WO2009133455A2 - Pharmaceutical composition of clodipogrel - Google Patents
Pharmaceutical composition of clodipogrel Download PDFInfo
- Publication number
- WO2009133455A2 WO2009133455A2 PCT/IB2009/005455 IB2009005455W WO2009133455A2 WO 2009133455 A2 WO2009133455 A2 WO 2009133455A2 IB 2009005455 W IB2009005455 W IB 2009005455W WO 2009133455 A2 WO2009133455 A2 WO 2009133455A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- clopidogrel
- aqueous injection
- formulation
- pharmaceutical aqueous
- injection formulation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
- A61K9/1075—Microemulsions or submicron emulsions; Preconcentrates or solids thereof; Micelles, e.g. made of phospholipids or block copolymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/4365—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system having sulfur as a ring hetero atom, e.g. ticlopidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/16—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing nitrogen, e.g. nitro-, nitroso-, azo-compounds, nitriles, cyanates
- A61K47/18—Amines; Amides; Ureas; Quaternary ammonium compounds; Amino acids; Oligopeptides having up to five amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/34—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyesters, polyamino acids, polysiloxanes, polyphosphazines, copolymers of polyalkylene glycol or poloxamers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/02—Inorganic compounds
Definitions
- the present invention relates to a pharmaceutical composition of clopidogrel or its pharmaceutically acceptable salts, particularly injectable pharmaceutical composition.
- the present invention also relates to an injectable formulation and processes for preparation of an injectable formulation of clopidogrel besylate.
- Clopidogrel is an inhibitor of ADP -induced platelet aggregation acting by direct inhibition of adenosine diphosphate (ADP) binding to its receptor and of the subsequent ADP-mediated activation of the glycoprotein GPIIb/IIIa complex. Chemically it is (+)- (S)-methyl 2-(2-chlorophenyl)-
- Clopidogrel is a platelet aggregation inhibitor that has selective irreversible inhibition of adenosine diphosphate (ADP)-induced platelet aggregation, acting by direct inhibition of adenosine diphosphate (ADP) binding to its receptor and of the subsequent ADP-mediated activation of the glycoprotein GPHb/IIIa complex.; with no significant effect on thromboxane A2 or prostacyclin synthesis, or phospholipase A activity.
- Clopidogrel is indicated for reduction of atherothrombotic events like recent myocardial infarction, recent stroke or established peripheral arterial disease, acute coronary syndrome.
- Clopidogrel is official in USP. The official monograph mentions the limits on related compounds A, B and C, the limits of which are mentioned in Table Ia. Throughout the specification, related compound A is mentioned as impurity A 5 related compound B is mentioned as impurity B and related compound C is mentioned as impurity C.
- Clopidogrel is available as its bisulphate salt in the form of immediate release tablets equivalent to 75 and 100 mg base. It is marketed under the brand name i "Plavix®" by Sanofi Aventis US. Typically the oral formulation takes more than 2 hours to show its pharmacological effect.
- Intravenous injections are known to provide the activity immediately.
- the parenteral route of administration has advantage over oral administration for immediate release, as there is no lag time corresponding to the drug reaching sufficient concentration in the systemic circulation to start surgery in cases of coronary intervention. Coronary intervention is a medical emergency, during which the rapid achievement of therapeutic drug concentrations in the blood and rapid onset of action is a priority and to achieve the same, intravenous administration is the best preferred route.
- Clopidogrel is a new platelet antiaggregatory, particularly described in the patent EP 099802, EP281459 and US4847265. These patents describe a composition of clopidogrel or its pharmaceutically acceptable salts for parenteral administration, in the form of fluid Injection. This type of formulation containing a salt solution of clopidogrel in an isotonic solvent is difficult to apply. Indeed, this type of solution with a pH of less than 2 makes it very painful injection.
- EP 1105102 describes a injectable aqueous solutions containing a salt of clopidogrel, pluronic F68, a basic pH modifier and solutol HS 15, a lyophilized formulations containing these and kits containing such lyophilized compositions in two parts.
- the application also discloses that it is necessary to achieve a lyophilisate since the salts of clopidogrel are unstable in aqueous solution and there is sharp deterioration.
- the formulation of the present invention is stable in the form of ready-to-use aqueous injection although not lyophilized.
- the various variables which affect the formulation are mainly the pH, the quantity of salts present, the type and quantity of excipients in the formulation, the type of cryoprotection chosen, as well as the temperatures, pressure and time chosen for the freezing, sublimation and drying operations and also these different variables influence the physical state of the freeze- dried product obtained, namely: vitreous amorphous, soft amorphous, crystalline or a combination of these states.
- the present invention seeks to overcome these problems.
- pH of about 3-7 is ideal in order to have patient compatability.
- Clopidogrel and its salts are soluble only at lower pH. Also, clopidogrel being lipophilic has a poor water solubility at the pH intended for injection formulation.
- the present invention overcomes these shortcomings and provides a ready-to-use stable aqueous clopidogrel composition that could be administered.
- an impediment to the preparation of an aqueous clopidogrel composition is that such compositions may not be adequately stable at ambient temperatures.
- the principal object of the present invention is to provide stable ready-to-use aqueous injection formulation comprising clopidogrel besylate.
- Another object of the present invention is to provide ready-to-use aqueous injection formulation comprising clopidogrel besylate.
- One more object of the present invention is to provide the stable pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a ready-to-use aqueous clopidogrel composition free of co solvent.
- Another object of present invention describes the pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a stable ready-to-use aqueous clopidogrel composition with enhanced stability at elevated temperatures such that accidental exposure of the composition to elevated temperatures for a brief time would be less likely to result in unacceptable levels of degradation.
- Another object of the present invention describes process for preparation of the pharmaceutical aqueous injection formulation of clopidogrel or its salts.
- aqueous compositions containing clopidogrel having enhanced stability at room temperature as well as upon a transient exposure to elevated temperatures.
