WO2012054861A1 - Glp-1 polymer conjugates having a releasable linkage - Google Patents
Glp-1 polymer conjugates having a releasable linkage Download PDFInfo
- Publication number
- WO2012054861A1 WO2012054861A1 PCT/US2011/057335 US2011057335W WO2012054861A1 WO 2012054861 A1 WO2012054861 A1 WO 2012054861A1 US 2011057335 W US2011057335 W US 2011057335W WO 2012054861 A1 WO2012054861 A1 WO 2012054861A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- glp
- water
- daltons
- polymer
- soluble polymer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*(C)*=*CCNC(CCCC(Nc(cc1)cc(C(COC(N*(C)C)=O)c2c3)c1-c2ccc3NC(CCCC(NCC*(C)*)=O)=O)=*)=*C Chemical compound C*(C)*=*CCNC(CCCC(Nc(cc1)cc(C(COC(N*(C)C)=O)c2c3)c1-c2ccc3NC(CCCC(NCC*(C)*)=O)=O)=*)=*C 0.000 description 4
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/605—Glucagons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
Definitions
- GLP-1 POLYMER CONJUGATES HAVING A RELEASABLE LINKAGE
- the present invention relates generally to polymer-active agent conjugates having a releasable linkage to thereby release the active agent in vivo.
- the invention relates to, among other things, methods for synthesizing the conjugates, methods for purifying the conjugates, and so on.
- Active agents that are polypeptides are often delivered via injection rather than orally. In this way, the polypeptide is introduced into the systemic circulation without exposure to the proteolytic environment of the stomach. Injection of polypeptides, however, has several drawbacks. For example, many polypeptides have a relatively short half-life, thereby necessitating repeated injections, which are often inconvenient and painful. Moreover, some polypeptides can elicit one or more immune responses with the consequence that the patient's immune system attempts to destroy or otherwise neutralize the immunogenic polypeptide. Of course, once the polypeptide has been destroyed or otherwise neutralized, the polypeptide cannot exert its intended pharmacodynamic activity. Thus, delivery of active agents such as polypeptides is often problematic even when these agents are administered by injection.
- PEGylated active agents such as PEGASYS ® PEGylated interferon alpha-2a (Hoffmann-La Roche, Nutley, NJ), PEG-INTRON ® PEGylated interferon alpha-2b (Schering Corp., Kennilworth, NJ), and NEULASTATM PEG-filgrastim (Amgen Inc., Thousand Oaks, CA) demonstrates that administration of a conjugated form of an active agent can have significant advantages over the unconjugated counterpart. Small molecules such as PEGASYS ® PEGylated interferon alpha-2a (Hoffmann-La Roche, Nutley, NJ), PEG-INTRON ® PEGylated interferon alpha-2b (Schering Corp., Kennilworth, NJ), and NEULASTATM PEG-filgrastim (Amgen Inc., Thousand Oaks, CA) demonstrates that administration of a conjugated form of an active agent can have significant advantages over the unconjugated counterpart. Small molecules such as
- conjugation of a polymer to an active agent to result in a commercially relevant drug is often challenging.
- conjugation can result in the polymer being attached at or near a site on the active agent that is necessary for pharmacologic activity (e.g., at or near a binding site).
- Such conjugates may therefore have unacceptably low activity due to, for example, the steric effects introduced by the polymer.
- Attempts to remedy conjugates having unacceptably low activity can be frustrated when the active agent has few or no other sites suited for attachment to a polymer.
- additional PEGylation alternatives have been desired.
- PEGylation wherein the native active agent (or a moiety having increased activity compared to the PEGylated active agent) is released.
- reversible PEGylation has been disclosed in the field of cancer chemotherapies. See Greenwald (1997) Exp. Opin. Ther. Patents 7(6):601-609.
- U.S. Patent Application Publication No. 2005/0079155 describes conjugates using reversible linkages. As described in this publication, reversible linkages can be effected through the use of an enzyme substrate moiety. It has been pointed out, however, that approaches relying on enzymatic activity are dependent on the availability of enzymes. See Peleg-Schulman (2004) J. Med. Chem. 47:4897-4904. Patient variability around the amount and activity of these enzymes can introduce inconsistent performance of the conjugate among different populations. Thus, additional approaches that do not rely on enzymatic processes for polymer release have been described as being desirable.
- GLP-1 can sometimes be administered to a patient to address or otherwise ameliorate this disorder. Due to the relatively short half-life of GLP-1, however, it would be advantageous to increase the in vivo half-life of these proteins by, for example, reversible PEGylation. Thus, the present invention seeks to solve this and other needs in the art.
- POLY 1 is a first water-soluble polymer
- POLY is a second water-soluble polymer
- X 1 is a first spacer moiety
- X is a second spacer moiety
- H a is an ionizable hydrogen atom
- R 1 is H or an organic radical
- R 2 is H or an organic radical
- R el when present, is a first electron altering group
- R e2 when present, is a second electron altering group
- Y 1 is O or S
- Y 2 is O or S
- GLP-1 is a residue of an amine-containing GLP-1 moiety.
- compositions comprising the conjugates are provided.
- a GLP-1 - water polymer conjugate is provided, the conjugate having an in vivo half-life increased by a factor of at least 1.5 as compared to the in vivo half-life of the GLP-1 moiety not conjugated to the water-soluble polymer.
- a GLP-1 -water soluble polymer conjugate is provided, the conjugate having an in vivo half-life increased by a factor of at least 2 as compared to the in vivo half-life of a GLP-1 moiety not conjugated to the water-soluble polymer.
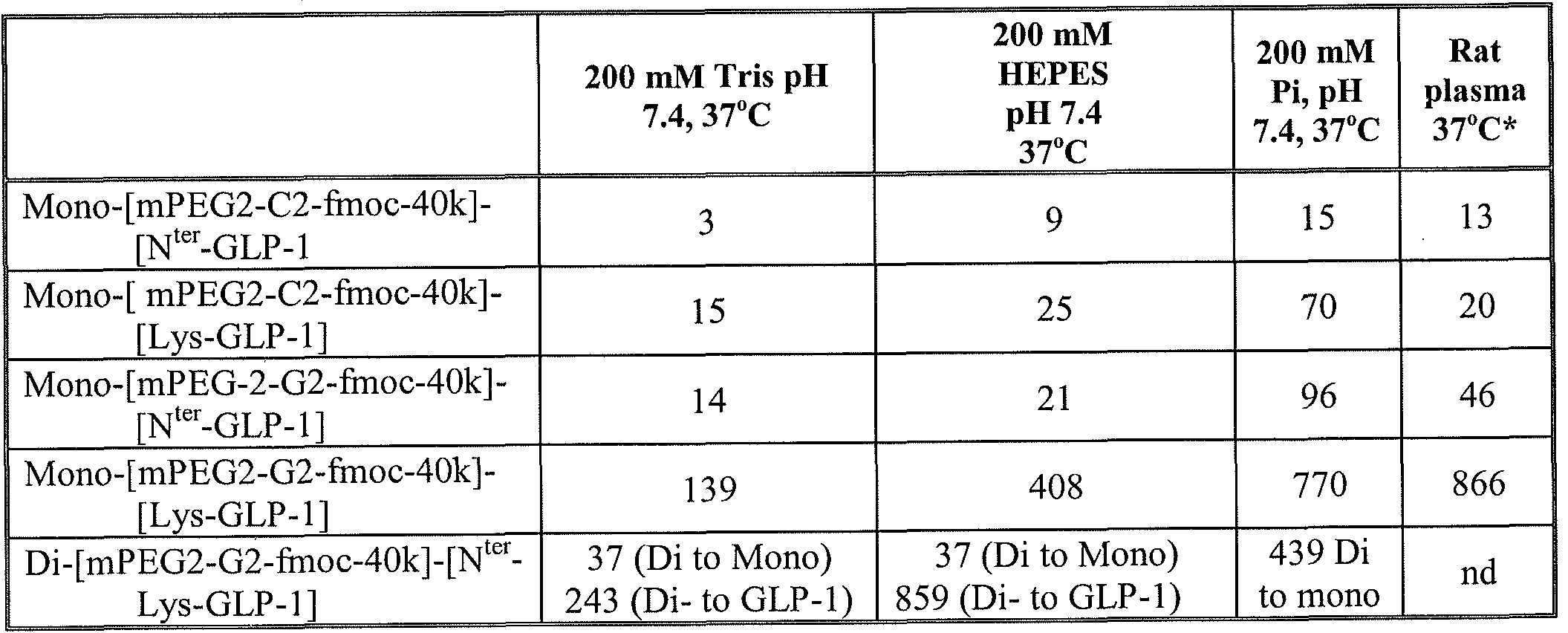
- FIG. 1 is a typical SP-HP cation exchange purification profile of
- FIG. 2 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-C2-fmoc-40K]-[N ter -GLP-l], as further described in Example 1.
- FIG. 3 is a typical SP-HP cation exchange purification profile of
- FIG. 4 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l], as further described in Example 2.
- FIG. 5 is a typical SP-HP cation exchange purification profile of
- FIG. 6 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-G2-fmoc-40K]-[N ter -GLP-l], as further described in Example 3.
- FIG. 7 is a typical SP-HP cation exchange purification profile of
- FIG. 8 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l], as further described in Example 4.
- FIG. 9 is a typical SP-HP cation exchange purification profile of di-[mPEG2-
- FIG. 10 is a reverse phase HPLC chromatogram of a purified composition of di-[mPEG2-G2-fmoc-40K]-[N ter -Lys-GLP-l], as further described in Example 5.
- FIG. 11A is a plot showing the decrease on mono-[mPEG2-G2-fmoc-40k]-
- FIG.llB reveals the stable level of GLP-1 (7-36) as a result of PEG linker release throughout the plasma incubation period. Both FIG. 11A and FIG. 11B are described in further detail in Example 6.
- FIG. 12 a plot of mice treated with native (i.e., unconjugated) GLP-1 peptide that lost in vivo effect after four hours of dosing, as further described in Example 8.
- FIG. 13A shows an eight-hour plot and FIG. 13B shows a 48-hour plot of the results of the results of saline, and several GLP-1 -containing conjugates, as further described in Example 8.
- FIG. 14A and FIG. 14B is a pair of plots showing GLP-1 conjugate's percent glucose lowering activity in db/db mice (relative to saline control at the 24 hour time point) and the in vitro ti /2 .
- GLP-1 conjugate's percent glucose lowering activity in db/db mice relative to saline control at the 24 hour time point
- in vitro ti /2 an inverse relationship appears between a GLP-1 conjugate's glucose lowering in mice and the in vitro ti /2 .
- FIG. 15A, FIG. 15B, FIG. 15C, and FIG. 15D are each plots showing the plasma concentration curves of tested conjugates following intraperitoneally (i.p.) of GLP-1 (7-36), PEG-GLP-1 , PEG, and GLP-1 (9-36), respectively.
- excipient includes a single excipient as well as two or more of the same or different excipients, and the like.
- GLP-1 moiety refers to those peptides
- GLP-1 moiety Prior to conjugation, the GLP-1 moiety has at least one electrophilic group or nucleophilic group suitable for reaction with a water soluble polymer.
- GLP-1 moiety encompasses both the GLP-1 moiety prior to conjugation as well as the GLP-1 moiety residue following conjugation. As will be explained in further detail below, one of ordinary skill in the art can determine whether any given moiety has GLP-1 activity.
- the terms "peptide,” “polypeptide,” and “protein,” refer to polymers comprised of amino acid monomers linked by amide bonds.
- Peptides may include the standard 20 a-amino acids that are used in protein synthesis by cells (i. e. natural amino acids), as well as non-natural amino acids (non-natural amino acids nay be found in nature, but not used in protein synthesis by cells, e.g., ornithine, citrulline, and sarcosine, or may be chemically synthesized), amino acid analogs, and peptidomimetics.
- the amino acids may be D- or L-optical isomers.
- Peptides may be formed by a condensation or coupling reaction between the a-carbon carboxyl group of one amino acid and the amino group of another amino acid.
- the terminal amino acid at one end of the chain (amino terminal) therefore has a free amino group, while the terminal amino acid at the other end of the chain (carboxy terminal) has a free carboxyl group.
- the peptides may be non-linear, branched peptides or cyclic peptides.
- the peptides may optionally be modified or protected with a variety of functional groups or protecting groups, including on the amino and/or carboxy terminus.
- Leucine is Leu or L; Isoleucine is He or I; Methionine is Met or M; Valine is Val or V; Serine is Ser or S; Proline is Pro or P; Threonine is Thr or T; Alanine is Ala or A;
- Tyrosine is Tyr or Y; Histidine is His or H; Glutamine is Gin or Q; Asparagine is Asn or N; Lysine is Lys or K; Aspartic Acid is Asp or D; Glutamic Acid is Glu or E; Cysteine is Cys or C; Tryptophan is Trp or W; Arginine is Arg or R; and Glycine is Gly or G.
- therapeutic peptide fragment refers to a peptide that comprises a truncation at the amino-terminus and/or a truncation at the carboxyl-terminus of a therapeutic peptide as defined herein.
- therapeutic peptide fragment or “fragments of therapeutic peptides” also encompasses amino-terminal and/or carboxyl-terminal truncations of therapeutic peptide variants and therapeutic peptide derivatives.
- Therapeutic peptide fragments may be produced by synthetic techniques known in the art or may arise from in vivo protease activity on longer peptide sequences. It will be understood that therapeutic peptide fragments retain some or all of the therapeutic activities of the therapeutic peptides.
- therapeutic peptide variants or “variants of therapeutic peptides” refer to therapeutic peptides having one or more amino acid
- substitutions including conservative substitutions and non-conservative substitutions, amino acid deletions (either internal deletions and/or C- and/or N- terminal truncations), amino acid additions (either internal additions and/or C- and/or N- terminal additions, e.g., fusion peptides), or any combination thereof.
- Variants may be naturally occurring (e.g. homologs or orthologs), or non-natural in origin.
- the term "therapeutic peptide variants" may also be used to refer to therapeutic peptides incorporating one or more non-natural amino acids, amino acid analogs, and peptidomimetics. It will be understood that, in accordance with the invention, therapeutic peptide fragments retain some or all of the therapeutic activities of the therapeutic peptides.
- therapeutic peptide derivatives or “derivatives of therapeutic peptides” as used herein refer to therapeutic peptides, therapeutic peptide fragments, and therapeutic peptide variants that have been chemically altered other than through covalent attachment of a water-soluble polymer. It will be understood that, in accordance with the invention, therapeutic peptide derivatives retain some or all of the therapeutic activities of the therapeutic peptides.
- amino terminus protecting group or “N-terminal protecting group,” “carboxy terminus protecting group” or “C-terminal protecting group;” or “side chain protecting group” refer to any chemical moiety capable of addition to and optionally removal from a functional group on a peptide (e.g., the TV-terminus, the
- PEG polyethylene glycol
- poly(ethylene glycol) poly(ethylene glycol)
- PEGs for use in accordance with the invention comprise the following structure "-0(CH 2 CH 2 0) m -" where (m) is 2 to 4000.
- PEG also includes "-CH 2 CH 2 -0(CH 2 CH 2 0) m -CH 2 CH 2 -" and "-(CH 2 CH 2 0) m -,” depending upon whether or not the terminal oxygens have been displaced.
- the atoms comprising the spacer moiety when covalently attached to a water-soluble polymer segment, do not result in the formation of an oxygen-oxygen bond (i.e., an "-0-0-" or peroxide linkage).
- PEG includes structures having various tenninal or "end capping” groups and so forth.
- PEG also means a polymer that contains a majority, that is to say, greater than 50%, of -CH 2 CH 2 0- monomelic subunits.
- the PEG can take any number of a variety of molecular weights, as well as structures or geometries such as “branched,” “linear,” “forked,” “multifunctional,” and the like, to be described in greater detail below.
- end-capped or “terminally capped” are interchangeably used herein to refer to a terminal or endpoint of a polymer having an end-capping moiety.
- the end-capping moiety comprises a hydroxy or Ci -20 alkoxy group.
- examples of end-capping moieties include alkoxy (e.g., methoxy, ethoxy and benzyloxy), as well as aryl, heteroaryl, cyclo, heterocyclo, and the like.
- saturated, unsaturated, substituted and unsubstituted forms of each of the foregoing are envisioned.
- the end-capping group can also be a silane.
- the end-capping group can also advantageously comprise a detectable label.
- the amount or location of the polymer and/or the moiety (e.g., active agent) of interest to which the polymer is coupled can be determined by using a suitable detector.
- suitable detector include, without limitation, fluorescers,
- chemiluminescers include photometers, films,
- Non-naturally occurring with respect to a polymer or water-soluble polymer means a polymer that in its entirety is not found in nature.
- a non-naturally occurring polymer or water-soluble polymer may, however, contain one or more subunits or portions of a subunit that are naturally occurring, so long as the overall polymer structure is not found in nature.
- water-soluble polymer is any polymer that is soluble in water at room temperature. Typically, a water-soluble polymer will transmit at least about 75%, more preferably at least about 95% of light, transmitted by the same solution after filtering. On a weight basis, a water-soluble polymer will preferably be at least about 35% (by weight) soluble in water, more preferably at least about 50% (by weight) soluble in water, still more preferably about 70% (by weight) soluble in water, and still more preferably about 85% (by weight) soluble in water. It is still more preferred, however, that the water-soluble polymer is about 95% (by weight) soluble in water and most preferred that the water-soluble polymer is completely soluble in water.
- Molecular weight in the context of a water-soluble polymer of the invention can be expressed as either a number average molecular weight or a weight average molecular weight. Unless otherwise indicated, all references to molecular weight herein refer to the weight average molecular weight. Both molecular weight determinations, number average and weight average, can be measured using gel permeation chromatography or other liquid chromatography techniques.
- the polymers of the invention are typically polydisperse (i.e., number average molecular weight and weight average molecular weight of the polymers are not equal), possessing low polydispersity values of preferably less than about 1.2, more preferably less than about 1.15, still more preferably less than about 1.10, yet still more preferably less than about 1.05, and most preferably less than about 1.03.
- carboxylic acid is a moiety having a -C-OH functional group [also represented as a "-COOH” or -C(0)OH], as well as moieties that are derivatives of a carboxylic acid, such derivatives including, for example, protected carboxylic acids.
- carboxylic acid includes not only the acid form, but corresponding esters and protected forms as well.
- protecting groups suited for a carboxylic acid and any other functional group described herein reference is made to Greene et al, "PROTECTIVE GROUPS IN ORGANIC SYNTHESIS” 3 rd Edition, John Wiley and Sons, Inc., New York, 1999.
- reactive and activated when used in conjunction with a particular functional group, refer to a reactive functional group that reacts readily with an electrophile or a nucleophile on another molecule. This is in contrast to those groups that require strong catalysts or highly impractical reaction conditions in order to react (i.e., a "nonreactive” or “inert” group).
- protected refers to the presence of a moiety (i.e., the protecting group) that prevents or blocks reaction of a particular chemically reactive functional group in a molecule under certain reaction conditions.
- the protecting group will vary depending upon the type of chemically reactive functional group being protected as well as the reaction conditions to be employed and the presence of additional reactive or protecting groups in the molecule, if any.
- Protecting groups known in the art can be found in Greene et ah, supra.
- spacer or "spacer moiety” are used herein to refer to an atom or a collection of atoms optionally appearing between one moiety and another.
- the spacer moieties may be hydrolytically stable or may include one or more physiologically hydrolyzable or enzymatically releasable linkages.
- An "organic radical" as used herein includes, for example, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, aryl and substituted aryl.
- Alkyl refers to a hydrocarbon chain, typically ranging from about 1 to 20 atoms in length. Such hydrocarbon chains are preferably but not necessarily saturated and may be branched or straight chain, although typically straight chain is preferred. Exemplary alkyl groups include methyl, ethyl, propyl, butyl, pentyl, 1-methylbutyl, 1-ethylpropyl, 3- methylpentyl, and the like. As used herein, "alkyl” includes cycloalkyl when three or more carbon atoms are referenced and lower alkyl.
- “Lower alkyl” refers to an alkyl group containing from 1 to 6 carbon atoms, and may be straight chain or branched, as exemplified by methyl, ethyl, n-butyl, z ' so-butyl, and tert-butyl.
- Cycloalkyl refers to a saturated or unsaturated cyclic hydrocarbon chain, including bridged, fused, or spiro cyclic compounds, preferably made up of 3 to about 12 carbon atoms, more preferably 3 to about 8 carbon atoms.
- Non-interfering substituents are those groups that, when present in a molecule, are typically non-reactive with other functional groups contained within the molecule.
- substituted refers to a moiety (e.g., an alkyl group) substituted with one or more non-interfering substituents, such as, but not limited to: C 3 -C 8 cycloalkyl, e.g., cyclopropyl, cyclobutyl, and the like; halo, e.g., fluoro, chloro, bromo, and iodo; cyano; alkoxy, lower phenyl; substituted phenyl; and the like, for one or more hydrogen atoms.
- substituents such as, but not limited to: C 3 -C 8 cycloalkyl, e.g., cyclopropyl, cyclobutyl, and the like
- halo e.g., fluoro, chloro, bromo, and iodo
- cyano alkoxy, lower phenyl; substituted phenyl; and the like, for one or more hydrogen atoms
- Substituted aryl is aryl having one or more non-interfering groups as a substituent. For substitutions on a phenyl ring, the substituents may be in any orientation (i.e., ortho, meta, or para).
- “Substituted ammonium” is ammonium having one or more non-interfering groups (e.g., an organic radical) as a substituent.
- Alkoxy refers to an -O-R group, wherein R is alkyl or substituted alkyl, preferably C1-C20 alkyl (e.g., methoxy, ethoxy, propyloxy, benzyl, etc.), more preferably Ci- C 7 alkyl.
- alkenyl refers to a branched or unbranched hydrocarbon group of 2 to 15 atoms in length, containing at least one double bond.
- alkenyl include (without limitation) ethenyl, «-propenyl, isopropenyl, n-butenyl, z ' so-butenyl, octenyl, decenyl, tetradecenyl, and the like.
- alkynyl refers to a branched or unbranched hydrocarbon group of 2 to 15 atoms in length, containing at least one triple bond.
- exemplary alkynyl include (without limitation) ethynyl, n-butynyl, z ' so-pentynyl, octynyl, decynyl, and so forth.
- Aryl means one or more aromatic rings, each of 5 or 6 core carbon atoms.
- Aryl includes multiple aryl rings that may be fused, as in naphthyl, or unfused, as in biphcnyl. Aryl rings may also be fused or unfused with one or more cyclic hydrocarbon, heteroaryl, or heterocyclic rings. As used herein, "aryl” includes heteroaryl.
- An aromatic-containing moiety e.g., Ar 1 , Ar 2 , and so forth, means a structure containing aryl.
- Heteroaryl is an aryl group containing from one to four heteroatoms, preferably N, O, or S, or a combination thereof. Heteroaryl rings may also be fused with one or more cyclic hydrocarbon, heterocyclic, aryl, or heteroaryl rings.
- Heterocycle or “heterocyclic” means one or more rings of 5-12 atoms, preferably 5-7 atoms, with or without unsaturation or aromatic character and having at least one ring atom which is not a carbon.
- Preferred heteroatoms include sulfur, oxygen, and nitrogen.
- Substituted heteroaryl is heteroaryl having one or more non-interfering , groups as substituents.
- Substituted heterocycle is a heterocycle having one or more side chains formed from non-interfering substituents.
- Electrophile refers to an ion or atom or collection of atoms, which may be ionic, having an electrophilic center, i.e., a center that is electron seeking, capable of reacting with a nucleophile.
- Nucleophile refers to an ion or atom or collection of atoms that may be ionic having a nucleophilic center, i.e., a center that is seeking an electrophilic center or with an electrophile.
- a "physiologically cleavable” as well as a “hydrolyzable” bond is a relatively weak bond that reacts with water (i.e., is hydrolyzed) under physiological conditions.
- the tendency of a bond to hydrolyze in water will depend not only on the general type of linkage connecting two central atoms but also on the substituents attached to these central atoms.
- Exemplary hydrolyzable bonds include, but are not limited to, carboxylate ester, phosphate ester, anhydride, acetal, ketal, acyloxyalkyl ether, imine, and ortho esters.
- a “releasable linkage” includes, but is not limited to, a physiologically cleavable bond, a hydrolyzable bond, and an enzymatically degradable linkage.
- a “releasable linkage” is a linkage that may undergo either hydrolysis or cleavage by some other mechanism (e.g., enzyme-catalyzed, acid-catalyzed, base-catalyzed, and so forth) under physiological conditions.
- a “releaseable linkage” can involve an elimination reaction that has a base abstraction of a proton, (e.g., an ionizable hydrogen atom, H a ), as the driving force.
- a “releaseable linkage” is synonymous with a “degradable linkage.”
- An "enzymatically releasable linkage” means a linkage that is subject to degradation by one or more enzymes.
- a “hydrolytically stable” linkage or bond refers to a chemical bond, typically a covalent bond, which is substantially stable in water, that is to say, does not undergo hydrolysis under physiological conditions to any appreciable extent over an extended period of time.
- hydrolytically stable linkages include but are not limited to the following: carbon-carbon bonds (e.g., in aliphatic chains), ethers, amides, and the like.
- a hydrolytically stable linkage is one that exhibits a rate of hydrolysis of less than about 1 -2% per day under physiological conditions. Hydrolysis rates of representative chemical bonds can be found in most standard chemistry textbooks. It must be pointed out that some linkages can be hydrolytically stable or hydrolyzable, depending upon (for example) adjacent and neighboring atoms and ambient conditions.
- One of ordinary skill in the art can determine whether a given linkage or bond is hydrolytically stable or hydrolyzable in a given context by, for example, placing a linkage-containing molecule of interest under conditions of interest and testing for evidence of hydrolysis (e.g., the presence and amount of two molecules resulting from the cleavage of a single molecule). Other approaches known to those of ordinary skill in the art for determining whether a given linkage or bond is hydrolytically stable or hydrolyzable can also be used.
- active agent biologically active agent
- pharmacologically active agent are used interchangeably herein and are defined to include any agent, drug, compound, composition of matter or mixture that provides some pharmacologic, often beneficial, effect that can be demonstrated in vivo or in vitro. This includes food
- these terms further include any physiologically or pharmacologically active substance that produces a localized or systemic effect in a patient.
- “Pharmaceutically acceptable excipient” or “pharmaceutically acceptable carrier” refers to an excipient that can be included in the compositions of the invention and that causes no significant adverse toxicological effects to the patient.
- “Pharmacologically effective amount,” “physiologically effective amount,” and “therapeutically effective amount” are used interchangeably herein to mean the amount of a polymer-active agent conjugate ⁇ typically present in a pharmaceutical preparation— that is needed to provide a desired level of active agent and/or conjugate in the bloodstream or in a target tissue. The exact amount will depend upon numerous factors, e.g., the particular active agent, the components and physical characteristics of the pharmaceutical preparation, intended patient population, patient considerations, and the like, and can readily be determined by one of ordinary skill in the art, based upon the information provided herein and available in the relevant literature.
- Multifunctional in the context of a polymer means a polymer having 3 or more functional groups contained therein, where the functional groups may be the same or different. Multifunctional polymers will typically contain from about 3-100 functional groups, or from 3- 50 functional groups, or from 3-25 functional groups, or from 3-15 functional groups, or from 3 to 10 functional groups, or will contain 3, 4, 5, 6, 7, 8, 9 or 10 functional groups within the polymer.
