WO2011073437A2 - Bacteriocin- and prebiotic-based cosmetic or dermatological compositions - Google Patents
Bacteriocin- and prebiotic-based cosmetic or dermatological compositions Download PDFInfo
- Publication number
- WO2011073437A2 WO2011073437A2 PCT/EP2010/070167 EP2010070167W WO2011073437A2 WO 2011073437 A2 WO2011073437 A2 WO 2011073437A2 EP 2010070167 W EP2010070167 W EP 2010070167W WO 2011073437 A2 WO2011073437 A2 WO 2011073437A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- plantaricin
- skin
- bacteriocin
- agents
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/73—Polysaccharides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/60—Sugars; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/10—Anti-acne agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q15/00—Anti-perspirants or body deodorants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/005—Preparations for sensitive skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/008—Preparations for oily skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/006—Antidandruff preparations
Definitions
- the present invention relates to compositions comprising, in a physiologically acceptable medium, the combination of at least one bacteriocin and at least one prebiotic. It also relates to cosmetic uses and processes involving the combination of at least one bacteriocin and at least one prebiotic. It still concerns the therapeutic use of this association.
- the cutaneous flora is composed of several species identified so far: Staphylococci (S.epidermis, S.hominis, S.haemolyticus, S.aureus), Corynebacteria (C.lipophiles, C.urealyticum, C.minutissimum, C.jeikeium) , Propionibacteria (P. acnes, P. avidum, P. granulosum, P.propionicum), Micrococci (M.luteus, M.varians), Streptococci (groups A, B and G), Brevibacteries.
- Staphylococci S.epidermis, S.hominis, S.haemolyticus, S.aureus
- Corynebacteria C.lipophiles, C.urealyticum, C.minutissimum, C.jeikeium
- Propionibacteria P. acnes, P. avidum, P. granul
- axillary microflora is mainly composed of Gram + bacteria, such as staphylococci (eg, S.epidermidis, S.hominis, etc.), coryneforms (more particularly C.xerosis), micrococci (eg, M.Luteus) and Propionibacteria. .
- staphylococci eg, S.epidermidis, S.hominis, etc.
- coryneforms more particularly C.xerosis
- micrococci eg, M.Luteus
- Propionibacteria Propionibacteria.
- the axillary bad odors are attributed to the microbial biotransformation of natural, odorless secretions into volatile odorants. Strong axillary odors are mainly related to the presence of C. xerosis in this area.
- antibiotics or bactericides raises the problem of the non-specificity of action aimed indifferently at the pathogenic flora and the resident flora, and the problem of the risk of the appearance of bacterial resistance, as well as skin tolerance problems (irritations, allergies). , .
- the application FR 2 916 634 proposes the combination of a fructooligosaccharide and an inducer of antimicrobial peptides to improve the quality of the microbial flora or restore its equilibrium. There remains, however, the need for improved cosmetic compositions and treatments to restore a normal balance of cutaneous microbial flora.
- the subject of the present invention is therefore the combination of at least one bacteriocin and at least one prebiotic, as well as a cosmetic and / or dermatological composition comprising this combination.
- the present invention also relates to the use of said combination as a cosmetic agent.
- composition comprising said combination in a physiologically acceptable medium.
- Another subject of the invention also relates to a cosmetic process for treating keratin materials, comprising the application of such a composition to keratin materials.
- “Physiologically acceptable medium” means a medium compatible with the skin and / or its integuments or mucous membranes which has a pleasant color, odor and feel and which does not generate unacceptable discomfort (tingling, tightness, redness), likely to divert the consumer from using this composition.
- keratin materials is meant the skin (body, face, eye area, scalp), hair, eyelashes, eyebrows, hair, nails, lips, mucous membranes.
- Bacteriocins are peptide compounds naturally synthesized by certain bacteria. They play an important role in the competition between bacterial strains.
- the bacteriocins can be bactericidal, that is to say eliminate certain microorganisms and / or they can be bacteriostatic, that is to say, inhibit the growth of certain microorganisms.
- the bactericidal or bacteriostatic activity is directed against certain species close to the producing strain.
- the bacteriocins are generally cationic and amphiphilic peptides, responsible for the permeabilization of the membrane of the target cells. Indeed, they bind to certain membrane receptors and cause the formation of pores. The membrane is thus made permeable to certain compounds such as ions or molecules. This is usually lethal to the target bacteria.
- the bacteriocins are classified according to structural data in four classes, as proposed by Klaenhammer (1993). These four classes are:
- Class I. Lantibiotics peptides smaller than 5 kDa, heat-stable and containing unusual sulfur-containing amino acids formed post-translationally, ie lanthionine, ⁇ -methyl lanthionine, dehydrobutyrin and dehydroalanine. They can be divided into two types: class Ia which comprises elongated hydrophobic cationic peptides containing up to 34 amino acids and class Ib which comprises negatively charged or no net charge globular peptides and containing up to 19 amino acids (McAuliffe et al., 2001, Twomey et al., 2002). There are also two-component class I bacteriocins such as cytolysin, lacticin 3147 and plantaricin W (see Table 3).
- Class I bacteriocins which may be used according to the invention may be nisin A, epidermine, subtilin, lactocin S, salivaricin A or plantaricin C.
- class I bacteriocins described in Tables 1, 2 and 3 below can be used.
- Table 1 Examples of class la bacteriocins
- Class II Peptides less than 10 kDa, heat stable, not containing modified amino acids.
- the sequences of some bacteriocins belonging to this class are shown in Table 4.
- Table 4 Sequences of some class II bacteriocins Class Ma: "Pediocin-like bacteriocin"
- ASGLAGM (SEQ ID NO: 3)
- Piscicoline 126 MKTVKELSVKEMQLTTGGKYYGNGVSCNKNGCTVDWSKAIGIIGNNAAANLTT
- GFLGGAIPGKC SEQ ID NO: 7
- Mb class "Two-peptides bacteriocin"
- ABP-1 18 a KRGPNCVGNFLGGLFAGAAAGVPLGPAGIVGGANLGMVGGALTCL (SEQ ID NO: 1]
- Lactocin 705 a MDNLNKFKKLSDNKLQATIGG (SEQ ID NO: 1 1)
- Lactococcin MN M IRGTGKGLAAAMVSGAAMGGAIGAFGGPVGAIMGAWGGAVGGAMKYSI
- Plantaricin EF E FNRGGYNFGKSVRHVVDAIGSVAGIRGILKSIR (SEQ ID NO: 15)
- Plantaricin A MKIQIKGMKQLSNKEMQKIVGGKSSAYSLQMGATAIKQVKKLFKKWGW (SEQ ID NO: 1
- Lactococcin 972 MKTKSLVLALSAVTLFSAGGIVAQAEGTWQHGYGVSSAYSNYHHGSKTHSATV
- the bacteriocins of the subclass Ma are peptides having a structure similar to that of pediocin, which was the first bacteriocin of this group described.
- the bacteriocins of the subclass Ma contain between 27 and 48 amino acids and all have a hydrophobic N-terminal portion containing the YGNGV consensus sequence as well as a disulfide bridge and a less conserved, hydrophobic or amphiphilic C-terminal portion which determines the specificity of action (Fimland et al., 2000, Richard et al., 2006).
- This class may also be more particularly defined as possessing in particular a conserved N-terminal sequence YGNGVxCxxxxCxV (Klaenhammer, 1993, Ennahar et al., 2000, Nés and Holo, 2000). They all have anti-Listeria activity. Some bacteriocins of this subclass also contain a second bridge disulfide in their C-terminal domain that appears to be important in the stabilization of the tertiary structure. It also appears to provide better antimicrobial activity, better resistance to high temperature exposure, and a broader spectrum of action (Eijsink et al., 1998, Fimland et al., 2000; al., 2006, Richard et al., 2006).
- Subclass Mb comprises the bacteriocins in need of two peptides for activity.
- Two types of class Mb bacteriocins can be distinguished: type E (Enhancing) where the function of one of the two peptides is to increase the activity of the other and type S (Synergy) where the two peptides are complementary .
- the subclass Ile contains the bacteriocins that can not be classified in the other subclasses.
- class Ma bacteriocins that can be used according to the invention, mention may be made of the bacteriocins of Table 5. It is also possible to mention coagulin. Table 5: Examples of class Ma bacteriocins
- class Mb bacteriocins that can be used according to the invention, mention may be made of lactococcin G, lactacin, lactocaine 705, thermophilin and plantaricin. EF, leucocin, carnocine, acidocine. In particular, mention may be made of the bacteriocins listed in Table 6 below.
- Lavoisier Plantaricin 149 is a class II bacteriocin of sequence YSLQMGATAIKQVKKLFKKKGG (SEQ ID NO: 20).
- bacteriocins Proteins larger than 30 kDa and sensitive to heat. The structure and mode of action of these bacteriocins differs completely from other bacteriocins produced by lactic acid bacteria. To date, four bacteriocins of this class have been identified: helveticin J produced by Lactobacillus helveticus A, enterolysin A produced by Enterococcus faecium, zoocin A produced by Spreptococcus zooepidemicus and millericin B produced by Streptococcus milleri (Nilsen et al. al., 2003; Papagianni 2003; Nigutova et al. 2007).
- class III bacteriocins that can be used according to the invention, mention may be made of the bacteriocins listed in Table 8 below.
- the bacteriocins used in the present invention can be produced by any suitable method known to those skilled in the art. They can be synthesized easily by the skilled person using conventional techniques of peptide synthesis, solid phase or in solution (M. Bodanszky, Principles of Peptide Synthesis, 2nd ed., 1993, Springer-Verlag Edition).
- the bacteriocins used according to the invention can also be produced by microorganisms, using bioengineering methods. In this case, it may be necessary to extract and purify the peptide producing microorganisms, before formulation.
- the bacteriocins used according to the invention are synthetic peptides.
- the bacteriocin used is a class I bacteriocin.
- the class I bacteriocin may especially be chosen from those mentioned above.
- class I bacteriocin may be selected from the group consisting of the bacteriocins listed in Tables 1, 2 and 3.
- class I bacteriocin is selected from the group consisting of nisin A , epidermin, salivaricin A, plantaricin C and gallidermin.
- the bacteriocin used is a class II bacteriocin, in particular a class Ma, Mb or Ile bacteriocin.
- the subject of the invention is the use of at least one of the class I bacteriocins mentioned above, more particularly a class Ma bacteriocin chosen from the group consisting of the bacteriocins listed in Table 4. or 5.
- the class Ma bacteriocin is selected from the group consisting of pediocin PA-1, sakacin A and sakacin P.
- the bacteriocin used is a bacteriocin of Mb class.
- the subject of the invention is the use of at least one of the class Mb bacteriocins mentioned above, more particularly a class Mb bacteriocin selected from the group consisting of bacteriocins listed in Table 4 or 6.
- the bacteriocin used is a class Ile bacteriocin.
- the subject of the invention is the use of at least one of the Ile class bacteriocins mentioned above, more particularly an Ile-class bacteriocin chosen from the group consisting of the bacteriocins listed in Table 4. or 7.
- the bacteriocin used is a plantaricin class II. More preferably, the plantaricin class II is selected from plantaricin A, plantaricin C19 and plantaricin 149.
- the bacteriocin in the combination is a type III bacteriocin.
- Type III bacteriocin may more particularly be selected from the group consisting of helveticin J, enterolysin A, zooein A and millericin B.
- the combination according to the invention comprises a single bacteriocin.
- the combination according to the invention comprises several bacteriocins. It can be in particular a mixture comprising several bacteriocins of the same class, or comprising bacteriocins of different classes (for example, class I + class II, class I + class III, class II + class III).
- the combination according to the invention may therefore comprise the mixture of the following bacteriocins:
- Class II bacteriocin is a class II plantaricin, in particular plantaricin A, plantaricin C19 or plantaricin 149;
- class I bacteriocin selected from the group consisting of nisin A, epidermin, salivaricin A, plantaricin C and gallidermine
- class II bacteriocin selected from the group consisting of pediocin PA-1, sakacin A and sakacin P.
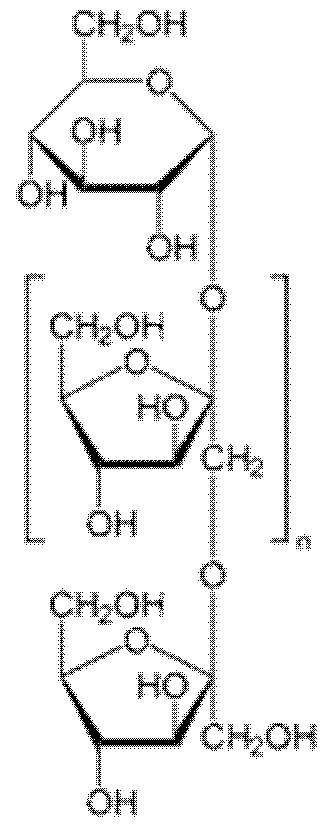
- the "prebiotics” are oligosaccharides and / or polysaccharides consisting of 2 to 100 sugar units, preferably 2 to 30 sugar units.
- the first official definition given by Gibson et al. in 1995 is the following "non-digestible substance that induces a physiological effect beneficial to the host by specifically stimulating the growth and / or activity of a limited number of bacterial populations already established in the colon".
- one or more prebiotics are used in order to maintain a favorable environment for the development of endogenous cutaneous flora.
- prebiotics are used to guide the metabolism of the entire skin flora to maintain a good balance of the latter while not promoting the development of certain pathogenic bacteria.
- the prebiotics used in the context of the present invention are those likely to be metabolized by the beneficial bacteria of the skin flora.
- the prebiotic used in the compositions of the invention is a prebiotic metabolized by the beneficial seeds of the skin, but which are not metabolizable by the undesirable germs responsible for skin disorders, including cosmetic or dermatological treatment. is desired.
- prebiotics modulate the composition and / or the activity of the natural ecosystem, giving priority to microorganisms having a positive effect on the skin.
- a mixture of prebiotics is used in the compositions, uses and processes according to the invention.
- Prebiotics contain carbohydrates. They are more specifically composed of oligosaccharides.
- the oligosaccharides may be produced in particular from glucose, galactose, xylose, maltose, sucrose, lactose, starch, xylan, hemicellulose, inulin, gums, especially from acacia gum, or a mixture thereof.
- Inulin is particularly abundant in the rhizomes of plants, notably the Jerusalem artichoke and the chicory from which it is extracted industrially. It is also found in other plants belonging to the family Asteraceae such as Jerusalem artichokes or dahlias onions and burdock. It is considered a soluble dietary fiber.
- the fructo-oligosaccharides correspond to the general formula G (F) n where G is a glucose, F fructose unit and n ranges from 1 to 10.
- the fructo-oligosaccharides (FOS) are produced either by partial enzymatic hydrolysis inulin (eg Raftilose® from Orafti-Belgium), or by enzymatic synthesis from sucrose (eg Actilight® from Beghin Meiiji industries-France).
- the prebiotic used in the invention corresponds to a mixture of FOS.
- GF2 + GF3 + GF4 blends such as the Quantom FOS95 from Quantum Hi-Tech or Actilight® from Beghin Meiiji Industries-France, the latter corresponding to a GF2 37%, GF3 53% and GF4 mixture. %.
- Gluco-oligosaccharides are oligosaccharides consisting of a sequence of glucose units linked at a1 -6 and which may also contain ⁇ 1 -2, ⁇ 1-3 bonds; a1 -4. They are synthesized by a transglucosylation reaction catalyzed by enzymes of the glucan saccharase family.
- Pyrodextrins are a mixture of oligosaccharides derived from the hydrolysis of starch. Isomaltooligosaccharide
- glucose oligomers bound to a (1-6) with a degree of polymerization of between 2 and 5.
- the Showa Sango lsomalto900P can be used.
- the xylo-oligosaccharide oligosaccharides consist of ⁇ (1 -4) -linked xylose.
- the TOS 100 from Yakult Honsha Co.Ltd. Japan.
- the prebiotic component used in the combination according to the invention corresponds to a mixture of prebiotics as defined above.
- it may be a mixture of prebiotics of the same type.
- FOS in particular a GF2 + GF3 + GF4 mixture, such as Quantom FOS95 from Quantum Hi-Tech or Actilight® from Beghin Meiiji Industries-France, the latter corresponding to a mixture GF2 37%, GF3 53% and GF4 10%.
- the invention may be a mixture of prebiotics of different types.
- the invention relates in particular to the use of a mixture of inulin with a GOS, an FOS, oligosaccharides derived from soya, pyrodextrins, an isomaltooligosachharide, a xylooligosaccharide and / or a transgalactooligosaccharide.
- a GOS such as Bioline Gova Ingredients.
- the invention also relates to the use of a mixture of a GOS with an inulin, a FOS, oligosaccharides derived from soybean, pyrodextrins, an isomaltooligosachharide, a xylooligosaccharide and / or a transgalactooligosaccharide. More particularly, it is possible to use a mixture of GOS, FOS and probiotics (Lactobacillus) such as Solabia Ecoskin.
- the present invention relates to the combination of at least one bacteriocin with at least one prebiotic.
- the combination comprises at least one prebiotic chosen from among inulin, an FOS, a GOS, an isomaltooligosaccharide and a xylooligosaccharide.
- the prebiotic is chosen from an FOS, an isomaltooligosaccharide and a xylooligosaccharide.
- the prebiotic is a mixture of prebiotics, for example an inulin + GOS or GOS + FOS + probiotic mixture.
- the combination also comprises a class I bacteriocin, in particular a bacteriocin chosen from Tables 1, 2 and 3, and in particular a bacteriocin chosen from the group consisting of nisin A, epidermin, salivaricin A, plantaricin C and gallidermin.
- the combination comprises at least one class II bacteriocin, in particular class Ma, in particular a bacteriocin chosen from Tables 4 and 5, and in particular a bacteriocin selected from the group consisting of pediocin PA-1, sakacin A and sakacin P.
- class II bacteriocin is a class II plantaricin, in particular a plantaricin class II selected from plantaricin A, plantaricin C19 and plantaricin 149.
- the bacteriocin is a plantaricin class II, in particular a class II plantaricin chosen from plantaricin A, plantaricin C19 and plantaricin 149 and the prebiotic is selected from an FOS, an isomaltooligosaccharide and a xylooligosaccharide.
- the invention relates to the combination of nisin A with a GOS, in particular GOS marketed by Solabia under the name Bioecolia.
- the invention relates to the combination of nisin A with a FOS, in particular the Actilight mentioned above.
- the invention relates to the combination of epidermidine with a GOS, in particular GOS marketed by Solabia under the name Bioecolia.
- the invention relates to the combination of plantaricin 149 with a FOS, in particular the Actilight mentioned above.
- the invention also relates to the use of the combination as described above, as a cosmetic active.
- the combination according to the invention relates to the use of the combination according to the invention as a skin care and / or cleansing agent, in particular as a care agent for sensitive, irritated skin, presenting eczema or atopic dermatitis, or as a care agent and / or cleansing for oily skin, in particular oily skin with acne or hyperseborrheic tendency. It also relates to the use of the combination according to the invention as anti-dandruff agent or deodorant agent.
- the invention also relates to the use of an association as described above for the manufacture of a composition (cosmetic and / or dermatological) intended to prevent and / or treat acne and / or hyperseborrhoea.
- the invention also relates to the use of an association as described above for the manufacture of a composition (cosmetic and / or dermatological) intended to prevent and / or treat skin irritation, eczema , or atopic dermatitis.
- the invention further relates to the use of a bacteriocin as a skin care and / or cleansing agent, in particular as a care agent for sensitive, irritated skin with eczema. or atopic dermatitis or as a care agent and / or cleansing agent for oily skin, in particular oily skin with an acne or hyperseborrhoeic tendency.
- a bacteriocin as defined above, for the manufacture of a composition for the treatment of sensitive skin, irritated skin, eczema, or atopic dermatitis.
- the invention also relates to a cosmetic and / or dermatological composition comprising, in a physiologically acceptable medium, the combination as described above.
- the invention also relates to a cosmetic and / or dermatological composition
- a cosmetic and / or dermatological composition comprising, in a physiologically acceptable medium, at least one type III bacteriocin.
- Each bacteriocin present in the compositions according to the invention is advantageously present at a concentration of between 0.0001% and 1% of active material relative to the total weight of the composition.
- the prebiotics are used at a level of between 0.01% and 70% by weight relative to the total weight. of the composition containing them. Preferably, they are present at a level of between 0.01% and 20% by weight relative to the total weight of the composition.
- the cosmetic and / or dermatological compositions according to the invention may be deodorant compositions, for the care and / or the cleaning of the skin, in particular for the care of sensitive, irritated skin, presenting with eczema or atopic dermatitis or for the care and / or the cleansing of oily skin, in particular oily skin with an acne or hyperseborrhoeic tendency.
- the compositions according to the invention can also be compositions for the treatment of films.
- compositions according to the invention may be in any of the galenic forms conventionally used in the intended application.
- those skilled in the art will take care to choose the components and any additional ingredients and / or active agents, and / or their quantity, in such a way that the advantageous properties of the combination of at least one bacteriocin and at least one a prebiotic according to the invention are not, or not substantially, impaired by the addition envisaged.
- composition according to the invention is a deodorant composition
- it may be in the form of suspension, dispersion, solution, gel, emulsions (in particular of liquid or semi-liquid, soft, semi-liquid consistency).
- solid or solid in particular oil-in-water (O / W), wax-in-water or water-in-oil (W / O) emulsion, or multiple (E / H / E or polyol / H / E or H / E / H), in the form of cream, paste, foam, microemulsion, dispersion of vesicles, in particular ionic lipids or not, wax / aqueous phase dispersions, two-phase or multiphase lotion, pulp, especially soft dough.
- compositions according to the invention may be in the form of a protection, treatment or care composition for the face, for the hands or for the body (for example, day creams, night cream, make-up removal cream, anti-solar composition, body protection or care milk, after-sun milks, lotion, gel or mousse for skin care); an aftershave composition.
- compositions are compositions for coating the skin of the body or of the face, more particularly compositions for makeup or care of the skin of the body or of the face, such as, for example, foundations or hair compositions. body makeup.
- compositions may in particular be packaged in pressurized form in an aerosol device or in a pump bottle; packaged in a device provided with a perforated wall including a grid; packaged in a device provided with a ball-on applicator; packaged in the form of sticks (sticks), in the form of loose or compacted powder; or else applied on wipes.
- the compositions may also be in the form of soaps or gels (rinsed or not rinsed). They contain in this respect the ingredients generally used in this type of products and well known to those skilled in the art.
- the compositions according to the invention can be anhydrous.
- An anhydrous composition according to the invention may especially be in the form of a stick, a cream, a soft-solid or an aerosol.
- anhydrous composition is meant a composition containing less than 2% by weight of water, or even less than 0.5% of water, and especially free of water, the water not being added during the preparation of the composition but corresponding to the residual water provided by the mixed ingredients.
- the compositions according to the invention can be solid, in particular in the form of a stick or a stick.
- solid composition it is meant that the measurement of the maximum force measured in texturometry during the insertion of a probe into the sample of formula must be at least 0.25 Newton, in particular at least equal to 0.30 Newton, especially at least 0.35 Newton, appreciated under precise measurement conditions as follows.
- the formulas are poured hot in pots 4 cm in diameter and 3 cm deep. Cooling is done at room temperature. The hardness of the formulas is measured after 24 hours of waiting.
- the pots containing the samples are characterized in texturometry using a texturometer such as that marketed by Rhéo TA-XT2, according to the following protocol: a ball-type stainless steel probe of diameter 5 mm is brought into contact with the sample at a speed of 1 mm / s.
- the measurement system detects the interface with the sample with a detection threshold equal to 0.005 newtons.
- the probe sinks 0.3 mm into the sample at a rate of 0.1 mm / s.
- the meter records the evolution of the force measured in compression over time, during the penetration phase.
- the hardness of the sample corresponds to the average of the maximum values of the force detected during the penetration, on at least 3 measurements.
- compositions according to the invention intended for cosmetic use may comprise at least one aqueous phase. They are in particular formulated in aqueous lotions or in water-in-oil, oil-in-water or multiple emulsion emulsions (triple oil-in-water-in-oil or water-in-oil-in-water emulsion). such emulsions are known and described for example by C. FOX in "Cosmetics and Toiletries" - november 1986 - Vol 101 - pages 101-122).
- the aqueous phase of said compositions contains water and in general other solvents soluble or miscible in water.
- Solvents that are soluble or miscible in water include short-chain monohydric alcohols, for example C 1 -C 4 such as ethanol, isopropanol; diols or polyols such as ethylene glycol, 1,2-propylene glycol, 1,3-butylene glycol, hexylene glycol, diethylene glycol, dipropylene glycol, 2-ethoxyethanol, diethylene glycol monomethyl ether, triethylene glycol monomethyl ether and sorbitol. More particularly, propylene glycol and glycerol, propane 1,3 diol will be used.
- composition according to the invention preferably has a pH of between 3 and 9, depending on the support chosen.
- Emulsifiers a) Oil-in-water emulsifiers
- nonionic emulsifiers such as esters of oxyalkylenated fatty acids and glycerol (more particularly polyoxyethylenated); oxyalkylenated fatty acid and sorbitan esters; oxyalkylenated fatty acid esters (oxyethylenated and / or oxypropylenated); oxyalkylenated fatty alcohol ethers (oxyethylenated and / or oxypropylenated); sugar esters such as sucrose stearate; and mixtures thereof such as the mixture of glyceryl stearate and PEG-40 stearate.
- emulsifiers that can be used in water-in-oil emulsions or triple emulsions (O / W / H or W / O / W), there may be mentioned by way of example alkyl dimethicone copolyols corresponding to formula (I) next CH,
- R 1 denotes a linear or branched C 12 -C 20 alkyl group and preferably
- R2 denotes the grouping: ⁇ CnH 2 n ⁇ (-OC2H 4 -) x ⁇ (-OC3H6-) y ⁇ O-R3,
- R3 denotes a hydrogen atom or a linear or branched alkyl radical having from 1 to 12 carbon atoms
- a is an integer from 1 to about 500;
- b is an integer from 1 to about 500;
- n is an integer from 2 to 12 and preferably 2 to 5;
- x denotes an integer ranging from 1 to about 50 and preferably from 1 to 30;
- y denotes an integer ranging from 0 to about 49 and preferably 0 to 29 provided that when y is different from zero the ratio x / y is greater than 1 and preferably varies from 2 to 1 1.
- CETYL PEG / PPG-10/1 DIMETHICONE and more particularly the mixture CETYL PEG / PPG-10/1 DIMETICONE AND DIMETHICONE (INCI name) as the product sold under the trade name ABIL EM90 by GOLDSCHMIDT or the mixture (POLYGLYCERYL-4- STEARATE and CETYL PEG / PPG-10 (AND) DIMETHICONE (AND) HEXYL LAURATE) as the product sold under the trade name ABIL WE09 by the same society.
- dimethicone copolyols corresponding to the following formula (II) CH 3 - S i -Si -O Si - C + Si (ii)
- R 4 denotes the grouping: ⁇ C m H 2 m ⁇ (-OC2H 4 -) s ⁇ (-OC3H6-) t ⁇ O-R 5 ,
- R 5 denotes a hydrogen atom or a linear or branched alkyl radical having from 1 to 12 carbon atoms
- d denotes an integer ranging from 1 to about 500
- n is an integer ranging from 2 to 12 and preferably 2 to 5,
- s denotes an integer ranging from 1 to about 50, and preferably from 1 to 30; t denotes an integer ranging from 0 to about 50 and preferably from 0 to 30; provided that the sum s + t is greater than or equal to 1.
- dimethicone copolyols of formula (II) PEG-18 / PPG-18 DIMETHICONE and more particularly the mixture CYCLOPENTASILOXANE (and) PEG-18 / PPG-18 DIMETHICONE (INCI name) such as the product sold by Dow Corning under the trade name Silicone DC 5225 C or KF-6040 from Shin Etsu.
- nonionic emulsifiers derived from fatty acid and polyol alkylpolyglycosides (APG), sugar esters and mixtures thereof.
- APG alkylpolyglycosides
- sugar esters and mixtures thereof.
- nonionic emulsifiers derived from fatty acid and polyol it is possible in particular to use the fatty acid and polyol esters, the fatty acid having in particular a C 8 -C 24 alkyl chain, and the polyols being, for example, glycerol and sorbitan.
- esters of isostearic acid and of polyols examples include esters of isostearic acid and of polyols, esters of stearic acid and of polyols, and mixtures thereof, in particular esters of isostearic acid and of glycerol, and / or sorbitan.
- esters of stearic acid and of polyols mention may in particular be made of polyethylene glycol esters such as PEG-30 dipolyhydroxystearate, such as the product sold under the name Arlacel P135 by the company ICI.
- glycerol and / or sorbitan esters examples include polyglycerol isostearate, such as the product sold under the name Isolan Gl 34 by the company Goldschmidt; sorbitan isostearate, such as the product sold under the name Arlacel 987 by the company ICI; sorbitan isostearate and glycerol, such as the product sold under the name Arlacel 986 by the company ICI, the mixture of sorbitan isostearate and polyglycerol isostearate (3 moles) sold under the name Arlacel 1690 by the Unigema company. and their mixtures.
- polyglycerol isostearate such as the product sold under the name Isolan Gl 34 by the company Goldschmidt
- sorbitan isostearate such as the product sold under the name Arlacel 987 by the company ICI
- sorbitan isostearate and glycerol such as the product sold under the name Arlacel 986 by the company ICI, the
- the emulsifier may also be chosen from alkylpolyglycosides having an HLB of less than 7, for example those represented by the following general formula (1):
- RO- (G) x (1) in which R represents a branched and / or unsaturated alkyl radical containing from 14 to 24 carbon atoms, G represents a reduced sugar containing from 5 to 6 carbon atoms, and x denotes a value ranging from 1 to 10 and preferably from 1 to 4, and G particularly denotes glucose, fructose or galactose.
- the unsaturated alkyl radical may comprise one or more ethylenic unsaturations, and in particular one or two ethylenic unsaturations.
- This alkylpolyglucoside may be used in admixture with a co-emulsifier, more particularly with a fatty alcohol and in particular a fatty alcohol having the same fatty chain as that of the alkylpolyglucoside, that is to say comprising from 14 to 24 carbon atoms. carbon and having a branched and / or unsaturated chain, and for example isostearyl alcohol when the alkylpolyglucoside is isostearylglucoside, and oleyl alcohol when the alkylpolyglucoside is oleylglucoside, optionally in the form of a self-emulsifying composition, as described for example in WO-A-92/06778.
- the mixture of isostearyl glucoside and isostearyl alcohol sold under the name Montanov WO 18 by the company SEPPIC and the mixture octyldodecanol and octyldodecylxyloside sold under the name FLUDANOV 20X by the company SEPPIC may be used.
- succinic-terminated polyolefins such as ester-terminated polyisobutylenes and their salts, especially diethanolamine salts, such as the products sold under the names Lubrizol 2724, Lubrizol 2722 and Lubrizol 5603 by the company Lubrizol or the product CHEMCINNATE 2000.
- the total amount of emulsifiers in the composition will preferably be in the composition according to the invention at contents of active material ranging from 1 to 8% by weight. weight and more particularly from 2 to 6% by weight relative to the total weight of the composition.
- the non-rinsed compositions according to the invention may contain at least one immiscible organic liquid phase in water called the fatty phase.
- This generally comprises one or more hydrophobic compounds which render said phase immiscible in water.
- Said phase is liquid (in the absence of structuring agent) at ambient temperature (20-25 ° C.).
- the organic liquid immiscible organic phase in accordance with the invention is generally constituted generally comprises at least one volatile oil and / or a non-volatile oil and optionally at least one structuring agent.
- oil is meant a fatty substance that is liquid at ambient temperature (25 ° C.) and atmospheric pressure (760 mmHg, ie 105 Pa).
- the oil can be volatile or non-volatile.
- volatile oil means an oil capable of evaporating on contact with the skin or keratin fiber in less than one hour at ambient temperature and atmospheric pressure.
- volatile oils of the invention are volatile cosmetic oils which are liquid at ambient temperature and have a non-zero vapor pressure at ambient temperature and atmospheric pressure, in particular from 0.13 Pa to 40,000 Pa (10 -3 to 300 mmHg), in particular ranging from 1.3 Pa to 13 000 Pa (0.01 to 100 mmHg), and more particularly ranging from 1.3 Pa to 1300 Pa (0.01 to 10 mmHg).
- non-volatile oil is meant an oil remaining on the skin or keratin fiber at room temperature and atmospheric pressure for at least several hours and having in particular a vapor pressure of less than 10 -3 mmHg (0.13 Pa).
- the oil may be chosen from all physiologically acceptable and in particular physiologically acceptable oils, in particular mineral, animal, vegetable and synthetic oils; in particular volatile and / or nonvolatile hydrocarbon and / or silicone and / or fluorinated oils and mixtures thereof.
- hydrocarbon-based oil means an oil comprising mainly carbon and hydrogen atoms and optionally one or more functional groups chosen from hydroxyl, ester, ether and carboxylic functions.
- the oil has a viscosity of 0.5 to 100,000 mPa.s, preferably 50 to 50,000 mPa.s and more preferably 100 to 30,000 mPa.s.
- volatile hydrocarbon oils chosen from hydrocarbon-based oils containing from 8 to 16 carbon atoms, and in particular Cs-C16 isoalkanes of petroleum origin (also known as isoparaffins), such as isododecane (also called 2,2,4,4, 6-pentamethylheptane), isodecane, isohexadecane, and for example the oils sold under the trade names Isopars or permetyls, branched C 6 -C 16 esters, isohexyl neopentanoate, and mixtures thereof.
- Other volatile hydrocarbon oils such as petroleum distillates, especially those sold under the name Shell or by Shell, may also be used; volatile linear alkanes such as those described in the patent application of Cognis DE10 2008 012 457.
- volatile silicones for example volatile linear or cyclic silicone oils, in particular those having a viscosity of ⁇ 8 centistokes (8 ⁇ 10 -6 m 2 / s), and having in particular from 2 to 7 silicon atoms, these silicones possibly comprising Alkyl or alkoxy groups having from 1 to 10 carbon atoms
- volatile silicone oil that may be used in the invention, mention may be made especially of octamethylcyclotetrasiloxane, decamethylcyclopentasiloxane, dodecamethylcyclohexasiloxane, heptamethylhexyltrisiloxane and heptamethyloctyl. trisiloxane, hexamethyl disiloxane, octamethyl trisiloxane, decamethyl tetrasiloxane, dodecamethylpentasiloxane
- R represents an alkyl group comprising from 2 to 4 carbon atoms and one or more hydrogen atoms of which may be substituted by a fluorine or chlorine atom.
- non-volatile oil that may be used in the invention, mention may be made of:
- hydrocarbon oils of animal origin such as perhydrosqualene
- vegetable hydrocarbon oils such as liquid triglycerides of fatty acids of 4 to 24 carbon atoms such as triglycerides of heptanoic or octanoic acids or oils oils of wheat germ, olive oil, sweet almond oil , palm, colza, cotton, alfalfa, poppy, pumpkin, squash, black currant, evening primrose, millet, barley, quinoa, rye, safflower,nadooulier, passionflower , musk rose, sunflower, maize, soya, squash, grape seed, sesame, hazelnut, apricot, macadamia, castor, avocado, caprylic / capric acid triglycerides as those sold by Stearineries Dubois or those sold under the names Miglyol 810, 812 and 818 by Dynamit Nobel, jojoba oil, shea butter; linear or branched hydrocarbons of mineral or synthetic origin
- esters in particular of fatty acids, such as the oils of formula R 1 COOR 2 in which R 1 represents the residue of a linear or branched higher fatty acid containing from 1 to 40 carbon atoms and R 2 represents a hydrocarbon chain, in particular branched, containing 1 to 40 carbon atoms with R 1 + R 2> 10 such as, for example, purcellin oil (cetostearyl octanoate), isononyl isononanoate, isopropyl myristate, isopropyl palmitate, alcohol benzoate C12 to C15, hexyl laurate, diisopropyl adipate, isononyl isononanoate, ethyl 2-hexyl palmitate, octyl 2-dodecyl stearate, octyl erucate 2- dodecyl, isostearyl isostearate, tridecyl trimellitate; octanoates,
- branched-chain and / or unsaturated carbon-chain liquid fatty alcohols having from 12 to 26 carbon atoms, such as octyl dodecanol, isostearyl alcohol, 2-butyloctanol, 2-hexyl decanol and 2-undecyl; pentadecanol, oleic alcohol;
- higher fatty acids such as oleic acid, linoleic acid, linolenic acid;
- fluorinated oils that may be partially hydrocarbon-based and / or silicone-based, such as fluorosilicone oils, fluorinated polyethers or fluorinated silicones, as described in document EP-A-847752; silicone oils, such as non-volatile, linear or cyclic polydimethylsiloxanes (PDMS); polydimethylsiloxanes comprising alkyl, alkoxy or phenyl groups, during or at the end of the silicone chain, groups having from 2 to 24 carbon atoms; phenyl silicones such as phenyl trimethicones, phenyl dimethicones, phenyl trimethylsiloxy diphenyl siloxanes, diphenyl dimethicones, diphenyl methyldiphenyl trisiloxanes, 2-phenylethyl trimethylsiloxysilicates, and
- compositions according to the invention comprising a fatty phase may additionally contain at least one structuring agent of said fatty phase, which may preferably be chosen from waxes, pasty compounds, mineral or organic lipophilic gelling agents and mixtures thereof.
- the wax is generally a lipophilic compound, solid at room temperature (25 ° C.), with a reversible solid / liquid state change, having a melting point greater than or equal to 30 ° C. and up to 200 ° C and especially up to 120 ° C.
- the waxes that are suitable for the invention may have a melting point greater than or equal to 45 ° C., and in particular greater than or equal to 55 ° C.
- the melting temperature corresponds to the temperature of the most endothermic peak observed in thermal analysis (DSC) as described in the ISO 1 1357-3 standard; 1999.
- the melting point of wax can be measured using a differential scanning calorimeter (DSC), for example the calorimeter sold under the name "MDSC 2920" by the company TA Instruments.
- the measurement protocol is as follows:
- a sample of 5 mg of wax placed in a crucible is subjected to a first temperature rise from -20 ° C. to 100 ° C., at the heating rate of 10 ° C./min, and is then cooled from 100 ° C. to -20 ° C at a cooling rate of 10 ° C / minute and finally subjected to a second temperature rise from -20 ° C to 100 ° C at a heating rate of 5 ° C / minute.
- the melting point of the compound is the value of the temperature corresponding to the peak apex of the curve representing the variation of the difference in power absorbed as a function of the temperature.
- the waxes that may be used in the compositions according to the invention are chosen from solid waxes at room temperature of animal, vegetable, mineral or synthetic origin, and mixtures thereof.
- Illustrative waxes suitable for the invention include hydrocarbon waxes such as beeswax, lanolin wax, and Chinese insect wax, rice bran wax, Carnauba wax Candellila wax, Ouricury wax, Alfa wax, berry wax, shellac wax, Japanese wax and sumac wax; montan wax, orange and lemon waxes, refined sunflower wax marketed under the name SUNFLOWER WAX by KOSTER KEUNEN, microcrystalline waxes, paraffins and ozokerite; polyethylene waxes, waxes obtained by Fisher-Tropsch synthesis and waxy copolymers and their esters.
- hydrocarbon waxes such as beeswax, lanolin wax, and Chinese insect wax, rice bran wax, Carnauba wax Candellila wax, Ouricury wax, Alfa wax, berry wax, shellac wax, Japanese wax and sumac wax
- montan wax, orange and lemon waxes refined sunflower wax marketed under the name SUNFLOWER WAX by KOSTER
- waxes obtained by catalytic hydrogenation of animal or vegetable oils having linear or branched C 8 -C 32 fatty chains may also be made.
- isomericized jojoba oil such as oil. trans-isomerized partially hydrogenated jojoba manufactured or marketed by Desert Whale under the trade name lso-Jojoba-50®, hydrogenated sunflower oil, hydrogenated castor oil, hydrogenated coconut oil, coconut oil hydrogenated lanolin, and the di- (trimethylol-1, 1, 1 propane) tetrastearate sold under the name Hest 2T-4S® by the company HETERENE.
- silicone waxes C 3 -C 4 ALKYL DIMETHICONE
- fluorinated waxes It is also possible to use the waxes obtained by hydrogenation of castor oil esterified with cetyl alcohol sold under the names Phytowax Ricin 16L64® and 22L73® by the company SOPHIM. Such waxes are described in the application FR-A-2792190.
- a C 2 -C 4 alkyl group comprising 20 to 40 carbon atoms
- Such a wax is especially sold under the names "Kester Wax K 82 P®", “Hydroxypolyester K 82 P®” and “Kester Wax K 80 P®” by the company Koster Keunen.
- micro-waxes that can be used in the compositions according to the invention, mention may be made in particular of carnauba micro-waxes such as that marketed under the name MicroCare 350® by the company MICRO POWDERS, and synthetic wax micro-waxes such as marketed under the name MicroEase 1 14S® by the company MICRO POWDERS, micro waxes consisting of a mixture of carnauba wax and polyethylene wax such as those marketed under the names Micro Care 300® and 310® by the company MICRO POWDERS, micro waxes consisting of a mixture of carnauba wax and synthetic wax such as that sold under the name Micro Care 325® by MICRO POWDERS, polyethylene micro-waxes such as those sold under the names Micropoly.
- carnauba micro-waxes such as that marketed under the name MicroCare 350® by the company MICRO POWDERS
- synthetic wax micro-waxes such as marketed under the name MicroEase 1 14S® by the
- composition according to the invention will preferably comprise a wax content (s) ranging from 3% to 20% by weight relative to the total weight of the composition, in particular from 5% to 15%, more particularly from 6% to 15%.
- anhydrous solid compositions in the form of a stick use will be made of polyethylene micro-waxes in the form of shape-factor crystallites of at least 2 having a melting point of 70.degree. at 1 10 ° C and preferably 70 to 100 ° C, to reduce or eliminate the presence of layers in the solid composition,
- These needle crystallites and in particular their dimensions can be characterized visually according to the following method.
- the wax is deposited on a microscope slide, which is placed on a hot plate.
- the blade and the wax are heated to a temperature generally at least 5 ° C higher than that of the melting point of the wax or wax mixture considered (e).
- the liquid thus obtained and the microscope slide are allowed to cool to solidify.
- the observation of the crystallites is carried out using a Leica DMLB100 type optical microscope, with a target selected according to the size of the objects to be viewed, and in polarized light.
- the dimensions of the crystallites are measured using an image analysis software such as those marketed by Microvision.
- the crystallite polyethylene waxes according to the invention preferably have an average length ranging from 5 to 10 ⁇ .
- average length is meant the dimension given by the statistical size distribution at half of the population, called D50.
- PERFOMALEN 400 POLYETHYLENE and PERFORMALENE 500-L POLYETHYLENE waxes from NEW PHASE TECHNOLOGIES will be used.
- the term "pasty compound” is intended to mean a lipophilic fat compound with a reversible solid / liquid state change, having in the solid state an anisotropic crystalline organization and comprising at a temperature of 23 ° C. a fraction liquid and a solid fraction.
- the pasty compound is preferably chosen from synthetic compounds and compounds of plant origin.
- a pasty compound can be obtained synthetically from starting materials of plant origin.
- linear or branched oligomers homo or copolymers of alkyl (meth) acrylates preferably having a C 8 -C 30 alkyl group,
- homo and copolymeric oligomers of vinyl esters having C 8 -C 30 alkyl groups and Homo and copolymer oligomers of vinyl ethers having alkyl groups
- liposoluble polyethers resulting from the polyetherification between one or more C2-C100, preferably C2-C50, diols,
- esters it is particularly preferred:
- esters of an oligomeric glycerol in particular the diglycerol esters, in particular the adipic acid and glycerol condensates, for which part of the hydroxyl groups of the glycerols have reacted with a mixture of fatty acids such as the acid stearic acid, capric acid, stearic acid and isostearic acid and 12-hydroxystearic acid, especially like those marketed under the trademark Softisan 649 by the company Sasol,
- non-crosslinked polyesters resulting from the polycondensation between a linear or branched C 4 -C 50 dicarboxylic acid or polycarboxylic acid and a C2-C50 diol or polyol,
- esters of diol dimer and diacid dimer if appropriate, esterified on their (their) function (s) alcohol (s) or acid (s) free (s) by acidic radicals or alcohols such as Plandool-G,
- inorganic lipophilic gelling agents there may be mentioned optionally modified clays, such as hectorites modified with a C10 to C22 ammonium chloride, such as hectorite modified with di-stearyl-dimethyl ammonium chloride such as, for example, sold under the name Bentone 38V® by the company ELEMENTIS. It is also possible to mention fumed silica optionally treated with hydrophobic surface, the particle size of which is less than 1 ⁇ . It is indeed possible to chemically modify the surface of the silica, by chemical reaction generating a decrease in the number of silanol groups present on the surface of the silica.
- optionally modified clays such as hectorites modified with a C10 to C22 ammonium chloride, such as hectorite modified with di-stearyl-dimethyl ammonium chloride such as, for example, sold under the name Bentone 38V® by the company ELEMENTIS.
- fumed silica optionally treated with hydrophobic surface, the particle size
- hydrophobic silica is then obtained.
- the hydrophobic groups may be trimethylsiloxyl groups, which are especially obtained by treatment of fumed silica in the presence of hexamethyldisilazane. Silicas thus treated are called “Silica silylate” according to the CTFA (8th edition, 2000).
- silica are for example sold under the references Aerosil R812® by the company Degussa, Cab-O-Sil TS-530® by the company Cabot, dimethylsilyloxyl or polydimethylsiloxane groups, which are especially obtained by treatment of fumed silica in the presence of polydimethylsiloxane or dimethyldichlorosilane.
- Silicas thus treated are called "Silica dimethyl silylate" according to the CTFA (8th edition, 2000). They are for example marketed under the references Aerosil R972®, and Aerosil R974® by the company DEGUSSA, CAB-O-SIL TS-610® and CAB-O-SIL TS-720® by CABOT.
- the hydrophobic fumed silica has in particular a particle size that can be nanometric to micrometric, for example ranging from about 5 to 200 nm.
- Polymeric organic lipophilic gelling agents are, for example, partially or fully crosslinked elastomeric organopolysiloxanes of three-dimensional structure, such as those marketed under the names KSG6®, KSG16® and KSG18® by the company SHIN-ETSU, by Trefil E-505C® and Trefil E-506C® by DOW-CORNING, Gransil SR-CYC®, SR DMF10®, SR-DC556®, SR 5CYC gel®, SR DMF 10 gel® and SR DC 556 gel® by GRANT INDUSTRIES , SF 1204® and JK 1 13® by the company GENERAL ELECTRIC; ethylcellulose such as that sold under the name Ethocel® by Dow Chemical; galactomannans having from one to six, and in particular from two to four, hydroxyl groups per sac, substituted by a saturated or unsaturated alkyl chain, such as guar gum alkylated by C1-C6 alky
- Lipophilic gelling agents that may also be mentioned include polymers having a weight average molecular weight of less than 100,000, comprising a) a polymer backbone having hydrocarbon-based repeating units provided with at least one heteroatom, and optionally b) at least one fatty chain. pendant and / or at least one chain optionally functionalized fatty acid, having from 6 to 120 carbon atoms and being bonded to these hydrocarbon-based units, as described in applications WO-A-02/056847, WO-A-02/47619, the content of which is incorporated as a reference; in particular polyamide resins (especially comprising alkyl groups having from 12 to 22 carbon atoms) such as those described in US-A-5783657 whose contents are incorporated by reference.
- lipophilic gelling agents that can be used in the compositions according to the invention, mention may also be made of dextrin and fatty acid esters, such as dextrin palmitates, in particular such as those sold under the names Rheopearl TL® or Rheopearl KL. ® by the company CHIBA FLOUR. It is also possible to use polyorganosiloxane-type silicone polyamides such as those described in US-A-5,874,069, US-A-5,919,441, US-A-6,051,216 and US-A-5,981,680.
- silicone polymers can belong to the following two families:
- polyorganosiloxanes comprising at least two groups capable of establishing hydrogen interactions, these two groups being located in the polymer chain, and / or
- polyorganosiloxanes comprising at least two groups capable of establishing hydrogen interactions, these two groups being located on grafts or branches.
- compositions of the invention may contain at least one salt or antiperspirant complex.
- the antiperspirant salts or complexes in accordance with the invention are generally chosen from aluminum or zirconium salts or complexes. They are preferably chosen from aluminum halohydrates; aluminum and zirconium halohydrates, complexes of zirconium hydroxychloride and of aluminum hydroxychloride with or without an amino acid such as those described in patent US-3792068.
- aluminum chlorohydrate in activated or non-activated form, aluminum chlorohydrex, chlorohydrex polyethylene glycol aluminum complex, aluminum chlorohydrexpropyleneglycol complex, aluminum dichlorohydrate, aluminum dichlorohydrex complex polyethylene glycol, aluminum complex dichlorohydrex propylene glycol, sesquichlorohydrate aluminum, sesquichlorohydrex aluminum polyethylene glycol complex, sesquichlorohydrex propylene glycol aluminum complex, aluminum sulphate buffered with sodium aluminum lactate.
- aluminum and zirconium salts there may be mentioned in particular aluminum zirconium octachlorohydrate, aluminum zirconium pentachlorohydrate, aluminum zirconium tetrachlorohydrate, aluminum zirconium trichlorohydrate.
- the complexes of zirconium hydroxychloride and aluminum hydroxychloride with an amino acid are generally known under the name ZAG (when the amino acid is glycine).
- ZAG when the amino acid is glycine.
- the antiperspirant salts or complexes may be present in the composition according to the invention in a proportion of approximately 0.5 to 25% by weight relative to the total weight of the composition.
- compositions according to the invention may also contain one or more additional deodorant active agents.
- Deodorant active means any substance capable of masking, absorbing, improving and / or reducing the unpleasant odor resulting from the decomposition of human sweat by bacteria
- the deodorant active agents may be bacteriostatic agents or bactericidal agents acting on the germs of axillary odors, such as 2,4,4'-trichloro-2'-hydroxydiphenyl ether ( ® Triclosan), 2,4-dichloro-2'- hydroxydiphenyl ether, 3 ', 4', 5'-trichlorosalicylanilide, 1 - (3 ', 4'-dichlorophenyl) -3- (4'-chlorophenyl) urea ( ® Triclocarban) or 3,7,1 1 -trimethyldodeca -2, 5,10-trienol (Farnesol ®); quaternary ammonium salts such as cetyltrimethylammonium salts, cetylpyridinium salts, DPTA (1,3-diaminopropanetetraacetic acid), 1,2-decanediol (SYMCLARIOL from Symrise), g
- zinc salts such as zinc salicylate, zinc gluconate, zinc pidolate; zinc sulphate, zinc chloride, zinc lactate, zinc phenolsulfonate; zinc ricinoleate
- salicylic acid and its derivatives such as n-octanoyl-5-salicylic acid
- the deodorant active agents may preferably be present in the compositions according to the invention in weight concentrations ranging from 0.01 to 5% by weight relative to the total weight of the composition. Suspending agents
- suspending agents which are preferably chosen from hydrophobic modified montmorillonite clays, such as hydrophobic modified bentonites or hectorites.
- CTFA name reaction product of bentonite and quaternary ammonium chloride stearalkonium chloride
- the suspending agents are preferably present in amounts ranging from 0.1 to 5% by weight and more preferably from 0.2 to 2% by weight relative to the total weight of the composition.
- compositions according to the invention will contain, in addition, an organic powder.
- organic powder means any solid that is insoluble in the medium at room temperature (25 ° C.).
- Organic powders that may be used in the composition of the invention include, for example, polyamide particles and especially those sold under the names ORGASOL by Atochem; polyethylene powders; microspheres based on acrylic copolymers, such as as those in ethylene glycol dimethacrylate / lauryl methacrylate copolymer sold by Dow Corning under the name Polytrap; polymethyl methacrylate microspheres, sold under the name MICROSPHERE M-100 by the company Matsumoto or under the name COVABEAD LH85 by the company Wackherr; hollow polymethyl methacrylate microspheres (granulometry: 6.5 - 10.5 ⁇ ) sold under the name Ganzpearl GMP 0800 by Ganz Chemical; methyl methacrylate / ethylene glycol dimethacrylate copolymer microbeads (size: 6.5-10.5 ⁇ ) sold under the name Ganzpe
- microspheres marketed under the name MICROPEARL F 80 ED by the company Matsumoto powders of natural organic materials such as starch powders, especially corn starch, wheat or rice, crosslinked or otherwise, such as starch powders crosslinked with octenylsuccinate anhydride, sold under the name DRY -FLO by the company National Starch; silicone resin microbeads such as those sold under the name Tospearl by the company Toshiba Silicone, in particular Tospearl 240; amino acid powders such as the Lauroyllysine powder marketed under the name Amihope LL-1 1 by the company Ajinomoto; the particles of microdispersion of wax, which preferably have average dimensions of less than 1 ⁇ and especially ranging from 0.02 ⁇ to 1 ⁇ , and which consist essentially
- the cosmetic compositions according to the invention may furthermore comprise cosmetic adjuvants chosen from softeners, antioxidants, opacifiers, stabilizers, moisturizing agents, vitamins, bactericides, preservatives, polymers, perfumes and thickeners. , propellants or any other ingredient usually used in cosmetics for this type of application.
- cosmetic adjuvants chosen from softeners, antioxidants, opacifiers, stabilizers, moisturizing agents, vitamins, bactericides, preservatives, polymers, perfumes and thickeners.
- propellants or any other ingredient usually used in cosmetics for this type of application propellants or any other ingredient usually used in cosmetics for this type of application.
- the thickeners may be selected from guar gums and modified or unmodified celluloses such as hydroxypropyl guar gum, cetylhydroxyethylcellulose, silicas such as Bentone Gel MIO sold by the company NL INDUSTRIES or the Veegum Ultra, sold by POLYPLASTIC.
- the thickeners may also be cationic, for example POLYQUATERNIUM-37 sold under the name Salcare SC95 (Polyquaternium-37 (And) Mineral Oil (And) PPG-1 Trideceth-6) or Salcare SC96 (Polyquaternium-37 (And) Propylene Glycol Dicaprylate / Dicaprate (And) PPG-1 -Trideceth-6) or other crosslinked cationic polymer such as for example those of CTFA name Ethylacrylate / Dimethylamino Ethyl Methacrylate Cationic Emulsion Copolymer.
- the amounts of these various constituents that may be present in the composition according to the invention are those conventionally used in compositions for the treatment of perspiration. aerosols
- compositions according to the invention may be further pressurized and packaged in an aerosol device consisting of:
- Propellants generally used in this type of product and well known to those skilled in the art are, for example, dimethyl ether (DME); volatile hydrocarbons such as n-butane, propane, isobutane, and mixtures thereof, optionally with at least one chlorinated and / or fluorinated hydrocarbon; among these are the compounds sold by the company Dupont de Nemours under the names Freon® and Dymel®, and in particular monofluorotrichloromethane, difluorodichloromethane, tetrafluorodichloroethane and 1,1-difluoroethane sold especially under the trade name Dymel 152 A by the company DUPONT.
- DME dimethyl ether
- volatile hydrocarbons such as n-butane, propane, isobutane, and mixtures thereof, optionally with at least one chlorinated and / or fluorinated hydrocarbon
- Freon® and Dymel® the compounds sold by the company Dupont de Nemours under the names
- compositions containing the perlite particles as defined above and the propellant (s) may be in the same compartment or in different compartments in the aerosol container.
- concentration of propellant generally varies from 5 to 95% by weight pressurized and more preferably from 50 to 85% by weight relative to the total weight of the pressurized composition.
- the dispensing means which forms part of the aerosol device, is generally constituted by a dispensing valve controlled by a dispensing head, it same comprising a nozzle by which the aerosol composition is vaporized.
- the container containing the pressurized composition may be opaque or transparent. It may be glass, polymeric material or metal, optionally covered with a layer of protective varnish.
- the invention also relates to a cosmetic process for treating body odors, in particular axillary odors or foot odors, which consists in applying to the keratin materials a composition as described above (and of course containing the combination according to US Pat. 'invention).
- This process is especially used to treat axillary odors caused by C. xerosis bacteria or foot smells caused by B. linens.
- Destroying or inhibiting odor-causing bacteria, especially axillary odors or feet, with a bacteriocin while using a prebiotic that confers a selective advantage on the beneficial germs of the skin, can reduce and / or to remove or prevent the development of body odor without destroying the ecosystem of the treated area.
- a composition according to the invention comprising a class II plantaricin, more particularly a class II plantaricin chosen from plantaricin A, plantaricin C19 and plantaricin, is applied. .
- composition according to the invention comprising a prebiotic chosen from a xylooligosaccharide and an isomalto-oligosaccharide is applied.
- the cosmetic process for treating body odors according to the invention comprises the application of a composition comprising
- bacteriocin selected from plantaricin A, plantaricin C19 and plantaricin 149, and
- composition for the care and / or cleaning of the skin chosen from a xylooligosaccharide and an isomalto-oligosaccharide.
- composition according to the invention contains a physiologically acceptable medium as defined above.
- the composition has a pH preferably close to that of the skin, between 4 and 7.
- compositions according to the invention may be in any of the galenical forms conventionally used for topical application and especially in the form of aqueous, hydroalcoholic, oil-in-water (O / W) or water-in-oil ( E / H) or multiple (triple: W / O / W or W / E / H), aqueous gels, or dispersions of a fatty phase in an aqueous phase using spherules, or lipid vesicles of ionic and / or nonionic type (liposomes, niosomes, oleosomes), or thin films.
- These compositions are prepared according to the usual methods.
- composition according to the invention may be more or less fluid and have the appearance of a white or colored cream, an ointment, a milk, a lotion, a serum, a paste, a foaming gel, a treatment, a tonic or a mousse.
- the composition according to the invention is formulated in the form of a foaming gel. It can optionally be applied to the skin in the form of an aerosol. It can also be in solid form, and for example in the form of a stick. It may be a care composition, an exfoliating composition or cleaning or scrubbing, or a shaving composition such as a shaving foam.
- composition used according to the invention comprises an oily phase
- it preferably contains at least one oil. It may also contain other fatty substances.
- oils that can be used in the composition of the invention, mention may be made for example of:
- hydrocarbon oils of animal origin such as perhydrosqualene
- hydrocarbon-based oils of vegetable origin such as liquid triglycerides of fatty acids containing from 4 to 10 carbon atoms, for instance triglycerides of heptanoic or octanoic acids or, for example, sunflower, corn or soybean oils, squash, grape seed, sesame, hazelnut, apricot, macadamia, arara, sunflower, castor oil, avocado, triglycerides of caprylic / capric acids such as those sold by Stearineries Dubois or those sold under the names Miglyol 810, 812 and 818 by the company Dynamit Nobel, jojoba oil, shea butter oil;
- esters and synthetic ethers in particular of fatty acids, such as the oils of formulas R'COOR 2 and R'OR 2 in which R 'represents the residue of a fatty acid comprising from 8 to 29 carbon atoms, and R 2 represents a hydrocarbon chain, branched or unbranched, containing from 3 to 30 carbon atoms, for example Purcellin oil, isononyl isononanoate, isopropyl myristate or 2-ethyl palmitate; hexyl, octyl-2-dodecyl stearate, octyl-2-dodecyl erucate, isostearyl isostearate; hydroxylated esters such as isostearyl lactate, octyl hydroxystearate, octyldodecyl hydroxystearate, diisostearyl malate, triisocetyl citrate, heptanoates, octan
- linear or branched hydrocarbons of mineral or synthetic origin such as paraffin oils, volatile or not, and their derivatives, petroleum jelly, polydecenes, hydrogenated polyisobutene such as sesam oil;
- fatty alcohols having from 8 to 26 carbon atoms such as cetyl alcohol, stearyl alcohol and their mixture (cetylstearyl alcohol), octyldodecanol, 2-butyloctanol, 2-hexyldecanol, 2-undecylpentadecanol, oleic alcohol or linoleic alcohol;
- silicone oils such as volatile or non-volatile polymethylsiloxanes (PDMS) with a linear or cyclic silicone chain, which are liquid or pasty at room temperature, in particular cyclopolydimethylsiloxanes (cyclomethicones) such as cyclohexasiloxane; polydimethylsiloxanes containing alkyl groups, alkoxy or phenyl, during or at the end of the silicone chain, groups having from 2 to 24 carbon atoms; phenyl silicones such as phenyltrimethicones, phenyldimethicones, phenyltrimethylsiloxydiphenylsiloxanes, diphenyl-dimethicones, diphenylmethyldiphenyltrisiloxanes, 2-phenylethyltrimethylsiloxysilicates, and polymethylphenylsiloxanes;
- PDMS volatile or non-volatile polymethylsiloxanes
- hydrocarbon oil is meant in the list of oils mentioned above, any oil predominantly comprising carbon and hydrogen atoms, and optionally ester, ether, fluoro, carboxylic acid and / or alcohol groups.
- the other fatty substances that may be present in the oily phase are, for example, fatty acids containing from 8 to 30 carbon atoms, such as stearic acid, lauric acid, palmitic acid and oleic acid; waxes such as lanolin, beeswax, carnauba or candelilla wax, paraffin waxes, lignite waxes or microcrystalline waxes, ceresin or ozokerite, synthetic waxes such as polyethylene waxes, waxes Fischer-Tropsch; silicone resins such as trifluoromethyl-C1-4-alkyldimethicone and trifluoropropyldimethicone; and silicone elastomers such as the products sold under the names KSG by the company Shin-Etsu, under the names "Trefil", "BY29” or "EPSX” by Dow Corning or under the names "Gransil” by the company Grant industries.
- fatty acids containing from 8 to 30 carbon atoms such as stea
- the composition according to the invention is a water-in-oil (W / O) or oil-in-water (O / W) emulsion.
- W / O water-in-oil
- O / W oil-in-water
- the proportion of the oily phase of the emulsion may range from 5 to 80% by weight, and preferably from 5 to 50% by weight relative to the total weight of the composition.
- the emulsions generally contain at least one emulsifier chosen from amphoteric, anionic, cationic or nonionic emulsifiers, used alone. or in mixture, and optionally a co-emulsifier.
- the emulsifiers are suitably selected according to the emulsion to be obtained (W / O or O / W).
- the emulsifier and the co-emulsifier are generally present in the composition, in a proportion ranging from 0.3 to 30% by weight, and preferably from 0.5 to 20% by weight relative to the total weight of the composition.
- emulsifiers examples include dimethicone copolyols such as the mixture of cyclomethicone and dimethicone copolyol, sold under the name "DC 5225 C" by Dow Corning, and alkyl dimethicone copolyols such as Laurylmethicone copolyol sold under the name Dow Corning 5200 Formulation Aid by Dow Corning and Cetyl dimethicone copolyol sold under the name Abil EM 90 R by Goldschmidt.
- dimethicone copolyols such as the mixture of cyclomethicone and dimethicone copolyol, sold under the name "DC 5225 C" by Dow Corning
- alkyl dimethicone copolyols such as Laurylmethicone copolyol sold under the name Dow Corning 5200 Formulation Aid by Dow Corning and Cetyl dimethicone copolyol sold under the name Abil EM 90 R by Goldschmid
- a crosslinked solid elastomeric organopolysiloxane comprising at least one oxyalkylene group such as those obtained according to the procedure of Examples 3, 4 and 8 of US Pat. No. 5,412,004 and examples US-A-5,81 1,487, in particular the product of Example 3 (synthetic example) of US-A-5,412,004, and such as that sold under the reference KSG 21 by Shin Etsu.
- emulsifiers for example, of nonionic emulsifiers such as oxyalkylenated (more particularly polyoxyethylenated) fatty acid esters of glycerol; oxyalkylenated fatty acid and sorbitan esters; oxyalkylenated fatty acid esters (oxyethylenated and / or oxypropylenated); oxyalkylenated fatty alcohol ethers (oxyethylenated and / or oxypropylenated); sugar esters such as sucrose stearate; and mixtures thereof such as the mixture of glyceryl stearate and PEG-40 stearate.
- nonionic emulsifiers such as oxyalkylenated (more particularly polyoxyethylenated) fatty acid esters of glycerol; oxyalkylenated fatty acid and sorbitan esters; oxyalkylenated fatty acid esters (oxyethyl
- the composition for the care and / or cleaning of the skin according to the invention may comprise at least one additional skin care ingredient and / or active ingredient, in particular for the treatment of oily skin.
- additional active ingredients and ingredients described in patent applications WO2004 / 105736 and DE10324567, incorporated herein by reference, among which depigmenting agents, preservatives, antimicrobial agents, antiperspirants, metal chelators, filters UV, hydrolysed proteins, antioxidants, vitamins, anti-inflammatory agents, anti-irritant agents, moisturizing agents, plant extracts, cosmetic adjuvants, and mixtures thereof.
- vitamin C and its derivatives there may be mentioned in particular vitamin C and its derivatives and in particular the vit CG, CP and 3-0 ethyl vitamin C, alpha and beta arbutin, lucinol and its derivatives, kojic acid, resorcinol and its derivatives, tranexamic acid and its derivatives, gentisic acid, homogentisate, methyl gentisate or homogentisate, dioic acid, D calcium panthetine sulfonate, lipoic acid, ellagic acid, vitamin B3, tranexamic acid, lipoic acid, linoleic acid and its derivatives, ceramides and their homologs, plant derivatives such as chamomile, bearberry, the family of aloe (vera, ferox, bardensis) , of mulberry tree, skullcap, without this list being exhaustive.
- Humidifying or moisturizing agents include glycerol and its derivatives, urea and its derivatives, in particular Hydrovance marketed by National Starch, lactic acids, hyaluronic acid, AHAs, BHAs and sodium pidolate. , xylitol, serine, sodium lactate, ectoin and its derivatives, chitosan and its derivatives, collagen, plankton, imperata cylindra extract marketed under the name Moist 24 by Sederma.
- additional ingredients and / or active agents for the care of oily skin, in particular with acne prone skin, and / or acne skin, and / or hyperseborrhoeic skin, such as mattifying agents, abrasive fillers or exfoliating agents, desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebum-regulating agents, antioxidants, cicatrizing agents, astringent agents, and mixtures thereof. More particularly, it will be chosen to incorporate into the composition according to the invention at least one additional skin care ingredient and / or active agent chosen from depigmenting agents, metal chelators, hydrolysed proteins, vitamins and plant extracts.
- mattifying agents mattifying agents, abrasive fillers or exfoliating agents, desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebo-regulating agents, antioxidants, cicatrizing agents, astringent agents, and their agents. mixtures.
- the composition may be a care composition and / or cleaning and / or makeup, intended to be rinsed or not rinsed. Preferably, it will be a non-rinsed skincare composition. When the composition comprises exfoliating agents or abrasive fillers, it will preferably be rinsed.
- matting agent agents intended to make the skin visibly duller, less glossy.
- the matting effect of the agent and / or of the composition containing it can in particular be evaluated using a gonioreflectometer, by measuring the ratio R between the specular reflection and the diffuse reflection.
- a value of R less than or equal to 2 generally reflects a matting effect.
- the matting agent may especially be chosen from a rice starch or a maize starch, kaolinite, silicas, talc, an extract of pumpkin seeds, cellulose microbeads, vegetable fibers, synthetic fibers, in particular polyamides, microspheres of expanded acrylic copolymers, polyamide powders, silica powders, polytetrafluoroethylene powders, silicone resin powders, acrylic copolymer powders, wax powders, polyethylene powders, powders crosslinked organopolysiloxane elastomer coated with silicone resin, talc / titanium dioxide / alumina / silica composite powders, amorphous mixed silicate powders, acrylic polymer powders, silicate particles and especially mixed silicate particles, and mixtures thereof.
- mattifying agents examples include:
- starch from rice or maize, in particular starch octenyl succinate aluminum sold under the name Dry Flo® by the company National Starch,
- - fibers such as silk, cotton, wool, flax, cellulose fibers extracted in particular from wood, vegetables or algae, polyamide (Nylon®), modified cellulose, poly-p-phenylene terephtamide, acrylic, polyolefin, glass, silica, aramid, carbon, Teflon®, insoluble collagen, polyesters, polyvinyl chloride or vinylidene, polyvinyl alcohol, polyacrylonitrile, chitosan , polyurethane, polyethylene phthalate, fibers formed from a mixture of polymers, synthetic resorbable fibers, and mixtures thereof described in patent application L'OREAL EP 1 151 742;
- microspheres of expanded acrylic copolymers such as those sold by Expancel under the names Expancel 551®,
- Polyamide powders such as, for example, the Atofina-type Orgasol Nylon 12 particles having a mean size of 10 microns and a refractive index 1, 54,
- silica powders such as Silica beads SB150 from
- polytetrafluoroethylene powders such as Clariant ceridust 9205F PTFE of average size 8 microns and refractive index 1, 36, silicone resin powders such as GE Silicone Silicon resin Tospearl 145A having a mean size of 4.5 microns and a refractive index of 1.41,
- acrylic copolymers in particular of methyl poly (meth) acrylate, such as PMMA Jurymer MBI particles of Nihon Junyoki having an average size of 8 microns and a refractive index of 1.49, or Micropearl M100® and F80 particles; ED® from Matsumoto Yushi-Seiyaku;
- wax powders such as Paraffin wax microease particles 1 14S of micropowders of average size 7 microns and refractive index 1, 54,
- polyethylene powders especially comprising at least one ethylene / acrylic acid copolymer, and in particular consisting of ethylene / acrylic acid copolymers such as the Sumitomo Flobeads EA 209 particles (of average size 10 microns and refractive index 1, 48 )
- elastomeric crosslinked organopolysiloxane powders coated with silicone resin in particular with silsesquioxane resin, as described, for example, in US Pat. No. 5,538,793.
- Such elastomer powders are sold under the names "KSP-100” and "KSP-100". -101 ",” KSP-102 “,” KSP-103 “,” KSP-104 “,” KSP-105 "by the company SHIN ETSU, and
- talc / titanium dioxide / alumina / silica composite powders such as those sold under the name Coverleaf AR-80 by Catalyst & chemicals,
- silica powders such as, for example, the porous silica microspheres sold under the name "Silica Beads SB-700" marketed by the company MYOSHI, the "SUNSPHERE® H51", “SUNSPHERE® H33", “SUNSPHERE® H53” marketed by ASAHI GLASS; amorphous silica microspheres coated with polydimethylsiloxane sold under the name "SA Sunsphere® H-33” and “SA Sunsphere® H-53” sold by ASAHI GLASS; amorphous mixed silicate powders, in particular aluminum and magnesium, such as, for example, that marketed under the name "NEUSILIN UFL2" by the company Sumitomo.
- polyamide powders for example "ORGASOL® 4000” marketed by the company ATOCHEM, and
- acrylic polymer powders in particular polymethyl methacrylate, for example "COVABEAD® LH85” marketed by WACKHERR; polymethyl methacrylate / ethylene glycol dimethacrylate, such as, for example, “DOW CORNING 5640 MICROSPONGE® SKIN OIL ADSORBER” marketed by Dow Corning, or “Ganzpearl® GMP-0820” marketed by Ganz Chemical; allyl polymethacrylate / ethylene glycol dimethacrylate, such as for example "POLY-PORE® L200" or “POLY-PORE® E200” marketed by AMCOL; ethylene glycol dimethacrylate / lauryl methacrylate copolymer, such as, for example, "POLYTRAP® 6603" marketed from Dow Corning;
- silicate particles such as alumina silicate
- mixed silicate particles such as:
- aluminum magnesium silicate particles such as saponite or magnesium aluminum silicate, hydrated with a sodium sulphate sold under the trade name Sumecton® by the company Kunimine;
- an extract of pumpkin seeds a rice or maize starch, kaolinite, silicas, talc, polyamide powders, polyethylene powders and copolymer powders. acrylics, microspheres of expanded acrylic copolymers, silicone resin microbeads, mixed silicate particles and mixtures thereof. Abrasive loads or exfoliating agents
- exfoliating agents that may be used in rinsed compositions according to the invention, mention may be made, for example, of exfoliant or scrubbing particles of mineral, vegetable or organic origin.
- exfoliant or scrubbing particles of mineral, vegetable or organic origin.
- beads or polyethylene powder, nylon powder, polyvinyl chloride powder, pumice, ground apricot kernels or nut shells can be used. sawdust, glass beads, alumina, and mixtures thereof.
- Exfogreen® from Solabia (bamboo extract), strawberry akenes (Greentech strawberry Akancies), peach kernel powder, apricot kernel powder, and finally the field of vegetable powders with abrasive effect include the cranberry powder.
- peach kernel powder As abrasive fillers or preferred exfoliation agents according to the invention, mention may be made of peach kernel powder, apricot kernel powder, cranberry kernel powder, strawberry achene extracts, bamboo extracts.
- associated active agents that can be used in the compositions of the invention, particular mention may be made of desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebo-regulating agents, antioxidants and mixtures thereof.
- the composition according to the invention will comprise as additional active agent at least one sebum-regulating agent.
- the composition may further optionally comprise an antimicrobial agent.
- compositions More preferably it may further comprise a desquamating agent. And to further increase the efficacy and / or the tolerance of said compositions, it may additionally add soothing or anti-inflammatory agents, antioxidants, cicatrizing agents, astringent agents, and mixtures thereof.
- sibo-regulating or anti-seborrhoeic agents is meant in particular agents capable of regulating the activity of the sebaceous glands.
- retinoic acid benzoyl peroxide, sulfur, vitamin B6 (or pyridoxine), selenium chloride, sea fennel;
- capryloyl glycine, sarcosine and cinnamomum zeylanicum extract marketed in particular by the company SEPPIC under the trademark Sepicontrol® A5;
- zinc salicylate such as zinc pyrrolidone carboxylate (or zinc pidolate), zinc lactate, zinc aspartate, zinc carboxylate, zinc cysteate;
- copper salts such as copper sulphate or copper pyrrolidone carboxylate
- algae extracts laminaria saccharina such as that sold under the name Phlorogine® by the company Biotechmarine;
- linseed extract such as that sold under the name Linumine® by the company Lucas Meyer;
- Phellodendron extracts such as those sold under the name Phellodendron extract BG by the company Maruzen or Oubaku liquid B by the company Ichimaru Pharcos;
- clove extract such as that sold under the name Clove Extract Powder by the company Maruzen;
- argan oil such as that sold under the name Lipofructyl® by Serobiological Laboratories
- lactic protein filtrates such as that sold under the name Normaseb® by the company Sederma
- algae laminaria extracts such as that sold under the name Laminarghane® by the company Biotechmarine;
- cane sugar extracts such as the product sold under the name Policasonol® by Sabinsa;
- oligosaccharides of algae laminaria digitata such as that sold under the name Phycosaccharide AC by the company Codif;
- sulphonated shale oil such as that sold under the name Ichtyol Pale by the company Ichthyol;
- sebacic acid in particular sold in the form of a sodium polyacrylate gel under the name Sebosoft by Sederma;
- glucomannans extracted from konjac tubers and modified with alkylsulphonate chains such as the one sold under the name Biopol Beta by the company Arch Chemical;
- Extracts of Sophora angustifolia such as those sold under the name Sophora powder or Sophora extract by the company Bioland;
- glycine grafted on an undecylenic chain such as that sold under the name Lipacide UG OR by the company Seppic;
- trialkyl citrate (C12-C13) sold under the name COSMACOL® ECI by the company Sasol
- Ci 4 -C 5 trialkyl citrate
- 10-hydroxydecanoic acid and in particular mixtures of 10-hydroxydecanoic acid, of sebacic acid and of 1,1-decandiol, such as that sold under the name Acnacidol® BG by the company Vincience; specific PPAR- ⁇ activators, such as those described in application WO 2005/053632;
- sapogenins or plant extracts containing them in particular extracts of Dioscorea rich in diosgenin;
- seboregulating agents that can be used according to the invention, mention may be made of:
- zinc salts such as zinc pyrrolidone carboxylate (or zinc pidolate), zinc lactate, zinc aspartate, zinc carboxylate, zinc cysteate;
- copper salts such as copper sulphate or copper pyrrolidone carboxylate
- algae extracts laminaria saccharina such as that sold under the name Phlorogine by the company Biotechmarine;
- sapogenins or plant extracts containing them in particular extracts of Dioscorea rich in diosgenin,
- antiperspirants such as: aluminum and / or zirconium salts; complexes of zirconium hydroxychloride and hydroxychloride of aluminum with an amino acid such as those described in US-3792068 commonly known as "ZAG complexes".
- ZAG complexes Such complexes are generally known by the name ZAG (when the amino acid is Glycine).
- the ZAG complexes usually have an Al / Zr quotient ranging from about 1.67 to 12.5 and a Metal / Cl quotient ranging from about 0.73 to 1.93.
- aluminum salts mention may be made of aluminum chlorohydrate, chlorohydroxyl aluminum, chlorohydroxyl aluminum PEG, aluminum chlorohydrex PG, aluminum dichlorohydrate, aluminum dichlorohydrex PEG, aluminum dichlorohydrex PG, aluminum sesquichlorohydrate, aluminum sesquichlorohydrex PEG, aluminum sesquichlorohydrex PG, alum salts, aluminum sulfate, aluminum zirconium octachlorohydrate, aluminum zirconium pentachlorohydrate, aluminum zirconium tetrachlorohydrate, aluminum zirconium trichlorohydrate and more particularly the aluminum hydrochloride marketed by the company REHEIS under the name MICRODRY ALUMINUM CHLOROHYDRATE or by the company GUILINI CHEMIE under the name ALOXICOLL PF 40.
- Aluminum and zirconium salts are for example that marketed by the company REHEIS under the name REACH AZP-908-SUF, activated aluminum salt for example that marketed by the company REHEIS s or the REACH 103 denomination or by WESTWOOD under the name WESTCHLOR 200.
- deodorant active agents also include zinc salts such as zinc sulphate, zinc chloride, zinc lactate, zinc phenolsulphonate; chlorhexidine and salts; diglycerol monocaprate, diglycerol monolaurate, glycerol monolaurate; salts of polyhexamethylene biguanide.
- zinc salts such as zinc sulphate, zinc chloride, zinc lactate, zinc phenolsulphonate; chlorhexidine and salts; diglycerol monocaprate, diglycerol monolaurate, glycerol monolaurate; salts of polyhexamethylene biguanide.
- anti-microbial agents agents having effects on the specific flora of oily skin, such as, for example, P acnes. These effects can be either bactericidal or bacterial anti-adhesion (prevents and / or reduces the adhesion of microorganisms) or acting on the biofilm of bacteria to prevent their multiplication.
- a hop cone extract HOP CO2-TO extract from Flavex
- a Millepertius extract ST John's Wort CO2-TO Flavex extract
- Asian acid extracts of scutellaria baicolensis roots as in Zurogin BMB-CF
- piroctone olamine citrollic acid
- sperillic acid ethylhexylglycerine
- caprylate / gluceryl caprate (ABITEC Capmul)
- calcium sodium phosphosilicate as Bioactive Schott's glasspowder
- Schott's Actysse first BG Ciba's silicon oxides
- Metashines salver derivatives
- phytanetriol and its derivatives as described in patent application EP 1 529 523, vegetable oils such as wheat germ oil , calendula oil, castor oil, olive oil, avocado oil, sweet almond oil, peanut oil, jojoba oil, sesame oil, apricot kernel oil, sunflower oil, macadamia oil, described in patent EP 1 133 979, or other fatty substances such as cocoamphodiacetate disodium, cocoate of oxyethylenated glyceryl (7 EO), Poloxamers, hexadecenylsuccinate 18, octoxyglyceryl palmitate, octoxyglyceryl behenate, dioctyl adipate, PPG-15 stearyl ether, C12-C13 di-alcohol tartrate branched derivatives described in EP 1 129 694.
- vegetable oils such as wheat germ oil , calendula oil, castor oil, olive oil, avocado oil, sweet almond oil, peanut oil, jo
- P acnnes In particular with respect to the propagation of P acnnes, mention may be made of pentylene glycol, nylon-66 (polyamide 66 fibers), rice bran oil, Celvol 540 PV alcohol (Polyvinyl alcohol 72962) , Akorex L from Karlshamns, fructose derivatives.
- compositions of the invention will use an agent chosen from caprylyl glycol, zinc derivatives including zinc pidolate (Zincidone® from Solabia), copper derivatives, octoglycerine or octoxyglycerine, and 10-hydroxy-2-decanoic acid, and mixtures thereof.
- zinc derivatives including zinc pidolate (Zincidone® from Solabia)
- copper derivatives octoglycerine or octoxyglycerine
- 10-hydroxy-2-decanoic acid and mixtures thereof.
- treating agent any compound capable of acting:
- ⁇ -hydroxy acids in particular salicylic acid and its derivatives (including n-octanoyl-5-salicylic acid); ⁇ -hydroxy acids, such as glycolic, citric, lactic, tartaric, malic or mandelic acids; urea; gentisic acid and its derivatives; oligofucoses; cinnamic acid; Saphora japonica extract; resveratrol and certain jasmonic acid derivatives; or on the enzymes involved in the desquamation or degradation of corneodesmosomes, glycosidases, stratum corneum chymotryptic enzyme (SCCE) or even other proteases (trypsin, chymotrypsin-like).
- SCCE stratum corneum chymotryptic enzyme
- aminosulfonic compounds and in particular (N-2-hydroxyethylpiperazine-N-2-ethane) sulfonic acid (HEPES); 2-oxothiazolidine-4-carboxylic acid derivatives (procysteine); glycine-type alpha amino acid derivatives (as described in EP-0 852 949, as well as the sodium methyl glycine diacetate marketed by BASF under the trade name TRILON M); honey ; sugar derivatives such as O-octanoyl-6-D-maltose and N-acetyl glucosamine.
- HEP-0 852 949 2-oxothiazolidine-4-carboxylic acid derivatives
- TRILON M glycine-type alpha amino acid derivatives
- honey sugar derivatives such as O-octanoyl-6-D-maltose and N-acetyl glucosamine.
- desquamating agents that can be used in the composition according to the invention include oligofructoses, EDTA and its derivatives, laminaria extract, o-linoleyl-6D-glucose, (3-hydroxy-2-pentylcyclopentyl) acetic acid, glycerol trilactate, O-octanyl-6'-D-maltose, S carboxymethyl cysteine, the silicon derivatives of salicylate as in patent EP 0 796 861, oligofucase as in patent EP 0 218 200, salts 5-acyl salicylic acid, active agents having effects on transglutaminase as in patent EP 0 899 330, jasmonic acid and derivatives as in patent applications EP 1 333 022 and EP 1 333 021, Exfolactive® from Silab (ficus opuntia indica flower extract), Soypon O® from Kawaken fine chemicals (sodium cocoyl sarcosinate)
- Preferred desquamating agents include beta-hydroxy acids such as n-octanoyl-5-salicylic acid; urea; glycolic, citric, lactic, tartaric, malic or mandelic acids; (N-2-hydroxyethylpiperazine-N-2-ethane) sulfonic acid (HEPES); Saphora japonica extract; honey ; N-acetyl glucosamine; sodium methyl glycine diacetate, and mixtures thereof.
- beta-hydroxy acids such as n-octanoyl-5-salicylic acid; urea; glycolic, citric, lactic, tartaric, malic or mandelic acids; (N-2-hydroxyethylpiperazine-N-2-ethane) sulfonic acid (HEPES); Saphora japonica extract; honey ; N-acetyl glucosamine; sodium methyl glycine diacetate, and mixtures thereof.
- a desquamating agent chosen from n-octanoyl 5-salicylic acid; urea; (N-2-hydroxyethylpiperazine-N-2-ethane) sulfonic acid (HEPES); Saphora japonica extract; honey ; Nacetyl glucosamine; sodium methyl glycine diacetate, and mixtures thereof.
- soothing agents that may be used in the composition according to the invention, mention may be made of: procyannidol oligomers, vitamins E, C, B5, B3, dextran sulphate, caffeine and its derivatives, pentacyclic triterpenes and plant extracts containing them, b-glycyrrhetinic acid and its salts or derivatives (stearyl glycyrrhetate, 3-stearoyloxy glycyrrhetic acid, glycyrrhetinic acid monoglucuronide) and the plants containing it (eg Glycyrrhiza glabra), oleanolic acid and its salts, ursolic acid and its salts, boswellic acid and its salts, betulinic acid and its salts, an extract of Paeonia suffruticosa and / or lactiflora, the phycosshacharides of Codif, an extract of Laminaria saccharina
- Anti-inflammatory agents chosen from a rose extract, a clove extract, dextran as in Vincience's Modulene®, a mint extract such as Silab's Calmiskin®, a mixture of an extract of waterlily alba and sodium palmitoylproline such as Seppic Seppicalm VG®, anise derivatives, an extract of Paeonia suffruticosa and / or lactiflora, and mixtures thereof.
- Anti-inflammatory agents chosen from a rose extract, a clove extract, dextran as in Vincience'
- anti-inflammatory agents mentioned in the applications WO2004 / 105736 and DE10324567, incorporated in the present invention by reference.
- antiinflammatory agents include cortisone, hydrocortisone, indomethacin, betamethasone, azealic acid, acetaminophen, diclofenac, clobetasol propionate and mixtures thereof.
- azelaic acid mention will be made of azelaic acid.
- Antioxidants By this expression is meant to define the agents having an antioxidant activity (which prevent the oxidation of squalene and the formation of comedones).
- tocopherol and its esters there may be mentioned in particular tocopherol and its esters, in particular tocopherol acetate; BHT and BHA.
- polyphenols such as polyphenols, tannic acid, epoigallocathechins and natural extracts containing them, anthocyanins, rosemary extracts, olive leaf extracts, green tea, resveratrol and its derivatives, Pycnogenol ergothineine, N acetylcysteine, biotin, chelants, idebenone, plant extracts Pronital Proniten Bioprotect TM from Provital, antiradicals such as vitamin E, coenzyme Q10, bioflavonoids, SOD, phytantriol, lignans, melatonin, pidolates, glutathione.
- cicatrizing agents mention may in particular be made of:
- Preferred healing agents according to the invention are tamanu oil, sodium acexamate, honey extracts, horsetail extracts, helichryse extracts, and mixtures thereof.
- Astringent agents By “astringent agents” is meant according to the invention agents for controlling the dilation of the sebaceous follicles.
- Preferred astringent agents according to the invention are scutellaria extract, ulmary extract, close-up queen extract, gentian extract, burdock extract and mixtures thereof. According to a particular embodiment of the invention, it will be possible to add to these ingredients and / or active agents for the treatment of oily skin other agents intended to embellish the appearance and / or the texture of the skin. In particular, it will be possible to add, in the compositions of the invention, active agents used to prevent and / or treat the signs of skin aging.
- the additional active ingredient (s) and / or ingredient (s) used in the composition according to the invention may represent from 0.0001 to 20%, preferably from 0.01 to 10% and better still from 0.01 to 5% by weight relative to to the total weight of the composition.
- the composition according to the invention is intended for the care and / or the treatment of persons having a phototype skin of III to VI.
- skin imperfections of oily skin may be more or less visible depending on the phototype of the subjects, especially on the skins of subjects to high phototype, ie skin of subjects with phototype III to VI, defined according to the Fitzpatrick classification which is based on the reactivity of the skin to the effects of solar radiation:
- compositions according to the invention are preferably cosmetic compositions for the care of oily skin.
- compositions are intended for topical application on the face and / or the body.
- the composition is applied to the areas of the face or the forehead having a gloss of the skin. It may also be shaving compositions or aftershave, especially for men.
- the cosmetic and / or dermatological composition according to the invention may also contain adjuvants which are customary in the cosmetic or dermatological field, such as hydrophilic or lipophilic gelling agents, preservatives, solvents, perfumes, fillers, UV filters and bactericides. , odor absorbers, dyestuffs, plant extracts, salts, antioxidants, basic agents, acids, nonionic, anionic, cationic surfactants.
- adjuvants which are customary in the cosmetic or dermatological field, such as hydrophilic or lipophilic gelling agents, preservatives, solvents, perfumes, fillers, UV filters and bactericides. , odor absorbers, dyestuffs, plant extracts, salts, antioxidants, basic agents, acids, nonionic, anionic, cationic surfactants.
- these various adjuvants are those conventionally used in the field under consideration, and for example from 0.01 to 20% of the total weight of the composition.
- These adjuvants depending on their nature, can be introduced into the fatty phase, into the aqueous phase and / or into the lipid vesicles.
- fillers which can be used in the composition of the invention, mention may be made, for example, besides the pigments, the silica powder; talcum; polyamide particles and especially those sold under the name ORGASOL by the Atochem company; polyethylene powders; micro-spheres based on acrylic copolymers, such as those made of ethylene glycol dimethacrylate / lauryl methacrylate copolymer sold by Dow Corning under the name Polytrap; expanded powders such as hollow microspheres and in particular the microspheres sold under the name Expancel by the company Kemanord Plast or under the name Micropearl F 80 ED by the company Matsumoto; silicone resin microbeads such as those sold under the name Tospearl by the company Toshiba Silicone; and their mixtures.
- These fillers may be present in amounts ranging from 0 to 20% by weight and preferably from 1 to 10% by weight relative to the total weight of the composition.
- hydrophilic or lipophilic gelling agents there may be mentioned in particular carbopol, luvigel, Hostacerin AMPS, Simulgel, Sepigel, xanthan, guar, cellulose gums, alginates and mixtures thereof. We can also mention hectorites.
- Antioxidants include polyphenols, tannic acid, epoigallocathechins and natural extracts containing them, anthocyanins, rosemary extracts, olive leaf extracts, green tea, resveratrol and its derivatives. derivatives, Pycnogenol, ergothineine, N acetylcysteine, biotin, chelants, idebenone, plant extracts Pronital Proniten Bioprotect TM from Provital, antiradicals such as vitamin E, coenzyme Q10, bioflavonoids , SOD, phytantriol, lignans, melatonin, pidolates, glutathione.
- the composition used according to the invention contains at least one UV filter (or sunscreen) which may be a chemical filter or a physical filter or a mixture of such filters.
- UV filter or sunscreen
- families the names correspond to the CTFA nomenclature of the filters: anthranilates, in particular menthyl anthranilate; benzophenones, in particular benzophenone-1, benzophenone-3, benzophenone-5, benzophenone-6, benzophenone-8, benzophenone-9, benzophenone-12, and preferentially benzophenone-2 (Oxybenzone), or benzophenone-4 (Uvinul MS40 available from BASF); benzylidenecamphers, in particular 3-benzylidene camphor, benzylidenecamphosulfonic acid, camphor benzalkonium methosulphate, polyacrylamidomethylbenzylidene camphor, terephthalylidene di
- benzimidazoles in particular benzimidazilate (Neo Heliopan AP available from Haarmann and Reimer), or phenylbenzimidazole sulfonic acid (Eusolex 232 available from Merck); benzotriazoles, in particular trisiloxane drometzol, or methylenebisbenzotriazolyltetramethylbutylphenol (Tinosorb M available from Ciba); cinnamates, in particular cinoxate, DEA methoxycinnamate, diisopropyl methylcinnamate, dimethoxycinnamate glyceryl ethylhexanoate, isopropyl methoxycinnamate, isoamyl cinnamate, and preferably ethocrylene (Uvinul N35 available from BASF), octyl methoxycinnamate (Parsol MCX available from Hoffmann La Roche), or octocrylene
- the amount of filters depends on the desired end use. It may range, for example, from 1 to 30% by weight and better still from 2 to 15% by weight relative to the total weight of the composition.
- compositions according to the invention may be applied directly to the skin or, alternatively, to occlusive or non-occlusive type cosmetic supports intended to be applied in a localized manner to the skin.
- non-limiting cosmetic media there may be mentioned a patch, a wipe, a roll-on and a pen.
- the support may be an occlusive support.
- the support consists of a thermoplastic material chosen from high and low density polyethylenes, polypropylenes, polyvinyl chlorides, copolymers of ethylene and vinyl acetate, polyesters and polyurethanes. , or a complex of such materials. These materials may also be present in laminated form with at least one sheet of metal such as aluminum foil.
- the support layer may be of any suitable thickness which will provide the desired support and protection functions.
- the thickness of the support layer is from about 20 ⁇ m to about 1.5 mm.
- the support layer is sufficiently flexible so as to perfectly fit the profile of the skin, and not to cause the user, a feeling of discomfort.
- the carrier is non-occlusive. In the latter case, it is advantageous to use a support consisting of a paper, a porous or perforated thermoplastic material, a woven fabric, a nonwoven fabric or a perforated nonwoven fabric.
- compositions according to the invention may be combined with orally administered compositions containing cosmetic active agents with a beneficial effect on the appearance of the skin.
- the invention also relates to a composition as described above, for the care and / or cleaning of sensitive skin, irritated, or having eczema or atopic dermatitis. In particular, it aims at a composition as described above. before to prevent and / or treat skin irritation, eczema, or atopic dermatitis.
- It also relates to a cosmetic process for the care and / or the cleaning of sensitive skin, irritated, or presenting with eczema or atopic dermatitis.
- It also relates to a cosmetic skin care process, implementing a composition as defined above, characterized in that it aims to reduce skin imperfections, including skin imperfections of oily skin.
- cosmetic skin care product particularly for the purpose of mattifying the skin, comprising the topical application thereof, of a composition as defined above.
- composition according to the invention may in particular be used to correct or prevent lesions and / or skin imperfections encountered in an acne or acne-prone skin, especially a subject with oily acne-prone skin.
- the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructo-oligosaccharide as a prebiotic.
- the invention therefore relates to a composition as described above, to reduce imperfections of oily skin.
- An aspect of the invention is more particularly the use of such a composition for the care and / or cleansing of the skin, especially oily skin, and more specifically for oily acne-prone skin.
- the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructooligosaccharide as a prebiotic.
- the invention also relates to a composition comprising the combination as described above, for the care and / or the cleaning of acne-prone or acne-prone skin, in particular fatty skin with an acne-prone tendency.
- the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructo-oligosaccharide as a prebiotic.
- the invention also relates to a cosmetic process for the care of oily skin, comprising the application to the skin of a composition comprising the combination as defined above.
- the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructooligosaccharide as a prebiotic.
- the invention also relates to a method for the care and / or cleaning of oily skin, comprising the application to the skin of a composition comprising the combination of a bacteriocin and a prebiotic as defined above .
- the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructo-oligosaccharide as a prebiotic.
- the composition described above is applied to areas of the face or forehead with a gloss of the skin.
- the composition according to the invention is applied to the skin of persons whose skin is of phototype III to VI.
- the invention furthermore relates to a composition as described above, in particular as a dermatological composition, intended for the preventive and / or curative treatment of acne and / or hyperseborrhoea.
- the invention therefore also relates to the use of the combination of a bacteriocin and a prebiotic for the preparation of a composition intended for the preventive and / or curative treatment of acne and / or hyperseborrhoea, and or intended to correct disorders associated with acne and / or hyperseborrhea.
- the invention furthermore relates to a plantaricin class II, in particular plantaricin 149, for use in the preventive and / or curative treatment of skin disorders related to P. acnes, in particular acne.
- the invention also relates to a cosmetic and / or dermatological composition comprising, in a physiologically acceptable medium, plantaricin 149.
- the invention also relates to a composition, in particular a dermatological composition, comprising, in a physiologically acceptable medium, a class II plantaricin, in particular the plantaricin peptide 149, for use in the preventive and / or curative treatment of skin-related disorders.
- a composition in particular a dermatological composition, comprising, in a physiologically acceptable medium, a class II plantaricin, in particular the plantaricin peptide 149, for use in the preventive and / or curative treatment of skin-related disorders.
- P. acnes especially in the treatment of acne.
- the composition for use in the preventive or curative treatment of disorders related to P. acnes is characterized in that it further comprises an additional asset for the care of the skin.
- fatty oils selected from desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebo-regulating agents, antioxidants, healing agents, astringent agents, and mixtures thereof.
- the invention relates to a cosmetic process for the care and / or cleaning of the skin, in particular of oily skin, comprising the topical application to said skin of a cosmetic composition comprising a physiologically acceptable medium. , a class II plantaricin, in particular plantaricin 149.
- the cosmetic process is intended to reduce and / or eliminate cutaneous imperfections related to P. acnes, in particular chosen from an irregular relief, dilated pores, comedones and / or blackheads.
- the composition according to the invention is used as an antibacterial active agent, to reduce the amount of acne-causing germs present on the skin. the skin, or to limit their development, and in particular to reduce the amount of P. / Acnes and / or P. Granulosum, or to limit the development of one of these germs. It may especially be in the form of a cream or an anti-acne gel.
- the dermatological composition according to the invention can thus be used in a subject having acne or hyperseborrhoea.
- the dermatological composition according to the invention can also be used for delaying or slowing the progression or preventing a further progression of acne, improving the aesthetic appearance of the skin of the user.
- the dermatological composition of the present invention may finally be administered to a person who does not have acne, but whose skin, especially oily skin, is acne-prone.
- the amount of the combination of at least one bacteriocin and at least one prebiotic in the dermatological composition according to the invention will advantageously be in the ranges described above, to obtain the desired effect.
- those skilled in the art are able to establish the effective amount of said combination in the dermatological composition according to the invention, and will ensure that the additional ingredients and / or active ingredients introduced into said composition do not affect the advantageous effects of the association.
- the invention relates to a method of dermatological treatment of the disorders of the skin indicated above, comprising the topical administration of a composition comprising at least the combination according to the invention, as described above. , to a person in need.
- composition can be applied in any way that can be envisaged by those skilled in the art.
- it may be envisaged to apply the composition directly to the skin or to dilute it beforehand in a suitable solvent.
- said methods or said uses according to the invention further comprise a rinsing step after application to the skin of the composition.
- the composition can be rinsed after a pause, advantageously between 5 and 30 minutes.
- the invention therefore also relates to a composition comprising the combination as defined above, intended for the treatment of desquamative disorders of the hair and / or the scalp.
- the desquamative states of the hair and / or the scalp are, for example, dandruff, seborrheic dermatitis.
- said desquamative states of the scalp are those induced by the yeast of the genus Malassezia spp., And more particularly M. furfur.
- compositions for the treatment of the desquamative states of the hair and / or the scalp according to the invention may be intended for a cosmetic or pharmaceutical application, particularly dermatological.
- compositions according to the invention are intended for a cosmetic application.
- compositions according to the invention are generally applied to the scalp or the hair.
- composition of the invention may be in any galenical form normally used, particularly in cosmetology.
- a composition for treating the desquamative conditions of the hair and / or the preferred scalp of the invention is a cosmetic composition intended for topical application.
- the composition for the treatment of the desquamative states of the hair and / or scalp according to the invention, after application to human hair and scalp can be rinsed or not rinsed with water or with a shampoo.
- compositions can be in any form conventionally used in the field concerned and for example in the form of an aqueous or oily solution or of the lotion or serum type dispersion, emulsions of liquid or semi-liquid consistency of the milk type, obtained by dispersion of a fatty phase in an aqueous phase (O / W) or conversely ( E / H), or suspensions or emulsions of soft consistency of the cream or gel type aqueous or anhydrous, or microcapsules or microparticles, or vesicular dispersions of ionic and / or nonionic type.
- aqueous or oily solution or of the lotion or serum type dispersion emulsions of liquid or semi-liquid consistency of the milk type, obtained by dispersion of a fatty phase in an aqueous phase (O / W) or conversely ( E / H), or suspensions or emulsions of soft consistency of the cream or gel type aqueous or anhydrous, or microcapsule
- the composition that may be used according to the invention may also be a composition for hair care, and in particular a shampoo, a setting lotion, a treatment lotion, a cream or a styling gel, a dye composition (especially oxidation dyes). optionally in the form of coloring shampoos, restructuring lotions for the hair, a permanent composition (in particular a composition for the first time of a perm), a preparation (lotion, gel, shampoo), anti-hair loss, a pest control shampoo, etc. .
- compositions for treating the desquamative states of the hair and / or scalp that can be used according to the invention are those conventionally used in the fields under consideration.
- compositions for the treatment of the desquamative states of the hair and / or the scalp which can be used according to the invention may also consist of solid preparations constituting soaps or cleaning bars.
- compositions for the treatment of the desquamative states of the hair and / or the scalp which can be used according to the invention can also be packaged in the form of an aerosol composition also comprising a propellant under pressure.
- compositions for the treatment of the desquamative states of the hair and / or scalp according to the invention contain a physiologically acceptable medium.
- this medium contains an aqueous phase containing water and optionally at least one water-miscible organic solvent such as C2-C6 monoalcohols such as ethanol, isopropanol, n-butanol, polyols such as propylene glycol, glycerol or glycol ethers.
- This medium may contain an oily phase containing one or more liquid fats which are liquid at room temperature (25 ° C.) and atmospheric pressure, immiscible with water, called "oils".
- the physiologically acceptable medium may, in addition, comprise at least one surfactant chosen from nonionic surfactants, anionic surfactants, cationic surfactants, amphoteric surfactants, and mixtures thereof.
- compositions according to the invention may further contain, as washing base, at least one surfactant chosen from anionic, nonionic and amphoteric surfactants and mixtures thereof.
- anionic surfactant usable in the present invention mention may especially be made of salts, in particular alkali metal salts such as sodium salts, ammonium salts, amine salts, aminoalcohol salts or salts thereof.
- alkaline earth metal salts for example of magnesium, of the following types: alkyl sulphates, alkyl ether sulphates, alkyl amido ether sulphates, alkylaryl polyethersulphates, monoglyceride sulphates; alkylsulfonates, alkylamidesulfonates, alkylarylsulfonates, ⁇ -olefinsulfonates, paraffin sulfonates, alkylsulfosuccinates, alkylethersulfosuccinates, alkylamidesulfosuccinates, alkylsulphoacetates, acylsarcosinates and acylglutamates, alkyl and acyl groups; of all these compounds having from
- alkyl monoesters and polyglycoside dicarboxylic acids such as alkyl glucoside citrates, alkyl polyglycoside tartrates and polyglycoside sulfosuccinates may also be used as anionic surfactants.
- Another group of anionic surfactants that can be used in the composition of the present invention is that of acyl lactylates, the acyl group of which contains from 8 to 20 carbon atoms.
- alkyl-D-galactoside-uronic acids and their salts polyoxyalkylenated (C6-C2 4) ether carboxylic acids, polyoxyalkylenated (C6-C2 4) aryl (C6-C2 4 poly (oxyalkylenated) ether carboxylic acids, poly (oxyalkylenated) (C 6 -C 2 4 ) alkyl amidoether carboxylic acids and their salts, in particular those containing from 2 to 50 ethylene oxide units, and mixtures thereof.
- polyoxyalkylenated (C6-C2 4) ether carboxylic acids polyoxyalkylenated (C6-C2 4) aryl (C6-C2 4 poly (oxyalkylenated) ether carboxylic acids, poly (oxyalkylenated) (C 6 -C 2 4 ) alkyl amidoether carboxylic acids and their salts, in particular those containing from 2 to 50 ethylene oxide units, and mixtures thereof.
- Anionic surfactants are preferably alkyl sulphates, alkyl ether sulphates and alkyl ether carboxylates, and mixtures thereof, in particular in the form of alkali metal or alkaline earth metal salts, ammonium, amine or aminoalcohol.
- amphoteric surfactant that may be used in the present invention, mention may be made of derivatives of secondary or tertiary aliphatic amines, in which the aliphatic group is a linear or branched chain containing from 8 to 22 carbon atoms and containing at least one anionic group such as for example, a carboxylate, sulfonate, sulfate, phosphate or phosphonate group.
- Ri represents an alkyl group derived from an acid Ra-COOH present in the hydrolysed coconut oil, a heptyl, nonyl or undecyl group, .
- R2 represents a beta-hydroxyethyl group, and
- R3 represents a carboxymethyl group
- B represents -CH 2 CH 2 OX ⁇
- X ' represents the group -CH 2 CH 2 -COOH or a hydrogen atom
- Y ' represents -COOH or the group -CH 2 -CHOH-SO 3 H
- R 1 ' represents an alkyl group of an R 1 -COOH acid present in coconut oil or in hydrolysed linseed oil, an alkyl group, especially C 17 and its iso form, an unsaturated C 17 group.
- coco-amphodiacetate disodium lauro-amphodiacetate disodium
- caprylamphodiacetate disodium caprylo-amphodiacetate disodium
- coco-ampho-dipropionate disodium lauro-ampho disodium dipropionate
- disodium capryl-ampho-dipropionate disodium caprylo-amphodipropionate
- lauro-amphodipropionic acid cocoamino-dipropionic acid.
- cocoamphodiacetate marketed by Rhodia under the trade name MIRANOL® C2M concentrate.
- C 8 -C 20 alkylbetaines C 8 -C 20 alkylamido (C 6 -C 8 ) alkylbetaines, alkylamphodiacetates and mixtures thereof are preferably used.
- nonionic surfactant that can be used in the composition according to the invention, mention may be made of the known compounds described in particular in the book "Handbook of Surfactants” by MR PORTER, published by Blackie & Son (Glasgow and London), 1991, pp. -178). It is chosen in particular from alcohols, alpha-diols, alkyl (C1 -20) phenols or polyethoxylated, polypropoxylated or polyglycerolated fatty acids, having a fatty chain comprising, for example, from 8 to 18 carbon atoms, the number ethylene oxide or propylene oxide groups which can range from 2 to 50 and the number of glycerol groups that can range from 2 to 30, their mixtures.
- nonionic surfactant which can be used in the invention, of ethylene oxide and propylene oxide condensates on fatty alcohols; polyethoxylated fatty amides preferably having from 2 to 30 ethylene oxide units; polyglycerolated fatty amides comprising on average from 1 to 5 glycerol groups and in particular from 1, 5 to 4; ethoxylated sorbitan fatty acid esters having 2 to 30 ethylene oxide units; fatty acid esters of sucrose; polyethylene glycol fatty acid esters; the (C6 to C2 4) alkylpolyglycosides; N- (C6-24 alkyl) glucamine derivatives; amine oxides such as (C 1 -C 4 alkyl) amine oxides or N- (C 10 -C 4 acyl) aminopropylmorpholine oxides; their mixtures.
- nonionic surfactants mentioned above are preferably used the (C6 to C2 4) polyg
- the washing base contains at least one anionic surfactant and at least one amphoteric or nonionic surfactant.
- the total amount of surfactant is generally in the range from 0.01% to 50% by weight, preferably from 0.1% to 25% by weight relative to the total weight of the composition.
- the total amount of surfactants (or washing base) is in particular chosen from 4% to 50% by weight, for example 8% to 25% by weight, based on the total weight of the composition.
- composition for the treatment of the desquamative states of the hair and / or the scalp according to the invention may, in addition, contain at least one additional ingredient conventionally used in the fields under consideration and chosen from principles cosmetic active agents having a beneficial effect on the hair and / or the scalp such as zinc salts of organic acid (acetate, glycolate, lactate, gluconate or citrate) or mineral (chloride and sulphate), vitamins (E, C , B2, B5, F), UV filters, anti-radical agents, preservatives, ceramides, plant extracts and formulation additives such as anionic, nonionic, cationic or amphoteric film-forming polymers, polymeric thickeners of aqueous phase or oily phase, nonpolymeric thickeners of aqueous phase such as hydroxylated or nonhydroxylated fatty acid salts or amides, pearlescent agents, opacifying agents, dyes soluble in the medium, pigments, fillers perfumes, oils of mineral, vegetable and / or synthetic origin, esters
- the pH of the composition for treating the desquamative states of the hair and / or scalp of the present invention is chosen in the range from 2 to 1 1 and preferably from 3 to 10, for example from 5 to 8.
- the composition for the treatment of desquamative conditions of the hair and / or scalp comprises at least one other active against the desquamative states of the scalp.
- the other agents for combating the desquamative states of the scalp are preferably chosen from pyridinethione salts such as zinc pyrithione, 1-hydroxy-2-pyrrolidone derivatives such as piroctone and piroctone olamine; selenium sulfides such as selenium disulfide; climbazole, undecylenic acid; ketoconazole, cyclopirox or their mixtures.
- the additional active agent or the mixture of additional active agents may represent from 0.001% to 10% by weight relative to the total weight of the composition and preferably from 0.1 to 5% by weight.
- the subject of the invention is a method of cosmetic treatment of the scalp for combating the desquamative states of the scalp, characterized in that a composition is applied to the hair and / or the scalp. for the treatment of desquamative conditions of the hair and / or scalp as defined above.
- the subject of the invention is also a process for the cosmetic treatment of the hair and / or the scalp, for combating dandruff or seborrheic dermatitis, characterized in that a composition is applied to the hair and / or the scalp. as described above.
- the method of cosmetic treatment of the desquamative states of the hair and / or scalp according to the invention is particularly suitable when said desquamative states of the scalp are induced by the yeast of the genus Malassezia spp., In particular M. furfur.
- Demineralized water qs 100g This milk is suitable for people with sensitive skin and can be used for a daily application on the body, for a moisturizing and protective effect.
- This sweet and fresh cream helps fight against acne problems with good efficiency.
- This composition is introduced into an aerosol can. Pressurized by introducing under pressure 35 g of dimethyl ether. The spray is applied on the scalp.
- the formula is pressurized with isobutane.
- the inhibitory action of the plantaricin A, plantaricin C19 and plantaricin 149 peptides was evaluated on the growth of C. xerosis germs (CIP 100653 - the main germ responsible for unpleasant body odors) and S. epidermidis (ATCC 155 commensal beneficial bacterium found in the microbial flora of the skin).
- C. xerosis germs CIP 100653 - the main germ responsible for unpleasant body odors
- S. epidermidis ATCC 155 commensal beneficial bacterium found in the microbial flora of the skin.
- the action of plantaricin 149 has also been tested on P. acnes germ (CIP 531 17 - germ found in people whose skin has a tendency to fat or hyperseborrheic and responsible for the development of acne).
- the peptides were at a concentration of 100 mg / L in DMSO.
- the test is based on the detection of the activity in solid medium and the confirmation of this activity in a liquid medium.
- the experiments on C. xerosis were carried out at 26 ° C, a pre-study having shown that this strain forms aggregates at 35 ° C,
- each strain tested covered the agar of a culture dish in a solid medium. Wells were made in the agar and the products to be tested were deposited in these wells.
- the products tested are the peptides mentioned above, at a concentration of 100 mg / L in DMSO, Irgasan 200 mg / L used as a positive control (also called Triclosan, this molecule is a recognized bactericide used especially in the treatment of bad smells), and water or DMSO as a negative control.
- Plantaricin 149 has an inhibitory effect on the growth of P. acnes.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Dermatology (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Chemical & Material Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Cosmetics (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Description
COMPOSITIONS COSMETIQUES OU DERMATOLOGIQUES A BASE DE COSMETIC OR DERMATOLOGICAL COMPOSITIONS BASED ON
BACTERIOCINES ET DE PREBIOTIQUES BACTERIOCINS AND PREBIOTICS
La présente invention concerne des compositions comprenant, dans un milieu physiologiquement acceptable, l'association d'au moins une bactériocine et d'au moins un prébiotique. Elle concerne également des utilisations et procédés cosmétiques mettant en oeuvre l'association d'au moins une bactériocine et d'au moins un prébiotique. Elle concerne encore l'utilisation thérapeutique de cette association. The present invention relates to compositions comprising, in a physiologically acceptable medium, the combination of at least one bacteriocin and at least one prebiotic. It also relates to cosmetic uses and processes involving the combination of at least one bacteriocin and at least one prebiotic. It still concerns the therapeutic use of this association.
De nombreuses bactéries se développent naturellement sur la peau. Elles constituent la flore commensale résidente de la peau et participent activement au maintien de sa santé. Elles jouent notamment un rôle important dans la résistance à la colonisation de micro-organismes saprophites et potentiellement pathogènes. La flore cutanée est composée de plusieurs espèces identifiées à ce jour: Staphylocoques (S.epidermis, S.hominis, S.haemolyticus, S.aureus), Corynebacteries (C.lipophiles ; C.urealyticum ; C.minutissimum ; C.jeikeium), Propionibacterie (P. acnés; P.avidum ; P. granulosum, P.propionicum), Microcoques (M.luteus; M.varians), Streptocoques (groupes A, B et G), Brevibacteries. Many bacteria grow naturally on the skin. They constitute the resident commensal flora of the skin and actively participate in maintaining its health. In particular, they play an important role in the resistance to colonization of saprophite and potentially pathogenic microorganisms. The cutaneous flora is composed of several species identified so far: Staphylococci (S.epidermis, S.hominis, S.haemolyticus, S.aureus), Corynebacteria (C.lipophiles, C.urealyticum, C.minutissimum, C.jeikeium) , Propionibacteria (P. acnes, P. avidum, P. granulosum, P.propionicum), Micrococci (M.luteus, M.varians), Streptococci (groups A, B and G), Brevibacteries.
Dans le domaine cosmétique, il est bien connu d'utiliser en application topique des produits déodorants contenant des substances actives de type anti-transpirant ou de type bactéricide pour diminuer, voire supprimer, les odeurs corporelles, en particulier les odeurs axillaires, généralement désagréables. La microflore axillaire est principalement composée de bactéries Gram+, comme les staphylocoques (e.g., S.epidermidis, S.hominis, etc.), les corynéformes (plus particulièrement C.xerosis), les microcoques (e.g., M.Luteus) et de Propionibacteries. Les mauvaises odeurs axillaires sont attribuées à la biotransformation microbienne des sécrétions naturelles, sans odeur, en produits volatils odorants. Les fortes odeurs axillaires sont principalement liées à la présence de C. xerosis sur cette zone. In the field of cosmetics, it is well known to use, in topical application, deodorant products containing active substances of the anti-perspirant or bactericidal type in order to reduce or even eliminate body odors, in particular axillary odors, which are generally unpleasant. The axillary microflora is mainly composed of Gram + bacteria, such as staphylococci (eg, S.epidermidis, S.hominis, etc.), coryneforms (more particularly C.xerosis), micrococci (eg, M.Luteus) and Propionibacteria. . The axillary bad odors are attributed to the microbial biotransformation of natural, odorless secretions into volatile odorants. Strong axillary odors are mainly related to the presence of C. xerosis in this area.
De nombreux désordres cutanés sont liés à la rupture de l'équilibre écologique de la flore résidente suite à la colonisation de la peau par des germes exogènes ou à la prolifération anormale d'une souche endogène. On peut notamment observer ce phénomène dans le développement des odeurs corporelles, notamment les odeurs axillaires et des pieds (principalement C. Xerosis pour les odeurs axillaires et B. linens pour les odeurs des pieds), chez les personnes à peau sensible, notamment en cas de dermatite atopique, eczéma (germe responsable : S.aureus), dans la production de pellicules (principalement M. furfur), ou encore chez les personnes dont la peau est à tendance grasse ou hyperséborrhéique (P. acnés) et de l'acné (germe responsable: P. acnés). Pour combattre ces microorganismes, il est courant d'utiliser des antibiotiques ou bactéricides. L'utilisation de ces composés pose cependant le problème de la non spécificité d'action visant indifféremment la flore pathogène et la flore résidente, et le problème du risque d'apparition de résistances bactériennes, ainsi que des problèmes de tolérance cutanée (irritations, allergies, ...). Many skin disorders are related to the rupture of the ecological balance of the resident flora following the colonization of the skin by exogenous germs or at the abnormal proliferation of an endogenous strain. In particular, this phenomenon can be observed in the development of body odors, especially axillary and foot smells (mainly C. Xerosis for axillary smells and B. linens for foot smells), in people with sensitive skin, especially in cases atopic dermatitis, eczema (responsible germ: S.aureus), in the production of dandruff (mainly M. furfur), or in people whose skin is prone to fatty or hyperseborrhoeic (P. acnes) and acne (germ responsible: P. acnes). To combat these microorganisms, it is common to use antibiotics or bactericides. The use of these compounds, however, raises the problem of the non-specificity of action aimed indifferently at the pathogenic flora and the resident flora, and the problem of the risk of the appearance of bacterial resistance, as well as skin tolerance problems (irritations, allergies). , ...).
Il persiste un besoin pour des composés qui appliqués sur la peau permettent la croissance et/ou la survie des bactéries désirées de la microflore cutané (flore résidente) et s'opposent à la croissance et/ou à la survie des bactéries non désirées présentes sur la peau (germes exogènes). There is still a need for compounds that apply to the skin to allow the growth and / or survival of the desired bacteria in the skin microflora (resident flora) and oppose the growth and / or survival of unwanted bacteria present on the skin. skin (exogenous germs).
La demande FR 2 916 634 propose l'association d'un fructo-oligosaccharide et d'un inducteur de peptides antimicrobiens pour améliorer la qualité de la flore microbienne ou rétablir son équilibre. II subsiste toutefois le besoin de disposer de compositions et de traitements cosmétiques améliorés permettant de retrouver un équilibre normal de la flore microbienne cutanée. The application FR 2 916 634 proposes the combination of a fructooligosaccharide and an inducer of antimicrobial peptides to improve the quality of the microbial flora or restore its equilibrium. There remains, however, the need for improved cosmetic compositions and treatments to restore a normal balance of cutaneous microbial flora.
La Demanderesse a découvert d'une manière surprenante et inattendue qu'on pouvait obtenir cet effet en utilisant l'association d'au moins un prébiotique et d'au moins une bactériocine. The Applicant has surprisingly and unexpectedly discovered that this effect can be achieved by using the combination of at least one prebiotic and at least one bacteriocin.
Cette découverte constitue la base de l'invention. La présente invention a donc pour objet l'association d'au moins une bactériocine et d'au moins un prébiotique, ainsi qu'une composition cosmétique et/ou dermatologique comprenant cette association. This discovery is the basis of the invention. The subject of the present invention is therefore the combination of at least one bacteriocin and at least one prebiotic, as well as a cosmetic and / or dermatological composition comprising this combination.
La présente invention a également pour objet l'utilisation de ladite association à titre d'agent cosmétique. The present invention also relates to the use of said combination as a cosmetic agent.
Elle a en outre pour objet une composition cosmétique et/ou dermatologique comprenant ladite association dans un milieu physiologiquement acceptable. It also relates to a cosmetic and / or dermatological composition comprising said combination in a physiologically acceptable medium.
Un autre objet de l'invention concerne également un procédé cosmétique de traitement des matières kératiniques comprenant l'application d'une telle composition sur les matières kératiniques. Another subject of the invention also relates to a cosmetic process for treating keratin materials, comprising the application of such a composition to keratin materials.
D'autres objets de l'invention apparaîtront dans la suite de la description. Other objects of the invention will appear in the following description.
Par "milieu physiologiquement acceptable", on entend un milieu compatible avec la peau et/ou ses phanères ou muqueuses qui présente une couleur, une odeur et un toucher agréables et qui ne génère pas d'inconforts inacceptables (picotements, tiraillements, rougeurs), susceptibles de détourner la consommatrice d'utiliser cette composition. "Physiologically acceptable medium" means a medium compatible with the skin and / or its integuments or mucous membranes which has a pleasant color, odor and feel and which does not generate unacceptable discomfort (tingling, tightness, redness), likely to divert the consumer from using this composition.
Par "matières kératiniques", on entend la peau (corps, visage, contour des yeux, cuir chevelu), cheveux, cils, sourcils, poils, ongles, lèvres, muqueuses. By "keratin materials" is meant the skin (body, face, eye area, scalp), hair, eyelashes, eyebrows, hair, nails, lips, mucous membranes.
BACTERIOCINES bacteriocins
Les bactériocines sont des composés peptidiques synthétisés naturellement par certaines bactéries. Elles jouent un rôle important dans la compétition entre souches bactériennes. Les bactériocines peuvent être bactéricides, c'est-à-dire éliminer certains microorganismes et/ou elles peuvent être bactériostatiques, c'est-à-dire inhiber la croissance de certains micro-organismes. L'activité bactéricide ou bactériostatique est orientée contre certaines espèces proches de la souche productrice. Bacteriocins are peptide compounds naturally synthesized by certain bacteria. They play an important role in the competition between bacterial strains. The bacteriocins can be bactericidal, that is to say eliminate certain microorganisms and / or they can be bacteriostatic, that is to say, inhibit the growth of certain microorganisms. The bactericidal or bacteriostatic activity is directed against certain species close to the producing strain.
Les bactériocines sont généralement des peptides cationiques et amphiphiles, responsables de la perméabilisation de la membrane des cellules cibles. En effet, elles se fixent à certains récepteurs membranaires et y provoquent la formation de pores. La membrane est ainsi rendue perméable à certains composés tels les ions ou molécules. Cela est généralement létal pour la bactérie cible. The bacteriocins are generally cationic and amphiphilic peptides, responsible for the permeabilization of the membrane of the target cells. Indeed, they bind to certain membrane receptors and cause the formation of pores. The membrane is thus made permeable to certain compounds such as ions or molecules. This is usually lethal to the target bacteria.
Classification des bactériocines Classification of bacteriocins
Les bactériocines sont classifiées selon des données structurales en quatre classes, comme proposé par Klaenhammer (1993). Ces quatre classes sont : The bacteriocins are classified according to structural data in four classes, as proposed by Klaenhammer (1993). These four classes are:
Classe I. Les lantibiotiques : peptides de taille inférieure à 5 kDa, stables à la chaleur et qui contiennent des acides aminés inhabituels soufrés formés post- traductionnellement, c'est-à-dire la lanthionine, la β-méthyl lanthionine, la déhydrobutyrine et la déhydroalanine. Ils peuvent être divisés en deux types : la classe la qui comprend des peptides cationiques hydrophobes allongés contenant jusqu'à 34 acides aminés et la classe Ib qui comprend les peptides globulaires chargés négativement ou sans charge nette et contenant jusqu'à 19 acides aminés (McAuliffe et al., 2001 ; Twomey et al., 2002). Il existe également des bactériocines de classe I à deux composants comme la cytolysine, la lacticine 3147 et la plantaricine W (cf. tableau 3). Class I. Lantibiotics: peptides smaller than 5 kDa, heat-stable and containing unusual sulfur-containing amino acids formed post-translationally, ie lanthionine, β-methyl lanthionine, dehydrobutyrin and dehydroalanine. They can be divided into two types: class Ia which comprises elongated hydrophobic cationic peptides containing up to 34 amino acids and class Ib which comprises negatively charged or no net charge globular peptides and containing up to 19 amino acids (McAuliffe et al., 2001, Twomey et al., 2002). There are also two-component class I bacteriocins such as cytolysin, lacticin 3147 and plantaricin W (see Table 3).
A titre illustratif, on peut citer comme bactériocines de classe I utilisables selon l'invention la nisine A, l'epidermine, la subtiline, la lactocine S, la salivaricine A, la plantaricine C. By way of illustration, Class I bacteriocins which may be used according to the invention may be nisin A, epidermine, subtilin, lactocin S, salivaricin A or plantaricin C.
On peut en particulier utiliser les bactériocines de classe I décrites dans les tableaux 1 , 2 et 3 ci-dessous. Tableau 1 : exemples de bactériocines de classe la In particular, the class I bacteriocins described in Tables 1, 2 and 3 below can be used. Table 1: Examples of class la bacteriocins
* telles qu'elles figurent dans le chapitre "Bactériocines de bactéries lactiques" (Morisset et al.) du livre Bactéries Lactiques et probiotiques, LUQUET François-Marie, CORRIEU Georges; éd. Lavoisier * as they appear in the chapter "Bacteriocins of lactic acid bacteria" (Morisset et al.) of the book Lactic and Probiotic Bacteria, LUQUET François-Marie, CORRIEU Georges; ed. Lavoisier
Tableau 2: exemples de bactériocines de classe Ib Table 2: Examples of class Ib bacteriocins
* telles qu'elles figurent dans le chapitre "Bactériocines de bactéries lactiques" (Morisset et al.) du livre Bactéries Lactiques et probiotiques, LUQUET François-Marie, CORRIEU Georges; éd. Lavoisier * as they appear in the chapter "Bacteriocins of lactic acid bacteria" (Morisset et al.) of the book Lactic and Probiotic Bacteria, LUQUET François-Marie, CORRIEU Georges; ed. Lavoisier
Tableau 3: exemples de bactériocines de classe I à deux composants Table 3: Examples of two-component Class I bacteriocins
* telles qu'elles figurent dans le chapitre "Bactériocines de bactéries lactiques" (Morisset et al.) du livre Bactéries Lactiques et probiotiques, LUQUET François-Marie, CORRIEU Georges; éd. Lavoisier * as they appear in the chapter "Bacteriocins of lactic acid bacteria" (Morisset et al.) of the book Lactic and Probiotic Bacteria, LUQUET François-Marie, CORRIEU Georges; ed. Lavoisier
Classe II. Peptides de taille inférieure à 10 kDa, stables à la chaleur, ne contenant pas d'acides aminés modifiés. Les séquences de quelques bactériocines appartenant à cette classe se trouvent au tableau 4. Class II. Peptides less than 10 kDa, heat stable, not containing modified amino acids. The sequences of some bacteriocins belonging to this class are shown in Table 4.
Tableau 4: séquences de certaines bactériocines de classe II Classe Ma: « Pediocin-like bacteriocin » Table 4: Sequences of some class II bacteriocins Class Ma: "Pediocin-like bacteriocin"
Mésentéricine MTNMKSVEAYQQLDNQNLKKVVGGKYYGNGVHCTKSGCSVNWGEAASAGIH Y105 RLANGGNGFW (SEQ ID NO:1 ) Mesentericin MTNMKSVEAYQQLDNQNLKKVVGGKYYGNGVHCTKSGCSVNWGEAASAGIH Y105 RLANGGNGFW (SEQ ID NO: 1)
Sakacine P MEKFIELSLKEVTAITGGKYYGNGVHCGKHSCTVDWGTAIGNIGNNAAANWAT Sakacin P MEKFIELSLKEVTAITGGKYYGNGVHCGKHSCTVDWGTAIGNIGNNAAANWAT
GWNAGG (SEQ ID NO:2) GWNAGG (SEQ ID NO: 2)
Curvacine A MNNVKELSMTELQTITGGARSYGNGVYCNNKKCWVNRGEATQSIIGGMISGW Curvacin A MNNVKELSMTELQTITGGARSYGNGVYCNNKKCWVNRGEATQSIIGGMISGW
ASGLAGM (SEQ ID NO:3) ASGLAGM (SEQ ID NO: 3)
Piscicoline 126 MKTVKELSVKEMQLTTGGKYYGNGVSCNKNGCTVDWSKAIGIIGNNAAANLTT Piscicoline 126 MKTVKELSVKEMQLTTGGKYYGNGVSCNKNGCTVDWSKAIGIIGNNAAANLTT
GGAAGWNKG (SEQ ID NO:4) GGAAGWNKG (SEQ ID NO: 4)
Carnobactériocine MKSVKELNKKEMWWINGGAISYGNGVYCNKEKCWVNKAENKQAITGIVIGGW Bm 1 ASSLAGMGH (SEQ ID NO:5) Carnobacteriocin MKSVKELNKKEMWWGGAGAYGNGVYCNKEKCWVNKAENKQAITGIVIGGW Bm 1 ASSLAGMGH (SEQ ID NO: 5)
Pédiocine PA-1 MKKIEKLTEKE M AN 11 G G KY YG N G VTCG KH SCS VD WG KATTC 11 N N G AM AWAT Pediocin PA-1 MKKIEKLTEKE M AN 11 G G KY YG N G VTCG KH SCS VD WG KATTC 11 N N G AM AWAT
GGHQGNHKC (SEQ ID NO:6) GGHQGNHKC (SEQ ID NO: 6)
Entérocine A MKHLKILSIKETWLIYGGTTHSGKYYGNGVYCTKNKCTVDWAKATTCIAGMSIG Enterocin A MKHLKILSIKETWLIYGGTTHSGKYYGNGVYCTKNKCTVDWAKATTCIAGMSIG
GFLGGAIPGKC (SEQ ID NO:7) GFLGGAIPGKC (SEQ ID NO: 7)
Sakacine G MKNTRSLTIQEIKSITGGKYYGNGVSCNSHGCSVNWGQAWTCGVNHLANGGH Sakacin G MKNTRSLTIQEIKSITGGKYYGNGVSCNSHGCSVNWGQAWTCGVNHLANGGH
GGVC (SEQ ID NO:8) GGVC (SEQ ID NO: 8)
Classe Mb: « Two-peptides bacteriocin » Mb class: "Two-peptides bacteriocin"
ABP-1 18 a KRGPNCVGNFLGGLFAGAAAGVPLGPAGIVGGANLGMVGGALTCL (SEQ ID ABP-1 18 a KRGPNCVGNFLGGLFAGAAAGVPLGPAGIVGGANLGMVGGALTCL (SEQ ID
NO:9) NO: 9)
β KNGYGGSGNRWVHCGAGIVGGALIGAIGGPWSAVAGGISGGFTSCR (SEQ ID NO: 10) β KNGYGGSGNRWVHCGAGIVGGALIGAIGGPWSAVAGGISGGFTSCR (SEQ ID NO: 10)
Lactocine 705 a MDNLNKFKKLSDNKLQATIGG (SEQ ID NO: 1 1 ) Lactocin 705 a MDNLNKFKKLSDNKLQATIGG (SEQ ID NO: 1 1)
β MESNKLEKFANISNKDLNKITGG (SEQ ID NO: 12) β MESNKLEKFANISNKDLNKITGG (SEQ ID NO: 12)
Lactococcine MN M IRGTGKGLAAAMVSGAAMGGAIGAFGGPVGAIMGAWGGAVGGAMKYSI Lactococcin MN M IRGTGKGLAAAMVSGAAMGGAIGAFGGPVGAIMGAWGGAVGGAMKYSI
(SEQ ID NO: 13) (SEQ ID NO: 13)
N GSIWGAIAGGAVKGAIAASWTGNPVGIGMSALGGAVLGGVTYARPVH (SEQ ID NO: 14) ## STR1 ##
Plantaricine EF E FNRGGYNFGKSVRHVVDAIGSVAGIRGILKSIR (SEQ ID NO:15) Plantaricin EF E FNRGGYNFGKSVRHVVDAIGSVAGIRGILKSIR (SEQ ID NO: 15)
F VFHAYSARGVRNNYKSAVGPADWVISAVRGFIHG (SEQ ID NO: 16) F VFHAYSARGVRNNYKSAVGPADWVISAVRGFIHG (SEQ ID NO: 16)
Classe Ile Island Class
Plantaricine A MKIQIKGMKQLSNKEMQKIVGGKSSAYSLQMGATAIKQVKKLFKKWGW (SEQ Plantaricin A MKIQIKGMKQLSNKEMQKIVGGKSSAYSLQMGATAIKQVKKLFKKWGW (SEQ
ID NO: 17) ID NO: 17)
Lactococcine A MKNQLNFNIVSDEELSEANGGKLTFIQSTAAGDLYYNTNTHKYVYQQTQNAFG Lactococcin A MKNQLNFNIVSDEELSEANGGKLTFIQSTAAGDLYYNTNTHKYVYQQTQNAFG
AAANTIVNGWMGGAAGGFGLHH (SEQ ID NO:18) ## STR1 ##
Lactococcine 972 MKTKSLVLALSAVTLFSAGGIVAQAEGTWQHGYGVSSAYSNYHHGSKTHSATV Lactococcin 972 MKTKSLVLALSAVTLFSAGGIVAQAEGTWQHGYGVSSAYSNYHHGSKTHSATV
VN N NTG RQG KDTQRAG VWAKATVG RN LTEKASFYYN FW (SEQ ID NO: 19) VN N NTG RQG KDTQRAG VWAKATVG RN LTEKASFYYN FW (SEQ ID NO: 19)
La classe II est divisée en trois sous-classes. Les bactériocines de la sous-classe Ma sont des peptides ayant une structure similaire à celle de la pédiocine, qui fut la première bactériocine de ce groupe décrite. Les bactériocines de la sous-classe Ma contiennent entre 27 et 48 acides aminés et ont toutes une partie N-terminale hydrophobe contenant la séquence consensus YGNGV ainsi qu'un pont disulfure et une partie C-terminale moins conservée, hydrophobe ou amphiphile qui détermine la spécificité d'action (Fimland et al., 2000 ; Richard et al., 2006). Cette classe peut également plus particulièrement être définie comme possédant en particulier une séquence N-terminale conservée YGNGVxCxxxxCxV (Klaenhammer, 1993; Ennahar et al. 2000; Nés et Holo, 2000). Elles ont toutes une activité anti-Listeria. Certaines bactériocines de cette sous-classe contiennent également un deuxième pont disulfure dans leur domaine C-terminal qui semble être important dans la stabilisation de la structure tertiaire. Il semble par ailleurs qu'il leur conférerait une meilleure activité antimicrobienne, une meilleure résistance à l'exposition à des hautes températures et un spectre d'action plus large (Eijsink et al., 1998 ; Fimland et al., 2000 ; Drider et al., 2006 ; Richard et al., 2006). La sous-classe Mb comprend les bactériocines ayant besoin de deux peptides pour avoir une activité. Deux types de bactériocines de classe Mb peuvent être distingués : le type E (Enhancing) où la fonction d'un des deux peptides est d'augmenter l'activité de l'autre et le type S (Synergy) où les deux peptides sont complémentaires. La sous-classe Ile contient les bactériocines ne pouvant pas être classées dans les autres sous-classes. Class II is divided into three subclasses. The bacteriocins of the subclass Ma are peptides having a structure similar to that of pediocin, which was the first bacteriocin of this group described. The bacteriocins of the subclass Ma contain between 27 and 48 amino acids and all have a hydrophobic N-terminal portion containing the YGNGV consensus sequence as well as a disulfide bridge and a less conserved, hydrophobic or amphiphilic C-terminal portion which determines the specificity of action (Fimland et al., 2000, Richard et al., 2006). This class may also be more particularly defined as possessing in particular a conserved N-terminal sequence YGNGVxCxxxxCxV (Klaenhammer, 1993, Ennahar et al., 2000, Nés and Holo, 2000). They all have anti-Listeria activity. Some bacteriocins of this subclass also contain a second bridge disulfide in their C-terminal domain that appears to be important in the stabilization of the tertiary structure. It also appears to provide better antimicrobial activity, better resistance to high temperature exposure, and a broader spectrum of action (Eijsink et al., 1998, Fimland et al., 2000; al., 2006, Richard et al., 2006). Subclass Mb comprises the bacteriocins in need of two peptides for activity. Two types of class Mb bacteriocins can be distinguished: type E (Enhancing) where the function of one of the two peptides is to increase the activity of the other and type S (Synergy) where the two peptides are complementary . The subclass Ile contains the bacteriocins that can not be classified in the other subclasses.
A titre d'exemple de bactériocines de classe Ma utilisables selon l'invention, on peut citer les bactériocines du tableau 5. On peut également citer la coaguline. Tableau 5: exemples de bactériocines de classe Ma By way of example of class Ma bacteriocins that can be used according to the invention, mention may be made of the bacteriocins of Table 5. It is also possible to mention coagulin. Table 5: Examples of class Ma bacteriocins
* telles qu'elles figurent dans le chapitre "Bactériocines de bactéries lactiques" (Morisset et al.) du livre Bactéries Lactiques et probiotiques, LUQUET François-Marie, CORRIEU Georges; éd. Lavoisier * as they appear in the chapter "Bacteriocins of lactic acid bacteria" (Morisset et al.) of the book Lactic and Probiotic Bacteria, LUQUET François-Marie, CORRIEU Georges; ed. Lavoisier
A titre d'exemple de bactériocines de classe Mb utilisables selon l'invention, on peut citer la lactococcine G, la lactacine, la lactoccine 705, la thermophiline, la plantaricine EF, la leucocine, la carnocine, l'acidocine. On peut plus particulièrement citer les bactériocines listées dans le tableau 6 ci-dessous. By way of example of class Mb bacteriocins that can be used according to the invention, mention may be made of lactococcin G, lactacin, lactocaine 705, thermophilin and plantaricin. EF, leucocin, carnocine, acidocine. In particular, mention may be made of the bacteriocins listed in Table 6 below.
Tableau 6 : exemples de bactériocines de classe Mb Table 6: Examples of class Mb bacteriocins
* telles qu'elles figurent dans le chapitre "Bactériocines de bactéries lactiques" (Morisset et al.) du livre Bactéries Lactiques et probiotiques, LUQUET François-Marie, CORRIEU Georges; éd. Lavoisier * as they appear in the chapter "Bacteriocins of lactic acid bacteria" (Morisset et al.) of the book Lactic and Probiotic Bacteria, LUQUET François-Marie, CORRIEU Georges; ed. Lavoisier
A titre d'exemple de bactériocines de classe Ile utilisables selon l'invention, on peut citer les bactériocines listées dans le tableau 7 ci-dessous. By way of example of Ile-class bacteriocins which can be used according to the invention, mention may be made of the bacteriocins listed in Table 7 below.
Tableau 7 : exemples de bactériocines de classe Ile Table 7: Examples of class Ile class bacteriocins
Bactériocines (peptides) Producteurs Références Bacteriocins (peptides) Producers References
Acidocine B Lactobacillus acidophilus M46 Leer et al., 1995 Acidocin B Lactobacillus acidophilus M46 Leer et al., 1995
Carnobacterium divergens Carnobacterium divergens
Divergicine A Worobo et al., 1995 Divergicin A Worobo et al., 1995
LV13 LV13
Entérocine P Enterococcus faecium P13 Cintas et al., 1997 Enterococcus P Enterococcus faecium P13 Cintas et al., 1997
Bactériocine 31 Enterococcus faecalis 31 Tomita et al., 1996 Bacteriocin 31 Enterococcus faecalis 31 Tomita et al., 1996
Lactococcine 972 Lactococcus lactis IPLA 972 Martinez et al., 1999 Lactococcin 972 Lactococcus lactis IPLA 972 Martinez et al., 1999
Entérocine I Enterococcus faecium 6T1a Quadri et al., 1994 Enterococcus faecium Enterococcus 6T1a Quadri et al., 1994
Entérocine Q Enterococcus faecium L50 Cintas et al., 2000 Enterococcus faecium enterococci L50 Cintas et al., 2000
Lactobacillus acidophilus Lactobacillus acidophilus
Acidocine 8912 Kanatani et al., 1995b Acidocin 8912 Kanatani et al., 1995b
TK8912 TK8912
Bactériocine AS-48 Enterococcus faecalis Mendoza et al., 1999 Bacteriocin AS-48 Enterococcus faecalis Mendoza et al., 1999
Gasséricine A Lactobacillus gasseri LA39 Kawai et al., 1998 Gassericin A Lactobacillus gasseri LA39 Kawai et al., 1998
Lactococcine A Lactococcus lactis Van Belkum et al., 1991 c; Morgan et al., 1995Lactococcin A Lactococcus lactis Van Belkum et al., 1991c; Morgan et al., 1995
Lactococcine B Lactococcus lactis Van Belkum et al., 1992 Lactococcin B Lactococcus lactis Van Belkum et al., 1992
Lactococcus lactis spp. Lactococcus lactis spp.
Diacétine B Ali et al., 1995 Diacetin B Ali et al., 1995
diacetylactis UL720 diacetylactis UL720
Carnobacterium piscicola Holck et al., 1994; Carnobacterium piscicola Holck et al., 1994;
Carnobactériocine A Carnobacteriocin A
LV17A Worobo et al., 1994 LV17A Worobo et al., 1994
Lactobacillus amylovorus LMG Lactobacillus amylovorus LMG
Lactobine A Contreras et al., 1997 Lactobin A Contreras et al., 1997
P-13139 P-13139
Divergicine 750 Carnobacterium divergens 750 Holck et al., 1996 Divergicin 750 Carnobacterium divergens 750 Holck et al., 1996
Enterococcus faecium Bogovic-Matijasic et al., 1998; Enterococcus faecium Bogovic-Matijasic et al., 1998;
Entérocine B Enterococcus B
T136/C492 et BFE900 Franz et al., 1999 T136 / C492 and BFE900 Franz et al., 1999
Peptide A Lactobacillus acidophilus LF221 Tahara et al., 1997 Peptide A Lactobacillus acidophilus LF221 Tahara et al., 1997
Peptide B Lactobacillus acidophilus LF221 Tahara et al., 1997 Peptide B Lactobacillus acidophilus LF221 Tahara et al., 1997
Leuconostoc mesenteroides Leuconostoc mesenteroides
Leucocine B-TA33a Papathanasopoulos et al., 1998 Leucocin B-TA33a Papathanasopoulos et al., 1998
TA33a TA33a
Gasséricine B3 Lactobacillus gasseri JCM 2124 Tahara et al., 1997 Gassericin B3 Lactobacillus gasseri JCM 2124 Tahara et al., 1997
Brévicine Lactobacillus brevis Benoit et al., 1997 Brévicine Lactobacillus brevis Benoit et al., 1997
Lactobacillus plantarum Lactobacillus plantarum
Plantaricine 1.25β Ehrmann et al., 2000 Plantaricin 1.25β Ehrmann et al., 2000
TMW1.25 TMW1.25
Leuconostoc mesenteroides Revol-Junelles et al., 1996; Leuconostoc mesenteroides Revol-Junelles et al., 1996;
Mésentéricine B105 Mesentericin B105
spp. mesenteroides Y105 Hechard et al., 1999 spp. mesenteroides Y105 Hechard et al., 1999
* telles qu'elles figurent dans le chapitre "Bactériocines de bactéries lactiques" (Morisset et al.) du livre Bactéries Lactiques et probiotiques, LUQUET François-Marie, CORRIEU Georges; éd. Lavoisier La plantaricine 149 est une bactériocine de classe II de séquence YSLQMGATAIKQVKKLFKKKGG (SEQ ID NO:20). * as they appear in the chapter "Bacteriocins of lactic acid bacteria" (Morisset et al.) of the book Lactic and Probiotic Bacteria, LUQUET François-Marie, CORRIEU Georges; ed. Lavoisier Plantaricin 149 is a class II bacteriocin of sequence YSLQMGATAIKQVKKLFKKKGG (SEQ ID NO: 20).
Classe III. Protéines de taille supérieure à 30 kDa et sensibles à la chaleur. La structure et le mode d'action de ces bactériocines diffèrent complètement des autres bactériocines produites par les bactéries lactiques. A ce jour, quatre bactériocines de cette classe ont été identifiées : l'helveticin J produite par Lactobacillus helveticus A, l'enterolysin A produite par Enterococcus faecium, la zoocin A produite par Spreptococcus zooepidemicus et la millericin B produite par Streptococcus milleri (Nilsen et al., 2003 ; Papagianni, 2003 ; Nigutova et al., 2007). Class III. Proteins larger than 30 kDa and sensitive to heat. The structure and mode of action of these bacteriocins differs completely from other bacteriocins produced by lactic acid bacteria. To date, four bacteriocins of this class have been identified: helveticin J produced by Lactobacillus helveticus A, enterolysin A produced by Enterococcus faecium, zoocin A produced by Spreptococcus zooepidemicus and millericin B produced by Streptococcus milleri (Nilsen et al. al., 2003; Papagianni 2003; Nigutova et al. 2007).
A titre d'exemple de bactériocines de classe III utilisables selon l'invention, on peut citer les bactériocines listées dans le tableau 8 ci-dessous. By way of example of class III bacteriocins that can be used according to the invention, mention may be made of the bacteriocins listed in Table 8 below.
Tableau 8 : exemples de bactériocines de classe III Table 8: Examples of class III bacteriocins
Bactériocines (peptides) Producteurs Références Bacteriocins (peptides) Producers References
Hélvécitine J Lactobacillus helveticus 481 Joerger et Klaenhammer 1990 Helvecitin J Lactobacillus helveticus 481 Joerger and Klaenhammer 1990
Streptococcus milleri NMSCC Streptococcus milleri NMSCC
Milléricine B Beukes et al., 2000 Millericin B Beukes et al., 2000
061 061
Streptococcus zooepidermicus Streptococcus zooepidermicus
Zoocine A Simmonds et al., 1996 Zoocine A Simmonds et al., 1996
4881 * telles qu'elles figurent dans le chapitre "Bactériocines de bactéries lactiques" (Morisset et al.) du livre Bactéries Lactiques et probiotiques, LUQUET François-Marie, CORRIEU Georges; éd. Lavoisier Classe IV. Peptides requérant une partie carbohydratée ou lipidique pour avoir une activité. Aucune bactériocine de cette classe n'a été décrite. 4881 * as they appear in the chapter "Bacteriocins of lactic acid bacteria" (Morisset et al.) of the book Lactic and Probiotic Bacteria, LUQUET François-Marie, CORRIEU Georges; ed. Lavoisier Class IV. Peptides requiring a carbohydrate or lipid portion to have activity. No bacteriocin of this class has been described.
Les bactériocines utilisées dans la présente invention peuvent être produites par toute méthode adaptée connue de l'homme du métier. Elles peuvent être synthétisées sans difficulté par l'homme du métier en utilisant les techniques conventionnelles de synthèse peptidique en phase solide ou en solution (M. Bodanszky, Principles of Peptides Synthesis, 2eme éd., 1993, Edition Springer- Verlag). Les bactériocines utilisées selon l'invention peuvent également être produites par des microorganismes, en utilisant des méthodes de bioingénierie. Dans ce cas, il peut être nécessaire d'extraire et purifier le peptide des microorganismes producteurs, avant formulation. Dans un mode préféré de réalisation, les bactériocines utilisées selon l'invention sont des peptides synthétiques. The bacteriocins used in the present invention can be produced by any suitable method known to those skilled in the art. They can be synthesized easily by the skilled person using conventional techniques of peptide synthesis, solid phase or in solution (M. Bodanszky, Principles of Peptide Synthesis, 2nd ed., 1993, Springer-Verlag Edition). The bacteriocins used according to the invention can also be produced by microorganisms, using bioengineering methods. In this case, it may be necessary to extract and purify the peptide producing microorganisms, before formulation. In a preferred embodiment, the bacteriocins used according to the invention are synthetic peptides.
Selon un mode particulier de réalisation, la bactériocine utilisée est une bactériocine de classe I. La bactériocine de classe I peut notamment être choisie parmi celles mentionnées ci-dessus. En particulier, la bactériocine de classe I peut être choisie dans le groupe constitué des bactériocines listées dans les tableaux 1 , 2 et 3. Selon un autre mode de réalisation particulier, la bactériocine de classe I est choisie dans le groupe constitué de la nisine A, l'épidermine, la salivaricine A, la plantaricine C et la gallidermine. According to a particular embodiment, the bacteriocin used is a class I bacteriocin. The class I bacteriocin may especially be chosen from those mentioned above. In particular, class I bacteriocin may be selected from the group consisting of the bacteriocins listed in Tables 1, 2 and 3. According to another particular embodiment, class I bacteriocin is selected from the group consisting of nisin A , epidermin, salivaricin A, plantaricin C and gallidermin.
Selon un mode particulier de réalisation, la bactériocine utilisée est une bactériocine de classe II, en particulier une bactériocine de classe Ma, Mb ou Ile. En particulier, l'invention a pour objet l'utilisation d'au moins l'une des bactériocines de classe Ma mentionnées ci-dessus, plus particulièrement d'une bactériocine de classe Ma choisie dans le groupe constitué des bactériocines listées dans le tableau 4 ou 5. Dans un mode particulier de réalisation, la bactériocine de classe Ma est choisie dans le groupe constitué de la pédiocine PA-1 , la sakacine A et la sakacine P. Selon un mode particulier de réalisation, la bactériocine utilisée est une bactériocine de classe Mb. En particulier, l'invention a pour objet l'utilisation d'au moins l'une des bactériocines de classe Mb mentionnées ci-dessus, plus particulièrement d'une bactériocine de classe Mb choisie dans le groupe constitué des bactériocines listées dans le tableau 4 ou 6. Selon un autre mode particulier de réalisation, la bactériocine utilisée est une bactériocine de classe Ile. En particulier, l'invention a pour objet l'utilisation d'au moins l'une des bactériocines de classe Ile mentionnées ci-dessus, plus particulièrement d'une bactériocine de classe Ile choisie dans le groupe constitué des bactériocines listées dans le tableau 4 ou 7. According to a particular embodiment, the bacteriocin used is a class II bacteriocin, in particular a class Ma, Mb or Ile bacteriocin. In particular, the subject of the invention is the use of at least one of the class I bacteriocins mentioned above, more particularly a class Ma bacteriocin chosen from the group consisting of the bacteriocins listed in Table 4. or 5. In a particular embodiment, the class Ma bacteriocin is selected from the group consisting of pediocin PA-1, sakacin A and sakacin P. According to a particular embodiment, the bacteriocin used is a bacteriocin of Mb class. In particular, the subject of the invention is the use of at least one of the class Mb bacteriocins mentioned above, more particularly a class Mb bacteriocin selected from the group consisting of bacteriocins listed in Table 4 or 6. According to another particular embodiment, the bacteriocin used is a class Ile bacteriocin. . In particular, the subject of the invention is the use of at least one of the Ile class bacteriocins mentioned above, more particularly an Ile-class bacteriocin chosen from the group consisting of the bacteriocins listed in Table 4. or 7.
Selon un mode préféré de réalisation, la bactériocine utilisée est une plantaricine de classe II. Plus préférentiellement, la plantaricine de classe II est choisie parmi la plantaricine A, la plantaricine C19 et la plantaricine 149. According to a preferred embodiment, the bacteriocin used is a plantaricin class II. More preferably, the plantaricin class II is selected from plantaricin A, plantaricin C19 and plantaricin 149.
Selon un mode particulier de réalisation, la bactériocine dans l'association est une bactériocine de type III. La bactériocine de type III peut plus particulièrement être choisie dans le groupe constitué de l'helveticin J, l'enterolysin A, la zooein A et la millericin B. According to one particular embodiment, the bacteriocin in the combination is a type III bacteriocin. Type III bacteriocin may more particularly be selected from the group consisting of helveticin J, enterolysin A, zooein A and millericin B.
Selon un mode particulier de réalisation, l'association selon l'invention comprend une seule bactériocine. Selon un mode alternatif de réalisation, l'association selon l'invention comprend plusieurs bactériocines. Il peut s'agir en particulier d'un mélange comprenant plusieurs bactériocines d'une même classe, ou comprenant des bactériocines de classes différentes (par exemple, classe I + classe II; classe I + classe III; classe II + classe III). L'association selon l'invention peut donc comprendre le mélange des bactériocines suivantes: According to a particular embodiment, the combination according to the invention comprises a single bacteriocin. According to an alternative embodiment, the combination according to the invention comprises several bacteriocins. It can be in particular a mixture comprising several bacteriocins of the same class, or comprising bacteriocins of different classes (for example, class I + class II, class I + class III, class II + class III). The combination according to the invention may therefore comprise the mixture of the following bacteriocins:
- au moins l'une des bactériocines de classe I des tableaux 1 , 2 et 3 avec au moins une bactériocine de classe II, en particulier Ma, Mb ou Ile, plus particulièrement choisie parmi celles listées dans les tableaux 4 et 5. De préférence, la bactériocine de classe II est une plantaricine de classe II, en particulier la plantaricine A, la plantaricine C19 ou la plantaricine 149; at least one of the class I bacteriocides of Tables 1, 2 and 3 with at least one Class II bacteriocin, in particular Ma, Mb or Ile, more particularly chosen from those listed in Tables 4 and 5. Preferably Class II bacteriocin is a class II plantaricin, in particular plantaricin A, plantaricin C19 or plantaricin 149;
- au moins une bactériocines de classe I choisie dans le groupe constitué de la nisine A, l'épidermine, la salivaricine A, la plantaricine C et la gallidermine, avec une bactériocine de classe II choisie dans le groupe constitué de la pédiocine PA-1 , la sakacine A et la sakacine P. at least one class I bacteriocin selected from the group consisting of nisin A, epidermin, salivaricin A, plantaricin C and gallidermine, with class II bacteriocin selected from the group consisting of pediocin PA-1, sakacin A and sakacin P.
PREBIOTIQUES PREBIOTIC
Dans le cadre de la présente invention, les "prébiotiques" sont des oligosaccharides et/ou polysaccharides constitués de 2 à 100 unités sucres, de préférence de 2 à 30 unités sucres. La première définition officielle donnée par Gibson et al. en 1995 est la suivante "substance non digestible qui induit un effet physiologique bénéfique à l'hôte en stimulant de façon spécifique la croissance et/ou l'activité d'un nombre limité de populations bactériennes déjà établies dans le côlon". In the context of the present invention, the "prebiotics" are oligosaccharides and / or polysaccharides consisting of 2 to 100 sugar units, preferably 2 to 30 sugar units. The first official definition given by Gibson et al. in 1995 is the following "non-digestible substance that induces a physiological effect beneficial to the host by specifically stimulating the growth and / or activity of a limited number of bacterial populations already established in the colon".
Selon la présente invention, un ou plusieurs prébiotiques sont utilisés afin de maintenir un milieu favorable au développement de la flore endogène cutanée. Les prébiotiques sont en particulier utilisés pour orienter le métabolisme de l'ensemble de la flore cutané afin de maintenir un bon équilibre de cette dernière tout en ne favorisant pas le développement de certaines bactéries pathogènes. A ce titre, les prébiotiques mis en oeuvre dans le cadre de la présente invention sont ceux susceptibles d'être métabolisés par les bactéries bénéfiques de la flore cutanée. Selon un mode particulièrement préféré de réalisation, le prébiotique utilisé dans les compositions de l'invention est un prébiotique métabolisé par les germes bénéfiques de la peau, mais qui ne sont pas métabolisables par les germes indésirables responsables des désordres cutanés dont le traitement cosmétique ou dermatologique est souhaité. Selon ce mode de réalisation, les prébiotiques modulent la composition et/ou l'activité de l'écosystème naturel, en privilégiant les microorganismes exerçant un effet positif sur la peau. According to the present invention, one or more prebiotics are used in order to maintain a favorable environment for the development of endogenous cutaneous flora. In particular, prebiotics are used to guide the metabolism of the entire skin flora to maintain a good balance of the latter while not promoting the development of certain pathogenic bacteria. As such, the prebiotics used in the context of the present invention are those likely to be metabolized by the beneficial bacteria of the skin flora. According to a particularly preferred embodiment, the prebiotic used in the compositions of the invention is a prebiotic metabolized by the beneficial seeds of the skin, but which are not metabolizable by the undesirable germs responsible for skin disorders, including cosmetic or dermatological treatment. is desired. According to this embodiment, prebiotics modulate the composition and / or the activity of the natural ecosystem, giving priority to microorganisms having a positive effect on the skin.
Dans un mode particulier de réalisation, on met en oeuvre un mélange de prébiotiques dans les compositions, utilisations et procédés selon l'invention. In a particular embodiment, a mixture of prebiotics is used in the compositions, uses and processes according to the invention.
Les prébiotiques contiennent des carbohydrates. Ils sont plus spécifiquement composés d'oligosaccharides. Les oligosaccharides peuvent êtres produits notamment à partir de glucose, galactose, xylose, maltose, sucrose, lactose, amidon, xylane, l'hémicellulose, de l'inuline, des gommes, notamment à partir de gomme d'acacia, ou un de leurs mélanges. Prebiotics contain carbohydrates. They are more specifically composed of oligosaccharides. The oligosaccharides may be produced in particular from glucose, galactose, xylose, maltose, sucrose, lactose, starch, xylan, hemicellulose, inulin, gums, especially from acacia gum, or a mixture thereof.
On citera à titre d'exemple les ingrédients prébiotiques suivants. By way of example, the following prebiotic ingredients may be mentioned.
L'inuline inulin
L'inuline est particulièrement abondante dans les rhizomes de plantes notamment l'artichaut de Jérusalem et la chicorée dont elle est extraite industriellement. On la trouve aussi dans d'autres végétaux appartenant à la famille des Asteraceae tels les topinambours ou les oignons de dahlias et la bardane. Elle est considérée comme une fibre alimentaire soluble. Inulin is particularly abundant in the rhizomes of plants, notably the Jerusalem artichoke and the chicory from which it is extracted industrially. It is also found in other plants belonging to the family Asteraceae such as Jerusalem artichokes or dahlias onions and burdock. It is considered a soluble dietary fiber.
Les inulines sont des polymères linéaires polydispersés de formule générale GFn (G = glucose, F = Fructose, n variant de 2 à plus de 60), les unités de fructose étant liées entre elles par une liaison β (2→ 1 ). Les inulines correspondent donc à une chaîne d'unités fructose terminée par une unité glucose. The inulins are polydispersed linear polymers of general formula GFn (G = glucose, F = Fructose, n varying from 2 to more than 60), the fructose units being linked together by a β bond (2 → 1). Inulins therefore correspond to a chain of fructose units terminated by a glucose unit.
Parmi les inulines utilisables et disponibles dans le commerce, on peut citer l'Inutec H25P n compris entre 2 et 7 l'Inutec N25 d'Orafti (n moyen = 25). Among the usable and commercially available inulins, there may be mentioned Inutec H25P n between 2 and 7 Inutec N25 Orafti (n average = 25).
Les fructo-oligosaccharides Les fructo-oligosaccharides (ou FOS) répondent à la formule générale G(F)n ou G est une unité glucose, F de fructose et n varie de 1 à 10. Les fructo-oligosaccharides (FOS) sont produits soit par hydrolyse enzymatique partielle de l'inuline (ex : Raftilose® d'Orafti-Belgique), soit par synthèse enzymatique à partir de saccharose (ex : Actilight® de Beghin Meiiji industries-France). Fructooligosaccharides The fructo-oligosaccharides (or FOS) correspond to the general formula G (F) n where G is a glucose, F fructose unit and n ranges from 1 to 10. The fructo-oligosaccharides (FOS) are produced either by partial enzymatic hydrolysis inulin (eg Raftilose® from Orafti-Belgium), or by enzymatic synthesis from sucrose (eg Actilight® from Beghin Meiiji industries-France).
Selon un mode particulier de réalisation, le prébiotique mis en oeuvre dans l'invention correspond à un mélange de FOS. On peut en particulier citer les mélanges GF2 + GF3 + GF4 tels que le Quantom FOS95 de Quantum hi-Tech ou l'Actilight® de Beghin Meiiji industries-France, ce dernier correspondant à un mélange GF2 37%, GF3 53% et GF4 10%. According to a particular embodiment, the prebiotic used in the invention corresponds to a mixture of FOS. In particular, mention may be made of GF2 + GF3 + GF4 blends such as the Quantom FOS95 from Quantum Hi-Tech or Actilight® from Beghin Meiiji Industries-France, the latter corresponding to a GF2 37%, GF3 53% and GF4 mixture. %.
Les qluco-oliqosaccharides GOS GOS qluco-oliqosaccharides
Les gluco-oligosaccharides (ou GOS) sont des oligosaccharides constitués d'un enchaînement d'unités glucose liées en a1 -6 et pouvant contenir également des liaisons α1 -2, α1 -3 ; a1 -4. Ils sont synthétisés par une réaction de transglucosylation catalysée par des enzymes de la famille des glucanes-saccharases. Gluco-oligosaccharides (or GOS) are oligosaccharides consisting of a sequence of glucose units linked at a1 -6 and which may also contain α1 -2, α1-3 bonds; a1 -4. They are synthesized by a transglucosylation reaction catalyzed by enzymes of the glucan saccharase family.
Parmi les GOS utilisables et disponibles dans le commerce, on peut citer le Bioecolia® de Solabia. Among the GOS that can be used and commercially available, mention may be made of Bioecolia® from Solabia.
Oligosaccharides issus du soja Oligosaccharides from soy
Ils sont extraits directement à partir de la graine de soja et ne requièrent aucun traitement enzymatique. Ils contiennent naturellement du raffinose et du stachyose, dont la formule est [a-D-Gal-(1→6)-]m-a-D-Glu-(1→2)- -D-Fru avec m=1 pour le raffinose et n=2 pour le stachyose. They are extracted directly from the soybean and do not require any enzyme treatment. They naturally contain raffinose and stachyose, whose formula is [αD-Gal- (1 → 6) -] m- αD-Glu- (1 → 2) - -D-Fru with m = 1 for raffinose and n = 2 for stachyose.
Parmi les oligosaccharides issus utilisables, on peut citer le Soya-oligo de calpis food ind. Japan Pyrodextrines : Among the oligosaccharides that can be derived that may be used, mention may be made of Soya-oligo from calpis food ind. Japan Pyrodextrines:
Les pyrodextrines sont un mélange d'oligosaccharides provenant de l'hydrolyse de l'amidon. Isomaltooligosaccharide Pyrodextrins are a mixture of oligosaccharides derived from the hydrolysis of starch. Isomaltooligosaccharide
Ils sont produits à partir de l'amidon. Ce sont des oligomères de glucose liés en a(1 - 6) avec un degré de polymérisation compris entre 2 et 5. A titre d'exemple, on peut utiliser l'lsomalto900P de Showa Sango. They are produced from starch. These are glucose oligomers bound to a (1-6) with a degree of polymerization of between 2 and 5. By way of example, the Showa Sango lsomalto900P can be used.
Xylooliqosaccharide Xylooliqosaccharide
Les xylo-oligosaccharide sont des oligosaccharides constitués de xylose liées en β(1 -4). The xylo-oligosaccharide oligosaccharides consist of β (1 -4) -linked xylose.
A titre d'exemple, on peut utiliser le Xylo 95P de Suntory limited. Transqalactooliqosaccharide For example, the Xylo 95P from Suntory limited can be used. Transqalactooliqosaccharide
Ce sont des oligomères linéaires de galactose, de structure chimique a-D-Glucose- (1→4)-[ -D-Galactose-(1→6)-]n (2 < n < 5) obtenus par fermentation du lactose. They are linear oligomers of galactose, of chemical structure a-D-Glucose- (1 → 4) - [-D-Galactose- (1 → 6) -] n (2 <n <5) obtained by fermentation of lactose.
A titre d'exmple, on peut utiliser le TOS 100 de Yakult Honsha Co.Ltd. Japan. For example, the TOS 100 from Yakult Honsha Co.Ltd. Japan.
Mélange de prébiotiques Mix of prebiotics
Selon un mode particulier de réalisation, la composante prébiotique mise en oeuvre dans l'association selon l'invention correspond à un mélange de prébiotiques tels que définis ci-dessus. Dans une variante, il peut s'agir d'un mélange de prébiotiques de même type. Par exemple, comme mentionné ci-dessus, on peut utiliser un mélange de FOS, notamment un mélange GF2+GF3+GF4, tel que le Quantom FOS95 de Quantum hi-Tech ou l'Actilight® de Beghin Meiiji industries-France, ce dernier correspondant à un mélange GF2 37%, GF3 53% et GF4 10%. According to a particular embodiment, the prebiotic component used in the combination according to the invention corresponds to a mixture of prebiotics as defined above. In a variant, it may be a mixture of prebiotics of the same type. For example, as mentioned above, it is possible to use a mixture of FOS, in particular a GF2 + GF3 + GF4 mixture, such as Quantom FOS95 from Quantum Hi-Tech or Actilight® from Beghin Meiiji Industries-France, the latter corresponding to a mixture GF2 37%, GF3 53% and GF4 10%.
Selon une autre variante, il peut s'agir d'un mélange de prébiotiques de types différents. L'invention concerne notamment la mise en oeuvre d'un mélange d'inuline avec un GOS, un FOS, des oligosaccharides issus du soja, des pyrodextrines, un isomaltooligosachharide, un xylooligosaccharide et/ou un transgalactooligosaccharide. Selon un mode particulier de réalisation, on met en oeuvre un mélange d'inuline et d'un GOS tel que le Bioline de Gova Ingrédients. L'invention concerne également la mise en oeuvre d'un mélange d'un GOS avec une inuline, un FOS, des oligosaccharides issus du soja, des pyrodextrines, un isomaltooligosachharide, un xylooligosaccharide et/ou un transgalactooligosaccharide. Plus particulièrement, on peut mettre en oeuvre un mélange de GOS, de FOS et d'un probiotiques (Lactobacillus) tel que l'Ecoskin de Solabia. According to another variant, it may be a mixture of prebiotics of different types. The invention relates in particular to the use of a mixture of inulin with a GOS, an FOS, oligosaccharides derived from soya, pyrodextrins, an isomaltooligosachharide, a xylooligosaccharide and / or a transgalactooligosaccharide. According to a particular embodiment, we put in A mixture of inulin and a GOS such as Bioline Gova Ingredients. The invention also relates to the use of a mixture of a GOS with an inulin, a FOS, oligosaccharides derived from soybean, pyrodextrins, an isomaltooligosachharide, a xylooligosaccharide and / or a transgalactooligosaccharide. More particularly, it is possible to use a mixture of GOS, FOS and probiotics (Lactobacillus) such as Solabia Ecoskin.
ASSOCIATION ASSOCIATION
La présente invention vise l'association d'au moins une bactériocine avec au moins un prébiotique. The present invention relates to the combination of at least one bacteriocin with at least one prebiotic.
Selon un mode particulier de réalisation, l'association comprend au moins un prébiotique choisi parmi l'inuline, un FOS, un GOS, un isomaltooligosaccharide et un xylooligosaccharide. Selon un mode préféré de réalisation, le prébiotique est choisi parmi un FOS, un isomaltooligosaccharide et un xylooligosaccharide. Selon un autre mode de réalisation, le prébiotique est un mélange de prébiotiques, par exemple un mélange inuline + GOS ou GOS + FOS + probiotiques. Selon une première variante de ce mode de réalisation, l'association comprend également une une bactériocine de classe I, notamment une bactériocine choisie dans les tableaux 1 , 2 et 3, et en particulier une bactériocine choisie dans le groupe constitué de la nisine A, l'épidermine, la salivaricine A, la plantaricine C et la gallidermine. Selon une seconde variante de ce mode de réalisation, l'association comprend au moins une bactériocine de classe II, en particulier de classe Ma, notamment une bactériocine choisie dans les tableaux 4 et 5, et en particulier une bactériocine choisie dans le groupe constitué de la pédiocine PA-1 , la sakacine A et la sakacine P. Selon une forme de cette seconde variante, la bactériocine de classe II est une plantaricine de classe II, en particulier une plantaricine de classe II choisie parmi la plantaricine A, la plantaricine C19 et la plantaricine 149. According to a particular embodiment, the combination comprises at least one prebiotic chosen from among inulin, an FOS, a GOS, an isomaltooligosaccharide and a xylooligosaccharide. According to a preferred embodiment, the prebiotic is chosen from an FOS, an isomaltooligosaccharide and a xylooligosaccharide. According to another embodiment, the prebiotic is a mixture of prebiotics, for example an inulin + GOS or GOS + FOS + probiotic mixture. According to a first variant of this embodiment, the combination also comprises a class I bacteriocin, in particular a bacteriocin chosen from Tables 1, 2 and 3, and in particular a bacteriocin chosen from the group consisting of nisin A, epidermin, salivaricin A, plantaricin C and gallidermin. According to a second variant of this embodiment, the combination comprises at least one class II bacteriocin, in particular class Ma, in particular a bacteriocin chosen from Tables 4 and 5, and in particular a bacteriocin selected from the group consisting of pediocin PA-1, sakacin A and sakacin P. According to a form of this second variant, class II bacteriocin is a class II plantaricin, in particular a plantaricin class II selected from plantaricin A, plantaricin C19 and plantaricin 149.
Selon un mode particulièrement préféré de réalisation, la bactériocine est une plantaricine de classe II, en particulier une plantaricine de classe II choisie parmi la plantaricine A, la plantaricine C19 et la plantaricine 149 et le prébiotique est choisi parmi un FOS, un isomaltooligosaccharide et un xylooligosaccharide. According to a particularly preferred embodiment, the bacteriocin is a plantaricin class II, in particular a class II plantaricin chosen from plantaricin A, plantaricin C19 and plantaricin 149 and the prebiotic is selected from an FOS, an isomaltooligosaccharide and a xylooligosaccharide.
Selon un mode particulier de réalisation, l'invention concerne l'association de la nisine A avec un GOS, notamment le GOS commercialisé par Solabia sous la dénomination Bioecolia. According to a particular embodiment, the invention relates to the combination of nisin A with a GOS, in particular GOS marketed by Solabia under the name Bioecolia.
Selon un autre mode particulier de réalisation, l'invention concerne l'association de la nisine A avec un FOS, notamment l'Actilight mentionné ci-dessus. According to another particular embodiment, the invention relates to the combination of nisin A with a FOS, in particular the Actilight mentioned above.
Selon encore un autre mode de réalisation, l'invention concerne l'association de l'épidermidine avec un GOS, notamment le GOS commercialisé par Solabia sous la dénomination Bioecolia. Selon un autre mode particulier de réalisation, l'invention concerne l'association de la plantaricine 149 avec un FOS, notamment l'Actilight mentionné ci-dessus. According to yet another embodiment, the invention relates to the combination of epidermidine with a GOS, in particular GOS marketed by Solabia under the name Bioecolia. According to another particular embodiment, the invention relates to the combination of plantaricin 149 with a FOS, in particular the Actilight mentioned above.
L'invention concerne également l'utilisation de l'association telle que décrite ci- dessus, à titre d'actif cosmétique. The invention also relates to the use of the combination as described above, as a cosmetic active.
En particulier, elle vise l'utilisation de l'association selon l'invention à titre d'agent de soin et/ou de nettoyage de la peau, en particulier à titre d'agent de soin pour les peaux sensibles, irritées, présentant de l'eczéma ou une dermatite atopique, ou à titre de d'agent de soin et/ou de nettoyage pour les peaux grasses, en particulier les peaux grasses à tendance acnéique ou hyperséborrhéique. Elle vise aussi l'utilisation de l'association selon l'invention à titre d'agent anti-pelliculaire ou d'agent déodorant. In particular, it relates to the use of the combination according to the invention as a skin care and / or cleansing agent, in particular as a care agent for sensitive, irritated skin, presenting eczema or atopic dermatitis, or as a care agent and / or cleansing for oily skin, in particular oily skin with acne or hyperseborrheic tendency. It also relates to the use of the combination according to the invention as anti-dandruff agent or deodorant agent.
L'invention vise encore l'utilisation d'une association telle que décrite ci-dessus pour la fabrication d'une composition (cosmétique et/ou dermatologique) destinée à prévenir et/ou à traiter l'acné et/ou l'hyperséborrhée. L'invention vise encore l'utilisation d'une association telle que décrite ci-dessus pour la fabrication d'une composition (cosmétique et/ou dermatologique) destinée à prévenir et/ou à traiter l'irritation de la peau, l'eczéma, ou encore la dermatite atopique. The invention also relates to the use of an association as described above for the manufacture of a composition (cosmetic and / or dermatological) intended to prevent and / or treat acne and / or hyperseborrhoea. The invention also relates to the use of an association as described above for the manufacture of a composition (cosmetic and / or dermatological) intended to prevent and / or treat skin irritation, eczema , or atopic dermatitis.
L'invention concerne en outre l'utilisation d'une bactériocine à titre d'agent de soin et/ou de nettoyage de la peau, en particulier à titre d'agent de soin pour les peaux sensibles, irritées, présentant de l'eczéma ou une dermatite atopique ou à titre de d'agent de soin et/ou de nettoyage pour les peaux grasses, en particulier les peaux grasses à tendance acnéique ou hyperséborrhéique. L'invention vise également l'utilisation d'une bactériocine telle que définie ci-dessus, pour la fabrication d'une composition destinée au traitement des peaux sensibles, des peaux irritées, de l'eczéma, ou encore de la dermatite atopique. L'invention concerne également une composition cosmétique et/ou dermatologique comprenant, dans un milieu physiologiquement acceptable, l'association telle que décrite ci-dessus. The invention further relates to the use of a bacteriocin as a skin care and / or cleansing agent, in particular as a care agent for sensitive, irritated skin with eczema. or atopic dermatitis or as a care agent and / or cleansing agent for oily skin, in particular oily skin with an acne or hyperseborrhoeic tendency. The invention also relates to the use of a bacteriocin as defined above, for the manufacture of a composition for the treatment of sensitive skin, irritated skin, eczema, or atopic dermatitis. The invention also relates to a cosmetic and / or dermatological composition comprising, in a physiologically acceptable medium, the combination as described above.
Selon une variante, l'invention concerne également une composition cosmétique et/ou dermatologique comprenant dans un milieu physiologiquement acceptable au moins une bactériocine de type III. According to one variant, the invention also relates to a cosmetic and / or dermatological composition comprising, in a physiologically acceptable medium, at least one type III bacteriocin.
Chaque bactériocine présente dans les compositions selon l'invention est avantageusement présente à une concentration comprise entre 0,0001 % et 1 % en Matière Active par rapport au poids total de la composition. Each bacteriocin present in the compositions according to the invention is advantageously present at a concentration of between 0.0001% and 1% of active material relative to the total weight of the composition.
Selon un mode particulier de réalisation, les prébiotiques (c'est-dire, le prébiotique ou le mélange de prébiotiques tels que définis ci-dessus) sont utilisés à un taux compris entre 0,01 et 70% en poids par rapport au poids total de la composition les contenant. De préférence, ils sont présents à un taux compris entre 0,01 et 20 % en poids par rapport au poids total de la composition. Les compositions cosmétiques et/ou dermatologiqes selon l'invention peuvent être des compositions déodorantes, pour le soin et/ou le nettoyage de la peau, en particulier pour le soin des peaux sensibles, irritées, présentant de l'eczéma ou une dermatite atopique ou pour le soin et/ou le nettoyage des peaux grasses, en particulier des peaux grasses à tendance acnéique ou hyperséborrhéique. Les compositions selon l'invention peuvent également être des compositions pour le traitement des pellicules. According to a particular embodiment, the prebiotics (that is, the prebiotic or the mixture of prebiotics as defined above) are used at a level of between 0.01% and 70% by weight relative to the total weight. of the composition containing them. Preferably, they are present at a level of between 0.01% and 20% by weight relative to the total weight of the composition. The cosmetic and / or dermatological compositions according to the invention may be deodorant compositions, for the care and / or the cleaning of the skin, in particular for the care of sensitive, irritated skin, presenting with eczema or atopic dermatitis or for the care and / or the cleansing of oily skin, in particular oily skin with an acne or hyperseborrhoeic tendency. The compositions according to the invention can also be compositions for the treatment of films.
FORMES GALENIQUES GALENIC FORMS
Les compositions selon l'invention peuvent se présenter selon toutes les formes galéniques classiquement utilisées dans l'application envisagée. Bien entendu, l'homme du métier veillera à choisir les composants et éventuels ingrédients et/ou actifs complémentaires, et/ou leur quantité, de manière telle que les propriétés avantageuses de l'association d'au moins une bactériocine et d'au moins un prébiotique selon l'invention ne soient pas, ou substantiellement pas, altérées par l'adjonction envisagée. The compositions according to the invention may be in any of the galenic forms conventionally used in the intended application. Of course, those skilled in the art will take care to choose the components and any additional ingredients and / or active agents, and / or their quantity, in such a way that the advantageous properties of the combination of at least one bacteriocin and at least one a prebiotic according to the invention are not, or not substantially, impaired by the addition envisaged.
* Composition déodorante Lorsque la composition selon l'invention est une composition déodorante, elle peut se présenter sous forme de suspension, de dispersion, de solution, de gel, d'émulsions (notamment de consistance liquide ou semi-liquide, molle, semi-solide ou solide), notamment émulsion huile-dans-eau (H/E), cire-dans-eau ou eau-dans- huile (E/H), ou multiple (E/H/E ou polyol/H/E ou H/E/H), sous forme de crème, de pâte, de mousse, de microémulsion, de dispersion de vésicules notamment de lipides ioniques ou non, de dispersions cire/phase aqueuse, de lotion biphasé ou multiphase, de pâte, notamment de pâte souple. Elle peut notamment se présenter sous forme de gel aqueux ou de solution aqueuse ou hydroalcoolique. Les compositions selon l'invention peuvent se présenter sous la forme d'une composition de protection, de traitement ou de soin pour le visage, pour les mains ou pour le corps (par exemple crèmes de jour, crème de nuit, crème démaquillante, composition anti-solaire, lait corporels de protection ou de soin, laits après-solaire, lotion, gel ou mousse pour le soin de la peau); une composition après-rasage. * Deodorant composition When the composition according to the invention is a deodorant composition, it may be in the form of suspension, dispersion, solution, gel, emulsions (in particular of liquid or semi-liquid, soft, semi-liquid consistency). solid or solid), in particular oil-in-water (O / W), wax-in-water or water-in-oil (W / O) emulsion, or multiple (E / H / E or polyol / H / E or H / E / H), in the form of cream, paste, foam, microemulsion, dispersion of vesicles, in particular ionic lipids or not, wax / aqueous phase dispersions, two-phase or multiphase lotion, pulp, especially soft dough. It may especially be in the form of an aqueous gel or an aqueous or hydroalcoholic solution. The compositions according to the invention may be in the form of a protection, treatment or care composition for the face, for the hands or for the body (for example, day creams, night cream, make-up removal cream, anti-solar composition, body protection or care milk, after-sun milks, lotion, gel or mousse for skin care); an aftershave composition.
Elles peuvent également être utilisées pour le maquillage. They can also be used for makeup.
Selon un mode de réalisation, les compositions sont des compositions de revêtement de la peau du corps ou du visage plus particulièrement des compositions de maquillage ou de soin de la peau du corps ou du visage telles que par exemple des fonds de teint ou des compositions de maquillage du corps. According to one embodiment, the compositions are compositions for coating the skin of the body or of the face, more particularly compositions for makeup or care of the skin of the body or of the face, such as, for example, foundations or hair compositions. body makeup.
L'homme du métier pourra choisir la forme galénique appropriée, ainsi que sa méthode de préparation, sur la base de ses connaissances générales, en tenant compte d'une part de la nature des constituants utilisés, notamment de leur solubilité dans le support, et d'autre part de l'application envisagée pour chaque composition. Those skilled in the art may choose the appropriate dosage form, as well as its method of preparation, on the basis of its general knowledge, taking into account, on the one hand, the nature of the constituents used, in particular their solubility in the support, and on the other hand, the intended application for each composition.
Les compositions peuvent être notamment conditionnées sous forme pressurisée dans un dispositif aérosol ou dans un flacon pompe ; conditionnée dans un dispositif muni d'une paroi ajourée notamment une grille ; conditionnées dans un dispositif muni d'un applicateur à billes ("roll-on") ; conditionnées sous forme de bâtonnets (sticks), sous forme de poudre libre ou compactée; ou encore appliquées sur des lingettes. Les compositions peuvent également se présenter sous la forme de savons ou de gels (rincés ou non rincés). Elles contiennent à cet égard les ingrédients généralement utilisés dans ce type de produits et bien connus de l'homme de l'art. Selon une autre forme particulière de l'invention, les compositions selon l'invention peuvent être anhydres. Une composition anhydre selon l'invention peut notamment se présenter sous la forme d'un stick, d'une crème, d'un soft-solid ou d'un aérosol. The compositions may in particular be packaged in pressurized form in an aerosol device or in a pump bottle; packaged in a device provided with a perforated wall including a grid; packaged in a device provided with a ball-on applicator; packaged in the form of sticks (sticks), in the form of loose or compacted powder; or else applied on wipes. The compositions may also be in the form of soaps or gels (rinsed or not rinsed). They contain in this respect the ingredients generally used in this type of products and well known to those skilled in the art. According to another particular form of the invention, the compositions according to the invention can be anhydrous. An anhydrous composition according to the invention may especially be in the form of a stick, a cream, a soft-solid or an aerosol.
On entend par composition anhydre une composition contenant moins de 2 % en poids d'eau, voire moins de 0,5 % d'eau, et notamment exempte d'eau, l'eau n'étant pas ajoutée lors de la préparation de la composition mais correspondant à l'eau résiduelle apportée par les ingrédients mélangés. Selon une autre forme particulière de l'invention, les compositions selon l'invention peuvent être solides en particulier sous forme de bâtonnet ou stick. By anhydrous composition is meant a composition containing less than 2% by weight of water, or even less than 0.5% of water, and especially free of water, the water not being added during the preparation of the composition but corresponding to the residual water provided by the mixed ingredients. According to another particular form of the invention, the compositions according to the invention can be solid, in particular in the form of a stick or a stick.
Par «composition solide», on entend que la mesure de la force maximale mesurée en texturométrie lors de l'enfoncement d'une sonde dans l'échantillon de formule doit être au moins égale à 0,25 Newton, en particulier au moins égal à 0,30 Newton, notamment au moins égale 0,35 Newton, appréciée dans des conditions de mesure précises comme suit. Les formules sont coulées à chaud dans des pots de 4 cm de diamètre et 3 cm de fond. Le refroidissement est fait à température ambiante. La dureté des formules réalisées est mesurée après 24 heures d'attente. Les pots contenant les échantillons sont caractérisés en texturométrie à l'aide d'un texturomètre tel que celui commercialisé par la société Rhéo TA-XT2, selon le protocole suivant : une sonde de type bille en inox de diamètre 5 mm est amenée au contact de l'échantillon à une vitesse de 1 mm/s. Le système de mesure détecte l'interface avec l'échantillon avec un seuil de détection égal à 0,005 newtons. La sonde s'enfonce de 0,3 mm dans l'échantillon, à une vitesse de 0,1 mm/s. L'appareil de mesure enregistre l'évolution de la force mesurée en compression au cours du temps, pendant la phase de pénétration. La dureté de l'échantillon correspond à la moyenne des valeurs maximales de la force détectée pendant la pénétration, sur au moins 3 mesures. By "solid composition" it is meant that the measurement of the maximum force measured in texturometry during the insertion of a probe into the sample of formula must be at least 0.25 Newton, in particular at least equal to 0.30 Newton, especially at least 0.35 Newton, appreciated under precise measurement conditions as follows. The formulas are poured hot in pots 4 cm in diameter and 3 cm deep. Cooling is done at room temperature. The hardness of the formulas is measured after 24 hours of waiting. The pots containing the samples are characterized in texturometry using a texturometer such as that marketed by Rhéo TA-XT2, according to the following protocol: a ball-type stainless steel probe of diameter 5 mm is brought into contact with the sample at a speed of 1 mm / s. The measurement system detects the interface with the sample with a detection threshold equal to 0.005 newtons. The probe sinks 0.3 mm into the sample at a rate of 0.1 mm / s. The meter records the evolution of the force measured in compression over time, during the penetration phase. The hardness of the sample corresponds to the average of the maximum values of the force detected during the penetration, on at least 3 measurements.
Phase aqueuse Les compositions selon l'invention destinées à l'usage cosmétique peuvent comporter au moins une phase aqueuse. Elles sont notamment formulées en lotions aqueuses ou en émulsion eau-dans-huile, huile-dans-eau, ou en émulsion multiple (émulsion triple huile-dans-eau-dans-huile ou eau-dans-huile-dans-eau (de telles émulsions sont connues et décrites par exemple par C. FOX dans « Cosmetics and Toiletries » - november 1986 - Vol 101 - pages 101 -1 12). Aqueous phase The compositions according to the invention intended for cosmetic use may comprise at least one aqueous phase. They are in particular formulated in aqueous lotions or in water-in-oil, oil-in-water or multiple emulsion emulsions (triple oil-in-water-in-oil or water-in-oil-in-water emulsion). such emulsions are known and described for example by C. FOX in "Cosmetics and Toiletries" - november 1986 - Vol 101 - pages 101-122).
La phase aqueuse des dites compositions contient de l'eau et en général d'autres solvants solubles ou miscibles dans l'eau. Les solvants solubles ou miscibles dans l'eau comprennent les mono alcools à chaîne courte par exemple en Ci-C4 comme l'éthanol, l'isopropanol ; les diols ou les polyols comme l'éthylèneglycol, le 1 ,2- propylèneglycol, le 1 ,3-butylène glycol, l'hexylèneglycol, le diéthylèneglycol, le dipropylene glycol, le 2-éthoxyéthanol, le diéthylène glycol monométhyléther, le triéthylène glycol monométhyléther et le sorbitol. On utilisera plus particulièrement le propylèneglycol et la glycérine, le propane 1 ,3 diol . The aqueous phase of said compositions contains water and in general other solvents soluble or miscible in water. Solvents that are soluble or miscible in water include short-chain monohydric alcohols, for example C 1 -C 4 such as ethanol, isopropanol; diols or polyols such as ethylene glycol, 1,2-propylene glycol, 1,3-butylene glycol, hexylene glycol, diethylene glycol, dipropylene glycol, 2-ethoxyethanol, diethylene glycol monomethyl ether, triethylene glycol monomethyl ether and sorbitol. More particularly, propylene glycol and glycerol, propane 1,3 diol will be used.
La composition selon l'invention a de préférence un pH compris entre 3 et 9, selon le support choisi. The composition according to the invention preferably has a pH of between 3 and 9, depending on the support chosen.
Emulsionnants a) Emulsionnants huile-dans-eau Comme émulsionnants pouvant être utilisés dans les émulsions huile-dans-eau ou émulsions triples huile-dans-eau-dans-huile, on peut citer par exemple les émulsionnants non ioniques tels que les esters d'acides gras et de glycérol oxyalkylénés (plus particulièrement polyoxyéthylénés) ; les esters d'acides gras et de sorbitan oxyalkylénés ; les esters d'acides gras oxyalkylénés (oxyéthylénés et/ou oxypropylénés) ; les éthers d'alcools gras oxyalkylénés (oxyéthylénés et/ou oxypropylénés) ; les esters de sucres comme le stéarate de sucrose ; et leurs mélanges tels que le mélange de stéarate de glycéryle et de stéarate de PEG-40. Emulsifiers a) Oil-in-water emulsifiers As emulsifiers that can be used in oil-in-water emulsions or oil-in-water-in-oil triple emulsions, mention may be made, for example, of nonionic emulsifiers such as esters of oxyalkylenated fatty acids and glycerol (more particularly polyoxyethylenated); oxyalkylenated fatty acid and sorbitan esters; oxyalkylenated fatty acid esters (oxyethylenated and / or oxypropylenated); oxyalkylenated fatty alcohol ethers (oxyethylenated and / or oxypropylenated); sugar esters such as sucrose stearate; and mixtures thereof such as the mixture of glyceryl stearate and PEG-40 stearate.
On peut citer également les mélanges émulsionnants alcool gras/alkylpolyglycoside tels que sont décrits dans les demandes WO92/06778, WO95/13863 et WO98/47610 comme les produits commerciaux vendus par la société SEPPIC sous les appellation MONTANOV ®. b) Emulsionnants eau-dans-huile Mention may also be made of the fatty alcohol / alkylpolyglycoside emulsifying mixtures as described in applications WO92 / 06778, WO95 / 13863 and WO98 / 47610, such as the commercial products sold by the company SEPPIC under the name MONTANOV®. (b) Water-in-oil emulsifiers
Parmi les émulsionnants pouvant être utilisés dans les émulsions eau-dans-huile ou émulsions triples (H/E/H ou E/H/E), on peut citer à titre d'exemple les alkyl dimethicone copolyols répondant à la formule (I) suivante CH, Among the emulsifiers that can be used in water-in-oil emulsions or triple emulsions (O / W / H or W / O / W), there may be mentioned by way of example alkyl dimethicone copolyols corresponding to formula (I) next CH,
I I
CH3 - Si Si -Si - O- ■Si - (i) CH 3 - S i If -Si - O- ■ Si - (i)
I I
CH., CH.,
CH, CH,
Ri R Ri R
dans lesquelles : in which :
Ri désigne un groupement alkyle linéaire ou ramifié en C12-C20 et de préférence en R 1 denotes a linear or branched C 12 -C 20 alkyl group and preferably
R2 désigne le groupement :~CnH2n~(-OC2H4-)x~(-OC3H6-)y~O— R3, R2 denotes the grouping: ~ CnH 2 n ~ (-OC2H 4 -) x ~ (-OC3H6-) y ~ O-R3,
R3 désigne un atome d'hydrogène ou un radical akyle linéaire ou ramifié comportant de 1 à 12 atomes de carbone ; R3 denotes a hydrogen atom or a linear or branched alkyl radical having from 1 to 12 carbon atoms;
a est un nombre entier allant de 1 à environ 500 ; a is an integer from 1 to about 500;
b désigne un nombre entier allant de 1 à environ 500 ; b is an integer from 1 to about 500;
n est un nombre entier allant de 2 à 12 et de préférence 2 à 5 ; n is an integer from 2 to 12 and preferably 2 to 5;
x désigne un nombre entier allant de 1 à environ 50 et de préférence de 1 à 30 ; y désigne un nombre entier allant de 0 à environ 49 et de préférence 0 à 29 sous réserve que lorsque y est différent de zéro le ratio x/y est supérieur à 1 et de préférence varie de 2 à 1 1 . x denotes an integer ranging from 1 to about 50 and preferably from 1 to 30; y denotes an integer ranging from 0 to about 49 and preferably 0 to 29 provided that when y is different from zero the ratio x / y is greater than 1 and preferably varies from 2 to 1 1.
Parmi les émulsionnants alkyldimethicone copolyols de formule (I) préférés, on citera plus particulièrement le CETYL PEG/PPG-10/1 DIMETHICONE et plus particulièrement le mélange CETYL PEG/PPG-10/1 DIMETICONE AND DIMETHICONE (nom INCI) comme le produit vendu sous le nom commercial ABIL EM90 par la société GOLDSCHMIDT ou bien le mélange (POLYGLYCERYL-4- STEARATE and CETYL PEG/PPG-10 (AND) DIMETHICONE (AND) HEXYL LAURATE) comme le produit vendu sous le nom commercial ABIL WE09 par la même société. Among the preferred alkyldimethicone copolyol emulsifiers of formula (I), there will be mentioned more particularly CETYL PEG / PPG-10/1 DIMETHICONE and more particularly the mixture CETYL PEG / PPG-10/1 DIMETICONE AND DIMETHICONE (INCI name) as the product sold under the trade name ABIL EM90 by GOLDSCHMIDT or the mixture (POLYGLYCERYL-4- STEARATE and CETYL PEG / PPG-10 (AND) DIMETHICONE (AND) HEXYL LAURATE) as the product sold under the trade name ABIL WE09 by the same society.
Parmi les émulsionnants eau-dans-huile, on peut citer également les dimethicone copolyols répondant à la formule (II) suivante CH3 - Si -Si -O Si - C+ Si (il) Among the water-in-oil emulsifiers, mention may also be made of dimethicone copolyols corresponding to the following formula (II) CH 3 - S i -Si -O Si - C + Si (ii)
CH. R. CH. R.
dans lesquelles in which
R4 désigne le groupement :~CmH2m~(-OC2H4-)s~(-OC3H6-)t~O— R5, R 4 denotes the grouping: ~ C m H 2 m ~ (-OC2H 4 -) s ~ (-OC3H6-) t ~ O-R 5 ,
R5 désigne un atome d'hydrogène ou un radical akyle linéaire ou ramifié comportant de 1 à 12 atomes de carbone ; R 5 denotes a hydrogen atom or a linear or branched alkyl radical having from 1 to 12 carbon atoms;
c est un nombre entier allant de 1 à environ 500 it is an integer ranging from 1 to about 500
d désigne un nombre entier allant de 1 à environ 500, d denotes an integer ranging from 1 to about 500,
m est un nombre entier allant de 2 à 12 et de préférence 2 à 5 , m is an integer ranging from 2 to 12 and preferably 2 to 5,
s désigne un nombre entier allant de 1 à environ 50, et de préférence de 1 à 30 ; t désigne un nombre entier allant de 0 à environ 50 et de préférence de 0 à 30 ; sous réserve que la somme s+t soit supérieure ou égal à 1 . s denotes an integer ranging from 1 to about 50, and preferably from 1 to 30; t denotes an integer ranging from 0 to about 50 and preferably from 0 to 30; provided that the sum s + t is greater than or equal to 1.
Parmi ces émulsionnants dimethicone copolyols de formule (II) préférentiels on utilisera particulièrement le PEG-18/PPG-18 DIMETHICONE et plus particulièrement le mélange CYCLOPENTASILOXANE (and) PEG-18/PPG-18 DIMETHICONE (nom INCI) tel que le produit vendu par la société Dow Corning sous la dénomination commerciale Silicone DC 5225 C ou KF-6040 de la société Shin Etsu. Selon une forme particulièrement préféré, on utilisera un mélange d'au moins un émulsionnant de formule (I) et d'au moins un émulsionnant de formule (II). Among these preferred emulsifiers dimethicone copolyols of formula (II), PEG-18 / PPG-18 DIMETHICONE and more particularly the mixture CYCLOPENTASILOXANE (and) PEG-18 / PPG-18 DIMETHICONE (INCI name) such as the product sold by Dow Corning under the trade name Silicone DC 5225 C or KF-6040 from Shin Etsu. According to a particularly preferred form, use will be made of a mixture of at least one emulsifier of formula (I) and at least one emulsifier of formula (II).
On utilisera plus particulièrement un mélange de PEG-18/PPG-18 Dimethicone et Cetyl PEG/PPG-10/1 DIMETHICONE et encore plus particulièrement un mélange de (CYCLOPENTASILOXANE (and) PEG-18/PPG-18 Dimethicone) et de Cetyl PEG/PPG-10/1 DIMETICONE and Dimethicone ou de (Polyglyceryl-4-stearate and Cetyl PEG/PPG-10 (and) Dimethicone (and) Hexyl Laurate). Parmi les émulsionnants eau-dans-huile, on peut citer également les émulsionnants non ioniques dérivés d'acide gras et de polyol, les alkylpolyglycosides (APG), les esters de sucres et leurs mélanges. Comme émulsionnants non ioniques dérivés d'acide gras et de polyol, on peut utiliser notamment les esters d'acide gras et de polyol, l'acide gras ayant notamment une chaîne alkyle en C8-C24, et les polyols étant par exemple le glycérol et le sorbitan. Comme esters d'acide gras et de polyol, on peut citer notamment les esters d'acide isostéarique et de polyols, les esters d'acide stéarique et de polyols, et leurs mélanges, en particulier les esters d'acide isostéarique et de glycérol et/ou de sorbitan. Comme esters d'acide stéarique et de polyols, on peut citer notamment les esters de polyéthylèneglycol comme le PEG-30 Dipolyhydroxystearate tel que le produit commercialisé sous le nom Arlacel P135 par la société ICI. A mixture of PEG-18 / PPG-18 Dimethicone and Cetyl PEG / PPG-10/1 DIMETHICONE and even more particularly a mixture of (CYCLOPENTASILOXANE (and) PEG-18 / PPG-18 Dimethicone) and Cetyl PEG will be used more particularly. / PPG-10/1 DIMETICONE and Dimethicone or (Polyglyceryl-4-stearate and Cetyl PEG / PPG-10 (and) Dimethicone (and) Hexyl Laurate). Among the water-in-oil emulsifiers, mention may also be made of nonionic emulsifiers derived from fatty acid and polyol, alkylpolyglycosides (APG), sugar esters and mixtures thereof. As nonionic emulsifiers derived from fatty acid and polyol, it is possible in particular to use the fatty acid and polyol esters, the fatty acid having in particular a C 8 -C 24 alkyl chain, and the polyols being, for example, glycerol and sorbitan. Examples of fatty acid and polyol esters that may be mentioned include esters of isostearic acid and of polyols, esters of stearic acid and of polyols, and mixtures thereof, in particular esters of isostearic acid and of glycerol, and / or sorbitan. As esters of stearic acid and of polyols, mention may in particular be made of polyethylene glycol esters such as PEG-30 dipolyhydroxystearate, such as the product sold under the name Arlacel P135 by the company ICI.
Comme esters de glycérol et/ou de sorbitan, on peut citer par exemple l'isostéarate de polyglycérol, tel que le produit commercialisé sous la dénomination Isolan Gl 34 par la société Goldschmidt ; l'isostéarate de sorbitan, tel que le produit commercialisé sous la dénomination Arlacel 987 par la société ICI ; l'isostéarate de sorbitan et le glycérol, tel que le produit commercialisé sous la dénomination Arlacel 986 par la société ICI, , le mélange d'isostéarate de sorbitan et d'isostéarate de polyglycérol (3 moles) commercialisé sous la dénomination Arlacel 1690 par la société Unigema. et leurs mélanges. Examples of glycerol and / or sorbitan esters that may be mentioned are polyglycerol isostearate, such as the product sold under the name Isolan Gl 34 by the company Goldschmidt; sorbitan isostearate, such as the product sold under the name Arlacel 987 by the company ICI; sorbitan isostearate and glycerol, such as the product sold under the name Arlacel 986 by the company ICI, the mixture of sorbitan isostearate and polyglycerol isostearate (3 moles) sold under the name Arlacel 1690 by the Unigema company. and their mixtures.
L'émulsionnant peut être choisi aussi parmi les alkylpolyglycosides ayant un HLB inférieur à 7, par exemple ceux représentés par la formule générale (1 ) suivante : The emulsifier may also be chosen from alkylpolyglycosides having an HLB of less than 7, for example those represented by the following general formula (1):
R-O-(G)x (1 ) dans laquelle R représente un radical alkyle ramifié et/ou insaturé, comportant de 14 à 24 atomes de carbone, G représente un sucre réduit comportant de 5 à 6 atomes de carbone, et x désigne une valeur allant de 1 à 10 et de préférence de 1 à 4, et G désigne notamment le glucose, le fructose ou le galactose. RO- (G) x (1) in which R represents a branched and / or unsaturated alkyl radical containing from 14 to 24 carbon atoms, G represents a reduced sugar containing from 5 to 6 carbon atoms, and x denotes a value ranging from 1 to 10 and preferably from 1 to 4, and G particularly denotes glucose, fructose or galactose.
Le radical alkyle insaturé peut comprendre une ou plusieurs insaturations éthyléniques, et en particulier une ou deux insaturations éthyléniques. The unsaturated alkyl radical may comprise one or more ethylenic unsaturations, and in particular one or two ethylenic unsaturations.
Comme alkylpolyglycosides de ce type, on peut citer les alkylpolyglucosides (G=glucose dans la formule (I)), et notamment les composés de formule (I) dans laquelle R représente plus particulièrement un radical oléyle (radical insaturé en C18) ou isostéaryle (radical saturé en C18), G désigne le glucose, x est une valeur allant de 1 à 2, notamment l'isostéaryl-glucoside, l'oléyl-glucoside et leurs mélanges. Cet alkylpolyglucoside peut être utilisé en mélange avec un co-émulsionnant, plus spécialement avec un alcool gras et notamment un alcool gras ayant la même chaîne grasse que celle de l'alkylpolyglucoside, c'est-à-dire comportant de 14 à 24 atomes de carbone et ayant une chaîne ramifiée et/ou insaturée, et par exemple l'alcool isostéarylique quand l'alkylpolyglucoside est l'isostéaryl-glucoside, et l'alcool oléylique quand l'alkylpolyglucoside est l'oleyl-glucoside, éventuellement sous forme d'une composition autoémulsionnante, comme décrit par exemple dans le document WO-A-92/06778. On peut utiliser par exemple le mélange d'isostéaryl-glucoside et d'alcool isostéarylique, commercialisé sous la dénomination Montanov WO 18 par la société SEPPIC ainsi que le mélange octyldodécanol et octyldodecylxyloside commercialisé sous la dénomination FLUDANOV 20X par la société SEPPIC. As alkylpolyglycosides of this type, mention may be made of alkylpolyglucosides (G = glucose in formula (I)), and in particular the compounds of formula (I) in which R represents more particularly an oleyl radical (unsaturated C18 radical) or isostearyl radical ( saturated C18 radical), G denotes glucose, x is a value ranging from 1 to 2, especially isostearyl glucoside, oleyl glucoside and mixtures thereof. This alkylpolyglucoside may be used in admixture with a co-emulsifier, more particularly with a fatty alcohol and in particular a fatty alcohol having the same fatty chain as that of the alkylpolyglucoside, that is to say comprising from 14 to 24 carbon atoms. carbon and having a branched and / or unsaturated chain, and for example isostearyl alcohol when the alkylpolyglucoside is isostearylglucoside, and oleyl alcohol when the alkylpolyglucoside is oleylglucoside, optionally in the form of a self-emulsifying composition, as described for example in WO-A-92/06778. For example, the mixture of isostearyl glucoside and isostearyl alcohol sold under the name Montanov WO 18 by the company SEPPIC and the mixture octyldodecanol and octyldodecylxyloside sold under the name FLUDANOV 20X by the company SEPPIC may be used.
On peut également citer les polyoléfines à terminaison succinique, comme les polyisobutylènes à terminaison succinique estérifiée et leurs sels, notamment les sels de diéthanolamine, tels que les produits commercialisés sous les dénominations Lubrizol 2724, Lubrizol 2722 et Lubrizol 5603 par la société Lubrizol ou le produit commercial CHEMCINNATE 2000. Mention may also be made of succinic-terminated polyolefins, such as ester-terminated polyisobutylenes and their salts, especially diethanolamine salts, such as the products sold under the names Lubrizol 2724, Lubrizol 2722 and Lubrizol 5603 by the company Lubrizol or the product CHEMCINNATE 2000.
La quantité totale en émulsionnants dans la composition sera de préférence dans la composition selon l'invention à des teneurs en matière active allant de 1 à 8% en poids et plus particulièrement de 2 à 6% en poids par rapport au poids total de la composition. The total amount of emulsifiers in the composition will preferably be in the composition according to the invention at contents of active material ranging from 1 to 8% by weight. weight and more particularly from 2 to 6% by weight relative to the total weight of the composition.
Phase grasse Fatty phase
Les compositions non rincées selon l'invention peuvent contenir au moins une phase liquide organique non-miscible dans l'eau appelée phase grasse. Celle-ci comprend en général un ou plusieurs composés hydrophobes qui rendent ladite phase non- miscible dans l'eau. Ladite phase est liquide (en l'absence d'agent structurant) à température ambiante (20-25 °C). De manière préférentielle, la phase organique liquide organique non-miscible dans l'eau conforme à l'invention est généralement constituée comprend généralement au moins une huile volatile et/ou une huile non volatile et éventuellement au moins un agent structurant. Par « huile », on entend un corps gras liquide à température ambiante (25 °C) et pression atmosphérique (760mm de Hg soit 105 Pa). L'huile peut être volatile ou non volatile. The non-rinsed compositions according to the invention may contain at least one immiscible organic liquid phase in water called the fatty phase. This generally comprises one or more hydrophobic compounds which render said phase immiscible in water. Said phase is liquid (in the absence of structuring agent) at ambient temperature (20-25 ° C.). Preferably, the organic liquid immiscible organic phase in accordance with the invention is generally constituted generally comprises at least one volatile oil and / or a non-volatile oil and optionally at least one structuring agent. By "oil" is meant a fatty substance that is liquid at ambient temperature (25 ° C.) and atmospheric pressure (760 mmHg, ie 105 Pa). The oil can be volatile or non-volatile.
Par « huile volatile », on entend au sens de l'invention une huile susceptible de s'évaporer au contact de la peau ou de la fibre kératinique en moins d'une heure, à température ambiante et pression atmosphérique. Les huiles volatiles de l'invention sont des huiles cosmétiques volatiles, liquides à température ambiante, ayant une pression de vapeur non nulle, à température ambiante et pression atmosphérique, allant en particulier de 0,13 Pa à 40 000 Pa (10"3 à 300 mm de Hg), en particulier allant de 1 ,3 Pa à 13 000 Pa (0,01 à 100 mm de Hg), et plus particulièrement allant de 1 ,3 Pa à 1300 Pa (0,01 à 10 mm de Hg). For the purposes of the invention, the term "volatile oil" means an oil capable of evaporating on contact with the skin or keratin fiber in less than one hour at ambient temperature and atmospheric pressure. The volatile oils of the invention are volatile cosmetic oils which are liquid at ambient temperature and have a non-zero vapor pressure at ambient temperature and atmospheric pressure, in particular from 0.13 Pa to 40,000 Pa (10 -3 to 300 mmHg), in particular ranging from 1.3 Pa to 13 000 Pa (0.01 to 100 mmHg), and more particularly ranging from 1.3 Pa to 1300 Pa (0.01 to 10 mmHg). ).
Par « huile non volatile », on entend une huile restant sur la peau ou la fibre kératinique à température ambiante et pression atmosphérique au moins plusieurs heures et ayant notamment une pression de vapeur inférieure à 10"3 mm de Hg (0,13 Pa). L'huile peut être choisie parmi toutes les huiles physiologiquement acceptables et en particulier physiologiquement acceptables, notamment les huiles minérales, animales, végétales, synthétiques ; en particulier les huiles hydrocarbonées et/ou siliconées et/ou fluorées volatiles ou non volatiles et leurs mélanges. By "non-volatile oil" is meant an oil remaining on the skin or keratin fiber at room temperature and atmospheric pressure for at least several hours and having in particular a vapor pressure of less than 10 -3 mmHg (0.13 Pa). . The oil may be chosen from all physiologically acceptable and in particular physiologically acceptable oils, in particular mineral, animal, vegetable and synthetic oils; in particular volatile and / or nonvolatile hydrocarbon and / or silicone and / or fluorinated oils and mixtures thereof.
Plus précisément, par « huile hydrocarbonée », on entend une huile comportant principalement des atomes de carbone et d'hydrogène et éventuellement une ou plusieurs fonctions choisies parmi les fonctions hydroxyle, ester, éther, carboxylique. Généralement, l'huile présente une viscosité de 0,5 à 100 000 mPa.s, de préférence de 50 à 50 000 mPa.s et de préférence encore de 100 à 30 000 mPa.s. More precisely, the term "hydrocarbon-based oil" means an oil comprising mainly carbon and hydrogen atoms and optionally one or more functional groups chosen from hydroxyl, ester, ether and carboxylic functions. Generally, the oil has a viscosity of 0.5 to 100,000 mPa.s, preferably 50 to 50,000 mPa.s and more preferably 100 to 30,000 mPa.s.
A titre d'exemple d'huile volatile utilisable dans l'invention, on peut citer : By way of example of volatile oil that may be used in the invention, mention may be made of:
les huiles hydrocarbonées volatiles choisies parmi les huiles hydrocarbonées ayant de 8 à 16 atomes de carbones, et notamment les isoalcanes en Cs-Ci6 d'origine pétrolière (appelées aussi isoparaffines) comme l'isododécane (encore appelé 2,2,4,4,6-pentaméthylheptane), l'isodécane, l'isohexadécane, et par exemple les huiles vendues sous les noms commerciaux d'Isopars ou de Permetyls, les esters ramifiés en Cs-Ci6, le néopentanoate d'iso-hexyle, et leurs mélanges. D'autres huiles hydrocarbonées volatiles comme les distillais de pétrole, notamment ceux vendus sous la dénomination Shell Soit par la société SHELL, peuvent aussi être utilisées ; les alcanes linéaires volatils comme ceux décrits dans la demande de brevet de la société Cognis DE10 2008 012 457. volatile hydrocarbon oils chosen from hydrocarbon-based oils containing from 8 to 16 carbon atoms, and in particular Cs-C16 isoalkanes of petroleum origin (also known as isoparaffins), such as isododecane (also called 2,2,4,4, 6-pentamethylheptane), isodecane, isohexadecane, and for example the oils sold under the trade names Isopars or permetyls, branched C 6 -C 16 esters, isohexyl neopentanoate, and mixtures thereof. Other volatile hydrocarbon oils such as petroleum distillates, especially those sold under the name Shell or by Shell, may also be used; volatile linear alkanes such as those described in the patent application of Cognis DE10 2008 012 457.
les silicones volatiles, comme par exemple les huiles de silicones linéaires ou cycliques volatiles, notamment celles ayant une viscosité < 8 centistokes (8 10"6 m2/s), et ayant notamment de 2 à 7 atomes de silicium, ces silicones comportant éventuellement des groupes alkyle ou aikoxy ayant de 1 à 10 atomes de carbone. Comme huile de silicone volatile utilisable dans l'invention, on peut citer notamment l'octaméthyl cyclotétrasiloxane, le décaméthyl cyclopentasiloxane, le dodécaméthyl cyclohexasiloxane, l'heptaméthyl hexyltrisiloxane, l'heptaméthyloctyl trisiloxane, l'hexaméthyl disiloxane, l'octaméthyl trisiloxane, le décaméthyl tétrasiloxane, le dodécaméthyl pentasiloxane volatile silicones, for example volatile linear or cyclic silicone oils, in particular those having a viscosity of <8 centistokes (8 × 10 -6 m 2 / s), and having in particular from 2 to 7 silicon atoms, these silicones possibly comprising Alkyl or alkoxy groups having from 1 to 10 carbon atoms As volatile silicone oil that may be used in the invention, mention may be made especially of octamethylcyclotetrasiloxane, decamethylcyclopentasiloxane, dodecamethylcyclohexasiloxane, heptamethylhexyltrisiloxane and heptamethyloctyl. trisiloxane, hexamethyl disiloxane, octamethyl trisiloxane, decamethyl tetrasiloxane, dodecamethylpentasiloxane
et leurs mélanges. On peut également citer les huiles linéaires alkyltrisiloxanes volatiles de formule générale (I) : and their mixtures. It is also possible to mention volatile alkyltrisiloxane linear oils of general formula (I):
où R représente un groupe alkyle comprenant de 2 à 4 atomes de carbone et dont un ou plusieurs atomes d'hydrogène peuvent être substitués par un atome de fluor ou de chlore. where R represents an alkyl group comprising from 2 to 4 carbon atoms and one or more hydrogen atoms of which may be substituted by a fluorine or chlorine atom.
Parmi les huiles de formule générale (I), on peut citer : Among the oils of general formula (I), mention may be made of:
le 3-butyl 1 ,1 ,1 ,3,5,5,5-heptaméthyl trisiloxane, 3-butyl 1,1,1,5,5,5-heptamethyltrisiloxane,
le 3-propyl 1 ,1 ,1 ,3,5,5,5-heptaméthyl trisiloxane, et 3-propyl-1,1,1,5,5,5-heptamethyltrisiloxane, and
le 3-éthyl 1 ,1 ,1 ,3,5,5,5-heptaméthyl trisiloxane, 3-ethyl-1,1,1,5,5,5-heptamethyltrisiloxane,
correspondant aux huiles de formule (I) pour lesquelles R est respectivement un groupe butyle, un groupe propyle ou un groupe éthyle. corresponding to the oils of formula (I) for which R is respectively a butyl group, a propyl group or an ethyl group.
A titre d'exemple d'huile non volatile utilisable dans l'invention, on peut citer : As an example of a non-volatile oil that may be used in the invention, mention may be made of:
les huiles hydrocarbonées d'origine animale telles que le perhydrosqualène ; les huiles hydrocarbonées végétales telles que les triglycérides liquides d'acides gras de 4 à 24 atomes de carbone comme les triglycérides des acides heptanoïque ou octanoïque ou encore les huiles les huiles de germe de blé, d'olive, l'huile d'amande douce, de palme, de colza, de coton, de luzerne, de pavot, de potimarron, de courge, de cassis, d'onagre, de millet, d'orge, de quinoa, de seigle, de carthame, de bancoulier, de passiflore, de rosier muscat, de tournesol, de maïs, de soja, de courge, de pépins de raisin, de sésame, de noisette, d'abricot, de macadamia, de ricin, d'avocat, les triglycérides des acides caprylique/caprique comme ceux vendus par la société Stearineries Dubois ou ceux vendus sous les dénominations Miglyol 810, 812 et 818 par la société Dynamit Nobel, l'huile de jojoba, de beurre de karité ; les hydrocarbures linéaires ou ramifiés, d'origine minérale ou synthétique tels que les huiles de paraffine et leurs dérivés, la vaseline, les polydécènes, les polybutènes, le polyisobutène hydrogéné tel que le Parleam, le squalane ; hydrocarbon oils of animal origin such as perhydrosqualene; vegetable hydrocarbon oils such as liquid triglycerides of fatty acids of 4 to 24 carbon atoms such as triglycerides of heptanoic or octanoic acids or oils oils of wheat germ, olive oil, sweet almond oil , palm, colza, cotton, alfalfa, poppy, pumpkin, squash, black currant, evening primrose, millet, barley, quinoa, rye, safflower, bancoulier, passionflower , musk rose, sunflower, maize, soya, squash, grape seed, sesame, hazelnut, apricot, macadamia, castor, avocado, caprylic / capric acid triglycerides as those sold by Stearineries Dubois or those sold under the names Miglyol 810, 812 and 818 by Dynamit Nobel, jojoba oil, shea butter; linear or branched hydrocarbons of mineral or synthetic origin such as liquid paraffins and derivatives thereof, petroleum jelly, polydecenes, polybutenes, hydrogenated polyisobutene such as Parleam, squalane;
les éthers de synthèse ayant de 10 à 40 atomes de carbone ; synthetic ethers having from 10 to 40 carbon atoms;
- les esters de synthèse notamment d'acides gras comme les huiles de formule R1COOR2 dans laquelle Ri représente le reste d'un acide gras supérieur linéaire ou ramifié comportant de 1 à 40 atomes de carbone et R2 représente une chaîne hydrocarbonée notamment ramifiée contenant de 1 à 40 atomes de carbone avec Ri + R2 > 10 comme par exemple l'huile de Purcellin (octanoate de cétostéaryle), l'isononanoate d'isononyle, le myristate d'isopropyle, le palmitate d'isopropyle, le benzoate d'alcool en C12 à C15, le laurate d'hexyle, l'adipate de diisopropyle, l'isononanoate d'isononyle, le palmitate d'éthyl 2-hexyle, le stéarate d'octyl 2- dodécyle, l'érucate d'octyl 2-dodécyle, l'isostéarate d'isostéaryle, le tridecyl trimellitate ; les octanoates, décanoates ou ricinoléates d'alcools ou de polyalcools comme le dioctanoate de propylène glycol ; les esters hydroxylés comme l'isostéaryl lactate, l'octyl hydroxy stéarate, l'hydroxy stéarate d'octyl dodécyle, le diisostéaryl malate, le citrate de triisocétyle, des heptanoates, octanoates, décanoates d'alcools gras ; des esters de polyol comme le dioctanoate de propylène glycol, le diheptanoate de néopentyl glycol, le diisononanoate de diéthylène glycol ; et les esters du pentaérythritol comme le tétra-isostéarate de pentaérythrytyle ; synthetic esters, in particular of fatty acids, such as the oils of formula R 1 COOR 2 in which R 1 represents the residue of a linear or branched higher fatty acid containing from 1 to 40 carbon atoms and R 2 represents a hydrocarbon chain, in particular branched, containing 1 to 40 carbon atoms with R 1 + R 2> 10 such as, for example, purcellin oil (cetostearyl octanoate), isononyl isononanoate, isopropyl myristate, isopropyl palmitate, alcohol benzoate C12 to C15, hexyl laurate, diisopropyl adipate, isononyl isononanoate, ethyl 2-hexyl palmitate, octyl 2-dodecyl stearate, octyl erucate 2- dodecyl, isostearyl isostearate, tridecyl trimellitate; octanoates, decanoates or ricinoleates of alcohols or polyalcohols such as propylene glycol dioctanoate; hydroxylated esters such as isostearyl lactate, octyl hydroxy stearate, octyl dodecyl hydroxystearate, diisostearyl malate, triisocetyl citrate, heptanoates, octanoates, decanoates of fatty alcohols; polyol esters such as propylene glycol dioctanoate, neopentyl glycol diheptanoate, diethylene glycol diisononanoate; and pentaerythritol esters such as pentaerythritol tetraisostearate;
- des alcools gras liquides à température ambiante à chaîne carbonée ramifiée et/ou insaturée ayant de 12 à 26 atomes de carbone comme l'octyl dodécanol, l'alcool isostéarylique, le 2-butyloctanol, le 2-hexyl décanol, le 2-undécyl pentadécanol, l'alcool oléique ; branched-chain and / or unsaturated carbon-chain liquid fatty alcohols having from 12 to 26 carbon atoms, such as octyl dodecanol, isostearyl alcohol, 2-butyloctanol, 2-hexyl decanol and 2-undecyl; pentadecanol, oleic alcohol;
- les acides gras supérieurs tels que l'acide oléique, l'acide linoléique, l'acide linolénique ; higher fatty acids such as oleic acid, linoleic acid, linolenic acid;
- les carbonates ; - carbonates;
- les acétates ; - acetates;
- les citrates ; - citrates;
- les huiles fluorées éventuellement partiellement hydrocarbonées et/ou siliconées comme les huiles fluorosiliconées, les polyéthers fluorés, les silicones fluorées telles que décrit dans le document EP-A-847752; - les huiles siliconées comme les polydiméthylsiloxanes (PDMS) non volatiles, linéaires ou cycliques ; les polydiméthylsiloxanes comportant des groupements alkyle, alcoxy ou phényle, pendant ou en bout de chaîne siliconée, groupements ayant de 2 à 24 atomes de carbone ; les silicones phénylées comme les phényl triméthicones, les phényl diméthicones, les phényl triméthylsiloxy diphényl siloxanes, les diphényl diméthicones, les diphényl méthyldiphényl trisiloxanes, les 2-phényl éthyl triméthyl-siloxysilicates, et fluorinated oils that may be partially hydrocarbon-based and / or silicone-based, such as fluorosilicone oils, fluorinated polyethers or fluorinated silicones, as described in document EP-A-847752; silicone oils, such as non-volatile, linear or cyclic polydimethylsiloxanes (PDMS); polydimethylsiloxanes comprising alkyl, alkoxy or phenyl groups, during or at the end of the silicone chain, groups having from 2 to 24 carbon atoms; phenyl silicones such as phenyl trimethicones, phenyl dimethicones, phenyl trimethylsiloxy diphenyl siloxanes, diphenyl dimethicones, diphenyl methyldiphenyl trisiloxanes, 2-phenylethyl trimethylsiloxysilicates, and
leurs mélanges. Agent structurant their mixtures. Structuring agent
Les compositions non rincées selon l'invention comprenant une phase grasse peuvent contenir en plus au moins un agent structurant de ladite phase grasse qui peut être choisi de préférence parmi les cires, les composés pâteux, les gélifiants lipophiles minéraux ou organiques et leurs mélanges. The non-rinsed compositions according to the invention comprising a fatty phase may additionally contain at least one structuring agent of said fatty phase, which may preferably be chosen from waxes, pasty compounds, mineral or organic lipophilic gelling agents and mixtures thereof.
Il est entendu que la quantité en ces composés peut être ajustée par l'homme du métier de manière à ne pas porter préjudice à l'effet recherché dans le cadre de la présente invention. It is understood that the amount of these compounds can be adjusted by those skilled in the art so as not to damage the desired effect in the context of the present invention.
Cire(s) Wax (es)
La cire est d'une manière générale un composé lipophile, solide à température ambiante (25 °C), à changement d'état solide/liquide réversible, ayant un point de fusion supérieur ou égal à 30 °C pouvant aller jusqu'à 200 °C et notamment jusqu'à 120 °C. The wax is generally a lipophilic compound, solid at room temperature (25 ° C.), with a reversible solid / liquid state change, having a melting point greater than or equal to 30 ° C. and up to 200 ° C and especially up to 120 ° C.
En particulier, les cires convenant à l'invention peuvent présenter un point de fusion supérieur ou égal à 45 °C, et en particulier supérieur ou égal à 55 °C. In particular, the waxes that are suitable for the invention may have a melting point greater than or equal to 45 ° C., and in particular greater than or equal to 55 ° C.
Au sens de l'invention, la température de fusion correspond à la température du pic le plus endothermique observé en analyse thermique (DSC) telle que décrite dans la norme ISO 1 1357-3 ; 1999. Le point de fusion de la cire peut être mesuré à l'aide d'un calorimètre à balayage différentiel (DSC), par exemple le calorimètre vendu sous la dénomination « MDSC 2920 » par la société TA Instruments. For the purposes of the invention, the melting temperature corresponds to the temperature of the most endothermic peak observed in thermal analysis (DSC) as described in the ISO 1 1357-3 standard; 1999. The melting point of wax can be measured using a differential scanning calorimeter (DSC), for example the calorimeter sold under the name "MDSC 2920" by the company TA Instruments.
Le protocole de mesure est le suivant : The measurement protocol is as follows:
Un échantillon de 5 mg de cire disposé dans un creuset est soumis à une première montée en température allant de -20 °C à 100 °C, à la vitesse de chauffe de 10 °C/minute, puis est refroidi de 100 °C à -20 °C à une vitesse de refroidissement de 10 °C/minute et enfin soumis à une deuxième montée en température allant de -20 °C à 100 °C à une vitesse de chauffe de 5 °C/minute. Pendant la deuxième montée en température, on mesure la variation de la différence de puissance absorbée par le creuset vide et par le creuset contenant l'échantillon de cire en fonction de la température. Le point de fusion du composé est la valeur de la température correspondant au sommet du pic de la courbe représentant la variation de la différence de puissance absorbée en fonction de la température. A sample of 5 mg of wax placed in a crucible is subjected to a first temperature rise from -20 ° C. to 100 ° C., at the heating rate of 10 ° C./min, and is then cooled from 100 ° C. to -20 ° C at a cooling rate of 10 ° C / minute and finally subjected to a second temperature rise from -20 ° C to 100 ° C at a heating rate of 5 ° C / minute. During the second temperature rise, the variation of the power difference absorbed by the empty crucible and the crucible containing the wax sample as a function of temperature is measured. The melting point of the compound is the value of the temperature corresponding to the peak apex of the curve representing the variation of the difference in power absorbed as a function of the temperature.
Les cires susceptibles d'être utilisées dans les compositions selon l'invention sont choisies parmi les cires solides à température ambiante d'origine animale, végétale, minérale ou de synthèse et leurs mélanges. The waxes that may be used in the compositions according to the invention are chosen from solid waxes at room temperature of animal, vegetable, mineral or synthetic origin, and mixtures thereof.
A titre illustratif des cires convenant à l'invention, on peut notamment citer les cires hydrocarbonées comme la cire d'abeille, la cire de lanoline, et les cires d'insectes de Chine, la cire de son de riz, la cire de Carnauba, la cire de Candellila, la cire d'Ouricury, la cire d'Alfa, la cire de berry, la cire de shellac, la cire du Japon et la cire de sumac; la cire de montan, les cires d'orange et de citron, Cire de Tournesol raffinée commercialisée sous la dénomination SUNFLOWER WAX par KOSTER KEUNEN, les cires microcristallines, les paraffines et l'ozokérite; les cires de polyéthylène, les cires obtenues par la synthèse de Fisher-Tropsch et les copolymères cireux ainsi que leurs esters. Illustrative waxes suitable for the invention include hydrocarbon waxes such as beeswax, lanolin wax, and Chinese insect wax, rice bran wax, Carnauba wax Candellila wax, Ouricury wax, Alfa wax, berry wax, shellac wax, Japanese wax and sumac wax; montan wax, orange and lemon waxes, refined sunflower wax marketed under the name SUNFLOWER WAX by KOSTER KEUNEN, microcrystalline waxes, paraffins and ozokerite; polyethylene waxes, waxes obtained by Fisher-Tropsch synthesis and waxy copolymers and their esters.
On peut aussi citer des cires obtenues par hydrogénation catalytique d'huiles animales ou végétales ayant des chaînes grasses, linéaires ou ramifiées, en Cs-C32- Parmi celles-ci, on peut notamment citer l'huile de jojoba isomérisée telle que l'huile de jojoba partiellement hydrogénée isomérisée trans fabriquée ou commercialisée par la société Désert Whale sous la référence commerciale lso-Jojoba-50®, l'huile de tournesol hydrogénée, l'huile de ricin hydrogénée, l'huile de coprah hydrogénée, l'huile de lanoline hydrogénée, et le tétrastéarate de di-(triméthylol-1 ,1 ,1 propane) vendu sous la dénomination de Hest 2T-4S® par la société HETERENE. Mention may also be made of waxes obtained by catalytic hydrogenation of animal or vegetable oils having linear or branched C 8 -C 32 fatty chains. Among these, mention may be made especially of isomericized jojoba oil such as oil. trans-isomerized partially hydrogenated jojoba manufactured or marketed by Desert Whale under the trade name lso-Jojoba-50®, hydrogenated sunflower oil, hydrogenated castor oil, hydrogenated coconut oil, coconut oil hydrogenated lanolin, and the di- (trimethylol-1, 1, 1 propane) tetrastearate sold under the name Hest 2T-4S® by the company HETERENE.
On peut encore citer les cires de silicone (C3o-45 ALKYL DIMETHICONE), les cires fluorées. On peut également utiliser les cires obtenues par hydrogénation d'huile de ricin estérifiée avec l'alcool cétylique vendues sous les dénominations de Phytowax ricin 16L64® et 22L73® par la société SOPHIM. De telles cires sont décrites dans la demande FR-A- 2792190. Comme cire, on peut utiliser un (hydroxystéaryloxy)stéarate d'alkyle en C2o-C4o (le groupe alkyle comprenant de 20 à 40 atomes de carbone), seul ou en mélange. Mention may also be made of silicone waxes (C 3 -C 4 ALKYL DIMETHICONE) and fluorinated waxes. It is also possible to use the waxes obtained by hydrogenation of castor oil esterified with cetyl alcohol sold under the names Phytowax Ricin 16L64® and 22L73® by the company SOPHIM. Such waxes are described in the application FR-A-2792190. As wax, a C 2 -C 4 (alkyl group comprising 20 to 40 carbon atoms) can be used as a wax (hydroxystearyloxy) stearate, alone or in mixture.
Une telle cire est notamment vendue sous les dénominations « Kester Wax K 82 P® », « Hydroxypolyester K 82 P® » et « Kester Wax K 80 P® » par la société KOSTER KEUNEN. Such a wax is especially sold under the names "Kester Wax K 82 P®", "Hydroxypolyester K 82 P®" and "Kester Wax K 80 P®" by the company Koster Keunen.
Comme micro-cires pouvant être utilisées dans les compositions selon l'invention, on peut citer notamment les micro cires de carnauba telles que celle commercialisée sous la dénomination de MicroCare 350® par la société MICRO POWDERS, les micro cires de cire synthétique telles que celle commercialisée sous la dénomination de MicroEase 1 14S® par la société MICRO POWDERS, les micro cires constituées d'un mélange de cire de carnauba et de cire de polyéthylène telles que celles commercialisées sous les dénominations de Micro Care 300® et 310® par la société MICRO POWDERS, les micro cires constituées d'un mélange de cire de carnauba et de cire synthétique telles que celle commercialisée sous la dénomination Micro Care 325® par la société MICRO POWDERS, les micro cires de polyéthylène telles que celles commercialisées sous les dénominations de Micropoly 200®, 220®, 220L® et 250S® par la société MICRO POWDERS, les produits commerciaux PERFOMALEN 400 POLYETHYLENE et PERFORMALENE 500-L POLYETHYLENE de NEW PHASE TECHNOLOGIES, le PERFORMALENE 655 POLYETHYLENE ou les cires de paraffine comme la cire ayant pour nom INCI , MICROCRISTALLINE WAX and SYNTHETIC WAX et vendue sous le nom commercial MICROLEASE par la Société SOCHIBO. ; les micro cires de polytétrafluoroéthylène telles que celles commercialisées sous les dénominations de Microslip 519® et 519 L® par la société MICRO POWDERS. La composition selon l'invention comprendra de préférence une teneur en cire(s) allant de 3 % à 20% en poids par rapport au poids total de la composition, en particulier de 5 à 15%, plus particulièrement de 6 à 15%. As micro-waxes that can be used in the compositions according to the invention, mention may be made in particular of carnauba micro-waxes such as that marketed under the name MicroCare 350® by the company MICRO POWDERS, and synthetic wax micro-waxes such as marketed under the name MicroEase 1 14S® by the company MICRO POWDERS, micro waxes consisting of a mixture of carnauba wax and polyethylene wax such as those marketed under the names Micro Care 300® and 310® by the company MICRO POWDERS, micro waxes consisting of a mixture of carnauba wax and synthetic wax such as that sold under the name Micro Care 325® by MICRO POWDERS, polyethylene micro-waxes such as those sold under the names Micropoly. 200®, 220®, 220L® and 250S® by the company MICRO POWDERS, the commercial products PERFOMALEN 400 POLYETHYLENE and PERFORMALENE 500-L POLYETHYLENE from NEW PHASE TECHNOLOGIES, the PERFORMALENE 655 POLYETHYLENE or the paraffin waxes such as the wax INCI, MICROCRYSTALLINE WAX and SYNTHETIC WAX and sold under the trade name MICROLEASE by SOCHIBO. ; polytetrafluoroethylene micro waxes such as those sold under the names Microslip 519® and 519 L® by the company Micro Powders. The composition according to the invention will preferably comprise a wax content (s) ranging from 3% to 20% by weight relative to the total weight of the composition, in particular from 5% to 15%, more particularly from 6% to 15%.
Selon une forme particulière de l'invention, dans le cadre des compositions solides anhydres sous forme de stick, on utilisera des micro-cires de polyéthylène sous forme de cristallites de facteur de forme au moins égal à 2 ayant un point de fusion allant de 70 à 1 10°C et de préférence 70 à 100°C, afin de réduire voire supprimer la présence de strates dans la composition solide, Ces cristallites en aiguilles et notamment leurs dimensions peuvent être caractérisées visuellement selon la méthode suivante. According to one particular form of the invention, in the context of the anhydrous solid compositions in the form of a stick, use will be made of polyethylene micro-waxes in the form of shape-factor crystallites of at least 2 having a melting point of 70.degree. at 1 10 ° C and preferably 70 to 100 ° C, to reduce or eliminate the presence of layers in the solid composition, These needle crystallites and in particular their dimensions can be characterized visually according to the following method.
La cire est déposée sur une lame de microscope, laquelle est posée sur une platine chauffante. La lame et la cire sont chauffées à une température généralement au moins supérieure de 5 °C à celle du point de fusion de la cire ou du mélange de cire considéré(e). A la fin de la fonte, le liquide ainsi obtenu et la lame de microscope sont laissés refroidir pour se solidifier. L'observation des cristallites est réalisée à l'aide d'un microscope optique de type Leica DMLB100, avec un objectif sélectionné en fonction de la taille des objets à visualiser, et en lumière polarisée. Les dimensions des cristallites sont mesurées à l'aide d'un logiciel d'analyse d'images tel que ceux commercialisés par la société Microvision. Les cires de polyéthylène cristallites conformes à l'invention possèdent de préférence une longueur moyenne allant de 5 à 10 μιτι. Par « longueur moyenne », on désigne la dimension donnée par la distribution granulométrique statistique à la moitié de la population, dite D50. The wax is deposited on a microscope slide, which is placed on a hot plate. The blade and the wax are heated to a temperature generally at least 5 ° C higher than that of the melting point of the wax or wax mixture considered (e). At the end of the melting, the liquid thus obtained and the microscope slide are allowed to cool to solidify. The observation of the crystallites is carried out using a Leica DMLB100 type optical microscope, with a target selected according to the size of the objects to be viewed, and in polarized light. The dimensions of the crystallites are measured using an image analysis software such as those marketed by Microvision. The crystallite polyethylene waxes according to the invention preferably have an average length ranging from 5 to 10 μιτι. By "average length" is meant the dimension given by the statistical size distribution at half of the population, called D50.
On utilisera plus particulièrement un mélange de cires PERFOMALEN 400 POLYETHYLENE et PERFORMALENE 500-L POLYETHYLENE de NEW PHASE TECHNOLOGIES Composés pâteux More particularly, a mixture of PERFOMALEN 400 POLYETHYLENE and PERFORMALENE 500-L POLYETHYLENE waxes from NEW PHASE TECHNOLOGIES will be used.
Par « composé pâteux » au sens de la présente invention, on entend un composé gras lipophile à changement d'état solide/liquide réversible, présentant à l'état solide une organisation cristalline anisotrope, et comportant à la température de 23 °C une fraction liquide et une fraction solide. For the purposes of the present invention, the term "pasty compound" is intended to mean a lipophilic fat compound with a reversible solid / liquid state change, having in the solid state an anisotropic crystalline organization and comprising at a temperature of 23 ° C. a fraction liquid and a solid fraction.
Le composé pâteux est de préférence choisi parmi les composés synthétiques et les composés d'origine végétale. Un composé pâteux peut être obtenu par synthèse à partir de produits de départ d'origine végétale. The pasty compound is preferably chosen from synthetic compounds and compounds of plant origin. A pasty compound can be obtained synthetically from starting materials of plant origin.
Le composé pâteux peut avantageusement choisi parmi : The pasty compound may advantageously be chosen from:
- la lanoline et ses dérivés, - lanolin and its derivatives,
- les composés siliconés polymères ou non, polymeric or non-polymeric silicone compounds,
- les composés fluorés polymères ou non, polymeric or non-polymeric fluorinated compounds,
- les polymères vinyliques, notamment : vinyl polymers, in particular:
- les homopolymères d'oléfines, homopolymers of olefins,
- les copolymères d'oléfines, copolymers of olefins,
- les homopolymères et copolymères de diènes hydrogénés, homopolymers and copolymers of hydrogenated dienes,
- les oligomères linéaires ou ramifiés, homo ou copolymères de (méth)acrylates d'alkyles ayant de préférence un groupement alkyle en C8-C30, linear or branched oligomers, homo or copolymers of alkyl (meth) acrylates preferably having a C 8 -C 30 alkyl group,
- les oligomères homo et copolymères d'esters vinyliques ayant des groupements alkyles en C8-C30, et - les oligomères homo et copolymères de vinyléthers ayant des groupements alkyles homo and copolymeric oligomers of vinyl esters having C 8 -C 30 alkyl groups, and Homo and copolymer oligomers of vinyl ethers having alkyl groups
- les polyéthers liposolubles résultant de la polyéthérification entre un ou plusieurs diols en C2-C100, de préférence en C2-C50, the liposoluble polyethers resulting from the polyetherification between one or more C2-C100, preferably C2-C50, diols,
- les esters, the esters,
- leurs mélanges. - their mixtures.
Parmi les esters, on préfère notamment : Among the esters, it is particularly preferred:
- les esters d'un glycérol oligomère, notamment les esters de diglycérol, en particulier les condensais d'acide adipique et de glycérol, pour lesquels une partie des groupes hydroxyles des glycérols ont réagi avec un mélange d'acides gras tels que l'acide stéarique, l'acide caprique, l'acide stéarique et l'acide isostéarique et l'acide 12- hydroxystéarique, à l'image notamment de ceux commercialisé sous la marque Softisan 649 par la société Sasol, esters of an oligomeric glycerol, in particular the diglycerol esters, in particular the adipic acid and glycerol condensates, for which part of the hydroxyl groups of the glycerols have reacted with a mixture of fatty acids such as the acid stearic acid, capric acid, stearic acid and isostearic acid and 12-hydroxystearic acid, especially like those marketed under the trademark Softisan 649 by the company Sasol,
- le propionate d'arachidyle commercialisé sous la marque Waxenol 801 par Alzo,arachidyl propionate sold under the name Waxenol 801 by Alzo,
- les esters de phytostérol, the phytosterol esters,
- les triglycérides d'acides gras et leurs dérivés, triglycerides of fatty acids and their derivatives,
- les esters de pentaérythritol, pentaerythritol esters,
- les polyesters non réticulés résultant de la polycondensation entre un acide dicarboxylique ou un polyacide carboxylique linéaire ou ramifié en C4-C50 et un diol ou un polyol en C2-C50, non-crosslinked polyesters resulting from the polycondensation between a linear or branched C 4 -C 50 dicarboxylic acid or polycarboxylic acid and a C2-C50 diol or polyol,
- les esters aliphatiques d'ester résultant de l'estérification d'un ester d'acide hydroxycarboxylique aliphatique par un acide carboxylique aliphatique, aliphatic ester esters resulting from the esterification of an aliphatic hydroxycarboxylic acid ester with an aliphatic carboxylic acid,
- les polyesters résultant de l'estérification, par un acide polycarboxylique, d'un ester d'acide hydroxy carboxylique aliphatique, ledit ester comprenant au moins deux groupes hydroxyle tels que les produits Risocast DA-H ®, et Risocast DA-L ®, polyesters resulting from the esterification, by a polycarboxylic acid, of an aliphatic hydroxycarboxylic acid ester, said ester comprising at least two hydroxyl groups such as the Risocast DA-H ® and Risocast DA-L ® products,
- les esters de dimère diol et dimère diacide, le cas échéant, estérifiés sur leur(s) fonction(s) alcool(s) ou acide(s) libre(s) par des radicaux acides ou alcools tels que le Plandool-G, the esters of diol dimer and diacid dimer, if appropriate, esterified on their (their) function (s) alcohol (s) or acid (s) free (s) by acidic radicals or alcohols such as Plandool-G,
- leurs mélanges. Parmi les composés pâteux d'origine végétale, on choisira de préférence un mélange de stérols de soja et de pentaérythritol oxyéthyléné (5OE) oxypropyléné (5 OP), commercialisé sous la référence Lanolide par la société VEVY. Gélifiants lipophiles - their mixtures. Among the pasty compounds of plant origin, a mixture of soy sterols and oxyethylenated (5OE) oxypropylene pentaerythritol (5 PO), marketed under the reference Lanolide by the company VEVY, will preferably be chosen. Lipophilic gelling agents
Gélifiants minéraux Mineral gelling agents
Comme gélifiant lipophile minéral, on peut citer les argiles éventuellement modifiées comme les hectorites modifiées par un chlorure d'ammonium en C10 à C22, comme l'hectorite modifiée par du chlorure de di-stéaryl di-méthyl ammonium telle que, par exemple, celle commercialisée sous la dénomination de Bentone 38V® par la société ELEMENTIS. On peut également citer la silice pyrogénée éventuellement traitée hydrophobe en surface dont la taille des particules est inférieure à 1 μιτι. Il est en effet possible de modifier chimiquement la surface de la silice, par réaction chimique générant une diminution du nombre de groupes silanol présents à la surface de la silice. On peut notamment substituer des groupes silanol par des groupements hydrophobes : on obtient alors une silice hydrophobe. Les groupements hydrophobes peuvent être des groupements triméthylsiloxyle, qui sont notamment obtenus par traitement de silice pyrogénée en présence de l'hexaméthyldisilazane. Des silices ainsi traitées sont dénommées « Silica silylate » selon le CTFA (8ème édition, 2000). Elles sont par exemple commercialisées sous les références Aerosil R812® par la société DEGUSSA, CAB-O-SIL TS-530® par la société CABOT, des groupements diméthylsilyloxyle ou polydiméthylsiloxane, qui sont notamment obtenus par traitement de silice pyrogénée en présence de polydiméthylsiloxane ou du diméthyldichlorosilane. Des silices ainsi traitées sont dénommées « Silica diméthyl silylate » selon le CTFA (8ème édition, 2000). Elles sont par exemple commercialisées sous les références Aerosil R972®, et Aerosil R974® par la société DEGUSSA, CAB-O-SIL TS-610® et CAB-O-SIL TS-720® par la société CABOT. La silice pyrogénée hydrophobe présente en particulier une taille de particules pouvant être nanométrique à micrométrique, par exemple allant d'environ de 5 à 200 nm. Gélifiants organiques As inorganic lipophilic gelling agents, there may be mentioned optionally modified clays, such as hectorites modified with a C10 to C22 ammonium chloride, such as hectorite modified with di-stearyl-dimethyl ammonium chloride such as, for example, sold under the name Bentone 38V® by the company ELEMENTIS. It is also possible to mention fumed silica optionally treated with hydrophobic surface, the particle size of which is less than 1 μιτι. It is indeed possible to chemically modify the surface of the silica, by chemical reaction generating a decrease in the number of silanol groups present on the surface of the silica. In particular, it is possible to substitute silanol groups with hydrophobic groups: a hydrophobic silica is then obtained. The hydrophobic groups may be trimethylsiloxyl groups, which are especially obtained by treatment of fumed silica in the presence of hexamethyldisilazane. Silicas thus treated are called "Silica silylate" according to the CTFA (8th edition, 2000). They are for example sold under the references Aerosil R812® by the company Degussa, Cab-O-Sil TS-530® by the company Cabot, dimethylsilyloxyl or polydimethylsiloxane groups, which are especially obtained by treatment of fumed silica in the presence of polydimethylsiloxane or dimethyldichlorosilane. Silicas thus treated are called "Silica dimethyl silylate" according to the CTFA (8th edition, 2000). They are for example marketed under the references Aerosil R972®, and Aerosil R974® by the company DEGUSSA, CAB-O-SIL TS-610® and CAB-O-SIL TS-720® by CABOT. The hydrophobic fumed silica has in particular a particle size that can be nanometric to micrometric, for example ranging from about 5 to 200 nm. Organic gelling agents
Les gélifiants lipophiles organiques polymériques sont par exemple les organopolysiloxanes élastomériques partiellement ou totalement réticulés, de structure tridimensionnelle, comme ceux commercialisés sous les dénominations de KSG6®, KSG16® et de KSG18® par la société SHIN-ETSU, de Trefil E-505C® et Trefil E-506C® par la société DOW-CORNING, de Gransil SR-CYC®, SR DMF10®, SR-DC556®, SR 5CYC gel®, SR DMF 10 gel® et de SR DC 556 gel® par la société GRANT INDUSTRIES, de SF 1204® et de JK 1 13® par la société GENERAL ELECTRIC ; l'éthylcellulose comme celle vendue sous la dénomination Ethocel® par la société DOW CHEMICAL ; les galactommananes comportant de un à six, et en particulier de deux à quatre, groupes hydroxyle par ose, substitués par une chaîne alkyle saturée ou non, comme la gomme de guar alkylée par des chaînes alkyle en C1 à C6, et en particulier en C1 à C3 et leurs mélanges. Les copolymères séquencés de type « dibloc », « tribloc » ou « radial » du type polystyrène/polyisoprène, polystyrène/polybutadiène tels que ceux commercialisés sous la dénomination Luvitol HSB® par la société BASF, du type polystyrène/copoly(éthylène-propylène) tels que ceux commercialisés sous la dénomination de Kraton® par la société SHELL CHEMICAL CO ou encore du type polystyrène/copoly(éthylène-butylène), les mélanges de copolymères tribloc et radial (en étoile) dans l'isododécane tels que ceux commercialisé par la société PENRECO sous la dénomination Versagel® comme par exemple le mélange de copolymère tribloc butylène/éthylène/styrène et de copolymère étoile éthylène/propylène/styrène dans l'isododécane (Versagel M 5960). Comme gélifiant lipophile, on peut encore citer les polymères de masse moléculaire moyenne en poids inférieure à 100 000, comportant a) un squelette polymérique ayant des motifs de répétition hydrocarbonés pourvus d'au moins un hétéroatome, et éventuellement b) au moins une chaîne grasse pendante et/ou au moins une chaîne grasse terminale éventuellement fonctionnalisées, ayant de 6 à 120 atomes de carbone et étant liées à ces motifs hydrocarbonés, telles que décrites dans les demandes WO-A-02/056847, WO-A-02/47619 dont le contenu est incorporé à titre de référence; en particulier les résines de polyamides (notamment comprenant des groupes alkyles ayant de 12 à 22 atomes de carbone) telles que celles décrites dans US-A-5783657 dont le contenu est incorporé à titre de référence. Polymeric organic lipophilic gelling agents are, for example, partially or fully crosslinked elastomeric organopolysiloxanes of three-dimensional structure, such as those marketed under the names KSG6®, KSG16® and KSG18® by the company SHIN-ETSU, by Trefil E-505C® and Trefil E-506C® by DOW-CORNING, Gransil SR-CYC®, SR DMF10®, SR-DC556®, SR 5CYC gel®, SR DMF 10 gel® and SR DC 556 gel® by GRANT INDUSTRIES , SF 1204® and JK 1 13® by the company GENERAL ELECTRIC; ethylcellulose such as that sold under the name Ethocel® by Dow Chemical; galactomannans having from one to six, and in particular from two to four, hydroxyl groups per sac, substituted by a saturated or unsaturated alkyl chain, such as guar gum alkylated by C1-C6 alkyl chains, and in particular C1 groups; C3 and mixtures thereof. Block copolymers of the "diblock", "triblock" or "radial" type of the polystyrene / polyisoprene, polystyrene / polybutadiene type, such as those sold under the name Luvitol HSB® by the company BASF, of the polystyrene / copoly (ethylene-propylene) type such as those sold under the name Kraton® by Shell Chemical Co., or else from the polystyrene / copoly (ethylene-butylene) type, mixtures of triblock and radial (star) copolymers in isododecane, such as those marketed by the company PENRECO company under the name Versagel® such as, for example, the mixture of butylene / ethylene / styrene triblock copolymer and ethylene / propylene / styrene star copolymer in isododecane (Versagel M 5960). Lipophilic gelling agents that may also be mentioned include polymers having a weight average molecular weight of less than 100,000, comprising a) a polymer backbone having hydrocarbon-based repeating units provided with at least one heteroatom, and optionally b) at least one fatty chain. pendant and / or at least one chain optionally functionalized fatty acid, having from 6 to 120 carbon atoms and being bonded to these hydrocarbon-based units, as described in applications WO-A-02/056847, WO-A-02/47619, the content of which is incorporated as a reference; in particular polyamide resins (especially comprising alkyl groups having from 12 to 22 carbon atoms) such as those described in US-A-5783657 whose contents are incorporated by reference.
Parmi les gélifiants lipophiles pouvant être utilisés dans les compositions selon l'invention, on peut encore citer les esters de dextrine et d'acide gras, tels que les palmitates de dextrine, notamment tels que ceux commercialisés sous les dénominations Rheopearl TL® ou Rheopearl KL® par la société CHIBA FLOUR. On peut également utiliser les polyamides siliconés du type polyorganosiloxane tels que ceux décrits dans les documents US-A-5,874,069, US-A-5,919,441 , US-A-6,051 ,216 et US-A-5,981 ,680. Among the lipophilic gelling agents that can be used in the compositions according to the invention, mention may also be made of dextrin and fatty acid esters, such as dextrin palmitates, in particular such as those sold under the names Rheopearl TL® or Rheopearl KL. ® by the company CHIBA FLOUR. It is also possible to use polyorganosiloxane-type silicone polyamides such as those described in US-A-5,874,069, US-A-5,919,441, US-A-6,051,216 and US-A-5,981,680.
Ces polymères siliconés peuvent appartenir aux deux familles suivantes : These silicone polymers can belong to the following two families:
- des polyorganosiloxanes comportant au moins deux groupes capables d'établir des interactions hydrogène, ces deux groupes étant situés dans la chaîne du polymère, et/ou polyorganosiloxanes comprising at least two groups capable of establishing hydrogen interactions, these two groups being located in the polymer chain, and / or
- des polyorganosiloxanes comportant au moins deux groupes capables d'établir des interactions hydrogène, ces deux groupes étant situés sur des greffons ou ramifications. polyorganosiloxanes comprising at least two groups capable of establishing hydrogen interactions, these two groups being located on grafts or branches.
Sels ou complexes anti-transpirants Salts or antiperspirant complexes
Selon une forme particulière de l'invention, les compositions de l'invention peuvent contenir au moins un sel ou complexe anti-transpirant. According to one particular form of the invention, the compositions of the invention may contain at least one salt or antiperspirant complex.
Par "agent de traitement de la transpiration", on entend toute substance qui, à elle seule, a pour effet de diminuer la sensation sur la peau d'humidité liée à la sueur humaine, de masquer la sueur humaine. Les sels ou complexes anti-transpirants conformes à l'invention sont choisis généralement parmi les sels ou complexes d'aluminium et/ou de zirconium. Il sont de préférence choisis parmi les halohydrates d'aluminium ; les halohydrates d'aluminium et de zirconium, les complexes d'hydroxychlorure de zirconium et d'hydroxychlorure d'aluminium avec ou sans un acide aminé tels que ceux décrits dans le brevet US- 3792068. By "sweat-treating agent" is meant any substance which, by itself, has the effect of diminishing the sensation on the skin of moisture associated with human sweat, of masking human sweat. The antiperspirant salts or complexes in accordance with the invention are generally chosen from aluminum or zirconium salts or complexes. They are preferably chosen from aluminum halohydrates; aluminum and zirconium halohydrates, complexes of zirconium hydroxychloride and of aluminum hydroxychloride with or without an amino acid such as those described in patent US-3792068.
Parmi les sels d'aluminium, on peut citer en particulier le chlorhydrate d'aluminium sous forme activée ou non activée, l'aluminium chlorohydrex, le complexe aluminium chlorohydrex polyéthylèneglycol, le complexe aluminium chlorohydrex propylèneglycol, l'aluminium dichlorohydrate, le complexe aluminium dichlorohydrex polyéthylèneglycol, le complexe aluminium dichlorohydrex propylèneglycol, l'aluminium sesquichlorohydrate, le complexe aluminium sesquichlorohydrex polyéthylèneglycol, le complexe aluminium sesquichlorohydrex propylèneglycol, le sulfate d'aluminium tamponné par le lactate de sodium et d'aluminium. Among the aluminum salts, mention may in particular be made of aluminum chlorohydrate in activated or non-activated form, aluminum chlorohydrex, chlorohydrex polyethylene glycol aluminum complex, aluminum chlorohydrexpropyleneglycol complex, aluminum dichlorohydrate, aluminum dichlorohydrex complex polyethylene glycol, aluminum complex dichlorohydrex propylene glycol, sesquichlorohydrate aluminum, sesquichlorohydrex aluminum polyethylene glycol complex, sesquichlorohydrex propylene glycol aluminum complex, aluminum sulphate buffered with sodium aluminum lactate.
Parmi les sels d'aluminium et de zirconium, on peut citer en particulier l'aluminium zirconium octachlorohydrate, l'aluminium zirconium pentachlorohydrate, l'aluminium zirconium tetrachlorohydrate, l'aluminium zirconium trichlorohydrate. Among the aluminum and zirconium salts, there may be mentioned in particular aluminum zirconium octachlorohydrate, aluminum zirconium pentachlorohydrate, aluminum zirconium tetrachlorohydrate, aluminum zirconium trichlorohydrate.
Les complexes d'hydroxychlorure de zirconium et d'hydroxychlorure d'aluminium avec un acide aminé sont généralement connus sous l'appellation ZAG (lorsque l'acide aminé est la glycine). Parmi ces produits on peut citer les complexes aluminium zirconium octachlorohydrex glycine, aluminium zirconium pentachlorohydrex glycine, aluminium zirconium tetrachlorohydrex glycine et aluminium zirconium trichlorohydrex glycine. The complexes of zirconium hydroxychloride and aluminum hydroxychloride with an amino acid are generally known under the name ZAG (when the amino acid is glycine). Among these products, mention may be made of aluminum complexes of zirconium octachlorohydrex glycine, aluminum zirconium pentachlorohydrex glycine, aluminum zirconium tetrachlorohydrex glycine and aluminum zirconium trichlorohydrex glycine.
Les sels ou complexes anti-transpirants peuvent être présents dans la composition selon l'invention à raison d'environ 0,5 à 25% en poids par rapport au poids total de la composition. The antiperspirant salts or complexes may be present in the composition according to the invention in a proportion of approximately 0.5 to 25% by weight relative to the total weight of the composition.
Actifs déodorants Les compositions selon l'invention peuvent également contenir en plus un ou plusieurs actifs déodorants additionnels. Deodorant active ingredients The compositions according to the invention may also contain one or more additional deodorant active agents.
On appelle « actif déodorant », toute substance capable de masquer, absorber, améliorer et/ou réduire l'odeur désagréable résultant de la décomposition de la sueur humaine par des bactéries Deodorant active means any substance capable of masking, absorbing, improving and / or reducing the unpleasant odor resulting from the decomposition of human sweat by bacteria
Les actifs déodorants peuvent être des agents bactériostatiques ou des agents bactéricides agissant sur les germes des odeurs axillaires, comme le 2,4,4'-trichloro- 2'-hydroxydiphényléther (®Triclosan), le 2,4-dichloro-2'-hydroxydiphényléther, le 3',4',5'-trichlorosalicylanilide, la 1 -(3',4'-dichlorophenyl)-3-(4'-chlorophenyl)urée (®Triclocarban) ou le 3,7,1 1 -triméthyldodéca-2, 5,10-triénol (®Farnesol) ; les sels d'ammonium quaternaires comme les sels de cetyltrimethylammonium, les sels de cétylpyridinium , le DPTA (acide 1 ,3-diaminopropanetétraacétique), le 1 ,2 decanediol (SYMCLARIOL de la société Symrise), les dérivés de glycérine comme par exemple le Caprylic/Capric Glycerides (CAPMUL MCM de Abitec), le Caprylate ou caprate de Glycerol (DERMOSOFT GMCY et DERMOSOFT GMC respectivement de STRAETMANS), le Polyglyceryl-2 Caprate (DERMOSOFT DGMC de STRAETMANS) les dérivés de biguanide comme les sels de polyhexaméthylène biguanide, la chlorhexidine et ses sels; 4-Phenyl-4,4-dimethyl-2butanol (SYMDEO MPP de Symrise). The deodorant active agents may be bacteriostatic agents or bactericidal agents acting on the germs of axillary odors, such as 2,4,4'-trichloro-2'-hydroxydiphenyl ether ( ® Triclosan), 2,4-dichloro-2'- hydroxydiphenyl ether, 3 ', 4', 5'-trichlorosalicylanilide, 1 - (3 ', 4'-dichlorophenyl) -3- (4'-chlorophenyl) urea ( ® Triclocarban) or 3,7,1 1 -trimethyldodeca -2, 5,10-trienol (Farnesol ®); quaternary ammonium salts such as cetyltrimethylammonium salts, cetylpyridinium salts, DPTA (1,3-diaminopropanetetraacetic acid), 1,2-decanediol (SYMCLARIOL from Symrise), glycerin derivatives, for example Caprylic acid; / Capric Glycerides (CAPMUL MCM from Abitec), Caprylate or Glycerol caprate (DERMOSOFT GMCY and DERMOSOFT GMC respectively from STRAETMANS), Polyglyceryl-2 Caprate (DERMOSOFT DG ™ from STRAETMANS) Biguanide derivatives such as polyhexamethylene biguanide salts, chlorhexidine and its salts; 4-Phenyl-4,4-dimethyl-2-butanol (SYMDEO MPP from Symrise).
Parmi les actifs déodorants conformes à l'invention, on peut aussi citer égalementAmong the deodorant active agents according to the invention, mention may also be made of
- les sels de zinc comme le salicylate de zinc, le gluconate de zinc, le pidolate de zinc ; le sulfate de zinc, le chlorure de zinc, le lactate de zinc, le phénolsulfonate de zinc ; ricinoléate de zinc zinc salts such as zinc salicylate, zinc gluconate, zinc pidolate; zinc sulphate, zinc chloride, zinc lactate, zinc phenolsulfonate; zinc ricinoleate
- le bicarbonate de sodium ; - sodium bicarbonate;
- l'acide salicylique et ses dérivés tels que l'acide n-octanoyl-5-salicylique salicylic acid and its derivatives such as n-octanoyl-5-salicylic acid
- Zéolites d'argent ou sans argent - Silver zeolites or without silver
- L'alun. Les actifs déodorants peuvent être présents de préférence dans les compositions selon l'invention dans des concentrations pondérales allant 0,01 à 5% en poids par rapport au poids total de la composition. Agents de suspension - Moon. The deodorant active agents may preferably be present in the compositions according to the invention in weight concentrations ranging from 0.01 to 5% by weight relative to the total weight of the composition. Suspending agents
Afin d'améliorer l'homogénéité du produit, on peut utiliser en plus un ou plusieurs agents de suspension qui sont choisis de préférence parmi les argiles montmorillonites modifiées hydrophobes comme les bentonites ou hectorites modifiées hydrophobes. On peut citer par exemple le produit Stearalkonium Bentonite (nom CTFA) (produit de réaction de la bentonite et de l'ammonium quaternaire chlorure de stéaralkonium) tel que le produit commercial vendu sous le nom TIXOGEL MP 250 par la société Sud Chemie Rheologicals, United Catalysts Inc ou le produit Disteardimonium Hectorite (nom CTFA) (produit de réaction de l'hectorite et du chlorure de distéaryldimonium) vendu sous le nom de Bentone 38 ou Bentone Gel par la société Elementis Specialities. In order to improve the homogeneity of the product, it is also possible to use one or more suspending agents which are preferably chosen from hydrophobic modified montmorillonite clays, such as hydrophobic modified bentonites or hectorites. Mention may be made, for example, of the product Stearalkonium Bentonite (CTFA name) (reaction product of bentonite and quaternary ammonium chloride stearalkonium chloride), such as the commercial product sold under the name Tixogel MP 250 by the company Sud Chemie Rheologicals, United Catalysts Inc. or the product Disteardimonium Hectorite (CTFA name) (reaction product of hectorite and distearyldimium chloride) sold under the name Bentone 38 or Bentone Gel by Elementis Specialties.
Les agents de suspension sont présents de préférence dans des quantités allant de 0,1 à 5% en poids et plus préférentiellement de 0.2 à 2% en poids par rapport au poids total de la composition. The suspending agents are preferably present in amounts ranging from 0.1 to 5% by weight and more preferably from 0.2 to 2% by weight relative to the total weight of the composition.
Poudre organique Organic powder
Selon une forme particulière de l'invention, les compositions selon l'invention contiendront en plus une poudre organique. According to one particular form of the invention, the compositions according to the invention will contain, in addition, an organic powder.
On entend dans la présente demande par « poudre organique », tout solide insoluble dans le milieu à température ambiante (25°C). Comme poudres organiques qui peuvent être utilisées dans la composition de l'invention, on peut citer par exemple, les particules de polyamide et notamment celles vendues sous les dénominations ORGASOL par la société Atochem ; les poudres de polyéthylène ; les microsphères à base de copolymères acryliques, telles que celles en copolymère diméthacrylate d'éthylène glycol/ methacrylate de lauryle vendues par la société Dow Corning sous la dénomination de POLYTRAP ; les microsphères de polyméthacrylate de méthyle, commercialisées sous la dénomination MICROSPHERE M-100 par la société Matsumoto ou sous la dénomination COVABEAD LH85 par la société Wackherr ; les microsphères de poly methacrylate de methyle creuses (granulometrie : 6,5 - 10,5 μ) commercialisées sous la dénomination GANZPEARL GMP 0800 par Ganz Chemical; micro-billes de copolymère methacrylate de methyle/dimethacrylate d'ethylene glycol (taille: 6.5-10.5 μ) commercialisées sous la dénomination GANZPEARL GMP 0820 par Ganz Chemical ou MICROSPONGE 5640 par la société Amcol Health & Beauty Solutions; les poudres de copolymère éthylène-acrylate, comme celles commercialisées sous la dénomination FLOBEADS par la société Sumitomo Seika Chemicals ; les poudres expansées telles que les microsphères creuses et notamment, les microsphères formées d'un terpolymère de chlorure de vinylidène, d'acrylonitrile et de méthacrylate et commercialisées sous la dénomination EXPANCEL par la société Kemanord Plast sous les références 551 DE 12 (granulométrie d'environ 12 m et masse volumique 40 kg/m3), 551 DE 20 (granulométrie d'environ 30 μιτι et masse volumique 65 kg/m3), 551 DE 50 (granulométrie d'environ 40 μιτι), ou les microsphères commercialisées sous la dénomination MICROPEARL F 80 ED par la société Matsumoto ; les poudres de matériaux organiques naturels tels que les poudres d'amidon, notamment d'amidons de maïs, de blé ou de riz, réticulés ou non, telles que les poudres d'amidon réticulé par l'anhydride octénylsuccinate, commercialisées sous la dénomination DRY-FLO par la société National Starch ; les microbilles de résine de silicone telles que celles commercialisées sous la dénomination TOSPEARL par la société Toshiba Silicone, notamment TOSPEARL 240 ; les poudres d'aminoacides telles que la poudre de Lauroyllysine commercialisée sous la dénomination AMIHOPE LL-1 1 par la Société Ajinomoto ; les particules de microdispersion de cire, qui ont de préférence des dimensions moyennes inférieures à 1 μιτι et notamment allant de 0,02 μιτι à 1 μιτι, et qui sont constituées essentiellement d'une cire ou d'un mélange de cires, telles que les produits commercialisés sous la dénomination Aquacer par la société Byk Cera, et notamment : Aquacer 520 (mélange de cires synthétiques et naturelles), Aquacer 514 ou 513 (cire de polyéthylène), Aquacer 51 1 (cire polymérique), ou telles que les produits commercialisés sous la dénomination Jonwax 120 par la société Johnson Polymer (mélange de cires de polyéthylène et de paraffine) et sous la dénomination Ceraflour 961 par la société Byk Cera (cire de polyéthylène modifiée micronisée) ; et leurs mélanges. As used herein, the term "organic powder" means any solid that is insoluble in the medium at room temperature (25 ° C.). Organic powders that may be used in the composition of the invention include, for example, polyamide particles and especially those sold under the names ORGASOL by Atochem; polyethylene powders; microspheres based on acrylic copolymers, such as as those in ethylene glycol dimethacrylate / lauryl methacrylate copolymer sold by Dow Corning under the name Polytrap; polymethyl methacrylate microspheres, sold under the name MICROSPHERE M-100 by the company Matsumoto or under the name COVABEAD LH85 by the company Wackherr; hollow polymethyl methacrylate microspheres (granulometry: 6.5 - 10.5 μ) sold under the name Ganzpearl GMP 0800 by Ganz Chemical; methyl methacrylate / ethylene glycol dimethacrylate copolymer microbeads (size: 6.5-10.5 μ) sold under the name Ganzpearl GMP 0820 by Ganz Chemical or Microsponge 5640 by the company Amcol Health & Beauty Solutions; ethylene-acrylate copolymer powders, such as those sold under the name FLOBEADS by Sumitomo Seika Chemicals; expanded powders such as hollow microspheres and in particular microspheres formed of a terpolymer of vinylidene chloride, acrylonitrile and methacrylate and sold under the name EXPANCEL by the company Kemanord Plast under the references 551 DE 12 (particle size distribution). about 12 m and density 40 kg / m 3), 551 DE 20 (particle size of about 30 μιτι and density 65 kg / m 3), 551 DE 50 (particle size of about 40 μιτι), or the microspheres marketed under the name MICROPEARL F 80 ED by the company Matsumoto; powders of natural organic materials such as starch powders, especially corn starch, wheat or rice, crosslinked or otherwise, such as starch powders crosslinked with octenylsuccinate anhydride, sold under the name DRY -FLO by the company National Starch; silicone resin microbeads such as those sold under the name Tospearl by the company Toshiba Silicone, in particular Tospearl 240; amino acid powders such as the Lauroyllysine powder marketed under the name Amihope LL-1 1 by the company Ajinomoto; the particles of microdispersion of wax, which preferably have average dimensions of less than 1 μιτι and especially ranging from 0.02 μιτι to 1 μιτι, and which consist essentially of a wax or a mixture of waxes, such as the products marketed under the name Aquacer by Byk Cera, and in particular: Aquacer 520 (mixture of synthetic and natural waxes), Aquacer 514 or 513 (polyethylene wax), Aquacer 51 1 (polymeric wax), or such as products sold under the name Jonwax 120 by Johnson Polymer (a mixture of polyethylene wax and paraffin wax) and under the name Ceraflour 961 by Byk Cera (micronized modified polyethylene wax); and their mixtures.
Additifs additives
Les compositions cosmétiques selon l'invention peuvent comprendre en outre des adjuvants cosmétiques choisis parmi les adoucissants, les antioxydants, les opacifiants, les stabilisants, les agents hydratants, les vitamines, des bactéricides, les conservateurs, les polymères, les parfums, les agents épaississants, des agents propulseurs ou tout autre ingrédient habituellement utilisé en cosmétique pour ce type d'application. Bien entendu, l'homme de métier veillera à choisir ce ou ces éventuels composés complémentaires de manière telle que les propriétés avantageuses attachées intrinsèquement à la composition conforme à l'invention ne soient pas, ou substantiellement pas, altérées par la ou les adjonctions envisagées. Les épaississants, de préférence non ioniques, peuvent être choisis parmi les gommes de guar et celluloses modifiées ou non modifiées telles que la gomme de guar hydroxypropylée, la cétylhydroxyéthylcellulose, les silices comme par exemple la Bentone Gel MIO vendue par la société NL INDUSTRIES ou la Veegum Ultra, vendue par la société POLYPLASTIC. The cosmetic compositions according to the invention may furthermore comprise cosmetic adjuvants chosen from softeners, antioxidants, opacifiers, stabilizers, moisturizing agents, vitamins, bactericides, preservatives, polymers, perfumes and thickeners. , propellants or any other ingredient usually used in cosmetics for this type of application. Of course, those skilled in the art will take care to choose this or these optional additional compounds in such a way that the advantageous properties intrinsically attached to the composition according to the invention are not, or not substantially, impaired by the addition or additions envisaged. The thickeners, preferably nonionic, may be selected from guar gums and modified or unmodified celluloses such as hydroxypropyl guar gum, cetylhydroxyethylcellulose, silicas such as Bentone Gel MIO sold by the company NL INDUSTRIES or the Veegum Ultra, sold by POLYPLASTIC.
Les épaississants peuvent également être cationiques comme par exemple le POLYQUATERNIUM-37 commercialisé sous la dénomination Salcare SC95 (Polyquaternium-37 (And) Minerai Oil (And) PPG-1 Trideceth-6) ou Salcare SC96 (Polyquaternium-37 (And) Propylene Glycol Dicaprylate/Dicaprate (And) PPG- 1 -Trideceth-6) ou d'autre polymère cationiques réticulés comme par exemple ceux de nom CTFA Copolymère Ethylacrylate / Dimethylamino Ethyl Methacrylate Cationique En Emulsion. Les quantités de ces différents constituants pouvant être présents dans la composition selon l'invention sont celles classiquement utilisées dans compositions pour le traitement de la transpiration. Aérosols The thickeners may also be cationic, for example POLYQUATERNIUM-37 sold under the name Salcare SC95 (Polyquaternium-37 (And) Mineral Oil (And) PPG-1 Trideceth-6) or Salcare SC96 (Polyquaternium-37 (And) Propylene Glycol Dicaprylate / Dicaprate (And) PPG-1 -Trideceth-6) or other crosslinked cationic polymer such as for example those of CTFA name Ethylacrylate / Dimethylamino Ethyl Methacrylate Cationic Emulsion Copolymer. The amounts of these various constituents that may be present in the composition according to the invention are those conventionally used in compositions for the treatment of perspiration. aerosols
Les compositions selon l'invention peuvent encore être pressurisées et être conditionnées dans un dispositif aérosol constitué par : The compositions according to the invention may be further pressurized and packaged in an aerosol device consisting of:
(A) un récipient comprenant une composition anti-transpirante telle que définie précédemment, (A) a container comprising an antiperspirant composition as defined above,
(B) au moins un agent propulseur et un moyen de distribution de la dite composition aérosol. (B) at least one propellant and a means for dispensing said aerosol composition.
Les propulseurs généralement utilisés dans ce type de produits et bien connus de l'homme de l'art, sont comme par exemple le diméthyléther (DME) ; les hydrocarbures volatils tels que le n-butane, le propane, l'isobutane, et leurs mélanges, éventuellement avec au moins un hydrocarbure chloré et/ou fluoré; parmi ces derniers on peut citer les composés vendus par la société Dupont de Nemours sous les dénominations Fréon® et Dymel®, et en particulier le monofluorotrichlorométhane, le difluorodichlorométhane, le tétrafluorodichloroéthane et le 1 ,1 -difluoroéthane vendu notamment sous la dénomination commerciale DYMEL 152 A par la société DUPONT. On peut également utiliser en tant qu'agent propulseur le gaz carbonique, le protoxyde d'azote, l'azote ou l'air comprimé. Les compositions contenant les particules de perlite telles que définies précédemment et le ou les agents propulseurs peuvent se trouver dans le même compartiment ou dans des compartiments différents dans le récipient aérosol. Selon l'invention, la concentration en agent propulseur varie généralement de 5 à 95% en poids pressurisée et plus préférentiellement de 50 à 85% en poids par rapport au poids total de la composition pressurisée. Propellants generally used in this type of product and well known to those skilled in the art are, for example, dimethyl ether (DME); volatile hydrocarbons such as n-butane, propane, isobutane, and mixtures thereof, optionally with at least one chlorinated and / or fluorinated hydrocarbon; among these are the compounds sold by the company Dupont de Nemours under the names Freon® and Dymel®, and in particular monofluorotrichloromethane, difluorodichloromethane, tetrafluorodichloroethane and 1,1-difluoroethane sold especially under the trade name Dymel 152 A by the company DUPONT. It is also possible to use as propellant carbon dioxide, nitrous oxide, nitrogen or compressed air. The compositions containing the perlite particles as defined above and the propellant (s) may be in the same compartment or in different compartments in the aerosol container. According to the invention, the concentration of propellant generally varies from 5 to 95% by weight pressurized and more preferably from 50 to 85% by weight relative to the total weight of the pressurized composition.
Le moyen de distribution, qui forme une partie du dispositif aérosol, est généralement constitué par une valve de distribution commandée par une tête de distribution, elle même comprenant une buse par laquelle la composition aérosol est vaporisée. Le récipient contenant la composition pressurisée peut être opaque ou transparent. Il peut être en verre, en matériau polymérique ou en métal, recouvert éventuellement d'une couche de vernis protecteur. The dispensing means, which forms part of the aerosol device, is generally constituted by a dispensing valve controlled by a dispensing head, it same comprising a nozzle by which the aerosol composition is vaporized. The container containing the pressurized composition may be opaque or transparent. It may be glass, polymeric material or metal, optionally covered with a layer of protective varnish.
L'invention concerne également un procédé cosmétique pour traiter les odeurs corporelles, notamment les odeurs axillaires ou les odeurs des pieds, qui consiste à appliquer sur les matières kératiniques une composition telle que décrite ci-dessus (et contenant bien entendu l'association selon l'invention). The invention also relates to a cosmetic process for treating body odors, in particular axillary odors or foot odors, which consists in applying to the keratin materials a composition as described above (and of course containing the combination according to US Pat. 'invention).
Ce procédé est en particulier utilisé pour traiter les odeurs axillaires dues à la bactérie C. xerosis ou les odeurs des pieds dues à B. linens. Le fait de détruire ou d'inhiber les bactéries responsables des mauvaises odeurs, et en particulier des mauvaises odeurs axillaires ou des pieds, avec une bactériocine tout en utilisant un prébiotique conférant un avantage sélectif aux les germes bénéfiques de la peau, permet de réduire et/ou d'ôter ou de prévenir le développement des odeurs corporelles sans pour autant détruire l'écosystème de la zone traitée. This process is especially used to treat axillary odors caused by C. xerosis bacteria or foot smells caused by B. linens. Destroying or inhibiting odor-causing bacteria, especially axillary odors or feet, with a bacteriocin while using a prebiotic that confers a selective advantage on the beneficial germs of the skin, can reduce and / or to remove or prevent the development of body odor without destroying the ecosystem of the treated area.
Selon un mode préféré de réalisation du procédé cosmétique pour traiter les odeurs corporelles, on applique une composition selon l'invention comprenant une plantaricine de classe II, plus particulièrement une plantaricine de classe II choisie parmi la plantaricine A, la plantaricine C19 et la plantaricine 149. According to a preferred embodiment of the cosmetic process for treating body odors, a composition according to the invention comprising a class II plantaricin, more particularly a class II plantaricin chosen from plantaricin A, plantaricin C19 and plantaricin, is applied. .
Selon un autre mode préféré de réalisation du procédé cosmétique pour traiter les odeurs corporelles, on applique une composition selon l'invention comprenant un prébiotique choisi parmi un xylooligosaccharide et un isomalto-oligosaccharide. According to another preferred embodiment of the cosmetic process for treating body odors, a composition according to the invention comprising a prebiotic chosen from a xylooligosaccharide and an isomalto-oligosaccharide is applied.
Selon une forme particulièrement préférée, le procédé cosmétique pour traiter les odeurs corporelles selon l'invention comprend l'application d'une composition comprenant According to a particularly preferred form, the cosmetic process for treating body odors according to the invention comprises the application of a composition comprising
- une bactériocine choisie parmie la plantaricine A, la plantaricine C19 et la plantaricine 149, et a bacteriocin selected from plantaricin A, plantaricin C19 and plantaricin 149, and
- un prébiotique choisi parmi un xylooligosaccharide et un isomalto-oligosaccharide. * Composition pour le soin et/ou le nettoyage de la peau a prebiotic chosen from a xylooligosaccharide and an isomalto-oligosaccharide. * Composition for the care and / or cleaning of the skin
La composition selon l'invention contient un milieu physiologiquement acceptable tel que défini ci-avant. Selon un mode préféré de réalisation de l'invention, la composition a un pH de préférence proche de celui de la peau, compris entre 4 et 7. The composition according to the invention contains a physiologically acceptable medium as defined above. According to a preferred embodiment of the invention, the composition has a pH preferably close to that of the skin, between 4 and 7.
Les compositions selon l'invention peuvent se présenter sous toutes les formes galéniques classiquement utilisées pour une application topique et notamment sous forme de solutions aqueuses, hydroalcooliques, d'émulsions huile-dans-eau (H/E) ou eau-dans-huile (E/H) ou multiple (triple : E/H/E ou H/E/H), de gels aqueux, ou de dispersions d'une phase grasse dans une phase aqueuse à l'aide de sphérules, ou des vésicules lipidiques de type ionique et/ou non ionique (liposomes, niosomes, oléosomes), ou de films fins. Ces compositions sont préparées selon les méthodes usuelles. The compositions according to the invention may be in any of the galenical forms conventionally used for topical application and especially in the form of aqueous, hydroalcoholic, oil-in-water (O / W) or water-in-oil ( E / H) or multiple (triple: W / O / W or W / E / H), aqueous gels, or dispersions of a fatty phase in an aqueous phase using spherules, or lipid vesicles of ionic and / or nonionic type (liposomes, niosomes, oleosomes), or thin films. These compositions are prepared according to the usual methods.
En outre, la composition selon l'invention peut être plus ou moins fluide et avoir l'aspect d'une crème blanche ou colorée, d'une pommade, d'un lait, d'une lotion, d'un sérum, d'une pâte, d'un gel moussant, d'un soin, d'un tonique ou d'une mousse. De manière particulièrement préférée, la composition selon l'invention est formulée sous forme de gel moussant. Elle peut être éventuellement appliquée sur la peau sous forme d'aérosol. Elle peut aussi se présenter sous forme solide, et par exemple sous forme de stick. Il pourra s'agir d'une composition de soin, d'une composition exfoliante ou de nettoyage ou de gommage, ou d'une composition de rasage telle qu'une mousse à raser. In addition, the composition according to the invention may be more or less fluid and have the appearance of a white or colored cream, an ointment, a milk, a lotion, a serum, a paste, a foaming gel, a treatment, a tonic or a mousse. In a particularly preferred manner, the composition according to the invention is formulated in the form of a foaming gel. It can optionally be applied to the skin in the form of an aerosol. It can also be in solid form, and for example in the form of a stick. It may be a care composition, an exfoliating composition or cleaning or scrubbing, or a shaving composition such as a shaving foam.
Quand la composition utilisée selon l'invention comporte une phase huileuse, celle-ci contient de préférence au moins une huile. Elle peut contenir en outre d'autres corps gras. When the composition used according to the invention comprises an oily phase, it preferably contains at least one oil. It may also contain other fatty substances.
Comme huiles utilisables dans la composition de l'invention, on peut citer par exemple : As oils that can be used in the composition of the invention, mention may be made for example of:
- les huiles hydrocarbonées d'origine animale, telles que le perhydrosqualène ; - les huiles hydrocarbonées d'origine végétale, telles que les triglycérides liquides d'acides gras comportant de 4 à 10 atomes de carbone comme les triglycérides des acides heptanoïque ou octanoïque ou encore, par exemple les huiles de tournesol, de maïs, de soja, de courge, de pépins de raisin, de sésame, de noisette, d'abricot, de macadamia, d'arara, de tournesol, de ricin, d'avocat, les triglycérides des acides caprylique/caprique comme ceux vendus par la société Stearineries Dubois ou ceux vendus sous les dénominations Miglyol 810, 812 et 818 par la société Dynamit Nobel, l'huile de jojoba, l'huile de beurre de karité; hydrocarbon oils of animal origin, such as perhydrosqualene; hydrocarbon-based oils of vegetable origin, such as liquid triglycerides of fatty acids containing from 4 to 10 carbon atoms, for instance triglycerides of heptanoic or octanoic acids or, for example, sunflower, corn or soybean oils, squash, grape seed, sesame, hazelnut, apricot, macadamia, arara, sunflower, castor oil, avocado, triglycerides of caprylic / capric acids such as those sold by Stearineries Dubois or those sold under the names Miglyol 810, 812 and 818 by the company Dynamit Nobel, jojoba oil, shea butter oil;
- les esters et les éthers de synthèse, notamment d'acides gras, comme les huiles de formules R'COOR2 et R'OR2 dans laquelle R' représente le reste d'un acide gras comportant de 8 à 29 atomes de carbone, et R2 représente une chaîne hydrocarbonée, ramifiée ou non, contenant de 3 à 30 atomes de carbone, comme par exemple l'huile de Purcellin, l'isononanoate d'isononyle, le myristate d'isopropyle, le palmitate d'éthyl-2-hexyle, le stéarate d'octyl-2-dodécyle, l'érucate d'octyl-2- dodécyle, l'isostéarate d'isostéaryle ; les esters hydroxylés comme l'isostéaryl lactate, l'octylhydroxystéarate, l'hydroxystéarate d'octyldodécyle, le diisostéaryl- malate, le citrate de triisocétyle, les heptanoates, octanoates, décanoates d'alcools gras ; les esters de polyol, comme le dioctanoate de propylène glycol, le diheptanoate de néopentylglycol et le diisononanoate de diéthylèneglycol ; et les esters du pentaérythritol comme le tétraisostéarate de pentaérythrityle ; esters and synthetic ethers, in particular of fatty acids, such as the oils of formulas R'COOR 2 and R'OR 2 in which R 'represents the residue of a fatty acid comprising from 8 to 29 carbon atoms, and R 2 represents a hydrocarbon chain, branched or unbranched, containing from 3 to 30 carbon atoms, for example Purcellin oil, isononyl isononanoate, isopropyl myristate or 2-ethyl palmitate; hexyl, octyl-2-dodecyl stearate, octyl-2-dodecyl erucate, isostearyl isostearate; hydroxylated esters such as isostearyl lactate, octyl hydroxystearate, octyldodecyl hydroxystearate, diisostearyl malate, triisocetyl citrate, heptanoates, octanoates, decanoates of fatty alcohols; polyol esters, such as propylene glycol dioctanoate, neopentyl glycol diheptanoate and diethylene glycol diisononanoate; and pentaerythritol esters such as pentaerythrityl tetraisostearate;
- les hydrocarbures linéaires ou ramifiés, d'origine minérale ou synthétique, tels que les huiles de paraffine, volatiles ou non, et leurs dérivés, la vaseline, les polydécènes, le polyisobutène hydrogéné tel que l'huile de parléam ; linear or branched hydrocarbons of mineral or synthetic origin, such as paraffin oils, volatile or not, and their derivatives, petroleum jelly, polydecenes, hydrogenated polyisobutene such as parleam oil;
- les alcools gras ayant de 8 à 26 atomes de carbone, comme l'alcool cétylique, l'alcool stéarylique et leur mélange (alcool cétylstéarylique), l'octyldodécanol, le 2- butyloctanol, le 2-hexyldécanol, le 2-undécylpentadécanol, l'alcool oléique ou l'alcool linoléique ; fatty alcohols having from 8 to 26 carbon atoms, such as cetyl alcohol, stearyl alcohol and their mixture (cetylstearyl alcohol), octyldodecanol, 2-butyloctanol, 2-hexyldecanol, 2-undecylpentadecanol, oleic alcohol or linoleic alcohol;
- les huiles fluorées partiellement hydrocarbonées et/ou siliconées comme celles décrites dans le document JP-A-2-295912 ; partially fluorinated hydrocarbon oils and / or silicone oils such as those described in document JP-A-2-295912;
- les huiles de silicone comme les polyméthylsiloxanes (PDMS) volatiles ou non à chaîne siliconée linéaire ou cyclique, liquides ou pâteux à température ambiante, notamment les cyclopolydiméthylsiloxanes (cyclométhicones) telles que la cyclohexasiloxane ; les polydiméthylsiloxanes comportant des groupements alkyle, alcoxy ou phényle, pendant ou en bout de chaîne siliconée, groupements ayant de 2 à 24 atomes de carbone ; les silicones phénylées comme les phényltriméthicones, les phényldiméthicones, les phényltriméthylsiloxydiphényl-siloxanes, les diphényl- diméthicones, les diphénylméthyldiphényl trisiloxanes, les 2-phényléthyltriméthyl- siloxysilicates, et les polyméthylphénylsiloxanes ; silicone oils such as volatile or non-volatile polymethylsiloxanes (PDMS) with a linear or cyclic silicone chain, which are liquid or pasty at room temperature, in particular cyclopolydimethylsiloxanes (cyclomethicones) such as cyclohexasiloxane; polydimethylsiloxanes containing alkyl groups, alkoxy or phenyl, during or at the end of the silicone chain, groups having from 2 to 24 carbon atoms; phenyl silicones such as phenyltrimethicones, phenyldimethicones, phenyltrimethylsiloxydiphenylsiloxanes, diphenyl-dimethicones, diphenylmethyldiphenyltrisiloxanes, 2-phenylethyltrimethylsiloxysilicates, and polymethylphenylsiloxanes;
- leurs mélanges. - their mixtures.
On entend par huile hydrocarbonée dans la liste des huiles citées ci-dessus, toute huile comportant majoritairement des atomes de carbone et d'hydrogène, et éventuellement des groupements ester, éther, fluoré, acide carboxylique et/ou alcool. By hydrocarbon oil is meant in the list of oils mentioned above, any oil predominantly comprising carbon and hydrogen atoms, and optionally ester, ether, fluoro, carboxylic acid and / or alcohol groups.
Les autres corps gras pouvant être présents dans la phase huileuse sont par exemple les acides gras comportant de 8 à 30 atomes de carbone, comme l'acide stéarique, l'acide laurique, l'acide palmitique et l'acide oléique ; les cires comme la lanoline, la cire d'abeille, la cire de Carnauba ou de Candellila, les cires de paraffine, de lignite ou les cires microcristallines, la cérésine ou l'ozokérite, les cires synthétiques comme les cires de polyéthylène, les cires de Fischer-Tropsch ; les résines de silicone telles que la trifluorométhyl-C1 -4-alkyldimethicone et la trifluoropropyldimethicone ; et les élastomères de silicone comme les produits commercialisés sous les dénominations KSG par la société Shin-Etsu, sous les dénominations "Trefil", "BY29" ou "EPSX" par la société Dow Corning ou sous les dénominations "Gransil" par la société Grant Industries. The other fatty substances that may be present in the oily phase are, for example, fatty acids containing from 8 to 30 carbon atoms, such as stearic acid, lauric acid, palmitic acid and oleic acid; waxes such as lanolin, beeswax, carnauba or candelilla wax, paraffin waxes, lignite waxes or microcrystalline waxes, ceresin or ozokerite, synthetic waxes such as polyethylene waxes, waxes Fischer-Tropsch; silicone resins such as trifluoromethyl-C1-4-alkyldimethicone and trifluoropropyldimethicone; and silicone elastomers such as the products sold under the names KSG by the company Shin-Etsu, under the names "Trefil", "BY29" or "EPSX" by Dow Corning or under the names "Gransil" by the company Grant industries.
Ces corps gras peuvent être choisis de manière variée par l'homme du métier afin de préparer une composition ayant les propriétés, par exemple de consistance ou de texture, souhaitées. Selon un mode particulier de réalisation de l'invention, la composition selon l'invention est une émulsion eau-dans-huile (E/H) ou huile-dans- eau (H/E). La proportion de la phase huileuse de l'émulsion peut aller de 5 à 80 % en poids, et de préférence de 5 à 50 % en poids par rapport au poids total de la composition. These fatty substances may be chosen in a varied manner by those skilled in the art in order to prepare a composition having the desired properties, for example of consistency or texture. According to a particular embodiment of the invention, the composition according to the invention is a water-in-oil (W / O) or oil-in-water (O / W) emulsion. The proportion of the oily phase of the emulsion may range from 5 to 80% by weight, and preferably from 5 to 50% by weight relative to the total weight of the composition.
Les émulsions contiennent généralement au moins un émulsionnant choisi parmi les émulsionnants amphotères, anioniques, cationiques ou non ioniques, utilisés seuls ou en mélange, et éventuellement un co-émulsionnant. Les émulsionnants sont choisis de manière appropriée suivant l'émulsion à obtenir (E/H ou H/E). L'émulsionnant et le co-émulsionnant sont généralement présents dans la composition, en une proportion allant de 0,3 à 30 % en poids, et de préférence de 0,5 à 20 % en poids par rapport au poids total de la composition. The emulsions generally contain at least one emulsifier chosen from amphoteric, anionic, cationic or nonionic emulsifiers, used alone. or in mixture, and optionally a co-emulsifier. The emulsifiers are suitably selected according to the emulsion to be obtained (W / O or O / W). The emulsifier and the co-emulsifier are generally present in the composition, in a proportion ranging from 0.3 to 30% by weight, and preferably from 0.5 to 20% by weight relative to the total weight of the composition.
Pour les émulsions E/H, on peut citer par exemple comme émulsionnants les dimethicone copolyols tels que le mélange de cyclomethicone et de dimethicone copolyol, vendu sous la dénomination "DC 5225 C" par la société Dow Corning, et les alkyl-dimethicone copolyols tels que le Laurylmethicone copolyol vendu sous la dénomination "Dow Corning 5200 Formulation Aid" par la société Dow Corning et le Cetyl dimethicone copolyol vendu sous la dénomination Abil EM 90R par la société Goldschmidt. On peut aussi utiliser comme tensioactif d'émulsions E/H, un organopolysiloxane solide élastomère réticulé comportant au moins un groupement oxyalkyléné, tel que ceux obtenus selon le mode opératoire des exemples 3, 4 et 8 du document US-A-5,412,004 et des exemples du document US-A-5,81 1 ,487, notamment le produit de l'exemple 3 (exemple de synthèse) du brevet US-A- 5,412,004, et tel que celui commercialisé sous la référence KSG 21 par la société Shin Etsu. For the W / O emulsions, examples of emulsifiers that may be mentioned include dimethicone copolyols such as the mixture of cyclomethicone and dimethicone copolyol, sold under the name "DC 5225 C" by Dow Corning, and alkyl dimethicone copolyols such as Laurylmethicone copolyol sold under the name Dow Corning 5200 Formulation Aid by Dow Corning and Cetyl dimethicone copolyol sold under the name Abil EM 90 R by Goldschmidt. It is also possible to use, as surfactant of W / O emulsions, a crosslinked solid elastomeric organopolysiloxane comprising at least one oxyalkylene group, such as those obtained according to the procedure of Examples 3, 4 and 8 of US Pat. No. 5,412,004 and examples US-A-5,81 1,487, in particular the product of Example 3 (synthetic example) of US-A-5,412,004, and such as that sold under the reference KSG 21 by Shin Etsu.
Pour les émulsions H/E, on peut citer par exemple comme émulsionnants, les émulsionnants non ioniques tels que les esters d'acides gras et de glycérol oxyalkylénés (plus particulièrement polyoxyéthylénés) ; les esters d'acides gras et de sorbitan oxyalkylénés ; les esters d'acides gras oxyalkylénés (oxyéthylénés et/ou oxypropylénés) ; les éthers d'alcools gras oxyalkylénés (oxyéthylénés et/ou oxypropylénés) ; les esters de sucres comme le stéarate de sucrose ; et leurs mélanges tels que le mélange de stéarate de glycéryle et de stéarate de PEG-40. For O / W emulsions, mention may be made, for example, as emulsifiers, of nonionic emulsifiers such as oxyalkylenated (more particularly polyoxyethylenated) fatty acid esters of glycerol; oxyalkylenated fatty acid and sorbitan esters; oxyalkylenated fatty acid esters (oxyethylenated and / or oxypropylenated); oxyalkylenated fatty alcohol ethers (oxyethylenated and / or oxypropylenated); sugar esters such as sucrose stearate; and mixtures thereof such as the mixture of glyceryl stearate and PEG-40 stearate.
De façon avantageuse, s la composition pour le soin et/ou le nettoyage de la peau selon l'invention peut comprendre au moins un ingrédient et/ou actif additionnel de soin de la peau, en particulier de soin de la peau grasse. On pourra notamment associer les actifs et ingrédients additionnels décrits dans les demandes de brevet W02004/105736 et DE10324567, incorporées ici par référence, parmi lesquels les agents dépigmentants, les conservateurs, les agents antimicrobiens, les agents antitranspirants, les chélateurs de métaux, les filtres UV, les protéines hydrolysées, les antioxydants, les vitamines, les agents antiinflammatoires, les agents anti-irritants, les agents hydratants, les extraits végétaux, les adjuvants cosmétiques, et leurs mélanges. Advantageously, the composition for the care and / or cleaning of the skin according to the invention may comprise at least one additional skin care ingredient and / or active ingredient, in particular for the treatment of oily skin. It will be possible to combine the additional active ingredients and ingredients described in patent applications WO2004 / 105736 and DE10324567, incorporated herein by reference, among which depigmenting agents, preservatives, antimicrobial agents, antiperspirants, metal chelators, filters UV, hydrolysed proteins, antioxidants, vitamins, anti-inflammatory agents, anti-irritant agents, moisturizing agents, plant extracts, cosmetic adjuvants, and mixtures thereof.
Comme agents dépigmentants, on pourra citer notamment la vitamine C et ses dérivés et notamment la vit CG, CP et 3-0 éthyl vitamine C, l'alpha et la béta arbutine, le lucinol et ses dérivés, l'acide kojique, le résorcinol et ses dérivés, l'acide tranexamique et ses dérivés, l'acide gentisique, l'homogentisate, le méthyl gentisate ou l'homogentisate, l'acide dioique, le D calcium panthéteine sulfonate, l'acide lipoique, l'acide ellagique, la vitamine B3, l'acide tranexamique, l'acide lipoique, l'acide linoléique et ses dérivés, les céramides et leurs homologues, les dérivés de plantes comme la camomille, la busserole, la famille des aloe (vera, ferox, bardensis), de mûrier, de scutellaire, sans que cette liste soit exhaustive. As depigmenting agents, there may be mentioned in particular vitamin C and its derivatives and in particular the vit CG, CP and 3-0 ethyl vitamin C, alpha and beta arbutin, lucinol and its derivatives, kojic acid, resorcinol and its derivatives, tranexamic acid and its derivatives, gentisic acid, homogentisate, methyl gentisate or homogentisate, dioic acid, D calcium panthetine sulfonate, lipoic acid, ellagic acid, vitamin B3, tranexamic acid, lipoic acid, linoleic acid and its derivatives, ceramides and their homologs, plant derivatives such as chamomile, bearberry, the family of aloe (vera, ferox, bardensis) , of mulberry tree, skullcap, without this list being exhaustive.
Comme agents humectants ou hydratants, on peut citer notamment le glycérol et ses dérivés, l'urée et ses dérivés notamment l'Hydrovance commercialisée par National Starch, les acides lactiques, l'acide hyaluronique, les AHA, les BHA, le pidolate de sodium, le xylitol, la sérine, le lactate de sodium, l'ectoine et ses dérivés, le chitosane et ses dérivés, le collagène, le plancton, un extrait d'imperata cylindra commercialisé sous la dénomination Moist 24 par la société Sederma. Humidifying or moisturizing agents that may especially be mentioned include glycerol and its derivatives, urea and its derivatives, in particular Hydrovance marketed by National Starch, lactic acids, hyaluronic acid, AHAs, BHAs and sodium pidolate. , xylitol, serine, sodium lactate, ectoin and its derivatives, chitosan and its derivatives, collagen, plankton, imperata cylindra extract marketed under the name Moist 24 by Sederma.
On pourra également utiliser des ingrédients et/ou actifs additionnels pour le soin des peaux grasses, notamment à tendance acnéique, et/ou des peaux acnéiques, et/ou des peaux hyperséborrhéiques, tels que les agents matifiants, les charges abrasives ou agents exfoliants, les agents desquamants, les agents antimicrobiens, les agents apaisants, les agents anti-inflammatoires, les agents sébo-régulateurs, les agents antioxydants, les agents cicatrisants, les agents astringents, et leurs mélanges. Plus particulièrement, on choisira d'incorporer dans la composition selon l'invention au moins un ingrédient et/ou actif additionnel de soin de la peau choisi parmi les agents dépigmentants, les chélateurs de métaux, les protéines hydrolysées, les vitamines, les extraits végétaux, les agents matifiants, les charges abrasives ou agents exfoliants, les agents desquamants, les agents antimicrobiens, les agents apaisants, les agents anti-inflammatoires, les agents sébo-régulateurs, les agents antioxydants, les agents cicatrisants, les agents astringents, et leurs mélanges. It will also be possible to use additional ingredients and / or active agents for the care of oily skin, in particular with acne prone skin, and / or acne skin, and / or hyperseborrhoeic skin, such as mattifying agents, abrasive fillers or exfoliating agents, desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebum-regulating agents, antioxidants, cicatrizing agents, astringent agents, and mixtures thereof. More particularly, it will be chosen to incorporate into the composition according to the invention at least one additional skin care ingredient and / or active agent chosen from depigmenting agents, metal chelators, hydrolysed proteins, vitamins and plant extracts. , mattifying agents, abrasive fillers or exfoliating agents, desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebo-regulating agents, antioxidants, cicatrizing agents, astringent agents, and their agents. mixtures.
La composition pourra être une composition de soin et/ou de nettoyage et/ou de maquillage, destinée à être rincée ou non rincée. De préférence, il s'agira d'une composition de soin non rincée. Lorsque la composition comprendra des agents exfoliants ou charges abrasives, elle sera de préférence rincée. The composition may be a care composition and / or cleaning and / or makeup, intended to be rinsed or not rinsed. Preferably, it will be a non-rinsed skincare composition. When the composition comprises exfoliating agents or abrasive fillers, it will preferably be rinsed.
Ingrédients et actifs additionnels pour le soin des peaux grasses Ingredients and additional active ingredients for the care of oily skin
Agents matifiants Matifying agents
Par 'agent matifiant', on entend des agents destinés à rendre la peau visiblement plus mate, moins brillante. By "matting agent" is meant agents intended to make the skin visibly duller, less glossy.
L'effet matifiant de l'agent et/ou de la composition le contenant peut notamment être évalué à l'aide d'un gonioréflectomètre, en mesurant le rapport R entre la réflexion spéculaire et la réflexion diffuse. Une valeur de R inférieure ou égale à 2 traduit généralement un effet matifiant. The matting effect of the agent and / or of the composition containing it can in particular be evaluated using a gonioreflectometer, by measuring the ratio R between the specular reflection and the diffuse reflection. A value of R less than or equal to 2 generally reflects a matting effect.
L'agent matifiant pourra notamment être choisi parmi un amidon de riz ou un amidon de maïs, la kaolinite, les silices, le talc, un extrait de graines de potiron, des microbilles de cellulose, des fibres végétales, des fibres synthétiques, en particulier de polyamides, des microsphères de copolymères acryliques expansées, des poudres de polyamides, les poudres de silice, les poudres de polytétrafluoroéthylène, les poudres de résine de silicone, les poudres de copolymères acryliques, les poudres de cire, les poudres de polyéthylène, les poudres d'organopolysiloxane réticulé élastomère enrobées de résine de silicone, les poudres composites de talc/dioxyde de titane/alumine/silice, les poudres de silicates mixtes amorphes, les poudres de polymères acryliques, les particules de silicate et notamment de silicate mixte, et leurs mélanges. The matting agent may especially be chosen from a rice starch or a maize starch, kaolinite, silicas, talc, an extract of pumpkin seeds, cellulose microbeads, vegetable fibers, synthetic fibers, in particular polyamides, microspheres of expanded acrylic copolymers, polyamide powders, silica powders, polytetrafluoroethylene powders, silicone resin powders, acrylic copolymer powders, wax powders, polyethylene powders, powders crosslinked organopolysiloxane elastomer coated with silicone resin, talc / titanium dioxide / alumina / silica composite powders, amorphous mixed silicate powders, acrylic polymer powders, silicate particles and especially mixed silicate particles, and mixtures thereof.
Comme exemples d'agents matifiants, on peut citer notamment : Examples of mattifying agents that may be mentioned include:
- l'amidon de riz ou de maïs, en particulier un aluminium starch octenyl succinate commercialisé sous la dénomination Dry Flo® par la société National Starch, starch from rice or maize, in particular starch octenyl succinate aluminum sold under the name Dry Flo® by the company National Starch,
- la kaolinite ; kaolinite;
- les silices ; - silicas;
- le talc ; - talc;
- un extrait de graines de potiron tel que commercialisé sous la dénomination Curbilene® par la société Indena ; an extract of pumpkin seeds as marketed under the name Curbilene® by the company Indena;
- des microbilles de cellulose telles que décrites dans la demande de brevet L'OREAL E P 1 562 562 ; cellulose microbeads as described in the patent application L'OREAL E P 1,562,562;
- des fibres, telles que des fibres de soie, de coton, de laine, de lin, de cellulose extraites notamment du bois, des légumes ou des algues, de polyamide (Nylon®), de cellulose modifiée, de poly-p-phénylène téréphtamide, en acrylique, de polyoléfine, de verre, de silice, d'aramide, de carbone, de Téflon®, de collagène insoluble, de polyesters, de polychlorure de vinyle ou de vinylidène, d'alcool polyvinylique, de polyacrylonitrile, de chitosane, de polyuréthane, de polyéthylène phtalate, des fibres formées d'un mélange de polymères, les fibres synthétiques résorbables, et leurs mélanges décrites dans la demande de brevet L'OREAL EP 1 151 742 ; - fibers, such as silk, cotton, wool, flax, cellulose fibers extracted in particular from wood, vegetables or algae, polyamide (Nylon®), modified cellulose, poly-p-phenylene terephtamide, acrylic, polyolefin, glass, silica, aramid, carbon, Teflon®, insoluble collagen, polyesters, polyvinyl chloride or vinylidene, polyvinyl alcohol, polyacrylonitrile, chitosan , polyurethane, polyethylene phthalate, fibers formed from a mixture of polymers, synthetic resorbable fibers, and mixtures thereof described in patent application L'OREAL EP 1 151 742;
- des microsphères de copolymères acryliques expansées telles que celles commercialisées par la société EXPANCEL sous les dénominations EXPANCEL 551®, microspheres of expanded acrylic copolymers such as those sold by Expancel under the names Expancel 551®,
- des charges à effet optique telles que décrites dans la demande de brevet FR 2 869 796, en particulier : optical effect charges as described in patent application FR 2 869 796, in particular:
- les poudres de polyamides (Nylon®), comme par exemple les particules de Nylon 12 du type Orgasol d'Atofina de taille moyenne 10 microns et d'indice de réfraction 1 ,54, polyamide powders (Nylon®), such as, for example, the Atofina-type Orgasol Nylon 12 particles having a mean size of 10 microns and a refractive index 1, 54,
- les poudres de silice, comme par exemple les Silica beads SB150 de silica powders, such as Silica beads SB150 from
Miyoshi de taille moyenne 5 microns et d'indice de réfraction 1 ,45, Miyoshi of average size 5 microns and refractive index 1, 45,
- les poudres de polytétrafluoroéthylène, comme les PTFE ceridust 9205F de Clariant de taille moyenne 8 microns et d'indice de réfraction 1 ,36, - les poudres de résine de silicone comme les Silicon resin Tospearl 145A de GE Silicone de taille moyenne 4,5 microns et d'indice de réfraction 1 ,41 ,polytetrafluoroethylene powders, such as Clariant ceridust 9205F PTFE of average size 8 microns and refractive index 1, 36, silicone resin powders such as GE Silicone Silicon resin Tospearl 145A having a mean size of 4.5 microns and a refractive index of 1.41,
- les poudres de copolymères acryliques, notamment de poly(méth)acrylate de méthyle, comme les particules PMMA Jurymer MBI de Nihon Junyoki de taille moyenne 8 microns et d'indice de réfraction 1 ,49, ou les particules Micropearl M100® et F 80 ED® de la société Matsumoto Yushi-Seiyaku ; powders of acrylic copolymers, in particular of methyl poly (meth) acrylate, such as PMMA Jurymer MBI particles of Nihon Junyoki having an average size of 8 microns and a refractive index of 1.49, or Micropearl M100® and F80 particles; ED® from Matsumoto Yushi-Seiyaku;
- les poudres de cire comme les particules Paraffin wax microease 1 14S de micropowders de taille moyenne 7 microns et d'indice de réfraction 1 ,54, wax powders such as Paraffin wax microease particles 1 14S of micropowders of average size 7 microns and refractive index 1, 54,
- les poudres de polyéthylène, notamment comprenant au moins un copolymère éthylène/acide acrylique, et en particulier constituées de copolymères éthylène/acide acrylique comme les particules Flobeads EA 209 de Sumitomo (de taille moyenne 10 microns et d'indice de réfraction 1 ,48),polyethylene powders, especially comprising at least one ethylene / acrylic acid copolymer, and in particular consisting of ethylene / acrylic acid copolymers such as the Sumitomo Flobeads EA 209 particles (of average size 10 microns and refractive index 1, 48 )
- les poudres d'organopolysiloxane réticulé élastomère enrobées de résine de silicone, notamment de résine silsesquioxane, comme décrit par exemple dans le brevet US 5 538 793. De telles poudres d'élastomère sont vendues sous les dénominations "KSP-100", "KSP-101 ", "KSP-102", "KSP-103", "KSP- 104", "KSP-105" par la société SHIN ETSU, et elastomeric crosslinked organopolysiloxane powders coated with silicone resin, in particular with silsesquioxane resin, as described, for example, in US Pat. No. 5,538,793. Such elastomer powders are sold under the names "KSP-100" and "KSP-100". -101 "," KSP-102 "," KSP-103 "," KSP-104 "," KSP-105 "by the company SHIN ETSU, and
- les poudres composites de talc/dioxyde de titane/alumine/silice comme celles vendues sous la dénomination Coverleaf AR-80 par la société Catalyst & chemicals, talc / titanium dioxide / alumina / silica composite powders, such as those sold under the name Coverleaf AR-80 by Catalyst & chemicals,
et leurs mélanges and their mixtures
- des composés absorbant et/ou adsorbant le sébum tels que décrit dans la même demande de brevet FR 2 869 796. On peut citer notamment : - Absorbent and / or adsorbent sebum compounds as described in the same patent application FR 2 869 796. Mention may in particular be made of:
- les poudres de silice, comme par exemple les microsphères de silice poreuses vendues sous la dénomination "SILICA BEADS SB-700" commercialisées par la société MYOSHI, les "SUNSPHERE® H51 ", "SUNSPHERE® H33", "SUNSPHERE® H53" commercialisées par la société ASAHI GLASS ; les microsphères de silice amorphe enrobées de polydiméthylsiloxane vendues sous la dénomination "SA SUNSPHERE® H- 33" et "SA SUNSPHERE® H-53" commercialisées par la société ASAHI GLASS ; - les poudres de silicates mixtes amorphes, notamment d'aluminium et de magnésium, comme par exemple celle commercialisée sous la dénomination "NEUSILIN UFL2" par la société Sumitomo. silica powders, such as, for example, the porous silica microspheres sold under the name "Silica Beads SB-700" marketed by the company MYOSHI, the "SUNSPHERE® H51", "SUNSPHERE® H33", "SUNSPHERE® H53" marketed by ASAHI GLASS; amorphous silica microspheres coated with polydimethylsiloxane sold under the name "SA Sunsphere® H-33" and "SA Sunsphere® H-53" sold by ASAHI GLASS; amorphous mixed silicate powders, in particular aluminum and magnesium, such as, for example, that marketed under the name "NEUSILIN UFL2" by the company Sumitomo.
- les poudres de polyamides (nylon®), comme par exemple "l'ORGASOL® 4000" commercialisé par la société ATOCHEM, et polyamide powders (nylon®), for example "ORGASOL® 4000" marketed by the company ATOCHEM, and
- les poudres de polymères acryliques, notamment de polyméthacrylate de méthyle, comme par exemple le "COVABEAD® LH85" commercialisé par la société WACKHERR ; de polyméthacrylate de méthyle/diméthacrylate d'éthylène glycol, comme par exemple le "DOW CORNING 5640 MICROSPONGE® SKIN OIL ADSORBER" commercialisé par la société DOW CORNING, ou le "GANZPEARL® GMP-0820" commercialisé par la société GANZ CHEMICAL ; de polyméthacrylate d'allyle/diméthacrylate d'éthylène glycol, comme par exemple le "POLY-PORE® L200" ou le "POLY-PORE® E200" commercialisés par la société AMCOL ; de copolymère diméthacrylate d'éthylène glycol/méthacrylate de lauryle, comme par exemple le "POLYTRAP® 6603" commercialisé de la société DOW CORNING ; acrylic polymer powders, in particular polymethyl methacrylate, for example "COVABEAD® LH85" marketed by WACKHERR; polymethyl methacrylate / ethylene glycol dimethacrylate, such as, for example, "DOW CORNING 5640 MICROSPONGE® SKIN OIL ADSORBER" marketed by Dow Corning, or "Ganzpearl® GMP-0820" marketed by Ganz Chemical; allyl polymethacrylate / ethylene glycol dimethacrylate, such as for example "POLY-PORE® L200" or "POLY-PORE® E200" marketed by AMCOL; ethylene glycol dimethacrylate / lauryl methacrylate copolymer, such as, for example, "POLYTRAP® 6603" marketed from Dow Corning;
- les particules de silicate, telle que la silicate d'alumine ; silicate particles, such as alumina silicate;
- les particules de silicates mixtes, telles que : mixed silicate particles, such as:
o les particules de silicate d'aluminium et de magnésium, telles que la saponite ou silicate de magnésium et d'aluminium hydraté avec un sulfate de sodium commercialisée sous la dénomination commerciale Sumecton® par la société Kunimine ; aluminum magnesium silicate particles, such as saponite or magnesium aluminum silicate, hydrated with a sodium sulphate sold under the trade name Sumecton® by the company Kunimine;
o le complexe silicate de magnésium, hydroxyéthylcellulose, huile de cumin noir, huile de courge et phospholipides ou Matipure® de Lucas Meyer. o magnesium silicate complex, hydroxyethylcellulose, black cumin oil, pumpkin oil and phospholipids or Matipure® by Lucas Meyer.
et leurs mélanges. and their mixtures.
Comme agents matifiants préférés, on pourra utiliser selon l'invention un extrait de graines de potiron, un amidon de riz ou de maïs, la kaolinite, des silices, le talc, les poudres de polyamides, les poudres de polyethylènes, les poudres de copolymères acryliques, les microsphères de copolymères acryliques expansées, les microbilles de résines de silicones, les particules de silicate mixte et leurs mélanges. Charges abrasives ou agents exfoliants As preferred matting agents, it is possible to use, according to the invention, an extract of pumpkin seeds, a rice or maize starch, kaolinite, silicas, talc, polyamide powders, polyethylene powders and copolymer powders. acrylics, microspheres of expanded acrylic copolymers, silicone resin microbeads, mixed silicate particles and mixtures thereof. Abrasive loads or exfoliating agents
Comme agents exfoliants utilisables dans des compositions rincées selon l'invention, on peut citer par exemple des particules exfoliantes ou gommantes d'origine minérale, végétale ou organique. Ainsi, on peut utiliser par exemple des billes ou de la poudre de polyéthylène, de la poudre de nylon, de la poudre de polychlorure de vinyle, de la pierre ponce, des broyats de noyaux d'abricots ou de coques de noix, de la sciure de bois, des billes de verre, l'alumine, et leurs mélanges. As exfoliating agents that may be used in rinsed compositions according to the invention, mention may be made, for example, of exfoliant or scrubbing particles of mineral, vegetable or organic origin. Thus, for example, beads or polyethylene powder, nylon powder, polyvinyl chloride powder, pumice, ground apricot kernels or nut shells can be used. sawdust, glass beads, alumina, and mixtures thereof.
On peut citer aussi l'Exfogreen® de Solabia (extrait de bambou), des extraits d'akenes de fraises (Akènes de fraise de Greentech), de la poudre de noyau de pêche, la poudre de noyau d'abricot, et enfin dans le domaine des poudres végétales à effet abrasif, citons la poudre de noyaux d'airelles (cranberry). Exfogreen® from Solabia (bamboo extract), strawberry akenes (Greentech strawberry Akènes), peach kernel powder, apricot kernel powder, and finally the field of vegetable powders with abrasive effect include the cranberry powder.
Comme charges abrasives ou agents exfoliants préférés selon l'invention, on citera la poudre de noyaux de pêche, la poudre de noyaux d'abricot, la poudre de noyaux d'airelles, les extraits d'akènes de fraise, les extraits de bambou. As abrasive fillers or preferred exfoliation agents according to the invention, mention may be made of peach kernel powder, apricot kernel powder, cranberry kernel powder, strawberry achene extracts, bamboo extracts.
Par l'expression actif additionnel de soin des peaux grasses , on entend, dans le cadre de la présente invention, un composé qui a par lui-même, c'est-à-dire ne nécessitant pas l'intervention d'un agent extérieur pour l'activer, une activité biologique qui peut être en particulier : By the additional active expression of care of oily skin is meant, in the context of the present invention, a compound which has by itself, that is to say not requiring the intervention of an external agent to activate it, a biological activity that can be in particular:
- une activité desquamante (qui permet l'ouverture des comédons), et/ou - desquamating activity (which allows the opening of comedones), and / or
- une activité anti-microbienne (notamment sur P. acnés), et/ou an antimicrobial activity (especially on P. acnes), and / or
- une activité apaisante ou anti-inflammatoire, et/ou a soothing or anti-inflammatory activity, and / or
- une activité sébo-régulatrice, et/ou a sebo-regulatory activity, and / or
- une activité anti-oxydante (qui empêche l'oxydation du squalène et la formation des comédons) an antioxidant activity (which prevents the oxidation of squalene and the formation of comedones)
- une activité cicatrisante ; - a healing activity;
- une activité astringente. - an astringent activity.
Comme exemples d'actifs associés utilisables dans les compositions de l'invention, on pourra donc notamment citer les agents desquamants, les agents antimicrobiens, les agents apaisants, les agents anti-inflammatoires, les agents sébo-régulateurs, les agents antioxydants et leurs mélanges. As examples of associated active agents that can be used in the compositions of the invention, particular mention may be made of desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebo-regulating agents, antioxidants and mixtures thereof.
De préférence la composition selon l'invention comprendra comme actif additionnel au moins un agent sébo-régulateur. Selon un mode particulier, la composition pourra comprendre en outre éventuellement un agent antimicrobien. Preferably, the composition according to the invention will comprise as additional active agent at least one sebum-regulating agent. In a particular embodiment, the composition may further optionally comprise an antimicrobial agent.
De façon encore préférée elle pourra comprendre en plus un agent desquamant. Et pour augmenter encore l'efficacité et/ou la tolérance desdites compositions, on pourra rajouter en outre des agents apaisants ou anti-inflammatoires, des agents antioxydants, des agents cicatrisants, des agents astringents, et leurs mélanges. More preferably it may further comprise a desquamating agent. And to further increase the efficacy and / or the tolerance of said compositions, it may additionally add soothing or anti-inflammatory agents, antioxidants, cicatrizing agents, astringent agents, and mixtures thereof.
Comme exemples de composés, pour chaque classe, on pourra citer notamment : Agents sébo-régulateurs ou anti-séborrhéiques As examples of compounds, for each class, mention may be made in particular of: Sebo-regulating or anti-seborrheic agents
Par agents "séborégulateurs ou anti-séborrhéiques", on entend notamment des agents capables de réguler l'activité des glandes sébacées. By "sebo-regulating or anti-seborrhoeic agents" is meant in particular agents capable of regulating the activity of the sebaceous glands.
On peut citer notamment : We can mention in particular:
- l'acide rétinoïque, le peroxyde de benzoyle, le soufre, la vitamine B6 (ou pyridoxine), le chlorure de sélénium, la criste marine ; retinoic acid, benzoyl peroxide, sulfur, vitamin B6 (or pyridoxine), selenium chloride, sea fennel;
- les mélanges d'extrait de canelle, de thé et d'octanoylglycine tel que le Sepicontrol A5 TEA de chez Seppic ; mixtures of cinnamon extract, tea and octanoylglycine such as Sepicontrol A5 TEA from Seppic;
- le mélange de capryloyl glycine, de sarcosine et d'extrait de cinnamomum zeylanicum commercialisé notamment par la société SEPPIC sous la dénomination commerciale Sepicontrol® A5 ; the mixture of capryloyl glycine, sarcosine and cinnamomum zeylanicum extract marketed in particular by the company SEPPIC under the trademark Sepicontrol® A5;
- d'autres sels de zinc, en plus du salicylate de zinc, tels que le pyrrolidone carboxylate de zinc (ou pidolate de zinc), le lactate de zinc, l'aspartate de zinc, le carboxylate de zinc, le cystéate de zinc ; other zinc salts, in addition to zinc salicylate, such as zinc pyrrolidone carboxylate (or zinc pidolate), zinc lactate, zinc aspartate, zinc carboxylate, zinc cysteate;
- des sels de cuivre, tel que le sulfate de cuivre ou le pyrrolidone carboxylate de cuivre ; copper salts, such as copper sulphate or copper pyrrolidone carboxylate;
- des extraits de végétaux des espèces Arnica montana, Cinchona succirubra, Eugenia caryophyllata, Humulus lupulus, Hypericum perforatum, Mentha piperita, Rosmarinus officinalis, Salvia oficinalis et Thymus vulga s, tous commercialisés par exemple par la société MARUZEN ; plant extracts of the species Arnica montana, Cinchona succirubra, Eugenia caryophyllata, Humulus lupulus, Hypericum perforatum, Mentha piperita, Rosmarinus officinalis, Salvia oficinalis and Thymus vulga s, all marketed for example by the company MARUZEN;
- les extraits de reine des prés (spiraea ulamaria) tel que celui vendu sous la dénomination Sébonormine® par la société Silab ; - Meadowwort extracts (Spiraea ulamaria) such as that sold under the name Sébonormine® by the company Silab;
- les extraits d'algue laminaria saccharina tel que celui vendu sous la dénomination Phlorogine® par la société Biotechmarine ; algae extracts laminaria saccharina such as that sold under the name Phlorogine® by the company Biotechmarine;
- les mélanges d'extraits de racines de pimprenelle (sanguisorba officinalis/poterium officinale), de rhizomes de gingembre (zingiber officinalis) et d'écorce de cannelier (cinnamomum cassia) tel que celui vendu sous la dénomination Sebustop® par la société Solabia ; mixtures of extracts of burnet root (Sanguisorba officinalis / Poterium officinale), of ginger rhizomes (Zingiber officinalis) and of cinnamon bark (Cinnamomum cassia), such as that sold under the name Sebustop® by the company Solabia;
- les extraits de graines de lin tel que celui vendu sous la dénomination Linumine® par la société Lucas Meyer ; linseed extract, such as that sold under the name Linumine® by the company Lucas Meyer;
- les extraits de Phellodendron tels que ceux vendu sous la dénomination Phellodendron extract BG par la société Maruzen ou Oubaku liquid B par la société Ichimaru Pharcos ; Phellodendron extracts such as those sold under the name Phellodendron extract BG by the company Maruzen or Oubaku liquid B by the company Ichimaru Pharcos;
- les mélanges d'huile d'argan, d'extrait de serenoa serrulata (saw palmetto) et d'extrait de graines de sésame tel que celui vendu sous la dénomination Regu SEB® par la société Pentapharm ; mixtures of argan oil, of Serenoa serrulata (saw palmetto) extract and of sesame seed extract, such as that sold under the name Regu SEB® by the company Pentapharm;
- les mélanges d'extraits d'epilobe, de terminalia chebula, de capucine et de zinc biodisponible (microalgues) tel que celui vendu sous la dénomination Seborilys® par la société green tech ; mixtures of extracts of willowweed, terminalia chebula, nasturtium and bioavailable zinc (microalgae), such as that sold under the name Seborilys® by the company green tech;
- les extraits de Pygeum afrianum tel que celui vendu sous la dénomination Pygeum afrianum sterolic lipid extract par la société Euromed ; extracts of Pygeum afrianum, such as that sold under the name Pygeum afrianum sterolic lipid extract by the company Euromed;
- les extraits de serenoa serrulata tels que ceux vendus sous les dénomination Viapure Sabal par la société Actives International, ou ceux vendus par la société extracts of serenoa serrulata such as those sold under the name Viapure Sabal by the company Actives International, or those sold by the company
Euromed ; Euromed;
- les mélanges d'extraits de plantain, de berberis aquifolium et de salicylate de sodium tels que celui vendu sous la dénomination Seboclear® par la société Rahn ; mixtures of extracts of plantain, berberis aquifolium and sodium salicylate, such as the product sold under the name Seboclear® by the company Rahn;
- l'extrait de clou de girofle tel que celui vendu sous la dénomination Clove extract Powder par la société Maruzen ; clove extract, such as that sold under the name Clove Extract Powder by the company Maruzen;
- l'huile d'argan telle que celle vendue sous la dénomination Lipofructyl® par les Laboratoires Sérobiologiques ; - les filtrats de protéine lactique tels que celui vendu sous la dénomination Normaseb® par la société Sederma ; argan oil such as that sold under the name Lipofructyl® by Serobiological Laboratories; the lactic protein filtrates, such as that sold under the name Normaseb® by the company Sederma;
- les extraits d'algue laminaria, tel que celui vendu sous la dénomination Laminarghane® par la société Biotechmarine ; algae laminaria extracts, such as that sold under the name Laminarghane® by the company Biotechmarine;
- les extraits de sucre de canne, tel que celui commercialisé sous la dénomination Policasonol® par la société Sabinsa ; cane sugar extracts, such as the product sold under the name Policasonol® by Sabinsa;
-les oligosaccharides d'algue laminaria digitata tel que celui vendu sous la dénomination Phycosaccharide AC par la société Codif ; oligosaccharides of algae laminaria digitata such as that sold under the name Phycosaccharide AC by the company Codif;
- l'huile de schiste sulfonée, telle que celle vendue sous la dénomination Ichtyol Pale par la société Ichthyol ; sulphonated shale oil, such as that sold under the name Ichtyol Pale by the company Ichthyol;
- les extraits d'ulmaire (spiraea ulmaria) tels que celui vendu sous la dénomination Cytobiol Ulmaire par la société Libiol ; - extracts of ulmaire (spiraea ulmaria) such as that sold under the name Cytobiol Ulmaire by the company Libiol;
- l'acide sébacique, notamment vendu sous la forme d'un gel de polyacrylate de sodium sous la dénomination Sebosoft par la société Sederma ; sebacic acid, in particular sold in the form of a sodium polyacrylate gel under the name Sebosoft by Sederma;
- les glucomannanes extraits de tubercule de konjac et modifié par des chaînes alkylsulfonates tel que celui vendu sous la dénomination Biopol Beta par la société Arch Chemical ; glucomannans extracted from konjac tubers and modified with alkylsulphonate chains, such as the one sold under the name Biopol Beta by the company Arch Chemical;
- les extraits de Sophora angustifolia, tels que ceux vendus sous la dénomination Sophora powder ou Sophora extract par la société Bioland ; extracts of Sophora angustifolia, such as those sold under the name Sophora powder or Sophora extract by the company Bioland;
- les extraits de cinchona succirubra bark tel que celui vendu sous la dénomination le Red bark HS par la société Alban Muller ; extracts of Cinchona succirubra bark such as that sold under the name Red Bark HS by the company Alban Muller;
- les extraits de quillaja saponaria tel que celui vendu sous la dénomination Panama wood HS par la société Alban Muller ; extracts of quillaja saponaria, such as the product sold under the name Panama wood HS by Alban Muller;
- la glycine greffée sur chaîne undécylénique, telle que celle vendue sous la dénomination Lipacide UG OR par la société Seppic ; glycine grafted on an undecylenic chain, such as that sold under the name Lipacide UG OR by the company Seppic;
- le mélange d'acide oléanolique et d'acide nordihydroguaïarétique, tel que celui vendus sous la forme d'un gel sous la dénomination AC.Net par la société Sederma ; the mixture of oleanolic acid and nordihydroguaiaretic acid, such as that sold in the form of a gel under the name AC.Net by Sederma;
- le citrate de trialkyle(Ci2-Ci3) vendu sous la dénomination COSMACOL® ECI par la société Sasol ; le citrate de trialkyle(Ci4-Ci5) vendu sous la dénomination COSMACOL® ECL par la société Sasol ; trialkyl citrate (C12-C13) sold under the name COSMACOL® ECI by the company Sasol; trialkyl citrate (Ci 4 -C 5 ) sold under the name COSMACOL® ECL by the company Sasol;
- l'acide 10-hydroxydécanoïque, et notamment les mélanges d'acide 10- hydroxydécanoïque, d'acide sébacique et de 1 ,10-décandiol tels que celui vendu sous la dénomination Acnacidol® BG par la société Vincience ; - les activateurs PPAR-γ spécifiques comme ceux décrits dans la demande WO 2005/053632 ; 10-hydroxydecanoic acid, and in particular mixtures of 10-hydroxydecanoic acid, of sebacic acid and of 1,1-decandiol, such as that sold under the name Acnacidol® BG by the company Vincience; specific PPAR-γ activators, such as those described in application WO 2005/053632;
- des extraits de plantes du genre Silybum ; extracts of plants of the genus Silybum;
- des sapogénines ou extraits végétaux en contenant, en particulier les extraits de Dioscorées riches en diosgénine ; et sapogenins or plant extracts containing them, in particular extracts of Dioscorea rich in diosgenin; and
- des extraits d'Eugenia caryophyllata contenant de l'eugénol et du glucoside d'eugényle, et leurs mélanges. extracts of Eugenia caryophyllata containing eugenol and eugenyl glucoside, and mixtures thereof.
Comme agents séborégulateurs préférés utilisables selon l'invention, on citera : As preferred seboregulating agents that can be used according to the invention, mention may be made of:
- la criste marine ; - sea fennel;
- les mélanges d'extrait de canelle, de thé et d'octanoylglycine tel que le Sepicontrol A5 TEA de chez Seppic ; mixtures of cinnamon extract, tea and octanoylglycine such as Sepicontrol A5 TEA from Seppic;
- le mélange de capryloyl glycine, de sarcosine et d'extrait de cinnamomum zeylanicum commercialisé notamment par la société SEPPIC sous la dénomination commerciale Sepicontrol A5® ; the mixture of capryloyl glycine, sarcosine and cinnamomum zeylanicum extract marketed in particular by the company SEPPIC under the trade name Sepicontrol A5®;
- des sels de zinc, tels que le pyrrolidone carboxylate de zinc (ou pidolate de zinc), le lactate de zinc, l'aspartate de zinc, le carboxylate de zinc, le cystéate de zinc ; zinc salts, such as zinc pyrrolidone carboxylate (or zinc pidolate), zinc lactate, zinc aspartate, zinc carboxylate, zinc cysteate;
- des sels de cuivre, tels que le sulfate de cuivre ou le pyrrolidone carboxylate de cuivre; copper salts, such as copper sulphate or copper pyrrolidone carboxylate;
- les extraits de reine des prés (spiraea ulamaria) tel que celui vendu sous la dénomination Sébonormine® par la société Silab ; - Meadowwort extracts (Spiraea ulamaria) such as that sold under the name Sébonormine® by the company Silab;
- les extraits d'algue laminaria saccharina tel que celui vendu sous la dénomination Phlorogine par la société Biotechmarine ; algae extracts laminaria saccharina such as that sold under the name Phlorogine by the company Biotechmarine;
- les mélanges d'extraits de racines de pimprenelle (sanguisorba officinalis/poterium officinale), de rhizomes de gingembre (zingiber officinalis) et d'écorce de cannelier - mixtures of extracts of burnet root (Sanguisorba officinalis / Poterium officinale), ginger rhizomes (Zingiber officinalis) and cinnamon bark
(cinnamomum cassia) tel que celui vendu sous la dénomination Sebustop® par la société Solabia ; (cinnamomum cassia) such as that sold under the name Sebustop® by the company Solabia;
- les sapogénines ou extraits végétaux en contenant, en particulier les extraits de Dioscorées riches en diosgénine, sapogenins or plant extracts containing them, in particular extracts of Dioscorea rich in diosgenin,
et leurs mélanges. and their mixtures.
On peut encore citer les antitranspirants, tels que : les sels d'aluminium et/ou de zirconium ; les complexes d'hydroxychlorure de zirconium et d'hydroxychlorure d'aluminium avec un acide aminé tels que ceux décrits dans le brevet US-3792068 communément connus sous l'appellation "ZAG complexes". De tels complexes sont généralement connus sous l'appellation ZAG (lorsque l'acide aminé est la Glycine). Les complexes ZAG présentent d'ordinaire un quotient Al/Zr allant d'environ 1 ,67 à 12,5 et un quotient Métal/Cl allant d'environ 0,73 à 1 ,93. Parmi ces produits on peut citer l'aluminium zirconium octachlorohydrex GLY, l'aluminium zirconium pentachlorohydrex GLY, l'aluminium zirconium tetrachlorohydrate GLY et l'aluminium zirconium trichlorohydrate-GLY. Mention may also be made of antiperspirants, such as: aluminum and / or zirconium salts; complexes of zirconium hydroxychloride and hydroxychloride of aluminum with an amino acid such as those described in US-3792068 commonly known as "ZAG complexes". Such complexes are generally known by the name ZAG (when the amino acid is Glycine). The ZAG complexes usually have an Al / Zr quotient ranging from about 1.67 to 12.5 and a Metal / Cl quotient ranging from about 0.73 to 1.93. Among these products, mention may be made of aluminum zirconium octachlorohydrex GLY, aluminum zirconium pentachlorohydrex GLY, aluminum zirconium tetrachlorohydrate GLY and aluminum zirconium trichlorohydrate-GLY.
Parmi les sels d'aluminium, on peut citer le chlorhydrate d'aluminium, l'aluminium chlorohydrex, l'aluminium chlorohydrex PEG, l'aluminium chlorohydrex PG, l'aluminium dichlorohydrate, l'aluminium dichlorohydrex PEG, l'aluminium dichlorohydrex PG, l'aluminium sesquichlorohydrate, l'aluminium sesquichlorohydrex PEG, l'aluminium sesquichlorohydrex PG, les sels d'alun, l'aluminium sulfate, l'aluminium zirconium octachlorohydrate, l'aluminium zirconium pentachlorohydrate, l'aluminium zirconium tetrachlorohydrate, l'aluminium zirconium trichlorohydrate et plus particulièrement l'aluminium chlorhydrate commercialisé par la société REHEIS sous la dénomination MICRODRY ALUMINUM CHLOROHYDRATE ou par la société GUILINI CHEMIE sous la dénomination ALOXICOLL PF 40. Des sels d'aluminium et de zirconium sont par exemple celui commercialisé par la société REHEIS sous la dénomination REACH AZP-908-SUF , des sel d'aluminium activés par exemple celui commercialisé par la société REHEIS sous la dénomination REACH 103 ou par la société WESTWOOD sous la dénomination WESTCHLOR 200. Among the aluminum salts, mention may be made of aluminum chlorohydrate, chlorohydroxyl aluminum, chlorohydroxyl aluminum PEG, aluminum chlorohydrex PG, aluminum dichlorohydrate, aluminum dichlorohydrex PEG, aluminum dichlorohydrex PG, aluminum sesquichlorohydrate, aluminum sesquichlorohydrex PEG, aluminum sesquichlorohydrex PG, alum salts, aluminum sulfate, aluminum zirconium octachlorohydrate, aluminum zirconium pentachlorohydrate, aluminum zirconium tetrachlorohydrate, aluminum zirconium trichlorohydrate and more particularly the aluminum hydrochloride marketed by the company REHEIS under the name MICRODRY ALUMINUM CHLOROHYDRATE or by the company GUILINI CHEMIE under the name ALOXICOLL PF 40. Aluminum and zirconium salts are for example that marketed by the company REHEIS under the name REACH AZP-908-SUF, activated aluminum salt for example that marketed by the company REHEIS s or the REACH 103 denomination or by WESTWOOD under the name WESTCHLOR 200.
Parmi les autres actifs déodorants, on peut citer également les sels de zinc comme le sulfate de zinc, le chlorure de zinc, le lactate de zinc, le phénolsulfonate de zinc ; la chlorhexidine et les sels; le monocaprate de diglycérol, le monolaurate de diglycérol , monolaurate de glycérol ; les sels de polyhexaméthylène biguanide. Other deodorant active agents also include zinc salts such as zinc sulphate, zinc chloride, zinc lactate, zinc phenolsulphonate; chlorhexidine and salts; diglycerol monocaprate, diglycerol monolaurate, glycerol monolaurate; salts of polyhexamethylene biguanide.
Agents anti-microbiens Anti-microbial agents
Par agents anti-microbiens, on entend des agents ayant des effets sur la flore spécifique des peaux grasses, tels que par exemple le P acnés. Ces effets peuvent être soit bactéricides, soit anti-adhésion bactérienne (prévient et/ou réduit l'adhésion des micro-organismes) soit agissant sur le biofilm des bactéries pour éviter leur multiplication. By anti-microbial agents is meant agents having effects on the specific flora of oily skin, such as, for example, P acnes. These effects can be either bactericidal or bacterial anti-adhesion (prevents and / or reduces the adhesion of microorganisms) or acting on the biofilm of bacteria to prevent their multiplication.
On peut citer notamment les actifs et conservateurs à activité anti-microbienne cités dans la demande DE10324567, incorporée dans la présente invention par référence. Mention may in particular be made of the active agents and preservatives with antimicrobial activity mentioned in application DE10324567, incorporated in the present invention by reference.
On peut citer également : un extrait de cône de houblon (HOP CO2-TO extract de Flavex), un extrait de Millepertius (ST John's Wort CO2-TO extract de Flavex), l'acide asiatique, les extraits de racines de scutellaria baicolensis comme dans le BMB - CF de Naturogin, la piroctone olamine, l'acide citrollique, l'acide spérillique, l'ethylhexylglycerine (le Sensiva De Shulke), le caprylate/caprate de gluceryl (Capmul de ABITEC), le calcium sodium phosphosilicate comme Bioactive glasspowder de Schott, les Actysse premier BG de Schott, les oxydes de silicium de Ciba, les Métashines (les dérivés d'argent), les extraits de busserole comme la Gatuline equalizing de Gattefossé, l'acide 10-hydroxy-2 decanoique comme l'Acnacidol P de Vincience, l'ursolate de sodium, l'acide azélaique, le di-iodo-methyl P tolylsulfone ou l'Amical Flowable de Angus, la Malachite de Maprecos, le Zincare de Elementis GMBH, l'arlatone dioic de Unichema ; l'acide ellagique ; le 2,4,4'-trichloro-2'-hydroxy diphényl éther (ou triclosan), la 1 -(3',4'-dichlorophenyl)-3-(4'-chlorophenyl)urée (ou triclocarban), le 3,4,4'-trichlorocarbanilide, le 3',4',5'-trichlorosalicylanilide, le phénoxyéthanol, le phénoxypropanol, le phénoxyisopropanol, l'hexamidine iséthionate, le métronidazole et ses sels, le miconazole et ses sels, l'itraconazole, le terconazole, l'éconazole, le ketoconazole, le saperconazole, le fluconazole, le clotrimazole, le butoconazole, l'oxiconazole, le sulfaconazole, le sulconazole, le terbinafine, le ciclopirox, le ciclopiroxolamine, l'acide undécylenique et ses sels, le peroxyde de benzoyle, l'acide 3-hydroxy benzoïque, l'acide 4-hydroxy benzoïque, l'acide phytique, la N-acétyl-L-cystéine, l'acide lipoïque, l'acide azélaïque et ses sels, l'acide arachidonique, le résorcinol, le 3,4,4'-trichlorocarbanalide, l'octopirox ou piroctone olamine, l'octoxyglycérine ou octoglycérine, l'octanoylglycine (Lipacid C8G® de Seppic), le caprylyl glycol, l'acide 10-hydroxy-2-décanoïque, le dichlorophenyl imidazol dioxolan et ses dérivés décrits dans la demande de brevet W09318743, les dérivés de zinc et en particulier le pidolate de zinc (Zincidone® de Solabia), les dérivés de cuivre, l'acide salicylique et d'autres dérivés de l'acide salicylique, l'iodopropynyl butylcarbamate, le 3,7,1 1 -triméthyldodéca-2, 5,10-triénol ou farnesol, les phytosphingosines ; le Sepicontrol® de Seppic, un extrait d'arganier connnne l'Argapure LS9710®, le Sebosoft® de Sederma, les sels d'ammonium quaternaires comme les sels de cetyltrimethylammonium, les sels de cétylpyridinium, l'éthanol... et leurs mélanges. We can also mention: a hop cone extract (HOP CO2-TO extract from Flavex), a Millepertius extract (ST John's Wort CO2-TO Flavex extract), Asian acid, extracts of scutellaria baicolensis roots as in Naturogin BMB-CF, piroctone olamine, citrollic acid, sperillic acid, ethylhexylglycerine (Sensiva De Shulke), caprylate / gluceryl caprate (ABITEC Capmul), calcium sodium phosphosilicate as Bioactive Schott's glasspowder, Schott's Actysse first BG, Ciba's silicon oxides, Metashines (silver derivatives), Bearberry extracts like Gattefossé's Gatuline equalizing, 10-hydroxy-2 decanoic acid as well as Vincient Acnacidol P, sodium ursolate, azelaic acid, di-iodomethyl P tolylsulfone or Angus Flowable Amicale, Malachite from Maprecos, Zincare from Elementis GMBH, arlatone dioic from Unichema ; ellagic acid; 2,4,4'-trichloro-2'-hydroxy-diphenyl ether (or triclosan), 1- (3 ', 4'-dichlorophenyl) -3- (4'-chlorophenyl) urea (or triclocarban), 3 4,4'-trichlorocarbanilide, 3 ', 4', 5'-trichlorosalicylanilide, phenoxyethanol, phenoxypropanol, phenoxyisopropanol, hexamidine isethionate, metronidazole and its salts, miconazole and its salts, itraconazole, terconazole, econazole, ketoconazole, saperconazole, fluconazole, clotrimazole, butoconazole, oxiconazole, sulfaconazole, sulconazole, terbinafine, ciclopirox, ciclopiroxolamine, undecylenic acid and its salts, benzoyl peroxide, 3-hydroxy benzoic acid, 4-hydroxy benzoic acid, phytic acid, N-acetyl-L-cysteine, lipoic acid, azelaic acid and its salts, acid arachidonic acid, resorcinol, 3,4,4'-trichlorocarbanalide, octopirox or piroctone olamine, octoxyglycerine or octoglycerine, octanoylglycine (Lipacid C8G® from Seppic), ca prylyl glycol, 10-hydroxy-2-decanoic acid, dichlorophenyl imidazol dioxolan and its derivatives described in patent application WO9318743, zinc derivatives and in particular zinc pidolate (Zincidone® from Solabia), derivatives of copper, salicylic acid and other acid derivatives salicylic acid, iodopropynyl butylcarbamate, 3,7,1 1 -trimethyldodeca-2,5,10-trienol or farnesol, phytosphingosines; Sepicontrol® from Seppic, an argan extract known as Argapure LS9710®, Sebosoft® from Sederma, quaternary ammonium salts such as cetyltrimethylammonium salts, cetylpyridinium salts, ethanol, and mixtures thereof. .
Comme agents prévenant et/ou réduisant l'adhésion des micro-organismes, on peut citer notamment : le phytanetriol et ses dérivés tels que décrits dans la demande de brevet EP 1 529 523, les huiles végétales telles de l'huile de germe de blé, l'huile de calendula, l'huile de ricin, l'huile d'olive, l'huile d'avocat, l'huile d'amande douce, l'huile d'arachide, l'huile de jojoba, l'huile de sésame, l'huile d'amande d'abricot, l'huile de tournesol, l'huile de macadamia, décrites dans le brevet EP 1 133 979, ou encore d'autres corps gras tels que le cocoamphodiacétate disodique, le cocoate de glycéryle oxyéthyléné (7 OE), les Poloxamers, l'hexadécénylsuccinate 18, le palmitate d'octoxyglycéryle, le béhénate d'octoxyglycéryle, l'adipate de dioctyle , le PPG-15 stéaryl éther, le tartrate de di-alcools C12-C13 ramifiés décrits dans le brevet EP 1 129 694. As agents that prevent and / or reduce the adhesion of microorganisms, mention may be made in particular of: phytanetriol and its derivatives as described in patent application EP 1 529 523, vegetable oils such as wheat germ oil , calendula oil, castor oil, olive oil, avocado oil, sweet almond oil, peanut oil, jojoba oil, sesame oil, apricot kernel oil, sunflower oil, macadamia oil, described in patent EP 1 133 979, or other fatty substances such as cocoamphodiacetate disodium, cocoate of oxyethylenated glyceryl (7 EO), Poloxamers, hexadecenylsuccinate 18, octoxyglyceryl palmitate, octoxyglyceryl behenate, dioctyl adipate, PPG-15 stearyl ether, C12-C13 di-alcohol tartrate branched derivatives described in EP 1 129 694.
En particulier vis-à-vis de la propagation du P acnés, on peut citer le pentylène glycol, le nylon-66 (fibres de polyamides 66), l'huile de son de riz , le Celvol 540 PV alcohol (Polyvinyl alcohol 72962), l'Akorex L de Karlshamns, les dérivés de fructose. In particular with respect to the propagation of P acnnes, mention may be made of pentylene glycol, nylon-66 (polyamide 66 fibers), rice bran oil, Celvol 540 PV alcohol (Polyvinyl alcohol 72962) , Akorex L from Karlshamns, fructose derivatives.
On peut également citer certains tensioactifs ayant un effet antimicrobien comme le cocoampho acétate de sodium ou diacetate disodium tel que le Miranol C2M CONC NP, les bétaines comme la cocoyl bétaine Genagen KB de Clariant, le lauryl ether sulfate de sodium comme l'Emal 270 D de Kao, le décyl glucoside comme le Plantacare 2000 UP, les malate de dialcools C12-13 ramifiés comme le Cosmacol EMI, les monoesters de propylène glycol comme monolaurate, monocaprylate, monocaprate de propylène glycol, le sodium lauroyl oat amino acid comme le Proteol OAT, le lauryl dimethylamine betaine comme le Empigen BB/LS ainsi que les polyammonium quaternaires comme le Quaternium-24 ou Bardac 2050 de Lonza et ceux décrits dans le brevet L'OREAL FR 0 108 283. Comnne agents antimicrobiens préférés, on utilisera dans les compositions de l'invention un agent choisi parmi le caprylyl glycol, les dérivés de zinc dont le pidolate de zinc (Zincidone® de Solabia), les dérivés de cuivre, l'octoglycérine ou octoxyglycérine, l'acide 10-hydroxy-2-décanoïque, et leurs mélanges. Mention may also be made of certain surfactants having an antimicrobial effect such as cocoampho sodium acetate or diacetate disodium such as Miranol C2M CONC NP, betaines such as cocoyl betaine Genagen KB from Clariant, sodium lauryl ether sulfate such as Emal 270 D Kao, decyl glucoside such as Plantacare 2000 UP, C12-13 dialkyl malate branched as Cosmacol EMI, propylene glycol monoesters such as monolaurate, monocaprylate, propylene glycol monocaprate, sodium lauroyl oat amino acid such as Proteol OAT , lauryl dimethylamine betaine such as Empigen BB / LS as well as quaternary polyammoniums such as Quaternium-24 or Bardac 2050 from Lonza and those described in the L'OREAL patent FR 0 108 283. As preferred antimicrobial agents, the compositions of the invention will use an agent chosen from caprylyl glycol, zinc derivatives including zinc pidolate (Zincidone® from Solabia), copper derivatives, octoglycerine or octoxyglycerine, and 10-hydroxy-2-decanoic acid, and mixtures thereof.
Agents desquamants Desquamating agents
Par "agent desquamant", on entend tout composé capable d'agir : By "desquamating agent" is meant any compound capable of acting:
- soit directement sur la desquamation en favorisant l'exfoliation, tel que les β- hydroxyacides, en particulier l'acide salicylique et ses dérivés (dont l'acide n-octanoyl 5-salicylique) ; les α-hydroxyacides, tels que les acides glycolique, citrique, lactique, tartrique, malique ou mandélique ; l'urée ; l'acide gentisique et ses dérivés ; les oligofucoses ; l'acide cinnamique ; l'extrait de Saphora japonica ; le resvératrol et certains dérivés d'acide jasmonique ; - soit sur les enzymes impliquées dans la desquamation ou la dégradation des cornéodesmosomes, les glycosidases, la stratum corneum chymotryptic enzym (SCCE) voire d'autres protéases (trypsine, chymotrypsine-like). On peut citer les composés aminosulfoniques et en particulier l'acide (N-2 hydroxyéthylpiperazine-N- 2-éthane) sulfonique (HEPES) ; les dérivés de l'acide 2-oxothiazolidine-4- carboxylique (procystéine) ; les dérivés d'acides alpha aminés de type glycine (tels que décrits dans EP-0 852 949, ainsi que le méthyl glycine diacétate de sodium commercialisé par BASF sous la dénomination commerciale TRILON M) ; le miel ; les dérivés de sucre tels que l'O-octanoyl-6-D-maltose et la N-acétyl glucosamine. Comme autres agents desquamants utilisables dans la composition selon l'invention, on peut citer les oligofructoses, l'EDTA et ses dérivés, les laminaria extract, le o- linoleyl-6D-glucose, le (3-hydroxy-2pentylcyclopentyl)acetic acid, le trilactate de glycérol, l'O-octanyl-6'-D-maltose, la S carboxyméthyl cystéine, les dérivés siliciés de salicylate comme dans le brevet EP 0 796 861 , les oligofucase comme dans le brevet EP 0 218 200, les sels d'acide 5-acyl salicylique, des actifs ayant des effets sur la transglutaminase comme dans le brevet EP 0 899 330, l'acide jasmonique et dérivés comme dans les demandes de brevet EP 1 333 022 et EP 1 333 021 , Exfolactive® de Silab (extrait de fleur de ficus opuntia indica), le Soypon O® de Kawaken fine chemicals (sodium cocoyl sarcosinate) or directly on desquamation by promoting exfoliation, such as β-hydroxy acids, in particular salicylic acid and its derivatives (including n-octanoyl-5-salicylic acid); α-hydroxy acids, such as glycolic, citric, lactic, tartaric, malic or mandelic acids; urea; gentisic acid and its derivatives; oligofucoses; cinnamic acid; Saphora japonica extract; resveratrol and certain jasmonic acid derivatives; or on the enzymes involved in the desquamation or degradation of corneodesmosomes, glycosidases, stratum corneum chymotryptic enzyme (SCCE) or even other proteases (trypsin, chymotrypsin-like). Mention may be made of aminosulfonic compounds and in particular (N-2-hydroxyethylpiperazine-N-2-ethane) sulfonic acid (HEPES); 2-oxothiazolidine-4-carboxylic acid derivatives (procysteine); glycine-type alpha amino acid derivatives (as described in EP-0 852 949, as well as the sodium methyl glycine diacetate marketed by BASF under the trade name TRILON M); honey ; sugar derivatives such as O-octanoyl-6-D-maltose and N-acetyl glucosamine. Other desquamating agents that can be used in the composition according to the invention include oligofructoses, EDTA and its derivatives, laminaria extract, o-linoleyl-6D-glucose, (3-hydroxy-2-pentylcyclopentyl) acetic acid, glycerol trilactate, O-octanyl-6'-D-maltose, S carboxymethyl cysteine, the silicon derivatives of salicylate as in patent EP 0 796 861, oligofucase as in patent EP 0 218 200, salts 5-acyl salicylic acid, active agents having effects on transglutaminase as in patent EP 0 899 330, jasmonic acid and derivatives as in patent applications EP 1 333 022 and EP 1 333 021, Exfolactive® from Silab (ficus opuntia indica flower extract), Soypon O® from Kawaken fine chemicals (sodium cocoyl sarcosinate)
Comme agents desquamants préférés, on pourra citer les beta-hydroxyacides, tel que l'acide n-octanoyl 5-salicylique ; l'urée ; les acides glycolique, citrique, lactique, tartrique, malique ou mandélique ; l'acide (N-2 hydroxyéthylpiperazine-N-2-éthane) sulfonique (HEPES) ; l'extrait de Saphora japonica ; le miel ; le N-acétyl glucosamine ; le méthyl glycine diacétate de sodium, et leurs mélanges. Encore plus préférentiellement on utilisera dans les compositions de l'invention un agent desquamant choisi parmi l'acide n-octanoyl 5-salicylique ; l'urée ; l'acide (N-2 hydroxyéthylpiperazine-N-2-éthane) sulfonique (HEPES) ; l'extrait de Saphora japonica ; le miel ; le Nacétyl glucosamine ; le méthyl glycine diacétate de sodium, et leurs mélanges. Preferred desquamating agents include beta-hydroxy acids such as n-octanoyl-5-salicylic acid; urea; glycolic, citric, lactic, tartaric, malic or mandelic acids; (N-2-hydroxyethylpiperazine-N-2-ethane) sulfonic acid (HEPES); Saphora japonica extract; honey ; N-acetyl glucosamine; sodium methyl glycine diacetate, and mixtures thereof. Even more preferentially, it will be used in the compositions of the invention a desquamating agent chosen from n-octanoyl 5-salicylic acid; urea; (N-2-hydroxyethylpiperazine-N-2-ethane) sulfonic acid (HEPES); Saphora japonica extract; honey ; Nacetyl glucosamine; sodium methyl glycine diacetate, and mixtures thereof.
Agents apaisants ou anti-irritants Soothing or anti-irritant agents
On peut citer notamment les agents apaisants ou anti-irritants cités dans les demandes WO2004/105736 et DE10324567, incorporées dans la présente invention par référence. Mention may in particular be made of the soothing or anti-irritant agents cited in the applications WO2004 / 105736 and DE10324567, incorporated in the present invention by reference.
Comme agents apaisants particuliers utilisables dans la composition selon l'invention, on peut citer : les oligomères procyannidoliques, les vitamines E, C, B5, B3, le sulfate de dextran, la caféine et ses dérivés, les triterpènes pentacycliques et les extraits de plantes les contenant, l'acide b-glycyrrhétinique et ses sels ou dérivés (le stearyl glycyrrhetate, l'acide 3-stéaroyloxy glycyrrhétique, l'acide glycyrrhétinique monoglucuronide) ainsi que les plantes en contenant (ex : Glycyrrhiza glabra), l'acide oléanolique et ses sels, l'acide ursolique et ses sels, l'acide boswellique et ses sels, l'acide bétulinique et ses sels, un extrait de Paeonia suffruticosa et/ou lactiflora, les phycosshacharides de la société Codif, un extrait de Laminaria saccharina, les extraits de Centella asiatica, l'huile de Canola, le bisabolol, le diesterphosphorique de vitamine E et C comme le Sepivital EPC® de Seppic, les extraits de camomille, l'allantoïne, les huiles insaturées en oméga 3 telles que les huiles de rosier musca, de cassis, d'Ecchium, de poisson, l'huile de calophilum, des extraits de plancton, la capryloyl glycine, le Seppicalm VG® (nymphéa alba et sodium palmitoylproline) de Seppic, un extrait de Pygeum, un extrait de Boswellia serrata, un extrait de Centipeda cunnighami, un extrait d'Helianthus annus en particulier l'Hélioxine de Silab, un extrait de Linum usitatissimum comme la Sensiline de Silab, les tocotriénols, les extraits de Cola nitida, le piperonal, un extrait de clou de girofle, un extrait d'Epilobium angustifolium, l'aloe vera, un extrait de Bacopa moniera, les phytostérols, l'eau de bleuet, l'eau de rose, le dextran comme dans Modulène® de Vincience, un extrait de menthe, en particulier de feuilles de menthe comme le Calmiskin® de Silab, les dérivés d'anis, les bactéries filamenteuse comme Vitreoscilla filiformis tel que décrit dans le brevet EP 761 204, un extrait de rose comme l'Herbasol rose extract, le Stimutex AS de Pentapharm, les sels alcalino- terreux notamment le strontium, la niacinamide, et leurs mélanges. As particular soothing agents that may be used in the composition according to the invention, mention may be made of: procyannidol oligomers, vitamins E, C, B5, B3, dextran sulphate, caffeine and its derivatives, pentacyclic triterpenes and plant extracts containing them, b-glycyrrhetinic acid and its salts or derivatives (stearyl glycyrrhetate, 3-stearoyloxy glycyrrhetic acid, glycyrrhetinic acid monoglucuronide) and the plants containing it (eg Glycyrrhiza glabra), oleanolic acid and its salts, ursolic acid and its salts, boswellic acid and its salts, betulinic acid and its salts, an extract of Paeonia suffruticosa and / or lactiflora, the phycosshacharides of Codif, an extract of Laminaria saccharina , extracts of Centella asiatica, canola oil, bisabolol, vitamin E and C diesterphosphoric acid, such as Seppic's Sepivital EPC®, chamomile extracts, allantoin, unsaturated 3 such as rose bush, black currant, ecchium, fish, calophilum oil, plankton extracts, capryloyl glycine, Seppic Seppicalm VG® (waterlily alba and sodium palmitoylproline), an extract of Pygeum, an extract of Boswellia serrata, an extract of Centipeda cunnighami, an extract of Helianthus annus, in particular Helioxin of Silab, an extract Linum usitatissimum such as Silab Sensilin, tocotrienols, Cola nitida extracts, piperonal, clove extract, Epilobium angustifolium extract, aloe vera, Bacopa moniera extract, phytosterols, l cornflower water, rose water, dextran as in Vincience Modulène®, mint extract, especially mint leaves such as Silab's Calmiskin®, anise derivatives, filamentous bacteria such as Vitreoscilla filiformis as described in patent EP 761 204, a pink extract such as Herbasol rose extract, Stimutex AS from Pentapharm, alkaline earth salts including strontium, niacinamide, and mixtures thereof.
Comme agents apaisants préférés, on utilisera un agent choisi parmi un extrait de rose, un extrait de clou de girofle, le dextran comme dans Modulène® de Vincience, un extrait de menthe comme le Calmiskin® de Silab, un mélange d'un extrait de nymphéa alba et sodium palmitoylproline comme le Seppicalm VG® de Seppic, des dérivés d'anis, un extrait de Paeonia suffruticosa et/ou lactiflora, et leurs mélanges. Agents anti-inflammatoires As preferred soothing agents, use will be made of an agent chosen from a rose extract, a clove extract, dextran as in Vincience's Modulene®, a mint extract such as Silab's Calmiskin®, a mixture of an extract of waterlily alba and sodium palmitoylproline such as Seppic Seppicalm VG®, anise derivatives, an extract of Paeonia suffruticosa and / or lactiflora, and mixtures thereof. Anti-inflammatory agents
On peut citer notamment les agents anti-inflammatoires cités dans les demandes WO2004/105736 et DE10324567, incorporées dans la présente invention par référence. Comme agents anti-inflammatoires particuliers utilisables selon l'invention, on peut citer la cortisone, l'hydrocortisone, l'indométhacine, la bétaméthasone, l'acide azéalique, l'acétominophène, le diclofénac, le propionate de clobetasol et leurs mélanges. Comme agent anti-inflammatoire préféré, on citera l'acide azélaique. In particular, mention may be made of the anti-inflammatory agents mentioned in the applications WO2004 / 105736 and DE10324567, incorporated in the present invention by reference. Particular antiinflammatory agents that can be used according to the invention include cortisone, hydrocortisone, indomethacin, betamethasone, azealic acid, acetaminophen, diclofenac, clobetasol propionate and mixtures thereof. As a preferred anti-inflammatory agent, mention will be made of azelaic acid.
Agents antioxydants On entend définir par cette expression les agents ayant une activité anti-oxydante (qui empêchent l'oxydation du squalène et la formation des comédons). Antioxidants By this expression is meant to define the agents having an antioxidant activity (which prevent the oxidation of squalene and the formation of comedones).
On peut citer notamment le tocophérol et ses esters, en particulier l'acétate de tocophérol ; le BHT et le BHA. There may be mentioned in particular tocopherol and its esters, in particular tocopherol acetate; BHT and BHA.
On peut également citer les polyphénols, l'acide tannique, l'époigallocathéchines et les extraits naturels en contenant, les anthocyanes, les extraits de romarin, les extraits de feuilles d'olivier, le thé vert, le resvératrol et ses dérivés, le Pycnogénol, l'ergothinéine, la N acétylcystéine, la biotine, les chélatants, l'idébénone, des extraits végétaux corne le Pronalen Bioprotect TM de la société Provital, les antiradicalaires comme la vitamine E, le co enzyme Q10, les bioflavonoides, les SOD, le phytantriol, les lignanes, la mélatonine, les pidolates, le gluthation. There may also be mentioned polyphenols, tannic acid, epoigallocathechins and natural extracts containing them, anthocyanins, rosemary extracts, olive leaf extracts, green tea, resveratrol and its derivatives, Pycnogenol ergothineine, N acetylcysteine, biotin, chelants, idebenone, plant extracts Pronital Proniten Bioprotect TM from Provital, antiradicals such as vitamin E, coenzyme Q10, bioflavonoids, SOD, phytantriol, lignans, melatonin, pidolates, glutathione.
Agents cicatrisants Healing agents
Comme exemples d'agents cicatrisants, on peut citer notamment : As examples of cicatrizing agents, mention may in particular be made of:
l'allantoine, l'urée, l'huile de germe de blé, certains acides aminés comme l'hydroxyproline, l'arginine, la sérine, et aussi des extraits de lys blanc (ex : le Phytélène Lys 37EG 16295 de Indena), un extrait de levures comme le cicatrisant LS 7225B de LS (Cognis), l'huile de tamanu, l'extrait de saccharomyces cerevisiae ou Biodynes TRF de Arch Chemical, les extraits d'avoine, le chitosane et dérivés, les extraits de carotte, l'extrait d'artemia ou GP4G de Vincience, l'acexamate de sodium, des extraits de lavandin, des extraits de miel ou de propolis, l'acide ximeninique et ses sels tel que acido ximeninico de Indena, l'huile de rosa rugosa, le Souci Ami Liposolible d'Alban Muller, des extraits de prêle, l'herbasol citron de Cosmetochem, des extraits d'helichryse, des bétaglucan et dérivés, du beurre de karité et ses fractions purifiées, les exopolysaccharides modifiés et les Polyaminosaccharides alkylsulfonnés. allantoin, urea, wheat germ oil, certain amino acids such as hydroxyproline, arginine, serine, and also extracts of white lily (eg Phytelene Lys 37EG 16295 from Indena), a yeast extract such as the LS 7225B healing agent from LS (Cognis), tamanu oil, Saccharomyces cerevisiae extract or Biodynes TRF from Arch Chemical, oat extracts, chitosan and derivatives, carrot extracts, Vincenzo artemia or GP4G extract, sodium acexamate, lavandin extracts, extracts of honey or propolis, ximeninic acid and its salts, such as Indena's acidoximininico, rosa rugosa oil the Alban Muller Liposolble-Souri, horsetail extracts, cosmetochem lemon herbasol, helichryse extracts, betaglucan and derivatives, shea butter and purified fractions thereof, modified exopolysaccharides and alkylsulfonated polyaminosaccharides.
Comme agents cicatrisants préférés selon l'invention, on utilisera l'huile de tamanu, l'acexamate de sodium, des extraits de miel, des extraits de prêle, des extraits d'helichryse, et leurs mélanges. Preferred healing agents according to the invention are tamanu oil, sodium acexamate, honey extracts, horsetail extracts, helichryse extracts, and mixtures thereof.
Agents astringents Par "agents astringents", on entend selon l'invention des agents permettant de lutter contre la dilatation des follicules sébacés. Astringent agents By "astringent agents" is meant according to the invention agents for controlling the dilation of the sebaceous follicles.
Comme agents astringents utilisables dans la composition selon l'invention, on peut citer le Laricyl LS8865® de Cognis, le Phytofirm LS9120® de Cognis , le Tanlex VEA/B® de Ichimaru Pharcos, la laponite, les sels d'aluminium, les extraits de centella (ex Plantactiv centella de Cognis), le Varisoft 432 CG® de Degussa, les extraits de marron dinde, les extraits de mauve, d'Hammamelis, l'Almondermin LS 3380® de Cognis, les extraits de bardane, l'Extrapone 9 specia®l de Symrise, les extraits de scutellaria, les extrais d'Ulmaire (ex Cytobiol Ulmaire de Libiol), l'Herb extract B1348® de Bell flavors & fragrances, les extraits d'acacia, d'orme, de saule blanc, de canelle, de bouleau, de reine des prés, les sapogénines de panama, le phenolsulfonate de zinc de Interchemical, des extraits de gentiane, de concombre, de noyer, le mélange Epilami de Alban Muller. As astringent agents that can be used in the composition according to the invention, mention may be made of Laricyl LS8865® from Cognis, Phytofirm LS9120® from Cognis, Tanlex VEA / B® from Ichimaru Pharcos, laponite, aluminum salts, extracts of centella (formerly Plantactiv centella of Cognis), Varisoft 432 CG® by Degussa, extracts of chestnut turkey, extracts of mauve, Hammamelis, Almondermin LS 3380® from Cognis, extracts of burdock, Extrapone 9 Symrise specia®l, scutellaria extracts, Ulmaire extracts (ex Cytobiol Ulmaire de Libiol), Bell flavors & fragrances B1348® Herb extract, acacia, elm and white willow extracts , cinnamon, birch, meadowsweet, panama sapogenins, Interchemical zinc phenolsulfonate, gentian extracts, cucumber, walnut, Alban Muller's Epilami blend.
Comme agents astringents préférés selon l'invention, on utilisera les extrais de scutellaria, les extraits d'ulmaire, les extraits de reine des près, les extraits de gentiane, les extraits de bardane et leurs mélanges. Selon un mode particulier de l'invention, on pourra encore ajouter à ces ingrédients et/ou actifs de soin de la peau grasse d'autres agents destinés à embellir l'aspect et/ou la texture de la peau. En particulier, on pourra ajouter dans les compositions de l'invention des actifs utilisés pour prévenir et/ou traiter les signes du vieillissement cutané. Preferred astringent agents according to the invention are scutellaria extract, ulmary extract, close-up queen extract, gentian extract, burdock extract and mixtures thereof. According to a particular embodiment of the invention, it will be possible to add to these ingredients and / or active agents for the treatment of oily skin other agents intended to embellish the appearance and / or the texture of the skin. In particular, it will be possible to add, in the compositions of the invention, active agents used to prevent and / or treat the signs of skin aging.
Le ou les actifs et/ou ingrédients additionnels utilisés dans la composition selon l'invention peuvent représenter de 0,0001 à 20%, de préférence de 0,01 à 10% et mieux, de 0,01 à 5% en poids par rapport au poids total de la composition. Selon un mode particulier, la composition selon l'invention est destinée au soin et/ou au traitement de personnes ayant une peau de phototype de III à VI. The additional active ingredient (s) and / or ingredient (s) used in the composition according to the invention may represent from 0.0001 to 20%, preferably from 0.01 to 10% and better still from 0.01 to 5% by weight relative to to the total weight of the composition. According to one particular embodiment, the composition according to the invention is intended for the care and / or the treatment of persons having a phototype skin of III to VI.
En effet, les imperfections cutanées des peaux grasses peuvent être plus ou moins visibles selon le phototype des sujets, en particulier sur les peaux de sujets à phototype élevé, c'est-à dire des peaux de sujets à phototype III à VI, définis selon la classification de Fitzpatrick qui repose sur la réactivité de la peau aux effets du rayonnement solaire : Indeed, skin imperfections of oily skin may be more or less visible depending on the phototype of the subjects, especially on the skins of subjects to high phototype, ie skin of subjects with phototype III to VI, defined according to the Fitzpatrick classification which is based on the reactivity of the skin to the effects of solar radiation:
I brûle toujours, ne bronze jamais I always burn, never bronze
II brûle toujours, bronze peu He still burns, little bronze
III brûle modérément, bronze progressivement III burns moderately, bronze gradually
IV brûle faiblement, bronze très facilement IV burns weakly, bronze very easily
V brûle rarement, bronze intensément V rarely burns, intensely bronze
VI ne brûle jamais, fortement pigmenté. VI never burns, strongly pigmented.
Les compositions selon l'invention sont préférentiellement des compositions cosmétiques de soin de la peau grasse. The compositions according to the invention are preferably cosmetic compositions for the care of oily skin.
Ces compositions sont destinées à une application topique sur le visage et/ou le corps. En particulier, la composition est appliquée sur les zones du visage ou du front présentant une brillance de la peau. Il pourra également s'agir de compositions de rasage ou d'après-rasage, en particulier pour hommes. These compositions are intended for topical application on the face and / or the body. In particular, the composition is applied to the areas of the face or the forehead having a gloss of the skin. It may also be shaving compositions or aftershave, especially for men.
La composition cosmétique et/ou dermatologique selon l'invention peut contenir également des adjuvants habituels dans le domaine cosmétique ou dermatologique, tels que les gélifiants hydrophiles ou lipophiles, les conservateurs, les solvants, les parfums, les charges, les filtres UV, les bactéricides, les absorbeurs d'odeur, les matières colorantes, les extraits végétaux, les sels, les antioxydants, les agents basiques, les acides, les tensioactifs non ioniques, anioniques, cationiques. The cosmetic and / or dermatological composition according to the invention may also contain adjuvants which are customary in the cosmetic or dermatological field, such as hydrophilic or lipophilic gelling agents, preservatives, solvents, perfumes, fillers, UV filters and bactericides. , odor absorbers, dyestuffs, plant extracts, salts, antioxidants, basic agents, acids, nonionic, anionic, cationic surfactants.
Les quantités de ces différents adjuvants sont celles classiquement utilisées dans le domaine considéré, et par exemple de 0,01 à 20 % du poids total de la composition. Ces adjuvants, selon leur nature, peuvent être introduits dans la phase grasse, dans la phase aqueuse et/ou dans les vésicules lipidiques. The amounts of these various adjuvants are those conventionally used in the field under consideration, and for example from 0.01 to 20% of the total weight of the composition. These adjuvants, depending on their nature, can be introduced into the fatty phase, into the aqueous phase and / or into the lipid vesicles.
Comme charges qui peuvent être utilisées dans la composition de l'invention, on peut citer par exemple, outre les pigments, la poudre de silice ; le talc ; les particules de polyamide et notamment celles vendues sous la dénomination ORGASOL par la société Atochem ; les poudres de polyéthylène ; les micro-sphères à base de copolymères acryliques, telles que celles en copolymère diméthacrylate d'éthylène glycol/ methacrylate de lauryle vendues par la société Dow Corning sous la dénomination de POLYTRAP ; les poudres expansées telles que les microsphères creuses et notamment, les microsphères commercialisées sous la dénomination EXPANCEL par la société Kemanord Plast ou sous la dénomination MICROPEARL F 80 ED par la société Matsumoto ; les microbilles de résine de silicone telles que celles commercialisées sous la dénomination TOSPEARL par la société Toshiba Silicone ; et leurs mélanges. Ces charges peuvent être présentes dans des quantités allant de 0 à 20 % en poids et de préférence de 1 à 10 % en poids par rapport au poids total de la composition. As fillers which can be used in the composition of the invention, mention may be made, for example, besides the pigments, the silica powder; talcum; polyamide particles and especially those sold under the name ORGASOL by the Atochem company; polyethylene powders; micro-spheres based on acrylic copolymers, such as those made of ethylene glycol dimethacrylate / lauryl methacrylate copolymer sold by Dow Corning under the name Polytrap; expanded powders such as hollow microspheres and in particular the microspheres sold under the name Expancel by the company Kemanord Plast or under the name Micropearl F 80 ED by the company Matsumoto; silicone resin microbeads such as those sold under the name Tospearl by the company Toshiba Silicone; and their mixtures. These fillers may be present in amounts ranging from 0 to 20% by weight and preferably from 1 to 10% by weight relative to the total weight of the composition.
Comme gélifiants hydrophiles ou lipophiles, on peut citer notamment les carbopol, les luvigel, l'Hostacerin AMPS, le Simulgel, les Sepigel, les gommes de xanthane, de guar, de cellulose, les alginates et leurs mélanges. On peut citer aussi les hectorites. As hydrophilic or lipophilic gelling agents, there may be mentioned in particular carbopol, luvigel, Hostacerin AMPS, Simulgel, Sepigel, xanthan, guar, cellulose gums, alginates and mixtures thereof. We can also mention hectorites.
Comme agents antioxydants, on peut citer notamment les polyphénols, l'acide tannique, l'époigallocathéchines et les extraits naturels en contenant, les anthocyanes, les extraits de romarin, les extraits de feuilles d'olivier, le thé vert, le resvératrol et ses dérivés, le Pycnogénol, l'ergothinéine, la N acétylcystéine, la biotine, les chélatants, l'idébénone, des extraits végétaux corne le Pronalen Bioprotect TM de la société Provital, les antiradicalaires comme la vitamine E, le co enzyme Q10, les bioflavonoides, les SOD, le phytantriol, les lignanes, la mélatonine, les pidolates, le gluthation. Antioxidants that may be mentioned include polyphenols, tannic acid, epoigallocathechins and natural extracts containing them, anthocyanins, rosemary extracts, olive leaf extracts, green tea, resveratrol and its derivatives. derivatives, Pycnogenol, ergothineine, N acetylcysteine, biotin, chelants, idebenone, plant extracts Pronital Proniten Bioprotect TM from Provital, antiradicals such as vitamin E, coenzyme Q10, bioflavonoids , SOD, phytantriol, lignans, melatonin, pidolates, glutathione.
Selon un mode préféré de réalisation de l'invention, la composition utilisée selon l'invention contient au moins un filtre UV (ou filtre solaire) qui peut être un filtre chimique ou un filtre physique ou un mélange de tels filtres. A titre d'illustration et de façon non limitative, on peut citer les familles suivantes (les noms correspondent à la nomenclature CTFA des filtres) : les anthranilates, en particulier l'anthranilate de menthyle ; les benzophénones, en particulier la benzophénone-1 , la benzophénone-3, la benzophénone-5, la benzophénone-6, la benzophénone-8, la benzophénone-9, la benzophénone-12, et préférentiellement la Benzophénone-2 (Oxybenzone), ou la Benzophénone-4 (Uvinul MS40 disponible chez B.A.S.F.) ; les benzylidènes-camphres, en particulier le 3-benzylidène-camphre, l'acide benzylidènecampho-sulfonique, le benzalkoniumméthosulfate de Camphre, le polyacrylamidométhylbenzylidène camphre, l'acide téréphthalylidène di-camphre sulfonique , et préférentiellement le 4-méthylbenzylidène camphre (Eusolex 6300 disponible chez Merck) ; les benzimidazoles, en particulier le benzimidazilate (Neo Heliopan AP disponible chez Haarmann et Reimer), ou l'acide phénylbenzimidazole sulfonique (Eusolex 232 disponible chez Merck) ; les benzotriazoles, en particulier le drométrizole trisiloxane, ou le méthylène bisbenzotriazolyltétraméthylbutylphénol (Tinosorb M disponible chez Ciba) ; les cinnamates, en particulier le cinoxate, le DEA méthoxycinnamate, le méthylcinnamate de diisopropyle, le glycéryl éthylhexanoate de diméthoxycinnamate, le méthoxycinnamate d'isopropyle, le cinnamate d'isoamyle, et préférentiellement l'éthocrylène (Uvinul N35 disponible chez B.A.S.F.), l'octylméthoxycinnamate (Parsol MCX disponible chez Hoffmann La Roche), ou l'octocrylène (Uvinul 539 disponible chez B.A.S.F.) ; les dibenzoylméthanes, en particulier le butyl méthoxydibenzoylméthane (Parsol 1789) ; les imidazolines, en particulier l'éthylhexyl diméthoxybenzylidène dioxoimidazoline ; les PABA, en particulier l'éthyl Dihydroxypropyl PABA, l'éthylhexyldiméthyl PABA, le glycéryl PABA, le PABA, le PEG-25 PABA, et préférentiellement la diéthylhexylbutamido- triazone (Uvasorb HEB disponible chez 3V Sigma), l'éthyl hexyltriazone (Uvinul T150 disponible chez B.A.S.F.), ou l'éthyl PABA (benzocaïne) ; les salicylates, en particulier le salicyclate de dipropylèneglycol, le salicylate d'éthylhexyle, l'homosalate, ou le TEA salicylate ; les t azines, en particulier l'anisot azine (Tinosorb S disponible chez Ciba) ; le drométrizole trisiloxane, l'oxyde de zinc, le dioxyde de titane, l'oxyde de zinc, de fer, de zirconium, de cerium enrobés ou non. According to a preferred embodiment of the invention, the composition used according to the invention contains at least one UV filter (or sunscreen) which may be a chemical filter or a physical filter or a mixture of such filters. By way of illustration and in a nonlimiting manner, mention may be made of the following families (the names correspond to the CTFA nomenclature of the filters): anthranilates, in particular menthyl anthranilate; benzophenones, in particular benzophenone-1, benzophenone-3, benzophenone-5, benzophenone-6, benzophenone-8, benzophenone-9, benzophenone-12, and preferentially benzophenone-2 (Oxybenzone), or benzophenone-4 (Uvinul MS40 available from BASF); benzylidenecamphers, in particular 3-benzylidene camphor, benzylidenecamphosulfonic acid, camphor benzalkonium methosulphate, polyacrylamidomethylbenzylidene camphor, terephthalylidene di-camphorsulfonic acid, and preferentially 4-methylbenzylidene camphor (Eusolex 6300 available). at Merck); benzimidazoles, in particular benzimidazilate (Neo Heliopan AP available from Haarmann and Reimer), or phenylbenzimidazole sulfonic acid (Eusolex 232 available from Merck); benzotriazoles, in particular trisiloxane drometzol, or methylenebisbenzotriazolyltetramethylbutylphenol (Tinosorb M available from Ciba); cinnamates, in particular cinoxate, DEA methoxycinnamate, diisopropyl methylcinnamate, dimethoxycinnamate glyceryl ethylhexanoate, isopropyl methoxycinnamate, isoamyl cinnamate, and preferably ethocrylene (Uvinul N35 available from BASF), octyl methoxycinnamate (Parsol MCX available from Hoffmann La Roche), or octocrylene (Uvinul 539 available from BASF); dibenzoylmethanes, especially butyl methoxydibenzoylmethane (Parsol 1789); imidazolines, in particular ethylhexyl dimethoxybenzylidene dioxoimidazoline; PABAs, in particular ethyl dihydroxypropyl PABA, ethylhexyldimethyl PABA, glyceryl PABA, PABA, PEG-25 PABA, and preferentially diethylhexylbutamido triazone (Uvasorb HEB available from 3V Sigma), ethyl hexyltriazone (Uvinul T150 available from BASF), or ethyl PABA (benzocaine); salicylates, especially dipropylene glycol salicyclate, ethylhexyl salicylate, homosalate, or TEA salicylate; azines, in particular anisot azine (Tinosorb S available from Ciba); trisiloxane drometzol, zinc oxide, titanium dioxide, zinc oxide, iron oxide, zirconium oxide, cerium oxide coated or uncoated.
La quantité de filtres dépend de l'utilisation finale souhaitée. Elle peut aller par exemple de 1 à 30% en poids et mieux de 2 à 15% en poids par rapport au poids total de la composition. The amount of filters depends on the desired end use. It may range, for example, from 1 to 30% by weight and better still from 2 to 15% by weight relative to the total weight of the composition.
Bien entendu, l'homme du métier veillera à choisir ce ou ces éventuels ingrédients et/ou actifs complémentaires, et/ou leur quantité, de manière telle que les propriétés avantageuses de l'association d'au moins une bactéhocine et d'au moins un prébiotique selon l'invention ne soient pas, ou substantiellement pas, altérées par l'adjonction envisagée. Les compositions selon l'invention pourront être appliquées directement sur la peau ou, de façon alternative, sur des supports cosmétiques de type occlusif ou non occlusif, destinés à être appliqués de façon localisée sur la peau. A titre d'exemples de supports cosmétiques, non limitatifs, on peut notamment citer un patch, une lingette, un roll-on et un stylo. Of course, those skilled in the art will take care to choose this or these optional ingredients and / or additional active ingredients, and / or their quantity, in such a way that the properties Advantageous of the combination of at least one bacteriocin and at least one prebiotic according to the invention are not, or not substantially, impaired by the addition envisaged. The compositions according to the invention may be applied directly to the skin or, alternatively, to occlusive or non-occlusive type cosmetic supports intended to be applied in a localized manner to the skin. As examples of non-limiting cosmetic media, there may be mentioned a patch, a wipe, a roll-on and a pen.
Le support peut être un support 'occlusif. A titre d'exemple, le support est constitué d'un matériau thermoplastique, choisi parmi les polyéthylènes haute et basse densité, les polypropylènes, les polychlorures de vinyle, les copolymères d'éthylène et d'acétate de vinyle, les polyesters et les polyuréthanes, ou d'un complexe de tels matériaux. Ces matériaux peuvent également être présents sous forme stratifiée avec au moins une feuille de métal telle qu'une feuille d'aluminium. The support may be an occlusive support. By way of example, the support consists of a thermoplastic material chosen from high and low density polyethylenes, polypropylenes, polyvinyl chlorides, copolymers of ethylene and vinyl acetate, polyesters and polyurethanes. , or a complex of such materials. These materials may also be present in laminated form with at least one sheet of metal such as aluminum foil.
La couche support peut être de toute épaisseur appropriée qui procurera les fonctions de support et de protection souhaitées. De préférence, l'épaisseur de la couche support est comprise entre environ 20 pm et environ 1 ,5 mm. Avantageusement, la couche support est suffisamment flexible de manière à pouvoir épouser parfaitement le profil de la peau, et à ne pas provoquer chez l'utilisateur, une sensation d'inconfort. De préférence toutefois, le support est 'non occlusif. Dans cette dernière hypothèse, on utilise avantageusement un support constitué d'un papier, d'un matériau thermoplastique poreux ou perforé, d'un tissé, d'un non tissé, d'un non tissé perforé. The support layer may be of any suitable thickness which will provide the desired support and protection functions. Preferably, the thickness of the support layer is from about 20 μm to about 1.5 mm. Advantageously, the support layer is sufficiently flexible so as to perfectly fit the profile of the skin, and not to cause the user, a feeling of discomfort. Preferably, however, the carrier is non-occlusive. In the latter case, it is advantageous to use a support consisting of a paper, a porous or perforated thermoplastic material, a woven fabric, a nonwoven fabric or a perforated nonwoven fabric.
Selon un autre mode de réalisation de l'invention, lesdites compositions selon l'invention peuvent être associées à des compositions administrées par voie orale, contenant des actifs cosmétiques à effet bénéfique sur l'aspect de la peau. L'invention concerne également une composition telle que décrite ci-avant, pour le soin et/ou de nettoyage des peaux sensibles, irritées, ou présentant de l'eczéma ou une dermatite atopique. En particulier, elle vise une composition telle que décrite ci- avant pour prévenir et/ou à traiter l'irritation de la peau, l'eczéma, ou encore la dermatite atopique. According to another embodiment of the invention, said compositions according to the invention may be combined with orally administered compositions containing cosmetic active agents with a beneficial effect on the appearance of the skin. The invention also relates to a composition as described above, for the care and / or cleaning of sensitive skin, irritated, or having eczema or atopic dermatitis. In particular, it aims at a composition as described above. before to prevent and / or treat skin irritation, eczema, or atopic dermatitis.
Elle concerne également un procédé cosmétique pour le soin et/ou le nettoyage des peaux sensibles, irritées, ou présentant de l'eczéma ou une dermatite atopique. It also relates to a cosmetic process for the care and / or the cleaning of sensitive skin, irritated, or presenting with eczema or atopic dermatitis.
Elle concerne encore un procédé cosmétique de soin de la peau, mettant en œuvre une composition telle que définie ci-avant, caractérisé en ce qu'il vise à atténuer les imperfections cutanées, notamment les imperfections cutanées des peaux grasses. It also relates to a cosmetic skin care process, implementing a composition as defined above, characterized in that it aims to reduce skin imperfections, including skin imperfections of oily skin.
On entend par « imperfections cutanées des peaux grasses », notamment, la brillance, la luisance de la peau, la dilation des pores de la peau, un grain de peau épais, l'ensemble pouvant conduire notamment à des comédons Elle concerne encore un procédé cosmétique de soin de la peau grasse, en particulier en vue de matifier la peau, comprenant l'application topique sur celle-ci, d'une composition elle que défini ci-avant. The term "skin imperfections of oily skin", in particular, the shine, the shine of the skin, the expansion of the pores of the skin, a thick skin texture, all of which may lead in particular to comedones. cosmetic skin care product, particularly for the purpose of mattifying the skin, comprising the topical application thereof, of a composition as defined above.
La composition selon l'invention peut notamment être utilisée pour corriger ou prévenir les lésions et/ou imperfections de la peau rencontrées chez un sujet acnéique ou dont la peau est à tendance acnéique, notamment un sujet à peau grasse à tendance acnéique. Dans ce cadre, la composition selon l'invention comprendra de préférence la plantaricine 149 en tant que bactériocine et/ou un fructo-oligosaccharide en tant que prébiotique. The composition according to the invention may in particular be used to correct or prevent lesions and / or skin imperfections encountered in an acne or acne-prone skin, especially a subject with oily acne-prone skin. In this context, the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructo-oligosaccharide as a prebiotic.
L'invention concerne donc une composition telle que décrite ci-dessus, pour atténuer les imperfections des peaux grasses. Un aspect de l'invention vise plus particulièrement l'utilisation d'une telle composition pour le soin et/ou le nettoyage de la peau, notamment de la peau grasse, et plus spécifiquement de la peau grasse à tendance acnéique. Dans ce cadre, la composition selon l'invention comprendra de préférence la plantaricine 149 en tant que bactériocine et/ou un fructo- oligosaccharide en tant que prébiotique. L'invention concerne également une composition comprenant l'association telle que décrite ci-dessus, pour le soin et/ou le nettoyage des peaux acnéiques ou à tendance acnéique, notamment des peaux grasses à tendance acnéique. Dans ce cadre, la composition selon l'invention comprendra de préférence la plantaricine 149 en tant que bactériocine et/ou un fructo-oligosaccharide en tant que prébiotique. The invention therefore relates to a composition as described above, to reduce imperfections of oily skin. An aspect of the invention is more particularly the use of such a composition for the care and / or cleansing of the skin, especially oily skin, and more specifically for oily acne-prone skin. In this context, the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructooligosaccharide as a prebiotic. The invention also relates to a composition comprising the combination as described above, for the care and / or the cleaning of acne-prone or acne-prone skin, in particular fatty skin with an acne-prone tendency. In this context, the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructo-oligosaccharide as a prebiotic.
L'invention concerne encore un procédé cosmétique pour le soin de la peau grasse, comprenant l'application sur la peau d'une composition comprenant l'association telle que définie ci-dessus. Dans ce cadre, la composition selon l'invention comprendra de préférence la plantaricine 149 en tant que bactériocine et/ou un fructo- oligosaccharide en tant que prébiotique. The invention also relates to a cosmetic process for the care of oily skin, comprising the application to the skin of a composition comprising the combination as defined above. In this context, the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructooligosaccharide as a prebiotic.
L'invention concerne également un procédé pour le soin et/ou le nettoyage de la peau grasse, comprenant l'application sur la peau d'une composition comprenant l'association d'une bactériocine et d'un prébiotique telle que définie ci-dessus. Dans ce cadre, la composition selon l'invention comprendra de préférence la plantaricine 149 en tant que bactériocine et/ou un fructo-oligosaccharide en tant que prébiotique. The invention also relates to a method for the care and / or cleaning of oily skin, comprising the application to the skin of a composition comprising the combination of a bacteriocin and a prebiotic as defined above . In this context, the composition according to the invention will preferably comprise plantaricin 149 as a bacteriocin and / or a fructo-oligosaccharide as a prebiotic.
Selon un mode particulier de réalisation, la composition décrite ci-dessus est appliquée sur des zones du visage ou du front présentant une brillance de la peau. According to a particular embodiment, the composition described above is applied to areas of the face or forehead with a gloss of the skin.
Selon un autre mode particulier de réalisation, la composition selon l'invention est appliquée sur la peau de personnes dont la peau est de phototype III à VI. L'invention concerne en outre une composition telle que décrite ci-dessus, notammment en tant que composition dermatologique, destinée à traiter à titre préventif et/ou curatif l'acné et/ou l'hyperséborrhée. L'invention concerne donc également l'utilisation de l'association d'une bactériocine et d'un prébiotique pour la préparation d'une composition destinée à traiter à titre préventif et/ou curatif l'acné et/ou une hyperséborrhée, et/ou destinée à corriger les désordres associés à l'acné et/ou à une hyperséborrhée. L'invention concerne par ailleurs une plantaricine de classe II, en particulier la plantaricine 149, pour une utilisation dans le traitement préventif et/ou curatif des désordres cutanés liés à P. acnés, en particulier l'acné. L'invention concerne également une composition cosmétique et/ou dermatologique, comprenant dans un milieu physiologiquement acceptable, la plantaricine 149. According to another particular embodiment, the composition according to the invention is applied to the skin of persons whose skin is of phototype III to VI. The invention furthermore relates to a composition as described above, in particular as a dermatological composition, intended for the preventive and / or curative treatment of acne and / or hyperseborrhoea. The invention therefore also relates to the use of the combination of a bacteriocin and a prebiotic for the preparation of a composition intended for the preventive and / or curative treatment of acne and / or hyperseborrhoea, and or intended to correct disorders associated with acne and / or hyperseborrhea. The invention furthermore relates to a plantaricin class II, in particular plantaricin 149, for use in the preventive and / or curative treatment of skin disorders related to P. acnes, in particular acne. The invention also relates to a cosmetic and / or dermatological composition comprising, in a physiologically acceptable medium, plantaricin 149.
L'inverntion concerne en outre une composition, en particulier dermatologique, comprenant dans un milieu physiologiquement acceptable, une plantaricine de classe II, en particulier le peptide plantaricine 149, pour une utilisation dans le traitement à titre préventif et/ou curatif des désordres cutanés liés à P. acnés, en particulier dans le traitement l'acné. The invention also relates to a composition, in particular a dermatological composition, comprising, in a physiologically acceptable medium, a class II plantaricin, in particular the plantaricin peptide 149, for use in the preventive and / or curative treatment of skin-related disorders. P. acnes, especially in the treatment of acne.
Selon un mode particulier de réalisation, la composition pour une utilisation dans le traitement préventif ou curatif des désordres liés à P. acnés (en particulier l'acné) est caractérisée en ce qu'elle comprend en outre un actif additionnel pour le soin des peaux grasses choisi parmi les agents desquamants, les agents antimicrobiens, les agents apaisants, les agents anti-inflammatoires, les agents sébo-régulateurs, les agents antioxydants, les agents cicatrisants, les agents astringents, et leurs mélanges. According to a particular embodiment, the composition for use in the preventive or curative treatment of disorders related to P. acnes (in particular acne) is characterized in that it further comprises an additional asset for the care of the skin. fatty oils selected from desquamating agents, antimicrobial agents, soothing agents, anti-inflammatory agents, sebo-regulating agents, antioxidants, healing agents, astringent agents, and mixtures thereof.
Selon un autre aspect, l'invention concerne un procédé cosmétique de soin et/ou de nettoyage de la peau, en particulier de la peau grasse, comprenant l'application topique sur ladite peau, d'une composition cosmétique comprenant dans un milieu physiologiquement acceptable, une plantaricine de classe II, en particulier la plantaricine 149. According to another aspect, the invention relates to a cosmetic process for the care and / or cleaning of the skin, in particular of oily skin, comprising the topical application to said skin of a cosmetic composition comprising a physiologically acceptable medium. , a class II plantaricin, in particular plantaricin 149.
Avantageusement, le procédé cosmétique est destiné à réduire et/ou éliminer les imperfections cutanées liées à P. acnés en particulier choisies parmi un relief irrégulier, des pores dilatés, les comédons et/ou points noirs. Advantageously, the cosmetic process is intended to reduce and / or eliminate cutaneous imperfections related to P. acnes, in particular chosen from an irregular relief, dilated pores, comedones and / or blackheads.
Selon un autre aspect, la composition selon l'invention est utilisée à titre d'actif antibactérien, pour diminuer la quantité de germes responsables de l'acné présents sur la peau, ou pour limiter leur développement, et notamment pour diminuer la quantité de P. /Acnés et/ou P. Granulosum, ou pour limiter le développement de l'un de ces germes. Elle peut notamment se présenter sous la forme d'une crème ou d'un gel anti-acné. In another aspect, the composition according to the invention is used as an antibacterial active agent, to reduce the amount of acne-causing germs present on the skin. the skin, or to limit their development, and in particular to reduce the amount of P. / Acnes and / or P. Granulosum, or to limit the development of one of these germs. It may especially be in the form of a cream or an anti-acne gel.
On entend désigner par le terme "traitement", et toutes ses déclinaisons, le traitement curatif ou préventif. La composition dermatologique selon l'invention peut ainsi être utilisée chez un sujet présentant de l'acné ou une hyperséborrhée. La composition dermatologique selon l'invention peut aussi être utilisée pour retarder ou ralentir la progression ou prévenir une progression plus en avant de l'acné, améliorant l'aspect esthétique de la peau de l'utilisateur. La composition dermatologique de la présente invention peut enfin être administrée à une personne ne présentant pas d'acné, mais dont la peau, notamment une peau grasse, est à tendance acnéique. What is meant by the term "treatment", and all its variations, curative or preventive treatment. The dermatological composition according to the invention can thus be used in a subject having acne or hyperseborrhoea. The dermatological composition according to the invention can also be used for delaying or slowing the progression or preventing a further progression of acne, improving the aesthetic appearance of the skin of the user. The dermatological composition of the present invention may finally be administered to a person who does not have acne, but whose skin, especially oily skin, is acne-prone.
La quantité de l'association d'au moins une bactériocine et d'au moins un prébiotique dans la composition dermatologique selon l'invention sera avantageusement comprise dans les intervalles décrits précédemment, pour obtenir l'effet désiré. Bien entendu, l'homme du métier est à même d'établir la quantité efficace de ladite association dans la composition dermatologique selon l'invention, et veillera à ce que les ingrédients et/ou actifs additionnels introduits dans ladite composition n'altère pas les effets avantageux de l'association. The amount of the combination of at least one bacteriocin and at least one prebiotic in the dermatological composition according to the invention will advantageously be in the ranges described above, to obtain the desired effect. Of course, those skilled in the art are able to establish the effective amount of said combination in the dermatological composition according to the invention, and will ensure that the additional ingredients and / or active ingredients introduced into said composition do not affect the advantageous effects of the association.
Selon un autre aspect, l'invention concerne une méthode de traitement dermatologique des troubles de la peau indiqués ci-dessus, comprenant l'administration topique d'une composition comprenant au moins l'association selon l'invention, telle que décrite ci-dessus, à une personne en ayant besoin. According to another aspect, the invention relates to a method of dermatological treatment of the disorders of the skin indicated above, comprising the topical administration of a composition comprising at least the combination according to the invention, as described above. , to a person in need.
La composition peut être appliquée selon toutes les façons envisageables par l'homme du métier. En particulier, il peut être envisagé d'appliquer la composition directement sur la peau ou de la diluer au préalable dans un solvant adéquat. Selon un mode particulier, lesdits procédés ou lesdites utilisations selon l'invention comprennent en outre une étape de rinçage après application sur la peau de la composition. Ainsi, après application sur la peau, la composition peut être rincée, après un temps de pause, compris avantageusement entre 5 et 30 minutes. The composition can be applied in any way that can be envisaged by those skilled in the art. In particular, it may be envisaged to apply the composition directly to the skin or to dilute it beforehand in a suitable solvent. According to a particular embodiment, said methods or said uses according to the invention further comprise a rinsing step after application to the skin of the composition. Thus, after application to the skin, the composition can be rinsed after a pause, advantageously between 5 and 30 minutes.
* Composition pour le traitement des désordres desquamatifs des cheveux ou du cuir chevelu * Composition for the treatment of desquamative disorders of the hair or scalp
L'invention concerne donc également une composition comprenant l'association telle que définie ci-avant, destinée au traitement des désordres desquamatifs des cheveux et/ou du cuir chevelu. The invention therefore also relates to a composition comprising the combination as defined above, intended for the treatment of desquamative disorders of the hair and / or the scalp.
Selon l'invention, les états desquamatifs des cheveux et/ou du cuir chevelu sont par exemple les pellicules, la dermite séborrhéique. De préférence, les dits états desquamatifs du cuir chevelu sont ceux induits par la levure du genre Malassezia spp., et plus particulièrement M. furfur. According to the invention, the desquamative states of the hair and / or the scalp are, for example, dandruff, seborrheic dermatitis. Preferably, said desquamative states of the scalp are those induced by the yeast of the genus Malassezia spp., And more particularly M. furfur.
Les compositions pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu selon l'invention peuvent être destinées à une application cosmétique ou pharmaceutique, particulièrement dermatologique. De préférence les compositions selon l'invention sont destinées à une application cosmétique. The compositions for the treatment of the desquamative states of the hair and / or the scalp according to the invention may be intended for a cosmetic or pharmaceutical application, particularly dermatological. Preferably the compositions according to the invention are intended for a cosmetic application.
Les compositions selon l'invention sont généralement appliquées sur le cuir chevelu ou les cheveux. The compositions according to the invention are generally applied to the scalp or the hair.
Selon le mode d'administration, la composition de l'invention peut se présenter sous toutes les formes galéniques normalement utilisées, particulièrement en cosmétologie. Une composition pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu préférée de l'invention est une composition cosmétique destinée à une application topique. La composition pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu suivant l'invention, après application sur les cheveux et cuir chevelu humains peut être rincée ou non rincée à l'eau ou par un shampooing. Elle peut se présenter sous toute forme classiquement utilisée dans le domaine concerné et par exemple sous forme de solution aqueuse ou huileuse ou de dispersion du type lotion ou sérum, d'émulsions de consistance liquide ou semi-liquide du type lait, obtenues par dispersion d'une phase grasse dans une phase aqueuse (H/E) ou inversement (E/H), ou de suspensions ou émulsions de consistance molle du type crème ou gel aqueux ou anhydres, ou encore de microcapsules ou microparticules, ou de dispersions vésiculaires de type ionique et/ou non ionique. Ces compositions sont préparées selon les méthodes usuelles. Depending on the mode of administration, the composition of the invention may be in any galenical form normally used, particularly in cosmetology. A composition for treating the desquamative conditions of the hair and / or the preferred scalp of the invention is a cosmetic composition intended for topical application. The composition for the treatment of the desquamative states of the hair and / or scalp according to the invention, after application to human hair and scalp can be rinsed or not rinsed with water or with a shampoo. It can be in any form conventionally used in the field concerned and for example in the form of an aqueous or oily solution or of the lotion or serum type dispersion, emulsions of liquid or semi-liquid consistency of the milk type, obtained by dispersion of a fatty phase in an aqueous phase (O / W) or conversely ( E / H), or suspensions or emulsions of soft consistency of the cream or gel type aqueous or anhydrous, or microcapsules or microparticles, or vesicular dispersions of ionic and / or nonionic type. These compositions are prepared according to the usual methods.
La composition utilisable selon l'invention peut aussi être une composition pour soins capillaires, et notamment un shampooing, une lotion de mise en plis, une lotion traitante, une crème ou un gel coiffant, une composition de teintures (notamment teintures d'oxydation) éventuellement sous forme de shampooings colorants, des lotions restructurantes pour les cheveux, une composition de permanente (notamment une composition pour le premier temps d'une permanente), une préparation (lotion, gel, shampooing) antichute des cheveux, un shampooing antiparasitaire, etc. The composition that may be used according to the invention may also be a composition for hair care, and in particular a shampoo, a setting lotion, a treatment lotion, a cream or a styling gel, a dye composition (especially oxidation dyes). optionally in the form of coloring shampoos, restructuring lotions for the hair, a permanent composition (in particular a composition for the first time of a perm), a preparation (lotion, gel, shampoo), anti-hair loss, a pest control shampoo, etc. .
Les quantités des différents constituants des compositions pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu utilisables selon l'invention sont celles classiquement utilisées dans les domaines considérés. The amounts of the various constituents of the compositions for treating the desquamative states of the hair and / or scalp that can be used according to the invention are those conventionally used in the fields under consideration.
Les compositions pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu utilisables selon l'invention peuvent également consister en des préparations solides constituant des savons ou des pains de nettoyage. The compositions for the treatment of the desquamative states of the hair and / or the scalp which can be used according to the invention may also consist of solid preparations constituting soaps or cleaning bars.
Les compositions pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu utilisables selon l'invention peuvent aussi être conditionnées sous forme d'une composition pour aérosol comprenant également un agent propulseur sous pression. The compositions for the treatment of the desquamative states of the hair and / or the scalp which can be used according to the invention can also be packaged in the form of an aerosol composition also comprising a propellant under pressure.
Les compositions pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu selon l'invention contiennent un milieu physiologiquement acceptable. En particulier, ce milieu contient une phase aqueuse contenant de l'eau et éventuellement au moins un solvant organique miscible à l'eau comme les monoalcools en C2 à C6 tels que l'éthanol, l'isopropanol, le n-butanol, les polyols comme le propylèneglycol, le glycérol ou les éthers de glycol. Ce milieu peut contenir une phase huileuse contenant un ou plusieurs corps gras liquides à température ambiante (25°C) et pression atmosphérique, non miscibles à l'eau, appelées "huiles". The compositions for the treatment of the desquamative states of the hair and / or scalp according to the invention contain a physiologically acceptable medium. In particular, this medium contains an aqueous phase containing water and optionally at least one water-miscible organic solvent such as C2-C6 monoalcohols such as ethanol, isopropanol, n-butanol, polyols such as propylene glycol, glycerol or glycol ethers. This medium may contain an oily phase containing one or more liquid fats which are liquid at room temperature (25 ° C.) and atmospheric pressure, immiscible with water, called "oils".
Le milieu physiologiquement acceptable peut, en outre, comprendre au moins un tensioactif choisi parmi les tensioactifs non-ioniques, les tensioactifs anioniques, les tensioactifs cationiques, les tensioactifs amphotères, et leurs mélanges. The physiologically acceptable medium may, in addition, comprise at least one surfactant chosen from nonionic surfactants, anionic surfactants, cationic surfactants, amphoteric surfactants, and mixtures thereof.
Les compositions selon l'invention peuvent, en outre, contenir comme base lavante au moins un agent tensioactif choisi parmi les agents tensioactifs anioniques, non- ioniques, amphotères et leurs mélanges. The compositions according to the invention may further contain, as washing base, at least one surfactant chosen from anionic, nonionic and amphoteric surfactants and mixtures thereof.
Comme agent tensioactif anionique utilisable dans la présente invention, on peut notamment mentionner les sels, en particulier les sels de métaux alcalins tels que les sels de sodium, les sels d'ammonium, les sels d'amines, les sels d'aminoalcools ou les sels de métaux alcalino-terreux, par exemple, de magnésium, des types suivants : les alkylsulfates, les alkyléthersulfates, les alkylamidoéthersulfates, les alkylaryl- polyéthersulfates, les monoglycéride- sulfates ; les alkylsulfonates, les alkylamidesulfonates, les alkylaryl-sulfonates, les cc-oléfine-sulfonates, les paraffine- sulfonates, les alkylsulfosuccinates, les alkyléthersulfosuccinates, les alkylamide- sulfosuccinates, les alkylsulfo-acétates, les acylsarcosinates et les acylglutamates, les groupes alkyle et acyle de tous ces composés comportant de 6 à 24 atomes de carbone et le groupe aryle désignant de préférence un groupe phényle ou benzyle, leurs mélanges. As anionic surfactant usable in the present invention, mention may especially be made of salts, in particular alkali metal salts such as sodium salts, ammonium salts, amine salts, aminoalcohol salts or salts thereof. alkaline earth metal salts, for example of magnesium, of the following types: alkyl sulphates, alkyl ether sulphates, alkyl amido ether sulphates, alkylaryl polyethersulphates, monoglyceride sulphates; alkylsulfonates, alkylamidesulfonates, alkylarylsulfonates, α-olefinsulfonates, paraffin sulfonates, alkylsulfosuccinates, alkylethersulfosuccinates, alkylamidesulfosuccinates, alkylsulphoacetates, acylsarcosinates and acylglutamates, alkyl and acyl groups; of all these compounds having from 6 to 24 carbon atoms and the aryl group preferably denoting a phenyl or benzyl group, their mixtures.
On peut également utiliser comme agent tensioactif anionique, les monoesters d'alkyle en C6-C24 et d'acides polyglycoside-dicarboxyliques tels que les glucoside- citrates d'alkyle, les polyglycoside-tartrates d'alkyle et les polyglycoside- sulfosuccinates d'alkyle, les alkylsulfosuccinamat.es, les acyliséthionates et les N- acyltaurates, le groupe alkyle ou acyle de tous ces composés comportant de 12 à 20 atomes de carbone, leurs mélanges. Un autre groupe d'agents tensioactifs anioniques utilisable dans la composition de la présente invention est celui des acyl-lactylates dont le groupe acyle comporte de 8 à 20 atomes de carbone. C 6 -C 2 4 alkyl monoesters and polyglycoside dicarboxylic acids such as alkyl glucoside citrates, alkyl polyglycoside tartrates and polyglycoside sulfosuccinates may also be used as anionic surfactants. alkyl, alkylsulfosuccinamates, acylisethionates and N-acyltaurates, the alkyl or acyl group of all these compounds having from 12 to 20 carbon atoms, and mixtures thereof. Another group of anionic surfactants that can be used in the composition of the present invention is that of acyl lactylates, the acyl group of which contains from 8 to 20 carbon atoms.
En outre, on peut encore citer les acides alkyl-D-galactoside-uroniques et leurs sels ainsi que les acides alkyl(C6-C24)éther-carboxyliques polyoxyalkylénés, les acides alkyl(C6-C24)aryl(C6-C24)éther-carboxyliques poly-oxyalkylénés, les acides alkyl(C6- C24)amidoéther-carboxyliques poly-oxyalkylénés et leurs sels, en particulier ceux comportant de 2 à 50 motifs oxyde d'éthylène, et leurs mélanges. Furthermore, mention may also be made of alkyl-D-galactoside-uronic acids and their salts, polyoxyalkylenated (C6-C2 4) ether carboxylic acids, polyoxyalkylenated (C6-C2 4) aryl (C6-C2 4 poly (oxyalkylenated) ether carboxylic acids, poly (oxyalkylenated) (C 6 -C 2 4 ) alkyl amidoether carboxylic acids and their salts, in particular those containing from 2 to 50 ethylene oxide units, and mixtures thereof.
On utilise de préférence comme agent tensioactif anionique, les alkylsulfates, les alkyléthersulfates et les alkyléthercarboxylates, leurs mélanges, en particulier sous forme de sels de métaux alcalins ou alcalino-terreux, d'ammonium, d'amine ou d'aminoalcool. Anionic surfactants are preferably alkyl sulphates, alkyl ether sulphates and alkyl ether carboxylates, and mixtures thereof, in particular in the form of alkali metal or alkaline earth metal salts, ammonium, amine or aminoalcohol.
Comme agent tensioactif amphotère utilisable dans la présente invention, on peut citer les dérivés d'amines aliphatiques secondaires ou tertiaires, dans lesquels le groupe aliphatique est une chaîne linéaire ou ramifiée comportant de 8 à 22 atomes de carbone et contenant au moins un groupe anionique tel que, par exemple, un groupe carboxylate, sulfonate, sulfate, phosphate ou phosphonate. On peut citer en particulier les alkyl(C8-C2o)bétaïnes, les sulfobétaïnes, les alkyl(C8-2o)amidoalkyl(C6- CsJ-bétaïnes, les alkyl(C8-C2o)amidoalkyl(C6-C8)sulfo-bétaïnes et leurs mélanges. Parmi les dérivés d'amines, on peut citer les produits commercialisés sous la dénomination MIRANOL®, tels que décrits dans les brevets US 2 528 378 et US 2 781 354 et classés dans le dictionnaire CTFA, 3ème édition, 1982, sous les dénominations Amphocarboxy-glycinate et Amphocarboxypropionate de structures respectives (2) et (3) : As amphoteric surfactant that may be used in the present invention, mention may be made of derivatives of secondary or tertiary aliphatic amines, in which the aliphatic group is a linear or branched chain containing from 8 to 22 carbon atoms and containing at least one anionic group such as for example, a carboxylate, sulfonate, sulfate, phosphate or phosphonate group. Mention may in particular be made of (C 8 -C 20) alkyl betaines, sulphobetaines, (C 8 -C 20) alkylamido (C 6 -C 8) alkyl betaines, (C 8 -C 20) alkylamido (C 6 -C 8) alkyl sulphobetaines and their Among the amine derivatives, mention may be made of the products sold under the name MIRANOL®, as described in patents US 2 528 378 and US 2 781 354 and classified in the CTFA dictionary, 3rd edition, 1982, under the terms Amphocarboxy-glycinate and Amphocarboxypropionate denominations of respective structures (2) and (3):
Ri-CONHCH2CH2-N(R2)(R3)(CH2COO-) (2) R-CONHCH 2 CH 2 -N (R 2) (R 3) (CH 2 COO) (2)
dans laquelle : in which :
.Ri représente un groupe alkyle dérivé d'un acide Ra-COOH présent dans l'huile de coprah hydrolysée, un groupe heptyle, nonyle ou undécyle, . R2 représente un groupe bêta-hydroxyéthyle, et Ri represents an alkyl group derived from an acid Ra-COOH present in the hydrolysed coconut oil, a heptyl, nonyl or undecyl group, . R2 represents a beta-hydroxyethyl group, and
. R3 représente un groupe carboxyméthyle ; et . R3 represents a carboxymethyl group; and
Ri'-CONHCH2CH2-N(B)(C) (3) Ri'-CONHCH 2 CH 2 -N (B) (C) (3)
dans laquelle : in which :
. B représente -CH2CH2OX\ . B represents -CH 2 CH 2 OX \
. C représente -(CH2)Z-Y\ avec z = 1 ou 2, . C represents - (CH 2 ) Z -Y \ with z = 1 or 2,
. X' représente le groupe -CH2CH2-COOH ou un atome d'hydrogène, . X 'represents the group -CH 2 CH 2 -COOH or a hydrogen atom,
. Y' représente -COOH ou le groupe -CH2-CHOH-SO3H, . Y 'represents -COOH or the group -CH 2 -CHOH-SO 3 H,
. Ri' représente un groupe alkyle d'un acide Ri'-COOH présent dans l'huile de coprah ou dans l'huile de lin hydrolysée, un groupe alkyle, notamment en C17 et sa forme iso, un groupe en C17 insaturé. . R 1 'represents an alkyl group of an R 1 -COOH acid present in coconut oil or in hydrolysed linseed oil, an alkyl group, especially C 17 and its iso form, an unsaturated C 17 group.
Ces composés sont classés dans le dictionnaire CTFA, 5ème édition, 1993, sous les dénominations coco-amphodiacétate de disodium, lauro-amphodiacétate de disodium, caprylamphodiacétate de disodium, caprylo-amphodiacétate de disodium, coco-ampho-dipropionate de disodium, lauro-ampho-dipropionate de disodium, capryl-ampho-dipropionate de disodium, caprylo-ampho-dipropionate de disodium, acide lauro-ampho-dipropionique, acide coco-ampho-dipropionique. A titre d'exemple, on peut citer le cocoamphodiacétate commercialisé par la société RHODIA sous la dénomination commerciale MIRANOL® C2M concentré. These compounds are classified in the CTFA dictionary, 5th edition, 1993, under the names coco-amphodiacetate disodium, lauro-amphodiacetate disodium, caprylamphodiacetate disodium, caprylo-amphodiacetate disodium, coco-ampho-dipropionate disodium, lauro-ampho disodium dipropionate, disodium capryl-ampho-dipropionate, disodium caprylo-amphodipropionate, lauro-amphodipropionic acid, cocoamino-dipropionic acid. By way of example, mention may be made of cocoamphodiacetate marketed by Rhodia under the trade name MIRANOL® C2M concentrate.
Parmi les agents tensioactifs amphotères, on utilise de préférence les (alkyle en C8- 20)-bétaïnes, les (alkyle en C8-C2o)-amido(alkyle en C6-C8)bétaïnes, les alkylamphodiacétates et leurs mélanges. Of the amphoteric surfactants, C 8 -C 20 alkylbetaines, C 8 -C 20 alkylamido (C 6 -C 8 ) alkylbetaines, alkylamphodiacetates and mixtures thereof are preferably used.
Comme agent tensioactif non-ionique utilisable dans la composition selon l'invention, on peut citer les composés connus décrits notamment dans le livre "Handbook of Surfactants" par M.R. PORTER, éditions Blackie & Son (Glasgow and London), 1991 , pp 1 16-178). Il est choisi notamment parmi les alcools, les alpha-diols, les alkyl(C1 -20)phénols ou les acides gras polyéthoxylés, polypropoxylés ou polyglycérolés, ayant une chaîne grasse comportant, par exemple, de 8 à 18 atomes de carbone, le nombre de groupements oxyde d'éthylène ou oxyde de propylène pouvant aller notamment de 2 à 50 et le nombre de groupements glycérol pouvant aller notamment de 2 à 30, leurs mélanges. As nonionic surfactant that can be used in the composition according to the invention, mention may be made of the known compounds described in particular in the book "Handbook of Surfactants" by MR PORTER, published by Blackie & Son (Glasgow and London), 1991, pp. -178). It is chosen in particular from alcohols, alpha-diols, alkyl (C1 -20) phenols or polyethoxylated, polypropoxylated or polyglycerolated fatty acids, having a fatty chain comprising, for example, from 8 to 18 carbon atoms, the number ethylene oxide or propylene oxide groups which can range from 2 to 50 and the number of glycerol groups that can range from 2 to 30, their mixtures.
On peut également citer comme agent tensioactif non-ionique utilisable dans l'invention les condensais d'oxyde d'éthylène et d'oxyde de propylène sur des alcools gras ; les amides gras polyéthoxylés ayant de préférence de 2 à 30 motifs d'oxyde d'éthylène ; les amides gras polyglycérolés comportant en moyenne de 1 à 5 groupements glycérol et en particulier de 1 ,5 à 4 ; les esters d'acides gras du sorbitane éthoxylés ayant de 2 à 30 motifs d'oxyde d'éthylène ; les esters d'acides gras du saccharose ; les esters d'acides gras de polyéthylèneglycol ; les (alkyle en C6-C24)polyglycosides ; les dérivés de N-(alkyle en C6-24)glucamine ; les oxydes d'amines tels que les oxydes d'(alkyle en Cio-Ci4)amines ou les oxydes de N-(acyle en Cio-Ci4)-aminopropylmorpholine ; leurs mélanges. Parmi les tensioactifs non-ioniques cités ci-dessus, on utilise de préférence les (alkyle en C6-C24)polyglycosides. Mention may also be made, as nonionic surfactant, which can be used in the invention, of ethylene oxide and propylene oxide condensates on fatty alcohols; polyethoxylated fatty amides preferably having from 2 to 30 ethylene oxide units; polyglycerolated fatty amides comprising on average from 1 to 5 glycerol groups and in particular from 1, 5 to 4; ethoxylated sorbitan fatty acid esters having 2 to 30 ethylene oxide units; fatty acid esters of sucrose; polyethylene glycol fatty acid esters; the (C6 to C2 4) alkylpolyglycosides; N- (C6-24 alkyl) glucamine derivatives; amine oxides such as (C 1 -C 4 alkyl) amine oxides or N- (C 10 -C 4 acyl) aminopropylmorpholine oxides; their mixtures. Among the nonionic surfactants mentioned above, are preferably used the (C6 to C2 4) polyglycosides.
Selon un mode de réalisation particulier de l'invention, la base lavante contient au moins un tensioactif anionique et au moins un tensioactif amphotère ou non-ionique. According to a particular embodiment of the invention, the washing base contains at least one anionic surfactant and at least one amphoteric or nonionic surfactant.
La quantité totale d'agent tensioactif est généralement comprise dans la gamme allant de 0,01 % à 50% en poids, de préférence de 0,1 % à 25 % en poids par rapport au poids total de la composition. En particulier, lorsque la composition pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu de l'invention se présente sous forme d'un shampooing, la quantité totale d'agents tensioactifs (ou base lavante) est en particulier choisie de 4 % à 50 % en poids, par exemple de 8 % à 25 % en poids, rapportée au poids total de la composition. The total amount of surfactant is generally in the range from 0.01% to 50% by weight, preferably from 0.1% to 25% by weight relative to the total weight of the composition. In particular, when the composition for treating the desquamative states of the hair and / or scalp of the invention is in the form of a shampoo, the total amount of surfactants (or washing base) is in particular chosen from 4% to 50% by weight, for example 8% to 25% by weight, based on the total weight of the composition.
La composition pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu selon l'invention peut, en outre, contenir au moins un ingrédient additionnel utilisé classiquement dans les domaines considérés et choisi parmi des principes actifs cosmétiques ayant un effet bénéfique sur les cheveux et/ou le cuir chevelu comme les sels de zinc d'acide organique (acétate, glycolate, lactate, gluconate ou citrate) ou minéral (chlorure et le sulfate), les vitamines (E, C, B2, B5, F), les filtres UV, les agents anti-radicalaires, les conservateurs, les céramides, les extraits de végétaux et des additifs de formulation tels que des polymères filmogènes anioniques, non ioniques, cationiques ou amphotères, des épaississants polymériques de phase aqueuse ou de phase huileuse, des épaississants non- polymériques de phase aqueuse comme des sels ou des amides d'acide gras hydroxylés ou non, des agents nacrants, des agents opacifiants, des colorants solubles dans le milieu, des pigments, des charges, des parfums, des huiles d'origine minérale, végétale et/ou synthétique, des esters d'acides et/ou d'alcools gras, des cires, des agents de stabilisation de pH comme les acides, les bases, les sels, des solvants organiques, des silicones, des électrolytes, et leurs mélanges. Les quantités des différents ingrédients additionnels de la composition selon l'invention sont celles généralement utilisées dans les domaines considérés et sont en particulier comprises dans la gamme allant de 0,001 à 20% du poids total de la composition. En outre, cette composition est préparée selon les méthodes usuelles. The composition for the treatment of the desquamative states of the hair and / or the scalp according to the invention may, in addition, contain at least one additional ingredient conventionally used in the fields under consideration and chosen from principles cosmetic active agents having a beneficial effect on the hair and / or the scalp such as zinc salts of organic acid (acetate, glycolate, lactate, gluconate or citrate) or mineral (chloride and sulphate), vitamins (E, C , B2, B5, F), UV filters, anti-radical agents, preservatives, ceramides, plant extracts and formulation additives such as anionic, nonionic, cationic or amphoteric film-forming polymers, polymeric thickeners of aqueous phase or oily phase, nonpolymeric thickeners of aqueous phase such as hydroxylated or nonhydroxylated fatty acid salts or amides, pearlescent agents, opacifying agents, dyes soluble in the medium, pigments, fillers perfumes, oils of mineral, vegetable and / or synthetic origin, esters of acids and / or fatty alcohols, waxes, pH stabilizing agents such as acids, bases, salts, organic solvents silicones, electrolytes, and mixtures thereof. The amounts of the various additional ingredients of the composition according to the invention are those generally used in the fields in question and are in particular in the range of 0.001 to 20% of the total weight of the composition. In addition, this composition is prepared according to the usual methods.
Bien entendu l'homme du métier veillera à choisir les éventuels ingrédients additionnels et/ou leur quantité de telle manière que les propriétés avantageuses de l'association d'au moins une bactériocine et d'au moins un prébiotique selon l'invention, à savoir l'activité vis-à-vis des états desquamatifs des cheveux et/ou du cuir chevelu ne soit pas ou substantiellement pas, altérée par l'adjonction envisagée. Of course, those skilled in the art will take care to choose the possible additional ingredients and / or their quantity in such a way that the advantageous properties of the combination of at least one bacteriocin and at least one prebiotic according to the invention, namely the activity vis-à-vis the desquamative states of the hair and / or the scalp is not or substantially not, impaired by the addition envisaged.
Avantageusement, le pH de la composition pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu de la présente invention est choisi dans la gamme allant de 2 à 1 1 et préférentiel lement de 3 à 10 par exemple de 5 à 8. Selon encore un autre aspect, la composition pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu comprend au moins un autre actif contre les états desquamatifs du cuir chevelu. Les autres agents de lutte contre les états desquamatifs du cuir chevelu sont choisis de préférence parmi les sels de pyridinethione comme le zinc pyrithione, les dérivés de 1 -hydroxy-2-pyrrolidone comme la piroctone et la piroctone olamine ; les sulfures de sélénium comme le disulfure de sélénium ; le climbazole, l'acide undécylénique ; le Kétoconazole , le cyclopirox ou leurs mélanges. En pratique, l'agent actif additionnel ou le mélange d'agents actifs additionnels peuvent représenter de 0,001 % à 10% en poids par rapport au poids total de la composition et préférentiel lement de 0,1 à 5% en poids. Selon encore un autre aspect, l'invention a pour objet un procédé de traitement cosmétique du cuir chevelu destiné à lutter contre les états desquamatifs du cuir chevelu, caractérisé en ce qu'on applique sur les cheveux et/ou le cuir chevelu, une composition pour le traitement des états desquamatifs des cheveux et/ou du cuir chevelu telle que définie précédemment. Advantageously, the pH of the composition for treating the desquamative states of the hair and / or scalp of the present invention is chosen in the range from 2 to 1 1 and preferably from 3 to 10, for example from 5 to 8. According to yet another aspect, the composition for the treatment of desquamative conditions of the hair and / or scalp comprises at least one other active against the desquamative states of the scalp. The other agents for combating the desquamative states of the scalp are preferably chosen from pyridinethione salts such as zinc pyrithione, 1-hydroxy-2-pyrrolidone derivatives such as piroctone and piroctone olamine; selenium sulfides such as selenium disulfide; climbazole, undecylenic acid; ketoconazole, cyclopirox or their mixtures. In practice, the additional active agent or the mixture of additional active agents may represent from 0.001% to 10% by weight relative to the total weight of the composition and preferably from 0.1 to 5% by weight. According to yet another aspect, the subject of the invention is a method of cosmetic treatment of the scalp for combating the desquamative states of the scalp, characterized in that a composition is applied to the hair and / or the scalp. for the treatment of desquamative conditions of the hair and / or scalp as defined above.
L'invention a encore pour objet un procédé de traitement cosmétique des cheveux et/ou du cuir chevelu, pour lutter contre les pellicules ou la dermite séborrhéique, caractérisé en ce qu'on applique sur les cheveux et/ou le cuir chevelu une composition telle que décrite ci-dessus. The subject of the invention is also a process for the cosmetic treatment of the hair and / or the scalp, for combating dandruff or seborrheic dermatitis, characterized in that a composition is applied to the hair and / or the scalp. as described above.
Le procédé de traitement cosmétique des états desquamatifs des cheveux et/ou du cuir chevelu selon l'invention est particulièrement adapté lorsque les dits états desquamatifs du cuir chevelu sont induits par la levure du genre Malassezia spp., en particulier M. furfur. The method of cosmetic treatment of the desquamative states of the hair and / or scalp according to the invention is particularly suitable when said desquamative states of the scalp are induced by the yeast of the genus Malassezia spp., In particular M. furfur.
L'homme du métier pourra adapter l'intensité des effets recherchés en sélectionnant les bactériocines les plus actives contre un ou plusieurs germes responsables des effets mentionnés ci-dessus. De même, l'homme du métier pourra adapter les effets recherchés en sélectionnant les prébiotiques les mieux métabolisés par la flore bénéfique de la peau, des cheveux ou du cuir chevelu. Those skilled in the art can adapt the intensity of the desired effects by selecting the most active bacteriocins against one or more germs responsible for the effects mentioned above. Similarly, the skilled person can adapt the desired effects by selecting the prebiotics best metabolized by the beneficial flora of the skin, hair or scalp.
Les exemples qui suivent servent à illustrer la présente invention. Les quantités sont données en pourcentage massique par rapport au poids total de la composition. Les composés sont, selon le cas, cités en noms chimiques ou en noms CTFA (International Cosmetic Ingrédient Dictionary and Handbook). The following examples serve to illustrate the present invention. The amounts are given as a percentage by weight relative to the total weight of the composition. The compounds are, as the case may be, listed in chemical names or CTFA names (International Cosmetic Ingredient Dictionary and Handbook).
EXEMPLES EXAMPLES
Exemple 1 : Lait de soin hydratant Example 1: Moisturizing care milk
La formule suivante est préparée: The following formula is prepared:
Nisine 0,01 g Nisin 0.01 g
Actilight® 0,1 g Actilight® 0.1 g
Alcool stéarylique 5,1 g Stearyl alcohol 5.1 g
Glyceryl stéarate 1 ,5g Glyceryl stearate 1, 5g
Dimethicone 2,0g Dimethicone 2.0g
Vaseline 4,0g Vaseline 4.0g
Huile de vaseline 1 1 g Vaseline oil 1 1 g
10 PEG-100 stéarate 1 ,5g 10 PEG-100 stearate 1, 5g
Glycérine 8,0g Glycerin 8,0g
Eau déminéralisée qsp 100g Ce lait est adapté aux personnes à peaux sensibles et peut être utilisé à raison d'une application quotidienne sur le corps, pour un effet hydratant et protecteur. Demineralized water qs 100g This milk is suitable for people with sensitive skin and can be used for a daily application on the body, for a moisturizing and protective effect.
Exemple 2: crème anti-acné (émulsion huile/eau) epidermidine 1 g EXAMPLE 2 Anti-Acne Cream (Oil / Water Emulsion) Epidermidine 1 g
Bioecolia® de Solabia de 3 g Solabia Bioecolia® of 3 g
Polyacryloyldiméthyltaurate d'ammonium 0,40 g Polyacryloyldimethyltaurate ammonium 0.40 g
Gomme de xanthane 0,20 g 0.20 g xanthan gum
Conservateurs 0,80 g Preservatives 0.80 g
EDTA disodique 0,05 g Disodium EDTA 0.05 g
Glycérol 5,00 g Glycerol 5.00 g
Mélange d'alcool cétéarylique/tartrate de dimyristyle/C12-15 Pareth-7/ PPG-25- Laureth-25 1 ,50 g Alcool stéarylique 1 ,00 g Cetearyl Alcohol Blend / Dimyristyl Tartrate / C12-15 Pareth-7 / PPG-25-Laureth-25 1, 50 g Stearyl alcohol 1, 00 g
Mélange de stéarate de glycéryle / stéarate de PEG-100 2,00 g Mixture of glyceryl stearate / PEG-100 stearate 2.00 g
Cyclohexasiloxane 10,00 g Cyclohexasiloxane 10.00 g
Méthoxycinnamate d'éthylhexyle 1 ,00 g Ethylhexyl methoxycinnamate 1.00 g
Parfum 0,01 g Perfume 0.01 g
Eau déminéralisée qsp 100g Demineralized water qs 100g
Cette crème douce et fraîche permet de lutter contre les problèmes d'acné avec une bonne efficacité. This sweet and fresh cream helps fight against acne problems with good efficiency.
Exemple 3: spray antipelliculaire Example 3 Dandruff Spray
On réalise une composition contenant A composition containing
- Nisine 0,5 g - Nisin 0.5 g
- Bioecolia® de Solabia 1 ,5 g - Bioecolia® Solabia 1, 5 g
- 1 g d'AQ 38S (Eastman Chemicals) 1 g of AQ 38S (Eastman Chemicals)
- Eau qsp 65 g - Water qs 65 g
On introduit cette composition dans un bidon aérosol. On pressurise en introduisant sous pression 35 g de diméthyléther. Le spray est appliqué sur le cuir chevelu. This composition is introduced into an aerosol can. Pressurized by introducing under pressure 35 g of dimethyl ether. The spray is applied on the scalp.
Exemple 4: Emulsions pour Roll-on Example 4 Emulsions for Roll-on
Ingrédients (Nom INCI) Roll-on Roll-on Ingredients (INCI name) Roll-on Roll-on
Emulsion DEO Emulsion AT Emulsion DEO Emulsion AT
ALUMINUM CHLOROHYDRATE ALUMINUM CHLOROHYDRATE
0 30 0 30
CHLORHYDROL 50 (SUMIT REHEIS) CHLORHYDROL 50 (SUMIT REHEIS)
EXPANSED MILLED PERLITE 6,5 1 EXPANSED MILLED PERLITE 6.5 1
(OPTIMAT 1430 OR - Word Minerais) (OPTIMAT 1430 OR - Word Minerals)
Pédiocine PA-1 0, 1 0, 1 Pediocin PA-1 0, 1 0, 1
Actilight -Beghin Meiji 0, 1 0, 1 Actilight -Beghin Meiji 0, 1 0, 1
POLY DIMETHYLSILOXANE POLY DIMETHYLSILOXANE
(VISCOSITE: 350 CST) (VISCOSITY: 350 CST)
0,5 0,5 0.5 0.5
(DOW CORNING 200 FLUID 350 CST - Dow Corning) (Dow Corning 200 Fluid 350 CST - Dow Corning)
CETEARYL ALCOHOL 2,5 2,5 CETEARYL ALCOHOL 2,5 2,5
CETEARETH-33 1 ,25 1 ,25 PPG-15 STEARYL ETHER 3 3 (ARLAMOL E Croda) CETEARETH-33 1, 25 1, 25 PPG-15 STEARYL ETHER 3 3 (ARLAMOL E Croda)
EAU PERMUTEE qsp 100 qsp 100 PERMUTE WATER qs 100 qs 100
On note une efficacité déodorante après un jour d'usage. Deodorant efficiency is noted after one day of use.
Exemple 5: composition pour aérosol anhydre déodorant EXAMPLE 5 Composition for Anhydrous Deodorant Aerosol
La formule est pressurisée avec de l'isobutane. The formula is pressurized with isobutane.
On note une efficacité déodorante après un jour d'usage. Deodorant efficiency is noted after one day of use.
Exemple 5: aérosol anhydre antitranspirant Example 5 Anhydrous Antiperspirant Aerosol
Nom INCI Référence commerciale Quantités en % en poids INCI name Commercial reference Quantities in% by weight
ALUMINUM ALUMINUM
5,25 5.25
CHLOROHYDRATE REACH 103 (SUMIT REHEIS) CHLOROHYDRATE REACH 103 (SUMIT REHEIS)
STEARALKON IUM TIXOGEL MP 250 (rockwood STEARALKON IUM TIXOGEL MP 250 (rockwood
0,39 0.39
BENTONITE additives) BENTONITE additives)
Nisine Nisaplin (Danisco) 0, 1 Nisin Nisaplin (Danisco) 0, 1
Raftilose® Orafti 1 Raftilose® Orafti 1
ISOPROPYL PALMITATE ISOPROPYL PALMITATE (Cognis) 0,9 ISOPROPYL PALMITATE ISOPROPYL PALMITATE (Cognis) 0.9
ISOBUTANE ISOBUTANO (Repsol) 85 ISOBUTANE ISOBUTANO (Repsol) 85
DOW CORNING 245 FLUID (Dow DOW CORNING 245 FLUID (Dow
CYCLOPENTASILOXANE QSP CYCLOPENTASILOXANE QSP
corning) corning)
CYCLOPENTASILOXANE DOW CORNING 1501 FLUID (dow CYCLOPENTASILOXANE DOW CORNING 1501 FLUID (dow
1 ,35 1, 35
(and) DIMETHICONOL corning) (and) DIMETHICONOL corning)
CITROFLEX 2 CITROFLEX 2
TRIETHYL CITRATE (Vertellus) 1 ,05 La formule est pressurisée avec de l'isobutane. TRIETHYL CITRATE (Vertellus) 1, 05 The formula is pressurized with isobutane.
On note une efficacité déodorante après un jour d'usage. Deodorant efficiency is noted after one day of use.
Exemple 6: Stick anhydre déodorant Example 6 Deodorant Anhydrous Stick
Exemple 7: évaluation de l'efficacité des bactériocines Example 7: Evaluation of the Efficacy of Bacteriocins
L'action inhibitrice des peptides plantaricine A, plantaricine C19 et plantaricine 149 a été évaluée sur la croissance des germes C. xerosis (CIP 100653 - principal germe responsable des mauvaises odeurs corporelles) et S. epidermidis (ATCC 155 bactérie commensale bénéfique trouvée dans la flore microbienne de la peau). L'action de la plantaricine 149 a également été testée sur le germe P. acnés (CIP 531 17 - germe trouvé chez les personnes dont la peau est à tendance grasse ou hyperséborrhéique et responsable du développement de l'acné). Les peptides étaient à une concentration de 100 mg/L dans du DMSO. The inhibitory action of the plantaricin A, plantaricin C19 and plantaricin 149 peptides was evaluated on the growth of C. xerosis germs (CIP 100653 - the main germ responsible for unpleasant body odors) and S. epidermidis (ATCC 155 commensal beneficial bacterium found in the microbial flora of the skin). The action of plantaricin 149 has also been tested on P. acnes germ (CIP 531 17 - germ found in people whose skin has a tendency to fat or hyperseborrheic and responsible for the development of acne). The peptides were at a concentration of 100 mg / L in DMSO.
Le test est basé sur la détection de l'activité en milieu solide et la confirmation de cette activité en milieu liquide. Les expériences sur C. xerosis ont été réalisées à 26°C, une pré-étude ayant permis de montrer que cette souche forme des agrégats à 35°C, The test is based on the detection of the activity in solid medium and the confirmation of this activity in a liquid medium. The experiments on C. xerosis were carried out at 26 ° C, a pre-study having shown that this strain forms aggregates at 35 ° C,
Spécifiquement, chaque souche testée recouvrait la gélose d'une boîte de culture en milieu solide. Des puits ont été réalisés dans la gélose et les produits à tester ont été déposés dans ces puits. Les produits testés sont les peptides mentionnés ci-dessus, à une concentration de 100 mg/L dans le DMSO, l'Irgasan à 200 mg/L utilisé comme témoin positif (encore appelée Triclosan, cette molécule est un bactéricide reconnu utilisé notamment dans le traitement des mauvaises odeurs), et l'eau ou du DMSO comme témoin négatif. Ces cultures solides ont permis de montrer l'innocuité des solvants et l'efficacité du contrôle positif sur tous les germes testés. Specifically, each strain tested covered the agar of a culture dish in a solid medium. Wells were made in the agar and the products to be tested were deposited in these wells. The products tested are the peptides mentioned above, at a concentration of 100 mg / L in DMSO, Irgasan 200 mg / L used as a positive control (also called Triclosan, this molecule is a recognized bactericide used especially in the treatment of bad smells), and water or DMSO as a negative control. These solid cultures made it possible to show the safety of the solvents and the effectiveness of the positive control on all the germs tested.
Les bactériocines testées ont toutes présenté un effet inhibiteur sur la croissance de C. xerosis (retard de croissance de 16, 22 and 46 heures pour la plantaricine A, plantaricine C19 et plantaricine 149, respectivement). Cependant, aucune des plantaricines testées n'a d'effet inhibiteur sur la croissance de la souche bénéfique S. epidermidis. The bacteriocins tested all showed an inhibitory effect on the growth of C. xerosis (16, 22 and 46 hours growth retardation for plantaricin A, plantaricin C19 and plantaricin 149, respectively). However, none of the plantaricins tested has an inhibitory effect on the growth of the beneficial S. epidermidis strain.
La plantaricine 149 a un effet inhibiteur sur la croissance de P. acnés. Plantaricin 149 has an inhibitory effect on the growth of P. acnes.
L'effet des peptides sur la croissance de ces différentes souches a également été évalué d'un point de vue quantitatif en milieu liquide. The effect of the peptides on the growth of these different strains was also evaluated from a quantitative point of view in a liquid medium.
L'action de la plantaricine A, la plantaricine C19 et la plantaricine 149 vis-à-vis de C. xerosis et leur innocuité sur S. epidermidis a été confirmée en milieu liquide. The action of plantaricin A, plantaricin C19 and plantaricin 149 against C. xerosis and their safety on S. epidermidis was confirmed in a liquid medium.
Les tests en milieu liquide ont également montré que la plantaricine 149 a également un fort effet inhibiteur sur la croissance de P. acnés (observation d'un retard de croissance de 19h en présence du peptide). Tests in liquid medium also showed that plantaricin 149 also has a strong inhibitory effect on the growth of P. acnes (observation of a 19h growth retardation in the presence of the peptide).
Claims
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0959154A FR2954140A1 (en) | 2009-12-17 | 2009-12-17 | COSMETIC OR DERMATOLOGICAL COMPOSITIONS BASED ON BACTERIOCINS AND PREBIOTICS |
| FR0959154 | 2009-12-17 | ||
| US29098809P | 2009-12-30 | 2009-12-30 | |
| US61/290,988 | 2009-12-30 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2011073437A2 true WO2011073437A2 (en) | 2011-06-23 |
| WO2011073437A3 WO2011073437A3 (en) | 2012-08-23 |
Family
ID=42338230
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2010/070167 Ceased WO2011073437A2 (en) | 2009-12-17 | 2010-12-17 | Bacteriocin- and prebiotic-based cosmetic or dermatological compositions |
Country Status (2)
| Country | Link |
|---|---|
| FR (1) | FR2954140A1 (en) |
| WO (1) | WO2011073437A2 (en) |
Cited By (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011073438A3 (en) * | 2009-12-17 | 2012-08-16 | L'oreal | Cosmetic method for treating body odours using a bacteriocin based composition |
| ITRM20110406A1 (en) * | 2011-07-28 | 2013-01-29 | Giuliani Spa | PLANTARICINE AND BIOMASS INCLUDING PLANTARS FOR USE IN THE COSMETIC AND MEDICAL FIELD. |
| JP2013516981A (en) * | 2010-01-12 | 2013-05-16 | ジュリアーニ・エッセ・ピ・ア | Method for preparing biomass containing plantaricin and its use in the medical field |
| CN103622901A (en) * | 2013-11-21 | 2014-03-12 | 广州立白企业集团有限公司 | Skin cleaner composition capable of regulating microecological balance of skin and preparation method thereof |
| WO2013122931A3 (en) * | 2012-02-14 | 2014-03-13 | The Procter & Gamble Company | Topical use of a skin-commensal prebiotic agent and compositions containing the same |
| FR2996138A1 (en) * | 2012-09-28 | 2014-04-04 | Biochimie Appliquee Soc | Composition, useful for cutaneous production of antimicrobic peptides, for preparing soothing foaming gel, cream for skin irritation and post depilatory foam and to treat skin disorders e.g. dermatitis, comprises prebiotic oligosaccharides |
| WO2015005787A1 (en) * | 2013-07-11 | 2015-01-15 | Micreos Human Health B.V. | Combination treatment for atopic dermatitis |
| EP3049093A2 (en) * | 2013-09-25 | 2016-08-03 | L'Oréal | Bacteria of the stenotrophomonas genus and/or growth inducer of said bacteria for preventing and/or treating atopic dermatitis |
| EP3485869A1 (en) * | 2017-11-16 | 2019-05-22 | DSM IP Assets B.V. | Use of phytantriol as an antimicrobial agent |
| WO2019173782A1 (en) * | 2018-03-08 | 2019-09-12 | Plexus Worldwide Llc | Compositions and methods for skin renewal |
| US10806769B2 (en) | 2016-03-31 | 2020-10-20 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| US10874700B2 (en) | 2016-03-31 | 2020-12-29 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| CN113038959A (en) * | 2018-11-16 | 2021-06-25 | S-生物医药有限公司 | Composition for treating oxidative stress-related skin diseases and skin aging and diagnosis of oxidative stress-related skin diseases |
| IT202000005122A1 (en) * | 2020-03-10 | 2021-09-10 | Marina Acampora | Prebiotic conditioner based on inulin and tannins for the characterization of cosmetic hair products |
| WO2022186286A1 (en) * | 2021-03-05 | 2022-09-09 | 株式会社林原 | Hair setting power enhancer |
| US11564879B2 (en) | 2016-11-23 | 2023-01-31 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| CN116370605A (en) * | 2023-05-15 | 2023-07-04 | 山东博森医学工程技术有限公司 | Medicine for improving proliferation and phagocytic capacity of immune cells |
| WO2023148292A1 (en) * | 2022-02-03 | 2023-08-10 | Embion Technologies Sa | Biomass extract for skin applications |
| CN116712526A (en) * | 2023-07-04 | 2023-09-08 | 云曼(深圳)健康科技有限公司 | Anti-hair loss hair growth essence and preparation method thereof |
| CN117098527A (en) * | 2021-01-12 | 2023-11-21 | 贝甘明治公司 | Cosmetic method and composition based on short-chain fructooligosaccharides and natural starch |
| JP2024502375A (en) * | 2021-01-12 | 2024-01-18 | ベギン,メイジ | Cosmetic methods and compositions based on short-chain fructooligosaccharides and raw starches |
| CN119530103A (en) * | 2025-01-21 | 2025-02-28 | 广东科隆生物科技有限公司 | A kind of glutamine aminotransferase culture medium and its preparation method and fermentation process |
| US12539324B2 (en) | 2016-03-31 | 2026-02-03 | Gojo Industries, Inc. | Antimicrobial peptide stimulating sanitizing composition |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR3097436B1 (en) * | 2019-06-24 | 2023-10-13 | Oreal | Cosmetic composition comprising a combination of at least one oligosaccharide and/or polysaccharide associated with a mannose monosaccharide |
| FR3118711A1 (en) * | 2021-01-12 | 2022-07-15 | Beghin Meiji | COSMETIC METHOD AND COMPOSITION BASED ON SHORT CHAIN FRUCTO-OLIGOSACCHARIDES |
Citations (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2528378A (en) | 1947-09-20 | 1950-10-31 | John J Mccabe Jr | Metal salts of substituted quaternary hydroxy cycloimidinic acid metal alcoholates and process for preparation of same |
| US2781354A (en) | 1956-03-26 | 1957-02-12 | John J Mccabe Jr | Imidazoline derivatives and process |
| US3792068A (en) | 1971-04-02 | 1974-02-12 | Procter & Gamble | Dry powder aerosol antiperspirant composition incorporating dry powder antiperspirant active complex and process for its preparation |
| EP0218200A2 (en) | 1985-10-11 | 1987-04-15 | D. Swarovski & Co. | Decorative glass body |
| JPH02295912A (en) | 1989-05-10 | 1990-12-06 | Shiseido Co Ltd | Cosmetic |
| WO1992006778A1 (en) | 1990-10-17 | 1992-04-30 | Societe D'exploitation De Produits Pour Les Industries Chimiques (S.E.P.P.I.C.) | Use of fatty alcohol based compositions for preparing emulsions, method of preparing emulsions and emulsions so obtained |
| WO1993018743A1 (en) | 1992-03-20 | 1993-09-30 | Janssen Pharmaceutica N.V. | Agent for regulating the greasiness of the skin |
| US5412004A (en) | 1991-11-21 | 1995-05-02 | Kose Corporation | Silicone polymer, paste-like silicone composition, and w/o-type cosmetic composition comprising the same |
| WO1995013863A1 (en) | 1993-11-19 | 1995-05-26 | Societe D'exploitation De Produits Pour Les Industries Chimiques, S.E.P.P.I.C. | Alkylglycoside-containing concentrate and uses thereof |
| US5538793A (en) | 1993-12-28 | 1996-07-23 | Shin-Etsu Chemical Co., Ltd. | Silicone rubber particles coated with silicone resin |
| EP0761204A1 (en) | 1995-09-07 | 1997-03-12 | L'oreal | Use of extracts of filamentous, non photosynthetic bacteria and the composition containing them |
| EP0796861A1 (en) | 1996-03-22 | 1997-09-24 | L'oreal | Silated derivatives of salicilic acid with desquamative properties |
| EP0847752A1 (en) | 1996-11-26 | 1998-06-17 | L'oreal | Without rub off topical composition containing a fluorosilicon compound |
| EP0852949A2 (en) | 1997-03-31 | 1998-07-15 | Shiseido Company Limited | Use of alpha-amino-acids for enhancing desmosomal degradation or stratum corneum desquamation |
| US5783657A (en) | 1996-10-18 | 1998-07-21 | Union Camp Corporation | Ester-terminated polyamides of polymerized fatty acids useful in formulating transparent gels in low polarity liquids |
| US5811487A (en) | 1996-12-16 | 1998-09-22 | Dow Corning Corporation | Thickening silicones with elastomeric silicone polyethers |
| WO1998047610A1 (en) | 1997-04-21 | 1998-10-29 | Societe D'exploitation De Produits Pour Les Industries Chimiques - Seppic | Composition based on alkylpolyglycosides and fatty alcohols, and uses thereof |
| US5874069A (en) | 1997-01-24 | 1999-02-23 | Colgate-Palmolive Company | Cosmetic composition containing silicon-modified amides as thickening agents and method of forming same |
| EP0899330A1 (en) | 1997-08-29 | 1999-03-03 | L'oreal | Polypeptide isolated from the epidermis and its use |
| US5919441A (en) | 1996-04-01 | 1999-07-06 | Colgate-Palmolive Company | Cosmetic composition containing thickening agent of siloxane polymer with hydrogen-bonding groups |
| US5981680A (en) | 1998-07-13 | 1999-11-09 | Dow Corning Corporation | Method of making siloxane-based polyamides |
| US6051216A (en) | 1997-08-01 | 2000-04-18 | Colgate-Palmolive Company | Cosmetic composition containing siloxane based polyamides as thickening agents |
| FR2792190A1 (en) | 1999-04-16 | 2000-10-20 | Sophim | Non-greasy wax-ester emollients for use in skin care preparations obtained by interesterification of triglycerides with an alcohol, distilling off residual alcohol, decolorizing and fridge or hydrogenating the product |
| EP1129694A1 (en) | 2000-02-15 | 2001-09-05 | L'oreal | Use of components for modifying physico-chemical properties of the skin and/or the mucosal tissue as adhesion prevention/decreasing adhesion agents for microorganisms |
| EP1133979A2 (en) | 2000-02-15 | 2001-09-19 | L'oreal | Use of specific fatty compounds for modifying the physico-chemical properties of the skin and/or the mucous membrane as agents for preventing or reducing the adhesion of micro-organisms thereon |
| EP1151742A2 (en) | 2000-05-04 | 2001-11-07 | L'oreal | Use of fibres in a make-up or skin-care composition for giving a matt skin appearance |
| WO2002047619A2 (en) | 2000-12-12 | 2002-06-20 | L'oreal S.A. | Cosmetic composition comprising a polymer and fibres |
| WO2002056847A1 (en) | 2001-01-17 | 2002-07-25 | L'oreal | Cosmetic composition comprising a polymer and a fluorinated oil |
| EP1333022A2 (en) | 2002-02-04 | 2003-08-06 | L'oreal | Compounds containing derivatives of cyclopentane and their use to stimulate skin peeling |
| EP1333021A2 (en) | 2002-02-04 | 2003-08-06 | L'oreal | Compositions containing derivatives of jasmonic acid and their use for stimulating skin peeling |
| WO2004105736A1 (en) | 2003-05-30 | 2004-12-09 | Symrise Gmbh & Co. Kg | Use of diphenylmethane derivatives as tyrosinase inhibitors |
| DE10324567A1 (en) | 2003-05-30 | 2004-12-23 | Symrise Gmbh & Co. Kg | Use of dihydroxyphenylmethane derivatives, e.g. styrylresorcinol, for control of microorganisms that cause dandruff, body odor, acne and mycoses |
| EP1529523A1 (en) | 2003-11-07 | 2005-05-11 | L'oreal | Cosmetic use of phytantriol as an agent for preventing or reducing the adhesion of microorganisms on the skin and/or mucosal surfaces |
| WO2005053632A2 (en) | 2003-12-01 | 2005-06-16 | Galderma Research & Development, S.N.C. | Use of ppar activators in cosmetics and dermatology |
| FR2869796A1 (en) | 2004-05-07 | 2005-11-11 | Oreal | COSMETIC COMPOSITION FOR MAKE-UP AND / OR SKIN CARE, IN PARTICULAR FACE |
| FR2916634A1 (en) | 2007-06-01 | 2008-12-05 | Jean Noel Thorel | Synergistic combination, useful e.g. as regulating agent of microbial flora of skin and to prepare cosmetic or pharmaceutical composition to treat oily or mixed skin, of fructo-oligosaccharides and inducer of antimicrobial peptide |
| DE102008012457A1 (en) | 2007-06-19 | 2008-12-24 | Cognis Ip Management Gmbh | Hydrocarbon mixture, useful e.g. in decorative cosmetics, preferably lipsticks, lip gloss, eye shade, mascara, eye pencil, nail polish and make-up formulations and eye shade, comprises linear hydrocarbons |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB8811760D0 (en) * | 1988-05-18 | 1988-06-22 | Thomae Gmbh Dr K | Antibiotic |
| DE58905744D1 (en) * | 1988-07-15 | 1993-11-04 | Thomae Gmbh Dr K | METHOD FOR OBTAINING, ISOLATING AND PURIFYING EPIDERMINE. |
| DE3938140A1 (en) * | 1989-11-16 | 1991-08-08 | Beiersdorf Ag | DESODRATING COSMETIC AGENTS |
| FR2678166B1 (en) * | 1991-06-27 | 1993-10-22 | Bioeurope | COSMETIC COMPOSITIONS CONTAINING GLUCOOLIGOSACCHARIDES. |
| US6645506B1 (en) * | 1997-04-18 | 2003-11-11 | Ganeden Biotech, Inc. | Topical compositions containing extracellular products of Pseudomonas lindbergii and Emu oil |
| EP1174118A1 (en) * | 2000-06-28 | 2002-01-23 | Cognis France S.A. | Use of inulin and derivatives thereof |
| FR2831059B1 (en) * | 2001-10-18 | 2004-07-23 | Jean Noel Thorel | USE OF FRUCTO-OLIGOSACCHARIDES AS COSMETIC ACTIVE INGREDIENTS, COMPOSITIONS COMPRISING SAME AND METHOD OF CONSERVATION USING THE SAME |
| JP4509517B2 (en) * | 2002-09-19 | 2010-07-21 | ロート製薬株式会社 | Agent that inhibits Staphylococcus aureus skin adhesion and promotes Staphylococcus epidermidis skin adhesion |
| FR2847165A1 (en) * | 2002-11-15 | 2004-05-21 | Jean Noel Thorel | Topical cosmetic or pharmaceutical compositions, containing nisin as non-irritating preservative having broad antimicrobial spectrum, also effective against greasy skin and acne |
| KR100536522B1 (en) * | 2003-03-07 | 2005-12-14 | (주)바이오솔루션 | Novel bacteriocin isolated from human skin and the use as therapeutic agent of an acne |
| JP2005089355A (en) * | 2003-09-17 | 2005-04-07 | Ichimaru Pharcos Co Ltd | Isomaltoligosaccharide-containing skin normal flora improving agent |
| WO2005091933A2 (en) * | 2004-03-04 | 2005-10-06 | E-L Management Corporation | Skin treatment method with lactobacillus extract |
| CN101336680A (en) * | 2007-07-02 | 2009-01-07 | 威海紫光科技园有限公司 | Fig fruit jam and production method thereof |
| GB0714030D0 (en) * | 2007-07-18 | 2007-08-29 | Novacta Biosystems Ltd | The use of type-B lantibiotic-based compounds having antimicrobial activity |
-
2009
- 2009-12-17 FR FR0959154A patent/FR2954140A1/en not_active Withdrawn
-
2010
- 2010-12-17 WO PCT/EP2010/070167 patent/WO2011073437A2/en not_active Ceased
Patent Citations (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2528378A (en) | 1947-09-20 | 1950-10-31 | John J Mccabe Jr | Metal salts of substituted quaternary hydroxy cycloimidinic acid metal alcoholates and process for preparation of same |
| US2781354A (en) | 1956-03-26 | 1957-02-12 | John J Mccabe Jr | Imidazoline derivatives and process |
| US3792068A (en) | 1971-04-02 | 1974-02-12 | Procter & Gamble | Dry powder aerosol antiperspirant composition incorporating dry powder antiperspirant active complex and process for its preparation |
| EP0218200A2 (en) | 1985-10-11 | 1987-04-15 | D. Swarovski & Co. | Decorative glass body |
| JPH02295912A (en) | 1989-05-10 | 1990-12-06 | Shiseido Co Ltd | Cosmetic |
| WO1992006778A1 (en) | 1990-10-17 | 1992-04-30 | Societe D'exploitation De Produits Pour Les Industries Chimiques (S.E.P.P.I.C.) | Use of fatty alcohol based compositions for preparing emulsions, method of preparing emulsions and emulsions so obtained |
| US5412004A (en) | 1991-11-21 | 1995-05-02 | Kose Corporation | Silicone polymer, paste-like silicone composition, and w/o-type cosmetic composition comprising the same |
| WO1993018743A1 (en) | 1992-03-20 | 1993-09-30 | Janssen Pharmaceutica N.V. | Agent for regulating the greasiness of the skin |
| WO1995013863A1 (en) | 1993-11-19 | 1995-05-26 | Societe D'exploitation De Produits Pour Les Industries Chimiques, S.E.P.P.I.C. | Alkylglycoside-containing concentrate and uses thereof |
| US5538793A (en) | 1993-12-28 | 1996-07-23 | Shin-Etsu Chemical Co., Ltd. | Silicone rubber particles coated with silicone resin |
| EP0761204A1 (en) | 1995-09-07 | 1997-03-12 | L'oreal | Use of extracts of filamentous, non photosynthetic bacteria and the composition containing them |
| EP0796861A1 (en) | 1996-03-22 | 1997-09-24 | L'oreal | Silated derivatives of salicilic acid with desquamative properties |
| US5919441A (en) | 1996-04-01 | 1999-07-06 | Colgate-Palmolive Company | Cosmetic composition containing thickening agent of siloxane polymer with hydrogen-bonding groups |
| US5783657A (en) | 1996-10-18 | 1998-07-21 | Union Camp Corporation | Ester-terminated polyamides of polymerized fatty acids useful in formulating transparent gels in low polarity liquids |
| EP0847752A1 (en) | 1996-11-26 | 1998-06-17 | L'oreal | Without rub off topical composition containing a fluorosilicon compound |
| US5811487A (en) | 1996-12-16 | 1998-09-22 | Dow Corning Corporation | Thickening silicones with elastomeric silicone polyethers |
| US5874069A (en) | 1997-01-24 | 1999-02-23 | Colgate-Palmolive Company | Cosmetic composition containing silicon-modified amides as thickening agents and method of forming same |
| EP0852949A2 (en) | 1997-03-31 | 1998-07-15 | Shiseido Company Limited | Use of alpha-amino-acids for enhancing desmosomal degradation or stratum corneum desquamation |
| WO1998047610A1 (en) | 1997-04-21 | 1998-10-29 | Societe D'exploitation De Produits Pour Les Industries Chimiques - Seppic | Composition based on alkylpolyglycosides and fatty alcohols, and uses thereof |
| US6051216A (en) | 1997-08-01 | 2000-04-18 | Colgate-Palmolive Company | Cosmetic composition containing siloxane based polyamides as thickening agents |
| EP0899330A1 (en) | 1997-08-29 | 1999-03-03 | L'oreal | Polypeptide isolated from the epidermis and its use |
| US5981680A (en) | 1998-07-13 | 1999-11-09 | Dow Corning Corporation | Method of making siloxane-based polyamides |
| FR2792190A1 (en) | 1999-04-16 | 2000-10-20 | Sophim | Non-greasy wax-ester emollients for use in skin care preparations obtained by interesterification of triglycerides with an alcohol, distilling off residual alcohol, decolorizing and fridge or hydrogenating the product |
| EP1129694A1 (en) | 2000-02-15 | 2001-09-05 | L'oreal | Use of components for modifying physico-chemical properties of the skin and/or the mucosal tissue as adhesion prevention/decreasing adhesion agents for microorganisms |
| EP1133979A2 (en) | 2000-02-15 | 2001-09-19 | L'oreal | Use of specific fatty compounds for modifying the physico-chemical properties of the skin and/or the mucous membrane as agents for preventing or reducing the adhesion of micro-organisms thereon |
| EP1151742A2 (en) | 2000-05-04 | 2001-11-07 | L'oreal | Use of fibres in a make-up or skin-care composition for giving a matt skin appearance |
| WO2002047619A2 (en) | 2000-12-12 | 2002-06-20 | L'oreal S.A. | Cosmetic composition comprising a polymer and fibres |
| WO2002056847A1 (en) | 2001-01-17 | 2002-07-25 | L'oreal | Cosmetic composition comprising a polymer and a fluorinated oil |
| EP1333022A2 (en) | 2002-02-04 | 2003-08-06 | L'oreal | Compounds containing derivatives of cyclopentane and their use to stimulate skin peeling |
| EP1333021A2 (en) | 2002-02-04 | 2003-08-06 | L'oreal | Compositions containing derivatives of jasmonic acid and their use for stimulating skin peeling |
| WO2004105736A1 (en) | 2003-05-30 | 2004-12-09 | Symrise Gmbh & Co. Kg | Use of diphenylmethane derivatives as tyrosinase inhibitors |
| DE10324567A1 (en) | 2003-05-30 | 2004-12-23 | Symrise Gmbh & Co. Kg | Use of dihydroxyphenylmethane derivatives, e.g. styrylresorcinol, for control of microorganisms that cause dandruff, body odor, acne and mycoses |
| EP1529523A1 (en) | 2003-11-07 | 2005-05-11 | L'oreal | Cosmetic use of phytantriol as an agent for preventing or reducing the adhesion of microorganisms on the skin and/or mucosal surfaces |
| WO2005053632A2 (en) | 2003-12-01 | 2005-06-16 | Galderma Research & Development, S.N.C. | Use of ppar activators in cosmetics and dermatology |
| FR2869796A1 (en) | 2004-05-07 | 2005-11-11 | Oreal | COSMETIC COMPOSITION FOR MAKE-UP AND / OR SKIN CARE, IN PARTICULAR FACE |
| FR2916634A1 (en) | 2007-06-01 | 2008-12-05 | Jean Noel Thorel | Synergistic combination, useful e.g. as regulating agent of microbial flora of skin and to prepare cosmetic or pharmaceutical composition to treat oily or mixed skin, of fructo-oligosaccharides and inducer of antimicrobial peptide |
| DE102008012457A1 (en) | 2007-06-19 | 2008-12-24 | Cognis Ip Management Gmbh | Hydrocarbon mixture, useful e.g. in decorative cosmetics, preferably lipsticks, lip gloss, eye shade, mascara, eye pencil, nail polish and make-up formulations and eye shade, comprises linear hydrocarbons |
Non-Patent Citations (4)
| Title |
|---|
| "Silica silylate", 2000 |
| C. FOX, COSMETICS AND TOILETRIES, vol. 101, November 1986 (1986-11-01), pages 101 - 112 |
| M. BODANSZKY: "Principles of Peptides Synthesis", 1993, SPRINGER-VERLAG |
| M.R. PORTER: "Handbook of Surfactants", 1991, BLACKIE & SON, pages: 116 - 178 |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011073438A3 (en) * | 2009-12-17 | 2012-08-16 | L'oreal | Cosmetic method for treating body odours using a bacteriocin based composition |
| JP2013516981A (en) * | 2010-01-12 | 2013-05-16 | ジュリアーニ・エッセ・ピ・ア | Method for preparing biomass containing plantaricin and its use in the medical field |
| ITRM20110406A1 (en) * | 2011-07-28 | 2013-01-29 | Giuliani Spa | PLANTARICINE AND BIOMASS INCLUDING PLANTARS FOR USE IN THE COSMETIC AND MEDICAL FIELD. |
| WO2013014687A1 (en) * | 2011-07-28 | 2013-01-31 | Giuliani S.P.A. | Plantaricins and biomass comprising plantaricins for use in cosmetic and medical fields |
| WO2013122931A3 (en) * | 2012-02-14 | 2014-03-13 | The Procter & Gamble Company | Topical use of a skin-commensal prebiotic agent and compositions containing the same |
| WO2013122932A3 (en) * | 2012-02-14 | 2014-08-07 | The Procter & Gamble Company | Topical use of a skin-commensal prebiotic agent and compositions containing the same |
| CN104105470A (en) * | 2012-02-14 | 2014-10-15 | 宝洁公司 | Topical use of a skin-commensal prebiotic agent and compositions containing the same |
| FR2996138A1 (en) * | 2012-09-28 | 2014-04-04 | Biochimie Appliquee Soc | Composition, useful for cutaneous production of antimicrobic peptides, for preparing soothing foaming gel, cream for skin irritation and post depilatory foam and to treat skin disorders e.g. dermatitis, comprises prebiotic oligosaccharides |
| US10357547B2 (en) | 2013-07-11 | 2019-07-23 | Micreos Human Health B.V. | Combination treatment for atopic dermatitis |
| EP3789033A1 (en) * | 2013-07-11 | 2021-03-10 | Micreos Human Health B.V. | Combination treatment for atopic dermatitis |
| WO2015005787A1 (en) * | 2013-07-11 | 2015-01-15 | Micreos Human Health B.V. | Combination treatment for atopic dermatitis |
| EP3049093A2 (en) * | 2013-09-25 | 2016-08-03 | L'Oréal | Bacteria of the stenotrophomonas genus and/or growth inducer of said bacteria for preventing and/or treating atopic dermatitis |
| CN103622901A (en) * | 2013-11-21 | 2014-03-12 | 广州立白企业集团有限公司 | Skin cleaner composition capable of regulating microecological balance of skin and preparation method thereof |
| CN103622901B (en) * | 2013-11-21 | 2016-01-20 | 广州立白企业集团有限公司 | Composition of skin cleanser of a kind of energy regulation of skin microecological balance and preparation method thereof |
| US11998575B2 (en) | 2016-03-31 | 2024-06-04 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| US11633451B2 (en) | 2016-03-31 | 2023-04-25 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| US12539324B2 (en) | 2016-03-31 | 2026-02-03 | Gojo Industries, Inc. | Antimicrobial peptide stimulating sanitizing composition |
| US10806769B2 (en) | 2016-03-31 | 2020-10-20 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| US10874700B2 (en) | 2016-03-31 | 2020-12-29 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| US11564879B2 (en) | 2016-11-23 | 2023-01-31 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| JP7187740B2 (en) | 2017-11-16 | 2022-12-13 | ディーエスエム アイピー アセッツ ビー.ブイ. | new use |
| CN109939090A (en) * | 2017-11-16 | 2019-06-28 | 帝斯曼知识产权资产管理有限公司 | novel use |
| EP3485869A1 (en) * | 2017-11-16 | 2019-05-22 | DSM IP Assets B.V. | Use of phytantriol as an antimicrobial agent |
| JP2019089760A (en) * | 2017-11-16 | 2019-06-13 | ディーエスエム アイピー アセッツ ビー.ブイ.Dsm Ip Assets B.V. | New use |
| WO2019173782A1 (en) * | 2018-03-08 | 2019-09-12 | Plexus Worldwide Llc | Compositions and methods for skin renewal |
| CN113038959A (en) * | 2018-11-16 | 2021-06-25 | S-生物医药有限公司 | Composition for treating oxidative stress-related skin diseases and skin aging and diagnosis of oxidative stress-related skin diseases |
| IT202000005122A1 (en) * | 2020-03-10 | 2021-09-10 | Marina Acampora | Prebiotic conditioner based on inulin and tannins for the characterization of cosmetic hair products |
| JP2024502375A (en) * | 2021-01-12 | 2024-01-18 | ベギン,メイジ | Cosmetic methods and compositions based on short-chain fructooligosaccharides and raw starches |
| CN117098527A (en) * | 2021-01-12 | 2023-11-21 | 贝甘明治公司 | Cosmetic method and composition based on short-chain fructooligosaccharides and natural starch |
| WO2022186286A1 (en) * | 2021-03-05 | 2022-09-09 | 株式会社林原 | Hair setting power enhancer |
| WO2023148292A1 (en) * | 2022-02-03 | 2023-08-10 | Embion Technologies Sa | Biomass extract for skin applications |
| CN116370605A (en) * | 2023-05-15 | 2023-07-04 | 山东博森医学工程技术有限公司 | Medicine for improving proliferation and phagocytic capacity of immune cells |
| CN116370605B (en) * | 2023-05-15 | 2023-09-12 | 山东博森医学工程技术有限公司 | Medicine for improving proliferation and phagocytic capacity of immune cells |
| CN116712526A (en) * | 2023-07-04 | 2023-09-08 | 云曼(深圳)健康科技有限公司 | Anti-hair loss hair growth essence and preparation method thereof |
| CN119530103A (en) * | 2025-01-21 | 2025-02-28 | 广东科隆生物科技有限公司 | A kind of glutamine aminotransferase culture medium and its preparation method and fermentation process |
Also Published As
| Publication number | Publication date |
|---|---|
| FR2954140A1 (en) | 2011-06-24 |
| WO2011073437A3 (en) | 2012-08-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2011073437A2 (en) | Bacteriocin- and prebiotic-based cosmetic or dermatological compositions | |
| EP1847247B1 (en) | Compositions comprising a hydroxylated diphenyl-methane derivative | |
| JP5922327B2 (en) | Kit for cosmetic prescription | |
| US20080008673A1 (en) | Compositions comprising at least one C-glycoside derivative | |
| EP2694026B1 (en) | Cosmetic method for treating human body odors using a 4-(3-ethoxy-4-hydroxyphenyl)alkylketone or 2-ethoxy 4-hydroxyalkylphenol compound | |
| WO2008110672A1 (en) | Use of a c-glycoside derivative as an anti-oxidant agent | |
| FR2822377A1 (en) | USE OF FIBERS AS ANTI-IRRITANT AGENT IN A COSMETIC OR DERMATOLOGICAL COMPOSITION | |
| FR2978035A1 (en) | USE AS ANTITRANSPIRANT OF A MULTIVALENT CATION SALT WITHOUT ANTITRANSPIRANT HALOGEN ALUMINUM OR COMPOUND LIKELY TO REACT WITH THIS SALT TO PRODUCE ANTITRANSPIRANT EFFECT | |
| FR2992210A1 (en) | MATIFYING EFFECT COMPOSITION COMPRISING HYDROPHOBIC AEROGEL PARTICLES AND SILICONE ELASTOMER PARTICLES | |
| FR2977151A1 (en) | Use of an alkali silicate as antiperspirants contains a composition comprising aluminum chlorohydrate and zirconium | |
| JP2016529275A (en) | Cosmetic composition comprising a sulfonic acid polymer, a hydrophobic polysaccharide and a silicone surfactant | |
| FR2940053A1 (en) | Cosmetic antioxidant and antiradical agent, useful for protecting skin and/or exoskeleton against oxidative stress, contains anisic acid or derivative | |
| EP3166583A1 (en) | Cosmetic use of spiculisporic acid as a deodorant active agent | |
| FR2913598A1 (en) | Use of C-glycoside derivative e.g. as agent intended to reduce the appearance/visibility of pores, for tightening the pores/decreasing the size of pores, and as agent for tightening/ refining the skin texture | |
| FR2990850A1 (en) | COSMETIC COMPOSITION COMPRISING THE ASSOCIATION OF A LIPOPHILIC DERIVATIVE OF SALICYLIC ACID, AN ANTI-TRANSPIRANT ALUMINUM SALT OR COMPLEX, AND AN ACETIC N, DIACIDAL AMINO ACID SALT | |
| FR2978032A1 (en) | PROCESS FOR TREATING HUMAN TRANSPIRATION USING MULTIVALENT CATION HALIDE AND NONION NITROGEN SALT | |
| FR2900047A1 (en) | USE OF A DIPHENYL-METHANE DERIVATIVE FOR THE CARE OF OILY SKINS | |
| NL2037617B1 (en) | Cosmetic composition | |
| FR2976485A1 (en) | Use of water-dispersible flocculant polymer comprising in side chain of non-quaternary amino groups carried by a side substituent directly attached to a main chain, as an antiperspirant active ingredient | |
| FR2961392A1 (en) | USE OF AMINO ACID DERIVATIVES AS A TREATMENT AGENT FOR HUMAN PERSPIRATION; NEW COMPOUNDS; COMPOSITIONS CONTAINING THEM | |
| FR2940121A1 (en) | Composition for the care of greasy skin, especially for eliminating bacteria responsible for acne, contains a combination of phthalamidoperhexanoic acid and zinc salicylate | |
| FR2996756A1 (en) | COSMETIC USE OF A MONO-UNSATURATED FATTY ACID OR ONE OF ITS SALTS AND / OR ITS ESTERS AS DEODORANT ACTIVE | |
| FR3013980A1 (en) | COMPOSITION BASED ON SEA WATER AND ALGAE EXTRACTS WITH ANTI-AGING SKIN ACTIVITY | |
| US20260014070A1 (en) | Cosmetic composition | |
| FR2939685A1 (en) | Composition, useful e.g. in dermatological composition for treating acne and/or rosacea, reducing the skin imperfections, preferably for oily skin, comprises hydroxylated diphenyl-methane derivative and phenanthrenol compound, in a medium |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10796380 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 10796380 Country of ref document: EP Kind code of ref document: A2 |