WO2007141806A1 - Pharmaceutical formulations comprising oxcarbazepine and methods thereof - Google Patents
Pharmaceutical formulations comprising oxcarbazepine and methods thereof Download PDFInfo
- Publication number
- WO2007141806A1 WO2007141806A1 PCT/IN2007/000223 IN2007000223W WO2007141806A1 WO 2007141806 A1 WO2007141806 A1 WO 2007141806A1 IN 2007000223 W IN2007000223 W IN 2007000223W WO 2007141806 A1 WO2007141806 A1 WO 2007141806A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- dosage form
- oxcarbazepine
- solid
- solvent
- solid dosage
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/141—Intimate drug-carrier mixtures characterised by the carrier, e.g. ordered mixtures, adsorbates, solid solutions, eutectica, co-dried, co-solubilised, co-kneaded, co-milled, co-ground products, co-precipitates, co-evaporates, co-extrudates, co-melts; Drug nanoparticles with adsorbed surface modifiers
- A61K9/146—Intimate drug-carrier mixtures characterised by the carrier, e.g. ordered mixtures, adsorbates, solid solutions, eutectica, co-dried, co-solubilised, co-kneaded, co-milled, co-ground products, co-precipitates, co-evaporates, co-extrudates, co-melts; Drug nanoparticles with adsorbed surface modifiers with organic macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
Definitions
- Oxcarbazepine is the drug of choice for such kind of seizures as it has certain advantages over the other available anticonvulsants. With Oxcarbazepine there is a constant relationship between dosage and serum level control. It has linear kinetics and no dosage adjustment is necessary in the course of treatment with Oxcarbazepine. Less protein binding occurs with Oxcarbazepine thus increasing the efficacy of the drug and lowering the chance of interaction with other protein bound medications.
- a solid oral pharmaceutical formulation comprising solid dispersion or premix product of oxcarbazepine, wherein oxcarbazepine used in the formulation has a particle size of more than 15 microns, preferably 15 to 20 microns.
- step 8 Dry the above granules of step 8 into the same FBP at 60oC for 30 mins or till LOD reaches below 2 % w/w.
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
A solid dispersion or premix product comprising an effective amount of an active ingredients and an effective amount of one or more hydrophilic polymers wherein the solid dispersion product can either be a co-precipitate or a co-evaporate. The solid dispersion or premix product is used for the manufacture of a dosage form having improved bioavailability of said one or more active ingredients by oral administration to a patient in need thereof.
Description
PHARMACEUTICAL FORMULATIONS COMPRISING OXCARBAZEPINE
AND METHODS THEREOF
FIELD OF THE INVENTION The present invention relates to bioavailable and stable solid oral formulations of Oxcarbazepine.
The present invention in particular relates to solid dispersion or premix of oxcarbazepine, which yield an increased dissolution leading to the supersaturated concentration of the drug at the absorption site thereby effectively solving the bioavailability and additionally the degradation problems associated with Oxcarbazepine formulations.
BACKGROUND OF THE INVENTION Epilepsy is a disease, which is characterized by seizures and the medical treatment of epilepsy focuses not only on better control of seizures, but it is increasingly interested in diminishing the adverse effects of medication and optimizing quality of life.
Oxcarbazepine is an agent of first choice to prevent seizures in patients with epilepsy. It is thought that Oxcarbazepine works to stop or stabilize some of the electrical activity of the brain, keeping the nerve cells of the brain from becoming overexcited. Seizures occur when there isHoo much electrical activity in the brain.
Oxcarbazepine is indicated for use as monotherapy or adjunctive therapy in the treatment of partial seizures in adults and as monotherapy in the treatment of partial seizures in children aged 4 years and above with epilepsy, and as adjunctive therapy in children aged 2 years and above with epilepsy.
Oxcarbazepine is the drug of choice for such kind of seizures as it has certain advantages over the other available anticonvulsants. With Oxcarbazepine there is a constant relationship between dosage and serum level control.
It has linear kinetics and no dosage adjustment is necessary in the course of treatment with Oxcarbazepine. Less protein binding occurs with Oxcarbazepine thus increasing the efficacy of the drug and lowering the chance of interaction with other protein bound medications.
Routine monitoring of blood values and liver functions is not required, its absorption is hundred % and it can be taken with or without food, but the most common problem associated with it is its poor solubility and degradation. This poor solubility and degradation sometimes results in poor bioavailability.
The therapeutic effect of drug depends on the drug concentration at the site of action. The absorption of the drug into the systemic circulation is a prerequisite to reach the site of action for all drugs, except those drugs that are applied at the site of action, or those that are intravenously injected. After oral administration (gastro intestinal route) many factors determine the bioavailability. Since only dissolved drugs can pass the gastrointestinal membrane, dissolution is one of those factors. However drug metabolism in the intestinal lumen, the intestinal wall and the liver may also reduce its bioavailability. In general it can be stated that the rate of absorption and therefore the onset and extent of the clinical effects is determined by the dissolution of the drug and the subsequent transport over the intestinal membrane and passage of the liver.
As per the Biopharmaceutical Classification System there are four classes of drugs out of which class I drugs dissolve rapidly in an aqueous environment and are rapidly transported over the absorbing membrane and doesn't require any formulation strategy to increase the absorption. When the release of the active from the formulation is slower than the gastric emptying rate, good in-vitro-in-vivo-correlation can be expected. The absorption rate of Class-II drugs can be enhanced by accelerating the dissolution. Class II drugs show in-vitro-in-vivo-correlation as long as the in-vivo dissolution rate is same as in-vitro. However since the dissolution rate is critical for class-II drugs, the in-vivo absorption can be affected by several physiological fluctuations, like the volume and pH of the intestinal juices, the presence of bile salts, food, enzymes and bacteria, the motility of gut and the viscosity in the gut lumen. For
class-Ill drags the absorption is rate limiting and in-vitro dissolution experiments cannot be used to predict in-vivo absorption. Also for Class-IV drugs no in-vitro-in- vivo-correlation can be expected. It is up to the formulation scientist to increase the extent of absorption but also to improve the in-vitro-in-vivo correlation. This will reduce the patient-to-patient variability and improve the bioavailability and predictability of pharmacokinetic parameters.
