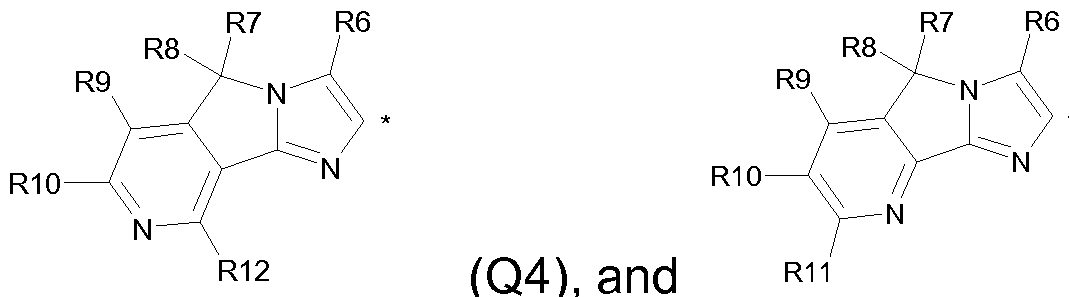WO2013045607A1 - Quinazoline linked heteroaromatic tricycle derivatives as pde10a enzyme inhibitors - Google Patents
Quinazoline linked heteroaromatic tricycle derivatives as pde10a enzyme inhibitors Download PDFInfo
- Publication number
- WO2013045607A1 WO2013045607A1 PCT/EP2012/069179 EP2012069179W WO2013045607A1 WO 2013045607 A1 WO2013045607 A1 WO 2013045607A1 EP 2012069179 W EP2012069179 W EP 2012069179W WO 2013045607 A1 WO2013045607 A1 WO 2013045607A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fluoro
- imidazo
- quinazolin
- ethyl
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 Cc1c(C(*)(*)[n]2c-3nc(C*)c2*)c-3c(*)c(*)c1O Chemical compound Cc1c(C(*)(*)[n]2c-3nc(C*)c2*)c-3c(*)c(*)c1O 0.000 description 2
- GNSNZIQPKXSYJA-UHFFFAOYSA-N CC(c(c(N)c1)cc(F)c1F)=O Chemical compound CC(c(c(N)c1)cc(F)c1F)=O GNSNZIQPKXSYJA-UHFFFAOYSA-N 0.000 description 1
- NNYQZLMGCMVTJA-UHFFFAOYSA-N CC(c(cc(c(F)c1)F)c1NC(CCc1c[n](Cc2c-3cccc2)c-3n1)=O)=O Chemical compound CC(c(cc(c(F)c1)F)c1NC(CCc1c[n](Cc2c-3cccc2)c-3n1)=O)=O NNYQZLMGCMVTJA-UHFFFAOYSA-N 0.000 description 1
- GWMFSNVPMFPPNM-UHFFFAOYSA-N OC(CCc1c[n](Cc2c-3cccc2)c-3n1)=O Chemical compound OC(CCc1c[n](Cc2c-3cccc2)c-3n1)=O GWMFSNVPMFPPNM-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains three hetero rings
- C07D471/14—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
Definitions
- the present invention provides heteroaromatic compounds that are PDE10A enzyme inhibitors, and as such are useful to treat neurodegenerative and psychiatric disorders. Especially, the invention provides compounds that are highly selective for PDE10 over other PDE subtypes. The present invention also provides pharmaceutical compositions comprising compounds of the invention and methods of treating disorders using the compounds of the invention.
- cyclic-adenosine monophosphate cyclic-adenosine monophosphate
- cGMP cyclic- guanosine monophosphate
- Intracellular cAMP and cGMP are generated by adenyl and guanyl cyclases, and are degraded by cyclic nucleotide phosphodiesterases (PDEs) via hydrolysis of the cyclic nucleotides into their respective nucleotide monophosphates.
- PDEs cyclic nucleotide phosphodiesterases
- Phosphodieasterase 10A is a dual-specificity phosphodiesterase that can convert both cAMP to AMP and cGMP to GMP (Soderling, S. et al. Proc. Natl. Acad. Sci. 1999, 96, 7071 -7076). PDE10A is primarily expressed in the neurons in the striatum, n. accumbens and in the olfactory tubercle (Kotera, J. et al. Biochem. Biophys. Res. Comm. 1999, 261, 551 -557 and Seeger, T.F. et al. Brain Research, 2003, 985, 1 13-126).
- MSN medium spiny neurons
- D 2 D 2 dopamine receptors
- the Di class of neurons is part of the 'direct' striatal output pathway, which broadly functions to facilitate behavioral responses.
- the D 2 class of neurons is part of the 'indirect' striatal output pathway, which functions to suppress behavioral responses that compete with those being facilitated by the 'direct' pathway.
- Dopamine D 2 receptor antagonism is well established in the treatment of schizophrenia.
- D 2 receptor antagonism has been the mainstay in psychosis treatment and all effective antipsychotic drugs antagonise D 2 receptors.
- the effects of D 2 are likely to be mediated primarily through neurons in the striatum, nucleus accumbens and olfactory tubercle, since these areas receive the densest dopaminergic projections and have the strongest expression of D 2 receptors (Konradi, C. and Heckers, S. Society of Biological Psychiatry, 2001 , 50, 729-742).
- PDE10A in this context, has the desired expression profile with high and relatively specific expression in neurons in striatum, nucleus accumbens and olfactory tubercle, PDE10A inhibition is likely to have effects similar to D 2 receptor antagonism and therefore have antipsychotic effects.
- PDE10A inhibition is expected to mimic D 2 receptor antagonism in part, it might be expected to have a different profile.
- the D 2 receptor has signaling components besides cAMP (Neve, K. A. et al. Journal of Receptors and Signal Transduction 2004, 24, 165-205), wherefore interference with cAMP through PDE10A inhibition may reduce the risk of the extrapyramidal side effects that are seen with strong D 2 antagonism.
- PDE10A inhibition may have some effects not seen with D 2 receptor antagonism.
- PDE10A is also expressed in Di receptors expressing striatal neurons (Seeger, T. F. et al. Brain Research, 2003, 985, 1 13-126).
- PDE10A inhibition is likely to also have effects that mimic Di receptor agonism.
- PDE10A inhibition will not only increase cAMP in cells, but might also be expected to increase cGMP levels, since PDE10A is a dual specificity phosphodiesterase.
- cGMP activates a number of target protein in cells like cAMP and also interacts with the cAMP signaling pathways.
- PDE10A inhibition is likely to mimic D 2 receptor antagonism in part and therefore has antipsychotic effect, but the profile might differ from that observed with classical D 2 receptor antagonists.
- the PDE10A inhibitor papaverine is shown to be active in several antipsychotic models. Papaverine potentiated the cataleptic effect of the D 2 receptor antagonist haloperidol in rats, but did not cause catalepsy on its own (WO 03/093499). Papaverine reduced hyperactivity in rats induced by PCP, while reduction of amphetamine induced hyperactivity was insignificant (WO 03/093499). These models suggest that PDE10A inhibition has the classic antipsychotic potential that would be expected from the theoretical considerations outlined above. WO 03/093499 further discloses the use of selective PDE10 inhibitors for the treatment of associated neurologic and psychiatric disorders.
- PDE10A inhibition reverses subchronic PCP-induced deficits in attentional set-shifting in rats (Rodefer et al. Eur. J. Neurosci. 2005, 4, 1070-1076). This model suggests that PDE10A inhibition might alleviate cognitive deficits associated with schizophrenia.
- the tissue distribution of PDE10A indicates that PDE10A inhibitors can be used to raise levels of cAMP and/or cGMP within cells that express the PDE10A enzyme, especially neurons that comprise the basal ganglia, and the PDE10A inhibitors of the present invention would therefore be useful in treating a variety of associated neuropsychiatric conditions involving the basal ganglia such as neurological and psychiatric disorders, schizophrenia, bipolar disorder, psychosis, obsessive compulsive disorder and addiction, and may have the benefit of not possessing unwanted side effects, which are associated with the current therapies on the market.
- Pyrrolodihydroisoquinolines and variants thereof are disclosed as inhibitors of PDE10 in WO 05/03129 and WO 05/02579.
- Piperidinyl-substituted quinazolines and isoquinolines that serve as PDE10 inhibitors are disclosed in WO 05/82883.
- WO 06/1 1040 discloses substituted quinazoline and isoquinoline compounds that serve as inhibitors of PDE10.
- US 20050182079 discloses substituted tetrahydroisoquinolinyl derivatives of quinazoline and isoquinoline that serve as effective phosphodiesterase (PDE) inhibitors.
- PDE phosphodiesterase
- US 20060019975 discloses piperidine derivatives of quinazoline and isoquinoline that serve as effective phosphodiesterase (PDE) inhibitors.
- US 20060019975 also relates to compounds that are selective inhibitors of PDE10.
- WO 06/028957 discloses cinnoline derivatives as inhibitors of PDE10 for the treatment of psychiatric and neurological syndromes.
- WO09/152825 discloses phenylimidazole derivatives as compounds that serve as inhibitors of PDE10.
- the present invention provides compounds that are PDE10A enzyme inhibitors and thus useful for treatment for neurodegenerative and/or psychiatric disorders, which are not efficacious in all patients. Hence, there remains a need for alternative methods of treatment. Summary of the Invention
- the objective of the present invention is to provide compounds that are selective PDE10A enzyme inhibitors.
- a further objective of the present invention is to provide compounds which have such activity, and which have improved solubility, metabolic stability and/or bioavailability compared to prior art compounds.
- Another objective of the invention is to provide an effective treatment, in particular long-term treatment, of a human patient, without causing the side effects typically associated with current therapies for neurological and psychiatric disorders.
- Embodiments of the invention in a first embodiment (E1 ) the present invention relates to compounds of formula I:
- R6, R7, R8, R9, R10, R1 1 and R12 are individually selected from the group consisting of hydrogen, halogen, Ci-C6 alkyl and Ci-C 4 alkoxy, and wherein * denotes the attachment point, and tautomers and pharmaceutically acceptable salts thereof, and polymorphic forms thereof.
- R1 is selected from the group consisting of hydrogen, methyl, methoxy, methylamine and triflourmethyl.
- R2, R3, R4 and R5 are selected from the group consisting of fluorine and bromine.
- R6, R7, R8, R9, R10, R1 1 and R12 are individually selected from the group consisting of hydrogen, fluorine and methoxy.
- the invention provides a compound of formula I, or a pharmaceutically acceptable salt thereof, for use as a medicament.
- the present invention provides a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a compound of formula I and a pharmaceutically acceptable carrier, diluent or excipient.
- the present invention provides the use of a compound of formula I, or a pharmaceutically acceptable salt thereof, for the preparation of a medicament for the treatment of a neurodegenerative or psychiatric disorder.
- the present invention provides a method of treating a subject suffering from a neurodegenerative disorder, comprising administering to the subject a therapeutically effective amount of a compound of formula I.
- the present invention provides a method of treating a subject suffering from a psychiatric disorder, comprising administering to the subject a therapeutically effective amount of a compound of formula I.
- the present invention provides a method of treating a subject suffering from a drug addiction, such as an alcohol, amphetamine, cocaine, or opiate addiction.
- a drug addiction such as an alcohol, amphetamine, cocaine, or opiate addiction.
- halo and halogen are used interchangeably and refer to fluorine, chlorine, bromine or iodine.
- C1 -C6 alkyl refers to a straight-chain or branched saturated hydrocarbon having from one to six carbon atoms, inclusive. Examples of such groups include, but are not limited to, methyl, ethyl, 1 -propyl, 2-propyl, 1 - butyl, 2-butyl, 2-methyl-2-propyl, 2-methyl-1 -butyl, and n-hexyl.
- C1 -C6 hydroxyalkyl refers to a C1 -C6 alkyl group as defined above which is substituted with one hydroxy group.
- halo(Ci - C6)alkyl refers to a C1 -C6 alkyl group as defined above which is substituted with up to three halogen atoms, such as trifluoromethyl.
- C1 -C6 alkoxy refers to a straight-chain or branched saturated alkoxy group having from one to six carbon atoms, inclusive, with the open valency on the oxygen. Examples of such groups include, but are not limited to, methoxy, ethoxy, n-butoxy, 2-methyl-pentoxy and n-hexyloxy.
- C3-C8 cycloalkyi typically refers to cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl.
- C1 -C6 alkyl(C3-C8)cycloalkyl refers to a C3-C8 cycloalkyi as defined above which is substituted with a straight-chain or branched C1-C6 alkyl. Examples of such groups include, but are not limited to, cyclopropylmethyl.
- heterocycloalkyl refers to a four to eight membered ring containing carbon atoms and up to three N, O or S atoms, provided that the four to eight membered ring does not contain adjacent O or adjacent S atoms.
- the open valency is on either the heteroatom or carbon atom. Examples of such groups include, but are not limited to, azetidinyl, oxetanyl, piperazinyl, morpholinyl, thiomorpholinyl and [1 ,4]diazepanyl.
- hydroxyheterocycloalkyl refers to a heterocycloalkyl as defined above which is substituted with one hydroxy group.
- C1-C6 alkyl-heterocycloalkyl refers to a heterocycloalkyl as defined above which is substituted with a C1-C6 alkyl group.
- Examples of such groups include, but are not limited to, tetrahydropyran-4-yl-methyl and 2-morpholin-4-yl-ethyl.
- aryl refers to a phenyl ring, optionally substituted with halogen, Ci- C6 alkyl, C1-C6 alkoxy or halo(Ci-Ce)alkyl as defined above. Examples of such groups include, but are not limited to, phenyl and 4-chlorophenyl.
- d-Cearylalkyl refers to an aryl as defined above which is substituted with a straight-chain or branched C1-C6 alkyl. Examples of such groups include, but are not limited to, benzyl and 4-chlorobenzyl.
- the present invention further provides certain embodiments of the invention that are described below.
- HET is a heteroaromatic group of formula II containing 2 nitrogen atoms. In another embodiment of the invention, HET is a heteroaromatic group of formula II containing 3 nitrogen atoms. In yet another embodiment of the invention, HET is a heteroaromatic group of formula II containing 4 nitrogen atoms. In a further embodiment one or more of the hydrogen atoms of the compound of formula I have been substituted by deuterium.
- the compound of formula I is selected among the following specific compounds listed in Table 1 and Table2, in the form of the free base, one or more tautomers thereof or a pharmaceutically acceptable salt thereof.
