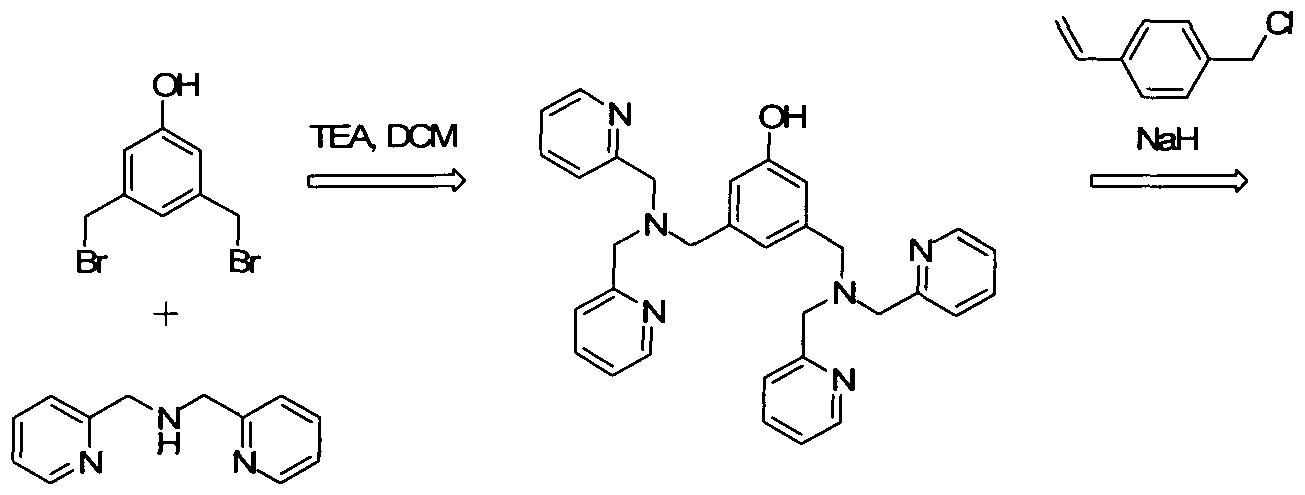
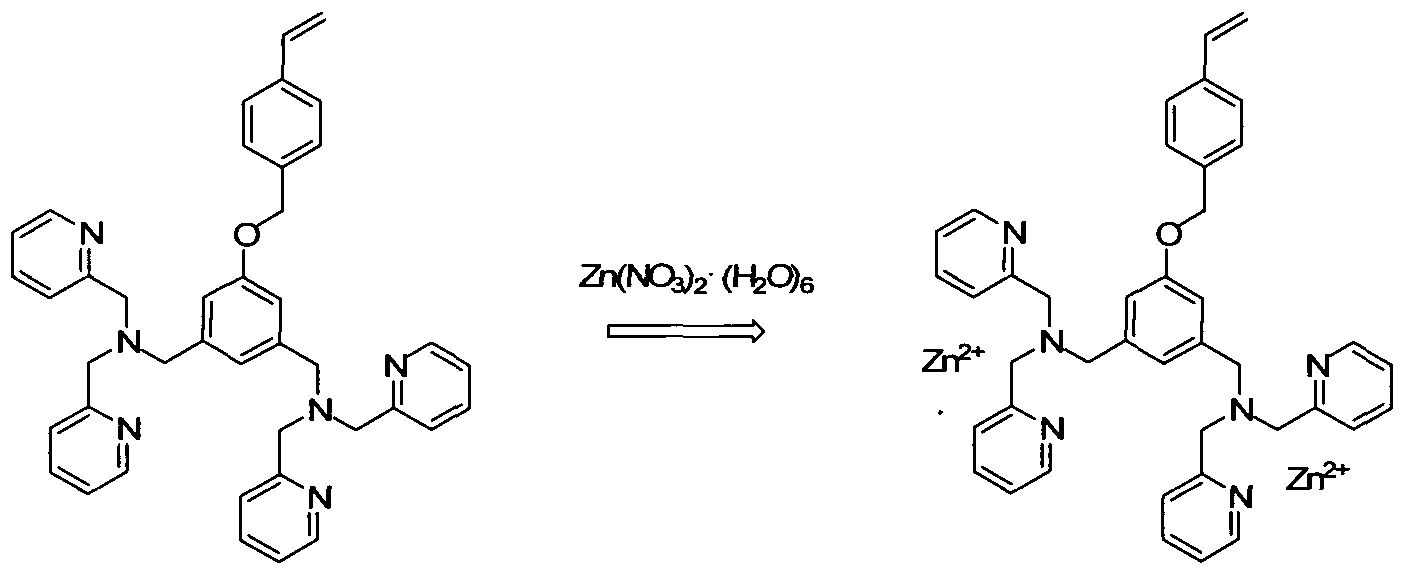
WO2008139204A2 - Functional group imprinted polymers - Google Patents
Functional group imprinted polymers Download PDFInfo
- Publication number
- WO2008139204A2 WO2008139204A2 PCT/GB2008/001681 GB2008001681W WO2008139204A2 WO 2008139204 A2 WO2008139204 A2 WO 2008139204A2 GB 2008001681 W GB2008001681 W GB 2008001681W WO 2008139204 A2 WO2008139204 A2 WO 2008139204A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- polymer
- functional group
- functional monomer
- phosphate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *P(O)(OC1(C2)C(C3)CC2CC3C1)=O Chemical compound *P(O)(OC1(C2)C(C3)CC2CC3C1)=O 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/38—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving specific interaction not covered by one or more of groups B01D15/265 and B01D15/30 - B01D15/36, e.g. affinity, ligand exchange or chiral chromatography
- B01D15/3852—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving specific interaction not covered by one or more of groups B01D15/265 and B01D15/30 - B01D15/36, e.g. affinity, ligand exchange or chiral chromatography using imprinted phases or molecular recognition
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/268—Polymers created by use of a template, e.g. molecularly imprinted polymers
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6803—General methods of protein analysis not limited to specific proteins or families of proteins
- G01N33/6842—Proteomic analysis of subsets of protein mixtures with reduced complexity, e.g. membrane proteins, phosphoproteins, organelle proteins
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2600/00—Assays involving molecular imprinted polymers/polymers created around a molecular template
Definitions
- the present invention relates to a novel class of imprinted polymers, termed 'functional group imprinted polymers' (FIPs), which selectively recognise molecules due to the presence of one or several predetermined functional groups, irrespective of the type, class, substructure or size of the molecule.
- FEPs functional group imprinted polymers'
- polymeric matrices containing recognition sites of pre- determined specificity for a target molecule.
- These polymeric matrices can involve covalent or non-covalent linkage of monomers or a hybrid network thereof.
- these polymeric matrices comprise polymerisable functional monomers which have functionality enabling them to interact with a target molecule.
- the polymerisable functional monomers are allowed to form a complex with a template molecule and then are co-polymerised with a cross-linker in the presence of a porogenic solvent.
- MIPs molecularly imprinted polymers
- MIPs have been developed that bind specifically to cholesterol, L-menthol, the pesticide bentazone, the barbiturate cyclobarbital, or the herbicide 2,4-dichlorophenoxyacetic acid) or a class of molecules (for example, ⁇ -lactam antibiotics).
- Binding selectivity of known MIP technology is represented in Figure 2.
- Molecular imprinting has received considerable attention over the last few decades, and the technology has been successfully applied to a broad range of areas including: biosensors; solid-phase extraction; immunoassays; and chiral separation.
- MIPs have been prepared for the selective recognition of a protein.
- Exemplary approaches to achieve selective MIP formation include employing a combination of metal complex binding sites and substrate inhibitors as template, post-functionalisation and interpenetrating network generation, application of biomolecule substructures (e.g. consensus DNA sequences) as a partial molecular imprint approach to biomolecule recognition and the inclusion of MIPs as part of a polymer composite to facilitate imprinting of large target substrates.
- biomolecule substructures e.g. consensus DNA sequences
- MIP technology is tailored to producing MIPs that bind specifically to a particular target molecule, analogously to antibodies.
- MIPs are unable to discriminate between substrates based solely on the nature of the functional groups present.
- MIPs have been produced according to the 'epitope approach' methodology. These MIPs do exhibit a certain degree of substrate promiscuity, since they are designed to recognise molecules bearing the same structural motif.
- MIPs as described above are not designed to, and cannot, recognise substrates based on the functional groups alone.
- proteomics An exemplary application requiring recognition on the basis of the presence of a functional group lies within the field of proteomics. It is well known that protein phosphorylation is an important post-translational modification that underlies the regulation of signal transduction pathways involved in all cellular processes. Phosphoproteomics, a subdiscipline of proteomics, focusses on the analysis of phosphorylated proteins and it is widely accepted that determination of the phosphoproteome is an enormously challenging task.
- MS mass spectrometry
- IMAC Immobilised Metal Ion Affinity Chromatography
- TiO 2 titanium dioxide affinity chromatography
- IMAC is not necessarily bind all phosphorylated species present in a sample. For example, multiply-phosphorylated species bind with greater affinity than singly-phosphorylated species, and indeed the latter may often not even be retained on a column. Furthermore, IMAC is not selective for phosphorylated residues (the acidic residues aspartate and glutamate are also bound, as are histidine residues).
- Chemical derivatisation is another method of phosphopeptide enrichment. This method is generally not favoured by biologists since it involves a number of synthetic steps, with the implications of incomplete reactions, side-reactions etc.
- Example of proposed derivatisation methods have included: reaction of phosphate esters with ⁇ - diazo substituted resins (with prior protection of carboxylate-bearing residues); base- catalysed ⁇ -elimination of phosphorylated serine or threonine residues to form a Michael Acceptor with subsequent reaction with a nucleophile to effect linkage to a biotinylated affinity tag; and a multi-step protocol involving conversion of a phosphate ester to a phosphoramidate and subsequent derivatisation.
- the invention provides an imprinted polymer comprising a porous polymeric network formed from a functional monomer comprising a chemical moiety capable of interacting with a pre-determined functional group and a crosslinker, wherein the imprinted polymer is capable of selectively recognising and binding to a plurality of molecules which possess the pre-determined functional group, wherein apart from possessing the pre-determined functional group, the plurality of molecules may differ in at least one of molecular class, structure, substructure or size.
- the polymeric network of the imprinted polymer is arranged such that the chemical moieties of the functional monomer form recognition sites capable of binding selectively to the predetermined functional group. Preferably, this binding is highly selective for the pre-determined functional group.
- FIPs functional group imprinted polymers'
- FIPs are designed to recognise, with minimal structural discrimination, compounds exhibiting one or more specific predetermined functional groups whilst at the same time exhibiting selectivity over all compounds with deviating functional group compositions. Discrimination by compound type, class, substructure or size is suppressed as part of the unique recognition and selection process exhibited by FIPs of the invention. This is attributable to the choice of template molecule and porogenic solvent used when forming the polymer, choice of functional monomer, type of polymer backbone and polymer network structure.
- the porous polymeric network formed from the functional monomer, linked by a crosslinker may be covalent, non-covalent or a hybrid covalent/non-covalent network.
- the porous polymeric network is a. macroporous polymeric network.
- a macroporous network is taken to be a network comprising pores with a mean diameter of > 50nm.
- the porous network may however comprise a distribution of pore sizes and some pores of diameter less than 50nm may be present.
- the presence of a macroporous structure is important in enabling accessibility of all sizes of analyte, thereby enabling the imprinted polymer of the invention to be selective on the basis of the presence or absence of the pre-determined function group, but not on the basis of molecular size.
- a macroporous polymeric network will enable access to recognition sites within the polymeric network for molecules of any size able to permeate the macroporous network.
- Molecules possessing the pre-determined functional group will bind selectively to the recognition sites, whereas those not possessing the predetermined functional group will not bind and will diffuse out of the polymeric network.
- the inventors have determined that it is possible to achieve sufficient macroporosity to negate size discrimination whilst also achieving high selectivity for a pre-determined functional group.
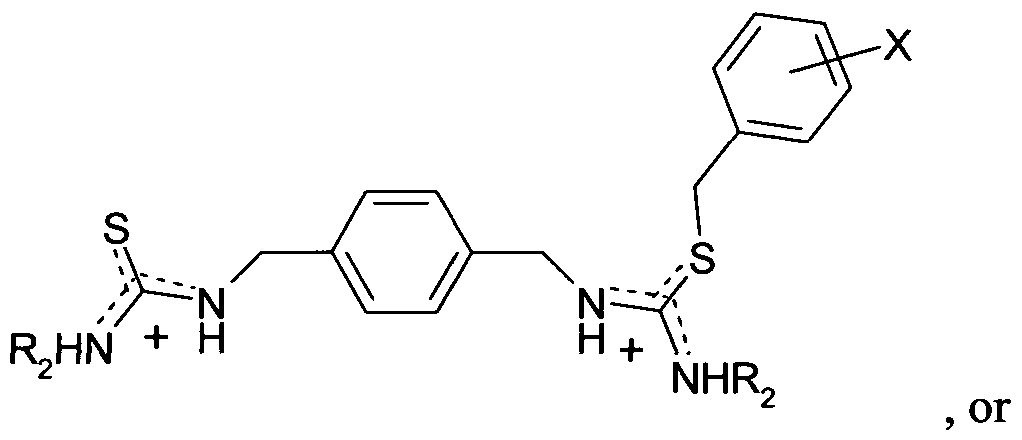
- the pre-determined functional group is selected from the group including, but not limited to: acetal; acid chloride; aldehyde; alkene; alkyne; amide; amidine; amine; ammonium; azide; azo; carbonate; carboxylic acid; carboxylate; ester; ether; guanidine; guanidinium; hydroxyl; isocyanate; isothiocyanate; imidazole; imide; imine; ketal; ketone; nitrate; nitrile; nitro; nitroso; phosphate; phosphate ester; peroxy; phosphine; phosphonate; phosphono; phosphonium; pyridyl; sulphate; sulfonate; sulfone; sulfonic acid; thiol; thiourea; thiourethane; thiouronium; urea; urethane; and uronium.
