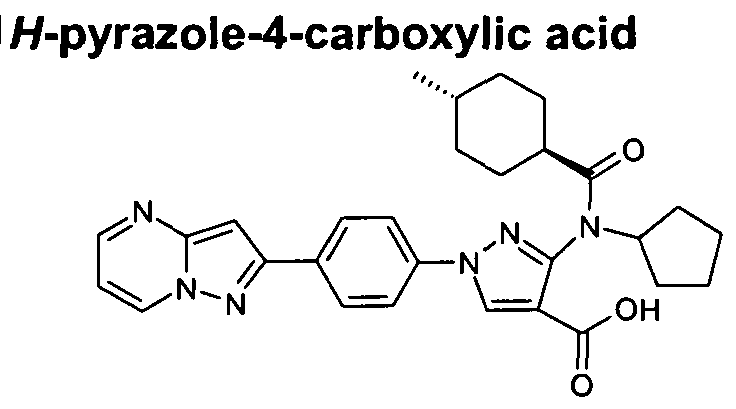WO2007039146A1 - 4-carboxy pyrazole derivatives as anti-viral agents - Google Patents
4-carboxy pyrazole derivatives as anti-viral agents Download PDFInfo
- Publication number
- WO2007039146A1 WO2007039146A1 PCT/EP2006/009238 EP2006009238W WO2007039146A1 WO 2007039146 A1 WO2007039146 A1 WO 2007039146A1 EP 2006009238 W EP2006009238 W EP 2006009238W WO 2007039146 A1 WO2007039146 A1 WO 2007039146A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pyrazole
- amino
- carboxylic acid
- carbonyl
- methylcyclohexyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C[C@](CC1)CC[C@@]1C(*(C1CCOCC1)C(C(C(O)=O)=C1)=*C*1c(cc1)ccc1-c1cc(cccc2)c2[o]1)=O Chemical compound C[C@](CC1)CC[C@@]1C(*(C1CCOCC1)C(C(C(O)=O)=C1)=*C*1c(cc1)ccc1-c1cc(cccc2)c2[o]1)=O 0.000 description 2
- QPCSSZCJLBAUJZ-UHFFFAOYSA-N CC(C)N(C(C1CCC(C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c([o]c1c2)nc1ccc2N Chemical compound CC(C)N(C(C1CCC(C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c([o]c1c2)nc1ccc2N QPCSSZCJLBAUJZ-UHFFFAOYSA-N 0.000 description 1
- VYRFHGZAGJTPDY-UHFFFAOYSA-N CC(C)N(C(C1CCC(C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1nc2ncccc2cc1 Chemical compound CC(C)N(C(C1CCC(C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1nc2ncccc2cc1 VYRFHGZAGJTPDY-UHFFFAOYSA-N 0.000 description 1
- RGMBUHJLAFOSGZ-UHFFFAOYSA-N CC(C)N(C(C1CCCCC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1cc2ncccc2[o]1 Chemical compound CC(C)N(C(C1CCCCC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1cc2ncccc2[o]1 RGMBUHJLAFOSGZ-UHFFFAOYSA-N 0.000 description 1
- GSJDSYWREIKDCH-IYARVYRRSA-N CC(C)N(C([C@H](CC1)CC[C@@H]1C(F)(F)F)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1n[n]2c(N)ccnc2c1 Chemical compound CC(C)N(C([C@H](CC1)CC[C@@H]1C(F)(F)F)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1n[n]2c(N)ccnc2c1 GSJDSYWREIKDCH-IYARVYRRSA-N 0.000 description 1
- XMINRHGNSQJMIZ-KESTWPANSA-N CC(C)N(C([C@H]1CC[C@H](C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c([o]c1ccc2)cc1c2N Chemical compound CC(C)N(C([C@H]1CC[C@H](C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c([o]c1ccc2)cc1c2N XMINRHGNSQJMIZ-KESTWPANSA-N 0.000 description 1
- XUHXAUQHHZEXDS-KESTWPANSA-N CC(C)N(C([C@H]1CC[C@H](C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1c[n](ccnc2)c2n1 Chemical compound CC(C)N(C([C@H]1CC[C@H](C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1c[n](ccnc2)c2n1 XUHXAUQHHZEXDS-KESTWPANSA-N 0.000 description 1
- YNIRJJZRLIHYRX-IYARVYRRSA-N CC(C)N(C([C@H]1CC[C@H](C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c1ccc(-c2c[n](cccc3)c3n2)[s]1 Chemical compound CC(C)N(C([C@H]1CC[C@H](C)CC1)=O)c(c(C(O)=O)c1)n[n]1-c1ccc(-c2c[n](cccc3)c3n2)[s]1 YNIRJJZRLIHYRX-IYARVYRRSA-N 0.000 description 1
- MKBWYMGKSNSAKO-UHFFFAOYSA-N CC(C)N(C(c(c(Cl)c1)ccc1Cl)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1n[n](cccn2)c2c1 Chemical compound CC(C)N(C(c(c(Cl)c1)ccc1Cl)=O)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1n[n](cccn2)c2c1 MKBWYMGKSNSAKO-UHFFFAOYSA-N 0.000 description 1
- SJJIQLGLOOSGTP-UHFFFAOYSA-N CC(CC1)CCC1C(N(C(C)OC(c1c2)=O)c1n[n]2-c1ccc(-c2cc3ccccc3[o]2)nc1)=O Chemical compound CC(CC1)CCC1C(N(C(C)OC(c1c2)=O)c1n[n]2-c1ccc(-c2cc3ccccc3[o]2)nc1)=O SJJIQLGLOOSGTP-UHFFFAOYSA-N 0.000 description 1
- SQXBOCNNAOFSJA-AFARHQOCSA-N CCOC(C(C(N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=[I]C1)=CN1c(cc1)ccc1-c1nc2ccccc2[o]1)=O Chemical compound CCOC(C(C(N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=[I]C1)=CN1c(cc1)ccc1-c1nc2ccccc2[o]1)=O SQXBOCNNAOFSJA-AFARHQOCSA-N 0.000 description 1
- SEJARARDGJBNNI-UHFFFAOYSA-N CCOC(c1c[nH]nc1N(C(C)C)C(c1ccc(C)cc1)=O)=O Chemical compound CCOC(c1c[nH]nc1N(C(C)C)C(c1ccc(C)cc1)=O)=O SEJARARDGJBNNI-UHFFFAOYSA-N 0.000 description 1
- MVRVYRQYDNDMTM-XUTJKUGGSA-N CCOC(c1c[n](-c(cc2)ccc2-c2c[n](cc[s]3)c3n2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2c[n](cc[s]3)c3n2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O MVRVYRQYDNDMTM-XUTJKUGGSA-N 0.000 description 1
- ACNSKZONSZTEAV-AFARHQOCSA-N CCOC(c1c[n](-c(cc2)ccc2-c2c[n](cccc3CN)c3n2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2c[n](cccc3CN)c3n2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O ACNSKZONSZTEAV-AFARHQOCSA-N 0.000 description 1
- ZFPUYGAJTNEKDU-AQYVVDRMSA-N CCOC(c1c[n](-c(cc2)ccc2-c2c[n](ccnc3)c3n2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2c[n](ccnc3)c3n2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O ZFPUYGAJTNEKDU-AQYVVDRMSA-N 0.000 description 1
- QLKPNOMJLUFUGI-AQYVVDRMSA-N CCOC(c1c[n](-c(cc2)ccc2-c2cc(c(N)ccc3)c3[o]2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2cc(c(N)ccc3)c3[o]2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O QLKPNOMJLUFUGI-AQYVVDRMSA-N 0.000 description 1
- NEBDLBFCZNLCNM-UHFFFAOYSA-N CCOC(c1c[n](-c(cc2)ccc2-c2cc(nccc3)c3[o]2)nc1N(C(C)C)C(c1c(C)cc(C)cc1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2cc(nccc3)c3[o]2)nc1N(C(C)C)C(c1c(C)cc(C)cc1)=O)=O NEBDLBFCZNLCNM-UHFFFAOYSA-N 0.000 description 1
- RMHFFAKAQNILAI-AFARHQOCSA-N CCOC(c1c[n](-c(cc2)ccc2-c2cc3ccccc3[s]2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2cc3ccccc3[s]2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O RMHFFAKAQNILAI-AFARHQOCSA-N 0.000 description 1
- USNOLWHRTHJGSF-JKIUYZKVSA-N CCOC(c1c[n](-c(cc2)ccc2-c2cc3ncc(C)cc3[o]2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2cc3ncc(C)cc3[o]2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O USNOLWHRTHJGSF-JKIUYZKVSA-N 0.000 description 1
- RKYCZSRLFGEFGM-VIQNHZKISA-N CCOC(c1c[n](-c(cc2)ccc2-c2cc3ncccc3[o]2)nc1N(C1COCC1)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2cc3ncccc3[o]2)nc1N(C1COCC1)C([C@H]1CC[C@H](C)CC1)=O)=O RKYCZSRLFGEFGM-VIQNHZKISA-N 0.000 description 1
- SCNYAVYZKXLAJS-AQYVVDRMSA-N CCOC(c1c[n](-c(cc2)ccc2-c2n[n]3c(Cl)cccc3c2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2n[n]3c(Cl)cccc3c2)nc1N(C(C)C)C([C@H]1CC[C@H](C)CC1)=O)=O SCNYAVYZKXLAJS-AQYVVDRMSA-N 0.000 description 1
- LFXICWBOLCNGQE-ZQRQZVKFSA-N CCOC(c1c[n](-c(cc2)ccc2-c2nc(nccc3)c3[s]2)nc1N(C(C)C)C(C1C=C[C@H](C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c(cc2)ccc2-c2nc(nccc3)c3[s]2)nc1N(C(C)C)C(C1C=C[C@H](C)CC1)=O)=O LFXICWBOLCNGQE-ZQRQZVKFSA-N 0.000 description 1
- ZFFRCGQPGJKBCZ-UHFFFAOYSA-N CCOC(c1c[n](-c2ccc(-c3c(C)[o]nc3C)c(Cl)c2)nc1N(C1CCOCC1)C(C1CCC(C)CC1)=O)=O Chemical compound CCOC(c1c[n](-c2ccc(-c3c(C)[o]nc3C)c(Cl)c2)nc1N(C1CCOCC1)C(C1CCC(C)CC1)=O)=O ZFFRCGQPGJKBCZ-UHFFFAOYSA-N 0.000 description 1
- OJCIOXHXRLDQME-UHFFFAOYSA-N CCOC(c1c[n](Cc2ccccc2)nc1NC1CCCC1)=O Chemical compound CCOC(c1c[n](Cc2ccccc2)nc1NC1CCCC1)=O OJCIOXHXRLDQME-UHFFFAOYSA-N 0.000 description 1
- VUHOQWCDBXCPPW-MEMLXQNLSA-N C[C@H](CC1)CC[C@@H]1C(N(C1CCOCC1)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1N(Cc1ccccc11)C1=O)=O Chemical compound C[C@H](CC1)CC[C@@H]1C(N(C1CCOCC1)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1N(Cc1ccccc11)C1=O)=O VUHOQWCDBXCPPW-MEMLXQNLSA-N 0.000 description 1
- PBHSKNOEECJUDG-JWSDMNKESA-N C[C@H](CC1)CC[C@@H]1C(N(C1COCC1)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1c[n](cccc2)c2n1)=O Chemical compound C[C@H](CC1)CC[C@@H]1C(N(C1COCC1)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1c[n](cccc2)c2n1)=O PBHSKNOEECJUDG-JWSDMNKESA-N 0.000 description 1
- MMGLMLSAUKDZID-IRJFHVNHSA-N C[C@H](CC1)CC[C@@H]1C(N(Cc1n[o]nc1C)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1n[n](cccn2)c2c1)=O Chemical compound C[C@H](CC1)CC[C@@H]1C(N(Cc1n[o]nc1C)c(c(C(O)=O)c1)n[n]1-c(cc1)ccc1-c1n[n](cccn2)c2c1)=O MMGLMLSAUKDZID-IRJFHVNHSA-N 0.000 description 1
- KHBDZOISBUHLLR-UHFFFAOYSA-N Cc(cnc1c2)c[n]1nc2-c(cc1)ccc1Br Chemical compound Cc(cnc1c2)c[n]1nc2-c(cc1)ccc1Br KHBDZOISBUHLLR-UHFFFAOYSA-N 0.000 description 1
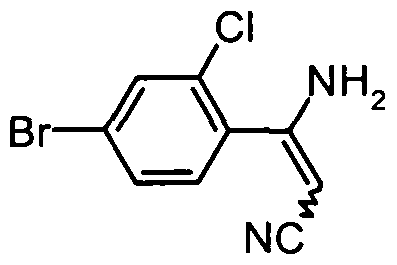
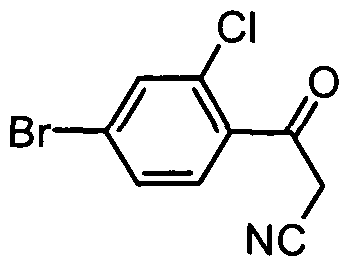
- VIPPLCMBACIPKZ-UHFFFAOYSA-N N#CCC(c(c(Cl)c1)ccc1Br)=O Chemical compound N#CCC(c(c(Cl)c1)ccc1Br)=O VIPPLCMBACIPKZ-UHFFFAOYSA-N 0.000 description 1
- RVBGKMVEVJJIPC-UHFFFAOYSA-N Nc1cc([o]c(-c(cc2)ccc2Br)c2)c2cc1 Chemical compound Nc1cc([o]c(-c(cc2)ccc2Br)c2)c2cc1 RVBGKMVEVJJIPC-UHFFFAOYSA-N 0.000 description 1
- LMHYHFRRKPCHSL-UHFFFAOYSA-N Nc1ccc[n]2c1nc(-c(cc1)ccc1Br)c2 Chemical compound Nc1ccc[n]2c1nc(-c(cc1)ccc1Br)c2 LMHYHFRRKPCHSL-UHFFFAOYSA-N 0.000 description 1
- RYVFNBCWCZCHSC-UHFFFAOYSA-N O=C(c1ccccc1C1)N1c(cc1)ccc1Br Chemical compound O=C(c1ccccc1C1)N1c(cc1)ccc1Br RYVFNBCWCZCHSC-UHFFFAOYSA-N 0.000 description 1
- OEXUGYKRHKIWDS-MXVIHJGJSA-N OC(c1c[n](-c(cc2)ccc2-c2n[n](cccn3)c3c2)nc1N(C1CCOCC1)C([C@H](CC1)CC[C@@H]1C(F)(F)F)=O)=O Chemical compound OC(c1c[n](-c(cc2)ccc2-c2n[n](cccn3)c3c2)nc1N(C1CCOCC1)C([C@H](CC1)CC[C@@H]1C(F)(F)F)=O)=O OEXUGYKRHKIWDS-MXVIHJGJSA-N 0.000 description 1
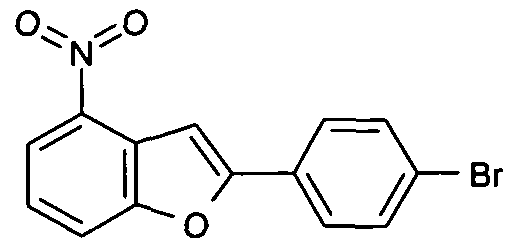
- URLGIBYHHSRMLZ-UHFFFAOYSA-N [O-][N+](c1ccc2[o]c(-c(cc3)ccc3Br)cc2c1)=O Chemical compound [O-][N+](c1ccc2[o]c(-c(cc3)ccc3Br)cc2c1)=O URLGIBYHHSRMLZ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing aromatic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/10—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D493/00—Heterocyclic compounds containing oxygen atoms as the only ring hetero atoms in the condensed system
- C07D493/02—Heterocyclic compounds containing oxygen atoms as the only ring hetero atoms in the condensed system in which the condensed system contains two hetero rings
- C07D493/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
Definitions
- the present invention relates to novel 4-carboxy pyrazole derivatives useful as anti-viral agents. Specifically, the present invention involves novel inhibitors of Hepatitis C Virus (HCV) replication.
- HCV Hepatitis C Virus
- HCV infection is responsible for 40-60% of all chronic liver disease and 30% of all liver transplants.
- Chronic HCV infection accounts for 30% of all cirrhosis, end-stage liver disease, and liver cancer in the U.S. The CDC estimates that the number of deaths due to
- HCV will minimally increase to 38,000/year by the year 2010.
- Alpha-interferon (alone or in combination with ribavirin) has been widely used since its approval for treatment of chronic HCV infection.
- adverse side effects are commonly associated with this treatment: flu-like symptoms, leukopenia, thrombocytopenia, depression from interferon, as well as anemia induced by ribavirin (Lindsay, K.L. (1997) Hepatology 26 (suppl 1 ): 71S-77S).
- HCV post-transfusion non A, non-B hepatitis
- NANBH non-B hepatitis
- this virus was assigned as a new genus in the Flaviviridae family.
- flaviviruses e.g. yellow fever virus and Dengue virus types 1-4
- pestiviruses e.g.
- HCV bovine viral diarrhea virus, border disease virus, and classic swine fever virus
- the HCV genome is approximately 9.6 kilobases (kb) with a long, highly conserved, noncapped 5 1 nontranslated region (NTR) of approximately 340 bases which functions as an internal ribosome entry site (IRES) (Wang CY et al 'An RNA pseudoknot is an essential structural element of the internal ribosome entry site located within the hepatitis C virus 5' noncoding region' RNA- A Publication of the RNA Society. 1 (5): 526-537, 1995 JuI.). This element is followed by a region which encodes a single long open reading frame (ORF) encoding a polypeptide of -3000 amino acids comprising both the structural and nonstructural viral proteins.
- ORF long open reading frame
- this RNA Upon entry into the cytoplasm of the cell, this RNA is directly translated into a polypeptide of -3000 amino acids comprising both the structural and nonstructural viral proteins.
- This large polypeptide is subsequently processed into the individual structural and nonstructural proteins by a combination of host and virally-encoded proteinases (Rice, CM. (1996) in B.N. Fields, D.M.Knipe and P.M. Howley (eds) Virology 2 nd Edition, p931-960; Raven Press, N.Y.).
- 3' NTR which roughly consists of three regions: an - 40 base region which is poorly conserved among various genotypes, a variable length poly(U)/polypyrimidine tract, and a highly conserved 98 base element also called the "3 1 X-tail" (Kolykhalov, A. et al (1996) J. Virology 70:3363-3371; Tanaka, T. et al (1995) Biochem Biophys. Res. Commun. 215:744-749; Tanaka, T. et al (1996) J. Virology 70:3307-3312; Yamada, N. et al (1996) Virology 223:255-261).
- the 3 1 NTR is predicted to form a stable secondary structure which is essential for HCV growth in chimps and is believed to function in the initiation and regulation of viral RNA replication.
- the NS5B protein (591 amino acids, 65 kDa) of HCV (Behrens, S.E. et al (1996) EMBO J. 15:12-22), encodes an RNA-dependent RNA polymerase (RdRp) activity and contains canonical motifs present in other RNA viral polymerases.
- the NS5B protein is fairly well conserved both intra-typically (-95-98% amino acid (aa) identity across 1b isolates) and inter-typically (-85% aa identity between genotype 1a and 1b isolates).
- the essentiality of the HCV NS5B RdRp activity for the generation of infectious progeny virions has been formally proven in chimpanzees (A. A. Kolykhalov et al.. (2000) Journal of Virology, 74(4): 2046-2051).
- inhibition of NS5B RdRp activity is predicted to be useful to treat HCV infection.
- the present invention involves novel 4-carboxy pyrazole compounds represented hereinbelow, pharmaceutical compositions comprising such compounds and use of the compounds in treating viral infection, especially HCV infection.
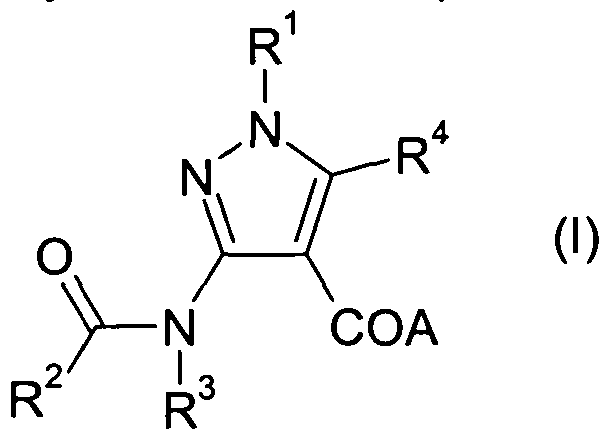
- the present invention provides at least one chemical entity chosen from compounds of Formula (I) :
- A represents hydroxy
- R 1 represents -R x -R ⁇ ;
- R x represents phenyl (optionally substituted by halo, methyl, ethyl, methoxy or trifluoromethyl) or a 5 or 6-membered heteroaryl bonded through a ring carbon atom to the nitrogen atom of the pyrazole;
- R ⁇ represents heteroaryl, bonded such that when R x is phenyl, R y is in the para-position;
- R 2 represents phenyl substituted by one or more substituents selected from -C 1-6 alkyl, halo, - OR A , -SR A , -C(O)NR B R C , -C(O)R 0 , -CO 2 H, -CO 2 R 0 , -NR B R C , -NR E C(O)R D , -NR E CO 2 R D , - NR E C(O)NR F R G , -SO 2 NR F R G , -SO 2 R 0 , nitro, cyano, and heterocyclyl, or two substituents on neighbouring carbon atoms of the phenyl ring together form a 4-, 5- or 6-membered ring containing one or more heteroatoms selected from O, S and N; or R 2 represents C 5 .
- R 3 represents heterocyclyl; or phenyl optionally substituted by one or more substituents selected from -C 1-6 alkyl, halo, -OR A , -SR A , -C(O)NR B R C , -C(O)R 0 , -CO 2 H, -CO 2 R 0 , -NR B R C , -
- R 3 represents -C 1-6 alkyl optionally substituted by one or more substituents selected from -0R A , -SR A , -C(O)NR B R C , -C(O)R 0 , -CO 2 H, -CO 2 R 0 , -NR B R C , -
- R 4 represents hydrogen
- R A represents hydrogen, -C 1-6 alkyl, arylalkyl, heteroarylalkyl, aryl, heterocyclyl or heteroaryl;
- R B and R c independently represent hydrogen, -C 1-6 alkyl, aryl, heterocyclyl or heteroaryl; or R B and R c together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
- R° is selected from the group consisting of -C 1-6 alkyl, aryl, heterocyclyl, heteroaryl, arylalkyl, and heteroarylalkyl;
- R E represents hydrogen or -C ⁇ alkyl
- R F and R G are independently selected from the group consisting of hydrogen, -C 1-6 alkyl, aryl, heteroaryl, arylalkyl, and heteroarylalkyl; or R F and R G together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
- references herein to therapy and/or treatment includes, but is not limited to prevention, retardation, prophylaxis, therapy and cure of the disease. It will further be appreciated that references herein to treatment or prophylaxis of HCV infection includes treatment or prophylaxis of HCV-associated disease such as liver fibrosis, cirrhosis and hepatocellular carcinoma.
- a method for the treatment of a human or animal subject with viral infection, particularly HCV infection which method comprises administering to said human or animal subject an effective amount of at least one chemical entity chosen from compounds of Formula (I) and pharmaceutically acceptable salts, solvates and esters thereof.
- the chemical entities of the present invention may contain one or more asymmetric carbon atoms and may exist in racemic, diastereoisomeric, and optically active forms. All of these racemic compounds, enantiomers and diastereoisomers are contemplated to be within the scope of the present invention.
- R x represents thienyl; pyridinyl; pyridazinyl; or phenyl optionally substituted by halo, methyl, methoxy or trifluoromethyl.
- R x represents 2,5 thienyl.
- R x represents phenyl optionally substituted by halo or methyl.
- R x represents phenyl, 3-chlorophenyl, or 3-methylphenyl, for example phenyl or 3- chlorophenyl.
- R x represents a heteroaryl group, that group is bonded through one of its ring carbon to the R ⁇ group.
- R ⁇ represents a heteroaryl group containing 2 fused rings.
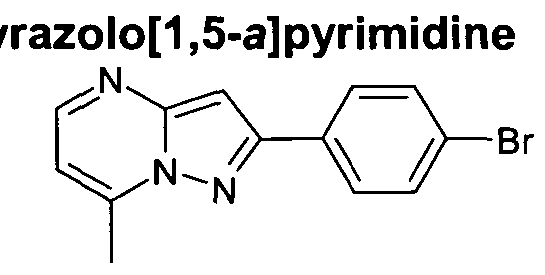
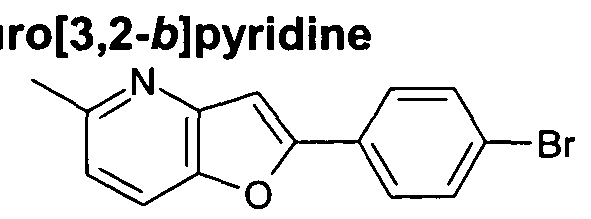
- R ⁇ represents optionally substituted pyrazolopyrimidine, furopyridine, imidazopyridine, benzoxazole, imidazothiazole, benzofuran or pyrazolopyridine, for example optionally substituted pyrazolopyrimidine, furopyridine, imidazopyridine, benzoxazole, or imidazothiazole
- R ⁇ represents optionally substituted pyrazolo[1 ,5- a]pyrimidin-2-yl, furo[3,2-b]pyridin-2-yl, imidazo[1 ,2-a]pyridin-2-yl, benzoxazol-2-yl, imidazo[2,1-6][1 ,3]thiazol-6-yl, 4-furo[3,2- ⁇ ]pyridin-6-yl, 4-furo[3,2-b]pyridin-5-yl, 1- benzofuran-2-
- R ⁇ represents optionally substituted pyrazolo[1 ,5-a]pyrimidin-2-yl, furo[3,2- ⁇ ]pyridin-2-yl, imidazo[1 ,2-a]pyridin-2-yl, benzoxazol-2-yl, or imidazo[2,1-b][1 ,3]thiazol-6-yl.
- R x represents a 6-membered heteroaryl bonded through a ring carbon atom to the nitrogen atom of the pyrazole
- R ⁇ is bonded so as to be in the para-position.
- R 2 represents phenyl substituted by one or more substituents selected from - Ci- ⁇ alkyI, halo, -OR A , -SR A , -C(O)NR B R C , -C(O)R 0 , -CO 2 H, -CO 2 R 0 , -NR B R C , -NR E C(O)R D , - NR E CO 2 R D , -NR E C(O)NR F R G , -SO 2 NR F R G , -SO 2 R 0 , nitro, cyano, and heterocyclyl; or R 2 represents C 5-7 cycloalkyl optionally substituted by one or more substitutents selected from - C 1-6 alkyl,
- R 2 represents phenyl substituted by one or more substituents selected from or halo, or R 2 represents C 6 cycloalkyl optionally substituted by one or more C 1-4 alkyl or trifluoromethyl substituents (for example one or more C 1-4 alkyl substituents).
- R 2 represents frans-4-methylcyclohexyl or trans- 4-trifluoromethylcyclohexyl, for example frans-4-methylcyclohexyl.
- R 2 represents a phenyl group substituted by two substituents on neighbouring carbon atoms of the phenyl ring and which together form a 5- or 6-membered ring (including the intervening phenyl ring carbon atoms) containing one or more heteroatoms selected from O, S and N.
- the ring is 5-membered.
- the ring contains one heteroatom.
- the heteroatom is O.
- the ring contains a carbon-carbon double bond.
- the R2 group may be a benzofuran group.
- R 3 represents heterocyclyl; or phenyl optionally substituted by one or more substituents selected from -C ⁇ alkyl, halo, -0R A , -SR A , -C(O)NR B R C , -C(O)R 0 , -CO 2 H, - CO 2 R 0 , -NR B R C , -NR E C(O)R°, -NR E CO 2 R°, -NR E C(O)NR F R G , -SO 2 NR F R G , -SO 2 R 0 , nitro, cyano, and heterocyclyl; or R 3 represents -C h alky!
- R 3 represents 1-methylethyl, 2-methylpropyl, 1-ethylpropyl, 1 ,3-dimethylbutyl, ethyl, propyl, 2-ethylbutyl, cyclopentyl, tetrahydro-2H-pyran-4-yl, tetrahydro-2/-/-pyran-4- ylmethyl, tetrahydrofuran-3-yl, 2-methoxyethyl, 2-(methoxy)-1-[(methoxy)methyl]ethyl, 1 ,3- oxazol-2-yl methyl, 2-(phenyloxy)ethyl, 1 ,3-thiazol-4-ylmethyl, (4-methyl-1 ,2,5-oxadiazol-3- yl)methyl, 2-(ethyloxy)ethyl, (2-methyl-1 ,3-thiazol-4-yl)methyl, butyl, (3,5-dimethyl-4- is
- R 3 represents 1-methylethyl, 2-methylpropyl, 1-ethylpropyl, 1 ,3-dimethylbutyl, ethyl, propyl, 2-ethylbutyl, cyclopentyl, tetrahydro-2H-pyran-4-yl, tetrahydro-2H-pyran-4-ylmethyl, tetrahydrofuran-3-yl, 2-methoxyethyl, or 2-(methoxy)-1-[(methoxy)methyl]ethyl.
- R 3 represents 1-methylethyl, ethyl, tetrahydro-2H-pyran-4-yl, tetrahydrofuran-3-yl or 2-(methoxy)-1- [(methoxy)methyl]ethyl. It is to be understood that the present invention covers all combinations of aspects, suitable, convenient and preferred groups described herein.
- acetyl refers to -C(O)CH 3 .
- acetylamino refers to -N(H)C(O)CH 3 .
- alkyl refers to an optionally substituted hydrocarbon group.
- the alkyl hydrocarbon group may be linear, branched or cyclic, saturated or unsaturated. Where the alkyl group is linear or branched, examples of such groups include methyl, ethyl, n-propyl, 1 -methylethyl (isopropyl), n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, neopentyl or hexyl and the like.
- alkyl hydrocarbon group is unsaturated, it will be understood that there will be a minimum of 2 carbon atoms in the group, for example an alkenyl or alkynyl group. Where the alkyl hydrocarbon group is cyclic, it will be understood that there will be a minimum of 3 carbon atoms in the group. In one aspect, alkyl moieties are Unless otherwise stated, optional substituents include -C h alky!
- alkenyl refers to a linear or branched hydrocarbon group containing one or more carbon-carbon double bonds. In one aspect the alkenyl group has from 2 to 6 carbon atoms. Examples of such groups include ethenyl, propenyl, butenyl, pentenyl or hexenyl and the like.
- alkynyl refers to a linear or branched hydrocarbon group containing one or more carbon-carbon triple bonds. In one aspect the alkynyl group has from 2 to 6 carbon atoms. Examples of such groups include ethynyl, propynyl, butynyl, pentynyl or hexynyl and the like.
- cycloalkyl refers to an optionally substituted, cyclic hydrocarbon group.
- the hydrocarbon group may be saturated or unsaturated, monocyclic or bridged bicyclic.
- examples of such groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl and the like.
- examples of such groups include cyclobutenyl, cyclopentenyl, cyclohexenyl, cycloheptenyl or cyclooctenyl and the like.
- the cycloalkyl group has from 5 to 7 carbon atoms.
- cycloalkyl moieties are cyclohexenyl, cyclopentenyl and cyclohexyl.
- -C ⁇ alkyl unsubstituted
- alkoxy refers to an -O-alkyl group wherein alkyl is as defined herein. Examples of such groups include methoxy, ethoxy, propoxy, butoxy, pentoxy or hexoxy and the like.
- aryl refers to an optionally substituted aromatic group with at least one ring having a conjugated pi-electron system, containing up to two conjugated or fused ring systems.
- Aryl includes carbocyclic aryl and biaryl groups, all of which may be optionally substituted.
- aryl moieties contain 6-10 carbon atoms.
- aryl moieties are unsubstituted, monosubstituted, disubstituted or trisubstituted phenyl.
- aryl substituents are selected from the group consisting of -C ⁇ alkyl, halo, -0R A , -SR A , -C(O)NR B R C , -C(O)R 0 , -CO 2 H, -CO 2 R 0 , -NR B R C , -NR E C(0)R°, -NR E CO 2 R°, -NR E S0 2 R°, -NR E C(0)NR F R G , -S0 2 NR F R G , -SO 2 R 0 , nitro, cyano, heterocyclyl, - CF 3 , -OCF 3 and phenyl.
- they may be selected from -C h alky!, halo, -0R A , -SR A , -C(O)NR 6 R 0 , -C(O)R 0 , -CO 2 H, -CO 2 R 0 , -NR B R C , -NR E C(O)R°, -NR 6 CO 2 R 0 , -NR ⁇ C(O)NR F R G , -S0 2 NR F R G , -SO 2 R 0 , nitro, cyano, heterocyclyl, -CF 3 , -OCF 3 and phenyl.
- arylalkyl refers to an aryl group attched to the parent molecular moiety through an alkyl group.
- carbonyl refers to -C(O)-.
- cyano refers to -CN.
- halogen or halo refer to a fluorine, chlorine, bromine or iodine atom. References to "fluoro”, “chloro”, “bromo” or “iodo” should be construed accordingly.
- heteroaryl refers to an optionally substituted, 5, 6, 8, 9 or 10 membered, aromatic group comprising one to four heteroatoms selected from
- heteroaryl moieties are unsubstituted, monosubstituted, disubstituted or trisubstituted (where applicable) pyridine, pyrazine, thiazole, thiophene, furan, oxadiazole, oxazole, isoxazole, pyrimidine, pyridazine, benzodioxole, benzofuran, benzodioxin, indole, benzimidazole, benzofuran, indole, indazole, isoindole, benzothiophene, benzothiazole, benzothiadiazine, benzoxazole, benzisoxazole, benzisothiazole,
- Each heteroaryl group may be attached at any ring carbon or may be attached through nitrogen when the nitrogen is part of a 5-membered ring.
- Each heteroaryl group may be optionally substituted on a carbon atom by one or more substituents.
- heteroaryl substituents are selected from the group consisting of -C 1-6 alkyl, halo, -OR A , -SR A , -C(O)NR B R C , -C(O)R D , -CO 2 R 0 , -NR B R C , -NR E C(O)R D , -NR E CO 2 R D , -NR E SO 2 R D , -NR E C(O)NR F R G , -SO 2 NR F R G , -SO 2 R 0 , oxo, nitro, cyano, heterocyclyl, -CF 3 and phenyl.
- -C 1-6 alkyl halo, -OR A , -SR A , -C(O)NR 6 R 0 , -C(O)R 0 , -CO 2 R 0 , -NR B R C , -NR E C(O)R°, -NR E CO 2 R°, -NR E C(O)NR F R G , -SO 2 NR F R G , -SO 2 R 0 , oxo, nitro, cyano, heterocyclyl, -CF 3 and phenyl.
- heteroarylalkyl refers to a heteroaryl group attached to the parent molecular moiety through an alkyl group.
- heterocyclic and “heterocyclyl” refer to an optionally substituted, 5 or 6 membered, saturated or partially saturated, cyclic group containing 1 or 2 heteroatoms selected from N, optionally substituted by hydrogen, -C 1-6 alkyl, -C(O)R 0 , -C(O)NR B R C , -C(O)OR 4 , -SO 2 R 0 , aryl or heteroaryl; O; and S, optionally substituted by one or two oxygen atoms.
- Ring carbon atoms may be optionally substituted by -C 1-6 alkyl, -0R E , -C(O)R 0 , or -SO 2 R 0 .
- heterocyclic moieties are unsubstituted or monosubstituted tetrahydro-2H-pyran-4-yl, piperidinyl and tetrahydrofuran- 3-yl.
- nitro refers to -NO 2 .
- Ac refers to “acetyl”
- Et refers to “ethyl”
- iPr refers to “isopropyl”
- Me refers to “methyl”
- OBn refers to "benzyloxy”
- Ph refers to "phenyl”.
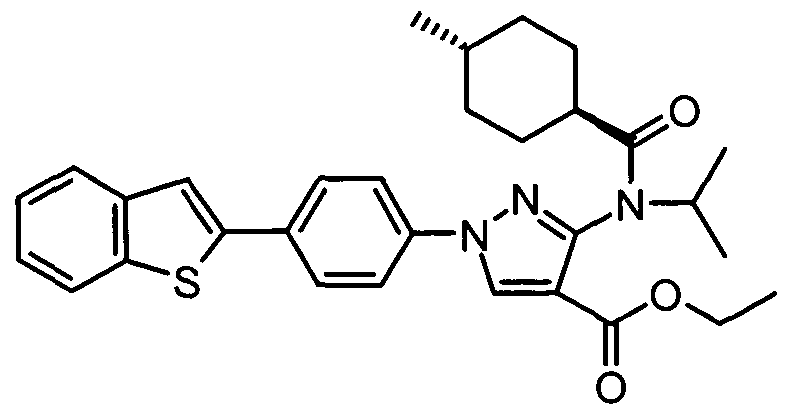
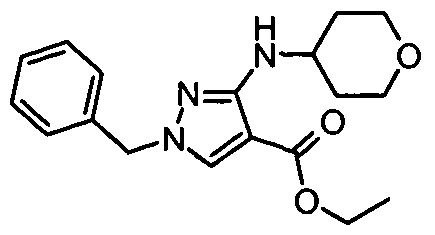
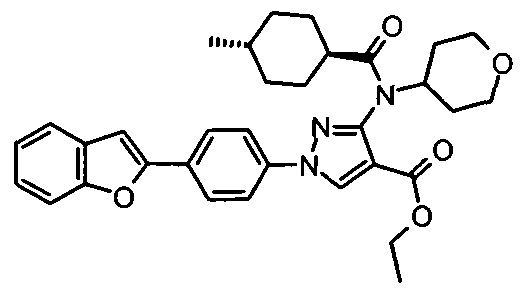
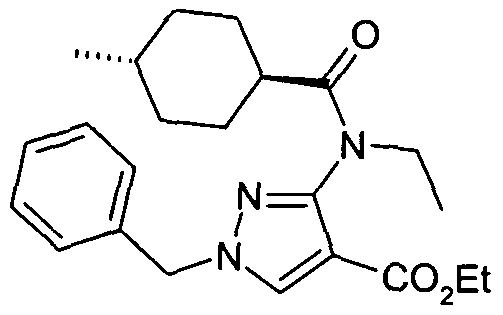
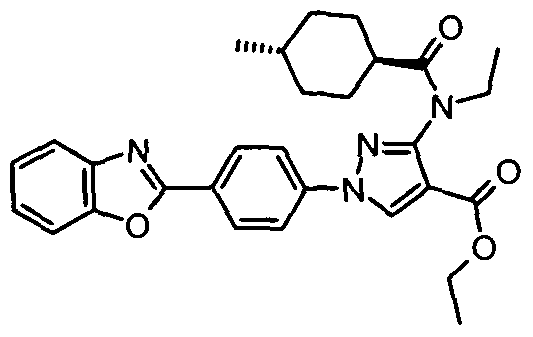
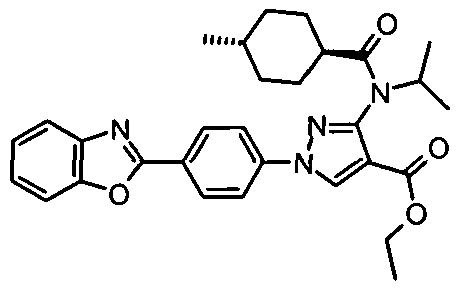
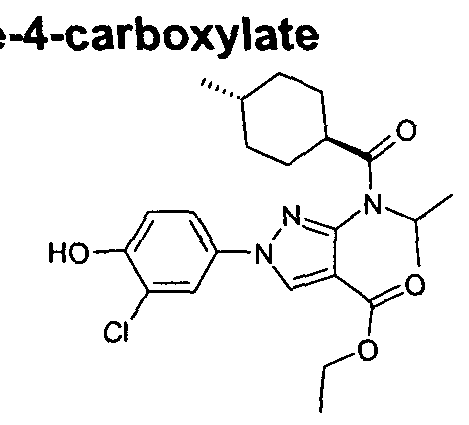
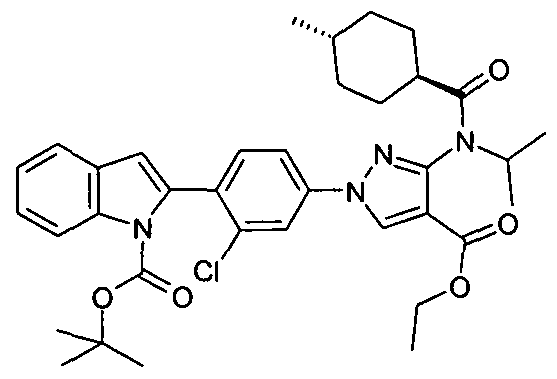
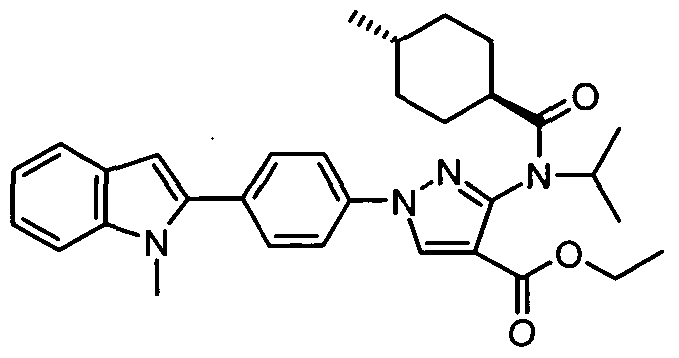
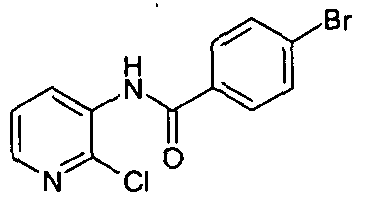
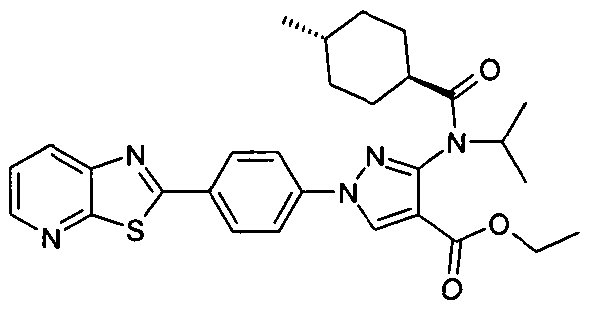
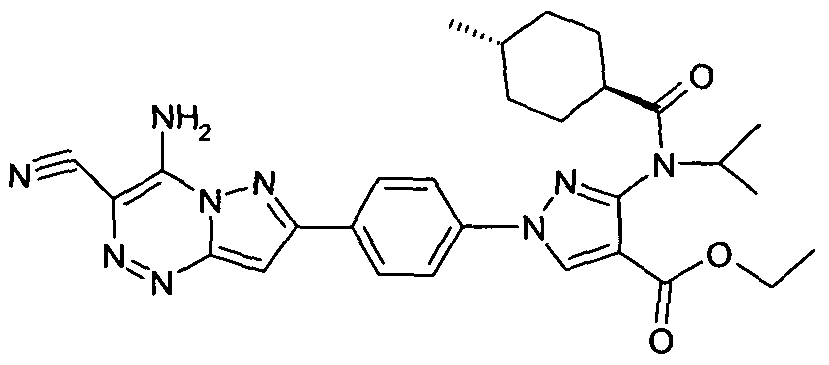
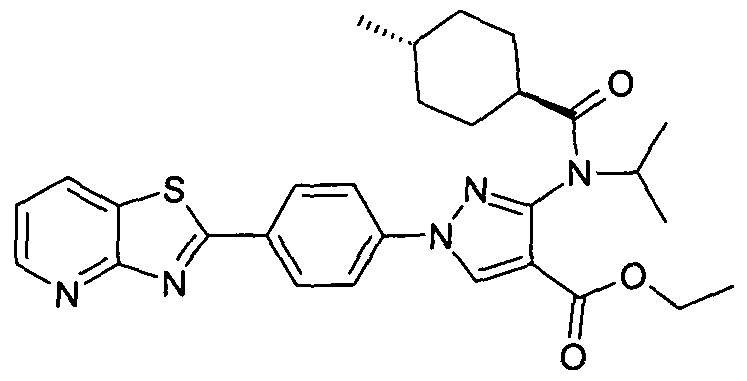
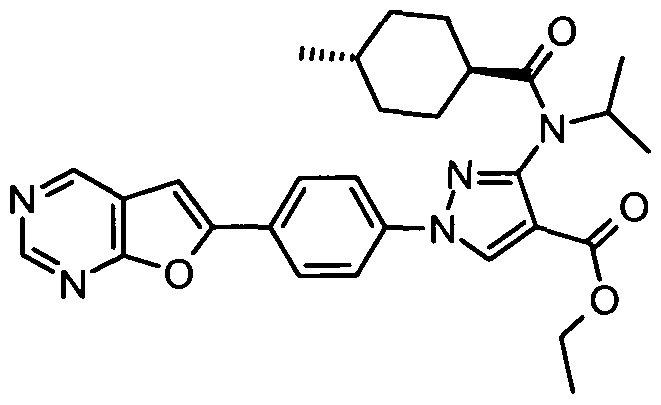
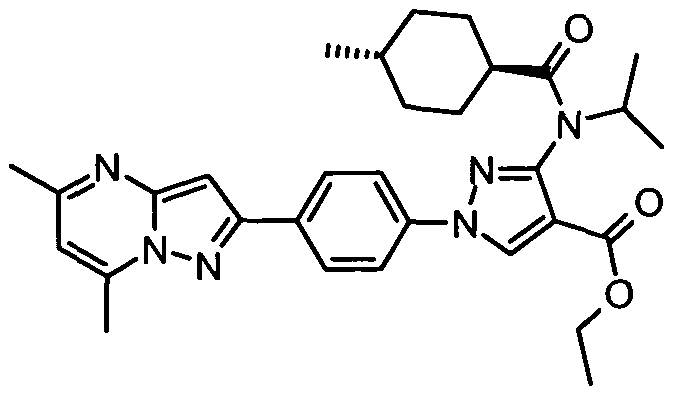
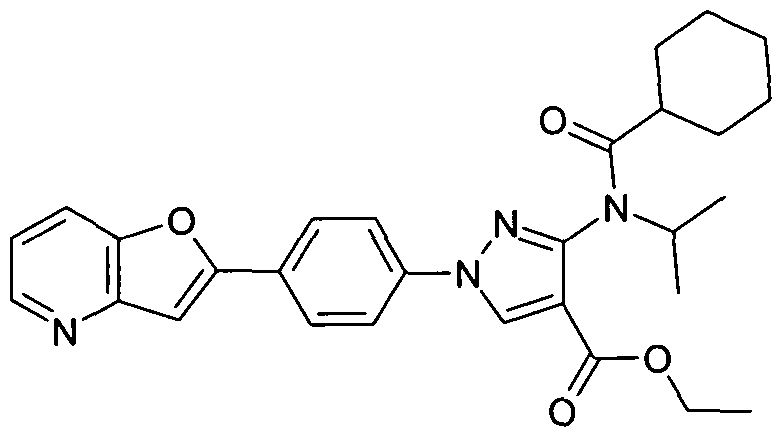
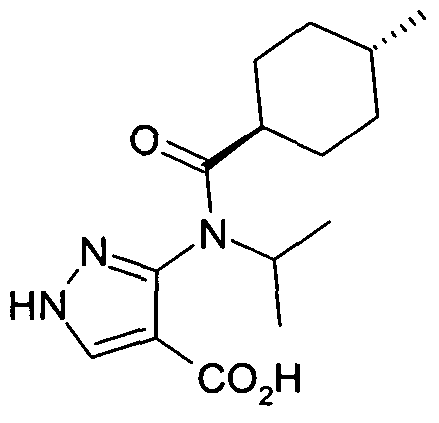
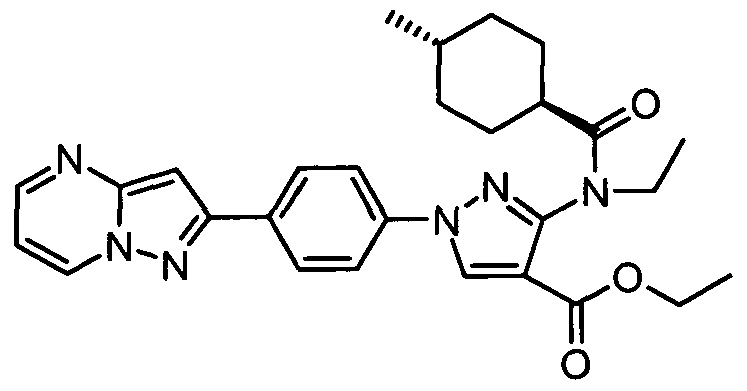
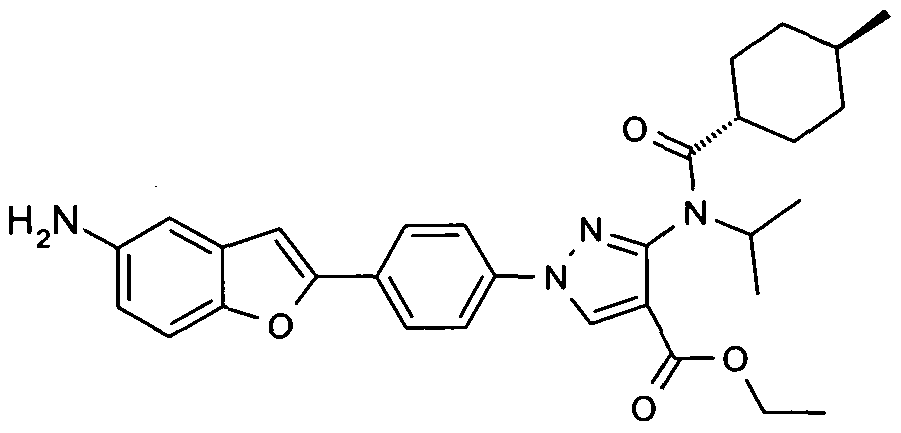
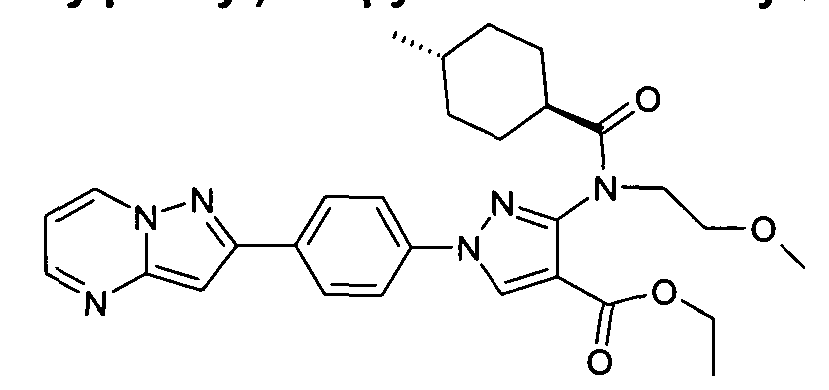
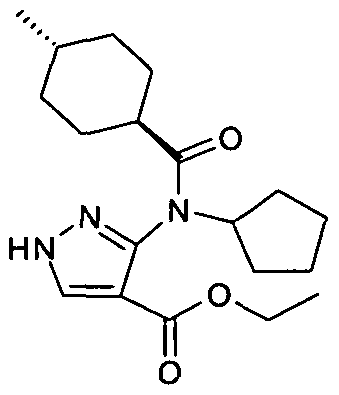
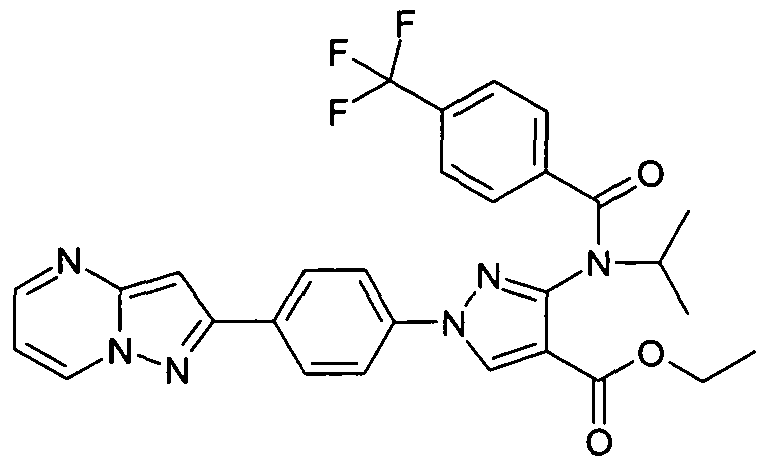
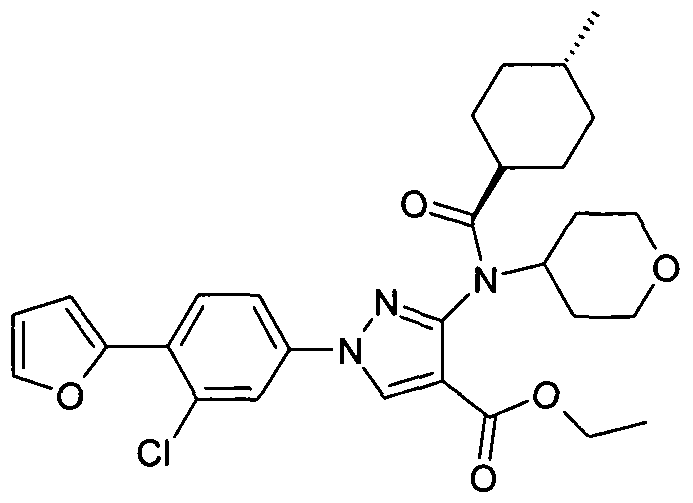
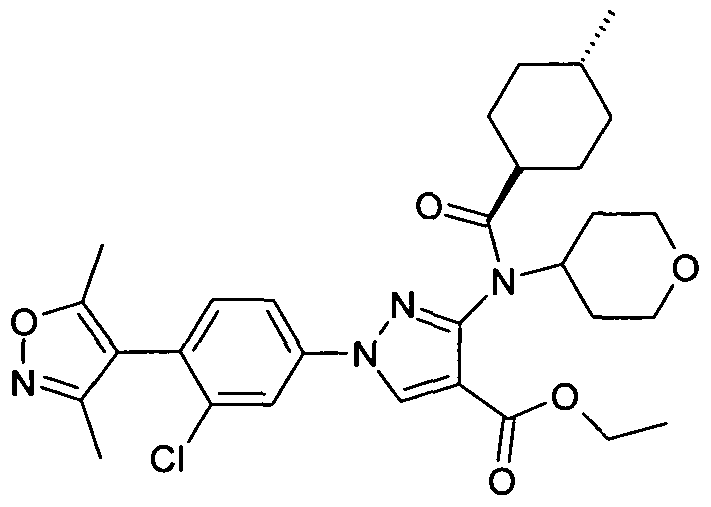
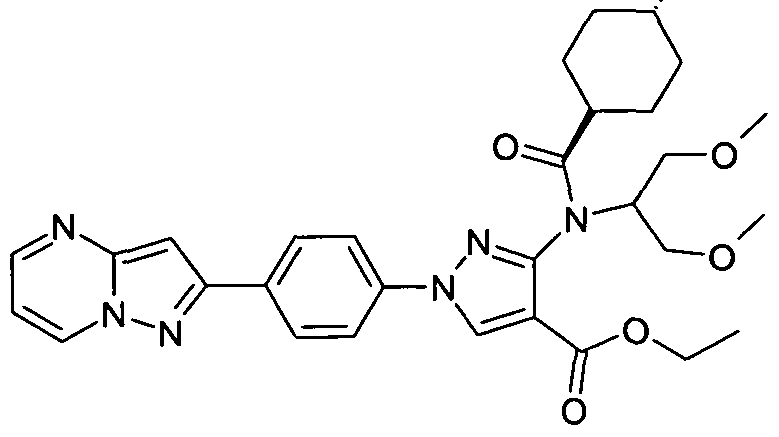
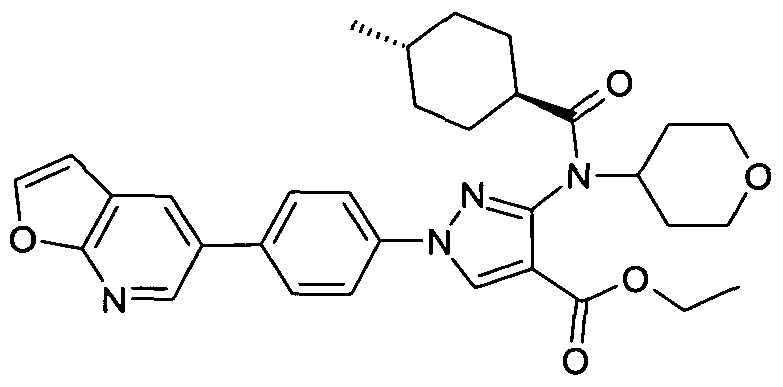
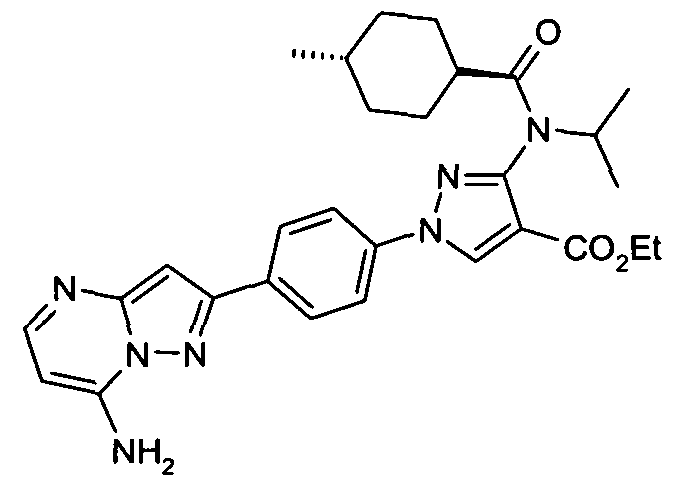
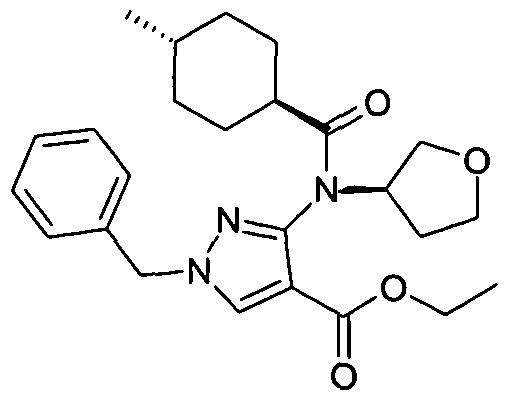
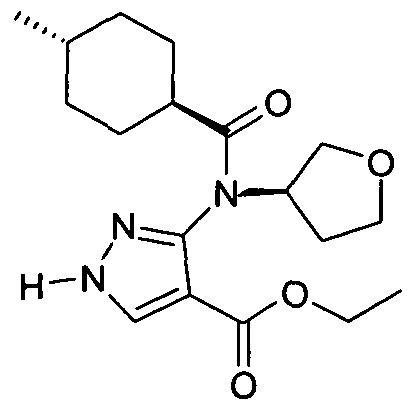
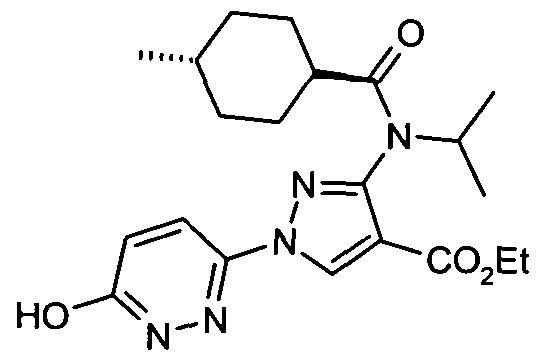
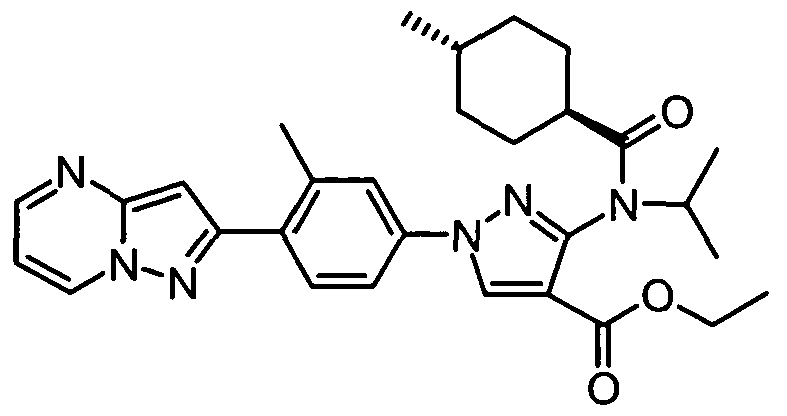
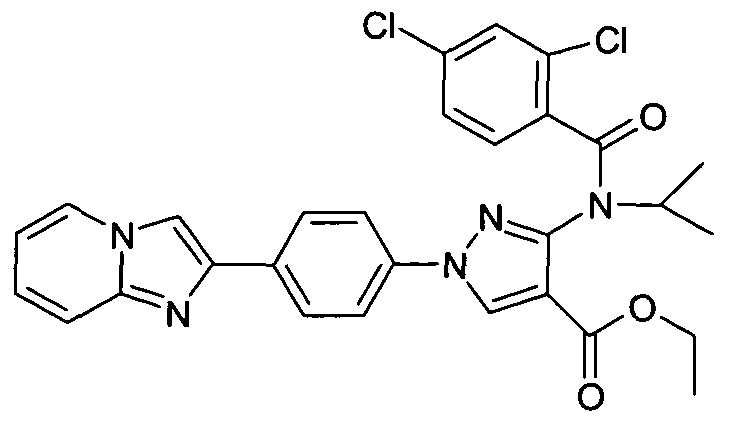
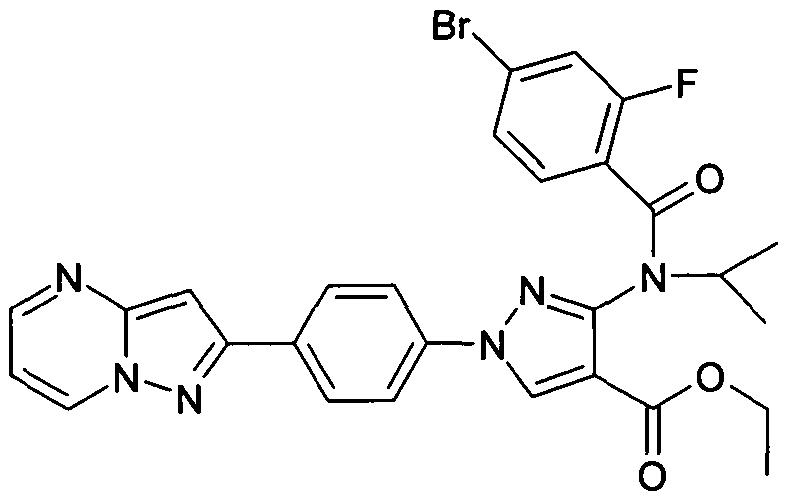
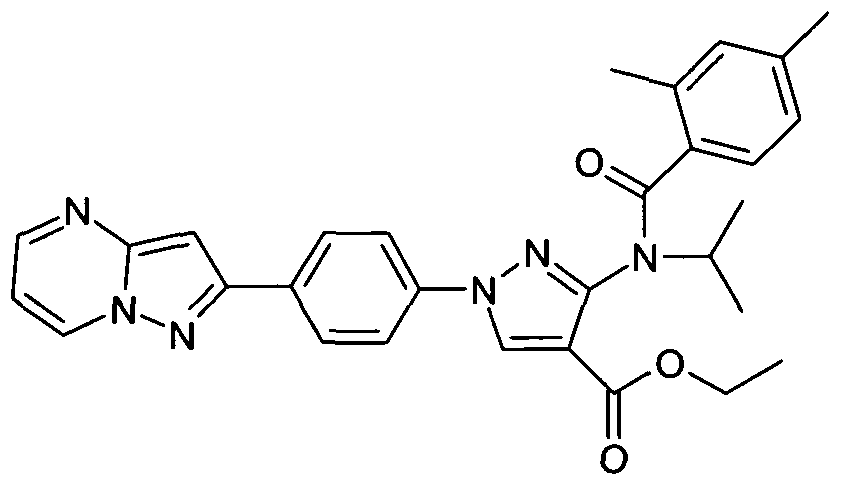
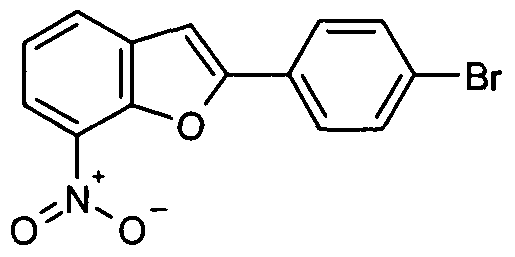
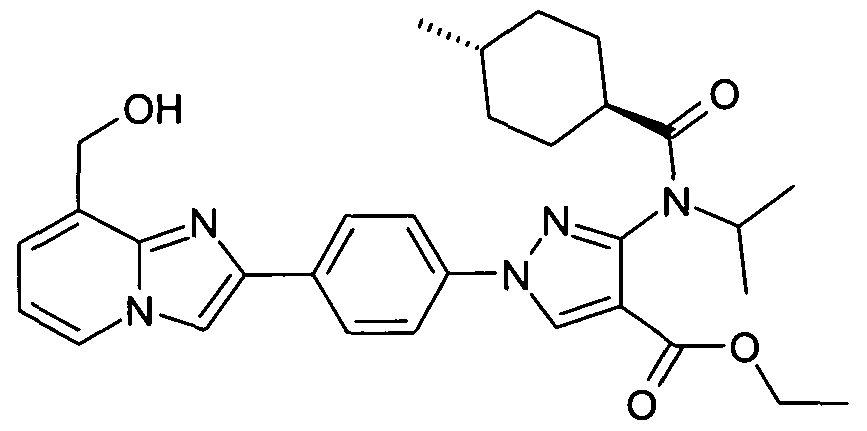
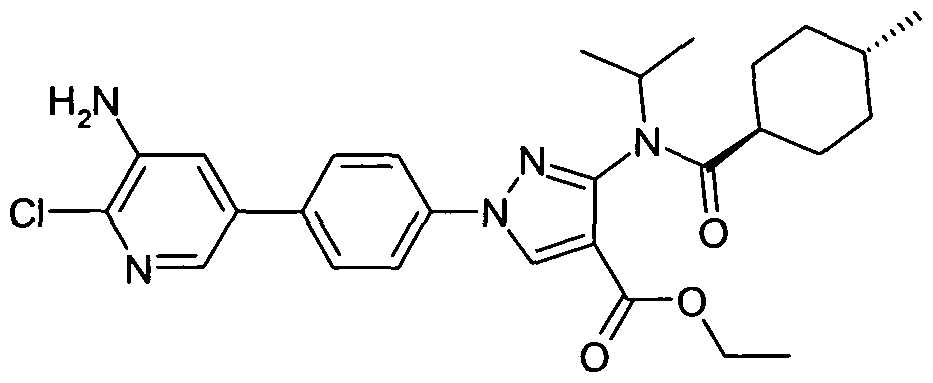
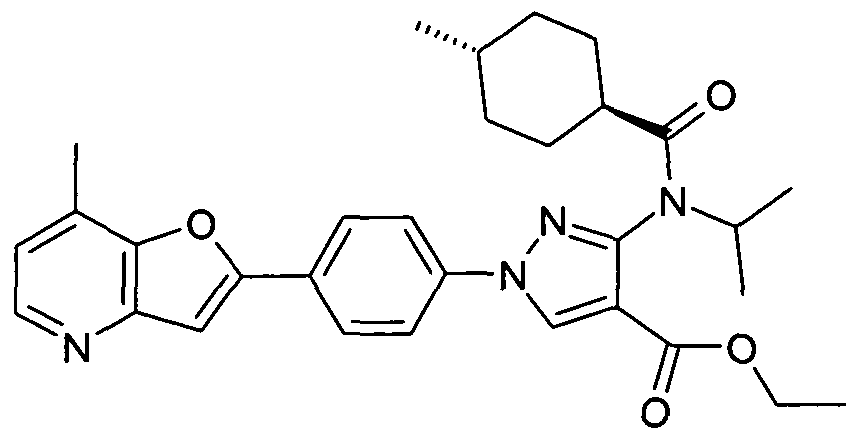
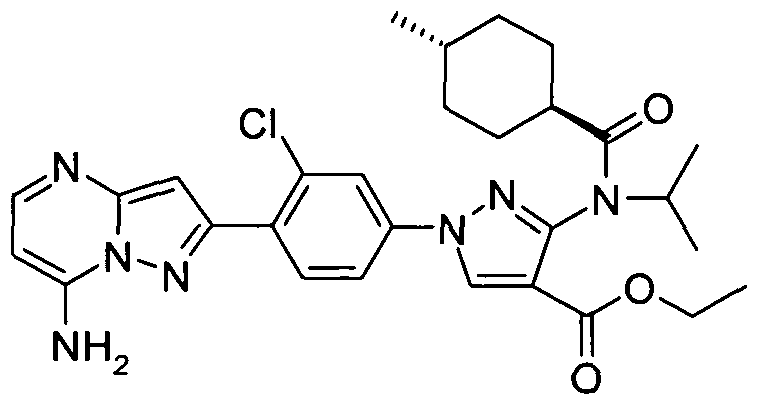
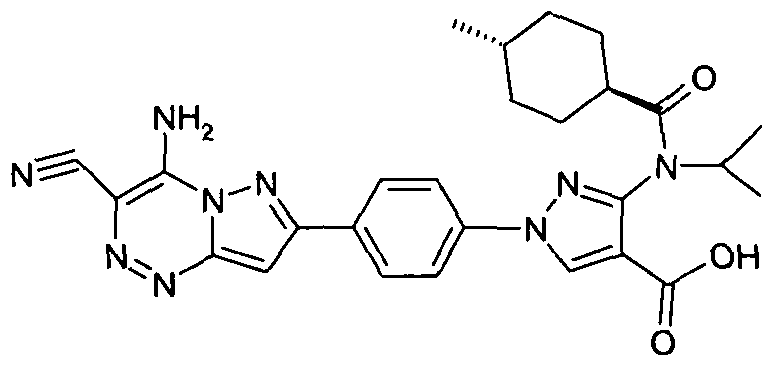
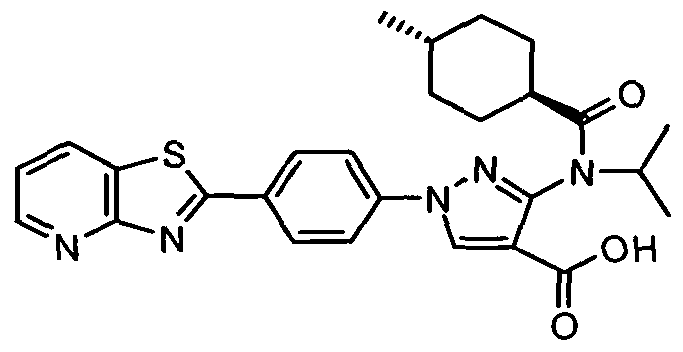
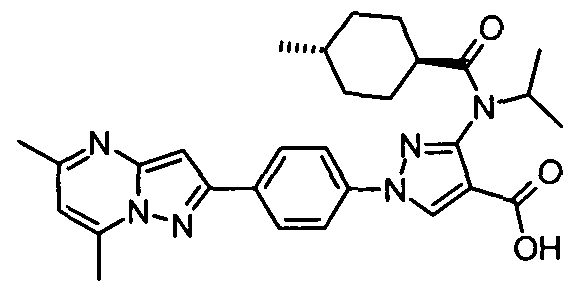
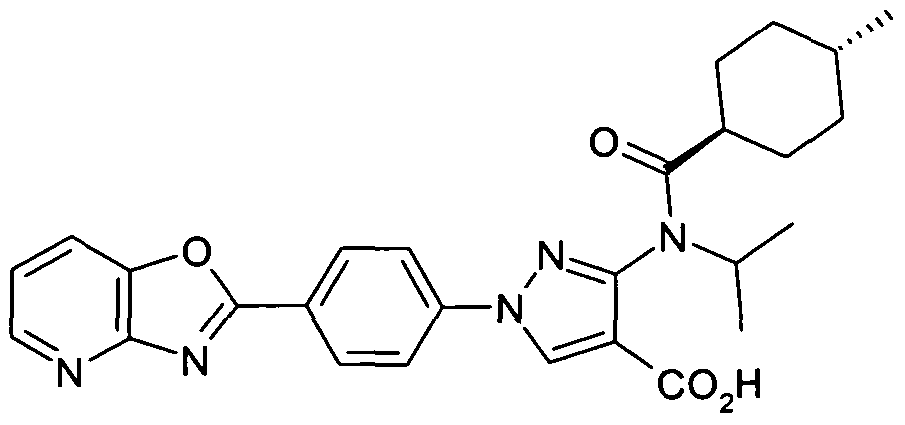
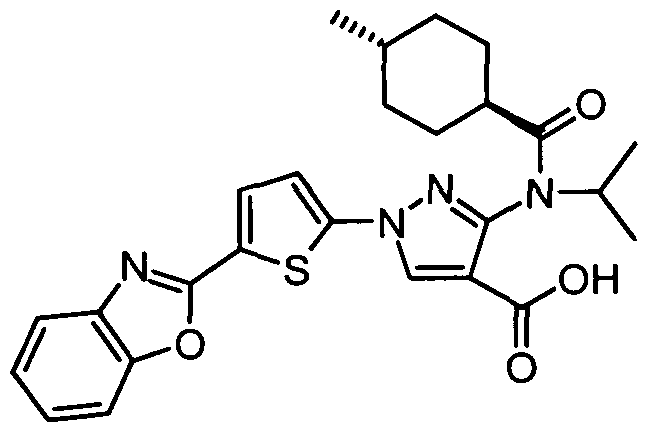
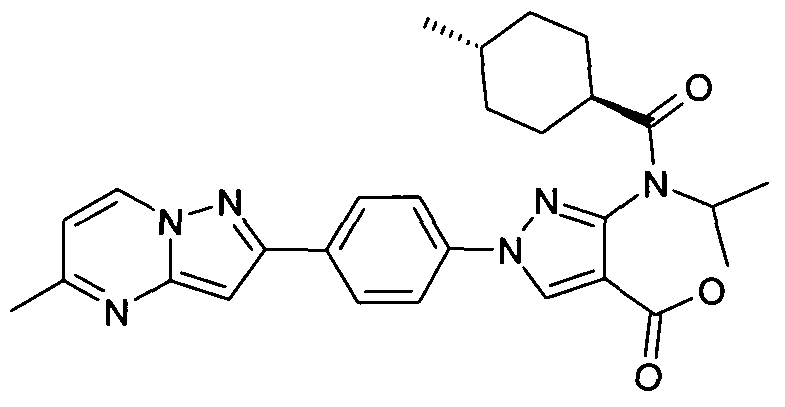
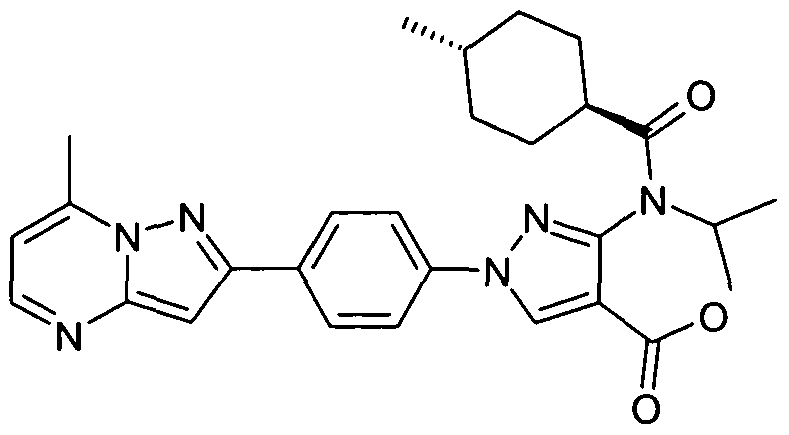
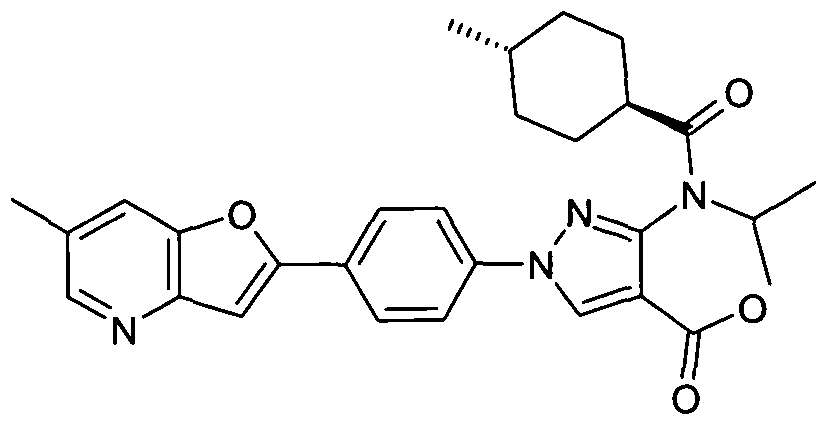
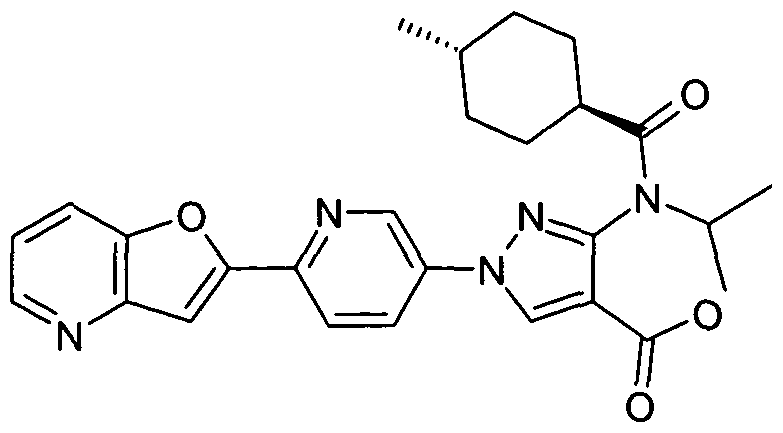
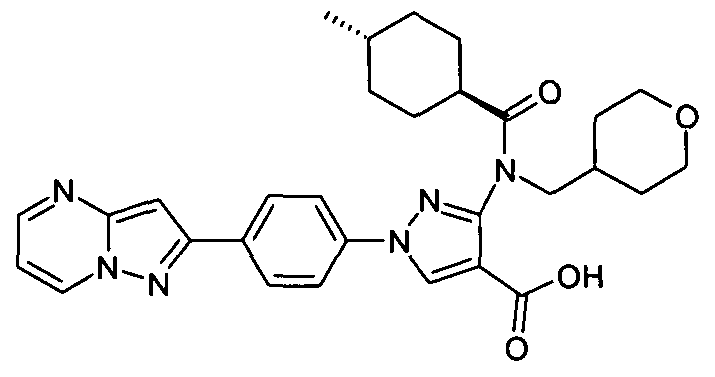
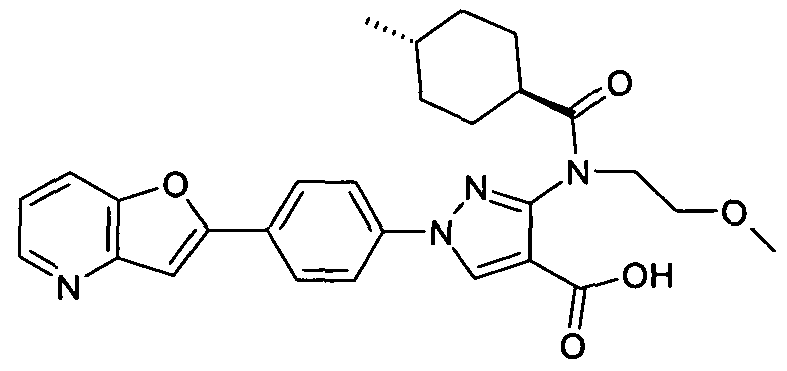
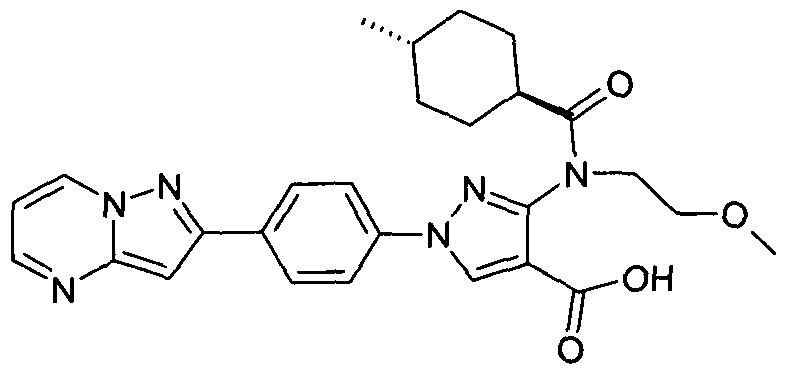
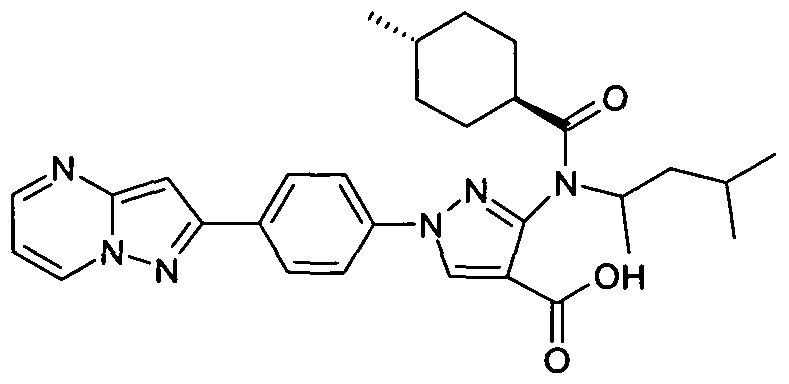
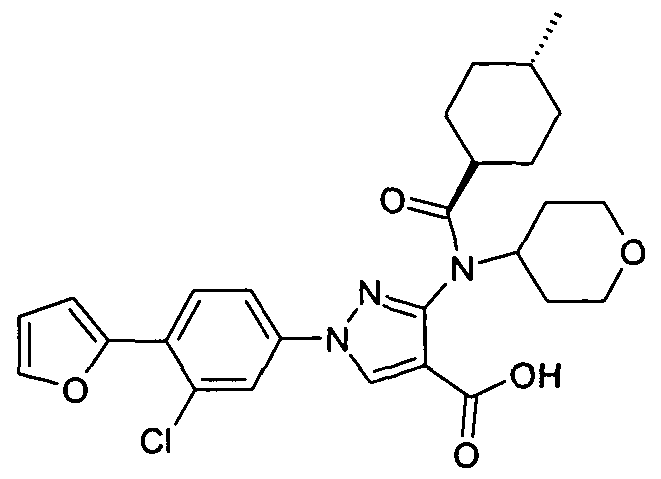
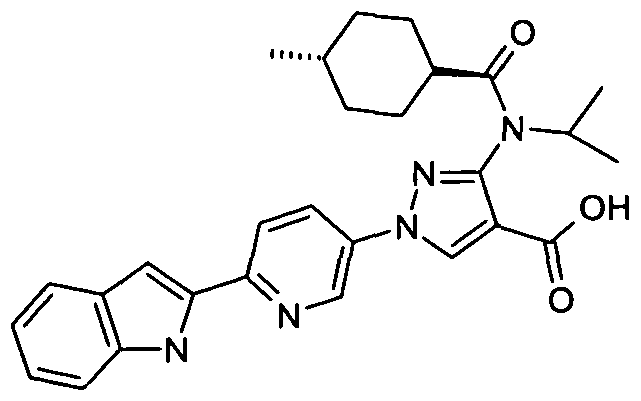
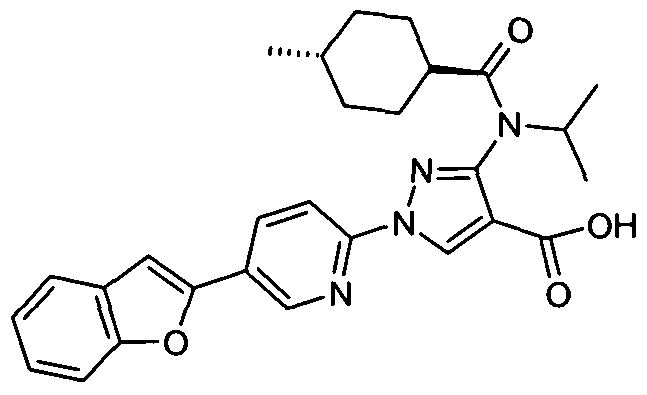
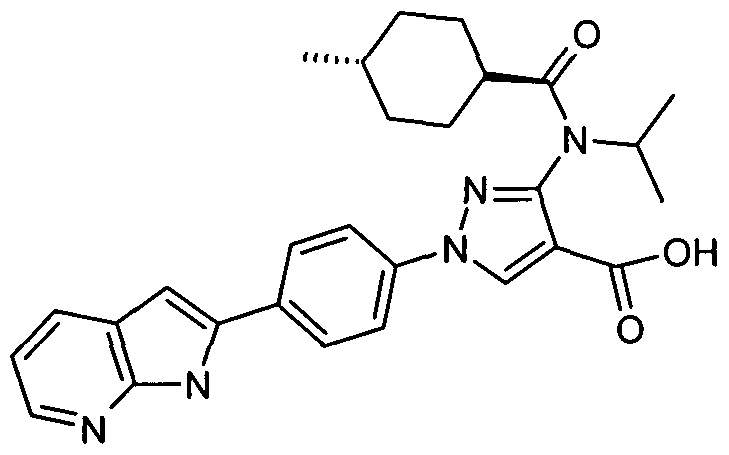
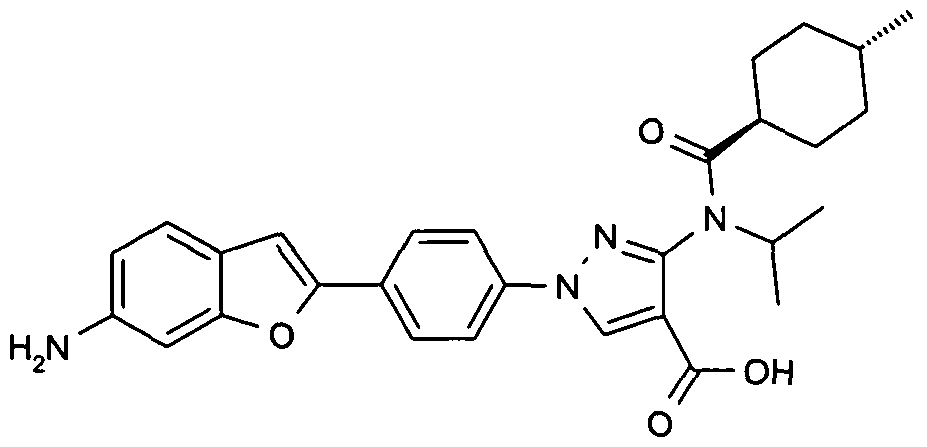
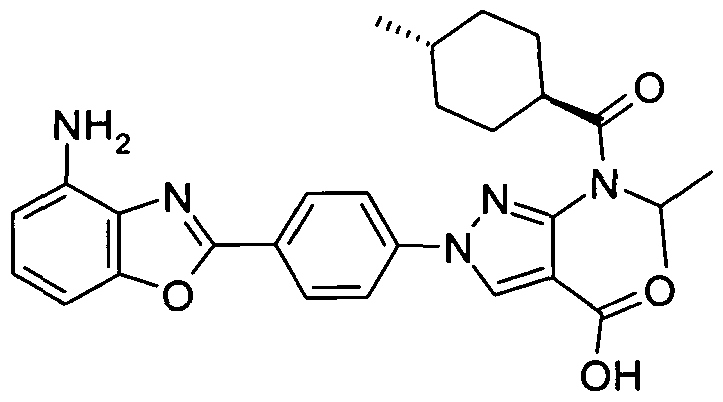
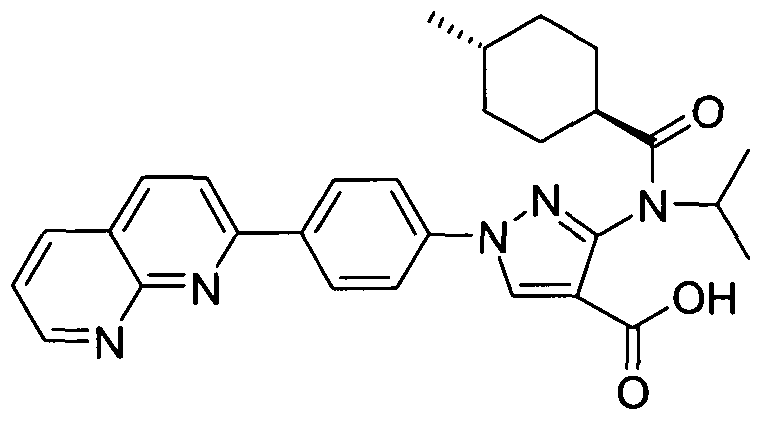
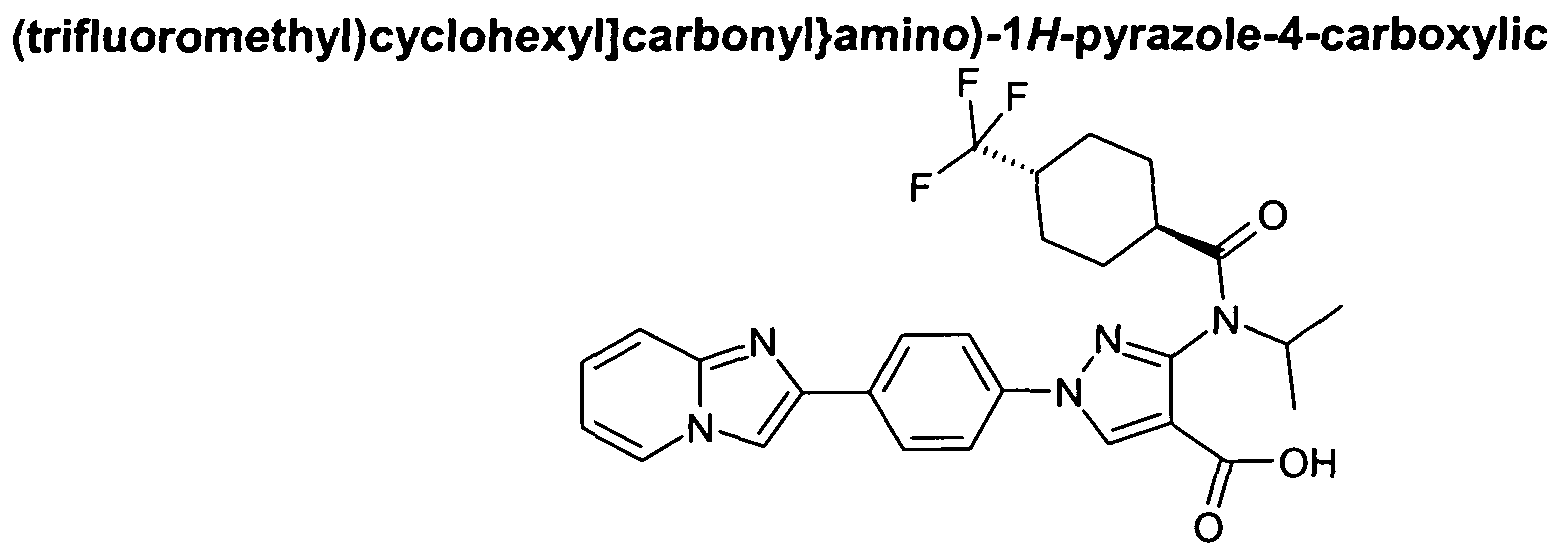
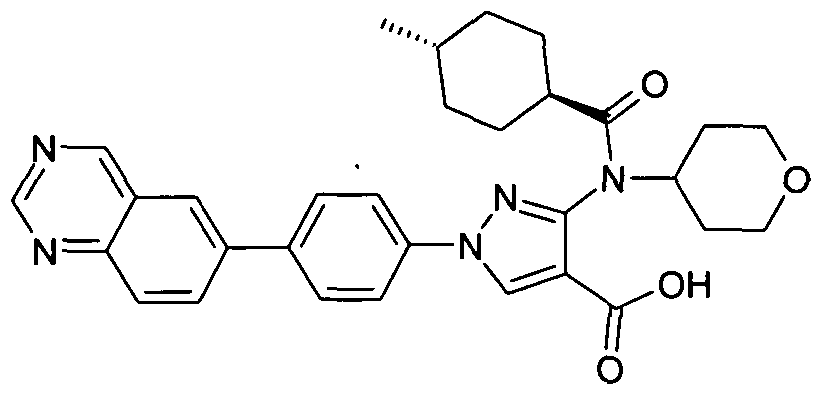
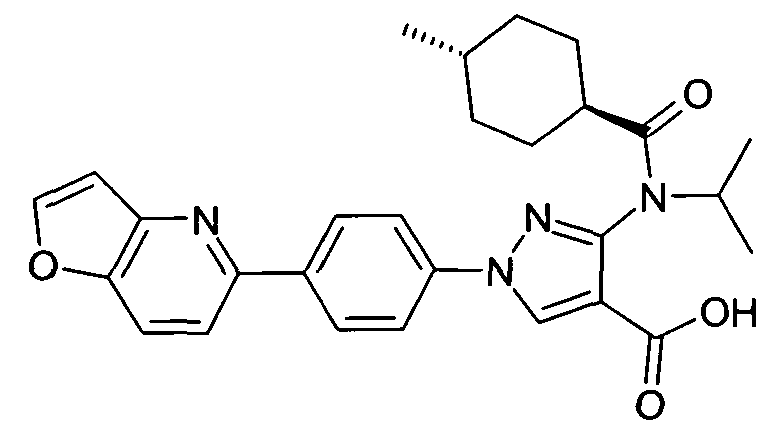
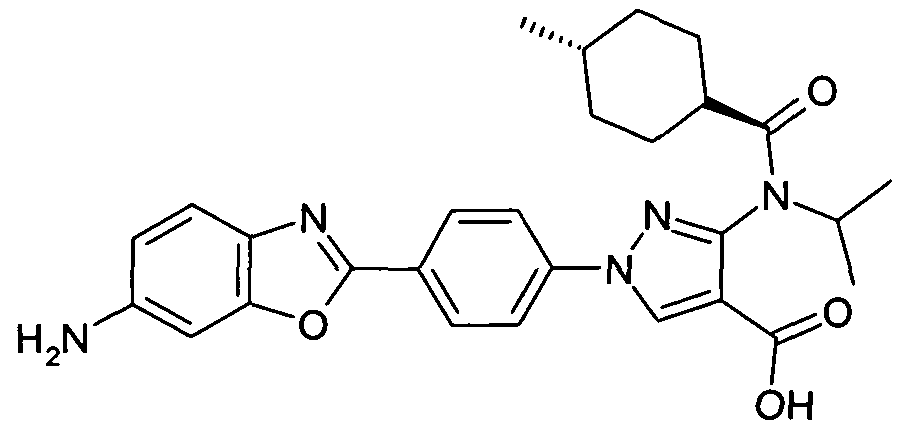
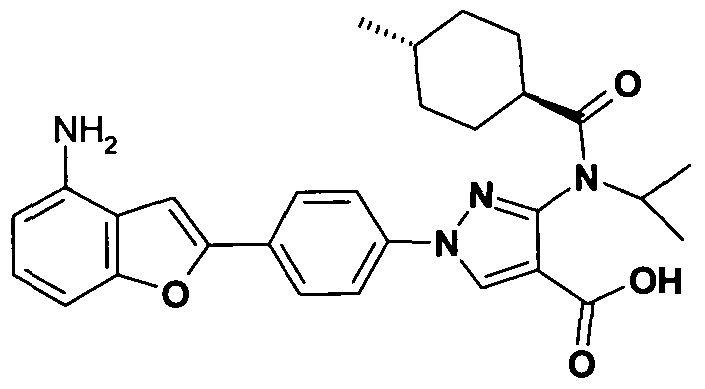
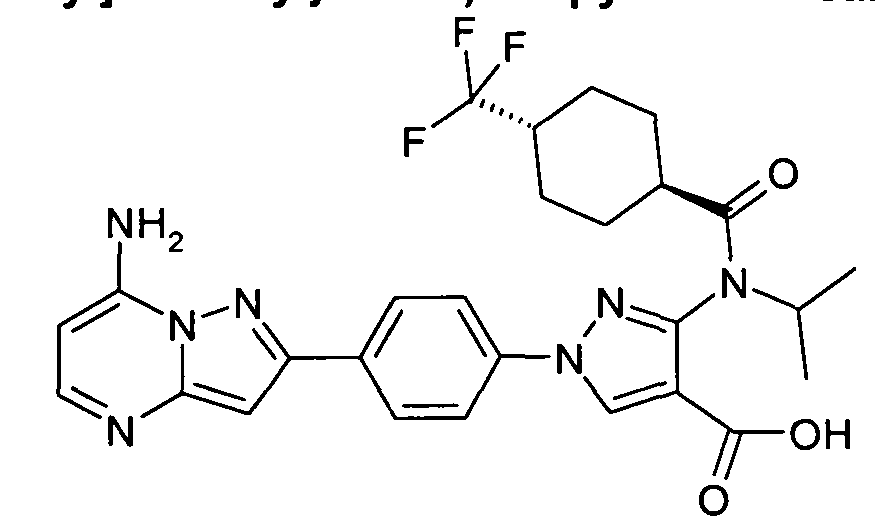
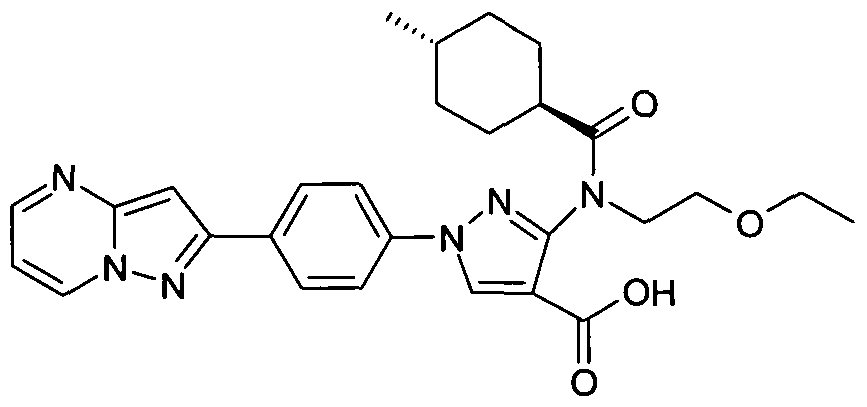
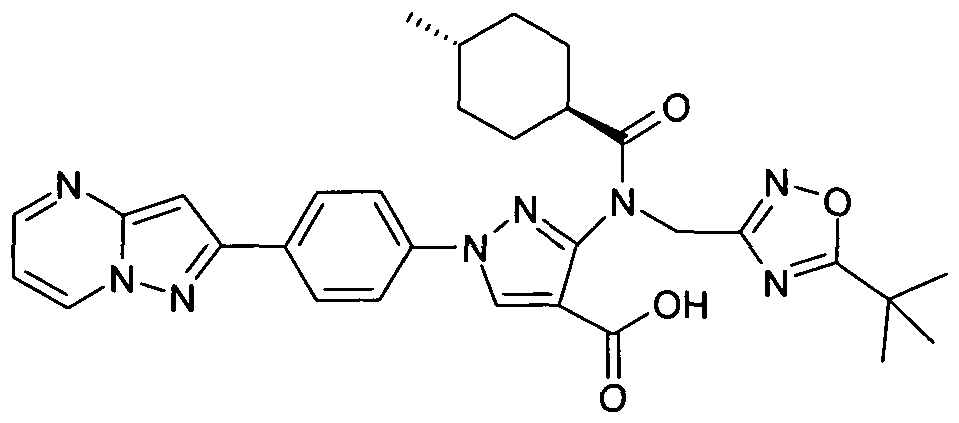
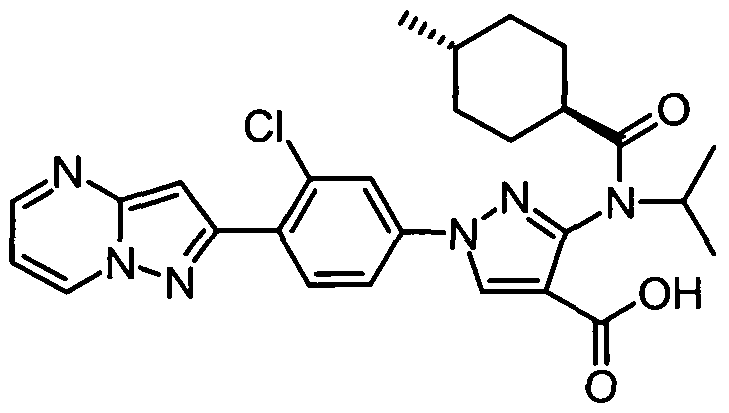
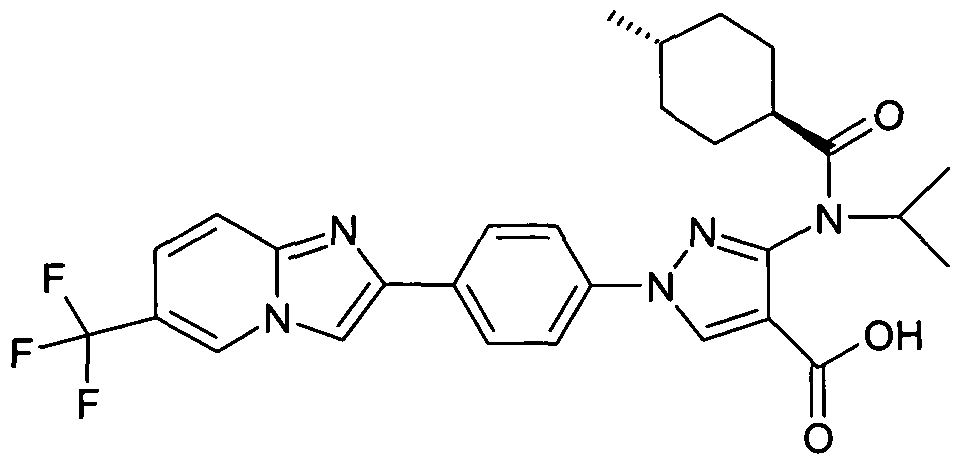
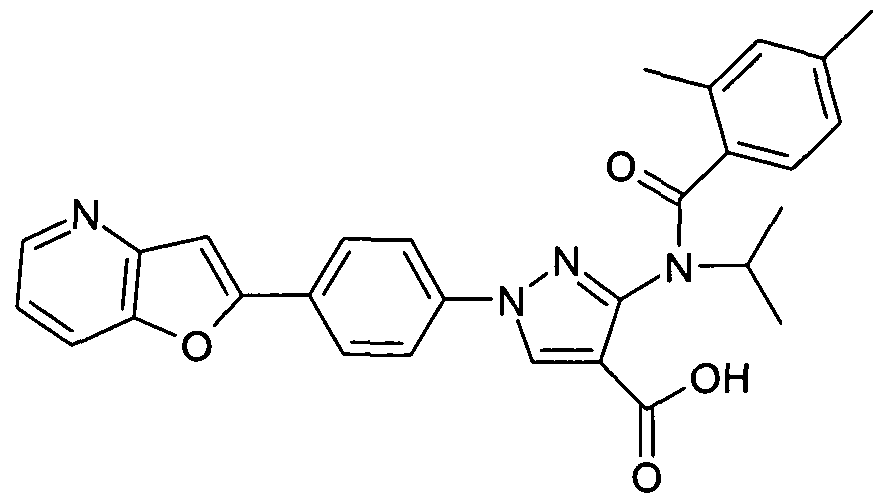
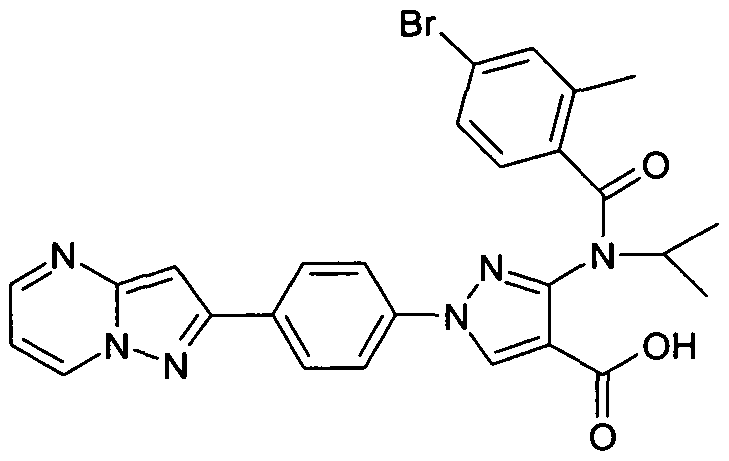
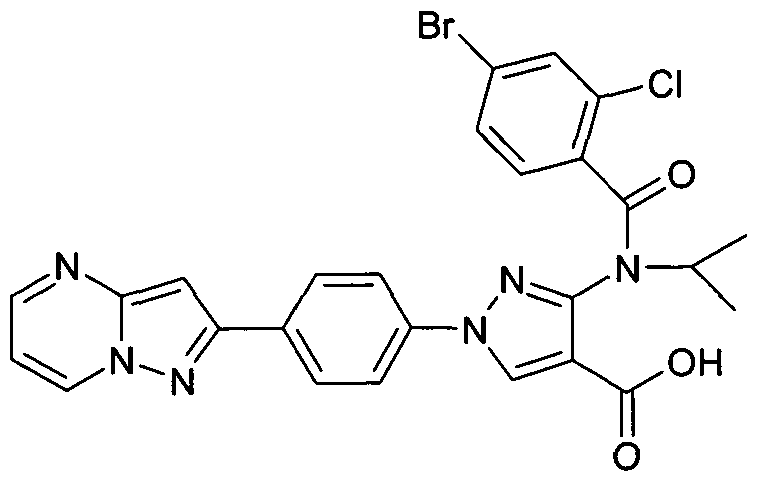
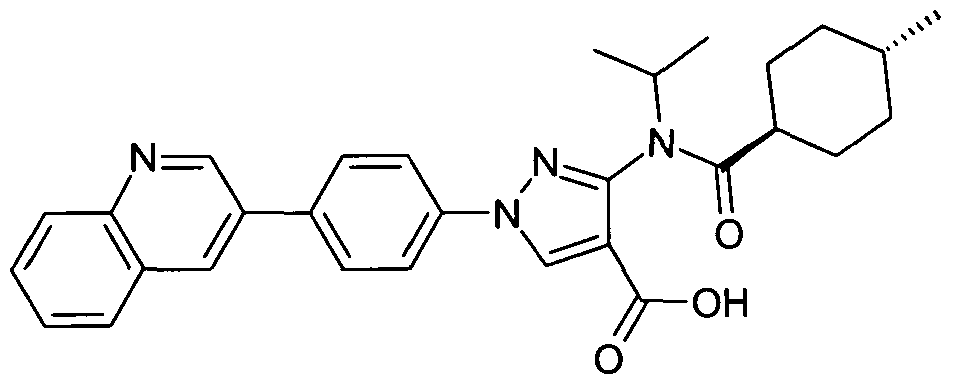
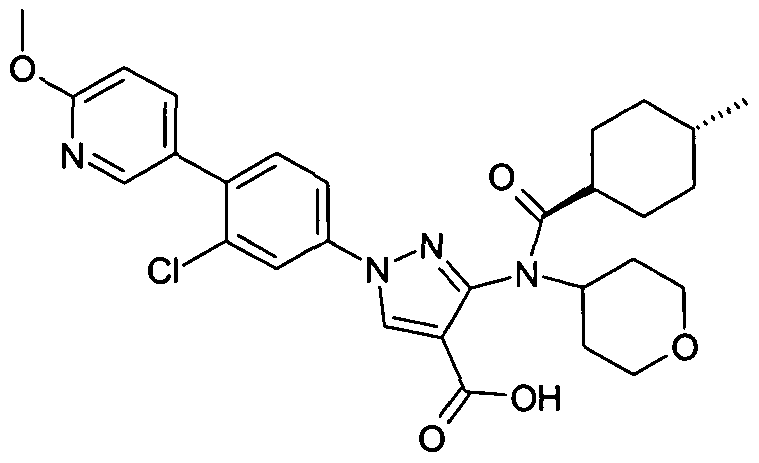
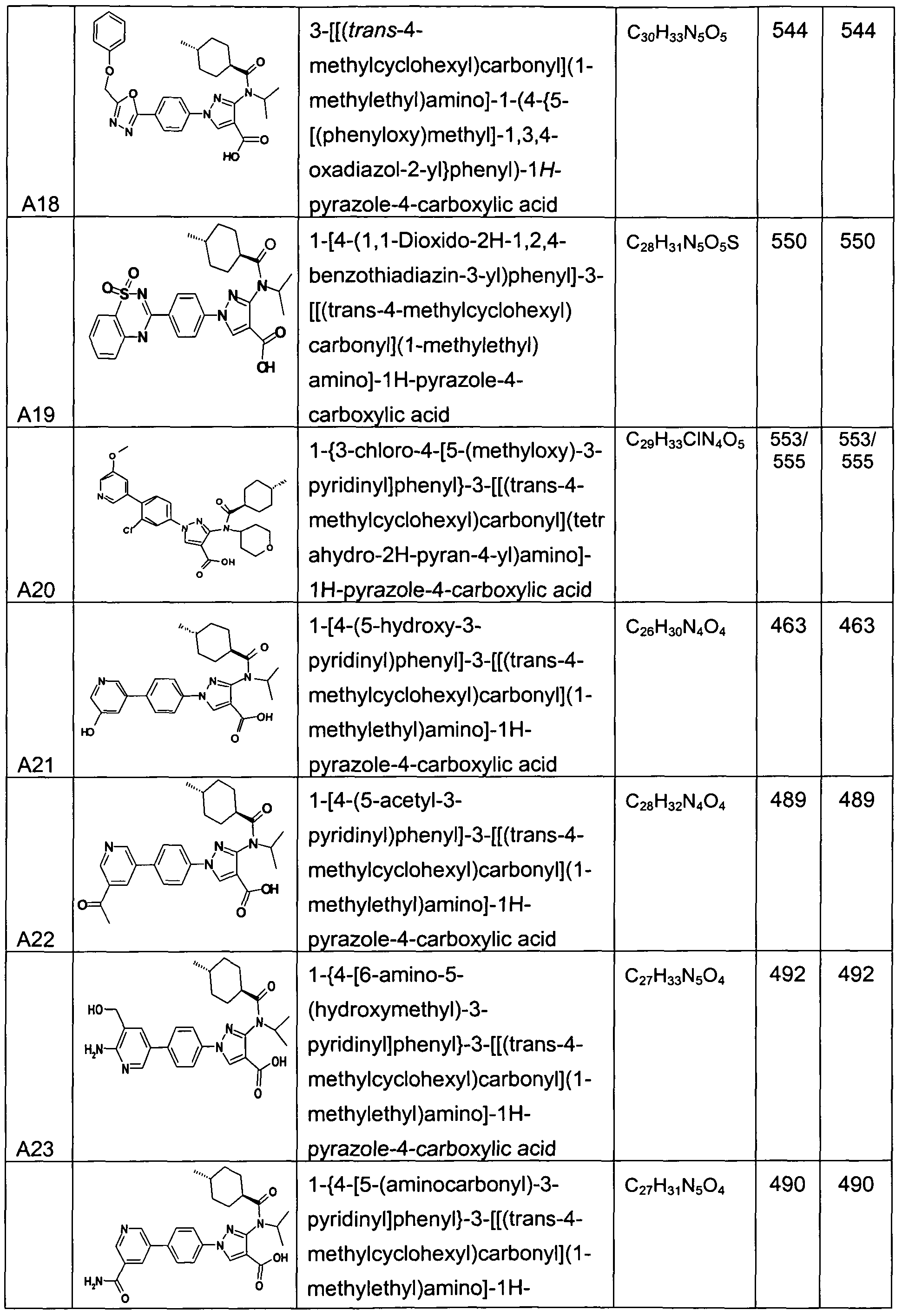
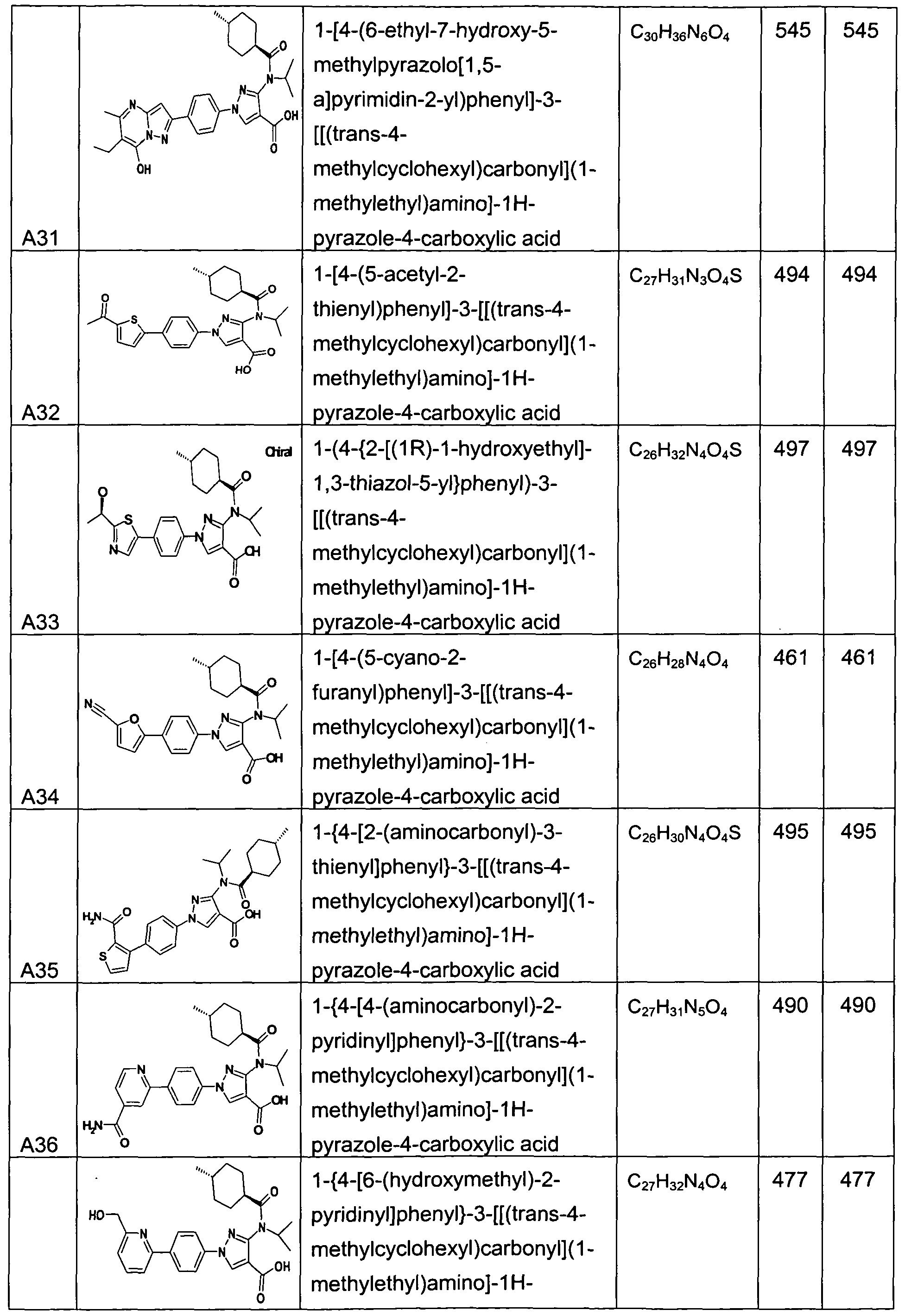
- chemical entities useful in the present invention may be chosen from compounds of Formula (I) selected from the group consisting of:
- Suitable pharmaceutically acceptable salts of the compounds of Formula (I) include acid salts, for example sodium, potassium, calcium, magnesium and tetraalkylammonium and the like, or mono- or di- basic salts with the appropriate acid for example organic carboxylic acids such as acetic, lactic, tartaric, malic, isethionic, lactobionic and succinic acids; organic sulfonic acids such as methanesulfonic, ethanesulfonic, benzenesulfonic and p-toluenesulfonic acids and inorganic acids such as hydrochloric, sulfuric, phosphoric and sulfamic acids and the like.
- organic carboxylic acids such as acetic, lactic, tartaric, malic, isethionic, lactobionic and succinic acids
- organic sulfonic acids such as methanesulfonic, ethanesulfonic, benzenesulfonic and p-toluen
- the present invention also relates to solvates of the compounds of Formula (I), for example hydrates.
- the present invention also relates to pharmaceutically acceptable esters of the compounds of Formula (I), for example carboxylic acid esters -COOR, in which R is selected from straight or branched chain alkyl, for example n-propyl, n-butyl, alkoxyalkyl (e.g. methoxymethyl), aralkyl (e.g. benzyl), aryloxyalkyl (e.g. phenoxymethyl), aryl (e.g. phenyl optionally substituted by halogen, -C 1-4 alkyl or -C ⁇ alkoxy or amino); or for example - CH 2 OC(O)R' or -CH 2 OCO 2 R' in which R' is alkyl (e.g.