- Clopidogrel is official in USP. The official monograph mentions the limits on related compounds A, B and C, the limits of which are mentioned in Table Ia. Throughout the specification, related compound A is mentioned as impurity A, related compound B is mentioned as impurity B and related compound C is mentioned as impurity C.
- a stable pharmaceutical aqueous injection formulation of clopidogrel besylate is one that contains not more than 1.5 % of impurity C and not more than 1.5 % of total impurity (excluding impurity C), when stored at 2 to 8 0 C for atleast 18 months.
- a physico-chemical solubilization technique such as miceller solubilization by means of surface-active agents, i.e., the use of surfactant micelles to solubilize and transport the therapeutic agent is an important method of increasing solubility of a practically insoluble ingredient.
- Micelles are agglomerates of colloidal dimensions formed by amphiphilic compounds under certain conditions. Micelles, and pharmaceutical compositions containing micelles, have been extensively studied and are described in detail in the literature. In aqueous solution, micelles can incorporate lipophillic therapeutic agents in the hydrocarbon core of the micelle, or can entangle the agents at various positions within the micelle walls.
- Solubilization of hydrophobic drugs with low aqueous solubility has been a major area of interest in recent years.
- Various solubilization techniques involve usage of cosolvents and surfactants along with pH adjustments.
- the present invention includes an approach of miceller system, uses surfactant micelles to solubilize and transport the therapeutic agent.
- Clopidogrel base or salt forms mixed with a surfactant under stirring gets creamy dispersion also called creamy nano dispersion.
- This dispersion is diluted with a buffer to attain a pH of 3 to 8 and stabilizing the dispersion with a stabilizing agent.
- the colloidal dispersion is diluted to reach the required concentration with water of injection.
- This diluted colloidal dispersion with appropriate pH is then packaged into 10 ml vials.
- the solution in the vial can be directly administered as an injection or can be diluted with IV infusion prior to administration.
- This mixture when diluted with intravenous infusion prior to administration forms a clear surfactant-drug concentrate that converts into nano size surfactant micelles, in which Clopidogrel base or salts is entrapped.
- the dose of the injection formulation of the present invention can be from about 3 mg/ml to about 100 mg/ml In one of the preferred embodiment, the dose of the injection formulation is
- the formulation of the present invention is stabilized by addition of sodium EDTA as stabilizing agent.
- the amount of stabilizing agent used in the formulation is in the range from 0.01 to 0.5 %w/w. Preferably, the amount of stabilizing agent used is 0.13 %w/w.
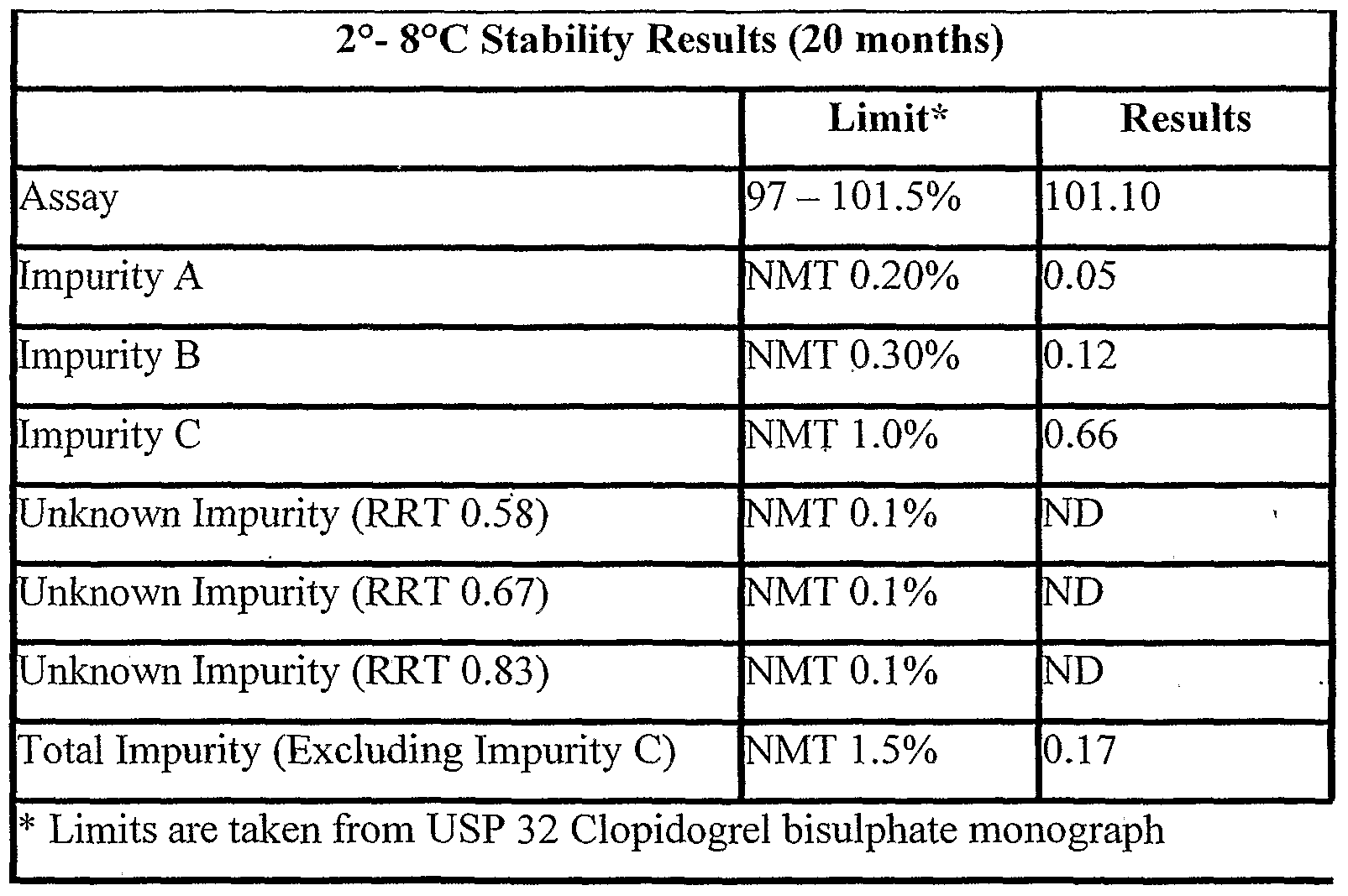
- the real time stability data in the temperature range of 2 to 8 0 C for 20 months for the formulation prepared in accordance with example 1 is given in Table 1 a.
