WO2004083242A1 - Human granulocyte-colony stimulating factor conjugate having enhanced stability in blood and process for the preparation thereof - Google Patents
Human granulocyte-colony stimulating factor conjugate having enhanced stability in blood and process for the preparation thereof Download PDFInfo
- Publication number
- WO2004083242A1 WO2004083242A1 PCT/KR2004/000569 KR2004000569W WO2004083242A1 WO 2004083242 A1 WO2004083242 A1 WO 2004083242A1 KR 2004000569 W KR2004000569 W KR 2004000569W WO 2004083242 A1 WO2004083242 A1 WO 2004083242A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- peg
- conjugate
- csf
- csf derivative
- derivative
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21J—FIBREBOARD; MANUFACTURE OF ARTICLES FROM CELLULOSIC FIBROUS SUSPENSIONS OR FROM PAPIER-MACHE
- D21J3/00—Manufacture of articles by pressing wet fibre pulp, or papier-mâché, between moulds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/52—Cytokines; Lymphokines; Interferons
- C07K14/53—Colony-stimulating factor [CSF]
- C07K14/535—Granulocyte CSF; Granulocyte-macrophage CSF
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B30—PRESSES
- B30B—PRESSES IN GENERAL
- B30B3/00—Presses characterised by the use of rotary pressing members, e.g. rollers, rings, discs
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B30—PRESSES
- B30B—PRESSES IN GENERAL
- B30B9/00—Presses specially adapted for particular purposes
- B30B9/28—Presses specially adapted for particular purposes for forming shaped articles
-
- G—PHYSICS
- G09—EDUCATION; CRYPTOGRAPHY; DISPLAY; ADVERTISING; SEALS
- G09B—EDUCATIONAL OR DEMONSTRATION APPLIANCES; APPLIANCES FOR TEACHING, OR COMMUNICATING WITH, THE BLIND, DEAF OR MUTE; MODELS; PLANETARIA; GLOBES; MAPS; DIAGRAMS
- G09B1/00—Manually or mechanically operated educational appliances using elements forming, or bearing, symbols, signs, pictures, or the like which are arranged or adapted to be arranged in one or more particular ways
- G09B1/32—Manually or mechanically operated educational appliances using elements forming, or bearing, symbols, signs, pictures, or the like which are arranged or adapted to be arranged in one or more particular ways comprising elements to be used without a special support
- G09B1/36—Manually or mechanically operated educational appliances using elements forming, or bearing, symbols, signs, pictures, or the like which are arranged or adapted to be arranged in one or more particular ways comprising elements to be used without a special support the elements being connectible by corresponding projections and recesses
Definitions
- the present invention relates to a conjugate of a human granulocyte- colony stimulating factor ( G-CSF) derivative modified with a polyethylene glycol (PEG) and a method for the preparation thereof.
- G-CSF human granulocyte- colony stimulating factor
- PEG polyethylene glycol
- G-CSFs Granulocyte-colony stimulating factors
- cytokines that stimulate the expansion and differentiation of bone marrow progenitor cells and leukocytes. They also activate mature myeloid cells to mediate a variety of anti-microbial activities and inflammatory responses. They have been widely used in clinical transplantation which induces hematopoietic stem cell mobilization and increases myeloid progenitor cell population. It has been reported that the purified G-CSF is highly active in inducing granulocyte differentiation and suppresses stem cell self-replication in myeloid leukemia cells (Nicola et. al, JBC 258(14): 9017-9023, 1983).
- G-CSF is a glycoprotein, consisting of 174 amino acids, which plays an important role in hematopoietic cell proliferation, differentiation of hematopoietic precursor cells, and activation of mature neutrophilic granulocytes.
- hG-CSF gene transcribes into two kinds of mRNA consisting of 207 and 204 amino acids, respectively, that contain a leader sequence consisting of 30 amino acids for their secretion at the terminal amino residue (Nagata et al., Nature 319: 415-418, 1986; Nagata et al., EMBO J. 5(3): 575-581, 1986; Souza et al, Science 232: 61-65, 1986).
- the difference in the two hG-CSFs is the presence or absence of three amino acids (Val-Ser-Glu) at the 35 th amino acid residue starting from the terminal amino residue. Accordingly, a mature hG- CSF protein has 174 or 177 amino acids.
- hG-CSF consisting of 174 amino acids shows a 20-fold higher activity than hG-CSF consisting of 177 amino acids. However, whether both hG-CSFs are actually expressed in vivo is not elucidated at this time.
- hG-CSF does not contain the amino acid sequences (Asn-X-Ser/Thr) relating to N-glycosylation, but has an O-glycosylation site at the 133 rd amino acid residue, threonine.
- glycosylated hG-CSF glycoprotein is prepared from mammalian cells using a genomic DNA or cDAN encoding hG-CSF.
- the glycosylated hG-CSF thus produced has O-glycosidic type sugar chain structures, but it has been known that the sugar moiety is not essential for hG-CSF activity (Lawrence, M. et al., Science 232: 61, 1986). Further, since these methods need to employ an expensive culture medium and complicated purification process to obtain the glycosylated hG-CSF, they are uneconomical.
- hG-CSF non-glycosylated hG-CSF using a prokaryotic cell.
- E. coli which produces hG- CSF consisting of 175 or 178 amino acids according to the characteristics of E. coli cell.
- These products have an extra methionine residue added at the terminal amino residue by the action of the ATG codon existing at the site of initiation codon.
- the additional methionine can trigger harmful immune response (EP Patent Publication No. 256,843).
- most of the expressed hG-CSF accumulates in cytoplasm in the form of insoluble inclusion bodies and must be converted into an active form through refolding during purification.
- hG-CSF exists partially in a reduced form, or forms an intermolecular disulfide coupling body or a defective disulfide coupling body. It is difficult to remove these by-products, which markedly lower the yield. In particular, it is extremely difficult to remove undesirable hG-CSF by-products such as misfolded hG-CSF.
- hG-CSF has five cysteine residues, four of which participate in disulfide bonding, while the remaining cysteine in the unbound free form reduces the folding titer and stability in solution. Further, non-glycosylated hG-CSF expressed in E. coli is apt to aggregate at a high concentration because of its low solubility.
- the present inventors have endeavored to develop a method for producing a soluble hG-CSF having no methionine residue added at the terminal amino residue thereof by using a microorganism.
- the present inventors have previously generated a new signal peptide of E. coli thermostable enterotoxin II (Korean Patent Application No. 98-38061) and found that this new secretory signal peptide can be used for the mass-production of the native form of hG-CSF. Further, the present inventors have found that when an hG-CSF derivative obtained by replacing the 17 th cysteine residue with other amino acid ligated to the modified E.
- thermostable enterotoxin II signal sequence is expressed, hG-CSF is produced in a wild-type form having no methionine residue added at its terminal amino residue in the periplasm of E. coli (Korean Patent No. 356140).
- a protein when a protein has a small molecular weight, it may be lost in the process of clearing intestinal tract, kidney, spleen or liver through filteration, but such pegylation may reduce the clearance loss (Knauf, M. J. et al, J. Biol. Chem. 263: 15064, 1988). Many studies to prolong the in vivo half-life, increase the solubility or reduce the immune response by coupling PEG to a biologically active peptide or a protein have been actively progressed.
- pegylation increases the solubility of peptide drug and prevents the hydrolysis thereof, thereby increasing the serum stability of the peptide drug without incurring any immune response due to its low antigenecity (Sade et al., J. Fermentation Bioengineering 71: 1370139, 1991).
- pegylated polypeptides have the disadvantages of lowering both the activity and production yield of an active substance when the molecular weight of PEG increases.
- PEG is covalently linked to one or more of free lysine residues existing at the surface of a protein, but if PEG binds to an active site directly involved in protein activity, the protein activity decreases. Further, since the binding of PEG to a lysine residue occurs in a random fashion, many kinds of pegylated protein complexes may co-exist as a mixture, and therefore, a purification process of a desired complex becomes more complicated and difficult (EP Patent Publication Nos: 0401384 and 0335423).