- R' is f-butyl
- any alkyl moiety present in such esters preferably contains 1 to 18 carbon atoms, particularly 1 to 4 carbon atoms.
- Any aryl moiety present in such esters preferably comprises a phenyl group.
- the compound of Formula (Ia) is in the form of parent compound, a salt or a solvate.
- the term "pharmaceutically acceptable” used in relation to an ingredient (active ingredient such as an active ingredient, a salt thereof or an excipient) which may be included in a pharmaceutical formulation for administration to a patient refers to that ingredient being acceptable in the sense of being compatible with any other ingredients present in the pharmaceutical formulation and not being deleterious to the recipient thereof.
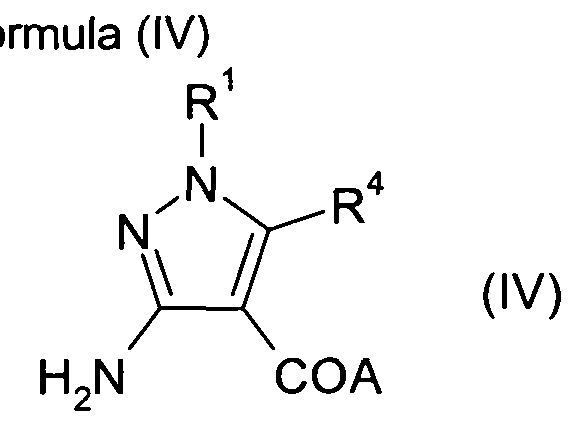
- A is a protected hydroxy group, for example an alkoxy, benzyloxy or silyloxy group and R 1 , R 2 , R 3 and R 4 are as defined above for Formula (I).
- R 1 , R 2 , R 3 and R 4 are as defined above for Formula (I)
- an appropriate base for example aqueous sodium hydroxide or lithium hydroxide, optionally in a solvent such as methanol, tetrahydrofuran or combinations thereof.
- the temperature is in the range 25 to 100 0 C, more preferably 50 to 100°C.
- R 1 , R 2 , R 3 and R 4 are as defined above for Formula (I)
- an appropriate acid for example trifluoroacetic acid.
- the reaction is carried out in a solvent, for example dichloromethane.
- the temperature is in the range 0 to 50 0 C, more preferably 15 to 30 0 C.
- A is silyloxy
- R 1 , R 2 , R 3 and R 4 are as defined above for Formula (I)
- a suitable fluoride source for example tetrabutylammonium fluoride.
- the reaction is carried out in a suitable solvent, for example tetrahydrofuran.
- the temperature is in the range 0 to 5O 0 C, more preferably 15 to 30 0 C.
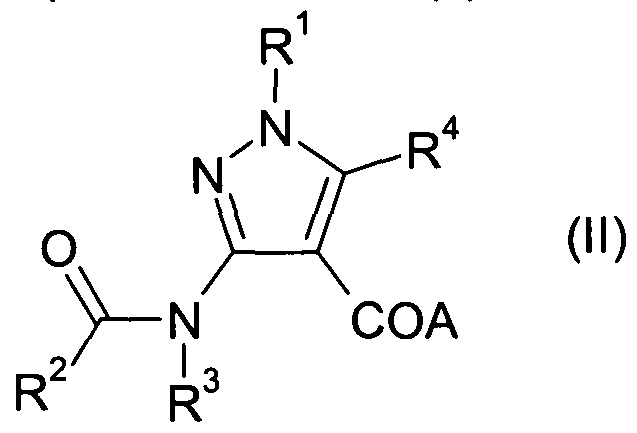
- Compounds of Formula (I) in which A is hydroxy, or (II) in which A is an alkoxy, benzyloxy or silyloxy group may be prepared by reaction of a compound of Formula (III) in which A is hydroxy or an alkoxy, benzyloxy or silyloxy group, and R 1 , R 3 and R 4 are as defined above for Formula (I); with a suitable acylating agent, for example R 2 -C(0)-Y, wherein Y is a halo atom, for example chloro or bromo, and R 2 is as defined above for Formula (I).
- a suitable acylating agent for example R 2 -C(0)-Y, wherein Y is a halo atom, for example chloro or bromo, and R 2 is as defined above for Formula (I).
- the reaction may be carried out in a suitable solvent, for example dichloromethane, in the presence of a suitable base, for example pyridine or triethylamine and thereafter removing any protecting group if desired.
- a suitable solvent for example dichloromethane
- a suitable base for example pyridine or triethylamine
- R 2 represents an aliphatic group
- a phosphine such as triphenylphosphine may optionally be used.
- Suitable protecting groups can be found, but are not restricted to, those found in T W Greene and P G M Wuts 'Protective Groups in Organic Synthesis', 3 rd Ed (1999), J Wiley and Sons.
- a compound of Formula (II) may be prepared by appropriate manipulation of another compound of Formula (II).
- a compound of Formula (II) in which any substituent comprises -C ⁇ alkenyl may be prepared from a suitable aldehyde or ketone substituent and a phosphorous ylid generated from a phosphonium salt in the presence of a suitable base, such as potassium tert-butoxide, in a suitable solvent such as THF.
- a suitable base such as potassium tert-butoxide
- THF a suitable solvent
- the trans and cis isomers may be separated by standard techniques known in the art.
- a compound of Formula (II) in which any substituent comprises -C 2-4 alkyl may be prepared by reduction of an alkenyl substituent, for example using hydrogen, optionally under pressure, in the presence of a suitable catalyst such as palladium on carbon, in a suitable solvent such as ethanol.
- a compound of Formula (II) in which any substituent comprises -C(O)NR A R B may be prepared by reacting a suitable acid substituent with an amine (HNR A R B ) in the presence of a coupling agent such as HATU, in the presence of a suitable base such as triethylamine, in a suitable solvent such as DMF.
- a compound of Formula (II) in which any substituent comprises -C(O)NR A R B may be prepared by reacting a suitable acid chloride substituent with an amine (HNR A R B ) in the presence of a suitable base such as triethylamine, in a suitable solvent such as dichloromethane.
- a compound of Formula (II) in which any substituent comprises -NR E C(O)R D may be prepared by reacting a suitable amine substituent with a carboxylic acid (R 0 CO 2 H) in the presence of a coupling agent such as HATU, in the presence of a suitable base such as triethylamine, in a suitable solvent such as DMF.
- a compound of Formula (II) in which any substituent comprises -NR E C(O)R D may be prepared by reacting a suitable amine substituent with an acid chloride in the presence of a suitable base such as triethylamine in a suitable solvent such as dichloromethane.
- a compound of Formula (II) in which any substituent comprises -NR E SO 2 R D may be prepared by reacting an amine substituent with a suitable sulfonyl chloride in the presence of a suitable base such as triethylamine, in a suitable solvent such as dichloromethane.
- a compound of Formula (II) in which any substituent comprises -SO 2 NR F R G may be prepared by reacting a sulfonyl chloride substituent with a suitable amine (HNR F R G ) in the presence of a suitable base such as triethylamine, in a suitable solvent such as dichloromethane.
- a compound of Formula (II) in which any substituent comprises -SO 2 R A may be prepared by oxidation of a compound in which a substituent represents -SR A , using for example oxone, sodium periodate, 3-chloro perbenzoic acid, or hydrogen peroxide.
- a suitable solvent such as dichloromethane
- A is an alkoxy, benzyloxy or silyloxy, and R 1 and R 4 are as defined above for
- Formula (I) by treatment with a suitable vinyl ether, or a suitable aldehyde or a suitable ketone in the presence of a suitable acid, such as acetic acid, and a suitable reducing agent such as sodium triacetoxyborohydride, in a suitable solvent such as dichloromethane.
- a suitable acid such as acetic acid
- a suitable reducing agent such as sodium triacetoxyborohydride
- compounds of Formula (III) in which A is an alkoxy, benzyloxy or silyloxy group may be prepared from compounds of Formula (IV) in which A is an alkoxy, benzyloxy or silyloxy, and R 1 and R 4 are as defined above for Formula (I), by treatment with a suitable alkylating agent R 3 -X where X is a halo group such as chloride, bromide or iodide, or X is a sulphonate ester such as methanesulfonate, in suitable solvent such as dimethylformamide in the presence of a suitable base such as triethylamine.
- a suitable alkylating agent R 3 -X where X is a halo group such as chloride, bromide or iodide, or X is a sulphonate ester such as methanesulfonate, in suitable solvent such as dimethylformamide in the presence of a suitable base such as triethylamine.
- Compounds of Formula (III) in which A is hydroxy may be prepared from compounds of Formula (III) in which A is an alkoxy, benyloxy or silyloxy group, for example by treatment with an appropriate base, acid or fluoride source as described in relation to the preparation of compounds of Formula (I) from compounds of Formula (II).
- R 1 is as defined above for Formula (I) with compounds of Formula (Vl) in which R 4 and A are as defined above for Formula (II) and R' is -C 1-4 alkyl (such as ethyl) in a suitable solvent such as ethanol or xylene, preferably in the temperature range 50-75 0 C.
- R 1 is as defined above for Formula (I) and P is a suitable nitrogen protecting group such as benzylidine, with compounds of Formula (Vl) in a suitable solvent such as ethanol or xylene, preferably in the temperature range 50-75 0 C.
- a suitable acid such as hydrochloric acid
- a suitable solvent such as ethanol
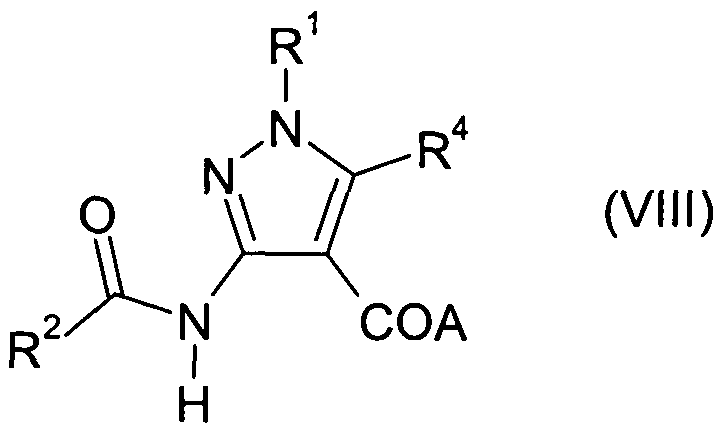
- Compounds of Formula (II) may also be prepared by reaction of a compound of Formula (VIII) in which A is an alkoxy, benzyloxy or silyloxy group, and R 1 , R 2 and R 4 are as defined above for Formula (I); with a suitable alkylating agent R 3 -X in which X is a halo atom such as chloro, bromo or iodo, or X is a sulphonate ester such as methanesulfonate, in a suitable solvent such as dimethylformamide, in the presence of a suitable base such as sodium hydride or sodium f-butoxide, optionally in the presence of triethylamine.
- a suitable alkylating agent R 3 -X in which X is a halo atom such as chloro, bromo or iodo, or X is a sulphonate ester such as methanesulfonate, in a suitable solvent such as dimethylform
- Compounds of Formula (VIII) may be prepared from compounds of Formula (IV) by reaction with a suitable acylating agent, for example R 2 -C(O)-Y, in which Y is a halo atom, preferably chloro or bromo, and R 2 is as defined above for Formula (I).
- a suitable acylating agent for example R 2 -C(O)-Y, in which Y is a halo atom, preferably chloro or bromo, and R 2 is as defined above for Formula (I).
- the reaction is carried out in a suitable solvent, for example dichloromethane, optionally in the presence of a suitable base, for example pyridine or triethylamine.
- a suitable solvent for example dichloromethane
- a suitable base for example pyridine or triethylamine.
- a phosphine such as triphenylphosphine may optionally be used in place of an amine base.
- A is a hydroxyl or an alkoxy, benzyloxy or silyloxy group
- R 2 , R 3 and R 4 are as defined above for Formula (I); by treatment with an aryl or heteroaryl boronic acid (R ⁇ -R x - boronic acid) in the presence of a copper reagent such as copper (II) acetate.
- a copper reagent such as copper (II) acetate.
- the reaction is carried out in the presence of a base, such as pyridine, in air, and in a suitable solvent such as dichloromethane or THF, and at a temperature in the range 10-30 0 C.
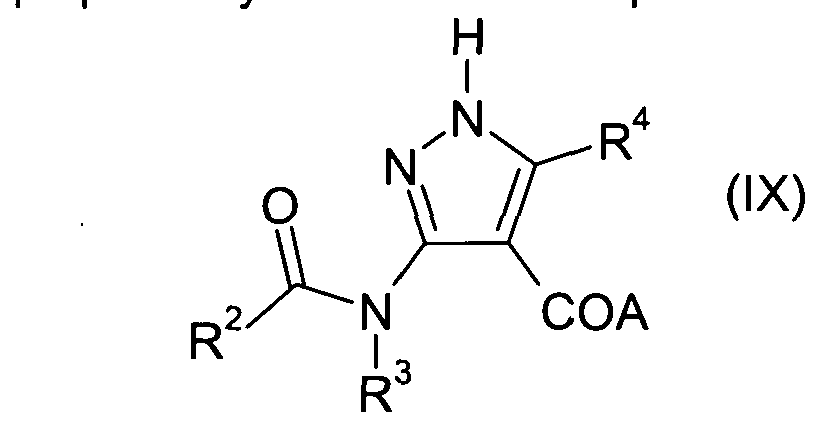
- compounds of Formula (II) may be prepared by reaction of compounds of Formula (IX) with an aryl or heteroaryl halide or triflate (R ⁇ -R x -halide or R ⁇ -R x -triflate) in the presence of a copper catalyst such as copper (I) iodide.
- the reaction is carried out in the presence of a base such as potassium carbonate, and in the presence of a reagent such as trans- ⁇ ,2-diaminocyclohexane or trans-N,N'-dimethyl-1 ,2-cyclohexanediamine, or a combination thereof in a suitable solvent such as dioxane, DMF or pyridine or a combination thereof, and at a temperature in the range 90-180 0 C.
- a base such as potassium carbonate
- a reagent such as trans- ⁇ ,2-diaminocyclohexane or trans-N,N'-dimethyl-1 ,2-cyclohexanediamine, or a combination thereof
- a suitable solvent such as dioxane, DMF or pyridine or a combination thereof
- This reaction can be suitably be performed in a microwave oven.
- R x , R 2 , R 3 , R 4 and A are as defined for Formula (II) by reaction with a suitable heteroaryl boronic acid, R ⁇ -boronic acid, in the presence of a palladium catalyst such as palladium (II) acetate, a reagent such as 2- dicyclohexylphosphino-2'(N,N-dimethylamino)-biphenyl, and an additional reagent such as cesium fluoride, in a suitable solvent such as dioxane.
- a palladium catalyst such as palladium (II) acetate
- a reagent such as 2- dicyclohexylphosphino-2'(N,N-dimethylamino)-biphenyl
- an additional reagent such as cesium fluoride
- Z represents B(OH) 2 , and R x , R 2 , R 3 , R 4 and A are as defined for Formula (II), by reaction with a suitable heteroaryl halide R ⁇ -hal, in which suitably the halide is bromo or iodo, in the presence of a palladium catalyst such as palladium (II) acetate, a reagent such as 2- dicyclohexylphosphino-2'(N,N-dimethylamino)-biphenyl, and an additional reagent such as cesium fluoride, in a suitable solvent such as dioxane.
- a suitable heteroaryl halide R ⁇ -hal in which suitably the halide is bromo or iodo
- a palladium catalyst such as palladium (II) acetate
- a reagent such as 2- dicyclohexylphosphino-2'(N,N-dimethylamino)
- Compounds of Formula (II)' in which Z is halo may be prepared by reaction of a compound of Formula (IX) with a boronic acid of Formula Z-R x -boronic acid under the conditions described above for the preparation of compounds of Formula (I) and (II) from (IX) and R ⁇ -R x -boronic acid.
- R 1 represents a 4-ethynylphenyl derivative
- R 2 , R 3 , R 4 and A are as defined above for Formula (II)
- a suitable pyridine the pyridine being substituted with adjacent hydroxy and iodo groups
- a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide
- a suitable solvent such as triethylamine or DMF.
- the temperature is in the range 50-80 C.
- R 1 represents a 4-(pyrrolopyridine)phenyl
- R 1 represents a 4-(pyrrolopyridine)phenyl
- a compound of Formula (II)" in which R 1 represents 4- ethynylphenyl with an appropriate pyridine (the pyridine being substituted by adjacent amino and iodo groups), in the presence of a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide, in a suitable solvent such as triethylamine.
- a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide
- the temperature is in the range 50-80 C.
- pyrrolopyridine synthesis see Heterocycles (1986) 24, 31 , Tetrahedron (2003) 59, 1571 , Synlett (1992) 515.
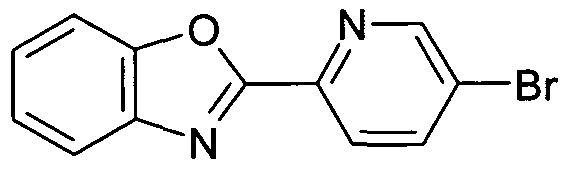
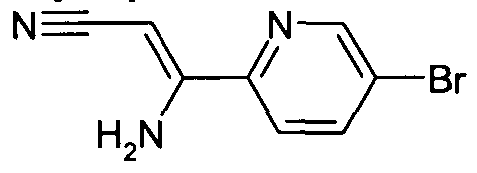
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(oxazolopyridine)phenyl may be prepared by reacting a compound of Formula (II)" in which R 1 represents 4-carboxyphenyl with an appropriate pyridine derivative (the pyridine being substituted with adjacent amino and hydroxyl groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-200 0 C (see for example J. Med. Chem. (1978) 21 , 1158).
- the acid chloride of the 4-carboxyphenyl may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) in a microwave reactor in a suitable solvent such as dioxan (see for example Tetrahedron Letters (2003) 44, 175).
- Compounds of Formula (II) in which R 1 represents a 4- (oxazolopyridine)phenyl may also be prepared by reacting the 4-carboxyphenyl derivative with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(thiazolopyridine)phenyl may be prepared by reacting a compound of Formula (II)" in which R 1 represents 4-phenyl-COCI with an appropriate pyridine (the pyridine being substituted with adjacent amino and chloro groups), in the presence of a suitable base such as pyridine, and then in a second step cyclised using a reagent such as Lawesson's reagent in a suitable solvent such as DMPU, at a suitable temperature such as 90-1 10 0 C.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(thiazolopyridine)phenyl may be prepared by reacting a compound of Formula (II)" in which R 1 represents 4-carboxyphenyl with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-200 0 C (see for example J. Med. Chem. (1978) 21 , 1158).
- the acid chloride of the 4-carboxyphenyl may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in a microwave reactor in a suitable solvent such as dioxan (see for example Tetrahedron Letters (2003) 44, 175).
- the 4-carboxyphenyl compound may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(2,3-dihydro-1,1-dioxo-1 ,2- benzisothiazol-2(3H)-yl)phenyl derivative may be prepared by treatment of a compound of Formula (II)' in which Z represents 4-halo with a 2,3-dihydro-1 ,2-benzisothiazole 1 ,1-dioxide derivative in the presence of copper (I) iodide with a suitable base such as potassium carbonate, and in the presence of a reagent such as trans- ⁇ ,2-diaminocyclohexane or trans- N.N'-dimethyl-i ⁇ -cyclohexanediamine, or a combination thereof in a suitable solvent such as dioxan, DMF or pyridine or a combination thereof, and at a temperature in the range 90- 160 0 C.
- R 1 represents a 4-(2,3-dihydro-1,
- 2-yl)phenyl derivative may be prepared by treatment of a compound of Formula (II)" in which R 1 represents a 4-aminophenyl derivative with a phenyl-1 ,2-di-aldehyde derivative in acetic acid optionally with a suitable solvent such as dichloromethane.
- compounds of Formula (I) or (II) in which in which R 1 represents a 4-(1-oxo-1 ,3-dihydro-2H- isoindol-2-yl)phenyl derivative may also be prepared by treatment of a compound of Formula (II)" in which R 1 represents a 4-aminophenyl derivative with a phenyl-1 ,2-di-aldehyde derivative in acetic acid optionally with a suitable solvent such as dichloromethane.
- R 1 represents 4-aminophenyl with a suitable phenyl derivative (this being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine, in a suitable solvent such as acetonitrile.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-((1-oxo-1 ,3-dihydro-2H-isoindol- 2-yl)pyridine)phenyl derivative may be prepared by treatment of a compound of Formula (II)" in which R 1 represents 4-aminophenyl with an appropriate pyridine derivative (the pyridine being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine in a suitable solvent such as acetonitrile.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(pyrazolopyrimidine)phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents 4-(5-amino-1 H- pyrazole)phenyl with 1 ,1 ,3,3-tetramethoxypropane in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-110 0 C.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(5-methylpyrazolopyrimidine)- phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents 4- (5-amino-1 H-pyrazole)phenyl with 4,4-dimethoxy-2-butanone in a suitable solvent such as ethanol, suitably the temperature is in the range 60-70°C.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(7-methylpyrazolopyrimidine)- phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents 4- (5-amino-1 H-pyrazole)phenyl with 4,4-dimethoxy-2-butanone in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-110 0 C.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(6- methylpyrazolopyrimidine)phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents 4-(5-amino-1 H-pyrazole)phenyl with 1 ,1 ,3,3-tetraethoxy-2- methylpropane in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-110°C.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(6- flouropyrazolopyrimidine)phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents 4-(5-amino-1 H-pyrazole)phenyl with 3-diethylamino-2-fluoro-2-propenal in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-110 0 C.
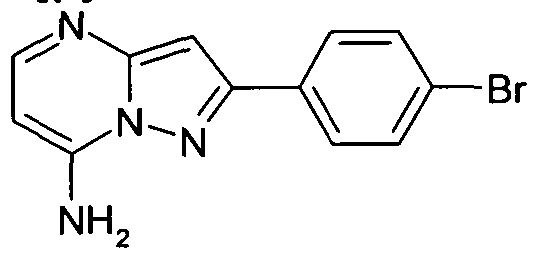
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(7- aminopyrazolopyrimidine)phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents 4-(5-amino-1 H-pyrazole)phenyl with 3-dimethylamino acrylonitrile in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-11O 0 C.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(imidazo[1 ,3]thiazol-6-yl)phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents a 4- (bromoacetophenone) with 2-aminothiazole in a suitable solvent such as ethanol or acetone, suitably the temperature is in the range 50-70 0 C.
- Compounds of Formula (I) or (II) in which R 1 represents a 4-(imidazo[1 ,2-a]pyridine-2- yl)phenyl may be prepared by treating a compound of Formula (II)" in which R 1 represents a 4-(bromoacetophenone) with 2-aminopyridine in a suitable solvent such as ethanol or acetone, suitably the temperature is in the range 50-70 0 C.
- a 4-(imidazo[1 ,3]thiazol-6-yl)phenyl derivative may be prepared by analogy to methods described in Journal of European Chemistry (1995) 30, 901 or JCS Perkin 1 (1989) 643.
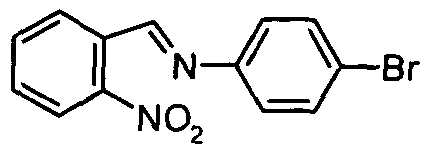
- a 2-(4-bromophenyl)-2H indazole derivative may be prepared by analogy to methods described in Farmaco Ed Sci (1981) 36, 1037 or J. Chem Soc. Perkin Trans 2 (1975), 1185, for example by treating 2-nitrobenzaldehyde with 4-bromoaniline in a suitable solvent such as methanol, and in a separate step reacting the imine with a phosphite such as triethylphosphite, in a microwave reactor at 210°C for 20 mins.
- a suitable solvent such as methanol
- a 2-(4-bromophenyl)imidazo[1 ,2-a]pyridine derivative may be prepared by analogy to methods described in Tetrahedron Letters (2001) 42, 3077.
- a 2-(4-bromophenyl)-1H-benzimidazole derivative may be prepared by analogy to methods described in J. Heterocyclic Chem. (1994) 31 , 957.
- a 2-(4-bromophenyl)-1 H-benzoxazole derivative may be prepared by analogy to methods described in Tetrahedron Letters (2003) 44, 175.
- a 2-(4-bromophenyl)-1H-benzothiazole derivatives may be prepared by analogy to methods described in Tetrahedron Letters (2003) 44, 175 or Synth. Commun. (1990) 20, 3379.
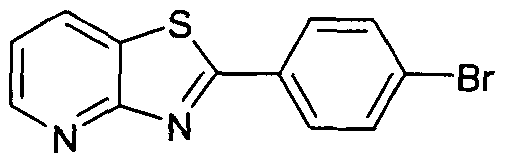
- a 4-(furopyridine)phenyl bromide may be prepared by treatment of a 4-ethynylphenyl bromide with a suitable pyridine (the pyridine being substituted with adjacent hydroxy and iodo groups), with a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide, in a suitable solvent such as triethylamine or DMF.
- a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide
- a suitable solvent such as triethylamine or DMF.
- the temperature is in the range 50-80 0 C.
- a 4-(oxazolopyridine)phenyl derivative may be prepared by reacting a 4-carboxyphenyl bromide with an appropriate pyridine derivative (the pyridine being substituted with adjacent amino and hydroxyl groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-200 0 C (see for example J. Med. Chem. (1978) 21, 1158).
- the acid chloride of the 4-carboxyphenyl bromide may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) in a microwave reactor in a suitable solvent such as dioxane (see for example
- a 4-(oxazolopyridine)phenyl may also be prepared by reacting a 4-carboxyphenyl bromide derivative with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
- a 4-(thiazolopyridine)phenyl bromide may be prepared by reacting an appropriate 4- carboxyphenyl bromide with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-200 0 C (see for example J. Med. Chem. (1978) 21, 1158).
- the acid chloride of the 4-carboxyphenyl may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in a microwave reactor in a suitable solvent such as dioxan (see for example Tetrahedron Letters (2003) 44, 175).
- the 4-carboxyphenyl bromide may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
- a 4-(thiazolopyridine)-phenyl bromide may be prepared by reacting an appropriate 4- bromophenyl-COCI with an appropriate pyridine (the pyridine being substituted with adjacent amino and chloro groups), in the presence of a suitable base such as pyridine, and then in a second step is cyclised using a reagent such as Lawesson's reagent in a suitable solvent such as DMPU 1 at a suitable temperature such as 90-11O 0 C.
- a 4-(1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl bromide derivative may be prepared by treatment of an appropriate 4-aminophenyl bromide derivative with a phenyl-1 ,2-di-aldehyde derivative in acetic acid optionally with a suitable solvent such as dichloromethane.
- a 4-(1- oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl bromide derivative may also be prepared by treatment of an appropriate 4-aminophenyl bromide with a suitable phenyl derivative (this being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine, in a suitable solvent such as acetonitrile.
- a 4-(1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)pyridine bromide may be prepared by treatment of an appropriate 4-aminophenyl bromide with an appropriate pyridine derivative (the pyridine being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine in a suitable solvent such as acetonitrile.
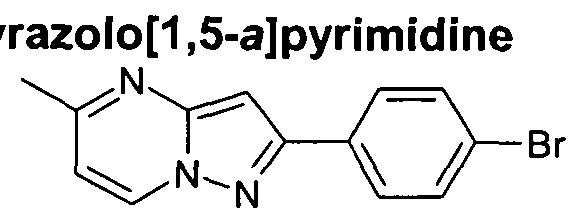
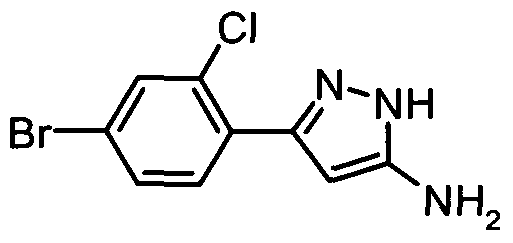
- a 4-(pyrazolopyrimidine)phenyl bromide may be prepared by treating a 3-(4-bromophenyl)- 1 H-pyrazole-5-amine with 1 ,1 ,3,3-tetramethoxypropane in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-110 0 C.
- A is an alkoxy, benzyloxy or silyloxy group, and R 2 , R 3 and R 4 are as defined above for Formula (I); and P is a suitable protecting group.
- Suitable protecting groups include, but are not restricted to, benzyl and tert-butyloxycarbonyl .
- Benzyl groups can be removed by hydrogenation using hydrogen gas with a catalyst such as palladium on carbon in a suitable solvent such as ethanol, optionally in the presence of a suitable acid, for example hydrochloric acid, and optionally conducting the reaction under pressure
- tert- Butyloxycarbonyl groups may be removed using an acid such as hydrochloric acid or trifluoroacetic acid.
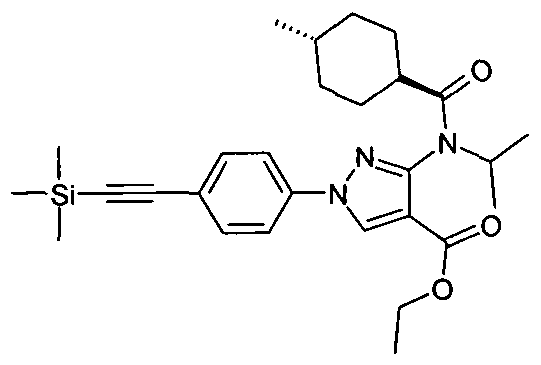
- compounds of Formula (X) may be prepared from compounds of Formulae (III), (IV) or (VIII) in which the group R 1 is a protecting group instead of a group as defined for Formula (I), by application of the synthetic routes described above in relation to the synthesis of compounds of Formula (II).
- Esters of compounds of Formula (I), in which A is -OR where R is selected from straight or branched chain alkyl, aralkyl, aryloxyalkyl, or aryl may also be prepared by esterification of a compound of Formula (I) in which A is hydroxy by standard literature procedures for esterification.
- compounds of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) which exist as diastereoisomers may optionally be separated by techniques well known in the art, for example by column chromatography or recrystallisation. For example, the formation of an ester using a chiral alcohol, separation of the resulting diastereoisomers, and subsequent hydrolysis of the ester to yield the individual enantiomeric acid of Formula (I) (II), (III), (IV), (VIII), (IX) and (X).
- racemic compounds of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) may be optionally resolved into their individual enantiomers. Such resolutions may conveniently be accomplished by standard methods known in the art. For example, a racemic compound of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) may be resolved by chiral preparative HPLC.
- racemic compounds of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) which contain an appropriate acidic or basic group, such as a carboxylic acid group or amine group may be resolved by standard diastereoisomeric salt formation with a chiral base or acid reagent respectively as appropriate. Such techniques are well established in the art.
- a racemic compound may be resolved by treatment with a chiral acid such as (R)-(-)-1 ,1 '-binaphthyl-2,2'-diyl-hydrogen phosphate or (-)-di-O,O'-p-tolyl-L-tartaric acid, in a suitable solvent, for example isopropanol.
- a suitable solvent for example isopropanol.
- the free enantiomer may then be obtained by treating the salt with a suitable base, for example triethylamine, in a suitable solvent, for example methyl tert-butyl ether.
- racemic acid compounds may be resolved using a chiral base, for example (S)-alpha methylbenzylamine, (S)-alpha phenylethylamine, (1S, 2S)-(+)-2-amino-1-phenyl-1,3-propane-diol, (-) ephidrine, quinine, brucine.
- a chiral base for example (S)-alpha methylbenzylamine, (S)-alpha phenylethylamine, (1S, 2S)-(+)-2-amino-1-phenyl-1,3-propane-diol, (-) ephidrine, quinine, brucine.
- Individual enantiomers of Formula (II), (III), (IV), (VIII), (IX) and/or (X) may then be progressed to an enantiomeric compound of Formula (I) by the chemistry described above in respect of racemic compounds.
- the DMF was removed in vacuo and the residue partitioned between DCM and water.
- the DCM layer was separated using a hydrophobic frit and concentrated. This was purified by silica gel chromatography eluting with a gradient of ethyl acetatexyclohexane (5:95 to 100:0) followed by ethyl acetate: methanol (100:0 to 50:50) to give the title compound.
- the reaction was stirred at room temperature for 18 hours.
- the catalyst was removed by filtration through Celite under a stream of nitrogen and the filtrate concentrated.
- the residue was dissolved in DCM and basified with saturated sodium bicarbonate solution.
- the mixture was extracted with DCM and the organic extract washed with brine, dried (MgSO 4 ) and concentrated to give the title compound.
- Tetrahydro-3-furanol 14.71 g was dissolved in anhydrous DCM (400 mL) and was treated with pyridine (16.95 mL). The mixture was cooled to -10°C under an atmosphere of nitrogen.
- Trifluoromethane sulfonic anhydride (51.6 g) was added dropwise ensuring the internal temperature of the reaction did not rise above 0 ° C. The reaction was then stirred at -10 ° C for
- reaction mixture was quenched with aqueous 2M HCI (200 mL) and the organics were separated, dried (Na 2 SO 4 ) and filtered. This solution was then filtered in portions through pre-packed Celite cartridges (2 X 10 g) to give title compound as a DCM solution.
- the filtrate was passed through a hydrophobic frit to remove residual water and was concentrated in vacuo and purified directly by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Virology (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Anti-viral agents of compounds of Formula (I): wherein A, R1, R2, R3 and R4 are as defined in the specification, processes for their preparation and their use in HCV treatment are provided.
Description
4-CARBOXY PYRAZOLE DERIVATIVES AS ANTI-VIRAL AGENTS
FIELD OF THE INVENTION
The present invention relates to novel 4-carboxy pyrazole derivatives useful as anti-viral agents. Specifically, the present invention involves novel inhibitors of Hepatitis C Virus (HCV) replication.
BACKGROUND OF THE INVENTION
Infection with HCV is a major cause of human liver disease throughout the world. In the US, an estimated 4.5 million Americans are chronically infected with HCV. Although only 30% of acute infections are symptomatic, greater than 85% of infected individuals develop chronic, persistent infection. Treatment costs for HCV infection have been estimated at $5.46 billion for the US in 1997. Worldwide over 200 million people are estimated to be infected chronically. HCV infection is responsible for 40-60% of all chronic liver disease and 30% of all liver transplants. Chronic HCV infection accounts for 30% of all cirrhosis, end-stage liver disease, and liver cancer in the U.S. The CDC estimates that the number of deaths due to
HCV will minimally increase to 38,000/year by the year 2010.
