WO2009030871A1 - Pyrrolopyrimidine derivatives having hsp90 inhibitory activity - Google Patents
Pyrrolopyrimidine derivatives having hsp90 inhibitory activity Download PDFInfo
- Publication number
- WO2009030871A1 WO2009030871A1 PCT/GB2007/003353 GB2007003353W WO2009030871A1 WO 2009030871 A1 WO2009030871 A1 WO 2009030871A1 GB 2007003353 W GB2007003353 W GB 2007003353W WO 2009030871 A1 WO2009030871 A1 WO 2009030871A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cyano
- chloro
- phenyl
- pyrrolo
- methoxy
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- HWVWUTPVXCDQKA-UHFFFAOYSA-N CCCOC[n]1c2nc(SCC(O)=O)nc(-c(cc(c(C#N)c3)OC)c3Cl)c2c(C#N)c1 Chemical compound CCCOC[n]1c2nc(SCC(O)=O)nc(-c(cc(c(C#N)c3)OC)c3Cl)c2c(C#N)c1 HWVWUTPVXCDQKA-UHFFFAOYSA-N 0.000 description 1
- LODSQJJTKFBOGC-UHFFFAOYSA-N CCOC(CSc1nc(-c(cc(c(C#N)c2)OC)c2Cl)c(c(C#N)c[n]2COCC[Si+](C)(C)C)c2n1)=O Chemical compound CCOC(CSc1nc(-c(cc(c(C#N)c2)OC)c2Cl)c(c(C#N)c[n]2COCC[Si+](C)(C)C)c2n1)=O LODSQJJTKFBOGC-UHFFFAOYSA-N 0.000 description 1
- MKNBXEANHDZWIH-UHFFFAOYSA-N CNC(CSc1nc(-c(cc(c(OC)c2)OCCOC)c2Cl)c(c(C#N)c[n]2COCC[Si+](C)(C)C)c2n1)=O Chemical compound CNC(CSc1nc(-c(cc(c(OC)c2)OCCOC)c2Cl)c(c(C#N)c[n]2COCC[Si+](C)(C)C)c2n1)=O MKNBXEANHDZWIH-UHFFFAOYSA-N 0.000 description 1
- HSHIGDDEOXUENZ-UHFFFAOYSA-N COc(c(C#N)c1)cc(-c2c(c(C#N)c[n]3COCC[Si+](C)(C)C)c3nc(SCC(O)=O)n2)c1Cl Chemical compound COc(c(C#N)c1)cc(-c2c(c(C#N)c[n]3COCC[Si+](C)(C)C)c3nc(SCC(O)=O)n2)c1Cl HSHIGDDEOXUENZ-UHFFFAOYSA-N 0.000 description 1
- PLWBOXUCHMALEZ-UHFFFAOYSA-N COc(c(OC)c1)cc(-c2c(c(C#N)c[nH]3)c3nc(SCC(NCC(F)(F)F)=O)n2)c1Cl Chemical compound COc(c(OC)c1)cc(-c2c(c(C#N)c[nH]3)c3nc(SCC(NCC(F)(F)F)=O)n2)c1Cl PLWBOXUCHMALEZ-UHFFFAOYSA-N 0.000 description 1
- ORYXSMHHAFKYEL-UHFFFAOYSA-N C[SH-]CCOC[n]1c2nc(SC)nc(Cl)c2c(Br)c1 Chemical compound C[SH-]CCOC[n]1c2nc(SC)nc(Cl)c2c(Br)c1 ORYXSMHHAFKYEL-UHFFFAOYSA-N 0.000 description 1
- LDDILCYXONMHNY-UHFFFAOYSA-N C[Si](C)(C)CCOC[n]1c2nc(SC)nc(Cl)c2c(C(O)=O)c1 Chemical compound C[Si](C)(C)CCOC[n]1c2nc(SC)nc(Cl)c2c(C(O)=O)c1 LDDILCYXONMHNY-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- This invention relates to substituted bicyclic pyrrolopyrimidine compounds having HSP90 inhibitory activity, to the use of such compounds in medicine, in relation to diseases which are responsive to inhibition of HSP90 activity such as cancers, and to pharmaceutical compositions containing such compounds.
- Molecular chaperones maintain the appropriate folding and conformation of proteins and are crucial in regulating the balance between protein synthesis and degradation. They have been shown to be important in regulating many important cellular functions, such as cell proliferation and apoptosis.
- Hsps heat shock proteins
- Hsps A number of multigene families of Hsps exist, with individual gene products varying in cellular expression, function and localization. They are classified according to molecular weight, e.g., Hsp70, Hsp90, and Hsp27.
- diseases in humans can be acquired as a result of protein misfolding.
- therapies which disrupt the molecular chaperone machinery may prove to be beneficial.
- misfolded proteins can cause protein aggregation resulting in neurodegenerative disorders.
- misfolded proteins may result in loss of wild type protein function, leading to deregulated molecular and physiological functions in the cell.
- Hsps have also been implicated in cancer. For example, there is evidence of differential expression of Hsps which may relate to the stage of tumour progression. As a result of the involvement of Hsp90 in various critical oncogenic pathways and the discovery that certain natural products with anticancer activity are targeting this molecular chaperone suggests that inhibiting the function of Hsp90 may be useful in the treatment of cancer.
- Hsp90 constitutes about 1-2% of total cellular protein. In cells, it forms dynamic multi-protein complexes with a wide variety of accessory proteins (referred to as co-chaperones) which appear responsible for regulating the chaperone function. It is essential for cell viability and it exhibits dual chaperone functions.
- co-chaperones accessory proteins
- Hsp90 forms a core component of the cellular stress response by interacting with many proteins after their native conformation has been altered.
- Environmental stresses such as heat shock, heavy metals or alcohol, generate localised protein unfolding. Hsp90 (in concert with other chaperones) binds these unfolded proteins allowing adequate refolding and preventing nonspecific aggregation (Smith et al., 1998).
- Hsp90 may also play a role in buffering against the effects of mutation, presumably by correcting the inappropriate folding of mutant proteins.
- Hsp90 also has an important regulatory role.
- Hsp90 plays a housekeeping role in the cell, maintaining the conformational stability and maturation of many client proteins.
- steroid hormone receptors e.g. estrogen receptor, progesterone receptor
- Ser/Thr or tyrosine kinases e.g. Her2, Raf-1 , CDK4, and Lck
- a collection of apparently unrelated proteins e.g.
- Hsp90 is responsible for stabilising and activating mutated kinases where the wild type kinase is not an Hsp90 client (for an example see the B-Raf story published in da Rocha Dias et al., 2005). All of these proteins play key regulatory roles in many physiological and biochemical processes in the cell. New client proteins of Hsp90 are being constantly identified; see http://www.picard.ch/downloads/Hsp90interactors.pdf for the most up to date list.
- Hsp90 The highly conserved Hsp90 family in humans consists of four genes, namely the cytosolic Hsp90 ⁇ and Hsp90 ⁇ isoforms, GRP94 in the endoplasmic reticulum and Hsp75/TRAP1 in the mitochondrial matrix. Apart from the differences in sub-cellular localisation, very little is known about the differences in function between Hsp90 ⁇ / ⁇ , GRP94 and TRAP1.
- Hsp90 participates in a series of complex interactions with a range of client and regulatory proteins. Although the precise molecular details remain to be elucidated, biochemical and X-ray crystallographic studies carried out over the last few years have provided increasingly detailed insights into the chaperone function of Hsp90.
- Hsp90 is an ATP-dependent molecular chaperone, with dimerisation of the nucleotide binding domains being essential for ATP hydrolysis, which is in turn essential for chaperone function. Binding of ATP results in the formation of a toroidal dimer structure in which the N terminal domains are brought into closer contact with each other resulting in a conformational switch known as the 'clamp mechanism'. This conformational switching is, in part, regulated by the various co-chaperones associated with Hsp90.
- geldanamycin The predominant mechanism of action of geldanamycin, 17AAG, and radicicol involves binding to Hsp90 at the ATP binding site located in the N-terminal domain of the protein, leading to inhibition of the intrinsic ATPase activity of Hsp90.
- Hsp90 ATPase activity by 17AAG induces the loss of p23 from the chaperone-client protein complex interrupting the chaperone cycle. This leads to the formation of a Hsp90-client protein complex that targets these client proteins for degradation via the ubiquitin proteasome pathway.
- Treatment with Hsp90 inhibitors leads to selective degradation of important proteins (for example Her2, Akt, estrogen receptor and CDK4) involved in cell proliferation, cell cycle regulation and apoptosis, processes which are fundamentally important in cancer.
- important proteins for example Her2, Akt, estrogen receptor and CDK4
- Hsp90 function has been shown to cause selective degradation of important signalling proteins involved in cell proliferation, cell cycle regulation and apoptosis, processes which are fundamentally important and which are commonly deregulated in cancer.
- An attractive rationale for developing drugs against this target for use in the clinic is that by simultaneously depleting proteins associated with the transformed phenotype, one may obtain a strong antitumour effect and achieve a therapeutic advantage against cancer versus normal cells. These events downstream of Hsp90 inhibition are believed to be responsible for the antitumour activity of Hsp90 inhibitors in cell culture and animal models.
- Hsp90 inhibitors therefore resensitise strains which have become resistant to, for example, azole antifungal agents (e.g. fluconazole) as well as newer agents such as echinocandins.
- azole antifungal agents e.g. fluconazole
- PCT/GB2007/000831 is concerned with a class of cyano pyrrolopyrimidines which inhibit HSP90 activity. This invention relates to a subset of compounds within the PCT/GB2007/000831 class, but which are not specifically disclosed therein.
- R is cyano or methoxy
- R 1 and R 2 are independently selected from hydrogen, optionally substituted C 1 -C 3 alkyl, and optionally substituted C 3 -C 6 cycloalkyl; or R 1 and R 2 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C 1 -C 3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C 1 -C 3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine;
- R 3 and R 4 are independently selected from hydrogen, C 1 -C 3 alkyl in which one or more hydrogens are optionally replaced by fluorine, and cyclopropyl; or R 2 and R 3 taken together with the carbon to which they are attached form a 3- to 6-membered ring cycloalkyl ring;
- n 1, 2 or 3;
- R 5 and R 6 are independently selected from hydrogen, C 1 -C 3 alkyl, and C 3 -C 6 cycloalkyl; or R 5 and R 6 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C 1 -C 3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C 1 -C 3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine; or (ii) Ci-C 3 alkoxy in which one or more hydrogens in the alkyl part are optionally replaced by fluorine.
- the nitrogens in the fused pyrimidine ring present in the compounds of the invention may be oxidesed to form N-oxides.
- Such N-oxides substantially retain the HSP90 inhibitory activity of the parent compounds, and are thus form part of the invention.
- Any unqualified reference herein to a compound which falls within formula (I) (“compounds of the invention") is to be construed as a reference to that compound, irrespective of whether it is or is not in the form of an N-oxide.
- compounds of the invention which may exist in one or more stereoisomeric form, because of the presence of asymmetric atoms or rotational restrictions, can exist as a number of stereoisomers with R or S stereochemistry at each chiral centre or as atropisomeres with R or S stereochemistry at each chiral axis.
- the invention includes all such enantiomers and diastereoisomers and mixtures thereof. Any unqualified reference herein to a compound which falls within formula (I) ("compounds of the invention”) is to be construed as a reference to that compound, irrespective of whether it is or is not in the form of a pure stereoisomer or a mixture of stereoisomers.
- the invention provides the use of a compound of compound of the invention in the preparation of a composition for inhibition of HSP90 activity in vitro or in vivo.
- the invention also provides a method of treatment of diseases which are responsive to inhibition of HSP90 activity in mammals, which method comprises administering to the mammal an amount of a compound of the invention effective to inhibit said HSP90 activity.
- the in vivo use, and method, of the invention is applicable to the treatment of diseases in which HSP90 activity is implicated, including use for immunosuppression or the treatment of viral disease, drug resistant fungal infection (since HSP90 inhibitors are able to resensitise strains which have become resistant to, for example, azole antifungal agents (e.g.
- fluconazole as well as newer agents such as echinocandins
- protozoal infections inflammatory diseases such as rheumatoid arthritis, asthma, multiple sclerosis, Type I diabetes, lupus, psoriasis and inflammatory bowel disease; cystic fibrosis angiogenesis-related disease such as diabetic retinopathy, haemangiomas, and endometriosis; or for protection of normal cells against chemotherapy-induced toxicity; or diseases where failure to undergo apoptosis is an underlying factor; or protection from hypoxia-ischemic injury due to elevation of Hsp70 in the heart and brain; scrapie/CJD, Huntingdon's or Alzheimer's disease. Use for the treatment of cancer is especially indicated.
- substituted as applied to any moiety herein means substituted with at least one substituent, for example selected from (CVC ⁇ Jalkyl, (C 1 -C 6 JaIkOXy (including methylenedioxy and ethylenedioxy substitution on adjacent carbon atoms of a carbocyclic or heterocyclic ring), hydroxy, hydroxy(C 1 -C 6 )alkyl, mercapto, mercapto(C 1 -C 6 )alkyl, (CrCeJalkylthio, monocyclic carbocyclic of 3-6 ring carbon atoms, monocyclic heterocyclic of 5 or 6 ring atoms, halo (including fluoro and chloro), trifluoromethyl, trifluoromethoxy, nitro, nitrile
- the optional substituent contains an alkyl radical
- that alkyl radical may be substituted by a monocyclic carbocyclic group of 3-6 ring carbon atoms, or a monocyclic heterocyclic group of 5 or 6 ring atoms.
- the optional substituent is or comprises a monocyclic carbocyclic group of 3-6 ring carbon atoms, or a monocyclic heterocyclic group of 5 or 6 ring atoms, that ring may itself be substituted by any of the non-cyclic optional substituents listed above.
- An "optional substituenf may be one of the substituent groups encompassed in the above description.
- salt includes base addition, acid addition and quaternary salts.
- Compounds of the invention which are acidic can form salts, including pharmaceutically or veterinarily acceptable salts, with bases such as alkali metal hydroxides, e.g. sodium and potassium hydroxides; alkaline earth metal hydroxides e.g. calcium, barium and magnesium hydroxides; with organic bases e.g. N-ethyl piperidine, dibenzylamine and the like.
- bases such as alkali metal hydroxides, e.g. sodium and potassium hydroxides; alkaline earth metal hydroxides e.g. calcium, barium and magnesium hydroxides; with organic bases e.g. N-ethyl piperidine, dibenzylamine and the like.
- Those compounds (I) which are basic can form salts, including pharmaceutically or veterinarily acceptable salts with inorganic acids, e.g.
- hydrohalic acids such as hydrochloric or hydrobromic acids, sulphuric acid, nitric acid or phosphoric acid and the like
- organic acids e.g. with acetic, tartaric, succinic, fumaric, maleic, malic, salicylic, citric, methanesulphonic and p-toluene sulphonic acids and the like.
- variable substituents in compounds of the invention is cyano or methoxy. Currently methoxy is preferred.
- Ri and R 2 are independently selected from hydrogen; optionally substituted Ci-C 3 alkyl i.e. methyl, ethyl, n- or iso-propyl; and optionally substituted C 3 -C 6 cycloalkyl i.e.
- Ri and R 2 may be selected from, for example, halo, amino, mono- or di- (Ci-C 3 alkyl)amino, hydroxyl, methoxy, or cyano.
- R 1 and R 2 are independently selected from hydrogen, methyl, trifluoromethyl, ethyl, 2-trifluoroethyl, isopropyl, tert-butyl, cyclopropyl, cyclobutyl, hydroxymethyl and hydroxyethyl.
- Ri and R 2 taken together with the nitrogen to which they are attached form a, 3-, 4-, 5- or 6-membered ring optionally substituted by fluoro, chloro, bromo, cyano, methyl, trifluoromethyl, ethyl, hydroxymethyl, hydroxy, or hydroxyethyl.
- the ring formed by R 1 and R 2 and the nitrogen to which they are attached may be, for example, a piperidine, homopiperidine, piperazine, or morpholine ring.
- R 3 and R 4 are independently selected from hydrogen, C 1 -C 3 alkyl ie methyl, ethyl or n- or iso- propyl, in which one or more hydrogens are optionally replaced by fluorine, and cyclopropyl; or R 2 and R 3 taken together with the carbon to which they are attached form a 3- to 6-membered cycloalkyl ring i.e. a cylopropyl, cyclobutyl, cyclopentyl or cyclohexyl ring.
- R 3 and R 4 are each hydrogen, or one of R 3 and R 4 is hydrogen and the other is methyl.
- n 1 , 2 or 3.
- Z is (i) -NR 5 R 6 wherein R 5 and R 6 are independently selected from hydrogen, Ci-C 3 alkyl i.e. methyl, ethyl, or n- or iso-propyl, and C 3 -C 6 cycloalkyl i.e.
- Z may be -NR 5 R 6 wherein R 5 and R 6 are independently selected from hydrogen, methyl, trifluoromethyl, ethyl, and cyclopropyl; or R 5 and R 6 taken together with the nitrogen to which they are attached form a 5- or 6-membered ring such as a piperidine, piperazine, or morpholine ring, optionally substituted by chloro, bromo, cyano, methyl, trifluoromethyl, hydroxymethyl or hydroxyethyl.
- Z is methoxy, trifluoromethoxy, or ethoxy.
- the compounds of the invention are inhibitors of HSP90 and are useful in the treatment of diseases which are responsive to inhibition of HSP90 activity, as referred to above.
- diseases include cancers; viral diseases such as Hepatitis C (HCV) (Waxman, 2002); Immunosuppression such as in transplantation (Bijlmakers, 2000 and Yorgin, 2000); Antiinflammatory diseases (Bucci, 2000) such as Rheumatoid arthritis, Asthma, MS, Type I Diabetes, Lupus, Psoriasis and Inflammatory Bowel Disease; Cystic fibrosis (Fuller, 2000); Angiogenesis-related diseases (Hur, 2002 and Kurebayashi, 2001 ): diabetic retinopathy, haemangiomas, psoriasis, endometriosis and tumour angiogenesis.
