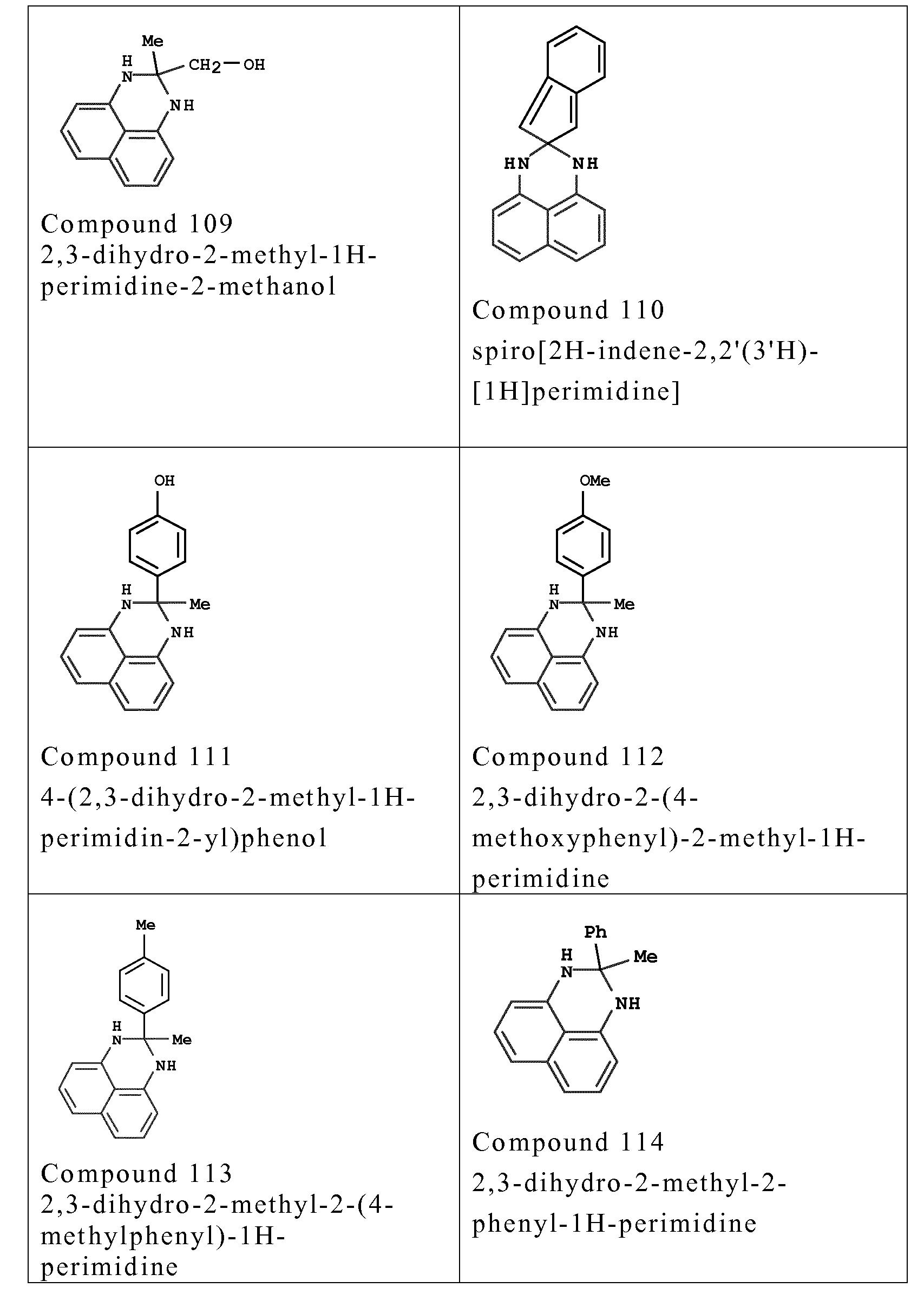
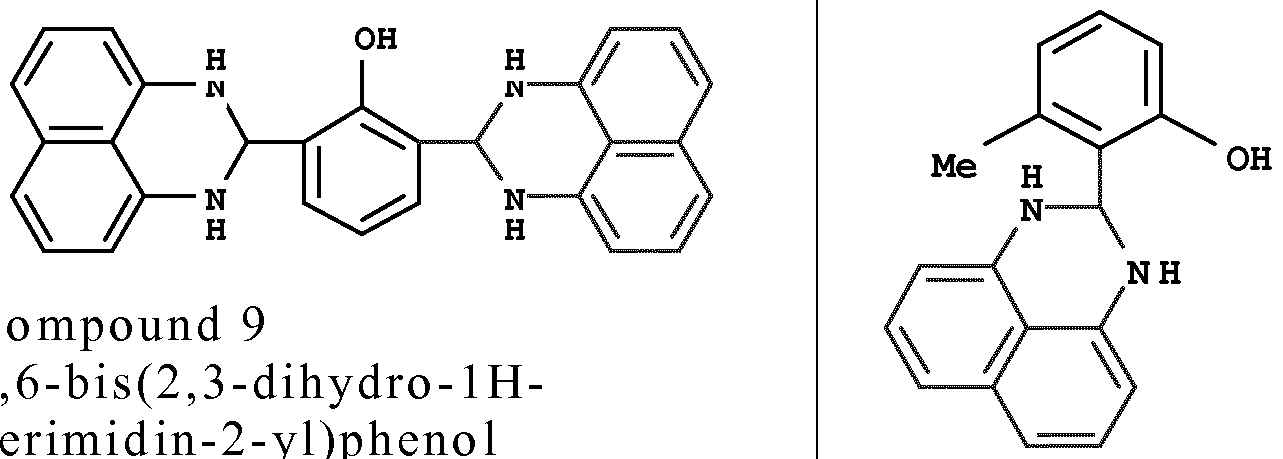
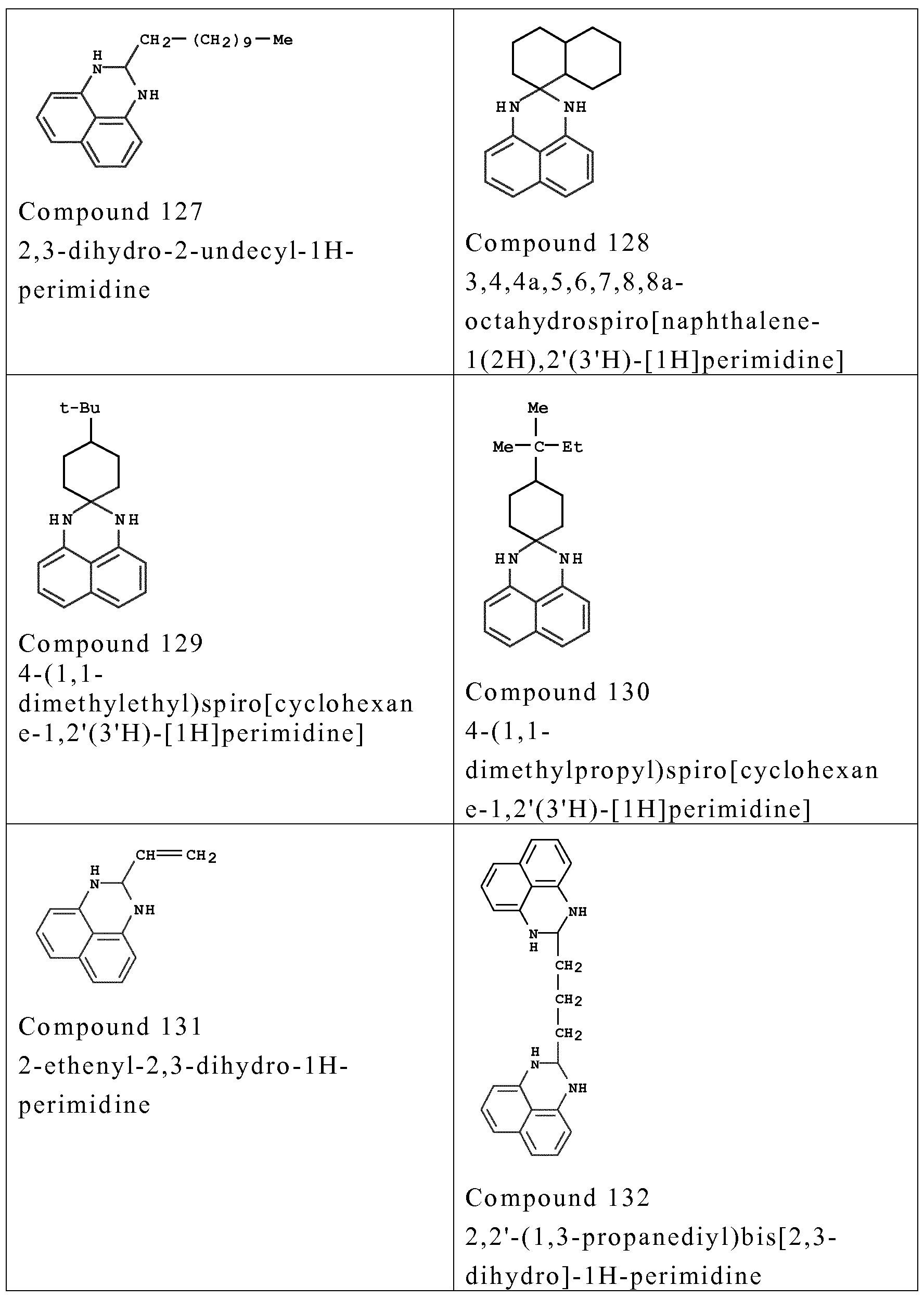
- composition according to the invention also comprises one or more couplers chosen from the couplers of formula (I) below and the addition salts thereof, isomers thereof, tautomers thereof, solvates thereof and dimers thereof:
- s denotes 0 or 1
- p denotes 0 or 1
- Ri, R 2 , R 3 , R 4 , R 5 , R 6 , R7 and Rs denote, independently of each other, a hydrogen atom; an amino group; a halogen atom; a hydroxyl group; a Ci-Cs alkoxy group; a C 2 -C 5 acylamino group; a group CHO, a linear, branched or cyclic Ci-Cis alkyl or C 2 -Cis alkylene group, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen, an aryl group, the alkyl, alkylene or aryl groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic acid, Ci-Cis alkyl ester, carbamate, carboxamide, amino, alkylamino, dialkylamino, carboxyl, alkoxy and halogen atoms, C 3 -C 6 cycloal
- the groups R 7 and Rs may form, with the atom that bears them, a ring optionally comprising one or more heteroatoms, this ring possibly being substituted with a C i -C 6 alkyl group or a hydroxyl group, or possibly being fused with one or more other rings optionally bearing one or more heteroatoms, one or more carbon atoms o f the ring(s) possibly being replaced with one or more carbonyl groups .
- R 7 may also denote a radical derived from a perimidinyl group of formula (I), thus leading to a dimer; the two perimidinyl groups are then preferably linked via the carbon atoms in position 2,
- R 7 may denote a linear or branched C 1 - C 1 0 alkylene chain, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen and/or optionally comprising a ring, or alternatively a saturated or unsaturated ring, optionally comprising one or more heteroatoms; a chain comprising several and preferably two saturated or unsaturated rings linked together via a covalent bond or via a heteroatom, the chains or the ring possibly being substituted with a hydroxyl group or with a perimidinyl radical o f formula (I) preferably via the carbon atom in position 2,
- R 4 when R 4 is in position 7 and R 5 is in position 6, then they can form, together with the carbon atoms that bear them, a 5 - to 8- membered ring optionally comprising one or more heteroatoms .
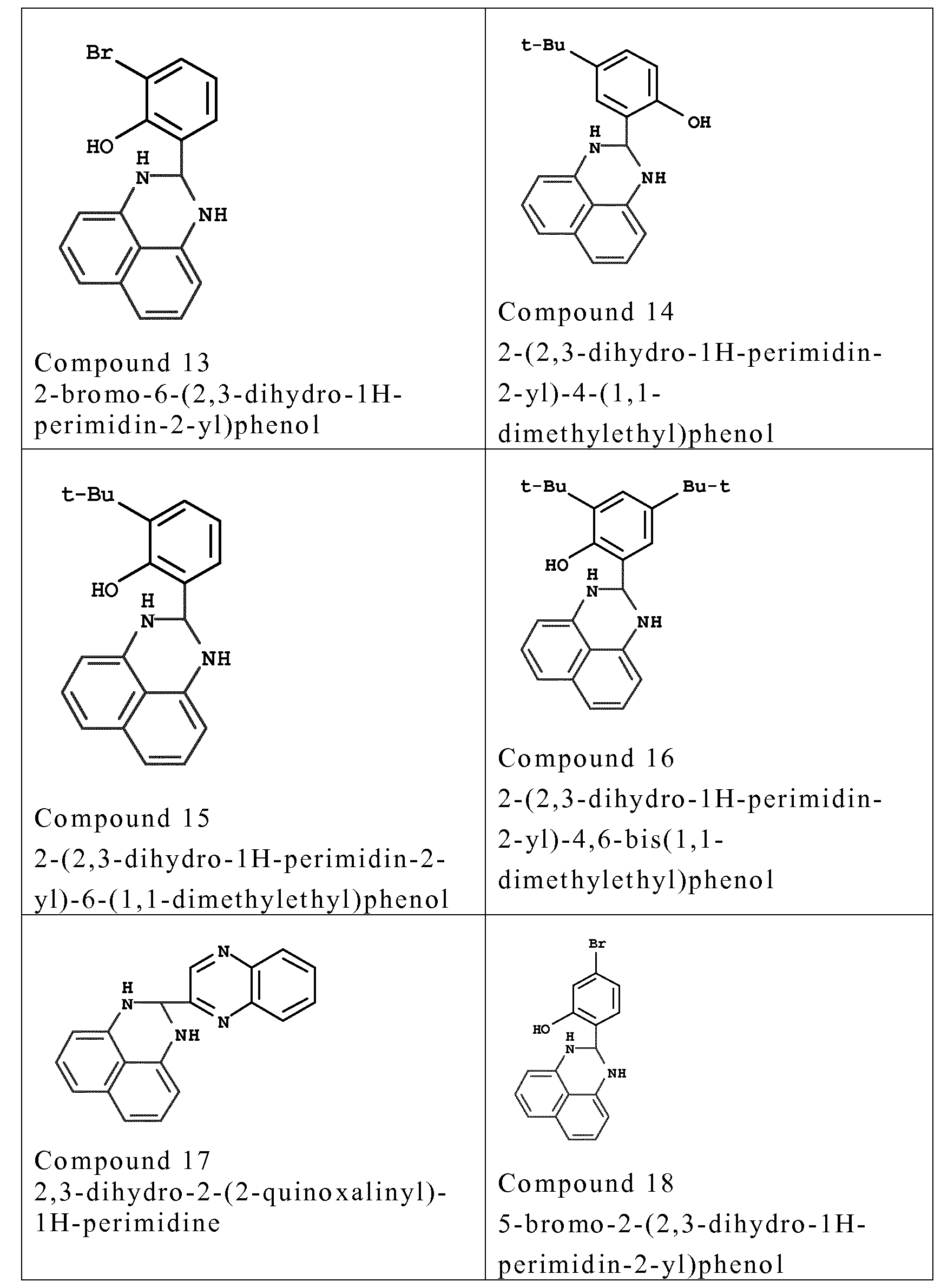
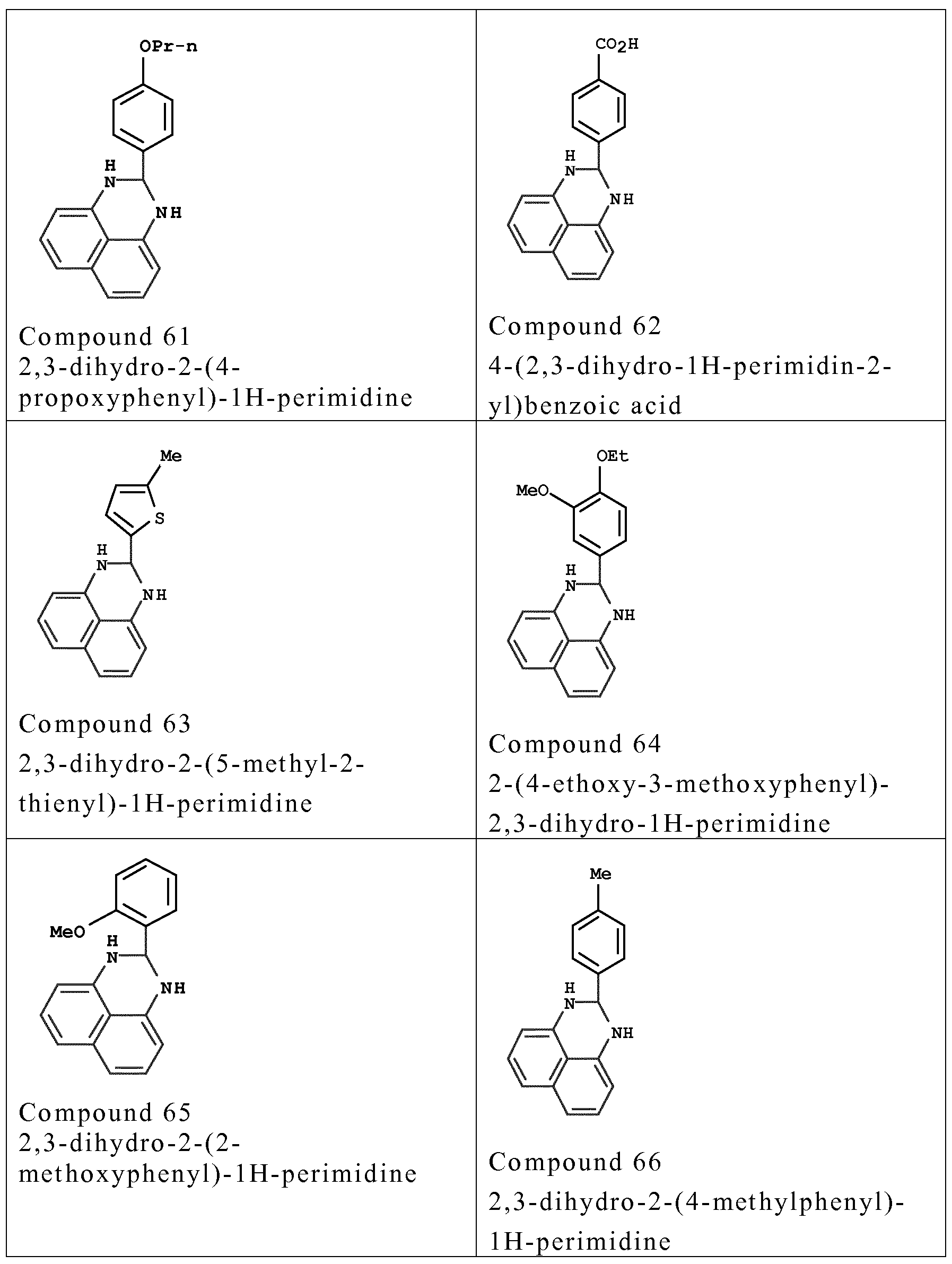
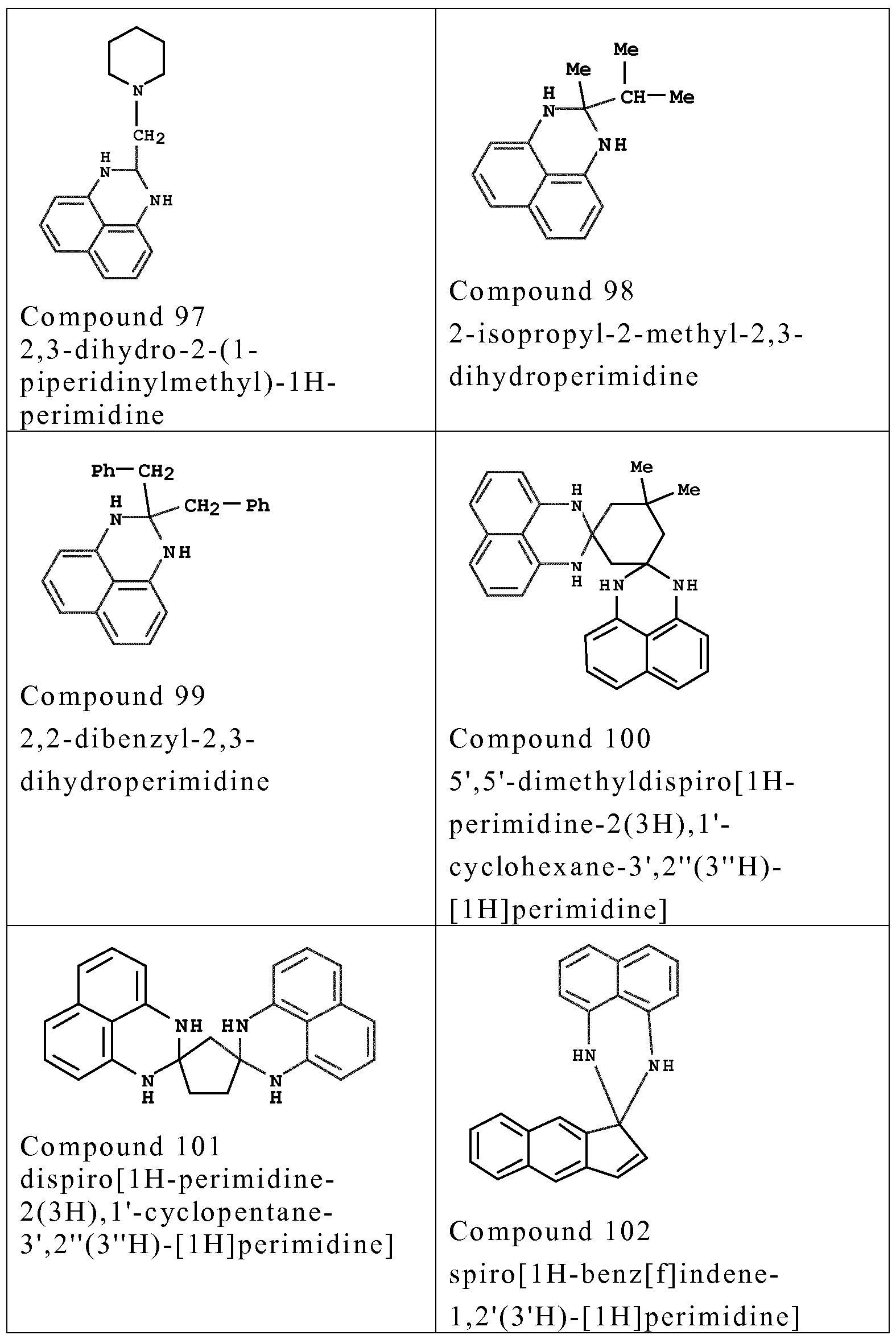
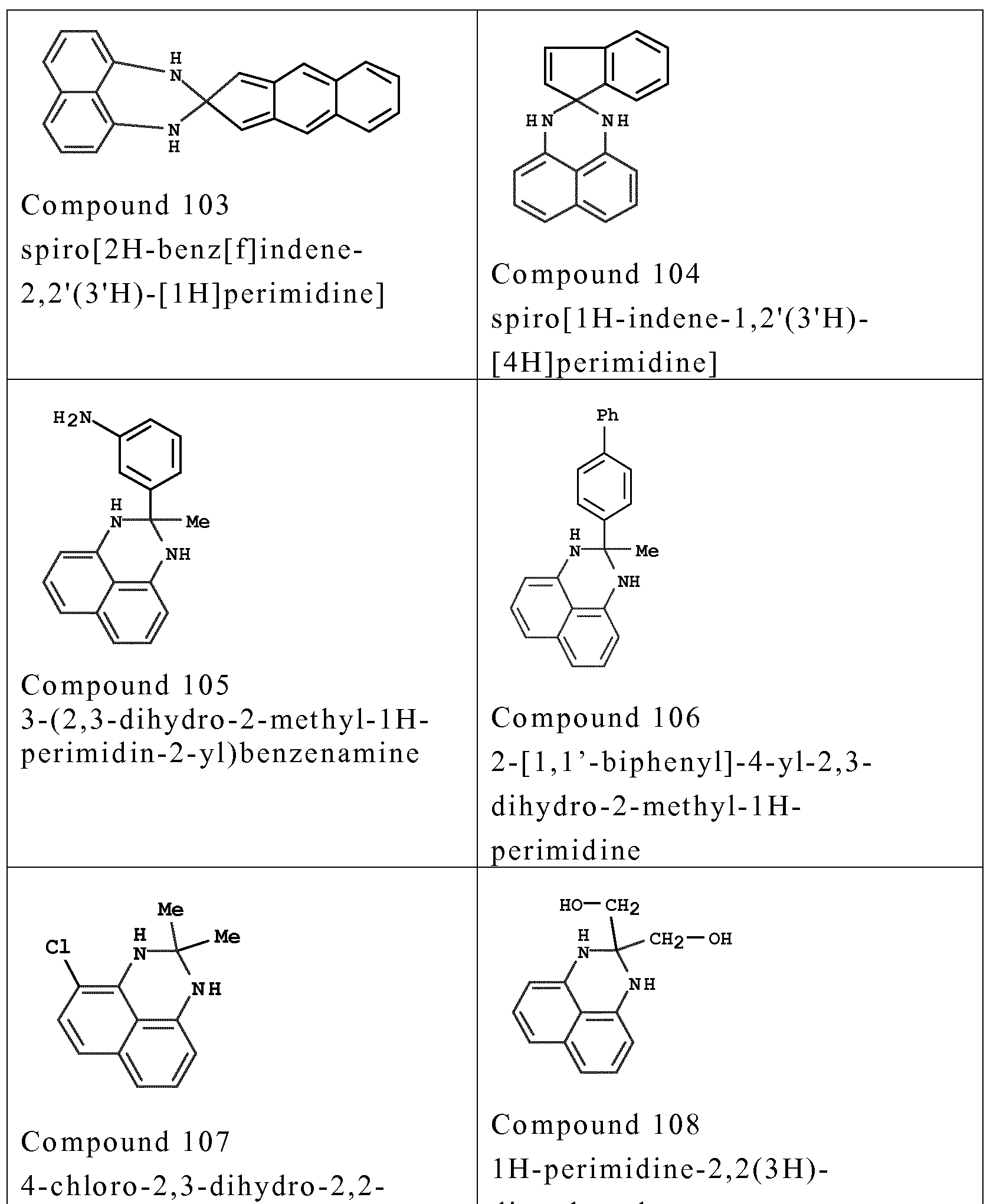
- couplers o f formula (I) are chosen from the fo llowing compounds :
- p denotes 1 .
- p denotes 0.
- R3 and R 6 represent a hydrogen atom.
- At least one of the groups Ri and R 2 represent a hydrogen atom.
- R 4 and R 5 represent a hydrogen atom.
- composition o f the invention may also comprise one or more additional couplers other than the couplers that are usually used, of formula (I) as described previously.
- couplers mention may be made especially o f meta-phenylenediamines, meta-aminopheno ls, meta-dipheno ls, naphthalene-based couplers and heterocyclic couplers, and also the addition salts thereof.
- Examples that may be mentioned include 1,3- dihydroxybenzene, 1 ,3-dihydroxy-2-methylbenzene, 4-chloro- 1 ,3- dihydroxybenzene, 2,4-diamino- 1 -(P-hydroxyethyloxy)benzene, 2- amino -4 -( ⁇ - hydroxy ethy lamino) - 1 -methoxybenzene, 1,3- diaminobenzene, 1 ,3-bis(2,4-diaminophenoxy)propane, 3- ureidoaniline, 3-ureido- 1 -dimethylaminobenzene, sesamol, 1- ⁇ - hydroxyethylamino-3,4-methylenedioxybenzene, a-naphthol, 2- methyl- 1 -naphthol, 6-hydroxyindole, 4-hydroxyindole, 4-hydroxy-N- methylindole, 2-amino-3-hydroxypyridine, 6- hydroxy
- addition salts of the oxidation bases and couplers that can be used within the context of the invention are especially chosen from the addition salts with an acid such as the hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, phosphates and acetates.
- an acid such as the hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, phosphates and acetates.
- the oxidation base(s) each advantageously represent from
- composition 0.0001% to 10% by weight relative to the total weight of the composition, and preferably from 0.005% to 5% by weight relative to the total weight of the composition.
- the coupler(s) each advantageously represent from 0.0001%) to 10% by weight relative to the total weight of the composition, and preferably from 0.005% to 5% by weight relative to the total weight of the composition.
- composition according to the invention may optionally comprise one or more synthetic or natural direct dyes, chosen from ionic or nonionic species, preferably cationic or nonionic species .
- Examples o f suitable synthetic direct dyes include azo direct dyes; (poly)methine dyes such as cyanins, hemicyanins and styryls; carbonyl dyes; azine dyes; nitro(hetero)aryl dyes; tri(hetero)arylmethane dyes; porphyrin dyes; and phthalocyanin dyes, alone or as mixtures.
- azo direct dyes include azo direct dyes; (poly)methine dyes such as cyanins, hemicyanins and styryls; carbonyl dyes; azine dyes; nitro(hetero)aryl dyes; tri(hetero)arylmethane dyes; porphyrin dyes; and phthalocyanin dyes, alone or as mixtures.
- Use may also be made of extracts or decoctions comprising these natural dyes and in particular henna-based poultices or extracts .
- the direct dye(s) more particularly represent from 0.00 1 % to 10% by weight and preferably from 0.005 % to 5 % by weight of the total weight of the composition.
- composition according to the invention may be an anhydrous or aqueous composition. It is preferably aqueous.
- aqueous composition is understood to mean a composition comprising more than 5 % by weight o f water, preferably more than 10% by weight of water and more advantageously still more than 20% by weight of water.
- the cosmetic composition according to the invention may optionally comprise one or more organic solvents.
- Examples o f organic so lvents that may be mentioned include linear or branched C 2 - C 4 alkano ls, such as ethanol and isopropano l; glycero l; polyo ls and polyo l ethers, for instance 2-butoxyethano l, propylene glyco l, dipropylene glyco l, propylene glyco l monomethyl ether, diethylene glyco l monomethyl ether and monoethyl ether, and also aromatic alcoho ls, for instance benzyl alcohol or phenoxyethano l, and mixtures thereof.
- the organic so lvent(s), if they are present, represent a content usually ranging from 1 % to 40% by weight and preferably from 5 % to 30% by weight relative to the weight of the composition.
- composition according to the invention may also contain various adjuvants conventionally used in hair dye compositions, such as anionic, cationic, nonionic, amphoteric or zwitterionic polymers or mixtures thereo f; mineral thickeners, and in particular fillers such as clays or talc; organic thickeners with, in particular, anionic, cationic, nonionic and amphoteric polymeric associative thickeners other than the polymers mentioned previously; antioxidants; penetrants; sequestrants; fragrances; dispersants; film-forming agents; ceramides; preserving agents; opacifiers .
- adjuvants conventionally used in hair dye compositions such as anionic, cationic, nonionic, amphoteric or zwitterionic polymers or mixtures thereo f; mineral thickeners, and in particular fillers such as clays or talc; organic thickeners with, in particular, anionic, cationic, nonionic and amphoteric polymeric associative thickeners other
- the above adjuvants are generally present in an amount for each o f them o f between 0.01 % and 20% by weight relative to the weight of the composition.
- the composition may also comprise one or more mineral or organic thickeners .
- the mineral thickeners may be chosen from organophilic clays and mineral fillers .
- the organophilic clay may be chosen from montmoriUonite, bentonite, hectorite, attapulgite and sepiolite, and mixtures thereof.
- the clay is preferably a bentonite or a hectorite .