- the pre-determined functional group is a phosphate or phosphate ester functional group. More preferably, the pre-determined functional group is the phosphate monoester functional group.
- the functional monomer comprises a polymerisable group and one or more chemical moieties capable of interacting with the pre-determined functional group, the one or more chemical moieties being selected from the following: acetal; acid chloride; aldehyde; alkene; alkyne; amide; amidine; amidinium; amine; polyamine; ammonium; azide; azo; carbonate; carboxylic acid; carboxylate; cyclodextrin; ester; ether; guanidine; guanidinium; hydroxyl; isocyanate; isothiocyanate; imidazole; imide; imine; ketal; ketone; nitrate; nitrile; nitro; nitroso; phosphate; phosphate ester; peroxy; phosphine; phosphonate; phosphono; phosphonium; pyridyl; polypyrrole; porphyrin; sulphate; sulfonate;
- the functional monomer is capable of binding strongly to a phosphate or phosphate monoester functional group and comprises one or more chemical moieties selected from amide, amidine, amidinium, carboxylic acid, polyamine, guanidinium, guanidine, polypyrrole, porphyrin, urea, urethane, thiourea, thiourethane and thiouronium as well as metal pyridine dipicolyl amines, metal cyclens (3, 4 and 5), metal coordination enes and metal guanidine chelates.
- chemical moieties selected from amide, amidine, amidinium, carboxylic acid, polyamine, guanidinium, guanidine, polypyrrole, porphyrin, urea, urethane, thiourea, thiourethane and thiouronium as well as metal pyridine dipicolyl amines, metal cyclens (3, 4 and 5), metal coordination enes and metal guanidine
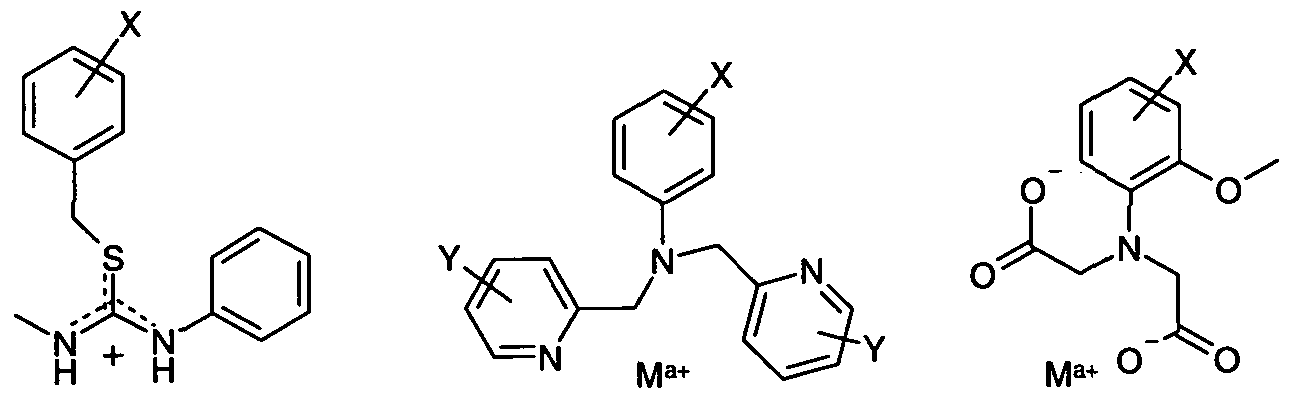
- the functional monomer comprises a thiouronium group, bis(pyridine-2-ylmethyl)amino, (diacetic acid)amino, l,2-bis(o-aminophenoxy)ethane -NJsfJV' JV'-tetraace ⁇ c acid.
- (diacetic acid)amino is l-(o- aminophenoxy)methane- ⁇ N-diacetic acid.
- the thiouronium group has the formula -SC(NHR 2 )(NHR 3 ), wherein R 2 and R 3 are each independently selected from hydrogen, Ci -6 alkyl or 5- or 6-membered aryl or heteroaryl.
- a chelated metal ion is present where the functional monomer comprises a metal pyridine dipicolyl amine, a metal cyclen (3, 4 and 5), a metal coordination ene, metal guanidine chelate, bis(pyridine-2-ylmethyl)amino, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-7V,./V'-diacetic acid) or l,2-bis(o- aminophenoxy)ethane -N,N,N',N'-tet ⁇ aace ⁇ c acid.
- the functional monomer comprises a metal pyridine dipicolyl amine, a metal cyclen (3, 4 and 5), a metal coordination ene, metal guanidine chelate, bis(pyridine-2-ylmethyl)amino, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-7V,./V
- the chelated metal ion is Al 3+ , Fe 3+ , Ca 2+ , Zr 4+ , Ni 2+ , Co 2+ , Cu 2+ , Mn 2+/3+ , Mg 2+ , Ca 2+ , Zn 2+ or Ga 3+ .
- the metal ion is Zn 2+ and for a (diacetic acid)amino group or 1 ,2-bis(o-aminophenoxy)ethane acid the metal ion is Ga 3+ .
- the functional monomer comprises the following structure:
- A comprises (CH 2 ) n , wherein n is 0 or 1 ;
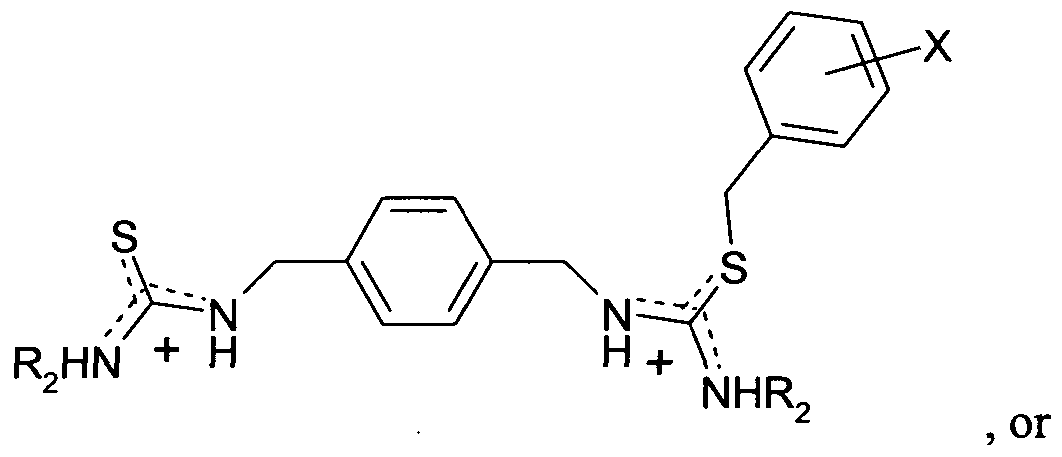
- B comprises (a) a thiouronium group of formula -SC(NHR 2 )(NHR 3 ), bis(pyridine-2- ylmethyl)amino optionally substituted on one or both pyridine rings with Y, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-NJV-diacetic acid), 1 ,2-bis(o-aminophenoxy)ethane-AUV,./V v JV -tetraacetic acid, or an analogue, derivative or ring-fused variant thereof; (b) a ditopic thiouronium group of formula:
- R 2 and R 3 are each independently selected from hydrogen, Ci -6 alkyl (preferably methyl) optionally substituted with Y, Ci -6 haloalkyl (preferably CF 3 or CH 2 CF 3 ), optionally substituted 5- or 6-membered aryl, heteroaryl (preferably phenyl), alkylaryl (preferably benzyl) or alkylheteroaryl; and R 4 is Ci -6 alkyl (preferably methyl) or
- X comprises hydrogen or a polymerisable group
- each occurrence of Y independently comprises hydrogen or a polymerisable group; provided that at least one occurrence of X or Y must be a polymerisable group.
- V is -(CH 2 )- W-(CH 2 )-, wherein W is 1-, 3-, 5- substituted phenyl.
- the preferred option for group B is option (a) as set out above.
- the polymerisable group is a group that can be polymerised by free radical chemistry, for example an acrylate, methacrylate, acrylamide, methacrylamide, or a vinyl group.
- the polymerisable group may be a group that is polymerisable by ring-opening metathesis polymerisation, for example a norbornene or other strained cyclic alkene derivative.
- the polymerisable group comprises a vinyl moiety, such that where X comprises a polymerisable group and n is 0, the functional monomer comprises a styrene group and where n is 1 the functional monomer comprises an alpha-methyl styrene group.
- X is para to -A-B.
- X is hydrogen
- R 1 is hydrogen
- n is 1
- B is -SC(NHR 2 )(NHR 3 ) wherein R 2 is CH 2 CHCH 2 and R 3 is benzyl.
- the functional monomer comprises one of the following structures:
- the functional monomer has the structure:
- the crosslinker is water-soluble.
- the crosslinker comprises an acrylate, a methacrylate, an acrylamide, a methacrylamide, a styrene, a polyethylene glycol (PEG) derivative bearing a polymerisable group at each end of the PEG chain or a 1,2-disubstituted alkene such as a maleimide or a vinyl ether.
- the cross linker is ethylene glycol dimethacrylate, a polyethylene glycol derivative of formula: wherein Z is a polymerisable group
- the polyethylene glycol derivative is a poly(ethylene glycol) diacrylate, a poly(ethylene glycol) dimethacrylate, a poly(ethylene glycol) diacrylamide or a poly(ethylene glycol) dimethacrylamide.
- Z is a norbenene.
- the functional monomer is custom-made.
- the polymer is formed from a functional monomer and at least one co-monomer.
- the co-monomer may be a second functional monomer as defined above or it may be any other monomer comprising a polymerisable group.
- the polymer comprises two or more types of functional monomer, each being capable of interacting with a different pre- determined functional group.
- the polymer is capable of selectively recognising a molecule which possesses one or more of a pre-determined selection of two or more functional groups.
- the polymer comprises two or more types of crosslinker as defined above.
- the present invention provides a method of preparing an imprinted polymer which is capable of selectively recognising and binding to a molecule which possesses a pre-determined functional group, wherein the imprinted polymer is capable of selectively recognising and binding to a plurality of molecules which possess the pre-determined functional group, wherein the plurality of molecules may differ in at least one of molecular type, structure, substructure or size, the method comprising the steps of: a) mixing a template molecule possessing the pre-determined functional group and a functional monomer comprising a chemical moiety capable of interacting with the pre-determined functional group and allowing complex formation; b) providing a porogenic solvent and a cross-linker to the mixture provided in step a) and allowing polymerisation to occur; and c) removing the template molecule thereby providing the imprinted polymer.
- step a) the template molecule undergoes a self-assembly process with one or more functional monomers that will constitute a functional group's binding site within the imprinted polymer.
- This self-assembly process leads to the formation of a template- binding site complex in which the functional monomers are spatially oriented for optimal binding to the pre-determined functional group. Minimisation of influence of substrate size, type etc on binding in the produced imprinted polymer is attributable to the choice of template molecule, functional monomer(s) and polymer backbone.
- the template molecule allows pre-organisation of binding sites (i.e. functional monomers) in solution in such a way that the structural and electronic features of the pre-determined functional group to be imprinted are recognised and during polymerisation become inscribed into the polymer matrix.
- binding sites i.e. functional monomers
- the polymer backbone allows the self-assembly and imprinting process to take place without adverse effect and the polymer backbone is compatible with the solvent or solvent mixtures to be used in the process of applying the FIP to an application.
- the polymer network forms in such a way that it produces pores and pathways which are large enough to ensure accessibility for all compounds that need to be recognised by the FIP.
- the functional monomer, crosslinker and porogenic solvent are selected to ensure formation of a macroporous network. This is achieved by tailoring the solubility of the functional monomer and crosslinker in the porogenic solvent as well as by tailoring the structure of the template molecule. With regard to stability, in order to achieve a macroporous structure the functional monomer, the crosslinker and any co-monomers or co-crosslinkers used should be soluble in the porogenic solvent. As the polymerization reaction proceeds, this solubility will be lost and polymer will begin to precipitate.
- a polymerisation initiator is provided in step b).
- the polymerization initiator may be a thermal or photochemical initiator or a redox initiator.
- Azo initiators and peroxides are preferred for thermal and photochemical polymerisation and are chosen to be soluble in the pre-polymerisation mixture with a half-life consistent with the preferred polymerisation temperature (0 0 C to 65°C typically).
- the initiator is azobisisobutyronitrile.
- Redox initiators are those commonly used to produce hydrogels and are typically persulfates, in combination with amines or Fe salts.
- Ring-opening metathesis polymerisation (ROMP) initiation requires specific ROMP catalysts, such as Grubbs and Hoveyda-type catalysts and other functional group tolerant metathesis catalysts known in the literature.
- the pre-determined functional group is a phosphate group or a phosphate monoester group.