- the real time stability data shows that the impurities A, B and C are within the prescribed USP limits for clopidogrel bisulphate.
- the formulation of the present invention had a limit of 0.66 % of impurity C, which is not more than 1.0 % of the USP limit for clopidogrel bisulphate.
- the accelerated stability studies conducted at 40 °C/75 %RH and 50 °C/80 %RH of the formulation prepared in accordance with example 1 is provided. See Table Ib.
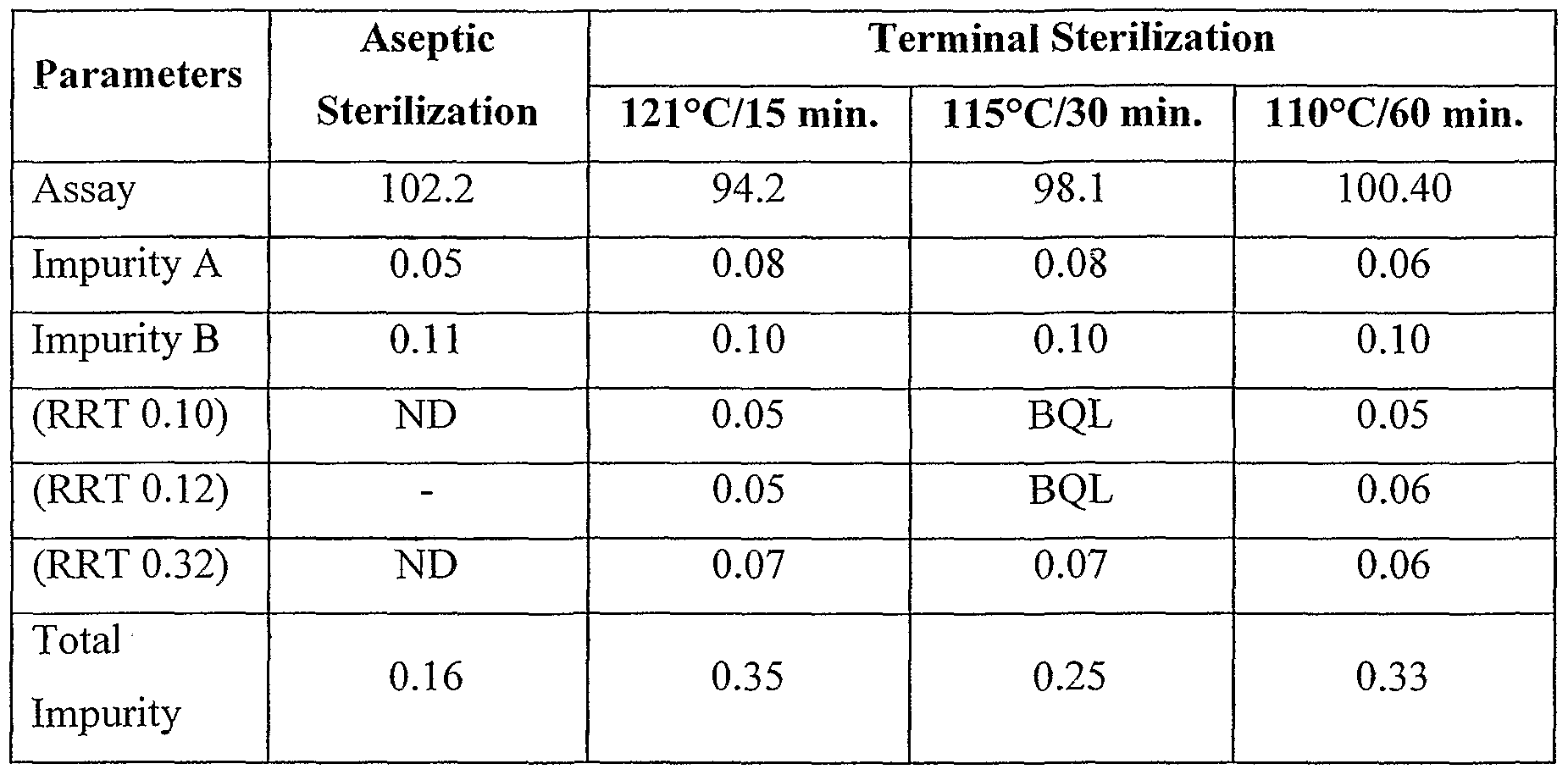
- the formulation of the present invention cab be sterilized either aseptically or terminally.
- the effect of aseptic as well as terminal sterilization on impurities A and B is provided. See Table 2.
- Clopidogrel or its salts in particular but not limited to Clopidogrel Besylate is highly acidic and thus a pharmaceutically acceptable buffer system is to be used so that the pH is attained between 3-8.
- Second aspect of the present invention describes the pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a ready-to-use aqueous clopidogrel composition in the pH range of 3 to 8.
- the pH of the formulation is maintained between about pH 5 to 6, preferably 5.7, so that ease of intravenous administration is achieved, increasing patient compliance.
- Fourth aspect of the present invention also describes process for preparation of the pharmaceutical aqueous injection formulation of clopidogrel or its salts.
- Fifth aspect of the present invention also describes preparation of an intravenous formulation of Clopidogrel and its pharmaceutically acceptable salts in order to provide a fast acting treatment in cardiovascular related surgical interventions.