- These clays can be modified with a chemical compound chosen from quaternary ammoniums, tertiary amines, amine acetates, imidazo lines, amine soaps, fatty sulfates, alkylarylsulfonates and amine oxides, and mixtures thereof.
- Organophilic clays that may be mentioned include quaternium- 1 8 bentonites, such as those so ld under the names Bentone 3 , Bentone
- the mineral thickeners are chosen from inorganic fillers, in particular kao linite.
- the organic thickeners may be chosen from associative or non- associative thickening polymers, in particular associative thickening polymers.
- the term "associative thickening polymer” means a thickening polymer comprising at least one C 8 - C 30 fatty chain and at least one hydrophilic unit.
- the associative thickening polymers are chosen from polyurethane polyethers comprising in their chain both hydrophilic blo cks usually o f po lyoxyethylenated nature and hydrophobic blo cks, which may be aliphatic sequences alone and/or cycloaliphatic and/or aromatic sequences,
- Aculyn 46® is a po lycondensate of polyethylene glyco l containing 150 or 1 80 mo l o f ethylene oxide, of stearyl alcoho l and o f methylenebis(4-cyclohexyl isocyanate) (SMDI), at 15 % by weight in a matrix o f maltodextrin (4%) and water (8 1 %);
- Aculyn 44® is a polycondensate of polyethylene glyco l containing 150 or 1 80 mo l o f ethylene oxide, o f decyl alcoho l and of methylenebis(4-cyclohexyl isocyanate) (SMDI), at 35 % by weight in a mixture of propylene glyco l (39%) and water (26%)] .
- the thickeners represent from 1 % to 30% by weight relative to the weight of the composition.
- composition according to the invention may comprise one or more chemical oxidizing agents .
- the chemical oxidizing agent(s) are chosen from hydrogen peroxide, urea peroxide, alkali metal bromates or ferricyanides, peroxygenated salts, for instance persulfates, perborates, peracids and precursors thereof, and percarbonates o f alkali metals or alkaline-earth metals, and peracids and precursors thereo f.
- One or more redox enzymes such as laccases, peroxidases and 2-electron oxidoreductases (such as uricase), optionally in the presence o f the respective donor or cofactor thereof, may also be used as oxidizing agent.
- This oxidizing agent advantageously consists o f hydrogen peroxide, in particular in aqueous solution (aqueous hydrogen peroxide so lution), the concentration of which may vary, more particularly from 0. 1 % to 50% by weight, even more preferentially from 0.5 % to 20% by weight and better still from 1 % to 15 % by weight relative to the composition.
- composition used in the process according to the invention corresponds to the composition as defined previously and that is free of chemical oxidizing agent; this composition also being used in the presence of at least one chemical oxidizing agent.
- the process according to the invention consists in applying to the said fibres a composition according to the invention, in the presence of at least one chemical oxidizing agent.
- composition used in the process according to the invention i. e. used in the presence o f at least one chemical oxidizing agent
- the temperature during the dyeing process is conventionally between room temperature (between 15 °C and 25 ° C) and 80°C and preferably between room temperature and 60°C .
- the human keratinous fibres are advantageously rinsed with water. They can optionally be washed with a shampoo, fo llowed by rinsing with water, before being dried or left to dry.
- composition applied in the process according to the invention i. e. used in the presence o f at least one chemical o xidizing agent
- compositions preferably two or three compositions .
- the composition applied in the process according to the invention results from the mixing o f two compositions.
- composition (A) comprising at least one oxidation base, at least one coupler chosen from the couplers o f formula (I) and at least one basifying agent is mixed with a composition (B) comprising at least one chemical oxidizing agent; at least one of the compositions (A) and (B) comprising at least one fatty substance, and at least one of the compositions (A) and (B) comprising at least one surfactant, the fatty substance content o f the composition according to the invention, resulting from the mixing o f compositions (A) and (B), comprising at least 25 % by weight of fatty substance.
- At least one of the compositions (A) and (B) is advantageously aqueous.
- composition (A) is aqueous.
- composition (B) is also aqueous .
- composition (A) preferably comprises at least 50% by weight of fatty substances, and more preferably still at least 50% by weight of fatty substances that are liquid at room temperature (25 °C), relative to the weight of this composition (A) .
- composition (A) is a direct emulsion (oil-in-water:
- compositions (A) and (B) are preferably mixed together in a weight ratio (A)/(B) ranging from 0.2 to 10 and better still from 0.5 to 2.
- the composition used in the process according to the invention results from the mixing of three compositions.
- the three compositions are aqueous or alternatively at least one of them is anhydrous.
- anhydrous cosmetic composition is understood to mean a cosmetic composition exhibiting a water content of less than 5% by weight, preferably of less than 2% by weight and more preferably still of less than 1% by weight, relative to the weight of the said composition.
- water present in the composition is more particularly "bound water”, such as water of crystallization in salts, or traces of water absorbed by the raw materials used in the preparation of the compositions according to the invention.
- ⁇ ' aqueous compositions
- ⁇ ' anhydrous composition
- the anhydrous composition ( ⁇ ') then preferably comprises at least one fatty substance, and more preferentially at least one liquid fatty substance.
- Composition ( ⁇ ') then preferably comprises at least one oxidation base and at least one coupler chosen from the couplers of formula (I).
- Composition (C) then preferably comprises at least one chemical oxidizing agent.
- the basifying agent(s) are included in compositions ( ⁇ ') and/or ( ⁇ ') and preferably only in composition ( ⁇ ').
- the surfactant(s) they are included in at least one of compositions ( ⁇ '), ( ⁇ ') and (C).
- the composition according to the invention i.e. the composition derived from the mixing of the three compositions ( ⁇ '), ( ⁇ ') and (C), has a fatty substance content of at least 25% by weight relative to the weight of the composition derived from the mixing of the abovementioned three compositions.
- compositions ( ⁇ '), ( ⁇ ') and (C) are preferably mixed together in a weight ratio [(A')+(B')]/(C) ranging from 0.2 to 10 and more particularly from 0.5 to 2 and in a weight ratio ( ⁇ ')/( ⁇ ') ranging from 0.5 to 10 and preferably from 1 to 5.
- the invention relates to a first multi-compartment device comprising a first compartment containing composition (A) as described above and at least a second compartment containing composition (B) as described above; the compositions (A) and (B) of the compartments being intended to be mixed together before application to give a composition according to the invention; the amount of fatty substance of which represents at least 25% by weight relative to the weight of the formulation resulting from the mixing of compositions (A) and (B).
- the invention also relates to a second multi-compartment device comprising a first compartment containing composition ( ⁇ ') as described above and a second compartment containing a cosmetic composition ( ⁇ ') as described above and at least a third compartment comprising composition (C) as described above, the compositions of the compartments being intended to be mixed together before application to give the composition according to the invention; the amount of fatty substance in the composition representing at least 25% by weight relative to the weight of the composition of the invention, i.e. resulting from the mixing of compositions ( ⁇ '), ( ⁇ ') and (C).