Various approaches like solubility buffers, surfactants, and complex forming excipients like cyclodextrin; solvent depositions and micronisation have been used in the prior art to enhance the dissolution of a poorly soluble drug. One particular approach is to use a dosage form in which drug molecules are already dissolved in an aqueous solution, but this will require large amounts of water to dissolve the complete drug dose.
For a number of drugs reproducible and extensive absorption after oral administration can be established by using .surfactants. However their use is limited for example surfactants or most cyclodextrin derivatives can cause irritation in the gastro intestinal tract and are therefore highly undesirable.
US 20040197402 disclose the use of anionic, cationic and nonionic surfactants to enhance the dissolution of Oxcarbazepine. One major disadvantage associated with the use of surfactants is irritation of gastrointestinal lining leading to the patient's discomfort, which may also result in the unexpected behavior of formulation in vivo.
Another approach to deal with drugs that are poorly soluble is to increase their dissolution rate. Often the absorption of lipophilic drugs is decelerated by the slow rate of dissolution from the solid - drug particles. Dispersion of the drug as very fine particles will increase the surface area available for dissolution. According to the classical Noyes-Whitney equation this will increase the dissolution rate. Particle size reduction may go to the nano-scale. However even this size reduction will not lead to concentrations above the maximum solubility of the drug in the intestinal fluids.
US7037525 discloses a tablet formulation comprising a therapeutically effective amount of Oxcarbazepine in a finely grounded state with a median particle size of approximately 2 to 12 microns.
This sort of systems has processing difficulties and moreover size reductions don't ' lead to concentrations above the maximum solubility of the drug in the intestinal fluid.
The above prior art approaches reveal that various attempts like surfactants, and micronizations have been done in the prior art to increase the dissolution and enhance the stability of the poorly soluble and degradation prone drugs. Unfortunately all the above-mentioned prior art approaches are either very cumbersome or don't result in a dissolution enhanced as well as a stable formulation.
Alternatively solid dispersion or a premix of drug can be prepared in which drug remains mixed with the matrix material at a molecular level thus yielding a bioavailable and stable formulation, wherein the solid dispersion or premix product may be prepared by co-evaporation or co-precipitation.
SUMMARY OF THE INVENTION
The present invention addresses the solubility and bioavailability and also the degradation problems of the prior art and provides a simple composition of Oxcarbazepine, which are easier to prepare and yet have excellent dissolution and stability characteristics.
It has been surprisingly found that essentially preparing a solid dispersion or premix of drug which consists of at least two components, generally a hydrophilic matrix and a hydrophobic drug, which remain mixed at a molecular level, wherein the solid dispersion or the premix product can either be a co-precipitate or a co-evaporate, not only results in the enhanced dissolution of the drug but also in a stable formulation.
An object of the invention is to provide a bioavailable and stable formulation without having the drawback of formulations of the prior art.
One further objective of the invention is to provide a formulation, which is degradation resistant and hence color stable.
Yet another objective of the invention is to provide an immediate release formulation, which utilizes hydrophilic polymer and a non-solvent in such a manner that results in an enhanced dissolution and enhanced stability.
In accordance with another embodiment of the present invention, there is provided a solid oral pharmaceutical formulation comprising solid dispersion or premix product of oxcarbazepine, wherein oxcarbazepine used in the formulation has a particle size of more than 15 microns, preferably 15 to 20 microns.
The present invention is a simple solution to the solubility and stability issues associated with the Oxcarbazepine, resulting in a formulation which is simple to manufacture, cost effective, does not utilize costly techniques and yet gives excellent formulation characteristics in the form of improved solubility and bioavailability.
It is further demonstrated that the degradation issues related to Oxcarbazepine formulations are also surprisingly solved by the present compositions and moreover, since drug is entrapped in the lattice of the polymer at a molecular level therefore better protection is provided to the drug particle.
DETAILED DESCRIPTION OF THE INVENTION
Poor solubility and degradation associated with some drugs during the formulation is a major problem, which not only affects the processing characteristics and machinability of formulations but also affects the bioavailability and stability of the final formulation. When a formulation of a poorly soluble and degradation sensitive drug like Oxcarbazepine is to be manufactured, special precautions have to be taken as processing may also affect the dissolution behavior of drug.
It has been surprisingly found that essentially preparing a solid dispersion or premix of drug which consists of at least two components, generally a hydrophilic matrix and a hydrophobic drug which- remain mixed at a molecular level, wherein the solid dispersion or premix product can either be co-precipitate or a co-evaporate, not only results in the enhanced dissolution of the drug but it has also been further surprisingly observed that the degradation and discoloration problems associated with the formulation of Oxcarbazepine are easily solved by the compositions of the present invention.
The term, solid dispersion defines a system in a solid state (as opposed to a liquid or gaseous state) comprising at least two components, wherein one component is dispersed more or less evenly throughout the other component or components. When said dispersion of the components is such that the system is chemically and physically uniform or homogenous throughout or consists of one phase (as defined in thermodynamics), such a dispersion is called a "solid solution". Solid solutions are preferred physical systems because the components therein readily form liquid solutions when brought into contact with a liquid medium such as gastric juice. This increased propensity for dissolution may be attributed to at least in part to the fact that the energy required for dissolving the components from a solid solution is less than required for dissolving the components from a crystalline or microcrystalline solid phase.
