WO2007103521A2 - Preventing norvaline and norleucine misincorporation in recombinant proteins - Google Patents
Preventing norvaline and norleucine misincorporation in recombinant proteins Download PDFInfo
- Publication number
- WO2007103521A2 WO2007103521A2 PCT/US2007/005963 US2007005963W WO2007103521A2 WO 2007103521 A2 WO2007103521 A2 WO 2007103521A2 US 2007005963 W US2007005963 W US 2007005963W WO 2007103521 A2 WO2007103521 A2 WO 2007103521A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seed
- culture
- leucine
- norvaline
- fermentor
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P21/00—Preparation of peptides or proteins
- C12P21/02—Preparation of peptides or proteins having a known sequence of two or more amino acids, e.g. glutathione
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/20—Bacteria; Culture media therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/32—Amino acids
Definitions
- the invention relates to methods of preventing norvaline raisincorporation in recombinant proteins.
- the invention provides a method for preventing misincorporation of norvaline in place of leucine in recombinant proteins.
- the method can be utilized in the production of any recombinant protein, and may be used with both large and small scale production.
- the method involves feeding the culture with the amino acids leucine, isoleucine, and methionine and is particularly suited for the prevention of norvaline and norleucine misincorporation in recombinant proteins without compromising cell densities and titers.
- the invention is directed to a method for preventing norvaline and norleucine misincorporation into a recombinant protein comprising providing a culture of host cells, which inducibly express the recombinant protein, an amount of amino acids leucine, isoleucine, and methionine sufficient to maintain a constant amino acid concentration of each of the amino acids throughout induction of recombinant protein expression.
- the leucine, isoleucine, and methionine are present in a glucose feed.
- the glucose feed comprises 5 g/L leucine, 6.3 g/L isoleucine, and 4 g/L methionine.
- the method further comprises supplying leucine and isoleucine to the culture at glucose exhaustion.
- the recombinant protein is TFPI or ala-TFPI.
- the culture is in a fermentor.
- the host cells are E. cailL
- FIG. 1 Map of plasmid pMON37621.
- the invention provides a method for preventing misincorporation of norvaline in place of leucine in recombinant proteins.
- the method involves feeding the culture with leucine, isoleucine, and methionine. Addition of methionine prevents incorporation of norleucine.
- Leucine inhibits and represses the enzymes involved in the synthesis of both norvaline and norleucine.
- leucine also affects other cellular functions and results in lower cell densities and titers at the end of fermentation. Addition of isoleucine results in greatly increased amount of recombinant protein production.
- the present method prevents norvaline and norleucine incorporation without compromising on cell densities and titers achieved in the absence of leucine.
- Methods of the invention can be used in the production of any recombinant protein and can be used with both large and small scale production. It is particularly useful for use with high density fermentation, for example, of E. coli host cells.
- One embodiment of the invention is illustrated in the examples below for the production of recombinant ala-TFPI (TFPI as shown in SEQ ED NO:1 comprising an N-terminal alanine). These examples are provided for purposes of illustration only and are not intended to limit the scope of the invention.
- the initial volume in the fermentor is 6400 L.
- Seed 1 Shake Flask.
- the fermentation process begins by thawing a frozen vial of a Working Cell Bank. The contents of this vial (1 mL) is used to inoculate 0.5 L of seed 1 medium in a seed 1 shake flask. The flask is incubated at 37 ⁇ 2° C. and mixed at 200 RPM. The culture is grown for approximately 9 generations until a cell density of 0.9- 1.7 OD is reached. The Seed 1 culture is then transferred to the Seed 2 fermentor.
- Seed 2-30 L Fermentor The 30 L of Seed 2 medium is inoculated with the 0.5 L contents of the seed 1 culture.
- the seed 2 medium is essentially the same as the Seed 1 medium, except for the addition of 0.1 mL/L Ucon antifoam.