Due to the high degree of variability in the viral surface antigens, existence of multiple viral genotypes, and demonstrated specificity of immunity, the development of a successful vaccine in the near future is unlikely. Alpha-interferon (alone or in combination with ribavirin) has been widely used since its approval for treatment of chronic HCV infection. However, adverse side effects are commonly associated with this treatment: flu-like symptoms, leukopenia, thrombocytopenia, depression from interferon, as well as anemia induced by ribavirin (Lindsay, K.L. (1997) Hepatology 26 (suppl 1 ): 71S-77S). This therapy remains less effective against infections caused by HCV genotype 1 (which constitutes -75% of all HCV infections in the developed markets) compared to infections caused by the other 5 major HCV genotypes. Unfortunately, only -50-80% of the patients respond to this treatment (measured by a reduction in serum HCV RNA levels and normalization of liver enzymes) and, of responders, 50-70% relapse within 6 months of cessation of treatment. Recently, with the introduction of pegylated interferon (Peg-IFN), both initial and sustained response rates have improved substantially, and combination treatment of Peg-IFN with ribavirin constitutes the gold standard for therapy. However, the side effects associated with combination therapy and the impaired response in patients with genotype 1 present opportunities for improvement in the management of this disease.
First identified by molecular cloning in 1989 (Choo, Q-L et al (1989) Science 244:359-362), HCV is now widely accepted as the most common causative agent of post-transfusion non A, non-B hepatitis (NANBH) (Kuo, G et al (1989) Science 244:362-364). Due to its genome structure and sequence homology, this virus was assigned as a new genus in the Flaviviridae family. Like the other members of the Flaviviridae, such as flaviviruses (e.g. yellow fever virus and Dengue virus types 1-4) and pestiviruses (e.g. bovine viral diarrhea
virus, border disease virus, and classic swine fever virus) (Choo, Q-L et al (1989) Science 244:359-362; Miller, R.H. and R.H. Purcell (1990) Proc. Natl. Acad. Sci. USA 87:2057-2061 ), HCV is an enveloped virus containing a single strand RNA molecule of positive polarity. The HCV genome is approximately 9.6 kilobases (kb) with a long, highly conserved, noncapped 51 nontranslated region (NTR) of approximately 340 bases which functions as an internal ribosome entry site (IRES) (Wang CY et al 'An RNA pseudoknot is an essential structural element of the internal ribosome entry site located within the hepatitis C virus 5' noncoding region' RNA- A Publication of the RNA Society. 1 (5): 526-537, 1995 JuI.). This element is followed by a region which encodes a single long open reading frame (ORF) encoding a polypeptide of -3000 amino acids comprising both the structural and nonstructural viral proteins.
Upon entry into the cytoplasm of the cell, this RNA is directly translated into a polypeptide of -3000 amino acids comprising both the structural and nonstructural viral proteins. This large polypeptide is subsequently processed into the individual structural and nonstructural proteins by a combination of host and virally-encoded proteinases (Rice, CM. (1996) in B.N. Fields, D.M.Knipe and P.M. Howley (eds) Virology 2nd Edition, p931-960; Raven Press, N.Y.). Following the termination codon at the end of the long ORF, there is a 3' NTR which roughly consists of three regions: an - 40 base region which is poorly conserved among various genotypes, a variable length poly(U)/polypyrimidine tract, and a highly conserved 98 base element also called the "31 X-tail" (Kolykhalov, A. et al (1996) J. Virology 70:3363-3371; Tanaka, T. et al (1995) Biochem Biophys. Res. Commun. 215:744-749; Tanaka, T. et al (1996) J. Virology 70:3307-3312; Yamada, N. et al (1996) Virology 223:255-261). The 31 NTR is predicted to form a stable secondary structure which is essential for HCV growth in chimps and is believed to function in the initiation and regulation of viral RNA replication.
The NS5B protein (591 amino acids, 65 kDa) of HCV (Behrens, S.E. et al (1996) EMBO J. 15:12-22), encodes an RNA-dependent RNA polymerase (RdRp) activity and contains canonical motifs present in other RNA viral polymerases. The NS5B protein is fairly well conserved both intra-typically (-95-98% amino acid (aa) identity across 1b isolates) and inter-typically (-85% aa identity between genotype 1a and 1b isolates). The essentiality of the HCV NS5B RdRp activity for the generation of infectious progeny virions has been formally proven in chimpanzees (A. A. Kolykhalov et al.. (2000) Journal of Virology, 74(4): 2046-2051). Thus, inhibition of NS5B RdRp activity (inhibition of RNA replication) is predicted to be useful to treat HCV infection.
Based on the foregoing, there exists a significant need to identify synthetic or biological compounds for their ability to inhibit HCV.
SUMMARY OF THE INVENTION
The present invention involves novel 4-carboxy pyrazole compounds represented hereinbelow, pharmaceutical compositions comprising such compounds and use of the compounds in treating viral infection, especially HCV infection.
DETAILED DESCRIPTION OF THE INVENTION
The present invention provides at least one chemical entity chosen from compounds of Formula (I) :
A represents hydroxy;
R1 represents -Rx-Rγ;
Rx represents phenyl (optionally substituted by halo, methyl, ethyl, methoxy or trifluoromethyl) or a 5 or 6-membered heteroaryl bonded through a ring carbon atom to the nitrogen atom of the pyrazole;
Rγ represents heteroaryl, bonded such that when Rx is phenyl, Ry is in the para-position;
R2 represents phenyl substituted by one or more substituents selected from -C1-6alkyl, halo, - ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)RD, -NRECO2RD, - NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, and heterocyclyl, or two substituents on neighbouring carbon atoms of the phenyl ring together form a 4-, 5- or 6-membered ring containing one or more heteroatoms selected from O, S and N; or R2 represents C5. /cycloalkyl optionally substituted by one or more substitutents selected from -C1-6alkyl, =CH(CH2)tH, -ORA, -SRA, -C(O)NR8R0, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, - NRECO2R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a C1-2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C^spiro substituent; or R2 represents Cs-ycycloalkylmethyl, the cycloalkyl portion being optionally substituted by one or more substitutents selected from -d-βalkyl, =CH(CH2),H, - 0RA, -SRA, -C(O)NR6R0 -C(O)R0, -CO2H, -CO2R0, -NR6R0, -NREC(0)R°, -NREC02R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a d.2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4SpJrO substituent;
t represents 0, 1 , 2, 3 or 4;
R3 represents heterocyclyl; or phenyl optionally substituted by one or more substituents selected from -C1-6alkyl, halo, -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -
NREC(O)RD, -NRECO2R°, -NRESO2R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, and heterocyclyl; or R3 represents -C1-6alkyl optionally substituted by one or more substituents selected from -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -
NREC(0)RD, -NREC02R°, -NRESO2RD, -NREC(0)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, phenyl, heteroaryl and heterocyclyl;
R4 represents hydrogen;
RA represents hydrogen, -C1-6alkyl, arylalkyl, heteroarylalkyl, aryl, heterocyclyl or heteroaryl;
RB and Rc independently represent hydrogen, -C1-6alkyl, aryl, heterocyclyl or heteroaryl; or RB and Rc together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
R° is selected from the group consisting of -C1-6alkyl, aryl, heterocyclyl, heteroaryl, arylalkyl, and heteroarylalkyl;
RE represents hydrogen or -C^alkyl;
RF and RG are independently selected from the group consisting of hydrogen, -C1-6alkyl, aryl, heteroaryl, arylalkyl, and heteroarylalkyl; or RF and RG together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
and salts, solvates and esters thereof.
There is provided as a further aspect of the present invention at least one chemical entity chosen from compounds of Formula (I) and pharmaceutically acceptable salts, solvates and esters thereof for use in human or veterinary medical therapy, particularly in the treatment or prophylaxis of viral infection, particularly HCV infection.
It will be appreciated that reference herein to therapy and/or treatment includes, but is not limited to prevention, retardation, prophylaxis, therapy and cure of the disease. It will further be appreciated that references herein to treatment or prophylaxis of HCV infection includes treatment or prophylaxis of HCV-associated disease such as liver fibrosis, cirrhosis and hepatocellular carcinoma.
In a further or alternative aspect there is provided a method for the treatment of a human or animal subject with viral infection, particularly HCV infection, which method comprises administering to said human or animal subject an effective amount of at least one chemical entity chosen from compounds of Formula (I) and pharmaceutically acceptable salts, solvates and esters thereof.
According to another aspect of the invention, there is provided the use of at least one chemical entity chosen from compounds of Formula (I) and pharmaceutically acceptable salts, solvates and esters thereof in the manufacture of a medicament for the treatment and/or prophylaxis of viral infection, particularly HCV infection.
It will be appreciated that the chemical entities of the present invention may contain one or more asymmetric carbon atoms and may exist in racemic, diastereoisomeric, and optically active forms. All of these racemic compounds, enantiomers and diastereoisomers are contemplated to be within the scope of the present invention.
In one aspect, Rx represents thienyl; pyridinyl; pyridazinyl; or phenyl optionally substituted by halo, methyl, methoxy or trifluoromethyl. In another aspect, Rx represents 2,5 thienyl. In another aspect, Rx represents phenyl optionally substituted by halo or methyl. In a further aspect Rx represents phenyl, 3-chlorophenyl, or 3-methylphenyl, for example phenyl or 3- chlorophenyl.
In one aspect of the invention when Rx represents a heteroaryl group, that group is bonded through one of its ring carbon to the Rγ group.
In one aspect, Rγ represents a heteroaryl group containing 2 fused rings. In another aspect, Rγ represents optionally substituted pyrazolopyrimidine, furopyridine, imidazopyridine, benzoxazole, imidazothiazole, benzofuran or pyrazolopyridine, for example optionally substituted pyrazolopyrimidine, furopyridine, imidazopyridine, benzoxazole, or imidazothiazole In a further aspect, Rγ represents optionally substituted pyrazolo[1 ,5- a]pyrimidin-2-yl, furo[3,2-b]pyridin-2-yl, imidazo[1 ,2-a]pyridin-2-yl, benzoxazol-2-yl, imidazo[2,1-6][1 ,3]thiazol-6-yl, 4-furo[3,2-ό]pyridin-6-yl, 4-furo[3,2-b]pyridin-5-yl, 1- benzofuran-2-yl (for example 7-amino-1-benzofuran-2-yl), or pyrazolo[1 ,5-a]pyridin-2-yl (for example 7-chloropyrazolo[1,5-a]pyridin-2-yl). In a further aspect, Rγ represents optionally substituted pyrazolo[1 ,5-a]pyrimidin-2-yl, furo[3,2-ύ]pyridin-2-yl, imidazo[1 ,2-a]pyridin-2-yl, benzoxazol-2-yl, or imidazo[2,1-b][1 ,3]thiazol-6-yl.
In one aspect, when Rx represents a 6-membered heteroaryl bonded through a ring carbon atom to the nitrogen atom of the pyrazole, Rγ is bonded so as to be in the para-position.
In one aspect, R2 represents phenyl substituted by one or more substituents selected from - Ci-βalkyI, halo, -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)RD, - NRECO2RD, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, and heterocyclyl; or R2 represents C5-7cycloalkyl optionally substituted by one or more substitutents selected from - C1-6alkyl, =CH(CH2)tH, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, - NREC(O)R°, -NRECO2R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a C1-2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4spiro substituent. In a further aspect, R2 represents phenyl substituted by one or more substituents selected from
or halo, or R2 represents C6cycloalkyl optionally substituted by one or more C1-4alkyl or trifluoromethyl substituents (for example one or more C1-4alkyl substituents). In a further aspect, R2 represents frans-4-methylcyclohexyl or trans- 4-trifluoromethylcyclohexyl, for example frans-4-methylcyclohexyl.
In one aspect, R2 represents a phenyl group substituted by two substituents on neighbouring carbon atoms of the phenyl ring and which together form a 5- or 6-membered ring (including the intervening phenyl ring carbon atoms) containing one or more heteroatoms selected from O, S and N. In one aspect the ring is 5-membered. In a further aspect, the ring contains one heteroatom. In a further aspect, the heteroatom is O. In a further aspect, the ring contains a carbon-carbon double bond. For example, the R2 group may be a benzofuran group.
In one aspect, R3 represents heterocyclyl; or phenyl optionally substituted by one or more substituents selected from -C^alkyl, halo, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, - CO2R0, -NRBRC, -NREC(O)R°, -NRECO2R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, and heterocyclyl; or R3 represents -Chalky! optionally substituted by one or more substituents selected from -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, - NREC(0)R°, -NRECO2R°, -NREC(0)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, phenyl, heteroaryl and heterocyclyl;
In one aspect, R3 represents 1-methylethyl, 2-methylpropyl, 1-ethylpropyl, 1 ,3-dimethylbutyl, ethyl, propyl, 2-ethylbutyl, cyclopentyl, tetrahydro-2H-pyran-4-yl, tetrahydro-2/-/-pyran-4- ylmethyl, tetrahydrofuran-3-yl, 2-methoxyethyl, 2-(methoxy)-1-[(methoxy)methyl]ethyl, 1 ,3- oxazol-2-yl methyl, 2-(phenyloxy)ethyl, 1 ,3-thiazol-4-ylmethyl, (4-methyl-1 ,2,5-oxadiazol-3- yl)methyl, 2-(ethyloxy)ethyl, (2-methyl-1 ,3-thiazol-4-yl)methyl, butyl, (3,5-dimethyl-4- isoxazolyl)methyl, 3-methylbutyl, [5-(1,1-dimethylethyl)-1 ,2,4-oxadiazol-3-yl]methyl, (1- methyl-1H-imidazol-2-yl)methyl, or (5-chloro-2-thienyl)methyl. In one aspect, R3 represents 1-methylethyl, 2-methylpropyl, 1-ethylpropyl, 1 ,3-dimethylbutyl, ethyl, propyl, 2-ethylbutyl, cyclopentyl, tetrahydro-2H-pyran-4-yl, tetrahydro-2H-pyran-4-ylmethyl, tetrahydrofuran-3-yl, 2-methoxyethyl, or 2-(methoxy)-1-[(methoxy)methyl]ethyl. In a further aspect, R3 represents 1-methylethyl, ethyl, tetrahydro-2H-pyran-4-yl, tetrahydrofuran-3-yl or 2-(methoxy)-1- [(methoxy)methyl]ethyl.
It is to be understood that the present invention covers all combinations of aspects, suitable, convenient and preferred groups described herein.
As used herein, "acetyl" refers to -C(O)CH3.
As used herein, "acetylamino" refers to -N(H)C(O)CH3.
As used herein unless otherwise specified, "alkyl" refers to an optionally substituted hydrocarbon group. The alkyl hydrocarbon group may be linear, branched or cyclic, saturated or unsaturated. Where the alkyl group is linear or branched, examples of such groups include methyl, ethyl, n-propyl, 1 -methylethyl (isopropyl), n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, neopentyl or hexyl and the like. Where the alkyl hydrocarbon group is unsaturated, it will be understood that there will be a minimum of 2 carbon atoms in the group, for example an alkenyl or alkynyl group. Where the alkyl hydrocarbon group is cyclic, it will be understood that there will be a minimum of 3 carbon atoms in the group. In one aspect, alkyl moieties are
Unless otherwise stated, optional substituents include -Chalky! (unsubstituted), =CH(CH2)tH, fluoro, -CF3, -ORA, -SRA, -C(O)NR8R0, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(0)R°, -NRECO2RD, -NRESO2RD, -NREC(0)NRFRG, -S02NRFRG, -SO2R0, nitro, cyano, oxo, aryl, heteroaryl and heterocyclyl. For example, substituents include -C1-6alkyl (unsubstituted), =CH(CH2),H, fluoro, -CF3, -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(0)R°, -NRECO2R°, - NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, oxo, aryl, heteroaryl and heterocyclyl.
As used herein, the term "alkenyl" refers to a linear or branched hydrocarbon group containing one or more carbon-carbon double bonds. In one aspect the alkenyl group has from 2 to 6 carbon atoms. Examples of such groups include ethenyl, propenyl, butenyl, pentenyl or hexenyl and the like.
As used herein, the term "alkynyl" refers to a linear or branched hydrocarbon group containing one or more carbon-carbon triple bonds. In one aspect the alkynyl group has from 2 to 6 carbon atoms. Examples of such groups include ethynyl, propynyl, butynyl, pentynyl or hexynyl and the like.
As used herein unless otherwise specified, "cycloalkyl" refers to an optionally substituted, cyclic hydrocarbon group. The hydrocarbon group may be saturated or unsaturated, monocyclic or bridged bicyclic. Where the cycloalkyl group is saturated, examples of such groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl and the like. Where the cycloalkyl group is unsaturated, examples of such groups include cyclobutenyl, cyclopentenyl, cyclohexenyl, cycloheptenyl or cyclooctenyl and the like. In one aspect, the cycloalkyl group has from 5 to 7 carbon atoms. In one aspect, cycloalkyl moieties are cyclohexenyl, cyclopentenyl and cyclohexyl. Unless otherwise stated, the cycloalkyl group may be substituted by one or more optional substituents including -C1-6alkyl
(unsubstituted), =CH(CH2)tH, fluoro, -CF3, -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(0)R°, -NRECO2R°, -NRESO2RD, -NREC(0)NRFRG, -S02NRFRG, -SO2R0, nitro, cyano, oxo, phenyl and heterocyclyl. For example, they include -Cβalkyl (unsubstituted), =CH(CH2),H, fluoro, -CF3, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NRECO2R°, -NREC(0)NRFRG, -S02NRFRG, -SO2R0, nitro, cyano, oxo, phenyl and heterocyclyl.
As used herein, the term "alkoxy" refers to an -O-alkyl group wherein alkyl is as defined herein. Examples of such groups include methoxy, ethoxy, propoxy, butoxy, pentoxy or hexoxy and the like.
As used herein, "aryl" refers to an optionally substituted aromatic group with at least one ring having a conjugated pi-electron system, containing up to two conjugated or fused ring systems. "Aryl" includes carbocyclic aryl and biaryl groups, all of which may be optionally substituted. In one aspect, "aryl" moieties contain 6-10 carbon atoms. In one aspect, "aryl" moieties are unsubstituted, monosubstituted, disubstituted or trisubstituted phenyl. In one aspect, unless otherwise stated, "aryl" substituents are selected from the group consisting of -C^alkyl, halo, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(0)R°, -NRECO2R°, -NRES02R°, -NREC(0)NRFRG, -S02NRFRG, -SO2R0, nitro, cyano, heterocyclyl, - CF3, -OCF3 and phenyl. For example they may be selected from -Chalky!, halo, -0RA, -SRA, -C(O)NR6R0, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NR6CO2R0, -NRεC(O)NRFRG, -S02NRFRG, -SO2R0, nitro, cyano, heterocyclyl, -CF3, -OCF3 and phenyl.
As used herein, "arylalkyl" refers to an aryl group attched to the parent molecular moiety through an alkyl group.
As used herein, "carbonyl" refers to -C(O)-.
As used herein, "cyano" refers to -CN.
As used herein, "halogen" or "halo" refer to a fluorine, chlorine, bromine or iodine atom. References to "fluoro", "chloro", "bromo" or "iodo" should be construed accordingly.
As used herein, unless otherwise specified, "heteroaryl" refers to an optionally substituted, 5, 6, 8, 9 or 10 membered, aromatic group comprising one to four heteroatoms selected from
N, O and S, wherein S may be optionally substituted by one or two oxygen atoms, with at least one ring having a conjugated pi-electron system, containing up to two conjugated or fused ring systems. In one aspect, "heteroaryl" moieties are unsubstituted, monosubstituted, disubstituted or trisubstituted (where applicable) pyridine, pyrazine, thiazole, thiophene, furan, oxadiazole, oxazole, isoxazole, pyrimidine, pyridazine, benzodioxole, benzofuran, benzodioxin, indole, benzimidazole, benzofuran, indole, indazole, isoindole, benzothiophene,
benzothiazole, benzothiadiazine, benzoxazole, benzisoxazole, benzisothiazole, benzotriazole, furopyridine, furopyrimidine, furopyridazine, furopyrazine, furotriazine, pyrrolopyridine, pyrrolopyrimidine, pyrrolopyridazine, pyrrolopyrazine, pyrrolotriazine, thienopyridine, thienopyrimidine, thienopyridazine, thienopyrazine, thienotriazine, thiazolopyridine, thiazolopyrimidine, thiazolopyridazine, thiazolopyrazine, thiazolotriazine, oxazolopyridine, oxazolopyrimidine, oxazolopyridazine, oxazolopyrazine, oxazolotriazine, imidazopyridine, imidazopyrimidine, imidazopyridazine, imidazopyrazine, imidazotriazine, pyrazolopyridine, pyrazolopyrimidine, pyrazolopyridazine, pyrazolopyrazine, pyrazolotriazine, triazolopyridine, triazolopyrimidine, triazolopyridazine, triazolopyrazine, quinoline, naphthyridine, quinoxaline, quinazoline, isoquinoline, cinnoline, pyridopyridazine, pyridopyrimidine, pyridopyrazine, pyrazinopyrazine, pteridine, pyrazinopyridazine, pyrimidopyridazine, pyrimidopyrimidine, imidazothiazole, thiazolooxazole. All isomers of the above heteroaryls are within the scope of this invention. Each heteroaryl group may be attached at any ring carbon or may be attached through nitrogen when the nitrogen is part of a 5-membered ring. Each heteroaryl group may be optionally substituted on a carbon atom by one or more substituents. In one aspect, unless otherwise stated, "heteroaryl" substituents are selected from the group consisting of -C1-6alkyl, halo, -ORA, -SRA, -C(O)NRBRC, -C(O)RD, -CO2R0, -NRBRC, -NREC(O)RD, -NRECO2RD, -NRESO2RD, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, oxo, nitro, cyano, heterocyclyl, -CF3 and phenyl. For example they may be selected from the group consisting of -C1-6alkyl, halo, -ORA, -SRA, -C(O)NR6R0, -C(O)R0, -CO2R0, -NRBRC, -NREC(O)R°, -NRECO2R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, oxo, nitro, cyano, heterocyclyl, -CF3 and phenyl.
As used herein, "heteroarylalkyl" refers to a heteroaryl group attached to the parent molecular moiety through an alkyl group.
As used herein, "heterocyclic" and "heterocyclyl" refer to an optionally substituted, 5 or 6 membered, saturated or partially saturated, cyclic group containing 1 or 2 heteroatoms selected from N, optionally substituted by hydrogen, -C1-6alkyl, -C(O)R0, -C(O)NRBRC, -C(O)OR4, -SO2R0, aryl or heteroaryl; O; and S, optionally substituted by one or two oxygen atoms. Ring carbon atoms may be optionally substituted by -C1-6alkyl, -0RE, -C(O)R0, or -SO2R0. In one aspect, unless otherwise stated, "heterocyclic" moieties are unsubstituted or monosubstituted tetrahydro-2H-pyran-4-yl, piperidinyl and tetrahydrofuran- 3-yl.
As used herein, "nitro" refers to -NO2.
As used herein, "oxo" refers to =0.
As used herein, "Ac" refers to "acetyl", "Et" refers to "ethyl", "iPr" refers to "isopropyl", "Me" refers to "methyl", "OBn" refers to "benzyloxy", and "Ph" refers to "phenyl".
In one aspect, chemical entities useful in the present invention may be chosen from compounds of Formula (I) selected from the group consisting of:
1-[4-(1/-/-lndol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H- pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -[4-(1 H-Benzimidazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid;
1-[3-Chloro-4-(1 H-indol-2-yl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(2H-lndazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]- 1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 -oxo-1 ,3-dihydro-2H- isoindol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1 ,1-Dioxido-1 ,2-benzisothiazol-2(3H)-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1 ,3-Benzoxazol-2-yl)phenyl]-3-[[(f/'ans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1-Benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran- 4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
^^-(i-Benzothien^-yOphenyll-S-^frans^-methylcyclohexyOcarbony^i- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-[1 ,3]thiazolo[5,4-ό]pyridin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(1-methyl-1 H-indol-2- yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(6-Fluoro-1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 -[4-(1 -oxo-1 ,3- dihydro-2H-isoindol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1 ,3-Benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(1 ,3-Benzoxazol-2-yl)phenyl]-3-{ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1 H- pyrazole-4-carboxylic acid; 1 -[4-(1.S-Benzothiazol^-yOphenylj-S-IKfrans^-methylcyclohexyOcarbony^i - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-pyra2olo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid; 3-{Ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-furo[3,2-/)]pyridin-2-ylphenyl)-1 H- pyrazole-4-carboxylic acid;
3-{Ethyl[(fra/7S-4-methylcyclohexyl)carbonyl]amino}-1-(4-imidazo[1 ,2-a]pyridin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro- 2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Fluoro-1 H-indol-2-yl)phenyl]-3-[[(fraA7s-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(3-Chloro-2H-indazol-2-yl)phenyl]-3-[[(fraA7s-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-[4-(1 ,3-Benzothiazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
^^-(i-Benzofuran^-yOphenyll-S-tKfrans^-methylcyclohexyOcarbony^i- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(6-Chloro-7aH-imidazo[4,5-/3]pyridin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Amino-5-methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[( fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Amino-1 ,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-(4-Furo[3,2-ό]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][(3R)-tetrahydro-3- furanyl]amino}-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-/3]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][(3S)-tetrahydro-3- furanyl]amino}-1 H-pyrazole-4-carboxylic acid;
1-[4-(4-Amino-3-cyanopyrazolo[5,1-c][1 ,2,4]triazin-7-yl)phenyl]-3-[[(^rans-4- methylcyclohexyOcarbony^i-methylethyOamino]-! H-pyrazole-4-carboxylic acid;
1-[4-(2H-lndazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-
1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-cf]pyrimidin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -(4-Furo[2,3-c/]pyrimidin-6-ylphenyl)-3-[[(fra/7s-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[2,1-b][1 ,3]thiazol-6-ylphenyl)-3-[[(fra/?s-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5,7-Dimethylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[1 ,2-a]pyπmidin-2-ylphenyl)-3-[[(fraπs-4-rnethylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-c]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid
1 -(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-{(1 -methylethyl)[(4-methylphenyl)carbonyl]amino}-1 H- pyrazole-4-carboxylic acid; 3-[(Cyclohexylcarbonyl)(1 -methylethyl)amino]-1 -(4-furo[3,2-b]pyridin-2-ylphenyl)-1 H- pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-[1 ,3]oxazolo[4,5-fc]pyridin-
2-ylphenyl)-1 /-/-pyrazole-4-carboxylic acid;
3-{Ethyl[(frans-4-methylcydohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)- 1 H-pyrazole-4-carboxylic acid;
3-[[(^rans-4-Methylcyclohexyl)carbonyl](tetrahydro-3-furanyl)amino]-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(fra/7s-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 1 -(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-[[(^ans-4-methylcyclohexyl)carbonyl] (tetrahydro-3- furanyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro-3- furanyl)amino]-1 H-pyrazole-4-carboxylic acid; i-^^I .S-Benzoxazol^-yO^-thienyll-S-t^trans^-methylcyclohexyOcarbony^i- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1 ,3-Benzoxazol-2-yl)-3-chlorophenyl]-3-[[(frans-4-methylcyclohexyl) carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(5-methylpyrazolo[1 ,5- a]pyrimidin-2-yl)phenyl]-1 /-/-pyrazole-4-carboxylic acid; 3-[[(^rans-4-Methylcyclohexyl)carbonyl](1-methylethyl)arnino]-1-[4-(7-methylpyrazolo[1 ,5- a]pyrimidin-2-yl)phenyl]-1 /-/-pyrazole-4-carboxylic acid;
3-[[(^ans-4-Methylcyclohexyl)c^rbonyl](1-methylethyl)amino]-1-[4-(5-methylfuro[3,2-
/)]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6-methylfuro[3,2- ft]pyridin-2-yl)phenyl]-1 /-/-pyrazole-4-carboxylic acid;
1-[6-(1 ,3-Benzoxazol-2-yl)-3-pyridinyl]-3-[[(fraA7s-4-methylcyclohexyl)carbonyl] (1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(6-Furo[3,2-b]pyridin-2-yl-3-pyridinyl)-3-[[(frans-4-methylcyclohexyl)carbonyl] (1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(6-pyrazolo[1 ,5-a]pyrimidin-2- y l-3-pyrid inyl )- 1 H-pyrazole-4-carboxylic acid;
1-[6-(6-Fluoropyrazolo[1 ,5-a]pyrimidin-2-yl)-3-pyridinyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-ylmethyl)amino]-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2--5]pyridin-2-ylphenyl)-3-{[(f/'ans-4-methylcyclohexyl)carbonyl][2-
(methyloxy)ethyl]amino}-1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[2,1 -ύ][1 ,3]thiazol-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl]
(tetrahydro-3-furanyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[2,1 -ύ][1 ,3]thiazol-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl) carbonyl](tetrahydro-2/-/-pyran-4-yl)amino]-1/-/-pyrazole-4-carboxylic acid; 3-{(2-Ethylbutyl)[(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[(frans-4-Methylcyclohexyl)carbonyl][2-(methyloxy)ethyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(^ans-4-Methylcyclohexyl)carbonyl](propyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](2-methylpropyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{(1-Ethylpropyl)[(^ans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-{(1 ,3-Dimethylbutyl)[(fra/7s-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{Cyclopentyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{(1 -Methylethyl)[(4-methylphenyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(4-Chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dichlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-((1-Methylethyl){[4-(trifluoromethyl)phenyl]carbonyl}amino)-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(3,4-Dichlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-[3-Chloro-4-(2-furanyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran- 4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[3-Chloro-4-(3,5-dimethyl-4-isoxazolyl)phenyl]-3-[[(frans-4-methylcyclohexyl) carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
3-([(frans-4-Methylcyclohexyl)carbonyl]{2-(methyloxy)-1 -[(methyloxy)methyl] ethyl}amino)-1 -
(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 1-(4-Furo[3,2--3]pyridin-2-ylphenyl)-3-([(frans-4-methylcyclohexyl)carbonyl]{2-(methyloxy)-1-
[(methyloxy)methyl]ethyl}amino)-1 H-pyrazole-4-carboxylic acid;
1-[4-(1-Benzofuran-6-yl)phenyl]-3-[[(fraA?s-4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-
4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(1 -Benzofuran-5-yl)phenyl]-3-[[(fraA7s-4-methylcyclohexyl)carbonyl] (tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1-Benzofuran-6-yl)-3-chlorophenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro-
2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[2,3-i>]pyridin-5-ylphenyl)-3-[[(^a/7S-4-nnethylcyclohexyl)carbonyl] (tetrahydro-2/-/- pyran-4-yl)amino]-1 /-/-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(1H-pyrrolo[3,2-b]pyridin-
2-yl)phenyl]-1 /-/-pyrazole-4-carboxylic acid; 3-[[(fraA7S-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(1H-pyrrolo[2,3-b]pyridin-
2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-[6-(1-Benzofuran-2-yl)-3-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
^^-(IH-lndol^-yO-S-pyridinylJ-S-^frans^-methylcyclohexyOcarbony^i-methylethyOarnino]- 1 H-pyrazole-4-carboxylic acid;
1-[5-(1-Benzofuran-2-yl)-2-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[5-(1 H-lndol-2-yl)-2-pyridinyl]-3-[[(frans-4-methylcydohexyl)carbonyl](1-methylethyl)amino]-
1 H-pyrazole-4-carboxylic acid; ^^-(i-Benzofuran^-yO-S-pyridazinylJ-S-^fraπs^-methylcyclohexyOcarbony^i- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Aminopyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4-methyl cyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(^rans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6-methylpyrazolo[1 ,5- a]pyrimidin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Ethylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-((1-Methylethyl){[frans-4-(trifluoromethyl)cyclohexyl]carbonyl}amino)-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)arnino]-1-[4-(3-methyl-1H-indol-2- yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(7-oxo-5,7-dihydro-6H- pyrrolo[3,4-ό]pyridin-6-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(5-Fluoro-1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(5-oxo-5,7-dihydro-6H- pyrrolo[3,4-ύ]pyridin-6-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[2,3-/9]pyridin-2-ylphenyl)-3-[[(^ans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-[4-(3-Methyl-1-benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(^aπs-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-[1,3]oxazolo[4,5-b]pyridin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(4-oxo-1 ,4- dihydrothieno[3,2-cdpyrimidin-6-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(2-Chlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(4-Fluorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)- 1 /-/-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(1 /-/-pyrrolo[3,2-/)]pyridin- 2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6-methylfuro[3,2- b]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-{(1 -Methylethyl)[(2-methylphenyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Hydroxy-5-methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(fra/is-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid;
3-[[(3-Chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 /-/-pyrazole-4-carboxylic acid;
3-[[(4-Bromophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 /-/-pyrazole-4-carboxylic acid; 3-[[(2,4-Difluorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-rnethylethyl)amino]-1-(4-{5-[(phenyloxy)methyl]-
1 ,3,4-oxadiazol-2-yl}phenyl)-1 /-/-pyrazole-4-carboxylic acid; or i-^-CI .I-Dioxido^H-i ^^-benzothiadiazin-S-yOphenyll-S-^trans^-methylcyclohexyl) carbonyl](1-methylethyl) amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(6-Amino-1-benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(4-Amino-1 ,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcydohexyl)carbonyl](1-methylethyl)arnino]-1-[4-(1 ,8-naphthyridin-2- yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1-[4-(3-methyl-1 H- indol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(8-Aminoimidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[(f/-ans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(4-Aminothieno[3,2-c(]pyrirnidin-6-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; i^-μ-CMethylaminoJthienotS^-c/lpyrimidin-θ-yπphenylJ-S-ttC^ans^- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -(4-Pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-((1 -methylethyl){[frans-4- (trifluoromethyOcyclohexylJcarbonylJamino)-! H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[2,1-b][1 ,3]thiazol-6-ylphenyl)-3-((1-methylethyl){[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-((1-methylethyl){[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid;
1-(4-lmida2o[1 ,2--3]pyrida2in-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-(4-lmidazo[1 ,2-a]pyrazin-2-ylphenyl)-3-[[(^ar7s-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[8-(Hydroxymethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1/-/-pyrazole-4-carboxylic acid;
1-{4-[8-(Aminomethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ^-aJpyridin-δ-ylphenyO-S-^frarts^-methylcydohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-fe]pyridin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2fy- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(^ans-4-Methylcyclohexyl)carbonyl](tetrahydro-2/-/-pyran-4-yl)amino]-1 -[4-(6- quinazolinyl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-ib]pyridin-5-ylphenyl)-3-[[(fra/7s-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(6-Aminoimidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
^^-(θ-Amino-I .S-benzoxazol^-yOphenyll-S-t^frans^-methylcyclohexyOcarbonylKI- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[(Cyclohexylacetyl)(1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1H- pyrazole-4-carboxylic acid; 3-[(Cyclopentylacetyl)(1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1H- pyrazole-4-carboxylic acid;
1 -[4-(4-Amino-1 -benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Amino-1-benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Aminopyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-((1-methylethyl){[^/'ans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 ,3-oxazol-2-ylmethyl)amino]-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-{[(frans-4-Methylcyclohexyl)carbonyl][2-(phenyloxy)ethyl]amino}-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 ,3-thiazol-4-ylmethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[(frans-4-Methylcyclohexyl)carbonyl][(4-rnethyl-1 ,2,5-oxadiazol-3-yl)methyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[2-(Ethyloxy)ethyl][(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[(frans-4-Methylcyclohexyl)carbonyl][(2-methyl-1 ,3-thia2θl-4-yl)methyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 /-/-pyrazole-4-carboxylic acid;
S-iButy^frans^-methylcyclohexyOcarbonyOaminoJ-i^-pyrazoloII .S-alpyrimidin^-ylphenyl)-
1 H-pyrazole-4-carboxylic acid; 3-{[(3,5-Dimethyl-4-isoxazolyl)methyl][(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 /-/-pyrazole-4-carboxylic acid;
3-{(3-Methylbutyl)[(frans-4-methylcyclohexyl)carbonyl]arnino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{{[5-(1 ,1-Dimethylethyl)-1 ,2,4-oxadiazol-3-yl]methyl}[(frans-4- methylcyclohexyljcarbonyllaminoj-i -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4- carboxylic acid;
3-{[(^ans-4-Methylcyclohexyl)carbonyl][(1-methyl-1H-imidazol-2-yl)methyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 /-/-pyrazole-4-carboxylic acid;
3-{[(5-Chloro-2-thienyl)methyl][(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Chloropyrazolo[1 ,5-a]pyridin-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[5-(6-Chloroimidazo[1 ,2-a]pyridin-2-yl)-2-thienyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -(5-lmidazo[1 ,2-a]pyrimidin-2-yl-2-thienyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(5-lmidazo[1 ,2-a]pyridin-2-yl-2-thienyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(5-pyrazolo[1 ,5-a]pyrimidin-2- yl-2-thienyl)-1 H-pyrazole-4-carboxylic acid;
1-[4-(2-Chloroimidazo[2,1-/j][1 ,3]thiazol-6-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(3-Chloro-4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(^ans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(3-methyl-4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dichlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-furo[3,2-/)]pyridin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-{4-[6- (trifluoromethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-1 H-pyrazole-4-carboxylic acid;
3-[[(fra/is-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(6-methylimidazo[1 ,2- a]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[5-(1 ,3-Benzoxazol-2-yl)-3-furanyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(2,4-Dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-imidazo[1 ,2-a]pyridin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dimethylphenyl)carbonyl](1-rnethylethyl)amino]-1-(4-furo[3,2-ό]pyridin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid;
3-[[(4-Bromo-2-fluorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(4-Bromo-2-methylphenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
S-t^-Chloro^-fluorophenyOcarbony^i-methylethyOarninol-i^-pyrazoloII .S-alpyrimidin^- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dimethylphenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[(1 -Benzofuran-5-ylcarbonyl)(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(4-Bromo-2-chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(2-Bromo-4-chlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; i-μ-CS-Amino-θ-chloro-S-pyridinyOphenyll-S-IKfrans^-methylcyclohexyOcarbony^i- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(3-quinolinyl)phenyl]-1H- pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6-quinoxalinyl)phenyl]-1H- pyrazole-4-carboxylic acid;
1-[4-(4-Amino-6-quinolinyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; i-μ-Cδ-lsoquinolinyOphenyll-S-^frans^-methylcydohexyOcarbony^i-methylethyOamino]-
1 H-pyrazole-4-carboxylic acid;
S-^frans^-MethylcyclohexyOcarbony^i-methylethyOarninol-i-^β-quinolinyOphenyll-IH- pyrazole-4-carboxylic acid;
3-[[(fraπs-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(7-methylfuro[3,2- ό]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-{3-Chloro-4-[6-(methyloxy)-3-pyridinyl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(7-Hydroxy-1 -benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; ^^-(S-Amino-i-benzofuran^-yOphenyll-S-^frans^-methylcyclohexyOcarbony^i- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Aminopyrazolo[1,5-a]pyrimidin-2-yl)-3-chlorophenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(^ans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-[1 ,3]oxazolo[4,5-c]pyridin- 2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-{3-Chloro-4-[5-(methyloxy)-3-pyridinyl]phenyl}-3-[[(trans-4- methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Hydroxy-3-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Acetyl-3-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-{4-[6-Amino-5-(hydroxymethyl)-3-pyridinyl]phenyl}-3-[[(trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[5-(Aminocarbonyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[5-(Hydroxymethyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[6-(Hydroxymethyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[6-(Aminocarbonyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(trans-4-Methylcyclohexyl)carbonyl](1-methylethyl)arnino]-1-[4-(1 H-pyrazolo[3,4-b]pyridin-
5-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-{4-[5-(Aminocarbonyl)-2-furanyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[{[2Λ-Bis(methyloxy)phenyl]carbonyl}(1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin- 2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-[4-(6-Ethyl-7-hydroxy-5-methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Acetyl-2-thienyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-
1 H-pyrazole-4-carboxylic acid; 1-(4-{2-[(1 R)-1-Hydroxyethyl]-1 ,3-thiazol-5-yl}phenyl)-3-[[(trans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Cyano-2-furanyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[2-(Aminocarbonyl)-3-thienyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[4-(Aminoc«rbonyl)-2-pyridinyl]phenyl}-3-[[(trans-4-rnethylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[6-(Hydroxymethyl)-2-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -[4-(5-Acetyl-2-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[5-(Hydroxymethyl)-2-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(6-Acetyl-3-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(^ans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-{4-[6-(methyloxy)-3- pyridinyl]phenyl}-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)annino]-1-(4-pyrido[2,3-b]pyrazin-7- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[{[4-Chloro-2-(methyloxy)phenyl]carbonyl}(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 A/-pyrazole-4-carboxylic acid; 1 -^-(e-Chloro-Z-hydroxy-S-methylpyrazolofi ,5-a]pyrimidin-2-yl)phenyl]-3-[[(fr-aA?s-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
and salts, solvates and esters, and individual enantiomers thereof where appropriate.
Also included in the present invention are pharmaceutically acceptable salt complexes. The present invention also covers the pharmaceutically acceptable salts of the compounds of Formula (I). Suitable pharmaceutically acceptable salts of the compounds of Formula (I) include acid salts, for example sodium, potassium, calcium, magnesium and tetraalkylammonium and the like, or mono- or di- basic salts with the appropriate acid for example organic carboxylic acids such as acetic, lactic, tartaric, malic, isethionic, lactobionic and succinic acids; organic sulfonic acids such as methanesulfonic, ethanesulfonic, benzenesulfonic and p-toluenesulfonic acids and inorganic acids such as hydrochloric, sulfuric, phosphoric and sulfamic acids and the like.
The present invention also relates to solvates of the compounds of Formula (I), for example hydrates.
The present invention also relates to pharmaceutically acceptable esters of the compounds of Formula (I), for example carboxylic acid esters -COOR, in which R is selected from straight or branched chain alkyl, for example n-propyl, n-butyl, alkoxyalkyl (e.g. methoxymethyl), aralkyl (e.g. benzyl), aryloxyalkyl (e.g. phenoxymethyl), aryl (e.g. phenyl optionally substituted by halogen, -C1-4alkyl or -C^alkoxy or amino); or for example - CH2OC(O)R' or -CH2OCO2R' in which R' is alkyl (e.g. R' is f-butyl). Unless otherwise specified, any alkyl moiety present in such esters preferably contains 1 to 18 carbon atoms, particularly 1 to 4 carbon atoms. Any aryl moiety present in such esters preferably comprises a phenyl group.
In one aspect, the compound of Formula (Ia) is in the form of parent compound, a salt or a solvate.
As used herein, the term "pharmaceutically acceptable" used in relation to an ingredient (active ingredient such as an active ingredient, a salt thereof or an excipient) which may be included in a pharmaceutical formulation for administration to a patient, refers to that ingredient being acceptable in the sense of being compatible with any other ingredients present in the pharmaceutical formulation and not being deleterious to the recipient thereof.
Throughout the specification and the claims which follow, unless the context requires otherwise, the word 'comprise', and variations such as 'comprises' and 'comprising', will be understood to imply the inclusion of a stated integer or step or group of integers but not to the exclusion of any other integer or step or group of integers or steps.
It will further be appreciated that certain compounds of the present invention may exist in different tautomeric forms. All tautomers are contemplated to be within the scope of the present invention.
PROCESSES
Compounds of Formula (I) in which A is hydroxy may be prepared from a compound of Formula (II)
in which A is a protected hydroxy group, for example an alkoxy, benzyloxy or silyloxy group and R1, R2, R3 and R4 are as defined above for Formula (I). For example when A is methoxy or ethoxy, and R1, R2, R3 and R4 are as defined above for Formula (I), by treatment with an appropriate base, for example aqueous sodium hydroxide or lithium hydroxide, optionally in a solvent such as methanol, tetrahydrofuran or combinations thereof. Suitably, the temperature is in the range 25 to 1000C, more preferably 50 to 100°C. Alternatively, when A is methoxy or ethoxy and R1, R2, R3 and R4 are as defined above for Formula (I), by treatment with lithium iodide in a suitable solvent such as pyridine, lutidine or collidine, suitably in the temperature range 100-1700C.
For example when A is tert-butoxy, and R1, R2, R3 and R4 are as defined above for Formula (I), by treatment with an appropriate acid, for example trifluoroacetic acid. Suitably, the reaction is carried out in a solvent, for example dichloromethane. Suitably, the temperature is in the range 0 to 500C, more preferably 15 to 300C.
For example when A is silyloxy, and R1, R2, R3 and R4 are as defined above for Formula (I), by treatment with a suitable fluoride source for example tetrabutylammonium fluoride. The reaction is carried out in a suitable solvent, for example tetrahydrofuran. Suitably, the temperature is in the range 0 to 5O0C, more preferably 15 to 300C.
Compounds of Formula (I) in which A is hydroxy, or (II) in which A is an alkoxy, benzyloxy or silyloxy group may be prepared by reaction of a compound of Formula (III)
in which A is hydroxy or an alkoxy, benzyloxy or silyloxy group, and R1, R3 and R4 are as defined above for Formula (I); with a suitable acylating agent, for example R2-C(0)-Y, wherein Y is a halo atom, for example chloro or bromo, and R2 is as defined above for Formula (I). The reaction may be carried out in a suitable solvent, for example dichloromethane, in the presence of a suitable base, for example pyridine or triethylamine and thereafter removing any protecting group if desired. Where R2 represents an aliphatic group, a phosphine such as triphenylphosphine may optionally be used. Suitable protecting groups can be found, but are not restricted to, those found in T W Greene and P G M Wuts 'Protective Groups in Organic Synthesis', 3rd Ed (1999), J Wiley and Sons.
In a further aspect, a compound of Formula (II) may be prepared by appropriate manipulation of another compound of Formula (II). For example, a compound of Formula (II) in which any substituent comprises -C^alkenyl may be prepared from a suitable aldehyde or ketone substituent and a phosphorous ylid generated from a phosphonium salt in the presence of a suitable base, such as potassium tert-butoxide, in a suitable solvent such as THF. Optionally, the trans and cis isomers may be separated by standard techniques known in the art. For example, a compound of Formula (II) in which any substituent comprises -C2-4alkyl may be prepared by reduction of an alkenyl substituent, for example using hydrogen, optionally under pressure, in the presence of a suitable catalyst such as palladium on carbon, in a suitable solvent such as ethanol.
For example, a compound of Formula (II) in which any substituent comprises -C(O)NRARB may be prepared by reacting a suitable acid substituent with an amine (HNRARB) in the presence of a coupling agent such as HATU, in the presence of a suitable base such as triethylamine, in a suitable solvent such as DMF. Alternatively, a compound of Formula (II) in which any substituent comprises -C(O)NRARB may be prepared by reacting a suitable acid chloride substituent with an amine (HNRARB) in the presence of a suitable base such as triethylamine, in a suitable solvent such as dichloromethane. For example, a compound of Formula (II) in which any substituent comprises -NREC(O)RD may be prepared by reacting a suitable amine substituent with a carboxylic acid (R0CO2H) in the presence of a coupling agent such as HATU, in the presence of a suitable base such as triethylamine, in a suitable solvent such as DMF. Alternatively, a compound of Formula (II) in which any substituent comprises -NREC(O)RD may be prepared by reacting a suitable amine substituent with an acid chloride in the presence of a suitable base such as triethylamine in a suitable solvent such as dichloromethane.
For example, a compound of Formula (II) in which any substituent comprises -NRESO2RD may be prepared by reacting an amine substituent with a suitable sulfonyl chloride in the presence of a suitable base such as triethylamine, in a suitable solvent such as dichloromethane. For example, a compound of Formula (II) in which any substituent comprises -SO2NRFRG may be prepared by reacting a sulfonyl chloride substituent with a suitable amine (HNRFRG) in the presence of a suitable base such as triethylamine, in a suitable solvent such as dichloromethane. For example, a compound of Formula (II) in which any substituent comprises -(CH2)nSRA, wherein n=0, 1 , 2, 3 or 4 may be prepared by reacting a thiol -(CH2)nSH substituent or a thiolate salt (for example sodium thioalkoxide) substituent with an alkyl halide RAX wherein X is a halo atom such as bromo, in a suitable solvent such as DMF, in the presence of a suitable base such as triethylamine. For example, a compound of Formula (II) in which any substituent comprises -SO2RA may be prepared by oxidation of a compound in which a substituent represents -SRA, using for example oxone, sodium periodate, 3-chloro perbenzoic acid, or hydrogen peroxide. For example, a compound of Formula (II) in which any substituent comprises -(CH2)nORA may be prepared by reacting an alcohol -(CH2)nOH substituent, wherein n=0, 1 , 2, 3, or 4, with an alkyl halide RAX wherein X is a halo atom such as bromo in the presence of a suitable base such as triethylamine or sodium hydride, or a compound of Formula (II) in which any substituent comprises -(CH2)PORA may prepared by reacting an alkyl halide -(CH2)pX substituent, where p=1 , 2 or 3 and X is a halo atom such as bromo, with an alcohol RAOH, optionally in the presence of a base such as triethylamine or sodium hydride, or with an alkoxide (for example sodium methoxide) in a suitable solvent such as DMF. For example, a compound of Formula (II) in which any substituent comprises -(CH2)nNRARB, where n=0,1 , 2, 3, or 4, may be prepared by reacting an amine -(CH2)nNHRB substituent with an alkyl halide RAX wherein X is a halo atom such as bromo, in the presence of a suitable base such as triethylamine or sodium hydride, or a compound of Formula (II) in which any substituent comprises -(CH2)nNRARB wherein n=0,1 , 2, 3, or 4, may be prepared by reacting an aldehyde substituent with an amine, in the presence of a reducing agent such as sodium triacetoxyborohydride, in a suitable solvent such as dichloromethane.
Compounds of Formula (III) in which A is an alkoxy, benzyloxy or silyloxy group may be prepared from compounds of Fo
Formula (I), by treatment with a suitable vinyl ether, or a suitable aldehyde or a suitable
ketone in the presence of a suitable acid, such as acetic acid, and a suitable reducing agent such as sodium triacetoxyborohydride, in a suitable solvent such as dichloromethane. Alternatively, compounds of Formula (III) in which A is an alkoxy, benzyloxy or silyloxy group may be prepared from compounds of Formula (IV) in which A is an alkoxy, benzyloxy or silyloxy, and R1 and R4 are as defined above for Formula (I), by treatment with a suitable alkylating agent R3-X where X is a halo group such as chloride, bromide or iodide, or X is a sulphonate ester such as methanesulfonate, in suitable solvent such as dimethylformamide in the presence of a suitable base such as triethylamine.
Compounds of Formula (III) in which A is hydroxy may be prepared from compounds of Formula (III) in which A is an alkoxy, benyloxy or silyloxy group, for example by treatment with an appropriate base, acid or fluoride source as described in relation to the preparation of compounds of Formula (I) from compounds of Formula (II).
Compounds of Formula (IV) may be prepared by reaction of compounds of Formula (V)
R1— O .COA
Ri-NHNH2 (V) R4>=<CN (Vl)
in which R1 is as defined above for Formula (I) with compounds of Formula (Vl) in which R4 and A are as defined above for Formula (II) and R' is -C1-4alkyl (such as ethyl) in a suitable solvent such as ethanol or xylene, preferably in the temperature range 50-750C.
Compounds of Formula (IV) may also be prepared by reaction of compounds of Formula (VII)
Ri-NH-N=P (VII)
in which R1 is as defined above for Formula (I) and P is a suitable nitrogen protecting group such as benzylidine, with compounds of Formula (Vl) in a suitable solvent such as ethanol or xylene, preferably in the temperature range 50-750C. This is followed by treatment with a suitable acid, such as hydrochloric acid, in a suitable solvent, such as ethanol to effect removal of the protecting group and cyclisation.
Compounds of Formula (V), (Vl) and (VII) are commercially available or well known in the art.
Compounds of Formula (II) may also be prepared by reaction of a compound of Formula (VIII)
in which A is an alkoxy, benzyloxy or silyloxy group, and R1, R2 and R4 are as defined above for Formula (I); with a suitable alkylating agent R3-X in which X is a halo atom such as chloro, bromo or iodo, or X is a sulphonate ester such as methanesulfonate, in a suitable solvent such as dimethylformamide, in the presence of a suitable base such as sodium hydride or sodium f-butoxide, optionally in the presence of triethylamine.
Compounds of Formula (VIII) may be prepared from compounds of Formula (IV) by reaction with a suitable acylating agent, for example R2-C(O)-Y, in which Y is a halo atom, preferably chloro or bromo, and R2 is as defined above for Formula (I). Suitably, the reaction is carried out in a suitable solvent, for example dichloromethane, optionally in the presence of a suitable base, for example pyridine or triethylamine. For example, where R2 represents an aliphatic group, a phosphine such as triphenylphosphine may optionally be used in place of an amine base.
Compounds of Formula (I) in which A is hydroxy, or (II) in which A is an alkoxy, benzyloxy or silyloxy group, may be prepared by reaction of a compound Formula (IX)
in which A is a hydroxyl or an alkoxy, benzyloxy or silyloxy group, and R2, R3 and R4 are as defined above for Formula (I); by treatment with an aryl or heteroaryl boronic acid (Rγ-Rx- boronic acid) in the presence of a copper reagent such as copper (II) acetate. Suitably, the reaction is carried out in the presence of a base, such as pyridine, in air, and in a suitable solvent such as dichloromethane or THF, and at a temperature in the range 10-300C. Alternatively, compounds of Formula (II) may be prepared by reaction of compounds of Formula (IX) with an aryl or heteroaryl halide or triflate (Rγ-Rx-halide or Rγ-Rx-triflate) in the presence of a copper catalyst such as copper (I) iodide. Suitably, the reaction is carried out in the presence of a base such as potassium carbonate, and in the presence of a reagent such as trans-λ ,2-diaminocyclohexane or trans-N,N'-dimethyl-1 ,2-cyclohexanediamine, or a combination thereof in a suitable solvent such as dioxane, DMF or pyridine or a combination thereof, and at a temperature in the range 90-1800C. This reaction can be suitably be performed in a microwave oven.
Compounds of Formula (I) in which A is hydroxy, or (II) in which A is an alkoxy, benzyloxy or silyloxy group, may be prepared by reaction of a compound of Formula (II)'
in which Z represents a chloro, bromo or iodo substituent, and Rx, R2, R3, R4 and A are as defined for Formula (II) by reaction with a suitable heteroaryl boronic acid, Rγ-boronic acid, in the presence of a palladium catalyst such as palladium (II) acetate, a reagent such as 2- dicyclohexylphosphino-2'(N,N-dimethylamino)-biphenyl, and an additional reagent such as cesium fluoride, in a suitable solvent such as dioxane.
Compounds of Formula (I) in which A is hydroxy, or (II) in which A is an alkoxy, benzyloxy or silyloxy group, may also be prepared by reaction of a compound of Formula (II)'
in which Z represents B(OH)2, and Rx, R2, R3, R4 and A are as defined for Formula (II), by reaction with a suitable heteroaryl halide Rγ-hal, in which suitably the halide is bromo or iodo, in the presence of a palladium catalyst such as palladium (II) acetate, a reagent such as 2- dicyclohexylphosphino-2'(N,N-dimethylamino)-biphenyl, and an additional reagent such as cesium fluoride, in a suitable solvent such as dioxane.
Compounds of Formula (II)' in which Z is halo may be prepared by reaction of a compound of Formula (IX) with a boronic acid of Formula Z-Rx-boronic acid under the conditions described above for the preparation of compounds of Formula (I) and (II) from (IX) and Rγ-Rx-boronic acid.
Boronic acids Z-Rx-boronic acid, Rγ-Rx-boronic acid and Rγ-boronic acid are commercially available or may be prepared by analogy to methods provided in Organometallics (1983) 2, 1316, Chem Revs. (1995) 95, 2457, Journal of Org Chem (2004) 69, 1999, SynLett (2004) (5), 892, Bioorg Med Chem (2005) 13, 2305, Tetrahedron Letters (2004) 44, 9359 and Tetrahedron Letters (2005) 45, 6657.
Compounds of Formula (I) or (II) in which R1 represents a 4-(furopyridine)phenyl, may be prepared by treatment of a compound of Formula (II)"
in which R1 represents a 4-ethynylphenyl derivative, and R2, R3, R4 and A are as defined above for Formula (II), with a suitable pyridine (the pyridine being substituted with adjacent hydroxy and iodo groups), with a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide, in a suitable solvent such as triethylamine or DMF. Suitably the temperature is in the range 50-80 C. For examples of furopyridine synthesis see Bioorganic and Medicinal Chemistry Letters (2002) 12, 1399, Synthesis (1986) 749.
Many compounds of Formula (I) or (II) in which R1 represents a 4-(pyrrolopyridine)phenyl, may be prepared by treatment of a compound of Formula (II)" in which R1 represents 4- ethynylphenyl with an appropriate pyridine (the pyridine being substituted by adjacent amino and iodo groups), in the presence of a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide, in a suitable solvent such as triethylamine. Suitably the temperature is in the range 50-80 C. For examples of pyrrolopyridine synthesis see Heterocycles (1986) 24, 31 , Tetrahedron (2003) 59, 1571 , Synlett (1992) 515.
Compounds of Formula (I) or (II) in which R1 represents phenyl substituted by a 4- imidazo[1 ,2-a]pyridine-2-yl, may be prepared by analogy to methods described in Tetrahedron Letters (2001) 42, 3077.
Compounds of Formula (I) or (II) in which R1 represents a 4-(1-H-benzimidazol-2-yl)phenyl derivative may be prepared by analogy to methods described in J. Heterocyclic Chem. (1994) 31 , 957.
Compounds of Formula (I) or (II) in which R1 represents a 4-(1 ,3-benzoxazol-2-yl)phenyl, then derivative may be prepared by analogy to methods described in Tetrahedron Letters (2003) 44, 175.
Compounds of Formula (I) or (II) in which R1 represents a 4-(1 ,3-benzothiazol-2-yl)phenyl, then derivative may be prepared by analogy to methods described in Tetrahedron Letters (2003) 44, 175 or Synth. Commun. (1990) 20, 3379.
Compounds of Formula (I) or (II) in which R1 represents a 4-(oxazolopyridine)phenyl may be prepared by reacting a compound of Formula (II)" in which R1 represents 4-carboxyphenyl with an appropriate pyridine derivative (the pyridine being substituted with adjacent amino and hydroxyl groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-200 0C (see for example J. Med. Chem. (1978) 21 , 1158). Alternatively, the acid chloride of the 4-carboxyphenyl may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) in a microwave reactor in a suitable solvent such as dioxan (see for example Tetrahedron Letters (2003) 44, 175). Compounds of Formula (II) in which R1 represents a 4- (oxazolopyridine)phenyl may also be prepared by reacting the 4-carboxyphenyl derivative with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
Compounds of Formula (I) or (II) in which R1 represents a 4-(thiazolopyridine)phenyl, may be prepared by reacting a compound of Formula (II)" in which R1 represents 4-phenyl-COCI with an appropriate pyridine (the pyridine being substituted with adjacent amino and chloro groups), in the presence of a suitable base such as pyridine, and then in a second step cyclised using a reagent such as Lawesson's reagent in a suitable solvent such as DMPU, at a suitable temperature such as 90-1 10 0C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(thiazolopyridine)phenyl, may be prepared by reacting a compound of Formula (II)" in which R1 represents 4-carboxyphenyl with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-2000C (see for example J. Med. Chem. (1978) 21 , 1158). Alternatively, the acid chloride of the 4-carboxyphenyl may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in a microwave reactor in a suitable solvent such as dioxan (see for example Tetrahedron Letters (2003) 44, 175). In another alternative the 4-carboxyphenyl compound may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
Compounds of Formula (I) or (II) in which R1 represents a 4-(2,3-dihydro-1,1-dioxo-1 ,2- benzisothiazol-2(3H)-yl)phenyl derivative, may be prepared by treatment of a compound of Formula (II)' in which Z represents 4-halo with a 2,3-dihydro-1 ,2-benzisothiazole 1 ,1-dioxide derivative in the presence of copper (I) iodide with a suitable base such as potassium carbonate, and in the presence of a reagent such as trans-λ ,2-diaminocyclohexane or trans- N.N'-dimethyl-i ^-cyclohexanediamine, or a combination thereof in a suitable solvent such as dioxan, DMF or pyridine or a combination thereof, and at a temperature in the range 90- 160 0C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(1-oxo-1 ,3-dihydro-2H-isoindol-
2-yl)phenyl derivative, may be prepared by treatment of a compound of Formula (II)" in which R1 represents a 4-aminophenyl derivative with a phenyl-1 ,2-di-aldehyde derivative in acetic acid optionally with a suitable solvent such as dichloromethane. Alternatively, compounds of Formula (I) or (II) in which in which R1 represents a 4-(1-oxo-1 ,3-dihydro-2H- isoindol-2-yl)phenyl derivative may also be prepared by treatment of a compound of Formula
(II)" in which R1 represents 4-aminophenyl with a suitable phenyl derivative (this being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine, in a suitable solvent such as acetonitrile.
Compounds of Formula (I) or (II) in which R1 represents a 4-((1-oxo-1 ,3-dihydro-2H-isoindol- 2-yl)pyridine)phenyl derivative, may be prepared by treatment of a compound of Formula (II)" in which R1 represents 4-aminophenyl with an appropriate pyridine derivative (the pyridine being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine in a suitable solvent such as acetonitrile.
Compounds of Formula (I) or (II) in which R1 represents a 4-(pyrazolopyrimidine)phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents 4-(5-amino-1 H- pyrazole)phenyl with 1 ,1 ,3,3-tetramethoxypropane in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-1100C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(5-methylpyrazolopyrimidine)- phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents 4- (5-amino-1 H-pyrazole)phenyl with 4,4-dimethoxy-2-butanone in a suitable solvent such as ethanol, suitably the temperature is in the range 60-70°C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(7-methylpyrazolopyrimidine)- phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents 4- (5-amino-1 H-pyrazole)phenyl with 4,4-dimethoxy-2-butanone in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-1100C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(6- methylpyrazolopyrimidine)phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents 4-(5-amino-1 H-pyrazole)phenyl with 1 ,1 ,3,3-tetraethoxy-2- methylpropane in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-110°C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(6- flouropyrazolopyrimidine)phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents 4-(5-amino-1 H-pyrazole)phenyl with 3-diethylamino-2-fluoro-2-propenal in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-1100C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(7- aminopyrazolopyrimidine)phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents 4-(5-amino-1 H-pyrazole)phenyl with 3-dimethylamino acrylonitrile in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-11O0C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(imidazo[1 ,3]thiazol-6-yl)phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents a 4- (bromoacetophenone) with 2-aminothiazole in a suitable solvent such as ethanol or acetone, suitably the temperature is in the range 50-700C.
Compounds of Formula (I) or (II) in which R1 represents a 4-(imidazo[1 ,2-a]pyridine-2- yl)phenyl, may be prepared by treating a compound of Formula (II)" in which R1 represents a 4-(bromoacetophenone) with 2-aminopyridine in a suitable solvent such as ethanol or acetone, suitably the temperature is in the range 50-700C.
A 4-(imidazo[1 ,3]thiazol-6-yl)phenyl derivative may be prepared by analogy to methods described in Journal of European Chemistry (1995) 30, 901 or JCS Perkin 1 (1989) 643.
Suitable methods for the preparation of compounds the above discussed Rγ derivatives may be found in the chemical literature, for example those described in Comprehensive Heterocyclic Chemistry, Edited by A.R. Katritzky and CW. Rees, Pergamon 1984, and Heterocyclic Chemistry, Edited by J.A. Joules and K. Mills, 4th Ed, Blackwell Science.
Compounds of Formula Rγ-Rx-hal or Rγ-hal for use in the preparation of compounds of Formula (II) are available commercially or may be prepared by methods well known in the art. Some examples of heteroaryl halide preparation are given below (but are not limited to these examples).
A 2-(4-bromophenyl)-2H indazole derivative may be prepared by analogy to methods described in Farmaco Ed Sci (1981) 36, 1037 or J. Chem Soc. Perkin Trans 2 (1975), 1185, for example by treating 2-nitrobenzaldehyde with 4-bromoaniline in a suitable solvent such as methanol, and in a separate step reacting the imine with a phosphite such as triethylphosphite, in a microwave reactor at 210°C for 20 mins.
A 2-(4-bromophenyl)imidazo[1 ,2-a]pyridine derivative may be prepared by analogy to methods described in Tetrahedron Letters (2001) 42, 3077.
A 2-(4-bromophenyl)-1H-benzimidazole derivative may be prepared by analogy to methods described in J. Heterocyclic Chem. (1994) 31 , 957.
A 2-(4-bromophenyl)-1 H-benzoxazole derivative may be prepared by analogy to methods described in Tetrahedron Letters (2003) 44, 175.
A 2-(4-bromophenyl)-1H-benzothiazole derivatives may be prepared by analogy to methods described in Tetrahedron Letters (2003) 44, 175 or Synth. Commun. (1990) 20, 3379.
A 4-(furopyridine)phenyl bromide, may be prepared by treatment of a 4-ethynylphenyl bromide with a suitable pyridine (the pyridine being substituted with adjacent hydroxy and iodo groups), with a suitable catalyst such as bis(triphenylphosphine)palladium (II) chloride and copper (I) iodide, in a suitable solvent such as triethylamine or DMF. Suitably the temperature is in the range 50-80 0C. For examples of furopyridine synthesis see Bioorganic and Medicinal Chemistry Letters (2002) 12, 1399, Synthesis (1986) 749.
A 4-(oxazolopyridine)phenyl derivative may be prepared by reacting a 4-carboxyphenyl bromide with an appropriate pyridine derivative (the pyridine being substituted with adjacent amino and hydroxyl groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-2000C (see for example J. Med. Chem. (1978) 21, 1158).
Alternatively, the acid chloride of the 4-carboxyphenyl bromide may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) in a microwave reactor in a suitable solvent such as dioxane (see for example
Tetrahedron Letters (2003) 44, 175). A 4-(oxazolopyridine)phenyl may also be prepared by reacting a 4-carboxyphenyl bromide derivative with an appropriate pyridine (the pyridine being substituted with adjacent amino and hydroxyl groups) using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
A 4-(thiazolopyridine)phenyl bromide may be prepared by reacting an appropriate 4- carboxyphenyl bromide with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in the presence of an acid such as polyphosphoric acid at temperatures in the range 180-2000C (see for example J. Med. Chem. (1978) 21, 1158). Alternatively, the acid chloride of the 4-carboxyphenyl may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), in a microwave reactor in a suitable solvent such as dioxan (see for example Tetrahedron Letters (2003) 44, 175). In another alternative the 4-carboxyphenyl bromide may be reacted with an appropriate pyridine (the pyridine being substituted with adjacent amino and thiol groups), using a suitable coupling agent such as HATU, and then in a second step cyclised using an appropriate reagent such as phosphorous oxychloride.
A 4-(thiazolopyridine)-phenyl bromide may be prepared by reacting an appropriate 4- bromophenyl-COCI with an appropriate pyridine (the pyridine being substituted with adjacent amino and chloro groups), in the presence of a suitable base such as pyridine, and then in a
second step is cyclised using a reagent such as Lawesson's reagent in a suitable solvent such as DMPU1 at a suitable temperature such as 90-11O0C.
A 4-(1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl bromide derivative may be prepared by treatment of an appropriate 4-aminophenyl bromide derivative with a phenyl-1 ,2-di-aldehyde derivative in acetic acid optionally with a suitable solvent such as dichloromethane. A 4-(1- oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl bromide derivative may also be prepared by treatment of an appropriate 4-aminophenyl bromide with a suitable phenyl derivative (this being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine, in a suitable solvent such as acetonitrile.
A 4-(1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)pyridine bromide may be prepared by treatment of an appropriate 4-aminophenyl bromide with an appropriate pyridine derivative (the pyridine being substituted with adjacent methyl ester and bromomethyl groups), in the presence of a suitable base such as diisopropylethylamine in a suitable solvent such as acetonitrile.
A 4-(pyrazolopyrimidine)phenyl bromide may be prepared by treating a 3-(4-bromophenyl)- 1 H-pyrazole-5-amine with 1 ,1 ,3,3-tetramethoxypropane in a suitable solvent such as acetic acid, suitably the temperature is in the range 90-1100C.
Compounds of Formula (IX) in which A is an alkoxy, benzyloxy or silyloxy group may be prepared by deprotection of a compound of Formula (X)
in which A is an alkoxy, benzyloxy or silyloxy group, and R2, R3 and R4 are as defined above for Formula (I); and P is a suitable protecting group. Suitable protecting groups include, but are not restricted to, benzyl and tert-butyloxycarbonyl . Benzyl groups can be removed by hydrogenation using hydrogen gas with a catalyst such as palladium on carbon in a suitable solvent such as ethanol, optionally in the presence of a suitable acid, for example hydrochloric acid, and optionally conducting the reaction under pressure, tert- Butyloxycarbonyl groups may be removed using an acid such as hydrochloric acid or trifluoroacetic acid.
It will be understood by those skilled in the art that compounds of Formula (X) may be prepared from compounds of Formulae (III), (IV) or (VIII) in which the group R1 is a protecting group instead of a group as defined for Formula (I), by application of the synthetic routes described above in relation to the synthesis of compounds of Formula (II).
Esters of compounds of Formula (I), in which A is -OR where R is selected from straight or branched chain alkyl, aralkyl, aryloxyalkyl, or aryl, may also be prepared by esterification of a compound of Formula (I) in which A is hydroxy by standard literature procedures for esterification.
It will be appreciated that compounds of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) which exist as diastereoisomers may optionally be separated by techniques well known in the art, for example by column chromatography or recrystallisation. For example, the formation of an ester using a chiral alcohol, separation of the resulting diastereoisomers, and subsequent hydrolysis of the ester to yield the individual enantiomeric acid of Formula (I) (II), (III), (IV), (VIII), (IX) and (X).
It will be appreciated that racemic compounds of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) may be optionally resolved into their individual enantiomers. Such resolutions may conveniently be accomplished by standard methods known in the art. For example, a racemic compound of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) may be resolved by chiral preparative HPLC. Alternatively, racemic compounds of Formula (I), (II), (III), (IV), (VIII), (IX) and (X) which contain an appropriate acidic or basic group, such as a carboxylic acid group or amine group may be resolved by standard diastereoisomeric salt formation with a chiral base or acid reagent respectively as appropriate. Such techniques are well established in the art. For example, a racemic compound may be resolved by treatment with a chiral acid such as (R)-(-)-1 ,1 '-binaphthyl-2,2'-diyl-hydrogen phosphate or (-)-di-O,O'-p-tolyl-L-tartaric acid, in a suitable solvent, for example isopropanol. The free enantiomer may then be obtained by treating the salt with a suitable base, for example triethylamine, in a suitable solvent, for example methyl tert-butyl ether. Alternatively, racemic acid compounds may be resolved using a chiral base, for example (S)-alpha methylbenzylamine, (S)-alpha phenylethylamine, (1S, 2S)-(+)-2-amino-1-phenyl-1,3-propane-diol, (-) ephidrine, quinine, brucine. Individual enantiomers of Formula (II), (III), (IV), (VIII), (IX) and/or (X) may then be progressed to an enantiomeric compound of Formula (I) by the chemistry described above in respect of racemic compounds.
With appropriate manipulation and protection of any chemical functionality, synthesis of compounds of Formula (I) is accomplished by methods analogous to those above and to those described in the Experimental section. Suitable protecting groups can be found, but are not restricted to, those found in T W Greene and P G M Wuts 'Protective Groups in Organic Synthesis', 3rd Ed (1999), J Wiley and Sons.
Various of the synthetic procedures described above in general terms (and below in specific terms) may involve heating the reactants. It will be appreciated that heating may be carried out by various conventional methods but also with use of a microwave oven.
EXAMPLES ABBREVIATIONS
STRATA cartridge Dual action SPE cartridge available from Phenomenex
SPE solid phase extraction column
TFA trifluoroacetic acid
HPLC high pressure liquid chromatography
DCM dichloromethane
DMF N.N-dimethylformamide
THF tetrahydrofuran
EtOAc ethyl acetate
AcOH acetic acid
DME 1 ,2-dimethoxyethane
DMPU 1 ,3-dimethyl-3,4,5,6-tetrahydro-2(1 H)-pyrimidinone
OASIS HLB cartridge Sample extraction cartridge available from Waters h hours
HATU O-(7-Azabenzotriazol-1 -yl)-N,N, N', N'-tetramethyluronium- hexafluorophosphate
DPPF diphenylphosphinoferrocene
TBDMS tert-butyldimethylsilyl
HCI hydrochloric acid
RT room temperature
DMAP 4-(Dimethylamino)pyridine
SCX-2 propylsulphonic acid ion exchange cartridge
NH2 SPE aminopropyl ion exchange cartdridge
MDAP HPLC reverse phase HPLC on a C18 column using a two-solvent gradient elution with (A) water containing formic acid (0.1%) and (B) acetonitrile-water (95:5 v/v) containing formic acid
(0.05%) as the eluents, and analysis of the fractions by electrospray mass spectroscopy.
ISCO Companion Automated flash chromatography equipment with fraction analysis by UV absorption available from Presearch. Greenhouse A greenhouse parallel synthesiser as manafactured by Radleys
Discovery Biotage Biotage flash equipment for use with pre-packed silica cartridges
All mass spectroscopy was performed using electrospray as the method of ionisation.
Intermediate 1
To a solution of absolute alcohol (200 mL) was added a sodium ethoxide solution (2.1 M in ethanol, 212 mL) followed by the ethyl 3-amino-1H-pyrazole-4-carboxylate (100 g) with stirring. To this solution was added benzyl chloride (80 g) dropwise. After completion of the addition, the mixture was heated at a temperature of 8O0C under a nitrogen atmosphere with stirring. After 1 h the mixture was filtered under vacuum while at elevated temperature. The filtrate was then reduced to approx. % volume and allowed to cool. The product crystallized out and was dried under high vacuum to give the title compound. MS calcd for (C13H15N3O2 + H)+: 246 MS found (electrospray): (M+H)+ = 246 Ref 1 Chem. Pharm. Bull.; 1972, 20(2), 391-398
Intermediate 2 Ethyl 3-[(1 -methylethyl)amino]-1 -(phenylmethyl)-i W-pyrazole-4-carboxylate
To a stirred solution of the amino Intermediate 1 (15 g) in dry dichloromethane (300 mL) under nitrogen was added sequentially, 2-methoxy propene (23.4 mL), glacial acetic acid (14 mL), followed by sodium triacetoxyborohydride (25.8 g) portionwise. The mixture was stirred at room temperature for 2.5 h. The reaction mixture was then diluted with water (300 mL) and the pH of the reaction adjusted to 7 using solid sodium bicarbonate with care (effervescence). The dichloromethane was then separated and the aqueous layer re- extracted with dichloromethane (100 mL). The combined organics were washed with brine, dried over sodium sulphate, filtered and evaporated. The crude product was then loaded onto a 400 g Biotage column (silica) and eluted with cyclohexane / ethyl acetate (9:1). Fractions containing the required compound were evaporated to give the title compound. MS calcd for (C16H21N3O3 + H)+: 288 MS found (electrospray): (M+H)+ = 288
Intermediate 3
Ethyl 3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(phenylmethyl)- 1 W-pyrazole-4-carboxylate
To a stirred solution of Intermediate 2 (20 g) and triphenylphosphine (36.5 g) in dry dichloromethane (100 mL) was added a solution of frans-4-methylcyclohexanecarbonyl chloride2 (18.5 g) in dry dichloromethane (100 mL). The mixture was heated at reflux with a bath temperature of 6O0C under a nitrogen atmosphere. After 16 h a further portion of trans- 4-methylcyclohexanecarbonyl chloride (4.8 g) was added and heating at reflux continued for 16 h. The mixture was cooled to room temperature and saturated sodium bicarbonate solution (200 mL) was added cautiously to the vigorously stirred reaction mixture. The organic phase was separated, dried over Na2SO4, filtered and evaporated to a yellow oil. This was purified by flash chromatography over silica (Biotage, 40Og silica cartridge) eluting with DCM/cyclohexane (1 :1 ), followed by cyclohexane/ethyl acetate (7:1 ). Product fractions were combined and evaporated to give a solid which was taken up in ethanol (400 mL) and in two portions passed through 2x70 g aminopropyl capped silica SPE columns. Product fractions were combined, evaporated and dried under high vacuum to give the title compound.
MS calcd for (C24H33N3O3 + H)+: 412 MS found (electrospray): (M+H)+ = 412 Ref 2: WO 2004/052885.
Intermediate 4
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4- carboxylate
A solution of Intermediate 3 (24 g) in ethanol (750 mL) was stirred with cone HCI (15 mL) and a catalyst of 10% palladium on carbon (7.5 g, wetted) under an atmosphere of hydrogen.
After 24 h, some starting material remained. The catalyst was removed by filtration through
Celite under a stream of nitrogen. The filtrate was added to fresh catalyst (7 g) and stirred under hydrogen atmosphere for 18 h. Catalyst was removed by filtration through Celite under a stream of nitrogen and the filtrate was evaporated under vacuum. The residue was dissolved in dichloromethane and stirred with saturated sodium bicarbonate solution. The organic phase was separated, dried over Na2SO4, filtered and evaporated to give the title compound.
