WO2004044141A2 - Conjugated oligomeric compounds and their use in gene modulation - Google Patents
Conjugated oligomeric compounds and their use in gene modulation Download PDFInfo
- Publication number
- WO2004044141A2 WO2004044141A2 PCT/US2003/035088 US0335088W WO2004044141A2 WO 2004044141 A2 WO2004044141 A2 WO 2004044141A2 US 0335088 W US0335088 W US 0335088W WO 2004044141 A2 WO2004044141 A2 WO 2004044141A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- oligomeric compound
- conjugate moiety
- oligomeric
- nucleic acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C1COC(CC(*)(*)*)C(*)C1 Chemical compound *C1COC(CC(*)(*)*)C(*)C1 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1137—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against enzymes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H21/00—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids
- C07H21/04—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids with deoxyribosyl as saccharide radical
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1138—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against receptors or cell surface proteins
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering nucleic acids [NA]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/315—Phosphorothioates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
- C12N2310/3513—Protein; Peptide
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
- C12N2310/3515—Lipophilic moiety, e.g. cholesterol
Definitions
- the present invention provides modified oligomeric compounds that modulate gene expression via an RNA interference pathway.
- the oligomeric compounds of the invention include one or more modifications thereon resulting in differences in various physical properties and attributes compared to wild type nucleic acids.
- the modified oligonucleotides are used alone or in compositions to modulate the targeted nucleic acids.
- the modifications include the attachment of one or more conjugate moieties to the oligomeric compound.
- the conjugate moieties can modify or enhance the pharmacokinetic and phamacodynamic properties of the attached oligomeric compound.
- dsRNA double-stranded RNA
- Cosuppression since the expression of both the introduced gene and the homologous endogenous gene was suppressed (Napoli et al., Plant Cell, 1990, 2, 279-289; Jorgensen et al, Plant Mol. Biol, 1996, 31, 957-973). [0003] Cosuppression has since been found to occur in many species of plants, fungi, and has been particularly well characterized in Neurospora crassa, where it is known as “quelling” (Cogoni and Macino. Genes Dev. 2000, 10, 638-643; Guru, Nature, 2000, 404, 804-808).
- PCT publication WO 01/48183 discloses methods of inhibiting expression of a target gene in a nematode worm involving feeding to the worm a food organism which is capable of producing a double-stranded RNA structure having a nucleotide sequence substantially identical to a portion of the target gene following ingestion of the food organism by the nematode, or by introducing a DNA capable of producing the double-stranded RNA structure (Bogaert et al., 2001).
- dsRNA double-stranded RNA interference
- dsRNA double-stranded RNA
- RNAi short interfering RNAs
- siRNAs short interfering RNAs
- the Drosophila embryo extract system has been exploited, using green fluorescent protein and luciferase tagged siRNAs, to demonstrate that siRNAs can serve as primers to transform the target mRNA into dsRNA.
- the nascent dsRNA is degraded to eliminate the incorporated target mRNA while generating new siRNAs in a cycle of dsRNA synthesis and degradation.
- Evidence is also presented that mRNA-dependent siRNA incorporation to form dsRNA is carried out by an RNA-dependent RNA polymerase activity (RdRP) (Lipardi et al., Cell, 2001, 107, 297- 307).
- RdRP RNA- dependent RNA polymerase activity
- RNA interference RNA interference
- secondary siRNAs RNA-directed RNA polymerase (RdRP)
- RdRP RNA-directed RNA polymerase
- RNA oligomers of antisense polarity can be potent inducers of gene silencing.
- antisense RNAs act independently of the RNAi genes rde-1 and rde-4 but require the mutator/RNAi gene mut-7 and a putative DEAD box RNA helicase, mut-14.
- elegans has demonstrated modification of the internucleotide linkage (phosphorothioate) to not interfere with activity (Parrish et al, Molecular Cell, 2000, 6, 1077-1087.) It was also shown by Parrish et al, that chemical modification like 2'-amino or 5-iodouridine are well tolerated in the sense strand but not the antisense strand of the dsRNA suggesting differing roles for the 2 strands in RNAi. Base modification such as guanine to inosine (where one hydrogen bond is lost) has been demonstrated to decrease RNAi activity independently of the position of the modification (sense or antisense). Some "position independent" loss of activity has been observed following the introduction of mismatches in the dsRNA trigger.
- RNA-DNA heteroduplexes did not serve as triggers for RNAi.
- dsRNA containing 2'-F-2'-deoxynucleosides appeared to be efficient in triggering RNAi response independent of the position (sense or antisense) of the 2'-F-2'- deoxynucleosides.
- RNA interference pathway for modulation of gene expression is an effective means for modulating the levels of specific gene products and, thus, would be useful in a number of therapeutic, diagnostic, and research applications involving gene silencing.
- the present invention therefore provides oligomeric compounds and compositions thereof useful for modulating gene expression pathways, including those relying on mechanisms of action such as RNA interference and dsRNA enzymes, as well as antisense and non- antisense mechanisms.
- RNA interference and dsRNA enzymes as well as antisense and non- antisense mechanisms.
- antisense and non- antisense mechanisms One having skill in the art, once armed with this disclosure will be able, without undue experimentation, to identify preferred oligomeric compounds for these uses.
- compositions comprising a first oligomeric compound and a second oligomeric compound, wherein at least a portion of the first oligomeric compound is capable of hybridizing with at least a portion of the second oligomeric compound, wherein at least a portion of the first oligomeric compound is capable of hybridizing to a target nucleic acid, and wherein at least one of the first and second oligomeric compounds comprises at least one conjugate moiety.
- compositions comprising a first oligomeric compound capable of hybridizing to a target nucleic acid; optionally, a second oligomeric compound hybridizable to the first oligomeric compound; at least one protein comprising at least a portion of a RNA-induced silencing complex
- composition comprises at least one oligomeric compound comprising at least one conjugate moiety.
- the present invention further provides oligomeric compounds comprising a first region and a second region, wherein the first region is capable of hybridizing with the second region, wherein a portion of the oligomeric compound is capable of hybridizing to a target nucleic acid, and wherein the oligomeric compound further comprises at least one conjugate moiety.
- compositions comprising any of the above compositions or oligomeric compounds and a pharmaceutically acceptable carrier.
- Methods for modulating the expression of a target nucleic acid in a cell comprise contacting the cell with any of the above compositions or oligomeric compounds.
- Methods of treating or preventing a disease or condition associated with a target nucleic acid comprise administering to a patient having or predisposed to the disease or condition a therapeutically effective amount of any of the above compositions or oligomeric compounds.
- oligomeric compounds of the invention are believed to modulate gene expression by hybridizing to a nucleic acid target resulting in loss of normal function of the target nucleic acid.
- target nucleic acid or “nucleic acid target” is used for convenience to encompass any nucleic acid capable of being targeted including without limitation DNA, RNA (including pre-mRNA and mRNA or portions thereof) transcribed from such DNA, and also cDNA derived from such RNA.
- modulation of gene expression is effected via modulation of a RNA associated with the particular gene RNA.
- the invention provides for modulation of a target nucleic acid where the target nucleic acid is a messenger RNA.
- the messenger RNA is degraded by the RNA interference mechanism as well as other mechanism wherein double stranded RNA/RNA structures are recognized and degraded, cleaved or otherwise rendered inoperable.
- RNA to be interfered with can include replication and transcription.
- Replication and transcription for example, can be from an endogenous cellular template, a vector, a plasmid construct or otherwise.
- the functions of RNA to be interfered with can include functions such as translocation of the RNA to a site of protein translation, translocation of the RNA to sites within the cell which are distant from the site of RNA synthesis, translation of protein from the RNA, splicing of the RNA to yield one or more RNA species, and catalytic activity or complex formation involving the RNA which may be engaged in or facilitated by the RNA.
- modulation and modulation of expression mean either an increase (stimulation) or a decrease (inhibition) in the amount or levels of a nucleic acid molecule encoding the gene, e.g., DNA or RNA. Inhibition is often the preferred form of modulation of expression and mRNA is often a preferred target nucleic acid.
- the present invention provides, wter alia, oligomeric compounds and compositions containing the same wherein the oligomeric compoimd includes one or more conjugate moieties.
- the oligomeric compounds of the present invention can be covalently attached, optionally through one or more linkers, to one or more conjugate moieties.
- the resulting conjugate compounds can have modified or enhanced pharmacokinetic, pharamcodynamic, and other properties compared with non-conjugated oligomeric compounds.
- a conjugate moiety that can modify or enhance the pharmacokinetic properties of an oligomeric compound can improve cellular distribution, bioavailability, metabolism, excretion, permeability, and/or cellular uptake of the oligomeric compound.
- a conjugate moiety that can modify or enhance pharmacodynamic properties of an oligomeric compound can improve activity, resistance to degradation, sequence-specific hybridization, uptake, and the like.
- conjugate moieties can include lipophilic molecules
- aromatic and non-aromatic including steroid molecules; proteins (e.g., antibodies, enzymes, serum proteins); peptides; vitamins (water-soluble or lipid-soluble); polymers (water-soluble or lipid-soluble); small molecules including drugs, toxins, reporter molecules, and receptor ligands; carbohydrate complexes; nucleic acid cleaving complexes; metal chelators (e.g., porphyrins, texaphyrins, crown ethers, etc.); intercalators including hybrid photonuclease/intercalators; crosslinking agents (e.g., photoactive, redox active), and combinations and derivatives thereof.
- proteins e.g., antibodies, enzymes, serum proteins
- peptides e.g., vitamins (water-soluble or lipid-soluble); polymers (water-soluble or lipid-soluble); small molecules including drugs, toxins, reporter molecules, and receptor ligands; carbohydrate complexes; nucleic acid cleaving complexes
- Lipophilic conjugate moieties can be used, for example, to counter the hydrophilic nature of an oligomeric compound and enhance cellular penetration.
- Lipophilic moieties include, for example, steroids and related compounds such as cholesterol (U.S. Pat. No. 4,958,013 and Letsinger et al., Proc. Natl. Acad. Sci.
- thiocholesterol (Oberhauser et al, Nucl Acids Res., 1992, 20, 533), lanosterol, coprostanol, stigmasterol, ergosterol, calciferol, cholic acid, deoxycholic acid, estrone, estradiol, estratriol, progesterone, stilbestrol, testosterone, androsterone, deoxycorticosterone, cortisone, 17-hydroxycorticosterone, their derivatives, and the like.
- Other lipophilic conjugate moieties include aliphatic groups, such as, for example, straight chain, branched, and cyclic alkyls, alkenyls, and alkynyls.
- the aliphatic groups can have, for example, 5 to about 50, 6 to about 50, 8 to about 50, or 10 to about 50 carbon atoms.
- Example aliphatic groups include undecyl, dodecyl, hexadecyl, heptadecyl, octadecyl, nonadecyl, terpenes, bornyl, adamantyl, derivatives thereof and the like.
- one or more carbon atoms in the aliphatic group can be replaced by a heteroatom such as O, S, or N (e.g., geranyloxyhexyl).
- a heteroatom such as O, S, or N (e.g., geranyloxyhexyl).
- suitable lipophilic conjugate moieties include aliphatic derivatives of glycerols such as alkylglycerols, bis(alkyl)glycerols, tris(alkyl)glycerols, monoglycerides, diglycerides, and triglycerides.
- the lipophilic conjugate is di-hexyldecyl-rac-glycerol or 1,2-di-O- hexyldecyl-rac-glycerol (Manoharan et al., Tetrahedron Lett., 1995, 36, 3651; Shea, et al., Nuc. Acids Res., 1990, 18, 3777) or phosphonates thereof.
- Saturated and unsaturated fatty functionalities such as, for example, fatty acids, fatty alcohols, fatty esters, and fatty amines, can also serve as lipophilic conjugate moieties.
- the fatty functionalities can contain from about 6 carbons to about 30 or about 8 to about 22 carbons.
- Example fatty acids include, capric, caprylic, lauric, palmitic, myristic, stearic, oleic, linoleic, linolenic, arachidonic, eicosenoic acids and the like.
- lipophilic conjugate groups can be polycyclic aromatic groups having from 6 to about 50, 10 to about 50, or 14 to about 40 carbon atoms.
- Example polycyclic aromatic groups include pyrenes, purines, acridines, xanthenes, fluorenes, phenanthrenes, anthracenes, quinolines, isoquinolines, naphthalenes, derivatives thereof and the like.
- Other suitable lipophilic conjugate moieties include menthols, trityls
- oligomeric compounds e.g., dimethoxytrityl (DMT)
- DMT dimethoxytrityl
- phenoxazines e.g., lipoic acid, phospholipids, ethers, thioethers (e.g., hexyl-S-tritylthiol), derivatives thereof and the like.
- Preparation of lipophilic conjugates of oligomeric compounds are well-described in the art, such as in, for example, VMware-Behmoaras et al, EMBO J., 1991, 10, 1111; Kabanov et al., FEBSLett., 1990, 259, 327; Svinarchuk et al, Biochimie, 1993, 75, 49; (Mishra et al., Biochim. Biophys. Acta, 1995, 1264, 229, and Manoharan et al., Tetrahedron Lett., 1995, 36, 3651.
- Oligomeric compounds containing conjugate moieties with affinity for low density lipoprotein (LDL) can help provide an effective targeted delivery system.
- High expression levels of receptors for LDL on tumor cells makes LDL an attractive carrier for selective delivery of drugs to these cells (Rump, et al., Bioconjugate Chem., 1998, 9, 341; Firestone, Bioconjugate Chem., 1994, 5, 105; Mishra, et al., Biochim. Biophys. Acta, 1995, 1264, 229).
- Moieties having affinity for LDL include many lipophilic groups such as steroids (e.g., cholesterol), fatty acids, derivatives thereof and combinations thereof.
- conjugate moieties having LDL affinity can be dioleyl esters of cholic acids such as chenodeoxycholic acid and lithocholic acid.
- Conjugate moieties can also include vitamins. Vitamins are known to be transported into cells by numerous cellular transport systems. Typically, vitamins can be classified as water soluble or lipid soluble. Water soluble vitamins include thiamine, riboflavin, nicotinic acid or niacin, the vitamin B 6 pyridoxal group, pantothenic acid, biotin, folic acid, the B ]2 cobamide coenzymes, inositol, choline and ascorbic acid. Lipid soluble vitamins include the vitamin A family, vitamin D, the vitamin E tocopherol family and vitamin K (and phytols). Related compounds include retinoid derivatives such as tazarotene and etretinate. [0040] In some embodiments, the conjugate moiety includes folic acid
- (folate) and/or one or more of its various forms such as dihydrofolic acid, tetrahydrofolic acid, folinic acid, pteropolyglutamic acid, dihydrofolates, tetrahydrofolates, tetrahydropterins, 1-deaza, 3-deaza, 5-deaza, 8-deaza, 10-deaza, 1,5-dideaza, 5,10-dideaza, 8,10-dideaza and 5,8-dideaza folate analogs, and antifolates.
- Folate is involved in the biosynthesis of nucleic acids and therefore impacts the survival and proliferation of cells.
- Folate cofactors play a role in the one-carbon transfers that are needed for the biosynthesis of pyrimidine nucleosides.
- Folate receptors also tend to be overexpressed in many human cancer cells, and folate- mediated targeting of oligonucleotides to ovarian cancer cells has been reported (Li, et al, Pharm. Res. 1998, 15, 1540, which is incorporated herein by reference in its entirety).
- Preparation of folic acid conjugates of nucleic acids are described in, for example, U.S. Pat. No. 6,528,631, which is incorporated herein by reference in its entirety.
- Vitamin conjugate moieties include, for example, vitamin A (retinol) and/or related compounds.
- the vitamin A family (retinoids), including retinoic acid and retinol, are typically absorbed and transported to target tissues through their interaction with specific proteins such as cytosol retinol-binding protein type II (CRBP-II), retinol-binding protein (RBP), and cellular retinol-binding protein (CRBP).
- CRBP-II cytosol retinol-binding protein type II
- RBP retinol-binding protein
- CRBP cellular retinol-binding protein
- the vitamin A family of compounds can be attached to oligomeric compounds via acid or alcohol functionalities found in the various family members.
- conjugation of an N-hydroxy succinimide ester of an acid moiety of retinoic acid to an amine function on a linker pendant to an oligonucleotide can result in linkage of vitamin A compound to the oligomeric compound via an amide bond.
- retinol can be converted to its phosphoramidite, which is useful for 5' conjugation.
- alpha-Tocopherol (vitamin E) and the other tocopherols (beta through zeta) can be conjugated to oligomeric compounds to enhance uptake because of their lipophilic character.
- vitamin D, and its ergosterol precursors can be conjugated to oligomeric compounds through their hydroxyl groups by first activating the hydroxyl groups to, for example, hemisuccinate esters. Conjugation can then be effected directly to the oligomeric compound or to an arninolinker pendant from the oliogmeric compound.
- vitamins that can be conjugated to oligomeric compounds in a similar manner on include thiamine, riboflavin, pyridoxine, pyridoxamine, pyridoxal, deoxypyridoxine.
- Lipid soluble vitamin K's and related quinone-containing compounds can be conjugated via carbonyl groups on the quinone ring.
- the phytol moiety of vitamin K can also serve to enhance binding of the oligomeric compounds to cells.
- Pyridoxal (vitamin B 6 ) has specific B 6 -binding proteins. The role of these proteins in pyridoxal transport has been studied by Zhang et al., Proc. Natl. Acad. Sci. USA, 1991, 88, 10407.
- Other pyridoxal family members include pyridoxine, pyridoxamine, pyridoxal phosphate, and pyridoxic acid.
- Pyridoxic acid, niacin, pantothenic acid, biotin, folic acid and ascorbic acid can be conjugated to oligomeric compounds, for example, using N-hydroxysuccinimide esters that are reactive with aminolinkers located on the oliogmeric compound, as described above for retinoic acid.
- Vitamin conjugate moieties can also be used to facilitate the targeting of specific cells or tissues.
- vitamin D and analogs thereof can assist in transporting conjugated oligomeric compounds to keratinocytes, dermal fibroblasts, and other cells containing vitamin D 3 nuclear receptors.
- Vitamin A and other retinoids can be used to target cells with retinoid X receptors. Accordingly, vitamin-containing conjugate moieties can be useful in treating, for example, skin disorders such as psoriasis.
- Conjugate moieties can also include polymers.
- Polymers can provide added bulk and various functional groups to affect permeation, cellular transport, and localization of the conjugated oligomeric compound. For example, increased hydrodynamic radius caused by conjugation of an oligomeric compound with a polymer can help prevent entry into the nucleus and encourage localization in the cytoplasm.
- the polymer does not substantially reduce cellular uptake or interfere with hybridization to a complementary strand or other target.
- the conjugate polymer moiety has, for example, a molecular weight of less than about 40, less than about 30, or less than about 20 kDa.
- polymer conjugate moieties can be water-soluble and optionally further comprise other conjugate moieties such as peptides, carbohydrates, drugs, reporter groups, or further conjugate moieties.
- polymer conjugates include polyethylene glycol (PEG) and copolymers and derivatives thereof. Conjugation to PEG has been shown to increase nuclease stability of an oligomeric compound.
- PEG conjugate moieties can be of any molecular weight including for example, about 100, about 500, about 1000, about 2000, about 5000, about 10,000 and higher.
- the PEG conjugate moieties contains at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 15, at least 20, or at least 25 ethylene glycol residues.
- the PEG conjugate moiety contains from about 4 to about 10, about 4 to about 8, about 5 to about 7, or about 6 ethylene glycol residues.
- the PEG conjugate moiety can also be modified such that a terminal hydroxyl is replaced by alkoxy, carboxy, acyl, amido, or other functionality.
- Other conjugate moieties such as reporter groups including, for example, biotin or fluorescein can also be attached to a PEG conjugate moiety.
- Copolymers of PEG are also suitable as conjugate moieties.
- PEG conjugate moieties and preparation of corresponding conjugated oligomeric compounds is described in, for example, U.S. Pat. Nos. 4,904,582 and 5,672,662, each of which is incorporated by reference herein in its entirety. Oligomeric compounds conjugated to one or more PEG moieties are available commercially.
- polymers suitable as conjugate moieties include polyamines, polypeptides, polymethacrylates (e.g., hydroxylpropyl methacrylate (HPMA)), poly(L-lactide), poly(DL lactide-co-glycolide (PGLA), polyacrylic acids, polyethylenimines (PEI), polyalkylacrylic acids, polyurethanes, polyacrylamides, N- alkylacrylamides, polyspermine (PSP), polyethers, cyclodextrins, derivatives thereof and co-polymers thereof.
- Many polymers, such as PEG and polyamines have receptors present in certain cells, thereby facilitating cellular uptake.
- Polyamines and other amine-containing polymers can exist in protonated form at physiological pH, effectively countering an anionic backbone of some oligomeric compounds, effectively enhancing cellular permeation.
- Some example polyamines include polypeptides (e.g., polylysine, polyornithine, polyhistadine, polyarginine, and copolymers thereof), triethylenetetraamine, spermine, polyspermine, spermidine, synnorspermidine, C-branched spermidine, and derivatives thereof.
- Preparation and biological activity of polyamine conjugates are described, for example, in Guzaev, et al, Bioorg. Med. Chem. Lett., 1998, 8, 3671; Corey, et al, J Am.