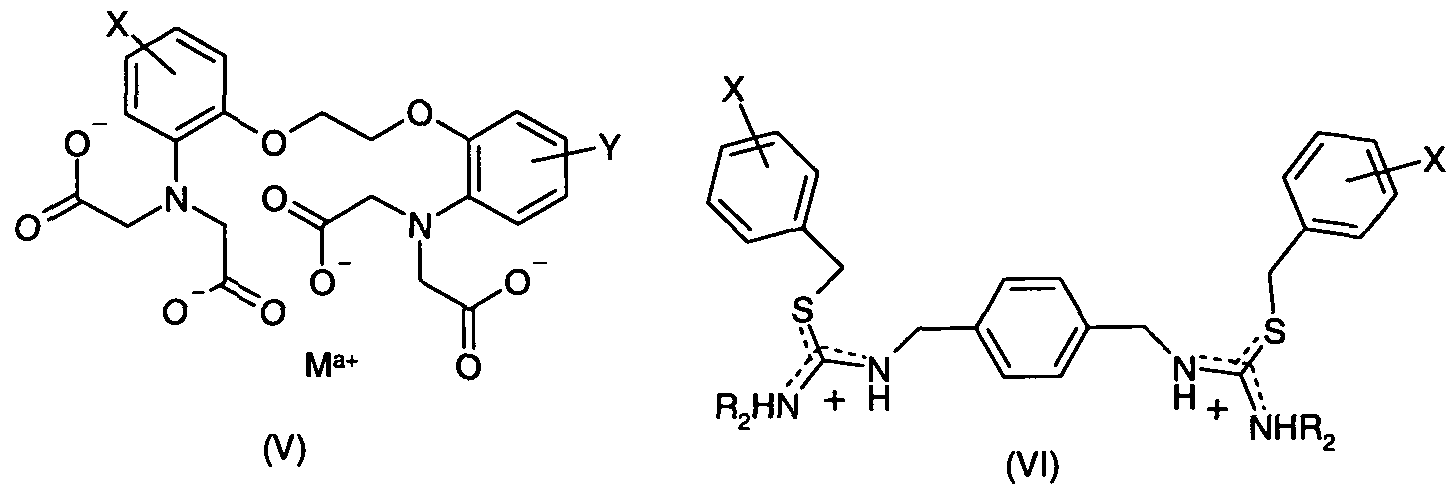
- the template molecule is phosphate-containing compound of formula (VI):
- R 5 is a C 1-20 linear, branched, cyclic or polycyclic hydrocarbon, a dendron or dendrimer, a linear or branched polymer chain, a cyclodextrin or a polypeptide chain; and wherein Z + is a positively charged counter ion, for example an ammonium ion of formula N(R 6 ) 4"1" , wherein each occurrence of R 6 is independently a Ci -20 linear, branched or cyclic hydrocarbon or bulky group selected from a dendron or dendrimer, a linear or branched polymer chain, a cyclodextrin and a polypeptide chain.
- modification of the template molecule with a large sacrificial substituent causes the template molecule itself to act as a pore forming agent.
- Use of space generating substituents such as dendrons and dendrimers, linear and branched polymer chains, cyclodextrins, phosphorylated proteins even is made on the basis of the maximum size of analyte for which functional group selectivity is required.
- such modified templates can be removed/degraded to facilitate their complete extraction from the FIP in step c) and maximise the loading level of the FIP at the same time.
- R 5 is adamantyl
- the functional monomer is a functional monomer as defined above in respect of the first aspect of the invention.
- the porogenic solvent is CHCl 3 , water, a water/solvent mixture wherein the solvent is an alcohol, DMF, DMSO, N-Methyl-2-pyrrolidone (NMP), a water-miscible ionic liquid or acetonitrile or an aqueous buffer, for example TRIS, HEPES, with a background electrolytes (typically NaCl).
- the solvent is an alcohol, DMF, DMSO, N-Methyl-2-pyrrolidone (NMP), a water-miscible ionic liquid or acetonitrile or an aqueous buffer, for example TRIS, HEPES, with a background electrolytes (typically NaCl).
- porogenic solvent is related to the choice of crosslinker, functional monomer, template molecule, co-monomer and co-crosslinker.
- the porogen provides solubility for all monomers, crosslinkers and the template molecule and is also compatible with the polymerisation mechanism. During polymerisation, the porogen generates phase separation so that large droplets of porogen are entrapped in a precipitating polymer matrix. These droplets are freed once the network is set and the porous structure has become permanent through cross-linking.
- the cross-linker is an acrylate, a methacrylate, an acrylamide, a methacrylamide, a styrene, or a 1,2-disubstituted alkenes such as a maleimide or a vinyl ether.
- the cross linker is ethylene glycol dimethacrylate.
- the template molecule is removed in step c) by TFA extraction.
- the present invention provides an imprinted polymer as produced by the second aspect of the invention.
- the present invention provides a method of separating molecules which possess a pre-determined functional group from molecules which do not possess the pre-determined functional group, wherein the method comprises exposing a mixture of molecules to an imprinted polymer according to the first aspect of the invention or as produced by a method according to the second aspect of the invention.
- the method is a chromatographic method and the imprinted polymer is used as a chromatographic stationary phase or in a solid-phase extraction mode.
- all the molecules within the mixture of molecules are soluble in the same solvent, which is preferably an aqueous solvent of high polarity.
- the method is a method of separating out phosphoproteins, phospholipids and/or phosphorylated metabolites from a biological sample.
- Useful solvents during the analysis are those which dissolve the analyte and at the same time minimise non-specific interactions between the analyte and the polymer matrix.
- the present invention provides a functional monomer for use in production of an imprinted polymer, the functional monomer having the structure:
- A comprises (CH 2 ) n , wherein n is 0 or 1 ;
- B comprises (a) a thiouronium group of formula -SC(NHR 2 )(NHR 3 ), bis(pyridine-2- ylmethyl)amino optionally substituted on one or both pyridine rings with Y, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-N,N'-diacetic acid), l,2-bis(o-aminophenoxy)ethane-A r ,N,N' r A ⁇ '-tetraacetic acid, or an analogue, derivative or ring-fused variant thereof; (b) a ditopic thiouronium group of formula:
- V disubstituted with (bis(pyridine-2-ylmethyl)amino), wherein V is an alkyl, haloalkyl, aryl, heteroaryl, naphthalene, anthracene or -(CH 2 )-W-(CH 2 )- group, ⁇ herein W is aryl, heteroaryl, naphthalene or anthracene; Ri comprises hydrogen or OR 4 ;
- R 2 and R 3 are each independently selected from hydrogen, Ci -6 alkyl (preferably methyl) optionally substituted with Y, Ci -6 haloalkyl (preferably CF 3 or CH 2 CF 3 ), optionally substituted 5- or 6-membered aryl or heteroaryl (preferably phenyl); and R 4 is Ci -6 alkyl (preferably methyl) or
- X comprises hydrogen or a polymerisable group
- each occurrence of Y independently comprises hydrogen or a polymerisable group; provided that at least one occurrence of X or Y must be a polymerisable group.
- V is -(CH 2 )- W-(CH 2 )-, wherein W is 1-, 3-, 5- substituted phenyl.
- the functional monomer is as defined above in respect of the first aspect of the invention.
- Figure 1 shows a schematic representation of a conventional molecular imprinting protocol. The individual steps shown are i) complexation in solution; ii) polymerisation; and iii) removal of template molecule.
- Figure 2 shows a schematic representation of selective binding as exhibited by conventional imprinted polymers. As illustrated, the aim of conventional molecular imprinting is to achieve selectivity for one molecule over all other molecules.
- Figure 3 shows a schematic representation of a general protocol for production of a functional group imprinted polymer according to the present invention. The individual steps shown are i) complexation in solution; ii) polymerisation; and iii) removal of template molecule.
- FIG. 4 shows a schematic representation of functional group selectivity as exhibited by a functional group imprinted polymer (FIP) of the present invention.
- FIP functional group imprinted polymer
- Figure 5 shows a schematic representation of a protocol for production of a functional group imprinted polymer according to the present invention.
- Figure 6 shows an experimental design used for comparing selectivity of an imprinted polymer of the invention with that of a conventional MIP.
- NIP non-imprinted polymer
- Figure 8 shows the analyte concentration of TBA 1-naphthyl phosphate and TBA 1- naphthyl acetate in (a) NIP and (b) FIP equilibrations.
- Figures 9 and 10 show the relative values of a phosphorylated substrate and a carboxylated substrate removed during the wash step and extraction step of equilibration experiments.
- the meanings of terms used herein are explained below, and the present invention is illustrated in detail by reference to the following non-limiting examples.
- alkyl as used herein means a straight or branched alkyl group, preferably having 1 to 20 carbon atoms, more preferably having 1 to 6 carbons, most preferably being methyl.
- haloalkyl as used herein means an alkyl group as substituted by one or more halogen (e.g. Cl 1 , Br, F, I).
- a haloalkyl is a C 1-6 haloalkyl, for example -CF 3 or -CH 2 CF 3 .
- 5-6 membered aryl as used herein means a monocyclic aryl group constituted by 5 or 6.
- an aryl group is phenyl.
- 5-6 membered heteroaryl used herein means a monocyclic heteroaryl group, in which the number of atoms forming the ring is 5 or 6 and wherein one or more ring atom is a heteroatom selected from the group consisting of a nitrogen atom, a sulphur atom and an oxygen atom, for example pyridine.
- An aryl or heteroaryl as defined above is optionally substituted with one or more substituents preferably selected from methyl, -CF 3 , -CN or NO 2 , halogen (eg. F, Cl, Br or I) or -NH Z .
- substituents preferably selected from methyl, -CF 3 , -CN or NO 2 , halogen (eg. F, Cl, Br or I) or -NH Z .
- 'functional group' is used herein in accordance with its standard meaning in the art, i.e. a group of atoms within a molecule responsible for a chemical characteristic of that molecule. Where a molecule is referred to as comprising a functional group, it should be appreciated that the functional group does not necessarily stand alone, but may form part of a larger molecule. In the context of the present application, a 'functional group' may also be referred to as a 'chemical moiety'.
- the term 'polymerisable group' refers to any functional group which is capable of reacting with another polymerisable group in a polymerisation reaction.
- a polymerisable group may be one that is capable of undergoing free radical polymerization, for example an acrylate, methacrylate, acrylamide, methacrylamide or a vinyl group.
- the polymerisable group may be a one that is capable of undergoing ring-opening metathesis polymerisation, for example a norbornene or other strained cyclic alkene derivative.
- crosslinker' refers to any molecule that is capable of linking polymer chains. Linkage can be by covalent or non-covalent (e.g. ionic) bonding.
- Reagents The following reagents were purchased from Aldrich Chemical Company, and used without further purification: anhydrous chloroform (>99% contains amylenes as stabilizer); ethylene glycol dimethacrylate (98%); 1-naphthyl phosphate (99%); tetrabutylammonium hydroxide-30-hydrate (98%); 4-vinylbenzyl chloride (90%).
- 2,2'-Azo-bis-isobutyronitrile (>98%) was purchased from Aldrich Chemical Company and recrystallised from hexanes prior to use.
- Tetrabutylammonium hydroxide 40 wt.% solution in water was purchased from Acros and used without further purification.
- l-Methyl-3-phenyl-2 -thiourea was purchased from Fluorochem and used without further purification.
- 2-Acetonaphthone was purchased from Fluka and used without further purification.
- Trifluoroacetic acid (98%) was purchased from Apollo Scientific Ltd, and used without further purification.
- Solvents All solvents (apart from those listed in the preceding paragraph) were obtained from BDH Laboratory Supplies and, unless otherwise specified, used without further purification. All water was purified by simple distillation prior to use.
- HPLC HPLC was performed using a Laserchrom system equipped with degasser, tertiary gradient pump, variable wavelength UV detector, Phenomenex Synergi 4U max C1212 column and column heater (set to 35° C). Data was processed using Data Apex Clarity Lite software. For all experiments, volume of the injection loop was 20 ⁇ L and the flow rate set to 1 mL/ min. HPLC-grade solvents were used.
- IR Infrared spectra were recorded on a Satellite FTIR spectrometer (manufactured by Mattson) using KBr plates.
- MS High resolution mass spectra were recorded by the Imperial College mass spectrometry service using chemical ionisation with ammonium ions (NH 4 + ) or using electrospray mass spectrometry.
- TLC Pre-coated silica gel F 254 / 366 Merck Kieselgel 60 A plates were used, and visualised using UV light (254 nm).
- Melting Points Melting points were determined using a Sanyo Gallenkamp hot stage apparatus, and are uncorrected.
- Elemental analysis was carried out by Stephen Boyer at the London Metropolitan University
- 1-Adamantyl phosphoric acid was produced according to the teaching of B ⁇ hringer, H.; Vogt, H., Arch. Pharm. (Weinheim). 1977, 310, (11), 894-905.
- ⁇ phenylisothiouronium chloride (2.0 eq, 0.211 mmol, 67.3 mg) were taken up in anhydrous CHCl 3 (600 ⁇ L).
- the vial was temporarily sealed under an inert atmosphere using a rubber septum, and ethylene glycol dimethacrylate ⁇ EDMA> (30 eq to tetrabutylammonium 1-adamantyl phosphate, 3.18 mmol, 600 ⁇ L) and azobis- ' c obutyronitrile ⁇ AIBN> (2 mol% to all double bonds, 0.131 mmol, 21.5 mg) were added.
- the polymer was dried in vacuo and then stirred in 5% v/v TFA in CHCl 3 (150 mL) at rt for 20 h. Following this, the polymer was once again dried in vacuo and washed by means of soxhlet extraction using 5% v/v acetic acid in CHCl 3 (100 mL) over 23 h (oil bath: 90-100°C). Finally, the polymer was further washed by means of soxhlet extraction using CHCl 3 (150 mL) over 67 h (oil bath: 90-100°C). The polymer was then dried in vacuo to afford a free- flowing, off-white solid (390 mg).
- the protocol for preparing an imprinted polymer (FIP) of the invention is represented schematically in figure 5.
- the polymer was dried in vacuo and then stirred in 5% v/v TFA in CHCl 3 (150 mL) at it for 20 h. Following this, the polymer was once again dried in vacuo and washed by means of soxhlet extraction using 5% v/v acetic acid in CHCl 3 (100 mL) over 23 h (oil bath: 90- 100° C). Finally, the polymer was further washed by means of soxhlet extraction using CHCl 3 (150 mL) over 67 h (oil bath: 90-100°C). The polymer was then dried in vacuo to afford a free-flowing, off-white solid (401 mg).