- a "difunctional” polymer means a polymer having two functional groups contained therein, either the same (i.e., homodifunctional) or different (i.e., heterodifunctional).
- Branched in reference to the geometry or overall structure of a polymer, refers to polymer having 2 or more polymer “arms.”
- a branched polymer may possess 2 polymer arms, 3 polymer arms, 4 polymer arms, 6 polymer arms, 8 polymer arms or more.
- One particular type of highly branched polymer is a dendritic polymer or dendrimer, which, for the purposes of the invention, is considered to possess a structure distinct from that of a branched polymer.
- a "dendrimer” or dendritic polymer is a globular, size monodisperse polymer in which all bonds emerge radially from a central focal point or core with a regular branching pattern and with repeat units that each contribute a branch point. Dendrimers exhibit certain dendritic state properties such as core encapsulation, making them unique from other types of polymers.
- a basic or acidic reactant described herein includes neutral, charged, and any corresponding salt forms thereof.
- the term "patient,” refers to a living organism suffering from or prone to a condition that can be prevented or treated by administration of a conjugate as provided herein, and includes both humans and animals.
- drug release rate means a rate (stated as a half-life) in which half of the total amount of polymer- active agent conjugates in a system will cleave into the active agent and a polymeric residue.
- the "halo" designator e.g., fluoro, chloro, iodo, bromo, and so forth
- the suffix "ide” e.g., fluoride, chloride, iodide, bromide, and so forth
- the halogen exists in its independent ionic form (e.g., such as when a leaving group leaves a molecule).
- the present invention comprises (among other things) conjugates having a releasable linkage.
- a water-soluble polymer for use herein can be, for example, other water-soluble polymers such as other poly(alkylene glycols) [also referred to as "poly(alkyleneoxides)"], such as poly(propylene glycol) ("PPG"), copolymers of ethylene glycol and propylene glycol and the like, poly(olefinic alcohol), poly(vinylpyrrolidone), poly(hydroxyalkylmethacrylamide), poly(hydroxyalkylmefhacrylate), poly(saccharides), poly(a-hydroxy acid), poly( vinyl alcohol), polyphosphazene, polyoxazoline, poly(N-acryloylmorpholine), such as described in U.S.
- the water soluble polymer can be a homopolymer, copolymer, terpolymer, nonrandom block polymer, and random block polymer of any of the foregoing.
- a water-soluble polymer can be linear, but can also be in other forms (e.g., branched, forked, and the like) as will be described in further detail below.
- a water-soluble polymer has from 1 to about 300 termini.
- each water-soluble polymer in the overall structure can be the same or different. It is preferred, however, that all water-soluble polymers in the overall structure are of the same type. For example, it is preferred that all water-soluble polymers within a given structure are poly(ethylene glycol) polymers.
- the weight average molecular weight of any given water-soluble polymer will typically be in the following range: 100 Daltons to about 150,000 Daltons. Exemplary ranges, however, include weight-average molecular weights in the following ranges: in the range of from about 880 Daltons to about 5,000 Daltons; in the range of greater than 5,000 Daltons to about 100,000 Daltons; in the range of from about 6,000 Daltons to about 90,000 Daltons; in the range of from about 10,000 Daltons to about 85,000 Daltons; in the range of greater than 10,000 Daltons to about 85,000 Daltons; in the range of from about 20,000 Daltons to about 85,000 Daltons; in the range of from about 53,000 Daltons to about 85,000 Daltons; in the range of from about 25,000 Daltons to about 120,000 Daltons; in the range of from about 29,000 Daltons to about 120,000 Daltons; in the range of from about 35,000 Daltons to about 120,000 Daltons; in the range of about 880 Daltons to about 60,000 Daltons; in the range of about 440 Dal
- Exemplary weight-average molecular weights for the water-soluble polymer include about 100 Daltons, about 200 Daltons, about 300 Daltons, about 400 Daltons, about 440 Daltons, about 500 Daltons, about 600 Daltons, about 700 Daltons, about 750 Daltons, about 800 Daltons, about 900 Daltons, about 1 ,000 Daltons, about 1,500 Daltons, about 2,000 Daltons, about 2,200 Daltons, about 2,500 Daltons, about 3,000 Daltons, about 4,000 Daltons, about 4,400 Daltons, about 4,500 Daltons, about 5,000 Daltons, about 5,500 Daltons, about 6,000 Daltons, about 7,000 Daltons, about 7,500 Daltons, about 8,000 Daltons, about 9,000 Daltons, about 10,000 Daltons, about 11 ,000 Daltons, about 12,000 Daltons, about 13,000 Daltons, about 14,000 Daltons, about 15,000 Daltons, about 16,000 Daltons, about 17,000 Daltons, about 18,000 Daltons, about 19,000 Daltons, about 20,000 Daltons, about 22,500 Daltons, about 25,000 Daltons, about
- Branched versions of the water-soluble polymer e.g., a branched 40,000 Dalton water-soluble polymer comprised of two 20,000 Dalton polymers
- Branched versions of the water-soluble polymer having a total weight average molecular weight of any of the foregoing can also be used.
- the polymeric reagent used to prepare the conjugate will comprise at least one water-soluble polymer having a total size in the range suited for the desired rate of release of the conjugate formed therefrom.
- a conjugate having a relatively long release rate can be prepared from a polymeric reagent having a size suited for (a) extended circulation prior to release of the active agent from the conjugate, and (b) moderately rapid in vivo clearance of the species liberated from the conjugate upon release from the conjugate.
- the polymeric reagent would typically have a lower molecular weight.
- the PEG When a PEG is used as the water-soluble polymer(s) in the polymeric reagent, the PEG typically comprises a number of (OCH 2 CH 2 ) monomers [or (CH 2 CH 2 0) monomers, depending on how the PEG is defined].
- the number of repeating units is identified by the subscript "n" in "(OCH 2 CH 2 ) n .”
- the value of (n) typically falls within one or more of the following ranges: from 2 to about 3400, from about 4 to about 1500, from about 100 to about 2300, from about 100 to about 2270, from about 136 to about 2050, from about 225 to about 1930, from about 450 to about 1930, from about 1200 to about 1930, from about 568 to about 2727, from about 660 to about 2730, from about 795 to about 2730, from about 795 to about 2730, from about 909 to about 2730, and from about 1 ,200 to about 1 ,900.
- n the number of repeating units
- Each water-soluble polymer is typically biocompatible and non-immunogenic.
- a substance is considered biocompatible if the beneficial effects associated with use of the substance alone or with another substance (e.g., an active agent) in connection with living tissues (e.g., administration to a patient) outweighs any deleterious effects as evaluated by a clinician, e.g., a physician.
- another substance e.g., an active agent
- living tissues e.g., administration to a patient
- non-immunogenicity a substance is considered non-immunogenic if use of the substance alone or with another substance in connection with living tissues does not produce an immune response (e.g., the formation of antibodies) or, if an immune response is produced, that such a response is not deemed clinically significant or important as evaluated by a clinician. It is particularly preferred that the water-soluble polymers described herein as well as conjugates of active agents and the polymers are biocompatible and non-immunogenic.
- free or nonbound PEG is a linear polymer terminated at each end with hydroxyl groups:
- (m 1 ) typically ranges from zero to about 4,000, preferably from about 20 to about 1 ,000.
- Another type of free or nonbound PEG useful in the present invention is methoxy-PEG-OH, or mPEG in brief, in which one terminus is the relatively inert methoxy group, while the other terminus is a hydroxyl group.
- the structure of mPEG is given below.
- Multi-armed or branched PEG molecules such as those described in U.S.
- Patent No. 5,932,462 can also be used as the PEG polymer.
- PEG can have the structure:
- poly a and poly b are PEG backbones (either the same or different), such as methoxy poly(ethylene glycol);
- R" is a nonreactive moiety, such as H, methyl or a PEG backbone
- the branched PEG polymer is methoxy poly( ethylene glycol) disubstituted lysine.
- the PEG can comprise a forked PEG.
- An example of a free or nonbound forked PEG is represented by the following formula:
- X is a spacer moiety and each Z is an activated terminal group linked to CH by a chain of atoms of defined length.
- the chain of atoms linking the Z functional groups to the branching carbon atom serve as a tethering group and may comprise, for example, alkyl chains, ether chains, ester chains, amide chains and combinations thereof.
- U.S. Patent No. 6,362,254 discloses various forked PEG structures capable of use in the present invention.
- the PEG polymer may comprise a pendant PEG molecule having reactive groups, such as carboxyl, covalently attached along the length of the PEG rather than at the end of the PEG chain.
- the pendant reactive groups can be attached to the PEG directly or through a spacer moiety, such as an alkylene group.
- each water-soluble polymer in the polymeric reagent can also be prepared with one or more weak or releasable linkages in the polymer, including any of the above described polymers.
- PEG can be prepared with ester linkages in the polymer that are subject to hydrolysis. As shown below, this hydrolysis results in cleavage of the polymer into fragments of lower molecular weight:
- hydrolytically releasable linkages useful as a releasable linkage within a polymer backbone, include carbonate linkages; imine linkages resulting, for example, from reaction of an amine and an aldehyde (see, e.g., Ouchi et al.
- phosphate ester linkages formed, for example, by reacting an alcohol with a phosphate group; hydrazone linkages which are typically formed by reaction of a hydrazide and an aldehyde; acetal linkages that are typically formed by reaction between an aldehyde and an alcohol; ortho ester linkages that are, for example, formed by reaction between a formate and an alcohol; amide linkages formed by an amine group, e.g., at an end of a polymer such as PEG, and a carboxyl group of another PEG chain; urethane linkages formed from reaction of, e.g., a PEG with a terminal isocyanate group and a PEG alcohol; peptide linkages formed by an amine group, e.g., at an end of a polymer such as PEG, and a carboxyl group of a peptide; and oligonucleotide linkages formed by, for example
- poly( ethylene glycol) or PEG represents or includes all the above forms of PEG.
- water-soluble polymer refers both to a molecule as well as the residue of water-soluble polymer that has been attached to another moiety.
- the following description of a water-soluble polymer are applicable not only to the polymeric reagent, but to the corresponding conjugates formed using the described polymeric reagents.
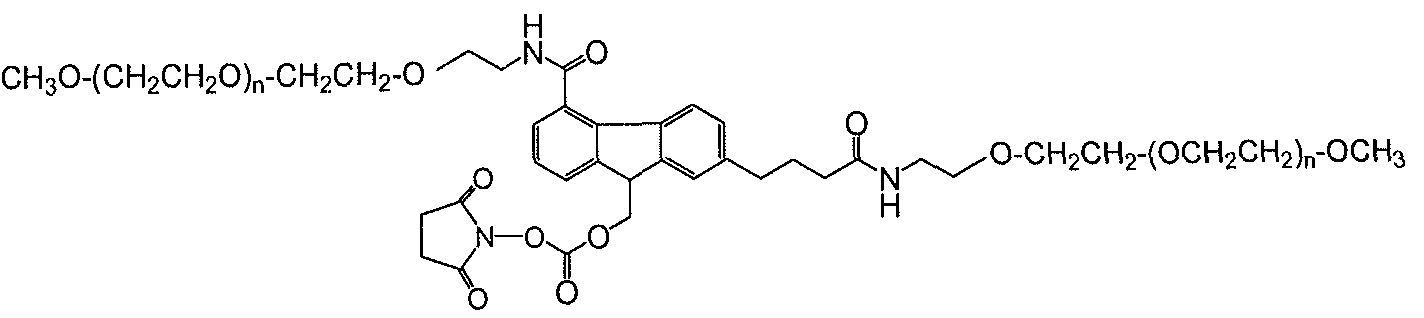
- the functional group of the polymeric reagents used to form the conjugates described herein is a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage.
- the invention is not limited with respect to the specific functional group so long as the functional group is capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage.
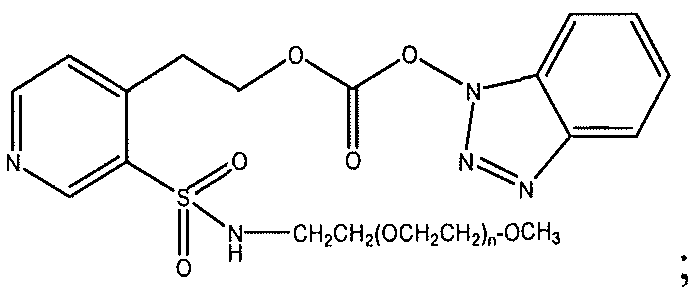
- Exemplary functional groups capable of reacting with an amino group of an active agent include those functional groups selected from the group consisting of active carbonates such as N-succinimidyl, 1-benzotriazolyl, imidazole, carbonate halides (such as carbonate chloride and carbonate bromide), phenolates (such as jo-nitrophenolate) and so forth. Also, as a special case, if the active agent is available with the active amine group converted into an isocyanate or isothiocyanate group, then the functional group of the polymeric reagent can be hydroxyl as the reaction of these components provides a releasable carbamate linkage.
- active carbonates such as N-succinimidyl, 1-benzotriazolyl, imidazole, carbonate halides (such as carbonate chloride and carbonate bromide), phenolates (such as jo-nitrophenolate) and so forth.
- the functional group of the polymeric reagent can be hydroxyl as the reaction of these components
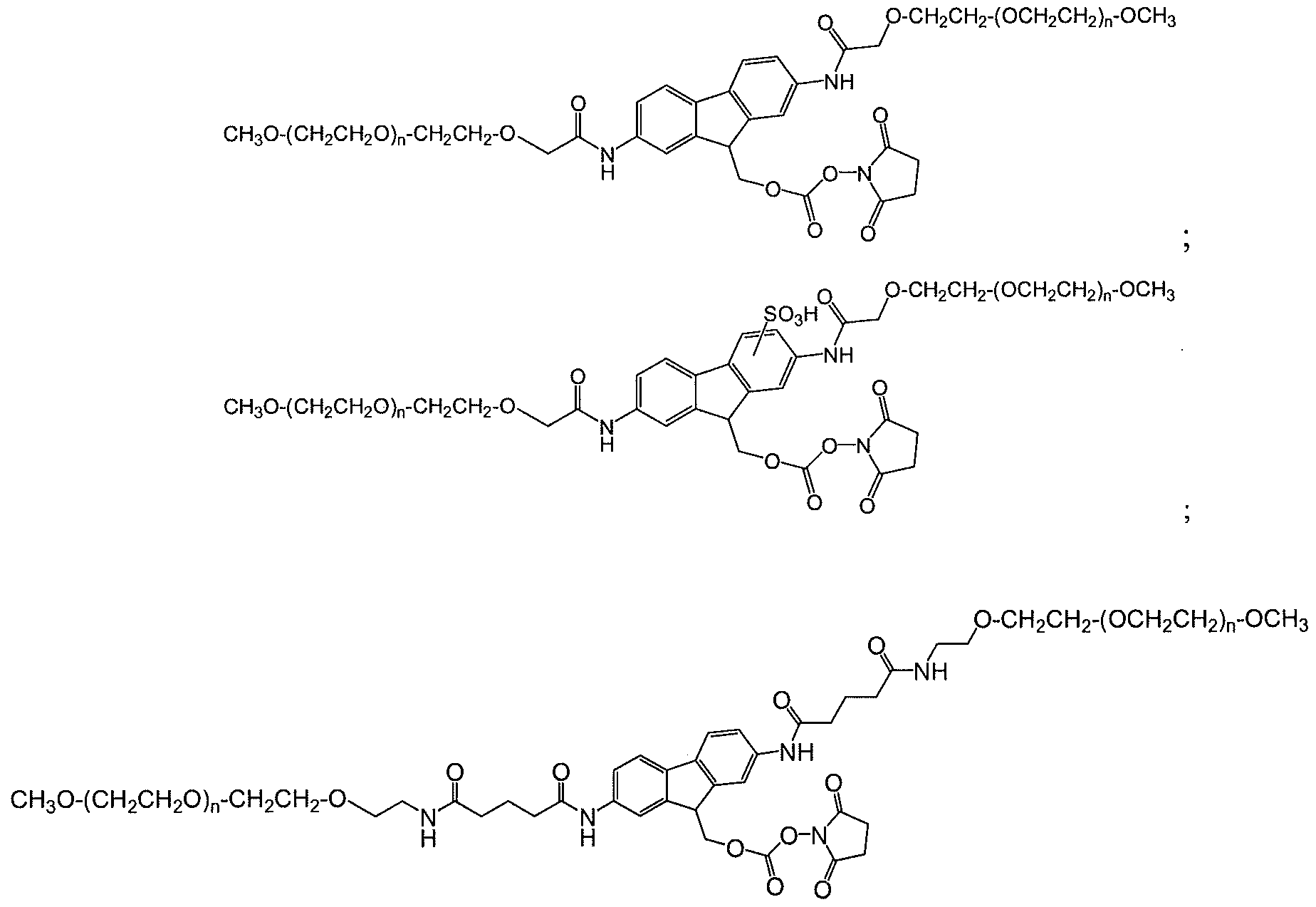
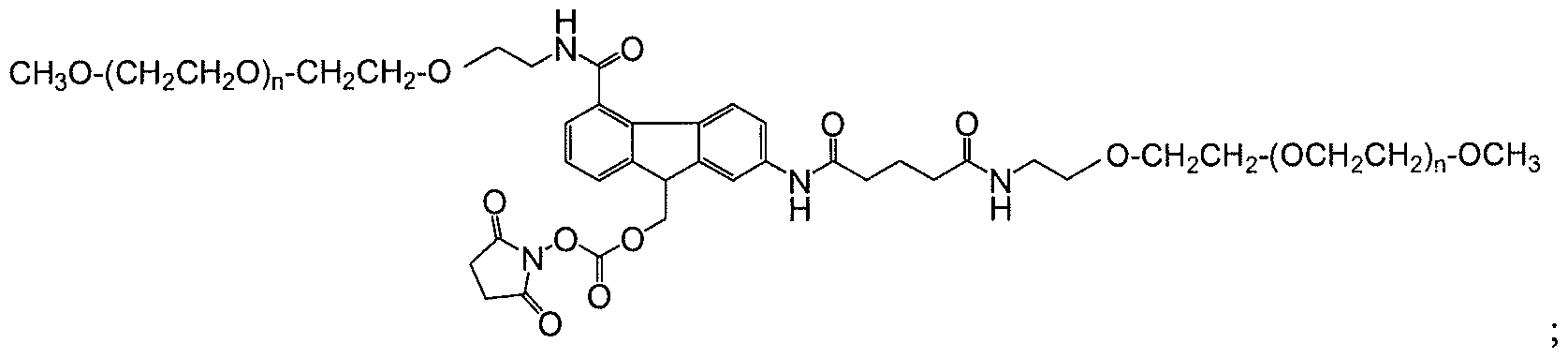
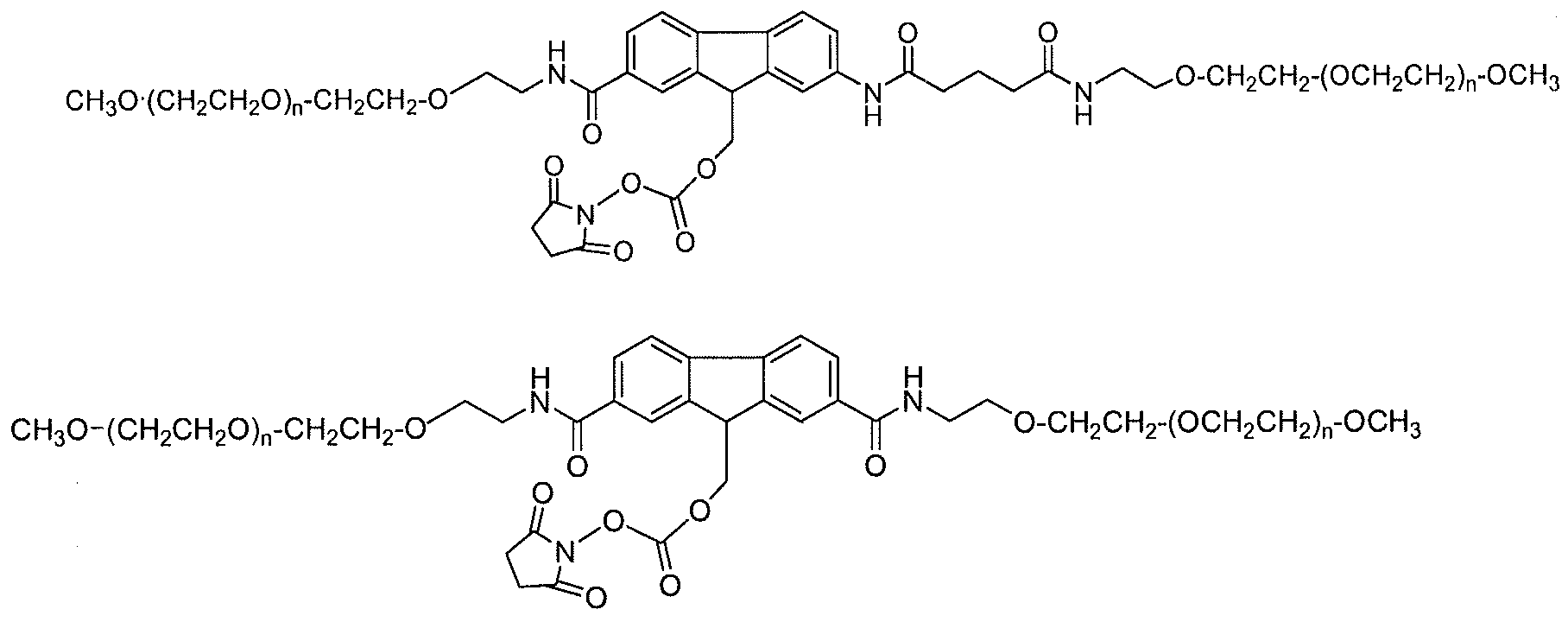
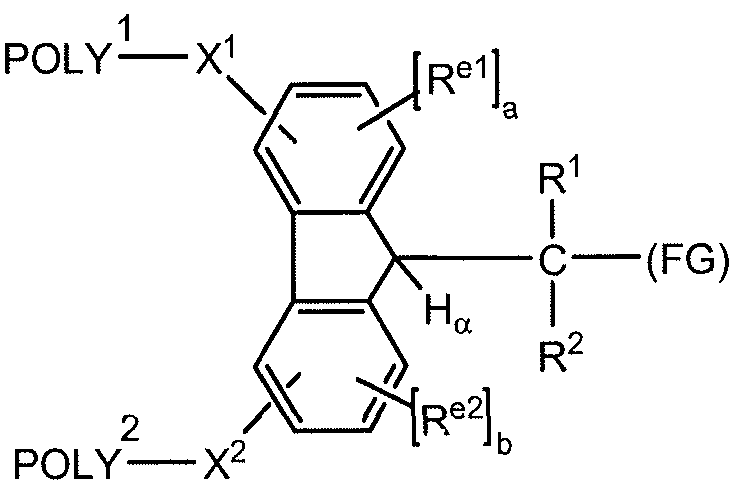
- An exemplar polymeric reagent has the following structure:
- POLY is a first water-soluble polymer
- POLY 2 is a second water-soluble polymer
- X 1 is a first spacer moiety
- X 2 is a second spacer moiety
- H a is an ionizable hydrogen atom
- R 1 is H or an organic radical
- R 2 is H or an organic radical
- R el when present, is a first electron altering group
- R e2 when present, is a second electron altering group
- FG is a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage.
- a releasable linkage such as a carbamate linkage.
- (FG) is a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage
- R 1 is H or an organic radical
- R 2 is H or an organic radical.
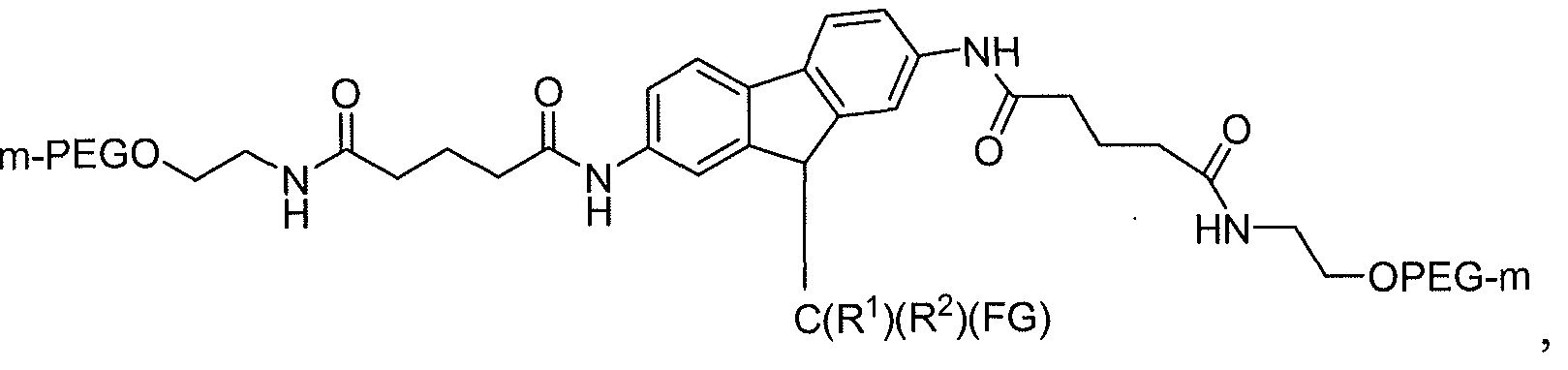
- Still other exemplary polymeric reagents have the structure:
- (n) is independently an integer from 4 to 1500.
- polymeric reagents can be prepared in any number of ways.
- polymeric reagents are not limited to the specific technique or approach used in their preparation. Exemplary approaches for preparing the polymeric reagents are described in the literature.
- polymeric reagents are useful for conjugation to biologically active agents.
- an amino group e.g., primary amine
- an active agent will react with the functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage.