- the Seed 2 fermentation preferably is carried out at a temperature of 37 ⁇ 2° C, with an air sparge of >2 LPM to maintain the dissolved oxygen above 10% of air saturation.
- the initial pH of the medium preferably is 7.2 ⁇ 0.2. When the cells have grown approximately 6 generations to a density of 0.9-1.7 OD, the Seed 2 culture is transferred to the 10,000 L fermentor.
- the production fermentor is controlled for the following parameters.
- the temperature of the growth phase preferably is 37 ⁇ 2° C.
- the temperature set point is changed from 37° C. to 30° C. approximately 0.5 hours before the induction of TFPI or TFPI analog expression, and the temperature of the expression phase preferably is 30 ⁇ 2° C.
- pH is controlled by the addition of concentrated NH40H and preferably is maintained at 6.9 ⁇ 0.2.
- a temporary pH spike to approximately pH 7.4 ⁇ 0.2 occurs when the initial supply of glucose is depleted and just prior to the start of the nutrient feed.
- dissolved oxygen preferably 0.1-0.5 atm
- Cell growth in the 10,000 L production fermentation begins as a simple batch culture, using the glucose of the starting medium. Glucose is depleted when the cell density reaches approximately 40 OD, as indicated by a pH increase to 7.4 ⁇ 0.2. At that time, a glucose/nutrient feed is started.
- the nutrient feed contains 550 g/L glucose, 18 g/L sodium polyphosphate, 6.65 g/L magnesium sulfate, and 4 g/L methionine. The nutrient feed rate is increased exponentially.
- the temperature set point is changed so that the culture reaches 30 ⁇ 2° C. within one hour after induction.
- the culture is harvested approximately 12 hours after induction.
- the TFPI or TFPI analog concentration at harvest is approximately 5 g/L TFPI as determined by SDS-PAGE analysis.
- Purified water is added to the collected cells to a volume of ⁇ 10,000 liters, DTPA is added to a final concentration of 1 mM and processed through the BTUX- 510 centrifuge as previously described. This wash step is repeated a total of two times.
- the flask is incubated at 37 ⁇ 2° C. and mixed at 200 RPM.
- the culture is grown for approximately 9 generations until a cell density of 0.9-1.7 OD is reached.
- a development seed stock is then frozen and used for inoculation of the seed culture of all 10 L fermentations.
- the contents of the development stock are used to inoculate 200 mL of culture in a 1 L shake flask.
- the flask is incubated at 37 ⁇ 2° C. and mixed at 200 RPM.
- the culture is grown for approximately 9 generations until a cell density of 0.9-1.7 OD is reached.
- [27] 10 L Production Fermentor A volume of the seed flask is transferred to the 10 L fermentor containing 6.9 L of medium to obtain an intial cell density of 0.006 OD 550 .
- the production medium composition is the same as shown in Table 3 for the 10,000 L fermentor.
- the 10 L production fermentor is controlled and operated the same as the 10,000 L fermentor as described in sections 13, 14 and 15 above.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Microbiology (AREA)
- General Health & Medical Sciences (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- General Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Tropical Medicine & Parasitology (AREA)
- Virology (AREA)
- Biomedical Technology (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Sampling And Sample Adjustment (AREA)
- Accommodation For Nursing Or Treatment Tables (AREA)
- Other Investigation Or Analysis Of Materials By Electrical Means (AREA)
Abstract
Methods of preventing norvaline and norleucine misincorporation in recombinant proteins.
Description
PREVENTING NORVALINE AND NORLEUCINE MISINCORPORATION IN RECOMBINANT PROTEINS
FIELD OF THE INVENTION
[01] The invention relates to methods of preventing norvaline raisincorporation in recombinant proteins.