- the bound substrate was then released by stirring the FIP and NIP in 5% v/v TFA in CHCl 3 (150 mL) for 27 h at rt. Afterwards the FIP and NIP particles were isolated via vacuum filtration, and washed through with CHCl 3 (50 mL). The filtrate was then concentrated in vacuo and dried in a vacuum oven (35-45°C, 36 h), and then analysed by HPLC. The relative value of each substrate removed during the substrate extraction step is shown in Figure 10.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Analytical Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Physics & Mathematics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Bioinformatics & Computational Biology (AREA)
- Biophysics (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Biomedical Technology (AREA)
- Organic Chemistry (AREA)
- Immunology (AREA)
- Biotechnology (AREA)
- Cell Biology (AREA)
- Microbiology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Food Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Polyesters Or Polycarbonates (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
Abstract
The present invention relates to a novel class of imprinted polymers, termed 'functional group imprinted polymers' (FIPs), which selectively recognise molecules due to the presence of one or several predetemined funtional groups, irrespective of the type, class, substructure or size of the molecule.
Description
FUNCTIONAL GROUP IMPRINTED POLYMERS
The present invention relates to a novel class of imprinted polymers, termed 'functional group imprinted polymers' (FIPs), which selectively recognise molecules due to the presence of one or several predetermined functional groups, irrespective of the type, class, substructure or size of the molecule.
Molecular imprinting is a technique that has been known since the 1970s, which involves the preparation of polymeric matrices containing recognition sites of pre- determined specificity for a target molecule. These polymeric matrices can involve covalent or non-covalent linkage of monomers or a hybrid network thereof. Typically, these polymeric matrices comprise polymerisable functional monomers which have functionality enabling them to interact with a target molecule. The polymerisable functional monomers are allowed to form a complex with a template molecule and then are co-polymerised with a cross-linker in the presence of a porogenic solvent. Subsequent removal of the template molecule from the resulting polymer affords binding molecules that are spatially arranged in accordance with the original template molecule, with cavities that are complementary to the size and shape of the template molecule. Such molecularly imprinted polymers (MIPs) are capable of re-binding the template molecule, with selectivity over structurally related molecules, even successfully competing with antibodies. A typical molecular imprinting protocol is shown in Figure 1.
The aim of previously published work on MIPs has been to optimise MIP technology so as to make the binding sites as uniform and 'monoclonal' as possible in order that they exhibit specificity for just one particular molecule (for example, MEPs have been developed that bind specifically to cholesterol, L-menthol, the pesticide bentazone, the barbiturate cyclobarbital, or the herbicide 2,4-dichlorophenoxyacetic acid) or a class of molecules (for example, β-lactam antibiotics). Binding selectivity of known MIP technology is represented in Figure 2.
Molecular imprinting has received considerable attention over the last few decades, and the technology has been successfully applied to a broad range of areas including: biosensors; solid-phase extraction; immunoassays; and chiral separation.
One area in which there has been much recent activity is the application of MIPs to protein recognition. There are numerous examples in which MIPs have been prepared for the selective recognition of a protein. Exemplary approaches to achieve selective MIP formation include employing a combination of metal complex binding sites and substrate inhibitors as template, post-functionalisation and interpenetrating network generation, application of biomolecule substructures (e.g. consensus DNA sequences) as a partial molecular imprint approach to biomolecule recognition and the inclusion of MIPs as part of a polymer composite to facilitate imprinting of large target substrates.
Turner et al. {Biotechnol Prog. 2006, 22(6), 1474-1489) and Bossi et al. (Biosens, Bioelectron. 2007, 22(6), 1131-1137) have reviewed the area of MIPs and protein imprinting. Both articles are comprehensive reviews summarising the most important strategies for preparing MIPs for protein recognition. A wide range of selectivities have been claimed in these studies usually making only limited comparison to related or other proteins or peptide structure on the way. References cited in both publications are included explicitly herein as part of the summary of the state of art.
Nonetheless, there are limitations of known MIP technology and conventional MIPs are unsuited to certain applications, in particular those where it is desirable to achieve recognition of a molecule based on the presence of one or more pre-determined functional groups, but irrespective of the overall structure, size or class of molecule. For example, known MIP technology is tailored to producing MIPs that bind specifically to a particular target molecule, analogously to antibodies. However, MIPs are unable to discriminate between substrates based solely on the nature of the functional groups present. MIPs have been produced according to the 'epitope approach' methodology. These MIPs do exhibit a certain degree of substrate promiscuity, since they are designed to recognise molecules bearing the same
structural motif. This is typified by the work of Minoura et al., wherein a MIP imprinted with a tripeptide (the 'epitope') was used to bind a nonapeptide containing that same tripeptide sequence. (Rachkov, A et al, J. Chromatogr. 2000, 889, 111- 118). The 'epitope-approach' technique has been taken up more recently by others such as Nishino et al. to produce protein-specific sorbents and chromatographic separations thereof.
Generally a wide range of selectivities have been claimed in studies of MEPs for protein recognition, usually making only limited comparison to related or other proteins. The MIPs as described above are not designed to, and cannot, recognise substrates based on the functional groups alone.
An exemplary application requiring recognition on the basis of the presence of a functional group lies within the field of proteomics. It is well known that protein phosphorylation is an important post-translational modification that underlies the regulation of signal transduction pathways involved in all cellular processes. Phosphoproteomics, a subdiscipline of proteomics, focusses on the analysis of phosphorylated proteins and it is widely accepted that determination of the phosphoproteome is an enormously challenging task.
Traditional methods of analysing and studying phosphoproteins include 32P radiolabelling, 31P NMR and immunob lotting. However, mass spectrometry (MS) is now the method of choice in contemporary phosphoproteomics, and a number of highly specialised MS experiments have been developed for analysis of the phosphoproteome. The very definition of phosphoproteomics demands that every phosphorylated component be detected and identified. Yet despite the advancements in MS technology, analysis of the phosphoproteome is still a complex task. Therefore, to properly benefit from current MS/ phosphoproteomics technology, there has arisen the need to enrich for phosphorylated species from complex biological mixtures prior to MS analysis. The two currently preferred methods of selecting for phosphorylated species are Immobilised Metal Ion Affinity Chromatography (IMAC) and titanium dioxide affinity chromatography (TiO2).
IMAC exploits the affinity of negatively-charged phosphates for the positively- charged Fe"1' Al111 or Ga111 metal ions. A sample containing phosphopeptides is exposed, typically by passing through a column, to Fe111, Al111 or Ga111 metal ions bound to a chelating support (typically iminodiacetic acid or nitrilotriacetic acid). Phosphopeptides bind to the metal ions and are retained on the column whilst unbound non-phosphorylated compounds are separated by elution through the column. After separation, the phosphopeptides are removed using a salt or pH gradient. A drawback of IMAC is that it does not necessarily bind all phosphorylated species present in a sample. For example, multiply-phosphorylated species bind with greater affinity than singly-phosphorylated species, and indeed the latter may often not even be retained on a column. Furthermore, IMAC is not selective for phosphorylated residues (the acidic residues aspartate and glutamate are also bound, as are histidine residues). It has been reported that methyl esterifϊcation of acidic residues prior to IMAC enrichment can improve selectivity for phosphate monoesters over carboxylates, but with this there are complications due to incomplete esterification, side-reactions (for example, with asparagine residues), and increased sample complexity hindering subsequent MS analysis.
In titanium dioxide affinity chromatography, phosphopeptides are bound to a titanium dioxide stationary phase under acidic conditions, and subsequently desorbed under conditions of high pH. Drawbacks of this method again include the preferential enrichment for multiply-phosphorylated peptides, the fact that it does not reliably bind all phosphopeptides present in a sample and non-selectivity for phosphate monoesters, with aspartate and glutamate also being bound.
Chemical derivatisation is another method of phosphopeptide enrichment. This method is generally not favoured by biologists since it involves a number of synthetic steps, with the implications of incomplete reactions, side-reactions etc. Example of proposed derivatisation methods have included: reaction of phosphate esters with α- diazo substituted resins (with prior protection of carboxylate-bearing residues); base- catalysed β-elimination of phosphorylated serine or threonine residues to form a Michael Acceptor with subsequent reaction with a nucleophile to effect linkage to a
biotinylated affinity tag; and a multi-step protocol involving conversion of a phosphate ester to a phosphoramidate and subsequent derivatisation.
Over the last few years there has been a significant increase in the number of publications geared towards optimising phosphopeptide enrichment, underlining its importance and relevance in contemporary proteomics. And yet, as pointed out in a recent comparison of the current methods (Bodenmiller, B.; Mueller, L. N.; Mueller,
M.; Domon, B.; Aebersold, R., Nat. Methods 2007, 4, (3), 231-237), "...at present, no single method is sufficient for a comprehensive phosphoproteome analysis". Similarly, it is desirable to separate phospholipids from a sample containing non- phospholipids and phosphorylated metabolites from non-phosphorylated metabolites.
Thus, there is a real requirement for a simple and effective enrichment method that is specific for phosphorylated species over non-phosphorylated species and that binds all phosphorylated species present in a complex mixture. It has now been determined that by careful selection of monomers, crosslinkers, template molecules and porogenic solvents, an imprinted polymer can be produced which contains recognition sites which are highly specific for compounds possessing a pre-determined functional group, but which do not discriminate on the basis of the overall structure of the compound. The present invention thus provides a class of imprinted polymers (referred to as 'Functional Group Imprinted Polymers') which can achieve selection based only on the presence of a pre-determined functional group. In contrast to conventional MIPs, which are designed to exhibit selectivity for one particular molecule or molecular substructure, the FIPs of the invention exploit "sub-molecular recognition", exhibiting recognition for a specific functional group rather than the overall structure of a molecule.
Therefore, in a first aspect, the invention provides an imprinted polymer comprising a porous polymeric network formed from a functional monomer comprising a chemical moiety capable of interacting with a pre-determined functional group and a crosslinker, wherein the imprinted polymer is capable of selectively recognising and binding to a plurality of molecules which possess the pre-determined functional
group, wherein apart from possessing the pre-determined functional group, the plurality of molecules may differ in at least one of molecular class, structure, substructure or size.
The polymeric network of the imprinted polymer is arranged such that the chemical moieties of the functional monomer form recognition sites capable of binding selectively to the predetermined functional group. Preferably, this binding is highly selective for the pre-determined functional group.
These 'functional group imprinted polymers' (FIPs) are designed to recognise, with minimal structural discrimination, compounds exhibiting one or more specific predetermined functional groups whilst at the same time exhibiting selectivity over all compounds with deviating functional group compositions. Discrimination by compound type, class, substructure or size is suppressed as part of the unique recognition and selection process exhibited by FIPs of the invention. This is attributable to the choice of template molecule and porogenic solvent used when forming the polymer, choice of functional monomer, type of polymer backbone and polymer network structure.
In a preferred embodiment, the porous polymeric network formed from the functional monomer, linked by a crosslinker may be covalent, non-covalent or a hybrid covalent/non-covalent network.
Preferably, the porous polymeric network is a. macroporous polymeric network. A macroporous network is taken to be a network comprising pores with a mean diameter of > 50nm. The porous network may however comprise a distribution of pore sizes and some pores of diameter less than 50nm may be present. The presence of a macroporous structure is important in enabling accessibility of all sizes of analyte, thereby enabling the imprinted polymer of the invention to be selective on the basis of the presence or absence of the pre-determined function group, but not on the basis of molecular size. A macroporous polymeric network will enable access to recognition sites within the polymeric network for molecules of any size able to permeate the
macroporous network. Molecules possessing the pre-determined functional group will bind selectively to the recognition sites, whereas those not possessing the predetermined functional group will not bind and will diffuse out of the polymeric network. Advantageously, the inventors have determined that it is possible to achieve sufficient macroporosity to negate size discrimination whilst also achieving high selectivity for a pre-determined functional group.
In a preferred embodiment the pre-determined functional group is selected from the group including, but not limited to: acetal; acid chloride; aldehyde; alkene; alkyne; amide; amidine; amine; ammonium; azide; azo; carbonate; carboxylic acid; carboxylate; ester; ether; guanidine; guanidinium; hydroxyl; isocyanate; isothiocyanate; imidazole; imide; imine; ketal; ketone; nitrate; nitrile; nitro; nitroso; phosphate; phosphate ester; peroxy; phosphine; phosphonate; phosphono; phosphonium; pyridyl; sulphate; sulfonate; sulfone; sulfonic acid; thiol; thiourea; thiourethane; thiouronium; urea; urethane; and uronium.
Preferably, the pre-determined functional group is a phosphate or phosphate ester functional group. More preferably, the pre-determined functional group is the phosphate monoester functional group.
In a preferred embodiment, the functional monomer comprises a polymerisable group and one or more chemical moieties capable of interacting with the pre-determined functional group, the one or more chemical moieties being selected from the following: acetal; acid chloride; aldehyde; alkene; alkyne; amide; amidine; amidinium; amine; polyamine; ammonium; azide; azo; carbonate; carboxylic acid; carboxylate; cyclodextrin; ester; ether; guanidine; guanidinium; hydroxyl; isocyanate; isothiocyanate; imidazole; imide; imine; ketal; ketone; nitrate; nitrile; nitro; nitroso; phosphate; phosphate ester; peroxy; phosphine; phosphonate; phosphono; phosphonium; pyridyl; polypyrrole; porphyrin; sulphate; sulfonate; sulfone; sulfonic acid; thiol; thiourea; thiourethane; thiouronium; urea; urethane; uronium.
Preferably, the functional monomer is capable of binding strongly to a phosphate or phosphate monoester functional group and comprises one or more chemical moieties selected from amide, amidine, amidinium, carboxylic acid, polyamine, guanidinium, guanidine, polypyrrole, porphyrin, urea, urethane, thiourea, thiourethane and thiouronium as well as metal pyridine dipicolyl amines, metal cyclens (3, 4 and 5), metal coordination enes and metal guanidine chelates.
More preferably, the functional monomer comprises a thiouronium group, bis(pyridine-2-ylmethyl)amino, (diacetic acid)amino, l,2-bis(o-aminophenoxy)ethane -NJsfJV' JV'-tetraaceύc acid.. Preferably, (diacetic acid)amino is l-(o- aminophenoxy)methane-ΛζN-diacetic acid. Preferably, the thiouronium group has the formula -SC(NHR2)(NHR3), wherein R2 and R3 are each independently selected from hydrogen, Ci-6 alkyl or 5- or 6-membered aryl or heteroaryl.