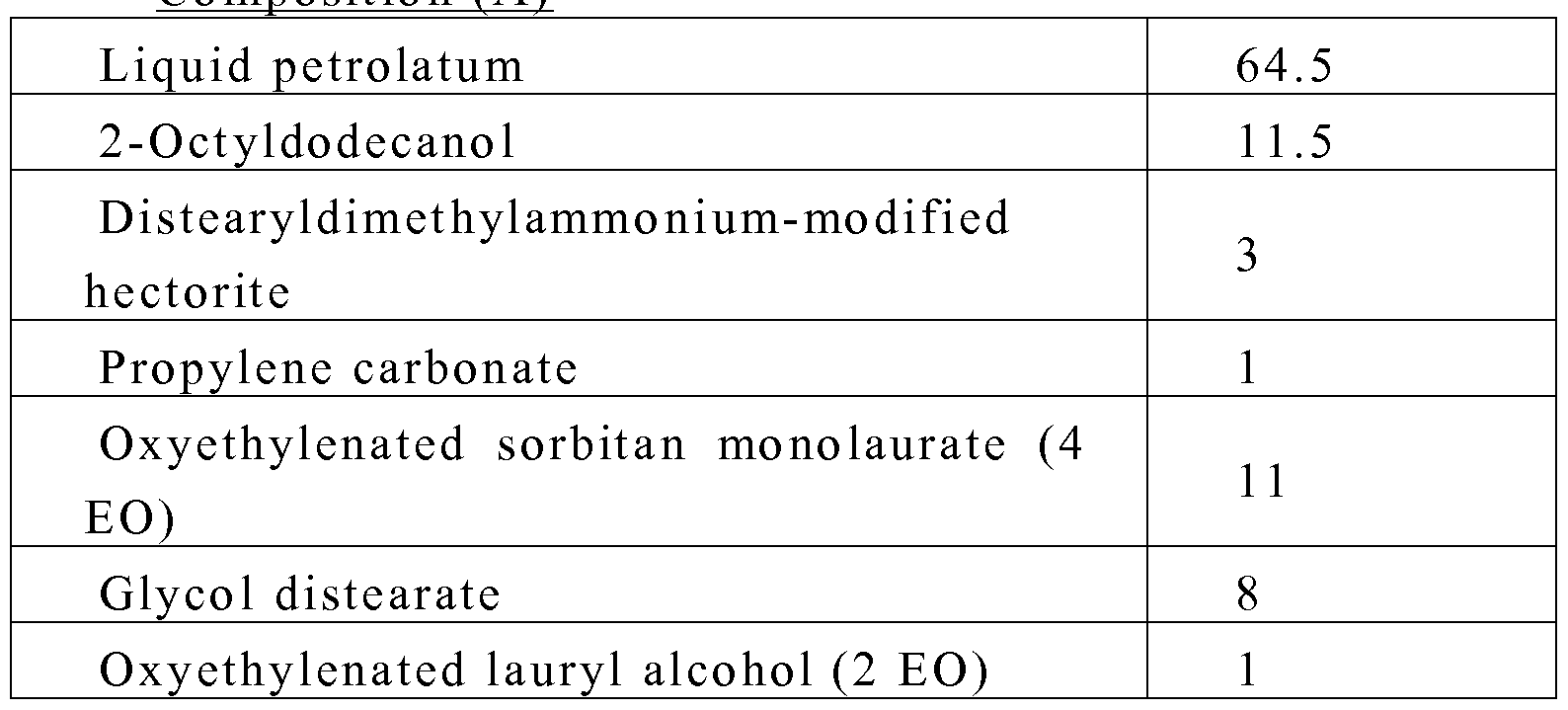
- compositions are prepared in which the amounts are expressed in grams of materials in their given state.
- Vitamin C L-ascorbic acid 0.25
- composition A -10 parts of composition A
- Each of the mixtures is then applied to locks of natural hair containing 90% white hairs.
- the “mixture/lock” bath ratio is respectively 10/1 (g/g).
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Cosmetics (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
Abstract
The present invention relates to a cosmetic composition for treating keratin fibres, in particular human keratin fibres such as the hair, comprising : at least 25 % by weight, relative to the total weight of the composition, of one or more fatty substances, one or more surfactants, one or more basifying agents, one or more oxidation bases, and one or more couplers of formula (I) below, addition salts thereof, isomers thereof, tautomers thereof, solvates thereof and dimers thereof: The invention also relates to processes using the composition according to the invention and to a multi-compartment device.
Description
Oxidation dye composition comprising a particular coupler in a medium rich in fatty substances, and processes and device suitable therefor The present invention relates to a composition for the oxidation dyeing o f keratin fibres, in particular o f human keratin fibres such as the hair, comprising at least 25 % by weight, relative to the total weight of the composition, o f one or more fatty substances, one or more surfactants, one or more basifying agents, one or more oxidation bases and one or more particular couplers .
The present invention also relates to dyeing processes using this composition in the presence o f at least one chemical oxidizing agent, and to a multi-compartment device suitable for using this composition.
The present invention relates to the field o f the dyeing o f keratinous fibres and more particularly to the field of hair dyeing.
Many people have sought for a long time to modify the co lour of their hair and in particular to mask their white hair. Essentially two types of dyeing have been developed to do this .
The first type o f dyeing is "permanent" or oxidation dyeing, which uses dye compositions containing oxidation dye precursors, generally known as oxidation bases. These oxidation bases are co lourless or weakly coloured compounds, which, when combined with oxidizing products, may give rise to coloured compounds via a process of oxidative condensation.
It is also known that the shades obtained with these oxidation bases may be varied by combining them with couplers or co louration modifiers, the latter being chosen especially from aromatic meta- diamines, meta-aminopheno ls, meta-dipheno ls and certain heterocyclic compounds such as indo le compounds. The variety o f the mo lecules used as oxidation bases and couplers allows a rich palette o f co lours to be obtained.
The second type o f dyeing is " semi-permanent" dyeing or direct dyeing, which consists in applying direct dyes to the keratin
fibres, the said dyes being coloured and colouring mo lecules that have affinity for the fibres, and then leaving them to take, to allow the mo lecules to penetrate by diffusion to the interior o f the fibre, and then rinsing the fibres .
In order to perform these co lorations, the direct dyes generally used are chosen from nitrobenzene, anthraquinone, nitropyridine, azo , xanthene, acridine, azine and triarylmethane direct dyes .
The use o f perimidine couplers is known in hair dyeing, especially in document US 5 922 086. However, it has been observed that, in standard formulations, compositions using this type of coupler give weak and very selective co lorations.
There is thus a real need for dye compositions and/or dyeing processes that give satisfactory levels o f co loration in terms o f power and selectivity.
This aim is achieved by the present invention, one subj ect o f which is especially a composition for the oxidation dyeing keratin fibres, in particular o f human keratin fibres such as the hair, comprising :
- at least 25 % by weight, relative to the total weight of the composition, of one or more fatty substances,
- one or more surfactants,
- one or more basifying agents,
- one or more oxidation bases, and
- one or more couplers o f formula (I) below, addition salts thereo f, isomers thereof, tautomers thereof, so lvates thereof and dimers thereof:
in which:
s denotes 0 or 1
p denotes 0 or 1
and s+p = 1
Ri, R2, R3, R4, R5, R6, R7 and Rs denote, independently of each other, a hydrogen atom; an amino group; a halogen atom; a hydroxyl group; a Ci-Cs alkoxy group; a C2-C5 acylamino group; a group CHO, a linear, branched or cyclic Ci-Cis alkyl or C2-Cis alkylene group, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen, an aryl group, the alkyl, alkylene or aryl groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic acid, Ci-Cis alkyl ester, carbamate, carboxamide, amino, alkylamino, dialkylamino, carboxyl, alkoxy and halogen atoms, C3-C6 cycloalkyl, 5- to 8-membered heterocycle comprising one or more heteroatoms such as nitrogen, sulfur or oxygen, one or more carbon atoms of the cycloalkyl or heterocyclic groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic acid, Ci-Cis alkyl ester, carbamate, carboxamide, amino, alkylamino, dialkylamino, carboxyl, alkoxy and halogen atoms,
the groups R7 and Rs may form, with the atom that bears them, a ring optionally comprising one or more heteroatoms, this ring possibly being substituted with a Ci-C6 alkyl group or a hydroxyl group, or possibly being fused with one or more other rings optionally bearing one or more heteroatoms, one or more carbon atoms of the ring(s) possibly being replaced with one or more carbonyl groups.
R7 may also denote a radical derived from a perimidinyl group of formula (I), thus leading to a dimer; the two perimidinyl groups are then preferably linked via the carbon atoms in position 2,
or alternatively R7 may denote a linear or branched C1-C10 alkylene chain, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen and/or optionally comprising a ring, or alternatively a saturated or unsaturated ring, optionally comprising one or more heteroatoms; a chain comprising several and preferably two saturated or unsaturated
rings linked together via a covalent bond or via a heteroatom, the chains or the ring possibly being substituted with a hydroxyl group or with a perimidinyl radical o f formula (I) preferably via the carbon atom in position 2,
when R4 is in position 7 and R5 is in position 6, then they can form, together with the carbon atoms that bear them, a 5 - to 8- membered ring optionally comprising one or more heteroatoms .
The present invention also relates to dyeing processes using this composition in the presence o f at least one chemical oxidizing agent, and to a multi-compartment device suitable for using this composition.
Thus, the use o f the composition according to the invention in the presence o f at least one chemical oxidizing agent leads to strong co lorations with little selectivity. Furthermore, the co lorations obtained remain stable, in particular with respect to light.
Other characteristics and advantages o f the invention will emerge more clearly on reading the description and the examples that fo llow.
In the text hereinbelow, and unless otherwise indicated, the limits of a range of values are included in that range.
The human keratinous fibres treated by the method according to the invention are preferably the hair.
The expression "at least one" is equivalent to the expression "one or more" .
Fatty substances
The composition according to the invention comprises one or more fatty substances .
The term "fatty substance" means an organic compound that is inso luble in water at ordinary room temperature (25 °C) and at atmospheric pressure (760 mmHg) (solubility o f less than 5 %, preferably 1 % and even more preferentially 0. 1 %) . They exhibit, in their structure, at least one hydrocarbon chain comprising at least 6 carbon atoms or a sequence o f at least two siloxane groups. In addition, the fatty substances are generally so luble in organic so lvents
under the same temperature and pressure conditions, for instance chloroform, ethano l, benzene, liquid petrolatum or decamethylcyclopentasiloxane. These fatty substances are neither polyoxyethylenated nor polyglycerolated.
More particularly, the fatty substances are chosen from C6 - C i 6 hydrocarbons, hydrocarbons containing more than 16 carbon atoms, non-silicone oils o f animal origin, plant oils o f triglyceride type, synthetic triglycerides, fluoro oils, fatty alcoho ls, esters of fatty acids and/or of fatty alcoho ls other than triglycerides, and plant waxes, non- silicone waxes and silicones .
It is recalled that, for the purposes o f the invention, the fatty alcoho ls, fatty esters and fatty acids more particularly contain one or more linear or branched, saturated or unsaturated hydrocarbon-based groups comprising 6 to 30 carbon atoms, which are optionally substituted, in particular with one or more (in particular 1 to 4) hydroxyl groups. If they are unsaturated, these compounds may comprise one to three conjugated or unconjugated carbon-carbon double bonds .
As regards the C6 - C i 6 hydrocarbons, they are linear or branched, and possibly cyclic, and are preferably alkanes. Examp les that may be mentioned include hexane, dodecane and isoparaffins such as isohexadecane and isodecane.
Mention may be made, as hydrocarbon oils of animal origin, o f perhydrosqualene.
The triglyceride oils o f p lant or synthetic origin are preferably chosen from liquid fatty acid triglycerides containing from 6 to 30 carbon atoms, for instance heptanoic or octanoic acid triglycerides, or alternatively, for examp le, sunflower oil, corn oil, soybean oil, marrow oil, grapeseed oil, sesame seed oil, hazelnut oil, apricot oil, macadamia oil, arara oil, castor oil, avocado oil, caprylic/capric acid triglycerides, for instance those so ld by the company Stearineries Dubois or those so ld under the names Miglyo l® 8 1 0, 8 12 and 8 1 8 by the company Dynamit Nobel, jojoba oil and shea butter oil.
The linear or branched hydrocarbons o f mineral or synthetic origin containing more than 16 carbon atoms are preferably chosen
from liquid paraffins, petroleum j elly, liquid petrolatum, polydecenes, and hydrogenated polyisobutene such as Parleam® .
The fluoro oils may be chosen from perfluoromethylcyclopentane and perfluoro- 1 ,3 -dimethylcyclohexane, so ld under the names Flutec® PC I and Flutec® PC3 by the company BNFL Fluorochemicals; perfluoro- 1 ,2-dimethylcyclobutane; perfluoroalkanes such as dodecafluoropentane and tetradecafluorohexane, so ld under the names PF 5050® and PF 5060® by the company 3M, or bromoperfluorooctyl so ld under the name Foralkyl® by the company Atochem; nonafluoromethoxybutane and nonafluoroethoxyisobutane; perfluoromorpholine derivatives such as 4-trifluoromethyl perfluoromorpholine sold under the name PF 5052® by the company 3M .
The fatty alcoho ls that may be used in the composition according to the invention are saturated or unsaturated, and linear or branched, and comprise from 6 to 30 carbon atoms and more particularly from 8 to 30 carbon atoms. Examples that may be mentioned include cetyl alcoho l, stearyl alcoho l and the mixture thereo f (cetylstearyl alcoho l), octyldodecanol, 2-butyloctanol, 2- hexyldecanol, 2-undecylpentadecanol, oleyl alcoho l and lino leyl alcoho l.
The wax(es) that may be used in the composition are chosen especially from carnauba wax, candelilla wax, esparto grass wax, paraffin wax, ozokerite, plant waxes, for instance olive wax, rice wax, hydrogenated jojoba wax or the absolute waxes o f flowers such as the essential wax o f blackcurrant blo ssom sold by the company Bertin (France), animal waxes, for instance beeswaxes, or modified beeswaxes (cerabellina); other waxes or waxy starting materials that may be used according to the invention are especially marine waxes such as the product sold by the company Sophim under the reference M82, and polyethylene waxes or polyo lefin waxes in general.
The fatty acids that may be used in the composition may be saturated or unsaturated and contain from 6 to 30 carbon atoms and in particular from 9 to 30 carbon atoms. They are more particularly
chosen from myristic acid, palmitic acid, stearic acid, behenic acid, oleic acid, linoleic acid, linolenic acid and isostearic acid.
As regards the esters o f a fatty acid and/or of a fatty alcoho l, which are advantageously different from the triglycerides mentioned above, mention may be made especially o f esters o f saturated or unsaturated, linear or branched C i -C26 aliphatic mono- or polyacids and o f saturated or unsaturated, linear or branched C i -C26 aliphatic mono- or polyalcoho ls, the total carbon number o f the esters more particularly being greater than or equal to 1 0.
Among the monoesters, mention may be made o f dihydroabietyl behenate; octyldodecyl behenate; isocetyl behenate; cetyl lactate; C 1 2-C 1 5 alkyl lactate; isostearyl lactate; lauryl lactate; lino leyl lactate; o leyl lactate; (iso)stearyl octanoate; isocetyl octanoate; octyl octanoate; cetyl octanoate; decyl o leate; iso cetyl isostearate; isocetyl laurate; isocetyl stearate; isodecyl octanoate; isodecyl o leate; isononyl isononanoate; isostearyl palmitate; methylacetyl ricino leate; myristyl stearate; octyl isononanoate; 2- ethylhexyl isononanoate; octyl palmitate; octyl pelargonate; octyl stearate; octyldodecyl erucate; o leyl erucate; ethyl and isopropyl palmitates, 2-ethylhexyl palmitate, 2-octyldecyl palmitate, alkyl myristates such as isopropyl, butyl, cetyl, 2-octyldodecyl, myristyl or stearyl myristate, hexyl stearate, butyl stearate, isobutyl stearate; dioctyl malate, hexyl laurate, 2-hexyldecyl laurate.
Still within the context of this variant, esters of C4-C22 dicarboxylic or tricarboxylic acids and of C i -C22 alcoho ls and esters of monocarboxylic, dicarboxylic or tricarboxylic acids and o f C2-C26 dihydroxy, trihydroxy, tetrahydroxy or pentahydroxy alcoho ls may also be used.
Mention may in particular be made of: diethyl sebacate; diisopropyl sebacate; diisopropyl adipate; di(n-propyl) adipate; dioctyl adipate; diisostearyl adipate; dioctyl maleate; glyceryl undecylenate; octyldodecyl stearoyl stearate; pentaerythrityl monoricino leate; pentaerythrityl tetraisononanoate; pentaerythrityl tetrapelargonate; pentaerythrityl tetraisostearate; pentaerythrityl
tetraoctanoate; propylene glyco l dicaprylate; propylene glyco l dicaprate; tridecyl erucate; triisopropyl citrate; triisostearyl citrate; glyceryl trilactate; glyceryl trioctanoate; trioctyldodecyl citrate; trio leyl citrate; propylene glyco l dio ctanoate; neopentyl glyco l diheptanoate; diethylene glyco l diisononanoate; and polyethylene glyco l distearates.
Among the esters mentioned above, it is preferred to use ethyl, isopropyl, myristyl, cetyl or stearyl palmitate, 2-ethylhexyl palmitate,
2-octyldecyl palmitate, alkyl myristates such as isopropyl, butyl, cetyl or 2-octyldodecyl myristate, hexyl stearate, butyl stearate, isobutyl stearate; dio ctyl malate, hexyl laurate, 2-hexyldecyl laurate, isononyl isononanoate or cetyl octanoate.
The composition may also comprise, as fatty ester, sugar esters and diesters of C6 - C30 and preferably C 12 - C22 fatty acids . It is recalled that the term " sugar" means oxygen-bearing hydrocarbon-based compounds bearing several alcoho l functions, with or without aldehyde or ketone functions, and which comprise at least 4 carbon atoms. These sugars can be monosaccharides, oligosaccharides or polysaccharides .
Mention may be made, as suitable sugars, for example, o f sucrose (or saccharose), glucose, galactose, ribose, fucose, maltose, fructose, mannose, arabino se, xylose, lactose and their derivatives, in particular alkyl derivatives, such as methyl derivatives, for example methylglucose.
The esters o f sugars and o f fatty acids can be chosen in particular from the group consisting of the esters or mixtures of esters of sugars described above and of saturated or unsaturated and linear or branched C6 - C30 and preferably C 12 - C22 fatty acids. If they are unsaturated, these compounds may comprise one to three conjugated or unconjugated carbon-carbon double bonds .
The esters according to this alternative form can also be chosen from mono-, di-, tri- and tetraesters and polyesters, and mixtures thereof.
These esters can, for example, be o leates, laurates, palmitates, myristates, behenates, cocoates, stearates, lino leates, lino lenates, caprates, arachidonates or their mixtures, such as, in particular, oleate/palmitate, oleate/stearate or palmitate/stearate mixed esters .
More particularly, use is made o f mono- and diesters and in particular mono- or di-o leate, -stearate, -behenate, -oleate/palmitate, - lino leate, -lino lenate or -oleate/stearate of sucrose, glucose or methylglucose.
Mention may be made, by way o f example, of the product sold under the name Glucate® DO by Amerchol, which is a methylglucose dio leate.
Examples o f esters or mixtures o f esters of sugar and o f fatty acid that may also be mentioned include :
- the products so ld under the names F 160, F 140, F 1 10, F90, F70 and SL40 by Crodesta, respectively denoting sucrose palmitate/stearates formed of 73 % monoester and 27% di- and triester, of 6 1 %) monoester and 39% di-, tri- and tetraester, of 52% monoester and 48%o di-, tri- and tetraester, of 45 % monoester and 55 % di-, tri- and tetraester, and o f 39% monoester and 61 % di-, tri- and tetraester, and sucrose mono laurate;
- the products sold under the name Ryoto Sugar Esters, for example referenced B370 and corresponding to sucrose behenate formed of 20% monoester and 80% diester, triester and polyester;
- the sucrose monopalmitate/stearate-dipalmitate/stearate so ld by Go ldschmidt under the name Tegosoft® PSE .
The silicones that can be used in the composition of the present invention are vo latile or non- vo latile, cyclic, linear or branched silicones, which are unmodified or mo dified with organic groups, having a viscosity from 5 x 10"6 to 2.5 m2/s at 25 °C, and preferably l x l O"5 to 1 m2/s .
The silicones which can be used in accordance with the invention can be provided in the form of oils, waxes, resins or gums .
Preferably, the silicone is chosen from polydialkylsiloxanes, in particular polydimethylsiloxanes (PDMSs), and organomodified
polysiloxanes comprising at least one functional group chosen from poly(oxyalkylene) groups, amino groups and alkoxy groups.