BACKGROUND
[02] Previous efforts to prevent norleucine misincorporation into recombinant proteins involved supplementing medium with leucine and/or methionine (e.g., U.S. Patent 5,599,690; Apostol et ah, J. Biol. Chem. 272, 28980-88, 1997) or supplementing the medium with combinations of leucine, methionine, and cysteine (e.g., U.S. Patent 5,622,845). There is a need in the art for methods of preventing misincorporation of norvaline and norleucine in recombinant proteins.
SUMMARY OF THE INVENTION
[03] The invention provides a method for preventing misincorporation of norvaline in place of leucine in recombinant proteins. The method can be utilized in the production of any recombinant protein, and may be used with both large and small scale production. The method involves feeding the culture with the amino acids leucine, isoleucine, and methionine and is particularly suited for the prevention of norvaline and norleucine misincorporation in recombinant proteins without compromising cell densities and titers.
[04] Accordingly, in one embodiment, the invention is directed to a method for preventing norvaline and norleucine misincorporation into a recombinant protein comprising providing a culture of host cells, which inducibly express the recombinant protein, an amount of amino acids leucine, isoleucine, and methionine sufficient to maintain a constant amino acid concentration of each of the amino acids throughout induction of recombinant protein expression.
[05] In certain embodiments of the method, the leucine, isoleucine, and methionine are present in a glucose feed.
[06] In additional embodiments of the method, the glucose feed comprises 5 g/L leucine, 6.3 g/L isoleucine, and 4 g/L methionine.
[071 I*1 still further embodiments, the method further comprises supplying leucine and isoleucine to the culture at glucose exhaustion.
[08] In other embodiments, the recombinant protein is TFPI or ala-TFPI.
[09] In yet an additional embodiment, the culture is in a fermentor.
[10] In certain embodiments of the subject invention, the host cells are E. cailL
[11] These and other embodiments of the subject invention will readily occur to those of skill in the art in view of the disclosure herein.
BRIEF DESCRIPTION OF THE DRAWINGS
[12] FIG. 1. Map of plasmid pMON37621.
DETAILED DESCRIPTION OF THE INVENTION
[13] The invention provides a method for preventing misincorporation of norvaline in place of leucine in recombinant proteins. The method involves feeding the culture with leucine, isoleucine, and methionine. Addition of methionine prevents incorporation of norleucine. Leucine inhibits and represses the enzymes involved in the synthesis of both norvaline and norleucine. However, leucine also affects other cellular functions and results in lower cell densities and titers at the end of fermentation. Addition of isoleucine results in greatly increased amount of recombinant protein production. Thus, the present method prevents norvaline and norleucine incorporation without compromising on cell densities and titers achieved in the absence of leucine.
[14] Methods of the invention can be used in the production of any recombinant protein and can be used with both large and small scale production. It is particularly useful for use with high density fermentation, for example, of E. coli host cells. One embodiment of the invention is illustrated in the examples below for the production of recombinant ala-TFPI (TFPI as shown in SEQ ED NO:1 comprising an N-terminal
alanine). These examples are provided for purposes of illustration only and are not intended to limit the scope of the invention.
[15] All patents, patent applications, and references cited in this disclosure are expressly incorporated herein by reference.
EXAMPLE l
Fermentation Conditions
[16] The following procedures were used in Example 2, below.
[17] Preparation of the Master and Working Cell Bank, Master and Working Cell Banks were prepared from production strains as follows for use in the experiments described below. See also US 2005/0037475. To prepare a Master Cell Bank, a frozen vial of parent MON210/pMON37621 cells (ATCC Accession No. PTA-5564) is thawed and grown in a shake flask for approximately 9 generations in the defined production seed medium with spectinomycin. Vials of cells with 10% glycerol are frozen and maintained at —70° C. Working Cell Banks are prepared by thawing a master stock vial and growing the cells as described for the Master Cell Bank. MON210/pMON37621 cells comprise pMON37621, shown in FIG. 1, which comprises a coding sequence for ala-TFPI,
[18] Fermentation Conditions. Growth of seed cultures for inoculation of the 10,000 L fermentor comprises two stages: (1) Seed 1 shake flask and (2) Seed 2 fermentor. The composition of the media used during the fermentation process are listed in Table 1, which also lists the components of medium used for large scale production of ala- TFPI. KOH and H2SO4 are used to adjust the medium pH of the Seed 1 and Seed 2 media. NH4OH and H2SO4 are used to adjust pH of fermentor medium. NH4OH also is used to control pH during the fermentation.
