WO2010132169A1 - Fungicidal active compound compositions for turf treatment - Google Patents
Fungicidal active compound compositions for turf treatment Download PDFInfo
- Publication number
- WO2010132169A1 WO2010132169A1 PCT/US2010/031239 US2010031239W WO2010132169A1 WO 2010132169 A1 WO2010132169 A1 WO 2010132169A1 US 2010031239 W US2010031239 W US 2010031239W WO 2010132169 A1 WO2010132169 A1 WO 2010132169A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- accoiding
- active compound
- fungicide
- compound combinations
- compositions
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/34—Shaped forms, e.g. sheets, not provided for in any other sub-group of this main group
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/44—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a nitrogen atom attached to the same carbon skeleton by a single or double bond, this nitrogen atom not being a member of a derivative or of a thio analogue of a carboxylic group, e.g. amino-carboxylic acids
- A01N37/50—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a nitrogen atom attached to the same carbon skeleton by a single or double bond, this nitrogen atom not being a member of a derivative or of a thio analogue of a carboxylic group, e.g. amino-carboxylic acids the nitrogen atom being doubly bound to the carbon skeleton
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/28—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N<
- A01N47/38—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N< containing the group >N—CO—N< where at least one nitrogen atom is part of a heterocyclic ring; Thio analogues thereof
Definitions
- Fungicidal active compound compositions for turf treatment are provided.
- the piescnt invention iclatcs to novel compositions comprising a fungicide that inhibits mitochondrial respiration at the QoI site, (QoI inhibitor), a contact fungicide, a dcmcthylation inhibitoi and pigments, which aic highly suitable foi conti oiling unwanted phytopathogenic fungi Moreovci, the fungicidal compositions ate able Io contiol mold and fungus infection in glasses, in paiticulai tuif glasses
- noithein tcgions such as Canada and the not them United States, which aie subject to temperate climates in the late summer and eaily fail and snow conditions in late fall and cat Iy wintci cciiain moulds such as dollar spot, Sclei otinia homeoc ⁇ pa, and snow molds, such as Microdochitim nivale, Typhula yernata and T ⁇ plnda Uhik ⁇ iemis, represent a chronic pioblcm foi turf glass giowcts
- Typical piograms consist of thtee applications pnor to peimancnt snow covei and an additional application afler the snow covet is gone in the spring
- Several commercial fungicide pioducts have been appioved foi use against dollar spot and snow mould species
- COMPASSTM (irifloxystiobin; ( ⁇ E)- ⁇ (meihoxyimino)-2-[[[(E)-[I-[3- (trifluoiomethylphcnyl]cthylidine]-amino]oxy]methyl]bcnzenacelic acid methyl cstei) is an aromatic dioxime fungicide which has been appiovcd foi the contioE of leaf spot, Fm ⁇ intn patch and blown patch diseases in tut f gtass Trifloxystiobi ⁇ is a quinone outside inhibitoi (QoI) and a member of the class of aromatic dioxime fungicides described in U S Pat No 5,238,956 BCS 09-6004
- GREEN GT 1 M iprodione; 3-(3,5-dichioropheny])W-(l-methylcthyl)-2 s 4-dioxo-l- imidazolidinccaboximide
- lpiodionc is a member of the class of 3-phc ⁇ yl hydantoin compounds described in U. S, Pat No. 3,755,350,
- iprodione to control the spread of snow mold in turf grasses (especially the gray snow molds T. lncamata and T ishikariemh) is highly variable due to the varying amounts of fungus inoculums, the varying length of snow cover under which snow mould inoculums thrive and the varying winlcr temperatures that exist across the country from year to year.
- a iclativcly high dose may be required as well as multiple applications.
- Phthalocyanines ate known pigments having many applications, such as pigments for inks and coatings and even for turf grass paints Sec, for example, DE 2,51 1,077 and .JP 03/221576 Copper phthalocyaninc has been used, but only in combination with certain othei active ingredients, foi enhancing turf quality.
- U.S. Pat. No. 5,599,804 describes a method of combating fungi and enhancing turf quality in turf grass by applying in specific ratios ccitain phthalocyanines in combination with phosphorous acid or an alkaline earth metal salt thereof or with certain r ⁇ onocster salts of phosphorous acid U S .
- Pal No 5,643,852 describes a method of enhancing turf quality in turf giass by applying in specific ratios certain phthalocyanines in combination with (i) phosphorous acid or an alkaline earth metal salt thereof or certain monoester salts of phosphorous acid and (ii) certain ethylene bisdiihiocarbamatc contact fungicides
- U.S. Pat. No. 5,336,661 describes a method of treating bent grass and enhancing turf quality by applying in specific iatios (i) ccitain monoester salts of phosphorous acid and (ii) a metallic ethylene bisdilhiocarbaniate contact fungicide.
- This patent also describes a specific composition containing a combination of aluminum tris(O cthylphosphonate) (fosctyl-al) and a manganese-zinc ethylene bisdithiocarbamate complex (mancozeb) used in a foim (FORE fungicide) believed to have contained an unknown amount of the phthalocya ⁇ ine compound Pigment Blue 15
- the picscnt invention provides active compound combinations or compositions which achieve the object at least in some aspects, BCS 09-6004
- the active compound combinations or compositions accoiding to the invention do not only exhibit an additive effect of the activity of the individual components, but a synergistic effect. Therefore, f ⁇ stly, the customary application rate of the individual substances can be reduced.