MS calcd for (C17H27N3O3 + H)+: 322 MS found (electrospray): (M+H)+ = 322
Intermediate 5
Ethyl 1 -(4-bromophenyl)-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
Intermediate 4 (2.0 g) was dissolved in DCM (60 ml_) and (4-bromophenyl)boronic acid (2.5 g) added. Pyridine (1 mL) was added followed by copper (II) acetate (1.69 g). The reaction mixture was stirred at room temperature for 24 h and then diluted with DCM (60 mL) and washed with 2M aqueous HCI solution, saturated sodium bicarbonate solution, brine, dried (Na2SO4) and concentrated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C23H30BrN3O3 + H)+: 476/478 MS found (electrospray): (M+H)+ = 476/478
Intermediate 6
Ethyl 1 -[4-(1 -benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
Intermediate 5 (200 mg), 1-benzofuran-2-ylboronic acid (102 mg), cesium fluoride (191 mg), palladium (II) acetate (18.8 mg) and 2-dicyclohexylphosphino-2'-(N,N-dimethylamino)- biphenyl (50 mg) were dissolved in dioxane (4 mL) and stirred at room temperature for 24 h. More 1-benzofuran-2-ylboronic acid (102 mg) was added and the mixture stirred at room temperature for 24 h. The reaction mixture was diluted with water and extracted with ethyl acetate. The organic extract was washed with saturated sodium bicarbonate solution, brine, dried (Na2SO4) and concentrated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give a mixture containing the title compound. Further purification by MDAP HPLC gave the title compound. MS calcd for (C3IH35N3O4 + H)+: 514 MS found (electrospray): (M+H)+ = 514
Intermediate 7
Ethyl 1 -[4-(1 Φenzothien^-yOphenyll-S-lKfraπs-A-methylcyclohexyOcarbonylHI - methylethyl)amino]-1W-pyrazole-4-carboxylate
Intermediate 5 (200 mg), 1-benzothien-2-ylboronic acid (112 mg), cesium fluoride (191 mg), palladium (II) acetate (18.8 mg) and 2-dicyclohexylphosphino-2'-(N,N-dimethylamino)- biphenyl (50 mg) were dissolved in dioxane (4 ml.) and stirred at room temperature for 5 days. The solvent was removed in vacuo and the residue partitioned between water and ethyl acetate. The organic extract was washed with brine, dried (Na2SO4) and concentrated to give a gum. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C31H35N3O3S + H)+: 530
MS found (electrospray): (M+H)+ = 530
Intermediate 8
1 ,1 -Dimethylethyl 2-(4-{4-[(ethyloxy)carbonyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazol-1 -yl}phenyl)-1 H-indole-1 - carboxylate
Intermediate 5 (200 mg), 1-(tert-butoxycarbonyl)-1 H-indol-2-ylboronic acid (165 mg), cesium fluoride (191 mg), palladium (II) acetate (18.8 mg) and 2-dicyclohexylphosphino-2'-(N,N- dimethylamino)-biphenyl (50 mg) were dissolved in dioxane (4 mL) and stirred at room temperature for 24 h. The reaction mixture was diluted with water and extracted with ethyl acetate. The organic extract was washed with saturated sodium bicarbonate solution, brine, dried (Na2SO4) and concentrated to give a gum. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C36H44N4O5 + H)+: 613 MS found (electrospray): (M+H)+ = 613
Intermediate 9
Ethyl 1 -(phenylmethyl)-3-(tetrahydro-2H-pyran-4-ylamino)-1 H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 1 (10 g) dissolved in anhydrous DCM (150 mL) was added acetic acid (7 mL). Tetrahydro-4H-pyran-4-one (6.6 mL) was added dropwise followed by portionwise addition of sodium triacetoxyborohydride (17.3 g). The reaction mixture was left to stir under N2 for 20 h and then partitioned between DCM and sat. NaHCO3 solution. The layers were separated using a hydrophobic frit and the organic solvent removed by evaporation. The resultant solid was purified by flash chromatography on silica (Biotage) using cyclohexane and EtOAc (4:1) as elution solvent. The relevant fractions were combined and the solvent removed to give the title compound. MS calcd for (C18H23N3O3 + H)+: 329 MS found (electrospray): (M+H)+ = 329
Intermediate 10
Ethyl 3-[[(4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1- (phenylmethyl)-1H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 9 (11.63 g) dissolved in anhydrous DCM (100 mL) was added triethylamine (7.4 mL) followed by frans-4-methylcyclohexanecarbonyl chloride2 (5.58 g). The reaction was left to stir at reflux for 20 h and then more triethylamine (1.23 mL) and fr-aA7s-4-methylcyclohexanecarbonyl chloride (1.42 g) added. The reaction was left to stir for 16 h at reflux. A further portion of triethylamine (1.74 mL) and trans-4- methylcyclohexanecarbonyl chloride2 (2 g) was added and the reaction left to stir at reflux for a further 4 h. After cooling to room temperature the mixture was washed with 1 M aqueous HCI, water and saturated sodium bicarbonate solution. The organic phase was dried using a hydrophobic frit and the solvent removed by evaporation. The resultant solid was triturated with acetonitrile to give the title compound. MS calcd for (C21H27N3O3 + H)+: 453 MS found (electrospray): (M+H)+ = 453 Ref 2: WO 2004/052885
Intermediate 11
A solution of Intermediate 10 (5.1 g, 11.3 mmol) in ethanol (200 ml_) and concentrated hydrochloric acid (2 mL) was subjected to atmospheric hydrogenation using 10% palladium on carbon catalyst (1.35 g) for 16 h. The mixture was filtered through a Celite pad, the pad washed with ethanol and the combined organics evaporated. The residue was partitioned between DCM and saturated sodium bicarbonate solution, passed through a hydrophobic frit and the organic layer evaporated to give the title compound. MS calcd for (C19H29N3O4 + H)+: 364 MS found (electrospray): (M+H)+ = 364.
Intermediate 12
Ethyl 1-(4-bromophenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2W-pyran-
4-yl)amino]-1H-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 5 using Intermediate 11 to give the title compound. MS calcd for (C25H32BrN3O4 + H)+: 518/520 MS found (electrospray): (M+H)+ = 518/520
Intermediate 13
Ethyl 1 -[4-(1 -benzofuran^-yOphenylJ-S-t^fra/is^-methylcyclohexyOcarbonyl] (tetrahydro-2W-pyran-4-yl)amino]-1H-pyrazole-4-carboxylate
Intermediate 12 (200 mg), 1-benzofuran-2-ylboronic acid (93.7 mg), cesium fluoride (175.9 mg), palladium (II) acetate (18 mg), and 2-dicyclohexylphosphino-2'-(N,N-dimethylamino)- biphenyl (50 mg) were dissolved in dioxane (4 mL) and stirred at room temperature for 4 h.
More palladium (II) acetate (18 mg) and 2-dicyclohexylphosphino-2'-(N,N-dimethylamino)- biphenyl (50 mg) was added and the mixture heated at 8O0C for 1 day. The reaction mixture was diluted with water and extracted with ethyl acetate. The organic extract was washed with saturated sodium bicarbonate solution, brine, dried (Na2SO4) and concentrated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C33H37N3O5 + H)+: 556 MS found (electrospray): (M+H)+ = 556.
Intermediate 14
Ethyl 1 -[4-(1.S-benzoxazol^-yOphenylJ-a-fKfrans^-methylcyclohexyOcarbonyl]
(tetrahydro-2W-pyran-4-yl)amino]-1f/-pyrazole-4-carboxylate
A mixture of Intermediate 11 (100 mg), Intermediate 21 (83 mg), copper (I) iodide (12.5 mg), fraA7s-(1 R,2R)-N,N'-bismethyl-1 ,2-cyclohexanediamine (18 mg) and potassium carbonate (91 mg) in DMF (3 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated to give a gum. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C32H36N4O5 + H)+: 557 MS found (electrospray): (M+H)+ = 557.
Intermediate 15 Ethyl 3-{[(fraπs-4-methylcyclohexyl)carbonyl]amino}-1-(phenylmethyl)-1H-pyrazole-4- carboxylate
To a stirred solution of Intermediate 1 (10 g) dissolved in anhydrous DCM (100 mL) was added frans-4-methylcyclohexanecarbonyl chloride2 (7.87 g) and triethylamine (8.6 mL). The reaction mixture was heated at reflux for 4 h. The reaction mixture was then diluted with DCM and partitioned with saturated NaHCO3 solution. The layers were separated and the organic dried with Na2SO4. The organic solvent was removed by evaporation and the residue triturated with diethyl ether and filtered to give the title compound. MS calcd for (C21H27N3O3 + H)+: 369 MS found (electrospray): (M+H)+ = 369 Ref 2: WO 2004/052885
Intermediate 16
Ethyl 3-{ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(phenyl methyl)-1 H- pyrazole-4-carboxylate
A stirred solution of Intermediate 15 (1.19 g) dissolved in DMF (25 mL) was cooled to O0C before addition of 60% sodium hydride in mineral oil (0.15 g). The reaction was left to stir under nitrogen for 30 mins before addition of ethyl iodide (0.78 mL). The reaction was stirred at room temperature for 3 h. More sodium hydride (50 mg) was added followed by ethyl iodide (0.2 mL) and the reaction left to stir for 16 h. The solvent was removed by evaporation and the resulting gum was partitioned between DCM and water. The layers were separated using a hydrophobic frit and the organic solvent removed by evaporation. The crude material was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C23H3IN3O3 + H)+: 397 MS found (electrospray): (M+H)+ = 397
Intermediate 17 Ethyl 3-{ethyl[(?rans-4-methylcyclohexyl)carbonyl]amino}-1 W-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 11 using Intermediate 16 to give the title compound. MS calcd for (C16H25N3O3 + H)+: 307 MS found (electrospray): (M+H)+ = 307
Intermediate 18
Ethyl 1-[4-(1, 3-benzoxazol-2-y I) phenyl] -3-{ethy I [(frans-4- methylcyclohexyl)carbonyl]amino}-1W-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 14 using Intermediate 17 and Intermediate 21 to give the title compound. MS calcd for (C29H32N4O4 + H)+: 501 MS found (electrospray): (M+H)+ = 501.
Intermediate 19
Ethyl 1 -(4-imidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1AV-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), 2-(4-bromophenyl)imidazo[1 ,2-a]pyridine3 (93 mg), copper (I) iodide (14 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H35N5O3 + H)+: 514 MS found (electrospray): (M+H)+ = 514 Ref 3: Burkholder, Conrad; Dolbier, William R.; Medebielle, Maurice; Ait-Mohand, Samia, Tetrahedron Lett., 42, 17, 2001 , 3077 - 3080.
Intermediate 20
Ethyl 1 -[4-(1 H-benzimidazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), 2-(4-bromophenyl)-1/-/-benzimidazole4 (93 mg), copper (I) iodide (14 mg), trans-{λ R, 2R)-N, N'-bismethyl-1 ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2 mL) was heated at 180°C in a Reactivial for 24 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H35N5O3 + H)+: 514 MS found (electrospray): (M+H)+ = 514
Ref 4: Kauffman, Joel M.; Khalaj, AIi; Litak, Peter T.; Novinski, John A.; Bajwa, Gurdip S, J. Heterocycl. Chem., 31 , 4, 1994, 957-966.
Intermediate 21
2-(4-Bromophenyl)-1,3-benzoxazole
A mixture of 2-aminophenol (196 mg) and 4-bromobenzoyl chloride (438 mg) in dioxane (2.5 mL) was heated in a microwave at 21O0C for 15 mins. The cooled reaction mixture was added to 1 M sodium hydroxide solution (40 mL). The solid was filtered, washed with water and dried in vacuo to give the title compound. MS calcd for (C13H8BrNO + H)+: 274/276 MS found (electrospray): (M+H)+ = 274/276
Intermediate 22
Ethyl 1 -[4-(1 ^-benzoxazol^-yOphenylJ-S-Hffrans^-methylcyclohexyOcarbonylHI - methylethyl)amino]-1H-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 4 and Intermediate 21 to give the title compound. MS calcd for (C30H34N4O4 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 23 2-(4-Bromophenyl)-1 ,3-benzothiazole
A mixture of 2-aminothiophenol (225 mg) and 4-bromobenzoyl chloride (438 mg) in dioxane (2.5 mL) was heated in a microwave reactor at 21O0C for 15 mins. The cooled reaction mixture was added to 1M sodium hydroxide solution (40 mL). The solid was filtered, washed with water and dried in vacuo to give the title compound. MS calcd for (C13H8BrNS + H)+: 290/292 MS found (electrospray): (M+H)+ = 290/292
Intermediate 24 Ethyl 1 -[4-(1 ,3-benzothiazol-2-yl)phenyl]-3-[[(fra/is-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 4 and Intermediate 23 to give the title compound. MS calcd for (C30H34N4O3S+ H)+: 531 MS found (electrospray): (M+H)+ = 531
Intermediate 25 (4-Bromophenyl)[(1£)-(2-nitrophenyl)methylidene]amine
A mixture of 2-nitrobenzaldehyde (2 g) and 4-bromoaniline (2.27 g) in methanol (25 ml.) was heated at reflux for 4 h and then allowed to cool to room temperature. The solid was filtered and dried in vacuo to give the title compound. MS calcd for (C13H9BrN2O2 + H)+: 305/307 MS found (electrospray): (M+H)+ = 305/307
Intermediate 26
2-(4-Bromophenyl)-2H-indazole
Intermediate 25 (210 mg) was dissolved in triethylphosphite (2 ml_) and the mixture heated in a microwave reactor at 21O0C for 15 mins. The triethylphosphite was removed in vacuo and the residue purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C13H9BrN2 + H)+: 273/275 MS found (electrospray): (M+H)+ = 273/275
Intermediate 27
Ethyl 1-[4-(2H-indazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1H-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 4 and Intermediate 26 to give the title compound. MS calcd for (C30H35N5O3+ H)+: 514 MS found (electrospray): (M+H)+ = 514
Intermediate 28 Ethyl 3-{ethyl[(fr3ns-4-methylcyclohexyl)carbonyl]arnino}-1 -(4-imidazo[1 ,2-a]pyridin-2- ylphenyl)-1W-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 17 and 2-(4-bromophenyl)imidazo[1,2-a]pyridine3 to give the title compound.
MS calcd for (C29H33N5O3+ H)+: 500
MS found (electrospray): (M+H)+ = 500
Ref:3: Burkholder, Conrad; Dolbier, William R.; Medebielle, Maurice; Ait-Mohand, Samia,
Tetrahedron Lett., 42, 17, 2001, 3077 - 3080.
Intermediate 29
Ethyl 1-(4-imidazo[1,2-a]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl) carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1W-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 11 and 2-(4-bromophenyl)imidazo[1 ,2-a]pyridine3 to give the title compound.
MS calcd for (C32H37N5O4+ H)+: 556
MS found (electrospray): (M+H)+ = 556
Ref:3: Burkholder, Conrad; Dolbier, William R.; Medebielle, Maurice; Ait-Mohand, Samia,
Tetrahedron Lett., 42, 17, 2001, 3077 - 3080.
Intermediate 30
Ethyl 1 -[4-(1 ,1 -dioxido-1 ,2-benzisothiazol-2(3H)-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A solution of Intermediate 5 (108 mg), 2,3-dihydro-1 ,2-benzisothiazole 1 ,1 -dioxide (35 mg), copper (I) iodide (4 mg), potassium carbonate (58 mg) and (IR^RJ-Λ/./V-dimethyl-i ^- cyclohexanediamine (12 mg) in DMF (4 mL) was heated in a microwave at 100° C, 300 W for 1 h and then evaporated to dryness. The residue was dissolved in ethyl acetate and washed sequentially with aqueous sodium bicarbonate solution, water and 2M HCI, dried using a
hydrophobic frit and evaporated to dryness. The crude product was then purified by MDAP HPLC to give the title compound. MS calcd for (C30H36N4O5 + H)+: 565 MS found (electrospray): (M+H)+ = 565
Intermediate 31
2-(4-Bromophenyl)-2,3-dihydro-1 H-isoindol-1 -one
To 4-bromoaniline (0.50 g) and o-phthaldialdehyde (0.43 g) dissolved in DCM (25 mL) was added glacial acetic acid (5 mL). The reaction was stirred at room temperature for 3 h and then evaporated to dryness. The residue was purified by 5 g Strata cartridge eluted with
DCM to give the title compound.
MS calcd for (C14H10BrNO + H)+: 288/290
MS found (electrospray): (M+H)+ = 288/290
Intermediate 32
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2W-pyran-4-yl)amino]-1 -[4-(1 - oxo-1,3-dihydro-2W-isoindol-2-yl)phenyl]-1H-pyrazole-4-carboxylate
Intermediate 11 (124 mg), Intermediate 31 (108 mg), copper (I) iodide (10 mg), potassium carbonate (93 mg) and (1R,2R)-Λ/,Λ/l-dimethyl-1 ,2-cyclohexanediamine (20 mg) in DMF (4 mL) were heated in a Reactivial at 130° C for 24 h and then evaporated to dryness. The residue was dissolved in ethyl acetate and washed sequentially with aqueous sodium bicarbonate solution, water and 2M HCI, dried with a hydrophobic frit and evaporated to dryness. The residue was purified by 10 g silica SPE cartridge eluting with a gradient of DCM in cyclohexane, then a gradient of EtOAc in DCM to give the title compound. MS calcd for (C33H38N4O5 + H)+: 571 MS found (electrospray): (M+H)+ = 571
Intermediate 33
Ethyl 1 -[4-({[(1 ,1 -dimethylethyl)oxy]carbonyl}amino)phenyl]-3-[[(fra/ιs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A solution of Intermediate 4 ( .50 g), [4-({[(1 ,1-di rmethylethyl)oxy]carbonyl}amino)phenyl] boronic acid (1.66 g), copper (II) acetate (1.68 g) and pyridine (0.75 ml.) in DCM (30 ml_) was stirred at room temperature under nitrogen for 24 h. The solution was washed sequentially with aqueous sodium bicarbonate solution, water and 2M HCI, dried using a hydrophobic frit and evaporated to dryness. This crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C28H40N4O5 + H)+: 513 MS found (electrospray): (M+H)+ = 513
Intermediate 34
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -{4- aminophenyl}-1H-pyrazole-4-carboxylate
To a solution of Intermediate 33 (1.14 g) in dioxane (5 mL) was slowly added a solution of
HCI in dioxane (4N, 10 mL) and the reaction stirred at room temperature for 16 h under nitrogen. A further portion of HCI in dioxane (10 mL) was added and stirring continued for 2 days. The solvents were evaporated and the mixture was partitioned between sodium bicarbonate solution (30 mL, 8% solution) and ethyl acetate and the aqueous layer further extracted with ethyl acetate (3 x 20 mL). The organic extract was dried (Na2SO4), evaporated, and purified by silica SPE, eluted with DCM, then DCM:ethyl acetate (9:1, then 8:2) to give the title compound. MS calcd for (C23H32N4O3 + H)+: 413 MS found (electrospray): (M+H)+ = 413
Intermediate 35
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 -oxo-1 ,3- dihydro-2H-isoindol-2-yl)phenyl]-1H-pyrazole-4-carboxylate
To Intermediate 34 (226 mg) and o-phthaldialdehyde (80 mg) dissolved in DCM (15 mL) was added glacial acetic acid (3 mL). The reaction was stirred at room temperature under nitrogen for 16 h and then evaporated to dryness. The residue was purified by 10 g silica SPE cartridge with a gradient of DCM in cyclohexane, then a gradient of EtOAc in DCM, then by 5g Strata cartridge eluted with DCM and EtOAc and finally by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C3IH36N4O4 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 36
Methyl 5-fluoro-2-methylbenzoate
A solution 5-fluoro-2-methylbenzoic acid (1.0 g) and concentrated sulphuric acid (0.25 mL) in methanol (25 mL) was stirred at reflux for 3 h, neutralised with aqueous sodium bicarbonate solution and evaporated to dryness. The residue was dissolved in ethyl acetate and the solution washed sequentially with aqueous sodium bicarbonate solution and brine, dried with a hydrophobic frit and evaporated to dryness to give the title compound. 1H NMR (CDCI3) δ 7.63 (1 H,dd), 7.22 (1 H,dd), 7.11 (1 H,dt), 3.91 (3H, s), 2.57 (3H,s).
Intermediate 37
Methyl 2-(bromomethyl)-5-fluorobenzoate
To a solution of Intermediate 36 (526 mg) and N-bromosuccinimide (550 mg) in carbon tetrachloride (25 mL) was added benzoyl peroxide (10 mg). The reaction was stirred at reflux under nitrogen for 18 h, cooled to 0° C, filtered and the filtrate evaporated to dryness. The residue was purified on a 20 g silica SPE cartridge with a gradient of DCM in cyclohexane, then a gradient of EtOAc in DCM, to give the title compound. 1H NMR (DMSO) δ 7.73-7.65 (2H, m), 7.52 (1 H, td), 5.04 (2H, s), 3.92 (3H, s).
Intermediate 38
2-(4-Bromophenyl)-6-fluoro-2, e
Intermediate 37 (626 mg), 4-bromoaniline (478 mg) and diisopropylethylamine (665 uL) were dissolved in acetonitrile (15 ml_). The reaction was stirred at room temperature under nitrogen for 16 h and then evaporated to dryness. The residue was purified by Strata cartridge eluted with dichloromethane, then by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. 1H NMR (CD3OD) δ 7.55 (1H, dd), 7.43 (1H, dd), 7.12 (1H, td), 7.07 (2H, d), 6.40 (2H, d), 4.51 (2H, s)
Intermediate 39 2-(4-Bromophenyl)furo[3,2-b]pyridine
To 4-bromophenylactetylene (1.65 g) was added 2-iodo-3-hydroxypyridine (2.02 g), bis(triphenylphosphine)palladium dichloride (575 mg), copper (I) iodide (225 mg) and triethylamine (35 ml_). The reaction was stirred at 5O0C, under nitrogen, for 3 h. The reaction was cooled, diluted with DCM (250 ml_) and washed with saturated ammonium chloride solution, brine and then concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C13H8BrNO + H)+: 274/276
MS found (electrospray): (M+H)+ = 274/276
Intermediate 40
Ethyl 3-{ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-furo[3,2-b]pyridin-2- ylphenyl)-1H-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 17 and Intermediate 39. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane and then further purified by MDAP HPLC to give the title compound. MS calcd for (C29H32N4O4+ H)+: 501 MS found (electrospray): (M+H)+ = 501
Intermediate 41
Ethyl 1 -(4-furo[3,2-/>]pyridin-2-ylphenyl)-3-[[(frans-4- methylcyclohexyOcarbonylKtetrahydro^H-pyran^-ylJaminol-IW-pyrazole^- carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 11 and Intermediate 39. The crude product was purified by ISCO companion reverse phase (C- 18) chromatography using a gradient elution with (A) water containing formic acid (0.1%) and (B) acetonitrile containing formic acid (0.05%) to give the title compound. MS calcd for (C32H36N4O5+ H)+: 557 MS found (electrospray): (M+H)+ = 557
Intermediate 42
2-(4-Bromophenyl)pyrazolo[1,5-a]pyrimidine
A solution of 3-(4-bromophenyl)-1H-pyrazol-5-amine (1.19 g) in acetic acid (20 ml_) was treated with 1 ,1 ,3,3-tetramethoxypropane (615 mg) and the mixture heated at 11O0C for 1.5 h. On cooling to room temperature the precipitated solid was isolated by filtration, washed with water and allowed to dry in air to give the title compound. MS calcd for (C12H8BrN3+ H)+: 274/276 MS found (electrospray): (M+H)+ = 274/276
Intermediate 43
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 4 and Intermediate 42 to give the title compound. MS calcd for (C29H34N6O3+ H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 44
Ethyl 1 -(^iodophenyO^HHfrans^-methylcyclohexyOcarbonyl^i -methylethyl)amino]- 1 H-pyrazole-4-carboxylate
To a solution of Intermediate 4 (4.0 g) in anhydrous DCM (300 ml_) at room temperature was added 4-iodophenyl boronic acid (4.6 g), copper (II) acetate (3.3 g) and pyridine (1.97 ml_). The reaction was stirred at room temperature for 16 h.
The reaction was diluted with DCM (200 ml_) and washed with sat. aq. sodium bicarbonate solution, water, brine, dried (Na2SO4) and evaporated. This was purified by chromatography on silica gel using a cyclohexane/ethyl acetate gradient to give the title compound. MS calcd for (C23H3OlN3CVH)+ : 524 MS found (electrospray): (M+H)+ = 524
Intermediate 45
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -{4- [(trimethylsilyl)ethynyl]phenyl}-1 H-pyrazole-4-carboxylate
A solution of Intermediate 44 (5.12 g) in anhydrous THF (20 ml.) was degassed then treated with trimethylsilyl acetylene (2.5 ml_), triethylamine (5.5 ml_), dichlorobis(triphenylphosphine)palladium (0.36 g) and copper (I) iodide (0.094 g). The reaction was stirred and heated at 550C under nitrogen for 1.5 h. The reaction was allowed to cool, diluted with DCM (70 ml_), filtered and evaporated to give a solid which was purified by chromatography on silica gel using a cyclohexane /ethyl acetate gradient to give the title compound. MS calcd for (C28H39N3O3SiH-H)+ : 494 MS found (electrospray): (M+H)+ = 494
Intermediate 46
Ethyl 1 -(4-ethynylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1/7-pyrazole-4-carboxylate
A solution of Intermediate 45 (4.7 g) in ethanol (90 ml_) was stirred at room temperature and treated with potassium carbonate (0.66 g) and the reaction stirred for 4 h. The reaction was evaporated and the residue dissolved in DCM (200 mL) and washed with saturated aqueous sodium bicarbonate solution, brine, dried (Na2SO4) and evaporated to give a solid which was purified by chromatography on silica gel using a cyclohexane /ethyl acetate gradient to give the title compound. MS calcd for (C25H3IN3CVH)+ : 422 MS found (electrospray): (M+H)+ = 422
Intermediate 47
Ethyl 1 -(4-furo[3,2-ϋ]pyridin-2-ylphenyl)-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
A solution of Intermediate 46 (3.49 g) in triethylamine (25 mL) was divided into 5 equal portions and each portion placed in a Reactivial and each treated with 3-hydroxy-2-iodo- pyridine (0.18 g), copper (I) iodide (0.016 g) and dichlorobis(triphenylphosphine)palladium (0.058 g). The Reactivials were stirred and heated at 7O0C for 3 h, then allowed to cool and the contents of each one combined, diluted with DCM and washed with saturated ammonium chloride solution, brine, dried (Na2SO4) and evaporated to give a foam. The foam was purified by chromatography on silica gel using a cyclohexane /ethyl acetate gradient to give the title compound. MS calcd for (C30H34N4O4+ H)+ : 515 MS found (electrospray): (M+H)+ = 515
Intermediate 48
Ethyl 1 -{3-chloro-4-[(phenylmethyl)oxy]phenyl}-3-[[(trans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
To Intermediate 4 (5 g) was added copper (II) acetate (4.24 g), pyridine (2.46 g) and 4- benzyloxy-3-chlorophenyl boronic acid (8.2 g). The reaction was stirred at room temperature, in air for 16 h. The mixture was then partitioned between DCM and 2N HCI, passed through a hydrophobic frit and the organic phase concentrated. The crude material was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H36CIN3O4 + H)+: 538/540 MS found (electrospray): (M+H)+ = 538/540
Intermediate 49
Ethyl 1-(3-chloro-4-hydroxyphenyl)-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole
Intermediate 48 (6.8 g) in EtOAc (270 ml.) was subjected to atmospheric pressure hydrogenation with 5% Platinum on carbon, degussa type (2 g, 30 wt.%) for 16 h. The mixture was then filtered through Celite and concentrated. The crude product was then purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C23H30CIN3O4 + H)+: 448/50 MS found (electrospray): (M+H)+ = 448/50
Intermediate 50
Ethyl 1-(3-chloro-4-{[(trifluoromethyl)sulfonyl]oxy}phenyl)-3-[[(trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 49 (2 g) and pyridine (0.71 g) in dry DCM (20 mL) at O0C, under a nitrogen atmosphere, was added trifluoromethanesulphonic anhydride (1.39 g).
The reaction was stirred for 30 mins then quenched with saturated sodium bicarbonate solution. The mixture was extracted with ethyl acetate and the organic phase washed with water, 2N HCI, then brine. The solution was dried over sodium sulphate and concentrated to give the title compound. MS calcd for (C23H30CIF3N3O6S + H)+: 580/582 MS found (electrospray): (M+H)+ = 580
Intermediate 51
1 ,1 -Dimethylethyl 2-(2-chloro-4-{4-[(ethyloxy)carbonyl]-3-[[(trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazol-1 -yl}phenyl)-1 H-indole-1 - carboxylate
To Intermediate 50 (100 mg) was added 1-(tert-butoxycarbonyl)indole-2-boronic acid (67.5 mg), cesium fluoride (78.5 mg), palladium (II) acetate (7.7 mg), 2-dicyclohexylphosphino-2'- (N,N-dimethylamino)biphenyl (20 mg) and dry 1 ,4-dioxane (2 ml_). The reaction was stirred at room temperature, under nitrogen, for 24 h. The reaction was diluted with water, extracted with ethyl acetate and the organic phase washed with saturated sodium bicarbonate solution, then brine, dried over sodium sulphate and concentrated. The crude product was then purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C36H43CIN4O5 + H)+: 647/649 MS found (electrospray): (M+H)+ = 647/649
Intermediate 52 Ethyl 1 -[4-(3-chloro-2H-indazol-2-yl)phenyl]-3-[[(^ans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), 2-(4-bromophenyl)-3-chloro-2H-indazole (104 mg), copper (I) iodide (14 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 ml_) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This was purified by MDAP HPLC to give the title compound. MS calcd for (C30H34CIN5O3+ H)+: 548/550
MS found (electrospray): (M+H)+ = 548/550
Intermediate 53
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 -methyl-1 H- indol-2-yl)phenyl]-1 H-pyrazole-4-carboxylate
Intermediate 5 (200 mg), (1 -methyl-1 H-indol-2-yl)boronic acid (110 mg) cesium fluoride (191 mg) palladium (II) acetate (18.8 mg) and 2-dicyclohexylphosphino-2'-(N,N-dimethylamino)- biphenyl (50 mg) were dissolved in dioxane (4 mL) and stirred at room temperature for 24 h. The reaction was then heated at 100°C for 2 h. After cooling to room temperature the mixture was diluted with water and extracted with ethyl acetate. The organic extract was washed with sodium bicarbonate solution brine, dried (Na2SO4) and concentrated. This residue was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C32H38N4O3 + H)+: 527 MS found (electrospray): (M+H)+ = 527
Intermediate 54
1,1 -Dimethylethyl 2-(4-{4-[(ethyloxy)carbonyl]-3-[[(fraπs-4-methylcyclohexyl) carbonyl](1-methylethyl)amino]-1H-pyrazol-1-yl}phenyl)-5-fluoro-1H-indole-1- carboxylate
Intermediate 5 (200 mg), (1-{[(1 ,1-dimethylethyl)oxy]carbonyl}-5-fluoro-1H-indol-2-yl)boronic acid (176 mg), cesium fluoride (191 mg), palladium (II) acetate (18.8 mg) and 2- dicyclohexylphosphino-2'-(N,N-dimethylamino)-biphenyl (50 mg) were dissolved in dioxane (4 mL) and stirred at room temperature for 2 h. The mixture was diluted with water and extracted with ethyl acetate. The organic extract was washed with sodium bicarbonate solution brine, dried (Na2SO4) and concentrated. This residue was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C36H43FN4O5 + H)+: 631 MS found (electrospray): (M+H)+ = 631
Intermediate 55
Ethyl 1 -[4-(1 ,3-benzothiazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl) carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1W-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 11 and Intermediate 23 to give the title compound. MS calcd for (C32H36N4O4S+ H)+: 573 MS found (electrospray): (M+H)+ = 573
Intermediate 56
4-Bromo-Λ/-(2-chloro-3-pyridinyl)benzamide
To a solution of 3-amino-2-chloropyridine (2.57 g) in dry pyridine (20 ml.) was added dropwise at -1O0C 4-bromobenzoyl chloride (4.38 g.) in dry dichloromethane (5 ml_). The reaction mixture was stirred for 2 h and then water (200 mL) added. The solid was filtered, washed with water and dried overnight to give the title compound. MS calcd for (C12H8BrCIN2O + H)+: 311/313 MS found (electrospray): (M+H)+ = 311/313
Intermediate 57
2-(4-Bromophenyl)[1,3]thiazolo[5,4-/)]pyridine
N ^
Intermediate 56 (1 g) was dissolved in DMPU (5 mL). To this was added Lawesson's reagent (1.3 g). The reaction was heated for 12 h at 10O0C. The reaction mixture was partitioned between ethyl acetate and water. The ethyl acetate extract was washed with water three times and then dried over sodium sulphate, filtered and evaporated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C12H7BrN2S + H)+: 291/293 MS found (electrospray): (M+H)+ = 291/293
Intermediate 58
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-
Prepared by a similar method to that described for Intermediate 19 using Intermediate 4 and Intermediate 57 to give the title compound. MS calcd for (C18H23N3O3 + H)+: 532 MS found (electrospray): (M+H)+ = 532
Intermediate 59
Ethyl 1-[4-(6-chloro-7aH-imidazo[4,5-b]pyridin-2-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (95 mg), 2-(4-bromophenyl)-6-chloro-7a/-/-imidazo[4,5-b]pyridine (100 mg), copper (I) iodide (6 mg), trans-C\ R, 2R)-N, N'-bismethyl-1 ,2-cyclohexane diamine (17 mg) and potassium carbonate (82 mg) in DMF (1.5 mL) was heated at 16O0C in a Reactivial for 8 h under a nitrogen atmosphere. The DMF was evaporated under vacuum and the residue partitioned between DCM and a water/methanol/2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by chromatography over silica (SPE), eluting with cyclohexane/EtOAc (2:1 ) to give the title compound. MS calcd for (C29H33CIN6O3 + H)+: 549/551 MS found (electrospray): (M+H)+ = 549/551
Intermediate 60
Ethyl 1 -[4-(7-amino-5-methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (80 mg), 2-(4-bromophenyl)-5-methylpyrazolo[1 ,5-a]pyrimidin-7- amine (83 mg), copper (I) iodide (5 mg), f/-ans-(1 R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (14 mg) and potassium carbonate (69 mg) in DMF (1.5 mL) was heated at 16O0C in a Reactivial for 8 h under a nitrogen atmosphere. The DMF was evaporated under vacuum
and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by chromatography over silica (ISCO Companion), eluting with cyclohexane/EtOAc (1 :1 ) to give the title compound. MS calcd for (C30H37N7O3 + H)+: 544 MS found (electrospray): (M+H)+ = 544
Intermediate 61
Ethyl 1 -[4-(5-amino-1 ,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (148 mg), 2-(4-bromophenyl)-1 ,3-benzoxazol-5-arnine (96 mg), copper (I) iodide (6 mg), trans-tf R, 2R)-N, N'-bismethyl-1 ,2-cyclohexane diamine (19 mg) and potassium carbonate (92 mg) in DMF (1.5 ml.) was heated at 16O0C in a Reactivial for 7 h under a nitrogen atmosphere. The DMF was evaporated under vacuum and the residue partitioned between DCM and brine. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by chromatography over silica (Biotage), eluting with cyclohexane/EtOAc (3:1) to give the title compound.
MS calcd for (C30H35N5O4 + H)+: 530
MS found (electrospray): (M+H)+ = 530
Intermediate 62
Ethyl i-t^furo^^-fclpyridin^-ylphenyO-S^Kfrans^-methylcyclohexyOcarbonyllKSR)- tetrahydro-3-furanyl]amino}-1H-pyrazole-4-carboxylate
A mixture of Intermediate 170 (200 mg), Intermediate 39 (131 mg), copper (I) iodide (9 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (135 mg) and potassium carbonate (132 mg) in DMF (3 mL) was heated at 16O0C in a Reactivial for 4 h under a nitrogen atmosphere. The DMF was evaporated under vacuum and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by chromatography over silica (ISCO Companion), eluting with EtOAc/cyclohexane (gradient from 0% to 100%) to give the title compound. MS calcd for (C31H34N4O5 + H)+: 543 MS found (electrospray): (M+H)+ = 543
Intermediate 63
Ethyl 1-(4-furo[3,2-b]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][(3S)- tetrahydro-3-furanyl]amino}-1/Y-pyrazole-4-carboxylate
A mixture of Intermediate 172 (200 mg), Intermediate 39 (131 mg), copper (I) iodide (9 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (135 mg) and potassium carbonate (132 mg) in DMF (3 mL) was heated at 16O0C in a Reactivial for 8 h under a nitrogen atmosphere. The DMF was evaporated under vacuum and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by chromatography over silica (ISCO Companion), eluting with EtOAc/cyclohexane (gradient from 0% to 100%) to give the title compound. MS calcd for (C3IH34N4O5 + H)+: 543 MS found (electrospray): (M+H)+ = 543
Intermediate 64
Ethyl 1-[4-(4-amino-3-cyanopyrazolo[5,1-c][1,2,4]triazin-7-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (115 mg), 4-amino-7-(4-bromophenyl)pyrazolo[5,1- c][1,2,4]triazine-3-carbonitrile (95 mg), copper (I) iodide (6 mg), ^a/7s-(1 R,2R)-N,N'- bismethyl-1 ,2-cyclohexane diamine (85 mg) and potassium carbonate (83 mg) in DMF (1.5 mL) was heated at 16O0C in a Reactivial for 3 h under a nitrogen atmosphere. The DMF was evaporated under vacuum and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by chromatography over silica (SPE), eluting with EtOAc/cyclohexane (gradient from 25% to
100%) then methanol, to give the title compound.
MS calcd for (C29H33N9O3 + H)+: 556
MS found (electrospray): (M+H)+ = 556
Intermediate 65
4-Bromophenylcarboxylic acid (0.7 g) in phosphorous oxychloride (5 mL) was treated with 2- amino-3-pyridinyl bis(1-methylethyl)dithiocarbamate (0.81 g). The reaction mixture heated at 700C for 2 h and then cooled and basified at O0C. The aqueous layer was then extracted with ethyl acetate. The organics then separated, and washed with water, dried over sodium sulphate, filtered and evaporated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C12H7BrN2S + H)+: 291/293 MS found (electrospray): (M+H)+ = 291/293
Intermediate 66
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-
[1 ,3]thiazolo[4,5-b]pyridin-2-ylphenyl)-1f/-pyrazole-4-carboxylate
Prepared by a similar method to that described for Intermediate 19 using Intermediate 4 and
Intermediate 65 to give the title compound.
MS calcd for (C29H33N5O3S + H)+: 532
MS found (electrospray): (M+H)+ = 532
Intermediate 67
6-(4-Bromophenyl)furo[3,2-cr]pyrimidine
To 4-bromophenylactetylene (0.3 g) was added 4-iodo-5-pyrimidinol (0.4 g), bis(triphenylphosphine)palladium dichloride (120 mg), copper (I) iodide (120 mg) and triethylamine (20 mL). The reaction was stirred at 650C, under nitrogen, for 3 h. The reaction was cooled, diluted with EtOAc (100 mL) and washed with saturated ammonium chloride solution, brine and then concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C13H8BrNO + H)+: 276/278 MS found (electrospray): (M+H)+ = 276/278
Intermediate 68
Ethyl 1-(4-furo[3,2-d]pyrimidin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (50 mg), Intermediate 67 (40 mg), copper (I) iodide (7 mg), trans- (1R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (10 mg) and potassium carbonate (50 mg) in DMF (2.0 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The crude material was purified by SPE silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C29H32N4O4+ H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 69 6-(4-Bromophenyl)furo[2,3-d]pyrimidine
To 4-bromophenylactetylene (0.9 g) was added 4-iodo-5-pyrimidinol (1.2 g), bis(triphenylphosphine)palladium dichloride (120 mg), copper (I) iodide (0.12 g) and triethylamine (25 mL). The reaction was stirred at 9O0C, under nitrogen, for 4 h. The reaction was cooled, diluted with EtOAc (100 mL) and washed saturated ammonium chloride solution, brine, dried over sodium sulphate, filtered and then concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C13H8BrNO + H)+: 275/277 MS found (electrospray): (M+H)+ = 275/277
Intermediate 70
Ethyl 1-(4-furo[2,3-d]pyrimidin-6-ylphenyl)-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 69 (90 mg), copper (I) iodide (14 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (3.0 ml_) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The crude material was purified by SPE silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C29H32N4O4+ H)+: 516 MS found (electrospray): (M+H)+ = 516
Intermediate 71
Ethyl 1 -(4-imidazo[2,1 -b][1 ,3]thiazol-6-ylphenyl)-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (200 mg), 6-(4-iodophenyl)imidazo[2,1-/b][1 ,3]thiazole (235 mg), copper (I) iodide (28 mg), fraπs-N,N'-bismethyl-1 ,2-cyclohexane diamine (42 mg) and potassium carbonate (207 mg) in DMF (4 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C28H33N5O3S + H)+: 520 MS found (electrospray): (M+H)+ = 520
Intermediate 72
Ethyl 1 -[4-(5,7-dimethylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (200 mg), 2-(4-bromophenyl)-5,7-dimethylpyrazolo[1 ,5- a]pyrimidine (217 mg), copper (I) iodide (28 mg), trans-N, N'-bismethyl-1 ,2-cyclohexane diamine (42 mg) and potassium carbonate (207 mg) in DMF (4 mL) was heated at 16O0C in a
Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between
DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C3IH38N6O3 + H)+: 543 MS found (electrospray): (M+H)+ = 543
Intermediate 73
Ethyl 1 -(4-imidazo[1 ,2-a]pyrimidin-2-ylphenyl)-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (200 mg), 2-(4-iodophenyl)imidazo[1 ,2-a]pyrimidine (231 mg), copper (I) iodide (28 mg), fraπs-N,N'-bismethyl-1 ,2-cyclohexane diamine (42 mg) and potassium carbonate (207 mg) in DMF (4 mL) was heated at 16O0C in a Reactivial for 18 h.
The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This was purified by silica gel chromatography eluting with a gradient of ethyl acetatexyclohexane (5:95 to 100:0) followed by ethyl acetate: methanol (100:0 to 50:50) to give the title compound.
MS calcd for (C29H34N6O3 + H)+: 515
MS found (electrospray): (M+H)+ = 515
Intermediate 74
2-(4-Bromophenyl)furo[3,2-c]pyridine
3-lodo-4-hydroxypyridine (122 mg), 4-bromophenylacetylene (1 10 mg), copper iodide (10 mg) and bis(triphenylphosphino)palladium chloride (20 mg) were dissolved in DCM (10 mL) and triethylamine (1 mL). The reaction mixture was stirred at room temperature under nitrogen for 3 days and then evaporated to dryness. The residue was taken into DCM (50 mL), washed with saturated sodium chloride solution (50 mL), dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 10 g silica SPE, eluted with a gradient of cyclohexane in DCM, followed by a gradient of DCM in ethyl acetate. The product containing fractions were concentrated to give the title compound. MS calcd for (C13H8BrNO + H)+: 274 / 276 MS found (electrospray): (M+H)+ = 274 / 276
Intermediate 75
Ethyl 3-{(1 -methylethyl)[(4-methylphenyl)carbonyl]amino}-1 -(phenylmethyl)-i H- pyrazole-4-carboxylate
Intermediate 2 (2.9 g) was dissolved in dry DCM (20 mL) and treated with triphenylphosphine (5.3 g) and p-toluolyl chloride (2.67 mL) in dry DCM (20 mL). The mixture was heated at reflux temperature for 18 hours. The reaction was basified with saturated sodium bicarbonate solution. The mixture was extracted with DCM and the organic extract dried (MgSO4) and concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of DCM in cyclohexane followed by ethyl acetate in cyclohexane to give the title compound. MS calcd for (C24H27N3O3 + H)+: 406 MS found (electrospray): (M+H)+ = 406
Intermediate 76 Ethyl 3-{(1 -methylethyl)[(4-methylphenyl)carbonyl]amino}-1 H-pyrazole-4-carboxylate
Intermediate 75 (3.61 g) was dissolved in ethanol (120 mL) and concentrated HCI (2.5 mL) and treated with a catalyst of 10% Pd on carbon (1.2 g) under an atmosphere of hydrogen.
The reaction was stirred at room temperature for 18 hours. The catalyst was removed by filtration through Celite under a stream of nitrogen and the filtrate concentrated. The residue was dissolved in DCM and basified with saturated sodium bicarbonate solution. The mixture was extracted with DCM and the organic extract washed with brine, dried (MgSO4) and concentrated to give the title compound.
MS calcd for (C17H2iN3O3 + H)+: 316 MS found (electrospray): (M+H)+ = 316
Intermediate 77
Ethyl 1 -(4-furo[3,2-b]pyridin-2-ylphenyl)-3-{(1 -methylethyl)[(4- methylphenyl)carbonyl]amino}-1H-pyrazole-4-carboxylate
Intermediate 76 (115 mg), Intermediate 39 (100 mg), frans-(1R,2R)N,N'-bismethyl1,2- cyclohexanediamine (21 mg) and potassium carbonate (100 mg) were dissolved in dry DMF (4 ml_) and treated with copper iodide (10 mg). The reaction was heated at 1100C in a Reactivial for 18 hours. The reaction mixture was treated with frans-(1 R,2R)N,N'-bismethyl- 1 ,2-cyclohexanediamine (21 mg) and copper iodide (10 mg) and was heated at 155°C in the Reactivial for 18 hours. The DMF was removed in vacuo and the residue partitioned between water and ethyl acetate. The mixture was extracted with ethyl acetate and the organic extract washed with brine, dried (MgSO4) and concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H28N4O4 + H)+: 509 MS found (electrospray): (M+H)+ = 509
Intermediate 78
Ethyl 3-[(cyclohexylcarbonyl)(1-methylethyl)amino]-1-(phenylmethyl)-1W-pyrazole-4- carboxylate
Intermediate 2 (2.8 g) was dissolved in dry DCM (20 mL) and treated with triphenylphosphine (5.12 g) and cyclohexanecarbonylchloride (2.61 mL) in dry DCM (20 mL). The reaction was heated at reflux temperature for 18 hours. The reaction mixture was treated with further cyclohexanecarbonylchloride (0.65 mL). The reaction was heated at reflux temperature for 18 hours and then was basified with saturated sodium bicarbonate solution. The mixture was extracted with DCM and the organic extract dried (MgSO4) and concentrated. The residue was purified by 5Og silica SPE cartridge eluting with a gradient of DCM in cyclohexane, then a gradient of ethyl acetate in DCM. The crude product was further purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C23H31N3O3 + H)+: 398 MS found (electrospray): (M+H)+ = 398
Intermediate 79
Ethyl 3-[(cyclohexylcarbonyl)(1 -methylethyl)amino]-1 H-pyrazole-4-carboxylate
Intermediate 78 (3.19 g) was dissolved in a mixture of ethanol (110 ml.) and concentrated HCI (2.5 ml.) and treated with a catalyst of 10% Pd on carbon (1.2 g) under an atmosphere of hydrogen. The reaction was stirred at room temperature for 18 hours. The catalyst was removed by filtration through Celite under a stream of nitrogen and the filtrate concentrated. The residue was dissolved in DCM and basified with saturated sodium bicarbonate solution. The mixture was extracted with DCM and the organic extract washed with brine, dried (MgSO4) and concentrated to give the title compound. MS calcd for (C16H25N3O3 + H)+: 308 MS found (electrospray): (M+H)+ = 308
Intermediate 80 Ethyl 3-[(cyclohexylcarbonyl)(1-methylethyl)amino]-1-(4-furo[3,2-b]pyridin-2-ylphenyl)- 1 H-pyrazole-4-carboxylate
Intermediate 79 (112 mg), Intermediate 39 (100 mg), frans-(1R,2R)N,N'-bismethyl1 ,2- cyclohexanediamine (21 mg) and potassium carbonate (100 mg) were dissolved in dry DMF (4 ml.) and treated with copper iodide (10 mg). The reaction was heated at 1100C in a Reactivial for 18 hours. The DMF was removed in vacuo and the residue partitioned between water and ethyl acetate. The mixture was extracted with ethyl acetate and the organic extract dried (MgSO4) and concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C29H32N4O4 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Intermediate 81 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(phenylmethyl)-i H- pyrazole-4-carboxylic acid
Intermediate 3 (2.95 g) was dissolved in THF (25 mL) and ethanol (25 mL) and treated with LiOH (2N, 28 mL). The reaction was stirred at room temperature for 18 hours. The reaction mixture was treated with more LiOH (2M, 6 mL) and the reaction stirred at room temperature for 18 hours. The ethanol and THF were removed in vacuo and the residue acidified with 2N HCI solution. The mixture was extracted with ethyl acetate and the organic extract washed with brine, dried (MgSO4) and concentrated to give the title compound. MS calcd for (C22H29N3O3+ H)+: 384 MS found (electrospray): (M+H)+ = 384
Intermediate 82
S-ttttrans^-MethylcyclohexyOcarbonylKI-methylethyOaminoJ-IH-pyrazole^- carboxylic acid
Intermediate 81 (2.75 g) was dissolved in ethanol (120 mL) and concentrated HCI (2.5 mL) and treated with a catalyst of 10% Pd on carbon (0.78 g) under a hydrogen atmosphere. The reaction was stirred at room temperature for 18 hours. The catalyst was removed by filtration through Celite under a stream of nitrogen and the filtrate concentrated to give the title compound. MS calcd for (C15H23N3O3 + H)+: 294 MS found (electrospray): (M+H)+ = 294
Intermediate 83 2-(4-Bromophenyl)[1,3]oxazolo[4,5-b]pyridine
2-Amino-3-hydroxypyridine (1 g), 4-bromobenzoic acid (3.65 g) and polyphosphoric acid (2.5 g) were heated at 185°C for 20 minutes. After cooling slightly, ice water was added to the reaction which resulted in a precipitate which was collected by filtration. The filtrate was neutralised with saturated bicarbonate solution. The mixture was extracted with
dichloromethane and the organic extract dried (MgSO4) and concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C12H7BrN2O + H)+: 275/277 MS found (electrospray): (M+H)+ = 275/277
Intermediate 84
Ethyl 3-{ethyl[(trans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylate
To Intermediate 17 (64 mg) was added Intermediate 42 (62 mg), copper (I) iodide (9.5 mg), rac-1 ,2-dimethylcyclohexane diamine (141 mg) and potassium carbonate (68.8 mg) and DMF (2.5ml_). The reaction was heated at 16O0C in a Reactivial for 18 hours. The DMF was removed and the residue partitioned between DCM and water. The organic phase was then concentrated and purified by a 12g silica ISCO Companion flash column eluting with ethyl acetate in cyclohexane (10-100%) to give the title compound. MS calcd for (C28H32N6O3 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Intermediate 85
Ethyl S-tKtrans^-methylcyclohexyOcarbonylKtetrahydro-a-furanyOaminol-i^- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylate
To Intermediate 96 (200 mg) was added Intermediate 42 (172 mg) copper (I) iodide (26.2 mg), rac-1 ,2-dimethylcyclohexane diamine (39.2 mg) and potassium carbonate (190 mg) and
DMF (5 ml_). The reaction was heated at 16O0C in a Reactivial overnight. The DMF was removed, the residue partitioned between DCM and water, and then passed through a hydrophobic frit. The organic phase was then concentrated and purified by a 4Og silica ISCO
Companion flash column eluting with ethyl acetate in cyclohexane (10-100%) to give the title compound.
MS calcd for (C30H34N6O4 + H)+: 543 MS found (electrospray): (M+H)+ = 543
Intermediate 86 2-(5-Bromo-2-thienyl)-1,3-benzoxazole
To 5-bromo-2-thiophene carboxylic acid (1 g) was added chloroform (5 mL) and thionyl chloride (2 mL). The reaction was heated at 660C, under nitrogen, for 2 hours. The solvent was carefully evaporated and a portion of the resultant acid chloride (454 mg) was suspended with 2-aminophenol (200 mg) in 1,4-dioxane (2.5 mL). The mixture was heated in a microwave at 21O0C for 15 mins. To the cooled reaction was added 1N sodium hydroxide (40 mL) and filtered. The precipitate was then purified by 4Og silica ISCO companion flash column eluting with EtOAc in cyclohexane (gradient from 0-50%) to give the title compound.
MS calcd for (C11H6BrNOS + H)+: 281/283 MS found (electrospray): (M+H)+ = 281/283
Intermediate 87
Ethyl 1 -[5-(1 ^-benzoxazol^-yl^-thienyll-S-Kftrans-A-methylcyclohexyOcarbonylHI - methylethyl)amino]-1H-pyrazole-4-carboxylate
To Intermediate 4 (200 mg) was added Intermediate 86 (180 mg), copper (I) iodide (28 mg), rac-1 ,2-dimethylcyclohexane diamine (42 mg), potassium carbonate (200 mg) and DMF (5 mL). The reaction was heated at 16O0C in a Reactivial for overnight. The DMF was removed and the residue partitioned between DCM and water. The organic phase was then concentrated and purified by a 4Og silica ISCO Companion flash column eluting with ethyl acetate in cyclohexane (gradient from 0-50%) to give the title compound. MS calcd for (C28H32N4O5S + H)+: 521 MS found (electrospray): (M+H)+ = 521
Intermediate 88 2-(4-Bromo-2-chlorophenyl)-1 ,3-benzoxazole
2-Aminophenol (195.1 mg) and 4-bromo-2-chloro benzoyl chloride (500 mg) were suspended in 1 ,4-dioxane (2.5 mL) and the mixture heated in a microwave at 21O0C for 15 mins. To the
cooled reaction was added 1 N sodium hydroxide (40 mL) and the precipitate was filtered and washed with water. The precipitate was then dried on a rotary evaporator to give the title compound.
MS calcd for (Ci3H7BrCINO + H)+: 308/309/311 MS found (electrospray): (M+H)+ = 308/309/311
Intermediate 89
Ethyl 1 -[4-(1 ,3-benzoxazol-2-yl)-3-chlorophenyl]-3-[[(trans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
To intermediate 4 (200 mg) was added Intermediate 88 (211 mg), copper (I) iodide (28 mg), rac-1 ,2-dimethylcyclohexane diamine (42 mg), potassium carbonate (206 mg) and DMF (5 mL). The reaction was heated at 16O0C in a Reactivial for overnight. The DMF was removed and the residue partitioned between DCM and water. The organic phase was then concentrated and purified by a 4Og silica ISCO Companion flash column eluting with ethyl acetate in cyclohexane (gradient from 0-50%) to give the title compound. MS calcd for (C30H33CIN4O4 + H)+: 549/551 MS found (electrospray): (M+H)+ = 549/551
Intermediate 90
Ethyl 1 -[4-(5-amino-1 -benzofuran-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 286 (165 mg), Intermediate 4 (184 mg), potassium carbonate (158 mg), R,R'-bis(methylamino)cyclohexane (35 mg) and copper (I) iodide (11.6 mg) in DMF (2.3 mL) was stirred and irradiated in a CEM Explorer microwave reactor at 1600C for 1.5 h. A further quantity of copper (I) iodide (10 mg) was added and microwave heating was continued for 2.5 h. The resulting mixture was evaporated to dryness under reduced pressure and the residue was partitioned between 100 mL portions of water and ethyl acetate. The organic layer was collected, washed with brine and dried (MgSO4). Solvent was removed under reduced pressure and the residue was purified by ISCO Companion
silica chromatography eluting with ethyl acetate/cyclohexane (a gradient of 0-100%) to give the title compound.
MS calcd for (C31H36N4O4 + H)+: 529
MS found (electrospray): (M+H)+ = 529
Intermediate 91
Ethyl 1-(4-furo[3,2-b]pyridin-2-ylphenyl)-3-[[(trans-4-methylcyclohexyl) carbonyl](tetrahydro-3-furanyl)amino]-1H-pyrazole-4-carboxylate
To Intermediate 95 (200 mg) was added Intermediate 39 (173 mg), copper (I) iodide (26.2 mg), rac-1,2-dimethylcyclohexane diamine (39.2 mg), potassium carbonate (190 mg) and DMF (5 ml_). The reaction was heated at 16O0C in a Reactivial for overnight. The DMF was removed and the residue partitioned between DCM and water and passed through a hydrophobic frit. The organic phase was then concentrated and purified by a 4Og silica ISCO Companion flash column eluting with ethyl acetate in cyclohexane (gradient from 10-100%) to give the title compound. MS calcd for (C31H34N4O5 + H)+: 543 MS found (electrospray): (M+H)+ = 543
Intermediate 92
Ethyl 3-[[(trans-4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylate
To Intermediate 11 (150 mg) was added Intermediate 42 (124 mg), copper (I) iodide (18.8 mg), rac-1 ,2-dimethylcyclohexane diamine (28 mg), potassium carbonate (138 mg) and DMF (3.75 ml_). The reaction was heated at 16O0C in a Reactivial for overnight. The DMF was removed and the residue partitioned between DCM and water. The organic phase was then concentrated and purified by a 40g silica ISCO Companion flash column eluting with ethyl acetate in cyclohexane (gradient from 10-100%) to give the title compound. MS calcd for (C31 H36N6O4 + H)+: 557 MS found (electrospray): (M+H)+ = 557
Intermediate 93
Ethyl 1 -(4-imidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(trans-4-methylcyclohexyl) carbonyl](tetrahydro-3-furanyl)amino]-1H-pyrazole-4-carboxylate
To Intermediate 96 (200 mg) was added 2-(4-bromophenyl)imidazo[1 ,2-a]pyridine5 (172 mg), copper (I) iodide (9.5 mg), rac-1 ,2-dimethylcyclohexane diamine (141 mg), potassium carbonate (68.8 mg) and DMF (2.5 ml_). The reaction was heated at 16O0C in a Reactivial for 18 hours. The DMF was removed and the residue partitioned between DCM and water.
The organic phase was then concentrated and purified by a 4Og silica ISCO Companion flash column eluting with ethyl acetate in cyclohexane (gradient from 10-100%) to give the title compound.
MS calcd for (C28H32N6O3 + H)+: 501
MS found (electrospray): (M+H)+ = 501
Ref 3. Tetrahedron Lett. 42, (17) 2001 , 3077-3080
Intermediate 94
Tetrahydro-3-furanyl trifluoromethanesulfonate
Tetrahydro-3-furanol (14.71 g) was dissolved in anhydrous DCM (400 mL) and was treated with pyridine (16.95 mL). The mixture was cooled to -10°C under an atmosphere of nitrogen.
Trifluoromethane sulfonic anhydride (51.6 g) was added dropwise ensuring the internal temperature of the reaction did not rise above 0°C. The reaction was then stirred at -10°C for
55 minutes. The reaction mixture was quenched with aqueous 2M HCI (200 mL) and the organics were separated, dried (Na2SO4) and filtered. This solution was then filtered in portions through pre-packed Celite cartridges (2 X 10 g) to give title compound as a DCM solution.
TLC (Silica gel 60 F254) eluted with EtOAc, plate developed with potassium permanganate solution.
Tetrahydro-3-furanol Rf = 0.45. Title compound Rf = 0.88.
Intermediate 95
Ethyl S-lKfraπs-A-methylcyclohexyOcarbonylHtetrahydro-S-furanylJaminoH-
Intermediate 15 (20.55 g) was dissolved in anhydrous THF (400 ml_) and anhydrous DMF (50 mL) and was cooled to -78°C under a nitrogen atmosphere. Potassium bis(trimethylsilyl)amide (0.5M solution in toluene, 150 mL) was added dropwise via a dropping funnel over 45 minutes. The reaction mixture was then stirred at -78°C for 1.25 h. Intermediate 94 (167 mL) was then added dropwise over 1 h. The reaction was then allowed to warm to -10°C over 2 h. Intermediate 94 (40 mL) was added dropwise and the reaction was stirred at -10°C for 1 h. The reaction was quenched with aqueous 2M HCI (200 mL) and was extracted with EtOAc (700 mL). The organics were washed with brine, dried (Na2SO4), filtered and evaporated in vacuo. The crude product was purified by flash chromatography over silica (Biotage, 400 g silica cartridge) eluting with cyclohexane/EtOAc (3:1). Product containing fractions were combined and evaporated in vacuo. The residue was re-purified by flash chromatography over silica (Biotage, 200 g silica cartridge) eluting with cyclohexane/EtOAc (3:1) to give the title compound. MS calcd for (C25H33N3O4+ H)+: 440 MS found (electrospray): (M+H)+ = 440
Intermediate 96
Ethyl-S-^fra/is^-methylcyclohexyOcarbonylJttetrahydro-S-furanyOaminoJ-IH- pyrazole-4-carboxylate
A solution of Intermediate 95 (5.86 g) in ethanol (250 mL) and concentrated hydrochloric acid (3 mL) was subjected to atmospheric hydrogenation using 10% palladium on carbon catalyst (1.5 g) overnight. The mixture was filtered through a Celite pad and the filtrate was neutralised with saturated aqueous sodium carbonate solution (100 mL). The mixture was filtered and evaporated in vacuo to near dryness. The residue was partitioned between EtOAc / water and the organics were separated, dried (Na2SO4), filtered and evaporated in vacuo to give the title compound. MS calcd for (C18H27N3O4+ H)+: 350
MS found (electrospray): (M+H)+ = 350
Intermediate 97 2-(4-Bromophenyl)-5-methylpy
A solution of 5-amino-3-(4-bromophenyl)pyrazole (237 mg) and 4,4-dimethoxy-2-butanone (133 μl_) in ethanol (5 mL) was heated at reflux temperature for 16 hours. White crystals were removed from the mixture, washed with methanol, filtered and dried under vacuum to give the title compound. MS calcd for (C13H10BrN3 + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Intermediate 98 2-(4-Bromophenyl)-6-methyl
A solution of 5-amino-3-(4-bromophenyl)pyrazole (300 mg) and 1 ,1-3,3-tetraethoxy-2- methylpropane (300 mg) in acetic acid (5 mL) was heated at 11O0C for 3 hours. On cooling to room temperature an off white precipitate was collected by filtration, washed with cold acetic acid and dried under vacuum to give the title compound. MS calcd for (C13H10BrN3 + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Intermediate 99 2-(4-Bromophenyl)pyrazolo[1,5-a]pyrimidin-7 -amine
A solution of 5-amino-3-(4-bromophenyl)pyrazole (300 mg) and 3-
(dimethylamino)acrylonitrile (123 mg) in acetic acid (5 mL) was heated at 110°C for 3 hours. Solvent was evaporated under vacuum and the residue was dissolved in 2N sodium bicarbonate solution. The resultant white precipitate was collected by filtration, washed with water and then cold isopropanol and dried under vacuum to give the title compound. MS calcd for (C12H9BrN4 + H)+: 289/291 MS found (electrospray): (M+H)+ = 289/291
Intermediate 100 Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(5- methylpyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 97 (98 mg), copper iodide (6 mg), trans- N,N'-dimethyl-1 ,2-diaminocyclohexane (18 mg) and potassium carbonate (86 mg) was stirred in dry DMF (1.5 mL) under a nitrogen atmosphere at 16O0C. After 4 hours, the solvent was evaporated then the residue was partitioned between water and DCM. The organic phase was evaporated and the residue was purified by chromatography over silica (ISCO Companion), eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C30H36N6O3 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 101 2-(4-Bromophenyl)-7-methylpy
A solution of 5-amino-3-(4-bromophenyl)pyrazole (237 mg) and 4,4-dimethoxy-2-butanone (133 μl_) in ethanol (5 mL) was heated at reflux temperature for 16 hours. White crystals were removed from the mixture and the remaining solution was evaporated under vacuum.
Purification of the residue by chromatography over silica using an eluant of cyclohexane/EtOAc (5:1 ) gave the title compound.
MS calcd for (C13H10BrN3 + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Intermediate 102
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(7- methylpyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-1W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (41 mg), Intermediate 101 (37 mg), copper iodide (3 mg), trans-
N,N'-dimethyl-1 ,2-diaminocyclohexane (7.5 mg) and potassium carbonate (35 mg) was stirred in dry DMF (0.6 mL) under a nitrogen atmosphere at 16O0C. After 4 hours, the solvent was evaporated and the residue was partitioned between water and DCM. The organic
phase was evaporated and the residue was purified by chromatography over silica gel, eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C30H36N6O3 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 103 2-(4-Bromophenyl)-5-methylfu
(4-Bromophenyl)ethyne (781 mg), 6-iodo-2-picolin-5-ol (1.01 g) and copper iodide (109 mg) were stirred in triethylamine (17 ml_) under an atmosphere of nitrogen. Pd(PPh3)2CI2 (276 mg) was added and the mixture was heated at 9O0C. After 10 minutes a thick precipitate formed and the reaction mixture was diluted with more triethylamine (10 ml_). Vigorous stirring was maintained for 5 hours and the mixture was cooled to room temperature and partitioned between EtOAc and brine. The organic phase was separated, dried (Na2SO4) and evaporated under vacuum. The residue was purified by chromatography over silica
(ISCO Companion) eluting with a gradient of EtOAc in cyclohexane to give the title compound.
MS calcd for (C14H10BrNO + H)+: 288/290
MS found (electrospray): (M+H)+ = 288/290
Intermediate 104
Ethyl 3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(5- methylfuro[3,2-Jb]pyridin-2-yl)phenyl]-1W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 103 (89 mg), copper iodide (6 mg), trans- N,N'-dimethyl-1 ,2-diaminocyclohexane (18 mg) and potassium carbonate (86 mg) was stirred in dry DMF (1.5 ml.) under a nitrogen atmosphere at 16O0C. After 3 hours, inorganic solids were removed by filtration and the filtrate was evaporated. The residue was partitioned between water and DCM, the organic phase was separated and evaporated under vacuum. The residue was purified by chromatography over silica (ISCO Companion), eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C31H36N4O4 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 105
2-(4-Bromophenyl)-6-methylfuro[3,2-b]pyridine
A mixture of (4-bromophenyl)ethyne (272 mg), 2-iodo-5-methyl-3-pyridinol (360 mg), copper iodide (38 mg) and Pd(PPh3)2CI2 (96 mg) was stirred in triethylamine (10 ml.) under an atmosphere of nitrogen at 9O0C. After 4 hours, solvent was removed under vacuum, and the residue was partitioned between water and DCM. The organic phase was separated, evaporated under vacuum and purified by chromatography over silica eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C14H10BrNO + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Intermediate 106
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(6- methylfuro[3,2-b]pyridin-2-yl)phenyl]-1AV-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 105 (89 mg), copper iodide (6 mg), trans- N,N'-dimethyl-1 ,2-diaminocyclohexane (18 mg) and potassium carbonate (86 mg) was stirred in dry DMF (1.5 mL) under a nitrogen atmosphere at 16O0C for 3 hours. The mixture was cooled to room temperature, solids were removed by filtration and the filtrate solution was evaporated under vacuum. The residue was purified by chromatography over silica gel, eluting with a gradient of EtOAc in cyclohexane. This was dissolved in Et2O and re- evaporated under vacuum to give the title compound. MS calcd for (C3IH36N4O4 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 107
2-(5-Bromo-2-pyridinyl)-1,3-benzoxazole
A mixture of 2-aminophenol (149 mg) and 5-bromo-2-pyridinecarbonyl chloride (300 mg) in 1 ,4-dioxane (2 mL) was heated in a microwave reactor for 1.5 minutes at 1750C and then 10 minutes at 2000C. The reaction mixture was poured into 2M sodium hydroxide solution (3 mL) and stirred for several minutes. A solid was collected by filtration, washed with water and dried under vacuum to give the title compound. MS calcd for (C12H7BrN2O + H)+: 275/277
MS found (electrospray): (M+H)+ = 275/277
Intermediate 108
Ethyl 1 -[6-(1 ,3-benzoxazol-2-yl)-3-pyridinyl]-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 • methylethyl)amino]-1 W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 107 (117 mg), copper iodide (6 mg), fraA7s-N,N'-dimethyl-1 ,2-diaminocyclohexane (18 mg) and potassium carbonate (86 mg) was stirred in dry DMF (1.5 ml_) under a nitrogen atmosphere at 16O0C. After 2.5 hours, inorganic solids were removed by filtration and the filtrate solution was evaporated. The residue was purified by chromatography over silica gel, eluting with cyclohexane/EtOAc (5:1 ) to give the title compound.
MS calcd for (C29H33N5O4 + H)+: 516
MS found (electrospray): (M+H)+ = 516
Intermediate 109
2-(5-Bromo-2-pyridinyl)furo[3,2-b]pyridine
5-Bromo-2-ethynylpyridine (590 mg), 2-iodo-3-hydroxypyridine (716 mg) and copper iodide (79 mg) were stirred in triethylamine (13 mL) under a nitrogen atmosphere. Pd(PPh3)CI2 (202 mg) was added and the mixture was heated at 9O0C for 1.75 hours. The solvent was evaporated under vacuum, and the residue was partitioned between EtOAc/methanol and water. The organic phase was separated, dried (Na2SO4) and evaporated under vacuum.
The residue was purified by chromatography over silica gel eluting with DCM, cyclohexane/EtOAc (2:1), Et2O then EtOAc. As the combined product fractions were evaporating a solid precipitated from the solution. This was filtered off and dried under vacuum to give the title compound.
MS calcd for (C12H7BrN2O + H)+: 275/277
MS found (electrospray): (M+H)+ = 275/277
Intermediate 110
Ethyl 1 -(6-furo[3,2-fa]pyridin-2-yl-3-pyridinyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 109 (102 mg), copper iodide (6 mg), trans-N, N'-dimethyl-1 ,2-diaminocyclohexane (18 mg) and potassium carbonate (86 mg) was stirred in dry DMF (1.5 ml_) under a nitrogen atmosphere at 16O0C for 2.5 hours. The mixture was cooled, and partitioned between DCM and water. The organic phase was separated, evaporated and the resultant residue was purified by chromatography over silica gel, eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C29H33N5O4 + H)+: 516 MS found (electrospray): (M+H)+ = 516
Intermediate 111 3-Amino-3-(5-bromo-2-pyridinyl)-2-propenenitrile
To a solution of 5-bromo-2-pyridinecarbonitrile (1.83 g) and acetonitrile (1.04 mL) in dry toluene (40 mL) was added potassium f-butoxide (3.37 g). The mixture was stirred under nitrogen at room temperature over one weekend. The mixture was partitioned between Et2O and sodium bicarbonate solution. The organic phase was separated, dried (Na2SO4), filtered and evaporated under vacuum. The residue was purified by chromatography over silica gel eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C8H6N3Br + H)+: 224/226
MS found (electrospray): (M+H)+ = 224/226
Intermediate 112
Intermediate 111 (600 mg) and hydrazine monohydrate (252 mg) were stirred at 9O0C in a solvent of isopropanol for 4 hours. 2M Hydrochloric acid (2 mL) was added then the reaction mixture was cooled to room temperature overnight. A precipitate was collected by filtration and washed with a small amount of isopropanol to give the title compound. MS calcd for (C8H7N4Br + H)+: 239/241
MS found (electrospray): (M+H)+ = 239/241
Intermediate 113
2-(5-Bromo-2-pyridinyl)pyrazolo[1,5-a]pyrimidine
Intermediate 112 (625 mg) and 1 ,1 )3,3-tetra(methyloxy)propane (448 μl_) were heated in a solvent of acetic acid (9 ml_) at 11O0C for 2.5 hours. The mixture was cooled to room temperature and diluted with water (10 ml_). A precipitate was collected by filtration, washed with water and dried under vacuum to give the title compound. MS calcd for (C11H7N4Br + H)+: 275/277 MS found (electrospray): (M+H)+ = 275/277
Intermediate 114
Ethyl 3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(6-pyrazolo[1 ,5- a]pyrimidin-2-yl-3-pyridinyl)-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (200 mg), Intermediate 113 (256 mg), copper iodide (12 mg), frans-N, N'-dimethyl-1 ,2-diaminocyclohexane (35 mg) and potassium carbonate (171 mg) was stirred in dry DMF (2 ml_) under a nitrogen atmosphere at 16O0C for 2.5 hours. Solvent was removed under vacuum and the residue was partitioned between DCM and water. The organic phase was separated, evaporated and the resultant residue was purified by chromatography over silica gel, eluting with a gradient of EtOAc in cyclohexane to give the title compound.
MS calcd for (C28H33N7O3 + H)+: 516 MS found (electrospray): (M+H)+ = 516
Intermediate 115 2-(4-Bromophenyl)-6-fluoropyrazolo[1 ,5-a]pyrimidine
A solution of 5-amino-3-(4-bromophenyl)pyrazole (262 mg) and 3-(diethylamino)-2-fluoro-2- propenal (160 mg) in acetic acid (4 mL) was heated at 11O0C for 3 hours. The mixture was cooled to room temperature and a thick white precipitate was collected by filtration. The solid was washed with water and dried under vacuum to give the title compound. MS calcd for (C12H7BrFN3 + H)+: 292/294 MS found (electrospray): (M+H)+ = 292/294
Intermediate 116
Ethyl 1 -[4-(6-fluoropyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (74 mg), Intermediate 115 (68 mg), copper iodide (4.4 mg), frans-N, N'-dimethyl-i ^-diaminocyclohexane (9.8 mg) and potassium carbonate (48 mg) was stirred in dry DMF (1.5 mL) under a nitrogen atmosphere. The reaction mixture was heated at 11O0C for 1.5 hours, then 13O0C for 1 hour and then at 16O0C for 45 mins. Solvent was removed under vacuum and the residue was partitioned between DCM and water. The organic phase was separated, evaporated and the resultant residue was purified by chromatography over silica gel, eluting with a gradient of EtOAc in cyclohexane to give the title compound.
MS calcd for (C29H33FN6O3 + H)+: 533 MS found (electrospray): (M+H)+ = 533
Intermediate 117
Ethyl 3-[(triphenylmethyl)amino]-1 H-pyrazole-4-carboxylate
To a stirred solution of ethyl 3-amino-1 H-pyrazole-4-carboxylate (20 g) in anhydrous dichloromethane (150 mL), triethylamine (21.58 mL) was added. This was stirred, under nitrogen, in an ice bath, until the temperature of the reaction mixture reached 50C. To this, a solution of trityl chloride (37.76 g) in anhydrous dichloromethane (100 mL) was added dropwise, maintaining the reaction temperature at 50C. The reaction mixture was stirred at 50C, under nitrogen, for 3 h. The reaction mixture was then partitioned between 5% citric acid solution and DCM. The organic phase was extracted, and then dried using a hydrophobic frit. The solvent was then removed in vacuo. This residue was then purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C25H23N3O2H-H)+: 398 MS found (electrospray): (M+H)+ = 398
Intermediate 118
Ethyl 1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-3-[(triphenylmethyl)amino]-1 H- pyrazole-4-carboxylate
Intermediate 117 (500 mg), Intermediate 42 (379 mg), potassium carbonate (418 mg), and frans-N,N-dimethylcyclohexane-1 ,2-diamine (86 mg) were dissolved in anhydrous DMF (12.5 ml_), and then copper (I) iodide (58 mg) was added. The reaction was stirred at 16O0C, under nitrogen, for 16 h. The DMF was removed in vacuo, and the residue partitioned between water and DCM. The layers were separated using a hydrophobic frit, and the organic layer concentrated. This was then purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C37H3ON6CVH)+: 591 MS found (electrospray): (M+H)+ = 591
Intermediate 119
Ethyl 3-{[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 118 (98 mg) in chloroform (8 ml_), trans-Λ- methylcyclohexanecarbonyl chloride2 (53 mg) was added. The reaction was stirred at 650C, under nitrogen, for 16 h. The reaction mixture was then cooled, and the solid filtered, washed with DCM, and dried to give the title compound.