Organopolysiloxanes are defined in greater detail in Walter Noll's Chemistry and Technology of Silicones (1968), Academic Press. They may be volatile or non-volatile.
When they are volatile, the silicones are more particularly chosen from those with a boiling point of between 60°C and 260°C, and even more particularly from:
(i) cyclic polydialkylsiloxanes containing from 3 to 7 and preferably from 4 to 5 silicon atoms. They are, for example, octamethylcyclotetrasiloxane, sold in particular under the name Volatile Silicone® 7207 by Union Carbide or Silbione® 70045 V2 by Rhodia, decamethylcyclopentasiloxane, sold under the name Volatile Silicone® 7158 by Union Carbide and Silbione® 70045 V5 by Rhodia, and mixtures thereof.
Mention may also be made of cyclocopolymers of the dimethylsiloxane/methylalkylsiloxane type, such as Volatile Silicone® FZ 3109, sold by Union Carbide, having the formula:
D"-D' D"-D'-
CH3 CH3 withD": — Si-O— with D' -Si- I C
Mention may also be made of mixtures of cyclic polydialkylsiloxanes with organic compounds derived from silicon, such as the mixture of octamethylcyclotetrasiloxane and tetra(trimethylsilyl)pentaerythritol (50/50) and the mixture of octamethylcyclotetrasiloxane and oxy- 1 , 1 '-bis(2,2,2 ',2' ,3,3 '- hexatrimethylsilyloxy)neopentane;
(ii) linear volatile polydialkylsiloxanes containing 2 to 9 silicon atoms and having a viscosity of less than or equal to 5 x 10"6 m2/s at 25°C. An example is decamethyltetrasiloxane sold in particular under the name SH 200 by the company Toray Silicone. Silicones
belonging to this category are also described in the article published in Cosmetics and Toiletries, Vol. 91, Jan. 76, pp. 27-32, Todd & Byers, Volatile Silicone Fluids for Cosmetics .
Use is preferably made of non-volatile polydialkylsiloxanes, polydialkylsiloxane gums and resins, polyorganosiloxanes modified with the organofunctional groups above, and mixtures thereof.
These silicones are more particularly chosen from polydialkylsiloxanes, among which mention may be made mainly of polydimethylsiloxanes having trimethylsilyl end groups. The viscosity of the silicones is measured at 25°C according to ASTM standard 445 Appendix C.
Mention may be made, among these polydialkylsiloxanes, without implied limitation, of the following commercial products:
- the Silbione® oils of the 47 and 70047 series or the Mirasil oils sold by Rhodia, such as, for example, the oil 70047 V 500000;
- the oils of the Mirasil® series sold by Rhodia;
- the oils of the 200 series from Dow Corning, such as DC200 having a viscosity of 60000 mm2/s;
- the Viscasil® oils from General Electric and certain oils of the SF series (SF 96, SF 18) from General Electric.
Mention may also be made of polydimethylsiloxanes having dimethylsilanol end groups known under the name of dimethiconol (CTFA), such as the oils of the 48 series from Rhodia.
Mention may also be made, in this category of polydialkylsiloxanes, of the products sold under the names Abil Wax® 9800 and 9801 by Goldschmidt, which are polydi(Ci- C2o)alkylsiloxanes.
The silicone gums which can be used in accordance with the invention are in particular polydialkylsiloxanes and preferably polydimethylsiloxanes having high number-average molecular weights of between 200000 and 1 000000, used alone or as a mixture in a solvent. This solvent can be chosen from volatile silicones, polydimethylsiloxane (PDMS) oils, polyphenylmethylsiloxane (PPMS)
oils, isoparaffins, polyisobutylenes, methylene chloride, pentane, dodecane, tridecane or their mixtures .
Products which can be used more particularly in accordance with the invention are mixtures, such as :
- the mixtures formed from a polydimethylsiloxane hydroxylated at the chain end, or dimethicono l (CTFA), and from a cyclic polydimethylsiloxane, also known as cyclomethicone (CTFA), such as the product Q2 1401 sold by Dow Corning;
- the mixtures o f a po lydimethylsiloxane gum and o f a cyclic silicone, such as the product SF 1214 Silicone Fluid from General
Electric; this product is an SF 30 gum corresponding to a dimethicone, having a number-average mo lecular weight o f 500 000, dissolved in the oil SF 1202 Silicone Fluid corresponding to decamethy Icy clop entasiloxane;
- the mixtures of two PDMSs with different viscosities, and more particularly of a PDMS gum and a PDMS oil, such as the product
SF 1236 from General Electric. The product SF 1236 is the mixture of a gum SE 30 defined above having a viscosity o f 20 m2/s and of an oil
SF 96 with a viscosity o f 5 x 10"6 m2/s. This product preferably comprises 15 % of gum SE 30 and 85 % of an oil SF 96.
The organopolysiloxane resins that can be used in accordance with the invention are crosslinked siloxane systems containing the fo llowing units :
R2Si02/2 , Rs SiO i/2 , RSi03/2 and Si04/2
in which R represents an alkyl containing 1 to 16 carbon atoms. Among these products, the ones that are particularly preferred are those in which R denotes a C i -C4 lower alkyl group, more particularly methyl.
Mention may be made, among these resins, of the product sold under the name Dow Corning 593 or those so ld under the names
Silicone Fluid S S 4230 and S S 4267 by General Electric, which are silicones of dimethyl/trimethylsiloxane structure.
Mention may also be made o f the resins o f the trimethylsiloxysilicate type, so ld in particular under the names X22- 4914, X21 -5034 and X21 -5037 by Shin-Etsu.
The organomodified silicones that can be used in accordance with the invention are silicones as defined above and comprising in their structure one or more organofunctional groups attached via a hydrocarbon-based group.
In addition to the silicones described above, the organomodified silicones can be polydiarylsiloxanes, in particular polydiphenylsiloxanes, and polyalkylarylsiloxanes functionalized by the abovementioned organofunctional groups.
The polyalkylarylsiloxanes are chosen particularly from linear and/or branched polydimethyl/methylphenylsiloxanes and polydimethyl/diphenylsiloxanes with a viscosity of from lxlO"5 to 5xl0"2 m2/s at 25°C.
Among these polyalkylarylsiloxanes, examples that may be mentioned include the products sold under the following names:
- Silbione® oils of the 70641 series from Rhodia;
- oils of the Rhodorsil® 70633 and 763 series from Rhodia;
- the oil Dow Corning 556 Cosmetic Grade Fluid from Dow Corning;
- the silicones of the PK series from Bayer, such as the product PK20;
- the silicones of the PN and PH series from Bayer, such as the products PN1000 and PH1000;
- certain oils of the SF series from General Electric, such as SF 1023, SF 1154, SF 1250 and SF 1265.
Mention may be made, among the organomodified silicones, of polyorganosiloxanes comprising:
- polyethyleneoxy and/or polypropyleneoxy groups optionally comprising C6-C24 alkyl groups, such as the products named dimethicone copolyol sold by Dow Corning under the name DC 1248 or the oils Silwet® L 722, L 7500, L 77 and L 711 by Union Carbide, and the (Ci2)alkyl methicone copolyol sold by Dow Corning under the name Q25200;
- substituted or unsubstituted amino groups, such as the products sold under the names GP 4 Silicone Fluid and GP 7100 by
Genesee or the products sold under the names Q2 8220 and Dow Corning 929 or 939 by Dow Corning. The substituted amino groups are in particular C 1 - C 4 aminoalkyl groups;
- alkoxylated groups, such as the product so ld under the name Silicone Copolymer F-755 by SWS Silicones, and Abil Wax® 2428 , 2434 and 2440 by Goldschmidt.
More particularly, the fatty substances are chosen from compounds that are liquid or pasty at room temperature and at atmospheric pressure.
Preferably, the fatty substance is a compound which is liquid at the temperature of 25 °C and at atmospheric pressure.
The fatty substances are advantageously chosen from C6 - C i 6 hydrocarbons, hydrocarbons containing more than 16 carbon atoms, non-silicone oils o f animal origin, plant oils o f triglyceride type, synthetic triglycerides, fluoro oils, fatty alcoho ls, esters of fatty acids and/or of fatty alcoho ls other than triglycerides, and plant waxes, non- silicone waxes and silicones, and a mixture thereof.
Preferably, the fatty substance(s) are chosen from liquid petrolatum, polydecenes, fatty alcoho ls and liquid esters o f a fatty acid and/or of a fatty alcohol, or mixtures thereof.
Even more preferentially, the fatty substances are chosen from liquid petrolatum and octyldodecanol.
The composition according to the invention has a fatty substance content of at least 25 % by weight. More particularly, the fatty substance concentration ranges from 25 % to 80% by weight, preferably from 30% to 70% by weight and even more advantageously from 30% to 60% by weight relative to the total weight o f the composition.
Surfactants
The composition according to the invention also comprises one or more surfactants .
In particular, the surfactant(s) are chosen from anionic, amphoteric, zwitterionic, cationic and nonionic surfactants, and preferentially nonionic surfactants .
The term " anionic surfactant" means a surfactant comprising, as ionic or ionizable groups, only anionic groups. These anionic groups are preferably chosen from the groups -C(0)OH, -C(0)0-, - S03H, -S(0)20-, -OS(0)2OH, -OS(0)20-, -P(0)OH2, -P(0)20-, - P(0)02-, -P(OH)2, =P(0)OH, -P(OH)0-, =P(0)0-, =POH and =PO-, the anionic parts comprising a cationic counterion such as an alkali metal, an alkaline-earth metal or an ammonium.
As examp les of anionic surfactants that may be used in the composition according to the invention, mention may be made of alkyl sulfates, alkyl ether sulfates, alkylamido ether sulfates, alkylaryl polyether sulfates, monoglyceride sulfates, alkylsulfonates, alkylamidesulfonates, alkylarylsulfonates, a-o lefin sulfonates, paraffin sulfonates, alkyl sulfo succinates, alkyl ether sulfo succinates, alkylamide sulfo succinates, alkyl sulfoacetates, acylsarcosinates, acylglutamates, alkyl sulfosuccinamates, acylisethionates and N- acyltaurates, polyglycoside polycarboxylic acid and alkyl monoester salts, acyl lactylates, salts of D-galactoside uronic acids, salts of alkyl ether carboxylic acids, salts o f alkylaryl ether carboxylic acids, salts of alkylamido ether carboxylic acids; and the corresponding non- salified forms of all these compounds; the alkyl and acyl groups of all these compounds comprising from 6 to 24 carbon atoms and the aryl group denoting a phenyl group .
These compounds can be oxyethylenated and then preferably comprise from 1 to 50 ethylene oxide units.
The salts o f C6-C24 alkyl monoesters o f polyglycoside- polycarboxylic acids can be chosen from C6-C24 alkyl polyglycoside- citrates, C6-C24 alkyl polyglycoside-tartrates and C6-C24 alkyl poly glycoside-sulfo succinates .
When the anionic surfactant(s) are in salt form, it (they) may be chosen from alkali metal salts such as the sodium or potassium salt and preferably the sodium salt, ammonium salts, amine salts and in particular amino alcoho l salts, or alkaline earth metal salts such as the magnesium salts .
Examples of amino alcohol salts that may especially be mentioned include monoethanolamine, diethanolamine and triethanolamine salts, monoisopropanolamine, diisopropanolamine or triisopropanolamine salts, 2-amino-2-methyl- 1 -propanol salts, 2- amino-2-methyl- 1 ,3-propanediol salts and tris(hydroxymethyl)amino methane salts.
Use is preferably made of alkali metal or alkaline earth metal salts and in particular of sodium or magnesium salts.
Use is preferably made, among the anionic surfactants mentioned, of (C6-C24)alkyl sulfates, (C6-C24)alkyl ether sulfates comprising from 2 to 50 ethylene oxide units, in particular in the form of alkali metal, ammonium, aminoalcohol and alkaline earth metal salts, or a mixture of these compounds.
In particular, it is preferable to use (Ci2-C2o)alkyl sulfates, (Ci2-C2o)alkyl ether sulfates comprising from 2 to 20 ethylene oxide units, in particular in the form of alkali metal, ammonium, aminoalcohol and alkaline earth metal salts, or a mixture of these compounds. Better still, it is preferable to use sodium lauryl ether sulfate comprising 2.2 mol of ethylene oxide.
The amphoteric or zwitterionic surfactant(s), which are preferably (a) non-silicone surfactant(s), which can be used in the present invention can in particular be derivatives of secondary or tertiary, optionally quaternized aliphatic amines, in which derivatives the aliphatic group is a linear or branched chain comprising from 8 to 22 carbon atoms, said amine derivatives comprising at least one anionic group, such as, for example, a carboxylate, sulfonate, sulfate, phosphate or phosphonate group. Mention may in particular be made of (C8-C2o)alkyl betaines, sulfobetaines, (C8-C2o)alkylamido(C3- Cs)alkyl betaines and (C8-C2o)alkylamido(C6-C8)alkyl sulfobetaines.
Among the secondary or tertiary, optionally quaternized aliphatic amine derivatives that can be used, as defined above, mention may also be made of the compounds of respective structures (Al) and (A2):
Ra-C(0)-NH-CH2-CH2-N+(Rb)(Rc)-CH2C(0)0-, M+, X" (Al)
in which formula (A l ) :
Ra represents a C 1 0 - C30 alkyl or alkenyl group derived from an acid RaCOOH preferably present in hydrolysed coconut oil, a heptyl, nonyl group or undecyl group;
Rb represents a β-hydroxyethyl group ; and
Rc represents a carboxymethyl group;
M+ represents a cationic counterion derived from an alkali metal or alkaline-earth metal, such as sodium, an ammonium ion or an ion derived from an organic amine, and
X" represents an organic or inorganic anionic counterion, such as that chosen from halides, acetates, phosphates, nitrates, (C i - C4)alkyl sulfates, (C i -C4)alkyl- or (C i -C4)alkylarylsulfonates, in particular methyl sulfate and ethyl sulfate; or alternatively M+ and X" are absent;
Ra ' -C(0)-NH-CH2-CH2-N(B)(B') (A2)
in which formula (A2) :
B represents the group -CH2-CH2-0-X';
B' represents the group -(CH2)zY', with z = 1 or 2;
X* represents the group -CH2-C(0)OH, -CH2-C(0)OZ ' , -CH2- CH2-C(0)OH or -CH2-CH2-C(0)OZ ' , or a hydrogen atom;
Y* represents the group -C(0)OH, -C(0)OZ ' , -CH2-CH(OH)- S O3H or the group -CH2-CH(OH)-S03-Z ' ;
Z' represents a cationic counterion derived from an alkali metal or alkaline-earth metal, such as sodium, an ammonium ion or an ion derived from an organic amine;
Ra' represents a C 1 0 - C30 alkyl or alkenyl group of an acid Ra' - C(0)OH which is preferably present in coconut oil or in hydrolysed linseed oil, or an alkyl group, especially a C 1 7 group , and its iso form, or an unsaturated C 1 7 group .
These compounds are classified in the CTFA dictionary, 5th edition, 1993 , under the names disodium cocoamphodiacetate, disodium lauroamphodiacetate, disodium caprylamphodiacetate, disodium capryloamphodiacetate, disodium cocoamphodipropionate, disodium lauroamphodipropionate, disodium
caprylamphodipropionate, disodium capryloamphodipropionate, lauroamphodipropionic acid and cocoamphodipropionic acid.
By way o f example, mention may be made o f the cocoamphodiacetate sold by the company Rhodia under the trade name Mirano l® C2M Concentrate.
Among the amphoteric or zwitterionic surfactants mentioned above, use is preferably made o f (C 8 - C2o)alkylbetaines such as cocobetaine, and (C8 - C2o)alkylamido(C 3 -C8)alkylbetaines such as cocamidopropylbetaine, and mixtures thereof. More preferably, the amphoteric or zwitterionic surfactant(s) are chosen from cocamidopropyl betaine and coco betaine.
The cationic surfactant(s) which can be used in the composition according to the invention comprise, for example, salts of primary, secondary or tertiary fatty amines, which are optionally polyoxyalkylenated, quaternary ammonium salts and mixtures thereof.
Examples of quaternary ammonium salts that may especially be mentioned include :
- those corresponding to the general formula (A3) below :
N X
(A3)
in which formula (A3) :
Rs to Rn , which may be identical or different, represent a linear or branched aliphatic group comprising from 1 to 30 carbon atoms, or an aromatic group such as aryl or alkylaryl, it being understood that at least one o f the groups Rs to Rn comprises from 8 to 30 carbon atoms and preferably from 12 to 24 carbon atoms; and
X" represents an organic or inorganic anionic counterion, such as that chosen from halides, acetates, phosphates, nitrates, (C i - C4)alkyl sulfates, (C i -C4)alkyl or (C i -C4)alkylaryl sulfonates, in particular methyl sulfate and ethyl sulfate .
The aliphatic groups o f Rs to Rn may also comprise heteroatoms especially such as oxygen, nitrogen, sulfur and halogens .
The aliphatic groups of Rs to Rn are chosen, for example, from C1-C30 alkyl, C1-C30 alkoxy, polyoxy(C2-Ce)alkylene, C1-C30 alkylamide, (Ci2-C22)alkylamido(C2-C6)alkyl, (Ci2-C22)alkylacetate, C1-C30 hydroxyalkyl, X" is an anionic counterion chosen from halides, phosphates, acetates, lactates, (Ci-C4)alkyl sulfates, and (Ci-C4)alkyl- or (Ci-C4)alkylarylsulfonates.
Among the quaternary ammonium salts of formula (A3), preference is given firstly to tetraalkylammonium chlorides, for instance dialkyldimethylammonium or alkyltrimethylammonium chlorides in which the alkyl group contains approximately from 12 to 22 carbon atoms, in particular behenyltrimethylammonium chloride, distearyldimethylammonium chloride, cetyltrimethylammonium chloride, benzyldimethylstearylammonium chloride, or else, secondly, distearoylethylhydroxyethylmethylammonium met ho sulfate, dipalmitoylethylhydroxyethylammonium methosulfate or distearoylethylhydroxyethylammonium methosulfate, or else, lastly, palmitylamidopropyltrimethylammonium chloride or stearamidopropyldimethyl(myristyl acetate)ammonium chloride, sold under the name Ceraphyl® 70 by the company Van Dyk;
(A4)
in which formula (A4):
R12 represents an alkenyl or alkyl group comprising from 8 to 30 carbon atoms, for example tallow fatty acid derivatives;
R represents a hydrogen atom, a C1-C4 alkyl group or an alkenyl or alkyl group comprising from 8 to 30 carbon atoms;
Ri4 represents a C1-C4 alkyl group;
Ri5 represents a hydrogen atom or a C1-C4 alkyl group;
X" represents an organic or inorganic anionic counterion, such as that chosen from halides, phosphates, acetates, lactates, (Ci- C4)alkyl sulfates, (Ci-C4)alkyl or (Ci-C4)alkylaryl sulfonates.
Ri2 and R13 preferably denote a mixture of alkenyl or alkyl groups comprising from 12 to 21 carbon atoms, for example tallow fatty acid derivatives, Ri4 denotes a methyl group, and R15 denotes a hydrogen atom. Such a product is sold, for example, under the name Rewoquat® W 75 by the company Rewo;
- di- or triquaternary ammonium salts, in particular of formula (A5) below:
2+
1
R16— — (CH — N— R '21 2X~
R, R„
(A5)
in which formula (A5):
Ri6 denotes an alkyl group comprising approximately from 16 to 30 carbon atoms, which is optionally hydroxylated and/or interrupted by one or more oxygen atoms;
Ri7 is chosen from hydrogen, an alkyl group comprising from 1 to 4 carbon atoms or a group -(CH2)3-N+(Ri6a)(Ri7a)(Ri8a), X";
Ri6a, Ri7a, Ri8a, Ri8, Ri9, R2o and R21 , which may be identical or different, are chosen from hydrogen and an alkyl group comprising from 1 to 4 carbon atoms; and
X", which may be identical or different, represents an organic or inorganic anionic counterion, such as that chosen from halides, acetates, phosphates, nitrates, (Ci-C4)alkyl sulfates, (Ci-C4)alkyl or (Ci-C4)alkylaryl sulfonates, in particular methyl sulfate and ethyl sulfate.