- EP Patent Publication No: 0733067 has disclosed a method for preparing a uniform G-CSF composition which comprises a PEG-G-CSF complex prepared by a selective coupling of PEG to a terminal amino residue of G-CSF.
- a fusion protein of PEG-G-CSF complex thus prepared shows enhanced stability, but this method does not use a wild-type G-CSF and produces an inactive protein having an extra methionine residue added at the terminal amino residue in the form of an inclusion body.
- they have the problems of triggering harmful immune response, requiring a refolding process and a markedly low yield.
- the present inventors have developed a PEG conjugate of an hG-CSF derivative having the 17 amino acid of the wild-type hG-CSF replaced with serine without an extra methionine residue added at the terminal amino residue.
- the PEG-hG-CSF derivative conjugate of the present invention has a prolonged in vivo half-life without inducing an immune response in a subject, while minimizing the biological activity loss.
- a primary object of the present invention is to provide a PEG conjugate of an hG-CSF derivative having a prolonged in vivo half-life and an improved biological activity without inducing an immune response in a subject; and a method for the preparation thereof.
- Another object of the present invention is to provide a pharmaceutical composition comprising said PEG conjugate of hG-CSF derivative.
- a further object of the present invention is to provide a method for enhancing the in vivo stability and prolonging the circulating half-life of hG- CSF, without sacrificing the activity thereof.
- a PEG-hG-CSF derivative conjugate having a prolonged in vivo half-life which is prepared by modifying the terminal amino residue of hG-CSF derivative with one PEG molecule; and a method for the preparation thereof.
- a pharmaceutical composition of hG-CSF having a prolonged in vivo half-life in comparison with unmodified hG-CSF which comprises said PEG- hG-CSF derivative conjugate and a pharmaceutically acceptable carrier.
- a method for enhancing the in vivo half-life of hG-CSF which comprises specifically modifying the terminal amino residue of hG-CSF derivative with one PEG molecule.
- Fig. 1 SDS-PAGE results of PEG-hG-CSF derivative conjugates
- Fig. 2 a size exclusion chromatogram of PEG-hG-CSF derivative conjugates
- Fig. 3 a result for comparing the peptide map of a PEG-hG-CSF derivative conjugate (B) with that of unmodified hG-CSF (A);
- Fig. 4 in vivo activities of PEG-hG-CSF derivative conjugates;
- Fig. 5 a pharmacokinetic graph showing the enhanced circulating half- life of a PEG-hG-CSF derivative conjugate.
- the present invention relates to a conjugate of human G-CSF derivative (Korean Patent No: 356140) modified with PEG and a method for the preparation thereof.
- the PEG conjugate of an hG-CSF derivative is a protein conjugate prepared by selectively modifying the terminal amino residue of an hG-CSF derivative with PEG, wherein the terminal amino residue does not necessarily participate in the binding of hG-CSF to a receptor thereof (Hill et, al., PNAS 90: 5167, 1993).
- the hG-CSF derivative employed in the present invention may be either a natural one isolated from a mammal or a chemically synthesized recombinant. Further, it may also be obtained from prokaryotic or eukaryotic cells transformed with DNA coding the hG-CSF derivative by genetic engineering. For this purpose, E. coli, yeast (e.g., S. cerevisias) and mammalian cells (e.g., Chinese hamster ovary cell, CHO) can be employed as a host.
- E. coli, yeast e.g., S. cerevisias
- mammalian cells e.g., Chinese hamster ovary cell, CHO
- the protein product expressed from hG-CSF derivative-transformed mammalian cells or eukaryotic cells may be glycosylated or non-glycosylated with a host cell-derived carbohydrate.
- the protein product may include an incipient methionine residue at -1 position starting from the translation initiation codon.
- a recombinant hG- CSF derivative obtained by replacing the 17 th amino acid residue of wild-type hG-CSF with serine according to the method described in Korean Patent No. 356140.
- PEG employable in the present invention mat be any one of known soluble PEG polymers. In the process of modifying a protein with a PEG polymer having a reactive group, the PEG binding occurs at the ⁇ -amino group of lysine, cysteine or histidine residue depending on the nature of the reactive group of PEG. But, when such a PEG polymer having a reactive group is used, two more of PEG molecules may bind to a hG-CSF derivative, leading to the loss of its protein activity.
- the binding between the hG-CSF derivative and PEG preferably occurs in a molar ratio of 1:1, and a suitable PEG polymer for this purpose is soluble in an aqueous environment such as a physiological environment and has a single reactive aldehyde group which specifically binds to the ⁇ -amino group of the terminal amino residue of hG-CSF derivative.
- a PEG has the aldehyde reactive group at one end and an alkoxy group at the other end, and the terminal amino residue is threonine.
- the molecular weight of said PEG is preferably, but not to be limited to, in the range of 2 to 100 kDa, more preferably, 10 to 40 kDa; and said PEG may be branched or not.
- a pegylation reaction involving the amino group of the hG-CSF derivative and the aldehyde group of the PEG is carried out in the presence of a reducing agent.
- the alkoxy-PEG-aldehyde is methoxy-PEG-aldehyde
- the reducing agent is cyanoborohydride (NaCNBH 3 ), sodium borohydride, dimethylamine borate or pyridine borate.
- the HPLC peak pattern of PEG-hG-CSF derivative conjugate differs from that of unmodified hG-CSF in specific ways due to the binding of PEG to one of protease cleavage fragments of PEG conjugate.
- the protease may be, but not limited to, trypsin, subtilisin or endoproteinase Glu-C; and, more preferably, endoproteinase Glu-C.
- the biological activity of the inventive PEG-hG-CSF derivative conjugate is measured according to the conventional methods well-known in the art (Baldwin et al., Ada Endocrinologica. 119: 326, 1988; Clark et al., J. Biol.
- the present invention provides a pharmaceutical composition of hG-CSF having a prolonged in vivo half-life, which comprises the inventive PEG-hG-CSF derivative conjugate and a pharmaceutically acceptable carrier.
- the pharmaceutical composition of the present invention can be administered via various routes including oral, transdermal, subcutaneous, intravenous and intramuscular introduction, and injection is more preferred.
- the composition of the invention may be formulated so as to provide quick, sustained or delayed release of the active ingredient after it is administered to a patient, by employing any one of the procedures well-known in the art.
- the formulation may be in the form of a tablet, pill, powder, sachet, elixer, suspension, emulsion, solution, syrup, aerosol, soft and hard gelatin capsule, sterile injectable solution, sterile packaged powder and the like.
- Suitable carriers, excipients or diluents are lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, alginates, gelatin, calcium phosphate, calcium silicate, polyvinylpyrrolidone, cellulose, methylcellulose, microcrystalline magnesium stearate and mineral oil.
- the formulation may additionally include fillers, anti-agglutinating agents, lubricating agents, wetting agents, flavoring agents, emulsifiers, preservatives and the like.
- a typical daily dose of the inventive PEG- hG-CSF derivative conjugate as an effective ingredient may range from about 0.01 to 100 g/kg body weight (calculating the mass of protein alone, without chemical modification). Owing to the enhanced stability of PEG-hG-CSF derivative conjugate of the invention, the total number and frequency of the administration of the formulation comprising the inventive PEG-G-CSF derivative conjugate can be considerably reduced.
- HM10411 Recombinant hG-CSF derivative, HM10411 was prepared according to the method described in Korea Patent No: 356140.
- the hG-CSF derivative thus prepared has the 17 th amino acid of wild-type G-CSF replaced with serine ( 17 S-G-CSF).
- HM10411 was dissolved in 100 mM potassium phosphate buffer (pH 6.0) to a concentration of 5 mg/m.£, distributed into three vials, and then, adjusted to a final volume of 5 m-6.
- An activated methoxy-PEG-aldehyde (Shearwater, USA) which has 20 kDa or 40 kDa of PEG was added to each of two vials in an amount corresponding to a HM10411 :methoxy-PEG-aldehyde molar ratio of 1 :4.