- the active compound combinations according to the invention offer a high degice of activity against phytopathogens even in cases whcic the individual compounds are employed in amounts where, foi their pait, they no longer show (sufficient) activity. In principle, this allows, firstly, the activity spectrum to be broadened and, secondly, belter safely during handling,
- the active compound combinations according to the invention have further surprising properties which, in a wider sense, may also be called synergistic, such as, for example: broadening of the activity spechum, for example to resistant pathogens of plant diseases; lower application iatcs of the active compounds; sufficient control of pests with the aid of the active compound combinations according to the invention even at application rales where the individual compounds show no oi virtually no activity; advantageous behavioui during formulation or during use, for example during grinding, sieving, emulsifying, dissolving or application; improved storage stability and light stability; advantageous iesidue formation; improved loxicological or ecotoxicological behavioui; improved properties of the plant, for example better growth, increased harvest yields, a better developed root system, a largci leaf area, greener leaves, stronger' shoots, less seed required, lower' phototoxicity, mobilization of the defence system of the plant, good compatibility with plants.
- synergistic such as, for example: broadening of the activity spechum, for
- the use of the active compound combinations or compositions accoiding to the invention contributes considerably to keeping young plant stems healthy, which inci cases, fot example, the winter survival of the cereal seed treated, and also safeguards quality and yield.
- the active compound combinations according to the invention may contribute to enhanced systemic action. Even if the individual compounds of the combination have no sufficient systemic properties, the active compound combinations according to the invention may stilt have this property. In a siniHai manner, the active compound combinations according to the invention may result in higher persistency of the fungicidal action.
- QoI inhibitor fungicides include azoxystrobin, cncstrobin, picoxystrobin, pyraclostrobi ⁇ , krcsoxim-methyl, trifloxysliobin, dimoxystrobin, metominostiobi ⁇ , orysastrobin, famoxadone, fluoxastrobin, fcnamidone, pyribcncaib, preferred are azoxystrobin, pyiaclostrobin, trifloxystrobi ⁇ and fluoxastiobin, particular preferred is irifloxystiobin.
- Contact fungicides include PP-fungicidcs such as fludioxinil, dicatboimides such as iprodione, chlozoiinate, procymidone, vi ⁇ clozolin, AH-fungicides such as biphenyl, chloioneb, dicloian, quintozcne (PCNB), tccnazenc (TCNB), tolclofos-methyl, dithiocarbamates such as ferbam, mancozeb, mancb, metiram, propincb, thiiam, zi ⁇ eb and ziram, phthalimidcs such as captan, captafol and folpet, chloronilriles such as chlorothalonil, sulfamides such as dichlofluanid and toiyfluanid, guanidines, such as dodine, guazatinc and iminooctadine, triazine
- DMI-f ⁇ ngicidcs include triazoies such as azaconazole, bitcrtanol, bromuconazolc, cyproconazoic, difcnoconazolc, diniconazole, diniconazole-M, epoxiconazofe, etaconazole, fenbuconazolc, fluquinconazole, flusilazoie, flutiiafol, hexaconazole, imibcnco ⁇ azole, ipconazolc, metconazolc, myclobutanil, penconazole, propiconazole, prothioconazole, simcconazolc, tcbuconazole, tetiaconazole, triad imefon, triadimenol and tiiticonazole, piefe ⁇ ed arc prothioconazole, tebuconazole,
- composition according to the invention comprises as compound B) i) trifloxystrobin as QoI inhibitor fungicide, ii) iprodione as contact fungicide and iii) tiitriconazolc as DMl- fu ⁇ gicide.
- composition according to the invention comprises i) trifloxystrobin as QoI inhibitor fungicide and ii) iprodione as contact fungicide.
- Suitable pigments A) include phthalocyanincs
- Suitable phthalocyanines are substituted copper phthalocyanincs which may be substituted from 1 to 4 times on each isoindolc gioup independently,
- suitable substituents for the isoindole groups of copper phthaiocyanine dyes include but are not limited to halogen, unsubstituted or substituted lower alkyl, lower alkoxy, alkylamino, alkylthio, ammonium, BCS 09-6004
- the ionic or ionizablc substituents can have as counterions the alkali metals, preferably lithium, sodium, or potassium, the alkaline earth metals, prcfciably beryllium, magnesium, calcium, strontium, and barium, and various ammonium ions
- lower alkyl and lower aikoxy are genet ally meant alkyl groups of from 1 to 6 carbon atoms and aikoxy groups of from 1 to 6 caibon atoms
- Suitable copper phthalocyanines aic commercially available and include but aie not limited to Pigment Blue 16, Vat Blue 29, Pigment Blue 15, Hcliogcn Green GG, Ingrain Blue 14, Ingrain Blue 5, Ingrain Blue 1 , Pigment Gieen 37, and Pigment Green 7.
- the copper phthalocya ⁇ ine is Pigment Blue 15, also known as phthalocyanine blue and Green pigment (Green 7)
- composition according to the invention comprises as compound A) a green pigment (Gieen 7)
- the term "active compound combination” means various possible combinations of the active compounds mentioned above, such as, for example, rcadymixcs, tank mixes (which is to be understood as meaning spray mixtures prepared prior to application from the foundations of the individual active compounds by mixing and diluting) or combinations thereof (for example, a binary rcadymix of two of the active compounds mentioned above is converted with a formulation of the third individual substance into a tank mix).
- the individual active compounds may also be used successively, i.c one after the other, within a reasonable interval of a few hours or days, and, in the treatment of seed, also, for example, by applying a plurality of layers comprising different active compounds.
- compositions comprising the active compound combinations according to the invention.
- the compositions are fungicidal compositions comprising agriculturally suitable carriers or extenders.
- carrier' is to be understood as meaning a natural or- synthetic, organic or inorganic substance which is mixed oi combined with the active compounds for better applicability, in particular for application to plants ot plant parts or seeds
- the carrier which may be solid or liquid, is generally inert and should be suitable for use in agriculture.
- Suitable solid carriers arc: for example ammonium salts and natural ground minerals, such as kaolins, clays, talc, chalk, quartz, attapulgite, monlmorillonile or diatomaceous earth, and ground synthetic minerals, such as finely divided silica, alumina and natural or synthetic silicates, resins, waxes, solid fertilizers, water, alcohols, especially butanol, organic solvents, mineral oils and vegetable oils, and also derivatives thereof. It is also possible to use mixtures of such carriers.
