WO2009026544A1 - Ampicillin and amoxicillin-based poly(anhydride-amides) - Google Patents
Ampicillin and amoxicillin-based poly(anhydride-amides) Download PDFInfo
- Publication number
- WO2009026544A1 WO2009026544A1 PCT/US2008/074070 US2008074070W WO2009026544A1 WO 2009026544 A1 WO2009026544 A1 WO 2009026544A1 US 2008074070 W US2008074070 W US 2008074070W WO 2009026544 A1 WO2009026544 A1 WO 2009026544A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- polymer
- formula
- ampicillin
- pharmaceutically acceptable
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D499/00—Heterocyclic compounds containing 4-thia-1-azabicyclo [3.2.0] heptane ring systems, i.e. compounds containing a ring system of the formula:, e.g. penicillins, penems; Such ring systems being further condensed, e.g. 2,3-condensed with an oxygen-, nitrogen- or sulfur-containing hetero ring
- C07D499/21—Heterocyclic compounds containing 4-thia-1-azabicyclo [3.2.0] heptane ring systems, i.e. compounds containing a ring system of the formula:, e.g. penicillins, penems; Such ring systems being further condensed, e.g. 2,3-condensed with an oxygen-, nitrogen- or sulfur-containing hetero ring with a nitrogen atom directly attached in position 6 and a carbon atom having three bonds to hetero atoms with at the most one bond to halogen, e.g. an ester or nitrile radical, directly attached in position 2
- C07D499/44—Compounds with an amino radical acylated by carboxylic acids, attached in position 6
- C07D499/48—Compounds with an amino radical acylated by carboxylic acids, attached in position 6 with a carbon chain, substituted by hetero atoms or by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, attached to the carboxamido radical
- C07D499/58—Compounds with an amino radical acylated by carboxylic acids, attached in position 6 with a carbon chain, substituted by hetero atoms or by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, attached to the carboxamido radical substituted in alpha-position to the carboxamido radical
- C07D499/64—Compounds with an amino radical acylated by carboxylic acids, attached in position 6 with a carbon chain, substituted by hetero atoms or by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, attached to the carboxamido radical substituted in alpha-position to the carboxamido radical by nitrogen atoms
- C07D499/68—Compounds with an amino radical acylated by carboxylic acids, attached in position 6 with a carbon chain, substituted by hetero atoms or by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, attached to the carboxamido radical substituted in alpha-position to the carboxamido radical by nitrogen atoms with aromatic rings as additional substituents on the carbon chain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- Bacterial infections are a safety issue with medical implants, including stainless steel materials; such infections are extremely difficult to treat due to the development of resistant biof ⁇ lm networks that are not susceptible to traditional antibiotics (Patel R. Biofilms and antimicrobial resistance. Clin Orthop Relat Res, 2005, 437, 41-47). Such infections often lead to multiple revision surgeries, and in some cases, death (Campoccia D, et al., Biomaterials, 2006, 27, 2331- 2339).
- bacterial infections are systemically treated with a course of antibiotics.
- systemic treatments expose the entire body to the drug such that the implant site may not receive the dosage necessary to kill the bacteria (Burrows LL, Khoury AE. Infection of medical devices. In: Wnek GE, Bowlin GL., editor. Encyclopedia of Biomaterials and Biomedical Engineering. New York: Marcel Dekkar; 2004.839-848).
- this lowered dose results in the development of resistant bacteria, and ultimately biof ⁇ lm formation (Hanssen AD, et al., Clin Orthop Relat Res, 2005, 437, 111-114).
- Localized antibiotic therapy can also be used to treat bacterial infections associated with medical implants.
- the most commonly studied localized antibiotic therapy is the physical admixture of antimicrobials within polymer matrices to treat bacterial infections (see von Eiff C, et al., Int J Artif Organs, 2005, 28, 1146-56; Garvin K, Feschuk, Clin Orthop Relat Res, 2005, 437, 105- 110; Gollwitzer H, et al., J Antimicrob Chemother, 2003, 51, 585-591 and Sharkawi T, et al., JBioact and Compat Pol, 2005, 20, 153-168).
- the present invention provides compounds that have antibacterial properties. Accordingly there is provided a compound of the invention which is a compound of formula I:
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12; or a salt thereof.
- n 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12; and each R is independently H or OH; or a salt thereof.
- each R is OH. In certain embodiments, each R is H.
- one R is H and one R is OH.
- n 4, 5, 6, 7 or 8.
- n 6, 7 or 8.
- n 8
- the invention also provides a pharmaceutical composition comprising a compound of the invention and a pharmaceutically acceptable diluent or carrier.
- the invention also provides a medical device having a coating that comprises a compound of the invention. In one embodiment the invention also provides a medical device comprising a compound of the invention.
- the medical device is an orthopedic implant. hi certain embodiments, the medical device is a knee joint or a hip joint.
- the invention also provides a method to promote an anti-bacterial effect in an animal comprising administering a compound of the invention to the animal.
- the antibacterial effect is against gram negative and/or gram positive bacteria.
- the invention also provides a method to promote an antibacterial effect comprising contacting bacteria (e.g., in vivo or in vitro) with a compound of the invention.
- the invention also provides a method to reduce biofilm formation on a surface comprising contacting the surface with a compound of the invention.
- the compound of the invention is incorporated into a coating that is on the surface.
- the invention also provides a compound of the invention for use in medical therapy.
- the invention also provides the use of a compound of the invention for the manufacture of a medicament for producing an anti- bacterial effect in a mammal.
- the invention also provides a compound of the invention, for the prophylactic or therapeutic treatment of a bacterial infection.
- the invention also provides processes and intermediates disclosed herein that are useful for preparing compounds of formula (I) or (II) or salts thereof.
- the compounds, compositions and methods of the invention are useful for preventing biofilm formation and for treating antibiotic resistant bacterial infections, such as those infections associated with medical implants.
- polymeric materials for biological applications such as medical devices (e.g., artificial organs, wound dressings, sutures, pacemakers, intravascular catheters, contact lenses, orthopedic implants) and drug delivery systems has evoked increasing interest.
- medical devices e.g., artificial organs, wound dressings, sutures, pacemakers, intravascular catheters, contact lenses, orthopedic implants
- drug delivery systems has evoked increasing interest.
- Numerous polymeric biomaterials are implanted each year in humans, yet bacterial infections associated with these medical implants remain a significant issue. For example, nearly half of the two million nosocomial infections occurring in the United States are associated with medical implants.
- the presence of a foreign body, such as a prosthetic implant provides a site for microbial adhesion and subsequent colonization, often leading to device-related infections.
- Current clinical therapies to prevent biomaterial- related infections involve the lengthy administration of antibiotics at high systemic concentrations. Unfortunately, repeated failures of this therapeutic approach require device removal to ensure eradication of the infection and to avoid relapse.
- degradable biomaterials were designed to release antimicrobials upon polymer degradation to locally prevent infections.
- This work describes the synthesis of biodegradable antimicrobial-based polymers in which the antimicrobial drugs ampicillin and amoxicillin are chemically incorporated into the polymeric backbone by anhydride and amide bonds, rather than attached as a side group or physically admixed.
- Antibacterial properties of the polymers were examined using gram-negative (Escherichia col ⁇ ) and gram- positive (Staphylococcus aureus) bacterial strains.
- E. coli is the major cause of infections in biliary stents and urinary catheters, and S. aureus is responsible for infections associated with dental and orthopedic implants and fixation devices.
- Antibiotic molecules (up to nearly 82% by weight) were incorporated into a polymeric backbone.
- the polymers were prepared under mild conditions using solution polymerization methods. This milder polymerization method was chosen over typical melt-condensation processes because of the thermal instability of the antibiotics and polymer precursors. No glass transition temperatures (T g ) were observed for the polymers in the measured range from - 10 °C up to polymer decomposition ( ⁇ 200 °C).
- T g glass transition temperatures
- the antimicrobial-based polymers displayed enhanced solubility in organic solvents relative to the antibiotics alone. This factor is significant, as it determines processability of polymer into solvent-cast films, microspheres, and fibers that may be used as controlled release devices.
- biodegradable antimicrobial-based poly(anhydride-amides) display promise as implants or coatings to reduce implant failure resulting from bacterial infections.
- a salt of a compound of formula I or II can be useful as an intermediate for isolating or purifying a compound of formula I or ⁇ .
- a compound of formula I or II as a pharmaceutically acceptable acid or base salt
- pharmaceutically acceptable salts are organic acid addition salts formed with acids which form a physiological acceptable anion, for example, tosylate, methanesulfonate, acetate, citrate, malonate, tartrate, succinate, benzoate, ascorbate, ⁇ -ketoglutarate, and ⁇ -glycerophosphate.
- Suitable inorganic salts may also be formed, including hydrochloride, sulfate, nitrate, bicarbonate, and carbonate salts.
- salts may be obtained using standard procedures well known in the art, for example by reacting a sufficiently basic compound such as an amine with a suitable acid affording a physiologically acceptable anion.
- a sufficiently basic compound such as an amine
- a suitable acid affording a physiologically acceptable anion.
- Alkali metal (for example, sodium, potassium or lithium) or alkaline earth metal (for example calcium) salts of carboxylic acids can also be made.
- the compounds of the invention are useful in a variety of applications where antibacterial activity is desired. Examples of such applications include, but are not limited to, medical, dental and cosmetic uses.
- the compounds of the invention may be prepared in accordance with methods commonly employed in the field of synthetic chemistry. The compounds can be readily processed into pastes or solvent cast to yield films, coatings, microspheres and fibers with different geometric shapes for design of various medical implants.
- the compounds of the invention can be incorporated into and/or released from medical implants such as, for example, vascular grafts and stents, bone plates, sutures, Implantable sensors, implantable drug delivery devices, orthopedic devices (e.g. replacement joints such as knees and hips), and stents for tissue regeneration.
- the diacid compounds of formula (I) or (II) can be released by hydrolysis of a corresponding anhydride polymer.
- the compounds of formula (I) or (II) can also be incorporated (e.g. dispersed) into the matrix of such an anhydride polymer or another polymer, and be released from that polymer over time (e.g. upon hydrolysis of the polymer).
- Compounds of the invention can also be incorporated into oral formulations and into products such as skin moisturizers, cleansers, pads, plasters, lotions, creams, gels, ointments, solutions, shampoos, tanning products and lipsticks for topical application.
- Useful dosages of the compounds can be determined by comparing their in vitro activity, and in vivo activity of the compounds of the invention in animal models. Additionally, useful dosages can be determined by measuring the rate of hydrolysis for a given polymer under various physiological conditions.
- the amount of a polymer required for use in treatment will vary not only with the particular polymer selected but also with the route of administration, the nature of the condition being treated and the age and condition of the patient and will be ultimately at the discretion of the attendant physician or clinician.
- the desired dose may conveniently be presented in a single dose or as divided doses administered at appropriate intervals, for example, as two, three, four or more sub-doses per day.
- the sub-dose itself may be further divided, e.g., into a number of discrete loosely spaced administrations.
- the compounds of formula I or II can be formulated as pharmaceutical compositions and administered to a mammalian host, such as a human patient in a variety of forms adapted to the chosen route of administration, e.g., orally or parenterally, by intravenous, intramuscular, topical or subcutaneous routes.
- the present compounds may be systemically administered, e.g., orally, in combination with a pharmaceutically acceptable vehicle such as an inert diluent or an assimilable edible carrier. They may be enclosed in hard or soft shell gelatin capsules, may be compressed into tablets, or may be incorporated directly with the food of the patient's diet.
- a pharmaceutically acceptable vehicle such as an inert diluent or an assimilable edible carrier.
- the active compound may be combined with one or more excipients and used in the form of ingestible tablets, buccal tablets, troches, capsules, elixirs, suspensions, syrups, wafers, and the like.
- the tablets, troches, pills, capsules, and the like may also contain the following: binders such as gum tragacanth, acacia, corn starch or gelatin; excipients such as dicalcium phosphate; a disintegrating agent such as corn starch, potato starch, alginic acid and the like; a lubricant such as magnesium stearate; and a sweetening agent such as sucrose, fructose, lactose or aspartame or a flavoring agent such as peppermint, oil of wintergreen, or cherry flavoring may be added.
- a liquid carrier such as a vegetable oil or a polyethylene glycol.
- any material used in preparing any unit dosage form should be pharmaceutically acceptable and substantially non-toxic in the amounts employed, hi addition, the active compound may be incorporated into sustained-release preparations and devices.
- the active compound may also be administered intravenously or intraperitoneally by infusion or injection.
- Solutions of the active compound or its salts can be prepared in water, optionally mixed with a nontoxic surfactant.
- Dispersions can also be prepared in glycerol, liquid polyethylene glycols, triacetin, and mixtures thereof and in oils. Under ordinary conditions of storage and use, these preparations contain a preservative to prevent the growth of microorganisms.
- the pharmaceutical dosage forms suitable for injection or infusion can include sterile aqueous solutions or dispersions or sterile powders comprising the active ingredient which are adapted for the extemporaneous preparation of sterile injectable or infusible solutions or dispersions, optionally encapsulated in liposomes.
- the ultimate dosage form should be sterile, fluid and stable under the conditions of manufacture and storage.
- the liquid carrier or vehicle can be a solvent or liquid dispersion medium comprising, for example, water, ethanol, a polyol (for example, glycerol, propylene glycol, liquid polyethylene glycols, and the like), vegetable oils, nontoxic glyceryl esters, and suitable mixtures thereof.
- the proper fluidity can be maintained, for example, by the formation of liposomes, by the maintenance of the required particle size in the case of dispersions or by the use of surfactants.