[19] USP purified water is used throughout the fermentation process. The selective antibiotic spectinomycin is used during the preparation of the Working Cell Bank and is not used during the inoculum preparation or fermentation process.
Table 1. Fermentation Media Composition and Concentration
1 The initial volume in the fermentor is 6400 L.
2Actual glucose concentration at start is 34-42 g/L due to moisture content
(<9%) of Cerelose.
33.5 L of Ucon antifoam added at start. Additional antifoam (up to 12.5 L) may be added during the fermentation.
[20] Seed 1 — Shake Flask. The fermentation process begins by thawing a frozen vial of a Working Cell Bank. The contents of this vial (1 mL) is used to inoculate 0.5 L of seed 1 medium in a seed 1 shake flask. The flask is incubated at 37±2° C. and mixed at 200
RPM. The culture is grown for approximately 9 generations until a cell density of 0.9- 1.7 OD is reached. The Seed 1 culture is then transferred to the Seed 2 fermentor.
[21] Seed 2-30 L Fermentor. The 30 L of Seed 2 medium is inoculated with the 0.5 L contents of the seed 1 culture. The seed 2 medium is essentially the same as the Seed 1 medium, except for the addition of 0.1 mL/L Ucon antifoam. The Seed 2 fermentation preferably is carried out at a temperature of 37±2° C, with an air sparge of >2 LPM to maintain the dissolved oxygen above 10% of air saturation. The initial pH of the medium preferably is 7.2±0.2. When the cells have grown approximately 6 generations to a density of 0.9-1.7 OD, the Seed 2 culture is transferred to the 10,000 L fermentor.
[22] 10,000 L Production Fermentor. The entire contents of the Seed 2 fermentor are transferred to the 10,000 L fermentor containing approximately 6,400 L of production medium. The production medium composition is shown in Table 3. The production fermentor is controlled for the following parameters. The temperature of the growth phase preferably is 37±2° C. The temperature set point is changed from 37° C. to 30° C. approximately 0.5 hours before the induction of TFPI or TFPI analog expression, and the temperature of the expression phase preferably is 30±2° C. pH is controlled by the addition of concentrated NH40H and preferably is maintained at 6.9±0.2. A temporary pH spike to approximately pH 7.4±0.2 occurs when the initial supply of glucose is depleted and just prior to the start of the nutrient feed. Finally, dissolved oxygen (preferably 0.1-0.5 atm) is controlled by adjusting agitation rate, sparge rate, and the proportion of oxygen in the sparge gas.
[23] Cell growth in the 10,000 L production fermentation begins as a simple batch culture, using the glucose of the starting medium. Glucose is depleted when the cell density reaches approximately 40 OD, as indicated by a pH increase to 7.4±0.2. At that time, a glucose/nutrient feed is started. The nutrient feed contains 550 g/L glucose, 18 g/L sodium polyphosphate, 6.65 g/L magnesium sulfate, and 4 g/L methionine. The nutrient feed rate is increased exponentially.
[24] When the cell density reaches approximately 100 OD, the culture is induced to produce TFPI or TFPI analog by the addition of IPTG (e.g., 187+/-3 g of IPTG per
fermentation, nominal volume=9500 L) and the glucose/nutrient feed rate is reduced to limit the glucose level during the expression phase. The temperature set point is changed so that the culture reaches 30±2° C. within one hour after induction. The culture is harvested approximately 12 hours after induction. The TFPI or TFPI analog concentration at harvest is approximately 5 g/L TFPI as determined by SDS-PAGE analysis.
