WO2012110402A1 - Trisazo compounds, processes for preparing them and use thereof - Google Patents
Trisazo compounds, processes for preparing them and use thereof Download PDFInfo
- Publication number
- WO2012110402A1 WO2012110402A1 PCT/EP2012/052239 EP2012052239W WO2012110402A1 WO 2012110402 A1 WO2012110402 A1 WO 2012110402A1 EP 2012052239 W EP2012052239 W EP 2012052239W WO 2012110402 A1 WO2012110402 A1 WO 2012110402A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- phenyl
- trione
- tribenzo
- tris
- hydroxy
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC(C*)=CCN Chemical compound CC(C*)=CCN 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B35/00—Disazo and polyazo dyes of the type A<-D->B prepared by diazotising and coupling
- C09B35/378—Trisazo dyes of the type
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G5/00—Recording members for original recording by exposure, e.g. to light, to heat, to electrons; Manufacture thereof; Selection of materials therefor
- G03G5/02—Charge-receiving layers
- G03G5/04—Photoconductive layers; Charge-generation layers or charge-transporting layers; Additives therefor; Binders therefor
- G03G5/06—Photoconductive layers; Charge-generation layers or charge-transporting layers; Additives therefor; Binders therefor characterised by the photoconductive material being organic
- G03G5/0664—Dyes
- G03G5/0675—Azo dyes
- G03G5/0687—Trisazo dyes
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G5/00—Recording members for original recording by exposure, e.g. to light, to heat, to electrons; Manufacture thereof; Selection of materials therefor
- G03G5/02—Charge-receiving layers
- G03G5/04—Photoconductive layers; Charge-generation layers or charge-transporting layers; Additives therefor; Binders therefor
- G03G5/06—Photoconductive layers; Charge-generation layers or charge-transporting layers; Additives therefor; Binders therefor characterised by the photoconductive material being organic
- G03G5/0664—Dyes
- G03G5/0675—Azo dyes
- G03G5/0687—Trisazo dyes
- G03G5/069—Trisazo dyes containing polymethine or anthraquinone groups
Definitions
- Trisazo compounds process for their preparation and their use
- the invention relates to novel trisazo compounds which are used as pigments e.g. can be used as Fotoieiter in the charge-generating layer of an electrophotographic device, processes for their preparation and their use in an electrophotographic device.
- Such electrophotographic apparatuses have been known for some time and in the simplest case consist of a layer containing an organic photogenerating compound and one provided with a conductive layer
- organic photoconductors used in such electrophotographic devices must meet a variety of partially exclusive criteria of photosensitivity, conductivity, and mechanical strength.
- the division of the photoconductive layer into a charge generating layer and an overlying charge transport layer enabled the use of new ones
- azo pigments are used in the charge-generating layer in addition to phthalocyanines, squarines, perylenes and a number of other substances.
- phthalocyanines squarines
- perylenes a number of other substances.
- diamines and naphtholic couplers have been used to prepare electrophotography
- Trisazo pigments e.g. tris (4-aminopheny! amine as an amine component (Law, K.-Y. et al., US-A 5,422,211 1995).
- the synthesis of trisazo dyes by coupling the
- Diazonium salt of the corresponding aromatic Triaminoverbundung with a variety of possible naphtholic couplers allows a broad optimization of the electrophotographic properties of the compounds by a suitable choice of triamines and couplers.
- Trisazo dyes the backbone of the 5H-tribenzo (a, f, k) trinden-5,10, 5-trione, are described in the patent DE 196 54 492 A1. Starting from the trinitro compound of the 5H-tribenzo (a, f, k), 5,10,15-trione, which has been reported by R. Seka et al. Already synthesized in 943, the corresponding tris-amino compound and the diazonium salt were prepared and trisazo compounds which absorb in the wavelength range above 500 nm with magenta couplers customary in the field of azochrome and for use as Charge generation Mater 'tal suitable with green or red lasers for analog copiers with white light source or digital copiers and printers.
- Modern digital printers or copiers use as their light source laser diodes which radiate in the near IR range.
- the charge-generating materials used in the OPC drums of these devices are predominantly phthalocyanine pigments and occasionally trisazo pigments.
- toner grain Essentially determined by the toner grain. At the present time, however, there are already toners of such small grain size that more and more the wavelength of the laser used becomes the limiting factor. Blue lasers would be e.g. able to ensure a higher resolution.
- Phthalocyanine pigments and azo compounds with magenta and cyan couplers are already described as charge generation materials for this range (US 2003049551A, Xerox) but have neither sufficient absorption in the short wavelength blue nor are they sufficiently active electrophotographically.
- the invention therefore an object of the invention to find new compounds that are used as dyes or charge-generating material in electrophotographic
- R 4l R5 and RB are identical or different hydrogen, lower alkyl, lower alkoxy or halogen and
- R-i, R2 and R3 are identical or different radicals (couplers) of the general formula 2
- R A represents a phenyl radical which is mono- or polysubstituted by lower alkyl
- R is a lower alkyl radical, preferably methyl, ethyl or propyl, or an optionally mono- or polysubstituted by lower alkyl, lower alkoxy or halogen
- R 1 to R 3 are the same or different per se
- aryl group (R a ) may be mono- or polysubstituted with R and the substituents for R - may be as defined under R a -.
- radicals R 1 to R 3 are identical.
- Preferred radicals R a are phenyl, optionally mono- or polysubstituted with substituents R, which are selected from Cl, F, methoxy, phenoxy and / or phenylcarbonyl.
- Preferred radicals R are phenyl or methyl.
- the radicals R 4 , R 5 and R 6 are preferably hydrogen.
- lower alkyl alkyl radicals having 1-8 carbon atoms, such as methyl, ethyl, propyl, isopropyl, butyl, sec-butyl, pentyl, etc.
- lower alkoxy alkoxy radicals having 1-8 carbon atoms, such as, methoxy, ethoxy, propoxy, butoxy, etc.
- Halogen is a fluoro, chloro, bromo or iodo substituent.