- the prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal, and the like. In many cases, it will be preferable to include isotonic agents, for example, sugars, buffers or sodium chloride. Prolonged absorption of the injectable compositions can be brought about by the use in the compositions of agents delaying absorption, for example, aluminum monostearate and gelatin.
- Sterile injectable solutions are prepared by incorporating the active compound in the required amount in the appropriate solvent with various of the other ingredients enumerated above, as required, followed by filter sterilization, hi the case of sterile powders for the preparation of sterile injectable solutions, the preferred methods of preparation are vacuum drying and the freeze drying techniques, which yield a powder of the active ingredient plus any additional desired ingredient present in the previously sterile-filtered solutions.
- the present compounds may be applied in pure form, i.e., when they are liquids. However, it will generally be desirable to administer them to the skin as compositions or formulations, in combination with a dermatologically acceptable carrier, which may be a solid or a liquid.
- a dermatologically acceptable carrier which may be a solid or a liquid.
- Useful solid carriers include finely divided solids such as talc, clay, microcrystalline cellulose, silica, alumina and the like.
- Useful liquid carriers include water, alcohols or glycols or water-alcohol/glycol blends, in which the present compounds can be dissolved or dispersed at effective levels, optionally with the aid of non-toxic surfactants.
- Adjuvants such as fragrances and additional antimicrobial agents can be added to optimize the properties for a given use.
- the resultant liquid compositions can be applied from absorbent pads, used to impregnate bandages and other dressings, or sprayed onto the affected area using pump-type or aerosol sprayers.
- Thickeners such as synthetic polymers, fatty acids, fatty acid salts and esters, fatty alcohols, modified celluloses or modified mineral materials can also be employed with liquid carriers to form spreadable pastes, gels, ointments, soaps, and the like, for application directly to the skin of the user.
- useful dermatological compositions which can be used to deliver the compounds of formula I or ⁇ to the skin are known to the art; for example, see Jacquet et al. (U.S. Pat. No. 4,608,392), Geria (U.S. Pat. No. 4,992,478), Smith et al. (U.S. Pat. No. 4,559,157) and Wortzman (U.S. Pat. No. 4,820,508).
- DMF Dimethylformamide
- acetic anhydride dichloromethane
- diethyl ether diethyl ether
- pyridine pyridine
- triethylamine purchased from Aldrich (Milwaukee, WI). All other reagents and solvents were purchased from Fisher Scientific (Fair Lawn, NJ) and used as received. Triethylamine was dried over calcium hydride, all other reagents were used without further purification.
- the ampicillin-based diacid (2) was prepared in DMF with pyridine as a base at 0°C.
- the polymer (1) was synthesized using triphosgene as a coupling agent and triethylamine as an acid acceptor at 0 °C under nitrogen.
- a 5 % (w/v) ampicillin-based poly(anhydride-amide) (1) solution was prepared in DMF for solvent-casting.
- the polymer solution 150 ⁇ L was pipetted onto 316L stainless steel coupons (McMaster-Carr, cut to 10 x 30 mm by Rutgers Physics Department Machine Shop).
- the solvent was evaporated using one of three methods: i) ambient conditions overnight; ii) heating samples to 60 °C for two hours; and iii) heating under vacuum to 60 °C for 45 minutes. Additionally, the ampicillin-based diacid precursor was precipitated onto stainless steel substrates for comparison to the polymer coatings. Peel tests were conducted using the x-cut method in American Standard
- Two 10 mm X-cuts were scratched into coated coupons using a fresh razor blade.
- Permacel P-99 tape was adhered to the surface and peeled away at a 180° angle after 90 seconds.
- Each sample was examined for coating removal and ranked according to the following scale: 5A- no peeling or removal; 4A-trace peeling or removal; 3A-jagged removal along incisions; 2A-jagged removal along most of the incisions; 1 A-removal from most of the area of the X under the tape; and 0 A-removal beyond the area of the X.
- Sessile contact angle measurements were completed using deionized water on a model 250 Rame-Hart goniometer (Mountain Lakes, NJ) with DROPimage software. One water drop was placed on each coating and measured in real time with a camera. The contact angle was measured as the average value from five water droplets with three angle measurements taken for each drop. The final contact angle value is the average of at least 15 measurements.
- Staphylococcus aureus was obtained from ATCC (Manassas, VA). The coatings were exposed to the Staphylococcus aureus bacteria cells at the required density for biofilm formation (10 6 colony forming units (CFU)/mL) for 24 hours at 37 °C. Adherent bacteria cells were recovered and quantified compared to negative uncoated controls. In Vitro Degradation with Bacteria Present
- the HPLC method was developed to separate the free ampicillin released from the ampicillin diacid at 210 nm on a Gemini Cl 8 column (15O x 4.6 mm, 5 ⁇ m, Phenomenex, Torrance, CA).
- the method was a linear gradient with a mobile phase consisting of 20 mM monobasic potassium phosphate pH 4.0 and methanol.
- Five point calibration curves were generated for both the ampicillin and the ampicillin diacid with concentrations ranging from 0.005 mg/mL to 0.5 mg/mL.
- the degradation media was diluted in monobasic potassium phosphate, if needed, to calculate release within the calibration curve.
- Cytotoxicity of the polymer coatings was analyzed by culturing cells in media containing degraded polymer. Individual ampicillin-based polymer- coated stainless steel plates were placed in 50 mL of cell culture media for 48 hours. Media consisted of Dulbecco's Modified Eagle's Medium (DMEM; Sigma, St. Louis, MO), 10% v/v fetal bovine serum (Atlanta Biologicals, Lawrenceville, GA), penicillin/streptomycin, and L-glutamine.
- DMEM Dulbecco's Modified Eagle's Medium
- L929 mouse areolar/adipose fibroblasts (Department of Biomedical Engineering, Rutgers University, Piscataway, New Jersey) were sustained in a cell culture incubator at 37°C and an atmosphere containing 5% CO 2. Confluent fibroblasts were removed from the cell culture flask via trypsinization. After media removal, trypsin (2 mg/mL, Sigma, St. Louis, MO) was placed on cells and incubated at 37°C for five minutes to allow cell detachment from the surface of the flask. Trypsin activity was ceased with addition of media and the solution centrifuged for 2 min at 2000 rpm (Thermo Electron 5682 3L GP, Franklin, MA).
- Cells were seeded at a concentration of 100,000 cells/well in media containing polymer degradation media and analyzed after 24, 48, and 72 hours. All samples were studied in triplicate. A standard curve was generated with cells seeded at 25,000, 50,000, 100,000, and 500,000 cells/well in cell culture media without polymer, analyzed after 24 hours. Imaging was performed with a fluorescent microscope (Olympus 1X81,
- Fluorescent intensity was quantified using a fluorescence plate reader (Cytofluor ® Series 4000, Applied Biosystems, Woodinville, CA) at 485 nm excitation and 530 nm emission. For the three days, live cell numbers were quantified against the standard curve using Microsoft Excel ®. Results and Discussion
- the quality of the polymer coatings was further assessed by an ASTM peel test (ASTM D 3359. West Conshohocken, PA: ASTM International). The results from the qualitative peel test are provided in the following table.
- Peel results showed trace coating removal along x-cut incisions, but overall, the coatings strongly adhere to the stainless steel substrates. These results may be due to a nonspecific chemical interaction between the steel and the coating, ultimately causing the coating to strongly adhere to the stainless steel surface without modification.
- a jagged removal along the x-cut incisions was noted.
- the vacuum and heat processed samples were typically more robust.
- the contact angle of the polymer coatings was about 45°, regardless of the solvent evaporation method. These coatings have intermediate contact angles, which are amenable to degradation, yet not immediately solubilized in aqueous media.
- the decomposition temperatures of the polymer coatings prepared by various processing methods were measured by TGA to ensure polymer stability during processing.
- Polymer (1) displayed a slight increase in the decomposition temperature following the coating preparation, increasing to a Tj of about 210
- the polymer does not decompose.
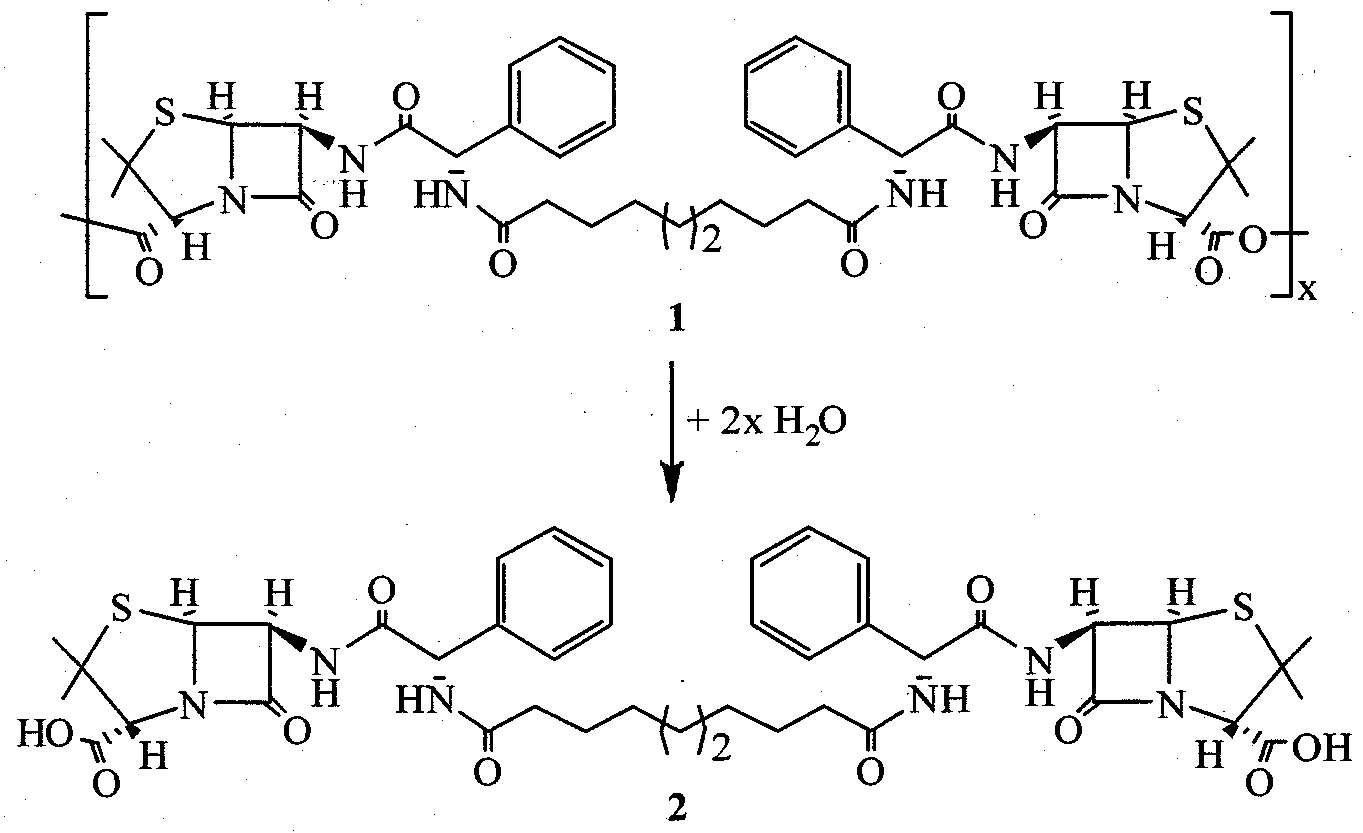
- the hydrolytic degradation scheme for the polymer (1) is shown below.
- the anhydride bonds are most labile, whereas the amide bonds in the diacid (2) intermediate are expected to hydrolyze more slowly and possibly require enzymatic degradation.
- Polymer 1 was found to hydrolyze in a more controlled manner with more than 70 % of the ampicillin-based polymer (1) coating degraded in 60 hours. Additional release data was obtained from a Varian USP 7 Dissolution Instrument with the ability to separate degradation products by HPLC. Ln these studies, the free ampicillin drug was not detected because the degradation media was PBS at pH 7.4. The conditions were not amenable to amide bond hydrolysis and there was no ampicillin peak observed in the HPLC chromatogram (results not shown). Again, the diacid intermediate (2) was the final degradation product released from the coatings.