- Example polypeptide conjugates of oligonucleotides are provided in, for example, Wei, et al., Nucleic Acids Res., 1996, 24, 655 and Zhu, et al., Antisense Res. Dev., 1993, 3, 265.
- Dendrimeric polymers can also be used as conjugate moieties, such as described in U.S. Pat. No. 5,714,166, which is incorporated herein by reference in its entirety.
- amine-containing moieties can also serve as suitable conjugate moieties due to, for example, the formation of cationic species at physiological conditions.
- Example amine-containing moieties include 3-aminopropyl, 3-(N,N-dimethylamino)propyl, 2-(2-(N,N-dimethylamino)ethoxy)ethyl, 2-(N-(2-aminoethyl)-N- methylaminooxy)ethyl, 2-(l-imidazolyl)ethyl, and the like.
- the G-clamp moiety can also serve as an amine-containing conjugate moiety (Lin, et al., J. Am. Chem. Soc, 1998, 120, 8531).
- Conjugate moieties can also include peptides. Suitable peptides can have from 2 to about 30, 2 to about 20, 2 to about 15, or 2 to about 10 amino acid residues. Amino acid residues can be naturally or non-naturally occurring, including both D and L isomers.
- peptide conjugate moieties are pH sensitive peptides such as fusogenic peptides.
- Fusogenic peptides can facilitate endosomal release of agents such as oligomeric compounds to the cytoplasm. It is believed that fusogenic peptides change conformation in acidic pH, effectively destabilizing the endosomal membrane thereby enhancing cytoplasmic delivery of endosomal contents.
- Example fusogenic peptides include peptides derived from polymyxin B, influenza HA2, GALA, KALA, EALA, melittin-derived peptide, a-helical peptide or Alzheimer ⁇ -amyloid peptide, and the like.
- oligonucleotides conjugated to fusogenic peptides are described in, for example, Bongartz, et al., Nucleic Acids Res., 1994, 22, 4681 and U.S. Pat. Nos. 6,559,279 and 6,344,436.
- peptides that can serve as conjugate moieties include delivery peptides which have the ability to transport relatively large, polar molecules (including peptides, oligonucleotides, and proteins) across cell membranes.
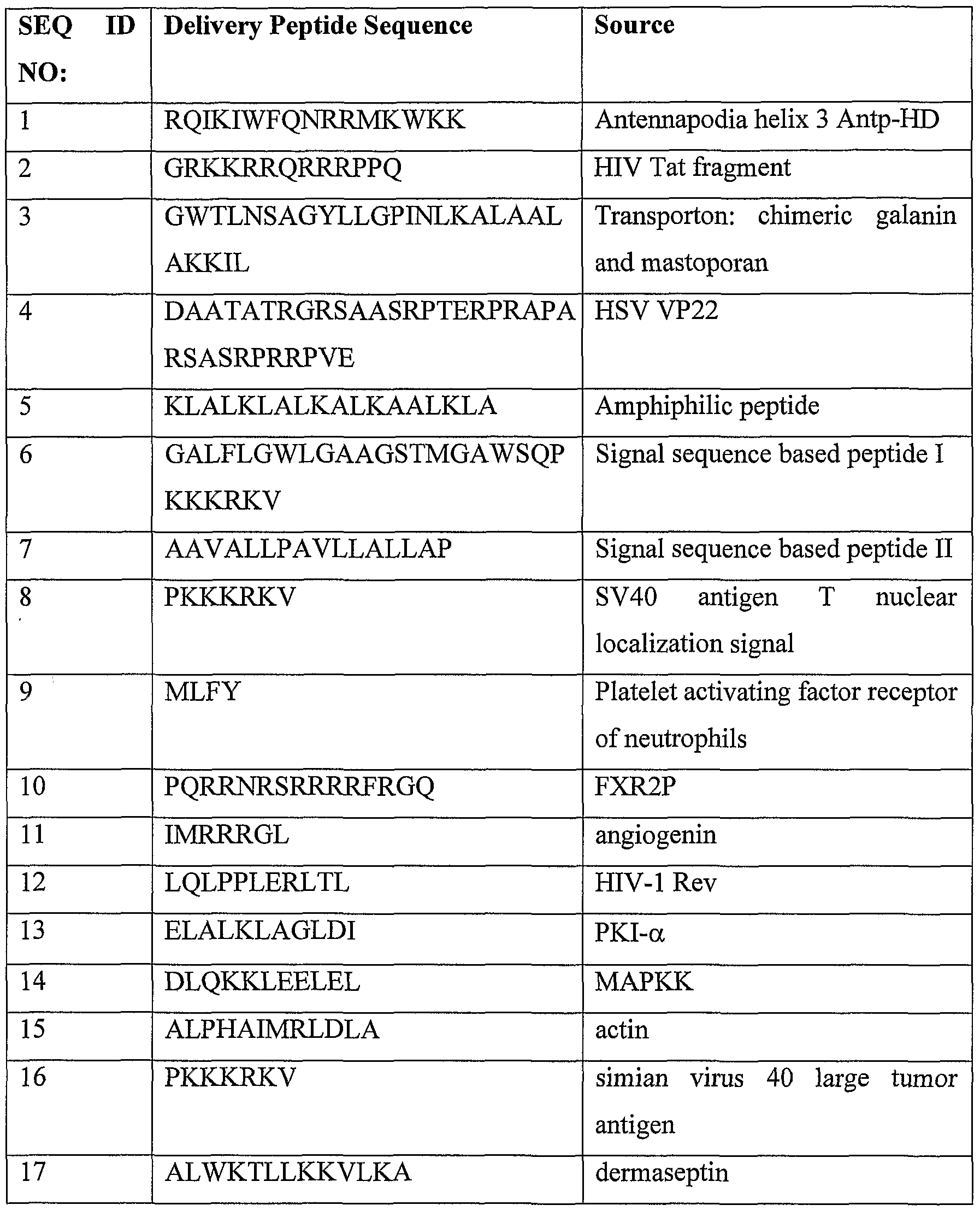
- Example delivery peptides include Tat peptide from HIV Tat protein and Ant peptide from Drosophila antenna protein. Conjugation of Tat and Ant with oligonucleotides is described in, for example, Astriab-Fisher, et al., Biochem. Pharmacol, 2000, 60, 83. These and other delivery peptides that can be used as conjugate moieties are provided below in Table I.
- Conjugated delivery peptides can help control localization of oligomeric compounds to specific regions of a cell, including, for example, the cytoplasm, nucleus, nucleolus, and endoplasmic reticulum (ER).
- Nuclear localization can be effected by conjugation of a nuclear localization signal (NLS).
- cytoplasmic localization can be facilitated by conjugation of a nuclear export signal (NES).
- NLS nuclear localization signal
- NES nuclear export signal
- Peptides suitable for localization of conjugated oligomeric compounds in the nucleus include, for example, N,N-dipalmitylglycyl-apo E peptide or N,N- dipalmitylglycyl-apolipoprotein E peptide (dpGapoE) (Liu, et al, Arterioscler. Thromb. Vase. Biol, 1999, 19, 2207; Chaloin, et al., Biochem. Biophys. Res. Commun., 1998, 243, 601).
- dpGapoE N,N-dipalmitylglycyl-apo E peptide
- dpGapoE N,N- dipalmitylglycyl-apolipoprotein E peptide
- Nucleus or nucleolar localization can also be facilitated by peptides having arginine and/or lysine rich motifs, such as in HIV-1 Tat, FXR2P, and angiogenin derived peptides (Lixin, et al, Biochem. Biophys. Res. Commun., 2001, 284, 185). Additionally, the nuclear localization signal (NLS) peptide derived from SV40 antigen T (Branden, et al., Nature Biotech, 1999, 17, 784) can be used to deliver conjugated oligomeric compounds to the nucleus of a cell.
- NLS nuclear localization signal
- Suitable peptides with nuclear or nucleolar localization properties are described in, for example, Antopolsky, et al., Bioconjugate Chem., 1999, 10, 598; Zanta, et al., Proc. Natl. Acad. Sci. USA, 1999 (simian virus 40 large tumor antigen); Hum. Mol. Genetics, 2000, 9, 1487; and FEBSLett., 2002, 532, 36).
- the delivery peptide for nucleus or nucleolar localization comprises at least three consecutive arginine residues or at least four consecutive arginine residues.
- Nuclear localization can also be facilitated by peptide conjugates containing RS, RE, or RD repeat motifs (Cazalla, et al., Mol Cell. Biol, 2002, 22, 6871).
- the peptide conjugate contains at least two RS, RE, or RD motifs.
- Localization of oligomeric compounds to the ER can be effected by, for example, conjugation to the signal peptide KDEL (SEQ ID NO: 18) (Arar, et al., Bioconjugate Chem., 1995, 6, 573; Pichon, et al., Mol. Pharmacol. 1997, 57, 431). [0057] Cytoplasmic localization of oligomeric compounds can be facilitated by conjugation to peptides having, for example, a nuclear export signal (NES) (Meunier, et al., Nucleic Acids Res., 1999, 27, 2730).
- NES nuclear export signal
- NES peptides include the leucine-rich NES peptides derived from HIV-1 Rev (Henderson, et al., Exp. Cell Res., 2000, 256, 213), transcription factor III A, MAPKK, PKI-alpha, cyclin Bl, and actin (Wada, et al., EMBO J., 1998, 17, 1635) and related proteins.
- Antimicrobial peptides such as dermaseptin derivatives, can also facilitate cytoplasmic localization (Hariton-Gazal, et al., Biochemistry, 2002, 41, 9208).
- Peptides containing RG and/or KS repeat motifs can also be suitable for directing oligomeric compounds to the cytoplasm.
- the peptide conjugate moieties contain at least two RG motifs, at least two KS motifs, or at least one RG and one KS motif.
- "peptide” includes not only the specific molecule or sequence recited herein (if present), but also includes fragments thereof and molecules comprising all or part of the recited sequence, where desired functionality is retained. In some embodiments, peptide fragments contain no fewer than 6 amino acids. Peptides can also contain conservative amino acid substitutions that do not substantially change its functional characteristics.
- Peptides also include homologous peptides. Homology can be measured according to percent identify using, for example, the BLAST algorithm (default parameters for short sequences). For example, homologous peptides can have greater than 50, 60, 70, 80, 90, 95, or 99 percent identity. Methods for conjugating peptides to oligomeric compounds such as oligonucleotides is described in, for example, U.S. Pat. No. 6,559,279, which is incorporated herein by reference in its entirety.
- nucleic acids can also serve as conjugate moieties that can affect localization of conjugated oligomeric compounds in a cell.
- nucleic acid conjugate moieties can contain poly A, a motif recognized by poly A binding protein (PABP), which can localize poly A-containing molecules in the cytoplasm (Gorlach, et al., Exp. Cell Res., 1994, 211, 400.
- PABP poly A binding protein
- the nucleic acid conjugate moiety contains at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 15, at least 20, and at least 25 consecutive A bases.
- the nucleic acid conjugate moiety can also contain one or more AU-rich sequence elements (AREs).
- AREs AU-rich sequence elements
- AREs are recognized by ELAV family proteins which can facilitate localization to the cytoplasm (Bollig, et al, Biochem. Bioophys. Res. Commun., 2003, 301, 665).
- Example AREs include UUAUUUAUU and sequences containing multiple repeats of this motif.
- the nucleic acid conjugate moiety contains two or more AU or AUU motifs.
- the nucleic acid conjugate moiety can also contain one or more CU-rich sequence elements (CREs) (Wein, et al, Eur. J. Biochem., 2003, 270, 350) which can bind to proteins HuD and/or HuR of the ELAV family of proteins.
- CREs CU-rich sequence elements
- the nucleic acid conjugate moiety contains the motif (CUUU)n .
- n can be 1 to about 20, 1 to about 15, or 1 to about 11.
- the (CUUU) n motif can optionally be followed or preceded by one or more U. In some embodiments, n is about 9 to about 12 or about 11.
- the nucleic acid conjugate moiety can also include substrates of hnRNP proteins (heterogeneous nuclear ribonucleoprotein), some of which are involved in shuttling nucleic acids between the nucleus and cytoplasm, (e.g., nhRNP Al and nhRNP K; see, e.g., Mili, et al, Mol. Cell Biol, 2001, 21, 7307).
- hnRNP substrates include nucleic acids containing the sequence UAGGA/U or (GG)ACUAGC(A).
- Other nucleic acid conjugate moieties can include Y strings or other tracts that can bind to, for example, linRNP I .
- the nucleic acid conjugate can contain at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 15, at least 20, and at least 25 consecutive pyrimidine bases. In other embodiments the nucleic acid conjugate can contain greater than 50, greater than 60, greater than 70, greater than 80, greater than 90, or greater than 95 percent pyrimidine bases.
- nucleic acid conjugate moieties can include pumilio (puf protein) recognition sequences such as described in Wang, et al., Cell, 2002, 110, 501.
- Example pumilio recognition sequences can include UGUANAUR, where N can be any base and R can be a purine base.
- nucleic acid conjugate moieties conataining AREs and/or CREs can facilitate localization of conjugated oligomeric compounds to the cytoplasm (e.g., hnRNP Al or K) or nucleus (e.g., hnRNP I).
- nucleus localization can be facilitated by nucleic acid conjugate moieties containing polypyrimidine tracts.
- Small molecule conjugate moieties often have specific interactions with certain receptors or other biomolecules, thereby allowing targeting of conjugated oligomeric compounds to specific cells or tissues.
- Example small molecule conjugate moieties include mycophenolic acid (inhibitor of inosine-5'-monophosphate dihydrogenase; useful for treating psoriasis and other skin disorders), curcumin (has therapeutic applications to psoriasis, cancer, bacterial and viral diseases).
- small molecule conjugate moieties can be ligands of serum proteins such as human serum albumin (HSA).
- HSA human serum albumin
- ligands of HSA include, for example, arylpropionic acids, ibuprofen, warfarin, phenylbutazone, suprofen, carprofen, fenfufen, ketoprofen, aspirin, indomethacin, (S)-(+)-pranoprofen, dansylsarcosine, 2,3,5-triiodobenzoic acid, flufenamic acid, folinic acid, benzothiadiazide, chlorothiazide, diazepines, indomethicin, barbituates, cephalosporins, sulfa drugs, antibacterials, antibiotics (e.g., puromycin and pamamycin), and the like. Oligonucleotide-drug conjugates and their preparation are described in, for example, WO 00/76554, which is incorporated herein by reference in its entirety.
- small molecule conjugates can target or bind certain receptors or cells.
- T-cells are known to have exposed amino groups that can form Schiff base complexes with appropriate molecules.
- small molecules containing functional groups such as aldehydes that can interact or react with exposed amino groups can also be suitable conjugate moieties.
- Tucaresol and related compounds can be conjugated to oligomeric compounds in such as way as to leave the aldehyde free to interact with T-cell targets. Interaction of tucaresol with T-cells in believed to result in therapeutic potentiation of the immune system by Schiff-base formation (Rhodes, et al., Nature, 1995, 377, 6544).
- Reporter groups that are suitable as conjugate moieties include any moiety that can be detected by, for example, spectroscopic means.
- Example reporter groups include dyes, flurophores, phosphors, radiolabels, and the like.
- the reporter group is biotin, flourescein, rhodamine, coumarin, or related compounds. Reporter groups can also be attached to other conjugate moieties.
- conjugate moieties can include proteins, subunits, or fragments thereof. Proteins include, for example, enzymes, reporter enzymes, antibodies, receptors, and the like. In some embodiments, protein conjugate moieties can be antibodies or fragaments thereof (Kuijpers, et al, Bioconjugate Chem., 1993, 4, 94). Antibodies can be designed to bind to desired targets such as tumor and other disease-related antigens. In further embodiments, protein conjugate moieties can be serum proteins such as HAS or glycoproteins such as asialoglycoprotein (Rajur, et al., Bioconjugate Chem., 1997, 6, 935).
- oligomeric compounds can be conjugated to RNAi-related proteins, RNAi-related protein complexes, subunits, and fragments thereof.
- oligomeric compounds can be conjugated to Dicer or RISC.
- Other conjugate moieties can include, for example, oligosaccharides and carbohydrate clusters such as Tyr-Glu-Glu-(aminohexyl GalNAc) 3 (YEE(ahGalNAc) 3 ; a glycotripeptide that binds to Gal/GalNAc receptors on hepatocytes, see, e.g., Duff, et al., Methods Eanzymol, 2000, 313, 297); lysine-based galactose clusters (e.g., L 3 G 4 ; Biessen, et al., E>ev.
- conjugates can include oligosaccharides that can bind to carbohydrate recognition domains (CRD) found on the asiologlycoprotein-receptor (ASGP-R).
- CCD carbohydrate recognition domains
- ASGP-R asiologlycoprotein-receptor
- Example conjugate moieties containing oligosaccharides and/or carbohydrate complexes are provided in U.S. Pat. No. 6,525,031, which is incorporated herein by reference in its entirey.
- Intercalators and minor groove binders can also be suitable as conjugate moieties.
- the MGB can contain repeating DPI (l,2-dihydro-3H-pyrrolo(2,3-e)indole-7-carboxylate) subunits or derivatives thereof (Lukhtanov, et al., Bioconjugate Chem., 1996, 7, 564 and Afonina, et al., Proc. Natl. Acad. Sci. USA, 1996, 93, 3199).
- Suitable intercalators include, for example, polycyclic aromatics such as naphthalene, perylene, phenanthridine, benzophenanthridine, phenazine, anthraquinone, acridine, and derivatives thereof.
- Hybrid intercalator/ligands include the photonuclease/intercalator ligand 6-[[[9-[[6- (4-nitrobenzamido)hexyl]amino]acridin-4-yl]carbonyl]amino]hexan oyl- pentafluorophenyl ester. This compound is both an acridine moiety that is an intercalator and a p-nitro benzamido group that is a photonuclease.
- cleaving agents can serve as conjugate moieties. Cleaving agents can facilitate degradation of target, such as target nucleic acids, by hydrolytic or redox cleavage mechamisms.
- Cleaving groups that can be suitable as conjugate moieties include, for example, metallocomplexes, peptides, amines, enzymes, and constructs containing constituents of the active sites of nucleases such as imidazole, guanidinium, carboxyl, amino groups, etx.).
- Example metallocomplexes include, for example, Cu-terpyridyl complexes, Fe-porphyrin complexes, Ru-complexes, and lanthanide complexes such as various Eu(III) complexes (Hall, et al., Chem. Biol, 1994, 1, 185; Huang, et al., J. Biol. Inorg.
- nuclease active site constituents include bisimiazole and histamine.
- Cross-linking agents can also serve as conjugate moieties.
- Cross- linking agents facilitate the covalent linkage of the conjugated oligomeric compounds with other compounds.
- cross-linking agents can covalently link double-stranded nucleic acids, effectively increasing duplex stability and modulating pharmacokinetic properties.
- cross-linking agents can be photoactive or redox active.
- Example cross-linking agents include psoralens which can facilitate interstrand cross-linking of nucleic acids by photoactivation (Lin, et al, Faseb J, 1995, 9, 1371).
- cross-linking agents include, for example, mitomycin C and analogs thereof (Maruenda, et al., Bioconjugate Chem., 1996, 7, 541; Maruenda, et al., Anti-Cancer Drug Des., 1997, 12, 473; and Huh, et al, Bioconjugate Chem., 1996, 7, 659).
- Cross-linking mediated by mitomycin C can be effected by reductive activation, such as, for example, with biological reductants (e.g., NADPH-cytochrome c reductase/NADPH system).
- photo-crosslinking agents include aryl azides such as, for example, N-hydroxysucciniimidyl-4- azidobenzoate (HSAB) and N-succinimidyl-6(-4'-azido-2'-nitrophenyl- amino)hexanoate (SANPAH).
- HSAB N-hydroxysucciniimidyl-4- azidobenzoate
- SANPAH N-succinimidyl-6(-4'-azido-2'-nitrophenyl- amino)hexanoate
- Aryl azides conjugated to oligonucleotides effect crosslinking with nucleic acids and proteins upon irradiation. They can also crosslink with earner proteins (such as KLH or BSA).
- conjugate moieties include, for example, polyboranes, carboranes, metallopolyboranes, metallocarborane, derivatives thereof and the like (see, e.g., U.S. Pat. No. 5,272,250, which is incorporated herein by reference in its entirety),
- Conjugate moieties can be attached to the oligomeric compound directly or through a linking moiety (linker or tether).
- Linkers are bifunctional moieties that serve to covalently connect a conjugate moiety to an oligomeric compound.
- the linker comprises a chain structure or an oligomer of repeating units such as ethylene glyol or amino acid units.
- the linker can have at least two functionalities, one for attaching to the oligomeric compound and the other for attaching to the conjugate moiety.
- Example linker functionalities can be electrophilic for reacting with nucleophilic groups on the oligomer or conjugate moiety, or nucleophilic for reacting with electrophilic groups.
- linker functionalities include amino, hydroxyl, carboxylic acid, thiol, phosphoramidate, phophate, phosphite, unsaturations (e.g., double or triple bonds), and the like.
- Some example linkers include 8-amino-3,6-dioxaoctanoic acid (ADO), succinimidyl 4-(N-maleimidomethyl)cyclohexane-l-carboxylate (SMCC), 6- aminohexanoic acid (AHEX or AHA), 6-aminohexyloxy, 4-aminobutyric acid, 4- aminocyclohexylcarboxylic acid, succinimidyl 4-(N-maleimidomethyl)cyclohexane- l-carboxy-(6-amido-caproate) (LCSMCC), succinimidyl m-maleimido-benzoylate (MBS), succinimidyl N- ⁇ -maleimido-cap
- a wide variety of futher linker groups are known in the art that can be useful in the attachment of conjugate moieties to oligomeric compounds.