- novel tnsazo compounds can be prepared by coupling a diazonium salt of an aromatic triamino compound of general formula 3
- the trisdiazonium salts of formula 3 are prepared by diazotization of the corresponding triamines also under conditions known per se, e.g. by adding sodium nitrite to the hydrochloric acid solution of the respective
- Triamino compound at below 5 ° C and addition of an aqueous alkali or
- the triamino compounds are prepared in a known manner from the trinitro compounds by reduction, e.g. under
- the coupler compounds of general formula 2 are commercially available, e.g. by Acros Organics, Belgium.
- the abovementioned new trisazo dyes are used as pigments individually or as a mixture of at least two components in different mixing ratios, each component being able to be up to 90% in the charge generation layer
- the electrophotographic device comprises at least one surface provided with a conductive surface, an overlying charge generator layer containing the abovementioned trisazo compounds, optionally applied as a dispersion, and an overlying charge transport layer comprising at least one applied conventional charge transport compound, such as N, N " -Bis (3 methylphenyl -N, N 'bis-containing ipheny ⁇ benzidine.
- the new trisazo compounds lead when used as photosensitive
- Substances in a carrier generation layer of an electrophotographic device surprisingly result in drastically increased photosensitivity at 400 to 480 nm, preferably at 400 to 450 nm, in the emission of blue
- Laser diodes With this they can e.g. find use in laser printers.
- Trisazo dyes 1 to 8 were determined in a bilayer system.
- an aluminized polyester substrate was coated with a dispersion which contains the respective trisazo compound as pigments in a polyvinyl butyral solution in methylene chloride. The obtained in this way after drying
- Carrier generation layers were coated (with a solution of N, N "-bis- 3- methylphenylJ-NN '-bis-ipheny ⁇ benzidine in polycarbonate Z200 in methylene chloride and measured after drying.
Landscapes
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Photoreceptors In Electrophotography (AREA)
Abstract
Description
Trisazoverbindungen, Verfahren zu ihrer Herstellung und ihre Verwendung Trisazo compounds, process for their preparation and their use
Die Erfindung betrifft neue Trisazoverbindungen, die als Pigmente z.B. als Fotoieiter in der ladungserzeugenden Schicht einer elektrofotografischen Vorrichtung eingesetzt werden können, Verfahren zu ihrer Herstellung sowie ihre Verwendung in einer elektrofotografischen Vorrichtung. The invention relates to novel trisazo compounds which are used as pigments e.g. can be used as Fotoieiter in the charge-generating layer of an electrophotographic device, processes for their preparation and their use in an electrophotographic device.
Derartige elektrofotografische Vorrichtungen sind seit längerer Zeit bekannt und bestehen im einfachsten Falle aus einer Schicht, die eine organische fotoieitende Verbindung enthält und einem mit einer leitfähigen Schicht versehenen Such electrophotographic apparatuses have been known for some time and in the simplest case consist of a layer containing an organic photogenerating compound and one provided with a conductive layer
Basismaterial. Base material.
Die in derartigen elektrofotografischen Vorrichtungen verwendeten organischen Fotoleiter müssen jedoch einer Vielzahl sich teilweise ausschließende Kriterien hinsichtlich Lichtempfindlichkeit, Leitfähigkeit und mechanischer Festigkeit gerecht werden. However, the organic photoconductors used in such electrophotographic devices must meet a variety of partially exclusive criteria of photosensitivity, conductivity, and mechanical strength.
Die Aufteilung der fotoleitenden Schicht in eine ladungserzeugende Schicht und eine darüberliegende Ladungstransportschicht ermöglichte den Einsatz neuer The division of the photoconductive layer into a charge generating layer and an overlying charge transport layer enabled the use of new ones
lichtempfindlicher Materialien unabhängig von der Optimierung der Photosensitive materials regardless of the optimization of the
Ladungstransport-Eigenschaften und der mechanischen Parameter. Charge transport properties and mechanical parameters.
Als lichtempfindliche Substanzen werden in der ladungserzeugenden Schicht neben Phthalocyaninen, Squarinen, Perylenen und einer Reihe weiterer Substanzen auch Azopigmente eingesetzt. Eine breite Palette von Diaminen und naphtholischen Kupplern wurden zur Herstellung von für die Elektrofotografie geeigneten As photosensitive substances, azo pigments are used in the charge-generating layer in addition to phthalocyanines, squarines, perylenes and a number of other substances. A wide range of diamines and naphtholic couplers have been used to prepare electrophotography
Bisazopigmenten eingesetzt. Später wurden auch Trisazopigmente z.B. tris(4- aminopheny!)amin als Aminkomponente (Law, K.-Y. et. al. US-A 5,422,211 1995) eingesetzt. Die Synthese von Trisazo-Farbstoffen durch Kupplung des Bisazo pigments used. Later, trisazo pigments, e.g. tris (4-aminopheny!) amine as an amine component (Law, K.-Y. et al., US-A 5,422,211 1995). The synthesis of trisazo dyes by coupling the
Diazoniumsalzes der entsprechenden aromatischen Triaminoverbundung mit einer Vielzahl möglicher naphtholischer Kuppler erlaubt eine breite Optimierung der elektrofotografischen Eigenschaften der Verbindungen durch geeignete Wahl von Triaminen und Kupplern. Diazonium salt of the corresponding aromatic Triaminoverbundung with a variety of possible naphtholic couplers allows a broad optimization of the electrophotographic properties of the compounds by a suitable choice of triamines and couplers.
Trisazo-Farbstoffe, deren Grundgerüst das 5H-Tribenzo(a,f,k)trinden-5,10, 5-trion darstellt, werden in der Patentschrift DE 196 54 492 A1 beschrieben. Ausgehend von der Trinitroverbindung des 5H-Tribenzo(a,f,k)trinden-5,10,15-trion, welche von R.Seka et al. bereits 943 synthetisiert wurde, wurden die entsprechende Tris- Amino-Verbindung und das Diazoniumsalz hergestellt und mit in der Azochemie üblichen Purpurkupplern zu Trisazo-Verbindungen, welche im Wellenlängenbereich oberhalb von 500 nm absorbieren und für den Einsatz als Ladungserzeugungsmater'tal für analoge Kopiergeräte mit Weißlichtquelle oder digitale Kopierer und Printer mit grünen oder roten Lasern geeignet sind. Trisazo dyes, the backbone of the 5H-tribenzo (a, f, k) trinden-5,10, 5-trione, are described in the patent DE 196 54 492 A1. Starting from the trinitro compound of the 5H-tribenzo (a, f, k), 5,10,15-trione, which has been reported by R. Seka et al. Already synthesized in 943, the corresponding tris-amino compound and the diazonium salt were prepared and trisazo compounds which absorb in the wavelength range above 500 nm with magenta couplers customary in the field of azochrome and for use as Charge generation Mater 'tal suitable with green or red lasers for analog copiers with white light source or digital copiers and printers.
