WO2005063760A1 - C-8 LINKED PYRROLO[2,1-c][1,4]BENZODIAZEPINE-ACRIDONE/ACRIDINE HYBRIDS - Google Patents
C-8 LINKED PYRROLO[2,1-c][1,4]BENZODIAZEPINE-ACRIDONE/ACRIDINE HYBRIDS Download PDFInfo
- Publication number
- WO2005063760A1 WO2005063760A1 PCT/IN2003/000464 IN0300464W WO2005063760A1 WO 2005063760 A1 WO2005063760 A1 WO 2005063760A1 IN 0300464 W IN0300464 W IN 0300464W WO 2005063760 A1 WO2005063760 A1 WO 2005063760A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- methoxy
- oxy
- hybrid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*(CCOc(cc(c1c2)N=C[C@](CC(C3)=C)N3C1=O)c2OC)Oc(c(OC)c1)cc(N=CC(C2)N3CC2=C)c1C3=O Chemical compound C*(CCOc(cc(c1c2)N=C[C@](CC(C3)=C)N3C1=O)c2OC)Oc(c(OC)c1)cc(N=CC(C2)N3CC2=C)c1C3=O 0.000 description 5
- IWNPELIYRIZFDK-UHFFFAOYSA-N CC(c(cccc12)c1Nc(cccc1)c1C2=O)=O Chemical compound CC(c(cccc12)c1Nc(cccc1)c1C2=O)=O IWNPELIYRIZFDK-UHFFFAOYSA-N 0.000 description 1
- CYTGKLYIGZYXGV-UHFFFAOYSA-N CC(c1c2nc(cccc3)c3cc2ccc1)=O Chemical compound CC(c1c2nc(cccc3)c3cc2ccc1)=O CYTGKLYIGZYXGV-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
- A61K31/5517—1,4-Benzodiazepines, e.g. diazepam or clozapine condensed with five-membered rings having nitrogen as a ring hetero atom, e.g. imidazobenzodiazepines, triazolam
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
Definitions