- Polyethyleneglycol 660 12-hydroxy stereate (Solutol HS 15) is the surfactant used in the present invention.
- the formulation contains water as a vehicle and thus issues for the intravenous administration are taken care of.
- stabilizing agents for injection/solution include EDTA(ethylene diamine tetraacetic acid) , para-hydroxybenzoic acid ester derivatives, alcohol, benzalkonium chloride, phenol derivatives, thiomerosal, acetic anhydride, ascorbic acid , sorbic acid, boric acid, adipic acid, sodium carboxylate, lauryl sulfate, retinol, tocopherol or sodium ascorbate, sulfite compounds, amino acid such as L-cysteine, thiodipropionic acid, thiolactic acid, and monothioglycerol, sulfurous acid, sulfite, ascorbate, L-cysteine, and tocopherol.
- EDTA is the stabilizer used in the present invention
- compositions of the present invention include borate buffers, citrate buffers, phosphate buffers, citric acid/phosphate buffers, carbonate/carbonic acid buffers, succinate/succinic acid buffers, and tris(hydroxymethyl)aminomethane/hydrochloric acid buffers.
- Pharmaceutically acceptable carbonate buffers include CaCO 3 , and Na 2 CO 3 .
- Pharmaceutically acceptable phosphate buffers include Na 3 PO 4 , Na 2 HPO 4 , NaH 2 PO 4 , K 3 PO 4 , K 2 HPO 4 , and KH 2 PO 4 or mixture thereof.
- dibasic sodium phosphate and monobasic potassium phosphate is used as buffers in the present invention.
- Clopidogrel besylate is added to Solutol HS 15 /T ween 80 Super refined and mixed till creamy nano dispersion is formed.
- Other ingredients like dibasic sodium phosphate, monobasic potassium phosphate, sodium metabisulphite, EDTA Sodium are dissolved in small amount of water and added to drug mixture with continuous stirring.
- Preservatives like methyl paraben, propyl paraben are dissolved in hot water and after cooling to room temperature, added to drug mixture. Finally, the volume is made up with water for injection.
- Table Ia Table Ia:
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biophysics (AREA)
- Dispersion Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Molecular Biology (AREA)
- Inorganic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The present invention discloses a pharmaceutical composition of clopidogrel or its pharmaceutically acceptable salts, particularly injectable pharmaceutical composition.
Description
PHARMACEUTICAL COMPOSITION OF CLOPIDOGREL FILED OF THE INVENTION
The present invention relates to a pharmaceutical composition of clopidogrel or its pharmaceutically acceptable salts, particularly injectable pharmaceutical composition. The present invention also relates to an injectable formulation and processes for preparation of an injectable formulation of clopidogrel besylate.
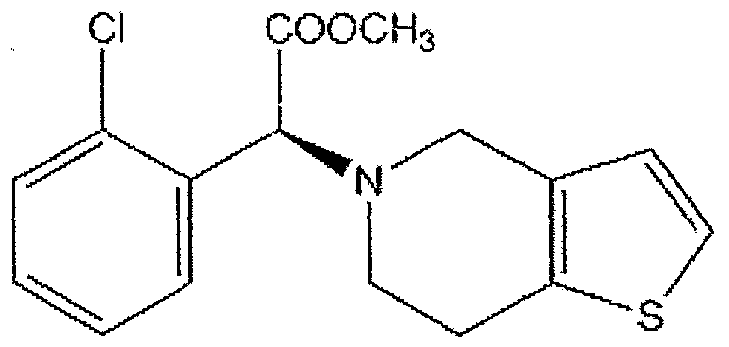
Clopidogrel is an inhibitor of ADP -induced platelet aggregation acting by direct inhibition of adenosine diphosphate (ADP) binding to its receptor and of the subsequent ADP-mediated activation of the glycoprotein GPIIb/IIIa complex. Chemically it is (+)- (S)-methyl 2-(2-chlorophenyl)-
2-(6,7-dihydrothieπo[3,2-c]pyridin-5(4H)-yl)acetate. The structural formula is as follows:
BACKGROUND OF THE INVENTION Clopidogrel is a platelet aggregation inhibitor that has selective irreversible inhibition of adenosine diphosphate (ADP)-induced platelet aggregation, acting by direct inhibition of adenosine diphosphate (ADP) binding to its receptor and of the subsequent ADP-mediated activation of the glycoprotein GPHb/IIIa complex.; with no significant effect on thromboxane A2 or prostacyclin synthesis, or phospholipase A activity. Clopidogrel is indicated for reduction of atherothrombotic events like recent myocardial infarction, recent stroke or established peripheral arterial disease, acute coronary syndrome.
Clopidogrel is official in USP. The official monograph mentions the limits on related compounds A, B and C, the limits of which are mentioned in Table Ia. Throughout the specification, related compound A is mentioned as impurity A5 related compound B is mentioned as impurity B and related compound C is mentioned as impurity C.
Clopidogrel is available as its bisulphate salt in the form of immediate release tablets equivalent to 75 and 100 mg base. It is marketed under the brand name i
"Plavix®" by Sanofi Aventis US. Typically the oral formulation takes more than 2 hours to show its pharmacological effect. Intravenous injections are known to provide the activity immediately. The parenteral route of administration has advantage over oral administration for immediate release, as there is no lag time corresponding to the drug reaching sufficient concentration in the systemic circulation to start surgery in cases of coronary intervention. Coronary intervention is a medical emergency, during which the rapid achievement of therapeutic drug concentrations in the blood and rapid onset of action is a priority and to achieve the same, intravenous administration is the best preferred route. The medication is used particularly for the reduction of accidents related to atherosclerosis (myocardial infarction, stroke, arthritis, vascular death) in patients who have had a recent myocardial infarction, stroke or with a recent arterial obliterans proved in the lower limbs. There is need of injection dosage form for patients who are in urgency for medication and for whom oral administration is difficult. Intravenous injection should be aqueous based so that biocompatibility is achieved. There is a problem in making a stable aqueous liquid product for Clopidogrel. There is no injection formulation available in the market for Clopidogrel as of today. The aqueous solution has only a limited shelf-life at room temperature. Clopidogrel aqueous solution can be administered parenterally at a maximum concentration of not more than 150 mg/mL.
Clopidogrel is a new platelet antiaggregatory, particularly described in the patent EP 099802, EP281459 and US4847265. These patents describe a composition of clopidogrel or its pharmaceutically acceptable salts for parenteral administration, in the form of fluid Injection. This type of formulation containing a salt solution of clopidogrel in an isotonic solvent is difficult to apply. Indeed, this type of solution with a pH of less than 2 makes it very painful injection.
US6284277 describes a pharmaceutical formulation provided in the form of a freeze-dried/lyophilized product of clopidogrel or its pharmaceutically acceptable salts containing mannitol and alanine. The process of production is costly and critical to achieve.
WO2008134600 describes a pharmaceutical composition containing clopidogrel. present as a free base or pharmaceutically acceptable salt thereof, sulfoalkylether cyclodextrin. The application also discloses in table 14c and 14d that solutol shows a synergistic effect with cyclodextrin in increasing the solubility at
concentrations of between 1 to 5 %w/v. Use of cyclodextrin is essential for the invention that is disclosed in this application.
EP 1105102 describes a injectable aqueous solutions containing a salt of clopidogrel, pluronic F68, a basic pH modifier and solutol HS 15, a lyophilized formulations containing these and kits containing such lyophilized compositions in two parts. The application also discloses that it is necessary to achieve a lyophilisate since the salts of clopidogrel are unstable in aqueous solution and there is sharp deterioration.
The formulation of the present invention is stable in the form of ready-to-use aqueous injection although not lyophilized. In Freeze-Drying and lyophilization the various variables which affect the formulation are mainly the pH, the quantity of salts present, the type and quantity of excipients in the formulation, the type of cryoprotection chosen, as well as the temperatures, pressure and time chosen for the freezing, sublimation and drying operations and also these different variables influence the physical state of the freeze- dried product obtained, namely: vitreous amorphous, soft amorphous, crystalline or a combination of these states. The present invention seeks to overcome these problems.
It is desirable to provide pharmaceutical formulations of (clopidogrel) lipophillic therapeutic agents wherein poor water-solubility of these lipophillic agents often results in major difficulties in formulation, particularly when easily sterilizable and administrable homogeneous aqueous solutions are needed.
For injection formulations, pH of about 3-7 is ideal in order to have patient compatability. Clopidogrel and its salts are soluble only at lower pH. Also, clopidogrel being lipophilic has a poor water solubility at the pH intended for injection formulation.
The present invention overcomes these shortcomings and provides a ready-to-use stable aqueous clopidogrel composition that could be administered. However, an impediment to the preparation of an aqueous clopidogrel composition is that such compositions may not be adequately stable at ambient temperatures.
The formulation of the present invention is ready-to-use and should be stored in a temperature of about 2 to 8 0C. SUMMARY OF THE INVENTION
The principal object of the present invention is to provide stable ready-to-use aqueous injection formulation comprising clopidogrel besylate.
Another object of the present invention is to provide ready-to-use aqueous injection formulation comprising clopidogrel besylate.
One more object of the present invention is to provide the stable pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a ready-to-use aqueous clopidogrel composition free of co solvent.
Another object of present invention describes the pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a stable ready-to-use aqueous clopidogrel composition with enhanced stability at elevated temperatures such that accidental exposure of the composition to elevated temperatures for a brief time would be less likely to result in unacceptable levels of degradation. Another object of the present invention describes process for preparation of the pharmaceutical aqueous injection formulation of clopidogrel or its salts.
Another object of the present invention describes to prepare an intravenous formulation of Clopidogrel and its pharmaceutically acceptable salts in order to provide a fast acting treatment in reduction of atherothrombotic events like recent myocardial infarction, recent stroke or established peripheral arterial disease, acute coronary syndrome.
DETAILED DESCRIPTION OF THE TABLES Table Ia: Real time stability data Table Ib: Accelerated stability data Table 2: Effect of sterilization DETAILED DESCRIPTION OF THE INVENTION
This invention relates to pharmaceutical formulations of Clopidogrel and or its pharmaceutically acceptable salts in particular but not limited to Clopidogrel Besylate in which such agents are solubilized in largely aqueous vehicles for use of such formulations. The formulations are stable in aqueous-based vehicles so that patient compliance can be achieved, and have therapeutically and commercially useful concentrations of active ingredient.
In the present invention, the inventors have found, surprisingly, that it may be possible to prepare aqueous compositions containing clopidogrel having enhanced stability at room temperature as well as upon a transient exposure to elevated temperatures.
Clopidogrel is official in USP. The official monograph mentions the limits on related compounds A, B and C, the limits of which are mentioned in Table Ia. Throughout the specification, related compound A is mentioned as impurity A, related
compound B is mentioned as impurity B and related compound C is mentioned as impurity C.
For the purpose of this invention, a stable pharmaceutical aqueous injection formulation of clopidogrel besylate is one that contains not more than 1.5 % of impurity C and not more than 1.5 % of total impurity (excluding impurity C), when stored at 2 to 8 0C for atleast 18 months.
The poor water-solubility of lipophillic agents like clopidogrel often results in major difficulties in formulation, particularly when easily sterilizable and administrable homogeneous aqueous solutions are needed. A physico-chemical solubilization technique such as miceller solubilization by means of surface-active agents, i.e., the use of surfactant micelles to solubilize and transport the therapeutic agent is an important method of increasing solubility of a practically insoluble ingredient. Micelles are agglomerates of colloidal dimensions formed by amphiphilic compounds under certain conditions. Micelles, and pharmaceutical compositions containing micelles, have been extensively studied and are described in detail in the literature. In aqueous solution, micelles can incorporate lipophillic therapeutic agents in the hydrocarbon core of the micelle, or can entangle the agents at various positions within the micelle walls.
Solubilization of hydrophobic drugs with low aqueous solubility has been a major area of interest in recent years. Various solubilization techniques involve usage of cosolvents and surfactants along with pH adjustments. Applications of cyclodextrins
(CDs) and microemulsions have also drawn attention in the field of solubilization techniques. The present invention does not include usage of co solvents.
First aspect of the present invention describes the pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a ready-to-use aqueous clopidogrel composition free of co solvent.
The present invention includes an approach of miceller system, uses surfactant micelles to solubilize and transport the therapeutic agent. Clopidogrel base or salt forms mixed with a surfactant under stirring gets creamy dispersion also called creamy nano dispersion. This dispersion is diluted with a buffer to attain a pH of 3 to 8 and stabilizing the dispersion with a stabilizing agent. The colloidal dispersion is diluted to reach the required concentration with water of injection. This diluted colloidal dispersion with appropriate pH is then packaged into 10 ml vials. The solution in the vial can be directly administered as an injection or can be diluted with IV infusion prior to administration. This mixture when diluted with intravenous infusion prior to
administration forms a clear surfactant-drug concentrate that converts into nano size surfactant micelles, in which Clopidogrel base or salts is entrapped.
The dose of the injection formulation of the present invention can be from about 3 mg/ml to about 100 mg/ml In one of the preferred embodiment, the dose of the injection formulation is
15mg/ml.
In one embodiment of this aspect, the formulation of the present invention is stabilized by addition of sodium EDTA as stabilizing agent. The amount of stabilizing agent used in the formulation is in the range from 0.01 to 0.5 %w/w. Preferably, the amount of stabilizing agent used is 0.13 %w/w. The real time stability data in the temperature range of 2 to 8 0C for 20 months for the formulation prepared in accordance with example 1 is given in Table 1 a.
The real time stability data shows that the impurities A, B and C are within the prescribed USP limits for clopidogrel bisulphate. In another embodiment, the formulation of the present invention had a limit of 0.66 % of impurity C, which is not more than 1.0 % of the USP limit for clopidogrel bisulphate.
In yet another embodiment, the accelerated stability studies conducted at 40 °C/75 %RH and 50 °C/80 %RH of the formulation prepared in accordance with example 1 is provided. See Table Ib. The accelerated stability studies show that the impurities A and B are within the prescribed USP limits for clopidogrel bisulphate. The formulation of the present invention cab be sterilized either aseptically or terminally. The effect of aseptic as well as terminal sterilization on impurities A and B is provided. See Table 2. Clopidogrel or its salts in particular but not limited to Clopidogrel Besylate is highly acidic and thus a pharmaceutically acceptable buffer system is to be used so that the pH is attained between 3-8. Being an intravenous injection, the patient compliance is important, thus pH of the formulation is to be maintained throughout the shelf-life so that less pain is caused to the patient while injecting the aqueous solution of the drug. The solution pH is controlled by either the salt form of the drug, strong acids/bases such as hydrochloric acid or sodium hydroxide, or a buffer such as glycine, citrate, acetate, histidine, phosphate, tris(hydroxymethy)aminomethane (TRIS), or carbonate in which case the buffer concentration must be high enough to maintain the desired pH but must be balanced by in vivo tolerability considerations.
Second aspect of the present invention describes the pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a ready-to-use aqueous clopidogrel composition in the pH range of 3 to 8.
In one embodiment of this aspect, the pH of the formulation is maintained between about pH 5 to 6, preferably 5.7, so that ease of intravenous administration is achieved, increasing patient compliance.
Third aspect of the present invention describes the pharmaceutical aqueous injection formulation of clopidogrel or its salts, wherein the formulation provides a ready-to-use aqueous clopidogrel composition with enhanced stability at elevated temperatures such that accidental exposure of the composition to elevated temperatures for a brief time would be less likely to result in unacceptable levels of degradation.
Fourth aspect of the present invention also describes process for preparation of the pharmaceutical aqueous injection formulation of clopidogrel or its salts.
Fifth aspect of the present invention also describes preparation of an intravenous formulation of Clopidogrel and its pharmaceutically acceptable salts in order to provide a fast acting treatment in cardiovascular related surgical interventions.
Surfactants that may be used with the present formulation are but not limited to sorbitan monooleate (Span 80), Polyoxyethylene 20 sorbitan monooleate (tween 80), d- α-tocopheryl polyethylene glycol 1000 succinate (Vitamin E TPGS), Polyethyleneglycol 660 12-hydroxy stereate (Solutol HS 15), Poly(ethyleneoxide)/Poly-(Propyleneoxide)/poly (ethyleneoxide) triblock copolymers (poloxamers, pluronics), Poloxamer 407 (pluronic F- 127), Mono and di fatty acid esters of PEG 300, Monostearic acid ester of PEG 400 or 1750 (polyoxyl 40 stearate), propylene glycol dicaprylate/dicaprate(Miglyol 840), Medium chain mono and diglycerides (capmul, imwitor 742), Medium chain triglycerides (Caprylic and capric triglycerides, Labrafac, Miglyol 810, 812, Crodamol GTCC-PN, Softison 378), Long chain monoglycerides (Glyceryl monooleate, Glyceryl monolinoleate), Polyoxyethylene castor oil derivatives (Cremophor EL and Cremophor RH 401), Polyethylene glycol and glycerol ethoxylates, Mono-, di-; and triglycerides and mono- and di -fatty acid esters of PEG (also contains glycerol and PEG) (Softigen 767, Labrosol, Labrafil Ml 944CS, Labrafil M-2125CS, Gelucire 44/14), all types of Phospholipids.