- an Hsp90 inhibitor of the invention may protect normal cells against chemotherapy-induced toxicity and be useful in diseases where failure to undergo apoptosis is an underlying factor.
- Such an Hsp90 inhibitor may also be useful in diseases where the induction of a cell stress or heat shock protein response could be beneficial, for example, protection from hypoxia-ischemic injury due to elevation of Hsp70 in the heart (Hutter, 1996 and Trost, 1998) and brain (Plumier, 1997 and Rajder, 2000).
- Hsp90 inhibitor - induced increase in Hsp70 levels could also be useful in diseases where protein misfolding or aggregation is a major causal factor, for example, neurogenerative disorders such as scrapie/CJD, Huntingdon's and Alzheimer's (Sittler, 2001 ; Trazelt, 1995 and Winklhofer, 2001)". Accordingly, the invention also includes:
- a pharmaceutical or veterinary composition comprising a compound of the invention, together with a pharmaceutically or veterinarily acceptable carrier.
- a method of treatment of diseases or conditions which are responsive to inhibition of HSP90 activity in mammals which method comprises administering to the mammal an amount of a compound of the invention effective to inhibit said HSP90 activity.
- a suitable dose for orally administrable formulations will usually be in the range of 0.1 to 3000 mg, once, twice or three times per day, or the equivalent daily amount administered by infusion or other routes.
- optimum dose levels and frequency of dosing will be determined by clinical trials as is conventional in the art.
- the compounds with which the invention is concerned may be prepared for administration by any route consistent with their pharmacokinetic properties.
- the orally administrable compositions may be in the form of tablets, capsules, powders, granules, lozenges, liquid or gel preparations, such as oral, topical, or sterile parenteral solutions or suspensions.
- Tablets and capsules for oral administration may be in unit dose presentation form, and may contain conventional excipients such as binding agents, for example syrup, acacia, gelatin, sorbitol, tragacanth, or polyvinyl-pyrrolidone; fillers for example lactose, sugar, maize-starch, calcium phosphate, sorbitol or glycine; tabletting lubricant, for example magnesium stearate, talc, polyethylene glycol or silica; disintegrants for example potato starch, or acceptable wetting agents such as sodium lauryl sulphate.
- the tablets may be coated according to methods well known in normal pharmaceutical practice.
- Oral liquid preparations may be in the form of, for example, aqueous or oily suspensions, solutions, emulsions, syrups or elixirs, or may be presented as a dry product for reconstitution with water or other suitable vehicle before use.
- Such liquid preparations may contain conventional additives such as suspending agents, for example sorbitol, syrup, methyl cellulose, glucose syrup, gelatin hydrogenated edible fats; emulsifying agents, for example lecithin, sorbitan monooleate, or acacia; non-aqueous vehicles (which may include edible oils), for example almond oil, fractionated coconut oil, oily esters such as glycerine, propylene glycol, or ethyl alcohol; preservatives, for example methyl or propyl p- hydroxybenzoate or sorbic acid, and if desired conventional flavouring or colouring agents.
- suspending agents for example sorbitol, syrup, methyl cellulose, glucose syrup, gelatin hydrogenated edible fats
- emulsifying agents for example lecithin, sorbitan monooleate, or acacia
- non-aqueous vehicles which may include edible oils
- almond oil fractionated coconut oil
- oily esters such as glycerine, propy
- the drug may be made up into a cream, lotion or ointment.
- Cream or ointment formulations which may be used for the drug are conventional formulations well known in the art, for example as described in standard textbooks of pharmaceutics such as the British Pharmacopoeia.
- the active ingredient may also be administered parenterally in a sterile medium.
- the drug can either be suspended or dissolved in the vehicle.
- adjuvants such as a local anaesthetic, preservative and buffering agents can be dissolved in the vehicle.
- Compounds of the invention may be administered together with other classes opf pharmaceutically active drugs.
- combination therapy with two or more different classes of anticancer agent is a recognised and widespread practice.
- the present compounds may be used in such combination therapy, particularly where the other drug(s) have a mode of action different from HSP90 inhibition.
- Flash chromatography was performed with pre-packed silica gel cartridges (Strata SI-1; 61 A, Phenomenex, Cheshire UK or 1ST Flash II, 54A, Argonaut, Hengoed, UK). Thin layer chromatography was conducted with 5 x 10 cm plates coated with Merck Type 60 F 254 silica gel.
- LC/MS Method A
- LC/MS Method A
- column Phenomenex Luna 3u C18(2) 30 x 4.6 mm
- Buffer A prepared by dissolving 1.93g ammonium acetate in 2.5 L HPLC grade H 2 O and adding 2 mL formic acid.
- Buffer B prepared by adding 132 mL buffer A to 2.5 L of HPLC grade acetonitrile and adding 2 mL formic acid; elution gradient 95:5 to 5:95 buffer A : buffer.B over 3.75 minutes.
- Flow rate 2.0 ml_/min) Retention Times (RT) are reported in minutes, lonisation is positive unless otherwise stated.
- Some compounds of the invention were purified by preparative HPLC. Preparative HPLC purifications were performed on a Waters FractionLynx MS Autopurification system with a Gemini ® 5 ⁇ M C18(2), 100 mm * 20 mm i.d. column from Phenomenex, running at a flow rate of 20 mL min '1 with UV diode array detection (210 - 400 nm) and mass-directed collection. Various gradients (1 to 4) were used and these are shown in table 1. The pH of the eluant and the solvent gradient depends on the characteristics of the compound being purified.
- Solvent B 95% v/v HPLC grade acetonitrile + 5% v/v Solvent A + 0.08% v/v formic acid.
- Solvent A HPLC grade Water + 10 mM ammonium acetate + 0.08% v/v ammonia solution.
- Solvent B 95% v/v HPLC grade acetonitrile + 5% v/v Solvent A + 0.08% v/v ammonia solution.
- the mass spectrometer was a Waters Micromass ZQ2000 spectrometer operating in positive or negative ion electrospray ionisation modes, with a molecular weight scan range of 150 to 1000.
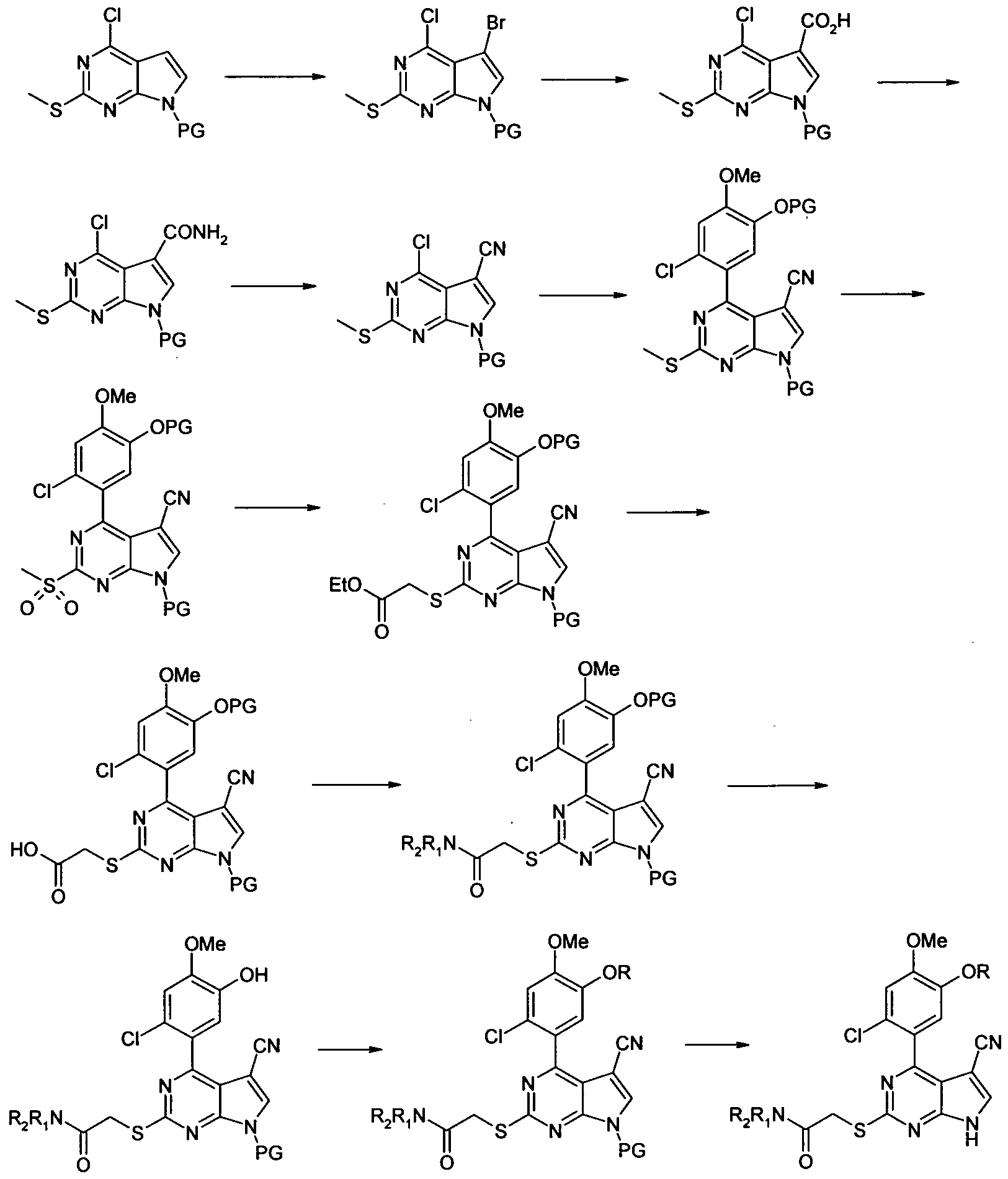
- Step 1 ⁇ chloro ⁇ -methylsulfanyl-Z ⁇ -trimethylsilanyl-ethoxymethyO-ZH-pyrrolo ⁇ .S-dJpyrimidine
- Ethyl thioglycolate (0.31 ml_, 2.78 mmol) was added to a mixture of NaH (46 mg, 1.15 mmol, 60% dispersion in mineral oil) in THF (10 ml.) at O 0 C under N 2 .
- 4-(2-Chloro-4-methoxy-5- methoxymethoxy-phenyl)-2-methanesulfonyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidine-5-carbonitrile (1.28 g, 2.32 mmol) dissolved in THF (10 mL) was then added dropwise.
- reaction mixture After stirring at ambient temperature for 5 minutes, the reaction mixture was cooled with an ice-water bath and diisopropyldiazodicarboxylate (55 ⁇ L, 0.281 mmol) was added and reaction mixture allowed to warm to ambient temperature and stir for 1.5 h. The reaction mixture was partitioned between ethyl acetate (20 mL) and saturated aqueous ammonium chloride solution (20 ml_).
- the title compound was made by way of method of example 22 step 3 (diazotization and quench with aqueous Iodine / sodium iodide solution).
- the title compound was prepared by way of the method of example 26 step 1 (boronic acid formation and subsequent cross coupling).
- Step 5 4-(2-Chloro-4-cyano-5-methoxy-phenyl)-2-methanesulfonyl-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile
- the title compound was prepared by way of the method of example 1 step 10 (oxidation with mcpba).
- the binding affinity for HSP90 of the compounds of the invention can be measured using a Fluorescence Polarization Assay.
- Fluorescence polarization also known as fluorescence an i sot ropy ⁇ measures the rotation of a fluorescing species in solution, where the larger molecule the more polarized the fluorescence emission.
- fluorophore When the fluorophore is excited with polarized light, the emitted light is also polarized.
- the molecular size is proportional to the polarization of the fluorescence emission.
- Test compound is added to the assay plate, left to equilibrate and the anisotropy measured again. Any change in anisotropy is due to competitive binding of compound to HSP90, thereby releasing probe.
- Chemicals are of the highest purity commercially available and all aqueous solutions are made up in AR water.
- BSA bovine serum albumen
- the Z' factor is calculated from zero controls and positive wells. It typically gives a value of 0.7 - 0.9.
- Control wells are at either side of the 96 well plates, where 40 ⁇ l of medium is added.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
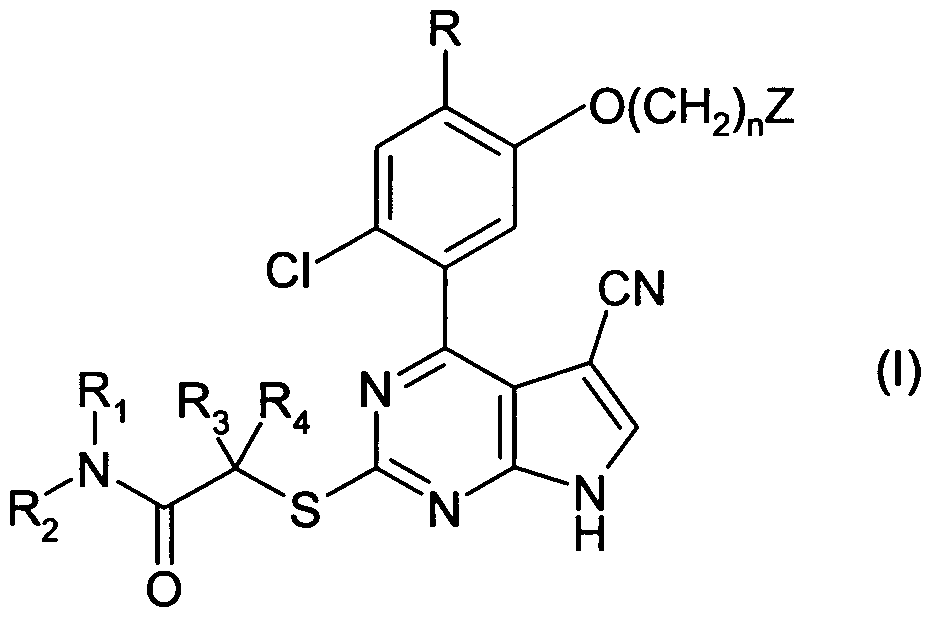
Compounds of formula (I) are inhibitors of HSP90, and are useful inter alia in the treatment of cancers, wherein R is cyano or methoxy; R1 and R2 are independently selected from hydrogen, optionally substituted C1-C3 alkyl, and optionally substituted C3-C6 cycloalkyl; or R1 and R2 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C1-C3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine; R3 and R4 are independently selected from hydrogen, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, and cyclopropyl; or R2 and R3 taken together with the carbon to which they are attached form a 3- to 6-membered ring cycloalkyl ring; n is 1, 2 or 3; and Z is (i) -NR5R6 wherein R5 and R6 are independently selected from hydrogen, C1-C3 alkyl, and C3-C6 cycloalkyl; or R5 and R6 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C1-C3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine; or (ii) C1-C3 alkoxy in which one or more hydrogens in the alkyl part are optionally replaced by fluorine.
Description
PYRROLOPYRIMIDINE DERIVATIVES HAVING HSP90 INHIBITORY ACTIVITY
This invention relates to substituted bicyclic pyrrolopyrimidine compounds having HSP90 inhibitory activity, to the use of such compounds in medicine, in relation to diseases which are responsive to inhibition of HSP90 activity such as cancers, and to pharmaceutical compositions containing such compounds.
Background to the invention
Molecular chaperones maintain the appropriate folding and conformation of proteins and are crucial in regulating the balance between protein synthesis and degradation. They have been shown to be important in regulating many important cellular functions, such as cell proliferation and apoptosis.
Heat Shock Proteins (Hsps)
Exposure of cells to a number of environmental stresses, including heat shock, alcohols, heavy metals and oxidative stress, results in the cellular accumulation of a number of chaperones, commonly known as heat shock proteins (Hsps). Induction of Hsps protects the cell against the initial stress insult, enhances recovery and leads to maintenance of a stress tolerant state. It has also become clear, however, that certain Hsps may also play a major molecular chaperone role under normal, stress-free conditions by regulating the correct folding, degradation, localization and function of a growing list of important cellular proteins.
A number of multigene families of Hsps exist, with individual gene products varying in cellular expression, function and localization. They are classified according to molecular weight, e.g., Hsp70, Hsp90, and Hsp27. Several diseases in humans can be acquired as a result of protein misfolding. Hence the development of therapies which disrupt the molecular chaperone machinery may prove to be beneficial. In some conditions (e.g., Alzheimer's disease, prion diseases and Huntington's disease), misfolded proteins can cause protein aggregation resulting in neurodegenerative disorders. Also, misfolded proteins may result in loss of wild type protein function, leading to deregulated molecular and physiological functions in the cell.
Hsps have also been implicated in cancer. For example, there is evidence of differential expression of Hsps which may relate to the stage of tumour progression. As a result of the
involvement of Hsp90 in various critical oncogenic pathways and the discovery that certain natural products with anticancer activity are targeting this molecular chaperone suggests that inhibiting the function of Hsp90 may be useful in the treatment of cancer.
HSP90
Hsp90 constitutes about 1-2% of total cellular protein. In cells, it forms dynamic multi-protein complexes with a wide variety of accessory proteins (referred to as co-chaperones) which appear responsible for regulating the chaperone function. It is essential for cell viability and it exhibits dual chaperone functions. When cells undergo various environmental cellular stresses, Hsp90 forms a core component of the cellular stress response by interacting with many proteins after their native conformation has been altered. Environmental stresses, such as heat shock, heavy metals or alcohol, generate localised protein unfolding. Hsp90 (in concert with other chaperones) binds these unfolded proteins allowing adequate refolding and preventing nonspecific aggregation (Smith et al., 1998). In addition, recent results suggest that Hsp90 may also play a role in buffering against the effects of mutation, presumably by correcting the inappropriate folding of mutant proteins. However, Hsp90 also has an important regulatory role. Under normal physiological conditions, together with its endoplasmic reticulum homologue GRP94, Hsp90 plays a housekeeping role in the cell, maintaining the conformational stability and maturation of many client proteins. These can be subdivided into three groups: (a) steroid hormone receptors (e.g. estrogen receptor, progesterone receptor) (b) Ser/Thr or tyrosine kinases (e.g. Her2, Raf-1 , CDK4, and Lck), and (c) a collection of apparently unrelated proteins, e.g. mutant p53 and the catalytic subunit of telomerase hTERT. It has also been shown recently that Hsp90 is responsible for stabilising and activating mutated kinases where the wild type kinase is not an Hsp90 client (for an example see the B-Raf story published in da Rocha Dias et al., 2005). All of these proteins play key regulatory roles in many physiological and biochemical processes in the cell. New client proteins of Hsp90 are being constantly identified; see http://www.picard.ch/downloads/Hsp90interactors.pdf for the most up to date list.