- Sodium cyanoborohydride NaCNBH 3 , Sigma, USA
- was added thereto to a final concentration of 20 mM and the reaction mixture was stirred at 4 ° C for 18 hrs.
- ethanolamine was added thereto to a final concentration of 50 mM to inactivate unreacted PEG.
- the reaction mixture was subjected to
- the PEG modified HM10411 was further purified from the elution fraction obtained in Example 2 as follows.
- the column effluent was subjected to size exclusion chromatography.
- the concentrated effluent was loaded onto Superdex 200 (Pharmacia) column equilibrated with 10 mM sodium phosphate buffer (pH 7.0) and eluted with the same buffer at a flow rate of 1 m /min. Since the tri- and di-PEG-HM10411 conjugates which eluted earlier than the mono-PEG-HM10411 conjugate were removed to obtain the purified mono-PEG-HM10411 conjugate.
- the mono-PEG-HM10411 conjugate was analyzed for the state of its modification and purity by SDS-PAGE using 15% criterion gel (Bio-Rad) stained with Coomassie blue, and also for the protein concentration by measuring absorbance at 280 nm using size exclusion chromatography according to Beer-Lambert theory (Bollag et al., Protein Methods Chapter 3, press in Wiley-Liss).
- the respective appearance molecular weights of 20 kDa and 40 kDa mono-PEG-HM10411 conjugates were about 50 kDa and 120 kDa.
- Fig. 1 shows the SDS-PAGE results of 20 kDa and 40 kDa mono-PEG-
- HM10411 conjugates wherein lane 1 is 3.0 ⁇ g of a molecular weight marker (Invitron, bench marker, 15, 20, 25, 30, 40, 50, 60, 70, 80, 90, 100, 120, 160, and 220 kDa starting from the bottom band); lane 2, 10 ⁇ g of unmodified HM10411; lane 3, 10 ⁇ g of 40 kDa mono-PEG-HM10411 conjugate; and lane 4, 10 ⁇ g of 20 kDa mono-PEG-HM10411 conjugate.
- a band of PEG-HM10411 conjugate was observed at a position corresponding to a molecular weight calculated by adding the molecular weight of HM10411 to the molecular weight of PEG. Further, since there was no band containing other proteins besides the PEG-HM10411 conjugate, it was found that the purified PEG-HM10411 conjugate had a high purity.
- Fig. 2 shows the results of size exclusion chromatography for each sample using HPLC (Hewlett-Packard, HPllOl).
- HPLC Hewlett-Packard, HPllOl
- the effluent was loaded onto an analyzing column (Biosep SEC-S3000, Phenomenex) equilibrated with the buffer mixed with a mixture of 100 mM sodium phosphate (pH 6.9) and 20% ethanol, eluted with the same buffer at a flow rate of 0.6 m ⁇ /min, and measured the absorbance at 214 nm.
- the retention times of the unmodified HM10411, 20 kDa and 40 kDa mono-PEG-HM10411 conjugates were 11.832, 10.184 and 9.694 min, respectively. Further, it was found that the PEG modified HM10411 was observed as a single peak in a highly pure form.
- Each of the protein cleavage fragments were injected into Vydac 238TP54 C4 column (4.6x 250 mm, 5 ⁇ of particle size, 300 A of small hole size) and the peptide map thereof was determined by an acetonitrile linear gradient method using 0.1% trifluoroacetic acid through HPLC.
- HM10411 has seven Glu-C cleavage sites, eight peaks are theoretically expected to be detected, but actually Glu-C cleavage did not occur at all Glu-C cleavage sites and it was not possible to completely detect all peaks due to the insufficient resolution of the column.
- the peak containing the terminal amino residue of HM10411 has been well defined in previous reports (Herman, et al., Pharm. Biotechnol. 9: 303, 1996), it should be possible to determine the amino acid sequences of the respective peaks obtained by Glu-c cleavage fractionation and to compare the peak patterns of unmodified HM10411 and PEG-HM10411 conjugate for the difference in their amino acid sequences.
- HL-60 cells Human myelogenous originated cell line, HL-60 cells (ATCC CCL-240, Promyelocytic leukemia patient/36 yr old Caucasian female) were cultivated in RPMI1640 medium supplemented with 10% fetal bovine serum (FBS), and then, the number of cells were adjusted to about 2.2x 10 5 cells/ml DMSO (dimethylsulfoxide, culture grade, Sigma) was added to the cells to a final concentration of 1.25% (v/v).
- FBS fetal bovine serum
- DMSO treated culture solution having about 2x IO 4 cells per well was added to a 96-well plate (low evaporation 96 well plate, Corning) and incubated at 37 ° C for about 48 hrs in a 5% C0 2 incubator.
- concentration of each sample was determined by using a G-CSF ELISA kit (Enzyme Linked Immunosorbent Assay, R&D systems), and each sample was diluted with RPMI1640 medium at an appropriate ratio to a concentration of 500 ng/m ⁇ .
- the resulting solution was subjected to 10 cycles of sequential half dilution with RPMI 1640 medium.
- Fig. 4 shows the results of measuring the in vivo activities. As can be seen from Fig. 4, all samples stimulated the growth of G-CSF sensitive cells, which suggesting that they exhibit in vivo activity. ⁇ Table 1>
- the inventive PEG modified HM10411 conjugates showed a lower activity than the wild-type G-CSF (Filgrastim) or the unmodified HM10411.
- 20 kDa PEG-HM10411 conjugate showed similar activity to that observed for 20 kDa PEG-G-CSF (Neulasta), and 40 kDa PEG-HM10411 conjugate, an activity lower than those observed for 20 kDa PEG modified proteins (Neulasta and 20 kDa PEG-HM10411).
- mice Male Sprague Dawly (SD) rat groups (7-week old, average body weight 200-250 g) were used in the following experiments. Rats received subcutaneous injection of 100 g/kg of Filgrastim, unmodified HM10411 (control), Neulasta and 40 kDa PEG-HM 10411 conjugate, respectively. Blood samples were taken from the control group at 0.5, 1, 2, 4, 6, 12, 24, 30 and 48 hrs after the injection, and the samples of the test groups, 1, 6, 12, 24, 30, 48 and 72 hrs after the injection. Blood samples were collected in an eppendorf tube coated with heparin to prevent blood coagulation, and subjected to high- speed micro centrifugation at 4 ° C, 10,000* g for 5 min to remove cells. The protein concentration in sera was measured by ELISA using an antibody specific for G-CSF.
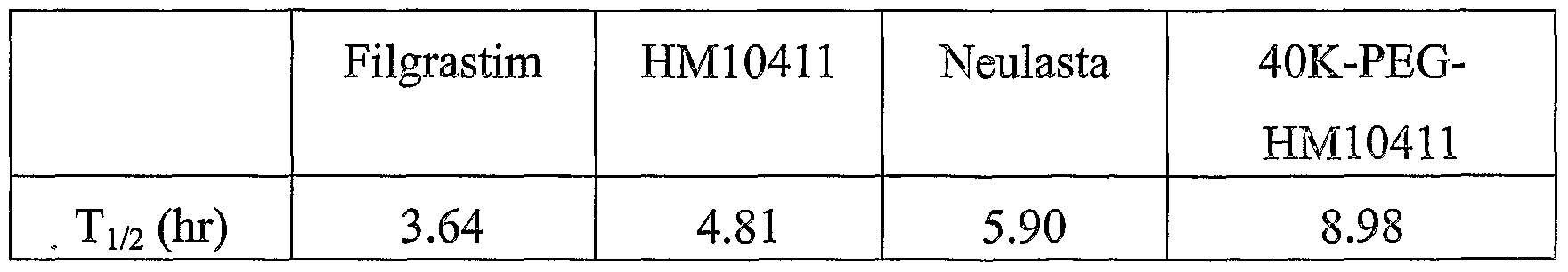
- the half-life of 40 kDa PEG- HM10411 conjugate was longer than those of wild-type G-CSF and its derivative. Particularly, the half-life of 40 kDa PEG-HM 10411 conjugate was about 2.5-fold higher than that of Fligrastim and about 0.5-fold higher than that of Neulasta.