In one embodiment, Polyethyleneglycol 660 12-hydroxy stereate (Solutol HS 15) is the surfactant used in the present invention.
The formulation contains water as a vehicle and thus issues for the intravenous administration are taken care of.
The formulation has been stabilized by using stabilizing agents. In general, examples of stabilizing agents for injection/solution include EDTA(ethylene diamine tetraacetic acid) , para-hydroxybenzoic acid ester derivatives, alcohol, benzalkonium chloride, phenol derivatives, thiomerosal, acetic anhydride, ascorbic acid , sorbic acid, boric acid, adipic acid, sodium carboxylate, lauryl sulfate, retinol, tocopherol or sodium ascorbate, sulfite compounds, amino acid such as L-cysteine, thiodipropionic acid, thiolactic acid, and monothioglycerol, sulfurous acid, sulfite, ascorbate, L-cysteine, and tocopherol. N-acetyl amino acid , tocopherol, sodium formaldehyde, or tertiary-butyl hydroquinone, sodium sulfite, sodium phosphate or mixture thereof.
In one embodiment, EDTA is the stabilizer used in the present invention
Pharmaceutically acceptable buffers which may be used in the compositions of the present invention include borate buffers, citrate buffers, phosphate buffers, citric acid/phosphate buffers, carbonate/carbonic acid buffers, succinate/succinic acid buffers, and tris(hydroxymethyl)aminomethane/hydrochloric acid buffers. Pharmaceutically acceptable carbonate buffers include CaCO3, and Na2CO3. Pharmaceutically acceptable phosphate buffers include Na3PO4, Na2HPO4, NaH2PO4, K3PO4, K2HPO4, and KH2PO4 or mixture thereof.
In one embodiment, dibasic sodium phosphate and monobasic potassium phosphate is used as buffers in the present invention.
The invention described here is demonstrated in examples illustrated below. These examples are provided as illustration only and therefore should not be construed as limitation of the scope of invention.
EXAMPLES Example I
Brief Manufacturing process:
Clopidogrel besylate is added to Solutol HS 15 /T ween 80 Super refined and mixed till creamy nano dispersion is formed. Other ingredients like dibasic sodium phosphate, monobasic potassium phosphate, sodium metabisulphite, EDTA Sodium are dissolved in small amount of water and added to drug mixture with continuous stirring. Preservatives like methyl paraben, propyl paraben are dissolved in hot water and after cooling to room temperature, added to drug mixture. Finally, the volume is made up with water for injection. Table Ia:
Table Ib:
Claims
1. A ready-to-use stable pharmaceutical aqueous injection formulation comprising clopidogrel or salt thereof.
2. Pharmaceutical aqueous injection formulation comprising of clopidogrel besylate in an amount from about 1 mg/ml to about 225 mg/ml.
3. A pharmaceutical aqueous injection formulation according to claim 1 or 2, further comprising a solubilizer, buffering agent and stabilizing agent.
4. A pharmaceutical aqueous injection formulation according to claim 3, wherein the stabilizing agent used is sodium EDTA. 5. A pharmaceutical aqueous injection formulation according to claim 4, wherein the amount of sodium EDTA used is 0.01 to 0.
5 %w/w.
6. A pharmaceutical aqueous injection formulation according to claim 3, wherein the solubilizer is Polyethylene glycol 660 12-hydroxy stereate.
7. A pharmaceutical aqueous injection formulation according to claim 1 or 2, wherein pH of the formulation is between about pH 3 to 8.
8. A stable pharmaceutical aqueous injection formulation of clopidogrel besylate, such that the formulation contains not more than 1.5 % of impurity C and not more than 1.5 % of total impurity (excluding impurity C), when stored at 2 to 8 0C for atleast 18 months.
9. A stable pharmaceutical ready-to-use aqueous injection formulation comprising
(a) clopidogrel besylate 0.5 to 15 %w/w, (b) sodium EDTA is 0.01 to 0.5 %w/w as stabilizing agent, (c) phosphate buffer 0.1 to 1.0 %w/w as buffering agent, the pH of the final formulation being from 5 to 6 and the formulation is devoid of a co solvent.
10. Process for the preparation of pharmaceutical aqueous injection formulation comprising : i. mixing clopidogrel or its salt with solubilizer under stirring to get nano dispersion; ii. diluting the dispersion of step (i) with a buffer to attain a pH of 3 to 8; iii. adding to the dispersion of step (ii) a stabilizing agent; and iv. diluting the dispersion of step (iii) to reach the required concentration with water for injection.
11. Process for the preparation of pharmaceutical aqueous injection formulation according to claim 10, wherein the clopidogrel salt is clopidogrel besylate, solubilizer used is polyethylene glycol 660 12-hydroxy stereate, buffering agent used is phosphate buffer and stabilizing agent used is sodium EDTA.
12. Use of stable pharmaceutical aqueous injection formulation according to claim 1 or 2 with pH maintained from 3 to 8 for the ease of intravenous administration and for better patient compliance to provide a fast acting treatment for reduction of atherothrombotic events like recent myocardial infarction, recent stroke or established peripheral arterial disease, acute coronary syndrome.