- POLY 1 is a first water-soluble polymer
- POLY 2 is a second water-soluble polymer
- X 1 is a first spacer moiety
- X 2 is a second spacer moiety
- H a is an ionizable hydrogen atom
- R 1 is H or an organic radical
- R 2 is H or an organic radical
- R el when present, is a first electron altering group
- R e2 when present, is a second electron altering group
- Y 1 is O or S
- Y 2 is O or S
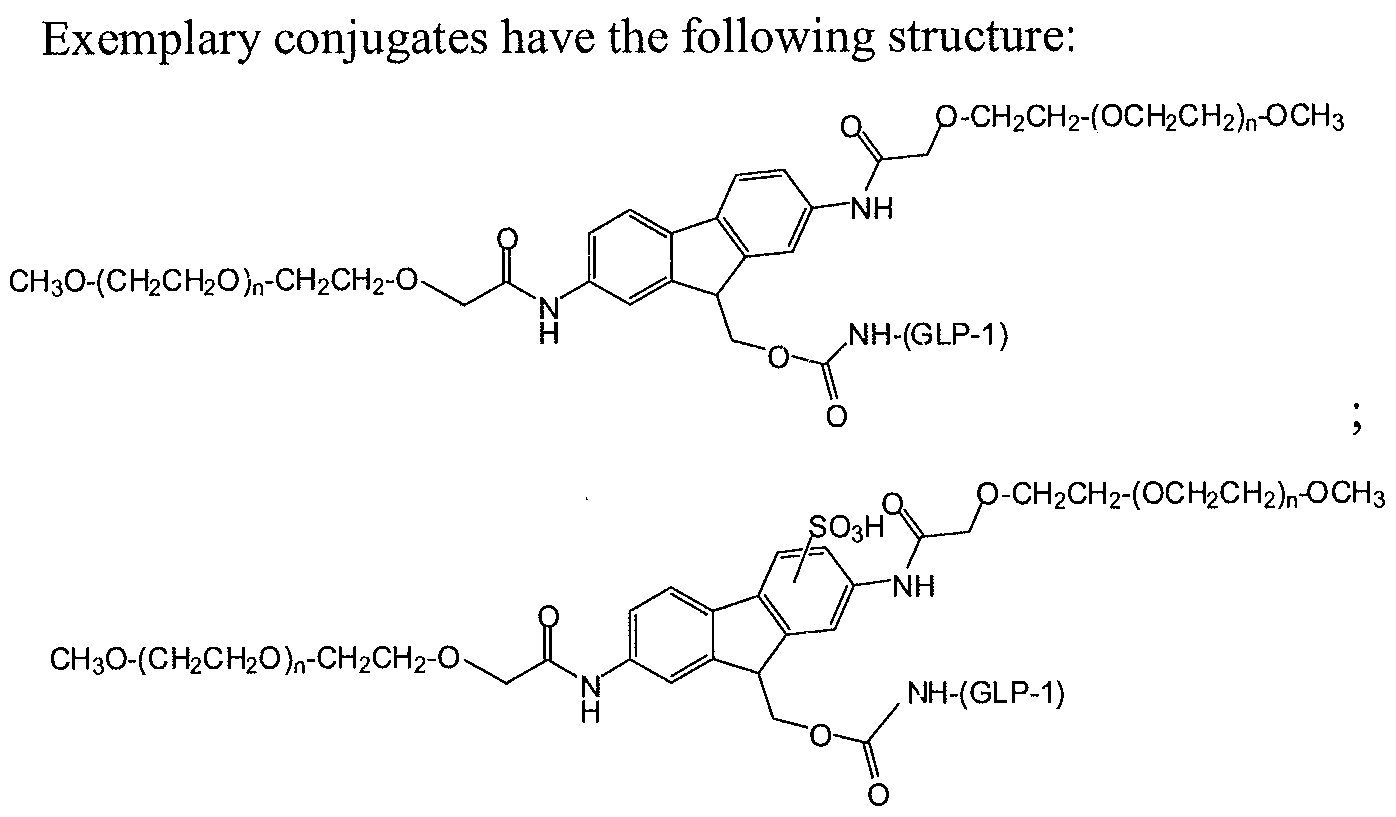
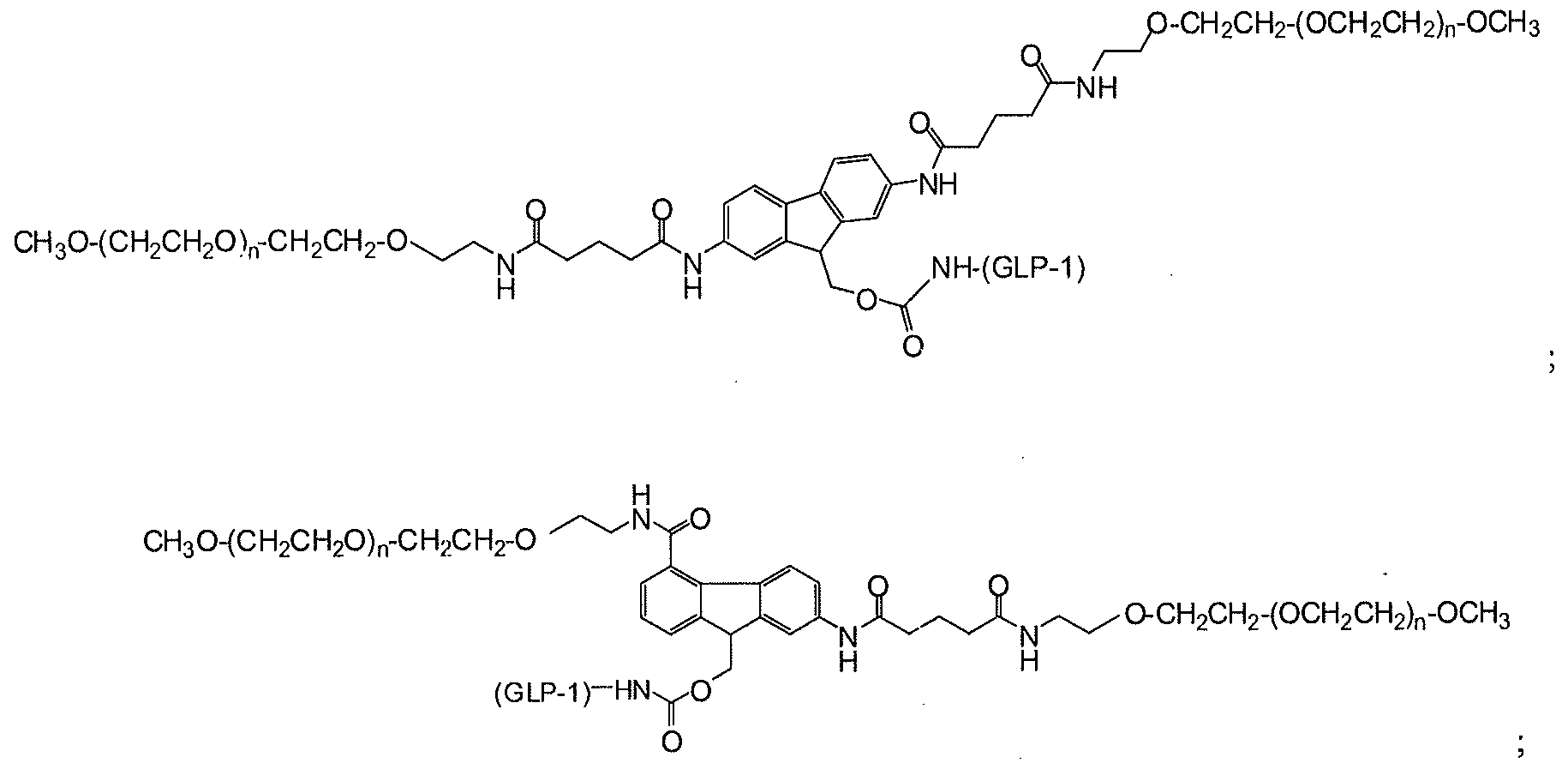
- GLP-1 is a residue of GLP-1 moiety.
- (n) is independently an integer from 4 to 1500, and (GLP-1) is a residue of a GLP-1 moiety.
- the biologically active agent to which a polymeric reagent as described herein can be conjugated is an amine-containing biologically active agent.
- the biologically active agent will be a macromolecule, such as a polypeptide, having a molecular weight greater than about 3,500 Daltons.
- Pharmacologically active polypeptides represent a preferred type of biologically active agent. It should be understood that for purposes of the present discussion, the term "polypeptide" will be generic for oligopeptides and proteins.
- the amine to which the polymeric reagent couples to can be on the N-terminus or an amine-containing side chain of an amino acid (such as lysine) within the polypeptide.
- the invention also provides for a method of preparing a conjugate comprising the step of contacting a polymeric reagent with a biologically active agent under conditions suitable to form a covalent attachment between the polymer and the biologically active agent.
- the polymer is added to the active agent or surface at an equimolar amount (with respect to the desired number of groups suitable for reaction with the reactive group) or at a molar excess.
- the polymeric reagent can be added to the target active agent at a molar ratio of about 1 : 1 (polymeric reagent: active agent), 1.5:1, 2:1, 3:1, 4: 1 , 5: 1 , 6: 1 , 8: 1, or 10: 1.
- the conjugation reaction is allowed to proceed until substantially no further conjugation occurs, which can generally be determined by monitoring the progress of the reaction over time. Progress of the reaction can be monitored by withdrawing aliquots from the reaction mixture at various time points and analyzing the reaction mixture by SDS-PAGE or MALDI-TOF mass spectrometry or any other suitable analytical method. Once a plateau is reached with respect to the amount of conjugate formed or the amount of unconjugated polymer remaining, the reaction is assumed to be complete. Typically, the conjugation reaction takes anywhere from minutes to several hours (e.g., from 5 minutes to 24 hours or more).
- the resulting product mixture is preferably, but not necessarily, purified to separate out excess reagents, unconjugated reactants (e.g., active agent) undesired multi-conjugated species, and free or unreacted polymer.
- the resulting conjugates can then be further characterized using analytical methods such as MALDI, capillary electrophoresis, gel electrophoresis, and/or chromatography.
- the conjugates can be purified to obtain/isolate different conjugated species.
- the product mixture can be purified to obtain the distribution of water-soluble polymer segments per active agent.
- the product mixture can be purified to obtain an average of anywhere from one to five PEGs per active agent (e.g., polypeptide).
- the strategy for purification of the final conjugate reaction mixture will depend upon a number of factors, including, for example, the molecular weight of the polymer employed, the particular active agent, the desired dosing regimen, and the residual activity and in vivo properties of the individual conjugate(s).
- conjugates having different molecular weights can be isolated using gel filtration chromatography. That is to say, gel filtration chromatography is used to fractionate differently numbered polymer-to-active agent ratios (e.g., 1-mer, 2-mer, 3-mer, and so forth, wherein “ 1-mer” indicates 1 polymer to active agent, "2-mer” indicates two polymers to active agent, and so on) on the basis of their differing molecular weights (where the difference corresponds essentially to the average molecular weight of the water-soluble polymer segments).
- polymer-to-active agent ratios e.g., 1-mer, 2-mer, 3-mer, and so forth, wherein “ 1-mer” indicates 1 polymer to active agent, "2-mer” indicates two polymers to active agent, and so on
- the resulting reaction mixture will likely contain unmodified protein (MW 100 kDa), mono- PEGylated protein (MW 120 kDa), di-PEGylated protein (MW 140 kDa), and so forth.
- RP-HPLC reverse phase-high performance liquid chromatography
- ion exchange chromatography using an ion exchange column, e.g., a SepharoseTM ion exchange column available from Amersham Biosciences. Either approach can be used to separate polymer-active agent isomers having the same molecular weight (positional isomers).
- An amine-containing biologically active agent for use in coupling to a polymer as presented herein is a GLP-1 moiety.
- GLP-1 itself (and not in a conjugate form), "GLP-1 " shall be understood to designate the peptide having the truncated "7-36" amino acid sequence (SEQ ID NO: l): NH 2 -His 7 -Ala 8 -Glu 9 -Gly 10 - ⁇
- GLP-1 (7-37)OH an exemplary GLP-1 moiety, shall be understood to designate the peptide having the following amino acid sequence (SEQ ID NO:l): NH 2 -His 7 -
- GLP-1 moieties for use in connection with the present invention include GLP-1 (1-36), GLP-1 analogs (such as those described in WO 91/1 1457), GLP-1 derivatives, GLP-1 moieties described in U.S. Published Patent Application No. 2004/0235710, GLP-1 biologically active fragments, extended GLP-1 (see, for example, WO 03/058203, in particular with respect to the extended glucagon-like peptide- 1 analogs described therein), N-terminal truncated fragments of GLP-1 (such as those described in EP 0 699 686, and exendins (including, for example, exendin-4 and analogs thereof).
- exendins are peptides that were first isolated from the salivary secretions of the Gila-monster and the Mexican Beaded Lizard.
- the exendins have a degree of similarity to several members of the GLP family, with the highest homology, 53%, to GLP-l(7-36)NH 2 (Goke, et al, J. Biol. Chem., 268: 19650-55, 1993).
- Particular exendins for use in the present invention include exendin-3 and exendin-4
- Exendin-3 (synthetic extendin-4 is also known as Exenatide).
- Exendin-3 (1-39) has the following amino acid sequence (SEQ ID NO:3): His-Ser-Asp-Gly-Thr-Phe-Thr-Ser-Asp-Leu-Ser-Lys-Gln- Met-Glu-Glu-Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-T -Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser- Gly-Ala-Pro-Pro-Pro-Ser-NH 2 .
- exendin-4 (1-39) corresponds to (SEQ ID NO:4): His-Gly-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Leu-Ser-Lys-Gln-Met-Glu-Glu- Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-Trp-Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser-Gly-Ala-Pro-Pro- Pro-Ser wherein the C-terminus serine is amidated.
- the GLP-1 moiety may be obtained from either non-recombinant methods or from recombinant methods, and the invention is not limited in this regard.
- GLP-1 moieties are commercially available, e.g., hGLP-1 , rExtendin-4, and rHuGLP-1 are available from ProSpecTany Techno Gene LTD (Rehovot, Israel); and (Ser8)GLP-l(7- 36)amide, hGLP-lamide, and hGLP-1 (7-36)Lys(biotin)amide are available from American Peptide Co., Sunnyvale, CA.
- Methods for preparing GLP-1 moieties are well-known, and are described, e.g., in U.S. Patent Nos. 5,118,666; 5,120,712; and 5,523,549.
- GLP-1 moieties can be prepared using standard methods of solution or solid phase peptide synthesis such as those described in Dugas H., Penny, C, Bioorganic Chemistry, Springer Verlag, New York, p. 54-92 (1981); Merrifield (1962) Chem Soc. 85:2149, and Stewart and Young, Solid Phase Peptide Synthesis, Freeman, San Francisco.
- Arg tosyl
- Asp cyclohexyl
- Glu cyclohexyl
- Ser benzyl
- Thr benzyl
- Tyr 4- bromocarbobenzoxy
- BOC deprotection may be carried out with trifluoroacetic acid in methylene chloride.
- the resulting peptide may be deprotected and cleaved from the resin using, e.g., anhydrous HF containing 10% meta-cresol.
- a GLP-1 moiety as described herein is prepared by constructing the nucleic acid encoding the desired polypeptide or fragment, cloning the nucleic acid into an expression vector, transforming a host cell ⁇ e.g. , plant, bacteria such as Escherichia coli, yeast such as Saccharomyces cerevisiae, or mammalian cell such as Chinese hamster ovary cell or baby hamster kidney cell), and expressing the nucleic acid to produce the desired polypeptide or fragment.
- a host cell ⁇ e.g. , plant, bacteria such as Escherichia coli, yeast such as Saccharomyces cerevisiae, or mammalian cell such as Chinese hamster ovary cell or baby hamster kidney cell
- the expression can occur via exogenous expression (when the host cell naturally contains the desired genetic coding) or via endogenous expression.
- Methods for producing and expressing recombinant polypeptides in vitro and in prokaryotic and eukaryotic host cells are known to those of ordinary skill in the art. See, for example, U.S. Patent No. 4,868,122.
- GLP-1 is typlically expressed in E. coli (since is does not require glycosylation for activity).
- nucleic acid sequences that encode an epitope tag or other affinity binding sequence can be inserted or added in-frame with the coding sequence, thereby producing a fusion protein comprised of the desired peptide and a polypeptide suited for binding.
- Fusion proteins can be identified and purified by first running a mixture containing the fusion protein through an affinity column bearing binding moieties (e.g., antibodies) directed against the epitope tag or other binding sequence in the fusion proteins, thereby binding the fusion protein within the column. Thereafter, the fusion protein can be recovered by washing the column with the appropriate solution (e.g. , acid) to release the bound fusion protein.
- binding moieties e.g., antibodies
- the GLP-1 moiety is not in the form of a fusion protein. See, for example, Dillon et al. (1993) Endocrinology 133:1907- 1910.
- GLP-1 activity For any given moiety, it is possible to determine whether that moiety possesses GLP-1 activity.
- Various assays may be used to assess bioactivity, including in- vitro and in-vivo assays that measure GLP-1 receptor binding activity or receptor activation. See, for example, EP 0 619 322 and U.S. Patent No. 5,120,712 for descriptions of assessing GLP-1 activity.
- a receptor-signaling assay may also be used to assess GLP-1 activity, such as described in Zlokarnik et al. (1998) Science 279:84-88.
- GLP-1 activity can also be used to determine whether a given moiety has GLP-1 activity. Such methods are useful for determining the GLP-1 activity of both the moiety itself (and therefore can be used as a "GLP-1 moiety"), as well as that of the corresponding polymer-moiety conjugate. For example, one can determine whether a given moiety is an agonist of the human GLP-1 receptor by assessing whether that moiety stimulates the formation of cAMP in a suitable medium containing the human GLP-1 receptor. The potency of such moiety is determined by calculating the EC50 value from a dose response curve.
- BHK cells baby hamster kidney cells
- expressing the cloned human GLP-1 receptor can be grown in DMEM media containing penicillin, streptomycin, calf serum, and Geneticin.
- the cells are then washed in phosphate buffered saline and harvested.
- Plasma membranes are then prepared from the cells by homogenization, and the homogenate is then centrifuged to produce a pellet.
- the resulting pellet is suspended by homogenization in a suitable buffer, centrifuged, and then washed.
- the cAMP receptor assay is then carried out by measuring cyclic AMP
- cAMP in response to the test insulinotropic moiety.
- cAMP can be quantified using the AlphaScreenTM cAMP Kit (Perkin Elmer). Incubations are typically carried out in microtiter plates in buffer, with addition of, e.g., ATP, GTP, IB MX (3-isobutyl-l-methylxanthine, Tween-20, BSA, acceptor beads, and donor beads incubated with biotinylated cAMP.
- Counting may be carried out, e.g., using the FusionTM instrument (Perkin Elmer).
- Concentration-response curves are then plotted for the individual insulinotropic moieties under evaluation, and their EC50 values determined.
- Biologically active fragments, deletion variants, substitution variants or addition variants of any of the foregoing that maintain at least some degree of GLP-1 activity can also serve as a GLP-1 moiety in the conjugates of the invention.
- GLP-1 moieties for use in connection with the present invention include any of the GLP-1 moieties described herein modified via methylation, N-terminal modification and/or glycosylation.
- a GLP-1 moiety may possess one or more methyl or other lower alkyl groups at one or more positions of the GLP-1 sequence.
- groups include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, pentyl, and so forth.
- Sites of modification include residues corresponding to positions 7, 8, 9, and/or 10 [based on a GLP-1 (7-36) numbering convention], with the 7 and/or 9 positions being preferred.
- DPP IV dipeptidyl peptidase IV
- GLP-1 N-methylated GLP-1
- alpha-methylated GLP-1 alpha-me-GLP-1
- desamidated GLP-1 desamino-GLP-1
- imidazole-lactic acid substituted GLP-1 imi-GLP-1
- the GLP-1 moieties described herein may also contain one or more glycosides.
- the GLP-1 moiety is preferably modified by introduction of a monosaccharide, a disaccharide or a trisaccharide.
- any site on the GLP-1 moiety may be modified by introduction of a saccharide, preferably, the saccharide is introduced at a residue or residues corresponding to any one or more of positions 7, 8, or 9 [based on a GLP-1 (7-36) numbering convention] to protect the peptide against DPP IV proteolysis.
- additional glycosides may be introduced, e.g., at any one or more of positions 22, 23 and 24 [again, based on a GLP-1 (7-36) numbering convention] to increase the helicity through the central portion of the peptide, as well as provide additional resistance to proteolysis.
- Glycosylated GLP-1 moieties are prepared using conventional Fmoc chemistry and solid phase peptide synthesis techniques, where the desired protected glycoamino acids are prepared prior to peptide synthesis and then introduced into the peptide chain at the desired position during peptide synthesis.
- Preparation of amino acid glycosides is described in U.S. Patent No. 5,767,254. Briefly, alpha and beta selective glycosylations of serine and threonine residues are carried out using the Koenigs-Knorr reaction and Lemieux's in situ anomerization methodology with Schiff base intermediates. Deprotection of the Schiff base glycoside is then carried out using mildly acidic conditions or hydrogenolysis.
- Monosaccharides that may be used for introduction at one or more amino acid residues of GLP-1 include glucose (dextrose), fructose, galactose, and ribose. Additional monosaccharides suitable for use include glyceraldehydes, dihydroxyacetone, erythrose, threose, erythrulose, arabinose, lyxose, xylose, ribulose, xylulose, allose, altrose, mannose, as well as others. Glycosides, such as mono-, di-, and trisaccharides for use in modifying a GLP-1 moiety, may be naturally occurring or may be synthetic.
- the present invention also includes pharmaceutical preparations comprising a conjugate as provided herein in combination with a pharmaceutical excipient.
- a conjugate as provided herein in combination with a pharmaceutical excipient.
- the conjugate itself will be in a solid form (e.g., a precipitate), which can be combined with a suitable pharmaceutical excipient that can be in either solid or liquid form.
- suitable pharmaceutical excipients include, without limitation, those selected from the group consisting of carbohydrates, inorganic salts, antimicrobial agents, antioxidants, surfactants, buffers, acids, bases, and combinations thereof.
- a carbohydrate such as a sugar, a derivatized sugar such as an alditol, aldonic acid, an esterified sugar, and/or a sugar polymer may be present as an excipient.
- Specific carbohydrate excipients include, for example: monosaccharides, such as fructose, maltose, galactose, glucose, D-mannose, sorbose, and the like; disaccharides, such as lactose, sucrose, trehalose, cellobiose, and the like; polysaccharides, such as raffinose, melezitose,
- maltodextrins dextrans, starches, and the like
- alditols such as mannitol, xylitol, maltitol, lactitol, xylitol, sorbitol (glucitol), pyranosyl sorbitol, myoinositol, and the like.
- the excipient can also include an inorganic salt or buffer such as citric acid, sodium chloride, potassium chloride, sodium sulfate, potassium nitrate, sodium phosphate monobasic, sodium phosphate dibasic, and combinations thereof.
- an inorganic salt or buffer such as citric acid, sodium chloride, potassium chloride, sodium sulfate, potassium nitrate, sodium phosphate monobasic, sodium phosphate dibasic, and combinations thereof.
- the preparation may also include an antimicrobial agent for preventing or deterring microbial growth.
- antimicrobial agents suitable for the present invention include benzalkonium chloride, benzethonium chloride, benzyl alcohol, cetylpyridinium chloride, chlorobutanol, phenol, phenylethyl alcohol, phenylmercuric nitrate, thimersol, and combinations thereof.
- An antioxidant can be present in the preparation as well. Antioxidants are used to prevent oxidation, thereby preventing the deterioration of the conjugate or other components of the preparation. Suitable antioxidants for use in the present invention include, for example, ascorbyl palmitate, butylated hydroxyanisole, butylated hydroxytoluene, hypophosphorous acid, monothioglycerol, propyl gallate, sodium bisulfite, sodium
- a surfactant may be present as an excipient.
- exemplary surfactants include: polysorbates, such as “Tween 20” and “Tween 80,” and pluronics such as F68 and F88 (both of which are available from BASF, Mount Olive, New Jersey); sorbitan esters; lipids, such as phospholipids such as lecithin and other phosphatidylcholines, phosphatidylethanolamines (although preferably not in liposomal form), fatty acids and fatty esters; steroids, such as cholesterol; and chelating agents, such as EDTA, zinc and other such suitable cations.
- Acids or bases may be present as an excipient in the preparation.
- acids that can be used include those acids selected from the group consisting of hydrochloric acid, acetic acid, phosphoric acid, citric acid, malic acid, lactic acid, formic acid, trichloroacetic acid, nitric acid, perchloric acid, phosphoric acid, sulfuric acid, fumaric acid, and combinations thereof.
- Suitable bases include, without limitation, bases selected from the group consisting of sodium hydroxide, sodium acetate, ammonium hydroxide, potassium hydroxide, ammonium acetate, potassium acetate, sodium phosphate, potassium phosphate, sodium citrate, sodium formate, sodium sulfate, potassium sulfate, potassium fumerate, and combinations thereof.
- the pharmaceutical preparations encompass all types of formulations and in particular those that are suited for injection, e.g., powders that can be reconstituted as well as suspensions and solutions.
- the amount of the conjugate (i.e., the conjugate formed between the active agent and the polymer described herein) in the composition will vary depending on a number of factors, but will optimally be a therapeutically effective dose when the composition is stored in a unit dose container (e.g., a vial).
- the pharmaceutical preparation can be housed in a syringe.
- a therapeutically effective dose can be determined experimentally by repeated administration of increasing amounts of the conjugate in order to determine which amount produces a clinically desired endpoint.
- the amount of any individual excipient in the composition will vary depending on the activity of the excipient and particular needs of the composition. Typically, the optimal amount of any individual excipient is determined through routine
- the excipient will be present in the composition in an amount of about 1% to about 99% by weight, preferably from about 5%-98% by weight, more preferably from about 15-95% by weight of the excipient, with concentrations less than 30% by weight most preferred.
- the pharmaceutical preparations of the present invention are typically, although not necessarily, administered via injection and are therefore generally liquid solutions or suspensions immediately prior to administration.
- the pharmaceutical preparation can also take other forms such as syrups, creams, ointments, tablets, powders, and the like.
- Other modes of administration are also included, such as pulmonary, rectal, transdermal, transmucosal, oral, intrathecal, subcutaneous, intra-arterial, and so forth.
- the conjugates can be administered parenterally by intravenous injection, or less preferably by intramuscular or by subcutaneous injection.
- Suitable formulation types for parenteral administration include ready- for-injection solutions, dry powders for combination with a solvent prior to use, suspensions ready for injection, dry insoluble compositions for combination with a vehicle prior to use, and emulsions and liquid concentrates for dilution prior to administration, among others.
- the invention also provides a method for administering a conjugate as provided herein to a patient suffering from a condition that is responsive to treatment with conjugate.
- the method comprises administering, generally via injection, a therapeutically effective amount of the conjugate (preferably provided as part of a pharmaceutical preparation).
- the method of administering may be used to treat any condition that can be remedied or prevented by administration of the particular conjugate.
- Those of ordinary skill in the art appreciate which conditions a specific conjugate can effectively treat.
- the actual dose to be administered will vary depend upon the age, weight, and general condition of the subject as well as the severity of the condition being treated, the judgment of the health care professional, and conjugate being administered.
- Therapeutically effective amounts are known to those skilled in the art and/or are described in the pertinent reference texts and literature.
- a therapeutically effective amount will range from about 0.001 mg to 100 mg, preferably in doses from 0.01 mg/day to 75 mg/day, and more preferably in doses from 0.10 mg/day to 50 mg/day.
- the unit dosage of any given conjugate (again, preferably provided as part of a pharmaceutical preparation) can be administered in a variety of dosing schedules depending on the judgment of the clinician, needs of the patient, and so forth.
- the specific dosing schedule will be known by those of ordinary skill in the art or can be determined
- Exemplary dosing schedules include, without limitation, administration five times a day, four times a day, three times a day, twice daily, once daily, three times weekly, twice weekly, once weekly, twice monthly, once monthly, and any combination thereof. Once the clinical endpoint has been achieved, dosing of the composition is halted.
- SDS-PAGE was used to analyze the purity and assess the molecular weights of GLP-1 conjugates. Samples were analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) using Bio-Rad system (Mini-PROTEAN III Precast Gel Electrophoresis System) ⁇ and Invitrogen system (XCell SureLock Mini-Cell). Samples were mixed with sample buffer. Then, the prepared samples were loaded onto a gel and run for approximately thirty minutes.