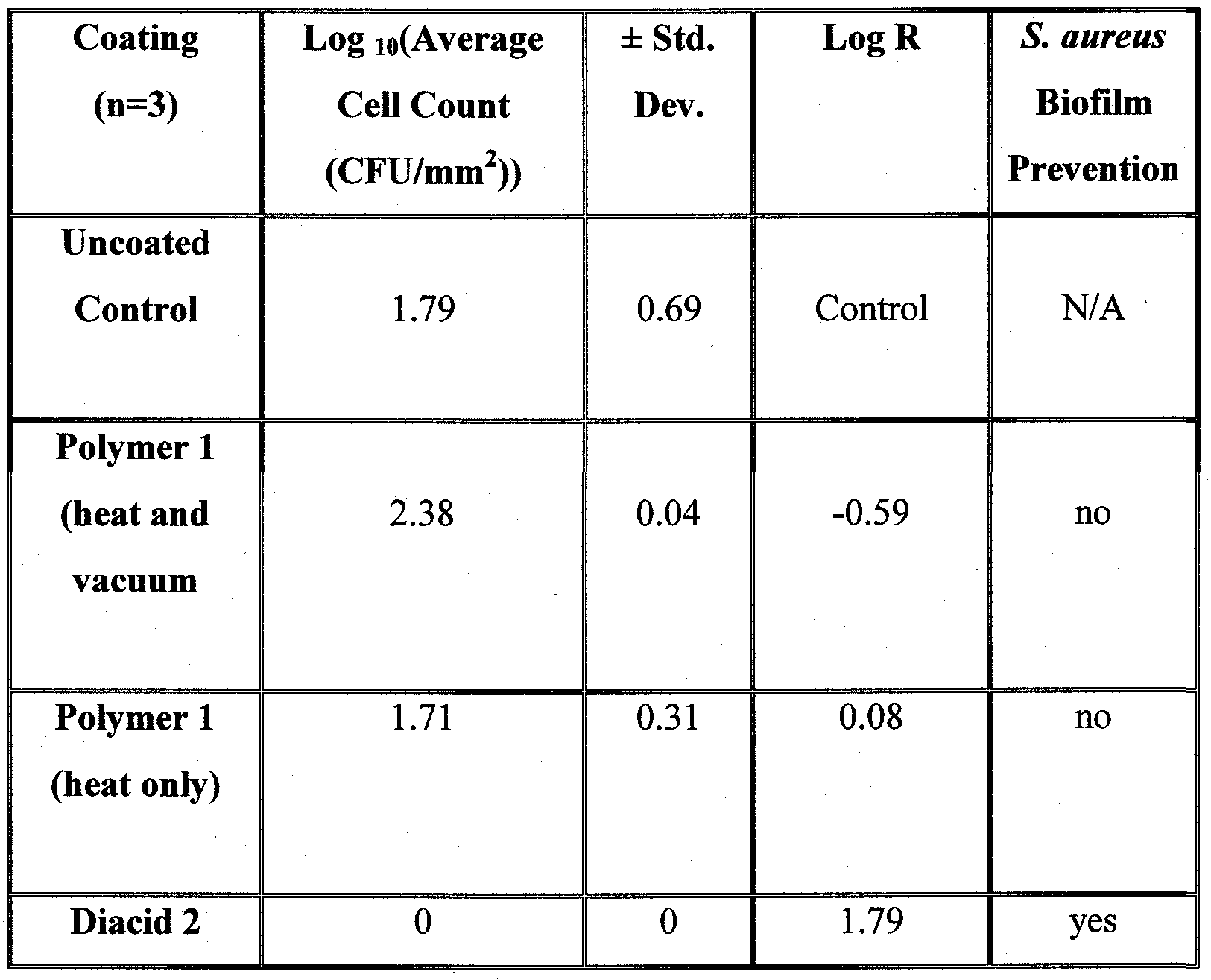
- Staphylococcus aureus growth was monitored during exposure to diacid (2) and polymer (1) coatings, as well as uncoated stainless steel controls.
- the bacteria cells were seeded to facilitate biofilm growth and were exposed to the coatings for 24 hours. The results of the study are shown in the following table.
- the ampicillin-based diacid (2) demonstrated complete prevention of Staphylococcus aureus biofilm.
- polymer 1 itself did not inhibit biofilm formation, indicating that the diacid 2 is a bioactive compounds.
- No significant difference in bacterial growth was observed between the uncoated control and the polymer samples. Given that Staphylococcus aureus does not grow on the diacid (2) coating, over longer time periods, it was determined that the diacid will prevent bacteria cell growth and biofilm formation.
- Fibroblasts were tested in media containing degraded polymer coatings through heat or through a heat and vacuum methods. Cell proliferation was quantified through 72 hours of culture. Fibroblast culture growth was not impeded by the use of polyampicillin in the cell culture medium. All samples showed positive growth throughout the duration of testing with neither polymer impeding cell proliferation and amplification.
- Cell morphology was compared to cells cultured in the control, which contained media without any polymer present.
- the cells displayed their natural, heterogeneous morphology. Generally, the fibroblasts readily attached and remained attached to the surface despite washing and showed characteristic spreading and extensions.
- cells cultured in media containing polymer 1 coatings prepared with fast solvent evaporation (heat and vacuum) did not extend and spread fibers as frequently as the control and polymer 1 coatings prepared with slow solvent evaporation (heat). Nonetheless, cells showed stellate morphology through 72 hours, proving a low cytotoxicity to the polymer degradation products.
- Ampicillin-based poly(anhydride-amide) coatings may improve current clinical therapies for bacterial infections.
- the coatings have strong adhesion properties to 316L stainless steel, which have not been demonstrated in any other polymer system.
- the in vitro degradation studies yielded the ampicillin-based diacid as the final degradation product, in PBS pH 7.4.
- the polymer (1) itself was found to be inactive against bacteria growth, it ws found to degrade to the more hydrophilic diacid (2), which completely eradicated Staphylococcus aureus growth and biofilm formation.
- Example 2 Poly(anhydride-amides) that include thermally sensitive ⁇ -lactam antibiotics were prepared by solution polymerization.
- Biodegradable antimicrobial-based polymers in which antimicrobial drugs, namely ampicillin and amoxicillin, were chemically incorporated into the polymeric backbone by anhydride and amide bonds were designed to locally prevent infections associated, e.g., with medical devices, by controlled hydrolytic degradation.
- the in vitro degradation of the polymer into bioactive products was measured and the antibacterial properties examined using gram-negative (Escherichia col ⁇ ) and gram-positive (Staphylococcus aureus) bacteria.
- the cytotoxicity of the polymers and bioactive degradation products were screened using fibroblast cells.
- the polymer degradation products demonstrated antibacterial activity with no cytotoxic effects on fibroblasts.
- the biodegradable antimicrobial-based poly(anhydride-amides) display promise as implants or implant coatings to reduce implant failure resulting from bacterial infections.
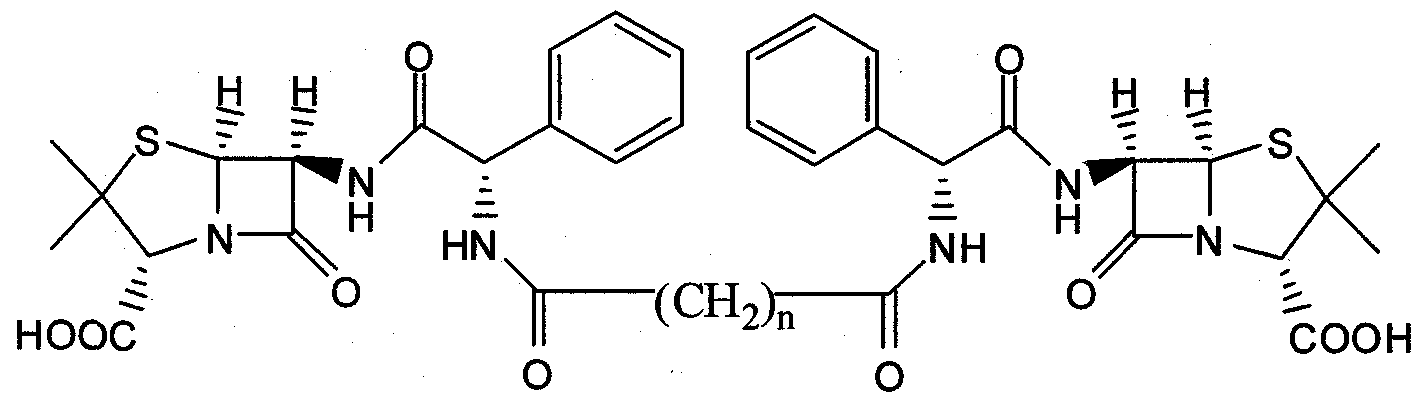
- antimicrobial-based poly(anhydride-amides), 4 comprised of either ampicillin (a) or amoxicillin (b).
- the polymer precursor or diacid (3) was synthesized by direct coupling of the antibiotic (1) (ampicillin, a, or amoxicillin, b) with sebacoyl chloride (2) in dimethylformamide (DMF) containing pyridine.
- the antibiotic (1) ampicillin, a, or amoxicillin, b
- sebacoyl chloride (2) in dimethylformamide (DMF) containing pyridine.
- DMF dimethylformamide
- pyridine acts as acid acceptor by neutralizing the HCl by-product.
- the 1 H and 13 C NMR spectra clearly indicate that the pure products are obtained.
- the polymers could not be generated via melt-condensation conditions typically used to generate drug-containing polyanhydrides. Therefore, the diacids were polymerized under mild conditions in methylene chloride at 0 0 C using triphosgene (1/3 equivalents) as a coupling agent and triethylamine as acid acceptor to form the poly(anhydride-amide) (4) in nearly quantitative yields.
- the chemical incorporation of the ⁇ -lactam antibiotics, ampicillin Ia and amoxicillin Ib, into polymeric backbones yielded polymers with drug loadings of nearly 82 % by weight.
- poly(anhydride-amides) synthesized from these antimicrobials ranged from 14,400-24,800 Da. These moderate molecular weights are similar to other poly(anhydride-ester) molecular weights obtained by melt-polymerization methods.
- Polymers 4 displayed no glass transition temperatures (T g ) from -10 °C to the temperatures just below decomposition (-200 "C).
- the ampicillin-based poly(anhydride-amide) (4a) displayed a higher decomposition temperature (T d ) of 235 °C than the amoxicillin-based polymer (4b) with T d 190 °C.
- the antimicrobial- containing polymers (4) were relatively more soluble in organic solvents than the free antibiotic (1); this characteristic is particularly significant for polymer processability.
- S. aureus (gram-positive) was the most susceptible organism; test compounds showed more potent activity against gram- positive than gram-negative bacteria.
- the slightly smaller inhibition zones on the plates containing E. coli compared to those with S. aureus reflects the difficulty for the test compounds to diffuse across the extra outer membrane present in the gram-negative E. coli.
- the diacids 3 themselves exhibited an inhibitory effect on the bacteria, displaying inhibition zones with diameters in the range of 10-15 mm for E. coli and 31-34 mm for & aureus. This phenomenon is notable because the NMR and in vitro degradation studies indicated that free antibiotic (1) was not formed from diacid 3 in media alone. Inhibition zones were not necessarily expected for the polymers (4), given that their large size may not allow for diffusion through the bacterial membrane. However, small inhibition zones were observed surrounding the polymer samples. These results indicate that the hydrolysis of the polymer (4) into the diacids (3) is sufficient to generate inhibitory concentration of diacid.
- Ampicillin and amoxicillin were obtained from MP Biomedicals (Solon, OH).
- Dimethylformamide (DMF), dimethylacetamide (DMA, anhydrous), pyridine, triethylamine, acetic anhydride, methylene chloride (anhydrous), and diethyl ether were purchased from Fisher (Fair Lawn, NJ). All other fine chemicals and solvents were obtained from Aldrich (Milwaukee, WI).
- Triethylamine was dried over calcium hydride (CaH 2 ) and all other reagents were used with no further purification.
- Weight-averaged molecular weights (M w ) and polydispersity indices (PDI) were determined by gel permeation chromatography (GPC) on a Waters liquid chromatography system consisting of a Waters 410 refractive index detector, a Waters 510 LC pump, and an ISS 200 advanced sample processor.
- GPC gel permeation chromatography
- An IBM Thinkcenter computer running Waters Empower software was used for data collection and processing. Polymers were dissolved (5 mg/mL) in dimethylformamide (DMF) containing 0.1 trifluoroacetic acid (TFA) and filtered through 0.45 ⁇ m poly(tetrafluoroethylene) (PTFE) syringe filters (Whatman, Clifton, NJ) before elution.
- DMF dimethylformamide
- TFA trifluoroacetic acid
- PTFE poly(tetrafluoroethylene)
- Sessile-drop contact angles of phosphate buffer solution on polymer disks were measured using an automated Rame-hart goniometer (Model 250, Netcong, NJ) with DROPimage Advanced software on a Dell Dimension 3000 computer. Angles were measured on three different disks for each polymer and an averaged value was taken.
- Diacid Synthesis (3) Diacids (3) were prepared by reaction of the appropriate antimicrobial 1 with sebacoyl chloride (2) in the presence of a base (pyridine). The preparation of 3a is provided as an example. Ampicillin (Ia) (3.5 g, 10 mmol) was added to DMF (100 mL) and pyridine (4.0 mL, 50 mmol) to afford a suspension. Sebacoyl chloride (2) (1.1 mL, 5.2 mmol) dissolved in dimethylformamide (10 mL) was added dropwise over 5 minutes to the stirring reaction mixture at 0 °C to afford a clear solution.
- Ampicillin (Ia) 3.5 g, 10 mmol
- DMF 100 mL
- pyridine 4.0 mL, 50 mmol
- the reaction was stirred for 2 h at 0 °C, poured over water (400 mL) and acidified to pH ⁇ 2 using 0.1 N hydrochloric acid solution while stirring.
- the white solid (diacid, 3) that formed was isolated by vacuum filtration, washed with water (3x100 mL) and dried overnight under vacuum at room temperature.
- Polymer pellets were prepared by pressing ground polymers (14.0 ⁇ 0.5 mg) into 6 mm diameter x 0.5 mm thick disks in an IR pellet die (International Crystal Laboratories, Garf ⁇ eld, NJ) with a bench-top hydraulic press (Carver model M, Wabash, IN). A pressure of 5,000 psi was applied for 5 min at room temperature. No change in polymer color was observed upon applying pressure.
- Degradation media consisted of phosphate buffer solution (PBS) containing 0.1 M potassium hydrogen phosphate and 0.1 M potassium dihydrogen phosphate.
- PBS phosphate buffer solution
- the pH was adjusted to 7.4 with 1 M sodium hydroxide and/or 1 N hydrochloric acid solutions and pH-measurements were performed on an Accumet® ARl 5 pH-meter (Fisher Scientific, Fair Lawn, NJ).