[25] Cell Harvest, Dissolution ofRefractile Bodies, and Refolding of the Expressed TFPI Cell Harvest. For cell harvest, the fermentation broth is adjusted to pH 5.5-6 and the delivery of oxygen and glucose are discontinued. Agitation is reduced, and the broth temperature is decreased to 5-10° C. The chelator DTPA is added to a final concentration of 1 mM. The DTPA addition is made using a stock solution that has been pH adjusted to 5.5-6.0 with citric acid. The harvest culture is fed to a BTUX-510 centrifuge operated at a flow rate to minimize loss of solids in the supernatant. Solids containing the harvested cells are continually pumped to a tank until the fermentor is empty. Purified water is added to the collected cells to a volume of ~10,000 liters, DTPA is added to a final concentration of 1 mM and processed through the BTUX- 510 centrifuge as previously described. This wash step is repeated a total of two times.
EXAMPLE 2
[26] Fermentation Conditions. Growth of seed cultures for inoculation of the 10 L fermentor comprises two stages: (1) Expansion of working cell bank and freezing of development cell bank and (2) Seed 1 shake flask. The composition of the media used during the fermentation process are the same as listed in Table 1. KOH and H2SO4 are used to adjust the medium pH of the Seed 1 media. NH4OH and H2SO4 are used to adjust pH of fermentor medium. NH4OH also is used to control pH during the fermentation. Seed 1 — Shake Flask. The fermentation process begins by thawing a frozen vial of a Working Cell Bank. The contents of this vial (1 mL) is used to inoculate 0.5 L of seed 1 medium in a seed 1 shake flask. The flask is incubated at 37±2° C. and mixed at 200 RPM. The culture is grown for approximately 9 generations until a cell density of 0.9-1.7 OD is reached. A development seed stock is then frozen and used for inoculation of the seed culture of all 10 L fermentations. The contents of the development stock are used to inoculate 200 mL of culture in a 1 L shake flask. The flask is incubated at 37±2° C. and mixed at 200 RPM. The culture is grown for approximately 9 generations until a cell density of 0.9-1.7 OD is reached.
[27] 10 L Production Fermentor. A volume of the seed flask is transferred to the 10 L fermentor containing 6.9 L of medium to obtain an intial cell density of 0.006 OD550. The production medium composition is the same as shown in Table 3 for the 10,000 L fermentor. The 10 L production fermentor is controlled and operated the same as the 10,000 L fermentor as described in sections 13, 14 and 15 above.
Effects of supplementation fermentation
[28] Four types of experiments were carried out to determine the effect of supplementing the fermentation runs with isoleucine, leucine, and methionine, added either in the feed only or in the feed and also as a bolus at the beginning of stage 2.