The solid dispersion or the premix product of the invention is prepared" by a solvent method and may be either a co-precipitate or a co-evaporate
A co-precipitate is defined as a1 solid dispersion, which is prepared by the addition of a non-solvent to the solution of drug and matrix.
A non-solvent is defined as a carrier, which is not soluble in the solution of the drug and the polymer.
A co-evaporate is defined as a solid dispersion, prepared by evaporating solution of drug and polymer by vacuum drying, spray drying, spray granulating, freeze drying and spray-freeze drying or by any other similar methods.
The solvent method for preparing co-precipitate typically comprises of following steps: a) Preparing a solution of a hydrophilic polymer or a mixture of polymers in a solvent and then charging the drug in this reaction mass till a clear solution appears b) Adding a non-solvent to the solution mixture of step (a) c) Distilling off the solvents till a pasty mass is obtained d) Adding a solvent to this pasty mass while heating e) Filtering the slurry thus obtained and then drying it.
When the solid dispersion or the premix is of the co-evaporate type then drug and the polymer are dissolved in a common solvent and then the solvents are distilled off either by spray drying or by spray granulating.
The particles of the solid dispersion or premix product can be formulated into pharmaceutical dosage forms comprising a therapeutically effective amount of particles. Although mainly pharmaceutically dosage forms like tablets and capsules are envisaged, the particles of the present invention can also be given in the form of a sachet. Preferred dosage forms are those adapted for oral administration shaped as a tablet. They can be produced by conventional tabletting techniques with conventional ingredients or excipients and with conventional tabletting machines.
When the compositions are in the form of a tablet, it may include one or more tabletting lubricants such as magnesium stearate, stearic acid, palmitic acid, calcium stearate, talc, polyethylene glycol, carnauba wax and the likes and mixtures thereof.
Further the tablet may comprise pharmaceutically acceptable disintegrants for example crospovidone, sodium starch glycolate, croscarmellose and its salts, low substituted
hydroxypropyl cellulose, microcrystalline cellulose, modified starches, pregelatinised starch, calclim carboxymethyl cellulose, guar gum, corn starch, alginates and natural starch and the like or mixtures thereof. The weight percent of disintegrant in the solid dosage form is preferably in the range of 0.1% to 10% and more preferably in the range of2% to 8%.
Glidants used according to the present invention may suitable be one or more selected from the group comprising of silica, magnesium trisilicate, powdered cellulose, starch, talc; sodium stearyl fumarate, tribasic calcium phosphate or mixtures thereof. Preferably the glidant is talc or silica or mixtures of both. The weight percent of glidant in the solid dosage form is preferably in the range of 0.1% to 3% and more preferably in the range of 0.5% to 2.1%.
The hydrophilic polymers for use herein include but are not limited to HPMC. It is also contemplated that other polymers like poly vinyl pyrrolidone, hydroxy propyl cellulose, hydroxy ethyl cellulose, poly vinyl alcohol, sodium carboxy methyl cellulose, starches such as corn starch, modified corn starch, sugars, gum acacia and the like may be used.
Non-solvents for use herein include but are not limited to crospovidone. It is further contemplated that other polymers like croscarmellose sodium, sodium starch glycolate and pregelatinised starch and the like may be used.
Tablet formulated according to the invention allows not only for enhanced dissolution of the drug from the formulation but also results in a stable formulation.
The following examples illustrate the invention.
Example 1 Preparation of Oxcarbazepine Co-precipitate
Procedure: '