- a review of many of the useful linker groups can be found in, for example, Antisense Research and Applications, S. T. Crooke and B. Lebleu, Eds., CRC Press, Boca Raton, Fla., 1993, p. 303-350.
- a disulfide linkage has been used to link the 3' terminus of an oligonucleotide to a peptide (Corey, et al., Science 1987, 238, 1401; Zuckermann, et al, J Am. Chem. Soc. 1988, 110, 1614; and Corey, et al., J Am.
- N-Fmoc-O- DMT-3 -amino- 1,2-propanediol is commercially available from Clontech Laboratories (Palo Alto, Calif.) under the name 3'-Amine. It is also commercially available under the name 3'-Amino-Modifier reagent from Glen Research Corporation (Sterling, Va.).
- This reagent was also utilized to link a peptide to an oligonucleotide as reported by Judy, et al., Tetrahedron Letters 1991, 32, 879.
- a similar commercial reagent for linking to the 5 '-terminus of an oligonucleotide is 5'- Amino-Modifier C6. These reagents are available from Glen Research Corporation (Sterling, Va.). These compounds or similar ones were utilized by Krieg, et al, Antisense Research and Development 1991, 1, 161 to link fluorescein to the 5'- terminus of an oligonucleotide.
- Linkers and their use in preparation of conjugates of oligomeric compounds are provided throughout the art such as in WO 96/11205 and WO 98/52614 and U.S. Pat. Nos. 4,948,882; 5,525,465; 5,541,313; 5,545,730; 5,552,538; 5,580,731; 5,486,603; 5,608,046; 4,587,044; 4,667,025; 5,254,469; 5,245,022; 5,112,963; 5,391,723; 5,510475; 5,512,667; 5,574,142; 5,684,142; 5,770,716; 6,096,875; 6,335,432; and 6,335,437, each of which is incorporated by reference in its entirety.
- Conjugate moieties can be attached to any position of the oligomeric compound.
- conjugate moieties can be attached to the terminus of an oligomeric compound such as a 5' or 3' terminal residue of a nucleic acid.
- Conjugate moieties can also be attached to internal residues of the oligomeric compounds.
- conjugate moieties can be attached to one or both strands.
- a double-stranded oligomeric compound contains a conjugate moiety attached to the sense strand.
- a double-stranded oligomeric compound contains a conjugate moiety attached to the antisense strand.
- conjugate moieties can be attached to heterocyclic base moieties (e.g., purines and pyrimidines), monomeric subunits (e.g., sugar moieties), or monomeric subunit linkages (e.g., phosphodiester linkages) of nucleic acid molecules.
- Conjugation to purines or derivatives thereof can occur at any position including, endocyclic and exocyclic atoms.
- the 2-, 6-, 7-, or 8-positions of a purine base are attached to a conjugate moiety. Conjugation to pyrimidines or derivatives thereof can also occur at any position.
- the 2-, 5-, and 6-positions of a pyrimidine base can be substituted with a conjugate moiety.
- Conjugation to sugar moieties of nucleosides can occur at any carbon atom.
- Example carbon atoms of a sugar moiety that can be attached to a conjugate moiety include the 2', 3', and 5' carbon atoms.
- the 1 ' position can also be attached to a conjugate moiety, such as in an abasic residue.
- Internucleosidic linkages can also bear conjugate moieties.
- the conjugate moiety can be attached directly to the phosphorus atom or to an O, N, or S atom bound to the phosphorus atom.
- the conjugate moiety can be attached to the nitrogen atom of the amine or amide or to an adjacent carbon atom.
- an oligomeric compound is attached to a conjugate moiety by contacting a reactive group (e.g., OH, SH, amine, carboxyl, aldehyde, and the like) on the oligomeric compound with a reactive group on the conjugate moiety.
- a reactive group e.g., OH, SH, amine, carboxyl, aldehyde, and the like
- one reactive group is electrophilic and the other is nucleophilic.
- an electrophilic group can be a carbonyl-containing functionality and a nucleophilic group can be an amine or thiol.
- hybridization or “hybridizing” means the pairing of complementary strands of oligomeric compounds. Pairing typically involves hydrogen bonding, which may be Watson-Crick, Hoogsteen or reversed Hoogsteen hydrogen bonding, between complementary nucleoside or nucleotide bases (nucleobases) of the strands of oligomeric compounds. For example, adenine and thymine are complementary nucleobases that pair through the formation of hydrogen bonds. Hybridization can occur under varying circumstances.
- An oligomeric compound of the invention is specifically hybridizable when binding of the compound to the target nucleic acid interferes with the normal function of the target nucleic acid to cause a loss of activity, and there is a sufficient degree of complementarity to avoid non-specific binding of the oligomeric compound to non-target nucleic acid sequences under conditions in which specific binding is desired, i.e., under physiological conditions in the case of in vivo assays or therapeutic treatment, and under conditions in which assays are performed in the case of in vitro assays.
- stringent hybridization conditions or “stringent conditions” refers to conditions under which an oligomeric compound of the invention will hybridize to its target sequence, but to a minimal number of other sequences. Stringent conditions are sequence-dependent and will vary with different circumstances and in the context of this invention; “stringent conditions” under which oligomeric compounds hybridize to a target sequence are determined by the nature and composition of the oligomeric compounds and the assays in which they are being investigated.
- “Complementary,” as used herein, refers to the capacity for precise pairing of two nucleobases regardless of where the two are located. For example, if a nucleobase at a certain position of an oligomeric compound is capable of hydrogen bonding with a nucleobase at a certain position of a target nucleic acid, then the position of hydrogen bonding between the oligonucleotide and the target nucleic acid is considered to be a complementary position.
- the oligomeric compound and the target nucleic acid are complementary to each other when a sufficient number of complementary positions in each molecule are occupied by nucleobases that can hydrogen bond with each other.
- oligomeric and “complementary” are terms which are used to indicate a sufficient degree of precise pairing or complementarity over a sufficient number of nucleobases such that stable and specific binding occurs between the oligonucleotide and a target nucleic acid.
- sequence of the oligomeric compound need not be 100% complementary to that of its target nucleic acid to be specifically hybridizable.
- an oligomeric compound may hybridize over one or more segments such that intervening or adjacent segments are not involved in the hybridization event (e.g., a loop structure or hairpin structure).
- oligomeric compounds of the present invention comprise at least 70% sequence complementarity to a target region within the target nucleic acid, in further embodiments they comprise 90% sequence complementarity and in yet further embodiments they comprise 95% sequence complementarity to the target region within the target nucleic acid sequence to which they are targeted.
- an oligomeric compound in which 18 of 20 nucleobases of the oligomeric compound are complementary to a target region, and would therefore specifically hybridize would represent 90 percent complementarity.
- the remaining noncomplementary nucleobases may be clustered or interspersed with complementary nucleobases and need not be contiguous to each other or to complementary nucleobases.
- an oligomeric compound which is 18 nucleobases in length having 4 (four) noncomplementary nucleobases which are flanked by two regions of complete complementarity with the target nucleic acid would have 77.8% overall complementarity with the target nucleic acid and would thus fall within the scope of the present invention.
- Percent complementarity of an oligomeric compound with a region of a target nucleic acid can be determined routinely using BLAST programs (basic local alignment search tools) and PowerBLAST programs known in the art (Altschul et al, J. Mol. Biol, 1990, 215, 403-410; Zhang and Madden, Genome Res., 1997, 7, 649-656).
- Targeting an oligomeric compound to a particular nucleic acid molecule, in the context of this invention, can be a multistep process.
- the process can begin with the identification of a target nucleic acid whose function is to be modulated.
- This target nucleic acid can be, for example, a mRNA transcribed from a cellular gene whose expression is associated with a particular disorder or disease state, or a nucleic acid molecule from an infectious agent.
- the targeting process usually also includes determination of at least one target region, segment, or site within the target nucleic acid for the interaction to occur such that the desired effect, e.g., modulation of expression, will result.
- region is defined as a portion of the target nucleic acid having at least one identifiable structure, function, or characteristic.
- segments Within regions of target nucleic acids are segments.
- Segments are defined as smaller or sub-portions of regions within a target nucleic acid.
- Sites as used in the present invention, are defined as positions within a target nucleic acid.
- region, segment, and site can also be used to describe an oligomeric compound of the invention such as, for example, a gapped oligomeric compound having 3 separate segments.
- the translation initiation codon is typically 5'-AUG (in transcribed mRNA molecules; 5'-ATG in the corresponding DNA molecule), the translation initiation codon is also referred to as the "AUG codon,” the “start codon” or the "AUG start codon”.
- a minority of genes have a translation initiation codon having the RNA sequence 5'-GUG, 5'-UUG or 5'-CUG, and 5'-AUA, 5'-ACG and 5'-CUG have been shown to function in vivo.
- translation initiation codon and “start codon” can encompass many codon sequences, even though the initiator amino acid in each instance is typically methionine (in eukaryotes) or formylmethionine (in prokaryotes). It is also known in the art that eukaryotic and prokaryotic genes may have two or more alternative start codons, any one of which may be preferentially utilized for translation initiation in a particular cell type or tissue, or under a particular set of conditions.
- start codon and “translation initiation codon” refer to the codon or codons that are used in vivo to initiate translation of an mRNA transcribed from a gene encoding a nucleic acid target, regardless of the sequence(s) of such codons. It is also known in the art that a translation termination codon (or "stop codon") of a gene may have one of three sequences, i.e., 5'-UAA, 5'-UAG and 5'-UGA (the corresponding DNA sequences are 5'-TAA, 5'-TAG and 5'-TGA, respectively).
- start codon region and “translation initiation codon region” refer to a portion of such an mRNA or gene that encompasses from about 25 to about 50 contiguous nucleotides in either direction (i.e., 5' or 3') from a translation initiation codon.
- stop codon region and “translation termination codon region” refer to a portion of such an mRNA or gene that encompasses from about 25 to about 50 contiguous nucleotides in either direction (i.e., 5' or 3') from a translation termination codon.
- ORF open reading frame
- a suitable region is the intragenic region encompassing the translation initiation or termination codon of the open reading frame (ORF) of a gene.
- target regions include the 5' untranslated region (5'UTR), known in the art to refer to the portion of an mRNA in the 5' direction from the translation initiation codon, and thus including nucleotides between the 5' cap site and the translation initiation codon of an mRNA (or corresponding nucleotides on the gene), and the 3' untranslated region (3'UTR), known in the art to refer to the portion of an mRNA in the 3' direction from the translation termination codon, and thus including nucleotides between the translation termination codon and 3' end of an mRNA (or corresponding nucleotides on the gene).
- 5'UTR 5' untranslated region
- 3'UTR 3' untranslated region
- the 5' cap site of an mRNA comprises an N7 -methylated guanosine residue joined to the 5'-most residue of the mRNA via a 5'-5' triphosphate linkage.
- the 5' cap region of an mRNA is considered to include the 5' cap structure itself as well as the first 50 nucleotides adjacent to the cap site. It is also preferred to target the 5' cap region.
- introns regions which are excised from a transcript before it is translated.
- exons regions which are excised from a transcript before it is translated.
- targeting splice sites i.e., intron-exon junctions or exon-intron junctions, may also be particularly useful in situations where aberrant splicing is implicated in disease, or where an overproduction of a particular splice product is implicated in disease. Aberrant fusion junctions due to rearrangements or deletions are also suitable target sites.
- fusion transcripts produced via the process of splicing of two (or more) mRNAs from different gene sources are known as "fusion transcripts". It is also known that introns can be effectively targeted using oligomeric compounds targeted to, for example, pre-mRNA.
- RNA transcripts can be produced from the same genomic region of DNA. These alternative transcripts are generally known as "variants”. More specifically, “pre-mRNA variants” are transcripts produced from the same genomic DNA that differ from other transcripts produced from the same genomic DNA in either their start or stop position and contain both intronic and exonic sequences.
- pre-mRNA variants Upon excision of one or more exon or intron regions, or portions thereof during splicing, pre-mRNA variants produce smaller "mRNA variants". Consequently, mRNA variants are processed pre-mRNA variants and each unique pre-mRNA variant must always produce a unique mRNA variant as a result of splicing. These mRNA variants are also known as "alternative splice variants”. If no splicing of the pre-mRNA variant occurs then the pre-mRNA variant is identical to the mRNA variant.
- variants can be produced through the use of alternative signals to start or stop transcription and that pre-mRNAs and mRNAs can possess more that one start codon or stop codon.
- Variants that originate from a pre-mRNA or mRNA that use alternative start codons are known as "alternative start variants" of that pre-mRNA or mRNA.
- Those transcripts that use an alternative stop codon are known as “alternative stop variants” of that pre-mRNA or mRNA.
- One specific type of alternative stop variant is the "polyA variant” in which the multiple transcripts produced result from the alternative selection of one of the "polyA stop signals" by the transcription machinery, thereby producing transcripts that terminate at unique polyA sites.
- target nucleic acids are also suitable target nucleic acids.
- the locations on the target nucleic acid to which compounds and compositions of the invention hybridize are herein below referred to as "preferred target segments.”
- preferred target segment is defined as at least an 8-nucleobase portion of a target region to which an active antisense oligomeric compound is targeted. While not wishing to be bound by theory, it is presently believed that these target segments represent portions of the target nucleic acid that are accessible for hybridization.
- oligomeric compounds are chosen which are sufficiently complementary to the target, i.e., hybridize sufficiently well and with sufficient specificity, to give the desired effect.
- a series of nucleic acid duplexes comprising the antisense strand oligomeric compounds of the present invention and its complement sense strand compound can be designed for a specific target or targets.
- the ends of the strands may be modified by the addition of one or more natural or modified nucleobases to form an overhang.
- the sense strand of the duplex is designed and synthesized as the complement of the antisense strand and may also contain modifications or additions to either terminus.
- both strands of the duplex would be complementary over the central nucleobases, each having overhangs at one or both termini.
- the combination of an antisense strand and a sense strand is identified as a complementary pair of siRNA oligonucleotides.
- This complementary pair of siRNA oligonucleotides can include additional nucleotides on either of their 5' or 3' ends. Further they can include other molecules or molecular structures on their 3' or 5' ends such as a phosphate group on the 5' end.
- compounds of the invention include a phosphate group on the 5' end of the antisense strand compound.
- compounds can include a phosphate group on the 5' end of the sense strand compound.
- compounds can include additional nucleotides such as a two base overhang on the 3' end.
- an siRNA complementary pair of oligonucleotides can comprise an antisense strand oligomeric compound having the sequence CGAGAGGCGGACGGGACCG (SEQ ID NO: 19) and having a two-nucleobase overhang of deoxythymidine(dT) and its complement sense strand having the sequence GCTCTCCGCCTGCCCTGGC (SEQ ID NO: 20) also having a two- nucleobase overhang of deoxythymidine.
- oligonucleotides can have approximately the following structure: cgagaggcggacgggaccgTT Antisense Strand I 1 I I 1 I I I I I I I I I I I I I I TTgctctccgcctgccctggc Sense Strand
- a single oligonucleotide having both the antisense portion as a first region in the oligonucleotide and the sense portion as a second region in the oligonucleotide is selected.
- the first and second regions are linked together by either a nucleotide linker (a string of one or more nucleotides that are linked together in a sequence) or by a non-nucleotide linker region or by a combination of both a nucleotide and non-nucleotide structure.
- the oligonucleotide when folded back on itself, would be complementary at least between the first region, the antisense portion, and the second region, the sense portion.
- the oligonucleotide can have a palindrome within it structure wherein the first region, the antisense portion in the 5' to 3' direction, is complementary to the second region, the sense portion in the 3' to 5' direction.
- the invention includes oligonucleotide/protein compositions. Such compositions have both an oligonucleotide component and a protein component.
- the oligonucleotide component includes at least one oligonucleotide, for example, either the antisense or the sense oligonucleotide.
- the oligonucleotide component is an antisense oligonucleotide (e.g., complementary to the target nucleic acid).
- the oligonucleotide component can also include both the antisense and the sense strand oligonucleotides.
- the protein component of the composition comprises at least one protein that forms a portion of the RNA-induced silencing complex, i.e., the RISC complex.
- RISC is a ribonucleoprotein complex that contains an oligonucleotide component and proteins of the Argonaute family of proteins, among others.
- the Argonaute proteins make up a highly conserved family whose members have been implicated in RNA interference and the regulation of related phenomena. Members of this family have been shown to possess the canonical PAZ and Piwi domains, thought to be a region of protein-protein interaction. Other proteins containing these domains have been shown to effect target cleavage, including the RNAse, Dicer.
- the Argonaute family of proteins includes, but depending on species, is not necessary limited to, elF2Cl and elF2C2.
- elF2C2 is also known as human GERp95.
- at least the antisense oligonucleotide strand is bound to the protein component of the RISC complex.
- the complex can also include the sense strand oligonucleotide. Carmell, et al, Genes and Development, 2002, 16, 2733-2742.
- RISC complex can interact with one or more of the translation machinery components.
- Translation machinery components include but are not limited to proteins that effect or aid in the translation of an RNA into protein including the ribosomes or polyribosome complex. Therefore, in further embodiments of the invention, the oligonucleotide component of the invention is associated with a RISC protein component and further associates with the translation machinery of a cell.
- Such interaction with the translation machinery of the cell can include interaction with structural and enzymatic proteins of the translation machinery including, but not limited to, the polyribosome and ribosomal subunits.
- the oligonucleotide of the invention can be associated with cellular factors such as transporters or chaperones. These cellular factors can be protein, lipid or carbohydrate based and can have structural or enzymatic functions that may or may not require the complexation of one or more metal ions.
- the oligonucleotide of the invention itself can have one or more moieties that is bound to the oligonucleotide which facilitates the active or passive transport, localization, or compartmentalization of the oligonucleotide.
- Cellular localization includes, but is not limited to, localization to within the nucleus, the nucleolus, or the cytoplasm.
- Compartmentalization includes, but is not limited to, any directed movement of the oligonucleotides of the invention to a cellular compartment including the nucleus, nucleolus, mitochondrion, or imbedding into a cellular membrane surrounding a compartment or the cell itself.
- the oligonucleotide of the invention is associated with cellular factors that affect gene expression, more specifically those involved in RNA modifications. These modifications include, but are not limited to, posttrascriptional modifications such as methylation. Furthermore, the oligonucleotide of the invention itself can have one or more moieties which are bound to the oligonucleotide and facilitate the posttranscriptional modification. [00107] Forms of oligomeric compound of the invention include single- stranded, double-stranded, circular or hairpin oligomeric compounds that can contain structural elements such as internal or terminal bulges or loops.
- the oligomeric compound is a single-stranded antisense oligonucleotide that binds to a RISC complex, a double stranded antisense/sense pair of oligonucleotide, or a single strand oligonucleotide that includes both an antisense portion and a sense portion.
- a RISC complex a double stranded antisense/sense pair of oligonucleotide
- a single strand oligonucleotide that includes both an antisense portion and a sense portion.
- dsRNA double-stranded RNA
- the compounds and compositions of the invention are used to modulate the expression of a target nucleic acid.
- “Modulators” are those oligomeric compounds that decrease or increase the expression of a nucleic acid molecule encoding a target and which comprise at least an 8-nucleobase portion that is complementary to a preferred target segment.
- the screening method comprises the steps of contacting a preferred target segment of a nucleic acid molecule encoding a target with one or more candidate modulators, and selecting for one or more candidate modulators which decrease or increase the expression of a nucleic acid molecule encoding a target. Once it is shown that the candidate modulator or modulators are capable of modulating (e.g.
- the modulator may then be employed in further investigative studies of the function of a target, or for use as a research, diagnostic, or therapeutic agent in accordance with the present invention.
- oligomeric compound refers to a polymeric structure capable of hybridizing a region of a nucleic acid molecule. This term includes oligonucleotides, oligonucleosides, oligonucleotide analogs, oligonucleotide mimetics and combinations of these. Oligomeric compounds are routinely prepared linearly but can be joined or otherwise prepared to be circular and may also include branching. Oligomeric compounds can be hybridized to form double stranded compounds that can be blunt ended or may include overhangs.
- an oligomeric compound can comprise a plurality of oligomeric residues where the residues contain a monomeric subunit such as a sugar moiety or related group, linkage connecting monomeric subunits, and heterocyclic base moiety.
- an oligomeric compound can comprise a backbone of linked monomeric subunits such as a sugar or surrogate where each linked monomeric subunit is directly or indirectly attached to a heterocyclic base moiety.
- the linkages joining the monomeric subunits, the sugar moieties or surrogates, and the heterocyclic base moieties can be independently modified giving rise to a plurality of motifs for the resulting oligomeric compounds including hemimers, gapmers and chimeras.
- nucleoside is a base-sugar combination.
- the base portion of the nucleoside is normally a heterocyclic base moiety.
- the two most common classes of such heterocyclic bases are purines and pyrimidines.
- Nucleotides are nucleosides that further include a phosphate group covalently linked to the sugar portion of the nucleoside.
- the phosphate group can be linked to either the 2', 3' or 5' hydroxyl moiety of the sugar.
- the phosphate groups covalently link adjacent nucleosides to one another to form a linear polymeric compound.