Preferably, a chelated metal ion is present where the functional monomer comprises a metal pyridine dipicolyl amine, a metal cyclen (3, 4 and 5), a metal coordination ene, metal guanidine chelate, bis(pyridine-2-ylmethyl)amino, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-7V,./V'-diacetic acid) or l,2-bis(o- aminophenoxy)ethane -N,N,N',N'-tetτaaceύc acid. Preferably, the chelated metal ion is Al3+, Fe3+, Ca2+, Zr4+, Ni2+, Co2+, Cu2+, Mn2+/3+, Mg2+, Ca2+, Zn2+ or Ga3+. Preferably, for bis(pyridine-2-ylmethyl)amino the metal ion is Zn2+ and for a (diacetic acid)amino group or 1 ,2-bis(o-aminophenoxy)ethane
acid the metal ion is Ga3+.
In a preferred embodiment, the functional monomer comprises the following structure:
(I) wherein A comprises (CH2)n, wherein n is 0 or 1 ;
B comprises (a) a thiouronium group of formula -SC(NHR2)(NHR3), bis(pyridine-2- ylmethyl)amino optionally substituted on one or both pyridine rings with Y, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-NJV-diacetic acid), 1 ,2-bis(o-aminophenoxy)ethane-AUV,./VvJV -tetraacetic acid, or an analogue, derivative or ring-fused variant thereof; (b) a ditopic thiouronium group of formula:
(c) V disubstituted with (bis(pyridine-2-yhnethyl)amino), wherein V is an alkyl, haloalkyl, aryl, heteroaryl, naphthalene, anthracene or -(CH2)-W-(CH2)- group, wherein W is aryl, heteroaryl, naphthalene or anthracene; > R] comprises hydrogen or OR4;
R2 and R3 are each independently selected from hydrogen, Ci-6 alkyl (preferably methyl) optionally substituted with Y, Ci-6 haloalkyl (preferably CF3 or CH2CF3), optionally substituted 5- or 6-membered aryl, heteroaryl (preferably phenyl), alkylaryl (preferably benzyl) or alkylheteroaryl; and R4 is Ci-6 alkyl (preferably methyl) or
X comprises hydrogen or a polymerisable group; and each occurrence of Y independently comprises hydrogen or a polymerisable group; provided that at least one occurrence of X or Y must be a polymerisable group.
Preferably, V is -(CH2)- W-(CH2)-, wherein W is 1-, 3-, 5- substituted phenyl.
The preferred option for group B is option (a) as set out above.
Preferably, the polymerisable group is a group that can be polymerised by free radical chemistry, for example an acrylate, methacrylate, acrylamide, methacrylamide, or a vinyl group. Alternatively, the polymerisable group may be a group that is polymerisable by ring-opening metathesis polymerisation, for example a norbornene or other strained cyclic alkene derivative.
In a preferred embodiment, the polymerisable group comprises a vinyl moiety, such that where X comprises a polymerisable group and n is 0, the functional monomer comprises a styrene group and where n is 1 the functional monomer comprises an alpha-methyl styrene group.
Preferably, X is para to -A-B.
In a preferred embodiment, X is hydrogen, R1 is hydrogen, n is 1, B is -SC(NHR2)(NHR3) wherein R2 is CH2CHCH2 and R3 is benzyl.
Preferably, the functional monomer comprises one of the following structures:
(VII) wherein Ma+ is a chelated metal ion.
Preferably, for formula (III) and (VII) M aa+ i •s Z ΓT-n 2z++ and for formulae (IV) and (V) M *-a+ is Ga 3+
Even more preferably, the functional monomer has the structure:
In a preferred embodiment, the crosslinker is water-soluble. Preferably, the crosslinker comprises an acrylate, a methacrylate, an acrylamide, a methacrylamide, a styrene, a polyethylene glycol (PEG) derivative bearing a polymerisable group at each end of the PEG chain or a 1,2-disubstituted alkene such as a maleimide or a vinyl ether. In a preferred embodiment, the cross linker is ethylene glycol dimethacrylate, a polyethylene glycol derivative of formula:
wherein Z is a polymerisable group
Preferably, the polyethylene glycol derivative is a poly(ethylene glycol) diacrylate, a poly(ethylene glycol) dimethacrylate, a poly(ethylene glycol) diacrylamide or a poly(ethylene glycol) dimethacrylamide. In another preferred embodiment, Z is a norbenene.
In a preferred embodiment, the functional monomer is custom-made.
In a preferred embodiment, the polymer is formed from a functional monomer and at least one co-monomer. The co-monomer may be a second functional monomer as defined above or it may be any other monomer comprising a polymerisable group.
hi certain preferred embodiment, the polymer comprises two or more types of functional monomer, each being capable of interacting with a different pre-
determined functional group. Thus, the polymer is capable of selectively recognising a molecule which possesses one or more of a pre-determined selection of two or more functional groups.
In a further preferred embodiment, the polymer comprises two or more types of crosslinker as defined above.
In a second aspect, the present invention provides a method of preparing an imprinted polymer which is capable of selectively recognising and binding to a molecule which possesses a pre-determined functional group, wherein the imprinted polymer is capable of selectively recognising and binding to a plurality of molecules which possess the pre-determined functional group, wherein the plurality of molecules may differ in at least one of molecular type, structure, substructure or size, the method comprising the steps of: a) mixing a template molecule possessing the pre-determined functional group and a functional monomer comprising a chemical moiety capable of interacting with the pre-determined functional group and allowing complex formation; b) providing a porogenic solvent and a cross-linker to the mixture provided in step a) and allowing polymerisation to occur; and c) removing the template molecule thereby providing the imprinted polymer.
In step a), the template molecule undergoes a self-assembly process with one or more functional monomers that will constitute a functional group's binding site within the imprinted polymer. This self-assembly process leads to the formation of a template- binding site complex in which the functional monomers are spatially oriented for optimal binding to the pre-determined functional group. Minimisation of influence of substrate size, type etc on binding in the produced imprinted polymer is attributable to the choice of template molecule, functional monomer(s) and polymer backbone.
Advantageously, in an imprinted polymer of the invention or produced according to the method of the invention, the template molecule allows pre-organisation of binding
sites (i.e. functional monomers) in solution in such a way that the structural and electronic features of the pre-determined functional group to be imprinted are recognised and during polymerisation become inscribed into the polymer matrix. The polymer backbone allows the self-assembly and imprinting process to take place without adverse effect and the polymer backbone is compatible with the solvent or solvent mixtures to be used in the process of applying the FIP to an application.
The polymer network forms in such a way that it produces pores and pathways which are large enough to ensure accessibility for all compounds that need to be recognised by the FIP. In particular, the functional monomer, crosslinker and porogenic solvent are selected to ensure formation of a macroporous network. This is achieved by tailoring the solubility of the functional monomer and crosslinker in the porogenic solvent as well as by tailoring the structure of the template molecule. With regard to stability, in order to achieve a macroporous structure the functional monomer, the crosslinker and any co-monomers or co-crosslinkers used should be soluble in the porogenic solvent. As the polymerization reaction proceeds, this solubility will be lost and polymer will begin to precipitate. Continued polymerization around this precipitated polymer leads to the formation of a porous polymer network. The choice of specific functional monomers, crosslinkers, co-monomers and co-crosslinkers can be used to alter the point at which the initial precipitation occurs and consequently tailor the pore size in the polymer network.
Preferably, a polymerisation initiator is provided in step b). The polymerization initiator may be a thermal or photochemical initiator or a redox initiator. Azo initiators and peroxides are preferred for thermal and photochemical polymerisation and are chosen to be soluble in the pre-polymerisation mixture with a half-life consistent with the preferred polymerisation temperature (00C to 65°C typically). Preferably, the initiator is azobisisobutyronitrile. Redox initiators are those commonly used to produce hydrogels and are typically persulfates, in combination with amines or Fe salts. Ring-opening metathesis polymerisation (ROMP) initiation requires specific ROMP catalysts, such as Grubbs and Hoveyda-type catalysts and other functional group tolerant metathesis catalysts known in the literature.
Preferably, the pre-determined functional group is a phosphate group or a phosphate monoester group.
Preferably, the template molecule is phosphate-containing compound of formula (VI):
O Il R-O-P-O Z+
OH (Vl) wherein R5 is a C1-20 linear, branched, cyclic or polycyclic hydrocarbon, a dendron or dendrimer, a linear or branched polymer chain, a cyclodextrin or a polypeptide chain; and wherein Z+ is a positively charged counter ion, for example an ammonium ion of formula N(R6)4"1", wherein each occurrence of R6 is independently a Ci-20 linear, branched or cyclic hydrocarbon or bulky group selected from a dendron or dendrimer, a linear or branched polymer chain, a cyclodextrin and a polypeptide chain.
Advantageously, modification of the template molecule with a large sacrificial substituent causes the template molecule itself to act as a pore forming agent. Use of space generating substituents such as dendrons and dendrimers, linear and branched polymer chains, cyclodextrins, phosphorylated proteins even is made on the basis of the maximum size of analyte for which functional group selectivity is required.
Preferably, such modified templates can be removed/degraded to facilitate their complete extraction from the FIP in step c) and maximise the loading level of the FIP at the same time.
In a preferred embodiment, R5 is adamantyl.
In a preferred embodiment, the functional monomer is a functional monomer as defined above in respect of the first aspect of the invention.
In a preferred embodiment, the porogenic solvent is CHCl3, water, a water/solvent mixture wherein the solvent is an alcohol, DMF, DMSO, N-Methyl-2-pyrrolidone
(NMP), a water-miscible ionic liquid or acetonitrile or an aqueous buffer, for example TRIS, HEPES, with a background electrolytes (typically NaCl).
The choice of porogenic solvent (porogen) is related to the choice of crosslinker, functional monomer, template molecule, co-monomer and co-crosslinker. The porogen provides solubility for all monomers, crosslinkers and the template molecule and is also compatible with the polymerisation mechanism. During polymerisation, the porogen generates phase separation so that large droplets of porogen are entrapped in a precipitating polymer matrix. These droplets are freed once the network is set and the porous structure has become permanent through cross-linking.
Preferably, the cross-linker is an acrylate, a methacrylate, an acrylamide, a methacrylamide, a styrene, or a 1,2-disubstituted alkenes such as a maleimide or a vinyl ether. In a preferred embodiment, the cross linker is ethylene glycol dimethacrylate.
Preferably, the template molecule is removed in step c) by TFA extraction.
In a third aspect, the present invention provides an imprinted polymer as produced by the second aspect of the invention.
In a fourth aspect, the present invention provides a method of separating molecules which possess a pre-determined functional group from molecules which do not possess the pre-determined functional group, wherein the method comprises exposing a mixture of molecules to an imprinted polymer according to the first aspect of the invention or as produced by a method according to the second aspect of the invention.
In a preferred embodiment, the method is a chromatographic method and the imprinted polymer is used as a chromatographic stationary phase or in a solid-phase extraction mode.
In a preferred embodiment, all the molecules within the mixture of molecules are soluble in the same solvent, which is preferably an aqueous solvent of high polarity. In a preferred embodiment, the method is a method of separating out phosphoproteins, phospholipids and/or phosphorylated metabolites from a biological sample.
Useful solvents during the analysis are those which dissolve the analyte and at the same time minimise non-specific interactions between the analyte and the polymer matrix.
In a fifth aspect, the present invention provides a functional monomer for use in production of an imprinted polymer, the functional monomer having the structure:
(I) wherein A comprises (CH2)n, wherein n is 0 or 1 ;
B comprises (a) a thiouronium group of formula -SC(NHR2)(NHR3), bis(pyridine-2- ylmethyl)amino optionally substituted on one or both pyridine rings with Y, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-N,N'-diacetic acid), l,2-bis(o-aminophenoxy)ethane-Ar,N,N'rA^'-tetraacetic acid, or an analogue, derivative or ring-fused variant thereof; (b) a ditopic thiouronium group of formula:
(c) V disubstituted with (bis(pyridine-2-ylmethyl)amino), wherein V is an alkyl, haloalkyl, aryl, heteroaryl, naphthalene, anthracene or -(CH2)-W-(CH2)- group, ^herein W is aryl, heteroaryl, naphthalene or anthracene;
Ri comprises hydrogen or OR4;
R2 and R3 are each independently selected from hydrogen, Ci-6 alkyl (preferably methyl) optionally substituted with Y, Ci-6 haloalkyl (preferably CF3 or CH2CF3), optionally substituted 5- or 6-membered aryl or heteroaryl (preferably phenyl); and R4 is Ci-6 alkyl (preferably methyl) or
X comprises hydrogen or a polymerisable group; and each occurrence of Y independently comprises hydrogen or a polymerisable group; provided that at least one occurrence of X or Y must be a polymerisable group. Preferably, V is -(CH2)- W-(CH2)-, wherein W is 1-, 3-, 5- substituted phenyl.
Preferably, the functional monomer is as defined above in respect of the first aspect of the invention.
All preferred features of each of the aspects of the invention apply to all other aspects mutatis mutandis.
The invention may be put into practice in various ways and a number of specific embodiments will be described by way of example to illustrate the invention with reference to the accompanying drawings in which:
Figure 1 shows a schematic representation of a conventional molecular imprinting protocol. The individual steps shown are i) complexation in solution; ii) polymerisation; and iii) removal of template molecule.
Figure 2 shows a schematic representation of selective binding as exhibited by conventional imprinted polymers. As illustrated, the aim of conventional molecular imprinting is to achieve selectivity for one molecule over all other molecules.
Figure 3 shows a schematic representation of a general protocol for production of a functional group imprinted polymer according to the present invention. The individual steps shown are i) complexation in solution; ii) polymerisation; and iii) removal of template molecule.
Figure 4 shows a schematic representation of functional group selectivity as exhibited by a functional group imprinted polymer (FIP) of the present invention. As illustrated, an FIP of the invention is designed to recognise molecules bearing a particular predetermined functional group whilst allowing variation in the remainder of the structure, with selectivity over molecules with deviating functional group compositions.