- diacids were dissolved in deuterated dimethylsulfoxide
- Antimicrobial Assays Polymers and Bacterial Strain. The inhibition zone test of polymers and their degradation products against the bacteria
- Bacteria inocula were suspended in nutrient broth (EMD Chemicals, Gibbstwon, NJ) and culture turbidity adjusted to match that of a 0.5 McFarland standard (see below) to give a bacterial count of approximately 10 colony forming units/mL.
- McFarland 0.5 turbidity standard was prepared according to the protocol described by the National Committee for Clinical Laboratory Standards (NCCLS) (Performance standards for antimicrobial susceptibility testing, hi National Comittee for Clinical Laboratory Standards (NCCLS); Eds.; document M100-S9: Wayne, PA, 1999; 21). Briefly, 0.5 mL of a 1.175% (wt/v) barium chloride dihydrate (BaCl 2 -2H 2 O) (Aldrich, Milwaukee, WI) solution was added to 99.5 mL of 1% (v/v) sulfuric acid (Fisher, Fair Lawn, NJ). The McFarland standard was stored at room temperature in the dark.
- NCCLS National Committee for Clinical Laboratory Standards
- BaCl 2 -2H 2 O Aldrich, Milwaukee, WI
- Disk Diffusion Method The Mueller-Hinton II agar plate surface was inoculated with the broth culture of Escherichia coli and Staphylococcus aureus using a cotton swab (Fisher, Fair Lawn, NJ). Sterile paper disks (6 mm-diameter, Becton Dickinson, Sparks, MD) impregnated with 20 ⁇ L of the test solutions were placed and gently pressed down onto the plates using sterile forceps. Only four disks were placed on each plate to minimize overlap of the inhibition zones. The plates were incubated (Precision Scientific Incubator, South Plainfield, NJ) at 37 °C for 24 h under aerobic conditions. After incubation, the plates were examined and inhibition zones surrounding the disks were measured in millimeters using a ruler. All determinations were carried out in triplicate.
- Cytotoxicity Studies Polymer Samples. Cytotoxicity of the polymers was analyzed by culturing cells in media containing the polymer. To prepare the polymer stock solution, the polymers were first sterilized by UV irradiation for 300 s and dissolved in DMSO to 40 mg/niL. The stock solution was then diluted with cell culture medium to three different final concentrations (1, 0.1 and 0.01 mg/mL). Cell culture media consisted of Dulbecco's Modified Eagle's Medium (Sigma, St. Louis, MO) with 10 % v/v fetal bovine serum, 1 % v/v glutamine, and 50 LVmL of penicillin and streptomycin. Media containing DMSO at the concentration used to prepare the polymer solutions was used as control.
- Dulbecco's Modified Eagle's Medium Sigma, St. Louis, MO
- Media containing DMSO at the concentration used to prepare the polymer solutions was used as control.
- Calcein AM is a cell membrane permeable fluorescent marker only for live cell cytoplasms (Cutler, S., Garcia, A. Biomaterials 2003, 24, 1759-1770).
- Calcein AM staining solution (8 ⁇ M, Molecular Probe, Carlsbad, CA) for 40 min at 4 °C. Fluorescent intensity was measured using a fluorescence plate reader
- Fibroblast growth in polymer solutions was statistically compared to controls at each time point in triplicate. Student's /-test was performed using
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
In one aspect the invention relates to an antibacterial compound of formula II: wherein n is 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12; R is H or OH; or a salt thereof. In other aspects the invention also provides devices that include the compound as well as methods for producing an antibacterial effect with the compound or its salt.
Description
AMPICILLIN AND AMOXICILLIN-BASED POLY(ANHYDRIDE-
AMIDES)
Government Funding
The invention described herein was made with government support under Grant Number DE 13207 awarded by the National Institutes of Health. The United States Government has certain rights in the invention. Related Application^)
This patent document claims the benefit of priority of U.S. application serial No. 60/957,359, filed August 22, 2007, which application is herein incorporated by reference.
Background of the Invention Bacterial infections are a safety issue with medical implants, including stainless steel materials; such infections are extremely difficult to treat due to the development of resistant biofϊlm networks that are not susceptible to traditional antibiotics (Patel R. Biofilms and antimicrobial resistance. Clin Orthop Relat Res, 2005, 437, 41-47). Such infections often lead to multiple revision surgeries, and in some cases, death (Campoccia D, et al., Biomaterials, 2006, 27, 2331- 2339).
Typically, bacterial infections are systemically treated with a course of antibiotics. However, systemic treatments expose the entire body to the drug such that the implant site may not receive the dosage necessary to kill the bacteria (Burrows LL, Khoury AE. Infection of medical devices. In: Wnek GE, Bowlin GL., editor. Encyclopedia of Biomaterials and Biomedical Engineering. New York: Marcel Dekkar; 2004.839-848). Often, this lowered dose results in the development of resistant bacteria, and ultimately biofϊlm formation (Hanssen AD, et al., Clin Orthop Relat Res, 2005, 437, 111-114). Localized antibiotic therapy can also be used to treat bacterial infections associated with medical implants. The most commonly studied localized antibiotic therapy is the physical admixture of antimicrobials within polymer matrices to treat bacterial infections (see von Eiff C, et al., Int J Artif Organs,
2005, 28, 1146-56; Garvin K, Feschuk, Clin Orthop Relat Res, 2005, 437, 105- 110; Gollwitzer H, et al., J Antimicrob Chemother, 2003, 51, 585-591 and Sharkawi T, et al., JBioact and Compat Pol, 2005, 20, 153-168). Polymer coatings for medical device applications have been characterized throughout the literature (see Roosjen A, et al., J Biomed Mater Res Part B, Appl Biomater, 2005, 73B, 347-354; Tedjo C, et al., J Biomed Mater Res, 2007, 82A, 479-491; Song R, et al., Polymer, 2005, 46, 1643-1652; Sarisuta N, et al., IntJPharm, 1999, 186:109-118; and Ruggeri V, et al., J Biomed Mater Res, 2007, 81A, 287- 298).
In spite of the above reports, there is currently a need for agents and methods that are useful for treating bacterial infections. For example, there is a need for agents and methods that are useful for treating bacterial infections associated with medical implants (e.g., orthopedic implants).
Summary of the Invention
In one embodiment, the present invention provides compounds that have antibacterial properties. Accordingly there is provided a compound of the invention which is a compound of formula I:
(I) wherein n is 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12; or a salt thereof.
Certain embodiments of the invention provide a compound of formula EI:
(H)
wherein n is 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12; and each R is independently H or OH; or a salt thereof.
In certain embodiments, each R is OH. In certain embodiments, each R is H.
In certain embodiments, one R is H and one R is OH.
In certain embodiments, n is 4, 5, 6, 7 or 8.
In certain embodiments, n is 6, 7 or 8.
In certain embodiments, n is 8. In one embodiment the invention also provides a pharmaceutical composition comprising a compound of the invention and a pharmaceutically acceptable diluent or carrier.
In one embodiment the invention also provides a medical device having a coating that comprises a compound of the invention. In one embodiment the invention also provides a medical device comprising a compound of the invention.
In certain embodiments, the medical device is an orthopedic implant. hi certain embodiments, the medical device is a knee joint or a hip joint.
In one embodiment the invention also provides a method to promote an anti-bacterial effect in an animal comprising administering a compound of the invention to the animal. hi certain embodiments, the antibacterial effect is against gram negative and/or gram positive bacteria. hi one embodiment the invention also provides a method to promote an antibacterial effect comprising contacting bacteria (e.g., in vivo or in vitro) with a compound of the invention. hi one embodiment the invention also provides a method to reduce biofilm formation on a surface comprising contacting the surface with a compound of the invention. hi certain embodiments, the compound of the invention is incorporated into a coating that is on the surface.
In one embodiment the invention also provides a compound of the invention for use in medical therapy. hi one embodiment the invention also provides the use of a compound of the invention for the manufacture of a medicament for producing an anti- bacterial effect in a mammal. hi one embodiment the invention also provides a compound of the invention, for the prophylactic or therapeutic treatment of a bacterial infection.
The invention also provides processes and intermediates disclosed herein that are useful for preparing compounds of formula (I) or (II) or salts thereof. The compounds, compositions and methods of the invention are useful for preventing biofilm formation and for treating antibiotic resistant bacterial infections, such as those infections associated with medical implants.
Detailed Description
The use of polymeric materials for biological applications such as medical devices (e.g., artificial organs, wound dressings, sutures, pacemakers, intravascular catheters, contact lenses, orthopedic implants) and drug delivery systems has evoked increasing interest. Numerous polymeric biomaterials are implanted each year in humans, yet bacterial infections associated with these medical implants remain a significant issue. For example, nearly half of the two million nosocomial infections occurring in the United States are associated with medical implants. The presence of a foreign body, such as a prosthetic implant, provides a site for microbial adhesion and subsequent colonization, often leading to device-related infections. Current clinical therapies to prevent biomaterial- related infections involve the lengthy administration of antibiotics at high systemic concentrations. Unfortunately, repeated failures of this therapeutic approach require device removal to ensure eradication of the infection and to avoid relapse.
As described herein, degradable biomaterials were designed to release antimicrobials upon polymer degradation to locally prevent infections. This work describes the synthesis of biodegradable antimicrobial-based polymers in which the antimicrobial drugs ampicillin and amoxicillin are chemically incorporated into the polymeric backbone by anhydride and amide bonds, rather
than attached as a side group or physically admixed. Antibacterial properties of the polymers were examined using gram-negative (Escherichia colϊ) and gram- positive (Staphylococcus aureus) bacterial strains. E. coli is the major cause of infections in biliary stents and urinary catheters, and S. aureus is responsible for infections associated with dental and orthopedic implants and fixation devices.
Antibiotic molecules (up to nearly 82% by weight) were incorporated into a polymeric backbone. The polymers were prepared under mild conditions using solution polymerization methods. This milder polymerization method was chosen over typical melt-condensation processes because of the thermal instability of the antibiotics and polymer precursors. No glass transition temperatures (Tg) were observed for the polymers in the measured range from - 10 °C up to polymer decomposition (~ 200 °C). Notably, the antimicrobial-based polymers displayed enhanced solubility in organic solvents relative to the antibiotics alone. This factor is significant, as it determines processability of polymer into solvent-cast films, microspheres, and fibers that may be used as controlled release devices.
In vitro hydrolytic degradation and antimicrobial susceptibility assays of these materials showed that antimicrobial compounds were rapidly released and inhibited the growth of gram-negative (E. coli) and gram-positive (S. aureus) bacteria. These bacteria are responsible for a wide range of medical device failures as a consequence of infection. The biodegradable antimicrobial-based polymers show promise as biomaterials that may reduce implant failure due to their antimicrobial properties.
As these polymers can be used for use as medical implants or implant coatings, their cytotoxicity was evaluated in vitro as a mean to evaluate material biocompatibility. The antimicrobial-based polymers and their degradation products showed no cytotoxic effects when in contact with fibroblast cells. Similar fibroblast proliferation and morphology was observed with different polymer concentrations in the range 0.01-0.1 mg/mL as compared to the controls. Based upon these results, the biodegradable antimicrobial-based poly(anhydride-amides) display promise as implants or coatings to reduce implant failure resulting from bacterial infections.
In cases where compounds are sufficiently basic or acidic, a salt of a compound of formula I or II can be useful as an intermediate for isolating or purifying a compound of formula I or π. Additionally, administration of a compound of formula I or II as a pharmaceutically acceptable acid or base salt may be appropriate. Examples of pharmaceutically acceptable salts are organic acid addition salts formed with acids which form a physiological acceptable anion, for example, tosylate, methanesulfonate, acetate, citrate, malonate, tartrate, succinate, benzoate, ascorbate, α-ketoglutarate, and α-glycerophosphate. Suitable inorganic salts may also be formed, including hydrochloride, sulfate, nitrate, bicarbonate, and carbonate salts.
Pharmaceutically acceptable salts may be obtained using standard procedures well known in the art, for example by reacting a sufficiently basic compound such as an amine with a suitable acid affording a physiologically acceptable anion. Alkali metal (for example, sodium, potassium or lithium) or alkaline earth metal (for example calcium) salts of carboxylic acids can also be made.
The compounds of the invention are useful in a variety of applications where antibacterial activity is desired. Examples of such applications include, but are not limited to, medical, dental and cosmetic uses. The compounds of the invention may be prepared in accordance with methods commonly employed in the field of synthetic chemistry. The compounds can be readily processed into pastes or solvent cast to yield films, coatings, microspheres and fibers with different geometric shapes for design of various medical implants. The compounds of the invention can be incorporated into and/or released from medical implants such as, for example, vascular grafts and stents, bone plates, sutures, Implantable sensors, implantable drug delivery devices, orthopedic devices (e.g. replacement joints such as knees and hips), and stents for tissue regeneration. As described below, the diacid compounds of formula (I) or (II) can be released by hydrolysis of a corresponding anhydride polymer. In one embodiment of the invention, the compounds of formula (I) or (II) can also be incorporated (e.g. dispersed) into the matrix of such an anhydride polymer or
another polymer, and be released from that polymer over time (e.g. upon hydrolysis of the polymer).
Compounds of the invention can also be incorporated into oral formulations and into products such as skin moisturizers, cleansers, pads, plasters, lotions, creams, gels, ointments, solutions, shampoos, tanning products and lipsticks for topical application.
Useful dosages of the compounds can be determined by comparing their in vitro activity, and in vivo activity of the compounds of the invention in animal models. Additionally, useful dosages can be determined by measuring the rate of hydrolysis for a given polymer under various physiological conditions. The amount of a polymer required for use in treatment will vary not only with the particular polymer selected but also with the route of administration, the nature of the condition being treated and the age and condition of the patient and will be ultimately at the discretion of the attendant physician or clinician. The desired dose may conveniently be presented in a single dose or as divided doses administered at appropriate intervals, for example, as two, three, four or more sub-doses per day. The sub-dose itself may be further divided, e.g., into a number of discrete loosely spaced administrations.