- the increased in-blood half-life may prolong its stability and biological activity, and therefore, may reduce the number of injection to a patient.
Landscapes
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Mechanical Engineering (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Zoology (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Educational Technology (AREA)
- Toxicology (AREA)
- Educational Administration (AREA)
- Gastroenterology & Hepatology (AREA)
- Physics & Mathematics (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Theoretical Computer Science (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Business, Economics & Management (AREA)
- Manufacturing & Machinery (AREA)
- Peptides Or Proteins (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
A conjugate of a hG-CSF derivative modified with a PEG shows an enhanced in vivo stability and prolonged half-life in blood, while reducing the possibility of inducing an immune response.
Description
HUMAN GRANULOCYTE-COLONY STIMULATING FACTOR
CONJUGATE HAVING ENHANCED STABILITY IN BLOOD AND PROCESS FOR THE PREPARATION THEREOF
Field of the Invention
The present invention relates to a conjugate of a human granulocyte- colony stimulating factor ( G-CSF) derivative modified with a polyethylene glycol (PEG) and a method for the preparation thereof.
Background of the Invention
Granulocyte-colony stimulating factors (G-CSFs) are cytokines that stimulate the expansion and differentiation of bone marrow progenitor cells and leukocytes. They also activate mature myeloid cells to mediate a variety of anti-microbial activities and inflammatory responses. They have been widely used in clinical transplantation which induces hematopoietic stem cell mobilization and increases myeloid progenitor cell population. It has been reported that the purified G-CSF is highly active in inducing granulocyte differentiation and suppresses stem cell self-replication in myeloid leukemia cells (Nicola et. al, JBC 258(14): 9017-9023, 1983). Recently, recombinant G-CSF proteins, sold under the brand names of Filgrastim and Neulasta, have been increasingly employed in the treatment of various diseases including cancer and hematopoietic disorders. Human G-CSF is a glycoprotein, consisting of 174 amino acids, which plays an important role in hematopoietic cell proliferation, differentiation of hematopoietic precursor cells, and activation of mature neutrophilic granulocytes. hG-CSF gene transcribes into two kinds of mRNA consisting of 207 and 204 amino acids, respectively, that contain a leader sequence consisting of 30 amino acids for their secretion at the terminal amino residue (Nagata et al.,
Nature 319: 415-418, 1986; Nagata et al., EMBO J. 5(3): 575-581, 1986; Souza et al, Science 232: 61-65, 1986). The difference in the two hG-CSFs is the presence or absence of three amino acids (Val-Ser-Glu) at the 35th amino acid residue starting from the terminal amino residue. Accordingly, a mature hG- CSF protein has 174 or 177 amino acids. hG-CSF consisting of 174 amino acids shows a 20-fold higher activity than hG-CSF consisting of 177 amino acids. However, whether both hG-CSFs are actually expressed in vivo is not elucidated at this time. hG-CSF does not contain the amino acid sequences (Asn-X-Ser/Thr) relating to N-glycosylation, but has an O-glycosylation site at the 133rd amino acid residue, threonine.
In order to purify G-CSF, a method which comprises culturing a G-CSF producing cell and extracting G-CSF from a culture supernatant has been widely employed; however, the method suffers from various problems associated with the low yield of G-CSF and complicated purification process. It has been reported that the amino acid sequence of hG-CSF and a gene encoding the same can be established by using the existing methods (Korean Patent Laid-Open Publication Nos. 91-5624 and 92-2312), and a method for preparing a substance showing hG-CSF activity by using genetic engineering is developed (Korean Patent Nos. 47178, 53723 and 57582). In the previously reported methods, a glycosylated hG-CSF glycoprotein is prepared from mammalian cells using a genomic DNA or cDAN encoding hG-CSF. The glycosylated hG-CSF thus produced has O-glycosidic type sugar chain structures, but it has been known that the sugar moiety is not essential for hG-CSF activity (Lawrence, M. et al., Science 232: 61, 1986). Further, since these methods need to employ an expensive culture medium and complicated purification process to obtain the glycosylated hG-CSF, they are uneconomical.
Further, widely employed in a method for producing non-glycosylated hG-CSF using a prokaryotic cell is a process using E. coli which produces hG- CSF consisting of 175 or 178 amino acids according to the characteristics of E. coli cell. These products have an extra methionine residue added at the
terminal amino residue by the action of the ATG codon existing at the site of initiation codon. However, it has been reported that the additional methionine can trigger harmful immune response (EP Patent Publication No. 256,843). Furthermore, most of the expressed hG-CSF accumulates in cytoplasm in the form of insoluble inclusion bodies and must be converted into an active form through refolding during purification. Such a refolding process is not efficient, and therefore, hG-CSF exists partially in a reduced form, or forms an intermolecular disulfide coupling body or a defective disulfide coupling body. It is difficult to remove these by-products, which markedly lower the yield. In particular, it is extremely difficult to remove undesirable hG-CSF by-products such as misfolded hG-CSF. In addition, hG-CSF has five cysteine residues, four of which participate in disulfide bonding, while the remaining cysteine in the unbound free form reduces the folding titer and stability in solution. Further, non-glycosylated hG-CSF expressed in E. coli is apt to aggregate at a high concentration because of its low solubility.
To solve these problems, the present inventors have endeavored to develop a method for producing a soluble hG-CSF having no methionine residue added at the terminal amino residue thereof by using a microorganism. The present inventors have previously generated a new signal peptide of E. coli thermostable enterotoxin II (Korean Patent Application No. 98-38061) and found that this new secretory signal peptide can be used for the mass-production of the native form of hG-CSF. Further, the present inventors have found that when an hG-CSF derivative obtained by replacing the 17th cysteine residue with other amino acid ligated to the modified E. coli thermostable enterotoxin II signal sequence is expressed, hG-CSF is produced in a wild-type form having no methionine residue added at its terminal amino residue in the periplasm of E. coli (Korean Patent No. 356140).
Meanwhile, the problem of using a peptide drug in vivo is that its half- life is too low to exert a satisfactory therapeutic effect. Accordingly, there have been several attempts to develop a peptide drug having an enhanced half-
life in blood to reduce the injection interval so that a high pharmacological efficacy and quality of patient's life can be maintained. One of the most widely used methods for improving the stability of a protein is pegylation, which involves chemical modification of a polypeptide with a highly soluble moiety such as polyethylene glycol (PEG). When linked to a peptide drug specifically or non-specifically, PEG increases the solubility of the peptide drug to water or an organic solvent, reduces the immune response and enhances in vivo stability. Further, when a protein has a small molecular weight, it may be lost in the process of clearing intestinal tract, kidney, spleen or liver through filteration, but such pegylation may reduce the clearance loss (Knauf, M. J. et al, J. Biol. Chem. 263: 15064, 1988). Many studies to prolong the in vivo half-life, increase the solubility or reduce the immune response by coupling PEG to a biologically active peptide or a protein have been actively progressed. It has been well known that, pegylation increases the solubility of peptide drug and prevents the hydrolysis thereof, thereby increasing the serum stability of the peptide drug without incurring any immune response due to its low antigenecity (Sade et al., J. Fermentation Bioengineering 71: 1370139, 1991). However, such pegylated polypeptides have the disadvantages of lowering both the activity and production yield of an active substance when the molecular weight of PEG increases.