Figure 5 shows a schematic representation of a protocol for production of a functional group imprinted polymer according to the present invention.
Figure 6 shows an experimental design used for comparing selectivity of an imprinted polymer of the invention with that of a conventional MIP.
Figure 7 shows the concentration of (a) TBA 1-naphthyl phosphate and (b) TBA 1- naphthyl acetate in non-imprinted polymer (NIP) and FIP equilibrations (i.e. in NIP and FEP supernatant at time = 0 and at equilibrium).
Figure 8 shows the analyte concentration of TBA 1-naphthyl phosphate and TBA 1- naphthyl acetate in (a) NIP and (b) FIP equilibrations.
Figures 9 and 10 show the relative values of a phosphorylated substrate and a carboxylated substrate removed during the wash step and extraction step of equilibration experiments.
The meanings of terms used herein are explained below, and the present invention is illustrated in detail by reference to the following non-limiting examples.
The term "alkyl" as used herein means a straight or branched alkyl group, preferably having 1 to 20 carbon atoms, more preferably having 1 to 6 carbons, most preferably being methyl.
The term "haloalkyl" as used herein means an alkyl group as substituted by one or more halogen (e.g. Cl1, Br, F, I). Preferably, a haloalkyl is a C1-6 haloalkyl, for example -CF3 or -CH2CF3.
The term "5-6 membered aryl" as used herein means a monocyclic aryl group constituted by 5 or 6. Preferably, an aryl group is phenyl.
The term "5-6 membered heteroaryl" used herein means a monocyclic heteroaryl group, in which the number of atoms forming the ring is 5 or 6 and wherein one or more ring atom is a heteroatom selected from the group consisting of a nitrogen atom, a sulphur atom and an oxygen atom, for example pyridine.
An aryl or heteroaryl as defined above is optionally substituted with one or more substituents preferably selected from methyl, -CF3, -CN or NO2, halogen (eg. F, Cl, Br or I) or -NHZ.
The term 'functional group' is used herein in accordance with its standard meaning in the art, i.e. a group of atoms within a molecule responsible for a chemical characteristic of that molecule. Where a molecule is referred to as comprising a functional group, it should be appreciated that the functional group does not necessarily stand alone, but may form part of a larger molecule. In the context of the present application, a 'functional group' may also be referred to as a 'chemical moiety'.
The term 'polymerisable group' refers to any functional group which is capable of reacting with another polymerisable group in a polymerisation reaction. A polymerisable group may be one that is capable of undergoing free radical polymerization, for example an acrylate, methacrylate, acrylamide, methacrylamide or a vinyl group. Alternatively, the polymerisable group may be a one that is capable of undergoing ring-opening metathesis polymerisation, for example a norbornene or other strained cyclic alkene derivative.
The term 'crosslinker' refers to any molecule that is capable of linking polymer chains. Linkage can be by covalent or non-covalent (e.g. ionic) bonding.
Examples
A FIP selective for monophosphate esters
An FIP imprinted with the 1-adamantyl phosphate ester template molecule was synthesized and binding selectivity experiments were conducted by pitting 1- napththylmonophosphate ester against 1-naphthyl acetate
Reagents and Apparatus
Reagents: The following reagents were purchased from Aldrich Chemical Company, and used without further purification: anhydrous chloroform (>99% contains amylenes as stabilizer); ethylene glycol dimethacrylate (98%); 1-naphthyl phosphate (99%); tetrabutylammonium hydroxide-30-hydrate (98%); 4-vinylbenzyl chloride (90%).
2,2'-Azo-bis-isobutyronitrile (>98%) was purchased from Aldrich Chemical Company and recrystallised from hexanes prior to use.
Tetrabutylammonium hydroxide 40 wt.% solution in water was purchased from Acros and used without further purification. l-Methyl-3-phenyl-2 -thiourea was purchased from Fluorochem and used without further purification.
2-Acetonaphthone (>99%) was purchased from Fluka and used without further purification.
1 -naphthalene acetic acid (97%) was purchased from Sigma and used without further purification. Acetic acid (99.5%) was purchased from BDH Laboratory Supplies, and used without further purification.
Trifluoroacetic acid (98%) was purchased from Apollo Scientific Ltd, and used without further purification.
Solvents: All solvents (apart from those listed in the preceding paragraph) were obtained from BDH Laboratory Supplies and, unless otherwise specified, used without further purification. All water was purified by simple distillation prior to use.
HPLC: HPLC was performed using a Laserchrom system equipped with degasser, tertiary gradient pump, variable wavelength UV detector, Phenomenex Synergi 4U max C1212 column and column heater (set to 35° C). Data was processed using Data Apex Clarity Lite software. For all experiments, volume of the injection loop was 20 μL and the flow rate set to 1 mL/ min. HPLC-grade solvents were used.
For all experiments, a gradient method was used according to the following:
NMR: All 1H NMR spectra were recorded on either a Bruker DRX 400 (400 MHz), Bruker Avance 500 (500 MHz) or a Jeol GSX (270 MHz) spectrometer. The reference peaks used were: CHCl3 (8H = 7.26 ppm); H2O (6H = 4.79 ppm); DMSO (6H = 2.50 ppm) unless otherwise indicated. All 13C NMR spectra were recorded on either a Bruker DRX 400 (400 MHz) or a Bruker Avance 500 (500 MHz)spectrometer. The reference peaks used were: CHCl3 (δc = 77.16 ppm); DMSO
(δc = 39.52 ppm). All 31P NMR spectra were recorded on either a Bruker DRX 400 (400 MHz) or a Bruker Avance 500 (500 MHz)spectrometer.
All NMR spectra were processed using MestRe-C software (version 4.8.1.1). Deuterated chloroform (99.8%) and deuterated dimethyl sulphoxide (99.8%) were purchased from Merck, and deuterium oxide (>99.9%) was purchased from Aldrich Chemical Company; all were used as received.
IR: Infrared spectra were recorded on a Satellite FTIR spectrometer (manufactured by Mattson) using KBr plates.
MS: High resolution mass spectra were recorded by the Imperial College mass spectrometry service using chemical ionisation with ammonium ions (NH4 +) or using electrospray mass spectrometry.
TLC: Pre-coated silica gel F254/366 Merck Kieselgel 60 A plates were used, and visualised using UV light (254 nm).
Melting Points: Melting points were determined using a Sanyo Gallenkamp hot stage apparatus, and are uncorrected.
Elemental Analysis: Elemental analysis was carried out by Stephen Boyer at the London Metropolitan University
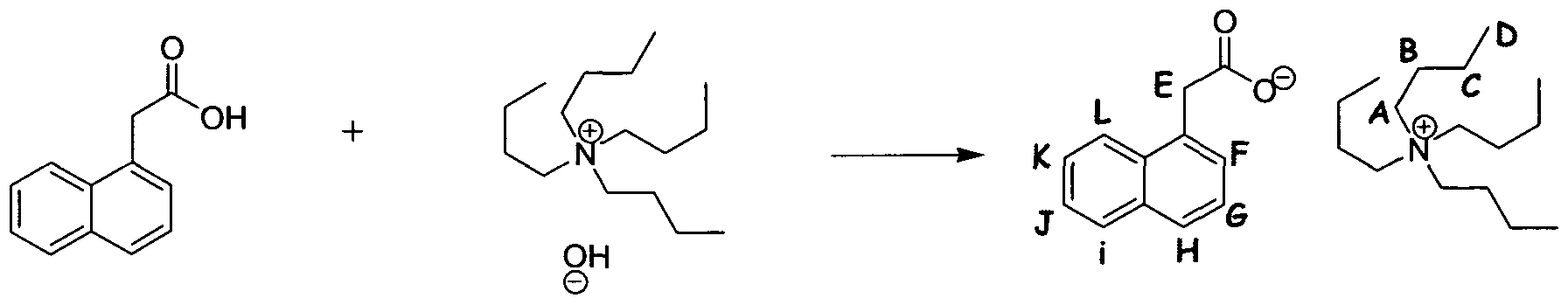
Preparation of tetrabutylammonium (TBA) 1-adamantyl phosphate
1-Adamantyl phosphoric acid was produced according to the teaching of Bδhringer, H.; Vogt, H., Arch. Pharm. (Weinheim). 1977, 310, (11), 894-905.
1-adamantanyl phosphoric acid (2.30 mmol, 533 mg) was taken up in a mixture of distilled water (60 mL) and MeOH (20 mL), and tetrabutylammonium hydroxide-30- hydrate (1.0 eq, 2.295 mmol, 1.836 g) added. This was stirred at it for 1 h, then concentrated in vacuo and lyophilised to afford a cream-coloured solid. This was recrystallised from EtOAc/hexanes and further lyophilised to afford an off-white solid
(561 mg, 52 %). The filtrate was left to stand at rt for several days, after which time a second crop precipitated (316 mg, 29%).
1H NMR (DMSO-d6, 400 MHz) δH = 0.93 [t, 3JHH = 7.3 Hz, 12H, (D)], 1.31 [sex, 3JHH = 7.4 Hz, 8H, (C)], 1.51-1.63 [m, 14H, (G, B)], 1.88 [m, 6H, (E)], 2.00 [s, 3H, (F)], 3.16 [m, 8H, (A)]; 13C NMR (DMSOd6, 400 MHz) δc = 13.43 (D), 19.14 (C), 22.97 (B), 30.21 (F), 36.14 (G), 43.50 [3JCp = 4.21 Hz] (E), 57.41 (A); 31P NMR
(DMSOd6, 400 MHz) δP = -3.52; 31P NMR (CDCl3, 500 MHz) δP = -2.29; Elemental analysis: Calculated (Ci6H36N-CiOH16O4P) C 65.93 %, H 11.67 %, N 2.96 %; Found C 64.21 %, H 10.85 %, N 2.83 %
Synthesis of (4-VJnVIbCnZyI)-I -methyl-3-phenylisothiouronium chloride
To a stirring solution of l-methyl-3-phenyl-2-thiourea (3.01 mmol, 500 mg) in anhydrous MeOH (5 mL) was added 4-vinylbenzyl chloride (1.2 eq, 3.61 mmol, 509 μL) in MeOH (5 mL). The resulting orange solution heated at reflux in inert conditions for 2 h. The red/pink reaction mixture was concentrated in vacuo {water bath >40°C} to a dark purple oil. This was triturated with diethyl ether to afford an off-white solid, which was isolated via vacuum filtration, washed with diethyl ether (200 mL), and dried under vacuum {40°C, 9 h} (828 mg, 87 %).
1H NMR (CDCl3, 400 MHz): δH = 3.14 [d, 3JHH = 4.4 Hz, 3H, CH3, (HF.)], 3.41 [d, 3JHH = 3.5 Hz, 3H, CH3 (HF)], 4.16 [s, 2H, CH2 (HE.)], 4.60 [s, 2H, CH2 (HE)], 5.27-
5.29 [d, 3JHH = 10.9 Hz, CH, (H11-)], 5.30-5.33 [d, 3JHH = 10.9 Hz, CH, (H8)], 5.75-
5.79 [d, 3JHH = 17.6 Hz, CH, (HA.)], 5.72-5.77 [d, 3JHH = 17.6 Hz, CH, (HA)], 6.66-
6.73 [dd, 3JHH = 10.8, 17.6 Hz, CH, (Hc0], 6.64-6.70 [dd, 3JHH = 10.8, 17.6 Hz, CH,
(Hc)], 7.17-7.49 [m, 9H, aromatic (HD., HD) and (Hr, H1)], 10.18 [m, IH, (HG-, HG)], 11.40 [s, IH, (HH)], 12.19 [s, IH, (HH-)]; '3C NMR (CDCl3, 500 MHz): δc = 33.10
(Cv), 37.02 (Cx), 114.72 (C2), 126.60 (aromatic), 126.93 (aromatic), 128.42
(aromatic), 129.25 (aromatic), 129.57 (aromatic), 132.74 (aromatic), 135.41
(aromatic), 135.96 (aromatic), 137.64 (Cγ), 166.89 (Cw); IR (thin film): vmax =
2560 cm"1 (broad, =N+- stretch), 1510 cm"1 (aromatic); mp 135-136°C; Elemental analysis: Calculated (Ci7H19N2SCl) C 72.05%, H 6.76 %, N 9.88%; Found C 65.51%,
H 6.28%, N 8.99%; HR-MS (CI, NH4 +): Found 283.127; Calculated (CnH19N2S)
283.418; TLC (UV, silica gel, hexane/ EtOAc, 1:1): i?/0.30
Preparation of an Imprinted Polymer having specificity for Phosphate Monoesters ('Phospho-specific FIP'V
Inside a screw-top vial equipped with magnetic stirrer bar, tetrabutylammonium 1- adamantyl phosphate (50.2 mg, 0.106 mmol) and (4-vinylbenzyl)- l-methyl-3-
■ phenylisothiouronium chloride (2.0 eq, 0.211 mmol, 67.3 mg) were taken up in anhydrous CHCl3 (600 μL). The vial was temporarily sealed under an inert atmosphere using a rubber septum, and ethylene glycol dimethacrylate <EDMA> (30 eq to tetrabutylammonium 1-adamantyl phosphate, 3.18 mmol, 600 μL) and azobis- 'cobutyronitrile <AIBN> (2 mol% to all double bonds, 0.131 mmol, 21.5 mg) were
added. The mixture was stirred at 250C under an inert atmosphere for 15 min until all components were solubilised, and the system degassed using a cycle of vacuum- freeze- thaw x 3. Under a positive flow of nitrogen the rubber septum was replaced by a screw-cap lid, and the vial then heated at 60°C with stirring for 36 h. This afforded a shiny, pale yellow-coloured solid which was subsequently ground using a pestle and mortar into an off-white, free-flowing fine solid. This was washed by means of soxhlet extraction using 5% v/v acetic acid in CHCl3 (250 mL) over 89 h (oil bath: 90-100°C). The polymer was dried in vacuo and then stirred in 5% v/v TFA in CHCl3 (150 mL) at rt for 20 h. Following this, the polymer was once again dried in vacuo and washed by means of soxhlet extraction using 5% v/v acetic acid in CHCl3 (100 mL) over 23 h (oil bath: 90-100°C). Finally, the polymer was further washed by means of soxhlet extraction using CHCl3 (150 mL) over 67 h (oil bath: 90-100°C). The polymer was then dried in vacuo to afford a free- flowing, off-white solid (390 mg).