- the present invention provides novel pyrrolo-[2,l-c][l,4]benzodiazepine hybrids useful as anti-tumour agents.
- the present invention also provides a process for the preparation of new pyrrolo[2,l-c][l,4]benzodiazepine hybrids as antitumour agents.
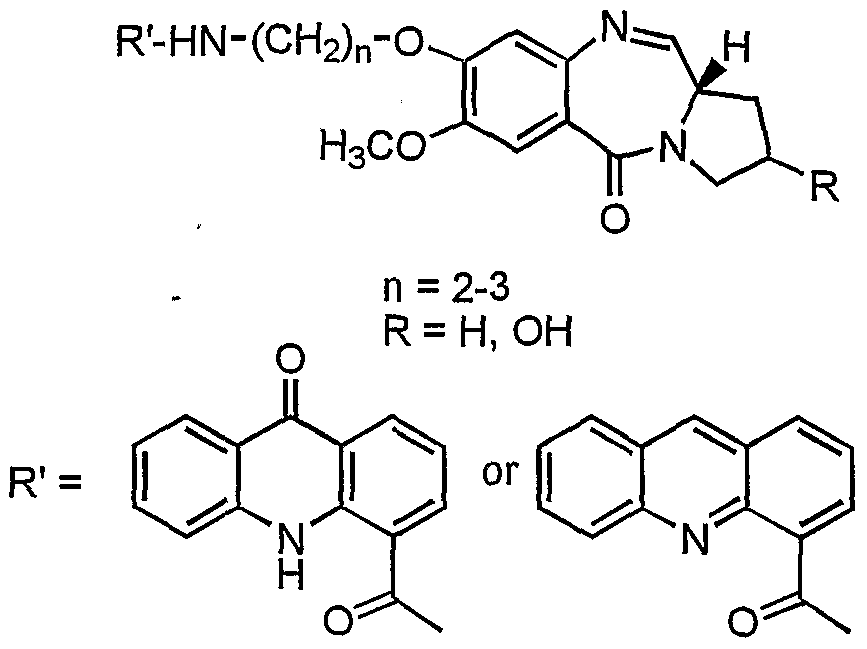
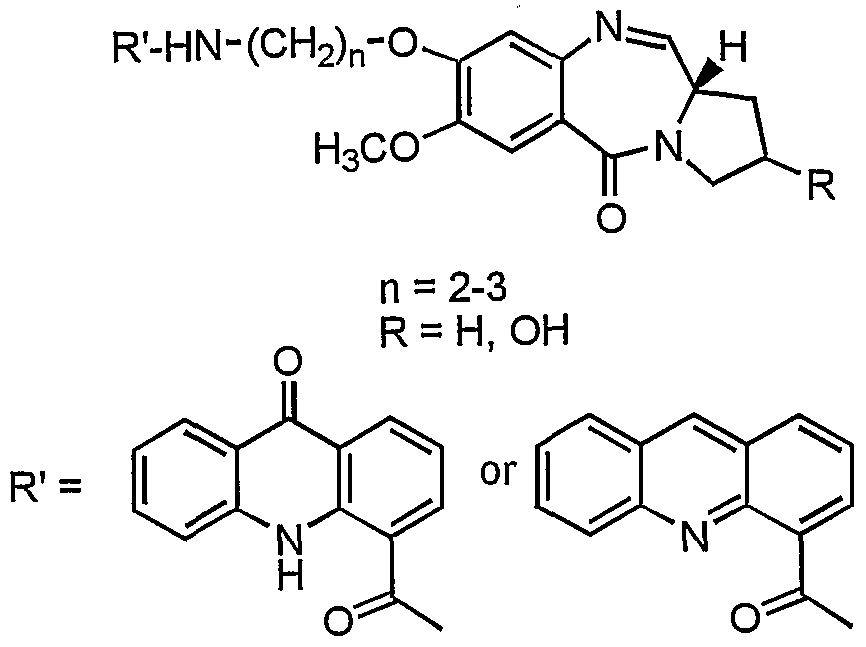
- the present invention provides a process for the preparation of 7-methoxy-8-[n'- (4"-acridonylcarboxamido)alkyl]-oxy-( 11 aS) ⁇ 1,2,3,11 a-tetraydro-5H- ⁇ yrrolo [2, 1 -c] [l,4]benzodiazepin-5-one and 7-methoxy-8-[n'-(4"-acridinylcarboxamido)-alkyl]-oxy- (llaS)-l,2,3,lla-tetraydro-5H-pyrrolo[2,l-c][l,4]benzodiazepin-5-one with aliphatic chain length variation of these compounds and it also describes the DNA binding, anticancer (antitumour) activity.
- the structural formula of the novel pyrrolo[2,l-c]-[l,4]benzodiazepines of the invention is given below:
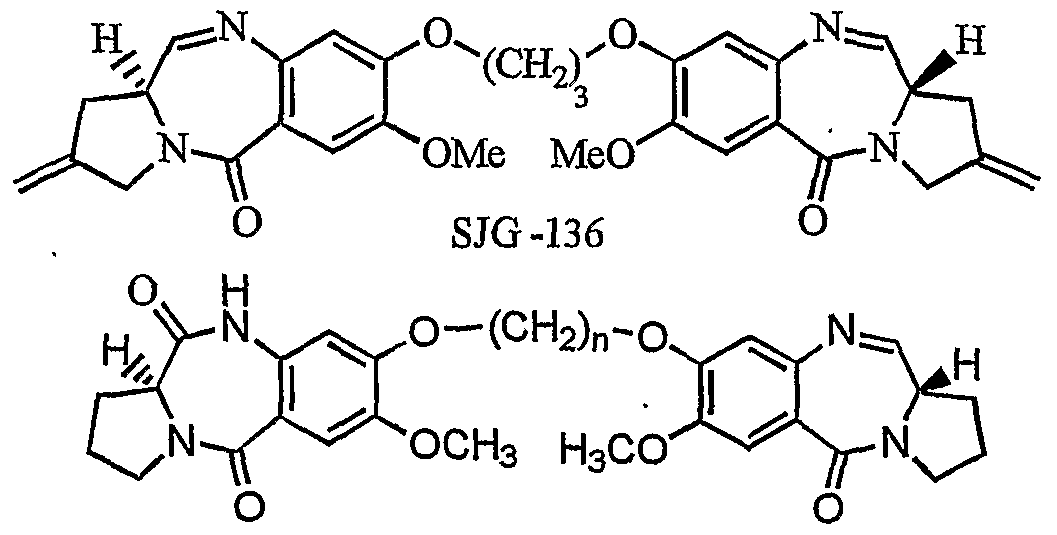
- PBD dimers have been developed that comprises two C2-exo-methylene substituted DC-81 subunits tethered through their C ⁇ 8 position via an inert propanedioxy linker (Gregson, S. I; Howard, P. W.; Hartely, J. A.; Brooks, N. A.; Adams, L. I; Jenkins, T. C; Kelland, L. R. and Thurston, D. E. J. Med. Chem. 2001, 44, 737). Recently, a noncross- linking mixed imine-amide PBD dimers have been synthesized that have significant DNA binding ability and potent antitumour activitiy.