- this linear polymeric structure can be joined to form a circular structure by hybridization or by formation of a covalent bond, however, open linear structures are generally suitable.
- the phosphate groups are commonly referred to as forming the internucleoside linkages of the oligonucleotide.
- the normal internucleoside linkage of RNA and DNA is a 3' to 5' phosphodiester linkage.
- oligonucleotide refers to an oligomer or polymer of ribonucleic acid (RNA) or deoxyribonucleic acid (DNA). This term includes oligonucleotides composed of naturally-occurring nucleobases, sugars and covalent internucleoside linkages.
- oligonucleotide analog refers to oligonucleotides that have one or more non-naturally occurring portions which function in a similar manner to oligonulceotides. Such non-naturally occurring oligonucleotides can be advantageous with respect to, for example, enhanced cellular uptake, enhanced affinity for nucleic acid target and increased stability in the presence of nucleases.
- oligonucleoside refers to nucleosides that are joined by internucleoside linkages that do not have phosphorus atoms. Internucleoside linkages of this type include short chain alkyl, cycloalkyl, mixed heteroatom alkyl, mixed heteroatom cycloalkyl, one or more short chain heteroatomic and one or more short chain heterocyclic.
- internucleoside linkages include but are not limited to siloxane, sulfide, sulfoxide, sulfone, acetal, formacetal, thioformacetal, methylene formacetal, thioformacetal, alkeneyl, sulfamate; methyleneimino, methylenehydrazino, sulfonate, sulfonamide, amide and others having mixed N, O, S and CH 2 component parts.
- nucleosides of the oligomeric compounds of the invention can have a variety of other modification so long as these other modifications either alone or in combination with other nucleosides enhance one or more of the desired properties described above.
- these nucleotides can have sugar portions that correspond to natiirally-occurring sugars or modified sugars.
- Representative modified sugars include carbocyclic or acyclic sugars, sugars having substituent groups at one or more of their 2', 3 ' or 4' positions and sugars having substituents in place of one or more hydrogen atoms of the sugar. Additional nucleosides amenable to the present invention having altered base moieties and or altered sugar moieties are disclosed in United States Patent 3,687,808 and PCT application PCT/US 89/02323.
- Altered base moieties or altered sugar moieties also include other modifications consistent with the spirit of this invention.
- Such oligonucleotides are best described as being structurally distinguishable from, yet functionally interchangeable with, naturally occurring or synthetic wild type oligonucleotides. All such oligonucleotides are comprehended by this invention so long as they function effectively to mimic the structure of a desired RNA or DNA strand.
- a class of representative base modifications include tricyclic cytosine analog, termed "G clamp" (Lin, et al, J. Am. Chem. Soc. 1998, 120, 8531).
- oligonucleotides of the invention also can include phenoxazine-substituted bases of the type disclosed by Flanagan, et al, Nat. Biotechnol 1999, 17(1), 48-52.
- the oligomeric compounds in accordance with this invention can comprise from about 8 to about 80 nucleobases (i.e. from about 8 to about 80 linked nucleosides).
- nucleobases i.e. from about 8 to about 80 linked nucleosides.
- the invention embodies oligomeric compounds of 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, or 80 nucleobases in length.
- the oligomeric compounds of the invention are N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N
- the oligomeric compounds of the invention are N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N
- oligomeric compounds are oligonucleotides from about 12 to about 50 nucleobases, or from about 15 to about 30 nucleobases.
- RNA oligomers can be synthesized by methods disclosed herein or purchased from various RNA synthesis companies such as for example Dharmacon
- the oligomeric compounds used in accordance with this invention may be conveniently and routinely made through the well-known technique of solid phase synthesis.
- Equipment for such synthesis is sold by several vendors including, for example, Applied Biosystems
- the complementary strands are annealed.
- the single strands are aliquoted and diluted to a concentration of 50 uM.
- 30 uL of each strand is combined with 15uL of a 5X solution of annealing buffer.
- the final concentration of the buffer is 100 mM potassium acetate, 30 mM HEPES-KOH pH 7.4, and 2mM magnesium acetate.
- the final volume is 75 uL.
- This solution is incubated for 1 minute at 90°C and then centrifuged for 15 seconds. The tube is allowed to sit for 1 hour at 37°C at which time the dsRNA duplexes are used in experimentation.
- the final concentration of the dsRNA compound is 20 uM.
- This solution can be stored frozen (-20°C) and freeze- thawed up to 5 times.
- the desired synthetic duplexes are evaluated for their ability to modulate target expression.
- they are treated with synthetic duplexes comprising at least one oligomeric compound of the invention.
- synthetic duplexes comprising at least one oligomeric compound of the invention.
- For cells grown in 96-well plates, wells are washed once with 200 ⁇ L OPTI-MEM-1 reduced-serum medium (Gibco BRL) and then treated with 130 ⁇ L of OPTI-MEM-1 containing 12 ⁇ g/mL LIPOFECTIN (Gibco BRL) and the desired dsRNA compound at a final concentration of 200 nM. After 5 hours of treatment, the medium is replaced with fresh medium. Cells are harvested 16 hours after treatment, at which time RNA is isolated and target reduction measured by RT-PCR.
- nucleoside is a base-sugar combination.
- the base portion of the nucleoside is normally a heterocyclic base.
- the two most common classes of such heterocyclic bases are the purines and the pyrimidines.
- Nucleotides are nucleosides that further include a phosphate group covalently linked to the sugar portion of the nucleoside.
- the phosphate group can be linked to either the T, 3' or 5' hydroxyl moiety of the sugar.
- the phosphate groups covalently link adjacent nucleosides to one another to form a linear polymeric compound.
- linear compounds are generally suitable.
- linear compounds may have internal nucleobase complementarity and may therefore fold in a manner as to produce a fully or partially double-stranded compound.
- the phosphate groups are commonly referred to as forming the internucleoside linkage or in conjunction with the sugar ring the backbone of the oligonucleotide.
- the normal internucleoside linkage that makes up the backbone of RNA and DNA is a 3' to 5' phosphodiester linkage.
- antisense oligomeric compounds useful in this invention include oligonucleotides containing modified e.g. non-naturally occurring internucleoside linkages.
- oligonucleotides having modified internucleoside linkages include internucleoside linkages that retain a phosphorus atom and internucleoside linkages that do not have a phosphorus atom.
- modified oligonucleotides that do not have a phosphorus atom in their internucleoside backbone can also be considered to be oligonucleosides.
- modification of the internucleotide linkage In the C. elegans system, modification of the internucleotide linkage
- oligomeric compounds of the invention can also have one or more modified internucleoside linkages.
- An example phosphorus containing modified internucleoside linkage is the phosphorothioate internucleoside linkage.
- Modified oligonucleotide backbones containing a phosphorus atom therein include, for example, phosphorothioates, chiral phosphorothioates, phosphorodithioates, phosphotriesters, aminoalkylphosphotriesters, methyl and other alkyl phosphonates including 3'-alkylene phosphonates, 5'-alkylene phosphonates and chiral phosphonates, phosphinates, phosphoramidates including 3 '-amino phosphoramidate and aminoalkylphosphoramidates, thionophosphoramidates, thiono- alkylphosphonates, thionoalkylphosphotriesters, selenophosphates and borano- phosphates having normal 3 '-5' linkages, 2 '-5' linked analogs of these, and those having inverted polarity wherein one or more internucleotide linkages is a 3' to 3', 5' to 5
- Oligonucleotides having inverted polarity comprise a single 3' to 3' linkage at the 3'-most internucleotide linkage i.e. a single inverted nucleoside residue which may be abasic (the nucleobase is missing or has a hydroxyl group in place thereof).
- Various salts, mixed salts and free acid forms are also included. [00128] Representative United States patents that teach the preparation of the above phosphorus-containing linkages include, but are not limited to, U.S.
- Modified oligonucleotide backbones that do not include a phosphorus atom therein have backbones that are formed by short chain alkyl or cycloalkyl internucleoside linkages, mixed heteroatom and alkyl or cycloalkyl internucleoside linkages, or one or more short chain heteroatomic or heterocyclic internucleoside linkages.
- morpholino linkages formed in part from the sugar portion of a nucleoside
- siloxane backbones sulfide, sulfoxide and sulfone backbones
- formacetal and thioformacetal backbones methylene formacetal and thioformacetal backbones
- riboacetal backbones alkene containing backbones; sulfamate backbones; methyleneimino and methylenehydrazino backbones; sulfonate and sulfonamide backbones; amide backbones; and others having mixed N, O, S and CH 2 component parts.
- Representative United States patents that teach the preparation of the above oligonucleosides include, but are not limited to, U.S.: 5,034,506; 5,166,315 5,185,444; 5,214,134; 5,216,141; 5,235,033; 5,264,562; 5,264,564; 5,405,938 5,434,257; 5,466,677; 5,470,967; 5,489,677; 5,541,307; 5,561,225; 5,596,086 5,602,240; 5,610,289; 5,602,240; 5,608,046; 5,610,289; 5,618,704; 5,623,070 5,663,312; 5,633,360; 5,677,437; 5,792,608; 5,646,269 and 5,677,439, certain of which are commonly owned with this application, and each of which is herein incorporated by reference.
- oligonucleotide mimetics Another group of oligomeric compounds amenable to the present invention includes oligonucleotide mimetics.
- mimetic as it is applied to oligonucleotides is intended to include oligomeric compounds wherein only the furanose ring or both the furanose ring and the internucleotide linkage are replaced with novel groups, replacement of only the furanose ring is also referred to in the art as being a sugar surrogate.
- the heterocyclic base moiety or a modified heterocyclic base moiety is maintained for hybridization with an appropriate target nucleic acid.
- PNA peptide nucleic acid
- the sugar-backbone of an oligonucleotide is replaced with an amide containing backbone, in particular an aminoethylglycine backbone.
- the nucleobases are retained and are bound directly or indirectly to aza nitrogen atoms of the amide portion of the backbone.
- Representative United States patents that teach the preparation of PNA oligomeric compounds include, but are not limited to, U.S.: 5,539,082; 5,714,331; and 5,719,262, each of which is herein incorporated by reference. Further teaching of PNA oligomeric compounds can be found in Nielsen et al, Science, 1991, 254, 1497-1500.
- PNA peptide nucleic acids
- the backbone in PNA compounds is two or more linked aminoethylglycine units which gives PNA an amide containing backbone.
- the heterocyclic base moieties are bound directly or indirectly to aza nitrogen atoms of the amide portion of the backbone.
- Representative United States patents that teach the preparation of PNA compounds include, but are not limited to, U.S.: 5,539,082; 5,714,331; and 5,719,262, each of which is herein incorporated by reference. Further teaching of PNA compounds can be found in Nielsen et al, Science, 1991, 254, 1497-1500.
- PNA has been modified to incorporate numerous modifications since the basic PNA structure was first prepared.
- the basic structure is shown below:
- Bx is a heterocyclic base moiety
- T 4 is hydrogen, an amino protecting group, -C(O)R 5 , substituted or unsubstituted C 1 -C 10 alkyl, substituted or unsubstituted C 2 -C ⁇ o alkenyl, substituted or unsubstituted C 2 -C 1 o alkynyl, alkylsulfonyl, arylsulfonyl, a chemical functional group, a reporter group, a conjugate group, a D or L ⁇ -amino acid linked via the ⁇ - carboxyl group or optionally through the ⁇ -carboxyl group when the amino acid is aspartic acid or glutamic acid or a peptide derived from D, L or mixed D and L amino acids linked through a carboxyl group, wherein the substituent groups are selected from hydroxyl, amino, alkoxy, carboxy, benzyl, phenyl, nitro, thiol, thioalkoxy, halogen, alkyl, ary
- T 5 is -OH, -N(Z ⁇ )Z 2 , R 5 , D or L ⁇ -amino acid linked via the ⁇ -amino group or optionally through the ⁇ -amino group when the amino acid is lysine or ornithine or a peptide derived from D, L or mixed D and L amino acids linked through an amino group, a chemical functional group, a reporter group or a conjugate group;
- Zi is hydrogen, CpC 6 alkyl, or an amino protecting group
- R 5 is a carbonyl protecting group; and n is from 2 to about 50.
- Another class of oligonucleotide mimetic that has been studied is based on linked morpholino units (morpholino nucleic acid) having heterocyclic bases attached to the morpholino ring.
- a number of linking groups have been reported that link the morpholino monomeric units in a morpholino nucleic acid.
- a class of linking groups have been selected to give a non-ionic oligomeric compound. The non-ionic morpholino-based oligomeric compounds are less likely to have undesired interactions with cellular proteins.
- Morpholino-based oligomeric compounds are non-ionic mimics of oligonucleotides which are less likely to form undesired interactions with cellular proteins (Dwaine A. Braasch and David R. Corey, Biochemistry, 2002, 41(14), 4503-4510). Morpholino-based oligomeric compounds are disclosed in United States Patent 5,034,506, issued July 23, 1991. The morpholino class of oligomeric compounds have been prepared having a variety of different linking groups joining the monomeric subunits. [00136] Morpholino nucleic acids have been prepared having a variety of different linking groups (L 2 ) joining the monomeric subunits. The basic formula is shown below:
- Ti is hydroxyl or a protected hydroxyl
- T 5 is hydrogen or a phosphate or phosphate derivative
- L 2 is a linking group; and n is from 2 to about 50.
- a further class of oligonucleotide mimetic is referred to as cyclohexenyl nucleic acids (CeNA).
- CeNA DMT protected phosphoramidite monomers have been prepared and used for oligomeric compound synthesis following classical phosphoramidite chemistry. Fully modified CeNA oligomeric compounds and oligonucleotides having specific positions modified with CeNA have been prepared and studied (see Wang et al, J. Am. Chem. Soc, 2000, 122, 8595-8602).
- CeNA monomers In general the incorporation of CeNA monomers into a DNA chain increases its stability of a DNA/RNA hybrid. CeNA oligoadenylates formed complexes with RNA and DNA complements with similar stability to the native complexes.
- the study of incorporating CeNA structures into natural nucleic acid structures was shown by NMR and circular dichroism to proceed with easy conformational adaptation. Furthermore the incorporation of CeNA into a sequence targeting RNA was stable to serum and able to activate E. Coli RNase resulting in cleavage of the target RNA strand.
- each Bx is a heterocyclic base moiety
- Tj is hydroxyl or a protected hydroxyl
- T2 is hydroxyl or a protected hydroxyl.
- Another class of oligonucleotide mimetic can be prepared from one or more anhydrohexitol nucleosides (see, Wouters and Herdewijn, Bioorg. Med. Chem. Lett., 1999, 9, 1563-1566) and would have the general formula:
- a further example modification includes Locked Nucleic Acids
- LNAs in which the 2'-hydroxyl group is linked to the 4' carbon atom of the sugar ring thereby forming a 2'-C,4'-C-oxymethylene linkage thereby forming a bicyclic sugar moiety.
- the linkage is a methylene (-CH -) worship group bridging the 2' oxygen atom and the 4' carbon atom wherein n is 1 or 2 (Singh et al, Chem. Commun., 1998, 4, 455-456).
- LNA has been shown to form exceedingly stable LNA:LNA duplexes
- LNA:LNA hybridization was shown to be the most thermally stable nucleic acid type duplex system, and the RNA-mimicking character of LNA was established at the duplex level.
- Tm +15/+11) toward DNA complements.
- Tm +15/+11
- the universality of LNA- mediated hybridization has been stressed by the formation of exceedingly stable LNALNA duplexes.
- the RNA-mimicking of LNA was reflected with regard to the N-type conformational restriction of the monomers and to the secondary structure of the LNA:RNA duplex.
- LNAs also form duplexes with complementary DNA, RNA or LNA with high thermal affinities.
- Circular dichroism (CD) spectra show that duplexes involving fully modified LNA (esp. LNA:RNA) structurally resemble an A-form RNA:RNA duplex.
- Nuclear magnetic resonance (NMR) examination of an LNA:DNA duplex confirmed the 3'-endo conformation of an LNA monomer. Recognition of double-stranded DNA has also been demonstrated suggesting strand invasion by LNA.
- Studies of mismatched sequences show that LNAs obey the Watson-Crick base pairing rules with generally improved selectivity compared to the corresponding unmodified reference strands.
- Novel types of LNA-oligomeric compounds, as well as the LNAs, are useful in a wide range of diagnostic and therapeutic applications. Among these are antisense applications, PCR applications, strand-displacement oligomers, substrates for nucleic acid polymerases and generally as nucleotide based drugs.
- antisense applications PCR applications
- strand-displacement oligomers substrates for nucleic acid polymerases
- substrates for nucleic acid polymerases and generally as nucleotide based drugs.
- nucleotide based drugs generally as nucleotide based drugs.
- Potent and nontoxic antisense oligonucleotides containing LNAs have been described (Wahlestedt et al., Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 5633- 5638.) The authors have demonstrated that LNAs confer several desired properties to antisense agents.
- LNA/DNA copolymers were not degraded readily in blood serum and cell extracts. LNA/DNA copolymers exhibited potent antisense activity in assay systems as disparate as G-protein-coupled receptor signaling in living rat brain and detection of reporter genes in Escherichia coli. Lipofectin-mediated efficient delivery of LNA into living human breast cancer cells has also been accomplished.
- LNA monomers adenine, cytosine, guanine, 5-methyl-cytosine, thymine and uracil, along with their oligomerization, and nucleic acid recognition properties have been described (Koshkin et al, Tetrahedron, 1998, 54, 3607-3630). LNAs and preparation thereof are also described in WO 98/39352 and WO 99/14226.
- oligonucleotide mimetic incorporate a phosphorus group in a backbone the backbone.
- This class of olignucleotide mimetic is reported to have useful physical and biological and pha ⁇ nacological properties in the areas of inhibiting gene expression (antisense oligonucleotides, ribozymes, sense oligonucleotides and triplex-forming oligonucleotides), as probes for the detection of nucleic acids and as auxiliaries for use in molecular biology.
- Oligomeric compounds of the invention may also contain one or more substituted sugar moieties.
- oligomeric compounds comprise a sugar substituent group selected from: OH; F; O-, S-, or N-alkyl; O-, S-, or N- alkenyl; O-, S- or N-alkynyl; or O-alkyl-O-alkyl, wherein the alkyl, alkenyl and alkynyl may be substituted or unsubstituted Ci to Cio alkyl or C 2 to Cio alkenyl and alkynyl.
- Example sugar substituents include O[(CH 2 ) n O] m CH 3 , O(CH 2 ) n OCH 3 , O(CH 2 ) n NH 2 , 0(CH 2 ) n CH 3 , 0(CH 2 ) n ONH 2 , and O(CH 2 ) n ON[(CH 2 ) n CH 3 ] 2 , where n and m are from 1 to about 10.
- oligonucleotides can comprise a sugar substituent group selected from: Ci to Cio lower alkyl, substituted lower alkyl, alkenyl, alkynyl, alkaryl, aralkyl, O-alkaryl or O-aralkyl, SH, SCH 3 , OCN, Cl, Br, CN, CF 3 , OCF 3 , SOCH 3 , SO 2 CH 3 , ONO 2 , NO 2 , N 3 , NH 2 , heterocycloalkyl, heterocycloalkaryl, aminoalkylamino, polyalkylamino, substituted silyl, an RNA cleaving group, a reporter group, an intercalator, a group for improving the pharmacokinetic properties of an oligonucleotide, or a group for improving the pharmacodynamic properties of an oligonucleotide, and other substituents having similar properties.
- a sugar substituent group selected from: Ci to Cio lower alkyl, substituted
- An example modification includes 2'-methoxyethoxy (2'-O- CH 2 CH 2 OCH 3 , also known as 2'-O-(2-methoxyethyl) or 2'-MOE) (Martin et al, Helv. Chim. Acta, 1995, 78, 486-504) i.e., an alkoxyalkoxy group.
- a further example modification includes 2'-dimethylaminooxyethoxy, i.e., a O(CH 2 ) 2 ON(CH ) 2 group, also known as 2'-DMAOE, as described in examples hereinbelow, and 2'- dimethylaminoethoxyethoxy (also known in the art as 2'-O-dimethyl-amino-ethoxy- ethyl or 2'-DMAEOE), i.e., 2'-O-CH 2 -O-CH 2 -N(CH 3 ) 2 .
- 2'-Sugar substituent groups may be in the arabino (up) position or ribo (down) position.
- An example 2'-arabino modification is 2'-F.
- Similar modifications may also be made at other positions on the oligomeric compound, particularly the 3' position of the sugar on the 3' terminal nucleoside or in 2'-5' linked oligonucleotides and the 5' position of 5' terminal nucleotide.
- Oligomeric compounds may also have sugar mimetics such as cyclobutyl moieties in place of the pentofuranosyl sugar.
- Representative United States patents that teach the preparation of such modified sugar structures include, but are not limited to, U.S.: 4,981,957; 5,118,800; 5,319,080; 5,359,044; 5,393,878; 5,446,137; 5,466,786; 5,514,785; 5,519,134; 5,567,811; 5,576,427; 5,591,722; 5,597,909; 5,610,300; 5,627,053; 5,639,873; 5,646,265; 5,658,873; 5,670,633; 5,792,747; and 5,700,920, certain of which are commonly owned with the instant application, and each of which is herein incorporated by reference in its entirety.