The protocol for preparing an imprinted polymer (FIP) of the invention is represented schematically in figure 5.
Preparation of corresponding non-imprinted, control polymer ('NIP') Inside a screw-top vial equipped with magnetic stirrer bar, (4-vinylbenzyl)-l-methyl- 3-phenylisothiouronium chloride (2.0 eq, 0.211 mmol, 67.3 mg) was taken up in anhydrous CHCl3 (600 μL). The vial was temporarily sealed under an inert atmosphere using a rubber septum, and ethylene glycol dimethacrylate <EDMA> (3.18 mmol, 600 μL) and azobis-isobutyronitrile <AIBN> (2 mol% to~all double bonds, 0.131 mmol, 21.5 mg) were added. The mixture was stirred at 25°C under an inert atmosphere for 15 min until all components were solubilised, and the system degassed using a cycle of vacuum-freeze-thaw x 3. Under a positive flow of nitrogen the rubber septum was replaced by a screw-cap lid, and the vial then heated at 60"C with stirring for 36 h. This afforded a shiny, pale yellow-coloured solid which was subsequently ground using a pestle and mortar into an off-white, free-flowing fine solid. This was washed by means of soxhlet extraction using 5% v/v acetic acid in ^HCl3 (250 mL) over 89 h (oil bath: 90-1000C). The polymer was dried in vacuo and
then stirred in 5% v/v TFA in CHCl3 (150 mL) at it for 20 h. Following this, the polymer was once again dried in vacuo and washed by means of soxhlet extraction using 5% v/v acetic acid in CHCl3 (100 mL) over 23 h (oil bath: 90- 100° C). Finally, the polymer was further washed by means of soxhlet extraction using CHCl3 (150 mL) over 67 h (oil bath: 90-100°C). The polymer was then dried in vacuo to afford a free-flowing, off-white solid (401 mg).
Batch equilibration experiment hi order to evaluate whether the aforementioned FIP would bind a phosphorylated substrate other than its template molecule (i.e. other than 1-adamantyl phosphate) and do so with selectivity over an isosteric substrate a batch equilibration experiment was carried out using competing isosteric substrates tetrabutylammonium 1-naphthyl phosphate ('P') and tetrabutylammonium 1-naphthyl acetate ('OAc') (carboxylate is an isostere of phosphate monoester).
Preparation of tetrabutylammonium 1-naphthyl phosphate
To 1-napthyl phosphate (1.05 g, 4.70 mmol) was added tetrabutylammonium hydroxide <40 wt% solution in water> (1.0 eq, 4.70 mmol, 3.05 mL) and MeOH (10 mL). The mixture was stirred at rt for 3 h, then concentrated in vacuo and subsequently lyophilised for 12 h, thus affording a rigid, pale yellow solid. This was ground in a pestle and mortar, yielding a free-flowing, pale yellow-coloured solid. 1.800 g, 82 %
1H NMR (DMSO-d6, 400 MHz): δH = 0.90-0.94 [t, 3JHH = 7.4 Hz, 12H, (HD)], 1.24-
1.34 [sex, 3JHH = 7.3 Hz, 8H, (Hc), 1.51-1.58 [m, 8H, (H8)], 3.13-3.17 [m, 8H, (HA)], 7.22-7.31 [t, 3JHH = 7.9 Hz, IH, (aromatic)], 7.35-7.456 [b, 3H, (aromatic), 7.54- 7.56 [d, IH, (aromatic)], 7.78-7.806 (d, 3JHH = 7.9 Hz, IH, (aromatic), 8.20-8.22 [d, * H, 3JHH = 8.2 Hz, (aromatic)].
Preparation of tetrabutylammonium 1-naphthyl acetate
To naphthalene acetic acid (26.9 mmol, 5.00 g) was added tetrabutylammonium hydroxide <40 wt% solution in water> (1.0 eq, 26.85 mmol, 17.43 mL) and MeOH (10 mL). The mixture was stirred at rt for 3 h, and then concentrated in vacuo to afford a pale yellow-coloured oil. This was lyophilised for 12 h, and then heated at 60°C under vacuum for 18 h. This afforded an off-white crystalline material (8.74 g, 76 %)
1H NMR (DMSO-de, 400 MHz): δH = 0.91-0.94 [t, 3JHH = 7.3 Hz, 12H, (Hn)], 1.24- 1.34 [sex, 3JHH = 7.3 Hz, 8H, (Hc), 1.51-1.58 [m, 8H, (H,,)], 3.12-3.17 [m, 8H, (HA)], 3.56 [s, 2H, (HE)], 7.22-7.23 [d, 3JHH = 6.8 Hz, IH, (aromatic)], 7.33-7.36 [t, 3JHH = 8.1 Hz, IH, (aromatic), 7.40-7.45 [m, 2H, (aromatic)], 7.64-7.66 (d, 3JHH = 8.2 Hz, IH, (aromatic), 7.81-7.84 [m, IH, (aromatic)], 8.18-8.20 [m, IH, (aromatic).
Equilibration Experiment Into separate screw-top vials was placed the FIP (387 mg, estimated to contain 110 μmol binding sites) and the NIP (387 mg), and to each vial was added CHCl3 (5 mL) and 79.2 μL of a 63.1 mM stock solution of tetrabutylammonium 1-naphthyl phosphate and tetrabutylammonium 1-naphthyl acetate (~5 μmol of each species). The vials were screwed closed, and then gently agitated using a laboratory shaker at rt. At t=324 h, the supernatant was analysed by HPLC (using 2-acetonaphthone as an internal standard), and the FIP and NTP were then removed via vacuum filtration.
The concentration of each substrate present at equilibrium is shown in Tables 1 and 2 and Figures 7 and 8.
Table 1 - Concentrations of TBA 1-naphthyl phosphate and TBA 1-naphthyl acetate for NIP equilibration:
Table 2 - Concentrations of TBA 1-naphthyl phosphate and TBA 1-naphthyl acetate for NIP equilibration:
The FIP and NIP were then washed by means of soxhlet extraction using 5% v/v acetic acid in CHCl3 (250 mL) over 24 h (oil bath: 90-100°C), and the resulting solution analysed by HPLC.
The relative value of each substrate removed during the wash step is shown in Figure 9.
The bound substrate was then released by stirring the FIP and NIP in 5% v/v TFA in CHCl3 (150 mL) for 27 h at rt. Afterwards the FIP and NIP particles were isolated via vacuum filtration, and washed through with CHCl3 (50 mL). The filtrate was then concentrated in vacuo and dried in a vacuum oven (35-45°C, 36 h), and then analysed by HPLC.
The relative value of each substrate removed during the substrate extraction step is shown in Figure 10.
Conclusion The above results demonstrate (Tables 1 and 2 and Figures 7-10) that both the FIP and the NIP retain a phosphorylated species other than that which was used as template molecule. However, importantly the FIP shows clear selectivity towards the molecule carrying the phosphate ester group, which only differs, in comparison to the competing molecule, in this functional group (phosphate against carboxylate), not the size, shape or class.
Preparation of alternative FIPs
The protocol described above for preparation of an imprinted polymer is also used to prepare phospho-specific imprinted polymers using alternative functional monomers. The synthesis of certain functional monomers that are used in the preparation of phospho-specific imprinted polymers is set out in schemes 1 to 4 below:
Scheme 1 : Preparation of (4-formyl-2-methoxy-phenylazanediylVbis-acetate
Scheme 3 - preparation of l-(2-aminophenoxy)-2-(2-aminophenoxy-5-vinyl')ethane-
N, N, N', N'-tetraacetate
Synthetic route starting from the commercially available tetramethyl-5-formyl-5'- methyl-bis-(2-aminophenoxymethylene)-7V,N,iVy/V-tetraacetate):
Saponification G
Scheme 4 - Preparation of [3-[(Bis-pyridin-2-ylmethyl-amino)-methyll-5-(4- vinvI-benzyloxy)-benzyll-bis-pyridiii-2-ylmethyl-ainine
It should be understood that various changes and modifications can be made to the embodiments described herein without departing from the spirit and scope of the present invention
Claims
1. An imprinted polymer comprising a porous polymeric network formed from a functional monomer comprising a chemical moiety capable of interacting with a pre- determined functional group and a crosslinker, wherein the imprinted polymer is capable of selectively recognising and binding to a plurality of molecules which possess the pre-determined functional group, wherein apart from possessing the predetermined functional group, the plurality of molecules may differ in at least one of molecular class, structure, substructure or size.
2. A polymer according to claim 1, wherein the porous polymeric network formed from the functional monomer, linked by a crosslinker is covalent, non- covalent or a hybrid covalent/non-covalent network.
3. A polymer according to claim 1 or 2, wherein the porous polymeric network is a macroporous polymeric network.
4. A polymer according to any preceding claim, wherein the pre-determined functional group is selected from the group including, but not limited to: acetal; acid chloride; aldehyde; alkene; alkyne; amide; amidine; amine; ammonium; azide; azo; carbonate; carboxylic acid; carboxylate; ester; ether; guanidine; guanidinium; hydroxyl; isocyanate; isothiocyanate; imidazole; imide; imine; ketal; ketone; nitrate; nitrile; nitro; nitroso; phosphate; phosphate ester; peroxy; phosphine; phosphonate; phosphono; phosphonium; pyridyl; sulphate; sulfonate; sulfone; sulfonic acid; thiol; thiourea; thiourethane; thiouronium; urea; urethane; and uranium.
5. A polymer according to any preceding claim, wherein the pre-determined functional group is a phosphate or phosphate ester functional group.
6. A polymer according to any preceding claim, wherein the functional monomer comprises a polymerisable group and one or more chemical moieties capable of interacting with the pre-determined functional group, the one or more chemical moieties being selected from the following: acetal; acid chloride; aldehyde; alkene; alkyne; amide; amidine; amidinium; amine; polyamine; ammonium; azide; azo; carbonate; carboxylic acid; carboxylate; cyclodextrin; ester; ether; guanidine; guanidinium; hydroxyl; isocyanate; isothiocyanate; imidazole; imide; imine; ketal; ketone; nitrate; nitrile; nitro; nitroso; phosphate; phosphate ester; peroxy; phosphine; phosphonate; phosphono; phosphonium; pyridyl; polypyrrole; porphyrin; sulphate; sulfonate; sulfone; sulfonic acid; thiol; thiourea; thiourethane; thiouronium; urea; urethane; uronium.
7. A polymer according to claim 6, wherein the functional monomer is capable of binding strongly to a phosphate or phosphate monoester functional group and comprises one or more chemical moieties selected from amide, amidine, amidinium, carboxylic acid, polyamine, guanidinium, guanidine, polypyrrole, porphyrin, urea, urethane, thiourea, thiourethane and thiouronium as well as metal pyridine dipicolyl amines, metal cyclens (3, 4 and 5), metal coordination enes and metal guanidine chelates.
8. A polymer according to claim 7, wherein the functional monomer comprises a thiouronium group, bis(pyridine-2-ylmethyl)amino, (diacetic acid)amino, l,2-bis(o- aminophenoxy)ethane -N,N,N',N-tetraacetic acid.. Preferably, (diacetic acid)amino is l-(o-aminophenoxy)methane-N,N -diacetic acid. Preferably, the thiouronium group has the formula -SC(NHR2)(NHR3), wherein R2 and R3 are each independently selected from hydrogen, Ci-6 alkyl or 5- or 6-membered aryl or heteroaryl.
9. A polymer according to claim 8, wherein a chelated metal ion is present, wherein the functional monomer comprises a metal pyridine dipicolyl amine, a metal cyclen (3, 4 and 5), a metal coordination ene, metal guanidine chelate, bis(pyridine-2- ylmethyl)amino, a (diacetic acid)amino group or 1 ,2-bis(o-aminophenoxy)ethane - N,N,N',N'-tetraacetic acid and wherein the chelated metal ion is Al3+, Fe3+, Ca2+, Zr4+, Ni2+, Co2+, Cu2+, Mn2+/3+, Mg2+, Ca2+, Zn2+ or Ga3+.
10. A polymer according to any preceding claim, wherein the functional monomer comprises the following structure:
(I) wherein A comprises (CH2)n, wherein n is 0 or 1 ;
B comprises (a) a thiouronium group of formula -SC(NHR2)(NHR3), bis(pyridine-2- ylmethyl)amino optionally substituted on one or both pyridine rings with Y, a
(diacetic acid)amino group (for example l-(o-aminophenoxy)methane-Nr/V'-diacetic acid), l,2-bis(o-aminophenoxy)ethane-ΛWiV'^V-tetraacetic acid, or an analogue, derivative or ring- fused variant thereof;
(b) a ditopic thiouronium group of formula:
(c) V disubstituted with (bis(pyridine-2-ylmethyl)amino), wherein V is an alkyl, haloalkyl, aryl, heteroaryl, naphthalene, anthracene or -(CH2)-W-(CH2)- group, wherein W is aryl, heteroaryl, naphthalene or anthracene; Ri comprises hydrogen or OR4;
R2 and R3 are each independently selected from hydrogen, Ci-6 alkyl optionally substituted with Y, Ci-6 haloalkyl, optionally substituted 5- or 6-membered aryl or heteroaryl; and R4 is Ci-6 alkyl or
X comprises hydrogen or a polymerisable group; and each occurrence of Y independently comprises hydrogen or a polymerisable group; provided that at least one occurrence of X or Y must be a polymerisable group
11. A polymer according to any preceding claim, wherein the functional monomer comprises one of the following structures:
(H) (III) (IV)
(VII) wherein Ma is a chelated metal ion.