The highly conserved Hsp90 family in humans consists of four genes, namely the cytosolic Hsp90α and Hsp90β isoforms, GRP94 in the endoplasmic reticulum and Hsp75/TRAP1 in the mitochondrial matrix. Apart from the differences in sub-cellular localisation, very little is known about the differences in function between Hsp90α/β, GRP94 and TRAP1. Initial reports
suggesting that certain client proteins were chaperoned by a specific Hsp90 (e.g. Her2 by Grp94 alone) appear to have been erroneous.
Hsp90 participates in a series of complex interactions with a range of client and regulatory proteins. Although the precise molecular details remain to be elucidated, biochemical and X-ray crystallographic studies carried out over the last few years have provided increasingly detailed insights into the chaperone function of Hsp90.
Following earlier controversy on this issue, it is now clear that Hsp90 is an ATP-dependent molecular chaperone, with dimerisation of the nucleotide binding domains being essential for ATP hydrolysis, which is in turn essential for chaperone function. Binding of ATP results in the formation of a toroidal dimer structure in which the N terminal domains are brought into closer contact with each other resulting in a conformational switch known as the 'clamp mechanism'. This conformational switching is, in part, regulated by the various co-chaperones associated with Hsp90.
Hsp90 as a Therapeutic Target
Due to its involvement in regulating a number of signalling pathways that are crucially important in driving the phenotype of a tumour, and the discovery that certain bioactive natural products exert their effects via Hsp90 activity, the molecular chaperone Hsp90 is currently being assessed as a new target for anticancer drug development.
The predominant mechanism of action of geldanamycin, 17AAG, and radicicol involves binding to Hsp90 at the ATP binding site located in the N-terminal domain of the protein, leading to inhibition of the intrinsic ATPase activity of Hsp90.
Inhibition of Hsp90 ATPase activity by 17AAG induces the loss of p23 from the chaperone-client protein complex interrupting the chaperone cycle. This leads to the formation of a Hsp90-client protein complex that targets these client proteins for degradation via the ubiquitin proteasome pathway. Treatment with Hsp90 inhibitors leads to selective degradation of important proteins (for example Her2, Akt, estrogen receptor and CDK4) involved in cell proliferation, cell cycle regulation and apoptosis, processes which are fundamentally important in cancer.
The preclinical development of 17AAG as an anticancer agent has been well documented and is currently undergoing Phase Il clinical trials. Phase I clinical trials results have been published. Of these trials, the most positive used a maximum dose of 450mg/m2/week achieved with PD marker responses in the majority of patients and possible antitumour activity in two patients
Inhibition of Hsp90 function has been shown to cause selective degradation of important signalling proteins involved in cell proliferation, cell cycle regulation and apoptosis, processes which are fundamentally important and which are commonly deregulated in cancer. An attractive rationale for developing drugs against this target for use in the clinic is that by simultaneously depleting proteins associated with the transformed phenotype, one may obtain a strong antitumour effect and achieve a therapeutic advantage against cancer versus normal cells. These events downstream of Hsp90 inhibition are believed to be responsible for the antitumour activity of Hsp90 inhibitors in cell culture and animal models.
Recent work has shown that the acetylation status of Hsp90 also plays a role in the control of the chaperone cycle. Inhibition of HDAC6 by either small molecule inhibitors or through siRNA gene targeting interrupts the chaperone cycle. Such treatments cause client protein degradation in a fashion analogous to small molecule ATP site inhibitors.
Recent reports (see Cowen et. al. Science 309, 2185 (2005) and Heitman, Science 309, 2175, 2005) also indicate that Hsp90 is required both for the emergence of fungal isolates resistant to antifungal agents, and for continued drug resistance once this has occurred. Hsp90 inhibitors therefore resensitise strains which have become resistant to, for example, azole antifungal agents (e.g. fluconazole) as well as newer agents such as echinocandins.
Brief description of the invention
Our co-pending patent international patent application no. PCT/GB2007/000831 is concerned with a class of cyano pyrrolopyrimidines which inhibit HSP90 activity. This invention relates to a subset of compounds within the PCT/GB2007/000831 class, but which are not specifically disclosed therein.
Detailed description of the invention
According to the invention, there is provided a compound of formula (I) or a salt, especially a pharmaceutically or veterinarily acceptable salt, thereof:
R is cyano or methoxy;
R1 and R2 are independently selected from hydrogen, optionally substituted C1-C3 alkyl, and optionally substituted C3-C6 cycloalkyl; or R1 and R2 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C1-C3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine;
R3 and R4 are independently selected from hydrogen, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, and cyclopropyl; or R2 and R3 taken together with the carbon to which they are attached form a 3- to 6-membered ring cycloalkyl ring;
n is 1, 2 or 3;
Z is
(i) -NR5R6 wherein R5 and R6 are independently selected from hydrogen, C1-C3 alkyl, and C3-C6 cycloalkyl; or R5 and R6 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C1-C3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine; or
(ii) Ci-C3 alkoxy in which one or more hydrogens in the alkyl part are optionally replaced by fluorine.
In common with many organic compounds useful in medicine, at least some of the compounds of the invention are expected to be recoverable as crystalline hydrates and solvates. Such hydrates and solvates are of course merely specific physico-chemical forms of the active compounds of the invention and therefore form part of the invention. Any unqualified reference herein to a compound which falls within formula (I) ("compounds of the invention") is to be construed as a reference to that compound, irrespective of whether it is or is not in the form of a hydrate or solvate. The term 'solvate' is used herein to describe a molecular complex comprising the compound of the invention and a stoichiometric amount of one or more pharmaceutically acceptable solvent molecules, for example, ethanol. The term 'hydrate' is employed when said solvent is water.
Likewise, the nitrogens in the fused pyrimidine ring present in the compounds of the invention may be oxidesed to form N-oxides. Such N-oxides substantially retain the HSP90 inhibitory activity of the parent compounds, and are thus form part of the invention. Any unqualified reference herein to a compound which falls within formula (I) ("compounds of the invention") is to be construed as a reference to that compound, irrespective of whether it is or is not in the form of an N-oxide.
Furthermore, compounds of the invention which may exist in one or more stereoisomeric form, because of the presence of asymmetric atoms or rotational restrictions, can exist as a number of stereoisomers with R or S stereochemistry at each chiral centre or as atropisomeres with R or S stereochemistry at each chiral axis. The invention includes all such enantiomers and diastereoisomers and mixtures thereof. Any unqualified reference herein to a compound which falls within formula (I) ("compounds of the invention") is to be construed as a reference to that compound, irrespective of whether it is or is not in the form of a pure stereoisomer or a mixture of stereoisomers.
In another aspect, the invention provides the use of a compound of compound of the invention in the preparation of a composition for inhibition of HSP90 activity in vitro or in vivo.
The invention also provides a method of treatment of diseases which are responsive to inhibition of HSP90 activity in mammals, which method comprises administering to the mammal an amount of a compound of the invention effective to inhibit said HSP90 activity.
The in vivo use, and method, of the invention is applicable to the treatment of diseases in which HSP90 activity is implicated, including use for immunosuppression or the treatment of viral disease, drug resistant fungal infection (since HSP90 inhibitors are able to resensitise strains which have become resistant to, for example, azole antifungal agents (e.g. fluconazole) as well as newer agents such as echinocandins), protozoal infections, inflammatory diseases such as rheumatoid arthritis, asthma, multiple sclerosis, Type I diabetes, lupus, psoriasis and inflammatory bowel disease; cystic fibrosis angiogenesis-related disease such as diabetic retinopathy, haemangiomas, and endometriosis; or for protection of normal cells against chemotherapy-induced toxicity; or diseases where failure to undergo apoptosis is an underlying factor; or protection from hypoxia-ischemic injury due to elevation of Hsp70 in the heart and brain; scrapie/CJD, Huntingdon's or Alzheimer's disease. Use for the treatment of cancer is especially indicated.
Unless otherwise specified in the context in which it occurs, the term "substituted" as applied to any moiety herein means substituted with at least one substituent, for example selected from (CVCβJalkyl, (C1-C6JaIkOXy (including methylenedioxy and ethylenedioxy substitution on adjacent carbon atoms of a carbocyclic or heterocyclic ring), hydroxy, hydroxy(C1-C6)alkyl, mercapto, mercapto(C1-C6)alkyl, (CrCeJalkylthio, monocyclic carbocyclic of 3-6 ring carbon atoms, monocyclic heterocyclic of 5 or 6 ring atoms, halo (including fluoro and chloro), trifluoromethyl, trifluoromethoxy, nitro, nitrile
(-CN), oxo, -COOH, -COORA, -CORA, -SO2RA, -CONH2, -SO2NH2, -CONHRA, -SO2NHRA, -C0NRARB, -SO2NRARB, -NH2, -NHRA, -NRARB, -OCONH2, -OCONHRA , -0C0NRARB, -NHCORA, -NHCOORA, -NRBCOORA, -NHSO2ORA, -NRBSO2ORA, -NHCONH2, -NRAC0NH2, -NHCONHR8, -NRACONHRB, -NHCONRARB or -NRACONRARB wherein RA and RB are independently a
group. In the case where the optional substituent contains an alkyl radical, that alkyl radical may be substituted by a monocyclic carbocyclic group of 3-6 ring carbon atoms, or a monocyclic heterocyclic group of 5 or 6 ring atoms. In the case where the optional substituent is or comprises a monocyclic carbocyclic group of 3-6 ring carbon atoms, or a monocyclic heterocyclic group of 5 or 6 ring
atoms, that ring may itself be substituted by any of the non-cyclic optional substituents listed above. An "optional substituenf may be one of the substituent groups encompassed in the above description.
As used herein the term "salt" includes base addition, acid addition and quaternary salts. Compounds of the invention which are acidic can form salts, including pharmaceutically or veterinarily acceptable salts, with bases such as alkali metal hydroxides, e.g. sodium and potassium hydroxides; alkaline earth metal hydroxides e.g. calcium, barium and magnesium hydroxides; with organic bases e.g. N-ethyl piperidine, dibenzylamine and the like. Those compounds (I) which are basic can form salts, including pharmaceutically or veterinarily acceptable salts with inorganic acids, e.g. with hydrohalic acids such as hydrochloric or hydrobromic acids, sulphuric acid, nitric acid or phosphoric acid and the like, and with organic acids e.g. with acetic, tartaric, succinic, fumaric, maleic, malic, salicylic, citric, methanesulphonic and p-toluene sulphonic acids and the like. Any unqualified reference herein to a compound which falls within formula (I) is to be construed as a reference to that compound, irrespective of whether it is or is not in the form of salt.
For a review on suitable salts, see Handbook of Pharmaceutical Salts: Properties, Selection, and Use by Stahl and Wermuth (Wiley-VCH, Weinheim, Germany, 2002).
The variable substituents in compounds of the invention R is cyano or methoxy. Currently methoxy is preferred.
Ri and R2 are independently selected from hydrogen; optionally substituted Ci-C3 alkyl i.e. methyl, ethyl, n- or iso-propyl; and optionally substituted C3-C6 cycloalkyl i.e. cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl; or R1 and R2 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, Ci-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(CrC3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine. Optional substituents in Ri and R2 may be selected from, for example, halo, amino, mono- or di- (Ci-C3alkyl)amino, hydroxyl, methoxy, or cyano. In some currently preferred embodiments, R1 and R2 are independently selected from hydrogen, methyl, trifluoromethyl, ethyl, 2-trifluoroethyl, isopropyl, tert-butyl, cyclopropyl, cyclobutyl, hydroxymethyl and hydroxyethyl. In other
embodiments, Ri and R2 taken together with the nitrogen to which they are attached form a, 3-, 4-, 5- or 6-membered ring optionally substituted by fluoro, chloro, bromo, cyano, methyl, trifluoromethyl, ethyl, hydroxymethyl, hydroxy, or hydroxyethyl. The ring formed by R1 and R2 and the nitrogen to which they are attached may be, for example, a piperidine, homopiperidine, piperazine, or morpholine ring.
R3 and R4 are independently selected from hydrogen, C1-C3 alkyl ie methyl, ethyl or n- or iso- propyl, in which one or more hydrogens are optionally replaced by fluorine, and cyclopropyl; or R2 and R3 taken together with the carbon to which they are attached form a 3- to 6-membered cycloalkyl ring i.e. a cylopropyl, cyclobutyl, cyclopentyl or cyclohexyl ring. Currently it is preferred that R3 and R4 are each hydrogen, or one of R3 and R4 is hydrogen and the other is methyl.
n is 1 , 2 or 3. Currently preferred are compounds wherein n is 2.
Z is (i) -NR5R6 wherein R5 and R6 are independently selected from hydrogen, Ci-C3 alkyl i.e. methyl, ethyl, or n- or iso-propyl, and C3-C6 cycloalkyl i.e. cylopropyl, cyclobutyl, cyclopentyl or cyclohexyl; or R5 and R6 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring, such as a piperidine, homopiperidine, piperazine, or morpholine ring, optionally substituted by chloro, bromo, cyano, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C1-C3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine; or (ii) C1-C3 alkoxy i.e. methoxy, ethoxy, or n- or iso-propoxy, in which one or more hydrogens in the alkyl part are optionally replaced by fluorine. For example Z may be -NR5R6 wherein R5 and R6 are independently selected from hydrogen, methyl, trifluoromethyl, ethyl, and cyclopropyl; or R5 and R6 taken together with the nitrogen to which they are attached form a 5- or 6-membered ring such as a piperidine, piperazine, or morpholine ring, optionally substituted by chloro, bromo, cyano, methyl, trifluoromethyl, hydroxymethyl or hydroxyethyl. In some preferred embodiments, Z is methoxy, trifluoromethoxy, or ethoxy.
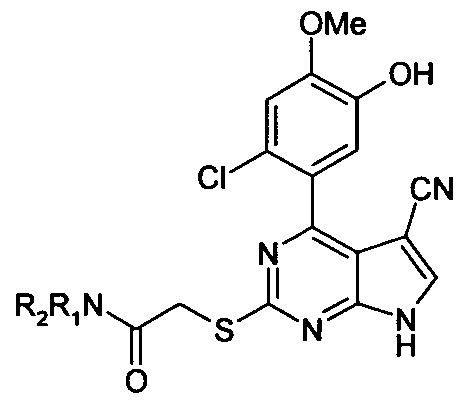
Specific examples of compounds of the invention include:
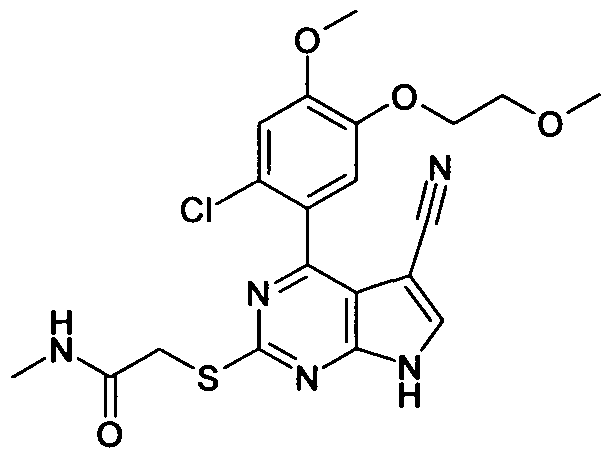
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N- dimethyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- cyclopropyl-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- cyclopropyl-acetamide;
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- cyclopropyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- isopropyl-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- isopropyl-acetamide;
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- isopropyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-ethyl- acetamide;
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- ethyl-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- ethyl-acetamide;
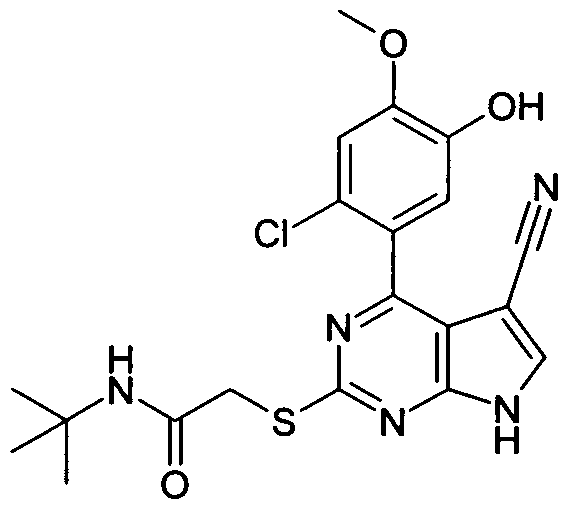
N-tert-Butyl-2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-acetamide;
N-tert-Butyl-2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-
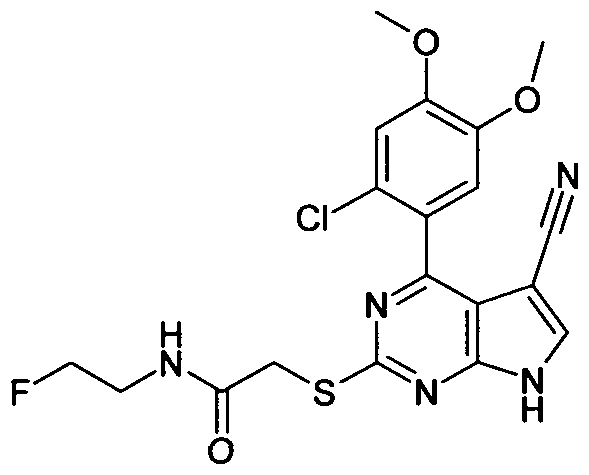
(2-fluoro-ethyl)-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-(2- fluoro-ethyl)-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N-methyl-acetamide;
2-{4-[2-Chloro-5-(2-diethylamino-ethoxy)-4-methoxy-phenyl]-5-cyano-7H- pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl}-N,N-dimethyl-acetamide; -
2-{4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxy)-phenyl]-5-cyano-7H-pyrro
lo[2,3-d]pyrimidin-2-ylsulfanyl}-N-methyl-acetamide;
2-{4-[2-Chloro-5-(2-hydroxy-ethoxy)-4-methoxy phenyl]-5-cyano-7H-pyrro lo[2,3-d]pyrimidin-2-ylsulfanyl}-N-methyl-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-
N,N-dimethyl-propionamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-(2,2,2- trifluofluoro-ethyl)-acetamide;
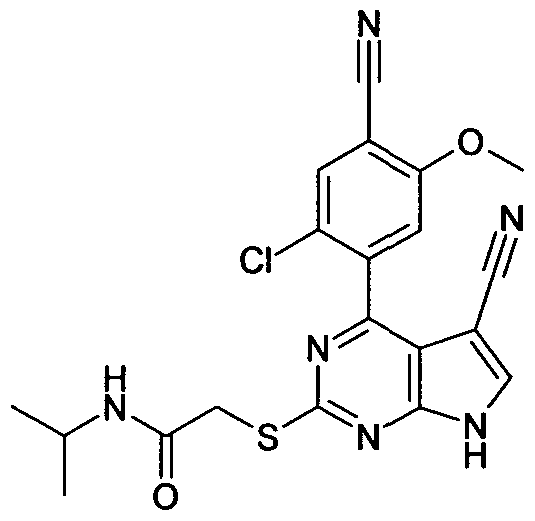
2-[4-[2-Chloro-4-cyano-5-methoxy-phenyl]-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulphanyl]-N- isopropyl-acetamide;
and salts thereof.