1. Charge Methylene chloride to the S.S reactor at 25-30° C.
2. Charge Methyl alcohol in S.S reactor of step (1) at 25-30° C.
3. Stir the solvent mixture for 10 mins at 25-30 ° C. 4. Heat the solvent mixture to 40-50 ° C.
5. Prepare HPMC solution by adding HPMC into a solvent mixture of step (4) at 40-500 C.
6. Charge oxcarbazepine to reaction mass of step (5) at 40-50 ° C.
7. Stir the reaction mass of step (6) till clear solution appears.
8. Charge crospovidone to reaction mass of step (7) at 40-50 ° C.
9. Stir the reaction mass of step (8) for 10-15 mins.
10. Distill off the solvents from reaction mass of step (9) under vacuum at 50 ° C till a pasty mass is obtained.
11. Add Isopropyl Alcohol to the pasty mass obtained from step (10) at 35-40 ° C.
12. Cool the reaction mixture of step (11) to 25-30 ° C.
13. Stir the reaction mass of step (12) for 30 mins at 25-30° C.
14. Filter the slurry mass of step (13) under vacuum at 25-30 ° C.
15. Dry the solid mass at 60 ° C. (LOD at 105 ° C for 2 hrs~NMT 1.5 %w/w)
16. Sift the dried solid mass through # 20 ASTM.
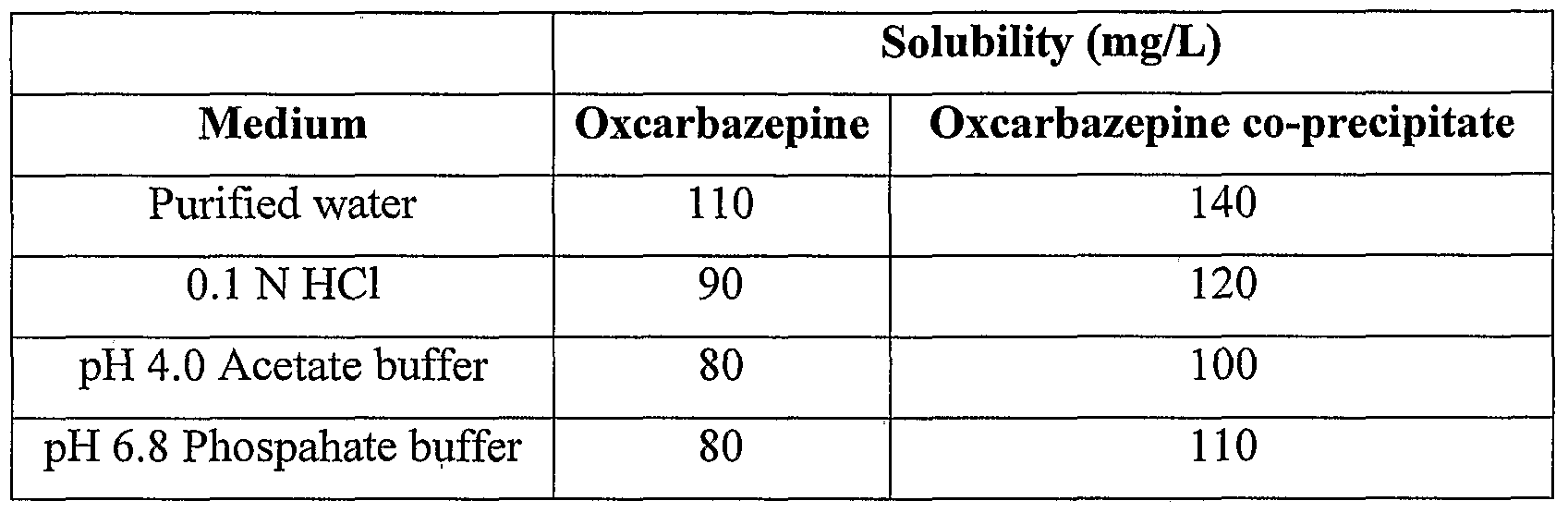
Solubility of Oxcarbazepine co-precipitate (Premix) and as such Oxcarbazepine API was carried out in different media. The solubility of Oxcarbazepine Co-precipitate was found to be more as compared to as such Oxcarbazepine API. The results of solubility study are appended in below table,
Table 1 Solubility data of Oxcarbazepine and Oxcarbazepine co-precipitate in different Media
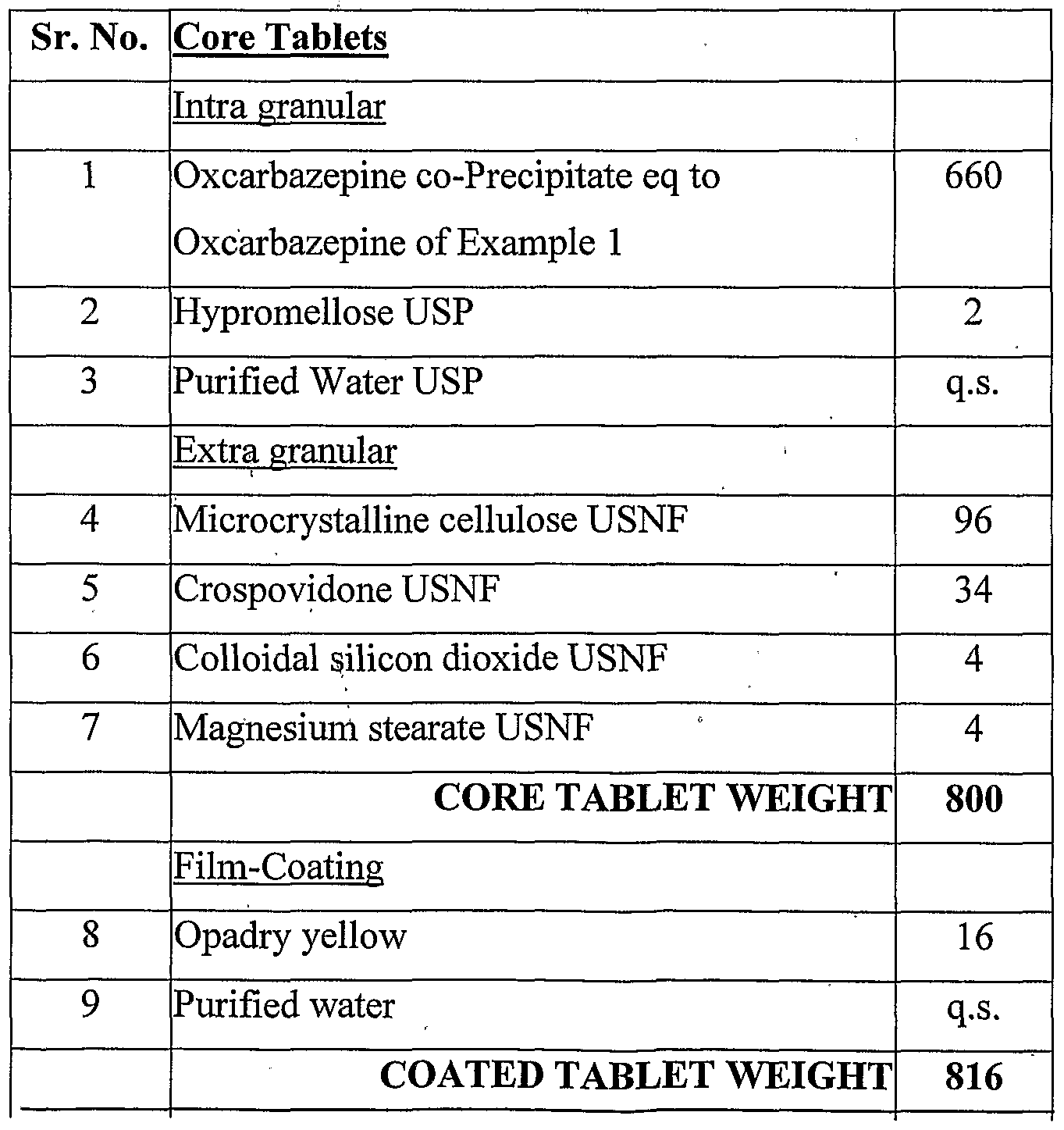
Example 2 Preparation of the tablets of the coprecipitate of Oxcarbazepine
1. Load the Oxcarbazepine co-Precipitate of Example 1 into the rapid mixer granulator (RMG) and mix for 5 mins with impeller 'ON' and chopper 'OFF'.
2. Prepare the binder solution by dispersing Hypromellose in purified water under stirring and continue stirring for 30 mins or till a clear solution forms.
3. , Add the binder solution of step (2) slowly to the material of step (1) at impeller ON" for 2-3 mins till granules mass formed.
4. Mix the above granules at impeller 'ON' and chopper 'ON' for 2-3 mins.
5. Unload the above material from RMG. 6. Sift the Wet mass of step (5) through #14 ASTM to get the wet granules. 7. Granules of step (6) is dried in a tray drier at 60°C till LOD (at 105° C ) reaches less than 2%Sift the dried granules through #20 ASTM. 8 Sift the Extragranular portion (crospovidone, microcrystalline cellulose, colloidal silicon dioxide) through #40 ASTM. 9. Mix the granules of step (8) and material of step (9) in a suitable blender.
10. Weigh and sift the Magnesium stearate through # 60 ASTM and add to the blend of step (10) mix it for 5 mins.
11. Unload the final blend from the blender and Compress the final blend into tablets. 12. Coat the compressed tablets using Opadry Yellow dispersion in purified water at a target weight gain of 2 % w/w.
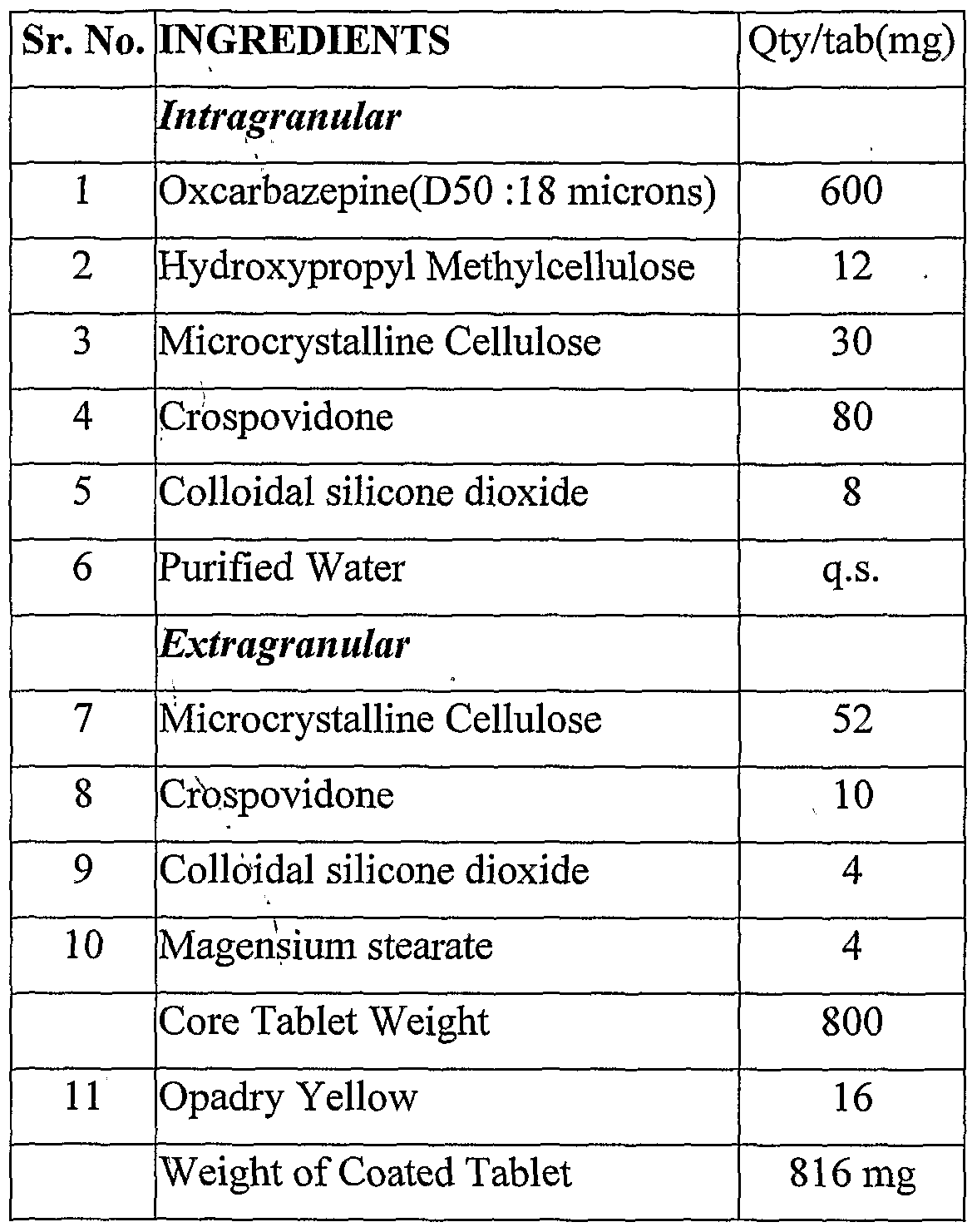
Example 3 Composition of Oxcarbazepine Tablets (Using Normal RMG Strategy)
Procedure:
1. Sift Ingredients 1 ,3,4 and 5 through 40 # ASTM.
2. Dissolve ingredient 2 in purified water under stirring.
3. Load the material of step (1) in RMG and mix for 10 minutes at impeller on and chopper off.
4. Add the granulating fluid of step (2) into the material of step(3) into RMG for a period of 1-2 minutes at impeller on and chopper off.
5. Further mix the mass of step (4) at impeller on and chopper on for 3-4 minutes or till a suitable granular mass formed. 6. Discharge the wet mass then dry the same in Fluid bed dryer at inlet of 60 degree C for 15 mins or till a LOD of less than 2 % w/w.
7. Sift the dried granules through 30 # ASTM.
8. Sift the extragranular excepients 7, 8 and 9 through sieve # 40.
9. Mix the sifted granules of step (7) and sifted excepients of step (8) in double cone mixer for 10 minutes.
10. Sift ingredient 10 through 60 # ASTM and add to the material of step (9) in Double cone blender and mix for 5 minutes.
11. Compress the final blend into tablets at a target tablet weight of 800 mg.
12. Coat the compressed tablets using Opadry Yellow dispersion in purified water at a target weigh't gain of 2 % w/w.
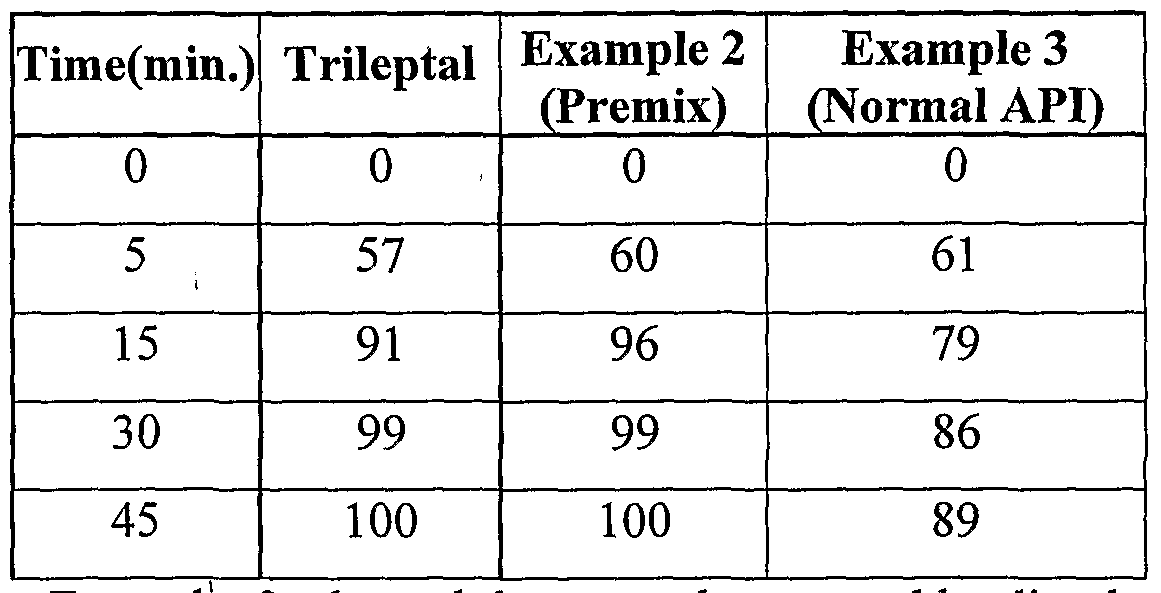
Dissolution of both the batches were carried out together with the reference product Trileptal Tablets 600 mg (Oxcarbazepine Tablets) in Purified water with 1 % SLS in 900 ml at 60 RPM using USP Apparatus II (Paddle). The dissolution results are appended in the below table,
^ Table 2
Comparative dissolution profile of Example 2 and 3 with Trileptal Tablets 600 mg
We found that Example 2 showed better and comparable dissolution profile as compared to Example 3 with respect to Reference Trileptal Tablets by using the Oxcarbazepine co-precipitatέ.
Accelerated stability study of Example 2 was carried out and found to be highly stable. The data are appended in below table,
Table 3 Accelerated Stability data of Example 2
Procedure:
1. Dissolve HPMC 3 cps and 15 cps in Methanol under stirring and continue stirring till clear solution appears. Divide the solution into two parts.
2. Disperse Oxcarbazepine into the solution of step 1 (Part 1)
3. Sift the dispersion of step 2 through # 200 (75 micron)
4. Dissolve the residue left above # 200 sieve (75 micron) of step 3 in the solution of step 1 (Part 2)
5. Add approx. 20 % of the Oxcarbazepine dispersion of step 2 under stirring to the solution of step 4 and continue stirring for 30 mins or till solution becomes clear.
6. Sift Crospovidone and MCC together through # 40 sieve (425 micron) and load the material into Low Shear Mixer like FBP and mix for 5 mins. 7. Granulate the above blend of step 6 using the remaining oxcarbazepine dispersion of step 2 onto FBP.
8. Further granulate the above granules of step 7 using oxcarbazepine dissolved in Methanol : HPMC solution of step 5 onto FBP
9. Dry the above granules of step 8 into the same FBP at 60oC for 30 mins or till LOD reaches below 2 % w/w.
10. Sift the dried granules of step 9 through # 25 sieve (710 micron)
11. Mix the sifted granules of step 10 with the extra granular crospovidone sifted through # 40 sieve (425 micron) and mix the blend in Non-shear tumble type blender like DCB for 20 mins. '
12. Add Mg Stearate and Aerosil (Sifted through # 60 sieve - 250 micron) to the DCB of step 11 and mix for 5 mins.
13. Compress the final blend of step 7 using 18.5 X 8.0 mm Ovaloid shaped punch
Film-coating 1. Add Purified water to a SS .tank.
2. Add Opadry II (Yellow color) to the purified water of step 1 while stirring and continue stirring for 1 hour. 3. Transfer the core tablets into the coating pan and pre warm the tablets at 40 °C for half an hour.
4. Spray the coating suspension onto the core tablets to achieve 2.5 % w/w build up. 5. Cure the coated tablets of step 4 at 40 °C for around 20 mins or till the constant tablet weight is achieved.
It will be understood that various modifications may be made to the embodiments and examples disclosed herein. Therefore the above description and examples should not be construed as limiting, but nierely as exemplification of the various embodiments. Those skilled in the art will envision other modifications within scope and spirit of the claims appended hereto.
Claims
1. A solid dispersion or premix product, which may be a co-precipitate or a co-evaporate comprising:
(i) an effective amount of Oxcarbazepine and (ii) an effective amount of a hydrophilic polymer or a mixture of two or more hydrophilic polymers, (iii) and /or a non-solvent
2. The solid dispersion or premix product of claim 1 wherein the hydrophilic polymer can be chosen from Hydroxypropylmethyl cellulose, hydroxypropyl cellulose, polyvinyl pyrrolidone or any similarly functioning material or mixtures thereof.
3. The solid dispersion or premix product of claim 3 wherein the most preferable hydrophilic polymer is hydrxypropylmethylcellulose.
4. The solid1 dispersion or premix product of claim 1, wherein the non- solvent is selected from a group comprising crospovidone, croscarmellose sodium, sodium starch glycolate and pregelatinised starch and the likes or mixtures thereof.
5. The solid dispersion or premix product of claim 4 wherein the most preferred non-solvent is cropovidone.
6. A solid dosage form comprising solid dispersion or premix product of claim 1 and one or more pharmaceutical excipients.
7. A solid dosage form of claim 6, wherein one or more pharmaceutical excipients are selected from the group comprising of diluents, lubricants, glidants, disintegrants, flavoring agents and solubilizers.
8. A solid dosage form of claim 7, wherein disintegrants are selected from the group comprising of croscarmellose sodium, crospovidone, sodium starch glycolate, low substitute hydroxypropyl cellulose, calcium carboxymethyl cellulose, guar gum, silica, pregelatinised starch, corn starch, alginates or mixtures thereof.
9. A solid dosage form of claim 8 wherein preferable disintegrant is crospovidone.
10. A solid dosage form of claim 7 wherein the lubricants are selected form the group comprising of stearic acid or its derivatives such as magnesiumstearate, calcium stearate, aluminium stearate, polyethylene glycols, sodium stearyl fumarate or mixtures thereof.
11. A solid dosage form of claim 10 wherein the lubricant is preferably magnesium stearate.
12. A solid dispersion product comprising particles of Oxcarbazepine, wherein the oxcarbazepine particles have a median particle size of more than 15 microns.
13. A solid dispersion product as per claim 12, wherein the oxcarbazepine has a preferable median particle size of 15 to 20 microns.
14. A process for the preparation of solid dispersion product and then a solid dosage form of Oxcarbazepine comprising
a) Preparing a solution of a hydrophilic polymer or a mixture of polymers in a solvent and then charging the drug in this reaction mass till a clear solution appears and/or b) Adding a non-solvent to the solution mixture of step (a) and c) Distilling off the solvents till a pasty mass is obtained and d) Adding a solvent to this pasty mass while heating and e) Filtering the slurry thus obtained and then drying it. and f) Mixing these particles of a solid dispersion product with pharmaceutically acceptable adjuncts and g) Formulating the same into suitable oral dosage form like tablets, capsules or sachet. And h) Coating the tablets
15. A process for preparing a solid dosage form as per claim 14, wherein the hydrophilic polymer is selected from hydroxypropylmethyl cellulose, hydroxypropyl cellulose, polyvinyl pyrrolidone or any other acceptable hydrophilic polymer or mixtures thereof.
16. A process for preparing a solid dosage form as per claim 15, wherein the hydrophilic polymer is preferably hydroxypropylmethyl cellulose.
17. A process for preparing a solid dosage form as per claim 14 wherein the non-solvent is selected from a group comprising crospovidone, croscarmellose sodium, sodium starch glycolate and pregelatinised starch and the likes or mixtures thereof.
18. A process for preparing a solid dosage form as per claim 17 wherein the preferable non-solvent is crospovidone.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN1334/DEL/2006 | 2006-06-02 | ||
| IN1334DE2006 | 2006-06-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007141806A1 true WO2007141806A1 (en) | 2007-12-13 |
Family
ID=38578822
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2007/000223 Ceased WO2007141806A1 (en) | 2006-06-02 | 2007-06-04 | Pharmaceutical formulations comprising oxcarbazepine and methods thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2007141806A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008092046A3 (en) * | 2007-01-26 | 2009-03-12 | Isp Investments Inc | Amorphous oxcarbazepine and the production thereof |
| WO2009013594A3 (en) * | 2007-07-25 | 2009-08-13 | Archimica Srl | Process for the preparation of controlled-release solid formulations containing oxcarbazepine, and formulations obtainable by said process |
| EP2200588A1 (en) | 2007-09-25 | 2010-06-30 | Solubest Ltd. | Compositions comprising lipophilic active compounds and method for their preparation |
| EP2441438A1 (en) * | 2010-10-15 | 2012-04-18 | Sanovel Ilac Sanayi ve Ticaret A.S. | Sachet formulations of eslicarbazepine |
| US12133911B2 (en) | 2015-06-09 | 2024-11-05 | Capsugel Belgium Nv | Formulations to achieve rapid dissolution of drug from spray-dried dispersions in capsules |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002094774A2 (en) * | 2001-05-18 | 2002-11-28 | Ranbaxy Laboratories Limited | Oxcarbazepine dosage forms |
| WO2003043602A1 (en) * | 2001-11-20 | 2003-05-30 | Korea Dds Pharmaceutical Co., Ltd. | Solid dispersions containing substituted cyclodextrin and insoluble drug and their preparations |
| US20050175690A1 (en) * | 2003-12-29 | 2005-08-11 | David Edgren | Novel drug compositions and dosage forms |
| WO2007011349A1 (en) * | 2005-07-15 | 2007-01-25 | Teva Pharmaceutical Industries Ltd. | Novel granulation process and granulate produced therefrom |
| WO2007029093A2 (en) * | 2005-09-05 | 2007-03-15 | Ranbaxy Laboratories Limited | Pharmaceutical dosage forms of oxcarbazepine |
-
2007
- 2007-06-04 WO PCT/IN2007/000223 patent/WO2007141806A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002094774A2 (en) * | 2001-05-18 | 2002-11-28 | Ranbaxy Laboratories Limited | Oxcarbazepine dosage forms |
| WO2003043602A1 (en) * | 2001-11-20 | 2003-05-30 | Korea Dds Pharmaceutical Co., Ltd. | Solid dispersions containing substituted cyclodextrin and insoluble drug and their preparations |
| US20050175690A1 (en) * | 2003-12-29 | 2005-08-11 | David Edgren | Novel drug compositions and dosage forms |
| WO2007011349A1 (en) * | 2005-07-15 | 2007-01-25 | Teva Pharmaceutical Industries Ltd. | Novel granulation process and granulate produced therefrom |
| WO2007029093A2 (en) * | 2005-09-05 | 2007-03-15 | Ranbaxy Laboratories Limited | Pharmaceutical dosage forms of oxcarbazepine |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008092046A3 (en) * | 2007-01-26 | 2009-03-12 | Isp Investments Inc | Amorphous oxcarbazepine and the production thereof |
| WO2009013594A3 (en) * | 2007-07-25 | 2009-08-13 | Archimica Srl | Process for the preparation of controlled-release solid formulations containing oxcarbazepine, and formulations obtainable by said process |
| JP2010534234A (en) * | 2007-07-25 | 2010-11-04 | アルキミカ ソシエタ ア レスポンサビリタ リミタータ | Method for preparing controlled release solid formulation containing oxcarbazepine and formulation obtained by the method |
| EP2200588A1 (en) | 2007-09-25 | 2010-06-30 | Solubest Ltd. | Compositions comprising lipophilic active compounds and method for their preparation |
| EP2601935A1 (en) * | 2007-09-25 | 2013-06-12 | Solubest Ltd. | Compositions comprising lipophilic active compounds and method for their preparation |
| US9254268B2 (en) | 2007-09-25 | 2016-02-09 | Solubest Ltd. | Compositions comprising lipophilic active compounds and method for their preparation |
| EP2200588B1 (en) * | 2007-09-25 | 2019-03-20 | Solubest Ltd. | Compositions comprising lipophilic active compounds and method for their preparation |
| EP2441438A1 (en) * | 2010-10-15 | 2012-04-18 | Sanovel Ilac Sanayi ve Ticaret A.S. | Sachet formulations of eslicarbazepine |
| TR201008460A1 (en) * | 2010-10-15 | 2012-05-21 | Sanovel İlaç San.Ve Ti̇c.A.Ş. | Sachet formulations of eslicarbazepine. |
| US12133911B2 (en) | 2015-06-09 | 2024-11-05 | Capsugel Belgium Nv | Formulations to achieve rapid dissolution of drug from spray-dried dispersions in capsules |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| FI87424C (en) | Process for the preparation of solid nifedipine preparations which are readily biodegradable and which have a delayed effect | |
| EP4031110B1 (en) | Dosage forms for tyk2 inhibitors | |
| JP5484910B2 (en) | Revaprazan-containing solid dispersion and method for producing the same | |
| EP3766485A1 (en) | Once daily formulation of lacosamide | |
| CN113573712A (en) | Pharmaceutical composition of nilotinib | |
| US12343430B2 (en) | Methods of improving pharmaceutical substance solubilization and products thereof | |
| WO2011002857A2 (en) | 3-cyanoquinoline tablet formulations and uses thereof | |
| KR20090092288A (en) | Pharmaceutical formulation comprising neurokinin antagonist | |
| JP2021059551A (en) | Pharmaceutical composition comprising phenylaminopyrimidine derivative | |
| WO2008104852A2 (en) | Pharmaceutical compositions comprising adsorbate of fenofibrate | |
| JP2004525887A (en) | New fenofibrate tablets | |
| WO2003039527A1 (en) | Controlled release tablets of metformin | |
| JP2008540644A (en) | New granulation method and granulated material produced therefrom | |
| WO2007141806A1 (en) | Pharmaceutical formulations comprising oxcarbazepine and methods thereof | |
| CN106511291A (en) | Acotiamide hydrochloride controlled release tablet and preparation method thereof | |
| CN113368032A (en) | Pharmaceutical composition, oral solid preparation and preparation method and application thereof | |
| AU2011328307B2 (en) | Pharmaceutical composition for treating HCV infections | |
| EP2190415A2 (en) | Pharmaceutical compositions of fenofibrate | |
| RU2390332C2 (en) | Solid pharmaceutical composition | |
| US20250352552A1 (en) | Pharmaceutical composition comprising meloxicam | |
| US20250352478A1 (en) | Pharmaceutical composition comprising meloxicam | |
| WO2026035689A1 (en) | Vamorolone solid oral dosage forms | |
| US20250352479A1 (en) | Pharmaceutical composition comprising meloxicam | |
| US20100285126A1 (en) | Pharmaceutical compositions of fenofibrate | |
| CN120420289A (en) | Pharmaceutical composition for treating chronic kidney disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07736614 Country of ref document: EP Kind code of ref document: A1 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 07736614 Country of ref document: EP Kind code of ref document: A1 |