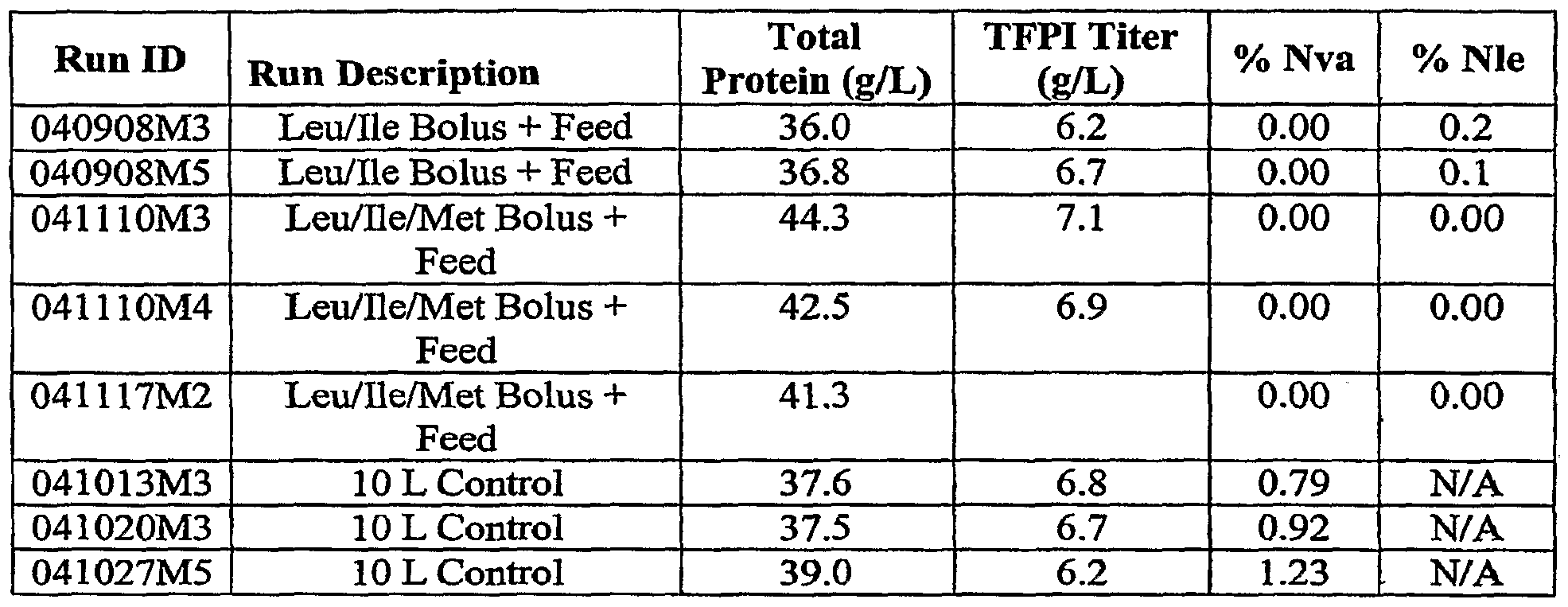
[29] Experiment type 1. Addition of the Ile/Leu/Met as a bolus in second stage of fermentation and in the feed, or addition of Ile/Leu as a bolus and in the feed. The data is presented in Table 2.
Table 2.
[30] Experiment type 2. Addition of Ile/Leu/Met or Leu/Ile in the feed only in stage 2. The data is shown in Table 3.
Table 3.
[31] Experiment type 3. In these experiments the fermentor was run with an initial seed in the presence of Leu/Ile/Met/Val/Thr. The use of Leu, He, and Met from the beginning prevents any norvaline or norleucine from being produced at all, so there is none to be incorporated even at a very low level in the recombinant protein product. The addition of the valine and the threonine were used to help cell growth.
[32] Experiment type 4. These experiments demonstrate the benefit of adding the lie into the feed in terms of the final amount of TFPI produced.
Claims
1. A method of preventing norvaline and norleucine misincorporation into a recombinant protein comprising providing a culture of host cells which inducibly express the recombinant protein an amount of amino acids leucine, isoleucine, and methionine sufficient to maintain a constant amino acid concentration of each of the amino acids throughout induction of recombinant protein expression.
2. The method of embodiment 1 wherein the leucine, isoleucine, and methionine are present in a glucose feed.
3. The method of embodiment 2 wherein the glucose feed comprises 5 g/L leucine, 6.3 g/L isoleucine, and 4 g/L methionine.