Compounds of the invention may be synthesised by the methods described in, or analogous to, the Examples herein.
The compounds of the invention are inhibitors of HSP90 and are useful in the treatment of diseases which are responsive to inhibition of HSP90 activity, as referred to above. Such diseases include cancers; viral diseases such as Hepatitis C (HCV) (Waxman, 2002); Immunosuppression such as in transplantation (Bijlmakers, 2000 and Yorgin, 2000); Antiinflammatory diseases (Bucci, 2000) such as Rheumatoid arthritis, Asthma, MS, Type I Diabetes, Lupus, Psoriasis and Inflammatory Bowel Disease; Cystic fibrosis (Fuller, 2000); Angiogenesis-related diseases (Hur, 2002 and Kurebayashi, 2001 ): diabetic retinopathy, haemangiomas, psoriasis, endometriosis and tumour angiogenesis. Also an Hsp90 inhibitor of the invention may protect normal cells against chemotherapy-induced toxicity and be useful in diseases where failure to undergo apoptosis is an underlying factor. Such an Hsp90 inhibitor may also be useful in diseases where the induction of a cell stress or heat shock protein response could be beneficial, for example, protection from hypoxia-ischemic injury due to elevation of Hsp70 in the heart (Hutter, 1996 and Trost, 1998) and brain (Plumier, 1997 and Rajder, 2000). An Hsp90 inhibitor - induced increase in Hsp70 levels could also be useful in diseases where protein misfolding or aggregation is a major causal factor, for example, neurogenerative disorders such as scrapie/CJD, Huntingdon's and Alzheimer's (Sittler, 2001 ; Trazelt, 1995 and Winklhofer, 2001)".
Accordingly, the invention also includes:
(i) A pharmaceutical or veterinary composition comprising a compound of the invention, together with a pharmaceutically or veterinarily acceptable carrier.
(ii) The use of a compound a compound of the invention in the preparation of a composition for composition for inhibition of HSP90 activity in vitro or in vivo.
(iii). A method of treatment of diseases or conditions which are responsive to inhibition of HSP90 activity in mammals which method comprises administering to the mammal an amount of a compound of the invention effective to inhibit said HSP90 activity.
It will be understood that the specific dose level for any particular patient will depend upon a variety of factors including the activity of the specific compound employed, the age, body weight, general health, sex, diet, time of administration, route of administration, rate of excretion, drug combination and the causative mechanism and severity of the particular disease undergoing therapy. In general, a suitable dose for orally administrable formulations will usually be in the range of 0.1 to 3000 mg, once, twice or three times per day, or the equivalent daily amount administered by infusion or other routes. However, optimum dose levels and frequency of dosing will be determined by clinical trials as is conventional in the art.
The compounds with which the invention is concerned may be prepared for administration by any route consistent with their pharmacokinetic properties. The orally administrable compositions may be in the form of tablets, capsules, powders, granules, lozenges, liquid or gel preparations, such as oral, topical, or sterile parenteral solutions or suspensions. Tablets and capsules for oral administration may be in unit dose presentation form, and may contain conventional excipients such as binding agents, for example syrup, acacia, gelatin, sorbitol, tragacanth, or polyvinyl-pyrrolidone; fillers for example lactose, sugar, maize-starch, calcium phosphate, sorbitol or glycine; tabletting lubricant, for example magnesium stearate, talc, polyethylene glycol or silica; disintegrants for example potato starch, or acceptable wetting agents such as sodium lauryl sulphate. The tablets may be coated according to methods well known in normal pharmaceutical practice. Oral liquid preparations may be in the form of, for example, aqueous or oily suspensions, solutions, emulsions, syrups or elixirs, or may be presented as a dry product for reconstitution with water or other suitable vehicle before use.
Such liquid preparations may contain conventional additives such as suspending agents, for example sorbitol, syrup, methyl cellulose, glucose syrup, gelatin hydrogenated edible fats; emulsifying agents, for example lecithin, sorbitan monooleate, or acacia; non-aqueous vehicles (which may include edible oils), for example almond oil, fractionated coconut oil, oily esters such as glycerine, propylene glycol, or ethyl alcohol; preservatives, for example methyl or propyl p- hydroxybenzoate or sorbic acid, and if desired conventional flavouring or colouring agents.
For topical application to the skin, the drug may be made up into a cream, lotion or ointment. Cream or ointment formulations which may be used for the drug are conventional formulations well known in the art, for example as described in standard textbooks of pharmaceutics such as the British Pharmacopoeia.
The active ingredient may also be administered parenterally in a sterile medium. Depending on the vehicle and concentration used, the drug can either be suspended or dissolved in the vehicle. Advantageously, adjuvants such as a local anaesthetic, preservative and buffering agents can be dissolved in the vehicle.
Compounds of the invention may be administered together with other classes opf pharmaceutically active drugs. For example, for the treatment of cancers, combination therapy with two or more different classes of anticancer agent is a recognised and widespread practice. The present compounds may be used in such combination therapy, particularly where the other drug(s) have a mode of action different from HSP90 inhibition.
The following examples illustrate the preparation and activities of specific compounds of the invention and are not intended to be limiting of the full scope of the invention.
General Procedures
All reagents obtained from commercial sources were used without further purification.
Anhydrous solvents were obtained from commercial sources and used without further drying. Flash chromatography was performed with pre-packed silica gel cartridges (Strata SI-1; 61 A,
Phenomenex, Cheshire UK or 1ST Flash II, 54A, Argonaut, Hengoed, UK). Thin layer chromatography was conducted with 5 x 10 cm plates coated with Merck Type 60 F254 silica gel.
Some of the compounds of the present invention were characterized by LC/MS (Method A) using a Hewlett Packard 1100 series LC/MS linked to quadripole detector (ionization mode: electron spray positive or negative; column: Phenomenex Luna 3u C18(2) 30 x 4.6 mm; Buffer A prepared by dissolving 1.93g ammonium acetate in 2.5 L HPLC grade H2O and adding 2 mL formic acid. Buffer B prepared by adding 132 mL buffer A to 2.5 L of HPLC grade acetonitrile and adding 2 mL formic acid; elution gradient 95:5 to 5:95 buffer A : buffer.B over 3.75 minutes. Flow rate = 2.0 ml_/min) Retention Times (RT) are reported in minutes, lonisation is positive unless otherwise stated.
Some compounds of the invention were characterised by an alternative LC/MS method (Method B):
Details for method B:
Instrument: Waters 2695 pump and 2700 sample manager
Waters ZQ2000, M/z range 100 to 900 amu
Column: Gemini 5μm, C18 110A, 30 mm x 2mm from Phenomenex. Pt no 00A-
4435-BO
Temperature: Ambient Mobile Phase: A - Water + 10 mMol / ammonium formate + 0.04% (v/v) formic acid at pH ca 3.5
B - 100% Acetonitrile + 0.04% (v/v) formic acid Injection Volume 1OuL Gradient:
Detection: UV detection from 220 to 400nm (1 :3 split MS to UV)
Nuclear magnetic resonance (NMR) analysis was performed with a Brucker DPX-400 MHz NMR spectrometer at 400 MHz. The spectral reference was the known chemical shift of the solvent. Proton NMR data is reported as follows: chemical shift (δ) in ppm, multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, p = pentet, m = multiplet, dd = doublet of doublet, br = broad), integration, coupling constant.
Some compounds of the invention were purified by preparative HPLC. Preparative HPLC purifications were performed on a Waters FractionLynx MS Autopurification system with a Gemini® 5 μM C18(2), 100 mm * 20 mm i.d. column from Phenomenex, running at a flow rate of 20 mL min'1 with UV diode array detection (210 - 400 nm) and mass-directed collection. Various gradients (1 to 4) were used and these are shown in table 1. The pH of the eluant and the solvent gradient depends on the characteristics of the compound being purified.
At pH 4:Solvent A: HPLC grade Water + 1OmM ammonium acetate + 0.08% v/v formic acid.
Solvent B: 95% v/v HPLC grade acetonitrile + 5% v/v Solvent A + 0.08% v/v formic acid.
At pH 9: Solvent A: HPLC grade Water + 10 mM ammonium acetate + 0.08% v/v ammonia solution.
Solvent B: 95% v/v HPLC grade acetonitrile + 5% v/v Solvent A + 0.08% v/v ammonia solution..
The mass spectrometer was a Waters Micromass ZQ2000 spectrometer operating in positive or negative ion electrospray ionisation modes, with a molecular weight scan range of 150 to 1000.
Table 1 Preparative HPLC gradients
Chemical names were generated using AutoNom 2000.
Some compounds of the invention can be made (by way of example) by following the route outlined in scheme 1 (PG = protecting group). Suitable protecting groups, experimental methods, reagents and product isolation methods will be known to those skilled in the art of organic synthesis. It is understood that other methods can also be used.
Scheme 1
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide
Step 1 ^chloro^-methylsulfanyl-Z^-trimethylsilanyl-ethoxymethyO-ZH-pyrrolo^.S-dJpyrimidine
To a mixture of sodium hydride (276 mg; 6.89 mmol) in DMF (1OmL) at 00C was added drop- wise a solution of 4-chloro-2-methylsulfanyl-7H-pyrrolo[2,3-d]pyrimidine [prepared as detailed in Davoll. J. J. Chem. Soc. 1960, pp131-138] (1.145 g; 5.74 mmol) in anhydrous DMF (20 ml_). When addition was complete, 2-(trimethylsilyl)ethoxymethyl chloride (1.32 ml_; 7.46 mmol) was added drop-wise and the reaction mixture was stirred at 00C for 1.5 hours then allowed to warm to ambient temperature. The reaction mixture was partitioned between water (100 ml_) and ethyl acetate (100 ml_). The organic phase was dried over Na2SO4 then filtered and filtrate solvents evaporated in vacuo. The crude product was purified by flash chromatography on silica gel (7Og) eluting with a solvent gradient of 0 to 5% ethyl acetate in hexane to afford product as colourless oil (2.04g). LC/MS (METHOD A): RT = 2.88 min; m/z - 332, 330 [M+H]+. Total run time 3.75 mins.
Step 2
To a solution of 4-chloro-2-methylsulfanyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidine (0.5 g, 1.516 mmol) (example 1 step 2) in DMF (14 ml_) at O0C was added dropwise a solution of N-bromosuccinimide (270 mg, 1.516 mmol) in DMF (6 ml_). After 5 minutes the reaction was allowed to warm to ambient temperature. The solution was partitioned between EtOAc (2 x 40 mL) and water (40 mL). The combined organics were passed through a hydrophobic frit and evaporated in vacuo. The crude product was purified by flash chromatography on SiO2 (50 g) eluting with Hexane - 5% EtOAc/Hexane (gradient) to afford the desired product as a white solid, 433 mg, 70%. LC/MS (METHOD A): RT = 3.112 min; m/z = 410, 408 [M+H]\ Total run time 3.75 mins
Step 3
4-chloro-2-methylsulfanyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidine-5-carboxylic acid
To a solution of n-butyl lithium (2.5M in hexanes, 0.24 mL, 0.59 mmol) in THF (0.5 mL) at O0C was added slowly dropwise a solution of 5-Bromo-4-chloro-2-methylsulfanyl-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine (200mg, 0.489 mmol) in THF (2 mL). After 2 minutes, crushed solid CO2 was added and the mixture was left to warm to ambient temperature. Acetic acid was added then water (20 mL) and the mixture extracted with EtOAc (2 x 20 mL). The combined organics were passed through a hydrophobic frit and evaporated in vacuo to afford the desired crude product as a white solid, 167 mg, 91%. LC/MS (METHOD A): RT=2.664 min; m/z = 374 [M+H]\ Total run time 3.75 mins
Step 4
4-Chloro-2-methylsulfanyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidine-5-carboxylic acid amide
To a solution of 4-chloro-2-methylsulfanyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidiner5-carboxylic acid (100mg, 0.268 mmol) in CH2CI2 (1.5 mL) was added oxalyl, chloride (2M in CH2CI2, 0.17 mL, 0.349 mmol) followed by a few drops of DMF. After 10 min the reaction mixture was evaporated in vacuo then re-dissolved in CH2CI2 (3 mL). Aqueous ammonia solution (2 mL) was added and the mixture was stirred vigorously for 15 minutes. Water (10 mL), and CH2CI2 (10 mL) were added and the resultant phases separated. The aqueous phase was extracted with further CH2CI2 (15 mL). The combined organics were passed through a hydrophobic frit and evaporated in vacuo. The crude product was applied to a column of SiO2 (20 g) eluting with CH2CI2 - 5% MeOH/CH2CI2 (gradient) to afford the title compound as a yellow solid, 77 mg, 77%. LC/MS (METHOD A): RT = 2.47 min; m/z = 373, 375 [M+H]+. Total run time 3.75 mins.
Step 5
^chloro^-methylsulfanyl-Z^-trimethylsilanyl-ethoxymethyO-ZH-pyrrolop.S- d]pyrimidine-5-carbonitrile
To a solution of 4-chloro-2-methylsulfanyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidine-5-carboxylic acid amide (73 mg, 0.196 mmol) in CH2CI2 at O0C was added Et3N followed by TFAA (0.03 mL, 0.21 mmol) slowly dropwise. The stirred reaction mixture was the allowed to warm to ambient temperature. Further CH2CI2 (5 mL) was then added and the organic phase was washed with sat. NaHCO3 solution (15 mL). The organic layer was passed through a hydrophobic frit and evaporated in vacuo. The crude product was purified by flash chromatography on SiO2 (20 g) eluting with Hexane - 20% EtOAc/Hexane (gradient) to afford the title compound as a white solid, 60 mg, 86%. LC/MS (METHOD A): RT = 2.84 min; m/z = 357, 355 [M+H]+. Total run time 3.75 mins.
Step 6 5-Bromo-2-methoxy-phenol
To a solution of 5-Bromo-2-methoxy-benzaldehyde (15 g, 69.8 mmol) in CH2CI2 (200 ml_), mCPBA (19.0 g, 82.4 mmol) was added and the resultant mixture stirred at RT for 48 h. This was then partitioned between CH2CI2 (150 ml.) and sat. NaHCO3 solution (400 mL). The organic phase was dried (Na2SO4) and evaporated in vacuo. The residue was then dissolved in a minimum of EtOAc and passed through a plug of SiO2 washing through with further EtOAc. The filtrate was evaporated in vacuo and redissolved in MeOH (50 mL). 1M LiOH aq. solution (50 mL) was added and the mixture stirred for 10 min. 2M HCI (aq) was then added cautiously to acidify the reaction mixture to pH 6-7. This was extracted with EtOAc (3 x 100 mL) and the combined organics dried (Na2SO4) and evaporated in vacuo. The resultant crude product was purified by flash chromatography on SiO2 eluting with Hexane then 10% EtOAc/Hexane to afford the title compound as a white solid, 10.21 g, 72%. LC/MS (METHOD A): RT=2.11 Min; m/z = 201 , 203 [M-H]. Total run time 3.75 mins.
Step 7 5-Bromo-4-chloro-2-methoxy-phenol
To a solution of 5-Bromo-2-methoxy-phenol (10.08 g, 49.66 mmol) in MeCN (110 mL), TFA (1.15 mL, 14.9 mmol) and NCS (7.29 g, 54.63 mmol) were added sequentially and the resultant mixture stirred at RT for 16 h. This was then partitioned between EtOAc (200 mL) and brine (400 mL). The organic phase was dried (Na2SO4) and evaporated in vacuo. The resultant crude product was purified by flash chromatography on SiO2 eluting with Hexane then 10% EtOAc/Hexane to afford the title compound as a white solid, 10.5 g, 89%. LC/MS (METHOD A): RT=2.28 Min; m/z = 235, 237 [M-H]. Total run time 3.75 mins.
Step 8 1-Bromo-2<:hloro-4-methoxy-5-methoxyrnethoxy-benzene
To a solution of 5-Bromo-4-chloro-2-methoxy-phenol (1.0 g, 4.21 mmol) in dimethoxymethane (28 mL) and CHCI3 (28 mL) at O0C under N2 P2O5 (5.68 g, 40 mmol) was added in one portion. After 5 min. the reaction was allowed to warm to RT. The reaction mixture was then poured on to ice and extracted with CH2CI2 (2 x 50 mL). The combined organic phases were dried (Na2SO4) and evaporated in vacuo. The resultant crude product was purified by flash chromatography on SiO2 eluting with Hexane-10% EtOAc/Hexane (gradient) to afford the title compound as a white solid, 1.03 g, 87%. LC/MS (METHOD A): RT=2.57 Min; no mass detected. Total run time 3.75 mins.