The compounds of formula I or II can be formulated as pharmaceutical compositions and administered to a mammalian host, such as a human patient in a variety of forms adapted to the chosen route of administration, e.g., orally or parenterally, by intravenous, intramuscular, topical or subcutaneous routes.
Thus, the present compounds may be systemically administered, e.g., orally, in combination with a pharmaceutically acceptable vehicle such as an inert diluent or an assimilable edible carrier. They may be enclosed in hard or soft shell gelatin capsules, may be compressed into tablets, or may be incorporated directly with the food of the patient's diet. For oral therapeutic administration, the active compound may be combined with one or more excipients and used in the form of ingestible tablets, buccal tablets, troches, capsules, elixirs, suspensions, syrups, wafers, and the like.
The tablets, troches, pills, capsules, and the like may also contain the following: binders such as gum tragacanth, acacia, corn starch or gelatin;
excipients such as dicalcium phosphate; a disintegrating agent such as corn starch, potato starch, alginic acid and the like; a lubricant such as magnesium stearate; and a sweetening agent such as sucrose, fructose, lactose or aspartame or a flavoring agent such as peppermint, oil of wintergreen, or cherry flavoring may be added. When the unit dosage form is a capsule, it may contain, in addition to materials of the above type, a liquid carrier, such as a vegetable oil or a polyethylene glycol. Various other materials may be present as coatings or to otherwise modify the physical form of the solid unit dosage form. For instance, tablets, pills, or capsules may be coated with gelatin, wax, shellac or sugar and the like. A syrup or elixir may contain the active compound, sucrose or fructose as a sweetening agent, methyl and propylparabens as preservatives, a dye and flavoring such as cherry or orange flavor. Of course, any material used in preparing any unit dosage form should be pharmaceutically acceptable and substantially non-toxic in the amounts employed, hi addition, the active compound may be incorporated into sustained-release preparations and devices.
The active compound may also be administered intravenously or intraperitoneally by infusion or injection. Solutions of the active compound or its salts can be prepared in water, optionally mixed with a nontoxic surfactant. Dispersions can also be prepared in glycerol, liquid polyethylene glycols, triacetin, and mixtures thereof and in oils. Under ordinary conditions of storage and use, these preparations contain a preservative to prevent the growth of microorganisms.
The pharmaceutical dosage forms suitable for injection or infusion can include sterile aqueous solutions or dispersions or sterile powders comprising the active ingredient which are adapted for the extemporaneous preparation of sterile injectable or infusible solutions or dispersions, optionally encapsulated in liposomes. In all cases, the ultimate dosage form should be sterile, fluid and stable under the conditions of manufacture and storage. The liquid carrier or vehicle can be a solvent or liquid dispersion medium comprising, for example, water, ethanol, a polyol (for example, glycerol, propylene glycol, liquid polyethylene glycols, and the like), vegetable oils, nontoxic glyceryl esters, and suitable mixtures thereof. The proper fluidity can be maintained, for example,
by the formation of liposomes, by the maintenance of the required particle size in the case of dispersions or by the use of surfactants. The prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal, and the like. In many cases, it will be preferable to include isotonic agents, for example, sugars, buffers or sodium chloride. Prolonged absorption of the injectable compositions can be brought about by the use in the compositions of agents delaying absorption, for example, aluminum monostearate and gelatin. Sterile injectable solutions are prepared by incorporating the active compound in the required amount in the appropriate solvent with various of the other ingredients enumerated above, as required, followed by filter sterilization, hi the case of sterile powders for the preparation of sterile injectable solutions, the preferred methods of preparation are vacuum drying and the freeze drying techniques, which yield a powder of the active ingredient plus any additional desired ingredient present in the previously sterile-filtered solutions.
For topical administration, the present compounds may be applied in pure form, i.e., when they are liquids. However, it will generally be desirable to administer them to the skin as compositions or formulations, in combination with a dermatologically acceptable carrier, which may be a solid or a liquid. Useful solid carriers include finely divided solids such as talc, clay, microcrystalline cellulose, silica, alumina and the like. Useful liquid carriers include water, alcohols or glycols or water-alcohol/glycol blends, in which the present compounds can be dissolved or dispersed at effective levels, optionally with the aid of non-toxic surfactants. Adjuvants such as fragrances and additional antimicrobial agents can be added to optimize the properties for a given use. The resultant liquid compositions can be applied from absorbent pads, used to impregnate bandages and other dressings, or sprayed onto the affected area using pump-type or aerosol sprayers.
Thickeners such as synthetic polymers, fatty acids, fatty acid salts and esters, fatty alcohols, modified celluloses or modified mineral materials can also be employed with liquid carriers to form spreadable pastes, gels, ointments, soaps, and the like, for application directly to the skin of the user.
Examples of useful dermatological compositions which can be used to deliver the compounds of formula I or π to the skin are known to the art; for example, see Jacquet et al. (U.S. Pat. No. 4,608,392), Geria (U.S. Pat. No. 4,992,478), Smith et al. (U.S. Pat. No. 4,559,157) and Wortzman (U.S. Pat. No. 4,820,508).
The ability of a compound of the invention to prevent biofilm formation can be determined as described hereinbelow.
The invention will now be illustrated by the following non-limiting Examples. In the event that a reference designator for a structure is assigned to different structures in different Examples, the reference designator should be read in the context of the specific Example in which it is included.
Example 1 Materials Ampicillin was purchased from MP Biomedicals (Solon, OH). N5N-
Dimethylformamide (DMF), acetic anhydride, dichloromethane, diethyl ether, pyridine, and triethylamine were purchased from Aldrich (Milwaukee, WI). All other reagents and solvents were purchased from Fisher Scientific (Fair Lawn, NJ) and used as received. Triethylamine was dried over calcium hydride, all other reagents were used without further purification. Ampicillin-based Poly(anhydride-amide) Synthesis
Briefly, the ampicillin-based diacid (2) was prepared in DMF with pyridine as a base at 0°C. The polymer (1) was synthesized using triphosgene as a coupling agent and triethylamine as an acid acceptor at 0 °C under nitrogen. The resulting polymer (1) was isolated by pouring over diethyl ether, drying under vacuum at room temperature, and had a Mw = 86,000 and PDI = 1.1.
A 5 % (w/v) ampicillin-based poly(anhydride-amide) (1) solution was prepared in DMF for solvent-casting. The polymer solution (150 μL) was pipetted onto 316L stainless steel coupons (McMaster-Carr, cut to 10 x 30 mm by Rutgers Physics Department Machine Shop). The solvent was evaporated using one of three methods: i) ambient conditions overnight; ii) heating samples to 60 °C for two hours; and iii) heating under vacuum to 60 °C for 45 minutes. Additionally, the ampicillin-based diacid precursor was precipitated onto stainless steel substrates for comparison to the polymer coatings. Peel tests were conducted using the x-cut method in American Standard
Test Method (ASTM) D 3359-02 (n=5) (ASTM D 3359. West Conshohocken, PA: ASTM International; 1998). Two 10 mm X-cuts were scratched into coated coupons using a fresh razor blade. Permacel P-99 tape was adhered to the surface and peeled away at a 180° angle after 90 seconds. Each sample was examined for coating removal and ranked according to the following scale: 5A- no peeling or removal; 4A-trace peeling or removal; 3A-jagged removal along incisions; 2A-jagged removal along most of the incisions; 1 A-removal from most of the area of the X under the tape; and 0 A-removal beyond the area of the X. Sessile contact angle measurements were completed using deionized water on a model 250 Rame-Hart goniometer (Mountain Lakes, NJ) with DROPimage software. One water drop was placed on each coating and measured in real time with a camera. The contact angle was measured as the average value from five water droplets with three angle measurements taken for each drop. The final contact angle value is the average of at least 15 measurements.
Digital microscope images were taken using a Keyence VHX-100 Digital Microscope (Woodcliff Lake, NJ). The coating surfaces were examined up to a magnification of 300Ox to monitor coating uniformity. In Vitro Degradation Study hi vitro degradation studies were conducted at the Office of Science and Engineering Labs in the Division of Chemistry and Materials Science at the Food
and Drug Administration in Rockville, MD. Polymer-coated stainless steel coupons were placed into individual chambers and degraded in the SOTAX USP 4 CE 7 Drug Dissolution Testing System (Allschwil, Switzerland) with a CP 7 ceramic pump, Agilent 8453 UV detection, and WinSOTAX software (see Brown W. Diss Tech, 2005, 12(2), 28-30; and Beyssac E, et al., Diss Tech, 2005, 12(2), 23-25). The samples were degraded at 37 °C in 0.1 M phosphate buffer solution (PBS) pH 7.4. In-line UV detection measurements were taken every five minutes for 4 hours and every hour subsequently at 210 nm. Preliminary experiments were done on a Varian 400-DS USP 7 Dissolution Instrument (Palo Alto, CA) under the same conditions outlined above and with the degradation media separated by HPLC with UV detection. Bacterial Adherence
Bacterial adherence testing was completed at Innovotech Inc. (Calgary, Alberta, Canada) using the Biofilm Eradication Surface Testing (BEST) Assay method. Staphylococcus aureus (ATCC 29213) was obtained from ATCC (Manassas, VA). The coatings were exposed to the Staphylococcus aureus bacteria cells at the required density for biofilm formation (106 colony forming units (CFU)/mL) for 24 hours at 37 °C. Adherent bacteria cells were recovered and quantified compared to negative uncoated controls. In Vitro Degradation with Bacteria Present
Polymer-coated coupons were degraded in BHI (Brain Heart Infusion) media (Fisher, Fair Lawn, NJ) with S. aureus cells for five days (cells were maintained by Linda Rosenberg, Department of Food Science, Rutgers University). The media was sampled at each time point, filtered through 0.45 μm surfactant-free cellulose acetate (SFCA) syringe filters (Nalgene, Rochester, NY), and stored at room temperature until HPLC analysis. The samples were transferred to HPLC vials and analyzed on a Waters 2695 Separations module with a 2487 Dual Wavelength UV Detector and Empower 2 software. The HPLC method was developed to separate the free ampicillin released from the ampicillin diacid at 210 nm on a Gemini Cl 8 column (15O x 4.6 mm, 5 μm, Phenomenex, Torrance, CA). The method was a linear gradient with a mobile phase consisting of 20 mM monobasic potassium phosphate pH 4.0 and
methanol. Five point calibration curves were generated for both the ampicillin and the ampicillin diacid with concentrations ranging from 0.005 mg/mL to 0.5 mg/mL. The degradation media was diluted in monobasic potassium phosphate, if needed, to calculate release within the calibration curve. Before HPLC analysis, the samples were filtered once more through 0.45 μm PTFE syringe filters (Nalgene, Rochester, NY). Full system automation and data analysis were completed using Empower 2 software Build 2154. Complete ampicillin release was ensured by observing complete polymer degradation and calculating the total ampicillin content in the polymer. In Vitro Cytotoxicity
Cytotoxicity of the polymer coatings was analyzed by culturing cells in media containing degraded polymer. Individual ampicillin-based polymer- coated stainless steel plates were placed in 50 mL of cell culture media for 48 hours. Media consisted of Dulbecco's Modified Eagle's Medium (DMEM; Sigma, St. Louis, MO), 10% v/v fetal bovine serum (Atlanta Biologicals, Lawrenceville, GA), penicillin/streptomycin, and L-glutamine.
L929 mouse areolar/adipose fibroblasts (Department of Biomedical Engineering, Rutgers University, Piscataway, New Jersey) were sustained in a cell culture incubator at 37°C and an atmosphere containing 5% CO2. Confluent fibroblasts were removed from the cell culture flask via trypsinization. After media removal, trypsin (2 mg/mL, Sigma, St. Louis, MO) was placed on cells and incubated at 37°C for five minutes to allow cell detachment from the surface of the flask. Trypsin activity was ceased with addition of media and the solution centrifuged for 2 min at 2000 rpm (Thermo Electron 5682 3L GP, Franklin, MA). Cells were seeded at a concentration of 100,000 cells/well in media containing polymer degradation media and analyzed after 24, 48, and 72 hours. All samples were studied in triplicate. A standard curve was generated with cells seeded at 25,000, 50,000, 100,000, and 500,000 cells/well in cell culture media without polymer, analyzed after 24 hours. Imaging was performed with a fluorescent microscope (Olympus 1X81,
Center Valley, PA) with a 10x phase-contrast objective and cell morphology assessed by observation at 24, 48, and 72 hours.
Cell growth was quantified using Calcein AM staining as a live cell assessment every 24 hours for 72 hours. At each time point, cell culture media was removed and cells washed twice with phosphate buffered saline (PBS, pH 7.4, MP Biomedical, Aurora, OH) and refrigerated with 200 μL Calcein AM stain (8 μM, Molecular Probe, Carsbad, CA) for 30 minutes at 4°C. Fluorescent intensity was quantified using a fluorescence plate reader (Cytofluor ® Series 4000, Applied Biosystems, Woodinville, CA) at 485 nm excitation and 530 nm emission. For the three days, live cell numbers were quantified against the standard curve using Microsoft Excel ®. Results and Discussion
Coating Preparation and Characterization
Smooth, uniform polymer coatings containing an average of 12 mg ampicillin-based poly(anhydride-amide) were solvent-cast onto 316L stainless steel. Several parameters were investigated to produce uniform polymer coatings: solvent evaporation rate, time, and temperature. Coatings with solvent evaporation at ambient conditions (room temperature and atmospheric pressure) typically produced cracked surfaces. Solvent evaporation with heat typically produced fewer cracks than ambient evaporation, but the faster evaporation rate typically produced a more uniform coating. Solvent evaporation with heat and vacuum typically provided reproducibly produced smooth, uniform polymer coatings.