Usually, PEG is covalently linked to one or more of free lysine residues existing at the surface of a protein, but if PEG binds to an active site directly involved in protein activity, the protein activity decreases. Further, since the binding of PEG to a lysine residue occurs in a random fashion, many kinds of pegylated protein complexes may co-exist as a mixture, and therefore, a purification process of a desired complex becomes more complicated and difficult (EP Patent Publication Nos: 0401384 and 0335423). To solve such problems, US Patent No: 5,766,897 and International Patent Publication No: WO 00/42175 teach that PEG-maleimide can be used for the selective binding of PEG to a cysteine residue to prevent PEG from interacting with an active site
of human growth hormone (hGH) in a coupling reaction of hGH to PEG. However, this coupling reaction demands at least one free cysteine residue which is not engaged in a disulfide bonding, but all four cysteine residues of hGH are involved in the formation of disulfide bonding. Accordingly, the method described above performs a pegylation reaction using a hGH derivative having an extra cysteine residue added by insertion. However, several problems have been reported that the protein employed in the pegylation reaction is not a wild-type and it is very difficult to express and purify the protein having an additional free cysteine residue. EP Patent Publication No: 0733067 has disclosed a method for preparing a uniform G-CSF composition which comprises a PEG-G-CSF complex prepared by a selective coupling of PEG to a terminal amino residue of G-CSF. A fusion protein of PEG-G-CSF complex thus prepared shows enhanced stability, but this method does not use a wild-type G-CSF and produces an inactive protein having an extra methionine residue added at the terminal amino residue in the form of an inclusion body. Thus, they have the problems of triggering harmful immune response, requiring a refolding process and a markedly low yield.
To solve the mentioned problems, the present inventors have developed a PEG conjugate of an hG-CSF derivative having the 17 amino acid of the wild-type hG-CSF replaced with serine without an extra methionine residue added at the terminal amino residue. The PEG-hG-CSF derivative conjugate of the present invention has a prolonged in vivo half-life without inducing an immune response in a subject, while minimizing the biological activity loss.
Summary of the Invention
Accordingly, a primary object of the present invention is to provide a PEG conjugate of an hG-CSF derivative having a prolonged in vivo half-life and an improved biological activity without inducing an immune response in a subject; and a method for the preparation thereof.
Another object of the present invention is to provide a pharmaceutical composition comprising said PEG conjugate of hG-CSF derivative.
A further object of the present invention is to provide a method for enhancing the in vivo stability and prolonging the circulating half-life of hG- CSF, without sacrificing the activity thereof.
In accordance with one aspect of the present invention, there is provided a PEG-hG-CSF derivative conjugate having a prolonged in vivo half-life which is prepared by modifying the terminal amino residue of hG-CSF derivative with one PEG molecule; and a method for the preparation thereof. In accordance with another aspect of the present invention, there is provided a pharmaceutical composition of hG-CSF having a prolonged in vivo half-life in comparison with unmodified hG-CSF, which comprises said PEG- hG-CSF derivative conjugate and a pharmaceutically acceptable carrier.
In accordance with still another aspect of the present invention, there is provided a method for enhancing the in vivo half-life of hG-CSF, which comprises specifically modifying the terminal amino residue of hG-CSF derivative with one PEG molecule.
Brief Description of the Drawings
The above and other objects and features of the present invention will become apparent from the following description of the invention, when taken in conjunction with the accompanying drawings which respectively show:
Fig. 1 : SDS-PAGE results of PEG-hG-CSF derivative conjugates;
Fig. 2: a size exclusion chromatogram of PEG-hG-CSF derivative conjugates;
Fig. 3: a result for comparing the peptide map of a PEG-hG-CSF derivative conjugate (B) with that of unmodified hG-CSF (A); Fig. 4: in vivo activities of PEG-hG-CSF derivative conjugates; and
Fig. 5: a pharmacokinetic graph showing the enhanced circulating half- life of a PEG-hG-CSF derivative conjugate.
etaiied HDegeiiption of the Invention
The present invention relates to a conjugate of human G-CSF derivative (Korean Patent No: 356140) modified with PEG and a method for the preparation thereof.
The PEG conjugate of an hG-CSF derivative is a protein conjugate prepared by selectively modifying the terminal amino residue of an hG-CSF derivative with PEG, wherein the terminal amino residue does not necessarily participate in the binding of hG-CSF to a receptor thereof (Hill et, al., PNAS 90: 5167, 1993).
The hG-CSF derivative employed in the present invention may be either a natural one isolated from a mammal or a chemically synthesized recombinant. Further, it may also be obtained from prokaryotic or eukaryotic cells transformed with DNA coding the hG-CSF derivative by genetic engineering. For this purpose, E. coli, yeast (e.g., S. cerevisias) and mammalian cells (e.g., Chinese hamster ovary cell, CHO) can be employed as a host. According to the characteristics of host cell used, the protein product expressed from hG-CSF derivative-transformed mammalian cells or eukaryotic cells may be glycosylated or non-glycosylated with a host cell-derived carbohydrate. Further, in case of expressing from hG-CSF derivative-transformed prokaryotic cells, the protein product may include an incipient methionine residue at -1 position starting from the translation initiation codon.
In a preferred embodiment of the present invention, a recombinant hG- CSF derivative obtained by replacing the 17th amino acid residue of wild-type hG-CSF with serine according to the method described in Korean Patent No. 356140. PEG employable in the present invention mat be any one of known
soluble PEG polymers. In the process of modifying a protein with a PEG polymer having a reactive group, the PEG binding occurs at the ε-amino group of lysine, cysteine or histidine residue depending on the nature of the reactive group of PEG. But, when such a PEG polymer having a reactive group is used, two more of PEG molecules may bind to a hG-CSF derivative, leading to the loss of its protein activity.
Accordingly, the binding between the hG-CSF derivative and PEG preferably occurs in a molar ratio of 1:1, and a suitable PEG polymer for this purpose is soluble in an aqueous environment such as a physiological environment and has a single reactive aldehyde group which specifically binds to the α-amino group of the terminal amino residue of hG-CSF derivative. Preferably, such a PEG has the aldehyde reactive group at one end and an alkoxy group at the other end, and the terminal amino residue is threonine. Further, the molecular weight of said PEG is preferably, but not to be limited to, in the range of 2 to 100 kDa, more preferably, 10 to 40 kDa; and said PEG may be branched or not.
To couple a PEG to an hG-CSF derivative, a pegylation reaction involving the amino group of the hG-CSF derivative and the aldehyde group of the PEG is carried out in the presence of a reducing agent. Preferably, the alkoxy-PEG-aldehyde is methoxy-PEG-aldehyde, and the reducing agent is cyanoborohydride (NaCNBH3), sodium borohydride, dimethylamine borate or pyridine borate. Further, it is preferable to employ the hG-CSF derivative and PEG in a molar ratio ranging from 1 : 1 to 1 :50 in the pegylation reaction. After the pegylation reaction is completed, whether a PEG-G-CSF derivative conjugate is generated or not can be examined by SDS-PAGE. Subsequently, a fraction containing the desired PEG-hG-CSF derivative conjugate may be isolated utilizing the difference in the appearance charge by using cation exchange chromatography. The resulting fraction thus obtained is subjected to size exclusion chromatography to purify the PEG-hG-CSF derivative conjugate. To examine whether PEG is specifically linked to the terminal amino
residue of hG-CSF derivative, the peptide map of PEG-hG-CSF derivative conjugate is drawn and compared with the previously reported peptide map of hG-CSF derivative (Journal of chromatography A, 808: 121, 1998; Analytical Biochemistry 122: 348, 1982; and Biotechnology and Applied Biochemistry 10: 326, 1988). Namely, when the PEG-hG-CSF derivative conjugate and unmodified hG-CSF are subjected to protease cleavage, the HPLC peak pattern of PEG-hG-CSF derivative conjugate differs from that of unmodified hG-CSF in specific ways due to the binding of PEG to one of protease cleavage fragments of PEG conjugate. The protease may be, but not limited to, trypsin, subtilisin or endoproteinase Glu-C; and, more preferably, endoproteinase Glu-C.
The biological activity of the inventive PEG-hG-CSF derivative conjugate is measured according to the conventional methods well-known in the art (Baldwin et al., Ada Endocrinologica. 119: 326, 1988; Clark et al., J. Biol.
Chem. 271(36): 21969, 1996; and Bozzola et al., J. endocrinol. Invest. 21: 768, 1998). As a result, the PEG-hG-CSF derivative conjugate shows a prolonged in vivo half-life, enhanced biological activity, and reduced toxicity and immunogenecity.
Accordingly, the present invention provides a pharmaceutical composition of hG-CSF having a prolonged in vivo half-life, which comprises the inventive PEG-hG-CSF derivative conjugate and a pharmaceutically acceptable carrier.