Moderne digitale Drucker oder Kopierer verwenden als Lichtquelle Laserdioden, welche im nahen IR-Bereich strahlen. Als Ladungserzeugungsmatenalien werden in den OPC Trommeln dieser Geräte überwiegend Phthalocyanin-Pigmente und vereinzelt Trisazo-Pigmente verwendet. Die Biidqualität der Verfahren, Modern digital printers or copiers use as their light source laser diodes which radiate in the near IR range. The charge-generating materials used in the OPC drums of these devices are predominantly phthalocyanine pigments and occasionally trisazo pigments. The bi-quality of the methods,
charakterisiert durch das erreichbare Auflösungsvermögen, wurde bisher im Characterized by the achievable resolution, was previously in
Wesentlichen durch die Tonerkörn igkeit bestimmt. Gegenwärtig gibt es jedoch bereits Toner mit so geringer Korngröße, dass mehr und mehr die Weilenlänge des verwendeten Lasers zum begrenzenden Faktor wird. Blaue Laser wären z.B. in der Lage ein höheres Auflösungsvermögen zu gewährleisten. Essentially determined by the toner grain. At the present time, however, there are already toners of such small grain size that more and more the wavelength of the laser used becomes the limiting factor. Blue lasers would be e.g. able to ensure a higher resolution.
Bisher waren diese jedoch zu kostenintensiv. Mit der zunehmenden Verbreitung der Blue ray DVD Technologie entstand aber ein Massenmarkt für derartige So far, these have been too costly. But with the increasing popularity of the Blue ray DVD technology, a mass market for such has emerged
Laserdioden, so dass deren Verwendung auch in Laserdruckern aus Kostensicht vertretbar wird. Erforderlich werden damit Ladungserzeugungsmatenalien mit einer ausgeprägten Absorption im Bereich von 400 bis 450 nm. Laser diodes, so that their use in laser printers from a cost point of view is justifiable. Thus, charge generation materials with a pronounced absorption in the range of 400 to 450 nm are required.
Phthalocyanin-Pigmente und Azo-Verbindungen mit Purpur und Blaugrünkupplern sind bereits als Ladungserzeugungsmatenalien für diesen Bereich beschrieben (US 2003049551A, Xerox), haben aber in dem kurzwelligen blauen Bereich weder eine ausreichende Absorption noch sind sie elektrofotografisch ausreichend aktiv. Phthalocyanine pigments and azo compounds with magenta and cyan couplers are already described as charge generation materials for this range (US 2003049551A, Xerox) but have neither sufficient absorption in the short wavelength blue nor are they sufficiently active electrophotographically.
Der Einsatz von handelsüblichen Azo-Geibkuppiern ergab jedoch nur eine geringe hypsochrome Verschiebung. However, the use of commercially available azo-geibkuppiern revealed only a small hypsochromic shift.
Der Erfindung lag deshalb die Aufgabe zugrunde, neue Verbindungen zu finden, die als Farbstoffe oder Ladungserzeugungsmaterial in elektrofotografischen The invention therefore an object of the invention to find new compounds that are used as dyes or charge-generating material in electrophotographic
Vorrichtungen im kurzwelligen (blauen) Bereich einsetzbar sind. Überraschend führte die Kupplung von Truxenchinon-Diazoniumsalzen mit an sich bekannten Gelbkupplern, welche bei der Herstellung von fotografischen Filmen Verwendung finden, zu entsprechenden Verbindungen, die als Pigmente geeignet sind. Gegenstand dieser Erfindung sind neue Trisazoverbindungen, der allgemeinen Formel 1 : Devices can be used in the short-wave (blue) range. Surprisingly, the coupling of truxenequinone diazonium salts with known yellow couplers used in the preparation of photographic films has resulted in corresponding compounds which are useful as pigments. The present invention relates to novel trisazo compounds of general formula 1:
Formel 1 worin bedeuten Formula 1 wherein mean
R4l R5 und RB gleich oder verschieden Wasserstoff, Niederalkyl, Niederalkoxy oder Halogen und R 4l R5 and RB are identical or different hydrogen, lower alkyl, lower alkoxy or halogen and
R-i, R2 und R3 gleiche oder unterschiedliche Reste (Kuppler) der allgemeinen Formel 2 R-i, R2 and R3 are identical or different radicals (couplers) of the general formula 2
Formel 2 wobei Formula 2 where
RA einen Phenylrest darstellt, der ein- oder mehrfach durch Niederalkyl R A represents a phenyl radical which is mono- or polysubstituted by lower alkyl
Niederalkoxy, Phenoxy, Phenylcarbonyl und/oder Halogen substituiert sein kann, und Lower alkoxy, phenoxy, phenylcarbonyl and / or halogen may be substituted, and
R einen Niederalkylrest vorzugsweise Methyl, Ethyl oder Propyl, oder einen optional ein- oder mehrfach durch Niederalkyl, Niederalkoxy oder Halogen R is a lower alkyl radical, preferably methyl, ethyl or propyl, or an optionally mono- or polysubstituted by lower alkyl, lower alkoxy or halogen
substituierten Phenylrest darstellt. represents substituted phenyl radical.
Vorzugsweise sind R1 bis R3 gleich oder verschieden an sich bekannte Preferably, R 1 to R 3 are the same or different per se
Kuppierverbindungen der allgemeinen Formeln 2a und 2b: Coupling compounds of general formulas 2a and 2b:
Formel 2a Formel 2b wobei die Arylgruppe (Ra) mit R ein- oder mehrfach substituiert sein kann und die Substituenten für R - wie unter Ra definiert - sein können. Wherein the aryl group (R a ) may be mono- or polysubstituted with R and the substituents for R - may be as defined under R a -.