- Naturally occurring pyrrolo[2,l-c][l,4]benzodiazepines belong to a group of antitumour antibiotics derived from Streptomyces species. Recently, there is much impetus for the PBD systems as they can recognize and bind to specific sequence of DNA.
- the main object of the present invention is to provide new pyrrolo[2,l-c][l,4]- benzodiazepine hybrids useful as antitumour agents.
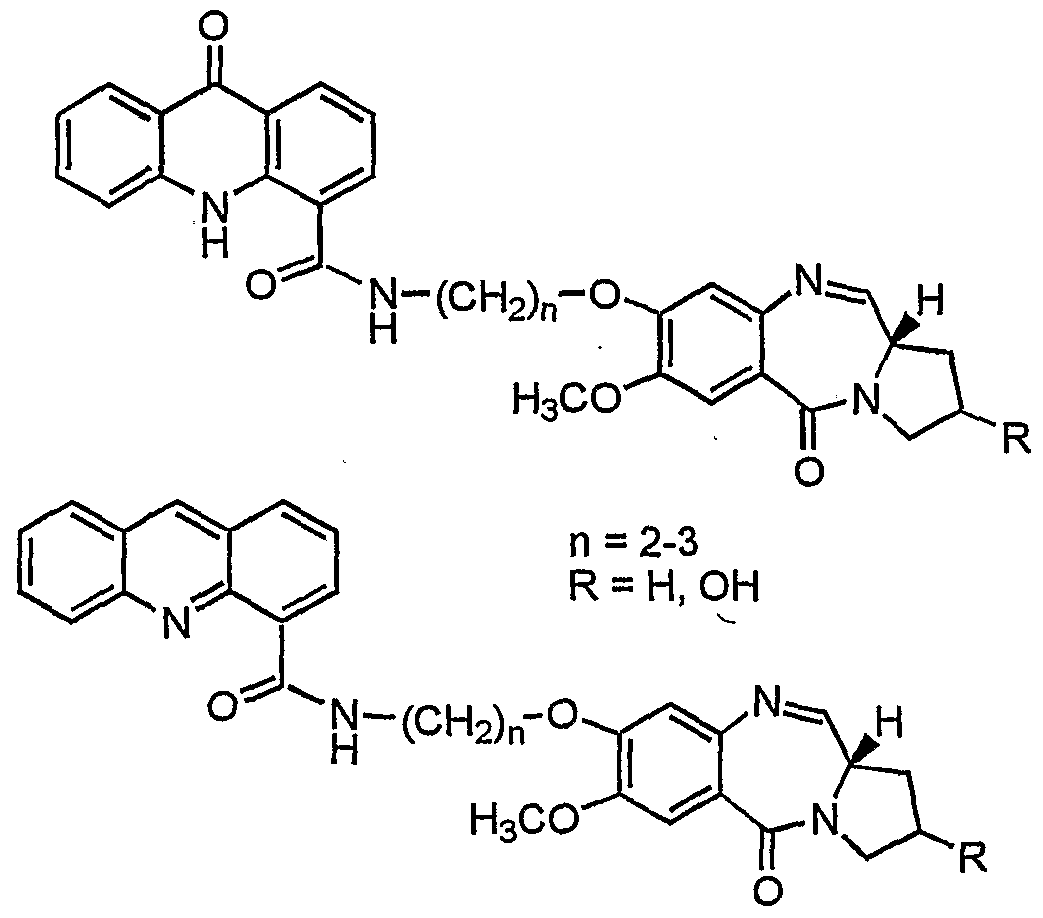
- the compound of the invention is selected from the group consisting .of 7-Methoxy-8-[2'-(4"-acridonylcarboxamido)ethyl]-oxy-
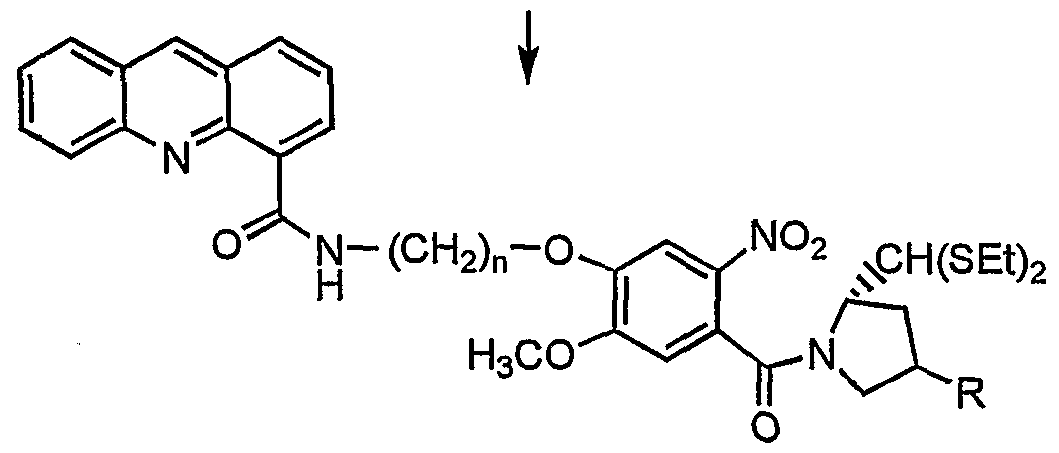
- Formula VII the process comprising reacting an acridone or an acridine acid with (2S)-N-[4-(n'- aminoalkyloxy)-5-methoxy-2-nitrobenzoyl]-pyrrolidine-2-carboxaldehyde diethyl thioacetal of formula I