MS calcd for (C26H28N6O3+H)+:473
MS found (electrospray): (M+H)+ = 473 Ref 2: WO 2004/052885
Intermediate 120
Ethyl S-Ittfrans^-methylcyclohexyOcarbonylKtetrahydro^H-pyran^-ylmethyOamino]-
To a stirred suspension of Intermediate 119 (57 mg) in anhydrous DMF (5 ml.) was added sodium hydride (60% dispersion in mineral oil) (11 mg). This was stirred at room temperature, under nitrogen, for 1 h. 4-Bromomethyltetrahydropyran (43 mg) was added, and the reaction was stirred at room temperature, under nitrogen, for 2 h. The reaction was stirred at 6O0C for 1 h. The reaction was then left to stir at 5O0C for 70 h. 2 Drops of water were then added to the reaction mixture, and the DMF was removed in vacuo. The residue was then partitioned between 2M HCI and DCM, and the organic phase removed using a hydrophobic frit, and concentrated to give the title compound. MS calcd for (C32H38N6CVH)+: 571
MS found (electrospray): (M+H)+ = 571
Intermediate 121
Ethyl 3-{[(fraπs-4-methylcyclohexyl)carbonyl][2-(methyloxy)ethyl]amino}-1- (phenylmethyl)-i H-pyrazole-4-carboxylate
Intermediate 15 (500 mg) was dissolved in anhydrous DMF (10 mL). The solution was stirred, under nitrogen, and cooled over ice, before sodium hydride (60% dispersion in mineral oil) (108 mg) was added. This was stirred under nitrogen for 15 minutes, and then 2- bromoethyl methyl ether (1.27 mL) was added, and the reaction stirred, under nitrogen, for 20 h. 2M Hydrochloric acid (1 mL) was then added, and the DMF was removed from the reaction mixture in vacuo, and the residue partitioned between 2M Hydrochloric acid and DCM. The layers were separated using a hydrophobic frit, and the solvent from the organic phase removed in vacuo. This was then purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C24H33N3CVH)+: 428 MS found (electrospray): (M+H)+ = 428
Intermediate 122 Ethyl 3-{[(fra/?s-4-methylcyclohexyl)carbonyl][2-(methyloxy)ethyl]amino}-1 H-pyrazole- 4-carboxylate
A solution of Intermediate 121 (432 mg) in ethanol (17.5 mL) and concentrated hydrochloric acid (0.3 mL) was subjected to atmospheric pressure hydrogenation using 10 wt % palladium on activated carbon catalyst (150 mg, wet) for 16 h. This was then filtered through a pad of Celite (10 g), which was washed with ethanol, and the organics concentrated. The residue was partitioned between saturated sodium bicarbonate solution and DCM, and passed through a hydrophobic frit. The organic phase was concentrated to give the title compound. MS calcd for (C17H27N3CVH)+: 338 MS found (electrospray): (M+H)+ = 338
Intermediate 123
Ethyl 1-(4-furo[3,2-b]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][2-
(methyloxy)ethyl]amino}-1
Intermediate 122 (80 mg), Intermediate 39 (71 mg), potassium carbonate (78 mg), and trans- N,N-dimethylcyclohexane-1 ,2-diamine (16 mg) were dissolved in anhydrous DMF (2 mL), and then copper (I) iodide (11 mg) was added. The reaction was stirred at 16O0C, under nitrogen, for 20 h. The DMF was then removed in vacuo, and the residue partitioned between water and DCM, and 4 drops of 2M Hydrochloric acid were added to neutralise the aqueous layer. The layers were separated using a hydrophobic frit, and the organic layer concentrated to give the crude title compound. MS calcd for (C30H34N4CVH)+: 531 MS found (electrospray): (M+H)+ = 531
Intermediate 124
Ethyl 1 -(4-imidazo[2,1 -b][1 ,3]thiazol-6-ylphenyl)-3-[[(frans-4- methylcyclohexyl)carbonyl](tetrahydro-3-furanyl)amino]-1W-pyrazole-4-carboxylate
To a mixture of Intermediate 96 (200 mg), 6-(4-iodophenyl)imidazo[2,1-b][1 ,3]thiazole (205 mg), frans-N,N-dimethylcyclohexane-1 ,2-diamine (39 mg) and potassium carbonate (190 mg) in anhydrous DMF (5 ml.) in a carousel reaction tube, copper (I) iodide (26 mg) was added. The reaction mixture was then stirred at 16O0C, under nitrogen, for 20 h. The reaction mixture was cooled to room temperature, and the DMF was removed in vacuo and the residue partitioned between DCM and water. The organic layer was separated using a hydrophobic frit and concentrated. This was then purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane, to give the title compound. MS calcd for (C29H33N5O4SH-H)+: 548 MS found (electrospray): (M+H)+ = 548
Intermediate 125
Ethyl 1-(4-imidazo[2,1 -Jb][1 ,3]thiazol-6-ylphenyl)-3-[[(fraπs-4- methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4- carboxylate
To a mixture of Intermediate 11 (200 mg), 6-(4-iodophenyl)imidazo[2,1-b][1,3]thiazole (197 mg), frans-N, N-dimethylcyclohexane-1 ,2-diamine (37 mg) and potassium carbonate (183 mg) in DMF (5 mL) in a carousel reaction tube, was added copper (I) iodide (25 mg). The reaction mixture was then stirred at 16O0C, under nitrogen, for 20 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The organic layer was separated using a hydrophobic frit and concentrated. This was then purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane, to give the title compound.
MS calcd for (C30H35N5O4SH-H)+: 562 MS found (electrospray): (M+H)+ = 562
Intermediate 126 Ethyl 3-{(2-ethylbutyl)[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1W-pyrazole-4-carboxylate
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (3 mL) was added sodium hydride (60% dispersion in mineral oil) (17 mg). This was stirred in a greenhouse plus reaction tube at room temperature, under nitrogen, for 0.5 h. 1-Bromo-2-ethylbutane (0.059 mL) was then added, and the reaction was stirred at 5O0C, under nitrogen, for 48 h. 2 drops of water were then added to the cooled reaction mixture, and the DMF was removed by evaporation to give the crude title compound. MS calcd for (C32H40Ne(VH)+: 557 MS found (electrospray): (M+H)+ = 557
Intermediate 127
Ethyl 3-{[(frans-4-methylcyclohexyl)carbonyl][2-(methyloxy)ethyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylate
A mixture of Intermediate 122 (100 mg), Intermediate 42 (89 mg), copper (I) iodide (14 mg), trans-N, N-dimethylcyclohexane-1 ,2-diamine (22.4 mL) and potassium carbonate (98 mg) in
DMF (2.5 mL) was stirred at 16O0C, in a Reactivial for 20 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The organic layer was separated using a hydrophobic frit and concentrated. This was then purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane, to give the title compound.
MS calcd for (C29H34N6CVH)+: 531 MS found (electrospray): (M+H)+ = 531
Intermediate 128 Ethyl 3-(cyclopentylamino)-1 -(phenylmethyl)-i H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 1 (1 g) in anhydrous DCM (20 mL) was added cyclopentanone (691 mg), acetic acid (740 mg), and sodium triacetoxyborohydride (1.74 g). The reaction mixture was then stirred at room temperature, under nitrogen, for 20 h. The mixture was then partitioned between saturated sodium bicarbonate solution and DCM, and the organic phase removed using a hydrophobic frit. This was concentrated in vacuo, and purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane, to give the title compound.
MS calcd for (C18H23N3(VH)+: 314 MS found (electrospray): (M+H)+ = 314
Intermediate 129
Ethyl 3-{cyclopentyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(phenylmethyl)-i H- pyrazole-4-carboxylate
To a stirred solution of Intermediate 128 (851 mg) in anhydrous DCM (13.6 mL), was added triphenylphosphine (1.068 g), and frans-4-methylcyclohexanecarbonyl chloride2 (654 mL). The reaction mixture was stirred at 450C, under nitrogen, for 20 h. Then, trans-4- methylcyclohexanecarbonyl chloride2 (218 mL) was added to the reaction mixture, which was then stirred at 450C, under nitrogen, for 16 h. The reaction mixture was allowed to cool, and then partitioned between saturated sodium bicarbonate solution and DCM, and the organic layer washed with brine, dried over Na2SO4 and concentrated in vacuo. This was then purified using ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane, to give the title compound.
MS calcd for (C26H35N3O3+H)+: 438
MS found (electrospray): (M+H)+ = 438
Ref 2: WO 2004/052885
Intermediate 130
Ethyl 3-{cyclopentyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1H-pyrazole-4- carboxylate
A solution of Intermediate 129 (1.045 g) in ethanol (42 mL) and concentrated hydrochloric acid (0.4 mL) was subjected to atmospheric pressure hydrogenation using 10 wt % palladium on activated carbon catalyst (315 mg, wet) for 24 h. This was then filtered through a pad of Celite (10 g), and the organics concentrated in vacuo. The residue was partitioned between saturated sodium bicarbonate solution and DCM, and passed through a hydrophobic frit. The organic phase was concentrated to give the title compound. MS calcd for (C18H29N3O3+H)+: 348
MS found (electrospray): (M+H)+ = 348
Intermediate 131
Ethyl 3-{cyclopentyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylate
Intermediate 130 (768 mg), Intermediate 42 (666 mg), potassium carbonate (733 mg), and frans-N,N-dimethylcyclohexane-1 ,2-diamine (151 mg) were dissolved in anhydrous DMF (19.2 ml_), and then copper (I) iodide (101 mg) was added. The reaction was stirred at 16O0C, under nitrogen, for 20 h. The reaction mixture was allowed to cool, and then the DMF was removed in vacuo, and the residue partitioned between water and DCM. The layers were separated using a hydrophobic frit, and the organic layer concentrated. This was then purified by ISCO companion silica chromatography, eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C3IH36N6CVH)+: 541
MS found (electrospray): (M+H)+ = 541
Intermediate 132
Ethyl 3-[(1 -methylethyl)(trifluoroacetyl)amino]-1 -(phenylmethyl)-i H-pyrazole-4- carboxylate
Intermediate 2 (1.0 g) was dissolved in diethyl ether (20 ml_) and cooled to O0C. Trifluoroacetic anhydride (0.58 ml_) was added dropwise and the reaction mixture was stirred for 1 h. The reaction mixture was warmed to room temperature and evaporated to dryness to give the title compound.
MS calcd for (C18H20F3N3O3+ H)+: 384 MS found (electrospray): (M+H)+ = 384
Intermediate 132 (0.74 g) in ethanol (25 mL) and concentrated hydrochloric acid (0.5 mL) was subjected to atmospheric pressure hydrogenation with 10% palladium on carbon (0.24 g, wet) overnight. The reaction was filtered through a Celite pad, and the filter cake washed with ethanol. The combined organics were evaporated to dryness. The residue was partitioned between DCM and saturated sodium bicarbonate solution, passed through a hydrophobic frit and the organics evaporated to dryness to give the title compound. MS calcd for (C11H14F3N3O3+ H)+: 294 MS found (electrospray): (M+H)+ = 294
Intermediate 134
Ethyl 3-[(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4- carboxylate
Copper (I) iodide (0.8 g), pyrazole intermediate 133 (3.6 g), Intermediate 42 (3.7 g), trans- N,N-dimethylcyclohexane-1 ,2-diamine (0.83 g) and potassium carbonate (4.3 g) were dissolved in DMF (125 mL). The reaction mixture was heated at 16O0C for 3 days. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organics were dried using sodium sulphate and evaporated to dryness. The residue was dissolved in DCM (-100 mL) and pulled through a frit under vacuum before loading onto a silica biotage cartridge. The cartridge was eluted with 5% EtOAc in DCM followed by 10% EtOAc in DCM. The appropriate fractions were evaporated to dryness to give the title compound. MS calcd for (C21H22N6O2+ H)+: 391 MS found (electrospray): (M+H)+ = 391
Intermediate 135
Ethyl 3-[[(4-chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1W-pyrazole-4-carboxylate
Intermediate 134 (50 mg), p-chlorobenzoyl chloride (25 μl_), and triphenylphosphine (67 mg) were dissolved in anhydrous DCM (2 ml_) and the mixture was heated at reflux overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 30 min. The organic layer was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C28H25CIN6O2+ H)+: 529/531 MS found (electrospray): (M+H)+ = 529/531
Intermediate 136
Ethyl 3-[[(2,4-dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1
Intermediate 134 (150 mg), 2,4-dichlorobenzoyl chloride (121 mg), and triphenylphosphine (201 mg) were dissolved in anhydrous DCM (5 ml.) and the mixture was heated at 4O0C overnight. 2,4-Dichlorobenzoyl chloride (30 μL) was added to the mixture which was stirred at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 hr. The organic was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C28H24CI2N6O3+ H)+: 563/565 MS found (electrospray): (M+H)+ = 563/565
Intermediate 137
Ethyl 3-((1 -methylethyl){[4-(trifluoromethyl)phenyl]carbonyl}amino)-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1W-pyrazole-4-carboxylate
Intermediate 134 (150 mg), 4-trifluoromethyl benzoyl chloride (120 mg), and triphenylphosphine (201 mg) were dissolved in anhydrous DCM (5 ml_) and the mixture was heated at 4O0C overnight. Further 4-trifluoromethyl benzoyl chloride (30 μl_) was added to the reaction and stirring was continued at 4O0C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 hr. The organic was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C29H25F3N6O3+ H)+: 563 MS found (electrospray): (M+H)+ = 563
Intermediate 138
Ethyl 3-[[(3,4-dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1
Intermediate 134 (150 mg), 3,4-dichlorobenzoyl chloride (121 mg), and triphenylphosphine (201 mg) were dissolved in anhydrous DCM (5 ml_) and the mixture was heated at 4O0C overnight. Further 3,4-dichlorobenzoyl chloride (30 μl_) was added to the reaction and stirring continued at 4O0C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic was separated and dried by passing through a hydrophobic frit and was then evaporated to dryness. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C28H24CI2N6O3+ H)+: 563/565 MS found (electrospray): (M+H)+ = 563/565
Intermediate 139
Ethyl 3-[isopropyl(4-methylbenzoyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)- 1 H-pyrazole-4-carboxylate
Copper (I) iodide (13.3 mg), Intermediate 76 (95 mg), Intermediate 42 (90 mg), trans- (1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (20 mg) and potassium carbonate (99 mg) were dissolved in DMF (3 mL) in a Reactivial. The reaction mixture was heated at 16O0C for 18 h. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic was dried by passing through a hydrophobic frit and was then evaporated to dryness. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C29H28N6O3+ H)+: 509 MS found (electrospray): (M+H)+ = 509
Intermediate 140 Ethyl-1 -(4-bromo-3-chlorophenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro- 2H-pyran-4-yl)amino]-1H-pyrazole-4-carboxylate
Intermediate 11 (4 g) was dissolved in anhydrous DMF (140 mL). 1 ,4-Dibromo-2- chlorobenzene (29.7 g), copper (I) iodide (500 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexanediamine (750 mg), and potassium carbonate (3.65 g) were added and the mixture stirred at 160°C overnight under an atmosphere of nitrogen. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated and filtered. The filtrate was passed through a hydrophobic frit to remove residual water and was concentrated in vacuo and purified directly by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C25H3IBrCIN3O4 + H)+: 552/554 MS found (electrospray): (M+H)+ = 552/554
Intermediate 141
Ethyl-1-[3-chloro-4-(2-furanyl)phenyl]-3-[[(frans-4-methylcyclohexyl) carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1H-pyrazole-4-carboxylate
Intermediate 140 (200 mg) was dissolved in DME (10 ml_). 2N sodium carbonate solution (5 mL) and Pd(PPh3)4 (20.9 mg) were added and the mixture was heated to 85°C under reduced pressure to deoxygenate the solution. The flask was flushed with nitrogen and the mixture was cooled slightly, then 2-furanylboronic acid (102 mg) was added. The reaction was stirred under an atmosphere of nitrogen at 85°C overnight. The mixture was diluted with
DCM/water which gave an emulsion. Saturated sodium bicarbonate solution was added and layers separated. The organics were dried (Na2SO4), filtered and evaporated in vacuo. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C29H34CIN3O5 + H)+: 540/542
MS found (electrospray): (M+H)+ = 540/542
Intermediate 142
Ethyl-1-[3-chloro-4-(3,5-dimethyl-4-isoxazolyl)phenyl]-3-[[(frans-4- methylcyclohexyOcarbonyll^etrahydro^H-pyran^-yOaminol-IW-pyrazole^- carboxylate
Intermediate 11 (200 mg) was dissolved in DME (10 mL). sodium carbonate solution (2N, 5 mL), Pd(PPh3)4 (20.9 mg) and 3,5-dimethylisoxazole-4-boronic acid (102 mg), were added and the mixture was heated to 85°C under reduced pressure to deoxygenate the solution. The flask was flushed with nitrogen and stirred under an atmosphere of nitrogen at 85°C for 1 day and then at 20°C for 1 day. Ethyl acetate and saturated sodium bicarbonate solution were added and the organics were separated. The aqueous layer was re-extracted with ethyl acetate and the combined organics were dried (Na2SO4), filtered and evaporated in vacuo to give the title compound. MS calcd for (C30H37CIN4O5+ H)+: 569/571
MS found (electrospray): (M+H)+ = 569/571
Intermediate 143
Ethyl 3-({2-(methyloxy)-1 -[(methyloxy)methyl]ethyl}amino)-1 -(phenylmethyl)-i H- pyrazole-4-carboxylate
To a stirred solution of Intermediate 1 (6.00 g) in anhydrous DCM (125 mL) containing acetic acid (4.00 mL) and 1 ,3-dimethoxyacetone (2.45 g) was added sodium triacetoxyborohydride (10.36 g) and the resultant mixture was stirred under nitrogen. Activated molecular sieves (4 angstrom) were added to the mixture after 22 h. More sodium triacetoxyborohydride (5.10 g) was added after 42 h. After a further 4 h the reaction mixture was filtered and the filtrate was washed with saturated aqueous sodium bicarbonate solution. The organic phase was separated using a hydrophobic frit and was then evaporated to leave a colourless oil. This was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C18H25N3O4 + H)+: 348 MS found (electrospray): (M+H)+ = 348
Intermediate 144 Ethyl-3-([(frans-4-methylcyclohexyl)carbonyl]{2-(methyloxy)-1-
[(methyloxy)methyl]ethyl}amino)-1-(phenylmethyl)-1H-pyrazole-4-carboxylate
Intermediate 143 (1.69 g) was dissolved in anhydrous DCM (23 mL). Triethylamine (1.261 mL) and frans-4-methyl-cyclohexanecarbonyl chloride (1.108 g) were added and the mixture was stirred at 50°C under a nitrogen atmosphere. Further portions of triethylamine (0.631 mL) and frans-4-methyl-cyclohexanecarbonyl chloride2 (554 mg) were added after 18 h, 23 h and 41 h and the reaction was then stirred at 50°C under an atmosphere of nitrogen for a further 18 h. The reaction was then quenched with saturated sodium bicarbonate solution (25 mL) and diluted with DCM (10 mL). The organic layer was separated using a
hydrophobic frit and evaporated in vacuo. The crude product was purified by flash chromatography over silica (Biotage, 90 g silica cartridge) eluting with cyclohexane/ethyl acetate (3:1 ), followed by cyclohexane/ethyl acetate (2:1) to give the title compound. MS calcd for (C26H37N3O5+ H)+: 472 MS found (electrospray): (M+H)+ = 472 Ref 2: WO 2004/052885
Intermediate 145
Ethyl-3-([(frans-4-methylcyclohexyl)carbonyl]{2-(methyloxy)-1- [(methyloxy)methyl]ethyl}amino)-1 H-pyrazole-4-carboxylate
A solution of Intermediate 144 (1.29 g) in ethanol (50 mL) was stirred with cone. HCI (8 drops) and a catalyst of 10% palladium on carbon (0.56 g, wet) under a hydrogen atmosphere at 50 psi. After 5 h the catalyst was removed by filtration through Celite and to the filtrate was added saturated aqueous sodium bicarbonate dropwise with stirring. The resulting milky suspension was concentrated to about half volume and was then partitioned between water and dichloromethane. The organic phase was separated and was then evaporated to give the title compound. MS calcd for (C19H3IN3O5 + H)+: 382 MS found (electrospray): (M+H)+ = 382
Intermediate 146
Ethyl-3-([(fraπs-4-methylcyclohexyl)carbonyl]{2-(methyloxy)-1- [(methyloxy)methyl]ethyl}amino)-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H- pyrazole-4-carboxylate
Intermediate 145 (183 mg) was dissolved in anhydrous DMF (4.5 mL). Intermediate 42 (144.7 mg), copper (I) iodide (21.9 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexanediamine (32.75 mg), and potassium carbonate (159.13 mg) were added and the mixture stirred at 160°C overnight in a Reactivial, then evaporated to dryness. The residue was partitioned between water and DCM, the organics separated using a hydrophobic frit
and the eluant evaporated in vacuo. The crude product was purified directly by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C31H38N6O5+ H)+: 575
MS found (electrospray): (M+H)+ = 575
Intermediate 147
Ethyl-1-(4-furo[3,2-to]pyridin-2-ylphenyl)-3-([(frans-4-methylcyclohexyl)carbonyl]{2-
(methyloxy)-1-[(methyloxy)methyl]ethyl}amino)-1H-pyrazo!e-4-carboxylate
Intermediate 145 (183 mg) was dissolved in anhydrous DMF (4.5 ml_). Intermediate 39 (144.7 mg), copper(l) iodide (21.9 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexanediamine (32.7 mg), and potassium carbonate (159.1 mg) were added and the mixture stirred at 160°C overnight in a Reactivial then evaporated to dryness. The residue was partitioned between water and DCM, the organics separated and evaporated in vacuo. The crude product was purified directly by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C31H38N6O5+ H)+: 575 MS found (electrospray): (M+H)+ = 575
Intermediate 148
6-(4,4,5,5-Tetramethyl-1 ,3,2-dioxaborolan-2-yl)-1 -benzofuran
To a stirred solution of 1-benzofuran-6-yl trifluoromethanesulfonate (3.60 g) in anhydrous dioxan (80 mL) under nitrogen was added PdCI2(dppf) (0.30 g), dppf (0.23g), potassium acetate (3.98 g) and bis(pinacolato)diboron (3.78 g) and the resultant mixture was heated at 8O0C for 19 h. The reaction mixture was diluted with ethyl acetate and then washed with saturated brine. The combined layers were filtered and then the organic phase was separated and was dried over sodium sulphate and evaporated. The residue was dissolved in dioxan (100 mL) and to this solution was added PdCI2(dppf) (0.30 g), dppf (0.23g), potassium acetate (3.98 g) and bis(pinacolato)diboron (3.78 g) and the resultant mixture was
heated at 8O0C for 19 h. The reaction mixture was diluted with ethyl acetate and then washed with saturated brine. The combined layers were filtered and then the organic phase was separated, dried over sodium sulphate and evaporated. The resultant oil was purified by flash chromatography on silica (Biotage) using cyclohexane / EtOAc (19:1) as elution solvent. Appropriate fractions were combined and then evaporated to give the title compound.
1H NMR (CDCI3) δ 7.97 (1 H, bs), 7.68 (1 H, d), 7.67 (1H, bs), 7.61 (1 H, d), 6.78 (1 H, m), 1.37 (12H, s).
Ref: Journal of Medicinal Chemistry (2004), 47(20), 4829-4837.
Intermediate 149
Ethyl 1 -[4-(1 -benzofuran-6-yl)phenyl]-3-[[(frans-4- methylcyclohexyOcarbonylKtetrahydro^H-pyran^-yOaminol-IH-pyrazole^- carboxylate
Sodium periodate (2.25 g) was added in portions to a mixture of Intermediate 148 (3.42 g; this material contains an impurity which is probably bis(pinacolato) diboron) and ammonium acetate (0.81 g) in acetone and water (1:1 , 80 mL) at room temperature under nitrogen. After 2 days the reaction mixture was concentrated by rotary evaporation and was then partitioned between saturated brine and EtOAc. The aqueous phase was separated and was extracted twice using EtOAc. The organic phases were combined then dried over sodium sulphate and finally evaporated to give an oil (0.30 g). This material was dissolved in a mixture of DME (10 mL) and sodium carbonate (2N, 5 mL) and to this solution was added Intermediate 12 (0.16 g) followed by Pd(PPh3)4 (18 mg). This mixture was heated at 1000C with stirring for 19 h and was then allowed to cool to room temperature. The reaction mixture was partitioned between saturated aqueous sodium bicarbonate solution and DCM. The aqueous phase was separated off and was extracted twice with DCM. The organic phases were all combined and passed through a hydrophobic frit and finally evaporated to give an oil. This oil was purified by flash chromatography on silica (Biotage) using cyclohexane and EtOAc (3:1) as elution solvent. Appropriate fractions were combined and then evaporated to give the title compound. MS calcd for (C13H15N3O2 + H)+: 556 MS found (electrospray): (M+H)+ = 556
Intermediate 150
Ethyl 1 -[4-(1 -benzofuran-5-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1H-pyrazole-4- carboxylate
To a mixture of Intermediate 12 (0.16 g) and 1 -benzofuran-5-yl boronic acid (0.15 g) in DME (10 mL) and sodium carbonate (2N, 5 imL) was added Pd(PPh3)4 (18 mg) and the resultant mixture was heated at 1000C with stirring under nitrogen. The reaction mixture was allowed to cool to room temperature and then saturated aqueous sodium bicarbonate (10 mL) and DCM (15 mL) were added with stirring. The aqueous layer was separated and was then extracted twice with DCM (2 X 15 mL). The organic phases were combined and evaporated and the crude product was purified by flash chromatography on silica (Biotage) using cyclohexane / EtOAc (3:1 ) as elution solvent. Appropriate fractions were combined and then evaporated to give the title compound. MS calcd for (C13H15N3O2 + H)+: 556 MS found (electrospray): (M+H)+ = 556
Intermediate 151
Ethyl 1-[4-(1-benzofuran-6-yl)-3-chlorophenyl]-3-[[(frans-4- methylcyclohexylJcarbonylKtetrahydro^W-pyran^-yOaminoJ-IW-pyrazole^- carboxylate
Sodium periodate (1.26 g) was added in portions to a mixture of crude Intermediate 148 (1.92 g; this material contains an impurity which is probably bis(pinacolato) diboron) and ammonium acetate (0.45 g) in acetone and water (1:1 ; 46 mL) at room temperature under nitrogen. After 2 days the reaction mixture was concentrated by rotary evaporation and was then partitioned between saturated brine and EtOAc. The aqueous phase was separated and was extracted twice using EtOAc. The organic phases were combined then dried over sodium sulphate and evaporated. The residue was triturated with a mixture of DCM, EtOAc and cyclohexane and filtered to give a white solid (0.15 g). This was combined with a further crop of material (0.13 g) which was obtained from the filtrate following flash chromatography on silica (Biotage) using cyclohexane and EtOAc (2:1) and then trituration with EtOAc/cyclohexane and dissolved in a mixture of DME (56 mL) and 2N sodium carbonate
(28 ml_). To this solution was added Intermediate 140 (0.16 g) followed by Pd(PPh3)4 (100 mg). This mixture was heated at 10O0C with stirring for 20 h and was then allowed to cool to room temperature. The reaction mixture was partitioned between saturated aqueous sodium bicarbonate solution and EtOAc. The aqueous phase was separated and was extracted twice with EtOAc. The organic phases were all combined, dried over sodium sulphate and evaporated to give a foam. This was purified by flash chromatography on silica (Biotage) using cyclohexane / EtOAc (4:1) as elution solvent. Appropriate fractions were combined and then evaporated to give the title compound. MS calcd for (C33H36CIN3O5 + H)+: 590/592 MS found (electrospray): (M+H)+ = 590/592
Intermediate 152
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1-[4-
(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-1H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 12 (0.65 g) in anhydrous dioxan (20 ml_) under nitrogen was added PdCI2(dppf) (0.028 g), dppf (0.021 g), potassium acetate (0.37 g) and bis(pinacolato) diboron (0.35 g) and the resultant mixture was heated at 8O0C for 19 h. The reaction mixture was allowed to cool to room temperature and was then diluted with EtOAc and washed with saturated brine. The organic phase was separated, dried over sodium sulphate and then evaporated. The crude product was purified by flash chromatography on silica (Biotage) using cyclohexane / EtOAc (2:1) as elution solvent. Appropriate fractions were combined and then evaporated to give the title compound (which contained 20% of the starting material, Intermediate 12). MS calcd for (C31H44BN3O6 + H)+: 566 MS found (electrospray): (M+H)+ = 566
Intermediate 153
Ethyl 1 -(4-furo[2,3-b]pyridin-5-ylphenyl)-3-[[(frans-4- methylcyclohexyOcarbonylKtetrahydro^H-pyran^-yOaminol-IW-pyrazole^- carboxylate
Sodium periodate (0.26 g) was added in portions to a stirred mixture of Intermediate 152 (0.68 g) and ammonium acetate (0.093 g) in acetone/water (1 :1 ; 5 ml_). After 19 h the reaction mixture was concentrated by evaporation and then partitioned between saturated brine and EtOAc. The aqueous phase was separated and then extracted with EtOAc. The organic phases were combined and then dried over sodium sulphate and evaporated to give a colourless gum (0.71 g). Part of this material (0.60 g) was dissolved in a DME/2N sodium carbonate (2:1 ; 60 ml_) and then 5-bromofuro[2,3-/)]pyridine (0.24 g) followed by Pd(PPh3)4 (0.070 g) were added and the resultant mixture was heated at 1000C for 19 h. The reaction mixture was allowed to cool to room temperature and was then partitioned between DCM and saturated aqueous sodium bicarbonate. The aqueous phase was separated and was extracted with DCM. The organic phases were combined and then evaporated. This crude product was partially purified by flash chromatography on silica (Biotage) using cyclohexane and EtOAc (2:1 ) as elution solvent. Appropriate fractions were combined and then evaporated and the residue was then further purified by MDAP HPLC to give the title compound.
MS calcd for (C32H36N4O5 + H)+: 557
MS found (electrospray): (M+H)+ = 557
Ref: Journal of Heterocyclic Chemistry, 35(6), 1305-1307; 1998
Intermediate 154
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 H- pyrrolo[2,3-b]pyridin-2-yl)phenyl]-1H-pyrazole-4-carboxylate
n-Butyl lithium (2.42 ml_, 1.6M solution in hexanes) was added dropwise to a solution of diisopropylamine (0.54 mL) in THF (dry, 5 mL) at -780C under nitrogen. This was stirred at -
780C for a further 30 mins before warming to -250C. A solution of 1-(phenylsulfonyl)-1H- pyrrolo[2,3-ύ]pyridine (500 mg) in dry THF (5 mL) was added dropwise, maintaining the internal temp at -3O0C to -2O0C. The resulting suspension was stirred at -250C for a further
25 mins. Triisopropyl borate (1.34 mL) was then added dropwise and the mixture stirred at - 2O0C for 2 h. This was allowed to warm to RT over 1 h, and was then acidified to pH 5.0 with
2N hydrochloric acid. Following extraction with EtOAc (3 x 30 mL) the combined organics were dried (Na2SO4) and evaporated to give crude boronic acid.
Tetrakis(triphenylphosphine)palladium (0) (18 mg) was added to a mixture of Intermediate 5
(150 mg) and the crude boronic acid (283 mg) in DME (10 mL) / 2N sodium carbonate solution (5 mL). The mixture was heated to 1000C under nitrogen for 4 h. On cooling 8% sodium bicarbonate solution (10 mL) and DCM (15 mL) were added. The layers were
separated and the aqueous fraction extracted further with DCM (2 x 10 mL), dried (aqueous extraction cartridge) and evaporated. This was purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1. to 8:2 to 7:3 to 6:4) to give the title compound MS calcd for (C30H35N5O4 + H)+: 514 MS found (electrospray): (M+H)+ = 514
Intermediate 155
Ethyl 3-((1 -methylethyl){[frans-4-(trif luoromethyl)cyclohexyl]carbonyl}amino)-1 -(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylate
Oxalyl chloride (0.84 mL) was added dropwise to an opaque solution of trans-4- (trifluoromethyl)cyclohexanecarboxylic acid6 (836 mg) in dry DCM (15 mL) at RT under nitrogen. The mixture was stirred at RT for 48 h and then the solvent was evaporated. A portion of the residue (64 mg) together with Intermediate 134 (84 mg), and triphenylphosphine (78 mg) were dissolved in DCM (3 mL) and the resultant solution was heated at 4O0C under nitrogen for 24 h. A further quantity of the residue (64 mg) was added and heating continued. After a further 4 h, a further quantity of residue (64 mg) was added and the reaction heated for a further 48 h. On cooling the reaction was diluted with DCM (5 mL), washed with 8% sodium bicarbonate solution (2 x 10 mL) dried through a hydrophobic frit and evaporated. The residue was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 1 :9) then EtOAc and finally MeCN/EtOAc (1 : 1 ) to give the title compound. MS calcd for (C29H31F3N6O3 + H)+: 569 MS found (electrospray): (M+H)+ = 569 Ref 5. EP 0 814 073 A
Intermediate 156
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(6-oxo-1 ,6- dihydro-3-pyridinyl)-1 W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (600 mg), 5-bromo-2(1 H)-pyridone (324 mg), copper (I) iodide (36 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (112 mg) and potassium carbonate (516 mg) in DMF (6 ml.) was heated at 12O0C in a Reactivial for 24 h. The DMF was evaporated and the residue partitioned between EtOAc (40 mL) and brine (20 mL). The aqueous fraction was extracted further with EtOAc (2 x 30 mL), dried (Na2SO4) and evaporated. The residue was purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient from 1 :1 to 1 :3) then EtOAc then EtOAc/MeCN (1 :1 ), then MeCN then MeCN/Acetone (1 :1) and finally acetone to give the title compound. MS calcd for (C22H30N4O4 + H)+: 415 MS found (electrospray): (M+H)+ = 415
Intermediate 157
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(6-
{[(trifluoromethyl)sulfonyl]oxy}-3-pyridinyl)-1H-pyrazole-4-carboxylate
Triflic anhydride (0.2 mL) was added dropwise to a solution of Intermediate 156 (453 mg) and 2, 6 di-te/f-butyl-4-methylpyridine (246 mg) in dry DCM (10 mL) at O0C under nitrogen. After 30 min the solvent was evaporated and the residue purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3) to give the title compound.
MS calcd for (C23H29F3N4O6S + H)+: 547 MS found (electrospray): (M+H)+ = 547
Intermediate 158 Ethyl 1 -[6-(1 -benzofuran^-ylJ-S-pyridinylJ-S-I^frans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1H-pyrazole-4-carboxylate
Tetrakis(triphenylphosphine)palladium (0) (22 mg) was added to a suspension of Intermediate 157 (215 mg) and benzofuran-2-boronic acid (96 mg) in DME (4 mL)/ 2N sodium carbonate solution (1.5 mL). The mixture was heated to 1000C under nitrogen for 2 h. On cooling, DCM (10 mL) was added and the layers separated. The aqueous fraction was extracted further with DCM (2 x 10 mL), the combined organic fractions dried (aqueous extraction cartridge) and evaporated. This was purified by SPE chromatography, eluting with
cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3 to 6:4 to 1 :1 ) to give the title compound.
MS calcd for (C30H34N4O4 + H)+: 515
MS found (electrospray): (M+H)+ = 515
Intermediate 159
1 ,1 -Dimethylethyl 2-(5-{4-[(ethyloxy)carbonyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazol-1 -yl}-2-pyridinyl)-1 H- indole-1 -carboxylate
Tetrakis (triphenylphosphine) palladium (0) (22 mg) was added to a suspension of Intermediate 157 (215 mg) and (1-{[(1 ,1-dimethylethyl)oxy]carbonyl}-1H-indol-2-yl) boronic acid (154 mg) in DME (4 mL) / 2N sodium carbonate solution (1.5 ml_). The mixture was heated to 1000C under nitrogen for 2 h. On cooling, DCM (10 mL) was added and the layers separated. The aqueous fraction was extracted further with DCM (2 x 10 mL), the combined organic fractions dried (hydrophobic frit) and evaporated. This was purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3 to 6:4 to 1 :1) to give the title compound. MS calcd for (C35H43N5O5 + H)+: 614 MS found (electrospray): (M+H)+ = 614
Intermediate 160
Ethyl 1 -[6-(1 H-indol-Σ-yO-a-pyridinyll-S-fKfra/is^-methylcyclohexylJcarbonylKI - methylethyl)amino]-1H-pyrazole-4-carboxylate
Intermediate 159 (214 mg) was treated with 4N hydrogen chloride in dioxan (5 mL) and the resulting solution left to stir at room temperature for 24 h. The solvent was evaporated, the residue basified with 8% sodium bicarbonate solution (10 mL), extracted with EtOAc (3 x 20 mL), dried (Na2SO4) and evaporated. This was purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3 to 6:4) to give the title compound.
MS calcd for (C30H35N5O3 + H)+: 514 MS found (electrospray): (M+H)+ = 514
Intermediate 161
Ethyl 1 -(5-chloro-2-pyridinyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (600 mg), 2-bromo-5-chloropyridine (356 mg), copper (I) iodide (36 mg), frans-(1 R,2R)-N,Nl-bismethyl-1 ,2- cyclohexane diamine (112 mg) and potassium carbonate (516 mg) in DMF (6 mL) was heated at 12O0C in a Reactivial for 24 h. The DMF was evaporated and the residue partitioned between EtOAc (30 mL) and brine (30 mL). The aqueous layer was extracted further with EtOAc (2 x 30 mL), the combined organics dried (Na2SO4) and evaporated. The residue was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3) to give the title compound.
MS calcd for (C22H29CIN4O3 + H)+: 433/435 MS found (electrospray): (M+H)+ = 433/435
Intermediate 162 Ethyl 1 -[5-(1 -benzof uran-2-yl)-2-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1W-pyrazole-4-carboxylate
Intermediate 161 (225 mg), 2-benzofuran boronic acid (169 mg), cesium fluoride (236 mg), 2-dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl (61 mg) and palladium (II) acetate (22 mg) in dioxan (7 mL) were stirred at room temperature under nitrogen for 24 h. The solvent was evaporated and the resulting oil dissolved in EtOAc (20 mL) and washed with 8% sodium bicarbonate solution (2 x 10 mL), dried (Na2SO4) and evaporated. This was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3) to give the title compound. MS calcd for (C30H34N4O4 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 163
1 ,1 -Dimethylethyl 2-(6-{4-[(ethyloxy)carbonyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazol-1 -yl}-3-pyridinyl)-1 H- indole-1-carboxylate
Intermediate 161 (225 mg), (1-{[(1 ,1-dimethylethyl)oxy]carbonyl}-1H-indol-2-yl)boronic acid (272 mg), cesium fluoride (236 mg), 2-dicyclohexylphosphino-2'-(N,N- dimethylamino)biphenyl (61 mg) and palladium (II) acetate (22 mg) in dioxan (7 ml_) were stirred at room temperature under nitrogen for 48 h. More (1-{[(1 ,1- dimethylethyl)oxy]carbonyl}-1H-indol-2-yl)boronic acid (136 mg), palladium (II) acetate (22 mg) and dioxan (6 ml.) were added and stirring continued for 24 h. The solvent was evaporated, the residue dissolved in EtOAc (20 ml.) and washed with 8% sodium bicarbonate solution (2 x 20 ml_), dried (Na2SO4) and evaporated. This was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2) to give the crude title compound. MS calcd for (C35H43N5O5 + H)+: 614 MS found (electrospray): (M+H)+ = 614
Intermediate 164
Ethyl 1 -[5-(1 H-indol^-yO^-pyridinyll-S-IKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1H-pyrazole-4-carboxylate
Intermediate 163 (182 mg) in 4N hydrogen chloride / dioxan (3 mL) was stirred at room temperature for 2 days and then evaporated. The resulting residue was basified with 8% sodium bicarbonate solution (10 mL) and extracted with EtOAc (3 x 20 mL), the combined organics dried (Na2SO4) and evaporated. This was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3) to give the crude title compound. MS calcd for (C30H35N5O3+ H)+: 514
MS found (electrospray): (IVMH)+ = 514
Intermediate 165
Ethyl 1 -[6-(1 -benzofuran-Σ-yO-S-pyridazinyll-a-tKfrans^-methylcyclohexyOcarbonylKI ■ methylethyl)amino]-1 H-pyrazole-4-carboxylate
Tetrakis(triphenylphosphine)palladium (0) (20 mg) was added to a mixture of Intermediate 174 (203 mg) and benzofuran-2-boronic acid (89 mg) in DME (4 ml.)/ 2N sodium carbonate solution (2.5 ml_). The mixture was heated to 1000C under nitrogen for 2 h. On cooling the solvent was evaporated and partitioned between EtOAc (10 ml.) and 8% sodium bicarbonate solution (10 mL). The aqueous layer was extracted further with EtOAc (2 x 10 ml_), dried (Na2SO4) and evaporated. This was purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3 to 6:4 to 1 :1 ) to give the title compound. MS calcd for (C29H33N45O4 + H)+: 516 MS found (electrospray): (M+H)+ = 516
Intermediate 166
Ethyl 1 -[4-(7-aminopyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (150 mg), Intermediate 99 (135 mg), copper (I) iodide (10 mg), trans-C\ R, 2R)-N, N'-bismethyl-1 ,2-cyclohexane diamine (26 mg) and potassium carbonate (129 mg) in DMF (1.5 mL) was heated at 16O0C in a Reactivial for 6 h. The DMF was evaporated and the residue partitioned between EtOAc (20 mL) and water (15 mL). The aqueous was extracted further with EtOAc (2 x 10 mL), dried (Na2SO4) and evaporated. The residue was purified by SPE chromatography eluting with cyclohexane to cyclohexane/EtOAc (gradient from 9:1 to 1 :9) to give the title compound. MS calcd for (C29H35N7O3 + H)+: 530 MS found (electrospray): (M+H)+ = 530
Intermediate 167
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(6- methylpyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (121 mg), Intermediate 98 (109 mg), copper (I) iodide (10 mg), fraπs-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (21 mg) and potassium carbonate (104 mg) in DMF (1.5 mL) was heated at 16O0C in a Reactivial for 6 h. The DMF was evaporated and the residue partitioned between EtOAc (20 mL) and water (15 mL). The aqueous was extracted further with EtOAc (2 x 10 mL), dried (Na2SO4) and evaporated. The residue was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 1 :1 ) to give the title compound. MS calcd for (C30H36N6O3 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 168
Ethyl 3-[[(frans-4-ethylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1W-pyrazole-4-carboxylate
A solution of Intermediate 134 (84 mg), frans-4-ethylcyclohexanecarbonyl chloride (53 mg) and triphenylphosphine (78 mg) in DCM (3 mL) was heated to 4O0C under nitrogen for 24 h. A further quantity of frans-4-ethylcyclohexanecarbonyl chloride (53 mg) was added and heating continued. After a further 4 h, a further quantity of frans-4-ethylcyclohexanecarbonyl chloride (53 mg) was added and the reaction heated for a further 48 h. On cooling the reaction was diluted with DCM (5 mL), washed with 8% sodium bicarbonate solution (2 x 10 mL) dried (hydrophobic frit) and evaporated. The residue was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 1 :9) then EtOAc to give the title compound. MS calcd for (C30H36N6O3 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 169
Ethyl 3-{[(frans-4-methylcyclohexyl)carbonyl][(3f?)-tetrahydro-3-furanyl]amino}-1-
(phenylmethyl)-1H-pyrazole-4-carboxylate
A mixture of S (+) 3-hydroxytetrahydrofuran (0.16 mol, 13.2 mL) and dry pyridine (15.8 ml_, 0.195 mol) in dry dichloromethane (400 mL) was cooled to -1O0C under nitrogen. Triflic anhydride (29 mL, 0.171 mol) was added via a dropping funnel ensuring the temperature did not exceed O0C. The solution was then stirred for one hour at -10°C for 1 h. The reaction mixture was then quenched with 2N hydrochloric acid, the organic layer separated, dried over sodium sulphate and then passed through a silica gel plug to remove colouration.
The resultant tetrahydro-3-furanyl trifluoromethanesulfonate solution was approximately 320 mL in volume and used in the following process.
To a stirred solution of Intermediate 15, 2Og (0.054 mol.) in dry THF (400 mL) and dry DMF (50 mL) at -780C was added a solution of potassium hexamethyldisilazide (140 mL, 0.5 M in toluene) over 0.75 h and the reaction allowed to stir for a further 1 h. The tetrahydro-3-furanyl trifluoromethanesulfonate solution (160 mL, corresponding to 0.07 moles.) was added dropwise over 1 h at -780C. The reaction mixture was then allowed to reach -1O0C over 2 h.
A further 30 mL (0.013 mol) of the tetrahydro-3-furanyl trifluoromethanesulfonate solution was then added to the reaction mixture and stirring continued for another 1 h at -1O0C. The reaction was then quenched by the addition of hydrochloric acid (2N, 200 mL) and extracted with ethyl acetate (500 mL). The organic layer was then separated, washed with brine, dried over sodium sulphate and evaporated to oil. The crude product was purified by silica chromatography (Flash 75 Biotage) eluting with cyclohexane/ethyl acetate (3:1) to give the title compound.
MS calcd for (C25H33N3O4+ H)+: 440 MS found (electrospray): (M+H)+ = 440
Intermediate 170 Ethyl S^^frans^-methylcyclohexyOcarbonyllttS^-tetrahydro-S-furanylJamino}^ H- pyrazole-4-carboxylate
Intermediate 169 (20 g) was dissolved in 800 ml_ of ethanol (800 mL) and concentrated hydrochloric acid (10 mL) was added under vacuum to 10% palladium on carbon (5g, wet) in a hydrogenation flask. The reaction mixture was then placed under atmospheric hydrogen and stirred at room temperature for 16 h. After this time the reaction was purged and filtered under nitrogen. The filtrate was then made basic with sodium bicarbonate solution. The reaction was then evaporated to low volume and extracted with ethyl acetate. The ethyl acetate was then separated, dried over sodium sulphate, filtered and evaporated to give the title compound.
MS calcd for (C18H27N3O4 + H)+:350 MS found (electrospray): (M+H)+= 350
Intermediate 171
Ethyl 3-{[(trans-4-methylcyclohexyl)carbonyl][(3S)-tetrahydro-3-furanyl]amino}-1-
(phenylmethyl)-1H-pyrazole-4-carboxylate
A solution of R(-)-3-hydroxytetrahydrofuran (6.65 mL, 0.008 mol) in dry dichloromethane (200 mL) and dry pyridine (7.9 mL, 0.82 mol) was cooled to -1O0C under nitrogen. Triflic anhydride (14.6 mL, 0.087 mol) was added via a dropping funnel ensuring the temperature did not exceed O0C. The solution was then stirred for one hour at -1O0C. The reaction mixture was then quenched with 2N hydrochloric acid. The organic layer was separated, dried over sodium sulphate and then passed through a silica gel plug to remove colouration. The resultant tetrahydro-3-furanyl trifluoromethanesulfonate solution was approximately 140 mL in volume and used in the following process.
To a stirred solution of intermediate 15 (10g, 0.054 mol.) in dry THF (200 mL) and dry DMF (25 mL) at -780C was added a solution of potassium hexamethyldisilazide in hexane (70 mL, 0.5M in toluene) over 0.75 h and the reaction allowed to stir for a further 1 h. 100 mL of the tetrahydro-3-furanyl trifluoromethanesulfonate solution (corresponding to 0.054 moles.) was then added dropwise over 1 h at -780C. The reaction mixture was then allowed to reach - 1O0C over 2 h. Another 40 mL (0.02mol) of the tetrahydro-3-furanyl trifluoromethanesulfonate solution was then added to the reaction mixture and stirring continued for 1 h at -1O0C. The reaction was then quenched with 2N hydrochloric acid (100 mL) and extracted with ethyl acetate (500 mL). The organic layer was then separated, washed with brine, dried over sodium sulphate, filtered and evaporated to an oil. The crude product was purified by ISCO silica chromatography eluting with ethyl acetate/cyclohexane (a gradient of 5-100%) to give the title compound. MS calcd for (C25H33N3O4+ H)+: 440
MS found (electrospray): (M+H)+ = 440
Intermediate172
Ethyl 3-{[(frans-4-methylcyclohexyl)carbonyl][(3S)-tetrahydro-3-furanyl]amino}-1W- pyrazole-4-carboxylate
A solution of Intermediate 171 (7.2 g) in 500 mL of ethanol (500 ml_) and concentrated hydrochloric acid (3.5 mL) was added under vacuum to 10% palladium on carbon (3g, wet) in a hydrogenation flask. The reaction mixture was then placed under atmospheric hydrogen and stirred at room temperature for 16 h. After this time the reaction was purged and filtered under nitrogen. The filtrate was then made basic with sodium bicarbonate solution and then evaporated to low volume and extracted with ethyl acetate. The ethyl acetate layer was then separated, dried over sodium sulphate, filtered and evaporated to give the title compound. MS calcd for (C18H27N3O4 + H)+: 350 MS found (electrospray): (M+H)+= 350
Intermediate 173
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(6-oxo-1 ,6- dihydro-3-pyridazinyl)-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (917 mg), 6-bromo-3-pyridazinol7(500 mg), copper (I) iodide (57 mg), frans-(1R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (100 mg) and potassium carbonate (790 mg) in DMF (5 mL) was heated at 16O0C for 24 h. The reaction was cooled and the DMF evaporated. The residue was partitioned between EtOAc (100 mL) and water (50 mL). The aqueous was extracted further with EtOAc (2 x 50 mL), dried (Na2SO4) and evaporated. The residue was purified by SPE chromatography eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 2:8) then EtOAc then MeCN then MeCN/acetone (1 :1) and finally MeOH to give the title compound. MS calcd for (C21H29N5O4 + H)+: 416 MS found (electrospray): (M+H)+ = 416
Ref 6: Ger. Offen. (1974) 58pp DE 2316821
Intermediate 174
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(6-
{[(trifluoromethyl)sulfonyl]oxy}-3-pyridazinyl)-1H-pyrazole-4-carboxylate
Triflic anhydride (0.12 mL) was added dropwise to a solution of Intermediate 173 (197 mg) and 2,6 di-terf-butyl-4-methylpyridine (149 mg) in dry DCM (5 mL) at O0C under nitrogen. After 1 h the solvent was evaporated and the residue purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient from 9:1 to 8:2 to 7:3 to 6:4) to give the title compound. MS calcd for (C22H28F3N5O6S + H)+: 548 MS found (electrospray): (M+H)+ = 548
Intermediate 175
Ethyl-1-{3-chloro-4-[6-(methyloxy)-3-pyridinyl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4- carboxylate
Intermediate 140 (200 mg) was dissolved in DME (10 mL). 2N sodium carbonate solution (5 mL), Pd(PPh3J4 (20.9 mg) and 6-methoxy-3-pyridinylboronic acid (110.7 mg) were added and the mixture was heated to 85°C under vacuum line. The flask was flushed with nitrogen and was stirred at 85°C overnight. The reaction was cooled and diluted with EtO Ac/saturated sodium bicarbonate solution. The layers were separated and the aqueous layer was re- extracted with ethyl acetate. The combined organics were dried (Na2SO4), filtered and evaporated in vacuo to give the title compound. MS calcd for (C31H37CIN4O5 + H)+: 581/583 MS found (electrospray): (M+H)+ = 581/583
Oxalyl chloride (4.59 mL) was added dropwise to a solution of trans-Λ- trifluoromethylcyclohexane carboxylic acid6 (6.85 g) in dry DCM (100 mL) at room temperature under nitrogen. The reaction was left to stir at room temperature for 24 h. The solvent was evaporated to give the title compound.
1H NMR (DMSO) (of methyl ester formed in situ by the addition of methanol) § 3.6 (3H, s),
1.10-1.46 (4H1 m), 1.79-1.95 (4H1 m), 2.19-2.4 (2H, m).
Ref 5: EP 0 814 073 A
Intermediate 177
(1 £)-1 -(4-Bromophenyl)ethanone O-(3-nitrophenyl)oxime
A mixture of O-(3-nitrophenyl)hydroxylamine (4.62 g) and 4-bromoacetophenone (5.97 g) in ethanol (100 mL) was stirred at room temperature and treated with concentrated hydrochloric acid (3 drops). After ca. 5 min. a pale yellow solid precipitated from the mixture. Stirring was continued for a further 30 minutes. The solid was collected by filtration, washed with cold ethanol, then cyclohexane to give the title compound.
1H NMR (d6-DMSO) δ 8.07 (1 H, m), 7.94 (1 H, m), 7.81 (2H, d), 7.75-7.65 (4H, m), 2.49 (3H1 s)
Intermediated
2-(4-Bromophenyl)-4-nitro-1-benzofuran
and Intermediate 245 2-(4-Bromophenyl)-6-nitro-1-benzofuran
A mixture of Intermediate 177 (5.00 g), formic acid (170 ml.) and concentrated hydrochloric acid (25 ml_) was stirred and heated at 800C for 18 h. The mixture was cooled to room temperature and the solid collected by filtration, washed with water and dried under vacuum at 400C overnight. A portion of the solid (1 g) was purified by ISCO Companion silica chromatography eluting with ethyl acetate/cyclohexane (gradient of 0-30%) to give Intermediate 178.
1H NMR (d6-DMSO) δ 8.24 (1 H, dd), 8.16 (1 H, d), 8.07 (2H, m), 8.04 (1 H, m) 7.76 (2H, d), 7.59 (1 H, t)
Further elution gave Intermediate 245. 1H NMR (d6-DMSO) .δ 8.57 (1 H, d), 8.19 (1 H, dd), 7.95 (2H, d), 7.91 (1 H, d) 7.78 (2H, d), 7.75 (1 H, s).
Intermediate 179
Ethyl 3-((1-methylethyl){[trans-4-(trifluoromethyl)cyclohexyl]carbonyl}amino)-1- (phenylmethyl)-i H-pyrazole-4-carboxylate
A solution of Intermediate 2 (3.5 g), Intermediate 176 (3.9 g) and PPh3 (6.4 g) in DCM (11 ml.) was heated at 500C for 2 days. After cooling, the mixture was stirred vigorously with saturated sodium bicarbonate solution for 1 h. The organic phase was separated, evaporated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-50% ethyl acetate in cyclohexane to give the title compound. MS calcd for (C24H30F3N3O3 + H)+: 466 MS found (electrospray): (M+H)+ = 466
Intermediate 180
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-1H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 5 (5.96 g) in anhydrous dioxan (200 mL) under nitrogen was added PdCI2 (dppf) (0.28 g), DPPF (0.21 g), potassium acetate (3.70 g) and
bis(pinacolato) diboron (3.50 g). This mixture was heated at 1000C for 2 h and then at 80°C for 19 h. The reaction was diluted with EtOAc and then washed with saturated brine. The organic phase was separated, dried (Na2SO4) and evaporated. The residue was purified using chromatography over silica gel (Biotage) eluting with EtOAc/cyclohexane (1 :2). Appropriate fractions were combined and then evaporated to give the title compound which was contaminated_with 20% of unreacted Intermediate 5. MS calcd for (C3IH44BN3O6 -H)+: 566 MS found (electrospray): (M -H)+ = 566
Intermediate 181
(4-{4-[(Ethyloxy)carbonyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2W- pyran-4-yl)amino]-1 H-pyrazol-1 -yl}phenyl)boronic acid
Sodium periodate (0.31 g) was added in portions to a stirred mixture of Intermediate 152 (0.74 g) and ammonium acetate (0.10 g) in acetone-water (1:1 ; 10 ml_). After 20 h, the reaction mixture was concentrated by evaporation and then was partitioned between EtOAc and brine. The aqueous phase was separated and extracted twice with EtOAc. The extracts and organic phase were combined, dried and finally evaporated to give the title compound.
TLC (Silica gel 60 F254) eluted with 1 :1 EtOAc/ cyclohexane, Rf = 0.05.
Intermediate 182
Ethyl 3-((1 -methylethyl){[trans-4-(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H- pyrazole-4-carboxylate
A solution of Intermediate 179 (3.1 g) in ethanol (120 mL) and cone. HCI (1.5 ml_) was stirred with wetted palladium (10% wt.) on charcoal catalyst (720 mg) at room temperature under an atmosphere of hydrogen for 16 h. The mixture was filtered through Celite, neutralised by addition of aqueous sodium bicarbonate and evaporated under vacuum. The residue was partitioned between water and DCM then the organic phase was separated using a hydrophobic frit and evaporated to give the title compound.
MS calcd for (C17H24F3N3O3 + H)+: 376 MS found (electrospray): (M+H)+ = 376
Intermediate 183 2-(4-Bromophenyl)-3-methyl-1H-indole
To phenyl hydrazine (200 mg) was added 1-(4-bromophenyl)-1-propanone (286 mg) and trichloroacetic acid (1 g). The mixture was stirred at 1000C for 5 minutes. The reaction was cooled by the addition of water and the precipitate was filtered, washed with water and dried to give the title compound.
MS calcd for (C15H12BrN + H)+: 286/288
MS found (electrospray): (M+H)+ = 286/288
Intermediate 184
Ethyl 3-[[(trans-4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1-[4-(3- methyl-1 H-indol-2-yl)phenyl]-1 H-pyrazole-4-carboxylate
To Intermediate 11 (100 mg) was added Intermediate 183 (86 mg), copper iodide (12 mg), (1 R1 2R)-1 ,2-bis(methylamino)cyclohexane (18 mg), potassium carbonate (91.2 mg) and DMF (3 ml_). The reaction was heated in a Reactivial at 16O0C overnight. The reaction was cooled, solvent removed and the residue was partitioned between DCM and water. The organics were separated using a hydrophobic frit and concentrated. The crude material was purified by 12 g silica ISCO Companion flash column using 0-100% EtOAc in cyclohexane to give the title compound. MS calcd for (C34H40N4O4 + H)+: 569 MS found (electrospray): (M+H)+ = 569
(1 £/Z)-1-(4-Bromophenyl)-2-(6-chloro-2-pyridinyl)ethanone oxime9 (832 mg) was dissolved in dimethoxyethane (10 mL) and cooled to O0C under nitrogen. Trifluoroacetic anhydride (0.36 mL) was added and the mixture allowed to warm to 150C. The solution was then cooled to O0C and triethylamine (0.71 mL) added. The reaction mixture was allowed to warm to room temperature and stirred for 1.5 h. Iron (II) chloride (6 mg) was added and the mixture heated at reflux for 3 h. The solvent was removed in vacuo and the residue purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C13H8BrCIN2 + H)+: 307/309/311 MS found (electrospray): (M+H)+ = 307/309/311 Ref 8: Bioorganic & Medicinal Chemistry; (2005), 13(18), 5346-5361
Intermediate 186 Ethyl 1 -[4-(7-chloropyrazolo[1 ,5-a]pyridin-2-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 185 (105 mg), copper (I) iodide (14 mg), trans-N, N'-bismethyl-i ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between chloroform and water. The chloroform layer was separated using a hydrophobic frit and concentrated. This residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H34CIN5O3 + H)+: 548/550 MS found (electrospray): (M+H)+ = 548/550
Intermediate 187
Ethyl 1 -[5-(6-chloroimidazo[1 ,2-a]pyridin-2-yl)-2-thienyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), 2-(5-bromo-2-thienyl)-6-chloroimidazo[1 ,2-a]pyridine (100 mg), copper (I) iodide (14 mg), ^aπs-N,N'-bismethyl-1 ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between chloroform and water. The chloroform layer was separated using a hydrophobic frit and concentrated. This residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C28H32CIN5O3S + H)+: 554/556 MS found (electrospray): (M+H)+ = 554/556
Intermediate 188
Ethyl 1 -(5-imidazo[1 ,2-a]pyridin-2-yl-2-thienyl)-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), 2-(5-bromo-2-thienyl)imidazo[1 ,2-a]pyridine (89 mg), copper (I) iodide (14 mg), trans-N, N'-bismethyl-i ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 mL) was heated at 16O0C in a Reactivial for 18 h.
The DMF was removed in vacuo and the residue partitioned between chloroform and water. The chloroform layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C28H33N5O3S + H)+: 520
MS found (electrospray): (M+H)+ = 520
Intermediate 189
3-(5-Bromo-2-thienyl)-3-oxopropanenitrile (1.8 g) was suspended in ethanol (50 mL) and treated with acetic acid (2 mL). Hydrazine monohydrate (0.758 mL) was added and the mixture heated at 7O0C for 6 h and then allowed to stand at room temperature for 18 h. The ethanol was removed in vacuo and the residue partitioned between DCM and saturated sodium hydrogen carbonate solution. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C7H6BrN3S + H)+: 244/246 MS found (electrospray): (M+H)+ = 244/246
Intermediate 190
2-(5-Bromo-2-thienyl)pyrazolo[1,5-a]pyrimidine
A solution of Intermediate 189 (603 mg) in acetic acid (7 mL) was treated with 1 ,1 ,3,3- tetramethoxypropane (0.488 mL) and the mixture heated at 11O0C for 2.5 h. The acetic acid was removed in vacuo and the residue partitioned between chloroform and saturated sodium hydrogen carbonate solution. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C10H6BrN3S+ H)+: 280/282 MS found (electrospray): (M+H)+ = 280/282
Intermediate 191
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(5-pyrazolo[1 ,5- a]pyrimidin-2-yl-2-thienyl)-1 H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 190 (96 mg), copper (I) iodide (14 mg), trans-N, N'-bismethyl-1 ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This residue was purified by ISCO Companion
silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C27H32N6O3S + H)+: 521
MS found (electrospray): (M+H)+ = 521
Intermediate 192
EthyM-[4-(2-chloroimidazo[2,1-b][1,3]thiazol-6-yl)phenyl]-3-[[(^ans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (200 mg), 6-(4-bromophenyl)-2-chloroimidazo[2,1- ύ][1 ,3]thiazole10 (214 mg), copper (I) iodide (28 mg), trans-N, N'-bismethyl-i ,2- cyclohexane diamine (42 mg) and potassium carbonate (207 mg) in DMF (4 ml.) was heated at 16O0C in a
Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between
DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This residue was purified by ISCO companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C28H32CIN5O3S + H)+: 554/556
MS found (electrospray): (M+H)+ = 554/556
Ref 9:European Journal of Medicinal Chemistry; (2001), 36(9), 743-746.
Intermediate 193
(2E/Z)-3-Amino-3-(4-bromo-2-chlorophenyl)-2-propenenitrile
4-Bromo-2-chlorobenzonitrile (3.16 g) was dissolved in toluene (80 ml_) and treated with acetonitrile (1.49 ml_). Potassium tert-butoxide (4.9 g) was added and the mixture stirred under nitrogen for 18 h. Diethyl ether (200 ml.) and saturated sodium bicarbonate solution (120 mL) were added and the mixture stirred vigorously for 10 min. The organic layer was separated, dried (Na2SO4) and concentrated in vacuo. The residue was triturated with dry ether and the solid filtered to give the title compound.
1H NMR (CDCI3) δ 7.63 (1 H, d), 7.47 (1H, dd), 7.26 (1 H, d), 4.93 (2H, br), 4.07 (1H, s)
Intermediate 193 (802 mg) was dissolved in chloroform (50 mL) and treated with 3N aqueous HCI solution (50 mL). The mixture was stirred vigorously at room temperature for 3 days. The chloroform layer was separated using a hydrophobic frit and concentrated to give the title compound.
MS calcd for (C9H5BrCINO - H)+: 256/258/260 MS found (electrospray): (M-H)+ = 256/258/260
Intermediate 195
3-(4-Bromo-2-chlorophenyl)-1H-pyrazol-5-amine
Intermediate 194 (606 mg) was suspended in isopropanol (20 mL) and treated with hydrazine monohydrate (0.341 mL). Acetic acid (0.2 mL) was added and the mixture heated at reflux for 6 h. The solvent was removed in vacuo and the residue partitioned between chloroform and saturated sodium bicarbonate solution. The chloroform layer was separated using a frit and concentrated. The residue was triturated with chloroform to give the title compound. MS calcd for (C9H7BrCIN3 - H)+: 272/274/276 MS found (electrospray): (M-H)+ = 272/274/276
Intermediate 196 2-(4-Bromo-2-chlorophenyl)pyrazolo[1,5-a]pyrimidine
A solution of Intermediate 195 (491 mg) in acetic acid (6 mL) was treated with 1 ,1 ,3,3- tetramethoxypropane (0.355 mL) and the mixture heated at 110°C for 3 h. The acetic acid was removed in vacuo and the residue partitioned between chloroform and saturated sodium hydrogen carbonate solution. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by SPE chromatography eluting with a stepped gradient of ethyl acetate in cyclohexane (10:90 to 70:30) to give the title compound. MS calcd for (C10H6BrN3S+ H)+: 308/310/312
MS found (electrospray): (M+H)+ = 308/310/312
Intermediate 197
Ethyl 1 -(3-chloro-4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-3-[[(frans-4- methylcyclohexyOcarbonylKI -methylethyl)amino]-1 W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (200 mg), Intermediate 196 (210 mg), copper (I) iodide (28 mg), frans-N,N'-bismethyl-1 ,2- cyclohexane diamine (42 mg) and potassium carbonate (207 mg) in DMF (4 mL) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C29H33CIN6O3 + H)+: 549/551 MS found (electrospray): (M+H)+ = 549/551
Intermediate 198 (2E/Z)-3-Amino-3-(4-bromo-2-methylphenyl)-2-propenenitrile
4-Bromo-2-methylbenzonitrile (2.0 g) was dissolved in toluene (40 mL) and treated with acetonitrile (1.02 mL). Potassium tert-butoxide (3.37 g) was added and the mixture stirred under nitrogen overnignt. Diethyl ether (150 mL) and saturated sodium bicarbonate solution (100 mL) were added and the mixture stirred vigorously for 10 min. The organic layer was separated, dried (Na2SO4) and concentrated in vacuo. The residue was triturated with ether and the solid filtered to give the title compound. 1H NMR (CDCI3) δ 7.41 (1H, br. d), 7.36 (1H, br. d), 7.14 (1 H, d), 4.86 (2H, br), 3.94 (1H, s).
Intermediate 199 3-(4-Bromo-2-methylphenyl)-3-oxopropanenitrile
Intermediate 198 (216 mg) was dissolved in chloroform (10 ml.) and treated with 3N aqueous HCL solution (8 ml_). The mixture was stirred vigorously at room temperature for 30 h. The chloroform layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C10H8BrNO - H)+: 236/238 MS found (electrospray): (M-H)+ = 236/238
Intermediate 200 3-(4-Bromo-2-methylphenyl)-1H-pyrazol-5-amine
Intermediate 199 (540 mg) was dissolved in ethanol (20 ml.) and treated with hydrazine monohydrate (0.22 ml_). The mixture was heated at reflux for 2 h. More hydrazine monohydrate (0.2 mL) was added followed by acetic acid (0.1 ml_). The mixture was heated at reflux for 6 h. The solvent was removed in vacuo and the residue partitioned between chloroform and saturated sodium bicarbonate solution. The chloroform layer was separated using a hydrophobic frit and concentrated. The residue was dissolved in DCM and loaded onto a 2Og silica SPE cartridge. The cartridge was eluted with ethyl acetate:cyclohexane (50:50, 75:25, 100:0) and then with ethyl acetate: methanol (95:5, 90:10, 80:20) to give the title compound.
MS calcd for (C10H10BrN3 - H)+: 252/254 MS found (electrospray): (M-H)+ = 252/254
Intermediate 201 2-(4-Bromo-2-methylphenyl)pyrazolo[1 ,5-a]pyrimidine
A solution of Intermediate 200 (559 mg) in acetic acid (8 mL) was treated with 1 ,1 ,3,3- tetramethoxypropane (0.437 mL) and the mixture heated at 110°C for 3 h. The acetic acid was removed in vacuo and the residue partitioned between chloroform and saturated sodium hydrogen carbonate solution. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C13H10BrN3+ H)+: 288/290
MS found (electrospray): (M+H)+ = 288/290
Intermediate 202
Ethyl a-IKfrans^-methylcyclohexyOcarbonylKI -methylethyl)amino]-1 -(3-methyl-4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (200 mg), Intermediate 201 (198 mg), copper (I) iodide (28 mg), trans-H, N -bismethyl-1 ,2- cyclohexane diamine (42 mg) and potassium carbonate (207 mg) in DMF (4 ml.) was heated at 16O0C in a Reactivial for 18 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C30H36N6O3 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 203
Ethyl 1 -(4-f uro[3,2-to]pyridin-2-ylphenyl)-3-[(1 -methylethyl)amino]-1 H-pyrazole-4- carboxylate
Copper (I) iodide (150 mg), Intermediate 133 (970 mg), Intermediate 39 (1.0 g), rac-1 ,2- dimethylcyclohexane diamine (225 mg) and potassium carbonate (1.14 g) were dissolved in DMF (28 ml_). The reaction mixture was heated at 16O0C for 2 days. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic was washed with brine and was dried by passing through a hydrophobic frit and evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C22H22N4O3+ H)+: 391 MS found (electrospray): (M+H)+ = 391
Intermediate 204 Ethyl 3-[[(2,4-dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-f uro[3,2-to]pyridin-2- ylphenyl)-1 H-pyrazole-4-carboxylate
Intermediate 203 (152 mg), 2,4-dichlorobenzoyl chloride (161 mg), and triphenylphosphine (252 mg) were dissolved in anhydrous DCM (5 mL) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C29H24CI2N4O4+ H)+: 563/565/566 MS found (electrospray): (M+H)+ = 563/565/566
Intermediate 205
Ethyl S-IKfrans-A-methylcyclohexyOcarbonylKI-methylethyOaminol-i^-IΘ-
(trifluoromethyl)imidazo[1,2-a]pyridin-2-yl]phenyl}-1W-pyrazole-4-carboxylate
Copper (I) iodide (13.3 mg), 2-(4-bromophenyl)-6-(trifluoromethyl)imidazo[1 ,2-a]pyridine (116 mg), ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4- carboxylate (100 mg) (a synthesis of which is described above as Intermediate 4), rac-1 ,2- dimethylcyclohexane diamine (20 mg) and potassium carbonate (99 mg) were dissolved in DMF (3 mL). The reaction mixture was heated at 16O0C in a reactivial overnight. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic was dried by passing through a hydrophobic frit and evaporated to dryness. The residue was loaded onto a 12 g silica SPE cartridge and was eluted with a gradient of 5% MeOH/DCM to give the title compound. MS calcd for (C3IH34F3N5O3+ H)+: 582 MS found (electrospray): (M+H)+ = 582
Intermediate 206
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(6- methylimidazo[1,2-a]pyridin-2-yl)phenyl]-1AV-pyrazole-4-carboxylate
Copper (I) iodide (13.3 mg), 2-(4-bromophenyl)-6-methylimidazo[1 ,2-a]pyridine (98 mg), ethyl 3-[[(fraA7S-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate (100 mg) (a synthesis of which is described above as Intermediate 4), rac-1 ,2- dimethylcyclohexane diamine (20 mg) and potassium carbonate (99 mg) were dissolved in DMF (3 ml_). The reaction mixture was heated at 16O0C in a reactivial overnight. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic was dried by passing through a hydrophobic frit and evaporated to dryness. The residue was loaded onto a 12 g silica SPE cartridge and was eluted with a gradient of 5% MeOH in DCM. The appropriate fractions were evaporated to dryness. The crude product was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C3IH37N5O3+ H)+: 528 MS found (electrospray): (M+H)+ = 528
Intermediate 207 2-(4-Bromo-2-furanyl)-1,3-benzoxazole
4-Bromo-2-furancarbonyl chloride (300 mg) and 2-aminophenol (156 mg) were dissolved in 1 ,4-dioxan (3 ml_) and heated in the microwave at 2100C for 45 min. This procedure was repeated 5 times. The reactions were combined and poured into 1 N NaOH solution which was extracted with DCM (x 3). The organic layer was dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C11H6NO2Br+ H)+: 264/266 MS found (electrospray): (M+H)+ = 264/266
Intermediate 208 Ethyl 1 -[5-(1 ,3-benzoxazol-2-yl)-3-furanyl]-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
Copper (I) iodide (34 mg), Intermediate 207 (215 mg), Intermediate 4 (238 mg), rac-1 ,2- dimethylcyclohexane diamine (20 mg) and potassium carbonate (245 mg) were dissolved in DMF (3 mL). The reaction mixture was heated at 16O0C in a reactivial for 18 h. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic was dried by passing through a hydrophobic frit and evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C28H32N4O5+ H)+: 505 MS found (electrospray): (M+H)+ = 505
Intermediate 209
Ethyl 1-(4-imidazo[1,2-a]pyridin-2-ylphenyl)-3-[(1-methylethyl)amino]-1H-pyrazole-4- carboxylate
Copper (I) iodide (143 mg), Intermediate 133 (917 mg), 2-(4-bromophenyl)imidazo[1 ,2- a]pyridine (940 mg), rac-1 ,2-dimethylcyclohexane diamine (213 mg) and potassium carbonate (1.1 g) were dissolved in DMF (28 mL). The reaction mixture was heated at 16O0C overnight. Copper (I) iodide (143 mg), Intermediate 133 (917 mg) and rac-1 ,2- dimethylcyclohexane diamine (213 mg) were added and the reaction was stirred for a further 24 h. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic was washed with brine and was dried by passing through a hydrophobic frit and evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C22H23N5O2+ H)+: 390 MS found (electrospray): (M+H)+ = 390
Intermediate 210
Ethyl 3-[[(2,4-dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-imidazo[1 ,2- a]pyridin-2-ylphenyl)-1H-pyrazole-4-carboxylate
Intermediate 209 (152 mg), 2,4-dichlorobenzoyl chloride (161 mg), and triphenylphosphine (252 mg) were dissolved in anhydrous DCM (5 mL) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 hr. The organic was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C29H25CI2N5O3+ H)+: 562/564/565 MS found (electrospray): (M+H)+ = 562/564/565
Intermediate 211
Ethyl 3-[[(2,4-dimethylphenyl)carbonyl](1 -methylethyl)amino]-1 -(phenylmethyl)-i H- pyrazole-4-carboxylate
Intermediate 2 (1.2 g), 2,4-dimethylbenzoyl chloride (0.8 g), and triphenylphosphine (2.2 g) were dissolved in anhydrous DCM (18 mL) and were heated at reflux for 88 h. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture. The organic was separated and extracted with DCM (x 3). The combined organics were dried by passing through a hydrophobic frit and were evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C25H29N3O3+ H)+: 420 MS found (electrospray): (M+H)+ = 420
Intermediate 212
Intermediate 211 (1.2 g) in ethanol (40 ml_) and cone. HCI (0.8 mL) was subjected to atmospheric pressure hydrogenation with 10% palladium on carbon (0.4 g wet) overnight. The reaction was filtered through a Celite pad, and the filter cake washed with ethanol. The combined organics were evaporated to dryness. The residue was partitioned between DCM and saturated sodium bicarbonate solution, passed through a hydrophobic frit and the organics evaporated to dryness to give the title compound. MS calcd for (C18H23N3O3+ H)+: 330 MS found (electrospray): (M+H)+ = 330
Intermediate 213
Ethyl 3-[[(2,4-dimethylphenyl)carbonyl](1 -methylethyl)amino]-1 -(4-furo[3,2-b]pyridin-2- ylphenyl)-1H-pyrazole-4-carboxylate
(90 mg) (a synthesis of which is described above as Intermediate 39), rac-1 ,2- dimethylcyclohexane diamine (20 mg) and potassium carbonate (99 mg) were dissolved in
DMF (3 mL). The reaction mixture was heated in a reactivial at 16O0C for 18 h. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic was dried by passing through a hydrophobic frit and evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound.