Besonders bevorzugt werden Verbindungen eingesetzt, in denen die Reste Ri bis R3 gleich sind. Bevorzugte Reste Ra sind Phenyl, gegebenenfalls ein- oder mehrfach substituiert mit Substituenten R, die ausgewählt sind aus Cl, F, Methoxy, Phenoxy und/oder Phenylcarbonyl. Particular preference is given to using compounds in which the radicals R 1 to R 3 are identical. Preferred radicals R a are phenyl, optionally mono- or polysubstituted with substituents R, which are selected from Cl, F, methoxy, phenoxy and / or phenylcarbonyl.
Bevorzugte Reste R sind Phenyl oder Methyl. Preferred radicals R are phenyl or methyl.
Die Reste R4, R5 und R6 sind bevorzugt Wasserstoff. The radicals R 4 , R 5 and R 6 are preferably hydrogen.
Unter Niederalkyl werden Alkylreste mit 1-8 Kohlenstoffatomen verstanden, wie Methyl, Ethyl, Propyl, Isopropyl, Butyl, sek.-Butyl, Pentyl usw.. By lower alkyl are meant alkyl radicals having 1-8 carbon atoms, such as methyl, ethyl, propyl, isopropyl, butyl, sec-butyl, pentyl, etc.
Unter Niederaikoxy werden Alkoxyreste mit 1-8 Kohlenstoffatomen verstanden, wie, Methoxy, Ethoxy, Propoxy, Butoxy usw.. By lower alkoxy are meant alkoxy radicals having 1-8 carbon atoms, such as, methoxy, ethoxy, propoxy, butoxy, etc.
Halogen ist ein Fluor-, Chlor-, Brom- oder Jodsubstituent. Halogen is a fluoro, chloro, bromo or iodo substituent.
Die neuen Tnsazoverbindungen können durch Kuppeln eines Diazoniumsalzes einer aromatischen Triaminoverbindung der allgemeinen Formel 3 The novel tnsazo compounds can be prepared by coupling a diazonium salt of an aromatic triamino compound of general formula 3
Formel 3 wobei R4 bis Rs wie unter Formel 1 genannt definiert ist, mit den Kupplern der Formel 2 hergestellt werden. Die Kupplung erfolgt unter solchen für die Kupplung allgemein bekannten Bedingungen, beispielsweise indem man bei Raumtemperatur in Formula 3 wherein R 4 to R s is defined as mentioned under formula 1, are prepared with the couplers of formula 2. The coupling is carried out under such well-known conditions for the coupling, for example by at room temperature in
Dimethylsulfoxid die Komponenten der Formeln 2 und 3 zusammen gibt und nach Zugabe von Natriumacetat-Trihydrat das Gemisch 4-5 Stunden unter Dimethyl sulfoxide, the components of the formulas 2 and 3 together and after addition of sodium acetate trihydrate, the mixture 4-5 hours under
Lichtausschluss rührt. Exclusion of light stirs.
Die Herstellung der Trisdiazoniumsalze der Formel 3 erfolgt durch die Diazotierung der entsprechenden Triamine ebenfalls unter an sich bekannten Bedingungen, z.B. durch Zugabe von Natriumnitrit zur salzsauren Lösung der jeweiligen The trisdiazonium salts of formula 3 are prepared by diazotization of the corresponding triamines also under conditions known per se, e.g. by adding sodium nitrite to the hydrochloric acid solution of the respective
Triaminoverbindung bei unter 5°C und Zugabe einer wässrigen Alkali- oder Triamino compound at below 5 ° C and addition of an aqueous alkali or
Ammoniumtetrafluoroboratlösung. Die Triaminoverbindungen werden in bekannter Weise aus den Trinitroverbindungen durch Reduktion hergestellt, z.B. unter Ammoniumtetrafluoroboratlösung. The triamino compounds are prepared in a known manner from the trinitro compounds by reduction, e.g. under
Verwendung von aktiviertem Raney-Nickel in Dimethylformamid bei 100°C bis 120°C und Zugabe von Hydrazinhydrat (98%ig). Die Trinitroverbindungen erhält man durch Nitrierung (Seka, R. et al. Monatsheft 74 (1943), S. 212) von durch Trimerisierung von Indan- 1 ,3-dion hergestellten Truxenchinonen (lonescu, . Chem. Ber. 60 Use of activated Raney nickel in dimethylformamide at 100 ° C to 120 ° C and addition of hydrazine hydrate (98%). The trinitro compounds are obtained by nitration (Seka, R. et al., Monatsheft 74 (1943), p. 212) of trichloroquinones prepared by trimerization of indane-1, 3-dione (Ionescu, Chem. Ber
(1927), S. 1232). (1927), p. 1232).
Die Kupplerverbindungen der allgemeinen Formel 2 sind kommerziell verfügbar, z.B. durch Acros Organics, Belgien. The coupler compounds of general formula 2 are commercially available, e.g. by Acros Organics, Belgium.
Die Herstellung der neuen Verbindungen ist einfach, so dass sehr wirksame und dauerhafte Systeme erhalten werden können, die, da kostengünstig, auch The preparation of the new compounds is simple, so that very effective and durable systems can be obtained, which, as low cost, too
ökonomisch sehr interessant sind. economically very interesting.