- emulsificrs and/or foam-formers are: for' example nonionic and anionic cmulsifiers, such as polyoxycthylene fatly acid esters, polyoxyethylcne fatty alcohol ethers, for example alkylaiyl polyglycol ethers, alkylsulphonates, alkyl sulphates, arylsulphonatcs, and also protein hydrolysates, Suitable dispcrsants are: for example lignosulphite waste liquors and mclhylccHulose.
- Suitable liquefied gaseous extenders or carriers are liquids which arc gaseous at ambient tcmperture and under atmospheric pressure, for example aeiosol piopcllants, such as butane, propane, nitrogen and carbon dioxide.
- Tackifiers such as carboxymethylcellulose and natural and synthetic polymers in the form of powders, granules and laticcs, such as gum aiabic, polyvinyl alcohol, polyvinyl acetate, or' else natural phospholipids, such as cephalins and lecithins and synthetic phospholipids can be used in the formulations.
- Other possible additives arc mineral and vegetable oils
- Suitable liquid solvents are essentially: aromatic compounds, such as xylene, toluene or alkylnaphthalenes, chlorinated aromatic compounds or chlorinated aliphatic hydrocarbons, such as chlorobenzenes, chloroelhylcncs or 1 methylene chloride, aliphatic hydrocarbons, such as cyclohexane oi 1 paraffins, for example mineral oil fractions, mineral and vegetable oils, alcohols, such as butanol or glycol, and also ethers and esters thereof, ketones, such as acetone, methyl ethyl ketone, methyl isobutyl ketone or cyclohexanonc, strongly potai solvents, such as dimethylformamidc and dimethyl sulphoxide, and also water 1 .
- aromatic compounds such as xylene, toluene or alkylnaphthalenes
- chlorinated aromatic compounds or chlorinated aliphatic hydrocarbons
- compositions according to the invention may comprise additional further components, such as, for example, surfactants.
- surfactants are emulsifieis, dispctsants or wetting agents having ionic or nonionic properties, or mixtures of these surfactants.
- salts of potyacrylic acid salts of lignosulphonic acid, salts of phenoisulphonic acid or ' naphthalenesulphonic acid, polycondensales of ethylene oxide with fatty alcohols or with fatty acids or with fatty amines, substituted phenols (preferably alkylphcnols or as y [phenols), sails of sulphosuccinic esters, taurine derivatives (preferably alkyl tamatcs), phosphoric esters of polyethoxylaicd alcohols or phenols, fatty esters of polyols, and derivatives of the compounds containing sulphates, suiphonates and phosphates
- a surfactant is required if one of the active compounds and/or one of the inert cairicrs is insoluble in water and when the application takes place in water.
- the proportion of surfactants is between 5 and 40 per cent by weight of the composition according to the invention .
- colorants such as inorganic pigments, for example iron oxide, titanium oxide, Prussian blue, and organic dyes, such as alizarin dyes, azo dyes and metal phthalocyaninc dyes, and trace nutrients, such as salts of iron, manganese, boron, copper, cobalt, molybdenum and zinc.
- inorganic pigments for example iron oxide, titanium oxide, Prussian blue
- organic dyes such as alizarin dyes, azo dyes and metal phthalocyaninc dyes
- trace nutrients such as salts of iron, manganese, boron, copper, cobalt, molybdenum and zinc.
- the active compounds can be combined with any solid or liquid additive customarily used for' fo ⁇ nulation purposes.
- compositions according to the invention comprise between 0,05 and 99 per cent by weight of the active compound combination according to the invention, preferably between 5 and 60 pes cent by weight, particularly preferably between 10 and 40 per cent by weight, vciy particularly picfcrably 20 and .30 per cent by weight.
- the composition according to the invention comprises between 2 and 10 per cent by weight, preferably between 4 and 9 per cent by weight, particularly preferably between 5 and 7 per cent by weight compound A), between 1 and 3,3 per cent by weight, preferably between 1,0 and 2,5 per cent by weight, particularly preferably between 1,2 and 2,0 per cent by weight compound Bi) and between 20 and 50 per cent by weight, preferably between 20 and 40 per cent by weight, particularly psefeiably between 20 and 30 per cent by weight compound Bii) and/or Biii), the sum of the compounds adding up to 100 per cent by weight.
- the fungicidal compositions according to the invention can be used for the curative or protective control of phytopalhogenic fungi
- the compositions according to the invention may be applied to healthy or diseased turfs. Prophylactic application to healthy turf may be helpful in preventing turf diseases. Application to turf containing one or more turf diseases is helpful in treating the one or more turf diseases.
- compositions arc also helpful in improving turf quality aftei snow cover release in spring
- the fungicidal compositions accoiding to the invention can be used for lurfgrass disease control on golf courses, sod farms, and institutional areas wheic fine tuif is grown
- the active compound combinations or compositions according to the invention can be used as such or, depending on their respective physical and/or chemical properties, in the form of their formulations or the use forms prepared therefrom, such as aerosols, capsule suspensions, cold- fogging concentrates, warm-fogging concentrates, encapsulated granules, fine granules, ilowable concentrates for the treatment of seed, ready ⁇ to ⁇ usc solutions, dustablc powdcis, emulsifiable concentrates, oil-in-water emulsions, water-in-oil emulsions, maciogranules, microgranules, oil- dispersiblc powders, oil-misctbEc flowable concentrates, oil-miscible liquids, foams, pastes, pesticide- coated seed, suspension concentrates, suspoemulsio ⁇ concentrates, soluble concentrates, suspensions, weltable powders, soluble powders, dusts and granules, water-
- the formulations mentioned can be prepared in a manner known per se, for example by mixing the active compounds or the active compound combinations with at least one solvent or diluent, emulsifier, dispersant and/or binder 1 or' fixing agent, water lepellanl, appropr iate siccatives and UV stabilizers and, if appropriate, dyes and pigments, and also further processing auxiliaries.
- the treatment accoiding to the invention of the plants and plant parts with the active compound combinations or compositions is carried out directly or by action on their surroundings, habitat or stosage space using customaiy treatment methods, for' example by dipping, spraying, atomizing, irrigating, evaporating, dusting, fogging, broadcasting, foaming, painting, spreading-on, watering (drenching), drip ungating and, in the case of propagation material, in particular in the case of seeds, furthermore as a powder for' diy seed treatment, a solution for seed treatment, a water-soluble powder for slurry treatment, by incrusting, by coating with one or more coats, etc,
- compositions according to the invention do not only comprise ready-to-use compositions which can be applied with suitable apparatus to the plant or the seed, but also commercial concentrates which have to be diluted with water priot to use
- active compound combinations accoiding to the invention can be present in commercial foimulations and in the use forms prepared from these formulations as a mixture with other active BCS 09-6004
- - 9 - compounds such as insecticides, alliactnnls, sterilanls, bactericides, acaricides, nematicides, fungicides, giowlh regulators or herbicides
- the picscnt invention can be practiced with all turfgtasscs, including cool season tuilgrasses and warm season lurfgiasses.
- cool season turfgrasscs ai"c bluegrasscs ⁇ Poa spp such as Kentucky blucgiass ⁇ Poa pratemis L ), rough bluegrass (Poa trivialis L.), Canada bluegrass (Poa compressa L), annual bluegrass (Poa annuci L,..), upland bluegrass (Poa glaticantha Gaudin), wood bluegiass (Poa nemoraUs L ), and bulbous biuegrass ⁇ Poa bulbosa L); the bentgrasses and redtop (Agrostis ⁇ p), such as creeping bcntgiass (Agrostis palustris Huds,), colonial bentgiass (Agrostis /emu's Sib
- Wedged wheatgiass such as fairway wheatgiass (Agropyron cristatum [L) Gacitn.), crested wheatgiass ⁇ Agropyron desertorum (Fisch.) Schult ), and western wheatgiass (Agropyron smilhii Rydb,)
- Othei cool season turfgrasscs include beachgrass (Ammoph ⁇ ' a breviligitlata Fein), smooth bromegtass (Broimis biennis Leyss ), cattails such as Timothy (Phleum pratense L ), sand cattail (Phleuin subulatum L), orchaidgrass (Dactylis gbmerata L), weeping alkaligrass ⁇ Puccitiellia distans (L.) Pail .) and crested dog's-tail (C ⁇ nosurus cristatus L )
- Examples of warm season turfgj asses include Bermudagrass ⁇ Cynodo ⁇ spp L. C. Rich), zoysiagiass (Zoysia spp WiIkL) 5 St. Augustine grass ⁇ Stenotaphrum secimdatuin Walt Kunlze), centipedcgrass
- the application rate of the active compound combinations according to the invention is BCS 09-6004
- the active compound combinations or compositions accoiding to the invention can thus be employed foi piotccting plants for a cctlain pes iod of time aftci treatment against attack by the pathogens mentioned
- the pet tod for which protection is piovided extends gencially for 1 to 28 days, piefeiably 1 to 14 days, afiei the licatme ⁇ t of the plants with the active compounds, Ot for up to 200 days after seed tieatment
- the good fungicidal activity of the active compound combinations according to the invention is demonsUated by the example below, Wlicicas the individual active compounds have weaknesses in the fungicidal activity, the combinations exhibit an activity which exceeds a simple addition of activity
- X is the efficacy when active compound A is applied at an application iatc of m g/ha,
- Y is the efficacy when active compound B is applied at an application rate of n g/ha
- Ej is the efficacy when the active compounds A and B aic applied at application rates of m and n g/ha, lespectivcly, and
- E 2 is the efficacy when the active compounds A and B and C aie applied at application iates of m and n and i g/ha, respectively,
- the efficacy is dctcimincd in % 0% means an efficacy wliieh co ⁇ esponds to that of the control, whereas an efficacy of 100% means that no infection is observed
- the activity of the combination is supeiadditive, i.e. a syneigistic effect is present.
- the efficacy actually observed must be greatei than the value, calculated using the formula given above, for the expected efficacy Ei .
- va ⁇ ous compositions compiisc a QoI inhibitor fungicide, a contact fungicide, and a dcmethylalion inhibitor with pigments over a composition containing three classes of fungicides but without pigments
- trials weie conducted at the following sites: 1. University of Massachusetts, Vermont Site; 2. Univeisily of Wisconsin, Sentry Site; .3, University of Wisconsin, Iron Mountain Site; 4. University of Wisconsin, Champion Ml Site; 5 University of Wisconsin, Edina MN Site; 6. Oregon State University, Sun River Site; 1, Washington State University, McCaIl Site; 8. Washington State Univcisity, Chewelah Site; and 9. Washington State University Pullman Site.
- a QoI inhibitor fungicide trifloxysttobin
- iprodione contact fungicide
- tiiticonazoie demcthylation inhibitor
- phthalocyanine Green pigment
- A trifloxystrobin
- B ipiodionc
- C Phthalocyaiiine Green pigment (Green 7);
- c IN STRATA* commercial standard without pigment (piopiconazole + f ⁇ udioxinil + chlorothaloiiil).
- a composition comprising A + B + C + D is the equivalent of INSTRATA* + a pigment.
- a direct comparison can be made to demonstrate the superior effects of a composition containing the three classes of fungicides with a pigment vs. a composition BCS 09-6004
- Table 2 shows the effect of a composition containing differing amounts of a QoI inhibitor fungicide (tiifloxystrobin), a contact fungicide (ipradione), a demethylation inhibitor (triticonazole) and a pigment (phthalocyanine Green pigment) in comparison with INSTRATA* on percentage disease infestation by site.
- a QoI inhibitor fungicide tiifloxystrobin
- a contact fungicide ipradione
- a demethylation inhibitor triticonazole
- a pigment phthalocyanine Green pigment
- composition containing the three classes of fungicides with a pigment cures about the same percentage of disease infestation as INSTRATA*.
- the composition disclosed is as effective as INSTRATA* in killing mold.
- the supetior benefits of a composition additionally comprising a pigment, such as a phthalocyanine include better recovery of the turf after the winter, during the spring green up; belter health of the turf; and better aesthetics (the turf looks visual better)
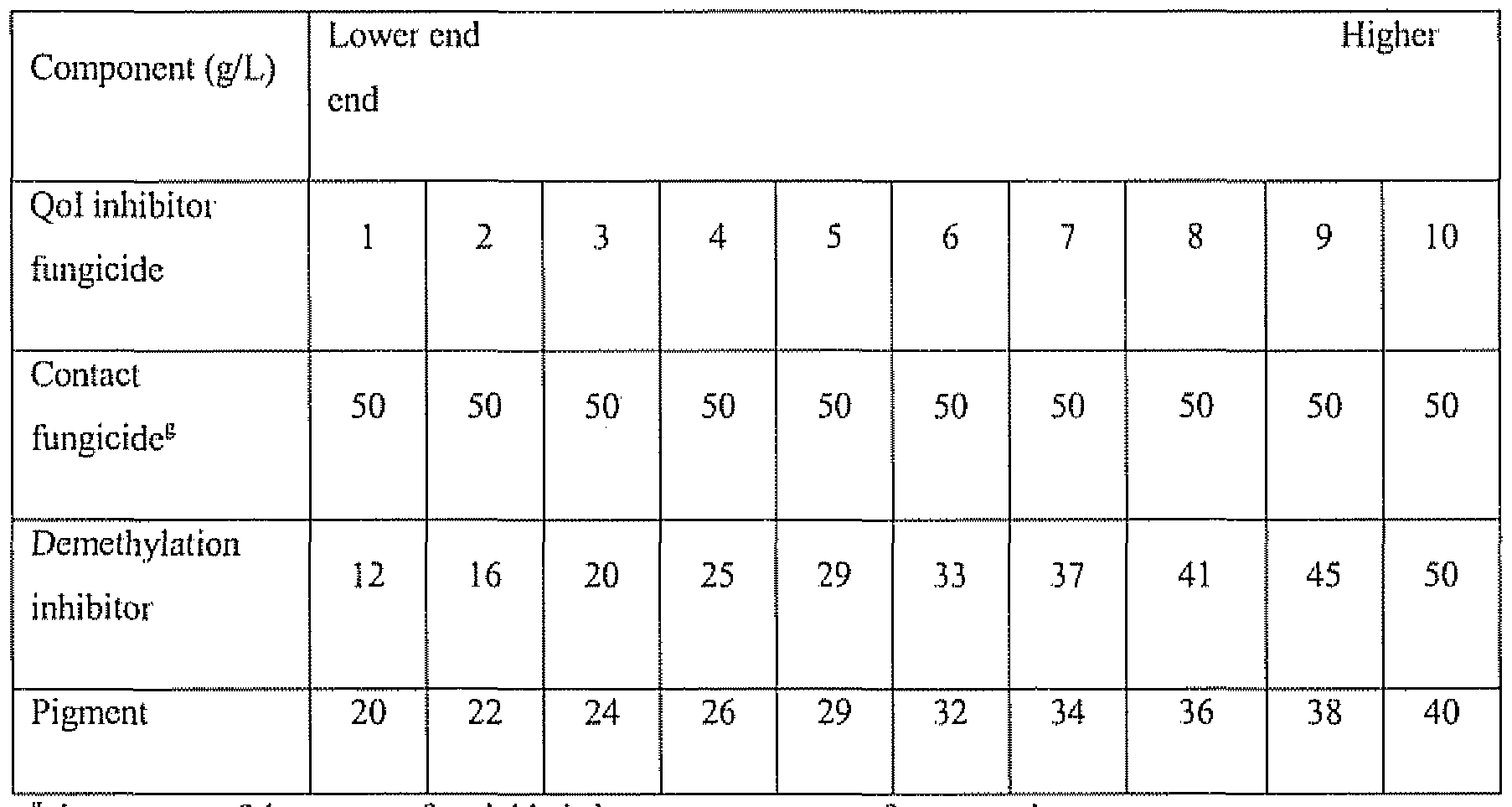
- the Qo ⁇ inhibitor fungicide, contact fungicide, the demcthylation inhibitor; and the pigment may be In any ratio within the composition. Examples of suitable ratios are shown in Table 3,
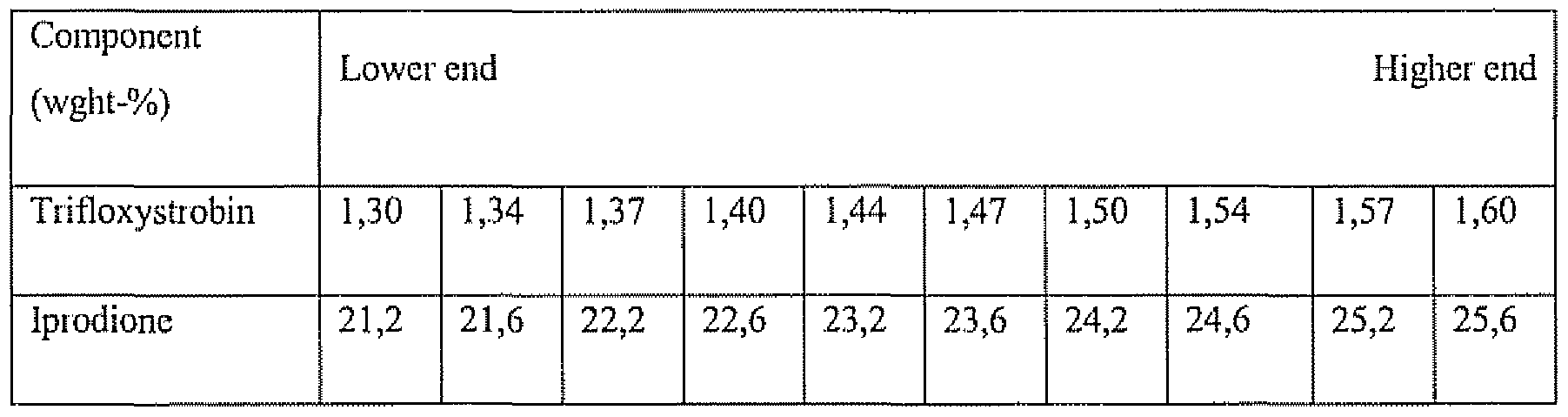
- compositions of trifloxystrobin, ipiodionc, and phthalocyanine Green pigment include those shown in Table 4, suspended in watci .
- Table 5 shows the superior effects of a composition containing 1,47 g/L of trifloxyslrobin, 29,41 g/L of iprodio ⁇ e, 3,14 g/L of ti iticonazole, and 6 g/L of phlhalocyaninc over a commercially available solution of INSTRATA*
- the four -component, composition and INSTRATA* weie applied to a bent grass/Po ⁇ plot at Westmounl Golf & Count! y Club in Kitchener, Ontario, Canada.
- Trials were set up to investigate the efficacy and effect on turf qualitiy of the combination of two active ingredients (Trifloxystrobin, QoI inhibitor fungicide and Iprodione, contact fungicide) and (he pigment (Phthalocyanine).
- the fungicidal compounds wcie applied individually, in combination BCS 09-6004
- Trials were conducted in various places in UK and Germany in 2008 and were between end of .July to early December depending on the individual disease.
- Trial design was a randomized complete-block with 4 replications and a plot area of 4 to 6,25 m 2 . Products were applied as spray application with 400 to 500 L water volume / ha.
- air pressurized sprayers witli a teejet or flatfan noozle were used, operated at 2,2 to 3,0 bar Two applications were ca ⁇ icd out at 28 day interval.
- Turf diseases Pink snow mold (Microd ⁇ chium nivale), Rust (Puccinia spp), Red thread (Laetis ⁇ Ia fucif ⁇ rmis) were assessed as % infected canopy area per plot. Turf quality (color and / or density) was judged on an ordinal scale Data is displayed as average over all assessments.
- c DACONIL* comma cial standard without pigment (chloiotlialonil)
- the data of table 6 shows an impiovemcnt of tuif quality in those plots whcie the the compounds A and B were combined with compound C
- the tieatmcnt with compounds A and B alone and the combination A + B achieved a lowci quality scoie than for A + B + C
- Table 7 shows the effect of a composition containing diffeiing amounts of a QoI inhibitor fungicide (tiifloxystrobin), a contact fungicide (iprodione) and a pigment (phthalocyanine Green pigment) in compaiison with DACONlL * or AMISTAR* on pcicentagc disease infestation by site
- Table 7 % diseased area, Indication Puccinia spp on turf
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Dentistry (AREA)
- Plant Pathology (AREA)
- Engineering & Computer Science (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Toxicology (AREA)
- Catching Or Destruction (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP10775231.3A EP2429287A4 (en) | 2009-05-15 | 2010-04-15 | Fungicidal active compound compositions for turf treatment |
| JP2012510815A JP5635080B2 (en) | 2009-05-15 | 2010-04-15 | Bactericidal active compounds and compositions for turf treatment |
| BRPI1010805A BRPI1010805A8 (en) | 2009-05-15 | 2010-04-15 | COMPOSITION OF ACTIVE FUNGICIDIAL COMPOUNDS FOR LAWN TREATMENT |
| MX2011011930A MX2011011930A (en) | 2009-05-15 | 2010-04-15 | Fungicidal active compound compositions for turf treatment. |
| KR1020117029557A KR101696208B1 (en) | 2009-05-15 | 2010-04-15 | Fungicidal active compound compositions for turf treatment |
| CN201080020663.XA CN102427723B (en) | 2009-05-15 | 2010-04-15 | Fungicidally active compound compositions for treating lawns |
| ZA2011/07904A ZA201107904B (en) | 2009-05-15 | 2011-10-28 | Fungicidal active compound compositions for turf treatment |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17871109P | 2009-05-15 | 2009-05-15 | |
| US61/178,711 | 2009-05-15 | ||
| EP09165641.3 | 2009-07-16 | ||
| EP09165641 | 2009-07-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010132169A1 true WO2010132169A1 (en) | 2010-11-18 |
Family
ID=41354003
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2010/031239 Ceased WO2010132169A1 (en) | 2009-05-15 | 2010-04-15 | Fungicidal active compound compositions for turf treatment |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP2429287A4 (en) |
| JP (1) | JP5635080B2 (en) |
| KR (1) | KR101696208B1 (en) |
| CN (1) | CN102427723B (en) |
| WO (1) | WO2010132169A1 (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102405930A (en) * | 2011-10-20 | 2012-04-11 | 迈克斯(如东)化工有限公司 | Bactericide composition capable of beautifying color for lawn and preparation method thereof |
| WO2013067103A3 (en) * | 2011-11-03 | 2013-06-27 | Bayer Cropscience Lp | Compositions and methods for enhancing plant quality |
| JP2014515378A (en) * | 2011-06-03 | 2014-06-30 | サンコー・エナジー・インコーポレーテッド | Oil-in-water emulsions of paraffin oil for controlling fungal pathogen infection in crop plants |
| WO2015079334A1 (en) * | 2013-11-26 | 2015-06-04 | Upl Limited | A method for controlling rust |
| US9226504B2 (en) | 2010-09-09 | 2016-01-05 | Suncor Energy Inc. | Synergistic paraffinic oil and boscalid fungicides |
| US9357768B2 (en) | 2006-10-05 | 2016-06-07 | Suncor Energy Inc. | Herbicidal composition with increased herbicidal efficacy |
| US9485988B2 (en) | 2008-06-26 | 2016-11-08 | Suncor Energy Inc. | Turfgrass fungicide formulation with pigment |
| US20170265465A1 (en) * | 2014-12-05 | 2017-09-21 | Board Of Trustees Of Michigan State University | Biorational treatment products and methods of using same |
| US9999219B2 (en) | 2004-05-18 | 2018-06-19 | Suncor Energy Inc. | Spray oil and method of use therof for controlling turfgrass pests |
| US12207655B2 (en) | 2019-02-15 | 2025-01-28 | Nutrien Ag Solutions (Canada) Inc. | Protoporphyrin IX derivatives and use thereof to improve the health of plants |
| US12369585B2 (en) | 2017-03-07 | 2025-07-29 | Upl Ltd | Fungicidal combinations |
| US12396455B2 (en) | 2012-06-04 | 2025-08-26 | Nutrien Ag Solutions (Canada) Inc. | Formulations containing paraffinic oil and anti-settling agent |
| WO2026012814A1 (en) | 2024-07-10 | 2026-01-15 | Basf Se | Compositions and methods to enhance crop yield and plant health |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118109316B (en) * | 2024-04-28 | 2024-07-02 | 云南省林业和草原科学院 | Isolation and application of rhizosphere fungus muelleri YAFEF-2016 of pinus massoniana |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006031631A1 (en) * | 2004-09-10 | 2006-03-23 | Syngenta Limited | Substituted isoxazoles as fungicides |
| US20060293188A1 (en) * | 2004-02-13 | 2006-12-28 | Norton Lawrence H | Method of improving grass quality |
| WO2008119439A2 (en) * | 2007-03-29 | 2008-10-09 | Syngenta Participations Ag | Fungicidal compositions comprising a carboxamide derivative, cyprodinil and an unsaturated fatty acid |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5643852A (en) * | 1993-01-13 | 1997-07-01 | North Carolina State University | Fungicidal compositions for the enhancement of turf quality |
| JP3363942B2 (en) * | 1993-03-24 | 2003-01-08 | 日清製粉株式会社 | Manufacturing method of organic fertilizer |
| US5506250A (en) * | 1994-11-14 | 1996-04-09 | Rhone-Poulenc Inc. | Method of treating turf |
| US5599804A (en) * | 1995-04-03 | 1997-02-04 | Rhone-Poulenc, Inc. | Fungicidal compositions for the enhancement of turf quality |
| US20050181949A1 (en) * | 2004-02-13 | 2005-08-18 | Norton Lawrence H. | Method of improving turfgrass quality |
| CN101106903A (en) * | 2005-02-04 | 2008-01-16 | 三井化学株式会社 | Plant disease control composition and control method thereof |
| CA2615547A1 (en) * | 2005-07-21 | 2007-01-25 | Syngenta Participations Ag | Fungicidal compositions comprising tebuconazole |
| CN101262762B (en) * | 2005-09-09 | 2012-07-04 | 拜尔农作物科学股份公司 | Solid formulations of antiseptic mixtures |
| JP5005699B2 (en) * | 2005-10-19 | 2012-08-22 | バイエル・クロツプサイエンス・インコーポレイテツド | Control with fungicides of fungi |
-
2010
- 2010-04-15 CN CN201080020663.XA patent/CN102427723B/en active Active
- 2010-04-15 WO PCT/US2010/031239 patent/WO2010132169A1/en not_active Ceased
- 2010-04-15 JP JP2012510815A patent/JP5635080B2/en active Active
- 2010-04-15 KR KR1020117029557A patent/KR101696208B1/en active Active
- 2010-04-15 EP EP10775231.3A patent/EP2429287A4/en not_active Withdrawn
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060293188A1 (en) * | 2004-02-13 | 2006-12-28 | Norton Lawrence H | Method of improving grass quality |
| WO2006031631A1 (en) * | 2004-09-10 | 2006-03-23 | Syngenta Limited | Substituted isoxazoles as fungicides |
| WO2008119439A2 (en) * | 2007-03-29 | 2008-10-09 | Syngenta Participations Ag | Fungicidal compositions comprising a carboxamide derivative, cyprodinil and an unsaturated fatty acid |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2429287A4 * |
Cited By (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9999219B2 (en) | 2004-05-18 | 2018-06-19 | Suncor Energy Inc. | Spray oil and method of use therof for controlling turfgrass pests |
| US9801369B2 (en) | 2006-10-05 | 2017-10-31 | Suncor Energy Inc. | Herbicidal composition with increased herbicidal efficacy |
| US9357768B2 (en) | 2006-10-05 | 2016-06-07 | Suncor Energy Inc. | Herbicidal composition with increased herbicidal efficacy |
| US9826738B2 (en) | 2008-06-26 | 2017-11-28 | Suncor Energy Inc. | Turfgrass fungicide formulation with pigment |
| US9485988B2 (en) | 2008-06-26 | 2016-11-08 | Suncor Energy Inc. | Turfgrass fungicide formulation with pigment |
| US9750249B2 (en) | 2010-09-09 | 2017-09-05 | Suncor Energy Inc. | Synergistic paraffinic oil and boscalid fungicides |
| US9226504B2 (en) | 2010-09-09 | 2016-01-05 | Suncor Energy Inc. | Synergistic paraffinic oil and boscalid fungicides |
| US9451773B2 (en) | 2011-06-03 | 2016-09-27 | Suncor Energy Inc. | Paraffinic oil-in-water emulsions for controlling infection of crop plants by fungal pathogens |
| JP2014515378A (en) * | 2011-06-03 | 2014-06-30 | サンコー・エナジー・インコーポレーテッド | Oil-in-water emulsions of paraffin oil for controlling fungal pathogen infection in crop plants |
| CN102405930A (en) * | 2011-10-20 | 2012-04-11 | 迈克斯(如东)化工有限公司 | Bactericide composition capable of beautifying color for lawn and preparation method thereof |
| US10517301B2 (en) | 2011-11-03 | 2019-12-31 | Bayer Cropscience Lp | Compositions and methods for enhancing plant quality |
| WO2013067103A3 (en) * | 2011-11-03 | 2013-06-27 | Bayer Cropscience Lp | Compositions and methods for enhancing plant quality |
| US9820490B2 (en) | 2011-11-03 | 2017-11-21 | Bayer Cropscience Lp | Compositions and methods for enhancing plant quality |
| US12396455B2 (en) | 2012-06-04 | 2025-08-26 | Nutrien Ag Solutions (Canada) Inc. | Formulations containing paraffinic oil and anti-settling agent |
| GB2532700B (en) * | 2013-11-26 | 2016-08-31 | Upl Ltd | A method for controlling rust |
| US10694744B2 (en) | 2013-11-26 | 2020-06-30 | Upl Limited | Method for controlling rust |
| WO2015079334A1 (en) * | 2013-11-26 | 2015-06-04 | Upl Limited | A method for controlling rust |
| CN105744834A (en) * | 2013-11-26 | 2016-07-06 | Upl 有限公司 | Methods for controlling rust |
| US10362782B2 (en) | 2013-11-26 | 2019-07-30 | Upl Limited | Method for controlling rust |
| EP3073826B1 (en) | 2013-11-26 | 2019-10-30 | UPL Limited | A method for controlling soybean rust |
| US10499638B2 (en) | 2013-11-26 | 2019-12-10 | Upl Limited | Method for controlling rust |
| GB2532700A (en) * | 2013-11-26 | 2016-05-25 | Upl Ltd | A method for controlling rust |
| US10694745B2 (en) | 2013-11-26 | 2020-06-30 | Upl Limited | Method for controlling rust |
| US9788544B2 (en) | 2013-11-26 | 2017-10-17 | Upl Limited | Method for controlling rust |
| US10701935B2 (en) | 2013-11-26 | 2020-07-07 | Upl Limited | Method for controlling rust |
| EP4014738B1 (en) | 2013-11-26 | 2024-10-30 | UPL Ltd | A method for controlling rust |
| EP4014741B1 (en) | 2013-11-26 | 2024-10-30 | UPL Ltd | A method for controlling rust |
| US20170265465A1 (en) * | 2014-12-05 | 2017-09-21 | Board Of Trustees Of Michigan State University | Biorational treatment products and methods of using same |
| US12369585B2 (en) | 2017-03-07 | 2025-07-29 | Upl Ltd | Fungicidal combinations |
| US12376592B2 (en) | 2017-03-07 | 2025-08-05 | Upl Ltd | Fungicidal combinations |
| US12207655B2 (en) | 2019-02-15 | 2025-01-28 | Nutrien Ag Solutions (Canada) Inc. | Protoporphyrin IX derivatives and use thereof to improve the health of plants |
| WO2026012814A1 (en) | 2024-07-10 | 2026-01-15 | Basf Se | Compositions and methods to enhance crop yield and plant health |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5635080B2 (en) | 2014-12-03 |
| CN102427723B (en) | 2014-12-31 |
| JP2012526813A (en) | 2012-11-01 |
| KR101696208B1 (en) | 2017-01-13 |
| KR20140014391A (en) | 2014-02-06 |
| EP2429287A1 (en) | 2012-03-21 |
| EP2429287A4 (en) | 2014-12-24 |
| CN102427723A (en) | 2012-04-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2010132169A1 (en) | Fungicidal active compound compositions for turf treatment | |
| US10231456B2 (en) | Methods and compositions for controlling weed infestation and improving grass quality | |
| US20230270113A1 (en) | Fungicidal mixtures | |
| US20120035221A1 (en) | Method of Fungal Pathogen Control in Grass or Turf | |
| EP2154959B1 (en) | Fungicide active ingredient combinations | |
| KR102334544B1 (en) | Active compound combinations | |
| EP2154968A2 (en) | Fungicidal substance combinations | |
| ES2320007T3 (en) | PROCEDURE TO IMPROVE THE QUALITY OF HERBS FOR LAWN. | |
| HU189691B (en) | Synergistic fungicide composition containing thiazolil and dithiocarbamate derivatives or tetrachloro-isophtalonitril or sulfamide derivatives or /dichloro-phenyl/-imide derivatives as active agents | |
| US8377850B2 (en) | Fungicidal compositions for turf treatment and improvement | |
| US20100291229A1 (en) | Fungicidal compositions and methods of enhancing plants such as turfgrass | |
| US8119564B2 (en) | Herbicidal control using sulfentrazone and metsulfuron | |
| US20130324582A1 (en) | Methods and compositions for reducing fungal infestation and improving grass quality | |
| US8119565B2 (en) | Herbicidal control using sulfentrazone and dithiopyr | |
| US20110250288A1 (en) | Fungicidal compositions and methods of enhancing turfgrass | |
| US20140005246A1 (en) | Use of dithiine-tetracarboximides as bird repellent | |
| CA2698279A1 (en) | Fungidical compositions and methods of enhancing turfgrass | |
| CA2703679A1 (en) | Fungicidal compositions and methods of enhancing plants such as turfgrass | |
| WO2015068158A1 (en) | Synergistic fungicidal composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080020663.X Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10775231 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2010775231 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010775231 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2011/011930 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012510815 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20117029557 Country of ref document: KR Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: PI1010805 Country of ref document: BR |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01E Ref document number: PI1010805 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: PI1010805 Country of ref document: BR Kind code of ref document: A2 Effective date: 20111111 |