- R b is O, S or NH
- R p and R q are each independently hydrogen or Ci-Cio alkyl
- R r is -R x -R y ; each R s , R , R u and R v is, independently, hydrogen, C(O)R w , substituted or unsubstituted Ci-Cio alkyl, substituted or unsubstituted C 2 -C ⁇ o alkenyl, substituted or unsubstituted C 2 -C ⁇ 0 alkynyl, alkylsulfonyl, arylsulfonyl, a chemical functional group or a conjugate group, wherein the substituent groups are selected from hydroxyl, amino, alkoxy, carboxy, benzyl, phenyl, nitro, thiol, thioalkoxy, halogen, alkyl, aryl, alkenyl and alkynyl; or optionally, R u and R v , together form a phthalimido moiety with the nitrogen atom to which they are attached; each R w is, independently,
- R k is hydrogen, a nitrogen protecting group or -R x -R y ;
- R p is hydrogen, a nitrogen protecting group or -R x -R y ;
- R x is a bond or a linking moiety
- R y is a chemical functional group, a conjugate group or a solid support medium; each R m and R réelle is, independently, H, a nitrogen protecting group, substituted or unsubstituted CpCio alkyl, substituted or unsubstituted C 2 -C ⁇ o alkenyl, substituted or unsubstituted C 2 -C ⁇ o alkynyl, wherein the substituent groups are selected from hydroxyl, amino, alkoxy, carboxy, benzyl, phenyl, nitro, thiol, thioalkoxy, halogen, alkyl, aryl, alkenyl, alkynyl; NH + , N(R U )(R V ), guanidino and acyl where said acyl is an acid amide or an ester; or R m and R n , together, are a nitrogen protecting group, are joined in a ring structure that optionally includes an additional heteroatom selected from N and O or are
- Rf, R g and Rh comprise a ring system having from about 4 to about 7 carbon atoms or having from about 3 to about 6 carbon atoms and 1 or 2 heteroatoms wherein said heteroatoms are selected from oxygen, nitrogen and sulfur and wherein said ring system is aliphatic, unsaturated aliphatic, aromatic, or saturated or unsaturated heterocyclic;
- R j is alkyl or haloalkyl having 1 to about 10 carbon atoms, alkenyl having 2 to about 10 carbon atoms, alkynyl having 2 to about 10 carbon atoms, aryl having 6 to about 14 carbon atoms, N(R k )(R m ) OR , halo, SR or CN; m a is 1 to about 10; each mb is, independently, 0 or 1 ; mc is 0 or an integer from 1 to 10; md is an integer from 1 to 10; provided that when mc is 0, md is greater than 1. [00156] Representative substituents groups of Formula I are disclosed in
- Example sugar substituent groups include O[(CH 2 ) n O] m CH 3 ,
- O(CH 2 ) n OCH 3 O(CH 2 )
- O(CH 2 ) offsetNH 2
- O(CH 2 ) n CH 3 O(CH 2 ) n ONH 2
- O(CH 2 ) n ONH 2 O(CH 2 ) n
- Oligomeric compounds may also include nucleobase (often referred to in the art simply as “base” or “heterocyclic base moiety”) modifications or substitutions.
- nucleobases include the purine bases adenine (A) and guanine (G), and the pyrimidine bases thymine (T), cytosine (C) and uracil (U).
- Modified nucleobases also referred herein as heterocyclic base moieties include other synthetic and natural nucleobases such as 5-methylcytosine (5-me-C), 5-hydroxymethyl cytosine, xanthine, hypoxanthine, 2- aminoadenine, 6-methyl and other alkyl derivatives of adenine and guanine, 2-propyl and other alkyl derivatives of adenine and guanine, 2-thiouracil, 2-thiothymine and 2-thiocytosine, 5-halouracil and cytosine, 5-propynyl (-C ⁇ C-CH 3 ) uracil and cytosine and other alkynyl derivatives of pyrimidine bases, 6-azo uracil, cytosine and thymine, 5-uracil (pseudouracil), 4-thiouracil, 8-halo, 8-amino, 8-thiol, 8-thioalkyl, 8-hydroxyl and other 8-sub
- Heterocyclic base moieties may also include those in which the purine or pyrimidine base is replaced with other heterocycles, for example 7-deaza-adenine, 7-deazaguanosine, 2-aminopyridine and 2-pyridone.
- Further nucleobases include those disclosed in United States Patent No. 3,687,808, those disclosed in The Concise Encyclopedia Of Polymer Science And Engineering, pages 858-859, Kroschwitz, J.I., ed.
- nucleobases are particularly useful for increasing the binding affinity of the oligomeric compounds of the invention. These include 5-substituted pyrimidines, 6-azapyrimidines and N-2, N-6 and O-6 substituted purines, including 2-aminopropyladenine, 5-propynyluracil and 5-propynylcytosine.
- 5-methylcytosine substitutions have been shown to increase nucleic acid duplex stability by 0.6-1.2°C (Sanghvi, Y.S., Crooke, S.T. and Lebleu, B., eds., Antisense Research and Applications, CRC Press, Boca Raton, 1993, pp. 276-278) and are examplery base substitutions, such as when combined with 2'-O- methoxyethyl sugar modifications.
- oligomeric compounds are prepared having polycyclic heterocyclic compounds in place of one or more heterocyclic base moieties.
- a number of tricyclic heterocyclic comounds have been previously reported. These compounds are routinely used in antisense applications to increase the binding properties of the modified strand to a target strand. The most studied modifications are targeted to guanosines hence they have been termed G- clamps or cytidine analogs. Many of these polycyclic heterocyclic compounds have the general formula:
- the gain in helical stability does not compromise the specificity of the oligonucleotides.
- the T m data indicate an even greater discrimination between the perfect match and mismatched sequences compared to dC5 me .
- the tethered amino group serves as an additional hydrogen bond donor to interact with the Hoogsteen face, namely the 06, of a complementary guanine thereby forming 4 hydrogen bonds. This means that the increased affinity of G-clamp is mediated by the combination of extended base stacking and additional specific hydrogen bonding.
- oligomeric compounds which are chimeric oligomeric compounds.
- Chimeric oligomeric compounds or “chimeras,” in the context of this invention are oligomeric compounds that contain two or more chemically distinct regions, each made up of at least one monomer unit, i.e., a nucleotide in the case of a nucleic acid based oligomer.
- Chimeric oligomeric compounds typically contain at least one region modified so as to confer increased resistance to nuclease degradation, increased cellular uptake, and/or increased binding affinity for the target nucleic acid.
- An additional region of the oligomeric compound may serve as a substrate for enzymes capable of cleaving RNA:DNA or RNA:RNA hybrids.
- RNase H is a cellular endonuclease which cleaves the RNA strand of an RNA:DNA duplex. Activation of RNase H, therefore, results in cleavage of the RNA target, thereby greatly enhancing the efficiency of inhibition of gene expression.
- RNA target can be routinely detected by gel electrophoresis and, if necessary, associated nucleic acid hybridization techniques known in the art.
- Chimeric oligomeric compounds of the invention may be formed as composite structures of two or more oligonucleotides, oligonucleotide analogs, oligonucleosides and/or oligonucleotide mimetics as described above. Such oligomeric compounds have also been referred to in the art as hybrids hemimers, gapmers or inverted gapmers.
- oligomeric compounds include nucleosides synthetically modified to induce a 3 '-endo sugar conformation.
- a nucleoside can incorporate synthetic modifications of the heterocyclic base, the sugar moiety or both to induce a desired 3 '-endo sugar conformation.
- These modified nucleosides are used to mimic RNA like nucleosides so that particular properties of an oligomeric compound can be enhanced while maintaining the desirable 3 '-endo conformational geometry.
- RNA type duplex A form helix, predominantly 3 '-endo
- RNA interference which is supported in part by the fact that duplexes composed of 2'- deoxy-2'-F-nucleosides appears efficient in triggering RNAi response in the C. elegans system.
- Properties that are enhanced by using more stable 3'-endo nucleosides include but aren't limited to modulation of pharmacokinetic properties through modification of protein binding, protein off-rate, absorption and clearance; modulation of nuclease stability as well as chemical stability; modulation of the binding affinity and specificity of the oligomer (affinity and specificity for enzymes as well as for complementary sequences); and increasing efficacy of RNA cleavage.
- the present invention provides oligomeric triggers of RNAi having one or more nucleosides modified in such a way as to favor a C3'-endo type conformation.
- Nucleoside conformation is influenced by various factors including substitution at the , 3' or 4'-positions of the pentofuranosyl sugar. Electronegative substituents generally prefer the axial positions, while sterically demanding substituents generally prefer the equatorial positions (Principles of Nucleic Acid Structure, Wolfgang Sanger, 1984, Springer-Verlag.) Modification of the 2' position to favor the 3 '-endo conformation can be achieved while maintaining the 2'-OH as a recognition element, as illustrated in Figure 2, below (Gallo et al., Tetrahedron (2001), 57, 5707-5713. Harry-O'kuru et al., J. Org.
- preference for the 3'-endo conformation can be achieved by deletion of the 2'-OH as exemplified by 2'deoxy- 2'F-nucleosides (Kawasaki et al, J. Med. Chem. (1993), 36, 831-841), which adopts the 3'-endo conformation positioning the electronegative fluorine atom in the axial position.
- oligomeric triggers of RNAi response might be composed of one or more nucleosides modified in such a way that conformation is locked into a C3'-endo type conformation, i.e. Locked Nucleic Acid (LNA, Singh et al, Chem. Commun. (1998), 4, 455-456), and ethylene bridged Nucleic Acids (ENA, Morita et al, Bioorganic & Medicinal Chemistry Letters (2002), 12, 73-76.)
- LNA Locked Nucleic Acid
- ENA ethylene bridged Nucleic Acids
- modified nucleosides and their oligomers can be estimated by various methods such as molecular dynamics calculations, nuclear magnetic resonance spectroscopy and CD measurements. Hence, modifications predicted to induce RNA like conformations, A-form duplex geometry in an oligomeric context, are selected for use in the modified oligoncleotides of the present invention.
- the synthesis of numerous of the modified nucleosides amenable to the present invention are known in the art (see for example, Chemistry of Nucleosides and Nucleotides Vol 1-3, ed. Leroy B.
- Nucleosides known to be inhibitors/substrates for RNA dependent RNA polymerases for example HCV NS5B [00176]
- the present invention is directed to oligonucleotides that are prepared having enhanced properties compared to native RNA against nucleic acid targets.
- a target is identified and an oligonucleotide is selected having an effective length and sequence that is complementary to a portion of the target sequence.
- Each nucleoside of the selected sequence is scrutinized for possible enhancing modifications.
- An example modification would be the replacement of one or more RNA nucleosides with nucleosides that have the same 3'-endo conformational geometry.
- the selected sequence can be further divided into regions and the nucleosides of each region evaluated for enhancing modifications that can be the result of a chimeric configuration. Consideration is also given to the 5' and 3'-termini as there are often advantageous modifications that can be made to one or more of the terminal nucleosides.
- the oligomeric compounds of the present invention include at least one 5'-modified phosphate group on a single strand or on at least one 5 '-position of a double stranded sequence or sequences. Further modifications are also considered such as internucleoside linkages, conjugate groups, substitute sugars or bases, substitution of one or more nucleosides with nucleoside mimetics and any other modification that can enhance the selected sequence for its intended target.
- RNA-.RNA duplexes are more stable and have higher melting temperatures (Tm's) than DNA:DNA duplexes (Sanger et al. Principles of Nucleic Acid Structure, 1984, Springer-Verlag; New York, NY.; Lesnik et al.
- RNA duplex RNA duplex
- the increased stability of RNA has been attributed to several structural features, most notably the improved base stacking interactions that result from an A-form geometry (Searle et al. Nucleic Acids Res, 1993, 21, 2051-2056).
- the presence of the hydroxyl in RNA biases the sugar toward a C3' endo pucker, i.e, also designated as Northern pucker, which causes the duplex to favor the A-form geometry.
- the 2' hydroxyl groups of RNA can form a network of water mediated hydrogen bonds that help stabilize the RNA duplex (Egli et al.
- deoxy nucleic acids prefer a C2' endo sugar pucker, i.e, also known as Southern pucker, which is thought to impart a less stable B-form geometry (Sanger, W. (1984) Principles of Nucleic Acid Structure, Springer-Verlag, New York, NY).
- B-form geometry is inclusive of both C2'-endo pucker and O4'-endo pucker. This is consistent with Berger, et. al.
- RNA:RNA duplexes and depending on their sequence may be either more or less stable than DNA:DNA duplexes (Searle et al, Nucleic Acids Res., 1993, 21, 2051- 2056).
- the structure of a hybrid duplex is intermediate between A- and B-form geometries, which may result in poor stacking interactions (Lane et al, Eur. J. Biochem., 1993, 215, 297-306; Fedoroff et al, J. Mol. Biol, 1993, 233, 509-523; Gonzalez et al, Biochemistry, 1995, 34, 4969-4982; Horton et al, J. Mol Biol, 1996, 264, 521-533).
- the stability of the duplex formed between a target RNA and a synthetic sequence is central to therapies such as but not limited to antisense and RNA interference as these mechanisms require the binding of a synthetic oligonucleotide strand to an RNA target strand.
- therapies such as but not limited to antisense and RNA interference as these mechanisms require the binding of a synthetic oligonucleotide strand to an RNA target strand.
- antisense effective inhibition of the mRNA requires that the antisense DNA have a very high binding affinity with the mRNA. Otherwise the desired interaction between the synthetic oligonucleotide strand and target mRNA strand will occur infrequently, resulting in decreased efficacyl
- One routinely used method of modifying the sugar puckering is the substitution of the sugar at the 2'-position with a substituent group that influences the sugar geometry.
- the influence on ring conformation is dependant on the nature of the substituent at the 2'-position.
- a number of different substituents have been studied to determine their sugar puckering effect. For example, 2'-halogens have been studied showing that the 2'-fluoro derivative exhibits the largest population (65%) of the C3'-endo form, and the 2'-iodo exhibits the lowest population (7%).
- the populations of adenosine (2'-OH) versus deoxyadenosine (2'-H) are 36% and 19%, respectively.
- the relative duplex stability can be enhanced by replacement of 2'-OH groups with 2'-F groups thereby increasing the C3'-endo population. It is assumed that the highly polar nature of the 2'-F bond and the extreme preference for C3'-endo puckering may stabilize the stacked conformation in an A-form duplex. Data from UV hypochromicity, circular dichroism, and 1H NMR also indicate that the degree of stacking decreases as the electronegativity of the halo substituent decreases. Furthermore, steric bulk at the 2' -position of the sugar moiety is better accommodated in an A-form duplex than a B-form duplex.
- a 2'-substituent on the 3'-terminus of a dinucleoside monophosphate is thought to exert a number of effects on the stacking conformation: steric repulsion, furanose puckering preference, electrostatic repulsion, hydrophobic attraction, and hydrogen bonding capabilities. These substituent effects are thought to be determined by the molecular size, electronegativity, and hydrophobicity of the substituent. Melting temperatures of complementary strands is also increased with the 2'-substituted adenosine diphosphates. It is not clear whether the 3'-endo preference of the conformation or the presence of the substituent is responsible for the increased binding. However, greater overlap of adjacent bases (stacking) can be achieved with the 3'-endo conformation.
- Oligonucleotides having the 2'-O- methoxyethyl substituent also have been shown to be antisense inhibitors of gene expression with promising features for in vivo use (Martin, P, Helv. Chim. Acta, 1995, 78, 486-504; Altmann et al, Chimia, 1996, 50, 168-176; Altmann et al, Biochem. Soc Trans., 1996, 24, 630-637; and Altmann et al, Nucleosides Nucleotides, 1997, 16, 917-926). Relative to DNA, the oligonucleotides having the 2'-MOE modification displayed improved RNA affinity and higher nuclease resistance.
- Chimeric oligonucleotides having 2'-MOE substituents in the wing nucleosides and an internal region of deoxy-phosphorothioate nucleotides have shown effective reduction in the growth of tumors in animal models at low doses.
- 2'-MOE substituted oligonucleotides have also shown outstanding promise as antisense agents in several disease states.
- One such MOE substituted oligonucleotide is presently being investigated in clinical trials for the treatment of CMV retinitis.
- alkyl means C ⁇ -C ⁇ 2 , preferably Ci-
- C 8 and more preferably C ⁇ -C 6 , straight or (where possible) branched chain aliphatic hydrocarbyl.
- heteroalkyl means C ⁇ -Cj 2 , preferably
- C ⁇ -C 8 and more preferably C ⁇ -C 6 , straight or (where possible) branched chain aliphatic hydrocarbyl containing at least one, and preferably about 1 to about 3, hetero atoms in the chain, including the terminal portion of the chain.
- Preferred heteroatoms include N, O and S.
- cycloalkyl means C 3 -Cj 2 , preferably
- alkenyl means C 2 -C ⁇ 2 , preferably C 2 -
- C 8 and more preferably C 2 -C 6 alkenyl, which may be straight or (where possible) branched hydrocarbyl moiety, which contains at least one carbon-carbon double bond.
- alkynyl means C -C ⁇ , preferably C 2 -
- C 8 and more preferably C 2 -C 6 alkynyl, which may be straight or (where possible) branched hydrocarbyl moiety, which contains at least one carbon-carbon triple bond.
- heterocycloalkyl means a ring moiety containing at least three ring members, at least one of which is carbon, and of which 1, 2 or three ring members are other than carbon.
- the number of carbon atoms varies from 1 to about 12, preferably 1 to about 6, and the total number of ring members varies from three to about 15, preferably from about 3 to about 8.
- Preferred ring heteroatoms are N, O and S.
- Preferred heterocycloalkyl groups include morpholino, thiomorpholino, piperidinyl, piperazinyl, homopiperidinyl, homopiperazinyl, homomorpholino, homothiomorpholino, pyrrolodinyl, tetrahydrooxazolyl, tetrahydroimidazolyl, tetrahydrothiazolyl, tetrahydroisoxazolyl, tetrahydropyrrazolyl, furanyl, pyranyl, and tetrahydroisothiazolyl.
- aryl means any hydrocarbon ring structure containing at least one aryl ring.
- Preferred aryl rings have about 6 to about 20 ring carbons.
- Especially preferred aryl rings include phenyl, napthyl, anthracenyl, and phenanthrenyl.
- hetaryl means a ring moiety containing at least one fully unsaturated ring, the ring consisting of carbon and non- carbon atoms.
- the ring system contains about 1 to about 4 rings.
- the number of carbon atoms varies from 1 to about 12, preferably 1 to about 6, and the total number of ring members varies from three to about 15, preferably from about 3 to about 8.
- Preferred ring heteroatoms are N, O and S.
- Prefened hetaryl moieties include pyrazolyl, thiophenyl, pyridyl, imidazolyl, tetrazolyl, pyridyl, pyrimidinyl, purinyl, quinazolinyl, quinoxalinyl, benzimidazolyl, benzothiophenyl, etc.
- a moiety is defined as a compound moiety, such as hetarylalkyl (hetaryl and alkyl), aralkyl (aryl and alkyl), etc, each of the sub-moieties is as defined herein.
- an electron withdrawing group is a group, such as the cyano or isocyanato group that draws electronic charge away from the carbon to which it is attached.
- Other electron withdrawing groups of note include those whose electronegativities exceed that of carbon, for example halogen, nitro, or phenyl substituted in the ortho- or para-position with one or more cyano, isothiocyanato, nitro or halo groups.
- halogen and halo have their ordinary meanings.
- Preferred halo (halogen) substituents are Cl, Br, and I.
- the aforementioned optional substituents are, unless otherwise herein defined, suitable substituents depending upon desired properties. Included are halogens (Cl, Br, I), alkyl, alkenyl, and alkynyl moieties, NO 2 , NH 3 (substituted and unsubstituted), acid moieties (e.g. -CO 2 H, -OSO 3 H 2 , etc.), heterocycloalkyl moieties, hetaryl moieties, aryl moieties, etc.
- the squiggle ( ⁇ ) indicates a bond to an oxygen or sulfur of the 5'-phosphate.
- Phosphate protecting groups include those described in US Patents
- the compounds and compositions of the invention are used to modulate the expression of a selected protein.
- “Modulators” are those oligomeric compounds and compositions that decrease or increase the expression of a nucleic acid molecule encoding a protein and which comprise at least an 8-nucleobase portion which is complementary to a preferred target segment.
- the screening method comprises the steps of contacting a prefened target segment of a nucleic acid molecule encoding a protein with one or more candidate modulators, and selecting for one or more candidate modulators which decrease or increase the expression of a nucleic acid molecule encoding a protein. Once it is shown that the candidate modulator or modulators are capable of modulating (e.g.
- oligomeric compounds of invention can be used combined with their respective complementary strand oligomeric compound to form stabilized double-stranded (duplexed) oligonucleotides. Double stranded oligonucleotide moieties have been shown to modulate target expression and regulate translation as well as RNA processing via an antisense mechanism.
- double-stranded moieties may be subject to chemical modifications (Fire et al. Nature, 1998, 391, 806-811; Timmons and Fire, Nature 1998, 395, 854; Timmons et al. Gene, 2001, 263, 103- 112; Tabara et al. Science, 1998, 282, 430-431; Montgomery et al, Proc. Natl. Acad. Sci. USA, 1998, 95, 15502-15507; Tuschl et al. Genes Dev., 1999, 13, 3191-3197; Elbashir et al. Nature, 2001, 411, 494-498; Elbashir et al. Genes Dev. 2001, 15, 188- 200 ; Nishikura et al.
- oligomeric compounds of the present invention are used to elucidate relationships that exist between proteins and a disease state, phenotype, or condition.
- These methods include detecting or modulating a target peptide comprising contacting a sample, tissue, cell, or organism with the oligomeric compounds and compositions of the present invention, measuring the nucleic acid or protein level of the target and or a related phenotypic or chemical endpoint at some time after treatment, and optionally comparing the measured value to a non-treated sample or sample treated with a further oligomeric compound of the invention.
- These methods can also be perfonned in parallel or in combination with other experiments to determine the function of unknown genes for the process of target validation or to determine the validity of a particular gene product as a target for treatment or prevention of a disease.
- kits Research Reagents, Diagnostics, and Therapeutics
- the oligomeric compounds and compositions of the present invention can additionally be utilized for diagnostics, therapeutics, prophylaxis and as research reagents and kits. Such uses allows for those of ordinary skill to elucidate the function of particular genes or to distinguish between functions of various members of a biological pathway.
- the oligomeric compounds and compositions of the present invention can be used as tools in differential and/or combinatorial analyses to elucidate expression patterns of a portion or the entire complement of genes expressed within cells and tissues.
- expression patterns within cells or tissues treated with one or more compounds or compositions of the invention are compared to control cells or tissues not treated with the compounds or compositions and the patterns produced are analyzed for differential levels of gene expression as they pertain, for example, to disease association, signaling pathway, cellular localization, expression level, size, structure or function of the genes examined. These analyses can be performed on stimulated or unstimulated cells and in the presence or absence of other compounds that affect expression patterns.
- Examples of methods of gene expression analysis known in the art include DNA anays or microanays (Brazma and Vilo, FEBSLett., 2000, 480, 17-24; Celis, et al, FEBS Lett., 2000, 480, 2-16), SAGE (serial analysis of gene expression)(Madden, et al, Drug Discov. Today, 2000, 5, 415-425), READS (restriction enzyme amplification of digested cDNAs) (Prashar and Weissman, Methods Enzymol, 1999, 303, 258-72), TOGA (total gene expression analysis) (Sutcliffe, et al, Proc. Natl. Acad. Sci.
- the compounds and compositions of the invention are useful for research and diagnostics, because these compounds and compositions hybridize to nucleic acids encoding proteins. Hybridization of the compounds and compositions of the invention with a nucleic acid can be detected by means known in the art. Such means may include conjugation of an enzyme to the compound or composition, radiolabelling or any other suitable detection means. Kits using such detection means for detecting the level of selected proteins in a sample may also be prepared. [00204] The specificity and sensitivity of compounds and compositions can also be harnessed by those of skill in the art for therapeutic uses. Antisense oligomeric compounds have been employed as therapeutic moieties in the treatment of disease states in animals, including humans.
- oligomeric compounds can be useful therapeutic modalities that can be configured to be useful in treatment regimes for the treatment of cells, tissues and animals, especially humans.
- an animal such as a human, suspected of having a disease or disorder that can be treated by modulating the expression of a selected protein is treated by administering the compounds and compositions.
- the methods comprise the step of administering to the animal in need of treatment, a therapeutically effective amount of a protein inhibitor.
- the protein inhibitors of the present invention effectively inhibit the activity of the protein or inhibit the expression of the protein.
- the activity or expression of a protein in an animal is inhibited by about 10%).
- the activity or expression of a protein in an animal is inhibited by about 30%.
- the activity or expression of a protein in an animal is inhibited by 50%) or more.
- the reduction of the expression of a protein can be measured in serum, adipose tissue, liver or any other body fluid, tissue or organ of the animal.
- the cells contained within the fluids, tissues or organs being analyzed contain a nucleic acid molecule encoding a protein and/or the protein itself.
- the compounds and compositions of the invention can be utilized in pharmaceutical compositions by adding an effective amount of the compound or composition to a suitable pharmaceutically acceptable diluent or canier.
- Use of the oligomeric compounds and methods of the invention may also be useful prophylactically.
- compositions of the invention may also be admixed, encapsulated, conjugated or otherwise associated with other molecules, molecule structures or mixtures of compounds, as for example, liposomes, receptor- targeted molecules, oral, rectal, topical or other formulations, for assisting in uptake, distribution and/or absorption.
- the compounds and compositions of the invention encompass any pharmaceutically acceptable salts, esters, or salts of such esters, or any other compound which, upon administration to an animal, including a human, is capable of providing (directly or indirectly) the biologically active metabolite or residue thereof. Accordingly, for example, the disclosure is also drawn to prodrugs and pharmaceutically acceptable salts of the oligomeric compounds of the invention, pharmaceutically acceptable salts of such prodrugs, and other bioequivalents.
- prodrugs and pharmaceutically acceptable salts of the oligomeric compounds of the invention pharmaceutically acceptable salts of such prodrugs, and other bioequivalents.
- prodrag indicates a therapeutic agent that is prepared in an inactive form that is converted to an active form (i.e, drug) within the body or cells thereof by the action of endogenous enzymes or other chemicals and/or conditions.
- prodrug versions of the oligonucleotides of the invention are prepared as SATE [(S-acetyl-2-thioethyl) phosphate] derivatives according to the methods disclosed in WO 93/24510 to Gosselin et al, published December 9, 1993 or in WO 94/26764 and U.S. 5,770,713 to Imbach et al.
- pharmaceutically acceptable salts refers to physiologically and pharmaceutically acceptable salts of the compounds and compositions of the invention: i.e, salts that retain the desired biological activity of the parent compound and do not impart undesired toxicological effects thereto.
- pharmaceutically acceptable salts and their uses are further described in U.S. Patent 6,287,860, which is incorporated herein in its entirety.
- the present invention also includes pharmaceutical compositions and formulations that include the compounds and compositions of the invention.
- the pharmaceutical compositions of the present invention may be administered in a number of ways depending upon whether local or systemic treatment is desired and upon the area to be treated. Administration may be topical (including ophthalmic and to mucous membranes including vaginal and rectal delivery), pulmonary, e.g., by inhalation or insufflation of powders or aerosols, including by nebulizer; intratracheal, intranasal, epidermal and transdermal), oral or parenteral.
- Parenteral administration includes intravenous, intraarterial, subcutaneous, intraperitoneal or intramuscular injection or infusion; or intracranial, e.g., intrathecal or intraventricular, administration.
- Pharmaceutical compositions and formulations for topical administration may include transdermal patches, ointments, lotions, creams, gels, drops, suppositories, sprays, liquids and powders.
- Conventional pharmaceutical caniers, aqueous, powder or oily bases, thickeners and the like may be necessary or desirable.
- Coated condoms, gloves and the like may also be useful.
- the pharmaceutical formulations of the present invention which may conveniently be presented in unit dosage form, may be prepared according to conventional techniques well known in the pharmaceutical industry.
- Such techniques include the step of bringing into association the active ingredients with the pharmaceutical canier(s) or excipient(s).
- the formulations are prepared by unifomily and intimately bringing into association the active ingredients with liquid carriers or finely divided solid carriers or both, and then, if necessary, shaping the product.
- compositions of the present invention may be formulated into any of many possible dosage forms such as, but not limited to, tablets, capsules, gel capsules, liquid syrups, soft gels, suppositories, and enemas.
- the compositions of the present invention may also be formulated as suspensions in aqueous, non-aqueous or mixed media.
- Aqueous suspensions may further contain substances which increase the viscosity of the suspension including, for example, sodium carboxymethylcellulose, sorbitol and/or dextran.
- the suspension may also contain stabilizers.
- compositions of the present invention include, but are not limited to, solutions, emulsions, foams and liposome-containing formulations.
- the pharmaceutical compositions and formulations of the present invention may comprise one or more penetration enhancers, carriers, excipients or other active or inactive ingredients.
- Emulsions are typically heterogenous systems of one liquid dispersed in another in the form of droplets usually exceeding 0.1 ⁇ m in diameter. Emulsions may contain additional components in addition to the dispersed phases, and the active drug that may be present as a solution in either the aqueous phase, oily phase or itself as a separate phase. Microemulsions are included as an embodiment of the present invention. Emulsions and their uses are well known in the art and are further described in U.S. Patent 6,287,860, which is incorporated herein in its entirety. [00217] Formulations of the present invention include liposomal formulations.
- liposome means a vesicle composed of amphiphilic lipids arranged in a spherical bilayer or bilayers. Liposomes are unilamellar or multilamellar vesicles which have a membrane formed from a lipophilic material and an aqueous interior that contains the composition to be delivered. Cationic liposomes are positively charged liposomes which are believed to interact with negatively charged DNA molecules to form a stable complex. Liposomes that are pH-sensitive or negatively-charged are believed to entrap DNA rather than complex with it. Both cationic and noncationic liposomes have been used to deliver DNA to cells.
- Liposomes also include "sterically stabilized" liposomes, a term which, as used herein, refers to liposomes comprising one or more specialized lipids that, when incorporated into liposomes, result in enhanced circulation lifetimes relative to liposomes lacking such specialized lipids.
- sterically stabilized liposomes are those in which part of the vesicle-forming lipid portion of the liposome comprises one or more glycolipids or is derivatized with one or more hydrophilic polymers, such as a polyethylene glycol (PEG) moiety.
- PEG polyethylene glycol
- compositions of the present invention may also include surfactants.
- surfactants used in drug products, formulations and in emulsions is well known in the art. Surfactants and their uses are further described in U.S. Patent 6,287,860, which is incorporated herein in its entirety.
- the present invention employs various penetration enhancers to effect the efficient delivery of nucleic acids, particularly oligonucleotides.
- penetration enhancers also enhance the permeability of lipophilic drugs.
- Penetration enhancers may be classified as belonging to one of five broad categories, i.e., surfactants, fatty acids, bile salts, chelating agents, and non-chelating non-surfactants. Penetration enhancers and their uses are further described in U.S. Patent 6,287,860, which is incorporated herein in its entirety.
- Formulations for topical administration include those in which the oligonucleotides of the invention are in admixture with a topical delivery agent such as lipids, liposomes, fatty acids, fatty acid esters, steroids, chelating agents and surfactants.
- a topical delivery agent such as lipids, liposomes, fatty acids, fatty acid esters, steroids, chelating agents and surfactants.
- Lipids and liposomes include neutral (e.g. dioleoylphosphatidyl DOPE ethanolamine, dimyristoylphosphatidyl choline DMPC, distearolyphosphatidyl choline) negative (e.g.
- dimyristoylphosphatidyl glycerol DMPG dimyristoylphosphatidyl glycerol DMPG
- cationic e.g. dioleoyltetramethylaminopropyl DOTAP and dioleoylphosphatidyl ethanolamine DOTMA.
- compounds and compositions of the invention may be encapsulated within liposomes or may form complexes thereto, in particular to cationic liposomes. Alternatively, they may be complexed to lipids, in particular to cationic lipids. Fatty acids and esters, pharmaceutically acceptable salts thereof, and their uses are further described in U.S. Patent 6,287,860, which is incorporated herein in its entirety. Topical formulations are described in detail in United States patent application 09/315,298 filed on May 20, 1999, which is incorporated herein by reference in its entirety.
- compositions and formulations for oral administration include powders or granules, microparticulates, nanoparticulates, suspensions or solutions in water or non-aqueous media, capsules, gel capsules, sachets, tablets or minitablets. Thickeners, flavoring agents, diluents, emulsifiers, dispersing aids or binders may be desirable.
- Oral formulations are those in which oligonucleotides of the invention are administered in conjunction with one or more penetration enhancers surfactants and chelators.
- Surfactants include fatty acids and/or esters or salts thereof, bile acids and/or salts thereof. Bile acids/salts and fatty acids and their uses are further described in U.S.
- Patent 6,287,860 which is incorporated herein in its entirety.
- penetration enhancers for example, fatty acids/salts in combination with bile acids/salts.
- a example combination is the sodium salt of lauric acid, capric acid and UDCA.
- Further penetration enhancers include polyoxyethylene-9-lauryl ether, polyoxyethylene-20-cetyl ether.
- Compounds and compositions of the invention may be delivered orally, in granular form including sprayed dried particles, or complexed to form micro or nanoparticles. Complexing agents and their uses are further described in U.S. Patent 6,287,860, which is incorporated herein in its entirety.
- compositions and formulations for parenteral, intrathecal or intraventricular administration may include sterile aqueous solutions that may also contain buffers, diluents and other suitable additives such as, but not limited to, penetration enhancers, carrier compounds and other pharmaceutically acceptable carriers or excipients.
- Certain embodiments of the invention provide pharmaceutical compositions containing one or more of the compounds and compositions of the invention and one or more other chemotherapeutic agents that function by a non- antisense mechanism.
- chemotherapeutic agents include but are not limited to cancer chemotherapeutic drags such as daunorubicin, daunomycin, dactinomycin, doxorabicin, epirubicin, idarubicin, esorabicin, bleomycin, mafosfamide, ifosfamide, cytosine arabinoside, bis-chloroethylnitrosurea, busulfan, mitomycin C, actinomycin D, mithramycin, prednisone, hydroxyprogesterone, testosterone, tamoxifen, dacarbazine, procarbazine, hexamethylmelamine, pentamethylmelamine, mitoxantrone, amsacrine, chlorambucil,
- chemotherapeutic agents When used with the oligomeric compounds of the invention, such chemotherapeutic agents may be used individually (e.g., 5-FU and oligonucleotide), sequentially (e.g., 5-FU and oligonucleotide for a period of time followed by MTX and oligonucleotide), or in combination with one or more other such chemotherapeutic agents (e.g., 5-FU, MTX and oligonucleotide, or 5-FU, radiotherapy and oligonucleotide).
- chemotherapeutic agents may be used individually (e.g., 5-FU and oligonucleotide), sequentially (e.g., 5-FU and oligonucleotide for a period of time followed by MTX and oligonucleotide), or in combination with one or more other such chemotherapeutic agents (e.g., 5-FU, MTX and oligonucleotide, or 5-FU, radiotherapy
- Anti-inflammatory drags including but not limited to nonsteroidal anti-inflammatory drugs and corticosteroids, and antiviral drags, including but not limited to ribivirin, vidarabine, acyclovir and ganciclovir, may also be combined in compositions of the invention. Combinations of compounds and compositions of the invention and other drugs are also within the scope of this invention. Two or more combined compounds such as two oligomeric compounds or one oligomeric compound combined with further compounds may be used together or sequentially. [00227] In another related embodiment, compositions of the invention may contain one or more of the compounds and compositions of the invention targeted to a first nucleic acid and one or more additional compounds such as antisense oligomeric compounds targeted to a second nucleic acid target.
- compositions of the invention may contain two or more oligomeric compounds and compositions targeted to different regions of the same nucleic acid target. Two or more combined compounds may be used together or sequentially
- compositions of the invention are believed to be within the skill of those in the art. Dosing is dependent on severity and responsiveness of the disease state to be treated, with the course of treatment lasting from several days to several months, or until a cure is effected or a diminution of the disease state is achieved. Optimal dosing schedules can be calculated from measurements of drag accumulation in the body of the patient. Persons of ordinary skill can easily determine optimum dosages, dosing methodologies and repetition rates. Optimum dosages may vary depending on the relative potency of individual oligonucleotides, and can generally be estimated based on EC 5 os found to be effective in in vitro and in vivo animal models.
- dosage is from 0.01 ug to 100 g per kg of body weight, and may be given once or more daily, weekly, monthly or yearly, or even once every 2 to 20 years. Persons of ordinary skill in the art can easily estimate repetition rates for dosing based on measured residence times and concentrations of the drug in bodily fluids or tissues. Following successful treatment, it may be desirable to have the patient undergo maintenance therapy to prevent the recurrence of the disease state, wherein the oligonucleotide is administered in maintenance doses, ranging from 0.01 ug to 100 g per kg of body weight, once or more daily, to once every 20 years.
- oligonucleotides 180 sec and preceded by the normal capping step. After cleavage from the CPG column and deblocking in concentrated ammonium hydroxide at 55°C (12-16 hr), the oligonucleotides were recovered by precipitating with >3 volumes of ethanol from a 1 M NFL- Ac solution. Phosphinate oligonucleotides are prepared as described in
- Alkyl phosphonate oligonucleotides are prepared as described in U.S.
- Patent 4,469,863 herein incorporated by reference.
- 3 '-Deoxy-3 '-methylene phosphonate oligonucleotides are prepared as described in U.S. Patents 5,610,289 or 5,625,050, herein incorporated by reference.
- Phosphoramidite oligonucleotides are prepared as described in U.S.
- Alkylphosphonothioate oligonucleotides are prepared as described in published PCT applications PCT/US94/00902 and PCT/US93/06976 (published as
- 3'-Deoxy-3'-amino phosphoramidate oligonucleotides are prepared as described in U.S. Patent 5,476,925, herein incorporated by reference.
- Phosphotriester oligonucleotides are prepared as described in U.S. Patent 5,023,243, herein incorporated by reference.
- Formacetal and thioformacetal linked oligonucleosides are prepared as described in U.S. Patents 5,264,562 and 5,264,564, herein incorporated by reference.
- Ethylene oxide linked oligonucleosides are prepared as described in
- RNA synthesis chemistry is based on the selective incorporation of various protecting groups at strategic intermediary reactions.
- a useful class of protecting groups includes silyl ethers.
- bulky silyl ethers are used to protect the 5 '-hydroxyl in combination with an acid-labile orthoester protecting group on the 2 '-hydroxyl.
- This set of protecting groups is then used with standard solid-phase synthesis technology. It is important to lastly remove the acid labile orthoester protecting group after all other synthetic steps.
- the early use of the silyl protecting groups during synthesis ensures facile removal when desired, without undesired deprotection of 2' hydroxyl.
- RNA oligonucleotides were synthesized. [00245] RNA oligonucleotides are synthesized in a stepwise fashion. Each nucleotide is added sequentially (3'- to 5 '-direction) to a solid support-bound oligonucleotide. The first nucleoside at the 3 '-end of the chain is covalently attached to a solid support.
- the nucleotide precursor, a ribonucleoside phosphoramidite, and activator are added, coupling the second base onto the 5 '-end of the first nucleoside.
- the support is washed and any unreacted 5 '-hydroxyl groups are capped with acetic anhydride to yield 5 '-acetyl moieties.
- the linkage is then oxidized to the more stable and ultimately desired P(V) linkage.
- the 5 '-silyl group is cleaved with fluoride. The cycle is repeated for each subsequent nucleotide.
- the methyl protecting groups on the phosphates are cleaved in 30 minutes utilizing 1 M disodium-2-carbamoyl-2-cyanoethylene-l,l- dithiolate trihydrate (S 2 Na 2 ) in DMF.
- the deprotection solution is washed from the solid support-bound oligonucleotide using water.
- the support is then treated with 40% methylamine in water for 10 minutes at 55 °C. This releases the RNA oligonucleotides into solution, deprotects the exocyclic amines, and modifies the T- groups.
- the oligonucleotides can be analyzed by anion exchange HPLC at this stage.
- the 2 '-orthoester groups are the last protecting groups to be removed.
- the ethylene glycol monoacetate orthoester protecting group developed by Dharmacon Research, Inc. (Lafayette, CO), is one example of a useful orthoester protecting group which, has the following important properties. It is stable to the conditions of nucleoside phosphoramidite synthesis and oligonucleotide synthesis. However, after oligonucleotide synthesis the oligonucleotide is treated with methylamine which not only cleaves the oligonucleotide from the solid support but also removes the acetyl groups from the orthoesters. The resulting 2-ethyl-hydroxyl substituents on the orthoester are less electron withdrawing than the acetylated precursor.
- the modified orthoester becomes more labile to acid-catalyzed hydrolysis. Specifically, the rate of cleavage is approximately 10 times faster after the acetyl groups are removed. Therefore, this orthoester possesses sufficient stability in order to be compatible with oligonucleotide synthesis and yet, when subsequently modified, permits deprotection to be canied out under relatively mild aqueous conditions compatible with the final RNA oligonucleotide product. [00248] Additionally, methods of RNA synthesis are well known in the art
- Chimeric oligonucleotides, oligonucleosides or mixed oligonucleotides/oligonucleosides of the invention can be of several different types. These include a first type wherein the "gap" segment of linked nucleosides is positioned between 5' and 3' "wing" segments of linked nucleosides and a second "open end” type wherein the "gap” segment is located at either the 3' or the 5' terminus of the oligomeric compound. Oligonucleotides of the first type are also known in the art as “gapmers” or gapped oligonucleotides. Oligonucleotides of the second type are also known in the art as “hemimers" or "wingmers”.
- Oligonucleotides are synthesized using the automated synthesizer and 2'-deoxy-5'-dimethoxytrityl-3'-O- phosphoramidite for the DNA portion and 5'-dimethoxytrityl-2'-O-methyl-3'-O- phosphoramidite for 5' and 3' wings.
- the standard synthesis cycle is modified by incorporating coupling steps with increased reaction times for the 5'-dimethoxytrityl- 2'-O-methyl-3'-O-phosphoramidite.
- the fully protected oligonucleotide is cleaved from the support and deprotected in concentrated ammonia (NH-.OH) for 12-16 hr at 55°C.
- the deprotected oligo is then recovered by an appropriate method (precipitation, column chromatography, volume reduced in vacuo and analyzed spetrophotometrically for yield and for purity by capillary electrophoresis and by mass spectrometry. [2'-O-(2-Methoxyethyl)] ⁇ [2'-deoxy]-[2 , -O-(Methoxyethyl)] Chimeric
- [00251] [2'-O-(2-methoxyethyl)] ⁇ [2'-deoxy]-[-2'-O-(methoxyethyl)] chimeric phosphorothioate oligonucleotides were prepared as per the procedure above for the 2'-O-methyl chimeric oligonucleotide, with the substitution of 2'-O-(methoxyethyl) amidites for the 2'-O-methyl amidites,
- [2'-O-(methoxyethyl) phosphodiester] chimeric oligonucleotides are prepared as per the above procedure for the 2'-O-methyl chimeric oligonucleotide with the substitution of 2'-O-(methoxyethyl) amidites forme 2'-O-methyl amidites, oxidation with iodine to generate the phosphodiester internucleotide linkages within the wing portions of the chimeric structures and sulfurization utilizing 3,H-1,2 benzodithiole- 3 -one 1,1 dioxide (Beaucage Reagent) to generate the phosphorothioate internucleotide linkages for the center gap.
- a series of nucleic acid duplexes comprising the antisense oligomeric compounds of the present invention and their complements can be designed to target a target.
- the ends of the strands may be modified by the addition of one or more natural or modified nucleobases to form an overhang.
- the sense strand of the dsRNA is then designed and synthesized as the complement of the antisense strand and may also contain modifications or additions to either terminus.
- both strands of the dsRNA duplex would be complementary over the central nucleobases, each having overhangs at one or both termini.
- a duplex comprising an antisense strand having the sequence CGAGAGGCGGACGGGACCG (SEQ ID NO: 19) and having a two- nucleobase overhang of deoxythymidine(dT) would have the following structure: cgagaggcggacgggaccgTT Antisense Strand I I I I I I I I I I I I I I I TTgctctccgcctgccctggc Complement Strand
- RNA strands of the duplex can be synthesized by methods disclosed herein or purchased from Dharmacon Research Inc., (Lafayette, CO). Once synthesized, the complementary strands are annealed. The single strands are aliquoted and diluted to a concentration of 50 uM. Once diluted, 30 uL of each strand is combined with 15uL of a 5X solution of annealing buffer. The final concentration of said buffer is 100 mM potassium acetate, 30 mM HEPES-KOH pH 7.4, and 2mM magnesium acetate. The final volume is 75 uL. This solution is incubated for 1 minute at 90°C and then centrifuged for 15 seconds.
- the tube is allowed to sit for 1 hour at 37°C at which time the dsRNA duplexes are used in experimentation.
- the final concentration of the dsRNA duplex is 20 uM.
- This solution can be stored frozen (-20°C) and freeze-thawed up to 5 times.
- oligonucleotides or oligonucleosides are recovered by precipitation out of 1 M NH OAc with >3 volumes of ethanol.
- Synthesized oligonucleotides were analyzed by electiospray mass spectroscopy (molecular weight determination) and by capillary gel electrophoresis and judged to be at least 70% full length material.
- the relative amounts of phosphorothioate and phosphodiester linkages obtained in the synthesis was determined by the ratio of conect molecular weight relative to the -16 amu product (+/-32 +/-48).
- Oligonucleotides were synthesized via solid phase P(III) phosphoramidite chemistry on an automated synthesizer capable of assembling 96 sequences simultaneously in a 96-well format.
- Phosphodiester internucleotide linkages were afforded by oxidation with aqueous iodine.
- Phosphorothioate internucleotide linkages were generated by sulfurization utilizing 3,H-1,2 benzodithiole-3-one 1,1 dioxide (Beaucage Reagent) in anhydrous acetonitrile.
- Standard base-protected beta-cyanoethyl-diiso-propyl phosphoramidites were purchased from commercial vendors (e.g.
- Non-standard nucleosides are synthesized as per standard or patented methods. They are utilized as base protected beta- cyanoethyldiisopropyl phosphoramidites.
- Oligonucleotides were cleaved from support and deprotected with concentrated NH 4 OH at elevated temperature (55-60°C) for 12-16 hours and the released product then dried in vacuo. The dried product was then re-suspended in sterile water to afford a master plate from which all analytical and test plate samples are then diluted utilizing robotic pipettors.
- oligonucleotide concentration was assessed by dilution of samples and UN absorption spectroscopy.
- the full-length integrity of the individual products was evaluated by capillary electrophoresis (CE) in either the 96- well format (Beckman P/ACETM MDQ) or, for individually prepared samples, on a commercial CE apparatus (e.g., Beckman P/ACETM 5000, ABI 270).
- Base and backbone composition was confirmed by mass analysis of the oligomeric compounds utilizing electrospray-mass spectroscopy. All assay test plates were diluted from the master plate using single and multi-channel robotic pipettors. Plates were judged to be acceptable if at least 85%) of the oligomeric compounds on the plate were at least 85% full length.
- Oligomeric compounds can be attached to conjugate moieties by a variety of methods known in the art. Numerous methods involve the incorporation of a modified residue into the oligomeric compound during solid-phase synthesis.
- the modified residue can include a functionality or linker group that would allow attachment of the conjugate moiety after completion of the coupling reactions.
- the modification is protected by an appropriate protecting group that can be selectively removed after the final coupling step.
- Oligonucleotides that are synthesized by routine solid-phase methods in the 3' to 5' direction are readily modified on the 5' end prior to deprotection and cleavage from the solid support.
- an electrophilic haloacetyl linker can be incorporated onto the 5' end of a solid-support bound oligonucleotide under reaction conditions similar to that for coupling reactions by treating the terminal 5' OH group with 2-cyanoethyl-((N-chloroacetyl)-6-aminohexyl)-(N,N-diisopropyl)- phosphoramidite (dissolved in, for example, MeCN) as described in U.S. Pat. No. 6,335,437.
- the resulting 5 '-haloacetyl modified oligonucleotide can be reacted with any amino or mercapto conjugate moiety prior to deprotection and cleavage from solid support.
- the 5 '-haloacetyl modified oligonucleotide can be reacted with thiocholesterol under reaction conditions provided in U.S. Pat. No. 6,445,437 to yield a 5' thiocholesterol conjugate.
- the conjugated oligonucleotide can then be treated with deprotecting and/or cleaving reagents for purification and isolation.
- An oligonucleotide possessing an amino group at its 5'-terminus is prepared using a DNA synthesizer and then is reacted with an active ester derivative of the conjugate moiety (e.g., cholic acid).
- Active ester derivatives are well known to those skilled in the art. Representative active esters include N-hydrosuccinimide esters, tetrafluorophenolic esters, pentafluorophenolic esters and pentachlorophenolic esters.
- active esters include N-hydrosuccinimide esters, tetrafluorophenolic esters, pentafluorophenolic esters and pentachlorophenolic esters.
- cholic acid the reaction of the amino group and the active ester produces an oligonucleotide in which cholic acid is attached to the 5 '-position through a linking group.
- the amino group at the 5'-tenninus can be prepared conveniently utilizing the commercially avaiable 5
- Cholic acid can also be attached to a 3 '-terminal amino group by reacting a 3 '-amino modified controlled pore glass (sold by Clontech Laboratories Inc., Palo Alto, Calif), with a cholic acid active ester.
- a 3 '-amino modified controlled pore glass sold by Clontech Laboratories Inc., Palo Alto, Calif
- Cholic acid can also be attached to both ends of a linked nucleoside sequence by reacting a 3',5'-diamino sequence with the cholic acid active ester.
- the required ohgonucleoside sequence is synthesized utilizing the 3'-Amino-Modifier and the 5'-Amino-Modifier C6 (or Aminolink-2) reagents noted above or by utilizing the above-noted 3'-amino modified controlled pore glass reagent in combination with the 5'-Amino-Modifier C2 (or Aminolink-2) reagents.
- An ohgonucleoside sequence bearing an aminolmker at the 2'-position of one or more selected nucleosides is prepared using a suitably functionalized nucleotide such as, for example, 5'-dimethoxytrityl-2'-O-( ⁇ -phthalimidylamino- pentyl)-2'-deoxyadeno sine-3'-N,N-diisopropyl-cyanoethoxy phosphoramidite. (See, e.g., Manoharan, et al. Tetrahedron Letters, 1991, 34, 7171).
- the nucleotide or nucleotides are attached to cholic acid or another substituent using an active ester or a thioiso-cyanate thereof.
- Conjugation (linking) of reporter enzymes, peptides, and proteins to oligomeric compounds is achieved by conjugation of the enzyme, peptide or protein to an amino linking group on the nucleoside.
- Conjugation of the peptide or protein can be carried out using EDC/sulfo-NHS (i.e, 1 -ethyl-3 (3 -dimethylaminopropyl-carbodiimide/N-hydroxysulfosuccinimide) to conjugate the carboxyl end of the reporter enzyme, peptide, or protein with an amino function, such as at a terminus or linking group, on the oligomeric compound.
- EDC/sulfo-NHS can be used to conjugate a carboxyl group of an aspartic or glutamic acid residue in the reporter enzyme, peptide or protein to the amino function in an oligomeric compound.
- conjugation can be carried out by linkage of the reporter enzyme, peptide or protein to the nucleoside sequence via a heterobifunctional linker such as m-maleimidobenzoyl-N-hydroxysulfosuccinimide ester (MBS) or succinimidyl 4-(N-maleimidomethyl)cyclohexane-l-carboxylate (SMCC) to link a thiol function on the reporter enzyme, peptide or protein to the amino function of the linking group on nucleoside sequence.
- MBS m-maleimidobenzoyl-N-hydroxysulfosuccinimide ester
- SMCC succinimidyl 4-(N-maleimidomethyl)cyclohexane-l-carboxylate
- conjugation can be carried out by conjugation of the peptide or protein to the sequence using a homobifunctional linker such as disuccinimidyl suberate (DSS) to link an amino function on the peptide or protein to the amino group of a linker on the oligomeric compound.
- a homobifunctional linker such as disuccinimidyl suberate (DSS)
- DSS disuccinimidyl suberate
- an oligonucleoside-succinimidyl conjugate is formed by reaction of the amino group of the linker on the oligomeric compound with a disuccinimidyl suberate linker.
- the disuccinimidyl suberate linker couples with the amine linker on the sequence to extend the size of the linker.
- the extended linker is then reacted with amine groups such as, for example, the amine of lysine or other available N-terminus amines, on reporter enzymes, peptides and proteins.
- an amino function can be introduced into the oligomeric compound by any known method.
- Conjugate moieties include proteins, peptides, cholic acid, biotin, fluoroscein, retinoic acid, folic acid, pyridoxal, tocopherol, phenanthroline, pyrene, acridine, porphyrin, hybrid/intercalator, bipyridine, aryl azide photocrosslinkers, imidazole, EDTA, cholesterol, digoxigenin, and others.
- RNA molecules conjugated with hexaethyleneglycol (18S).
- AS sense and antisense 21-mer oligonucleotides targeting cRaf were obatined from Dharmacon, Inc. (AS is antisense, Fl is fluoroscein, and Bi is biotin) and were annealed according to instractions provided by the supplier.
- cRaf mRNA expression levels (% UTC) in HUNECs at 18 hours post treatment were determined by RT-PCR for duplexes containing the above oligonucleotides A-F.
- Table III shows that duplexes containing at least one 18s conjugate moiety were effective in reducing cRaf mR ⁇ A levels. It is also interesting to note that the sense-only conjugate (B/C) was just as active as the duplex having no modifications (A/C) whereas duplexes containing a conjugate group on the antisense strand tended to be less active.
- the effect of conjugation of an oligonucleotide with respect to cellular uptake can be determined by the inhibition of ICAM-1 utilizing the method of Chiang, et al, J. Biol. Chem. 1991, 266, 18162. Utilizing this method, human lung epithelial carcinoma cells (A549 cells) are grown to confluence in 96 well plates. Medium is removed and the cells are washed with folic acid free medium three times.
- ICAM-1 specific antisense phosphorothioate oligonucleotide having the sequence 5'- TGG GAG CCA TAG CGA GGC-3' (SEQ ID NO:23), either free or conjugated, is added to the incubation medium.
- This oligonucleotide is an 18 base phosphorothioate oligonucleotide that targets the AUG translation initiation codon of the human ICAM-1 mRNA.
- the oligonucleotides are incubated with the A549 cells for 24 hours then ICAM-1 is induced by adding 2.5 ng/ml tumor necrosis factor- ⁇ to the medium. Cells are incubated an additional 15 hours in the presence of tumor necrosis factor- ⁇ and oligonucleotide.
- ICAM-1 expression is determined by a specific ELISA as described by Chiang, et al.
- oligomeric compounds on target nucleic acid expression can be tested in any of a variety of cell types provided that the target nucleic acid is present at measurable levels. This can be routinely determined using, for example, PCR or Northern blot analysis. The following cell types are provided for illustrative purposes, but other cell types can be routinely used, provided that the target is expressed in the cell type chosen. This can be readily determined by methods routine in the art, for example Northern blot analysis, ribonuclease protection assays, or RT- PCR. T-24 cells:
- the human transitional cell bladder carcinoma cell line T-24 was obtained from the American Type Culture Collection (ATCC) (Manassas, VA). T-24 cells were routinely cultured in complete McCoy's 5A basal media (Invitrogen Corporation, Carlsbad, CA) supplemented with 10%) fetal calf serum (Invitrogen Corporation, Carlsbad, CA), penicillin 100 units per mL, and streptomycin 100 micrograms per mL (Invitrogen Corporation, Carlsbad, CA). Cells were routinely passaged by trypsinization and dilution when they reached 90% confluence. Cells were seeded into 96-well plates (Falcon-Primaria #353872) at a density of 7000 cells/well for use in RT-PCR analysis.
- ATCC American Type Culture Collection
- cells may be seeded onto 100 mm or other standard tissue culture plates and treated similarly, using appropriate volumes of medium and oligonucleotide.
- A549 cells A549 cells:
- the human lung carcinoma cell line A549 was obtained from the
- A549 cells were routinely cultured in DMEM basal media (Invitrogen Corporation, Carlsbad, CA) supplemented with 10% fetal calf serum (Invitrogen Corporation, Carlsbad, CA), penicillin 100 units per mL, and streptomycin 100 micrograms per mL (Invitrogen Corporation, Carlsbad, CA). Cells were routinely passaged by trypsinization and dilution when they reached 90% confluence.
- DMEM basal media Invitrogen Corporation, Carlsbad, CA
- penicillin 100 units per mL Invitrogen Corporation, Carlsbad, CA
- streptomycin 100 micrograms per mL
- HEK cells [00283] Human embryonic keratinocytes (HEK) were obtained from the Clonetics Corporation (Walkersville, MD). HEKs were routinely maintained in Keratinocyte Growth Medium (Clonetics Corporation, Walkersville, MD) formulated as recommended by the supplier. Cells were routinely maintained for up to 10 passages as recommended by the supplier. Treatment with antisense oligomeric compounds:
- the concentration of oligonucleotide used varies from cell line to cell line.
- the cells are treated with a positive control oligonucleotide at a range of concentrations.
- the positive control oligonucleotide is selected from either ISIS 13920 (TCCGTCATCGCTCCTCAGGG, SEQ ID NO: 24) which is targeted to human H-ras, or ISIS 18078, (GTGCGCGCGAGCCCGAAATC, SEQ ID NO: 25) which is targeted to human Jun-N-terminal kinase-2 (JNK2).
- Both controls are 2'-O-methoxyethyl gapmers (2'-O-methoxyethyls shown in bold) with a phosphorothioate backbone.
- the positive control oligonucleotide is ISIS 15770, ATGCATTCTGCCCCCAAGGA, SEQ ID NO: 26, a 2'-O-methoxyethyl gapmer (2'-O-methoxyethyls shown in bold) with a phosphorothioate backbone which is targeted to both mouse and rat c-raf.
- the concentration of positive control oligonucleotide that results in 80% inhibition of c- H-ras (for ISIS 13920), JNK2 (for ISIS 18078) or c-raf (for ISIS 15770) mRNA is then utilized as the screening concentration for new oligonucleotides in subsequent experiments for that cell line. If 80% inhibition is not achieved, the lowest concentration of positive control oligonucleotide that results in 60% inhibition of c- H-ras, JNK2 or c-raf mRNA is then utilized as the oligonucleotide screening concentration in subsequent experiments for that cell line. If 60% inhibition is not achieved, that particular cell line is deemed as unsuitable for oligonucleotide transfection experiments.
- concentrations of antisense oligonucleotides used herein are from 50 nM to 300 nM.
- RNA analysis can be performed on total cellular RNA or poly(A)+ mRNA.
- the prefened method of RNA analysis of the present invention is the use of total cellular RNA as described in other examples herein. Methods of RNA isolation are well known in the art. Northern blot analysis is also routine in the art.
- PCR Real-time quantitative
- ABI PRISMTM 7600, 7700, or 7900 Sequence Detection System available from PE- Applied Biosystems, Foster City, CA and used according to manufacturer's instructions.
- Protein levels of a target can be quantitated in a variety of ways well known in the art, such as immunoprecipitation, Western blot analysis (immunoblorting), enzyme-linked immunosorbent assay (ELISA) or fluorescence- activated cell sorting (FACS).
- Antibodies directed to a target can be identified and obtained from a variety of sources, such as the MSRS catalog of antibodies (Aerie Corporation, Birmingham, MI), or can be prepared via conventional monoclonal or polyclonal antibody generation methods well known in the art.
- the oligomeric compounds are further investigated in one or more phenotypic assays, each having measurable endpoints predictive of efficacy in the treatment of a particular disease state or condition.
- Phenotypic assays, kits and reagents for their use are well known to those skilled in the art and are herein used to investigate the role and/or association of a target in health and disease.
- Representative phenotypic assays which can be purchased from any one of several commercial vendors, include those for determining cell viability, cytotoxicity, proliferation or cell survival (Molecular Probes, Eugene, OR; PerkinElmer, Boston, MA), protein-based assays including enzymatic assays (Panvera, LLC, Madison, WI; BD Biosciences, Franklin Lakes, NJ; Oncogene Research Products, San Diego, CA), cell regulation, signal transduction, inflammation, oxidative processes and apoptosis (Assay Designs Inc., Ann Arbor, MI), triglyceride accumulation (Sigma-Aldrich, St.
- cells determined to be appropriate for a particular phenotypic assay i.e, MCF-7 cells selected for breast cancer studies; adipocytes for obesity studies
- a target inhibitors identified from the in vitro studies as well as control compounds at optimal concentiations which are determined by the methods described above.
- treated and untreated cells are analyzed by one or more methods specific for the assay to determine phenotypic outcomes and endpoints.
- Phenotypic endpoints include changes in cell morphology over time or treatment dose as well as changes in levels of cellular components such as proteins, lipids, nucleic acids, hormones, saccharides or metals. Measurements of cellular status which include pH, stage of the cell cycle, intake or excretion of biological indicators by the cell, are also endpoints of interest.
- Analysis of the geneotype of the cell (measurement of the expression of one or more of the genes of the cell) after treatment is also used as an indicator of the efficacy or potency of the target inhibitors. Hallmark genes, or those genes suspected to be associated with a specific disease state, condition, or phenotype, are measured in both treated and untreated cells. In vivo studies
- the individual subjects of the in vivo studies described herein are warm-blooded vertebrate animals, which includes humans.
- the clinical trial is subjected to rigorous controls to ensure that individuals are not unnecessarily put at risk and that they are fully informed about their role in the study.
- Volunteers receive either the a target inhibitor or placebo for eight week period with biological parameters associated with the indicated disease state or condition being measured at the beginning (baseline measurements before any treatment), end (after the final treatment), and at regular intervals during the study period.
- biological parameters associated with the indicated disease state or condition include the levels of nucleic acid molecules encoding a target or a target protein levels in body fluids, tissues or organs compared to pre- treatment levels.
- Other measurements include, but are not limited to, indices of the disease state or condition being treated, body weight, blood pressure, serum titers of pharmacologic indicators of disease or toxicity as well as ADME (absorption, distribution, metabolism and excretion) measurements.
- Information recorded for each patient includes age (years), gender, height (cm), family history of disease state or condition (yes/no), motivation rating (some/moderate/great) and number and type of previous treatment regimens for the indicated disease or condition.
- Volunteers taking part in this study are healthy adults (age 18 to 65 years) and roughly an equal number of males and females participate in the study. Volunteers with certain characteristics are equally distributed for placebo and a target inhibitor treatment. In general, the volunteers treated with placebo have little or no response to treatment, whereas the volunteers treated with the target inhibitor show positive trends in their disease state or condition index at the conclusion of the study.
- the repetitive pipetting and elution steps may be automated using a
- Quantitation of a target mRNA levels was accomplished by real-time quantitative PCR using the ABI PRISMTM 7600, 7700, or 7900 Sequence Detection System (PE-Applied Biosystems, Foster City, CA) according to manufacturer's instractions.
- ABI PRISMTM 7600, 7700, or 7900 Sequence Detection System PE-Applied Biosystems, Foster City, CA
- This is a closed-tube, non-gel-based, fluorescence detection system which allows high-throughput quantitation of polymerase chain reaction (PCR) products in real-time.
- PCR polymerase chain reaction
- oligonucleotide probe that anneals specifically between the forward and reverse PCR primers, and contains two fluorescent dyes.
- a reporter dye e.g., FAM or JOE, obtained from either PE-Applied Biosystems, Foster City, CA, Operon Technologies Inc., Alameda, CA or Integrated DNA Technologies Inc., Coralville, IA
- a quencher dye e.g., TAMRA, obtained from either PE-Applied Biosystems, Foster City, CA, Operon Technologies Inc., Alameda, CA or Integrated DNA Technologies Inc., Coralville, IA
- reporter dye emission is quenched by the proximity of the 3' quencher dye.
- annealing of the probe to the target sequence creates a substrate that can be cleaved by the 5'- exonuclease activity of Taq polymerase.
- cleavage of the probe by Taq polymerase releases the reporter dye from the remainder of the probe (and hence from the quencher moiety) and a sequence-specific fluorescent signal is generated.
- additional reporter dye molecules are cleaved from their respective probes, and the fluorescence intensity is monitored at regular intervals by laser optics built into the ABI PRISMTM Sequence Detection System.
- a series of parallel reactions containing serial dilutions of mRNA from untreated control samples generates a standard curve that is used to quantitate the percent inhibition after antisense oligonucleotide treatment of test samples.
- primer-probe sets specific to the target gene being measured are evaluated for their ability to be "multiplexed" with a GAPDH amplification reaction.
- multiplexing both the target gene and the internal standard gene GAPDH are amplified concurrently in a single sample.
- mRNA isolated from untreated cells is serially diluted. Each dilution is amplified in the presence of primer-probe sets specific for GAPDH only, target gene only ("single-plexing"), or both (multiplexing).
- standard curves of GAPDH and target mRNA signal as a function of dilution are generated from both the single-plexed and multiplexed samples.
- PCR reagents were obtained from Invitrogen Corporation, (Carlsbad,
- RT-PCR reactions were carried out by adding 20 ⁇ L PCR cocktail (2.5x PCR buffer minus MgCl 2 , 6.6 mM MgCl 2 , 375 ⁇ M each of dATP, dCTP, dCTP and dGTP, 375 nM each of forward primer and reverse primer, 125 nM of probe, 4 Units RNAse inhibitor, 1.25 Units PLATINUM® Taq, 5 Units MuLV reverse transcriptase, and 2.5x ROX dye) to 96-well plates containing 30 ⁇ L total RNA solution (20-200 ng). The RT reaction was carried out by incubation for 30 minutes at 48°C.
- PCR cocktail 2.5x PCR buffer minus MgCl 2 , 6.6 mM MgCl 2 , 375 ⁇ M each of dATP, dCTP, dCTP and dGTP, 375 nM each of forward primer and reverse primer, 125 nM of probe, 4 Units RNAs
- RiboGreenTM working reagent 170 ⁇ L of RiboGreenTM working reagent (RiboGreenTM reagent diluted 1 :350 in lOmM Tris-HCl, 1 mM EDTA, pH 7.5) is pipetted into a 96- well plate containing 30 ⁇ L purified, cellular RNA. The plate is read in a CytoFluor 4000 (PE Applied Biosystems) with excitation at 485nm and emission at 530nm.
- Probes and primers are designed to hybridize to a human a target sequence, using published sequence information.
- RNAZOLTM TEL-TEST "B” Inc., Friendswood
- Total RNA was prepared following manufacturer's recommended protocols.
- RNA was fractionated by electrophoresis through 1.2% agarose gels containing 1.1% formaldehyde using a MOPS buffer system
- N+ nylon membranes (Amersham Pharmacia Biotech, Piscataway, NJ) by overnight capillary transfer using a Northern/Southern Transfer buffer system (TEL-TEST "B"
- RNA transfer was confirmed by UN visualization.
- Crosslinker 2400 (Stratagene, Inc, La Jolla, CA) and then probed using
- QUICKHYBTM hybridization solution (Stratagene, La Jolla, CA) using manufacturer's recommendations for stringent conditions.
- a human a target specific primer probe set is prepared by PCR To normalize for variations in loading and transfer efficiency membranes are stripped and probed for human glyceraldehyde-3- ⁇ hosphate dehydrogenase (GAPDH) R ⁇ A (Clontech, Palo Alto, CA).
- GPDH glyceraldehyde-3- ⁇ hosphate dehydrogenase
- oligomeric compounds are designed to target different regions of the human target R ⁇ A.
- the oligomeric compounds are analyzed for their effect on human target mR ⁇ A levels by quantitative real-time PCR as described in other examples herein. Data are averages from three experiments.
- the target regions to which these prefened sequences are complementary are herein refened to as "preferred target segments" and are therefore prefened for targeting by oligomeric compounds of the present , invention.
- the sequences represent the reverse complement of the prefened antisense oligomeric compounds.
- prefened target segments have been found by experimentation to be open to, and accessible for, hybridization with the antisense oligomeric compounds of the present invention, one of skill in the art will recognize or be able to ascertain, using no more than routine experimentation, further embodiments of the invention that encompass other oligomeric compounds that specifically hybridize to these prefened target segments and consequently inhibit the expression of a target.
- antisense oligomeric compounds include antisense oligomeric compounds, antisense oligonucleotides, ribozymes, external guide sequence (EGS) oligonucleotides, alternate splicers, primers, probes, and other short oligomeric compounds that hybridize to at least a portion of the target nucleic acid.
- GCS external guide sequence
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Organic Chemistry (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Wood Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Plant Pathology (AREA)
- Microbiology (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Virology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002505090A CA2505090A1 (en) | 2002-11-05 | 2003-11-04 | Conjugated oligomeric compounds and their use in gene modulation |
| AU2003295389A AU2003295389A1 (en) | 2002-11-05 | 2003-11-04 | Conjugated oligomeric compounds and their use in gene modulation |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US42376002P | 2002-11-05 | 2002-11-05 | |
| US60/423,760 | 2002-11-05 | ||
| US61624103A | 2003-07-09 | 2003-07-09 | |
| US10/616,241 | 2003-07-09 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2004044141A2 true WO2004044141A2 (en) | 2004-05-27 |
| WO2004044141A8 WO2004044141A8 (en) | 2004-08-26 |
| WO2004044141A3 WO2004044141A3 (en) | 2005-05-19 |
Family
ID=32314505
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2003/035088 Ceased WO2004044141A2 (en) | 2002-11-05 | 2003-11-04 | Conjugated oligomeric compounds and their use in gene modulation |
Country Status (3)
| Country | Link |
|---|---|
| AU (1) | AU2003295389A1 (en) |
| CA (1) | CA2505090A1 (en) |
| WO (1) | WO2004044141A2 (en) |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1800695A1 (en) * | 2005-12-21 | 2007-06-27 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Immuno-RNA-constructs |
| AU2006280600B2 (en) * | 2005-08-17 | 2012-01-19 | Bioneer Corporation | Sirna-hydrophilic polymer conjugates for intracellular delivery of siRNA and method thereof |
| US8324365B2 (en) | 2003-04-03 | 2012-12-04 | Korea Advanced Institute Of Science And Technology | Conjugate for gene transfer comprising oligonucleotide and hydrophilic polymer, polyelectrolyte complex micelles formed from the conjugate, and methods for preparation thereof |
| WO2014076195A1 (en) | 2012-11-15 | 2014-05-22 | Santaris Pharma A/S | Oligonucleotide conjugates |
| WO2014118267A1 (en) | 2013-01-30 | 2014-08-07 | Santaris Pharma A/S | Lna oligonucleotide carbohydrate conjugates |
| WO2014118272A1 (en) | 2013-01-30 | 2014-08-07 | Santaris Pharma A/S | Antimir-122 oligonucleotide carbohydrate conjugates |
| US8969543B2 (en) | 2003-04-03 | 2015-03-03 | Bioneer Corporation | SiRNA-hydrophilic polymer conjugates for intracellular delivery of siRNA and method thereof |
| US9045739B2 (en) | 2004-01-16 | 2015-06-02 | Fraunhofer-Gesellschaft Zur Forderung Der Angewandten Forschung E.V. | Immunokinases |
| US9394333B2 (en) | 2008-12-02 | 2016-07-19 | Wave Life Sciences Japan | Method for the synthesis of phosphorus atom modified nucleic acids |
| US9598458B2 (en) | 2012-07-13 | 2017-03-21 | Wave Life Sciences Japan, Inc. | Asymmetric auxiliary group |
| US9605019B2 (en) | 2011-07-19 | 2017-03-28 | Wave Life Sciences Ltd. | Methods for the synthesis of functionalized nucleic acids |
| US9617547B2 (en) | 2012-07-13 | 2017-04-11 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant |
| EP2363481B1 (en) | 2006-05-05 | 2017-04-12 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulating gene expression |
| US9744183B2 (en) | 2009-07-06 | 2017-08-29 | Wave Life Sciences Ltd. | Nucleic acid prodrugs and methods of use thereof |
| US9879265B2 (en) | 2013-06-27 | 2018-01-30 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugates |
| US9982257B2 (en) | 2012-07-13 | 2018-05-29 | Wave Life Sciences Ltd. | Chiral control |
| US10144933B2 (en) | 2014-01-15 | 2018-12-04 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having immunity induction activity, and immunity induction activator |
| US10149905B2 (en) | 2014-01-15 | 2018-12-11 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having antitumor effect and antitumor agent |
| US10160969B2 (en) | 2014-01-16 | 2018-12-25 | Wave Life Sciences Ltd. | Chiral design |
| US10266825B2 (en) | 2002-11-05 | 2019-04-23 | Ionis Pharmaceuticals, Inc. | Compositions comprising alternating 2′-modified nucleosides for use in gene modulation |
| US10322173B2 (en) | 2014-01-15 | 2019-06-18 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having anti-allergic activity, and anti-allergic agent |
| US10358643B2 (en) | 2014-01-30 | 2019-07-23 | Hoffmann-La Roche, Inc. | Poly oligomer compound with biocleavable conjugates |
| US10428019B2 (en) | 2010-09-24 | 2019-10-01 | Wave Life Sciences Ltd. | Chiral auxiliaries |
| US10478449B2 (en) | 2002-11-05 | 2019-11-19 | Ionis Pharmaceuticals, Inc. | 2′-methoxy substituted oligomeric compounds and compositions for use in gene modulations |
| CN114127306A (en) * | 2019-07-08 | 2022-03-01 | 加利福尼亚大学董事会 | Lipid-modified oligonucleotides and methods of use thereof |
| US20220088202A1 (en) * | 2019-01-23 | 2022-03-24 | Shanghai Jiaotong University | Nucleic Acid-Drug Conjugate, Drug Delivery System, Preparation Method Therefor and Use Thereof |
| US12472148B2 (en) | 2021-04-26 | 2025-11-18 | Celanese Eva Performance Polymers Llc | Implantable device for sustained release of a macromolecular drug compound |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19502912A1 (en) * | 1995-01-31 | 1996-08-01 | Hoechst Ag | G-Cap Stabilized Oligonucleotides |
| ATE216594T1 (en) * | 1995-09-01 | 2002-05-15 | Univ Washington | INTERACTIVE MOLECULAR CONJUGATES |
| US20020102267A1 (en) * | 1999-10-21 | 2002-08-01 | Lu Peter S. | CLASP-5 transmembrane protein |
| US20030190635A1 (en) * | 2002-02-20 | 2003-10-09 | Mcswiggen James A. | RNA interference mediated treatment of Alzheimer's disease using short interfering RNA |
| US6559279B1 (en) * | 2000-09-08 | 2003-05-06 | Isis Pharmaceuticals, Inc. | Process for preparing peptide derivatized oligomeric compounds |
-
2003
- 2003-11-04 WO PCT/US2003/035088 patent/WO2004044141A2/en not_active Ceased
- 2003-11-04 CA CA002505090A patent/CA2505090A1/en not_active Abandoned
- 2003-11-04 AU AU2003295389A patent/AU2003295389A1/en not_active Abandoned
Cited By (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10478449B2 (en) | 2002-11-05 | 2019-11-19 | Ionis Pharmaceuticals, Inc. | 2′-methoxy substituted oligomeric compounds and compositions for use in gene modulations |
| US10266825B2 (en) | 2002-11-05 | 2019-04-23 | Ionis Pharmaceuticals, Inc. | Compositions comprising alternating 2′-modified nucleosides for use in gene modulation |
| US8969543B2 (en) | 2003-04-03 | 2015-03-03 | Bioneer Corporation | SiRNA-hydrophilic polymer conjugates for intracellular delivery of siRNA and method thereof |
| US8324365B2 (en) | 2003-04-03 | 2012-12-04 | Korea Advanced Institute Of Science And Technology | Conjugate for gene transfer comprising oligonucleotide and hydrophilic polymer, polyelectrolyte complex micelles formed from the conjugate, and methods for preparation thereof |
| US9045739B2 (en) | 2004-01-16 | 2015-06-02 | Fraunhofer-Gesellschaft Zur Forderung Der Angewandten Forschung E.V. | Immunokinases |
| AU2006280600B2 (en) * | 2005-08-17 | 2012-01-19 | Bioneer Corporation | Sirna-hydrophilic polymer conjugates for intracellular delivery of siRNA and method thereof |
| WO2007071777A3 (en) * | 2005-12-21 | 2007-11-22 | Fraunhofer Ges Forschung | Immuno-rna-constructs |
| EP1800695A1 (en) * | 2005-12-21 | 2007-06-27 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Immuno-RNA-constructs |
| US8829178B2 (en) | 2005-12-21 | 2014-09-09 | Fraunhofer-Gesellschaft Zur Forderung Der Angewandten Forschung E.V. | Immuno-RNA-constructs |
| EP2363481B1 (en) | 2006-05-05 | 2017-04-12 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulating gene expression |
| US9695211B2 (en) | 2008-12-02 | 2017-07-04 | Wave Life Sciences Japan, Inc. | Method for the synthesis of phosphorus atom modified nucleic acids |
| US9394333B2 (en) | 2008-12-02 | 2016-07-19 | Wave Life Sciences Japan | Method for the synthesis of phosphorus atom modified nucleic acids |
| US10329318B2 (en) | 2008-12-02 | 2019-06-25 | Wave Life Sciences Ltd. | Method for the synthesis of phosphorus atom modified nucleic acids |
| US9744183B2 (en) | 2009-07-06 | 2017-08-29 | Wave Life Sciences Ltd. | Nucleic acid prodrugs and methods of use thereof |
| US10307434B2 (en) | 2009-07-06 | 2019-06-04 | Wave Life Sciences Ltd. | Nucleic acid prodrugs and methods of use thereof |
| US10428019B2 (en) | 2010-09-24 | 2019-10-01 | Wave Life Sciences Ltd. | Chiral auxiliaries |
| US9605019B2 (en) | 2011-07-19 | 2017-03-28 | Wave Life Sciences Ltd. | Methods for the synthesis of functionalized nucleic acids |
| US10280192B2 (en) | 2011-07-19 | 2019-05-07 | Wave Life Sciences Ltd. | Methods for the synthesis of functionalized nucleic acids |
| US9617547B2 (en) | 2012-07-13 | 2017-04-11 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant |
| US10167309B2 (en) | 2012-07-13 | 2019-01-01 | Wave Life Sciences Ltd. | Asymmetric auxiliary group |
| US9982257B2 (en) | 2012-07-13 | 2018-05-29 | Wave Life Sciences Ltd. | Chiral control |
| US9598458B2 (en) | 2012-07-13 | 2017-03-21 | Wave Life Sciences Japan, Inc. | Asymmetric auxiliary group |
| US10590413B2 (en) | 2012-07-13 | 2020-03-17 | Wave Life Sciences Ltd. | Chiral control |
| EP3406718A1 (en) | 2012-11-15 | 2018-11-28 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugates |
| US10077443B2 (en) | 2012-11-15 | 2018-09-18 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugates |
| US11155816B2 (en) | 2012-11-15 | 2021-10-26 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugates |
| WO2014076196A1 (en) | 2012-11-15 | 2014-05-22 | Santaris Pharma A/S | Anti apob antisense conjugate compounds |
| WO2014076195A1 (en) | 2012-11-15 | 2014-05-22 | Santaris Pharma A/S | Oligonucleotide conjugates |
| WO2014118272A1 (en) | 2013-01-30 | 2014-08-07 | Santaris Pharma A/S | Antimir-122 oligonucleotide carbohydrate conjugates |
| WO2014118267A1 (en) | 2013-01-30 | 2014-08-07 | Santaris Pharma A/S | Lna oligonucleotide carbohydrate conjugates |
| US10385342B2 (en) | 2013-06-27 | 2019-08-20 | Roche Innovation Center Copenhagen A/S | Methods of treatment using antisense oligomers and conjugates targeting PCSK9 |
| US11739332B2 (en) | 2013-06-27 | 2023-08-29 | Roche Innovation Center Copenhagen A/S | Antisense oligomers targeting PCSK9 |
| US10370668B2 (en) | 2013-06-27 | 2019-08-06 | Roche Innovation Center Copenhagen A/S | Manufacture of antisense oligomers and conjugates targeting PCSK9 |
| US12421516B2 (en) | 2013-06-27 | 2025-09-23 | Roche Innovation Center Copenhagen A/S | Antisense oligomers targeting PCSK9 |
| US10443058B2 (en) | 2013-06-27 | 2019-10-15 | Roche Innovation Center Copenhagen A/S | Antisense oligomers targeting PCSK9 |
| EP3591054A1 (en) | 2013-06-27 | 2020-01-08 | Roche Innovation Center Copenhagen A/S | Antisense oligomers and conjugates targeting pcsk9 |
| US9879265B2 (en) | 2013-06-27 | 2018-01-30 | Roche Innovation Center Copenhagen A/S | Oligonucleotide conjugates |
| US10322173B2 (en) | 2014-01-15 | 2019-06-18 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having anti-allergic activity, and anti-allergic agent |
| US10149905B2 (en) | 2014-01-15 | 2018-12-11 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having antitumor effect and antitumor agent |
| US10144933B2 (en) | 2014-01-15 | 2018-12-04 | Shin Nippon Biomedical Laboratories, Ltd. | Chiral nucleic acid adjuvant having immunity induction activity, and immunity induction activator |
| US10160969B2 (en) | 2014-01-16 | 2018-12-25 | Wave Life Sciences Ltd. | Chiral design |
| US10358643B2 (en) | 2014-01-30 | 2019-07-23 | Hoffmann-La Roche, Inc. | Poly oligomer compound with biocleavable conjugates |
| US20220088202A1 (en) * | 2019-01-23 | 2022-03-24 | Shanghai Jiaotong University | Nucleic Acid-Drug Conjugate, Drug Delivery System, Preparation Method Therefor and Use Thereof |
| CN114127306A (en) * | 2019-07-08 | 2022-03-01 | 加利福尼亚大学董事会 | Lipid-modified oligonucleotides and methods of use thereof |
| US12472148B2 (en) | 2021-04-26 | 2025-11-18 | Celanese Eva Performance Polymers Llc | Implantable device for sustained release of a macromolecular drug compound |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2004044141A8 (en) | 2004-08-26 |
| CA2505090A1 (en) | 2004-05-27 |
| AU2003295389A1 (en) | 2004-06-03 |
| WO2004044141A3 (en) | 2005-05-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7919612B2 (en) | 2′-substituted oligomeric compounds and compositions for use in gene modulations | |
| AU2010201712B2 (en) | 2'-substituted oligomeric compounds and compositions for use in gene modulations | |
| US20050119470A1 (en) | Conjugated oligomeric compounds and their use in gene modulation | |
| US20040161844A1 (en) | Sugar and backbone-surrogate-containing oligomeric compounds and compositions for use in gene modulation | |
| WO2004044141A2 (en) | Conjugated oligomeric compounds and their use in gene modulation | |
| AU2003290596A2 (en) | Sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation | |
| US20050042647A1 (en) | Phosphorous-linked oligomeric compounds and their use in gene modulation | |
| US20040147022A1 (en) | 2'-methoxy substituted oligomeric compounds and compositions for use in gene modulations | |
| US20220288100A1 (en) | 2'-methoxy substituted oligomeric compounds and compositions for use in gene modulations | |
| US20050032069A1 (en) | Oligomeric compounds having modified bases for binding to adenine and guanine and their use in gene modulation | |
| US20040171031A1 (en) | Sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation | |
| WO2004041924A2 (en) | Non-phosphorous-linked oligomeric compounds and their use in gene modulation | |
| US20040266706A1 (en) | Cross-linked oligomeric compounds and their use in gene modulation | |
| EP1560839A2 (en) | Chimeric oligomeric compounds and their use in gene modulation | |
| WO2004044135A2 (en) | Structural motifs and oligomeric compounds and their use in gene modulation | |
| WO2004044137A2 (en) | Modified oligomeric compounds and compositions for use in gene modulation | |
| US7812149B2 (en) | 2′-Fluoro substituted oligomeric compounds and compositions for use in gene modulations | |
| US20040171028A1 (en) | Phosphorous-linked oligomeric compounds and their use in gene modulation | |
| US20040254358A1 (en) | Phosphorous-linked oligomeric compounds and their use in gene modulation | |
| AU2011203091B2 (en) | Chimeric oligomeric compounds and their use in gene modulation | |
| US20160076031A1 (en) | Phosphorous-linked oligomeric compounds and their use in gene modulation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| CFP | Corrected version of a pamphlet front page |
Free format text: UNDER (54) PUBLISHED TITLE IN ENGLISH REPLACED BY CORRECT TITLE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2505090 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003295389 Country of ref document: AU |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 122 | Ep: pct application non-entry in european phase | ||
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| WWW | Wipo information: withdrawn in national office |
Country of ref document: JP |