MS calcd for (C3IH30N4O4+ H)+: 523
MS found (electrospray): (M+H)+ = 523
Intermediate 214
Ethyl 3-[[(4-bromo-2-fluorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylate
Intermediate 134 (150 mg), 4-bromo-2-fluorobenzoyl chloride (227 mg), and triphenylphosphine (252 mg) were dissolved in anhydrous DCM (5 ml.) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give impure title compound. MS calcd for (C28H24BrFN6O3+ H)+: 591/593 MS found (electrospray): (M+H)+ = 591/593
Intermediate 215
Ethyl 3-[[(4-bromo-2-methylphenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1,5- a] pyri mid i n-2-yl phenyl) -1 H-pyrazole-4-carboxylate
Intermediate 134 (150 mg), 4-bromo-2-methylbenzoyl chloride (224 mg), and triphenylphosphine (252 mg) were dissolved in anhydrous DCM (5 mL) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give impure title compound. MS calcd for (C29H27BrN6O3+ H)+: 587/589 MS found (electrospray): (M+H)+ = 587/589
Intermediate 216
Ethyl 3-[[(4-chloro-2-fluorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylate
Intermediate 134 (150 mg), 4-chloro-2-fluorobenzoyl chloride (185 mg), and triphenylphosphine (252 mg) were dissolved in anhydrous DCM (5 mL) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic layer was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give impure title compound. MS calcd for (C28H24CIFN6O3+ H)+: 547/549 MS found (electrospray): (M+H)+ = 547/549
Intermediate 217
Ethyl 3-[[(2,4-dimethylphenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylate
Copper (I) iodide (13 mg), Intermediate 212 (100 mg), Intermediate 42 (90 mg), rac-1 ,2- dimethylcyclohexane diamine (20 mg) and potassium carbonate (99 mg) were dissolved in DMF (3 mL). The reaction mixture was heated in a Reactivial at 16O0C for 18 h. The reaction mixture was evaporated to dryness and the residue partitioned between water and DCM. The organic layer was dried by passing through a hydrophobic frit and evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H30N6O3 H)+: 523 MS found (electrospray): (M+H)+ = 523
Intermediate 218
Ethyl 3-[(1 -benzofuran-5-ylcarbonyl)(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylate
Intermediate 134 (121 mg), 1-benzofuran-5-carbonyl chloride (75 mg), and triphenylphosphine (162 mg) were dissolved in anhydrous DCM (6 ml_) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H26N6O4+ H)+: 535 MS found (electrospray): (M+H)+ = 535
Intermediate 219
3-[[(4-Bromo-2-chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-
Intermediate 134 (150 mg), 4-bromo-2-chlorobenzoyl chloride (244 mg), and triphenylphosphine (252 mg) were dissolved in anhydrous DCM (5 ml.) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic layer was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C28H24BrCIN6O3+ H)+: 607/609/610/611 MS found (electrospray): (M+H)+ = 607/609/610/611
Intermediate 220
Ethyl 3-[[(2-bromo-4-chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1W-pyrazole-4-carboxylate
Intermediate 134 (150 mg), 4-chloro-2-bromobenzoyl chloride (243 mg), and triphenylphosphine (252 mg) were dissolved in anhydrous DCM (5 mL) and were heated at 450C overnight. The reaction mixture was cooled and sodium bicarbonate solution was added to the reaction mixture which was stirred at room temperature for 1 h. The organic layer was separated and dried by passing through a hydrophobic frit and was evaporated to dryness. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give impure title compound. MS calcd for (C28H24BrCIN6O3+ H)+: 607/609/610/611 MS found (electrospray): (M+H)+ = 607/609/610/611
Intermediate 221
(1 E) -1 -(4-Bromophenyl)ethanone O-(2-nitrophenyl)oxime
A solution of potassium te/f-butoxide (5.1 mL, 1.0M solution in THF) was added in 1 mL portions to a solution of (1£)-1-(4-bromophenyl)ethanone oxime (1.0 g) in dry THF (5 mL) at O0C under nitrogen. Initially a precipitate formed but this dissolves in ca. 10 mins. The reaction was allowed to warm to room temperature and then 2-chloro-nitrobenzene (735 mg) was added portionwise. The reaction was heated to 5O0C under nitrogen for 3 h. On cooling the mixture was poured into water (40 mL) and extracted with EtOAc (3 x 30 mL), dried (Na2SO4) and evaporated to give a solid. This was applied in the minimum volume of DCM to a 2Og silica SPE cartridge. Elution with cyclohexane to cyclohexane/EtOAc (95:5) gave the title compound. MS calcd for (C14H11BrN2O3+ H)+: 336/337 MS found (electrospray): (M+H)+ = 336/337
(1 E)-1-(4-Bromophenyl)ethanone O-(2-nitrophenyl)oxime (500 mg) (a synthesis of which is described above as Intermediate 221 )as a suspension in 1M HCI/Acetic acid (5 mL) was stirred at room temperature for 24 h. The solvent was then evaporated. The compound was suspended in 1 M HCI/Acetic acid (5 mL) and heated to 8O0C under nitrogen for 48 h. On cooling, the solvent was evaporated. The residue was co-evaporated with toluene (3 x 5 mL), then diethyl ether (2 x 5 mL) to give a solid. This was applied in the minimum volume of DCM to a 50 g silica SPE cartridge. Elution with cyclohexane to cyclohexane /EtOAc (10%) gave the title compound. MS calcd for (C14H8BrNO3+ NH4)+: 336/337 MS found (electrospray): (M+NH4)+ = 336/337
Intermediate 223 [2-(4-Bromophenyl)-1-benzofuran-7-yl]amine
Tin (II) chloride (948 mg) was added in portions to a solution of 2-(4-bromophenyl)-7-nitro-1- benzofuran (519 mg) in THF (20 mL) and cone. HCI (10 drops) at room temperature under nitrogen. After 5 h, a further quantity of tin (II) chloride (948 mg) was added and the reaction left to stir for 48 h. The reaction was diluted with water (50 mL) and basified with 2N NaOH (ca. 8 mL). EtOAc (100 mL) was added and the phases filtered through celite. The layers were separated and the aqueous extracted further with EtOAc (2 x 50 mL). The combined organics were dried (Na2SO4) and evaporated to give a solid. This was applied in the minimum volume of DCM to a 20 g silica SPE cartridge. Elution with cyclohexane to cylohexane/EtOAc (gradient 9:1 to 8:2 to 7:3 to 6:4 to 1 :1 ) gave the title compound. MS calcd for (C14H10BrNO+ H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Intermediate 224 EthyH -[4-(4-amino-1 -benzofuran-2-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A solution of Intermediate 4 (159 mg), Intermediate 223 (143 mg), frans-(1 R, 2R)-N1N'- bismethyl-1 ,2-cyclohexanediamine (30 mg) and copper iodide (10 mg) with potassium carbonate (137 mg) in DMF (2 ml.) were heated in a Reactivial at 16O0C for 24 h. On cooling the solvent was evaporated and the residue partitioned between EtOAc (30 ml_) and water (10 ml_). The organic layer washed again with water (10 ml_), dried (Na2SO4) and evaporated. This was applied in the minimum volume of DCM to a 2Og silica SPE cartridge eluting with cyclohexane then cyclohexane/EtOAc (gradient 9:1 to 8:2 to 7:3 to 6:4 to 1 :1) gave the title compound. MS calcd for (C31 H36N4O4+ H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 225
Ethyl 3-[(cyclopentylacetyl)(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 W-pyrazole-4-carboxylate
A solution of Intermediate 134 (80 mg), cyclopentyl acetyl chloride (45 μL) and triphenylphosphine (78 mg) in dry DCM (3 ml.) was heated to 450C under nitrogen for 24 h.
On cooling the mixture was partitioned between 8% sodium bicarbonate solution and the layers separated using an aqueous extraction cartridge. The DCM layer was applied directly to a 1O g silica SPE cartridge. Elution with cyclohexane then cyclohexane/EtOAc (gradient
9:1 to 8:2 to 7:3 to 6:4 to 1 :1 ) gave the title compound.
MS calcd for (C28H32N6O3+ H)+: 503
MS found (electrospray): (M+H)+ = 503
Intermediate 226
Ethyl 3-[(cyclohexylacetyl)(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1W-pyrazole-4-carboxylate
A solution of Intermediate 134 (80 mg), cyclohexyl acetyl chloride (45 μl_) and triphenylphosphine (78 mg) in DCM (dry, 3 mL) was heated to 4O0C under nitrogen for 24 h. A further 45 μl_ of cyclohexyl acetyl chloride was added and heating continued for 6 h. On cooling the mixture was diluted with DCM (5 mL) and was washed with 8% sodium bicarbonate solution (2 x 5 mL), dried (aqueous extraction cartridge) and evaporated. This was applied in the minimum volume of DCM to a 20 g silica SPE cartridge. Elution with cyclohexane to cyclohexane/EtOAc (gradient 9:1 to 8:2 to 7:3 to 6:4 to 1 :1 ) gave the title compound. MS calcd for (C29H34N6O3+ H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 227 2-(4-Bromophenyl)-6-nitroimidazo[1,2-a]pyridine
A mixture of 2-amino-5-nitropyridine (270 mg) and 2,4-dibromoacetophenone (540 mg) in acetone (2.5 mL) was heated in a microwave at 13O0C for 35 min. The cooled reaction mixture was filtered and washed with acetone. The solid was collected and dried in vacuo to give the title compound.
MS calcd for (Ci3H8BrN3O2+H)+ : 318/320 MS found (electrospray): (M+H)+ = 318/320
Intermediate 228 2-(4-Bromophenyl)imidazo[1 ,2-a]pyridin-6-amine
A solution of Intermediate 227 (2.61 g) in THF (125 mL) was treated with tin (II) chloride (3.6 g) and cone. HCI (1 mL) and stirred at room temperature for 2 h. The reaction was evaporated to dryness and the residue treated with 2M sodium hydroxide solution and chloroform. The resulting emulsion was filtered and washed with chloroform. The aqueous phase was separated from the filtrate and extracted with more chloroform (x2) and the
combined organic phases were dried (MgSO4) and evaporated to dryness to give the title compound.
MS calcd for (Ci3H10BrN3+H)+ : 288/290
MS found (electrospray): (M+H)+ = 288/290
Intermediate 229
Ethyl 1 -[4-(6-aminoimidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (170 mg), Intermediate 228 (150 mg), copper (I) iodide (10 mg), frans-(1 Rl2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (30 mg) and potassium carbonate (140 mg) in DMF (3.5 ml_) was heated at 16O0C in a Reactivial for 3 h. The DMF was removed in vacuo and the residue partitioned between ethyl acetate and water. The aqueous phase was separated and extracted with more ethyl acetate (x2) and the combined organic phases washed with brine, dried (MgSO4) and concentrated. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane followed by a gradient of methanol in dichloromethane to give the title compound. MS calcd for (C30H36N6O^H)+ : 529 MS found (electrospray): (M+H)+ = 529
Intermediate 230 2-(4-Bromophenyl)-6-nitro-1,3-benzoxazole
A mixture of 2-amino-5-nitrophenol (278 mg) and 4-bromobenzoyl chloride (439 mg) in dry dioxane (2.5 mL) was heated in a microwave at 21O0C for 15 min. The cooled reaction mixture was added to 1M sodium hydroxide solution (50ml). The solid was filtered, washed with water and dried in vacuo to give the title compound. MS calcd for (C13H7BrN2O3 + H)+: 318/319 MS found (electrospray): (M+H)+ = 318/319
Intermediate 231
2-(4-Bromophenyl)-1,3-benzoxazol-6-amine
A solution of Intermediate 230 (2.5 g) in THF (125 ml.) was treated with tin (II) chloride (3.42 g) and concentrated hydrochloric acid (1 ml_) and stirred at room temperature for 5 h. Tin (II) chloride (3.42 g) and cone. HCI (1 ml.) was added and the mixture stirred at room temperature for 18 h. Tin (II) chloride (800 mg) was added and the mixture stirred at room temperature for 18 h. The reaction was evaporated to dryness and the residue treated with 2M sodium hydroxide solution and ethyl acetate. The resulting emulsion was filtered and washed with ethyl acetate. The aqueous phase was separated from the filtrate and extracted with more ethyl acetate (x2) and the combined organic phases were dried (MgSO4) and evaporated to dryness to give the title compound. MS calcd for (C13H9BrN2CHH)+ : 289/291 MS found (electrospray): (M+H)+ = 289/291
Intermediate 232
Ethyl 1 -[4-(6-amino-1 ,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (222 mg), Intermediate 231 (200 mg), copper (I) iodide (13 mg), trans-Cl R, 2R)-N, N'-bismethyl-1 ,2-cyclohexane diamine (39 mg) and potassium carbonate (190 mg) in DMF (4 mL) was heated at 16O0C in a Reactivial for 3 h. The DMF was removed in vacuo and the residue partitioned between ethyl acetate and water. The aqueous phase was separated and extracted with more ethyl acetate (x2) and the combined organic phases washed with brine, dried (MgSO4) and concentrated. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H35N5O4H-H)+ : 530 MS found (electrospray): (M+H)+ = 530
To a mixture of 5-chlorofuro[3,2-b]pyridine (1.04 g) and sodium iodide (5.00 g) in anhydrous acetonitrile (90 mL) was added acetyl chloride (2.39 mL) and the resultant yellow suspension was heated at reflux for 20 h. The reaction mixture was allowed to cool to room temperature and was then poured onto a stirred mixture of 0.5M aqueous potassium carbonate (400 mL), 1.0M aqueous sodium sulphite (100 mL) and diethyl ether (700 mL). The organic phase was separated off and was dried (MgSO4) and evaporated to give an approximately 1 :4 mixture (of product : starting material) containing the title compound. TLC (Silica gel 60 F254) eluted with 1 :1 EtOAc/ cyclohexane. Product Rf = 0.58, starting material Rf = 0.53.
Intermediate 234
Ethyl 1 -(4-f uroIS^-Jblpyridin-S-ylphenyO-S-tKfrans^-methylcyclohexyOcarbonyllti - methylethyl)amino]-1 H-pyrazole-4-carboxylate
Tetrakistriphenylphosphine palladium (0.043 g) was added to a mixture of Intermediate 180 (0.13 g; this material contains approximately 80% of 5-chlorofuro[3,2-ύ]pyridine) and Intermediate 181 (0.28 g) in dioxan (3 mL) and 2N sodium carbonate (0.75 mL) and the mixture heated at 1000C for 4.5 h. The reaction mixture was evaporated to dryness and the residue was partitioned between water and DCM. The organic phase was separated off using a hydrophobic frit and then evaporated to dryness. The residue was purified by was purified by chromatography over silica gel (ISCO Companion), eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C30H34N4O4 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 235 Ethyl 1 -(4-imidazo[1 ,2-a]pyridin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
To a mixture of Intermediate 180 (0.12 g) and 6-bromoimidazo[1 ,2-a]pyridine (0.11 g) in dioxan (2 ml.) containing 2N sodium carbonate (0.5 ml_) was added tetrakistriphenyl phosphine palladium (0) (0.036 g) and the resultant mixture was stirred at room temperature for 0.5 h and was then heated in a microwave oven at 1000C for 0.25 h. The mixture was allowed to cool to room temperature and was then partitioned between EtOAc and water. The organic phase was separated off, dried (Na2SO4) and evaporated. The residue was purified by chromatography over silica gel (ISCO companion) using gradient elution with EtOAc/cyclohexane give the title compound. MS calcd for (C30H35N5O3 +H)+: 514 MS found (electrospray): (M +H)+ = 514
Intermediate 236 2-(4-Bromophenyl)-1-benzofuran-4-amine
To a solution of Intermediate 178 (450 mg) and cone. HCI (10 drops) in dry THF (20 mL) was added tin (II) chloride (540 mg) and the reaction stirred at room temperature under nitrogen for 20 h. A further 2 equivalents of tin (II) chloride (540mg) was added and the reaction stirred at room temperature under nitrogen for 2 days. The reaction was evaporated to dryness, diluted with water, extracted twice with EtOAc, filtered through a Celite cartridge, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 2 x 10 g SCX-2 ion exchange cartridges, eluted with dioxan, MeOH, 10% ammonia in methanol and finally 10% triethylamine in MeOH. The basic fractions were combined and evaporated, and then this crude product was purified by 40 g silica ISCO Companion chromatography to give the title compound. MS calcd for (C14H10BrNO + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Dichloropalladium bistriphenylphosphine (0.63 g) and copper (I) iodide (0.17 g) were added to a degassed solution of 5-bromo-2-iodophenol (5.29 g), trimethylsilyl acetylene (3.73 mL) and triethylamine (9.91 mL) in THF (50 mL). The resultant mixture was stirred at 5O0C under a nitrogen atmosphere for 2 h. The reaction mixture was allowed to cool to RT and was then evaporated to dryness. The residue was partially partitioned between water and DCM. The organic phase was separated using a hydrophobic frit and the aqueous phase was washed with DCM. The organic phases were combined, concentrated by evaporation and the resultant slurry was diluted with acetonitrile and then filtered. The filtrate was evaporated to dryness and the residue was purified by chromatography over silica gel (Biotage) eluting with EtOAc/cyclohexane (1 :19). Appropriate fractions were combined and then evaporated to give the title compound. MS calcd for (C10H12BrNOSi + H)+: 270/272 MS found (electrospray): (M+H)+ = 270/272
Intermediate 238 6-Bromofuro[3,2-b]pyridine
To a stirred solution of Intermediate 237 (1.48 g) in ethanol (40 mL) was added potassium carbonate (0.65 g) and the resultant mixture was allowed to stir at RT for 3 days. The reaction mixture was evaporated to dryness and the residue was partitioned between DCM and water (some insoluble material). The organic phase was separated using a hydrophobic frit and was then evaporated. The residue was purified by ISCO Companion silica chromatography eluting with EtOAc/cyclohexane (gradient of 0-38%) to give the title compound.
MS calcd for (C7H4BrNO + H)+: 198/200 MS found (electrospray): (M+H)+ = 198/200
Intermediate 239
Ethyl 1 -(4-furo[3,2-b]pyridin-2-ylphenyl)-3-((1 -methylethyl){[trans-4- (trifluoromethyl)cyclohexyl]carbonyl}amino)-1H-pyrazole-4-carboxylate
A stirred mixture of Intermediate 182 (175 mg), Intermediate 39 (115 mg), potassium carbonate (72 mg), copper (I) iodide (9.5 mg) and trans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (25 mg) in DMF (3 ml_) was heated in a Reactivial at 1600C for 5 h. The solvent was evaporated then the residue was partitioned between DCM and water. The organic phase was separated by hydrophobic frit, evaporated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-100% ethyl acetate in cyclohexane. Product fractions were evaporated to give the title compound. MS calcd for (C30H3IF3N4O4 + H)+: 569 MS found (electrospray): (M+H)+ = 569
Intermediate 240 2-(4-Bromophenyl)-4-nitro-1,3-benzoxazole
A mixture of 2-amino-3-nitrophenol (278 mg) and 4-bromobenzoylchloride (439 mg) in dry dioxan (2 ml.) was stirred and heated at 21O0C in a microwave reactor for 15 minutes. The reaction was then slowly added to a vigorously stirred 1 N NaOH solution (50 ml_). The precipitate was collected by filtration, washed with water and dried (vacuum oven) to give the title compound.
MS calcd for (C13H7BrN2O3 +H)+ :319/321
MS found (electrospray): (M +H)+ = 319/321
Intermediate 241 2-(4-Bromophenyl)-1,3-benzoxazol-4-amine
A solution of Intermediate 240 (0.51 g) in THF (25 mL) was stirred at 210C and treated with tin (II) chloride (0.7 g) and concentrated HCI (11 drops). The mixture was stirred for 5. More
tin (II) chloride (0.7 g) and c. HCI (11 drops) was added and the reaction stirred for 3 days. The reaction was diluted with water, basified with 2N NaOH solution and extracted with EtOAc (x3). The combined extracts were washed with brine, dried (Na2SO4) and concentrated. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C13H9BrN2O +H)+ :289/291 MS found (electrospray): (M +H)+ = 289/291
Intermediate 242 Ethyl 1-[4-(4-amino-1,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 241 (339 mg), Intermediate 4 (377 mg), potassium carbonate (323 mg), copper (I) iodide (22 mg) and frans-(1 R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (66 mg) in dry DMF (5 ml_) was stirred and heated at 17O0C in a Reactivial for 3.5 h. The reaction was allowed to cool and concentrated. The residue was partitioned between EtOAc/water and the aqueous phase separated and extracted with more EtOAc (x2). The combined extracts were washed with water, brine, dried (Na2SO4) and concentrated. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C30H35N5O4 +H)+ : 530 MS found (electrospray): (M +H)+ = 530
Intermediate 243
Ethyl 3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1-[4-(1 ,8- naphthyridin-2-yl)phenyl]-1H-pyrazole-4-carboxylate
A Reactivial was charged with 2-(4-bromophenyl)-1 ,8-naphthyridine (211 mg), Intermediate 4 (237 mg), copper (I) iodide (14 mg), potassium carbonate (204 mg), frans-(1 R,2R)-N,N'- bismethyl-1 ,2-cyclohexane diamine (11 mg) and dry DMF (4 ml.) under nitrogen. The
reaction was heated at 1050C for 18 h. More copper (I) iodide (14 mg) was added and the reaction heated at 12O0C for 3 days. The reaction was allowed to cool and evaporated in vacuo. The residue was partitioned between EtOAc/water and the organic phase washed with more water. The combined aqueous phases were extracted with more EtOAc and the combined organics washed with brine, dried (Na2SO4) and concentrated. This was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C3i H35N5O3 +H)+ :526 MS found (electrospray): (M +H)+ = 526
Intermediate 244
Ethyl 1 -[4-(4-amino-1 -benzofuran-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 236 (90 mg), potassium carbonate (86 mg), copper iodide (5 mg) and Λ/,/V-dimethyl-1 ,2-cyclohexanediamine (20 mg) in DMF (2 mL) was heated in a microwave at 100° C for 15 minutes, then at 110° C for 20 minutes. The reaction mixture was evaporated to dryness, taken into EtOAc, washed twice with water, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 12 g silica ISCO Companion chromatography eluted with 0-100% EtOAc in cyclohexane to give the title compound. MS calcd for (C31H36N4O4 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 245 - see Intermediate 178
Intermediate246 6-Amino-2-(4-bromophenyl)-1-benzofuran
To a solution of Intermediate 245 (420 mg) in THF (25 mL) was added tin (II) chloride (1.73 g) and concentrated hydrochloric acid (5 drops). The mixture was stirred at room temperature for 3 h. A further quantity of tin (II) chloride (1.73 g) was added and stirring at room temperature was continued for 18 h. A third batch of tin (II) chloride (1.73 g) was added together with concentrated hydrochloric acid (5 drops) and the resulting mixture was
stirred at room temperature for a further 24 h. The mixture was diluted to 150 mL with water, basified with 2M sodium hydroxide solution, and extracted with ethyl acetate (2 x 150 mL). The biphasic mixture was filtered through a pad of Celite to remove insoluble tin residues. The organic layer was collected, washed with saturated brine (50 mL), dried (MgSO4) and evaporated to dryness to give the title compound. MS calcd for (C14H10BrNO + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Intermediate 247 Ethyl 1-[4-(6-amino-1-benzofuran-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1W-pyrazole-4-carboxylate
A mixture of Intermediate 246 (120 mg), Intermediate 4 (134 mg), potassium carbonate (115 mg), R,R'-bis(methylamino)cyclohexane (25 mg) and copper (I) iodide (8.5 mg) in DMF (1.7 mL) was stirred and irradiated in a CEM Explorer microwave reactor at 1600C for 1 h. A further quantity of copper (I) iodide (8.5 mg) was added and microwave heating was continued for 2 h. The resulting mixture was evaporated to dryness under reduced pressure and the residue was partitioned between 50 mL portions of water and ethyl acetate. The organic layer was collected, washed with brine and dried (MgSO4). The solvent was removed under reduced pressure and the residue was purified by ISCO Companion silica chromatography eluting with ethyl acetate/cyclohexane (a gradient of 0-100%) to give the title compound.
MS calcd for (C31H36N4O4 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 248 2-(4-Bromophenyl)imidazo[1 ,2-a]pyridin-8-amine
To a solution of 2-(4-bromophenyl)-8-nitroimidazo[1 ,2-a]pyridine (128m mg) and cone. HCI (10 drops) in dry THF (20 ml_) was added tin (II) chloride (152 mg) and the reaction stirred at room temperature under nitrogen for 18 h. A further addition of tin (II) chloride (152 mg) was made and the reaction stirred at room temperature under nitrogen for a further 4 days. The reaction was evaporated to dryness, water was added (150 mL), extracted with EtOAc (2 x 100 mL), dried (MgSO4), filtered and evaporated to dryness to give a solid. This was purified by 5 g SCX-2 ion exchange cartridge eluted with MeOH, 10% 2M ammonia in MeOH and finally 10% triethylamine in MeOH. The final fraction was evaporated to give the title compound. MS caled for (C13H10BrN3 + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Intermediate 249
Ethyl 1 -[4-(8-aminoimidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (117 mg), Intermediate 248 (105 mg), potassium carbonate (100 mg), copper (I) iodide (6 mg) and Λ/./V-dimethyl-i ,2-cyclohexanediamine (22 mg) in DMF (2 mL) was heated in a microwave at 100° C for 15 minutes. The reaction was evaporated to dryness, taken into water, extracted twice with EtOAc, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 40 g silica ISCO Companion chromatography eluted with 0-60% EtOAc in cyclohexane to give the title compound. MS calcd for (C30H36N6O3 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 250
6-(4-Bromophenyl)thieno[3,2-d]pyrimidin-4-amine
A mixture of 6-(4-bromophenyl)-4-chlorothieno[3,2-c(]pyrimidine (150 mg) and 2N ammonia (2.2 mL) in THF (3 mL) was stirred at room temperature in a Reactivial for 20 h. 2N ammonia (2 mL) was added, and the mixture was stirred in the Reactivial at 600C for 70 h. The THF was evaporated under vacuum and the residue partitioned between DCM and 2N HCI solution. A precipitate appeared between the phases; it was isolated by filtration and dried to give the title compound.
MS calcd for (C12H8BrN3S + H)+: 306/308 MS found (electrospray): (M+H)+ = 306/308
Intermediate 251
Ethyl 1-[4-(4-aminothieno[3,2-d]pyrimidin-6-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1W-pyrazole-4-carboxylate
A mixture of Intermediate 4 (134 mg), Intermediate 250 (128 mg), copper (I) iodide (8 mg), trans-(\ R, 2R)-N, N'-bismethyl-1 ,2-cyclohexane diamine (119 mg) and potassium carbonate (115 mg) in DMF (2 ml_) was heated at 16O0C in a Reactivial for 6 h under an atmosphere of nitrogen. The DMF was evaporated under vacuum and the residue partitioned between
DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated.
The residue was purified by ISCO Companion silica chromatography, eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C29H34N6O3S + H)+: 547
MS found (electrospray): (M+H)+ = 547
Intermediate 252 6-(4-Bromophenyl)-Λ/-methylthieno[3,2-d]pyrimidin-4-amine
A mixture of 6-(4-bromophenyl)-4-chlorothieno[3,2-c/]pyrimidine (150 mg) and 2N methylamine (2.3mL) in THF (3 ml.) was stirred at room temperature in a Reactivial for 20 h. The THF was evaporated under vacuum and the residue partitioned between DCM and 2N HCI solution. A precipitate appeared between the phases; it was filtered and dried to give the title compound.
MS calcd for (C13H10BrN3S + H)+: 319/321 MS found (electrospray): (M+H)+ = 319/321
Intermediate 253 Ethyl 1 -{4-[4-(methylamino)thieno[3,2-d]pyrimidin-6-yl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (179 mg), Intermediate 252 (154 mg), copper (I) iodide (11 mg), trans-(\ R, 2R)-N, N'-bismethyl-1 ,2-cyclohexane diamine (159 mg) and potassium carbonate (154 mg) in DMF (2.5 ml.) was heated at 16O0C in a Reactivial for 6 h under an atmosphere of nitrogen. The DMF was evaporated under vacuum and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion silica chromatography, eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C30H36N6O3S + H)+: 561 MS found (electrospray): (M+H)+ = 561
Intermediate 254
Ethyl 1 -(phenylmethyl)-3-(tetrahydro-2H-pyran-4-yl{[frans-4-
(trifluoromethyl)cyclohexyl]ca -4-carboxylate
Intermediate 9 (1.401g) was dissolved in dry DCM (6 ml.) and treated with triphenylphosphine (2.228 g) and Intermediate 176 (1.365 g). The reaction was heated at reflux temperature for 24 h, then the mixture was basified with saturated sodium bicarbonate solution. The mixture was extracted with DCM and the organic phase was concentrated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C26H32F3N3O4+ H)+: 508 MS found (electrospray): (M+H)+ = 508
Intermediate 255
Ethyl 3-(tetrahydro-2H-pyran-4-yl{[fraπs-4- (trifluoromethyl)cyclohexyl]carbonyl}amino)-1H-pyrazole-4-carboxylate
A solution of Intermediate 254 (1.71 g) in ethanol (60 ml.) and cone. HCI (0.7 ml_) was stirred under an atmosphere of hydrogen with 10% palladium on carbon catalyst (0.36 g) overnight. The mixture was filtered through a Celite pad, the pad washed with ethanol and the combined organics evaporated. The residue was partitioned between DCM and saturated sodium bicarbonate solution, passed through a hydrophobic frit and the organic layer evaporated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of EtOAc in cyclohexane to give the title compound. MS calcd for (C19H26F3N3O4 + H)+: 418 MS found (electrospray): (M+H)+ = 418
Intermediate 256
Ethyl 1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[frans-4-
(trifluoromethyl)cyclohexyl] -carboxylate
A mixture of Intermediate 255 (150 mg), Intermediate 42 (84 mg), copper (I) iodide (15 mg), frans-(1 R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (42 mg) and potassium carbonate (60 mg) in DMF (2.5 ml_) was heated at 16O0C in a microwave reactor for 1 h. The DMF was evaporated under vacuum and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C31H33F3N6O4 + H)+: 611 MS found (electrospray): (M+H)+ = 611
Intermediate 257 Ethyl 1 -(4-furo[3,2-ib]pyridin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[frans-4- (trifluoromethyl)cyclohexyl] -carboxylate
A mixture of Intermediate 255 (150 mg), Intermediate 39 (84 mg), copper (I) iodide (15 mg), trans-(\ R,2R)-N,N'-bismethyl-1 ,2-cyclohexane diamine (42 mg) and potassium carbonate (60 mg) in DMF (2.5 ml.) was heated at 1600C in a microwave reactor for 2.5 h. The DMF was evaporated under vacuum then the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound.
MS calcd for (C32H33F3N4O5 + H)+: 611 MS found (electrospray): (M+H)+ = 611
Intermediate 258
Ethyl 1 -(4-imidazo[1 ,2-b]pyridazin-2-ylphenyl)-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A stirred mixture of Intermediate 4 (200 mg), 2-(4-bromophenyl)imidazo[1 ,2-b]pyridazine (112 mg), potassium carbonate (95 mg), copper (I) iodide (23 mg) and trans-(1 R,2R)-N,N'- bismethyl-1 ,2- cyclohexane diamine (35 mg) in DMF (3.5 mL) was heated in a microwave at 1600C for 1.5 h. More copper (I) iodide (23 mg) and more frans-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (35 mg) were added to the mixture and it was heated in a microwave at 1600C for 1.25 h. The mixture was evaporated under vacuum and the residue was partitioned between DCM and water. The organic phase was separated, evaporated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-100% ethyl acetate in cyclohexane. Product fractions were combined, evaporated and further purified by ISCO Companion silica chromatography eluting with a gradient of 0-4% MeOH in DCM to give the title compound. MS calcd for (C29H34N6O3 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 259
Ethyl 1 -(4-imidazo[1 ^-alpyrazin^-ylphenyO-S-Mfraπs^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 W-pyrazole-4-carboxylate
A stirred mixture of Intermediate 4 (115 mg), 2-(4-bromophenyl)imidazo[1 ,2-a]pyrazine (64 mg), potassium carbonate (55 mg), copper (I) iodide (14 mg) and frans-(1 R,2R)-N,N'- bismethyl-1 ,2- cyclohexane diamine (22 mg) in DMF (2 ml.) was heated in a microwave at 1600C for 1.5 h. The mixture was evaporated under vacuum and the residue was partitioned between DCM and water. The organic phase was separated, evaporated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-100% ethyl acetate in cyclohexane. Product fractions were combined and evaporated to give the title compound. MS calcd for (C29H34N6O3 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Intermediate 260 [2-(4-Bromophenyl)imidazo[1,
(2-Amino-3-pyridinyl)methanol (134 mg) and 2-bromo-1-(4-bromophenyl)ethanone (300 mg) were heated at reflux in ethanol (3 mL) overnight. A white solid was filtered off and dried under vacuum to give the title compound. MS calcd for (Ci4H11BrN2O + H)+: 303/305 MS found (electrospray): (M+H)+ = 303/305
Intermediate 261
Ethyl 1 -{4-[8-(hydroxymethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-3-[[(trans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A stirred mixture of Intermediate 4 (313 mg), Intermediate 260 (295 mg), potassium carbonate (160 mg), copper (I) iodide (36 mg) and frans-(1 R,2R)-N,N'-bismethyl-1,2- cyclohexane diamine (97 mg) in DMF (5 mL) was heated in a microwave at 1300C for 20 minutes and then at 1400C for a further 20 minutes. More copper (I) iodide (29 mg) and more frans-(1 R, 2R)-N, N'-bismethyl-1 ,2- cyclohexane diamine (53 mg) were added to the mixture and it was heated in a microwave at 16O0C for 20 minutes. The mixture was evaporated under vacuum, then the residue was partitioned between DCM and water. The organic phase was separated, evaporated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-80% ethyl acetate in cyclohexane. Product fractions were combined and evaporated to give the title compound.
MS calcd for (C3IH37N5O4 + H)+: 544 MS found (electrospray): (M+H)+ = 544
Intermediate 262
Ethyl 1 -{4-[8-(aminomethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-3-[[(trans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
To a solution of Intermediate 261 (171 mg) in dry THF (1.5 mL) at 00C were added triethylamine (66 uL) and tosyl chloride (67 mg). After 0.5 h more triethylamine (66 uL) and tosyl chloride (67 mg) were added. The mixture was warmed to room temperature, DMAP (3 mg) was added and stirring continued overnight. The mixture was partitioned between DCM and water. The organic phase was separated, evaporated and dissolved in a 0.5 M solution of ammonia in dioxane (6 mL). After 3 h, a 7 M solution of ammonia in methanol (2 mL) was added and the solution was stirred overnight. The mixture was evaporated to dryness, redissolved in a 7 M solution of ammonia in methanol (5 mL) and heated at 600C in a Reactivial for 5 h. The solvent was evaporated and the crude product was purified by SCX SPE ion exchange chromatography, then further purified by ISCO Companion silica chromatography eluting with a gradient of 0-40% methanol in DCM. Product fractions were evaporated to give the title compound. MS calcd for (C3IH38N6O3 + H)+: 543 MS found (electrospray): (M+H)+ = 543
Intermediate 263
Ethyl 1 -[4-(7-aminopyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-((1 -methylethyl){[trans-4-
(trifluoromethyl)cycloh rboxylate
A stirred mixture of Intermediate 182 (190 mg), Intermediate 99 (146 mg), potassium carbonate (80 mg), copper (I) iodide (9.5 mg) and fraA?s-(1 R,2R)-N,N'-bismethyl-1 ,2- cyclohexane diamine (28 mg) in DMF (3.5 mL) was heated in a microwave at 1000C for 20 minutes and then at 1200C for a further 25 minutes. The solvent was evaporated under
vacuum and the residue was partitioned between DCM and water. The organic phase was separated, evaporated under vacuum and purified by ISCO Companion silica chromatography eluting with a gradient of 0-100% ethyl acetate in cyclohexane. Product fractions were evaporated to give the title compound. MS calcd for (C29H32F3N7O3 + H)+: 584 MS found (electrospray): (M+H)+ = 584
Intermediate 264
Ethyl S-tttfrans^-methylcyclohexyOcarbonylKtetrahydro^H-pyran^-yOaminol-i-^-tθ- quinazolinyl)phenyl]-1 H-pyrazole-4-carboxylate
Tetrakistriphenylphosphine palladium (0.036 g) was added to a mixture of 6- bromoquinazoline (0.12 g) and Intermediate 181 (0.28 g) in DME (20 ml_) and 2N sodium carbonate (10 ml_). This mixture was heated to 1000C for 23 h. The reaction mixture was allowed to cool to room temperature and was then poured onto a mixture of EtOAc and saturated aqueous sodium bicarbonate. The aqueous phase was separated off and was extracted twice with EtOAc. The extracts and organic phase were combined, dried and evaporated to give the title compound. MS calcd for (C33H37N5O4 + H)+: 568 MS found (electrospray): (M+H)+ = 568
Intermediate 265
Ethyl 1 -(4-imidazo[1 ,2-a]pyridin-2-ylphenyl)-3-((1 -methylethyl){[trans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1H-pyrazole-4-carboxylate
A stirred mixture of Intermediate 182 (150 mg), 2-(4-bromophenyl)imidazo[1 ,2-a]pyridine (92 mg), potassium carbonate (60 mg), copper (I) iodide (15 mg) and trans-{\ R,2R)-N, N1- bismethyl-1 ,2- cyclohexane diamine (42 mg) in DMF (2.5 ml_) was heated in a microwave at 16O0C for 2.5 h. The solvent was evaporated under vacuum and the residue was partitioned between DCM and water. The organic phase was separated and purified by ISCO
Companion silica chromatography eluting with a gradient of 0-70% ethyl acetate in cyclohexane. Product fractions were evaporated to give the title compound. MS calcd for (C30H32F3N5O3 + H)+: 568 MS found (electrospray): (M+H)+ = 568
Intermediate 266
Ethyl 1 -(4-imidazo[2,1 -b][1 ,3]thiazol-6-ylphenyl)-3-((1 -methylethyl){[trans-4-
(trifluoromethyl)cyclohex rboxylate
A stirred mixture of Intermediate 182 (150 mg), 6-(4-bromophenyl)imidazo[2,1-b][1 ,3]thiazole (95 mg), potassium carbonate (60 mg), copper (I) iodide (15 mg) and trans-( 1 R, 2R)-N, N'- bismethyl-1 ,2-cyclohexane diamine (42 mg) in DMF (2.5 mL) was heated by microwave at 1600C for 2.5 h. The solvent was evaporated under vacuum then the residue was partitioned between DCM and water. The organic phase was separated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-70% ethyl acetate in cyclohexane. Product fractions were evaporated to give the title compound. MS calcd for (C28H30F3N5O3S + H)+: 574 MS found (electrospray): (M+H)+ = 574
Intermediate 267
Ethyl 1 -[4-(5-amino-6-chloro-3-pyridinyl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 180 (0.10 g), 2-chloro-3-amino-5-bromopyridine (30 mg), sodium carbonate (60 mg) and tetrakis(triphenylphosphine)palladium (20 mg) in dioxan (1.5 mL) and water (0.5 mL) was heated in a microwave at 110° C for 20 minutes. The reaction was evaporated to dryness, taken into EtOAc, washed with water, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 12 g silica ISCO Companion chromatography eluted with 0-100% EtOAc in cyclohexane to give the title compound. MS calcd for (C28H34CIN5O3 + H)+: 524/526 MS found (electrospray): (M+H)+ = 524/526
Intermediate 268
Ethyl 3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(3- quinolinyl)phenyl]-1/7-pyrazole-4-carboxylate
Intermediate 180 (0.10 g), 3-bromoquinoline (29 uL), sodium carbonate (0.20 g) and tetrakis(triphenylphosphine)palladium (23 mg) in 1 ,2-dimethoxyethane (15 ml_) and water (1 ml.) were heated at 90° C for 3 hours. The reaction was taken into DCM, washed with sodium bicarbonate solution, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 5 g silica SPE cartridge eluted with a gradient of cyclohexane to DCM, followed by a gradient of DCM to EtOAc to give the title compound. MS calcd for (C32H36N4O3 + H)+: 525 MS found (electrospray): (M+H)+ = 525
Intermediate 269
Ethyl 3-[[(frans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6- quinoxalinyl)phenyl]-1H-pyrazole-4-carboxylate
Intermediate 180 (0.10 g), 6-bromoquinoxaline (44 mg), sodium carbonate (60 mg) and tetrakis(triphenylphosphine)palladium (23 mg) in 1 ,4-dioxane (1.5 ml.) and water (0.5 mL) were heated in a microwave at 100° C for 15 minutes. The reaction was evaporated to dryness, taken into DCM, washed with sodium bicarbonate solution, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 5 g silica SPE cartridge eluted with a gradient of DCM to EtOAc to give the title compound. MS calcd for (C3i H35N5O3 + H)+: 526 MS found (electrospray): (M+H)+ = 526
Intermediate 270
Ethyl 1 -[4-(4-amino-6-quinolinyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
Intermediate 180 (0.10 g), 4-amino-6-bromoquinoline (47 mg), sodium carbonate (60 mg) and tetrakis(triphenylphosphine)palladium (23 mg) in 1 ,4-dioxan (1.5 ml_) and water (0.5 ml.) were heated in a microwave at 100° C for 15 minutes. The reaction was evaporated to dryness, taken into DCM, washed with sodium bicarbonate solution, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 5 g silica SPE cartridge eluted with a gradient of DCM to EtOAc to acetonitrile to acetone to MeOH to give the title compound. MS calcd for (C32H37N5O3 + H)+: 540 MS found (electrospray): (M+H)+ = 540
Intermediate 271
Ethyl 1 -[4-(6-isoquinolinyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylate
Intermediate 180 (0.10 g), 6-bromoisoquinoline (44 mg), sodium carbonate (60 mg) and tetrakis(triphenylphosphine)palladium (23 mg) in 1,4-dioxan (1.5 ml.) and water (0.5 mL) were heated in a microwave at 100° C for 15 minutes. The reaction was evaporated to dryness, taken into DCM, washed with sodium bicarbonate solution, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 5 g silica SPE cartridge eluted with a gradient of DCM to EtOAc to give the title compound. MS calcd for (C32H36N4O3 + H)+: 525 MS found (electrospray): (M+H)+ = 525
Intermediate 272
(4-{4-[(Ethyloxy)carbonyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazol-1 -yl}phenyl)boronic acid
A solution of Intermediate 180 (0.50 g), sodium periodate (0.61 g) and ammonium acetate (0.22 g) in acetone (40 ml_) and water (40 mL) was stirred at room temperature for 20 h. The reaction mixture was filtered through Celite, washed with acetone and evaporated. The organics were extracted twice with EtOAc, dried using a hydrophobic frit and evaporated to dryness to give the title compound. MS calcd for (C23H32BN3O5 + H)+: 442 MS found (electrospray): (M+H)+ = 442
Intermediate 273
Ethyl S-IKfra/js^-methylcyclohexyOcarbonyllti-methylethyOaminol-i-^-tθ- quinolinyl)phenyl]-1H-pyrazole-4-carboxylate
Intermediate 272 (0.10 g), 6-bromoquinoline (39 mg), sodium carbonate (60 mg) and tetrakis(triphenylphosphine)palladium (20 mg) in 1 ,4-dioxan (1.5 mL) and water (0.5 mL) were heated in a microwave at 110° C for 20 minutes, then at 160° C for 20 minutes. The reaction was evaporated to dryness, taken into DCM, washed with sodium bicarbonate solution, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 12 g silica ISCO Companion chromatography eluted with 0-100% EtOAc in cyclohexane to give the title compound. MS calcd for (C32H36N4O3 + H)+: 525 MS found (electrospray): (M+H)+ = 525
Intermediate 274 Ethyl 3-[[(trans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(7- methylfuro[3,2-b]pyridin-2-yl)phenyl]-1H-pyrazole-4-carboxylate
To a stirred solution of 2-chloro-3-hydroxy-4-picoline (250 mg) in THF (10 mL) was added a 4 M solution of HCI in dioxan (2 mL). The solvent was evaporated and the solid was stirred for 2 days with sodium iodide (1 g) in dry acetonitrile (7.5 mL) at 900C. The mixture was evaporated and partitioned between EtOAc and an aqueous solution of 10% potassium carbonate and 5% sodium bisulfite. The organic phase was separated, dried, evaporated then purified by SPE silica chromatography eluting with cyclohexane/ethyl acetate (5:1 ). Product fractions were combined and evaporated to give an impure solid containing some 2- iodo-3-hydroxy-4-picoline. This solid was dissolved in triethylamine (2 mL) with Intermediate 45 (95 mg), copper (I) iodide (10 mg) and Pd(PPh3J2CI2 (16 mg) then the mixture was stirred at 950C for 2 h. The solvent was evaporated then the residue was partitioned between water and DCM. The organic phase was separated, evaporated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-65% ethyl acetate in cyclohexane. Product fractions were evaporated to give the title compound. MS calcd for (C31H36N4O4 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Intermediate 275
Ethyl 1 -(5-imidazo[1 ,2-a]pyrimidin-2-yl-2-thienyl)-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), 2-(5-bromo-2-thienyl)imidazo[1,2-a]pyrimidine (90 mg), copper (I) iodide (14 mg), fraA7S-N,N'-bismethyl-1 ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 mL) was heated at 16O0C in a Reactivial for 18 h.
The DMF was removed in vacuo and the residue partitioned between chloroform and water. The chloroform layer was separated using a hydrophobic frit and concentrated. This residue was purified by SPE silica chromatography eluting with a stepped gradient of methanol in ethyl acetate (0:100 to 50:50) to give the title compound.
MS calcd for (C27H32N6O3S + H)+: 521
MS found (electrospray): (M+H)+ = 521 Intermediate 276
A mixture of dichloropalladium bistriphenylphosphine (0.48 g), copper (I) iodide (0.17 g), 3- iodo-1,2-benzenediol (3.10 g), 1 -bromo-4-ethynylbenzene (3.55 g), triethylamine (7.35 mL) and anhydrous THF (42 mL) was degassed in a stream of nitrogen for 5 min and was then heated to 500C under nitrogen for 3.5 h. The reaction was allowed to cool to room temperature and was then evaporated to dryness. The residue was purified by Biotage chromatography over silica gel eluting with a gradient of 5 to 40 % EtOAc/cyclohexane. Appropriate fractions were combined and then evaporated to give the title compound. MS calcd for (C14H9BrO2 -H)+: 287/289 MS found (electrospray): (M -H)+ = 287/289
Intermediate 277 2-(4-Bromophenyl)-1-benzofuran-7-ol
A stirred partial solution of Intermediate 276 (3.49 g) in a mixture of acetonitrile (100 mL) and saturated aqueous sodium bicarbonate (15 mL) was heated at 700C with stirring for 26 h. The reaction mixture was allowed to cool to room temperature, was evaporated to near dryness and then water and DCM were added with stirring. The aqueous layer was adjusted to pH1 using 2N HCI and the organic phase was separated off using a hydrophobic frit. The aqueous phase was extracted with DCM (x 10). The organic phase and extracts were combined and then evaporated. The residue was purified by Biotage chromatography over silica gel eluting with a gradient of EtOAc/cyclohexane. Appropriate fractions were combined and then evaporated to give the title compound MS calcd for (C14H9BrO2 -H)+: 287/289 MS found (electrospray): (M -H)+ = 287/289
Intermediate 278
Ethyl 1 -[4-(7-hydroxy-1 -benzofuran-2-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
To a stirred solution of Intermediate 4 (0.15 g), Intermediate 277 (0.15 g), copper (I) iodide (0.037 g), (1 R,2R)-Λ/,^dimethyl-1 ,2-cyclohexanediamine (0.041 g) and potassium carbonate (0.15 g) was heated at 1600C in DMF (5 ml.) under nitrogen for 20 h. The reaction mixture was evaporated to dryness and the residue was partitioned between DCM and 0.5M HCI. The organic phase was separated using a hydrophobic frit and was then evaporated. The residue was purified by Biotage chromatography over silica gel eluting with a gradient of 5 to 50% EtOAc/cyclohexane. Appropriate fractions were combined and then evaporated to give the title compound MS calcd for (C3IH35N3O5 -H)+: 530 MS found (electrospray): (M -H)+ = 530
Intermediate 279
1-(4-Bromophenyl)ethanoneoxime (8:1 mixture of E and Z isomers)
A mixture of 1-(4-bromophenyl)ethanone (5.0 g) and hydroxylamine hydrochloride (1.75 g) in pyridine (10 ml_) was stirred at RT for 3 h. The mixture was poured into 2M HCI (250 mL) and was extracted with ethyl acetate (2 x 150 mL). The combined organic extracts were washed with water and brine and dried (MgSO4). The solvent was removed in vacuo to give title compound.
1H NMR (d6-DMSO) δ 11.34 (major) + 10.79 (minor) (both s, total =1 H), 7.59 (m, 4H), 3.34 (3H, 2 x s)
Intermediate 280
1-(4-Bromophenyl)ethanone mixture of E and Z isomers)
A solution of Intermediate 279 in THF (10 mL) was stirred at O0C under nitrogen. 1.0M potassium tert-butoxide in THF (10 mL) was added and the resulting mixture was stirred
under nitrogen with ice-bath cooling for 1.5 h. A solution of 1-fluoro-4-nitrobenzene (1.41 g) in THF (5 mL) was added and the mixture was stirred at 500C for 4 h. The mixture was cooled to room temperature and diluted to 250 mL with water. The resulting mixture was extracted with ethyl acetate (2 x 250 mL). The organic extracts were combined, washed with brine and dried (MgSO4). The solvent was removed in vacuo to give title compound.
1H NMR (CDCI3) (major isomer) δ 8.29 (d, 2H), 7.83 (d, 2H), 7.72 (d, 2H), 7.53 (d, 2H), 3.34 (s, 3H).
Intermediate 281 2-(4-Bromophenyl)-5-nitro-1 -benzof uran
A mixture of Intermediate 280 (2.8 g), formic acid (100 mL) and concentrated hydrochloric acid (15 mL) was stirred and heated at 800C for overnight. The mixture was cooled to room temperature and the solid collected by filtration, washed with water and dried under vacuum at 400C overnight to give the title compound.
1H NMR (d6-DMSO) δ 8.64 (d, 1H), 8.24 (dd, 1H), 7.93 (d, 2H), 7.89 (d, 1 H), 7.76 (d, 2H), 7.72 (s, 1 H)
Intermediate 282 2-(4-Bromo-2-chlorophenyl)py mine
Intermediate 195 (1.38 g) was dissolved in acetic acid (20 mL) and was treated with 3- dimethylaminoacrylonitrile (0.433 mL). The mixture was heated at 11O0C for 1.5 h. The acetic acid was removed in vacuo and the residue was triturated with DCM. The solid was filtered off to give the title compound.
MS calcd for (Ci2H8BrCIN4 + H)+: 322/324/326 MS found (electrospray): (M+H)+ = 322/324/326
Intermediate 283 Ethyl 1 -[4-(7-aminopyrazolo[1 ,5-a]pyrimidin-2-yl)-3-chlorophenyl]-3-[[(fra/is-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1H-pyrazole-4-carboxylate
A mixture of Intermediate 4 (100 mg), Intermediate 282 (111 mg), copper (I) iodide (14 mg), trans-N, N'-bismethyl-i ,2- cyclohexane diamine (21 mg) and potassium carbonate (103 mg) in DMF (2.5 ml.) was heated at 16O0C in a Reactivial for 16 h. The DMF was removed in vacuo and the residue partitioned between chloroform and water. The chloroform layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion silica chromatography eluting with a gradient of ethyl acetate in cyclohexane to give the title compound. MS calcd for (C29H34CIN7O3 + H)+: 562/564 MS found (electrospray): (M+H)+ = 562/564
Intermediate 284 4-Bromo-Λ/-(4-hydroxy-3-pyridinyl)benzamide
To 4-bromobenzoyl chloride (749 mg) and 3-amino-4-hydroxypyridine hydrochloride (500 mg) was added pyridine (7.5 mL) and the suspension was stirred at RT overnight. Ice/water (10 mL) was added and the precipitate was filtered off and was washed with water. The solid was partitioned between DCM and 1 M HCI solution. The mixture was filtered off and dried in vacuum oven to give title compound. 1H NMR (d6-DMSO) δ 10.01 (s, 1 H), 8.98 (d, 1 H), 8.26 (dd, 1 H), 7.92 (dt, 2H), 7.77 (dt, 2H), 7.16 (d, 1 H). One exchangeable proton not seen.
Intermediate 285 2-(4-Bromophenyl)[1,3]oxazolo[4,5-c]pyridine
To intermediate 284 (331 mg) was added phosphorus (V) oxychloride (3 mL) and the mixture was heated at 1000C for 4 h. The reaction was cooled to RT and was evaporated in vacuo and was co-evaporated with toluene (x 4). The residue was dried in a vacuum dessicator over sodium hydroxide pellets overnight. Ethyl acetate was added and the slurry was added to stirred saturated sodium bicarbonate solution. The mixture was diluted with a little water and ethyl acetate until a clear bi-phasic mixture was obtained and the organics were separated and evaporated in vacuo. The residue was purified by ISCO Companion silica
chromatography eluting with a gradient 5% EtOAc/cyclohexane to 100% EtOAc to give the title compound.
MS calcd for (C12H7BrN2O + H)+: 274/276 MS found (electrospray): (M+H)+ = 274/276
Intermediate 286 2-(4-Bromophenyl)-1-benzofuran-5-amine
To a solution of Intermediate 281 (1.0 g) in THF (50 ml_) was added tin (II) chloride (4.13 g) and concentrated hydrochloric acid (20 drops). The mixture was stirred at room temperature overnight. A further quantity of tin (II) chloride (4.13 g) was added and stirring at room temperature was continued for 24 h. The mixture was diluted to 500 ml. with water, basified with 2M sodium hydroxide solution, and extracted with ethyl acetate (2 x 400 ml_). The biphasic mixture was filtered through a pad of Celite to remove insoluble tin residues. The organic layer was collected, washed with saturated brine (150 ml_), dried (MgSO4) and evaporated to dryness to give the title compound. MS calcd for (C14H10BrNO + H)+: 288/290 MS found (electrospray): (M+H)+ = 288/290
Example 1 i-^IH-lndol^-ylJphenyll-S-lKfrans^-methylcyclohexyOcarbonylKI- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 8 (210 mg) was dissolved in ethanol (20 ml_) and treated with sodium ethoxide in ethanol (21 wt.%, 3 ml_). The reaction was stirred at room temperature for 2 days and then heated at 7O0C for 2 h. Water (4 ml_) and THF (4 mL) were added and the reaction stirred at room temperature for 4 days. The organic solvents were removed in vacuo and the residue acidified with 2N HCI solution. The mixture was extracted with ethyl acetate and the organic extract washed with brine, dried (Na2SO4) and concentrated. The crude product was purified by MDAP HPLC to give the title compound. MS calcd for (C29H32N4O3 + H)+: 485 MS found (electrospray): (M+H)+ = 485
Example 2
1 -(4-lmidazo[1 ^-alpyridin^-ylphenyO-S-IKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 19 (118 mg) was dissolved in THF (2 mL) and ethanol (2 ml_). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 2 days. The ethanol and THF were removed in vacuo, the residue diluted with water and acidified to pH 4 with aqueous 2N HCI solution. The mixture was extracted with ethyl acetate, the organic extract dried (Na2SO4) and concentrated to give the title compound. MS calcd for (C28H3IN5O3 + H)+: 486 MS found (electrospray): (M+H)+ = 486
1H NMR (de-DMSO) δ 12.78 (1 H, br. s), 9.15 (1H, s), 8.55 (1 H, d), 8.49 (1 H, s), 8.13 (2H, d), 8.00 (2H, d), 7.60 (1 H, d), 7.30-7.24 (1 H, m), 6.95-6.89 (1 H, m), 4.75 (1 H, sept.), 2.01-1.89 (1H, m), 1.73-1.41 (5H, m) 1.31-1.10 (5H, m), 0.89 (3H, br. d) 0.74 (3H, d), 0.70-0.48 (2H, m)
Example 3
1 -[4-(1 H-Benzimidazol-2-yl)phenyl]-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 20 (22 mg) was dissolved in THF (2 mL) and ethanol (2 mL). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 3 days. The ethanol and THF were removed in vacuo, the residue diluted with water and acidified to pH 2 with aqueous 2N HCI solution. The mixture was extracted with ethyl acetate, the organic extract dried (Na2SO4) and concentrated to give the title compound.
MS calcd for (C28H31N5O3 + H)+: 486 MS found (electrospray): (M+H)+ = 486
Example 4
1 -[3-Chloro-4-(1 H-indol^-yOphenyll-S-IKtrans^-methylcyclohexyOcarbonylMI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 51 (30 mg) was dissolved in ethanol (5 mL) and added to sodium ethoxide (21 wt. %) in ethanol (1 mL). The reaction was stirred at 7O0C, under nitrogen, for 2 h and then cooled to room temperature. Water (1 mL) and THF (1 mL) were added and the mixture stirred overnight. The organic solvents were evaporated in vacuo and the residue acidified with 2N HCI solution. The mixture was extracted with ethyl acetate and the organic extract washed with brine, then dried over sodium sulphate and concentrated. The crude product was then purified by MDAP HPLC to give the title compound. MS calcd for (C29H3ICIN4O3 + H)+: 519/21 MS found (electrospray): (M+H)+ = 519/21
Example 5 i-^^H-lndazol^-yOphenyll-S-IKfrans^-methylcyclohexyOcarbonylJti- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 27 (125 mg) was dissolved in THF (3 mL) and ethanol (3 mL). Sodium hydroxide solution (2N, 1.5 mL) was added and the mixture stirred at room temperature for 24 h. The ethanol and THF were removed in vacuo and the residue partitioned between aqueous 2N HCI solution and DCM. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C28H3IN5O3 + H)+: 486 MS found (electrospray): (M+H)+ = 486
Example 6 S-I^fraπs^-MethylcyclohexyOcarbonylKI-methylethyOaminol-i -[4-(1-oxo-1 ,3-dihydro- 2H-isoindol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
To Intermediate 35 (56 mg) in ethanol (5 mL) was added lithium hydroxide (2M, 0.55 mL). The reaction was stirred at room temperature for 16 h and then evaporated to dryness. The
residue was acidified with aqueous 2M HCI solution and purified by OASIS cartridge eluted with water and MeOH, and then purified by 5 g silica SPE cartridge with a gradient of DCM in cyclohexane, then a gradient of EtOAc in DCM to give the title compound. MS calcd for (C29H32N4O4 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 7
1 -[4-(1 ,1 -Dioxido-1 ,2-benzisothiazol-2(3H)-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
To Intermediate 30 (10 mg) in ethanol (2 mL) and THF (3 ml_) was added lithium hydroxide (2M, 0.09 mL). The reaction was stirred at room temperature for 10 days and then evaporated to dryness. The residue was diluted with aqueous 2M HCI solution, and the mixture extracted with EtOAc. The organic extract was washed with 1 M HCI solution, dried using a hydrophobic frit and evaporated to dryness to give the title compound. MS calcd for (C28H32N4O5S + H)+: 537 MS found (electrospray): (M+H)+ = 537
1H NMR (MeOD) δ 8.85 (1 H, s), 7.88 (3H, t), 7.76 (1 H, td), 7.67 (2H, d), 7.64 (2H, d), 5.01 (2H, s), 4.83 (1 H, septet), 2.05 (1 H, tt), 1.79 (3H, d), 1.60 (3H, d), 1.40-0.55 (9H, m), 0.77 (3H, d) carboxylic acid proton not seen.
Example 8
1-(4-Furo[3,2-t)]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 47 (1.32 g) in ethanol (15 mL) and THF (15 mL) was treated with 2M sodium hydroxide solution (9.5 mL) and stirred at room temperature overnight. The reaction was evaporated to dryness and the residue partitioned between ethyl acetate and water and acidified to pH 3 with aqueous 2M HCI solution. The aqueous phase was separated and extracted with more ethyl acetate (x2) and the combined organic phases washed with brine, dried (Na2SO4) and evaporated to give a foam that was purified by chromatography on silica gel using a DCM / methanol gradient to give the title compound. MS calcd for (C28H30N4O^H)+ : 487 MS found (electrospray): (M+H)+ = 487
1H NMR (CDCI3) δ 8.68 (1 H, s), 8.60 (1 H, d), 8.10 (2H, d), 7.88 (3H, dd), 7.39 (1 H, s), 7.29 (1 H, m), 5.02 (1 H, m), 2.05 (1H, br), 1.93-1.60 (5H, br), 1.53-1.23 (5H, br), 1.05 (3H, br), 0.79 (3H1 d), 0.78-0.57 (2H, br) carboxylic acid proton not seen.
Example 9
1 -[4-(1.S-Benzoxazol^-yOphenyll-S-IKϊraπs^-methylcyclohexyOcarbonyl] tetrahydro-
2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 14 (100 mg) was dissolved in THF (1 ml_) and ethanol (1 ml_). Lithium hydroxide solution (2M, 1 ml.) was added and the mixture stirred at room temperature for 24 h. The mixture was acidified with aqueous 2N HCI solution and extracted with DCM. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was dissolved in dioxane and loaded on to a 5g aminopropyl SPE cartridge which had been preconditioned with dioxane. The cartridge was washed with 3 column volumes of dioxane and then with 10% acetic acid in dioxane to elute the product. Fractions containing the product were concentrated to give the title compound. MS calcd for (C30H32N4O5 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Example 10
1 -[4-(1 -Benzofuran^-yOphenyll-S-I^frans^-methylcyclohexyOcarbonyl] tetrahydro-2H- pyran-4-yl)amino]-1 AV-pyrazole-4-carboxylic acid
Intermediate 13 (10 mg) was dissolved in THF (1 mL) and ethanol (1 mL). Lithium hydroxide solution (2M, 1 mL) was added and the mixture stirred at room temperature for 24 h. The mixture was acidified with aqueous 2N HCI solution and extracted with DCM. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound.
MS calcd for (C31H33N3O5 + H)+: 528
MS found (electrospray): (M+H)+ = 528
Example 11
1 -[4-(1 -Benzothien^-yOphenylJ-S-IKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 7 (172 mg) was dissolved in THF (4 mL) and ethanol (2 ml_). Sodium hydroxide solution (2N1 1 mL) was added and the mixture stirred at room temperature for 3 days. The ethanol and THF were removed in vacuo and the residue partitioned between aqueous 2N HCI solution and DCM. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C29H31N3O3S + H)+: 502 MS found (electrospray): (M+H)+ = 502
1H NMR (CD3OD) δ 8.68 (1 H, s), 7.95-7.80 (6H1 m), 7.75 (1 H1 s), 7.39-7.30 (2H, m), 4.83 (1 H, sept), 2.14-2.04 (1 H, m), 1.86-1.76 (2H, m), 1.70-1.53 (3H, m), 1.42-1.22 (5H, m), 1.04- 0.96 (3H, m), 0.78 (3H, d), 0.75-0.54 (2H, m) carboxylic acid proton not seen.
Example 12
3-[[(fraπs-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-[1 ,3]thiazolo[5,4- b]pyridin-2-ylphenyl)-1H-pyrazole-4-carboxylic acid
Intermediate 58 (60 mg) was dissolved in tetrahydrofuran (2 mL) and ethanol (2 mL) and treated with sodium hydroxide solution (2N, 0.4 mL). The reaction mixture was stirred at room temperature for 18 h. The ethanol and THF were removed in vacuo, the residue diluted with water and acidified to pH 3 with aqueous 2N HCI solution. The mixture was extracted with DCM, the organic extract separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC gave the title compound.
MS calcd for (C27H29N5O3S + H)+: 504
MS found (electrospray): (M+H)+ = 504
Example 13
3-[[(fraπs-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 -methyl-1 W-indol-
2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Intermediate 53 (30 mg) was dissolved in THF (1 mL) and ethanol (1 mL). Lithium hydroxide solution (2M, 1 mL) was added and the mixture stirred at room temperature for 24 h. The
mixture was acidified with aqueous 2N HCI solution and extracted with DCM. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C3IH33N3O5 + H)+: 528 MS found (electrospray): (M+H)+ = 528
Example 14
1 -[4-(6-Fluoro-1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 4 (200 mg), Intermediate 38 (200 mg), copper (I) iodide (11 mg), potassium carbonate (188 mg) and (1R,2R)-/V,^dimethyl-1 ,2-cyclohexanediamine (36 mg) in DMF (4 ml.) were heated in a Reactivial at 180° C for 16 h then evaporated to dryness. The residue was dissolved in EtOAc and then washed sequentially with aqueous sodium bicarbonate solution, water and 1 M HCI, dried with a hydrophobic frit and then purified by 10 g silica SPE cartridge with a gradient of DCM in cyclohexane, then a gradient of DCM in methanol. The methanol fractions were evaporated to dryness. The residue was dissolved in ethanol (5 mL) and treated with lithium hydroxide (2M, 1 mL). The reaction was stirred at room temperature for 2 days and then evaporated to dryness. The residue was acidified with aqueous 2M HCI solution and extracted with DCM. The organic extract was washed with aqueousi M HCI solution dried with a hydrophobic frit and evaporated to dryness. The crude product was purified by HPLC using a Supelco ABZ++ 100 x 21.2 mm, 5 um; column eluted with 65 - 80% acetonitrile (containing 0.05% formic acid) and water over 30 minutes at 20 mL/min flow rate to give the title compound. MS calcd for (C29H31 FN4O4 + H)+: 519 MS found (electrospray): (M+H)+ = 519
Example 15 S-lKfraπs^-MethylcyclohexyOcarbonylKtetrahydro-ΣH-pyran^-yOaminol-i -[4-(1 -oxo- 1 ,3-dihydro-2H-isoindol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
To Intermediate 32 (92 mg) in ethanol (2 mL) and THF (2 mL) was added lithium hydroxide (2M, 0.8 mL). The reaction was stirred at room temperature for 16 h and then evaporated to
dryness. The residue was diluted with aqueous 2M HCI solution, and the mixture extracted with EtOAc. The organic extract was washed with 1 M HCI solution, dried using a hydrophobic frit and evaporated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C3IH34N4O5 + H)+: 543 MS found (electrospray): (M+H)+ = 543
The following examples were prepared in a similar manner using the appropriate starting reagents (see table)
Example 16
1 -[4-(1.S-Benzoxazol^-yOphenyll-S-l^frans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Example 17
1-[4-(1,3-Benzoxazol-2-yl)phenyl]-3-{ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}- 1 H-pyrazole-4-carboxylic acid
Example 18 1 -[4-(1 ,3-Benzothiazol-2-yl)phenyl]-3-[[(^rans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Example 19
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Example 20
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro- 2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid
Example 21
3-{Ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-furo[3,2-Jb]pyridin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid
Example 22
S^Ethyl^frans^-methylcyclohexyOcarbonyllamino^i^-imidazoti^-aJpyridin^- ylphenyl)-1 H-pyrazole-4-carboxylic acid
Example 23 1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(fraπs-4- methylcyclohexyOcarbonylKtetrahydro^H-pyran^-yOaminoJ-i H-pyrazole-4-carboxylic acid
Example 24
1 -[4-(5-Fluoro-1 H-indol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Example 25
1-[4-(3-Chloro-2H-indazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Example 26 i-^^I.S-Benzothiazol^-yOphenyll-a-IKfrans^-methylcyclohexyOcarbonylKtetrahydro- 2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid
Example 27 1 -[4-(1 -Benzofuran^-yOphenyll-S-t^trans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Table 1
Example 28
1-[4-(6-Chloro-7aH-imidazo[4,5-b]pyridin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 59 (90 mg) was dissolved in THF (0.5 ml.) and methanol (0.5 mL). Sodium hydroxide solution (2N, 0.5 mL) was added and the mixture stirred at room temperature for 3 h. The methanol and THF were evaporated under vacuum, and the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C27H29CIN6O3 + H)+: 521
MS found (electrospray): (M+H)+ = 521
1H NMR (MeOD) δ 9.00 (1 H, s), 8.37 (1 H, br.s), 8.32 (2H, d), 8.09 (2H, d), 8.04 (1 H1 br.s), 4.90-4.82 (1 H, m), 2.12-2.03 (1 H, m), 1.84-1.76 (2H1 m), 1.71-1.54 (3H, m), 1.42-1.29 (2H, m), 1.28 (3H, m), 1.03-0.90 (3H, m), 0.80-0.75 (3H, d), 0.75-0.53 (2H, m) carboxylic acid proton and amine proton not seen.
Example 29
1-[4-(7-Amino-5-methylpyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 60 (35 mg) was dissolved in THF (0.2 ml_) and methanol (0.2 ml_). Sodium hydroxide solution (2N, 0.2 mL) was added and the mixture stirred at room temperature for 8 h. The methanol and THF were evaporated under vacuum, and the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by ion exchange chromatography on aminopropyl capped silica, eluting with methanol then 10% 2N HCI / methanol, to give the title compound.
MS calcd for (C28H33N7O3 + H)+: 516 MS found (electrospray): (M+H)+ = 516 1H NMR (MeOD) δ 8.97 (1 H, s), 8.26 (2H, d), 8.01 (2H, d), 6.93 (1 H, s), 6.26 (1 H, s), 4.90- 4.80 (1 H, m), 2.58 (3H, s), 2.11-2.00 (1 H, m), 1.82-1.73 (2H, m), 1.70-1.50 (3H, m), 1.42- 1.28 (2H, m), 1.26 (3H, m), 1.04-0.91 (3H, m), 0.81-0.75 (3H, d), 0.75-0.51 (2H, m) carboxylic acid proton and amine protons not seen.
Example 30
1 -[4-(5-Amino-1.S-benzoxazol^-yOphenylJ-S-IKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 61 (60 mg) was dissolved in THF (0.3 mL) and methanol (0.3 mL). Sodium hydroxide solution (2N, 0.3 mL) was added and the mixture stirred at room temperature for 20 h. The methanol and THF were evaporated under vacuum, and the residue was partitioned between DCM and 2N HCI/methanol solution. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound.
MS calcd for (C28H31N5O4 + H)+: 502 MS found (electrospray): (M+H)+ = 502
1H NMR (MeOD) δ 9.05 (1 H, S)1 8.44 (2H, d), 8.12 (2H, d), 7.86 (1 H, d), 7.73 (1 H, s), 7.40 (1 H, d), 4.93-4.80 (1 H, m), 2.10-2.02 (1 H, m), 1.83-1.75 (2H, m), 1.70-1.54 (3H, m), 1.41- 1.30 (2H, m), 1.28 (3H, d), 0.99 (3H, d), 0.78 (3H, d), 0.75-0.54 (2H, m), carboxylic acid proton and amine protons not seen.
Example 31
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][(3/?)- tetrahydro-3-furanyl]amino}-1W-pyrazole-4-carboxylic acid
Intermediate 62 (150 mg) was dissolved in THF (0.7 mL) and methanol (0.7 mL). Sodium hydroxide solution (2N, 0.7 mL) was added and the mixture stirred at room temperature for 17 h. The methanol and THF were evaporated under vacuum, then the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C29H30N4O5 + H)+: 515 MS found (electrospray): (M+H)+ = 515 1H NMR (MeOD) δ 9.08 (1 H, s), 8.79 (1 H, d), 8.76 (1 H, d), 8.35 (2H, d), 8.12 (2H, d), 7.93 (1 H, t), 7.89 (1 H, s), 5.22-5.03 (1 H, m), 4.89-4.84 (1 H, m), 4.05-3.91 (1 H, m), 3.85-3.77 (1 H, m), 3.69-3.59 (2H, m), 2.33-2.10 (1 H, m), 2.10-2.00 (1 H, m), 1.91-1.71 (2H, m), 1.71-1.49 (3H, m), 1.49-1.24 (2H, m), 0.78 (3H, d), 0.78-0.55 (2H, m), carboxylic acid proton not seen.
Example 32 i^-Furo^^-blpyridin^-ylphenyO-a^Kfrans^-methylcyclohexyOcarbonyllltSS)- tetrahydro-3-furanyl]amino}-1 H-pyrazole-4-carboxylic acid
Intermediate 63 (151 mg) was dissolved in THF (0.7 mL) and methanol (0.7 mL). Sodium hydroxide solution (2N, 0.7 mL) was added and the mixture stirred at room temperature for 17 h. The methanol and THF were evaporated under vacuum, and the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C29H30N4O5 + H)+: 515
MS found (electrospray): (M+H)+ = 515
1H NMR (MeOD) δ 9.05 (1H, s), 8.67 (1H, d), 8.59 (1H, d), 8.29 (2H, d), 8.11 (2H, d), 7.80- 7.75 (1H, m), 7.74 (1 H, s), 5.21-5.02 (1 H, m), 4.97-4.79 (1 H, m), 4.05-3.91 (1 H, m), 3.83- 3.77 (1H, m), 3.70-3.59 (2H, m), 2.35-2.10 (1H, m), 2.10-2.00 (1H, m), 1.92-1.70 (2H1 m), 1.70-1.49 (3H, m), 1.47-1.23 (2H, m), 0.78 (3H1 d), 0.74-0.54 (2H, m), carboxylic acid proton not seen.
Example 33
1 -[4-(4-Amino-3-cyanopyrazolo[5,1 -c][1 ,2,4]triazin-7-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 64 (130 mg) was dissolved in THF (0.6 mL) and methanol (0.6 mL). Sodium hydroxide solution (2N, 0.6 mL) was added and the mixture stirred at room temperature for
15 h. The methanol and THF were evaporated under vacuum, then the residue was partitioned between DCM and 2N HCI / methanol solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound.
MS calcd for (C27H29N9O3 + H)+: 528
MS found (electrospray): (M+H)+ = 528 1H NMR (MeOD) δ 8.97 (1 H, s), 8.31 (2H, d), 8.00 (2H, d), 7.48 (1 H, s), 4.93-4.80 (1 H, m),
2.12-2.04 (1H, m), 1.85-1.76 (2H, m), 1.70-1.53 (3H1 m), 1.43-1.30 (2H, m), 1.30-1.23 (3H, m), 1.03-0.96 (3H, m), 0.78 (3H, m), 0.76-0.55 (2H, m), carboxylic acid proton and amine protons not seen.
Example 34 i-μ-^H-lndazol^-ytyphenyll-S-Hffrans^-methylcyclohexyOcarbonylHI- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 66 (130 mg) was dissolved in THF (4 mL) and ethanol (4 mL). Sodium hydroxide solution (2N, 0.4 mL) was added and the mixture stirred at room temperature for
24 h. The ethanol and THF were removed in vacuo and the residue partitioned between aqueous 2N HCI solution (pH=3) and DCM. The DCM layer was separated using a
hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound.
MS calcd for (C29H3IN3O3S + H)+: 504
MS found (electrospray): (M+H)+ = 504
Example 35
1-(4-Furo[3,2-cy]pyrimidin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 68 (0.03 g) in ethanol (2ml_) and THF (2 ml_) was treated with sodium hydroxide solution (2M, 1.0 ml_) and stirred at room temperature overnight. The reaction was evaporated to dryness and the residue partitioned between dichloromethane and water and acidified to pH 2 with aqueous 2N HCI solution. The organic phase was passed through a hydrophobic frit and evaporated to give a solid. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C28H30N4C^H)+ : 488 MS found (electrospray): (M+H)+ = 488
Example 36 1 -(4-Furo[2,3-</]pyrimidin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 70 (0.12 g) in ethanol (4mL) and THF (4 mL) was treated with sodium hydroxide solution (2M, 0.4 mL) and stirred at room temperature overnight. The reaction was evaporated to dryness and the residue partitioned between dichloromethane and water and acidified to pH 3 with aqueous 2M HCI solution. The organic phase was passed through a hydrophobic frit and evaporated to give a solid. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C28H30N4CVH)+ : 488 MS found (electrospray): (M+H)+ = 488
Example 37
1 -(4-lmidazo[2,1 -b][1.Slthiazol-θ-ylphenyO-S^Kfraπs^-methylcyclohexyOcarbonylHI - methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Intermediate 71 (192 mg) was dissolved in THF (4 mL) and ethanol (4 ml_). Sodium hydroxide solution (2N, 2 mL) was added and the mixture stirred at room temperature for 48 h. The ethanol and THF were removed in vacuo and the residue partitioned between aqueous 2N HCI solution and DCM. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C26H29N5O3S + H)+: 492 MS found (electrospray): (M+H)+ = 492
1H NMR (CD3OD) δ 8.65 (1 H, s), 7.98 (2H, d), 7.85-7.78 (3H, m), 7.51 (1 H1 d), 6.95 (1 H, d), 4.99 (1 H, sept) 2.10-2.00 (1 H, m), 1.90-1.20 (1OH, br. m), 1.02 (3H, br d), 0.77 (3H, d), 0.74- 0.58 (2H, br m) carboxylic acid proton not seen.
Example 38
1-[4-(5J-Dimethylpyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 72 (277 mg) was dissolved in THF (5 mL) and ethanol (5 mL). Sodium hydroxide solution (2N, 2.5 mL) was added and the mixture stirred at room temperature for
48 h. The ethanol and THF were removed in vacuo and the residue partitioned between aqueous 2N HCI solution and DCM. The DCM layer was separated using a hydrophobic frit and concentrated to give the title compound.
MS calcd for (C29H34N6O3 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Example 39
1 -(4-lmidazo[1 ,2-a]pyrimidin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 73 (81 mg) was dissolved in THF (2 mL) and ethanol (2 mL). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 48 h. The
ethanol and THF were removed in vacuo, the residue diluted with water and acidified to pH 3 with aqueous 2N HCI solution. The mixture was extracted with DCM, the organic phase separated using a hydrophobic frit and concentrated to give the title compound MS calcd for (C27H30N6O3 + H)+: 487 MS found (electrospray): (M+H)+ = 487
Example 40
1-(4-Furo[3,2-c]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 4 (28 mg), Intermediate 74 (24 mg), copper iodide (5 mg), potassium carbonate (25 mg) and (1R,2R)-Λ/,Λ/'-dimethyl-1 ,2-cyclohexanediamine (10 mg) dissolved in DMF (4 ml_) was heated at 160° C in a Reactivial for 2 days. The solvent was removed in vacuo and the residue acidified to pH 6, taken into ethyl acetate, washed with water, dried using a hydrophobic frit and evaporated to dryness. This residue was then dissolved in ethanol (3 ml_). Lithium hydroxide solution (2M, 0.5 ml.) was added and the reaction mixture stirred at room temperature for 20 hours. The reaction mixture was evaporated to dryness, acidified with 2N HCI solution, taken into ethyl acetate (50 mL), washed with HCI solution (2M, 50 mL), dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 5g silica SPE, eluted with a gradient of ethyl acetate in acetone. The product containing fractions were combined and dried to give the title compound.