The quality of the polymer coatings was further assessed by an ASTM peel test (ASTM D 3359. West Conshohocken, PA: ASTM International). The results from the qualitative peel test are provided in the following table.
Peel Test Analysis of Coating Stability
ASTM Peel Test Scale
5 A-no peeling or removal
4A-trace peeling or removal
3A-jagged removal along incisions
2A-jagged removal along most of the incisions
1 A-removal from most of the area of the X under the tape
OA-removal beyond the area of the X
Peel results showed trace coating removal along x-cut incisions, but overall, the coatings strongly adhere to the stainless steel substrates. These results may be due to a nonspecific chemical interaction between the steel and the coating, ultimately causing the coating to strongly adhere to the stainless steel surface without modification. Upon examination of the heat-processed samples, a jagged removal along the x-cut incisions was noted. Thus, the heat-processed samples typically produce a weaker coating. The vacuum and heat processed samples were typically more robust. The contact angle of the polymer coatings was about 45°, regardless of the solvent evaporation method. These coatings have intermediate contact angles, which are amenable to degradation, yet not immediately solubilized in aqueous media.
The decomposition temperatures of the polymer coatings prepared by various processing methods were measured by TGA to ensure polymer stability during processing. Polymer (1) displayed a slight increase in the decomposition
temperature following the coating preparation, increasing to a Tj of about 210
°C. Most importantly, during the coating process, the polymer does not decompose.
In Vitro Degradation Study
The hydrolytic degradation scheme for the polymer (1) is shown below.
+ 2x H2O
The anhydride bonds are most labile, whereas the amide bonds in the diacid (2) intermediate are expected to hydrolyze more slowly and possibly require enzymatic degradation. Initially, the samples were degraded in the SOTAX USP 4 Dissolution system, which measures cumulative degradation products with real-time UV detection. Decomposition temperatures as measured by TGA (n=3) for each coating condition are provided below.
Polymer 1 was found to hydrolyze in a more controlled manner with more than 70 % of the ampicillin-based polymer (1) coating degraded in 60 hours. Additional release data was obtained from a Varian USP 7 Dissolution Instrument with the ability to separate degradation products by HPLC. Ln these studies, the free ampicillin drug was not detected because the degradation media was PBS at pH 7.4. The conditions were not amenable to amide bond hydrolysis and there was no ampicillin peak observed in the HPLC chromatogram (results not shown). Again, the diacid intermediate (2) was the final degradation product released from the coatings. Bacterial Adherence Study
Staphylococcus aureus growth was monitored during exposure to diacid (2) and polymer (1) coatings, as well as uncoated stainless steel controls. The bacteria cells were seeded to facilitate biofilm growth and were exposed to the coatings for 24 hours. The results of the study are shown in the following table.
The ampicillin-based diacid (2) demonstrated complete prevention of Staphylococcus aureus biofilm. In contrast, polymer 1 itself did not inhibit biofilm formation, indicating that the diacid 2 is a bioactive compounds. No significant difference in bacterial growth was observed between the uncoated control and the polymer samples. Given that Staphylococcus aureus does not grow on the diacid (2) coating, over longer time periods, it was determined that the diacid will prevent bacteria cell growth and biofilm formation. Cytotoxicity Assessment
Fibroblasts were tested in media containing degraded polymer coatings through heat or through a heat and vacuum methods. Cell proliferation was quantified through 72 hours of culture. Fibroblast culture growth was not impeded by the use of polyampicillin in the cell culture medium. All samples showed positive growth throughout the duration of testing with neither polymer impeding cell proliferation and amplification.
Cell morphology was compared to cells cultured in the control, which contained media without any polymer present. The cells displayed their natural, heterogeneous morphology. Generally, the fibroblasts readily attached and
remained attached to the surface despite washing and showed characteristic spreading and extensions. However, cells cultured in media containing polymer 1 coatings prepared with fast solvent evaporation (heat and vacuum) did not extend and spread fibers as frequently as the control and polymer 1 coatings prepared with slow solvent evaporation (heat). Nonetheless, cells showed stellate morphology through 72 hours, proving a low cytotoxicity to the polymer degradation products.
Conclusions
Ampicillin-based poly(anhydride-amide) coatings may improve current clinical therapies for bacterial infections. The coatings have strong adhesion properties to 316L stainless steel, which have not been demonstrated in any other polymer system. The in vitro degradation studies yielded the ampicillin-based diacid as the final degradation product, in PBS pH 7.4. Although the polymer (1) itself was found to be inactive against bacteria growth, it ws found to degrade to the more hydrophilic diacid (2), which completely eradicated Staphylococcus aureus growth and biofilm formation.
Example 2 Poly(anhydride-amides) that include thermally sensitive β-lactam antibiotics were prepared by solution polymerization. Biodegradable antimicrobial-based polymers in which antimicrobial drugs, namely ampicillin and amoxicillin, were chemically incorporated into the polymeric backbone by anhydride and amide bonds were designed to locally prevent infections associated, e.g., with medical devices, by controlled hydrolytic degradation. The in vitro degradation of the polymer into bioactive products was measured and the antibacterial properties examined using gram-negative (Escherichia colϊ) and gram-positive (Staphylococcus aureus) bacteria. The cytotoxicity of the polymers and bioactive degradation products were screened using fibroblast cells. Overall, the polymer degradation products demonstrated antibacterial activity with no cytotoxic effects on fibroblasts. Based upon these results, the biodegradable antimicrobial-based poly(anhydride-amides) display promise as
implants or implant coatings to reduce implant failure resulting from bacterial infections.
Results and Discussion
Polymer Synthesis and Physicochemical Characterization. Based on the functional groups present in the antimicrobial molecules (1), the compounds were chemically incorporated into the polymer backbone through the more nucleophilic primary amine groups, whereas the carboxylic acids were used to ultimately generate the anhydride linkages as outlined in the scheme below.
R = H, a OH, b DMF O 0C Pyridine
4a-b
Synthesis of antimicrobial-based poly(anhydride-amides), 4 comprised of either ampicillin (a) or amoxicillin (b).
The polymer precursor or diacid (3) was synthesized by direct coupling of the antibiotic (1) (ampicillin, a, or amoxicillin, b) with sebacoyl chloride (2)
in dimethylformamide (DMF) containing pyridine. At low reaction temperatures, the primary amino group of the antibiotic reacts with the acyl chlorides of sebacoyl chloride (2) to form 3 in reasonable yields. In this conversion, pyridine acts as acid acceptor by neutralizing the HCl by-product. The 1H and 13C NMR spectra clearly indicate that the pure products are obtained. The potential for side reactions, particularly the phenol of amoxicillin to also react with the sebacoyl chloride, was a concern yet only the expected product (4b) was isolated and used in the polymerization reactions.
With the thermal instability of the antibiotics (1) and antimicrobial-based diacids (3), the polymers could not be generated via melt-condensation conditions typically used to generate drug-containing polyanhydrides. Therefore, the diacids were polymerized under mild conditions in methylene chloride at 0 0C using triphosgene (1/3 equivalents) as a coupling agent and triethylamine as acid acceptor to form the poly(anhydride-amide) (4) in nearly quantitative yields. The chemical incorporation of the β-lactam antibiotics, ampicillin Ia and amoxicillin Ib, into polymeric backbones yielded polymers with drug loadings of nearly 82 % by weight. Molecular weights of the poly(anhydride-amides) synthesized from these antimicrobials ranged from 14,400-24,800 Da. These moderate molecular weights are similar to other poly(anhydride-ester) molecular weights obtained by melt-polymerization methods. Polymers 4 displayed no glass transition temperatures (Tg) from -10 °C to the temperatures just below decomposition (-200 "C). The ampicillin-based poly(anhydride-amide) (4a) displayed a higher decomposition temperature (Td) of 235 °C than the amoxicillin-based polymer (4b) with Td 190 °C. Notably, the antimicrobial- containing polymers (4) were relatively more soluble in organic solvents than the free antibiotic (1); this characteristic is particularly significant for polymer processability.
In Vitro Degradation. Hydrolytic degradation of the poly(anhydride- amides) was measured on polymer disks immersed in PBS at 37 °C for up to 3 days, hi these experiments, only the polymer degradation intermediate (diacid 3) was observed in the degradation media; the free antibiotic (1) was not detected. After two days of in vitro incubation, the anhydride linkages of the polymers (4)
are completely hydrolyzed to yield the intermediate 3. However, no amide cleavage to release the free antibiotic (1) was observed in vitro; hydrolysis of amides typically requires amidases or extreme pH conditions.
Additional 1H NMR studies confirmed this observation, in which diacids (3) were dissolved in DMSO/PBS and monitored. Hydrolytic cleavage of the amide bonds of 3 to release the free antibiotic (1) and sebacic acid was investigated by incubating the diacids (3) at 37 °C for three days. The presence of free antibiotic (1) was not detected by NMR spectroscopy, indicating that the amide bond is stable under these conditions. Antimicrobial Assessment. The Kirby-Bauer or disk diffusion method is a relatively simple and reliable method used to test antimicrobial susceptibility for drugs and provides data that can reliably predict the effectiveness of the compound in vivo (Antibacterial susceptibility tests: dilution and disk diffusion methods. In Manual of Clinical Microbiology; Murray, P.R., Pfaller, M. A., Tenover, F.C., Baron, E.J.,Yolken, R.H., Eds.; ASM Press: Washington DC,
1999; 1526-1543; Performance standards for antimicrobial susceptibility testing. In National Comittee for Clinical Laboratory Standards (NCCLS); Eds.; document M100-S9: Wayne, PA, 1999; 21). The antimicrobial activity of the poly(anhydride-amides) as well as their degradation products against Escherichia coli and Staphylococcus aureus was investigated in vitro using the disk diffusion method. Paper disks impregnated with test solutions were placed on agar plates uniformly inoculated with either E. coli or S. aureus. As the compounds diffused out of the disk over a 24 h incubation period at 37 °C, a circular zone of inhibition was observed around the disk indicating that the compound may be either bactericidal or bacteriostatic. Antibacterial efficiency was assessed by measuring the zone of inhibition surrounding the polymer-impregnated disk. The diameters of the inhibition zones are reported in the Table below against S. aureus and E. coli strains.
Table. Zones of inhibition (mm) of test compounds against Escherichia coli, and Staphylococcus aureus. Inhibition zones were not observed for controls (media only).
Among the bacteria tested, S. aureus (gram-positive) was the most susceptible organism; test compounds showed more potent activity against gram- positive than gram-negative bacteria. The slightly smaller inhibition zones on the plates containing E. coli compared to those with S. aureus reflects the difficulty for the test compounds to diffuse across the extra outer membrane present in the gram-negative E. coli.
Unexpectedly, the diacids 3 themselves exhibited an inhibitory effect on the bacteria, displaying inhibition zones with diameters in the range of 10-15 mm for E. coli and 31-34 mm for & aureus. This phenomenon is notable because the NMR and in vitro degradation studies indicated that free antibiotic (1) was not formed from diacid 3 in media alone. Inhibition zones were not necessarily expected for the polymers (4), given that their large size may not allow for diffusion through the bacterial membrane. However, small inhibition zones were observed surrounding the polymer samples. These results indicate that the hydrolysis of the polymer (4) into the diacids (3) is sufficient to generate inhibitory concentration of diacid.
Cytotoxicity Assessment. The cytotoxicity of the polymers 4 and their degradation products were evaluated in vitro with L929 fibroblasts, the most predominant tissue cells in the body and the standard cells recommended by the
American Society for Testing and Materials (ASTM) in testing materials. For this study, polymer solutions of 0.1 mg/mL and 0.01 mg/mL were prepared in DMSO/media, then fibroblast morphology and proliferation evaluated. The range of polymer concentrations was chosen based on polymer cytotoxicity test protocols developed by the Uhrich laboratory (Schmalenberg, K., Frauchiger, L., Nikkhouy-Albers, L., Uhrich, K. Biomacromolecules 2001, 2, 851-855; Weber, N., Wendel, H., Kohn, J. J. Biomed. Mater. Res. 2005, 72A, 42-47; Sharma, R., Kohn, J., Moghe, P. J. Biomed. Mater. Res. 2004, 69A, 114-123). Solutions containing media and DMSO at the same concentrations used to prepare the polymer solutions were used as controls. Cellular morphologies were compared to the control sample every 24 h over a 3-day period, hi general, no significant morphological changes were observed for cells in polymer solutions of 0.01 mg/mL and 0.1 mg/mL relative to the controls of DMSO/media and media alone. For both 0.01 and 0.1 mg/mL polymer concentrations, the cells were attached onto the surface of the plate and grew with normal stellate morphology over the 3 days. Similarly to the controls of DMSO/media and media alone, high density of cells is observed by the end of the 3 -day period, hi this study, round appearance of cells is not an indication of cell death but of a high cell density as confirmed by live cell counts. Based on the in vitro degradation experiments described herein, the 3 -day cell cultivation time was sufficient to guarantee cell contact with the polymers and to allow complete cell cycles. Cells exhibited positive growth cycles in all the polymer solutions at every concentration and no statistical significance was observed for all polymers with respect to the control (Student's t-test, 95%). hi general, polymers 4 showed good results at both polymer concentrations with respect to cell proliferation/cytotoxicity.