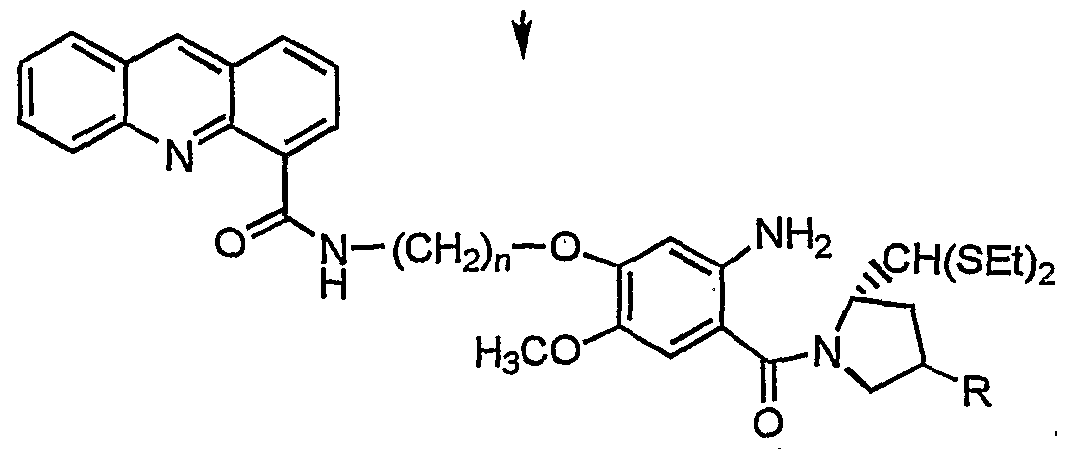
- Formula V isolating the compound of formula Il/formula V and then reducing the compounds of formula Il/formula V with SnCl 2 .2H 2 O in presence of an organic solvent up to a reflux temperature, isolating the (2S)-N- ⁇ 4-[n'-(4"-acridonylcarboxamido)-alkyl]-oxy-5-methoxy-2- aminobenzoyl ⁇ pyrroU-dine-2-carboxaldehydediethylthioacetal of formula III/(2S)-N- ⁇ 4-[n'- (4"-acridinylcarbox-arm ⁇ o)-alkyl]-oxy-5-methoxy-2-aminobenzoyl ⁇ pyrroUdine-2- carboxaldehyde diethyl thioacetal of formula VI where n is 2-3 by known methods,
- Formula HI Formula VI reacting compound of formula Ill/formula VI with a known deprotecting agents in a conventional manner to give novel pyrrolo[2,l-c][l,4]benzodiazepine hybrids of formula IV/formula VII wherein n and R are as stated above.
- the organic solvent used for the reaction of the acridone/acridine acid with compound of formula I comprises dimethyl furan.
- the compound of formula Il/formula V is isolated by washing with saturated NaHCO 3 , brine, drying and evaporation of the solvent.
- the organic solvent used during the reduction of compound of formula Il/formula V comprises methanol.
- the compound of formula Ill/formula V is isolated by adjusting the pH of the reaction mixture to about pH 8 with a saturated NaHCO 3 solution, diluting with ethyl acetate, filtering through ceUte and extracted an organic phase and drying the organic phase over Na 2 SO 4 .
- the deprotecting agent used for obtaining the compound of formula IV/formula Nil comprises HgCl 2 and CaCO 3 in MeC ⁇ -water (4:1).
- the process of the invention comprises reacting, an acridone or an acridine acid with (2S)- ⁇ -[4-(n'-ammoalkyloxy)-5-methoxy-2-nitrobenzoyl]-pyrroUdine-2-carboxaldehyde diethyl thioacetal of formula I
- the organic solvent used for the reaction of the acridone/acridine acid with compound of formula I comprises dimethyl furan and the compound of formula Il/formula V is isolated by washing with saturated NaHCO 3 , brine, drying and evaporation of the solvent.
- the organic solvent used during the reduction of compound of formula Il/formula V comprises methanol and the compound of formula Ill/formula V is isolated by adjusting the pH of the reaction mixture to about pH 8 with a saturated NaHCO 3 solution, diluting with ethyl acetate, filtering through celite and extracted an organic phase and drying the organic phase over Na 2 SO 4 .
- the deprotecting agent used for obtaining the compound of formula IV/formula VII comprises HgCl 2 and CaCO 3 in MeCN-water (4: 1).
- the precursors acridone acid (AtweU, G. J.; Cain, B. F.; Baguley, B. C; Finlay, G. J.; Denny, W. A. J. Med. Chem. 1984, 27, 1481), acridine acid (AtweU, G. J.; Rewcastle, G. W.; Baguley, B. C; Denny, W. A. J. Med Chem.
- Example 2 Compound acridone acid (0.239 g, 1 mmol) was taken in dry DMF (10 mL), EDCI (0.203g, 1.5 mmol) and HOBt(0.288 g, 1.5mmol) was added and the mixture was cooled at 0-5 °C and the mixture was stirred for 30 min. A solution of (4R)-hydroxy-(2S)-N-[4-(2'- aminoethyl)-oxy-5-methoxy-2-nitrobenzoyl]pyrrolidine-2-carboxaldehyde diethylthioacetal of formula I (0.459 g, 1 mmol) in DMF was added to it at the same temperature and the solution was stirred at room temperature for overnight.
- Example 4 Compound acridone acid (0.239 g, 1 mmol) was taken in dry DMF (10 mL), EDCI (0.203 g, 1.5 mmol)and HOBt (0.288 g, 1.5mmol) was added and the mixture was cooled at 0-5 °C and the mixture was stirred for 30 min. A solution of (4R)-hydroxy-(2S)-N-[4-(3'- aminopropyl)-oxy-5-methoxy-2-nitrobenzoyl]pyrroUdine-2-carboxaldehyde diethyl thioacetal of formula I (0.473 g, 5 mmol) in DMF was added to it at the same temperature and the solution was stirred at room temperature for overnight.