- RP-HPLC reversed phase high-performance liquid chromatography
- Agilent 1200 HPLC system Agilent 1200 HPLC system
- Samples were analyzed using a Intrada WP-RP column (3 ⁇ particle size, 150 x 4.6 mm, Silverton, Japan), and mobile phases consisting of 0.09% trifluoroacetic acid in water (buffer A) and 0.04% trifluoroacetic acid in acetonitrile (buffer B).
- the flow rate for the column was 0.5 ml/min.
- the protein and PEG-protein conjugates were eluted with a linear gradient 25%-65 % over 30 minutes, and were visualized using Diode Array detector at 220, 280, and 320 nm.
- dimers identified through RP-HPLC indicate protein dimer aggregates (and lack any polymeric component).
- HiTrap SP Sepharose HP cation exchange column (Amersham Biosciences) was used with the AKTAExplorerlOO system (GE Bioscience) to purify the PEG-GLP-1 conjugates.
- the conjugate solution was loaded on a column that was pre-equilibrated in 10 mM NaoAc buffer, pH 4.0 (buffer A) and then washed with ten column volumes of buffer A to remove any unreacted PEG reagent. Subsequently, a linear gradient of buffer A and buffer B (1 OmM NaOAc with 1.0 M NaCl buffer, pH 4.0) was applied over 20 column volume. The eluent was monitored by UV detector at 220nm, 280 nm, and 320 nm. The purity of individual fractions the purity of GLP-1 conjugates was determined by RP-HPLC and SDS-PAGE.
- mPEG 2 -C2-fmoc-40K-NHS stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging.
- 200 mG/mL stock solutions of were prepared in 2 mM HCl, and a molar ratio of 2.5: 1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM).
- MES buffer 0.5 M, pH 6.0
- a solution containing 1 M Hydroxyamine and 1 M glycine (pH 6.0) was added to the reaction mixture to final concentrations of 100 mM Hydroxyamine and 100 mM glycine to stop the PEGylation reaction and remove undesired imidazole PEG side product.
- the reaction was allowed to continue for one hour, and was diluted with H 2 0 to a conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
- the specificity of N-terminal PEGylation was determined to be 100% based on quantification of "signature" peptides. Briefly, protease digestion of PEG-containing compounds using trypsin and Lys-C created fragments. The Lys-C digestion created a mixture of peptide fragments 7-26, 27-34 and 35-36. If Lys26 was PEGylated, then no the digestion would not be expected to produce fragments 7-26 and 27-34 as the PEG component essentially protects the peptide from digestion at Lys26. Using this approach, it was determined that the specificity of N-terminal PEGylation was 100% (in view of the digestion pattern).
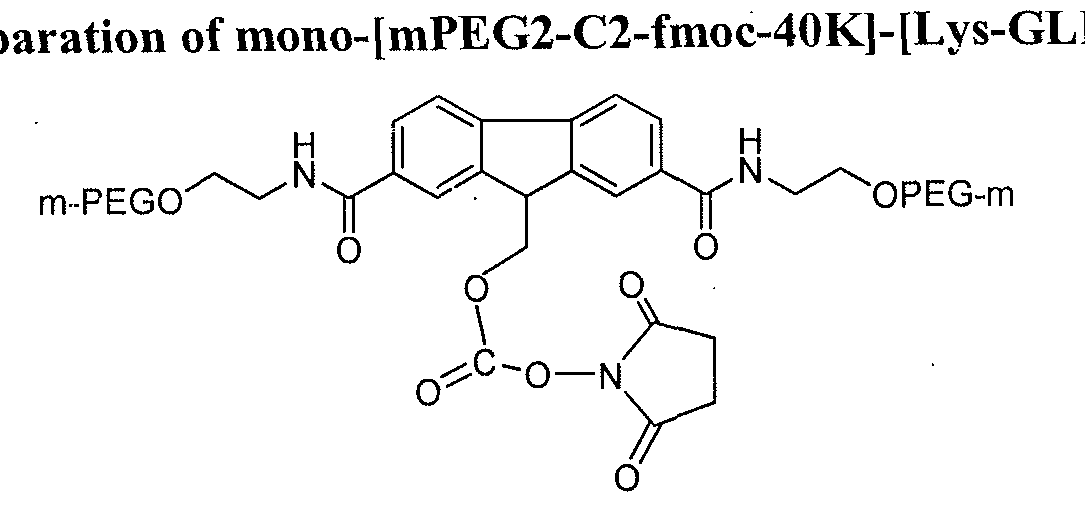
- a typical SP-HP cation exchange purification profile of mono-[mPEG2-C2- fmoc-40K]-[N ter -GLP-l] is provided in FIG. 1.
- the mono-[mPEG2-C2-fmoc-40K]-[N ter - GLP-1] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm and 225 nm).
- Purity analysis of mono-[mPEG2-C2-fmoc-40K]- [N ter -GLP-1] by reverse phase HPLC determined that the purity of the purified conjugate was determined to be 98.2 % at 280 nm.
- C2-fmoc-40K-NHS stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging.
- 200 mG/mL stock solutions of were prepared in 2 mM HCl, and a molar ratio of 1.5: 1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM).
- NaHC0 3 /Na 2 C0 3 buffer 1.0 M, pH 10.0
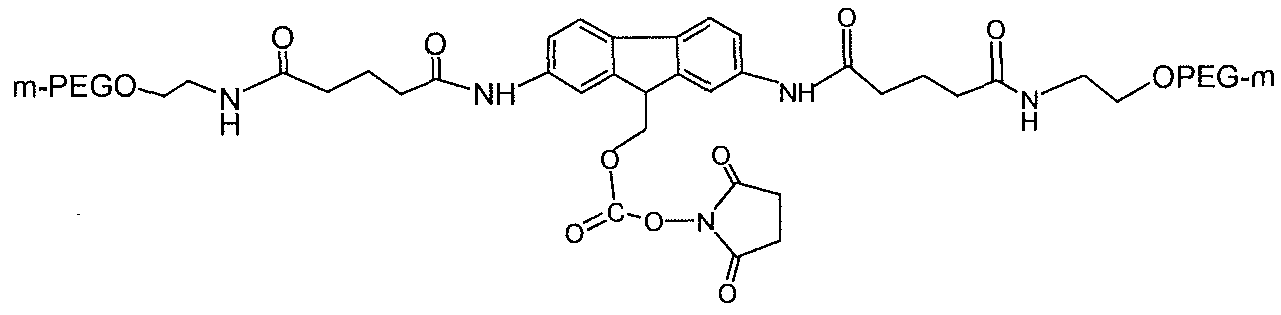
- a typical SP-HP cation exchange purification profile of mono-[mPEG2-C2- fmoc-40 ]-[Lys-GLP-l] is provided in FIG. 3.
- the mono-[mPEG.2-C2-fmoc-40K]-[Lys- GLP-1] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm and 225 nm.).
- Purity analysis of mono-[mPEG2-C2-fmoc-40K]- [Lys-GLP-1 ] by reverse phase HPLC determined that the purity of the purified conjugate was determined to be .99.8 % at 280 nm.
- MALDI-TOF analysis evidenced a peak at 43,966 Da, which agrees with the calculated molecular weight of mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l ].
- Another peak at 25,548 Da might represent a doubly charged conjugate.
- mPEG 2 -G2-fmoc-40K-NHS stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging.
- 200 mG/mL stock solutions of were prepared in 2 raM HC1, and a molar ratio of 2.5:1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM).
- MES buffer 0.5 M, pH 6.0
- a solution containing 1 M Hydroxyamine and 1 M glycine (pH 6.0) was added to the reaction mixture to final concentrations of 100 mM Hydroxyamine and 100 mM glycine to stop the PEGylation reaction and remove undesired imidazole PEG side product.
- the reaction was allowed to continue for one hour, and was diluted with H 2 0 to the conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
- a typical SP-HP cation exchange purification profile of mono-[mPEG2-G2- fmoc-40K]-[N ter -GLP-l] is provided in FIG. 5.
- the mono-[mPEG2-G2-fmoc-40K]-[N ter - GLP-1 ] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm and 225 nm).
- G2-fmoc-40K-NHS stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging.
- 200 mG/mL stock solutions of were prepared in 2 mM HC1, and a molar ratio of 1.5: 1 (PEG/GLP-1 ) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM).
- NaHC0 3 /Na 2 C0 3 buffer 1.0 M, pH 10.0
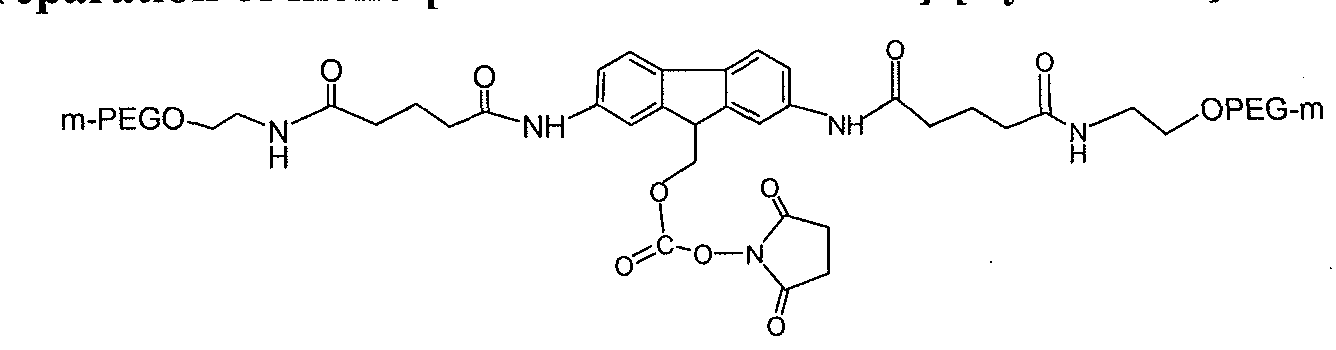
- a typical SP-HP cation exchange purification profile of mono-[mPEG2-G2- fmoc-40K]-[Lys-GLP-l] is provided in FIG. 7.
- the mono-[mPEG2-G2-fmoc-40K]-[Lys- GLP-1] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm 225 nm).
- Purity analysis of mono-[mPEG2-G2-fmoc-40K]-[Lys- GLP-1 ] by reverse phase HPLC determined that the purity of the purified conjugate was determined to be 99.8 % at 280 nm. The peak at 15.8 minute was GLP-1 (0.2 %). See
- FIG. 8 Purity analysis by 4-12 % NuPage Bis-Tris SDS-PAGE with Coomassie Blue staining resulted in an apparent large molecular weight (66-54 kDa) of mono-[mPEG2-G2- fmoc-40K]-[Lys-GLP-l], likely attributable to the slow mobility of the conjugate through the gel due to a high degree of PEG hydration.
- the PEG group bound to GLP-1 causes the conjugate to smear on the gel. Impurities were not detected.
- MALDI-TOF analysis evidenced a peak at 44,037 Da, which agrees with the calculated molecular weight of mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l ].
- Another peak at 25,474 Da might represent a doubly charged conjugate.
- mPEG 2 -G2-fmoc-40K-NHS stored at -80 °C under argon, is warmed to ambient temperature under nitrogen purging.
- 200 mG/mL stock solutions of were prepared in 2 mM HCl, and a molar ratio of 4.5:1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM).
- MES buffer 0.5 M, pH 6.0
- a solution containing 1 M Hydroxyamine and 1 M glycine (pH 6.0) was added to the reaction mixture to final concentrations of 100 mM Hydroxyamine and 100 mM glycine to stop the PEGylation reaction and remove undesired imidazole PEG side product.
- the reaction was allowed to continue for one hour, and was diluted with H 2 0 to the conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
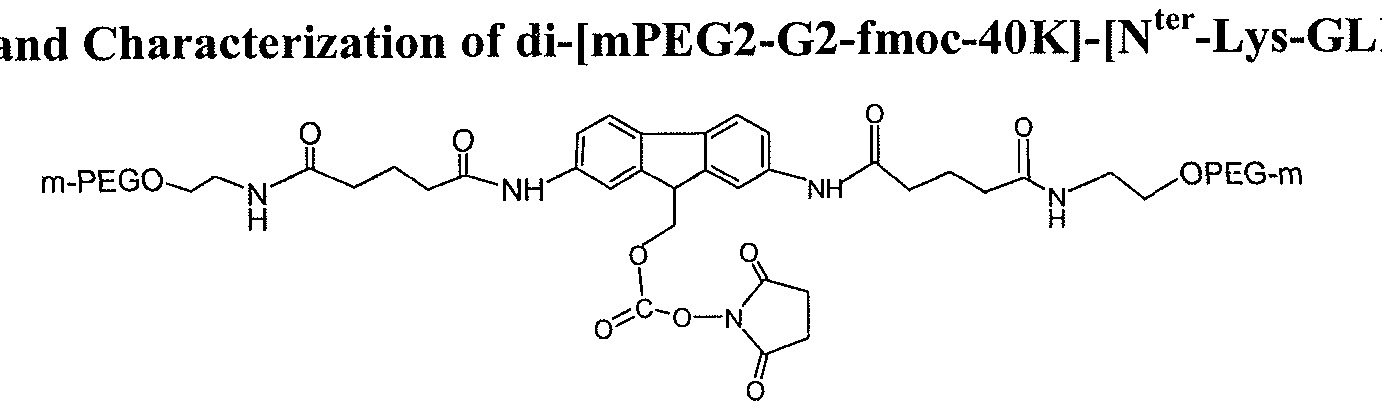
- FIG. 40 -[N ter -Lys-GLP-l] is provided in FIG. 9.
- the di-[mPEG2-G2-fmoc-40K]-[N ter -Lys- GLP-1], mono-mPEG2-G2-fmoc-40K-N ter -GLP-l and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm 225 nm).
- Purity analysis of di-[mPEG2-G2-fmoc-40K]-[N ter -Lys-GLP-l] by reverse phase HPLC determined that the purity of the purified conjugate is determined to be 100 % at 280 nm. See FIG. 10.
- the gradient settings for GLP-1 conjugates and PEG quantification were as follows: mobile phase A - 0.1 % fomic acid/H 2 0; and mobile phase B - 0.1 % fomic acid/acetonitrile, wherein the parameters for each step were as follows: step 0, total time - 0.00, flow rate 500 ⁇ /min, 80%A/20%B; step 1, total time - 1.00, flow rate 500 ⁇ /min, 80%A/20%B; step 2, total time - 6.00, flow rate 500 ⁇ /min, 55%A/45%B; step 3, total time - 1 1.00, flow rate 500 ⁇ /min, 35%A/65%B ' ; step 4, total time - 11.30, flow rate 500 ⁇ /min, 10%A/90%B; step 5, total time - 13.00, flow rate 500 ⁇ /min, 10%A/90%B; and step 6, total time - 13.01 , flow rate 750 ⁇ /min, 80%A/20%B.
- the gradient settings for GLP-1 (7-36) and GLP-1 (9-36) quantification were as follows: mobile phase A - 10 mM ammonium acetate/H 2 0; and mobile phase B - 10 mM ammonium acetate/acetonitrile, wherein the parameters for each step were as follows: step 0, total time - 0.00, flow rate 500 ⁇ /min, 75%A/25%B; step 1, total time - 1.00, flow rate 500 ⁇ /min, 75%A/25%B; step 2, total time - 8.00, flow rate 500 ⁇ /min, 55%A/45%B; step 3.
- Mass spectrometer parameters were set to conventional conditions and the parameters associated with the auto-injector were as follows: seat capillary volumne - 2.3 ⁇ ; eject speed - 200 ⁇ ; injection volume - 20.00 ⁇ ; syringe volume - ⁇ ; injection mode - standard; prefetch vial mode - off; draw speed - 200 ⁇ 7 ⁇ 6; and the draw position was 0.
- the plasma incubation and extraction procedure involved a 5 mL aliquot of freshly collected, heparin treated, EDTA-free pooled s/d rat plasma, or
- IXfreeze/thawed rat plasma into a 15 mL corning centrifuge tube. To this was added 1.2 mL of 5X PBS solution to 5 mL plasma, which was then mixed by inverting the tube. The final concentration of PBS in rat plasma was 1 X PBS. The tube was centrifuged and kept on ice until used. Immediately before plasma incubation, the plasma was pre-warmed to 37 °C for five minutes. Plasma incubation time points were set as follows: T 0, 0.25 hour, 0.5 hour, 1 hour, 2 hour, 3 hour, 4 hour, 6 hour, 8 hour, 12 hour, and 24 hour. For each time point, duplicate samples were prepared by labeling 2 1.5 mL Eppendorf tubes. Protease inhibitor mix (25 uL of 10X HALT) prepared according to conventional methods, was added to the tubes and tubes were kept on ice until use.
- Protease inhibitor mix 25 uL of 10X HALT
- protease inhibitor mix 100 uL of
- 10X Halt solution or 1/10 volume of the blood to be collected, was placed into pre-chilled, heparin coated tubes. Approximately 1 mL of blood was added to the tube and mixed gently by inverting the tube five times. Following collection, tubes containing samples were centrifuged at approximately 2500rpm for five minutes (at 2-6°C). Thereafter, two aliquots of resulting plasma (approximately 200 uL each) were transferred to pre-labeled tubes. The tubes were frozen on dry ice and stored at -80°C pending analysis.
- FIG. 11A shows the decrease of mono-[mPEG2-G2-fmoc-40K]-[N ter -GLP-l], and corresponding formation of free, released PEG, revealing the release of active GLP-1 peptide as a result of PEG linker release.
- FIG. 11B revealed a stable level of GLP-1 (7-36) as a result of PEG linker release throughout the plasma incubation period.
- Table 1 below provides a summary of the buffer and matrix effects as a function of ti /2 of PEG linker release of GLP-1 conjugates of Examples 1-5.
- test compounds Serial dilutions of test compounds (GLP-1 conjugates) were prepared in assay buffer containing protease inhibitors and equilibrium binding assays were conducted at room temperature for 3 hours using 00 pM 125 I-GLP-1 as the competitive radioligand. At the end of the incubation, cells were washed with phosphate buffered saline, solubilized in 0.2M NaOH and bound radioactivity was counted using a gamma counter. Non-specific binding was measured in the presence of excess cold GLP-1 (1000 nM) and subtracted from the total binding values to yield values for specific binding at each test compound concentration. IC 5 0 values for each compound were obtained from a non-linear regression fit of the dose-response curve.
- mice (BKS.Cg- Lepr db /Lepr db /0 ⁇ aHsd) were obtained from Harlan
- mice Male animals 10-12 weeks old and weighing 35-45 g were randomly assigned into different testing groups (five mice in each group). ⁇ 10 ⁇ 1 of blood was collected from mouse tail and the baseline blood glucose level in the animal was measured by a glucometer (OneTouch Ultra from LifeScan). Animals were injected intraperitoneally with either saline, test article, and an equimolar (based on GLP-1) of PEGylated GLP-1 conjugates (each equivalent to 75 ⁇ g of GLP-1). At different time points after compound administration, animal blood glucose levels were monitored using the glucometer. Food was removed during the whole study. FIG.
- FIG. 12 shows the results associated with mice treated with native (i.e., unconjugated) GLP-1 peptide, wherein glucose lowering effect in vivo was lost after four hours of dosing.
- FIG. 13A shows an eight-hour plot and FIG. 13B shows a 48-hour plot of the results. Data points are the average glucose levels from 5 mice, error bars represent standard error (SEM). In each of FIG. 13A and FIG.
- Example 13B Example 5, Example 4, Example 2, Example 3 and Example 1 , corresponds to di-[mPEG2-G2-frnoc-40K]-[N ter -Lys-GLP-l], mono-[mPEG2-G2-frnoc- 40K]-[Lys-GLP-l], mono-[mPEG2-C2-frnoc-40K]-[Lys-GLP-l], mono-[mPEG2-G2-frnoc- 40K]-[N ter -GLP-l ], mono-[mPEG2-C2-frnoc-40K]-[N ter -GLP-l ], respectively.
- Example 14B Example 5, Example 4, Example 2, Example 3 and Example 1, corresponds to di- [mPEG2-G2-fmoc-40K]-[N ter -Lys-GLP-l], mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l], mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l], mono-[mPEG2-G2-fmoc-40K]-[N ter -GLP-l], mono-[mPEG2-C2-fmoc-40K]-[N ter -GLP-l], respectively.
- FIG. 15A shows the plasma GLP-1 peptide concentration after intraperitoneal dosing. Compared to native GLP-1 peptide which was cleared from blood circulation quickly and undectable after eight hours postdosing, various PEG-GLP-1 conjugates could release GLP-1 over an extensively prolonged time. Released GLP-1 peptide could still be detected at 120 hours post dosing. In agreement with in vitro results, the plasma rates of free GLP-1 appearance from a conjugated form followed PEG linker hydrolysis chemistry quantitatively in vivo: conjugates with a relatively faster release rate released higher concentrations of GLP- 1 peptide than the slower-releasing conjugates.
- FIG. 15B shows the plasma concentration of PEG-GLP-1 conjugates.
- FIG. 15C and FIG. 15D show released PEG molecules and the degraded GLP-1 metabolite (9-36) peptide in the plasma post-dosing, respectively.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Gastroenterology & Hepatology (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Endocrinology (AREA)
- Toxicology (AREA)
- Zoology (AREA)
- Epidemiology (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Medicinal Preparation (AREA)
Abstract
The present invention provides GLP-1 -polymer conjugates having a releasable linkage. Methods of making conjugates and methods for administering conjugates are also provided.
Description
GLP-1 POLYMER CONJUGATES HAVING A RELEASABLE LINKAGE
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This application claims the benefit of priority under 35 U.S.C. § 119(e) to U.S.
Provisional Patent Application No. 61/406,040, filed October 22, 2010, the disclosure of which is incorporated herein by reference in its entirety.
FIELD OF THE INVENTION
[0002] The present invention relates generally to polymer-active agent conjugates having a releasable linkage to thereby release the active agent in vivo. In addition, the invention relates to, among other things, methods for synthesizing the conjugates, methods for purifying the conjugates, and so on.
BACKGROUND OF THE INVENTION
[0003] Scientists and clinicians face a number of challenges in their attempts to develop active agents into forms suited for delivery to a patient. Active agents that are polypeptides, for example, are often delivered via injection rather than orally. In this way, the polypeptide is introduced into the systemic circulation without exposure to the proteolytic environment of the stomach. Injection of polypeptides, however, has several drawbacks. For example, many polypeptides have a relatively short half-life, thereby necessitating repeated injections, which are often inconvenient and painful. Moreover, some polypeptides can elicit one or more immune responses with the consequence that the patient's immune system attempts to destroy or otherwise neutralize the immunogenic polypeptide. Of course, once the polypeptide has been destroyed or otherwise neutralized, the polypeptide cannot exert its intended pharmacodynamic activity. Thus, delivery of active agents such as polypeptides is often problematic even when these agents are administered by injection.
[0004] Some success has been achieved in addressing the problems of delivering active agents via injection. For example, conjugating the active agent to a water-soluble polymer has resulted in a polymer-active agent conjugate having reduced immunogenicity and antigenicity. In addition, these polymer-active agent conjugates often have greatly increased half-lives compared to their unconjugated counterparts as a result of decreased
clearance through the kidney and/or decreased enzymatic degradation in the systemic circulation. As a result of having a greater half-life, the polymer- active agent conjugate requires less frequent dosing, which in turn reduces the overall number of painful injections and inconvenient visits with a health care professional. Moreover, active agents that were only marginally soluble demonstrate a significant increase in water solubility when conjugated to a water-soluble polymer.
[0005] Due to its documented safety as well as its approval by the FDA for both topical and internal use, polyethylene glycol has been conjugated to active agents. When an active agent is conjugated to a polymer of polyethylene glycol or "PEG," the conjugated active agent is conventionally referred to as "PEGylated." The commercial success of PEGylated active agents such as PEGASYS® PEGylated interferon alpha-2a (Hoffmann-La Roche, Nutley, NJ), PEG-INTRON® PEGylated interferon alpha-2b (Schering Corp., Kennilworth, NJ), and NEULASTA™ PEG-filgrastim (Amgen Inc., Thousand Oaks, CA) demonstrates that administration of a conjugated form of an active agent can have significant advantages over the unconjugated counterpart. Small molecules such as
distearoylphosphatidylethanolamine (Zalipsky (1993) Bioconjug. Chem. 4(4):296-299) and fluorouracil (Ouchi et al. (1992) Drug Des. £>z',scov.9(l):93-105) have also been PEGylated. Harris et al. have provided a review of the effects of PEGylation on pharmaceuticals. Harris et al. (2003) Nat. Rev. Drug Discov. 2(3):214-221.
[0006] Despite these successes, conjugation of a polymer to an active agent to result in a commercially relevant drug is often challenging. For example, conjugation can result in the polymer being attached at or near a site on the active agent that is necessary for pharmacologic activity (e.g., at or near a binding site). Such conjugates may therefore have unacceptably low activity due to, for example, the steric effects introduced by the polymer. Attempts to remedy conjugates having unacceptably low activity can be frustrated when the active agent has few or no other sites suited for attachment to a polymer. Thus, additional PEGylation alternatives have been desired.
[0007] One suggested approach for solving this and other problems is "reversible
PEGylation" wherein the native active agent (or a moiety having increased activity compared to the PEGylated active agent) is released. For example, reversible PEGylation has been disclosed in the field of cancer chemotherapies. See Greenwald (1997) Exp. Opin. Ther. Patents 7(6):601-609. U.S. Patent Application Publication No. 2005/0079155 describes conjugates using reversible linkages. As described in this publication, reversible linkages can
be effected through the use of an enzyme substrate moiety. It has been pointed out, however, that approaches relying on enzymatic activity are dependent on the availability of enzymes. See Peleg-Schulman (2004) J. Med. Chem. 47:4897-4904. Patient variability around the amount and activity of these enzymes can introduce inconsistent performance of the conjugate among different populations. Thus, additional approaches that do not rely on enzymatic processes for polymer release have been described as being desirable.
[0008] Another approach for reversible PEGylation is described in U.S. Patent No.
7,060,259, which described (among other things) water-soluble prodrugs in which a biologically active agent is linked to a water-soluble non-immunogenic polymer by a hydrolyzable carbamate bond. As described therein, the biologically active agent can be readily released by the hydrolysis of the carbmate bond in vivo without the need for adding enzymes or catalytic materials.
[0009] Another approach for reversible PEGylation is described in Peleg-Schulman
(2004) J. Med. Chem. 47:4897-4904, WO 2004/089280 and U.S. Patent Application
Publication No. 2006/0171920. Yet another releaseable approach is described in U.S. Patent Application Publication No. 2006/0293499.
[0010] In the area of diabetes (e.g., adult-onset diabetes), GLP-1 can sometimes be administered to a patient to address or otherwise ameliorate this disorder. Due to the relatively short half-life of GLP-1, however, it would be advantageous to increase the in vivo half-life of these proteins by, for example, reversible PEGylation. Thus, the present invention seeks to solve this and other needs in the art.