Erfindungsgemäß werden die oben genannten neuen Trisazofarbstoffe als Pigmente einzeln oder als Gemisch von mindestens zwei Komponenten in unterschiedlichen Mischungsverhältnissen, wobei jede Komponente Hauptkomponente sein kann, mit einem Gehalt bis zu 90%, in die Ladungserzeugungsschicht einer According to the invention, the abovementioned new trisazo dyes are used as pigments individually or as a mixture of at least two components in different mixing ratios, each component being able to be up to 90% in the charge generation layer
elektrofotografischen Vorrichtung eingebracht. Der Schichtverbund dieser introduced electrophotographic device. The layer composite of this
elektrofotografischen Vorrichtung umfasst mindestens eine mit leitfähiger Oberfläche versehene Unterlage, eine darüberliegende Ladungserzeugerschicht, welche die obengenannten Trisazoverbindungen, gegebenenfalls als Dispersion aufgebracht, enthält und eine darüberliegende Ladungstransportschicht, welche mindestens eine aufgebrachte herkömmliche Ladungstransportverbindung, wie z.B. N,N"-Bis-(3- methylpheny -N.N'-bis-ipheny^-benzidin enthält. Die neuen Trisazoverbindungen führen bei Verwendung als lichtempfindliche The electrophotographic device comprises at least one surface provided with a conductive surface, an overlying charge generator layer containing the abovementioned trisazo compounds, optionally applied as a dispersion, and an overlying charge transport layer comprising at least one applied conventional charge transport compound, such as N, N " -Bis (3 methylphenyl -N, N 'bis-containing ipheny ^ benzidine. The new trisazo compounds lead when used as photosensitive
Substanzen in einer Ladungsträgererzeugungsschicht einer elektrofotografischen Vorrichtung überraschend zu einer drastisch gesteigerten Lichtempfindlichkeit bei 400 bis 480 nm, bevorzugt bei 400 bis 450 nm, der Emission von blauen Substances in a carrier generation layer of an electrophotographic device surprisingly result in drastically increased photosensitivity at 400 to 480 nm, preferably at 400 to 450 nm, in the emission of blue
Laserdioden. Damit können sie z.B. in Laserdruckern Verwendung finden. Laser diodes. With this they can e.g. find use in laser printers.
Die Erfindung soll durch Beispiele näher erläutert werden: Ausführungsbeispiel The invention will be explained in more detail by examples: embodiment
Herstellung von Trisazofarbstoffen der allgemeinen Formel 1 , worin Ri bis R3 = K bedeuten gemäß Tabelle 1 und R bis R5 = H sind: Preparation of trisazo dyes of the general formula 1 in which R 1 to R 3 = K in accordance with Table 1 and R 5 to R 5 = H are:
Beispiel 1 example 1
Herstellung von 2,7, 2-Triamino-5H-tribenzo(a,f , k)trinden-5, 10,15-trion a) Trimerisierung von lndan-1 ,3-dion 3,5g lndan-1 ,3-dion bei Raumtemperatur 15 min in konzentrierter Schwefelsäure rühren, 3 h stehen lassen, anschließend in 500g Eiswasser geben, absaugen und bis pH= 7 mit Wasser waschen. Das so erhaltene 5H-Tribenzo(a,f,k)t nden-5, 0, 5-trion in Wasser suspendieren, kochen und heiß absaugen, anschließend in Eisessig kochen und heiß absaugen, in Chloroform rühren, absaugen, mit Acezon waschen und aus Nitrobenzol Umkristallisieren. b) Nitrierung von 5H-Tribenzo(a,f,k)trinden-5,10,15-trion Preparation of 2,7,2-triamino-5H-tribenzo (a, f, k) trinde-5,10,15-trione a) Trimerization of indane-1,3-dione 3,5g indane-1,3-dione Stir in concentrated sulfuric acid at room temperature for 15 min, allow to stand for 3 h, then pour into 500 g of ice-water, suction and wash until pH = 7 with water. Suspend the 5H-tribenzo (a, f, k) t-5, 0, 5-trione in water, boil and siphon off hot, then boil in glacial acetic acid and suction hot, stir in chloroform, suction, wash with acezone and Recrystallize from nitrobenzene. b) Nitration of 5H-tribenzo (a, f, k) triene-5,10,15-trione
0,01 Mol (3,84g) 5H-Tribenzo(a,f,k)trinden-5, 0,15-trion werden in 150ml 0.01 mole (3.84 g) of 5H-tribenzo (a, f, k) trinde-5, 0.15-trione are dissolved in 150 ml
konzentrierter Schwefelsäure bei -10°C- -5°C suspendiert und gerührt bis alles gelöst ist. Dann tropft man 0,036 Mol 100%ige Salpetersäure zu und rührt eine Stunde bei dieser Temperatur und anschließend 12 h bei Raumtemperatur. Danach gießt man das 2,7,12-Trinitro-5H~Tribenzo(a,f,k)trinden-5)10,15-trion auf 1000g Eis, saugt ab, wäscht mehrmals mit Wasser und zum Schluß mit Aceton. Nach dem trocknen kristallisiert man aus Nitrobenzol um. c) Reduktion von 2,7,12-Trinitro-5H-Tribenzo(a,f,k)trinden-5,10l15-trion concentrated sulfuric acid at -10 ° C -5 ° C and stirred until everything is solved. Then added dropwise 0.036 mol of 100% nitric acid and stirred for one hour at this temperature and then 12 h at room temperature. Thereafter, the 2,7,12-trinitro-5H-tribenzo (a, f, k) trindene-5 ) 10,15-trione is poured onto 1000 g of ice, filtered off with suction, washed several times with water and finally with acetone. After drying, crystallized from nitrobenzene. c) Reduction of 2,7,12-trinitro-5H-tribenzo (a, f, k) trindee-5,10 l 15-trione
Ein Gemisch von 0,01 Mol (5,19g) 2,7,12-Trinitro-5H-Thbenzo(a)f,k)trinden-5,10,15- trion und aktivierten Raney-Nickel wird in 1000ml DMF bei 100- 120°C suspendiert. Danach tropft man unter Rühren und Stickstoffeinieitung bei dieser Temperatur 25 ml Hydrazinhydrat (98%ig) langsam zu. Anschließend wird das Gemisch eine Stunde bei 130- 140°C gerührt, dann abgesaugt, das Filtrat bis auf 40 ml eingeengt und das 2,7,12Triamino-5H-Tribenzo(a,f,k)trinden-5,10,15-trion mit Ether ausgefällt. A mixture of 0.01 mol (5.19 g) of 2,7,12-trinitro-5H-thbenzo (a ) f, k) trindee-5,10,15-trione and activated Raney nickel is dissolved in 1000 ml of DMF at 100 - 120 ° C suspended. Then added dropwise with stirring and Stickstoffeinieitung at this temperature, 25 ml of hydrazine hydrate (98%) slowly. The mixture is then stirred for one hour at 130-140 ° C., then filtered off with suction, the filtrate is concentrated to 40 ml and the 2,7,12-triamino-5H-tribenzo (a, f, k) is trindene-5,10,15- precipitated with ether.