- Example 6 Compound acridine acid (0.223 g, 1 mmol) was taken in dry DMF (10 mL), EDCI (0.203- g, 1.5 mmol)and HOBt (0.288 g, 1.5mmol) was added and the mixture was cooled at 0-5 °C and the mixture was stirred for 30 min. A solution of (4R)-hydroxy-(2S)-N-[4-(2'- aminoethyl)-oxy-5-methoxy-2-nitrobenzoyl]pyrrolidine-2-carboxaldehyde diethyl thioacetal of formula I (0.459 g, 1 mmol) in DMF was added to it at the same temperature and the solution was stirred at room temperature for overnight.
- Example 7 Compound acridine acid (0.223 g, 1 mmol) was taken in dry DMF (10 mL), EDCI (0.203 g, 1.5 mmol)and ⁇ OBt (0.288 g, 1.5mmol) was added and the mixture was cooled at 0-5 °C and the mixture was stirred for 30 min. A solution of (2S)-N-[4-(3'-amino-propyl)oxy- 5-methoxy-2-nitrobenzoyl]pyrroUdine-2-carboxaldehyde diethyl thioacetal of formula I (0.457 g, 1 mmol) in DMF was added to it at same temperature and the solution was stirred at room-temperature for overnight.
- IV 0 0 10 a One dose of IV at 10 molar concentration
- formula IV has shown to possess ⁇ 10 nano molar potency (at the LC 50 level) against one melanoma cancer (UACC-62) and 0.1 micro molar potency against colon cancer (HCC- 2998), CNS cancer (SNB-75), breast cancer (MDA-MB-435) and also have ⁇ 10 micro molar potency against two melanoma cancer cell lines (LOXIMVI, M14) and one renal cancer (SN12C).
- Table 2. logio GI50 logio TGI and logio LC50 mean graphs midpoints(MGJV ⁇ D) of in vitro cytotoxicity data for the compound IV against human tumour cell lines.
- Each cancer type represents the average of six to nine different cancer ceU lines. 333NF/03 MEAN GRAPH
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Epidemiology (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2003300720A AU2003300720A1 (en) | 2003-12-31 | 2003-12-31 | C-8 linked pyrrolo(2,1-c)(1,4)benzodiazepine-acridone/acridine hybrids |
| GB0614754A GB2424884B (en) | 2003-12-31 | 2003-12-31 | C-8 linked pyrrolo[2,1-c][1,4]benzodiazepine-acridone/acridine hybrids |
| PCT/IN2003/000464 WO2005063760A1 (en) | 2003-12-31 | 2003-12-31 | C-8 LINKED PYRROLO[2,1-c][1,4]BENZODIAZEPINE-ACRIDONE/ACRIDINE HYBRIDS |
| JP2005512763A JP4718328B2 (en) | 2003-12-31 | 2003-12-31 | C8-linked pyrrolo [2,1-c] [1,4] benzodiazepine-acridone / acridine hybrid |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/IN2003/000464 WO2005063760A1 (en) | 2003-12-31 | 2003-12-31 | C-8 LINKED PYRROLO[2,1-c][1,4]BENZODIAZEPINE-ACRIDONE/ACRIDINE HYBRIDS |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2005063760A1 true WO2005063760A1 (en) | 2005-07-14 |
Family
ID=34717581
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2003/000464 Ceased WO2005063760A1 (en) | 2003-12-31 | 2003-12-31 | C-8 LINKED PYRROLO[2,1-c][1,4]BENZODIAZEPINE-ACRIDONE/ACRIDINE HYBRIDS |
Country Status (4)
| Country | Link |
|---|---|
| JP (1) | JP4718328B2 (en) |
| AU (1) | AU2003300720A1 (en) |
| GB (1) | GB2424884B (en) |
| WO (1) | WO2005063760A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009016647A1 (en) * | 2007-08-01 | 2009-02-05 | Council Of Scientific & Industrial Research | Pyrrolo [2,1-c][1, 4] benzodiazepine-glycoside prodrugs useful as a selective anti tumor agent |
| JP2009515870A (en) * | 2005-11-10 | 2009-04-16 | カウンスィル オブ サイエンティフィック アンド インダストリアル リサーチ | Novel pyrrolo [2,1-c] [1,4] benzodiazepine hybrids and methods for their preparation |