Step 9
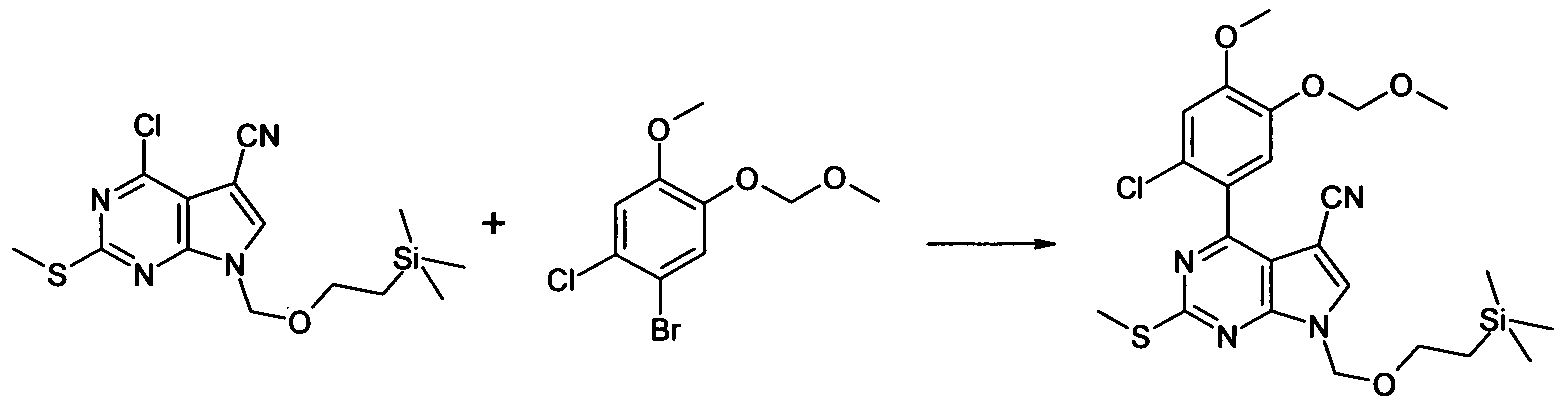
4-(2-Chloro-4-methoxy-5-methoxymethoxy-phenyl)-2-methylsulfanyl-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile
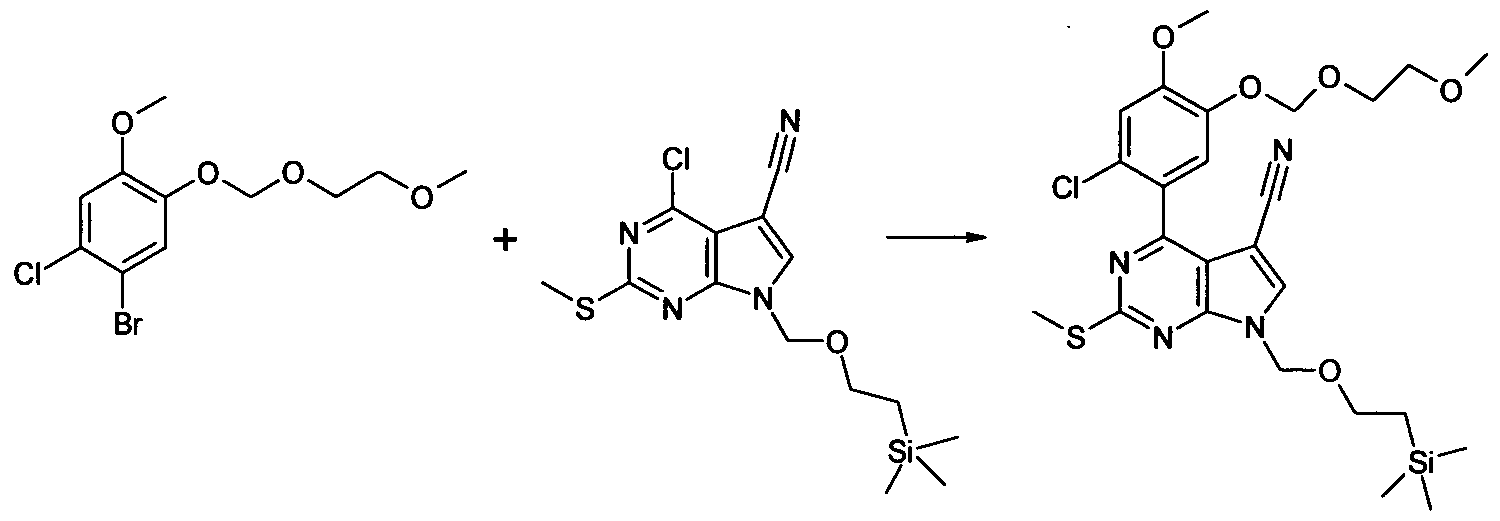
To a solution of 1-Bromo-2-chloro-4-methoxy-5-methoxymethoxy-benzene (2.27 g, 8.06 mmol) in THF at -780C under N2 triisopropylborate (3.72 mL, 16.12 mmol) was added followed by "BuLi (4.2 mL, 10.48 mL, 2.5N in Hexanes) dropwise. The reaction was then allowed to warm to room temperature and evaporated in vacuo. DMF, 4-chloro-2-methylsulfanyl-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile (2.0 g, 5.64 mmol), NaHCO3 (24 mL, 24.2 mmol, 1N aq. sol.), and PdCI2(PPh3J2 (280 mg, 0.4 mmol) were added sequentially and the resultant mixture degassed by bubbling N2 through it for 5 min. The reaction was then heated at 8O0C for 1.5h. The resultant mixture was then partitioned between EtOAc (250 mL) and brine
(250 ml_). The organic layer was dried (Na2SO4) and evaporated in vacuo to afford a crude oil. This was applied to a column of SiO2 (100g) eluting with Hexane - 30%EtOAc/Hexane (gradient) to afford the title compound as a yellow oil, 3.04 g, 98%. LC/MS (METHOD A): RT=2.84 Min; m/z = 521 , 523 [M+H]+. Total run time 3.75 mins.
Step 10
4-(2-Chloro-4-methoxy-5-methoxymethoxy-phenyl)-2-methanesulfonyl-7-(2- trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile
To an ice cooled solution of 4-(2-Chloro-4-methoxy-5-methoxymethoxy-phenyl)-2- methylsulfanyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile (3.04 g, 5.5 mmol) in CH2CI2 (100 mL) mCPBA (4.93 g, 22 mmol) dissolved in CH2CI2 (70 mL) was added dropwise. After the addition was complete the reaction was allowed to warm to RT. It was then washed sequentially with aq. sodiumthiosulfate solution (150 mL) and saturated NaHCO3 sol. (170 mL). The organic layer was dried (Na2SO4) and evaporated in vacuo to afford a crude oil. This was applied to a column of SiO2 (5Og) eluting with Hexane - 40%EtOAc/Hexane (gradient) to afford the title compound as a yellow foam, 2.5 g, 80%. LC/MS (METHOD A): RT=2.62 Min; m/z = 553, 555 [M+H]+. Total run time 3.75 mins.
Stepi 1
[4-(2-ChIoro-4-methoxy-5-methoxymethoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-acetic acid ethyl ester
Ethyl thioglycolate (0.31 ml_, 2.78 mmol) was added to a mixture of NaH (46 mg, 1.15 mmol, 60% dispersion in mineral oil) in THF (10 ml.) at O0C under N2. 4-(2-Chloro-4-methoxy-5- methoxymethoxy-phenyl)-2-methanesulfonyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidine-5-carbonitrile (1.28 g, 2.32 mmol) dissolved in THF (10 mL) was then added dropwise. The reaction was then allowed to warm to RT before being partitioned between EtOAc (2 x 100 mL) and aq. NH4CI sol. (100 mL). The organic layer was dried (Na2SO4) and evaporated in vacuo to afford a crude oil. This was applied to a column of SiO2 (70 g) eluting with Hexane - 40%EtOAc/Hexane (gradient) to afford the title compound as a yellow oil, 1.32 g, 96%. LC/MS (METHOD A): RT=2.84 Min; m/z = 593, 595 [M+H]+. Total run time 3.75 mins.
Step12
[4-(2-Chloro-4-methoxy-5-methoxymethoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-acetic acid
1 N NaOH aq. (4.46 mL, 4.46 mmol) was added to a solution of [4-(2-Chloro-4-methoxy-5- methoxymethoxy-phenyl)-5-cyano-7-(2-trimethyIsilanyl-ethoxymethyl)-7H-pyrrolo[2,3- d]pyrimidin-2-ylsulfanyl]-acetic acid ethyl ester (1.32 g, 2.23 mmol) in MeOH (20 mL) and stirred at RT for 2 h. The mixture was then evaporated in vacuo, water added (10OmL) and the resultant solution acidified to pH 5 by cautious addition of 1.2 N HCI sol. This misture was then extracted with EtOAc (2 x 100 mL). The combined organics were dried (Na2SO4) and evaporated in vacuo to afford the title compound as a yellow foam (1.27 g, 100%).
LC/MS (METHOD A): RT=2.63 Min; m/z = 565, 567 [M+H]+. Total run time 3.75 mins.
Step 13
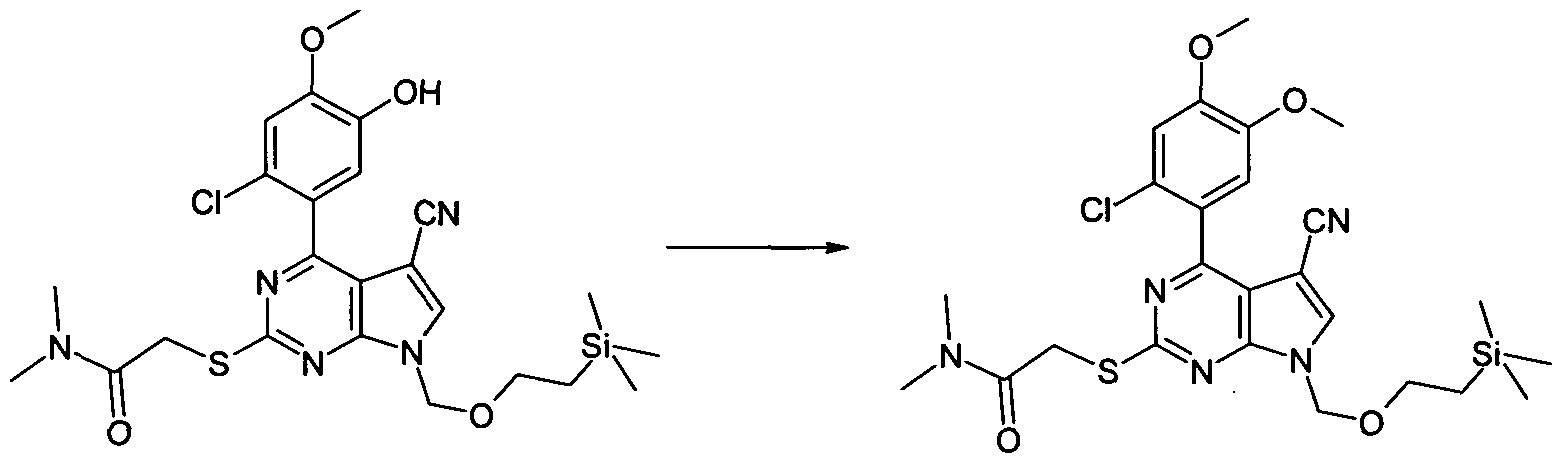
2-[4-(2-Chloro-4-methoxy-5-methoxymethoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide
To a solution of [4-(2-Chloro-4-methoxy-5-methoxymethoxy-phenyl)-5-cyano-7-(2- trimethylsilanyl-ethoxymethyO^H-pyrrolo^.S-dlpyrimidin^-ylsulfanyll-acetic acid (500 mg, 0.885 mmol) in anhydrous MeCN (10 ml.) triethylamine (0.19 mL, 1.33 mmol), dimethylamine (0.67 mL, 1.33 mmol, 2 M solution in THF) and HBTU (O-(benzotriazol-1-yl)-N,N,N\N'- tetramethyluronium hexafluorophosphate (504 mg, 1.33 mmol) were added sequentially. After 1 h the reaction mixture was partitioned between EtOAc (2 x 50 mL) and NH4CI solution (50 mL). The combined organics were dried (Na2SO4) and evaporated in vacuo. The resultant crude oil was dissolved in a minimum of EtOAc and applied to a column of SiO2 (50 g) eluting with Hexane-80% EtOAc/hexane (gradient) to afford the title compound as a white foam,670 mg,. 100%. LC/MS (METHOD A): RT=2.67 min; m/z = 592, 594 [M+H]+. Total run time 3.75 mins.
Step 14
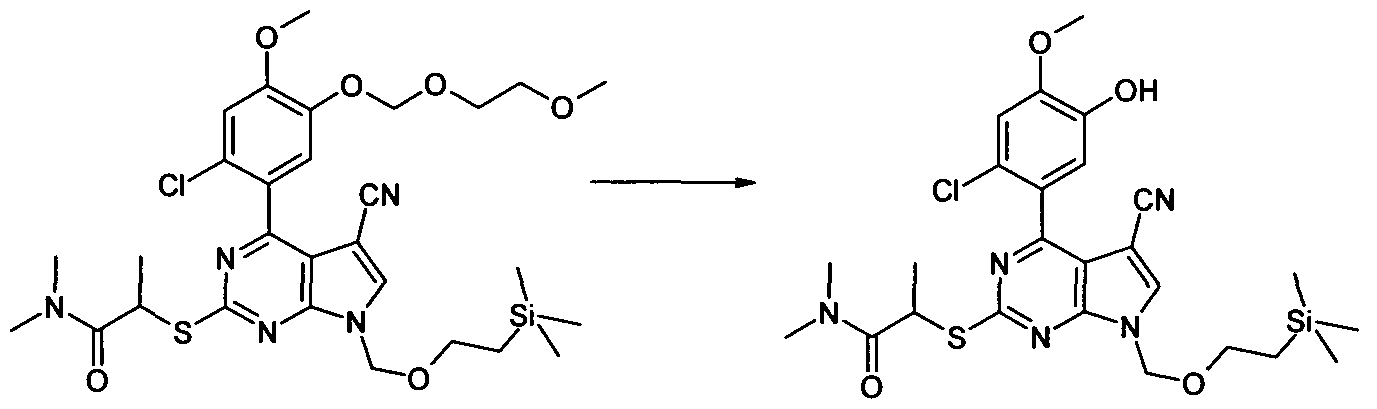
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7-(2-trimethy1silanyl-ethoxymethyl)-
7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide
To a solution of 2-[4-(2-Chloro-4-methoxy-5-methoxymethoxy-phenyl)-5-cyano-7-(2- trimethylsilanyl-ethoxymethyO-yH-pyrrolop.S-dJpyrimidin^-ylsulfanyll-N.N-dimethyl-acetamide (628 mg, 1.06 mmol) in 'PrOH (10 mL) PPTS (pyridinium p-toluenesulfonate) (53 mg, 0.212 mmol) was added and the mixture heated at 850C under N2 overnight. The reaction was allowed to cool and partitioned between EtOAc (2 x 20 mL) and NH4CI solution (20 mL). The dried (Na2SO4) organics were evaporated in vacuo to give a crude oil. This was applied to a cartridge of SiO2 (50 g) eluting with CH2CI2 - 5% MeOH/ CH2CI2 (gradient) to afford the title compound as a white foam, 411 mg, 71 %. LC/MS (METHOD A): RT=2.53 min; m/z = 548, 550 [M+H]+. Total run time 3.75 mins.
Step 15
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N,N-dimethyl-acetamide
To 2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H- pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide (60 mg, 0.109 mmol) under N2, anhydrous THF (2 mL) ethylene diamine (0.022 mL, 0.327 mmol) and 1N TBAF in THF (0.65 mL, 0.654 mmol) were added sequentially. The mixture was heated at 4O0C overnight. The reaction was allowed to cool and was then partitioned between EtOAc (30 mL) and water (30 mL). The organic layer was dried (Na2SO4) and evaporated in vacuo to give a crude oil (-60 mg). This was applied to a cartridge of SiO2 (10 g) eluting with Hexane - 100% EtOAc (gradient) to afford the title compound as a white solid, 16 mg, 35%.
LC/MS (METHOD A): RT=1.86 min; m/z = 418, 420 [M+H]\ Total run time 3.75 mins.