The pharmaceutical composition of the present invention can be administered via various routes including oral, transdermal, subcutaneous, intravenous and intramuscular introduction, and injection is more preferred. The composition of the invention may be formulated so as to provide quick, sustained or delayed release of the active ingredient after it is administered to a patient, by employing any one of the procedures well-known in the art. The formulation may be in the form of a tablet, pill, powder, sachet, elixer, suspension, emulsion, solution, syrup, aerosol, soft and hard gelatin capsule, sterile injectable solution, sterile packaged powder and the like. Examples of
suitable carriers, excipients or diluents are lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, alginates, gelatin, calcium phosphate, calcium silicate, polyvinylpyrrolidone, cellulose, methylcellulose, microcrystalline magnesium stearate and mineral oil. The formulation may additionally include fillers, anti-agglutinating agents, lubricating agents, wetting agents, flavoring agents, emulsifiers, preservatives and the like.
The amount of the active ingredient actually administered ought to be determined in light of various relevant factors including the condition to be treated, the chosen route of administration, the age, sex and body weight of the individual patient, and the severity of the patient's syndrome, especially, the kind of active ingredient. Preferably, a typical daily dose of the inventive PEG- hG-CSF derivative conjugate as an effective ingredient may range from about 0.01 to 100 g/kg body weight (calculating the mass of protein alone, without chemical modification). Owing to the enhanced stability of PEG-hG-CSF derivative conjugate of the invention, the total number and frequency of the administration of the formulation comprising the inventive PEG-G-CSF derivative conjugate can be considerably reduced.
The present invention is further defined in the following Examples. It should be understood that these Examples, while indicating preferred embodiments of the invention, are given by way of illustration only. From the above discussion and these Examples, one skilled in the art can ascertain the essential characteristics of this invention, and without departing from the spirit and scope thereof, can make various changes and modifications of the invention to adapt it to various usage and conditions.
Esample 1: Preparation of recombinant human G-CSF derivative
Recombinant hG-CSF derivative, HM10411 was prepared according to the method described in Korea Patent No: 356140. The hG-CSF derivative
thus prepared has the 17th amino acid of wild-type G-CSF replaced with serine (17S-G-CSF).
Example 2; Preparation of PEG-hG-C§F derivative conjugate
HM10411 was dissolved in 100 mM potassium phosphate buffer (pH 6.0) to a concentration of 5 mg/m.£, distributed into three vials, and then, adjusted to a final volume of 5 m-6. An activated methoxy-PEG-aldehyde (Shearwater, USA) which has 20 kDa or 40 kDa of PEG was added to each of two vials in an amount corresponding to a HM10411 :methoxy-PEG-aldehyde molar ratio of 1 :4. Sodium cyanoborohydride (NaCNBH3, Sigma, USA) was added thereto to a final concentration of 20 mM, and the reaction mixture was stirred at 4°C for 18 hrs. Then, ethanolamine was added thereto to a final concentration of 50 mM to inactivate unreacted PEG. To further remove unreacted PEG, the reaction mixture was subjected to
Sephadex G-25 column chromatography (Pharmacia). Namely, after the column was equilibrated with 2 CV (column volume) of 10 mM Tris-HCl buffer (pH 7.5), the reaction mixture was loaded onto the column and eluted with the same buffer. Elution fractions were analyzed for absorbance at 260 nm using a UV spectrophotometer. As a result, the PEG modified HM10411 which has a large molecular weight was eluted before unreacted PEG.
Example 3: Isolation of mono-PEG-hG-CSF derivative conjugate
The PEG modified HM10411 was further purified from the elution fraction obtained in Example 2 as follows.
A column packed with 3 m-6 of PolyWAX LP (Polywax Inc., USA) was equilibrated with 10 mM Tris-HCl buffer (pH 7.5). The elution fraction containing the PEG modified HM10411 was loaded onto the column at a flow rate of 1 m-£/min, and the column was washed with 5 CV (15 m&) equilibration
buffer. Tri-, di- and mono-PEG-HM10411 conjugates were fractionated in that order using a salt concentration gradient method (NaCl concentration: 0- >100%) using 10 CV (30 ml) of 1 M NaCl buffer for 30 min.
To further purify the mono-PEG modified HM10411 from the mixture, the column effluent was subjected to size exclusion chromatography. The concentrated effluent was loaded onto Superdex 200 (Pharmacia) column equilibrated with 10 mM sodium phosphate buffer (pH 7.0) and eluted with the same buffer at a flow rate of 1 m /min. Since the tri- and di-PEG-HM10411 conjugates which eluted earlier than the mono-PEG-HM10411 conjugate were removed to obtain the purified mono-PEG-HM10411 conjugate.
The mono-PEG-HM10411 conjugate was analyzed for the state of its modification and purity by SDS-PAGE using 15% criterion gel (Bio-Rad) stained with Coomassie blue, and also for the protein concentration by measuring absorbance at 280 nm using size exclusion chromatography according to Beer-Lambert theory (Bollag et al., Protein Methods Chapter 3, press in Wiley-Liss). The respective appearance molecular weights of 20 kDa and 40 kDa mono-PEG-HM10411 conjugates were about 50 kDa and 120 kDa.
Fig. 1 shows the SDS-PAGE results of 20 kDa and 40 kDa mono-PEG-
HM10411 conjugates, wherein lane 1 is 3.0 μg of a molecular weight marker (Invitron, bench marker, 15, 20, 25, 30, 40, 50, 60, 70, 80, 90, 100, 120, 160, and 220 kDa starting from the bottom band); lane 2, 10 μg of unmodified HM10411; lane 3, 10 μg of 40 kDa mono-PEG-HM10411 conjugate; and lane 4, 10 μg of 20 kDa mono-PEG-HM10411 conjugate. As shown in Fig. 1, a band of PEG-HM10411 conjugate was observed at a position corresponding to a molecular weight calculated by adding the molecular weight of HM10411 to the molecular weight of PEG. Further, since there was no band containing other proteins besides the PEG-HM10411 conjugate, it was found that the purified PEG-HM10411 conjugate had a high purity.
Fig. 2 shows the results of size exclusion chromatography for each sample using HPLC (Hewlett-Packard, HPllOl). The effluent was loaded
onto an analyzing column (Biosep SEC-S3000, Phenomenex) equilibrated with the buffer mixed with a mixture of 100 mM sodium phosphate (pH 6.9) and 20% ethanol, eluted with the same buffer at a flow rate of 0.6 m^/min, and measured the absorbance at 214 nm. The retention times of the unmodified HM10411, 20 kDa and 40 kDa mono-PEG-HM10411 conjugates were 11.832, 10.184 and 9.694 min, respectively. Further, it was found that the PEG modified HM10411 was observed as a single peak in a highly pure form.
Example 4: Peptide mapping
To examine whether 40 kDa PEG is specifically linked to the terminal amino residue of hG-CSF in the PEG-hG-CSF derivative conjugate prepared in Example 3, the unmodified HM10411 and 40 kDa mono-PEG-HM10411 conjugate were subjected to peptide mapping and their peak patterns were compared with each other. The peptide map of unmodified HM10411 was previously reported in the art (Herman, et al., Pharm. Biotechnol. 9: 303, 1996; Clogston, et al., Analytical Biochemistry 202: 376, 1992). After the 40 kDa mono-PEG-HM10411 conjugate and unmodified HM10411 were loaded onto a desalting column (Pharmacia biotch) and eluted with 0.1 M ammonium acetate buffer (pH 8.3), the column effluents were adjusted to a final concentration of 0.25 mg/m-6, respectively. 10 μg of endoproteinase Glu-C (Roche) protease dissolved in distilled water was added to the respective column effluents. The reaction mixtures were each incubated in a 37°C constant water bath for 19 hrs to cleavage the protein. Each of the protein cleavage fragments were injected into Vydac 238TP54 C4 column (4.6x 250 mm, 5 μ of particle size, 300 A of small hole size) and the peptide map thereof was determined by an acetonitrile linear gradient method using 0.1% trifluoroacetic acid through HPLC.
Since HM10411 has seven Glu-C cleavage sites, eight peaks are theoretically expected to be detected, but actually Glu-C cleavage did not occur
at all Glu-C cleavage sites and it was not possible to completely detect all peaks due to the insufficient resolution of the column. However, since the peak containing the terminal amino residue of HM10411 has been well defined in previous reports (Herman, et al., Pharm. Biotechnol. 9: 303, 1996), it should be possible to determine the amino acid sequences of the respective peaks obtained by Glu-c cleavage fractionation and to compare the peak patterns of unmodified HM10411 and PEG-HM10411 conjugate for the difference in their amino acid sequences.
As shown in Fig. 3, it was found that the peak (Tl) corresponding to the terminal amino residue of HM10411 which was eluted at about 28 min in the peptide map of unmodified HM10411 did not match the PEG-HM10411 conjugate, due to the change of peak's elution time when the sample passed through the column. These results suggest that PEG binds to the terminal amino residue of HM 10411 in a molar ratio of 1 : 1.
Experimental Example 1: Measurement of in vivo activity
In vivo activities of wild-type G-CSF (Filgrastim, Amgen), 20 kDa PEG-G-CSF (Neulasta, Amgen), unmodified HM10411, and 40 kDa PEG- HM10411 conjugate were measured and compared each other as follows.
Human myelogenous originated cell line, HL-60 cells (ATCC CCL-240, Promyelocytic leukemia patient/36 yr old Caucasian female) were cultivated in RPMI1640 medium supplemented with 10% fetal bovine serum (FBS), and then, the number of cells were adjusted to about 2.2x 105 cells/ml DMSO (dimethylsulfoxide, culture grade, Sigma) was added to the cells to a final concentration of 1.25% (v/v). 90 μl of the DMSO treated culture solution having about 2x IO4 cells per well was added to a 96-well plate (low evaporation 96 well plate, Corning) and incubated at 37 °C for about 48 hrs in a 5% C02 incubator. The concentration of each sample was determined by using a G-CSF
ELISA kit (Enzyme Linked Immunosorbent Assay, R&D systems), and each sample was diluted with RPMI1640 medium at an appropriate ratio to a concentration of 500 ng/mδ. The resulting solution was subjected to 10 cycles of sequential half dilution with RPMI 1640 medium.
10 μl of each sample prepared above was added to each well having HL-60 cells on cultivation, and the concentration was reduced by half from 50 ng/m-β. The well plates treated with protein samples were further incubated at 37°C for 48 hrs.
To examine the extent cell growth after the incubation, the number of cells were determined by measuring the absorbance at 670 nm using CellTiter96™ (Cat. No. G4100, Promega).
Fig. 4 shows the results of measuring the in vivo activities. As can be seen from Fig. 4, all samples stimulated the growth of G-CSF sensitive cells, which suggesting that they exhibit in vivo activity. <Table 1>
As can be seen in Table 1, it was found that the inventive PEG modified HM10411 conjugates showed a lower activity than the wild-type G-CSF (Filgrastim) or the unmodified HM10411. 20 kDa PEG-HM10411 conjugate showed similar activity to that observed for 20 kDa PEG-G-CSF (Neulasta), and 40 kDa PEG-HM10411 conjugate, an activity lower than those observed for 20 kDa PEG modified proteins (Neulasta and 20 kDa PEG-HM10411). From these results, it was found that the in vivo activity of hG-CSF derivative,
HM10411, gradually decreased as the molecular weight of the PEG moiety increased, which was due to the structural and locational inhibition of PEG when hG-CSF binds to a receptor existing on the surface of a target cell. Namely, as the molecular weight of PEG increases, such inhibition of PEG increases gradually.
Experimental Example 2; PharmacoMiietic analysis
In vivo stability and pharmacokinetic coefficient of 40 kDa PEG- HM10411 conjugate prepared in Example 3 were measured and compared with those of biologically active wild-type proteins as follows.
3 Male Sprague Dawly (SD) rat groups (7-week old, average body weight 200-250 g) were used in the following experiments. Rats received subcutaneous injection of 100 g/kg of Filgrastim, unmodified HM10411 (control), Neulasta and 40 kDa PEG-HM 10411 conjugate, respectively. Blood samples were taken from the control group at 0.5, 1, 2, 4, 6, 12, 24, 30 and 48 hrs after the injection, and the samples of the test groups, 1, 6, 12, 24, 30, 48 and 72 hrs after the injection. Blood samples were collected in an eppendorf tube coated with heparin to prevent blood coagulation, and subjected to high- speed micro centrifugation at 4°C, 10,000* g for 5 min to remove cells. The protein concentration in sera was measured by ELISA using an antibody specific for G-CSF.
As a result, pharmacokinetic values of test and control groups are described in Table 2, in which T1 2 means the half-life of hG-CSF in blood. <Table 2>
As can be seen in Table 2 and Fig. 5, the half-life of 40 kDa PEG-
HM10411 conjugate was longer than those of wild-type G-CSF and its derivative. Particularly, the half-life of 40 kDa PEG-HM 10411 conjugate was about 2.5-fold higher than that of Fligrastim and about 0.5-fold higher than that of Neulasta. When a protein having biological activity exerts a therapeutic effect, the increased in-blood half-life may prolong its stability and biological activity, and therefore, may reduce the number of injection to a patient. These results suggest that the binding of PEG to HM10411 shows far superior in vivo durability than the binding of PEG to wild-type hG-CSF from the point of in- blood stability, and the binding of 40 kDa PEG rather than 20 kDa PEG favors the maintenance of the biological activity in vivo, thereby inducing a dramatic change in the therapeutic effect of a protein drug.
While the embodiments of the subject invention have been described and illustrated, it is obvious that various changes and modifications can be made therein without departing from the spirit of the present invention which should be limited only by the scope of the appended claims.
Claims
1. A polyethylene glycol (PEG) conjugate of a human granulocyte-colony stimulating factor (hG-CSF) prepared by modifying the terminal amino residue of a hG-CSF derivative.
2. The conjugate of claim 1, wherein the molar ratio of the hG-CSF derivative to said PEG is 1 : 1
3. The conjugate of claim 1, wherein the hG-CSF derivative has the amino acid sequence of the wild-type hG-CSF except that the 17th amino acid is replaced with serine.
4. The conjugate of claim 1, wherein the amino acid at the terminal amino residue of the hG-CSF derivative is threonine.
5. The conjugate of claim 1, wherein the PEG binds to the α-amino group of the terminal amino residue of the hG-CSF derivative.
6. The conjugate of claim 1, wherein the molecular weight of said PEG ranges from 2 to 100 kDa.
7. The conjugate of claim 6, wherein the molecular weight of said PEG ranges from 10 to 40 kDa.
8. The conjugate of claim 1, wherein said PEG has a single aldehyde group
9. The conjugate of claim 8, wherein said PEG is an alkoxy-PEG-aldehyde.
10. The conjugate of claim 9, wherein the alkoxy-PEG-aldehyde is methoxy-PEG-aldehyde.
11. A method for preparing the conjugate of claim 1 , which comprises 1) mixing a hG-CSF derivative with a PEG and performing a pegylation reaction; and
2) purifying a conjugate of the hG-CSF derivative modified with said PEG.
12. The method of claim 11, wherein step 1) is carried out in the presence of a reducing agent.
13. The method of claim 12, wherein the reducing agent is selected from the group consisting of sodium cyanoborohydride (NaCNBH3), sodium borohydride, dimethylamine borate and pyridine borate.
14. The method of claim 11 , wherein said PEG has a single aldehyde group
15. The method of claim 14, wherein said PEG is an alkoxy-PEG-aldehyde.
16. The method of claim 15, wherein the alkoxy-PEG-aldehyde is methoxy- PEG-aldehyde.
17. The method of claim 11, wherein the hG-CSF derivative is mixed with said PEG in a molar ratio ranging from 1:1 to 1 :50.