| US11583590B2 (en) | 2017-09-29 | 2023-02-21 | Daiichi Sankyo Company, Limited | Antibody-pyrrolobenzodiazepine derivative conjugate and method of use thereof for treating a tumor |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0098098A2 (en) * | 1982-06-25 | 1984-01-11 | Development Finance Corporation Of New Zealand | Acridinecarboxamide compounds |
| WO2000012508A2 (en) * | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolbenzodiazepines |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9621795D0 (en) * | 1996-10-18 | 1996-12-11 | Xenova Ltd | Pharmaceutical compounds |
-
2003
- 2003-12-31 AU AU2003300720A patent/AU2003300720A1/en not_active Abandoned
- 2003-12-31 JP JP2005512763A patent/JP4718328B2/en not_active Expired - Fee Related
- 2003-12-31 WO PCT/IN2003/000464 patent/WO2005063760A1/en not_active Ceased
- 2003-12-31 GB GB0614754A patent/GB2424884B/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0098098A2 (en) * | 1982-06-25 | 1984-01-11 | Development Finance Corporation Of New Zealand | Acridinecarboxamide compounds |
| WO2000012508A2 (en) * | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolbenzodiazepines |
Non-Patent Citations (5)
| Title |
|---|
| GAMAGE S W ET AL: "A NEW SYNTHESIS OF SUBSTITUTED ACRIDINE-4CARBOXYLIC ACIDS AND THE ANTICANCER DRUG N-U2-(DIMETHYLAMINO)ETHYLACRIDINE-4-CARBOXAMIDE", TETRAHEDRON LETTERS, ELSEVIER SCIENCE PUBLISHERS, AMSTERDAM, NL, vol. 38, no. 4, 27 January 1997 (1997-01-27), pages 699 - 702, XP002051605, ISSN: 0040-4039 * |
| KAMAL A ET AL: "Design, Synthesis, and Evaluation of New Non-Crosslinking Pyrrolobenzodiazepine Dimers with Efficient DNA Binding Ability and Potent Antitumor Activity", JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 45, 2002, pages 4679 - 4688, XP002249808, ISSN: 0022-2623 * |
| SCHOFIELD P C ET AL: "Metabolism of N-(2-(dimethylamino)ethyl)acridine-4-carboxamide in cancer patients undergoing a phase I clinical trial", CANCER CHEMOTHERAPY AND PHARMACOLOGY, SPRINGER VERLAG, BERLIN, DE, vol. 44, no. 1, July 1999 (1999-07-01), pages 51 - 58, XP002238691, ISSN: 0344-5704 * |
| THURSTON D E ET AL: "SYNTHESIS OF DNA-INTERACTIVE PYRROLO2,1-C1,4BENZODIAZEPINES", CHEMICAL REVIEWS, AMERICAN CHEMICAL SOCIETY. EASTON, US, vol. 94, 1994, pages 433 - 465, XP001026336, ISSN: 0009-2665 * |
| THURSTON D E ET AL: "Synthesis of Sequence-Selective C8-Linked Pyrrolo(2,1-c)(1,4)benzodia zepine DNA Interstrand Cross-Linking Agents", JOURNAL OF ORGANIC CHEMISTRY, AMERICAN CHEMICAL SOCIETY. EASTON, US, vol. 61, no. 23, 1996, pages 8141 - 8147, XP002272010, ISSN: 0022-3263 * |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009515870A (en) * | 2005-11-10 | 2009-04-16 | カウンスィル オブ サイエンティフィック アンド インダストリアル リサーチ | Novel pyrrolo [2,1-c] [1,4] benzodiazepine hybrids and methods for their preparation |
| WO2009016647A1 (en) * | 2007-08-01 | 2009-02-05 | Council Of Scientific & Industrial Research | Pyrrolo [2,1-c][1, 4] benzodiazepine-glycoside prodrugs useful as a selective anti tumor agent |
| US11583590B2 (en) | 2017-09-29 | 2023-02-21 | Daiichi Sankyo Company, Limited | Antibody-pyrrolobenzodiazepine derivative conjugate and method of use thereof for treating a tumor |
| US11628223B2 (en) | 2017-09-29 | 2023-04-18 | Daiichi Sankyo Company, Limited | Antibody-drug conjugates comprising substituted benzo[e]pyrrolo[1,2-α][1,4]diazepines |