1H NMR (DMSO-cfe): δ 2.85 (s, 3H); 3.09 (s, 3H); 3.87 (s, 3H); 4.19 (s, 2H); 6.89 (s, 1 H); 7.14 (s,
1 H); 8.41 (s, 1H); 9.59 (s, 1H); 13.13 (brs, 1H).
Example 2
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]- N,N-dimethyl-acetamide
Step i
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H- pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide
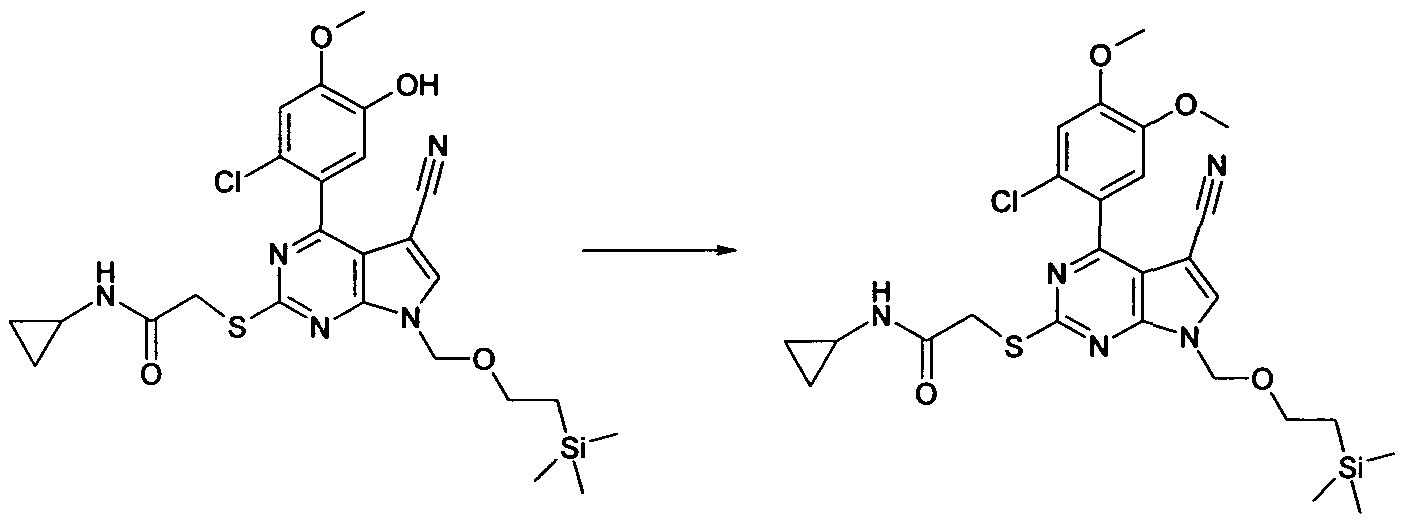
To a mixture of 2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide (80 mg, 0.15 mmol), Cs2CO3 (71 mg, 0.22 mmol) and anhydrous DMF (2 mL) under N2 was added MeI (0.01 ml_, 0.15mmol). The reaction was allowed to stir at ambient temperature for 2h. The reaction mixture was then partitioned between EtOAc (20 mL) and NH4CI solution (20 mL). The organic layer was washed with brine (20 mL) then dried (Na2SO4) and evaporated in vacuo to afford the title compound as a yellow oil, 96 mg, 100%. LC/MS (METHOD A): RT=2.64 min; m/z = 562, 564 [M+H]+. Total run time 3.75 mins.
Step 2
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyI]-
The title compound was prepared using the route outlined in scheme 1 and methods outlined in example 1 , step 15.
LC/MS (METHOD A): RT=1.99 min; m/z = 432, 434 [M+H]+. Total run time 3.75 mins. 1H NMR (DMSO-CZ6): δ 2.85 (s, 3H); 3.09 (s, 3H); 3.80 (s, 3H); 3.87 (s, 3H); 4,23 (s, 2H); 7.14 (s, 1 H); 7.19 (s, 1H); 8.44 (s, 1H); 13.16 (brs, 1H).
Example 3
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- cyclopropyl-acetamide
Step i
4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-2-methylsulfanyl-7-(2- trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile
The title compound was prepared using the route outlined in scheme 1 and using the methods outlined in example 1 , step 9. 1-Bromo-2-chloro-4-methoxy-ethoxymethoxybenzene was made from 5-Bromo-4-chloro-2-methoxy-phenol (example 1 , step 7) by reacting with 2- methoxyethoxymethyl chloride. LC/MS (METHOD A): RT=2.86 min; m/z = 565, 567 [M+H]+. Total run time 3.75 mins.
Step 2
4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-2-methanesulfonyl-7-(2- trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile
The title compound was prepared using the route outlined in scheme 1 and using the methods outlined in example 1 , step 10.
LC/MS (METHOD A): RT=2.63 min; m/z = 597, 599 [M+H]+. Total run time 3.75 mins.
Step 3
[4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5-cyano-7-(2- trimethylsilanyl-ethoxymethyO^H-pyrrolo^.S-dJpyrimidin^-ylsulfanyll-acetic acid ethyl ester
The title compound was prepared using the route outlined in scheme 1 and using the methods outlined in example 1 , step 11.
LC/MS (METHOD A): RT=2.82 min; m/z = 637, 639 [M+H]+. Total run time 3.75 mins.
Step 4
[4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5-cyano-7-(2- trimethylsilanyl-ethoxymethyO^H-pyrrolo^.S-dlpyrimidin^-ylsulfanylJ-acetic acid
/
The title compound was prepared using the route outlined in scheme 1 and using the methods outlined in example 1 , step 12.
LC/MS (METHOD A): RT=2.62 min; m/z = 609, 611 [M+H]\ Total run time 3.75 mins.
Step 5
2-[4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5-cyano-7-(2- trimethylsilanyl-ethoxymethyO^H-pyrrolop.S-dJpyrimidin^-ylsulfanylJ-N-cyclopropyl- acetamide
To a solution of the acid (679 mg, 1.2 mmol, 1 eq) in anhydrous MeCN (30 ml.) triethylamine (0.25 ml_, 1.8 mmol, 1.5 eq), cyclopropylamine (0.125 ml_, 1.8 mmol, 1.5 eq) and HBTU (684 mg, 1.8 mmol, 1.5 eq) were added sequentially. After 1h the reaction mixture was partitioned between EtOAc (2x80 mL) and NH4CI solution (80 mL). The combined organics were dried
(Na2SO4) and evaporated in vacuo. The resultant crude oil was dissolved in a minimum of EtOAc and applied to a column of SiO2 (7Og) eluting with Hexane-60% EtOAc/hexane (gradient) to afford the title compound as a white foam,770 mg, 100%. LC/MS (METHOD A): RT=2.66 min; m/z = 604, 606 [M+H]\ Total run time 3.75 mins.
Step 6
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-
7H-pvrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-cyclopropyl-acetamide
To a solution of the amide (770 mg, 1.2 mmol) in 1PrOH (11 mL) PPTS (302 mg, 1.2 mmol, 1 eq) was added and the mixture heated at 850C under N2 overnight. The reaction was allowed to cool and partitioned between EtOAc (70 mL) and NH4CI solution (70 mL). The dried (Na2SO4) organics were evaporated in vacuo to give a crude oil (~1g). This was dissolved in MeOH and pre-absorbed onto silica (10 mL). This was applied to a cartridge of SiO2 (50 g) eluting with 30%EtOAc - 80% EtOAc/Hexane (gradient) to afford the title compound as a colourless oil, 515 mg, 77%. LC/MS (METHOD A): RT=2.56 min; m/z = 560, 562 [M+H]+. Total run time 3.75 mins.
Step 7
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H- pyrrolofrS-dlpyrimidin^-ylsulfanyll-N-cyclopropyl-acetamide
To a mixture of the alcohol (80 mg, 0.14 mmol), Cs2CO3 (49 mg, 0.15 mmol) and anhydrous DMF (2 mL) under N2 was added MeI (0.01 ml_, 0.15mmol). The reaction was allowed to stir at RT for 1h. The reaction mixture was then partitioned between EtOAc (20 mL) and NH4CI solution (20 mL). The organic layer was washed with brine (20 mL) then dried (Na2SO4) and evaporated in vacuo to give a crude oil (-100 mg). This was dissolved in a minimum of CH2CI2 and applied to a cartridge of SiO2 (10 g) eluting with CH2CI2 - 5% MeOH / CH2CI2 (gradient) to afford the title compound as a colourless oil, 76 mg, 93%. LC/MS (METHOD A): RT=2.64 min; m/z = 574, 576 [M+H]+. Total run time 3.75 mins.
Step 8
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-yIsulfanyl]-N- cyclopropyl-acetamide
The title compound was prepared using the route outlined in scheme 1 and using the methods outlined in example 1, step 15.
LC/MS (METHOD A): RT=2.02 min; m/z = 444, 446 [M+H]+. Total run time 3.75 mins.
1H NMR (DMSO-de): δ 0.36 (m, 2H); 0.58 (m, 2H); 2.6 (m, 1H); 3.81 (s, 3H); 3.87 (s, 3H); 3.88
(s, 2H); 7.13 (s, 1H); 7.19 (s, 1 H); 8.17 (d, 1H, J = 4.0 Hz); 8.45 (s, 1H); 13.14 (brs, 1 H).
Example 4
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-cyclopropyl-acetamide
The title compound was prepared from 2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7-
(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-cyclopropyl- acetamide (example 3, step 6) using the methods outlined in example 1, step 15 (removal of
SEM protecting group).
LC/MS (METHOD A): RT=1.90 min; m/z = 430, 432 [M+H]+. Total run time 3.75 mins.
1H NMR (DMSO-de): δ 0.37 (m, 2H); 0.59 (m, 2H); 2.62 (m, 1H); 3.87 (s, 5H); 6:89 (s, 1H); 7.14
(s, 1H); 8.18 (d, 1H, J = 3.0 Hz); 8.42 (s, 1H); 9.58 (brs, 1H); 13.11 (brs, 1H).
Example 5
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-cyclopropyl -acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT=2.01 min; m/z = 458, 460 [M+H]+. Total run time 3.75 mins.
1H NMR (DMSO-de): δ 0.36 (m, 2H); 0.58 (m, 2H); 1.33 (t, 3H, J = 7.0 Hz); 2.61 (m, 1 H); 3.87 (s,
3H); 3.88 (s, 2H); 4.05 (q, 2H, J = 7.0 Hz); 7.11 (s, 1H); 7.18 (s, 1H); 8.17 (d, 1H, J = 4.0 Hz);
8.44 (s, 1 H); 13.13 (brs, 1H).
Example 6
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- isopropyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT=1.996 minutes m/z = 446.1 , 448.1 [M+H]+ (Run time 3.75mins) 1H NMR (DMSO-CZ6) δ 1.00 (d, 6H, J = 6.3 Hz), 3.79 (m, 1H), 3.80 (s, 3H), 3.86 (s, 3H), 3.89 (s, 2H), 7.14 (s, 1H), 7.19 (s, 1H), 7.95 (brd, 1H, J = 7.5 Hz), 8.44 (s, 1H), 13.06 (brs, 1H).
Example 7
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-isopropyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT=1.657 minutes, m/z = 432.1 [M+H]+ (Run time 3.75mins)
1H NMR (DMSO-CZ6) δ 1.01 (d, 6H, J = 6.6 Hz), 3.81 (m, 1H), 3.87 (s, 3H), 3.88 (s, 2H), 6.90 (s,
1H), 7.14 (s, 1H), 7.95 (brd, 1 H, J = 7.6 Hz), 8.42 (s, 1H), 9.57 (s, 1H), 13.11 (brs, 1H).
Example 8
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-isopropyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT = 2.089 minutes, m/z = 460.1/ 462.1 [M+H]+ (Run time 3.75mins) 1H NMR (DMSO-CZ6) δ 1.00 (d, 6H, J = 6.8 Hz), 1.32 (t, 3H, J = 7.1 Hz), 3.81 (m, 1H), 3.86 (s, 3H), 3.89 (s, 2H), 4.05 (q, 2H, J = 7.1 Hz), 7.12 (s, 1 H), 7.18 (s, 1 H), 7.94 (brd, J = 7.6 Hz), 8.44 (s, 1H), 13.1 (brs, 1H).
Example 9
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- ethyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT = 1.697 minutes, m/z = 432.1 [M+H]+ (Run time 3.75mins)
1H NMR (DMSO-CZ6) δ 0.97 (t, 3H, J = 7.3 Hz), 3.08 (m, 2H), 3.80 (s, 3H), 3.87 (s, 3H), 3.91 (s,
2H), 7.13 (s, 1H), 7.19 (s, 1H), 8.08 (t, 1 H, J = 5.0 Hz), 8.44 (s, 1H), 13.08 (brs, 1H).
Example 10
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-ethyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT = 1.797 minutes, m/z = 446.1 , 448.1 [M+H]+ (Run time 3.75mins) 1H NMR (DMSO-Cf6) δ 0.97 (t, 3H, J = 7.3 Hz), 1.33 (t, 3H, J = 6.7 Hz), 3.07 (m, 2H), 3.87 (s, 3H), 3.91 (s, 2H), 4.05 (q, 2H, J = 6.7 Hz), 7.10 (s, 1H), 7.18 (s, 1 H), 8.07 (t, 1 H, J = 5.0 Hz), 8.43 (s, 1 H), 13.10 (brs, 1H).
Example 11
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-ethyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT= 1.572 minutes, m/z = 418.1 [M+H]+ (Run time 3.75mins)
1H NMR (DMSO-Gf6) δ 0.98 (t, 3H, J = 7.3 Hz), 3.09 (m, 2H), 3.87 (s, 3H), 3.89 (s, 2H), 6.89 (s,
1 H), 7.13 (s, 1 H), 8.08 (t, 1 H, J = 5.0 Hz), 8.42 (s, 1 H), 9.58 (brs, 1 H), 13.11 (brs, 1H).
Example 12
N-tert-Butyl^-^-tZ-Chloro-S-hydroxy^-methoxy-phenyO-δ-cyano-TH-pyrrolop.S- d]pyrimidin-2-ylsulfanyl]-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD B): RT= 3.00 minutes, m/z = 446 [M+H]+ (Run time 5.20 mins)
1H NMR (MeOD) δ 8.12 (s, 1H), 7.11 (s, 1H), 6.97 (s, 1 H), 3.94 (s, 3H), 3.83 (s, 2H), 1.22 (s, 9H).
Example 13
N-tert-Butyl-2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanylj-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD B): RT= 3.13 minutes, m/z = 460 [M+H]+ (Run time 5.20 mins)
1H NMR (MeOD) δ 8.13 (s, 1 H), 7.67 (s, 1H), 7.14 (s, 2H), 3.92 (s, 3H), 3.87 (s, 3H), 3.85 (s,
2H), 1.22 (s, 9H).
Example 14
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-(2-fluoro-ethyl)-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD B): RT= 2.85 minutes, m/z = 436 [M+H]+ (Run time 5.20 mins)
1H NMR (DMSOd6) δ 3.35 (m, 1H), 3.42 (m, 1H), 3.86 (s, 3H), 3.95 (s, 2H), 4.33(m, 1H), 4.45
(m, 1H), 6.90 (s, 1H), 7.15 (s, 1H), 8.35 (m, 1H), 8.42 (s, 1H), 9.56 (s, 1H), 13.0-13.2 (brs, 1H).
Example 15
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-
(2-fluoro-ethyl)-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD B): RT= 2.94 minutes, m/z = 450 [M+H]+ (Run time 5.20 mins)
1H NMR (CDCI3) δ 3.51-3.60 (m, 2H1 CH2), 3.90-3.98 (m, 8H, 2 OCH3 + CH2), 4.35 (m, 1H), 4.45
(m, 1H), 7.00 (s, 1H1), 7.05 (s, 1H1), 7.38 (m, 1H), 7.81 (s, 1H), 10.60-11.00 (brs, 1H).
Example 16
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N-methyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD A): RT=1.79 min; m/z = 404, 406 [M+H]+. Total run time 3.75 mins.
1H NMR (DMSOd6): δ 2.61 (d, 3H, J = 4.5 Hz); 3.87 (s, 3H); 3.90 (s, 2H); 6.88 (s, 1H); 7.14 (s,
1 H); 8.03 (brs, 1H); 8.42 (s, 1H); 9.58 (s, 1H); 13.15 (brs, 1H).
Example 17
2-{4-[2-Chloro-5-(2-diethylamino-ethoxy)-4-methoxy-phenyl]-5-cyano-7H- pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl}-N,N-dimethyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3. Specifically 2-[4-(2-Chloro-5-hydroxy- 4-methoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N,N-dimethyl-acetamide (example 1 , step 14) was alkylated with 2-bromoethyl diethylamine followed by protecting group removal using by the method of example 1 step 15. LC/MS: RT=1.64 min; m/z = 517, 519 [M+H]+. Total run time 3.75 mins. 1H NMR (DMSOd6): δ 0.97 (d, 6H, J = 7.1 Hz), 2.59 (q, 4H, J = 7.1 Hz), 2.85 (s, 3H), 2.86 (m, 2H), 3.09 (S, 3H), 3.86 (s, 3H), 4.07 (t, 2H, J = 6.1 Hz), 4.22 (s, 2H)1 7.18 (s, 1H), 7.20 (s, 1H), 8.43 (s, 1H), 12.78 (brs 1H).
Example 18
2-{4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxy)-phenyl]-5-cyano-7H-pyrro lo[2,3-d]pyrimidin-2-ylsulfanyl}-N-methyl-acetamide
Step i
2-[4-(2-Chloro-4-methoxy-5-(2-methoxy-ethoxyl)-phenyl]-5-cyano-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-methyl-acetamide
2-methoxyethanol (20 μL; 0.224 mmol) was added to a mixture of 2-[4-(2-Chloro-5-hydroxy-4- methoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-N-methyl-acetamide (100 mg, 0.187 mmol; prepared by the methods outlined for example 1 ) and triphenylphosphine (74 mg; 0.281 mmol) in THF (3 ml_). After stirring at ambient temperature for 5 minutes, the reaction mixture was cooled with an ice-water bath and diisopropyldiazodicarboxylate (55 μL, 0.281 mmol) was added and reaction mixture allowed to warm to ambient temperature and stir for 1.5 h. The reaction mixture was partitioned between ethyl acetate (20 mL) and saturated aqueous ammonium chloride solution (20 ml_). The organic phase was separated and dried over anhydrous sodium sulphate, filtered and filtrate solvents removed in vacuo to afford a crude product which was purified by flash chromatography on silica gel (10 g), eluting with a gradient of dichloromethane to 5% methanol in dichloromethane to afford title compound as a pale yellow oil. LC/MS: RT=2.57 min; m/z = 592, 594 [M+H]+. Total run time 3.75 mins.
Step 2
2-{4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxy)-phenyl]-5-cyano-7H-pyrro lo[2,3-d]pyrimidin-2-ylsulfanyl}-N-methyl-acetamide
The title compound was prepared using the method outlined in example 1, step 15.
LC/MS: RT=1.79 min; m/z = 404, 406 [M+H]+. Total run time 3.75 mins.
1H NMR (d6 DMSO): 52.60 (d, 3H, J = 5.0 Hz); 3.30 (s, 3H); 3.66 (t, 2H, J = 4.5 Hz); 3.87 (s,
3H); 3.91 (s, 2H); 4.12 (t, 2H, J = 4.5 Hz); 7.13 (s, 1H); 7.20 (s, 1H); 8.02 (m, 1H); 8.44 (s, 1H);
13.18 (brs, 1H).