Materials. Ampicillin and amoxicillin were obtained from MP Biomedicals (Solon, OH). Dimethylformamide (DMF), dimethylacetamide (DMA, anhydrous), pyridine, triethylamine, acetic anhydride, methylene chloride (anhydrous), and diethyl ether were purchased from Fisher (Fair Lawn, NJ). All other fine chemicals and solvents were obtained from Aldrich (Milwaukee, WI). Triethylamine was dried over calcium hydride (CaH2) and all other reagents
were used with no further purification.
Methods and Equipment. Nuclear magnetic resonance (1H and 13C NMR) spectra were recorded on a Varian 300 MHz spectrometer. The samples (5-10 mg) were dissolved in a deuterated solvent (OMSO-dβ), which was also used as the internal reference. Infrared (IR) spectra were measured on a Thermo Nicolet/ Avatar 360 FT IR spectrometer, by solvent-casting samples from methylene chloride or methanol onto NaCl plates (if solid).
Weight-averaged molecular weights (Mw) and polydispersity indices (PDI) were determined by gel permeation chromatography (GPC) on a Waters liquid chromatography system consisting of a Waters 410 refractive index detector, a Waters 510 LC pump, and an ISS 200 advanced sample processor. An IBM Thinkcenter computer running Waters Empower software was used for data collection and processing. Polymers were dissolved (5 mg/mL) in dimethylformamide (DMF) containing 0.1 trifluoroacetic acid (TFA) and filtered through 0.45 μm poly(tetrafluoroethylene) (PTFE) syringe filters (Whatman, Clifton, NJ) before elution. Samples were resolved on two mixed-bed GPC columns (Polymer Laboratories, Amherst, MA) (Alltech Associates, Deerfield, IL) placed in series at 25 0C, with DMF containing 0.1% TFA as eluent at a flow rate of 0.8 niL/min. Molecular weights were calibrated relative to narrow molecular weight polystyrene standards (Polysciences, Dorval, Canada).
Thermal analyses were performed on a Perkin-Elmer system consisting of Pyris 1 DSC and TGA 7 analyzers with TAC 7/DX instrument controllers. Perkin-Elmer Pyris software was used for data collection on a Dell OptiPlex GXl 10 computer. For DSC, samples (5 mg) were heated under dry nitrogen gas. Data were collected at heating and cooling rates of 10 °C/min with a two-cycle minimum, starting from -10 °C to just below decomposition (-200 °C). Glass transition temperatures were calculated as half Cp extrapolated. For TGA, samples (10 mg) were heated under dry nitrogen gas. Data were collected at a heating rate of 10 °C/min. Decomposition temperatures were defined as the onset of decomposition.
Elemental analyses were provided by QTI (Whitehouse, NJ). Melting points were obtained with a Mel-Temp apparatus at a heating rate of 1 °C per minute.
Sessile-drop contact angles of phosphate buffer solution on polymer disks were measured using an automated Rame-hart goniometer (Model 250, Netcong, NJ) with DROPimage Advanced software on a Dell Dimension 3000 computer. Angles were measured on three different disks for each polymer and an averaged value was taken.
Poly(anhydride-amide) Precursors: Diacid Synthesis (3). Diacids (3) were prepared by reaction of the appropriate antimicrobial 1 with sebacoyl chloride (2) in the presence of a base (pyridine). The preparation of 3a is provided as an example. Ampicillin (Ia) (3.5 g, 10 mmol) was added to DMF (100 mL) and pyridine (4.0 mL, 50 mmol) to afford a suspension. Sebacoyl chloride (2) (1.1 mL, 5.2 mmol) dissolved in dimethylformamide (10 mL) was added dropwise over 5 minutes to the stirring reaction mixture at 0 °C to afford a clear solution. The reaction was stirred for 2 h at 0 °C, poured over water (400 mL) and acidified to pH~2 using 0.1 N hydrochloric acid solution while stirring. The white solid (diacid, 3) that formed was isolated by vacuum filtration, washed with water (3x100 mL) and dried overnight under vacuum at room temperature.
Ampicillin-sebacic Diacid (3a). Yield: 82% (white powder). 1H NMR (DMSO-<4): δ 9.10 (d, 2H, NH), 8.50 (d, 2H, NH), 7.40 (m, 1OH, ArH), 5.75 (d, 2H, CH), 5.55 (m, 2H, CH), 5.40 (d, 2H, CH), 4.20 (s, 2H, CH), 2.25 (t, 4H, CH2), 1.65-1.35 (m, 12H, CH2), 1.20 (s, 12H, CH3). 13C-NMR (DMSO-(I6): δ 173.7, 172.3, 170.5, 169.1, 138.6, 128.4, 127.8, 127.4, 70.6, 67.5, 64.0, 58.5,
55.7, 35.2, 30.7, 29.0, 26.9, 25.6IR (NaCl, cm"1): 3500-3100 (OH, COOH), 1785 (C=O, COOH), 1730 (C=O, amide I), 1645 (C=O, amide I), 1645 (C=O, amide I), 1535 (NH, amide II), 1510 (NH, amide II). Anal. Calcd.: C, 58.3%; H, 6.1%; N, 9.7%; S, 7.4%. Found: C, 57.7%; H, 6.3%; N, 9.5%; S, 7.4%. Td = 200 0C. Amoxicillin-sebacic Diacid (3b). Yield: 48% (white powder). 1H NMR
(DMSO-J6): δ 8.90 (d, 2H, NH), 8.30 (d, 2H, NH), 7.10 (d, 4H, ArH), 6.65 (d, 4H, ArH), 5.50 (m, 4H, CH), 5.35 (d, 2H, CH), 4.20 (s, 2H, CH), 2.15 (t, 4H,
CH2), 1.60-1.35 (m, 12H, CH2), 1.15 (s, 12H, CH3). 13C-NMR (DMSO-(I6): δ 173.9, 172.2, 171.0, 169.2, 157.0, 128.7, 128.7, 115.2, 70.6, 67.5, 64.0, 58.3, 55.3, 35.2, 30.6, 29.0, 26.9, 25.6 IR (NaCl, cm-1): 3550-3100 (OH, COOH), 1775 (C=O, COOH), 1730 (C=O, amide I), 1660 (C=O, amide I), 1645 (C=O, amide I), 1550 (NH, amide II), 1515 (NH, amide II). Anal. Calcd.: C, 56.2%; H, 5.8%; N, 9.3%; 0, 22.0%; S, 6.7%. Found: C, 55.8%; H, 6.2%; N, 8.7%; O, 22.2%; S, 7.1%. Td = 173 0C.
Polymer Synthesis (4). Diacid (3) (0.30 mmol) was dissolved 20 % (w/v) methylene chloride and triethylamine (1.3 mmol). Triphosgene (0.33 mmol) dissolved in methylene chloride (5 mL) was added dropwise at a rate of 1 drop/4 sec to the stirring reaction mixture at 0 °C to afford a suspension. The reaction was stirred for 2 h at 0 °C under nitrogen and poured over diethyl ether (100 mL). The solid (polymer 4) that formed was isolated by vacuum filtration, washed with acidic water (3x100 mL) and dried overnight under vacuum at room temperature. Any changes to the synthetic procedure are noted below. Polymers 4 are soluble in organic solvents such as methanol, dimethylformamide, and dimethylsulfoxide.
Polyjampicillin-sebacic] (4a). Yield: quantitative (yellow powder). 1H NMR (DMSO-J6): δ 9.10 (d, 2H, NH), 8.45 (d, 2H, NH), 7.40 (m, 1OH, ArH), 5.75 (d, 2H, CH), 5.55 (m, 2H, CH), 5.35 (d, 2H, CH), 4.20 (s, 2H, CH), 2.20 (t, 4H, CH2), 1.60-1.30 (m, 12H, CH2), 1.15 (s, 12H, CH3). IR (NaCl, cm'1): 1795, 1760 (C=O, anhydride), 1730 (C=O, amide I), 1650 (C=O, amide I), 1605 (C=O, amide I), 1540 (NH, amide II), 1515 (NH, amide D). Mw = 24800, PDI = 1.8. Td = 235 0C. Contact angle: 27 °. Poly[amoxicillin-sebacic] (4b). Dichloromethane^imethylacetamide
(1:1) was used as solvent. Yield: quantitative (orange powder). 1H NMR (DMSO-J6): δ 8.90 (d, 2H, NH), 8.30 (d, 2H, NH), 7.10 (d, 4H, ArH), 6.65 (d, 4H, ArH), 5.55 (m, 4H, CH), 5.35 (d, 2H, CH), 4.15 (s, 2H, CH), 2.15 (t, 4H, CH2), 1.55-1.30 (m, 12H, CH2), 1.15 (s, 12H, CH3). IR (NaCl, cm"1): 1800, 1760 (C=O, anhydride), 1720 (C=O, amide I), 1705 (C=O, amide I), 1645 (C=O, amide I), 1575 (NH, amide H), 1515 (NH, amide D). Mw = 14400, PDI = 1.2. Td = 190 0C. Contact angle: 32 °.
In Vitro Degradation: Sample and Media Preparation. Polymer pellets were prepared by pressing ground polymers (14.0 ± 0.5 mg) into 6 mm diameter x 0.5 mm thick disks in an IR pellet die (International Crystal Laboratories, Garfϊeld, NJ) with a bench-top hydraulic press (Carver model M, Wabash, IN). A pressure of 5,000 psi was applied for 5 min at room temperature. No change in polymer color was observed upon applying pressure.
Degradation media consisted of phosphate buffer solution (PBS) containing 0.1 M potassium hydrogen phosphate and 0.1 M potassium dihydrogen phosphate. The pH was adjusted to 7.4 with 1 M sodium hydroxide and/or 1 N hydrochloric acid solutions and pH-measurements were performed on an Accumet® ARl 5 pH-meter (Fisher Scientific, Fair Lawn, NJ).
In Vitro Degradation: Analysis. Hydrolytic degradation of polymers 4 was performed by placing the disks in 20 mL Wheaton glass scintillation vials (Fisher, Fair Lawn, NJ) with 10 mL of PBS, and incubating them at 37 0C with agitation using a controlled environment incubator-shaker (New Brunswick Scientific Co., Edison, NJ) at 60 rpm for 3 days. The buffer solution was replaced by fresh solution (10 mL) every 24 h. The spent media was analyzed by UV spectroscopy at each time point.
Diacid 3 concentration in the degradation media at each time point was determined by UV spectroscopy at λ = 209 run (3a) and 206 nm (3b). UV data were calibrated against standard solutions of known concentrations. Data (average of 3 samples per time point) were presented as a cumulative percent over time.
To further evaluate diacid 3 degradation into the corresponding antimicrobials (1), diacids were dissolved in deuterated dimethylsulfoxide
(DMSO-J6) and PBS (pH = 7.4), at 4:1 (v/v) and incubated at 37 °C over 3 days. Every 24 h, media was analyzed by 1H NMR and 13C NMR spectroscopy for the appearance of free antimicrobial (1).
Antimicrobial Assays: Polymers and Bacterial Strain. The inhibition zone test of polymers and their degradation products against the bacteria
Escherichia coli and Staphylococcus aureus was carried out according to the standardized protocol approved by the National Committee for Clinical
Laboratory Standards (NCCLS) (Antibacterial susceptibility tests: dilution and disk diffusion methods. In Manual of Clinical Microbiology; Murray, P.R., Pfaller, M.A., Tenover, F.C., Baron, E.J.,Yolken, R.H., Eds.; ASM Press: Washington DC, 1999; 1526-1543; Performance standards for antimicrobial susceptibility testing. In National Comittee for Clinical Laboratory Standards (NCCLS); Eds.; document M100-S9: Wayne, PA, 1999; 21).
Stock solutions of all polymers (4), diacids (3) and antimicrobials (1) (10 mg/mL) were prepared in dimethylsulfoxide (DMSO) and then diluted in PBS at 1 :4 (v/v) to give final concentrations of 2.0 mg/mL. A control containing DMSO/PBS was utilized to negate any antibacterial effect due to DMSO.
Two bacterial strains Escherichia coli B and Staphylococcus aureus were used. Bacteria inocula were suspended in nutrient broth (EMD Chemicals, Gibbstwon, NJ) and culture turbidity adjusted to match that of a 0.5 McFarland standard (see below) to give a bacterial count of approximately 10 colony forming units/mL.
Antimicrobial Assays. McFarland 0.5 turbidity standard was prepared according to the protocol described by the National Committee for Clinical Laboratory Standards (NCCLS) (Performance standards for antimicrobial susceptibility testing, hi National Comittee for Clinical Laboratory Standards (NCCLS); Eds.; document M100-S9: Wayne, PA, 1999; 21). Briefly, 0.5 mL of a 1.175% (wt/v) barium chloride dihydrate (BaCl2-2H2O) (Aldrich, Milwaukee, WI) solution was added to 99.5 mL of 1% (v/v) sulfuric acid (Fisher, Fair Lawn, NJ). The McFarland standard was stored at room temperature in the dark.
Mueller-Hinton II agar nutrient medium (Becton Dickinson, Sparks, MD) was prepared according to manufacturer's instructions. Briefly, agar (38 g) was suspended in 1 L of deionized water and heated to boil with agitation to complete dissolution. Solution was autoclaved (Amsco Scientific Series 3021 -S, Apex, NC) at 121 0C for 20 min and the sterile medium (pH = 7.2-7.4, 25 mL) poured at 50-60 °C onto 15 x 100 mm sterile petri dishes (Fisher, Fair Lawn, NJ) to an uniform thickness of 4 mm. Freshly prepared plates were refrigerated at 2 ° to 8 0C before use.