Beispiel 2 Example 2
Herstellung des 2,7, 2-Trisdiazonium-5H-tnbenzo(a,f,k)trinden-5, 0,15-trion- tristetrafluoro-borats Diazotierung Preparation of the 2,7,2-tris-diazonium-5H-tennobenzo (a, f, k) trindee-5,15-tri- intratra- trifluoroborate diazotization
0,01 Mo! (4,29g) 2)7,12-Triamino-5H-thbenzo(a,f,k)thnden-5,10,15-trion suspendiert man in einem Gemisch von 7o ml Wasser und 70 ml konzentrierter Salzsäure und lässt bei einer Temperatur unter 5°C eine Lösung von 0,075 Mol (5,17 g) 0.01 Mo! (4.29 g) 2 ) 7,12-triamino-5H-thbenzo (a, f, k) thnden-5,10,15-trione is suspended in a mixture of 70 ml of water and 70 ml of concentrated hydrochloric acid and allowed to stand at a Temperature below 5 ° C a solution of 0.075 mole (5.17 g)
Natriumnitrit in 30ml Wasser langsam zutropfen, wobei die Temperatur nicht überSodium nitrite in 30ml of water slowly dropwise, the temperature does not exceed
5°C steigen darf. Anschließend wird das Reaktionsgemisch bei dieser Temperatur 30 min gerührt und dann eine Lösung aus 0,045 Mol (4,71g) Ammoniumtetrafluoroborat in 30 ml Wasser zugetropft. Danach rührt man 30 min bei dieser Temperatur, saugt das 2,7,12-Thsdiazonium-5H-tribenzo(a,f,k)trinden-5,10,15-trion-tristetrafluoroborat ab, wäscht mit einem Gemisch Ethanol/ Ether (1 :1) und trocknet es. 5 ° C may rise. Subsequently, the reaction mixture is stirred at this temperature for 30 min and then added dropwise a solution of 0.045 mol (4.71 g) of ammonium tetrafluoroborate in 30 ml of water. The mixture is then stirred for 30 minutes at this temperature, sucks the 2,7,12-thsdiazonium-5H-tribenzo (a, f, k) trinden-5,10,15-trione tristetrafluoroborat, washed with a mixture of ethanol / ether ( 1: 1) and it dries.
Beispiel 3 Example 3
Herstellung der Zielverbindung 1 durch Kupplungsreaktion mit Verbindungen K 0,003 Mol (2,19g) des 2,7,12-Trisdiazonium-5H-tribenzo(a,f,k)trinden-5,10,15-trion- tristetrafluoroborats werden in 180 ml DMSO bei Raumtemperatur gelöst. Dazu tropft man eine Lösung aus 0,0091 Mol (2,7g) Kuppler K gemäß folgender Tabelle 1 und 80 ml DMSO schnell zu und gibt anschließend 0,0 8 mol (2,448 g ) Natriumacetat- trihydrat hinzu. Das Gemisch wird bei Raumtemperatur unter Lichtausschiuss fünf Stunden gerührt. Danach wird der Trisazofarbstoff (gemäß Tabelle 1) mit 11 Wasser ausgefällt, abgesaugt, dreimal mit siedendem Wasser sowie zweimal mit Methanoi gewaschen und getrocknet. Preparation of the Target Compound 1 by Coupling Reaction with Compounds K 0.003 mol (2.19 g) of the 2,7,12-trisdiazonium-5H-tribenzo (a, f, k) trindee-5,10,15-tri- intratrafluoroborate are dissolved in 180 ml DMSO dissolved at room temperature. A solution of 0.0091 mol (2.7 g) of coupler K in accordance with the following Table 1 and 80 ml of DMSO is added dropwise rapidly and then 0.08 mol (2.448 g) of sodium acetate are added dropwise. added trihydrate. The mixture is stirred at room temperature with Lichtausschiuss five hours. Thereafter, the trisazo dye (according to Table 1) is precipitated with 11 water, filtered off, washed three times with boiling water and twice with Methanoi and dried.
Zur Reinigung des Farbstoffes erhitzt man fünfmal mit 100 ml Methanol 30 min und saugt heiß ab. Anschließend wird fünfmal mit 100 ml eines Gemisches aus DMF und Methanol (1:1) 30 min gerührt und heiß abgesaugt. Zum Schluss wäscht man das Produkt nochmals mit Aceton und trocknet. Nachfolgende Trisazopigmente wurden gemäß Beispiel 3 synthetisiert, wobei die Kuppler K kommerziell verfügbar (Acros) waren und erfindungsgemäß eingesetzt wurden: To purify the dye, it is heated five times with 100 ml of methanol for 30 min and filtered off with suction. The mixture is then stirred five times with 100 ml of a mixture of DMF and methanol (1: 1) for 30 min and filtered off with suction while hot. Finally, the product is washed again with acetone and dried. Subsequent trisazo pigments were synthesized according to Example 3, wherein the couplers K were commercially available (acros) and were used according to the invention:
Tabelle 1 Table 1
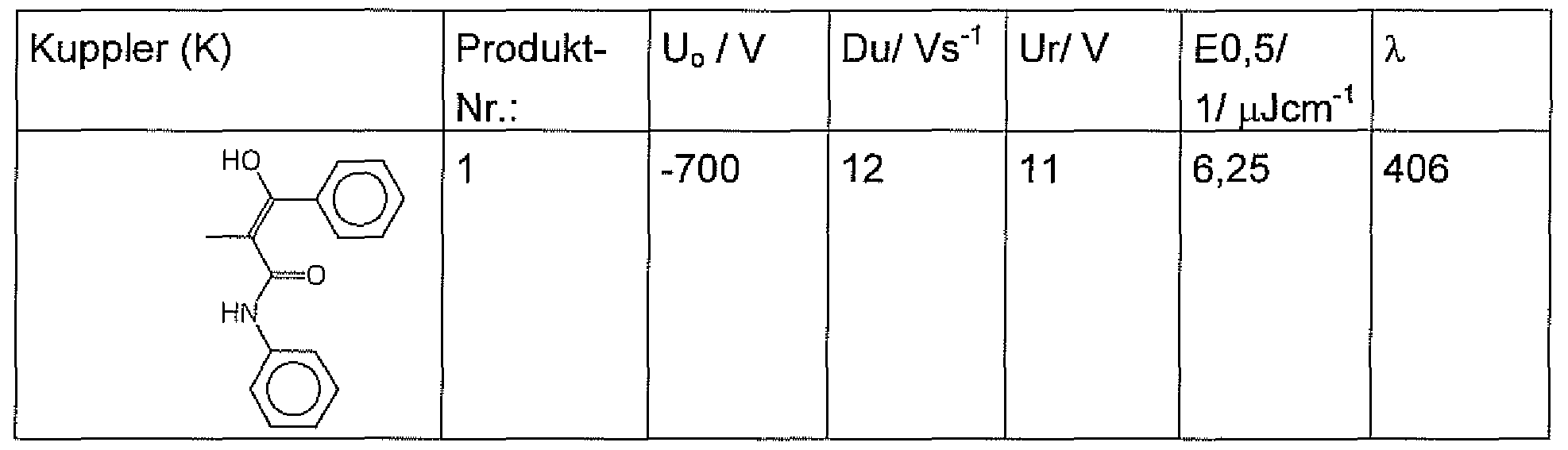
Produkt- Nr.: Struktur des Kupplers (K) Name des Trisazofarbstoffes Product No .: Structure of coupler (K) Name of trisazo dye
1 2,7,12-Tris-(2-hydroxy-2-phenyl-1 - phenylcarbamoyl-vinylazo))-5H-Tribenzo- (a,f ,k)trinden-5,10, 5-trion 1 2,7,12-tris (2-hydroxy-2-phenyl-1-phenylcarbamoyl-vinylazo)) - 5H-tribenzo (a, f, k) triene-5,10, 5-trione
2 2,7,12-Tris-(2-hydroxy-2-phenyl-1-(2- chloro-phenylcarbarnoyl-vinylazo))-5H- Tribenzo-(a,f,k)trinden-5, 10,15-trion 2,7,12-tris- (2-hydroxy-2-phenyl-1- (2-chloro-phenylcarbarnoyl-vinylazo)) - 5H-tribenzo (a, f, k) -trinde-5, 10, 15- trione
3 2,7,12-Tris-(2-hydroxy-2-phenyl-1 -(2,4- dichloro-phenylcarbamoyl-vinylazo))-5H- Thbenzo-(a,f,k)trinden-5, 10, 15-trion 2,7,12-Tris-{2- ydroxy-2-phenyl-1-(2- methoxy-phenylcarbamoyl-vinylazo))-5H- Tribenzo-(a,f,k)trinden-5, 10, 15-trion 3 2,7,12-tris- (2-hydroxy-2-phenyl-1 - (2,4-dichloro-phenylcarbamoyl-vinylazo)) - 5H-thbenzo- (a, f, k) -trinde-5, 10, 15-trione 2,7,12-Tris {2-ydroxy-2-phenyl-1- (2-methoxyphenylcarbamoyl-vinylazo)) - 5H-tribenzo (a, f, k) triene-5, 10, 15-trione
HN HN
CH30^^> CH 3 0 ^^>
2,7,12-Tris-(2-hydroxy-2-phenyi-1-(2-f!uoro- phenylcarba oyl-vinylazo))-5H-Tribenzo- (a,f,k)trinden-5, 10, 15-trion 2,7,12-tris- (2-hydroxy-2-phenyl-1- (2-fluoro-phenylcarbamoyl-vinylazo)) - 5H-tribenzo (a, f, k) -trinde-5, 10, 15-trione
2,7,12-Tris-(2-hydroxy~2-pheny[-1 -(4- phenoxy-phenylcarbamoyl-vinylazo))-5H- Tribenzo-(a,f,k)trinden-5, 10, 5-trion 2,7,12-tris- (2-hydroxy-2-phenyl-1 - (4-phenoxyphenylcarbamoyl-vinylazo)) - 5H-tribenzo (a, f, k) triene-5, 10, 5 trione
2,7, 2-Tris-(2-hydroxy-2-phenyl-1-(2- 2,7-tris (2-hydroxy-2-phenyl-1- (2-hydroxy)
HO v benzoyl-phenylcarbamoyl-vinylazo))-5H- Tribenzo-(a,ftk)trinden-5, 0, 15-trion HO v benzoyl-phenylcarbamoyl-vinylazo)) - 5H-tribenzo (a, f t k) -trene-5, 0, 15-trione
HN HN
2,7,12-Tris~(2-hydroxy-2-phenyl-1-(2- benzoyl-4-chloro-phenylcarbamoyl- vinylazo))-5H-Tribenzo-(a,f,k)trinden- 5,10,15-trion 2,7,12-tris - (2-hydroxy-2-phenyl-1- (2-benzoyl-4-chloro-phenylcarbamoyl-vinyl-azo)) - 5H-tribenzo (a, f, k) triene-5,10 , 15-trione
Beispiel 4 Example 4
Nachweis der elektrofotografischen Eigenschaften Die elektrofotografischen Eigenschaften der in Tabellel dargestellten Proof of the electrophotographic properties The electrophotographic properties of the tabular images
Trisazofarbstoffe 1 bis 8 wurden in einem Doppelschichtsystem bestimmt. Dazu wurde eine aluminisierte Polyesterunterlage mit einer Dispersion, die die jeweilige Trisazoverbindung als Pigmente in einer Polyvinylbutyrallösung in Methyienchlorid enthält, beschichtet. Die auf diese Weise nach dem Trocknen erhaltenen Trisazo dyes 1 to 8 were determined in a bilayer system. For this purpose, an aluminized polyester substrate was coated with a dispersion which contains the respective trisazo compound as pigments in a polyvinyl butyral solution in methylene chloride. The obtained in this way after drying
Ladungsträgererzeugungsschichten wurden mit einer Lösung von N,N"-Bis-(3- methylphenylJ-N.N'-bis-ipheny^-benzidin in Polycarbonat Z200 in Methylenchlorid beschichtet und nach Trocknung vermessen. Carrier generation layers were coated (with a solution of N, N "-bis- 3- methylphenylJ-NN '-bis-ipheny ^ benzidine in polycarbonate Z200 in methylene chloride and measured after drying.