- Table 1 lists compounds of the invention where HET is a 2-quinazolinyl group of formula I la and the corresponding IC 5 o values determined as described in the section "PDE10A inhibition assay”.
- Table 2 lists compounds of the invention where HET is a 4- quinazolinyl group of formula Mb and the corresponding IC 5 o values determined as described in the section "PDE10A inhibition assay".
- Each of the compounds constitutes an individual embodiment, of the present invention. It should be understood that the various aspects, embodiments, implementations and features of the invention mentioned herein may be claimed separately, or in any combination, as illustrated by the following non- limiting examples.
- the compounds of the present invention have an IC 5 o value of less than 50 nM, such as in the range of 0.2 - 20 nM, particularly in the range of 0.2 - 10 nM, such as in the range of 0.2 - 5 nM or in the range of 0.2 - 1 nM.
- the present invention also comprises salts of the compounds, typically, pharmaceutically acceptable salts.
- Such salts include pharmaceutically acceptable acid addition salts.
- Acid addition salts include salts of inorganic acids as well as organic acids.
- suitable inorganic acids include hydrochloric, hydrobromic, hydroiodic, phosphoric, sulfuric, sulfamic, nitric acids and the like.
- suitable organic acids include formic, acetic, trichloroacetic, trifluoroacetic, propionic, benzoic, cinnamic, citric, fumaric, glycolic, itaconic, lactic, methanesulfonic, maleic, malic, malonic, mandelic, oxalic, picric, pyruvic, salicylic, succinic, methane sulfonic, ethanesulfonic, tartaric, ascorbic, pamoic, bismethylene salicylic, ethanedisulfonic, gluconic, citraconic, aspartic, stearic, palmitic, EDTA, glycolic, p-aminobenzoic, glutamic, benzenesulfonic, p-tol
- the compounds of this invention may exist in unsolvated as well as in solvated forms with pharmaceutically acceptable solvents such as water, ethanol and the like.
- the solvated forms are considered equivalent to the unsolvated forms for the purposes of this invention.
- the present invention further provides a pharmaceutical composition comprising a therapeutically effective amount of a compound of formula I and a pharmaceutically acceptable carrier or diluent.
- the present invention also provides a pharmaceutical composition comprising a therapeutically effective amount of one of the specific compounds disclosed in the Experimental Section herein and a pharmaceutically acceptable carrier or diluent.
- the compounds of the invention may be administered alone or in combination with pharmaceutically acceptable carriers, diluents or excipients, in either single or multiple doses.
- pharmaceutical compositions according to the invention may be formulated with pharmaceutically acceptable carriers or diluents as well as any other known adjuvants and excipients in accordance with conventional techniques such as those disclosed in Remington: The Science and Practice of Pharmacy, 19 th Edition, Gennaro, Ed., Mack Publishing Co., Easton, PA, 1995.
- compositions may be specifically formulated for administration by any suitable route such as oral, rectal, nasal, pulmonary, topical (including buccal and sublingual), transdermal, intracisternal, intraperitoneal, vaginal and parenteral (including subcutaneous, intramuscular, intrathecal, intravenous and intradermal) routes. It will be appreciated that the route will depend on the general condition and age of the subject to be treated, the nature of the condition to be treated and the active ingredient.
- compositions for oral administration include solid dosage forms such as capsules, tablets, dragees, pills, lozenges, powders and granules. Where appropriate, the compositions may be prepared with coatings such as enteric coatings or they may be formulated so as to provide controlled release of the active ingredient such as sustained or prolonged release according to methods well known in the art.
- Liquid dosage forms for oral administration include solutions, emulsions, suspensions, syrups and elixirs.
- Pharmaceutical compositions for parenteral administration include sterile aqueous and nonaqueous injectable solutions, dispersions, suspensions or emulsions as well as sterile powders to be reconstituted in sterile injectable solutions or dispersions prior to use.
- Other suitable administration forms include, but are not limited to, suppositories, sprays, ointments, creams, gels, inhalants, dermal patches and implants.
- Typical oral dosages range from about 0.001 to about 100 mg/kg body weight per day. Typical oral dosages also range from about 0.01 to about 50 mg/kg body weight per day. Typical oral dosages further range from about 0.05 to about 10 mg/kg body weight per day. Oral dosages are usually administered in one or more dosages, typically, one to three dosages per day. The exact dosage will depend upon the frequency and mode of administration, the sex, age, weight and general condition of the subject treated, the nature and severity of the condition treated and any concomitant diseases to be treated and other factors evident to those skilled in the art.
- a typical unit dosage form for oral administration may contain from about 0.01 to about 1000 mg, from about 0.05 to about 500 mg, or from about 0.5 mg to about 200 mg.
- parenteral routes such as intravenous, intrathecal, intramuscular and similar administration
- typical doses are in the order of half the dose employed for oral administration.
- the present invention also provides a process for making a pharmaceutical composition
- a pharmaceutical composition comprising admixing a therapeutically effective amount of a compound of formula I and at least one pharmaceutically acceptable carrier or diluent.
- the compound utilized in the aforementioned process is one of the specific compounds disclosed in the Experimental Section herein.
- the compounds of this invention are generally utilized as the free substance or as a pharmaceutically acceptable salt thereof.
- One example is an acid addition salt of a compound having the utility of a free base.
- a compound of formula I contains a free base such salts are prepared in a conventional manner by treating a solution or suspension of a free base of formula I with a molar equivalent of a pharmaceutically acceptable acid.
- suitable organic and inorganic acids are described above.
- solutions of the compounds of formula I in sterile aqueous solution, aqueous propylene glycol, aqueous vitamin E or sesame or peanut oil may be employed.
- aqueous solutions should be suitably buffered if necessary and the liquid diluent first rendered isotonic with sufficient saline or glucose.
- the aqueous solutions are particularly suitable for intravenous, intramuscular, subcutaneous and intraperitoneal administration.
- the compounds of formula I may be readily incorporated into known sterile aqueous media using standard techniques known to those skilled in the art.
- Suitable pharmaceutical carriers include inert solid diluents or fillers, sterile aqueous solutions and various organic solvents.
- solid carriers include lactose, terra alba, sucrose, cyclodextrin, talc, gelatin, agar, pectin, acacia, magnesium stearate, stearic acid and lower alkyl ethers of cellulose.
- liquid carriers include, but are not limited to, syrup, peanut oil, olive oil, phospholipids, fatty acids, fatty acid amines, polyoxyethylene and water.
- the carrier or diluent may include any sustained release material known in the art, such as glyceryl monostearate or glyceryl distearate, alone or mixed with a wax.
- the pharmaceutical compositions formed by combining the compounds of formula I and a pharmaceutically acceptable carrier are then readily administered in a variety of dosage forms suitable for the disclosed routes of administration.
- the formulations may conveniently be presented in unit dosage form by methods known in the art of pharmacy.
- Formulations of the present invention suitable for oral administration may be presented as discrete units such as capsules or tablets, each containing a predetermined amount of the active ingredient, and optionally a suitable excipient.
- the orally available formulations may be in the form of a powder or granules, a solution or suspension in an aqueous or non-aqueous liquid, or an oil-in-water or water-in-oil liquid emulsion.
- the preparation may be tabletted, placed in a hard gelatin capsule in powder or pellet form or it may be in the form of a troche or lozenge.
- the amount of solid carrier will vary widely but will range from about 25 mg to about 1 g per dosage unit.
- the preparation may be in the form of a syrup, emulsion, soft gelatin capsule or sterile injectable liquid such as an aqueous or non-aqueous liquid suspension or solution.
- compositions of the invention may be prepared by conventional methods in the art.
- tablets may be prepared by mixing the active ingredient with ordinary adjuvants and/or diluents and subsequently compressing the mixture in a conventional tabletting machine prepare tablets.
- adjuvants or diluents comprise: corn starch, potato starch, talcum, magnesium stearate, gelatin, lactose, gums, and the like. Any other adjuvants or additives usually used for such purposes such as colorings, flavorings, preservatives etc. may be used provided that they are compatible with the active ingredients.
- the term "therapeutically effective amount" of a compound means an amount sufficient to cure, alleviate or partially arrest the clinical manifestations of a given disease and its complications in a therapeutic intervention comprising the administration of said compound.
- An amount adequate to accomplish this is defined as “therapeutically effective amount”.
- Effective amounts for each purpose will depend on the severity of the disease or injury as well as the weight and general state of the subject. It will be understood that determining an appropriate dosage may be achieved using routine experimentation, by constructing a matrix of values and testing different points in the matrix, which is all within the ordinary skills of a trained physician.
- treatment means the management and care of a patient for the purpose of combating a condition, such as a disease or a disorder.
- the term is intended to include the full spectrum of treatments for a given condition from which the patient is suffering, such as administration of the active compound to alleviate the symptoms or complications, to delay the progression of the disease, disorder or condition, to alleviate or relief the symptoms and complications, and/or to cure or eliminate the disease, disorder or condition as well as to prevent the condition, wherein prevention is to be understood as the management and care of a patient for the purpose of combating the disease, condition, or disorder and includes the administration of the active compounds to prevent the onset of the symptoms or complications. Nonetheless, prophylactic (preventive) and therapeutic (curative) treatments are two separate aspects of the invention.
- the patient to be treated is preferably a mammal, in particular a human being.
- the compounds of formula I are PDE10A enzyme inhibitors and as such are useful to treat associated neurological and psychiatric disorders.
- the invention thus provides a compound of formula I or a pharmaceutically acceptable acid addition salt thereof, as well as a pharmaceutical composition containing such a compound, for use in the treatment of a neurodegenerative disorder, psychiatric disorder or drug addiction in humans.
- the neurodegenerative disorder or condition involves neurodegeneration of striatal medium spiny neurons in a human.
- the neurodegenerative disorder or condition is Huntington's disease.
- the disorder is dyskinesia associated with dopamine agonist therapy.
- the psychiatric disorder is selected from the group consisting of schizophrenia, for example of the paranoid, disorganized, catatonic, undifferentiated, or residual type; schizophreniform disorder; schizoaffective disorder, for example of the delusional type or the depressive type; delusional disorder; substance-induced psychotic disorder, for example psychosis induced by alcohol, amphetamine, cannabis, cocaine, hallucinogens, inhalants, opioids, or phencyclidine; personality disorder of the paranoid type; and personality disorder of the schizoid type.
- schizophrenia for example of the paranoid, disorganized, catatonic, undifferentiated, or residual type
- schizophreniform disorder for example of the delusional type or the depressive type
- delusional disorder substance-induced psychotic disorder, for example psychosis induced by alcohol, amphetamine, cannabis, cocaine, hallucinogens, inhalants, opioids, or phencyclidine
- personality disorder of the paranoid type and personality
- This invention further provides a method of treating a drug addiction, for example an alcohol, amphetamine, cocaine, or opiate addiction, in a human, which method comprises administering to said human an amount of a compound of formula I effective in treating addiction, such as drug addiction.
- a drug addiction for example an alcohol, amphetamine, cocaine, or opiate addiction
- drug addiction means an abnormal desire for a drug and is generally characterized by motivational disturbances such a compulsion to take the desired drug and episodes of intense drug craving.
- Other disorders that can be treated according to the present invention are obsessive/compulsive disorders, non-insuline demanding diabetes mellitus (NIDDM), and Tourette's syndrome and other tic disorders as well as Attention Deficit/Hyperactivity Disorder (ADHD).
- NIDDM non-insuline demanding diabetes mellitus
- ADHD Attention Deficit/Hyperactivity Disorder
- the compounds of formula I or pharmaceutically acceptable salts thereof may be used in combination with one or more other drugs (including typical and atypical antpsychotic agent) in the treatment of diseases or conditions for which the compounds of the present invention have utility, where the combination of the drugs together are safer or more effective than either drug alone.
- the compounds of the present invention may be used in combination with one or more other drugs that treat, prevent, control, ameliorate, or reduce the risk of side effects or toxicity of the compounds of the present invention.
- the combinations, uses and methods of treatment of the invention may also provide advantages in treatment of patients who fail to respond adequately or who are resistant to other known treatments.
- compositions of the present invention include those that contain one or more other active ingredients, in addition to the compounds of the present invention.
- the combinations may be administered as part of a unit dosage form combination product, or as a kit or treatment protocol wherein one or more additional drugs are administered in separate dosage forms as part of a treatment regimen.
- neuroleptic agent refers to drugs, which have the effect on cognition and behaviour of antipsychotic agent drugs that reduce confusion, delusions, hallucinations, and psychomotor agitation in patients with psychoses.
- neuroleptic agents include, but are not limited to: typical antipsychotic drugs, including phenothiazines, further divided into the aliphatics, piperidines, and piperazines, thioxanthenes (e.g., cisordinol), butyrophenones (e.g., haloperidol), dibenzoxazepines (e.g., loxapine), dihydroindolones (e.g., molindone), diphenylbutylpiperidines (e.g., pimozide), and atypical antipsychotic drugs, including benzisoxazoles (e.g., risperidone), sertindole, olan
- neuroleptic agents for use in the invention are sertindole, olanzapine, risperidone, quetiapine, aripiprazole, haloperidol, clozapine, ziprasidone and osanetant.
- a "neurodegenerative disorder or condition” refers to a disorder or condition that is caused by the dysfunction and/or death of neurons in the central nervous system.
- the treatment of these disorders and conditions can be facilitated by administration of an agent which prevents the dysfunction or death of neurons at risk in these disorders or conditions and/or enhances the function of damaged or healthy neurons in such a way as to compensate for the loss of function caused by the dysfunction or death of at-risk neurons.
- neurotrophic agent refers to a substance or agent that has some or all of these properties.
- LC-MS were run on a Sciex API150EX equipped with APPI-source operating in positive ion mode.
- the HPLC consisted of Shimadzu LC10-ADvp LC pumps. SPD-M20A PDA detector (operating at 254 nM) and SCL-10A system controller. Autosampler was Gilson 215. Colomn oven was a Jones Chromatography 7990R and ELS detector was a Sedere Sedex 85.
- LC-conditions The column was a Waters Symmetry C-18. 4.6 x 30 mm. 3.5 ⁇ operating at 60 °C with 2.5 ml/min of a binary gradient consisting of water + 0.05 % TFA (A) and methanol + 0.05 % TFA. Gradient:
- LC-MS were run on a Sciex API300 equipped with APPI source operating in positive ion mode.
- the UPLC consisted of Waters Aquity including column mager. binary solvent manager, sample organizer. PDA detector (operating at 254 nM) and ELS detector.
- LC-conditions The column was a Waters Aquity UPLC BEH C-18. 2.1 x 50 mm. 1 .7 ⁇ operating at 60 °C with 1 .2 ml/min of a binary gradient consisting of water + 0.05 % TFA (A) and 95 % acetonitrile containing 5 % water + 0.03 % TFA. Gradient Time. min. %B
- LC-MS were run on an Agilent 1 100 LCMS system with ELS Detector, Pump G131 1A, Degasser G1379A,Well-plate Autosampler, G1367A, Column Oven G1316A, DAD G1315B,MSD G1946C or G1956A,Alltech ELSD 800,Column YMC ODS-AQ,Particle size 5 micrometer, Pore size 12 nm, Dimension 50 * 2.0 mm ID, Injection volume 2 microliter, Column temperature 50°C, Flow 0.8 ml/min.
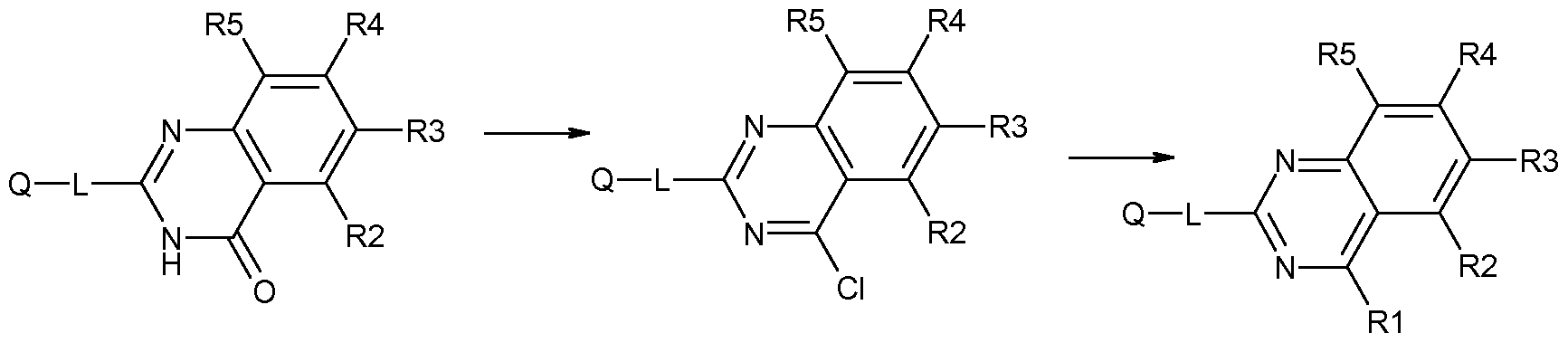
- This reaction is typically carried out in a solvent such as 1 -propanol, toluene, DMF, or acetonitrile, optionally in the presence of a carbonate base such as potassium carbonate or a tertiary amine base such as triethylamine or diisopropylethylannine (DIPEA), at a temperature ranging from about 0°C to about 200°C, optionally under pressure in a closed vessel.
- a solvent such as 1 -propanol, toluene, DMF, or acetonitrile
- a carbonate base such as potassium carbonate
- a tertiary amine base such as triethylamine or diisopropylethylannine (DIPEA)
- DIPEA diisopropylethylannine
- suitable solvents include benzene, chloroform, dioxane, ethyl acetate, 2-propanol and xy
- Thiol-compounds of formula IV are either commercially available or can be made by methods similar to those described in the literature e.g. Journal of Heterocyclic Chemistry 1977 14(5), 889-92, Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry 1979, 5, 1 132-6, Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio- Organic Chemistry 1997, 20, 2983-2988, Organic & Biomolecular Chemistry 2009, 7, 128-134.
- Electrophiles of formula III can be prepared as described in e.g. Venkatesan, A. et al. ChemMedChem 2008, 3, 1658-1661 .
- a transition metal catalyst such as palladium metal
- a hydrogen source such as hydrogen gas, ammonium hydrogen carbonate, or cyclohexadiene.
- the reduction can be performed using tosylhydrazide TsNHNH2 in dimethylformamide at elevated temperature e.g. 130 °C.
- Phosphonium salt of formula VII are readily available by reaction of compounds of formula V with triphenylphosphine by methods known to chemists skilled in the art.
- Aldehydes of formula VIII are available by methods described in the literature se e.g. Venkatesan, A. et al.
- This reaction is typically carried out in a suitable solvent, such as methanol and performed by mixing a solution of orthoacyl-anilide of formula XI in 7M NH3/MeOH solution and heating at 50°C for 6-24h.
- a suitable solvent such as methanol
- the orthoacyl-anilide of formula XI can be prepared from commercially available substituted anthranilic acids XII by standard organic synthesis methods known in the art as shown in scheme 5.
- Active PDE10A enzyme is prepared in a number of ways for use in PDE assays (Loughney, K. et al. Gene 1999, 234, 109-1 17; Fujishige, K. et al. Eur J Biochem. 1999, 266, 1 1 18-1 127 and Soderling, S. et al. Proc. Natl. Acad. Sci. 1999, 96, 7071 -7076).
- PDE10A can be expressed as full-length proteins or as truncated proteins, as long as they express the catalytic domain.
- PDE10A can be prepared in different cell types, for example insect cells or E. coli.
- catalytically active PDE10A is as follows: The catalytic domain of human PDE10A (amino acids 440-779 from the sequence with accession number NP 006652) is amplified from total human brain total RNA by standard RT-PCR and is cloned into the BamH1 and Xho1 sites of the pET28a vector (Novagen). Expression in coli is performed according to standard protocols. Briefly, the expression plasmids are transformed into the BL21 (DE3) E. coli strain, and 50 ml_ cultures inoculated with the cells allowed to grow to an OD600 of 0.4-0.6 before protein expression is induced with 0.5mM IPTG.
- a PDE1 OA assay may for example, be performed as follows: The assay is performed in 60 uL samples containing a fixed amount of the relevant PDE enzyme (sufficient to convert 20-25% of the cyclic nucleotide substrate), a buffer (50 mM HEPES7.6; 10mM MgCI 2 ; 0.02% Tween20), 0.1 mg/ml BSA, 225 pCi of 3 H-labelled cyclic nucleotide substrate, tritium labeled cAMP to a final concentration of 5 nM and varying amounts of inhibitors.
- Reactions are initiated by addition of the cyclic nucleotide substrate, and reactions are allowed to proceed for one hr at room temperature before being terminated through mixing with 15 uL 8 mg/mL yttrium silicate SPA beads (Amersham). The beads are allowed to settle for one hr in the dark before the plates are counted in a Wallac 1450 Microbeta counter. The measured signal can be converted to activity relative to an uninhibited control (100 %) and IC 5 o values can be calculated using the Xlfit extension to EXCEL.
- mice Male mice (NMRI, Charles River) weighing 20-25g are used. Eight mice are used in each group receiving the test compound (5 mg/kg) plus PCP (2.3 mg/kg) including the parallel control groups receiving the vehicle of the test compound plus PCP or vehicle injections only. The injection volumen is 10 ml/kg. The experiment is made in normal light conditions in an undisturbed room. The test substance is injected per oss 60 min before injection of PCP, which is administered subcutaneous.
- mice Immediately after injection of PCP the mice are placed individually in special designed test cage (20 cm x 32 cm). The activity is measured by 5X8 infrared light sources and photocells spaced by 4 cm. The light beams cross the cage 1 .8 cm above the bottom of the cage. Recording of a motility count requires interruption of adjacent light beams, thus avoiding counts induced by stationary movements of the mice.
- Motility is recorded in 5 min intervals for a period of 1 hour.
- the drug effect is calculated on the total counts during the 1 hour behavioral test period in the following manner:
- the mean motility induced by vehicle treatment in the absence of PCP is used as baseline.
- the 100 per cent effect of PCP is accordingly calculated to be total motility counts minus baseline.
- the response in groups receiving test compound is thus determined by the total motility counts minus baseline, expressed in per cent of the similar result recorded in the parallel PCP control group.
- the per cent responses are converted to per cent inhibition.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Neurosurgery (AREA)
- Biomedical Technology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Medicinal Chemistry (AREA)
- Neurology (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
This invention is directed to compounds of formula I which are PDE10A enzyme inhibitors, pharmaceutical composition comprising a therapeutically effective amount of a compound of the invention and a pharmaceutically acceptable carrier and processes for the preparation of the compounds of formula I. The present invention further provides a method of treating a subject suffering from a neurodegenerative disorder, a drug addiction or a psychiatric disorder comprising administering to the subject a therapeutically effective amount of a compound of formula I. Formula I, wherein Q is selected from the group consisting of (Q1), (Q2), (Q3), (Q4), and (Q5).
Description
Quinazoline linked heteroaromatic tricycle derivatives as PDE10A enzyme inhibitors
Field of the Invention
The present invention provides heteroaromatic compounds that are PDE10A enzyme inhibitors, and as such are useful to treat neurodegenerative and psychiatric disorders. Especially, the invention provides compounds that are highly selective for PDE10 over other PDE subtypes. The present invention also provides pharmaceutical compositions comprising compounds of the invention and methods of treating disorders using the compounds of the invention.
Background of the Invention
The cyclic nucleotides cyclic-adenosine monophosphate (cAMP) and cyclic- guanosine monophosphate (cGMP) function as intracellular second messengers regulating a vast array of processes in neurons. Intracellular cAMP and cGMP are generated by adenyl and guanyl cyclases, and are degraded by cyclic nucleotide phosphodiesterases (PDEs) via hydrolysis of the cyclic nucleotides into their respective nucleotide monophosphates.
Phosphodieasterase 10A (PDE10A) is a dual-specificity phosphodiesterase that can convert both cAMP to AMP and cGMP to GMP (Soderling, S. et al. Proc. Natl. Acad. Sci. 1999, 96, 7071 -7076). PDE10A is primarily expressed in the neurons in the striatum, n. accumbens and in the olfactory tubercle (Kotera, J. et al. Biochem. Biophys. Res. Comm. 1999, 261, 551 -557 and Seeger, T.F. et al. Brain Research, 2003, 985, 1 13-126).
Studies indicate that within the brain, PDE10 expression is expressed at high levels by the medium spiny neurons (MSN) of the caudate nucleus, the accumbens nucleus and the corresponding neurons of the olfactory tubercle. MSN express two functional classes of neurons: the Di class expressing Di dopamine receptors and the D2 class expressing D2 dopamine receptors. The Di class of neurons is part of the 'direct' striatal output pathway, which broadly
functions to facilitate behavioral responses. The D2 class of neurons is part of the 'indirect' striatal output pathway, which functions to suppress behavioral responses that compete with those being facilitated by the 'direct' pathway. Dopamine D2 receptor antagonism is well established in the treatment of schizophrenia. Since the 1950's, dopamine D2 receptor antagonism has been the mainstay in psychosis treatment and all effective antipsychotic drugs antagonise D2 receptors. The effects of D2 are likely to be mediated primarily through neurons in the striatum, nucleus accumbens and olfactory tubercle, since these areas receive the densest dopaminergic projections and have the strongest expression of D2 receptors (Konradi, C. and Heckers, S. Society of Biological Psychiatry, 2001 , 50, 729-742).
Because PDE10A, in this context, has the desired expression profile with high and relatively specific expression in neurons in striatum, nucleus accumbens and olfactory tubercle, PDE10A inhibition is likely to have effects similar to D2 receptor antagonism and therefore have antipsychotic effects.
While PDE10A inhibition is expected to mimic D2 receptor antagonism in part, it might be expected to have a different profile. The D2 receptor has signaling components besides cAMP (Neve, K. A. et al. Journal of Receptors and Signal Transduction 2004, 24, 165-205), wherefore interference with cAMP through PDE10A inhibition may reduce the risk of the extrapyramidal side effects that are seen with strong D2 antagonism. Conversely, PDE10A inhibition may have some effects not seen with D2 receptor antagonism. PDE10A is also expressed in Di receptors expressing striatal neurons (Seeger, T. F. et al. Brain Research, 2003, 985, 1 13-126).
Further, since Di receptor agonism leads to stimulation of adenylate cyclase and resulting increase in cAMP levels, PDE10A inhibition is likely to also have effects that mimic Di receptor agonism.
Finally, PDE10A inhibition will not only increase cAMP in cells, but might also be expected to increase cGMP levels, since PDE10A is a dual specificity phosphodiesterase. cGMP activates a number of target protein in cells like cAMP and also interacts with the cAMP signaling pathways.
In conclusion, PDE10A inhibition is likely to mimic D2 receptor antagonism in part and therefore has antipsychotic effect, but the profile might differ from that observed with classical D2 receptor antagonists.
The PDE10A inhibitor papaverine is shown to be active in several antipsychotic models. Papaverine potentiated the cataleptic effect of the D2 receptor antagonist haloperidol in rats, but did not cause catalepsy on its own (WO 03/093499). Papaverine reduced hyperactivity in rats induced by PCP, while reduction of amphetamine induced hyperactivity was insignificant (WO 03/093499). These models suggest that PDE10A inhibition has the classic antipsychotic potential that would be expected from the theoretical considerations outlined above. WO 03/093499 further discloses the use of selective PDE10 inhibitors for the treatment of associated neurologic and psychiatric disorders. Furthermore, PDE10A inhibition reverses subchronic PCP-induced deficits in attentional set-shifting in rats (Rodefer et al. Eur. J. Neurosci. 2005, 4, 1070-1076). This model suggests that PDE10A inhibition might alleviate cognitive deficits associated with schizophrenia.
The tissue distribution of PDE10A indicates that PDE10A inhibitors can be used to raise levels of cAMP and/or cGMP within cells that express the PDE10A enzyme, especially neurons that comprise the basal ganglia, and the PDE10A inhibitors of the present invention would therefore be useful in treating a variety of associated neuropsychiatric conditions involving the basal ganglia such as neurological and psychiatric disorders, schizophrenia, bipolar disorder, psychosis, obsessive compulsive disorder and addiction, and may have the benefit of not possessing unwanted side effects, which are associated with the current therapies on the market.
Furthermore, recent publications (WO 2005/120514, WO 2005012485, Cantin et al, Bioorganic & Medicinal Chemistry Letters 17 (2007) 2869-2873) suggest that PDE10A inhibitors may be useful for treatment of obesity and non-insulin dependent diabetes.
Furthermore, recent publications suggest that PDE10A inhibitors may be useful for the treatment of Huntingtons Disease (Giampa et al. PLoS One 2010, 5(10), Giampa et al. Neurobiology of Disease (2009), 34(3), 450-456, Hebb et al. Current Opinion in Pharmacology 2007, 7(1 ), 86-92.)
Pyrrolodihydroisoquinolines and variants thereof are disclosed as inhibitors of PDE10 in WO 05/03129 and WO 05/02579. Piperidinyl-substituted quinazolines and isoquinolines that serve as PDE10 inhibitors are disclosed in WO 05/82883. WO 06/1 1040 discloses substituted quinazoline and isoquinoline compounds that serve as inhibitors of PDE10. US 20050182079 discloses substituted tetrahydroisoquinolinyl derivatives of quinazoline and isoquinoline that serve as effective phosphodiesterase (PDE) inhibitors. In particular, US 20050182079 relates to said compounds, which are selective inhibitors of PDE10. Analogously, US 20060019975 discloses piperidine derivatives of quinazoline and isoquinoline that serve as effective phosphodiesterase (PDE) inhibitors. US 20060019975 also relates to compounds that are selective inhibitors of PDE10. WO 06/028957 discloses cinnoline derivatives as inhibitors of PDE10 for the treatment of psychiatric and neurological syndromes. WO09/152825 discloses phenylimidazole derivatives as compounds that serve as inhibitors of PDE10.
However, these disclosures do not pertain to the compounds of the invention, which are structurally unrelated to any of the known PDE10 inhibitors (Kehler, J. et al. Expert Opin. Ther. Patents 2007, 17, 147-158), and which have now been found by the inventors to be highly active and selective PDE10A enzyme inhibitors.
The present invention provides compounds that are PDE10A enzyme
inhibitors and thus useful for treatment for neurodegenerative and/or psychiatric disorders, which are not efficacious in all patients. Hence, there remains a need for alternative methods of treatment. Summary of the Invention
The objective of the present invention is to provide compounds that are selective PDE10A enzyme inhibitors.
A further objective of the present invention is to provide compounds which have such activity, and which have improved solubility, metabolic stability and/or bioavailability compared to prior art compounds.
Another objective of the invention is to provide an effective treatment, in particular long-term treatment, of a human patient, without causing the side effects typically associated with current therapies for neurological and psychiatric disorders.
Further objectives of the invention will become apparent upon reading the present specification.
Detailed description of the invention
Embodiments of the invention In a first embodiment (E1 ) the present invention relates to compounds of formula I:
wherein Ri is selected from hydrogen, amine, Ci-C6 alkyl; Ci-C4 alkoxy, and trifluoromethyl wherein R2, R3, R4 and R5 are individually selected from the group consisting of hydrogen; halogen and dioxolo wherein -L- is a linker selected from -CH2-CH2-, _c=c_ , -CH=CH-, -S-CH2- and -CH2-S- wherein Q is selected from the group consisting of
(Q4), and
wherein R6, R7, R8, R9, R10, R1 1 and R12 are individually selected from the group consisting of hydrogen, halogen, Ci-C6 alkyl and Ci-C4 alkoxy, and wherein * denotes the attachment point, and tautomers and pharmaceutically acceptable salts thereof, and polymorphic forms thereof.
In an embodiment (E2) of embodiment (E1 ) -L- is -CH2-CH2-.
In an embodiment (E3) of any of embodiments (E1 ) and (E2) R1 is selected from the group consisting of hydrogen, methyl, methoxy, methylamine and triflourmethyl.
In an embodiment (E4) of any of embodiments (E1 ) and (E2) R2, R3, R4 and R5 are selected from the group consisting of fluorine and bromine.
In an embodiment (E5) of any of embodiments (E1 ) and (E2) R6, R7, R8, R9, R10, R1 1 and R12 are individually selected from the group consisting of hydrogen, fluorine and methoxy.
In a specific embodiment (E6) of any of embodiments (E1 ), (E2), (E3), (E4) and (E5) the compound is selected from the group of compounds listed in Table 1 .
In an embodiment (E7) of any of the previous embodiments the invention provides a compound of formula I, or a pharmaceutically acceptable salt thereof, for use as a medicament.
In an embodiment (E8) of any of the embodiments (E1 )-(E6) the present invention provides a pharmaceutical composition comprising a therapeutically effective amount of a compound of formula I and a pharmaceutically acceptable carrier, diluent or excipient.
In an embodiment (E9) of any of the embodiments (E1 )-(E6) the present invention provides the use of a compound of formula I, or a pharmaceutically acceptable salt thereof, for the preparation of a medicament for the treatment of a neurodegenerative or psychiatric disorder.
Furthermore, in an embodiment (E10) of any of the embodiments (E1 )-(E6) the present invention provides a method of treating a subject suffering from a
neurodegenerative disorder, comprising administering to the subject a therapeutically effective amount of a compound of formula I.
In an embodiment (E1 1 ) of any of the embodiments (E1 )-(E6) the present invention provides a method of treating a subject suffering from a psychiatric disorder, comprising administering to the subject a therapeutically effective amount of a compound of formula I.
In an embodiment (E12) of any of the embodiments (E1 )-(E6) the present invention provides a method of treating a subject suffering from a drug addiction, such as an alcohol, amphetamine, cocaine, or opiate addiction.
Definition of substitutents
As used in the context of the present invention, the terms "halo" and "halogen" are used interchangeably and refer to fluorine, chlorine, bromine or iodine.
The term "C1 -C6 alkyl" refers to a straight-chain or branched saturated hydrocarbon having from one to six carbon atoms, inclusive. Examples of such groups include, but are not limited to, methyl, ethyl, 1 -propyl, 2-propyl, 1 - butyl, 2-butyl, 2-methyl-2-propyl, 2-methyl-1 -butyl, and n-hexyl. The expression "C1 -C6 hydroxyalkyl" refers to a C1 -C6 alkyl group as defined above which is substituted with one hydroxy group. The term "halo(Ci - C6)alkyl" refers to a C1 -C6 alkyl group as defined above which is substituted with up to three halogen atoms, such as trifluoromethyl.
The expression "C1 -C6 alkoxy" refers to a straight-chain or branched saturated alkoxy group having from one to six carbon atoms, inclusive, with the open valency on the oxygen. Examples of such groups include, but are not limited to, methoxy, ethoxy, n-butoxy, 2-methyl-pentoxy and n-hexyloxy.
The term "C3-C8 cycloalkyi" typically refers to cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl. The expression "C1 -C6 alkyl(C3-C8)cycloalkyl" refers to a C3-C8 cycloalkyi as defined above which is
substituted with a straight-chain or branched C1-C6 alkyl. Examples of such groups include, but are not limited to, cyclopropylmethyl.
The term "heterocycloalkyl" refers to a four to eight membered ring containing carbon atoms and up to three N, O or S atoms, provided that the four to eight membered ring does not contain adjacent O or adjacent S atoms. The open valency is on either the heteroatom or carbon atom. Examples of such groups include, but are not limited to, azetidinyl, oxetanyl, piperazinyl, morpholinyl, thiomorpholinyl and [1 ,4]diazepanyl. The term "hydroxyheterocycloalkyl" refers to a heterocycloalkyl as defined above which is substituted with one hydroxy group. The term "C1-C6 alkyl-heterocycloalkyl" refers to a heterocycloalkyl as defined above which is substituted with a C1-C6 alkyl group. Examples of such groups include, but are not limited to, tetrahydropyran-4-yl-methyl and 2-morpholin-4-yl-ethyl.
The term "aryl" refers to a phenyl ring, optionally substituted with halogen, Ci- C6 alkyl, C1-C6 alkoxy or halo(Ci-Ce)alkyl as defined above. Examples of such groups include, but are not limited to, phenyl and 4-chlorophenyl. The term "d-Cearylalkyl" refers to an aryl as defined above which is substituted with a straight-chain or branched C1-C6 alkyl. Examples of such groups include, but are not limited to, benzyl and 4-chlorobenzyl.
Additionally, the present invention further provides certain embodiments of the invention that are described below.
In one embodiment of the invention, HET is a heteroaromatic group of formula II containing 2 nitrogen atoms. In another embodiment of the invention, HET is a heteroaromatic group of formula II containing 3 nitrogen atoms. In yet another embodiment of the invention, HET is a heteroaromatic group of formula II containing 4 nitrogen atoms.
In a further embodiment one or more of the hydrogen atoms of the compound of formula I have been substituted by deuterium.
In separate embodiments of the invention, the compound of formula I is selected among the following specific compounds listed in Table 1 and Table2, in the form of the free base, one or more tautomers thereof or a pharmaceutically acceptable salt thereof. Table 1 lists compounds of the invention where HET is a 2-quinazolinyl group of formula I la and the corresponding IC5o values determined as described in the section "PDE10A inhibition assay". Table 2 lists compounds of the invention where HET is a 4- quinazolinyl group of formula Mb and the corresponding IC5o values determined as described in the section "PDE10A inhibition assay". Each of the compounds constitutes an individual embodiment, of the present invention. It should be understood that the various aspects, embodiments, implementations and features of the invention mentioned herein may be claimed separately, or in any combination, as illustrated by the following non- limiting examples.
Table 1 : Compounds of the invention and IC5o valu
Compound ICso
(nM)
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 0.64
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 2.5
2-[2-(4-Methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 3
2-[2-(4-Ethoxy-8-fluoro-quinazolin-2-yl)-ethyl]-7-methoxy-5H-imidazo[2, 1- 830 a]isoindole
6-[2-(5H-lmidazo[2, 1-a]isoindol-2-yl)-ethyl]-8-methyl-[1 ,3]dioxolo[4,5- 20 g]quinazoline
2-[2-(6J-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 1
Compound ICso
(nM)
{6J-Difluoro-2-[2-(5H-imidazo[2 -a]isoindol-2-yl)-ethyl]-quinazolin-4-yl}- 19 methyl-amine
6-Fluoro-2-[2-(5H-imidazo[2, 1-a]isoindol-2-yl)-ethyl]-4-methyl-quinazolin-7- 25 ylamine
2-[2-(6-Fluoro-7-methoxy-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1- 9 a]isoindole
{8-Fluoro-2-[2-(5H-imidazo[2 -a]isoindol-2-yl)-ethyl]-quinazolin-4-yl}-methyl- 27 amine
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-7-methoxy-5H-imidazo[2, 1- 22 a]isoindole
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-6-methoxy-5H-imidazo[2, 1- 12 a]isoindole
9-Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1- 9.7 a]isoindole
7-Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1- 0.52 a]isoindole
8-Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1- 17 a]isoindole
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-9-methoxy-5H-imidazo[2, 1- 1 1 a]isoindole
6-Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1- 8.9 a]isoindole
2-[2-(8-Bromo-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 6.8
2-[2-(6,8-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 7.5
Compound ICso
(nM)
2-[2-(6,8-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-7-fluoro-5H-imidazo[2, 1- 4.4 a]isoindole
7-Fluoro-2-[2-(8-fluoro-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 50
2-[2-(8-fluoro-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1-a]isoindole 300
2-[2-(8-Fluoro-4-trifluoromethyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2, 1- 7.5 a]isoindole
7-Fluoro-2-[2-(8-fluoro-4-trifluoromethyl-quinazolin-2-yl)-ethyl]-5H- imidazo[2, 1-a]isoindole
In a particular embodiment of the present invention the compounds of the present invention have an IC5o value of less than 50 nM, such as in the range of 0.2 - 20 nM, particularly in the range of 0.2 - 10 nM, such as in the range of 0.2 - 5 nM or in the range of 0.2 - 1 nM.
Pharmaceutically Acceptable Salts
The present invention also comprises salts of the compounds, typically, pharmaceutically acceptable salts. Such salts include pharmaceutically acceptable acid addition salts. Acid addition salts include salts of inorganic acids as well as organic acids.
Representative examples of suitable inorganic acids include hydrochloric, hydrobromic, hydroiodic, phosphoric, sulfuric, sulfamic, nitric acids and the like. Representative examples of suitable organic acids include formic, acetic, trichloroacetic, trifluoroacetic, propionic, benzoic, cinnamic, citric, fumaric, glycolic, itaconic, lactic, methanesulfonic, maleic, malic, malonic, mandelic, oxalic, picric, pyruvic, salicylic, succinic, methane sulfonic, ethanesulfonic, tartaric, ascorbic, pamoic, bismethylene salicylic, ethanedisulfonic, gluconic,
citraconic, aspartic, stearic, palmitic, EDTA, glycolic, p-aminobenzoic, glutamic, benzenesulfonic, p-toluenesulfonic acids, theophylline acetic acids, as well as the 8-halotheophyllines, for example 8-bromotheophylline and the like. Further examples of pharmaceutically acceptable inorganic or organic acid addition salts include the pharmaceutically acceptable salts listed in Berge, S.M. et al., J. Pharm. Sci. 1977, 66, 2, the contents of which are hereby incorporated by reference.
Furthermore, the compounds of this invention may exist in unsolvated as well as in solvated forms with pharmaceutically acceptable solvents such as water, ethanol and the like. In general, the solvated forms are considered equivalent to the unsolvated forms for the purposes of this invention.
Pharmaceutical compositions
The present invention further provides a pharmaceutical composition comprising a therapeutically effective amount of a compound of formula I and a pharmaceutically acceptable carrier or diluent. The present invention also provides a pharmaceutical composition comprising a therapeutically effective amount of one of the specific compounds disclosed in the Experimental Section herein and a pharmaceutically acceptable carrier or diluent.
The compounds of the invention may be administered alone or in combination with pharmaceutically acceptable carriers, diluents or excipients, in either single or multiple doses. The pharmaceutical compositions according to the invention may be formulated with pharmaceutically acceptable carriers or diluents as well as any other known adjuvants and excipients in accordance with conventional techniques such as those disclosed in Remington: The Science and Practice of Pharmacy, 19th Edition, Gennaro, Ed., Mack Publishing Co., Easton, PA, 1995. The pharmaceutical compositions may be specifically formulated for administration by any suitable route such as oral, rectal, nasal, pulmonary, topical (including buccal and sublingual), transdermal, intracisternal,
intraperitoneal, vaginal and parenteral (including subcutaneous, intramuscular, intrathecal, intravenous and intradermal) routes. It will be appreciated that the route will depend on the general condition and age of the subject to be treated, the nature of the condition to be treated and the active ingredient.
Pharmaceutical compositions for oral administration include solid dosage forms such as capsules, tablets, dragees, pills, lozenges, powders and granules. Where appropriate, the compositions may be prepared with coatings such as enteric coatings or they may be formulated so as to provide controlled release of the active ingredient such as sustained or prolonged release according to methods well known in the art. Liquid dosage forms for oral administration include solutions, emulsions, suspensions, syrups and elixirs. Pharmaceutical compositions for parenteral administration include sterile aqueous and nonaqueous injectable solutions, dispersions, suspensions or emulsions as well as sterile powders to be reconstituted in sterile injectable solutions or dispersions prior to use. Other suitable administration forms include, but are not limited to, suppositories, sprays, ointments, creams, gels, inhalants, dermal patches and implants.
Typical oral dosages range from about 0.001 to about 100 mg/kg body weight per day. Typical oral dosages also range from about 0.01 to about 50 mg/kg body weight per day. Typical oral dosages further range from about 0.05 to about 10 mg/kg body weight per day. Oral dosages are usually administered in one or more dosages, typically, one to three dosages per day. The exact dosage will depend upon the frequency and mode of administration, the sex, age, weight and general condition of the subject treated, the nature and severity of the condition treated and any concomitant diseases to be treated and other factors evident to those skilled in the art.
The formulations may also be presented in a unit dosage form by methods known to those skilled in the art. For illustrative purposes, a typical unit dosage form for oral administration may contain from about 0.01 to about
1000 mg, from about 0.05 to about 500 mg, or from about 0.5 mg to about 200 mg.
For parenteral routes such as intravenous, intrathecal, intramuscular and similar administration, typical doses are in the order of half the dose employed for oral administration.
The present invention also provides a process for making a pharmaceutical composition comprising admixing a therapeutically effective amount of a compound of formula I and at least one pharmaceutically acceptable carrier or diluent. In an embodiment, of the present invention, the compound utilized in the aforementioned process is one of the specific compounds disclosed in the Experimental Section herein. The compounds of this invention are generally utilized as the free substance or as a pharmaceutically acceptable salt thereof. One example is an acid addition salt of a compound having the utility of a free base. When a compound of formula I contains a free base such salts are prepared in a conventional manner by treating a solution or suspension of a free base of formula I with a molar equivalent of a pharmaceutically acceptable acid. Representative examples of suitable organic and inorganic acids are described above.
For parenteral administration, solutions of the compounds of formula I in sterile aqueous solution, aqueous propylene glycol, aqueous vitamin E or sesame or peanut oil may be employed. Such aqueous solutions should be suitably buffered if necessary and the liquid diluent first rendered isotonic with sufficient saline or glucose. The aqueous solutions are particularly suitable for intravenous, intramuscular, subcutaneous and intraperitoneal administration. The compounds of formula I may be readily incorporated into known sterile aqueous media using standard techniques known to those skilled in the art.
Suitable pharmaceutical carriers include inert solid diluents or fillers, sterile aqueous solutions and various organic solvents. Examples of solid carriers include lactose, terra alba, sucrose, cyclodextrin, talc, gelatin, agar, pectin, acacia, magnesium stearate, stearic acid and lower alkyl ethers of cellulose. Examples of liquid carriers include, but are not limited to, syrup, peanut oil, olive oil, phospholipids, fatty acids, fatty acid amines, polyoxyethylene and water. Similarly, the carrier or diluent may include any sustained release material known in the art, such as glyceryl monostearate or glyceryl distearate, alone or mixed with a wax. The pharmaceutical compositions formed by combining the compounds of formula I and a pharmaceutically acceptable carrier are then readily administered in a variety of dosage forms suitable for the disclosed routes of administration. The formulations may conveniently be presented in unit dosage form by methods known in the art of pharmacy.
Formulations of the present invention suitable for oral administration may be presented as discrete units such as capsules or tablets, each containing a predetermined amount of the active ingredient, and optionally a suitable excipient. Furthermore, the orally available formulations may be in the form of a powder or granules, a solution or suspension in an aqueous or non-aqueous liquid, or an oil-in-water or water-in-oil liquid emulsion.
If a solid carrier is used for oral administration, the preparation may be tabletted, placed in a hard gelatin capsule in powder or pellet form or it may be in the form of a troche or lozenge. The amount of solid carrier will vary widely but will range from about 25 mg to about 1 g per dosage unit. If a liquid carrier is used, the preparation may be in the form of a syrup, emulsion, soft gelatin capsule or sterile injectable liquid such as an aqueous or non-aqueous liquid suspension or solution.
The pharmaceutical compositions of the invention may be prepared by conventional methods in the art. For example, tablets may be prepared by mixing the active ingredient with ordinary adjuvants and/or diluents and
subsequently compressing the mixture in a conventional tabletting machine prepare tablets. Examples of adjuvants or diluents comprise: corn starch, potato starch, talcum, magnesium stearate, gelatin, lactose, gums, and the like. Any other adjuvants or additives usually used for such purposes such as colorings, flavorings, preservatives etc. may be used provided that they are compatible with the active ingredients.
Therapeutically effective amount
In the present context, the term "therapeutically effective amount" of a compound means an amount sufficient to cure, alleviate or partially arrest the clinical manifestations of a given disease and its complications in a therapeutic intervention comprising the administration of said compound. An amount adequate to accomplish this is defined as "therapeutically effective amount". Effective amounts for each purpose will depend on the severity of the disease or injury as well as the weight and general state of the subject. It will be understood that determining an appropriate dosage may be achieved using routine experimentation, by constructing a matrix of values and testing different points in the matrix, which is all within the ordinary skills of a trained physician.
In the present context, the term "treatment" and "treating" means the management and care of a patient for the purpose of combating a condition, such as a disease or a disorder. The term is intended to include the full spectrum of treatments for a given condition from which the patient is suffering, such as administration of the active compound to alleviate the symptoms or complications, to delay the progression of the disease, disorder or condition, to alleviate or relief the symptoms and complications, and/or to cure or eliminate the disease, disorder or condition as well as to prevent the condition, wherein prevention is to be understood as the management and care of a patient for the purpose of combating the disease, condition, or disorder and includes the administration of the active compounds to prevent
the onset of the symptoms or complications. Nonetheless, prophylactic (preventive) and therapeutic (curative) treatments are two separate aspects of the invention. The patient to be treated is preferably a mammal, in particular a human being.
Treatment of Disorders
As mentioned above, the compounds of formula I are PDE10A enzyme inhibitors and as such are useful to treat associated neurological and psychiatric disorders.
The invention thus provides a compound of formula I or a pharmaceutically acceptable acid addition salt thereof, as well as a pharmaceutical composition containing such a compound, for use in the treatment of a neurodegenerative disorder, psychiatric disorder or drug addiction in humans.
In one embodiment of the present invention, the neurodegenerative disorder or condition involves neurodegeneration of striatal medium spiny neurons in a human. In a specific embodiment of the present invention, the neurodegenerative disorder or condition is Huntington's disease. In a further embodiment the disorder is dyskinesia associated with dopamine agonist therapy.
In an embodiment the psychiatric disorder is selected from the group consisting of schizophrenia, for example of the paranoid, disorganized, catatonic, undifferentiated, or residual type; schizophreniform disorder; schizoaffective disorder, for example of the delusional type or the depressive type; delusional disorder; substance-induced psychotic disorder, for example psychosis induced by alcohol, amphetamine, cannabis, cocaine, hallucinogens, inhalants, opioids, or phencyclidine; personality disorder of the paranoid type; and personality disorder of the schizoid type.
This invention further provides a method of treating a drug addiction, for example an alcohol, amphetamine, cocaine, or opiate addiction, in a human, which method comprises administering to said human an amount of a compound of formula I effective in treating addiction, such as drug addiction.
The term "drug addiction", as used herein, means an abnormal desire for a drug and is generally characterized by motivational disturbances such a compulsion to take the desired drug and episodes of intense drug craving. Other disorders that can be treated according to the present invention are obsessive/compulsive disorders, non-insuline demanding diabetes mellitus (NIDDM), and Tourette's syndrome and other tic disorders as well as Attention Deficit/Hyperactivity Disorder (ADHD). The compounds of formula I or pharmaceutically acceptable salts thereof may be used in combination with one or more other drugs (including typical and atypical antpsychotic agent) in the treatment of diseases or conditions for which the compounds of the present invention have utility, where the combination of the drugs together are safer or more effective than either drug alone. Additionally, the compounds of the present invention may be used in combination with one or more other drugs that treat, prevent, control, ameliorate, or reduce the risk of side effects or toxicity of the compounds of the present invention. The combinations, uses and methods of treatment of the invention may also provide advantages in treatment of patients who fail to respond adequately or who are resistant to other known treatments.
Such other drugs may be administered, by a route and in an amount commonly used therefore, contemporaneously or sequentially with the compounds of the present invention. Accordingly, the pharmaceutical compositions of the present invention include those that contain one or more other active ingredients, in addition to the compounds of the present invention. The combinations may be administered as part of a unit dosage form combination product, or as a kit or treatment protocol wherein one or more
additional drugs are administered in separate dosage forms as part of a treatment regimen.
The term "neuroleptic agent" as used herein refers to drugs, which have the effect on cognition and behaviour of antipsychotic agent drugs that reduce confusion, delusions, hallucinations, and psychomotor agitation in patients with psychoses. Also known as major tranquilizers and antipsychotic drugs, neuroleptic agents include, but are not limited to: typical antipsychotic drugs, including phenothiazines, further divided into the aliphatics, piperidines, and piperazines, thioxanthenes (e.g., cisordinol), butyrophenones (e.g., haloperidol), dibenzoxazepines (e.g., loxapine), dihydroindolones (e.g., molindone), diphenylbutylpiperidines (e.g., pimozide), and atypical antipsychotic drugs, including benzisoxazoles (e.g., risperidone), sertindole, olanzapine, quetiapine, osanetant and ziprasidone.
Particularly preferred neuroleptic agents for use in the invention are sertindole, olanzapine, risperidone, quetiapine, aripiprazole, haloperidol, clozapine, ziprasidone and osanetant. As used herein, and unless otherwise indicated, a "neurodegenerative disorder or condition" refers to a disorder or condition that is caused by the dysfunction and/or death of neurons in the central nervous system. The treatment of these disorders and conditions can be facilitated by administration of an agent which prevents the dysfunction or death of neurons at risk in these disorders or conditions and/or enhances the function of damaged or healthy neurons in such a way as to compensate for the loss of function caused by the dysfunction or death of at-risk neurons. The term "neurotrophic agent" as used herein refers to a substance or agent that has some or all of these properties.
All references, including publications, patent applications and patents, cited herein are hereby incorporated by reference in their entirety and to the same extent as if each reference were individually and specifically indicated to be
incorporated by reference and were set forth in its entirety (to the maximum extent permitted by law).
Headings and sub-headings are used herein for convenience only, and should not be construed as limiting the invention in any way.
The use of any and all examples, or exemplary language (including "for instance", "for example", "e.g.", and "as such") in the present specification is intended merely to better illuminate the invention, and does not pose a limitation on the scope of invention unless otherwise indicated.
The citation and incorporation of patent documents herein is done for convenience only, and does not reflect any view of the validity, patentability and/or enforceability of such patent documents.
The present invention includes all modifications and equivalents of the subject-matter recited in the claims appended hereto, as permitted by applicable law. The invention disclosed herein is further illustrated by the following non- limiting examples.
Experimental Section
General Methods
Analytical LC-MS data were obtained using one of the following methods.
Method 131 :
A PE Sciex API 150EX instrument equipped with atmospheric pressure photo ionisation and a Shimadzu LC-8A SLC-10A LC system was used. Column: 4.6 x 30 mm Waters Symmetry C18 column with 3.5 μιτι particle size; Column temperature: 60 °C; Solvent system: A = water/trifluoroacetic acid (100:0.05)
and B = water/ acetonitrile/trifluoroacetic acid (5:95:0.035); Method: Linear gradient elution with A:B = 90:10 to 0:100 in 2.4 minutes and with a flow rate of 3.3 mL/min. Method 132
LC-MS were run on a Sciex API150EX equipped with APPI-source operating in positive ion mode. The HPLC consisted of Shimadzu LC10-ADvp LC pumps. SPD-M20A PDA detector (operating at 254 nM) and SCL-10A system controller. Autosampler was Gilson 215. Colomn oven was a Jones Chromatography 7990R and ELS detector was a Sedere Sedex 85.
LC-conditions: The column was a Waters Symmetry C-18. 4.6 x 30 mm. 3.5μ operating at 60 °C with 2.5 ml/min of a binary gradient consisting of water + 0.05 % TFA (A) and methanol + 0.05 % TFA. Gradient:
Total run time: 2.8 min Method 350
LC-MS were run on a Sciex API300 equipped with APPI source operating in positive ion mode. The UPLC consisted of Waters Aquity including column mamager. binary solvent manager, sample organizer. PDA detector (operating at 254 nM) and ELS detector.
LC-conditions: The column was a Waters Aquity UPLC BEH C-18. 2.1 x 50 mm. 1 .7 μιτι operating at 60 °C with 1 .2 ml/min of a binary gradient consisting of water + 0.05 % TFA (A) and 95 % acetonitrile containing 5 % water + 0.03 % TFA.
Gradient Time. min. %B
0.00 10.0
1 .00 100.0
1 .01 10.0
1 .15 10.0
Total run time 1 .15 min
Method WXV-AB05/ WXV-AB10
LC-MS were run on an Agilent 1 100 LCMS system with ELS Detector, Pump G131 1A, Degasser G1379A,Well-plate Autosampler, G1367A, Column Oven G1316A, DAD G1315B,MSD G1946C or G1956A,Alltech ELSD 800,Column YMC ODS-AQ,Particle size 5 micrometer, Pore size 12 nm, Dimension 50 * 2.0 mm ID, Injection volume 2 microliter, Column temperature 50°C, Flow 0.8 ml/min. Mobile phase: A=0.1 % TFA in water, B=0.05% TFA in Acetonitril,C=10 mmol/l NH4HCO3 in water, D=Acetonitril, Total run time 4.5 min, Gradien linear. UV Detection
Wavelength=254 nm,ELSD Detection Temperature: 50°C, Gas Pressure: 3.2 bar.
TIME A% B%
WXV-AB05 0 95 5
3,5 0 100
3,55 95 5
TIME A% B%
WXV-AB10 0 90 10
3,4 0 100
3,5 0 100
3,51 90 10
Preparative LC-MS-purification was performed on a PE Sciex API 150EX instrument with atmospheric pressure chemical ionization. Column: 50 X 20 mm YMC ODS-A with 5 μιτι particle size; Method: Linear gradient elution with A:B = 80:20 to 0:100 in 7 minutes and with a flow rate of 22.7 mL/minute. Fraction collection was performed by split-flow MS detection.
1H NMR spectra were recorded at 500.13 MHz on a Bruker Avance AV500 instrument or at 250.13 MHz on a Bruker Avance DPX250 instrument. TMS was used as internal reference standard. Chemical shift values are expressed in ppm. The following abbreviations are used for multiplicity of NMR signals: s = singlet, d = doublet, t = triplet, q = quartet, qui = quintet, h = heptet, dd = double doublet, dt = double triplet, dq = double quartet, tt = triplet of triplets, m = multiplet, br s = broad singlet and br = broad signal. Abbreviations are in accordance with to the ACS Style Guide: "The ACS Styleguide - A manual for authors and editors" Janet S. Dodd, Ed. 1997, ISBN: 0841234620.
Preparative LC-MS-purification was performed on a PE Sciex API 150EX instrument with atmospheric pressure chemical ionization. Column: 50 X 20 mm YMC ODS-A with 5 μιτι particle size; Method: Linear gradient elution with A:B = 80:20 to 0:100 in 7 minutes and with a flow rate of 22.7 mL/minute. Fraction collection was performed by split-flow MS detection. 1H NMR spectra were recorded at 500.13 MHz on a Bruker Avance AV500 instrument or at 250.13 MHz on a Bruker Avance DPX250 instrument. TMS was used as internal reference standard. Chemical shift values are expressed in ppm. The following abbreviations are used for multiplicity of NMR signals: s = singlet, d = doublet, t = triplet, q = quartet, qui = quintet, h = heptet, dd = double doublet, dt = double triplet, dq = double quartet, tt = triplet of triplets, m = multiplet, br s = broad singlet and br = broad signal.
Abbreviations are in accordance with to the ACS Style Guide: "The ACS Styleguide - A manual for authors and editors" Janet S. Dodd, Ed. 1997, ISBN: 0841234620. General: p-Toluene-sulfonyl hydrazide (98%) was from Avocado. 2-Phenyl - 1 H-lmidazole-4-Carbaldehyde was from ASDI. Solvents and standard chemicals were from sigma-aldrich.
Preparation of the compounds of the invention
Compounds of the general formula I of the invention may be prepared as described in the following reaction schemes. Unless otherwise indicated, in the reaction schemes and discussion that follow, Q, R1 -R1 1 , and -L- are as defined above.
Compounds of formula I, wherein -L- is -CH2-S-, can be prepared by the coupling of a nucleophile of formula IV with an electrophile of formula III (exemplified with the use of Q1 and analogues reactions can be used with Q2- Q5), where X is a leaving group, e.g. CI, Br, I, methanesulfonyl, 4- toluenesulfonyl, as shown in scheme 1 .
III
Scheme 1.
This reaction is typically carried out in a solvent such as 1 -propanol, toluene, DMF, or acetonitrile, optionally in the presence of a carbonate base such as potassium carbonate or a tertiary amine base such as triethylamine or diisopropylethylannine (DIPEA), at a temperature ranging from about 0°C to about 200°C, optionally under pressure in a closed vessel. Other suitable solvents include benzene, chloroform, dioxane, ethyl acetate, 2-propanol and xylene. Alternatively, solvent mixtures such as toluene/2-propanol can be used. Thiol-compounds of formula IV are either commercially available or can be made by methods similar to those described in the literature e.g. Journal of Heterocyclic Chemistry 1977 14(5), 889-92, Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry 1979, 5, 1 132-6, Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio- Organic Chemistry 1997, 20, 2983-2988, Organic & Biomolecular Chemistry 2009, 7, 128-134.
Electrophiles of formula III can be prepared as described in e.g. Venkatesan, A. et al. ChemMedChem 2008, 3, 1658-1661 .
Compounds of formula I, wherein -L- is -CH=CH- or -CH2-CH2- can be prepared by the reaction sequence shown in scheme 2 (exemplified with the use of Q1 and analogues reactions can be used with Q2-Q5) where X is a leaving group, e.g. CI, Br, I, methanesulfonyl, 4-toluenesulfonyl, as shown.
Scheme 2.
Specifically, compounds of formula I, wherein -L- is -CH2-CH2- can be prepared by reduction of an alkene of formula I, wherein -L- is -CH=CH- by hydrogenation using a transition metal catalyst, such as palladium metal, together with a hydrogen source, such as hydrogen gas, ammonium hydrogen carbonate, or cyclohexadiene. Alternatively the reduction can be performed using tosylhydrazide TsNHNH2 in dimethylformamide at elevated temperature e.g. 130 °C. Said alkenes of formula I, wherein -L- is -CH=CH- can be prepared by the Wittig reaction between a phosphonium salt of formula VII and an aldehyde of formula VIII in a suitable solvent, such as tetrahydrofuran or ethanol, in the presence of a suitable base, such as sodium hydride or 1 ,8- diazabicyclo[5.4.0]undec-7-ene. Phosphonium salt of formula VII are readily available by reaction of compounds of formula V with triphenylphosphine by methods known to chemists skilled in the art. Aldehydes of formula VIII are available by methods described in the literature se e.g. Venkatesan, A. et al. ChemMedChem 2008, 3, 1658-1661 .
Compounds of formula I, wherein R1 is an amine or an alkyl substituent, or an alkoxy substituent, or a phenyl or heteroaryl can be prepared by the reaction sequence shown in scheme 3.
Scheme 3.
Compounds of formula I, wherein -L- is -CH2-CH2- can be prepared by the method described in scheme 4, where an orthoacyl-anilide of formula XI is treated with ammonia under cyclisative conditions.
Scheme 4.
This reaction is typically carried out in a suitable solvent, such as methanol and performed by mixing a solution of orthoacyl-anilide of formula XI in 7M NH3/MeOH solution and heating at 50°C for 6-24h.
The orthoacyl-anilide of formula XI can be prepared from commercially available substituted anthranilic acids XII by standard organic synthesis methods known in the art as shown in scheme 5.
Preparation of intermediates
Syntheses of intermediates
6-Fluoro-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
2E
A solution of the compound 2A (50 g, 0.37 mol) in CCI4 (500 mL) was added the NBS (72.5 g, 0.408 mol) and the AIBN (1 .2 g, 0.037 mol), the reaction solution was refluxed for overnight, TLC indicated the reaction was completed, the reaction solution was filtered and concentrated in vacuum to give the crude product which was purified by flash silica chromatography eluting with petroleum ether/EtOAc (500:1 ) to give the desired compound 2B (30 g, yield: 40%) as a white solid.
To a solution of compound 2B (60 g, 0.28 mol) and compound 1 C (78 g, 0.44 mol) in DMF (500 mL) was added Cs2CO3 (150 g, 0.46 mol). The mixture was stirred at r.t (12 °C) for 2 hours. TLC indicated that all the starting material 2B was consumed completely. The mixture was poured into water (2 L), the precipitated product was filtered, washed with water (500 mL) and methanol (500 mL) to give the product 2C (85 g, yield: 92%) as a white solid.
A mixture of compound 2C (130 g, 0.466 mol) and KOH (80 g, 1 .43 mol) in EtOH (1 .8 L) was heated at 90°C for 0.5 hour. TLC showed that the reaction was completed. The mixture was cooled and filtered, the yellowish filtrate was concentrated in vacuum to give a brown solid, which was diluted with EtOAc (1 L) and water (300 ml_), the aqueous layer was extracted by EtOAc (300 ml_x5), the combined organic layer was washed by brine (300 ml_), dried over anhydrous Na2SO4, concentrated in vacuum to give the product 2D (65 g, crude) as brown solid, which is pure enough for next step reaction. A mixture of compound 2D (25 g, crude) and compound 1 F (43 g, 0.185 mol) in dry CH3CN (500 ml_) was stirred at r.t. (27°C) overnight. The resulting mixture was filtered and washed with MeCN, and dired in vacuum over to afford compound 2E (37 g, yield: 85%). A solution of compound 2E (23 g, 60 mmol) and Et3N (7.27 g, 72 mmol) in dry CH3CN (400 ml_) was stirred at 90°C -100 °C reflux for 18 hours. The reaction solution was concentrated. The residue was diluted with EtOAc, and washed with 20% aqueous potassium hydrogen carbonate solution. After filtration through a pad of Celite, the organic layer was dried (MgSO4) and concentrated in vacuum. The residue was purified by silica column chromatography (eluted with PE:EtOAc = 5:1 ) to give 6-Fluoro- 5H-imidazo[2,1 -a]isoindole-2-carbaldehyde (1 .1 g, yield: 10%) as a yellowish solid. 1H NMR (CDCI3 400 MHz): 59.9 (s, 1 H), 7.89 (s, 1 H), 7.69 (d, J = 7.6Hz, 1 H), 7.50-7.44 (m, 1 H), 7.1 1 (m, 1 H), 5.04 (s, 2H).
The following intermediates were made in a similar way:
5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
7-Fluoro-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
8- Fluoro-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
9- Fluoro-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
6- Methoxy-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
7- Methoxy-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
8-Methoxy-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
9-Methoxy-5H-imidazo[2,1 -a]isoindole-2-carbaldehyde
5H-lmidazo[1 ',2':1 ,5]pyrrolo[3,4-b]pyridine-2-carbaldehyde (i.e. 6-aza-5H- imidazo[2,1 -a]isoindole-2-carbaldehyde)
8H-3,6,8a-Triaza-cyclopenta[a]indene-2-carbaldehyde (i.e. 7-aza-5H- imidazo[2,1 -a]isoindole-2-carbaldehyde)
8H-3,5,8a-Triaza-cyclopenta[a]indene-2-carbaldehyde (i.e. 8-aza-5H- imidazo[2,1 -a]isoindole-2-carbaldehyde)
5H-lmidazo[1 ',2':1 ,2]pyrrolo[3,4-b]pyridine-2-carbaldehyde (i.e. 9-aza-5H- imidazo[2,1 -a]isoindole-2-carbaldehyde)
2-methyl-6-fluoro-4,8-dimethyl-quinazoline triphenylphosphonium chloride
e (669 mg, 2.69 mmol) and Tnphenylphosphine (846 mg, 3.22 mmol) was dissolved in Acetonitrile (10 mL, 200 mmol) and the mixture was refluxed over night. Evaporation in vacuo gave 2-methyl-6-fluoro-4,8-dimethyl-quinazoline triphenylphosphonium chloride (3. 1 .312 g; 100 %)
The following intermediates were made in a similar way:
4-Methyl-quinazoline triphenylphosphonium chloride
4-Ethoxy-8-fluoro-quinazoline triphenylphosphonium chloride
8-methyl-[1 ,3]dioxolo[4,5-g]quinazoline triphenylphosphonium chloride 8-Fluoro-4-methyl-quinazoline triphenylphosphonium chloride
7,8-difluoro-4-methyl-quinazoline triphenylphosphonium chloride
8-Fluoro-4-methyl-quinazoline triphenylphosphonium chloride
8-Bromo-4-methyl-quinazoline triphenylphosphonium chloride
6,8-Difluoro-4-methyl-quinazoline triphenylphosphonium chloride
6,8-Difluoro-4-methyl-quinazoline triphenylphosphonium chloride
8-Fluoro-4-trifluoromethyl-quinazoline triphenylphosphonium chloride
7-Fluoro-4-trifluoromethyl-quinazoline triphenylphosphonium chloride N-(2-Acetyl-4,5-difluoro-phenyl)-3-(5H-imidazo[2,1 -a]isoindol-2-yl)-propi onamide
A slurry of 3-(5H-lmidazo[2,1 -a]isoindol-2-yl)-propionic acid (0.303 g, 1 .33 mmol) in thionyl chloride (10 mL, 100 mmol) and one drop DMF was heated at 50 °C for 4h. The volatiles were removed in vacuo and the residue rotovaped twice from acetonitrile. The residue was dissolved in Toluene (40 mL, 400 mmol) where upon 1 -(2-Amino-4,5-difluoro-phenyl)-ethanone (0.227 g, 1 .33 mmol) was added followed by and the solution stirred at reflux for 12h. LCMS indicated messy product. The volatiles were removed in vacuo and water was added and the organics extracted with EtOAc. The combined organics were washed with water, brine, dried (MgSO4) filtered and the volatiles removed in vacuo. The residue was purified by flash chromatography Eluent EtOAc to EtOAc/MeOH/TEA, 87:10:3 and the product fractions collected and evaporated to yield [C] N-(2-Acetyl-4,5-difluoro-phenyl)-3-(5H-imidazo[2,1 - a]isoindol-2-yl)-propionamide (97 mg; Yield = 14%; Purity = 72.2%)which was used without further purification.
General procedure for preparing 2-[(E)-2-(4-methyl-quinazolin-2-yl)- vinyl]-5H-imidazo[2,1 -a]isoindoles (take 2-[(E)-2-(8-Fluoro-4-methyl- uinazolin-2-yl)-vinyl]-5H-imidazo[2,1 -a]isoindole as example):
To (8-Fluoro-4-methyl-quinazolin-2-ylmethyl)-triphenyl-phosphonium; chloride (760 mg, 1 .3 mmol) and 5H-lmidazo[2,1 -a]isoindole-2-carbaldehyde (279 mg, 1 .24 mmol) in Ethanol (20 mL, 300 mmol) was added 1 ,8- Diazabicyclo[5.4.0]undec-7-ene (220 uL, 1 .5 mmol), the reaction was stirred at RT for 6 hours. The reaction mixture was Poured into water, extracted with
DCM, washed with Brine, dried with Na2SO4, filtered and cone. In vacuo to give 495 mg crude material, which was purified by column chromatography on silicagel using to give 2-[(E)-2-(8-Fluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H- imidazo[2,1 -a]isoindole (30 mg).
The following compounds were made in an analogues way:
2-[(E)-2-(4-Methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 -a]isoindole
2-[(E)-2-(4-Ethoxy-8-fluoro-quinazolin-2-yl)-vinyl]-7-methoxy-5H-imidazo[2,1 - a]isoindole
6-[(E)-2-(5H-lmidazo[2,1 -a]isoindol-2-yl)-vinyl]-8-methyl-[1 ,3]dioxolo[4,5- g]quinazoline
2-[(E)-2-(6,7-Difluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 - a]isoindole
{6,7-Difluoro-2-[(E)-2-(5H-imidazo[2,1 -a]isoindol-2-yl)-vinyl]-quinazolin-4-yl}- methyl-amine
6- Fluoro-2-[(E)-2-(5H-imidazo[2,1 -a]isoindol-2-yl)-vinyl]-4-methyl-quinazolin-7- ylamine
2-[(E)-2-(6-Fluoro-7-methoxy-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 - a]isoindole
{8-Fluoro-2-[(E)-2-(5H-imidazo[2,1 -a]isoindol-2-yl)-vinyl]-quinazolin-4-yl}- methyl-amine
2-[(E)-2-(8-Fluoro-4-methyl-quinazolin-2-yl)-vinyl]-7-methoxy-5H-imidazo[2,1 - a]isoindole
2-[(E)-2-(8-Fluoro-4-methyl-quinazolin-2-yl)-vinyl]-6-methoxy-5H-imidazo[2,1 - a]isoindole
9-Fluoro-2-[(E)-2-(8-fluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 - a]isoindole
7- Fluoro-2-[(E)-2-(8-fluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 - a]isoindole
8-Fluoro-2-[(E)-2-(8-fluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 - a]isoindole
2-[(E)-2-(8-Fluoro-4-methyl-quinazolin-2-yl)-vinyl]-9-methoxy-5H-imidazo[2,1 - a]isoindole
6- Fluoro-2-[(E)-2-(8-fluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 - a]isoindole
2-[(E)-2-(8-Bromo-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 -a]isoin 2-[(E)-2-(6,8-Difluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 - a]isoindole
2-[(E)-2-(6,8-Difluoro-4-methyl-qui^
a]isoindole
7- Fluoro-2-[(E)-2-(8-fluoro-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 -a]isoindole 2-[(E)-2-(8-fluoro-quinazolin-2-yl)-vinyl]-5H-imidazo[2,1 -a]isoindole
2-[(E)-2-(8-Fluoro-4-trifluoromethyl-quinazolin-2-yl)-vinyl]-5H-imidazo[2 a]isoindole
7-Fluoro-2-[(E)-2-(8-fluoro-4-trifluoronnethyl-quinazolin-2-yl)-vinyl]-5H- imidazo[2,1 -a]isoindole
Syntheses of compounds of the invention
Example 1
General procedure for preparing 2-[2-(4-methyl-quinazolin-2-yl)ethyl]-5H- imidazo[2,1 -a]isoindoles (take 6-[2-(5H-lmidazo[2,1 -a]isoindol-2-yl)- ethyl]-8-methyl-1 ,3-dioxolo[4,5-g]quinazoline as example):
A solution of 6-[(E)-2-(5H-lmidazo[2,1 -a]isoindol-2-yl)-vinyl]-8-methyl-1 ,3- dioxolo[4,5-g]quinazoline (0.197 g, 0.448 mmol) in MeOH (20 mL), DCM (10 mL) and AcOH (5 mL) was treated with 10% Pd/C (50% wet) 100 mg and the slurry was shaken on the parr apparatus for 5h. The solids were removed and the volatiles removed in vacuo. The residue was purified by flash chromatography Eluent EtOAc:EtOAc/MeOH (9:1 ) and the product fractions collected and evaporated to yield 6-[2-(5H-lmidazo[2,1 -a]isoindol-2-yl)-ethyl]-
8-methyl-1 ,3-dioxolo[4,5g]quinazoline (0.061 g; Yield Purity = 96.3%) as a white solid.
Example 2
General procedure for preparing 2-[2-(4-methyl-quinazolin-2-yl)ethyl]-5H- imidazo[2,1 -a]isoindoles (take 2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)- eth l]-5H-imidazo[2,1-a]isoindole as example):
A solution of 2-[(E)-2-(8-Fluoro-4-methyl-quinazolin-2-yl)-vinyl]-5H- imidazo[2,1 -a]isoindole (30 mg) in methanol (5 ml_) was passed through a H- Cube® Continuous-flow Hydrogenation Reactor (ThalesNano) at a flow rate of 1 mL/min through a small cartridge of 10% Pd/C (THS01 1 1 1 ) with an internal temperature of 25°C and 1 bar of hydrogen pressure. Evaporation of the volatiles afforded 2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H- imidazo[2,1 -a]isoindole LC-MS: m/z = 345,1 (MH+), tR = 0,93 min, method = 131 .
The following compounds were made in an analogues way:
2-[2-(4-Methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole LC-MS: m/z = 327,2 (MH+), tR = 0,92 min, method = 131
2-[2-(4-Ethoxy-8-fluoro-quinazolin-2-yl)-ethyl]-7-methoxy-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 405,4 (MH+), tR = 1 ,21 min, method = 131
6-[2-(5H-lmidazo[2,1 -a]isoindol-2-yl)-ethyl]-8-methyl-[1 ,3]dioxolo[4,5- g]quinazoline LC-MS: m/z = 371 (MH+), tR = 0,81 min, method = 131
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-7-methoxy-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 374,8 (MH+), tR = 0,99 min, method = 131
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-6-methoxy-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 374,8 (MH+), tR = 1 ,03 min, method = 131
9-Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 363,1 (MH+), tR = 0,92 min, method = 131
7- Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 363,2 (MH+), tR = 0,93 min, method = 131
8- Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 363,1 (MH+), tR = 0,95 min, method = 131
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-9-methoxy-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 374,8 (MKT), tR = 1 min, method = 131
6- Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 363 (MH+), tR = 0,94 min, method = 131
2-[2-(8-Bromo-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole LC- MS: mlz = 405,3 (MKT), tR = 1 ,05 min, method = 131
2-[2-(6,8-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole LC-MS: m/z = 363,3 (MH+), tR = 0,95 min, method = 131
2-[2-(6,8-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-7-fluoro-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 381 ,2 (MH+), tR = 0,99 min, method = 131
7-Fluoro-2-[2-(8-fluoro-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole LC- MS: m/z = 349,2 (MKT), tR = 0,86 min, method = 131
2-[2-(8-fluoro-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole LC-MS: mlz = 331 ,1 (MH+), tR = 1 ,3 min, method = 131
2-[2-(8-Fluoro-4-thfluoromethyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 399,1 (MKT), tR = 1 ,14 min, method = 131
7- Fluoro-2-[2-(8-fluoro-4-thfluoromethyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole LC-MS: m/z = 417,4 (MKT), tR = 0,95 min, method = 131
Example 3
{8-Fluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-quinazolin-4-yl}- meth l-amine:
A solution of 8-Fluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-3H- quinazolin-4-one (200 mg, 0.6 mmol) in Acetonitrile (6.4 mL, 120 mmol) was
treated with phosphonitrilic chloride trimer (201 mg, 0.577 mmol) and Triethylamine (402 uL, 2.89 mmol). The solution was allowed to stir at RT o/n. Methylamine (0.0359 g, 1 .15 mmol) (2M solution in THF) was added and the solution allowed to stir at RT o/n. The volatiles were removed in vacuo and the residue purified by column chromatography 100% EtOAc to MeOH:EtOAc:TEA 5:93:2. This yielded a mixture of the starting material and the product. The fractions were evaporated and the residue purified by prep HPLC. The product fractions were evaporated to yield
{8-Fluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-quinazolin-4-yl}-methyl- amine (1 12 mg; Yield = 50.4%; Purity = 93.4%) as an off white solid.
LC-MS: m/z = 360,2 (MKT), tR = 0,58 min, method = 131
The following compounds were made in an analogues way:
{6,7-Difluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-quinazolin-4-yl}- methyl-amine LC-MS: m/z = 378,3 (MKT), tR = 0,62 min, method = 131
Example 4
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1- a isoindole:
A slurry of 8-Fluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-3H-quinazolin- 4-one (0.244 g, 0.704 mmol) in Phosphoryl chloride (10 ml_, 100 mmol) was heated at reflux for 30 mins. LCMS indicated complete conversion to 2-[2-(4- Chloro-8-fluoro-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole. The excess Phosphoryl chloride was removed in vacuo and the residue evaporated from acetonitrile 3 times. The residue was dissolved in dry THF (30 ml_) where upon 1 ,1 '-bis(diphenylphosphino)ferrocene-palladium(ll) dichloride dichloromethane complex (0.0575 g, 0.0704 mmol) was added followed by methylmagnesium bromide (0.924 g, 7.75 mmol). The solution was stirred for 6h. Water was added and the organics extracted with EtOAc. The combined
organics were washed with water, brine, dried (MgSO4) filtered and the volatiles removed in vacuo. The residue was purified by flash chromatography Eluent EtOAc 100% to EtOAc:MeOH (9:1 ) and the product fractions collected and evaporated to yield 2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H- imidazo[2,1 -a]isoindole (55 mg).
Example 5 2-[2-(6,7-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1- a]isoindole
A solution of N-(2-Acetyl-4,5-difluoro-phenyl)-3-(5H-imidazo[2,1 -a]isoindol-2- yl)-propionamide (97 mg, 0.18 mmol) in 7M NH3/MeOH was heated in MW for 30 mins at 130°C, LCMS indicated incomplete reaction. Heated a further 30 mins at 130°C then the volatiles evaporated and the residue was purified by flash chromatography Eluent EtOAc:EtOAc:MeOH:TEA 85:10:5 and the product fractions collected and evaporated to yield 2-[2-(6,7-Difluoro-4-methyl- quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole (21 mg; Yield = 29%; Purity = 91 .2%). LC-MS: m/z = 363,1 (MH+), tR = 1 ,03 min, method = 131
The following compounds where isolated from the same experiment:
6-Fluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-4-methyl-quinazolin-7- ylamine (1 .6 mg; Yield = 2.0%), LC-MS: m/z = 360,2 (MKT), tR = 0,6 m method = 131
2-[2-(6-Fluoro-7-methoxy-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole (1 .7 mg; Yield = 2.1 %; Purity = 85.1 %), LC-MS: m/z = 374,9 (MKT), tR = 0,99 min, method = 131
Pharmacological Testing PDE10A enzyme
Active PDE10A enzyme is prepared in a number of ways for use in PDE assays (Loughney, K. et al. Gene 1999, 234, 109-1 17; Fujishige, K. et al. Eur J Biochem. 1999, 266, 1 1 18-1 127 and Soderling, S. et al. Proc. Natl. Acad. Sci. 1999, 96, 7071 -7076). PDE10A can be expressed as full-length proteins or as truncated proteins, as long as they express the catalytic domain. PDE10A can be prepared in different cell types, for example insect cells or E. coli. An example of a method to obtain catalytically active PDE10A is as follows: The catalytic domain of human PDE10A (amino acids 440-779 from the sequence with accession number NP 006652) is amplified from total human brain total RNA by standard RT-PCR and is cloned into the BamH1 and Xho1 sites of the pET28a vector (Novagen). Expression in coli is performed according to standard protocols. Briefly, the expression plasmids are transformed into the BL21 (DE3) E. coli strain, and 50 ml_ cultures inoculated with the cells allowed to grow to an OD600 of 0.4-0.6 before protein expression is induced with 0.5mM IPTG. Following induction, the cells are incubated overnight at room temperature, after which the cells are collected by centrifugation. Cells expressing PDE10A are resuspended in 12 ml_ (50 mM TRIS-HCI-pH8.0, 1 mM MgCI2 and protease inhibitors). The cells are lysed by sonication, and after all cells are lysed, TritonX100 is added according to Novagen protocols. PDE10A is partially purified on Q sepharose and the most active fractions were pooled.
PDE10A inhibition assay
A PDE1 OA assay may for example, be performed as follows: The assay is performed in 60 uL samples containing a fixed amount of the relevant PDE enzyme (sufficient to convert 20-25% of the cyclic nucleotide substrate), a buffer (50 mM HEPES7.6; 10mM MgCI2; 0.02% Tween20), 0.1 mg/ml BSA, 225 pCi of 3H-labelled cyclic nucleotide substrate, tritium labeled cAMP to a final concentration of 5 nM and varying amounts of inhibitors. Reactions are
initiated by addition of the cyclic nucleotide substrate, and reactions are allowed to proceed for one hr at room temperature before being terminated through mixing with 15 uL 8 mg/mL yttrium silicate SPA beads (Amersham). The beads are allowed to settle for one hr in the dark before the plates are counted in a Wallac 1450 Microbeta counter. The measured signal can be converted to activity relative to an uninhibited control (100 %) and IC5o values can be calculated using the Xlfit extension to EXCEL.
Phencyclidine (PCP) induced hyperactivity
Male mice (NMRI, Charles River) weighing 20-25g are used. Eight mice are used in each group receiving the test compound (5 mg/kg) plus PCP (2.3 mg/kg) including the parallel control groups receiving the vehicle of the test compound plus PCP or vehicle injections only. The injection volumen is 10 ml/kg. The experiment is made in normal light conditions in an undisturbed room. The test substance is injected per oss 60 min before injection of PCP, which is administered subcutaneous.
Immediately after injection of PCP the mice are placed individually in special designed test cage (20 cm x 32 cm). The activity is measured by 5X8 infrared light sources and photocells spaced by 4 cm. The light beams cross the cage 1 .8 cm above the bottom of the cage. Recording of a motility count requires interruption of adjacent light beams, thus avoiding counts induced by stationary movements of the mice.
Motility is recorded in 5 min intervals for a period of 1 hour. The drug effect is calculated on the total counts during the 1 hour behavioral test period in the following manner:
The mean motility induced by vehicle treatment in the absence of PCP is used as baseline. The 100 per cent effect of PCP is accordingly calculated to be total motility counts minus baseline. The response in groups receiving test compound is thus determined by the total motility counts minus baseline, expressed in per cent of the similar result recorded in the parallel PCP control
group. The per cent responses are converted to per cent inhibition.
Claims
What is claimed:
1 . A compound having the structure of formula I
wherein Ri is selected from hydrogen, amine, Ci-C6 alkyl; Ci-C4 alkoxy, and trifluoromethyl wherein R2, R3, R4 and R5 are individually selected from the group consisting of hydrogen; halogen and dioxolo wherein -L- is a linker selected from -CH2-CH2-, , -CH=CH-
S-CH2-and -CH2-S- wherein Q is selected from the group consisting of
(Q4), and (Q5)
wherein R6, R7, R8, R9, R10, R1 1 and R12 are individually selected from the group consisting of hydrogen, halogen, C1-C6 alkyl and Ci-C4 alkoxy and wherein * denotes the attachment point, and tautomers and pharmaceutically acceptable salts thereof, and polymorphic forms thereof
The compound of claim 1 wherein the linker -L- is -CH2-CH2-.
The compound of claim 1 wherein R1 is selected from the group consisting of hydrogen, methyl, methoxy, methylamine and triflourmethyl.
The compound of claim 1 wherein one or more of R2, R3, R4 and R5 is a halogen selected from the group consisting of fluorine and bromine.
5. The compound of claim 1 wherein R6, R7, R8, R9, R10, R1 1 and R12 are individually selected from the group consisting of hydrogen, fluorine and methoxy.
6. The compound of any of claims 1 -5 wherein Q is
7. The compound of any of claims 1 -6 selected from the group of
compounds consisting of
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
2-[2-(4-Methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 -a]isoindole;
2-[2-(4-Ethoxy-8-fluoro-quinazolin-2-yl)-ethyl]-7-nnethoxy-5H- imidazo[2,1 -a]isoindole;
6-[2-(5H-lmidazo[2,1 -a]isoindol-2-yl)-ethyl]-8-methyl-[1 ,3]dioxolo[4,5- g]quinazoline;
2-[2-(6,7-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
{6,7-Difluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-quinazolin-4- yl}-methyl-annine;
6- Fluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-4-methyl- quinazolin-7-ylamine;
2-[2-(6-Fluoro-7-methoxy-4-methyl-quinazolin-2-yl)-ethyl]-5H- imidazo[2,1 -a]isoindole;
{8-Fluoro-2-[2-(5H-imidazo[2,1 -a]isoindol-2-yl)-ethyl]-quinazolin-4-yl}- methyl-amine;
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-7-methoxy-5H- imidazo[2,1 -a]isoindole;
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-6-methoxy-5H- imidazo[2,1 -a]isoindole;
9-Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
7- Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
8- Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
2-[2-(8-Fluoro-4-methyl-quinazolin-2-yl)-ethyl]-9-methoxy-5H- imidazo[2,1 -a]isoindole;
6- Fluoro-2-[2-(8-fluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
2-[2-(8-Bromo-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
2-[2-(6,8-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
2-[2-(6,8-Difluoro-4-methyl-quinazolin-2-yl)-ethyl]-7-fluoro-5H- imidazo[2,1 -a]isoindole;
7- Fluoro-2-[2-(8-fluoro-quinazolin-2-yl)-ethyl]-5H-imidazo[2,1 - a]isoindole;
2-[2-(8-fluoro-quinazolin-2-yl)-ethyl]-5H-innidazo[2,1 -a]isoindole;
2-[2-(8-Fluoro-4-trifluoromethyl-quinazolin-2-yl)-ethyl]-5H-imidazo[^ a]isoindole; and
7-Fluoro-2-[2-(8-fluoro-4-thfluoronnethyl-quinazolin-2-yl)-ethyl]-5H- imidazo[2,1 -a]isoindole
The compound of any of claims 1 -7, or a pharmaceutically acceptable salt thereof, for use as a medicament.
9. A pharmaceutical composition comprising a therapeutically effective amount of the compound of any of claims 1 -7, and a pharmaceutically acceptable carrier, diluent or excipient.
10. The compound of any of claims 1 -7, or a pharmaceutically acceptable salt thereof, for the preparation of a medicament for the treatment of a neurodegenerative or psychiatric disorder.
1 1 . A method of treating a subject suffering from a neurodegenerative or psychiatric disorder, comprising administering to the subject a therapeutically effective amount of a compound of any of claims 1 -7.
12. The compound of any one of claims 1 -7, for use in the treatment of a neurodegenerative or psychiatric disorder, alone or in combination with one or more neuroleptic agents such as sertindole, olanzapine, risperidone, quetiapine, aripiprazole, haloperidol, clozapine, ziprasidone and osanetant, selected from the group consisting of ADHD, cognitive deficits, cognitive deficits associated with schizophrenia, schizophrenia, schizoaffective disorder, bipolar disorder, OCD, obesity, non-insulin dependent diabetes, addiction and Huntington's disease.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161541113P | 2011-09-30 | 2011-09-30 | |
| DKPA201100750 | 2011-09-30 | ||
| US61/541,113 | 2011-09-30 | ||
| DKPA201100750 | 2011-09-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013045607A1 true WO2013045607A1 (en) | 2013-04-04 |
Family
ID=47994312
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2012/069179 Ceased WO2013045607A1 (en) | 2011-09-30 | 2012-09-28 | Quinazoline linked heteroaromatic tricycle derivatives as pde10a enzyme inhibitors |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2013045607A1 (en) |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003093499A2 (en) | 2002-05-03 | 2003-11-13 | Pfizer Products Inc. | Therapeutic use of selective pde10 inhibitors |
| WO2005003129A1 (en) | 2003-06-30 | 2005-01-13 | Altana Pharma Ag | Pyrrolodihydroisoquinolines as pde10 inhibitors |
| WO2005002579A1 (en) | 2003-06-30 | 2005-01-13 | Altana Pharma Ag | Pyrrolo-dihydroisoquinoline derivatives as pde10 inhibitors |
| WO2005012485A2 (en) | 2003-07-31 | 2005-02-10 | Bayer Pharmaceuticals Corporation | Methods for treating diabetes and related disorders using pde10a inhibitors |
| US20050182079A1 (en) | 2004-02-18 | 2005-08-18 | Pfizer Inc | Tetrahydroisoquinolinyl derivatives of quinazoline and isoquinoline |
| WO2005120514A1 (en) | 2004-06-07 | 2005-12-22 | Pfizer Products Inc. | Phosphodiesterase 10 inhibition as treatment for obesity-related and metabolic syndrome-related conditions |
| US20060019975A1 (en) | 2004-07-23 | 2006-01-26 | Pfizer Inc | Novel piperidyl derivatives of quinazoline and isoquinoline |
| WO2006028957A1 (en) | 2004-09-03 | 2006-03-16 | Memory Pharmaceuticals Corporation | 4-substituted 4, 6-dialkoxy-cinnoline derivatives as phospodiesterase 10 inhibitors for the treatment of psychiatric or neurological syndroms |
| WO2006072828A2 (en) * | 2005-01-07 | 2006-07-13 | Pfizer Products Inc. | Heteroaromatic quinoline compounds and their use as pde10 inhibitors |
| WO2008001182A1 (en) * | 2006-06-26 | 2008-01-03 | Pfizer Products Inc. | Tricyclic heteroaryl compounds as pde10 inhibitors |
| WO2009152825A1 (en) | 2008-06-20 | 2009-12-23 | H. Lundbeck A/S | Novel phenylimidazole derivatives as pde10a enzyme inhibitors |
| WO2010030027A1 (en) * | 2008-09-10 | 2010-03-18 | Mitsubishi Tanabe Pharma Corporation | Aromatic nitrogen-containing 6-membered ring compounds and their use |
| WO2012065612A1 (en) * | 2010-11-19 | 2012-05-24 | H. Lundbeck A/S | Imidazole derivatives as pde10a enzyme inhibitors |
-
2012
- 2012-09-28 WO PCT/EP2012/069179 patent/WO2013045607A1/en not_active Ceased
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003093499A2 (en) | 2002-05-03 | 2003-11-13 | Pfizer Products Inc. | Therapeutic use of selective pde10 inhibitors |
| WO2005003129A1 (en) | 2003-06-30 | 2005-01-13 | Altana Pharma Ag | Pyrrolodihydroisoquinolines as pde10 inhibitors |
| WO2005002579A1 (en) | 2003-06-30 | 2005-01-13 | Altana Pharma Ag | Pyrrolo-dihydroisoquinoline derivatives as pde10 inhibitors |
| WO2005012485A2 (en) | 2003-07-31 | 2005-02-10 | Bayer Pharmaceuticals Corporation | Methods for treating diabetes and related disorders using pde10a inhibitors |
| US20050182079A1 (en) | 2004-02-18 | 2005-08-18 | Pfizer Inc | Tetrahydroisoquinolinyl derivatives of quinazoline and isoquinoline |
| WO2005082883A2 (en) | 2004-02-18 | 2005-09-09 | Pfizer Products Inc. | Tetrahydroisoquinolinyl derivatives of quinazoline and isoquinoline |
| WO2005120514A1 (en) | 2004-06-07 | 2005-12-22 | Pfizer Products Inc. | Phosphodiesterase 10 inhibition as treatment for obesity-related and metabolic syndrome-related conditions |
| US20060019975A1 (en) | 2004-07-23 | 2006-01-26 | Pfizer Inc | Novel piperidyl derivatives of quinazoline and isoquinoline |
| WO2006011040A1 (en) | 2004-07-23 | 2006-02-02 | Pfizer Products Inc. | Quinazolin-4-yl- piperidine and cinnolin-4-yl- piperidine derivatives as pde10 inhibitors for the treatment of cns disorders |
| WO2006028957A1 (en) | 2004-09-03 | 2006-03-16 | Memory Pharmaceuticals Corporation | 4-substituted 4, 6-dialkoxy-cinnoline derivatives as phospodiesterase 10 inhibitors for the treatment of psychiatric or neurological syndroms |
| WO2006072828A2 (en) * | 2005-01-07 | 2006-07-13 | Pfizer Products Inc. | Heteroaromatic quinoline compounds and their use as pde10 inhibitors |
| WO2008001182A1 (en) * | 2006-06-26 | 2008-01-03 | Pfizer Products Inc. | Tricyclic heteroaryl compounds as pde10 inhibitors |
| WO2009152825A1 (en) | 2008-06-20 | 2009-12-23 | H. Lundbeck A/S | Novel phenylimidazole derivatives as pde10a enzyme inhibitors |
| WO2010030027A1 (en) * | 2008-09-10 | 2010-03-18 | Mitsubishi Tanabe Pharma Corporation | Aromatic nitrogen-containing 6-membered ring compounds and their use |
| WO2012065612A1 (en) * | 2010-11-19 | 2012-05-24 | H. Lundbeck A/S | Imidazole derivatives as pde10a enzyme inhibitors |
Non-Patent Citations (22)
| Title |
|---|
| "Remington: The Science and Practice of Pharmacy", 1995, MACK PUBLISHING CO. |
| BERGE, S.M. ET AL., J. PHARM. SCI., vol. 66, 1977, pages 2 |
| CANTIN ET AL., BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 17, 2007, pages 2869 - 2873 |
| FUJISHIGE, K. ET AL., EUR J BIOCHEM., vol. 266, 1999, pages 1118 - 1127 |
| GIAMPA ET AL., NEUROBIOLOGY OF DISEASE, vol. 34, no. 3, 2009, pages 450 - 456 |
| GIAMPA ET AL., PLOS ONE, vol. 5, 2010, pages 10 |
| HEBB ET AL., CURRENT OPINION IN PHARMACOLOGY, vol. 7, no. 1, 2007, pages 86 - 92 |
| JANET S. DODD: "ACS Style Guide", 1997, article "The ACS Styleguide - A manual" |
| JOURNAL OF HETEROCYCLIC CHEMISTRY, vol. 14, no. 5, 1977, pages 889 - 92 |
| JOURNAL OF THE CHEMICAL SOCIETY, PERKIN TRANSACTIONS 1: ORGANIC AND BIO- ORGANIC CHEMISTRY, vol. 20, 1997, pages 2983 - 2988 |
| JOURNAL OF THE CHEMICAL SOCIETY, PERKIN TRANSACTIONS 1: ORGANIC AND BIO-ORGANIC CHEMISTRY, vol. 5, 1979, pages 1132 - 6 |
| KEHLER, J. ET AL., EXPERT OPIN. THER. PATENTS, vol. 17, 2007, pages 147 - 158 |
| KONRADI, C.; HECKERS, S., SOCIETY OF BIOLOGICAL PSYCHIATRY, vol. 50, 2001, pages 729 - 742 |
| KOTERA, J. ET AL., BIOCHEM. BIOPHYS. RES. COMM., vol. 261, 1999, pages 551 - 557 |
| LOUGHNEY, K. ET AL., GENE, vol. 234, 1999, pages 109 - 117 |
| NEVE, K. A. ET AL., JOURNAL OF RECEPTORS AND SIGNAL TRANSDUCTION, vol. 24, 2004, pages 165 - 205 |
| ORGANIC & BIOMOLECULAR CHEMISTRY, vol. 7, 2009, pages 128 - 134 |
| RODEFER ET AL., EUR. J. NEUROSCI., vol. 4, 2005, pages 1070 - 1076 |
| SEEGER, T. F. ET AL., BRAIN RESEARCH, vol. 985, 2003, pages 113 - 126 |
| SEEGER, T.F. ET AL., BRAIN RESEARCH, vol. 985, 2003, pages 113 - 126 |
| SODERLING, S. ET AL., PROC. NATL. ACAD. SCI., vol. 96, 1999, pages 7071 - 7076 |
| VENKATESAN, A. ET AL., CHEMMEDCHEM, vol. 3, 2008, pages 1658 - 1661 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2640731B1 (en) | Imidazole derivatives as pde10a enzyme inhibitors | |
| CA2783727C (en) | 2-arylimidazole derivatives as pde10a enzyme inhibitors | |
| EP2513106A1 (en) | Heteroaromatic aryl triazole derivatives as pde10a enzyme inhibitors | |
| WO2013050527A1 (en) | Quinazoline derivatives as pde10a enzyme inhibitors | |
| US8796304B2 (en) | Tricyclic imidazole compounds as PDE10 inhibitors | |
| US9801878B2 (en) | Quinoline derivatives as PDE10A enzyme inhibitors | |
| WO2013045607A1 (en) | Quinazoline linked heteroaromatic tricycle derivatives as pde10a enzyme inhibitors | |
| HK1189593B (en) | Imidazole derivatives as pde10a enzyme inhibitors | |
| OA16430A (en) | Imidazole derivatives as PDE10A ensyme inhibitors | |
| WO2013127817A1 (en) | Imidazole derivatives as pde10a enzyme inhibitors | |
| MX2013005592A (en) | Imidazole derivatives as pde10a enzyme inhibitors. | |
| WO2013107856A1 (en) | Imidazole derivatives as pde10a enzyme inhibitors | |
| NZ626279B2 (en) | Quinoline derivatives as pde10a enzyme inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12766314 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12766314 Country of ref document: EP Kind code of ref document: A1 |