Antimicrobial Assays: Disk Diffusion Method. The Mueller-Hinton II agar plate surface was inoculated with the broth culture of Escherichia coli and Staphylococcus aureus using a cotton swab (Fisher, Fair Lawn, NJ). Sterile paper disks (6 mm-diameter, Becton Dickinson, Sparks, MD) impregnated with 20 μL of the test solutions were placed and gently pressed down onto the plates using sterile forceps. Only four disks were placed on each plate to minimize overlap of the inhibition zones. The plates were incubated (Precision Scientific Incubator, South Plainfield, NJ) at 37 °C for 24 h under aerobic conditions. After incubation, the plates were examined and inhibition zones surrounding the disks were measured in millimeters using a ruler. All determinations were carried out in triplicate.
Cytotoxicity Studies: Polymer Samples. Cytotoxicity of the polymers was analyzed by culturing cells in media containing the polymer. To prepare the polymer stock solution, the polymers were first sterilized by UV irradiation for 300 s and dissolved in DMSO to 40 mg/niL. The stock solution was then diluted with cell culture medium to three different final concentrations (1, 0.1 and 0.01 mg/mL). Cell culture media consisted of Dulbecco's Modified Eagle's Medium (Sigma, St. Louis, MO) with 10 % v/v fetal bovine serum, 1 % v/v glutamine, and 50 LVmL of penicillin and streptomycin. Media containing DMSO at the concentration used to prepare the polymer solutions was used as control.
Cytotoxicity Studies: Fibroblast Cultures. L929 mouse areolar/adipose fibroblasts (Department of Biomedical Engineering, Rutgers University, Piscataway, NJ) were incubated in cell culture medium at 37 °C in a humidified atmosphere (5 % CO2). Confluent fibroblasts were detached by trypsin (0.02 mg/mL, Sigma, St. Louis, MO) and incubated at 37 °C for 5 min. Trypsin activity was stopped by adding cold media, cell pellets were obtained by centrifugation (Thermo Electron 5682 3L GP, Franklin, MA) at 2000 rpm for 2 min. Cells were seeded onto the wells of a 24-well plate containing the polymer solutions at a density of 5 x 104 cells per well and incubated for 3 days. All experiments were performed in triplicate for each polymer solution.
Cytotoxicity Studies: Cell Morphology and Proliferation. Fibroblast morphology in each solution was observed using a fluorescent microscope
(1X81, Olympus, Japan) equipped with 1OX phase-contrast objective at days 1, 2 and 3.
Cell growth in each solution was evaluated every 24 h, after imaging.
Cell numbers were measured using Calcein AM staining as a live-dead assay. Calcein AM solution is a cell membrane permeable fluorescent marker only for live cell cytoplasms (Cutler, S., Garcia, A. Biomaterials 2003, 24, 1759-1770).
At each time point, medium was removed and fibroblasts were gently washed twice with PBS (MP Biomedical, Aurora, OH) then incubated with 200 μL of
Calcein AM staining solution (8 μM, Molecular Probe, Carlsbad, CA) for 40 min at 4 °C. Fluorescent intensity was measured using a fluorescence plate reader
(Cytofluor® Series 4000, Applied Biosystems, Woodinville, CA) at 485 nm excitation and 530 nm emission. Live cell numbers were calculated against a standard curve. All polymers were assayed in triplicate.
Fibroblast growth in polymer solutions was statistically compared to controls at each time point in triplicate. Student's /-test was performed using
Microsoft EXCEL® software at an accepted significance of p < 0.05.
All publications, patents, and patent documents are incorporated by reference herein, as though individually incorporated by reference. The invention has been described with reference to various specific and preferred embodiments and techniques. However, it should be understood that many variations and modifications may be made while remaining within the spirit and scope of the invention.
Claims
1. A compound of formula II:
wherein n is 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12; and each R is independently H or OH; or a salt thereof.
2. The compound of claim 1 , wherein each R is OH.
3. A compound of formula I :
4. The compound of any one of claims 1-3, wherein n is 4, 5, 6, 7, or 8.
5. The compound of claim 4, wherein n is 6, 7, or 8.
6. The compound of claim 5, wherein n is 8.
7. A pharmaceutical composition comprising a compound of formula I or II as described in any one of claims 1-6; or a pharmaceutically acceptable salt thereof; and a pharmaceutically acceptable diluent or carrier.
8. A medical device having a coating that comprises a compound of formula I or II as described in any one of claims 1-6; or a pharmaceutically acceptable salt thereof.
9. A medical device comprising a compound of formula I or II as described in any one of claims 1-6; or a pharmaceutically acceptable salt thereof.
10. The medical device of claim 8 or 9, which is an orthopedic implant.
11. The medical device of claim 8 or 9, which is a knee joint or a hip joint.
12. A method to promote an anti-bacterial effect in an animal comprising administering a compound of formula I or II as described in any one of claims 1- 6; or a pharmaceutically acceptable salt thereof to the animal.
13. A method to promote an antibacterial effect comprising contacting bacteria with a compound of formula I or II as described in any one of claims 1- 6, or with a salt thereof.
14. A method to reduce biofilm formation on a surface comprising contacting the surface with a compound of formula I or II as described in any one of claims 1-6; or a salt thereof.
15. The method of claim 14, wherein the compound of formula I or II or the salt thereof is incorporated into a coating that is on the surface.
16. A compound of formula I or II as described in any one of claims 1 -6, or a pharmaceutically acceptable salt thereof for use in medical therapy.
17. The use of a compound of formula I or II as described in any one of claims 1-6, or a pharmaceutically acceptable salt thereof for the manufacture of a medicament for producing an anti-bacterial effect in a mammal.
18. The compound of formula I or II as described in any one of claims 1-6; or a pharmaceutically acceptable salt thereof, for the prophylactic or therapeutic treatment of a bacterial infection.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US95735907P | 2007-08-22 | 2007-08-22 | |
| US60/957,359 | 2007-08-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009026544A1 true WO2009026544A1 (en) | 2009-02-26 |
Family
ID=40378699
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2008/074070 Ceased WO2009026544A1 (en) | 2007-08-22 | 2008-08-22 | Ampicillin and amoxicillin-based poly(anhydride-amides) |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2009026544A1 (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7985415B2 (en) | 1997-09-10 | 2011-07-26 | Rutgers, The State University Of New Jersey | Medical devices employing novel polymers |
| US8221790B2 (en) | 2000-07-27 | 2012-07-17 | Rutgers, The State University Of New Jersey | Therapeutic polyesters and polyamides |
| US8263060B2 (en) | 2005-05-23 | 2012-09-11 | Rutgers, The State University Of New Jersey | Fast degrading polymers |
| US8361453B2 (en) | 2006-06-06 | 2013-01-29 | Rutgers, The State University Of New Jersey | Iodinated polymers |
| US8747832B2 (en) | 2007-04-12 | 2014-06-10 | Rutgers, The State University Of New Jersey | Biodegradable polyanhydrides with natural bioactive molecules |
| US9108070B2 (en) | 2006-09-13 | 2015-08-18 | Polymerix Corporation | Active agents and their oligomers and polymers |
| US9144579B2 (en) | 2012-08-17 | 2015-09-29 | Rutgers, The State University Of New Jersey | Polyesters and methods of use thereof |
| US9387250B2 (en) | 2013-03-15 | 2016-07-12 | Rutgers, The State University Of New Jersey | Therapeutic compositions for bone repair |
| US9782432B2 (en) | 2012-10-25 | 2017-10-10 | Rutgers, The State University Of New Jersey | Polymers and methods thereof for wound healing |
| US9862672B2 (en) | 2013-05-29 | 2018-01-09 | Rutgers, The State University Of New Jersey | Antioxidant-based poly(anhydride-esters) |
| US10023521B2 (en) | 2014-06-13 | 2018-07-17 | Rutgers, The State University Of New Jersey | Process and intermediates for preparing poly(anhydride-esters) |
| US10251869B2 (en) * | 2014-04-23 | 2019-04-09 | Case Western Reserve University | Compositions and methods of inhibiting metallo-β-lactamases |
| US10543162B2 (en) | 2015-04-10 | 2020-01-28 | Rutgers, The State University Of New Jersey | Kojic acid polymers |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050100516A1 (en) * | 2003-11-07 | 2005-05-12 | Formulated Solutions, Llc | Hydroalcohol based self-tanning composition |
| US20060013851A1 (en) * | 2002-02-07 | 2006-01-19 | Giroux Karen J | Therapeutic polyanhydride compounds for drug delivery |
-
2008
- 2008-08-22 WO PCT/US2008/074070 patent/WO2009026544A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060013851A1 (en) * | 2002-02-07 | 2006-01-19 | Giroux Karen J | Therapeutic polyanhydride compounds for drug delivery |
| US20050100516A1 (en) * | 2003-11-07 | 2005-05-12 | Formulated Solutions, Llc | Hydroalcohol based self-tanning composition |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7985415B2 (en) | 1997-09-10 | 2011-07-26 | Rutgers, The State University Of New Jersey | Medical devices employing novel polymers |
| US8221790B2 (en) | 2000-07-27 | 2012-07-17 | Rutgers, The State University Of New Jersey | Therapeutic polyesters and polyamides |
| US8241668B2 (en) | 2000-07-27 | 2012-08-14 | Rutgers, The State University Of New Jersey | Therapeutic polyesters and polyamides |
| US8263060B2 (en) | 2005-05-23 | 2012-09-11 | Rutgers, The State University Of New Jersey | Fast degrading polymers |
| US8361453B2 (en) | 2006-06-06 | 2013-01-29 | Rutgers, The State University Of New Jersey | Iodinated polymers |
| US9108070B2 (en) | 2006-09-13 | 2015-08-18 | Polymerix Corporation | Active agents and their oligomers and polymers |
| US10092578B2 (en) | 2006-09-13 | 2018-10-09 | Polymerix Corporation | Active agents and their oligomers and polymers |
| US8747832B2 (en) | 2007-04-12 | 2014-06-10 | Rutgers, The State University Of New Jersey | Biodegradable polyanhydrides with natural bioactive molecules |
| US9144579B2 (en) | 2012-08-17 | 2015-09-29 | Rutgers, The State University Of New Jersey | Polyesters and methods of use thereof |
| US9782432B2 (en) | 2012-10-25 | 2017-10-10 | Rutgers, The State University Of New Jersey | Polymers and methods thereof for wound healing |
| US9387250B2 (en) | 2013-03-15 | 2016-07-12 | Rutgers, The State University Of New Jersey | Therapeutic compositions for bone repair |
| US9862672B2 (en) | 2013-05-29 | 2018-01-09 | Rutgers, The State University Of New Jersey | Antioxidant-based poly(anhydride-esters) |
| US10251869B2 (en) * | 2014-04-23 | 2019-04-09 | Case Western Reserve University | Compositions and methods of inhibiting metallo-β-lactamases |
| US10023521B2 (en) | 2014-06-13 | 2018-07-17 | Rutgers, The State University Of New Jersey | Process and intermediates for preparing poly(anhydride-esters) |
| US10543162B2 (en) | 2015-04-10 | 2020-01-28 | Rutgers, The State University Of New Jersey | Kojic acid polymers |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2009026544A1 (en) | Ampicillin and amoxicillin-based poly(anhydride-amides) | |
| Weyell et al. | Tailor-made material characteristics of bacterial cellulose for drug delivery applications in dentistry | |
| US8361453B2 (en) | Iodinated polymers | |
| CN105778126B (en) | Genipin cross-linked biogel and preparation method and application thereof | |
| US7772352B2 (en) | Bioabsorbable and biocompatible polyurethanes and polyamides for medical devices | |
| CN1211130C (en) | Antibacterial polyphosphazene derivatives and their applications | |
| US7411031B2 (en) | Synthesis of polyanhydrides | |
| US20120277852A1 (en) | Coating compositions, methods and coated devices | |
| RS54815B1 (en) | ANTIBIOTIC AND DISPERSANT SUBSTANCE | |
| US8263763B2 (en) | Chemically modified polyaminosaccharide by a hydrocarbyl sultone compound | |
| Leśniak-Ziółkowska et al. | Antibacterial and cytocompatible coatings based on poly (adipic anhydride) for a Ti alloy surface | |
| CZ258598A3 (en) | Neovascularization inhibitor | |
| KR20230126734A (en) | Medical devices, hydrogels and their manufacturing methods and applications | |
| KR101820306B1 (en) | Gallic Acid-Chitosan Complexes and Composition for Wound Healing Comprising Them | |
| Raj et al. | Drug loaded chitosan/aloe vera nanocomposite on Ti for orthopedic applications | |
| Zhang et al. | An antibacterial and biocompatible piperazine polymer | |
| Ismail et al. | Mechanical and antibacterial activities study of gellan gum/virgin coconut oil film embedded norfloxacin | |
| TW202448489A (en) | Wound healing dressings and formulations and methods of use thereof | |
| Mansjur et al. | The effect of coating chitosan from cuttlefish bone (Sepia Sp.) on the surface of orthodontic mini-screw | |
| EP4047012A1 (en) | Antimicrobial cationic peptoid and n-subtituted peptidic copolymers, preparation and uses thereof | |
| US11376345B2 (en) | Hydrogel composition and its uses | |
| Mo et al. | Facile fabrication of chitosan/polysialic acid composite hydrogels crosslinked with tannic acid to enhance wound healing after cesarean section | |
| Quevedo et al. | Study of the synergistic properties of copaiba oil co-electrospun with poly (L-co-D, L lactic acid) and natural rubber latex for application in bioactive wound dressings | |
| RU2815508C1 (en) | Biologically active polymer medical composition | |
| Liu et al. | Ciprofloxacin-Loaded Poly (Trimethylene Carbonate) Implants in |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08798525 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 08798525 Country of ref document: EP Kind code of ref document: A1 |