13. A pharmaceutical aqueous injection formulation comprising clopidogrel besylate according to any of the preceeding claims, such that the formulation is administered in a dose from 3 mg/ml to 100 mg/ml.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN956/MUM/2008 | 2008-05-01 | ||
| IN956MU2008 | 2008-05-01 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2009133455A2 true WO2009133455A2 (en) | 2009-11-05 |
| WO2009133455A3 WO2009133455A3 (en) | 2010-02-25 |
Family
ID=41255485
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2009/005455 Ceased WO2009133455A2 (en) | 2008-05-01 | 2009-05-01 | Pharmaceutical composition of clodipogrel |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2009133455A2 (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011101865A3 (en) * | 2010-02-19 | 2011-10-27 | Cadila Healthcare Limited | Stable pharmaceutical compositions of clopidogrel for parenteral delivery |
| CN114025746A (en) * | 2019-05-10 | 2022-02-08 | 福多兹制药公司 | Injectable polymer nanoparticle compositions of antithrombotic agents and methods thereof |
| WO2023012479A1 (en) | 2021-08-03 | 2023-02-09 | Liqmeds Worldwide Limited | An oral pharmaceutical solution of clopidogrel |
| CN117503720A (en) * | 2024-01-02 | 2024-02-06 | 济南舜景医药科技有限公司 | Clopidogrel bisulfate tablet and preparation method thereof |
| IL298129B1 (en) * | 2020-08-28 | 2024-07-01 | Cybin Uk Ltd | Injectable formulation comprising the fumarate salt of dimethyltryptamine or deuterated dimethyltryptamine |
| US12318477B2 (en) | 2021-11-18 | 2025-06-03 | Cybin Uk Ltd | Injectable and inhalable formulations |
| US12343327B2 (en) | 2020-08-28 | 2025-07-01 | Cybin Uk Ltd | Injectable formulations |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2782455B3 (en) * | 1998-08-20 | 2000-09-15 | Sanofi Sa | INJECTABLE PHARMACEUTICAL COMPOSITION BASED ON A PHARMACEUTICALLY ACCEPTABLE SALT OF CLOPIDOGREL OR TICLOPIDINE |
| WO2004074215A1 (en) * | 2003-02-03 | 2004-09-02 | Sunil Sadanand Nadkarni | Process for preparation of clopidogrel, its salts and pharmaceutical compositions |
| AU2006336417A1 (en) * | 2005-05-10 | 2007-08-02 | Elan Pharma International, Limited | Nanoparticulate clopidogrel formulations |
| CN100562309C (en) * | 2006-07-14 | 2009-11-25 | 北京赛科药业有限责任公司 | Injectable composition of clopidogrel and its salt and its preparation method |
| WO2008060934A2 (en) * | 2006-11-14 | 2008-05-22 | Acusphere, Inc. | Formulations of tetrahydropyridine antiplatelet agents for parenteral or oral administration |
| CA2685331C (en) * | 2007-04-27 | 2016-07-05 | Cydex Pharmaceuticals, Inc. | Formulations containing clopidogrel and sulfoalkyl ether cyclodextrin and methods of use |
-
2009
- 2009-05-01 WO PCT/IB2009/005455 patent/WO2009133455A2/en not_active Ceased
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011101865A3 (en) * | 2010-02-19 | 2011-10-27 | Cadila Healthcare Limited | Stable pharmaceutical compositions of clopidogrel for parenteral delivery |
| CN114025746A (en) * | 2019-05-10 | 2022-02-08 | 福多兹制药公司 | Injectable polymer nanoparticle compositions of antithrombotic agents and methods thereof |
| CN114025746B (en) * | 2019-05-10 | 2023-08-25 | 福多兹制药公司 | Injectable polymer nanoparticle compositions of antithrombotic agents and methods thereof |
| IL298129B1 (en) * | 2020-08-28 | 2024-07-01 | Cybin Uk Ltd | Injectable formulation comprising the fumarate salt of dimethyltryptamine or deuterated dimethyltryptamine |
| IL298129B2 (en) * | 2020-08-28 | 2024-11-01 | Cybin Uk Ltd | Injectable formulation comprising the fumarate salt of dimethyltryptamine or deuterated dimethyltryptamine |
| US12343327B2 (en) | 2020-08-28 | 2025-07-01 | Cybin Uk Ltd | Injectable formulations |
| WO2023012479A1 (en) | 2021-08-03 | 2023-02-09 | Liqmeds Worldwide Limited | An oral pharmaceutical solution of clopidogrel |
| US12318477B2 (en) | 2021-11-18 | 2025-06-03 | Cybin Uk Ltd | Injectable and inhalable formulations |
| CN117503720A (en) * | 2024-01-02 | 2024-02-06 | 济南舜景医药科技有限公司 | Clopidogrel bisulfate tablet and preparation method thereof |
| CN117503720B (en) * | 2024-01-02 | 2024-03-15 | 济南舜景医药科技有限公司 | Clopidogrel bisulfate tablet and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2009133455A3 (en) | 2010-02-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11433062B2 (en) | Stable nimodipine parenteral formulation | |
| WO2009133455A2 (en) | Pharmaceutical composition of clodipogrel | |
| AU2015256331B2 (en) | Formulations of Cyclophosphamide liquid concentrate | |
| NZ538255A (en) | Aqueous 2,6-diisopropylphenol pharmaceutical compositions | |
| ES2782106T3 (en) | Improved formulations of levosimendan for intravenous administration as an infusion or injection and as an infusion concentrate | |
| US20170014343A1 (en) | Stable pharmaceutical composition of clopidogrel free base for oral and parenteral delivery | |
| US8481589B2 (en) | Taxoid-based compositions | |
| US10799486B2 (en) | Stable nimodipine parenteral formulation | |
| US10278946B2 (en) | Liquid formulation of cabazitaxel | |
| US20200163947A1 (en) | Methods of treatment using nimodipine parenteral formulations | |
| CA3123002A1 (en) | Stable formulations of anesthetics and associated dosage forms | |
| US20200397769A1 (en) | Stable nimodipine parenteral formulation | |
| US12414943B2 (en) | Nimodipine parenteral administration | |
| WO2019006134A1 (en) | Stable nimodipine parenteral formulation | |
| AU2024262981A1 (en) | Injectable compositions of metolazone | |
| HK40077176A (en) | Nimodipine formulation and method of treating conditions | |
| WO2011101865A2 (en) | Stable pharmaceutical compositions of clopidogrel for parenteral delivery |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| DPE2 | Request for preliminary examination filed before expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2325/MUMNP/2010 Country of ref document: IN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 09738468 Country of ref document: EP Kind code of ref document: A2 |