12. A polymer according to claim 11, wherein the functional monomer has the structure:
13. A polymer according to any preceding claim, wherein the cross linker comprises an acrylate, a methacrylate, an acrylamide, a methacrylamide, a styrene, a polyethylene glycol derivative bearing a polymerisable group at each end of the PEG chain or a 1 ,2-disubstituted alkene such as a maleimide or a vinyl ether.
14. A polymer according to any preceding claim, wherein the polymer is formed from a functional monomer and at least one co-monomer, wherein the co-monomer may be a second functional monomer as defined above or any other monomer comprising a polymerisable group.
15. A polymer according to any preceding claim, wherein the polymer comprises two or more types of crosslinker as defined above.
16. A method of preparing an imprinted polymer which is capable of selectively recognising and binding to a molecule which possesses a pre-determined functional group, wherein the imprinted polymer is capable of selectively recognising and binding to a plurality of molecules which possess the pre-determined functional group, wherein the plurality of molecules may differ in at least one of molecular type, class, substructure or size, the method comprising the steps of: a) mixing a template molecule possessing the pre-determined functional group and a functional monomer comprising a chemical moiety capable of interacting with the pre-determined functional group and allowing complex formation; b) providing a porogenic solvent and a cross-linker to the mixture provided in step a) and allowing polymerisation to occur; and c) removing the template molecule thereby providing the imprinted polymer.
17. A method according to claim 16, wherein a polymerisation initiator is provided in step b).
18. A method according to claim 16 or 17, wherein the pre-determined functional group is a phosphate group or a phosphate monoester group.
19. A method according to any of claims 16 to 18, wherein the template molecule is phosphate-containing compound of formula (VI):
O Il R-O-P-O Z+
OH (Vl) wherein R5 is a Ci-20 linear, branched, cyclic or polycyclic hydrocarbon, a dendron or dendrimer, a linear or branched polymer chain, a cyclodextrin or a polypeptide chain; and wherein Z+ is a positively charged counter ion, for example an ammonium ion of formula N(RO)4+, wherein each occurrence of R6 is independently a Ci-20 linear, branched or cyclic hydrocarbon or bulky group selected from a dendron or dendrimer, a linear or branched polymer chain, a cyclodextrin and a polypeptide chain.
20. A method according to any of claims 16 to 19, wherein the functional monomer is as defined in any of claims 7 to 12.
21. A method according to any of claims 16 to 20, wherein the porogenic solvent is CHCl3, water, a water/solvent mixture wherein the solvent is an alcohol, DMF, DMSO, N-Methyl-2-pyrrolidone (NMP), a water-miscible ionic liquid or acetonitrile or an aqueous buffer, for example TRIS, HEPES, with a background electrolytes (typically NaCl).
22. A method according to any of claims 16 to 21, wherein the cross-linker comprises an acrylate, a methacrylate, an acrylamide, a methacrylamide, a styrene, a polyethylene glycol derivative bearing a polymerisable group at each end of the PEG chain or a 1,2-disubstituted alkene such as a maleimide or a vinyl ether.
23. An imprinted polymer as produced by method of any of claims 16 to 22.
24. A method of separating molecules which possess a pre-determined functional group from molecules which do not possess the pre-determined functional group, wherein the method comprises exposing a mixture of molecules to an imprinted polymer according to any of claims 1 to 15 or 23.
25. A method according to claim 24, wherein the method is a chromatographic method and the imprinted polymer is used as a chromatographic stationary phase or in a solid-phase extraction mode.
26. A functional monomer for use in production of an imprinted polymer, the functional monomer having the structure:
(I) wherein A comprises (CH2)n, wherein n is 0 or 1 ;
B comprises (a) a thiouronium group of formula -SC(NHR2)(NHR3), bis(pyridine-2- ylmethyl)amino optionally substituted on one or both pyridine rings with Y, a (diacetic acid)amino group (for example l-(o-aminophenoxy)methane-Nr/V'-diacetic acid), l,2-bis(o-aminophenoxy)ethane-Nr/V,N',N'-tetraacetic acid, or an analogue, derivative or ring-fused variant thereof; (b) a ditopic thiouronium group of formula:
(c) V disubstituted with (bis(pyridine-2-ylmethyl)amino), wherein V is an alkyl, haloalkyl, aryl, heteroaryl, naphthalene, anthracene or -(CH2)-W-(CH2)- group, wherein W is aryl, heteroaryl, naphthalene or anthracene; Ri comprises hydrogen or OR4;
R2 and R3 are each independently selected from hydrogen, Ci-6 alkyl optionally substituted with Y, Ci-6 haloalkyl, optionally substituted 5- or 6-membered aryl or heteroaryl; and R4 is Ci-6 alkyl or
X comprises hydrogen or a polymerisable group; and each occurrence of Y independently comprises hydrogen or a polymerisable group; provided that at least one occurrence of X or Y must be a polymerisable group.
27. A functional monomer as defined in any of claims 7 to 12.
28. A polymer a method or a functional monomer substantially as described herein with reference to or as described in any of the accompanying figures or examples.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0709336A GB0709336D0 (en) | 2007-05-15 | 2007-05-15 | Functional group imprinted polymers |
| GB0709336.2 | 2007-05-15 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2008139204A2 true WO2008139204A2 (en) | 2008-11-20 |
| WO2008139204A3 WO2008139204A3 (en) | 2009-02-26 |
Family
ID=38219452
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB2008/001681 Ceased WO2008139204A2 (en) | 2007-05-15 | 2008-05-15 | Functional group imprinted polymers |
Country Status (2)
| Country | Link |
|---|---|
| GB (1) | GB0709336D0 (en) |
| WO (1) | WO2008139204A2 (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102250285A (en) * | 2010-05-19 | 2011-11-23 | 中国科学院大连化学物理研究所 | Semi-covalent molecularly imprinted polymer used for selectivity separating phenols, preparation method thereof, and application thereof |
| WO2012125117A1 (en) * | 2011-03-16 | 2012-09-20 | Biomotif Ab | Method and kit for analyzing samples |
| CN103193826A (en) * | 2013-04-15 | 2013-07-10 | 中国科学院化学研究所 | Nanocluster, as well as preparation method and application thereof |
| CN104069837A (en) * | 2014-06-27 | 2014-10-01 | 广东石油化工学院 | Adsorbing agent for removing trace benzaldehyde impurities in styrene |
| CN104587969A (en) * | 2015-02-03 | 2015-05-06 | 吉林大学 | Preparation method of carbon-base adsorbing material capable of selectively adsorbing copper ions |
| CN104892839A (en) * | 2015-06-23 | 2015-09-09 | 嘉兴学院 | Surface molecular imprinting polyion liquid of reduced graphene oxide for detecting bisphenol A and preparation method and application of surface molecular imprinting polyion liquid |
| CN105017482A (en) * | 2015-06-23 | 2015-11-04 | 嘉兴学院 | Surface molecularly imprinted poly(ionic liquid) for detecting 4-nonyl phenol and its preparation method and use |
| CN107652461A (en) * | 2017-11-13 | 2018-02-02 | 李辉 | A kind of technology of preparing with thing trace new material and the application altogether of composition metal Polyphenols |
| CN111036181A (en) * | 2019-12-26 | 2020-04-21 | 南京师范大学 | Molecularly imprinted silica gel polymer and preparation method and application thereof |
| CN114917881A (en) * | 2022-05-19 | 2022-08-19 | 中国烟草总公司郑州烟草研究院 | Heterocyclic amine molecularly imprinted composite material and preparation method and application thereof |
| CN115093525A (en) * | 2022-06-13 | 2022-09-23 | 哈尔滨工业大学 | Multifunctional monomer covalent organic framework molecularly imprinted polymer and preparation method and application thereof |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109021171B (en) * | 2018-07-26 | 2020-06-12 | 河南师范大学 | Aqueous preparation method of tylosin tartrate surface molecularly imprinted polymer and its application |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6849701B2 (en) * | 2001-03-02 | 2005-02-01 | E. I. Du Pont De Nemours And Company | Droplet polymerization method for synthesis of molecularly imprinted polymers |
| US20040009872A1 (en) * | 2002-07-15 | 2004-01-15 | Gordon Cohen | Method using solvents for improved microporous polymeric adsorbents |
-
2007
- 2007-05-15 GB GB0709336A patent/GB0709336D0/en not_active Ceased
-
2008
- 2008-05-15 WO PCT/GB2008/001681 patent/WO2008139204A2/en not_active Ceased
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102250285A (en) * | 2010-05-19 | 2011-11-23 | 中国科学院大连化学物理研究所 | Semi-covalent molecularly imprinted polymer used for selectivity separating phenols, preparation method thereof, and application thereof |
| WO2012125117A1 (en) * | 2011-03-16 | 2012-09-20 | Biomotif Ab | Method and kit for analyzing samples |
| CN103193826A (en) * | 2013-04-15 | 2013-07-10 | 中国科学院化学研究所 | Nanocluster, as well as preparation method and application thereof |
| CN103193826B (en) * | 2013-04-15 | 2016-01-06 | 中国科学院化学研究所 | Nanocluster and preparation method thereof and application |
| CN104069837A (en) * | 2014-06-27 | 2014-10-01 | 广东石油化工学院 | Adsorbing agent for removing trace benzaldehyde impurities in styrene |
| CN104587969A (en) * | 2015-02-03 | 2015-05-06 | 吉林大学 | Preparation method of carbon-base adsorbing material capable of selectively adsorbing copper ions |
| CN105017482A (en) * | 2015-06-23 | 2015-11-04 | 嘉兴学院 | Surface molecularly imprinted poly(ionic liquid) for detecting 4-nonyl phenol and its preparation method and use |
| CN104892839A (en) * | 2015-06-23 | 2015-09-09 | 嘉兴学院 | Surface molecular imprinting polyion liquid of reduced graphene oxide for detecting bisphenol A and preparation method and application of surface molecular imprinting polyion liquid |
| CN107652461A (en) * | 2017-11-13 | 2018-02-02 | 李辉 | A kind of technology of preparing with thing trace new material and the application altogether of composition metal Polyphenols |
| CN107652461B (en) * | 2017-11-13 | 2020-12-11 | 苏州知瑞光电材料科技有限公司 | Preparation technology and application of a new composite metal polyphenols co-complex imprinted material |
| CN111036181A (en) * | 2019-12-26 | 2020-04-21 | 南京师范大学 | Molecularly imprinted silica gel polymer and preparation method and application thereof |
| CN111036181B (en) * | 2019-12-26 | 2022-02-22 | 南京师范大学 | Molecularly imprinted silica gel polymer and preparation method and application thereof |
| CN114917881A (en) * | 2022-05-19 | 2022-08-19 | 中国烟草总公司郑州烟草研究院 | Heterocyclic amine molecularly imprinted composite material and preparation method and application thereof |
| CN115093525A (en) * | 2022-06-13 | 2022-09-23 | 哈尔滨工业大学 | Multifunctional monomer covalent organic framework molecularly imprinted polymer and preparation method and application thereof |
| CN115093525B (en) * | 2022-06-13 | 2024-04-05 | 哈尔滨工业大学 | Multifunctional monomer covalent organic framework molecularly imprinted polymer and preparation method and application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2008139204A3 (en) | 2009-02-26 |
| GB0709336D0 (en) | 2007-06-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2008139204A2 (en) | Functional group imprinted polymers | |
| US5310648A (en) | Composition of matter comprising an imprinted matrix exhibiting selective binding interactions through chelated metals | |
| US7208557B2 (en) | Functional monomers for molecular recognition and catalysis | |
| US6379599B1 (en) | Process for the preparation of molecularly imprinted polymers useful for separation of enzymes | |
| DK2212020T3 (en) | EMBROIDERED POLYMERS WITH AFFINITY FOR PHOSHORIORED PEPTIDES AND PROTEINS | |
| EP2276567B1 (en) | Enantioselective zwitterionic ion-exchange material | |
| JP2002511924A (en) | Cation exchange stationary phase based on bifunctional crown ether for liquid chromatography | |
| Qader et al. | Peptide imprinted receptors for the determination of the small cell lung cancer associated biomarker progastrin releasing peptide | |
| CN108003287B (en) | Preparation method of protein affinity imprinted hydrogel polymer based on acrylamide metal chelating monomer | |
| Yang et al. | An artificial receptor synthesized by surface-confined imprinting for the recognition of acetylation on histone H4 K16 | |
| WO2016137953A1 (en) | Enrichment of lysophosphatidic acids with templated polymeric materials | |
| Rajkumar et al. | Development of fructosyl valine binding polymers by covalent imprinting | |
| US5541342A (en) | Separation of amino acids, amino-acid-based monomer, and process for the preparation thereof, as well as polymer material and process for the preparation thereof | |
| Araki et al. | Enantioselective polymer prepared by surface imprinting technique using a bifunctional molecule | |
| Trochimczuk et al. | Novel chelating resins with aminothiophosphonate ligands | |
| JP5249582B2 (en) | Imprint polymer and use thereof | |
| Galaev et al. | Metal-copolymer complexes of N-isopropylacrylamide for affinity precipitation of proteins | |
| US20200061579A1 (en) | Molecular Imprinted Polymers for Chemosensing | |
| Wu et al. | Comparison of monofunctional and multifunctional monomers in phosphate binding molecularly imprinted polymers | |
| CN109851717A (en) | A kind of preparation method and application of anionic PSSS/PSA microballoon | |
| EP0706520A1 (en) | Novel cross-linking agents and use thereof | |
| Sellergren | Enantiomer separations using designed imprinted chiral phases | |
| Spivak | Selectivity in molecularly imprinted matrices | |
| CN114555202A (en) | Materials and methods for separation based on halogen bonding | |
| Ramström | Synthesis and selection of functional and structural monomers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08750613 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 08750613 Country of ref document: EP Kind code of ref document: A2 |