18. The method of claim 11, wherein the conjugate has the hG-CSF derivative:PEG molar ratio of 1 : 1
19. A pharmaceutical composition of a hG-CSF having a prolonged half- life, which comprises the conjugate of claim 1 and a pharmaceutically acceptable carrier.
20. A method for prolonging the in vivo half-life of a hG-CSF, which comprises modifying the terminal amino residue of an hG-CSF derivative with a PEG.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020030017867A KR20040083268A (en) | 2003-03-21 | 2003-03-21 | Human granulocyte-colony stimulating factor conjugate having enhanced stability in blood and process for the preparation thereof |
| KR10-2003-0017867 | 2003-03-21 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004083242A1 true WO2004083242A1 (en) | 2004-09-30 |
Family
ID=33028844
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2004/000569 Ceased WO2004083242A1 (en) | 2003-03-21 | 2004-03-17 | Human granulocyte-colony stimulating factor conjugate having enhanced stability in blood and process for the preparation thereof |
Country Status (2)
| Country | Link |
|---|---|
| KR (1) | KR20040083268A (en) |
| WO (1) | WO2004083242A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006094530A1 (en) * | 2005-03-11 | 2006-09-14 | Siegfried Ltd. | Di-polymer protein conjugates and processes for their preparation |
| EP1834963A1 (en) * | 2006-03-13 | 2007-09-19 | Siegfried Ltd. | Di-polymer protein conjugates and processes for their preparation |
| WO2008060002A1 (en) * | 2006-11-17 | 2008-05-22 | Dong-A Pharm. Co., Ltd. | Polyethylene glycol-g-csf conjugate |
| WO2012021088A1 (en) * | 2010-08-13 | 2012-02-16 | Closed Joint Stock Company "Biocad" | A novel conjugate of granulocyte colony-stimulating factor (g-csf) with polyethylene glycol |
| CN102485742A (en) * | 2010-12-02 | 2012-06-06 | 山东新时代药业有限公司 | Preparation method and separation and purification method of polyethylene glycol single modified recombinant human granulocyte-colony stimulating factor |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7737260B2 (en) | 2003-11-13 | 2010-06-15 | Hanmi Pharm. Co., Ltd | Protein complex using an immunoglobulin fragment and method for the preparation thereof |
| KR100735784B1 (en) * | 2005-07-20 | 2007-07-06 | 재단법인 목암생명공학연구소 | Human Granulocyte Colony Stimulator Variants and Chemical Conjugates thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5824778A (en) * | 1988-12-22 | 1998-10-20 | Kirin-Amgen, Inc. | Chemically-modified G-CSF |
| KR20030045415A (en) * | 2001-12-04 | 2003-06-11 | 선바이오(주) | Conjugates of granulocyte-colony stimulating factor and polyethylene glycol derivatives |
-
2003
- 2003-03-21 KR KR1020030017867A patent/KR20040083268A/en not_active Ceased
-
2004
- 2004-03-17 WO PCT/KR2004/000569 patent/WO2004083242A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5824778A (en) * | 1988-12-22 | 1998-10-20 | Kirin-Amgen, Inc. | Chemically-modified G-CSF |
| KR20030045415A (en) * | 2001-12-04 | 2003-06-11 | 선바이오(주) | Conjugates of granulocyte-colony stimulating factor and polyethylene glycol derivatives |
Non-Patent Citations (2)
| Title |
|---|
| LEE K.C. ET AL.: "Identification of the modifying sites of mono-PEGylated salmon calcitonins by capillary electrophoresis and MALDI-TOF mass spectrometry", J. CHROMATOGR. B., vol. 754, no. 1, 2001, pages 259 - 263, XP004232013 * |
| PARK, T.G. ET AL.: "Preparation and characterization of mono-PEGylated epidermal growth factor: evaluation of in vitro biologic activity", PHARM. RES., vol. 19, no. 6, 2002, pages 845 - 851, XP002904848 * |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006094530A1 (en) * | 2005-03-11 | 2006-09-14 | Siegfried Ltd. | Di-polymer protein conjugates and processes for their preparation |
| WO2006095029A3 (en) * | 2005-03-11 | 2006-11-30 | Siegfried Ltd | Di-polymer protein conjugates and processes for their preparation |
| EP1834963A1 (en) * | 2006-03-13 | 2007-09-19 | Siegfried Ltd. | Di-polymer protein conjugates and processes for their preparation |
| WO2007104768A3 (en) * | 2006-03-13 | 2007-11-08 | Siegfried Ltd | Di-polymer protein conjugates and processes for their preparation |
| WO2008060002A1 (en) * | 2006-11-17 | 2008-05-22 | Dong-A Pharm. Co., Ltd. | Polyethylene glycol-g-csf conjugate |
| US8329191B2 (en) | 2006-11-17 | 2012-12-11 | Dong-A-Pharm. Co., Ltd. | Three-branched polyethylene glycol-G-CSF conjugate |
| WO2012021088A1 (en) * | 2010-08-13 | 2012-02-16 | Closed Joint Stock Company "Biocad" | A novel conjugate of granulocyte colony-stimulating factor (g-csf) with polyethylene glycol |
| RU2446173C1 (en) * | 2010-08-13 | 2012-03-27 | Зао "Биокад" | New functional, high-purity stable conjugate of granulocyte colony-stimulating factor (g-csf) and polyethylene glycol with prolonged biological action, applicable for medical purposes, and based immunobiological agent |
| EA019043B1 (en) * | 2010-08-13 | 2013-12-30 | Закрытое Акционерное Общество "Биокад" | NEW CONJUGATE OF GRANULOCITAR COLONIUS STIMULATION FACTOR (G-CSF) WITH POLYETHYLENE GLYCOL |
| CN102485742A (en) * | 2010-12-02 | 2012-06-06 | 山东新时代药业有限公司 | Preparation method and separation and purification method of polyethylene glycol single modified recombinant human granulocyte-colony stimulating factor |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20040083268A (en) | 2004-10-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU736067B2 (en) | Erythropoietin derivatives | |
| US8932828B2 (en) | Method for preparing recombinant granulocyte colony stimulating factor cysteine muteins | |
| EP1121155B1 (en) | Site-directed dual pegylation of proteins | |
| JP5336372B2 (en) | G-CSF site-specific monoconjugate | |
| EP0442724B1 (en) | Modified hIL-6 | |
| CN100528235C (en) | Conjugates of erythropoietin | |
| US6586398B1 (en) | Chemically modified novel erythropoietin stimulating protein compositions and methods | |
| KR100203824B1 (en) | Compositions and methods for stimulating megakaryocyte growth and differentiation | |
| KR100961859B1 (en) | G-CS conjugate | |
| CA2460489A1 (en) | Pegylated and diglycosylated erythropoietin | |
| JP2000508356A (en) | Improved interferon-polymer conjugate | |
| EP1762574B1 (en) | Chemical modification of proteins to improve biocompatibility and bioactivity | |
| Long et al. | Design of homogeneous, monopegylated erythropoietin analogs with preserved in vitro bioactivity | |
| KR100507796B1 (en) | Peg-biologically active polypeptide homodimer conjugate having enhanced half life in blood and process for the preparation thereof | |
| JP2002509691A (en) | Production and use of recombinant protein multimers with altered biological activity | |
| WO2004083242A1 (en) | Human granulocyte-colony stimulating factor conjugate having enhanced stability in blood and process for the preparation thereof | |
| KR100694994B1 (en) | Human granulocyte colony forming factor homologue | |
| KR20110014564A (en) | Double-stranded polyethylene glycol-modified growth hormones, methods of making and uses thereof | |
| JP4782834B2 (en) | Granulocyte colony stimulating factor (G-CSF) variant and chemically conjugated polypeptide thereof | |
| CA2092542A1 (en) | Hemopoietic stem cell multiplier | |
| MXPA01003764A (en) | Site-directed dual pegylation of proteins for improved bioactivity and biocompatibility | |
| MXPA00001208A (en) | Chemical modification of proteins to improve biocompatibility and bioactivity |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| 122 | Ep: pct application non-entry in european phase |