SUMMARY OF THE INVENTION
[0011] In one or more embodiments of the invention, a conjugate of the following formula is provided:
POLY1 is a first water-soluble polymer;
POLY is a second water-soluble polymer;
X1 is a first spacer moiety;
X is a second spacer moiety;
Ha is an ionizable hydrogen atom;
R1 is H or an organic radical;
R2 is H or an organic radical;
(a) is either zero or one;
(b) is either zero or one;
Rel, when present, is a first electron altering group;
Re2, when present, is a second electron altering group; and
Y1 is O or S;
Y2 is O or S; and
(GLP-1) is a residue of an amine-containing GLP-1 moiety.
[0012] In one or more embodiments of the invention, methods for preparing conjugates are provided.
[0013] In one or more embodiments of the invention, pharmaceutical preparations comprising the conjugates are provided.
[0014] In one or more embodiments of the invention, methods for administering the conjugates are provided.
[0015] In one or more embodiments of the invention, a GLP-1 - water polymer conjugate is provided, the conjugate having an in vivo half-life increased by a factor of at least 1.5 as compared to the in vivo half-life of the GLP-1 moiety not conjugated to the water-soluble polymer.
[0016] In one or more embodiments of the invention, a GLP-1 -water soluble polymer conjugate is provided, the conjugate having an in vivo half-life increased by a factor of at least 2 as compared to the in vivo half-life of a GLP-1 moiety not conjugated to the water-soluble polymer.
BRIEF DESCRIPTION OF THE FIGURES
[0017] FIG. 1 is a typical SP-HP cation exchange purification profile of
mono-[mPEG2-C2-fmoc-40K]-[Nter-GLP-l], as further described in Example 1.
[0018] FIG. 2 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-C2-fmoc-40K]-[Nter-GLP-l], as further described in Example 1.
[0019] FIG. 3 is a typical SP-HP cation exchange purification profile of
mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l ], as further described in Example 2.
[0020] FIG. 4 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l], as further described in Example 2.
[0021] FIG. 5 is a typical SP-HP cation exchange purification profile of
morio-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l], as further described in Example 3.
[0022] FIG. 6 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l], as further described in Example 3.
[0023] FIG. 7 is a typical SP-HP cation exchange purification profile of
mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l], as further described in Example 4.
[0024] FIG. 8 is a reverse phase HPLC chromatogram of a purified composition of mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l], as further described in Example 4.
[0025] FIG. 9 is a typical SP-HP cation exchange purification profile of di-[mPEG2-
G2-fmoc-40 ]-[Nter-Lys-GLP-l], as further described in Example 5.
[0026] FIG. 10 is a reverse phase HPLC chromatogram of a purified composition of di-[mPEG2-G2-fmoc-40K]-[Nter-Lys-GLP-l], as further described in Example 5.
[0027] FIG. 11A is a plot showing the decrease on mono-[mPEG2-G2-fmoc-40k]-
[Nter-GLP-1] and the corresponding formation of free, released PEG, revealing the release of active GLP-1 peptide as a result of PEG linker release. FIG.llB reveals the stable level of GLP-1 (7-36) as a result of PEG linker release throughout the plasma incubation period. Both FIG. 11A and FIG. 11B are described in further detail in Example 6.
[0028] FIG. 12 a plot of mice treated with native (i.e., unconjugated) GLP-1 peptide that lost in vivo effect after four hours of dosing, as further described in Example 8.
[0029] FIG. 13A shows an eight-hour plot and FIG. 13B shows a 48-hour plot of the results of the results of saline, and several GLP-1 -containing conjugates, as further described in Example 8.
[0030] FIG. 14A and FIG. 14B is a pair of plots showing GLP-1 conjugate's percent glucose lowering activity in db/db mice (relative to saline control at the 24 hour time point) and the in vitro ti/2. As further described in Example 8, an inverse relationship appears between a GLP-1 conjugate's glucose lowering in mice and the in vitro ti/2.
[0031] FIG. 15A, FIG. 15B, FIG. 15C, and FIG. 15D are each plots showing the plasma concentration curves of tested conjugates following intraperitoneally (i.p.) of GLP-1 (7-36), PEG-GLP-1 , PEG, and GLP-1 (9-36), respectively.
DETAILED DESCRIPTION OF THE INVENTION
[0032] Before describing the present invention in detail, it is to be understood that this invention is not limited to particular polymers, synthetic techniques, active agents, and the like, as such may vary.
[0033] It must be noted that, as used in this specification and the claims, the singular forms "a," "an," and "the" include plural referents unless the context clearly dictates otherwise. Thus, for example, reference to a "polymer" includes a single polymer as well as two or more of the same or different polymers, reference to a "conjugate" refers to a single conjugate as well as two or more of the same or different conjugates, reference to an
"excipient" includes a single excipient as well as two or more of the same or different excipients, and the like.
[0034] In describing and claiming the present invention, the following terminology will be used in accordance with the definitions described below.
[0035] The term "GLP-1 moiety," as used herein, refers to those peptides,
polypeptides and proteins having GLP-1 activity, including (for example) GLP-1. Prior to conjugation, the GLP-1 moiety has at least one electrophilic group or nucleophilic group suitable for reaction with a water soluble polymer. In addition, the term "GLP-1 moiety" encompasses both the GLP-1 moiety prior to conjugation as well as the GLP-1 moiety residue following conjugation. As will be explained in further detail below, one of ordinary skill in the art can determine whether any given moiety has GLP-1 activity.
[0036] As used herein, the terms "peptide," "polypeptide," and "protein," refer to polymers comprised of amino acid monomers linked by amide bonds. For use herein, each of "peptide," "polypeptide" and "protein" will be referred to as "peptide." Peptides may include the standard 20 a-amino acids that are used in protein synthesis by cells (i. e. natural amino acids), as well as non-natural amino acids (non-natural amino acids nay be found in nature, but not used in protein synthesis by cells, e.g., ornithine, citrulline, and sarcosine, or may be chemically synthesized), amino acid analogs, and peptidomimetics. The amino acids may be D- or L-optical isomers. Peptides may be formed by a condensation or coupling reaction between the a-carbon carboxyl group of one amino acid and the amino group of another amino acid. The terminal amino acid at one end of the chain (amino terminal) therefore has a free amino group, while the terminal amino acid at the other end of the chain (carboxy terminal) has a free carboxyl group. Alternatively, the peptides may be non-linear, branched peptides or cyclic peptides. Moreover, the peptides may optionally be modified or protected with a variety of functional groups or protecting groups, including on the amino and/or carboxy terminus.
[0037] Amino acid residues in peptides are abbreviated as follows: Phenylalanine is
Phe or F; Leucine is Leu or L; Isoleucine is He or I; Methionine is Met or M; Valine is Val or V; Serine is Ser or S; Proline is Pro or P; Threonine is Thr or T; Alanine is Ala or A;
Tyrosine is Tyr or Y; Histidine is His or H; Glutamine is Gin or Q; Asparagine is Asn or N; Lysine is Lys or K; Aspartic Acid is Asp or D; Glutamic Acid is Glu or E; Cysteine is Cys or C; Tryptophan is Trp or W; Arginine is Arg or R; and Glycine is Gly or G.
[0038] The terms "therapeutic peptide fragment" or "fragments of therapeutic peptides" refer to a peptide that comprises a truncation at the amino-terminus and/or a truncation at the carboxyl-terminus of a therapeutic peptide as defined herein. The terms "therapeutic peptide fragment" or "fragments of therapeutic peptides" also encompasses amino-terminal and/or carboxyl-terminal truncations of therapeutic peptide variants and therapeutic peptide derivatives. Therapeutic peptide fragments may be produced by synthetic techniques known in the art or may arise from in vivo protease activity on longer peptide sequences. It will be understood that therapeutic peptide fragments retain some or all of the therapeutic activities of the therapeutic peptides.
[0039] As used herein, the terms "therapeutic peptide variants" or "variants of therapeutic peptides" refer to therapeutic peptides having one or more amino acid
substitutions, including conservative substitutions and non-conservative substitutions, amino
acid deletions (either internal deletions and/or C- and/or N- terminal truncations), amino acid additions (either internal additions and/or C- and/or N- terminal additions, e.g., fusion peptides), or any combination thereof. Variants may be naturally occurring (e.g. homologs or orthologs), or non-natural in origin. The term "therapeutic peptide variants" may also be used to refer to therapeutic peptides incorporating one or more non-natural amino acids, amino acid analogs, and peptidomimetics. It will be understood that, in accordance with the invention, therapeutic peptide fragments retain some or all of the therapeutic activities of the therapeutic peptides.
[0040] The terms "therapeutic peptide derivatives" or "derivatives of therapeutic peptides" as used herein refer to therapeutic peptides, therapeutic peptide fragments, and therapeutic peptide variants that have been chemically altered other than through covalent attachment of a water-soluble polymer. It will be understood that, in accordance with the invention, therapeutic peptide derivatives retain some or all of the therapeutic activities of the therapeutic peptides.
[0041] As used herein, the terms "amino terminus protecting group" or "N-terminal protecting group," "carboxy terminus protecting group" or "C-terminal protecting group;" or "side chain protecting group" refer to any chemical moiety capable of addition to and optionally removal from a functional group on a peptide (e.g., the TV-terminus, the
C-terminus, or a functional group associated with the side chain of an amino acid located within the peptide) to allow for chemical manipulation of the peptide.
[0042] "PEG," "polyethylene glycol" and "poly(ethylene glycol)" as used herein, are meant to encompass any water-soluble poly(ethylene oxide). Typically, PEGs for use in accordance with the invention comprise the following structure "-0(CH2CH20)m-" where (m) is 2 to 4000. As used herein, PEG also includes "-CH2CH2-0(CH2CH20)m-CH2CH2-" and "-(CH2CH20)m-," depending upon whether or not the terminal oxygens have been displaced. When the PEG further comprises a spacer moiety (to be described in greater detail below), the atoms comprising the spacer moiety, when covalently attached to a water-soluble polymer segment, do not result in the formation of an oxygen-oxygen bond (i.e., an "-0-0-" or peroxide linkage). Throughout the specification and claims, it should be remembered that the term "PEG" includes structures having various tenninal or "end capping" groups and so forth. The term "PEG" also means a polymer that contains a majority, that is to say, greater than 50%, of -CH2CH20- monomelic subunits. With respect to specific forms, the PEG can take any number of a variety of molecular weights, as well as structures or geometries such as
"branched," "linear," "forked," "multifunctional," and the like, to be described in greater detail below.
[0043] The terms "end-capped" or "terminally capped" are interchangeably used herein to refer to a terminal or endpoint of a polymer having an end-capping moiety.
Typically, although not necessarily, the end-capping moiety comprises a hydroxy or Ci-20 alkoxy group. Thus, examples of end-capping moieties include alkoxy (e.g., methoxy, ethoxy and benzyloxy), as well as aryl, heteroaryl, cyclo, heterocyclo, and the like. In addition, saturated, unsaturated, substituted and unsubstituted forms of each of the foregoing are envisioned. Moreover, the end-capping group can also be a silane. The end-capping group can also advantageously comprise a detectable label. When the polymer has an end- capping group comprising a detectable label, the amount or location of the polymer and/or the moiety (e.g., active agent) of interest to which the polymer is coupled can be determined by using a suitable detector. Such labels include, without limitation, fluorescers,
chemiluminescers, moieties used in enzyme labeling, colorimetric (e.g., dyes), metal ions, radioactive moieties, and the like. Suitable detectors include photometers, films,
spectrometers, and the like.
[0044] "Non-naturally occurring" with respect to a polymer or water-soluble polymer means a polymer that in its entirety is not found in nature. A non-naturally occurring polymer or water-soluble polymer may, however, contain one or more subunits or portions of a subunit that are naturally occurring, so long as the overall polymer structure is not found in nature.
[0045] The term "water-soluble polymer" is any polymer that is soluble in water at room temperature. Typically, a water-soluble polymer will transmit at least about 75%, more preferably at least about 95% of light, transmitted by the same solution after filtering. On a weight basis, a water-soluble polymer will preferably be at least about 35% (by weight) soluble in water, more preferably at least about 50% (by weight) soluble in water, still more preferably about 70% (by weight) soluble in water, and still more preferably about 85% (by weight) soluble in water. It is still more preferred, however, that the water-soluble polymer is about 95% (by weight) soluble in water and most preferred that the water-soluble polymer is completely soluble in water.
[0046] Molecular weight in the context of a water-soluble polymer of the invention, such as PEG, can be expressed as either a number average molecular weight or a weight average molecular weight. Unless otherwise indicated, all references to molecular weight
herein refer to the weight average molecular weight. Both molecular weight determinations, number average and weight average, can be measured using gel permeation chromatography or other liquid chromatography techniques. Other methods for measuring molecular weight values can also be used, such as the use of end-group analysis or the measurement of colligative properties (e.g., freezing-point depression, boiling-point elevation, or osmotic pressure) to determine number average molecular weight or the use of light scattering techniques, ultracentrifugation or viscometry to determine weight average molecular weight. The polymers of the invention are typically polydisperse (i.e., number average molecular weight and weight average molecular weight of the polymers are not equal), possessing low polydispersity values of preferably less than about 1.2, more preferably less than about 1.15, still more preferably less than about 1.10, yet still more preferably less than about 1.05, and most preferably less than about 1.03.
O II
[0047] As used herein, the term "carboxylic acid" is a moiety having a -C-OH functional group [also represented as a "-COOH" or -C(0)OH], as well as moieties that are derivatives of a carboxylic acid, such derivatives including, for example, protected carboxylic acids. Thus, unless the context clearly dictates otherwise, the term carboxylic acid includes not only the acid form, but corresponding esters and protected forms as well. With regard to protecting groups suited for a carboxylic acid and any other functional group described herein, reference is made to Greene et al, "PROTECTIVE GROUPS IN ORGANIC SYNTHESIS" 3rd Edition, John Wiley and Sons, Inc., New York, 1999.
[0048] The terms "reactive" and "activated" when used in conjunction with a particular functional group, refer to a reactive functional group that reacts readily with an electrophile or a nucleophile on another molecule. This is in contrast to those groups that require strong catalysts or highly impractical reaction conditions in order to react (i.e., a "nonreactive" or "inert" group).
[0049] The terms "protected," "protecting group," and "protective group" refer to the presence of a moiety (i.e., the protecting group) that prevents or blocks reaction of a particular chemically reactive functional group in a molecule under certain reaction conditions. The protecting group will vary depending upon the type of chemically reactive functional group being protected as well as the reaction conditions to be employed and the presence of additional reactive or protecting groups in the molecule, if any. Protecting groups known in the art can be found in Greene et ah, supra.
[0050] As used herein, the term "functional group" or any synonym thereof i s meant to encompass protected forms thereof.
[0051] The terms "spacer" or "spacer moiety" are used herein to refer to an atom or a collection of atoms optionally appearing between one moiety and another. The spacer moieties may be hydrolytically stable or may include one or more physiologically hydrolyzable or enzymatically releasable linkages.
[0052] An "organic radical" as used herein includes, for example, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, aryl and substituted aryl.
[0053] "Alkyl" refers to a hydrocarbon chain, typically ranging from about 1 to 20 atoms in length. Such hydrocarbon chains are preferably but not necessarily saturated and may be branched or straight chain, although typically straight chain is preferred. Exemplary alkyl groups include methyl, ethyl, propyl, butyl, pentyl, 1-methylbutyl, 1-ethylpropyl, 3- methylpentyl, and the like. As used herein, "alkyl" includes cycloalkyl when three or more carbon atoms are referenced and lower alkyl.
[0054] "Lower alkyl" refers to an alkyl group containing from 1 to 6 carbon atoms, and may be straight chain or branched, as exemplified by methyl, ethyl, n-butyl, z'so-butyl, and tert-butyl.
[0055] "Cycloalkyl" refers to a saturated or unsaturated cyclic hydrocarbon chain, including bridged, fused, or spiro cyclic compounds, preferably made up of 3 to about 12 carbon atoms, more preferably 3 to about 8 carbon atoms.
[0056] "Non-interfering substituents" are those groups that, when present in a molecule, are typically non-reactive with other functional groups contained within the molecule.
[0057] The term "substituted" as in, for example, "substituted alkyl," refers to a moiety (e.g., an alkyl group) substituted with one or more non-interfering substituents, such as, but not limited to: C3-C8 cycloalkyl, e.g., cyclopropyl, cyclobutyl, and the like; halo, e.g., fluoro, chloro, bromo, and iodo; cyano; alkoxy, lower phenyl; substituted phenyl; and the like, for one or more hydrogen atoms. "Substituted aryl" is aryl having one or more non-interfering groups as a substituent. For substitutions on a phenyl ring, the substituents may be in any orientation (i.e., ortho, meta, or para). "Substituted ammonium" is ammonium having one or more non-interfering groups (e.g., an organic radical) as a substituent.
[0058] "Alkoxy" refers to an -O-R group, wherein R is alkyl or substituted alkyl, preferably C1-C20 alkyl (e.g., methoxy, ethoxy, propyloxy, benzyl, etc.), more preferably Ci- C7 alkyl.
[0059] As used herein, "alkenyl" refers to a branched or unbranched hydrocarbon group of 2 to 15 atoms in length, containing at least one double bond. Exemplary alkenyl include (without limitation) ethenyl, «-propenyl, isopropenyl, n-butenyl, z'so-butenyl, octenyl, decenyl, tetradecenyl, and the like.
[0060] The term "alkynyl" as used herein refers to a branched or unbranched hydrocarbon group of 2 to 15 atoms in length, containing at least one triple bond. Exemplary alkynyl include (without limitation) ethynyl, n-butynyl, z'so-pentynyl, octynyl, decynyl, and so forth.
[0061] "Aryl" means one or more aromatic rings, each of 5 or 6 core carbon atoms.
Aryl includes multiple aryl rings that may be fused, as in naphthyl, or unfused, as in biphcnyl. Aryl rings may also be fused or unfused with one or more cyclic hydrocarbon, heteroaryl, or heterocyclic rings. As used herein, "aryl" includes heteroaryl. An aromatic-containing moiety (e.g., Ar1, Ar2, and so forth), means a structure containing aryl.
[0062] "Heteroaryl" is an aryl group containing from one to four heteroatoms, preferably N, O, or S, or a combination thereof. Heteroaryl rings may also be fused with one or more cyclic hydrocarbon, heterocyclic, aryl, or heteroaryl rings.
[0063] "Heterocycle" or "heterocyclic" means one or more rings of 5-12 atoms, preferably 5-7 atoms, with or without unsaturation or aromatic character and having at least one ring atom which is not a carbon. Preferred heteroatoms include sulfur, oxygen, and nitrogen.
[0064] "Substituted heteroaryl" is heteroaryl having one or more non-interfering , groups as substituents.
[0065] "Substituted heterocycle" is a heterocycle having one or more side chains formed from non-interfering substituents.
[0066] "Electrophile" refers to an ion or atom or collection of atoms, which may be ionic, having an electrophilic center, i.e., a center that is electron seeking, capable of reacting with a nucleophile.
[0067] "Nucleophile" refers to an ion or atom or collection of atoms that may be ionic having a nucleophilic center, i.e., a center that is seeking an electrophilic center or with an electrophile.
[0068] A "physiologically cleavable" as well as a "hydrolyzable" bond is a relatively weak bond that reacts with water (i.e., is hydrolyzed) under physiological conditions. The tendency of a bond to hydrolyze in water will depend not only on the general type of linkage connecting two central atoms but also on the substituents attached to these central atoms. Exemplary hydrolyzable bonds include, but are not limited to, carboxylate ester, phosphate ester, anhydride, acetal, ketal, acyloxyalkyl ether, imine, and ortho esters.
[0069] A "releasable linkage" includes, but is not limited to, a physiologically cleavable bond, a hydrolyzable bond, and an enzymatically degradable linkage. Thus, a "releasable linkage" is a linkage that may undergo either hydrolysis or cleavage by some other mechanism (e.g., enzyme-catalyzed, acid-catalyzed, base-catalyzed, and so forth) under physiological conditions. For example, a "releaseable linkage" can involve an elimination reaction that has a base abstraction of a proton, (e.g., an ionizable hydrogen atom, Ha), as the driving force. For purposes herein, a "releaseable linkage" is synonymous with a "degradable linkage."
[0070] An "enzymatically releasable linkage" means a linkage that is subject to degradation by one or more enzymes.
[0071] A "hydrolytically stable" linkage or bond refers to a chemical bond, typically a covalent bond, which is substantially stable in water, that is to say, does not undergo hydrolysis under physiological conditions to any appreciable extent over an extended period of time. Examples of hydrolytically stable linkages include but are not limited to the following: carbon-carbon bonds (e.g., in aliphatic chains), ethers, amides, and the like.
Generally, a hydrolytically stable linkage is one that exhibits a rate of hydrolysis of less than about 1 -2% per day under physiological conditions. Hydrolysis rates of representative chemical bonds can be found in most standard chemistry textbooks. It must be pointed out that some linkages can be hydrolytically stable or hydrolyzable, depending upon (for example) adjacent and neighboring atoms and ambient conditions. One of ordinary skill in the art can determine whether a given linkage or bond is hydrolytically stable or hydrolyzable in a given context by, for example, placing a linkage-containing molecule of interest under conditions of interest and testing for evidence of hydrolysis (e.g., the presence and amount of two molecules resulting from the cleavage of a single molecule). Other approaches known to
those of ordinary skill in the art for determining whether a given linkage or bond is hydrolytically stable or hydrolyzable can also be used.
[0072] The terms "active agent," "biologically active agent" and "pharmacologically active agent" are used interchangeably herein and are defined to include any agent, drug, compound, composition of matter or mixture that provides some pharmacologic, often beneficial, effect that can be demonstrated in vivo or in vitro. This includes food
supplements, nutrients, nutriceuticals, drugs, proteins, vaccines, antibodies, vitamins, and other beneficial agents. As used herein, these terms further include any physiologically or pharmacologically active substance that produces a localized or systemic effect in a patient.
[0073] "Pharmaceutically acceptable excipient" or "pharmaceutically acceptable carrier" refers to an excipient that can be included in the compositions of the invention and that causes no significant adverse toxicological effects to the patient.
[0074] "Pharmacologically effective amount," "physiologically effective amount," and "therapeutically effective amount" are used interchangeably herein to mean the amount of a polymer-active agent conjugate ~ typically present in a pharmaceutical preparation— that is needed to provide a desired level of active agent and/or conjugate in the bloodstream or in a target tissue. The exact amount will depend upon numerous factors, e.g., the particular active agent, the components and physical characteristics of the pharmaceutical preparation, intended patient population, patient considerations, and the like, and can readily be determined by one of ordinary skill in the art, based upon the information provided herein and available in the relevant literature.
[0075] "Multifunctional" in the context of a polymer means a polymer having 3 or more functional groups contained therein, where the functional groups may be the same or different. Multifunctional polymers will typically contain from about 3-100 functional groups, or from 3- 50 functional groups, or from 3-25 functional groups, or from 3-15 functional groups, or from 3 to 10 functional groups, or will contain 3, 4, 5, 6, 7, 8, 9 or 10 functional groups within the polymer. A "difunctional" polymer means a polymer having two functional groups contained therein, either the same (i.e., homodifunctional) or different (i.e., heterodifunctional).
[0076] "Branched," in reference to the geometry or overall structure of a polymer, refers to polymer having 2 or more polymer "arms." A branched polymer may possess 2 polymer arms, 3 polymer arms, 4 polymer arms, 6 polymer arms, 8 polymer arms or more. One particular type of highly branched polymer is a dendritic polymer or dendrimer, which,
for the purposes of the invention, is considered to possess a structure distinct from that of a branched polymer.
[0077] A "dendrimer" or dendritic polymer is a globular, size monodisperse polymer in which all bonds emerge radially from a central focal point or core with a regular branching pattern and with repeat units that each contribute a branch point. Dendrimers exhibit certain dendritic state properties such as core encapsulation, making them unique from other types of polymers.
[0078] A basic or acidic reactant described herein includes neutral, charged, and any corresponding salt forms thereof.
[0079] The term "patient," refers to a living organism suffering from or prone to a condition that can be prevented or treated by administration of a conjugate as provided herein, and includes both humans and animals.
[0080] As used herein, "drug release rate" means a rate (stated as a half-life) in which half of the total amount of polymer- active agent conjugates in a system will cleave into the active agent and a polymeric residue.
[0081] "Optional" and "optionally" mean that the subsequently described
circumstance may or may not occur, so that the description includes instances where the circumstance occurs and instances where it does not.
[0082] As used herein, the "halo" designator (e.g., fluoro, chloro, iodo, bromo, and so forth) is generally used when the halogen is attached to a molecule, while the suffix "ide" (e.g., fluoride, chloride, iodide, bromide, and so forth) is used when the halogen exists in its independent ionic form (e.g., such as when a leaving group leaves a molecule).
[0083] In the context of the present discussion, it should be recognized that the definition of a variable provided with respect to one structure or formula is applicable to the same variable repeated in a different structure, unless the context dictates otherwise.
[0084] As previously stated, the present invention comprises (among other things) conjugates having a releasable linkage.
[0085] Before describing exemplary conjugates of the invention, embodiments of a water-soluble polymer and a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage, will be discussed.
[0086] With respect to a given water-soluble polymer, each water-soluble polymer
(e.g., POLY, POLY and POLY ) can comprise any polymer so long as the polymer is water- soluble and non-peptidic. Although preferably a poly(ethylene glycol), a water-soluble polymer for use herein can be, for example, other water-soluble polymers such as other poly(alkylene glycols) [also referred to as "poly(alkyleneoxides)"], such as poly(propylene glycol) ("PPG"), copolymers of ethylene glycol and propylene glycol and the like, poly(olefinic alcohol), poly(vinylpyrrolidone), poly(hydroxyalkylmethacrylamide), poly(hydroxyalkylmefhacrylate), poly(saccharides), poly(a-hydroxy acid), poly( vinyl alcohol), polyphosphazene, polyoxazoline, poly(N-acryloylmorpholine), such as described in U.S. Patent No. 5,629,384. The water soluble polymer can be a homopolymer, copolymer, terpolymer, nonrandom block polymer, and random block polymer of any of the foregoing. In addition, a water-soluble polymer can be linear, but can also be in other forms (e.g., branched, forked, and the like) as will be described in further detail below. In the context of being present within an overall structure, a water-soluble polymer has from 1 to about 300 termini.
[0087] In instances where the polymeric reagent comprises two or more water-soluble polymers, each water-soluble polymer in the overall structure can be the same or different. It is preferred, however, that all water-soluble polymers in the overall structure are of the same type. For example, it is preferred that all water-soluble polymers within a given structure are poly(ethylene glycol) polymers.
[0088] Although the weight-average molecular weight of any individual
water-soluble polymer can vary, the weight average molecular weight of any given water-soluble polymer will typically be in the following range: 100 Daltons to about 150,000 Daltons. Exemplary ranges, however, include weight-average molecular weights in the following ranges: in the range of from about 880 Daltons to about 5,000 Daltons; in the range of greater than 5,000 Daltons to about 100,000 Daltons; in the range of from about 6,000 Daltons to about 90,000 Daltons; in the range of from about 10,000 Daltons to about 85,000 Daltons; in the range of greater than 10,000 Daltons to about 85,000 Daltons; in the range of from about 20,000 Daltons to about 85,000 Daltons; in the range of from about 53,000 Daltons to about 85,000 Daltons; in the range of from about 25,000 Daltons to about 120,000 Daltons; in the range of from about 29,000 Daltons to about 120,000 Daltons; in the range of from about 35,000 Daltons to about 120,000 Daltons; in the range of about 880 Daltons to about 60,000 Daltons; in the range of about 440 Daltons to about 40,000 Daltons; in the range of about 440 Daltons to about 30,000 Daltons; and in the range of from about 40,000 Daltons
to about 120,000 Daltons. For any given water-soluble polymer, PEGs having a molecular weight in one or more of these ranges are preferred.