MS calcd for (C28H30N4O4 + H)+: 487 MS found (electrospray): (M+H)+ = 487 1H NMR (CD3OD): δ 8.88 (1 H, s), 8.69 (1 H, s), 8.42 (1 H, d), 8.08 (2H, d), 7.95 (2H, d), 7.66 (1H, d), 7.40 (1H1 s), 4.87-4.78 (1H, m), 2.21-2.16 (1 H, m), 1.92-1.78 (2H, m), 1.65-1.55 (3H, m), 1.32-0.97 (8H, m), 0.75 (3H, d), 0.72-0.52 (2H, m) carboxylic acid proton not seen.
Example 41 1 -(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-{(1 -methylethyl)[(4- methylphenyl)carbonyl]amino}-1 H-pyrazole-4-carboxylic acid
Intermediate 77 (57 mg) was dissolved in THF (0.75 mL) and ethanol (0.75 mL) and treated with NaOH (2N, 0.45 mL). The reaction was stirred at room temperature for 18 hours. The
ethanol and THF were removed in vacuo and the residue acidified with 2N HCI solution. The mixture was extracted with ethyl acetate and the organic extract dried (MgSO4) and concentrated. The crude product was purified by ISCO companion silica chromatography eluting with a gradient of methanol in dichloromethane. The crude product was further purified by MDAP HPLC to give the title compound. MS calcd for (C28H24N4O4 + H)+: 481 MS found (electrospray): (M+H)+ = 481
Example 42 3-[(Cyclohexylcarbonyl)(1 -methylethyl)amino]-1 -(4-furo[3,2-b]pyιϊdin-2-ylphenyl)-1 H- pyrazole-4-carboxylic acid
Intermediate 80 (112 mg) was dissolved in THF (1 mL) and ethanol (1 ml_) and treated with NaOH (2N, 1.1 mL). The reaction was stirred at room temperature for 18 hours. The ethanol and THF were removed in vacuo and the residue acidified with 2N HCI solution. The mixture was extracted with ethyl acetate followed by DCM followed by chloroform and the organic extracts dried (MgSO4) and concentrated. The residue was purified by 5g silica SPE cartridge eluting with a gradient of methanol in chloroform to give the title compound. MS calcd for (C27H28N4O4 + H)+: 473 MS found (electrospray): (M+H)+ = 473
Example 43
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-[1 ,3]oxazolo[4,5- b]pyridin-2-ylphenyl)-1 W-pyrazole-4-carboxylic acid
Intermediate 82 (150 mg), Intermediate 83 (141 mg), frans-(1 R,2R)N,N'-bismethyl-1 ,2- cyclohexanediamine (29 mg) and potassium carbonate (212 mg) were dissolved in dry DMF (4 mL) and treated with copper iodide (10 mg). The reaction was heated at 155°C in a Reactivial for 18 hours. The DMF was removed in vacuo and the residue acidified with 2N HCI solution. The mixture was extracted with ethyl acetate and the organic extract dried (MgSO4) and concentrated. The crude product was purified by ISCO companion silica
chromatography eluting with a gradient of methanol in dichloromethane. The crude product was further purified by MDAP HPLC. The crude product was further purified by 5g aminopropyl SPE cartridge eluting with methanol followed by 10% acetic acid in methanol to give the title compound. MS calcd for (C27H29N5O4 + H)+: 488 MS found (electrospray): (M+H)+ = 488
Example 44
S^Ethyl^frans^-methylcyclohexyOcarbonyllaminoH^-pyrazoloti^-alpyrimidin^- ylphenyO-IH-pyrazole^-carboxylic acid
To Intermediate 84 (30 mg) was added THF (1 ml_), EtOH (1 mL) and LiOH (2M, 1 mL). The reaction was then stirred at room temperature overnight. The solvent was evaporated, water
(10 mL) added and the pH adjusted to pH 4-5. The solution was extracted with DCM and the organic phase concentrated. The crude product was purified by 1g NH2 SPE loaded in 1 ,4- dioxane, the column washed 3 times with 1 ,4-dioxane then eluted with 10% acetic acid in
1 ,4-dioxane to give the title compound.
MS calcd for (C26H28N6O3 + H)+: 473
MS found (electrospray): (M+H)+ = 473
Example 45
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-3-furanyl)amino]-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To Intermediate 85 (100 mg) was added THF (3 mL), EtOH (1 mL) and LiOH (2M, 1 mL). The reaction was then stirred at room temperature overnight. The solvent was removed, then acidified with hydrochloric acid (2N, -1 mL) then partitioned between water and DCM. The organic phase was separated with a hydrophobic frit and concentrated. The crude product was then purified by MDAP HPLC to give the title compound. MS calcd for (C28H30N6O4 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Example 46
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To intermediate 92 (100 mg) was added THF (3 ml_), EtOH (1 mL) and LiOH (2M, 1 mL). The reaction was then stirred at room temperature overnight. The solvent was removed, then acidified with hydrochloric acid (2N, -1 mL) and partitioned between water and DCM. The organic phase was separated with a hydrophobic frit and concentrated. The aqueous phase was extracted further with EtOAc and the organic phase combined with the first and concentrated. The crude product was then purified by MDAP HPLC to give the title compound.
MS calcd for (C29H32N6O4 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Example 47
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-[[(trans-4-methylcyclohexyl)carbonyl] (tetrahydro-3-furanyl)amino]-1 H-pyrazole-4-carboxylic acid
To Intermediate 91 (100 mg) was added THF (3 mL), EtOH (1 mL) and LiOH (2M, 1 mL). The reaction was then stirred at room temperature overnight. The solvent was removed, then acidified with hydrochloric acid (2N, -1 mL) and partitioned between water and DCM.
The organic phase was separated with a hydrophobic frit and concentrated. The crude product was then purified by MDAP HPLC to give the title compound.
MS calcd for (C29H30N5O5 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Example 48
1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(fraπs-4- methylcyclohexyl)carbonyl](tetrahydro-3-furanyl)amino]-1 H-pyrazole-4-carboxylic acid
To Intermediate 93 (100 mg) was added THF (3 ml_), EtOH (1 mL) and LiOH (2M, 1 ml_). The reaction was then stirred at room temperature overnight. The solvent was removed, then acidified with hydrochloric acid (2N, -1 mL) then partitioned between water and DCM. The organic phase was separated with a hydrophobic frit and concentrated. The aqueous phase was extracted further with EtOAc and the organic phases combined and concentrated to give the title compound. MS calcd for (C29H31N5O4 + H)+: 514 MS found (electrospray): (M+H)+ = 514
Example 49
1 -[5-(1.S-Benzoxazol^-yO^-thienyll-S-IIttrans^-methylcyclohexyOcarbony^i - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
To Intermediate 87 (170 mg) was added THF (3 mL), EtOH (2 mL) and LiOH (2M, 2 mL). The reaction was then stirred at room temperature over a weekend. The solvent was removed, hydrochloric acid (2N, 2 mL) added and then partitioned between water and EtOAc. The organic phase was separated, dried and concentrated. The crude product was then purified by MDAP HPLC, then dissolved in 1 ,4-dioxane and loaded onto a NH2 SPE cartridge. The cartridge was washed with 1,4-dioxane and eluted with 10% acetic acid in 1 ,4-dioxane to give the title compound. MS calcd for (C26H28N4O4S + H)+: 492 MS found (electrospray): (M+H)+ = 492
Example 50
1 -[4-(1 ^-Benzoxazol^-yO-S-chlorophenyll-S-Hftrans^-methylcyclohexyl) carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
To Intermediate 89 (110 mg) was added THF (1 mL), EtOH (1 mL) and LiOH (2M, 1 ml_). The reaction was then stirred at room temperature over a weekend. The solvent was removed, then neutralised with hydrochloric acid (2N, 2 mL) and partitioned between water and EtOAc. The organic phase was separated, dried and concentrated. The crude product was then purified by HPLC, firstly with a LC-ABZ+plus 100x21.2 mm ID 5 uM column (Solvent A 0.1% TFA in water, Solvent B 0.05% TFA in MeCN) eluting with a 50-100% B gradient over 30 mins at 20 mL/min. Secondly with a Supelcosil LC-ABZ+plus 100x21.2 mm ID 5 uM column (Solvent A 0.1 % formic acid in water, Solvent B 95% MeCN 5% 0.1% formic acid in water) with a 50-90% B gradient over 25 mins at 20 mL/min to give the title compound.
MS calcd for (C26H28N4O4S + H)+: 492 MS found (electrospray): (M+H)+ = 492
Example 51
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(5- methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Intermediate 100 (80 mg) and 2M lithium hydroxide solution (375 μL) were stirred in THF (0.75 mL) and methanol (0.75 mL) at ambient temperature for 16 hours. The solvent was evaporated under vacuum, and the residue was partitioned between DCM/methanol and 2M hydrochloric acid. The organic phase was separated and evaporated under vacuum. The residue was agitated in Et2O until a crystalline solid formed, then solvent was evaporated to give the title compound as a powder. MS calcd for (C28H32N6O3 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 52
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(7- methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Intermediate 102 (41 mg) and 2M lithium hydroxide solution (190 μl_) were stirred in THF (0.4 ml.) and methanol (0.4 mL) at ambient temperature for 16 hours. The solvent was evaporated under vacuum, then the residue was partitioned between DCM/methanol and 2M hydrochloric acid. The organic phase was separated, evaporated under vacuum and purified by MDAP HPLC to give a gum. This was freeze dried from dioxane to give the title compound as an amorphous solid. MS calcd for (C28H32N6O3 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 53
S-tttfrans^-MethylcyclohexyOcarbonylKI-methylethyOaminol-i-^S-methylfuro^^- b]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Intermediate 104 (113 mg) and 2M lithium hydroxide solution (525 μL) were stirred in THF (1 mL) and methanol (1 mL) at ambient temperature for 16 hours. The solvent was evaporated under vacuum, and the residue was partitioned between DCM/methanol and 2M hydrochloric acid. The organic phase was separated, evaporated under vacuum and purified by MDAP HPLC to give the title compound as a solid. MS calcd for (C29H32N4O6 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 54
S-t^fraπs^-MethylcyclohexyOcarbonylKI-methylethyOaminol-i-^-tβ-methylfurotS^- Jb]pyridin-2-yl)phenyl]-1H-pyrazole-4-carboxylic acid
Intermediate 106 (106 mg) and 2M lithium hydroxide solution (0.5 ml.) were stirred in THF (0.5 mL) and methanol (0.5 mL) at ambient temperature for 5 hours. The solvent was evaporated under vacuum, and the residue was partitioned between DCM and 2M hydrochloric acid. The organic phase was separated, evaporated under vacuum and purified by ion exchange chromatography over aminopropyl capped silica gel eluting with methanol then 10% 2M hydrochloric acid in methanol. The product obtained was further purified by MDAP HPLC to give the title compound as a solid. MS calcd for (C29H32N4O6 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 55
1 -[6-(1.S-Benzoxazol^-yO-S-pyridinyll-S-I^frans-A-methylcyclohexyOcarbonyl] (1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 108 (150 mg) and 2M lithium hydroxide solution (0.75 mL) were stirred in THF
(0.75 mL) and methanol (0.75 mL) at ambient temperature overnight. The solvent was evaporated under vacuum, and the residue was partitioned between DCM and 2M hydrochloric acid. The organic phase was separated, evaporated under vacuum and freeze dried from dioxane to give the title compound as an amorphous solid. MS calcd for (C27H29N5O4 + H)+: 488 MS found (electrospray): (M+H)+ = 488
Example 56 1 -(G-Furoβ^-ϋlpyridin^-yl-SφyridinyO-S-lKfrans^-methylcyclohexyOcarbonyl] (1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 110 (97 mg) and 2M lithium hydroxide solution (0.5 ml.) were stirred in THF (0.5 ml_) and methanol (0.5 ml_) at ambient temperature for 4 hours. The solvent was evaporated under vacuum, and the residue was partitioned between DCM/methanol and 2M hydrochloric acid. The organic phase was separated, evaporated under vacuum and triturated in Et2O. A powder was filtered off and dried under vacuum to give the title compound. MS calcd for (C27H29N5O4 + H)+: 488 MS found (electrospray): (M+H)+ = 488
Example 57
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(6-pyrazolo[1 ,5- a]pyrimidin-2-yl-3-pyridinyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 114 (200 mg) and 2M lithium hydroxide solution (1 ml.) were stirred in THF (1 ml_) and methanol (1 mL) at 230C for 18 hours. The solvent was evaporated under vacuum, and the residue was partitioned between DCM and dilute citric acid. The organic phase was separated and evaporated under vacuum to give the title compound as a solid.
MS calcd for (C26H29N7O3 + H)+: 488
MS found (electrospray): (M+H)+ = 488
Example 58
1-[6-(6-Fluoropyrazolo[1,5-a]pyrimidin-2-yl)-3-pyridinyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 116 (9 mg) and sodium hydroxide solution (2M, 45 μL) were stirred in THF (0.1 mL) and methanol (0.1 mL) at ambient temperature for 16 hours. The mixture was partitioned
between chloroform and 2M hydrochloric acid. The organic phase was separated, evaporated under vacuum and purified by ion exchange over aminopropyl capped silica gel, eluting with methanol then 10% 2M hydrochloric acid in methanol. Product fractions were evaporated to give a gum which was freeze dried from dioxane to give the title compound as an amorphous solid.
MS calcd for (C27H29FN6O3 + H)+: 505 MS found (electrospray): (M+H)+ = 505
Example 59 S-t^fraπs^-MethylcyclohexyOcarbonylKtetrahydro^H-pyran^-ylmethyOaminoJ-i -(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylic acid
Impure Intermediate 120 (109 mg) was dissolved in THF (4 mL) and ethanol (2 ml_). Lithium hydroxide solution (2M, 2 mL) was added and the mixture stirred at room temperature for 24 h. The solvents were removed in vacuo, and the residue partitioned between hydrochloric acid (2M, 4 mL) and DCM. The organic layer was separated using a hydrophobic frit, and concentrated in vacuo. The residue was purified by MDAP HPLC to give the title compound.
MS calcd for (C30H34N6CVH)+: 543
MS found (electrospray): (M+H)+ = 543
Example 60 i^-Furo^^-blpyridin^-ylphenyO-a^^frans^-methylcyclohexyOcarbonyllp-
(methyloxy)ethyl]amino}-1 W-pyrazole-4-carboxylic acid
Crude Intermediate 123 (194 mg) was dissolved in THF (3 mL) and ethanol (3 mL). Lithium hydroxide solution (2M, 3 mL) was added and the mixture stirred at room temperature for 16 h. Hydrochloric acid (2M1 3 mL) was added, and the mixture was partitioned between water and DCM. The organic layer was separated using a hydrophobic frit, and concentrated in vacuo. This was purified by MDAP HPLC. Further purification by mass directed HPLC using a Suppelco ABZ+ column (Ci8, 100 x 20 mm, 20 mL/min flow rate) and eluting with mobile phase A (0.2% aqueous TFA) and mobile phase B (95% aqueous MeCN containing 0.1% TFA), [gradient of 20 - 100% mobile phase B over 40 minutes, injected in DMSO:MeOH 1 :1 , volume of 500 ul x2) gave the title compound.
MS calcd for (C28H30N4(VH)+: 503 MS found (electrospray): (M+H)+ = 503
Example 61
1 -(4-lmidazo[2,1 -b][1 ,3]thiazol-6-ylphenyl)-3-[[(.raπs-4- methylcyclohexyl)carbonyl](tetrahydro-3-furanyl)amino]-1H-pyrazole-4-carboxylic acid
Intermediate 124 (180 mg) was dissolved in THF (3 ml_) and ethanol (3 ml_). Lithium hydroxide solution (2M1 3 mL) was added and the mixture stirred at room temperature for 20 h. Hydrochloric acid (2M, 3 mL) was added, and the mixture was partitioned between water and DCM. The organic layer was separated using a hydrophobic frit, and concentrated in vacuo to give the title compound.
MS calcd for (C27H29N5O^H)+: 520
MS found (electrospray): (M+H)+ = 520
Example 62
1 -(4-lmidazo[2,1 -b][1 ,3]thiazol-6-ylphenyl)-3-[[(£rans-4-rnethylcyclohexyl) carbonylKtetrahydro^H-pyran^-yOaminol-IH-pyrazole^-carboxylic acid
Intermediate 125 (38 mg) was dissolved in THF (2 mL) and ethanol (2 mL). Lithium hydroxide solution (2M, 2 mL) was added and the mixture stirred at room temperature for 20 h. Hydrochloric acid (2M, 2 mL) was added, and the mixture was partitioned between water and DCM. The organic layer was separated using a hydrophobic frit, and concentrated in vacuo to give the title compound. MS calcd for (C28H31 N5O4S+H)+: 534 MS found (electrospray): (M+H)+ = 534
Example 63
3-{(2-Ethylbutyl)[(fraπs-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylic acid
Impure Intermediate 126 was dissolved in THF (1 ml.) and ethanol (1 ml_). Lithium hydroxide solution (2M, 1 mL) was added and the mixture stirred at room temperature for 96 h. Hydrochloric acid (2M, 1 ml.) was added, and the mixture was partitioned between water and DCM. The organic layer was separated using a hydrophobic frit, and concentrated in vacuo. This was then purified by MDAP HPLC to give the title compound. MS calcd for (C30H36N6CVH)+: 529 MS found (electrospray): (M+H)+ = 529
1 H NMR (CDCI3) δ 8.73 (1H, d), 8.64 (1 H, s), 8.51 (1H, dd), 8.15 (2H, d), 7.86 (2H, d), 7.05 (1 H, s), 6.86 (1H, dd), 4.17 (2H, br), 2.15 (1 H, t), 1.85 (2H1 m), 1.65-1.56 (5H, m), 1.48-1.27 (6H, m), 0.88-0.62 (1OH, m)
Example 64
3-{[(fraπs-4-Methylcyclohexyl)carbonyl][2-(methyloxy)ethyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylic acid
Intermediate 127 (64 mg) was dissolved in THF (1 mL) and ethanol (1 mL). Lithium hydroxide solution (2M1 1 mL) was added, and the mixture stirred at room temperature for 20 h. Hydrochloric acid (2M, 1 mL) was added, and the mixture was partitioned between water and DCM. The layers were stirred, and then the organic layer separated using a hydrophobic frit, and concentrated in vacuo to give the title compound. MS calcd for (C27H30N6CVH)+: 503 MS found (electrospray): (M+H)+ = 503
Example 65
3-[[(frans-4-Methylcyclohexyl)carbonyl](propyl)amino]-1-(4-pyrazolo[1,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL) in a greenhouse plus reaction tube, was added sodium hydride (60% dispersion in mineral oil) (17 mg). This was stirred at room temperature, under nitrogen, for 15 minutes. 1- lodopropane (180 mg) was added, and the reaction was stirred at 5O0C, under nitrogen, for 20 h. The reaction was then allowed to cool to room temperature, and water (0.5 mL) was added, and the mixture stirred for 45 minutes. The solvent was then removed by evaporation. The residue was then dissolved in THF (1 mL) and ethanol (1 mL). Lithium hydroxide (2N, 1 mL) was added, and the reaction mixture was stirred at room temperature for 20 h. 2N Hydrochloric acid (1 mL) was added, and the mixture was partitioned between water and DCM, the layers stirred for 30 minutes, and the organic phase separated using a hydrophobic frit. This was concentrated by evaporation, and then purified by MDAP HPLC to give the title compound. MS calcd for (C27H30N6CVH)+: 487 MS found (electrospray): (M+H)+ = 487
1 H NMR (MeOD) δ 8.95 (1 H, d), 8.90 (1 H, s), 8.51 (1 H, d),
8.20 (2H, d), 7.95 (2H, d), 7.12 (1 H, s), 7.01 (1 H, dd), 3.66 (2H, br), 2.17 (1 H, tt), 1.81 (2H, d), 1.64-1.47 (6H, m), 1.32 (1 H, m), 0.92 (3H, t), 0.79 (3H, d), 0.70 (2H, dq)
Example 66
3-[[(frans-4-Methylcyclohexyl)carbonyl](2-methylpropyl)amino]-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL) in a greenhouse plus reaction tube, was added sodium hydride (60% dispersion in mineral oil) (17 mg). This was stirred at room temperature, under nitrogen, for 15 minutes. 1-lodo-2- methylpropane (195 mg) was added, and the reaction was stirred at 5O0C, under nitrogen, for 20 h. The reaction was then allowed to cool to room temperature, water (0.5 mL) was added, and the mixture stirred for 45 minutes. The solvent was removed by evaporation. The residue was then dissolved in THF (1 mL) and ethanol (1 mL). Lithium hydroxide (2M, 1 mL) was added, and the reaction mixture was stirred at room temperature for 20 h.
Hydrochloric acid (2M1 1 mL) was added, and the mixture was partitioned between water and DCM, the layers stirred for 30 minutes, and the organic phase separated using a hydrophobic frit. This was concentrated by evaporation, and then purified by MDAP HPLC to give the title compound. MS calcd for (C28H32N6C^H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 67
3-{(1 -Ethylpropyl)[(trans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL) in a greenhouse plus reaction tube, was added sodium hydride (60% dispersion in mineral oil) (17 mg). This was stirred at room temperature, under nitrogen, for 15 minutes. 3- Bromopentane (160 mg) was added, and the reaction was stirred at 5O0C, under nitrogen, for 20 h. Water (0.5 mL) was added, and the mixture stirred at room temperature for 45 minutes. The solvent was then removed by evaporation. The residue was then dissolved in THF (1 mL) and ethanol (1 mL). Lithium hydroxide (2M, 1 mL) was added, and the reaction mixture was stirred at room temperature for 20 h. Hydrochloric acid (2M, 1 mL) was added, and the mixture was partitioned between water and DCM, the layers stirred for 30 minutes, and the organic phase separated using a hydrophobic frit. This was concentrated by evaporation, and then purified by MDAP HPLC to give the title compound. MS calcd for (C29H34N6O3+H)+: 515 MS found (electrospray): (M+H)+ = 515
Example 68
3-{(1 ^-DimethylbutyOKfrans^-methylcyclohexyOcarbonyllaminoH -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL) in a greenhouse plus reaction tube, was added sodium hydride (60% dispersion in mineral oil) (17 mg). This was stirred at room temperature, under nitrogen, for 15 minutes. 2-Bromo-4-
methylpentane (175 mg) was added, and the reaction was stirred at 5O0C, under nitrogen, for 20 h. Water (0.5 mL) was added, and the mixture stirred at room temperature for 45 minutes. The solvent was then removed by evaporation. The residue was dissolved in THF (1 mL) and ethanol (1 mL). Lithium hydroxide (2M, 1 mL) was added, and the reaction mixture was stirred at room temperature for 20 h. Hydrochloric acid (2M, 1 mL) was added, and the mixture was partitioned between water and DCM, the layers stirred for 30 minutes, and the organic phase separated using a hydrophobic frit. This was concentrated by evaporation, and then purified by MDAP HPLC to give the title compound. MS calcd for (C30H36N6CVH)+: 529 MS found (electrospray): (M+H)+ = 529
Example 69
3-{Cyclopentyl[(fraπs-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1
Intermediate 131 (521 mg) was dissolved in THF (7 mL) and ethanol (7 mL). Lithium hydroxide (2M, 7 mL) was added and the reaction mixture stirred at room temperature for 16 h. 2M Hydrochloric acid (7 mL) was added, and the mixture was partitioned between water and DCM. The layers were stirred, and then the organic phase was separated using a hydrophobic frit, and concentrated by evaporation. The residue was then purified using a 10g aminopropyl capped silica SPE column eluting with 10% HCI in methanol. The fractions containing product were combined and concentrated in vacuo to give the title compound. MS calcd for (C29H32N6CVH)+: 513 MS found (electrospray): (M+H)+ = 513 1H NMR (CD3OD) δ 9.10 (1 H, d), 8.93 (1H, s), 8.58 (1H,dd), 8.23 (2H, d), 7.97 (2H, d), 7.18 (1 H1 s), 7.10 (1 H, dd), 4.82 (1 H, m), 2.04 (2H, m), 1.90-1.22 (14H, m), 0.78 (3H, d), 0.70 (2H, m), carboxylic acid proton not seen.
Example 70 3-{(1-Methylethyl)[(4-methylphenyl)carbonyl]amino}-1-(4-pyrazolo[1,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 139 (146 mg) was dissolved in tetrahydrofuran (1.5 ml_) and ethanol (1.5 ml_) and treated with sodium hydroxide solution (2N, 1.1 ml_). The reaction mixture was stirred at room temperature for 42 h. The reaction mixture was evaporated to dryness, the residue diluted with water and acidified with aqueous 2N HCI solution. The mixture was extracted with DCM, the organic extract separated using a hydrophobic frit and evaporated to dryness to give the title compound. MS calcd for (C27H24N6O3+ H)+: 481 MS found (electrospray): (M+H)+ = 481
Example 71
3-[[(4-Chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 W-pyrazole-4-ca
Intermediate 135 (55 mg) was dissolved in tetrahydrofuran (0.7 ml.) and ethanol (0.7 ml.) and treated with sodium hydroxide solution (2N, 0.4 ml_). The reaction mixture was stirred at room temperature for 18 h. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic fractions were dried using a hydrophobic frit and evaporated to dryness to give the title compound.
MS calcd for (C26H21CIN6O3+ H)+: 501/503 MS found (electrospray): (M+H)+ = 501/503
Example 72
3-[[(2,4-Dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 136 (65 mg) was dissolved in tetrahydrofuran (1.5 ml.) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature for 40 h. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic fractions were dried using a hydrophobic frit and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H20CI2N6O3+ H)+: 535/537
MS found (electrospray): (M+H)+ = 535/537
1H NMR (DMSO 1 H, temperature 378K) δ 9.04 (1 H, m), 8.73 (1 H1 s), 8.53 (1 H, m), 8.15 (2H, d), 7.90 (2H, d), 7.45 (1H, br), 7.41 (1 H, s), 7.28 (1 H, br m), 7.19 (1 H,s), 7.02 (1 H, m), 4.73 (1H, br), 1.29 (6H, d) carboxylic acid proton not seen.
Example 73
3-((1-Methylethyl){[4-(trifluoromethyl)phenyl]carbonyl}amino)-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 W-
Intermediate 137 (125 mg) was dissolved in tetrahydrofuran (1.5 ml.) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature for 40 h. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic fractions were dried using a hydrophobic frit and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound.
MS calcd for (C27H2IF3N6O3+ H)+: 535 MS found (electrospray): (M+H)+ = 535
Example 74
3-[[(3,4-Dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 138 (104 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature for 40 h. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic fractions were dried using a hydrophobic frit and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound.
MS calcd for (C26H20CI2N6O3+ H)+: 535/537 MS found (electrospray): (M+H)+ = 535/537
Example 75
1-[3-Chloro-4-(2-furanyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro-
2W-pyran-4-yl)amino]-1 AY-pyrazole-4-carboxylic acid
Intermediate 141 (132 mg) was dissolved in THF (6 mL) and ethanol (3 ml_). Sodium hydroxide solution (2N, 1.5 mL) was added and the mixture was left to stand at room temperature for 3 days. The ethanol and THF were removed in vacuo, the residue partitioned between aqueous 1 M HCI and DCM. A solid precipitated out and this was filtered off and dried in a vacuum oven at 40°C to give the title compound. MS calcd for (C27H30CIN3O5+ H)+: 512/514 MS found (electrospray): (M+H)+ = 512/514
Example 76 1 -[3-Chloro-4-(3,5-dimethyl-4-isoxazolyl)phenyl]-3-[[(frans-4-methylcyclohexyl) carbonylKtetrahydro^H-pyran^-yOaminol-IH-pyrazole^-carboxylic acid
Intermediate 142 (240 mg) was dissolved in THF (12 mL) and ethanol (6 mL). Sodium hydroxide solution (2N, 3 mL) was added and the mixture was left to stand at room temperature for 3 days. The ethanol and THF were removed in vacuo, the residue partitioned between aqueous 1 M HCI and DCM, passed through a hydrophobic frit and the organic layer evaporated in vacuo. The crude product was purified by MDAP HPLC and then further purified using the following system:
Column: Zorbax SB Phenyl 150x21.2 mm ID 7 um, Eluent: 0.1 %TFA/water and 0.05%TFA/acetonitrile,
Gradient: 40-70% in 25 minutes at 20 mL/min, 30 minute cycle time.
This gave the title compound.
MS calcd for (C28H33CIN4O5+ H)+: 541/543
MS found (electrospray): (M+H)+ = 541/543
Example 77
3-([(frans-4-Methylcyclohexyl)carbonyl]{2-(methyloxy)-1-[(methyloxy)methyl] ethyl}amino)-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 146 (120.2 mg) was dissolved in THF (6 mL) and methanol (3 ml_). Sodium hydroxide solution (2N, 1.5 mL) was added and the mixture was left to stand at room temperature for overnight. The methanol and THF were removed in vacuo, the residue partitioned between aqueous 1 M HCI and DCM, passed through a hydrophobic frit and the organic layer evaporated in vacuo. The crude product was purified by MDAP HPLC to give title compound.
MS calcd for (C29H34N6O5+ H)+: 547 MS found (electrospray): (M+H)+ = 547
Example 78 1 -(4-Furo[3,2-/)]pyridin-2-ylphenyl)-3-([(frans-4-methylcyclohexyl)carbonyl]{2- (methyloxy)-i -[(methyloxy)methyl]ethyl}amino)-1 H-pyrazole-4-carboxylic acid
Intermediate 147 (222.2 mg) was dissolved in THF (12 mL) and methanol (6 mL). Sodium hydroxide solution (2N, 3 mL) was added and the mixture was left to stand at room temperature overnight. The methanol and THF were removed in vacuo, the residue partitioned between aqueous 1 M HCI and DCM1 passed through a hydrophobic frit and the organic layer evaporated in vacuo. The crude product was purified by MDAP HPLC to give title compound. MS calcd for (C29H34N6O5+ H)+: 547 MS found (electrospray): (M+H)+ = 547
1 H NMR (CDCI3): δ 8.62 (1 H, s), 8.59 (1 H, d), 8.08 (2H, d), 7.89-7.83 (3H, m), 7.37 (1 H, s), 7.29 (1 H, dd), 5.09 (1 H, quintet), 3.90-3.19 (1OH, m), 2.13 (1 H, m), 1.88-1.20 (8H, m), 0.78 (3H, d), 0.76-0.58 (1 H, m). Carboxylic acid proton is assumed to be exchanged with moisture in the solvent.
Example 79
1-t4-(1-Benzofuran-6-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 W-pyrazole-4-carboxylic acid
To a stirred solution of Intermediate 149 (0.12 g) in THF (2 ml_) and ethanol (2 mL) was added sodium hydroxide (2N, 1 mL). The resultant pale yellow solution was stirred at room temperature for 0.5 h and was then allowed to stand at room temperature overnight. The reaction mixture was evaporated to dryness and the residue was partitioned between 0.5M hydrochloric acid and dichloromethane. The organic phase was separated using a hydrophobic frit and was then evaporated. The crude product was then purified by MDAP
HPLC to give the title compound.
MS calcd for (C3IH33CIN3O5 + H)+: 528
MS found (electrospray): (M+H)+ = 528
Example 80
1 -[4-(1 -Benzofuran-S-yOphenyll-S-IKfrans^-methylcyclohexyOcarbonyl] (tetrahydro-
2H-pyran-4-yl)amino]-1 W-pyrazole-4-carboxylic acid
To a stirred solution of Intermediate 150 (0.18 g) in ethanol (2 mL) and THF (2 mL) was added sodium hydroxide (2N, 1 mL) and the resultant mixture was allowed to stir for 1 h and was then allowed to stand for 2 days. The reaction mixture was concentrated to a volume of about 1 mL to give a white suspension. DCM and 2N HCI were added with stirring and then the solid was filtered off and dried under suction to give title compound. MS calcd for (C31H33N3O5 + H)+: 528 MS found (electrospray): (M+H)+ = 528
Example 81
1 -[4-(1 -Benzof uran-6-yl)-3-chlorophenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](tetrahydro-2W-pyran-4-yl)amino]-1W-pyrazole-4-carboxylic acid
To a stirred solution of Intermediate 151 (0.64 g) in THF (5 ml_) and ethanol (5 mL) was added sodium hydroxide (2N, 2.5 mL). The resultant pale solution was stirred at room temperature for 0.5 h and was then allowed to stand at room temperature overnight. The reaction mixture was evaporated to dryness and the residue was partitioned between 0.5M hydrochloric acid and dichloromethane. The organic phase was separated using a hydrophobic frit and was then evaporated. The crude product was then purified by MDAP HPLC. Trituration with DCM and acetonitrile gave the title compound. MS calcd for (C3IH32CIN3O5 + H)+: 562/564 MS found (electrospray): (M+H)+ = 562/564
Example 82 i^-Furop.S-blpyridin-S-ylphenyO-S-IKfrans^-methylcyclohexylJcarbonyl] (tetrahydro-2H-pyran-4-yl)amino]-1 W-pyrazole-4-carboxylic acid
To a stirred solution of Intermediate 153 (0.10 g) in THF (2 mL) and ethanol (2 mL) was added sodium hydroxide (2N, 1 mL). The resultant pale yellow solution was stirred at room temperature for 0.5 h and was then allowed to stand at room temperature overnight. The reaction mixture was evaporated to dryness and the residue was triturated with a mixture of water and dichloromethane. The solid was filtered off and dried in vacuo at 450C to give the title compound.
MS calcd for (C30H32N4O5 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Example 83
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 H-pyrrolo[3,2- jb]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Copper iodide (4 mg) and dichlorobis (triphenylphosphine) palladium (16 mg) were added to a solution of Intermediate 46 (200 mg) and ethyl (2-bromo-3-pyridinyl) carbamate8 (232 mg) in triethylamine (2 ml.) and DMF (4 mL). This mixture was stirred at room temperature and then heated in a Reactivial at 7O0C for 5 h. On cooling the solvent was evaporated, the residue dissolved in EtOAc (40 mL), washed with water (2 x 20 mL), dried (Na2SO4) and evaporated. This was purified by SPE chromatography, eluting with cyclohexane then cyclohexane/EtOAc (gradient 9:1 to 1:9) then EtOAc. Sodium ethoxide in ethanol (21 wt %, 0.5 mL) was added to a solution of this product in EtOH (3 mL). The mixture was heated to reflux under nitrogen for 2 h. The reaction was allowed to cool and left to stir at room temperature for 48 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 1g OASIS cartridge, eluted with water (3 x column volumes) then MeOH (3 x column volumes) Appropriate MeOH fractions were combined and evaporated. Further purification was by MDAP HPLC. The sample obtained was dissolved in MeOH (2 mL) and treated with lithium hydroxide solution (2N, 0.5 mL), and stirred at room temperauture for 3 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 1g OASIS cartridge, using water (3 x column volumes) then MeOH (3 x column volumes) as eluent. Appropriate MeOH fractions combined and evaporated to give the title compound.
MS calcd for (C28H3IN5O3 + H)+: 486
MS found (electrospray): (M+H)+ = 486
Ref 7: Chem. Pharm. Bull 35 (5) 1823-8, 1987.
Example 84
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 H-pyrrolo[2,3- b]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1 mL) was added to a solution of Intermediate 154 (69 mg) in THF (1 mL) and EtOH (1 mL) and the mixture stirred at room temperature for 24 h. The
solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 1g OASIS cartridge and eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated. Further purification was by MDAP HPLC to give the title compound. MS calcd for (C28H3I N5O3 + H)+: 486 MS found (electrospray): (M+H)+ = 486
Example 85
1 -[6-(1 -Benzof uran-2-yl)-3-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1H-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1.5 mL) was added to a solution of Intermediate 162 (196 mg) in THF (1.5 mL) and EtOH (1.5 mL) and the mixture was stirred at room temperature for
24 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 6g OASIS cartridge and eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated to give the title compound.
MS calcd for (C28H30N4O4 + H)+: 487
MS found (electrospray): (M+H)+ = 487
Example 86
1-[6-(1H-lndol-2-yl)-3-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N1 1 mL) was added to a solution of Intermediate 160 (147 mg) in THF (1.5 mL) and EtOH (1.5 mL) and the mixture stirred at room temperature for 24 h. A further 0.5 mL of 2N lithium hydroxide solution was added and stirring continued for a further 4 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 6g OASIS cartridge and eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated to give the title compound.
MS calcd for (C28H3IN5O3 + H)+: 486 MS found (electrospray): (M+H)+ = 486
Example 87
1 -[5-(1 -Benzofuran^-yO^-pyridinyll-S-t^frans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1.5 mL) was added to a solution of Intermediate 162 (231 mg) in THF (1.5 mL) and EtOH (1.5 mL) and the mixture stirred at room temperature for 48 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 6g OASIS cartridge and eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated to give the title compound. MS calcd for (C28H30N4O4 + H)+: 487 MS found (electrospray): (M+H)+ = 487
Example 88
1 -[5-(1 H-lndol-Z-yO^-pyridinylJ-S-IKfrans^-methylcyclohexyOcarbonyllti - methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1 mL) was added to a solution of Intermediate 164 (58 mg) in
THF (1 mL) and EtOH (1 mL) and the mixture was stirred at room temperature for 24 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 6g OASIS cartridge and eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated to give the title compound. MS calcd for (C28H31N5O3 + H)+: 486 MS found (electrospray): (M+H)+ = 486
Example 89
1 -[6-(1 -Benzofuran^-yO-a-pyridazinylJ-a-IKfrans-A-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1.5 mL) was added to a solution of Intermediate 165 (97 mg) in THF (1.5 mL) and EtOH (1.5 mL) and the mixture was stirred at room temperature for 24 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to an OASIS cartridge eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated. Further purification was by MDAP HPLC to give the title compound. MS calcd for (C27H29N5O4 + H)+: 488 MS found (electrospray): (M+H)+ = 488
Example 90
1-[4-(7-Aminopyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4-methyl cyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1 mL) was added to a solution of Intermediate 166 (30 mg) in THF (1.5 mL) and EtOH (1.5mL) and the mixture was stirred at room temperature for 24 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 1g OASIS cartridge and eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated to give the title compound. MS calcd for (C27H3iN7O3 + H)+: 502 MS found (electrospray): (M+H)+ = 502
1H NMR (d6-DMSO) δ 12.4 (1H, br. s), 9.12 (1H, s), 8.3-8.4 (2H, br), 8.25 (2H, d), 8.1 (1 H, br.s), 8.06 (2H, d), 7.01 (1 H, s), 6.2 (1 H, d), 4.75 (1 H, m), 2.01-1.89 (1 H, m), 1.73-1.41 (5H, m) 1.31-1.10 (5H, m), 0.89 (3H, br. d) 0.74 (3H, d), 0.70-0.48 (2H, m)
Example 91
S-t^traπs-A-MethylcyclohexyOcarbonylKI-methylethyOaminol-i-^β- methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1 mL) was added to a solution of Intermediate 167 (156 mg) in THF (2 mL) and EtOH (2 mL) and the mixture was stirred at room temperature for 24 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 1g OASIS cartridge eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated to give the title compound. MS calcd for (C28H32N6O3 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 92
3-[[(fraπs-4-Ethylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1 mL) was added to a solution of Intermediate 168 (47 mg) in THF (1.5 mL) and EtOH (1.5 mL) and the mixture was stirred at room temperature for 5 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. This was extracted with EtOAc (3 x 20 mL), dried (Na2SO4) and evaporated. Purification by MDAP HPLC gave the title compound. MS calcd for (C28H32N6O3+ H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 93
3-((1-Methylethyl){[frans-4-(trifluoromethyl)cyclohexyl]carbonyl}amino)-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1 mL) was added to a solution of Intermediate 155 (32 mg) in THF (1.5 mL) and EtOH (1.5 mL) and the mixture was stirred at room temperature for 5 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. This was extracted with EtOAc (3 x 10 mL), dried (Na2SO4) and evaporated. Purification by MDAP HPLC gave the title compound. MS calcd for (C27H27F3N6O3+ H)+: 541 MS found (electrospray): (M+H)+ = 541
Example 94
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 W-pyrrolo[2,3- Jb]pyridin-2-yl)phenyl]-1 W-pyrazole-4-carboxylic acid
Lithium hydroxide solution (2N, 1 mL) was added to a solution of Intermediate 154 (69 mg) in THF (1 mL) / EtOH (1 mL) and the mixture stirred at room temperature for 24 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N hydrochloric acid. The resulting suspension was applied to a 1 g OASIS cartridge and eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated. Further purification by MDAP HPLC gave the title compound. MS calcd for (C28H3IN5O3 + H)+: 486 MS found (electrospray): (M+H)+ = 486
Example 95
1 -[4-(6-Amino-1 -benzofuran^-yOphenylJ-S-IKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1H-pyrazole-4-carboxylic acid
To a solution of Intermediate 247 (121 mg) in THF (3 mL) and ethanol (3 mL) was added 2M aqueous sodium hydroxide solution (1.5 mL). The mixture was left to stand at room temperature for 24 h and then evaporated to dryness under reduced pressure. The residue was dissolved in water (10 mL) and treated with 2M hydrochloric acid to give a gum. The supernatant was decanted off and the residue was dissolved in a minimum of THF and purified by ISCO Companion silica chromatography eluting with dichloromethane/methanol (gradient of 0-25%) to give the title compound. MS calcd for (C29H32N4O4 + H)+: 501 MS found (electrospray): (M+H)+ = 501
1H NMR (MeOD) δ 8.88 (1 H, s), 7.96 (2H, d), 7.89 (2H, d), 7.33 (1 H, d), 7.11 (1 H, s), 6.88 (1 H, d), 6.70 (1 H, dd), 4.83 (1 H, m), 2.07 (1 H, m), 1.78 (2H, m), 1.70-1.54 (3H, m), 1.41-1.30 (2H, m), 1.30 (3H, d), 1.02 (3H, d), 0.81 (3H1 d), 0.77-0.58 (2H, m), carboxylic acid proton and amine protons not seen.
Example 96
1 -[4-(4-Amino-1.S-benzoxazol^-yOphenyll-S-lKfraπs^-methylcyclohexyOcarbonylHI - methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
A solution of Intermediate 242 (517.8 mg) in THF/ethanol (1/1 , 20 mL) was stirred at 210C and treated with 2M lithium iodide solution (10 mL). The resulting suspension was stirred overnight, and then concentrated. The residue was partitioned between EtOAc/water and the aqueous phase separated and acidified to pH2 with 2M HCI and then extracted with EtOAc (x3). The combined extracts were washed with brine, dried (Na2SO4) and concentrated. This was purified by ISCO Companion silica chromatography eluting with a gradient of methanol in DCM, followed by trituration with diethyl ether to give the title compound.
MS calcd for (C28H31N5O4 H-H)+ :502 MS found (electrospray): (M +H)+ = 502 1H NMR (MeOD) δ 9.00 (1 H, s), 8.35 ( 2H, d), 8.06 (2H, d), 7.13 (1 H, tr), 6.91 (1 H, d), 6.64 (1 H1 d), 4.90-4.82 (2H, m), 2.09 (1 H, m), 1.80 (2H, m), 1.71-1.54 (3H, m), 1.28 (4H, m), 1.03-
0.90 (3H1 m), 0.80-0.75 (3H, d), 0.75-0.55 (2H, m) carboxylic acid proton and amine protons not seen.
Example 97 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 ,8-naphthyridin-2- yl)phenyl]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 243 (129 mg) in THF/ethanol (1/1 , 4 mL) was stirred at 210C and treated with 2N sodium hydroxide solution (0.86 mL) and stirred overnight. The reaction was evaporated in vacuo and the residue partitioned between EtOAc/water and acidified to pH 3 with 2N HCI. The phases were separated and the aqueous phase extracted with more EtOAc (x2). The combined organics were washed with brine, dried (Na2SO4) and concentrated. This was purified by ISCO Companion silica chromatography eluting with a gradient of methanol in dichloromethane to give the title compound. MS calcd for (C29H3IN5O3 H-H)+ :498 MS found (electrospray): (M +H)+ = 498
Example 98
S-Hftrans^-MethylcyclohexyOcarbonylHtetrahydro^Hφyran^-yOaminol-i-^-p- methyl-1H-indol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
To Intermediate 184 was added 2M lithium hydroxide (1 mL), THF (1 mL) and EtOH (1 mL). The reaction was stirred at room temperature for 24 h. The reaction was neutralised with 2M HCI, partitioned with DCM and the organics separated and concentrated to give the title compound.
MS calcd for (C32H36N4O4 + H)+: 541 MS found (electrospray): (M+H)+ = 541
Example 99
1 -[4-(8-Aminoimidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[( trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 249 (130 mg) in ethanol (5 mL) and 2M lithium hydroxide (1 mL) was stirred at room temperature for 3 days. The reaction was evaporated to dryness, acidified to approximately pH 6 with 2M HCI, diluted with water and extracted twice with EtOAc. The combined organics were dried using a hydrophobic frit and evaporated to dryness to give the title compound. MS calcd for (C28H32N6O3 + H)+: 501 MS found (electrospray): (M+H)+ = 501
1H NMR (MeOD) δ 8.84 (1 H, s), 8.02 (1 H, s), 7.96 (2H, d), 7.82 (2H, d), 7.68 (1 H1 dd), 6.63 (1 H, t), 6.40 (1 H, dd), 4.77-4.88 (1 H, septet), 2.03-2.14 (1 H, m), 1.75-1.84 (2H, m), 1.50-1.64 (3H, m), 1.18-1.40 (5H, m), 0.99 (3H, d), 0.72 (3H1 d), 0.51-0.73 (2H, m). Carboxylic acid and amine protons not seen.
Example 100
1-[4-(4-Aminothieno[3,2-d]pyrimidin-6-yl)phenyl]-3-[[(trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 251 (72 mg) was dissolved in THF (0.4 mL) and methanol (0.4 mL). 2N sodium hydroxide solution (0.4 mL) was added and the mixture stirred at room temperature for 24 h. The methanol and THF were evaporated under vacuum, then the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C27H30N6O3S + H)+: 519 MS found (electrospray): (M+H)+ = 519
Example 101
1-{4-[4-(Methylamino)thieno[3,2-d]pyrimidin-6-yl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 253 (169 mg) was dissolved in THF (0.8 mL) and methanol (0.8 ml_). 2N sodium hydroxide solution (0.8 mL) was added and the mixture stirred at room temperature for 24 h. The methanol and THF were evaporated under vacuum, then the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound.
MS calcd for (C28H32N6O3S + H)+: 533 MS found (electrospray): (M+H)+ = 533
Example 102
1-(4-Pyrazolo[1,5-a]pyrimidin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[frans-4- (trifluoromethyl)cyclohexy arboxylic acid
Intermediate 256 (141 mg) was dissolved in THF (0.6 mL) and methanol (0.6 mL). 2N sodium hydroxide solution (0.6 mL) was added and the mixture stirred at room temperature for 24 h. The methanol and THF were evaporated under vacuum, then the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by ISCO Companion eluting with a gradient of methanol in DCM to give the title compound. MS calcd for (C29H29F3N6O4+ H)+: 583 MS found (electrospray): (M+H)+ = 583
Example 103 1-(4-Furo[3,2-to]pyridin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[fraπs-4- (trifluoromethyl)cyclohexyl]carbonyl}amino)-1H-pyrazole-4-carboxylic acid
Intermediate 257 (52 mg) was dissolved in THF (0.3 ml_) and methanol (0.3 mL). 2N sodium hydroxide solution (0.3 mL) was added and the mixture stirred at room temperature for 24 h. 2N sodium hydroxide solution (0.4 mL), THF (0.4 mL) and methanol (0.4mL) were added, and the reaction was stirred at room temperature for 1 more day. The methanol and THF were evaporated under vacuum, then the residue was partitioned between DCM and 2N HCI solution. The DCM layer was separated using a hydrophobic frit and concentrated. The residue was purified by NH2 SPE ion exchange cartridge, eluting with 10% 2M HCI in methanol. This was purified further by ISCO Companion silica chromatography eluting with a gradient of methanol in DCM, followed by reverse phase chromatography using an ABZ column÷ (100X21.2mm, ID 5um), 15 mL/min, 20 minute cycle time, eluting with a gradient of 0.1% TFA/water (A) in 0.05% TFA/acetonitrile (B) (gradient of 3-70% A: B) ) over 25 minutes (multiple injections). This gave the title compound. MS calcd for (C30H29F3N4O5 + H)+: 583 MS found (electrospray): (M+H)+ = 583
Example 104
1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-((1 -methylethyl){[frans-4- acid
A solution of Intermediate 265 (163 mg) and 2M sodium hydroxide solution (1.5 mL) in THF (1.5 mL) and MeOH (1.5 mL) was stirred overnight at room temperature. The solvent was removed under vacuum then the residue was partitioned between 2M HCI and DCM containing some methanol to aid solubility. The organic phase was separated, evaporated and purified by ISCO Companion silica chromatography eluting with a gradient of 0-30% methanol in DCM. Product fractions were evaporated and freeze dried from dioxane to give the title compound.
MS calcd for (C28H28F3N5O3 + H)+: 540 MS found (electrospray): (M+H)+ = 540
Example 105
1 -(4-lmidazo[2,1 -b][1 ,3]thiazol-6-ylphenyl)-3-((1 -methylethyl){[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1AV-pyrazole-4-carboxylic acid
Prepared from Intermediate 266 (165mg) using a similar method to that described for Example 104 to give the title compound. MS calcd for (C26H26F3N5O3S + H)+: 545 MS found (electrospray): (M+H)+ = 545
Example 106
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-((1-methylethyl){[frans-4- (trifluoromethyl)cyclohex rboxylic acid
A solution of Intermediate 239 (45 mg) and 2M sodium hydroxide solution (0.2 mL) in THF (0.3 mL) and MeOH (0.2 mL) was stirred overnight at room temperature. The solvent was removed under vacuum then the residue was partitioned between 2M HCI and DCM. The organic phase was separated, evaporated and freeze dried from dioxane to give the title compound. MS calcd for (C28H27F3N4O4 + H)+: 541 MS found (electrospray): (M+H)+ = 541
Example 107
1 -(4-lmidazo[1 ^-blpyridazin^-ylphenyO-S-I^frans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 258 (108 mg) and 2M sodium hydroxide solution (0.5 mL) in THF (0.75 mL) and MeOH (0.75 mL) was stirred overnight at room temperature. The solvent was removed under vacuum then the residue was partitioned between 2M HCI and EtOAc. The organic phase was separated and purified by NH2 SPE ion exchange chromatography eluting with MeOH then 10% 2M HCI in MeOH. Combined product fractions were evaporated and freeze dried from dioxan to give the title compound. MS calcd for (C27H30N6O3 + H)+: 487 MS found (electrospray): (M+H)+ = 487
Example 108
1 -(4-lmidazo[1 ,2-a]pyrazin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
A solution of Intermediate 259 (56 mg) and 2M sodium hydroxide solution (0.27 mL) in THF (0.5 mL) and MeOH (0.5 mL) was stirred overnight at room temperature. The solvent was removed under vacuum then the residue was partitioned between 2M HCI and chloroform containing some methanol to aid solubility. The organic phase was separated by hydrophobic frit and evaporated to give the title compound. MS calcd for (C27H30N6O3 + H)+: 487 MS found (electrospray): (M+H)+ = 487
Example 109
1-{4-[8-(Hydroxymethyl)imidazo[1,2-a]pyridin-2-yl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 261 (100 mg) and 2M sodium hydroxide solution (0.5 mL) in THF (0.5 mL) and MeOH (0.5 mL) was stirred overnight at room temperature. The solvent was removed under vacuum then the residue was partitioned between 2M HCI and DCM containing some methanol to aid solubility. The organic phase was separated and purified by NH2 SPE ion exchange chromatography. Combined product fractions were evaporated and freeze dried from dioxan to give the title compound. MS calcd for (C29H33N5O4 + H)+: 516 MS found (electrospray): (M+H)+ = 516
Example 110
1-{4-[8-(Aminomethyl)imidazo[1,2-a]pyridin-2-yl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
A solution of Intermediate 262 (30.5 mg) and 2M sodium hydroxide solution (0.15 mL) in THF (0.5 mL) and MeOH (0.5 mL) was stirred overnight at room temperature. More 2M NaOH was added (0.05 mL) and stirring continued over one more night. The mixture was neutralised with 2M HCI (0.2 mL) and solvent was removed under vacuum. The residue was dissolved in aqueous DMSO and purified by Oasis reverse phase chromatography column, eluting with water then methanol. Combined product fractions were evaporated and the residue was freeze dried from dioxan containing some methanol to aid solubility to give the title compound.
MS calcd for (C29H34N6O3 + H)+: 515 MS found (electrospray): (M+H)+ = 515
Example 111
1 -(4-lmidazo[1 ^-alpyridin-β-ylphenyO-S-lKfrans^-methylcyclohexyOcarbonylHI - methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Intermediate 235 (55 mg) was dissolved in ethanol (0.5 mL) and THF (0.5 ml.) and to this solution was added 2N sodium hydroxide (0.25 mL). The resultant mixture was allowed to stand for 2 days. The reaction mixture was evaporated to dryness and the residue was purified using MDAP HPLC. Appropriate fractions were combined and evaporated to give the title compound.
MS calcd for (C28H3IN5O3 + H)+: 486
MS found (electrospray): (M+H)+ = 486
Example 112
1-(4-Furo[3,2-b]pyridin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro- 2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid
To a mixture of Intermediate 238 (0.12 g) and Intermediate 181 (0.28 g) in DME (20 mL) containing 2N sodium carbonate (10 mL) was added tetrakistriphenyl phosphine palladium (0) (0.036g) and the resultant mixture was heated to 1000C for 24 h. The mixture was allowed to cool to room temperature and was then partitioned between EtOAc and saturated aqueous sodium bicarbonate solution. The aqueous phase was separated and was extracted twice with EtOAc. The extracts and organic phase were combined then dried and finally evaporated.
The residue was dissolved in ethanol (10 mL) and THF (10 mL) and to this solution was added 2N sodium hydroxide (5 mL). The resultant solution was stirred at room temperature for 0.5 h and was then allowed to stand for 2 days. The reaction mixture was evaporated to dryness and the residue was partitioned between water and DCM. The aqueous phase was adjusted to ~pH7 using 2N HCI and then the layers were stirred. The organic phase was separated and then evaporated to give crude product. This material was partially purified using MDAP HPLC. Appropriate fractions were combined and evaporated and the residue was re-purified using MDAP HPLC. Appropriate fractions were again combined and evaporated and the residue was purified by reverse phase HPLC (Column: Supleco ABZ++, 100X21.2mm, 5μm. Flow Rate: 20ml/min using a two-solvent gradient elution with (A) water
containing formic acid (0.1%) and (B) acetonitrile-water (95:5 v/v) containing formic acid (0.05%) as the eluents) to give the title compound. MS calcd for (C30H32N4O5 + H)+: 529 MS found (electrospray): (M+H)+ = 529
Example 113
S-t^frans^-MethylcyclohexyOcarbony^tetrahydro-ΣH-pyran-A-yOaminol-i-^^e- quinazolinyl)phenyl]-1 H-pyrazole-4-carboxylic acid
Intermediate 264 (0.43 g) was dissolved in ethanol (10 ml.) and THF (10 mL) and to this solution was added 2N sodium hydroxide (5 mL). The resultant solution was stirred at room temperature for 0.5 h and was then allowed to stand for 2 days. The reaction mixture was evaporated to dryness and the residue was partitioned between water and DCM. The aqueous phase was adjusted to pH3 using 2N HCI and the layers were stirred. The organic phase was separated and evaporated to give crude product. This material was partially purified using MDAP HPLC. Appropriate fractions were combined and evaporated and the residue was re-purified using MDAP HPLC. Appropriate fractions were again combined and evaporated and the residue was crystallised from acetonitrile to give the title compound. MS calcd for (C31H33N5O4 + H)+: 540 MS found (electrospray): (M+H)+ = 540
Example 114
1-(4-Furo[3,2-/j]pyridin-5-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 /Y-pyrazole-4-carboxylic acid
Intermediate 234 (0.61 g) was dissolved in ethanol (2 mL) and THF (2 mL) and to this solution was added 2N sodium hydroxide (1 mL). The resultant solution was stirred at room temperature for 0.5 h and was then allowed to stand for 20 h. The reaction mixture was evaporated to dryness and the residue was purified using MDAP HPLC. Appropriate fractions were combined and evaporated and the residue was then freeze-dried from dioxan to give the title compound.
MS calcd for (C28H30N4O4 + H)+: 487
MS found (electrospray): (M+H)+ = 487
1H NMR (DMSO) δ 12.88 (1H, v.b.s), 9.19 (1 H, s), 8.38 (1H, br.d), 8.30 (2H, 1/2AA1BB'), 8.16
(1 H, d), 8.05 (2H, 1/2AA1BB'), 8.00 (1 H, d), 7.23 (1 H, dd), 4.75 (1 H, m), 1.96 (1 H, br.t.t),
1.72-0.48 (18H, 7 x m+d).
Example 115
1 -[4-(6-Aminoimidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[( frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Intermediate 229 (148 mg) was dissolved in THF (1.5 ml.) and ethanol (1.5 ml_). Lithium hydroxide solution (2M, 1.4 ml.) was added and the mixture stirred at room temperature for 18 h. The ethanol and THF were removed in vacuo, the residue diluted with water and extracted with ethyl acetate (x 2). The aqueous phase was acidified to pH 3 with 2M HCI solution and extracted with ethyl acetate (x 3). The combined organic phases were dried (MgSO4) and concentrated. This was purified by chromatography on silica gel using a DCM/ MeOH gradient to give the title compound. MS calcd for (C28H32N6O3+H)+ : 501 MS found (electrospray): (M+H)+ = 501
Example 116
1 -[4-(6-Amino-1.S-benzoxazol^-ylJphenyll-S-IKfraπs^-methylcyclohexylJcarbonylHI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 232 (320 mg) was dissolved in THF (2 ml_) and ethanol (2 ml_). Lithium hydroxide solution (2M, 3 mL) was added and the mixture stirred at room temperature for 18 h. The ethanol and THF were removed in vacuo, the residue diluted with water and extracted with ethyl acetate (x 2). The aqueous phase was acidified to pH3 with 2M HCI solution and extracted with ethyl acetate (x 3). The combined organic phases were dried
(MgSO4) and concentrated. This was purified by chromatography on silica gel using a DCM/ MeOH gradient to give the title compound. MS calcd for (C28H31 N5O4+H)+ : 502 MS found (electrospray): (M+H)+ = 502
Example 117
3-[(Cyclohexylacetyl)(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-
1W-pyrazole-4-carboxylic acid
A solution of Intermediate 226 (79 mg) in EtOH (1 ml.) and THF (1 mL) was treated with 2N lithium hydroxide solution (1 mL). This was left to stir at RT for 24 h. The solvent was then evaporated. The residue was acidified to pH 1.0 with 2N HCI and was extracted with EtOAc (3 x 10 mL), dried (Na2SO4) and evaporated. The material was purified by MDAP HPLC to give the title compound. MS calcd for (C27H30N6O3+ H)+: 487 MS found (electrospray): (M+H)+ = 487
Example 118
S-^CyclopentylacetyOti-methylethyOaminol-i^-pyrazoloti.S-alpyrimidin^-ylphenyl)- IH-pyrazole-4-carboxylic acid
2N Lithium hydroxide (1 mL) was added to a solution of Intermediate 225 (51 mg) in EtOH (1 mL) and THF (1 mL). This was stirred at RT for 24 h. The solvent was evaporated and acidified to pH 1.0 with 2N HCI. This was extracted with EtOAc (3 x 10 mL), dried (Na2SO4) and evaporated. The material was purified by MDAP HPLC to give the title compound. MS calcd for (C26H28N6O3+ H)+: 473 MS found (electrospray): (M+H)+ = 473
Example 119
1 -[4-(4-Amino-1 -benzofuran^-yOphenyll-a-IKfrans^-methylcyclohexylJcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
2N Lithium hydroxide (1 ml_) was added to a solution of Intermediate 244 (151 mg) in EtOH (2 ml_) and THF (1 ml_). This was stirred at RT for 48 h. The solvent was evaporated and acidified to pH 1.0 with 2N HCI. The resulting suspension was applied to a 6g OASIS HLB cartridge. This was eluted with water (3 x column volumes) then MeOH (3 x column volumes). The appropriate MeOH fractions were combined and evaporated to give the title compound. MS calcd for (C29H32N4O4+ H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 120
1 -[4-(7-Amino-1 -benzofuran^-yOphenyll-S-IKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 224 (53 mg) in THF (1.5 mL) and EtOH (1.5 mL) was treated with 2N lithium hydroxide solution (0.5 mL). The resulting suspension was stirred at RT for 24 h and was then evaporated and acidified with 2N HCI. The resulting solid was filtered off and air dried. The material was re-dissolved in EtOH (2 mL) and treated with 2N lithium hydroxide solution (1 mL) and left to stir for a further 24 h. The solvent was evaporated and the residue acidified to pH 1.0 with 2N HCI. The resulting suspension was applied to a 1 g OASIS HLB cartridge. This was eluted with water (3 x column volumes) then MeOH (3 x column volumes). Appropriate MeOH fractions were combined and evaporated. The material was purified further by MDAP HPLC to give the title compound. MS calcd for (C29H32N4O4+ H)+: 501 MS found (electrospray): (M+H)+ = 501 1H NMR (DMSO-de) δ 9.19 (1 H, s), 8.04-8.14 (4H, dd), 7.39 (1 H, s), 6.95 (1 H, t), 6.82 (1 H, dm), 6.57 (1H, dm), 5.46 (2H, s br), 4.74 (1H, m), 1.96 (1H, m), 1.1-1.7 (1OH, m), 0.89 (3H, m), 0.74 (3H, m), 0.5-0.7 (2H, m), carboxylic acid proton not seen.
Example 121
1 -[4-(7-Aminopyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-((1 -methylethyl){[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1H-pyrazole-4-carboxylic acid
A solution of Intermediate 263 (30 mg) and 2M sodium hydroxide solution (0.2 ml_) in THF (0.2 mL) and MeOH (0.2 mL) was stirred overnight at room temperature. Solvent was removed under vacuum then the residue was partitioned between 2M HCI and DCM. The organic phase was separated and purified by NH2 SPE ion exchange chromatography. Combined product fractions were evaporated and freeze dried from dioxan to give the title compound.
MS calcd for (C27H28F3N7O3 + H)+: 556
MS found (electrospray): (M+H)+ = 556
1 H NMR (CD3OD) δ 8.99 (1 H, s), 8.29 (2H, d), 8.23 (1 H, d), 8.03 (2H, d), 7.04 (1 H, s), 6.44
(1H, d), 4.88-4.81 (1H, m, partially obscured by solvent water peak), 2.19-1.99 (2H, m), 1.96- 1.82 (4H, m), 1.70-1.59 (1 H, m), 1.47-1.37 (1 H, m), 1.28 (3H, br. d), 1.15-1.03 (2H, m), 1.01 (3H, br. d). Carboxylic acid and NH2 protons are assumed to be exchanged with moisture in the solvent.
Example 122 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 ,3-oxazol-2-ylmethyl)amino]-1 -(4- pyrazolo[1,5-a]pyrimidin-2-ylphenyl)-1W-pyrazole-4-carboxylic acid
To Intermediate 119 (200 mg) was added anhydrous DMF (8 mL), and the resulting suspension was stirred at room temperature, under nitrogen for 5 minutes. Sodium hydride (60% dispersion in mineral oil) (34 mg) was then added, and the reaction mixture stirred at room temperature, under nitrogen for 25 minutes. 2-Chloromethyl oxazole (249 mg) was added, and the reaction was stirred at 5O0C, under nitrogen for 20 h. Water (2 mL) was added to quench the reaction, and the mixture was stirred for 15 minutes before the solvent was removed by evaporation. The residue was suspended in THF (2 mL) and ethanol (2 mL), and 2M lithium hydroxide solution (2 mL) was then added. The reaction mixture was
stirred at room temperature for 24 h. The mixture was neutralised using 2M HCI, and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was then concentrated by evaporation, and purified using ISCO Companion Ci8 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water ( containing 0.1% formic acid) to give the title compound. MS calcd for (C28H27N7(VH)+: 526 MS found (electrospray): (M+H)+= 526
1H NMR (DMSO- d6) δ 12.90 (1 H, br), 9.18-9.13 (2H, m), 8.56 (1 H1 dd), 8.20 (2H, d), 8.03 (1H, s), 7.97 (2H1 d), 7.34 (1H, s), 7.09-7.05 (2H, m), 4.98-4.91 (2H, br), 2.20-2.12 (1H, m), 1.74 (2H, d), 1.59 (2H, d), 1.40 (2H, q), 1.31-1.20 (1 H, m), 0.75 (3H, d), 0.66 (2H, q).
Example 123
3-{[(frans-4-Methylcyclohexyl)carbonyl][2-(phenyloxy)ethyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1H-pyrazole-4-carboxylic acid
To Intermediate 119 (200 mg) was added anhydrous DMF (8 mL), and the resulting suspension was stirred at room temperature, under nitrogen for 5 minutes. Sodium hydride (60% dispersion in mineral oil) (34 mg) was then added, and the reaction mixture stirred at room temperature, under nitrogen for 25 minutes. Beta-chlorophenetole (331 mg) was added, and the reaction was stirred at 5O0C, under nitrogen for 20 h. Water (2 mL) was added to quench the reaction, and the mixture was stirred for 15 minutes before the solvent was removed by evaporation. The residue was then suspended in THF (2 mL) and ethanol (2 mL), and 2M lithium hydroxide solution (2 mL) was then added. The reaction mixture was stirred at room temperature for 24 h, and then neutralised using 2M HCI, and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was then concentrated by evaporation, and purified using ISCO Companion C18 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water ( containing 0.1% formic acid) to give the title compound. MS calcd for (C32H32N6O4+H)+: 565 MS found (electrospray): (M+H)+= 565
Example 124
3-[[(frans-4-Methylcyclohexyl)carbonyl](1,3-thiazol-4-ylmethyl)amino]-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylic acid
To Intermediate 119 (200 mg) was added anhydrous DMF (8 ml_), and the resulting suspension was stirred at room temperature, under nitrogen for 5 minutes. Sodium hydride (60% dispersion in mineral oil) (34 mg) was then added, and the reaction mixture stirred at room temperature, under nitrogen for 25 minutes. 4-(Chloromethyl)thiazole hydrochloride (360 mg) was partitioned between sat. NaHCO3 solution and toluene (2 ml_), and the organic layer was separated and added to the reaction mixture. The reaction was then stirred at 5O0C, under nitrogen for 20 h. Water (2 mL) was added to quench the reaction, and the mixture was stirred before the solvent was removed by evaporation. The residue was then suspended in THF (2 mL) and ethanol (2 mL), and 2M lithium hydroxide solution (2 mL) was then added. The reaction mixture was stirred at room temperature for 24 h, and then neutralised using 2M HCI, and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was then concentrated by evaporation, and purified using ISCO Companion C18 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water (containing 0.1% formic acid) to give the title compound. MS calcd for (C28H27N7O3S+H)+: 542 MS found (electrospray): (M+H)+= 542
1H NMR (de-DMSO) δ 9.17-9.13 (2H, m), 8.97 (1 H1 d), 8.56 (1 H, dd), 8.20 (2H, d), 7.99 (2H, d), 7.51 (1 H, s), 7.34 (1 H, s), 7.07 (1 H, dd), 5.04-4.93 (2H, br), 2.24-2.16 (1 H, m), 1.81-1.73 (2H, br), 1.64-1.57 (2H, br), 1.42 (2H, q), 1.31-1.21 (1 H, br), 0.76 (3H, d), 0.68 (2H, q) carboxylic acid proton not seen.
Example 125
3-{[(frans-4-Methylcyclohexyl)carbonyl][(4-methyl-1,2,5-oxadia2θl-3-yl)methyl]amino}- 1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 AV-pyrazole-4-carboxylic acid
To Intermediate 119 (200 mg) was added anhydrous DMF (8 mL), and the resulting suspension was stirred at room temperature, under nitrogen for 5 minutes. Sodium hydride (60% dispersion in mineral oil) (34 mg) was then added, and the reaction mixture stirred at room temperature, under nitrogen for 25 minutes. 3-(Chloromethyl)-4-methyl-1 ,2,5-
oxadiazole (280 mg) was added to the reaction mixture. The reaction was then stirred at 5O0C, under nitrogen for 20 h. Water (2 mL) was added to quench the reaction, and the mixture was stirred for 15 minutes before the solvent was removed by evaporation. The residue was then suspended in THF (2 mL) and ethanol (2 mL), and 2M lithium hydroxide solution (2 mL) was then added. The reaction mixture was stirred at room temperature for 24 h, and then neutralised using 2M HCI, and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was then concentrated by evaporation, and purified using ISCO Companion Ci8 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water (containing 0.1 % formic acid) to give the title compound. MS calcd for (C28H28N8O4H-H)+: 541 MS found (electrospray): (M+H)+= 541
Example 126 3-{[2-(Ethyloxy)ethyl][(fraπs-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL), was added sodium hydride (60% dispersion in mineral oil) (17 mg). The reaction mixture was stirred at room temperature, under nitrogen for 15 minutes, and then 2-bromoethyl ethyl ether (162 mg) was added. The reaction mixture was then stirred at 5O0C, under nitrogen for 24 h. Water (0.5 mL) was then added to the reaction mixture, and the solvent was removed by evaporation. The residue was then suspended in THF (1 mL) and ethanol (1 mL), and 2M lithium hydroxide solution (2 mL) was added. The reaction was stirred at room temperature for 20 h, before being neutralised with 2M HCI (2 mL) and partitioned between water and dichloromethane. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was concentrated by evaporation to give a residue which was then purified by ISCO Companion C18 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water (containing 0.1% formic acid) to give the title compound.
MS calcd for (C28H32N6O4-HH)+: 517 MS found (electrospray): (M+H)+= 517
Example 127 S-^fraπs^-MethylcyclohexyOcarbonyll^-methyl-I.S-thiazoM-yOmethyllamino}^^- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 ml_), was added sodium hydride (60% dispersion in mineral oil) (17 mg). The reaction mixture was stirred at room temperature, under nitrogen for 15 minutes, and then 4-(chloromethyl)-2-methyl-1 ,3- thiazole (158 mg) was added. The reaction mixture was then stirred at 5O0C, under nitrogen for 24 h. Water (0.5 mL) was then added to the reaction mixture, and the solvent was removed by evaporation. The residue was then suspended in THF (1 mL) and ethanol (1 mL), and 2M lithium hydroxide solution (2 mL) was added . The reaction was stirred at room temperature for 20 h, before being neutralised with 2M HCI (2 mL) and partitioned between water and dichloromethane. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was concentrated by evaporation to give a residue which was then purified by ISCO Companion C18 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water (containing 0.1% formic acid) to give the title compound. MS calcd for (C29H29N7O3S+H)+: 556 MS found (electrospray): (M+H)+= 556
Example 128
3-{Butyl[(fraπs-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1,5-a]pyrimidin-2- ylphenyl)-1H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL), sodium hydride (60% dispersion in mineral oil) (17 mg) was added. The reaction mixture was stirred at room temperature, under nitrogen for 15 minutes, and then 1-iodobutane (195 mg) was added. The reaction mixture was stirred, under nitrogen, at 5O0C for 20 h, and then water (0.5 mL) was added. The mixture was stirred for 45 minutes, and then the solvent was removed by evaporation to give a residue, which was then suspended in THF (1 mL) and ethanol (1 mL). To this stirred suspension, 2M lithum hydroxide solution (1 mL) was added, and the reaction mixture was stirred at room temperature for 20 h, before being neutralised with 2M HCI (1 mL) and partitioned between water and dichloromethane. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was
then concentrated by evaporation to give a residue which was purified by MDAP HPLC to give the title compound. MS calcd for (C28H32N6(VH)+: 501 MS found (electrospray): (M+H)+= 501 1H NMR Cd6-DMSO) δ 9.18 (1H, s), 9.16 (1H, d), 8.57 (1H, dd), 8.21 (2H, d), 8.03 (2H, d), 7.35 (1 H, s), 7.08 (1 H, dd), 3.66-3.52 (2H, br), 2.10-2.12 (1 H, m), 1.73-1.65 (2H, br), 1.62- 1.54 (2H, br), 1.47-1.35 (4H1 m), 1.32-1.23 (3H, m), 0.84 (3H1 1), 0.74 (3H1 d), 0.63 (2H1 q) carboxylic acid proton not seen.
Example 129
3-{[(3,5-Dimethyl-4-isoxazolyl)methyl][(fraπs-4-methylcyclohexyl)carbonyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL), was added sodium hydride (60% dispersion in mineral oil) (17 mg). The reaction mixture was stirred at room temperature, under nitrogen for 15 minutes, and then 4-(chloromethyl)-3,5- dimethylisoxazole (154 mg) was added. The reaction mixture was then stirred at 500C1 under nitrogen for 24 h. Water (0.5 mL) was then added to the reaction mixture, and the solvent was removed by evaporation. The residue was then suspended in THF (1 mL) and ethanol (1 mL), and 2M lithium hydroxide solution (2 mL) was added. The reaction was stirred at room temperature for 20 h, before being neutralised with 2M HCI (2 mL) and partitioned between water and dichloromethane. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was concentrated by evaporation to give a residue which was then purified by ISCO Companion C18 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water (containing 0.1% formic acid) to give the title compound. MS calcd for (C3oH3iN7θ4+H)+: 554 MS found (electrospray): (M+H)+= 554
Example 130
3-{(3-Methylbutyl)[(fraπs-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL), was added sodium hydride (60% dispersion in mineral oil) (17 mg). The reaction mixture was stirred at room temperature under nitrogen for 15 minutes, and then 1-iodo-3-methylbutane (210 mg) was added. The mixture was stirred under nitrogen at 5O0C for 20 h, and then water (0.5 mL) was added and this was stirred for a further 45 minutes. The solvent was removed by evaporation and the residue was suspended in THF (1 mL) and ethanol (1 mL). To this stirred suspension, 2M lithum hydroxide solution (1 mL) was added, and this was stirred at room temperature for 20 h, before being neutralised with 2M HCI (1 mL) and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was then concentrated by evaporation to give a residue which was purified by MDAP HPLC to give the title compound. MS calcd for (C29H34N6CyI-H)+: 515 MS found (electrospray): (M+H)+= 515
1H NMR (DMSO-d6) δ 9.17-9.14 (2H, m), 8.56 (1H, dd), 8.22 (2H, d), 8.03 (2H, d), 7.34 (1 H, s), 7.07 (1H, dd), 3.70-3.52 (2H, br), 2.10-2.02 (1H, m), 1.72-1.64 (2H, m), 1.62-1.54 (3H, m), 1.37-1.30 (4H, m), 1.27-1.20 (1 H, m), 0.84 (6H, d), 0.74 (3H, d), 0.63 (2H, q), carboxylic acid proton not seen.
Example 131
3-{{[5-(1 ,1 -Dimethylethyl)-1 ,2,4-oxadiazol-3-yl]methyl}[( fraπs-4- methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H- pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL), was added sodium hydride (60% dispersion in mineral oil) (17 mg). The reaction mixture was stirred at room temperature under nitrogen for 15 minutes, and then 5-(tert-butyl)-3-(chloromethyl)- 1 ,2,4-oxadiazole (185 mg) was added. The reaction mixture was then stirred at 5O0C under nitrogen for 24 h. Water (0.5 mL) was then added to the reaction mixture, and the solvent was removed by evaporation. The residue was then suspended in THF (1 mL) and ethanol (1 mL), before being neutralised with 2M HCI (2 mL) and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit.