Such compounds are, for example, Finquat CT-P, provided by Finetex (Quaternium 89), or Finquat CT, provided by Finetex (Quaternium 75);
- quaternary ammonium salts containing one or more ester functions, such as those of formula (A6) below:
in which formula (A6):
R22 is chosen from Ci-C6 alkyl groups and Ci-C6 hydroxyalkyl or dihydroxyalkyl groups,
R23 is chosen from:
O
- the group : -c
linear or branched, saturated or unsaturated C1-C22 hydrocarbon-based groups R27,
- a hydrogen atom,
R25 is chosen from:
linear or branched, saturated or unsaturated Ci-C6 hydrocarbon-based groups R29,
- a hydrogen atom,
R24, R26 and R28, which may be identical or different, are chosen from linear or branched, saturated or unsaturated C7-C21 hydrocarbon-based groups;
r, s and t, which may be identical or different, are integers ranging from 2 to 6,
rl and tl, which may be identical or different, are equal to 0 or 1, with r2+rl=2r and tl+t2=2t,
y is an integer ranging from 1 to 10,
x and z, which may be identical or different, are integers ranging from 0 to 10,
X" represents an organic or inorganic anionic counterion, with the proviso that the sum x + y + z is from 1 to 15, that when x is 0, then R23 denotes R27 and that when z is 0, then R25 denotes R29.
The alkyl groups R22 may be linear or branched, and more particularly linear.
Preferably, R22 denotes a methyl, ethyl, hydroxyethyl or dihydroxypropyl group, and more particularly a methyl or ethyl group .
Advantageously, the sum x + y + z ranges from 1 to 1 0.
When R23 is a hydrocarbon-based group R27 , it may be long and may contain from 12 to 22 carbon atoms, or may be short and may contain from 1 to 3 carbon atoms .
When R25 is a hydrocarbon-based group R29 , it preferably contains 1 to 3 carbon atoms .
Advantageously, R24 , R26 and R28 , which may be identical or different, are chosen from linear or branched, saturated or unsaturated C 1 1 -C2 1 hydrocarbon-based groups, and more particularly from linear or branched, saturated or unsaturated C n -C2 1 alkyl and alkenyl groups .
x and z, which may be identical or different, are preferably 0 or 1 .
y is advantageously equal to 1 .
Preferably, r, s and t, which may be identical or different, equal 2 or 3 , and even more particularly are equal to 2.
The anionic counterion X" is preferably a halide, such as chloride, bromide or iodide; a (C i - C4)alkyl sulfate or a (C i - C4)alkylsulfonate or (C i -C4)alkylarylsulfonate. However, it is possible to use methanesulfonate, phosphate, nitrate, tosylate, an anion derived from an organic acid, such as acetate or lactate, or any other anion that is compatible with the ammonium containing an ester function.
The anionic counterion X" is even more particularly chloride, methyl sulfate or ethyl sulfate.
Use is made more particularly in the composition according to the invention of the ammonium salts of formula (A6) in which:
- R22 denotes a methyl or ethyl group ,
- x and y are equal to 1 ,
- z is equal to 0 or 1 ,
- r, s and t are equal to 2 ,
- R23 is chosen from:
O
p
the group 26
methyl, ethyl or C 1 4 - C22 hydrocarbon-based groups,
a hydrogen atom,
- R25 is chosen from:
O the group R28 ^
a hydrogen atom,
- R24 , R26 and R28 , which may be identical or different, are chosen from linear or branched, saturated or unsaturated C 1 3-C 1 7 hydrocarbon-based groups, and more particularly from linear or branched, saturated or unsaturated C 1 3-C 1 7 alkyl and alkenyl groups .
Advantageously, the hydrocarbon radicals are linear.
Among the compounds o f formula (A6), examples that may be mentioned include salts, especially the chloride or methyl sulfate, of diacyloxyethyldimethyl ammonium,
diacyloxyethylhydroxyethylmethylammonium,
monoacyloxyethyldihydroxyethylmethylammonium,
triacyloxyethylmethylammonium or monoacyloxyethylhydroxyethyldimethylammonium, and mixtures thereo f. The acyl groups preferably contain 14 to 1 8 carbon atoms and are obtained more particularly from a plant oil such as palm oil or sunflower oil. When the compound contains several acyl groups, these groups may be identical or different.
These products are obtained, for example, by direct esterification o f triethano lamine, triisopropanolamine, an alkyldiethano lamine or an alkyldiisopropanolamine, which are optionally oxyalkylenated, with fatty acids or with fatty acid mixtures of plant or animal origin, or by transesterification of the methyl esters thereo f. This esterification is fo llowed by a quaternization by means of an alkylating agent such as an alkyl halide, preferably methyl or ethyl halide, a dialkyl sulfate, preferably dimethyl or diethyl sulfate,
methyl methanesulfonate, methyl /?ara-toluenesulfonate, glycol chlorohydrin or glycerol chlorohydrin.
Such compounds are sold, for example, under the names Dehyquart® by the company Henkel, Stepanquat® by the company Stepan, Noxamium® by the company Ceca or Rewoquat® WE 18 by the company Rewo-Witco.
The composition according to the invention may contain, for example, a mixture of quaternary ammonium salts of monoesters, diesters and triesters with a weight majority of diester salts.
It is also possible to use the ammonium salts containing at least one ester function that are described in patents US-A-4 874 554 and US-A-4 137 180.
Use may be made of behenoylhydroxypropyltrimethylammonium chloride sold by KAO under the name Quatarmin BTC 131.
Preferably, the ammonium salts containing at least one ester function contain two ester functions.
Among the cationic surfactants that may be present in the composition according to the invention, it is more particularly preferred to choose cetyltrimethylammonium, behenyltrimethylammonium and dipalmitoylethylhydroxyethylmethyl- ammonium salts, and mixtures thereof, and more particularly behenyltrimethylammonium chloride, cetyltrimethylammonium chloride, and dipalmitoylethylhydroxyethylammonium methosulfate, and mixtures thereof.
Examples of nonionic surfactants that can be used in the composition used according to the invention are described, for example, in the Handbook of Surfactants by M.R. Porter, published by Blackie & Son (Glasgow and London), 1991, pp. 116-178. They are especially chosen from alcohols, a-diols and (Ci-C2o)alkylphenols, these compounds being polyethoxylated, polypropoxylated or polyglycerolated, and containing at least one fatty chain comprising, for example, from 8 to 18 carbon atoms, it being possible for the number of ethylene oxide and/or propylene oxide groups to especially
range from 2 to 50, and for the number o f glycerol groups to especially range from 2 to 30.
Mention may also be made o f copolymers of ethylene oxide and propylene oxide, optionally oxyethylenated sorbitan fatty acid esters, sucrose fatty acid esters, polyoxyalkylenated fatty acid esters, optionally oxyalkylenated alkyl polyglycosides, alkyl glucoside esters, derivatives o f N-alkylglucamine and of N-acylmethylglucamine, aldobionamides and amine oxides.
The nonionic surfactants are chosen more particularly from mono- or polyoxyalkylenated or mono- or polyglycero lated nonionic surfactants. The oxyalkylene units are more particularly oxyethylene or oxypropylene units, or their combination, preferably oxyethylene units.
Examples o f oxyalkylenated nonionic surfactants that may be mentioned include :
oxyalkylenated (Cs- C 24)alkylphenols;
saturated or unsaturated and linear or branched oxyalkylenated C 8 - C30 alcoho ls;
saturated or unsaturated and linear or branched oxyalkylenated C 8 - C30 amides;
esters of saturated or unsaturated and linear or branched C 8 - C30 acids and of polyethylene glycols;
polyoxyethylenated esters o f saturated or unsaturated and linear or branched C 8 - C30 acids and of sorbito l;
saturated or unsaturated oxyethylenated vegetable oils;
condensates o f ethylene oxide and/or of propylene oxide, inter alia, alone or as mixtures;
oxyethylenated and/or oxypropylenated silicones .
The surfactants contain a number o f mo les o f ethylene oxide and/or of propylene oxide of between 1 and 100, preferably between 2 and 50 and preferably between 2 and 30. Advantageously, the nonionic surfactants do not comprise any oxypropylene units.
In accordance with a preferred embo diment of the invention, the oxyalkylenated nonionic surfactants are chosen from
oxyethylenated C8-C30 alcohols comprising from 1 to 100 mol of ethylene oxide; and polyoxyethylenated esters of saturated or unsaturated and linear or branched C8-C30 acids and of sorbitol comprising from 1 to 100 mol of ethylene oxide.
As examples of monoglycerolated or polyglycerolated nonionic surfactants, monoglycerolated or polyglycerolated C8-C40 alcohols are preferably used.
In particular, the monoglycerolated or polyglycerolated C8-C40 alcohols correspond to the formula (A7) below:
R290-[CH2-CH(CH2OH)-0]m-H (A7)
in which formula (A7):
R29 represents a linear or branched C8-C40 and preferably C8- C30 alkyl or alkenyl radical; and
m represents a number ranging from 1 to 30 and preferably from 1 to 10.
As examples of compounds of formula (A7) that are suitable within the context of the invention, mention may be made of lauryl alcohol containing 4 mol of glycerol (INCI name: Polyglyceryl-4 Lauryl Ether), lauryl alcohol comprising 1.5 mol of glycerol, oleyl alcohol comprising 4 mol of glycerol (INCI name: Polyglyceryl-4 Oleyl Ether), oleyl alcohol comprising 2 mol of glycerol (INCI name: Polyglyceryl-2 Oleyl Ether), cetearyl alcohol comprising 2 mol of glycerol, cetearyl alcohol comprising 6 mol of glycerol, oleocetyl alcohol comprising 6 mol of glycerol, and octadecanol comprising 6 mol of glycerol.
The alcohol of formula (A8) may represent a mixture of alcohols in the same way that the value of m represents a statistical value, which means that, in a commercial product, several species of polyglycerolated fatty alcohols may coexist in the form of a mixture.
Use is more preferably made, among the mono- or polyglycerolated alcohols, of the Cs/Cio alcohol comprising 1 mol of glycerol, the C10/C12 alcohol comprising 1 mol of glycerol and the C12 alcohol comprising 1.5 mol of glycerol.
Preferably, the surfactant used in the composition o f the invention is chosen from nonionic monooxyalkylenated or polyoxyalkylenated and monoglycerolated or polyglycero lated surfactants, more particularly monooxyethylenated or polyoxyethylenated, or monooxypropylenated or po lyoxypropylenated, nonionic surfactants, or a combination thereo f, more particularly monooxyethylenated or polyoxyethylenated.
Preferably, the surfactant(s) are chosen from nonionic surfactants or from anionic surfactants . More particularly, the surfactant(s) present in the composition are chosen from nonionic surfactants .
More preferably still, the nonionic surfactants are chosen from polyoxyethylenated sorbito l esters and polyoxyethylenated fatty alcoho ls, and mixtures thereof.
In the composition of the invention, the amount of surfactant(s) in the composition preferably ranges from 0. 1 % to 50% by weight and better still from 0.5 % to 20% by weight relative to the total weight of the composition.
Basifying agents
As indicated previously, the composition according to the invention comprises one or more basifying agents .
The basifying agent(s) may be mineral or organic or hybrid. For the purposes o f the present invention, the term "mineral compound" means any compound bearing in its structure one or more elements from co lumns 1 to 13 of the Periodic Table o f the Elements other than hydrogen.
According to one particular embodiment of the invention, the mineral basifying agent contains one or more elements from columns 1 and 2 of the Perio dic Table of the Elements other than hydrogen.
In one preferred variant, the mineral basifying agent has the fo llowing structure :
(Z1 x-)m(Z2 y+)n
in which:
Z2 denotes a metal from columns 1 to 13 and preferably 1 or 2 of the Periodic Table of the Elements, such as sodium or potassium;
Zix" denotes an anion chosen from the ions C03 2 , OH", HC03 2", Si03 2", HP04 2", P04 3" and B407 2", and preferably from the ions C03 2 , OH" and Si03 2";
x denotes 1, 2 or 3;
y denotes 1, 2, 3 or 4;
m and n denote, independently of each other, 1, 2, 3 or 4;
with n.y = m.x.
Preferably, the mineral basifying agent corresponds to the following formula (Zix")m(Z2 y+)n, in which Z2 denotes a metal from columns 1 and 2 of the Periodic Table of the Elements; Zix" denotes an anion chosen from the ions C03 2", OH" and Si03 2 , x is 1, y denotes 1 or 2, and m and n denote, independently of each other, 1 or 2 with n.y = m.x.
The mineral basifying agent(s) are preferably chosen from aqueous ammonia, alkali carbonates or bicarbonates such as sodium carbonate, potassium carbonate, sodium bicarbonate, potassium bicarbonate, sodium hydroxide (soda), potassium hydroxide (potash), sodium metasilicate and potassium metasilicate, and a mixture thereof.
More particularly, the basifying agent is a mineral base chosen from alkali metal carbonates.
The organic basifying agent(s) are preferably chosen from organic amines with a pKb at 25°C of less than 12, preferably less than 10 and even more advantageously less than 6. It should be noted that it is the pKb corresponding to the functional group of highest basicity. In addition, the organic amines do not comprise any alkyl or alkenyl fatty chains comprising more than ten carbon atoms.
The organic basifying agent(s) are chosen, for example, from alkanolamines, oxyethylenated and/or oxypropylenated ethylenediamines, amino acids and the compounds of formula (III) below:
Rx Rz
\
N-W-N
Ry Rt (III)
in which formula (III) W is a divalent Ci-C6 alkylene residue optionally substituted with a hydroxyl group or a Ci-C6 alkyl radical; and/or optionally interrupted with one or more heteroatoms such as O, or NRu; Rx, Ry, Rz, Rt and Ru, which may be identical or different, represent a hydrogen atom or a Ci-C6 alkyl, Ci-C6 hydroxyalkyl or Ci- C6 aminoalkyl radical.
Examples of amines of formula (II) that may be mentioned include 1 ,3-diaminopropane, 1 ,3-diamino-2-propanol, spermine and spermidine.
The term "alkanolamine" means an organic amine comprising a primary, secondary or tertiary amine function, and one or more linear or branched Ci-Cs alkyl groups bearing one or more hydroxyl radicals.
Organic amines chosen from alkanolamines, such as mono-, di- or trialkanolamines, comprising from one to three identical or different C1-C4 hydroxyalkyl radicals, are suitable in particular for the implementation of the invention.
Among the compounds of this type, mention may be made of monoethanolamine (MEA), diethanolamine, triethanolamine, monoisopropanolamine, diisopropanolamine, N,N- dimethylethanolamine, 2-amino-2-methyl- 1 -propanol, triisopropanolamine, 2 -amino -2 -methyl- 1 ,3 -propanediol, 3 -amino - 1 ,2- propanediol, 3-dimethylamino- 1 ,2-propanediol and tris(hydroxymethyl)amino methane.
More particularly, the amino acids which can be used are of natural or synthetic origin, in their L, D or racemic form, and comprise at least one acid functional group chosen more particularly from carboxylic acid, sulfonic acid, phosphonic acid or phosphoric acid functional groups. The amino acids can be in the neutral or ionic form.
Mention may in particular be made, as amino acids which can be used in the present invention, of aspartic acid, glutamic acid,
alanine, arginine, ornithine, citrulline, asparagine, carnitine, cysteine, glutamine, glycine, histidine, lysine, iso leucine, leucine, methionine, N-phenylalanine, proline, serine, taurine, threonine, tryptophan, tyrosine and valine.
Advantageously, the amino acids are basic amino acids comprising an additional amine functional group optionally included in a ring or in a ureido functional group .
Such basic amino acids are preferably chosen from those corresponding to formula (IV) below :
NH,
/ 2
R CH2— CH ^
C02H
(IV)
-(CH2)3NH2
-(CH2)2NH2 ; -(CH2)2NHCONH2.
The compounds corresponding to formula (IV) are histidine, lysine, arginine, ornithine and citrulline.
The organic amine may also be chosen from organic amines o f heterocyclic type. Besides histidine that has already been mentioned in the amino acids, mention may in particular be made o f pyridine, piperidine, imidazole, triazo le, tetrazole and benzimidazole.
The organic amine may also be chosen from amino acid dipeptides. As amino acid dipeptides that may be used in the present invention, mention may be made especially o f carnosine, anserine and balenine.
The organic amine may also be chosen from compounds comprising a guanidine function. As amines o f this type that can be used in the present invention, besides arginine, which has already been mentioned as an amino acid, mention may be made especially o f
creatine, creatinine, 1 , 1 -dimethylguanidine, 1 , 1 -diethylguanidine, glycocyamine, metformin, agmatine, N-amidinoalanine, 3 - guanidinopropionic acid, 4-guanidinobutyric acid and 2- ([ amino (imino)methyl] amino) ethane- 1 - sulfonic acid.
Hybrid compounds that may be mentioned include the salts o f the amines mentioned previously with acids such as carbonic acid or hydrochloric acid.
Guanidine carbonate or monoethanolamine hydrochloride may be used in particular.
Preferably, the basifying agent(s) present in the composition of the invention are chosen from alkanolamines, amino acids in neutral or ionic form, in particular basic amino acids, and preferably corresponding to those of formula (IV), and compounds comprising a guanidine function, and mixtures thereof.
Even more preferentially, the basifying agent(s) are chosen from alkano lamines such as monoethanolamine (MEA) .
Advantageously, the composition according to the invention has a content of organic basifying agent(s) ranging from 0.01 % to 30% by weight and preferably from 0. 1 % to 20% by weight relative to the weight of the said composition.
According to a first particular embodiment, the composition according to the invention or else the process according to the invention does not use aqueous ammonia, or a salt thereo f, as basifying agent.
According to a second embodiment, if the composition or the process according to the invention does use aqueous ammonia, or a salt thereo f, as basifying agent, its content should not exceed 0.03 %> by weight (expressed as NH3) and preferably should not exceed 0.01 % by weight, relative to the weight of the composition of the invention.
Preferably, if the composition comprises aqueous ammonia, or a salt thereof, then the amount of basifying agent(s) other than the aqueous ammonia is greater than that of the aqueous ammonia (expressed as NH3) .
Oxidation bases
The composition according to the invention comprises one or more oxidation bases .
The oxidation bases may be chosen from heterocyclic bases and benzenic bases, and mixtures thereof.
By way o f examp le, the oxidation bases are chosen from para- phenylene diamines, bis (phenyl) alky lene diamines, para-aminophenols, ortho-aminopheno ls and heterocyclic bases, and the addition salts thereo f.
Among the para-phenylenediamines, examples that may be mentioned include para-phenylenediamine, para-tolylenediamine, 2- chloro-para-phenylenediamine, 2, 3 -dimethyl-para-pheny lene diamine, 2, 6 -dimethyl-para-pheny lene diamine, 2,6-diethyl-para- phenylenediamine, 2,5 -dimethyl-para-phenylenediamine, N,N- dimethyl-para-phenylenediamine, Ν,Ν-diethyl-para-pheny lene diamine, Ν,Ν-dipropyl-para-pheny lene diamine, 4 -amino -N,N- diethyl- 3 - methylaniline, N,N-bis(P -hydroxy ethyl) -para-phenylenediamine, 4- N,N-bis( -hydroxy ethyl) amino -2 -met hylaniline, 4-N,N-bis( - hy droxy ethyl) amino -2 -chloro aniline, 2- β -hydroxy ethyl-para- phenylenediamine, 2- fluoro -para-phenylenediamine, 2-isopropyl-para- phenylenediamine, N-( -hydroxypropyl)-para-phenylenediamine, 2- hydroxymethyl-para-phenylenediamine, N, N- dimethyl- 3 -methyl-par a- phenylenediamine, N-ethyl-N-( -hydroxy ethyl) -para- phenylenediamine, N-( ,y-dihydroxypropyl)-para-phenylenediamine, N- (4 '-aminophenyl) -para-phenylenediamine, N-phenyl-para- phenylenediamine, 2- -hydroxyethyloxy-para-phenylenediamine, 2-β- acetylaminoethyloxy-para-phenylenediamine, N- (β-methoxy ethyl) - para-phenylenediamine, 4-aminophenylpyrrolidine, 2-thienyl-para- phenylenediamine, 2- -hydroxyethylamino-5 -aminotoluene and 3 - hydroxy- 1 -(4'-aminophenyl)pyrro lidine, and the addition salts thereo f with an acid.
Among the para-phenylenediamines mentioned above, para- phenylenediamine, para-tolylenediamine, 2-isopropyl-para- phenylenediamine, 2- -hydroxyethyl-para-phenylenediamine, 2-β- hydroxyethyloxy-para-phenylenediamine, 2, 6 -dimethyl-par a-
phenylenediamine, 2,6-diethyl-para-phenylenediamine, 2, 3 -dimethyl- par a-phenylenediamine, N, N-bis( - hydroxy ethyl) -par a- phenylenediamine, 2-chloro-para-phenylenediamine and 2-β- acetylaminoethyloxy-para-phenylenediamine, and the addition salts thereo f with an acid, are particularly preferred.
Among the bis(phenyl)alkylenediamines, examples that may be mentioned include N,N'-bis( -hydroxyethyl)-N,N'-bis(4'- aminophenyl)- l ,3 -diaminopropanol, N, N'-bis( -hydroxy ethyl)-N, N'- bis(4'-aminophenyl)ethylenediamine, N,N'-bis(4- aminophenyl)tetramethylenediamine, N, N'-bis( -hydroxy ethyl) -N,N '- bis(4- amino phenyl)tetramethylenediamine, N,N'-bis(4- methylaminophenyl)tetramethylenediamine, N,N'-bis(ethyl)-N,N'- bis(4'-amino-3 '-methylphenyl)ethylenediamine and l , 8-bis(2,5 - diaminophenoxy)-3 ,6-dioxaoctane, and the addition salts thereof.
Among the para-aminophenols, examples that may be mentioned include para-aminophenol, 4-amino-3 -methylpheno l, 4- amino-3 -fluoropheno l, 4-amino-3 -chlorophenol, 4-amino-3 - hydroxymethylpheno l, 4-amino-2-methylpheno l, 4-amino-2- hydroxymethylpheno l, 4-amino-2-methoxymethylpheno l, 4-amino-2- amino methylpheno l, 4-amino-2-( -hydroxyethylaminomethyl)pheno l and 4-amino-2-fluorophenol, and the addition salts thereof with an acid.
Mention may be made, among ortho-aminopheno ls, by way o f example, o f 2-aminophenol, 2-amino-5 -methylpheno l, 2-amino-6- methylpheno l, 5 -acetamido-2-aminopheno l and their addition salts.
Mention may be made, among heterocyclic bases, by way o f example, o f pyridine derivatives, pyrimidine derivatives and pyrazole derivatives.
Among the pyridine derivatives that may be mentioned are the compounds described, for examp le, in patents GB 1 026 978 and GB 1 153 196, for instance 2,5 -diaminopyridine, 2-(4- methoxyphenyl)amino-3 -aminopyridine and 3 ,4-diaminopyridine, and the addition salts thereof.
Other pyridine oxidation bases that are useful in the present invention are the 3-aminopyrazolo[ 1 ,5-a]pyridine oxidation bases or the addition salts thereof described, for example, in patent application FR 2 801 308. Examples that may be mentioned include pyrazolo[ 1 ,5- a]pyrid-3-ylamine, 2-acetylaminopyrazolo[ 1 ,5-a]pyrid-3-ylamine, 2- morpholin-4-ylpyrazolo[ 1 ,5-a]pyrid-3-ylamine, 3-aminopyrazolo[ 1 ,5- a]pyridine-2-carboxylic acid, 2-methoxypyrazolo[ 1 ,5-a]pyrid-3- ylamine, (3-aminopyrazolo[ 1 ,5-a]pyrid-7-yl)methanol, 2-(3- aminopyrazolo[ 1 ,5-a]pyrid-5-yl)ethanol, 2-(3-aminopyrazolo[ 1 ,5- a]pyrid-7-yl)ethanol, (3-aminopyrazolo[ 1 ,5-a]pyrid-2-yl)methanol, 3,6-diaminopyrazolo[ 1 ,5-a]pyridine, 3,4-diaminopyrazolo[ 1 ,5- ajpyridine, pyrazolo[ 1 ,5-a]pyridine-3 ,7-diamine, 7-morpholin-4- ylpyrazolo[l ,5-a]pyrid-3-ylamine, pyrazolo[l ,5-a]pyridine-3,5- diamine, 5-morpholin-4-ylpyrazolo[l,5-a]pyrid-3-ylamine, 2-[(3- aminopyrazolo[ 1 ,5-a]pyrid-5-yl)(2-hydroxyethyl)amino]ethanol, 2-[(3- aminopyrazolo[ 1 ,5-a]pyrid-7-yl)(2-hydroxyethyl)amino]ethanol, 3- aminopyrazolo[ 1 ,5-a]pyridin-5-ol, 3-aminopyrazolo[ 1 ,5-a]pyridin-4- ol, 3-aminopyrazolo[ 1 ,5-a]pyridin-6-ol and 3-aminopyrazolo[ 1 ,5- a]pyridin-7-ol, and the addition salts thereof.
Among the pyrimidine derivatives that may be mentioned are the compounds described, for example, in patents DE 2359399, JP 88- 169571, JP 05-63124 and EP 0 770 375 or patent application WO 96/15765, such as 2,4,5,6-tetraaminopyrimidine, 4-hydroxy-2,5,6- triaminopyrimidine, 2-hydroxy-4,5,6-triaminopyrimidine, 2,4- dihydroxy-5,6-diaminopyrimidine, 2,5,6-triaminopyrimidine and addition salts thereof, and tautomeric forms thereof, when a tautomeric equilibrium exists.
Among the pyrazole derivatives that may be mentioned are the compounds described in patents DE 3843892 and DE 4133957 and patent applications WO 94/08969, WO 94/08970, FR-A-2733 749 and DE 19543988, such as 4,5-diamino- 1 -methylpyrazole, 4,5-diamino- 1 - ( -hydroxyethyl)pyrazole, 3,4-diaminopyrazole, 4,5-diamino- 1 -(4 '- chlorobenzyl)pyrazole, 4,5-diamino- 1 ,3-dimethylpyrazole, 4,5- diamino -3 -methyl- 1 -phenylpyrazole, 4,5-diamino - 1 -methyl- 3-
phenylpyrazole, 4 -amino - 1 ,3-dimethyl-5-hydrazinopyrazole, 1 -benzyl- 4,5-diamino-3-methylpyrazole, 4,5-diamino-3-tert-butyl- 1 - methylpyrazole, 4,5-diamino- 1 -tert-butyl-3-methylpyrazole, 4,5- diamino- 1 -(β -hydro xyethyl)-3-methylpyrazole, 4,5-diamino - 1 - ethy 1-3- methylpyrazole, 4,5-diamino - 1 - ethy 1-3- (4'-methoxyphenyl)pyrazole, 4,5-diamino - 1 - ethy 1-3 -hydro xymethylpyrazole, 4,5-diamino -3- hydroxymethyl- 1 -methylpyrazole, 4,5-diamino-3-hydroxymethyl- 1 - isopropylpyrazole, 4,5-diamino-3-methyl- 1 -isopropylpyrazole, 4- amino-5-(2'-aminoethyl)amino- 1 ,3-dimethylpyrazole, 3,4,5- triaminopyrazole, l-methyl-3,4,5-triaminopyrazole, 3,5-diamino-l- methyl-4-methylaminopyrazole and 3,5-diamino-4-( - hydroxyethyl)amino- 1 -methylpyrazole, and the addition salts thereof. 4,5-Diamino- 1 -( -methoxyethyl)pyrazole may also be used.
Use will preferably be made of a 4,5-diaminopyrazole and even more preferentially of 4,5-diamino- 1 -( -hydroxyethyl)pyrazole and/or a salt thereof.
Mention may also be made, as pyrazole derivatives, of diamino-N,N-dihydropyrazolopyrazolones and in particular those described in application FR-A-2886 136, such as the following compounds and the addition salts thereof: 2,3-diamino-6,7-dihydro- lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 -one, 2-amino-3-ethylamino-6,7- dihydro- lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 - one, 2-amino-3- isopropylamino-6,7-dihydro-lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 -one, 2- amino-3-(pyrrolidin-l-yl)-6,7-dihydro-lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1-one, 4,5-diamino-l,2-dimethyl-l,2-dihydropyrazol-3-one, 4,5- diamino- 1 ,2-diethyl- 1 ,2-dihydropyrazol-3-one, 4,5-diamino - 1 ,2-di(2- hydroxy ethyl) - 1 ,2-dihydropyrazol-3-one, 2-amino-3-(2- hydroxyethyl)amino-6,7-dihydro-lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 -one, 2-amino-3-dimethylamino-6,7-dihydro- lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1-one, 2,3-diamino-5,6,7,8-tetrahydro-lH,6H-pyridazino[l,2- ajpyrazol- 1-one, 4 -amino - 1 ,2-diethyl-5-(pyrrolidin- 1 -yl)- 1 ,2- dihydropyrazol-3-one, 4-amino-5-(3-dimethylaminopyrrolidin- 1 -yl)- 1 ,2-diethyl- 1 ,2-dihydropyrazo 1-3 -one, 2,3-diamino-6-hydroxy-6,7- dihydro-lH,5H-pyrazolo[ 1 ,2-a]pyrazol- 1 -one.
Use will preferably be made of 2,3-Diamino-6,7-dihydro- lH,5H-pyrazolo[l,2-a]pyrazol-l-one and/or a salt thereof.
As heterocyclic bases, use will preferably be made of 4,5- diamino- 1 - (β -hydro xyethyl)pyrazole and/or 2,3-diamino-6,7-dihydro- lH,5H-pyrazolo[l,2-a]pyrazol-l-one and/or a salt thereof.
Couplers of formula (I)
The composition according to the invention also comprises one or more couplers chosen from the couplers of formula (I) below and the addition salts thereof, isomers thereof, tautomers thereof, solvates thereof and dimers thereof:
in which:
s denotes 0 or 1
p denotes 0 or 1
and s+p = 1
Ri, R2, R3, R4, R5, R6, R7 and Rs denote, independently of each other, a hydrogen atom; an amino group; a halogen atom; a hydroxyl group; a Ci-Cs alkoxy group; a C2-C5 acylamino group; a group CHO, a linear, branched or cyclic Ci-Cis alkyl or C2-Cis alkylene group, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen, an aryl group, the alkyl, alkylene or aryl groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic acid, Ci-Cis alkyl ester, carbamate, carboxamide, amino, alkylamino, dialkylamino, carboxyl, alkoxy and halogen atoms, C3-C6 cycloalkyl, 5- to 8-membered heterocycle comprising one or more heteroatoms such as nitrogen, sulfur or oxygen, one or more carbon atoms of the cycloalkyl or heterocyclic groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic
acid, C 1 - C 1 8 alkyl ester, carbamate, carboxamide, amino, alkylamino , dialkylamino, carboxyl, alkoxy and halogen atoms,
the groups R7 and Rs may form, with the atom that bears them, a ring optionally comprising one or more heteroatoms, this ring possibly being substituted with a C i -C6 alkyl group or a hydroxyl group, or possibly being fused with one or more other rings optionally bearing one or more heteroatoms, one or more carbon atoms o f the ring(s) possibly being replaced with one or more carbonyl groups .
R7 may also denote a radical derived from a perimidinyl group of formula (I), thus leading to a dimer; the two perimidinyl groups are then preferably linked via the carbon atoms in position 2,
or alternatively R7 may denote a linear or branched C 1 - C 1 0 alkylene chain, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen and/or optionally comprising a ring, or alternatively a saturated or unsaturated ring, optionally comprising one or more heteroatoms; a chain comprising several and preferably two saturated or unsaturated rings linked together via a covalent bond or via a heteroatom, the chains or the ring possibly being substituted with a hydroxyl group or with a perimidinyl radical o f formula (I) preferably via the carbon atom in position 2,
when R4 is in position 7 and R5 is in position 6, then they can form, together with the carbon atoms that bear them, a 5 - to 8- membered ring optionally comprising one or more heteroatoms .
Preferably the couplers o f formula (I) are chosen from the fo llowing compounds :
Compound 7 Compound 8 spiro[2H-indene-2,2'(3*H)- spiro[acenaphthylene- [lHJperimidine] 1,3-dihydro l(2H),2'(3*H)-[lH]perimi
2-one
Compound 9
Compound 10
2-(2,3-dihydro-lH-perimidin-2- yl)-3-methylphenol
Compound 11 Compound 12
2-(2,3-dihydro-lH-perimidin-2- 3-(2,3-dihydro-lH-perimidin-2- yl)-6-methoxy-4-(2-propen- 1 - yl)-4-hydroxybenzoic acid yl)phenol
pyrro - -y - -perm ne t eny - -perm ne
Compound 37
4-ethylspiro[cyclohexane- l,2 3^-[lH]perimidine] Compound 38
2-[4-(2-furanyl)phenyl]-2, dihydro- lH-perimidine
2,3-dihydro-2-(lH-imidazol-
Compound 39
5-yl)- lH-perimidine
2-[4-(l,l- dimethylethyl)cyclohexyl]- 2,3-dihydro-lH-perimidine
Compound 41
2-(2, 3 -dihydro -1 H-perimidin- Compound 42
Compound 49 Compound 50
2-cy clohexy 1-2,3 -dihydro- spiro[lH-perimidine-2(3H),2'- lH-perimidine tricyclo[3.3.1.13 ,7]decane]
2,3-dihydro-2,2,4,8-
Compound 51
tetramethyl- 1 H-perimi
4-(2,3-dihydro-lH- perimidin-2-yl)-2-iodo-6
methoxyphenol
Compound 54
perimidine
3-(2,3-dihydro-2-methyl- lH-perimidin-2-yl)phenol Compound 134
4-(2,3-dihydro- lH-perimidin-2- yl)-2-methoxyphenol
Compound 136
Compound 135
3-(2,3-dihydro-2-methyl-lH- 2-(2,3-dihydro-lH- perimidin-2-yl)-6-methyl-2H- perimidin-2-yl)-6- pyran-4-ol
methoxyphenol
II
Compound 138
Compound 137
2,3-dihydro-lH-perimidine- 2,3-dihydro-2-methyl-2-pentyl- 2-carboxylic acid lH-perimidine
2 -met hylspiro [cy clohexane- spiro[cyclobutane-l ,2'(3Ή)-
1.2'(3^-[lH]perimidine] [lHJperimidine]
NH 2 NH 2 H2 H2
S03H
Compound 360
4,5 -diamino- 1 - Compound 361
naphthalenesulfonic acid 1 ,2-dihydro-5 ,6- acenaphthylenedi amine
Compound 362
1 , 8-naphthalenediamine and also the addition salts thereo f, isomers thereof, tautomers thereo f and so lvates thereof.
According to one embodiment of the invention, p denotes 1 .
According to another embodiment of the invention, p denotes 0.
In a first variant of the invention, in formula (I), R3 and R6 represent a hydrogen atom.
In a second variant of the invention, in formula (I), at least one of the groups Ri and R2 represent a hydrogen atom.
In a third variant o f the invention, in formula (I), R4 and R5 represent a hydrogen atom.
Additional couplers
The composition o f the invention may also comprise one or more additional couplers other than the couplers that are usually used, of formula (I) as described previously.
Among these couplers, mention may be made especially o f meta-phenylenediamines, meta-aminopheno ls, meta-dipheno ls,
naphthalene-based couplers and heterocyclic couplers, and also the addition salts thereof.
Examples that may be mentioned include 1,3- dihydroxybenzene, 1 ,3-dihydroxy-2-methylbenzene, 4-chloro- 1 ,3- dihydroxybenzene, 2,4-diamino- 1 -(P-hydroxyethyloxy)benzene, 2- amino -4 -(β- hydroxy ethy lamino) - 1 -methoxybenzene, 1,3- diaminobenzene, 1 ,3-bis(2,4-diaminophenoxy)propane, 3- ureidoaniline, 3-ureido- 1 -dimethylaminobenzene, sesamol, 1-β- hydroxyethylamino-3,4-methylenedioxybenzene, a-naphthol, 2- methyl- 1 -naphthol, 6-hydroxyindole, 4-hydroxyindole, 4-hydroxy-N- methylindole, 2-amino-3-hydroxypyridine, 6- hydroxybenzomorpholine, 3,5-diamino-2,6-dimethoxypyridine, 1-N- (P-hydroxyethyl)amino-3,4-methylenedioxybenzene, 2,6-bis(P- hydroxyethylamino)toluene, 6-hydroxyindoline, 2,6-dihydroxy-4- methylpyridine, 1 -H-3-methylpyrazol-5-one, l-phenyl-3- methylpyrazol-5-one, 2,6-dimethylpyrazolo[ 1 ,5-b]- 1 ,2,4-triazole, 2,6- dimethyl[3,2-c]-l,2,4-triazole and 6-methylpyrazolo[ 1 ,5- ajbenzimidazole, the addition salts thereof with an acid, and mixtures thereof.
In general, the addition salts of the oxidation bases and couplers that can be used within the context of the invention are especially chosen from the addition salts with an acid such as the hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, phosphates and acetates.
The oxidation base(s) each advantageously represent from
0.0001% to 10% by weight relative to the total weight of the composition, and preferably from 0.005% to 5% by weight relative to the total weight of the composition.
The coupler(s) each advantageously represent from 0.0001%) to 10% by weight relative to the total weight of the composition, and preferably from 0.005% to 5% by weight relative to the total weight of the composition.
Additional dyes
The composition according to the invention may optionally comprise one or more synthetic or natural direct dyes, chosen from ionic or nonionic species, preferably cationic or nonionic species .
Examples o f suitable synthetic direct dyes that may be mentioned include azo direct dyes; (poly)methine dyes such as cyanins, hemicyanins and styryls; carbonyl dyes; azine dyes; nitro(hetero)aryl dyes; tri(hetero)arylmethane dyes; porphyrin dyes; and phthalocyanin dyes, alone or as mixtures.
Mention may be made, among the natural direct dyes which can be used according to the invention, o f lawsone, juglone, alizarin, purpurin, carminic acid, kermesic acid, purpurogallin, protocatechaldehyde, indigo, isatin, curcumin, spinulo sin, apigenidin and orceins. Use may also be made of extracts or decoctions comprising these natural dyes and in particular henna-based poultices or extracts .
When they are present, the direct dye(s) more particularly represent from 0.00 1 % to 10% by weight and preferably from 0.005 % to 5 % by weight of the total weight of the composition.
The composition according to the invention may be an anhydrous or aqueous composition. It is preferably aqueous. The term "aqueous composition" is understood to mean a composition comprising more than 5 % by weight o f water, preferably more than 10% by weight of water and more advantageously still more than 20% by weight of water.
Solvents
The cosmetic composition according to the invention may optionally comprise one or more organic solvents.
Examples o f organic so lvents that may be mentioned include linear or branched C 2 - C 4 alkano ls, such as ethanol and isopropano l; glycero l; polyo ls and polyo l ethers, for instance 2-butoxyethano l, propylene glyco l, dipropylene glyco l, propylene glyco l monomethyl ether, diethylene glyco l monomethyl ether and monoethyl ether, and also aromatic alcoho ls, for instance benzyl alcohol or phenoxyethano l, and mixtures thereof.
The organic so lvent(s), if they are present, represent a content usually ranging from 1 % to 40% by weight and preferably from 5 % to 30% by weight relative to the weight of the composition.
Other additives
The composition according to the invention may also contain various adjuvants conventionally used in hair dye compositions, such as anionic, cationic, nonionic, amphoteric or zwitterionic polymers or mixtures thereo f; mineral thickeners, and in particular fillers such as clays or talc; organic thickeners with, in particular, anionic, cationic, nonionic and amphoteric polymeric associative thickeners other than the polymers mentioned previously; antioxidants; penetrants; sequestrants; fragrances; dispersants; film-forming agents; ceramides; preserving agents; opacifiers .
The above adjuvants are generally present in an amount for each o f them o f between 0.01 % and 20% by weight relative to the weight of the composition.
According to one embodiment o f the invention, the composition may also comprise one or more mineral or organic thickeners .
The mineral thickeners may be chosen from organophilic clays and mineral fillers .
The organophilic clay may be chosen from montmoriUonite, bentonite, hectorite, attapulgite and sepiolite, and mixtures thereof.
The clay is preferably a bentonite or a hectorite .
These clays can be modified with a chemical compound chosen from quaternary ammoniums, tertiary amines, amine acetates, imidazo lines, amine soaps, fatty sulfates, alkylarylsulfonates and amine oxides, and mixtures thereof.
Organophilic clays that may be mentioned include quaternium- 1 8 bentonites, such as those so ld under the names Bentone 3 , Bentone
38 and Bentone 38V by Rheox, Tixogel VP by United Catalyst and
Claytone 34, Claytone 40 and Claytone XL by Southern Clay; stearalkonium bentonites, such as those sold under the names Bentone
27 by Rheox, Tixogel LG by United Catalyst and Claytone AF and
Claytone APA by Southern Clay; quaternium- 1 8/benzalkonium bentonites, such as those so ld under the names Claytone HT and Claytone PS by Southern Clay; quaternium- 1 8 hectorites, such as those so ld under the names Bentone Gel DOA, Bentone Gel EC05 , Bentone Gel EUG, Bentone Gel IPP, Bentone Gel ISD, Bentone Gel S S71 , Bentone Gel VS 8 and Bentone Gel VS38 by Rheox, and Simagel M and Simagel SI 345 by Biophil.
Preferably, the mineral thickeners are chosen from inorganic fillers, in particular kao linite.
The organic thickeners may be chosen from associative or non- associative thickening polymers, in particular associative thickening polymers.
According to the invention, the term "associative thickening polymer" means a thickening polymer comprising at least one C 8 - C 30 fatty chain and at least one hydrophilic unit.
Preferably, the associative thickening polymers are chosen from polyurethane polyethers comprising in their chain both hydrophilic blo cks usually o f po lyoxyethylenated nature and hydrophobic blo cks, which may be aliphatic sequences alone and/or cycloaliphatic and/or aromatic sequences,
Such polyurethane polyethers are so ld especially by the company Rohm & Haas under the names Aculyn 46® and Aculyn 44® [Aculyn 46® is a po lycondensate of polyethylene glyco l containing 150 or 1 80 mo l o f ethylene oxide, of stearyl alcoho l and o f methylenebis(4-cyclohexyl isocyanate) (SMDI), at 15 % by weight in a matrix o f maltodextrin (4%) and water (8 1 %); Aculyn 44® is a polycondensate of polyethylene glyco l containing 150 or 1 80 mo l o f ethylene oxide, o f decyl alcoho l and of methylenebis(4-cyclohexyl isocyanate) (SMDI), at 35 % by weight in a mixture of propylene glyco l (39%) and water (26%)] .
When they are present, the thickeners represent from 1 % to 30% by weight relative to the weight of the composition.
Chemical oxidizing agents
The composition according to the invention may comprise one or more chemical oxidizing agents .
More particularly, the chemical oxidizing agent(s) are chosen from hydrogen peroxide, urea peroxide, alkali metal bromates or ferricyanides, peroxygenated salts, for instance persulfates, perborates, peracids and precursors thereof, and percarbonates o f alkali metals or alkaline-earth metals, and peracids and precursors thereo f.
One or more redox enzymes such as laccases, peroxidases and 2-electron oxidoreductases (such as uricase), optionally in the presence o f the respective donor or cofactor thereof, may also be used as oxidizing agent.
This oxidizing agent advantageously consists o f hydrogen peroxide, in particular in aqueous solution (aqueous hydrogen peroxide so lution), the concentration of which may vary, more particularly from 0. 1 % to 50% by weight, even more preferentially from 0.5 % to 20% by weight and better still from 1 % to 15 % by weight relative to the composition.
Processes according to the invention
The composition used in the process according to the invention corresponds to the composition as defined previously and that is free of chemical oxidizing agent; this composition also being used in the presence of at least one chemical oxidizing agent.
The process according to the invention consists in applying to the said fibres a composition according to the invention, in the presence of at least one chemical oxidizing agent.
The composition used in the process according to the invention (i. e. used in the presence o f at least one chemical oxidizing agent) is applied to wet or dry keratin fibres.
It is usually left in place on the fibres for a time generally o f from 1 minute to 1 hour and preferably from 5 minutes to 30 minutes .
The temperature during the dyeing process is conventionally between room temperature (between 15 °C and 25 ° C) and 80°C and preferably between room temperature and 60°C .
On conclusion o f the treatment, the human keratinous fibres are advantageously rinsed with water. They can optionally be washed with a shampoo, fo llowed by rinsing with water, before being dried or left to dry.
The composition applied in the process according to the invention (i. e. used in the presence o f at least one chemical o xidizing agent) is generally prepared by mixing at least two compositions, preferably two or three compositions .
In a first variant o f the invention, the composition applied in the process according to the invention (thus used with at least one chemical oxidizing agent) results from the mixing o f two compositions.
In particular, a composition (A) comprising at least one oxidation base, at least one coupler chosen from the couplers o f formula (I) and at least one basifying agent is mixed with a composition (B) comprising at least one chemical oxidizing agent; at least one of the compositions (A) and (B) comprising at least one fatty substance, and at least one of the compositions (A) and (B) comprising at least one surfactant, the fatty substance content o f the composition according to the invention, resulting from the mixing o f compositions (A) and (B), comprising at least 25 % by weight of fatty substance.
At least one of the compositions (A) and (B) is advantageously aqueous.
Preferably, composition (A) is aqueous. Preferably, composition (B) is also aqueous .
In this variant, composition (A) preferably comprises at least 50% by weight of fatty substances, and more preferably still at least 50% by weight of fatty substances that are liquid at room temperature (25 °C), relative to the weight of this composition (A) .
Preferably, composition (A) is a direct emulsion (oil-in-water:
O/W) or an inverse emulsion (water-in-oil : W/O), and preferably a direct emulsion (O/W) .
In this variant, compositions (A) and (B) are preferably mixed together in a weight ratio (A)/(B) ranging from 0.2 to 10 and better still from 0.5 to 2.
In a second variant of the invention, the composition used in the process according to the invention (thus in the presence of at least one chemical oxidizing agent) results from the mixing of three compositions. In particular, the three compositions are aqueous or alternatively at least one of them is anhydrous.
More particularly, within the meaning of the invention, the term "anhydrous cosmetic composition" is understood to mean a cosmetic composition exhibiting a water content of less than 5% by weight, preferably of less than 2% by weight and more preferably still of less than 1% by weight, relative to the weight of the said composition. It should be noted that the water present in the composition is more particularly "bound water", such as water of crystallization in salts, or traces of water absorbed by the raw materials used in the preparation of the compositions according to the invention.
Preferably, use is made of two aqueous compositions (Β') and (C) and one anhydrous composition (Α').
The anhydrous composition (Α') then preferably comprises at least one fatty substance, and more preferentially at least one liquid fatty substance.
Composition (Β') then preferably comprises at least one oxidation base and at least one coupler chosen from the couplers of formula (I).
Composition (C) then preferably comprises at least one chemical oxidizing agent.
According to this preferred embodiment of the second variant, the basifying agent(s) are included in compositions (Α') and/or (Β') and preferably only in composition (Β'). As regards the surfactant(s), they are included in at least one of compositions (Α'), (Β') and (C).
According to this preferred embodiment, the composition according to the invention, i.e. the composition derived from the
mixing of the three compositions (Α'), (Β') and (C), has a fatty substance content of at least 25% by weight relative to the weight of the composition derived from the mixing of the abovementioned three compositions.
In this variant, compositions (Α'), (Β') and (C) are preferably mixed together in a weight ratio [(A')+(B')]/(C) ranging from 0.2 to 10 and more particularly from 0.5 to 2 and in a weight ratio (Α')/(Β') ranging from 0.5 to 10 and preferably from 1 to 5.
Devices
Finally, the invention relates to a first multi-compartment device comprising a first compartment containing composition (A) as described above and at least a second compartment containing composition (B) as described above; the compositions (A) and (B) of the compartments being intended to be mixed together before application to give a composition according to the invention; the amount of fatty substance of which represents at least 25% by weight relative to the weight of the formulation resulting from the mixing of compositions (A) and (B).
The invention also relates to a second multi-compartment device comprising a first compartment containing composition (Α') as described above and a second compartment containing a cosmetic composition (Β') as described above and at least a third compartment comprising composition (C) as described above, the compositions of the compartments being intended to be mixed together before application to give the composition according to the invention; the amount of fatty substance in the composition representing at least 25% by weight relative to the weight of the composition of the invention, i.e. resulting from the mixing of compositions (Α'), (Β') and (C).
The examples that follow serve to illustrate the invention without, however, being limiting in nature.
EXAMPLE
The following compositions are prepared in which the amounts are expressed in grams of materials in their given state.
Composition (B)
Propylene glycol 6.2
Ethyl alcohol 8.25
Hexylene glycol (2-methyl-2,4- pentanediol) 3
Dipropylene glycol 3
Monoethanolamine 14.5
Sodium metabisulfite 0.7
Vitamin C: L-ascorbic acid 0.25
Diethylenetriaminepentaacetic acid,
pentasodium salt, as a 40% aqueous
solution 1
Hydroxyethylcellulose (Natrosol 250
HHR, Aqualon) 3.5
2xl0"2
Oxidation base mol
Coupler of formula (I) 2xl0"2 mol
qs
Water 100 g
Com osition (C)
At the time of use, the following are mixed (by weight):
-10 parts of composition A
-4 parts of composition B
-16 parts of composition C
Each of the mixtures is then applied to locks of natural hair containing 90% white hairs.
The "mixture/lock" bath ratio is respectively 10/1 (g/g).
The leave-on time is 30 minutes at 27°C. After this time, the locks are rinsed and then washed with Elseve multivitamin shampoo, and dried.
Example Oxidation base Coupler of formula Dyeing
(I) result
1 4-(3- 1,8- Bluish
Aminopyrazolo[ 1 ,5- Naphthalene diamine grey a]pyridin-2-yl)-l,l- (compound 362)
dimethylpiperazin-
1-ium chloride
hydrochloride
2 4-(3- 2,3-Dihydro-2- Violet
Aminopyrazolo[ 1 ,5- methyl-lH- a]pyridin-2-yl)-l,l- perimidine-2- dimethylpiperazin- methanol
1-ium chloride (compound 109)
hydrochloride
3 p-Phenylenediamine 2,3-Dihydro-2- Violet- methyl-lH- brown perimidine-2- methanol
(compound 109)
4 4-(3- 1H-Perimidine- Violet
Aminopyrazolo[ 1 ,5- 2,2(3H)-dimethanol a]pyridin-2-yl)-l,l- (compound 108)
dimethylpiperazin- 1-ium chloride
hydrochloride
5 2-[(3- 1H-Perimidine- Red-
Aminopyrazolo[ 1 ,5- 2,2(3H)-dimethanol brown a]pyridin-2- (compound 108)
yl)oxy]ethanol
hydrochloride
6 p-Phenylenediamine 1H-Perimidine- Violet- 2,2(3H)-dimethanol brown (compound 108)
All the shades obtained are strong.
Claims
1. Composition for dyeing keratin fibres, in particular human keratin fibres such as the hair, comprising:
- at least 25% by weight, relative to the total weight of the composition, of one or more fatty substances,
- one or more surfactants,
- one or more basifying agents,
- one or more oxidation bases, and
- one or more couplers of formula (I) below, addition salts thereof, isomers thereof, tautomers thereof, solvates thereof and dimers thereof:
s denotes 0 or 1
p denotes 0 or 1
and s+p = 1
Ri, R2, R3, R4, R5, R6, R7 and Rs denote, independently of each other, a hydrogen atom; an amino group; a halogen atom; a hydroxyl group; a Ci-Cs alkoxy group; a C2-C5 acylamino group; a group CHO, a linear, branched or cyclic Ci-Cis alkyl or C2-Cis alkylene group, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen, an aryl group, the alkyl, alkylene or aryl groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic acid, Ci-Cis alkyl ester, carbamate, carboxamide, amino, alkylamino, dialkylamino, carboxyl, alkoxy and halogen atoms, C3-C6 cycloalkyl,
5 - to 8-membered heterocycle comprising one or more heteroatoms such as nitrogen, sulfur or oxygen, one or more carbon atoms o f the cycloalkyl or heterocyclic groups possibly being substituted with one or more groups chosen from hydroxyl, nitro, nitrile, cyano, carboxylic acid, C 1 - C 1 8 alkyl ester, carbamate, carboxamide, amino, alkylamino , dialkylamino, carboxyl, alkoxy and halogen atoms,
the groups R7 and Rs may form, with the atom that bears them, a ring optionally comprising one or more heteroatoms, this ring possibly being substituted with a C i -C6 alkyl group or a hydroxyl group, or possibly being fused with one or more other rings optionally bearing one or more heteroatoms, one or more carbon atoms o f the ring(s) possibly being replaced with one or more carbonyl groups .
R7 may also denote a radical derived from a perimidinyl group of formula (I), thus leading to a dimer; the two perimidinyl groups are then preferably linked via the carbon atoms in position 2,
or alternatively R7 may denote a linear or branched C 1 - C 1 0 alkylene chain, one or more carbon atoms possibly being replaced with one or more heteroatoms such as oxygen, sulfur or nitrogen and/or optionally comprising a ring, or alternatively a saturated or unsaturated ring, optionally comprising one or more heteroatoms; a chain comprising several and preferably two saturated or unsaturated rings linked together via a covalent bond or via a heteroatom, the chains or the ring possibly being substituted with a hydroxyl group or with a perimidinyl radical o f formula (I) preferably via the carbon atom in position 2,
when R4 is in position 7 and R5 is in position 6, then they can form, together with the carbon atoms that bear them, a 5 - to 8- membered ring optionally comprising one or more heteroatoms .
2. Composition according to the preceding claim, characterized in that the fatty substance(s) are chosen from compounds that are liquid or pasty at room temperature and at atmospheric pressure.
3. Composition according to any o f the preceding claims, characterized in that the fatty substance(s) are chosen from C6 - C i 6 hydrocarbons, hydrocarbons containing more than 1 6 carbon atoms,
non-silicone oils o f animal origin, plant oils o f triglyceride type, synthetic triglycerides, fluoro oils, fatty alcoho ls, esters o f fatty acids and/or of fatty alcoho ls other than triglycerides, and plant waxes, non- silicone waxes and silicones, and a mixture thereof.
4. Composition according to any one o f the preceding claims, characterized in that the fatty substance(s) are chosen from liquid petrolatum, polydecenes, fatty alcohols, and liquid esters of fatty acids or of fatty alcohols, and mixtures thereof.
5. Composition according to any one o f the preceding claims, characterized in that the concentration of fatty substances ranges from
25 % to 80% by weight, preferably from 30% to 70%> by weight and more advantageously still from 30% to 60% by weight relative to the total weight of the composition.
6. Composition according to any one o f the preceding claims, characterized in that the surfactant(s) are chosen from nonionic surfactants, in particular monooxyalkylenated or polyoxyalkylenated and monoglycerolated or polyglycerolated nonionic surfactants .
7. Composition according to any one o f the preceding claims, characterized in that the coupler(s) o f formula (I) are chosen from the fo llowing compounds :
Compound 7 Compound 8 spiro[2H-indene-2,2'(3*H)- spiro[acenaphthylene- [lHJperimidine] 1,3-dihydro l(2H),2'(3*H)-[lH]perimi
2-one
Compound 9
Compound 10
2-(2,3-dihydro-lH-perimidin-2- yl)-3-methylphenol
Compound 11 Compound 12
2-(2,3-dihydro-lH-perimidin-2- 3-(2,3-dihydro-lH-perimic yl)-6-methoxy-4-(2-propen- 1 - yl)-4-hydroxybenzoic acid yl)phenol
Compound 25
2-(2-furanyl)-2,3-dihydro-2-methyl- Compound 26
lH-perimidine 2,3-dihydro-2-methyl-2-(2- pyridinyl)- lH-perimidine
4-(2,3-dihydro-lH-perimidin-2-yl)-N-
[[4-(2,3-dihydro-lH-perimidin-2- Compound 28
yl)phenyl]methyl]benzenemethanamine l-(2,3-dihydro-2-methyl- lH-perimidin-2-yl)-2- propanone
2,2-dihexyl-2,3-dihydro-lH- perimidine Compound 30
Compound 37
4-ethylspiro[cyclohexane- l,2 3^-[lH]perimidine] Compound 38
2-[4-(2-furanyl)phenyl]-2, dihydro- lH-perimidine
2,3-dihydro-2-(lH-imidazol-
Compound 39
5-yl)- lH-perimidine
2-[4-(l,l- dimethylethyl)cyclohexyl]- 2,3-dihydro-lH-perimidine
Compound 41
2-(2, 3 -dihydro -1 H-perimidin- Compound 42
y ro- -per m ne
Compound 49 Compound 50
2-cy clohexy 1-2,3 -dihydro- spiro[lH-perimidine-2(3H),2'- lH-perimidine tricyclo[3.3.1.13,7]decane]
2,3-dihydro-2,2,4,8-
Compound 51
tetramethyl- 1 H-perimi
4-(2,3-dihydro-lH- perimidin-2-yl)-2-iodo-6
methoxyphenol
Compound 54
perimidine
3-(2,3-dihydro-2-methyl- lH-perimidin-2-yl)phenol Compound 134
4-(2,3-dihydro- lH-perimidin-2- yl)-2-methoxyphenol
Compound 136
Compound 135
3-(2,3-dihydro-2-methyl-lH- 2-(2,3-dihydro-lH- perimidin-2-yl)-6-methyl-2H- perimidin-2-yl)-6- pyran-4-ol
methoxyphenol
II
Compound 138
Compound 137
2,3-dihydro-lH-perimidine- 2,3-dihydro-2-methyl-2-pentyl- 2-carboxylic acid lH-perimidine
2 -met hylspiro [cy clohexane- spiro[cyclobutane-l ,2'(3Ή)-
1.2'(3^-[lH]perimidine] [lHJperimidine]
NH 2 NH 2 H2 H2
S03H
Compound 360
4,5 -diamino- 1 - Compound 361
naphthalenesulfonic acid 1 ,2-dihydro-5 ,6- acenaphthylenediamine
Compound 362
1 , 8-naphthalenediamine
8. Composition according to any one o f the preceding claims, characterized in that p denotes 1 .
9. Composition according to any one o f the preceding claims, characterized in that p denotes 0.
10. Composition according to any one o f the preceding claims, characterized in that the oxidation base(s) are chosen from para- phenylene diamines, bis (phenyl) alky lene diamines, para-aminophenols, ortho-aminopheno ls and heterocyclic bases, and the addition salts thereo f.
1 1 . Composition according to any one o f the preceding claims, characterized in that the basifying agent is an organic amine with a pKb o f less than 12 and preferably less than 10.
12. Composition according to any one o f the preceding claims, characterized in that the basifying agent is an organic amine chosen from alkanolamines, basic amino acids, compounds comprising a guanidine function, and mixtures thereof.
13. Composition according to any one o f the preceding claims, characterized in that the basifying agent is an alkano lamine, in particular monoethanolamine.
14. Composition according to any one of Claims 1 to 8 , characterized in that the basifying agent is a mineral base chosen from aqueous ammonia, sodium carbonate, potassium carbonate, sodium hydroxide, potassium hydroxide, sodium metasilicate and potassium metasilicate, and a mixture thereof.
15. Composition according to the preceding claim, characterized in that the basifying agent is a mineral base chosen from alkaline carbonates .
16. Composition according to any one o f the preceding claims, characterized in that it comprises at least one chemical oxidizing agent, preferentially hydrogen peroxide.
17. Process for dyeing keratin fibres, in particular human keratin fibres, which consists in applying to the said fibres a composition according to any one of Claims 1 to 15 , in the presence o f at least one chemical oxidizing agent.
1 8. Process according to the preceding claim, characterized in that the composition results from the mixing o f two compositions; preferably o f a composition (A) comprising at least one oxidation base, at least one coupler of formula (I) and at least one basifying agent and o f a composition (B) comprising at least one chemical oxidizing agent, at least one o f the compositions (A) and (B) comprising at least one fatty substance, and at least one o f the compositions (A) and (B) comprising at least one surfactant; the fatty substance content of the composition resulting from the mixing o f compositions (A) and (B) representing at least 25 % by weight relative to the total weight.
19. Process according to Claim 17, characterized in that the composition results from the mixing o f three compositions, preferably of two aqueous compositions (Β ' ) and (C) and o f an anhydrous composition (Α' ), the anhydrous composition (Α' ) comprising at least one fatty substance, the composition (Β ') comprising at least one
oxidation base and at least one coupler chosen from the couplers of formula (I), the composition (C) comprising at least one chemical oxidizing agent, one or more basifying agents being included in the compositions (Α') and/or (Β') and preferably only in the composition (Β')> one or more surfactants being included in at least one of the compositions (Α'), (Β') and (C), the fatty substance content of the composition resulting from the mixing of the three compositions (Α'), (Β') and (C) representing at least 25% by weight relative to the total weight.
20. Multi-compartment device comprising a first compartment containing composition (A) as described in Claim 18 and at least a second compartment containing composition (B) as described in Claim 18, the compositions of the compartments being intended to be mixed together before application, on condition that the amount of fatty substance represents at least 25% by weight relative to the weight of the composition resulting from the mixing of (A) and (B)
21. Multi-compartment device comprising a first compartment containing composition (Α') as described in Claim 19, and a second compartment containing a cosmetic composition (Β') as described in Claim 19 and at least a third compartment comprising composition (C) as described in Claim 19, the compositions of the compartments being intended to be mixed together before application, on condition that the amount of fatty substance represents at least 25% by weight relative to the weight of the composition derived from the mixing of (Α'), (Β') and (C).
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR1161571A FR2983714A1 (en) | 2011-12-13 | 2011-12-13 | OXIDATION COLORING COMPOSITION COMPRISING A PARTICULAR COUPLER IN A BODY-RICH MEDIUM, METHODS AND APPROPRIATE DEVICE |
| FR1161571 | 2011-12-13 | ||
| US201261585007P | 2012-01-10 | 2012-01-10 | |
| US61/585,007 | 2012-01-10 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2013087631A2 true WO2013087631A2 (en) | 2013-06-20 |
| WO2013087631A3 WO2013087631A3 (en) | 2015-01-22 |
Family
ID=45688755
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2012/075094 Ceased WO2013087631A2 (en) | 2011-12-13 | 2012-12-11 | Oxidation dye composition comprising a particular coupler in a medium rich in fatty substances, and processes and device suitable therefor |
Country Status (2)
| Country | Link |
|---|---|
| FR (1) | FR2983714A1 (en) |
| WO (1) | WO2013087631A2 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8884012B1 (en) * | 2013-08-26 | 2014-11-11 | Xerox Corporation | Dye compound and method of making the compound |
| CN104418811A (en) * | 2013-08-20 | 2015-03-18 | 江南大学 | 2,3-dihydronaphthalene-embedded m-diazabenzene analogue, synthetic method, drug composition and application thereof |
| US9193869B2 (en) | 2013-08-26 | 2015-11-24 | Xerox Corporation | Dye compounds, method of making the compounds and ink composition employing the compounds |
| CN104739836B (en) * | 2014-09-30 | 2017-03-29 | 三峡大学 | A kind of micromolecular inhibitor and its application |
| US9738811B2 (en) | 2013-08-26 | 2017-08-22 | Xerox Corporation | Phase change inks containing wax-soluble near-infrared dyes |
| US20170314007A1 (en) * | 2014-09-30 | 2017-11-02 | China Three Gorges University | Medicament design pocket of ornithine decarboxylase and application of medicament design pocket |
| CN113337096A (en) * | 2021-06-23 | 2021-09-03 | 西北师范大学 | Attapulgite-graphene oxide composite powder and preparation method and application thereof |
| CN114773220A (en) * | 2021-12-21 | 2022-07-22 | 西安阿伯塔资环分析测试技术有限公司 | Rigid double-tail surfactant, preparation method thereof and clean fracturing fluid |
Citations (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1026978A (en) | 1962-03-30 | 1966-04-20 | Schwarzkopf Verwaltung G M B H | Method of dyeing hair |
| GB1153196A (en) | 1965-07-07 | 1969-05-29 | Schwarzkopf Verwaltung G M B H | Method of Dyeing Hair |
| DE2359399A1 (en) | 1973-11-29 | 1975-06-12 | Henkel & Cie Gmbh | Tetraaminopyrimidines as developers in oxidation hair dyes - esp. used with meta aminophenol couplers for blue shading dyes |
| US4137180A (en) | 1976-07-02 | 1979-01-30 | Lever Brothers Company | Fabric treatment materials |
| JPS63169571A (en) | 1987-01-06 | 1988-07-13 | Nec Corp | Tone detector |
| US4874554A (en) | 1986-07-10 | 1989-10-17 | Henkel Kommanditgesellschaft Auf Aktien | Quaternary ammonium compounds |
| DE3843892A1 (en) | 1988-12-24 | 1990-06-28 | Wella Ag | OXIDATION HAIR AGENTS CONTAINING DIAMINOPYRAZOL DERIVATIVES AND NEW DIAMINOPYRAZOLE DERIVATIVES |
| JPH0563124A (en) | 1991-09-03 | 1993-03-12 | Mitsubishi Electric Corp | Hybrid integrated circuit device |
| DE4133957A1 (en) | 1991-10-14 | 1993-04-15 | Wella Ag | HAIR DYE CONTAINING AMINOPYRAZOLE DERIVATIVES AND NEW PYRAZOLE DERIVATIVES |
| WO1994008969A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Process for producing 4,5-diamino pyrazole derivatives, their use for colouring hair and novel pyrazole derivatives |
| WO1994008970A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Oxidation hair dye containing 4,5-diaminopyrazole derivatives and novel 4,5-diaminopyrazole derivatives and process for their production |
| WO1996015765A1 (en) | 1994-11-17 | 1996-05-30 | Henkel Kommanditgesellschaft Auf Aktien | Oxidation dyes |
| FR2733749A1 (en) | 1995-05-05 | 1996-11-08 | Oreal | COMPOSITIONS FOR DYEING KERATINIC FIBERS CONTAINING DIAMINO PYRAZOLES, DYEING PROCESS, NOVEL DIAMINO PYRAZOLES, AND PREPARATION METHOD THEREOF |
| EP0770375A1 (en) | 1995-10-21 | 1997-05-02 | GOLDWELL GmbH | Hair dyeing composition |
| DE19543988A1 (en) | 1995-11-25 | 1997-05-28 | Wella Ag | Oxidative hair dye composition |
| US5922086A (en) | 1995-04-24 | 1999-07-13 | Clariant Gmbh | Hair colorants based on perimidine derivatives |
| FR2801308A1 (en) | 1999-11-19 | 2001-05-25 | Oreal | KERATIN FIBER DYEING COMPOSITIONS CONTAINING 3-AMINO PYRAZOLO- [1, (- a] -PYRIDINES, DYEING PROCESS, NOVEL 3-AMINO PYRAZOLO- [1,5-a] -PYRIDINES |
| FR2886136A1 (en) | 2005-05-31 | 2006-12-01 | Oreal | COMPOSITION FOR DYING KERATIN FIBERS COMPRISING AT LEAST ONE DIAMINO-N, N-DIHYDRO-PYRAZOLONE DERIVATIVE AND A CATIONIC OXIDATION DYE |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE4434165A1 (en) * | 1994-09-24 | 1996-03-28 | Cassella Ag | Hair dye |
| US6296672B1 (en) * | 1995-12-22 | 2001-10-02 | Novozymes A/S Patents | Enzymatic method for textile dyeing |
| BR0207949A (en) * | 2001-03-08 | 2004-07-27 | Ciba Sc Holding Ag | Porous Material Staining Method |
| DE102005062645A1 (en) * | 2005-12-23 | 2007-06-28 | Henkel Kgaa | Composition for oxidative dyeing of keratin fibers, especially human hair, contains a naphthoxathiol-2-one derivative as coupler |
| FR2940105B1 (en) * | 2008-12-19 | 2011-04-08 | Oreal | COMPOSITION COMPRISING A PARTICULAR OXYETHYLENE FIBER AND SURFACTANT, METHOD FOR COLORING THE SAME AND DEVICES THEREFOR |
-
2011
- 2011-12-13 FR FR1161571A patent/FR2983714A1/en not_active Withdrawn
-
2012
- 2012-12-11 WO PCT/EP2012/075094 patent/WO2013087631A2/en not_active Ceased
Patent Citations (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1026978A (en) | 1962-03-30 | 1966-04-20 | Schwarzkopf Verwaltung G M B H | Method of dyeing hair |
| GB1153196A (en) | 1965-07-07 | 1969-05-29 | Schwarzkopf Verwaltung G M B H | Method of Dyeing Hair |
| DE2359399A1 (en) | 1973-11-29 | 1975-06-12 | Henkel & Cie Gmbh | Tetraaminopyrimidines as developers in oxidation hair dyes - esp. used with meta aminophenol couplers for blue shading dyes |
| US4137180A (en) | 1976-07-02 | 1979-01-30 | Lever Brothers Company | Fabric treatment materials |
| US4874554A (en) | 1986-07-10 | 1989-10-17 | Henkel Kommanditgesellschaft Auf Aktien | Quaternary ammonium compounds |
| JPS63169571A (en) | 1987-01-06 | 1988-07-13 | Nec Corp | Tone detector |
| DE3843892A1 (en) | 1988-12-24 | 1990-06-28 | Wella Ag | OXIDATION HAIR AGENTS CONTAINING DIAMINOPYRAZOL DERIVATIVES AND NEW DIAMINOPYRAZOLE DERIVATIVES |
| JPH0563124A (en) | 1991-09-03 | 1993-03-12 | Mitsubishi Electric Corp | Hybrid integrated circuit device |
| DE4133957A1 (en) | 1991-10-14 | 1993-04-15 | Wella Ag | HAIR DYE CONTAINING AMINOPYRAZOLE DERIVATIVES AND NEW PYRAZOLE DERIVATIVES |
| WO1994008969A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Process for producing 4,5-diamino pyrazole derivatives, their use for colouring hair and novel pyrazole derivatives |
| WO1994008970A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Oxidation hair dye containing 4,5-diaminopyrazole derivatives and novel 4,5-diaminopyrazole derivatives and process for their production |
| WO1996015765A1 (en) | 1994-11-17 | 1996-05-30 | Henkel Kommanditgesellschaft Auf Aktien | Oxidation dyes |
| US5922086A (en) | 1995-04-24 | 1999-07-13 | Clariant Gmbh | Hair colorants based on perimidine derivatives |
| FR2733749A1 (en) | 1995-05-05 | 1996-11-08 | Oreal | COMPOSITIONS FOR DYEING KERATINIC FIBERS CONTAINING DIAMINO PYRAZOLES, DYEING PROCESS, NOVEL DIAMINO PYRAZOLES, AND PREPARATION METHOD THEREOF |
| EP0770375A1 (en) | 1995-10-21 | 1997-05-02 | GOLDWELL GmbH | Hair dyeing composition |
| DE19543988A1 (en) | 1995-11-25 | 1997-05-28 | Wella Ag | Oxidative hair dye composition |
| FR2801308A1 (en) | 1999-11-19 | 2001-05-25 | Oreal | KERATIN FIBER DYEING COMPOSITIONS CONTAINING 3-AMINO PYRAZOLO- [1, (- a] -PYRIDINES, DYEING PROCESS, NOVEL 3-AMINO PYRAZOLO- [1,5-a] -PYRIDINES |
| FR2886136A1 (en) | 2005-05-31 | 2006-12-01 | Oreal | COMPOSITION FOR DYING KERATIN FIBERS COMPRISING AT LEAST ONE DIAMINO-N, N-DIHYDRO-PYRAZOLONE DERIVATIVE AND A CATIONIC OXIDATION DYE |
Non-Patent Citations (4)
| Title |
|---|
| "CTFA dictionary", 1993 |
| M.R. PORTER: "Handbook of Surfactants", 1991, BLACKIE & SON, pages: 116 - 178 |
| TODD; BYERS: "Volatile Silicone Fluids for Cosmetics", COSMETICS AND TOILETRIES, vol. 91, 1976, pages 27 - 32 |
| WALTER: "Noll's Chemistry and Technology of Silicones", 1968, ACADEMIC PRESS |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104418811A (en) * | 2013-08-20 | 2015-03-18 | 江南大学 | 2,3-dihydronaphthalene-embedded m-diazabenzene analogue, synthetic method, drug composition and application thereof |
| CN104418811B (en) * | 2013-08-20 | 2017-05-31 | 江南大学 | The dihydro perimidine analog of one class 2,3, its synthetic method, pharmaceutical composition and purposes |
| US8884012B1 (en) * | 2013-08-26 | 2014-11-11 | Xerox Corporation | Dye compound and method of making the compound |
| JP2015042639A (en) * | 2013-08-26 | 2015-03-05 | ゼロックス コーポレイションXerox Corporation | Dye compound and method of making this compound |
| US9193869B2 (en) | 2013-08-26 | 2015-11-24 | Xerox Corporation | Dye compounds, method of making the compounds and ink composition employing the compounds |
| US9738811B2 (en) | 2013-08-26 | 2017-08-22 | Xerox Corporation | Phase change inks containing wax-soluble near-infrared dyes |
| DE102014216738B4 (en) * | 2013-08-26 | 2021-04-01 | Xerox Corporation | PERIMIDINE COUPLER AND METHOD OF MANUFACTURING THEREOF |
| CN104739836B (en) * | 2014-09-30 | 2017-03-29 | 三峡大学 | A kind of micromolecular inhibitor and its application |
| US20170314007A1 (en) * | 2014-09-30 | 2017-11-02 | China Three Gorges University | Medicament design pocket of ornithine decarboxylase and application of medicament design pocket |
| CN113337096A (en) * | 2021-06-23 | 2021-09-03 | 西北师范大学 | Attapulgite-graphene oxide composite powder and preparation method and application thereof |
| CN114773220A (en) * | 2021-12-21 | 2022-07-22 | 西安阿伯塔资环分析测试技术有限公司 | Rigid double-tail surfactant, preparation method thereof and clean fracturing fluid |
| CN114773220B (en) * | 2021-12-21 | 2024-01-26 | 西安阿伯塔资环分析测试技术有限公司 | Rigid double-tail surfactant, preparation method thereof and clean fracturing fluid |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2013087631A3 (en) | 2015-01-22 |
| FR2983714A1 (en) | 2013-06-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8828099B2 (en) | Dyeing or lightening process using a composition rich in fatty substances comprising a solid alcohol and a solid ester, compositions and device | |
| US9044412B2 (en) | Dye composition using a long-chain ether of an alkoxylated fatty alcohol and a cationic polymer, processes and devices using the same | |
| US8808400B2 (en) | Dyeing or lightening process using a composition rich in fatty substances comprising an alcohol having at least 20 carbons, compositions and device | |
| WO2014207097A1 (en) | Cosmetic composition for lightening or dyeing the hair, comprising two basic agents, an acid and an oxidizing agent | |
| EP2846761A2 (en) | Composition comprising (2,5-diaminophenyl)ethanol, an alkylpolyglucoside nonionic surfactant, an oxyethylenated sorbitan ester or a polyalkoxylated or polyglycerolated fatty alcohol in a medium rich in fatty substances, dyeing process and device therefore | |
| WO2012146530A1 (en) | Dye composition using a meta-aminophenol-derived coupler in a medium rich in fatty substances, processes and devices | |
| WO2013087631A2 (en) | Oxidation dye composition comprising a particular coupler in a medium rich in fatty substances, and processes and device suitable therefor | |
| WO2013083642A1 (en) | Oil-rich aqueous composition and its use in an oxidative colouring or bleaching method | |
| WO2012146527A2 (en) | Dye composition using a 1-hydroxynaphthalene coupler and a heterocyclic base in a fatty-substance-rich medium, dyeing process and device therefor | |
| US9066890B2 (en) | Dye composition comprising an alkoxylated fatty alcohol ether and a fatty alcohol | |
| WO2012146531A1 (en) | Dye composition using a phenol-derived coupler in a medium rich in fatty substances, processes and devices | |
| WO2013004787A1 (en) | Dye composition using a long-chain ether of an alkoxylated fatty alcohol and glycerol, processes and devices using the same | |
| US9028562B2 (en) | Dye composition using a 2-hydroxynaphthalene, (acylamino)phenol or quinoline coupler in a fatty-substance-rich medium, dyeing process and device therefor | |
| WO2012163898A1 (en) | Dye composition using a (hydroxy)indoline coupler in a medium rich in fatty substances, process and devices | |
| WO2013144244A2 (en) | Dye composition using (2,5-diaminophenyl)ethanol and an aliphatic fatty alcohol in a medium rich in fatty substances, dyeing process and device | |
| EP2950774A2 (en) | Dye composition using at least one coupler of meta-phenylenediamine type substituted in position 4 in a medium comprising a fatty substance, processes and device | |
| WO2013004786A2 (en) | Dye composition using a long-chain ether of an alkoxylated fatty alcohol and a pyrazolone oxidation base, processes and devices using the same | |
| US9585824B2 (en) | Dye composition using at least one coupler of meta-phenylenediamine type substituted in position 2 in a medium comprising a fatty substance, processes and device | |
| WO2017050994A1 (en) | Dye composition using at least one oxidation base and at least one particular aminobenzene coupler, processes and device | |
| WO2014118231A2 (en) | Dye composition using at least one coupler of meta-aminophenol type in a medium comprising a fatty substance, processes and device | |
| WO2013092484A2 (en) | Oxidation dyeing process rich in fatty substances and a metal catalyst, and device therefor | |
| WO2013087634A2 (en) | Oxidative dyeing composition comprising a specific perimidine coupler in a medium with high fatty substance content, methods and suitable device | |
| WO2017051020A1 (en) | Dye composition comprising a resorcinol-based coupler and an oxidation base | |
| WO2013092482A2 (en) | Oxidation dye composition rich in fatty substances, processes and suitable device for same | |
| WO2014118093A2 (en) | Dye composition comprising at least one particular lipophilic meta-aminophenol coupler in a medium rich in fatty substances, processes and devices |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12798327 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12798327 Country of ref document: EP Kind code of ref document: A2 |