Example 19
2-{4-[2-Chloro-5-(2-hydroxy-ethoxy)-4-methoxy phenyl]-5-cyano-7H-pyrro lo[2,3-d]pyrimidin-2-ylsulfanyI}-N-methyl-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS: RT=1.76 min; m/z = 448 [M+H]+. Total run time 3.75 mins.
1H NMR (MeOD): δ 2.77 (s, 3H), 3.92 (t, 2H, J = 4.5 Hz), 3.98 (s, 3H), 3.98 (s, 2H), 4.15 (t, 2H, J
= 4.5 Hz), 7.16 (s, 1H), 7.20 (s, 1H), 8.16 (s, 1H).
Example 20
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-
(2,2,2-trifluofluoro-ethyl)-acetamide
The title compound was prepared using the route outlined in scheme 1 using appropriate reagents and methods as outlined in Examples 1 , 2 and 3.
LC/MS (METHOD B): RT= 3.23 minutes, m/z = 486 [M+H]+ (Run time 5.20 mins)
1H NMR (CDCI3) δ 3.85 (m, 2H), 3.93 (s, 3H), 3.95 (s, 3H), 3.96 (s, 2H), 7.00 (s, 1H), 7.04 (s,
1 H), 7.56 (t, 1H, J = 7.0 Hz), 10.88 (brs, 1H).
Example 21
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N,N-dimethyl-propionamide
Stepi
[4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5-cyano-7-(2- trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-propionic acid ethyl ester
To a stirred suspension of sodium hydride (24 mg, 60% in oil, 0.60 mmol) at 00C in anhydrous THF (4.5 ml.) was added 2-mercaptopropionic acid ethyl ester (0.078 mL, 0.60 mmol) followed by a solution of the 4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-2- methanesulfonyl-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile (example 3 step 2) (300 mg, 0.50 mmol) in THF (3 mL). The mixture was allowed to warm to ambient temperature and stirred for 1.5 hours. The reaction mixture was diluted with water (50 mL) then extracted with EtOAc (2 * 50 mL). The combined organic extracts were washed with brine (100 mL), dried (Na2SO4), filtered and evaporated in vacuo to a crude yellow oil that was used in the next reaction without further purification or characterisation.
Step 2
[4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5-cyano-7-(2- trimethylsilanyl-ethoxymethyO^H-pyrroloP.S-dlpyrimidin^-ylsulfanyll-propionic acid
[4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5-cyano-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-propionic acid ethyl ester was dissolved in a mixture of methanol (10 mL) and 2N NaOH aq. (10 mL) and stirred for 18 hours. The reaction mixture was reduced in vacuo to approximately 10 mL then partitioned between water (30 mL) and EtOAc (30 mL). The organic layer was discarded. The aqueous layer was acidified to pH 2 by addition of 1.2 N HCI aq. and then extracted with EtOAc (2 * 30 mL). The combined organic extracts were washed with brine (60 mL), dried (Na2SO4), filtered and evaporated in vacuo to afford the title compound as a colorless oil which was used crude in the next reaction without further purification. LC/MS (METHOD A): RT=2.61 Min; m/z = 624 [M+H]+. Total run time 3.75 mins.
Step 3
2-[4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5-cyano-7-(2- trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N-dimethyl- propionamide
To a solution of the crude [4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxymethoxy)-phenyl]-5- cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-propionic acid (assume 0.50 mmol) in anhydrous MeCN (10 ml) was sequentially added triethylamine (0.21 ml, 1.5 mmol), dimethylamine hydrochloride (63 mg, 0.75 mmol) and HBTU (288 mg, 0.75 mmol) then the reaction mixture was stirred for 18 hours. The reaction mixture was partitioned between EtOAc (50ml) and saturated NaHCO3 sol. (50 ml). The organic extract was washed with saturated brine (50 ml_), dried (Na2SO4), filtered and evaporated in vacuo. The resultant crude oil was purified by flash column chromatography over silica gel (eluting with 2:1 to 3:1 EtOAc/hexane) to afford the title compound as a white foam, 48 mg, 15% over 3 steps. LC/MS (METHOD A): RT=2.62 Min; m/z = 650, 652 [M+H]+. Total run time 3.75 mins.
Step 4
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-
7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N-dimethyI-propionamide
To a solution of the amide (80 mg, 0.12 mmol) in 1PrOH (3 ml) was added PPTS (31 mg, 0.12 mmol) and the mixture heated at 850C under N2 for 18 hours. The reaction was allowed to cool and partitioned between EtOAc (30 ml) and saturated NaHCO3 sol. (30 ml). The organic extract was washed with saturated brine (30 mL) and then dried (Na2SO4), filtered and evaporated in
vacuo to give a crude oil. Purification by flash column chromatography over silica gel (eluting with 1 :1 to 2:1 EtOAc/hexane) afforded the title compound as a white foam, 44 mg, 64%. LC/MS (METHOD A): RT=2.53 Min; m/z = 562, 564 [M+H]+. Total run time 3.75 mins.
Step 5
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N,N-dimethyl-propionamide
To a solution of the pyrrolopyrimidine (44 mg, 0.078 mmol) under N2 in anhydrous THF (2 ml) was added sequentially ethylene diamine (0.0154 ml, 0.234 mmol) then 1N TBAF in THF (0.46 ml, 0.46 mmol). The mixture was then heated at 4O0C for 24 hours. The reaction mixture was then diluted with saturated NaHCO3 sol. (30 mL) and the resultant mixture extracted with EtOAc (2 * 30 ml). The organic extracts were combined and washed with saturated brine (60 mL), dried (Na2SO4), and then evaporated in vacuo to give a crude oil. Purification by flash column chromatography (eluting with 2:1 EtOAc/hexane then neat EtOAC) afforded the title compound as a white foam, 20 mg, 59%.
LC/MS (METHOD A): RT=1.85 Min; m/z = 432, 434 [M+H]\ Total run time 3.75 mins. 1H NMR (DMSO-cfe): δ 1.49 (d, J = 6.9 Hz, 3H); 2.85 (s, 3H); 3.08 (s, 3H); 3.87 (s, 3H); 4.97 (q, J = 6.9 Hz, 1H); 6.88 (s, 1H); 7.14 (s, 1 H); 8.42 (s, 1H); 9.59 (s, 1H); 13.15 (brs, 1H).
Example 22
2-[4-[2-Chloro-4-cyano-5-methoxy-phenyl]-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulphanyl]-N-isopropyl-acetamide
Step i 4-Amino-5-chloro-2-methoxybenzamide
4-amino-5-chloro-2-methoxybenzoic acid (commercially available) (600mg, 2.98 mmol) was added to sulfamide (715 mg; 7.44 mmol, 2.5 equiv) and the mixture was then dissolved in pyridine (2.9 ml) and heated under nitrogen atmosphere for 2.5 hours. Reaction mixture was allowed to cool to ambient temperature and the pyridine was removed in vacuo. The resulting solids were washed with 10% MeOH in dichloromethane. Filtered and dried to afford a cream solid 550mg; 92%.
Step 2 4-Amino-5-chloro-2-methoxybenzonitrile
4-Amino-5-chloro-2-methoxybenzamide was added to acetonitrile) and POCI3 (excess) was added to the resulting suspension and this mixture heated to 80 0C for 3 hours (reaction mixture was homogeneous after 1.5 hours). Reaction mixture was allowed to cool to room temperature, then poured into ice water. After stirring for 2 hours a yellow solid was filtered off, this was dried overnight at 500C.
Step 3 4-lodo-5-chloro-2-methoxybenzonitrile
The title compound was made by way of method of example 22 step 3 (diazotization and quench with aqueous Iodine / sodium iodide solution).
Step 4
4-(2-Chloro-4-cyano-5-methoxy-phenyl)-2-methylsulfanyl-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile
The title compound was prepared by way of the method of example 26 step 1 (boronic acid formation and subsequent cross coupling).
LC/MS (METHOD A): RT=2.884 min; m/z = 486, 488 [M+H]\ Total run time 3.75 mins.
Step 5 4-(2-Chloro-4-cyano-5-methoxy-phenyl)-2-methanesulfonyl-7-(2-trimethylsilanyl- ethoxymethyl)-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile
LC/MS (METHOD A): RT=2.581 min; m/z = 518, 520 [M+H]+. Total run time 3.75 mins.
Step 6 [4-[2-Chloro-4-cyano-5-methoxy-phenyl]-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H- pyrrolo[2,3-d]pyrimidin-2-ylsulphanyl]-acetic acid ethyl ester
The title compound was prepared by way of the method of example 1 step 11. LC/MS (METHOD A): RT=2.768 min; m/z = 558, 560 [M+H]+. Total run time 3.75 mins.
Step 7
[4-[2-Chloro-4-cyano-5-methoxy-phenyl]-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-7H- pyrrolo[2,3-d]pyrimidin-2-ylsulphanyl]-acettc acid
The title compound was prepared by way of the method of example 1 step 12. LC/MS (METHOD A): RT=2.586 min; m/z = 530, 532 [M+H]+. Total run time 3.75 mins.
Step 8
2-[4-[2-Chloro-4-cyano-5-methoxy-phenyl]-5-cyano-7-(2-trimethylsilanyl-ethoxymethyl)-
7H-pyrrolo[2,3-d]pyrimidin-2-ylsulphanyl]-N-isopropyl-acetamide
The title compound was prepared by way of the method of example 1 step 13. LC/MS (METHOD A): RT=2.645 min; m/z = 571, 573 [M+H]+. Total run time 3.75 mins.
Step 9
2-[4-[2-Chloro-4-cyano-5-methoxy-phenyl]-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulphanyl]-N-isopropyl-acetamide
The title compound was prepared by way of the method of example 1 step 15.
LC/MS (METHOD A): RT=2.103 min; m/z = 441 [M+H]+. Total run time 3.75 mins.
1H NMR (DMSO-d6): δ 1.00 (d, 6H, J = 6.6 Hz), 3.80 (m, 1H), 3.91 (s, 2H), 3.97 (s, 3H), 7.52 (s,
1H). 7.96 (brd, 1H 1 J = 7.3 Hz), 8.18 (s, 1H), 8.50 (s, 1H), 13.3 (brs, 1H).
The binding affinity for HSP90 of the compounds of the invention can be measured using a Fluorescence Polarization Assay.
Fluorescence Polarization Assay
Fluorescence polarization {also known as fluorescence an i sot ropy} measures the rotation of a fluorescing species in solution, where the larger molecule the more
polarized the fluorescence emission. When the fluorophore is excited with polarized light, the emitted light is also polarized. The molecular size is proportional to the polarization of the fluorescence emission.
The fluoroscein-labelled probe VER-00051001-
binds to HSP90 { full-length human, full-length yeast or N-terminal domain HSP90 } and the anisotropy {rotation of the probe:protein complex} is measured.
Test compound is added to the assay plate, left to equilibrate and the anisotropy measured again. Any change in anisotropy is due to competitive binding of compound to HSP90, thereby releasing probe.
Materials
Chemicals are of the highest purity commercially available and all aqueous solutions are made up in AR water.
1 ) Costar 96-well black assay plate #3915
2) Assay buffer of (a)10OmM Tris pH7.4; (b) 2OmM KCI; (c) 6mM MgCI2 Stored at room temperature.
3) BSA (bovine serum albumen) 10 mg/ml (New England Biolabs # B9001 S)
4) 20 mM probe in 100 % DMSO stock concentration. Stored in the dark at RT. Working concentration is 200 nM diluted in AR water and stored at 4 0C. Final concentration in assay 8O nM.
5) E. coli expressed human full-length HSP90 protein, purified >95% (see, e.g., Panaretou et al., 1998) and stored in 50μL aliquots at -8O0C .
Protocol
1) Add 100μl 1x buffer to wells 11A and 12A (=FP BLNK)
2) Prepare assay mix - all reagents are kept on ice with a lid on the bucket as the probe is light-sensitive.
i. Final Cone"
• 1x Hsp90 FP Buffer 10 ml 1x
• BSA 10mg/ml (NEB) 5.0 μl 5 μg/ml
• Probe 200μM 4.0 μl 80 nM
• Human full-length Hsp90 6.25 μl 200 nM
3) Aliquot 100μl assay mix to all other wells
4) Seal plate and leave in dark at room temp for 20 minutes to equilibrate
Compound Dilution Plate - 1 x 3 dilution series
1 ) In a clear 96-well v-bottom plate - {# VWR 007/008/257} add 10 μl 100% DMSO to wells B1 to H11
2) To wells A1 to A11 add 17.5μl 100% DMSO
3) Add 2.5 μl cpd to A1. This gives 2.5 mM {50x} stock cpd - assuming cpds 20 mM.
4) Repeat for wells A2 to A10. Control in columns 11 and 12.
5) Transfer 5 μl from row A to row B- not column 12. Mix well.
6) Transfer 5 μl from row B to row C. Mix well.
7) Repeat to row G.
8) Do not add any compound to row H - this is the 0 row.
9) This produces a 1x3 dilution series from 50 μM to 0.07 μM.
10) In well B12 prepare 20 μl of 100 μM standard compound.
11 ) After first incubation the assay plate is read on a Fusion™ α-FP plate reader (Packard BioScience, Pangboume, Berkshire.UK).
12) After the first read, 2 μl of diluted compound is added to each well for columns 1 to 10. In column 11 {provides standard curve} only add compound B11 - H11. Add 2 μl of 10OmM standard cpd to wells B12 - H12 {is positive control }
13) The Z' factor is calculated from zero controls and positive wells. It typically gives a value of 0.7 - 0.9.
All compounds exemplified above had IC50 values of < 200 nM as determined by the Fluorescence Polarization Assay described above. By way of example, table 1 lists the affinity of specific compounds of the invention.
Table 1
Growth Inhibition Assay
A growth inhibition assay was also employed for the evaluation of candidate HSP90 inhibitors:
Assessment of cytotoxicity by Sulforhodamine B (SRB) assay: calculation of 50% inhibitory concentration (ICsn).
Day 1
1 ) Determine cell number by haemocytometer.
2) Using an 8 channel multipipettor, add 160μl of the cell suspension (3600 cells/well or 2 x 104 cells/ml) to each well of a 96-well microtitre plate.
3) Incubate overnight at 370C in a CO2 incubator.
Day 2
4) Stock solutions of drugs are prepared, and serial dilutions of each drug are performed in medium to give final concentrations in wells.
5) Using a multipipettor, 40μl of drug (at 5x final concentration) is added to quadruplicate wells.
6) Control wells are at either side of the 96 well plates, where 40μl of medium is added.
7) Incubate plates in CO2 incubator for 4 days (48 hours).
Day 6
8) Tip off medium into sink and immerse plate slowly into 10% ice cold trichloroacetic acid (TCA). Leave for about 30mins on ice.
9) Wash plates three times in tap water by immersing the plates into baths of tap water and tipping it off.
10) Dry in incubator.
11 ) Add 10Oμl of 0.4% SRB in 1 % acetic acid to each well (except the last row (right hand) of the 96 well plate, this is the 0% control, ie no drug, no stain. The first row will be the 100% control with no drug, but with stain). Leave for 15 mins.
12) Wash off unbound SRB stain with four washes of 1% acetic acid.
13) Dry plates in incubator.
14) Solubilise SRB using 100μl of 1OmM Tris base and put plates on plate shaker for 5 mins.
15) Determine absorbance at 540nm using a plate reader. Calculate mean absorbance for quadruplicate wells and express as a percentage of value for control, untreated wells.
16) Plot % absorbance values versus log drug concentration and determine the IC50
Compounds tested were assigned one of two values A or B. A IC50 < 0.50 μM. B IC50 > 0.50 μM
By way of illustration, the compound of example 1 gave an IC50 in the "A" range as described above when the effect on BT474 cell growth was tested as described above.
Claims
1. A compound of formula (I), or a salt thereof:
R is cyano or methoxy;
Ri and R2 are independently selected from hydrogen, optionally substituted C1-C3 alkyl, and optionally substituted C3-C6 cycloalkyl; or R1 and R2 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C1-C3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine;
R3 and R4 are independently selected from hydrogen, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, and cyclopropyl; or R2 and R3 taken together with the carbon to which they are attached form a 3- to 6-membered ring cycloalkyl ring;
n is 1 , 2 or 3; and
Z is
(i) -NR5R6 wherein R5 and R6 are independently selected from hydrogen, C1-C3 alkyl, and C3-C6 cycloalkyl; or R5 and R6 taken together with the nitrogen to which they are attached form a 3- to 7-membered ring optionally substituted by chloro, bromo, cyano, C1-C3 alkyl in which one or more hydrogens are optionally replaced by fluorine, or hydroxy(C1-C3 alkyl)- in which one or more hydrogens in the alkyl part are optionally replaced by fluorine; or (ii) C1-C3 alkoxy in which one or more hydrogens in the alkyl part are optionally replaced by fluorine.
2. A compound as claimed in claim 1 wherein n is 2.
3. A compound as claimed in claim 1 or claim 2 wherein Z is -NR5R6 wherein R5 and R6 are independently selected from hydrogen, methyl, trifluoromethyl, ethyl, and cyclopropyl; or R5 and R6 taken together with the nitrogen to which they are attached form a 5- or 6-membered ring optionally substituted by chloro, bromo, cyano, methyl, trifluoromethyl, hydroxymethyl or hydroxyethyl.
4. A compound as claimed in claim 1 or claim 2 wherein Z is methoxy, trifluoromethoxy, or ethoxy.
5. A compound as claimed in any of the preceding claims R3 and R4 are each hydrogen, or one of R3 and R4 is hydrogen and the other is methyl.
6. A compound as claimed in any of the preceding claims wherein R1 and R2 are independently selected from hydrogen, methyl, trifluoromethyl, ethyl, 2-trifluoroethyl, isopropyl, tert-butyl, cyclopropyl, cyclobutyl, hydroxymethyl and hydroxyethyl.