The organic phase was concentrated by evaporation to give a residue which was then purified by ISCO Companion Ci8 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water (containing 0.1% formic acid) to give the title compound. MS calcd for (C3IH34N8CVH)+: 583 MS found (electrospray): (M+H)+= 583
Example 132
3-{[(frans-4-Methylcyclohexyl)carbonyl][(1 -methyl-1 W-imidazol-2-yl)methyl]amino}-1 - (4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 ml_), was added sodium hydride (60% dispersion in mineral oil) (17 mg). The reaction mixture was stirred at room temperature under nitrogen for 15 minutes, and then 2-(chloromethyl)-1 -methyl-1 H- imidazole hydrochloride (177 mg) was added. The reaction mixture was then stirred at 5O0C under nitrogen for 24 h. Water (0.5 mL) was then added to the reaction mixture, and the solvent was removed by evaporation. The residue was then suspended in THF (1 mL) and ethanol (1 mL), and 2M lithium hydroxide solution (2 mL) was added. The reaction was stirred at room temperature for 20 h, before being neutralised with 2M HCI (2 mL) and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was concentrated by evaporation to give a residue which was then purified by MDAP HPLC to give the title compound. MS calcd for (C29H30N8CVH)+: 539 MS found (electrospray): (M+H)+= 539
Example 133
3-{[(5-Chloro-2-thienyl)methyl][(traπs-4-methylcyclohexyl)carbonyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
To a stirred suspension of Intermediate 119 (100 mg) in anhydrous DMF (4 mL), was added sodium hydride (60% dispersion in mineral oil) (17 mg). The reaction mixture was stirred at room temperature under nitrogen for 15 minutes, and then 2-chloro-5-
(chloromethyl)thiophene (177 mg) was added. The reaction mixture was then stirred at 5O0C under nitrogen for 24 h. Water (0.5 ml.) was then added to the reaction mixture, and the solvent was removed by evaporation. The residue was then suspended in THF (1 mL) and ethanol (1 mL), and 2M lithium hydroxide solution (2 mL) was added. The reaction was stirred at room temperature for 20 h, before being neutralised with 2M HCI (2 mL) and partitioned between water and DCM. The layers were stirred for 30 minutes, and then separated using a hydrophobic frit. The organic phase was concentrated by evaporation to give a residue which was then purified by ISCO Companion C18 chromatography eluting with a gradient of acetonitrile (containing 0.05% formic acid) in water (containing 0.1% formic acid) to give the title compound.
MS calcd for (C29H27CIN6O3S+^*: 575/577
MS found (electrospray): (M+H)+= 575/577
1H NMR (CDCI3) δ 8.73 (1 H, d), 8.60 (1 H, s), 8.51 (1 H1 d), 8.14 (2H, d), 7.80 (2H1 d), 7.06
(1 H, s), 6.88 (1 H, dd), 6.72-6.63 (2H, br), 5.10-4.89 (2H, br), 2.26-2.13 (1H, m), 1.94-1.79 (2H, m), 1.72-1.53 (4H, m), 1.41-1.28 (1 H, m), 0.77 (3H, d), 0.75-0.62 (2H, m), carboxylic acid proton not seen.
Example 134
1-[4-(7-Chloropyrazolo[1,5-a]pyridin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyOcarbonylKI -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 186 (30 mg) was dissolved in THF (1 mL) and ethanol (1 mL). Sodium hydroxide solution (2N, 0.5 mL) was added and the mixture stirred at room temperature for 18 h. The reaction mixture was acidified with 2N aqueous HCI solution and extracted with DCM. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C28H30CIN5O3 + H)+: 520/522 MS found (eiectrospray): (M+H)+ = 520/522
Example 135
1-[5-(6-chloroimidazo[1,2-a]pyridin-2-yl)-2-thienyi]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Intermediate 187 (118 mg) was dissolved in THF (2 ml.) and ethanol (2 mL). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 48 h. The ethanol and THF were removed in vacuo and the residue dissolved in water and the pH adjusted to ~2 with aqueous 2N HCI solution. The mixture was extracted with chloroform. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H28CIN5O3S + H)+: 526/528 MS found (electrospray): (M+H)+ = 526/528
Example 136
1 -(5-imidazo[1 ,2-a]pyrimidin-2-yl-2-thienyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Intermediate 275 (92 mg) was dissolved in THF (2 mL) and ethanol (2 mL). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 2 days. The ethanol and THF were removed in vacuo and the residue partitioned between chloroform and 0.1 M aqueous HCI solution. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound.
MS calcd for (C25H28N6O3S + H)+: 493 MS found (electrospray): (M+H)+ = 493
Example 137 1 -(5-imidazo[1 ,2-a]pyridin-2-yl-2-thienyl)-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 188 (61 mg) was dissolved in THF (2 mL) and ethanol (2 mL). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 48 h. The ethanol and THF were removed in vacuo and the residue dissolved in water and the pH adjusted to ~2 with aqueous 2N HCI solution. The mixture was extracted with chloroform. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H29N5O3S + H)+: 492 MS found (electrospray): (M+H)+ = 492 1H NMR (de-DMSO) δ 8.87 (1 H, br. s), 8.52 (1 H, br. d), 8.34 (1 H, s), 8.30 (1 H, s), 7.54 (1 H, br. d), 7.50 (1H, d), 7.45 (1 H, d), 7.27 (1 H, dd), 6.92 (1 H1 dd), 4.71 (1 H, m), 1.96 (1 H, m), 1.68 (2H, br. d), 1.56 (2H, br. d), 1.50-0.50 (14H, br. m).
Example 138 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(5-pyrazolo[1 ,5- a]pyrimidin-2-yl-2-thienyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 191 (106 mg) was dissolved in THF (2 mL) and ethanol (2 mL). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 2 days. The ethanol and THF were removed in vacuo and the residue partitioned between 2N aqueous HCI solution and DCM. The organic layer was separated using a hydrophobic frit and concentrated give the title compound.
MS calcd for (C25H28N6O3S + H)+: 493
MS found (electrospray): (M+H)+ = 493 1H NMR (d6-DMSO) δ 12.85 (1 H, v.br. s), 9.15 (1H, s), 9.09 (1 H1 br. d), 8.55 (1 H, dd), 7.71
(1 H, d), 7.65 (1 H, d), 7.21 (1H, s), 7.06 (1H, dd), 4.73 (1 H, m), 1.93 (1 H, m), 1.70-0.50 (18H, br. m).
Example 139 1 -[4-(2-Chloroimidazo[2,1 -b][1 ,3]thiazol-6-yl)phenyl]-3-[[(fraπs-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 192 (63 mg) was dissolved in THF (2 mL) and ethanol (2 ml_). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 18 h. The ethanol and THF were removed in vacuo and the residue partitioned between 2N aqueous HCI solution and DCM. The organic layer was separated using a hydrophobic frit and concentrated. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H28CIN5O3S + H)+: 526/528 MS found (electrospray): (M+H)+ = 526/528
1H NMR (CDCI3) δ 8.60 (1 H, s), 7.94 (2H, d), 7.79 (2H, d), 7.76 (1 H, s), 7.47 (1 H, s), 4.99 (1 H, m), 2.03 (1 H1 m), 1.90-0.50 (18H1 br. m) carboxylic acid proton not seen.
Example 140
1-(3-Chloro-4-pyrazolo[1,5-a]pyrimidin-2-ylphenyl)-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 197 (167 mg) was dissolved in THF (4 mL) and ethanol (4 mL). Sodium hydroxide solution (2N, 2 mL) was added and the mixture stirred at room temperature for 66 h. The ethanol and THF were removed in vacuo and the residue partitioned between 2N aqueous HCI solution and DCM. The organic layer was separated using a hydrophobic frit and concentrated. The residue was dissolved in methanol and applied to a 1O g aminopropyl SPE Ion exchange cartridge. The cartridge was eluted with two column volumes of methanol followed by six column volumes of methanol/2N aqueous HCI (9:1). Fractions containing the product were combined and concentrated. Further purification by MDAP HPLC gave the title compound. MS calcd for (C27H29CIN6O3 + H)+: 521/523 MS found (electrospray): (M+H)+ = 521/523
1H NMR (CDCI3) δ 8.77 (1H1 br. d), 8.65 (1H1 s), 8.56 (1H1 dd), 8.13 (1H1 d), 7.98 (1 H, d), 7.76 (1 H1 dd), 7.32 (1 H1 s), 6.92 (1 H, dd), 5.00 (1H1 m), 2.00 (1 H1 m), 1.90-0.55 (18H, br. m) carboxylic acid proton not seen.
Example 141
3-[[(rrans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(3-methyl-4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylic acid
Intermediate 202 (271 mg) was dissolved in THF (4 mL) and ethanol (4 ml_). Sodium hydroxide solution (2N, 2 mL) was added and the mixture stirred at room temperature for 66 h. The ethanol and THF were removed in vacuo and the residue partitioned between 2N aqueous HCI solution and DCM. The organic layer was separated using a hydrophobic frit and concentrated to give the title compound. MS calcd for (C28H32N6O3 + H)+: 501 MS found (electrospray): (M+H)+ = 501
1H NMR (CDCI3) δ 8.77 (1H, br. d), 8.62 (1H, s), 8.55 (1H, dd), 7,87 (1H, d), 7.72 (1H, d), 7.67 (1H, dd), 6.93 (1 H, s), 6.90 (1 H, dd), 4.99 (1 H, m), 2.68 (3H, s), 2.02 (1 H, m), 1.90-0.55 (18H, br. m) carboxylic acid proton not seen.
Example 142
3-[[(2,4-Dichlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-furo[3,2-b]pyridin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 204 (109 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit. The aqueous was decanted off leaving undissolved material on the frit which was dissolved in MeOH. The DCM and MeOH fractions were combined and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C27H20CI2N4O4+ H)+: 535/536/537 MS found (electrospray): (M+H)+ = 535/536/537
1H NMR (DMSO-d6 at 378K) δ 8.75 (1 H, br.s), 8.53 (1 H, dd), 8.10 (2H, d), 7.97 (3H1 m), 7.58 (1 H, d), 7.49-7.37 (2H, br.m), 7.37-7.21 (2H, m), 4.69 (1 H, br), 1.29 (6H, d) carboxylic acid proton not seen.
Example 143
S-Hffrans^-MethylcyclohexyOcarbonylHI-methylethyOaminol-i-^-rβ- (trifluoromethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-1 H-pyrazole-4-carboxylic acid
Intermediate 205 (137 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N1 1.0 mL). The reaction mixture was stirred at room temperature for 2 days. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C29H30F3N5O3+ H)+: 554 MS found (electrospray): (M+H)+ = 554
Example 144
S-lKfrans^-MethylcyclohexyOcarbonylHI-methylethyOaminol-i-^-fβ- methylimidazo[1 ,2-a]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
Intermediate 206 (74 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature for 2 days. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C29H33N5O3+ H)+: 500 MS found (electrospray): (M+H)+ = 500
Example 145
1 -[5-(1 ,3-Benzoxazol-2-yl)-3-furanyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 208 (55 mg) was dissolved in tetrahydrofuran (1.5 ml_) and ethanol (1.5 ml.) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. DCM (12 mL) and 2N HCI were added and the reaction mixture was stirred at room temperature for 30 min. The organic was separated and dried using a hydrophobic frit and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H28N4O5+ H)+: 477 MS found (electrospray): (M+H)+ = 477
Example 146
3-[[(2,4-Dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-imidazo[1 ,2-a]pyridin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 210 (128 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit. The aqueous was decanted off leaving undissolved material on the frit which was dissolved in MeOH. The DCM and MeOH fractions were combined and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C27H21CI2N5O3+ H)+: 534/535/536 MS found (electrospray): (M+H)+ = 534/535/536
Example 147
3-[[(2,4-Dimethylphenyl)carbonyl](1-methylethyl)amino]-1-(4-furo[3,2-b]pyridin-2- ylphenyl)-1 W-pyrazole-4-carboxylic acid
Intermediate 213 (102 mg) was dissolved in tetrahydrofuran (1.3 ml_) and ethanol (1.3 mL) and treated with sodium hydroxide solution (2N, 0.8 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was dried using a hydrophobic frit and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C29H26N4O4+ H)+: 495 MS found (electrospray): (M+H)+ = 495
Example 148
3-[[(4-Bromo-2-fluorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 214 (82 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N1 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit and the organic was evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H20BrFN6O3+ H)+: 563/565 MS found (electrospray): (M+H)+ = 563/565
Example 149
3-[[(4-Bromo-2-methylphenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 215 (32 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit and the organic was evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C27H23BrN6O3+ H)+: 559/561 MS found (electrospray): (M+H)+ = 559/561
Example 150
3-[[(4-Chloro-2-fluorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 216 (52 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit and the organic was evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H20CIFN6O3+ H)+: 519/521 MS found (electrospray): (M+H)+ = 519/521
Example 151
3-[[(2,4-Dimethylphenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-
Intermediate 217 (75 mg) was dissolved in tetrahydrofuran (1.0 ml_) and ethanol (1.0 mL) and treated with sodium hydroxide solution (2N, 0.6 mL). The reaction mixture was stirred at room temperature for 42 h. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit and the organic was evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C28H26N6O3+ H)+: 495 MS found (electrospray): (M+H)+ = 495
Example 152
3-[(1 -Benzofuran-5-ylcarbonyl)(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 218 (76 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit. The aqueous was decanted off leaving undissolved material on the frit which was dissolved in MeOH. The DCM and MeOH fractions were combined and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C28H22N6O4+ H)+: 507 MS found (electrospray): (M+H)+ = 507
Example 153
3-[[(4-Bromo-2-chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
Intermediate 219 (16 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 ml_) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit and the organic was evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H20BrCIN6O3+ H)+: 579/581/582/583 MS found (electrospray): (M+H)+ = 579/581/582/583
Example 154
3-[[(2-Bromo-4-chlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1 W-pyrazole-4-carboxylic acid
Intermediate 220 (128 mg) was dissolved in tetrahydrofuran (1.5 mL) and ethanol (1.5 mL) and treated with sodium hydroxide solution (2N, 1.0 mL). The reaction mixture was stirred at room temperature overnight. The reaction mixture was evaporated to dryness and was partitioned between 2N HCI and DCM. The organic was separated and dried using a hydrophobic frit. The aqueous was decanted off leaving undissolved material on the frit which was dissolved in MeOH. The DCM and MeOH fractions were combined and evaporated to dryness. The residue was purified by MDAP HPLC to give the title compound. MS calcd for (C26H20BrCIN6O3+ H)+: 579/581/582/583 MS found (electrospray): (M+H)+ = 579/581/582/583
Example 155
1-[4-(5-Amino-6-chloro-3-pyridinyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
A solution of Intermediate 267 (47 mg) and 2M lithium hydroxide (0.5 ml_) in ethanol (2 ml_) was stirred at room temperature for 10 h and then evaporated to dryness. The residue was diluted with water, acidified to pH 5 with 2M HCI, extracted with EtOAc, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by MDAP HPLC to give the title compound as a white solid. MS calcd for (C26H30CIN5O3 + H)+: 496/498 MS found (electrospray): (M+H)+ = 496/498
Example 156
S-IKfrans^-MethylcyclohexyOcarbonylKI-methylethylJaminol-i-μ-β- quinolinyl)phenyl]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 268 (130 mg) and 2M lithium hydroxide (1 ml_) in ethanol (5 ml_) was stirred at room temperature for 18 h and then evaporated to dryness. The residue was diluted with water, acidified to pH 4 with 2M HCI, extracted with EtOAc, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by MDAP HPLC to give the title compound as a white solid. MS calcd for (C30H32N4O3 + H)+: 497 MS found (electrospray): (M+H)+ = 497
Example 157
S-IKfraπs^-MethylcyclohexyOcarbonylKI-methylethyOaminol-i-t^θ- quinoxalinyl)phenyl]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 269 (59 mg) and 2M lithium hydroxide (0.5 mL) in ethanol (2 mL) was stirred at room temperature for 18 h and then evaporated to dryness. The residue was
diluted with water, acidified to pH 5 with 2M HCI, extracted twice with EtOAc, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 12 g silica ISCO Companion chromatography eluted with 0-100% MeOH in DCM to give the title compound. MS calcd for (C29H3IN5O3 + H)+: 498 MS found (electrospray): (M+H)+ = 498
Example 158 i-^^-Amino-θ-quinolinyOphenyπ-a-tKfrans^-methylcyclohexyOcarbonylKI- methylethyl)amino]-1H-pyrazole-4-carboxylic acid
A solution of Intermediate 270 (73 mg) and 2M lithium hydroxide (0.5 mL) in ethanol (2 mL) was stirred at room temperature for 18 h and then evaporated to dryness. The residue was diluted with water and acidified to pH 5 with 2M HCI, then purified by Oasis HLB cartridge eluted with water (2 x column volumes), dioxan (2 x column volumes) and MeOH (2 x column volumes). The organic components were combined and evaporated, then the crude product was purified by 12 g silica ISCO Companion chromatography eluted with 0-100% MeOH in DCM to give the title compound. MS calcd for (C30H33N5O3 + H)+: 512 MS found (electrospray): (M+H)+ = 512
Example 159
1-[4-(6-lsoquinolinyl)phenyl]-3-[[(fraπs-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
A solution of Intermediate 271 (71 mg) and 2M lithium hydroxide (0.5 mL) in ethanol (2 mL) was stirred at room temperature for 24 h and then evaporated to dryness. The residue was diluted with water and acidified to pH 6 with 2M HCI, extracted twice with EtOAc, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 12 g silica ISCO Companion chromatography eluted with 0-100% MeOH in DCM to give the title compound. MS calcd for (C30H32N4O3 + H)+: 497
MS found (electrospray): (M+H)+ = 497
Example 160
S-^fraπs^-MethylcyclohexyOcarbonylKI-methylethyOaminol-i-t^β- quinolinyl)phenyl]-1H-pyrazole-4-carboxylic acid
A solution of Intermediate 273 (37 mg) and 2M lithium hydroxide (0.5 ml_) in ethanol (2 mL) was stirred at room temperature for 3 days and then evaporated to dryness. The residue was diluted with water and acidified to pH 6 with 2M HCI, extracted twice with EtOAc, dried using a hydrophobic frit and evaporated to dryness. The crude product was purified by 2 g silica SPE cartridge eluted with a gradient of DCM to EtOAc to give the title compound. MS calcd for (C30H32N4O3 + H)+: 497 MS found (electrospray): (M+H)+ = 497
Example 161
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(7-methylfuro[3,2- b]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 274 (49 mg) and 2M sodium hydroxide solution (0.23 mL) in THF (0.5 mL) and MeOH (0.5 mL) was stirred overnight at room temperature. Solvent was evaporated under vacuum and the residue was partitioned between 2M HCI and DCM containing some methanol to aid solubility. The organic phase was separated, evaporated and purified by MDAP HPLC. Combined product fractions were evaporated and freeze dried from dioxan to give the title compound. MS calcd for (C29H32N4O4 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 162
1-{3-Chloro-4-[6-(methyloxy)-3-pyridinyl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](tetrahydro-2W-pyran-4-yl)amino]-1H-pyrazole-4-carboxylic acid
Intermediate 175 (265 mg) was dissolved in THF (12 mL) and ethanol (6 ml_). 2N sodium hydroxide solution (3 mL) was added and the mixture was left to stand at room temperature overnight. The ethanol and THF were removed in vacuo, the residue partitioned between aqueous 1 M HCI and DCM, passed through a hydrophobic frit and the organic layer evaporated in vacuo. The crude product was purified by MDAP HPLC to give title compound.
MS calcd for (C29H33CIN4O5+ H)+: 554/556 MS found (electrospray): (M+H)+ = 554/556
Example 163 1 -[4-(7-Hydroxy-1 -benzofuran-Z-ylJphenyll-S-IKfrans^-methylcyclohexylJcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
Intermediate 278 (0.18 g) was dissolved in ethanol (2 mL) and THF (2 mL) and to this solution was added 2N sodium hydroxide (0.75 mL). The resultant solution was stirred at room temperature for 0.5 h and was then allowed to stand for 20 h. The reaction mixture was evaporated to dryness and the residue was partitioned between DCM and 0.5M HCI. The organic phase was separated using a hydrophobic frit and was then evaporated to dryness. The residue was purified using MDAP HPLC. Appropriate fractions were combined and evaporated and the residue was freeze-dried from dioxan to give the title compound. MS calcd for (C29H31N3O5 + H)+: 502 MS found (electrospray): (M+H)+ = 502
1H NMR (CDCI3) δ 10.17 (1 H, v.br.s), 9.19 (1 H, s), 8.07 (4H, AA'BB'), 7.47 (1 H, s), 7.09 (1 H, dd), 7.05 (1 H, dd), 6.78 (1 H, dd), 4.75 (1 H, m), 1.96 (1 H, tt), 1.72-0.49 (18H, 6 X m+d). One exchangeable proton is not seen.
Example 164
1 -[4-(5-Amino-1 -benzofuran^-yOphenyll-S-tKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid
To a solution of Intermediate 90 (186 mg) in THF (4 mL) and ethanol (4 ml.) was added 2M aqueous sodium hydroxide solution (2 mL). The mixture was stirred at room temperature for 24 h and then evaporated to dryness under reduced pressure. The residue was partitioned between 2M HCI and DCM. A gum was collected from the interface of the two layers. This was dissolved in MeOH and was evaporated to give a solid which was washed with diethyl ether and dried under vacuum at 400C overnight to give the title compound. MS calcd for (C29H32N4O4 + H)+: 501 MS found (electrospray): (M+H)+ = 501
Example 165 1 -[4-(7-Aminopyrazolo[1 ,5-a]pyrimidin-2-yl)-3-chlorophenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 W-pyrazole-4-carboxylic acid
Intermediate 283 (9 mg) was dissolved in THF (2 mL) and ethanol (2 mL). Sodium hydroxide solution (2N, 1 mL) was added and the mixture stirred at room temperature for 48 h. The reaction was partitioned between 2N aqueous HCI solution and ethyl acetate. The organic layer was separated and dried (Na2SO4) and was concentrated. The residue was freeze- dried from1 ,4-dioxan to give the title compound.
MS calcd for (C27H30CIN7O3 + H)+: 536/538
MS found (electrospray): (M+H)+ = 536/538 1H NMR (MeOD) δ 9.04 (s, 1H), 8.25 (d, 1 H), 8.23 (d, 1H), 8.14 (d, 1 H), 7.99 (dd, 1 H), 7.09
(s, 1 H), 6.43 (d, 1 H), 4.80 - 4.87 (m, 1 H), 2.05 (tt, 1 H), 1.75 - 1.82 (m, 2H), 0.87 - 0.91 (m,
3H), 0.79 (d, 6H), 0.54 - 1.73 (m, 7H + large excess), carboxylic acid and amino protons not seen.
Example 166
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -(4-[1 ,3]oxazolo[4,5- c]pyridin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid
A solution of Intermediate 285 (127 mg) in dry DMF (5 ml.) was added to Intermediate 82 (123.2 mg), followed by copper (I) iodide (19.2 mg), frans-N, N'-bismethyl-1 ,2- cyclohexane diamine (28.7 mg) and potassium carbonate (139.6 mg). This was heated at 16O0C under nitrogen for 17 h. The DMF was removed in vacuo and the residue partitioned between DCM and water. The DCM layer was separated using a hydrophobic frit and concentrated. This residue was purified by ISCO Companion C-18 reverse phase chromatography eluting with a gradient of 5% acetonitrile (0.05% formic acid)/ water (0.1% formic acid) to 100% acetonitrile (0.05% formic acid) to give the title compound. MS calcd for (C30H34CIN5O3 + H)+: 488 MS found (electrospray): (M+H)+ = 488 The following examples are also within the scope of this invention:
The chemical entities according to the invention may be formulated for administration in any convenient way, and the invention therefore also includes within its scope pharmaceutical compositions for use in therapy, comprising a compound of formula (I) or a pharmaceutically acceptable salt or solvate thereof in admixture with one or more pharmaceutically acceptable diluents or carriers.
The chemical entities of the present invention can be administered by different routes including intravenous, intraperitoneal, subcutaneous, intramuscular, oral, topical, transdermal, or transmucosal administration. For systemic administration, oral administration is preferred. For oral administration, for example, the chemical entities can be formulated into conventional oral dosage forms such as capsules, tablets and liquid preparations such as syrups, elixirs and concentrated drops.
Alternatively, injection (parenteral administration) may be used, e.g., intramuscular, intravenous, intraperitoneal, and subcutaneous. For injection, the chemical entities of the invention are formulated in liquid solutions, preferably, in pharmaceutically compatible buffers or solutions, such as saline solution, Hank's solution, or Ringer's solution. In addition, the chemical entities may be formulated in solid form and redissolved or suspended immediately prior to use. Lyophilized forms can also be produced.
Systemic administration can also be by transmucosal or transdermal means. For transmucosal or transdermal administration, penetrants appropriate to the barrier to be permeated are used in the formulation. Such penetrants are generally known in the art, and include, for example, for transmucosal administration, bile salts and fusidic acid derivatives. In addition, detergents may be used to facilitate permeation. Transmucosal administration, for example, may be through nasal sprays, rectal suppositories, or vaginal suppositories.
For topical administration, the compounds of the invention can be formulated into ointments, salves, gels, or creams, as is generally known in the art.
The amounts of various chemical entities to be administered can be determined by standard procedures taking into account factors such as the compound (IC50) potency, (EC50) efficacy, and the biological half-life (of the chemical entity), the age, size and weight of the patient, and the disease or disorder associated with the patient. The importance of these and other factors to be considered are known to those of ordinary skill in the art.
Amounts administered also depend on the routes of administration and the degree of oral bioavailability. For example, for chemical entities with low oral bioavailability, relatively higher doses will have to be administered. Oral administration is a preferred method of administration of the present chemical entities.
Preferably the composition is in unit dosage form. For oral application, for example, a tablet, or capsule may be administered, for nasal application, a metered aerosol dose may be administered, for transdermal application, a topical formulation or patch may be administered and for transmucosal delivery, a buccal patch may be administered. In each case, dosing is such that the patient may administer a single dose.
Each dosage unit for oral administration contains suitably from 0.01 to 500 mg/Kg, and preferably from 0.1 to 50 mg/Kg, of a compound of Formula (I) or a pharmaceutically acceptable salt thereof, calculated as the free base. The daily dosage for parenteral, nasal, oral inhalation, transmucosal or transdermal routes contains suitably from 0.01 mg to 100 mg/Kg, of a compound of Formula(l). A topical formulation contains suitably 0.01 to 5.0% of a compound of Formula (I). The active ingredient may be administered from 1 to 6 times per day, preferably once, sufficient to exhibit the desired activity, as is readily apparent to one skilled in the art.
Chemical entities of Formula (I) which are active when given orally can be formulated as syrups, tablets, capsules and lozenges. A syrup formulation will generally consist of a suspension or solution of the compound or salt in a liquid carrier for example, ethanol, peanut oil. olive oil, glycerine or water with a flavoring or coloring agent. Where the composition is in the form of a tablet, any pharmaceutical carrier routinely used for preparing solid formulations may be used. Examples of such carriers include magnesium stearate, terra alba, talc, gelatin, acacia, stearic acid, starch, lactose and sucrose. Where the composition is in the form of a capsule, any routine encapsulation is suitable, for example using the aforementioned carriers in a hard gelatin capsule shell. Where the composition is in the form of a soft gelatin shell capsule any pharmaceutical carrier routinely used for preparing dispersions or suspensions may be considered, for example aqueous gums, celluloses, silicates or oils, and are incorporated in a soft gelatin capsule shell.
Typical parenteral compositions consist of a solution or suspension of a compound or salt in a sterile aqueous or non-aqueous carrier optionally containing a parenterally acceptable oil, for example polyethylene glycol, polyvinylpyrrolidone, lecithin, arachis oil or sesame oil.
Typical compositions for inhalation are in the form of a solution, suspension or emulsion that may be administered as a dry powder or in the form of an aerosol using a conventional non- CFC propellant such as 1 ,1 ,1 ,2-tetrafluoroethane or 1 ,1 ,1 ,2,3,3,3-heptafluoropropane.
A typical suppository formulation comprises a compound of Formula (I) or a pharmaceutically acceptable salt thereof which is active when administered in this way, with a binding and/or lubricating agent, for example polymeric glycols, gelatins, cocoa-butter or other low melting vegetable waxes or fats or their synthetic analogs.
Typical dermal and transdermal formulations comprise a conventional aqueous or nonaqueous vehicle, for example a cream, ointment, lotion or paste or are in the form of a medicated plaster, patch or membrane.
No unacceptable toxicological effects are expected when compounds of the present invention are administered in accordance with the present invention.
ASSAY
The potential for chemical entities of the invention to inhibit NS5B wildtype HCV polymerase activity may be demonstrated, for example, using the following in vitro assay:
In Vitro Detection of inhibitors of HCV RNA-dependent RNA Polymerase Activity
Incorporation of [33P]-GMP into RNA was followed by absorption of the biotin labelled RNA polymer by streptavidin containing SPA beads. A synthetic template consisting of biotinylated 13mer-oligoG hybridised to polyrC was used as a homopolymer substrate.
Reaction Conditions were 0.5 μM [33P]-GTP (20 Ci/mMol), 1 mM Dithiothreitol, 20 mM MgCI2, 5mM MnCI2, 20 mM Tris-HCI, pH7.5, 1.6 μg/mL polyC/0.256 μM biotinylated oligoG13, 10% glycerol, 0.01 % NP-40, 0.2 u/μL RNasin and 50 mM NaCI.
HCV RNA Polymerase (Recombinant full-length NS5B (Lohmann et al, J. Virol. 71 (11), 1997, 8416 'Biochemical properties of hepatitis C virus NS5B RNA-dependent RNA polymerase and identification of amino acid sequence motifs essential for enzymatic activity') expressed in baculovirus and purified to homogeneity) was added to 4 nM final concentration.
5x concentrated assay buffer mix was prepared using 1M MnCI2 (0.25 ml_), glycerol (2.5mL), 10% NP-40 (0.025 mL) and Water (7.225 mL), Total 10 mL
2x concentrated enzyme buffer contained 1 M-Tris-HCI, pH7.5 (0.4 mL), 5M NaCI (0.2 mL), 1M-MgCI2 (0.4 mL), glycerol (1 mL), 10% NP-40 (10 μL), 1 M DTT (20 μL) and water (7.97 mL), Tote/ 1O mL
Substrate Mix was prepared using 5x Concentrated assay Buffer mix (4μL), [33P]-GTP (10 μCi/μL, 0.02μL), 25 μM GTP (0.4 μL), 40 u/μL RNasin (0.1 μL), 20 μg/mL polyrC/biotinylated- oligorG (1.6 μL), and Water (3.94 μL), Total 10 μL.
Enzyme Mix was prepared by adding 1mg/mL full-length NS5B polymerase (1.5 μL) to 2.81 mL 2x-concentrated enzyme buffer.
The Assay was set up using compound (1 μL), Substrate Mix (10 μL), and Enzyme Mix (added last to start reaction) (10 μL), Total is μL.
The reaction was performed in a U-bottomed, white, 96-well plate. The reaction was mixed on a plate-shaker, after addition of the Enzyme, and incubated for 1h at 22°C. After this time, the reaction was stopped by addition of 40 μl_ 1.875 mg/mL streptavidin SPA beads in 0.1 M EDTA. The beads were incubated with the reaction mixture for 1 h at 22°C after which 120 μL 0.1 M EDTA in PBS was added. The plate was sealed, mixed centrifuged and incorporated radioactivity determined by counting in a Trilux (Wallac) or Topcount (Packard) Scintillation Counter.
After subtraction of background levels without enzyme, any reduction in the amount of radioactivity incorporated in the presence of a compound, compared to that in the absence, was taken as a measure of the level of inhibition. Ten concentrations of compounds were tested in three- or fivefold dilutions. From the counts, percentage of inhibition at highest concentration tested or IC50S for the compounds were calculated using Grafιt3, Grafit4 or Grafitδ software packages or a data evaluation macro for Excel based on XLFit software (IDBS).
The exemplified compounds had an IC50 of <10μM in the above described assay. In one aspect, compounds have an IC50 of <1 μM, for example <0.2μM. Accordingly, the compounds of the invention are of potential therapeutic benefit in the treatment and prophylaxis of HCV.
The pharmaceutical compositions according to the invention may also be used in combination with other therapeutic agents, for example immune therapies (eg. Interferon, such as Interferon alfa-2a (Roferon-A; Hoffmann-La Roche), inteferon alpha-2b (Intron-A;
Schering-Plough), interferon alfacon-1 (Infergen; Intermune), peginterferon alpha-2b (Peg-
Intron; Schering-Plough) or peginterferon alpha-2a (Pegasys; Hoffmann-La Roche)), therapeutic vaccines, antifibrotic agents, anti-inflammatory agents such as corticosteroids or NSAIDs, bronchodilators such as beta-2 adrenergic agonists and xanthines (e.g. theophylline), mucolytic agents, anti-muscarinics, anti-leukotrienes, inhibitors of cell adhesion
(e.g. ICAM antagonists), anti-oxidants (eg N-acetylcysteine), cytokine agonists, cytokine antagonists, lung surfactants and/or antimicrobial and anti-viral agents (eg ribavirin and amantidine). The compositions according to the invention may also be used in combination with gene replacement therapy.
The invention thus provides, in a further aspect, a combination comprising at least one chemical entity of Formula (I) together with at least one other therapeutically active agent, especially Interferon, ribavirin and/or an additional anti-HCV agent.
The combinations referred to above may conveniently be presented for use in the form of a pharmaceutical formulation and thus pharmaceutical formulations comprising a combination
as defined above together with a pharmaceutically acceptable carrier thereof represent a further aspect of the invention.
The individual components of such combinations may be administered either sequentially or simultaneously in separate or combined pharmaceutical formulations. Appropriate doses of known therapeutic agents will be readily appreciated by those skilled in the art.
All publications, including but not limited to patents and patent applications cited in this specification are herein incorporated by reference as if each individual publication were specifically and individually indicated to be incorporated by reference as though fully set forth.
The application of which this description and claims forms part may be used as a basis for priority in respect of any subsequent application. The claims of such subsequent application may be directed to any feature or combination of features described herein. They may take the form of product, composition, process, or use claims and may include, by way of example and without limitation, the following claims:
Claims
1. At least one chemical entity chosen from compounds Formula (I) :
A represents hydroxy;
R1 represents -Rx-Rγ;
Rx represents phenyl (optionally substituted by halo, methyl, ethyl, methoxy or trifluoromethyl) or a 5 or 6-membered heteroaryl bonded through a ring carbon atom to the nitrogen atom of the pyrazole;
Rγ represents heteroaryl, bonded such that when Rx is phenyl, Rγ is in the para-position;
R2 represents phenyl substituted by one or more substituents selected from -C1-6alkyl, halo, -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NREC02R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, and heterocyclyl, or two substituents on neighbouring carbon atoms of the phenyl ring together form a A-, 5- or 6-membered ring containing one or more heteroatoms selected from O, S and N; or R2 represents
=CH(CH2),H, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(0)R°, -NRECO2R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a Ci.2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4spiro substituent; or R2 represents Cs-ycycloalkylmethyl, the cycloalkyl portion being optionally substituted by one or more substitutents selected from -Chalky!, =CH(CH2),H, - 0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NREC02R°, - NREC(O)NRFRG, -S02NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a C1-2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4spiro substituent;
t represents O, 1 , 2, 3 or 4;
R3 represents heterocyclyl; or phenyl optionally substituted by one or more substituents selected from -C1-6alkyl, halo, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H1
-CO2R0, -NRBRC, -NREC(O)RD, -NRECO2RD, -NRESO2RD, -NREC(O)NRFRG, -SO2NRFRG, - SO2R0, nitro, cyano, and heterocyclyl; or R3 represents
optionally substituted by one or more substituents selected from -0RA, -SRA, -C(O)NRBRC, -C(O)RD, -CO2H, -CO2R0, -NRBRC, -NREC(O)RD, -NRECO2RD, -NRES02R°, -NREC(0)NRFRG, -SO2NRFRG, - SO2R0, fluoro, nitro, cyano, oxo, phenyl, heteroaryl and heterocyclyl;
R4 represents hydrogen;
RA represents hydrogen, -Chalky!, arylalkyl, heteroarylalkyl, aryl, heterocyclyl or heteroaryl;
RB and Rc independently represent hydrogen, -C1-6alkyl, aryl, heterocyclyl or heteroaryl; or RB and Rc together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
RD is selected from the group consisting of -C^alkyl, aryl, heterocyclyl, heteroaryl, arylalkyl, and heteroarylalkyl;
RE represents hydrogen or -C1-6alkyl;
RF and RG are independently selected from the group consisting of hydrogen, -C1-6alkyl, aryl, heteroaryl, arylalkyl, and heteroarylalkyl; or RF and RG together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
and salts, solvates and esters thereof.
2. At least one chemical entity as claimed in claim 1 chosen from compounds of
Formula (I) selected from the group consisting of:
1 -[4-(1 H-lndol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H- pyrazole-4-carboxylic acid; 1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid; 1-[4-(1H-Benzimidazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[3-Chloro-4-(1 H-indol-2-yl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(2H-lndazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-
1 /-/-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 -oxo-1 ,3-dihydro-2H- isoindol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid; 1-[4-(1,1-Dioxido-1 ,2-benzisothiazol-2(3H)-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
^(^FurotS^-^pyridin^-ylphenyO-S-^frans^-methylcyclohexyOcarbonylKI- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1 ,3-Benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 /-/-pyrazole-4-carboxylic acid; 1-[4-(1-Benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2/-/-pyran-
4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1-Benzothien-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid;
S-lt^rans^-MethylcyclohexyOcarbonylJCI-methylethyOaminol-i^-ti .SJthiazolotδ^-^pyridin- 2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcydohexyl)carbonyl](1-methylethyl)amino]-1-[4-(1-methyl-1 H-indol-2- yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(6-Fluoro-1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 -[4-(1 -oxo-1 ,3- dihydro-2H-isoindol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(1 ,3-Benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1 ,3-Benzoxazol-2-yl)phenyl]-3-{ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1H- pyrazole-4-carboxylic acid;
1 -[4-(1 ,3-Benzothiazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 )5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 1-(4-Furo[3,2-/)]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
3-{Ethyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-furo[3,2-ft]pyridin-2-ylphenyl)-1 H- pyrazole-4-carboxylic acid;
3-{Ethyl[(frans-4-rnethylcyclohexyl)carbonyl]amino}-1-(4-imidazo[1 ,2-a]pyridin-2-ylphenyl)- 1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro-
2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(5-Fluoro-1 H-indol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -[4-(3-Chloro-2H-indazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1 ,3-Benzothiazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(1 -Benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(6-Chloro-7aH-imidazo[4,5-/j]pyridin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Amino-5-methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- rnethylcyclohexyl)carbonyl](1-methylethyl)amino]-1/-/-pyrazole-4-carboxylic acid;
1 -[4-(5-Amino-1 ,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-(4-Furo[3,2-/5]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][(3R)-tetrahydro-3- furanyl]amino}-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-i»]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][(3S)-tetrahydro-3- furanyl]amino}-1 /-/-pyrazole-4-carboxylic acid;
1-[4-(4-Amino-3-cyanopyrazolo[5,1-c][1,2,4]triazin-7-yl)phenyl]-3-[[(^ans-4- methylcyclohexylJcarbonylKI -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(2H-lndazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-
1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-c/]pyrimidin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-(4-Furo[2,3-c(]pyrimidin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[2,1-b][1,3]thiazol-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5,7-Dimethylpyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-3-[[(fraA7s-4- methylcyclohexyOcarbony^i -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ,2-a]pyrimidin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-c]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid 1 -(4-Furo[3,2-/)]pyridin-2-ylphenyl)-3-{(1 -methylethyl)[(4-methylphenyl)carbonyl]amino}-1 H- pyrazole-4-carboxylic acid;
3-[(Cyclohexylcarbonyl)(1 -methylethyl)amino]-1 -(4-furo[3,2-b]pyridin-2-ylphenyl)-1 H- pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-[1,3]oxazolo[4,5-d]pyridin- 2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{Ethyl[(^ans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-3-furanyl)amino]-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro-3- furanyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ^-alpyridin^-ylphenyO-S-t^frans^-methylcyclohexyOcarbonyl] (tetrahydro-3- furanyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[5-(1,3-Benzoxazol-2-yl)-2-thienyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1,3-Benzoxazol-2-yl)-3-chlorophenyl]-3-[[(fraA7s-4-methylcyclohexyl) carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(^ans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(5-methylpyrazolo[1 ,5- a]pyrimidin-2-yl)phenyl]-1 /-/-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(7-methylpyrazolo[1 ,5- a]pyrimidin-2-yl)phenyl]-1 /-/-pyrazole-4-carboxylic acid;
S-IfC^ans^-MethylcyclohexyOcarbony^i-methylethyOaminol-i-^-CS-methylfurotS^- to]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(^ans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6-methylfuro[3,2- jb]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-[6-(1,3-Benzoxazol-2-yl)-3-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl] (1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(6-Furo[3,2-/?]pyridin-2-yl-3-pyridinyl)-3-[[(frans-4-methylcyclohexyl)carbonyl] (1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1-(6-pyrazolo[1 ,5-a]pyrimidin-2- yl-3-pyridinyl)-1 H-pyrazole-4-carboxylic acid;
1-[6-(6-Fluoropyrazolo[1 ,5-a]pyrimidin-2-yl)-3-pyridinyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-ylmethyl)amino]-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-/j]pyridin-2-ylphenyl)-3-{[(frans-4-methylcyclohexyl)carbonyl][2-
(methyloxy)ethyl]amino}-1 H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[2,1-b][1,3]thiazol-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl]
(tetrahydro-3-furanyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-(4-lmidazo[2,1-b][1,3]thiazol-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl) carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
3-{(2-Ethylbutyl)[(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[(frans-4-Methylcyclohexyl)carbonyl][2-(methyloxy)ethyl]amino}-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
S-^frans^-MethylcyclohexyOcarbonylKpropyOaminol-i^-pyrazololi .S-aJpyrimidin^- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](2-methylpropyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-{(1 -Ethylpropyl)[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{(1 ,3-Dimethylbutyl)[(frans-4-methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{Cyclopentyl[(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{(1-Methylethyl)[(4-methylphenyl)carbonyl]amino}-1-(4-pyrazolo[1,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(4-Chlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 /-/-pyrazole-4-carboxylic acid; 3-((1-Methylethyl){[4-(trifluorornethyl)phenyl]carbonyl}amino)-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(3,4-Dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 /-/-pyrazole-4-carboxylic acid;
1-[3-Chloro-4-(2-furanyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2/-/-pyran- 4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[3-Chloro-4-(3,5-dimethyl-4-isoxazolyl)phenyl]-3-[[(frans-4-methylcyclohexyl) carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
3-([(frans-4-Methylcyclohexyl)carbonyl]{2-(methyloxy)-1 -[(methyloxy)methyl] ethyl}amino)-1 -
(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 1 -(4-Furo[3,2-jb]pyridin-2-ylphenyl)-3-([(frans-4-methylcyclohexyl)carbonyl]{2-(methyloxy)-1 -
[(methyloxy)methyl]ethyl}amino)-1 H-pyrazole-4-carboxylic acid;
1-[4-(1-Benzofuran-6-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-
4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(1 -Benzofuran-5-yl)phenyl]-3-[[(fra/7S-4-methylcyclohexyl)carbonyl] (tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(1-Benzofuran-6-yl)-3-chlorophenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro-
2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[2,3-b]pyridin-5-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl] (tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(1H-pyrrolo[3,2-b]pyridin-
2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 H-pyrrolo[2,3-ό]pyridin-
2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-[6-(1-Benzofuran-2-yl)-3-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[6-(1H-lndol-2-yl)-3-pyridinyl]-3-[[(^ans-4-rnethylcyclohexyl)carbonyl](1-rnethylethyl)arnino]-
1 H-pyrazole-4-carboxylic acid;
1-[5-(1-Benzofuran-2-yl)-2-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -[5-(1 H-lndol-2-yl)-2-pyridinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 -methylethyl)amino]-
1 H-pyrazole-4-carboxylic acid;
1 -[6-(1 -Benzofuran-2-yl)-3-pyridazinyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Aminopyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4-methyl cyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(6-methylpyrazolo[1 ,5- a]pyrimidin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(^ans-4-Ethylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrinniclin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-((1-Methylethyl){[fraπs-4-(trifluoromethyl)cyclohexyl]carbonyl}amino)-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(3-methyl-1 H-indol-2- yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(7-oxo-5,7-dihydro-6H- pyrrolo[3,4--)]pyridin-6-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(5-Fluoro-1 -oxo-1 ,3-dihydro-2H-isoindol-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(fraπs-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(5-oxo-5,7-dihydro-6H- pyrrolo[3,4-ό]pyridin-6-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[2,3-ό]pyridin-2-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -[4-(3-Methyl-1 -benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-rnethylethyl)amino]-1-(4-[1 ,3]oxazolo[4,5-/)]pyridin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(4-oxo-1 ,4- dihydrothieno[3,2-cflpyrimidin-6-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(2-Chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(4-Fluorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrirnidin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(1 H-pyrrolo[3,2-b]pyridin-
2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6-methylfuro[3,2- ύ]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-{(1 -Methylethyl)[(2-methylphenyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Hydroxy-5-methylpyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1-methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(3-Chlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(4-Bromophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Difluorophenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- yl phenyl )-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)∞rbonyl](1-methylethyl)amino]-1-(4-{5-[(phenyloxy)methyl]- 1 ,3,4-oxadiazol-2-yl}phenyl)-1 H-pyrazole-4-carboxylic acid; or
1 -[4-(1 , 1 -Dioxido-2H-1 ,2,4-benzothiadiazin-3-yl)phenyl]-3-[[(trans-4-methylcyclohexyl) carbonyl](1-methylethyl) amino]-1 H-pyrazole-4-carboxylic acid;
^^-(θ-Amino-i-benzofuran^-yOphenylJ-S-I^frans-A-methylcyclohexyOcarbony^i- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(4-Amino-1 ,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 3-[[(^ans-4-Methylcyclohexyl)carbonyl](1-methylethyl)arnino]-1-[4-(1 ,8-naphthyridin-2- yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2/-/-pyran-4-yl)amino]-1-[4-(3-methyl-1H- indol-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-[4-(8-Aminoirnidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[(^ans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(4-Aminothieno[3,2-c/]pyrimidin-6-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1.{4.[4-(Methylamino)thieno[3,2-cf]pyrimidin-6-yl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -(4-Pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[^rans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-t»]pyridin-2-ylphenyl)-3-(tetrahydro-2H-pyran-4-yl{[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[1 ,2-a]pyridin-2-ylphenyl)-3-((1-methylethyl){[frans-4- (trifluoromethyOcyclohexyllcarbonylJamino)-! H-pyrazole-4-carboxylic acid;
1-(4-lmidazo[2,1-ύ][1,3]thiazol-6-ylphenyl)-3-((1-methylethyl){[frans-4-
(trifluoromethyOcyclohexyllcarbonylJaminoJ-i H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-b]pyridin-2-ylphenyl)-3-((1-methylethyl){[^ans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 H-pyrazole-4-carboxylic acid; 1 -(4-lmidazo[1 ,2-ύ]pyridazin-2-ylphenyl)-3-[[(^aA7S-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ,2-a]pyrazin-2-ylphenyl)-3-[[(^ans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[8-(Hydroxymethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-3-[[(frans-4- methylcyclohexyOcarbony^i-methylethyOamino]-! H-pyrazole-4-carboxylic acid;
1-{4-[8-(Aminomethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-3-[[(frans-4- methylcyclohexyOcarbony^i-methylethyOaminoJ-i H-pyrazole-4-carboxylic acid;
1 -(4-lmidazo[1 ^-alpyridin-θ-ylphenyO-S-IKfrans^-methylcyclohexyOcarbonylKI - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-(4-Furo[3,2-fe]pyridin-6-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](tetrahydro-2H- pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1-[4-(6- quinazolinyl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-(4-Furo[3,2-ό]pyridin-5-ylphenyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(6-Aminoimidazo[1 ,2-a]pyridin-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -[4-(6-Amino-1 ,3-benzoxazol-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid;
3-[(Cyclohexylacetyl)(1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1/-/- pyrazole-4-carboxylic acid; 3-[(Cyclopentylacetyl)(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H- pyrazole-4-carboxylic acid;
1-[4-(4-Amino-1-benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid;
1-[4-(7-Amino-1-benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid;
1 -[4-(7-Aminopyrazolo[1 ,5-a]pyrimidin-2-yl)phenyl]-3-((1 -methylethyl){[frans-4-
(trifluoromethyl)cyclohexyl]carbonyl}amino)-1 /-/-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 ,3-oxazol-2-ylmethyl)amino]-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 /-/-pyrazole-4-carboxylic acid; 3-{[(ϊrans-4-Methylcyclohexyl)carbonyl][2-(phenyloxy)ethyl]amino}-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(fΛans-4-Methylcyclohexyl)carbonyl](1 ,3-thiazol-4-ylmethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[(frans-4-Methylcyclohexyl)carbonyl][(4-methyl-1 ,2,5-oxadiazol-3-yl)methyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[2-(Ethyloxy)ethyl][(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[(frans-4-Methylcyclohexyl)carbonyl][(2-methyl-1 ,3-thiazol-4-yl)methyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-{Butyl[(frans-4-rnethylcyclohexyl)carbonyl]arnino}-1-(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid;
3-{[(3>5-Dimethyl-4-isoxazolyl)rnethyl][(^ans-4-methylcyclohexyl)carbonyl]amino}-1-(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{(3-Methylbutyl)[(frans-4-methylcyclohexyl)carbonyl]arnino}-1-(4-pyrazolo[1 ,5-a]pyrirriidin- 2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
S-Wδ-CI .I-DimethylethyO-i ^^-oxadiazol-S-ylJmethy^C^ans^- methylcyclohexyl)carbonyl]amino}-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4- carboxylic acid;
3-{[(frans-4-Methylcyclohexyl)carbonyl][(1 -methyl-1 H-imidazol-2-yl)methyl]amino}-1 -(4- pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-{[(5-Chloro-2-thienyl)methyl][(frans-4-methylcyclohexyl)carbonyl]amino}-1-(4-pyrazolo[1,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Chloropyrazolo[1 ,5-a]pyridin-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-[5-(6-Chloroimidazo[1 ,2-a]pyridin-2-yl)-2-thienyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -(5-lmidazo[1 ,2-a]pyrimidin-2-yl-2-thienyl)-3-[[(frans-4-nnethylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1 -(5-lmidazo[1 ,2-a]pyridin-2-yl-2-thienyl)-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; S-t^frans^-MethylcyclohexyOcarbony^i-methylethyOaminol-I^S-pyrazoloti .S-aJpyrimidin^- yl-2-thienyl)-1 /-/-pyrazole-4-carboxylic acid;
1-[4-(2-Chloroimidazo[2,1-ό][1 ,3]thiazol-6-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-(3-Chloro-4-pyrazolo[1 ,5-a]pyrimidin-2-ylphenyl)-3-[[(^ans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(3-methyl-4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dichlorophenyl)carbonyl](1-methylethyl)amino]-1-(4-furo[3,2-/j]pyridin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid; 3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -{4-[6-
(trifluoromethyl)imidazo[1 ,2-a]pyridin-2-yl]phenyl}-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(6-methylimidazo[1 ,2- a]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1 -[5-(1.S-Benzoxazol^-yO-S-furanylJ-S-^frans^-methylcyclohexyOcarbony^i - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dichlorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-imidazo[1 ,2-a]pyridin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dimethylphenyl)carbonyl](1-methylethyl)amino]-1-(4-furo[3,2-ύ]pyridin-2-ylphenyl)-
1 H-pyrazole-4-carboxylic acid; 3-[[(4-Bromo-2-fluorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(4-Bromo-2-methylphenyl)carbonyl](1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(4-Chloro-2-fluorophenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2,4-Dimethylphenyl)carbonyl](1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[(1-Benzofuran-5-ylcarbonyl)(1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[[(4-Bromo-2-chlorophenyl)∞rbonyl](1-methylethyl)arnino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
3-[[(2-Bromo-4-chlorophenyl)c^rbonyl](1-methylethyl)arnino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-2- ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Amino-6-chloro-3-pyridinyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(3-quinolinyl)phenyl]-1H- pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(6-quinoxalinyl)phenyl]-1/-/- pyrazole-4-carboxylic acid;
1-[4-(4-Amino-6-quinolinyl)phenyl]-3-[[(fraA7S-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-[4-(6-lsoquinolinyl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-
1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 -[4-(6-quinolinyl)phenyl]-1 H- pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(7-methylfuro[3,2- /)]pyridin-2-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-{3-Chloro-4-[6-(methyloxy)-3-pyridinyl]phenyl}-3-[[(frans-4- methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1H-pyrazole-4-carboxylic acid;
1 -[4-(7-Hydroxy-1 -benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 /-/-pyrazole-4-carboxylic acid; 1-[4-(5-Amino-1-benzofuran-2-yl)phenyl]-3-[[(frans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(7-Aminopyrazolo[1 ,5-a]pyrimidin-2-yl)-3-chlorophenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-[1,3]oxazolo[4,5-c]pyridin- 2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
1-{3-Chloro-4-[5-(methyloxy)-3-pyridinyl]phenyl}-3-[[(trans-4- methylcyclohexyl)carbonyl](tetrahydro-2H-pyran-4-yl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Hydroxy-3-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1-[4-(5-Acetyl-3-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[6-Amino-5-(hydroxymethyl)-3-pyridinyl]phenyl}-3-[[(trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[5-(Aminocarbonyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid ;
1-{4-[5-(Hydroxymethyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[6-(Hydroxymethyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -{4-[6-(Aminocarbonyl)-3-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(trans-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-[4-(1 H-pyrazolo[3,4-b]pyridin-
5-yl)phenyl]-1 H-pyrazole-4-carboxylic acid;
1-{4-[5-(Aminocarbonyl)-2-furanyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[{[2,4-Bis(methyloxy)phenyl]carbonyl}(1-methylethyl)amino]-1-(4-pyrazolo[1 ,5-a]pyrimidin-
2-ylphenyl)-1 H-pyrazole-4-carboxylic acid;
i-^-CΘ-Ethyl-y-hydroxy-S-methylpyrazolofi .S-alpyrimidin^-yOphenylJ-S-IfCtrans^- methylcyclohexyOcarbonylKI -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Acetyl-2-thienyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1-methylethyl)amino]-
1 H-pyrazole-4-carboxylic acid; 1 -(4-{2-[(1 R)-1 -Hydroxyethyl]-1 ,3-thiazol-5-yl}phenyl)-3-[[(trans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(5-Cyano-2-furanyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[2-(Aminocarbonyl)-3-thienyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[4-(Aminocarbonyl)-2-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[6-(Hydroxymethyl)-2-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid; 1 -[4-(5-Acetyl-2-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1 - methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-{4-[5-(Hydroxymethyl)-2-pyridinyl]phenyl}-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
1-[4-(6-Acetyl-3-pyridinyl)phenyl]-3-[[(trans-4-methylcyclohexyl)carbonyl](1- methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
3-[[(frans-4-Methylcyclohexyl)carbonyl](1-methylethyl)arnino]-1-{4-[6-(methyloxy)-3- pyridinyl]phenyl}-1 H-pyrazole-4-carboxylic acid;
3-[[(fraA7s-4-Methylcyclohexyl)carbonyl](1-methylethyl)amino]-1-(4-pyrido[2>3-b]pyrazin-7- ylphenyl)-1 H-pyrazole-4-carboxylic acid; 3-[{[4-Chloro-2-(methyloxy)phenyl]carbonyl}(1 -methylethyl)amino]-1 -(4-pyrazolo[1 ,5- a]pyrimidin-2-ylphenyl)-1 /-/-pyrazole-4-carboxylic acid;
1-[4-(6-Chloro-7-hydroxy-5-methylpyrazolo[1,5-a]pyrimidin-2-yl)phenyl]-3-[[(frans-4- methylcyclohexyl)carbonyl](1 -methylethyl)amino]-1 H-pyrazole-4-carboxylic acid;
and salts, solvates and esters, and individual enantiomers thereof where appropriate.
3. A method of treating or preventing viral infection which comprises administering to a subject in need thereof, an effective amount of at least one chemical entity chosen from compounds of Formula (I)
R1 represents -Rx-Rγ;
Rx represents phenyl (optionally substituted by halo, methyl, ethyl, methoxy or trifluoromethyl) or a 5 or 6-membered heteroaryl bonded through a ring carbon atom to the nitrogen atom of the pyrazole;
Rγ represents heteroaryl, bonded such that when Rx is phenyl, Rγ is in the para-position;
R2 represents phenyl substituted by one or more substituents selected from -C1-6alkyl, halo, -ORA, -SRA, -C(O)NR6R0, -C(O)RD, -CO2H, -CO2R0, -NRBRC, -NREC(0)RD, -NRECO2RD, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, and heterocyclyl, or two substituents on neighbouring carbon atoms of the phenyl ring together form a A-, 5- or 6-membered ring containing one or more heteroatoms selected from O, S and N; or R2 represents C^/cycloalkyl optionally substituted by one or more substitutents selected from -C1-6alkyl, =CH(CH2)tH, -0RA, -SRA, -C(0)NRBRc, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NREC02R°, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a C1-2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4spiro substituent; or R2 represents C5-7cycloalkylmethyl, the cycloalkyl portion being optionally substituted by one or more substitutents selected from -Chalky!, =CH(CH2)tH, - 0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NREC02R°, - NREC(0)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a Ci-2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4spiro substituent;
t represents O, 1 , 2, 3 or 4;
R3 represents heterocyclyl; or phenyl optionally substituted by one or more substituents selected from -C1-6alkyl, halo, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NRECO2R°, -NR6SO2R0, -NREC(0)NRFRG, -SO2NRFRG, - SO2R0, nitro, cyano, and heterocyclyl; or R3 represents -C^alkyl optionally substituted by one or more substituents selected from -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NRECO2R°, -NRES02R°, -NREC(O)NRFRG, -SO2NRFRG, - SO2R0, fluoro, nitro, cyano, oxo, phenyl, heteroaryl and heterocyclyl;
R4 represents hydrogen;
RA represents hydrogen, -C1-6alkyl, arylalkyl, heteroarylalkyl, aryl, heterocyclyl or heteroaryl;
RB and Rc independently represent hydrogen, -C1-6alkyl, aryl, heterocyclyl or heteroaryl; or RB and Rc together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
RD is selected from the group consisting of -C1-6alkyl, aryl, heterocyclyl, heteroaryl, arylalkyl, and heteroarylalkyl;
RE represents hydrogen or -C1-ealkyl;
RF and RG are independently selected from the group consisting of hydrogen, -C1-6alkyl, aryl, heteroaryl, arylalkyl, and heteroarylalkyl; or RF and RG together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
and salts, solvates and esters thereof.
4. A method as claimed in claim 3 which involves inhibiting HCV replication.
5. A method as claimed in claim 3 in which the chemical entity is administered in an oral dosage form.
6. At least one chemical entity chosen from compounds of Formula (I)
A represents hydroxy;
R1 represents -Rx-Rγ;
Rx represents phenyl (optionally substituted by halo, methyl, ethyl, methoxy or trifluoromethyl) or a 5 or 6-membered heteroaryl bonded through a ring carbon atom to the nitrogen atom of the pyrazole;
Rγ represents heteroaryl, bonded such that when Rx is phenyl, Rγ is in the para-position;
R2 represents phenyl substituted by one or more substituents selected from -C1-6alkyl, halo, -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)RD, -NRECO2R°,
-NREC(O)NRFRG, -SO2NRFRG, -SO2R0, nitro, cyano, and heterocyclyl, or two substituents on
neighbouring carbon atoms of the phenyl ring together form a 4-, 5- or 6-membered ring containing one or more heteroatoms selected from O, S and N; or R2 represents C5-7cycloalkyl optionally substituted by one or more substitutents selected from -C^alkyl, =CH(CH2),H, -ORA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)RD, -NRECO2RD, -NREC(O)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a Ci-2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4spiro substituent; or R2 represents C5-7cycloalkylmethyl, the cycloalkyl portion being optionally substituted by one or more substitutents selected from -C1-6alkyl, =CH(CH2),H, - 0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NREC02R°, - NREC(O)NRFRG, -SO2NRFRG, -SO2R0, fluoro, nitro, cyano, oxo, and heterocyclyl, or wherein two substituents may together form a C1-2alkylene bridge substituent, or wherein two alkyl substituents on the same carbon atom may together form a C3-4spiro substituent;
t represents O, 1 , 2, 3 or 4;
R3 represents heterocyclyl; or phenyl optionally substituted by one or more substituents selected from -C1-6alkyl, halo, -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(0)R°, -NR8CO2R0, -NRESO2R°, -NREC(O)NRFRG, -S02NRFRG, - SO2R0, nitro, cyano, and heterocyclyl; or R3 represents -Chalky! optionally substituted by one or more substituents selected from -0RA, -SRA, -C(O)NRBRC, -C(O)R0, -CO2H, -CO2R0, -NRBRC, -NREC(O)R°, -NRECO2R°, -NRESO2R°, -NREC(O)NRFRG, -SO2NRFRG, - SO2R0, fluoro, nitro, cyano, oxo, phenyl, heteroaryl and heterocyclyl;
R4 represents hydrogen;
RA represents hydrogen, -C1-6alkyl, arylalkyl, heteroarylalkyl, aryl, heterocyclyl or heteroaryl;
RB and Rc independently represent hydrogen, -Ci-6alkyl, aryl, heterocyclyl or heteroaryl; or RB and Rc together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
R° is selected from the group consisting of -C1-6alkyl, aryl, heterocyclyl, heteroaryl, arylalkyl, and heteroarylalkyl;
RE represents hydrogen or -C1-6alkyl;
RF and RG are independently selected from the group consisting of hydrogen, -C1-6alkyl, aryl, heteroaryl, arylalkyl, and heteroarylalkyl; or RF and RG together with the nitrogen atom to which they are attached form a 5 or 6 membered saturated cyclic group;
and salts, solvates and esters thereof;
for use in medical therapy.
7. A compound as claimed in claim 6 wherein the medical therapy is the treatment of viral infection.
8. A compound as claimed in claim 7 wherein the viral infection is HCV.
9. A pharmaceutical formulation comprising at least one chemical entity chosen from compounds of Formula (I) and pharmaceutically acceptable salts, solvates and esters thereof as defined in claim 1 in conjunction with at least one pharmaceutically acceptable diluent or carrier.
10. A process for the preparation of a compound of Formula (I) as defined in claim 1 , comprising deprotection of a compound of Formula (II)
11. A process as claimed in claim 10 in which A is ethoxy.
12. Use of at least one chemical entity chosen from compounds of Formula (I) and pharmaceutically acceptable salts, solvates and esters thereof as claimed in claim 1 , in the manufacture of a medicament for the treatment and/or prophylaxis of viral infection.
13. Use as claimed in claim 12 wherein the viral infection is HCV.
14. A combination comprising at least one chemical entity as defined in Claim 1 , together with at least one other therapeutically active agent.
15. A combination as claimed in Claim 14, wherein the other therapeutically active agent is selected from Interferon, ribavirin and/or an additional anti-HCV agent.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0519474A GB0519474D0 (en) | 2005-09-23 | 2005-09-23 | Compounds |
| GB0519474.1 | 2005-09-23 | ||
| GB0605467A GB0605467D0 (en) | 2006-03-17 | 2006-03-17 | Compounds |
| GB0605467.0 | 2006-03-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007039146A1 true WO2007039146A1 (en) | 2007-04-12 |
Family
ID=37744166
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2006/009238 Ceased WO2007039146A1 (en) | 2005-09-23 | 2006-09-21 | 4-carboxy pyrazole derivatives as anti-viral agents |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2007039146A1 (en) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009076747A1 (en) | 2007-12-19 | 2009-06-25 | Boehringer Ingelheim International Gmbh | Viral polymerase inhibitors |
| WO2010080874A1 (en) | 2009-01-07 | 2010-07-15 | Scynexis, Inc. | Cyclosporine derivative for use in the treatment of hcv and hiv infection |
| WO2010052448A3 (en) * | 2008-11-05 | 2010-09-10 | Ucb Pharma S.A. | Fused pyrazine derivatives as kinase inhibitors |
| US8030334B2 (en) | 2008-06-27 | 2011-10-04 | Novartis Ag | Organic compounds |
| US8080569B2 (en) * | 2006-11-16 | 2011-12-20 | Sanofi-Aventis | Imidazo[2,1-b]thiazoles and their use as pharmaceuticals |
| WO2012035421A3 (en) * | 2010-09-17 | 2012-09-07 | Purdue Pharma L.P. | Pyridine compounds and the uses thereof |
| JP2013540134A (en) * | 2010-10-20 | 2013-10-31 | ビオタ サイエンティフィック マネージメント ピーティーワイ リミテッド | Viral polymerase inhibitor |
| WO2014053450A1 (en) | 2012-10-02 | 2014-04-10 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| WO2015065243A1 (en) | 2013-11-01 | 2015-05-07 | Общество с ограниченной ответственностью "Уральский центр биофармацевтических технологий" | 2-methylsulphanyl-6-nitro-7-oxo-1, 2, 4-triazolo [5, 1-c] [1, 2, 4] triazinide l-arginine dihydrate active toward west nile virus |
| WO2015179392A1 (en) * | 2014-05-21 | 2015-11-26 | Bristol-Myers Squibb Company | 2-(aryl- or heteroaryl-)phenyl (aza)benzofuran compounds for the treatment of hepatitis c |
| EP2957562A1 (en) * | 2014-06-20 | 2015-12-23 | Masarykova univerzita | Pyrazolotriazines as inhibitors of nucleases |
| US20160060261A1 (en) * | 2011-03-30 | 2016-03-03 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1h-pyrazolo [3, 4-b] pyridine and pyrazolo [3, 4-b] pyridine derivatives as protein kinase inhibitors |
| CN107056653A (en) * | 2017-06-09 | 2017-08-18 | 丽珠医药集团股份有限公司 | Synthetic method of the blonanserin intermediate to fluorobenzoyl acetonitrile |
| CN109535092A (en) * | 2018-12-24 | 2019-03-29 | 温州大学 | A kind of preparation method of benzotriazole analog derivative |
| CN109734686A (en) * | 2019-01-07 | 2019-05-10 | 浙江万里学院 | A kind of catalytic synthesis method of 2-substituted benzofuran compounds |
| CN110498759A (en) * | 2019-09-12 | 2019-11-26 | 天津瑞岭化工有限公司 | The synthetic method of isoindoline ketone compound |
| CN114213424A (en) * | 2021-12-30 | 2022-03-22 | 杭州澳赛诺生物科技有限公司 | Synthetic method of furan [3, 2-b ] pyridine derivative |
| CN114907335A (en) * | 2022-02-25 | 2022-08-16 | 陕西维世诺新材料有限公司 | 2- (benzothiophene-2-yl) benzo [ d ] oxazole derivative, preparation method and application |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003037895A1 (en) * | 2001-11-02 | 2003-05-08 | Glaxo Group Limited | 4-(6-membered)-heteroaryl acyl pyrrolidine derivatives as hcv inhibitors |
| WO2004096210A1 (en) * | 2003-05-01 | 2004-11-11 | Glaxo Group Limited | Acylated indoline and tetrahydroquinoline derivatives as hcv inhibitors |
| WO2005092863A1 (en) * | 2004-03-26 | 2005-10-06 | Smithkline Beecham Corporation | 4-carbox pyrazole derivates useful as anti-viral agents |
| WO2006050034A1 (en) * | 2004-10-29 | 2006-05-11 | Schering Corporation | Substituted 5-carboxyamide pyrazoles and [1,2,4]triazoles as antiviral agents |
| WO2006100106A1 (en) * | 2005-03-21 | 2006-09-28 | Smithkline Beecham Corporation | 3-carboxy pyrroles as anti-viral agents |
-
2006
- 2006-09-21 WO PCT/EP2006/009238 patent/WO2007039146A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003037895A1 (en) * | 2001-11-02 | 2003-05-08 | Glaxo Group Limited | 4-(6-membered)-heteroaryl acyl pyrrolidine derivatives as hcv inhibitors |
| WO2004096210A1 (en) * | 2003-05-01 | 2004-11-11 | Glaxo Group Limited | Acylated indoline and tetrahydroquinoline derivatives as hcv inhibitors |
| WO2005092863A1 (en) * | 2004-03-26 | 2005-10-06 | Smithkline Beecham Corporation | 4-carbox pyrazole derivates useful as anti-viral agents |
| WO2006050034A1 (en) * | 2004-10-29 | 2006-05-11 | Schering Corporation | Substituted 5-carboxyamide pyrazoles and [1,2,4]triazoles as antiviral agents |
| WO2006100106A1 (en) * | 2005-03-21 | 2006-09-28 | Smithkline Beecham Corporation | 3-carboxy pyrroles as anti-viral agents |
Cited By (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8080569B2 (en) * | 2006-11-16 | 2011-12-20 | Sanofi-Aventis | Imidazo[2,1-b]thiazoles and their use as pharmaceuticals |
| WO2009076747A1 (en) | 2007-12-19 | 2009-06-25 | Boehringer Ingelheim International Gmbh | Viral polymerase inhibitors |
| US8791141B2 (en) | 2008-06-27 | 2014-07-29 | Novartis Ag | Organic compounds |
| US8030334B2 (en) | 2008-06-27 | 2011-10-04 | Novartis Ag | Organic compounds |
| US9242963B2 (en) | 2008-06-27 | 2016-01-26 | Novartis Ag | Organic compounds |
| WO2010052448A3 (en) * | 2008-11-05 | 2010-09-10 | Ucb Pharma S.A. | Fused pyrazine derivatives as kinase inhibitors |
| WO2010080874A1 (en) | 2009-01-07 | 2010-07-15 | Scynexis, Inc. | Cyclosporine derivative for use in the treatment of hcv and hiv infection |
| WO2012035421A3 (en) * | 2010-09-17 | 2012-09-07 | Purdue Pharma L.P. | Pyridine compounds and the uses thereof |
| US20130303526A1 (en) * | 2010-09-17 | 2013-11-14 | Purdue Pharma L.P. | Pyridine Compounds and the Uses Thereof |
| US9056832B2 (en) | 2010-09-17 | 2015-06-16 | Purdue Pharma L.P. | Pyridine compounds and the users thereof |
| US9611222B2 (en) | 2010-09-17 | 2017-04-04 | Purdue Pharma L.P. | Pyridine compounds and the uses thereof |
| JP2013540134A (en) * | 2010-10-20 | 2013-10-31 | ビオタ サイエンティフィック マネージメント ピーティーワイ リミテッド | Viral polymerase inhibitor |
| US9669028B2 (en) * | 2011-03-30 | 2017-06-06 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1H-pyrazolo [3, 4-B] pyridine and pyrazolo [3, 4-B] pyridine derivatives as protein kinase inhibitors |
| US20160060261A1 (en) * | 2011-03-30 | 2016-03-03 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1h-pyrazolo [3, 4-b] pyridine and pyrazolo [3, 4-b] pyridine derivatives as protein kinase inhibitors |
| WO2014053450A1 (en) | 2012-10-02 | 2014-04-10 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US10435374B2 (en) | 2012-10-02 | 2019-10-08 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US11548854B2 (en) | 2012-10-02 | 2023-01-10 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US11332448B2 (en) | 2012-10-02 | 2022-05-17 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US10961201B2 (en) | 2012-10-02 | 2021-03-30 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US10689348B2 (en) | 2012-10-02 | 2020-06-23 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US9802899B2 (en) | 2012-10-02 | 2017-10-31 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US9790227B2 (en) | 2013-11-01 | 2017-10-17 | Limited Liability Company “Ural Center Of Biopharmaceutical Technology” | 2-methylsulphanyl-6-nitro-7-oxo-1, 2, 4-triazolo [5, 1-C] [1, 2, 4] triazinide L-arginine dihydrate active toward west nile virus |
| WO2015065243A1 (en) | 2013-11-01 | 2015-05-07 | Общество с ограниченной ответственностью "Уральский центр биофармацевтических технологий" | 2-methylsulphanyl-6-nitro-7-oxo-1, 2, 4-triazolo [5, 1-c] [1, 2, 4] triazinide l-arginine dihydrate active toward west nile virus |
| US9840511B2 (en) | 2014-05-21 | 2017-12-12 | Bristol-Meyers Squibb Company | 2-(aryl or heteroaryl-)phenyl (AZA)benzofuran compounds for the treatment of hepatitis C |
| WO2015179392A1 (en) * | 2014-05-21 | 2015-11-26 | Bristol-Myers Squibb Company | 2-(aryl- or heteroaryl-)phenyl (aza)benzofuran compounds for the treatment of hepatitis c |
| EP2957562A1 (en) * | 2014-06-20 | 2015-12-23 | Masarykova univerzita | Pyrazolotriazines as inhibitors of nucleases |
| CN107056653A (en) * | 2017-06-09 | 2017-08-18 | 丽珠医药集团股份有限公司 | Synthetic method of the blonanserin intermediate to fluorobenzoyl acetonitrile |
| CN109535092A (en) * | 2018-12-24 | 2019-03-29 | 温州大学 | A kind of preparation method of benzotriazole analog derivative |
| CN109535092B (en) * | 2018-12-24 | 2021-05-11 | 温州大学 | Preparation method of benzotriazole derivatives |
| CN109734686A (en) * | 2019-01-07 | 2019-05-10 | 浙江万里学院 | A kind of catalytic synthesis method of 2-substituted benzofuran compounds |
| CN109734686B (en) * | 2019-01-07 | 2022-07-01 | 浙江万里学院 | A kind of catalytic synthesis method of 2-substituted benzofuran compounds |
| CN110498759A (en) * | 2019-09-12 | 2019-11-26 | 天津瑞岭化工有限公司 | The synthetic method of isoindoline ketone compound |
| CN114213424A (en) * | 2021-12-30 | 2022-03-22 | 杭州澳赛诺生物科技有限公司 | Synthetic method of furan [3, 2-b ] pyridine derivative |
| CN114907335A (en) * | 2022-02-25 | 2022-08-16 | 陕西维世诺新材料有限公司 | 2- (benzothiophene-2-yl) benzo [ d ] oxazole derivative, preparation method and application |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2007088148A1 (en) | Derivatives of thiophene carboxilic acid as antiviral agent | |
| CN102427729B (en) | Inhibitors of hepatitis C virus replication | |
| WO2007039146A1 (en) | 4-carboxy pyrazole derivatives as anti-viral agents | |
| US7348425B2 (en) | Inhibitors of HCV replication | |
| US7456165B2 (en) | HCV NS5B inhibitors | |
| EP2086966B1 (en) | 2-carboxy thiophene derivatives as anti viral agents | |
| US7153848B2 (en) | Inhibitors of HCV replication | |
| AU2005259894C1 (en) | Pyrrolotriazine kinase inhibitors | |
| AU2008273017B2 (en) | Heterocyclic compounds useful as Raf kinase inhibitors | |
| KR100851178B1 (en) | Inhibitors of hepatitis C virus RNA-dependent RNA polymerase, compositions and treatments using the same | |
| US20120121540A1 (en) | Certain Nitrogen Containing Bicyclic Chemical Entities For Treating Viral Infections | |
| EP1971599A1 (en) | Antiviral 2-carboxy-thiophene compounds | |
| AU2009291993A1 (en) | Compounds for the treatment of hepatitis C | |
| WO2008017688A1 (en) | 2-carboxy thiophene derivatives as anti-viral agents | |
| JP2014510105A (en) | Azole compounds as PIM inhibitors | |
| CA2593450A1 (en) | Indole derivatives for treating viral infections | |
| MX2010010268A (en) | Polysubstituted derivatives of 6-heteroaryl-imidazo[l,2- î] pyridines, and preparation and therapeutic use thereof. | |
| KR20160128420A (en) | Trka kinase inhibitors, compositions and methods thereof | |
| JP2013521237A (en) | Inhibitor of hepatitis C virus NS5B polymerase | |
| WO2009155388A1 (en) | Imidazopyridine and imidazopyrazine compounds useful as kinase inhibitors | |
| EP2307411A1 (en) | Triazolopyridine compounds useful as kinase inhibitors | |
| WO2008043791A2 (en) | Thiophene derivatives for treating hepatitis c | |
| TW202340181A (en) | Heterocyclic compounds and uses thereof | |
| EP1663105B1 (en) | Cycloalkyl heterocycles for treating hepatitis c virus | |
| WO2008125599A1 (en) | 2-carboxy thiophene derivatives as anti-viral agents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 06792238 Country of ref document: EP Kind code of ref document: A1 |