Die Ergebnisse sind in Tabelle 2 dargestellt. Alle Verbindungen emittieren im blauen Bereich. The results are shown in Table 2. All connections emit in the blue range.
Tabelle 2 Table 2
Darin bedeuten: In this mean:
Du = Dunkelabfall You = dark waste
Uo = Aufladepotentiai Uo = Aufladepotentiai
E0,5= 1/ Lichtmenge, die notwendig ist, Uo auf 50% zu reduzieren Ur = Restpotential mit 6 μϋ/ cm2 bei Weißlicht E0,5 = 1 / amount of light necessary to reduce Uo to 50% Ur = residual potential at 6 μϋ / cm 2 for white light
λ =Messwellenlänge λ = measuring wavelength
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE201110004379 DE102011004379B4 (en) | 2011-02-18 | 2011-02-18 | Trisazo compounds, process for their preparation and their use |
| DE102011004379.9 | 2011-02-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012110402A1 true WO2012110402A1 (en) | 2012-08-23 |
Family
ID=45774158
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2012/052239 Ceased WO2012110402A1 (en) | 2011-02-18 | 2012-02-09 | Trisazo compounds, processes for preparing them and use thereof |
Country Status (2)
| Country | Link |
|---|---|
| DE (1) | DE102011004379B4 (en) |
| WO (1) | WO2012110402A1 (en) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5422211A (en) | 1993-04-30 | 1995-06-06 | Xerox Corporation | Imaging members with trisazo photogenerating materials |
| DE19654492A1 (en) | 1996-12-17 | 1998-06-18 | Syntec Ges Fuer Chemie Und Tec | Tris:azo compounds useful as photoconductors in charge generating layer |
| JP2001288239A (en) * | 2000-04-06 | 2001-10-16 | Inst Of Physical & Chemical Res | Polymer comprising triindole derivative and optical element |
| US20030049551A1 (en) | 2001-09-07 | 2003-03-13 | Xerox Corporation | Blue diode laser sensitive photoreceptor |
| EP1980593A2 (en) * | 2007-04-04 | 2008-10-15 | Xerox Corporation | Colourant compounds for phase change inks |
-
2011
- 2011-02-18 DE DE201110004379 patent/DE102011004379B4/en not_active Expired - Fee Related
-
2012
- 2012-02-09 WO PCT/EP2012/052239 patent/WO2012110402A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5422211A (en) | 1993-04-30 | 1995-06-06 | Xerox Corporation | Imaging members with trisazo photogenerating materials |
| DE19654492A1 (en) | 1996-12-17 | 1998-06-18 | Syntec Ges Fuer Chemie Und Tec | Tris:azo compounds useful as photoconductors in charge generating layer |
| JP2001288239A (en) * | 2000-04-06 | 2001-10-16 | Inst Of Physical & Chemical Res | Polymer comprising triindole derivative and optical element |
| US20030049551A1 (en) | 2001-09-07 | 2003-03-13 | Xerox Corporation | Blue diode laser sensitive photoreceptor |
| EP1980593A2 (en) * | 2007-04-04 | 2008-10-15 | Xerox Corporation | Colourant compounds for phase change inks |
Non-Patent Citations (2)
| Title |
|---|
| LONESCU, M., CHEM. BER., vol. 60, 1927, pages 1232 |
| SEKA, R. ET AL., MONATSHEFT, vol. 74, 1943, pages 212 |
Also Published As
| Publication number | Publication date |
|---|---|
| DE102011004379A1 (en) | 2012-08-23 |
| DE102011004379B4 (en) | 2014-08-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE2829543A1 (en) | ELECTROPHOTOGRAPHIC RECORDING MATERIAL | |
| DE2829606B2 (en) | Bisazo compounds, processes for their production and their use for the production of electrophotographic recording materials | |
| JPS58182640A (en) | Electrophotographic receptor of composite type | |
| DE3447685A1 (en) | DISAZO CONNECTIONS, METHOD FOR THEIR PRODUCTION AND AN ELECTROPHOTOGRAPHIC ELEMENT CONTAINING THESE CONNECTIONS | |
| DE3617948A1 (en) | ELECTROPHOTOGRAPHIC RECORDING MATERIAL | |
| DE3220772A1 (en) | LIGHT SENSITIVE ELEMENT FOR ELECTROPHOTOGRAPHIC PURPOSES | |
| DE69917510T2 (en) | Electrophotographic photosensitive member, apparatus unit and electrophotographic apparatus | |
| DE3602987A1 (en) | LIGHT SENSITIVE MATERIAL CONTAINING A DISAZO CONNECTION FOR USE IN ELECTROPHOTOGRAPHY | |
| DE2947760C2 (en) | ||
| JPH08110649A (en) | Electrophotographic photoreceptor | |
| DE3301453C2 (en) | ||
| DE10020779B4 (en) | Phthalocyanines, process for making the same and electrophotographic photoreceptor in which it is used | |
| DE102011004379B4 (en) | Trisazo compounds, process for their preparation and their use | |
| DE3521652C2 (en) | ||
| DE19808088A1 (en) | New tris(hydroxynaphthylazo) bridged triphenylamine pigments for electrophotographic charge generating layer | |
| DE60105801T2 (en) | naphthol | |
| DE10152122B4 (en) | Phthalocyanine composition, process for its preparation and electrophotographic photoreceptor | |
| DE3522896C2 (en) | ||
| DE69122303T2 (en) | Electrophotographic, light-sensitive element, electrophotographic apparatus and facsimile apparatus | |
| DE2352112A1 (en) | SENSITIZER FOR USE IN AN ELECTROPHOTOGRAPHIC LIGHT SENSITIVE MATERIAL | |
| DE4029565B4 (en) | Electrophotographic recording material and electrophotographic printing plate precursors containing a phthalocyanine pigment and a thiobarbituric acid derivative | |
| DE602004010216T2 (en) | Stilbene derivatives, processes for their preparation and electrophotographic photoconductors | |
| DE3804280A1 (en) | PHOTO SENSITIVE ELEMENT | |
| JPS59229564A (en) | Electrophotographic sensitive body | |
| DE4126622A1 (en) | ELECTROPHOTOGRAPHIC PHOTO RECEPTOR |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12706499 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12706499 Country of ref document: EP Kind code of ref document: A1 |