[0089] Exemplary weight-average molecular weights for the water-soluble polymer include about 100 Daltons, about 200 Daltons, about 300 Daltons, about 400 Daltons, about 440 Daltons, about 500 Daltons, about 600 Daltons, about 700 Daltons, about 750 Daltons, about 800 Daltons, about 900 Daltons, about 1 ,000 Daltons, about 1,500 Daltons, about 2,000 Daltons, about 2,200 Daltons, about 2,500 Daltons, about 3,000 Daltons, about 4,000 Daltons, about 4,400 Daltons, about 4,500 Daltons, about 5,000 Daltons, about 5,500 Daltons, about 6,000 Daltons, about 7,000 Daltons, about 7,500 Daltons, about 8,000 Daltons, about 9,000 Daltons, about 10,000 Daltons, about 11 ,000 Daltons, about 12,000 Daltons, about 13,000 Daltons, about 14,000 Daltons, about 15,000 Daltons, about 16,000 Daltons, about 17,000 Daltons, about 18,000 Daltons, about 19,000 Daltons, about 20,000 Daltons, about 22,500 Daltons, about 25,000 Daltons, about 30,000 Daltons, about 35,000 Daltons, about 40,000 Daltons, about 45,000 Daltons, about 50,000 Daltons, about 55,00 Daltons, about 60,000 Daltons, about 65,000 Daltons, about 70,000 Daltons, and about 75,000 Daltons. Branched versions of the water-soluble polymer (e.g., a branched 40,000 Dalton water-soluble polymer comprised of two 20,000 Dalton polymers) having a total weight average molecular weight of any of the foregoing can also be used.
[0090] The polymeric reagent used to prepare the conjugate will comprise at least one water-soluble polymer having a total size in the range suited for the desired rate of release of the conjugate formed therefrom. For example, a conjugate having a relatively long release rate can be prepared from a polymeric reagent having a size suited for (a) extended circulation prior to release of the active agent from the conjugate, and (b) moderately rapid in vivo clearance of the species liberated from the conjugate upon release from the conjugate. Likewise, when the conjugate has a relatively fast release rate, then the polymeric reagent would typically have a lower molecular weight.
[0091] When a PEG is used as the water-soluble polymer(s) in the polymeric reagent, the PEG typically comprises a number of (OCH2CH2) monomers [or (CH2CH20) monomers, depending on how the PEG is defined]. As used throughout the description, the number of repeating units is identified by the subscript "n" in "(OCH2CH2)n." Thus, the value of (n) typically falls within one or more of the following ranges: from 2 to about 3400, from about 4 to about 1500, from about 100 to about 2300, from about 100 to about 2270, from about 136 to about 2050, from about 225 to about 1930, from about 450 to about 1930, from about 1200
to about 1930, from about 568 to about 2727, from about 660 to about 2730, from about 795 to about 2730, from about 795 to about 2730, from about 909 to about 2730, and from about 1 ,200 to about 1 ,900. For any given polymer in which the molecular weight is known, it is possible to determine the number of repeating units (i.e., "n") by dividing the total weight-average molecular weight of the polymer by the molecular weight of the repeating monomer.
[0092] Each water-soluble polymer is typically biocompatible and non-immunogenic.
With respect to biocompatibility, a substance is considered biocompatible if the beneficial effects associated with use of the substance alone or with another substance (e.g., an active agent) in connection with living tissues (e.g., administration to a patient) outweighs any deleterious effects as evaluated by a clinician, e.g., a physician. With respect to
non-immunogenicity, a substance is considered non-immunogenic if use of the substance alone or with another substance in connection with living tissues does not produce an immune response (e.g., the formation of antibodies) or, if an immune response is produced, that such a response is not deemed clinically significant or important as evaluated by a clinician. It is particularly preferred that the water-soluble polymers described herein as well as conjugates of active agents and the polymers are biocompatible and non-immunogenic.
[0093] In one form useful, free or nonbound PEG is a linear polymer terminated at each end with hydroxyl groups:
HO-CH2CH20-(CH2CH20)m-CH2CH2-OH
wherein (m1) typically ranges from zero to about 4,000, preferably from about 20 to about 1 ,000.
[0094] The above polymer, alpha-, omega-dihydroxylpoly(ethylene glycol), can be represented in brief form as HO-PEG-OH where it is understood that the -PEG- symbol can represent the following structural unit:
-CH2CH20-(CH2CH20)m.-CH2CH2- where (m') is as defined as above.
[0095] Another type of free or nonbound PEG useful in the present invention is methoxy-PEG-OH, or mPEG in brief, in which one terminus is the relatively inert methoxy group, while the other terminus is a hydroxyl group. The structure of mPEG is given below.
CH30-CH2CH20-(CH2CH20)m-CH2CH2-
where (m') is as described above.
[0096] Multi-armed or branched PEG molecules, such as those described in U.S.
Patent No. 5,932,462, can also be used as the PEG polymer. For example, PEG can have the structure:
P°iya— P
R"— C
I
polyb— Q wherein:
polya and polyb are PEG backbones (either the same or different), such as methoxy poly(ethylene glycol);
R" is a nonreactive moiety, such as H, methyl or a PEG backbone; and
P and Q are nonreactive linkages. In a preferred embodiment, the branched PEG polymer is methoxy poly( ethylene glycol) disubstituted lysine.
[0097] In addition, the PEG can comprise a forked PEG. An example of a free or nonbound forked PEG is represented by the following formula:
Z
/
PEG-X-C-H
\
Z wherein: X is a spacer moiety and each Z is an activated terminal group linked to CH by a chain of atoms of defined length. The chain of atoms linking the Z functional groups to the branching carbon atom serve as a tethering group and may comprise, for example, alkyl chains, ether chains, ester chains, amide chains and combinations thereof. U.S. Patent No. 6,362,254, discloses various forked PEG structures capable of use in the present invention.
[0098] The PEG polymer may comprise a pendant PEG molecule having reactive groups, such as carboxyl, covalently attached along the length of the PEG rather than at the end of the PEG chain. The pendant reactive groups can be attached to the PEG directly or through a spacer moiety, such as an alkylene group.
[0099] In addition to the above-described forms of PEG, each water-soluble polymer in the polymeric reagent can also be prepared with one or more weak or releasable linkages in the polymer, including any of the above described polymers. For example, PEG can be
prepared with ester linkages in the polymer that are subject to hydrolysis. As shown below, this hydrolysis results in cleavage of the polymer into fragments of lower molecular weight:
-PEG-C02-PEG- + H20 ► -PEG-C02H + HO-PEG-
[0100] Other hydrolytically releasable linkages, useful as a releasable linkage within a polymer backbone, include carbonate linkages; imine linkages resulting, for example, from reaction of an amine and an aldehyde (see, e.g., Ouchi et al. (1997) Polymer Preprints 38(l):582-3); phosphate ester linkages formed, for example, by reacting an alcohol with a phosphate group; hydrazone linkages which are typically formed by reaction of a hydrazide and an aldehyde; acetal linkages that are typically formed by reaction between an aldehyde and an alcohol; ortho ester linkages that are, for example, formed by reaction between a formate and an alcohol; amide linkages formed by an amine group, e.g., at an end of a polymer such as PEG, and a carboxyl group of another PEG chain; urethane linkages formed from reaction of, e.g., a PEG with a terminal isocyanate group and a PEG alcohol; peptide linkages formed by an amine group, e.g., at an end of a polymer such as PEG, and a carboxyl group of a peptide; and oligonucleotide linkages formed by, for example, a phosphoramidite group, e.g., at the end of a polymer, and a 5' hydroxyl group of an oligonucleotide.
[0101] It is understood by those of ordinary skill in the art that the term poly( ethylene glycol) or PEG represents or includes all the above forms of PEG.
[0102] Those of ordinary skill in the art will recognize that the foregoing discussion concerning substantially water-soluble polymers is by no means exhaustive and is merely illustrative, and that all polymeric materials having the qualities described above are contemplated. As used herein, the term "water-soluble polymer" refers both to a molecule as well as the residue of water-soluble polymer that has been attached to another moiety. The following description of a water-soluble polymer are applicable not only to the polymeric reagent, but to the corresponding conjugates formed using the described polymeric reagents.
[0103] The functional group of the polymeric reagents used to form the conjugates described herein is a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage. The invention is not limited with respect to the specific functional group so long as the functional group is capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage. Exemplary functional groups capable of reacting with an amino group of an active agent include those functional groups selected from the group consisting of active
carbonates such as N-succinimidyl, 1-benzotriazolyl, imidazole, carbonate halides (such as carbonate chloride and carbonate bromide), phenolates (such as jo-nitrophenolate) and so forth. Also, as a special case, if the active agent is available with the active amine group converted into an isocyanate or isothiocyanate group, then the functional group of the polymeric reagent can be hydroxyl as the reaction of these components provides a releasable carbamate linkage.
[0104] Exemplary polymeric reagents will now be discussed in further detail. It must be remembered that while stereochemistry is not specifically shown in any formulae or structures (whether for a polymeric reagent, conjugate, or any other formula or structure), the provided formulae and structures contemplate both enantiomers, as well as compositions comprising mixtures of each enantiomer in equal amounts (i.e., a racemic mixture) and unequal amounts.
f0105| An exemplar polymeric reagent has the following structure:
wherein:
POLY is a first water-soluble polymer;
POLY2 is a second water-soluble polymer;
X1 is a first spacer moiety;
X2 is a second spacer moiety;
Ha is an ionizable hydrogen atom;
R1 is H or an organic radical;
R2 is H or an organic radical;
(a) is either zero or one;
(b) is either zero or one;
Rel, when present, is a first electron altering group;
Re2, when present, is a second electron altering group; and
(FG) is a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage.
[0106] Exemplary polymeric reagents fall within the following formulae:
wherein, in each instance: (FG) is a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage; R1 is H or an organic radical; and R2 is H or an organic radical.
[0107] Still other exemplary polymeric reagents have the structure:
wherein each of POLY1, POLY2, X1, X2, R1, R2, Ha and (FG) is as previously defined, and Rel is a first electron altering group; and Re2 is a second electron altering group.
[0108] Still other exemplary polymeric reagents fall within the following structures
wherein, for each structure and in each instance, (n) is independently an integer from 4 to 1500.
[0109] The polymeric reagents can be prepared in any number of ways.
Consequently, synthesis of the polymeric reagents is not limited to the specific technique or approach used in their preparation. Exemplary approaches for preparing the polymeric reagents are described in the literature.
[0110] The above-described polymeric reagents are useful for conjugation to biologically active agents. For example, an amino group (e.g., primary amine) on an active agent will react with the functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage.
[0111] Exemplary conjugates include those of the following formulae:
POLY1 is a first water-soluble polymer;
POLY2 is a second water-soluble polymer;
X1 is a first spacer moiety;
X2 is a second spacer moiety;
Ha is an ionizable hydrogen atom;
R1 is H or an organic radical;
R2 is H or an organic radical;
(a) is either zero or one;
(b) is either zero or one;
Rel, when present, is a first electron altering group; Re2, when present, is a second electron altering group; Y1 is O or S;
Y2 is O or S; and
(GLP-1) is a residue of GLP-1 moiety.
[0112]
C H30 ·( CH 2CH 20)n-C H2C H2-0
wherein, for each structure and in each instance, (n) is independently an integer from 4 to 1500, and (GLP-1) is a residue of a GLP-1 moiety.
[0113] The biologically active agent to which a polymeric reagent as described herein can be conjugated, is an amine-containing biologically active agent. Typically, the biologically active agent will be a macromolecule, such as a polypeptide, having a molecular weight greater than about 3,500 Daltons. Pharmacologically active polypeptides represent a preferred type of biologically active agent. It should be understood that for purposes of the present discussion, the term "polypeptide" will be generic for oligopeptides and proteins. With regard to polypeptides, the amine to which the polymeric reagent couples to can be on the N-terminus or an amine-containing side chain of an amino acid (such as lysine) within the polypeptide.
[0114] The invention also provides for a method of preparing a conjugate comprising the step of contacting a polymeric reagent with a biologically active agent under conditions suitable to form a covalent attachment between the polymer and the biologically active agent. Typically, the polymer is added to the active agent or surface at an equimolar amount (with
respect to the desired number of groups suitable for reaction with the reactive group) or at a molar excess. For example, the polymeric reagent can be added to the target active agent at a molar ratio of about 1 : 1 (polymeric reagent: active agent), 1.5:1, 2:1, 3:1, 4: 1 , 5: 1 , 6: 1 , 8: 1, or 10: 1. The conjugation reaction is allowed to proceed until substantially no further conjugation occurs, which can generally be determined by monitoring the progress of the reaction over time. Progress of the reaction can be monitored by withdrawing aliquots from the reaction mixture at various time points and analyzing the reaction mixture by SDS-PAGE or MALDI-TOF mass spectrometry or any other suitable analytical method. Once a plateau is reached with respect to the amount of conjugate formed or the amount of unconjugated polymer remaining, the reaction is assumed to be complete. Typically, the conjugation reaction takes anywhere from minutes to several hours (e.g., from 5 minutes to 24 hours or more). The resulting product mixture is preferably, but not necessarily, purified to separate out excess reagents, unconjugated reactants (e.g., active agent) undesired multi-conjugated species, and free or unreacted polymer. The resulting conjugates can then be further characterized using analytical methods such as MALDI, capillary electrophoresis, gel electrophoresis, and/or chromatography.
[0115] It is possible to characterize the degree of attachment (that is, the number
— often expressed in terms of an average number in the context of a composition of conjugates ~ of polymeric reagents that became attached to the protein) of a conjugate. To determine the average number of water-soluble polymer molecules on protein conjugates, analytical techniques such as SDS-PAGE, SEC, IEC, MALDI-TOF, and so forth can be used. Spectrophotometric detection of the residual primary amine on a protein using TNBSA following water-soluble polymer attachment has led to the qualitative and quantitative estimation of degree of attachment. With respect to GLP-1 -water-soluble polymer conjugates, the degree of attachment was qualitatively described as low, medium, and high based on SDS-PAGE.
[0116] With respect to polymer-active agent conjugates, the conjugates can be purified to obtain/isolate different conjugated species. Alternatively, and more preferably for lower molecular weight (e.g., less than about 20 kiloDaltons, more preferably less than about 10 kiloDaltons) polymers, the product mixture can be purified to obtain the distribution of water-soluble polymer segments per active agent. For example, the product mixture can be purified to obtain an average of anywhere from one to five PEGs per active agent (e.g., polypeptide). The strategy for purification of the final conjugate reaction mixture will depend upon a number of factors, including, for example, the molecular weight of the
polymer employed, the particular active agent, the desired dosing regimen, and the residual activity and in vivo properties of the individual conjugate(s).
[0117] If desired, conjugates having different molecular weights can be isolated using gel filtration chromatography. That is to say, gel filtration chromatography is used to fractionate differently numbered polymer-to-active agent ratios (e.g., 1-mer, 2-mer, 3-mer, and so forth, wherein " 1-mer" indicates 1 polymer to active agent, "2-mer" indicates two polymers to active agent, and so on) on the basis of their differing molecular weights (where the difference corresponds essentially to the average molecular weight of the water-soluble polymer segments). For example, in an exemplary reaction where a 100 kDa protein is randomly conjugated to a polymeric reagent having a molecular weight of about 20 kDa, the resulting reaction mixture will likely contain unmodified protein (MW 100 kDa), mono- PEGylated protein (MW 120 kDa), di-PEGylated protein (MW 140 kDa), and so forth.
While this approach can be used to separate PEG and other polymer conjugates having different molecular weights, this approach is generally ineffective for separating positional isomers having different polymer attachment sites within the protein. For example, gel filtration chromatography can be used to separate from each other mixtures of PEG 1 -mers, 2-mers, 3-mers, and so forth, although each of the recovered PEG-mer compositions may contain PEGs attached to different reactive amino groups (e.g., lysine residues) within the active agent.
[0118] Gel filtration columns suitable for carrying out this type of separation include
Superdex™ and Sephadex™ columns available from Amersham Biosciences (Piscataway, NJ). Selection of a particular column will depend upon the desired fractionation range desired. Elution is generally carried out using a suitable buffer, such as phosphate, acetate, or the like. The collected fractions may be analyzed by a number of different methods, for example, (i) optical density (OD) at 280 ran for protein content, (ii) bovine serum albumin (BSA) protein analysis, (iii) iodine testing for PEG content [Sims et al. (1980) Anal. Biochem, 107:60-63], and (iv) sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS PAGE), followed by staining with barium iodide.
[0119] Separation of positional isomers is carried out by reverse phase
chromatography using a reverse phase-high performance liquid chromatography (RP-HPLC) CI 8 column (Amersham Biosciences or Vydac) or by ion exchange chromatography using an ion exchange column, e.g., a Sepharose™ ion exchange column available from Amersham
Biosciences. Either approach can be used to separate polymer-active agent isomers having the same molecular weight (positional isomers).
[0120] An amine-containing biologically active agent for use in coupling to a polymer as presented herein is a GLP-1 moiety.
[0121] With respect to "GLP-1 " itself (and not in a conjugate form), "GLP-1 " shall be understood to designate the peptide having the truncated "7-36" amino acid sequence (SEQ ID NO: l): NH2-His7-Ala8-Glu9-Gly10-^
Leu^-Glu^-Gly^-Gln^-Ala^-Ala^^
Gly35-Arg36-NH2). GLP-1 (7-37)OH, an exemplary GLP-1 moiety, shall be understood to designate the peptide having the following amino acid sequence (SEQ ID NO:l): NH2-His7-
Gln23-Ala24-Ala25-Lys26-Glu2^^
Other exemplary GLP-1 moieties for use in connection with the present invention include GLP-1 (1-36), GLP-1 analogs (such as those described in WO 91/1 1457), GLP-1 derivatives, GLP-1 moieties described in U.S. Published Patent Application No. 2004/0235710, GLP-1 biologically active fragments, extended GLP-1 (see, for example, WO 03/058203, in particular with respect to the extended glucagon-like peptide- 1 analogs described therein), N-terminal truncated fragments of GLP-1 (such as those described in EP 0 699 686, and exendins (including, for example, exendin-4 and analogs thereof).
[0122] With respect to exendins, exendins are peptides that were first isolated from the salivary secretions of the Gila-monster and the Mexican Beaded Lizard. The exendins have a degree of similarity to several members of the GLP family, with the highest homology, 53%, to GLP-l(7-36)NH2 (Goke, et al, J. Biol. Chem., 268: 19650-55, 1993). ' Particular exendins for use in the present invention include exendin-3 and exendin-4
(synthetic extendin-4 is also known as Exenatide). Exendin-3 (1-39) has the following amino acid sequence (SEQ ID NO:3): His-Ser-Asp-Gly-Thr-Phe-Thr-Ser-Asp-Leu-Ser-Lys-Gln- Met-Glu-Glu-Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-T -Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser- Gly-Ala-Pro-Pro-Pro-Ser-NH2. The amino acid sequence of exendin-4 (1-39) corresponds to (SEQ ID NO:4): His-Gly-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Leu-Ser-Lys-Gln-Met-Glu-Glu- Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-Trp-Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser-Gly-Ala-Pro-Pro- Pro-Ser wherein the C-terminus serine is amidated.
[0123] Regardless of the GLP-1 moiety used in connection with the present invention, the GLP-1 moiety may be obtained from either non-recombinant methods or from
recombinant methods, and the invention is not limited in this regard. Moreover, several GLP-1 moieties are commercially available, e.g., hGLP-1 , rExtendin-4, and rHuGLP-1 are available from ProSpecTany Techno Gene LTD (Rehovot, Israel); and (Ser8)GLP-l(7- 36)amide, hGLP-lamide, and hGLP-1 (7-36)Lys(biotin)amide are available from American Peptide Co., Sunnyvale, CA. Methods for preparing GLP-1 moieties are well-known, and are described, e.g., in U.S. Patent Nos. 5,118,666; 5,120,712; and 5,523,549.
[0124] Briefly, however, GLP-1 moieties can be prepared using standard methods of solution or solid phase peptide synthesis such as those described in Dugas H., Penny, C, Bioorganic Chemistry, Springer Verlag, New York, p. 54-92 (1981); Merrifield (1962) Chem Soc. 85:2149, and Stewart and Young, Solid Phase Peptide Synthesis, Freeman, San
Francisco, p. 24-66 (1969). Peptide synthesizers are available from, e.g., Applied
Biosystems, Foster City, CA. Solid phase synthesizers are typically used according to manufacturers' instructions for blocking interfering groups, protecting certain amino acids, coupling, decoupling, and capping unreacted amino acids. BOC-amino acids and other reagents are commercially available from Applied Biosystems, Foster City CA. Sequential BOC (tert-butoxycarbonyl or "BOC") chemistry using double coupling protocols are applied to the starting p-methyl benzhydryl amine resins for the production of C-terminal
carboxamides. For the production of C-terminal acids, the corresponding PAM
(oxymethylphenylacetamidomethyl or "PAM") resin is used. Asn, Gin, an Arg are coupled using preformed hydroxybenzotriazole esters. Suitable side chain protecting groups include: Arg (tosyl), Asp (cyclohexyl), Glu (cyclohexyl), Ser (benzyl), Thr (benzyl), and Tyr (4- bromocarbobenzoxy). BOC deprotection may be carried out with trifluoroacetic acid in methylene chloride. Following synthesis, the resulting peptide may be deprotected and cleaved from the resin using, e.g., anhydrous HF containing 10% meta-cresol.
[0125] Exemplary recombinant methods used to prepare a GLP-1 moiety (whether a human GLP-1 or a different peptide having GLP-1 activity) include the following, among others as will be apparent to one skilled in the art. Typically, a GLP-1 moiety as described herein is prepared by constructing the nucleic acid encoding the desired polypeptide or fragment, cloning the nucleic acid into an expression vector, transforming a host cell {e.g. , plant, bacteria such as Escherichia coli, yeast such as Saccharomyces cerevisiae, or mammalian cell such as Chinese hamster ovary cell or baby hamster kidney cell), and expressing the nucleic acid to produce the desired polypeptide or fragment. The expression can occur via exogenous expression (when the host cell naturally contains the desired genetic coding) or via endogenous expression. Methods for producing and expressing recombinant
polypeptides in vitro and in prokaryotic and eukaryotic host cells are known to those of ordinary skill in the art. See, for example, U.S. Patent No. 4,868,122. Often, GLP-1 is typlically expressed in E. coli (since is does not require glycosylation for activity).
[0126] Yet further methods for making recombinant GLP-1 moieties are described in
WO 03/099847 and U.S. Patent No. 7,498,148.
[0127] To facilitate identification and purification of the recombinant polypeptide, nucleic acid sequences that encode an epitope tag or other affinity binding sequence can be inserted or added in-frame with the coding sequence, thereby producing a fusion protein comprised of the desired peptide and a polypeptide suited for binding. Fusion proteins can be identified and purified by first running a mixture containing the fusion protein through an affinity column bearing binding moieties (e.g., antibodies) directed against the epitope tag or other binding sequence in the fusion proteins, thereby binding the fusion protein within the column. Thereafter, the fusion protein can be recovered by washing the column with the appropriate solution (e.g. , acid) to release the bound fusion protein. These and other methods for identifying and purifying recombinant peptides are known to those of ordinary skill in the art. In one or more embodiments of the present invention, the GLP-1 moiety is not in the form of a fusion protein. See, for example, Dillon et al. (1993) Endocrinology 133:1907- 1910.
[0128] For any given moiety, it is possible to determine whether that moiety possesses GLP-1 activity. Various assays may be used to assess bioactivity, including in- vitro and in-vivo assays that measure GLP-1 receptor binding activity or receptor activation. See, for example, EP 0 619 322 and U.S. Patent No. 5,120,712 for descriptions of assessing GLP-1 activity. A receptor-signaling assay may also be used to assess GLP-1 activity, such as described in Zlokarnik et al. (1998) Science 279:84-88.
[0129] Other methods known to those of ordinary skill in the art can also be used to determine whether a given moiety has GLP-1 activity. Such methods are useful for determining the GLP-1 activity of both the moiety itself (and therefore can be used as a "GLP-1 moiety"), as well as that of the corresponding polymer-moiety conjugate. For example, one can determine whether a given moiety is an agonist of the human GLP-1 receptor by assessing whether that moiety stimulates the formation of cAMP in a suitable medium containing the human GLP-1 receptor. The potency of such moiety is determined by calculating the EC50 value from a dose response curve. As an example, BHK cells (baby hamster kidney cells) expressing the cloned human GLP-1 receptor can be grown in DMEM
media containing penicillin, streptomycin, calf serum, and Geneticin. The cells are then washed in phosphate buffered saline and harvested. Plasma membranes are then prepared from the cells by homogenization, and the homogenate is then centrifuged to produce a pellet. The resulting pellet is suspended by homogenization in a suitable buffer, centrifuged, and then washed. The cAMP receptor assay is then carried out by measuring cyclic AMP
(cAMP) in response to the test insulinotropic moiety. cAMP can be quantified using the AlphaScreen™ cAMP Kit (Perkin Elmer). Incubations are typically carried out in microtiter plates in buffer, with addition of, e.g., ATP, GTP, IB MX (3-isobutyl-l-methylxanthine, Tween-20, BSA, acceptor beads, and donor beads incubated with biotinylated cAMP.
Counting may be carried out, e.g., using the Fusion™ instrument (Perkin Elmer).
Concentration-response curves are then plotted for the individual insulinotropic moieties under evaluation, and their EC50 values determined.
[0130] Nonlimiting examples of GLP-1 moieties for use in connection with the present invention include any of SEQ ID NO: 1 , SEQ ID NO: 2 and SEQ ID NO: 3; and SEQ ID NO:4; truncated versions thereof; hybrid variants, and peptide mimetics having GLP-1 activity. Biologically active fragments, deletion variants, substitution variants or addition variants of any of the foregoing that maintain at least some degree of GLP-1 activity can also serve as a GLP-1 moiety in the conjugates of the invention.
[0131] Still further GLP-1 moieties for use in connection with the present invention include any of the GLP-1 moieties described herein modified via methylation, N-terminal modification and/or glycosylation.
[0132] With respect to methylation, a GLP-1 moiety may possess one or more methyl or other lower alkyl groups at one or more positions of the GLP-1 sequence. Examples of such groups include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, pentyl, and so forth. Sites of modification include residues corresponding to positions 7, 8, 9, and/or 10 [based on a GLP-1 (7-36) numbering convention], with the 7 and/or 9 positions being preferred.
Introduction of one or more methyl groups is expected to modify the dipeptidyl peptidase IV (DPP IV) recognition site, such that the modified GLP-1 is protected against degradation by DPP IV.
[0133] Introduction of N-terminal modifications to GLP-1 may be carried out as described by Gallwitz et al. (2000) Regulatory Peptides 86(1-3):103-11 1. Illustrative GLP-1 moieties with alterations at the N-terminus include N-methylated GLP-1 (N-me-GLP-1), alpha-methylated GLP-1 (alpha-me-GLP-1), desamidated GLP-1 (desamino-GLP-1), and
imidazole-lactic acid substituted GLP-1 (imi-GLP-1), among others, and are suitable for use in the present invention.
[0134] Methods for synthesizing various other analogues of GLP-1 moieties, including methyl-derivatives as described above, as well examples of such additional GLP-1 analogues are described in WO 2000/34332, WO 2004/074315 and WO 2005/058955.
[0135] With respect to glycosylation, the GLP-1 moieties described herein may also contain one or more glycosides. Although any glycoside can be used, the GLP-1 moiety is preferably modified by introduction of a monosaccharide, a disaccharide or a trisaccharide. Although any site on the GLP-1 moiety may be modified by introduction of a saccharide, preferably, the saccharide is introduced at a residue or residues corresponding to any one or more of positions 7, 8, or 9 [based on a GLP-1 (7-36) numbering convention] to protect the peptide against DPP IV proteolysis. Further, additional glycosides may be introduced, e.g., at any one or more of positions 22, 23 and 24 [again, based on a GLP-1 (7-36) numbering convention] to increase the helicity through the central portion of the peptide, as well as provide additional resistance to proteolysis.
[0136] Glycosylated GLP-1 moieties are prepared using conventional Fmoc chemistry and solid phase peptide synthesis techniques, where the desired protected glycoamino acids are prepared prior to peptide synthesis and then introduced into the peptide chain at the desired position during peptide synthesis. Preparation of amino acid glycosides is described in U.S. Patent No. 5,767,254. Briefly, alpha and beta selective glycosylations of serine and threonine residues are carried out using the Koenigs-Knorr reaction and Lemieux's in situ anomerization methodology with Schiff base intermediates. Deprotection of the Schiff base glycoside is then carried out using mildly acidic conditions or hydrogenolysis.
[0137] Monosaccharides that may be used for introduction at one or more amino acid residues of GLP-1 include glucose (dextrose), fructose, galactose, and ribose. Additional monosaccharides suitable for use include glyceraldehydes, dihydroxyacetone, erythrose, threose, erythrulose, arabinose, lyxose, xylose, ribulose, xylulose, allose, altrose, mannose, as well as others. Glycosides, such as mono-, di-, and trisaccharides for use in modifying a GLP-1 moiety, may be naturally occurring or may be synthetic.
[0138] The present invention also includes pharmaceutical preparations comprising a conjugate as provided herein in combination with a pharmaceutical excipient. Generally, the conjugate itself will be in a solid form (e.g., a precipitate), which can be combined with a suitable pharmaceutical excipient that can be in either solid or liquid form.
[0139] Exemplary excipients include, without limitation, those selected from the group consisting of carbohydrates, inorganic salts, antimicrobial agents, antioxidants, surfactants, buffers, acids, bases, and combinations thereof.
[0140] A carbohydrate such as a sugar, a derivatized sugar such as an alditol, aldonic acid, an esterified sugar, and/or a sugar polymer may be present as an excipient. Specific carbohydrate excipients include, for example: monosaccharides, such as fructose, maltose, galactose, glucose, D-mannose, sorbose, and the like; disaccharides, such as lactose, sucrose, trehalose, cellobiose, and the like; polysaccharides, such as raffinose, melezitose,
maltodextrins, dextrans, starches, and the like; and alditols, such as mannitol, xylitol, maltitol, lactitol, xylitol, sorbitol (glucitol), pyranosyl sorbitol, myoinositol, and the like.
[0141] The excipient can also include an inorganic salt or buffer such as citric acid, sodium chloride, potassium chloride, sodium sulfate, potassium nitrate, sodium phosphate monobasic, sodium phosphate dibasic, and combinations thereof.
[0142] The preparation may also include an antimicrobial agent for preventing or deterring microbial growth. Nonlimiting examples of antimicrobial agents suitable for the present invention include benzalkonium chloride, benzethonium chloride, benzyl alcohol, cetylpyridinium chloride, chlorobutanol, phenol, phenylethyl alcohol, phenylmercuric nitrate, thimersol, and combinations thereof.
[0143] An antioxidant can be present in the preparation as well. Antioxidants are used to prevent oxidation, thereby preventing the deterioration of the conjugate or other components of the preparation. Suitable antioxidants for use in the present invention include, for example, ascorbyl palmitate, butylated hydroxyanisole, butylated hydroxytoluene, hypophosphorous acid, monothioglycerol, propyl gallate, sodium bisulfite, sodium
formaldehyde sulfoxylate, sodium metabisulfite, and combinations thereof.
[0144] A surfactant may be present as an excipient. Exemplary surfactants include: polysorbates, such as "Tween 20" and "Tween 80," and pluronics such as F68 and F88 (both of which are available from BASF, Mount Olive, New Jersey); sorbitan esters; lipids, such as phospholipids such as lecithin and other phosphatidylcholines, phosphatidylethanolamines (although preferably not in liposomal form), fatty acids and fatty esters; steroids, such as cholesterol; and chelating agents, such as EDTA, zinc and other such suitable cations.
[0145] Acids or bases may be present as an excipient in the preparation. Nonlimiting examples of acids that can be used include those acids selected from the group consisting of
hydrochloric acid, acetic acid, phosphoric acid, citric acid, malic acid, lactic acid, formic acid, trichloroacetic acid, nitric acid, perchloric acid, phosphoric acid, sulfuric acid, fumaric acid, and combinations thereof. Examples of suitable bases include, without limitation, bases selected from the group consisting of sodium hydroxide, sodium acetate, ammonium hydroxide, potassium hydroxide, ammonium acetate, potassium acetate, sodium phosphate, potassium phosphate, sodium citrate, sodium formate, sodium sulfate, potassium sulfate, potassium fumerate, and combinations thereof.
[0146] The pharmaceutical preparations encompass all types of formulations and in particular those that are suited for injection, e.g., powders that can be reconstituted as well as suspensions and solutions. The amount of the conjugate (i.e., the conjugate formed between the active agent and the polymer described herein) in the composition will vary depending on a number of factors, but will optimally be a therapeutically effective dose when the composition is stored in a unit dose container (e.g., a vial). In addition, the pharmaceutical preparation can be housed in a syringe. A therapeutically effective dose can be determined experimentally by repeated administration of increasing amounts of the conjugate in order to determine which amount produces a clinically desired endpoint.
[0147] The amount of any individual excipient in the composition will vary depending on the activity of the excipient and particular needs of the composition. Typically, the optimal amount of any individual excipient is determined through routine
experimentation, i.e., by preparing compositions containing varying amounts of the excipient (ranging from low to high), examining the stability and other parameters, and then
determining the range at which optimal performance is attained with no significant adverse effects.
[0148] Generally, however, the excipient will be present in the composition in an amount of about 1% to about 99% by weight, preferably from about 5%-98% by weight, more preferably from about 15-95% by weight of the excipient, with concentrations less than 30% by weight most preferred.
[0149] These foregoing pharmaceutical excipients along with other excipients are described in "Remington: The Science & Practice of Pharmacy", 19th ed., Williams &
Williams, (1995), the "Physician's Desk Reference", 52"d ed., Medical Economics, Montvale, NJ (1998), and Kibbe, A.H., Handbook of Pharmaceutical Excipients, 3rd Edition, American Pharmaceutical Association, Washington, D.C., 2000.
[0150] The pharmaceutical preparations of the present invention are typically, although not necessarily, administered via injection and are therefore generally liquid solutions or suspensions immediately prior to administration. The pharmaceutical preparation can also take other forms such as syrups, creams, ointments, tablets, powders, and the like. Other modes of administration are also included, such as pulmonary, rectal, transdermal, transmucosal, oral, intrathecal, subcutaneous, intra-arterial, and so forth.
[0151] As previously described, the conjugates can be administered parenterally by intravenous injection, or less preferably by intramuscular or by subcutaneous injection.
Suitable formulation types for parenteral administration include ready- for-injection solutions, dry powders for combination with a solvent prior to use, suspensions ready for injection, dry insoluble compositions for combination with a vehicle prior to use, and emulsions and liquid concentrates for dilution prior to administration, among others.
[0152] The invention also provides a method for administering a conjugate as provided herein to a patient suffering from a condition that is responsive to treatment with conjugate. The method comprises administering, generally via injection, a therapeutically effective amount of the conjugate (preferably provided as part of a pharmaceutical preparation). The method of administering may be used to treat any condition that can be remedied or prevented by administration of the particular conjugate. Those of ordinary skill in the art appreciate which conditions a specific conjugate can effectively treat. The actual dose to be administered will vary depend upon the age, weight, and general condition of the subject as well as the severity of the condition being treated, the judgment of the health care professional, and conjugate being administered. Therapeutically effective amounts are known to those skilled in the art and/or are described in the pertinent reference texts and literature. Generally, a therapeutically effective amount will range from about 0.001 mg to 100 mg, preferably in doses from 0.01 mg/day to 75 mg/day, and more preferably in doses from 0.10 mg/day to 50 mg/day.
[0153] The unit dosage of any given conjugate (again, preferably provided as part of a pharmaceutical preparation) can be administered in a variety of dosing schedules depending on the judgment of the clinician, needs of the patient, and so forth. The specific dosing schedule will be known by those of ordinary skill in the art or can be determined
experimentally using routine methods. Exemplary dosing schedules include, without limitation, administration five times a day, four times a day, three times a day, twice daily, once daily, three times weekly, twice weekly, once weekly, twice monthly, once monthly,
and any combination thereof. Once the clinical endpoint has been achieved, dosing of the composition is halted.
[0154] It is to be understood that while the invention has been described in conjunction with the preferred specific embodiments thereof, that the foregoing description as well as the experimental that follow are intended to illustrate and not limit the scope of the invention. Other aspects, advantages and modifications within the scope of the invention will be apparent to those skilled in the art to which the invention pertains.
[0155] All articles, books, patents, patent publications and other publications referenced herein are hereby incorporated by reference in their entireties.
EXPERIMENTAL
[0156] The practice of the invention will employ, unless otherwise indicated, conventional techniques of organic synthesis and the like, which are understood by one of ordinary skill in the art and are explained in the literature. Such techniques are fully explained in the literature. See, for example, J. March, Advanced Organic Chemistry:
Reactions Mechanisms and Structure, 4th Ed. (New York): Wiley- Interscience, 1992), supra.
[0157] In the following examples, efforts have been made to ensure accuracy with respect to numbers used (e.g., amounts, temperatures, and so forth), but some experimental error and deviation should be accounted for. Unless otherwise indicated, temperature is in degrees Celsius and pressure is at or near atmospheric pressure at sea level. Each of the following examples is considered to be instructive to one of ordinary skill in the art for carrying out one or more embodiments described herein.
[0158] Recombinant-GLP-1 used in the experimental was obtained from LGM
Pharma Inc. (Boca Raton, FL 33487, USA), CAS# 107444-51-9 Batch No. 20090326.
[0159] SDS-PAGE was used to analyze the purity and assess the molecular weights of GLP-1 conjugates. Samples were analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) using Bio-Rad system (Mini-PROTEAN III Precast Gel Electrophoresis System)^ and Invitrogen system (XCell SureLock Mini-Cell). Samples were mixed with sample buffer. Then, the prepared samples were loaded onto a gel and run for approximately thirty minutes.
[0160] To analyze the purity of GLP-1 conjugates and monitor the PEGylation reactions, reversed phase high-performance liquid chromatography (RP-HPLC) was performed on an Agilent 1200 HPLC system (Agilent). Samples were analyzed using a Intrada WP-RP column (3 μηι particle size, 150 x 4.6 mm, Silverton, Japan), and mobile phases consisting of 0.09% trifluoroacetic acid in water (buffer A) and 0.04% trifluoroacetic acid in acetonitrile (buffer B). The flow rate for the column was 0.5 ml/min. The protein and PEG-protein conjugates were eluted with a linear gradient 25%-65 % over 30 minutes, and were visualized using Diode Array detector at 220, 280, and 320 nm.
[0161] Molecular weights GLP-1 compounds were analyzed with MALDI-TOF spectrophotometer, and sites of PEGylation were confirmed with peptide mapping using Trypsin/Glu-C. The digestion product was analyzed by LC/MALDI-TOF.
[0162] When present, dimers identified through RP-HPLC indicate protein dimer aggregates (and lack any polymeric component).
[0163] Cation exchange chromatography was used to purify GLP-1 conjugates. A
HiTrap SP Sepharose HP cation exchange column (Amersham Biosciences) was used with the AKTAExplorerlOO system (GE Bioscience) to purify the PEG-GLP-1 conjugates. For each conjugate solution prepared, the conjugate solution was loaded on a column that was pre-equilibrated in 10 mM NaoAc buffer, pH 4.0 (buffer A) and then washed with ten column volumes of buffer A to remove any unreacted PEG reagent. Subsequently, a linear gradient of buffer A and buffer B (1 OmM NaOAc with 1.0 M NaCl buffer, pH 4.0) was applied over 20 column volume. The eluent was monitored by UV detector at 220nm, 280 nm, and 320 nm. The purity of individual fractions the purity of GLP-1 conjugates was determined by RP-HPLC and SDS-PAGE.
[0164] The fractions of the purified GLP-1 conjugates from cation exchange chromatography were pooled and further purified with CG-71 S reverse phase
chromatography. When cation exchange chromatography was conducted, a HiTrap SP Sepharose HP cation exchange column (Roam Haas) with the AKTAExplorerlOO system (GE Bioscience). For each conjugate solution prepared, the conjugate solution was loaded on the CG71 S column that was pre-equilibrated in 5 % NaoAc/H20 v/v (buffer A) and then washed with ten column volumes of buffer A to remove undesired salt. Subsequently, a linear gradient of buffer A and buffer B (5 % NaoAc/acetonitrile v/v) was applied over 10 column volume. The eluent was monitored by UV detector at 220, 280, and 320 nm. The purity of individual fractions of GLP-1 conjugates was analyzed by RP-HPLC and SDS-PAGE. The
fractions were pooled and lyophilized, and the lyohpilyzed powder of GLP-1 conjugates was re-suspended in 2 mM HCl for endotoxin assay (LAL assay, Charles River, Inc), and aliquoted for further characterization.
Example 1
Prepa
mPEG2-C2-fomc-40K-N-Hydroxysuccinimide Derivative, 40kDa, ("mPEG2-C2-40K-NHS")
[0165] mPEG2-C2-fmoc-40K-NHS, stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging. 200 mG/mL stock solutions of were prepared in 2 mM HCl, and a molar ratio of 2.5: 1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM). MES buffer (0.5 M, pH 6.0) was added to the PEG-GLP-1 mixture to a final concentration of 20 mM, and the PEGylation was allowed to react for 3 hours. A solution containing 1 M Hydroxyamine and 1 M glycine (pH 6.0) was added to the reaction mixture to final concentrations of 100 mM Hydroxyamine and 100 mM glycine to stop the PEGylation reaction and remove undesired imidazole PEG side product. The reaction was allowed to continue for one hour, and was diluted with H20 to a conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
[0166] As for the specificity of N-terminal PEGylation, the specificity was determined to be 100% based on quantification of "signature" peptides. Briefly, protease digestion of PEG-containing compounds using trypsin and Lys-C created fragments. The Lys-C digestion created a mixture of peptide fragments 7-26, 27-34 and 35-36. If Lys26 was PEGylated, then no the digestion would not be expected to produce fragments 7-26 and 27-34 as the PEG component essentially protects the peptide from digestion at Lys26. Using this approach, it was determined that the specificity of N-terminal PEGylation was 100% (in view of the digestion pattern).
[0167] A typical SP-HP cation exchange purification profile of mono-[mPEG2-C2- fmoc-40K]-[Nter-GLP-l] is provided in FIG. 1. The mono-[mPEG2-C2-fmoc-40K]-[Nter- GLP-1] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm and 225 nm). Purity analysis of mono-[mPEG2-C2-fmoc-40K]- [Nter-GLP-1] by reverse phase HPLC determined that the purity of the purified conjugate was determined to be 98.2 % at 280 nm. The peak at 15.81 minutes was GLP-1 presumably released during analysis. See FIG. 2. Purity as determined by 4-12 % NuPage Bis-Tris SDS-PAGE with Coomassie Blue Staining (gel not shown) indicated 20 μg of purified mono- [mPEG2-C2-fmoc-40K]-[Nter-GLP-l]. The apparent large molecular weight of the conjugate, about 66-54 kDa, was a result of the slow mobility of the conjugate through the gel due to a high degree of PEG hydration. Finally, MALDI-TOF analysis (spectrum not shown) evidenced a peak at 26,450 Da, which might represent a doubly charged conjugate. A signal around 50 kDa representing singly charged conjugate was visible but weak, likely due to suppression of ionization as a result of high conjugate concentration.
Example 2
Prep P-l]
[0168] mPEG2-C2-fomc-40K-N-Hydroxysuccinimide Derivative, 40kDa, ("mPEG2-
C2-fmoc-40K-NHS"), stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging. 200 mG/mL stock solutions of were prepared in 2 mM HCl, and a molar ratio of 1.5: 1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM). NaHC03/Na2C03 buffer (1.0 M, pH 10.0) was added to the PEG-GLP-1 mixture to a final concentration of 20 mM, and the PEGylation was allowed to react for 20 minutes. To the reaction mixture a final concentration of 100 mM glycine was added to stop the PEGylation for 30 minutes, followed with diluting with H20 to the conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
[0169] As for positional isomer quantification, the specificity of lysine attachment was 100 %, based on analysis using LC-QTrap (Agilant 1 100/Fiimigan QTrap Deca).
Differentiation between attachment at Lys26-GLP-1 and Lys34-GLP-1 was not determined.
[0170] A typical SP-HP cation exchange purification profile of mono-[mPEG2-C2- fmoc-40 ]-[Lys-GLP-l] is provided in FIG. 3. The mono-[mPEG.2-C2-fmoc-40K]-[Lys- GLP-1] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm and 225 nm.). Purity analysis of mono-[mPEG2-C2-fmoc-40K]- [Lys-GLP-1 ] by reverse phase HPLC determined that the purity of the purified conjugate was determined to be .99.8 % at 280 nm. The peak at 15.8 minute was GLP-1 (0.2 %). See FIG. 4. Purity analysis mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l ] by 4-12 % NuPage Bis-Tris SDS-PAGE with Coomassie Blue staining (gel not shown) resulted in an apparent large molecular weight (about 66-54 kDa) of mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l], likely attributable to the slow mobility of the conjugate through the gel due to a high degree of PEG hydration. A band at 200 kDa might indicate conjugate association. Finally, MALDI-TOF analysis (spectrum not shown) evidenced a peak at 43,966 Da, which agrees with the calculated molecular weight of mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l ]. Another peak at 25,548 Da might represent a doubly charged conjugate.
Example 3
Synthesis and Characterization of mono-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l]
mPEG2-G2-fomc-40K-N-Hydroxysuccinimide Derivative, 40kDa, ("mPEG2-G2-40 -NHS")
[0171] mPEG2-G2-fmoc-40K-NHS, stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging. 200 mG/mL stock solutions of were prepared in 2 raM HC1, and a molar ratio of 2.5:1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM). MES buffer (0.5 M, pH 6.0) was added to the PEG-GLP-1 mixture to a final concentration of 20 mM, and the PEGylation was allowed to react for three hours. A solution containing 1 M Hydroxyamine and 1 M glycine (pH 6.0) was added to the reaction mixture to final concentrations of 100 mM Hydroxyamine
and 100 mM glycine to stop the PEGylation reaction and remove undesired imidazole PEG side product. The reaction was allowed to continue for one hour, and was diluted with H20 to the conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
[0172] As for the specificity of N-terminal PEGylation, not less than 90 % specificity was achieved, based on a measurement indicating that less than 10 % of species indicated a lysine PEGylation.
[0173] A typical SP-HP cation exchange purification profile of mono-[mPEG2-G2- fmoc-40K]-[Nter-GLP-l] is provided in FIG. 5. The mono-[mPEG2-G2-fmoc-40K]-[Nter- GLP-1 ] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm and 225 nm). Purity analysis of mono-[mPEG2-G2-fmoc-40K]- [Nter-GLP-1] by reverse phase HPLC determined that the purity of the purified conjugate was determined to be 99.2 % at 280 nm with 0.8 % free GLP-1 peptide released during lyophilization (retention Time 15.4 minute). See FIG. 6. Purity analysis of mono-[mPEG2- G2-fmoc-40K]-[Nter-GLP-l] by 4-12 % NuPage Bis-Tris SDS-PAGE with Coomassie Blue staining (gel not shown) resulted in the PEG group bound to GLP-1 causing the conjugate to smear on the gel. Impurities were not detected. Finally, MALDI-TOF analysis (spectrum not shown) for evidenced a peak at 44,408 Da, which agrees with the calculated molecular weight of the conjugate. Another peak at 25,183 Da might represent a doubly charged conjugate.
Example 4
Preparation of mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l]
[0174] mPEG2-G2-fomc-40K-N-Hydroxysuccinimide Derivative, 40kDa, ("mPEG2-
G2-fmoc-40K-NHS"), stored at -80 °C under argon, was warmed to ambient temperature under nitrogen purging. 200 mG/mL stock solutions of were prepared in 2 mM HC1, and a molar ratio of 1.5: 1 (PEG/GLP-1 ) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM). NaHC03/Na2C03 buffer (1.0 M, pH 10.0) was
added to the PEG-GLP-1 mixture to a final concentration of 20 mM, and the PEGylation was allowed to react for 20 minutes. To the reaction mixture a final concentration of 100 mM glycine was added to stop the PEGylation for 30 minutes, followed with diluting with H20 to the conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
[0175] As for positional isomer quantification, the specificity of lysine attachment was 100 %, based on analysis using LC-QTrap (Agilant 1 100/Finnigan QTrap Deca).
Differentiation between attachment at Lys26-GLP-1 and Lys34-GLP-1 was not determined.
[0176] A typical SP-HP cation exchange purification profile of mono-[mPEG2-G2- fmoc-40K]-[Lys-GLP-l] is provided in FIG. 7. The mono-[mPEG2-G2-fmoc-40K]-[Lys- GLP-1] and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm 225 nm). Purity analysis of mono-[mPEG2-G2-fmoc-40K]-[Lys- GLP-1 ] by reverse phase HPLC determined that the purity of the purified conjugate was determined to be 99.8 % at 280 nm. The peak at 15.8 minute was GLP-1 (0.2 %). See
FIG. 8. Purity analysis by 4-12 % NuPage Bis-Tris SDS-PAGE with Coomassie Blue staining resulted in an apparent large molecular weight (66-54 kDa) of mono-[mPEG2-G2- fmoc-40K]-[Lys-GLP-l], likely attributable to the slow mobility of the conjugate through the gel due to a high degree of PEG hydration. Without wishing to be bound by theory, the PEG group bound to GLP-1 causes the conjugate to smear on the gel. Impurities were not detected. Finally, MALDI-TOF analysis (spectrum not shown) evidenced a peak at 44,037 Da, which agrees with the calculated molecular weight of mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l ]. Another peak at 25,474 Da might represent a doubly charged conjugate.
Example 5
Synthesis a P-l]
mPEG2-G2-fomc-40K-N-Hydroxysuccinimide Derivative, 40kDa, ("mPEG2-G2-40K-NHS")
[0177] mPEG2-G2-fmoc-40K-NHS, stored at -80 °C under argon, is warmed to ambient temperature under nitrogen purging. 200 mG/mL stock solutions of were prepared
in 2 mM HCl, and a molar ratio of 4.5:1 (PEG/GLP-1) was added to GLP-1 solution to reach a final GLP-1 concentration of 1.5 mG/mL (0.45 mM). MES buffer (0.5 M, pH 6.0) was added to the PEG-GLP-1 mixture to a final concentration of 20 mM, and the PEGylation was allowed to react for 3 hours. A solution containing 1 M Hydroxyamine and 1 M glycine (pH 6.0) was added to the reaction mixture to final concentrations of 100 mM Hydroxyamine and 100 mM glycine to stop the PEGylation reaction and remove undesired imidazole PEG side product. The reaction was allowed to continue for one hour, and was diluted with H20 to the conductivity below 0.5 mS/cm (25 °C). pH was then adjusted to 4.0 using glacial acetic acid prior to column chromatography purification.
[0178] A typical SP-HP cation exchange purification profile of di-[mPEG2-G2-fmoc-
40 ]-[Nter-Lys-GLP-l] is provided in FIG. 9. The di-[mPEG2-G2-fmoc-40K]-[Nter-Lys- GLP-1], mono-mPEG2-G2-fmoc-40K-Nter-GLP-l and unreacted PEG are indicated and the lines correspond to absorbance at various wavelengths (e.g., 280 nm 225 nm). Purity analysis of di-[mPEG2-G2-fmoc-40K]-[Nter-Lys-GLP-l] by reverse phase HPLC determined that the purity of the purified conjugate is determined to be 100 % at 280 nm. See FIG. 10. Purity analysis of di-[mPEG2-G2-fmoc-40K]-[Nter-Lys-GLP-l] by 4-12 % NuPage Bis-Tris SDS- PAGE with Coomassie Blue staining (gel not shown) indicated that the PEG group bound to GLP-1 caused the conjugate to smear on the gel. Impurities were not detected.
Example 6
Stability of
mono-[mPEG2-C2-fmoc-40K]-[Nter-GLP-l], mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l], mono-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l], and
mono- [mPEG2-G2-f moc-40K]- f Lys-GLP- 1 ]
(Compounds of Examples 1-4)
[0179] Mono-[mPEG2-C2-fmoc-40K]-[Nter-GLP-l], mono-[mPEG2-C2-fmoc-40K]- [Lys-GLP-1], mono-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l], and mono-[mPEG2-C2-40K]- [Lys-GLP-1] were shown
[0180] The gradient settings for GLP-1 conjugates and PEG quantification were as follows: mobile phase A - 0.1 % fomic acid/H20; and mobile phase B - 0.1 % fomic acid/acetonitrile, wherein the parameters for each step were as follows: step 0, total time -
0.00, flow rate 500 μΐ/min, 80%A/20%B; step 1, total time - 1.00, flow rate 500 μΐ/min, 80%A/20%B; step 2, total time - 6.00, flow rate 500 μΐ/min, 55%A/45%B; step 3, total time - 1 1.00, flow rate 500 μΐ/min, 35%A/65%B'; step 4, total time - 11.30, flow rate 500 μΐ/min, 10%A/90%B; step 5, total time - 13.00, flow rate 500 μΐ/min, 10%A/90%B; and step 6, total time - 13.01 , flow rate 750 μΐ/min, 80%A/20%B. An Intrada reverse phase column (WP-RP 3 μηι, 5mm x 150 mm) was used.
[0181] The gradient settings for GLP-1 (7-36) and GLP-1 (9-36) quantification were as follows: mobile phase A - 10 mM ammonium acetate/H20; and mobile phase B - 10 mM ammonium acetate/acetonitrile, wherein the parameters for each step were as follows: step 0, total time - 0.00, flow rate 500 μΐ/min, 75%A/25%B; step 1, total time - 1.00, flow rate 500 μΐ/min, 75%A/25%B; step 2, total time - 8.00, flow rate 500 μΐ/min, 55%A/45%B; step 3. total time - 8.30, flow rate 750 μΐ/min, 5%A/95%B; step 4, total time - 10.30, flow rate 750 μΐ/min, 5%A/95%B; step 5, total time - 10.31 , flow rate 750 μΐ/min, 75%A/25%B; and step 6, total time - 12.30, flow rate 750 μΐ/min, 75%A/25%B. An Intrada reverse phase column (WP-RP 3 μιη, 5mm x 150 mm) was used.
[0182] Mass spectrometer parameters were set to conventional conditions and the parameters associated with the auto-injector were as follows: seat capillary volumne - 2.3μί; eject speed - 200μΐνηΊΐηυΐε; injection volume - 20.00μΤ; syringe volume - ΙΟΟμί; injection mode - standard; prefetch vial mode - off; draw speed - 200μΙ7ηιίηυί6; and the draw position was 0.
[0183] Briefly, the plasma incubation and extraction procedure involved a 5 mL aliquot of freshly collected, heparin treated, EDTA-free pooled s/d rat plasma, or
IXfreeze/thawed rat plasma into a 15 mL corning centrifuge tube. To this was added 1.2 mL of 5X PBS solution to 5 mL plasma, which was then mixed by inverting the tube. The final concentration of PBS in rat plasma was 1 X PBS. The tube was centrifuged and kept on ice until used. Immediately before plasma incubation, the plasma was pre-warmed to 37 °C for five minutes. Plasma incubation time points were set as follows: T=0, 0.25 hour, 0.5 hour, 1 hour, 2 hour, 3 hour, 4 hour, 6 hour, 8 hour, 12 hour, and 24 hour. For each time point, duplicate samples were prepared by labeling 2 1.5 mL Eppendorf tubes. Protease inhibitor mix (25 uL of 10X HALT) prepared according to conventional methods, was added to the tubes and tubes were kept on ice until use.
[0184] Stock solution (50 uL of 100 X) was pippetted into plasma, mixed briefly by inverting the 15 mL centrifuge tube, and incubation started. At each incubation time point,
200 uL was pipetted to the tubes containing 25 uL inhibitor solution, and mixed by pipeting 5 times. Immediately thereafter, 225 uL of 100 % acetonitrile was added to each tube, and mixed by shaking several times. Tubes were then centrifuged at 13,000 rpm for ten minutes, 4 °C. The supernatant (200 uL from each tube) was pippetted to a new tube. A conventional reconstitution solution (200 uL) was added to the tube, and the total volume was 400 uL. The total volume was mixed by pippetting. An aliquot of this solution (200 uL) was added to a polypropylene insert, placed into amber HPLC vials, and labeled.
[0185] With respect to blood sample collection, blood samples were collected into tubes coated with sodium heparin. The sodium heparin coated tubes were previously prepared, allowed to air dry overnight and then stored at 2-8 °C pending use.
[0186] With repect to plasma sample preparation, protease inhibitor mix (100 uL of
10X Halt) solution, or 1/10 volume of the blood to be collected, was placed into pre-chilled, heparin coated tubes. Approximately 1 mL of blood was added to the tube and mixed gently by inverting the tube five times. Following collection, tubes containing samples were centrifuged at approximately 2500rpm for five minutes (at 2-6°C). Thereafter, two aliquots of resulting plasma (approximately 200 uL each) were transferred to pre-labeled tubes. The tubes were frozen on dry ice and stored at -80°C pending analysis.
[0187] Ex vivo plasma stability study of the conjugates of Examples 1 -4 (quantified by LC/MS) were conducted. Typical release kinetics of the conjugates are exemplied by mono-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l], which results are set forth in FIG. 11A. In the results set forth in FIG. 11 A, were obtained by spiking mono-[mPEG2-G2-fmoc-40K]-[Nter- GLP-1 ] at 1000 ng/mL in freshly prepared rat plasma and incubated at 37 °C. Plasma samples were taken at preset time intervals and analytes were quantified. FIG. 11A shows the decrease of mono-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l], and corresponding formation of free, released PEG, revealing the release of active GLP-1 peptide as a result of PEG linker release. FIG. 11B revealed a stable level of GLP-1 (7-36) as a result of PEG linker release throughout the plasma incubation period.
[0188] Table 1 below provides a summary of the buffer and matrix effects as a function of ti/2 of PEG linker release of GLP-1 conjugates of Examples 1-5. The reported stabilities (ti/2) simultaneous monitored and quantified released GLP-1 peptide, conjugates, and released free PEG. Quantification of GLP-1 analytes were done with HPLC and LC/MS, and Tl/2 were calculated from the slope of First Order kinetic fitting ln(Co/Ct)=£T with p<0.05 and RA2>0.98.
Table 1
Buffer and Matrix Effects
ti 2 of PEG linker release of GLP-1 conjugates of Examples 1-5.
nd = not determined
* 80% fresh plasma/20 % 5X PBS; pH increased from 7.7 to 7.9 over 24 hr
Example 7
In vitro receptor binding
[0189] Methods: The binding affinity of GLP-1 conjugates was measured in vitro using competitive radioligand displacement assays in RINm5F cells that endogenously express the GLP-1 receptor. Confluent mono-layers of cells in 24- well plates were washed with assay buffer containing 50 mM HEPES, 10 mM Dextrose, 15 mM Sodium acetate, 5 mM KC1, 120 mM NaCl, 1.2 mM MgS04, 10 mM CaCl2, 0.1% BSA, pH 7.8. Serial dilutions of test compounds (GLP-1 conjugates) were prepared in assay buffer containing protease inhibitors and equilibrium binding assays were conducted at room temperature for 3 hours using 00 pM 125I-GLP-1 as the competitive radioligand. At the end of the incubation, cells were washed with phosphate buffered saline, solubilized in 0.2M NaOH and bound radioactivity was counted using a gamma counter. Non-specific binding was measured in the presence of excess cold GLP-1 (1000 nM) and subtracted from the total binding values to yield values for specific binding at each test compound concentration. IC50 values for each compound were obtained from a non-linear regression fit of the dose-response curve.
[0190] For GLP-1 conjugates with a releasable linker, binding affinities were measured for both the intact conjugate and the free peptide following its in-vitro release. Conjugates were incubated in buffer containing 200 mM lysine at 37C, for a duration of time
that allowed for >90% release of the free peptide from the conjugate. Table 2 provides a summary of the data obtained in this in vitro study.
Table 2
Summar of bindin affinit IC50 values for GLP-1 con u ates.
Example 8
Pharmacodynamic Effects
[0191] Diabetic mice (BKS.Cg- Leprdb/Leprdb/0\aHsd) were obtained from Harlan
Laboratories and acclimated for two weeks in testing facility before experiments. Male animals 10-12 weeks old and weighing 35-45 g were randomly assigned into different testing groups (five mice in each group). ~10μ1 of blood was collected from mouse tail and the baseline blood glucose level in the animal was measured by a glucometer (OneTouch Ultra from LifeScan). Animals were injected intraperitoneally with either saline, test article, and an equimolar (based on GLP-1) of PEGylated GLP-1 conjugates (each equivalent to 75 μg of GLP-1). At different time points after compound administration, animal blood glucose levels were monitored using the glucometer. Food was removed during the whole study. FIG. 12 shows the results associated with mice treated with native (i.e., unconjugated) GLP-1 peptide, wherein glucose lowering effect in vivo was lost after four hours of dosing. FIG. 13A shows an eight-hour plot and FIG. 13B shows a 48-hour plot of the results. Data points are the average glucose levels from 5 mice, error bars represent standard error (SEM). In each of FIG. 13A and FIG. 13B, Example 5, Example 4, Example 2, Example 3 and Example 1 , corresponds to di-[mPEG2-G2-frnoc-40K]-[Nter-Lys-GLP-l], mono-[mPEG2-G2-frnoc- 40K]-[Lys-GLP-l], mono-[mPEG2-C2-frnoc-40K]-[Lys-GLP-l], mono-[mPEG2-G2-frnoc- 40K]-[Nter-GLP-l ], mono-[mPEG2-C2-frnoc-40K]-[Nter-GLP-l ], respectively.
[0192] Using data from this experiment as well as in vitro studies, it is possible to compare the relationship between in vitro release kinetics and biologic response. For example, there is an inverse relationship between a GLP-1 conjugate's glucose lowering in mice and the in vitro ti/2 (relative to a saline control and as determined at the 24 hour time
point). The relationship is depicted in FIG. 14A and FIG. 14B. In each FIG. 14A and FIG.
14B, Example 5, Example 4, Example 2, Example 3 and Example 1, corresponds to di- [mPEG2-G2-fmoc-40K]-[Nter-Lys-GLP-l], mono-[mPEG2-G2-fmoc-40K]-[Lys-GLP-l], mono-[mPEG2-C2-fmoc-40K]-[Lys-GLP-l], mono-[mPEG2-G2-fmoc-40K]-[Nter-GLP-l], mono-[mPEG2-C2-fmoc-40K]-[Nter-GLP-l], respectively.
Example 9
Pharmacokinetics of PEGylated GLP-1 In Vivo
[0193] A pharmacokinetic study was conducted using C57B1/6 mice (males, weighing
20-25g; from Harlan Laboratories). Animals were acclimated in the testing facility for at least 72 hours. Each animal was dosed intraperitoneally 1.875mg/kg (GLP-1 equivalent) of testing conjugate or native GLP-1 peptide. At different time points after dosing, mouse blood samples were collected through cardiac puncture and plasma was separated by centrifugation. Plasma PEG-GLP-1 conjugate, released GLP-1 (7-36), inactivated (9-36) GLP-1 as well as the released PEG were quantified by LC MS/MS on an ABI4000QTrap system.
Pharmacokinetic samples extraction, LC MS/MS instrument settings, and quantitation of analytes are detailed in Example 6.
[0194] FIG. 15A shows the plasma GLP-1 peptide concentration after intraperitoneal dosing. Compared to native GLP-1 peptide which was cleared from blood circulation quickly and undectable after eight hours postdosing, various PEG-GLP-1 conjugates could release GLP-1 over an extensively prolonged time. Released GLP-1 peptide could still be detected at 120 hours post dosing. In agreement with in vitro results, the plasma rates of free GLP-1 appearance from a conjugated form followed PEG linker hydrolysis chemistry quantitatively in vivo: conjugates with a relatively faster release rate released higher concentrations of GLP- 1 peptide than the slower-releasing conjugates. FIG. 15B shows the plasma concentration of PEG-GLP-1 conjugates. FIG. 15C and FIG. 15D show released PEG molecules and the degraded GLP-1 metabolite (9-36) peptide in the plasma post-dosing, respectively.
[0195] The pharmacokinetic profiles of PEG-GLP-1 conjugates and GLP-1 peptide were analyzed by Pheonix WinNonLin 6.2 (Pharsight; Sunnyvale, CA) software using non- compartmental analysis (NCA). Table 3 shows the appearent PK parameters after
intraperitoneal dosing at 1.875 mg/kg. All PEG-GLP-1 conjugates tested showed increased half-lives (HL or Tl/2) and Mean Residence Time (MRT) compared to native GLP-1 peptide.
All tested PEG-GLP-1 conjugates also showed dramatically decreased clearance (CL) and increased plasma concentration area under the curve (AUC) compared to GLP-1 peptide.
Table 3
Pharmacokinetic Parameters after Intraperitoneal Dosing at 1.875 mg/kg
HL_Lambd . AUMONF_ob MRTIM=_
Compound Tmax Cmax AUCall Vz_F_obs CI_F_obs
a_z s obs
(hr) (hr) (ng/mL) (hr*ng/mL) (mL/kg) (mL hr/kg) (hr*hr*ng/m) (hr)
0.53 0.1 310.0 192.6 7602.3 9956.5 143.1 0.76 -l] 20.20 4.0 2637.5 76572.2 698.6 24.0 2297885.5 29.38
28.41 8.0 4345.0 195190.5 368.5 9.0 8337452.5 39.99
9.22 4.0 4250.5 75322.7; 330.1 24.8 1012864.8 13.41
8.30 4.0 3747.5 79500.8 283.2 23.7 1104559.8 13.94
Claims
1. A composition comprising conjugate comprising an N-terminus-containing and lysine residue-containing GLP-1 moiety to which is covalently attached: (i) via a first linker to a first water-soluble, non-peptidic polymer having a molecular weight of greater than 5,000 Daltons at the N-terminus; and (ii) via a second linker to a second water-soluble, non-peptidic polymer having a molecular weight of greater than 5,000 Daltons at the lysine residue, wherein each of the first linker and second linker is a releasable linker.
2. The composition of claim 1 , wherein the N-terminus-containing and lysine residue-containing GLP-1 moiety is GLP-1 (7-36).
3. The composition of claim 1 , wherein the N-terminus-containing and lysine residue-containing GLP-1 moiety is Lys26/34-GLP-l (7-36).
4. The composition of claim 1 , wherein the composition lacks GLP-1 conjugates having covalent attachment of a fatty acid.
5. The composition of claim 1 , wherein the composition lacks GLP-1 conjugates having covalent attachment of a carbohydrate.
6. The composition of claim 1, wherein the composition lacks GLP-1 conjugates having covalent attachment of albumin.
7. The composition of claim 1 , wherein the non-peptidic, water-soluble polymer is linear.
8. The composition of claim 1 , wherein the non-peptidic, water-soluble polymer is branched.
9. The composition of claim 1 , wherein each of the first non-peptidic, water-soluble polymer and second non-peptidic, water-soluble polymer is, prior to attachment to the N-terminus-containing and lysine residue-containing GLP-1 is encompassed by the following structure:
wherein:
POLY is a first water-soluble polymer;
POLY2 is a second water-soluble polymer;
X1 is a first spacer moiety;
X is a second spacer moiety;
Ha is an ionizable hydrogen atom;
R1 is H or an organic radical;
R is H or an organic radical;
(a) is either zero or one;
(b) is either zero or one;
Rel, when present, is a first electron altering group;
Re2, when present, is a second electron altering group; and
(FG) is a functional group capable of reacting with an amino group of an active agent to form a releasable linkage, such as a carbamate linkage.
10. The composition of claim 1, wherein each of the first non-peptidic, water-soluble polymer and second non-peptidic, water-soluble polymer has a weight-average molecular weight in a range of from about 500 Daltons to about 100,000 Daltons.
1 1. The composition of claim 1 , wherein each of the first non-peptidic, water-soluble polymer and second non-peptidic, water-soluble polymer has a weight-average molecular weight in a range of from about 2,000 Daltons to about 50,000 Daltons.
12. The composition of claim 1 , wherein each of the first non-peptidic, water-soluble polymer and second non-peptidic, water-soluble polymer has a weight-average molecular weight in a range of from about 6,000 Daltons to about 40,000 Daltons.
13. The composition of claim 1, further comprising a pharmaceutically acceptable excipient.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US40604010P | 2010-10-22 | 2010-10-22 | |
| US61/406,040 | 2010-10-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012054861A1 true WO2012054861A1 (en) | 2012-04-26 |
Family
ID=44947199
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2011/057335 Ceased WO2012054861A1 (en) | 2010-10-22 | 2011-10-21 | Glp-1 polymer conjugates having a releasable linkage |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2012054861A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10046058B2 (en) | 2014-12-02 | 2018-08-14 | Rezolute, Inc. | Use of hydrophobic organic acids to increase hydrophobicity of proteins and protein conjugates |
| US10220075B2 (en) | 2015-06-04 | 2019-03-05 | Rezolute, Inc. | Amine pegylation methods for the preparation of site-specific protein conjugates |
Citations (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4868122A (en) | 1987-03-05 | 1989-09-19 | Vogelbusch Gesellschaft M.B.H. | Arrangement for growing permanent forms of microorganisms |
| WO1991011457A1 (en) | 1990-01-24 | 1991-08-08 | Buckley Douglas I | Glp-1 analogs useful for diabetes treatment |
| US5118666A (en) | 1986-05-05 | 1992-06-02 | The General Hospital Corporation | Insulinotropic hormone |
| US5120712A (en) | 1986-05-05 | 1992-06-09 | The General Hospital Corporation | Insulinotropic hormone |
| EP0619322A2 (en) | 1993-04-07 | 1994-10-12 | Pfizer Inc. | Prolonged delivery of peptides |
| EP0699686A2 (en) | 1994-08-30 | 1996-03-06 | Eli Lilly And Company | Biologically active fragments of glucagon-like insulinotropic peptide |
| US5523549A (en) | 1994-05-25 | 1996-06-04 | Ceramic Powders, Inc. | Ferrite compositions for use in a microwave oven |
| US5629384A (en) | 1994-05-17 | 1997-05-13 | Consiglio Nazionale Delle Ricerche | Polymers of N-acryloylmorpholine activated at one end and conjugates with bioactive materials and surfaces |
| US5767254A (en) | 1992-12-15 | 1998-06-16 | The Arizona Board Of Regents On Behalf Of The University Of Arizona | Method for making amino acid glycosides and glycopeptides |
| US5932462A (en) | 1995-01-10 | 1999-08-03 | Shearwater Polymers, Inc. | Multiarmed, monofunctional, polymer for coupling to molecules and surfaces |
| WO2000034332A1 (en) | 1998-12-07 | 2000-06-15 | Societe De Conseils De Recherches Et D'applications Scientifiques Sas | Glp-1 analogues |
| US6362254B2 (en) | 1998-03-12 | 2002-03-26 | Shearwater Corporation | Poly(ethylene glycol) derivatives with proximal reactive groups |
| WO2003058203A2 (en) | 2002-01-08 | 2003-07-17 | Eli Lilly And Company | Extended glucagon-like peptide-1 analogs |
| WO2003099847A2 (en) | 2002-05-24 | 2003-12-04 | Restoragen, Inc. | Method for enzymatic production of glp-1 (7-36) amide peptides |
| WO2004074315A2 (en) | 2003-02-19 | 2004-09-02 | Societe De Conseils De Recherches Et D'applications Scientifiques (S.C.R.A.S.) | Analogues of glp-1 |
| WO2004089280A2 (en) | 2003-04-08 | 2004-10-21 | Yeda Research And Development Co. Ltd. | Reversible pegylated drugs |
| US20040235710A1 (en) | 2001-08-28 | 2004-11-25 | Defelippis Michael Rosario | Pre-mixes of glp-1 and basal insulin |
| US20050079155A1 (en) | 2003-03-20 | 2005-04-14 | Xencor, Inc. | Generating protein pro-drugs using reversible PPG linkages |
| WO2005058955A1 (en) | 2003-12-16 | 2005-06-30 | Societe De Conseils De Recherches Et D'applications Scientifiques S.A.S. | Analogues of glp-1 |
| US7060259B2 (en) | 1999-12-23 | 2006-06-13 | Nektar Therapeutics Al, Corporation | Hydrolytically degradable carbamate derivatives of poly(ethylene glycol) |
| US20060293499A1 (en) | 2005-06-16 | 2006-12-28 | Bentley Michael D | Conjugates having a degradable linkage and polymeric reagents useful in preparing such conjugates |
| WO2007075534A2 (en) * | 2005-12-16 | 2007-07-05 | Nektar Therapeutics Al, Corporation | Polymer conjugates of glp-1 |
| US7498148B2 (en) | 2002-05-24 | 2009-03-03 | Restoragen, Inc. | Method for universal enzymatic production of bioactive peptides |
-
2011
- 2011-10-21 WO PCT/US2011/057335 patent/WO2012054861A1/en not_active Ceased
Patent Citations (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5118666A (en) | 1986-05-05 | 1992-06-02 | The General Hospital Corporation | Insulinotropic hormone |
| US5120712A (en) | 1986-05-05 | 1992-06-09 | The General Hospital Corporation | Insulinotropic hormone |
| US4868122A (en) | 1987-03-05 | 1989-09-19 | Vogelbusch Gesellschaft M.B.H. | Arrangement for growing permanent forms of microorganisms |
| WO1991011457A1 (en) | 1990-01-24 | 1991-08-08 | Buckley Douglas I | Glp-1 analogs useful for diabetes treatment |
| US5767254A (en) | 1992-12-15 | 1998-06-16 | The Arizona Board Of Regents On Behalf Of The University Of Arizona | Method for making amino acid glycosides and glycopeptides |
| EP0619322A2 (en) | 1993-04-07 | 1994-10-12 | Pfizer Inc. | Prolonged delivery of peptides |
| US5629384A (en) | 1994-05-17 | 1997-05-13 | Consiglio Nazionale Delle Ricerche | Polymers of N-acryloylmorpholine activated at one end and conjugates with bioactive materials and surfaces |
| US5523549A (en) | 1994-05-25 | 1996-06-04 | Ceramic Powders, Inc. | Ferrite compositions for use in a microwave oven |
| EP0699686A2 (en) | 1994-08-30 | 1996-03-06 | Eli Lilly And Company | Biologically active fragments of glucagon-like insulinotropic peptide |
| US5932462A (en) | 1995-01-10 | 1999-08-03 | Shearwater Polymers, Inc. | Multiarmed, monofunctional, polymer for coupling to molecules and surfaces |
| US6362254B2 (en) | 1998-03-12 | 2002-03-26 | Shearwater Corporation | Poly(ethylene glycol) derivatives with proximal reactive groups |
| WO2000034332A1 (en) | 1998-12-07 | 2000-06-15 | Societe De Conseils De Recherches Et D'applications Scientifiques Sas | Glp-1 analogues |
| US7060259B2 (en) | 1999-12-23 | 2006-06-13 | Nektar Therapeutics Al, Corporation | Hydrolytically degradable carbamate derivatives of poly(ethylene glycol) |
| US20040235710A1 (en) | 2001-08-28 | 2004-11-25 | Defelippis Michael Rosario | Pre-mixes of glp-1 and basal insulin |
| WO2003058203A2 (en) | 2002-01-08 | 2003-07-17 | Eli Lilly And Company | Extended glucagon-like peptide-1 analogs |
| WO2003099847A2 (en) | 2002-05-24 | 2003-12-04 | Restoragen, Inc. | Method for enzymatic production of glp-1 (7-36) amide peptides |
| US7498148B2 (en) | 2002-05-24 | 2009-03-03 | Restoragen, Inc. | Method for universal enzymatic production of bioactive peptides |
| WO2004074315A2 (en) | 2003-02-19 | 2004-09-02 | Societe De Conseils De Recherches Et D'applications Scientifiques (S.C.R.A.S.) | Analogues of glp-1 |
| US20050079155A1 (en) | 2003-03-20 | 2005-04-14 | Xencor, Inc. | Generating protein pro-drugs using reversible PPG linkages |
| WO2004089280A2 (en) | 2003-04-08 | 2004-10-21 | Yeda Research And Development Co. Ltd. | Reversible pegylated drugs |
| US20060171920A1 (en) | 2003-04-08 | 2006-08-03 | Yeda Research And Development Co., Ltd. | Reversible pegylated drugs |
| WO2005058955A1 (en) | 2003-12-16 | 2005-06-30 | Societe De Conseils De Recherches Et D'applications Scientifiques S.A.S. | Analogues of glp-1 |
| US20060293499A1 (en) | 2005-06-16 | 2006-12-28 | Bentley Michael D | Conjugates having a degradable linkage and polymeric reagents useful in preparing such conjugates |
| WO2007075534A2 (en) * | 2005-12-16 | 2007-07-05 | Nektar Therapeutics Al, Corporation | Polymer conjugates of glp-1 |
Non-Patent Citations (20)
| Title |
|---|
| "Physician's Desk Reference", 1998, MEDICAL ECONOMICS |
| "Remington: The Science & Practice of Pharmacy", 1995, WILLIAMS & WILLIAMS |
| DILLON ET AL., ENDOCRINOLOGY, vol. 133, 1993, pages 1907 - 1910 |
| DUGAS H., PENNY, C.: "Bioorganic Chemistry", 1981, SPRINGER VERLAG, pages: 54 - 92 |
| GALLWITZ ET AL., REGULATORY PEPTIDES, vol. 86, no. 1-3, 2000, pages 103 - 111 |
| GOKE ET AL., J. BIOT. CHEM., vol. 268, 1993, pages 19650 - 55 |
| GREENE ET AL.: "PROTECTIVE GROUPS IN ORGANIC SYNTHESIS", 1999, JOHN WILEY AND SONS, INC. |
| GREENWALD, EXP. OPIN. THER. PATENTS, vol. 2, no. 6, 1997, pages 601 - 609 |
| HARRIS ET AL., NAT. REV. DRUG DISCOV., vol. 2, no. 3, 2003, pages 214 - 22 1 |
| J. MARCH: "Advanced Organic Chemistry: Reactions Mechanisms and Structure", 1992, WILEY-INTERSCIENCE |
| KIBBE, A.H.: "Handbook of Pharmaceutical Excipients", 2000, AMERICAN PHARMACEUTICAL ASSOCIATION |
| MERRIFIELD, CHEM SOC., vol. 85, 1962, pages 2149 |
| OUCHI ET AL., DRUG DES. DISCOV., vol. 2, no. 1, 1992, pages 93 - 105 |
| OUCHI ET AL., POLYMER PREPRINTS, vol. 38, no. 1, 1997, pages 582 - 3 |
| PELEG-SCHULMAN, J NLED. CHEM., vol. 47, 2004, pages 4897 - 4904 |
| PELEG-SCHULMAN, J. MED. CHEM., vol. 47, 2004, pages 4897 - 4904 |
| SIMS ET AL., ANAL. BIOCHEM, vol. 107, 1980, pages 60 - 63 |
| STEWART, YOUNG: "Solid Phase Peptide Synthesis", 1969, FREEMAN, pages: 24 - 66 |
| ZALIPSKY, BIOCONJUG. CHEM., vol. 4, no. 4, 1993, pages 296 - 299 |
| ZLOKARNIK ET AL., SCIENCE, vol. 279, 1998, pages 84 - 88 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10046058B2 (en) | 2014-12-02 | 2018-08-14 | Rezolute, Inc. | Use of hydrophobic organic acids to increase hydrophobicity of proteins and protein conjugates |
| US10702611B2 (en) | 2014-12-02 | 2020-07-07 | Rezolute, Inc. | Use of hydrophobic organic acids to increase hydrophobicity of proteins and protein conjugates |
| US10220075B2 (en) | 2015-06-04 | 2019-03-05 | Rezolute, Inc. | Amine pegylation methods for the preparation of site-specific protein conjugates |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7104735B2 (en) | Conjugation of IL-2 moiety and polymer | |
| JP5606738B2 (en) | Factor IX moiety-polymer conjugate having a releasable linkage | |
| EP3139947B1 (en) | Conjugates of il-15 and branched polyethylene glycol | |
| KR20170125839A (en) | Conjugates of an il-7 moiety and an polymer | |
| JP6626045B2 (en) | Conjugate of polymer with cholinesterase moiety | |
| WO2013020079A2 (en) | Conjugates of an il-11 moiety and a polymer | |
| WO2006102659A2 (en) | CONJUGATES OF AN hGH MOIETY AND A POLYMER | |
| WO2012054861A1 (en) | Glp-1 polymer conjugates having a releasable linkage | |
| WO2012054822A1 (en) | Pharmacologically active polymer-glp-1 conjugates | |
| WO2012064845A2 (en) | G-csf polymer conjugates having a releasable linkage | |
| WO2012064867A1 (en) | Pharmacologically active polymer-g-csf conjugates | |
| HK40053166A (en) | Conjugates of an il-2 moiety and a polymer | |
| HK1130191B (en) | Factor ix moiety-polymer conjugates having a releaseable linkage |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11782280 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 11782280 Country of ref document: EP Kind code of ref document: A1 |