| US12246196B2 (en) | 2017-09-29 | 2025-03-11 | Daiichi Sankyo Company, Limited | Antibody-drug conjugates comprising substituted benzo[e]pyrrolo[1,2-a][1,4]diazepines |
| US12350344B2 (en) | 2017-09-29 | 2025-07-08 | Daiichi Sankyo Company, Limited | Methods of treating a tumor by administering a claudin-6 (CLDN6) or CLDN9 antibody-pyrrolobenzodiazepine derivative conjugate |
Also Published As
| Publication number | Publication date |
|---|---|
| GB0614754D0 (en) | 2006-09-06 |
| AU2003300720A1 (en) | 2005-07-21 |
| GB2424884A (en) | 2006-10-11 |
| GB2424884B (en) | 2008-06-11 |
| JP2007528344A (en) | 2007-10-11 |
| JP4718328B2 (en) | 2011-07-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2520898C (en) | Non-cross-linking pyrrolo(2,1-c)(1,4)benzodiazepines as potential antitumour agents and process thereof | |
| EP2061795B1 (en) | Pyrrolo[2,1-c][1,4]benzodiazepine hybrids and a process for the preparation thereof | |
| US7189710B2 (en) | C2-fluoro pyrrolo [2,1−c][1,4]benzodiazepine dimers | |
| WO2011117668A9 (en) | Carbazole linked pyrrolo[2,1-c][1,4]benzodiazepine hybrids as potential anticancer agents and process for the preparation thereof | |
| EP2265613B1 (en) | Quinazoline linked pyrrolo[2,1-c][1, 4]benzodiazepine hybrids as potential anticancer agents and process for the preparation thereof | |
| US7056913B2 (en) | C8—linked pyrrolo[2,1-c][1,4]benzodiazepine-acridone/acridine hybrids | |
| US7312210B2 (en) | Pyrrolo[2,1-c][1,4]benzodiazepine compounds and processes for the preparation thereof | |
| US6800622B1 (en) | Pyrene-linked pyrrolo[2,1-c][1,4]benzodiazepine hybrids useful as anti-cancer agents | |
| US6951853B1 (en) | Process for preparing pyrrolo[2, 1-c] [1,4] benzodiazepine hybrids | |
| EP1608663B1 (en) | Pyrrolo (2,1-c)(1,4) benzodiazepines dimers as antitumour agents and process thereof | |
| EP1608650B1 (en) | Pyrene-linked pyrrolo(2,1-c)(1,4)benzodiazepine derivatives useful as anticancer agents | |
| US7015215B2 (en) | Pyrrolo[2,1-c][1,4] benzodiazepines compounds and process thereof | |
| WO2005063760A1 (en) | C-8 LINKED PYRROLO[2,1-c][1,4]BENZODIAZEPINE-ACRIDONE/ACRIDINE HYBRIDS | |
| WO2005063759A1 (en) | PROCESS FOR PREPARING PYRROLO[2, 1-c] [1, 4] BENZODIAZEPINE HYBRIDS | |
| US6683073B1 (en) | Pyrimidine linked pyrrolo[2,1-c][1,4]benzodiazepines as potential antitumour agents | |
| US8461150B2 (en) | Chalcone linked pyrrolo[2,1-c][1, 4]benzodiazepine hybrids as potential anticancer agents and process for the preparation thereof | |
| EP2271648B1 (en) | Isoxazoline linked pyrrolo [2,1-c][1,4]benzodiazepine hybrids as potential anticancer agents and the process for preparattion thereof | |
| WO2010052732A1 (en) | Cinnamido-pvrrolor[2,1-c][1,4]benzodiazepines as potential anticancer agents and process for the preparation thereof | |
| WO2004087712A1 (en) | NEW PYRIMIDINE LINKED PYRROLO[2,1-c][1,4]BENZODIAZEPINES AS POTENTIAL ANTITUMOUR AGENTS | |
| WO2009118749A1 (en) | Benzophenone-piperazine linked pyrrolo[2,1-c][1,4]benzodiazepine hybrids as potential anticancer agents and process for the preparation thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 1267/DELNP/2005 Country of ref document: IN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005512763 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 0614754.0 Country of ref document: GB Ref document number: 0614754 Country of ref document: GB |
|
| 122 | Ep: pct application non-entry in european phase |