7. A compound as claimed in any of claims 1 to 5 wherein R1 and R2 taken together with the nitrogen to which they are attached form a, 3-, 4-, 5- or 6-membered ring optionally substituted by fluoro, chloro, bromo, cyano, methyl, trifluoromethyl, ethyl, hydroxymethyl, hydroxy, or hydroxyethyl.
8. A compound as claimed in claim 1 which is selected from the group consisting of 2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N,N-dimethyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N,N- dimethyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- cyclopropyl-acetamide; 2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- cyclopropyl-acetamide;
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- cyclopropyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- isopropyl-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- isopropyl-acetamide;
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- isopropyl-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-ethyl- acetamide;
2-[4-(2-Chloro-5-ethoxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- ethyl-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N- ethyl-acetamide;
N-tert-Butyl-2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanyl]-acetamide;
N-tert-Butyl-2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2- ylsulfanylj-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-
(2-fluoro-ethyl)-acetamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-(2- fluoro-ethyl)-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N-methyl-acetamide;
2-{4-[2-Chloro-5-(2-diethylamino-ethoxy)-4-methoxy-phenyl]-5-cyano-7H- pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl}-N,N-dimethyl-acetamide;
2-{4-[2-Chloro-4-methoxy-5-(2-methoxy-ethoxy)-phenyl]-5-cyano-7H-pyrro lo[2,3-d]pyrimidin-2-ylsulfanyl}-N-methyl-acetamide;
2-{4-[2-Chloro-5-(2-hydroxy-ethoxy)-4-methoxy phenyl]-5-cyano-7H-pyrro lo[2,3-d]pyrimidin-2-ylsulfanyl}-N-methyl-acetamide;
2-[4-(2-Chloro-5-hydroxy-4-methoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]py rimidin-2-ylsulfanyl]-N,N-dimethyl-propionamide;
2-[4-(2-Chloro-4,5-dimethoxy-phenyl)-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulfanyl]-N-(2,2,2- trifluofluoro-ethyl)-acetamide;
2-[4-[2-Chloro-4-cyano-5-methoxy-phenyl]-5-cyano-7H-pyrrolo[2,3-d]pyrimidin-2-ylsulphanyl]-N- isopropyl-acetamide;
and salts thereof.
9. A pharmaceutical or veterinary composition comprising a compound as claimed in any of claims 1 to 8, together with one or more pharmaceutically or veterinarily acceptable carriers and/or excipients.
10. The use of a compound as claimed in any of claims 1 to 9 in the preparation of a composition for inhibition of HSP90 activity in vitro or in vivo
11. A method of treatment of diseases which are responsive to inhibition of HSP90 activity in mammals, which method comprises administering to the mammal an amount of a compound as claimed in any of claims 1 to 9 effective to inhibit said HSP90 activity.
12. The use as claimed in claim 10 or a method as claimed claim 11 for immunosuppression or the treatment of viral disease, drug resistant fungal infection, parasitic infection, inflammatory diseases such as rheumatoid arthritis, asthma, multiple sclerosis, Type I diabetes, lupus, psoriasis and inflammatory bowel disease; cystic fibrosis angiogenesis-related disease such as diabetic retinopathy, haemangiomas, and endometriosis; or for protection of normal cells against chemotherapy-induced toxicity; or diseases where failure to undergo apoptosis is an underlying factor; or protection from hypoxia-ischemic injury due to elevation of Hsp70 in the heart and brain; scrapie/CJD, Huntingdon's or Alzheimer's disease.
13. The use as claimed in claim 10 or a method as claimed claim 11 , for the treatment of cancer.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/GB2007/003353 WO2009030871A1 (en) | 2007-09-07 | 2007-09-07 | Pyrrolopyrimidine derivatives having hsp90 inhibitory activity |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/GB2007/003353 WO2009030871A1 (en) | 2007-09-07 | 2007-09-07 | Pyrrolopyrimidine derivatives having hsp90 inhibitory activity |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009030871A1 true WO2009030871A1 (en) | 2009-03-12 |
Family
ID=39409806
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB2007/003353 Ceased WO2009030871A1 (en) | 2007-09-07 | 2007-09-07 | Pyrrolopyrimidine derivatives having hsp90 inhibitory activity |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2009030871A1 (en) |
Cited By (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011027081A2 (en) | 2009-09-03 | 2011-03-10 | Sanofi-Aventis | Novel derivatives of 5,6,7,8-tetrahydroindolizine inhibiting hsp90, compositions containing same, and use thereof |
| JP2015134797A (en) * | 2008-12-10 | 2015-07-27 | ウィスタ ラボラトリーズ リミテッド | 3,6-disubstituted xanthylium salt |
| US9266892B2 (en) | 2012-12-19 | 2016-02-23 | Incyte Holdings Corporation | Fused pyrazoles as FGFR inhibitors |
| US9388185B2 (en) | 2012-08-10 | 2016-07-12 | Incyte Holdings Corporation | Substituted pyrrolo[2,3-b]pyrazines as FGFR inhibitors |
| US9533984B2 (en) | 2013-04-19 | 2017-01-03 | Incyte Holdings Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US9533954B2 (en) | 2010-12-22 | 2017-01-03 | Incyte Corporation | Substituted imidazopyridazines and benzimidazoles as inhibitors of FGFR3 |
| US9580423B2 (en) | 2015-02-20 | 2017-02-28 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US9611267B2 (en) | 2012-06-13 | 2017-04-04 | Incyte Holdings Corporation | Substituted tricyclic compounds as FGFR inhibitors |
| US9708318B2 (en) | 2015-02-20 | 2017-07-18 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US9890156B2 (en) | 2015-02-20 | 2018-02-13 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US10611762B2 (en) | 2017-05-26 | 2020-04-07 | Incyte Corporation | Crystalline forms of a FGFR inhibitor and processes for preparing the same |
| US10851105B2 (en) | 2014-10-22 | 2020-12-01 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US11174257B2 (en) | 2018-05-04 | 2021-11-16 | Incyte Corporation | Salts of an FGFR inhibitor |
| CN114259564A (en) * | 2021-11-30 | 2022-04-01 | 清华大学 | Novel use of HSP90 inhibitors to block STAT3 mitochondrial transport and to treat asthma |
| US11407750B2 (en) | 2019-12-04 | 2022-08-09 | Incyte Corporation | Derivatives of an FGFR inhibitor |
| US11466004B2 (en) | 2018-05-04 | 2022-10-11 | Incyte Corporation | Solid forms of an FGFR inhibitor and processes for preparing the same |
| US11566028B2 (en) | 2019-10-16 | 2023-01-31 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US11591329B2 (en) | 2019-07-09 | 2023-02-28 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US11607416B2 (en) | 2019-10-14 | 2023-03-21 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US11628162B2 (en) | 2019-03-08 | 2023-04-18 | Incyte Corporation | Methods of treating cancer with an FGFR inhibitor |
| US11897891B2 (en) | 2019-12-04 | 2024-02-13 | Incyte Corporation | Tricyclic heterocycles as FGFR inhibitors |
| US11939331B2 (en) | 2021-06-09 | 2024-03-26 | Incyte Corporation | Tricyclic heterocycles as FGFR inhibitors |
| US12012409B2 (en) | 2020-01-15 | 2024-06-18 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US12065494B2 (en) | 2021-04-12 | 2024-08-20 | Incyte Corporation | Combination therapy comprising an FGFR inhibitor and a Nectin-4 targeting agent |
| US12122767B2 (en) | 2019-10-01 | 2024-10-22 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US12428420B2 (en) | 2021-06-09 | 2025-09-30 | Incyte Corporation | Tricyclic heterocycles as FGFR inhibitors |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6635762B1 (en) * | 1998-06-19 | 2003-10-21 | Pfizer Inc. | Monocyclic-7H-pyrrolo[2,3-d]pyrimidine compounds, compositions, and methods of use |
| WO2006105372A2 (en) * | 2005-03-30 | 2006-10-05 | Conforma Therapeutics Corporation | Alkynyl pyrrolopyrimidines and related analogs as hsp90-inhibitors |
| WO2007104944A1 (en) * | 2006-03-11 | 2007-09-20 | Vernalis (R & D) Ltd. | Pyrrolopyrimidine derivatives used as hsp90 inhibitors |
-
2007
- 2007-09-07 WO PCT/GB2007/003353 patent/WO2009030871A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6635762B1 (en) * | 1998-06-19 | 2003-10-21 | Pfizer Inc. | Monocyclic-7H-pyrrolo[2,3-d]pyrimidine compounds, compositions, and methods of use |
| WO2006105372A2 (en) * | 2005-03-30 | 2006-10-05 | Conforma Therapeutics Corporation | Alkynyl pyrrolopyrimidines and related analogs as hsp90-inhibitors |
| WO2007104944A1 (en) * | 2006-03-11 | 2007-09-20 | Vernalis (R & D) Ltd. | Pyrrolopyrimidine derivatives used as hsp90 inhibitors |
Cited By (54)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015134797A (en) * | 2008-12-10 | 2015-07-27 | ウィスタ ラボラトリーズ リミテッド | 3,6-disubstituted xanthylium salt |
| US10399955B2 (en) | 2008-12-10 | 2019-09-03 | Wista Laboratories Ltd. | 3,6-disubstituted xanthylium salts |
| US9549933B2 (en) | 2008-12-10 | 2017-01-24 | Wista Laboratories Ltd. | 3,6-disubstituted xanthylium salts |
| WO2011027081A2 (en) | 2009-09-03 | 2011-03-10 | Sanofi-Aventis | Novel derivatives of 5,6,7,8-tetrahydroindolizine inhibiting hsp90, compositions containing same, and use thereof |
| US10213427B2 (en) | 2010-12-22 | 2019-02-26 | Incyte Corporation | Substituted imidazopyridazines and benzimidazoles as inhibitors of FGFR3 |
| US10813930B2 (en) | 2010-12-22 | 2020-10-27 | Incyte Corporation | Substituted imidazopyridazines and benzimidazoles as inhibitors of FGFR3 |
| US9533954B2 (en) | 2010-12-22 | 2017-01-03 | Incyte Corporation | Substituted imidazopyridazines and benzimidazoles as inhibitors of FGFR3 |
| US10131667B2 (en) | 2012-06-13 | 2018-11-20 | Incyte Corporation | Substituted tricyclic compounds as FGFR inhibitors |
| US11840534B2 (en) | 2012-06-13 | 2023-12-12 | Incyte Corporation | Substituted tricyclic compounds as FGFR inhibitors |
| US12534463B2 (en) | 2012-06-13 | 2026-01-27 | Incyte Corporation | Substituted tricyclic compounds as FGFR inhibitors |
| US11053246B2 (en) | 2012-06-13 | 2021-07-06 | Incyte Corporation | Substituted tricyclic compounds as FGFR inhibitors |
| US9611267B2 (en) | 2012-06-13 | 2017-04-04 | Incyte Holdings Corporation | Substituted tricyclic compounds as FGFR inhibitors |
| US9388185B2 (en) | 2012-08-10 | 2016-07-12 | Incyte Holdings Corporation | Substituted pyrrolo[2,3-b]pyrazines as FGFR inhibitors |
| US9745311B2 (en) | 2012-08-10 | 2017-08-29 | Incyte Corporation | Substituted pyrrolo[2,3-b]pyrazines as FGFR inhibitors |
| US9266892B2 (en) | 2012-12-19 | 2016-02-23 | Incyte Holdings Corporation | Fused pyrazoles as FGFR inhibitors |
| US10040790B2 (en) | 2013-04-19 | 2018-08-07 | Incyte Holdings Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US9533984B2 (en) | 2013-04-19 | 2017-01-03 | Incyte Holdings Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US10947230B2 (en) | 2013-04-19 | 2021-03-16 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US11530214B2 (en) | 2013-04-19 | 2022-12-20 | Incyte Holdings Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US10450313B2 (en) | 2013-04-19 | 2019-10-22 | Incyte Holdings Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US10851105B2 (en) | 2014-10-22 | 2020-12-01 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US9890156B2 (en) | 2015-02-20 | 2018-02-13 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US10016438B2 (en) | 2015-02-20 | 2018-07-10 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US10738048B2 (en) | 2015-02-20 | 2020-08-11 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US11667635B2 (en) | 2015-02-20 | 2023-06-06 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US10251892B2 (en) | 2015-02-20 | 2019-04-09 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US10214528B2 (en) | 2015-02-20 | 2019-02-26 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US11014923B2 (en) | 2015-02-20 | 2021-05-25 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US9801889B2 (en) | 2015-02-20 | 2017-10-31 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US11173162B2 (en) | 2015-02-20 | 2021-11-16 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US9580423B2 (en) | 2015-02-20 | 2017-02-28 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US10632126B2 (en) | 2015-02-20 | 2020-04-28 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US9708318B2 (en) | 2015-02-20 | 2017-07-18 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| US10611762B2 (en) | 2017-05-26 | 2020-04-07 | Incyte Corporation | Crystalline forms of a FGFR inhibitor and processes for preparing the same |
| US11472801B2 (en) | 2017-05-26 | 2022-10-18 | Incyte Corporation | Crystalline forms of a FGFR inhibitor and processes for preparing the same |
| US12024517B2 (en) | 2018-05-04 | 2024-07-02 | Incyte Corporation | Salts of an FGFR inhibitor |
| US11174257B2 (en) | 2018-05-04 | 2021-11-16 | Incyte Corporation | Salts of an FGFR inhibitor |
| US12473286B2 (en) | 2018-05-04 | 2025-11-18 | Incyte Corporation | Salts of an FGFR inhibitor |
| US11466004B2 (en) | 2018-05-04 | 2022-10-11 | Incyte Corporation | Solid forms of an FGFR inhibitor and processes for preparing the same |
| US11628162B2 (en) | 2019-03-08 | 2023-04-18 | Incyte Corporation | Methods of treating cancer with an FGFR inhibitor |
| US11591329B2 (en) | 2019-07-09 | 2023-02-28 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US12122767B2 (en) | 2019-10-01 | 2024-10-22 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US12083124B2 (en) | 2019-10-14 | 2024-09-10 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US11607416B2 (en) | 2019-10-14 | 2023-03-21 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US11566028B2 (en) | 2019-10-16 | 2023-01-31 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US11897891B2 (en) | 2019-12-04 | 2024-02-13 | Incyte Corporation | Tricyclic heterocycles as FGFR inhibitors |
| US11407750B2 (en) | 2019-12-04 | 2022-08-09 | Incyte Corporation | Derivatives of an FGFR inhibitor |
| US12168660B2 (en) | 2019-12-04 | 2024-12-17 | Incyte Corporation | Derivatives of an FGFR inhibitor |
| US12012409B2 (en) | 2020-01-15 | 2024-06-18 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| US12065494B2 (en) | 2021-04-12 | 2024-08-20 | Incyte Corporation | Combination therapy comprising an FGFR inhibitor and a Nectin-4 targeting agent |
| US11939331B2 (en) | 2021-06-09 | 2024-03-26 | Incyte Corporation | Tricyclic heterocycles as FGFR inhibitors |
| US12428420B2 (en) | 2021-06-09 | 2025-09-30 | Incyte Corporation | Tricyclic heterocycles as FGFR inhibitors |
| CN114259564A (en) * | 2021-11-30 | 2022-04-01 | 清华大学 | Novel use of HSP90 inhibitors to block STAT3 mitochondrial transport and to treat asthma |
| CN114259564B (en) * | 2021-11-30 | 2023-03-14 | 清华大学 | Novel use of HSP90 inhibitors for blocking STAT3 mitochondrial transport and for treating asthma |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2009030871A1 (en) | Pyrrolopyrimidine derivatives having hsp90 inhibitory activity | |
| CA2537135C (en) | Pyrimidothiophene compounds | |
| AU2014400628B2 (en) | Aminopyridazinone compounds as protein kinase inhibitors | |
| CN104470919B (en) | 2-(azaindole-2-base) benzimidazole as PAD4 inhibitor | |
| EP2651417B1 (en) | Ask1 inhibiting pyrrolopyrimidine derivatives | |
| EP2920162B1 (en) | Inhibitors of bruton's tyrosine kinase | |
| AU2017315343A1 (en) | Amino-pyrrolopyrimidinone compounds and methods of use thereof | |
| CA3088025A1 (en) | Inhibitors of the bcl6 btb domain protein-protein interaction and uses thereof | |
| EP2007767B1 (en) | Pyrrolopyrimidine derivatives used as hsp90 inhibitors | |
| AU2002366801B8 (en) | Pyrrolopyrimidine A2b selective antagonist compounds, their synthesis and use | |
| CA3042301A1 (en) | Degradation of protein kinases by conjugation of protein kinase inhibitors with e3 ligase ligand and methods of use | |
| TWI826509B (en) | Heteroaromatic compounds as vanin inhibitors | |
| JPH08507056A (en) | Pyrazolitriazine having interleukin-1 and tumor necrosis factor inhibitory activity | |
| CA2723237A1 (en) | Substituted amides, method of making, and use as btk inhibitors | |
| CN102781943A (en) | Benzodiazepine bromodomain inhibitor | |
| CN104768946A (en) | Fused pyrimidines as inhibitors of the p97 complex | |
| ZA200601715B (en) | Pyrimidothiophene compounds | |
| EP2079742A1 (en) | Pteridine derivatives as polo-like kinase inhibitors useful in the treatment of cancer | |
| WO2020114943A1 (en) | Heteroaromatic compounds as vanin inhibitors | |
| EP3080099B1 (en) | Inhibitors of bruton's tyrosine kinase | |
| CN107793371B (en) | A class of bromodomain recognition protein inhibitors and preparation method and use thereof | |
| US20220041592A1 (en) | Heteroaromatic compounds as vanin inhibitors | |
| WO2007034185A1 (en) | Purine compounds as hsp90 protein inhibitors for the treatment of cancer | |
| WO2008025947A1 (en) | 1h-pyrrolo[2,3-b]pyridine derivatives useful as hsp90 inhibitors | |
| EP2313413B1 (en) | Pyrazolo [1, 5-a] pyrimidine derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07823889 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 07823889 Country of ref document: EP Kind code of ref document: A1 |