4. The method of embodiment 2 further comprising supplying leucine and isoleucine to the culture at glucose exhaustion.
5. The method of embodiment 1 wherein the recombinant protein is TFPI or ala- TFPL
6. The method of embodiment 1 wherein the culture is in a fermentor.
7. The method of embodiment 1 wherein the host cells are E. coli.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP07752647A EP1996697A2 (en) | 2006-03-07 | 2007-03-07 | Preventing norvaline and norleucine misincorporation in recombinant proteins |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US77942906P | 2006-03-07 | 2006-03-07 | |
| US60/779,429 | 2006-03-07 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2007103521A2 true WO2007103521A2 (en) | 2007-09-13 |
| WO2007103521A3 WO2007103521A3 (en) | 2008-02-21 |
Family
ID=38475570
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2007/005963 Ceased WO2007103521A2 (en) | 2006-03-07 | 2007-03-07 | Preventing norvaline and norleucine misincorporation in recombinant proteins |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP1996697A2 (en) |
| WO (1) | WO2007103521A2 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014047311A1 (en) * | 2012-09-19 | 2014-03-27 | Genentech, Inc. | Methods and compositions for preventing norleucine misincorporation into proteins |
| US10808037B1 (en) | 2007-07-09 | 2020-10-20 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| JP2022514083A (en) * | 2018-12-20 | 2022-02-09 | サノフィ-アベンティス・ドイチュラント・ゲゼルシャフト・ミット・ベシュレンクテル・ハフツング | How to reduce misuptake of non-canonical branched chain amino acids |
| WO2024079114A1 (en) | 2022-10-11 | 2024-04-18 | UCB Biopharma SRL | Process for the production of recombinant proteins |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4522997B2 (en) * | 2003-08-13 | 2010-08-11 | ノバルティス バクシンズ アンド ダイアグノスティックス,インコーポレーテッド | Improved method for purifying TFPI and TFPI analogs |
| US8603781B2 (en) * | 2003-09-25 | 2013-12-10 | Monsanto Technology Llc | Prevention of incorporation of non-standard amino acids into protein |
-
2007
- 2007-03-07 WO PCT/US2007/005963 patent/WO2007103521A2/en not_active Ceased
- 2007-03-07 EP EP07752647A patent/EP1996697A2/en not_active Withdrawn
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11639395B2 (en) | 2007-07-09 | 2023-05-02 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US12528875B2 (en) | 2007-07-09 | 2026-01-20 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US12173080B1 (en) | 2007-07-09 | 2024-12-24 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US12145997B2 (en) | 2007-07-09 | 2024-11-19 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US12098211B2 (en) | 2007-07-09 | 2024-09-24 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US11999791B2 (en) | 2007-07-09 | 2024-06-04 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US11987638B1 (en) | 2007-07-09 | 2024-05-21 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US11987637B1 (en) | 2007-07-09 | 2024-05-21 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US10808037B1 (en) | 2007-07-09 | 2020-10-20 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US10906986B2 (en) | 2007-07-09 | 2021-02-02 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US11976127B1 (en) | 2007-07-09 | 2024-05-07 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| US11078294B2 (en) | 2007-07-09 | 2021-08-03 | Genentech, Inc. | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| AU2017203049B2 (en) * | 2012-09-19 | 2019-06-27 | Genentech, Inc. | Methods and compositions for preventing norleucine misincorporation into proteins |
| US11015214B2 (en) | 2012-09-19 | 2021-05-25 | Genentech, Inc. | Microorganisms comprising a mutant metA allele for reducing norleucine misincorporation into proteins |
| US10421984B2 (en) | 2012-09-19 | 2019-09-24 | Genentech, Inc. | Methods for reducing norleucine misincorporation into proteins using a microorganism comprising a mutant metA allele |
| WO2014047311A1 (en) * | 2012-09-19 | 2014-03-27 | Genentech, Inc. | Methods and compositions for preventing norleucine misincorporation into proteins |
| EP3502267A1 (en) * | 2012-09-19 | 2019-06-26 | F. Hoffmann-La Roche AG | Methods and compositions for preventing norleucine misincorporation into proteins |
| JP2019068810A (en) * | 2012-09-19 | 2019-05-09 | ジェネンテック, インコーポレイテッド | Methods and compositions for preventing norleucine misincorporation into proteins |
| US10179925B2 (en) | 2012-09-19 | 2019-01-15 | Genentech, Inc. | Compositions for preventing norleucine misincorporation into proteins |
| US9850514B2 (en) | 2012-09-19 | 2017-12-26 | Genentech, Inc. | Methods and compositions for preventing norleucine misincorporation into proteins |
| JP2015528314A (en) * | 2012-09-19 | 2015-09-28 | ジェネンテック, インコーポレイテッド | Method and composition for preventing erroneous incorporation of norleucine into protein |
| US20220064692A1 (en) * | 2018-12-20 | 2022-03-03 | Sanofi Aventis Deutschland Gmbh | Method for reducing misincorporation of non-canonical branched-chain amino acids |
| JP2022514083A (en) * | 2018-12-20 | 2022-02-09 | サノフィ-アベンティス・ドイチュラント・ゲゼルシャフト・ミット・ベシュレンクテル・ハフツング | How to reduce misuptake of non-canonical branched chain amino acids |
| JP7611144B2 (en) | 2018-12-20 | 2025-01-09 | サノフィ-アベンティス・ドイチュラント・ゲゼルシャフト・ミット・ベシュレンクテル・ハフツング | Methods for reducing misincorporation of non-canonical branched chain amino acids |
| WO2024079114A1 (en) | 2022-10-11 | 2024-04-18 | UCB Biopharma SRL | Process for the production of recombinant proteins |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1996697A2 (en) | 2008-12-03 |
| WO2007103521A3 (en) | 2008-02-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Nakano et al. | Influence of acetic acid on the growth of Escherichia coli K12 during high-cell-density cultivation in a dialysis reactor | |
| JP2564506B2 (en) | Method for producing heterologous protein | |
| KR101677328B1 (en) | A microorganism producing O-phosphoserine and a method for producing O-phosphoserine or L-cysteine using the same | |
| EP1360275B1 (en) | Culture of crypthecodinium cohnii and microorganisms derived therefrom | |
| WO2007103521A2 (en) | Preventing norvaline and norleucine misincorporation in recombinant proteins | |
| WO2025190352A1 (en) | Method for preparing recombinant human albumin with high expression and low o-glycosylation level | |
| CN106868079B (en) | Culture medium for fermenting polymyxin B sulfate and method for producing polymyxin B sulfate through fermentation | |
| US5334512A (en) | Fatty acid supplemented medium for recombinant production of human serum albumin by yeast | |
| Lee et al. | Bacteriorhodopsin production by cell recycle culture of Halobacterium halobium | |
| EP2611900B1 (en) | Alkaline feed | |
| EP2671952B1 (en) | Method for preparing cyclic lipopeptide compound | |
| CN111057672B (en) | Recombinant strain and application thereof | |
| US5866371A (en) | Process for using the yeast ADH II promoter system for the production of heterologous proteins in high yields | |
| EP1416947A1 (en) | Fermentation medium and method | |
| CN118166053B (en) | A method for preparing recombinant human albumin with low O-glycosylation level | |
| CN111909859A (en) | Pichia pastoris low-temperature culture medium | |
| CN112111408B (en) | Micafungin precursor WF11899A production strain and fermentation method | |
| Yoon et al. | Regulation of trp promoter for production of bovine somatotropin in recombinant Escherichia coli fed-batch fermentation | |
| WO2018064036A1 (en) | Methods of culturing aurantiochytrium using acetate as an organic carbon source | |
| EP1169432A1 (en) | Process for the production of yeast biomass comprising functionally deleted hxk2 genes | |
| CN103833828B (en) | Extraction method of insulin glargine precursor protein | |
| CN103119056B (en) | For the preparation of the modification method of recombinant human somatropin | |
| KR100255270B1 (en) | Do-stat fed batch culture for mass production of human parathyroid hormone | |
| CN121472346A (en) | Method for producing sirolimus by fermentation of streptomycete | |
| JP2002542262A (en) | Pseudomycin production by Pseudomonas syringe |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2007752647 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |