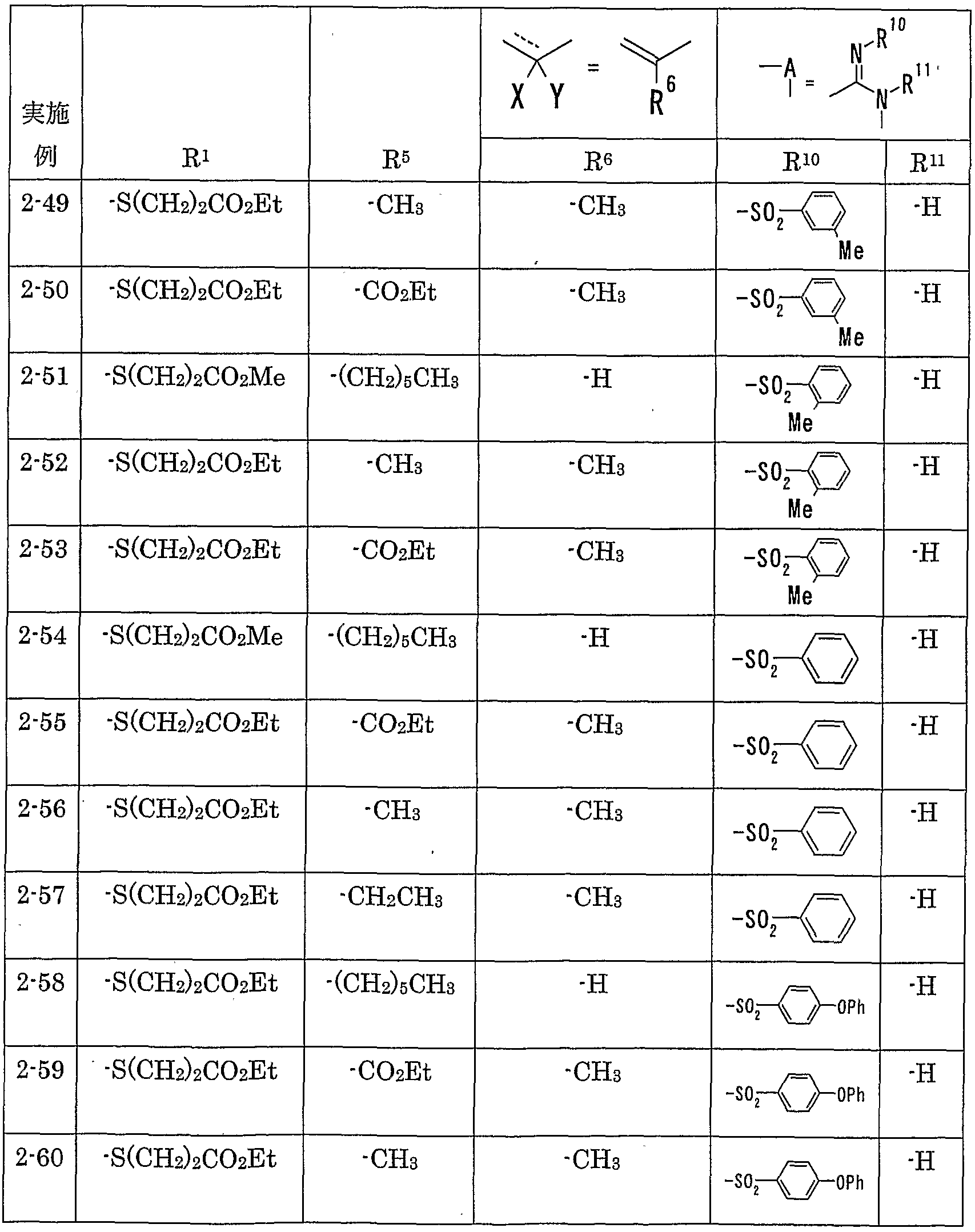
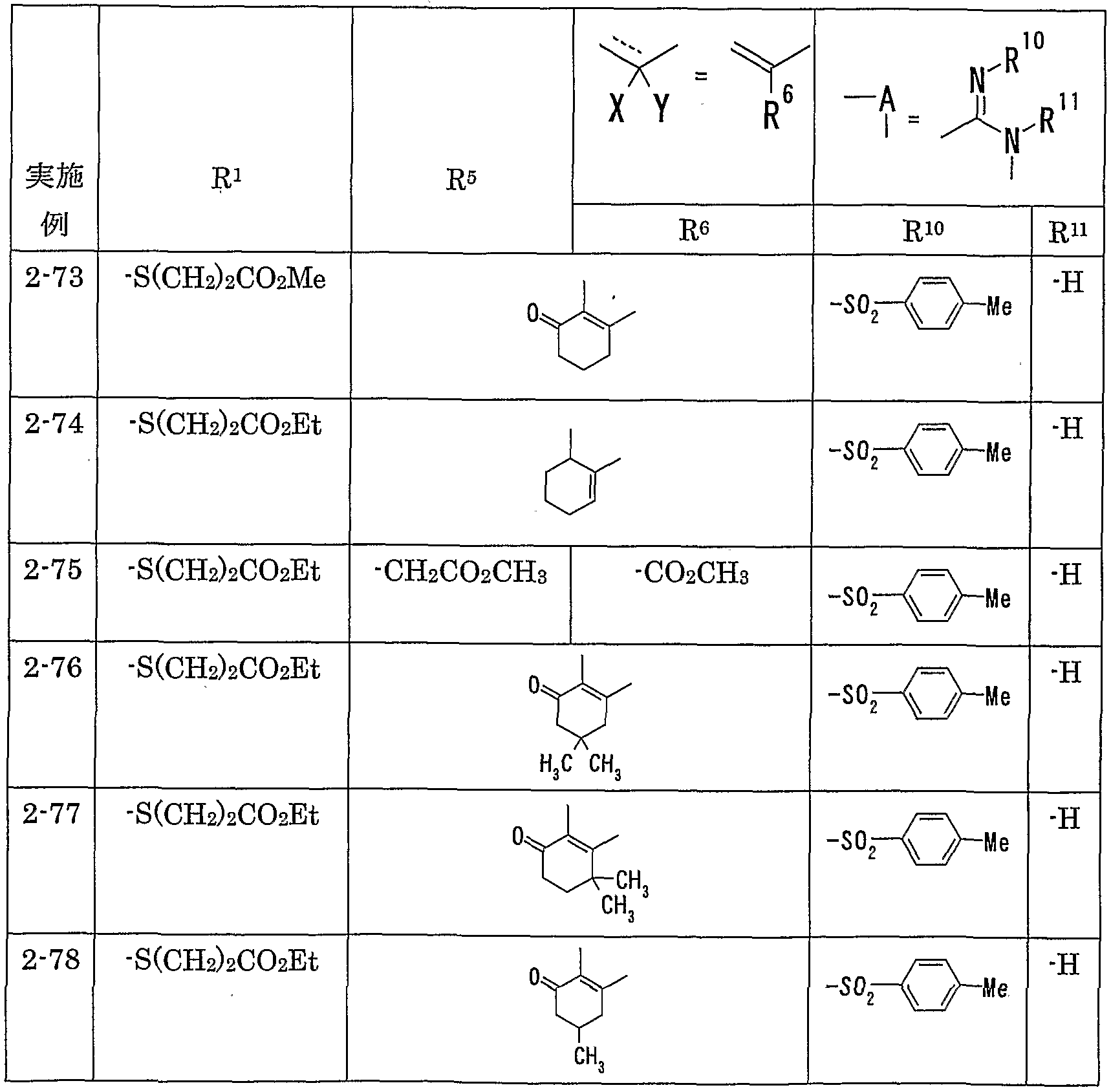
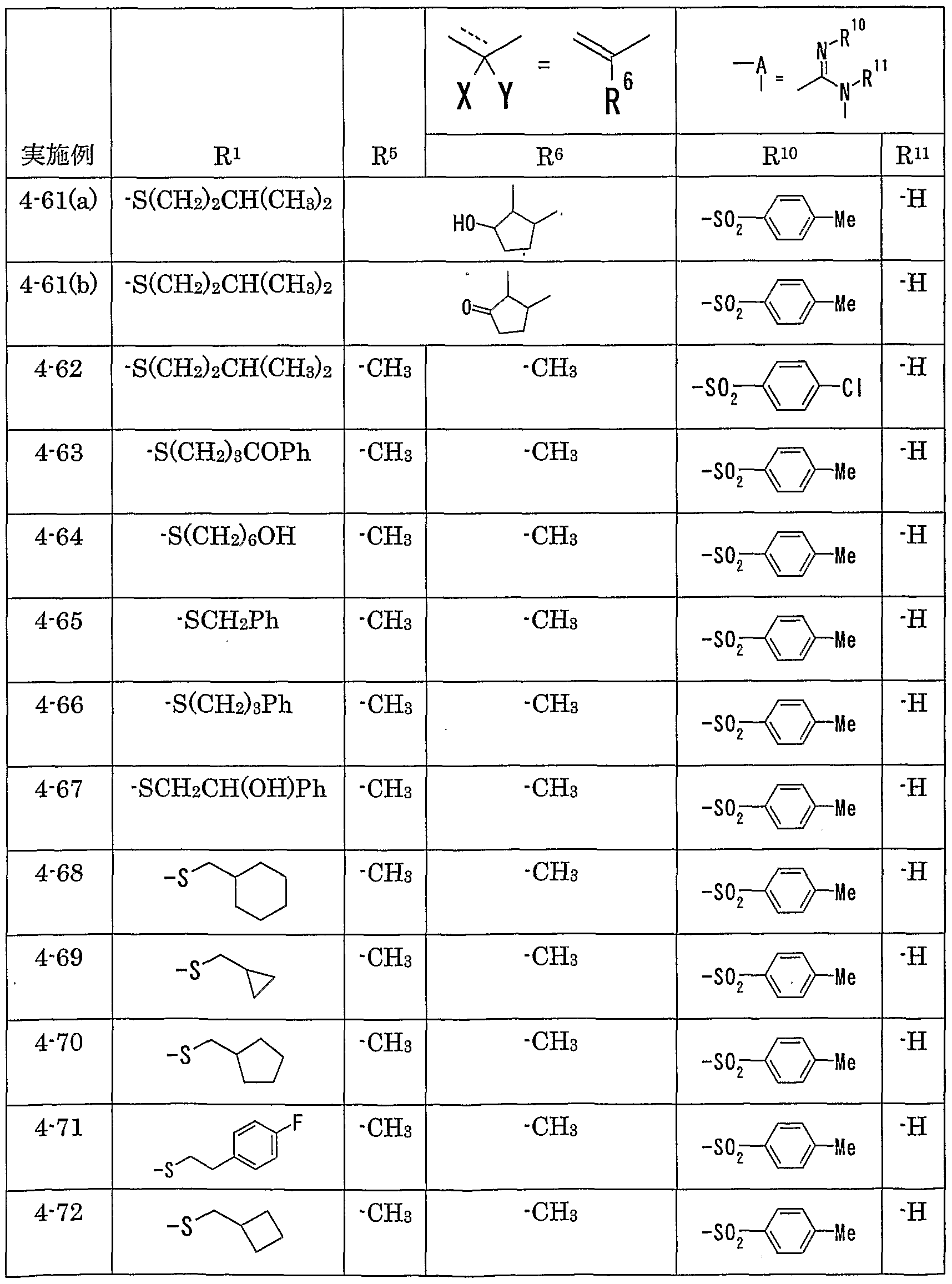
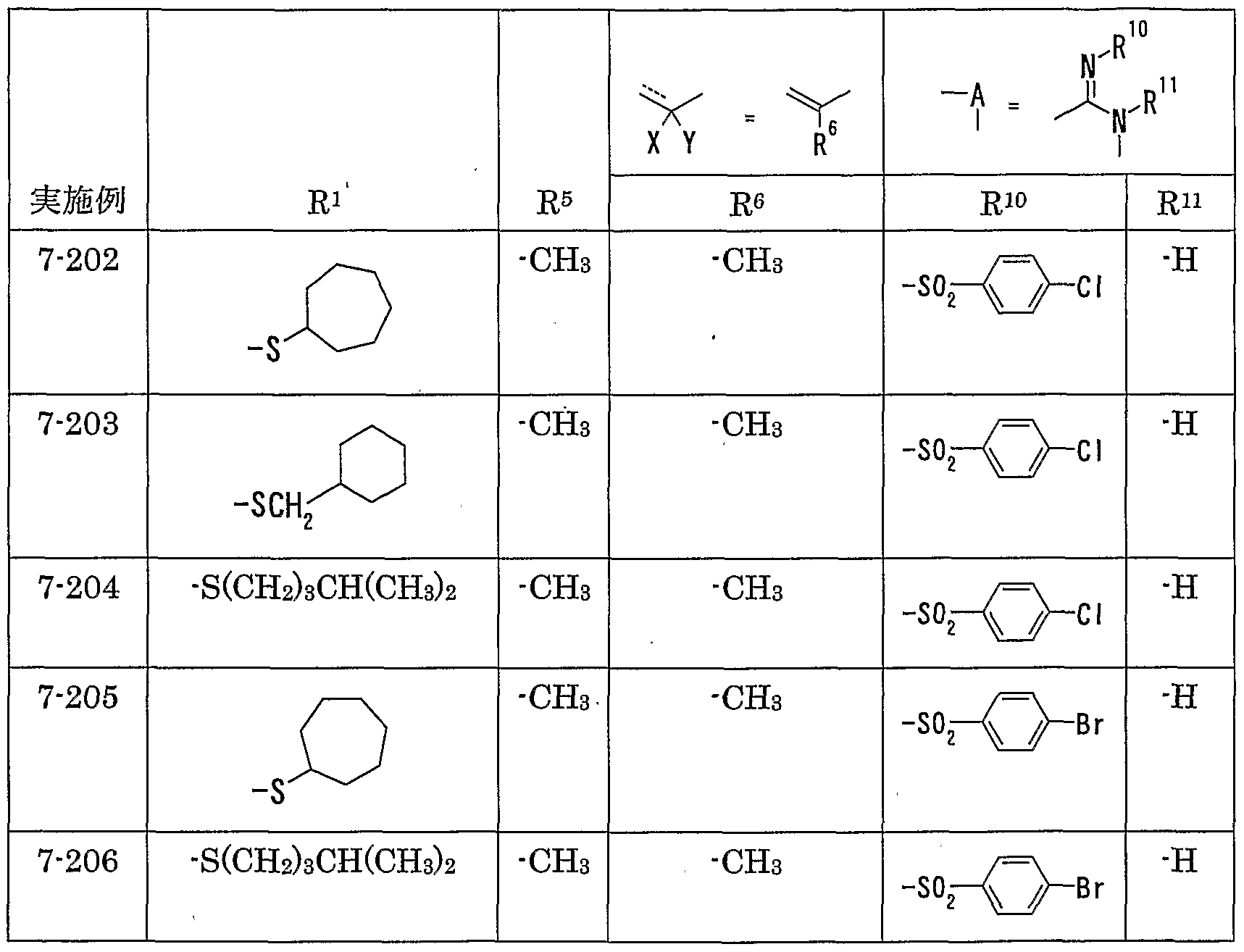
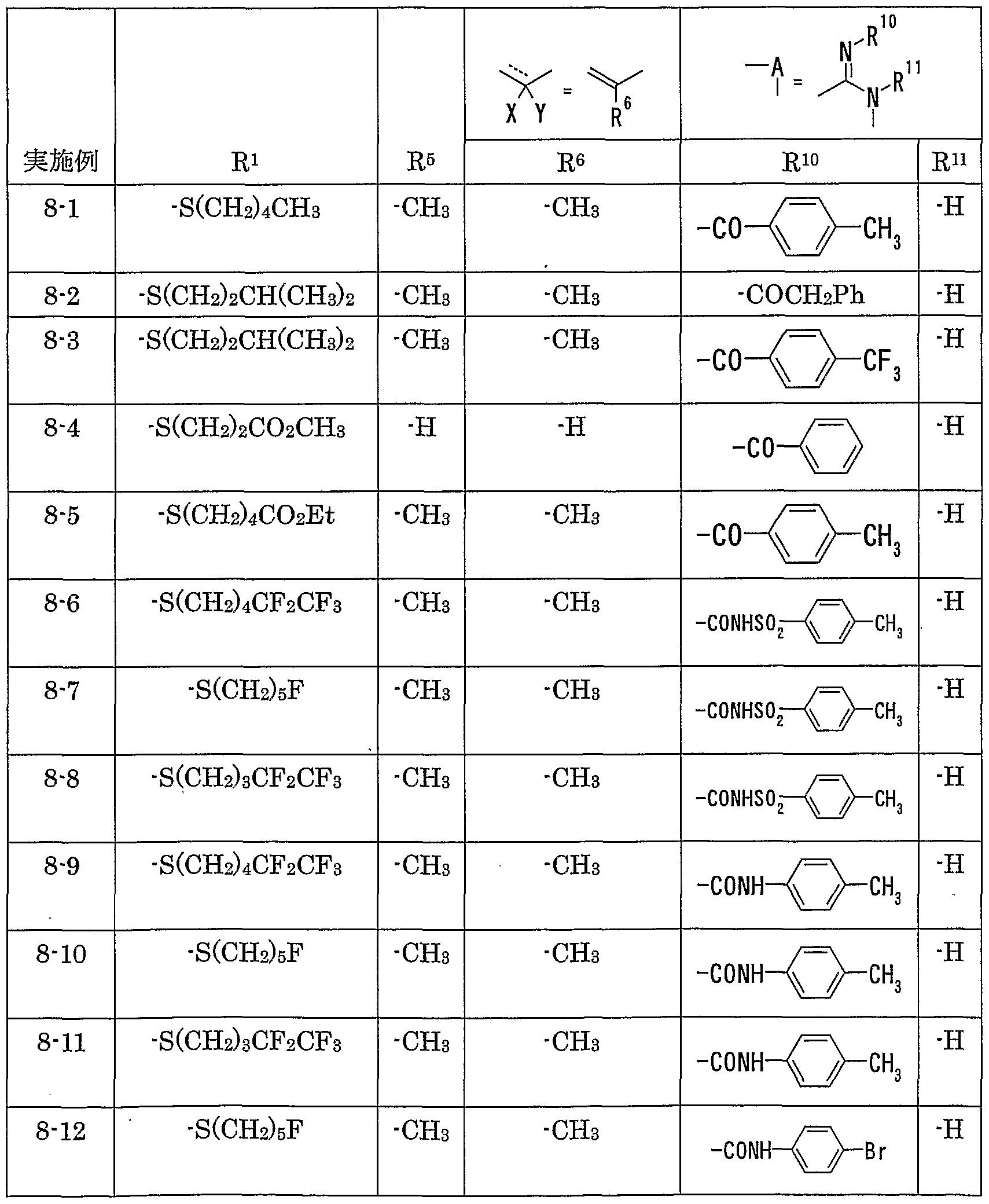
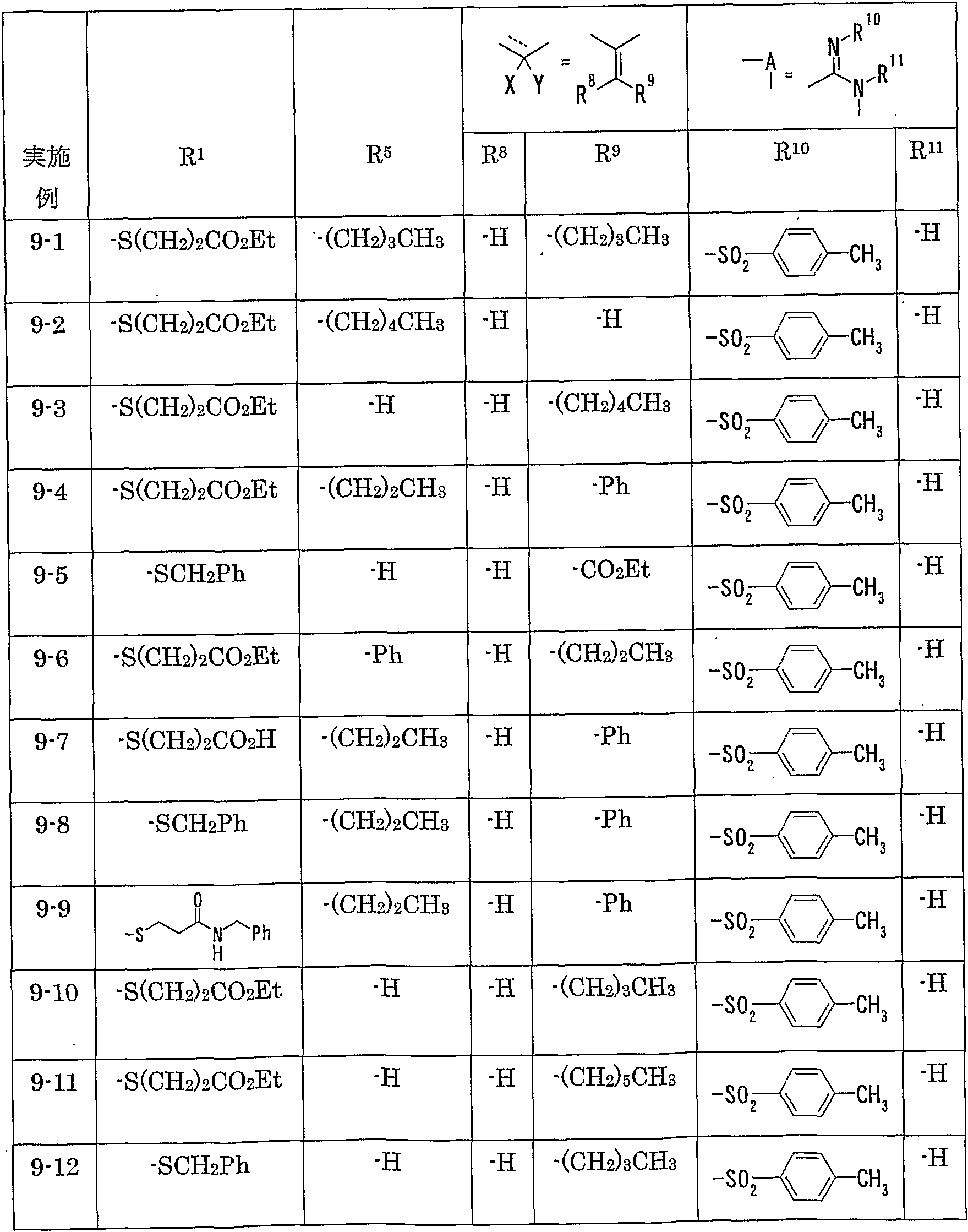
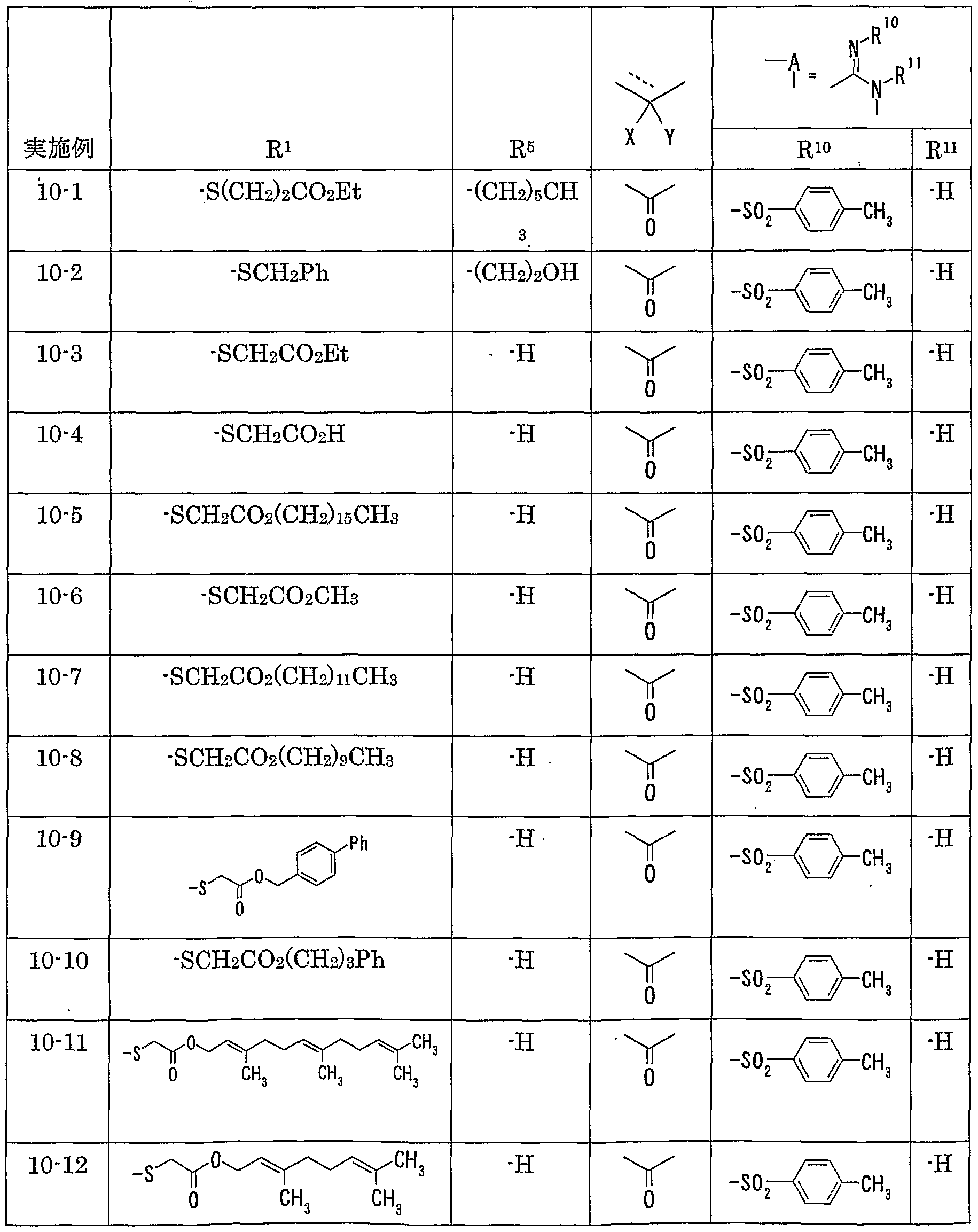
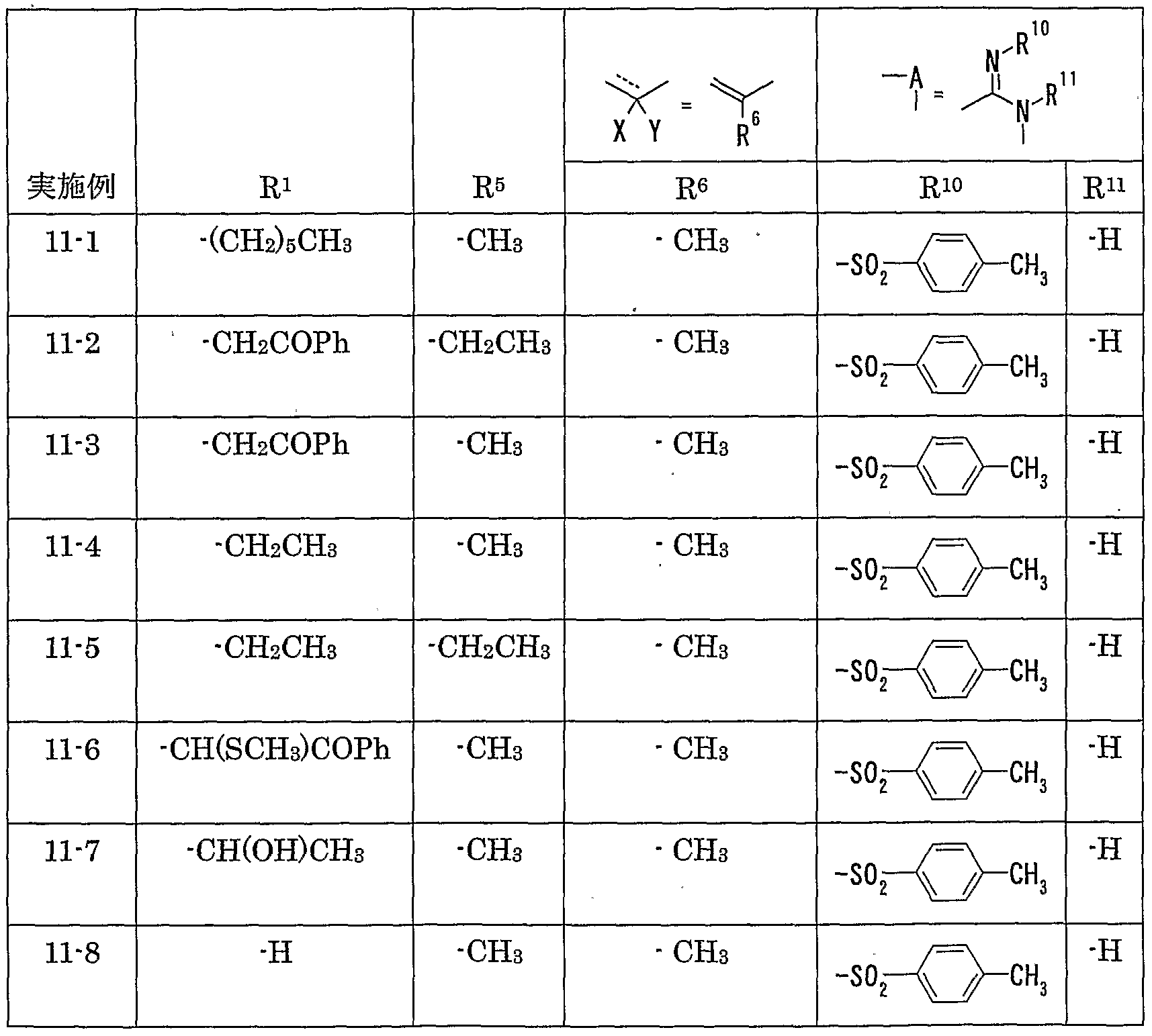
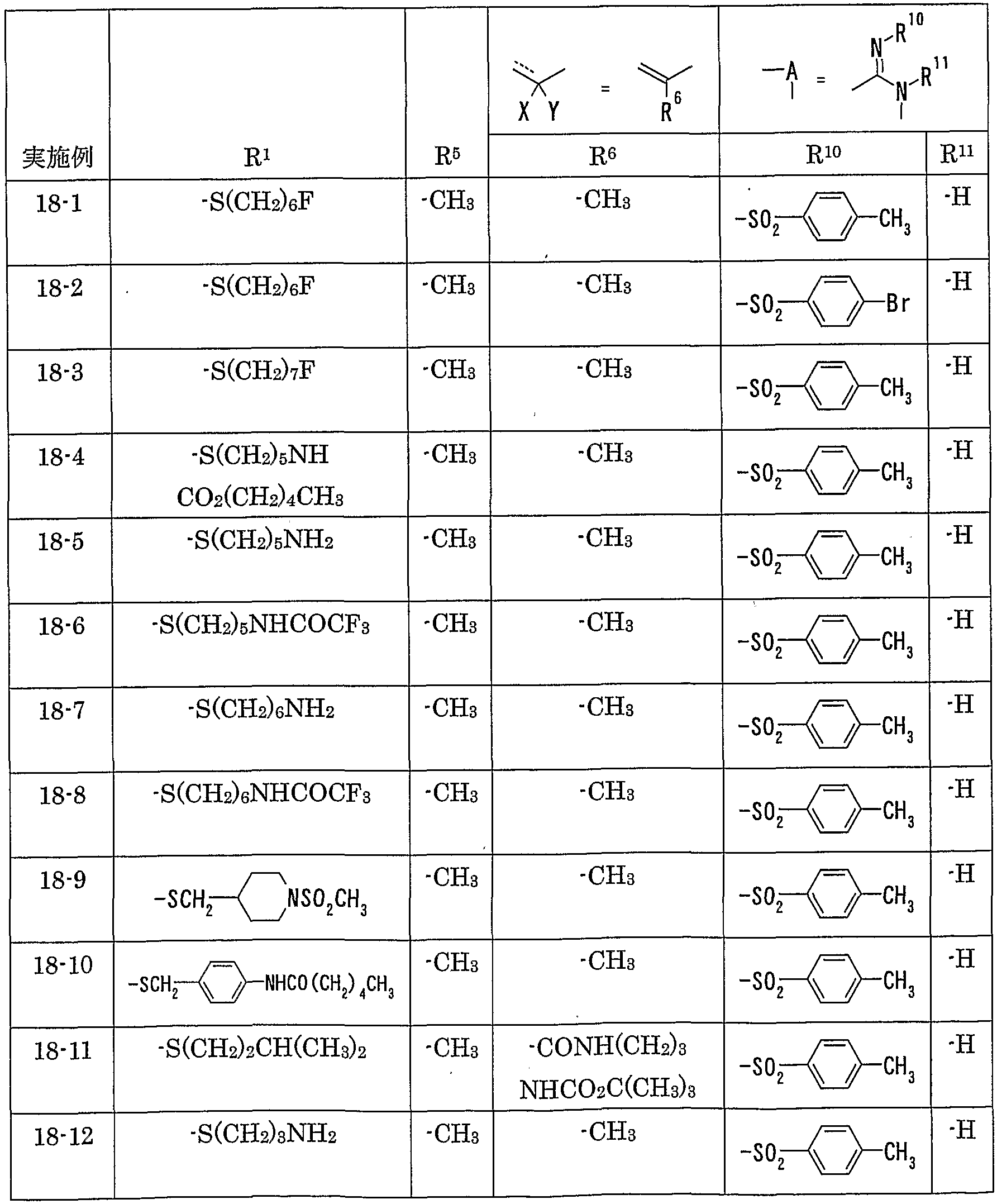
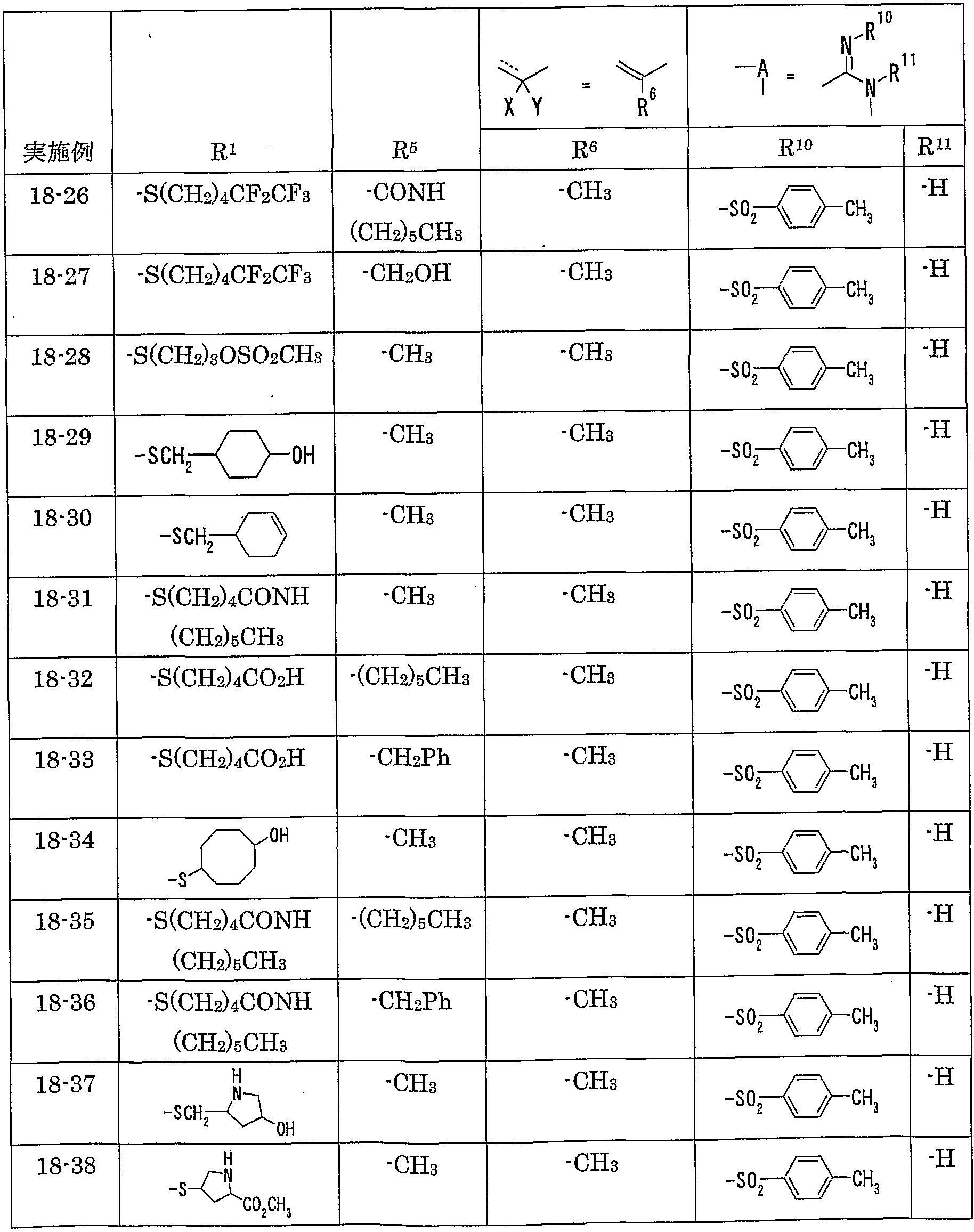
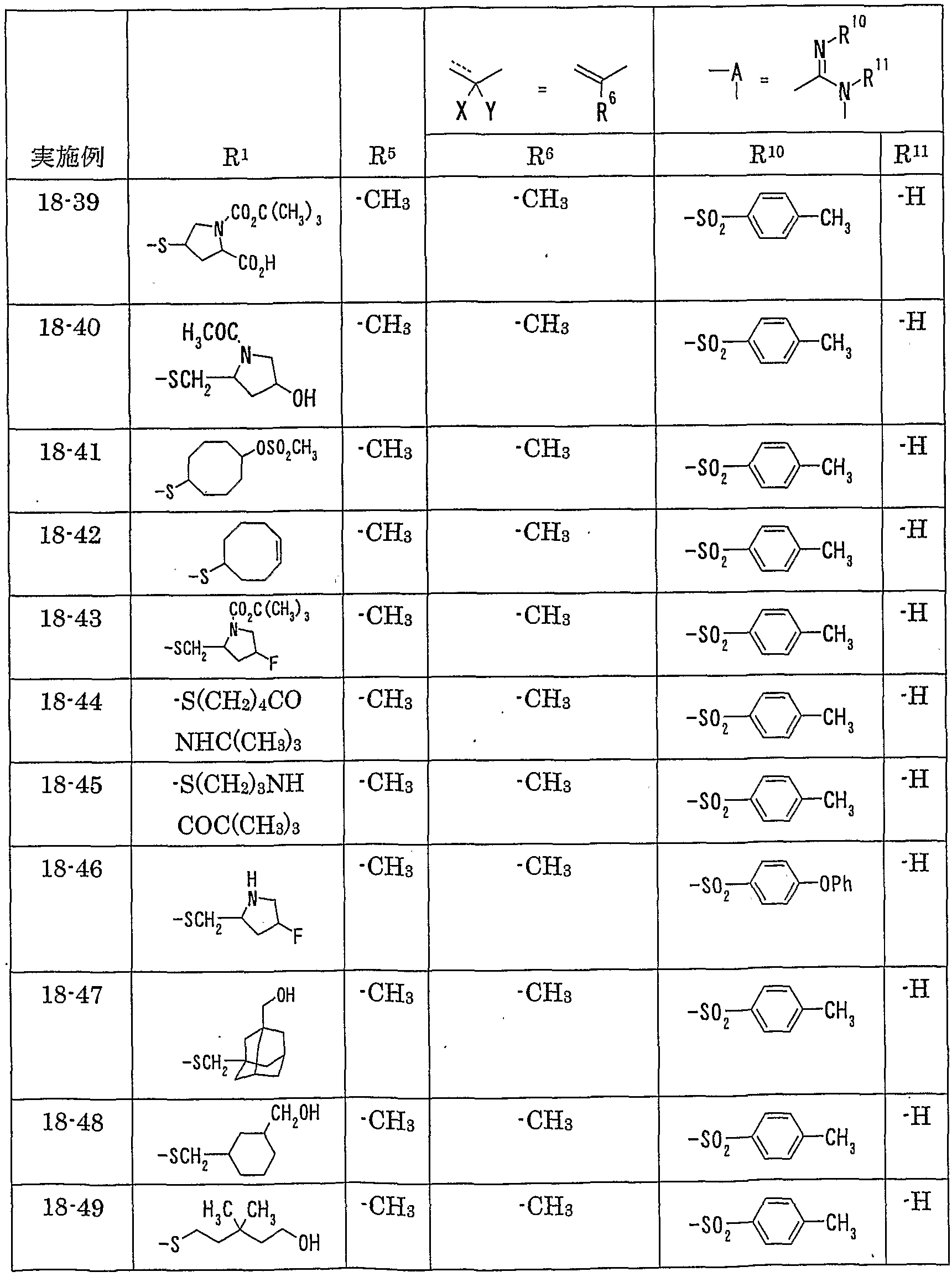
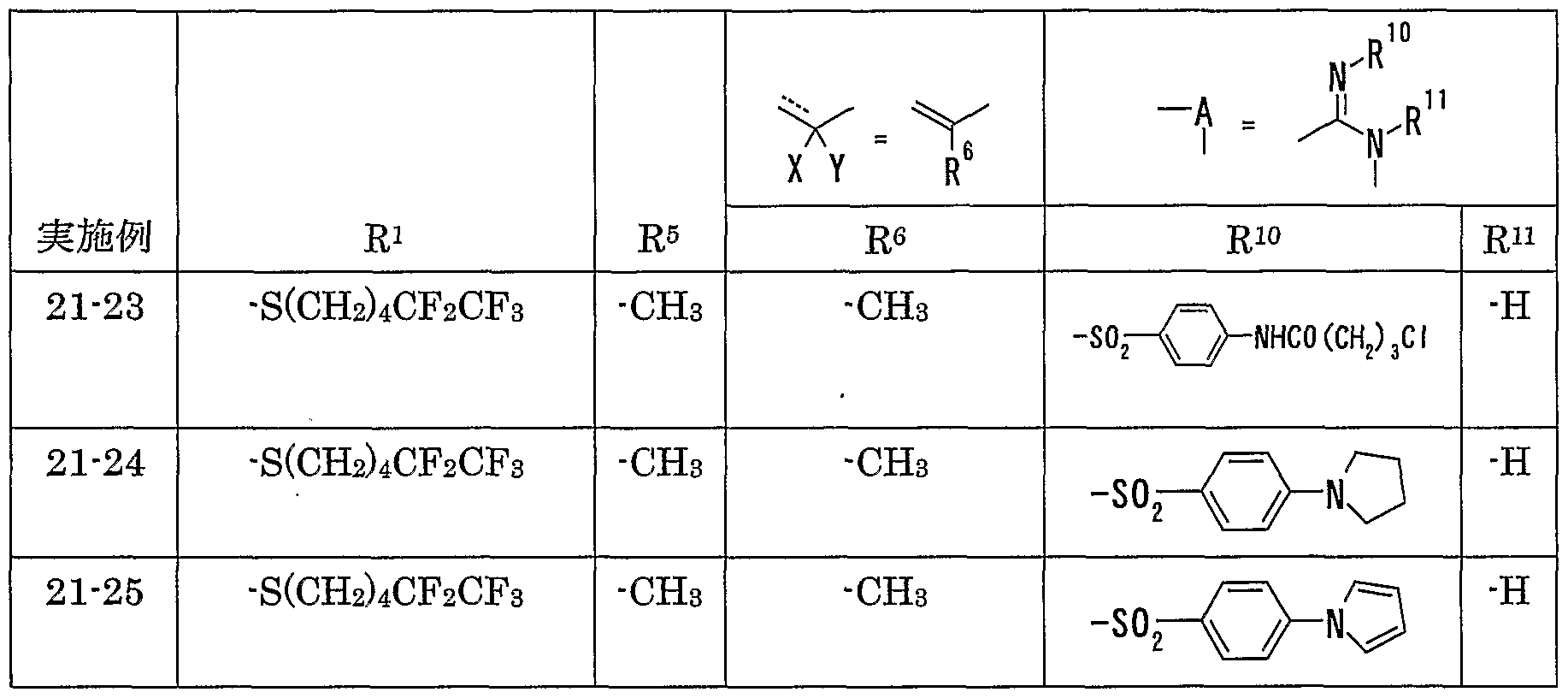
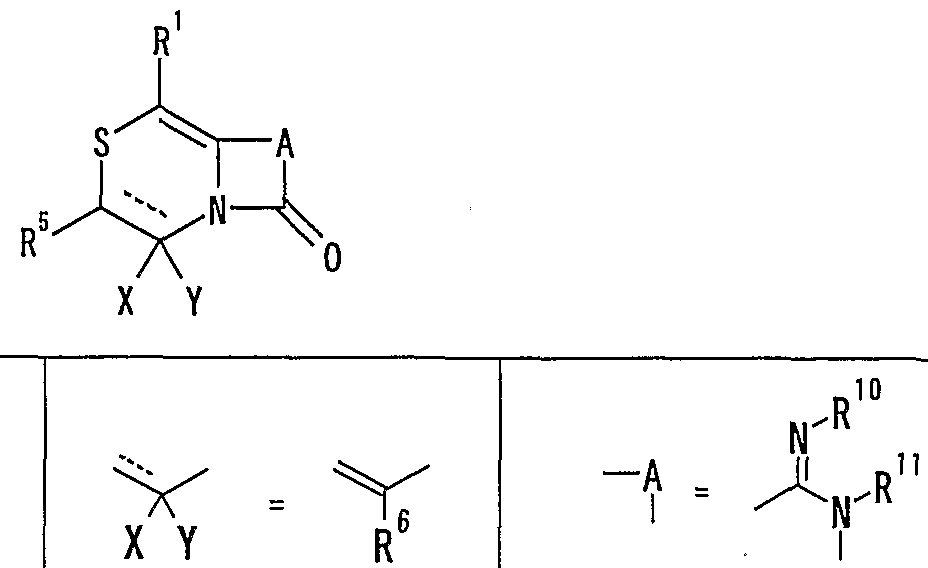
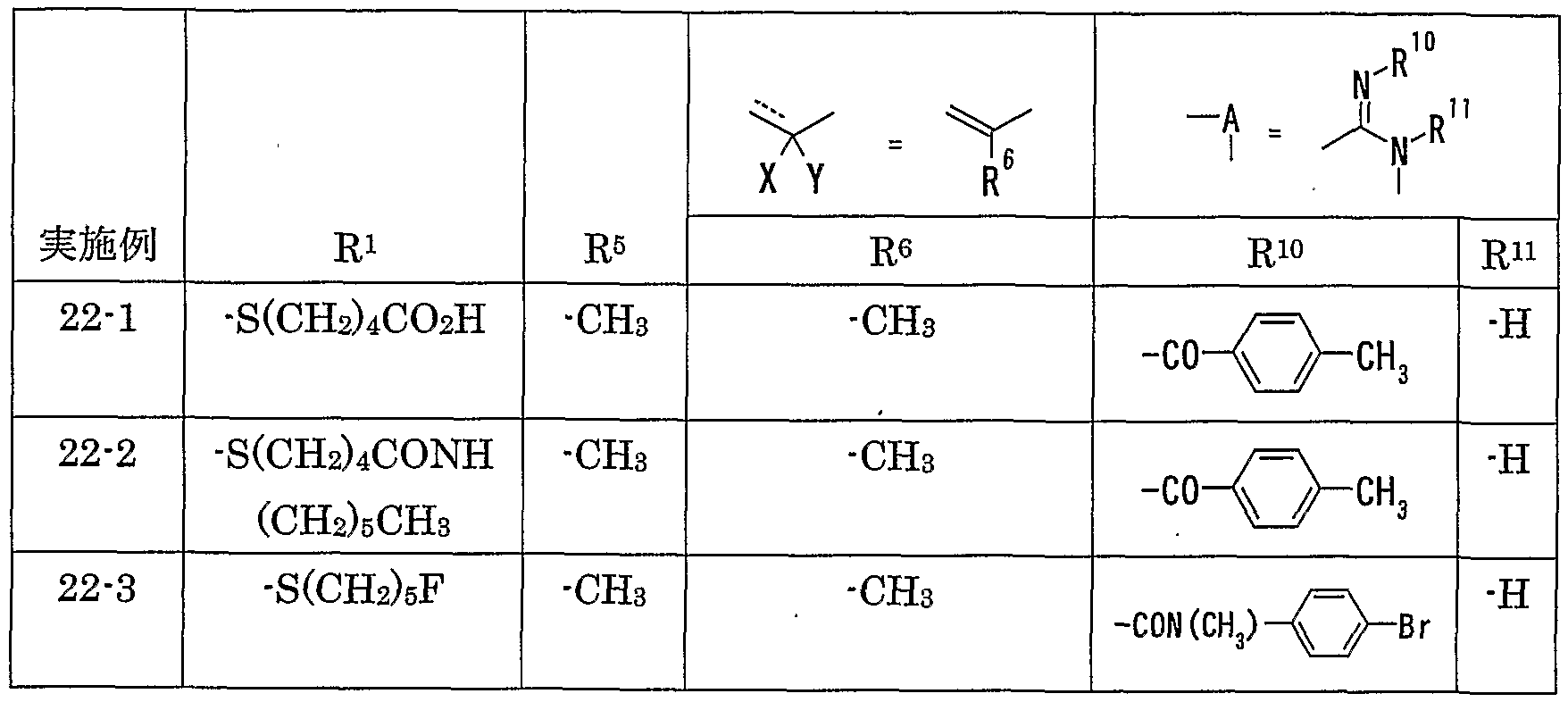
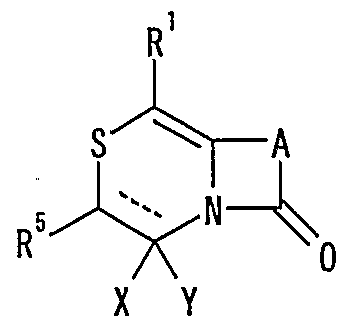
WO2002092606A1 - Fused imidazolidine derivatives, process for preparation of the same and use thereof - Google Patents
Fused imidazolidine derivatives, process for preparation of the same and use thereof Download PDFInfo
- Publication number
- WO2002092606A1 WO2002092606A1 PCT/JP2002/004640 JP0204640W WO02092606A1 WO 2002092606 A1 WO2002092606 A1 WO 2002092606A1 JP 0204640 W JP0204640 W JP 0204640W WO 02092606 A1 WO02092606 A1 WO 02092606A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- optionally substituted
- group
- hydrogen atom
- hydrocarbon group
- substituted hydrocarbon
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/54—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame
- A61K31/542—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/554—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having at least one nitrogen and one sulfur as ring hetero atoms, e.g. clothiapine, diltiazem
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6561—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing systems of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring or ring system, with or without other non-condensed hetero rings
Definitions
- the present invention relates to a novel condensed imidazolidine derivative useful as a medicament such as an agent for treating a joint disease, a method for producing the same, or an agent therefor.
- Osteoarthritis is a progressive disease with degeneration and destruction of articular cartilage caused by aging, obesity> mechanical stress, inflammation, etc., and mainly occurs in the knee and hip joints.
- anti-inflammatory drugs and hyaluronic acid preparations have been used as therapeutic agents mainly for the purpose of pain relief [Bad Etz Chin- (Bird, HA :), Drugs & Aging, 12, 87-95, 1998, Lund, B., Scandinavian Journal Rhematology (Scandinavian J. Rheumatol.), Vol. 27, pp. 32-37, 1998, and Ando Inoue, Diagnosis and treatment of osteoarthritis, Medical Shoin, 94-99, 1994].
- Cartilage is formed from chondrocytes and a cartilage matrix produced by chondrocytes.
- the matrix components are proteodalican, which has hydration ability and gives flexibility and elasticity to cartilage, and type II, which forms a reticulated skeleton. It is roughly divided into collagen.
- Cartilage destruction in osteoarthritis can be divided into two stages: proteodalican degradation followed by type II collagen degradation [Poole, AR], Takashi Fujisawa (translation), protein nucleic acid enzymes, 40, 520—531, 1995, Takei Doi et al., The Bone, 1, 293—297, 2000].
- Hymen ialdisine a compound derived from a marine sponge, has been reported to inhibit proteoglycan degradation from articular cartilage [Alison, MB (Alison, MB), The Journal of Pharmaceutical Sciences and Ande The Journal of Pharmacology and Experimental Therapeutics, Vol. 290, pp. 587-593, 1999], and a derivative of hymenialdisine, debromohymenialdisine (DBH), Inhibition of matrix meta-oral proteinase (MMP) mRNA production in articular chondrocyte culture system [Vasios, G. Proceedings of 1999 American College of Rheumatology Announcer Meeting, poster session! (Poster Session D), No. 1123], Protective effect on articular cartilage in animal models [Huibregtse, ⁇ ⁇ , et al., Proceedings of 1999 American College of Rheumatology Annual Meeting ), Poster Session D, No. 1102].
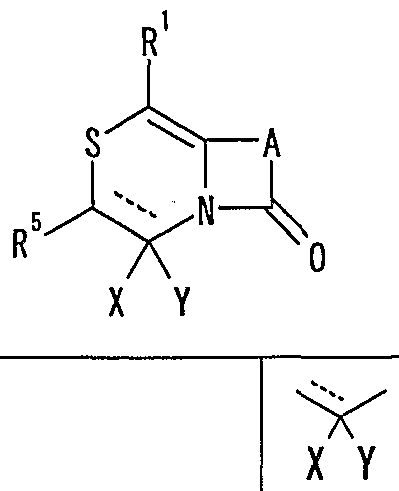
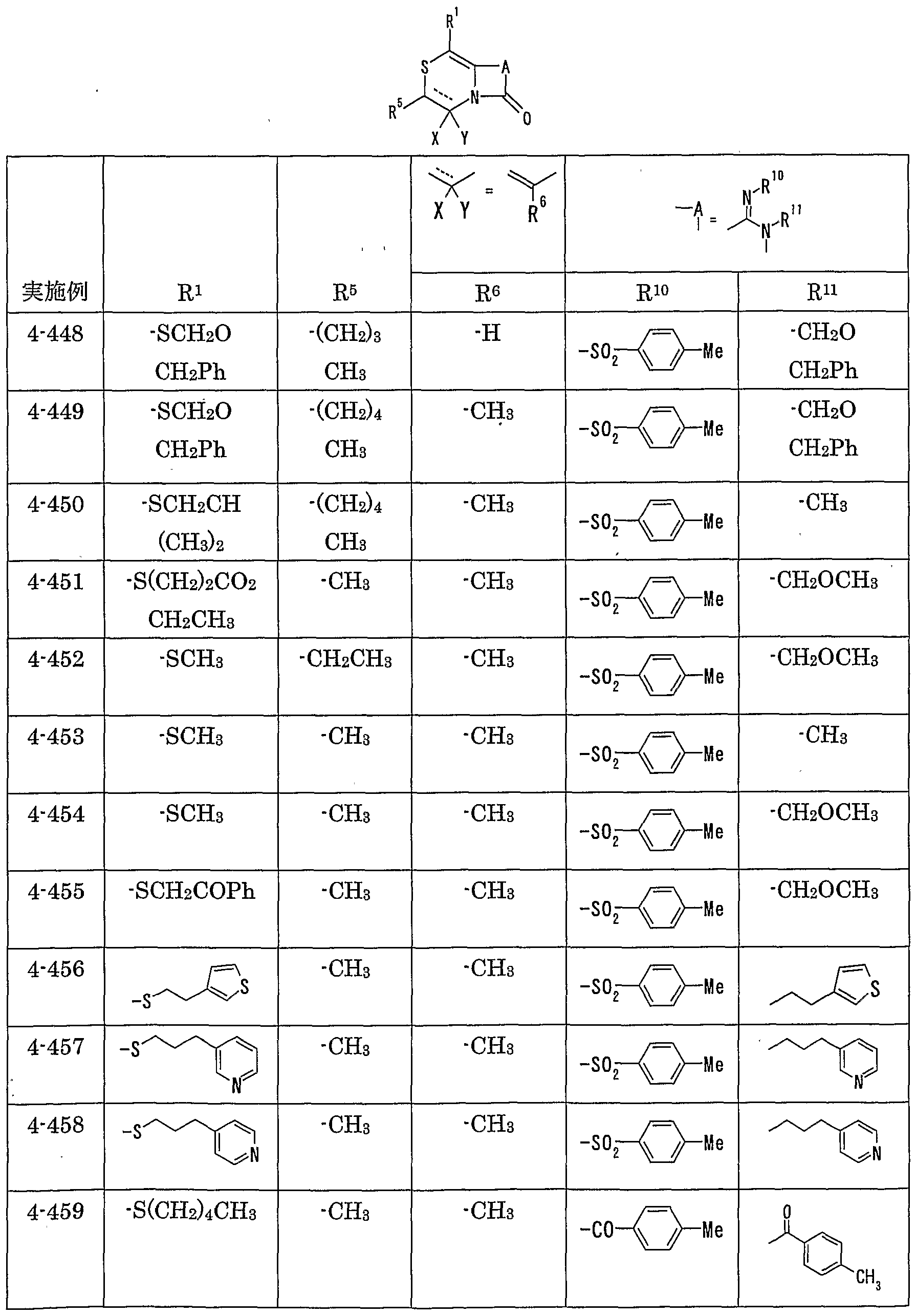
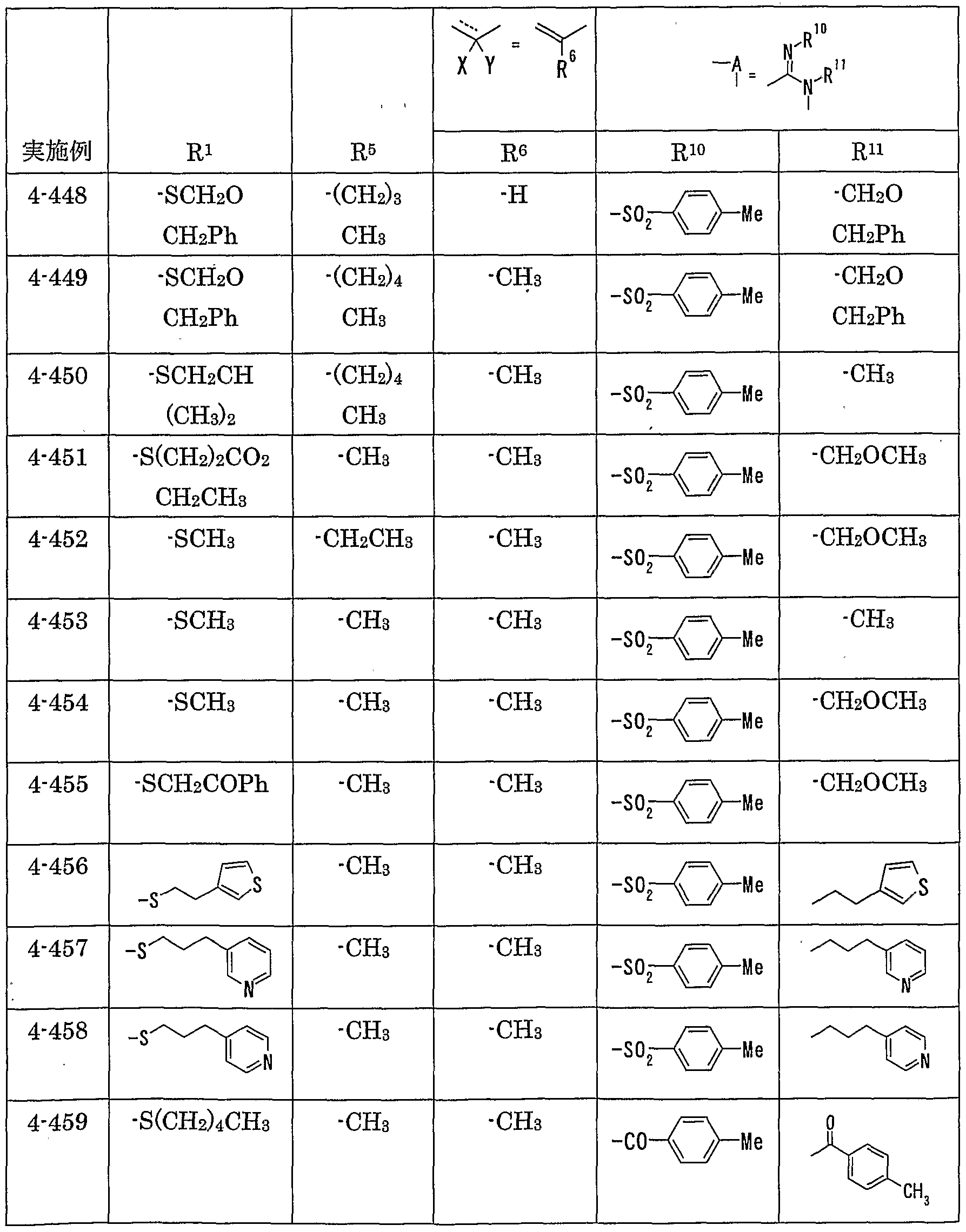
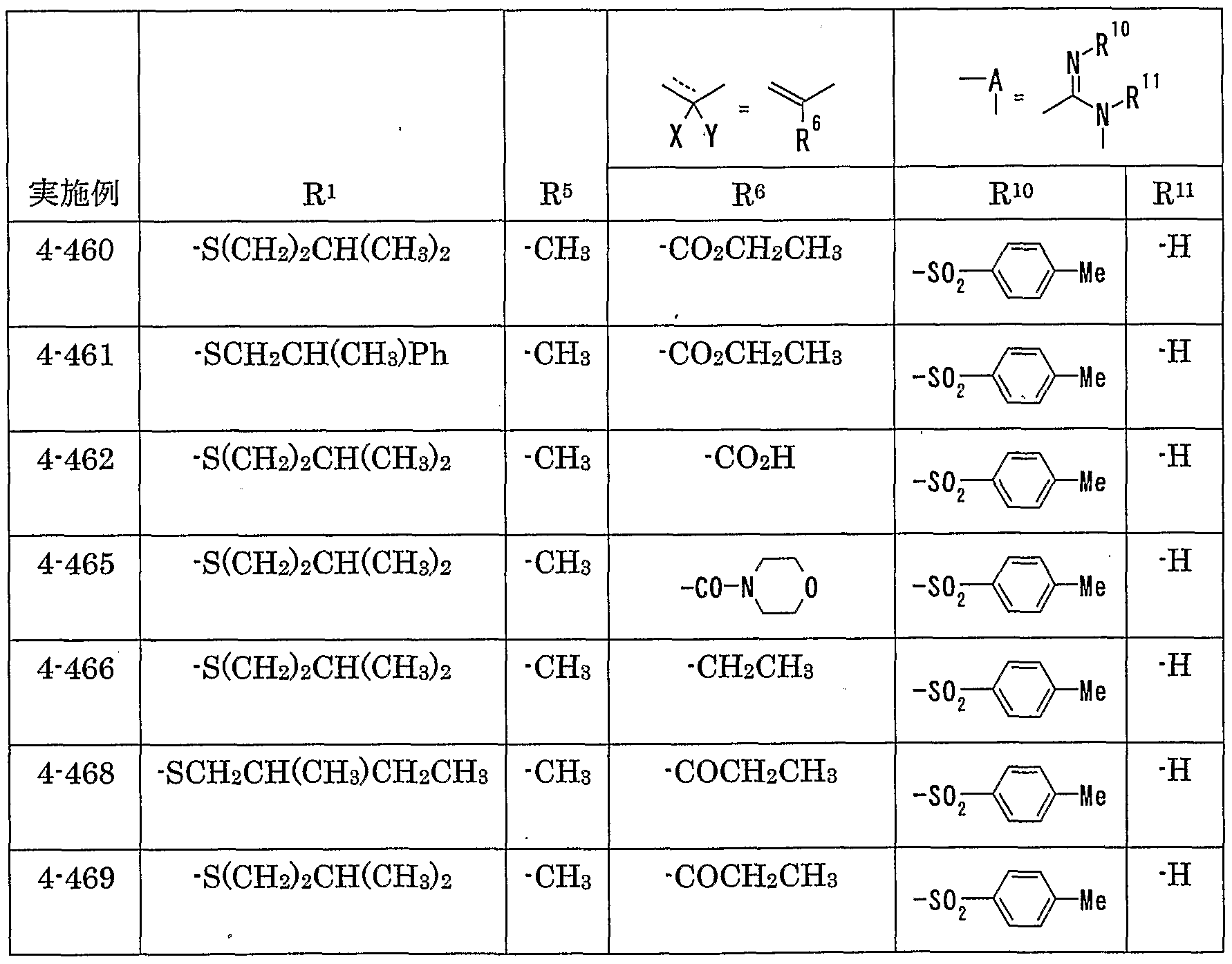
- MMP matrix meta
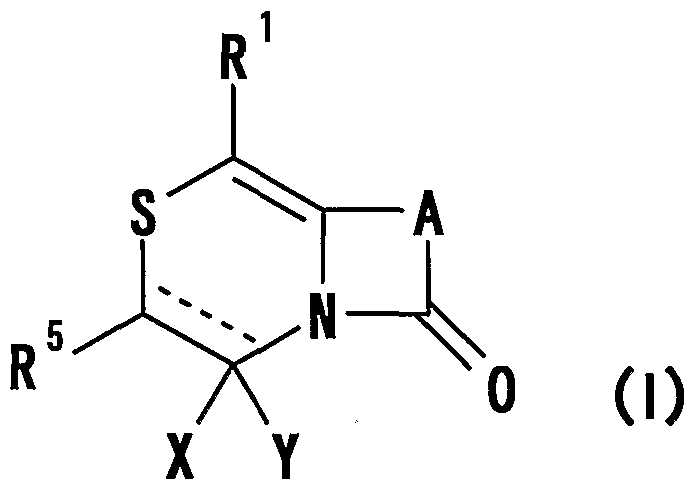
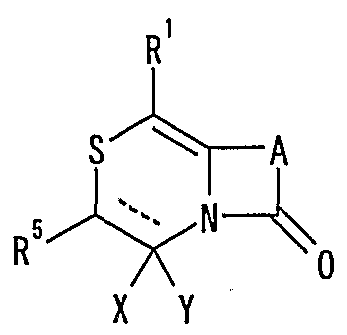
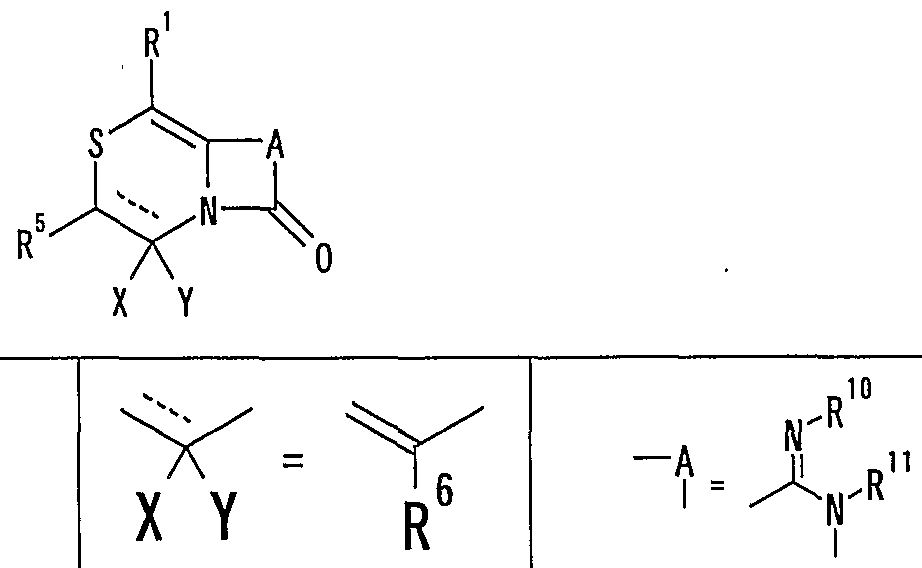
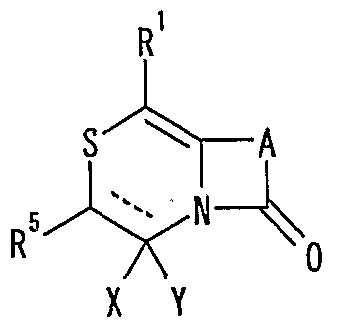
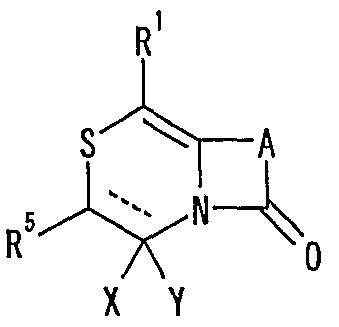
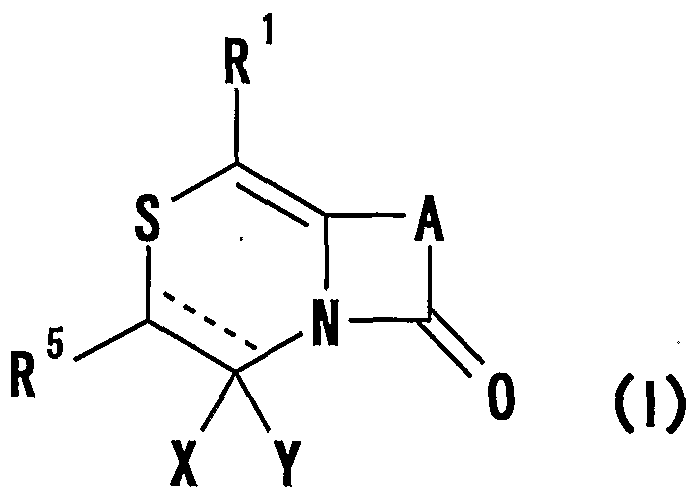
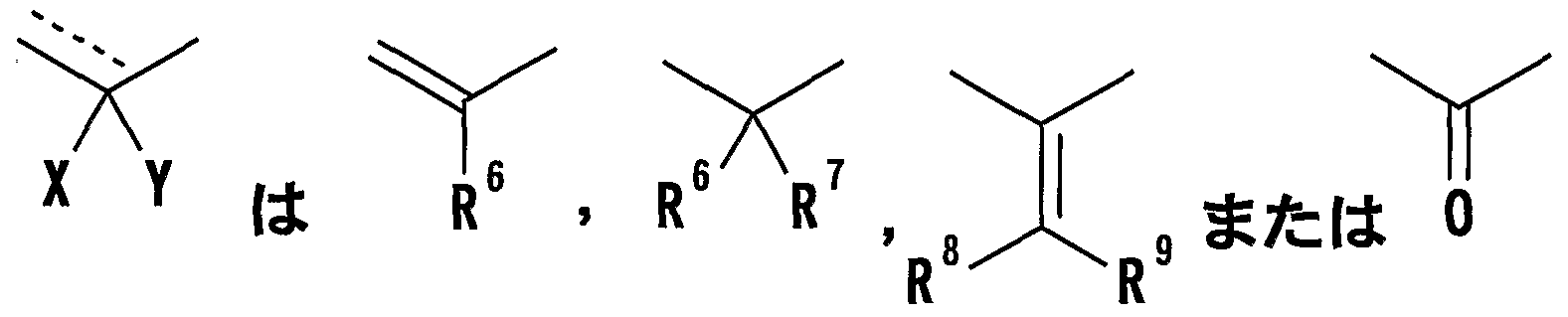
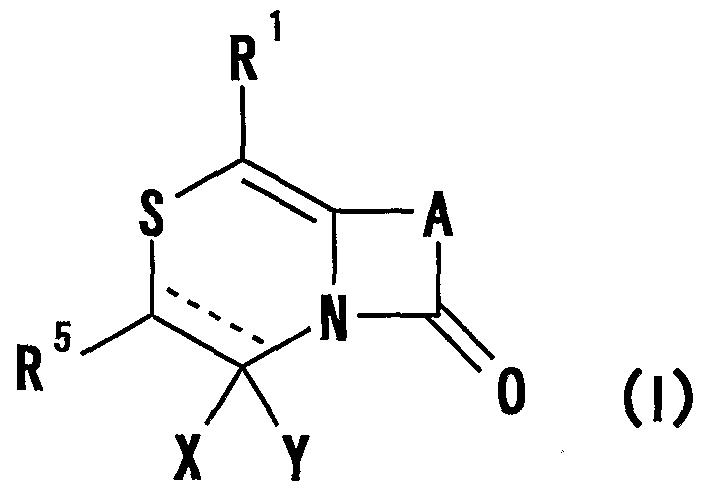
- R 1 represents one (S) n —; 2 or one NR 3 R 4 , n represents an integer of 0 to 2
- R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group or substituted
- R 3 and R 4 are the same or different and are each a hydrogen atom, an optionally substituted hydrocarbon group, an optionally substituted heterocyclic group, or A nitrogen-containing heterocyclic ring formed together with a nitrogen atom
- R 5 is a hydrogen atom, and an optionally substituted hydrocarbon
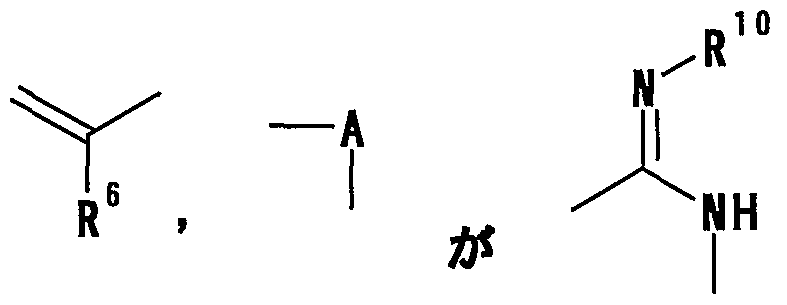
- R 6 represents a hydrogen atom, a hydrocarbon group which may be substituted, a cyano group, an acyl group, a propyloxyl group which may be esterified or amidated, or a heterocyclic group which may be substituted;
- R 7 is an optionally substituted hydroxyl group;
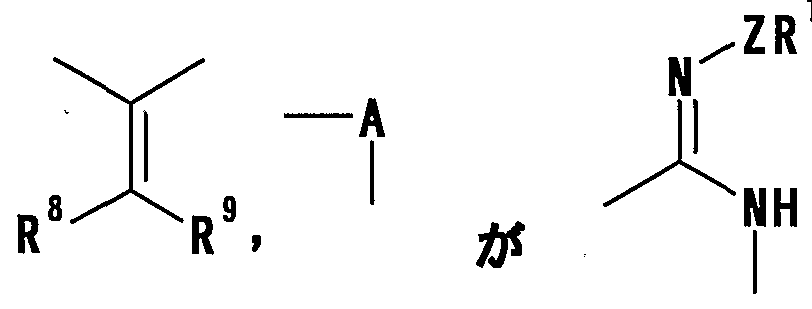
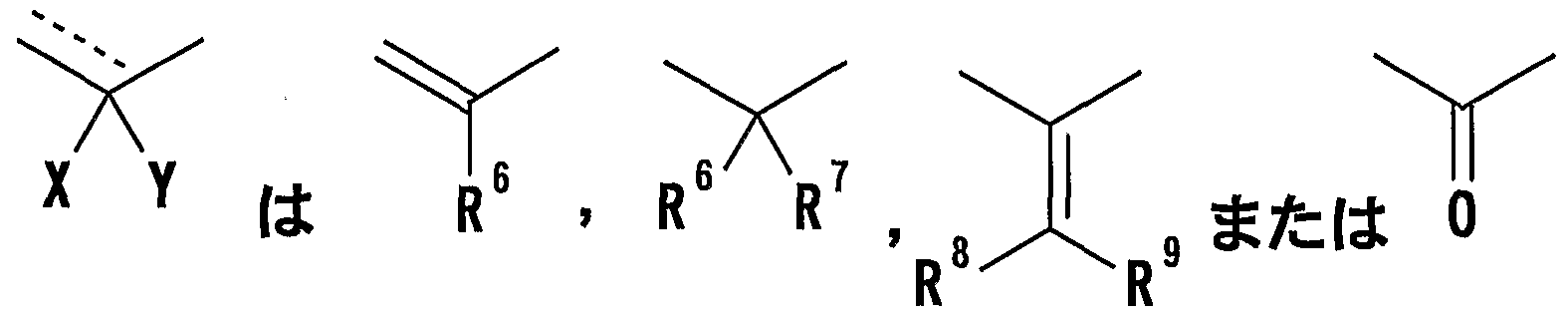
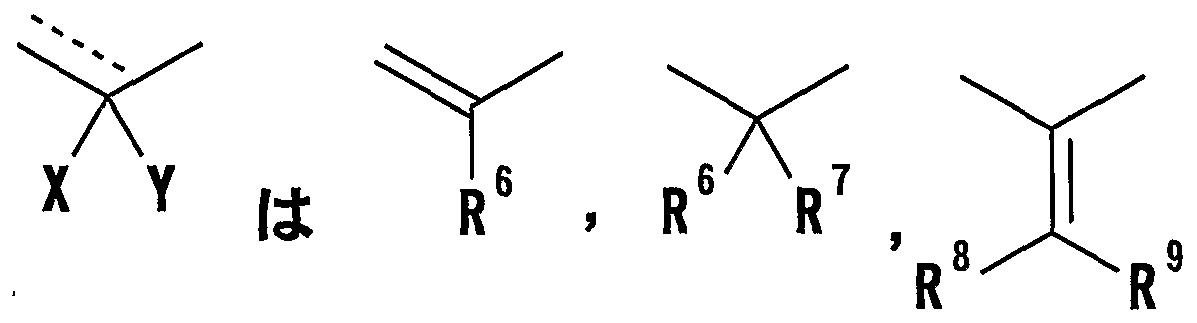
- R 8 and R 9 are each a hydrogen atom or an optionally substituted hydrocarbon group (R 5 and R 6 , R 5 and R 8 or R 8 And R 9 may be taken together to form an optionally substituted cyclic hydrocarbon or heterocyclic ring),
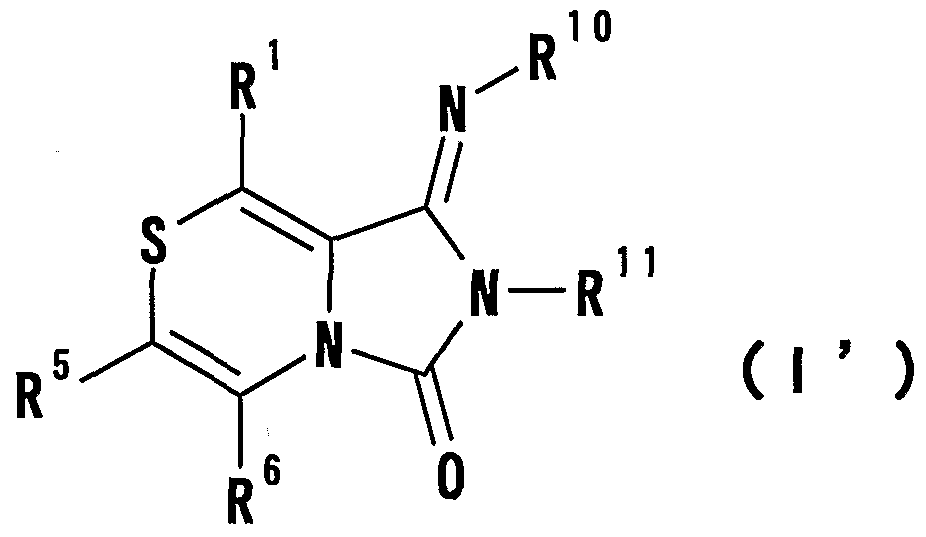
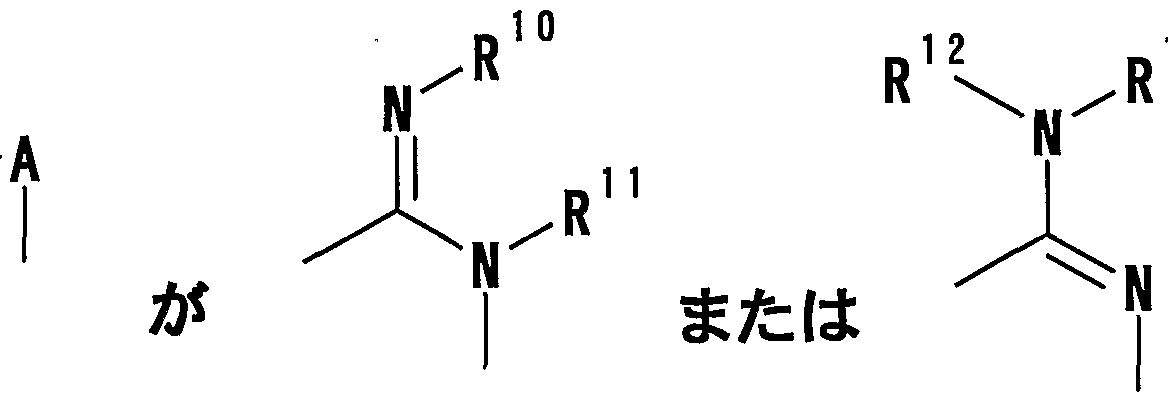
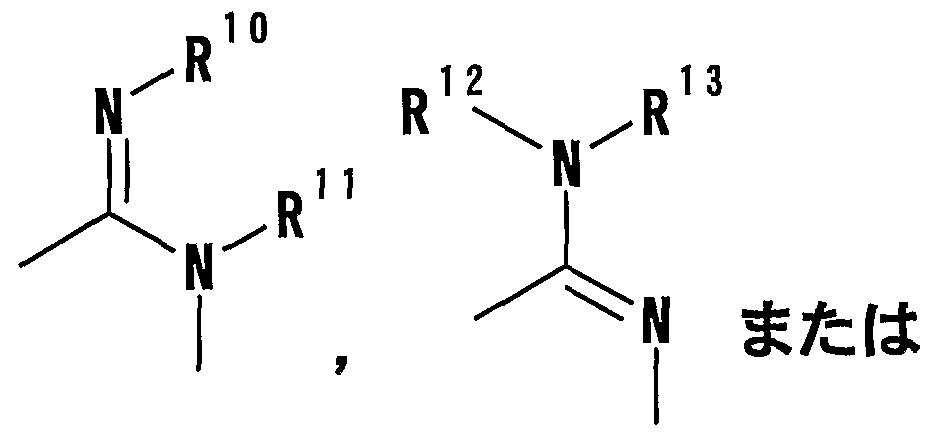
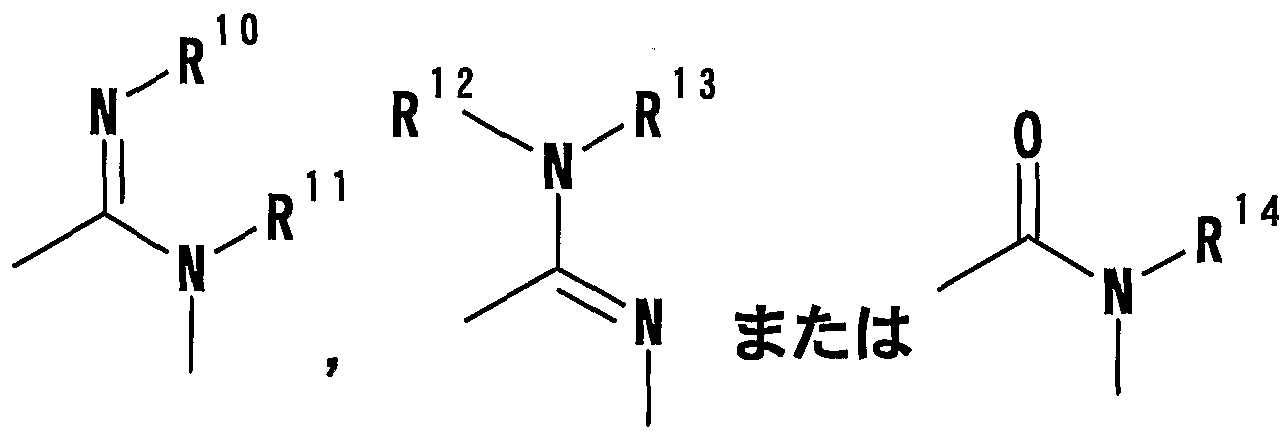
- R 1 Is a hydrogen atom, -ZR 15 (Z is _S0 2 —, SO—, _C ⁇ NR 18 S ⁇ 2 — (R 18 is 6 alkyl), — CONR 19 — (R 19 is —6 alkyl R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group) or — P ( ⁇ ) R 16 R 17 (R 16 and R 17 Represents an optionally substituted hydrocarbon group, an optionally substituted hydroxyl group or an optionally substituted amino group), and R 11 represents a hydrogen atom or an optionally substituted hydrocarbon group ( R 1Q and R 11 may together form a nitrogen-containing heterocyclic ring which may be substituted), R 12 and R 13 are each a hydrogen atom or an optionally substituted hydrocarbon group, or Together with the nitrogen atom to which it is attached forms a nitrogen-containing heterocycle, R 14 is a hydrogen atom, Or a hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described
- R 14 represents an optionally substituted hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described above).
- the present invention has been completed based on these findings. That is, the present invention
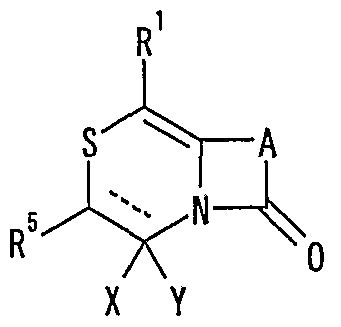
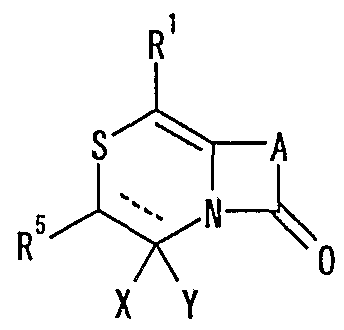
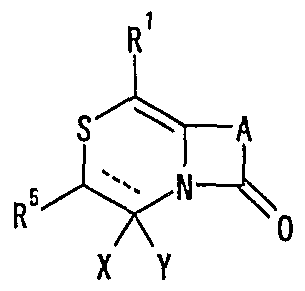
- R 1 represents one (S) n —R 2 or —NR 3 R 4 , n represents an integer of 0 to 2, R 2 represents a hydrogen atom, or a substituted or unsubstituted hydrocarbon group.
- R 3 and R 4 may be the same or different and represent a hydrogen atom, a hydrocarbon group which may be substituted, a heterocyclic group which may be substituted or
- R 5 represents a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, an esterified or amidated nitrogen-containing heterocyclic ring formed together with a nitrogen atom A xyl group or an optionally substituted heterocyclic group,
- R 6 represents a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, a carboxyl group which may be esterified or amidated, or a heterocyclic group which may be substituted
- 7 is an optionally substituted hydroxyl group
- R 8 and R 9 are each a hydrogen atom or an optionally substituted hydrocarbon group (R 5 and R 6 , R 5 and R 8 or R 8 R 9 may be taken together to form an optionally substituted cyclic hydrocarbon or heterocyclic ring),
- R 10 is a hydrogen atom
- _ZR 15 ( ⁇ one S0 2 -, -SO-, - CONR 18 SO 2 - (R 18 represents a CI- 6 alkyl), one CONR 19 - (R 19 is ( Or 6 represents an alkyl) or —CO—
- R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group) or — P (0)
- R 16 R 17 R 16 and R 17 each represent an optionally substituted hydrocarbon group, an optionally substituted hydroxyl group or an optionally substituted amino group
- R 11 represents a hydrogen atom or an optionally substituted hydrocarbon group.
- a hydrogen group (R 1 ° and R 11 may be taken together to form a nitrogen-containing heterocyclic ring which may be substituted);
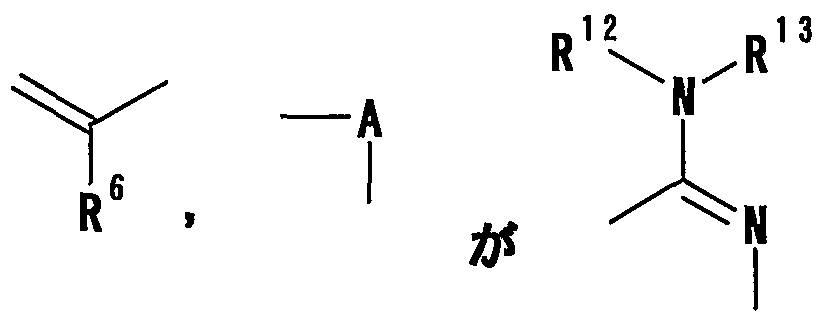
- R 12 and R 13 each represent a hydrogen atom or a carbon atom which may be substituted; hydrogen group, or a connexion nitrogen-containing heterocyclic ring such together with the nitrogen atom to which they are attached,
- R 14 is a hydrogen atom, Conversion are optionally substituted hydrocarbon group or also -.
- Z 15 (Z and R 15 are as defined above) shows the proviso, R 5 is a hydrogen atom, XY is
- R 14 represents an optionally substituted hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described above).
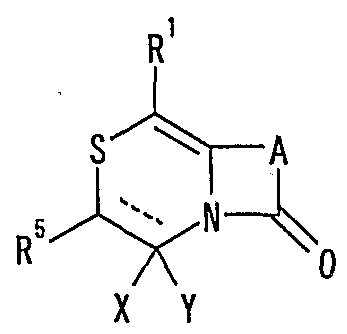
- R 1 is (S) n _R 2 or _NR 3 R 4 , n is an integer of 0 to 2, R 2 is a hydrogen atom, an optionally substituted hydrocarbon group or a substituted R 3 and R 4 may be the same or different and represent a hydrogen atom, an optionally substituted hydrocarbon group, an optionally substituted heterocyclic group, or a nitrogen atom to which they are bonded.
- R 5 is a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, a carboxyl group which may be esterified or amidated, or a substituted heterocyclic ring.
- a heterocyclic group which may be Is or
- R 6 represents a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, an esterified or amidated propyloxyl group or an optionally substituted heterocyclic group.
- R 7 is an optionally substituted hydroxyl group;
- R 8 and R 9 are each a hydrogen atom or an optionally substituted hydrocarbon group (R 5 and R 6 , R 5 and R 8 Or R 8 and R 9 may be taken together to form an optionally substituted cyclic hydrocarbon or heterocyclic ring),
- R 1 () is a hydrogen atom or _ZR 15 (Z - S0 2 - or - a CO-
- R 15 is a optionally substituted hydrocarbon group or an optionally substituted heterocyclic group
- R 11 represents a hydrogen atom or an optionally substituted hydrocarbon group
- R 12 and R 13 represent a hydrogen atom or an optionally substituted hydrocarbon group, respectively
- R 14 represents a hydrogen atom, an optionally substituted hydrocarbon group or one ZR 15 (Z and R 15 have the same meanings as described above)
- R 5 is a hydrogen atom,
- R 14 represents an optionally substituted hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described above). Or a salt thereof or a sulfoxide thereof;
- R 5 is an optionally substituted hydrocarbon group, a cyano group, an acyl group, a carboxyl group which may be esterified or amidated, or an optionally substituted heterocyclic group.
- R 5 is an optionally substituted hydrocarbon group, a cyano group, an acyl group, a carboxyl group which may be esterified or amidated, or an optionally substituted heterocyclic group.
- R 6 is a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group
- R 7 is an optionally substituted hydroxyl group or an optionally substituted heterocyclic group
- R 7 is an optionally substituted hydroxyl group
- R 8 and R 9 are each a hydrogen atom or a substituted
- R 10 is a hydrogen atom, one ZR 15 (Z one S0 2 -, One SO-, One C_ ⁇ _NR 18 S_ ⁇ 2 one (R 18 is (: 6 represents an alkyl), - CONR 19 - (R 19 ⁇ - a indicates to) or single CO- 6 alkyl, R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group) or - P (O) R 16 R 17 (R 16 and R 17 each represent an optionally substituted hydrocarbon group, an optionally substituted hydroxyl group or an optionally substituted amino group), and R 11 represents a hydrogen atom or a substituted R 1 Q and R 11 may be taken together to form a nitrogen-containing compound ring which may be substituted, and R 12 and R 13 are each a hydrogen atom or (1) a hydrocarbon group which may be substituted, or a nitrogen-containing heterocyclic ring together with a nitrogen atom to which they are bonded].
- R 1C1 represents a hydrogen atom or _ZR 15 (Z represents one S ⁇ 2 — or —C ⁇ one, and R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group. ), R 11 is a hydrogen atom or an optionally substituted hydrocarbon group, and R 12 and R 13 are each a hydrogen atom or an optionally substituted hydrocarbon group, or a nitrogen atom to which they are bonded. Together with a nitrogen-containing heterocycle].
- R 1Q is — S ⁇ 2 R 15 (R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group), and R 5 and R 6 are each substituted Or (8) a hydrocarbon group which may be substituted or an optionally substituted cyclic hydrocarbon or an optionally substituted heterocyclic ring which are formed together, and wherein R 11 is a hydrogen atom.
- R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group
- R 5 and R 6 are each substituted Or (8) a hydrocarbon group which may be substituted or an optionally substituted cyclic hydrocarbon or an optionally substituted heterocyclic ring which are formed together, and wherein R 11 is a hydrogen atom.
- R 1 is one SR 2 (R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group, or an optionally substituted heterocyclic group).
- R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group, or an optionally substituted heterocyclic group.
- Compound; (11) The compound according to the above (10), wherein R 2 is an optionally substituted hydrocarbon group;
- R 1 is one SR 2 (R 2 is an optionally substituted alkyl group having 1 to 8 carbon atoms, an alkenyl group optionally having 2 to 8 carbon atoms, A cycloalkyl group having 3 to 8 carbon atoms or an aralkyl group having 7 to 10 carbon atoms which may be substituted), wherein R 15 is an optionally substituted phenyl group, an optionally substituted benzyl group Or the optionally substituted phenyl group, the compound according to the above (9), wherein both R 5 and R 6 are methyl groups;
- R 1G is —COR 15 (R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group), and R 5 and R 6 are each substituted.
- a hydrocarbon group which may be optionally substituted or an optionally substituted cyclic hydrocarbon or an optionally substituted heterocyclic ring which are formed together, wherein R 11 is a hydrogen atom;
- a pharmaceutical composition comprising the compound (I) according to the above (1) or a salt thereof, a sulfoxide thereof or a prodrug thereof;
- composition according to the above (30) which is an agent for preventing or treating a disease caused by cartilage destruction; (35) the pharmaceutical composition according to the above (30), which is a matrix meta-oral proteinase gene expression inhibitor;
- a proteoglitin degradation inhibitor comprising a condensed imidazolidine derivative
- a cartilage matrix degrading enzyme production inhibitor comprising a condensed imidazolidine derivative
- a prophylactic / therapeutic agent for osteoarthritis comprising a condensed imidazolidine derivative
- a method for preventing or treating osteoarthritis which comprises administering to a mammal an effective amount of the compound (II) or a salt thereof or a sulfoxide or a prodrug thereof according to (1);
- (42) a method for preventing or treating a disease caused by cartilage destruction, which comprises administering an effective amount of the compound (I) or a salt thereof or a sulfoxide or a prodrug thereof according to the above (1) to a mammal;
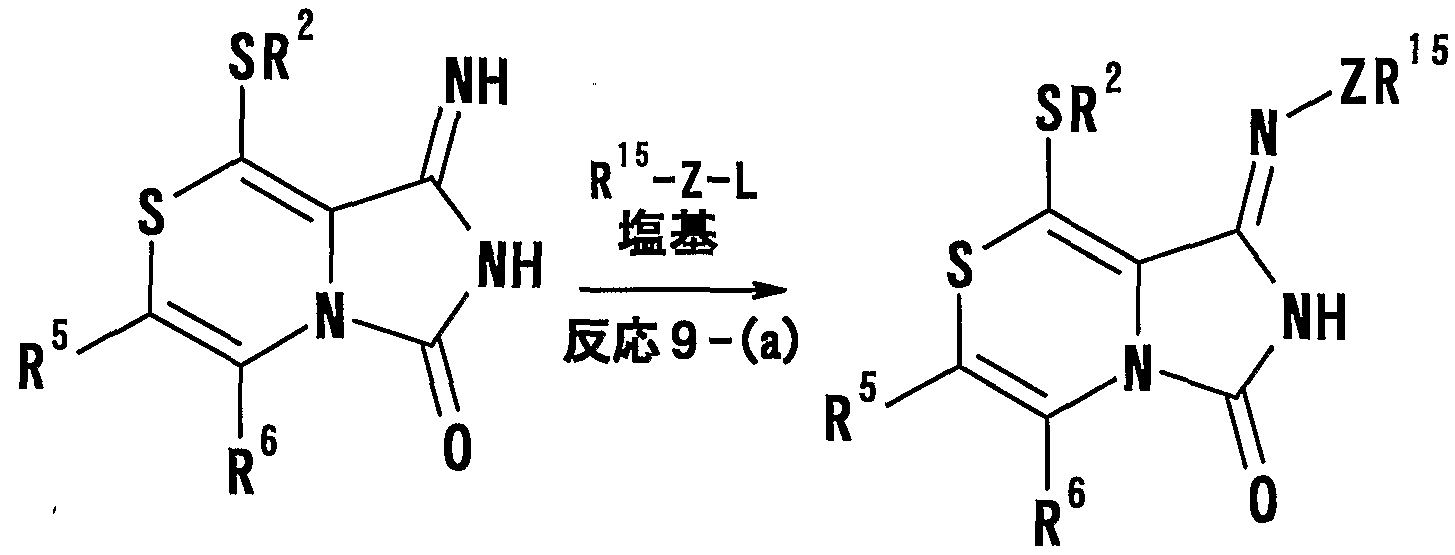
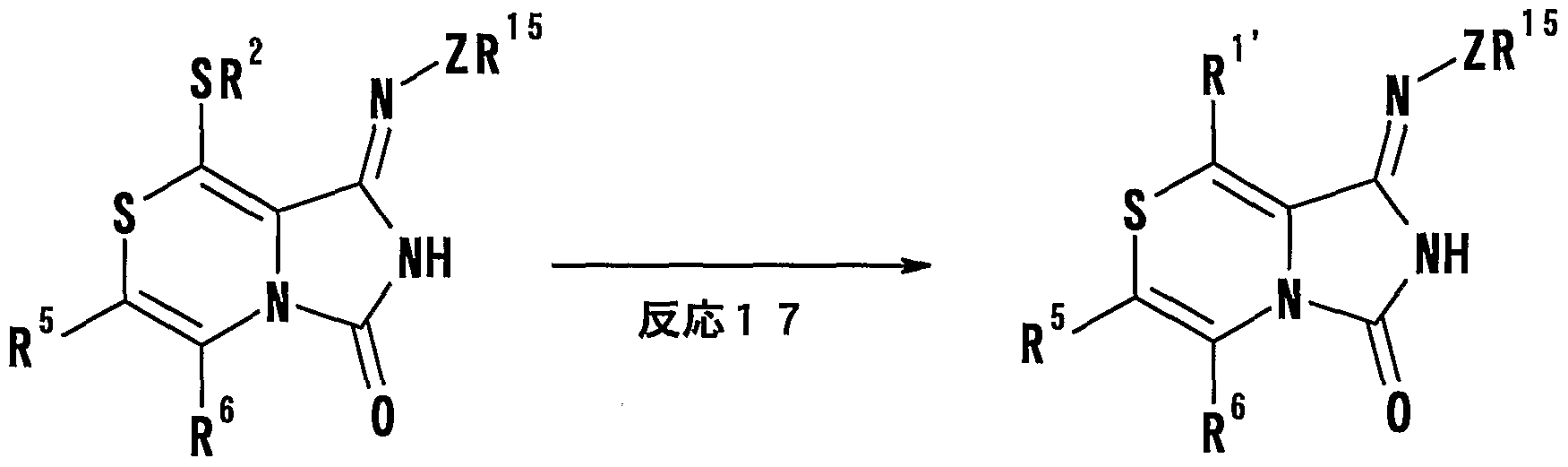
- [L is a leaving group, and R 5 and R 6 have the same meanings as described in the above (1)] or a salt thereof, and then subjected to a ring-forming reaction, and if desired, a hydrolysis reaction and a dehydration reaction.
- General formula characterized by conducting a reaction, an alkylation reaction, a substitution reaction and / or an oxidation reaction.
- R 1 represents — (S) n —R 2 or —NR 3 R 4 , n represents an integer of 0 to 2, R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group or R 3 and R 4 are the same or different hydrogen atoms, an optionally substituted hydrocarbon group, an optionally substituted heterocyclic group or an optionally substituted heterocyclic group, A nitrogen-containing heterocyclic ring formed together with a nitrogen atom is shown.
- Alkenyl group as the optionally substituted hydrocarbon group includes alkenyl groups having 2 to 20 carbon atoms (for example, vinyl, aryl, isoprobenyl, 1-probenyl, 1-butenyl, 2-butenyl, 3 -Butenyl, 1,3-butenyl, 1-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-1-propenyl, 2_methyl-2-propenyl 1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1,3-pentenyl, 2,4-pentenyl, 1,4-pentagenenyl, 1-methyl-1-butenyl, 2-methyl 1-butenyl, 3-methyl-1-butenyl, 1_methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl, 1-methyl-3-butenyl, 2-methyl 3-butenyl, 3-methyl-3-butenyl, 1-ethyl-1
- alkynyl group as the optionally substituted hydrocarbon group include an alkynyl group having 2 to 20 carbon atoms (eg, ethynyl, 1-propynyl, 2-propynyl,
- Examples of the "reyl group” as the optionally substituted hydrocarbon group include an aryl group having 6 to 10 carbon atoms (eg, phenyl, 0-tolyl, m-tolyl, p-tolyl, 2-,
- cycloalkyl group examples include cycloalkyl groups having 3 to 8 carbon atoms (eg, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, etc.). ). These may further be condensed with an aryl group such as a phenyl group, and examples thereof include indanyl, benzocyclohexyl, benzocycloheptyl, and benzocyclooctyl. Of these, preferred are cyclohexyl, cycloheptyl, cyclohexyl, indanyl, benzocycloheptyl and the like.
- This cycloalkyl group is bridged through a linear atom chain having 1 or 2 carbon atoms to form bicyclo [2.2.1] heptyl, bicyclo [2.2.2] octyl, bicyclo [ 3.2.1 octyl, bicyclo [3.3.1] nonyl, bicyclo [3.2.23 nonyl, etc. (preferably a cyclo group having a bridge through a linear atom chain having 1 or 2 carbon atoms) Hexyl, and more preferably bicyclo [2.2.1] heptyl) may form a crosslinked cyclic hydrocarbon residue. It also forms an adamantyl group. May be formed.
- cycloalkenyl group examples include a cycloalkenyl group having 3 to 8 carbon atoms (for example, 2-cyclopropenyl, 2-cyclobutenyl, 2-cyclopentenyl, 3-cyclopentenyl). Pentenyl, 2-cyclohexenyl, 3-cyclohexenyl, 2-cycloheptenyl, 3-cycloheptenyl, 4-cycloheptenyl; k 2-cyclooctenyl, 3-cyclooctenyl, 4-cyclooctenyl.
- aryl group such as a phenyl group, for example, indenyl, benzocyclohexenyl, benzocycloheptenyl, benzocyclooctenyl and the like.
- aryl group such as a phenyl group
- indenyl benzocyclohexenyl
- benzocycloheptenyl benzocyclooctenyl
- benzocyclooctenyl and the like.
- Hexenyl, 2-cycloheptenyl, cyclooctyl, indenyl and the like can be mentioned.
- substituent of the “optionally substituted hydrocarbon group” for R 2 include (1) a nitro group, (2) an oxo group, and (3) a halogen atom (eg, fluorine, chlorine, bromine, (4) cyano group, (5) methylene group, (6) optionally substituted lower alkyl group, (7) optionally substituted lower alkenyl group, (8) substituted (9) optionally substituted aralkyl group, (10) optionally substituted aralkyl group, (11) optionally substituted cycloalkyl group, (12) lower haloalkyl Group, (13) olepoxyl group which may be esterified or amidified, (14) thiol bamoyl group, (15) acyl group, (16) amidyl group, (17) hydroxyl group which may be substituted (18) an optionally substituted sulfanyl group, (19) a substituted An optionally substituted alkylsulfinyl group, (20) an optionally substituted aryl
- the substituent of the "optionally substituted hydrocarbon” and the "lower alkyl group” of the “optionally substituted lower alkyl group” for R 2 are, for example, methyl, ethyl, ⁇ _propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl And C1 to C4 alkyl groups.
- Examples of the substituent which the “lower alkyl group” may have include, for example, (1) a nitro group, (2) an oxo group, (3) a halogen atom (eg, fluorine, chlorine, bromine, etc.), (4) ) A cyano group, (5) a methylene group, (6) a lower alkyl group (for example, having 1 to 4 carbon atoms such as methyl, ethyl, n-propyl, isopropyl, butyl, isoptyl, sec-butyl, tert-butyl, etc.) (7) a lower alkenyl group (for example, bier, allyl, isopropyl, 1-butenyl, 2-butenyl, 3-methyl-2-butenyl, 3-methyl-3-butenyl, An alkenyl group having 2 to 5 carbon atoms such as 1-pentenyl and 2-pentenyl); (8) a lower alkynyl group (an alkynyl
- alkoxycarbonyl Amino e.g., methoxy Cal Poni Rua amino, ethoxy Cal Poni Rua amino, tert - Butoxycarponylamino, etc., aryloxylponylamino (for example, phenoxycarponylamino, naphthyloxycarponylamino, etc.), aralkyloxycarbonylamino (for example, benzyloxycarponylamino, phenethyloxycarponylamino, etc.) ), Alkylsulfonylamino (eg, methylsulfonylamino, ethylsulfonylamino, isopropylsulfonylamino, butylsulfonylamino, etc.), arylsulfonylamino (eg, benzenesulfonylamino, naphthylsulfony
- Examples of the “lower alkenyl group” of the “optionally substituted lower alkenyl group” as the substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, vinyl and allyl. ), Isoprobenyl, 1-butenyl, 2-butenyl, 3-methyl-2-butenyl, 3-methyl-3-butenyl, 1 pentenyl, 2-pentenyl and the like.
- the substituent which the “lower alkenyl group” may have include the “optionally substituted lower” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. The same number and the same as the substituents that the “alkyl group” may have.
- Examples of the “lower alkynyl group” of the “optionally substituted lower alkynyl group” as the substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, ethynyl, propargyl, 2- Examples thereof include alkynyl groups having 2 to 5 carbon atoms, such as lower alkynyl groups such as butynyl and 2-pentynyl. Examples of the substituent which the “lower alkynyl group” may have include a “optionally substituted lower alkyl group” as a substituent of the aforementioned “optionally substituted hydrocarbon” represented by R 2. And the same number and the same as the substituents that may be possessed.
- Examples of the “aryl group” of the “optionally substituted arylyl group” as the substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, phenylyl, 2_, 3-or 4 And aryl groups having 6 to 12 carbon atoms such as -tolyl, 1- or 2-naphthyl, 2-, 3- or 4-biphenyl.
- Examples of the substituent which the “aryl group” may have include a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents which the “optionally substituted lower alkyl group” may have.
- Examples of the “aralkyl group” of the “optionally substituted aralkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, benzyl, 2-phenethyl, 3-phenyl Examples thereof include aralkyl groups having 7 to 13 carbon atoms such as propyl, 1- or 2-naphthylmethyl, 2-, 3- or 4-biphenylmethyl.
- substituent which the “aralkyl group” may have include “optionally substituted lower alkyl” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2. And the same number and the same as the substituents which the group may have.
- Examples of the “cycloalkyl group” of the “optionally substituted cycloalkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, cyclopropyl, cyclobutyl, cyclopentyl, C3-C8 cycloalkyl groups such as cyclohexyl, cycloheptyl and cyclooctyl, bicyclo [2.2.1] heptyl, bicyclo [2.2.2] octyl, bicyclo [3.2.1] octyl , Bicyclo [3.3.1] Noel, picicyclo [3.2.2] Noel and adamantyl.
- Examples of the substituent which the “cycloalkyl group” may have include a “optionally substituted lower alkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents which may be possessed.
- lower haloalkyl group as a substituent of the “optionally substituted hydrocarbon” represented by R 2 , for example, fluoromethyl, difluoromethyl, trifluoromethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-Trifluoroethyl, 1,1,2,2-Tetrafluoroethyl, 1,1,2,2,2-pentafluorofluorethyl, 3-Fluoro mouth pill, 3,3-Diflu Trifluoropropyl, 3,3,3-trifluoropropyl, 2,2,3,3,3-pentafluoropropyl, 4-fluorobutyl, 4,4-difluorobutyl, 4,4,4-trifluorobutyl, 3,3,4,4,4-pentyl butyl, 5-fluoropentyl, 5,5-difluoropentyl, 5,5,5-trifluoropentyl, 4,4,5,5,5-
- alkoxycarbonyl group examples include, for example, those having 2 to 2 carbon atoms such as methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, tert-butoxycarbonyl, hexoxycarbonyl, dodecyloxycarbonyl, hexadecyoxycarbonyl and the like. 18 alkoxycarbonyl groups.
- the “alkoxy group” may have a substituent, and the substituent may be a “substituted hydrocarbon” represented by R 2 described above. The same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
- alkenyloxycarbonyl group examples include those having 2 carbon atoms such as allyloxycarbonyl, octa-2,6-genyloxycarbonyl, dodeforce-2,6,10-trienyloxycarbonyl. To 18 alkenyloxycarbonyl groups.
- the “alkenyloxycarbonyl group” may have a substituent, and the substituent may be a “substituted hydrocarbon” represented by R 2 described above as a substituent of “optionally substituted hydrocarbon”. And the same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
- aryloxycarbonyl group for example, phenoxycarbonyl, 1-naphthoxycarponyl, 21-naphthoxycarponyl and the like can be mentioned.
- the “aryloxycarbonyl group” may have a substituent, and the substituent may be a “substituted hydrocarbon” as a substituent of the “optionally substituted hydrocarbon” represented by R 2. And the same number and the same as the substituents which the lower alkyl group may have.
- aralkyloxycarbonyl group examples include aralkyloxycarbonyl groups such as benzyloxycarbonyl, phenethyloxycarbonyl, and 3-phenylpropoxycarbonyl.
- the “aralkyloxycarbonyl group” may have a substituent, and the substituent is the “substituted or substituted” represented by R 2 described above.
- the same number and the same as the substituents which the “optionally substituted lower alkyl group” may have as a substituent of the “optionally substituted hydrocarbon”.
- substituent of the “N-monosubstituted carpamoyl group” examples include lower alkyl (eg, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl, etc.), lower alkenyl (eg, Bier, Aryl, isopropenyl, propenyl, butenyl, pentenyl, hexenyl, etc., cycloalkyl (eg, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, etc.), aryl (eg, phenyl) , 1-naphthyl, 2-naphthyl), aralkyl (eg, benzyl, phenethyl, etc.), heterocyclic group (for example, “substituted as a substituent of“ optionally substituted hydro
- Lower alkyl, lower alkenyl, cycloalkyl, ⁇ Li - Le, Ararukiru, heterocyclic group may have a substituent, examples of the substituent group, represented by R 2 "which may be substituted As the substituent for the "good hydrocarbon", the same number and the same substituents as the substituent which the "optionally substituted lower alkyl group” may have.
- the substituent of the “N, N-disubstituent rubamoyl group” means a disubstituted group having two substituents on a nitrogen atom, and is the same as the substituent in the above “N-monosubstituent rubamoyl”.
- two substituents may be combined with a nitrogen atom to form a cyclic amino.
- examples of the cyclic amino group rubamoyl include N-pyrrolidylcarbonyl, N-imidazolylcarbonyl, and N-imidazolylcarbonyl.
- acyl group as a substituent of the “optionally substituted hydrocarbon” represented by R 2 , a hydrogen atom or a substituent having one of the above-mentioned monosubstituent rubamoyl groups on the nitrogen atom And carbonyl, such as a lower alkanoyl group such as formyl group, acetyl and propionyl, or an aroyl group such as benzoyl and naphthyl.
- an optionally substituted lower alkyl group As the substituent of the ⁇ optionally substituted hydroxyl group '' as the substituent of the ⁇ optionally substituted hydrocarbon '' represented by R 2 , an optionally substituted lower alkyl group, Lower alkenyl group which may be substituted, lower alkynyl group which may be substituted, aryl group which may be substituted, aralkyl group which may be substituted, cycloalkyl group which may be substituted, esterification Or an amidated propyloxyl group, an optionally substituted alkylsulfanylthiocarbonyl group, an optionally substituted aralkylsulfanylthiocarbonyl group, an optionally substituted alkylsulfonyl group, An arylsulfonyl group which may be substituted, an optionally substituted N-imidazolylthiol haponyl, an optionally substituted N-morpholylthio haponyl, a
- the “substituted hydrocarbon” represented by R 2 may be a “substituted hydrocarbon”.
- esterified or amidated carboxyl group refers to the optionally esterified or amidated lipoxyl as a substituent of the aforementioned “optionally substituted hydrocarbon” represented by R 2.
- alkylsulfanylthiocarponyl group include methylsulfanylthiocarponyl, ethylsulfanylthiocarponyl, tert-butoxysulfanylthiocarponyl, and the like.
- Examples of the "good aralkylsulfanylthiocarbonyl group” include benzylsulfanylthiocarbonyloxy and the like, and the "optionally substituted alkylsulfonyl group” include methylsulfonyl, ethylsulfonyl, isopropylsulfonyl, Examples of the “optionally substituted arylsulfonyl group” include benzenesulfonyl, p-toluenesulfonyl, m-toluenesulfonyl, o-toluenesulfonyl and the like.
- optionally substituted alkylsulfanylthiocarbonyl group includes, for example, the optionally substituted hydrocarbon represented by R 2 above.
- R 2 the optionally substituted hydrocarbon represented by R 2 above.
- substituents which the “lower alkyl group which may be substituted” may have. Examples of the substituent which may be possessed by the ⁇ optionally substituted N-imidazolylthiol compound '' and the ⁇ optionally substituted N-morpholylthio compound '' are those represented by R 2 described above.
- substituent of the "optionally substituted hydrocarbon” the same number and the same substituents as the “optionally substituted lower alkyl group” may have.
- hydroxyl protecting group examples include 2-tetrahydropyranyl, trimethylsilyl, triethylsilyl, tert-butyldiphenylsilyl and the like.
- Examples of the “optionally substituted sulfanyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, a sulfanyl group, methylsulfanyl, methysulfanyl, isopropylsulfanyl, tert- Examples thereof include an alkylsulfanyl group such as butylsulfanyl, an arylsulfanyl group such as phenylsulfanyl, and an aralkylsulfanyl group such as benzylsulfanyl.
- substituents which the “optionally substituted sulfanyl group” may have include “substituted or substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2.
- substituents which the “lower alkyl group which may be optionally present” may have.
- Examples of the “optionally substituted alkylsulfinyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, methylsulfinyl, ethylsulfinyl, isopropylsulfinyl and the like.
- Examples of the substituent which the “optionally substituted alkylsulfinyl group” may have include “substituted or substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2. The same number and the same as the substituents which the “lower alkyl group which may be optionally present” may have.
- Examples of the “optionally substituted arylsulfinyl group” as a substituent of the “optionally substituted hydrocarbon” for R 2 include, for example, benzenesulfinyl, toluenesulfinyl and the like.
- the "arylsulf optionally substituted” As has also been have substituents Iniru group ", it said; have the” optionally substituted lower alkyl group "as the substituent of the" hydrocarbon which may be substituted "represented by 2 And the same number and the same as the substituents which may be included.
- Examples of the “optionally substituted alkylsulfonyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, methylsulfonyl, ethylsulfonyl, isopropylsulfonyl, butylsulfonyl and the like. Is mentioned.
- Examples of the substituent which the “optionally substituted alkylsulfonyl group” may have include “substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2. The same numbers and the same as the substituents which the lower alkyl group which may be optionally possessed "may have.
- Examples of the "optionally substituted arylsulfonyl group" as a substituent of the "optionally substituted hydrocarbon” represented by R 2 include, for example, benzenesulfonyl, toluenesulfonyl, naphthylsulfonyl and the like. .
- Examples of the substituent which the “optionally substituted arylsulfonyl group” may have include a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. The same number and the same thing as the substituent which the "lower alkyl group which may be substituted" may have are mentioned.
- Examples of the “sulfonic acid group which may be esterified or amidated” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, a sulfonic acid group, methyl sulfonic acid, and sulfonic acid Ethyl, sulfonic acid amide, N-methylsulfonic acid amide and the like.
- amino or di-substituted amino with an amino group an alkyl group or an aralkyl group Groups, acylamino, alkoxyl propylonamino, aryloxycarbonylamino, aralkyloxycarbonylamino, alkylsulfonylamino, arylsulfonylamino and the like.
- Amino group j mono- or di-substituted with an alkyl group or an aralkyl group j includes, for example, methylamino, ethylamino, isopropylamino, dimethylamino, methylamino, ethylmethylamino, benzylamino, benzylmethylamino and the like.
- ⁇ Can be.
- the "amino group mono- or di-substituted by an alkyl group or an aralkyl group” May have a substituent, and the substituent may be a ⁇ optionally substituted lower alkyl group '' as a substituent of the ⁇ optionally substituted hydrocarbon '' represented by R 2 above. And the same number and the same as the substituents that may be possessed.
- Acylamino includes, for example, formylamino, acetylamino, propionylamino, butyrylamino, isoptyrylamino, valerylamino, isovaleramino, pivaloylamino, benzoylamino, phenacylamino and the like.
- the “acylamino” may have a substituent, and the substituent may be a “optionally substituted lower” as a substituent of the “optionally substituted hydrocarbon” shown in the above 2 The same number and the same as the substituents that the “alkyl group” may have.
- the “alkoxyl ponylamino” includes, for example, methoxycarbonylamino, ethoxycarbonylamino, tert-butoxycarponylamino and the like.
- the “alkoxy compound / amino” may have a substituent, and the substituent may be a substituent of the “optionally substituted hydrocarbon” represented by R 2 described above .
- the same number and the same as those of the substituent which the “optionally substituted lower alkyl group” may have are exemplified.
- aryloxycarbonylamino includes, for example, phenoxycarbonylamino, naphthyloxycarbonylamino and the like.
- the “aryloxycarbonylamino” may have a substituent, and the substituent may be a “optionally substituted hydrocarbon” represented by R 2 described above. The same number and the same as the substituents which the "lower alkyl group which may be substituted” may have.
- Alkyloxycarbonylamino includes, for example, benzyloxycarponylamino, phenethyloxycarbonylamino and the like.
- the “aralkyloxycarbonylamino” may have a substituent, and the substituent may be a “substituted hydrocarbon” as a substituent of the “optionally substituted hydrocarbon” for R 2.
- the same numbers and the same as the substituents that the “lower alkyl group which may be substituted” may have.
- Alkylsulfonylamino includes, for example, methylsulfonylamino, ethylsulfonylamino, isopropylsulfonylamino, butylsulfonylamino and the like.
- the “alkylsulfonylamino” may have a substituent, and the substituent may be a “substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” represented by R 2. The same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
- arylsulfonylamino includes, for example, benzenesulfonylamino, naphthylsulfonylamino and the like.
- the “arylsulfonylamino” may have a substituent, and the substituent may be a “optionally substituted hydrocarbon” represented by R 2 described above. The same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
- Examples of the “optionally substituted heterocyclic group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, an aliphatic monocyclic nitrogen-containing heterocyclic group and an aromatic monocyclic group. Examples include a cyclic nitrogen-containing heterocyclic group, a condensed nitrogen-containing heterocyclic group, an oxygen-containing monocyclic or condensed heterocyclic group, and a sulfur-containing monocyclic or condensed heterocyclic group.
- Aliphatic monocyclic nitrogen-containing heterocyclic group refers to, for example, aziridinyl, azetidinyl, pyrrolidinyl, imidazolidinyl, pyrazolidinyl, oxazolidinyl, isoxazolidinyl, thiazolidinyl, isothiazolidinyl, piperidyl, piperazyl, pyrazolyl, morpholinyl, morpholinyl Oxazinyl, thiazinyl, azepinyl, diazepinyl, oxazepinyl, thiazepinyl and the like.
- the “aliphatic monocyclic nitrogen-containing heterocyclic group” may have a substituent, and the substituent is a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents which the "optionally substituted lower alkyl group” may have.
- Aromatic monocyclic nitrogen-containing heterocyclic group means, for example, pyrrolyl, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, triazolyl, oxadiazolyl, thiadiazolyl, tetrazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazinyl, pyrazinyl Is mentioned.
- the “aromatic monocyclic nitrogen-containing heterocyclic group” may have a substituent, and the substituent may be a “optionally substituted hydrocarbon” represented by R 2 described above. As the substituent, the same number and the same as those of the "optionally substituted lower alkyl group" may be mentioned. You.
- “Fused nitrogen-containing heterocyclic group” is, for example, benzopyrrolidinyl, benzimidazolidinyl, benzopyrazolidinyl, benzoxazolidinyl, benzoisoxazolidinyl, benzothiazolidinyl, benzoisothiazolidinyl Benzopiperidyl, benzopiperidyl, benzopyrazinyl, benzomorpholinyl, benzothiomorpholyl, benzoxazinyl, benzothiazinyl, benzozepinyl, benzodiazepinyl, benzoxazepinyl, benzothiazepinel, indolyl, isoindolyl, 1H-indazolyl , Benzimidazolyl, benzoxazolyl, benzoisoxazolyl, benzothiazolyl, benzoisothiazolyl, m-benzotriazolyl, benzoxoxadiazolyl
- the “condensed nitrogen-containing heterocyclic group” may have a substituent, and the substituent may be a “substituted hydrocarbon” represented by R 2 described above.
- R 2 substituted hydrocarbon
- Oxygen-containing monocyclic or fused heterocyclic group means, for example, oxilanyl, oxenyl, oxolanyl, dioxolanyl, furyl, pyranyl, tetrahydroviranyl, dioxanyl, benzofuranyl, isobenzofuranyl, benzopyrael, isobenzopyranyl, benzol Dioxanyl, 7-oxabicyclo [2.2.1] heptyl), 9-oxabicyclo [3.3.1] nonyl, and the like.
- the “oxygen-containing monocyclic or condensed heterocyclic group” may have a substituent, and the substituent may be any of the above-mentioned “optionally substituted hydrocarbon” represented by R 2 .
- substituent “substituted The same numbers and the same as the substituents that the “lower alkyl group which may be substituted” may have.
- the “sulfur-containing monocyclic or condensed heterocyclic group” includes, for example, phenyl, thioxolanyl, tetrahydrothiopyrael, dithianil, benzothenyl and the like.
- the “sulfur-containing monocyclic or condensed heterocyclic group” may have a substituent, and the substituent may be a substituent of the “optionally substituted hydrocarbon” represented by R 2 described above.
- R 2 the same number and the same as the substituent which the "lower alkyl group which may be substituted” may have.
- alkylenedioxy group as a substituent of the "optionally substituted hydrocarbon” represented by, for example, methylenedioxy, ethylenedioxy and the like can be mentioned.
- Examples of the “optionally substituted cycloalkenyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include a cycloalkenyl group having 3 to 8 carbon atoms (eg, 2-cyclopropenyl, 2-cyclobutenyl, 2-cyclopentenyl, 2-cyclohexenyl, 3-cyclohexenyl, 2-cycloheptenyl, 3-cycloheptenyl, 2-cyclooctenyl, 4-cyclooctenyl and the like.
- Examples of the substituent which the “cycloalkenyl group” may have include a “optionally substituted lower alkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents that may be possessed.
- hydrocarbon group as the “optionally substituted hydrocarbon group” represented by R 3 and R 4 examples include the hydrocarbon as the “optionally substituted hydrocarbon group” represented by the aforementioned R 2 And the same.
- Preferable examples include an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like.
- the ⁇ optionally substituted heterocyclic group '' represented by R 3 or R 4 may be ⁇ optionally substituted '' as a substituent of the ⁇ optionally substituted hydrocarbon '' represented by R 2 And the same as the “good complex ring group”.
- Examples of the nitrogen-containing heterocyclic ring as “an optionally substituted nitrogen-containing heterocyclic ring represented by R 3 and R 4 together with the nitrogen atom to which they are bonded” include, in addition to a carbon atom, May contain 1 to 4 heteroatoms selected from atoms, sulfur atoms, nitrogen atoms, etc. 3- to 8-membered monocyclic heterocycles or condensed bicyclic or tricyclic heterocycles And fused heterocyclic groups. Among them, a preferable example is a 5- to 7-membered nitrogen-containing heterocyclic ring. Particularly preferred examples include azepier, piperidyl, piperazyl, N-methylbiperazyl, pyrrolidyl, morpholyl, and the like.
- R 3, R 4 are both nitrogen-containing heterocyclic ring which may be substituted, such connexion represent together with the nitrogen atom to which they are attached", "is substituted represented by R 2 And the same number as the substituents of the "optionally substituted hydrocarbon group".
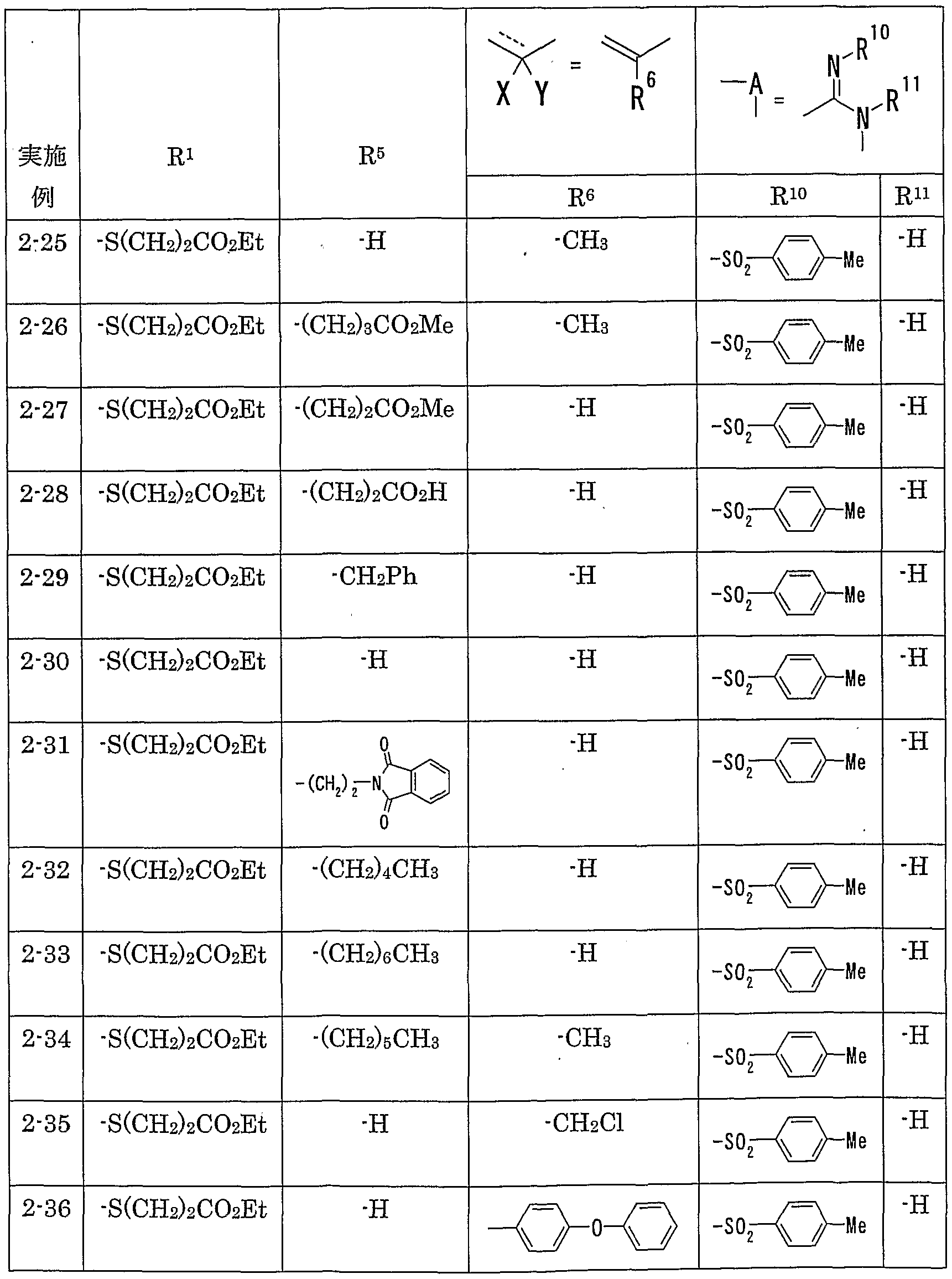
- R 1 is preferably —SR 2 (R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group), and in particular, R 2 is substituted. Those which are good hydrocarbon groups are preferred.
- R 2 is an optionally substituted alkyl group (optionally substituted alkyl group having 1 to 8 carbon atoms) and an optionally substituted alkenyl group (optionally substituted An alkenyl group having 2 or 8 carbon atoms, an aralkyl group which may be substituted (an aralkyl group having 7 to 10 carbon atoms which may be substituted), a cycloalkyl group which may be substituted (substituted) And a cycloalkyl group having 3 to 8 carbon atoms which may be substituted.
- R 2 is preferably an alkyl group having 1 to 8 carbon atoms which may be substituted, and more preferably an alkyl group having 4 to 8 carbon atoms which may be substituted by a halogen atom. Are preferred.
- the hydrocarbon group as the ⁇ optionally substituted hydrocarbon group '' represented by R 5 is the same as the hydrocarbon group as the ⁇ optionally substituted hydrocarbon group '' represented by the aforementioned R 2 Things.
- an alkyl group having 1 to 8 carbon atoms an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and a cycloalkyl having 3 to 8 carbon atoms
- the group is preferably a cycloalkenyl group having 3 to 8 carbon atoms.
- an alkyl group having 1 to 6 carbon atoms is preferable, and methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isobenzyl, 2-methylbutyl, hexyl, isobutyl Hexyl, 2-ethylbutyl, 1,1-dimethylbutyl, 2,2-dimethylbutyl, 3,3-dimethylbutyl, 2,2-dimethylpentyl, and 3,3-dimethylpentyl are preferred.
- acyl group represented by R 5 is the same as the “acyl group” as a substituent of the “optionally substituted hydrocarbon group” represented by R 2 .
- the “carboxyl group which may be esterified or amidated” represented by R 5 means “esterification or amide as a substituent of the aforementioned“ optionally substituted hydrocarbon group ”represented by R 2. And the like. Among them, an alkoxycarbonyl group having 2 to 18 carbon atoms such as methoxycarbonyl, ethoxycarbonyl, propoxyl-proponyl, tert-butoxycarbonyl, hexyloxyl-proponyl, dodecyloxycarbonyl, hexadecyloxycarbonyl and the like is preferable.
- the “optionally substituted heterocyclic group” represented by R 5 includes the same as the “optionally substituted heterocyclic group” represented by R 2 .
- the hydrocarbon group as the ⁇ optionally substituted hydrocarbon group '' represented by R 6 is the same as the hydrocarbon group as the ⁇ optionally substituted hydrocarbon group '' represented by the aforementioned R 2 Things.
- Preferable examples include an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Among them, alkyl groups having 1 to 6 carbon atoms, A phenyl group and a benzyl group are preferred. ,
- acyl group represented by R 6 is the same as the “acyl group” as a substituent of the aforementioned “optionally substituted hydrocarbon group” represented by R 2 .
- the ⁇ optionally esterified or amidated olepoxyl group '' represented by R 6 means ⁇ esterification or a hydrocarbon group which may be substituted '' represented by R 2 above. And the like.
- the cyclic hydrocarbon or heterocyclic ring formed by R 5 and R 6 together may contain, in addition to carbon atoms, one to three atoms selected from nitrogen, oxygen, sulfur and the like. Or an unsaturated 5- to 8-membered monocyclic carbocyclic or heterocyclic ring, or a bicyclic fused carbocyclic or heterocyclic ring containing these.
- cycloalkyl eg, cyclopentyl, cyclohexyl, cycloheptyl, etc.
- cycloalkenyl eg, cyclopentenyl, cyclohexenyl, cyclohexenyl, cycloheptenyl, etc.
- aromatic hydrocarbon ring eg, phenyl) , Naphthyl, etc.
- oxygen-containing ring for example, furyl, oxolanyl, tetrahydroviranyl, oxepinyl, benzofuranyl, benzopyranyl, etc.
- nitrogen-containing ring for example, pyrrolidyl, piperidyl, pyridyl, azepinyl, indolyl, quinolinyl, benzoxazepinyl, etc.
- Sulfur-containing rings eg, phenyl, tetrahydrothiopyranyl,
- Cyclic hydrocarbon or heterocyclic ring and R 5 and R 6 are you connexion formed such together may have a substituent, examples of the substituent group, be "substituted represented by R 2 The same number and the same as the substituents of the “good hydrocarbon group”.
- R 5 is preferably an optionally substituted hydrocarbon group, a cyano group, an acyl group, an optionally esterified or amidated propyloxyl group, or an optionally substituted heterocyclic group.
- An optionally substituted hydrocarbon group is preferred.
- alkyl groups having 1 to 12 carbon atoms alkenyl groups having 2 to 12 carbon atoms, alkynyl groups having 2 to 12 carbon atoms, aryl groups having 6 to 10 carbon atoms, and 7 carbon atoms.
- Preferred are an aralkyl group having 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, and a cycloalkenyl group having 3 to 8 carbon atoms.
- alkyl groups having 1 to 8 carbon atoms alkenyl groups having 2 to 8 carbon atoms, aryl groups having 6 to 10 carbon atoms, aralkyl groups having 7 to 10 carbon atoms, cycloalkyl groups having 3 to 8 carbon atoms And a cycloalkenyl group having 3 to 8 carbon atoms is preferred.
- a hydrocarbon group having 1 to 6 carbon atoms particularly an alkyl group having 1 to 6 carbon atoms is preferable, and methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, Preferred are isopentyl, 2-methylbutyl, hexyl, isohexyl, 2-ethylbutyl, 1,2-dimethylbutyl, 2,2-dimethylbutyl and 3,3-dimethylbutyl.
- R 6 is preferably an optionally substituted hydrocarbon group.
- an alkyl group having 1 to 8 carbon atoms an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like are preferable.
- a hydrocarbon group having 1 to 6 carbon atoms especially an alkyl group having 1 to 6 carbon atoms (methyl, ethyl, propyl, isopropyl, butyl, isoptyl, sec-butyl, tert-butyl, pentyl, isopentyl, 2 -Methylbutyl, hexyl, isohexyl, 2-ethylbutyl, 1,1-dimethylbutyl, 2,2-dimethylbutyl, 3,3-dimethylbutyl, etc., particularly methyl and ethyl) are preferred.
- a compound forming a benzene ring which may be substituted together with R 5 and R 6 or a cyclohexene ring which may be substituted may also be mentioned as a preferable example.
- Examples of the substituent of the ⁇ optionally substituted hydroxyl group '' represented by R 7 include an optionally substituted hydrocarbon group, an optionally substituted alkylsulfonyl group, and an optionally substituted An arylsulfonyl group, an optionally substituted alkyl group, and an optionally substituted arylcarbonyl group.
- a hydroxyl group is preferable.
- an alkyl group having 1 to 6 carbon atoms, a benzyl group and the like are preferable.
- a substituent of the optionally substituted hydrocarbon group as a substituent of the “optionally substituted hydroxyl group a substituent of the “optionally substituted hydrocarbon group” represented by R 2 above Similar numbers and similar ones are included.
- Examples of the optionally substituted alkylcarbonyl group and the optionally substituted arylcarbonyl group as the substituent of the ⁇ optionally substituted hydroxyl group '' represented by R 7 include, for example, Examples thereof include an alkylcarbonyl group having a good carbon number of 1 to 4, an optionally substituted benzoyl group, and the like. Among them, an acetyl group, a propionyl group, a benzoyl group and the like are preferable.
- substituent of the arylcarbonyl group which may be substituted include the same numbers and the same substituents as those of the “optionally substituted hydrocarbon group” for R 2 .
- Examples of the hydrocarbon group as the “optionally substituted hydrocarbon group” represented by R 8 and R 9 include the hydrocarbon as the “optionally substituted hydrocarbon group” represented by the aforementioned R 2 Examples are the same as the groups.
- Preferable examples include an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Among them, an alkyl group having 1 to 6 carbon atoms, a phenyl group, a benzyl group and the like are preferable.
- the substituent of the “optionally substituted hydrocarbon group” represented by R 8 and R 9 is the same as the substituent of the “optionally substituted hydrocarbon group” represented by R 2 described above. , And the like.
- the cyclic hydrocarbon or heterocyclic ring formed by R 5 and R 8 together may contain 1 to 3 atoms selected from nitrogen, oxygen, sulfur, etc., in addition to carbon. Or an unsaturated 5- to 8-membered monocyclic carbocyclic or heterocyclic ring, or a bicyclic fused carbocyclic or heterocyclic ring containing these.
- cycloalkyl eg, cyclopentyl, cyclohexyl, cycloheptyl, etc.
- cycloalkenyl eg, cyclopentenyl, cyclohexenyl, cyclohexenyl, cycloheptenyl, etc.
- aromatic hydrocarbon ring eg, phenyl
- oxygen-containing ring for example, frill, oxorael, tetrahydropyranyl, oxepinyl, benzofuranyl, benzopyranyl, etc.
- nitrogen-containing ring for example, pyrrolidyl, piperidyl, pyridyl, azepinyl, indolyl, quinolinyl, benzoxazepiel, etc.
- Sulfur-containing rings eg, phenyl, tetrahydrothiopyranyl, be
- the cyclic hydrocarbon or heterocyclic ring formed by R 5 and R 8 together may have a substituent, and the substituent may be ⁇ substituted or substituted '' represented by R 2 above. And the same number and the same as the substituents of the "optionally substituted hydrocarbon group".
- the cyclic hydrocarbon or heterocyclic ring formed by combining R 8 and R 9 may include one to three atoms selected from a nitrogen atom, an oxygen atom, a sulfur atom, and the like in addition to a carbon atom. Examples thereof include a saturated or unsaturated, 5- to 8-membered monocyclic carbocyclic or heterocyclic ring, or a bicyclic fused carbocyclic or heterocyclic ring containing these.
- cycloalkyl eg, cyclopentyl, cyclohexyl, cycloheptyl, etc.
- cycloalkenyl eg, cyclopentenyl, cyclohexenyl, cyclohexenyl, cycloheptenyl, etc.
- aromatic hydrocarbon ring eg, phenyl
- oxygen-containing ring for example, furyl, oxazolanyl, tetrahydropyranyl, oxepiel, benzofuranyl, benzopyranyl, etc.
- nitrogen-containing ring for example, pyrrolidyl, piperidyl, pyridyl, azepinyl, indolyl, quinolinyl, benzoxazepiel, etc.
- Sulfur-containing rings eg, phenyl, tetrahydrothioviranyl, benzo
- the cyclic hydrocarbon or heterocyclic ring formed by R 8 and R 9 together may have a substituent, and the substituent may be ⁇ optionally substituted '' represented by R 2 described above. And the same number and the same as the substituents of the "hydrocarbon group".
- an optionally substituted hydrocarbon group is preferable.
- an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, a phenyl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms are preferable. That is, an alkyl group having 1 to 6 carbon atoms, a phenyl group, a benzyl group and the like are preferable.
- RIG represents a hydrogen atom, one ZR 15 or —P (O) R 16 R 17 .
- the hydrocarbon group of the ⁇ optionally substituted hydrocarbon group '' represented by R 15 of ZR 15 examples include the same as the hydrocarbon group.
- an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and a cycloalkyl group having 3 to 8 carbon atoms are preferable.
- the substituent of the “optionally substituted hydrocarbon group” represented by 5 is the same as or similar to the substituent of the “optionally substituted hydrocarbon group” represented by R 2 above. Is mentioned. Of these, an optionally substituted alkyl group having 1 to 4 carbon atoms, an optionally substituted alkenyl group having 2 to 4 carbon atoms, a halogen atom such as fluorine, chlorine, and bromine, and having 1 to 4 carbon atoms Examples of the substituent include a methyl group, an ethyl group, a propyl group, an isopropyl group, a Bier group, and the like. Examples include fluorine, chlorine, bromine, a methoxy group, and an ethoxy group.
- - ZR 1 5 and R 1 5 in the "optionally substituted heterocyclic group" represented is as the substituent of the "optionally substituted hydrocarbon" represented by R 2 ' Heterocyclic group which may be substituted "and the like.
- a phenyl group which may be substituted a quinolyl group which may be substituted, and a benzoxazidi which may be substituted Azolyl group, optionally substituted pyridyl group, optionally substituted benzothiadiazolyl group, optionally substituted benzozoenyl group, optionally substituted oxazolidinyl group, optionally substituted
- An benzodioxanyl group, a substituted or unsubstituted dibenzofuranyl group and a morpholyl group are preferred, and particularly a optionally substituted phenyl group, an optionally substituted benzothiadiazolyl group and an optionally substituted
- a preferred example is a pyridyl group
- Z is -S0 2 _, -SO-, -CON 18 S0 2 - (R 18 is (:. 6 alkyl), -CONR 19 - (R 19 is - 6 alkyl) or - CO- shown R 18 and R the C Bok 6 alkyl represented by 1 9, for example, methyl, Echiru, propyl, iso-propyl, heptyl, isobutyl, sec- butyl, tert- butyl, pentyl, hexyl, and the like to.
- the “optionally substituted hydrocarbon group” represented by R 16 and R 17 is the same as the hydrocarbon group represented by the aforementioned R 2 3 ⁇ 4 “optionally substituted hydrocarbon group” And the like.
- an alkyl group having 1 to 12 carbon atoms an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, Examples thereof include a 3-8 cycloalkyl group and a C3-8 cycloalkenyl group.
- an alkyl group having 1 to 6 carbon atoms an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like are preferable.
- methyl, ethyl, phenyl, benzyl and the like are preferable.
- the substituent of the ⁇ optionally substituted hydrocarbon group '' represented by R 16 and R 17 is the same as the substituent of the ⁇ optionally substituted hydrocarbon group '' represented by R 2 And the like.
- R 16 and R 17 examples include, for example, an optionally substituted hydrocarbon group.
- an alkyl group having 1 to 8 carbon atoms examples thereof include an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Among them, 1 to 6 carbon atoms And an alkyl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like.
- substituent of the optionally substituted hydrocarbon group as the substituent of the “optionally substituted hydroxyl group” include the substituent of the “optionally substituted hydrocarbon group” represented by R 2 above. Similar numbers and similar ones are included.
- the “optionally substituted hydroxyl group” represented by R 16 and R 17 is preferably a hydroxyl group substituted by an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms, or the like.
- the ⁇ optionally substituted amino group '' represented by R 16 and R 17 includes the ⁇ substituted amino group '' as a substituent of the ⁇ optionally substituted hydrocarbon '' represented by R 2 above. And the like. " Preferably, methyl, ethyl, phenyl, benzyl and the like are mentioned.
- R 16 and R 17 are an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms, a hydroxyl group substituted with an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms. And a hydroxyl group substituted with methyl, ethyl, phenyl, benzyl, methyl, a hydroxyl group substituted with ethyl, and a hydroxyl group substituted with phenyl.
- ZR 15 Z and R 15 are as defined above
- Z is —S ⁇ 2 — or 1 CO—
- R 15 is an optionally substituted aryl group, an optionally substituted aralkyl group, and an optionally substituted heterocyclic ring. What is a group is preferable.
- Z is —SO 2 — or 1 CO—
- R 15 is an optionally substituted aralkyl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms which may be substituted, Those which are heterocyclic groups which may be substituted are preferred.
- Z is -S ⁇ 2-
- R 15 may be substituted with methyl, ethyl, propyl, isopropyl, vinyl, fluorine, chlorine, bromine, methoxy, ethoxy, etc.
- Phenyl, benzyl, phenyl, benzothiadiazolyl, or Z is —CO—
- R 15 is methyl, ethyl, propyl, isopropyl, vinyl, fluorine, chlorine, bromine, methoxy And a pyridyl group which may be substituted with an ethoxy group or the like.
- R 11 represents a hydrogen atom or a hydrocarbon group which may be substituted. .
- the hydrocarbon group of the ⁇ optionally substituted hydrocarbon group '' represented by R 11 is the same as the hydrocarbon group of the ⁇ optionally substituted hydrocarbon group '' represented by the aforementioned R 2 Things.
- Preferable examples include an alkyl group having 1 to 6 carbon atoms, an alkenyl group having 2 to 6 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Of these, an alkyl group having 1 to 6 carbon atoms, a benzyl group and the like are preferable, and an alkyl group having 1 to 4 carbon atoms is particularly preferable.
- the substituent of the ⁇ optionally substituted hydrocarbon group '' represented by R 11 is the same as or similar to the substituent of the ⁇ optionally substituted hydrocarbon group '' represented by R 2 One.
- R 11 is preferably a hydrogen atom.
- the nitrogen-containing heterocyclic ring which may be formed by combining R 1Q and R 11 is
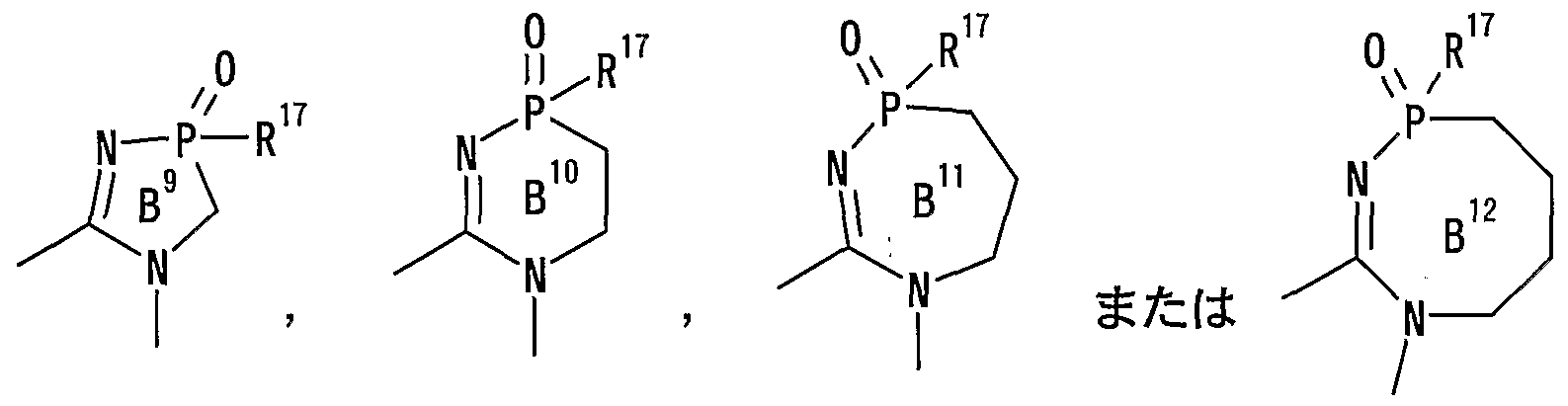
- ring B represents an optionally substituted 5- to 8-membered ring
- R 17 has the same meaning as described above, and ring B 1 to ring B 12 each represent a nitrogen-containing heterocyclic ring which may further have a substituent].
- the substituent which the ring B 1 to ring B 12 may have is the same as the substituent which the ⁇ optionally substituted hydrocarbon group '' represented by R 15 may have Means things.
- Examples of the nitrogen-containing heterocyclic ring which may be formed by R 1 Q and R 11 together include:
- ring B 2 and ring B 3 represent a nitrogen-containing heterocyclic ring which may further have a substituent.
- an alkyl group having 1 to 12 carbon atoms an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aryl group having 6 to 10 carbon atoms, and a 7 to 10 carbon atom
- an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms are preferable.
- substituent of the “optionally substituted hydrocarbon group” represented by R 12 and R 13 include the substitution of the “optionally substituted hydrocarbon group” represented by the aforementioned R 2 The same number and the same as the group can be mentioned.
- Examples of the nitrogen-containing heterocyclic ring which may be replaced together with R 12 and R 13 together with the nitrogen atom to which they are bonded include the aforementioned ⁇ R 3 and R 4 are both those wherein Nitrogen-containing heterocycles which may be substituted together with the nitrogen atom to be bonded ”. Particularly preferred examples include piperidyl, piperazyl, N-methylpiperazyl, pyrrolidyl, morpholyl and the like.
- Examples of the substituent of the “nitrogen-containing heterocyclic ring which may be substituted by both R 12 and R 13 forming a nitrogen atom to which they are bonded to —” are those represented by the aforementioned R 2 The same number and the same as the substituents of the “optionally substituted hydrocarbon group”.
- R 12 is a hydrogen atom, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aryl group having 6 to 10 carbon atoms.
- Preferred are a group, an aralkyl group having 7 to 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, a cycloalkenyl group having 3 to 8 carbon atoms, and the like.
- an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms are preferable.
- R 13 is preferably a hydrogen atom.
- the hydrocarbon group of the “optionally substituted hydrocarbon group” represented by R 14 the hydrocarbon group represented by the aforementioned “optionally substituted hydrocarbon group” represented by R 2 And the same as the group.
- an alkyl group having 1 to 12 carbon atoms an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, not having 6 carbon atoms
- an aryl group having 10 carbon atoms an aralkyl group having 7 to 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, and a cycloalkenyl group having 3 to 8 carbon atoms.
- an alkyl group having 1 to 4 carbon atoms and an aralkyl group having 7 to 10 carbon atoms are preferable.
- the substituent of the “optionally substituted hydrocarbon group” represented by R 14 is the same as or similar to the substituent of the “optionally substituted hydrocarbon group” represented by the aforementioned R 2. And the like.
- One ZR 15 represented by R 14 include the same as one ZR 15 represented by R 1Q.
- an optionally substituted hydrocarbon group is preferable.
- an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, A cycloalkenyl group having 3 to 8 carbon atoms and the like are preferable, and an alkyl group having 1 to 6 carbon atoms and an aralkyl group having 7 to 10 carbon atoms are particularly preferable.
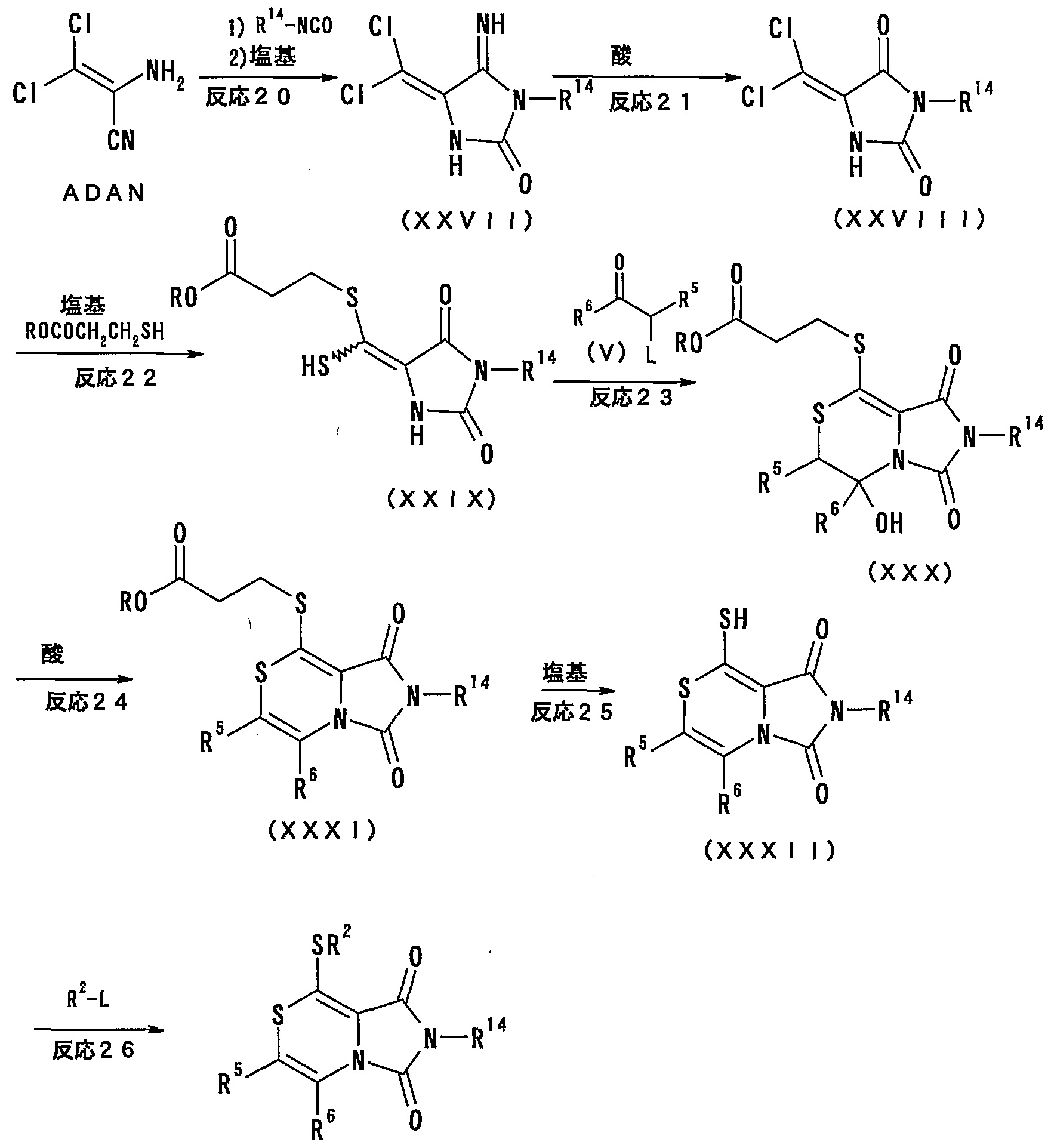
- R 1 is-(S) n — R 2 ,
- the isocyanate compound represented by R 1 G —NC ⁇ is used in an amount of 0.1 to 10 equivalents, preferably 0.2 to 5 equivalents, more preferably 1 to 2 equivalents, based on the amount of the raw material.
- the isocyanate compound represented by R 1 Q- NCO is preferably a sulfonyl isocyanate compound in which R 10 is R 15 SO 2 , more preferably a benzene ring in which R 15 may be substituted,
- benzenesulfonyl isocyanate, p-toluenesulfonyl isocyanate, P-fluorobenzenesulfonyl isocyanate, P-chlorobenzenesulfonyl isocyanate, and p-bromobenzenesulfonyl isocyanate are particularly preferable.
- isocyanates can be prepared from the corresponding amide by a method known per se.
- This reaction is advantageously performed in a solvent.
- These reactions are usually performed in a solvent that does not affect the reaction.
- Solvents are not specified as long as they do not affect the reaction, but amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, etc.), halogenated Hydrocarbon solvents (eg, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, chloroform benzene, etc.), nitrile solvents (eg, acetonitrile, etc.), ester solvents (eg, ethyl formate, Ethyl acetate, tert-butyl acetate, etc.), ketone solvents (eg, acetone, methyl ethyl ketone, methyl isobutyl
- octogenated hydrocarbon solvents eg, pyridine, 2, 4, 6- Aromatic bases such as trimethylpyridine, picoline, 4-dimethylaminopyridine, 2,6-lutidine, 1,8-diazabicyclo [5,4,0] -7-indenecene, and tertiary such as triethylamine and dimethylalanine Amin, etc.).
- pyridine 2, 4, 6- Aromatic bases such as trimethylpyridine, picoline, 4-dimethylaminopyridine, 2,6-lutidine, 1,8-diazabicyclo [5,4,0] -7-indenecene, and tertiary such as triethylamine and dimethylalanine Amin, etc.
- the base is used in an amount of 0 to 10 equivalents, preferably 0 to 1 equivalent, the reaction temperature is — 40 ° C to 15 ° C, preferably 0 ° C to 100 ° C, and the reaction time is 2 minutes to 96 hours, preferably 5 minutes to 60 hours.
- the mercapto compound represented by ROC ⁇ CH 2 CH 2 SH is used in an amount of 0.5 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 2 to 5 equivalents, based on the amount of the raw material.
- the base include alkali metal hydride or alkaline earth metal hydride (eg, sodium hydride, potassium hydride, calcium hydride, etc.), alkali metal alkoxide (eg, sodium methoxide, sodium ethoxide, Sodium tert-butoxide, etc.), inorganic bases (eg, sodium bicarbonate, sodium carbonate, sodium bicarbonate, sodium carbonate, sodium hydroxide, sodium hydroxide, sodium hydroxide), organic bases (eg, sodium tert-butoxide) Aromatic bases such as pyridine, 2,4,6-trimethylpyridine, picoline, 4-dimethylaminopyridine, 2,6-lutidine, 1,8_diazabicyclo [5,4,0] -7_indene, Ter
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent.
- Solvents are not specified as long as they do not affect the reaction, but amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, etc.), ether solvents (eg, , Tetrahydrofuran, getyl ether, dioxane, dimethoxyethane, etc.), sulfoxide solvents (eg, dimethyl sulfoxide), alcohol solvents (eg, methanol, ethanol, 2-propanol, tert-butanol, etc.), nitrile Solvent (eg, acetonitrile, etc.), ester solvent (eg, ethyl formate, ethyl acetate, tert-butyl acetate, etc.), hydrocarbon solvent (eg, hexane, benzene, toluene, etc.), halogenated carbon Hydrogen solvents (e.g., dichloromethane
- N, N-dimethylformamide, N, N-dimethylacetamide, methanol, ethanol, 2-propanol, tert-butanol, pyridine, tetrahydrofuran, dioxane, dimethoxyethane, acetonitrile, etc. preferable.
- the reaction temperature is from 140 to 150 ° C., preferably from 0 ° C. to 100 ° C.
- the reaction time is from 5 minutes to 96 hours, preferably from 30 minutes to 48 hours.
- the imidazolidine compound represented by the general formula (III) or a salt thereof is used as a raw material and reacted with a base to give an imidazolidine compound represented by the general formula (IV) (R represents the above alkyl group) or a compound thereof. Get the salt.
- Examples of the base include the bases shown in the above Reaction 2, and preferable examples are For example, an alkali metal hydride, an alkali metal alkoxide, an inorganic base, an organic base, an alkyl lithium, or an alkali metal amide is preferable. And 4-dimethylaminopyridine, methyllithium, butyllithium, lithium disopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably an amide solvent, an ether solvent, an alcohol solvent, a nitrile solvent, an organic base solvent Solvents, or a mixture thereof, among which N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol, acetonitrile, pyridine Etc. are particularly preferred.
- the reaction temperature is 140 ° C. to 150 ° C., preferably 2 Ot: to 120 ° C., and the reaction time is 5 minutes to 96 hours, preferably 30 minutes to 24 hours. .
- the above Reaction 2 and Reaction 3 can be advantageously performed by continuously performing the reaction without isolating and purifying the product during the reaction.
- the above 5-dichloromethylene imidazolidine compound represented by the general formula (II) or a salt thereof as a raw material with the above mercapto compound represented by the general formula ROCOCH 2 CH 2 SH in the presence of a base, the general formula An imidazolidine compound represented by (IV) or a salt thereof is obtained.
- a ketone compound having a leaving group L at the octyl position represented by the general formula (V) L represents a halogen atom such as chlorine, bromine or iodine or a sulfonyloxy group such as methanesulfonyloxy, P-toluenesulfonyloxy, trifluoromethanesulfonyloxy, etc.
- L represents a halogen atom such as chlorine, bromine or iodine
- a sulfonyloxy group such as methanesulfonyloxy, P-toluenesulfonyloxy, trifluoromethanesulfonyloxy, etc.
- the leaving group L is a halogen atom.
- the compound represented by the general formula (V) or a salt thereof, in which the leaving group L is a sulfonyloxy group is a compound known per se represented by the corresponding -hydroxyketone compound, for example, the method shown in Reaction 10 below. It can be prepared by sulfonylation of a hydroxyl group by the method described in the above.
- L is preferably a halogen atom such as chlorine, bromine or iodine, and more preferably chlorine or bromine. It is.
- the ketone compound or a salt thereof is used in an amount of 0.2 to 10 equivalents, preferably 1 to 5 equivalents, more preferably 1 to 3 equivalents, based on the raw material.
- the base include the bases shown in Reaction 2 above, and preferable examples thereof include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably, an amide solvent, an ether solvent, a ketone solvent, an ester solvent, and a nitrile solvent are used. Solvents, hydrocarbon solvents, halogenated hydrocarbon solvents, organic base solvents, etc., and mixtures thereof.
- N, N-dimethylformamide, N, N-dimethylileacetamide, titra Hydrofuran, dioxane, dimethoxyethane, acetate, 2-butenonone, ethyl acetate, tert-butyl acetate, acetatetonitrile, toluene, dichloromethane, chloroform, 1,2-dichloroethane, pyridine and the like are particularly preferred.
- the reaction temperature is from 140 ° C to 150 ° C, preferably from 0 to 100 ° C
- the reaction time is from 5 minutes to 96 hours, preferably from 30 minutes to 48 hours.
- the imidazothiazine compound represented by the general formula (VI) (R represents the above-mentioned alkyl group) or a salt thereof is subjected to a dehydration reaction in the presence of an acid, whereby the compound represented by the general formula (VII) is obtained.
- R represents the above alkyl group.
- Preferred examples of the acid used include, for example, octaacetic acid (eg, trifluoroacetic acid), inorganic acid (eg, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, perchloric acid, etc.), and organic carboxylic acids.
- Acids eg, formic acid, acetic acid, citric acid, tartaric acid, oxalic acid, etc.
- Lewis acids eg, zinc monoacetic acid, boron trifluoride etherate, etc.
- organic sulfonic acids eg, methanesulfonic acid, octanesulfonic acid, Benzenesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc.
- trifluoroacetic acid hydrochloric acid, sulfuric acid, phosphoric acid, acetic acid, methanesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc. Is particularly preferred.
- the acid is used in an amount of 0.05 to 10 equivalents, preferably 0.1 to 5 equivalents, and more preferably 0.2 to 2 equivalents, based on the raw material.
- the reaction is advantageously carried out by coexisting a dehydrating agent to remove the usually generated water.
- a dehydrating agent molecular sieves 4A, acid anhydride (eg, acetic anhydride, propionic anhydride, benzoic anhydride, etc.) are used.
- the amount is 5% to 10 times, preferably 10% to 5 times, or 10% by weight of the raw material.
- the solvent is not specified as long as it does not affect the reaction, but is preferably an organic acid-based solvent (eg, formic acid, acetic acid, propionic acid, etc.), an amide-based solvent (eg, N, N-dimethylformamide, N, N —Dimethylacetamide, etc.), ether solvents (eg, tetrahydrofuran, getylether, dioxane, dimethoxyethane, etc.), sulfoxide solvents (eg, dimethylsulfoxide, etc.), alcohol solvents (eg, methanol, ethanol, propanol, Isopropanol, isobutanol, tert-butanol, etc., nitrile solvents (eg,
- acetic acid, N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, tert-butanol, acetonitrile, ethyl acetate, tert-butyl acetate, toluene, xylenes, chloroform, 1,2- Dichloroethane is preferred.
- the reaction temperature is ⁇ 4 Ot: to 180 ° C, preferably 0 ° C to 120 ° C, and the reaction time is 5 minutes to 120 hours, preferably 10 minutes to 72 hours. .
- the imidazothiazine compound represented by the general formula (VIII) is obtained by reacting the imidazothiazine compound represented by the general formula (VII) (R represents the above alkyl group) or a salt thereof with a base. A compound or a salt thereof is obtained.
- Examples of the base include the bases shown in Reaction 2 above, and preferable examples thereof include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides.
- alkali metal hydrides sodium methoxide, sodium ethoxide, potassium tert-butoxide, sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, diisopropylethylamine, 4-dimethylaminopyridine, 1,8-diazavisic mouth [5,4,0] -7-Pendecene, methyllithium, butyllithium, lithium diisopropylamide, and lithium bistrimethylsilylamide are particularly preferred.
- the base is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents.
- These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, preferably an amide solvent, an ether solvent, an alcohol solvent, a nitrile solvent, an organic base. Solvent, or a mixture of these solvents. Among them, N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol, Acetonitrile, pyridine and the like are particularly preferred.
- the reaction temperature is ⁇ 40 ° C. to 15 Ot: preferably 20 ° C. to 120 °, and the reaction time is 5 minutes to 96 hours, preferably 30 minutes to 24 hours.
- L is a halogen atom such as chlorine, bromine, iodine, etc., or methanesulfonyloxy, P-toluenesulfonyl, trifluoromethanesulfonyloxy, etc. Sulfonyloxy group and the like are preferable.
- General formula: 2 a compound represented by one L or compound leaving group L is Nono androgenic atom of its salt corresponding alcohol Ichiru compound R 2 - 0 H or a hydroxyl by a method per se publicly known salts thereof Can be prepared by halogenating Among the compounds represented by the general formula R 2 —L or a salt thereof, the compound in which the leaving group L is a sulfonyloxy group can be converted from R 2 — ⁇ H to a compound represented by, for example, the method shown in Reaction 10 below. It can be prepared by sulfonylating a hydroxyl group by a known method.
- the compound represented by the above general formula R 2 -L or a salt thereof is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents to the starting material.
- the base include the bases shown in the above Reaction 2, and preferable examples thereof include, for example, hydrogenated metal alkoxides, metal alkoxides, inorganic bases, organic bases, alkyllithiums, and metal alkoxides.
- Amides and the like are preferable, and sodium hydride, sodium methoxide, sodium ethoxide, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, disopropylethylamine, pyridine, 4-dimethylaminopyridine , 1,8-Diazavicic mouth [5,4,0] -7-indene, methyllithium, butyllithium, lithium diisopropylamide, lithium bistrimethylsilylamide and the like are particularly preferable. Of these, liquid ones are also used as solvents.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, preferably an amide solvent, ether solvent, ketone solvent, ester solvent, nitrile Solvents, alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents, and the like, or a mixture thereof.
- the solvent shown in Reaction 2 above is used, preferably an amide solvent, ether solvent, ketone solvent, ester solvent, nitrile Solvents, alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents, and the like, or a mixture thereof.
- N, N-dimethylform Muamide, N-methylpyrrolidone, tetrahydrofuran, dioxane, dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred.
- reaction temperature is ⁇ 20 ° C. to 20 O, preferably 20 ° C. to 120 ° C.
- the reaction time is 5 minutes to 120 hours, preferably 20 minutes to 72 hours.
- Reaction 6 and reaction 7_ (a) can also be performed in a concatenated manner.
- the alcohol compound represented by the general formula R 2 —OH or a salt thereof is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, and more preferably 1 to 5 equivalents to the starting material.
- the dehydrating condensing agent include phosphine monoazodicarboxylates and phosphine monoazodicarboxylates reported in the Mitsunobu reaction [Synthes is, p. 1, 1989] [Journal of the Society of Synthetic Organic Chemistry] 55, 631, 1997] are advantageously used as the dehydrating condensing agent.
- Examples of the phosphine include triaryl phosphine, triaryl phosphine such as tri-0-tolyl phosphine, and the like, or trialkyl phosphine such as trimethyl phosphine and tributyl phosphine, among which triphenyl phosphine and tributyl phosphine are preferable.
- Examples of the azodicarboxylate include acetyl dicarboxylate, diisopropyl azodicarboxylate, and the like.
- azodicarboxylic acid amides include N, N, ⁇ ', ⁇ '-tetramethyl azodicalpoxamide, ⁇ , ⁇ , N', N'-tetraisopropyl azodicarpoxamide, 1, tri (azodicarponyl) And dipiperidine.
- the dehydrating condensing agent is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. It is preferable to use the same moles of phosphine and azodicarboxylic acid ester or azodicarboxylic acid amide.
- These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction.
- amide solvents Preferably used are amide solvents, ether solvents, ketone solvents, ester solvents, nitrile solvents, hydrocarbon solvents, octogenated hydrocarbon solvents, organic base solvents, etc., or a mixture thereof.
- the reaction temperature is between 120 ° C and 150 ° C, preferably between 20 ° C and 120 ° C, and the reaction time is between 5 minutes and 120 hours, preferably between 20 minutes and 60 hours. is there.
- a thiol compound represented by the general formula R 2 —SH (where R 2 represents an aryl group which may have a substituent) from an imidazothiazine compound represented by the general formula (VIII) or a salt thereof as a raw material.
- the thiol compound represented by the general formula R 2 —SH or a salt thereof is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material.
- the dehydrating condensing agent for example, the phosphine and azodicarboxylic acid ester or azodicarboxylic acid amide mentioned in the above reaction 7- (b) are advantageously used. Examples include acetyldiazolate azodicarboxylate and diisopropyl azodicarboxylate.
- azodicarboxylate amides examples include N, N, N ', N, -tetramethylazodicarpoxamide, 1,1- (azodicarpoxamide). Nyl) dipiperidine and the like are preferred.
- the dehydrating condensing agent is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. It is preferable that phosphine and azodicarboxylic acid ester or azodicarboxylic acid amide be used in the same mole number. These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably, an amide solvent, an ether solvent, a ketone solvent, an ester solvent, and nitrile are used. Solvent, hydrocarbon solvent, halogenated hydrocarbon solvent, organic base solvent, etc.
- the reaction temperature is -20 ° C to 120 ° C, preferably 20 ° C to 80 ° C, and the reaction time is 10 minutes to 240 hours, preferably 20 minutes to 120 hours.
- R 1 is -NR 3 R 4 ,
- R 1 is -SR 2 ,
- Imidazolidine compound represented by the general formula (X) (Z represents a S0 2.) Or As a salt as a raw material, Amin or its salt with a base represented by R 3 R 4 NH or R 12 R 13 NH To give an imidazolidine compound (Z represents S ⁇ 2 ) represented by the general formula (XI) or (XII) or a salt thereof.
- the amine represented by R 3 R 4 NH or R 12 R 13 NH or a salt thereof is 1 to 50 Equivalents, preferably 2 to 20 equivalents, more preferably 2 to 10 equivalents are used. Of these, liquid ones are also used as solvents. These amines are also advantageously used as salts with suitable acids.
- Examples of the acid include octaacetic acid (eg, trifluoroacetic acid), inorganic acid (eg, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, perchloric acid, etc.), and organic carboxylic acid (eg, formic acid, acetic acid) , Citric acid, tartaric acid, oxalic acid, etc.), organic sulfonic acids (eg, methanesulfonic acid, benzenesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc.) are used, among which hydrochloric acid, formic acid, acetic acid , Methanesulfonic acid, P-toluenesulfonic acid and the like are particularly preferred.
- octaacetic acid eg, trifluoroacetic acid
- inorganic acid eg, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric
- the acid is usually used in an equimolar amount to the amine.
- the base include the bases shown in the above Reaction 2, and preferable examples include, for example, hydrogenated alkali metal, alkoxide of alkali metal, inorganic base, organic base, and alkali metal amide.
- the base is used in an amount of 0.1 to 20 equivalents, preferably 0.5 to 10 equivalents, and more preferably 1 to 5 equivalents. These reactions are usually advantageously carried out in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above Reaction 2, etc. is used, and preferably an amide solvent, an ether solvent, an alcohol solvent, a nitrile solvent, Examples include organic base solvents and the like, or a mixed solvent thereof. Among them, N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol , Acetonitrile, pyridine and the like are particularly preferred.
- the reaction temperature is from ⁇ 40 ° C. to 150 ° C., preferably from 20 ° to 120 ° C. (: the reaction time is from 5 minutes to 96 hours, preferably from 30 minutes to 24 hours.
- R 1 is one SR 2 , XY is
- the base include the bases shown in Reaction 2 above, and preferable examples include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides.
- sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0]- 7-Pendecene, methyllithium, butyllithium, lithium disopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred.
- liquid ones are also used as solvents.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents.
- L is preferably a halogen atom such as chlorine, bromine or iodine, or a cyano group or an imidazole group.
- the compound represented by the above general formula R 15 —Z—L is used in an amount of 0.2 to 20 equivalents, preferably 1 to 1 equivalent to the raw material. 0 equivalents, more preferably 1 to 5 equivalents are used.
- These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, a nitrile solvent.
- the reaction temperature is from 180 ° C. to 150 ° C., preferably from 0 to 100 ° C., and the reaction time is from 1 minute to 120 hours, preferably from 5 minutes to 48 hours.
- Examples of the base include the bases shown in the above-mentioned Reaction 9-1 (a), and preferable examples include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkalis. Metal amides and the like are preferred, and sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazapicyclo 0]-7-Pendecene, methyllithium, butyllithium, And lithium pistrimethylsilylamide are particularly preferred. Of these, liquid ones are also used as solvents.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents.
- L is preferably a halogen atom such as chlorine, bromine, and iodine.
- the compound represented by the above general formula R 16 R 17 P ( ⁇ ) 1 L is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above reaction 9- (a) or the like is used, preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, Examples include nitrile-based solvents, alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents, and the like, or a mixed solvent thereof. Dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred.
- the reaction temperature is -80 to 150 ° C, preferably 0 to 100 ° C, and the reaction time is 1 minute to 120 hours, preferably 5 minutes to 48 hours.
- imidazothiazine compound represented by the general formula (XIII) or a salt thereof as a starting material the general formula R 18 _Z- NCO (Z represents S_ ⁇ 2 or a single bond.) Represented by the 'Isoshi Aneto acids or their By reacting with a salt, an imidazothiazine compound represented by the general formula (X ”) or a salt thereof is obtained.
- the reaction is advantageously performed in the presence of a base.
- a base The base shown in the above reaction 9-1 (a) and the like are preferable.
- Preferred examples include, for example, alkali metal hydride, alkali metal alkoxide, inorganic base, organic base, alkyl lithium, and alkali metal amide.
- Sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabisic D [5, 4, 0]- 7-Pendecene, methyllithium, butyllithium, lithium diisopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. Of these, liquid ones are also used as solvents.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents.
- the isocyanate represented by the general formula R 18 —Z-NCO is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material. These reactions are usually and advantageously effected in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above reaction 9-1 (a) is used, and preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, Examples include nitrile solvents, alcohol solvents, hydrocarbon solvents, organic base solvents, and mixtures thereof.N, N-dimethylformamide, N-methylpyrrolidone, tetrahydrofuran, dioxane , Dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred.
- the reaction temperature ranges from 80 ° C to 150 ° C, preferably from 0 ° C to 100 ° C, and the reaction time ranges from 1 minute to 120 hours, preferably from 5 minutes to 48 hours.
- condensing agent examples include N, N'-carbonyldiimidazole, benzotriazolyl-N-hydranide salt, N, N, -disocyanate.
- Preferred examples thereof include, for example, N, N dicyclohexylcarboimide, Particularly preferred are getyl cyanophosphate, and triethyl hydrochloride-3- (3-dimethylaminopropyl) carbodiimide.
- the condensing agent is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents.
- Examples of the base include the bases shown in the above reaction 9-1 (a). Preferred examples include alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, 7-alkyllithiums, and alkalis.
- Metal amides and the like are preferable, and sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0 ] -7-Pendecene, methyllithium, butyllithium, lithium disopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. Of these, liquid ones are also used as solvents.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. Formula R 1 5 -.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above reaction 9-1 (a) is used, and preferably an amide solvent, an ether solvent, a ketone solvent, or an ester solvent Solvents, nitrile-based solvents, alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents and the like, or a mixture thereof, among which N, N-dimethylformamide, N-methylpyrrolidone, tetrahydrofuran, Dioxane, dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred.
- Anti The reaction temperature is
- imidazothiazine compound represented by (X IV) Z represents a S_ ⁇ 2.
- a salt thereof as a raw material, subjecting the hydroxyl group in the formula a conversion reaction of the leaving group L, to Accordingly, imidazothiazine compound represented by the general formula (XV) (Z represents a S_ ⁇ 2.) or a salt thereof.
- the conversion reaction to the leaving group L ′ include a halogenation reaction with a halogenating agent and a sulfonic acid esterification reaction.
- halogenating agent examples include hydrogen halide (eg, hydrogen iodide, hydrogen bromide, hydrogen chloride, etc.) or halogenating agent (eg, phosphorus trichloride, phosphorus pentachloride, phosphorus oxychloride, phosphorus tribromide, Organophosphorus compounds such as phosphorus halides such as phosphorus bromide, triphenylphosphine monocarbon tetrachloride, triphenylphosphine monobromine, triphenylphosphine mono-N-bromosuccinimide, triphenylphosphite methyl iodide, or thionyl chloride, oxalyl chloride , Phosgene, etc.).
- hydrogen halide eg, hydrogen iodide, hydrogen bromide, hydrogen chloride, etc.
- halogenating agent eg, phosphorus trichloride, phosphorus pentachloride, phosphorus
- the halogenating agent is used in an amount of 1 to 10 equivalents, preferably 1 to 3 equivalents. Of these, liquid ones are also used as solvents. These halogenation reactions are advantageously performed in a solvent.
- Solvents include water, organic acid solvents (eg, formic acid, acetic acid, etc.), ketone solvents (eg, acetone, methylethyl ketone, methyl isobutyl ketone, etc.), amide solvents (eg, N, N-dimethylformamide) , Dimethylacetamide, etc.), halogenated hydrocarbon solvents (eg, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, cyclobenzene), alcohol solvents (eg, tert- Butanol), nitrile solvents (eg, acetonitrile), ester solvents (eg, ethyl formate, ethyl acetate,
- reaction temperature is ⁇ 20 to 150, preferably to 100 ° C.
- reaction time is 2 minutes to 48 hours, preferably 5 minutes to 10 hours.
- the hydroxyl group of the raw material is converted to a sulfonyl halide (eg, methanesulfonyl chloride, benzenesulfonyl chloride, P-toluenesulfonyl chloride, trifluoromethanesulfonyl chloride, etc.) or a sulfonic anhydride (eg, methanesulfone Acid anhydride, benzenesulfonic anhydride, P-toluenesulfonic anhydride, trifluoromethanesulfonic anhydride, etc.) in the presence of a suitable base.
- a sulfonyl halide eg, methanesulfonyl chloride, benzenesulfonyl chloride, P-toluenesulfonyl chloride, trifluoromethanesulfonyl chloride, etc.
- the sulfonyl halide or sulfonic anhydride is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, and more preferably 1 to 5 equivalents, based on the starting material.
- the base include the bases shown in the above Reaction 2, and preferable examples thereof include, for example, an alkali metal hydride, an inorganic base, an organic base, and an alkali metal amide.
- potassium triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-indene, lithium diisopropylamide, lithium pistrimethylsilylamide and the like.
- liquid ones are also used as solvents.
- the base is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents.
- the sulfonyloxy group formed in the sulfonic acid esterification reaction can be converted to a halogen group by reacting with an appropriate inorganic halide (eg, sodium iodide, potassium iodide, sodium bromide, potassium bromide, etc.). It is possible.
- the inorganic halide is used in an amount of 1 to 50 equivalents, preferably 1 to 20 equivalents, and more preferably 1 to 10 equivalents, based on the raw material. These reactions are usually performed in a solvent that does not affect the reaction.
- the solvent examples include those used in the above-mentioned halogenation reaction except for water, and among them, acetone, methylethylketone, N, N-dimethylformamide, dimethylacetamide, dichloromethane, and chloroform And 1,2-dichloroethane, acetonitrile, ethyl acetate, tert-butyl acetate, tetrahydrofuran, getyl ethere, dioxane, dimethoxyethane, hexane, toluene, pyridine, and the like, or a mixed solvent thereof. These are particularly preferably water-free solvents.
- the reaction temperature is from 120 to 200, preferably from 20 ° C. to 12 Ot: and the reaction time is from 5 minutes to 120 hours, preferably from 20 minutes to 72 hours.
- the imidazothiazine compound represented by the general formula (XV) (Z represents so 2 ) or a salt thereof is reacted with a base or an acid to obtain the compound represented by the general formula (XVI).
- Examples of the base include the bases shown in Reaction 2 above, and preferable examples thereof include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, and alkali metal amides. , Potassium tert-butoxide, sodium carbonate, potassium carbonate, diisopropylethylamine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-indene, lithium diisopropylamide, lithium pistrimethylsilylamide Etc. are particularly preferred.
- the base is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents.
- Examples of the acid include the acids shown in the above Reaction 5, among which, for example, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, formic acid, acetic acid, methanesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid And boron trifluoride etherate complex are particularly preferred. Of these, liquid ones are also used as solvents.
- the acid is used in an amount of 0.05 to 10 equivalents, preferably 0.1 to 5 equivalents, and more preferably 0.2 to 2 equivalents, based on the raw material. These reactions are advantageously carried out usually in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above Reaction 2 or Reaction 5 is used, and preferably, an organic acid solvent, an amide solvent, an ether solvent, or an alcohol solvent Solvents, nitrile-based solvents, hydrocarbon-based solvents, halogenated hydrocarbon-based solvents, organic base-based solvents, and the like, or a mixture thereof.
- reaction temperature is —40 ° C. to 200 ° C., preferably 0 to 12 O, and the reaction time is 5 minutes to 120 hours, preferably 30 minutes to 48 hours.
- R 1 is — SR 2 , Fifteen
- the imine represented by the general formula (IV) Dazorijin compounds Z is S 0 2, R indicates the alkyl group.
- the general formula (XV III) imidazolidine represented Ru compounds Z is S_ ⁇ 2, R is obtained.) or a salt thereof shows the alkyl group .
- Imida Zorijin compound represented by the general formula (XV III) Z is S_ ⁇ 2, R represents the alkyl group.
- imidazolidine compound represented by the general formula (XIX) Z is S 0 2, R represents the alkyl group.
- an imidazolysine compound represented by the general formula (XIX) or a salt thereof is used as a starting material, and a ketone having a leaving group L at the ⁇ -position represented by the above general formula (V)
- a ketone having a leaving group L at the ⁇ -position represented by the above general formula (V) By reacting the compound or a salt thereof in the presence of a base, an imidazothiazine compound represented by the general formula (XX) or a salt thereof is obtained.
- the imidazothiazine compound represented by the general formula (XX) ( ⁇ represents so 2 ) or a salt thereof is reacted with an acid to form the compound represented by the general formula (X) or (XXI).
- the acid include the acids shown in Reaction 5 above, among which, for example, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, formic acid, acetic acid, methanesulfonic acid, p-toluenesulfonic acid, camphorsulfonic acid And boron trifluoride etherate are particularly preferred. Of these, liquid ones are also used as solvents.
- the acid is used in an amount of 0.05 equivalents, preferably 10 equivalents, preferably 0.1 to 5 equivalents, more preferably 0.2 to 2 equivalents, based on the raw material.
- These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvents described in Reaction 5 above are used, and preferably, organic acid solvents, amide solvents, ether solvents, alcohol solvents, nitriles Solvents, hydrocarbon solvents, haptic hydrocarbon solvents, organic base solvents, etc., and mixtures thereof.
- reaction temperature is between 14O: to 200 ° C, preferably not 0 to 120 ° C, and the reaction time is 5 minutes to 120 hours, preferably 30 minutes to 48 hours.
- R is _SR 2 .
- imidazolidine compound represented by (Z is S_ ⁇ 2 are shown.
- a salt thereof as a raw material represented by the general formula (XX III) (R represents an alkyl group such as methyl, ethyl, propyl, isopropyl, 2-ethylhexyl) or a salt thereof in the presence of a base.
- R represents an alkyl group such as methyl, ethyl, propyl, isopropyl, 2-ethylhexyl
- Z represents so 2
- R 1 is a hydrogen atom, an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group,
- organometallic reagents or their equivalents examples include alkyl lithium (eg, methyl lithium, ethyl lithium, n-butyl lithium, sec-butyl lithium, tert-butyl lithium, etc.), alkyl magnesium hydride (eg, Methylmagnesium bromide, ethylmagnesium bromide, n-butylmagnesium bromide, n-pentylmagnesium bromide, n-hexylmagnesium bromide, etc., alkyl zinc halides (eg, methyl zinc bromide, getyl zinc bromide, etc.) Alkyl zinc (eg, dimethyl zinc, diethyl zinc, etc.), alkyl aluminum (eg, trimethyl aluminum, etc.) are used.
- alkyl lithium eg, methyl lithium, ethyl lithium, n-butyl lithium, sec-butyl lithium, tert-butyl lithium, etc.
- an arbitrary halide can be converted into an organolithium, an organomagnesium halide or an alkyl zinc halide by a method known per se as a raw material and used in this reaction.
- the metal hydride include lithium aluminum hydride, sodium borohydride, lithium borohydride, diisobutylaluminum hydride, and the like.
- the organic metal reagent or its equivalent or metal hydride is used in an amount of 0.5 to 20 equivalents, preferably 1 to 10 equivalents. These reactions can be advantageously performed in the presence of an appropriate organic phosphorus compound or the like.
- the organic phosphorus compound include triarylphosphine (eg, triffee).
- the organic phosphorus compound is used in an amount of 1 to 50 equivalents, preferably 1 to 10 equivalents, based on the amount of the raw material.
- reaction can be performed in the presence of an appropriate inorganic salt (eg, lithium iodide, lithium bromide, lithium chloride, cerium chloride, etc.).
- the inorganic salt is used in an amount of 1 to 50 equivalents, preferably 1 to 10 equivalents, based on the raw material.
- These reactions are usually performed in a solvent that does not affect the reaction.
- the solvent examples include amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, etc.), ether solvents (eg, tetrahydrofuran, getyl ether, dioxane, dimethoxetane, etc.), Hydrocarbon solvents (eg, hexane, benzene, toluene, etc.), organic base solvents (eg, pyridine, 2,4,6-trimethylpyridine, picoline, 2,6-lutidine, 1,8-diazabicyclo [5 , 4, 0] -7-dendene and the like, or a mixed solvent thereof.
- amide solvents eg, N, N-dimethylformamide, N, N-dimethylacetamide, etc.
- ether solvents eg, tetrahydrofuran, getyl ether, dioxane, dimethoxetane, etc.
- Hydrocarbon solvents
- reaction temperature is from 100 to 150, preferably from 140 to 120, and the reaction time is from 2 to 120 hours, preferably from 10 to 4 hours. 8 hours.
- An imidazothiazine compound represented by the general formula (XXV) or a salt thereof is obtained by reacting an imidazothiazine compound represented by the general formula (XX) (Z represents CO) or a salt thereof with an acid. Get the salt.
- Examples of the acid include the acids shown in the above Reaction 5, among which, for example, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, formic acid, acetic acid, methanesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid And boron trifluoride etherate complex are particularly preferred. Of these, liquid ones are also used as solvents.
- the acid is used in an amount of 0.05 to 10 equivalents, preferably 0.1 to 5 equivalents, more preferably 0.2 to 2 equivalents, based on the raw material. These reactions are usually advantageously performed in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above Reaction 2 or Reaction 5 is used, and preferably an organic acid solvent, an amide solvent, an ether solvent, or an alcohol.
- the reaction temperature is from ⁇ 40 to 200, preferably from 0 ° C. to 120 ° C., and the reaction time is from 5 minutes to 120 hours, preferably from 30 minutes to 48 hours.
- an imidazothiazine compound represented by the general formula (XXV) or a salt thereof as a raw material and reacting with the general formula R 14 — L (L represents a leaving group) or a salt thereof in the presence of a base
- a base include the bases shown in the above Reaction 2.
- Preferred examples are, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides.
- the base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents.
- L is preferably a halogen atom such as chlorine, bromine or iodine.
- R 1 4 is compound of represented by R 1 5 Z-, L is chlorine, bromine, a halogen atom such as iodine or Metansuru Honiruokishi, p - toluenesulfonyl O carboxymethyl, such as triflate Ruo b methanesulfonyl two Ruoki Shi Sulfonyloxy groups and the like are preferred.
- the compound represented by the above general formula R 14 -L or a salt thereof is used in an amount of 0.2 equivalents, preferably 1 to 10 equivalents, and more preferably 1 to 5 equivalents to the raw material. . These reactions are advantageously carried out usually in a solvent.
- the solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, Examples include tolyl solvents, alcohol solvents, hydrocarbon solvents, organic base solvents, and the like, and mixtures thereof, among which N, N_dimethylformamide, N-methylpyrrolidone, tetrahydrofuran, dioxane, dimethyloxetane, Acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitril, toluene, pyridine and the like are particularly preferred.
- the reaction temperature is from 180 to 150, preferably from 0 to 100 T, and the reaction time is from 1 to 120 hours, preferably from 5 to 48 hours.
- R 1 is — SR 2 ,
- an isocyanate represented by the general formula R 14 -NC ⁇ (R 14 represents an optionally substituted hydrocarbon group) is reacted in the presence of an acid, and then reacted with a base As a result, a 5-dichloromethyleneimidazolidine compound represented by the general formula (XXV II) or a salt thereof is obtained.
- the isocyanate compound represented by R 14 -NCO is used in an amount of 0.1 to 10 equivalents, preferably 0.2 to 5 equivalents, more preferably 1 to 2 equivalents, based on the amount of the raw material.
- the isocyanate compound represented by R 14 -NC 0 is preferably such that R 14 is, for example, methyl
- Particularly preferred is isocyanate which is a group, benzyl group, n-butoxycarbonylmethyl group, ethoxycarbonylmethyl group, ethoxycarbonylethyl group and the like.
- the acid catalyst examples include Lewis acids (eg, boron trifluoride ether complex, zinc chloride, aluminum chloride, titanium tetrachloride, tin tetrachloride, etc.), organic sulfonic acids (eg, methanesulfonic acid, octanesulfonic acid, benzene) Sulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc., haloacetic acid (eg, trifluoroacetic acid, etc.), inorganic acid (eg, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, perchloric acid, etc.) Among them, Lewis acids are preferable, and aluminum chloride is particularly preferable.
- Lewis acids eg, boron trifluoride ether complex, zinc chloride, aluminum chloride, titanium tetrachloride, tin tetrachloride, etc.
- organic sulfonic acids
- the acid is used in an amount of 0.1 to 10 equivalents, preferably 0.2 to 5 equivalents, more preferably 1 to 2 equivalents, based on the amount of the raw material.
- This reaction is advantageously performed in a solvent. These reactions are usually performed in a solvent that does not affect the reactions.
- Solvents are not specified as long as they do not affect the reaction, but amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, etc.), halogenated carbon Hydrogen-based solvents (eg, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, cyclobenzene), nitrile solvents (eg, acetonitrile, etc.), ester solvents (eg, ethyl formate, ethyl acetate) , Tert-butyl acetate, etc.), ketone solvents (eg, acetone, methyl ethyl ketone, methyl isobutyl ketone, etc.), ether solvents (eg, tetrahydrofuran, getyl ether, diisopropyl ether, dioxan, dimethyloxetane, etc
- halogenated hydrocarbon solvents halogenated hydrocarbon solvents, ester solvents, ether solvents, hydrocarbon solvents, and the like are preferable, and dichloromethane, chloroform, tetrahydrofuran, ethyl ether, and toluene are particularly preferable.
- the reaction temperature is ⁇ 40 ° C. to 150 ° C., preferably O to 100 ° C., and the reaction time is 2 minutes to 96 hours, preferably 5 minutes to 60 hours.
- Examples of the base to be used include the bases shown in the above Reaction 2, and preferable examples thereof include, for example, pyridine, 4-dimethylaminopyridine, 1,8-diazabicycline [5,4,0] -7-pentadecene , Triethylamine, Dimethylaniline, Sodium carbonate, Carbonated Liu And the like.
- the base is used in an amount of 0 to 10 equivalents, preferably 0 to 1 equivalent.
- the reaction temperature is 14 to 150, preferably Ot: to 100, and the reaction time is 2 to 96 hours, preferably 5 to 60 hours. is there.
- Examples of the acid to be used include the acids shown in the above Reaction 5, etc., among them, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, methanesulfonic acid, ⁇ -toluenesulfonic acid, camphorsulfonic acid and the like. preferable. Of these, liquid ones are also used as solvents.
- the acid is used in an amount of 0.1 to 20 equivalents, preferably 0.5 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material. These reactions are advantageously carried out usually in a solvent.
- the solvent is not specified as long as it does not affect the reaction.
- the solvent include, for example, water in addition to the solvent shown in Reaction 5 above, and preferably water, formic acid, acetic acid, ⁇ , ⁇ -dimethylformamide, Examples thereof include tetrahydrofuran, dioxane, dimethoxetane, dimethyl sulfoxide, methanol, ethanol, propanol, isopropanol, isobutanol, tert-butanol, and acetonitrile, or a mixed solvent thereof.
- the reaction temperature is between 140 ° C. and 180 ° C., preferably Ot: 120
- the reaction time is between 10 minutes and 72 hours, preferably between 30 minutes and 48 hours.
- a 5_dichloromethyleneimidazolidine compound represented by the general formula (XXVI II) or a salt thereof is used as a raw material, and expressed by a general formula R ⁇ COCH 2 CH 2 SH in the presence of a base.
- R represents an alkyl group such as methyl, ethyl, propyl, isopropyl, 2-ethylhexyl, etc.
- XX IX an imidazolidine compound represented by the general formula (XX IX). Or to obtain its salt.
- imidazo represented by the general formula (XX IX) Using a lysine compound (R represents the above-mentioned alkyl group) or a salt thereof as a raw material, a ketone compound represented by the general formula (V) having a leaving group L at the cH position or a salt thereof is reacted in the presence of a base. As a result, an imidazothiazine compound represented by the general formula (XX) (R represents the above alkyl group) or a salt thereof is obtained.
- an imidazothiazine compound represented by the general formula (XXX) (R represents the above alkyl group) or a salt thereof is subjected to a dehydration reaction in the presence of an acid.
- an imidazothiazine compound represented by the general formula (XXXI) (R represents the above alkyl group) or a salt thereof is obtained.
- an imidazothiazine compound represented by the general formula (XX XI) (R does not have the above-mentioned alkyl group) or a salt thereof is used as a raw material and reacted with a base to give a general compound.
- An imidazothiazine compound represented by the formula (XXXII) or a salt thereof is obtained.
- an imidazothiazine compound represented by the general formula (XXXII) is used as a starting material, and in the presence of a base, a compound represented by the general formula R 2 — L (L is a leaving group To obtain an imidazothiazine compound represented by the general formula (XXXIII).
- substituents may have functional groups that decompose and react under each reaction condition.However, their protection and deprotection after the reaction can be performed by a method known per se. it can. The conversion of various functional groups that may be present on each substituent can be performed by a method known per se.
- the hydrolysis is advantageously performed in the presence of, for example, an acid or a base.
- the acid include the acid shown in Reaction 5 above
- examples of the base include the base shown in Reaction 3 above.
- hydrochloric acid sulfuric acid, phosphoric acid, trifluoroacetic acid, methanesulfonic acid, P-toluenesulfone Acid, camphorsulfonic acid, etc.
- sodium hydride sodium methoxide, sodium ethoxide, potassium tert-butoxide, sodium carbonate, carbonic acid
- potassium hydroxide sodium methoxide, sodium ethoxide, potassium tert-butoxide
- sodium carbonate carbonic acid
- potassium hydroxide triethylamine, diisopropylethylamine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-pinedecene and the like are preferable.
- the acid or base is used in an amount of 0.1 to 20 equivalents, preferably 1 to 5 equivalents, based on the starting material.
- These reactions are usually advantageously performed in a solvent that does not affect the reaction, and water, the solvent shown in the above Reaction 2, or the like is used. Among them, water, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol, acetonitrile, pyridine and the like, or a mixed solvent containing these waters are preferable.
- the reaction temperature is from ⁇ 20 ° C. to 20 ° C., preferably from 20 ° C. to 120 ° C.
- the reaction time is from 5 minutes to 120 hours, preferably from 20 minutes to 72 hours.
- the starting compound when a functional group such as an esterified or amidated carboxyl group, a cyano group, or a carbonyl group is reduced to be converted to a formyl group, a hydroxymethyl group, an aminomethyl group, a hydroxyl group, or the like, the starting compound is used. It is obtained by subjecting it to a reduction reaction.
- a functional group such as an esterified or amidated carboxyl group, a cyano group, or a carbonyl group
- the starting compound is used. It is obtained by subjecting it to a reduction reaction.
- This reaction involves metal hydride (eg, lithium aluminum hydride, sodium aluminum hydride, lithium borohydride, sodium borohydride, sodium cyanoborohydride, etc.), aluminum hydride (eg, hydrogen Diisobutylaluminum, etc.), dipolane (eg, borane-tetrahydrofuran, polan-methanol, etc.), dialkylborane (eg, di-sec-butylbutyl, ditexylporan, 9-boravisic [3,3,1] norpolonene, etc.)
- the reaction is carried out by reacting with a reducing agent in an inert solvent.
- the inert solvent examples include ether solvents (eg, tetrahydrofuran, methyl ether, dioxane, dimethyloxetane, etc.), hexane, benzene, toluene and the like.
- the reducing agent is used in an amount of 1 to 20 equivalents, preferably 1 to 5 equivalents, and the reaction temperature is from 170 ° C to 1.50 ° C, preferably from 0 to 100 ° C.
- the reaction time is 10 minutes to 48 hours, preferably 1 hour to 20 hours.
- the desired product When the desired product is obtained in a free state by each of the above-mentioned reactions, it may be converted into a salt according to a conventional method, and when the desired product is obtained as a salt, the free form is obtained according to a conventional method Alternatively, it can be converted to another salt.
- the compound thus obtained or a salt thereof can be obtained by known means, for example, phase transfer, concentration, solvent extraction, fractionation,
- the salt of the compound described in the present specification is preferably a pharmacologically acceptable salt, for example, a salt with an inorganic base, a salt with an organic base, a salt with an inorganic acid, a salt with an organic acid, or a basic salt. Or a salt with an acidic amino acid.
- the salt with an inorganic base include alkali metal salts such as sodium salt and potassium salt; alkaline earth metal salts such as calcium salt and magnesium salt; and aluminum salt, ammonium salt and the like.
- alkali metal salts such as sodium salt and potassium salt
- alkaline earth metal salts such as calcium salt and magnesium salt
- Preferred examples of the salt with an organic base include, for example, trimethylamine, triethylamine, pyridine, picoline, ethanolamine, diethanolamine, triethanolamine, dicyclohexylamine, ⁇ , ⁇ '-dibenzylethylene. And salts with diamine and the like.
- salts with inorganic acids include salts with hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid, and the like.
- Preferred examples of salts with organic acids include, for example, formic acid, acetic acid, trifluoroacetic acid, fumaric acid, oxalic acid, tartaric acid, maleic acid, citric acid, succinic acid, malic acid, methanesulfonic acid, benzenesulfonic acid, -Salts with toluenesulfonic acid and the like.
- Preferred examples of the salt with a basic amino acid include, for example, salts with arginine, lysine, ordinine, and the like.
- Preferred examples of the salt with an acidic amino acid include, for example, aspartic acid, glutamic acid, and the like. Salts.
- the compound (I) of the present invention may be a hydrate or a non-hydrate.
- the compound (I) used in the present invention may have one asymmetric carbon in the skeleton and thus may have two kinds of optical isomers. Each of these isomers and a mixture thereof are also described in the present invention. Included in the invention. When the substituent has an asymmetric carbon atom or an unsaturated bond, a stereoisomer or a geometric isomer is similarly produced, and the isomers and mixtures thereof are also included in the present invention.
- the compound of the present invention has a sulfur atom in its structure, which may be oxidized by an oxidizing agent (eg, hydrogen peroxide or m-chloroperbenzoic acid) according to a conventional method. Sulfonated compounds are also included in the present invention.
- the compound of the present invention may have an amide, imide, amidino, guanidino or peridode group in its structure or as a substituent, and these compounds have a resonance structure in which the double bond is isomerized. In some cases, these resonance structural compounds are also included in the present invention.
- the compound (I) of the present invention may be used as a prodrug.
- a prodrug a compound which is converted into a compound (I) by a reaction with an enzyme or gastric acid under physiological conditions in a living body, that is, a compound which is oxidized enzymatically , A compound that undergoes reduction, hydrolysis, etc., and changes to the compound (I), and a compound, which undergoes hydrolysis, etc., due to stomach acid or the like, to change to the compound (I).
- Examples of the prodrug of the compound (I) include compounds in which the amino group of the compound (I) is acylated, alkylated, and phosphorylated (eg, the amino group of the compound (I) is icosanoylated, alanylated, pentylamino).
- Carponylated (5-methyl-2-oxo- 1,3-dioxolen-141-yl) methoxycarbonylated, tetrahydrofuranylated, pyrrolidylmethylated, bivaloyoxymethylated, tert-butylated compounds
- the hydroxyl group of compound (I) is acylated, alkylated, phosphorylated or borated (eg, the hydroxyl group of compound (I) is acetylated, palmitoylated, propanoylated, pivaloyylated, succinylated) , Fumarylation, aranylation, dimethylaminomethylcarbonylated compounds, etc.);
- Compounds e.g., compound (I) is converted to ethyl ester, phenyl ester, carboxymethyl ester, dimethylaminomethyl ester, pivaloyloxymethyl ester, Ethoxycarponyloxetyl ester
- Compounds that inhibit cartilage matrix degradation can be used as preventive and therapeutic agents for osteoarthritis and diseases caused by cartilage destruction.
- Compounds that inhibit proteoglycan degradation, compounds that inhibit type I collagen degradation, and that inhibit the production of cartilage matrix-degrading enzymes such as matrix meta-oral proteinase 13 (MMP-13) and aggrecanases Compound and MMP gene expression suppression The compound having an effect is used as a prophylactic / therapeutic agent for diseases caused by osteoarthritis and cartilage destruction.
- MMP-13 matrix meta-oral proteinase 13
- Compounds that inhibit proteodalican degradation, compounds that inhibit type II collagen degradation, compounds that inhibit the production of cartilage matrix-degrading enzymes such as MMP-13 and aglycanase, and those that inhibit MMP gene expression Preferred examples of the compound having the following are condensed imidazolidine derivatives, and more specifically, the compound (I) or a salt thereof or a sulfoxide thereof.
- the compound (I) or a salt thereof or a sulfoxide thereof has excellent proteoglycan release inhibitory activity, collagen II degradation inhibitory activity, and cartilage matrix degrading enzyme production inhibitory activity such as MMP-13 and aggrecanase.
- the therapeutic agent for cartilage disease of the present invention containing Compound (I) or a salt thereof as an active ingredient is particularly useful for cartilage disease before the lesion reaches the bone itself, in terms of preventing deterioration of the cartilage tissue condition. Yes, it can be applied not only to treatment of osteoarthritis, rheumatoid arthritis and diseases caused by cartilage destruction, but also to prevention of these diseases.
- the compound (I) or its salt or its sulfoxide has an excellent inhibitory effect on the expression of the MMP gene (particularly the MMP-13 gene), and is useful as a safe preventive and therapeutic agent for all MMP-related diseases. is there.
- compound (I) or a salt thereof or a sulfoxide thereof exhibits an inhibitory action on proteoglycan release, an inhibitory action on collagen type I degradation, and an inhibitory action on cartilage matrix degrading enzyme production such as MMP-13 and aglycanase.
- Substrates, specifically t inhibit the degradation of proteodalican and type II collagen. That is, it has an effect as a proteodalican degradation inhibitor, a type I collagen degradation inhibitor, and a cartilage matrix degrading enzyme inhibitor.
- cartilage destruction As a result, it has a strong inhibitory effect on cartilage destruction, so it can be used for various cartilage diseases (deformation) in mammals (eg, humans, rats, guinea pigs, mice, cats, dogs, puppies, cows, pigs, etc.). Osteoarthritis and diseases caused by cartilage destruction), for example, for the prevention and treatment of osteoarthritis involving cartilage, rheumatoid arthritis involving cartilage, or cartilage destruction in joints etc. in related diseases Can be used for various cartilage diseases (deformation) in mammals (eg, humans, rats, guinea pigs, mice, cats, dogs, puppies, cows, pigs, etc.). Osteoarthritis and diseases caused by cartilage destruction), for example, for the prevention and treatment of osteoarthritis involving cartilage, rheumatoid arthritis involving cartilage, or cartilage destruction in joints etc. in related diseases Can be used for
- Compound (I) or its salt or its sulfoxide has an excellent MMP gene (Especially the MMP-13 gene) has an expression-suppressing effect, and in addition to all MMP-related diseases, for example, the above-mentioned joint diseases (eg, osteoarthritis, rheumatoid arthritis, etc.), osteoporosis, Cancer (eg, primary, metastatic or recurrent, breast, prostate, cancer, stomach, lung, colon (colon, rectum, anal), esophagus, duodenum, head and neck (tongue) Cancer, pharyngeal cancer, laryngeal cancer), brain tumor, schwannoma, non-small cell lung cancer, small cell lung cancer, liver cancer, kidney cancer, bile duct cancer, uterine cancer (uterine body cancer, cervical cancer), ovarian cancer, bladder Cancer, skin cancer, hemangiomas, malignant lymphomas, malignant melanomas, thyroid cancer, bone tumors, hemangiomas, an
- the compound (I) or a salt thereof or a sulfoxide thereof when used as an agent for preventing or treating cartilage disease, the compound (I) or a salt thereof is mixed with a pharmaceutically acceptable carrier according to a conventional method to obtain a drug having a dosage form suitable as a medicament.
- Solid preparations such as tablets, capsules, granules, powders, and suppositories; or liquid preparations such as syrups, drops, solutions, suspensions, and injections, orally or parenterally It can be administered. .
- Pharmaceutically acceptable carriers include various organic or inorganic carrier materials commonly used as drug substances, such as excipients, lubricants, binders, and disintegrants in solid preparations; solvents and dissolution in liquid preparations. It is formulated as an auxiliary, suspending agent, tonicity agent, buffer, soothing agent, stabilizing agent, anticoagulant, and the like. If necessary, pharmaceutical additives such as preservatives, antioxidants, coloring agents and sweeteners can also be used.
- Preferred examples of the excipient include lactose, sucrose, D-mannitol, starch, crystalline cellulose, light gay anhydride, and the like.
- Preferred examples of the lubricant include magnesium stearate, calcium stearate, talc, colloidal silica and the like.
- Suitable examples of the binder include, for example, crystalline cellulose, sucrose, D-mannitol, dextrin, hydroxypropylcellulose, and hydroxypropylmethyl.
- cellulose bilipyrrolidone Preferable examples of the disintegrant include starch, carboxymethylcellulose, carboxymethylcellulose calcium, croscarmellose sodium, carboxymethylsuniform sodium and the like.
- Preferred examples of the solvent include water for injection, alcohol, propylene glycol, macrogol, sesame oil, corn oil and the like.
- solubilizing agent examples include, for example, polyethylene glycol, propylene glycol, D-mannitol, cyclodextrins, twins, benzyl benzoate, ethanol, trisaminomethane, cholesterol, triethanolamine, and carbonic acid.
- solubilizing agent include, for example, polyethylene glycol, propylene glycol, D-mannitol, cyclodextrins, twins, benzyl benzoate, ethanol, trisaminomethane, cholesterol, triethanolamine, and carbonic acid.
- Sodium and sodium citrate examples include, for example, polyethylene glycol, propylene glycol, D-mannitol, cyclodextrins, twins, benzyl benzoate, ethanol, trisaminomethane, cholesterol, triethanolamine, and carbonic acid.
- Sodium and sodium citrate examples include, for example, polyethylene glycol, propylene glycol, D-mannitol, cyclodext
- suspending agent examples include surfactants such as, for example, stearyltriethanolamine, sodium lauryl sulfate, laurylaminopropionic acid, lecithin, benzalkonium chloride, benzethonium chloride, and glycerin monostearate; And hydrophilic polymers such as benzyl alcohol, polyvinylpyrrolidone, sodium carboxymethylcellulose, methylcellulose, hydroxymethylcellulose, hydroxyethylcellulose, and hydroxypropylcellulose.
- tonicity agent include sodium chloride, glycerin, D-manniyl, glucose, sorbyl, and the like.
- Preferred examples of the buffer include buffers such as phosphate, acetate, carbonate, and citrate.
- Preferable examples of the soothing agent include benzyl alcohol and the like.
- Preferable examples of the stabilizer include polyethylene glycol, polylactic acid and the like.
- Preferred examples of the coagulant include dextran sulfate, heparin and the like.
- Preferred examples of preservatives include, for example, paraoxybenzoates, chlorobutanol, benzyl alcohol, phenethyl alcohol, dehydroacetic acid, sorbic acid and the like.
- Suitable examples of the antioxidant include, for example, sulfite, ascorbic acid and the like.
- the compound (I) or a salt thereof or a sulfoxide thereof is dissolved in a conventional aqueous diluent and used as a solution.
- Diluents include glucose aqueous solution, physiological saline, Ringer's solution, and nutritional supplement solution.
- the prophylactic / therapeutic agent of the present invention can also be produced by dissolving the suspension and shaping it into microcapsules, spheres, rods, needles, pellets, films and the like. Further, the prophylactic / therapeutic agent of the present invention may be formed into a sustained-release preparation containing compound (I) or a salt thereof or a sulfoxide thereof and a biodegradable high molecular compound.
- the preparation of the sustained-release preparation can be in accordance with the method described in JP-A-9-1265355.
- the ratio of containing compound (I) or a salt thereof or a sulfoxide thereof is about 0.1 to about 95% by weight, preferably about 1 to about 50% by weight, based on the whole preparation.
- Compound (I) or a salt thereof or a sulfoxide thereof is preferably used as an oral preparation.
- Compound (I) or a salt thereof or a sulfoxide thereof can be directly administered to a cartilage disease-affected site when formed into a topical preparation and administered. In this case, injection is preferable.
- Parenteral preparations for topical administration eg, injection preparations for intramuscular, subcutaneous, organ, joint, etc., solid preparations such as implants, granules, powders, liquid preparations such as suspensions, ointments, etc.
- topical administration eg, injection preparations for intramuscular, subcutaneous, organ, joint, etc., solid preparations such as implants, granules, powders, liquid preparations such as suspensions, ointments, etc.
- It can also be administered as
- compound (I) or a salt thereof or a sulfoxide thereof is dispersed in a dispersant (eg, a surfactant such as Tween 80, HCO-60, carboxymethyl cellulose, sodium alginate, hyaluronic acid).
- a dispersant eg, a surfactant such as Tween 80, HCO-60, carboxymethyl cellulose, sodium alginate, hyaluronic acid.
- preservatives eg, methyl paraben, propyl paraben, etc.
- tonicity agents eg, sodium chloride, mannitol, sorbitol, glucose, etc.
- buffering agents eg, calcium carbonate, etc.
- a practical injectable preparation can be obtained by preparing an aqueous suspension together with a pH adjuster (eg, sodium phosphate, potassium phosphate, etc.).
- vegetable oils such as sesame oil and corn oil, or mixtures of these with phospholipids such as lecithin, or dispersed with medium-chain fatty acid triglycerides (eg, migliol 812) are used as oily suspending agents. Make injections that can be used.
- a compound e.g., a hyaluronic acid formulation for injection (eg, Kaken Pharmaceutical Co., Ltd .: Aru Note)
- a dispersion medium It can be prepared by dispersing I) or a salt thereof or a sulfoxide thereof.
- Hyaluronic acid used in the dispersion medium may be a non-toxic salt thereof, for example, an alkali such as sodium or potassium.
- Metal salts, salts of alkaline earth metals such as magnesium, calcium and the like can be mentioned, and particularly, sodium salts are preferably used.
- Hyaluronic acid and non-toxic salts thereof have a molecular weight of about 200,000 to 500,000 (viscosity method), preferably about 500,000 to 300,000, and more preferably about 700,000 to 250,000. Things are used.
- the final concentration of hyaluronic acid or sodium hyaluronate in this dispersant is preferably less than 1% (W / V) as a viscosity, and is preferable in terms of various operations and ease of administration, and particularly about 0.02 to 1%. It is more preferably less than about 0.1 to 1% (W / V).
- the dispersion medium may contain a pH adjuster, a local anesthetic, an antibiotic, a solubilizer, an isotonic agent, an anti-adsorption agent, glycosaminodalican, a polysaccharide, etc. by a method known per se. Good.
- Preferred examples thereof include mannitol, sorbitol, salt, glycine, ammonium acetate, or a water-soluble protein that can be injected into a body fluid without substantially exhibiting pharmacological activity.
- glycosaminodalican examples include hyaluronic acid, chondroitin, chondroitin sulfate A, chondroitin sulfate C, dermatan sulfate, heparin, heparan sulfate, and keutane sulfate.
- polysaccharide examples include an acidic polysaccharide such as arginic acid.
- the above-mentioned water-soluble protein may be any protein which is soluble in water, physiological saline or buffer, and includes, for example, human serum albumin, human serum globulin, collagen, gelatin and the like.
- the pH adjuster include glycine, ammonium acetate, citrate, hydrochloric acid, sodium hydroxide and the like.
- the local anesthetic include chlorobutanol and xylocaine hydrochloride.
- Examples of the antibiotic include gentamicin and the like.
- the solubilizer include glycerin and polyethylene glycol 400.
- the tonicity agent include mannitol, sorbitol, sodium chloride and the like.
- the above-mentioned adsorption inhibitor include polyoxyethylene sorbitol monooleate.
- the content of the water-soluble protein is preferably 0.05 to 5 Omg, more preferably 0.5 to 2 Omg, per one administration of the preparation. Preferably, it is more preferably 0.75 to 1 O mg.
- the formulation is Acid or a salt thereof (eg, sodium phosphate, potassium phosphate, etc.).
- the concentration of sodium phosphate or potassium phosphate in the injection is about 0.1 mM to 50 mM, and about lmM to 10 mM. Is preferred.
- 'Aseptic preparations include, but are not particularly limited to, a method of sterilizing the entire production process, a method of sterilizing with gamma rays, and a method of adding a preservative.
- the agent of the present invention Since the agent of the present invention has an excellent cartilage destruction inhibitory activity (cartilage matrix degrading enzyme production inhibitor and proteodalican degradation inhibitor), it can be used for cartilage diseases (eg, osteoarthritis, osteoarthritis of the knee, rheumatoid arthritis). And the destruction of articular cartilage (disease caused by cartilage destruction) and the like in the similar diseases.
- the agent of the present invention can be used together with other therapeutic agents for joint diseases.
- Compound (I) or its salt or its sulfoxide is used as a cartilage destruction inhibitor (a cartilage matrix degrading enzyme production inhibitor and a proteodalican degradation inhibitor)
- Drugs to be combined include anti-inflammatory steroids (eg, prednisolone, hydrocortisone, methylprednisolone, dexamethasone, vesamethasone, etc.), non-steroidal anti-inflammatory analgesics
- hyaluronic acid preparations eg, sodium hyaluronate, etc.
- COX-II inhibitors eg, celecoxib, lefecoxib, etc.
- In-polymer preparations eg, Remicade, Enbrel, Kinneret, etc.
- the agent of the present invention is used as a safe and highly efficacious preparation suitable for prevention and treatment of cartilage disease, maintenance of cartilage tissue, and the like. For example, it is possible to efficiently exert the inhibitory effect on cartilage destruction locally, and to improve the quality of life of patients who are impaired in daily life due to fatigue due to joint cartilage or pain due to destruction.
- compound (I) or a salt thereof or a sulfoxide thereof is used as a prophylactic / therapeutic agent for cartilage disease
- the daily dose depends on the condition and weight of the patient, the method of administration, and the release rate from the preparation.
- Adult (weight 5 O kg) for administration 1 About 5 to about 2,000 mg, preferably about 30 to about 50 Omg, of the compound (I) or its salt or its sulfoxide per person, is administered in 1 to 3 divided doses.
- the dose may be determined according to the type and content of compound (I) or a salt thereof or a sulfoxide thereof, the dosage form, the duration of drug release, the animal to be administered (eg, Humans, rats, mice, cats, dogs, puppies, cattle, pigs, and other mammals) vary depending on the purpose of administration, but when applied by parenteral administration, for example, from about 0.1 to about 10 Omg of compound (I) or a salt thereof or a sulfoxide thereof may be released from the administration preparation.
- the animal to be administered eg, Humans, rats, mice, cats, dogs, puppies, cattle, pigs, and other mammals
- parenteral administration for example, from about 0.1 to about 10 Omg of compound (I) or a salt thereof or a sulfoxide thereof may be released from the administration preparation.
- the pharmaceutical composition of the present invention has low toxicity and can be used safely.
- the compound when the compound was orally administered to rats at a dose of 30 OmgZkg / day, no abnormal findings were observed.
- the following example compounds are excellent in absorbability when administered orally, and can be advantageously used for oral preparations.
- the present invention will be described in more detail with reference to Examples, but the present invention is not limited thereto.
- the percentage (%) in the solution is expressed by weight Z volume percent unless otherwise specified.
- the mixing ratio of the solvent is indicated by the volume ratio unless otherwise specified.
- the 1H and 13 C nuclear magnetic resonance (NMR) spectra were obtained using DPX-300 or AC-300 spectra using tetramethylsilane as an internal standard (Bulliki, Germany), The values were measured with a Gemini 200 or Mercury 300 spectrum meter (Varian, USA) and ⁇ 6 values are shown in ppm.
- s is singlet
- d is doublet
- t is triplet
- Q is quartet
- sept represents a sevenfold line
- m represents a multiple line
- b or br represents a wide line, respectively.
- dd is a double double line
- ddd is a double double double line
- dt is a double triple line
- dq is a double quadruple line
- brd ⁇ ⁇ wide double line, and so on They may be displayed in combination. Coupling constants are shown in Hz.
- ADDP (1, 1,-(azodicarbonyl) dipiperidine) 1, ⁇ -(azodicarponyl) dipiperidine
- DIAD diisopropyl azodicarboxylate
- DMAP 4_N, N-dimethhy1 am i nopyr idine
- ADAN (31.9 g, 0.233 mol, manufactured by Junsei Chemical Co., Ltd.) is dissolved in getyl ether (300 ml, hereinafter abbreviated as ether), and stirred at room temperature while stirring with P-toluenesulfonyl isocyanate (36.0 ml). , Purity 98%, manufactured by Tokyo Chemical Industry Co., Ltd.), and the mixture was stirred at room temperature for 14 hours. The resulting precipitate was collected by filtration, washed with diethyl ether, and dried to give a pale yellow powder of the title compound (74.3 g, 0.222 mol).
- Benzyl isocyanate (20 g) was added to a dichloromethane (400 ml) solution of ADAN (19.4 g), and aluminum chloride (18.9 g) was added in three portions. After stirring at the same temperature for 1 hour, water (150 ml) was added, and the organic solvent was distilled off. To the remaining aqueous layer were added ethyl acetate (150 ml), water (150 ml) and 0.5 M citrate (150 ml), and the mixture was stirred for 5 minutes. After the organic 'solvent was distilled off, the precipitate was collected by filtration, washed with water, and dried to obtain 58.6 g of a colorless powder.
- the obtained compound was dissolved in acetone (400 ml), carbonated lime (39.2 g) was added, and the mixture was heated under reflux for 50 minutes. After cooling to room temperature, water (400 ml) was added, and the mixture was adjusted to pH 3 with about 500 ml of IN hydrochloric acid. The precipitate was collected by filtration, washed with water (100 ml), and dried to give the title compound (31.2 g, 77%).
- R 10 is —ZR 15 (compound in which ⁇ is S ⁇ 2 and R 15 is 4_phenoxyphenyl group)
- R 10 is -ZR 15 (Z is S ⁇ 2 , R 15 is a 4-chlorophenyl group))
- Ethanol (310 ml) was added to the compound synthesized in Reference Example 1-1 (30.8 g, 92.2 mmol) and potassium carbonate (29.3 g, 0.212 mol), and the mixture was stirred at room temperature while stirring with ethyl 3_mercaptopropionate (24.2 g). ml, 0.180 mol) was added dropwise over about 6 hours, and the reaction mixture was stirred at room temperature for 12 hours. 1N Hydrochloric acid (240 ml) was added to the reaction solution, and the mixture was stirred for 30 minutes.
- R 10 is —ZR 15 (Z is S ⁇ 2 , R 15 is a p-bromophenyl group),
- N, N-dimethylaminopyridine (0.21 g, 1.72 mmol) was added 16 hours after the start of the reaction, and p-bromobenzenesulfonyl chloride (0.89 g, 3.43 mmol) was added 18 hours later.
- concentration of the reaction solution the residue was diluted with ethyl acetate (500 ml), washed with 0.3 N hydrochloric acid, 0.1 N hydrochloric acid, water, and saturated saline, and then dried over sodium sulfate.
- Ethanol (27 ml) was added to the compound synthesized in Reference Example 2-4 (1.35 g, 2.45 bandol) and carbonic acid rim (407 mg, 2.95 bandol), and the mixture was stirred at 80 ° C for 2 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate-THF (1: 1, 60 ml) and washed with 0.1N hydrochloric acid (60 ml). The aqueous layer was extracted with ethyl acetate (30 ml), combined with the organic layer, washed with brine, and dried over sodium sulfate.
- IR Infrared absorption spectrum (IR) (in KBr): 3069, 3027, 2965, 2936, 2911, 1481 cm- 1 .
- Methanesulfonyl chloride (0.652 ml, 8.42 mmol) was added to a solution of 4,4,5,5,5-pentafluoroethanol (1.00 g, 5.61 ol) in tetrahydrofuran (20.0 ml). And triethylamine (1.57 ml, 11.2 mmol) were sequentially added under ice-cooling. After stirring the reaction mixture at room temperature for 3 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was water washed to give after drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (1.42 g, 993 ⁇ 4) as an oil-like product.
- Methanesulfonyl chloride (1.95 ml, 25. 2) was added to a solution of 5,5,6,6,6-pentafluorohexanol (3.23 g, 16.8 t) obtained in Reference Example 8 in tetrahydrofuran (60.0 ml). mmol) and triethylamine (4.69 ml, 33.6 maraudal 01) were sequentially added under ice cooling. After stirring the reaction mixture at room temperature for 2 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (4.13 g, 913 ⁇ 4) as an oil.
- R 2 Gabe Njiru group in formula (XVI II), the synthesis of Z is S0 2, R 15 is p- Toruiru group, and R is an Echiru group
- ADAN (10.8 g, 79.1 mmol) was dissolved in dichloromethane (220 ml), and isocyanate was dissolved.
- Ethyl acetate (10.0 g, 75.1 dust ol) was added, and the mixture was cooled to, and aluminum chloride (2 g, 75.1 ol) was added in several portions.
- the precipitate was filtered, washed with ether, ice-cold water, and water, and dried to obtain a pale yellow powder (14.2 g, 46.6 mmol).
- Acetonitrile 60 ml was added to the compound synthesized in Reference Example 13-2 (3.05 g, 10.3 mmol) and carbonated carbonate (4.29 g, 31.0 ol) and suspended, and methyl 3-mercaptopropionate (2.34 ml) was added. , 20.7 mmol was added dropwise, and the mixture was stirred at room temperature for 2 hours, and further stirred at 8 for 40 minutes. After concentrating the reaction solution, 1N hydrochloric acid (31 ml), water (80 ml) and ethyl acetate (100 ml) were added and mixed, and the organic layer was extracted with a 2% aqueous sodium hydrogen carbonate solution.
- the aqueous layers were combined, adjusted to pH 3, and extracted with ethyl acetate.
- the organic layer was washed with a 7% saline solution and a saturated saline solution, and then dried with sodium sulfate.
- the solvent was distilled off under reduced pressure, methanol was added to the residue, and the resulting precipitate was collected by filtration, washed with methanol, and dried to give the title compound as an orange oil (3.20 g, 8.50 benzyl) .
- IR (KBr) V 3368, 3200, 3071, 2953, 2924, 2907, 2868, 1765, 1730, 1634, 1574, 1499 CDT 1 .
- IR (Br) V 3300-2800, 2961, 1703, 1671, 1570, 1507 cm “ 1 .
- N-Boc-6-amino-1-hexyl mesylate (purity 80.4, 6.39 g, 40.5 thigh 01) was obtained from 6-amino-1-hexanol (5.0 g, 40.5 thigh ol). A colored oil was obtained.
- Deoxo-Fluor TM (5.06 g, 22.9 mmol) was added dropwise over 30 minutes to a solution of 4-oxocyclohexanecarbethylate (2.29 g, 13.4 mmol) in dichloromethane (20 ml) with stirring with stirring for 30 minutes. After warming, the reaction was stirred at room temperature for 6 hours. The reaction solution was poured into ice (50 g), and extracted with ethyl acetate (100 ml) and getyl ether (30 ml).
- Lithium aluminum hydride (purity 80%, 0.402) was added to a solution of ethyl cis-4- (2-tetrahydroviranyl) oxycyclohexanecarboxylate (4.14 g, 15.4 benzyl) in THF (80 ml) while stirring at 0 ° C. g, 8.47 mmol) was added in several portions, and the mixture was stirred for 2.5 hours with TC.
- the extract was purified by extraction with ethyl acetate, and cis-4- (2-tetrahydropyranyl) oxycyclohexanemethanol (purity 90%) was added. , 3.67 g, 15.4 mmol, quant.).
- Cis-4- (2-tetrahydropyranyl) oxycyclohexanemethanol (purity 90%, 3.67 g, 15.4 mmol) and triethylamine (2.56 ml, 18.5 mmol) in THF (70 ml) were stirred at 0 with methane.
- a solution of sulfonyl chloride (1.87 g, 16.2 ml) in THF (5 ml) was added dropwise over 20 minutes, and the reaction solution was stirred at 0 ° C for 2.5 hours.
- the crude material (5.27 g) obtained by extraction with ethyl acetate was purified by silica gel (50 g) chromatography (elution solvent, ethyl acetate-hexane, 2: 8-3: 7), and cis-4- A pale yellow oil of (2-tetrahydrobilanyl) oxycyclohexylmethyl mesylate (3.76 g, 12.9 ol, 83.5%) was obtained.
- the reaction solution was poured into ice-water (about 80 g) in which sodium carbonate (1.51 g, 18.1 mol) was dissolved, and extracted with ethyl acetate-hexane (1: 1, 60 nil) and ethyl acetate (50 ml).
- the organic layer was washed with saturated sodium bicarbonate and saturated brine, dried over sodium sulfate, and concentrated to give a yellow oil.
- This was subjected to chromatography on silica gel (20 g) (ethyl acetate-hexane, 1: 9-4: 6), and the ethyl acetate-hexane (2: 8-3: 7) fraction was concentrated to dryness. Later, it was diluted with Jetil ether.
- Lithium aluminum hydride (purity 80, 2.41 g, 50.9 t) was added to a solution of 3,3-dimethyldaltaric anhydride (purity 953 ⁇ 4, 6.92 g, 46.2 t) in THF (140 ffll) while stirring at 0 ° C. ) was added in several portions, and the mixture was stirred (TC for 2 hours, room temperature for 13 hours, and heated to reflux for 4.5 hours. Ethyl acetate (20 ml) was added to the reaction solution, and the mixture was stirred.
- the reaction solution was neutralized by adding sodium hydrogen carbonate, and purified by extraction with ethyl acetate to obtain a colorless oil of adamantan-1-methyl acetate (10.8 g, quant.).
- Triethylamine (3.07 ml, 22.2 mmol) was added to a solution of adamantane-1-ethanol (3.33 g, 18.5 mmol) and methanesulfonyl chloride (2.24 g, 19.4 mmol) in THF (70 ml) for 20 minutes while stirring at 0. And dropped.
- the reaction solution was stirred at 0 ° C for 2 hours.
- the reaction solution was purified by extraction with ethyl ethyl diisopropyl acetate (1: 1) to give adamantan-toethyl methanesulfonate (5.30 g, 18.5 mmol, quant.) As a pale yellow oil.
- reaction solution was purified by extraction with ethyl acetate-hexane (1: 1), and adamantan methanesulfonate 1- Methyl (purity 903 ⁇ 4, 4.27 g, 15.8 mmol, quant.) was obtained as a pale yellow oil.
- a suspension of adamantane trimethyl methanesulfonate (purity 90%, 4.27 g, 15.8 mmol) and sodium iodide (7.10 g, 47.4 mmol) in 2-butanone (85 ml) was heated under reflux for 3.5 days.
- the reaction mixture was extracted with ethyl acetate-hexane (2: 3), purified by silica gel (40 g) chromatography (hexane), and purified with 1-odomethyladamantane (2.20 g, 7.96 bandol, 50.4%) as a white solid.
- the crude substance obtained by ethyl acetate extraction was purified by silica gel (20 g) chromatography (ethyl acetate-hexane, 2: 8-3: 7), and (2S, 4R) -N-Boc-4- (2 A colorless oil of -tetrahydrobilanyloxy) pyrrolidine-2-methyl mesylate (1.35 g, 3.55 mmol, 96.9%) was obtained.
- trans-2-hydroxymethylcyclohexane-1-methyltosylate (2.21 g, 7.41 mmol) and acetic anhydride (6 ml) in pyridine (12 ml) was stirred at room temperature for 11 hours.
- the reaction solution was concentrated to dryness to give trans-2-acetoxymethylcyclohexane-methyltosylate (2.54 g, 7.4 mmol, quant.) As a colorless oil.
- trans-2-Acetoxymethylcyclohexane-1-methyltosylate (2.54 g, 7.4 t ol) and sodium iodide (3.33 g, 22.2 t ol) suspended in 2-butane nonone (55 ml)
- the liquid was heated at reflux for 2.5 hours.
- the reaction mixture was extracted with ethyl acetate-hexane (1: 4), concentrated to dryness, and trans-2-iodomethylcyclohexane-1-methyl acetate (1.98 g, 6.68 mmol, 90.1) was obtained as a colorless oil. Obtained as a product.
- Lithium aluminum hydride (purity 803 ⁇ 4, 2.12 g) was added to a solution of dimethyl cis-cyclohexane-1,4-dicarbonate (8.19 g, 37.2 mmol) obtained above in THF (160 ml) while stirring at 0 ° C. , 44.7 mmol) was added in several portions, and the mixture was stirred at 0 ° C for 80 minutes. Extract with ethyl acetate-THF (4: 1), dry and concentrate to dryness to obtain cis-cyclohexane-1,4-dimethanol (purity 87%, 6.18 g, quant., 8% trans-isomer A colorless oil was obtained.
- Triethylamine (27.2) was added to a solution of 3_methyl-1,3_butanediol (20.66 g, purity 90%, 0.179 mol) and methanesulfonyl chloride (14.7 ml, 0.187 mol) in THF (300 ml) with stirring at 0 ° C. ml, 0.196 mol) was added dropwise over 80 minutes, and the reaction solution was stirred at 0 ° C for 1.5 hours.
- the reaction mixture was extracted with getyl ether-hexane (1: 1), washed with water, dried and concentrated to dryness to give 3-methyl-3-hydroxybutyl mesylate (purity 80.5%, 39.3 g, 0.174 mol). Obtained as a yellow oil.
- Methanesulfonyl chloride (2.09 ml, 27.1) was added to a solution of 6,6,7,7,7-pentafluorofuranol (3.72 g, 18.0 mmol) obtained in Reference Example 51 in tetrahydrofuran (80.0 ml). mmol) and triethylamine (5.03 ml, 36.1 bandol) were sequentially added under ice-cooling. After stirring the reaction mixture at room temperature for 3.5 hours, 1 hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried '(MgSO 4), solvent was evaporated under reduced pressure to give the title compound (3.80 g, 74%) as an oil.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Physical Education & Sports Medicine (AREA)
- Epidemiology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Rheumatology (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Immunology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
明細書 Specification
縮合ィミダゾリジン誘導体、 その製造法及び用途 技術分野 Condensed imidazolidine derivatives, their production methods and applications
本発明は関節疾患治療剤などの医薬として有用な新規な縮合ィミダゾリジン誘 導体、 その製法または剤に関する。 背景技術 The present invention relates to a novel condensed imidazolidine derivative useful as a medicament such as an agent for treating a joint disease, a method for producing the same, or an agent therefor. Background art
変形性関節症は加齢、肥満 > 力学的ストレス、 炎症等で引き起こされる関節軟骨 の変性や破壊を伴った進行性疾患で主に膝関節や股関節に生じる。 近年先進国で は高齢者の増加による有病率の増加が問題となっており、治療薬としては痛みの緩 和を主目的として抗炎症薬やヒアルロン酸製剤が用いられている [バード ·エツ チ · n—(Bird, H.A.:)、 ドラッグス ·アンド ·エイジング (Drugs &Aging)、 12巻、 87-95頁、 1998年、 ランド 'ビー(Lund, B.)、 スカンジナビアン ·ジャーナル · リューマトロジー(Scandinavian J. Rheumatol.), 27巻、 32— 37頁、 1998年、 及 び井上一、 変形性関節症の診かたと治療、 医学書院、 94一 99頁、 1994年] 。 Osteoarthritis is a progressive disease with degeneration and destruction of articular cartilage caused by aging, obesity> mechanical stress, inflammation, etc., and mainly occurs in the knee and hip joints. In recent years, in the developed countries, the increase in the prevalence due to the increase in the elderly has become a problem, and anti-inflammatory drugs and hyaluronic acid preparations have been used as therapeutic agents mainly for the purpose of pain relief [Bad Etz Chin- (Bird, HA :), Drugs & Aging, 12, 87-95, 1998, Lund, B., Scandinavian Journal Rhematology (Scandinavian J. Rheumatol.), Vol. 27, pp. 32-37, 1998, and Ando Inoue, Diagnosis and treatment of osteoarthritis, Medical Shoin, 94-99, 1994].
軟骨は軟骨細胞と軟骨細胞が産生する軟骨マトリックスから形成されており、そ のマトリックス構成成分は水和能を持ち軟骨に柔軟性や弾力性を与えるプロテオ ダリカンと網目状の骨格を構成する II型コラーゲンに大別される。 変形関節症に おける軟骨破壊は、 プロテオダリカン分解とそれに続く II型コラーゲン分解の 2 つの段階に分けられる [ポール 'エー 'アール (Poole, A.R.)、 藤沢隆ー (訳) 、 蛋白質核酸酵素、 40巻、 520— 531頁、 1995年、土井武ら、 ザ ·ポ一ン (The Bone)、 1 巻、 293— 297頁、 2000 年] 。 海産スポンジ由来の化合物ヒメ二アルジシン (hymen ialdisine) に関節軟骨からのプロテオグリカン分解抑制作用が報告され [ァリソン ·ェム ·ビー (Alison, M.B.), ザ ·ジャーナル ·ォブ ·ファーマコロジ 一 · アンド ·ェクスペリメンタル ·セラピューテイクス (The Journal of Pharmacology and Experimental Therapeutics)、 290巻、 587— 593頁、 1999年]、 さ ら に ヒメ二アルジシンの誘導体デブロ モ ヒ メ 二アルジシン (debromohymenialdisine; DBH) には関節軟骨細胞培養系でのマトリックスメタ口 プロティナ一ゼ (MMP) mRNA産生を抑制する作用 [ヴァシォス ·ジー (Vasios, G.) ら、 アメリカリュ一マチ学会 1999 年会予稿集 (Proceedings of 1999 American College of Rheumatology Annu l Meeting)、ポスターセッシヨン!) (Poster Session D) 、 No. 1123] 、動物モデルでの関節軟骨保護作用 [フイブレグッェ * ピー (Huibregtse, Β·)ら、 アメリカリューマチ学会 1999年会予稿集 (Proceedings of 1999 American College of Rheumatology Annual Meeting) 、 ポスタ一セッション D (Poster Session D) 、 No. 1102] が報告されている。 Cartilage is formed from chondrocytes and a cartilage matrix produced by chondrocytes.The matrix components are proteodalican, which has hydration ability and gives flexibility and elasticity to cartilage, and type II, which forms a reticulated skeleton. It is roughly divided into collagen. Cartilage destruction in osteoarthritis can be divided into two stages: proteodalican degradation followed by type II collagen degradation [Poole, AR], Takashi Fujisawa (translation), protein nucleic acid enzymes, 40, 520—531, 1995, Takei Doi et al., The Bone, 1, 293—297, 2000]. Hymen ialdisine, a compound derived from a marine sponge, has been reported to inhibit proteoglycan degradation from articular cartilage [Alison, MB (Alison, MB), The Journal of Pharmaceutical Sciences and Ande The Journal of Pharmacology and Experimental Therapeutics, Vol. 290, pp. 587-593, 1999], and a derivative of hymenialdisine, debromohymenialdisine (DBH), Inhibition of matrix meta-oral proteinase (MMP) mRNA production in articular chondrocyte culture system [Vasios, G. Proceedings of 1999 American College of Rheumatology Announcer Meeting, poster session! (Poster Session D), No. 1123], Protective effect on articular cartilage in animal models [Huibregtse, Β ·, et al., Proceedings of 1999 American College of Rheumatology Annual Meeting ), Poster Session D, No. 1102].
一方、縮合イミダゾリジン骨格を有する化合物としては、 除草活性を有する二環 性ヒダントイン誘導体に関する特許が報告されている [特顧平 10-501453、 WO 97/47626] が、 プロテオダリカン分解抑制作用の報告はない。 On the other hand, as a compound having a condensed imidazolidine skeleton, a patent on a bicyclic hydantoin derivative having herbicidal activity has been reported [Japanese Patent Application No. 10-501453, WO 97/47626]. There is no report.
また、 縮合イミダゾリジンの合成として、 m-イミダゾ [5,卜 c] [1,4]チアジン 1,3, 5 (2H, 6H)-トリオンの合成が報告されている [Chem. Ber.l00(2)巻、 564-577 頁、 1967年] が、 この化合物にはプロテオダリカン分解抑制作用の報告はない。 変形性関節症の患者にとって、軟骨基質の分解を抑制することにより関節破壊の 進行を阻止する薬剤は、鎮痛剤など対症療法薬と異なり根治治療薬となりうること から、 第一に必要な薬剤として求められている。 発明の開示 As a synthesis of condensed imidazolidines, the synthesis of m-imidazo [5, tric] [1,4] thiazine 1,3,5 (2H, 6H) -trione has been reported [Chem. Ber.l00 ( 2), pp. 564-577, 1967], but there is no report on the inhibitory effect of this compound on proteodarican degradation. For patients with osteoarthritis, drugs that inhibit the progression of joint destruction by suppressing cartilage matrix degradation can be curative drugs unlike symptomatic drugs such as analgesics. It has been demanded. Disclosure of the invention
本発明者らはかかる現状に鑑み、 種々検討した結果、 一般式 In view of this situation, the present inventors have conducted various studies and found that the general formula
[式中、 R1は、 一 (S) n— ; 2または一 NR3R4を、 nは 0ないし 2の整数を、 R2は水素原子、 置換されていてもよい炭化水素基または置換されていてもよい複 素環基を、 R3および R4は同一または異なって水素原子、 置換されていてもよい 炭化水素基、置換されていてもよい複素環基またはそれらが結合している窒素原子 と一緒に形成する含窒素複素環を、 R5は水素原子、 置換されていてもよい炭化水 素基、 シァノ基、 ァシル基、 エステル化もしくはアミド化されていてもよい力ルポ キシル基または置換されていてもよい複素環基を、 [Wherein, R 1 represents one (S) n —; 2 or one NR 3 R 4 , n represents an integer of 0 to 2, R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group or substituted Wherein R 3 and R 4 are the same or different and are each a hydrogen atom, an optionally substituted hydrocarbon group, an optionally substituted heterocyclic group, or A nitrogen-containing heterocyclic ring formed together with a nitrogen atom, R 5 is a hydrogen atom, and an optionally substituted hydrocarbon An alkyl group, a cyano group, an acyl group, an esterified or amidated hydroxyl group or an optionally substituted heterocyclic group,
,9また , 9 also
r 、R r, R
を、 R6は、 水素原子、 置換されていてもよい炭化水素基、 シァノ基、 ァシル基、 エステル化もしくはアミド化されていてもよい力ルポキシル基または置換されて いてもよい複素環基を、 R7は置換されていてもよい水酸基を、 R8および R9はそ れぞれ水素原子または置換されていてもよい炭化水素基を (R5と R6、 R5と R8 または R 8と R 9はそれぞれ一緒になつて置換されていてもよい環状炭化水素また は複素環を形成してもよい) 、 R 6 represents a hydrogen atom, a hydrocarbon group which may be substituted, a cyano group, an acyl group, a propyloxyl group which may be esterified or amidated, or a heterocyclic group which may be substituted; R 7 is an optionally substituted hydroxyl group; R 8 and R 9 are each a hydrogen atom or an optionally substituted hydrocarbon group (R 5 and R 6 , R 5 and R 8 or R 8 And R 9 may be taken together to form an optionally substituted cyclic hydrocarbon or heterocyclic ring),
を、 R1。は、 水素原子、 -ZR15 (Zは _S02—、 一 SO—、 _C〇NR18S〇 2— (R18は 6アルキルを示す) 、 — CONR19— (R19は — 6アルキルを 示す) または一 C〇一を、 R15は、 置換されていてもよい炭化水素基または置換 されていてもよい複素環基を示す) または— P (〇) R16R17 (R16および R17 はそれぞれ置換されていてもよい炭化水素基、置換されていてもよい水酸基または 置換されていてもよいアミノ基を示す) を、 R 11は水素原子または置換されてい てもよい炭化水素基を (R1Qと R 11は一緒になつて置換されていてもよい含窒素 複素環を形成してもよい) 、 R 12および R 13はそれぞれ水素原子または置換され ていてもよい炭化水素基、あるいはそれらが結合している窒素原子と一緒になつて 含窒素複素環を、 R14は、 水素原子、 置換されていてもよい炭化水素基または一 ZR15 (Zおよび R 15は、 前記と同意義を示す) を示す。 但し、 R 5が水素原子、 X Y が A, R 1. Is a hydrogen atom, -ZR 15 (Z is _S0 2 —, SO—, _C〇NR 18 S〇 2 — (R 18 is 6 alkyl), — CONR 19 — (R 19 is —6 alkyl R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group) or — P (〇) R 16 R 17 (R 16 and R 17 Represents an optionally substituted hydrocarbon group, an optionally substituted hydroxyl group or an optionally substituted amino group), and R 11 represents a hydrogen atom or an optionally substituted hydrocarbon group ( R 1Q and R 11 may together form a nitrogen-containing heterocyclic ring which may be substituted), R 12 and R 13 are each a hydrogen atom or an optionally substituted hydrocarbon group, or Together with the nitrogen atom to which it is attached forms a nitrogen-containing heterocycle, R 14 is a hydrogen atom, Or a hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described above). Where R 5 is a hydrogen atom, XY is
を示すとき、 R14は置換されていてもよい炭化水素基または一 ZR15 (Z、 R15 は、 前記と同意義を示す) を示す。 ]で表される化合物 (I) またはその塩、 また はそのスルホキシドである新規な縮合イミダゾリジン誘導体を初めて合成したと ころ、 これらの化合物がその特異な化学構造に基づいて、意外にも軟骨器官培養系 において優れたプロテオグリ力ン分解抑制作用を示すこと、さらに動物モデルにお いて膝関節軟骨からのプロテオダリカン遊離抑制作用を示す等の医薬としての優 れた性質を有していることを見い出し、 これらに基づいて本発明を完成した。 すなわち本発明は、 In the formula, R 14 represents an optionally substituted hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described above). The first synthesis of a novel condensed imidazolidine derivative, which is a compound (I) or a salt thereof, or a sulfoxide thereof represented by the formula: It has an excellent inhibitory effect on proteogliin degradation in the culture system, and has excellent properties as a pharmaceutical, such as an inhibitory effect on proteodalican release from knee cartilage in animal models. The present invention has been completed based on these findings. That is, the present invention
(1) 一般式 (1) General formula
[式中、 R1は、 一 (S) n— R2または— NR3R4を、 nは 0ないし 2の整数を、 R 2は水素原子、 置換されていてもよ,い炭化水素基または置換されていてもよい複 素環基を、 R3および R4は同一または異なって水素原子、 置換されていてもよい 炭化水素基、置換されていてもよい複素環基またはそれらが結合している窒素原子 と一緒に形成する含窒素複素環を、 R5は水素原子、 置換されていてもよい炭化水 素基、 シァノ基、 ァシル基、 エステル化もしくはアミド化されていてもよい力ルポ キシル基または置換されていてもよい複素環基を、 [Wherein, R 1 represents one (S) n —R 2 or —NR 3 R 4 , n represents an integer of 0 to 2, R 2 represents a hydrogen atom, or a substituted or unsubstituted hydrocarbon group. R 3 and R 4 may be the same or different and represent a hydrogen atom, a hydrocarbon group which may be substituted, a heterocyclic group which may be substituted or R 5 represents a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, an esterified or amidated nitrogen-containing heterocyclic ring formed together with a nitrogen atom A xyl group or an optionally substituted heterocyclic group,
を、 R6は、 水素原子、 置換されていてもよい炭化水素基、 シァノ基、 ァシル基、 エステル化もしくはアミド化されていてもよいカルボキシル基または置換されて いてもよい複素環基を、 R7は置換されていてもよい水酸基を、 R8および R9はそ れぞれ水素原子または置換されていてもよい炭化水素基を (R5と R6、 R5と R8 または R 8と R 9はそれぞれ一緒になつて置換されていてもよい環状炭化水素また は複素環を形成してもよい) 、 R 6 represents a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, a carboxyl group which may be esterified or amidated, or a heterocyclic group which may be substituted, 7 is an optionally substituted hydroxyl group, R 8 and R 9 are each a hydrogen atom or an optionally substituted hydrocarbon group (R 5 and R 6 , R 5 and R 8 or R 8 R 9 may be taken together to form an optionally substituted cyclic hydrocarbon or heterocyclic ring),
を、 R10は、 水素原子、 _ZR15 (Ζは一 S02—、 —SO—、 — CONR18SO 2— (R18は Ci— 6アルキルを示す) 、 一 CONR19— (R19は (: 6アルキルを 示す) または— CO—を、 R15は、 置換されていてもよい炭化水素基または置換 されていてもよい複素環基を示す) または— P (0) R16R17 (R 16および R 17 はそれぞれ置換されていてもよい炭化水素基、置換されていてもよい水酸基または 置換されていてもよいアミノ基を示す) を、 R11は水素原子または置換されてい てもよい炭化水素基を ( R 1 °と R 11は一緒になつて置換されていてもよい含窒素 複素環を形成してもよい) 、 R 12および R 13はそれぞれ水素原子または置換され ていてもよい炭化水素基、あるいはそれらが結合している窒素原子と一緒になつて 含窒素複素環を、 R14は、 水素原子、 置換されていてもよい炭化水素基または— Z 15 (Zおよび R 15は、 前記と同意義を示す) を示す。 但し、 R 5が水素原子、 X Y が The, R 10 is a hydrogen atom, _ZR 15 (Ζ one S0 2 -, -SO-, - CONR 18 SO 2 - (R 18 represents a CI- 6 alkyl), one CONR 19 - (R 19 is ( Or 6 represents an alkyl) or —CO—, and R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group) or — P (0) R 16 R 17 (R 16 and R 17 each represent an optionally substituted hydrocarbon group, an optionally substituted hydroxyl group or an optionally substituted amino group), and R 11 represents a hydrogen atom or an optionally substituted hydrocarbon group. A hydrogen group (R 1 ° and R 11 may be taken together to form a nitrogen-containing heterocyclic ring which may be substituted); R 12 and R 13 each represent a hydrogen atom or a carbon atom which may be substituted; hydrogen group, or a connexion nitrogen-containing heterocyclic ring such together with the nitrogen atom to which they are attached, R 14 is a hydrogen atom, Conversion are optionally substituted hydrocarbon group or also -. Z 15 (Z and R 15 are as defined above) shows the proviso, R 5 is a hydrogen atom, XY is
を示すとき、 R14は置換されていてもよい炭化水素基または一 ZR15 (Z、 R15 は、 前記と同意義を示す) を示す。 ]で表される化合物 (I) またはその塩または そのスルホキシド; In the formula, R 14 represents an optionally substituted hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described above). A compound (I) or a salt or a sulfoxide thereof represented by the formula:
(2) 一般式 (2) General formula
[式中、 R1は、 一 (S) n_R2または _NR3R4を、 nは 0ないし 2の整数を、 R2は水素原子、 置換されていてもよい炭化水素基または置換されていてもよい複 素環基を、 R 3および R4は同一または異なって水素原子、 置換されていてもよい 炭化水素基、置換されていてもよい複素環基またはそれらが結合している窒素原子 と一緒に形成する含窒素複素環を、 R 5は水素原子、 置換されていてもよい炭化水 素基、 シァノ基、 ァシル基、 エステル化もしくはアミド化されていてもよいカルボ キシル基または置換されていてもよい複素環基を、 は または Wherein R 1 is (S) n _R 2 or _NR 3 R 4 , n is an integer of 0 to 2, R 2 is a hydrogen atom, an optionally substituted hydrocarbon group or a substituted R 3 and R 4 may be the same or different and represent a hydrogen atom, an optionally substituted hydrocarbon group, an optionally substituted heterocyclic group, or a nitrogen atom to which they are bonded. And R 5 is a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, a carboxyl group which may be esterified or amidated, or a substituted heterocyclic ring. A heterocyclic group which may be Is or
R z 、R R z, R
'を、 R6は、 水素原子、 置換されていてもよい炭化水素基、 シァノ基、 ァシル基、 エステル化もしくはアミド化されていてもよい力ルポキシル基または置換されて いてもよい複素環基を、 R7は置換されていてもよい水酸基を、 R8および R9はそ れぞれ水素原子または置換されていてもよい炭化水素基を (R5と R6、 R5と R8 または R8と R 9はそれぞれ一緒になつて置換されていてもよい環状炭化水素また は複素環を形成してもよい) 、 R 6 represents a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, an esterified or amidated propyloxyl group or an optionally substituted heterocyclic group. R 7 is an optionally substituted hydroxyl group; R 8 and R 9 are each a hydrogen atom or an optionally substituted hydrocarbon group (R 5 and R 6 , R 5 and R 8 Or R 8 and R 9 may be taken together to form an optionally substituted cyclic hydrocarbon or heterocyclic ring),
を、 R1()は、 水素原子または _ZR15 (Zは— S02—または— CO—を、 R15 は、 置換されていてもよい炭化水素基または置換されていてもよい複素環基を示 す) を、 R 11は水素原子または置換されていてもよい炭化水素基を、 R12および R 13はそれぞれ水素原子または置換されていてもよい炭化水素基、 'あるいはそれ らが結合している窒素原子と一緒になつて含窒素複素環を、 R14は、 水素原子、 置換されていてもよい炭化水素基または一 ZR15 (Zおよび R15は、 前記と同意 義を示す) を示す。 伹し、 R 5が水素原子、 The, R 1 () is a hydrogen atom or _ZR 15 (Z - S0 2 - or - a CO-, R 15 is a optionally substituted hydrocarbon group or an optionally substituted heterocyclic group R 11 represents a hydrogen atom or an optionally substituted hydrocarbon group, R 12 and R 13 represent a hydrogen atom or an optionally substituted hydrocarbon group, respectively, or R 14 represents a hydrogen atom, an optionally substituted hydrocarbon group or one ZR 15 (Z and R 15 have the same meanings as described above) . And R 5 is a hydrogen atom,
を示すとき、 R14は置換されていてもよい炭化水素基または一 ZR15 (Z、 R15 は、 前記と同意義を示す) を示す。 ]で表される化合物 (I) .またはその塩または そのスルホキシド; In the formula, R 14 represents an optionally substituted hydrocarbon group or ZR 15 (Z and R 15 have the same meanings as described above). Or a salt thereof or a sulfoxide thereof;
(3) R 5が置換されていてもよい炭化水素基、 シァノ基、 ァシル基、 エステル化 もしくはアミド化されていてもよいカルボキシル基または置換されていてもよい 複素環基である前記 (1) または (2) 記載の化合物; (3) The above-mentioned (1), wherein R 5 is an optionally substituted hydrocarbon group, a cyano group, an acyl group, a carboxyl group which may be esterified or amidated, or an optionally substituted heterocyclic group. Or the compound according to (2);
(4) (Four)
[R6は、 水素原子、 置換されていてもよい炭化水素基,シァノ基、 ァシル基、 エス テル化もしくはアミド化されていてもよい力ルポキシル基または置換されていて もよい複素環基を、 R7は置換されていてもよい水酸基を、 R8および R9はそれぞ れ水素原子または置換されていてもよい炭化水素基を示す (R5と R6、 R5と R8 または R 8と R 9はそれぞれ一緒になつて置換されていてもよい環状炭化水素また は複素環を形成してもよい) ]である前記 (1) または (2) 記載の化合物; (5) [R 6 is a hydrogen atom, an optionally substituted hydrocarbon group, a cyano group, an acyl group, R 7 is an optionally substituted hydroxyl group or an optionally substituted heterocyclic group, R 7 is an optionally substituted hydroxyl group, and R 8 and R 9 are each a hydrogen atom or a substituted Represents an optionally substituted hydrocarbon group (R 5 and R 6 , R 5 and R 8, or R 8 and R 9 are taken together to form an optionally substituted cyclic hydrocarbon or heterocyclic ring, (5) the compound according to the above (1) or (2),
一 A One A
が But
[R10は、 水素原子、 一 ZR15 (Zは一 S02—、 一 SO—、 一 C〇NR18S〇2 一 (R18は(: 6アルキルを示す) 、 — CONR19— (R19は ^— 6アルキルを示 す) または一 CO—を、 R15は、 置換されていてもよい炭化水素基または置換さ れていてもよい複素環基を示す) または— P (O) R16R17 (R16および R17は それぞれ置換されていてもよい炭化水素基、置換されていてもよい水酸基または置 換されていてもよいアミノ基を示す) を、 R 11は水素原子または置換されていて もよい炭化水素基を (R 1 Qと R 11は一緒になつて置換されていてもよい含窒素複 素環を形成してもよい) 、 R 12および R 13はそれぞれ水素原子または置換されて いてもよい炭化水素基、あるいはそれらが結合している窒素原子と一緒になつて含 窒素複素環を示す]である前記 (1) 記載の化合物; [R 10 is a hydrogen atom, one ZR 15 (Z one S0 2 -, One SO-, One C_〇_NR 18 S_〇 2 one (R 18 is (: 6 represents an alkyl), - CONR 19 - (R 19 ^ - a indicates to) or single CO- 6 alkyl, R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group) or - P (O) R 16 R 17 (R 16 and R 17 each represent an optionally substituted hydrocarbon group, an optionally substituted hydroxyl group or an optionally substituted amino group), and R 11 represents a hydrogen atom or a substituted R 1 Q and R 11 may be taken together to form a nitrogen-containing compound ring which may be substituted, and R 12 and R 13 are each a hydrogen atom or (1) a hydrocarbon group which may be substituted, or a nitrogen-containing heterocyclic ring together with a nitrogen atom to which they are bonded]. A compound of
(6) (6)
一 A One A
が But
[R1C1は、 水素原子または _ZR15 (Zは一 S〇2—または— C〇一を、 R15は、 置換されていてもよい炭化水素基または置換されていてもよい複素環基を示す)を、 R 11は水素原子または置換されていてもよい炭化水素基を、 R 12および R 13はそ れぞれ水素原子または置換されていてもよい炭化水素基、あるいはそれらが結合し ている窒素原子と一緒になつて含窒素複素環を示す]である前記 (2) 記載の化合 物; . [R 1C1 represents a hydrogen atom or _ZR 15 (Z represents one S〇 2 — or —C〇 one, and R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group. ), R 11 is a hydrogen atom or an optionally substituted hydrocarbon group, and R 12 and R 13 are each a hydrogen atom or an optionally substituted hydrocarbon group, or a nitrogen atom to which they are bonded. Together with a nitrogen-containing heterocycle].
(7) 化合物 (I) が、 一般式 (7) When the compound (I) has the general formula
( )()
[記号は前記 (1) 記載と同意義を示す。 ]で表される化合物である前記 (1) 記載 の化合物; [The symbols are as defined in the above (1). ] The compound according to the above (1), which is a compound represented by the formula:
(8) 化合物 (I) が、 一般式 (8) When the compound (I) has the general formula
1 1
( ) ()
[記号は前記 (2) 記載と同意義を示す。 ]で表される化合物である前記 (2) 記載 の化合物; [The symbols are as defined in the above (2). ] The compound according to the above (2), which is a compound represented by the formula:
(9) R1Qが— S〇2R15 (R15は、置換されていてもよい炭化水素基または置換 されていてもよい複素環基を示す) であり、 R 5および R 6がそれぞれ置換されて いてもよい炭化水素基またはそれらが一緒になつて形成する置換されていてもよ い環状炭化水素または置換されていてもよい複素環で、 R 11が水素原子である前 記 (8) 記載の化合物; (9) R 1Q is — S〇 2 R 15 (R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group), and R 5 and R 6 are each substituted Or (8) a hydrocarbon group which may be substituted or an optionally substituted cyclic hydrocarbon or an optionally substituted heterocyclic ring which are formed together, and wherein R 11 is a hydrogen atom. A compound as described above;
(10) R1が、 一 SR2 (R2は、 水素原子、 置換されていてもよい炭化水素基ま たは置換されていてもよい複素環基を示す) である前記 (9) 記載の化合物; (1 1) R2が、 置換されていてもよい炭化水素基である前記 (1 0) 記載の化合 物; (10) The method according to the above (9), wherein R 1 is one SR 2 (R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group, or an optionally substituted heterocyclic group). Compound; (11) The compound according to the above (10), wherein R 2 is an optionally substituted hydrocarbon group;
(12) R5および R6が、 それぞれ置換されていてもよい炭素数 1ないし 6の炭 化水素基である前記 (9) ないし (1 1) 記載の化合物; (12) The compound according to the above (9) to (11), wherein R 5 and R 6 are each an optionally substituted hydrocarbon group having 1 to 6 carbon atoms;
(13) R6が、 メチルまたはェチルである前記 (12) 記載の化合物; (13) The compound according to the above (12), wherein R 6 is methyl or ethyl;
(14) R5および R6が一緒になつて置換されていてもよいベンゼン環を形成す る前記 (12) 記載の化合物; (14) The compound according to the above (12), wherein R 5 and R 6 together form an optionally substituted benzene ring;
(1 5) R15が、 置換されていてもよいァリール基、 置換されていてもよいァラ ルキル基または置換されていてもよい複素環基である前記 (9) ないし (14) 記 載の化合物; ( 15 ) The above-mentioned (9) to (14), wherein R 15 is an optionally substituted aryl group, an optionally substituted aralkyl group or an optionally substituted heterocyclic group. Compound;
(1 6) R15が、 置換されていてもよいフエニル基、 置換されていてもよいベン ジル基または置換されていてもよいチェニル基である前記 (1 5) 記載の化合物;(16) The compound according to the above ( 15 ), wherein R 15 is an optionally substituted phenyl group, an optionally substituted benzyl group or an optionally substituted phenyl group;
(17) R1が、 一 SR2 (R2は置換されていてもよい炭素数 1ないし 8のアルキ ル基、置換されていてもよい炭素数 2ないし 8のアルケニル基、置換されていても よい炭素数 3ないし 8のシクロアルキル基または置換されていてもよい炭素数 7 ないし 10のァラルキル基を示す)、 R15が、置換されていてもよいフエニル基、 置換されていてもよいベンジル基または置換されていてもよいチェニル基、 R5お よび R6がともにメチル基である前記 (9) 記載の化合物; (17) R 1 is one SR 2 (R 2 is an optionally substituted alkyl group having 1 to 8 carbon atoms, an alkenyl group optionally having 2 to 8 carbon atoms, A cycloalkyl group having 3 to 8 carbon atoms or an aralkyl group having 7 to 10 carbon atoms which may be substituted), wherein R 15 is an optionally substituted phenyl group, an optionally substituted benzyl group Or the optionally substituted phenyl group, the compound according to the above (9), wherein both R 5 and R 6 are methyl groups;
( 18 ) R2が置換されていてもよい炭素数 1ないし 8のアルキル基である前記( 1 7) 記載の化合物; (18) The compound according to the above (17), wherein R 2 is an optionally substituted alkyl group having 1 to 8 carbon atoms;
(1 9) R 2がハロゲン原子で置換されていてもよい炭素数 4ないし 8のアルキル 基である前記 (18) 記載の化合物; (19) The compound according to the above (18), wherein R 2 is an alkyl group having 4 to 8 carbon atoms which may be substituted with a halogen atom;
(20) R1Gがー COR15 (R15は、 置換されていてもよい炭化水素基または置 換されていてもよい複素環基を示す) であり、 R5および R6がそれぞれ置換され ていてもよい炭化水素基またはそれらが一緒になつて形成する置換されていても よい環状炭化水素または置換されていてもよい複素環で、 R11が水素原子である 前記 (8) 記載の化合物; (20) R 1G is —COR 15 (R 15 represents an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group), and R 5 and R 6 are each substituted. A hydrocarbon group which may be optionally substituted or an optionally substituted cyclic hydrocarbon or an optionally substituted heterocyclic ring which are formed together, wherein R 11 is a hydrogen atom;
(21) R1が、 一 SR2 (R2は、 水素原子、 置換されていてもよい炭化水素基ま たは置換されていてもよい複素環基を示す) である前記 (20) 記載の化合物; (22) R2が、 置換されていてもよい炭化水素基である前記 (21) 記載の化合 物; (21) The method according to the above (20), wherein R 1 is one SR 2 (R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group). Compound; (22) The compound according to the above (21), wherein R 2 is an optionally substituted hydrocarbon group;
(23) R5および R6が、 それぞれ置換されていてもよい炭素数 1ないし 6の炭 化水素基である前記 (20) ないし (22) 記載の化合物; (23) The compound according to the above (20) to (22), wherein R 5 and R 6 are each an optionally substituted hydrocarbon group having 1 to 6 carbon atoms;
(24) R6が、 メチルまたはェチルである前記 (23) 記載の化合物; (24) The compound according to the above (23), wherein R 6 is methyl or ethyl;
(25) R 5および R 6が一緒になつて置換されていてもよいベンゼン環を形成す る前記 (23) 記載の化合物; (25) The compound according to the above (23), wherein R 5 and R 6 together form an optionally substituted benzene ring;
(26) R15が、 置換されていてもよいァリール基、 置換されていてもよいァラ ルキル基または置換されていてもよい複素環基である前記 (20) ないし (25) 記載の化合物; (26) The compound according to any of the above (20) to (25), wherein R 15 is an optionally substituted aryl group, an optionally substituted aralkyl group or an optionally substituted heterocyclic group;
(27) R15が、 置換されていてもよいピリジル基である前記 (26) 記載の化 合物; (27) The compound according to the above (26), wherein R 15 is an optionally substituted pyridyl group;
(28) 4—プロモー N— {8— 〔 (5—フルォロペンチル)スルファニル〕 一 5, 6—ジメチルー 3—ォキソ—2, 3—ジヒドロ— 1H—イミダゾ [5, l_c][l, 4]チアジン _ 1—イリデン } ベンゼンスルホンアミド、 N— { 5, 6ージメチル - 3一ォキソ一 8— 〔 (4, 4, 4, 5, 5 _ペンタフルォロペンチル) スルファ ニル〕 —2, 3—ジヒドロー 1H—イミダゾ [5, 1 -c] [1, 4]チアジン一 1一 イリデン } (フエニル) メタンスルホンアミド、 N— {5, 6ージメチル— 3—ォ キソー 8— 〔 (4, 4, 4, 5, 5一ペン夕フルォロペンチル) スルファニル〕 ― 2, 3—ジヒドロ— 1H—イミダゾ [5, 1 - c] [1, 4]チアジン一 1一イリデン } —4—メチルベンゼンスルホンアミド、 4—ブロモー N— 〔8_ (イソペンチルス ルファ二ル) — 5, 6—ジメチル— 3_ォキソ一 2, 3—ジヒドロ— 1H—イミダ ゾ [5, 1 _ c] [1, 4]チアジン一 1.—イリデン〕 ベンゼンスルホンアミド、 また はそれらの塩; (28) 4-Promote N— {8— [(5-Fluoropentyl) sulfanyl] -1-5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, l_c] [l, 4] thiazine _ 1-ylidene} benzenesulfonamide, N— {5,6-dimethyl-3-oxo-1 8 — [(4,4,4,5,5_pentafluoropentyl) sulfanyl] —2,3-dihydro-1H —Imidazo [5,1-c] [1,4] thiazine-1-1ylidene} (phenyl) methanesulfonamide, N— {5,6-dimethyl-3-oxo-8— [(4,4,4,5 , 5 pentafluorofluoropentyl) sulfanyl] ― 2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-111-ylidene} —4-methylbenzenesulfonamide, 4-bromo-N— [8_ (Isopentylsulfanyl) — 5,6-dimethyl-3, oxo-1,2,3-dihydro-1H-imidazo [5,1_c] [ 1,4] thiazine 1.-ylidene] benzenesulfonamide or a salt thereof;
(29) 前記 (1) 記載の化合物 (I) またはその塩またはそのスルホキシドのプ ロドラッグ; (29) a prodrug of the compound (I) according to the above (1) or a salt thereof or a sulfoxide thereof;
(30) 前記 (1) 記載の化合物 (I) またはその塩またはそのスルホキシドまた はそのプロドラッグを含有することを特徴とする医薬組成物; (30) A pharmaceutical composition comprising the compound (I) according to the above (1) or a salt thereof, a sulfoxide thereof or a prodrug thereof;
(31) 軟骨基質分解酵素産生抑制剤である前記 (30) 記載の医薬組成物; (32) プロテオダリカン分解抑制剤である前記 (30) 記載の医薬組成物; (33) 変形性関節症予防治療剤である前記 (30) 記載の医薬組成物; (31) the pharmaceutical composition according to (30), which is a cartilage matrix degrading enzyme production inhibitor; (32) The pharmaceutical composition according to (30), which is a proteodalican degradation inhibitor; (33) the pharmaceutical composition according to (30), which is a osteoarthritis preventive or therapeutic agent;
(34)軟骨破壊に起因する疾患予防治療剤である前記(30)記載の医薬組成物; (35) マトリックスメタ口プロティナ一ゼ遺伝子発現抑制剤である前記 (30) 記載の医薬組成物; (34) The pharmaceutical composition according to the above (30), which is an agent for preventing or treating a disease caused by cartilage destruction; (35) the pharmaceutical composition according to the above (30), which is a matrix meta-oral proteinase gene expression inhibitor;
(36) マトリックスメタ口プロティナーゼ— 13遺伝子発現抑制剤である前記 (30) 記載の医薬組成物; (36) The pharmaceutical composition according to the above (30), which is a matrix meta-oral proteinase-13 gene expression inhibitor;
(37)縮合ィミダゾリジン誘導体を含有することを特徴とするプロテオグリ力ン 分解抑制剤; (37) a proteoglitin degradation inhibitor comprising a condensed imidazolidine derivative;
(38)縮合イミダゾリジン誘導体を含有することを特徴とする軟骨基質分解酵素 産生抑制剤; (38) a cartilage matrix degrading enzyme production inhibitor comprising a condensed imidazolidine derivative;
(39) 縮合イミダゾリジン誘導体を含有することを特徴とする変形性関節症予 防 ·治療剤; (39) a prophylactic / therapeutic agent for osteoarthritis, comprising a condensed imidazolidine derivative;
(40)縮合ィミダゾリジン誘導体を含有することを特徴とする軟骨破壊に起因す る疾患予防 ·治療剤; (40) an agent for preventing and treating diseases caused by cartilage destruction, characterized by containing a condensed imidazolidine derivative;
(41) 前記 (1) 記載の化合物 (Γ) またはその塩またはそのスルホキシドまた はそのプロドラッグの有効量を哺乳動物に投与することを特徴とする変形性関節 症予防 ·治療方法; (41) A method for preventing or treating osteoarthritis, which comprises administering to a mammal an effective amount of the compound (II) or a salt thereof or a sulfoxide or a prodrug thereof according to (1);
(42) 前記 (1) 記載の化合物 (I) またはその塩またはそのスルホキシドまた はそのプロドラッグの有効量を哺乳動物に投与することを特徴とする軟骨破壊に 起因する疾患の予防 ·治療方法; (42) a method for preventing or treating a disease caused by cartilage destruction, which comprises administering an effective amount of the compound (I) or a salt thereof or a sulfoxide or a prodrug thereof according to the above (1) to a mammal;
(43)変形性関節症予防 ·治療剤を製造するための前記(1)記載の化合物(I) またはその塩またはそのスルホキシドまたはそのプロドラッグの使用; (43) Use of the compound (I) according to the above (1) or a salt thereof or a sulfoxide or a prodrug thereof for the manufacture of a prophylactic or therapeutic agent for osteoarthritis;
(44)軟骨破壊に起因する疾患の予防 ·治療剤を製造するための前記 (1) 記載 の化合物(I)またはその塩またはそのスルホキシドまたはそのプロドラッグの使 用; (44) Use of the compound (I) or a salt thereof or a sulfoxide or a prodrug thereof according to the above (1) for producing an agent for preventing or treating a disease caused by cartilage destruction;
(45) 式 Equation (45)
[記号は前記 (1) 記載と同意義を示す]で表される化合物またはその塩と、 式 Wherein the symbol is as defined in the above (1), or a salt thereof;
[Lは脱離基を、 R5および R6は前記 (1) 記載と同意義を示す]で表される化合 物またはその塩とを環形成反応に付し、所望により加水分解反応、 脱水反応、 アル キル化反応、 置換反応または/および酸化反応を行うことを特徴とする一般式 [L is a leaving group, and R 5 and R 6 have the same meanings as described in the above (1)] or a salt thereof, and then subjected to a ring-forming reaction, and if desired, a hydrolysis reaction and a dehydration reaction. General formula characterized by conducting a reaction, an alkylation reaction, a substitution reaction and / or an oxidation reaction.
[記号は前記(1)記載と同意義を示す]で表される化合物またはその塩またはその スルホキシドの製造法; [The symbols are as defined in the above (1)], or a method for producing a compound thereof, a salt thereof, or a sulfoxide thereof;
等に関する。 Etc.
本明細書において、 R1は、 ― (S) n— R2または—NR3R4を、 nは 0ないし 2の整数を、 R2は水素原子、 置換されていてもよい炭化水素基または置換されて いてもよい複素環基を、 R3および R4は同一または異なって水素原子、 置換され ていてもよい炭化水素基、置換されていてもよい複素環基またはそれらが結合して いる窒素原子と一緒に形成する含窒素複素環を示す。 In the present specification, R 1 represents — (S) n —R 2 or —NR 3 R 4 , n represents an integer of 0 to 2, R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group or R 3 and R 4 are the same or different hydrogen atoms, an optionally substituted hydrocarbon group, an optionally substituted heterocyclic group or an optionally substituted heterocyclic group, A nitrogen-containing heterocyclic ring formed together with a nitrogen atom is shown.
R2で表される置換されていてもよい炭化水素基の 「炭化水素基」 としては、 ァ ルキル基、 アルケニル基、 アルキニル基、 ァリール基、 ァラルキル基、 シクロアル キル基、 シクロアルケニル基、 などが挙げられる。 Examples of the “hydrocarbon group” of the optionally substituted hydrocarbon group represented by R 2 include an alkyl group, an alkenyl group, an alkynyl group, an aryl group, an aralkyl group, and a cycloalkyl group. A kill group, a cycloalkenyl group, and the like.
置換されていてもよい炭化水素基としての 「アルキル基」 の例としては、 炭素数 1ないし 20のアルキル基 (例えばメチル、 ェチル、 プロピル、 イソプロピル、 ブ チル、 イソブチル、 sec-ブチル、 tert-ブチル、 ペンチル、 イソペンチル、 ネオべ ンチル、 tert-ペンチル、 1-メチルプチル、 2 -メチルブチル、 1, 2-ジメチルプ 口ピル、 1 -ェチルプロピル、 へキシル、 イソへキシル、 1 -メチルペンチル、 2 - メチルペンチル、 3-メチルペンチル、 1, 1-ジメチルブチル、 1, 2-ジメチルブ チル、 2, 2-ジメチルブチル、 3, 3-ジメチルブチル、 1, 3 -ジメチルブチル、 2, 3-ジメチルブチル、 1 -ェチルブチル、 2-ェチルブチル、 1, 1, 2-トリメチルプ 口ピル、 1, 2, 2-トリメチルプロピル、 1 -ェチル- 1 -メチルプロピル、 1-ェチ ル -2-メチルプロピル、 ヘプチル、 イソへプチル、 1 -メチルへキシル、 2-メチル へキシル、 3-メチルへキシル、 4-メチルへキシル、 1-ェチルペンチル、 2 -ェチ ルペンチル、 3-ェチルペンチル、 1 -プロピルプチル、 1, 1-ジメチルペンチル、 1, 2-ジメチルペンチル、 1, 3-ジメチルペンチル、 1, 4-ジメチルペンチル、 2, 2 -ジメチルペンチル、 2, 3-ジメチルペンチル、 2, 4 ジメチルペンチル、 3, Examples of the “alkyl group” as the optionally substituted hydrocarbon group include an alkyl group having 1 to 20 carbon atoms (eg, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl) , Pentyl, isopentyl, neopentyl, tert-pentyl, 1-methylbutyl, 2-methylbutyl, 1,2-dimethylpropyl, 1-ethylpropyl, hexyl, isohexyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 2,2-dimethylbutyl, 3,3-dimethylbutyl, 1,3-dimethylbutyl, 2,3-dimethylbutyl, 1-ethylbutyl , 2-ethylbutyl, 1,1,2-trimethylpropyl pill, 1,2,2-trimethylpropyl, 1-ethyl-1-methylpropyl, 1-ethyl-2-methylpropyl Heptyl, isoheptyl, 1-methylhexyl, 2-methylhexyl, 3-methylhexyl, 4-methylhexyl, 1-ethylpentyl, 2-ethylpentyl, 3-ethylpentyl, 1-propylbutyl, 1 1,1-dimethylpentyl, 1,2-dimethylpentyl, 1,3-dimethylpentyl, 1,4-dimethylpentyl, 2,2-dimethylpentyl, 2,3-dimethylpentyl, 2,4-dimethylpentyl, 3,
3-ジメチルペンチル、 3, 4-ジメチルペンチル、 4, 4-ジメチルペンチル、 1, 1 , 2 _トリメチルプチル、 1, 1, 3 -トリメチルプチル、 1, 2 , 2 -トリメチルプチ ル、 1, 2, 3 トリメチルブチル、 1, 3, 3 トリメチルプチル、 2, 2, 3 トリメ チルブチル、 2, 3, 3-トリメチルプチル、 1-ェチル -1-メチルブチル、 1-ェチ ル -2-メチルブチル、 1 -ェチル- 3 -メチルブチル、 2 -ェチル- 1 -メチルブチル、 2 -ェチル -2-メチルプチル、 2-ェチル _ 3 _メチルプチル、 ォクチル、 イソォクチ ル、 1-メチルヘプチル、 2-メチルヘプチル、 3-メチルヘプチル、 4 -メチルヘプ チル、 5 -メチルヘプチル、 1-ェチルへキシル、 2-ェチルへキシル、 3-ェチルへ キシル、 4 -ェチルへキシル、 1-プロピルペンチル、 2 -プロピルペンチル、 1 , 1 -ジメチルへキシル、 2, 2-ジメチルへキシル、 3, 3-ジメチルへキシル、 4,3-dimethylpentyl, 3,4-dimethylpentyl, 4,4-dimethylpentyl, 1,1,2-trimethylbutyl, 1,1,3-trimethylbutyl, 1,2,2-trimethylbutyl, 1,2 1,3,3-methylbutyl, 1,3,3-trimethylbutyl, 2,2,3-trimethylbutyl, 2,3,3-trimethylbutyl, 1-ethyl-1-methylbutyl, 1-ethyl-2-methylbutyl, 1- Ethyl-3-methylbutyl, 2-ethyl-1-methylbutyl, 2-ethyl-2-methylbutyl, 2-ethyl_3-methylbutyl, octyl, isooctyl, 1-methylheptyl, 2-methylheptyl, 3-methylheptyl, 4-methylheptyl, 5-methylheptyl, 1-ethylhexyl, 2-ethylhexyl, 3-ethylhexyl, 4-ethylhexyl, 1-propylpentyl, 2-propylpentyl, 1,1-dimethyl Xyl, 2,2-dimethyl Hexyl, 3, hexyl 3-dimethylaminopropyl, 4,
4-ジメチルへキシル、 5, 5-ジメチルへキシル、 3 -ェチル- 3 -メチルペンチル、 1, 1 -ジェチルブチル、 2, 2 -ジ'ェチルプチル、 1, 1, 2, 2-テ卜ラメチルブチル、 1, 1, 3, 3-テトラメチルプチレ、 2, 2, 3, 3-テトラメチルブチル、 1 , 1 -ジメ チル -2-ェチルプチル、 ノニル、 イソノニル、 1 -イソプチル- 3 -メチルブチル、 デ シル、 イソデシル、 ゥンデシル、 イソゥンデシル、 ドデシル、 イソドデシル、 トリ デシル、 イソトリデシル、 テトラデシル、 イソテトラデシル、 ペン夕デシル、 イソ ペン夕デシル、 へキサデシル、 イソへキサデシル、 ヘプ夕デシル、 イソヘプ夕デシ ル、 ォク夕デシル、 イソォクタデシル、 ノナデシル、 イソノナデシルなど) が挙げ られ、 好ましくは炭素数 1ないし 10のアルキル基 (例えば、 メチル、 ェチル、 プ 口ピル、 イソプロピル、 ブチル、 イソブチル、 sec-ブチル、 tert-ブチル、 ペンチ ル、イソペンチル、 2-メチルブチル、へキシル、イソへキシル、 2-ェチルプチル、 1, 1-ジメチルブチル、 2, 2-ジメチルブチル、 3, 3 -ジメチルブチル、 ヘプチル、 イソ ヘプチル、 1-メチルへキシル、 1-ェチルペンチル、 3-ェチルペンチル、 1-プロピ ルプチル、 ォクチル、 イソォクチル、 2 -プロピルペンチル、 ノエル、 イソノエル、4-dimethylhexyl, 5,5-dimethylhexyl, 3-ethyl-3-methylpentyl, 1,1-ethylethylbutyl, 2,2-di'ethylbutyl, 1,1,2,2-tetramethylbutyl, 1 1,1,3,3-tetramethylbutyle, 2,2,3,3-tetramethylbutyl, 1,1-dimethyl-2-ethylbutyl, nonyl, isononyl, 1-isobutyl-3-methylbutyl, Syl, isodecyl, pendecyl, isopendecyl, dodecyl, isododecyl, tridecyl, isotridecyl, tetradecyl, isotetradecyl, pendudecyl, isopendudecyl, hexadecyl, isohexadecyl, hepdudecyl, isohepdecyl, octyldecyl Decyl, isooctadecyl, nonadecyl, isononadecyl and the like, and preferably an alkyl group having 1 to 10 carbon atoms (eg, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl) , Isopentyl, 2-methylbutyl, hexyl, isohexyl, 2-ethylbutyl, 1,1-dimethylbutyl, 2,2-dimethylbutyl, 3,3-dimethylbutyl, heptyl, isoheptyl, 1-methylhexyl , 1-ethylpentyl, 3-ethyl Pentyl, 1-propyl Rupuchiru, Okuchiru, Isookuchiru, 2 - propyl pentyl, Noel, Isonoeru,
1-イソブチル -3-メチルプチル、 デシル、 イソデシルなど) が挙げられる。 1-isobutyl-3-methylbutyl, decyl, isodecyl, etc.).
置換されていてもよい炭化水素基としての 「アルケニル基」 としては、 炭素数 2 ないし 20のアルケニル基 (例えばビニル、 ァリル、 イソプロべニル、 1 -プロべ ニル、 1 ブテニル、 2-ブテニル、 3 -ブテニル、 1, 3-ブ夕ジェニル、 1 -メチル- 1-プロぺニル、 1-メチル -2-プロぺニル、 2 -メチル -1-プロぺニル、 2_メチル - 2-プロぺニル、 1 -ペンテニル、 2 -ペンテニル、 3 -ペンテニル、 4-ペンテニル、 1, 3-ペン夕ジェニル、 2, 4-ペン夕ジェニル、 1,4-ペンタジェニル、 1-メチル - 1 - ブテニル、 2 -メチル -1-ブテニル、 3 -メチル -1-ブテニル、 1_メチル -2-ブテ ニル、 2-メチル -2-ブテニル、 3 -メチル -2-ブテニル、 1 -メチル -3-ブテニル、 2 -メチル -3-ブテニル、 3 -メチル -3-ブテニル、 1-ェチル -1-プロぺニル、 1 - ェチル -2-プロぺニル、 1 -へキセニル、 2 _へキセニル、 3-へキセニル、 4_へキ セニル、 5-へキセニル、 1, 3-へキサジェニル、 1, 4-へキサジェニル、 1,5 -へキサ ジェニル、 2, 4 -へキサジェニル、 2, 5-へキサジェニル、 1 -メチル- 1 -ペンテニル、 “Alkenyl group” as the optionally substituted hydrocarbon group includes alkenyl groups having 2 to 20 carbon atoms (for example, vinyl, aryl, isoprobenyl, 1-probenyl, 1-butenyl, 2-butenyl, 3 -Butenyl, 1,3-butenyl, 1-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-1-propenyl, 2_methyl-2-propenyl 1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1,3-pentenyl, 2,4-pentenyl, 1,4-pentagenenyl, 1-methyl-1-butenyl, 2-methyl 1-butenyl, 3-methyl-1-butenyl, 1_methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl, 1-methyl-3-butenyl, 2-methyl 3-butenyl, 3-methyl-3-butenyl, 1-ethyl-1-propenyl, 1-ethyl-2-propenyl, 1-hexe 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 1,3-hexenyl, 1,4-hexenyl, 1,5-hexenyl, 2,4-hexyl Xaxenyl, 2,5-hexagenyl, 1-methyl-1-pentenyl,
2 -メチル -1-ペンテニル、 3-メチル -1-ペンテニル、 4-メチル -1-ペンテニル、 1 -メチル -2-ペンテニル、 2 -メチル -2-ペンテニル、 3 -メチル -2-ペンテニル、 4- メチル -2-ペンテニル、 1 -メチル -3-ペンテニル、 2 -メチル -3-ペンテニル、 3 -メ チル- 3-ペンテニル、 4 -メチル -3-ペンテニル、 1-メチル -4-ペンテニル、 2-メチ ル- 4-ペンテニル、 3-メチル -4-ペンテニル、 4_メチル -4-ペンテニル、 1 -ェチル- 1-ブテニル、 2 -ェチル- 1 -プテニル、 卜ェチル -2-ブテニル、 2-ェチル -2-ブテ ニル、 1-ェチル -3 -プテニル、 2-ェチル -3-ブテニル、 1-ヘプテニル、 2-ヘプテニ ル、 3 -へプテニル、 4 -ヘプテュル、 5-ヘプテニル、 6 -へプテニル、 1, 3 -へプタジ ェニル、 1, 4-ヘプ夕ジェニル、 1, 5-ヘプ夕ジェニル、 1,6-ヘプタジェニル、 2,4- ヘプタジェ二リレ、 2, 5 -ヘプタジェニル、 2, 6-ヘプタジェニル、 3, 5-ヘプタジェニル、 3,6-ヘプタジェニル、 4, 6 -ヘプタジェニル、 1-メチル- 1-へキセニル、 1-メチル - 2-へキセニル、 1 -メチル 3-へキセニル、 1-メチル -4-へキセニル、 1-メチ ル -5-へキセニル、 2-メチル -1-へキセニル、 2-メチル -2 -へキセニル、 2-メチル - 3-へキセニル、 2-メチル -4-へキセニル、 2-メチル -5-へキセニル、 3-メチル-2-Methyl-1-pentenyl, 3-methyl-1-pentenyl, 4-methyl-1-pentenyl, 1-methyl-2-pentenyl, 2-methyl-2-pentenyl, 3-methyl-2-pentenyl, 4- Methyl-2-pentenyl, 1-methyl-3-pentenyl, 2-methyl-3-pentenyl, 3-methyl-3-pentenyl, 4-methyl-3-pentenyl, 1-methyl-4-pentenyl, 2-methyl 4-pentenyl, 3-methyl-4-pentenyl, 4_methyl-4-pentenyl, 1-ethyl-1-butenyl, 2-ethyl-1-butenyl, triethyl-2-butenyl, 2-ethyl-2 -Bute Nil, 1-ethyl-3-butenyl, 2-ethyl-3-butenyl, 1-heptenyl, 2-heptenyl, 3-heptenyl, 4-heptenyl, 5-heptenyl, 6-heptenyl, 1,3-h Ptadenyl, 1,4-heptagenyl, 1,5-heptagenyl, 1,6-heptagenyl, 2,4-heptaphenyl, 2,5-heptagenyl, 2,6-heptagenyl, 3,5-heptagenyl , 3,6-heptagenyl, 4,6-heptagenyl, 1-methyl-1-hexenyl, 1-methyl-2-hexenyl, 1-methyl 3-hexenyl, 1-methyl-4-hexenyl, 1- Methyl-5-hexenyl, 2-methyl-1-hexenyl, 2-methyl-2-hexenyl, 2-methyl-3-hexenyl, 2-methyl-4-hexenyl, 2-methyl-5- Hexenyl, 3-methyl-
1 -へキセニル、 3-メチル -2-へキセニル、 3-メチル -3-へキセニル、 3 -メチル 4-へキセニル、 3-メチル -5-へキセニル、 4-メチル -1-へキセニル、 4-メチル-1-hexenyl, 3-methyl-2-hexenyl, 3-methyl-3-hexenyl, 3-methyl 4-hexenyl, 3-methyl-5-hexenyl, 4-methyl-1-hexenyl, 4 -Methyl-
2-へキセニル、 4-メチル -3-へキセニル、 4 -メチル -4-へキセニル、 4-メチル- 5-へ午セニル、 5 -メチル -1-へキセニル、 5-メチル -2-へキセニル、 5 -メチル-2-hexenyl, 4-methyl-3-hexenyl, 4-methyl-4-hexenyl, 4-methyl-5-hexenyl, 5-methyl-1-hexenyl, 5-methyl-2-hexenyl , 5-Methyl-
3 -へキセニル、 5-メチル- 4 -へキセニル、 5-メチル- 5 -へキセニル、 1-メチル- 2, 4 - へキサジェニル、 2-メチル -2, 4-へキサジェニル、 3 -メチル -2, 4-へキサジェニル、 4-メチル -2, 4-へキサジェニル、 5-メチル -2, 4-へキサジェニル、 5-メチル -2, 5 -へ キサジェニル、 1-ェチル -1-ペンテニル、 2-ェチル -1-ペンテニル、 3-ェチル- 1 -ペンテニル、 1 -ェチリレ -2-ペンテニル、 2-ェチル -2-ペンテニル、 3-ェチル -2- ペンテニル、 1-ェチル -3-ペンテニル、 2 -ェチル -3-ペンテニル、 3-ェチル -3-ぺ ンテニル、 1-ェチル -4-ペンテニル、 2-ェチル -4-ペンテニル、 3-ェチル -4-ペン テニル、 1-プロピル- 1-ブテニル、 1 -プロピル- 2-ブテニル、 1-プロピル- 3-ブテニ ル、 2 -ォクテニル、 3-ォクテニル、 4-ォクテニル、 5-ォクテニル、 6-ォクテニル、 7-ォクテニル、 卜メチル -2-ヘプテニル、 1-メチル -3-ヘプテニル、 1-メチル -4 -へ プテニル、 1 -メチル -5-ヘプテニル、 1-メチル -6-ヘプテニル、 6-メチル -2-ヘプテ ニル、 6-メチル -3-ヘプテニル、 6-メチル -4-ヘプテニル、 6-メチル -5-ヘプテニル、 6-メチル -6-ヘプテニル、 6-メチル -3, 5-ヘプ夕ジェニル、 6-メチル -2, 5-ヘプタジ ェニル、 4 -ェチル -2-へキセニル、 4-ェチル -3-へキセニル、 2 -ノネニル、 8-ノネ二 ル、 7-メチル -1-ォクテニル、 7-メチル -2-ォクテニル、 7-メチル -3-ォクテニル、 7 -メチル -4-ォクテニル、 7 -メチル -5-ォクテニル、 7-メチル -6 -ォクテニル、 7 -メ チル- 7-ォクテニル、 エチル -2-ヘプテニル、 1-ェチル -3-ヘプテニル、 1-ェチル - 4 -へプテニル、 卜ェチル -5-ヘプテニル、 1-ェチル -6-ヘプテニル、 5-ェチル -2- ヘプテニル、 5-ェチル -3-ヘプテニル、 5-ェチル -4-ヘプテニル、 5-ェチル -5-ヘプ テニル、 5-ェチル -6-ヘプテニル、 2-デセニル、 9-デセニル、 8-メチル -2-ノネニル、 、 8 -メチル -3-ノネニル、 8-メチル -4 -ノネニル、 8 -メチル- 5-ノネニル、 8 -メチル -6 - ノネニル、 8 -メチル -7-ノネニル、 8-メチル -8-ノネニル、 3, 7-ジメチル -2, 6-ォク タジェニル、 2-ゥンデセニル、 10-ゥンデセニル、 9-メチル -2-デセニル、 9-メチル -8-デセニル、 9-メチル -9 -デセニル、 2-ドデセニル、 1卜ドデセニル、 10-メチル -2 - ゥンデセニル、 10-メチル -9-ゥンデセニル、 10-メチル -10-ゥンデセニル、 2-トリ デセニル、 12-トリデセニル、 1卜メチル -2-ドデセニル、 11-メチル -10-ドデセニル、 11-メチル -1卜ドデセニル、 2-テ卜ラデセニル、 13 -テトラデセニル、 12-メチル _2 - トリデセニル、 12-メチル -11-トリデセニル、 12-メチル -12-トリデセニル、 2 -ペン 夕デセニル、 14-ペンタデセニル、 13-メチル -2-テトラデセニル、 13 -メチル -12- テトラデセニル、 13 -メチル -13-テトラデセエル、 3, 7, 1卜トリメチル- 2, 6, 10-ドデ カトリェニル、 2 -へキサデセニル、 15-へキサデセニル、 14-メチル -2-ペン夕デセ ニル、 14-メチル -13-ペンタデセニル、 14-メチル -14-ペンタデセニル、 2-ヘプ夕デ セニル、 16 -ヘプタデセニル、 15-メチル -2-へキサデセニル、 15-メチル -14-へキサ デセニル、 15-メチル -15-へキサデセニル、 2-ォクタデセニル、 17-ォクタデセニル、 9, 12-ォクタデカジエニル、 9, 12, 15-ォクタデカトリェニル、 9, 11 , 13-ォクタデカ トリェニル、 16-メチル -2-ヘプタデセニル、 16-メチル -15-ヘプタデセニル、 16- メチル- 16-へプ夕デセニル、 2-ノナデセニル、 18 -ノナデセニル、 17 -メチル -2-ォ クタデセニル、 17 -メチル -16-ォク夕デセニル、 17 -メチル -17-ォクタデセニル、 2- エイコセニル、 19-エイコセニル、 5, 8, 1 1 , 14-ィコサテトラエニル、 18 メチル -2- ノナデセニル、 18 -メチル -17-ノナデセニル、 18 -メチル -18 -ノナデセニル、 3, 7, 11, 15 -テトラメチル- 2, 6, 10, 14 -ドデカテトラェニルなど) が挙げられ、 好ま しくは炭素数 2ないし 1 0のアルケニル基(例えばビエル、 ァリル、 イソプロべ二 ル、 2 -メチル - 1 -プロぺニル、 3 -ブテニル、 4 -ペンテニル、 3 -メチル - 2 -ブテ ニル、 3 -メチル -3-ブテニル、 5-へキセニル、 4 -メチル -2 -ペンテニル、 4-メチル - 3 -べンテニル、 4-メチル- 4. -べンテニル、 6-ヘプテュル、 5-メチル- 2 -へキセニル、 5 -メチル- 3 -へキセニル、 5-メチル- 4 -へキセニル、 5 -メチル - 5 -へキセニル、 5- メチル -2, 4-へキサジェニル、 7-ォクテニル、 6 -メチル -2-ヘプテニル、 6-メチル- 3 - ヘプテニル、 6-メチル -4-ヘプテニル、 6-メチル -5-ヘプテニル、 6-メチル -6-ヘプ テニル、 8-ノネニル、 9-デセニル、 3, 7-ジメチル- 2, 6-ォク夕ジェニルなど) が挙 げられる。 3-hexenyl, 5-methyl-4-hexenyl, 5-methyl-5-hexenyl, 1-methyl-2,4-hexhexenyl, 2-methyl-2,4-hexenyl, 3-methyl-2 , 4-Hexenyl, 4-methyl-2, 4-hexenyl, 5-methyl-2, 4-hexenyl, 5-methyl-2,5-hexexenyl, 1-ethyl-1-pentenyl, 2-ethyl 1-pentenyl, 3-ethyl-1-pentenyl, 1-ethylile-2-pentenyl, 2-ethyl-2-pentenyl, 3-ethyl-2-pentenyl, 1-ethyl-3-pentenyl, 2-ethyl-3 -Pentenyl, 3-ethyl-3-pentenyl, 1-ethyl-4-pentenyl, 2-ethyl-4-pentenyl, 3-ethyl-4-pentenyl, 1-propyl-1-butenyl, 1-propyl-2 -Butenyl, 1-propyl-3-butenyl, 2-octenyl, 3-octenyl, 4-octenyl, 5-octenyl, 6-octenyl, 7-octyl Thenyl, trimethyl-2-heptenyl, 1-methyl-3-heptenyl, 1-methyl-4-heptenyl, 1-methyl-5-heptenyl, 1-methyl-6-heptenyl, 6-methyl-2-heptenyl , 6-methyl-3-heptenyl, 6-methyl-4-heptenyl, 6-methyl-5-heptenyl, 6-methyl-6-heptenyl, 6-methyl-3,5-heptenyl, 6-methyl-2 , 5-heptadienyl, 4-ethyl-2-hexenyl, 4-ethyl-3-hexenyl, 2-nonenyl, 8-nonenyl, 7-methyl-1-octenyl, 7-methyl-2-octenyl, 7-methyl-3-octenyl, 7-methyl-4-octenyl, 7-methyl-5-octenyl, 7-methyl-6-octenyl, 7-methyl-7-octenyl, ethyl-2-heptenyl, 1-ethyl -3-heptenyl, 1-ethyl -4 -Heptenyl, Tetyl-5-heptenyl, 1-Ethyl-6-heptenyl, 5-Ethyl-2-heptenyl, 5-Ethyl-3-heptenyl, 5-Ethyl-4-heptenyl, 5-Ethyl-5 -Heptenyl, 5-ethyl-6-heptenyl, 2-decenyl, 9-decenyl, 8-methyl-2-nonenyl, 8-methyl-3-nonenyl, 8-methyl-4-nonenyl, 8-methyl-5 -Nonenyl, 8-methyl-6-nonenyl, 8-methyl-7-nonenyl, 8-methyl-8-nonenyl, 3,7-dimethyl-2,6-octagenyl, 2-p-decenyl, 10-p-decenyl, 9 -Methyl-2-decenyl, 9-methyl-8-decenyl, 9-methyl-9-decenyl, 2-dodecenyl, 1-dodecenyl, 10-methyl-2-didecenyl, 10-methyl-9-dendecenyl, 10-methyl -10-Pendecenyl, 2-tridecenyl, 12-tridecenyl, 1-methyl-2-dodecenyl, 11-methyl-10-dodecenyl , 11-methyl-1-tododecenyl, 2-tetradecenyl, 13-tetradecenyl, 12-methyl_2-tridecenyl, 12-methyl-11-tridecenyl, 12-methyl-12-tridecenyl, 2-pentenodecenyl, 14 -Pentadecenyl, 13-methyl-2-tetradecenyl, 13-methyl-12-tetradecenyl, 13-methyl-13-tetradecenyl, 3,7,1 trimethyl-2,6,10-dodecatrienyl, 2-hexadecenyl, 15-hexadecenyl, 14-methyl-2-pentadecenyl, 14-methyl-13-pentadecenyl, 14-methyl-14-pentadecenyl, 2-heptadecenyl, 16-heptadecenyl, 15-methyl-2- Hexadecenyl, 15-methyl-14-hexadecenyl, 15-methyl-15-hexadecenyl, 2-octadecenyl, 17-octadecenyl, 9,12-octadecadienyl, 9,12,15-octadecatri Henil, 9, 11, 13-octade Trienyl, 16-methyl-2-heptadecenyl, 16-methyl-15-heptadecenyl, 16-methyl-16-hepcenyldecenyl, 2-nonadecenyl, 18-nonadecenyl, 17-methyl-2-octadecenyl, 17-methyl- 16-octenyl decenyl, 17-methyl-17-octadecenyl, 2-eicosenyl, 19-eicosenyl, 5,8,11,14-icosatetraenyl, 18-methyl-2-nonadecenyl, 18-methyl-17- Nonadecenyl, 18-methyl-18-nonadecenyl, 3,7,11,15-tetramethyl-2,6,10,14-dodecatetenyl), preferably an alkenyl group having 2 to 10 carbon atoms. (E.g., Bier, aryl, isopropyl, 2-methyl-1-propenyl, 3-butenyl, 4-pentenyl, 3-methyl-2-butenyl, 3-methyl-3-butenyl, 5-to- Xenyl, 4-methyl-2-pentenyl, 4-methyl-3- Tenenyl, 4-methyl-4-pentenyl, 6-heptur, 5-methyl-2-hexenyl, 5-methyl-3-hexenyl, 5-methyl-4-hexenyl, 5-methyl-5-to Xenyl, 5- Methyl-2,4-hexagenyl, 7-octenyl, 6-methyl-2-heptenyl, 6-methyl-3-heptenyl, 6-methyl-4-heptenyl, 6-methyl-5-heptenyl, 6-methyl-6 -Heptenyl, 8-nonenyl, 9-decenyl, 3,7-dimethyl-2,6-octenyl, etc.).
置換されていてもよい炭化水素基としての 「アルキニル基」 の例としては、 炭素 数 2ないし 2 0のアルキニル基(例えばェチニル、 1 -プロピニル、 2 -プロピニル、 Examples of the “alkynyl group” as the optionally substituted hydrocarbon group include an alkynyl group having 2 to 20 carbon atoms (eg, ethynyl, 1-propynyl, 2-propynyl,
1 -ブチニル、 2 -ブチニル、 3 -ブチニル、 1 _メチル - 2 -プロピニル、 1 -ペンチ ニル、 2 -ペンチニル、 3 -ペンチニル、 4 -ペンチニル、 1 -メチル - 3 -ブチェル、.1-butynyl, 2-butynyl, 3-butynyl, 1_methyl-2-propynyl, 1-pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 1-methyl-3-butenyl ,.
2 -メチル - 3 -ブチニル、 1 -へキシニル、 2 _へキシニル、 3 -へキシニル、 4 -へ キシニル、 5 _へキシニル、 1 -へプチニル、 2 -へプチニル、 3 -へプチニル、 4 _ ヘプチェル、 5 -へプチニル、 6-ヘプチェル、 2 -ォクチ二ル、 3 -ォクチ二ル、 4 - ォクチ二ル、 5 -ォクチニル、 6-ォクチ二ル、 7-ォクチニル、 2-ノニニル、 8-ノニ ニル、 2-デシニル、 9-デシニル、 2 -ゥンデシニル、 10-ゥンデシニル、 2-ドデシ二 ル、 11-ドデシ二ル、 2-トリデシニル、 12-トリデシニル、 2-テトラデシニル、 I Sテトラデシニル、 2-ペン夕デシニル、 14-ペン夕デシニル、 2_へキサデシニル、 15 - へキサデシニル、 2-ヘプタデシニル、 16-ヘプ夕デシニル、 2-ォクタデシニル、 17 - ォク夕デシニル、 2-ノナデシニル、 18-ノナデシニル、 2-エイコシニル、 19-エイコ シニルなど) が挙げられ、好ましくは炭素数 2ないし 8のアルケニル基 (例えばェ チニル、 1 -プロピニル、 2 -プロピニル、 2-ブチニル、 3 -プチニル、 2-ペンチニル、 4-ペンチニル、 2-へキシニル、 5 -へキシニル、 2_へプチニル、 6-へプチニル、 2 - ォクチエル、 7-ォクチニルなど) が挙げられる。 2-Methyl-3-butynyl, 1-hexynyl, 2_hexynyl, 3-hexynyl, 4-hexynyl, 5_hexynyl, 1-heptynyl, 2-heptynyl, 3-heptynyl, 4_ Heptchel, 5-heptinyl, 6-heptyl, 2-octinyl, 3-octinyl, 4-octinyl, 5-octinyl, 6-octinyl, 7-octinyl, 2-noninyl, 8-noni Nyl, 2-decinyl, 9-decinyl, 2-p-decinyl, 10-p-decinyl, 2-dodecyl, 11-dodecinyl, 2-tridecinyl, 12-tridecinyl, 2-tetradecinyl, IS-tetradecinyl, 2-pentydecinyl , 14-pentadecynyl, 2_hexadecynyl, 15-hexadecynyl, 2-heptadecynyl, 16-hepdecanedecynyl, 2-octadedecynyl, 17-octanedecynyl, 2-nonadecynyl, 18-nonadecynyl, 2-eicosynyl, 19-Eico Shinil And alkenyl groups having 2 to 8 carbon atoms (e.g., ethynyl, 1-propynyl, 2-propynyl, 2-butynyl, 3-butynyl, 2-pentynyl, 4-pentynyl, 2-hexynyl, 5-hexynyl, 2_heptynyl, 6-heptynyl, 2-octiel, 7-octynyl, etc.).
置換されていてもよい炭化水素基としての 「 リール基」 の例としては、 炭素数 6ないし 1 0のァリール基(例えばフエニル、 0—トリル、 m—トリル、 p—トリル、 2 -、 Examples of the "reyl group" as the optionally substituted hydrocarbon group include an aryl group having 6 to 10 carbon atoms (eg, phenyl, 0-tolyl, m-tolyl, p-tolyl, 2-,
3 -または 4-ビフエ二ル、 卜または 2-ナフチル、 アントリル、 フエナントリルなど) が挙げられ、 好ましくはフエニル、 p-トリル、 1-、 3_または 4-ビフエニル、 1 -ま たは 2 -ナフチルなどが挙げられる。 3- or 4-biphenyl, tri- or 2-naphthyl, anthryl, phenanthryl, etc.), preferably phenyl, p-tolyl, 1-, 3_ or 4-biphenyl, 1- or 2-naphthyl And the like.
置換されていてもよい炭ィヒ水素基としての「ァラルキル基」の例としては炭素数 7ないし 1 4のァラルキル基(例えばベンジル、 1-フエネチル、 2-フエネチル、 1_ メチル -2-フエネチル、 卜メチル -1-フエネチル、 1, 1-ジメチル -2-フエネチル、 1- フエニルプロピル、 2-フエニルプロピル、 3-フエニルプロピル、 卜メチル -2-フエ ニルプロピル、 2_メチル -2-フエニルプロピル、 卜フエニルブチル、 2-フエニルブ チル、 3 -フエニルブチル、 4_フエニルブチル、 1-フエ二ルペンチル、 2-フエニルぺ ンチル、 3-フエ二ルペンチル、 4 -フエ二ルペンチル、 5 フエ二ルペンチル、 3 -メチ ル- 3-フエニルブチル、 1, 1 -ジメチル -3-フエニルプロピル、 2, 2-ジメチル- 3 -フ ェニルプロピル、 1,卜ジメチル- 4-フエニルブチル、 2, 2-ジメチル -4-フエ二ルブチ ル、 3, 3-ジメチル- 4 -フエニルブチル、 4-メチル -4 -フエ二ルペンチル、 1-メチル- 5- フエ二ルペンチル、 2-メチル -5-フエ二ルペンチル、 3-メチル -5-フエ二ルペンチル、 4-メチル -5-フエ二ルペンチル、 1 -ナフチルメチル、 2-ナフチルメチル、 1-ナフチ ルェチル、 2-ナフチルェチル、 2-ビフエニルメチル、 3-ビフエニルメチル、 4 -ビフ ェニルメチル、 1-アントリルメチル、 2-アントリルメチル、 9-アントリルメチル、 フエナントリルメチルなど) が挙げられ、 好ましくはベンジル、 2-フエネチル、 2 - フエニルプロピル、 3-フエニルプロピル、 2 -メチル -2-フエニルプロピル、 4-フエ ニルブチル、 1 -ナフチルメチル、 2 -ナフチルメチル、 1 -ナフチルェチル、 2-ナフチ ルェチル、 2-ビフエ二ルメチル、 3-ビフエニルメチル、 4 -ビフエニルメチルなどが 挙げられる。 Examples of the "aralkyl group" as the optionally substituted hydrocarbon group include an aralkyl group having 7 to 14 carbon atoms (eg, benzyl, 1-phenethyl, 2-phenethyl, 1-methyl-2-phenethyl, Methyl-1-phenethyl, 1,1-dimethyl-2-phenethyl, 1- Phenylpropyl, 2-phenylpropyl, 3-phenylpropyl, trimethyl-2-phenylpropyl, 2_methyl-2-phenylpropyl, triphenylbutyl, 2-phenylbutyl, 3-phenylbutyl, 4_phenylbutyl, 1-phenylpentyl, 2-phenylpentyl, 3-phenylpentyl, 4-phenylpentyl, 5 phenylpentyl, 3-methyl-3-phenylbutyl, 1,1-dimethyl-3-phenylpropyl, 2,2-Dimethyl-3-phenylpropyl, 1,2-dimethyl-4-phenylbutyl, 2,2-dimethyl-4-phenylbutyl, 3,3-dimethyl-4-phenylbutyl, 4-methyl-4- Phenylpentyl, 1-methyl-5-phenylpentyl, 2-methyl-5-phenylpentyl, 3-methyl-5-phenylpentyl, 4-methyl-5-phenylpentyl, 1-naphthylmethyl, 2 -Naphthylmethyl 1-naphthylethyl, 2-naphthylethyl, 2-biphenylmethyl, 3-biphenylmethyl, 4-biphenylmethyl, 1-anthrylmethyl, 2-anthrylmethyl, 9-anthrylmethyl, phenanthrylmethyl, etc.) Preferably benzyl, 2-phenyl, 2-phenylpropyl, 3-phenylpropyl, 2-methyl-2-phenylpropyl, 4-phenylbutyl, 1-naphthylmethyl, 2-naphthylmethyl, 1-naphthylethyl, 2 -Naphthylethyl, 2-biphenylmethyl, 3-biphenylmethyl, 4-biphenylmethyl and the like.
置換されていてもよい炭化水素基としての「シクロアルキル基」の例としては炭 素数 3ないし 8のシクロアルキル基(例えばシクロプロピル、 シクロブチル、 シク 口ペンチル、 シクロへキシル、 シクロへプチル、 シクロォクチルなど)が挙げられ る。 これらはさらにフエニル基などのァリール基が縮合していてもよく、例えばィ ンダニル、 ベンゾシクロへキシル、 ベンゾシクロへプチル、 ベンゾシクロォクチル などが挙げられる。 なかでも、 好ましくはシクロへキシル、 シクロへプチル、 シク 口才クチル、 インダニル、 ベンゾシクロへプチルなどが挙げられる。 Examples of the "cycloalkyl group" as the optionally substituted hydrocarbon group include cycloalkyl groups having 3 to 8 carbon atoms (eg, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, etc.). ). These may further be condensed with an aryl group such as a phenyl group, and examples thereof include indanyl, benzocyclohexyl, benzocycloheptyl, and benzocyclooctyl. Of these, preferred are cyclohexyl, cycloheptyl, cyclohexyl, indanyl, benzocycloheptyl and the like.
また、 このシクロアルキル基は、炭素数 1または 2の直鎖状の原子鎖を介して架 橋し、 ビシクロ [2. 2. 1] ヘプチル、 ビシクロ [2. 2. 2] ォクチル、 ビシ クロ [3. 2. 1] ォクチル、 ビシクロ [3. 3. 1] ノニル、 ビシクロ [3. 2. 23 ノニルなど (好ましくは、 炭素数 1または 2の直鎖状の原子鎖を介した架橋を 有するシクロへキシルなど、 さらに好ましくは、 ビシクロ [2. 2. 1] ヘプチル など) の架橋環式炭化水素残基を形成していてもよい。 また、 ァダマンチル基を形 成していてもよい。 This cycloalkyl group is bridged through a linear atom chain having 1 or 2 carbon atoms to form bicyclo [2.2.1] heptyl, bicyclo [2.2.2] octyl, bicyclo [ 3.2.1 octyl, bicyclo [3.3.1] nonyl, bicyclo [3.2.23 nonyl, etc. (preferably a cyclo group having a bridge through a linear atom chain having 1 or 2 carbon atoms) Hexyl, and more preferably bicyclo [2.2.1] heptyl) may form a crosslinked cyclic hydrocarbon residue. It also forms an adamantyl group. May be formed.
置換されていてもよい炭化水素基としての「シクロアルケニル基」の例としては 炭素数 3ないし 8のシクロアルケニル基 (例えば 2-シクロプロべニル、 2-シクロ ブテニル、 2-シクロペンテニル、 3 -シクロペンテニル、 2-シクロへキセニル、 3 - シクロへキセニル、 2-シクロへプテニル、 3 -シクロへプテニル、 4-シクロへプテニ ; k 2-シク口ォクテニル、 3-シクロォクテニル、 4-シクロォクテニルが挙げられる。 これらはさらにフエニル基などのァリ一ル基が縮合していてもよく、例えばインデ ニル、 ベンゾシクロへキセニル、 ベンゾシクロへプテニル、 ベンゾシクロォクテ二 ルなどが挙げられる。 なかでも好ましくは 2-シクロへキセニル、 2-シクロへプテ ニル、 シクロォクチル、 ィンデニルなどが挙げられる。 Examples of the "cycloalkenyl group" as the optionally substituted hydrocarbon group include a cycloalkenyl group having 3 to 8 carbon atoms (for example, 2-cyclopropenyl, 2-cyclobutenyl, 2-cyclopentenyl, 3-cyclopentenyl). Pentenyl, 2-cyclohexenyl, 3-cyclohexenyl, 2-cycloheptenyl, 3-cycloheptenyl, 4-cycloheptenyl; k 2-cyclooctenyl, 3-cyclooctenyl, 4-cyclooctenyl. These may further be condensed with an aryl group such as a phenyl group, for example, indenyl, benzocyclohexenyl, benzocycloheptenyl, benzocyclooctenyl and the like. Hexenyl, 2-cycloheptenyl, cyclooctyl, indenyl and the like can be mentioned.
R 2で示される 「置換されていてもよい炭化水素基」 の置換基としては、 例えば (1)ニトロ基、 (2)ォキソ基、 (3)ハロゲン原子 (例えば、 フッ素、 塩素、 臭素、 ョ ゥ素等) 、 (4)シァノ基、 (5)メチレン基、 (6)置換されていてもよい低級アルキル 基、 (7)置換されていてもよい低級アルケニル基、 (8)置換されていてもよい低級ァ ルキニル基、 (9)置換されていてもよいァリール基、 (10)置換されていてもよいァ ラルキル基、 (11)置換されていてもよいシクロアルキル基、 (12)低級ハロアルキル 基、 (13)エステル化もしくはアミド化されていてもよい力ルポキシル基、 (14)チォ 力ルバモイル基、 (15)ァシル基、 (16)ァミジル基、 (17)置換されていてもよい水酸 基、 (18)置換されていてもよいスルファニル基、 (19)置換されていてもよいアルキ ルスルフィニル基、 (20)置換されていてもよいァリールスルフィニル基、 (21)置換 されていてもよいアルキルスルホニル基、(22)置換されていてもよいァリ一ルスル ホニル基、 (23)エステル化もしくはアミド化されていてもよいスルホン酸基、 (24) 置換されていてもよいアミノ基、(25)ニトロ基で置換されていてもよいグァニジル 基、 (26)置換されていてもよい複素環基、 (27)アルキレンジォキシ基、 (28) イミ ノ基、 (29)置換されていてもよいシクロアルケニル基等が挙げられ、 これらの中か ら選択される 1ないし 6個の置換基を置換可能な位置に有してもよい。 Examples of the substituent of the “optionally substituted hydrocarbon group” for R 2 include (1) a nitro group, (2) an oxo group, and (3) a halogen atom (eg, fluorine, chlorine, bromine, (4) cyano group, (5) methylene group, (6) optionally substituted lower alkyl group, (7) optionally substituted lower alkenyl group, (8) substituted (9) optionally substituted aralkyl group, (10) optionally substituted aralkyl group, (11) optionally substituted cycloalkyl group, (12) lower haloalkyl Group, (13) olepoxyl group which may be esterified or amidified, (14) thiol bamoyl group, (15) acyl group, (16) amidyl group, (17) hydroxyl group which may be substituted (18) an optionally substituted sulfanyl group, (19) a substituted An optionally substituted alkylsulfinyl group, (20) an optionally substituted arylsulfinyl group, (21) an optionally substituted alkylsulfonyl group, (22) an optionally substituted arylsulfonyl group (23) a sulfonic acid group which may be esterified or amidated, (24) an amino group which may be substituted, (25) a guanidyl group which may be substituted by a nitro group, and (26) a substituted guanidyl group. An optionally substituted heterocyclic group, (27) an alkylenedioxy group, (28) an imino group, and (29) an optionally substituted cycloalkenyl group. It may have from 6 to 6 substituents at substitutable positions.
R 2で示される 「置換されていてもよい炭化水素」 の置換基とレての 「置換され ていてもよい低級アルキル基」 の 「低級アルキル基」 とは、 例えば、 メチル、 ェチ レ、 η_プロピル、 イソプロピル、 ブチル、 イソブチル、 sec-ブチル、 ter t-ブチル などの炭素数 1ないし 4のアルキル基が挙げられる。 The substituent of the "optionally substituted hydrocarbon" and the "lower alkyl group" of the "optionally substituted lower alkyl group" for R 2 are, for example, methyl, ethyl, η_propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl And C1 to C4 alkyl groups.
該 「低級アルキル基」 の有していてもよい置換基としては、 例えば、 (1)ニトロ 基、 (2)ォキソ基、 (3)ハロゲン原子 (例えば、 フッ素、 塩素、 臭素等) 、 (4)シァ ノ基、 (5)メチレン基、 (6)低級アルキル基(例えば、メチル、ェチル、 n-プロピル、 イソプロピル、 プチル、 イソプチル、 sec -ブチル、 ter t-ブチルなどの炭素数 1な いし 4のアルキル基) 、 (7)低級アルケニル基 (例えば、 ビエル、 ァリル (al lyl)、 イソプロぺニル、 1-ブテニル、 2-ブテニル、 3-メチル -2-ブテニル、 3-メチル -3 - ブテニル、 1-ペンテニル、 2-ペンテニルなどの炭素数 2ないし 5のアルケニル基)、 (8)低級アルキニル基 (ェチニル、 プロパルギル、 2 -ブチェル、 2-ペンチニルなど の炭素数 2ないし 5のアルキニル基) 、 (9)ァリール基 (例えば、 フエニル、 2_、 3 -または 4-トリル、 卜または 2-ナフチル、 2-、 3-または 4-ビフエ二ルなどの炭素 数 6ないし 1 2のァリール基) 、 (10)ァラルキル基 (例えば、 ベンジル、 2-フエネ チル、 3-フエニルプロピル、 1-または 2-ナフチルメチル、 1- 3-または 4-ピフエ ニルメチルなどの炭素数 7ないし 1 3のァラルキル基)、(11)シクロアルキル基(例 えば、 シクロプルピル、 シクロブチル、 シクロペンチル、 シクロへキシル、 シクロ ヘプチル、 シクロォクチルなどの炭素数 3ないし 8のシクロアルキル基) 、 Q2) エステル化もしくはアミド化されていてもよい力ルポキシル基(例えば、カルポキ シル基、炭素数 2から 1 8アルコキシカルボニル基(例えば、メトキシカルボニル、 エトキシカルポニル、 プロポキシカルボニル、 ter t-ブトキシカルボニル、 へキシ ルォキシカルポニル、 ドデシルォキシカルポニル、へキサデシルォキシカルポニル など)、炭素数 2から 1 8のアルケニルォキシ力ルポニル基 (例えば、ァリル(al lyl) ォキシカルボニル、 ォクタ _2, 6-ジェニルォキシカルボニル、 ドデ力- 2, 6, 10-トリ ェニルォキシカルポニルなど) 、 ァリールォキシ力ルポニル基(例えば、 フエノキ シカルボニル、 1一ナフトキシカルポニル、 2—ナフトキシカルポニルなど) 、 ァ ラルキルォキシ力ルポニル基(例えば、 ベンジルォキシカルボニル、 フエネチルォ キシカルポニル、 3-フエニルプロポキシカルポニルなど) 、 力ルバモイル基、 前記 の低級アルキル基、 低級アルケニル基、 シクロアルキル基、 ァリール基、 ァラルキ ル基および複素環基 (後述する R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されていてもよい複素環基」 同様のものなど)で N—モノ 置換された力ルバモイル基、 前記の低級アルキル基、 低級アルケニル基、 シクロア ルキル基、ァリール基、ァラルキル基および複素環基(後述する R 2で示される「置 換されていてもよい炭化水素」の置換基としての「置換されていてもよい複素環基」 と同様のものなど) で N, N—ジ置換された力ルバモイル基、 環状アミノカルバモ ィル基 (例えば、 N -ピロリジルカルポニル、 N-イミダゾリルカルポニル、 N -ピ ベリジルカルポニル、 N-ピペラジルカルポニル、 N -メチル -Ν'-ピペラジルカル ポニル、 Ν-モルホリルカルポニルなど) )、 (13)置換されていてもよい水酸基(例 えば、前記の低級アルキル基、低級アルケニル基、シクロアルキル基、ァリール基、 ァラルキル基、エステル化もしくはアミド化されていてもよい力ルポキシル基で置 換されていてもよい水酸基、 アルキルスルファニルチオ力ルポニルォキシ基(例え ば、 メチルスルファ二ルチオ力ルポ二ルォキシ、ェチルスルファ二ルチオカルボ二 ルォキシ、 tert-ブトキシスルファ二ルチオカルポニルォキシなど) 、 ァラルキル スルファニルチオ力ルポニルォキシ基 (例えば、ベンジルスルファ二ルチオ力ルポ ニルォキシなど) 、 N-ィミダゾリルチオカルポニルォキシ基、 N-モルホリルチオ力 ルポニルォキシ基など) 、 (14)置換されていてもよいスルファニル基(例えば、 ス ルファニル、 アルキルスルファニル基 (例えば、 メチルスルファニル、 ェチルスル ファニル、 イソプロピルスルファニル、 tert-ブチルスルファニルなど) 、 ァリー ルスルファニル基 (例えば、 フエニルスルファニルなど) 、 ァラルキルスルファニ ル基 (例えば、ベンジルスルファニルなど)など)、 (15)アルキルスルホニル基(例 えば、 メチルスルホニル、 ェチルスルホニル、 イソプロピルスルホニル、 プチルス ルホニルなど) 、 (16)ァリールスルホニル基 (例えば、 ベンゼンスルフィエル、 ト ルエンスルフィニルなど) 、 (17)置換されていてもよいアミノ基 (例えば、 ァミノ 基、 アルキル基またはァラルキル基でモノまたはジ置換されたァミノ基 (例えば、 メチルァミノ、 ェチルァミノ、 イソプロピルアミノ、 ジメチルァミノ、 ジェチルァ ミノ、 ェチルメチルァミノ、 ベンジルァミノ、 ベンジルメチルァミノなど) 、 ァシ ルァミノ (例えば、 ホルミルァミノ、 ァセチルァミノ、 プロピオニルァミノ、 ブチ リルァミノ、 イソプチリルァミノ、 バレリルァミノ、 イソバレリルァミノ、 ピバロ ィルァミノ、 ベンゾィルァミノ、 フエナシルァミノなど) 、 アルコキシカルボニル ァミノ (例えば、 メトキシカルポニルァミノ、 エトキシカルポニルァミノ、 tert - ブトキシカルポニルァミノなど) 、 ァリールォキシ力ルポニルァミノ (例えば、 フ エノキシカルポニルァミノ、 ナフチルォキシカルポニルァミノ等) 、 ァラルキルォ キシカルボニルァミノ (例えば、 ベンジルォキシカルポニルァミノ、 フエネチルォ キシカルポニルァミノなど) 、 アルキルスルホニルァミノ (例えば、 メチルスルホ ニルァミノ、 ェチルスルホニルァミノ、 ィソプロピルスルホニルァミノ、 プチルス ルホニルァミノ等)、 ァリールスルホニルァミノ (例えばベンゼンスルホニルアミ ノ、ナフチルスルホニルァミノなど)など)、 (18)アルキレンジォキシ基 (例えば、 メチレンジォキシ、 エチレンジォキシなど) 、 (19)ィミノ基などが挙げられ、 これ らの中から選択される 1ないし 4個の置換基を置換可能な位置に有してもよい。 Examples of the substituent which the “lower alkyl group” may have include, for example, (1) a nitro group, (2) an oxo group, (3) a halogen atom (eg, fluorine, chlorine, bromine, etc.), (4) ) A cyano group, (5) a methylene group, (6) a lower alkyl group (for example, having 1 to 4 carbon atoms such as methyl, ethyl, n-propyl, isopropyl, butyl, isoptyl, sec-butyl, tert-butyl, etc.) (7) a lower alkenyl group (for example, bier, allyl, isopropyl, 1-butenyl, 2-butenyl, 3-methyl-2-butenyl, 3-methyl-3-butenyl, An alkenyl group having 2 to 5 carbon atoms such as 1-pentenyl and 2-pentenyl); (8) a lower alkynyl group (an alkynyl group having 2 to 5 carbon atoms such as ethynyl, propargyl, 2-butenyl and 2-pentynyl), ( 9) Aryl group (for example, Phenyl, 2_, 3-or 4-tolyl, tri- or 2-naphthyl, 2- or 3- or 4-biphenyl, etc., aryl group having 6 to 12 carbon atoms), (10) aralkyl group (for example, benzyl) Aralkyl groups having 7 to 13 carbon atoms, such as, 2-phenyl, 3-phenylpropyl, 1- or 2-naphthylmethyl, 1-3- or 4-piphenylmethyl), (11) cycloalkyl groups (eg For example, a cycloalkyl group having 3 to 8 carbon atoms such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl), Q2) a propyloxyl group which may be esterified or amidated (eg, a carboxyl group) A C 2 -C 18 alkoxycarbonyl group (for example, methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, tert-butoxyca Bonyl, hexyloxycarbonyl, dodecyloxycarbonyl, hexadecyloxycarbonyl, etc.), alkenyloxycarbonyl groups having 2 to 18 carbon atoms (for example, allyloxycarbonyl, octa_2, 6-genyloxycarbonyl, dodeforce-2,6,10-trienyloxycarbonyl, etc., aryloxycarbonyl group (for example, phenoxycarbonyl, 1-naphthoxycarbonyl, 2-naphthoxycarbonyl, etc.), An aralkyloxycarbonyl group (for example, benzyloxycarbonyl, phenethyloxycarbonyl, 3-phenylpropoxycarbonyl, etc.), a carbamoyl group, a lower alkyl group, a lower alkenyl group, a cycloalkyl group, an aryl group, an aralkyl group And a heterocyclic group (“R 2 An “optionally substituted hydrocarbon” as a substituent of “an optionally substituted heterocyclic group”, and the like) A substituted carbamoyl group, a lower alkyl group, a lower alkenyl group, a cycloalkyl group, an aryl group, an aralkyl group, and a heterocyclic group (the “optionally substituted hydrocarbon” represented by R 2 described below); N, N-disubstituted carbamoyl groups, cyclic aminocarbamoyl groups (eg, N-pyrrolidylcarbonyl, N- Imidazolyl carbonyl, N-pibelidyl carbonyl, N-piperazyl carbonyl, N-methyl- ピ '-piperazyl carbonyl, Ν-morpholyl carbonyl, etc.)), (13) an optionally substituted hydroxyl group (for example, The lower alkyl group, the lower alkenyl group, the cycloalkyl group, the aryl group, the aralkyl group, the esterified or amidated olepoxyl A hydroxyl group which may be replaced by an alkylsulfanylthio carbonyl group (for example, methylsulfanylthio carbonyl group, ethylsulfanyl thiocarboxy group, tert-butoxysulfanyl thiocarbonyloxy group, etc.), aralkyl sulfanylthio group A sulfonyl group (eg, benzylsulfanylthio group, N-imidazolylthiocarboxy group, N-morpholylthio group, etc.), (14) an optionally substituted sulfanyl group (eg, , Sulfanyl, alkylsulfanyl group (eg, methylsulfanyl, ethylsulfanyl, isopropylsulfanyl, tert-butylsulfanyl, etc.), arylsulfanyl group (eg, phenylsulfanyl, etc.), aral (15) alkylsulfonyl group (eg, methylsulfonyl, ethylsulfonyl, isopropylsulfonyl, butylsulfonyl, etc.), (16) arylsulfonyl group (eg, benzenesulfonyl group) (17) an amino group which may be substituted (for example, an amino group mono- or di-substituted by an amino group, an alkyl group or an aralkyl group (for example, methylamino, ethylamino, isopropylamino, dimethylamino) , Acetylamino, ethylmethylamino, benzylamino, benzylmethylamino, etc.), acylamino (for example, formylamino, acetylamino, propionylamino, butyrylamino, isoptyrylamino, valerelli) Amino, isovaleryl Rua Mino, Pibaro Iruamino, Benzoiruamino, etc. Fuenashiruamino), alkoxycarbonyl Amino (e.g., methoxy Cal Poni Rua amino, ethoxy Cal Poni Rua amino, tert - Butoxycarponylamino, etc., aryloxylponylamino (for example, phenoxycarponylamino, naphthyloxycarponylamino, etc.), aralkyloxycarbonylamino (for example, benzyloxycarponylamino, phenethyloxycarponylamino, etc.) ), Alkylsulfonylamino (eg, methylsulfonylamino, ethylsulfonylamino, isopropylsulfonylamino, butylsulfonylamino, etc.), arylsulfonylamino (eg, benzenesulfonylamino, naphthylsulfonylamino, etc.) (18) alkylenedioxy group (for example, methylenedioxy, ethylenedioxy, etc.), (19) imino group, etc., and one or more substituents selected from these are located at substitutable positions. do it Good.
R 2で示される,「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよい低級アルケニル基」 の 「低級アルケニル基」 としては、 例えば、 ビニ ル、 ァリル (al lyl)、 イソプロべニル、 1 -ブテニル、 2 -ブテニル、 3-メチル -2-ブテ ニル、 3 -メチル -3-ブテニル、 1 ペンテニル、 2-ペンテニルなどの炭素数 2ないし 5のアルケニル基が挙げられる。 該「低級アルケニル基」 の有していてもよい置換 基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基と しての「置換されていてもよい低級アルキル基」の有していてもよい置換基と同様 な数、 同様のものが挙げられる。 Examples of the “lower alkenyl group” of the “optionally substituted lower alkenyl group” as the substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, vinyl and allyl. ), Isoprobenyl, 1-butenyl, 2-butenyl, 3-methyl-2-butenyl, 3-methyl-3-butenyl, 1 pentenyl, 2-pentenyl and the like. Can be Examples of the substituent which the “lower alkenyl group” may have include the “optionally substituted lower” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. The same number and the same as the substituents that the “alkyl group” may have.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよい低級アルキニル基」 の 「低級アルキニル基」 としては、 例えば、 ェチ ニル、 プロパルギル、 2 -ブチニル、 2-ペンチニルなどの低級アルキニル基などの炭 素数 2ないし 5のアルキニル基が挙げられる。該「低級アルキニル基」 の有してい てもよい置換基としては、前記の R 2で示される「置換されていてもよい炭化水素」 の置換基としての「置換されていてもよい低級アルキル基」の有していてもよい置 換基と同様な数、 同様のものが挙げられる。 Examples of the “lower alkynyl group” of the “optionally substituted lower alkynyl group” as the substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, ethynyl, propargyl, 2- Examples thereof include alkynyl groups having 2 to 5 carbon atoms, such as lower alkynyl groups such as butynyl and 2-pentynyl. Examples of the substituent which the “lower alkynyl group” may have include a “optionally substituted lower alkyl group” as a substituent of the aforementioned “optionally substituted hydrocarbon” represented by R 2. And the same number and the same as the substituents that may be possessed.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいァリーリレ基」 の 「ァリール基」 としては、 例えば、 フエ二リレ、 2_、 3 - または 4-トリル、 1-または 2-ナフチル、 2-、 3-または 4-ビフエ二ルなどの炭素数 6ないし 1 2のァリール基が挙げられる。該「ァリ一ル基」 の有していてもよい置 換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基 としての「置換されていてもよい低級アルキル基」の有していてもよい置換基と同 様な数、 同様のものが挙げられる。 Examples of the “aryl group” of the “optionally substituted arylyl group” as the substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, phenylyl, 2_, 3-or 4 And aryl groups having 6 to 12 carbon atoms such as -tolyl, 1- or 2-naphthyl, 2-, 3- or 4-biphenyl. Examples of the substituent which the “aryl group” may have include a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents which the “optionally substituted lower alkyl group” may have.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいァラルキル基」 の 「ァラルキル基」 としては、 例えば、 ベンジル、 2- フエネチル、 3-フエニルプロピル、 1 -または 2-ナフチルメチル、 2-、 3-または 4- ビフエニルメチルなどの炭素数 7ないし 1 3のァラルキル基が挙げられる。該「ァ ラルキル基」 の有していてもよい置換基としては、 前記の R 2で示される 「置換さ れていてもよい炭化水素」の置換基としての「置換されていてもよい低級アルキル 基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 Examples of the “aralkyl group” of the “optionally substituted aralkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, benzyl, 2-phenethyl, 3-phenyl Examples thereof include aralkyl groups having 7 to 13 carbon atoms such as propyl, 1- or 2-naphthylmethyl, 2-, 3- or 4-biphenylmethyl. Examples of the substituent which the “aralkyl group” may have include “optionally substituted lower alkyl” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2. And the same number and the same as the substituents which the group may have.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいシクロアルキル基」 の 「シクロアルキル基」 としては、 例えば、 シク 口プルピル、 シクロブチル、 シクロペンチル、 シクロへキシル、 シクロへプチル、 シクロォクチルなどの炭素数 3ないし 8のシクロアルキル基、 ビシクロ [2. 2. 1] ヘプチル、 ビシクロ [2. 2. 2〕 ォクチル、 ビシクロ [3. 2. 1] ォクチ ル、 ビシクロ [3. 3. 1] ノエル、 ピシクロ [3. 2. 2] ノエルおよびァダマ ンチルが挙げられる。該「シクロアルキル基」の有していてもよい置換基としては、 前記の R2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換 されていてもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様の ものが挙げられる。 Examples of the “cycloalkyl group” of the “optionally substituted cycloalkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, cyclopropyl, cyclobutyl, cyclopentyl, C3-C8 cycloalkyl groups such as cyclohexyl, cycloheptyl and cyclooctyl, bicyclo [2.2.1] heptyl, bicyclo [2.2.2] octyl, bicyclo [3.2.1] octyl , Bicyclo [3.3.1] Noel, picicyclo [3.2.2] Noel and adamantyl. Examples of the substituent which the “cycloalkyl group” may have include a “optionally substituted lower alkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents which may be possessed.
' R 2で示される 「置換されていてもよい炭化水素」 の置換基としての,「低級ハロ アルキル基」 としては、 例えば、 フルォロメチル、 ジフルォロメチル、 トリフルォ ロメチル、 2 -フルォロェチル、 2, 2-ジフルォロェチル、 2, 2, 2 -トリフルォロェチル、 1, 1, 2, 2 -テトラフルォロェチル、 1, 1, 2, 2, 2-ペン夕フルォロェチル、 3-フルォロプ 口ピル、 3, 3-ジフルォロプロピル、 3, 3, 3-トリフルォロプロピル、 2,2,3,3, 3 -ペン タフルォロプロピル、 4 -フルォロブチル、 4, 4-ジフルォロブチル、 4,4,4-トリフル ォロブチル、 3, 3, 4, 4, 4-ペン夕フルォ口ブチル、 5-フルォロペンチル、 5, 5 -ジフル ォロペンチル、 5, 5, 5-トリフルォロペンチル、 4, 4, 5, 5, 5-ペン夕フルォロペンチル、 クロロメチル、 ジクロロメチル、 プロモメチルなどの 1ないし 6個のハロゲン基で 置換された低級アルキル基等が挙げられる。 R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「エステル 化もしくはアミド化されていてもよい力ルポキシル基」としては、力ルポキシル基、 アルコキシカルポニル基、アルケニルォキシカルポニル基、 ァリールォキシ力ルポ ニル基、 ァラルキルォキシカルポニル基、 力ルバモイル基、 N—モノ置換カルバモ ィル基、 N, N—ジ置換力ルバモイル基が挙げられる。 As the “lower haloalkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 , for example, fluoromethyl, difluoromethyl, trifluoromethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-Trifluoroethyl, 1,1,2,2-Tetrafluoroethyl, 1,1,2,2,2-pentafluorofluorethyl, 3-Fluoro mouth pill, 3,3-Diflu Trifluoropropyl, 3,3,3-trifluoropropyl, 2,2,3,3,3-pentafluoropropyl, 4-fluorobutyl, 4,4-difluorobutyl, 4,4,4-trifluorobutyl, 3,3,4,4,4-pentyl butyl, 5-fluoropentyl, 5,5-difluoropentyl, 5,5,5-trifluoropentyl, 4,4,5,5,5-pentyl 1 such as fluoropentyl, chloromethyl, dichloromethyl, bromomethyl Lower alkyl groups substituted with stone 6 halogen group. As the “optionally esterified or amidated hapoxyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 , an alkoxyl group, an alkoxycarbonyl group, an alkenyloxycarbonyl group And aralkyloxycarbonyl groups, alkaryloxycarbonyl groups, rubamoyl groups, N-monosubstituted carbamoyl groups, and N, N-disubstituted rubamoyl groups.
「アルコキシカルポニル基」 としては、 例えば、 メトキシカルポニル、 エトキシ 力ルポ二)レ、 プロポキシ力ルポニル、 tert-ブトキシカルポニル、 へキシルォキシ カルボニル、 ドデシルォキシカルポニル、 へキサデシルォキシカルポニルなどの炭 素数 2から 1 8のアルコキシカルボニル基が挙げられる。該「アルコキシ力ルポ二 ル基」 は置換基を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての「置換されていてもよい低級 アルキル基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 Examples of the “alkoxycarbonyl group” include, for example, those having 2 to 2 carbon atoms such as methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, tert-butoxycarbonyl, hexoxycarbonyl, dodecyloxycarbonyl, hexadecyoxycarbonyl and the like. 18 alkoxycarbonyl groups. The “alkoxy group” may have a substituent, and the substituent may be a “substituted hydrocarbon” represented by R 2 described above. The same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
「アルケニルォキシカルポニル基」 としては、 例えばァリル(al lyl)ォキシカル ポニル、 ォクタ- 2, 6-ジェニルォキシカルポニル、 ドデ力- 2, 6, 10-卜リエ二ルォキ シカルポニルなどの炭素数 2から 1 8のアルケニルォキシカルボニル基が挙げら れる。 該「アルケニルォキシカルポニル基」 は置換基を有していてもよく、 その置 換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基 としての「置換されていてもよい低級アルキル基」の有していてもよい置換基と同 様な数、 同様のものが挙げられる。 Examples of the “alkenyloxycarbonyl group” include those having 2 carbon atoms such as allyloxycarbonyl, octa-2,6-genyloxycarbonyl, dodeforce-2,6,10-trienyloxycarbonyl. To 18 alkenyloxycarbonyl groups. The “alkenyloxycarbonyl group” may have a substituent, and the substituent may be a “substituted hydrocarbon” represented by R 2 described above as a substituent of “optionally substituted hydrocarbon”. And the same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
「ァリ一ルォキシカルボニル基」 としては、 例えばフエノキシ力ルポニル、 1― ナフトキシカルポニル、 2一ナフトキシカルポニル等が挙げられる。該「ァリール ォキシカルポニル基」 は置換基を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されて いてもよい低級アルキル基」の有していてもよい置換基と同様な数、同様のものが 挙げられる。 As the “aryloxycarbonyl group”, for example, phenoxycarbonyl, 1-naphthoxycarponyl, 21-naphthoxycarponyl and the like can be mentioned. The “aryloxycarbonyl group” may have a substituent, and the substituent may be a “substituted hydrocarbon” as a substituent of the “optionally substituted hydrocarbon” represented by R 2. And the same number and the same as the substituents which the lower alkyl group may have.
「ァラルキルォキシカルポニル基」としては、例えばべンジルォキシカルポニル、 フェネチルォキシカルボニル、 3 -フエニルプロポキシ力ルポニルなどのァラルキル ォキシカルポニル基が挙げられる。該「ァラルキルォキシカルポニル基」 は置換基 を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されてい てもよい炭化水素」 の置換基としての 「置換されていてもよい低級アルキル基」 の 有していてもよい置換基と同様な数、 同様のものが挙げられる。 Examples of the “aralkyloxycarbonyl group” include aralkyloxycarbonyl groups such as benzyloxycarbonyl, phenethyloxycarbonyl, and 3-phenylpropoxycarbonyl. The “aralkyloxycarbonyl group” may have a substituent, and the substituent is the “substituted or substituted” represented by R 2 described above. The same number and the same as the substituents which the “optionally substituted lower alkyl group” may have as a substituent of the “optionally substituted hydrocarbon”.
「N—モノ置換カルパモイル基」 の置換基としては、 例えば低級アルキル (例、 メチル、 ェチル、 プロピル、 イソプロピル、 ブチル、 イソブチル、 tert—ブチル、 ペンチル、 へキシル等) 、 低級アルケニル (例、 ビエル、 ァリル(al lyl)、 イソプ ロぺニル、 プロぺニル、 ブテニル、 ペンテニル、 へキセニル等) 、 シクロアルキル (例、 シクロプロピル、 シクロブチル、 シクロペンチル、 シクロへキシル等) 、 ァ リ一ル (例、 フエニル、 1 一ナフチル、 2—ナフチル) 、 ァラルキル (例、 ベンジ ル、 フエネチル等) 、 複素環基 (例えば後述する R 2で示される 「置換されていて もよい炭化水素」 の置換基としての「置換されていてもよい複素環基」 と同様のも の等) 等が挙げられる。 該低級アルキル、 低級アルケニル、 シクロアルキル、 ァリ —ル、 ァラルキル、 複素環基は置換基を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換 されていてもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様の ものが挙げられる。 Examples of the substituent of the “N-monosubstituted carpamoyl group” include lower alkyl (eg, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl, etc.), lower alkenyl (eg, Bier, Aryl, isopropenyl, propenyl, butenyl, pentenyl, hexenyl, etc., cycloalkyl (eg, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, etc.), aryl (eg, phenyl) , 1-naphthyl, 2-naphthyl), aralkyl (eg, benzyl, phenethyl, etc.), heterocyclic group (for example, “substituted as a substituent of“ optionally substituted hydrocarbon ”shown by R 2 described below) The same as the "heterocyclic group which may be substituted") and the like. Lower alkyl, lower alkenyl, cycloalkyl, § Li - Le, Ararukiru, heterocyclic group may have a substituent, examples of the substituent group, represented by R 2 "which may be substituted As the substituent for the "good hydrocarbon", the same number and the same substituents as the substituent which the "optionally substituted lower alkyl group" may have.
「N, N—ジ置換力ルバモイル基」 の置換基とは、 窒素原子上に 2個の置換基を 有する力ルバモイル基を意味し、 上記した「N—モノ置換力ルバモイル」 における 置換基と同様のものが挙げられる。また、 2個の置換基が窒素原子と一緒になつて 環状アミノを形成する場合もあり、この様な場合の環状アミノ力ルバモイルとして は、 例えば N-ピロリジルカルポニル、 N-イミダゾリルカルボニル、 N-ピベリジ ルカルポニル、 N-ピペラジルカルポニル、 N-メチル -Ν'-ピペラジルカルポニル、 The substituent of the “N, N-disubstituent rubamoyl group” means a disubstituted group having two substituents on a nitrogen atom, and is the same as the substituent in the above “N-monosubstituent rubamoyl”. One. In some cases, two substituents may be combined with a nitrogen atom to form a cyclic amino.In such a case, examples of the cyclic amino group rubamoyl include N-pyrrolidylcarbonyl, N-imidazolylcarbonyl, and N-imidazolylcarbonyl. Piveridyl rucarbonyl, N-piperazylcarbonyl, N-methyl-Ν'-piperazylcarbonyl,
Ν-モルホリルカルポニルなどが挙げられる。 Ν-morpholylcarbonyl and the like.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての「ァシル基」 としては、 水素原子または前記の ΓΝ—モノ置換力ルバモイル基」 が窒素原子上に 1個有する置換基とカルポニルとが結合したものが挙げられ、 例えばホルミル基、 ァセチル、 プロピオニルなどの低級アルカノィル基、 またはべンゾィル、 ナフ卜ィ ルなどのァロイル基等が挙げられる。 As the “acyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 , a hydrogen atom or a substituent having one of the above-mentioned monosubstituent rubamoyl groups on the nitrogen atom And carbonyl, such as a lower alkanoyl group such as formyl group, acetyl and propionyl, or an aroyl group such as benzoyl and naphthyl.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよい水酸基」 の置換基としては、 置換されていてもよい低級アルキル基、 置換されていてもよい低級アルケニル基、置換されていてもよい低級アルキニル基、 置換されていてもよいァリール基、置換されていてもよいァラルキル基、置換され ていてもよいシクロアルキル基、エステル化もしくはアミド化された力ルポキシル 基、置換されていてもよいアルキルスルファニルチオカルポニル基、置換されてい てもよぃァラルキルスルファニルチオカルポニル基、置換されていてもよいアルキ ルスルホニル基、置換されていてもよいァリ一ルスルホニル基、置換されていても よい N-イミダゾリルチオ力ルポニル、 置換されていてもよい N-モルホリルチオ力 ルポニル、水酸基の保護基等が挙げられる。該「置換されていてもよい低級アルキ ル基」 、 「置換されていてもよい低級アルケニル基」 、 「置換されていてもよい低 級アルキニル基」 、 「置換されていてもよいァリール基」 、 「置換されていてもよ ぃァラルキル基」 、 「置換されていてもよいシクロアルキル基」 としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されて いてもよい低級アルキル基」 、 「置換されていてもよい低級アルケニル基」 、 「置 換されていてもよい低級アルキニル基」 、 「置換されていてもよいァリール基」 、 「置換されていてもよいァラルキル基」、 「置換されていてもよいシクロアルキル 基」 と同様のものが挙げられる。 該「エステル化もしくはアミド化されたカルポキ シル基」 とは、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基 としてのエステル化もしくはアミド化されていてもよい力ルポキシル基」からカル ポキシル基を除いたものが挙げられる。該「置換されていてもよいアルキルスルフ ァニルチオカルポニル基」 としては、 例えばメチルスルファ二ルチオ力ルポニル、 ェチルスルファ二ルチオ力ルポニル、 ter t-プトキシスルファ二ルチオカルポニル 等があげられ、該「置換されていてもよいァラルキルスルファニルチオカルボニル 基」 としては、 ベンジルスルファ二ルチオカルボニルォキシ等が挙げられ、 該「置 換されていてもよいアルキルスルホニル基」 としては、 メチルスルホニル、 ェチル スルホニル、 イソプロピルスルホニル、 プチルスルホニル等が挙げられ、 該「置換 されていてもよいァリ一ルスルホニル基」 としては、 ベンゼンスルホニル、 p—ト ルエンスルホニル、 m—トルエンスルホニル、 o—トルエンスルホニル等が挙げら れる。 該「置換されていてもよいアルキルスルファニルチオカルポニル基」 、 「置 換されていてもよいァラルキルスルファニルチオカルポニル基」、. 「置換されてい てもよいアルキルスルホニル基」、 「置換されていてもよいァリールスルホニル基」 の有していてもよい置換基としては、 前記の R 2で示される 「置換されていてもよ い炭化水素」 の置換基としての 「置換されていてもよい低級アルキル基」 の有して いてもよい置換基と同様な数、 同様のものが挙げられる。 「置換されていてもよい N -イミダゾリルチオ力ルポ二ル」 、 「置換されていてもよい N-モルホリルチオ力 ルポニル」 の有していてもよい置換基としては、 前記の R 2で示される 「置換され ていてもよい炭化水素」の置換基としての「置換されていてもよい低級アルキル基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 該「水酸基の保 護基」 としては、 例えば 2—テトラヒドロピラニル、 トリメチルシリル、 トリェチ ルシリル、 ter t -プチルジフエニルシリル等が挙げられる。 As the substituent of the `` optionally substituted hydroxyl group '' as the substituent of the `` optionally substituted hydrocarbon '' represented by R 2 , an optionally substituted lower alkyl group, Lower alkenyl group which may be substituted, lower alkynyl group which may be substituted, aryl group which may be substituted, aralkyl group which may be substituted, cycloalkyl group which may be substituted, esterification Or an amidated propyloxyl group, an optionally substituted alkylsulfanylthiocarbonyl group, an optionally substituted aralkylsulfanylthiocarbonyl group, an optionally substituted alkylsulfonyl group, An arylsulfonyl group which may be substituted, an optionally substituted N-imidazolylthiol haponyl, an optionally substituted N-morpholylthio haponyl, a hydroxyl-protecting group, and the like. The “optionally substituted lower alkyl group”, the “optionally substituted lower alkenyl group”, the “optionally substituted lower alkynyl group”, the “optionally substituted aryl group”, As the “optionally substituted aralkyl group” and the “optionally substituted cycloalkyl group”, the “substituted hydrocarbon” represented by R 2 may be a “substituted hydrocarbon”. Optionally substituted lower alkyl group, optionally substituted lower alkenyl group, optionally substituted lower alkynyl group, optionally substituted aryl group, and substituted And an optionally substituted aralkyl group "and a" optionally substituted cycloalkyl group ". The “esterified or amidated carboxyl group” refers to the optionally esterified or amidated lipoxyl as a substituent of the aforementioned “optionally substituted hydrocarbon” represented by R 2. Group "from which the carboxyl group has been removed. Examples of the "optionally substituted alkylsulfanylthiocarponyl group" include methylsulfanylthiocarponyl, ethylsulfanylthiocarponyl, tert-butoxysulfanylthiocarponyl, and the like. Examples of the "good aralkylsulfanylthiocarbonyl group" include benzylsulfanylthiocarbonyloxy and the like, and the "optionally substituted alkylsulfonyl group" include methylsulfonyl, ethylsulfonyl, isopropylsulfonyl, Examples of the "optionally substituted arylsulfonyl group" include benzenesulfonyl, p-toluenesulfonyl, m-toluenesulfonyl, o-toluenesulfonyl and the like. Said "optionally substituted alkylsulfanylthiocarbonyl group", "optionally substituted aralkylsulfanylthiocarbonyl group", and "substituted The optionally substituted alkylsulfonyl group and the optionally substituted arylsulfonyl group include, for example, the optionally substituted hydrocarbon represented by R 2 above. And the same number and the same as the substituents which the “lower alkyl group which may be substituted” may have. Examples of the substituent which may be possessed by the `` optionally substituted N-imidazolylthiol compound '' and the `` optionally substituted N-morpholylthio compound '' are those represented by R 2 described above. As the substituent of the "optionally substituted hydrocarbon", the same number and the same substituents as the "optionally substituted lower alkyl group" may have. Examples of the “hydroxyl protecting group” include 2-tetrahydropyranyl, trimethylsilyl, triethylsilyl, tert-butyldiphenylsilyl and the like.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいスルファニル基」 としては、 例えば、 スルファニル基、 メチルスルフ ァニル、 工チルスルファニル、 イソプロピルスルファニル、 tert -プチルスルファ ニル等のアルキルスルファニル基、フエニルスルファニル等のァリ一ルスルファ二 ル基、ベンジルスルファニル等のァラルキルスルファニル基等が挙げられる。該「置 換されていてもよいスルファニル基」の有していてもよい置換基としては、前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されて いてもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様のものが 挙げられる。 Examples of the “optionally substituted sulfanyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, a sulfanyl group, methylsulfanyl, methysulfanyl, isopropylsulfanyl, tert- Examples thereof include an alkylsulfanyl group such as butylsulfanyl, an arylsulfanyl group such as phenylsulfanyl, and an aralkylsulfanyl group such as benzylsulfanyl. Examples of the substituent which the “optionally substituted sulfanyl group” may have include “substituted or substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2. The same number and the same as the substituents which the “lower alkyl group which may be optionally present” may have.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいアルキルスルフィニル基」 としては、 例えば、 メチルスルフィニル、 ェチルスルフィニル、 イソプロピルスルフィニル等が挙げられる。 該「置換されて いてもよいアルキルスルフィニル基」の有していてもよい置換基としては、前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されて いてもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様のものが 挙げられる。 Examples of the “optionally substituted alkylsulfinyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, methylsulfinyl, ethylsulfinyl, isopropylsulfinyl and the like. Examples of the substituent which the “optionally substituted alkylsulfinyl group” may have include “substituted or substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2. The same number and the same as the substituents which the “lower alkyl group which may be optionally present” may have.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいァリ一ルスルフィニル基」としては、例えば、ベンゼンスルフィニル、 トルエンスルフィニル等が挙げられる。該「置換されていてもよいァリールスルフ ィニル基」 の有していてもい置換基としては、 前記の; 2で示される 「置換されて いてもよい炭化水素」 の置換基としての 「置換されていてもよい低級アルキル基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 Examples of the “optionally substituted arylsulfinyl group” as a substituent of the “optionally substituted hydrocarbon” for R 2 include, for example, benzenesulfinyl, toluenesulfinyl and the like. The "arylsulf optionally substituted" As has also been have substituents Iniru group ", it said; have the" optionally substituted lower alkyl group "as the substituent of the" hydrocarbon which may be substituted "represented by 2 And the same number and the same as the substituents which may be included.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいアルキルスルホニル基」 としては、 例えば、 メチルスルホニル、 ェチ ルスルホニル、イソプロピルスルホニル、プチルスルホニル等が挙げられる。該「置 換されていてもよいアルキルスルホニル基」 の有していてもよい置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換 されていてもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様の ものが挙げられる。 Examples of the “optionally substituted alkylsulfonyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, methylsulfonyl, ethylsulfonyl, isopropylsulfonyl, butylsulfonyl and the like. Is mentioned. Examples of the substituent which the “optionally substituted alkylsulfonyl group” may have include “substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” for R 2. The same numbers and the same as the substituents which the lower alkyl group which may be optionally possessed "may have.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいァリールスルホニル基」 としては、 例えば、 ベンゼンスルホニル、 ト ルエンスルホニル、 ナフチルスルホニル等が挙げられる。該「置換されていてもよ いァリ一ルスルホニル基」 の有していてもよい置換基としては、 前記の R 2で示さ れる 「置換されていてもよい炭化水素」 の置換基としての「置換されていてもよい 低級アルキル基」の有していてもよい置換基と同様な数、同様のものが挙げられる。 Examples of the "optionally substituted arylsulfonyl group" as a substituent of the "optionally substituted hydrocarbon" represented by R 2 include, for example, benzenesulfonyl, toluenesulfonyl, naphthylsulfonyl and the like. . Examples of the substituent which the “optionally substituted arylsulfonyl group” may have include a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. The same number and the same thing as the substituent which the "lower alkyl group which may be substituted" may have are mentioned.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「エステル 化もしくアミド化されてもよいスルホン酸基」 としては、 例えば、 スルホン酸基、 スルホン酸メチル、 スルホン酸ェチル、 スルホン酸アミド、 N-メチルスルホン酸ァ ミド等が挙げられる。 Examples of the “sulfonic acid group which may be esterified or amidated” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, a sulfonic acid group, methyl sulfonic acid, and sulfonic acid Ethyl, sulfonic acid amide, N-methylsulfonic acid amide and the like.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいアミノ基」 としては、 アミノ基、 アルキル基またはァラルキル基でモ ノまたはジ置換されたァミノ基、 ァシルァミノ、 アルコキシ力ルポニルァミノ、 ァ リールォキシカルボニルァミノ、 ァラルキルォキシカルボニルアミノ、 アルキルス ルホニルァミノあるいはァリールスルホニルァミノ等が挙げられる。 As the “optionally substituted amino group” as a substituent of the “optionally substituted hydrocarbon” for R 2 , amino or di-substituted amino with an amino group, an alkyl group or an aralkyl group Groups, acylamino, alkoxyl propylonamino, aryloxycarbonylamino, aralkyloxycarbonylamino, alkylsulfonylamino, arylsulfonylamino and the like.
「アルキル基またはァラルキル基でモノまたはジ置換されたァミノ基 j とは、例 えばメチルァミノ、 ェチルァミノ、 イソプロピルァミノ、 ジメチルァミノ、 ジェチ ルァミノ、 ェチルメチルァミノ、 ベンジルァミノ、 ベンジルメチルァミノ等が挙げ "An amino group j mono- or di-substituted with an alkyl group or an aralkyl group j includes, for example, methylamino, ethylamino, isopropylamino, dimethylamino, methylamino, ethylmethylamino, benzylamino, benzylmethylamino and the like.
\ 、 られる。 該 「アルキル基またはァラルキル基でモノまたはジ置換されたァミノ基」 は置換基を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換 されていてもよい炭化水素」の置換基としての「置換されていてもよい低級アルキ ル基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 \, Can be. The "amino group mono- or di-substituted by an alkyl group or an aralkyl group" May have a substituent, and the substituent may be a `` optionally substituted lower alkyl group '' as a substituent of the `` optionally substituted hydrocarbon '' represented by R 2 above. And the same number and the same as the substituents that may be possessed.
「ァシルァミノ」 とは例えばホルミルァミノ、 ァセチルァミノ、 プロピオニルァ ミノ、 プチリルアミノ、 イソプチリルァミノ、 バレリルァミノ、 イソバレリルアミ ノ、 ピバロィルァミノ、 ベンゾィルァミノ、 フエナシルァミノ等が挙げられる。 該 「ァシルァミノ」 は置換基を有していてもよく、 その置換基としては、 前記の 2 で示される 「置換されていてもよい炭化水素」 の置換基としての「置換されていて もよい低級アルキル基」の有していてもよい置換基と同様な数、 同様のものが挙げ られる。 "Acylamino" includes, for example, formylamino, acetylamino, propionylamino, butyrylamino, isoptyrylamino, valerylamino, isovaleramino, pivaloylamino, benzoylamino, phenacylamino and the like. The “acylamino” may have a substituent, and the substituent may be a “optionally substituted lower” as a substituent of the “optionally substituted hydrocarbon” shown in the above 2 The same number and the same as the substituents that the “alkyl group” may have.
「アルコキシ力ルポニルァミノ」 とは例えばメトキシカルボニルァミノ、 ェトキ シカルボニルァミノ、 ter卜ブトキシカルポニルァミノ等が挙げられる。 該 「アル コキシ力ルポニ^/アミノ」 は置換基を有していてもよく、その置換基としては、 前 記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換さ れていてもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様のも のが挙げられる。 The “alkoxyl ponylamino” includes, for example, methoxycarbonylamino, ethoxycarbonylamino, tert-butoxycarponylamino and the like. The “alkoxy compound / amino” may have a substituent, and the substituent may be a substituent of the “optionally substituted hydrocarbon” represented by R 2 described above . The same number and the same as those of the substituent which the “optionally substituted lower alkyl group” may have are exemplified.
「ァリールォキシカルポニルアミノ」 とは例えばフエノキシカルポニルァミノ、 ナフチルォキシカルボニルァミノ等が挙げられる。該「ァリールォキシカルポニル ァミノ」 は置換基を有していてもよく、 その置換基としては、 前記の R 2で示され る 「置換されていてもよい炭化水素」 の置換基としての「置換されていてもよい低 級アルキル基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 The "aryloxycarbonylamino" includes, for example, phenoxycarbonylamino, naphthyloxycarbonylamino and the like. The “aryloxycarbonylamino” may have a substituent, and the substituent may be a “optionally substituted hydrocarbon” represented by R 2 described above. The same number and the same as the substituents which the "lower alkyl group which may be substituted" may have.
「ァラルキルォキシカルボニルァミノ」 とは例えばベンジルォキシカルポニルァ ミノ、 フエネチルォキシ力ルポニルァミノ等が挙げられる。該「ァラルキルォキシ カルボニルァミノ」 は置換基を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されてい てもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様のものが挙 げられる。 “Aralkyloxycarbonylamino” includes, for example, benzyloxycarponylamino, phenethyloxycarbonylamino and the like. The “aralkyloxycarbonylamino” may have a substituent, and the substituent may be a “substituted hydrocarbon” as a substituent of the “optionally substituted hydrocarbon” for R 2. The same numbers and the same as the substituents that the “lower alkyl group which may be substituted” may have.
「アルキルスルホニルァミノ」 とは例えばメチルスルホニルァミノ、 ェチルスル ホニルァミノ、イソプロピルスルホニルァミノ、 プチルスルホニルァミノ等が挙げ られる。該「アルキルスルホニルァミノ」 は置換基を有していてもよく、 その置換 基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基と しての「置換されていてもよい低級アルキル基」の有していてもよい置換基と同様 な数、 同様のものが挙げられる。 "Alkylsulfonylamino" includes, for example, methylsulfonylamino, ethylsulfonylamino, isopropylsulfonylamino, butylsulfonylamino and the like. Can be The “alkylsulfonylamino” may have a substituent, and the substituent may be a “substituted hydrocarbon” as a substituent of the aforementioned “optionally substituted hydrocarbon” represented by R 2. The same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
「ァリールスルホニルァミノ」 とは例えばべンゼンスルホニルァミノ、 ナフチル スルホニルァミノ等が挙げられる。 該「ァリ一ルスルホニルァミノ」 は置換基を有 していてもよく、 その置換基としては、 前記の R 2で示される 「置換されていても よい炭化水素」 の置換基としての 「置換されていてもよい低級アルキル基」 の有し ていてもよい置換基と同様な数、 同様のものが挙げられる。 The "arylsulfonylamino" includes, for example, benzenesulfonylamino, naphthylsulfonylamino and the like. The “arylsulfonylamino” may have a substituent, and the substituent may be a “optionally substituted hydrocarbon” represented by R 2 described above. The same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよい複素環基」 としては、 例えば、 脂肪族単環式含窒素複素環基、 芳香族 単環式含窒素複素環基、縮合含窒素複素環基、含酸素単環式あるいは縮合複素環基、 含硫黄単環式あるいは縮合複素環基等が挙げられる。 Examples of the “optionally substituted heterocyclic group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include, for example, an aliphatic monocyclic nitrogen-containing heterocyclic group and an aromatic monocyclic group. Examples include a cyclic nitrogen-containing heterocyclic group, a condensed nitrogen-containing heterocyclic group, an oxygen-containing monocyclic or condensed heterocyclic group, and a sulfur-containing monocyclic or condensed heterocyclic group.
「脂肪族単環式含窒素複素環基」 とは、 例えばアジリジニル、 ァゼチジニル、 ピ ロリジニル、 イミダゾリジニル、 ピラゾリジニル、 ォキサゾリジニル、 イソキサゾ リジニル、 チアゾリジニル、 イソチアゾリジニル、 ピペリジル、 ピペラジル、 ピラ ジニル、 モルホリル、 チオモルホリル、 ォキサジニル、 チアジニル、 ァゼピニル、 ジァゼピニル、 ォキサゼピニル、 チアゼピニル等が挙げられる。 該「脂肪族単環式 含窒素複素環基」 は置換基を有していてもよく、 その置換基としては、 前記の R 2 で示される 「置換されていてもよい炭化水素」 の置換基としての「置換されていて もよい低級アルキル基」の有していてもよい置換基と同様な数、 同様のものが挙げ られる。 "Aliphatic monocyclic nitrogen-containing heterocyclic group" refers to, for example, aziridinyl, azetidinyl, pyrrolidinyl, imidazolidinyl, pyrazolidinyl, oxazolidinyl, isoxazolidinyl, thiazolidinyl, isothiazolidinyl, piperidyl, piperazyl, pyrazolyl, morpholinyl, morpholinyl Oxazinyl, thiazinyl, azepinyl, diazepinyl, oxazepinyl, thiazepinyl and the like. The “aliphatic monocyclic nitrogen-containing heterocyclic group” may have a substituent, and the substituent is a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents which the "optionally substituted lower alkyl group" may have.
「芳香族単環式含窒素複素環基」 とは、 例えばピロリル、 イミダゾリル、 ピラゾ リル、 ォキサゾリル、 イソキサゾリル、 チアゾリル、 イソチアゾリル、 トリアゾリ ル、ォキサジァゾリル、チアジアゾリル、テトラゾリル、 ピリジル、 ピリダジニル、 ピリミジニル、 ピラジニル、 トリアジニル等が挙げられる。 該「芳香族単環式含窒 素複素環基」 は置換基を有していてもよく、 その置換基としては、 前記の R 2で示 される 「置換されていてもよい炭化水素」 の置換基としての 「置換されていてもよ い低級アルキル基」の有していても^い置換基と同様な数、 同様のものが挙げられ る。 "Aromatic monocyclic nitrogen-containing heterocyclic group" means, for example, pyrrolyl, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, triazolyl, oxadiazolyl, thiadiazolyl, tetrazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazinyl, pyrazinyl Is mentioned. The “aromatic monocyclic nitrogen-containing heterocyclic group” may have a substituent, and the substituent may be a “optionally substituted hydrocarbon” represented by R 2 described above. As the substituent, the same number and the same as those of the "optionally substituted lower alkyl group" may be mentioned. You.
「縮合含窒素複素環基」 とは、 例えばべンゾピロリジニル、 ベンゾイミダゾリジ ニル、 ベンゾピラゾリジニル、 ベンゾォキサゾリジニル、 ベンゾイソキサゾリジニ ル、 ベンゾチアゾリジニル、 ベンゾイソチアゾリジニル、 ベンゾピペリジル、 ベン ゾピペラジル、 ベンゾピラジニル、 ベンゾモルホリル、 ベンゾチオモルホリル、 ベ ンゾォキサジニル、 ベンゾチアジニル、 ベンゾァゼピニル、 ベンゾジァゼピニル、 ベンゾォキサゼピニル、 ベンゾチアゼピエル、 インドリル、 イソインドリル、 1H- インダゾリル、 ベンゾイミダゾリル、 ベンゾォキサゾリル、 ベンゾイソォキサゾリ ル、 ベンゾチアゾリル、 ベンゾイソチアゾリル、 m-ベンゾトリアゾリル、 ベンゾ ォキサジァゾリル、 ベンゾチアジアゾリル、 キノリル、 イソキノリル、 キナゾリ二 ル、 キノキサリニル、 ナフチリジニル、 プリニル、 プテリジニル、 カルバゾリル、 カルポリニル、 ァクリジニル、 フエノキサジニル、 フエノチアジニル、 フエナジ二 ル、 インドリジニル、 ピロ口ピリダジニル、 ピロ口ピリミジニル、 ピロ口ピラジ二 ル、 ピロ口ピリジル、 ィミダゾピリダジニル、 ィミダゾピリミジニル、 ィミダゾピ ラジニル、 ィミダゾピリジル、 チエノピリダジニル、 チエノピリミジニル、 チエノ ピラジニル、 チエノピリジル、 チアゾロピリダジニル、 チアゾロピリミジニル、 チ ァゾロピラジニル、 チアゾロピリジル、 ピリダゾピリダジニル、 ピ Vダゾピリミジ ニル、 ピリダゾピラジニル、 ピリダゾピリジル、 トリアゾロピリジル等が挙げられ る。該「縮合含窒素複素環基」は置換基を有していてもよく、その置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換 されていてもよい低級アルキル基」 の有していてもよい置換基と同様な数、 同様の ものが挙げられる。 "Fused nitrogen-containing heterocyclic group" is, for example, benzopyrrolidinyl, benzimidazolidinyl, benzopyrazolidinyl, benzoxazolidinyl, benzoisoxazolidinyl, benzothiazolidinyl, benzoisothiazolidinyl Benzopiperidyl, benzopiperidyl, benzopyrazinyl, benzomorpholinyl, benzothiomorpholyl, benzoxazinyl, benzothiazinyl, benzozepinyl, benzodiazepinyl, benzoxazepinyl, benzothiazepinel, indolyl, isoindolyl, 1H-indazolyl , Benzimidazolyl, benzoxazolyl, benzoisoxazolyl, benzothiazolyl, benzoisothiazolyl, m-benzotriazolyl, benzoxoxadiazolyl, benzothiadiazolyl, quinolyl, iso Noryl, quinazolinyl, quinoxalinyl, naphthyridinyl, prenyl, pteridinyl, carbazolyl, carporinyl, acridinyl, phenoxazinyl, phenothiazinyl, fenazinyl, indolizinyl, piro-pyridazinyl, piro-mouth pyrimidinyl, piro-mouth pyrazinyl, piro-mouth pyrazinyl Imidazopyridazinyl, imidazopyrimidinyl, imidazopyrazinyl, imidazopyridyl, thienopyridazinyl, thienopyrimidinyl, thienopyrazinyl, thienopyridyl, thiazolopyridazinyl, thiazolopyrimidinyl, thiazolopizyl, thiazolopizyl Examples include pyridazopyridazinyl, pVdazopyrimidinyl, pyridazopyrazinyl, pyridazopyridyl, triazolopyridyl and the like. The “condensed nitrogen-containing heterocyclic group” may have a substituent, and the substituent may be a “substituted hydrocarbon” represented by R 2 described above. The same number and the same as the substituents which the lower alkyl group which may be substituted "may have.
「含酸素単環式あるいは縮合複素環基」 とは、 例えばォキシラニル、 ォキセ夕二 ル、 ォキゾラニル、 ジォキソラニル、 フリル、 ピラニル、 テトラヒドロビラニル、 ジォキサニル、 ベンゾフラニル、 イソベンゾフラニル、 ベンゾピラエル、 イソベン ゾピラニル、 ベンゾジォキサニル、 7ーォキサビシクロ [ 2 . 2 . 1 ]ヘプチル)、 9ーォキサビシクロ [ 3 . 3 . 1 ] ノニル等が挙げられる。 該「含酸素単環式ある いは縮合複素環基」 は置換基を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されてい てもよい低級アルキル基」の有していてもよい置換基と同様な数、 同様のものが挙 げられる。 "Oxygen-containing monocyclic or fused heterocyclic group" means, for example, oxilanyl, oxenyl, oxolanyl, dioxolanyl, furyl, pyranyl, tetrahydroviranyl, dioxanyl, benzofuranyl, isobenzofuranyl, benzopyrael, isobenzopyranyl, benzol Dioxanyl, 7-oxabicyclo [2.2.1] heptyl), 9-oxabicyclo [3.3.1] nonyl, and the like. The “oxygen-containing monocyclic or condensed heterocyclic group” may have a substituent, and the substituent may be any of the above-mentioned “optionally substituted hydrocarbon” represented by R 2 . As a substituent, "substituted The same numbers and the same as the substituents that the “lower alkyl group which may be substituted” may have.
「含硫黄単環式あるいは縮合複素環基」とは、例えばチェニル、チォキソラニル、 テトラヒドロチォピラエル、 ジチア二ル、 ベンゾチェニル等が挙げられる。 該「含 硫黄単環式あるいは縮合複素環基」は置換基を有していてもよく、その置換基とし ては、 前記の R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換されていてもよい低級アルキル基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 The “sulfur-containing monocyclic or condensed heterocyclic group” includes, for example, phenyl, thioxolanyl, tetrahydrothiopyrael, dithianil, benzothenyl and the like. The “sulfur-containing monocyclic or condensed heterocyclic group” may have a substituent, and the substituent may be a substituent of the “optionally substituted hydrocarbon” represented by R 2 described above. As the group, the same number and the same as the substituent which the "lower alkyl group which may be substituted" may have.
で示される 「置換されていてもよい炭化水素」 の置換基としての 「アルキレ ンジォキシ基」 としては、 例えば、 メチレンジォキシ、 エチレンジォキシ等が挙げ られる。 As the "alkylenedioxy group" as a substituent of the "optionally substituted hydrocarbon" represented by, for example, methylenedioxy, ethylenedioxy and the like can be mentioned.
R 2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換され ていてもよいシクロアルケニル基」としては炭素数 3ないし 8のシクロアルケニル 基 (例えば 2 -シクロプロぺニル、 2-シクロブテニル、 2 -シクロペンテニル、 2 -シ クロへキセニル、 3 -シクロへキセニル、 2-シクロへプテニル、 3 -シクロへプテニル、 2 -シクロォクテニル、 4 -シクロォクテニルなど) が挙げられる。 該「シクロアルケ ニル基」 の有していてもよい置換基としては、 前記の R 2で示される 「置換されて いてもよい炭化水素」 の置換基としての 「置換されていてもよい低級アルキル基」 の有していてもよい置換基と同様な数、 同様のものが挙げられる。 Examples of the “optionally substituted cycloalkenyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 include a cycloalkenyl group having 3 to 8 carbon atoms (eg, 2-cyclopropenyl, 2-cyclobutenyl, 2-cyclopentenyl, 2-cyclohexenyl, 3-cyclohexenyl, 2-cycloheptenyl, 3-cycloheptenyl, 2-cyclooctenyl, 4-cyclooctenyl and the like. Examples of the substituent which the “cycloalkenyl group” may have include a “optionally substituted lower alkyl group” as a substituent of the “optionally substituted hydrocarbon” represented by R 2 above. And the same number and the same as the substituents that may be possessed.
R 2で表される 「置換されていてもよい複素環基」 としては、 前記の R 2で表さ れる 「置換されていてもよい炭化水素」 の置換基としての「置換されていてもよい 複素環基」 と同様のものが挙げられる。 ' As the "optionally substituted heterocyclic group" represented by R 2, "may be substituted as the substituent of the" hydrocarbon which may be substituted "represented by R 2 Heterocyclic group ". '
R 3、 R 4で示される 「置換されていてもよい炭化水素基」 としての炭化水素基 としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 としての炭 化水素と同様のものが挙げられる。 好ましくは、 炭素数 1ないし 6のアルキル基、 炭素数 6ないし 1 0のァリール基、炭素数 7ないし 1 0のァラルキル基等が挙げら れる。Examples of the hydrocarbon group as the “optionally substituted hydrocarbon group” represented by R 3 and R 4 include the hydrocarbon as the “optionally substituted hydrocarbon group” represented by the aforementioned R 2 And the same. Preferable examples include an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like.
3 R 4で示される 「置換されていてもよい炭化水素基」 の置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基と同様な数、 同様なものが挙げられる。 3 As the substituent of the `` optionally substituted hydrocarbon group '' represented by R 4 , the same number as the substituent of the `` optionally substituted hydrocarbon group '' represented by the aforementioned R 2 , Similar ones can be mentioned.
R 3、 R 4で示される 「置換されていてもよい複素環基」 は、 前記の R 2で表され る 「置換されていてもよい炭化水素」 の置換基としての 「置換されていてもよい複 素環基」 と同様のものが挙げられる。 The `` optionally substituted heterocyclic group '' represented by R 3 or R 4 may be `` optionally substituted '' as a substituent of the `` optionally substituted hydrocarbon '' represented by R 2 And the same as the “good complex ring group”.
「R 3、 R 4がともにそれらが結合している窒素原子と一緒になつて表す置換さ れていてもよい含窒素複素環」 としての含窒素複素環の例としては、炭素原子以外 に酸素原子、硫黄原子、窒素原子等から選ばれたヘテロ原子が 1ないし 4個含まれ ても良い 3ないし 8員環の単環性複素環またはこれらに環構造の縮合した 2環性 または 3環性縮合複素環基などが挙げられる。中でも好ましい例は 5ないし 7員環 の含窒素複素環が挙げられる。特に好ましい例としては、ァゼピエル、ピペリジル、 ピペラジル、 N-メチルビペラジル、 ピロリジル、モルホリル、などが挙げられる。 Examples of the nitrogen-containing heterocyclic ring as “an optionally substituted nitrogen-containing heterocyclic ring represented by R 3 and R 4 together with the nitrogen atom to which they are bonded” include, in addition to a carbon atom, May contain 1 to 4 heteroatoms selected from atoms, sulfur atoms, nitrogen atoms, etc. 3- to 8-membered monocyclic heterocycles or condensed bicyclic or tricyclic heterocycles And fused heterocyclic groups. Among them, a preferable example is a 5- to 7-membered nitrogen-containing heterocyclic ring. Particularly preferred examples include azepier, piperidyl, piperazyl, N-methylbiperazyl, pyrrolidyl, morpholyl, and the like.
「R 3、 R 4がともにそれらが結合している窒素原子と一緒になつて表す置換さ れていてもよい含窒素複素環」 の置換基としては、 前記の R 2で示される 「置換さ れていてもよい炭化水素基」 の置換基と同様な数、 同様なものが挙げられる。 The substituent of the "R 3, R 4 are both nitrogen-containing heterocyclic ring which may be substituted, such connexion represent together with the nitrogen atom to which they are attached", "is substituted represented by R 2 And the same number as the substituents of the "optionally substituted hydrocarbon group".
R 1としては、 — S R 2 (R 2は水素原子、 置換されていてもよい炭化水素基また は置換されていてもよい複素環基を示す) が好ましく、 なかでも R 2が置換されて いてもよい炭化水素基であるものが好ましい。 とりわけ— S R 2において、 R 2が 置換されていてもよいアルキル基(置換されていてもよい炭素数 1ないし 8のアル キル基)、置換されていてもよいアルケニル基(置換されていてもよい炭素数 2な いし 8のアルケニル基) 、 置換されていてもよいァラルキル基(置換されていても よい炭素数 7ないし 1 0のァラルキル基)、置換されていてもよいシクロアルキル 基(置換されていてもよい炭素数 3ないし 8のシクロアルキル基)であるものが好 ましい。 特に一 S R 2において、 R 2が置換されていてもよい炭素数 1ないし 8の アルキル基であるものがより好ましく、とりわけハロゲン原子で置換されていても よい炭素数 4ないし 8のアルキル基であるものが好ましい。 R 1 is preferably —SR 2 (R 2 represents a hydrogen atom, an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group), and in particular, R 2 is substituted. Those which are good hydrocarbon groups are preferred. In particular, in SR 2 , R 2 is an optionally substituted alkyl group (optionally substituted alkyl group having 1 to 8 carbon atoms) and an optionally substituted alkenyl group (optionally substituted An alkenyl group having 2 or 8 carbon atoms, an aralkyl group which may be substituted (an aralkyl group having 7 to 10 carbon atoms which may be substituted), a cycloalkyl group which may be substituted (substituted) And a cycloalkyl group having 3 to 8 carbon atoms which may be substituted. In particular, in SR 2 , R 2 is preferably an alkyl group having 1 to 8 carbon atoms which may be substituted, and more preferably an alkyl group having 4 to 8 carbon atoms which may be substituted by a halogen atom. Are preferred.
R 5で示される 「置換されていてもよい炭化水素基」 としての炭化水素基として は、 前記の R 2で示される 「置換されていてもよい炭化水素基」 としての炭化水素 基と同様のものが挙げられる。好ましくは、 炭素数 1ないし 1 2のアルキル基、 炭 素数 2ないし 1 2のアルケニル基、炭素数 2ないし 1 2のアルキニル基、炭素数 6 ないし 1 0のァリ一ル基、炭素数 7ないし 1 0のァラルキル基、炭素数 3ないし 8 のシクロアルキル基、炭素数 3ないし 8のシクロアルケニル基等が挙げられる。な かでも、 炭素数 1ないし 8のアルキル基、 炭素数 2ない 8のアルケニル基、 炭素数 6ないし 1 0のァリール基、炭素数 7ないし 1 0のァラルキル基、炭素数 3ないし 8のシクロアルキル基、炭素数 3ないし 8のシクロアルケニル基が好ましい。 とり わけ、 炭素数 1ないし 6のアルキル基が好ましく、 メチル、 ェチル、 プロピル、 ィ ソプロピル、 ブチル、 イソブチル、 sec-ブチル、 ter t-ブチル、 ペンチル、 イソべ ンチル、 2 -メチルブチル、 へキシル、 イソへキシル、 2 -ェチルブチル、 1, 1 -ジメ チルブチル、 2, 2-ジメチルブチル、 3, 3-ジメチルブチル、 2, 2-ジメチルペンチル、 3, 3-ジメチルペンチルが好ましい。 The hydrocarbon group as the `` optionally substituted hydrocarbon group '' represented by R 5 is the same as the hydrocarbon group as the `` optionally substituted hydrocarbon group '' represented by the aforementioned R 2 Things. Preferably, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, and 6 carbon atoms And an aryl group having 7 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, and a cycloalkenyl group having 3 to 8 carbon atoms. Among them, an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and a cycloalkyl having 3 to 8 carbon atoms The group is preferably a cycloalkenyl group having 3 to 8 carbon atoms. In particular, an alkyl group having 1 to 6 carbon atoms is preferable, and methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isobenzyl, 2-methylbutyl, hexyl, isobutyl Hexyl, 2-ethylbutyl, 1,1-dimethylbutyl, 2,2-dimethylbutyl, 3,3-dimethylbutyl, 2,2-dimethylpentyl, and 3,3-dimethylpentyl are preferred.
R 5で示される 「置換されていてもよい炭化水素基」 の置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基と同様の数、 同様の ものが挙げられる。 As the substituent of the `` optionally substituted hydrocarbon group '' represented by R 5 , the same number as the substituent of the `` optionally substituted hydrocarbon group '' represented by R 2 , and the same Things.
R 5で示される 「ァシル基」 とは、 前記の R 2で示される 「置換されていてもよ い炭化水素基」 の置換基としての 「ァシル基」 と同様のものが挙げられる。 The “acyl group” represented by R 5 is the same as the “acyl group” as a substituent of the “optionally substituted hydrocarbon group” represented by R 2 .
R 5で示される「エステル化もしくはアミド化されていてもよいカルボキシル基」 とは、 前記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基として の「エステル化もしくはアミド化されていてもよい力ルポキシル基」 と同様のもの が挙げられる。 なかでもメトキシカルポニル、 エトキシカルポニル、 プロポキシ力 ルポニル、 ter t-ブトキシカルポニル、 へキシルォキシ力ルポニル、 ドデシルォキ シカルボニル、へキサデシルォキシカルポニルなどの炭素数 2から 1 8のアルコキ シカルボニル基が好ましい。 The “carboxyl group which may be esterified or amidated” represented by R 5 means “esterification or amide as a substituent of the aforementioned“ optionally substituted hydrocarbon group ”represented by R 2. And the like. Among them, an alkoxycarbonyl group having 2 to 18 carbon atoms such as methoxycarbonyl, ethoxycarbonyl, propoxyl-proponyl, tert-butoxycarbonyl, hexyloxyl-proponyl, dodecyloxycarbonyl, hexadecyloxycarbonyl and the like is preferable.
R 5で示される 「置換されていてもよい複素環基」 とは、 前記の R 2で示される 「置換されていてもよい複素環基」 と同様のものが挙げられる。 The “optionally substituted heterocyclic group” represented by R 5 includes the same as the “optionally substituted heterocyclic group” represented by R 2 .
R 6で示される 「置換されていてもよい炭化水素基」 としての炭化水素基として は、 前記の R 2で示される 「置換されていてもよい炭化水素基」 としての炭化水素 基と同様のものが挙げられる。好ましくは、 炭素数 1ないし 8のアルキル基、 炭素 数 2ないし 8のアルケニル基、炭素数 6ないし 1 0のァリ一ル基、炭素数 7ないし 1 0のァラルキル基等が挙げられる。 なかでも、 炭素数 1ないし 6のアルキル基、 フエニル基、 ベンジル基等が好ましい。 ,The hydrocarbon group as the `` optionally substituted hydrocarbon group '' represented by R 6 is the same as the hydrocarbon group as the `` optionally substituted hydrocarbon group '' represented by the aforementioned R 2 Things. Preferable examples include an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Among them, alkyl groups having 1 to 6 carbon atoms, A phenyl group and a benzyl group are preferred. ,
R 6で示される 「置換されていてもよい炭化水素基」 の置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基と同様の数、 同様の ものが挙げられる。 As the substituent of the `` optionally substituted hydrocarbon group '' represented by R 6 , the same number as the substituent of the `` optionally substituted hydrocarbon group '' represented by the aforementioned R 2 , the same Things.
R 6で示される 「ァシル基」 とは、 前記の; R 2で示される 「置換されていてもよ い炭化水素基」 の置換基としての 「ァシル基」 と同様のものが挙げられる。 The “acyl group” represented by R 6 is the same as the “acyl group” as a substituent of the aforementioned “optionally substituted hydrocarbon group” represented by R 2 .
R 6で示される「エステル化もしくはアミド化されていてもよい力ルポキシル基」 とは、 前記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基として の「エステル化もしくはアミド化されていてもよい力ルポキシル基」 と同様のもの が挙げられる。 The `` optionally esterified or amidated olepoxyl group '' represented by R 6 means `` esterification or a hydrocarbon group which may be substituted '' represented by R 2 above. And the like.
R 6で示される 「置換されていてもよい複素環基」 とは、 前記の R 2で表される 「置換されていてもよい炭ィヒ水素」の置換基としての「置換されていてもよい複素 環基」 と同様のものが挙げられる。 The "optionally substituted heterocyclic group" represented by R 6, even if "substituted as the substituent of the" optionally substituted charcoal I nonhydrogen "represented by R 2 And the same as the “good heterocyclic group”.
R 5と R 6とが一緒になつて形成する環状炭化水素または複素環としては、 炭素 原子以外に窒素原子、酸素原子、硫黄原子等から選ばれた原子を 1ないし 3個含ん でもよい、 飽和または不飽和の、 5ないし 8員環の単環式炭素環または複素環、 あ るいはこれらを含む 2環式縮合炭素環または複素環などが挙げられる。例えば、 シ クロアルキル (例えば、シクロペンチル、シクロへキシル、シクロへプチルなど)、 シクロアルケニル (例えば、 シクロペンテニル、 シクロへキセニル、 シクロへキサ ジェニル、 シクロヘプテニルなど) 、 芳香族炭化水素環 (例えば、 フエニル、 ナフ チルなど) 、 含酸素環 (例えば、 フリル、 ォキソラニル、 テトラヒドロビラニル、 ォキセピニル、 ベンゾフラニル、 ベンゾピラニルなど) 、 含窒素環(例えば、 ピロ リジル、 ピペリジル、 ピリジル、 ァゼピニル、 インドリル、 キノリニル、 ベンゾァ ゼピニルなど) 、 含硫黄環 (例えばチェニル、 テトラヒドロチォピラニル、 ベンゾ チェニルなど) などが挙げられ、 中でもシクロへキシル、 シクロペンテニル、 シク 口へキセニル、 フエニルなどが特に好ましい。 R 5と R 6とが一緒になつて形成す る環状炭化水素または複素環は置換基を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基と同様の数、 同様のものが挙げられる。 R 5としては、 置換されていてもよい炭化水素基、 シァノ基、 ァシル基、 エステ ル化もしくはアミド化されていてもよい力ルポキシル基または置換されていても よい複素環基が好ましく、特に置換されていてもよい炭化水素基が好ましい。なか でも、 炭素数 1ないし 1 2のアルキル基、 炭素数 2ないし 1 2のアルケニル基、 炭 素数 2ないし 1 2のアルキニル基、炭素数 6ないし 1 0のァリ一ル基、炭素数 7な いし 1 0のァラルキル基、炭素数 3ないし 8のシクロアルキル基、炭素数 3ないし 8のシクロアルケニル基等が好ましい。とりわけ、炭素数 1ないし 8のアルキル基、 炭素数 2ない 8のァレケニル基、炭素数 6ないし 1 0のァリール基、炭素数 7ない し 1 0のァラルキル基、炭素数 3ないし 8のシクロアルキル基、炭素数 3ないし 8 のシクロアルケニル基が好ましい。特に、 炭素数 1ないし 6の炭化水素基、 とりわ け、 炭素数 1ないし 6のアルキル基が好ましく、 メチル、 ェチル、 プロピル、 イソ プロピル、 ブチル、 イソブチル、 sec-ブチル、 tert-ブチル、 ペンチル、 イソペン チル、 2 -メチルブチル、 へキシル、 イソへキシル、 2 -ェチルプチル、 1,卜ジメチ ルブチル、 2, 2-ジメチルブチル、 3, 3-ジメチルブチルが好ましい。 The cyclic hydrocarbon or heterocyclic ring formed by R 5 and R 6 together may contain, in addition to carbon atoms, one to three atoms selected from nitrogen, oxygen, sulfur and the like. Or an unsaturated 5- to 8-membered monocyclic carbocyclic or heterocyclic ring, or a bicyclic fused carbocyclic or heterocyclic ring containing these. For example, cycloalkyl (eg, cyclopentyl, cyclohexyl, cycloheptyl, etc.), cycloalkenyl (eg, cyclopentenyl, cyclohexenyl, cyclohexenyl, cycloheptenyl, etc.), aromatic hydrocarbon ring (eg, phenyl) , Naphthyl, etc.), oxygen-containing ring (for example, furyl, oxolanyl, tetrahydroviranyl, oxepinyl, benzofuranyl, benzopyranyl, etc.), nitrogen-containing ring (for example, pyrrolidyl, piperidyl, pyridyl, azepinyl, indolyl, quinolinyl, benzoxazepinyl, etc.) ), Sulfur-containing rings (eg, phenyl, tetrahydrothiopyranyl, benzothenyl, etc.), among which cyclohexyl, cyclopentenyl, cyclohexenyl, phenyl and the like are particularly preferred. Masui. Cyclic hydrocarbon or heterocyclic ring and R 5 and R 6 are you connexion formed such together may have a substituent, examples of the substituent group, be "substituted represented by R 2 The same number and the same as the substituents of the “good hydrocarbon group”. R 5 is preferably an optionally substituted hydrocarbon group, a cyano group, an acyl group, an optionally esterified or amidated propyloxyl group, or an optionally substituted heterocyclic group. An optionally substituted hydrocarbon group is preferred. Among them, alkyl groups having 1 to 12 carbon atoms, alkenyl groups having 2 to 12 carbon atoms, alkynyl groups having 2 to 12 carbon atoms, aryl groups having 6 to 10 carbon atoms, and 7 carbon atoms. Preferred are an aralkyl group having 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, and a cycloalkenyl group having 3 to 8 carbon atoms. Among them, alkyl groups having 1 to 8 carbon atoms, alkenyl groups having 2 to 8 carbon atoms, aryl groups having 6 to 10 carbon atoms, aralkyl groups having 7 to 10 carbon atoms, cycloalkyl groups having 3 to 8 carbon atoms And a cycloalkenyl group having 3 to 8 carbon atoms is preferred. Particularly, a hydrocarbon group having 1 to 6 carbon atoms, particularly an alkyl group having 1 to 6 carbon atoms is preferable, and methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, Preferred are isopentyl, 2-methylbutyl, hexyl, isohexyl, 2-ethylbutyl, 1,2-dimethylbutyl, 2,2-dimethylbutyl and 3,3-dimethylbutyl.
R 6としては、 置換されていてもよい炭化水素基が好ましい。 なかでも、 炭素数 1ないし 8のアルキル基、炭素数 2ないし 8のアルケニル基、炭素数 6ないし 1 0 のァリール基、 炭素数 7ないし 1 0のァラルキル基等が好ましい。特に、 炭素数 1 ないし 6の炭化水素基、 とりわけ、 炭素数 1ないし 6のアルキル基(メチル、 ェチ ル、 プロピル、 イソプロピル、 ブチル、 イソプチル、 sec-ブチル、 tert-ブチル、 ペンチル、 イソペンチル、 2 -メチルブチル、 へキシル、 イソへキシル、 2 -ェチル プチル、 1, 1-ジメチルブチル、 2, 2-ジメチルブチル、 3, 3-ジメテルブチル等、 特に メチル、 ェチル) 等が好ましい。 R 6 is preferably an optionally substituted hydrocarbon group. Of these, an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like are preferable. In particular, a hydrocarbon group having 1 to 6 carbon atoms, especially an alkyl group having 1 to 6 carbon atoms (methyl, ethyl, propyl, isopropyl, butyl, isoptyl, sec-butyl, tert-butyl, pentyl, isopentyl, 2 -Methylbutyl, hexyl, isohexyl, 2-ethylbutyl, 1,1-dimethylbutyl, 2,2-dimethylbutyl, 3,3-dimethylbutyl, etc., particularly methyl and ethyl) are preferred.
また、 R 5と R 6とが一緒になつて置換されていてもよいベンゼン環、 置換され ていてもよいシクロへキセン環を'形成する化合物も好ましい例として挙げられる。 Further, a compound forming a benzene ring which may be substituted together with R 5 and R 6 or a cyclohexene ring which may be substituted may also be mentioned as a preferable example.
R 7で示される 「置換されていてもよい水酸基」 の置換基としては、 例えば、 置 換されていてもよい炭化水素基、置換されていてもよいアルキルスルホニル基、置 換されていてもよいァリ一ルスルホニル基、置換されていてもよいアルキル力ルポ ニル基、 置換されていてもよいァリールカルボニル基等が挙げられる。 R 7で示さ れる 「置換されていてもよい水酸基」 のなかでも、 水酸基が好ましい。 R 7で示される 「置換されていてもよい水酸基」 の置換基としての置換されてい てもよい炭化水素基の 「炭化水素基」 としては、 炭素数 1ないし 8のアルキル基、 炭素数 2ないし 8のアルケニル基、炭素数 6ないし 1 0のァリール基、.炭素数 7な いし 1 0のァラルキル基等が挙げられる。なかでも、炭素数 1ないし 6のアルキル 基、 ベンジル基等が好ましい。 「置換されていてもよい水酸基」 の置換基としての 置換されていてもよい炭化水素基の置換基としては、 前記の R 2で示される 「置換 されていてもよい炭化水素基」 の置換基と同様の数、 同様のものが挙げられる。 Examples of the substituent of the `` optionally substituted hydroxyl group '' represented by R 7 include an optionally substituted hydrocarbon group, an optionally substituted alkylsulfonyl group, and an optionally substituted An arylsulfonyl group, an optionally substituted alkyl group, and an optionally substituted arylcarbonyl group. Among the “optionally substituted hydroxyl groups” represented by R 7 , a hydroxyl group is preferable. As the `` hydrocarbon group '' of the optionally substituted hydrocarbon group as a substituent of the `` optionally substituted hydroxyl group '' for R 7 , an alkyl group having 1 to 8 carbon atoms, an alkyl group having 2 to 2 carbon atoms An alkenyl group having 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like. Of these, an alkyl group having 1 to 6 carbon atoms, a benzyl group and the like are preferable. As a substituent of the optionally substituted hydrocarbon group as a substituent of the “optionally substituted hydroxyl group”, a substituent of the “optionally substituted hydrocarbon group” represented by R 2 above Similar numbers and similar ones are included.
R 7で示される 「置換されていてもよい水酸基」 の置換基としての置換されてい てもよいアルキルスルホニル基、置換されていてもよいァリ一ルスルホニル基の例 としては、例えば置換されていてもよい炭素数 1ないし 4のアルキルスルホニル基、 置換されていてもよいベンゼンスルホニル基等が挙げられ、なかでもメタンスルホ ニル基、 トリフルォロメ夕ンスルホニル基、 ベンゼンスルホニル基、 トルエンスル ホニル基等が好ましい。 Optionally substituted alkylsulfonyl group as the substituent of the "hydroxy group which may be substituted" represented by R 7, examples of the optionally substituted § Li one Rusuruhoniru group, for example substituted And an optionally substituted alkylsulfonyl group, an optionally substituted benzenesulfonyl group, and the like, among which methanesulfonyl group, trifluoromethanesulfonyl group, benzenesulfonyl group, and toluenesulfonyl group are preferable. .
R 7で示される 「置換されていてもよい水酸基」 の置換基としての置換されてい てもよいアルキルカルポニル基、置換されていてもよいァリ一ルカルポニル基とし ては、例えば置換されていてもよい炭素数 1ないし 4のアルキルカルポニル基、置 換されていてもよいベンゾィル基等が挙げられ、なかでもァセチル基、 プロピオ二 ル基、 ベンゾィル基等が好ましい。 Examples of the optionally substituted alkylcarbonyl group and the optionally substituted arylcarbonyl group as the substituent of the `` optionally substituted hydroxyl group '' represented by R 7 include, for example, Examples thereof include an alkylcarbonyl group having a good carbon number of 1 to 4, an optionally substituted benzoyl group, and the like. Among them, an acetyl group, a propionyl group, a benzoyl group and the like are preferable.
R 7で示される 「置換されていてもよい水酸基」 の置換基としての置換されてい てもよいアルキルスルホニル基、置換されていてもよいァリールスルホニル基、置 換されていてもよいアルキルカルボニル基および置換されていてもよいァリール カルポニル基の置換基としては、 前記の R 2で示される 「置換されていてもよい炭 化水素基」 の置換基と同様の数、 同様のものが挙げられる。 An optionally substituted alkylsulfonyl group, an optionally substituted arylsulfonyl group, an optionally substituted alkylcarbonyl group as a substituent of the "optionally substituted hydroxyl group" for R 7 Examples of the substituent of the arylcarbonyl group which may be substituted include the same numbers and the same substituents as those of the “optionally substituted hydrocarbon group” for R 2 .
R 8および R 9で示される 「置換されていてもよい炭化水素基」 としての炭化水 素基としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 として の炭化水素基と同様のものが挙げられる。好ましくは、炭素数 1ないし 8のアルキ ル基、 炭素数 2ないし 8のアルケニル基、 炭素数 6ないし 1 0のァリール基、 炭素 数 7ないし 1 0のァラルキル基等が挙げられる。なかでも、炭素数 1ないし 6のァ ' ルキル基、 フエニル基、 ベンジル基等が好ましい。 R 8および R 9で示される 「置換されていてもよい炭化水素基」 の置換基として は、 前記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基と同様の 数、 同様のものが挙げられる。 Examples of the hydrocarbon group as the “optionally substituted hydrocarbon group” represented by R 8 and R 9 include the hydrocarbon as the “optionally substituted hydrocarbon group” represented by the aforementioned R 2 Examples are the same as the groups. Preferable examples include an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Among them, an alkyl group having 1 to 6 carbon atoms, a phenyl group, a benzyl group and the like are preferable. The substituent of the “optionally substituted hydrocarbon group” represented by R 8 and R 9 is the same as the substituent of the “optionally substituted hydrocarbon group” represented by R 2 described above. , And the like.
R 5と R 8とが一緒になつて形成する環状炭化水素または複素環としては、 炭素 原子以外に窒素原子、酸素原子、硫黄原子等から選ばれた原子を 1ないし 3個含ん でもよい、 飽和または不飽和の、 5ないし 8員環の単環式炭素環または複素環、 あ るいはこれらを含む 2環式縮合炭素環または複素環などが挙げられる。例えば、 シ クロアルキル (例えば、シクロペンチル、シクロへキシル、シクロへプチルなど)、 シクロアルケニル (例えば、 シクロペンテニル、 シクロへキセニル、 シクロへキサ ジェニル、 シクロヘプテニルなど) 、 芳香族炭化水素環 (例えば、 フエニル、 ナフ チルなど) 、 含酸素環 (例えば、 フリル、 ォキソラエル、 テトラヒドロピラニル、 ォキセピニル、 ベンゾフラニル、 ベンゾピラニルなど) 、 含窒素環 (例えば、 ピロ リジル、 ピペリジル、 ピリジル、 ァゼピニル、 インドリル、 キノリニル、 ベンゾァ ゼピエルなど) 、 含硫黄環 (例えばチェニル、 テトラヒドロチォピラニル、 ベンゾ チェニルなど) などが挙げられ、 中でもシクロへキセニル、 フエニルなどが特に好 ましい。 R 5と R 8とが一緒になつて形成する環状炭化水素ま,たは複素環は置換基 を有していてもよく、 その置換基としては、 前記の R 2で示される 「置換されてい てもよい炭化水素基」 の置換基と同様の数、 同様のものが挙げられる。 The cyclic hydrocarbon or heterocyclic ring formed by R 5 and R 8 together may contain 1 to 3 atoms selected from nitrogen, oxygen, sulfur, etc., in addition to carbon. Or an unsaturated 5- to 8-membered monocyclic carbocyclic or heterocyclic ring, or a bicyclic fused carbocyclic or heterocyclic ring containing these. For example, cycloalkyl (eg, cyclopentyl, cyclohexyl, cycloheptyl, etc.), cycloalkenyl (eg, cyclopentenyl, cyclohexenyl, cyclohexenyl, cycloheptenyl, etc.), aromatic hydrocarbon ring (eg, phenyl) , Naphthyl, etc.), oxygen-containing ring (for example, frill, oxorael, tetrahydropyranyl, oxepinyl, benzofuranyl, benzopyranyl, etc.), nitrogen-containing ring (for example, pyrrolidyl, piperidyl, pyridyl, azepinyl, indolyl, quinolinyl, benzoxazepiel, etc.) ), Sulfur-containing rings (eg, phenyl, tetrahydrothiopyranyl, benzothenyl, etc.), among which cyclohexenyl, phenyl and the like are particularly preferred. The cyclic hydrocarbon or heterocyclic ring formed by R 5 and R 8 together may have a substituent, and the substituent may be `` substituted or substituted '' represented by R 2 above. And the same number and the same as the substituents of the "optionally substituted hydrocarbon group".
R 8と R 9とがー緖になって形成する環状炭化水素または複素環としては、 炭素 原子以外に窒素原子、酸素原子、硫黄原子等から選ばれた原子を 1ないし 3個含ん でもよい、飽和または不飽和の、 5ないし 8員環の単環式炭素環または複素環、 あ るいはこれらを含む 2環式縮合炭素環または複素環などが挙げられる。例えば、 シ クロアルキル (例えば、シクロペンチル、シクロへキシル、シクロへプチルなど)、 シクロアルケニル (例えば、 シクロペンテニル、 シクロへキセニル、 シクロへキサ ジェニル、 シクロヘプテニルなど) 、 芳香族炭化水素環 (例えば、 フエニル、 ナフ チルなど) 、 含酸素環 (例えば、 フリル、 ォキゾラニル、 テトラヒドロピラニル、 ォキセピエル、 ベンゾフラニル、 ベンゾピラニルなど) 、 含窒素環 (例えば、 ピロ リジル、 ピペリジル、 ピリジル、 ァゼピニル、 インドリル、 キノリニル、 ベンゾァ ゼピエルなど) 、 含硫黄環(例えばチェニル、 テトラヒドロチォビラニル、 ベンゾ チェニルなど) などが挙げられる。 R 8と R 9とが一緒になつて形成する環状炭化 水素または複素環は置換基を有していてもよく、 その置換基としては、 前記の R 2 で示される 「置換されていてもよい炭化水素基」 の置換基と同様の数、 同様のもの が挙げられる。 The cyclic hydrocarbon or heterocyclic ring formed by combining R 8 and R 9 may include one to three atoms selected from a nitrogen atom, an oxygen atom, a sulfur atom, and the like in addition to a carbon atom. Examples thereof include a saturated or unsaturated, 5- to 8-membered monocyclic carbocyclic or heterocyclic ring, or a bicyclic fused carbocyclic or heterocyclic ring containing these. For example, cycloalkyl (eg, cyclopentyl, cyclohexyl, cycloheptyl, etc.), cycloalkenyl (eg, cyclopentenyl, cyclohexenyl, cyclohexenyl, cycloheptenyl, etc.), aromatic hydrocarbon ring (eg, phenyl) , Naphthyl, etc.), oxygen-containing ring (for example, furyl, oxazolanyl, tetrahydropyranyl, oxepiel, benzofuranyl, benzopyranyl, etc.), nitrogen-containing ring (for example, pyrrolidyl, piperidyl, pyridyl, azepinyl, indolyl, quinolinyl, benzoxazepiel, etc.) ), Sulfur-containing rings (eg, phenyl, tetrahydrothioviranyl, benzo) Chenyl, etc.). The cyclic hydrocarbon or heterocyclic ring formed by R 8 and R 9 together may have a substituent, and the substituent may be `` optionally substituted '' represented by R 2 described above. And the same number and the same as the substituents of the "hydrocarbon group".
R 8および R 9としては、 置換されていてもよい炭化水素基が好ましい。 なかで も、 炭素数 1ないし 8のアルキル基、 炭素数 2ないし 8のアルケニル基、 炭素数 6 ないし 1 0のァ "U—ル基、炭素数 7ないし 1 0のァラルキル基等が好ましい。 とり わけ、 炭素数 1ないし 6のアルキル基、 フエニル基、 ベンジル基等が好ましい。 As R 8 and R 9 , an optionally substituted hydrocarbon group is preferable. Of these, an alkyl group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms, a phenyl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms are preferable. That is, an alkyl group having 1 to 6 carbon atoms, a phenyl group, a benzyl group and the like are preferable.
R I Gは、 水素原子、 一 Z R 1 5または— P (O) R 1 6 R 1 7を示す。 RIG represents a hydrogen atom, one ZR 15 or —P (O) R 16 R 17 .
一 Z R 1 5の R 1 5で表される 「置換されていてもよい炭化水素基」 の炭化水素基 としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 としての炭 化水素基と同様のものが挙げられる。好ましくは、炭素数 1ないし 1 2のアルキル 基、 炭素数 2ないし 1 2のアルケニル基、 炭素数 2ないし 1 2のアルキニル基、 炭 素数 6ないし 1 0のァリール基、炭素数 7ないし 1 0のァラルキル基、炭素数 3な いし 8のシクロアルキル基、炭素数 3ないし 8のシクロアルケニル基等が挙げられ る。 なかでも、 炭素数 6ないし 1 0のァリール基、 炭素数 7ないし 1 0のァラルキ ル基、 炭素数 3ないし 8のシクロアルキル基等が好ましい。 As the hydrocarbon group of the `` optionally substituted hydrocarbon group '' represented by R 15 of ZR 15 , the `` optionally substituted hydrocarbon group '' represented by the aforementioned R 2 Examples include the same as the hydrocarbon group. Preferably, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aryl group having 6 to 10 carbon atoms, and a 7 to 10 carbon atom Aralkyl groups, cycloalkyl groups having 3 or 8 carbon atoms, cycloalkenyl groups having 3 to 8 carbon atoms, and the like. Of these, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and a cycloalkyl group having 3 to 8 carbon atoms are preferable.
5で示される 「置換されていてもよい炭化水素基」 の置換基としては、 前記 の R 2で示される 「置換されていてもよい炭化水素基」 の置換基と同様の数、 同様 のものが挙げられる。なかでも、置換されていてもよい炭素数 1ないし 4のアルキ ル基、 置換されていてもよい炭素数 2ないし 4のアルケニル基、 フッ素、 塩素、 臭 素などのハロゲン原子、炭素数 1ないし 4のアルコキシ基、炭素数 1ないし 4のハ 口アルキル基、 力ルポキシル基、 カルポキサミド基などが挙げられ、特に好ましい 置換基の例としては、 メチル基、 ェチル基、 プロピル基、 イソプロピル基、 ビエル 基、 フッ素、 塩素、 臭素、 メトキシ基、 エトキシ基などが挙げられる。 The substituent of the “optionally substituted hydrocarbon group” represented by 5 is the same as or similar to the substituent of the “optionally substituted hydrocarbon group” represented by R 2 above. Is mentioned. Of these, an optionally substituted alkyl group having 1 to 4 carbon atoms, an optionally substituted alkenyl group having 2 to 4 carbon atoms, a halogen atom such as fluorine, chlorine, and bromine, and having 1 to 4 carbon atoms Examples of the substituent include a methyl group, an ethyl group, a propyl group, an isopropyl group, a Bier group, and the like. Examples include fluorine, chlorine, bromine, a methoxy group, and an ethoxy group.
— Z R 1 5の R 1 5で表される 「置換されていてもよい複素環基」 とは、 前記の R 2 で表される 「置換されていてもよい炭化水素」 の置換基としての 「置換されていて もよい複素環基」 と同様のものが挙げられる。なかでも置換されていてもよいチェ ニル基、 置換されていてもよいキノリル基、置換されていてもよいべンゾォキサジ ァゾリル基、置換されていてもよいピリジル基、置換されていてもよいべンゾチア ジァゾリル基、置換されていてもよいべンゾチェニル基、置換されていてもよいォ キサゾリジニル基、置換されていてもよいべンゾジォキサニル基、置換されていて もよぃジベンゾフラニル基、 モルホリル基が好ましく、特に置換されていてもよい チェニル基、置換されていてもよいべンゾチアジアゾリル基、置換されていてもよ いピリジル基が好ましい例として挙げられる。 - ZR 1 5 and R 1 5 in the "optionally substituted heterocyclic group" represented is as the substituent of the "optionally substituted hydrocarbon" represented by R 2 ' Heterocyclic group which may be substituted "and the like. Among them, a phenyl group which may be substituted, a quinolyl group which may be substituted, and a benzoxazidi which may be substituted Azolyl group, optionally substituted pyridyl group, optionally substituted benzothiadiazolyl group, optionally substituted benzozoenyl group, optionally substituted oxazolidinyl group, optionally substituted An benzodioxanyl group, a substituted or unsubstituted dibenzofuranyl group and a morpholyl group are preferred, and particularly a optionally substituted phenyl group, an optionally substituted benzothiadiazolyl group and an optionally substituted A preferred example is a pyridyl group.
Zは—S02_、 -SO-, -CON 18S02- (R18は (: 6アルキル) 、 -CONR19- (R19は — 6アルキル) または— CO—を示す。 R18および R1 9で表される C卜6アルキルとしては、 例えば、 メチル、 ェチル、 プロピル、 イソ プロピル、 プチル、 イソブチル、 sec-ブチル、 tert-ブチル、 ペンチル、 へキシル 等が挙げられる。 Z is -S0 2 _, -SO-, -CON 18 S0 2 - (R 18 is (:. 6 alkyl), -CONR 19 - (R 19 is - 6 alkyl) or - CO- shown R 18 and R the C Bok 6 alkyl represented by 1 9, for example, methyl, Echiru, propyl, iso-propyl, heptyl, isobutyl, sec- butyl, tert- butyl, pentyl, hexyl, and the like to.
R16および R17で表される 「置換されていてもよい炭化水素基」 としては、 前 記の R2で示され ¾ 「置換されていてもよい炭化水素基」 としての炭化水素基と同 様のものが挙げられる。好ましくは、 炭素数 1ないし 12のアルキル基、 炭素数 2 ないし 12のアルケニル基、炭素数 2ないし 12のアルキニル基、炭素数 6ないし 10のァリール基、炭素数 7ないし 10のァラルキル基、炭素数 3ないし 8のシク 口アルキル基、炭素数 3ないし 8のシクロアルケニル基等が挙げられる。なかでも、 炭素数 1ないし 6のアルキル基、炭素数 6ないし 10のァリール基、炭素数 7ない し 10のァラルキル基等が好ましい。 特に、 メチル、 ェチル、 フエニル、 ベンジル などが好ましい。 The “optionally substituted hydrocarbon group” represented by R 16 and R 17 is the same as the hydrocarbon group represented by the aforementioned R 2 ¾ “optionally substituted hydrocarbon group” And the like. Preferably, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, Examples thereof include a 3-8 cycloalkyl group and a C3-8 cycloalkenyl group. Of these, an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like are preferable. Particularly, methyl, ethyl, phenyl, benzyl and the like are preferable.
R16および R17で表される 「置換されていてもよい炭化水素基」 の置換基とし ては、 前記の R2で示される 「置換されていてもよい炭化水素基」 の置換基と同様 の数、 同様のものが挙げられる。 The substituent of the `` optionally substituted hydrocarbon group '' represented by R 16 and R 17 is the same as the substituent of the `` optionally substituted hydrocarbon group '' represented by R 2 And the like.
R 16および R 17で表される「置換されていてもよい水酸基」 としては、例えば、 置換されていてもよい炭化水素基が挙げられる。 Examples of the “optionally substituted hydroxyl group” represented by R 16 and R 17 include, for example, an optionally substituted hydrocarbon group.
R 16および R 17で表される 「置換されていてもよい水酸基」 の置換基としての 置換されていてもよい炭化水素基の 「炭化水素基」 としては、 炭素数 1ないし 8の アルキル基、炭素数 2ないし 8のアルケニル基、炭素数 6ないし 10のァリール基、 炭素数 7ないし 10のァラルキル基等が挙げられる。なかでも、炭素数 1ないし 6 のアルキル基、炭素数 6ないし 10のァリール基、炭素数 7ないし 10のァラルキ ル基等が好ましい。 特に、 メチル、 ェチル、 プロピル、 プチル、 フエニル、 ベンジ ル等が好ましい。 「置換されていてもよい水酸基」の置換基としての置換されてい てもよい炭化水素基の置換基としては、 前記の R2で示される 「置換されていても よい炭化水素基」 の置換基と同様の数、 同様のものが挙げられる。 As the `` hydrocarbon group '' of the optionally substituted hydrocarbon group as a substituent of the `` optionally substituted hydroxyl group '' represented by R 16 and R 17 , an alkyl group having 1 to 8 carbon atoms, Examples thereof include an alkenyl group having 2 to 8 carbon atoms, an aryl group having 6 to 10 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Among them, 1 to 6 carbon atoms And an alkyl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, and the like. Particularly, methyl, ethyl, propyl, butyl, phenyl, benzyl and the like are preferable. Examples of the substituent of the optionally substituted hydrocarbon group as the substituent of the “optionally substituted hydroxyl group” include the substituent of the “optionally substituted hydrocarbon group” represented by R 2 above. Similar numbers and similar ones are included.
R 16および R 17で表される 「置換されていてもよい水酸基」 としては、 炭素数 1ないし 6のアルキル基、炭素数 6ないし 10のァリール基等で置換された水酸基 が好ましい。 The “optionally substituted hydroxyl group” represented by R 16 and R 17 is preferably a hydroxyl group substituted by an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms, or the like.
R16および R17で表される 「置換されていてもよいアミノ基」 としては、 前 記の R2で示される 「置換されていてもよい炭化水素」 の置換基としての 「置換さ れていてもよいアミノ基」 .と同様のものが挙げられる。好ましくは、 メチル、 ェチ ル、 フエニル、 ベンジル等が挙げられる。 The `` optionally substituted amino group '' represented by R 16 and R 17 includes the `` substituted amino group '' as a substituent of the `` optionally substituted hydrocarbon '' represented by R 2 above. And the like. " Preferably, methyl, ethyl, phenyl, benzyl and the like are mentioned.
R 16および R 17としては、 炭素数 1ないし 6のアルキル基、 炭素数 6ないし 1 0のァリール基、炭素数 1ないし 6のアルキル基で置換された水酸基、炭素数 6な いし 10のァリール基で置換された水酸基等が好ましく、 特にメチル、ェチル、 フ ェニル、 ベンジル、 メチルで置換された水酸基、 ェチルで置換された水酸基、 フエ ニルで置換された水酸基等が好ましい。 R 16 and R 17 are an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms, a hydroxyl group substituted with an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 10 carbon atoms. And a hydroxyl group substituted with methyl, ethyl, phenyl, benzyl, methyl, a hydroxyl group substituted with ethyl, and a hydroxyl group substituted with phenyl.
R1Qとしては一 ZR15 (Zおよび R15は前記と同意義を示す) が好ましい。 な かでも、 Zがー S〇2—または一 CO—であり、 R15が置換されていてもよいァリ —ル基、置換されていてもよいァラルキル基、置換されていてもよい複素環基であ るものが好ましい。 とりわけ、 Zがー SO 2—または一 CO—であり、 R 15が置換 されていてもよい炭素数 6ないし 10のァリール基、置換されていてもよい炭素数 7ないし 10のァラルキル基、置換されていてもよい複素環基であるものが好まし い。 特に、 Zがー S〇2—であり、 R 15がメチル基、 ェチル基、 プロピル基、 イソ プロピル基、 ビニル基、 フッ素、 塩素、 臭素、 メトキシ基、 エトキシ基などで置換 されていてもよいフエニル基、ベンジル基、チェニル基、ベンゾチアジアゾリル基、 または Zが—CO—であり、 R15がメチル基、 ェチル基、 プロピル基、 イソプロ ピル基、 ビニル基、 フッ素、 塩素、 臭素、 メトキシ基、 エトキシ基などで置換され ていてもよいピリジル基等が好ましい。 R11は、 水素原子または置換されていてもよい炭化水素基を示す。 。 R11で表 される 「置換されていてもよい炭化水素基」 の炭化水素基としては、 前記の R2で 示される「置換されていてもよい炭化水素基」 としての炭化水素基と同様のものが 挙げられる。好ましくは、 炭素数 1ないし 6のアルキル基、 炭素数 2ないし 6のァ ルケニル基、 炭素数 7ないし 10のァラルキル基等が挙げられる。 なかでも、 炭素 数 1ないし 6のアルキル基、ベンジル基等が好ましく、特に炭素数 1ないし 4のァ ルキル基等が好ましい。 R 11で示される 「置換されていてもよい炭化水素基」 の 置換基としては、 前記の R2で示される 「置換されていてもよい炭化水素基」 の置 換基と同様の数、 同様のものが挙げられる。 As R 1Q , one ZR 15 (Z and R 15 are as defined above) is preferable. Among them, Z is —S〇 2 — or 1 CO—, and R 15 is an optionally substituted aryl group, an optionally substituted aralkyl group, and an optionally substituted heterocyclic ring. What is a group is preferable. In particular, Z is —SO 2 — or 1 CO—, and R 15 is an optionally substituted aralkyl group having 6 to 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms which may be substituted, Those which are heterocyclic groups which may be substituted are preferred. In particular, Z is -S〇 2- , and R 15 may be substituted with methyl, ethyl, propyl, isopropyl, vinyl, fluorine, chlorine, bromine, methoxy, ethoxy, etc. Phenyl, benzyl, phenyl, benzothiadiazolyl, or Z is —CO—, and R 15 is methyl, ethyl, propyl, isopropyl, vinyl, fluorine, chlorine, bromine, methoxy And a pyridyl group which may be substituted with an ethoxy group or the like. R 11 represents a hydrogen atom or a hydrocarbon group which may be substituted. . The hydrocarbon group of the `` optionally substituted hydrocarbon group '' represented by R 11 is the same as the hydrocarbon group of the `` optionally substituted hydrocarbon group '' represented by the aforementioned R 2 Things. Preferable examples include an alkyl group having 1 to 6 carbon atoms, an alkenyl group having 2 to 6 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms. Of these, an alkyl group having 1 to 6 carbon atoms, a benzyl group and the like are preferable, and an alkyl group having 1 to 4 carbon atoms is particularly preferable. The substituent of the `` optionally substituted hydrocarbon group '' represented by R 11 is the same as or similar to the substituent of the `` optionally substituted hydrocarbon group '' represented by R 2 One.
R 11としては、 水素原子が好ましい。 R 11 is preferably a hydrogen atom.
R1Qと R11がー緖になって形成してもよい含窒素複素環とは、 すなわち、 式 1The nitrogen-containing heterocyclic ring which may be formed by combining R 1Q and R 11 is
で表される部分構造が The partial structure represented by
〔式中 B環は置換されていてもよい 5ないし 8員環を示す〕 (In the formula, ring B represents an optionally substituted 5- to 8-membered ring)
で表される 5ないし 8員の含窒素複素環であるものを意味する。 より具体的には、 式 Means a 5- to 8-membered nitrogen-containing heterocycle represented by More specifically, the expression
〔式中、 R17は前記と同意義を、 B1環ないし B12環はさらに置換基を有していて もよい含窒素複素環を示す〕 で表される環を意味する。 なお、 B1環ないし B12環 が有していてもよい置換基とは、 前記の R 15で示される 「置換されていてもよい 炭化水素基」 が有していてもよい置換基と同様のものを意味する。 [Wherein, R 17 has the same meaning as described above, and ring B 1 to ring B 12 each represent a nitrogen-containing heterocyclic ring which may further have a substituent]. In addition, the substituent which the ring B 1 to ring B 12 may have is the same as the substituent which the `` optionally substituted hydrocarbon group '' represented by R 15 may have Means things.
R 1 Qと R 11が一緒になつて形成してもよい含窒素複素環としては、 式 Examples of the nitrogen-containing heterocyclic ring which may be formed by R 1 Q and R 11 together include:
〔式中、 B 2環および B 3環はさらに置換基を有していてもよい含窒素複素環を示 す〕 [In the formula, ring B 2 and ring B 3 represent a nitrogen-containing heterocyclic ring which may further have a substituent.]
で表される 6または 7員の含窒素複素環が好ましい。 A 6- or 7-membered nitrogen-containing heterocyclic ring represented by
R12および R13で表される 「置換されていてもよい炭化水素基」 の炭化水素基 としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 としての炭 化水素基と同様のものが挙げられる。好ましくは、炭素数 1ないし 1 2のアルキル 基、 炭素数 2ないし 1 2のアルケニル基、 炭素数 2ないし 1 2のアルキニル基、 炭 素数 6ないし 1 0のァリール基、炭素数 7ないし 1 0のァラルキル基、炭素数 3な いし 8のシクロアルキル基、炭素数 3ないし 8のシクロアルケ二レ基等が挙げられ る。 なかでも、 炭素数 1ないし 1 2のアルキル基、 炭素数 2ないし 1 2のァルケ二 ル基、 炭素数 7ないし 1 0のァラルキル基が好ましい。 R 1 2および R 1 3で示され る 「置換されていてもよい炭化水素基」 の置換基としては、 前記の R 2で示される 「置換されていてもよい炭ィヒ水素基」の置換基と同様の数、同様のものが挙げられ る。 The hydrocarbon group of the “optionally substituted hydrocarbon group” represented by R 12 and R 13 Examples thereof include the same hydrocarbon groups as the aforementioned “optionally substituted hydrocarbon group” for R 2 . Preferably, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aryl group having 6 to 10 carbon atoms, and a 7 to 10 carbon atom An aralkyl group, a cycloalkyl group having 3 or 8 carbon atoms, a cycloalkenyl group having 3 to 8 carbon atoms, and the like. Of these, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms are preferable. Examples of the substituent of the “optionally substituted hydrocarbon group” represented by R 12 and R 13 include the substitution of the “optionally substituted hydrocarbon group” represented by the aforementioned R 2 The same number and the same as the group can be mentioned.
R 1 2および R 1 3がともにそれらが結合している窒素原子と一緒になつて示す置 換されていてもよい含窒素複素環の例としては前記の 「R 3、 R 4がともにそれら が結合している窒素原子と一緒になつて表す置換されていてもよい含窒素複素環」 の含窒素複素環と同様のものが挙げられる。特に好ましい例としては、ピペリジル、 ピペラジル、 N-メチルピペラジル、 ピロリジル、 モルホリルなどが挙げられる。 Examples of the nitrogen-containing heterocyclic ring which may be replaced together with R 12 and R 13 together with the nitrogen atom to which they are bonded include the aforementioned `` R 3 and R 4 are both those wherein Nitrogen-containing heterocycles which may be substituted together with the nitrogen atom to be bonded ”. Particularly preferred examples include piperidyl, piperazyl, N-methylpiperazyl, pyrrolidyl, morpholyl and the like.
「 R 1 2、 R 1 3がともにそれらが結合している窒素原子とー緖になつて表す置換 されていてもよい含窒素複素環」 の置換基としては、 前記の R 2で示される 「置換 されていてもよい炭化水素基」 の置換基と同様な数、 同様なものが挙げられる。 Examples of the substituent of the “nitrogen-containing heterocyclic ring which may be substituted by both R 12 and R 13 forming a nitrogen atom to which they are bonded to —” are those represented by the aforementioned R 2 The same number and the same as the substituents of the “optionally substituted hydrocarbon group”.
R 1 2としては、 水素原子、 炭素数 1ないし 1 2のアルキル基、 炭素数 2ないし 1 2のアルケニル基、炭素数 2ないし 1 2のアルキニル基、炭素数 6ないし 1 0の ァリ一ル基、炭素数 7ないし 1 0のァラルキル基、炭素数 3ないし 8のシクロアル キル基、 炭素数 3ないし 8のシクロアルケニル基等が好ましい。 とりわけ、 炭素数 1ないし 1 2のアルキル基、炭素数 2ないし 1 2のアルケニル基、,炭素数 7ないし 1 0のァラルキル基が好ましい。 R 12 is a hydrogen atom, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aryl group having 6 to 10 carbon atoms. Preferred are a group, an aralkyl group having 7 to 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, a cycloalkenyl group having 3 to 8 carbon atoms, and the like. Among them, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, and an aralkyl group having 7 to 10 carbon atoms are preferable.
R 1 3としては、 水素原子が好ましい。 R 13 is preferably a hydrogen atom.
R 1 4で表される 「置換されていてもよい炭化水素基」 の炭化水素基としては、 前記の R 2で示される 「置換されていてもよい炭化水素基」 としての炭^ ίヒ水素基と 同様のものが挙げられる。好ましくは、 炭素数 1ないし 1 2のアルキル基、 炭素数 2ないし 1 2のアルケニル基、炭素数 2ないし 1 2のアルキニル基、炭素数 6ない し 10のァリール基、炭素数 7ないし 10のァラルキル基、炭素数 3ないし 8のシ クロアルキル基、炭素数 3ないし 8のシクロアルケニル基等が挙げられる。なかで も、炭素数 1ないし 4のアルキル基、炭素数 7ないし 10のァラルキル基が好まし い。 R 14で示される 「置換されていてもよい炭化水素基」 の置換基としては、 前 記の R 2で示される 「置換されていてもよい炭化水素基」 の置換基と同様の数、 同 様のものが挙げられる。 R14で表される一 ZR15は、 R1Qで表される一 ZR15と 同様のものが挙げられる。 As the hydrocarbon group of the “optionally substituted hydrocarbon group” represented by R 14 , the hydrocarbon group represented by the aforementioned “optionally substituted hydrocarbon group” represented by R 2 And the same as the group. Preferably, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, not having 6 carbon atoms And an aryl group having 10 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, and a cycloalkenyl group having 3 to 8 carbon atoms. Among them, an alkyl group having 1 to 4 carbon atoms and an aralkyl group having 7 to 10 carbon atoms are preferable. The substituent of the “optionally substituted hydrocarbon group” represented by R 14 is the same as or similar to the substituent of the “optionally substituted hydrocarbon group” represented by the aforementioned R 2. And the like. One ZR 15 represented by R 14 include the same as one ZR 15 represented by R 1Q.
R14としては、 置換されていてもよい炭化水素基が好ましい。 なかでも、 炭素 数 1ないし 12のアルキル基、炭素数 2ないし 12のアルケニル基、炭素数 2ない し 12のアルキニル基、炭素数 7ないし 10のァラルキル基、炭素数 3ないし 8の シクロアルキル基、炭素数 3ないし 8のシクロアルケニル基等が好ましく、 とりわ け、炭素数 1ないし 6のアルキル基、炭素数 7ないし 10のァラルキル基が好まし い。 As R 14 , an optionally substituted hydrocarbon group is preferable. Among them, an alkyl group having 1 to 12 carbon atoms, an alkenyl group having 2 to 12 carbon atoms, an alkynyl group having 2 to 12 carbon atoms, an aralkyl group having 7 to 10 carbon atoms, a cycloalkyl group having 3 to 8 carbon atoms, A cycloalkenyl group having 3 to 8 carbon atoms and the like are preferable, and an alkyl group having 1 to 6 carbon atoms and an aralkyl group having 7 to 10 carbon atoms are particularly preferable.
が好ましく、 なかでも とりわけ が好ましい c Is preferred, especially Above all Is preferred c
が好ましく、 なかでも Is preferred, especially
が好ましい。 Is preferred.
化合物 (I) としては、 As the compound (I),
[式中、各記号は前記と同意義を示す。 ]で表される化合物が特に好ましく用いられ る。 具体例として、 [Wherein the symbols have the same meanings as described above. ] Is particularly preferably used. As a specific example,
4ーブロモー N_ {8— 〔 (5—フルォロペンチル) スルファニル〕 —5, 6—ジ メチルー 3—ォキソ一 2, 3—ジヒドロー 1 H—イミダゾ [5, 1 -c] [1, 4] チアジン一 ]·一イリデン }ベンゼンスルホンアミド、 N - {5, 6—ジメチルー 3 —ォキソ一8— 〔 (4, 4, 4, 5, 5—ペンタフルォロペンチル) スルファニル〕 —2, 3—ジヒドロ— 1H—イミダゾ [5, 1— c][l, 4]チアジン— 1一イリデ ン} (フエニル) メタンスルホンアミド、 N— {5, 6—ジメチル _ 3—ォキソ一 8 - 〔 (4, 4, 4, 5, 5一ペンタフルォロペンチル) スルファニル〕 -2, 3 —ジヒドロ— 1H—イミダゾ [5, 1 -c] [1, 4]チアジン一 1—イリデン } 一 4 一メチルベンゼンスルホンアミド、 4_ブロモ— N— 〔8— (イソペンチルスルフ ァニル)一 5, 6—ジメチルー 3—ォキソ一 2, 3—ジヒドロー 1H—イミダゾ [5, 1一 c][l, 4]チアジン— 1—イリデン〕 ベンゼンスルホンアミドなどが挙げら れる。 4-bromo-N_ {8— [(5-fluoropentyl) sulfanyl] —5,6-dimethyl-3-oxo-1,2,3-dihydro-1 H-imidazo [5,1-c] [1,4] thiazine-1] · 1-ylidene} benzenesulfonamide, N- {5,6-dimethyl-3—oxo-18 — [(4,4,4,5,5-pentafluoropentyl) sulfanyl] —2,3-dihydro—1H— Imidazo [5,1—c] [l, 4] thiazine—11-ylidene} (phenyl) methanesulfonamide, N— {5,6-dimethyl_3-oxo-1-8- [(4, 4, 4, 5,5-Pentafluoropentyl) sulfanyl] -2,3 -dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1 1-ylidene} 1-4 1-methylbenzenesulfonamide, 4_bromo-N- [8- (isopentylsulfanyl) -1, 6, -dimethyl-3-oxo-1,2,3-dihydro-1H-imidazo [5,11c] [l, 4] thiazine-1-ylidene] benzenesulfonamide and the like.
以下に一般式 (I) で表される化合物またはその塩の合成法について示す。 一般式 (I) において R1がー (S) n— R2、 Hereinafter, a method for synthesizing the compound represented by the general formula (I) or a salt thereof will be described. In the general formula (I), R 1 is-(S) n — R 2 ,
X Y がX Y
が である化合物 (I X) またはその塩の合成 Synthesis of compound (IX) or a salt thereof in which is
(V I I) (V I I)
( I I I) (I X) (I I I) (I X)
反応 1 2 — ァ ミ ノ — 3 , 3 — ジ ク ロ ロ ア ク リ ロ ニ ト リ ル (2-amino-3,3-dichloroacrylonitrile, 以下 AD ANと略することがある。 ) を 原料とし、 一般式 R1Q-N COで表されるイソシァネートと反応させることにより、 一般式 (I I) で表される 5-ジクロロメチレンイミダゾリジン化合物またはその 塩を得る。 Reaction 1 2 — Amino — 3, 3 — Dichloroacrylonitrile (2-amino-3,3-dichloroacrylonitrile, hereafter sometimes abbreviated as ADAN) By reacting with an isocyanate represented by R 1Q -NCO, a 5-dichloromethyleneimidazolidine compound represented by the general formula (II) or a salt thereof is obtained.
R1 G- NC〇で表されるイソシァネート化合物は原料に対して 0.1ないし 10 当量、好ましくは 0.2ないし 5当量、さらに好ましくは 1ないし 2当量を用いる。 R 1 Q-N C Oで表されるイソシァネ一ト化合物は好ましくは R 10が R 15 S O 2であ るスルホ二ルイソシァネート化合物であり、 さらに好ましくは R 15が置換されて いてもよいベンゼン環であり、 例えば、 ベンゼンスルホ二ルイソシァネート、 p- トルエンスルホニルイソシァネート、 P-フルォロベンゼンスルホ二ルイソシァネ一 ト、 P-クロ口ベンゼンスルホ二ルイソシァネート、 p-ブロモベンゼンスルホ二ルイ ソシァネートなどが特に好ましい。これらのイソシァネート類は対応するアミドか ら自体公知の方法で調製することができる。 この反応は溶媒中で有利に行われる。 . これらの反応は通常反応に影響を与えない溶媒中で行われる。溶媒としては、反応 に影響を与えないものであれば特定されないが、 アミド系溶媒(例、 N, N—ジメ チルホルムアミド、 N, N—ジメチルァセトアミド、 N-メチルピロリドンなど)、 ハロゲン化炭化水素系溶媒(例、ジクロロメタン、クロ口ホルム、四塩化炭素、 1,2- ジクロロェタン、 クロ口ベンゼンなど) 、 二トリル系溶媒(例、 ァセトニトリルな ど) 、 エステル系溶媒 (例、 ギ酸ェチル、 酢酸ェチル、 酢酸 tert-ブチルなど) 、 ケトン系溶媒 (例、ァセトン、メチルェチルケトン、メチルイソプチルケトンなど)、 エーテル系溶媒 (例、 テトラヒドロフラン、 ジェチルエーテル、 ジイソプロピルェ —テル、 ジォキサン、 ジメトキシェタンなど) 、 炭化水素系溶媒 (例、 へキサン、 ベンゼン、 トルエンなど) 、 芳香族有機塩基系溶媒 (例、 ピリジン、 2,4,6-トリメ チリレビリジン、 ピコリン、 2,6-ルチジン、 1,8-ジァザビシクロ [5, 4,0]- 7-ゥンデセ ンなど) など、 あるいはこれらの混合溶媒が挙げられる。 なかでも八ロゲン化炭化 水素系溶媒、 エステル系溶媒、 エーテル系溶媒、 炭化水素系溶媒などが好ましく、 ジクロロメタン、 クロ口ホルム、 テトラヒドロフラン、 ジェチルエーテル、 トルェ ンなどが特に好ましい。 これらの反応には適当な有機塩基(例、 ピリジン、 2, 4, 6- トリメチルピリジン、 ピコリン、 4-ジメチルァミノピリジン、 2, 6-ルチジン、 1,8- ジァザビシクロ [5, 4,0] -7-ゥンデセンなどの芳香族塩基、 トリェチルァミン、 ジメ チルァ二リンなどの 3級ァミンなど)の存在下に行われてもよい。塩基は 0ないし 10当量、好ましくは 0ないし 1当量を用い、反応温度は — 40°Cないし 15 ΟΤ 好ましくは 0°Cないし 100°C、反応時間は 2分間ないし 96時間、好ましくは 5 分間ないし 60時間である。 The isocyanate compound represented by R 1 G —NC〇 is used in an amount of 0.1 to 10 equivalents, preferably 0.2 to 5 equivalents, more preferably 1 to 2 equivalents, based on the amount of the raw material. The isocyanate compound represented by R 1 Q- NCO is preferably a sulfonyl isocyanate compound in which R 10 is R 15 SO 2 , more preferably a benzene ring in which R 15 may be substituted, For example, benzenesulfonyl isocyanate, p-toluenesulfonyl isocyanate, P-fluorobenzenesulfonyl isocyanate, P-chlorobenzenesulfonyl isocyanate, and p-bromobenzenesulfonyl isocyanate are particularly preferable. These isocyanates can be prepared from the corresponding amide by a method known per se. This reaction is advantageously performed in a solvent. These reactions are usually performed in a solvent that does not affect the reaction. Solvents are not specified as long as they do not affect the reaction, but amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, etc.), halogenated Hydrocarbon solvents (eg, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, chloroform benzene, etc.), nitrile solvents (eg, acetonitrile, etc.), ester solvents (eg, ethyl formate, Ethyl acetate, tert-butyl acetate, etc.), ketone solvents (eg, acetone, methyl ethyl ketone, methyl isobutyl ketone, etc.), ether solvents (eg, tetrahydrofuran, getyl ether, diisopropyl ether, dioxane, dimethoxy) ), Hydrocarbon solvents (eg, hexane, benzene, toluene, etc.), aromatic organic salts Base solvents (eg, pyridine, 2,4,6-trimethyl revilidine, picoline, 2,6-lutidine, 1,8-diazabicyclo [5,4,0] -7-indene), etc., or a mixture thereof Solvents. Among them, octogenated hydrocarbon solvents, ester solvents, ether solvents, hydrocarbon solvents and the like are preferable, and dichloromethane, chloroform, tetrahydrofuran, getyl ether, toluene and the like are particularly preferable. Suitable organic bases (eg, pyridine, 2, 4, 6- Aromatic bases such as trimethylpyridine, picoline, 4-dimethylaminopyridine, 2,6-lutidine, 1,8-diazabicyclo [5,4,0] -7-indenecene, and tertiary such as triethylamine and dimethylalanine Amin, etc.). The base is used in an amount of 0 to 10 equivalents, preferably 0 to 1 equivalent, the reaction temperature is — 40 ° C to 15 ° C, preferably 0 ° C to 100 ° C, and the reaction time is 2 minutes to 96 hours, preferably 5 minutes to 60 hours.
反応 2 Reaction 2
一般式 (I I) で表される 5-ジクロロメチレンイミダゾリジン化合物またはその 塩を原料とし、 塩基の存在下にー般式R〇C〇CH2CH2SHで表されるメルカ ブト化合物 (Rはメチル、 ェチル、 プロピル、 イソプロピル、 2-ェチルへキシルな どのアルキル基を表す。 ) と反応させることにより、 一般式 (I I I) で表される イミダゾリジン化合物 (Rは前記アルキル基を示す。 ) またはその塩を得る。 Starting from a 5-dichloromethyleneimidazolidine compound represented by the general formula (II) or a salt thereof, and in the presence of a base, a mercapto compound represented by the general formula R〇C〇CH 2 CH 2 SH (where R is Represents an alkyl group such as methyl, ethyl, propyl, isopropyl, 2-ethylhexyl, etc.), and is reacted with an imidazolidine compound represented by the general formula (III) (R represents the above alkyl group) or Obtain the salt.
ROC〇CH2CH2SHで表されるメルカブト化合物は原料に対して 0.5な いし 20当量、好ましくは 1ないし 10当量、 さらに好ましくは 2ないし 5当量を 用いる。塩基としては、 例えば、水素化アルカリ金属または水素化アルカリ土類金 属 (例、 水素化ナトリウム、 水素化カリウム、 水素化カルシウムなど) 、 アルカリ 金属のアルコキシド (例、 ナトリウムメトキシド、 ナトリウムエトキシド、 力リウ ム tert-ブトキシドなど)、無機塩基(例、炭酸水素ナトリゥム、炭酸ナトリゥム、 炭酸水素力リゥム、 炭酸力リゥム、 水酸化ナトリゥム、 水酸化力リゥム、 水酸化力 ルシゥムなど)、有機塩基(例、 ピリジン、 2, 4,6-トリメチルピリジン、 ピコリン、 4 -ジメチルァミノピリジン、 2, 6-ルチジン、 1,8_ジァザビシクロ[5,4,0]-7_ゥンデ センなどの芳香族塩基、 トリェチルァミン、 ジメチルァニリンなどの 3級ァミンな ど) 、 アルカリ金属 (例、 ナトリウム、 カリウムなど) 、 金属水素化物 (例、 水素 化リチウムアルミニウム、水素化ホウ素リチウム、水素化ジイソブチルアルミニゥ ムなど) 、 アルキルリチウム (例、 メチルリチウム、 ブチルリチウムなど) 、 また はアルカリ金属アミド (例、 リチウムジイソプロピルアミド、 リチウムピストリメ チルシリルアミド、ナトリウムビストリメチルシリルアミドなど)などが挙げられ、 中でも水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 アルキルリチウム、またはアルカリ金属アミドなどが好ましぐ水素化ナトリウム、 ナトリウムェ卜キシド、 カリウム tert-ブトキシド、 炭酸ナトリウム、炭酸水素力 リウム、 炭酸カリウム、 4-ジメチルァミノピリジン、 トリェチルァミン、 メチルリ チウム、 ブチルリチウム、 リチウムジイソプロピルアミド、 リチウムビストリメチ ルシリルアミドなどが特に好ましい。これらのうち液体のものは溶媒としても用い られる。塩基は 1ないし 5 0当量、 好ましくは 2ないし 1 0当量、 さらに好ましく は 2ないし 5当量を用いる。 これらの反応は通常溶媒中で有利に行われる。溶媒は 反応に影響を与えないものであれば特定されないが、 アミド系溶媒(例、 N, N— ジメチルホルムアミド、 N, N—ジメチルァセ卜アミド、 N-メチルピロリドンな ど)、エーテル系溶媒(例、テトラヒドロフラン、ジェチルエーテル、ジォキサン、 ジメトキシェタンなど)、スルホキシド系溶媒 (例、ジメチルスルホキシドなど)、 アルコール系溶媒 (例、 メタノール、 エタノール、 2-プロパノール、 tert-ブタノ ールなど) 、 二トリル系溶媒 (例、 ァセトニトリルなど) 、 'エステル系溶媒 (例、 ギ酸ェチル、 酢酸ェチル、 酢酸 ter t-ブチルなど) 、 炭化水素系溶媒(例、 へキサ ン、ベンゼン、 トルエンなど)、ハロゲン化炭化水素系溶媒 (例、ジクロロメタン、 クロ口ホルム、 四塩化炭素、 1, 2 -ジクロロェタン、 クロ口ベンゼンなど) 、 有機塩 基系溶媒 (例、 ピリジン、 2, 4, 6-トリメチルピリジン、 ピコリン、 4-ジメチルアミ ノピリジン、 2, 6 -ルチジン、 1,8-、クァザビシクロ [5, 4, 0] - 7 -ゥンデセンなどの芳香 族塩基、 トリェチルァミン、 ジメチルァニリンなどの 3級アミンなど) 、 液体アン モニァなど、 あるいはこれらの混合溶媒が挙げられる。 なかでも、 N, N—ジメチ ルホルムアミド、 N, N—ジメチルァセトアミド、 メタノール、 エタノール、 2- プロパノール、 ter t-ブ夕ノール、 ピリジン、 テ卜ラヒドロフラン、 ジォキサン、 ジメトキシェタン、 ァセトニトリルなどが好ましい。反応温度は 一 4 0 ^: ないし 1 5 O 、好ましくは 0 °Cないし 1 0 0 °C、 反応時間は 5分間ないし 9 6時間、 好 ましくは 3 0分間ないし 4 8時間である。 The mercapto compound represented by ROC〇CH 2 CH 2 SH is used in an amount of 0.5 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 2 to 5 equivalents, based on the amount of the raw material. Examples of the base include alkali metal hydride or alkaline earth metal hydride (eg, sodium hydride, potassium hydride, calcium hydride, etc.), alkali metal alkoxide (eg, sodium methoxide, sodium ethoxide, Sodium tert-butoxide, etc.), inorganic bases (eg, sodium bicarbonate, sodium carbonate, sodium bicarbonate, sodium carbonate, sodium hydroxide, sodium hydroxide, sodium hydroxide), organic bases (eg, sodium tert-butoxide) Aromatic bases such as pyridine, 2,4,6-trimethylpyridine, picoline, 4-dimethylaminopyridine, 2,6-lutidine, 1,8_diazabicyclo [5,4,0] -7_indene, Tertiary amines such as triethylamine, dimethylaniline, etc.), alkali metals (eg, sodium, potassium, etc.), metal hydrogenation (Eg, lithium aluminum hydride, lithium borohydride, diisobutylaluminum hydride, etc.), alkyl lithium (eg, methyl lithium, butyl lithium, etc.), or alkali metal amide (eg, lithium diisopropylamide, lithium piste) Sodium hydride, which is preferably an alkali metal hydride, an alkali metal alkoxide, an inorganic base, an organic base, an alkyllithium, or an alkali metal amide. Particularly preferred are sodium ethoxide, potassium tert-butoxide, sodium carbonate, potassium bicarbonate, potassium carbonate, 4-dimethylaminopyridine, triethylamine, methyllithium, butyllithium, lithium diisopropylamide, lithium bistrimethylsilylamide and the like. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent. Solvents are not specified as long as they do not affect the reaction, but amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, etc.), ether solvents (eg, , Tetrahydrofuran, getyl ether, dioxane, dimethoxyethane, etc.), sulfoxide solvents (eg, dimethyl sulfoxide), alcohol solvents (eg, methanol, ethanol, 2-propanol, tert-butanol, etc.), nitrile Solvent (eg, acetonitrile, etc.), ester solvent (eg, ethyl formate, ethyl acetate, tert-butyl acetate, etc.), hydrocarbon solvent (eg, hexane, benzene, toluene, etc.), halogenated carbon Hydrogen solvents (e.g., dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, ), Organic base solvents (eg, pyridine, 2,4,6-trimethylpyridine, picoline, 4-dimethylaminopyridine, 2,6-lutidine, 1,8-, quasabicyclo [5,4,0] -7 Aromatic bases such as -indene, tertiary amines such as triethylamine and dimethylaniline), liquid ammonia and the like, or a mixed solvent thereof. Among them, N, N-dimethylformamide, N, N-dimethylacetamide, methanol, ethanol, 2-propanol, tert-butanol, pyridine, tetrahydrofuran, dioxane, dimethoxyethane, acetonitrile, etc. preferable. The reaction temperature is from 140 to 150 ° C., preferably from 0 ° C. to 100 ° C., and the reaction time is from 5 minutes to 96 hours, preferably from 30 minutes to 48 hours.
反応 3 Reaction 3
一般式(I I I ) で表されるイミダゾリジン化合物またはその塩を原料とし、塩基 と反応させることにより、 一般式 ( I V) で表されるイミダゾリジン化合物 (Rは 上記アルキル基を示す。 ) またはその塩を得る。 The imidazolidine compound represented by the general formula (III) or a salt thereof is used as a raw material and reacted with a base to give an imidazolidine compound represented by the general formula (IV) (R represents the above alkyl group) or a compound thereof. Get the salt.
塩基としては、前記反応 2で示した塩基などが挙げられ、好ましい例としては例 えば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 アルキルリチウム、またはアル力リ金属アミドなどが好ましく、水素化ナトリゥム、 ナトリウムエトキシド、 カリウム tert-ブトキシド、 炭酸ナトリウム、 炭酸力リウ ム、 4 -ジメチルァミノピリジン、 メチルリチウム、 ブチルリチウム、 リチウムジィ ソプロピルアミド、 リチウムピストリメチルシリルアミドなどが特に好ましい。塩 基は 1ないし 5 0当量、好ましくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。 これらの反応は通常溶媒中で有利に行われる。溶媒は反応に影響 を与えないものであれば特定されないが、上記反応 2で示した溶媒などが用いられ、 好ましくはアミド系溶媒、 エーテル系溶媒、 アルコール系溶媒、 二トリル系溶媒、 有機塩基系溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも、 N, N— ジメチルホルムアミド、 テトラヒドロフラン、 ジォキサン、 ジメトキシェタン、 メ タノ一ル、 エタノール、 2-プロパノール、 ter t-ブタノール、 ァセトニトリル、 ピ リジンなどが特に好ましい。 反応温度は一 4 0 °Cないし 1 5 0 °C、 好ましくは 2 O t:ないし 1 2 0 °C、反応時間は 5分間ないし 9 6時間、好ましくは 3 0分間ない し 2 4時間である。 Examples of the base include the bases shown in the above Reaction 2, and preferable examples are For example, an alkali metal hydride, an alkali metal alkoxide, an inorganic base, an organic base, an alkyl lithium, or an alkali metal amide is preferable. And 4-dimethylaminopyridine, methyllithium, butyllithium, lithium disopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably an amide solvent, an ether solvent, an alcohol solvent, a nitrile solvent, an organic base solvent Solvents, or a mixture thereof, among which N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol, acetonitrile, pyridine Etc. are particularly preferred. The reaction temperature is 140 ° C. to 150 ° C., preferably 2 Ot: to 120 ° C., and the reaction time is 5 minutes to 96 hours, preferably 30 minutes to 24 hours. .
上記反応 2および反応 3は途中で生成物を単離精製することなく連続的に行う ことで、 有利に実施できる。 一般式 (I I ) で表される上記 5-ジクロロメチレン ィミダゾリジン化合物またはその塩を原料とし、塩基の存在下に一般式 R O C O C H 2 C H 2 S Hで表される上記メルカプト化合物と反応させることにより、 一般式 ( I V) で表されるイミダゾリジン化合物またはその塩を得る。 The above Reaction 2 and Reaction 3 can be advantageously performed by continuously performing the reaction without isolating and purifying the product during the reaction. By reacting the above 5-dichloromethylene imidazolidine compound represented by the general formula (II) or a salt thereof as a raw material with the above mercapto compound represented by the general formula ROCOCH 2 CH 2 SH in the presence of a base, the general formula An imidazolidine compound represented by (IV) or a salt thereof is obtained.
反応 4 Reaction 4
一般式(I V)で表されるイミダゾリジン化合物(Rは前記アルキル基を示す。) またはその塩を原料とし、 一般式(V) で表されるひ位に脱離基 Lを有するケトン 化合物 (Lは塩素、 臭素、 ヨウ素などのハロゲン原子、 あるいはメタンスルホニル ォキシ、 P-トルエンスルホニルォキシ、 トリフルォロメ夕ンスルホニルォキシなど のスルホニルォキシ基などを表す。) またはその塩と塩基の存在下に反応させるこ とにより、 一般式 (V I ) で表されるィミダゾチアジン化合物 (Rは上記アルキル 基を示す。 ) またはその塩を得る。 Starting from an imidazolidine compound represented by the general formula (IV) (R represents the above-mentioned alkyl group) or a salt thereof, a ketone compound having a leaving group L at the octyl position represented by the general formula (V) ( L represents a halogen atom such as chlorine, bromine or iodine or a sulfonyloxy group such as methanesulfonyloxy, P-toluenesulfonyloxy, trifluoromethanesulfonyloxy, etc.) or a salt thereof in the presence of a base By the reaction, an imidazothiazine compound represented by the general formula (VI) (R represents the above alkyl group) or a salt thereof is obtained.
一般式 (V)で表される化合物またはその塩のうち脱離基 Lがハロゲン原子であ る化合物は対応するケトン化合物から自体公知の方法で α位をハロゲン化するこ とで調製することができる。一般式 (V) で表される化合物またはその塩のうち脱 離基 Lがスルホニルォキシ基である化合物は対応する -ヒドロキシケトン化合物 から例えば後述の反応 1 0で示す方法に代表される自体公知の方法で水酸基をス ルホニル化することで調製することができる。 In the compound represented by the general formula (V) or a salt thereof, the leaving group L is a halogen atom. Can be prepared from the corresponding ketone compound by halogenating the α-position by a method known per se. The compound represented by the general formula (V) or a salt thereof, in which the leaving group L is a sulfonyloxy group, is a compound known per se represented by the corresponding -hydroxyketone compound, for example, the method shown in Reaction 10 below. It can be prepared by sulfonylation of a hydroxyl group by the method described in the above.
一般式 (V)で表される α位に脱離基 Lを有するケトン化合物またはその塩にお いて、 Lは好ましくは塩素、 臭素、 ヨウ素などのハロゲン原子であり、 さらに好ま しくは塩素または臭素である。 上記ケトン化合物またはその塩は原料に対して 0 . 2ないし 1 0当量、好ましくは 1ないし 5当量、 さらに好ましくは 1ないし 3当量 を用いる。塩基としては、 上記反応 2で示した塩基などが挙げられ、好ましい例と しては例えば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有 機塩基、 アルキルリチウム、 またはアルカリ金属アミドが好ましく、水素化ナ卜リ ゥム、ナトリウムメトキシド、ナ卜リゥムェトキシド、力リゥム tert-ブトキシド、 炭酸水素ナトリウム、 炭酸ナトリウム、 炭酸水素カリウム、 炭酸カリウム、 トリエ チルァミン、ジィソプロピルェチルアミン、ピリジン、 4-ジメチルァミノピリジン、 1,8-ジァザビシクロ[5,4,0]-7-ゥンデセン、 メチルリチウム、 ブチルリチウム、 リ チウムジイソプロピルアミド、リチウムピストリメチルシリルアミドなどが特に好 ましい。 これらのうち液体のものは溶媒としても用いられる。塩基は 1ないし 5 0 当量、 好ましくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。 これらの反応は通常溶媒中で有利に行われる。溶媒は反応に影響を与えないもので あれば特定されないが、上記反応 2で示した溶媒などが用いられ、好ましくはアミ ド系溶媒、 エーテル系溶媒、 ケトン系溶媒、 エステル系溶媒、 二トリル系溶媒、 炭 化水素系溶媒、 ハロゲン化炭化水素系溶媒、有機塩基系溶媒など、 あるいはこれら の混合溶媒が挙げられ、 なかでも、 N, N—ジメチルホルムアミド、 N, N—ジメ チリレアセトアミド、 チトラヒドロフラン、 ジォキサン、 ジメトキシェタン、 ァセト ン、 2-ブ夕ノン、 酢酸ェチル、 酢酸 tert -プチル、 ァセトニトリル、 トルエン、 ジ クロロメタン、クロ口ホルム、 1, 2-ジクロロェタン、ピリジンなどが特に好ましい。 反応温度は一 4 0 °Cないし 1 5 0 °C,好ましくは 0 ないし 1 0 0 °C、反応時間は 5分間ないし 9 6時間、 好ましくは 3 0分間ないし 4 8時間である。 反応 5 In the ketone compound having a leaving group L at the α-position represented by the general formula (V) or a salt thereof, L is preferably a halogen atom such as chlorine, bromine or iodine, and more preferably chlorine or bromine. It is. The ketone compound or a salt thereof is used in an amount of 0.2 to 10 equivalents, preferably 1 to 5 equivalents, more preferably 1 to 3 equivalents, based on the raw material. Examples of the base include the bases shown in Reaction 2 above, and preferable examples thereof include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides. , Sodium hydride, sodium methoxide, sodium methoxide, potassium tert-butoxide, sodium hydrogen carbonate, sodium carbonate, potassium hydrogen carbonate, potassium carbonate, triethylamine, disopropylethylamine, pyridine, 4 -Dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-indene, methyllithium, butyllithium, lithium diisopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably, an amide solvent, an ether solvent, a ketone solvent, an ester solvent, and a nitrile solvent are used. Solvents, hydrocarbon solvents, halogenated hydrocarbon solvents, organic base solvents, etc., and mixtures thereof. Among them, N, N-dimethylformamide, N, N-dimethylileacetamide, titra Hydrofuran, dioxane, dimethoxyethane, acetate, 2-butenonone, ethyl acetate, tert-butyl acetate, acetatetonitrile, toluene, dichloromethane, chloroform, 1,2-dichloroethane, pyridine and the like are particularly preferred. The reaction temperature is from 140 ° C to 150 ° C, preferably from 0 to 100 ° C, and the reaction time is from 5 minutes to 96 hours, preferably from 30 minutes to 48 hours. Reaction 5
一般式 (V I )で表されるイミダゾチアジン化合物(Rは上記アルキル基を示す。 ) またはその塩を原料とし、 酸の存在下に脱水反応に付すことにより、 一般式(V I I ) で表されるイミダゾチアジン化合物 (Rは上記アルキル基を示す。 ) を得る。 用 られる酸の好ましい例としては、例えば八口酢酸(例、 トリフルォロ酢酸な ど) 、 無機酸 (例、 塩化水素酸、 臭化水素酸、 硫酸、 リン酸、 過塩素酸など) 、 有 機カルボン酸(例、ギ酸、酢酸、クェン酸、酒石酸、シユウ酸など)、ルイス酸(例、 亜鉛一酢酸、 3フッ化ホウ素エーテル錯体など) 、 有機スルホン酸 (例、 メタンス ルホン酸、 オクタンスルホン酸、 ベンゼンスルホン酸、 P -トルエンスルホン酸、 カンファースルホン酸など)等が用いられ、中でもトリフルォロ酢酸、塩化水素酸、 硫酸、 リン酸、 酢酸、 メタンスルホン酸、 P -トルエンスルホン酸、 カンファース ルホン酸などが特に好ましい。これらのうち液体のものは溶媒としても用いられる。 酸は原料に対して 0 . 0 5ないし 1 0当量、好ましくは 0 . 1ないし 5当量、 さらに 好ましくは 0 . 2ないし 2当量を用いる。 通常生成した水を除くために脱水剤を共 存させることで反応が有利に行われる。脱水剤としてはモレキユラ一シーブス 4 A、 酸無水物 (例、 無水酢酸、 無水プロピオン酸、 無水安息香酸など) などが用いられ る。例えばモレキユラ一シ一ブス 4 A ( 1 Z 1 6〜2 0 0メッシュ)であれば原料 に対して 5重量%ないし 1 0倍量、好ましくは 1 0重量%ないし 5倍量を、 あるい は酸無水物であれば原料に対して 0 . 5ないし 2 0当量、 好ましくは 1ないし 1 0 当量、 さらに好ましくは 1ないし 5当量をそれぞれ用いる。 これらの反応は通常溶 媒中で有利に行われる。溶媒は反応に影響を与えないものであれば特定されないが、 好ましくは有機酸系溶媒(例、ギ酸、酢酸、プロピオン酸など)、アミド系溶媒(例、 N, N—ジメチルホルムアミド、 N, N—ジメチルァセトアミドなど) 、 エーテル 系溶媒(例、 テトラヒドロフラン、 ジェチルェ一テル、 ジォキサン、 ジメトキシェ タンなど) 、 スルホキシド系溶媒 (例、 ジメチルスルホキシドなど) 、 アルコール 系溶媒 (例、 メタノール、 エタノール、 プロパノール、 イソプロパノール、 イソブ タノール、 ter t-ブタノールなど)、 二トリル系溶媒 (例、 ァセトニトリルなど)、 エステル系溶媒 (例、 ギ酸ェチル、 酢酸ェチル、 酢酸 tert-ブチルなど) 、 炭化水 素系溶媒(例、 へキサン、 ベンゼン、 トルエン、 キシレン類など) 、 ハロゲン化炭 化水素系溶媒 (例、 ジクロロメタン、 クロ口ホルム、 四塩化炭素、 1, 2-ジクロロェ タン、 クロ口ベンゼンなど) など、 あるいはこれらの混合溶媒が挙げられる。 なか でも、 酢酸、 N, N—ジメチルホルムアミド、 テトラヒドロフラン、 ジォキサン、 ジメトキシェタン、 tert -ブ夕ノール、 ァセトニトリル、 酢酸ェチル、 酢酸 ter t- プチル、 トルエン、 キシレン類、 クロ口ホルム、 1, 2 -ジクロロェタンなどが好まし い。 反応温度は - 4 O t:ないし 1 8 0 °C、 好ましくは 0 °Cないし 1 2 0 °C, 反応時 間は 5分間ないし 1 2 0時間、 好ましくは 1 0分間ないし 7 2時間である。 The imidazothiazine compound represented by the general formula (VI) (R represents the above-mentioned alkyl group) or a salt thereof is subjected to a dehydration reaction in the presence of an acid, whereby the compound represented by the general formula (VII) is obtained. (Wherein R represents the above alkyl group). Preferred examples of the acid used include, for example, octaacetic acid (eg, trifluoroacetic acid), inorganic acid (eg, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, perchloric acid, etc.), and organic carboxylic acids. Acids (eg, formic acid, acetic acid, citric acid, tartaric acid, oxalic acid, etc.), Lewis acids (eg, zinc monoacetic acid, boron trifluoride etherate, etc.), organic sulfonic acids (eg, methanesulfonic acid, octanesulfonic acid, Benzenesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc.), among which trifluoroacetic acid, hydrochloric acid, sulfuric acid, phosphoric acid, acetic acid, methanesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc. Is particularly preferred. Of these, liquid ones are also used as solvents. The acid is used in an amount of 0.05 to 10 equivalents, preferably 0.1 to 5 equivalents, and more preferably 0.2 to 2 equivalents, based on the raw material. The reaction is advantageously carried out by coexisting a dehydrating agent to remove the usually generated water. As the dehydrating agent, molecular sieves 4A, acid anhydride (eg, acetic anhydride, propionic anhydride, benzoic anhydride, etc.) are used. For example, if the molecular weight is 4 A (1 Z 16 to 200 mesh), the amount is 5% to 10 times, preferably 10% to 5 times, or 10% by weight of the raw material. If it is an acid anhydride, it is used in an amount of 0.5 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents, based on the raw materials. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but is preferably an organic acid-based solvent (eg, formic acid, acetic acid, propionic acid, etc.), an amide-based solvent (eg, N, N-dimethylformamide, N, N —Dimethylacetamide, etc.), ether solvents (eg, tetrahydrofuran, getylether, dioxane, dimethoxyethane, etc.), sulfoxide solvents (eg, dimethylsulfoxide, etc.), alcohol solvents (eg, methanol, ethanol, propanol, Isopropanol, isobutanol, tert-butanol, etc., nitrile solvents (eg, acetonitrile, etc.), ester solvents (eg, ethyl formate, ethyl acetate, tert-butyl acetate, etc.), hydrocarbon solvents (eg, Hexane, benzene, toluene, xylenes, etc.), halogenated carbon Hydrogenated solvents (eg, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, cyclobenzene, etc.) and the like, and a mixed solvent thereof are exemplified. Among them, acetic acid, N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, tert-butanol, acetonitrile, ethyl acetate, tert-butyl acetate, toluene, xylenes, chloroform, 1,2- Dichloroethane is preferred. The reaction temperature is −4 Ot: to 180 ° C, preferably 0 ° C to 120 ° C, and the reaction time is 5 minutes to 120 hours, preferably 10 minutes to 72 hours. .
反応 6 Reaction 6
一般式(V I I )で表されるイミダゾチアジン化合物(Rは上記アルキル基を示 す。) またはその塩を原料とし、塩基と反応させることにより、一般式(V I I I ) で表されるイミダゾチアジン化合物またはその塩を得る。 The imidazothiazine compound represented by the general formula (VIII) is obtained by reacting the imidazothiazine compound represented by the general formula (VII) (R represents the above alkyl group) or a salt thereof with a base. A compound or a salt thereof is obtained.
塩基としては、上記反応 2で示した塩基などが挙げられ、好ましい例としては例 えば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 アルキルリチウム、またはアル力リ金属アミドなどが好ましく、水素化ナトリゥム、 ナトリウムメトキシド、 ナトリウムエトキシド、 カリウム tert-ブトキシド、水酸 化ナトリウム、 水酸化カリウム、 炭酸ナトリウム、 炭酸カリウム、 ジイソプロピル ェチルァミン、 4-ジメチルァミノピリジン、 1 , 8-ジァザビシク口 [5, 4, 0] - 7-ゥンデ セン、 メチルリチウ厶、 ブチルリチウム類、 リチウムジイソプロピルアミド、 リチ ゥムピストリメチルシリルアミドなどが特に好ましい。 塩基は 1ないし 2 0当量、 好ましくは 2ないし 1 0当量、 さらに好ましくは 2から 5当量を用いる。 これらの 反応は通常溶媒中で有利に行われる。溶媒は反応に影響を与えないものであれば特 定されないが、上記反応 2で示した溶媒などが用いられ、好ましくはアミド系溶媒、 エーテル系溶媒、 アルコール系溶媒、 二トリル系溶媒、 有機塩基系溶媒など、 ある いはこれらの混合溶媒が挙げられ、 なかでも、 N, N—ジメチルホルムアミド、 テ トラヒドロフラン、 ジォキサン、 ジメトキシェタン、 メタノール、 エタノール、 2- プロパノール、 tert-ブ夕ノール、ァセトニトリル、ピリジンなどが特に好ましい。 反応温度はー4 0 °Cないし 1 5 O t:、好ましくは 2 0 °Cないし 1 2 0 、反応時間 は 5分間ないし 9 6時間、 好ましくは 3 0分間ないし 2 4時間である。 Examples of the base include the bases shown in Reaction 2 above, and preferable examples thereof include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides. Preferably, sodium hydride, sodium methoxide, sodium ethoxide, potassium tert-butoxide, sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, diisopropylethylamine, 4-dimethylaminopyridine, 1,8-diazavisic mouth [5,4,0] -7-Pendecene, methyllithium, butyllithium, lithium diisopropylamide, and lithium bistrimethylsilylamide are particularly preferred. The base is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, preferably an amide solvent, an ether solvent, an alcohol solvent, a nitrile solvent, an organic base. Solvent, or a mixture of these solvents. Among them, N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol, Acetonitrile, pyridine and the like are particularly preferred. The reaction temperature is −40 ° C. to 15 Ot: preferably 20 ° C. to 120 °, and the reaction time is 5 minutes to 96 hours, preferably 30 minutes to 24 hours.
反応 7 - (a ) 一般式(V I I I )で表されるイミダゾチアジン化合物またはその塩を原料とし、 塩基の存在下に一般式 R 2— L (Lは脱離基を表す。 ) またはその塩と反応させる ことにより、 一般式 ( I X) (n=l) で表されるイミダゾチアジン化合物またはそ の塩を得る。 Reaction 7-(a) By reacting an imidazothiazine compound represented by the general formula (VIII) or a salt thereof as a raw material with a general formula R 2 —L (L represents a leaving group) or a salt thereof in the presence of a base, An imidazothiazine compound represented by the general formula (IX) (n = l) or a salt thereof is obtained.
一般式 R 2— Lで表される化合物またはぞの塩において、 Lは塩素、 臭素、 ヨウ 素などのハロゲン原子、 あるいはメタンスルホニルォキシ、 P-トルエンスルホニル 才キシ、 トリフルォロメタンスルホニルォキシなどのスルホニルォキシ基などが好 ましい。 一般式: 2一 Lで表される化合物またはその塩のうち脱離基 Lがノヽロゲン 原子である化合物は対応するアルコ一ル化合物 R 2— 0 Hまたはその塩から自体公 知の方法で水酸基をハロゲン化することで調製することができる。 一般式 R 2— L で表される化合物またはその塩のうち脱離基 Lがスルホニルォキシ基である化合 物は R 2—〇Hから例えば後述の反応 1 0で示す方法に代表される自体公知の方法 で水酸基をスルホニル化することで調製することができる。 In the compound represented by the general formula R 2 — L or a salt thereof, L is a halogen atom such as chlorine, bromine, iodine, etc., or methanesulfonyloxy, P-toluenesulfonyl, trifluoromethanesulfonyloxy, etc. Sulfonyloxy group and the like are preferable. General formula: 2 a compound represented by one L or compound leaving group L is Nono androgenic atom of its salt corresponding alcohol Ichiru compound R 2 - 0 H or a hydroxyl by a method per se publicly known salts thereof Can be prepared by halogenating Among the compounds represented by the general formula R 2 —L or a salt thereof, the compound in which the leaving group L is a sulfonyloxy group can be converted from R 2 —〇H to a compound represented by, for example, the method shown in Reaction 10 below. It can be prepared by sulfonylating a hydroxyl group by a known method.
上記一般式 R 2 - Lで表される化合物またはその塩は原料に対して 0 . 2ないし 2 0当量、好ましくは 1ないし 1 0当量、 さらに好ましくは 1ないし 5当量を用い る。塩基としては、 上記反応 2で示した塩基などが挙げられ、好ましい例としては 例えば、水素化アル力リ金属、アル力リ金属のアルコキシド、無機塩基、有機塩基、 アルキルリチウム、またはアル力リ金属アミドなどが好ましく、水素化ナトリウム、 ナトリウムメトキシド、 ナトリウムエトキシド、 カリウム ter t-ブ卜キシド、炭酸 ナトリウム、 炭酸カリウム、 トリェチルァミン、 ジィソプロピルェチルァミン、 ピ リジン、 4-ジメチルァミノピリジン、 1 , 8-ジァザビシク口 [5, 4, 0] -7-ゥンデセン、 メチルリチウム、 ブチルリチウム、 リチウムジイソプロピルアミド、 リチウムビス トリメチルシリルアミドなどが特に好ましい。これらのうち液体のものは溶媒とし ても用いられる。塩基は 1ないし 5 0当量、好ましくは 2ないし 1 0当量、 さらに 好ましくは 2ないし 5当量を用いる。これらの反応は通常溶媒中で有利に行われる。 溶媒は反応に影響を与えないものであれば特定されないが、上記反応 2で示した溶 媒などが用いられ、 好ましくはアミド系溶媒、 エーテル系溶媒、 ケトン系溶媒、 ェ ステル系溶媒、 二トリル系溶媒、 アルコール系溶媒、 炭化水素系溶媒、 有機塩基系 溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも N, N—ジメチルホル ムアミド、 N-メチルピロリドン、 テトラヒドロフラン、 ジォキサン、 ジメトキシ ェタン、アセトン、 2-ブ夕ノン、酢酸ェチル、酢酸 tert-ブチル、ァセトニトリル、 トルエン、 ピリジンなどが特に好ましい。反応温度は— 2 0 °Cないし 2 0 O , 好 ましくは 2 0 °Cないし 1 2 0 °C、反応時間は 5分間ないし 1 2 0時間、好ましくは 2 0分間ないし 7 2時間である。 また、 反応 6と反応 7 _ ( a ) は連結的に実施す ることも可能である。 The compound represented by the above general formula R 2 -L or a salt thereof is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents to the starting material. Examples of the base include the bases shown in the above Reaction 2, and preferable examples thereof include, for example, hydrogenated metal alkoxides, metal alkoxides, inorganic bases, organic bases, alkyllithiums, and metal alkoxides. Amides and the like are preferable, and sodium hydride, sodium methoxide, sodium ethoxide, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, disopropylethylamine, pyridine, 4-dimethylaminopyridine , 1,8-Diazavicic mouth [5,4,0] -7-indene, methyllithium, butyllithium, lithium diisopropylamide, lithium bistrimethylsilylamide and the like are particularly preferable. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, preferably an amide solvent, ether solvent, ketone solvent, ester solvent, nitrile Solvents, alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents, and the like, or a mixture thereof. Among them, N, N-dimethylform Muamide, N-methylpyrrolidone, tetrahydrofuran, dioxane, dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred. The reaction temperature is −20 ° C. to 20 O, preferably 20 ° C. to 120 ° C., and the reaction time is 5 minutes to 120 hours, preferably 20 minutes to 72 hours. . Reaction 6 and reaction 7_ (a) can also be performed in a concatenated manner.
反応 7— ( b ) Reaction 7— (b)
一般式(V I I I )で表されるイミダゾチアジン化合物またはその塩を原料とし、 一般式 R 2—〇Hで表されるアルコール化合物またはその塩と光延反応を応用した 脱水縮合反応に付すことにより、 一般式 (I X) (n=l) で表されるイミダゾチア ジン化合物またはその塩を得る。 By subjecting an imidazothiazine compound represented by the general formula (VIII) or a salt thereof as a raw material to a dehydration condensation reaction using a Mitsunobu reaction with an alcohol compound represented by the general formula R 2 —ΔH or a salt thereof, An imidazothiazine compound represented by the general formula (IX) (n = l) or a salt thereof is obtained.
一般式 R 2— O Hで表されるアルコール化合物またはその塩は原料に対して 0 . 2ないし 2 0当量、好ましくは 1ないし 1 0当量、 さらに好ましくは 1ないし 5当 量を用いる。 脱水縮合剤としては、 通常光延反応 [シンセシス (Synthes is) 、 1 頁、 1 9 8 1年] に報告されているホスフィン一ァゾジカルボン酸エステル、 ある いはホスフィン一ァゾジカルボン酸アミド [有機合成化学協会誌、 第 5 5巻、 6 3 1頁、 1 9 9 7年]などが脱水縮合剤として有利に用いられる。 ホスフィンとして は例えば、 トリフエニルホスフィン、 トリ- 0 -トリルホスフィンなどのトリァリー ルホスフィンなど、 あるいはトリメチルホスフィン、 トリブチルホスフィンなどの トリアルキルホスフィンなどが挙げられ、なかでもトリフエニルホスフィン、 トリ ブチルホスフィンなどが好ましい。ァゾジカルボン酸エステルとしては例えば、 ァ ゾジカルボン酸ジェチル、 ァゾジ力ルポン酸ジイソプロピルなどが挙げられる。 ァ ゾジカルボン酸アミドとしては例えば、 N, N, Ν', Ν'—テトラメチルァゾジカ ルポキサミド、 Ν, Ν, N', N'—テトライソプロピルァゾジカルポキサミド、 1,卜 (ァゾジカルポニル) ジピペリジンなどが挙げられる。脱水縮合剤は 1ないし 2 0 当量、 好ましくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。 ホスフィンとァゾジカルボン酸エステルあるいはァゾジカルボン酸アミドは同モ ル数用いるのが好ましい。 これらの反応は通常溶媒中で有利に行われる。溶媒は反 応に影響を与えないものであれば特定されないが、上記反応 2で示した溶媒などが 用いられ、 好ましくはアミド系溶媒、 エーテル系溶媒、 ケトン系溶媒、 エステル系 溶媒、 二トリル系溶媒、 炭化水素系溶媒、 八ロゲン化炭化水素系溶媒、 有機塩基系 溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも N, N—ジメチルホル ムアミド、 テトラヒドロフラン、 ジォキサン、 ジメトキシェタン、 アセトン、 2- ブタノン、 酢酸ェチル、 酢酸 tert -プチル、 ァセトニトリル、 トルエン、 クロロホ ルム、 1, 2-ジクロロェタン、 ピリジンなどが特に好ましい。 反応温度は一 2 0 °Cな いし 1 5 0 °C、好ましくは 2 0 °Cないし 1 2 0 °C、反応時間は 5分間ないし 1 2 0 時間、 好ましくは 2 0分間ないし 6 0時間である。 The alcohol compound represented by the general formula R 2 —OH or a salt thereof is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, and more preferably 1 to 5 equivalents to the starting material. Examples of the dehydrating condensing agent include phosphine monoazodicarboxylates and phosphine monoazodicarboxylates reported in the Mitsunobu reaction [Synthes is, p. 1, 1989] [Journal of the Society of Synthetic Organic Chemistry] 55, 631, 1997] are advantageously used as the dehydrating condensing agent. Examples of the phosphine include triaryl phosphine, triaryl phosphine such as tri-0-tolyl phosphine, and the like, or trialkyl phosphine such as trimethyl phosphine and tributyl phosphine, among which triphenyl phosphine and tributyl phosphine are preferable. . Examples of the azodicarboxylate include acetyl dicarboxylate, diisopropyl azodicarboxylate, and the like. Examples of azodicarboxylic acid amides include N, N, Ν ', Ν'-tetramethyl azodicalpoxamide, Ν, Ν, N', N'-tetraisopropyl azodicarpoxamide, 1, tri (azodicarponyl) And dipiperidine. The dehydrating condensing agent is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. It is preferable to use the same moles of phosphine and azodicarboxylic acid ester or azodicarboxylic acid amide. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction. Preferably used are amide solvents, ether solvents, ketone solvents, ester solvents, nitrile solvents, hydrocarbon solvents, octogenated hydrocarbon solvents, organic base solvents, etc., or a mixture thereof. N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, chloroform, 1,2-dichloroethane, pyridine, etc. Is particularly preferred. The reaction temperature is between 120 ° C and 150 ° C, preferably between 20 ° C and 120 ° C, and the reaction time is between 5 minutes and 120 hours, preferably between 20 minutes and 60 hours. is there.
反応 7 — ( c ) Reaction 7 — (c)
一般式(V I I I )で表されるイミダゾチアジン化合物またはその塩を原料とし、 一般式 R 2— S H (R 2は置換基を有しても良いァリール基を示す。 ) で表される チオール化合物またはその塩と光延反応を応用した脱水縮合反応に付すことによ り、 一般式 ( I X) (n=lまたは 2) で表されるイミダゾチアジン化合物またはそ の塩を得る。 A thiol compound represented by the general formula R 2 —SH (where R 2 represents an aryl group which may have a substituent) from an imidazothiazine compound represented by the general formula (VIII) or a salt thereof as a raw material. Alternatively, an imidazothiazine compound represented by the general formula (IX) (n = l or 2) or a salt thereof is obtained by subjecting the salt thereof to a dehydration condensation reaction applying the Mitsunobu reaction.
一般式 R 2— S Hで表されるチオール化合物またはその塩は原料に対して 0 . 2 ないし 2 0当量、好ましくは 1ないし 1 0当量、 さらに好ましくは 1ないし 5当量 を用いる。脱水縮合剤としては、 例えば上記反応 7— ( b ) で挙げられるホスフィ ンおよびァゾジカルボン酸エステルあるいはァゾジカルボン酸アミドなどが有利 に用いられ、 ホスフィンとしては例えば、 トリフエニルホスフィン、 トリプチルホ スフィンが、 ァゾジカルポン酸エステルとしては例えば、 ァゾジカルボン酸ジェチ リレ、 ァゾジ力ルポン酸ジイソプロピルなどが、 ァゾジカルボン酸アミドとしては例 えば、 N, N, N', N,-テトラメチルァゾジカルポキサミド、 1, 1- (ァゾジカルポ ニル) ジピペリジンなどが好ましい。脱水縮合剤は 1ないし 2 0当量、好ましくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。ホスフィンとァゾ ジカルボン酸エステルあるいはァゾジカルボン酸アミドは同モル数用いるのが好 ましい。 これらの反応は通常溶媒中で有利に行われる。溶媒は反応に影響を与えな いものであれば特定されないが、上記反応 2で示した溶媒などが用いられ、好まし くはアミド系溶媒、 エーテル系溶媒、 ケトン系溶媒、 エステル系溶媒、 二トリル系 溶媒、 炭化水素系溶媒、 ハロゲン化炭化水素系溶媒、 有機塩基系溶媒など、 あるい はこれらの混合溶媒が挙げられ、 なかでも N, N—ジメチルホルムアミド、 テトラ ヒドロフラン、 ジォキサン、 ジメトキシェタン、 アセトン、 2-ブ夕ノン、 酢酸ェチ ル、 酢酸 tert-ブチル、 ァセトニトリル、 トルエン、 クロ口ホルム、 1,2-ジクロロ ェタン、 ピリジンなどが特に好ましい。反応温度は— 20°Cないし 120°C、 好ま しくは 20°Cないし 80°C、反応時間は 10分間ないし 240時間、好ましくは 2 0分間ないし 120時間である。 The thiol compound represented by the general formula R 2 —SH or a salt thereof is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material. As the dehydrating condensing agent, for example, the phosphine and azodicarboxylic acid ester or azodicarboxylic acid amide mentioned in the above reaction 7- (b) are advantageously used. Examples include acetyldiazolate azodicarboxylate and diisopropyl azodicarboxylate. Examples of azodicarboxylate amides include N, N, N ', N, -tetramethylazodicarpoxamide, 1,1- (azodicarpoxamide). Nyl) dipiperidine and the like are preferred. The dehydrating condensing agent is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. It is preferable that phosphine and azodicarboxylic acid ester or azodicarboxylic acid amide be used in the same mole number. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably, an amide solvent, an ether solvent, a ketone solvent, an ester solvent, and nitrile are used. Solvent, hydrocarbon solvent, halogenated hydrocarbon solvent, organic base solvent, etc. Include mixed solvents of these, among which N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, chloroform Mouth form, 1,2-dichloroethane, pyridine and the like are particularly preferred. The reaction temperature is -20 ° C to 120 ° C, preferably 20 ° C to 80 ° C, and the reaction time is 10 minutes to 240 hours, preferably 20 minutes to 120 hours.
一般式 (I) において R1がー NR3R4、 In the general formula (I), R 1 is -NR 3 R 4 ,
X Y がX Y
である化合物 (X I) またはその塩および一般式 (I) において R1がー SR2、 In the compound (XI) or a salt thereof and the general formula (I), R 1 is -SR 2 ,
X Y がX Y
である化合物 (X I I) またはその塩の合成 Of compound (XII) or its salt
反応 8 Reaction 8
一般式 (X) で表されるイミダゾリジン化合物 (Zは S02を示す。 ) またはそ の塩を原料とし、 R3R4NHまたは R12R13NHで表されるァミンまたはその塩 と塩基の存在下に反応させることにより、 一般式 (X I) あるいは (X I I) で表 されるイミダゾリジン化合物 (Zは S〇2を示す。 ) またはその塩を得る。 Imidazolidine compound represented by the general formula (X) (Z represents a S0 2.) Or As a salt as a raw material, Amin or its salt with a base represented by R 3 R 4 NH or R 12 R 13 NH To give an imidazolidine compound (Z represents S 式2 ) represented by the general formula (XI) or (XII) or a salt thereof.
R3R4NHまたは R12R13NHで表されるァミンまたはその塩は 1ないし 50 当量、好ましくは 2ないし 2 0当量、さらに好ましくは 2ないし 1 0当量を用いる。 これらのうち液体のものは溶媒としても用いられる。これらのアミンは適当な酸と の塩としても有利に用いられる。酸としては、 例えば八口酢酸(例、 トリフルォロ 酢酸など)、無機酸(例、塩化水素酸、臭化水素酸、硫酸、 リン酸、過塩素酸など)、 有機カルボン酸 (例、 ギ酸、 酢酸、 クェン酸、 酒石酸、 シユウ酸など) 、 有機スル ホン酸 (例、 メタンスルホン酸、 ベンゼンスルホン酸、 P -トルエンスルホン酸、 カンファースルホン酸など) などが用いられ、 中でも塩化水素酸、 ギ酸、 酢酸、 メ タンスルホン酸、 P-トルエンスルホン酸などが特に好ましい。 酸はァミンに対し て通常等モル用いられる。塩基としては、上記反応 2で示した塩基などが挙げられ、 好ましい例としては例えば、水素化アル力リ金属、アル力リ金属類のアルコキシド、 無機塩基、 有機塩基、 またはアルカリ金属アミドなどが挙げられ、 水素化ナトリウ ム、 ナトリウムエトキシド、 カリウム tert-ブトキシド、 炭酸水素ナトリウム、 炭 酸水素カリウム、 炭酸ナトリウム、 炭酸カリウム、 トリェチルァミン、 ジイソプロ ピルェチルァミン、 ピリジン、 4-ジメチルァミノピリジン、 1, 8 -ジァザビシクロ [5, 4, 0] - 7-ゥンデセン、 リチウムジイソプロピルアミド、 リチウムビストリメチル シリルアミドなどが特に好ましい。 塩基は 0 . 1ないし 2 0当量、 好ましくは 0 . 5ないし 1 0当量、 さらに好ましくは 1ないし 5当量を用いる。 これらの反応は通 常溶媒中で有利に行われる。溶媒は反応に影響を与えないものであれば特定されな いが、 上記反応 2で示した溶媒などが用いられ、好ましくはアミド系溶媒、 エーテ ル系溶媒、 アルコール系溶媒、 二トリル系溶媒、 有機塩基系溶媒など、 あるいはこ れらの混合溶媒が挙げられ、 なかでも、 N, N—ジメチルホルムアミド、 テトラヒ ドロフラン、 ジォキサン、 ジメトキシェタン、 メタノール、 エタノール、 2 -プロパ ノール、 ter t-ブタノ一ル、 ァセトニトリル、 ピリジンなどが特に好ましい。 反応 温度は— 4 0 °Cないし 1 5 0 °C、 好ましくは 2 0 ないし 1 2 0 ° (:、 反応時間は 5 分間ないし 9 6時間、 好ましくは 3 0分間ないし 2 4時間である。 The amine represented by R 3 R 4 NH or R 12 R 13 NH or a salt thereof is 1 to 50 Equivalents, preferably 2 to 20 equivalents, more preferably 2 to 10 equivalents are used. Of these, liquid ones are also used as solvents. These amines are also advantageously used as salts with suitable acids. Examples of the acid include octaacetic acid (eg, trifluoroacetic acid), inorganic acid (eg, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, perchloric acid, etc.), and organic carboxylic acid (eg, formic acid, acetic acid) , Citric acid, tartaric acid, oxalic acid, etc.), organic sulfonic acids (eg, methanesulfonic acid, benzenesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc.) are used, among which hydrochloric acid, formic acid, acetic acid , Methanesulfonic acid, P-toluenesulfonic acid and the like are particularly preferred. The acid is usually used in an equimolar amount to the amine. Examples of the base include the bases shown in the above Reaction 2, and preferable examples include, for example, hydrogenated alkali metal, alkoxide of alkali metal, inorganic base, organic base, and alkali metal amide. Sodium hydride, sodium ethoxide, potassium tert-butoxide, sodium bicarbonate, potassium bicarbonate, sodium carbonate, potassium carbonate, triethylamine, diisopropylpyrethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-Pendecene, lithium diisopropylamide, lithium bistrimethylsilylamide and the like are particularly preferred. The base is used in an amount of 0.1 to 20 equivalents, preferably 0.5 to 10 equivalents, and more preferably 1 to 5 equivalents. These reactions are usually advantageously carried out in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above Reaction 2, etc. is used, and preferably an amide solvent, an ether solvent, an alcohol solvent, a nitrile solvent, Examples include organic base solvents and the like, or a mixed solvent thereof. Among them, N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol , Acetonitrile, pyridine and the like are particularly preferred. The reaction temperature is from −40 ° C. to 150 ° C., preferably from 20 ° to 120 ° C. (: the reaction time is from 5 minutes to 96 hours, preferably from 30 minutes to 24 hours.
一般式 ( I ) において R 1が一 S R 2、 X Y がIn the general formula (I), R 1 is one SR 2 , XY is
である化合物 (X) またはその塩の合成 Of compound (X) or its salt
(X I I I) (X) (X I I I) (X)
反 j¾、 9 - a ) Anti-j¾, 9-a)
一般式(X I I I )で表されるイミダゾチアジン化合物またはその塩を原料とし、 塩基の存在下に一般式 R 1 5— Z— L ( Zは S 02または C〇、 Lは脱離基を表す。) で表される化合物またはその塩と反応させることにより、 一般式 (X)で表される イミダゾチアジン化合物 (Zは S〇2または C Oを示す。 ) またはその塩を得る。 塩基としては、上記反応 2で示した塩基などが挙げられ、好ましい例としては例 えば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 アルキルリチウム、またはアル力リ金属ァミドなどが好ましく、水素化ナトリウム、 カリウム ter卜ブトキシド、 炭酸ナトリウム、 炭酸カリウム、 トリェチルァミン、 ジィソプロピルェチルァミン、 ピリジン、 4 -ジメチルァミノピリジン、 1, 8-ジァザ ビシクロ [5, 4, 0]- 7-ゥンデセン、 メチルリチウム、 ブチルリチウム、 リチウムジィ ソプロピルアミド、 リチウムピストリメチルシリルアミドなどが特に好ましい。 こ れらのうち液体のものは溶媒としても用いられる。塩基は 1ないし 5 0当量、好ま しくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。 一般式 R 1' 5— Z— Lで表される化合物において、 Lは塩素、 臭素、 ヨウ素などのハロゲン原 子、 あるいはシァノ基、 イミダゾール基などが好ましい。 上記一般式 R 1 5— Z— Lで表される化合物は原料に対して 0 . 2ないし 2 0当量、 好ましくは 1ないし 1 0当量、 さらに好ましくは 1ないし 5当量を用いる。 これらの反応は通常溶媒中で 有利に行われる。溶媒は反応に影響を与えないものであれば特定されないが、上記 反応 2で示した溶媒などが用いられ、 好ましくはアミド系溶媒、 エーテル系溶媒、 ケトン系溶媒、 エステル系溶媒、 二トリル系溶媒、 アルコール系溶媒、 炭化水素系 溶媒、 有機塩基系溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも N, N—ジメチルホルムアミド、 N-メチルピロリドン、 テトラヒドロフラン、 ジォキ サン、ジメトキシェタン、アセトン、 2-ブタノン、酢酸ェチル、酢酸 tert -プチル、 ァセトニトリル、 トルエン、 ピリジンなどが特に好ましい。 反応温度は一 8 0 °Cな いし 1 5 O :、好ましくは 0 ないし 1 0 0 °C、反応時間は 1分間ないし 1 2 0時 間、 好ましくは 5分ないし 4 8時間である。 Imidazothiazine compound represented by the general formula (XIII) or a salt thereof as a starting material, the general formula R 1 5 in the presence of a base - Z- L (Z is S 0 2 or C_〇, L is a leaving group represented. the compound or reacting it with a salt thereof), imidazothiazine compound represented by the general formula (X) (Z is obtained.) or a salt thereof shows a S_〇 2 or CO. Examples of the base include the bases shown in Reaction 2 above, and preferable examples include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides. Preferably, sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0]- 7-Pendecene, methyllithium, butyllithium, lithium disopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. In the compound represented by the general formula R 1 ′ 5 —Z—L, L is preferably a halogen atom such as chlorine, bromine or iodine, or a cyano group or an imidazole group. The compound represented by the above general formula R 15 —Z—L is used in an amount of 0.2 to 20 equivalents, preferably 1 to 1 equivalent to the raw material. 0 equivalents, more preferably 1 to 5 equivalents are used. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, a nitrile solvent. , Alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents and the like, and mixtures thereof. Among them, N, N-dimethylformamide, N-methylpyrrolidone, tetrahydrofuran, dioxane, dimethoxyethane, acetone, Particularly preferred are 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like. The reaction temperature is from 180 ° C. to 150 ° C., preferably from 0 to 100 ° C., and the reaction time is from 1 minute to 120 hours, preferably from 5 minutes to 48 hours.
(X I I I) (Χ' ) (X I I I) (Χ ')
反応 9一 (b ) Reaction 91 (b)
一般式(X I I I )で表されるイミダゾチアジン化合物またはその塩を原料とし、 塩基の存在下に一般式 R 1 6 R 1 7 P (〇) — L (Lは脱離基を表す。 ) で表される 化合物またはその塩と反応させることにより、 一般式 (X ') で表されるイミダゾ チアジン化合物またはその塩を得る。 Starting from the imidazothiazine compound represented by the general formula (XIII) or a salt thereof, in the presence of a base, represented by the general formula R 16 R 17 P (〇) —L (L represents a leaving group). By reacting with a compound represented by the formula or a salt thereof, an imidazothiazine compound represented by the general formula (X ') or a salt thereof is obtained.
塩基としては、 上記反応 9一 ( a ) で示した塩基などが挙げられ、 好ましい例と しては例えば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有 機塩基、 アルキルリチウム、 またはアルカリ金属アミドなどが好ましく、 水素化ナ トリウム、 カリウム tert-ブトキシド、 炭酸ナトリウム、 炭酸カリウム、 トリェチ ルァミン、 ジイソプロピルェチルァミン、 ピリジン、 4-ジメチルァミノピリジン、 1, 8 -ジァザピシクロ [5, 4, 0] - 7-ゥンデセン、 メチルリチウム、 プチルリチウム、 リ ド、リチウムピストリメチルシリルアミドなどが特に好 ましい。 これらのうち液体のものは溶媒としても用いられる。塩基は 1ないし 50 当量、 好ましくは 2ないし 10当量、 さらに好ましくは 2ないし 5当量を用いる。 一般式 R16R17P (〇) —Lで表される化合物において、 Lは塩素、 臭素、 ヨウ 素などのハロゲン原子などが好ましい。 上記一般式 R16R17P (〇) 一 Lで表さ れる化合物は原料に対して 0.2ないし 20当量、 好ましくは 1ないし 10当量、 さらに好ましくは 1ないし 5当量を用いる。これらの反応は通常溶媒中で有利に行 われる。溶媒は反応に影響を与えないものであれば特定されないが、上記反応 9— (a) で示した溶媒などが用いられ、 好ましくはアミド系溶媒、 エーテル系溶媒、 ケトン系溶媒、 エステル系溶媒、 二トリル系溶媒、 アルコール系溶媒、 炭化水素系 溶媒、 有機塩基系溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも N, N—ジメチルホルムアミド、 N -メチルピロリドン、 テ卜ラヒドロフラン、 ジォキ サン、ジメトキシェタン、アセトン、 2 -ブタノン、酢酸ェチル、酢酸 tert-プチル、 ァセトニトリル、 トルエン、 ピリジンなどが特に好ましい。反応温度は— 80 な いし 150 °C、好ましくは 0 ないし 100 °C、反応時間は 1分間ないし 120時 間、 好ましくは 5分ないし 48時間である。 Examples of the base include the bases shown in the above-mentioned Reaction 9-1 (a), and preferable examples include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkalis. Metal amides and the like are preferred, and sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazapicyclo 0]-7-Pendecene, methyllithium, butyllithium, And lithium pistrimethylsilylamide are particularly preferred. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. In the compound represented by the general formula R 16 R 17 P (〇) —L, L is preferably a halogen atom such as chlorine, bromine, and iodine. The compound represented by the above general formula R 16 R 17 P (〇) 1 L is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material. These reactions are advantageously carried out usually in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above reaction 9- (a) or the like is used, preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, Examples include nitrile-based solvents, alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents, and the like, or a mixed solvent thereof. Dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred. The reaction temperature is -80 to 150 ° C, preferably 0 to 100 ° C, and the reaction time is 1 minute to 120 hours, preferably 5 minutes to 48 hours.
(XIII) (XIII)
(X") (X ")
反応 9一 (C) Reaction 91 (C)
一般式(X I I I)で表されるイミダゾチアジン化合物またはその塩を原料とし、 一般式 R18_Z— NCO (Zは S〇2あるいは単結合を表す。 ) で表される'イソシ ァネート類またはその塩と反応させることにより、 一般式 (X" ) で表されるイミ ダゾチアジン化合物またはその塩を得る。 And imidazothiazine compound represented by the general formula (XIII) or a salt thereof as a starting material, the general formula R 18 _Z- NCO (Z represents S_〇 2 or a single bond.) Represented by the 'Isoshi Aneto acids or their By reacting with a salt, an imidazothiazine compound represented by the general formula (X ") or a salt thereof is obtained.
Zが単結合を示す場合、本反応は塩基の存在下に有利に行われる。塩基としては、 上記反応 9一 (a) で示した塩基などが挙げられ、 好ましい例としては例えば、 水 素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 アルキル リチウム、 またはアルカリ金属アミドなどが好ましく、 水素化ナトリウム、 力リウ ム tert-ブトキシド、 炭酸ナトリウム、 炭酸カリウム、 トリェチルァミン、 ジイソ プロピルェチルアミ.ン、 ピリジン、 4-ジメチルァミノピリジン、 1, 8-ジァザビシク D [5, 4, 0] -7-ゥンデセン、 メチルリチウム、 ブチルリチウム、 リチウムジイソプロ ピルアミド、 リチウムピストリメチルシリルアミドなどが特に好ましい。 これらの うち液体のものは溶媒としても用いられる。塩基は 1ないし 50当量、好ましくは 2ないし 10当量、 さらに好ましくは 2ないし 5当量を用いる。 一般式 R18— Z 一 NCOで表されるイソシァネート類は原料に対して 0.2ないし 20当量、 好ま しくは 1ないし 10当量、 さらに好ましくは 1ないし 5当量を用いる。 これらの反 応は通常溶媒中で有利に行われる。溶媒は反応に影響を与えないものであれば特定 されないが、 上記反応 9一 (a) で示した溶媒などが用いられ、 好ましくはアミド 系溶媒、 エーテル系溶媒、 ケトン系溶媒、 エステル系溶媒、 二トリル系溶媒、 アル コール系溶媒、 炭化水素系溶媒、 有機塩基系溶媒など、 あるいはこれらの混合溶媒 が挙げられ、 なかでも N, N—ジメチルホルムアミド、 N-メチルピロリドン、 テ トラヒドロフラン、 ジォキサン、 ジメトキシェタン、 アセトン、 2-ブ夕ノン、 酢酸 ェチル、 酢酸 tert -プチル、 ァセトニトリル、 トルエン、 ピリジンなどが特に好ま しい。反応温度は一 80°Cないし 150°C、好ましくは 0°Cないし 100°C、 反応 時間は 1分間ないし 120時間、 好ましくは 5分ないし 48時間である。 When Z represents a single bond, the reaction is advantageously performed in the presence of a base. As the base, The base shown in the above reaction 9-1 (a) and the like are preferable. Preferred examples include, for example, alkali metal hydride, alkali metal alkoxide, inorganic base, organic base, alkyl lithium, and alkali metal amide. Sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabisic D [5, 4, 0]- 7-Pendecene, methyllithium, butyllithium, lithium diisopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. The isocyanate represented by the general formula R 18 —Z-NCO is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material. These reactions are usually and advantageously effected in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above reaction 9-1 (a) is used, and preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, Examples include nitrile solvents, alcohol solvents, hydrocarbon solvents, organic base solvents, and mixtures thereof.N, N-dimethylformamide, N-methylpyrrolidone, tetrahydrofuran, dioxane , Dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred. The reaction temperature ranges from 80 ° C to 150 ° C, preferably from 0 ° C to 100 ° C, and the reaction time ranges from 1 minute to 120 hours, preferably from 5 minutes to 48 hours.
(XIII) (X) (XIII) (X)
反応 9— (d) Reaction 9— (d)
一般式(X I I I)で表されるイミダゾチアジン化合物またはその塩を原料とし、 縮合剤と塩基の存在下に一般式 R 1 5— C O 2 Hで表される化合物またはその塩と 反応させることにより、一般式 (X) で表されるイミダゾチアジン化合物またはそ の塩を得る。 Starting from an imidazothiazine compound represented by the general formula (XIII) or a salt thereof, Reaction with a compound represented by the general formula R 15 —CO 2 H or a salt thereof in the presence of a condensing agent and a base gives an imidazothiazine compound represented by the general formula (X) or a salt thereof .
縮合剤としては、 N, N' -カルボ二ルジィミダゾール、 ベンゾトリアゾリル- N-ヒド ン化物塩、 N,N,-ジシ トキシカルポニル _2 - エトキシ -1, 2-ジヒドロキノリン、 塩酸 1-ェチル -3- (3-ジメチルァミノプロピル) カルポジイミドなどが挙げられ、 好ましい例としては例えば、 N,N ジシクロへキ シルカルポジイミド、 シァノりん酸ジェチル、 塩酸 卜ェチル -3- (3-ジメチルアミ ノプロピル)カルポジイミドなどが特に好ましい。 縮合剤は 1ないし 5 0当量、 好 ましくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。塩基とし ては、 上記反応 9一 ( a ) で示した塩基などが挙げられ、 好ましい例としては例え ば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 7 ルキルリチウム、 またはアルカリ金属アミドなどが好ましく、 水素化ナトリウム、 カリウム ter卜ブトキシド、 炭酸ナトリウム、 炭酸カリウム、 トリェチルァミン、 ジイソプロピルェチルァミン、 ピリジン、 4-ジメチルァミノピリジン、 1, 8-ジァザ ビシクロ [5, 4, 0]- 7-ゥンデセン、 メチルリチウム、 ブチルリチウム、 リチウムジィ ソプロピルアミド、 リチウムピストリメチルシリルアミドなどが特に好ましい。 こ れらのうち液体のものは溶媒としても用いられる。塩基は 1ないし 5 0当量、好ま しくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。 一般式 R 1 5— C〇2Hで表される化合物は原料に対して 0 . 2ないし 2 0当量、 好ましくは 1 ないし 1 0当量、 さらに好ましくは 1ないし 5当量を用いる。 これらの反応は通常 溶媒中で有利に行われる。溶媒は反応に影響を与えないものであれば特定されない が、 上記反応 9一 ( a ) で示した溶媒などが用いられ、 好ましくはアミド系溶媒、 ェ一テル系溶媒、 ケトン系溶媒、 エステル系溶媒、 二トリル系溶媒、 アルコール系 溶媒、 炭化水素系溶媒、有機塩基系溶媒など、 あるいはこれらの混合溶媒が挙げら れ、 なかでも N, N—ジメチルホルムアミド、 N-メチルピロリドン、 テトラヒド 口フラン、 ジォキサン、 ジメトキシェタン、 アセトン、 2-ブ夕ノン、 酢酸ェチル、 酢酸 ter t-プチル、 ァセトニトリル、 トルエン、 ピリジンなどが特に好ましい。 反 応温度は— 80 °Cないし 150 °C、好ましくは 0 ないし 100 °C、反応時間は 1 分間ないし 120時間、 好ましくは 5分ないし 48時間である。 Examples of the condensing agent include N, N'-carbonyldiimidazole, benzotriazolyl-N-hydranide salt, N, N, -disocyanate. Toxicarbonyl_2-ethoxy-1,2-dihydroquinoline, 1-ethyl-3- (3-dimethylaminopropyl) carbodiimide hydrochloride, and the like. Preferred examples thereof include, for example, N, N dicyclohexylcarboimide, Particularly preferred are getyl cyanophosphate, and triethyl hydrochloride-3- (3-dimethylaminopropyl) carbodiimide. The condensing agent is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. Examples of the base include the bases shown in the above reaction 9-1 (a). Preferred examples include alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, 7-alkyllithiums, and alkalis. Metal amides and the like are preferable, and sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0 ] -7-Pendecene, methyllithium, butyllithium, lithium disopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. Formula R 1 5 -. C_〇 compound represented by the 2 H 0 2 to 2 0 equivalents relative to the starting, preferably 1 to 1 0 equivalents, more preferably 1 to use 5 equivalents. These reactions are advantageously carried out usually in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above reaction 9-1 (a) is used, and preferably an amide solvent, an ether solvent, a ketone solvent, or an ester solvent Solvents, nitrile-based solvents, alcohol-based solvents, hydrocarbon-based solvents, organic base-based solvents and the like, or a mixture thereof, among which N, N-dimethylformamide, N-methylpyrrolidone, tetrahydrofuran, Dioxane, dimethoxyethane, acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitrile, toluene, pyridine and the like are particularly preferred. Anti The reaction temperature is -80 ° C to 150 ° C, preferably 0 to 100 ° C, and the reaction time is 1 minute to 120 hours, preferably 5 minutes to 48 hours.
一般式 (I) において In general formula (I)
X Y がX Y
である化合物 (XVI) またはその塩および Compound (XVI) or a salt thereof and
X Y がX Y
である化合物 (XVI I) またはその塩の合成 Of compound (XVI I) or its salt
(XVI) (XVII) 反応 10 (XVI) (XVII) Reaction 10
一般式 (X IV) で表されるイミダゾチアジン化合物 (Zは S〇2を示す。 ) ま たはその塩を原料とし、 式中の水酸基を脱離基 L,への変換反応に付すことにより、 一般式 (XV) で表されるイミダゾチアジン化合物 (Zは S〇2を示す。 ) または その塩を得る。 ' 上記の脱離基 L'への変換反応には、 例えば、 ハロゲン化剤によるハロゲン化反 応、 スルホン酸エステル化反応などが挙げられる。ハロゲン化剤としては、例えば ハロゲン化水素 (例、 ヨウ化水素、 臭化水素、 塩化水素など) またはハロゲン化剤 (例、 三塩化リン、 五塩化リン、 ォキシ塩化リン、 三臭化リン、 五臭化リンなどの ハロゲン化リン、 トリフエニルホスフィン一四塩化炭素、 トリフエニルホスフィン 一臭素、 トリフエニルホスフィン一 N-ブロモコハク酸イミド、 トリフエニルホス フアイトーヨウ化メチルなどの有機リン化合物、 または塩化チォニル、塩化ォキザ リル、 ホスゲンなど) どが挙げられる。 ハロゲン化剤は 1ないし 1 0当量、好ま しくは 1ないし 3当量を用いる。これらのうち液体のものは溶媒としても用いられ る。 これらのハロゲン化反応は溶媒中で有利に行われる。 溶媒としては、 水、 有機 酸系溶媒 (例、 ギ酸、 酢酸など) 、 ケトン系溶媒 (例、 アセトン、 メチルェチルケ トン、 メチルイソプチルケトンなど) など、 アミド系溶媒 (例、 N, N—ジメチル ホルムアミド、 ジメチルァセトアミドなど) 、 ハロゲン化炭化水素系溶媒 (例、 ジ クロロメタン、 クロ口ホルム、 四塩化炭素、 1, 2-ジクロロェタン、 クロ口ベンゼン など) 、 アルコール系溶媒 (例、 ter t-ブタノールなど) 、 二トリル系溶媒 (例、 ァセトニトリルなど)、エステル系溶媒(例、ギ酸ェチル、酢酸ェチル、酢酸 tert- ブチルなど) 、 エーテル系溶媒 (例、 テトラヒドロフラン、 ジェチルエーテル、 ジ ォキサン、 ジメトキシェタンなど) 、 炭化水素系溶媒 (例、 へキサン、 ベンゼン、 トルエンなど) 、 芳香族有機塩基系溶媒(例、 ピリジン、 2, 4, 6-トリメチルピリジ ン、 ピコリン、 2, 6 -ルチジン、 1, 8-ジァザビシク口 [5, 4, 0] -7-ゥンデセンなど) な ど、 あるいはこれらの混合溶媒が挙げられる。 なかでも、 水、 アセトン、 N, N - ジメチルホルムアミド、 ジメチルァセトアミド、 ジクロロメタン、 クロ口ホルム、 四塩化炭素、 1, 2 -ジクロロェタン、 ジォキサン、 ピリジンなどが好ましい。 これら の反応には適当な無機酸 (例、 硫酸、 リン酸など) の存在下に行われてもよい。 反 応温度は— 2 0 ないし 1 5 0 、好ましくは ないし 1 0 0 °C、反応時間は 2 分間ないし 4 8時間、 好ましくは 5分間ないし 1 0時間である。 Formula imidazothiazine compound represented by (X IV) (Z represents a S_〇 2.) Or a salt thereof as a raw material, subjecting the hydroxyl group in the formula a conversion reaction of the leaving group L, to Accordingly, imidazothiazine compound represented by the general formula (XV) (Z represents a S_〇 2.) or a salt thereof. ' Examples of the conversion reaction to the leaving group L ′ include a halogenation reaction with a halogenating agent and a sulfonic acid esterification reaction. Examples of the halogenating agent include hydrogen halide (eg, hydrogen iodide, hydrogen bromide, hydrogen chloride, etc.) or halogenating agent (eg, phosphorus trichloride, phosphorus pentachloride, phosphorus oxychloride, phosphorus tribromide, Organophosphorus compounds such as phosphorus halides such as phosphorus bromide, triphenylphosphine monocarbon tetrachloride, triphenylphosphine monobromine, triphenylphosphine mono-N-bromosuccinimide, triphenylphosphite methyl iodide, or thionyl chloride, oxalyl chloride , Phosgene, etc.). The halogenating agent is used in an amount of 1 to 10 equivalents, preferably 1 to 3 equivalents. Of these, liquid ones are also used as solvents. These halogenation reactions are advantageously performed in a solvent. Solvents include water, organic acid solvents (eg, formic acid, acetic acid, etc.), ketone solvents (eg, acetone, methylethyl ketone, methyl isobutyl ketone, etc.), amide solvents (eg, N, N-dimethylformamide) , Dimethylacetamide, etc.), halogenated hydrocarbon solvents (eg, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, cyclobenzene), alcohol solvents (eg, tert- Butanol), nitrile solvents (eg, acetonitrile), ester solvents (eg, ethyl formate, ethyl acetate, tert-butyl acetate, etc.), ether solvents (eg, tetrahydrofuran, getyl ether, dioxane, dimethoxy) ), Hydrocarbon solvents (eg, hexane, benzene, toluene, etc.), aromatic organic bases Medium (eg, pyridine, 2,4,6-trimethylpyridin, picoline, 2,6-lutidine, 1,8-diazabi- sic [5,4,0] -7-indene), and the like A mixed solvent is mentioned. Of these, water, acetone, N, N-dimethylformamide, dimethylacetamide, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, dioxane, pyridine and the like are preferred. These reactions may be performed in the presence of a suitable inorganic acid (eg, sulfuric acid, phosphoric acid, etc.). The reaction temperature is −20 to 150, preferably to 100 ° C., and the reaction time is 2 minutes to 48 hours, preferably 5 minutes to 10 hours.
スルホン酸エステル化反応は原料の水酸基をハロゲン化スルホニル(例、塩化メ タンスルホニル、 塩化ベンゼンスルホニル、 塩化 P -トルエンスルホニル、 塩化卜 リフルォロメタンスルホニルなど) またはスルホン酸無水物 (例、 メタンスルホン 酸無水物、 ベンゼンスルホン酸無水物、 P -トルエンスルホン酸無水物、 トリフル ォロメタンスルホン酸無水物など)等と適当な塩基の存在下に反応させることによ り行われ、対応するスルホニルォキシ基へ変換する反応である。ハロゲン化スルホ ニルまたはスルホン酸無水物は原料に対して 0 . 2ないし 2 0当量、 好ましくは 1 ないし 1 0当量、 さらに好ましくは 1ないし 5当量を用いる。塩基としては、 上記 反応 2で示した塩基などが挙げられ、好ましい例としては例えば、水素化アルカリ 金属、 無機塩基、 有機塩基、 またはアルカリ金属アミドなどが好ましく、 水素化ナ トリウム、 炭酸ナトリウム、 炭酸カリウム、 トリェチルァミン、 ジイソプロピルェ チルアミン、ピリジン、 4-ジメチルァミノピリジン、 1, 8 -ジァザビシク口 [5, 4, 0] -7- ゥンデセン、 リチウムジイソプロピルアミド、 リチウムピストリメチルシリルアミ ドなどが特に好ましい。 これらのうち液体のものは溶媒としても用いられる。塩基 は 1ないし 2 0当量、好ましくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5 当量を用いる。スルホン酸エステル化反応で生成したスルホ二ルォキシ基は適当な 無機ハロゲン化物 (例、 ヨウ化ナトリウム、 ヨウ化カリウム、 臭化ナトリウム、 臭 化カリウムなど)と反応させることによりハロゲン基へ変換することが可能である。 無機ハロゲン化物は原料に対して 1ないし 5 0当量、好ましくは 1ないし 2 0当量、 さらに好ましくは 1ないし 1 0当量を用いる。これらの反応は通常反応に影響を与 えない溶媒中で行われる。溶媒としては、上記ハロゲン化反応に用いられる溶媒の うち水を除いたものなどが挙げられ、中でも、アセトン、メチルェチルケトン、 N, N—ジメチルホルムアミド、 ジメチルァセトアミド、 ジクロロメタン、 クロ口ホル ム、 1, 2-ジクロロエタン、 ァセトニトリル、 酢酸ェチル、 酢酸 ter t -プチル、 テト ラヒドロフラン、 ジェチルエーテレ、 ジォキサン、 ジメトキシェタン、 へキサン、 トルエン、 ピリジンなど、 あるいはこれらの混合溶媒が好ましい。 これらは無水溶 媒であることが特に好ましい。反応温度は一 2 0 ないし 2 0 0 、好ましくは 2 0 °Cないし 1 2 O t:、反応時間は 5分間ないし 1 2 0時間、好ましくは 2 0分間な いし 7 2時間である。 In the sulfonic acid esterification reaction, the hydroxyl group of the raw material is converted to a sulfonyl halide (eg, methanesulfonyl chloride, benzenesulfonyl chloride, P-toluenesulfonyl chloride, trifluoromethanesulfonyl chloride, etc.) or a sulfonic anhydride (eg, methanesulfone Acid anhydride, benzenesulfonic anhydride, P-toluenesulfonic anhydride, trifluoromethanesulfonic anhydride, etc.) in the presence of a suitable base. This is a reaction for converting to a group. The sulfonyl halide or sulfonic anhydride is used in an amount of 0.2 to 20 equivalents, preferably 1 to 10 equivalents, and more preferably 1 to 5 equivalents, based on the starting material. Examples of the base include the bases shown in the above Reaction 2, and preferable examples thereof include, for example, an alkali metal hydride, an inorganic base, an organic base, and an alkali metal amide. Particularly preferred are potassium, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-indene, lithium diisopropylamide, lithium pistrimethylsilylamide and the like. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. The sulfonyloxy group formed in the sulfonic acid esterification reaction can be converted to a halogen group by reacting with an appropriate inorganic halide (eg, sodium iodide, potassium iodide, sodium bromide, potassium bromide, etc.). It is possible. The inorganic halide is used in an amount of 1 to 50 equivalents, preferably 1 to 20 equivalents, and more preferably 1 to 10 equivalents, based on the raw material. These reactions are usually performed in a solvent that does not affect the reaction. Examples of the solvent include those used in the above-mentioned halogenation reaction except for water, and among them, acetone, methylethylketone, N, N-dimethylformamide, dimethylacetamide, dichloromethane, and chloroform And 1,2-dichloroethane, acetonitrile, ethyl acetate, tert-butyl acetate, tetrahydrofuran, getyl ethere, dioxane, dimethoxyethane, hexane, toluene, pyridine, and the like, or a mixed solvent thereof. These are particularly preferably water-free solvents. The reaction temperature is from 120 to 200, preferably from 20 ° C. to 12 Ot: and the reaction time is from 5 minutes to 120 hours, preferably from 20 minutes to 72 hours.
反応 1 1 Reaction 1 1
一般式 (XV) で表されるイミダゾチアジン化合物 (Zは s o 2を示す。 ) また はその塩を原料とし、 塩基または酸と反応させることにより、 一般式 (XV I ) ま たは (X V I I ) で表されるイミダゾチアジン化合物 (Zは s〇2を示す。 ) また はその塩を得る。 The imidazothiazine compound represented by the general formula (XV) (Z represents so 2 ) or a salt thereof is reacted with a base or an acid to obtain the compound represented by the general formula (XVI). Others imidazothiazine compound represented by (XVII) (Z represents a S_〇 2.) Or a salt thereof.
塩基としては、上記反応 2で示した塩基などが挙げられ、好ましい例としては例 えば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 またはアルカリ金属アミドなどが好ましく、 水素化ナトリウム、 カリウム tert - ブトキシド、 炭酸ナトリウム、 炭酸カリウム、 ジイソプロピルェチルァミン、 4- ジメチルァミノピリジン、 1, 8-ジァザビシクロ [5, 4, 0] - 7 -ゥンデセン、 リチウムジ イソプロピルアミド、 リチウムピストリメチルシリルアミドなどが特に好ましい。 塩基は 1ないし 2 0当量、好ましくは 2ないし 1 0当量、 さらに好ましくは 2ない し 5当量を用いる。酸としては上記反応 5で示した酸などが挙げられ、 中でも例え ば、 トリフルォロ酢酸、 塩化水素酸、 臭化水素酸、 硫酸、 ギ酸、 酢酸、 メタンスル ホン酸、 P-トルエンスルホン酸、 カンファースルホン酸、 3フッ化ホウ素エーテ ル錯体などが特に好ましい。 これらのうち液体のものは溶媒としても用いられる。 酸は原料に対して 0 . 0 5ないし 1 0当量、好ましくは 0 . 1ないし 5当量、 さらに 好ましくは 0 . 2ないし 2当量を用いる。 これらの反応は通常溶媒中で有利に行わ れる。溶媒は反応に影響を与えないものであれば特定されないが、上記反応 2ある いは反応 5で示した溶媒などが用いられ、好ましくは有機酸系溶媒、アミド系溶媒、 エーテル系溶媒、 アルコール系溶媒、 二トリル系溶媒、 炭化水素系溶媒、 ハロゲン 化炭化水素系溶媒、 有機塩基系溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも、 ギ酸、 酢酸、 N, N—ジメチルホルムアミド、 テトラヒドロフラン、 ジ ォキサン、 ジメ卜キシェタン、 ァセトニトリル、 ベンゼン、 トルエン、 キシレン、 ジクロロメタン、 クロ口ホルム、 1, 2-ジクロロェタン、 クロ口ベンゼン、 ピリジン などが特に好ましい。 反応温度は — 4 0 °Cないし 2 0 0 °C、 好ましくは 0 ない し 1 2 O 、反応時間は 5分間ないし 1 2 0時間、好ましくは 3 0分間ないし 4 8 時間である。 Examples of the base include the bases shown in Reaction 2 above, and preferable examples thereof include, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, and alkali metal amides. , Potassium tert-butoxide, sodium carbonate, potassium carbonate, diisopropylethylamine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-indene, lithium diisopropylamide, lithium pistrimethylsilylamide Etc. are particularly preferred. The base is used in an amount of 1 to 20 equivalents, preferably 2 to 10 equivalents, more preferably 2 to 5 equivalents. Examples of the acid include the acids shown in the above Reaction 5, among which, for example, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, formic acid, acetic acid, methanesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid And boron trifluoride etherate complex are particularly preferred. Of these, liquid ones are also used as solvents. The acid is used in an amount of 0.05 to 10 equivalents, preferably 0.1 to 5 equivalents, and more preferably 0.2 to 2 equivalents, based on the raw material. These reactions are advantageously carried out usually in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above Reaction 2 or Reaction 5 is used, and preferably, an organic acid solvent, an amide solvent, an ether solvent, or an alcohol solvent Solvents, nitrile-based solvents, hydrocarbon-based solvents, halogenated hydrocarbon-based solvents, organic base-based solvents, and the like, or a mixture thereof. Among them, formic acid, acetic acid, N, N-dimethylformamide, tetrahydrofuran, Dioxane, dimethyloxetane, acetonitrile, benzene, toluene, xylene, dichloromethane, chloroform, 1,2-dichloroethane, cyclobenzene, pyridine and the like are particularly preferred. The reaction temperature is —40 ° C. to 200 ° C., preferably 0 to 12 O, and the reaction time is 5 minutes to 120 hours, preferably 30 minutes to 48 hours.
一般式 ( I ) において R 1が— S R 2、 15 In the general formula (I), R 1 is — SR 2 , Fifteen
X Y が X Y
が But
である化合物 (X) またはその塩および R1がー SR2、 A compound (X) or a salt thereof, and R 1 is -SR 2 ,
15 Fifteen
X Y が »X Y »
である化合物 (XX I) またはその塩の合成 Of compound (XX I) or its salt
塩基 Base
反応 1 3 Reaction 1 3
(I V) (XV I I I) (I V) (XV I I I)
(X) (XX I) (X) (XX I)
反応 12 Reaction 12
前記記載の反応 7— (a) と同様の条件により、 一般式 (I V) で表されるイミ ダゾリジン化合物 (Zは S 02、 Rは前記アルキル基を示す。 ) またはその塩を原 料とし、 塩基の存在下に上記反応 7— ( a ) で示した一般式 R 2— L (Lは脱離基 を表す。 ) またはその塩と反応させることにより、 一般式 (XV I I I ) で表され るイミダゾリジン化合物 (Zは S〇2、 Rは前記アルキル基を示す。 ) またはその 塩を得る。 Under the same conditions as in Reaction 7- (a) described above, the imine represented by the general formula (IV) Dazorijin compounds (Z is S 0 2, R indicates the alkyl group.) Or a salt thereof as raw materials, the general formula R 2 shown in the presence of a base in the above reaction 7- (a) - L (L is by reacting with.) or a salt thereof represents a leaving group, the general formula (XV III) imidazolidine represented Ru compounds (Z is S_〇 2, R is obtained.) or a salt thereof shows the alkyl group .
反応 1 3 Reaction 1 3
前記記載の反応 3と同様の条件により、 一般式(XV I I I ) で表されるイミダ ゾリジン化合物 (Zは S〇2、 Rは前記アルキル基を示す。 ) またはその塩を原料 とし、 塩基と反応させることにより、 一般式 (X I X) で表されるイミダゾリジン 化合物 (Zは S 02、 Rは前記アルキル基を示す。 ) またはその塩を得る。 Under the same conditions as described in Reaction 3 of the description, Imida Zorijin compound represented by the general formula (XV III) (Z is S_〇 2, R represents the alkyl group.) Or a salt thereof as a starting material, the base and the reaction by, imidazolidine compound represented by the general formula (XIX) (Z is S 0 2, R represents the alkyl group.) is obtained or a salt thereof.
反応 1 4 . Reaction 1 4.
前記記載の反応 4と同様の条件により、 一般式(X I X) で表されるイミダゾリ ジン化合物またはその塩を原料とし、 上記一般式(V) で表される α位に脱離基 L を有するケトン化合物またはその塩と塩基の存在下に反応させることにより、一般 式 (X X) で表されるイミダゾチアジン化合物またはその塩を得る。 Under the same conditions as in Reaction 4 described above, an imidazolysine compound represented by the general formula (XIX) or a salt thereof is used as a starting material, and a ketone having a leaving group L at the α-position represented by the above general formula (V) By reacting the compound or a salt thereof in the presence of a base, an imidazothiazine compound represented by the general formula (XX) or a salt thereof is obtained.
反応 1 5 Reaction 1 5
一般式 (X X) で表されるイミダゾチアジン化合物 ( Ζは s o 2を示す。 ) また はその塩を原料とし、 酸と反応させることにより、 一般式 (X) または (XX I ) で表されるイミダゾチアジン化合物 (Zは s〇2を示す。 ) またはその塩を得る。 酸としては上記反応 5で示した酸などが挙げられ、 中でも例えば、 トリフルォロ 酢酸、 塩化水素酸、 臭化水素酸、 硫酸、 ギ酸、 酢酸、 メタンスルホン酸、 p -トル エンスルホン酸、カンファースルホン酸、 3フッ化ホウ素エーテル錯体などが特に 好ましい。 これらのうち液体のものは溶媒としても用いられる。酸は原料に対して 0 . 0 5力、ら 1 0当量、 好ましくは 0 . 1から 5当量、 さらに好ましくは 0 . 2から 2当量を用いる。 これらの反応は通常溶媒中で有利に行われる。溶媒は反応に影響 を与えないものであれば特定されないが、.上記反応 5で示した溶媒などが用いられ、 好ましくは有機酸系溶媒、 アミド系溶媒、 エーテル系溶媒、 アルコール系溶媒、 二 トリル系溶媒、炭化水素系溶媒、ハ口ゲン化炭化水素系溶媒、有機塩基系溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも、 ギ酸、 酢酸、 N, N—ジメチル ホルムアミド、 テトラヒドロフラン、.ジォキサン、 ジメトキシェタン、 ァセトニト リル、 ベンゼン、 トルエン、 キシレン、 ジクロロメタン、 クロ口ホルム、 1,2-ジク ロロェタン、 クロ口ベンゼン、 ピリジンなどが特に好ましい。 反応温度は一 4 O : ないし 200°C、好ましくは 0でないし 120°C、反応時間は 5分間ないし 120 時間、 好ましくは 30分間ないし 48時間である。 The imidazothiazine compound represented by the general formula (XX) (Ζ represents so 2 ) or a salt thereof is reacted with an acid to form the compound represented by the general formula (X) or (XXI). that imidazothiazine compound (Z denotes a S_〇 2.) or a salt thereof. Examples of the acid include the acids shown in Reaction 5 above, among which, for example, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, formic acid, acetic acid, methanesulfonic acid, p-toluenesulfonic acid, camphorsulfonic acid And boron trifluoride etherate are particularly preferred. Of these, liquid ones are also used as solvents. The acid is used in an amount of 0.05 equivalents, preferably 10 equivalents, preferably 0.1 to 5 equivalents, more preferably 0.2 to 2 equivalents, based on the raw material. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvents described in Reaction 5 above are used, and preferably, organic acid solvents, amide solvents, ether solvents, alcohol solvents, nitriles Solvents, hydrocarbon solvents, haptic hydrocarbon solvents, organic base solvents, etc., and mixtures thereof. Among them, formic acid, acetic acid, N, N-dimethyl Formamide, tetrahydrofuran, dioxane, dimethoxyethane, acetonitrile, benzene, toluene, xylene, dichloromethane, chloroform, 1,2-dichloroethane, cyclobenzene, pyridine and the like are particularly preferred. The reaction temperature is between 14O: to 200 ° C, preferably not 0 to 120 ° C, and the reaction time is 5 minutes to 120 hours, preferably 30 minutes to 48 hours.
一般式 (I) において R が _SR2、 In the general formula (I), R is _SR 2 ,
X Y が 0X Y is 0
である化合物 (XX I V) またはその塩の合成 Of compound (XXIV) or its salt
(XX I I ) (XX I V) (XX I I) (XX I V)
反応 16 Reaction 16
前記記載の反応 4と同様の条件により、一般式 (XX I I) で表されるイミダゾ リジン化合物 (Zは S〇2を示す。 ) またはその塩を原料とし、 一般式 (XX I I I) で表される 位に脱離基 Lを有するカルボン酸エステル化合物 (Rはメチル、 ェチル、 プロピル、 イソプロピル、 2-ェチルへキシルなどのアルキル基を表す。 ) またはその塩と塩基の存在下に反応させることにより、 一般式 (XX I V)で表さ れるイミダゾチアジン化合物 (Zは so2を示す。 ) またはその塩を得る。 Under the same conditions as described in Reaction 4 of the described general formula (XX II) imidazolidine compound represented by (Z is S_〇 2 are shown.) Or a salt thereof as a raw material, represented by the general formula (XX III) (R represents an alkyl group such as methyl, ethyl, propyl, isopropyl, 2-ethylhexyl) or a salt thereof in the presence of a base. To obtain an imidazothiazine compound represented by the general formula (XX IV) (Z represents so 2 ) or a salt thereof.
一般式 (I) において R1が水素原子、 置換されていてもよい炭化水素基または 置換されていてもよい複素環基であり、 In the general formula (I), R 1 is a hydrogen atom, an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group,
である化合物 (XV I ) またはその塩の合成 Of compound (XVI) or its salt
(X) (X V I ) 反応 1 7 (X) (XVI) reaction 1 7
一般式 (X) で表されるイミダゾチアジン化合物 (Zは s o 2を示す。 ) または その塩を原料とし、有機金属試薬またはその等価体あるいは金属水素化物と反応さ せることにより、 一般式 (X V I ) で表されるイミダゾチアジン化合物 (R 1 'は 置換されていてもよい炭化水素または水素原子を、 Zは s o 2を示す。 ) またはそ の塩を得る。 By reacting an imidazothiazine compound represented by the general formula (X) (Z represents so 2 ) or a salt thereof with an organometallic reagent or an equivalent thereof or a metal hydride, the general formula (X XVI) (wherein R 1 ′ represents an optionally substituted hydrocarbon or hydrogen atom, and Z represents so 2 ) or a salt thereof.
上記有機金属試藥またはその等価体としては例えば、 アルキルリチウム (例、 メ チルリチウム、 ェチルリチウム、 n-ブチルリチウム、 sec-ブチルリチウム、 ter t- ブチルリチウムなど) 、 アルキルマグネシウムハラィド (例、 メチルマグネシウム プロミド、 ェチルマグネシゥムブロミド、 n-ブチルマグネシウムブロミド、 n -ペン チルマグネシウムブロミド、 n-へキシルマグネシウムブロミドなど) 、 アルキル亜 鉛ハライド (例、 メチル亜鉛プロミド、 ジェチル亜鉛プロミドなど) 、 アルキル亜 鉛 (例、 ジメチル亜鉛、 ジ工チル亜鉛など) 、 アルキルアルミニウム (例、 トリメ チルアルミニウムなど)などが用いられる。あるいは任意のハロゲン化物を原料に 自体公知の方法により有機リチウム、有機マグネシウムハラィドまたはアルキル亜 鉛ハライドへと変換して本反応に用いることができる。金属水素化物としては例え ば、 水素化リチウムアルミニウム、 水素化ホウ素ナトリウム、 水素化ホウ素リチウ ム、水素化ジイソブチルアルミニウムなどが挙げられる。有機金属試薬またはその 等価体あるいは金属水素化物は 0 . 5ないし 2 0当量、 好ましくは 1ないし 1 0当 量を用いる。これらの反応は適当な有機リン化合物等の存在下に有利に行うことが できる。 有機リン化合物としては例えば、 トリァリ一ルホスフィン (例、 トリフエ ニルホスフィン、 トリ- 0 -トリルホスフィンなど)、 トリアルキルホスフィン(例、 トリメチルホスフィン、 トリブチルホスフィンなど) 、 あるいはトリアルキルホス ファイト (例、 トリメチルホスファイト、 トリェチルホスファイトなど) などが举 げられ、なかでもトリフエニルホスフィン、トリブチルホスフィンなどが好ましい。 有機リン化合物等は原料に対して 1ないし 5 0当量、好ましくは 1ないし 1 0当量 を用いる。またこれらの反応は適当な無機塩(例、ヨウ化リチウム、臭ィ匕リチウム、 塩化リチウム、塩化セリウムなど) 等の存在下に行うことができる。 無機塩は原料 に対して 1ないし 5 0当量、 好ましくは 1ないし 1 0当量を用いる。 これらの 反応は通常反応に影響を与えない溶媒中で行われる。 溶媒としてはアミド系溶媒 (例、 N, N—ジメチルホルムアミド、 N, N—ジメチルァセトアミドなど) 、 ェ 一テル系溶媒 (例、 テ卜ラヒドロフラン、 ジェチルエーテル、 ジォキサン、 ジメト キシェタンなど) 、 炭化水素系溶媒 (例、 へキサン、 ベンゼン、 トルエンなど) 、 有機塩基系溶媒(例、 ピリジン、 2, 4, 6 -トリメチルピリジン、 ピコリン、 2, 6-ルチ ジン、 1, 8-ジァザビシクロ [5, 4, 0] - 7-ゥンデセンなどの芳香族塩基など) など、 あ るいはこれらの混合溶媒が挙げられる。 なかでも、 テトラヒドロフラン、 ジェチル エーテル、 ジメトキシェタン、 N, N—ジメチルホルムアミド、 N, N—ジメチル ァセトアミドなどが好ましい。 これらは無水溶媒であることが特に好ましい。反応 の種類によっても異なるが、反応温度は一 1 0 0 ないし 1 5 0 、好ましくは一 4 0 °Cないし 1 2 0 、反応時間は 2分間ないし 1 2 0時間、好ましくは 1 0分間 ないし 4 8時間である。 Examples of the organometallic reagents or their equivalents include alkyl lithium (eg, methyl lithium, ethyl lithium, n-butyl lithium, sec-butyl lithium, tert-butyl lithium, etc.), alkyl magnesium hydride (eg, Methylmagnesium bromide, ethylmagnesium bromide, n-butylmagnesium bromide, n-pentylmagnesium bromide, n-hexylmagnesium bromide, etc., alkyl zinc halides (eg, methyl zinc bromide, getyl zinc bromide, etc.) Alkyl zinc (eg, dimethyl zinc, diethyl zinc, etc.), alkyl aluminum (eg, trimethyl aluminum, etc.) are used. Alternatively, an arbitrary halide can be converted into an organolithium, an organomagnesium halide or an alkyl zinc halide by a method known per se as a raw material and used in this reaction. Examples of the metal hydride include lithium aluminum hydride, sodium borohydride, lithium borohydride, diisobutylaluminum hydride, and the like. The organic metal reagent or its equivalent or metal hydride is used in an amount of 0.5 to 20 equivalents, preferably 1 to 10 equivalents. These reactions can be advantageously performed in the presence of an appropriate organic phosphorus compound or the like. Examples of the organic phosphorus compound include triarylphosphine (eg, triffee). Nylphosphine, tri-0-tolylphosphine, etc.), trialkylphosphine (eg, trimethylphosphine, tributylphosphine, etc.), or trialkylphosphite (eg, trimethylphosphite, triethylphosphite, etc.) Among them, triphenylphosphine, tributylphosphine and the like are preferable. The organic phosphorus compound is used in an amount of 1 to 50 equivalents, preferably 1 to 10 equivalents, based on the amount of the raw material. These reactions can be performed in the presence of an appropriate inorganic salt (eg, lithium iodide, lithium bromide, lithium chloride, cerium chloride, etc.). The inorganic salt is used in an amount of 1 to 50 equivalents, preferably 1 to 10 equivalents, based on the raw material. These reactions are usually performed in a solvent that does not affect the reaction. Examples of the solvent include amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, etc.), ether solvents (eg, tetrahydrofuran, getyl ether, dioxane, dimethoxetane, etc.), Hydrocarbon solvents (eg, hexane, benzene, toluene, etc.), organic base solvents (eg, pyridine, 2,4,6-trimethylpyridine, picoline, 2,6-lutidine, 1,8-diazabicyclo [5 , 4, 0] -7-dendene and the like, or a mixed solvent thereof. Among them, tetrahydrofuran, getyl ether, dimethoxyethane, N, N-dimethylformamide, N, N-dimethylacetamide and the like are preferable. It is particularly preferred that these are anhydrous solvents. Although it depends on the type of reaction, the reaction temperature is from 100 to 150, preferably from 140 to 120, and the reaction time is from 2 to 120 hours, preferably from 10 to 4 hours. 8 hours.
一般式 ( I ) において In the general formula (I)
である化合物 (X X V I ) またはその塩の合成 4 Of compound (XXVI) or its salt Four
反応 1 8 Reaction 1 8
一般式 (X X) で表されるイミダゾチアジン化合物 (Zは C Oを示す。 ) または その塩を原料とし、酸と反応させることにより、 一般式 (X XV) で表されるイミ ダゾチアジン化合物またはその塩を得る。 An imidazothiazine compound represented by the general formula (XXV) or a salt thereof is obtained by reacting an imidazothiazine compound represented by the general formula (XX) (Z represents CO) or a salt thereof with an acid. Get the salt.
酸としては前記反応 5で示した酸などが挙げられ、 中でも例えば、 トリフルォロ 酢酸、 塩化水素酸、 臭化水素酸、 硫酸、 ギ酸、 酢酸、 メタンスルホン酸、 P -トル エンスルホン酸、カンファースルホン酸、 3フッ化ホウ素ェ一テル錯体などが特に 好ましい。 これらのうち液体のものは溶媒としても用いられる。酸は原料に対して 0 . 0 5力、ら 1 0等量、 好ましくは 0 . 1から 5等量、 さらに好ましくは 0 . 2から 2等量を用いる。 これらの反応は通常溶媒中で有利に行われる。溶媒は反応に影響 を与えないものであれば特定されないが、上記反応 2あるいは反応 5で示した溶媒 などが用いられ、 好ましくは有機酸系溶媒、 アミド系溶媒、 ェ一テル系溶媒、 アル コール系溶媒、二トリル系溶媒、炭化水素系溶媒、ハロゲン化炭化水素系溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも、 ギ酸、 酢酸、 N, N—ジメチル ホルムアミド、 テトラヒドロフラン、 ジォキサン、 ジメトキシェタン、 ァセトニト リル、 ベンゼン、 トルエン、 キシレン、 ジクロロメタン、 クロ口ホルム、 1, 2-ジク ロロェタン、 クロ口ベンゼンなどが特に好ましい。反応温度は _ 4 0 ないし 2 0 0で、 好ましくは 0 °Cないし 1 2 0 °C、 反応時間は 5分間ないし 1 2 0時間、好ま' しくは 3 0分間から 4 8時間である。 Examples of the acid include the acids shown in the above Reaction 5, among which, for example, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, formic acid, acetic acid, methanesulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid And boron trifluoride etherate complex are particularly preferred. Of these, liquid ones are also used as solvents. The acid is used in an amount of 0.05 to 10 equivalents, preferably 0.1 to 5 equivalents, more preferably 0.2 to 2 equivalents, based on the raw material. These reactions are usually advantageously performed in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in the above Reaction 2 or Reaction 5 is used, and preferably an organic acid solvent, an amide solvent, an ether solvent, or an alcohol. Solvents, nitrile-based solvents, hydrocarbon-based solvents, halogenated hydrocarbon-based solvents, and the like, and mixtures thereof. Among them, formic acid, acetic acid, N, N-dimethylformamide, tetrahydrofuran, dioxane, dimethoxy Tan, acetonitrile, benzene, toluene, xylene, dichloromethane, chloroform, 1,2-dichloroethane and cyclobenzene are particularly preferred. The reaction temperature is from −40 to 200, preferably from 0 ° C. to 120 ° C., and the reaction time is from 5 minutes to 120 hours, preferably from 30 minutes to 48 hours.
反応 1 9 Reaction 1 9
一般式 (X X V) で表されるイミダゾチアジン化合物またはその塩を原料とし、 塩基の存在下に一般式 R 1 4— L (Lは脱離基を表す。 ) またはその塩と反応させ ることにより、 一般式(XXV I ) またはその塩で表されるイミダゾチアジン化合 物を得る。 塩基としては、上記反応 2で示した塩基などが挙げられ、好ましい例'としては例 えば、 水素化アルカリ金属、 アルカリ金属のアルコキシド、 無機塩基、 有機塩基、 アルキルリチウム、またはアル力リ金属アミドなどが好ましぐ水素化ナトリゥム、 カリウム tert -ブトキシド、 炭酸ナトリウム、 炭酸カリウム、 トリェチルァミン、 ジィソプロピルェチルァミン、 ピリジン、 4 -ジメチルァミノピリジン、 1, 8 -ジァザ ビシクロ [5, 4, 0] - 7-ゥンデセン、 メチルリチウム、 ブチルリチウム、 リチウムジィ ソプロピルアミド、 リチウムピストリメチルシリルアミドなどが特に好ましい。 こ れらのうち液体のものは溶媒としても用いられる。塩基は 1ないし 5 0当量、好ま しくは 2ないし 1 0当量、 さらに好ましくは 2ないし 5当量を用いる。 Lは塩素、 臭素、 ヨウ素などのハロゲン原子などが好ましい。 R 1 4が R 1 5 Z—で表される化 合物において、 Lは塩素、 臭素、 ヨウ素などのハロゲン原子、 あるいはメタンスル ホニルォキシ、 p -トルエンスルホニルォキシ、 トリフルォロメタンスルホ二ルォキ シなどのスルホニルォキシ基などが好ましい。 上記一般式 R 1 4— Lで表される化 合物またはその塩は原料に対して 0 . 2力 ^ら 2 0当量、好ましくは 1から 1 0当量、 さらに好ましくは 1から 5当量を用いる。これらの反応は通常溶媒中で有利に行わ れる。溶媒は反応に影響を与えないものであれば特定されないが、上記反応 2で示 した溶媒などが用いられ、好ましくはアミド系溶媒、 ェ一テル系溶媒、 ケトン系溶 媒、 エステル系溶媒、 二トリル系溶媒、 アルコール系溶媒、 炭化水素系溶媒、 有機 塩基系溶媒など、 あるいはこれらの混合溶媒が挙げられ、 なかでも N, N _ジメチ ルホルムアミド、 N-メチルピロリドン、 テトラヒドロフラン、 ジォキサン、 ジメト キシェタン、 アセトン、 2-ブタノン、 酢酸ェチル、 酢酸 tert -プチル、 ァセトニト リル、トルエン、ピリジンなどが特に好ましい。反応温度は一 8 0 °Cないし 1 5 0 、 好ましくは 0 °Cないし 1 0 0 T:、反応時間は 1分間ないし 1 2 0時間、好ましくは 5分間ないし 4 8時間である。 Using an imidazothiazine compound represented by the general formula (XXV) or a salt thereof as a raw material and reacting with the general formula R 14 — L (L represents a leaving group) or a salt thereof in the presence of a base As a result, an imidazothiazine compound represented by the general formula (XXVI) or a salt thereof is obtained. Examples of the base include the bases shown in the above Reaction 2. Preferred examples are, for example, alkali metal hydrides, alkali metal alkoxides, inorganic bases, organic bases, alkyllithiums, and alkali metal amides. Preferred are sodium hydride, potassium tert-butoxide, sodium carbonate, potassium carbonate, triethylamine, diisopropylethylamine, pyridine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] ]-7-Pendecene, methyllithium, butyllithium, lithium disopropylamide, lithium pistrimethylsilylamide and the like are particularly preferred. Of these, liquid ones are also used as solvents. The base is used in an amount of 1 to 50 equivalents, preferably 2 to 10 equivalents, and more preferably 2 to 5 equivalents. L is preferably a halogen atom such as chlorine, bromine or iodine. In R 1 4 is compound of represented by R 1 5 Z-, L is chlorine, bromine, a halogen atom such as iodine or Metansuru Honiruokishi,, p - toluenesulfonyl O carboxymethyl, such as triflate Ruo b methanesulfonyl two Ruoki Shi Sulfonyloxy groups and the like are preferred. The compound represented by the above general formula R 14 -L or a salt thereof is used in an amount of 0.2 equivalents, preferably 1 to 10 equivalents, and more preferably 1 to 5 equivalents to the raw material. . These reactions are advantageously carried out usually in a solvent. The solvent is not specified as long as it does not affect the reaction, but the solvent shown in Reaction 2 above is used, and preferably an amide solvent, an ether solvent, a ketone solvent, an ester solvent, Examples include tolyl solvents, alcohol solvents, hydrocarbon solvents, organic base solvents, and the like, and mixtures thereof, among which N, N_dimethylformamide, N-methylpyrrolidone, tetrahydrofuran, dioxane, dimethyloxetane, Acetone, 2-butanone, ethyl acetate, tert-butyl acetate, acetonitril, toluene, pyridine and the like are particularly preferred. The reaction temperature is from 180 to 150, preferably from 0 to 100 T, and the reaction time is from 1 to 120 hours, preferably from 5 to 48 hours.
—般式 ( I ) において R 1が— S R 2、 —In general formula (I), R 1 is — SR 2 ,
χ γ が 4 χ γ is 4
である化合物 (X X X I I I ) またはその塩の合成 Of compound (XXXIII) or its salt
(XXX I I I ) 反応 20 (XXX I I I) Reaction 20
AD ANを原料とし、 一般式 R14- NC〇 (R 14は置換されていてもよい炭化水 素基を示す)で表されるイソシァネートを酸の存在下に反応させ、次いで塩基と反 応させることにより、 一般式 (XXV I I) で表される 5 -ジクロロメチレンイミ ダゾリジン化合物またはその塩を得る。 Using ADA as a raw material, an isocyanate represented by the general formula R 14 -NC を (R 14 represents an optionally substituted hydrocarbon group) is reacted in the presence of an acid, and then reacted with a base As a result, a 5-dichloromethyleneimidazolidine compound represented by the general formula (XXV II) or a salt thereof is obtained.
R14- NCOで表されるイソシァネート化合物は原料に対して 0.1ないし 10 当量、好ましくは 0.2ないし 5当量、さらに好ましくは 1ないし 2当量を用いる。 R 14- N C 0で表されるィソシァネート化合物は好ましくは R 14が例えば、メチル 基、 ベンジル基、 n -ブトキシカルポニルメチル基、 ェトキシカルポニルメチル基、 エトキシカルポニルェチル基などであるィソシァネートなどが特に好ましい。上記 酸触媒としては例えば、 ルイス酸 (例、 3フッ化ホウ素エーテル錯体、 塩化亜鉛、 塩化アルミニウム、 四塩化チタン、 四塩化スズなど) 、 有機スルホン酸 (例、 メタ ンスルホン酸、 オクタンスルホン酸、 ベンゼンスルホン酸、 P -トルエンスルホン 酸、 カンファースルホン酸など) 、 ハロ酢酸 (例えば、 トリフルォロ酢酸など) 、 無機酸 (例、 塩化水素酸、 臭化水素酸、 硫酸、 リン酸、 過塩素酸など) などが挙げ られ、 中でもルイス酸が好ましく、塩化アルミニウムが特に好ましい。酸は原料に 対して 0 . 1ないし 1 0当量、好ましくは 0 . 2ないし 5当量、 さらに好ましくは 1 ないし 2当量を用いる。 この反応は溶媒中で有利に行われる。 これらの反応は通常 反応に影響を与えない溶媒中で行われる。溶媒としては、反応に影響を与えないも のであれば特定されないが、 アミド系溶媒 (例、 N, N—ジメチルホルムアミド、 N , N—ジメチルァセトアミド、 N-メチルピロリドンなど) 、 ハロゲン化炭化水素 系溶媒 (例、 ジクロロメタン、 クロ口ホルム、 四塩化炭素、 1, 2-ジクロロェタン、 クロ口ベンゼンなど) 、 二トリル系溶媒 (例、 ァセトニトリルなど) 、 エステル系 溶媒(例、ギ酸ェチル、酢酸ェチル、酢酸 tert-ブチルなど)、ケトン系溶媒(例、 アセトン、 メチルェチルケトン、 メチルイソプチルケトンなど) 、 エーテル系溶媒 (例、 テトラヒドロフラン、 ジェチルエーテル、 ジイソプロピルエーテル、 ジォキ サン、 ジメ卜キシェタンなど) 、 炭化水素系溶媒 (例、 へキサン、 ベンゼン、 トル ェンなど) 、 芳香族有機塩基系溶媒 (例、 ピリジン、 2, 4, 6-トリメチルピリジン、 ピコリン、 2, 6-ルチジン、 1, 8-ジァザビシク口 [5, 4, 0] -7-ゥンデセンなど) など、 あるいはこれらの混合溶媒が挙げられる。なかでもハロゲン化炭化水素系溶媒、ェ ステル系溶媒、エーテル系溶媒、炭化水素系溶媒などが好ましく、ジクロロメタン、 クロ口ホルム、 テトラヒドロフラン、 ェチルエーテル、 トルエンなどが特に好まし い。 反応温度は— 4 0 °Cないし 1 5 0 °C, 好ましくは O ないし 1 0 O 、 反応時 間は 2分間ないし 9 6時間、 好ましくは 5分間ないし 6 0時間である。 The isocyanate compound represented by R 14 -NCO is used in an amount of 0.1 to 10 equivalents, preferably 0.2 to 5 equivalents, more preferably 1 to 2 equivalents, based on the amount of the raw material. The isocyanate compound represented by R 14 -NC 0 is preferably such that R 14 is, for example, methyl Particularly preferred is isocyanate which is a group, benzyl group, n-butoxycarbonylmethyl group, ethoxycarbonylmethyl group, ethoxycarbonylethyl group and the like. Examples of the acid catalyst include Lewis acids (eg, boron trifluoride ether complex, zinc chloride, aluminum chloride, titanium tetrachloride, tin tetrachloride, etc.), organic sulfonic acids (eg, methanesulfonic acid, octanesulfonic acid, benzene) Sulfonic acid, P-toluenesulfonic acid, camphorsulfonic acid, etc., haloacetic acid (eg, trifluoroacetic acid, etc.), inorganic acid (eg, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, perchloric acid, etc.) Among them, Lewis acids are preferable, and aluminum chloride is particularly preferable. The acid is used in an amount of 0.1 to 10 equivalents, preferably 0.2 to 5 equivalents, more preferably 1 to 2 equivalents, based on the amount of the raw material. This reaction is advantageously performed in a solvent. These reactions are usually performed in a solvent that does not affect the reactions. Solvents are not specified as long as they do not affect the reaction, but amide solvents (eg, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, etc.), halogenated carbon Hydrogen-based solvents (eg, dichloromethane, chloroform, carbon tetrachloride, 1,2-dichloroethane, cyclobenzene), nitrile solvents (eg, acetonitrile, etc.), ester solvents (eg, ethyl formate, ethyl acetate) , Tert-butyl acetate, etc.), ketone solvents (eg, acetone, methyl ethyl ketone, methyl isobutyl ketone, etc.), ether solvents (eg, tetrahydrofuran, getyl ether, diisopropyl ether, dioxan, dimethyloxetane, etc.) ), Hydrocarbon solvents (eg, hexane, benzene, toluene), aromatic solvents Organic base solvents (eg, pyridine, 2,4,6-trimethylpyridine, picoline, 2,6-lutidine, 1,8-diazabicyclic [5,4,0] -7-dendene, etc.) A mixed solvent is mentioned. Of these, halogenated hydrocarbon solvents, ester solvents, ether solvents, hydrocarbon solvents, and the like are preferable, and dichloromethane, chloroform, tetrahydrofuran, ethyl ether, and toluene are particularly preferable. The reaction temperature is −40 ° C. to 150 ° C., preferably O to 100 ° C., and the reaction time is 2 minutes to 96 hours, preferably 5 minutes to 60 hours.
用いる塩基としては、上記反応 2で示した塩基などが挙げられ、好ましい例とし ては例えば、ピリジン、 4 -ジメチルァミノピリジン、 1 , 8 -ジァザビシク口 [5, 4, 0] -7- ゥンデセン、 トリェチルァミン、 ジメチルァニリン、 炭酸ナトリウム、 炭酸力リウ ムなどが挙げられる。塩基は 0ないし 10当量、好ましくは 0ないし 1当量を用レ 反応温度は一 4 O ないし 150で、好ましくは Ot:ないし 100 、反応時間は 2分間ないし 96時間、 好ましくは 5分間ないし 60時間である。 Examples of the base to be used include the bases shown in the above Reaction 2, and preferable examples thereof include, for example, pyridine, 4-dimethylaminopyridine, 1,8-diazabicycline [5,4,0] -7-pentadecene , Triethylamine, Dimethylaniline, Sodium carbonate, Carbonated Liu And the like. The base is used in an amount of 0 to 10 equivalents, preferably 0 to 1 equivalent.The reaction temperature is 14 to 150, preferably Ot: to 100, and the reaction time is 2 to 96 hours, preferably 5 to 60 hours. is there.
反応 21 Reaction 21
一般式 (XXVI I) で表される 5-ジクロロメチレンイミダゾリジン化合物ま たはその を酸加水分解反応に付すことにより一般式 (XXV I I I)で表される 5 -ジク口ロメチレンイミダゾリジン化合物またはその塩を得る。 A 5-dichloromethylene imidazolidine compound represented by the general formula (XXVI I) or a 5-dichloromethylene imidazolidine compound represented by the general formula (XXV III) by subjecting the compound to an acid hydrolysis reaction or Obtain the salt.
用いられる酸としては、例えば上記反応 5で示した酸などが挙げられ、 中でも塩 化水素酸、 臭化水素酸、 硫酸、 リン酸、 メタンスルホン酸、 Ρ-トルエンスルホン 酸、カンファースルホン酸などが好ましい。 これらのうち液体のものは溶媒として も用いられる。酸は原料に対して 0.1ないし 20当量、好ましくは 0.5ないし 1 0当量、 さらに好ましくは 1ないし 5当量を用いる。 これらの反応は通常溶媒中で 有利に行われる。溶媒は反応に影響を与えないものであれば特定されないが、例え ば上記反応 5で示した溶媒に加えて例えば水などが挙げられ、好ましくは水、ギ酸、 酢酸、 Ν, Ν—ジメチルホルムアミド、 テトラヒドロフラン、 ジォキサン、 ジメト キシェタン、 ジメチルスルホキシド、 メタノール、 エタノール、 プロパノール、 ィ ソプロパノール、 イソブタノール、 tert-ブタノール、 ァセトニトリルなど、 ある いはこれらの混合溶媒が挙げられる。反応温度は一 40°Cないし 180°C、好まし くは Ot:ないし 120 、反応時間は 10分間ないし 72時間、好ましくは 30分 間ないし 48時間である。 Examples of the acid to be used include the acids shown in the above Reaction 5, etc., among them, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, methanesulfonic acid, Ρ-toluenesulfonic acid, camphorsulfonic acid and the like. preferable. Of these, liquid ones are also used as solvents. The acid is used in an amount of 0.1 to 20 equivalents, preferably 0.5 to 10 equivalents, more preferably 1 to 5 equivalents, based on the amount of the raw material. These reactions are advantageously carried out usually in a solvent. The solvent is not specified as long as it does not affect the reaction.Examples of the solvent include, for example, water in addition to the solvent shown in Reaction 5 above, and preferably water, formic acid, acetic acid, Ν, Ν-dimethylformamide, Examples thereof include tetrahydrofuran, dioxane, dimethoxetane, dimethyl sulfoxide, methanol, ethanol, propanol, isopropanol, isobutanol, tert-butanol, and acetonitrile, or a mixed solvent thereof. The reaction temperature is between 140 ° C. and 180 ° C., preferably Ot: 120, and the reaction time is between 10 minutes and 72 hours, preferably between 30 minutes and 48 hours.
反応 22 Reaction 22
前記記載の反応 2と同様の条件により、 一般式 (XXVI I I) で表される 5_ ジクロロメチレンイミダゾリジン化合物またはその塩を原料とし、塩基の存在下に 一般式 R〇COCH2CH2SHで表されるメルカプト化合物 (Rはメチル、 ェチ ル、 プロピル、 イソプロピル、 2-ェチルへキシルなどのアルキル基を表す。 ) と反 応させることにより、 一般式 (XX I X) で表されるイミダゾリジン化合物または その塩を得る。 Under the same conditions as in Reaction 2 described above, a 5_dichloromethyleneimidazolidine compound represented by the general formula (XXVI II) or a salt thereof is used as a raw material, and expressed by a general formula R〇COCH 2 CH 2 SH in the presence of a base. (R represents an alkyl group such as methyl, ethyl, propyl, isopropyl, 2-ethylhexyl, etc.) to form an imidazolidine compound represented by the general formula (XX IX). Or to obtain its salt.
反応 23 Reaction 23
前記記載の反応 4と同様の条件 (こより、 一般式(XX IX) で表されるイミダゾ リジン化合物(Rは前記アルキル基を示す。 )またはその塩を原料とし、一般式 (V) で表される cH立に脱離基 Lを有するケトン化合物またはその塩と塩基の存在下に 反応させることにより、 一般式 (X XX) で表されるイミダゾチアジン化合物 (R は前記アルキル基を示す。 ) またはその塩を得る。 The same conditions as in Reaction 4 described above (hereinafter, imidazo represented by the general formula (XX IX)) Using a lysine compound (R represents the above-mentioned alkyl group) or a salt thereof as a raw material, a ketone compound represented by the general formula (V) having a leaving group L at the cH position or a salt thereof is reacted in the presence of a base. As a result, an imidazothiazine compound represented by the general formula (XX) (R represents the above alkyl group) or a salt thereof is obtained.
反応 2 4 Reaction 2 4
前記記載の反応 5と同様の条件により、一般式 (X X X) で表されるイミダゾチ ァジン化合物 (Rは上記アルキル基を示す。 ) またはその塩を原料とし、 酸の存在 下に脱水反応に付すことにより、 一般式(X X X I ) で表されるイミダゾチアジン 化合物 (Rは上記アルキル基を示す。 ) またはその塩を得る。 Under the same conditions as in Reaction 5 described above, an imidazothiazine compound represented by the general formula (XXX) (R represents the above alkyl group) or a salt thereof is subjected to a dehydration reaction in the presence of an acid. As a result, an imidazothiazine compound represented by the general formula (XXXI) (R represents the above alkyl group) or a salt thereof is obtained.
反応 2 5 Reaction 2 5
前記記載の反応 6と同様の条件により、 一般式(XX X I )で表されるイミダゾ チアジン化合物 (Rは上記アルキル基を未す。 ) またはその塩を原料とし、塩基と 反応させることにより、一般式(X X X I I ) で表されるイミダゾチアジン化合物 またはその塩を得る。 Under the same conditions as in Reaction 6 described above, an imidazothiazine compound represented by the general formula (XX XI) (R does not have the above-mentioned alkyl group) or a salt thereof is used as a raw material and reacted with a base to give a general compound. An imidazothiazine compound represented by the formula (XXXII) or a salt thereof is obtained.
反応 2 6 Reaction 2 6
前記記載の反応 7— ( a) と同様の条件により、 一般式 (XXX I I ) で表され るイミダゾチアジン化合物を原料とし、 塩基の存在下に一般式 R 2— L (Lは脱離 基を表す。 ) と反応させることにより、 一般式 (X X X I I I ) で表されるイミダ ゾチアジン化合物を得る。 Under the same conditions as in Reaction 7- (a) described above, an imidazothiazine compound represented by the general formula (XXXII) is used as a starting material, and in the presence of a base, a compound represented by the general formula R 2 — L (L is a leaving group To obtain an imidazothiazine compound represented by the general formula (XXXIII).
上記の各反応においては、置換基によっては各反応条件下で分解、反応する官能 基を有する場合がありうるが、それらの保護、 および反応後の脱保護等は自体公知 の方法で行うことができる。また各置換基上に存在し得る各種官能基の変換につい ても自体自体公知の方法で行うことができる。 In each of the above reactions, some substituents may have functional groups that decompose and react under each reaction condition.However, their protection and deprotection after the reaction can be performed by a method known per se. it can. The conversion of various functional groups that may be present on each substituent can be performed by a method known per se.
例えばエステル化された力ルポキシル基を加水分解してカルボキシル基へとす る場合には、 例えば、酸あるいは塩基の存在下に有利に行われる。酸としては上記 反応 5で示した酸が、塩基としては上記反応 3で示した塩基などが挙げられ、 中で も塩化水素酸、 硫酸、 リン酸、 トリフルォロ酢酸、 メタンスルホン酸、 P -トルェ ンスルホン酸、 カンファースルホン酸など、 水素化ナトリウム、 ナトリウムメトキ シド、 ナトリウムエトキシド、 カリウム tert-ブトキシド、 炭酸ナトリウム、 炭酸 カリウム、 水酸化ナトリウム、 水酸化カリウム、 トリェチルァミン、 ジイソプロピ ルェチルァミン、 4 -ジメチルァミノピリジン、 1, 8-ジァザビシクロ[5,4, 0] -7-ゥン デセンなどが好ましい。 酸あるいは塩基は原料に対して 0 . 1から 2 0当量、 好ま しくは 1から 5当量を用いる。これらの反応は通常反応に影響を与えない溶媒中で 有利に行われ、水、または上記反応 2で示した溶媒などが用いられる。なかでも水、 テトラヒドロフラン、 ジォキサン、 ジメトキシェタン、 メタノール、 エタノール、 2 -プロパノール、 ter t-ブ夕ノール、 ァセトニトリル、 ピリジンなど、 あるいはこ れらの含水混合溶媒が好ましい。反応温度は— 2 0 °Cないし 2 0 O t、好ましくは 2 0 °Cないし 1 2 0 °C、反応時間は 5分間ないし 1 2 0時間、好ましくは 2 0分間 ないし 7 2時間である。 For example, in the case where an esterified hydroxyl group is hydrolyzed into a carboxyl group, the hydrolysis is advantageously performed in the presence of, for example, an acid or a base. Examples of the acid include the acid shown in Reaction 5 above, and examples of the base include the base shown in Reaction 3 above. Among them, hydrochloric acid, sulfuric acid, phosphoric acid, trifluoroacetic acid, methanesulfonic acid, P-toluenesulfone Acid, camphorsulfonic acid, etc., sodium hydride, sodium methoxide, sodium ethoxide, potassium tert-butoxide, sodium carbonate, carbonic acid Potassium, sodium hydroxide, potassium hydroxide, triethylamine, diisopropylethylamine, 4-dimethylaminopyridine, 1,8-diazabicyclo [5,4,0] -7-pinedecene and the like are preferable. The acid or base is used in an amount of 0.1 to 20 equivalents, preferably 1 to 5 equivalents, based on the starting material. These reactions are usually advantageously performed in a solvent that does not affect the reaction, and water, the solvent shown in the above Reaction 2, or the like is used. Among them, water, tetrahydrofuran, dioxane, dimethoxyethane, methanol, ethanol, 2-propanol, tert-butanol, acetonitrile, pyridine and the like, or a mixed solvent containing these waters are preferable. The reaction temperature is from −20 ° C. to 20 ° C., preferably from 20 ° C. to 120 ° C., and the reaction time is from 5 minutes to 120 hours, preferably from 20 minutes to 72 hours.
例えばエステル化もしくはアミド化されたカルボキシル基、 シァノ基、カルボ二 ル基などの官能基を還元してホルミル基、 ヒドロキシメチル基、 アミノメチル基、 水酸基などへと変換する場合には、 原料化合物を還元反応に付すことで得られる。 本反応は、 金属水素化物 (例、 水素化リチウムアルミニウム、 水素化ナトリウムァ ルミ二ゥム、 水素化ホウ素リチウム、水素化ホウ素ナトリウム、 シァノ水素化ホウ 素ナトリウムなど) 、 水素化アルミニウム (例、 水素化ジイソプチルアルミニウム など) 、 ジポラン (例、 ボラン-テトラヒドロフラン、 ポラン -メタノールなど) 、 ジアルキルボラン (例、 ジ -sec-プチルポラン、 ジテキシルポラン、 9 -ボラビシク 口 [3, 3, 1]ノルポルネンなど) などの還元剤と不活性溶媒中で反応させることによ り行う。不活性溶媒としては、 エーテル系溶媒 (例、 テトラヒドロフラン、 ジェチ ルエーテル、 ジォキサン、 ジメ卜キシェタンなど) 、 へキサン、 ベンゼン、 トルェ ンなどが挙げられる。還元剤は 1ないし 2 0当量、好ましくは 1ないし 5当量を用 い、 反応温度は一 7 0 °Cないし 1. 5 0 °C、 好ましくは 0 ないし 1 0 0 である。 反応時間は 1 0分間ないし 4 8時間、 好ましくは 1時間ないし 2 0時間である。 前記の各反応によって、 目的物が遊離の状態で得られる場合には、常法にしたが つて塩に変換してもよく、 また目的物が塩として得られる場合には、常法に従って 遊離体または他の塩に変換することもできる。かくして得られる化合物またはそれ らの塩は公知の手段、例えば、転溶、濃縮、溶媒抽出、分留、 For example, when a functional group such as an esterified or amidated carboxyl group, a cyano group, or a carbonyl group is reduced to be converted to a formyl group, a hydroxymethyl group, an aminomethyl group, a hydroxyl group, or the like, the starting compound is used. It is obtained by subjecting it to a reduction reaction. This reaction involves metal hydride (eg, lithium aluminum hydride, sodium aluminum hydride, lithium borohydride, sodium borohydride, sodium cyanoborohydride, etc.), aluminum hydride (eg, hydrogen Diisobutylaluminum, etc.), dipolane (eg, borane-tetrahydrofuran, polan-methanol, etc.), dialkylborane (eg, di-sec-butylbutyl, ditexylporan, 9-boravisic [3,3,1] norpolonene, etc.) The reaction is carried out by reacting with a reducing agent in an inert solvent. Examples of the inert solvent include ether solvents (eg, tetrahydrofuran, methyl ether, dioxane, dimethyloxetane, etc.), hexane, benzene, toluene and the like. The reducing agent is used in an amount of 1 to 20 equivalents, preferably 1 to 5 equivalents, and the reaction temperature is from 170 ° C to 1.50 ° C, preferably from 0 to 100 ° C. The reaction time is 10 minutes to 48 hours, preferably 1 hour to 20 hours. When the desired product is obtained in a free state by each of the above-mentioned reactions, it may be converted into a salt according to a conventional method, and when the desired product is obtained as a salt, the free form is obtained according to a conventional method Alternatively, it can be converted to another salt. The compound thus obtained or a salt thereof can be obtained by known means, for example, phase transfer, concentration, solvent extraction, fractionation,
結晶化、 再結晶等によって反応溶液から単離精製することができる < 本明細書記載の化合物の塩としては、薬理学的に許容される塩が好ましく、例え ば無機塩基との塩、 有機塩基との塩、 無機酸との塩、 有機酸との塩、 塩基性または 酸性ァミノ酸との塩などが挙げられる。 It can be isolated and purified from the reaction solution by crystallization, recrystallization, etc. < The salt of the compound described in the present specification is preferably a pharmacologically acceptable salt, for example, a salt with an inorganic base, a salt with an organic base, a salt with an inorganic acid, a salt with an organic acid, or a basic salt. Or a salt with an acidic amino acid.
無機塩基との塩の好適な例としては、例えばナトリウム塩、カリウム塩などのァ ルカリ金属塩;カルシウム塩、 マグネシウム塩などのアル力リ土類金属塩;ならび にアルミニウム塩、 アンモニゥム塩などが挙げられる。 Preferable examples of the salt with an inorganic base include alkali metal salts such as sodium salt and potassium salt; alkaline earth metal salts such as calcium salt and magnesium salt; and aluminum salt, ammonium salt and the like. Can be
有機塩基との塩の好適な例としては、例えばトリメチルァミン、 トリェチルアミ ン、 ピリジン、 ピコリン、 エタノールァミン、 ジエタノールァミン、 卜リエ夕ノー リレアミン、 ジシクロへキシルァミン、 Ν, Ν' -ジベンジルエチレンジァミンなどと の塩が挙げられる。 Preferred examples of the salt with an organic base include, for example, trimethylamine, triethylamine, pyridine, picoline, ethanolamine, diethanolamine, triethanolamine, dicyclohexylamine, Ν, Ν'-dibenzylethylene. And salts with diamine and the like.
無機酸との塩の好適な例としては、 例えば塩酸、 臭化水素酸、 硝酸、 硫酸、 リン 酸などとの塩が挙げられる。 Preferable examples of salts with inorganic acids include salts with hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid, and the like.
有機酸との塩の好適な例としては、 例えばギ酸、 酢酸、 トリフルォロ酢酸、 フマ ル酸、 シユウ酸、 酒石酸、 マレイン酸、 クェン酸、 コハク酸、 リンゴ酸、 メタンス ルホン酸、 ベンゼンスルホン酸、 Ρ-トルエンスルホン酸などとの塩が挙げられる。 塩基性アミノ酸との塩の好適な例としては、 例えばアルギニン、 リジン、 オル二 チンなどとの塩が挙げられ、酸性アミノ酸との塩の好適な例としては、例えばァス パラギン酸、 グルタミン酸などとの塩が挙げられる。 Preferred examples of salts with organic acids include, for example, formic acid, acetic acid, trifluoroacetic acid, fumaric acid, oxalic acid, tartaric acid, maleic acid, citric acid, succinic acid, malic acid, methanesulfonic acid, benzenesulfonic acid, -Salts with toluenesulfonic acid and the like. Preferred examples of the salt with a basic amino acid include, for example, salts with arginine, lysine, ordinine, and the like. Preferred examples of the salt with an acidic amino acid include, for example, aspartic acid, glutamic acid, and the like. Salts.
本発明の化合物 (I ) は、 水和物であってもよく、 非水和物であってもよい。 ま た、 本発明で用いられる化合物(I ) は、 骨格中に 1つの不斉炭素を持ちうるため 2種の光学異性体が存在し得るが、それらの各異性体、およびそれらの混合物も本 発明に含まれる。 またその置換基に不斉炭素、不飽和結合を有する場合も同様に立 体異性体、 幾何異性体を生ずるが、 それらの各異性体、 およびそれらの混合物も本 発明に含まれる。 The compound (I) of the present invention may be a hydrate or a non-hydrate. In addition, the compound (I) used in the present invention may have one asymmetric carbon in the skeleton and thus may have two kinds of optical isomers. Each of these isomers and a mixture thereof are also described in the present invention. Included in the invention. When the substituent has an asymmetric carbon atom or an unsaturated bond, a stereoisomer or a geometric isomer is similarly produced, and the isomers and mixtures thereof are also included in the present invention.
本発明の化合物は、 その構造中に硫黄原子を有するが、 これらは酸化剤 (例、 過 酸化水素または m-クロ口過安息香酸など) を用いて常法に従い酸化されていても よく、 スルホキシド、 スルホン化された化合物も本発明に含まれる。本発明の化合 物は、 その構造中または置換基にアミド、 イミド、 アミジノ、 グァニジノあるいは ゥレイド基を有することがあるが、これらはその 2重結合が異性化した共鳴構造を とる場合があるが、これら共鳴構造化合物も本発明に含まれる。 The compound of the present invention has a sulfur atom in its structure, which may be oxidized by an oxidizing agent (eg, hydrogen peroxide or m-chloroperbenzoic acid) according to a conventional method. Sulfonated compounds are also included in the present invention. The compound of the present invention may have an amide, imide, amidino, guanidino or peridode group in its structure or as a substituent, and these compounds have a resonance structure in which the double bond is isomerized. In some cases, these resonance structural compounds are also included in the present invention.
本発明の化合物(I ) はプロドラッグとして用いてもよく、 かかるプロドラッグ としては、生体内における生理条件下で酵素や胃酸等による反応により化合物(I ) に変換する化合物、すなわち酵素的に酸化、還元、加水分解等を起こして化合物( I ) に変化する化合物、 胃酸等により加水分解などを起こして化合物( I ) に変化する 化合物をいう。 化合物 (I ) のプロドラッグとしては、 化合物 (I ) のァミノ基が ァシル化、 アルキル化、 りん酸化された化合物 (例、 化合物 (I ) のァミノ基がェ ィコサノィル化、 ァラニル化、 ペンチルァミノカルポニル化、 (5—メチル—2— ォキソ一 1, 3—ジォキソレン一 4一ィル) メトキシカルポニル化、 テトラヒドロ フラニル化、 ピロリジルメチル化、 ビバロイルォキシメチル化、 t e r t—ブチル 化された化合物など) ;化合物(I )の水酸基がァシル化、アルキル化、 りん酸化、 ほう酸化された化合物(例、化合物( I )の水酸基がァセチル化、パルミトイル化、 プロパノィル化、 ピバロイル化、 サクシ二ル化、 フマリル化、 ァラニル化、 ジメチ ルアミノメチルカルポニル化された化合物など) ;化合物 ( I ) のカルボキシル基 がエステル化、 アミド化された化合物 (例、 化合物 ( I ) の力ルポキシル基がェチ ルエステル化、 フエニルエステル化、 カルポキシメチルエステル化、 ジメチルアミ ノメチルエステル化、 ピバロイルォキシメチルエステル化、エトキシカルポニルォ キシェチルエステル化、 'フタリジルエステル化、 ( 5—メチル一 2 _ォキゾ一 1, 3 _ジォキソレン— 4—ィル)メチルエステル化、 シク口へキシルォキシ力ルポ二 ルェチルエステル化、 メチルアミド化された化合物など) ;等が挙げられる。 これ らの化合物は自体公知の方法によって化合物 I ) から製造することができる。 また、 化合物 (I ) のプロドラッグは、 広川書店 1 9 9 0年刊 「医薬品の開発」 第 7巻分子設計 1 6 3頁から 1 9 8頁に記載されているような、生理的条件で化合 物 ( I ) に変化するものであってもよい。 The compound (I) of the present invention may be used as a prodrug. As such a prodrug, a compound which is converted into a compound (I) by a reaction with an enzyme or gastric acid under physiological conditions in a living body, that is, a compound which is oxidized enzymatically , A compound that undergoes reduction, hydrolysis, etc., and changes to the compound (I), and a compound, which undergoes hydrolysis, etc., due to stomach acid or the like, to change to the compound (I). Examples of the prodrug of the compound (I) include compounds in which the amino group of the compound (I) is acylated, alkylated, and phosphorylated (eg, the amino group of the compound (I) is icosanoylated, alanylated, pentylamino). Carponylated, (5-methyl-2-oxo- 1,3-dioxolen-141-yl) methoxycarbonylated, tetrahydrofuranylated, pyrrolidylmethylated, bivaloyoxymethylated, tert-butylated compounds Compounds in which the hydroxyl group of compound (I) is acylated, alkylated, phosphorylated or borated (eg, the hydroxyl group of compound (I) is acetylated, palmitoylated, propanoylated, pivaloyylated, succinylated) , Fumarylation, aranylation, dimethylaminomethylcarbonylated compounds, etc.); Compounds (e.g., compound (I) is converted to ethyl ester, phenyl ester, carboxymethyl ester, dimethylaminomethyl ester, pivaloyloxymethyl ester, Ethoxycarponyloxetyl esterification, 'phthalidyl esterification, (5-methyl-1-oxo-1,3,3-dioxolen-4-yl) methyl esterification, hexyloxyl-propyl-2-ethyl esterification, methylamide And the like); and the like. These compounds can be produced from compound I) by a method known per se. The prodrug of compound (I) is compounded under physiological conditions as described in Hirokawa Shoten, 1990, “Development of Pharmaceuticals”, Vol. 7, Molecular Design, pp. 163 to 198. It may change to an object (I).
軟骨基質の分解を抑制する化合物は、 変形性関節症および軟骨破壊に起因する 疾患の予防 ·治療剤として用いることが可能である。 すなわち、 プロテオグリカ ン分解抑制作用を有する化合物、 Π 型コラーゲン分解抑制作用を有する化合物、 マトリックスメタ口プロティナ一ゼー 1 3 (MM P - 1 3 ) やァグリカナーゼな どの軟骨基質分解酵素産生抑制作用を有する化合物および MM P遺伝子発現抑制 作用を有する化合物は、 変形性関節症および軟骨破壊に起因する疾患の予防 ·治 療剤として用いられる。 プロテオダリカン分解抑制作用を有する化合物、 I I型コ ラーゲン分解抑制作用を有する化合物、 MM P— 1 3やァグリカナ一ゼなどの軟 骨基質分解酵素産生抑制作用を有する化合物および MM P遺伝子発現抑制作用を 有する化合物としては、 縮合ィミダゾリジン誘導体が好ましい例として挙げられ、 より具体的には当該化合物 (I ) またはその塩またはそのスルホキシドが好まし い例として挙げられる。 Compounds that inhibit cartilage matrix degradation can be used as preventive and therapeutic agents for osteoarthritis and diseases caused by cartilage destruction. Compounds that inhibit proteoglycan degradation, compounds that inhibit type I collagen degradation, and that inhibit the production of cartilage matrix-degrading enzymes such as matrix meta-oral proteinase 13 (MMP-13) and aggrecanases Compound and MMP gene expression suppression The compound having an effect is used as a prophylactic / therapeutic agent for diseases caused by osteoarthritis and cartilage destruction. Compounds that inhibit proteodalican degradation, compounds that inhibit type II collagen degradation, compounds that inhibit the production of cartilage matrix-degrading enzymes such as MMP-13 and aglycanase, and those that inhibit MMP gene expression Preferred examples of the compound having the following are condensed imidazolidine derivatives, and more specifically, the compound (I) or a salt thereof or a sulfoxide thereof.
当該化合物 (I ) またはその塩またはそのスルホキシドには優れたプロテオグ リカン遊離抑制活性、 I I型コラーゲン分解抑制作用および MM P— 1 3やァグリ カナーゼなどの軟骨基質分解酵素産生抑制活性があり、 病変しつつある軟骨組織 の状態の悪化を阻止するという点から、 化合物 (I ) またはその塩を有効成分と する本発明の軟骨疾患治療薬は、 病変が骨自体に至る以前の軟骨疾患に特に有用 であり、 変形性関節症や慢性関節リウマチ、 軟骨破壊に起因する疾患などの初期 段階の治療はもちろんのこと、 これらの疾患の予防にも応用できる。 The compound (I) or a salt thereof or a sulfoxide thereof has excellent proteoglycan release inhibitory activity, collagen II degradation inhibitory activity, and cartilage matrix degrading enzyme production inhibitory activity such as MMP-13 and aggrecanase. The therapeutic agent for cartilage disease of the present invention containing Compound (I) or a salt thereof as an active ingredient is particularly useful for cartilage disease before the lesion reaches the bone itself, in terms of preventing deterioration of the cartilage tissue condition. Yes, it can be applied not only to treatment of osteoarthritis, rheumatoid arthritis and diseases caused by cartilage destruction, but also to prevention of these diseases.
当該化合物 (I ) またはその塩またはそのスルホキシドには優れた MM P遺伝 子 (特に MM P— 1 3遺伝子) 発現抑制作用があり、 総ての MM P関連疾患安全 な予防、 治療薬として有用である。 The compound (I) or its salt or its sulfoxide has an excellent inhibitory effect on the expression of the MMP gene (particularly the MMP-13 gene), and is useful as a safe preventive and therapeutic agent for all MMP-related diseases. is there.
即ち、 化合物 (I ) またはその塩またはそのスルホキシドは、 プロテオグリカ ン遊離抑制作用、 Π型コラーゲン分解抑制作用および MM P— 1 3やァグリカナ ーゼなどの軟骨基質分解酵素産生抑制作用を示し、軟骨基質、具体的 tはプロテオ ダリカンおよび I I型コラーゲンの分解を阻止する。 すなわち、 プロテオダリカン 分解抑制剤、 Π 型コラーゲン分解抑制剤および軟骨基質分解酵素抑制剤としての 作用を有する。その結果、強い軟骨破壊抑制作用を有しているので、哺乳動物(例、 ヒト、 ラット、 モルモット、 マウス、 ネコ、 ィヌ、 ゥサギ、 牛、 豚など) におけ る種々の軟骨の疾患 (変形性関節症や軟骨破壊に起因する疾患) 、 例えば、 軟骨 が関与する変形性膝関節炎、 軟骨が関与する慢性関節リウマチ、 あるいはそれら の類縁疾患における関節等における軟骨の破壊等の予防ならびに治療のために使 用され得る。 That is, compound (I) or a salt thereof or a sulfoxide thereof exhibits an inhibitory action on proteoglycan release, an inhibitory action on collagen type I degradation, and an inhibitory action on cartilage matrix degrading enzyme production such as MMP-13 and aglycanase. Substrates, specifically t, inhibit the degradation of proteodalican and type II collagen. That is, it has an effect as a proteodalican degradation inhibitor, a type I collagen degradation inhibitor, and a cartilage matrix degrading enzyme inhibitor. As a result, it has a strong inhibitory effect on cartilage destruction, so it can be used for various cartilage diseases (deformation) in mammals (eg, humans, rats, guinea pigs, mice, cats, dogs, puppies, cows, pigs, etc.). Osteoarthritis and diseases caused by cartilage destruction), for example, for the prevention and treatment of osteoarthritis involving cartilage, rheumatoid arthritis involving cartilage, or cartilage destruction in joints etc. in related diseases Can be used for
また化合物 (I ) またはその塩またはそのスルホキシドには優れた MM P遺伝子 (特に MM P— 1 3遺伝子) 発現抑制作用があり、 総ての MM P関連疾患、 例え ば、 上記の関節疾患 (例えば、 変形関節症、 慢性関節リウマチなど) に加え、 骨粗 鬆症、癌(例えば、原発性、転移性または再発性の、乳癌、前立腺癌、勝癌、 胃癌、 肺癌、大腸癌(結腸癌、直腸癌、肛門癌)、食道癌、十二指腸癌、頭頸部癌(舌癌、 咽頭癌、 喉頭癌) 、 脳腫瘍、 神経鞘腫、 非小細胞肺癌、 肺小細胞癌、 肝臓癌、 腎臓 癌、 胆管癌、 子宮癌 (子宮体癌、 子宮頸癌) 、 卵巣癌、 膀胱癌、 皮膚癌、 血管腫、 悪性リンパ腫、 悪性黒色腫、 甲状腺癌、 骨腫瘍、 血管腫、 血管線維腫、 網膜肉腫、 陰茎癌、 小児固形癌、 力ポジ肉腫、 A I D Sに起因する力ポジ肉腫、 上顎洞腫瘍、 線維性組織球腫、 平滑筋肉腫、 横紋筋肉腫、 脂肪肉腫、 子宮筋腫、 骨芽細胞腫、 骨 肉腫、 軟骨肉腫、 癌性の中皮腫瘍、 '白血病などの腫瘍など) 、 歯周病、 角膜潰瘍、 慢性潰瘍、 病的骨吸収 (ベ一ジェット病など) 、 腎炎、 血管新生、 動脈瘤、 動脈硬 化、 肺気腫、 肝硬変、 自己免疫疾患 (クローン病、 シ: n—ダレン病など) 、 癌の浸 潤 -転移、 などの安全な予防、 治療薬として有用である。 Compound (I) or its salt or its sulfoxide has an excellent MMP gene (Especially the MMP-13 gene) has an expression-suppressing effect, and in addition to all MMP-related diseases, for example, the above-mentioned joint diseases (eg, osteoarthritis, rheumatoid arthritis, etc.), osteoporosis, Cancer (eg, primary, metastatic or recurrent, breast, prostate, cancer, stomach, lung, colon (colon, rectum, anal), esophagus, duodenum, head and neck (tongue) Cancer, pharyngeal cancer, laryngeal cancer), brain tumor, schwannoma, non-small cell lung cancer, small cell lung cancer, liver cancer, kidney cancer, bile duct cancer, uterine cancer (uterine body cancer, cervical cancer), ovarian cancer, bladder Cancer, skin cancer, hemangiomas, malignant lymphomas, malignant melanomas, thyroid cancer, bone tumors, hemangiomas, angiofibromas, retinal sarcomas, penile cancers, pediatric solid carcinomas, lipopositive sarcomas, lipopositive sarcomas caused by AIDS, Maxillary sinus tumor, fibrous histiocytoma, leiomyosarcoma, rhabdomyosarcoma, liposarcoma, child Fibroids, osteoblastomas, osteosarcomas, chondrosarcomas, carcinomatous mesothelial tumors, tumors such as leukemia), periodontal disease, corneal ulcers, chronic ulcers, pathological bone resorption (such as Beiget's disease), Useful as a safe preventive and therapeutic agent for nephritis, angiogenesis, aneurysm, arterial sclerosis, emphysema, cirrhosis, autoimmune diseases (Crohn's disease, Si: n-Darren's disease, etc.), cancer invasion and metastasis, etc. is there.
本発明において、 化合物 (I ) またはその塩またはそのスルホキシドを軟骨疾 患予防 ·治療剤として用いる場合、 常法に従って薬学的に許容される担体と配合 することにより、 医薬として適切な剤型の、 錠剤、 カプセル剤、 顆粒剤、 散剤、 座剤などの固形製剤;またはシロップ剤、 点滴剤、 液剤、 懸濁液剤、 注射剤など の液状製剤として製剤化することができ、 経口または非経口的に投与することが できる。 . In the present invention, when the compound (I) or a salt thereof or a sulfoxide thereof is used as an agent for preventing or treating cartilage disease, the compound (I) or a salt thereof is mixed with a pharmaceutically acceptable carrier according to a conventional method to obtain a drug having a dosage form suitable as a medicament. Solid preparations such as tablets, capsules, granules, powders, and suppositories; or liquid preparations such as syrups, drops, solutions, suspensions, and injections, orally or parenterally It can be administered. .
薬学的に許容される担体としては、 製剤素材として慣用の各種有機あるいは無 機担体物質が用いられ、 固形製剤における賦形剤、 滑沢剤、 結合剤、 崩壊剤;液 状製剤における溶剤、 溶解補助剤、 懸濁化剤、 等張化剤、 緩衝剤、 無痛化剤、 安 定化剤、抗凝固剤などとして配合される。 また必要に応じて、防腐剤、抗酸化剤、 着色剤、 甘味剤などの製剤添加物を用いることもできる。 賦形剤の好適な例とし ては、 例えば乳糖、 白糖、 D—マンニトール、 デンプン、 結晶セルロース、 軽質 無水ゲイ酸などが挙げられる。 滑沢剤の好適な例としては、 例えばステアリン酸 マグネシウム、 ステアリン酸カルシウム、 タルク、 コロイドシリカなどが挙げら れる。 結合剤の好適な例としては、 例えば結晶セルロース、 白糖、 D—マンニト —ル、 デキストリン、 ヒドロキシプロピルセルロース、 ヒドロキシプロピルメチ ルセルロース、 ポリビエルピロリドンなどが挙げられる。 崩壊剤の好適な例とし ては、 例えばデンプン、 カルポキシメチルセルロース、 カルポキシメチルセル口 ースカルシウム、 クロスカルメロースナトリウム、 カルポキシメチルス夕一チナ トリウムなどが挙げられる。 溶剤の好適な例としては、 例えば注射用水、 アルコ ール、 プロピレングリコール、 マクロゴール、 ゴマ油、 トウモロコシ油などが挙 げられる。 溶解補助剤の好適な例としては、 例えばポリエチレングリコール、 プ ロピレンダリコール、 D—マンニトール、 シクロデキストリン類、 ツイ一ン、 安 息香酸ベンジル、 エタノール、 トリスァミノメタン、 コレステロール、 トリエタ ノールァミン、 炭酸ナトリウム、 クェン酸ナトリウムなどが挙げられる。 懸濁化 剤の好適な例としては、 例えばステアリルトリエタノールァミン、 ラウリル硫酸 ナトリウム、 ラウリルアミノプロピオン酸、 レシチン、 塩化ベンザルコニゥム、 塩化べンゼトニゥム、 モノステアリン酸グリセリン、 などの界面活性剤;例えば ポリピニルアルコール、 ポリビニルピロリドン、 カルボキシメチルセルロースナ トリウム、 メチルセルロース、 ヒドロキシメチルセルロース、 ヒドロキシェチル セルロース、 ヒドロキシプロピルセルロースなどの親水性高分子などが挙げられ る。 等張化剤の好適な例としては、 例えば塩化ナトリウム、 グリセリン、 D—マ ンニ] ^一ル、 グルコース, ソルビ] ルなどが挙げられる。 緩衝剤の好適な例と しては、 例えばリン酸塩、 酢酸塩、 炭酸塩、 クェン酸塩などの緩衝液などが挙げ られる。 無痛化剤の好適な例としては、 例えばべンジルアルコールなどが挙げら れる。 安定化剤の好適な例としては、 例えばポリエチレングリコール, ポリ乳酸 などが挙げられる。 坊凝固剤の好適な例としては、 例えばデキストラン硫酸, へ パリンなどが挙げられる。 防腐剤の好適な例としては、 例えばパラォキシ安息香 酸エステル類、クロロブタノ一ル、ベンジルアルコール、フエネチルアルコール、 ' デヒドロ酢酸、 ソルビン酸などが挙げられる。 抗酸化剤の好適な例としては、 例 えば亜硫酸塩、 ァスコルビン酸などが挙げられる。 投与に当たっては、 これら化 合物( I )またはその塩またはそのスルホキシドを慣用の水性希釈剤中に溶解し、 液剤として用いる。 希釈剤としてはぶどう糖水溶液、 生理食塩水、 リンゲル液、 栄 養補給剤液などが含まれる。 Pharmaceutically acceptable carriers include various organic or inorganic carrier materials commonly used as drug substances, such as excipients, lubricants, binders, and disintegrants in solid preparations; solvents and dissolution in liquid preparations. It is formulated as an auxiliary, suspending agent, tonicity agent, buffer, soothing agent, stabilizing agent, anticoagulant, and the like. If necessary, pharmaceutical additives such as preservatives, antioxidants, coloring agents and sweeteners can also be used. Preferred examples of the excipient include lactose, sucrose, D-mannitol, starch, crystalline cellulose, light gay anhydride, and the like. Preferred examples of the lubricant include magnesium stearate, calcium stearate, talc, colloidal silica and the like. Suitable examples of the binder include, for example, crystalline cellulose, sucrose, D-mannitol, dextrin, hydroxypropylcellulose, and hydroxypropylmethyl. And cellulose bilipyrrolidone. Preferable examples of the disintegrant include starch, carboxymethylcellulose, carboxymethylcellulose calcium, croscarmellose sodium, carboxymethylsuniform sodium and the like. Preferred examples of the solvent include water for injection, alcohol, propylene glycol, macrogol, sesame oil, corn oil and the like. Preferable examples of the solubilizing agent include, for example, polyethylene glycol, propylene glycol, D-mannitol, cyclodextrins, twins, benzyl benzoate, ethanol, trisaminomethane, cholesterol, triethanolamine, and carbonic acid. Sodium and sodium citrate. Preferred examples of the suspending agent include surfactants such as, for example, stearyltriethanolamine, sodium lauryl sulfate, laurylaminopropionic acid, lecithin, benzalkonium chloride, benzethonium chloride, and glycerin monostearate; And hydrophilic polymers such as benzyl alcohol, polyvinylpyrrolidone, sodium carboxymethylcellulose, methylcellulose, hydroxymethylcellulose, hydroxyethylcellulose, and hydroxypropylcellulose. Preferable examples of the tonicity agent include sodium chloride, glycerin, D-manniyl, glucose, sorbyl, and the like. Preferred examples of the buffer include buffers such as phosphate, acetate, carbonate, and citrate. Preferable examples of the soothing agent include benzyl alcohol and the like. Preferable examples of the stabilizer include polyethylene glycol, polylactic acid and the like. Preferred examples of the coagulant include dextran sulfate, heparin and the like. Preferred examples of preservatives include, for example, paraoxybenzoates, chlorobutanol, benzyl alcohol, phenethyl alcohol, dehydroacetic acid, sorbic acid and the like. Suitable examples of the antioxidant include, for example, sulfite, ascorbic acid and the like. For administration, the compound (I) or a salt thereof or a sulfoxide thereof is dissolved in a conventional aqueous diluent and used as a solution. Diluents include glucose aqueous solution, physiological saline, Ringer's solution, and nutritional supplement solution.
適当な方法で化合物 (I ) またはその塩またはそのスルホキシドを分散させた 懸濁液を溶解し、 マイクロカプセル、 球状、 棒状、 針状、 ペレット状、 フィルム 状等に賦形して本発明の予防治療剤を製造することもできる。 また本発明の予防 '治療剤は、 化合物 (I ) またはその塩またはそのスルホキシドと生体内分解性高 分子化合物とを含む徐放性製剤に成形してもよい。 該徐放性製剤の調製は、 特開 平 9一 2 6 3 5 4 5号公報に記載の方法に準ずることができる。 Compound (I) or a salt thereof or a sulfoxide thereof was dispersed by an appropriate method. The prophylactic / therapeutic agent of the present invention can also be produced by dissolving the suspension and shaping it into microcapsules, spheres, rods, needles, pellets, films and the like. Further, the prophylactic / therapeutic agent of the present invention may be formed into a sustained-release preparation containing compound (I) or a salt thereof or a sulfoxide thereof and a biodegradable high molecular compound. The preparation of the sustained-release preparation can be in accordance with the method described in JP-A-9-1265355.
以上の製剤化において、 化合物 (I ) またはその塩またはそのスルホキシドを 含有させる割合は、製剤全体に対して約 0 . 1ないし約 9 5重量%、好ましくは約 1ないし約 5 0重量%である。 化合物 (I ) またはその塩またはそのスルホキシ ドは経口用製剤として用いることが好ましい。 化合物 (I ) またはその塩または そのスルホキシドは、 局所投与製剤に成形して投与すると軟骨疾患の患部に直接 投与することもできる。 この場合は、 注射剤とするのが好ましい。 局所投与用の 非経口剤 (例、 筋肉内、 皮下、 臓器、 関節部位などへの注射剤、 埋め込み剤、 顆 粒剤、 散剤等の固形製剤、 懸濁剤等の液剤、 軟膏剤等) などとして投与すること もできる。 In the above formulation, the ratio of containing compound (I) or a salt thereof or a sulfoxide thereof is about 0.1 to about 95% by weight, preferably about 1 to about 50% by weight, based on the whole preparation. . Compound (I) or a salt thereof or a sulfoxide thereof is preferably used as an oral preparation. Compound (I) or a salt thereof or a sulfoxide thereof can be directly administered to a cartilage disease-affected site when formed into a topical preparation and administered. In this case, injection is preferable. Parenteral preparations for topical administration (eg, injection preparations for intramuscular, subcutaneous, organ, joint, etc., solid preparations such as implants, granules, powders, liquid preparations such as suspensions, ointments, etc.) It can also be administered as
例えば、 注射剤とするには、 化合物 (I ) またはその塩またはそのスルホキシ ドを分散剤 (例、 Tween 8 0、 H C O - 6 0等の界面活性剤、 カルポキシメチル セルロース、 アルギン酸ナトリウム、 ヒアルロン酸等の多糖類、 ポリソルベート 等) 、 保存剤 (例、 メチルパラベン、 プロピルパラベン等) 、 等張化剤 (例、 塩 化ナトリウム、 マンニトール、 ソルビトール、 ブドウ糖等) 、 緩衝剤 (例、 炭酸 カルシウム等) 、 p H調整剤 (例、 リン酸ナトリウム、 リン酸カリウム等) 等と 共に水性懸濁剤とすることにより実用的な注射用製剤が得られる。また、ゴマ油、 コーン油などの植物油あるいはこれにレシチンなどのリン脂質を混合したもの、 あるいは中鎖脂肪酸トリグリセリド (例、 ミグリオ一ル 8 1 2等) と共に分散し て油性懸濁剤として実際に使用できる注射剤とする。 For example, to prepare an injection, compound (I) or a salt thereof or a sulfoxide thereof is dispersed in a dispersant (eg, a surfactant such as Tween 80, HCO-60, carboxymethyl cellulose, sodium alginate, hyaluronic acid). , Polysorbates, etc.), preservatives (eg, methyl paraben, propyl paraben, etc.), tonicity agents (eg, sodium chloride, mannitol, sorbitol, glucose, etc.), buffering agents (eg, calcium carbonate, etc.), A practical injectable preparation can be obtained by preparing an aqueous suspension together with a pH adjuster (eg, sodium phosphate, potassium phosphate, etc.). In addition, vegetable oils such as sesame oil and corn oil, or mixtures of these with phospholipids such as lecithin, or dispersed with medium-chain fatty acid triglycerides (eg, migliol 812) are used as oily suspending agents. Make injections that can be used.
とりわけ、 変形性関節症患者に対しては関節腔内へ局所投与剤として直接投与 するような場合には、 注射用ヒアルロン酸製剤 (例えば、 科研製薬製:アルッ注) を分散媒として、 化合物 (I ) またはその塩またはそのスルホキシドを分散する ことにより調製することができる。 分散媒中に用いられるヒアルロン酸は、 その 非毒性塩を用いてもよく、 その例としてはナトリウム、 カリウムなどのアルカリ 金属塩、 マグネシウム、 カルシウムなどのアルカリ土類金属塩があげられ、 とり わけナトリウム塩が好ましく用いられる。 ヒアルロン酸およびその非毒性塩とし ては分子量約 2 0万ないし 5 0 0万 (粘度法) 、 好ましくは約 5 0万ないし 3 0 0万、 さらに好ましくは約 7 0万ないし 2 5 0万のものが用いられる。 In particular, when osteoarthritis patients are to be administered directly into the joint cavity as a topical agent, a compound (e.g., a hyaluronic acid formulation for injection (eg, Kaken Pharmaceutical Co., Ltd .: Aru Note)) should be used as a dispersion medium. It can be prepared by dispersing I) or a salt thereof or a sulfoxide thereof. Hyaluronic acid used in the dispersion medium may be a non-toxic salt thereof, for example, an alkali such as sodium or potassium. Metal salts, salts of alkaline earth metals such as magnesium, calcium and the like can be mentioned, and particularly, sodium salts are preferably used. Hyaluronic acid and non-toxic salts thereof have a molecular weight of about 200,000 to 500,000 (viscosity method), preferably about 500,000 to 300,000, and more preferably about 700,000 to 250,000. Things are used.
本分散剤におけるヒアルロン酸またはヒアルロン酸ナトリゥムの最終濃度は 1 % (W/V) 未満が粘度として適当であり各種操作、 投与の容易さなどの点で 好ましく、とりわけ約 0 . 0 2ないし 1 %未満が好ましぐさらに好ましくは約 0 . 1ないし 1 % (W/V) である。 The final concentration of hyaluronic acid or sodium hyaluronate in this dispersant is preferably less than 1% (W / V) as a viscosity, and is preferable in terms of various operations and ease of administration, and particularly about 0.02 to 1%. It is more preferably less than about 0.1 to 1% (W / V).
上記分散媒には、 自体公知の方法により、 p H調節剤、局所麻酔剤、抗生物質、 溶解補助剤、 等張化剤、 吸着防止剤、 グリコサミノダリカン、 多糖類などを含有 させてもよい。 その好ましい例としては、 マンニ! ル、 ソルビトール、 食塩、 グリシン、 酢酸アンモニゥム、 あるいは実質的に薬理活性を示さずに体液内に注 入しうる水溶性蛋白などが挙げられる。 該グリコサミノダリカンとしては、 ヒア ルロン酸、 コンドロイチン、 コンドロイチン硫酸 A、 コンドロイチン硫酸 C、 デ ルマタン硫酸、 へパリン、 へパラン硫酸、. ケウタン硫酸などが挙げられる。 該多 糖類としては、 アルギニン酸のような酸性多糖類が挙げられる。 The dispersion medium may contain a pH adjuster, a local anesthetic, an antibiotic, a solubilizer, an isotonic agent, an anti-adsorption agent, glycosaminodalican, a polysaccharide, etc. by a method known per se. Good. Preferred examples thereof include mannitol, sorbitol, salt, glycine, ammonium acetate, or a water-soluble protein that can be injected into a body fluid without substantially exhibiting pharmacological activity. Examples of the glycosaminodalican include hyaluronic acid, chondroitin, chondroitin sulfate A, chondroitin sulfate C, dermatan sulfate, heparin, heparan sulfate, and keutane sulfate. Examples of the polysaccharide include an acidic polysaccharide such as arginic acid.
上記水溶性蛋白としては、 水、 生理食塩水または緩衝液に溶解するものであれ ばよく、 例えばヒト血清アルブミン、 ヒト血清グロブリン、 コラーゲン、 ゼラチ ンなどがあげられる。 上記 p H調節剤としては、 たとえばグリシン、 酢酸アンモ 二ゥム、 クェン酸、 塩酸、 水酸化ナトリウムなどがあげられる。 上記局所麻酔剤 としては、 例えばクロロブタノ一ル、 塩酸キシロカインなどがあげられる。 上記 抗生物質としては、 たとえばゲンタマイシンなどがあげられる。 上記溶解補助剤 としては、例えばグリセリン、ポリエチレングリコール 4 0 0などがあげられる。 上記等張化剤としては、 たとえばマンニトール、 ソルビトール、 塩化ナトリウム などがあげられる。 上記吸着防止剤としては、 たとえばポリオキシエチレンソル ビ夕ンモノォレエ一トなどがあげられる。 The above-mentioned water-soluble protein may be any protein which is soluble in water, physiological saline or buffer, and includes, for example, human serum albumin, human serum globulin, collagen, gelatin and the like. Examples of the pH adjuster include glycine, ammonium acetate, citrate, hydrochloric acid, sodium hydroxide and the like. Examples of the local anesthetic include chlorobutanol and xylocaine hydrochloride. Examples of the antibiotic include gentamicin and the like. Examples of the solubilizer include glycerin and polyethylene glycol 400. Examples of the tonicity agent include mannitol, sorbitol, sodium chloride and the like. Examples of the above-mentioned adsorption inhibitor include polyoxyethylene sorbitol monooleate.
さらに分散媒中に水溶性蛋白を含有する場合の該水溶性蛋白の含有量としては、 一回投与の製剤あたり、好ましくは 0 . 0 5ないし 5 O m g ,さらに 0 . 5ないし 2 O m gが好ましく、 より好ましくは 0 . 7 5ないし 1 O m gである。該製剤は、 リ ン酸またはその塩 (例えば、 リン酸ナトリウム、 リン酸カリウム等) を含有して いてもよい。 Further, when the water-soluble protein is contained in the dispersion medium, the content of the water-soluble protein is preferably 0.05 to 5 Omg, more preferably 0.5 to 2 Omg, per one administration of the preparation. Preferably, it is more preferably 0.75 to 1 O mg. The formulation is Acid or a salt thereof (eg, sodium phosphate, potassium phosphate, etc.).
注射剤にリン酸またはその塩が含まれる場合、 その注射剤中のリン酸ナトリウ ムあるいはリン酸カリウムの濃度は約 0 . I mMないし 5 0 O mMであり、約 l m Mないし 1 0 O mMのときが好ましい。 ' 無菌製剤にするには、製造全工程を無菌にする方法、ガンマ線で滅菌する方法、 防腐剤を添加する方法等が挙げられるが、 特に限定されない。 When phosphoric acid or a salt thereof is contained in an injection, the concentration of sodium phosphate or potassium phosphate in the injection is about 0.1 mM to 50 mM, and about lmM to 10 mM. Is preferred. 'Aseptic preparations include, but are not particularly limited to, a method of sterilizing the entire production process, a method of sterilizing with gamma rays, and a method of adding a preservative.
本発明の剤は、 優れた軟骨破壊抑制作用 (軟骨基質分解酵素産生抑制剤および プロテオダリカン分解抑制剤)を有するので、軟骨疾患(例えば、変形性関節症、 変形性膝関節炎、 慢性関節リウマチ、 およびそれらの類似疾患における関節軟骨 の破壊 (軟骨破壊に起因する疾患) 等) の予防、 治療に用いることができる。 本発明の剤は、 他の関節疾患治療剤と共に用いることもできる。 例えば、 化合 物 (I ) またはその塩またはそのスルホキシドが軟骨破壊抑制剤 (軟骨基質分解 酵素産生抑制剤およびプロテオダリカン分解抑制剤) として用いられる場合、 他 の関節疾患治療剤と組み合わせて用いることもできる。 該組み合される薬剤とし ては、 抗炎症ステロイド剤 (例、 プレドニゾロン、 ヒドロコルチゾン、 メチルプ レドニゾロン、 デキサメタゾン、 ベ夕メタゾン等) 、 非ステロイド性消炎鎮痛剤 Since the agent of the present invention has an excellent cartilage destruction inhibitory activity (cartilage matrix degrading enzyme production inhibitor and proteodalican degradation inhibitor), it can be used for cartilage diseases (eg, osteoarthritis, osteoarthritis of the knee, rheumatoid arthritis). And the destruction of articular cartilage (disease caused by cartilage destruction) and the like in the similar diseases. The agent of the present invention can be used together with other therapeutic agents for joint diseases. For example, when Compound (I) or its salt or its sulfoxide is used as a cartilage destruction inhibitor (a cartilage matrix degrading enzyme production inhibitor and a proteodalican degradation inhibitor), it should be used in combination with other therapeutic agents for joint diseases. Can also. Drugs to be combined include anti-inflammatory steroids (eg, prednisolone, hydrocortisone, methylprednisolone, dexamethasone, vesamethasone, etc.), non-steroidal anti-inflammatory analgesics
(例、 インドメタシン、 ジクロフエナク、 ロキソプロフェン、 イブプロフェン、 アスピリン、 ピロキシカム、 スリンダク等) あるいはヒアルロン酸製剤 (例、 ヒ アルロン酸ナトリウム等) 、 C O X— I I阻害剤 (例、 セレコキシブ、 レフェコ キシブ等) 、 抗サイト力イン高分子製剤 (例、 レミケイド、 ェンブレル、 キネレ ット等) などが挙げられる。 (Eg, indomethacin, diclofenac, loxoprofen, ibuprofen, aspirin, piroxicam, sulindac, etc.) or hyaluronic acid preparations (eg, sodium hyaluronate, etc.), COX-II inhibitors (eg, celecoxib, lefecoxib, etc.) In-polymer preparations (eg, Remicade, Enbrel, Kinneret, etc.) and the like.
本発明の剤は、 軟骨疾患の予防 ·治療、 軟骨組織の維持等に合致した、 安全で 効能の高い製剤として用いられる。 例えば軟骨破壊抑制作用を局所において効率 的に発揮させ、 関節軟骨の疲弊あるいは破壊による疼痛などにより日常生活に支 障をきたしている患者のクォリティーォブライフを改善することが可能である。 化合物 (I ) またはその塩またはそのスルホキシドを、 軟骨疾患の予防治療剤 として使用する場合、その 1日当たりの投与量は、患者の状態や体重、投与方法、 製剤からの放出速度により異なるが、 経口投与の場合、 成人 (体重 5 O k g ) 1 人当たり化合物 (I) またはその塩またはそのスルホキシドとして約 5から約 2 000mg、 好ましくは約 30から約 50 Omgであり、 1から 3回にわけて投 与する。 The agent of the present invention is used as a safe and highly efficacious preparation suitable for prevention and treatment of cartilage disease, maintenance of cartilage tissue, and the like. For example, it is possible to efficiently exert the inhibitory effect on cartilage destruction locally, and to improve the quality of life of patients who are impaired in daily life due to fatigue due to joint cartilage or pain due to destruction. When compound (I) or a salt thereof or a sulfoxide thereof is used as a prophylactic / therapeutic agent for cartilage disease, the daily dose depends on the condition and weight of the patient, the method of administration, and the release rate from the preparation. Adult (weight 5 O kg) for administration 1 About 5 to about 2,000 mg, preferably about 30 to about 50 Omg, of the compound (I) or its salt or its sulfoxide per person, is administered in 1 to 3 divided doses.
非経口投与の場合、 成人 (体重 5 Okg) 1人当たり化合物 (I) またはその 塩またはそのスルホキシドとして約 1から約 50 Omg、 好ましくは約 5から約 300mgであり、 1から 3回にわけて投与する。 In the case of parenteral administration, about 1 to about 50 mg, preferably about 5 to about 300 mg of compound (I) or its salt or its sulfoxide per adult (body weight 5 Okg) per person, administered in 1 to 3 doses I do.
本発明の医薬組成物が徐放性製剤である場合の投与量は、 化合物 (I) または その塩またはそのスルホキシドの種類と含量、 剤形、'薬物放出の持続時間、 投与 対象動物 (例、 ヒト、 ラット、 マウス、 ネコ、 ィヌ、 ゥサギ、 牛、 豚等の哺乳動 物)、投与目的により種々異なるが、例えば非経口投与により適用する場合には、 1週間に約 0. 1から約 10 Omgの化合物(I)またはその塩またはそのスルホ キシドが投与製剤から放出されるようにすればよい。 When the pharmaceutical composition of the present invention is a sustained-release preparation, the dose may be determined according to the type and content of compound (I) or a salt thereof or a sulfoxide thereof, the dosage form, the duration of drug release, the animal to be administered (eg, Humans, rats, mice, cats, dogs, puppies, cattle, pigs, and other mammals) vary depending on the purpose of administration, but when applied by parenteral administration, for example, from about 0.1 to about 10 Omg of compound (I) or a salt thereof or a sulfoxide thereof may be released from the administration preparation.
本発明の医薬組成物は低毒性で安全に使用することができる。 例えば、 化合物 を 30 OmgZkg/日の用量でラットに経口投与しても何等異常所見は認めら れなかった。 特に以下に示す実施例化合物は、 経口投与されたときの吸収性にお いて優れているので、 経口用製剤のために有利に使用できる。 The pharmaceutical composition of the present invention has low toxicity and can be used safely. For example, when the compound was orally administered to rats at a dose of 30 OmgZkg / day, no abnormal findings were observed. In particular, the following example compounds are excellent in absorbability when administered orally, and can be advantageously used for oral preparations.
以下に実施例を挙げて本発明を更に詳しく説明するが、 これによつて本発明が 限定されるものではない。 尚、 溶液におけるパーセント (%) は、 特に断りのな い限り、 重量 Z容量パーセントを表示する。 また、 溶媒の混合比率は、 特に断り のない限り、 容量比を表示する。 1Hおよび 1 3 C核磁気共鳴 (NMR) スペクトル は内部標準としてテトラメチルシランを用いて D P X— 3 0 0型あるいは A C— 3 0 0型スペクトルメ一夕一(ブル力一社製、 ドイツ)、 ジェミニ 200型あるいは マーキュリー 300型スペクトルメ一ター (バリアン社製、 米国) で測定し、 ± 6 値を ppmで示した。 実施例の lH- NMRスぺクトル中、 sは 1重線、 dは 2重線、 t は 3重線、 Qは 4重線、 quintは 5重線、 s ixtetは 6重線、 septは 7重線、 mは 多重線、 bまたは brは幅広線をそれぞれ意味する。 またこれらは例えば、 ddは 2 重 2重線、 dddは 2重 2重 2重線、 dtは 2重 3重線、 dqは 2重 4重線、 brd Φ畐広 2重線、 のように組み合わせて表示されることがある。 また結合定数 ひ) は Hz で示した。 また13 C- NMRスペクトル中、 CH3はメチル炭素、 CH2はメチレン炭素、 CH はメチン炭素、 Q は 4級炭素をそれぞれ意味する。 測定溶媒の重クロ口ホルムは CDC13と、 重ジメチルスルホキシドは DMS0_dsと略すことがある。 赤外吸光スぺク トル(IR)は SHIMADZU FTIR-8200PCで測定した。 質量分析のうち FAB-MSは JE0L JMS-AX505W (日本電子製、 日本)を、 SIMSは日立 D- 80B型 GC 2重収束質量分析計ま たは日立 M- 2000マススぺクトロメーター(日立製作所、日本)を用いて測定した。 融点 (mp. ) はャナコ機器開発研究所製融点測定器 MP- J8を用いて測定した。 また、 参考例および実施例中、 室温とは 0 °Cないし 3 O t:を示す。 Hereinafter, the present invention will be described in more detail with reference to Examples, but the present invention is not limited thereto. In addition, the percentage (%) in the solution is expressed by weight Z volume percent unless otherwise specified. In addition, the mixing ratio of the solvent is indicated by the volume ratio unless otherwise specified. The 1H and 13 C nuclear magnetic resonance (NMR) spectra were obtained using DPX-300 or AC-300 spectra using tetramethylsilane as an internal standard (Bulliki, Germany), The values were measured with a Gemini 200 or Mercury 300 spectrum meter (Varian, USA) and ± 6 values are shown in ppm. During l H- NMR scan Bae spectrum of Example, s is singlet, d is doublet, t is triplet, Q is quartet, quint the quintet, s Ixtet the sextet, sept Represents a sevenfold line, m represents a multiple line, and b or br represents a wide line, respectively. Also, for example, dd is a double double line, ddd is a double double double line, dt is a double triple line, dq is a double quadruple line, brd Φ 畐 wide double line, and so on. They may be displayed in combination. Coupling constants are shown in Hz. In the 13 C-NMR spectrum, CH 3 means methyl carbon, CH 2 means methylene carbon, CH means methine carbon, and Q means quaternary carbon. Heavy black port Holm measurement solvent and CDC1 3, deuterated dimethyl sulfoxide is sometimes abbreviated as DMS0_d s. The infrared absorption spectrum (IR) was measured with SHIMADZU FTIR-8200PC. Among mass spectrometry, FAB-MS is JE0L JMS-AX505W (manufactured by JEOL, Japan), and SIMS is Hitachi D-80B GC double focusing mass spectrometer or Hitachi M-2000 mass spectrometer (Hitachi, Ltd.). Japan). The melting point (mp.) Was measured using a melting point analyzer MP-J8 manufactured by Yanako Instruments Development Laboratory. In the reference examples and examples, room temperature refers to 0 ° C. to 3 Ot :.
なお参考例および実施例中以下のような略号を用いることがある. The following abbreviations may be used in Reference Examples and Examples.
ADAN (2-amino-3, 3-dichloroacryloni tri le) = 2-ァミノ- 3, 3-ジクロロアクリロ 二トリル ' ADAN (2-amino-3, 3-dichloroacrylonitrile) = 2-amino-3,3-dichloroacrylonitrile ''
ADDP (1, 1, - (azodicarbonyl) dipiperidine) = 1, Γ - (ァゾジカルポニル)ジピぺ リジン ADDP (1, 1,-(azodicarbonyl) dipiperidine) = 1, Γ-(azodicarponyl) dipiperidine
Boc (tert-butoxycarbonyl) - tert -ブトキシカルポニル Boc (tert-butoxycarbonyl)-tert-butoxycarbonyl
CSA (D_camphor- 10- sul fonic acid) = D-カンファ一- 10-スルホン酸 CSA (D_camphor-10-sul fonic acid) = D-camphor-10-sulfonic acid
DAST (diethyl aminosul fur tri f luoride) = 3フッ化ジェチルァミノ硫黄 Deoxo-Fluor™ (bis [(2-methoxylet yl) amino] sulfur trif luoride)= 3 フッ化 [(2-メ卜キシェチル)ァミノ]硫黄 DAST (diethyl aminosul fur tri f luoride) = getylamino sulfur trifluoride Deoxo-Fluor ™ (bis [(2-methoxyletyl) amino] sulfur trif luoride) = trifluoride [(2-methoxylethyl) amino] sulfur
DEAD (diethyl azodicarboxylate)= ァゾジ力ルポン酸ジェチル DEAD (diethyl azodicarboxylate) =
DHP (3, 4-dihydro-2H-pyran)= 3, 4-ジヒドロ- 2H-ピラン DHP (3, 4-dihydro-2H-pyran) = 3,4-dihydro-2H-pyran
DIAD (diisopropyl azodicarboxylate)= ァゾジ力ルポン酸ジイソプロピル DMAP (4-N, N-d i me t hy 1 am i nopyr i d i ne) = 4_N, N—ジメチレアミノピリジン DIAD (diisopropyl azodicarboxylate) = diisopropyl azodicarboxylate DMAP (4-N, N-dimethhy1 am i nopyr idine) = 4_N, N-dimethylaminopyridine
DMF (N, N-dimethylformamide)= Ν,Ν-ジメチルホルムアミド DMF (N, N-dimethylformamide) = Ν, Ν-dimethylformamide
DMSO (dimethylsulfoxide)= ジメチルスルホキシド DMSO (dimethylsulfoxide) = dimethylsulfoxide
HOBT (l-hydroxybenzotriazole)= 1 -ヒドロキシベンゾトリアゾ一ル HOBT (l-hydroxybenzotriazole) = 1-hydroxybenzotriazole
TFA (trifluoroacetic acid)= トリフルォロ酢酸 TFA (trifluoroacetic acid) = trifluoroacetic acid
THF (tetrahydrofuran)=テトラヒドロフラン THF (tetrahydrofuran) = tetrahydrofuran
p-TsOH (p-toluenesulfonic acid)= p -トルエンスルホン酸 p-TsOH (p-toluenesulfonic acid) = p-toluenesulfonic acid
WSC (l-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride) =1-ェチ ル -3 -(3 -ジメチルァミノプロピル)カルポジイミド塩酸塩 WSC (l-ethyl-3- (3-dimethylaminopropyl) carbodiimide hydrochloride) = 1-ethyl-3- (3-dimethylaminopropyl) carbodiimide hydrochloride
bp. (boiling point) =沸点 bp. (boiling point) = boiling point
quant, (quant i tat ive)=定量的収率 実施例 quant, (quant i tat ive) = quantitative yield
参考例 1-1 Reference example 1-1
式 (I I) において R1Qがー ZR15 (Zが S02、 R15が p-トルィル基) である 化合'物の合成 Synthesis of R 1Q gar ZR 15 (Z is S0 2, R 15 is p- Toruiru group) compound 'compound in the formula (II)
ADAN (31.9 g, 0.233 モル、 純正化学社製) をジェチルエーテル (300 ml、 以下エーテルと略すことがある。) に溶解し、 室温で撹拌しながら P-トルエンス ルホニルイソシァネート (36.0 ml, 純度 98%、 東京化成社製) を滴下後、 室温 で 14時間撹拌した。 生成した沈澱物を濾取し、 ジェチルェ一テルで洗浄後、 乾 燥して標題化合物の淡黄色の粉末 (74.3 g、 0.222モル) を得た。 ADAN (31.9 g, 0.233 mol, manufactured by Junsei Chemical Co., Ltd.) is dissolved in getyl ether (300 ml, hereinafter abbreviated as ether), and stirred at room temperature while stirring with P-toluenesulfonyl isocyanate (36.0 ml). , Purity 98%, manufactured by Tokyo Chemical Industry Co., Ltd.), and the mixture was stirred at room temperature for 14 hours. The resulting precipitate was collected by filtration, washed with diethyl ether, and dried to give a pale yellow powder of the title compound (74.3 g, 0.222 mol).
'H-NMR (300 MHz, 重クロ口ホルム-重ジメチルスルホキシド =9: 1中) : δ 2.44 (3Η, s), 7.34 (2Η, d, J= 8.2 Hz), 7.90 (2H, d, J= 8.2 Hz), 8.19 (1H, brs), ca. 10.2 (1H, br). 13C-NMR (75 MHz, 重クロ口ホルム-重ジメチルスルホキシド =9 : 1中): δ 21.6 (CH3) , 110.4 (Q), 111.3 (Q), 127.8 (Q), 127.9 (2CH) , 129.6 (2CH), 136.2 (Q), 144.8 (Q), 147.9 (Q). 'H-NMR (300 MHz, double-mouthed form-deuterated dimethyl sulfoxide = 9: 1): δ 2.44 (3Η, s), 7.34 (2Η, d, J = 8.2 Hz), 7.90 (2H, d, J = 8.2 Hz), 8.19 (1H, brs), ca. 10.2 (1H, br). 13 C-NMR (75 MHz, double-mouthed form-deuterated dimethyl sulfoxide = 9: 1 in): δ 21.6 (CH3), 110.4 (Q), 111.3 (Q), 127.8 (Q), 127.9 (2CH), 129.6 (2CH), 136.2 (Q), 144.8 (Q), 147.9 (Q).
元素分析値: CUH9N303SC12 として計算値: C, 39.54; H, 2.71; N, 12.57; S, 9.60; Cl, 21.22 (%), 実測値; C, 39.62; H, 2.48; N, 12.66; S, 9.63; C1, 21.00 (%) . Elemental analysis: C U H 9 N 3 0 3 SC 12 Calculated: C, 39.54; H, 2.71; N, 12.57; S, 9.60; Cl, 21.22 (%), found; C, 39.62; H, 2.48; N, 12.66; S, 9.63; C1, 21.00 (%).
参考例 1-2 Reference example 1-2
式 (I I) において R10がー ZR15 (Zが S02、 R 15が p-クロ口フエニル基) である化合物の合成 Synthesis of R 10 Gar ZR 15 (Z is S0 2, R 15 is p- black port phenyl group) compound which is in the formula (II)
ADAN (11.2 g, 82.0 imol) をジェチルェ一テル (120 ml) に溶解し、 室温 で撹拌しながら P-クロ口ベンゼンスルホ二ルイソシァネート (20 g, 純度 85%、 アルドリッチ社製) を約 10分かけて滴下する。 さらに室温で 14時間撹拌し、生 成した沈澱物を濾取し、 ジェチルェ一テルで洗浄後、 乾燥して標題化合物の淡黄 色の粉末 (27.3g、 76.8 mmol) を得た。 Dissolve ADAN (11.2 g, 82.0 imol) in getyl ether (120 ml) and add benzenesulfonyl isocyanate (20 g, 85% pure, Aldrich) for about 10 minutes while stirring at room temperature. And drop it. The mixture was further stirred at room temperature for 14 hours, and the formed precipitate was collected by filtration, washed with diethyl ether, and dried to obtain a pale yellow powder of the title compound (27.3 g, 76.8 mmol).
Ή-NMR (300 MHz, 重クロロホルム-重ジメチルスルホキシド =5: 1中): δ 7.53 (2Η, ddm, J= 8.8, 4.4 Hz), 7.97 (2H, ddm, J= 8.8, 5.2 Hz), 8.36 (1H, brs), 11.01 (1H, brs). Ή-NMR (300 MHz, deuterated chloroform-deuterated dimethyl sulfoxide = 5: 1): δ 7.53 (2Η, ddm, J = 8.8, 4.4 Hz), 7.97 (2H, ddm, J = 8.8, 5.2 Hz), 8.36 (1H, brs), 11.01 (1H, brs).
以下に参考例 1- l、l-2で合成した化合物と同様にして合成した化合物の一覧を 示す。 The following is a list of compounds synthesized in the same manner as the compounds synthesized in Reference Examples 1-1 and 1-2.
〔表 1〕 〔table 1〕
( I I ) (R "> = -ZR is) (I I) (R "> = -ZR is)
参考例ト 6 - -. ΓΠ : ί-Ι^ 式(I I) において R10が _ZR15 (∑が( 〇、 R 15がフエニル基) である化合 物の合成 Reference example 6--. ΓΠ: ί-Ι ^ Synthesis of a compound of the formula (II) wherein R 10 is _ZR 15 (∑ is (〇, R 15 is a phenyl group)
ADAN (19.4g)のジクロロメタン(400ml)溶液にベンゾィルイソシアナ一ト(20g) を加え とした後、塩化アルミニウム(18.9g)を三回に分けて加えた。同温にて 1 時間撹拌後、 水(150ml)を加え有機溶媒を留去した。 残った水層に酢酸ェチル (150ml)、 水(150ml)と 0.5Mクェン酸(150ml)を加え 5分撹拌した。 有機'溶媒を留 去後、 沈殿物をろ取し水で洗浄、 乾燥し 58.6gの無色粉末を得た。 Benzyl isocyanate (20 g) was added to a dichloromethane (400 ml) solution of ADAN (19.4 g), and aluminum chloride (18.9 g) was added in three portions. After stirring at the same temperature for 1 hour, water (150 ml) was added, and the organic solvent was distilled off. To the remaining aqueous layer were added ethyl acetate (150 ml), water (150 ml) and 0.5 M citrate (150 ml), and the mixture was stirred for 5 minutes. After the organic 'solvent was distilled off, the precipitate was collected by filtration, washed with water, and dried to obtain 58.6 g of a colorless powder.
得られた化合物をァセトン(400ml)に溶解し、炭酸力リゥム(39.2g)を加え 50分間 加熱還流した。室温に冷却後、水(400ml)を加え約 500mlの IN塩酸にて pH3とし た。 沈殿物をろ取し水(100ml)で洗浄、 乾燥して標題化合物(31.2g, 77%)を得た。 The obtained compound was dissolved in acetone (400 ml), carbonated lime (39.2 g) was added, and the mixture was heated under reflux for 50 minutes. After cooling to room temperature, water (400 ml) was added, and the mixture was adjusted to pH 3 with about 500 ml of IN hydrochloric acid. The precipitate was collected by filtration, washed with water (100 ml), and dried to give the title compound (31.2 g, 77%).
-腿 R (300 MHz, 重クロ口ホルム中): 57.49-7.56 (2H, m), 7.60-7.67 (1H, m), 8.05-8.10 (2H, m), 11.0 (1H, br s), 11.6 (1H, br s). -Thigh R (300 MHz, in double-mouthed holm): 57.49-7.56 (2H, m), 7.60-7.67 (1H, m), 8.05-8.10 (2H, m), 11.0 (1H, br s), 11.6 (1H, br s).
元素分析値:(^Η7Ν302(ΐ2 として計算値: C; 46.50, Η; 2.48, Ν; 14.79 (%)、 実 測値; C; 45.99, Η; 2.43, Ν; 14.66 (%) . Elemental analysis: (^ Η 7 Ν 3 0 2 (ΐ 2 Calculated: C; 46.50, Η; 2.48 , Ν; 14.79 (%), actual Hakachi; C; 45.99, Η; 2.43 , Ν; 14.66 ( %).
同様にして参考例卜 7、 1-8の化合物を合成した。 Similarly, the compounds of Reference Examples 7 and 1-8 were synthesized.
参考例 1-7 Reference Example 1-7
(式 (I I) において R10が— ZR15 (Ζが S〇2、 R15が 4_フエノキシフエ二 ル基である化合物)(In the formula (II), R 10 is —ZR 15 (compound in which Ζ is S〇 2 and R 15 is 4_phenoxyphenyl group)
- NMR (300 MHz, 重クロ口ホルム中) : <57.05 (2H, dt, J= 9.0, 2.5 Hz), 7.08 ' (2H, dq, J= 8.0, 1.9 Hz), 7.24 (1H, brt, J= 7.2 Hz), 7.42 (2H, brtt, J= 8.1, 1.9 Hz), 7.61 (1H, brs), 7.90 (2H, dt, J= 9.0, 2.3 Hz), 9.74 (1H, brs). 元素分析値: C1SH„N304SC12として計算値: C, 46.62; H, 2.69; N, 10.19; S, 7.78; Cl, 17.20 (%), 実測値; C, 46.42; H, 2.86; N, 9.97; S, 7.85; C1, 17.09 (%) · 参考例卜 8 -NMR (300 MHz, in double-mouthed form): <57.05 (2H, dt, J = 9.0, 2.5 Hz), 7.08 '(2H, dq, J = 8.0, 1.9 Hz), 7.24 (1H, brt, J = 7.2 Hz), 7.42 (2H, brtt, J = 8.1, 1.9 Hz), 7.61 (1H, brs), 7.90 (2H, dt, J = 9.0, 2.3 Hz), 9.74 (1H, brs). : C 1S H "N 3 0 4 SC1 2 calculated: C, 46.62; H, 2.69 ; N, 10.19; S, 7.78; Cl, 17.20 (%), Found; C, 46.42; H, 2.86 ; N , 9.97; S, 7.85; C1, 17.09 (%)
(式(I I) において R10がー ZR15 (Zが S〇2、 R15が 4-クロ口フエ二ル基) である化合物) (In the formula (II), R 10 is -ZR 15 (Z is S〇 2 , R 15 is a 4-chlorophenyl group))
Ή -腿 (200 MHz, 重ジメチルスルホキシド中) : <57.58 (1H, m), 7.68 (2H, d, J= Ή-thigh (200 MHz, in heavy dimethyl sulfoxide): <57.58 (1H, m), 7.68 (2H, d, J =
4.8 Hz), 8.15 (1H, d, J= 7.0 Hz), 11.28 (1H, brs), 11.90 (1H, br) . 4.8 Hz), 8.15 (1H, d, J = 7.0 Hz), 11.28 (1H, brs), 11.90 (1H, br).
元素分析値: CiAWClgとして計算値: C, 33.87; H, 1.71; N, 11.85; S, 9.04; CI, 29.99 ( ), 実測値; C, 34.09; H, 1.67; N, 11.80; S, 9.10; CI, 29.54Elemental analysis: Calculated as CiAWClg: C, 33.87; H, 1.71; N, 11.85; S, 9.04; CI, 29.99 (), observed; C, 34.09; H, 1.67; N, 11.80; S, 9.10; CI, 29.54.
(%) . (%).
参考例 2 - 1 Reference Example 2-1
式 (I I I) において R10が— ZR15 (Zが S〇2、 R15が p-トルィル基)、 R がェチル基である化合物の合成 Synthesis of a compound of the formula (III) wherein R 10 is —ZR 15 (Z is S〇 2 , R 15 is a p-tolyl group) and R is an ethyl group
参考例 1-1で合成した化合物 (30.8 g, 92.2mmol) および炭酸カリウム (29.3 g, 0.212 mol) にエタノール (310 ml) を加え、 室温で撹捽しながら 3_メルカプ トプロピオン酸ェチル (24.2 ml, 0.180 mol) を約 6時間かけて滴下し、 反応混 合物を室温で 12時間撹拌した。 反応液に 1N塩酸 (240 ml) を加えて 30分間 撹拌した。 混合物を濃縮してエタノールを除去し、 水 (300 ml) を加え、 生成し た沈澱物を濾取し、水で洗浄後、乾燥して標題化合物の淡黄色粉末(29.8 g、 56.3 imnol) を得た。 Ethanol (310 ml) was added to the compound synthesized in Reference Example 1-1 (30.8 g, 92.2 mmol) and potassium carbonate (29.3 g, 0.212 mol), and the mixture was stirred at room temperature while stirring with ethyl 3_mercaptopropionate (24.2 g). ml, 0.180 mol) was added dropwise over about 6 hours, and the reaction mixture was stirred at room temperature for 12 hours. 1N Hydrochloric acid (240 ml) was added to the reaction solution, and the mixture was stirred for 30 minutes. The mixture was concentrated to remove ethanol, water (300 ml) was added, the resulting precipitate was collected by filtration, washed with water, and dried to give a pale yellow powder of the title compound (29.8 g, 56.3 imnol). Obtained.
¾-NMR (300 MHz, 重クロ口ホルム中) : δ 1.25 (3Η, t, J= 7.1 Hz), L 27 (3H, t, J- 7.1 Hz), 2.43 (3H, s), 2.57 (2H, t, J= 7.3 Hz), 2.60'(2H, t, J= 6.7 Hz), 3.12 (2H, t, J= 6.7 Hz), 3.20 (2H, t, J= 7.3 Hz), 4.13 (2H, q, J= 7.1 Hz), 4.19 (2H, q, J= 7.1 Hz), 7.32 (2H, brd, J= 8.3 Hz), 7.89 (2H, brd, J= 8.3 Hz), 8.30 (1H, brs), 9.67 (1H, br). ¾-NMR (300 MHz, in double-mouthed form): δ 1.25 (3Η, t, J = 7.1 Hz), L 27 (3H, t, J- 7.1 Hz), 2.43 (3H, s), 2.57 (2H , t, J = 7.3 Hz), 2.60 '(2H, t, J = 6.7 Hz), 3.12 (2H, t, J = 6.7 Hz), 3.20 (2H, t, J = 7.3 Hz), 4.13 (2H, q, J = 7.1 Hz), 4.19 (2H, q, J = 7.1 Hz), 7.32 (2H, brd, J = 8.3 Hz), 7.89 (2H, brd, J = 8.3 Hz), 8.30 (1H, brs) , 9.67 (1H, br).
元素分析値: C21H„N307S3 として計算値: C, 47.62; H, 5.14; N, 7.93; S, 18.16 (%), 実測値; C, 47.51; H, 4.84; N, 8.19; S, 18.12 (%) . Elemental analysis: C 21 H と し て N 3 0 7 S 3 Calculated: C, 47.62; H, 5.14; N, 7.93; S, 18.16 (%), found; C, 47.51; H, 4.84; N, 8.19; S, 18.12 (%).
参考例 2-2 Reference Example 2-2
式 (I I I) において R1Qが水素原子、 Rがェチル基である化合物の合成 参考例 2-1で合成した化合物 (6.96 g, 純度 90%、 13.1 mmol) にエタノール (310 ml) を加え、 撹拌しながらギ酸アンモニゥム (3.93 g, 59.1 mmol) を加え て 80 で 12時間撹拌した。 混合物を濃縮してエタノールを除去後、 エーテル (300 ml) で希釈し、 0.3N塩酸 (100 ml) で 3回抽出した。 抽出水層を炭酸水 素ナトリウムを加えて pH 8に調整後、 酢酸ェチル (200 mlおよび 150 ml) で抽 出した。 酢酸ェチル層を飽和炭酸水素ナトリウム水溶液および飽和食塩水で洗浄 後、 硫酸ナトリウムで乾燥し、 減圧下に濃縮乾固した。 残渣にェ一テルを加え、 生成した沈澱物を濾取し、 エーテルで洗浄後、 乾燥して標題化合物の淡黄色粉末 (3.37 g、 9.00 mmol) を得た。 Synthesis of Compound (III) wherein R 1Q is a hydrogen atom and R is an ethyl group To the compound (6.96 g, purity 90%, 13.1 mmol) synthesized in Reference Example 2-1 was added ethanol (310 ml) and stirred. While adding ammonium formate (3.93 g, 59.1 mmol), the mixture was stirred at 80 for 12 hours. After the mixture was concentrated to remove ethanol, the mixture was diluted with ether (300 ml) and extracted three times with 0.3N hydrochloric acid (100 ml). The extracted aqueous layer was adjusted to pH 8 by adding sodium hydrogen carbonate, and extracted with ethyl acetate (200 ml and 150 ml). The ethyl acetate layer was washed with a saturated aqueous sodium hydrogen carbonate solution and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure. Ether was added to the residue, and the resulting precipitate was collected by filtration, washed with ether and dried to give a pale yellow powder of the title compound (3.37 g, 9.00 mmol) was obtained.
¾-NMR (200 MHz, 重クロ口ホルム中): <5 1.26 (3H, t, J= 7.2 Hz), 1.27 (3H, t, J= 7.2 Hz), 2.60 (2H, t, J= 7.0 Hz), 2.63 (2H, t, J= 7.0 Hz), 3.13 (2H, t, J= 6.9 Hz), 3.14 (2H, t, J= 7.0 Hz), 4.16 (2H, q, J= 7.1 Hz), 4.17 (2H, q, J= 7.4 Hz), 7.78 (1H, brs), 7.97 (1H, brs). ¾-NMR (200 MHz, in double-mouthed form): <5 1.26 (3H, t, J = 7.2 Hz), 1.27 (3H, t, J = 7.2 Hz), 2.60 (2H, t, J = 7.0 Hz) ), 2.63 (2H, t, J = 7.0 Hz), 3.13 (2H, t, J = 6.9 Hz), 3.14 (2H, t, J = 7.0 Hz), 4.16 (2H, q, J = 7.1 Hz), 4.17 (2H, q, J = 7.4 Hz), 7.78 (1H, brs), 7.97 (1H, brs).
元素分析値: C14H21N305S2 として計算値: C, 44.78; H, 5.64; N, 11.19; S, 17.08Elemental analysis: C 14 H 21 N 3 0 5 S 2 Calculated: C, 44.78; H, 5.64 ; N, 11.19; S, 17.08
(%), 実測値; C, 44.67; H, 5.49; N, 11.39; S, 17.11 (%) . ' 融点: 150.5- 152.5°C (%), Found; C, 44.67; H, 5.49; N, 11.39; S, 17.11 (%). 'Melting point: 150.5-152.5 ° C
参考例 2-3 Reference example 2-3
式(I I I)において R10が— ZR15(Zが S〇2、 R15が p-ブロモフエニル基)、In the formula (III), R 10 is —ZR 15 (Z is S〇 2 , R 15 is a p-bromophenyl group),
Rがェチル基である化合物の合成 Synthesis of compounds in which R is an ethyl group
参考例 2-2で合成した化合物 (12.8 g, 34.3 mmol) をテトラヒドロフラン (250 ml、 以下 THFと略すことがある。) に溶解し、 トリェチルァミン (14.3ml, 0.103 mol) および塩化 p_ブロモベンゼンスルホニル (9.84 g, 37.7腿01) を加えて室 温で 23時間撹拌した。 この間、 反応開始より 1 6時間後に N,N -ジメチルァミノ ピリジン (0.21 g, 1.72 mmol) を、 および 1 8時間後に塩化 p-ブロモベンゼン スルホニル (0.89 g, 3.43 mmol) を加えた。 反応液を濃縮後、 残渣を酢酸ェチル (500 ml)で希釈し、 0. 3N塩酸、 0. 1N塩酸、水、および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去し、 残渣にエーテルおよび酢酸 ェチルを加え、 生成した沈澱物を濾取し、 エーテルで洗浄後、 乾燥して標題化合 物の黄色粉末 (14.9 g、 25.1 匪 ol) を得た。 The compound synthesized in Reference Example 2-2 (12.8 g, 34.3 mmol) was dissolved in tetrahydrofuran (250 ml, hereinafter sometimes abbreviated as THF), and triethylamine (14.3 ml, 0.103 mol) and p_bromobenzenesulfonyl chloride were dissolved. (9.84 g, 37.7 thighs 01) was added, and the mixture was stirred at room temperature for 23 hours. During this period, N, N-dimethylaminopyridine (0.21 g, 1.72 mmol) was added 16 hours after the start of the reaction, and p-bromobenzenesulfonyl chloride (0.89 g, 3.43 mmol) was added 18 hours later. After concentration of the reaction solution, the residue was diluted with ethyl acetate (500 ml), washed with 0.3 N hydrochloric acid, 0.1 N hydrochloric acid, water, and saturated saline, and then dried over sodium sulfate. The solvent was distilled off under reduced pressure, ether and ethyl acetate were added to the residue, and the resulting precipitate was collected by filtration, washed with ether, and dried to give a yellow powder of the title compound (14.9 g, 25.1 ol). Obtained.
'H-NMR (200 MHz, 重クロ口ホルム中) : δ 1.25 (3Η, t, J= 7.2 Hz), 1.28 (3H, t, J= 7.1 Hz), 2.59 (2H, t, J= 7.2 Hz), 2.61 (2H, t, J= 6.4 Hz), 3.12 (2H, t, J= 6.6 Hz), 3.23 (2H, t, J= 7.2 Hz), 4.13 (2H, q, J= 7.1 Hz), 4.20 (2H, q, J= 7.2 Hz), 7.67 (2H, dt, J= 8.8, 2.2 Hz), 7.88 (2H, dt, J= 8.8, 2.0 Hz), 8.36 (1H, brs), 9.60 (1H, br). 'H-NMR (200 MHz, in double-mouthed form): δ 1.25 (3Η, t, J = 7.2 Hz), 1.28 (3H, t, J = 7.1 Hz), 2.59 (2H, t, J = 7.2 Hz) ), 2.61 (2H, t, J = 6.4 Hz), 3.12 (2H, t, J = 6.6 Hz), 3.23 (2H, t, J = 7.2 Hz), 4.13 (2H, q, J = 7.1 Hz), 4.20 (2H, q, J = 7.2 Hz), 7.67 (2H, dt, J = 8.8, 2.2 Hz), 7.88 (2H, dt, J = 8.8, 2.0 Hz), 8.36 (1H, brs), 9.60 (1H , br).
元素分析値: C20H24N3O7S3Br として計算値: C, 40.40; H, 4.07; N, 7.07; S, 16.18; Br, 13.44 (%)、 実測値; C, 40.33; H, 3.89; N, 6.99; S, 16.17; Br, 13.13 (%) . 融点: 118.0-118.5°C Elemental analysis: C 20 H 24 N 3 O 7 S 3 Br Calculated: C, 40.40; H, 4.07 ; N, 7.07; S, 16.18; Br, 13.44 (%), Found; C, 40.33; H , 3.89; N, 6.99; S, 16.17; Br, 13.13 (%). Melting point: 118.0-118.5 ° C
参考例 2-4 Reference Example 2-4
式(I I I)において R1()が _ZR15(Zが S〇2、R15が p-クロ口フエ二ル基)、 Rがェチル基である化合物の合成 ' Synthesis of a compound of the formula (III) wherein R 1 () is _ZR 15 (Z is S〇 2 , R 15 is a p-chlorophenyl group) and R is an ethyl group ′
参考例 2-3と同様にして標題化合物を得た。 The title compound was obtained in the same manner as in Reference Example 2-3.
'H-NMR (200 MHz, 重クロ口ホルム中) : (5 1.25 (3H, t, J= 7.0 Hz), 1.28 (3H, t, J= 7.1 Hz), 2.58 (2H, i, J= 7.1 Hz), 2.61 (2H, i, J= 6.6 Hz) , 3.13 (2H, t, J= 6.4 Hz),, 3.23 (2H, t, J= 7.2 Hz), 4.13 (2H, q, J= 7.1 Hz), 4.20 (2H, q, J= 7.1 Hz), 7.51 (2H, d, J= 8.6 Hz), 7.96 (2H, d, J= 8.8 Hz), 8.49 (1H, brs), 9.72 (1H, br). 'H-NMR (200 MHz, in double-mouthed form): (5 1.25 (3H, t, J = 7.0 Hz), 1.28 (3H, t, J = 7.1 Hz), 2.58 (2H, i, J = 7.1 Hz), 2.61 (2H, i, J = 6.6 Hz), 3.13 (2H, t, J = 6.4 Hz) ,, 3.23 (2H, t, J = 7.2 Hz), 4.13 (2H, q, J = 7.1 Hz) ), 4.20 (2H, q, J = 7.1 Hz), 7.51 (2H, d, J = 8.6 Hz), 7.96 (2H, d, J = 8.8 Hz), 8.49 (1H, brs), 9.72 (1H, br) ).
元素分析値: C20H24N307S3C1 として計算値: C, 43.67; H, 4.40; N, 7.64; S, 17.49; C1, 6.45 (%)、 実測値; C, 43.72; H, 4.39; N, 7.72; S, 17.52; C1, 16.33 (%) . 融点: 115.5- 117.0で Elemental analysis: C 20 H 24 N 3 0 7 S 3 C1 Calculated: C, 43.67; H, 4.40 ; N, 7.64; S, 17.49; C1, 6.45 (%), Found; C, 43.72; H , 4.39; N, 7.72; S, 17.52; C1, 16.33 (%). Melting point: 115.5-117.0
参考例 3-1 Reference example 3-1
式(I V)において R10が一 ZR15 (Zが S02、 R 15が p -クロ口フエニル基)、 Rがェチル基である化合物の合成 In the formula (IV), synthesis of a compound in which R 10 is one ZR 15 (Z is S 0 2 , R 15 is a p-chlorophenyl group) and R is an ethyl group
方法 A Method A
参考例 2-4で合成した化合物(1.35 g, 2.45匪 ol)および炭酸力リゥム(407 mg, 2.95匪 ol) にエタノール (27 ml) を加え、 80 °Cで 2時間撹拌した。 反応液を 濃縮後、 残渣を酢酸ェチルー THF (1:1, 60 ml) で希釈し、 0.1N塩酸 (60 ml) で洗浄した。 水層を酢酸ェチル (30ml) で抽出し、 有機層と合わせて飽和食塩水, で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去し、 残渣にメタノー ルを加え、 生成した沈澱物を濾取し、 メタノールで洗浄後、 乾燥して標題化合物 の黄褐色粉末 (588 mg、 1.24職 ol) を得た。 Ethanol (27 ml) was added to the compound synthesized in Reference Example 2-4 (1.35 g, 2.45 bandol) and carbonic acid rim (407 mg, 2.95 bandol), and the mixture was stirred at 80 ° C for 2 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate-THF (1: 1, 60 ml) and washed with 0.1N hydrochloric acid (60 ml). The aqueous layer was extracted with ethyl acetate (30 ml), combined with the organic layer, washed with brine, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, methanol was added to the residue, and the resulting precipitate was collected by filtration, washed with methanol, and dried to give a tan powder of the title compound (588 mg, 1.24 mmol). .
方法 B Method B
参考例卜 2で合成した化合物 (5.06 g, 14.3麵01) および炭酸カリウム (4.54 g, 32.8匪01) にエタノール (100 ml) を加え、 室温で撹拌しながら 3-メルカプ トプロピオン酸ェチル (3.84 ml, 28.5腿01) を滴下後、 80°Cで 4時間撹拌し た。 反応液に 1N塩酸 (37ml) を加えて混和し、 濃縮乾固後、 残渣を酢酸ェチル (200 ml) で希釈し、 水、 10%食塩水および飽和食塩水で洗浄後、 硫酸ナトリ ゥムで乾燥した。 溶媒を減圧下に溜去し、 残渣にメタノールを加え、 生成した沈 澱物を濾取し、メタノールで洗浄後、乾燥して標題化合物の黄褐色の粉末(7.9 g、 純度約 70%、 12.3 mmol) を得た。 To the compound synthesized in Reference Example 2 (5.06 g, 14.3 麵 01) and potassium carbonate (4.54 g, 32.8 band01), add ethanol (100 ml), and stir at room temperature while stirring ethyl 3-mercaptopropionate (3.84 g). Then, the mixture was stirred at 80 ° C for 4 hours. 1N Hydrochloric acid (37 ml) was added to the reaction mixture, mixed and concentrated to dryness. (200 ml), washed with water, 10% saline and saturated saline, and dried over sodium sulfate. The solvent was evaporated under reduced pressure, methanol was added to the residue, and the resulting precipitate was collected by filtration, washed with methanol, and dried to give a tan powder of the title compound (7.9 g, purity approximately 70%, 12.3 g). mmol).
Ή-NMR (200 MHz, 重ジメチルスルホキシド中): δ 1.18 (3Η, t, J=7.1Hz), 2.63 (2H, t, J= 6.9 Hz), 3.32 (2H, i, J= 7.1 Hz), 4.07 (2H, q, J= 7.1 Hz), 7.57 (2H, d, J= 8.4 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.31 (1H, brs), ca. 10.2 (1H, br). Ή-NMR (200 MHz, in heavy dimethyl sulfoxide): δ 1.18 (3Η, t, J = 7.1 Hz), 2.63 (2H, t, J = 6.9 Hz), 3.32 (2H, i, J = 7.1 Hz), 4.07 (2H, q, J = 7.1 Hz), 7.57 (2H, d, J = 8.4 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.31 (1H, brs), ca. 10.2 (1H, br ).
元素分析値: C!5H16N305S3C1 として計算値: C, 40.04; H, 3.58; N, 9.34; S, 21.38; C1, 7.88 (%), 実測値; C, 40.06; H, 3.61; N, 9.42; S, 21.26; C1, 7.76 (%) . 融点: 166.3-169.8°C Elemental analysis: Calculated as C 5 H 16 N 3 0 5 S 3 C1:! C, 40.04; H, 3.58; N, 9.34; S, 21.38; C1, 7.88 (%), Found; C, 40.06; H, 3.61; N, 9.42; S, 21.26; C1, 7.76 (%). Melting point: 166.3-169.8 ° C
参考例 3-2 Reference example 3-2
式 (I V) において R1()が— ZR15 (Zが S〇2、 R15が P-トルィル基)、 尺が ェチル基である化合物の合成 In the formula (IV), synthesis of a compound in which R 1 () is — ZR 15 (Z is S〇 2 , R 15 is a P-tolyl group), and the length is ethyl
参考例 1-1で合成した化合物 (45.5 g, 0.136 mol) および炭酸力リゥム (65.8 g, 0.476 mol) に N, N-ジメチルホルムアミド (450 ml、 以下 DMFと略すことがあ る。) を加え、 室温で撹拌しながら 3_メルカプトプロピオン酸ェチル (35.9 ml, To the compound synthesized in Reference Example 1-1 (45.5 g, 0.136 mol) and carbonated lime (65.8 g, 0.476 mol), N, N-dimethylformamide (450 ml, hereinafter sometimes abbreviated as DMF) was added. While stirring at room temperature, 3_ ethyl mercaptopropionate (35.9 ml,
0.272 mol) を約 2時間かけて滴下した。 反応混合物を室温で 12時間撹拌し、 さ らに 80°Cで 7時間撹拌した。 反応液を濃縮乾固後、 残渣に 2N塩酸 (340 ml) およびメタノール (100 ml) を加えて混和し、 生成した沈澱物を濾取し、 50% メタノール水およびエタノールで洗浄後、 乾燥して標題化合物の橙褐色の粉末 (55.3 g、 純度約 70%、 0.090モル) を得た。 0.272 mol) was added dropwise over about 2 hours. The reaction mixture was stirred at room temperature for 12 hours and further at 80 ° C. for 7 hours. After the reaction mixture was concentrated to dryness, 2N hydrochloric acid (340 ml) and methanol (100 ml) were added to the residue and mixed, and the resulting precipitate was collected by filtration, washed with 50% aqueous methanol and ethanol, and dried. An orange-brown powder of the title compound (55.3 g, purity about 70%, 0.090 mol) was obtained.
• Ή-NMR (300 MHz, 重ジメチルスルホキシド中): δ 1.18 (3Η, t, J=7.1Hz), 2.33• Ή-NMR (300 MHz, in heavy dimethyl sulfoxide): δ 1.18 (3Η, t, J = 7.1Hz), 2.33
(3H, s), 2.63 (2H, t, J= 7.1 Hz), 3.30 (1H, s), 3.32 (2H, t, J= 7.1 Hz), 4.07 (2H, q, J= 7.1 Hz), 7.28 (2H, d, J= 8.1 Hz), 7.74 (2H, d, J= 8.1 Hz), 8.75(3H, s), 2.63 (2H, t, J = 7.1 Hz), 3.30 (1H, s), 3.32 (2H, t, J = 7.1 Hz), 4.07 (2H, q, J = 7.1 Hz), 7.28 (2H, d, J = 8.1 Hz), 7.74 (2H, d, J = 8.1 Hz), 8.75
(1H, brd,' J- 2 Hz), 9.61 (1H, brs). (1H, brd, 'J-2 Hz), 9.61 (1H, brs).
参考例 3-3 Reference Example 3-3
式 (I V) において R10が— ZR15 (Zが CO、 R 15がフエニル基)、 Rがメチ ル基である化合物の合成 参考例 卜 6 で合成した化合物(2.85g)と 2-メルカプトプロピオン酸メチル (2.2ml) の DMF(50ml)溶液に炭酸カリウム(3.17g)を室温にて加え 1時間撹拌後、 70°Cにて 45分間撹拌した。沈殿物をろ別し、ろ液を減圧濃縮後酢酸ェチル (50ml) を加えた。 1N塩酸で pH 3とし、 生成した沈殿物をろ取し水 (30ml)で洗浄、 乾燥 して標題化合物 (3.27g, 90¾)を得た。 Synthesis of a compound of formula (IV) wherein R 10 is —ZR 15 (Z is CO, R 15 is a phenyl group) and R is a methyl group Reference Example To a solution of the compound synthesized in Example 6 (2.85 g) and methyl 2-mercaptopropionate (2.2 ml) in DMF (50 ml) was added potassium carbonate (3.17 g) at room temperature, and the mixture was stirred for 1 hour. And stirred for 45 minutes. The precipitate was separated by filtration, the filtrate was concentrated under reduced pressure, and ethyl acetate (50 ml) was added. The pH was adjusted to 3 with 1N hydrochloric acid, and the formed precipitate was collected by filtration, washed with water (30 ml), and dried to obtain the title compound (3.27 g, 90 °).
Ή-NMR (300 MHz, 重ジメチルスルホキシド中) : 52.77 (2H, t, J-7.0Hz), 3.52 (2H, t, J=7.0Hz), 3.63 (3H, s), 7.61-7.68 (2H, m), 7.70-7.77 (1H, m), 7.97-8.02 (2H, m), 11.0 (1H, s), 12.1 (1H, br s), 13.6 (1H, s). Ή-NMR (300 MHz, in heavy dimethyl sulfoxide): 52.77 (2H, t, J-7.0Hz), 3.52 (2H, t, J = 7.0Hz), 3.63 (3H, s), 7.61-7.68 (2H, m), 7.70-7.77 (1H, m), 7.97-8.02 (2H, m), 11.0 (1H, s), 12.1 (1H, br s), 13.6 (1H, s).
参考例 3-4 Reference Example 3-4
式(I V) において R10が— ZR15 (Zが S〇2、 R15が p-ブロモフエニル基)、 Rがェチル基である化合物の合成 Synthesis of a compound of the formula (IV) wherein R 10 is —ZR 15 (Z is S〇 2 , R 15 is a p-bromophenyl group) and R is an ethyl group
参考例 3 - 1と同様の方法に従って合成した。 Synthesized according to a method similar to that of Reference Example 3-1.
¾-NM (200 MHz, 重ジメチルスルホキシド中): δ 1.18 (3Η, t, J= 7.0Hz), 2.63 (2H, t, J= 7.0 Hz), 3.32 (2H, t, J= 7.10 Hz), 4.06 (2H, q, J= 7.1 Hz), 7.68 (2H, d, J= 8.4 Hz), 7.79 (2H, d, J= 8.4 Hz), 8.83 (1H, brs), 9.76 (1H, brs). 参考例 4 ¾-NM (200 MHz, in heavy dimethyl sulfoxide): δ 1.18 (3Η, t, J = 7.0 Hz), 2.63 (2H, t, J = 7.0 Hz), 3.32 (2H, t, J = 7.10 Hz), 4.06 (2H, q, J = 7.1 Hz), 7.68 (2H, d, J = 8.4 Hz), 7.79 (2H, d, J = 8.4 Hz), 8.83 (1H, brs), 9.76 (1H, brs). Reference example 4
メタンスルホン酸 インダン- 2-ィルエステル Methanesulfonic acid indane-2-yl ester
2 -インダノ一ル (1.00 g, 7.45 mmol) のテトラヒドロフラン (20.0 ml) 溶液 に、 メタンスルホニルクロリド(0.865 ml, 11.2讓01) とトリエチルァミン(2.08 ml, 14.9 mmol) を氷冷下順次加えた。 反応混合物を室温で 3時間かき混ぜた後、 IN塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (1.43 g, 90%) を無色結晶として得た。 To a solution of 2-indanol (1.00 g, 7.45 mmol) in tetrahydrofuran (20.0 ml) was added methanesulfonyl chloride (0.865 ml, 11.2 cas 01) and triethylamine (2.08 ml, 14.9 mmol) sequentially under ice cooling. . After stirring the reaction mixture at room temperature for 3 hours, IN hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (1.43 g, 90%) as colorless crystals.
赤外吸収スペクトル(IR) (KBr中) : 3069, 3027, 2965, 2936, 2911, 1481 cm一1.Infrared absorption spectrum (IR) (in KBr): 3069, 3027, 2965, 2936, 2911, 1481 cm- 1 .
'H-NMR (CDC13) : δ 3.02 (3H, s), 3.25 (2H, dd, J=16.8, 3.6 Hz), 3.38 (2H, dd, J=17.0, 6.0 Hz), 5.48-5.59 (1H, m), 7.17-7.29 (4H, m). 'H-NMR (CDC1 3) : δ 3.02 (3H, s), 3.25 (2H, dd, J = 16.8, 3.6 Hz), 3.38 (2H, dd, J = 17.0, 6.0 Hz), 5.48-5.59 (1H , m), 7.17-7.29 (4H, m).
参考例 5 Reference example 5
メタンスルホン酸 4, 4, 5, 5, 5-ペンタフルォロペンチルエステル Methanesulfonic acid 4,4,5,5,5-pentafluoropentyl ester
4, 4, 5, 5, 5-ペンタフルォ口-卜ペン夕ノール (1.00 g, 5.61 匪 ol) のテトラヒ ドロフラン(20.0ml)溶液に、メタンスルホニル クロリド(0.652 ml, 8.42 mmol) とトリエチルァミン (1.57 ml, 11.2 mmol) を氷冷下順次加えた。 反応混合物を 室温で 3時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水 洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (1.42 g, 99¾) を油 状物として得た。 Methanesulfonyl chloride (0.652 ml, 8.42 mmol) was added to a solution of 4,4,5,5,5-pentafluoroethanol (1.00 g, 5.61 ol) in tetrahydrofuran (20.0 ml). And triethylamine (1.57 ml, 11.2 mmol) were sequentially added under ice-cooling. After stirring the reaction mixture at room temperature for 3 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was water washed to give after drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (1.42 g, 99¾) as an oil-like product.
赤外吸収スペクトル(IR) (KBr中) : 3031, 2946, 912, 743 cm"1. Infrared absorption spectrum (IR) (in KBr): 3031, 2946, 912, 743 cm " 1 .
Ή-NMR (CDC13) : δ 1.98-2.35 (4Η, m), 3.05 (3H, s), 4.32 (2H, t, J=5.6 Hz). 参考例 6 Ή-NMR (CDC1 3): . Δ 1.98-2.35 (4Η, m), 3.05 (3H, s), 4.32 (2H, t, J = 5.6 Hz) Reference Example 6
5, 5, 6, 6, 6-ペン夕フルォ口へキサン二トリル 5, 5, 6, 6, 6-Penyu Fluorohexanenitrile
シアン化ナトリウム (5.03 g, 78.1画】) のジメチルスルホキシド (10.0 ml) の懸濁液に、参考例 5で得られたメタンスルホン酸 4, 4, 5, 5, 5-ペン夕フルォロぺ ンチルエステル (10.0 g, 39.0腿 ol) のジメチルスルホキシド (10.0 ml) 溶液 を加えた。反応混合物を室温で 17時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥 (MgS04)後、溶媒を減圧下に留去して題記化合物(5.25 g, 72%) を油状物として得た。 To a suspension of sodium cyanide (5.03 g, 78.1 fraction) in dimethyl sulfoxide (10.0 ml) was added the methanesulfonic acid 4,4,5,5,5-pentanofluoroethyl ester obtained in Reference Example 5. (10.0 g, 39.0 tmol) in dimethyl sulfoxide (10.0 ml) was added. After stirring the reaction mixture at room temperature for 17 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (5.25 g, 72%) as an oil.
赤外吸収スペクトル(IR) (KBr中) : 2959, 2251 cm-1. Infrared absorption spectrum (IR) (in KBr): 2959, 2251 cm- 1 .
Ή-N R (CDCI3) : δ 1.93-2.17 (2Η, m), 2.17-2.37 (2H, m), 2.51 (2H, t, J=7.0 Ή-NR (CDCI3): δ 1.93-2.17 (2Η, m), 2.17-2.37 (2H, m), 2.51 (2H, t, J = 7.0
Hz). Hz).
参考例 7 Reference Example 7
5, 5, 6, 6, 6 -ペン夕フルォ口へキサン酸 5,5,6,6,6-hexanoic acid
参考例 6で得られた 5, 5, 6, 6, 6-ペンタフルォ口へキサン二トリル(5.25 g, 28.1 mmol) を酢酸 (50.0ml) 及び濃塩酸 (25.0ml) に溶かし、 120°Cで 4時間かき混 ぜた。 反応混合物を氷水に加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (4.80 g, 83%) を油状物とし て得た。 The 5,5,6,6,6-pentafluoromouth hexanenitrile (5.25 g, 28.1 mmol) obtained in Reference Example 6 was dissolved in acetic acid (50.0 ml) and concentrated hydrochloric acid (25.0 ml), and the solution was heated at 120 ° C. Stir for 4 hours. The reaction mixture was added to ice water and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (4.80 g, 83%) was obtained as an oil.
赤外吸収スペクトル(IR) (KBr中) : 3700-2300, 1715 cm-1. Infrared absorption spectrum (IR) (in KBr): 3700-2300, 1715 cm- 1 .
Ή-NMR (CDCI3) : 51.86-2.04 (2H, m), 2.08-2.28 (2H, m), 2.50 (2H, t, J=7.0 Hz). Ή-NMR (CDCI3): 51.86-2.04 (2H, m), 2.08-2.28 (2H, m), 2.50 (2H, t, J = 7.0 Hz).
参考例 8 Reference Example 8
5, 5, 6, 6, 6-ペンタフルォ口-卜へキサノール 参考例 7で得られた 5, 5, 6, 6, 6 -ペン夕フルォ口へキサン酸 (4.80 g, 23.3匪01) のテトラヒドロフラン (50.0ml) 溶液に、 ォキサリル クロリド (2.68ml, 27.9 画 ol) と N,N-ジメチルホルムアミ ド (0.100ml) を順次加えた。 反応混合物を室 温で 4 時間かき混ぜた後、 溶媒を減圧下に留去した。 水素化ホウ素ナトリウム (1.76 g, 46.6 mmol) のテトラヒドロフラン (25.0 ml) 及び 1,2 -ジメトキシェ タン (20.0 ml) 溶液に、 残留物のテトラヒドロフラン (25.0 ml) 溶液を氷冷下 加えた。反応混合物を室温で 19時間かき混ぜた後、反応混合物を氷水に注ぎ、 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (3.23 g, 73%) を油状物として得た。 5,5,6,6,6-pentafluorohexanol To a solution of 5,5,6,6,6-pentanofluorhexaenoic acid (4.80 g, 23.3 maraudal 01) obtained in Reference Example 7 in tetrahydrofuran (50.0 ml) was added oxalyl chloride (2.68 ml, 27.9 ) And N, N-dimethylformamide (0.100 ml) were added sequentially. After the reaction mixture was stirred at room temperature for 4 hours, the solvent was distilled off under reduced pressure. To a solution of sodium borohydride (1.76 g, 46.6 mmol) in tetrahydrofuran (25.0 ml) and 1,2-dimethoxyethane (20.0 ml) was added a solution of the residue in tetrahydrofuran (25.0 ml) under ice-cooling. After stirring the reaction mixture at room temperature for 19 hours, the reaction mixture was poured into ice water, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), the solvent was evaporated reduced pressure the title compound (3.23 g, 73%) as an oil.
赤外吸収スペクトル(IR) (KBr中) : 3710-3050, 2953, 2884 cm"1. Infrared absorption spectrum (IR) (in KBr): 3710-3050, 2953, 2884 cm " 1 .
'Η - NMR (CDC13) : δ 1.36-1.80 (6Η, m), 1.92-2.22 (2H, m), 3.70 (2H, t, J=6.2 Hz). 'Η - NMR (CDC1 3) : δ 1.36-1.80 (6Η, m), 1.92-2.22 (2H, m), 3.70 (2H, t, J = 6.2 Hz).
参考例 9 Reference Example 9
メタンスルホン酸 5, 5, 6, 6, 6-ペンタフルォ口へキシルエステル Methanesulfonic acid 5,5,6,6,6-Pentafluorohexyl ester
参考例 8で得られた 5, 5, 6, 6, 6-ペン夕フルォロ-卜へキサノール(3.23 g, 16.8 腿 ol)のテトラヒドロフラン(60.0ml)溶液に、メタンスルホニル クロリ ド (1.95 ml, 25.2 mmol) とトリエチルァミン (4.69 ml, 33.6匪01) を氷冷下順次加えた。 反応混合物を室温で 2時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(4.13 g, 91¾) を油状物として得た。 Methanesulfonyl chloride (1.95 ml, 25. 2) was added to a solution of 5,5,6,6,6-pentafluorohexanol (3.23 g, 16.8 t) obtained in Reference Example 8 in tetrahydrofuran (60.0 ml). mmol) and triethylamine (4.69 ml, 33.6 maraudal 01) were sequentially added under ice cooling. After stirring the reaction mixture at room temperature for 2 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (4.13 g, 91¾) as an oil.
赤外吸収スペクトル(IR) (KBr中) : 3029, 2949, 1354, 1175, 974, 936 cm—1. Ή-NMR (CDC13) : δ 1.66-1.95 (4Η, m), 1.95-2.29 (2H, m), 3.03 (3H, s), 4.27 (2H, t, J=6.2 Hz). Infrared absorption spectrum (IR) (in KBr):. 3029, 2949, 1354, 1175, 974, 936 cm- 1 Ή-NMR (CDC1 3): δ 1.66-1.95 (4Η, m), 1.95-2.29 (2H , m), 3.03 (3H, s), 4.27 (2H, t, J = 6.2 Hz).
参考例 10 Reference Example 10
式(XVI I I) において R2がべンジル基、 Zが S02、 R15が p-トルィル基、 Rがェチル基である化合物の合成 R 2 Gabe Njiru group in formula (XVI II), the synthesis of Z is S0 2, R 15 is p- Toruiru group, and R is an Echiru group
参考例 3-2で合成した化合物 (1.09 g, 2.54腿01) をァセトニトリル (30 ml) に溶解し、 臭化べンジル (0.339 ml, 2.79删01) およびトリェチルァミン (1.06 ml, 7.61匪 ol) を加えて室温で 2時間半撹拌後、 .60 °Cで 19時間撹拌した。 こ の間 DMF (6 ml)、 臭化べンジル (0.339 ml, 2.79 miol) およびトリェチルアミ ン (1.06ml, 7.61顧 ol) を適宜加えた。 反応液を濃縮後、 残渣を酢酸ェチル (50 ml) で希釈し、 2%炭酸水素ナトリウム水溶液、 0.1N塩酸、 水および飽和食塩水 で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去し、 残渣をシリカゲ ル 60 (25g) のカラムクロマトグラフィーに付し、 酢酸ェチル:へキサン (4 0 : 60〜 50 : 50) の溶出画分を減圧下濃縮乾固し、 標題化合物の黄色油状 物 (0.95g, 1.83 mmol) を得た。 The compound synthesized in Reference Example 3-2 (1.09 g, 2.54 thigh 01) was dissolved in acetonitrile (30 ml), and benzyl dibromide (0.339 ml, 2.79 删 01) and triethylamine (1.06 ml, 7.61 bandol) were added. After stirring at room temperature for 2.5 hours, the mixture was further stirred at 60 ° C. for 19 hours. This During this period, DMF (6 ml), benzyl bromide (0.339 ml, 2.79 miol) and triethylamine (1.06 ml, 7.61) were added as appropriate. After concentrating the reaction solution, the residue was diluted with ethyl acetate (50 ml), washed with a 2% aqueous sodium hydrogen carbonate solution, 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. The solvent was evaporated under reduced pressure, and the residue was subjected to column chromatography on silica gel 60 (25 g). The fraction eluted with ethyl acetate: hexane (40:60 to 50:50) was concentrated to dryness under reduced pressure. As a result, a yellow oily substance (0.95 g, 1.83 mmol) of the title compound was obtained.
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 1.24 (3Η, t, J= 7.1 Hz), 2.43 (3H, s), 2.55 (2H, t, J= 7.3 Hz), 3.18 (2H, t, J= 7.3 Hz), 4.01 (2H, s), 4.13 (2H, q, J= 7.1 Hz), 7.32 (2H, d, J= 8.2 Hz), 7.89 (2H, d, J= 8.2 Hz), 7.97 (1H, brs), 9.76 (1H, br). 'H-NMR (300 MHz, in double-mouthed form): δ 1.24 (3Η, t, J = 7.1 Hz), 2.43 (3H, s), 2.55 (2H, t, J = 7.3 Hz), 3.18 (2H , t, J = 7.3 Hz), 4.01 (2H, s), 4.13 (2H, q, J = 7.1 Hz), 7.32 (2H, d, J = 8.2 Hz), 7.89 (2H, d, J = 8.2 Hz) ), 7.97 (1H, brs), 9.76 (1H, br).
参考例 Π Reference example Π
式 (X I X) において R2がべンジル基、 Zが S02、 R15が p-トルィル基、 R がェチル基である化合物の合成 Synthesis of Compound R 2 Gabe Njiru group, Z is S0 2, R 15 is p- Toruiru radical, R is a Echiru group in the formula (XIX)
,参考例 10で合成した化合物 (1.95 g, 3.75腿01) を THF (25 ml) およびメタ ノール (25 ml) に溶解し、 水酸化カリウム (0.99 g, 15.0 腿 ol) を加えて室温 で 40分間撹拌した。 反応液に 1N塩酸 (15 ml), 水 (25 ml) およびメタノール (10 ml) を加えて混和し、 室温で 30分間撹拌した。 生成した沈澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物の黄色粉末 (1.28g、 3.05画 ol) を 得た。 The compound synthesized in Reference Example 10 (1.95 g, 3.75 t1) was dissolved in THF (25 ml) and methanol (25 ml), and potassium hydroxide (0.99 g, 15.0 t) was added. Stirred for minutes. To the reaction solution, 1N hydrochloric acid (15 ml), water (25 ml) and methanol (10 ml) were added and mixed, followed by stirring at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give a yellow powder of the title compound (1.28 g, 3.05 mmol).
'H-NMR (300 MHz, 重クロ口ホルム中): <5 2.44 (3H, s), 4.53 (2H, s), 7.31 (5H, m), 7.34 (2H, d, J= 8.7 Hz), 7.78 (2H, d, J= 8.3 Hz), 8.12 (1H, brs) , 9.28 (1H, brs) , 12.54 (1H, brs). 'H-NMR (300 MHz, in double-mouthed form): <5 2.44 (3H, s), 4.53 (2H, s), 7.31 (5H, m), 7.34 (2H, d, J = 8.7 Hz), 7.78 (2H, d, J = 8.3 Hz), 8.12 (1H, brs), 9.28 (1H, brs), 12.54 (1H, brs).
元素分析値: C18H17N303S3として計算値: C, 51.53; H, 4.08; N, 10.02; S, 22.93 (%), 実測値; C, 51.53; H, 4.08; N, 10.02; S, 22.93 (%) . Elemental analysis: C 18 H 17 N 3 0 3 S 3 Calculated: C, 51.53; H, 4.08 ; N, 10.02; S, 22.93 (%), Found; C, 51.53; H, 4.08 ; N, 10.02; S, 22.93 (%).
参考例 12 Reference Example 12
式 (XXV I I) において R 14がエトキシカルポニルメチル基である化合物の合 成 Synthesis of a compound of the formula (XXV II) wherein R 14 is an ethoxycarbonylmethyl group
ADAN(10.8g、 79.1 mmol)をジクロロメタン(220 ml)に溶解し、 イソシアナ一ト 酢酸ェチル(10.0 g、 75.1塵 ol)を加え、 に冷却した後、塩化アルミニウム( 2 g, 75.1醒 ol)を数回に分けて加えた。 反応液を Otにて 3時間半撹拌後、 沈殿物 をろ過し、エーテル、氷冷水、および水で洗浄し、乾燥して淡黄色粉末(14.2 g, 46.6 mmol)を得た。 このようにして得た粉末(15.1 g、 56.4mmol)を酢酸ェチル(300 ml) に懸濁し、 1、リエチルァミン(15.6 ηιΚ 0.113 mol)を加えて、 室温で 12時間撹 拌した。 沈殿物をろ過し、 エーテルで洗浄し、 乾燥して標題化合物の灰白色粉末 (12.2 g, 45.8 匪 ol)を得た。 ADAN (10.8 g, 79.1 mmol) was dissolved in dichloromethane (220 ml), and isocyanate was dissolved. Ethyl acetate (10.0 g, 75.1 dust ol) was added, and the mixture was cooled to, and aluminum chloride (2 g, 75.1 ol) was added in several portions. After stirring the reaction solution at Ot for 3.5 hours, the precipitate was filtered, washed with ether, ice-cold water, and water, and dried to obtain a pale yellow powder (14.2 g, 46.6 mmol). The powder (15.1 g, 56.4 mmol) thus obtained was suspended in ethyl acetate (300 ml), 1, and lithiamine (15.6 ηιΚ 0.113 mol) was added thereto, followed by stirring at room temperature for 12 hours. The precipitate was filtered, washed with ether and dried to give an off-white powder of the title compound (12.2 g, 45.8 ol).
¾-NMR (300 MHz, 重ジメチルスルホキシド中) : δΐ.20 (3H, t, J= 7.1 Hz), 4.14 (2H, q, J= 7.1 Hz), 4.27 (2H, s), 8.74 (1H, brs). ¾-NMR (300 MHz, in heavy dimethyl sulfoxide): δΐ.20 (3H, t, J = 7.1 Hz), 4.14 (2H, q, J = 7.1 Hz), 4.27 (2H, s), 8.74 (1H, brs).
元素分析値: C8 N303C12 として計算値: C; 36.11 H; 3.41, N; 15.79; C1, 26.65 (%)、 実測値; C; 36.39 H; 3.41, ; 15.69; C1, 26.47 (%) . Elemental analysis: C 8 N 3 0 3 C1 2 Calculated: C; 36.11 H; 3.41, N; 15.79; C1, 26.65 (%), Found; C; 36.39 H; 3.41, ; 15.69; C1, 26.47 (%).
参考例 13-1 Reference Example 13-1
式 (XXV I I I) において R 14がエトキシカルボニルメチル基である化合物の 合成 Synthesis of compound of formula (XXVIII) wherein R 14 is an ethoxycarbonylmethyl group
参考例 12で合成した化合物 (14.7 g, 55.1 腿 ol) をエタノール (150 ml) に 溶解し、 1N塩酸 (150ml) を加え、 反応混合物を室温で 67時間撹拌した。 沈殿 物をろ過し、 水で洗浄して塩酸を除去し、 含水白色粉末を得た。 これをエタノー ル (200 ml) に懸濁し、 1N水酸化ナトリウム (100 ml) を加え、 室温で 1時間半 撹拌した。 反応液を濃縮してエタノールを留去し、 残水層に 1N塩酸 (100 ml) を加えて PH2に調整した。 生成した沈澱物を濾取し、 水で洗浄後、 乾燥して標題 化合物の白色粉末 (7.18 g、 30.0 腿 ol) を得た。 The compound synthesized in Reference Example 12 (14.7 g, 55.1 tmol) was dissolved in ethanol (150 ml), 1N hydrochloric acid (150 ml) was added, and the reaction mixture was stirred at room temperature for 67 hours. The precipitate was filtered, washed with water to remove hydrochloric acid, and a water-containing white powder was obtained. This was suspended in ethanol (200 ml), 1N sodium hydroxide (100 ml) was added, and the mixture was stirred at room temperature for 1.5 hours. The reaction solution was concentrated to distill off ethanol, and 1N hydrochloric acid (100 ml) was added to the remaining aqueous layer to adjust to PH2. The resulting precipitate was collected by filtration, washed with water, and dried to give the title compound as a white powder (7.18 g, 30.0 tmol).
'H-NMR (300 MHz, 重クロ口ホルム-重ジメチルスルホキシド, 9 : 1中) : δ 4.23 (2Η, s), 10.87 (1H, brs). 'H-NMR (300 MHz, double-mouthed form-deuterated dimethyl sulfoxide, in 9: 1): δ 4.23 (2Η, s), 10.87 (1H, brs).
元素分析値: C6H4N204C12 として計算値: C, 30.15; H, 1.69; N, 11.72; C1, 29.67 (%), 実測値; C, 30.32; H, 1.86; N, 11.73; C1, 29.66 ( ) . Elemental analysis: C 6 H 4 N 2 0 4 C1 2 Calculated: C, 30.15; H, 1.69 ; N, 11.72; C1, 29.67 (%), Found; C, 30.32; H, 1.86 ; N, 11.73; C1, 29.66 ().
参考例 13 - 2 Reference Example 13-2
式 (XXV I I I) において R14が tert-ブトキシカルポニルメチル基である化 合物の合成 Synthesis of a compound of the formula (XXVIII) wherein R 14 is a tert-butoxycarbonylmethyl group
参考例 13-1で合成した化合物 (7.25 g, 30.3 mmol) に酢酸 tert-ブチル (240 ml) を加えて溶解し、 3フッ化ホウ素エーテル錯体 (3.81 ml, 30.3 匪01) を加 えて室温で 7時間撹拌した。 反応液を氷冷水 (200 g) に注いで混合、 分配し、 水 層を酢酸ェチル(200ml)で抽出した。有機層を合わせて炭酸ナトリウム水溶液、 水および飽和食塩水で洗浄後、硫酸ナトリウムで乾燥し、減圧下に濃縮乾固した。 残渣にエーテルおよびジィソプロピルエーテルを加え、生成した沈澱物を濾取し、 ジイソプロピルエーテルで洗浄後、乾燥して標題化合物の白色粉末(6.17g、 20.9 腿 ol) を得た。 The compound synthesized in Reference Example 13-1 (7.25 g, 30.3 mmol) was added to tert-butyl acetate (240 ml), and the mixture was dissolved with boron trifluoride etherate (3.81 ml, 30.3 Band01) and stirred at room temperature for 7 hours. The reaction solution was poured into ice-cold water (200 g), mixed and partitioned, and the aqueous layer was extracted with ethyl acetate (200 ml). The organic layers were combined, washed with an aqueous solution of sodium carbonate, water and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure. Ether and diisopropyl ether were added to the residue, and the resulting precipitate was collected by filtration, washed with diisopropyl ether, and dried to give the title compound as a white powder (6.17 g, 20.9 tmol).
Ή-NMR (300 MHz, 重クロ口ホルム中): δ 1.48 (9Η, s), 4.22 (2H, s), 7.37 (1H, brs) . Ή-NMR (300 MHz, in double-mouthed form): δ 1.48 (9Η, s), 4.22 (2H, s), 7.37 (1H, brs).
元素分析値: C10HI2N204C12 として計算値: C, 40.70; H, 4.10; N, 9.49; C1, 24.03 (%) 実測値; C, 40.70; H, 4.12; N, 9.23; C1, 23.97 (%) . Elemental analysis: C 10 H I2 N 2 0 4 C1 2 Calculated: C, 40.70; H, 4.10 ; N, 9.49; C1, 24.03 (%) Found; C, 40.70; H, 4.12 ; N, 9.23 ; C1, 23.97 (%).
参考例 14 Reference Example 14
式 (XX I X) において R 14が tert-ブトキシカルボニルメチル基、 Rがメチル 基である化合物の合成 Synthesis of a compound of the formula (XX IX) wherein R 14 is a tert-butoxycarbonylmethyl group and R is a methyl group
参考例 13-2で合成した化合物 (3.05 g, 10.3 mmol) および炭酸力リゥム (4.29 g, 31.0誦 ol) にァセトニトリル (60 ml) を加えて懸濁し、 3-メルカプトプロピ オン酸メチル (2.34 ml, 20.7 mmol) を滴下後、 室温で 2時間撹拌し、 さらに 8 で 40分間撹拌した。 反応液を濃縮後、 1N塩酸 (31 ml), 水 (80 ml) およ び酢酸ェチル (100 ml) を加えて混和し、 有機層を 2 %炭酸水素ナトリウム水溶 液で抽出した。 水層を合わせて pH3に調整し、 酢酸ェチルで抽出した。 有機層を 7%食塩水および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥した。溶媒を減圧下 に溜去し、 残渣にメタノールを加え、 生成した沈澱物を濾取し、 メタノールで洗 浄後、 乾燥して標題化合物の橙色油状物 (3.20 g、 8.50 膽 ol) を得た。 Acetonitrile (60 ml) was added to the compound synthesized in Reference Example 13-2 (3.05 g, 10.3 mmol) and carbonated carbonate (4.29 g, 31.0 ol) and suspended, and methyl 3-mercaptopropionate (2.34 ml) was added. , 20.7 mmol) was added dropwise, and the mixture was stirred at room temperature for 2 hours, and further stirred at 8 for 40 minutes. After concentrating the reaction solution, 1N hydrochloric acid (31 ml), water (80 ml) and ethyl acetate (100 ml) were added and mixed, and the organic layer was extracted with a 2% aqueous sodium hydrogen carbonate solution. The aqueous layers were combined, adjusted to pH 3, and extracted with ethyl acetate. The organic layer was washed with a 7% saline solution and a saturated saline solution, and then dried with sodium sulfate. The solvent was distilled off under reduced pressure, methanol was added to the residue, and the resulting precipitate was collected by filtration, washed with methanol, and dried to give the title compound as an orange oil (3.20 g, 8.50 benzyl) .
Ή -匿 (300 MHz, 重クロ口ホルム中) : δ 1.45/1.49 (9Η, s), 2.71/2.75 (2H, t, J= 6.9/7.0 Hz), 3.24/3.58 (2H, t, J= 6.9/7.0 Hz), 3.72/3.74 (3H, s), 4.14 (1/2H, d, J= 17.9 Hz), 4.16 (1/2H, d, J= 17.9 Hz), 4.23 (2/2H, s), 5.28 (1/2H, d, J= 0.7 Hz), 6.14 (1/2H, brs), 7.36 (1/2H, brs), 7.74 (1/2H, brs). 元素分析値: C14H2。N206S2として計算値: C, 44.67; H, 5.35; N, 7.44; S, 17.04 (%)、 実測値: C, 44.77; H, 5.48; N, 7.27; S, 16.61 (%) · 参考例 15 " 3-{[ (イソプチルスルファニル)(5-{[(4-メチルフエニル)スルホニル]イミノ}-2- ォキソ -4-ィミダゾリジニリデン)メチル]スルファニル }プロピオン酸ェチル 参考例 3- 2で合成した化合物 (8.00 g, 18.6删 ol) を THF (80.0 ml) および D F (40.0 ml) に溶解し、 ヨウ化イソブチル (2.36 ml, 20.5 mmol) およびトリ ェチルァミン (7.79 ml, 55.9讓01) を加えて室温で 17時間撹拌した。 反応液を 濃縮後、 残渣を酢酸ェチルで希釈し、 水、 および飽和食塩水で洗浄後、 硫酸ナト リウムで乾燥した。 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマ トグラフィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合 物(6.05g, 67%)を黄色結晶として得た。酢酸ェチルから再結晶して融点 95- 97°C の黄色結晶を得た。 Ή-concealed (300 MHz, in double-mouthed form): δ 1.45 / 1.49 (9Η, s), 2.71 / 2.75 (2H, t, J = 6.9 / 7.0 Hz), 3.24 / 3.58 (2H, t, J = 6.9 / 7.0 Hz), 3.72 / 3.74 (3H, s), 4.14 (1 / 2H, d, J = 17.9 Hz), 4.16 (1 / 2H, d, J = 17.9 Hz), 4.23 (2 / 2H, s ), 5.28 (1 / 2H, d, J = 0.7 Hz), 6.14 (1 / 2H, brs), 7.36 (1 / 2H, brs), 7.74 (1 / 2H, brs) elemental analysis:. C 14 H 2 . N 2 0 6 S 2 Calculated: C, 44.67; H, 5.35 ; N, 7.44; S, 17.04 (%), Found: C, 44.77; H, 5.48 ; N, 7.27; S, 16.61 (%) · Reference Example 15 "3-{[(isobutylsulfanyl) (5-{[(4-methylphenyl) sulfonyl] imino} -2-oxo-4-imidazolidinylidene) methyl] sulfanyl} ethyl propionate Reference Example 3 -The compound synthesized in (2) (8.00 g, 18.6 mol) was dissolved in THF (80.0 ml) and DF (40.0 ml), and isobutyl iodide (2.36 ml, 20.5 mmol) and triethylamine (7.79 ml, 55.9 ml) were dissolved. ) Was added and the mixture was stirred at room temperature for 17 hours.After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed with water and saturated saline, and dried over sodium sulfate. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (6.05 g, 67%) as yellow crystals. Recrystallization from ethyl acetate gave yellow crystals with a melting point of 95-97 ° C.
IR (KBr) V: 3368, 3200, 3071, 2953, 2924, 2907, 2868, 1765, 1730, 1634, 1574, 1499 CDT1. IR (KBr) V: 3368, 3200, 3071, 2953, 2924, 2907, 2868, 1765, 1730, 1634, 1574, 1499 CDT 1 .
Ή -賺 (CDC13) : δ 1.00 (6Η, d, J=6.6 Hz), 1.25 (3H, t, J=6.8 Hz), 1.73-1.89 (1H, m), 2.43 (3H, s), 2.56 (2H, t, J=7.4 Hz), 2.80 (2H, d, J=6.6 Hz), 3.16 (2H, t, J=7.4 Hz), 4.13 (2H, q, J=7.0 Hz), 7.32 (2H, d, J=8.2 Hz), 7.56 (1H, bs), 7.89 (2H, d, J=8.4 Hz), 9.65 (1H, bs). E -賺(CDC1 3): δ 1.00 ( 6Η, d, J = 6.6 Hz), 1.25 (3H, t, J = 6.8 Hz), 1.73-1.89 (1H, m), 2.43 (3H, s), 2.56 (2H, t, J = 7.4 Hz), 2.80 (2H, d, J = 6.6 Hz), 3.16 (2H, t, J = 7.4 Hz), 4.13 (2H, q, J = 7.0 Hz), 7.32 (2H , d, J = 8.2 Hz), 7.56 (1H, bs), 7.89 (2H, d, J = 8.4 Hz), 9.65 (1H, bs).
元素分析値: C2。H27N305S3として Elemental analysis: C 2. H 27 N 3 0 5 As S 3
計算値 (¾) : C, 49.46; H, 5.60; N, 8.65; S, 19.81. Calculated value (¾): C, 49.46; H, 5.60; N, 8.65; S, 19.81.
実測値 (¾) : C, 49.42; H, 5.45; N, 8.60; S, 19.83. Found (¾): C, 49.42; H, 5.45; N, 8.60; S, 19.83.
参考例 16 Reference Example 16
N - {5- [(イソプチルスルファニル)(スルファニル)メチリデン] -2-ォキソ -4 -ィミ 参考例 15で合成した 3- {[ (イソプチルスルファニル)(5- {[ (4-メチルフエニル) スルホニル]ィミノ }- 2-ォキソ - 4-ィミダゾリジニリデン)メチル]スルファニル } プロピオン酸ェチル (1.69 g, 2.48讓 ol) の THF (17.0 ml) およびエタノール (17.0 ml) 懸濁液に、 水酸化カリウム (689 mg, 10.4匪 ol) を加えて、 室温で 40分間撹拌した。 反応液に 1N塩酸 (12.2 ml) を加えて混和し、 室温で 10分間 撹拌した。 生成した沈澱物を濾取し、 水、 エタノールで洗浄後、 乾燥して表題化 合物の黄色結晶 (1.03 g, 〜100« を得た。 酢酸ェチルから再結晶して融点 204- 205°Cの黄色結晶を得た。 N- {5-[(Isoptylsulfanyl) (sulfanyl) methylidene] -2-oxo-4-imimi 3-{[(isobutylsulfanyl) (5-{[(4-methylphenyl) Sulfonyl] imino} -2-oxo-4-imidazolidinylidene) methyl] sulfanyl} A suspension of ethyl (1,69 ml) in ethyl (17.0 ml) and propanolate (17.0 ml) in water was added to water. Potassium oxide (689 mg, 10.4 ol) was added and stirred at room temperature for 40 minutes. 1N Hydrochloric acid (12.2 ml) was added to the reaction solution, mixed, and stirred at room temperature for 10 minutes. The precipitate formed was collected by filtration, washed with water and ethanol, dried and titled The compound was recrystallized from ethyl acetate to give yellow crystals having a melting point of 204-205 ° C.
IR ( Br) V: 3300-2800, 2961, 1703, 1671, 1570, 1507 cm"1. IR (Br) V: 3300-2800, 2961, 1703, 1671, 1570, 1507 cm " 1 .
Ή-N R (CDC13) : 51.02 (6H, d, J=6.6 Hz), 1.90-2.04 (1H, m), 2.44 (3H, s), 3.21 (2H, d, J=6.6 Hz), 7.34 (2H, d, J=8.4 Hz), 7.78 (2H, d, J=8.4 Hz), 8.19 (1H, bs), 9.24 (1H, bs). Ή-NR (CDC1 3): 51.02 (6H, d, J = 6.6 Hz), 1.90-2.04 (1H, m), 2.44 (3H, s), 3.21 (2H, d, J = 6.6 Hz), 7.34 ( 2H, d, J = 8.4 Hz), 7.78 (2H, d, J = 8.4 Hz), 8.19 (1H, bs), 9.24 (1H, bs).
元素分析値: C15H19N303S3として Elemental analysis: as C 15 H 19 N 3 0 3 S 3
計算値 (¾) : C, 46.73; H, 4.97; N, 10.90; S, 24.95. Calculated (¾): C, 46.73; H, 4.97; N, 10.90; S, 24.95.
実測値 (¾) : C, 46.58; H, 4.87; N, 10.61; S, 24.70. Found (¾): C, 46.58; H, 4.87; N, 10.61; S, 24.70.
参考例 17 Reference Example 17
2-{[ (イソプチルスルファニル) (5 - {[(4-メチルフエニル)スルホニル]イミノ}-2 ォキソ -4-ィミダゾリジニリデン)メチル]スルファニル }プロピオン酸ェチル 参考例 16で合成した N- {5- [(イソプチルスルファニル) (スルファニル)メチリ デン] -2-ォキソ -4-イミダゾリジニリデン } -4-メチルベンゼンスルホンアミド (4.33 g, 11.2 匪 ol) を THF (43.0 ml) および DMF (21.5 ml) に溶解し、 2-ブ ロモプロピオン酸ェチル(1.61 ml, 12.4匪01)およびトリェチルァミン(4.70 ml, 33.7mmol) を加えて室温で 7時間撹拌した。 反応液を濃縮後、 残渣を酢酸ェチル で希釈し、 水、 および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を 減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに付し、 n-へ キサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (4.22 g, 11%) を黄色結 晶として得た。 酢酸ェチルから再結晶して融点 97-99 の黄色結晶を得た。 2-{[(isobutylsulfanyl) (5-{[(4-methylphenyl) sulfonyl] imino} -2oxo-4-imidazolidinylidene) methyl] sulfanyl} ethyl propionate N-synthesized in Reference Example 16 {5-[(Isoptylsulfanyl) (sulfanyl) methylidene] -2-oxo-4-imidazolidinylidene} -4-methylbenzenesulfonamide (4.33 g, 11.2 bandol) was added to THF (43.0 ml) and DMF ( 21.5 ml), and ethyl 2-bromopropionate (1.61 ml, 12.4 band01) and triethylamine (4.70 ml, 33.7 mmol) were added, followed by stirring at room temperature for 7 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed with water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (4.22 g, 11%) as yellow crystals. Recrystallization from ethyl acetate gave yellow crystals with a melting point of 97-99.
IR (KBr) V: 3258, 2959, 2932, 2870, 1750, 1634, 1566, 1497 cm—1. IR (KBr) V: 3258, 2959, 2932, 2870, 1750, 1634, 1566, 1497 cm— 1 .
Ή-NM (CDC13) : δ 0.912 (6Η, dd, J=6.6, 2.2 Hz), 1.23 (3H, t, J=7.2 Hz), 1.54 (3H, d, J=7.2 Hz), 1.64-1.80 (1H, ) , 2.43 (3H, s), 2.79 (2H, dd, J=6.6, 1.0 Hz), 3.93 (1H, q, J=7.4 Hz), 4.13 (2H, q, J=7.0 Hz), 7.30 (2H, d, J-8.4 Hz), 7.89 (2H, d, J=8.4 Hz), 7.91 (1H, bs), 7.67 (1H, bs). Ή-NM (CDC1 3): δ 0.912 (6Η, dd, J = 6.6, 2.2 Hz), 1.23 (3H, t, J = 7.2 Hz), 1.54 (3H, d, J = 7.2 Hz), 1.64-1.80 (1H,), 2.43 (3H, s), 2.79 (2H, dd, J = 6.6, 1.0 Hz), 3.93 (1H, q, J = 7.4 Hz), 4.13 (2H, q, J = 7.0 Hz), 7.30 (2H, d, J-8.4 Hz), 7.89 (2H, d, J = 8.4 Hz), 7.91 (1H, bs), 7.67 (1H, bs).
元素分析値: C2QH27N305S3として Elemental analysis: as C 2Q H 27 N 3 0 5 S 3
計算値 (¾) : C, 49.46; H, 5.60; N, 8.65; S, 19.81. Calculated (¾): C, 49.46; H, 5.60; N, 8.65; S, 19.81.
実測値 (¾) : C, 49.25; H, 5.60; N, 8.63; S, 19.86. 参考例 18 Found (¾): C, 49.25; H, 5.60; N, 8.63; S, 19.86. Reference Example 18
3 -ブロモ -2 -ケトグル夕ル酸ジメチル Dimethyl 3-bromo-2-ketoglurate
2—ケトグルタル酸 (25.3 g, 0.171 mol) と 濃塩酸 (14.3 ml, 0.171 mol) の メタノール (200 ml) 溶液を室温で 24時間攪拌した。 酢酸ェチルにより抽出し、 粗製の 2-ケトグル夕ル酸ジメチル (純度 72%, 30.4 g, 0.126 mol, 73¾) の淡黄 色油状物を得た。 A solution of 2-ketoglutaric acid (25.3 g, 0.171 mol) and concentrated hydrochloric acid (14.3 ml, 0.171 mol) in methanol (200 ml) was stirred at room temperature for 24 hours. Extraction with ethyl acetate gave a pale yellow oil of crude dimethyl 2-ketoglurate (purity 72%, 30.4 g, 0.126 mol, 73%).
Ή-N R (300MHz, CDC13): δ 2.69 (2Η, t, J= 6.5 Hz), 3.17 (2H, t, J= 6.5 Hz), 3.70 (3H, s), 3.89 (3H, s). Ή-NR (300MHz, CDC1 3 ): δ 2.69 (2Η, t, J = 6.5 Hz), 3.17 (2H, t, J = 6.5 Hz), 3.70 (3H, s), 3.89 (3H, s).
3 臭化トリメチルフエ二ルアンモニゥム(13.4 g, 34.9 mmol)のクロ口ホルム (100 ml)の懸濁液に室温で攪拌しながら上記 2-ケトグルタル酸ジメチル (純度 72%, 8.4 g, 34.9匪 ol)のクロ口ホルム (60 ml)溶液を 5分間かけて滴下し、 反 応液を室温で 3時間攪拌した。 反応液を濃縮してクロ口ホルムを留去後、 酢酸ェ チル(350 ml)で希釈し、 15%および飽和の食塩水で洗浄後、硫酸ナトリウムで乾燥、 濃縮して粗製の 3-ブロモ -2-ケトグルタル酸ジメチル(純度 70%, 11.8 g, 32.6 mmol, 93.5%)の黄色油状物を得た。 3 The above-mentioned dimethyl 2-ketoglutarate (purity 72%, 8.4 g, 34.9 ol) was added to a suspension of trimethylphenylammonium bromide (13.4 g, 34.9 mmol) in chloroform (100 ml) with stirring at room temperature. Was added dropwise over 5 minutes, and the reaction solution was stirred at room temperature for 3 hours. After concentrating the reaction solution and distilling off the chloroform form, dilute with ethyl acetate (350 ml), wash with 15% and saturated saline, dry over sodium sulfate and concentrate to obtain crude 3-bromo- A yellow oil of dimethyl 2-ketoglutarate (purity 70%, 11.8 g, 32.6 mmol, 93.5%) was obtained.
'H-NMR (200MHz, CDC13): 53.06 (1H, dd, J= 17.2, 5.8 Hz), 3.33 (1H, dd, J= 17.2, 9.2 Hz), 3.71 (3H, s), 3.95 (3H, s), 5.40 (1H, dd, J= 9.2, 5.8 Hz). 参考例 19 'H-NMR (200MHz, CDC1 3): 53.06 (1H, dd, J = 17.2, 5.8 Hz), 3.33 (1H, dd, J = 17.2, 9.2 Hz), 3.71 (3H, s), 3.95 (3H, s), 5.40 (1H, dd, J = 9.2, 5.8 Hz). Reference Example 19
N - Boc- 5-ァミノ-卜ペンチル メシレート N-Boc-5-aminopentyl mesylate
5 -ァミノ-卜ペンタノ一ル (純度 95%, 4.25 g, 39.1 mmol) と 重炭酸ジ tert- ブチル (8.81 g, 39.1 匪 ol) を THF-水 (1:1, 100 ml) 中でトリェチルァミン (13.6ml, 97.8腿 ol) と反応させて N-Boc 5-ァミノ-卜ペンタノール (純度 87 ¾, 9.10 g, 39.1 画 ol) の無色油状物を得た。 5-Aminopentanol (purity 95%, 4.25 g, 39.1 mmol) and ditert-butyl bicarbonate (8.81 g, 39.1 marl) were dissolved in THF-water (1: 1, 100 ml) in triethylamine (1: 1). 13.6 ml, 97.8 t ol) to give N-Boc 5-amino-pentanol (purity 87¾, 9.10 g, 39.1 fraction ol) as a colorless oil.
Ή-NMR (300MHz, CDC13): 61.U (9H, s), 1.4-1.6 (6H, m), 3.13 (2H, brq, J= 6.6 Hz), 3.65 (2H, brq, J= 2.4 Hz), 4.57 (1H, m). Ή-NMR (300MHz, CDC1 3 ): 61.U (9H, s), 1.4-1.6 (6H, m), 3.13 (2H, brq, J = 6.6 Hz), 3.65 (2H, brq, J = 2.4 Hz ), 4.57 (1H, m).
得られた 5-ァミノ-卜ペン夕ノール 3.92 g, 16.8匪 ol)およびメタンスルホ二 ルクロリ ド (1.45 ml, 18.5腿 ol)の THF (80 ml)溶液に 0 で攪拌しながらトリ エヂルァミン(2.79 ml, 20.1 mmol)を加え、 反応液を 0°Cで 2時間攪拌した。 反 応液を酢酸ェチル抽出して精製し、 N- Boc- 5-ァミノ- 1-ぺンチル メシレート(純度 87%, 5.46 g, 16.8 mmol) の無色油状物を得た。 To a solution of the resulting 5-aminopentanol (3.92 g, 16.8 ol) and methanesulfonyl chloride (1.45 ml, 18.5 t ol) in THF (80 ml) was stirred with 0 at 0 ° C. 20.1 mmol) was added, and the reaction solution was stirred at 0 ° C for 2 hours. The reaction solution was purified by extraction with ethyl acetate and purified with N-Boc-5-amino-1-pentyl mesylate (purity 87%, 5.46 g, 16.8 mmol) of a colorless oil.
Ή-NMR (300MHz, CDC13): 51.44 (9H, s), 1.56 (4H, m), 1.79 (2H, brquint, J= 7.5 Hz), 3.03 (3H, s), 3.13 (2H, brq, J= 6.3 Hz), 4.23 (2H, t, J= 6.4 Hz), 4.56 (1H, m). Ή-NMR (300MHz, CDC1 3 ): 51.44 (9H, s), 1.56 (4H, m), 1.79 (2H, brquint, J = 7.5 Hz), 3.03 (3H, s), 3.13 (2H, brq, J = 6.3 Hz), 4.23 (2H, t, J = 6.4 Hz), 4.56 (1H, m).
参考例 20 Reference Example 20
ヨウ化 N - Boc- 6-アミノ-卜へキシル N-Boc-6-amino-trihexyl iodide
参考例 19 と同様にして 6-ァミノ- 1 -へキサノール (5.0 g, 40.5腿 ol) から N - Boc-6 -ァミノ- 1-へキシル メシレート (純度 80.4, 6.39 g, 40.5腿01) の無 色油状物を得た。 In the same manner as in Reference Example 19, N-Boc-6-amino-1-hexyl mesylate (purity 80.4, 6.39 g, 40.5 thigh 01) was obtained from 6-amino-1-hexanol (5.0 g, 40.5 thigh ol). A colored oil was obtained.
'H-NMR (300MHz, CDC13): δ 1.34-1.50 (6Η, m), 1.44 (9H, s), 1.75 (2H, brquint, J= 6.8 Hz), 3.01 (3H, s), 3.11 (2H, brq, J= 6.4 Hz), 4.22 (2H, t, J= 6.4 Hz), 4.53 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 1.34-1.50 (6Η, m), 1.44 (9H, s), 1.75 (2H, brquint, J = 6.8 Hz), 3.01 (3H, s), 3.11 (2H , brq, J = 6.4 Hz), 4.22 (2H, t, J = 6.4 Hz), 4.53 (1H, br).
上記で得た N- Boc-6-ァミノ- 1-へキシル メシレート (純度 80.4%, 2.72 g, 7.40腿 ol)およびヨウ化ナトリウム(2.22 g, 14.8腿 ol) の アセトン (60 ml) 懸濁液を 2 時間加熱還流した。 反応液を酢酸ェチルにより抽出してヨウ化 N - Boc- 6-ァミノ- 1 -へキシル(94%, 2.57 g, 7.40 mmol)を褐色油状物として得た。 参考例 21 Suspension of N-Boc-6-amino-1-hexyl mesylate (purity 80.4%, 2.72 g, 7.40 t ol) and sodium iodide (2.22 g, 14.8 t ol) in acetone (60 ml) obtained above Was heated to reflux for 2 hours. The reaction solution was extracted with ethyl acetate to obtain N-Boc-6-amino-1-hexyl iodide (94%, 2.57 g, 7.40 mmol) as a brown oil. Reference Example 21
4,4-ジフルォロ- 1-ョ一ドシクロへキサン 4,4-difluoro-1-cyclohexane
シクロへキサン- 1, 4-ジオンモノエチレンケタール(8.84 g, 55.5腿 ol)のメタ ノール(170 ml)溶液に室温で攪拌しながら水素化ホウ素ナトリウム (純度 90%, 0.57 g, 13.6匪01) の粉末を加えて 15分間攪拌した。 反応液に水素化ホウ素ナ トリウム (70 mg, 1.67 mmol)を加えて 20分間攪拌した。 反応液を酢酸ェチル抽 出により精製し、 4-ォキソシクロへキサノ一ルエチレンケタール(8.44 g, 53.3 mmol, 96%)の淡褐色油状物を得た。 Sodium borohydride (purity 90%, 0.57 g, 13.6 band01) in a solution of cyclohexane-1,4-dionemonoethylene ketal (8.84 g, 55.5 tmol) in methanol (170 ml) at room temperature with stirring Was added and stirred for 15 minutes. Sodium borohydride (70 mg, 1.67 mmol) was added to the reaction solution, and the mixture was stirred for 20 minutes. The reaction solution was purified by extraction with ethyl acetate to obtain a light brown oil of 4-oxocyclohexanolethylene ketal (8.44 g, 53.3 mmol, 96%).
¾-NMR (200MHz, CDC13): 51.43 (6H, s), 1.47 (3H, s), 2.26 (1H, ddd, J= 14.4, 8.2, 4.8 Hz), 2.62 (1H, m), 3.06 (3H, s) , 3.75 (3H, s), 3.79 (2H, m), 4.43 (1H, dt, J= 12.8, 8.1 Hz), 5.26 (1H, m) . ¾-NMR (200MHz, CDC1 3 ): 51.43 (6H, s), 1.47 (3H, s), 2.26 (1H, ddd, J = 14.4, 8.2, 4.8 Hz), 2.62 (1H, m), 3.06 (3H , s), 3.75 (3H, s), 3.79 (2H, m), 4.43 (1H, dt, J = 12.8, 8.1 Hz), 5.26 (1H, m).
4 -ォキソシクロへキサノールエチレンケタール(4.05 g, 25.6 mmol)およびメタ ンスルホニルクロリド (3.47 g, 30.0膽 ol)の THF (80 ml)溶液に 0°Cで攪拌し ながらトリェチルァミン(4.85 ml, 35.0 mmol)の THF (10 ml)溶液を 30分間かけ て滴下した。 反応液を 0でで 90分間攪拌した。 反応液を酢酸ェチル抽出し、 粗製 の 4-ォキソシクロへキシルメシレ一トエチレンケタール (純度 65%, 6.55 g, 18 腿 ol, 70»の淡黄色油状物を得た。 The mixture was stirred at 0 ° C in a solution of 4-oxocyclohexanol ethylene ketal (4.05 g, 25.6 mmol) and methanesulfonyl chloride (3.47 g, 30.0 butyl) in THF (80 ml). While stirring, a solution of triethylamine (4.85 ml, 35.0 mmol) in THF (10 ml) was added dropwise over 30 minutes. The reaction was stirred at 0 for 90 minutes. The reaction solution was extracted with ethyl acetate to obtain a crude 4-oxocyclohexylmesylethylene ethylene ketal (purity 65%, 6.55 g, 18 ol, 70 »pale yellow oily substance).
Ή-NMR (200MHz, CDC13): δ 1.66 (2Η, m), 1.84 (2H, brq, J= 6.4 Hz), 1.99 (4H, brt, J= 5.9 Hz), 3.02 (3H, s), 3.95 (4H, brt, J= 2.0 Hz), 4.85 (1H, quint, J= 5.1 Hz). Ή-NMR (200MHz, CDC1 3 ): δ 1.66 (2Η, m), 1.84 (2H, brq, J = 6.4 Hz), 1.99 (4H, brt, J = 5.9 Hz), 3.02 (3H, s), 3.95 (4H, brt, J = 2.0 Hz), 4.85 (1H, quint, J = 5.1 Hz).
上記で得られた 4-ォキソシクロへキシルメシレー卜エチレンケタール(純度 65¾, 6.55 g, 18腿 ol)および p-トルエンスルホン酸水和物 (2.47g, 13.0龍 ol) の アセトン -水 (1:1, 130 ml)溶液を室温で 2.5時間、 50°C で 4時間攪拌した。 反応液を酢酸ェチル抽出し、 4-ォキソシクロへキシルメシレート (純度 90¾, 3.44 g, 16.1 mmol, 89.4%)の黄色油状物を得た。 The above obtained 4-oxocyclohexyl mesylate ethylene ketal (purity 65¾, 6.55 g, 18 liters) and p-toluenesulfonic acid hydrate (2.47 g, 13.0 liters) in acetone-water (1: 1, 130 ml) solution was stirred at room temperature for 2.5 hours and at 50 ° C for 4 hours. The reaction mixture was extracted with ethyl acetate to give 4-oxocyclohexyl mesylate (purity 90 °, 3.44 g, 16.1 mmol, 89.4%) as a yellow oil.
Ή-NMR (200MHz, CDC13): 62.15 (2H, m), 2.3-2.5 (4H, m), 2.62 (2H, m), 3.10 (3H, s), 5.13 (1H, tt, J= 5.4, 2.7 Hz). Ή-NMR (200MHz, CDC1 3 ): 62.15 (2H, m), 2.3-2.5 (4H, m), 2.62 (2H, m), 3.10 (3H, s), 5.13 (1H, tt, J = 5.4, 2.7 Hz).
4-ォキソシクロへキシルメシレート(純度 90¾, 3. 4 g, 16.1 mmol)のジクロ口 メタン (30 ml)の溶液に 0 で攪拌しながら Deoxo- Fluor™ (7.08 g, 32.0 mmol) のジクロロメタン (3 ml)溶液を 30分間かけて滴下し、 昇温後、 反応液を室温で 6時間攪拌した。反応液をジェチルエーテル (30 ml) で希釈後、 氷 (50 g)に注ぎ、 ジェチルェ一テル(50 ml)およびジイソプロピルェ一テル(50 ml)で抽出した。 有 機層を飽和炭酸水素ナトリウム、 水および飽和食塩水で洗浄し、 硫酸ナトリウム で乾燥後、濃縮して 4, 4-ジフルォロシクロへキシルメシレート (純度 92¾, 3.5g, 15匪 ol, 93%)を黄色油状物として得た。 A solution of 4-oxocyclohexyl mesylate (purity 90¾, 3.4 g, 16.1 mmol) in dichloromethane (30 ml) was stirred at 0 with Deoxo-Fluor ™ (7.08 g, 32.0 mmol) in dichloromethane (3 ml). ml) solution was added dropwise over 30 minutes, and after heating, the reaction solution was stirred at room temperature for 6 hours. The reaction solution was diluted with getyl ether (30 ml), poured into ice (50 g), and extracted with getyl ether (50 ml) and diisopropyl ether (50 ml). The organic layer is washed with saturated sodium bicarbonate, water and saturated saline, dried over sodium sulfate, concentrated and concentrated to 4,4-difluorocyclohexyl mesylate (purity 92¾, 3.5 g, 15 bandol, 93%) Was obtained as a yellow oil.
Ή-NMR (200MHz, CDC13): (51.92-2.18 (6H, m), 2.40 (2H, m), 3.04/3.05 (3H, s), 4.92/4.98 (1H, m). Ή-NMR (200MHz, CDC1 3 ): (51.92-2.18 (6H, m), 2.40 (2H, m), 3.04 / 3.05 (3H, s), 4.92 / 4.98 (1H, m).
4, 4-ジフルォロシクロへキシルメシレート (純度 92 , 3.5 g, 15腿 ol)および ヨウ化ナトリウム(4.50 g, 30腿 ol) の 2-ブ夕ノン (60 ml)懸濁液を 13時間加 熱還流した。 反応液をジィソプロピルエーテルージェチルエーテルにより抽出し て粗製の 4, 4-ジフルォ口-卜ョ一ドシクロへキサン(純度 45%, 4.9 g, 9.0 mmol, 60%)を褐色油状物として得た。 lH - NMR (300MHz, CDC13): 51.91 (2H, m), 2.08 (4H, i), 2.14 (2H, m), 4.40/4.46 (1H, m). A suspension of 4,4-difluorocyclohexyl mesylate (purity 92, 3.5 g, 15 t ol) and sodium iodide (4.50 g, 30 t ol) in 2-butaneone (60 ml) was heated for 13 hours. Refluxed. The reaction solution was extracted with disopropyl ether-ethyl ether to obtain crude 4,4-difluoromethylcyclohexane (purity: 45%, 4.9 g, 9.0 mmol, 60%) as a brown oil. . l H - NMR (300MHz, CDC1 3): 51.91 (2H, m), 2.08 (4H, i), 2.14 (2H, m), 4.40 / 4.46 (1H, m).
参考例 22 Reference Example 22
4,4-ジフルォ口-卜ョードメチルシクロへキサン 4,4-difluoro-methyl-cyclohexane
4-ォキソシクロへキサンカルポン酸ェチル(2.29 g, 13.4mmol)のジクロ口メタ ン (20 ml)の溶液に で攪拌しながら Deoxo - Fluor™ (5.06 g, 22.9 mmol)を 30 分間かけて滴下し、 昇温後、 反応液を室温で 6時間攪拌した。 反応液氷 (50 g)に 注ぎ、 酢酸ェチル(100 ml)およびジェチルェ一テル(30 ml)で抽出した。 有機層を 5%および 2¾炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄し、 硫酸ナト リウムで乾燥後、濃縮して粗製の 4, 4-ジフルォロシクロへキサンカルボン酸ェチ ル (2.79 g)を黄色油状物として得た。 この黄色油状物を THF (55 ml) に溶解し、 0°Cで攪拌しながら水素化リチウムアルミニウム (純度 80%, 636 mg, 13.4 mmol) を数回に分けて加え、 0°Cで 30分間攪拌した。 反応液にメタノールを徐々に加え て未反応の水素化リチウムアルミニウムを分解後 0.5M クェン酸 (28 ml)を加え てジェチルェ一テル-ジイソプロピルエーテル(1:1, 100 ml)で抽出した。 有機層 を洗浄、乾燥、濃縮して 4, 4-ジフルォロシクロへキサンメタノール (1.94g, 12.9 mmol, 96%)を黄色油状物として得た。 Deoxo-Fluor ™ (5.06 g, 22.9 mmol) was added dropwise over 30 minutes to a solution of 4-oxocyclohexanecarbethylate (2.29 g, 13.4 mmol) in dichloromethane (20 ml) with stirring with stirring for 30 minutes. After warming, the reaction was stirred at room temperature for 6 hours. The reaction solution was poured into ice (50 g), and extracted with ethyl acetate (100 ml) and getyl ether (30 ml). The organic layer was washed with 5% aqueous sodium hydrogencarbonate solution, water and saturated saline, dried over sodium sulfate, and concentrated to give crude 4,4-difluorocyclohexanecarboxylate (2.79 g). Obtained as a yellow oil. This yellow oil was dissolved in THF (55 ml), lithium aluminum hydride (purity 80%, 636 mg, 13.4 mmol) was added in several portions with stirring at 0 ° C, and the mixture was added at 0 ° C for 30 minutes. Stirred. Unreacted lithium aluminum hydride was decomposed by slowly adding methanol to the reaction solution, and then 0.5M citric acid (28 ml) was added thereto, followed by extraction with getyl ether-diisopropyl ether (1: 1, 100 ml). The organic layer was washed, dried and concentrated to give 4,4-difluorocyclohexanemethanol (1.94 g, 12.9 mmol, 96%) as a yellow oil.
Ή-NMR (200MHz, CDC13): 51.34 (2H, m), 1.55 (2H, m), 1.75 (1H, ), 1.80 (2H, m), 2.05-2.25 (2H, m), 3.53 (2H, m). Ή-NMR (200MHz, CDC1 3 ): 51.34 (2H, m), 1.55 (2H, m), 1.75 (1H,), 1.80 (2H, m), 2.05-2.25 (2H, m), 3.53 (2H, m).
4,4-ジフルォロシクロへキサンメタノール (1.94 g, 12.9 mmol)およびメタン スルホニルクロリド (1.64 g, 14.2 mmol)の THF (39 ml)溶液に 0°Cで攪拌しな がらトリェチルァミン(2.15 ml, 15.5 mmol)の THF (5 ml)溶液を 60分間かけて 滴下した後、 反応液を 0°Cで 60分間攪拌した。.反応液をジェチルエーテル-ジィ ソプロピルエーテル(1:1, 100 ml)で抽出し、 粗製の 4, 4-ジフルォロシクロへキ シルメチルメシレート (純度 90%, 2.79 g, 11 匪 ol, 85%)の無色油状物を得た。 Ή-N R (200MHz, CDC13): (51.39 (2H, brq, J- 12.1 Hz), 1.57 (1H, m), 1.70-2.10 (2H, m), 1.85 (2H, m), 2.19 (2H, brd, J= 16 Hz), 3.02 (3H, s), 4.08/4.13 (2H, d, J= 6.2 Hz). Triethylamine (2.15 ml, 15.5 mmol) in a solution of 4,4-difluorocyclohexanemethanol (1.94 g, 12.9 mmol) and methanesulfonyl chloride (1.64 g, 14.2 mmol) in THF (39 ml) while stirring at 0 ° C. After a THF (5 ml) solution was added dropwise over 60 minutes, the reaction solution was stirred at 0 ° C for 60 minutes. The reaction mixture was extracted with getyl ether-diisopropyl ether (1: 1, 100 ml) and the crude 4,4-difluorocyclohexylmethyl mesylate (purity 90%, 2.79 g, 11 bandol ol, 85 %) As a colorless oil. Ή-NR (200MHz, CDC1 3 ): (51.39 (2H, brq, J- 12.1 Hz), 1.57 (1H, m), 1.70-2.10 (2H, m), 1.85 (2H, m), 2.19 (2H, brd, J = 16 Hz), 3.02 (3H, s), 4.08 / 4.13 (2H, d, J = 6.2 Hz).
4, 4-ジフルォロシクロへキシルメチルメシレート (純度 90%, 2.79g, 11匪 ol) およびヨウ化ナトリウム(3.30 g, 22 imol) の アセトン (60 ml)懸濁液を 1.5 時間加熱還流後、 50°C で 16時間攪拌した。 反応液を酢酸ェチル -へキサン(1:1) により抽出して 4, 4-ジフルォロ- 1-ョードメチルシクロへキサン(51%酢酸ェチル 溶液, 5.6 g, 11 匪 ol, quant.)を褐色油状物として得た。 ' Ή-NMR (200MHz, CDC13): 61.35 (2H, brq, J= 10.6 Hz), 1.59 (2H, m), 1.80 (2H, m), 1.92 (2H, m), 2.23 (1H, m), 3.13/3.19 (2H, d, J= 6.2/5.8 Hz). 4, 4-difluorocyclohexylmethyl mesylate (Purity 90%, 2.79g, 11 ol) A suspension of sodium iodide (3.30 g, 22 imol) in acetone (60 ml) was heated under reflux for 1.5 hours, and then stirred at 50 ° C for 16 hours. The reaction mixture was extracted with ethyl acetate-hexane (1: 1) to give 4,4-difluoro-1-iodomethylcyclohexane (51% ethyl acetate solution, 5.6 g, 11 ol, quant.). Obtained as a brown oil. 'Ή-NMR (200MHz, CDC1 3): 61.35 (2H, brq, J = 10.6 Hz), 1.59 (2H, m), 1.80 (2H, m), 1.92 (2H, m), 2.23 (1H, m) , 3.13 / 3.19 (2H, d, J = 6.2 / 5.8 Hz).
参考例 23 Reference Example 23
ヨウ化 c i s_4- (2 -テトラヒドロピラニル)ォキシシクロへキシルメチル Cis_4- (2-tetrahydropyranyl) oxycyclohexylmethyl iodide
cis- 4 -ヒドロキシシクロへキサンカルボン酸 (4.00 g, 27.7 mmol) と 濃塩酸 (4.62 ml, 55.5匪 ol) のエタノール (80 ml) 溶液を室温で 19 時間攪拌した。 酢酸ェチルにより抽出で精製し、 cis- 4-ヒドロキシシクロへキサン力ルポン酸ェ チル(4.72 g, 27.4匪01, 98.9¾) の無色油状物を得た。 A solution of cis-4-hydroxycyclohexanecarboxylic acid (4.00 g, 27.7 mmol) and concentrated hydrochloric acid (4.62 ml, 55.5 mol / l) in ethanol (80 ml) was stirred at room temperature for 19 hours. Purification by extraction with ethyl acetate gave a colorless oily substance of cis-4-hydroxycyclohexanyl ethyl ruponate (4.72 g, 27.4 bandages 01, 98.9%).
¾-NMR (200MHz, CDC13): (51.26 (3H, t, J= 7.1 Hz), 1.36 (1H, brs), 1.67 (6H, m), 1.95 (2H, quint, J= 8.2 Hz), 2.38 (1H, m), 3.90 (1H, in), 4.14 (2H, q, J= 7.1 Hz). ¾-NMR (200MHz, CDC1 3 ): (51.26 (3H, t, J = 7.1 Hz), 1.36 (1H, brs), 1.67 (6H, m), 1.95 (2H, quint, J = 8.2 Hz), 2.38 (1H, m), 3.90 (1H, in), 4.14 (2H, q, J = 7.1 Hz).
cis- 4-ヒドロキシシクロへキサンカルボン酸ェチル (2.65 g, 15.4歷 ol) と 3, 4 -ジヒドロ- 2H-ピラン (以下 DHPと略記することがある, 1.72 ml, 18.5 mmol) のクロ口ホルム (80 ml) 溶液を Dカンファ一- 10-スルホン酸 (以下 CSAと略記 することがある, 357 mg, 1.54 mmol)と室温で 3 時間攪拌した。 酢酸ェチルによ り抽出で精製し、 cis- 4- (2-テトラヒドロピラエル)ォキシシクロへキサン力ルポ ン酸ェチル(4.14 g, 15.4 mmol, quant. )の無色油状物を得た。 Ethyl cis-4-hydroxycyclohexanecarboxylate (2.65 g, 15.4 olol) and 3,4-dihydro-2H-pyran (hereinafter abbreviated as DHP, 1.72 ml, 18.5 mmol) The solution was stirred with D-camphor-10-sulfonic acid (hereinafter sometimes abbreviated as CSA, 357 mg, 1.54 mmol) at room temperature for 3 hours. Purification by extraction with ethyl acetate gave a colorless oily substance of ethyl cis-4- (2-tetrahydropyrael) oxycyclohexane hexane propionate (4.14 g, 15.4 mmol, quant.).
cis-4-(2-テトラヒドロビラニル)ォキシシクロへキサンカルボン酸ェチル (4.14 g, 15.4讓 ol) の THF (80 ml)溶液に 0°Cで攪拌しながら水素化リチウム アルミニウム(純度 80%, 0.402 g, 8.47 mmol) の粉末を数回に分けて加え、 (TC で 2.5時間攪拌した。酢酸ェチルにより抽出で精製し、 cis- 4- (2-テトラヒドロピ ラニル)ォキシシクロへキサンメタノール(純度 90%, 3.67 g, 15.4 mmol, quant.) の淡黄色油状物を得た。 Lithium aluminum hydride (purity 80%, 0.402) was added to a solution of ethyl cis-4- (2-tetrahydroviranyl) oxycyclohexanecarboxylate (4.14 g, 15.4 benzyl) in THF (80 ml) while stirring at 0 ° C. g, 8.47 mmol) was added in several portions, and the mixture was stirred for 2.5 hours with TC. The extract was purified by extraction with ethyl acetate, and cis-4- (2-tetrahydropyranyl) oxycyclohexanemethanol (purity 90%) was added. , 3.67 g, 15.4 mmol, quant.).
Ή - NMR (200MHz, CDC13): δ 1.40-1.75 (11H, m), 1.84 (4H, m), 3.50 (2H, m), 3.50 (2H, d, J= 6.0 Hz), 3.88 (2H, m), 3.92 (1H, m), 4.66/4.95 (1H, m). 約 3:1 ァノマ一混合物 Ή - NMR (200MHz, CDC1 3 ): δ 1.40-1.75 (11H, m), 1.84 (4H, m), 3.50 (2H, m), 3.50 (2H, d, J = 6.0 Hz), 3.88 (2H, m), 3.92 (1H, m), 4.66 / 4.95 (1H, m). 3: 1 panoma mixture
c i s-4- (2-テトラヒドロピラニル)ォキシシクロへキサンメタノール (純度 90%, 3.67 g, 15.4 mmol)およびトリェチルァミン(2.56 ml, 18.5 mmol) の THF (70 ml) 溶液に 0 で攪拌しながらメタンスルホニルクロリド(1.87 g, 16.2薩 ol)の THF (5 ml)溶液を 20分間で滴下し、 反応液を 0°Cで 2.5時間攪拌した。 酢酸ェチルに より抽出して得た粗物質(5.27 g)をシリカゲル(50 g) のクロマトグラフィー(溶 出溶媒、 酢酸ェチルーへキサン、 2:8-3:7) により精製し、 cis- 4- (2-テトラヒド ロビラニル)ォキシシクロへキシルメチルメシレ一卜(3.76 g, 12.9匪 ol, 83.5%) の淡黄色油状物を得た。 Cis-4- (2-tetrahydropyranyl) oxycyclohexanemethanol (purity 90%, 3.67 g, 15.4 mmol) and triethylamine (2.56 ml, 18.5 mmol) in THF (70 ml) were stirred at 0 with methane. A solution of sulfonyl chloride (1.87 g, 16.2 ml) in THF (5 ml) was added dropwise over 20 minutes, and the reaction solution was stirred at 0 ° C for 2.5 hours. The crude material (5.27 g) obtained by extraction with ethyl acetate was purified by silica gel (50 g) chromatography (elution solvent, ethyl acetate-hexane, 2: 8-3: 7), and cis-4- A pale yellow oil of (2-tetrahydrobilanyl) oxycyclohexylmethyl mesylate (3.76 g, 12.9 ol, 83.5%) was obtained.
¾— NMR (300MHz, CDC13): δ 1.41 (2Η, in), 1.50-1.62 (8H, m), 1.70 (1H, m), 1.84 (4H, m), 3.01 (3H, s), 3.49 (2H, dt, J= 11.7, 5.1 Hz), 3.90 (2H, m), 4.06 (2H, d, J= 6.6 Hz), 4.65 (1H, t, J= 3.8 Hz). ¾- NMR (300MHz, CDC1 3) : δ 1.41 (2Η, in), 1.50-1.62 (8H, m), 1.70 (1H, m), 1.84 (4H, m), 3.01 (3H, s), 3.49 ( 2H, dt, J = 11.7, 5.1 Hz), 3.90 (2H, m), 4.06 (2H, d, J = 6.6 Hz), 4.65 (1H, t, J = 3.8 Hz).
c i s-4_ (2-テトラヒドロビラニル)ォキシシクロへキシルメチルメシレート (3.76 g, 12.9 mmol)およびヨウ化ナトリウム(3.85 g, 25.7蘭 ol) の アセトン (70 ml)懸濁液を 18時間加熱還流した。 反応液を酢酸ェチルにより抽出して精製 し、ヨウ化 cis- 4-(2-テトラヒドロビラニル)ォキシシクロへキシルメチル (3.84 g, 11.8 mmol, 91.8%)の淡黄色油状物を得た。 A suspension of ci s-4_ (2-tetrahydroviranyl) oxycyclohexylmethyl mesylate (3.76 g, 12.9 mmol) and sodium iodide (3.85 g, 25.7 ol) in acetone (70 ml) was heated at reflux for 18 hours. did. The reaction solution was extracted with ethyl acetate and purified to give a pale yellow oil of cis-4- (2-tetrahydroviranyl) oxycyclohexylmethyl iodide (3.84 g, 11.8 mmol, 91.8%).
Ή-NMR (200MHz, CDC13): δ 1.39 (2Η, brq, J= 8.4 Hz), 1.50-1.65 (8H, m), 1.69 (1H, m), 1.84 (4H, m), 3.13 (2H, d, J= 5.6 Hz), 3.48 (2H, dt, J= 11.4, 5.8 Hz), 3.84 (1H, m), 3.92 (1H, m), 4.65 (1H, brt, J= 4 Hz). Ή-NMR (200MHz, CDC1 3 ): δ 1.39 (2Η, brq, J = 8.4 Hz), 1.50-1.65 (8H, m), 1.69 (1H, m), 1.84 (4H, m), 3.13 (2H, d, J = 5.6 Hz), 3.48 (2H, dt, J = 11.4, 5.8 Hz), 3.84 (1H, m), 3.92 (1H, m), 4.65 (1H, brt, J = 4 Hz).
参考例 24 Reference Example 24
ヨウ化 cis- 5-ァセトキシシクロォクチル Cis-5-Acetoxycyclooctyl iodide
cis- 1,5 -シクロオクタンジオール (2.01 g, 13.7 mmol) と p-トルエンスルホ ニルクロリド(2.95 g, 15.0匪 ol) のピリジン (30 ml) 溶液を室温で 16時間攪 拌した。 反応液を濃縮乾固後、 酢酸ェチルにより抽出し、 残渣の油状物 (4.2 g) をシリカゲル(50 g)のクロマトグラフィー (酢酸ェチルーへキサン、 2:8-4:6) で 精製し、 cis-1, 5 -シクロオクタンジオールモノトシレート(2.27 g, 7.61 mmol, 55.7%) の無色油状物を得た。 A solution of cis-1,5-cyclooctanediol (2.01 g, 13.7 mmol) and p-toluenesulfonyl chloride (2.95 g, 15.0 marl) in pyridine (30 ml) was stirred at room temperature for 16 hours. The reaction mixture was concentrated to dryness, extracted with ethyl acetate, and the residual oil (4.2 g) was purified by silica gel (50 g) chromatography (ethyl acetate-hexane, 2: 8-4: 6) to give cis. A colorless oil of -1,5-cyclooctanediol monotosylate (2.27 g, 7.61 mmol, 55.7%) was obtained.
Ή-NMR (200MHz, CDC13): 51.40 (2H, brq,; J= 8.2 Hz), 1.59 (2H, m), 1.82 (8H, m), 2.45 (3H, s), 3.75 (1H, brt, J= 8.8 Hz), 4.59 (1H, m), 7.34 (2H, d, J= 8.8 Hz), 7.79 (2H, d, J= 8.0 Hz). Ή-NMR (200MHz, CDC1 3 ): 51.40 (2H, brq,; J = 8.2 Hz), 1.59 (2H, m), 1.82 (8H, m), 2.45 (3H, s), 3.75 (1H, brt, J = 8.8 Hz), 4.59 (1H, m), 7.34 (2H, d, J = 8.8 Hz), 7.79 (2H, d, J = 8.0 Hz).
cis- 1,5-シクロオクタンジオールモノトシレー卜 (0.89 g, 2.98腿01) と 無 水酢酸 (5 ml) のピリジン (10 ml) 溶液を室温で 12時間攪拌した。 反応液を濃縮 乾固して、 cis- 5-ァセトキシシクロォクチルトシレート(1.1 g, quant.) の無色 油状物を得た。 A pyridine (10 ml) solution of cis-1,5-cyclooctanediol monotosylate (0.89 g, 2.98 thigh 01) and anhydrous acetic acid (5 ml) was stirred at room temperature for 12 hours. The reaction solution was concentrated to dryness to obtain a colorless oily product of cis-5-acetoxycyclooctyl tosylate (1.1 g, quant.).
Ή-NMR (200MHz, CDC13): (51.44 (2H, m), 1.60-1.90 (10H, m), 2.00 (3H, s), 2.45 (3H, s), 4.61 (1H, m), 4.77 (1H, HI), 7.34 (2H, d, J= 8.0 Hz) , 7.79 (2H, d, J= 8.4 Hz). Ή-NMR (200MHz, CDC1 3 ): (51.44 (2H, m), 1.60-1.90 (10H, m), 2.00 (3H, s), 2.45 (3H, s), 4.61 (1H, m), 4.77 ( 1H, HI), 7.34 (2H, d, J = 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz).
cis- 5 -ァセトキシシクロォクチルトシレート(1.1 g, 2.98 mmol)およびヨウ化 ナトリウム(1.34 g, 8.94 mmol) の 2-ブ夕ノン (25 ml)懸濁液を 1.5時間加熱還 流した。反応液を酢酸ェチルにより抽出してヨウ化 cis- 5-ァセトキシシクロォク チル (純度 60 , 0.81 g, 1.6 mmol, 55%)の褐色油状物を得た。 A suspension of cis-5-acetoxycyclooctyl tosylate (1.1 g, 2.98 mmol) and sodium iodide (1.34 g, 8.94 mmol) in 2-butaneone (25 ml) was heated under reflux for 1.5 hours. . The reaction solution was extracted with ethyl acetate to obtain a brown oily substance of cis-5-acetoxycyclooctyl iodide (purity 60, 0.81 g, 1.6 mmol, 55%).
参考例 25 Reference Example 25
(2S, 4S) - N-Boc-4-フルォロ- 2 -ョ一ドメチルピロリジン . (2S, 4S) -N-Boc-4-fluoro-2-fluoromethylpyrrolidine.
Deoxd-Fluor™ (1.20 g, 5.42卿 ol)のジクロロメタン (10 ml)の溶液に 0 で 攪拌しながら (2S, 4R) -N-Boc-4-ヒドロキシピロリジン- 2-メチルトシレート (1.68 g, 4.52腿 ol)のジクロロメタン (10 ml)溶液を 45分間かけて滴下し、 昇 温後、 反応液を室温で 10時間攪拌した。 反応液を炭酸ナトリウム(1.51 g, 18.1 謹 ol)を溶解した氷水(約 80 g)に注ぎ、 酢酸ェチルーへキサン(1:1, 60 nil)およ び酢酸ェチル(50 ml)で抽出した。有機層を飽和炭酸水素ナトリゥムおよび飽和食 塩水で洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して黄色油状物を得た。 これをシ リカゲル(20 g)のクロマトグラフィ一(酢酸ェチル一へキサン、 1: 9-4: 6)に付し、 酢酸ェチルーへキサン(2:8- 3:7) の画分を濃縮乾固後、 ジェチルェ一テルで希釈 した。 析出物を濾取し、 ジェチルエーテルで洗浄後、 乾燥して(2S,4S)-N- Boc - 4_ フルォロピロリジン- 2-メチルトシレ一ト(1.30 g, 3.48匪01, 77.0%)を白色粉末 として得た。 A solution of Deoxd-Fluor ™ (1.20 g, 5.42 rol) in dichloromethane (10 ml) was stirred at 0 with (2S, 4R) -N-Boc-4-hydroxypyrrolidine-2-methyltosylate (1.68 g, A solution of 4.52 mol) in dichloromethane (10 ml) was added dropwise over 45 minutes, and after heating, the reaction solution was stirred at room temperature for 10 hours. The reaction solution was poured into ice-water (about 80 g) in which sodium carbonate (1.51 g, 18.1 mol) was dissolved, and extracted with ethyl acetate-hexane (1: 1, 60 nil) and ethyl acetate (50 ml). The organic layer was washed with saturated sodium bicarbonate and saturated brine, dried over sodium sulfate, and concentrated to give a yellow oil. This was subjected to chromatography on silica gel (20 g) (ethyl acetate-hexane, 1: 9-4: 6), and the ethyl acetate-hexane (2: 8-3: 7) fraction was concentrated to dryness. Later, it was diluted with Jetil ether. The precipitate was collected by filtration, washed with getyl ether, and dried to give (2S, 4S) -N-Boc-4_fluoropyrrolidine-2-methyltosylate (1.30 g, 3.48 bandages 01, 77.0%) Obtained as a white powder.
Ή-NMR (200MHz, CDC13): 51.43 (9H, s), 2.00 (1H, m), 2.28 (1H, brd, J= 18.4 Hz), 2.45 (3H, s), 3.58 (1H, brd, J= 27.4 Hz), 3.87 (1H, m), 4.14 (1H, m), 4.29 (1H, m), 5.17 (1H, brd, J= 52.4 Hz), 7.35 (2H, d, J= 8.0 Hz), 7.80 (2H, d, J= 8.4 Hz). Ή-NMR (200MHz, CDC1 3 ): 51.43 (9H, s), 2.00 (1H, m), 2.28 (1H, brd, J = 18.4 Hz), 2.45 (3H, s), 3.58 (1H, brd, J = 27.4 Hz), 3.87 (1H, m), 4.14 (1H, m), 4.29 (1H, m), 5.17 (1H, brd, J = 52.4 Hz), 7.35 (2H, d, J = 8.0 Hz), 7.80 (2H, d, J = 8.4 Hz).
元素分析値 C17H24N05SFとして計算値: C, 54.68; H, 6.48; N, 3.75; S, 8.59; F, 5.09 (%) 、 実測値: C, 54.61; H, 6.60; N, 3.70; S, 8.66; F, 4.80 (%) . Elemental analysis C 17 H 24 N0 5 SF Calculated: C, 54.68; H, 6.48 ; N, 3.75; S, 8.59; F, 5.09 (%), Found: C, 54.61; H, 6.60 ; N, 3.70; S, 8.66; F, 4.80 (%).
(2S,4S)- N-Boc-4_フルォロピロリジン- 2-メチルトシレート(1.20 g, 3.21 腿 ol)およびヨウ化ナトリウム(1.44 g, 9.64匪 ol) の アセトン (24 ml)懸濁液 を室温で 12時間攪拌後、 8時間加熱還流した。反応液をジイソプロピルエーテル により抽出して(2S, 4S) -N_Boc- 4_フルォロ- 2-ョードメチルピ口リジンの粗物質 (1.00 g)を黄色油状物として得た。 Suspension of (2S, 4S) -N-Boc-4_fluoropyrrolidine-2-methyltosylate (1.20 g, 3.21 tall ol) and sodium iodide (1.44 g, 9.64 marl ol) in acetone (24 ml) After the solution was stirred at room temperature for 12 hours, it was heated and refluxed for 8 hours. The reaction solution was extracted with diisopropyl ether to obtain a crude substance (1.00 g) of (2S, 4S) -N_Boc-4_fluoro-2-iodomethylpiperidine as a yellow oil.
参考例 26 Reference Example 26
5, 5-ジフルォロシクロォクチルトシレート 5,5-difluorocyclooctyl tosylate
塩化ォキザリル (1.27ml, 14.1匪 ol)のジクロロメタン (80 ml)の溶液に- 78°C で攪拌しながら ジメチルスルホキシド (1.36 ml, 19.0 mmol)のジクロロメタン (6 ml)溶液を 5分間かけて滴下し、 10分後、 cis_l,5 -シクロオクタンジオールモ ノトシレ一ト (2.10 g, 7.04讓 ol) のジクロロメタン (30 ml)溶液を 20分間か けて滴下した。 反応液を室温で 2 時間攪拌した後、 反応液にトリェチルァミン (7.12 ml, 51.4 mmol)を滴下した。 反応液を 30分間かけて 0°Cまで昇温し、 20 分間攪拌した。 反応液を氷水(100 g)に注ぎ、 上層を酢酸ェチル (100 ml)で抽出 した。 酢酸ェチル層を飽和食塩水で洗浄後、 下層と合わせ、 濃縮してジクロロメ タンを留去し、 酢酸ェチル (150 ml)で希釈後、 5%および飽和の食塩水で順次洗浄 し、 硫酸ナトリウムで乾燥後、 濃縮して黄色固形物を得た。 これをジェチルェ一 テル—ジィソプロピルェ一テル中で粉碎して濾取し、 ジィソプロピルエーテルで 洗浄後、乾燥して 5-ォキソシクロォクチルトシレート(1.63 g, 5.51匪01, 78.3%) を白色粉末として得た。 To a solution of oxalyl chloride (1.27 ml, 14.1 ol) in dichloromethane (80 ml) was added dropwise a solution of dimethyl sulfoxide (1.36 ml, 19.0 mmol) in dichloromethane (6 ml) over 5 minutes while stirring at -78 ° C. After 10 minutes, a solution of cis_l, 5-cyclooctanediol monotosylate (2.10 g, 7.04 benzyl) in dichloromethane (30 ml) was added dropwise over 20 minutes. After stirring the reaction solution at room temperature for 2 hours, triethylamine (7.12 ml, 51.4 mmol) was added dropwise to the reaction solution. The reaction solution was heated to 0 ° C over 30 minutes and stirred for 20 minutes. The reaction solution was poured into ice water (100 g), and the upper layer was extracted with ethyl acetate (100 ml). The ethyl acetate layer was washed with saturated saline, combined with the lower layer, concentrated to distill off dichloromethane, diluted with ethyl acetate (150 ml), washed successively with 5% and saturated saline, and washed with sodium sulfate. After drying, concentration gave a yellow solid. This is pulverized in diethyl propyl ether, collected by filtration, washed with diisopropyl ether, and dried to give 5-oxocyclooctyl tosylate (1.63 g, 5.51 bandat 01, 78.3%). Obtained as a white powder.
Ή-NMR (200MHz, CDC13): (51.71 (2H, m), 1.82 (2H, t, J= 5.0 Hz), 1.85 (2H, t, J= 5.0 Hz), 1.91 (2H, m), 2.26 (2H, ddd, J= 13.6, 8.8, 3.6 Hz), 2.51 (2H, ddd, J= 13.2, 8.8, 3.6 Hz), 4.30 (1H, quint, J= 5.9 Hz), 7.35 (2H, d, J= 8.8 Hz), 7.76 (2H, d, J= 8.4 Hz). Ή-NMR (200MHz, CDC1 3 ): (51.71 (2H, m), 1.82 (2H, t, J = 5.0 Hz), 1.85 (2H, t, J = 5.0 Hz), 1.91 (2H, m), 2.26 (2H, ddd, J = 13.6, 8.8, 3.6 Hz), 2.51 (2H, ddd, J = 13.2, 8.8, 3.6 Hz), 4.30 (1H, quint, J = 5.9 Hz), 7.35 (2H, d, J = 8.8 Hz), 7.76 (2H, d, J = 8.4 Hz).
元素分析値: C15H2004S として計算値: C, 60.79; H, 6.80; S, 10.82 (%) 、 実 測値: C, 60.79; H, 6.92; S, 10.95 (%) . Elemental analysis: C 15 H 20 0 4 S Calculated: C, 60.79; H, 6.80 ; S, 10.82 (%), actual Found: C, 60.79; H, 6.92; S, 10.95 (%).
5 -ォキソシクロォクチルトシレート(1.26 g, 4.25 mmol)のジクロロメタン (10 ml)の溶液に 0でで攪拌しながら Deoxo-Fluor™ (1.88 g, 8.50 mmol)のジクロ口 メタン (5 ml)溶液を 30分間かけて滴下し、 昇温後、 反応液を室温で 6日間攪拌 し、 さらに 2 日間加熱還流した。 反応液を炭酸ナトリウム(2.14 g, 25.5 mmol) を溶解した氷水(約 50 g)に注ぎ、 酢酸ェチルージェチルエーテル α:1, 0 ml) および酢酸ェチル (30 ml)で抽出した。有機層を飽和炭酸水素ナトリゥムおよび飽 和食塩水で洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して黄色油状物を得た。 これ をシリカゲル(15 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:9-4:6) に 付し、酢酸ェチルーへキサン(1:9) の画分を濃縮乾固して 5, 5-ジフルォロシクロ ォクチルトシレート (純度 85%, 0.48 g, 1.28腿 ol, 30%)を淡黄色油状物として 得た。 To a solution of 5-oxocyclooctyl tosylate (1.26 g, 4.25 mmol) in dichloromethane (10 ml) while stirring at 0 with Deoxo-Fluor ™ (1.88 g, 8.50 mmol) in dichloromethane (5 ml) The solution was added dropwise over 30 minutes. After the temperature was raised, the reaction solution was stirred at room temperature for 6 days, and further heated under reflux for 2 days. The reaction solution was poured into ice water (about 50 g) in which sodium carbonate (2.14 g, 25.5 mmol) was dissolved, and extracted with ethyl acetate-ethyl ether α: 1.0 ml) and ethyl acetate (30 ml). The organic layer was washed with saturated sodium bicarbonate and saturated saline, dried over sodium sulfate, and concentrated to give a yellow oil. This was subjected to chromatography on silica gel (15 g) (ethyl acetate-hexane, 1: 9-4: 6), and the ethyl acetate-hexane (1: 9) fraction was concentrated to dryness to give 5,5-hexane. Difluorocyclooctyl tosylate (purity 85%, 0.48 g, 1.28 t, 30%) was obtained as a pale yellow oil.
'H-NMR (200MHz, CDC13): (51.58 (2H, m), 1.68-2.12 (10H, m), 2.46 (3H, s), 4.63 (1H, tt, J= 8.0, 3.8 Hz), 7.34 (2H, d, J= 8.4 Hz), 7.79 (2H, d, J- 8.4 Hz) . 'H-NMR (200MHz, CDC1 3): (51.58 (2H, m), 1.68-2.12 (10H, m), 2.46 (3H, s), 4.63 (1H, tt, J = 8.0, 3.8 Hz), 7.34 (2H, d, J = 8.4 Hz), 7.79 (2H, d, J- 8.4 Hz).
参考例 27 Reference Example 27
3 -ョードメチルァダマン夕ン- 1-メチルァセテ一ト 3-Methyl methyl adaman-1-methyl acetate
ァダマンタン- 1,3-ジメタノール(1.53 g, 7.79 mmol)および p -トルエンスルホ ニルクロリド (1.69 g, 8.57腿 ol)の ピリジン (20 ml)溶液を室温で 14時間攪 拌した。 反応液にトリェチルァミン(0.063 ml, 0.455 腿 ol) を加え、 さらに 2 時間攪拌した。 反応液から酢酸ェチルにより抽出で得たゲル状物にジェチルエー テル加え、 析出物を除去した後、 母液をシリカゲル(25 g)のクロマトグラフィー (酢酸ェチルーへキサン、 2:8-4:6) に付した。酢酸ェチルーへキサン(4: 6) の画 分を濃縮乾固、 乾燥して 3 -ヒドロキシメチルァダマンタン- 1-メチルトシレート (1.72 g, 4.91 匪 ol, 63.0%)を無色油状物として得た。 A solution of adamantane-1,3-dimethanol (1.53 g, 7.79 mmol) and p-toluenesulfonyl chloride (1.69 g, 8.57 mol) in pyridine (20 ml) was stirred at room temperature for 14 hours. Triethylamine (0.063 ml, 0.455 t) was added to the reaction solution, and the mixture was further stirred for 2 hours. Getyl ether was added to the gel obtained by extraction from the reaction mixture with ethyl acetate, and the precipitate was removed. The mother liquor was subjected to silica gel (25 g) chromatography (ethyl acetate-hexane, 2: 8-4: 6). Attached. The fraction of ethyl acetate-hexane (4: 6) was concentrated to dryness and dried to give 3-hydroxymethyladamantane-1-methyltosylate (1.72 g, 4.91 hexane, 63.0%) as a colorless oil. Was.
¾-NMR (200MHz, CDC13): δ 1.25 (3Η, m), 1.44 (8H, br), 1.60 (2H, brs), 2.08 (2H, m), 2.45 (3H, s), 3.21 (2H, d, J= 6.0 Hz), 3.60 (2H, s), 7.34 (2H, d, J= 8.4 Hz), 7.77 (2H, d, J= 8.4 Hz). ' ¾-NMR (200MHz, CDC1 3 ): δ 1.25 (3Η, m), 1.44 (8H, br), 1.60 (2H, brs), 2.08 (2H, m), 2.45 (3H, s), 3.21 (2H, d, J = 6.0 Hz), 3.60 (2H, s), 7.34 (2H, d, J = 8.4 Hz), 7.77 (2H, d, J = 8.4 Hz).
3 -ヒドロキシメチルァダマンタン- 1-メチルトシレ一卜 (1.72 g, 4.91 mmol) と 無水酢酸 (5 ml) のピリジン (10 ml) 溶液を室温で 12時間攪拌した。 反応液 を濃縮乾固して、 3-ァセトキシメチルァダマンタン-卜メチルトシレート(1.89 g, 4.91 mmol, 98.0¾) の白色固形物を得た。 3-Hydroxymethyladamantane-1-methyltosylate (1.72 g, 4.91 mmol) And a solution of acetic anhydride (5 ml) in pyridine (10 ml) were stirred at room temperature for 12 hours. The reaction solution was concentrated to dryness to obtain a white solid of 3-acetoxymethyladamantane-methyltosylate (1.89 g, 4.91 mmol, 98.0%).
Ή-NMR (200MHz, CDC13): 51.27 (2H, s), 1.44 (8H, br), 1.60 (2H, brs), 2.06 (3H, s), 2.07 (2H, brs), 2.46 (3H, s), 3.59 (2H, s), 3.67 (2H, s), 7.35 (2H, d, J= 7.6 Hz) , 7.77 (2H, d, J= 8.4 Hz) . Ή-NMR (200MHz, CDC1 3 ): 51.27 (2H, s), 1.44 (8H, br), 1.60 (2H, brs), 2.06 (3H, s), 2.07 (2H, brs), 2.46 (3H, s ), 3.59 (2H, s), 3.67 (2H, s), 7.35 (2H, d, J = 7.6 Hz), 7.77 (2H, d, J = 8.4 Hz).
3 -ァセトキシメチルァダマンタン- 1-メチルトシレ一ト(1.51 g, 3.85 mmol)お よびヨウ化ナトリウム(1.73 g, 11.5腿 ol) の 2-ブタノン (40 ml)懸濁液を 18 時間加熱還流した。 反応液を酢酸ェチルにより抽出して褐色油状を得、 ついでシ リカゲル(15 g)のクロマトグラフィー (酢酸ェチルーへキサン、 0:100-10:90) に 付した。 酢酸ェチルーへキサン(3:97) の画分を濃縮乾固、 乾燥して 3-ョードメ チルァダマンタン- 1-メチルアセテート(768 mg, 2.21匪01, 57.3%)を無色油状物 として得た。 Heat a suspension of 3-acetoxymethyladamantane-1-methyltosylate (1.51 g, 3.85 mmol) and sodium iodide (1.73 g, 11.5 tmol) in 2-butanone (40 ml) for 18 hours. Refluxed. The reaction solution was extracted with ethyl acetate to obtain a brown oil, which was then subjected to silica gel (15 g) chromatography (ethyl acetate-hexane, 0: 100-10: 90). The fraction of ethyl acetate-hexane (3:97) was concentrated to dryness and dried to give 3-odomethytyl adamantane-1-methyl acetate (768 mg, 2.21 bandages 01, 57.3%) as a colorless oil.
Ή-NMR (200MHz, CDC13): δΐ.31 (2Η, brs), 1.43 (4H, brd, J= 2.2 Hz), 1.49 (4H, brs), 1.58 (2H, brs), 2.07 (3H, s), 2.07 (2H, brs), 3.05 (2H, s), 3.73 (2H, s). Ή-NMR (200MHz, CDC1 3 ): δΐ.31 (2Η, brs), 1.43 (4H, brd, J = 2.2 Hz), 1.49 (4H, brs), 1.58 (2H, brs), 2.07 (3H, s ), 2.07 (2H, brs), 3.05 (2H, s), 3.73 (2H, s).
参考例 28 Reference Example 28
3-ョ一ドメチルシクロへキサン- 1-メチルァセテ一ト 3-hydroxymethylcyclohexane-1-methyl acetate
シクロへキサン- 1, 3-ジカルポン酸 (c is/trans混合物、純度 90%, 2.6 g, 13.8 mmol)と 濃塩酸 (2.30 ml, 27.6 mmol) のメタノール (50 ml) 溶液を室温で 13 時間攪拌した。 反応液に炭酸水素ナトリウムを加えて中和後、 酢酸ェチル抽出に より精製し、 シクロへキサン- 1,3 -ジカルポン酸ジメチル (cis/trans 混合物、 純 度 96%, 2.85 g, 13.7 mmol, 99%)の無色油状物を得た。 A solution of cyclohexane-1,3-dicarponic acid (cis / trans mixture, purity 90%, 2.6 g, 13.8 mmol) and concentrated hydrochloric acid (2.30 ml, 27.6 mmol) in methanol (50 ml) was stirred at room temperature for 13 hours. did. The reaction mixture was neutralized with sodium bicarbonate and purified by extraction with ethyl acetate.Dimethyl cyclohexane-1,3-dicarbonate (cis / trans mixture, purity 96%, 2.85 g, 13.7 mmol, 99 %) As a colorless oil.
lH-NMR (200MHz, CDC13): δΐ.35 (2Η, m), 1.57 (3/2H, m), 1.72 (3/2H, m), 1.92 (1H, i), 1.97 (2H, t, J= 8.6 Hz), 2.23 (1H, dm, J= 12.8 Hz), 2.33 (2H, m) 2.69 (1H, quint, J= 5.8 Hz), 3.67 (3H, s), 3.68 (3H, s). lH-NMR (200MHz, CDC1 3 ): δΐ.35 (2Η, m), 1.57 (3 / 2H, m), 1.72 (3 / 2H, m), 1.92 (1H, i), 1.97 (2H, t, J = 8.6 Hz), 2.23 (1H, dm, J = 12.8 Hz), 2.33 (2H, m) 2.69 (1H, quint, J = 5.8 Hz), 3.67 (3H, s), 3.68 (3H, s).
シクロへキサン- 1,3-ジカルポン酸ジメチル(2.85 g, 13.7 mmol) の THF (60 ml)溶液に 0 で攪拌しながら水素化リチウムアルミニウム (純度 80¾, 729 mg, 15.4膽01) の粉末を数回に分けて加え、 0。Cで 90分間攪拌した。 酢酸ェチルによ り抽出、 濃縮乾固してシクロへキサン- 1,3-ジメタノール(cis/trans混合物、 純 度 90%, 2.20 g, 13.7 mmol, quant. )の無色油状物を得た。 While stirring at 0 with a solution of dimethyl cyclohexane-1,3-dicarbonate (2.85 g, 13.7 mmol) in THF (60 ml), several powders of lithium aluminum hydride (purity 80729, 729 mg, 15.4 膽 01) were added. Add 0 times. Stirred at C for 90 minutes. With ethyl acetate Extraction and concentration to dryness gave a colorless oil of cyclohexane-1,3-dimethanol (cis / trans mixture, purity 90%, 2.20 g, 13.7 mmol, quant.).
Ή-NMR (200MHz, CDC13): δ 0.89/0.66 (2Η, qd/q, J= 12.8, 4.0/12.1 Hz), 1.30-1.65 (7H, in), 1.74-1.91 (3H, m), 3.48/3.55 (4H, d, J= 6.2/6.8 Hz). 元素分析値: C12H2。02として計算値: C, 73.43; H, 10.27(%)、 実測値: C, 73.47; H, 10.56 (%) . Ή-NMR (200MHz, CDC1 3 ): δ 0.89 / 0.66 (2Η, qd / q, J = 12.8, 4.0 / 12.1 Hz), 1.30-1.65 (7H, in), 1.74-1.91 (3H, m), 3.48 /3.55 (4H, d, J = 6.2 / 6.8 Hz) elemental analysis:. C 12 H 2. 0 2 Calculated: C, 73.43; H, 10.27 (%), Found: C, 73.47; H, 10.56 (%).
シクロへキサン - 1, 3 -ジメタノール (純度 90%, 2.20 g, 13.7画01) のピリジン (25 ml)溶液に p-トルエンスルホニルクロリド (2.96 g, 15.1 ranol)のピリジン (5 ml)溶液を室温で 6時間かけて滴下し、 ついで反応液を 2時間攪拌した。 反応 液にトリェチルァミン(0.063 ml, 0.455職 ol) を加え、 さらに 2時間攪拌した。 反応液から酢酸ェチルにより抽出で得た粗物質(4.1 g)をシリ力ゲル (40 g)のクロ マトグラフィー (酢酸ェチルーへキサン、 2:8-4:6) で精製し、 3-ヒドロキシメチ ルシクロへキサン - 1 -メチルトシレート(cis/trans 混合物、 2.44 g, 8.17 mmol, 59.7%)を無色油状物として得た。 To a solution of cyclohexane-1,3-dimethanol (purity 90%, 2.20 g, 13.7 fraction 01) in pyridine (25 ml) was added a solution of p-toluenesulfonyl chloride (2.96 g, 15.1 ranol) in pyridine (5 ml). The mixture was added dropwise at room temperature over 6 hours, and then the reaction solution was stirred for 2 hours. Triethylamine (0.063 ml, 0.455 mol) was added to the reaction solution, and the mixture was further stirred for 2 hours. The crude substance (4.1 g) obtained by extraction with ethyl acetate from the reaction solution was purified by chromatography on silica gel (40 g) (ethyl acetate-hexane, 2: 8-4: 6) to give 3-hydroxymethyl Lecyclohexane-1-methyltosylate (cis / trans mixture, 2.44 g, 8.17 mmol, 59.7%) was obtained as a colorless oil.
Ή-NMR (200MHz, CDC13): (50.85/0.64 (2H, brq/q, J= 13.2/11.8 Hz), 1.24 (2H, m), 1.39 (1H, m), 1.42 (1H, t, J= 5.8 Hz), 1.62-1.82 (4H, m), 2.45 (3H, s), 3. 4/3.47 (2H, t, J= 5.8/7.2 Hz), 3.83/3.93 (2H, d, J= 5.8/7.0 Hz), 7.34 (2H, d, J= 8.2 Hz), 7.78 (2H, dt, J= 8.0, 1.8 Hz). Ή-NMR (200MHz, CDC1 3 ): (50.85 / 0.64 (2H, brq / q, J = 13.2 / 11.8 Hz), 1.24 (2H, m), 1.39 (1H, m), 1.42 (1H, t, J = 5.8 Hz), 1.62-1.82 (4H, m), 2.45 (3H, s), 3.4 / 3.47 (2H, t, J = 5.8 / 7.2 Hz), 3.83 / 3.93 (2H, d, J = 5.8 /7.0 Hz), 7.34 (2H, d, J = 8.2 Hz), 7.78 (2H, dt, J = 8.0, 1.8 Hz).
3 -ヒドロキシメチルシクロへキサン- 1-メチルトシレ一卜 (1.37g, 4.59腿 ol) と 無水酢酸 (5 ml) のピリジン (10 ml) 溶液を室温で 13時間攪拌した。 反応液 を濃縮乾固して、 3-ァセトキシメチルシクロへキサン- 1-メチルトシレート (cis/trans混合物、 1.52 g, 4.49 mmol, 98%) の無色油状物を得た。 A solution of 3-hydroxymethylcyclohexane-1-methyltosylate (1.37 g, 4.59 mol) and acetic anhydride (5 ml) in pyridine (10 ml) was stirred at room temperature for 13 hours. The reaction solution was concentrated to dryness to obtain a colorless oil of 3-acetoxymethylcyclohexane-1-methyltosylate (cis / trans mixture, 1.52 g, 4.49 mmol, 98%).
LH-NMR (300MHz, CDC13): δ 0.87/0.65 (2Η, q, J= 13.2/11.4 Hz) , 1.27/1.41 (2H, t, J= 11.6/6.0 Hz), 1.60-1.85 (5H, m), 1.98 (1H, m), 2.04 (3H, s), 2.45 (3H, s), 3.82/3.84 (2H, d/dd, J= 6.3/5.6, 0.9 Hz), 3.91/3.87 (2H, d, J= 6.6/3.6 Hz), 7.34 (2H, d, J= 7.8 Hz), 7.77 (2H, d, J= 8.1 Hz). LH-NMR (300MHz, CDC1 3 ): δ 0.87 / 0.65 (2Η, q, J = 13.2 / 11.4 Hz), 1.27 / 1.41 (2H, t, J = 11.6 / 6.0 Hz), 1.60-1.85 (5H, m ), 1.98 (1H, m), 2.04 (3H, s), 2.45 (3H, s), 3.82 / 3.84 (2H, d / dd, J = 6.3 / 5.6, 0.9 Hz), 3.91 / 3.87 (2H, d , J = 6.6 / 3.6 Hz), 7.34 (2H, d, J = 7.8 Hz), 7.77 (2H, d, J = 8.1 Hz).
3 -ァセトキシメチルシクロへキサン-卜メチルトシレート(1.52 g, 4.49腿 ol) およびヨウ化ナトリウム(2.06 g, 13.8 mmol) の アセトン (30 ml)懸濁液を 3 時間加熱還流した。 反応液を酢酸ェチル—ジイソプロピルエーテルにより抽出、 濃縮乾固して 3-ョードメチルシク口へキサン- 1-メチルァセテ一ト(c is/trans混 合物、 1.24 g, 4.18 imol, 91.2%)を無色油状物として得た。 A suspension of 3-acetoxymethylcyclohexane-methyltosylate (1.52 g, 4.49 mol) and sodium iodide (2.06 g, 13.8 mmol) in acetone (30 ml) was heated under reflux for 3 hours. The reaction solution was extracted with ethyl acetate-diisopropyl ether, Concentration to dryness yielded 3-odomethylcyclohexane-1-methylacetate (cis / trans mixture, 1.24 g, 4.18 imol, 91.2%) as a colorless oil.
'H-NMR (200MHz, CDC13): δ 0.90/0.70 (2Η, tq/q, J= 3.2, 12.4/12.0 Hz), 1.25-1.58 (4H, m), 1.65-1.80 (2H, m), 1.85/1.94 (1H, m), 2.06 (3H, s), 3.11/3.18 (2H, d, J= 6.2/6.6 Hz), 3.91/3.99 (2H, d, J= 6.4/7.4 Hz). 'H-NMR (200MHz, CDC1 3): δ 0.90 / 0.70 (2Η, tq / q, J = 3.2, 12.4 / 12.0 Hz), 1.25-1.58 (4H, m), 1.65-1.80 (2H, m), 1.85 / 1.94 (1H, m), 2.06 (3H, s), 3.11 / 3.18 (2H, d, J = 6.2 / 6.6 Hz), 3.91 / 3.99 (2H, d, J = 6.4 / 7.4 Hz).
参考例 29 Reference Example 29
3-フルォロメチルシク口へキサン- 1-メチルトシレート 3-fluoromethylcyclohexane-1-methyltosylate
Deoxo-Fluor™ (952 mg, 4.30腿 ol)のジクロロメタン (7 ml)の溶液に 0°Cで攪 拌しながら参考例 28 で得た 3-ヒドロキシメチルシクロへキサン- 1-メチルトシ レート(cis/trans混合物、 1.07 g, 3.59匪 ol)のジクロロメタン (4 ml)溶液を 80分間かけて滴下し、 反応液を 0°Cで 1.5時間攪拌した。 反応液を氷水 (約 50 g) に注ぎ、酢酸ェチル—ジィソプロピルエーテル(1: 1)およびジェチルエーテルで抽 出して得た油状物をシリカゲル ao g)のクロマトグラフィー (酢酸ェチルーへキ サン、 0:100-10:90) で精製して 3-フルォロメチルシクロへキサン-卜メチルトシ レート(cis/trans 混合物、 172 mg, 0.573匪 ol, 15.9%)を無色油状物として得 た。 ' The 3-hydroxymethylcyclohexane-1-methyl tosylate (cis / cis) obtained in Reference Example 28 was stirred at 0 ° C in a solution of Deoxo-Fluor ™ (952 mg, 4.30 tmol) in dichloromethane (7 ml). A solution of the trans mixture, 1.07 g, 3.59 bandol) in dichloromethane (4 ml) was added dropwise over 80 minutes, and the reaction solution was stirred at 0 ° C for 1.5 hours. The reaction solution was poured into ice water (about 50 g), and the oil obtained by extraction with ethyl acetate-diisopropyl ether (1: 1) and getyl ether was chromatographed on silica gel ao g) (ethyl acetate-hexane). 0: 100-10: 90) to give 3-fluoromethylcyclohexane-methyltosylate (cis / trans mixture, 172 mg, 0.573 bandol, 15.9%) as a colorless oil. '
Ή-NMR (200MHz, CDC13): ά 0.90/0.70 (2Η, q, J= 13.0/12.0 Hz) , 1.20-1.45 (3H, m), 1.60-1.85 (5H, m), 2.46 (3H, s), 3.84 (2H, d, J= 5.8 Hz), 4.20 (2H, dd, J= 47.2, 5.6 Hz), 7.35 (2H, d, J= 8.2 Hz), 7.78 (2H, d, J= 8.0 Hz). Ή-NMR (200MHz, CDC1 3 ): ά 0.90 / 0.70 (2Η, q, J = 13.0 / 12.0 Hz), 1.20-1.45 (3H, m), 1.60-1.85 (5H, m), 2.46 (3H, s ), 3.84 (2H, d, J = 5.8 Hz), 4.20 (2H, dd, J = 47.2, 5.6 Hz), 7.35 (2H, d, J = 8.2 Hz), 7.78 (2H, d, J = 8.0 Hz) ).
参考例 30 Reference Example 30
酢酸 5-ョ一ド- 3, 3-ジメチルペンチル Acetic acid 5-chloride-3,3-dimethylpentyl
3, 3-ジメチルダルタル酸無水物 (純度 95¾, 6.92 g, 46.2 腿 ol) の THF (140 ffll)溶液に 0°Cで攪拌しながら水素化リチウムアルミニウム (純度 80, 2.41 g, 50.9腿01) の粉末を数回に分けて加え、 (TCで 2時間、 室温で 13時間攪拌し、 さ らに 4.5時間加熱還流した。 反応液に酢酸ェチル (20 ml)を加えて攪拌後、 メタノ ール水で希釈し、 酢酸ェチル - THF (1:1)により抽出、 濃縮乾固して黄色油状物を 得た。 これをシリカゲル(50 g)のクロマトグラフィー (酢酸ェチルーへキサン、 4: 6および 5: 5) で精製して酢酸 5-ヒドロキシ- 3, 3-ジメチルぺンチル (純度 90 , 1.60 g, 8.3 mmol, 18 )を無色油状物として得た。 Ή-NMR (200MHz, CDC13): δθ.96 (6H, s), 1.56 (2H, t, J= 7.2 Hz), 1.60 (2H, t, J= 7.4 Hz), 2.04 (3H, s), 3.73 (2H, t, J= 7.5 Hz), 4.13 (2H, t, J= 7.5 Hz). Lithium aluminum hydride (purity 80, 2.41 g, 50.9 t) was added to a solution of 3,3-dimethyldaltaric anhydride (purity 95¾, 6.92 g, 46.2 t) in THF (140 ffll) while stirring at 0 ° C. ) Was added in several portions, and the mixture was stirred (TC for 2 hours, room temperature for 13 hours, and heated to reflux for 4.5 hours. Ethyl acetate (20 ml) was added to the reaction solution, and the mixture was stirred. The mixture was diluted with water, extracted with ethyl acetate-THF (1: 1) and concentrated to dryness to give a yellow oil, which was chromatographed on silica gel (50 g) (ethyl acetate-hexane, 4: 6 and Purification by 5: 5) gave 5-hydroxy-3,3-dimethylpentyl acetate (purity 90, 1.60 g, 8.3 mmol, 18) as a colorless oil. Ή-NMR (200MHz, CDC1 3 ): δθ.96 (6H, s), 1.56 (2H, t, J = 7.2 Hz), 1.60 (2H, t, J = 7.4 Hz), 2.04 (3H, s), 3.73 (2H, t, J = 7.5 Hz), 4.13 (2H, t, J = 7.5 Hz).
酢酸 5-ヒドロキシ- 3,3-ジメチルペンチル (純度 90¾, 1.60 g, 8.3i ol)および ρ-トルエンスルホニルクロリド (1.79 g, 9.09匪 ol)のピリジン(20 ml)溶液を室 温で 19時間攪拌した。 反応液から酢酸ェチルーへキサン(1:1)により抽出で得た 粗物質をシリカゲル(20 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1: 9-1: 3)で精製し、 5 -ァセ卜キシ- 3, 3 -ジメチルぺンチルトシレー h(1.82g, 5.54 mmol, 66.8%)を無色油状物として得た。 A solution of 5-hydroxy-3,3-dimethylpentyl acetate (purity 90¾, 1.60 g, 8.3 i ol) and ρ-toluenesulfonyl chloride (1.79 g, 9.09 marl ol) in pyridine (20 ml) was stirred at room temperature for 19 hours. did. The crude substance obtained by extracting the reaction solution with ethyl acetate-hexane (1: 1) was purified by chromatography on silica gel (20 g) (ethyl acetate-hexane, 1: 9-1: 3) to give 5-acetate. Toxyl-3,3-dimethylpentyl silyl h (1.82 g, 5.54 mmol, 66.8%) was obtained as a colorless oil.
Ή-N R (200MHz, CDC13): 50.91 (6H, s), 1.53 (2H, t, J= 7.3 Hz), 1.63 (2H, t, J= 7.2 Hz), 2.02 (3H, s), 2.46 (3H, s), 4.05 (2H, t, J= 7.4 Hz), 4.10 (2H, t, J= 7.3 Hz), 7.35 (2H, d, J= 8.4 Hz), 7.79 (2H, d, J= 8.4 Hz). Ή-NR (200MHz, CDC1 3 ): 50.91 (6H, s), 1.53 (2H, t, J = 7.3 Hz), 1.63 (2H, t, J = 7.2 Hz), 2.02 (3H, s), 2.46 ( 3H, s), 4.05 (2H, t, J = 7.4 Hz), 4.10 (2H, t, J = 7.3 Hz), 7.35 (2H, d, J = 8.4 Hz), 7.79 (2H, d, J = 8.4) Hz).
5-ァセ卜キシ -3, 3-ジメチルペンチルトシレート (1.82 g, 5.54 mmol)およびョ ゥ化ナトリウム(2.49 g, 16.6 mmol) の アセトン (40 ml)懸濁液を 3時間加熱還 流した。 反応液を酢酸ェチルーへキサン(1:1)により抽出して粗製の酢酸 5-ョー ド- 3, 3_ジメチルペンチル (純度 90%, 1.50 g, 4.75 mmol, 85.8%)を黄色油状物と して得た。 A suspension of 5-acetoxy-3,3-dimethylpentyl tosylate (1.82 g, 5.54 mmol) and sodium iodide (2.49 g, 16.6 mmol) in acetone (40 ml) was heated under reflux for 3 hours. . The reaction solution was extracted with ethyl acetate-hexane (1: 1) to give crude 5-chloride-3,3-dimethylpentyl acetate (purity 90%, 1.50 g, 4.75 mmol, 85.8%) as a yellow oil. I got it.
'H-NMR (300MHz, CDC13): 50.93 (6H, s), 1.57 (2H, t, J= 7.2 Hz), 1.94 (2H, m), 2.06 (3H, s), 3.16 (2H, m), 4.10 (2H, t, J= 7.2 Hz). 'H-NMR (300MHz, CDC1 3): 50.93 (6H, s), 1.57 (2H, t, J = 7.2 Hz), 1.94 (2H, m), 2.06 (3H, s), 3.16 (2H, m) , 4.10 (2H, t, J = 7.2 Hz).
参考例 31 Reference Example 31
ト(2 -ョードェチル) 7ダマンタン G (2-Edoetil) 7 Damantan
ァダマンタン-卜酢酸 (10.0 g, 51.0画 ol)と 濃塩酸 (6.37 ml, 76.4 mmol) の メタノール (150 ml) 溶液を室温で 2.5日間攪拌した。 反応液に炭酸水素ナトリ ゥムを加えて中和後、酢酸ェチル抽出により精製し、ァダマンタン- 1-酢酸メチル (10.8 g, quant.)の無色油状物を得た。 A solution of adamantane-acetic acid (10.0 g, 51.0 mmol) and concentrated hydrochloric acid (6.37 ml, 76.4 mmol) in methanol (150 ml) was stirred at room temperature for 2.5 days. The reaction solution was neutralized by adding sodium hydrogen carbonate, and purified by extraction with ethyl acetate to obtain a colorless oil of adamantan-1-methyl acetate (10.8 g, quant.).
Ή-NMR (200MHz, CDC13): (51.61 (6H, d, J= 2.6 Hz), 1.67 (6H, br), 1.97 (3H, br), 2.08 (2H, s), 3.65 (3H, s). Ή-NMR (200MHz, CDC1 3 ): (51.61 (6H, d, J = 2.6 Hz), 1.67 (6H, br), 1.97 (3H, br), 2.08 (2H, s), 3.65 (3H, s) .
ァダマンタン-卜酢酸メチル(10.8 g, 51.0匪 ol) の THF (200 ml)溶液に 0°C で攪拌しながら水素化リチウムアルミニウム (純度 80%, 1.45 g, 30.6 mmol) の 粉末を数回に分けて加え、 Otで 90分間攪拌した。酢酸ェチル抽出により精製し、 ァダマンタン-卜エタノール(9, 21 g, 51.0 mmol, quant.)の白色粉末を得た。 Ή-N R (300MHz, CDC"): 51.10 (1H, br), 1.38 (2H, t, J= 7.5 Hz), 1.52 (6H, d, J= 2.4 Hz), 1.63 (3H, brd, J= 13.5 Hz), 1.70 (3H, brd, J= 12.3 Hz), 1.94 (3H, brs), 3.71 (2H, t, J= 7.5 Hz). A solution of lithium aluminum hydride (purity 80%, 1.45 g, 30.6 mmol) in a solution of adamantane-methyl acetate (10.8 g, 51.0 bandol) in THF (200 ml) was stirred at 0 ° C. The powder was added in several portions and stirred with Ot for 90 minutes. Purification by ethyl acetate extraction gave a white powder of adamantane-ethanol (9, 21 g, 51.0 mmol, quant.). Ή-NR (300MHz, CDC "): 51.10 (1H, br), 1.38 (2H, t, J = 7.5 Hz), 1.52 (6H, d, J = 2.4 Hz), 1.63 (3H, brd, J = 13.5) Hz), 1.70 (3H, brd, J = 12.3 Hz), 1.94 (3H, brs), 3.71 (2H, t, J = 7.5 Hz).
元素分析値 C12H200として計算値: C, 79.94; H, 11.18 (%)、 実測値: C, 80.00; H, 11.35 (%) . Elemental analysis C 12 H 20 0 Calculated: C, 79.94; H, 11.18 (%), Found: C, 80.00; H, 11.35 (%).
ァダマンタン- 1-エタノール(3.33 g, 18.5 mmol)およびメタンスルホニルクロ リド (2.24 g, 19.4 mmol)の THF (70 ml)溶液に 0 で攪拌しながらトリェチル ァミン(3.07 ml, 22.2 mmol)を 20分間かけて滴下した。 反応液を 0°Cで 2時間攪 拌した。 反応液を酢酸ェチルージィソプロピルエーテル(1: 1)抽出により精製し、 メタンスルホン酸ァダマンタン-卜ェチル (5.30 g, 18.5 mmol, quant.)を淡黄色 油状物として得た。 Triethylamine (3.07 ml, 22.2 mmol) was added to a solution of adamantane-1-ethanol (3.33 g, 18.5 mmol) and methanesulfonyl chloride (2.24 g, 19.4 mmol) in THF (70 ml) for 20 minutes while stirring at 0. And dropped. The reaction solution was stirred at 0 ° C for 2 hours. The reaction solution was purified by extraction with ethyl ethyl diisopropyl acetate (1: 1) to give adamantan-toethyl methanesulfonate (5.30 g, 18.5 mmol, quant.) As a pale yellow oil.
'H-NMR (200MHz, CDC13): (51.54 (6H, d, J= 2.6 Hz), 1.56 (2H, t, J= 7.4 Hz), 1.67 (6H, m), 1.97 (3H, m), 3.00 (3H, s), 4.30 (2H, t, J= 7.5 Hz). 'H-NMR (200MHz, CDC1 3): (51.54 (6H, d, J = 2.6 Hz), 1.56 (2H, t, J = 7.4 Hz), 1.67 (6H, m), 1.97 (3H, m), 3.00 (3H, s), 4.30 (2H, t, J = 7.5 Hz).
メタンスルホン酸ァダマンタン-卜ェチル (5.30g, 18.5 mmol)およびヨウ化ナ トリウム(5.55 g, 37.0 mmol) の アセトン (100 ml)懸濁液を 3時間加熱還流し た。反応液を酢酸ェチル—へキサン(2: 3)により抽出して粗製の卜 (2-ョードエチ ル)ァダマンタン (純度 86%, 4.91 g, 14.6 mmol, 79.0 )を黄色固形物として得 た。 A suspension of adamantane-methaneethyl methanesulfonate (5.30 g, 18.5 mmol) and sodium iodide (5.55 g, 37.0 mmol) in acetone (100 ml) was heated under reflux for 3 hours. The reaction mixture was extracted with ethyl acetate-hexane (2: 3) to give crude 2- (ododoethyl) adamantane (purity 86%, 4.91 g, 14.6 mmol, 79.0) as a yellow solid.
Ή-腿 (200MHz, CDC13): δ 1.50 (6Η, d, J= 2.6 Hz), 1.65 (6H, m), 1.79 (2H, m), 1.96 (3H, m), 3.17 (2H, 1) . Ή- thigh (200MHz, CDC1 3): δ 1.50 (6Η, d, J = 2.6 Hz), 1.65 (6H, m), 1.79 (2H, m), 1.96 (3H, m), 3.17 (2H, 1) .
参考例 32 Reference Example 32
メタンスルホン酸ァダマンタン- 1-メチル Adamantane methanesulfonate-1-methyl
ァダマンタン-卜メタノール(2.72 g, 15.8 顏 ol)およびメタンスルホニルクロ リド (1.92 g, 16.6 mmol)の THF (50 ml)溶液に (TCで攪拌しながらトリェチル ァミン(2.63 ml, 19.0讓 ol) の THF (5 ml)溶液を 20分間かけて滴下した。 反応 液を 0°Cで 40分間攪拌した。 反応液を酢酸ェチルーへキサン(1:1)抽出により精 製し、 メタンスルホン酸ァダマンタン- 1-メチル (純度 90¾, 4.27 g, 15.8 mmol, quant. )を淡黄色油状物として得た。 To a solution of adamantane methanol (2.72 g, 15.8 mmol) and methanesulfonyl chloride (1.92 g, 16.6 mmol) in THF (50 ml) (while stirring with TC, was added THF of triethylamine (2.63 ml, 19.0 mmol) while stirring with TC. (5 ml) solution was added dropwise over 20 minutes The reaction solution was stirred for 40 minutes at 0 ° C. The reaction solution was purified by extraction with ethyl acetate-hexane (1: 1), and adamantan methanesulfonate 1- Methyl (purity 90¾, 4.27 g, 15.8 mmol, quant.) was obtained as a pale yellow oil.
'H-NMR (200MHz, CDC13): <51.57 (6H, d, J- 2.6 Hz), 1.70 (6H, brd, J= 8.4 Hz), 2.02 (3H, m), 2.99 (3H, s), 3.78 (2H, s). 'H-NMR (200MHz, CDC1 3): <51.57 (6H, d, J- 2.6 Hz), 1.70 (6H, brd, J = 8.4 Hz), 2.02 (3H, m), 2.99 (3H, s), 3.78 (2H, s).
メタンスルホン酸ァダマンタン-卜メチル (純度 90%, 4.27 g, 15.8 mmol)およ びヨウ化ナトリウム(7.10 g, 47.4 mmol) の 2-ブタノン (85 ml)懸濁液を 3.5 日間加熱還流した。反応液を酢酸ェチルーへキサン (2:3)により抽出後、シリカゲ ル (40 g)のクロマトグラフィー (へキサン) で精製し、 1-ョードメチルァダマン タン (2.20 g, 7.96匪 ol, 50.4%)を白色固形物として得た。 A suspension of adamantane trimethyl methanesulfonate (purity 90%, 4.27 g, 15.8 mmol) and sodium iodide (7.10 g, 47.4 mmol) in 2-butanone (85 ml) was heated under reflux for 3.5 days. The reaction mixture was extracted with ethyl acetate-hexane (2: 3), purified by silica gel (40 g) chromatography (hexane), and purified with 1-odomethyladamantane (2.20 g, 7.96 bandol, 50.4%) as a white solid.
^- R (200MHz, CDC13): 51.52 (6H, d, J= 3.0 Hz), 1.62 (6H, brd, J= 2.2 Hz), 1.96 (3H, brs), 3.02 (2H, s). ^ - R (200MHz, CDC1 3 ): 51.52 (6H, d, J = 3.0 Hz), 1.62 (6H, brd, J = 2.2 Hz), 1.96 (3H, brs), 3.02 (2H, s).
参考例 33 - 1 Reference Example 33-1
(2S,4R)- N-Boc- 2 -ヒドロキシメチル- 4- (2-テトラヒドロピラエルォキシ)ピロリ ジン (2S, 4R) -N-Boc-2-hydroxymethyl-4- (2-tetrahydropyraeroxy) pyrrolidine
(2S,4R)- N- Boc- 4 -ヒドロキシ- L-プロリン (5.03 g, 21.7 mmol)'と 濃塩酸 (0.90 ml, 10.9 讓 ol) のメタノール (100 ml) 溶液を室温で 15時間攪拌した。 反応液に炭酸水素ナトリゥム(0.91 g, 10.9 mmol)を加えて中和後、 酢酸ェチル抽 出により精製し、 (2S, 4R) -N-Boc-4-ヒドロキシ プロリンメチルエステル(3.45 g, 14.0腿 ol, 64.8¾) の無色油状物を得た。 A solution of (2S, 4R) -N-Boc-4-hydroxy-L-proline (5.03 g, 21.7 mmol) 'and concentrated hydrochloric acid (0.90 ml, 10.9 benzene) in methanol (100 ml) was stirred at room temperature for 15 hours. . The reaction mixture was neutralized with sodium hydrogen carbonate (0.91 g, 10.9 mmol), purified by extraction with ethyl acetate, and purified with (2S, 4R) -N-Boc-4-hydroxyproline methyl ester (3.45 g, 14.0 g). ol, 64.8%).
— NMR (300MHz, CDC13): 51.42 (6H, s), 1.46 (3H, s), 1.91 (1H, dd, J= 15.6, 3.3 Hz), 2.09 (1H, m), 2.31 (1H, brdd, J= 13.5, 8.1 Hz), 3.57 (1H, brd, J= - NMR (300MHz, CDC1 3) : 51.42 (6H, s), 1.46 (3H, s), 1.91 (1H, dd, J = 15.6, 3.3 Hz), 2.09 (1H, m), 2.31 (1H, brdd, J = 13.5, 8.1 Hz), 3.57 (1H, brd, J =
12.0 Hz), 3.64 (1H, dd, J= 11.4, 4.2 Hz), 3.74 (3H, s), 4.41 (1H, dt, J= 16.2,12.0 Hz), 3.64 (1H, dd, J = 11.4, 4.2 Hz), 3.74 (3H, s), 4.41 (1H, dt, J = 16.2,
8.4 Hz), 4.50 (1H, brs). 8.4 Hz), 4.50 (1H, brs).
(2S,4R)-N- Boc- 4-ヒドロキシ- L-プロリンメチルエステル (3.15 g, 12.8 mmol) と DHP (1.80 ml, 19.3 腿 ol) のジクロロメタン (60 ml) 溶液を CSA (298 mg, 1.28 mmol)と室温で 18 時間攪拌した。.酢酸ェチル抽出により精製し、 A solution of (2S, 4R) -N-Boc-4-hydroxy-L-proline methyl ester (3.15 g, 12.8 mmol) and DHP (1.80 ml, 19.3 t) in dichloromethane (60 ml) was treated with CSA (298 mg, 1.28 mmol) at room temperature for 18 hours. .Purified by ethyl acetate extraction
(2S,4R)- N- Boc-4 -テトラヒドロビラニルォキシ -L-プロリンメチルエステル(2S, 4R) -N-Boc-4-tetrahydrobilanyloxy-L-proline methyl ester
(5.00 g)の淡褐色粗油状物をァノマー混合物として得た。 (5.00 g) of a light brown crude oil was obtained as an anomeric mixture.
Ή-NMR (200MHz, CDC13): δΙΛΙ ( , s), 1.46 (3H, s), 1.5-1.6 (4H, m), 1.75 (2H, m), 2.08 (1H, m), 2.37 (1H, m), 3.45-3.60 (3H, m), 3.73 (3H, s), 3.84 (1H, m), 4.40 (2H, m), 4.65/4.96 (1H, m). Ή-NMR (200MHz, CDC1 3 ): δΙΛΙ (, s), 1.46 (3H, s), 1.5-1.6 (4H, m), 1.75 (2H, m), 2.08 (1H, m), 2.37 (1H, m), 3.45-3.60 (3H, m), 3.73 (3H, s), 3.84 (1H, m), 4.40 (2H, m), 4.65 / 4.96 (1H, m).
上記で得られた(2S, 4R) - N-Boc - 4 -テトラヒドロピラニルォキシ- L-プロリンメ チルエステルの粗油状物(5.00 g) の THF (80 ml)溶液に室温で攪拌しながら水素 化ホウ素ナトリウム (純度 90%, 538 mg, 12.8匪01) の粉末を加え、 ついで 60。C に加温し、 メタノール(2ml)の THF(8ml)溶液を 70分間かけて滴下した。 酢酸ェ チル抽出により精製し、 黄色油状物 (4.5 g)を得た。 これをシリカゲル (35 g)クロ マトグラフィ一(酢酸ェチル—へキサン、 2:8-4:6)で精製し、 (2S,4R)_N- Boc - 2 - ヒドロキシメチル -4-(2-テトラヒドロピラニルォキシ)ピロリジン (3.58g, 11.9 mmol, 92.8%)の無色油状物を得た。 A solution of the crude oil (5.00 g) of (2S, 4R) -N-Boc-4-tetrahydropyranyloxy-L-proline methyl ester obtained above in THF (80 ml) was stirred at room temperature at room temperature with borohydride. Add sodium (purity 90%, 538 mg, 12.8 bandages 01) powder and then 60. C., and methanol (2 ml) in THF (8 ml) was added dropwise over 70 minutes. Purification by ethyl acetate extraction gave a yellow oil (4.5 g). This was purified by silica gel (35 g) chromatography (ethyl acetate-hexane, 2: 8-4: 6) to give (2S, 4R) _N-Boc-2-hydroxymethyl-4- (2-tetrahydropyrane A colorless oil of (Roxy) pyrrolidine (3.58 g, 11.9 mmol, 92.8%) was obtained.
—NMR (200MHz, CDC13): δΐ.47 (9Η, s), 1.54 (4H, m), 1.6-1.8 (3H, m), 2.13 (1H, m), 3.40-3.60 (3H, m), 3.69 (1H, m) , 3.84 (1H, m), 4.10 (1H, m), 4.28 (1H, m), 4.66 (1H, i), .91 (1H, brdd, J= 16.6, 8.1 Hz). -NMR (200MHz, CDC1 3): δΐ.47 (9Η, s), 1.54 (4H, m), 1.6-1.8 (3H, m), 2.13 (1H, m), 3.40-3.60 (3H, m), 3.69 (1H, m), 3.84 (1H, m), 4.10 (1H, m), 4.28 (1H, m), 4.66 (1H, i), .91 (1H, brdd, J = 16.6, 8.1 Hz).
参考例 33 - 2 Reference Example 33-2
ヨウ化 (2S, 4R)-N-Boc_4-ヒドロキシピロリジン- 2-メチル (2S, 4R) -N-Boc_4-hydroxypyrrolidine-2-methyl iodide
(2S,4R)- N-Boc_2-ヒドロキシメチル- 4- (2-テトラヒドロピラニルォキシ)ピロ リジン(1.11 g, 15.4mmol)およびメタンスルホニルクロリド(0.473 g, 4.04 mmol) の THF (15 ml)溶液に 0°Cで攪拌しながらトリェチルァミン(0.663 ml, 4.78 mmol) の THF (5 ml)溶液を 30分間かけて滴下した。 反応液を 0°Cで 20分間攪拌した。 酢酸ェチル抽出により得た粗物質をシリカゲル(20 g)のクロマトグラフィー(酢 酸ェチルーへキサン、 2:8-3:7) により精製し、 (2S,4R)- N-Boc-4-(2-テトラヒド ロビラニルォキシ)ピロリジン- 2-メチルメシレート(1.35 g, 3.55 mmol, 96.9%) の無色油状物を得た。 (2S, 4R) -N-Boc_2-hydroxymethyl-4- (2-tetrahydropyranyloxy) pyrrolidine (1.11 g, 15.4 mmol) and methanesulfonyl chloride (0.473 g, 4.04 mmol) in THF (15 ml) While stirring at 0 ° C, a solution of triethylamine (0.663 ml, 4.78 mmol) in THF (5 ml) was added dropwise over 30 minutes. The reaction was stirred at 0 ° C for 20 minutes. The crude substance obtained by ethyl acetate extraction was purified by silica gel (20 g) chromatography (ethyl acetate-hexane, 2: 8-3: 7), and (2S, 4R) -N-Boc-4- (2 A colorless oil of -tetrahydrobilanyloxy) pyrrolidine-2-methyl mesylate (1.35 g, 3.55 mmol, 96.9%) was obtained.
Ή-NMR (200MHz, CDC13): δ 1.46 (9Η, s), 1.54 (4H, i), 1.69 (1H, m), 1.77 (1H, m), 2.11 (1H, m), 2.17 (1H, ni), 3.00 (3H, s), 3.51 (2H, m), 3.85 (1H, m), 4.16 (1H, m), 4.20-4.45 (3H, m), 4.55 (1H, m), 4.61 (1H, m). Ή-NMR (200MHz, CDC1 3 ): δ 1.46 (9Η, s), 1.54 (4H, i), 1.69 (1H, m), 1.77 (1H, m), 2.11 (1H, m), 2.17 (1H, ni), 3.00 (3H, s), 3.51 (2H, m), 3.85 (1H, m), 4.16 (1H, m), 4.20-4.45 (3H, m), 4.55 (1H, m), 4.61 (1H , m).
(2S,4R)-N-Boc- 4-ヒドロキシ- L-プロリンメチルエステル (1.78 g, 7.26腿〇1) の THF (80 ml)溶液に室温で攪拌しながら水素化ホウ素ナトリウム (純度 90%, 305 mg, 7.26 匪 ol) の粉末を加え、 ついで 60°Cに加温し、 メタノール(2 ml)の THF(5 ml)溶液を 90 分間かけて滴下した。 酢酸ェチル抽出により精製し、 (2S,4R)- N_Boc- 4-ヒドロキシ- 2-ヒドロキシメチルピロリジン (1.61 g, quant.) の無色油状物を得た。 A solution of (2S, 4R) -N-Boc-4-hydroxy-L-proline methyl ester (1.78 g, 7.26261) in THF (80 ml) was stirred at room temperature while stirring with sodium borohydride (purity 90%, 305 mg, 7.26 bandol) were added, and the mixture was heated to 60 ° C., and a solution of methanol (2 ml) in THF (5 ml) was added dropwise over 90 minutes. Purified by ethyl acetate extraction, A colorless oil of (2S, 4R) -N_Boc-4-hydroxy-2-hydroxymethylpyrrolidine (1.61 g, quant.) Was obtained.
'H-NMR (200MHz, CDC13): 51.48 (9H, s), 1.86 (1H, brs), 2.05 (1H, in), 3.44 (1H, dd, J= 12.0, 4.0 Hz), 3.52 (1H, m), 3.59 (1H, dd, J= 7.0, 2.8 Hz), 3.72 (1H, brt, J= 8.4 Hz), 4.14 (1H, m), 4.38 (1H, brs), 5.04 (1H, m). 'H-NMR (200MHz, CDC1 3): 51.48 (9H, s), 1.86 (1H, brs), 2.05 (1H, in), 3.44 (1H, dd, J = 12.0, 4.0 Hz), 3.52 (1H, m), 3.59 (1H, dd, J = 7.0, 2.8 Hz), 3.72 (1H, brt, J = 8.4 Hz), 4.14 (1H, m), 4.38 (1H, brs), 5.04 (1H, m).
(2S,4R)- N_Boc- 4 -ヒドロキシ -2-ヒドロキシメチルピロリジン (1.61 g, 7.26 腿 ol)および p -トルエンスルホニルクロリド(1.50 g, 7.62腿 ol)の ピリジン (16 ml)溶液を室温で 9時間攪拌した。酢酸ェチル抽出により得た粗物質をシリカゲル (25 g)のクロマトグラフィー(酢酸ェチルーへキサン、 2:8-4:6)により精製し、 (2S,4R)- N_Boc- 4 -ヒドロキシピロリジン- 2-メチルトシレート(1.10 g, 2.96 mmol, ' 40.8%)の無色油状物を得た。 A solution of (2S, 4R) -N_Boc-4-hydroxy-2-hydroxymethylpyrrolidine (1.61 g, 7.26 t) and p-toluenesulfonyl chloride (1.50 g, 7.62 t) in pyridine (16 ml) was added at room temperature to 9 ml. Stirred for hours. The crude material obtained by ethyl acetate extraction was purified by silica gel (25 g) chromatography (ethyl acetate-hexane, 2: 8-4: 6) to give (2S, 4R) -N_Boc-4- 4-hydroxypyrrolidine-2- A colorless oil of methyl tosylate (1.10 g, 2.96 mmol, '40 .8%) was obtained.
JH-N R (200MHz, CDC13): (51.38 (9H, s), 2.06 (2H, m), 2.44 (3H, s), 3.40 (1H, m), 3.44 (1H, m), 4.06 (1H, m), 4.11 (2H, brs), 4.45 (1H, ) , 7.34 (2H, d, J= 7.8 Hz), 7.76 (2H, d, J= 8.4 Hz). JH-N R (200MHz, CDC1 3): (51.38 (9H, s), 2.06 (2H, m), 2.44 (3H, s), 3.40 (1H, m), 3.44 (1H, m), 4.06 (1H , m), 4.11 (2H, brs), 4.45 (1H,), 7.34 (2H, d, J = 7.8 Hz), 7.76 (2H, d, J = 8.4 Hz).
(2S,4R)- N_Boc-4-ヒドロキシピロリジン- 2-メチルトシレート(1.10 g, 2.96 mmol)およびヨウ化ナトリウム(1.33 g, 8.88 mmol) の アセトン (25 ml)懸濁液 を 9時間加熱還流した。 反応液を酢酸ェチル—へキサン、 1:1)により抽出して精 製し、 ヨウ化 (2S,4R)- N- Boc- 4 -ヒドロキシピロリジン- 2-メチル (406 mg, 1.24 mmol, 41.9%)の無色油状物を得た。 A suspension of (2S, 4R) -N_Boc-4-hydroxypyrrolidine-2-methyltosylate (1.10 g, 2.96 mmol) and sodium iodide (1.33 g, 8.88 mmol) in acetone (25 ml) was heated at reflux for 9 hours. did. The reaction solution was purified by extraction with ethyl acetate-hexane (1: 1) and purified by (2S, 4R) -N-Boc-4-hydroxypyrrolidine-2-methyl iodide (406 mg, 1.24 mmol, 41.9% ) Was obtained as a colorless oil.
参考例 34 Reference example 34
trans- 2-ョ—ドメチルシク口へキサン- 1-メチルァセテ一ト trans-2-odemethylcyclohexane-1-methylacetate
trans-シクロへキサン- 1,2-ジカルボン酸 (10.1 , 55.9 腿 ol)と 濃塩酸 (7.00 ml, 83.9 腿01) のメタノール (150 ml) 溶液を室温で 1.5日間攪拌した。 反応液に炭酸水素ナトリウムを加えて中和後、 酢酸ェチル抽出により精製し、 trans-シクロへキサン- 1, 2-ジカルポン酸ジメチル(11.0 g, 54.8 腿 ol, 98.2%) の無色油状物を得た。 A solution of trans-cyclohexane-1,2-dicarboxylic acid (10.1, 55.9 t) and concentrated hydrochloric acid (7.00 ml, 83.9 t) in methanol (150 ml) was stirred at room temperature for 1.5 days. The reaction mixture was neutralized with sodium hydrogen carbonate, and purified by extraction with ethyl acetate to obtain a colorless oily substance of dimethyl trans-cyclohexane-1,2-dicarboxylate (11.0 g, 54.8 mol, 98.2%). Was.
'H-NMR (200MHz, CDC13): 51.32 (4H, m), 1.78 (2H, m), 2.06 (2H, brd, J= 10 Hz), 2.62 (2H, m), 3.67 (6H, s). ' trans-シクロへキサン- 1, 2-ジカルポン酸ジメチル(11.0 g, 54.8 mmol) の THF (200 ml)溶液に 0でで攪拌しながら水素化リチウムアルミニウム (純度 80%, 2.86 g, 60.4腿01) の粉末を数回に分けて加え、 0°Cで 100分間攪拌した。 酢酸ェチル により抽出、 濃縮後、 残渣にジイソプロピルエーテルおよびへキサンを加えた。 析出物を濾取し、 へキサンで洗浄後、 乾燥して trans-シクロへキサン- 1,2-ジメ タノール(5.77 g, 40.0 mmol, 71.6»の無色油状物を得た。 'H-NMR (200MHz, CDC1 3): 51.32 (4H, m), 1.78 (2H, m), 2.06 (2H, brd, J = 10 Hz), 2.62 (2H, m), 3.67 (6H, s) 'Dimethyl trans-cyclohexane-1,2-dicarbonate (11.0 g, 54.8 mmol) in THF (200 ml) A solution of lithium aluminum hydride (purity 80%, 2.86 g, 60.4 thigh 01) was added in several portions to the solution while stirring at 0, and the mixture was stirred at 0 ° C for 100 minutes. After extraction with ethyl acetate and concentration, diisopropyl ether and hexane were added to the residue. The precipitate was collected by filtration, washed with hexane, and dried to obtain a colorless oil of trans-cyclohexane-1,2-dimethanol (5.77 g, 40.0 mmol, 71.6 »).
Ή-NMR (300MHz, CDC13): 51.07 (2H, brt, J= 10.8 Hz), 1.25 (2H, m), 1.33 (2H, m), 1.63 (2H, brd, J= 13.2 Hz), 1.74 (2H, m), 3.56 (2H, dd, J= 11.0, 5.8 Hz), 3.63 (2H, dd, J= 10.8, 3.0 Hz). Ή-NMR (300MHz, CDC1 3 ): 51.07 (2H, brt, J = 10.8 Hz), 1.25 (2H, m), 1.33 (2H, m), 1.63 (2H, brd, J = 13.2 Hz), 1.74 ( 2H, m), 3.56 (2H, dd, J = 11.0, 5.8 Hz), 3.63 (2H, dd, J = 10.8, 3.0 Hz).
trans-シクロへキサン- 1,2 -ジメタノール(2.00 g, 13.9 mmol) の THF (65 ml) 溶液に水素化ナトリウム (純度 60%油性, 0.556 g, 13.9腿 ol) を室温で加え、 60分間攪拌した。 得られた懸濁液を p -トルエンスルホニルクロリド (2.73 g, 13.9讓 ol)の THF (20 ml)溶液に室温で 30分間かけて滴下し、 ついで反応液を 1 時間攪拌した。反応液から酢酸ェチルにより抽出で得た粗油状物をシリカゲル (40 g)のクロマトグラフィ一 (酢酸ェチルーへキサン、 2:8-4:6) で精製し、 trans - 2 - ヒドロキシメチルシクロへキサン-卜メチルトシレート(2.21 g, 7.41 mmol, 53.3¾)を無色油状物として得た。 To a solution of trans-cyclohexane-1,2-dimethanol (2.00 g, 13.9 mmol) in THF (65 ml) was added sodium hydride (purity 60% oily, 0.556 g, 13.9 tmol) at room temperature, and the mixture was added for 60 minutes. Stirred. The resulting suspension was added dropwise to a solution of p-toluenesulfonyl chloride (2.73 g, 13.9 carbonyl) in THF (20 ml) over 30 minutes at room temperature, and the reaction solution was stirred for 1 hour. The crude oil obtained by extraction from the reaction mixture with ethyl acetate was purified by chromatography on silica gel (40 g) (ethyl acetate-hexane, 2: 8-4: 6) to give trans-2-hydroxymethylcyclohexane- Trimethyl tosylate (2.21 g, 7.41 mmol, 53.3%) was obtained as a colorless oil.
-NMR (300MHz, CDC13): 61.18 (4H, m), 1.28 (2H, m), 1.59 (1H, m), 1.71 (4H, m), 1.74 (2H, m), 2.45 (3H, s), 3.51 (1H, dd, J= 11.1, 4.2 Hz), 3.54 (1H, dd, J= 10.8, 4.5 Hz), 4.05 (2H, d, J= 5.1 Hz), 7.34 (2H, d, J= 8.1 Hz), 7.78 (2H, d, J= 8.1 Hz). -NMR (300MHz, CDC1 3): 61.18 (4H, m), 1.28 (2H, m), 1.59 (1H, m), 1.71 (4H, m), 1.74 (2H, m), 2.45 (3H, s) , 3.51 (1H, dd, J = 11.1, 4.2 Hz), 3.54 (1H, dd, J = 10.8, 4.5 Hz), 4.05 (2H, d, J = 5.1 Hz), 7.34 (2H, d, J = 8.1 Hz), 7.78 (2H, d, J = 8.1 Hz).
trans-2-ヒドロキシメチルシクロへキサン- 1-メチルトシレ一ト (2.21 g, 7.41 醒 ol) と 無水酢酸(6 ml) のピリジン (12 ml) 溶液を室温で 11時間攪拌した。 反応液を濃縮乾固して、 trans- 2-ァセトキシメチルシクロへキサン-卜メチルトシ レート(2.54 g, 7.4 mmol, quant. )の無色油状物を得た。 A solution of trans-2-hydroxymethylcyclohexane-1-methyltosylate (2.21 g, 7.41 mmol) and acetic anhydride (6 ml) in pyridine (12 ml) was stirred at room temperature for 11 hours. The reaction solution was concentrated to dryness to give trans-2-acetoxymethylcyclohexane-methyltosylate (2.54 g, 7.4 mmol, quant.) As a colorless oil.
Ή-N R (200MHz, CDC13): δΐ.18 (4Η, brd, J= 6.2 Hz), 1.54 (2H, m), 1.70 (4H, m), 2.02 (3H, s), 2.45 (3H, s), 3.91 (2H, d, J- 4 Hz), 4.00 (2H, d, J= 4.2 Hz), 7.35 (2H, d, J= 8.0 Hz), 7.78 (2H, d, J= 8.4 Hz). Ή-NR (200MHz, CDC1 3 ): δΐ.18 (4Η, brd, J = 6.2 Hz), 1.54 (2H, m), 1.70 (4H, m), 2.02 (3H, s), 2.45 (3H, s ), 3.91 (2H, d, J-4 Hz), 4.00 (2H, d, J = 4.2 Hz), 7.35 (2H, d, J = 8.0 Hz), 7.78 (2H, d, J = 8.4 Hz).
trans- 2-ァセトキシメチルシク口へキサン- 1-メチルトシレ一ト(2.54 g, 7.4 腿 ol)およびヨウ化ナトリウム(3.33 g, 22.2腿 ol) の 2 -ブ夕ノン (55 ml)懸濁 液を 2.5時間加熱還流した。反応液を酢酸ェチル—へキサン(1:4)により抽出、濃 縮乾固して trans- 2 -ョードメチルシクロへキサン- 1-メチルアセテート(1.98 g, 6.68 mmol, 90.1 )を無色油状物として得た。 trans-2-Acetoxymethylcyclohexane-1-methyltosylate (2.54 g, 7.4 t ol) and sodium iodide (3.33 g, 22.2 t ol) suspended in 2-butane nonone (55 ml) The liquid was heated at reflux for 2.5 hours. The reaction mixture was extracted with ethyl acetate-hexane (1: 4), concentrated to dryness, and trans-2-iodomethylcyclohexane-1-methyl acetate (1.98 g, 6.68 mmol, 90.1) was obtained as a colorless oil. Obtained as a product.
'H-NMR (200MHz, CDC13): (51.02 (IH, i), 1.27 (4H, brt, J= 7.6 Hz), 1.53 (1H, m), 1.73 (4H, i), 2.07 (3H, s), 2.45 (3H, s), 3.30 (IH, dd, J= 10.2, 5.4 Hz), 3.34 (IH, dd, J= 9.8, 3.0 Hz), 3.99 (IH, dd, J= 11.4, 4.0 Hz), 4.06 (IH, dd, J= 11.2, 5.2 Hz). 'H-NMR (200MHz, CDC1 3): (51.02 (IH, i), 1.27 (4H, brt, J = 7.6 Hz), 1.53 (1H, m), 1.73 (4H, i), 2.07 (3H, s ), 2.45 (3H, s), 3.30 (IH, dd, J = 10.2, 5.4 Hz), 3.34 (IH, dd, J = 9.8, 3.0 Hz), 3.99 (IH, dd, J = 11.4, 4.0 Hz) , 4.06 (IH, dd, J = 11.2, 5.2 Hz).
参考例 35 Reference Example 35
(2S)- N- Boc-4-ョード _L-プロリンメチルエステル (2S)-N-Boc-4-Ed _L-proline methyl ester
(2S,4R)- N-Boc- 4-ヒドロキシ _L -プロリンメチルエステル(3.45 g, 14.0 mmol) およびメタンスルホニルクロリド (1.70 g, 14.7 mmol)の THF ^(?!!! 溶液にひ^ で攪拌しながらトリェチルァミン(2.33 ml, 16.8 画 ol)の THF (10 ml)溶液を 30 分間かけて滴下した。 反応液を (TCで 80分間攙拌した。 酢酸ェチル抽出により得 た油状物をジェチルエーテルから結晶化し、 (2S, 4R) - N-Boc- 4-メシルォキシ- L - プロリンメチルエステル(4.05 g, 12.5 腿 ol, 89.5%)の白色粉末を得た。 (2S, 4R) -N-Boc-4-hydroxy_L-proline methyl ester (3.45 g, 14.0 mmol) and methanesulfonyl chloride (1.70 g, 14.7 mmol) in THF ^ (? !!!!!) A solution of triethylamine (2.33 ml, 16.8 eol) in THF (10 ml) was added dropwise over 30 minutes, and the reaction mixture was stirred for 80 minutes with TC. To give a white powder of (2S, 4R) -N-Boc-4-mesyloxy-L-proline methyl ester (4.05 g, 12.5 t, 89.5%).
Ή-NMR (200MHz, CDC13): 51.43 (6H, s), 1.47 (3H, s), 2.26 (IH, ddd, J= 14.4, 8.2, 4.8 Hz), 2.62 (IH, m), 3.06 (3H, s), 3.75 (3H, s), 3.79 (2H, i), 4.43 (m, dt, J= 12.8, 8.1 Hz), 5.26 (IH, m) . Ή-NMR (200MHz, CDC1 3 ): 51.43 (6H, s), 1.47 (3H, s), 2.26 (IH, ddd, J = 14.4, 8.2, 4.8 Hz), 2.62 (IH, m), 3.06 (3H , s), 3.75 (3H, s), 3.79 (2H, i), 4.43 (m, dt, J = 12.8, 8.1 Hz), 5.26 (IH, m).
(2S,4R)- N- BQC- 4_メシルォキシ - L-プロリンメチルエステル(純度 80%, 1.45 g, 3.77匪 ol)およびヨウ化ナトリウム(1.70 g, 11.3 mmol) の 2-ブタノン (30 ml) 懸濁液を 38 時間加熱還流した。 反応液を酢酸ェチルにより抽出して精製し、 (2S)- N-Boc- 4-ョード - L -プロリンメチルエステル (純度 80%, 1.29 g, 2.90 mmol, 11%)の淡褐色油状物を得た。 2-butanone (30 ml) of (2S, 4R) -N-BQC-4_mesyloxy-L-proline methyl ester (purity 80%, 1.45 g, 3.77 marl) and sodium iodide (1.70 g, 11.3 mmol) The suspension was heated at reflux for 38 hours. The reaction solution was purified by extraction with ethyl acetate, and a light brown oily substance of (2S) -N-Boc-4-ode-L-proline methyl ester (purity 80%, 1.29 g, 2.90 mmol, 11%) was obtained. Was.
Ή -蘭 R (200MHz, CDC13): 51.43 (6H, s), 1.47 (3H, s), 2.40 (1H, m), 2.57/2.85 (IH, m), 3.74/3.76 (3H, s), 3.75 (IH, m), 3.98/4.08 (IH, m), 4.40 (2H, brquint, J= 5.6 Hz). E - Ran R (200MHz, CDC1 3): 51.43 (6H, s), 1.47 (3H, s), 2.40 (1H, m), 2.57 / 2.85 (IH, m), 3.74 / 3.76 (3H, s), 3.75 (IH, m), 3.98 / 4.08 (IH, m), 4.40 (2H, brquint, J = 5.6 Hz).
参考例 36 Reference Example 36
4 -ョードメチルシクロへキサン - 1-メタノール 4-Eodomethylcyclohexane-1-methanol
cis-シクロへキサン- 1,4-ジカルポン酸 (純度 90%, 9.80 g, 51.2 腿 ol、 8% trans -異性体との混合物)と 濃塩酸 (6.77 ml, 81.3丽 ol) のメタノール (150 ml) 溶液を室温で 1.5日間攪拌した。 反応液に炭酸水素ナトリウムを加えて中和 後、 酢酸ェチル—へキサン(1:1)抽出により精製し、 cis-シクロへキサン- 1,4 -ジ 力ルポン酸ジメチル (純度 91%, 11.2 g, quant.、 8¾ trans-異性体との混合物) の無色油状物を得た。 cis-cyclohexane-1,4-dicarbonic acid (purity 90%, 9.80 g, 51.2 t ol, 8% A mixture of trans-isomer) and concentrated hydrochloric acid (6.77 ml, 81.3 mol) in methanol (150 ml) was stirred at room temperature for 1.5 days. The reaction mixture was neutralized with sodium bicarbonate, and purified by extraction with ethyl acetate-hexane (1: 1) .Dimethyl cis-cyclohexane-1,4-dichloromethane (purity 91%, 11.2 g) , quant., a mixture with 8¾ trans-isomer).
'Η— NMR (200MHz, CDC13): δΐ.67 (4Η, m), 1.89 (4H, m), 2.47 (2H, i), 3.68 (6H, s). . 'Η- NMR (200MHz, CDC1 3 ): δΐ.67 (4Η, m), 1.89 (4H, m), 2.47 (2H, i), 3.68 (6H, s)..
上記で得られた cis -シクロへキサン- 1, 4-ジカルポン酸ジメチル(8.19 g, 37.2 mmol) の THF (160 ml)溶液に 0°Cで攪拌しながら水素化リチウムアルミニウム (純度 80¾, 2.12 g, 44.7 mmol) の粉末を数回に分けて加え、 0°Cで 80分間攪拌 した。 酢酸ェチル -THF (4:1)により抽出し、 乾燥、 濃縮乾固して cis-シクロへキ サン- 1,4-ジメタノール (純度 87%, 6.18 g, quant., 8% trans-異性体との混合物) の無色油状物を得た。 Lithium aluminum hydride (purity 80¾, 2.12 g) was added to a solution of dimethyl cis-cyclohexane-1,4-dicarbonate (8.19 g, 37.2 mmol) obtained above in THF (160 ml) while stirring at 0 ° C. , 44.7 mmol) was added in several portions, and the mixture was stirred at 0 ° C for 80 minutes. Extract with ethyl acetate-THF (4: 1), dry and concentrate to dryness to obtain cis-cyclohexane-1,4-dimethanol (purity 87%, 6.18 g, quant., 8% trans-isomer A colorless oil was obtained.
Ή-NMR (200MHz, CDC13): δ 1.35-1.60 (10Η, m), 1.70 (2H, m), 1.96 (2H, m), 3.55 (4H, d, J= 6.6 Hz). Ή-NMR (200MHz, CDC1 3 ): δ 1.35-1.60 (10Η, m), 1.70 (2H, m), 1.96 (2H, m), 3.55 (4H, d, J = 6.6 Hz).
上記で得られた cis-シクロへキサン- 1,4-ジメタノール (純度 87%, 2.1 g, 12.9 mmol, 8% trans-異性体との混合物)および p-トルエンスルホニルクロリド(2.79 g, 14.2匪 ol)のピリジン (30 ml) 溶液を室温で 15時間攪拌した。 反応液から酢酸 ェチルにより抽出で得た粗油状物をシリカゲル(30 g)のクロマトグラフィー (酢 酸ェチルーへキサン、 2:8- 4:6)で精製し、 4-ヒドロキシメチルシクロへキサン- 1 - メチルトシレート(2.40 g, 8.04歷 ol, 57.4¾, cis/ trans: 92:8混合物)を無色 油状物として得た。 The cis-cyclohexane-1,4-dimethanol obtained above (purity 87%, 2.1 g, 12.9 mmol, mixture with 8% trans-isomer) and p-toluenesulfonyl chloride (2.79 g, 14.2 ol) in pyridine (30 ml) was stirred at room temperature for 15 hours. The crude oil obtained by extraction from the reaction mixture with ethyl acetate was purified by chromatography on silica gel (30 g) (ethyl acetate-hexane, 2: 8-4: 6) to give 4-hydroxymethylcyclohexane-1. -Methyl tosylate (2.40 g, 8.04 olol, 57.4 ¾, cis / trans: 92: 8 mixture) was obtained as a colorless oil.
Ή-NM (300MHz, CDC13): 51.20 (1H, brt, J= 5 Hz), 1.23-1.43 (5H, m), 1.50 (3H, m), 1.66 (1H, m), 1.89 (1H, m), 2.45 (3H, s), 3.49 (2H, dd, J= 6.3, 5.1 Hz), 3.92 (2H, d, J= 6.9 Hz), 7.34 (2H, d,- J= 7.8 Hz), 7.78 (2H, d, J= 8.4 Hz). Ή-NM (300MHz, CDC1 3 ): 51.20 (1H, brt, J = 5 Hz), 1.23-1.43 (5H, m), 1.50 (3H, m), 1.66 (1H, m), 1.89 (1H, m ), 2.45 (3H, s), 3.49 (2H, dd, J = 6.3, 5.1 Hz), 3.92 (2H, d, J = 6.9 Hz), 7.34 (2H, d, -J = 7.8 Hz), 7.78 ( 2H, d, J = 8.4 Hz).
上記で得られた 4 -ヒドロキシメチルシクロへキサン- 1-メチルトシレー卜 (2.40 g, 8.04 mmol, cis/ trans= 92:8混合物)およびヨウ化ナトリウム(3.62 g, 24.1 mmol)のアセトン(50 ml)懸濁液を室温で 15時間攪拌した後、 3時間加熱還 流した。 反応液を酢酸ェチルにより抽出、 濃縮乾固して 4-ョ一ドメチルシクロへ キサン - 1 -メタノール(1.83 g, 7.20 mmol, 89.6¾, cis/ trans= 92:8混合物)を 淡褐色油状物として得た。 4-Hydroxymethylcyclohexane-1-methyltosylate (2.40 g, 8.04 mmol, cis / trans = 92: 8 mixture) obtained above and sodium iodide (3.62 g, 24.1 mmol) in acetone (50 ml) Stir the suspension at room temperature for 15 hours, then heat for 3 hours Shed. The reaction mixture was extracted with ethyl acetate and concentrated to dryness to give 4-hydroxymethylcyclohexane-1-methanol (1.83 g, 7.20 mmol, 89.6¾, cis / trans = 92: 8 mixture) as a pale brown oil. Was.
参考例 37 Reference Example 37
4-フルォロ- 4 -メチルペンチル メシレ一ト 4-Fluoro-4-methylpentyl mesylate
3_メチル - 1,3_ブタンジオール (20.66 g, 純度 90%, 0.179 mol)およびメタン スルホニルクロリド (14.7 ml, 0.187 mol)の THF (300 ml)溶液に 0°Cで攪拌し ながらトリェチルァミン(27.2 ml, 0.196 mol)を 80分間かけて滴下し、 反応液を 0°Cで 1.5時間攪拌した。反応液をジェチルェ一テル-へキサン(1: 1)で抽出し、水 洗、乾燥、濃縮乾固して、 3-メチル -3 -ヒドロキシプチル メシレート (純度 80.5%, 39.3 g, 0.174 mol)を黄色油状物として得た。 Triethylamine (27.2) was added to a solution of 3_methyl-1,3_butanediol (20.66 g, purity 90%, 0.179 mol) and methanesulfonyl chloride (14.7 ml, 0.187 mol) in THF (300 ml) with stirring at 0 ° C. ml, 0.196 mol) was added dropwise over 80 minutes, and the reaction solution was stirred at 0 ° C for 1.5 hours. The reaction mixture was extracted with getyl ether-hexane (1: 1), washed with water, dried and concentrated to dryness to give 3-methyl-3-hydroxybutyl mesylate (purity 80.5%, 39.3 g, 0.174 mol). Obtained as a yellow oil.
'H-NMR (300MHz, CDC13): 51.30 (6H, s), 1.43 (1H, brs), 1.96 (2H, t, J= 6.9 Hz), 3.03 (3H, s), 4.43 (2H, t, J= 6.9 Hz). 'H-NMR (300MHz, CDC1 3): 51.30 (6H, s), 1.43 (1H, brs), 1.96 (2H, t, J = 6.9 Hz), 3.03 (3H, s), 4.43 (2H, t, J = 6.9 Hz).
3 -メチル- 3-ヒドロキシブチル メシレート (純度 80.5%, 39.3 g, 0.174 mol) および シアン化ナトリウム (13.6 g, 0.269 mol) の N, N-ジメチルスルホキシド N, N-dimethylsulfoxide of 3-methyl-3-hydroxybutyl mesylate (80.5% purity, 39.3 g, 0.174 mol) and sodium cyanide (13.6 g, 0.269 mol)
(100 ml)懸濁液を 50 で 27時間,攪拌した。 酢酸ェチルで抽出して精製し、 4 - メチル -4-ヒドロキシパレロニトリル(0.174 mol、 酢酸ェチル含有、 44 g)を黄色 油状物として得た。 ' (100 ml) The suspension was stirred at 50 for 27 hours. Extraction and purification with ethyl acetate gave 4-methyl-4-hydroxypareronitrile (0.174 mol, containing ethyl acetate, 44 g) as a yellow oil. '
'H-NMR (200MHz, CDC13): 51.28 (6H, s), 1.85 (2H, t, J= 7.9 Hz), 2.48 (2H, t, J= 6.9 Hz). 'H-NMR (200MHz, CDC1 3): 51.28 (6H, s), 1.85 (2H, t, J = 7.9 Hz), 2.48 (2H, t, J = 6.9 Hz).
4-メチル -4-ヒドロキシバレロ二トリル (0.174 mol、 酢酸ェチル含有、 44 g) の 酢酸 (200 ml)および濃塩酸 (100 ml)溶液を 21 時間加熱還流した。 反応液を酢酸 ェチルで抽出して精製し、 4, 4-ジメチルブチロラクトン(純度 72 , 23 g, 0.146 mol)を無色油状物として得た。 - Ή-NMR (200MHz, CDC13): (51.44 (6H, s), 2.06 (2H, t, J- 8.3 Hz), 2.63 (2H, t, J= 8.1 Hz). A solution of 4-methyl-4-hydroxyvaleronitrile (0.174 mol, containing ethyl acetate, 44 g) in acetic acid (200 ml) and concentrated hydrochloric acid (100 ml) was heated under reflux for 21 hours. The reaction solution was extracted and purified with ethyl acetate to obtain 4,4-dimethylbutyrolactone (purity: 72, 23 g, 0.146 mol) as a colorless oil. - Ή-NMR (200MHz, CDC1 3): (51.44 (6H, s), 2.06 (2H, t, J- 8.3 Hz), 2.63 (2H, t, J = 8.1 Hz).
4, 4-ジメチルブチロラクトン (トルエン含有, 9.0 g, 52匪 )を THF (120 ml) に溶解し、 0°Cで攪拌しながら水素化リチウムアルミニウム (純度 80 , 3.04 g, 64 膽 ol) を数回に分けて加え、 0°Cで 2 時間攪拌した。 反応液を酢酸ェチル -THF'で 抽出して精製し、 粗製の 4-メチル -1,4-ペン夕ンジオール (純度 80%, 5.00 g, 34 腿 ol)を黄色油状物として得た。 4,4-Dimethylbutyrolactone (containing toluene, 9.0 g, 52 bandages) was dissolved in THF (120 ml), and lithium aluminum hydride (purity: 80, 3.04 g, 64 tonol) was added while stirring at 0 ° C. The mixture was added in portions and stirred at 0 ° C for 2 hours. Ethyl acetate -THF ' Extraction and purification yielded crude 4-methyl-1,4-pentanol (purity 80%, 5.00 g, 34 t ol) as a yellow oil.
4-メチル -1, 4-ペン夕ンジオール (純度 80%, 5.00 g, 34 mmol)およびメタンス ルホニルクロリ ド (3.31 il, 42.3匪 ol)の THF (100 ml)溶液に 0°Cで攪拌しな がらトリェチルァミン(6.57 il, 47.4ΜΙΟ1)の THF (10 ml)溶液を 20分間かけて 滴下し、 反応液を で 1時間攪拌した。 反応液を酢酸ェチルで抽出し、 水洗、 乾燥、濃縮乾固して、粗製の 4-メチル -4-ヒドロキシペンチル メシレート (8.2g, 約 40 mmol)を褐色油状物として得た。 While stirring at 0 ° C, a solution of 4-methyl-1,4-pentanol (purity 80%, 5.00 g, 34 mmol) and methanesulfonyl chloride (3.31 il, 42.3 bandol) in THF (100 ml) was stirred. A solution of triethylamine (6.57 il, 47.4ΜΙΟ1) in THF (10 ml) was added dropwise over 20 minutes, and the reaction solution was stirred with for 1 hour. The reaction solution was extracted with ethyl acetate, washed with water, dried and concentrated to dryness to obtain crude 4-methyl-4-hydroxypentyl mesylate (8.2 g, about 40 mmol) as a brown oil.
4-メチル -4 -ヒドロキシペンチル メシレート (8.2 g, 約 40 mmol)のジクロ口 メタン(5 ml)の溶液に _78でで攪拌しながら DAST (7.16 g, 40匪 ol)のジクロ口 メタン (4 ml)溶液を 50分間かけて滴下し、 0°Cまで昇温後、 反応液を室温で 10 分間攪拌した。 反応液を酢酸ェチルで抽出し、 水洗、 乾燥、 濃縮乾固して、 粗油 状物を得た。 これをシリカゲル(50 g)のクロマトグラフィー (酢酸ェチル一へキ サン、 1:4-1:3) により精製し、 4 -フルォロ- 4-メチルペンチル メシレート (3.97 g, 20.0腿 ol)を淡黄色油状物として得た。 A solution of 4-methyl-4-hydroxypentyl mesylate (8.2 g, about 40 mmol) in dichloromethane (5 ml) was stirred with _78 at _78 while DAST (7.16 g, 40 marl) in dichloromethane (4 ml). ) The solution was added dropwise over 50 minutes, the temperature was raised to 0 ° C, and the reaction solution was stirred at room temperature for 10 minutes. The reaction solution was extracted with ethyl acetate, washed with water, dried and concentrated to dryness to obtain a crude oil. This was purified by silica gel (50 g) chromatography (ethyl acetate 1-hexane, 1: 4-1: 3), and 4-fluoro-4-methylpentyl mesylate (3.97 g, 20.0 t) was pale yellow. Obtained as an oil.
Ή-NMR (200MHz, CDC13): 01.34 (3H, s), 1.41 (3H, s), 1.73 (2H, m), 1.89 (2H, m), 3.02 (3H, s), 4.27 (2H, t, J= 6.3 Hz). Ή-NMR (200MHz, CDC1 3 ): 01.34 (3H, s), 1.41 (3H, s), 1.73 (2H, m), 1.89 (2H, m), 3.02 (3H, s), 4.27 (2H, t , J = 6.3 Hz).
参考例 38 . Reference example 38.
trans-4-ョ一ドメチルシクロへキサン-卜メタノール trans-4-methoxymethylcyclohexane methanol
trans-シクロへキサン- 1,4-ジカルポン酸 (純度 97%, 9.62g, 54.2廳01)と 濃 塩酸 (6.77 ml, 81.3腿01) のメタノール (150 ml) 溶液を室温で 1.5日間攪拌 した。 反応液に炭酸水素ナトリウムを加えて中和後、 酢酸ェチル抽出により精製 し、 trans-シクロへキサン- 1,4-ジカルポン酸ジメチル(純度 99%, 10.9 g, quant.)の白色固形物を得た。 A solution of trans-cyclohexane-1,4-dicarponic acid (purity 97%, 9.62 g, 54.2 Cafe 01) and concentrated hydrochloric acid (6.77 ml, 81.3 thigh 01) in methanol (150 ml) was stirred at room temperature for 1.5 days. The reaction solution was neutralized by adding sodium hydrogen carbonate, and purified by extraction with ethyl acetate to obtain a white solid of dimethyl trans-cyclohexane-1,4-dicarbonate (purity 99%, 10.9 g, quant.). Was.
Ή-NMR (200MHz, CDC13): (51.45 (4H, dd, J= 11.4, 9.2 Hz), 2.05 (4H, brd, J= 7.0 Hz), 2.29 (2H, brt, J= 7 Hz), 3.67 (6H, s). Ή-NMR (200MHz, CDC1 3 ): (51.45 (4H, dd, J = 11.4, 9.2 Hz), 2.05 (4H, brd, J = 7.0 Hz), 2.29 (2H, brt, J = 7 Hz), 3.67 (6H, s).
trans-シクロへキサン- 1,4-ジカルボン酸ジメチル(7.76 g, 38.3腿 ol) の THF (150 ml)溶液に 0 で攪拌しながら水素化リチウムアルミニウム (純度 80%, 2.00 g, 42.2 mmol) の粉末を数回に分けて加え、 0°Cで 80分間攪拌した。 酢酸ェチル - THF (5:1)により抽出し、 乾燥、 濃縮乾固して trans-シクロへキサン- 1, 4-ジメ 夕ノール(5.48 g, 38.0匪 ol, 99.2%)の白色固形物を得た。 A solution of lithium aluminum hydride (purity 80%, 2.00 g, 42.2 mmol) was added to a solution of trans-cyclohexane-dimethyl 1,4-dicarboxylate (7.76 g, 38.3 mol) in THF (150 ml) while stirring at 0. The powder was added in several portions and stirred at 0 ° C for 80 minutes. Ethyl acetate -Extracted with THF (5: 1), dried and concentrated to dryness to obtain a white solid substance of trans-cyclohexane-1,4-dimethylethanol (5.48 g, 38.0 marl, 99.2%).
Ή— NMR (300MHz, CDC13): δ 0.98 (4Η, dd, J= 11.4, 9.2 Hz), 1.33 (2H, brs), 1.47 (2H, m), 1.85 (4H, brd, J= 6.8 Hz), 3.47 (4H, d, J= 6.2 Hz). Ή- NMR (300MHz, CDC1 3) : δ 0.98 (4Η, dd, J = 11.4, 9.2 Hz), 1.33 (2H, brs), 1.47 (2H, m), 1.85 (4H, brd, J = 6.8 Hz) , 3.47 (4H, d, J = 6.2 Hz).
trans-シクロへキサン- 1,4-ジメタノール(2.47 g, 17.1 mmol)および p-トルェ ンスルホニルクロリド(3.70 g, 18.8匪 ol)のピリジン (30ml) 溶液を室温で 7.5 時間攪拌した。反応液から酢酸ェチルにより抽出で得た粗油状物をシリカゲル (20 g)のクロマトグラフィ一 (酢酸ェチルーへキサン、 2:8-4:6) で精製し、 trans - 4- ヒドロキシメチルシクロへキサン- 1-メチルトシレー ト (3.00 g, 10.0 腿 ol, 58.5¾)を無色油状物として得た。 A solution of trans-cyclohexane-1,4-dimethanol (2.47 g, 17.1 mmol) and p-toluenesulfonyl chloride (3.70 g, 18.8 bandol) in pyridine (30 ml) was stirred at room temperature for 7.5 hours. The crude oil obtained by extraction with ethyl acetate from the reaction solution was purified by chromatography on silica gel (20 g) (ethyl acetate-hexane, 2: 8-4: 6) to give trans-4-hydroxymethylcyclohexane- 1-Methyl tosylate (3.00 g, 10.0 t, 58.5 5) was obtained as a colorless oil.
Ή-NMR (200MHz, CDC13): 50.94 (4H, dd, J= 11.2, 8.8 Hz), 1.42 (1H, m), 1.60 (1H, m), 1.79 (4H, m), 2.46 (3H, s), 3.44 (2H, brt, J= 5.7 Hz), 3.83 (2H, d, J= 6.2 Hz), 7.33 (2H, d, J- 8.0 Hz), 7.79 (2H, d, J= 8.4 Hz), Ή-NMR (200MHz, CDC1 3 ): 50.94 (4H, dd, J = 11.2, 8.8 Hz), 1.42 (1H, m), 1.60 (1H, m), 1.79 (4H, m), 2.46 (3H, s ), 3.44 (2H, brt, J = 5.7 Hz), 3.83 (2H, d, J = 6.2 Hz), 7.33 (2H, d, J- 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz),
trans- 4-ヒドロキシメチルシクロへキサン- 1-メチルトシレート (1.21 g, 4.05 丽 ol)およびヨウ化ナトリウム(1.82 g, 12.2匪 ol) の アセトン (25 ml)懸濁液 を 5時間加熱還流した。 反応液を酢酸ェチルにより抽出、 濃縮乾固して trans - 4- ョ一ドメチルシクロへキサン- 1-メタノール(0.92 g, 3.62 mmol, 89.4%)を淡黄色 油状物として得た。 A suspension of trans-4-hydroxymethylcyclohexane-1-methyltosylate (1.21 g, 4.05 olol) and sodium iodide (1.82 g, 12.2 ol) in acetone (25 ml) was heated to reflux for 5 hours. . The reaction solution was extracted with ethyl acetate and concentrated to dryness to obtain trans-4-hydromethylcyclohexane-1-methanol (0.92 g, 3.62 mmol, 89.4%) as a pale yellow oil.
'H-NMR (200MHz, CDC13): δ 1.00 (4Η, dd, J= 11.2, 8.8 Hz), 1.28 (1H, br), 1.84 (2H, m), 1.95 (2H, m), 3.11 (2H, d, J= 6.6 Hz), 3.47 (2H, m). 'H-NMR (200MHz, CDC1 3): δ 1.00 (4Η, dd, J = 11.2, 8.8 Hz), 1.28 (1H, br), 1.84 (2H, m), 1.95 (2H, m), 3.11 (2H , d, J = 6.6 Hz), 3.47 (2H, m).
参考例 39 Reference example 39
p-トルイル酸 5-ヒドロキシ- 4, 4-ジメチルぺンチル 5-hydroxy-4,4-dimethylpentyl p-toluate
2, 2-ジメチルダルタル酸 (98¾, 10.2 g, 62.3匪 ol)と濃塩酸 (7.79 ml, 93.5 mmol) のメタノール (150 ml) 溶液を室温で 1.5日間攪拌した。 反応液に炭酸水 素ナトリウムを加えて中和後、 酢酸ェチルーへキサン(2:1)抽出により精製し、 2, 2 -ジメチルダルタル酸ジメチル(9.00 g, 47.8匪 ol, 76.7%)の無色油状物を得 た。 A solution of 2,2-dimethyldartaric acid (98¾, 10.2 g, 62.3 bandol) and concentrated hydrochloric acid (7.79 ml, 93.5 mmol) in methanol (150 ml) was stirred at room temperature for 1.5 days. The reaction solution was neutralized with sodium hydrogen carbonate, and purified by extraction with ethyl acetate-hexane (2: 1). The colorless solution of dimethyl 2,2-dimethyldaltarate (9.00 g, 47.8 marl ol, 76.7%) was obtained. An oil was obtained.
Ή-NMR (300MHz, CDC13): 51.19 (6H, s), 1.88 (2H, m), 2.28 (2H, m), 3.66 (6H, s). 2, 2-ジメチルダルタル酸ジメチル(9.00 g, 47.8111101)の11^ (180 ml)溶液に 0°C で攪拌しながら水素化リチウムアルミニウム (純度 80 , 2.49 g, 52.6匪01) の 粉末を数回に分けて加え、 0°Cで 2時間攪拌した。酢酸ェチル - THF (3:1)により抽 出、脱水乾燥、濃縮乾固して 2, 2-ジメチルペンタン- 1, 5-ジオール (純度 90%, 6.99 g, quant.)の無色油状物を得た。 Ή-NMR (300MHz, CDC1 3 ): 51.19 (6H, s), 1.88 (2H, m), 2.28 (2H, m), 3.66 (6H, s). A powder of lithium aluminum hydride (purity 80, 2.49 g, 52.6 bandages 01) was added to a 11 ^ (180 ml) solution of dimethyl 2,2-dimethyldaltarate (9.00 g, 47.8111101) while stirring at 0 ° C. The mixture was added in portions and stirred at 0 ° C for 2 hours. Extracted with ethyl acetate-THF (3: 1), dehydrated, dried and concentrated to dryness to give a colorless oil of 2,2-dimethylpentane-1,5-diol (purity 90%, 6.99 g, quant.). Was.
¾-NMR (200MHz, CDC13): δθ.89 (6Η, s), 1.33 (2H, m), 1.42 (2H, m), 3.34 (2H, s), 3.65 (2H, t, J= 6.4 Hz). ¾-NMR (200MHz, CDC1 3 ): δθ.89 (6Η, s), 1.33 (2H, m), 1.42 (2H, m), 3.34 (2H, s), 3.65 (2H, t, J = 6.4 Hz ).
2,2-ジメチルペンタン- 1, 5-ジオール(純度 90%, 625 g, 4.25 龍 ol)および塩 化 P-トルオイル (724 mg, 4.68讓 ol)のピリジン (7 ml) 溶液を室温で 3時間攪 拌した。 反応液を濃縮乾固後、 酢酸ェチルに溶解し、 水洗、 脱水乾燥、 濃縮して 粗油状物を得た。 これをシリカゲル(10 g)のクロマトグラフィー (酢酸ェチルー へキサン、 1:9-2:8) で精製し、 p-トルィル酸 5-ヒドロキシ -4, 4-ジメチルペンチ ル(490 mg, ί.96匪 ol, 46.1 )を無色油状物として得た。 A solution of 2,2-dimethylpentane-1,5-diol (purity 90%, 625 g, 4.25 r. Ol) and chlorinated P-toluoyl (724 mg, 4.68 c. Ol) in pyridine (7 ml) for 3 hours at room temperature Stirred. The reaction solution was concentrated to dryness, dissolved in ethyl acetate, washed with water, dehydrated and dried, and concentrated to obtain a crude oil. This was purified by chromatography on silica gel (10 g) (ethyl acetate-hexane, 1: 9-2: 8) to give 5-hydroxy-4,4-dimethylpentyl p-toluate (490 mg, ί.96 Ol, 46.1) as a colorless oil.
¾— NMR (200MHz, CDC13): 50.91 (6H, s), 1.39 (2H, m), 1.73 (2H, m), 2.41 (3H, s), 3.36 (2H, s), 4.29 (2H, t, J= 6.8 Hz), 7.24 (2H, d, J= 8.0 Hz), 7.93 (2H, d, J= 8.0 Hz). ¾- NMR (200MHz, CDC1 3) : 50.91 (6H, s), 1.39 (2H, m), 1.73 (2H, m), 2.41 (3H, s), 3.36 (2H, s), 4.29 (2H, t , J = 6.8 Hz), 7.24 (2H, d, J = 8.0 Hz), 7.93 (2H, d, J = 8.0 Hz).
参考例 40 Reference example 40
1-メチルシク口へキサン- 1 -メ夕ノール 1-Methylcyclohexan-1-methanol
卜メチルシクロへキサンカルポン酸 (純度 98¾, 3.05 g, 21.2 匪 ol)と濃塩酸 (2.65 ml, 31.9匪 ol) のメタノール (50 ml) 溶液を室温で 2.5日間攪拌した。 反応液に炭酸水素ナトリウムを加えて中和後、 ジイソプロピルエーテル抽出によ り精製し、 1-メチルシクロへキサンカルボン酸メチル (純度 81.4¾, 3.00 g, 15.6 mmol, 73.8¾)の無色油状物を得た。 A methanol (50 ml) solution of trimethylcyclohexanecarponic acid (purity 98¾, 3.05 g, 21.2 bandol) and concentrated hydrochloric acid (2.65 ml, 31.9 bandol) was stirred at room temperature for 2.5 days. The reaction mixture was neutralized with sodium hydrogen carbonate and purified by extraction with diisopropyl ether to obtain a colorless oily substance of methyl 1-methylcyclohexanecarboxylate (purity: 81.4¾, 3.00 g, 15.6 mmol, 73.8¾). Was.
Ή-NMR (200MHz, CDC13): 61.15 (3H, s), 1.20-1.40 (5H, m), 1.54 (3H, m), 2.01 (2H, brdd, J= 8.4, 6.2 Hz), 3.68 (3H, s). Ή-NMR (200MHz, CDC1 3 ): 61.15 (3H, s), 1.20-1.40 (5H, m), 1.54 (3H, m), 2.01 (2H, brdd, J = 8.4, 6.2 Hz), 3.68 (3H , s).
卜メチルシクロへキサンカルポン酸メチル(純度 81. «, 3.00 g, 15.6 mmol) の THF (60 ml)溶液に 0°Cで攪拌しながら水素化リチウムアルミニウム (純度 80 , 503 nig, 10.6画01) の粉末を数回に分けて加え、 0°Cで 2時間攪拌した。 酢酸ェ チルにより抽出、 濃縮乾固して 1 -メチルシクロへキサン- 1-メタノール(純度 91.8¾, 1.95 g, 14.0 mmol, 89.7%)の無色油状物を得た。 Powder of lithium aluminum hydride (purity 80, 503 nig, 10.6 painting 01) in THF (60 ml) solution of methyl trimethylcyclohexanecarbonate (purity 81. «, 3.00 g, 15.6 mmol) at 0 ° C with stirring Was added in several portions, and the mixture was stirred at 0 ° C for 2 hours. Extract with ethyl acetate, concentrate to dryness and dry 1-methylcyclohexane-1-methanol (purity 91.8%, 1.95 g, 14.0 mmol, 89.7%).
'H-NMR (200MHz, CDC13): (50.91 (3H, s), 1.28 (6H, m), 1.37 (1H, br), 1.40-1.55 (4H, m), 3.35 (2H, brs). 'H-NMR (200MHz, CDC1 3): (50.91 (3H, s), 1.28 (6H, m), 1.37 (1H, br), 1.40-1.55 (4H, m), 3.35 (2H, brs).
参考例 41 Reference Example 41
5 -フルォロ- 2, 2-ジメチルペンタノ一ル 5-fluoro-2,2-dimethylpentanoyl
2, 2-ジメチルペンタン- 1, 5-ジオール (純度 90%, 5.82 g, 39.6 mmol)および 3,4-ジヒドロ- 2H-ピラン(4.10 ml, 43.6腿 ol)のジクロロメタン(100 ml) 溶液を CSA (920 mg, 3.96 mmol) 存在下に室温で 3· 5時間攪拌した。 酢酸ェチル -へキサ ン(2:1) で抽出して得た油状物をシリカゲル (90 g) クロマトグラフィー (酢酸 ェチル -へキサン、 15:85および 3: 7) に付し、酢酸ェチル -へキサン(3 :7) の画分 を濃縮乾固して 4, 4 ジメチル -5- (2-テトラヒドロピラニルォキシ)ペンタノ一ル および 2,2 -ジメチル- 5- (2-テトラヒドロピラニルォキシ)ペン夕ノールの 1 : 1 混合物 (3.00 g, 13.9顧 ol) を無色油状物として得た。 A solution of 2,2-dimethylpentane-1,5-diol (purity 90%, 5.82 g, 39.6 mmol) and 3,4-dihydro-2H-pyran (4.10 ml, 43.6 mol) in dichloromethane (100 ml) was treated with CSA. (920 mg, 3.96 mmol) and stirred at room temperature for 3.5 hours. The oil obtained by extraction with ethyl acetate-hexane (2: 1) was chromatographed on silica gel (90 g) (ethyl acetate-hexane, 15:85 and 3: 7) to give ethyl acetate-hexane. The fraction of xanthane (3: 7) is concentrated to dryness to give 4,4 dimethyl-5- (2-tetrahydropyranyloxy) pentanol and 2,2-dimethyl-5- (2-tetrahydropyranyloxy) A 1: 1 mixture of pennitol (3.00 g, 13.9 mmol) was obtained as a colorless oil.
'H-NMR (300MHz, CDC13): δ 0.88/0.89 (3Η, s), 0.91/0.91 (3H, s), 1.30 (2H, m), 1.50-1.62 (7H, m), 1.70 (1H, m), 1.82 (1H, m), 3.01 (1/2H, d, J= 9.0 Hz), 3.33 (2/2H, brd, J= 3.2 Hz) , 3.38 (1/2H, dt, J= 9.6, 6.6 Hz), 3.50 (1H, m), 3.62 (2/2H, brd, J= 6.6 Hz), 3.73 (1/2H, dt, J= 9.6, 6.9 Hz), 3.85 (1H, m), 4.57/4.54 (1H, m). 'H-NMR (300MHz, CDC1 3): δ 0.88 / 0.89 (3Η, s), 0.91 / 0.91 (3H, s), 1.30 (2H, m), 1.50-1.62 (7H, m), 1.70 (1H, m), 1.82 (1H, m), 3.01 (1 / 2H, d, J = 9.0 Hz), 3.33 (2 / 2H, brd, J = 3.2 Hz), 3.38 (1 / 2H, dt, J = 9.6, 6.6 Hz), 3.50 (1H, m), 3.62 (2 / 2H, brd, J = 6.6 Hz), 3.73 (1 / 2H, dt, J = 9.6, 6.9 Hz), 3.85 (1H, m), 4.57 / 4.54 (1H, m).
上記で得た油状物(3.00 g, 13.9 mmol)および p-トルエンスルホニルクロリド (1.18 g, 5.96腿 ol)のピリジン (30 ml) 溶液を室温で 3時間攪拌した。 反応液 から酢酸ェチル -へキサン(1: 1)により抽出して得た粗油状物をシリ力ゲル (40 g) のクロマトグラフィー (酢酸ェチルーへキサン、 10:90-15:85) で精製し、 4,4- ジメチル -5- (2-テトラヒドロビラ二ルォキシ)ぺンチルトシレート(純度 75%, 1.04 g, 2.11 mmol)を無色油状物として得た。 A solution of the above-obtained oil (3.00 g, 13.9 mmol) and p-toluenesulfonyl chloride (1.18 g, 5.96 mmol) in pyridine (30 ml) was stirred at room temperature for 3 hours. The crude oil obtained by extracting the reaction mixture with ethyl acetate-hexane (1: 1) was purified by chromatography on silica gel (40 g) (ethyl acetate-hexane, 10: 90-15: 85). There was obtained 4,4-dimethyl-5- (2-tetrahydrovinyliloxy) pentyl tosylate (purity 75%, 1.04 g, 2.11 mmol) as a colorless oil.
'H-NMR (200MHz, CDC13): (50.84 (3H, s), 0.86 (3H, s), 1.26 (2H, m), 1.45-1.65 (6H, m), 1.80 (2H, m), 2.45 (3H, s), 2.94 (1H, d, J= 9.2 Hz), 3.42 (1H, d, J= 9.6 Hz), 3.51 (1H, m), 3.80 (1H, m), 4.00 (2H, t, J= 6.6 Hz), 4.51 (1H, brs), 7.35 (2H, d, J= 8.0 Hz), 7.79 (2H, d, J= 8.4 Hz). 'H-NMR (200MHz, CDC1 3): (50.84 (3H, s), 0.86 (3H, s), 1.26 (2H, m), 1.45-1.65 (6H, m), 1.80 (2H, m), 2.45 (3H, s), 2.94 (1H, d, J = 9.2 Hz), 3.42 (1H, d, J = 9.6 Hz), 3.51 (1H, m), 3.80 (1H, m), 4.00 (2H, t, J = 6.6 Hz), 4.51 (1H, brs), 7.35 (2H, d, J = 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz).
上記で得た 4,4-ジメチル- 5- (2-テトラヒドロビラニルォキシ)ペンチルトシレ ート(純度 75%, 1.04 g, 2.11 mmol), フッ化テトラプチルアンモニゥム (2.28g, 7.03腿01) および水(0.190 ml, 10.5 mmol)のァセトニトリル(10 ml) 溶液を 80 で 4 時間攪拌した。 反応液を濃縮後、 酢酸ェチル (50 ml)で抽出した。 有機層を 水、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾 固して粗油状物を得た。 これをシリカゲル(10 g)のクロマトグラフィー (酢酸ェ チル一へキサン、 0:10-2:8) に付し、 酢酸ェチルーへキサン(1:19)の画分を濃縮 乾固して 5-フルオロ- 2, 2-ジメチルペンチル 2-テトラヒドロピラニルエーテル (389 mg, 1.78 mmol)を無色油状物として得た。 ' 4,4-Dimethyl-5- (2-tetrahydrobiranyloxy) pentyl tosyl obtained above A solution of acetate (purity 75%, 1.04 g, 2.11 mmol), tetrabutylammonium fluoride (2.28 g, 7.03 01) and water (0.190 ml, 10.5 mmol) in acetonitrile (10 ml) was stirred at 80 for 4 hours at 80 Stirred. After the reaction solution was concentrated, it was extracted with ethyl acetate (50 ml). The organic layer was washed with water, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to dryness to obtain a crude oil. This was subjected to silica gel (10 g) chromatography (ethyl acetate-hexane, 0: 10-2: 8), and the ethyl acetate-hexane (1:19) fraction was concentrated to dryness to give 5- Fluoro-2,2-dimethylpentyl 2-tetrahydropyranyl ether (389 mg, 1.78 mmol) was obtained as a colorless oil. '
]H-NMR (200MHz, CDC13): 60.91 (3Η, s), 0.92 (3H, s), 1.36 (2H, m), 1.50-1.65 (4H, m), 1.60 (1H, m), 1.70 (1H, m), 1.70 - 1.85 (2H, m), 3.00 (1H, d, J= 9.6 Hz), 3.48 (1H, d, J= 9.62 Hz), 3.51 (1H, m), 3.84 (1H, m), 4.42 (2H, dt, J= 47.6, 6.2 Hz), 4.56 (1H, i). ] H-NMR (200MHz, CDC1 3): 60.91 (3Η, s), 0.92 (3H, s), 1.36 (2H, m), 1.50-1.65 (4H, m), 1.60 (1H, m), 1.70 ( 1H, m), 1.70-1.85 (2H, m), 3.00 (1H, d, J = 9.6 Hz), 3.48 (1H, d, J = 9.62 Hz), 3.51 (1H, m), 3.84 (1H, m ), 4.42 (2H, dt, J = 47.6, 6.2 Hz), 4.56 (1H, i).
5-フルォロ- 2,2-ジメチルペンチル 2-テトラヒドロピラニルエーテル(1.10 g, 5.04 mmol)、 および CSA (117 mg, 0.504腿 ol)のメタノール(10 ml) 溶液を室温 で 5時間攪拌した。反応液を濃縮後、酢酸ェチルで抽出、精製し、 5-フルォロ- 2, 2- ジメチルペン夕ノールおよび酢酸ェチルの 1: 1混合物 (1.40 g)を無色油状物とし て得た。 A solution of 5-fluoro-2,2-dimethylpentyl 2-tetrahydropyranyl ether (1.10 g, 5.04 mmol) and CSA (117 mg, 0.504 mol) in methanol (10 ml) was stirred at room temperature for 5 hours. The reaction mixture was concentrated, extracted with ethyl acetate and purified to obtain a 1: 1 mixture (1.40 g) of 5-fluoro-2,2-dimethylpentanol and ethyl acetate as a colorless oil.
Ή-NMR (200MHz, CDC13): 50.90 (6H, s), 1.35 (2H, m), 1.73 (2H, m), 3.35 (2H, brd, J= 4.8 Hz), 4.44 (2H, brdt, J= 47.2, 6.2 Hz). Ή-NMR (200MHz, CDC1 3 ): 50.90 (6H, s), 1.35 (2H, m), 1.73 (2H, m), 3.35 (2H, brd, J = 4.8 Hz), 4.44 (2H, brdt, J = 47.2, 6.2 Hz).
参考例 42 Reference Example 42
2-ブロモ -5, 5-ジメチルシクロへキサン- 1,3-ジオン 2-bromo-5,5-dimethylcyclohexane-1,3-dione
5, 5 -ジメチルシクロへキサン- 1, 3-ジオン (5.00 g, 35.3 mmol) を 47%臭化水 素酸(5.34ml) 及び水 (25.0ml) に溶かし、臭素酸カリウム (2.0 , 12.0 mmol) の温水 (25.0ml) 溶液を加えた。 生成した沈澱物を濾取し、 水で洗浄後、 乾燥し て題記化合物 (8.46 g, ca.100¾) を合成した。 酢酸ェチル -ジェチルェ一テルか ら再結晶して融点 175-177°Cの無色結晶を得た。 5,5-Dimethylcyclohexane-1,3-dione (5.00 g, 35.3 mmol) was dissolved in 47% hydrobromic acid (5.34 ml) and water (25.0 ml), and potassium bromate (2.0, 12.0 mmol) was dissolved. ) In warm water (25.0 ml). The resulting precipitate was collected by filtration, washed with water, and dried to synthesize the title compound (8.46 g, ca. 100¾). Recrystallization from ethyl acetate-ethyl ether gave colorless crystals having a melting point of 175-177 ° C.
IR (KBr) V: 3700-2700, 2959, 2872, 2614, 1719, 1645, 1590 cm"1. IR (KBr) V: 3700-2700, 2959, 2872, 2614, 1719, 1645, 1590 cm " 1 .
Ή -醒 (CDC13: 01.12 (6H, s), 2.48 (4H, s), 6.58 (1H, bs). E - Awakening: (CDC1 3: 01.12 (6H, s), 2.48 (4H, s), 6.58 (1H, bs).
元素分析値: CAABr ^I^Oとして 計算値 (¾) : C, 42.80; H, 5.20. Elemental analysis: CAABr ^ I ^ O Calculated value (¾): C, 42.80; H, 5.20.
実測値 (¾) : C, 42.72; H, 4.84. Found (¾): C, 42.72; H, 4.84.
参考例 43 Reference Example 43
2 -プロモ- 4, 4-ジメチルシク口へキサン- ί, 3 -ジオン 2-Promo-4,4-dimethylcyclohexan-ί, 3-dione
参考例 42と同様の方法により、 4, 4-ジメチルシクロへキサン- 1,3-ジオン(5.00 g, 35.7誦01) から題記化合物 (7.53 g, 96%) を合成した。 酢酸ェチル-ジェチ ルエーテルから再結晶して融点 147- U9°Cの暗赤色結晶を得た。 By the same method as in Reference Example 42, the title compound (7.53 g, 96%) was synthesized from 4,4-dimethylcyclohexane-1,3-dione (5.00 g, 35.7 recited 01). Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 147-U9 ° C.
IR (KBr) V 3700-2200, 2967, 2932, 2872, 1574 cm"1. IR (KBr) V 3700-2200, 2967, 2932, 2872, 1574 cm " 1 .
Ή— NMR (CDC13) : δ 1.18 (6ΗΧ0.62, s), 1.32 (3HX0.38, s), 1.86 (2H, t, J=6.6 Hz), 2.63 (2HX0.38, t, J=6.2 Hz), 2.68 (2HX0.62, t, J=6.2 Hz), 6.42 (1H Ή- NMR (CDC1 3): δ 1.18 (6ΗΧ0.62, s), 1.32 (3HX0.38, s), 1.86 (2H, t, J = 6.6 Hz), 2.63 (2HX0.38, t, J = 6.2 Hz), 2.68 (2HX0.62, t, J = 6.2 Hz), 6.42 (1H
X0.38, bs), 6.53 (1HX0.62, bs). X0.38, bs), 6.53 (1HX0.62, bs).
元素分析値: CsHuO^rとして Elemental analysis: as CsHuO ^ r
計算値 (%) : C, 43.86; H, 5.06. Calculated value (%): C, 43.86; H, 5.06.
実測値 (%) : C, 43.68; H, 5.01. Found (%): C, 43.68; H, 5.01.
参考例 44 Reference example 44
2 -ブロモ -5-メチルシクロへキサン - 1, 3-ジオン 2-bromo-5-methylcyclohexane-1,3-dione
参考例 42と同様の方法により、 5-メチルシクロへキサン- 1,3-ジオン (5.り Og, In the same manner as in Reference Example 42, 5-methylcyclohexane-1,3-dione (5.
38.8匪01) から題記化合物 (7.45 g, 94¾) を合成した。 酢酸ェチル -ジェチルェ 一テルから再結晶して融点 173- 175°Cの暗赤色結晶を得た。 The title compound (7.45 g, 94¾) was synthesized from 38.8 bandits01). Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals with a melting point of 173-175 ° C.
IR (KBr) リ: 3700-2300, 2963, 2936, 2878, 2614, 1636, 1599 cm"1. IR (KBr): 3700-2300, 2963, 2936, 2878, 2614, 1636, 1599 cm " 1 .
Ή-NMR (CDC13) : δ\Λ2 (3Η, d, ゾ =6.0 Hz), 2.16-2.44 (3H, m), 2.58-2.80 (2H, m), 6.55 (1H, bs) . Ή-NMR (CDC1 3): δ \ Λ2 (3Η, d, zo = 6.0 Hz), 2.16-2.44 (3H , m), 2.58-2.80 (2H, m), 6.55 (1H, bs).
元素分析値: C7H902Brとして Elementary analysis: as C 7 H 9 0 2 Br
計算値 (%) : C, 41.00; H, 4.42. Calculated value (%): C, 41.00; H, 4.42.
実測値 (¾) : C, 41.31; H, 4.41. Found (¾): C, 41.31; H, 4.41.
参考例 45 Reference example 45
3-ピリジンスルホニル クロリド 3-pyridinesulfonyl chloride
方法 A Method A
3-ピリジンスルホン酸(l.OOg, 6.09腿01)の^^ジメチルホルムァミド(10.0 ml) の懸濁液に、 塩化チォニル (0.925 ml, 12.2 mmol) を氷冷下ゆっくりと滴下 した。反応混合物を室温で 50分間かき混ぜた後、 80°Cで 1時間かき混ぜた。反応 混合物を酢酸ェチルで希釈し、 飽和炭酸水素ナトリウム水溶液、 水、 飽和食塩水 で洗?争後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に全量 2.00 mlまで啬去し て、 次の反応にそのまま用いた。 ^^ Dimethylformamide (10.0g, l.OOg, 6.09 thigh 01) To this suspension, thionyl chloride (0.925 ml, 12.2 mmol) was slowly added dropwise under ice-cooling. The reaction mixture was stirred at room temperature for 50 minutes and then at 80 ° C for 1 hour. The reaction mixture was diluted with ethyl acetate, washed with a saturated aqueous solution of sodium hydrogencarbonate, water and saturated saline, and then dried over sodium sulfate. The solvent was removed under reduced pressure to a total volume of 2.00 ml, and used as it was in the next reaction.
方法 B Method B
3-ピリジンスルホン酸 (l.OOg, 6.09 mmol) と五塩化りん (2.64g, 12.2腿01) の混合物を 140°Cで 2時間かき混ぜた。 室温まで冷却後、 反応混合物を酢酸ェチ ルで希釈し、 濾過した。 瀘液を濃縮し、 残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物を油 状物として得、 次の反応にそのまま用いた。 A mixture of 3-pyridinesulfonic acid (l.OOg, 6.09 mmol) and phosphorus pentachloride (2.64 g, 12.2 g 01) was stirred at 140 ° C for 2 hours. After cooling to room temperature, the reaction mixture was diluted with ethyl acetate and filtered. The filtrate was concentrated, the residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound as an oil. Used directly in the reaction.
IR (KBr) V: 3652, 3434, 3065, 1742, 1719, 1567, 1468 cm"1. IR (KBr) V: 3652, 3434, 3065, 1742, 1719, 1567, 1468 cm " 1 .
Ή-NMR (CDC13) : 57.62 (1H, ddd, J=8.0, 4.8, 0.8 Hz), 8.34 (1H, ddd, J=8.0, Ή-NMR (CDC1 3): 57.62 (1H, ddd, J = 8.0, 4.8, 0.8 Hz), 8.34 (1H, ddd, J = 8.0,
2.2, 1.4 Hz), 8.98 (1H, dd, J=5.2, 1.4 Hz), 9.27 (1H, d, 1=2.2 Hz). 2.2, 1.4 Hz), 8.98 (1H, dd, J = 5.2, 1.4 Hz), 9.27 (1H, d, 1 = 2.2 Hz).
参考例 46 Reference Example 46
p-トルエンスルフィン酸 p-toluenesulfinic acid
p -トルエンスルフィン酸ナトリウム塩 (5.00 g, 27.2匪01) の温水 (40.0 ml) 溶液に濃塩酸 (6.80 ml) をゆっくりと加えた。 反応混合物を氷冷下で冷却後、 冷 蔵庫で 3.5時間放置した。生成した沈殿を濾取し、氷水で洗浄し題記化合物(3.12 g, 73%) を無色結晶として得た。 Concentrated hydrochloric acid (6.80 ml) was slowly added to a solution of p-toluenesulfinic acid sodium salt (5.00 g, 27.2 band01) in warm water (40.0 ml). After cooling the reaction mixture under ice cooling, it was left in a refrigerator for 3.5 hours. The resulting precipitate was collected by filtration and washed with ice water to give the title compound (3.12 g, 73%) as colorless crystals.
融点 74.0-75.0X 74.0-75.0X
元素分析値: C7H802Sとして Elemental analysis: as C 7 H 8 0 2 S
計算値 (%) : C, 53.82; H, 5.16; S, 20.53. Calculated value (%): C, 53.82; H, 5.16; S, 20.53.
実測値 (¾) : C, 53.64; H, 5.10; S, 20.68. Found (¾): C, 53.64; H, 5.10; S, 20.68.
参考例 47 Reference Example 47
P-トルエンスルフィニル クロリド P-toluenesulfinyl chloride
参考例 46で得られた P-トルエンスルフィン酸 (200 rag, 1.28 mmol) の N,N_ ジメチルホルムアミド (4.00 ml) 溶液に、 塩化チォニル (0.194 ml, 2.56匪〇1) を加えた。 反応混合物を室温で 1時間かき混ぜた後、 反応混合物を酢酸ェチルで 希釈し、水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(181 mg, 92¾) を油状物として得、 次の反応にそのまま用いた。 To a solution of P-toluenesulfinic acid (200 rag, 1.28 mmol) obtained in Reference Example 46 in N, N_dimethylformamide (4.00 ml) was added thionyl chloride (0.194 ml, 2.56 maraudal 1). After stirring the reaction mixture at room temperature for 1 hour, the reaction mixture is stirred with ethyl acetate. Diluted, washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (181 mg, 92¾) as an oil, it was used directly in the next reaction.
参考例 48 Reference Example 48
D - (+)-3-ブロモ-カンファ一- 8-スルホニル クロリド D-(+)-3-bromo-campha-1-sulfonyl chloride
D- (+)- 3-ブロモ-カンファ一- 8-スルホン酸 アンモニゥム (500 mg, 1.48 mmol) の N,N -ジメチルホルムアミド (5.00ml) 溶液に、 塩化チォニル (0.224ml, 2.96 mmol) を加えた。 反応混合物を室温で 1時間かき混ぜた後、 反応混合物を酢酸ェ チルで希釈し、 水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物を油 状物として得、 次の反応にそのまま用いた。 To a solution of D-(+)-3-bromo-1-camphor-8-sulfonic acid ammonium (500 mg, 1.48 mmol) in N, N-dimethylformamide (5.00 ml) was added thionyl chloride (0.224 ml, 2.96 mmol). Was. After stirring 1 hour the reaction mixture at room temperature, the reaction mixture was diluted with acetic acid E chill, washed with water, dried (MgSO 4), to give the title compound as an oil-like material and the solvent was evaporated under reduced pressure, the following Used directly in the reaction.
参考例 49 Reference 49
6, 6, 7, 7, 7 -ペン夕フルォロヘプ夕ンニトリル 6,6,7,7,7-Pen evening Fluorohep evening night nitrile
シアン化ナトリウム (1.59 g, 31.5 mmol) のジメチルスルホキシド (4.00 ml) の懸濁液に、参考例 9で得られたメタンスルホン酸 5, 5, 6, 6, 6-ペン夕フルォ口へ キシルエステル (7.10 g, 26.3 匪 ol) のジメチルスルホキシド (4.00 ml) 溶液 を加えた。反応混合物を室温で 19.5時間かき混ぜた後、水を加えてジェチルェ一 テルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記 化合物を油状物として得、 次の反応にそのまま用いた。 To a suspension of sodium cyanide (1.59 g, 31.5 mmol) in dimethylsulfoxide (4.00 ml) was added the methanesulfonic acid 5,5,6,6,6-pentenefluorohexyl ester obtained in Reference Example 9. (7.10 g, 26.3 bandol) in dimethyl sulfoxide (4.00 ml) was added. After stirring the reaction mixture at room temperature for 19.5 hours, water was added thereto, and the mixture was extracted with getyl ether. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure to give the title compound as an oil, it was used directly in the next reaction.
Ή-NMR (CDC13) : 51.68-1.83 (4H, m), 1.90-2.22 (2H, m), 2.38-2.46 (2H, m) . 参考例 50 Ή-NMR (CDC1 3): . 51.68-1.83 (4H, m), 1.90-2.22 (2H, m), 2.38-2.46 (2H, m) Reference Example 50
6, 6, 7, 7, 7-ペンタフルォロヘプタン酸 6, 6, 7, 7, 7-pentafluoroheptanoic acid
参考例 49で得られた 6, 6, 7, 7, 7-ペン夕フルォロヘプ夕ンニトリルを酢酸(30.0 ml) 及び濃塩酸 (15.0 ml) に溶かし、 120 で 2時間かき混ぜた。 反応混合物を 氷水に加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物を油状物として得、 次の反応にそのまま用いた。 6,6,7,7,7-Pentafluoroheptanil nitrile obtained in Reference Example 49 was dissolved in acetic acid (30.0 ml) and concentrated hydrochloric acid (15.0 ml), and the mixture was stirred at 120 for 2 hours. The reaction mixture was added to ice water and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was evaporated reduced pressure to give the title compound as an oil, was used directly in the next reaction.
IR (KBr) v: 3700-2200, 2955, 1715, 1661, 1603 cm"1. IR (KBr) v: 3700-2200, 2955, 1715, 1661, 1603 cm " 1 .
¾-NMR (CDC13) : (51.54-1.84 (4H, m), 1.92-2.24 (2H, m), 2.42 (2H, t, J=7.0 Hz). ¾-NMR (CDC1 3): (51.54-1.84 (4H, m), 1.92-2.24 (2H, m), 2.42 (2H, t, J = 7.0 Hz).
参考例 51 Reference Example 51
6,6,7, 7, 7-ペンタフルォ口-卜ヘプ夕ノール 参考例 50で得られた 6, 6, 7, 7, 7-ペン夕フルォロヘプタン酸のテトラヒドロフ ラン (80.0 ml) 溶液に、 ォキサリル クロリド (3.80 ml, 39.7 mmol) と N,N -ジ メチルホルムアミド (0.100 ml) を順次加えた。 反応混合物を室温で 3.5時間か き混ぜた後、 溶媒を減圧下に留去した。 水素化ホウ素ナトリウム (2.50 g, 66.1 mmol) のテトラヒドロフラン (40,0 ml) 及び 1, 2-ジメトキシェタン (30.0 ml) 溶液に、 残留物のテトラヒドロフラン (40.0ml) 溶液を氷冷下加えた。 反応混合 物を室温で 23時間かき混ぜた後、 反応混合物を氷水に注ぎ、 1N塩酸を加えて酢 酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して 題記化合物 (3.72 g, 69¾ (メタンスルホン酸 5, 5, 6, 6, 6-ペンタフルォ口へキシ ルエステルから算出)) を油状物として得た。 6,6,7,7,7-Pentafluoro mouth In a solution of 6,6,7,7,7-pentafluorofluorheptanoic acid obtained in Reference Example 50 in tetrahydrofuran (80.0 ml), oxalyl chloride (3.80 ml, 39.7 mmol) and N, N-dimethylformamide ( 0.100 ml). After stirring the reaction mixture at room temperature for 3.5 hours, the solvent was distilled off under reduced pressure. To a solution of sodium borohydride (2.50 g, 66.1 mmol) in tetrahydrofuran (40,0 ml) and 1,2-dimethoxyethane (30.0 ml) was added a solution of the residue in tetrahydrofuran (40.0 ml) under ice cooling. After stirring the reaction mixture at room temperature for 23 hours, the reaction mixture was poured into ice water, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after calculating the carboxy glycol ester and the solvent was distilled off under reduced pressure the title compound (3.72 g, 69¾ (methanesulfonic acid 5, 5, 6, 6, the 6- Pentafuruo port) ) Was obtained as an oil.
IR (KBr) V: 3700-2700, 2949, 2878, 1723 cm"1. IR (KBr) V: 3700-2700, 2949, 2878, 1723 cm " 1 .
Ή-NMR (CDC13) : δ 1.34 - 1.88 (6Η, m), 1.88-2.20 (2H, m), 3.68 (2H, t, J=6.2 Hz). . Ή-NMR (CDC1 3): δ 1.34 - 1.88 (6Η, m), 1.88-2.20 (2H, m), 3.68 (2H, t, J = 6.2 Hz)..
参考例 52 Reference Example 52
メタンスルホン酸 6, 6, 7, 7, 7 -ペン夕フルォ口へプチルエステル Methanesulfonic acid 6,6,7,7,7-Penta fluorester heptyl ester
参考例 51で得られた 6, 6, 7, 7, 7-ペン夕フルォ口-卜ヘプ夕ノール(3.72 g, 18.0 mmol)のテトラヒドロフラン(80.0ml)溶液に、メタンスルホニル クロリド(2.09 ml, 27.1 mmol) とトリエチルァミン (5.03 ml, 36.1匪 ol) を氷冷下順次加えた。 反応混合物を室温で 3.5時間かき混ぜた後、 1 塩酸を加えて酢酸ェチルで抽出し た。 抽出液を水洗、 乾燥'(MgS04) 後、 溶媒を減圧下に留去して題記化合物 (3.80 g, 74%) を油状物として得た。 Methanesulfonyl chloride (2.09 ml, 27.1) was added to a solution of 6,6,7,7,7-pentafluorofuranol (3.72 g, 18.0 mmol) obtained in Reference Example 51 in tetrahydrofuran (80.0 ml). mmol) and triethylamine (5.03 ml, 36.1 bandol) were sequentially added under ice-cooling. After stirring the reaction mixture at room temperature for 3.5 hours, 1 hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried '(MgSO 4), solvent was evaporated under reduced pressure to give the title compound (3.80 g, 74%) as an oil.
IR (KBr) V: 2948, 2878, 1734, 1674, 1356, 1194, 1177, 976, 945 cm"1. Ή-NMR (CDCI3) : 51.35-1.90 (6H, m), 1.90-2.26 (2H, m), 3.02 (3H, s), 4.25 (2H, t, J=6.6 Hz). IR (KBr) V: 2948, 2878, 1734, 1674, 1356, 1194, 1177, 976, 945 cm " 1. .-NMR (CDCI3): 51.35-1.90 (6H, m), 1.90-2.26 (2H, m ), 3.02 (3H, s), 4.25 (2H, t, J = 6.6 Hz).
参考例 53-1 Reference Example 53-1
5, 5, 5 -トリフルォロペンタン二トリル. , 5,5,5-Trifluoropentane nitrile.
シアン化ナトリウム (1.13 g, 18.7龍01) のジメチルスルホキシド (5.70 ml) の懸濁液に、 4-ブロモ -ί, 1, 1-トリフルォロブタン (3.64 g, 18.7 mmol) を加え た。反応混合物を室温で Π時間かき混ぜた後、水を加えてジェチルエーテルで抽 出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物を 油状物として得、 次の反応にそのまま用いた。 To a suspension of sodium cyanide (1.13 g, 18.7 dragon 01) in dimethyl sulfoxide (5.70 ml) was added 4-bromo-ί, 1,1-trifluorobutane (3.64 g, 18.7 mmol). Stir the reaction mixture at room temperature for Π hour, add water and extract with getyl ether. Issued. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure to give the title compound as an oil, it was used directly in the next reaction.
IR (KBr) V: 2944, 2251 cm"1. IR (KBr) V: 2944, 2251 cm " 1 .
]H-NMR (CDC13) : (51.88-2.05 (2H, m), 2.16-2.42 (2H, m), 2.49 (2H, t, J=7.0 Hz). ] H-NMR (CDC1 3) : (51.88-2.05 (2H, m), 2.16-2.42 (2H, m), 2.49 (2H, t, J = 7.0 Hz).
参考例 53-2 Reference Example 53-2
5, 5, 5-トリフルォロペンタン酸 5,5,5-trifluoropentanoic acid
参考例 53-1で得られた 5, 5, 5-トリフルォロペンタン二トリルを酢酸(20.0 ml) 及び濃塩酸 (10.0 ml) に溶かし、 120°Cで 2時間かき混ぜた。 反応混合物を氷水 に加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去して題記化合物を油状物として得、 次の反応にそのまま用いた。 5,5,5-Trifluoropentane nitrile obtained in Reference Example 53-1 was dissolved in acetic acid (20.0 ml) and concentrated hydrochloric acid (10.0 ml), and the mixture was stirred at 120 ° C for 2 hours. The reaction mixture was added to ice water and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure to give the title compound as an oil, it was used directly in the next reaction.
IR (KBr) V: 3600-2300, 1717 cm—1. IR (KBr) V: 3600-2300, 1717 cm— 1 .
Ή-NMR (CDCI3) : (51.82-2.00 (2H, m), 2.06-2.32 (2H, m), 2.49 (2H, t, J=7.2 Hz). Ή-NMR (CDCI3): (51.82-2.00 (2H, m), 2.06-2.32 (2H, m), 2.49 (2H, t, J = 7.2 Hz).
参考例 53-3 Reference Example 53-3
5, 5, 5-トリフルォロ -1_ペン夕ノール 5, 5, 5-Trifluoro-1_ Pen Pennol
参考例 53-2で得られた 5, 5, 5-トリフルォ口へキサン酸のテトラヒドロフラン (40.0 ml) 溶液に、 ォキサリル クロリド (2.93 ml, 25.5 mmol) と ジメチ ルホルムアミド (100 ml) を順次加えた。 反応混合物を室温で 2時間かき混ぜた 後、 溶媒を減圧下に留去した。 水素化ホウ素ナトリウム (1.93g, 51.0 mmol) の テトラヒドロフラン (20.0 ml) 及び 1,2-ジメトキシェタン (15.0 ml) 溶液に、 残留物のテトラヒドロフラン (20.0ml) 溶液を氷冷下加えた。 反応混合物を室温 で 20時間かき混ぜた後、 反応混合物を氷水に注ぎ、 1N塩酸を加えて酢酸ェチル で抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合 物 (680 mg, 26¾ (4-ブロモ -1, 1, 1-トリフルォロブタンから算出,)) を油状物とし て得た。 To a solution of 5,5,5-trifluorohexanoic acid in tetrahydrofuran (40.0 ml) obtained in Reference Example 53-2, oxalyl chloride (2.93 ml, 25.5 mmol) and dimethylformamide (100 ml) were sequentially added. . After stirring the reaction mixture at room temperature for 2 hours, the solvent was distilled off under reduced pressure. To a solution of sodium borohydride (1.93 g, 51.0 mmol) in tetrahydrofuran (20.0 ml) and 1,2-dimethoxyethane (15.0 ml) was added a solution of the residue in tetrahydrofuran (20.0 ml) under ice cooling. After stirring the reaction mixture at room temperature for 20 hours, the reaction mixture was poured into ice water, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (680 mg, 26¾ (4- bromo-1, 1, calculated from the 1-triflate Ruo Rob Tan,)) and Obtained as an oil.
IR (KBr) V: 3700-3100, 2951 cm—1. IR (KBr) V: 3700-3100, 2951 cm— 1 .
Ή-NMR (CDCI3) : (51.56-1.72 (4H, m), 2.00-2.25 (2H, m), 3.69 (2H, t, J=5.8 Hz). 参考例 53 - 4 Ή-NMR (CDCI3): (51.56-1.72 (4H, m), 2.00-2.25 (2H, m), 3.69 (2H, t, J = 5.8 Hz). Reference Example 53-4
メタンスルホン酸 5, 5, 5-トリフルォロペンチルエステル Methanesulfonic acid 5,5,5-trifluoropentyl ester
参考例 53-3 で得られた 5, 5, 5-トリフルォロ- 1-ペン夕ノール (680 mg, 4.88 ranol)のテトラヒドロフラン(15. Oml)溶液に、メタンスルホニル クロリド(0.555 ml, 7.18mmoi) とトリエチルァミン (1.33 ml, 9.57mmol) を氷冷下順次加えた。 反応混合物を室温で 2時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(790 mg, 75¾) を油状物として得た。 To a solution of 5,5,5-trifluoro-1-pentanol (680 mg, 4.88 ranol) obtained in Reference Example 53-3 in tetrahydrofuran (15.Oml) was added methanesulfonyl chloride (0.555 ml, 7.18 mmoi). Triethylamine (1.33 ml, 9.57 mmol) was sequentially added under ice cooling. After stirring the reaction mixture at room temperature for 2 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (790 mg, 75¾) as an oil.
IR (KBr) リ : 2949, 2888, 1360, 1337, 1258, 1175, 1146, 976, 939 cm—1. Ή-NMR (CDC13) : 51.62-1.96 (4H, m), 2.02-2.28 (2H, m), 3.03 (3H, s), 4.26 (2H, t, J=6.2 Hz). IR (KBr) Re:. 2949, 2888, 1360, 1337, 1258, 1175, 1146, 976, 939 cm- 1 Ή-NMR (CDC1 3): 51.62-1.96 (4H, m), 2.02-2.28 (2H, m), 3.03 (3H, s), 4.26 (2H, t, J = 6.2 Hz).
参考例 53-5 Reference Example 53-5
6, 6, 6-トリフルォ口へキサン二トリル 6,6,6-Trifluohexanenitrile
シアン化ナトリウム (950 mg, 18.8腿 ol) のジメチルスルホキシド (6.00 ml) の懸濁液に、 参考例 53- 4で得られたメタンスルホン酸 5, 5, 5 -トリフルォロペン チルエステル (2.76 g, 12.5醒 ol) のジメチルスルホキシド (4.00 ml) 溶液を 加えた。反応混合物を室温で 19.5時間かき混ぜた後、水を加えてジェチルエーテ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物を油状物として得、 次の反応にそのまま用いた。 To a suspension of sodium cyanide (950 mg, 18.8 t ol) in dimethyl sulfoxide (6.00 ml) was added the methanesulfonic acid 5,5,5-trifluoropentyl ester obtained in Reference Example 53-4 (2.76 g, 12.5 mmol). ol) in dimethylsulfoxide (4.00 ml). After stirring the reaction mixture at room temperature for 19.5 hours, water was added and extracted with getyl ether. The extract was washed with water, dried (MgSO 4), to give the title of compound as an oil and the solvent was evaporated under reduced pressure and used directly in the next reaction.
参考例 54 Reference example 54
6, 6, 6-トリフルォ口へキサン酸 6,6,6-Trifluohexanoic acid
参考例 53-5で得られた 6, 6, 6-トリフルォ口へキサン二卜リルを酢酸(8.00 ml) 及び濃塩酸 (16.0 ml) に溶かし、 120°Cで 2時間かき混ぜた。 反応混合物を氷水 に加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去して題記化合物 (1.03g, 49% (メタンスルホン酸 5, 5, 5 -トリフルォロペン チルエステルから算出)) を油状物として得た。 6,6,6-Trifluorohexanitolitol obtained in Reference Example 53-5 was dissolved in acetic acid (8.00 ml) and concentrated hydrochloric acid (16.0 ml), and the mixture was stirred at 120 ° C. for 2 hours. The reaction mixture was added to ice water and extracted with ethyl acetate. As an oil - the extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (calculated from Torifuruoropen Chiruesuteru) 1.03 g, 49% (methanesulfonic acid 5, 5, 5) Was.
IR (KBr) V: 3700-2300, 2949, 2884, 1715, 1661, 1605 cm -1. IR (KBr) V: 3700-2300, 2949, 2884, 1715, 1661, 1605 cm- 1 .
'H-NMR (CDC13) : (51.56-1.80 (4H, m), 2.04-2.22 (2H, m), 2.41 (2H, t, J=7.2 Hz) . 参考例 55 'H-NMR (CDC1 3) : (51.56-1.80 (4H, m), 2.04-2.22 (2H, m), 2.41 (2H, t, J = 7.2 Hz). Reference Example 55
6, 6, 6 -トリフルォ口- 1 -へキサノール 6,6,6-Trifluo-1-hexanol
参考例 54で得られた 6, 6, 6-トリフルォ口へキサン酸 (1.03 g, 6.13 mmol) の テトラヒドロフラン(10. Oml)溶液に、ォキサリル クロリド(0.704ml, 7.35腿01) と N,N-ジメチルホルムアミド (0.100 ml) を順次加えた。 反応混合物を室温で 1 時間かき混ぜた後、 溶媒を減圧下に留去した。 水素化ホウ素ナトリウム (464 mg, 12.3 mmol) のテトラヒドロフラン (5.00 ml) 及び 1, 2-ジメトキシェタン (4.00 ml) 溶液に、 残留物のテトラヒドロフラン (5.00ml) 溶液を氷冷下加えた。 反応 混合物を室温で 18時間かき混ぜた後、 反応混合物を氷水に注ぎ、 1N塩酸を加え て酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去 して題記化合物 (730 mg, 79¾) を油状物として得た。 In a solution of 6,6,6-trifluoromethylhexanoic acid (1.03 g, 6.13 mmol) obtained in Reference Example 54 in tetrahydrofuran (10.Oml), oxalyl chloride (0.704 ml, 7.35 t01) and N, N- Dimethylformamide (0.100 ml) was added sequentially. After stirring the reaction mixture at room temperature for 1 hour, the solvent was distilled off under reduced pressure. To a solution of sodium borohydride (464 mg, 12.3 mmol) in tetrahydrofuran (5.00 ml) and 1,2-dimethoxyethane (4.00 ml) was added a solution of the residue in tetrahydrofuran (5.00 ml) under ice cooling. After stirring the reaction mixture at room temperature for 18 hours, the reaction mixture was poured into ice water, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (730 mg, 79¾) as an oil.
IR (KBr) V: 3700-3050, 2948, 2874 cm"1. IR (KBr) V: 3700-3050, 2948, 2874 cm " 1 .
Ή-NMR (CDC13) : 51.34-1.70 (6H, m), 2.00-2.22 (2H, m), 3.67 (2H, t, J=6.2 Ή-NMR (CDC1 3): 51.34-1.70 (6H, m), 2.00-2.22 (2H, m), 3.67 (2H, t, J = 6.2
Hz). Hz).
参考例 56 Reference Example 56
メタンスルホン酸 6, 6, 6-トリフルォ口へキシルエステル Methanesulfonic acid 6,6,6-trifluorohexyl ester
参考例 55で得られた 6, 6, 6-トリフルォロ- 1 -へキサノール(730 mg, 4.86 mmol) のテトラヒドロフラン(15.0ml)溶液に、メタンスルホニル クロリド(0.376 ml, To a solution of 6,6,6-trifluoro-1-hexanol (730 mg, 4.86 mmol) obtained in Reference Example 55 in tetrahydrofuran (15.0 ml) was added methanesulfonyl chloride (0.376 ml,
4.86 mmol) とトリエチルァミン (1.02 ml, 7.29腿01) を氷冷下順次加えた。 反 応混合物を室温で 2時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(873 mg, 77%) を油状物として得た。 4.86 mmol) and triethylamine (1.02 ml, 7.29 thigh 01) were sequentially added under ice cooling. After stirring the reaction mixture at room temperature for 2 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain the solvent was distilled off under reduced pressure the title compound (873 mg, 77%) as an oil.
IR (KBr) V : 2949, 2878, 1177, 1144, 974, 947 cm一1. IR (KBr) V: 2949, 2878, 1177, 1144, 974, 947 cm- 1 .
Ή-NMR (CDC13) : (51.38-1.88 (6H, m), 2.00-2.24 (2H, m), 3.02 (3H, s), 4.24 (2H, t, J=6.4 Hz). Ή-NMR (CDC1 3): (51.38-1.88 (6H, m), 2.00-2.24 (2H, m), 3.02 (3H, s), 4.24 (2H, t, J = 6.4 Hz).
参考例 57 Reference Example 57
5 -フルォロペンタン二トリル 5-Fluoropentane nitrile
シアン化ナトリウム (2.12 g, 41.9醒01) のジメチルスルホキシド (6.00 ml) の懸濁液に、 4-ブロモ _1_フルォロブタン (4.42 g, 57.9 mmol) のジメチルスル ホキシド(4.00 ml)溶液を加えた。反応混合物を室温で 17.5時間かき混ぜた後、 水を加えてジェチルエーテルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物を油状物として得、 次の反応にそのまま用いた。 参考例 58 To a suspension of sodium cyanide (2.12 g, 41.9) in dimethyl sulfoxide (6.00 ml) was added 4-bromo_1_fluorobutane (4.42 g, 57.9 mmol) in dimethyl sulfoxide. A solution of foxide (4.00 ml) was added. After stirring the reaction mixture at room temperature for 17.5 hours, water was added and extracted with getyl ether. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure to give the title compound as an oil, it was used directly in the next reaction. Reference Example 58
5-フルォロペン夕ナ一ル 5-Fluoropen Pen
参考例 57で得られた 5-フルォロペンタン二トリルのジクロロメタン (200 ml) 溶液に、水素化ジイソブチルアルミニウム 1.5 mol/1 トルエン溶液(37.2 ml, 55.8 mmol) を- 70°Cで加えた。 反応混合物を- 70°Cでアルゴン気流下 2.5時間かき混ぜ た。 飽和塩化アンモニゥム水溶液 (100 ml) を加え、 反応混合物を室温で 1時間 かき混ぜた後、 塩酸を加えてジクロロメタンで抽出した。 抽出液を水洗、 乾燥 To a solution of 5-fluoropentanenitrile obtained in Reference Example 57 in dichloromethane (200 ml), diisobutylaluminum hydride 1.5 mol / 1 toluene solution (37.2 ml, 55.8 mmol) was added at -70 ° C. The reaction mixture was stirred at -70 ° C under an argon stream for 2.5 hours. A saturated aqueous solution of ammonium chloride (100 ml) was added, the reaction mixture was stirred at room temperature for 1 hour, hydrochloric acid was added, and the mixture was extracted with dichloromethane. Wash the extract with water and dry
( gS04) 後、 溶媒を減圧下に留去して題記化合物を油状物として得、 次の反応に そのまま用いた。 (GS0 4) After the solvent was evaporated under reduced pressure to give the title compound as an oil, was used directly in the next reaction.
参考例 59 Reference example 59
5-フルォ口-卜ペン夕ノール 5-Fluo mouth
水素化アルミニウムリチウム(1.59 g, 33.5 mmol)のジェチルエーテル(100 ml) 懸濁液に、 参考例 58 で得られた 5-フルォロペンタナールのジェチルエーテル (50.0 ml) 溶液を氷冷下加え、 反応混合物を室温で 1時間かき混ぜた。過剰の水 素化アルミニウムリチウムを氷冷下少量の水で分解し、 反応混合物を濾過、 ジェ チルェ一テルで洗浄した。 濾液を乾燥 ( gS04) 後、 溶媒を減圧下に留去して題記 化合物 (850 mg, 29% (4-ブロモ -1-フルォロブタンから算出)) を油状物として得 た。 To a suspension of lithium aluminum hydride (1.59 g, 33.5 mmol) in getyl ether (100 ml) was added a solution of 5-fluoropentanal in dimethyl ether (50.0 ml) obtained in Reference Example 58 on ice. The reaction mixture was stirred at room temperature for 1 hour. Excess lithium aluminum hydride was decomposed with a small amount of water under ice-cooling, and the reaction mixture was filtered and washed with ethyl ether. After the filtrate dried (GS0 4), to obtain solvent was distilled off under reduced pressure the title compound (850 mg, 29% (calculated from 4-bromo-1-Furuorobutan)) as an oil.
IR (KBr) V: 3700-3050, 2938, 2867 cm—1. IR (KBr) V: 3700-3050, 2938, 2867 cm— 1 .
'H-NMR (CDC13) : 51.46-1.56 (2H, i), 1.58-1.84 (4H, m), 3.67 (2H, t, J=6.3 Hz), 4.46 (2H, dt, J=47.4, 6.0 Hz). 'H-NMR (CDC1 3) : 51.46-1.56 (2H, i), 1.58-1.84 (4H, m), 3.67 (2H, t, J = 6.3 Hz), 4.46 (2H, dt, J = 47.4, 6.0 Hz).
参考例 60 Reference Example 60
メタンスルホン酸 5_フルォロペンチルエステル Methanesulfonic acid 5_fluoropentyl ester
参考例 59で得られた 5 -フルォロ-卜ペンタノ一ル (400mg, 3.77腿01) のテト ラヒドロフラン (10.0ml) 溶液に、 メタンスルホニル クロリド (0.350 ml, 4.52 腿 ol) とトリエチルァミン (0.788 ml, 5.65 mmol) を氷冷下順次加えた。 反応混 合物を室温で 45分かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出 液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (690 mg, 99¾) を油状物として得た。 To a solution of 5-fluoropentanol (400 mg, 3.77 thigh 01) obtained in Reference Example 59 in tetrahydrofuran (10.0 ml) was added methanesulfonyl chloride (0.350 ml, 4.52 thigh ol) and triethylamine (0.788 ml). , 5.65 mmol) were sequentially added under ice cooling. Reaction mixture After the mixture was stirred at room temperature for 45 minutes, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (690 mg, 99¾) as an oil.
IR ( Br) リ: 2946, 2919, 2874, 1352, 1175, 976, 955, 924 cm-1. IR (Br): 2946, 2919, 2874, 1352, 1175, 976, 955, 924 cm- 1 .
Ή-NMR (CDC ) : δ 1.50-1.62 (3Η, m), 1.66-1.88 (3H, m), 3.02 (3H, s), 4.25 (2H, t, J-6.3 Hz), 4.47 (2H, dt, 1=47.1, 6.0 Hz). Ή-NMR (CDC): δ 1.50-1.62 (3Η, m), 1.66-1.88 (3H, m), 3.02 (3H, s), 4.25 (2H, t, J-6.3 Hz), 4.47 (2H, dt) , 1 = 47.1, 6.0 Hz).
参考例 61 Reference Example 61
1-ブロモ -5-フルォロペン夕ン 1-bromo-5-fluoropen
例 1 Example 1
ジェチルァミノ硫黄トリフルオリド (67.9 g, 0.421 匪 ol) のジクロロメタン (200 ml) 溶液に、 5-ブロモ-卜ペンタノ一ル (67.0 g, 0.401 mmol) のジクロ口 メタン (100 ml) 溶液を- 70°Cでゆっくりと滴下した。 反応混合物を 22時間かけ て 0°Cまで昇温した後、 氷冷下飽和炭酸水素ナトリウムを加えてジクロロメタン で抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去し、 蒸留によ り精製して題記化合物 (34.2 g, 51¾) を油状物として得た。 To a solution of getylaminosulfur trifluoride (67.9 g, 0.421 ol) in dichloromethane (200 ml) was added a solution of 5-bromo-toppentanol (67.0 g, 0.401 mmol) in dichloromethane (100 ml) at -70 ° C. And slowly dripped. After the reaction mixture was heated to 0 ° C. over 22 hours, saturated sodium bicarbonate was added thereto under ice-cooling, and the mixture was extracted with dichloromethane. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure, the title compound was purified Ri by distillation (34.2 g, 51¾) as an oil.
'H-NMR (CDC13) : (51.52-1.64 (2H, m), 1.64-1.84 (2H, m), 1.92 (2H, quint, J=7.2 Hz), 3.43 (2H, t, J=6.9 Hz), 4.46 (2H, dt, J=47.4, 6.0 Hz). 'H-NMR (CDC1 3) : (51.52-1.64 (2H, m), 1.64-1.84 (2H, m), 1.92 (2H, quint, J = 7.2 Hz), 3.43 (2H, t, J = 6.9 Hz ), 4.46 (2H, dt, J = 47.4, 6.0 Hz).
例 2 Example 2
ジェチルァミノ硫黄トリフルオリド (50.0 g, 0.310 mmol) のジクロロメタン (200 ml) 溶液に、 5-ブロモ -卜ペン夕ノール (50.0 g, 0.299 mmol) のジクロ口 メタン (50.0 ml) 溶液を- 7(TCでゆっくりと滴下した。 反応混合物を 16.5時間か けて室温まで昇温した後、 飽和炭酸水素ナトリウムを加えてジクロロメタンで抽 出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去し、 蒸留により精 製して題記化合物 (30.7 g, 61¾) を油状物として得た。 To a solution of getylaminosulfur trifluoride (50.0 g, 0.310 mmol) in dichloromethane (200 ml) was added a solution of 5-bromo-topopenol (50.0 g, 0.299 mmol) in dichloromethane (50.0 ml) at -7 (TC after heating was slowly added dropwise. the reaction mixture to room temperature for 16.5 hours Placing and gave extracted with dichloromethane by the addition of saturated sodium bicarbonate. the extract was washed with water, dried (MgSO 4) after the solvent under reduced pressure The residue was purified by distillation to give the title compound (30.7 g, 61¾) as an oil.
参考例 62 Reference Example 62
へキサン酸 (4-ヒドロキシブチル)アミド Hexanoic acid (4-hydroxybutyl) amide
4 -ヒドロキシブチルァミン(1.00 g, 11.0 mmol)と n-へキサン酸(1.68 ml, 12.1 画 ol) の N,N -ジメチルホルムアミド (20.0 ml) 溶液に、 シァノりん酸ジェチル (1.96 ml, 12.1 mmol) 続いてトリェチルァミン (1.69 ml, 12.1 匪 ol) を加え た。反応混合物を室温で 18時間かき混ぜた後、水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (2.24 g, ca.l00¾) を油状物として得、 次の反応にそのまま用いた。 To a solution of 4-hydroxybutylamine (1.00 g, 11.0 mmol) and n-hexanoic acid (1.68 ml, 12.1 mmol) in N, N-dimethylformamide (20.0 ml) was added getyl cyanophosphate (1.96 ml, 12.1 ml). mmol) and then add triethylamine (1.69 ml, 12.1 ol). Was. After stirring the reaction mixture at room temperature for 18 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure to give the title compound (2.24 g, ca.l00¾) as an oil, it was used directly in the next reaction.
Ή-NMR (CDC13) : 50.896 (3H, t, J=6.6Hz), 1.16-1.44 (6H, m), 1.48-1.76 (4H, m), 2.16 (2H, t, J-7.6 Hz), 3.22-3.38 (2H, m), 3.69 (1H, bt, J=6.0 Hz), 4.08 (2H, t, J=6.2 Hz), 5.62 (1H, bs). Ή-NMR (CDC1 3): 50.896 (3H, t, J = 6.6Hz), 1.16-1.44 (6H, m), 1.48-1.76 (4H, m), 2.16 (2H, t, J-7.6 Hz), 3.22-3.38 (2H, m), 3.69 (1H, bt, J = 6.0 Hz), 4.08 (2H, t, J = 6.2 Hz), 5.62 (1H, bs).
参考例 63 Reference Example 63
メタンスルホン酸 4 -へキサノィルァミノブチル エステル Methanesulfonic acid 4-hexanoylaminobutyl ester
参考例 62で得られたへキサン酸 (4-ヒドロキシブチル)アミド (2.24 g, 11.0 腿 ol)のテトラヒドロフラン(50.0ml)溶液に、メタンスルホニル クロリド(1.02 ml, 13.2腿01) とトリエチルァミン (2.30ml, 16.5讓01) を氷冷下順次加えた。 反応混合物を室温で 2.5時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出し た。抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (2.78 g, 95¾) を油状物として得た。 In a solution of hexanoic acid (4-hydroxybutyl) amide (2.24 g, 11.0 t) obtained in Reference Example 62 in tetrahydrofuran (50.0 ml), methanesulfonyl chloride (1.02 ml, 13.2 t01) and triethylamine ( 2.30ml, 16.5case01) were sequentially added under ice-cooling. After stirring the reaction mixture at room temperature for 2.5 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (2.78 g, 95¾) as an oil.
LH-NMR (CDC13) : (50.897 (3H, t, J=7.5Hz), 1.22-1.40 (6H, m), 1.50-1.74 (4H, m), 2.16 (2H, t, J=7.2 Hz), 3.31 (2H, q, J=6.0 Hz), 2.02 (3H, s), 4.27 (2H, t, J=6.0 Hz), 5.50 (1H, bs). LH-NMR (CDC1 3): (50.897 (3H, t, J = 7.5Hz), 1.22-1.40 (6H, m), 1.50-1.74 (4H, m), 2.16 (2H, t, J = 7.2 Hz) , 3.31 (2H, q, J = 6.0 Hz), 2.02 (3H, s), 4.27 (2H, t, J = 6.0 Hz), 5.50 (1H, bs).
参考例 64 Reference Example 64
へキサン酸 [4- (2-ヒドロキシェチル)フエニル]アミド Hexanoic acid [4- (2-hydroxyethyl) phenyl] amide
4- (2-ヒドロキシェチル)フエニルァミン (500 mg, 3.57腿 ol) と n -へキサン 酸 (0.547 ml, 3.93 mmol) の N, N-ジメチルホルムアミド (10.0 ml) 溶液に、 シ ァノりん酸ジェチル (0.635 ml, 3.93 mmol) 続いてトリェチルァミン (0.548 ml, 3.93讓01) を加えた。 反応混合物を室温で 18時間かき混ぜた後、 水を加えて酢 酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して 題記化合物 (520 mg, 62%) を無色結晶として得、 次の反応にそのまま用いた。 IR (KBr) V: 3700-2600, 2930, 2872, 1653, 1593, 1532 cm"1. To a solution of 4- (2-hydroxyethyl) phenylamine (500 mg, 3.57 liters) and n-hexanoic acid (0.547 ml, 3.93 mmol) in N, N-dimethylformamide (10.0 ml) was added cyanophosphoric acid. Getyl (0.635 ml, 3.93 mmol) was added followed by triethylamine (0.548 ml, 3.93). After stirring the reaction mixture at room temperature for 18 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure to give the title compound (520 mg, 62%) as colorless crystals, it was used directly in the next reaction. IR (KBr) V: 3700-2600, 2930, 2872, 1653, 1593, 1532 cm " 1 .
Ή-NM (CDCI3) : 50.910 (3H, t, J=7.0Hz), 1.24-1.45 (4H, m), 1.64-1.82 (4H, m), 2.35 (2H, t, J=7.6 Hz), 2.84 (2H, t, J=6.6 Hz), 3.84 (2H, t, J=7.0 Hz), 7.12 (1H, bs), 7.19 (2H, d, J=8.4 Hz), 7.46 (2H, d, J=8.0 Hz). 参考例 65 Ή-NM (CDCI3): 50.910 (3H, t, J = 7.0Hz), 1.24-1.45 (4H, m), 1.64-1.82 (4H, m), 2.35 (2H, t, J = 7.6 Hz), 2.84 (2H, t, J = 6.6 Hz), 3.84 (2H, t, J = 7.0 Hz), 7.12 (1H, bs), 7.19 (2H, d, J = 8.4 Hz), 7.46 (2H, d, J = 8.0 Hz). Reference example 65
メタンスルホン酸 2-(4 -へキサノィルァミノフエニル)ェチルエステル Methanesulfonic acid 2- (4-hexanoylaminophenyl) ethyl ester
参考例 64で得られたへキサン酸 [4- (2-ヒドロキシェチル)フエニル]アミド (520 mg, 2.21 靈 ol) のテトラヒドロフラン (10.0 ml) 溶液に、 メタンスルホ ニル クロリド (0.205 ml, 2.65mmol) とトリエチルァミン(0.462 ml, 3.31 mmol) を氷冷下順次加えた。反応混合物を室温で 2時間かき混ぜた後、 1N塩酸を加えて 酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去し て題記化合物 (630 mg, 91¾) を合成した。 酢酸ェチル-ジェチルエーテルから再 結晶して融点 88.0-90. の無色結晶を得た。 Methanesulfonyl chloride (0.205 ml, 2.65 mmol) was added to a solution of hexanoic acid [4- (2-hydroxyethyl) phenyl] amide (520 mg, 2.21 ol) in tetrahydrofuran (10.0 ml) obtained in Reference Example 64. And triethylamine (0.462 ml, 3.31 mmol) were sequentially added under ice cooling. After stirring the reaction mixture at room temperature for 2 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (630 mg, 91¾) was synthesized. Recrystallization from ethyl acetate-getyl ether gave colorless crystals having a melting point of 88.0-90.
IR (KBr) V 3304, 2957, 2932, 2861, 1661, 1601, 1526 cm—1. IR (KBr) V 3304, 2957, 2932, 2861, 1661, 1601, 1526 cm— 1 .
Ή-NMR (CDC13) : 50.912 (3H, t, J=7.0Hz), 1.30-1.42 (4H, m), 1.65-1.82 (2H, m), 2.35 (2H, t, J=7.6 Hz), 2.87 (3H, s), 3.02 (2H, t, J=6.8 Hz), 4.39 (2H, t, J=6.6 Hz), 7.11 (IH, bs), 7.19 (2H, d, J=8.8 Hz), 7.48 (2H, d, J=8.4 Hz). 元素分析値: C15H23N04Sとして Ή-NMR (CDC1 3): 50.912 (3H, t, J = 7.0Hz), 1.30-1.42 (4H, m), 1.65-1.82 (2H, m), 2.35 (2H, t, J = 7.6 Hz), 2.87 (3H, s), 3.02 (2H, t, J = 6.8 Hz), 4.39 (2H, t, J = 6.6 Hz), 7.11 (IH, bs), 7.19 (2H, d, J = 8.8 Hz), 7.48 (2H, d, J = 8.4 Hz) elemental analysis:. as C 15 H 23 N0 4 S
計算値 (¾) : C, 57.48; H, 7.40; N, 4.47; S, 10.23. Calculated value (¾): C, 57.48; H, 7.40; N, 4.47; S, 10.23.
実測値 (¾) : C, 57.21; H, 7.41; N, 4.31; S, 9.85. Found (¾): C, 57.21; H, 7.41; N, 4.31; S, 9.85.
参考例 66 Reference Example 66
4 -へキサノィルァミノ安息香酸 ェチルエステル 、 4-hexanoylamino benzoic acid ethyl ester,
4 -ァミノ安息香酸ェチルエステル(1.00 g, 6.05 mmol)と n -へキサン酸(0.927 ml, 6.66 mmol) の N, N-ジメチルホルムアミド (5.00 ml) 溶液に、 シァノりん酸 ジェチル (1.08 ml, 6.66顧01) 続いてトリェチルァミン (0.928 ml, 6.66 mmol) を加えた。反応混合物を室温で 20時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(2.82 g, ca.100¾) を黄色無定形晶として得た。 To a solution of 4-ethylaminobenzoic acid ethyl ester (1.00 g, 6.05 mmol) and n-hexanoic acid (0.927 ml, 6.66 mmol) in N, N-dimethylformamide (5.00 ml) was added getyl cyanophosphate (1.08 ml, 6.66). 01) Subsequently, triethylamine (0.928 ml, 6.66 mmol) was added. After stirring the reaction mixture at room temperature for 20 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (2.82 g, ca.100¾) as a yellow amorphous crystals.
IR (KBr) ソ : 3326, 2959, 2932, 2863, 1715, 1701, 1597, 1534 cm"1. IR (KBr) Seo: 3326, 2959, 2932, 2863, 1715, 1701, 1597, 1534 cm " 1 .
'H-NMR (CDC") : (50.916 (3H, t, J=6.9 Hz), 1.30-1.48 (4H, m), 1.39 (3H, t, J=7.2 Hz), 1.68-1.82 (2H, m), 2.39 (3H, t, J=7.8Hz), 4.36 (2H, q, J=6.9Hz), 7.31 (1H, bs), 7.61 (2H, d, J=8.7 Hz), 8.01 (2H, d, J=8.7 Hz). ' 参考例 67 へキサン酸 (4-ヒドロキシメチルフエニル)アミド 'H-NMR (CDC "): (50.916 (3H, t, J = 6.9 Hz), 1.30-1.48 (4H, m), 1.39 (3H, t, J = 7.2 Hz), 1.68-1.82 (2H, m ), 2.39 (3H, t, J = 7.8Hz), 4.36 (2H, q, J = 6.9Hz), 7.31 (1H, bs), 7.61 (2H, d, J = 8.7 Hz), 8.01 (2H, d , J = 8.7 Hz). 'Example 67 Hexanoic acid (4-hydroxymethylphenyl) amide
参考例 66で得られた 4-へキサノィルァミノ安息香酸 ェチルエステル(2.82 g, 10.7mmol)のテトラヒドロフラン(6.00ml)溶液に、水素化ホウ素リチウム (389 mg, 16.1 匪 ol) を加えた。 反応混合物を室温で 24時間かき混ぜた。 さらに水素 化ホウ素リチウム (389 mg, 16.1 匪 ol) を反応混合物に加え、 室温で 17.5時間 かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (690 mg, 29%) を無定形晶として得た。 To a solution of 4-hexanoylaminobenzoic acid ethyl ester (2.82 g, 10.7 mmol) obtained in Reference Example 66 in tetrahydrofuran (6.00 ml), lithium borohydride (389 mg, 16.1 bandol) was added. The reaction mixture was stirred at room temperature for 24 hours. Further, lithium borohydride (389 mg, 16.1 bandol) was added to the reaction mixture, and the mixture was stirred at room temperature for 17.5 hours, and then added with 1N hydrochloric acid and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (690 mg, 29%) as amorphous crystals.
IR ( Br) V: 3700-2500, 2953, 2930, 2863, 1657, 1605, 1541 cm"1. IR (Br) V: 3700-2500, 2953, 2930, 2863, 1657, 1605, 1541 cm " 1 .
'H-NMR (CDC13) : 60.911 (3H, t, J=6.9Hz), 1.38-1.44 (4H, m), 1.60-1.80 (2H, m), 2.35 (2H, t, J=7.8 Hz), 4.65 (2H, s), 7.22 (1H, bs), 7.31 (2H, d, J=8.7 Hz), 7.50 (2H, d, J=8.1 Hz). 'H-NMR (CDC1 3) : 60.911 (3H, t, J = 6.9Hz), 1.38-1.44 (4H, m), 1.60-1.80 (2H, m), 2.35 (2H, t, J = 7.8 Hz) , 4.65 (2H, s), 7.22 (1H, bs), 7.31 (2H, d, J = 8.7 Hz), 7.50 (2H, d, J = 8.1 Hz).
参考例 68 Reference Example 68
へキサン酸 (4-クロロメチルフエニル)アミド Hexanoic acid (4-chloromethylphenyl) amide
参考例 67で得られたへキサン酸 (4-ヒドロキシメチルフエニル)アミド(690 mg, 3.12mmol) のテトラヒドロフラン (15.0ml) 溶液に、 メタンスルホニル クロリ ド (0.290 ml, 3.74讓 ol) とトリエチルァミン (0.652 ml, 4.68 mmol) を氷冷 下順次加えた。 反応混合物を室温で 22時間かき混ぜた後、 1N塩酸を加えて酢酸 ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題 記化合物 (640 mg, 85%) を合成した。 酢酸ェチル-ジェチルェ一テルから再結晶 して融点 75.0-77.0°Cの無色結晶を得た。 To a solution of hexanoic acid (4-hydroxymethylphenyl) amide (690 mg, 3.12 mmol) obtained in Reference Example 67 in tetrahydrofuran (15.0 ml) was added methanesulfonyl chloride (0.290 ml, 3.74 alcohol) and triethyl alcohol. Min (0.652 ml, 4.68 mmol) was added sequentially under ice cooling. After stirring the reaction mixture at room temperature for 22 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was evaporated under reduced pressure to synthesize the title compound (640 mg, 85%). Recrystallization from ethyl acetate-ethyl ether gave colorless crystals having a melting point of 75.0-77.0 ° C.
IR (KBr) リ: 3318, 3193, 3119, 3048, 2935, 2932, 2863, 1659, 1599, 1537 cm—1. lH-NMR (CDC13) : δ 0.911 (3Η, t, J=6.9Hz), 1.30-1.42 (4H, m), 1.68-1.80 (2H, m), 2.35 (2H, t, J=7.5 Hz), 4.56 (2H, s), 7.18 (1H, bs), 7.34 (2H, d, J=8. Hz), 7.52 (2H, d, J=8. Hz). IR (KBr) Re:. 3318, 3193, 3119, 3048, 2935, 2932, 2863, 1659, 1599, 1537 cm- 1 l H-NMR (CDC1 3): δ 0.911 (3Η, t, J = 6.9Hz) , 1.30-1.42 (4H, m), 1.68-1.80 (2H, m), 2.35 (2H, t, J = 7.5 Hz), 4.56 (2H, s), 7.18 (1H, bs), 7.34 (2H, d , J = 8. Hz), 7.52 (2H, d, J = 8. Hz).
元素分析値: C13H18N0C1 -0.2H20として Elemental analysis: as C 13 H 18 N0C1 -0.2H 20
計算値 (¾) : C, 64.17; H, 7.62; N, 5.76. Calculated value (¾): C, 64.17; H, 7.62; N, 5.76.
実測値 ( ) : C, 64.23; Η, 7.69; Ν, 5.44. 参考例 69 Observed value (): C, 64.23; Η, 7.69; Ν, 5.44. Reference Example 69
(2, 2, 2 -トリフルォロエトキシ)酢酸ェチルエステル (2,2,2-Trifluoroethoxy) acetic acid ethyl ester
水素化ナトリウム(1.43g, 35.6讓01) (60 油性) のテトラヒドロフラン(40.0 ml) の懸濁液に、 2, 2, 2-トリフルォロエタノール (3.00 g, 29.7腿01) のテトラ ヒドロフラン (10.0ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これにブロモ酢 酸ェチル (3.80 ml, 32.7丽 ol) のテトラヒドロフラン (10.0 ml) 溶液を加え、 さらに室温で 15.5時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで 抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (4.62 g, 85%) を油状物として得た。 To a suspension of sodium hydride (1.43 g, 35.6 0101) (60 oily) in tetrahydrofuran (40.0 ml) was added 2,2,2-trifluoroethanol (3.00 g, 29.7 thigh 01) in tetrahydrofuran (10.0 g). ml), and the mixture was stirred under ice-cooling for 5 minutes. To this was added a solution of ethyl bromoacetate (3.80 ml, 32.7 mol) in tetrahydrofuran (10.0 ml), and the mixture was further stirred at room temperature for 15.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (4.62 g, 85%) as an oil.
IR (KBr) V: 2961, 2926, 1755 cm"1. IR (KBr) V: 2961, 2926, 1755 cm " 1 .
Ή-NMR (CDC13) : (51.26 (3H, t, J-6.9 Hz), 3.79 (2H, s), 3.99 (2H, q, J=8.7 Hz), 4.26 (2H, s). Ή-NMR (CDC1 3): (51.26 (3H, t, J-6.9 Hz), 3.79 (2H, s), 3.99 (2H, q, J = 8.7 Hz), 4.26 (2H, s).
参考例 70 水素化アルミニウムリチウム(1.79g, 37.6mmol)のジェチルェ一テル(100 ml) 懸濁液に、 参考例 69で得られた(2, 2, 2-トリフルォロェトキシ)酢酸ェチルエス テル (4.62 g, 25.1 mmol) のジェチルエーテル (100 ml) 溶液を氷冷下加え、 反 応混合物を室温で 3時間かき混ぜた。 過剰の水素化アルミニウムリチウムを氷冷 下少量の水で分解し、 反応混合物を濾過、 ジェチルェ一テルで洗浄した。 濾液を 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (1.84 g, 51¾) を油状物 として得た。 Reference Example 70 A suspension of lithium aluminum hydride (1.79 g, 37.6 mmol) in ethyl ether (100 ml) was added to a suspension of ethyl (2,2,2-trifluoroethoxy) acetate obtained in Reference Example 69 (100 ml). A solution of 4.62 g, 25.1 mmol) in getyl ether (100 ml) was added under ice-cooling, and the reaction mixture was stirred at room temperature for 3 hours. Excess lithium aluminum hydride was decomposed with a small amount of water under ice-cooling, and the reaction mixture was filtered and washed with diethyl ether. After the filtrate dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (1.84 g, 51¾) was obtained as an oil.
¾-NMR (CDC13) : 51.95-2.06 (1H, m), 3.71-3.84 (4H, m), 3.90 (2H, q, J=8.8 Hz). ¾-NMR (CDC1 3): 51.95-2.06 (1H, m), 3.71-3.84 (4H, m), 3.90 (2H, q, J = 8.8 Hz).
参考例 71 Reference Example 71
メタンスルホン酸 2- (2, 2, 2-トリフルォロェトキシ)ェチルエステル Methanesulfonic acid 2- (2,2,2-trifluoroethoxy) ethyl ester
参考例 70 で得られた 2- (2, 2, 2-トリフルォロエトキシ)エタノール (1.84 g, 12.8應01) のテトラヒドロフラン (40.0ml) 溶液に、 メタンスルホニル クロリ ド (1.19 ml, 15.3 mmol) とトリエチルァミン (2.17 ml, 19.2 腿01) を氷冷下 順次加えた。 反応混合物を室温で 2時間かき混ぜた後、 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (2.62 g, 92%) を油状物として得た。 Methanesulfonyl chloride (1.19 ml, 15.3 mmol) was added to a solution of 2- (2,2,2-trifluoroethoxy) ethanol (1.84 g, 12.8 to 01) obtained in Reference Example 70 in tetrahydrofuran (40.0 ml). And triethylamine (2.17 ml, 19.2 thigh 01) were added sequentially under ice cooling. Stir the reaction mixture at room temperature for 2 hours, then add 1N hydrochloric acid and add ethyl acetate. Extracted. The obtained extract was washed with water, dried (MgSO 4), solvent was distilled off under reduced pressure the title of compound a (2.62 g, 92%) as an oil.
IR (KBr) V: 3029, 2924, 2853, 1354, 1279, 1173, 968, 924 cm—1. IR (KBr) V: 3029, 2924, 2853, 1354, 1279, 1173, 968, 924 cm— 1 .
Ή-NMR (CDC13) : 53.06 (3H, s), 3.91 (2H, q, J=12.6 Hz), 3.88-3.94 (2H, m), 4.37-4.43 (2H, m). Ή-NMR (CDC1 3): 53.06 (3H, s), 3.91 (2H, q, J = 12.6 Hz), 3.88-3.94 (2H, m), 4.37-4.43 (2H, m).
参考例 72 Reference Example 72
1 -ブロモ -4- (2, 2, 2-トリフルォロェトキシ)ブ夕ン 1-bromo-4- (2,2,2-trifluoroethoxy) butane
水素化ナトリウム (950 g, 23.8腿01) (60%油性) のテトラヒドロフラン (10.0 ml) の懸濁液に、 2, 2, 2 -トリフルォロエタノール (2.00 g, 19.8mmol) のテトラ ヒドロフラン (20.0 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 1,4-ジ ブロモブタン (1.20 ml, 9.90腿01) のテトラヒドロフラン (10.0,ml) 溶液を加. え、 さらに 80°Cで 16時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチル で抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合 物 (2.07 g, 40¾) を油状物として得た。 To a suspension of sodium hydride (950 g, 23.8 thigh 01) (60% oil) in tetrahydrofuran (10.0 ml) was added 2,2,2-trifluoroethanol (2.00 g, 19.8 mmol) in tetrahydrofuran (20.0 t ml), and the mixture was stirred under ice-cooling for 5 minutes. To this was added a solution of 1,4-dibromobutane (1.20 ml, 9.90 thigh 01) in tetrahydrofuran (10.0, ml), and the mixture was further stirred at 80 ° C for 16 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (2.07 g, 40¾) as an oil.
Ή-NMR (CDC13) : δ 1.64—2.06 (4Η, m), 3.45 (2Η, ί, J=6.6 Hz), 3.65 (2H, t, Ή-NMR (CDC1 3): δ 1.64-2.06 (4Η, m), 3.45 (2Η, ί, J = 6.6 Hz), 3.65 (2H, t,
J=6.2 Hz), 3.80 (2H, q, J=8.8 Hz). J = 6.2 Hz), 3.80 (2H, q, J = 8.8 Hz).
参考例 73 Reference Example 73
1 -ブロモ - 5-(2, 2, 2 -トリフルォロェトキシ)ペンタン 1-bromo-5- (2,2,2-trifluoroethoxy) pentane
水素化ナトリウム (475 g, 11.9mmol) (60 油性) のテトラヒドロフラン (10.0 ml) の懸濁液に、 2, 2, 2 -トリフルォロエタノール (1.00 g, 9.90腿01) のテトラ ヒドロフラン (10.0 mi) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 1,5 -ジ ブロモペンタン (1.50 ml, 10.9匪 ol) のテトラヒドロフラン (10.0 ml) 溶液を 加え、 さらに 80°Cで 16時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (2.23 g, 47¾) を油状物として得た。 To a suspension of sodium hydride (475 g, 11.9 mmol) (60 oily) in tetrahydrofuran (10.0 ml) was added 2,2,2-trifluoroethanol (1.00 g, 9.90 t01) in tetrahydrofuran (10.0 mi). ) The solution was added and stirred for 5 minutes under ice cooling. To this was added a solution of 1,5-dibromopentane (1.50 ml, 10.9 marl) in tetrahydrofuran (10.0 ml), and the mixture was further stirred at 80 ° C for 16 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title of compound a (2.23 g, 47¾) as an oil.
Ή-NMR (CDC13) : 61.38-1.73 (4H, in), 1.80-1.96 (2H, m), 3.42 (2H, t, J=6.8 Hz), 3.61 (2H, t, J=6.2 Hz), 3.80 (2H, q, J=8.8 Hz). Ή-NMR (CDC1 3): 61.38-1.73 (4H, in), 1.80-1.96 (2H, m), 3.42 (2H, t, J = 6.8 Hz), 3.61 (2H, t, J = 6.2 Hz), 3.80 (2H, q, J = 8.8 Hz).
参考例 74 Reference example 74
(3 -フルォロプロボキシ)酢酸ェチルエステル 水素化ナトリウム (622 g, 15.6raraol) (60%油性) のテトラヒドロフラン (5.00 ml) の懸濁液に、 グリコ一ル酸 ェチルエステル (1.50 g, 13.0 mmol) のテトラ ヒドロフラン (20.0 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに卜プロ モ— 3 -フルォロプロパン (1.87 g, 13.0 mmol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 さらに 60 で 19時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢 酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して 題記化合物 (680 mg, 15¾) を油状物として得た。 (3-Fluoropropoxy) acetic acid ethyl ester To a suspension of sodium hydride (622 g, 15.6raraol) (60% oil) in tetrahydrofuran (5.00 ml) was added a solution of ethyl gluconate (1.50 g, 13.0 mmol) in tetrahydrofuran (20.0 ml). The mixture was stirred for 5 minutes under ice cooling. To this was added a solution of tribromo-3-fluoropropane (1.87 g, 13.0 mmol) in tetrahydrofuran (5.00 ml), and the mixture was further stirred at 60 for 19 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (680 mg, 15¾) as an oil.
'H-NMR (CDC13) : 51.29 (3H, t, J=7.2 Hz), 2.01 (2H, dt, J=26.,l, 5.7 Hz), 3.67 (2H, t, J=6.3 Hz), 4.09 (2H, s), 4.23 (2H, q, J=6.9 Hz), 4.58 (2H, dt, J=47.1, 5.7 Hz). 'H-NMR (CDC1 3) : 51.29 (3H, t, J = 7.2 Hz), 2.01 (2H, dt, J = 26., l, 5.7 Hz), 3.67 (2H, t, J = 6.3 Hz), 4.09 (2H, s), 4.23 (2H, q, J = 6.9 Hz), 4.58 (2H, dt, J = 47.1, 5.7 Hz).
参考例 75 Reference Example 75
(4-フルォロブトキシ)酢酸ェチルエステル (4-Fluorobutoxy) acetic acid ethyl ester
水素化ナトリウム (415g, 10.4mmol) (60%油性) のテトラヒドロフラン (5.00 ml) の懸濁液に、 グリコール酸ェチルエステル (1.00 g, 8.65 腿 ol) のテトラ ヒドロフラン (10.0 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに卜プロ モ- 4-フルォロブタン (0.947 ml, 8.65 mmol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 さらに 60°Cで 14.5時間かき混ぜた。反応混合物に 1N塩酸を加えて 酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去し て題記化合物 (680 mg, 44¾) を油状物として得た。 To a suspension of sodium hydride (415 g, 10.4 mmol) (60% oil) in tetrahydrofuran (5.00 ml) was added a solution of ethyl glycolate (1.00 g, 8.65 t) in tetrahydrofuran (10.0 ml), and the mixture was cooled on ice. Stir for 5 minutes below. A solution of tripromo-4-fluorobutane (0.947 ml, 8.65 mmol) in tetrahydrofuran (5.00 ml) was added thereto, and the mixture was further stirred at 60 ° C for 14.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (680 mg, 44¾) as an oil.
IR (KBr) v: 2963, 2909, 2876, 1755, 1206, 1140 cm"1. IR (KBr) v: 2963, 2909, 2876, 1755, 1206, 1140 cm " 1 .
¾-N R (CDC13) : 51.29 (3H, t, J=7.0 Hz), 1.70-1.95 (4H, m), 3.58 (2H, t, J=6.2 Hz), 4.07 (2H, s), 4.22 (2H, q, J=7.4 Hz), 4.49 (2H, dt, J-47.2, 5.8 Hz). ¾-NR (CDC1 3): 51.29 (3H, t, J = 7.0 Hz), 1.70-1.95 (4H, m), 3.58 (2H, t, J = 6.2 Hz), 4.07 (2H, s), 4.22 ( 2H, q, J = 7.4 Hz), 4.49 (2H, dt, J-47.2, 5.8 Hz).
参考例 76 Reference Example 76
(5-フルォロペンチルォキシ)酢酸 ェチルエステル (5-Fluoropentyloxy) acetic acid ethyl ester
参考例 74と同様の方法により、 1-ブロモ -3-フルォロプロパンの代わりに 1 -ブ ロモ- 5-フルォロペンタン (2.41 g, 14.3 mmol) を用いて、 グリコ一ル酸ェチル エステル (3.00.g, 26.0 mmol) から題記化合物 (2.11 g, 84%) を油状物として 得た。 IR (KBr) v: 2964, 2870, 1755 cm-1. In the same manner as in Reference Example 74, 1-bromo-5-fluoropentane (2.41 g, 14.3 mmol) was used in place of 1-bromo-3-fluoropropane, and glycoethyl ester (3.00.g, 26.0 g) was used. mmol) to give the title compound (2.11 g, 84%) as an oil. IR (KBr) v: 2964, 2870, 1755 cm- 1 .
Ή-NMR (CDC13) : 51.26 (3H, t, J=6.0 Hz), 1.42-1.88 (6H, m), 3.55 (2H, t, J=6.2 Hz), 4.07 (2H, s), 4.22 (2H, q, J=7.4 Hz), 4.45 (2H, dt, J=47.4, 6.0 Hz). Ή-NMR (CDC1 3): 51.26 (3H, t, J = 6.0 Hz), 1.42-1.88 (6H, m), 3.55 (2H, t, J = 6.2 Hz), 4.07 (2H, s), 4.22 ( 2H, q, J = 7.4 Hz), 4.45 (2H, dt, J = 47.4, 6.0 Hz).
参考例 77 Reference Example 77
2- (3 -フルォロプロポキシ)エタノール 2- (3-fluoropropoxy) ethanol
水素化アルミニウムリチウム (295 mg, 6.21匪01) のジェチルエーテル (20.0 ml) 懸濁液に、 参考例 74で得られた(3-フルォロプロボキシ)酢酸ェチルエステ ル (680 mg, 4.14 mmol) のジェチルエーテル (10.0 ml) 溶液を氷冷下加え、 反 応混合物を室温で 2時間かき混ぜた。 過剰の水素化アルミニウムリチウムを氷冷 下少量の水で分解し、 反応混合物を濾過、 ジェチルエーテルで洗浄した。 濾液を 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (260 mg, 36¾) を油状物 として得た。 A suspension of lithium aluminum hydride (295 mg, 6.21 band01) in getyl ether (20.0 ml) was added to the (3-fluoropropoxy) acetate ethyl ester (680 mg, 4.14 mmol) obtained in Reference Example 74. )) In dimethyl ether (10.0 ml) was added under ice-cooling, and the reaction mixture was stirred at room temperature for 2 hours. Excess lithium aluminum hydride was decomposed with a small amount of water under ice cooling, and the reaction mixture was filtered and washed with getyl ether. After the filtrate dried (MgSO 4), the title compound solvent was evaporated under reduced pressure (260 mg, 36¾) was obtained as an oil.
Ή一 NMR (CDC13) : 51.99 (2H, dt, J=26.1, 5.7 Hz), 3.54-3.59 (2H, m), 3.62 (2H, t, J=6.0 Hz), 3.70-3.78 (2H, m), 4.56 (2H, dt, J=47.1, 5.7 Hz). 参考例 78 Ή one NMR (CDC1 3): 51.99 ( 2H, dt, J = 26.1, 5.7 Hz), 3.54-3.59 (2H, m), 3.62 (2H, t, J = 6.0 Hz), 3.70-3.78 (2H, m ), 4.56 (2H, dt, J = 47.1, 5.7 Hz).
2_ (4-フルォロブトキシ)エタノール 2_ (4-fluorobutoxy) ethanol
水素化アルミニウムリチウム (272 mg, 5.72匪01) のジェチルェ一テル (20.0 ml) 懸濁液に、 参考例 75で得られた(4-フルォロブトキシ)酢酸ェチルエステル (680 mg, 3.82 mmol) のジェチルェ一テル (10.0 ml) 溶液を氷冷下加え、 反応 混合物を室温で 3時間かき混ぜた。 過剰の水素化アルミニウムリチウムを氷冷下 少量の水で分解し、 反応混合物を濾過、 ジェチルエーテルで洗浄した。 濾液を乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (200 mg, 38¾) を油状物と して得た。 In a suspension of lithium aluminum hydride (272 mg, 5.72 band01) in ethyl acetate (20.0 ml), ethyl 4- (butyrolbutoxy) acetic acid ethyl ester (680 mg, 3.82 mmol) obtained in Reference Example 75 was added. (10.0 ml) The solution was added under ice-cooling, and the reaction mixture was stirred at room temperature for 3 hours. Excess lithium aluminum hydride was decomposed with a small amount of water under ice cooling, and the reaction mixture was filtered and washed with getyl ether. After filtrate Drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (200 mg, 38¾) was obtained as an oil.
IR (KBr) リ : 3700-3100, 2926, 2872, 1123, 1067 cm"1. IR (KBr): 3700-3100, 2926, 2872, 1123, 1067 cm " 1 .
Ή-NMR (CDC13) : δ 1.69-1.79 (4Η, m), 2.03 (1H, t, J=5.7 Hz), 3.50-3.58 (4H, m), 3.74 (2H, dt, J=5.1, 3.9 Hz), 4.48 (2H, dt, J=47. , 6.0 Hz). Ή-NMR (CDC1 3): δ 1.69-1.79 (4Η, m), 2.03 (1H, t, J = 5.7 Hz), 3.50-3.58 (4H, m), 3.74 (2H, dt, J = 5.1, 3.9 Hz), 4.48 (2H, dt, J = 47., 6.0 Hz).
参考例 79 Reference Example 79
2 -(5 -フルォロペンチルォキシ)エタノール 水素化アルミニウムリチウム (781 mg, 16.5 mmol) のジェチルエーテル (60.0 ml) 懸濁液に、 参考例 76で得られた(5-フルォロペンチルォキシ)酢酸ェチルェ ステル (2. llg, 11.0 mmol) のジェチルエーテル (20. Oral) 溶液を氷冷下加え、 反応混合物を室温で 50分間かき混ぜた。過剰の水素化アルミニウムリチウムを氷 冷下少量の水で分解し、 反応混合物を濾過、 ジェチルエーテルで洗浄した。 濾液 を乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (910 mg, 55¾) を油状 物として得た。 2- (5-fluoropentyloxy) ethanol A suspension of lithium aluminum hydride (781 mg, 16.5 mmol) in getyl ether (60.0 ml) was added to the (5-fluoropentyloxy) acetate ethyl ester (2. llg, 11.0 g) obtained in Reference Example 76. (mmol) of getyl ether (20. Oral) was added under ice cooling, and the reaction mixture was stirred at room temperature for 50 minutes. Excess lithium lithium hydride was decomposed with a small amount of water under ice cooling, and the reaction mixture was filtered and washed with getyl ether. After the filtrate dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (910 mg, 55¾) as an oil.
¾-NMR (CDC13) : δ 1.44-1.86 (6Η, m), 2.02 (1H, t, J=6.2 Hz), 3.50 (2H, t, J=6.6 Hz), 3.48-3.56 (2H, m), 3.68-3.78 (2H, m), 4.46 (2H, dt, 1=47.2, 5.8 Hz) . ¾-NMR (CDC1 3): δ 1.44-1.86 (6Η, m), 2.02 (1H, t, J = 6.2 Hz), 3.50 (2H, t, J = 6.6 Hz), 3.48-3.56 (2H, m) , 3.68-3.78 (2H, m), 4.46 (2H, dt, 1 = 47.2, 5.8 Hz).
参考例 80 Reference Example 80
メタンスルホン酸 2 -(3-フルォロプロボキシ)ェチルエステル Methanesulfonic acid 2- (3-fluoropropoxy) ethyl ester
参考例 77 で得られた 2-(3-フルォロプロボキシ)エタノール (260 mg, 2.13 mmol)のテトラヒドロフラン(10.0 ml)溶液に、メ夕ンスルホニル クロリド(0.198 il, 2.55 mmol) とトリエチルァミン (0.445 ml, 3.19 龍 ol) を氷冷下順次加え た。反応混合物を室温で 2.5時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(320 mg, 83%) を油状物として得た。 To a solution of 2- (3-fluoropropoxy) ethanol (260 mg, 2.13 mmol) obtained in Reference Example 77 in tetrahydrofuran (10.0 ml) was added methylsulfonyl chloride (0.198 il, 2.55 mmol) and triethyla. Min (0.445 ml, 3.19 dragon ol) was added sequentially under ice cooling. After stirring the reaction mixture at room temperature for 2.5 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (320 mg, 83%) as an oil.
Ή-NMR (CDC13) : δΐ.98 (2Η, dt, J=26.4, 6.0 Hz), 3.06 (3H, s), 3.63 (2H, t, J-6.3 Hz), 3.70-3.75 (2H, m), 4.35-4.39 (2H, m), 4.55 (2H, dt, J=47. , 6.0 Hz). Ή-NMR (CDC1 3): δΐ.98 (2Η, dt, J = 26.4, 6.0 Hz), 3.06 (3H, s), 3.63 (2H, t, J-6.3 Hz), 3.70-3.75 (2H, m ), 4.35-4.39 (2H, m), 4.55 (2H, dt, J = 47., 6.0 Hz).
参考例 81 Reference 81
メ夕ンスルホン酸 2- (4-フルォロブトキシ)ェチルエステル 2- (4-fluorobutoxy) ethyl ester of maleic sulfonic acid
参考例 78で得られた 2 - (4-フルォロブトキシ)エタノール (200 mg, 1.47 mmol) のテトラヒドロフラン(10.0ml)溶液に、メタンスルホニル クロリド(0.136 ml, 1.76 mmol) とトリエチルァミン (0.307 ml, 2.20 mmol) を氷冷下順次加えた。 反応混合物を室温で 1時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(242 mg, 77¾) を油状物として得た。 IR (KBr) リ : 2951, 2874, 1352, 1175, 974, 922 cm"1. In a solution of 2- (4-fluorobutoxy) ethanol (200 mg, 1.47 mmol) obtained in Reference Example 78 in tetrahydrofuran (10.0 ml), methanesulfonyl chloride (0.136 ml, 1.76 mmol) and triethylamine (0.307 ml, 2.20) were added. mmol) were sequentially added under ice cooling. After stirring the reaction mixture at room temperature for 1 hour, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (242 mg, 77¾) as an oil. IR (KBr): 2951, 2874, 1352, 1175, 974, 922 cm " 1 .
Ή-NMR (CDC13) : ά 1.66-1.88 (4H, m), 3.06 (3H, s), 3.54 (2H, t, J=6.3 Hz), 3.68-3.73 (2H, m), 4.34-4. 0 (2H, m), 4.45 (2H, dt, J=47.4, 6.0 Hz). 参考例 82 Ή-NMR (CDC1 3): ά 1.66-1.88 (4H, m), 3.06 (3H, s), 3.54 (2H, t, J = 6.3 Hz), 3.68-3.73 (2H, m), 4.34-4. 0 (2H, m), 4.45 (2H, dt, J = 47.4, 6.0 Hz). Reference example 82
メタンスルホン酸 2- (5-フルォロペンチルォキシ)ェチルエステル Methanesulfonic acid 2- (5-fluoropentyloxy) ethyl ester
参考例 79で得られた 2- (5-フルォロペンチルォキシ)エタノール(910mg, 6.06 mmol)のテトラヒドロフラン(30.0 ml)溶液に、メ夕ンスルホニル クロリド(0.563 ml, 7.27 mmol) とトリエチルァミン (1.27 ml, 9.09 mmol) を氷冷下順次加えた。 反応混合物を室温で 1時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(1.28 g, 93%) を油状物として得た。 To a solution of 2- (5-fluoropentyloxy) ethanol (910 mg, 6.06 mmol) obtained in Reference Example 79 in tetrahydrofuran (30.0 ml) was added methyl sulfonyl chloride (0.563 ml, 7.27 mmol) and triethyla. Min (1.27 ml, 9.09 mmol) was added sequentially under ice cooling. After stirring the reaction mixture at room temperature for 1 hour, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (1.28 g, 93%) as an oil.
IR (KBr) V: 3027, 2944, 2968, 1354, 1177, 1130, 1019, 974, 924 cm"1. — NMR (CDCI3) : (51.40-1.90 (6H, m), 3.06 (3H, s), 3.51 (2H, t, J-6. Hz), 3.66-3.74 (2H, m), 4.30-4.40 (2H, m), 4.45 (2H, dt, J=47.4, 6.4 Hz). 参考例 83 IR (KBr) V: 3027, 2944, 2968, 1354, 1177, 1130, 1019, 974, 924 cm " 1. — NMR (CDCI3): (51.40-1.90 (6H, m), 3.06 (3H, s), 3.51 (2H, t, J-6.Hz), 3.66-3.74 (2H, m), 4.30-4.40 (2H, m), 4.45 (2H, dt, J = 47.4, 6.4 Hz) .Reference example 83
(2-フルォロエトキシ)酢酸 ェチルエステル (2-Fluoroethoxy) acetic acid ethyl ester
水素化ナトリウム(2.14g, 53.4醒01) (60 油性)のテトラヒドロフラン(40.0 ml) の懸濁液に、 2-フルォロエタノール (3.00g, 44.5腿01) のテトラヒドロフ ラン (10.0 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これにブロモ酢酸ェチル (7.77 ml, 66.7 mmol) のテトラヒドロフラン (10.0 ml) 溶液を加え、 さらに室 温で 45時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(9.25 g, 60¾) を油状物として得た。 To a suspension of sodium hydride (2.14 g, 53.4 virgin 01) (60 oily) in tetrahydrofuran (40.0 ml), add a solution of 2-fluoroethanol (3.00 g, 44.5 thigh 01) in tetrahydrofuran (10.0 ml) And stirred under ice cooling for 5 minutes. To this was added a solution of ethyl bromoacetate (7.77 ml, 66.7 mmol) in tetrahydrofuran (10.0 ml), and the mixture was further stirred at room temperature for 45 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (9.25 g, 60¾) as an oil.
Ή-NMR (CDC13) : (51.31 (3H, t, J=7.2 Hz), 3.84 (2H, s), 3.84 (2H, dt, J=29.7, 3.9 Hz), 4.23 (2H, q, J=7.2 Hz), 4.61 (2H, dt, J=46.2, 4.2 Hz). Ή-NMR (CDC1 3): (51.31 (3H, t, J = 7.2 Hz), 3.84 (2H, s), 3.84 (2H, dt, J = 29.7, 3.9 Hz), 4.23 (2H, q, J = 7.2 Hz), 4.61 (2H, dt, J = 46.2, 4.2 Hz).
参考例 84 Reference Example 84
2- (2 -フルォロェ卜キシ)エタノール 2- (2-fluoroethoxy) ethanol
水素化アルミニウムリチウム(4.38g, 92.4腿01)のジェチルエーテル(400 ml) 懸濁液に、参考例 83で得られた(2-フルォロエトキシ)酢酸ェチルエステル(9.25 g, 61.6謹01) のジェチルエーテル (100 ml) 溶液を氷冷下加え、 反応混合物を 室温で 1.5時間かき混ぜた。 過剰の水素化アルミニゥムリチウムを氷冷下少量の 水で分解し、反応混合物を濾過、ジェチルエーテルで洗浄した。濾液を乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (1.87 g, 28%) を油状物として得た。 Ή-NMR (CDC13) : 53.51-3.84 (6H, m), 4.59 (2H, dt, J-47.7, 4.2 Hz). 参考例 85 A suspension of lithium aluminum hydride (4.38 g, 92.4 thigh 01) in getyl ether (400 ml) was added to the (2-fluoroethoxy) acetic acid ethyl ester (9.25) obtained in Reference Example 83. g, 61.6) was added under ice-cooling, and the reaction mixture was stirred at room temperature for 1.5 hours. Excess lithium aluminum hydride was decomposed with a small amount of water under ice-cooling, and the reaction mixture was filtered and washed with getyl ether. After the filtrate dried (MgSO 4), to give the title compound (1.87 g, 28%) as an oil and the solvent was evaporated under reduced pressure. Ή-NMR (CDC1 3): . 53.51-3.84 (6H, m), 4.59 (2H, dt, J-47.7, 4.2 Hz) Reference Example 85
メタンスルホン酸 2_(2-フルォロエトキシ)ェチルエステル Methanesulfonic acid 2_ (2-fluoroethoxy) ethyl ester
参考例 84で得られた 2_(2 -フルォロエトキシ)エタノール (1.87g, 17.3腿01) のテトラヒドロフラン (100ml) 溶液に、 メタンスルホニル クロリド (1.34ml, 20.8 mmol) とトリエチルァミン (2.89 ml, 20.8腿01) を氷冷下順次加えた。 反 応混合物を室温で 2.5時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(2.79 g, 87¾) を油状物として得た。 In a tetrahydrofuran (100 ml) solution of 2_ (2-fluoroethoxy) ethanol (1.87 g, 17.3 thigh 01) obtained in Reference Example 84, methanesulfonyl chloride (1.34 ml, 20.8 mmol) and triethylamine (2.89 ml, 20.8 thigh) were added. 01) was added sequentially under ice cooling. After stirring the reaction mixture at room temperature for 2.5 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (2.79 g, 87¾) as an oil.
Ή-NMR (CDC13) : 53.07 (3H, s), 3.69-3.84 (4H, m), 4.38-4.42 (2H, m), 4.58 (2H, dt, J=46.5, 5.4 Hz). Ή-NMR (CDC1 3): 53.07 (3H, s), 3.69-3.84 (4H, m), 4.38-4.42 (2H, m), 4.58 (2H, dt, J = 46.5, 5.4 Hz).
参考例 86 Reference Example 86
メタンスルホン酸テトラヒドロピラン -4_メチル Methanesulfonic acid tetrahydropyran-4_methyl
テトラヒドロピラン- 4, 4-ジカルポン酸 (純度 65, 3.04 g, 11.3匪 ol)のァセ トニトリル(300 ml)溶液をアルゴン雰囲気下、 酸化第 1 銅(84 mg, 0.567腿 ol) 存在下に 24時間加熱還流した。 反応液を濃縮してメタノール(60 ml)で溶解し、 濃塩酸(1.88ml, 22.6蘭 ol)を加えて室温で 25 時間攪拌した。 反応液を中和後、 食塩で飽和し、 酢酸ェチルで抽出した。 脱水乾燥後、 濃縮して褐色油状物 (3.0 g) を得た。 これを THF (60 ml)に溶解し、 0°Cで攪拌しながら水素化リチウムアルミ ニゥム (純度 80, 0.54 g, 11.3匪 ol) の粉末を数回に分けて加え、 0°Cで 60 分間攪拌した。 反応液に 0.5M クェン酸水溶液 (23 ml)を加え、 食塩で飽和し、 THF (50 ml)で 2回抽出した。 THFを留去し、 酢酸ェチル(100 ml)で希釈後、 乾燥、 濃縮して淡黄色油状物(1.68 g)を得た。 この油状物をクロ口ホルム (30 ml)に溶 解し、 0°Cで攪拌しながらメタンスルホニルクロリド (0.89 ml, 11.3顧01) を加 え、 次いでトリェチルァミン(1.72 ml, 12.4 mmol)を滴下し、 反応液を 0°Cで 60 分間攪拌した。 反応液を濃縮して酢酸ェチル (100 ml)で希釈し、 水および飽和食 塩水で洗浄し、 乾燥、 濃縮した。 残渣をシリカゲル (20 g)のクロマトグラフィー (酢酸ェチルーへキサン、 3:7-4:6)で精製し、 メタンスルホン酸テ卜ラヒドロピ ラン- 4-メチル(0.77 g, 3.96 mmol)を淡褐色油状物として得た。 A solution of tetrahydropyran-4,4-dicarponic acid (purity 65, 3.04 g, 11.3 ol) in acetonitrile (300 ml) was placed in an argon atmosphere in the presence of cuprous oxide (84 mg, 0.567 ol). Heated to reflux for an hour. The reaction solution was concentrated and dissolved with methanol (60 ml). Concentrated hydrochloric acid (1.88 ml, 22.6 ranol) was added and the mixture was stirred at room temperature for 25 hours. After neutralizing the reaction solution, it was saturated with sodium chloride and extracted with ethyl acetate. After dehydration and drying, the mixture was concentrated to give a brown oil (3.0 g). This was dissolved in THF (60 ml), and lithium aluminum hydride (purity 80, 0.54 g, 11.3 bandol) powder was added in several portions while stirring at 0 ° C, and the mixture was added at 0 ° C for 60 minutes. Stirred. A 0.5 M aqueous solution of cunic acid (23 ml) was added to the reaction solution, and the mixture was saturated with sodium chloride and extracted twice with THF (50 ml). The THF was distilled off, diluted with ethyl acetate (100 ml), dried and concentrated to obtain a pale yellow oil (1.68 g). This oil was dissolved in chloroform (30 ml), methanesulfonyl chloride (0.89 ml, 11.3) was added with stirring at 0 ° C, and then triethylamine (1.72 ml, 12.4 mmol) was added dropwise. The reaction solution is kept at 0 ° C for 60 Stirred for minutes. The reaction solution was concentrated, diluted with ethyl acetate (100 ml), washed with water and saturated saline, dried and concentrated. The residue was purified by silica gel (20 g) chromatography (ethyl acetate-hexane, 3: 7-4: 6), and tetrahydropyran-4-methyl methanesulfonate (0.77 g, 3.96 mmol) was added to a pale brown oil. Obtained as a product.
'H-NMR (200MHz, CDC13): 51.40 (2H, dq, J= 4.8, 12.0 Hz), 1.68 (2H, brdq, J= 12.4, 2.0 Hz), 2.03 (1H, m), 3.02 (3H, s), 3.40 (2H, dt, J= 2.2, 11.6 Hz), 4.00 (2H, brdd, J= 11,6, 3.9 Hz), 4.07 (2H, d, J= 6.6 Hz). 'H-NMR (200MHz, CDC1 3): 51.40 (2H, dq, J = 4.8, 12.0 Hz), 1.68 (2H, brdq, J = 12.4, 2.0 Hz), 2.03 (1H, m), 3.02 (3H, s), 3.40 (2H, dt, J = 2.2, 11.6 Hz), 4.00 (2H, brdd, J = 11,6, 3.9 Hz), 4.07 (2H, d, J = 6.6 Hz).
参考例 87 Reference Example 87
メタンスルホン酸 2- (テトラヒドロピラン- 4-ィル)ェチル Methanesulfonic acid 2- (tetrahydropyran-4-yl) ethyl
水素化ナトリウム (油性 60%、 0.40 g, 10.0腿 ol)のトルエン(15 ml)懸濁液に 室温で攪拌しながらエトキシカルポニルメチルホスホン酸ジェチル(2.05 ml, 10.0 mmol) のトルエン(5 ml)溶液を 20分間かけて滴下し、 反応液を 70分間攪拌 した。 この反応液にテトラヒドロピラン- 4-オン (0.983 ml, 10.0 mmol) のトル ェン(5 ml)溶裨を 50分間かけて滴下し、 反応液を室温で 2時間、 60°C で 2時間 攪拌した。 反応液に 1N塩酸 (10 ml)および水(40 ml)を加え、 酢酸ェチルで抽出 した。 有機層を水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮し て無色油状物 (約 2g) を得た。 A toluene (5 ml) solution of ethoxycarbonylmethylphosphonate getyl (2.05 ml, 10.0 mmol) was added to a suspension of sodium hydride (oil-based 60%, 0.40 g, 10.0 tmol) in toluene (15 ml) while stirring at room temperature. The solution was added dropwise over 20 minutes and the reaction solution was stirred for 70 minutes. To this reaction solution, a solution of tetrahydropyran-4-one (0.983 ml, 10.0 mmol) in toluene (5 ml) was added dropwise over 50 minutes, and the reaction solution was stirred at room temperature for 2 hours and at 60 ° C for 2 hours. did. 1N Hydrochloric acid (10 ml) and water (40 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate. The organic layer was washed with water and saturated saline, dried over sodium sulfate, and concentrated to give a colorless oil (about 2 g).
この無色油状物 (約 2 g) をメタノール (40ml)に溶解し、 10% パラジウム炭素 (324 mg, 52%含水) 存在下、 水素雰囲気下に室温で 4時間攪拌した。 反応液を濾 過して触媒を除去し、濃縮乾固して粗製のテトラヒドロピラン- 4-酢酸ェチル(1.7 g)を無色油状物として得た。 This colorless oil (about 2 g) was dissolved in methanol (40 ml), and the mixture was stirred for 4 hours at room temperature under a hydrogen atmosphere in the presence of 10% palladium on carbon (324 mg, containing 52% water). The reaction solution was filtered to remove the catalyst, and concentrated to dryness to obtain crude tetrahydropyran-4-ethyl acetate (1.7 g) as a colorless oil.
上記粗製のテトラヒドロピラン- 4-酢酸ェチル(1.7 g)を THF (30 ml)に溶解し、 0 で攪拌しながら水素化リチウムアルミニウム (純度 80%, 0.47 g, 10 mmol) の 粉末を数回に分けて加え、 (T で 4時間攪拌した。 反応液に 0.5M クェン酸水溶液 (20ml)を加え、食塩で飽和し、 THFおよび酢酸ェチルで抽出した。 THFを留去し、 酢酸ェチル(70 ml)で希釈後、 乾燥、 濃縮して粗製の 2- (テトラヒドロピラン- 4 - ィル)エタノールの淡褐色油状物を得た。 この油状物をクロ口ホルム(15 ml)に溶 解し、 0°Cで攪拌しながらメタンスルホニルクロリド (0.783 ml, 10.0 mmol) を 加え、 次いでトリェチルァミン(1.52 ml, 11.0 mmol)のクロ口ホルム(5 ml)溶液 を 15分かけて滴下し、 反応液を 0°Cで 65分間攪拌した。 反応液を濃縮して酢酸 ェチル(100 ml)で希釈し、 飽和炭酸水素ナトリウム水溶液および飽和食塩水で洗 浄し、 乾燥、 濃縮した。 残渣をシリカゲル (20 g)のクロマトグラフィー (酢酸ェ チルーへキサン、 2:8-4:6) で精製し、 酢酸ェチルーへキサン(3:7- 4:6)の画分を 濃縮乾固してメタンスルホン酸 2- (テトラヒドロビラン- 4-ィル)ェチル (0.84 g, 4.03 mmol)を淡黄色油状物として得た。 The above crude tetrahydropyran-4-ethyl acetate (1.7 g) was dissolved in THF (30 ml), and the powder of lithium aluminum hydride (purity 80%, 0.47 g, 10 mmol) was added several times while stirring at 0. The reaction mixture was stirred for 4 hours with T. To the reaction solution was added a 0.5 M aqueous solution of citric acid (20 ml), and the mixture was saturated with sodium chloride and extracted with THF and ethyl acetate. ), Dried and concentrated to give crude 2- (tetrahydropyran-4-yl) ethanol as a pale brown oil, which was dissolved in chloroform (15 ml). While stirring at ° C, methanesulfonyl chloride (0.783 ml, 10.0 mmol) was added, and then a solution of triethylamine (1.52 ml, 11.0 mmol) in chloroform (5 ml) was added. Was added dropwise over 15 minutes, and the reaction solution was stirred at 0 ° C for 65 minutes. The reaction solution was concentrated, diluted with ethyl acetate (100 ml), washed with a saturated aqueous solution of sodium hydrogencarbonate and brine, dried and concentrated. The residue was purified by silica gel (20 g) chromatography (ethyl acetate-hexane, 2: 8-4: 6), and the ethyl acetate-hexane (3: 7-4: 6) fraction was concentrated to dryness. This gave 2- (tetrahydrovirane-4-yl) ethyl methanesulfonate (0.84 g, 4.03 mmol) as a pale yellow oil.
Ή-NMR (200MHz, CDC13): 51.33 (2H, m), 1.64 (2H, brd, J= 14.0 Hz), 1.60 (1H, m), 1.70 (2H, brdd, J= 7.0, 4.4 Hz), 3.02 (3H, s), 3.99 (2H, dt, J= 1.8, 11.9 Hz), 3.96 (2H, brdd, J= 11.6, 4.4 Hz), 4.29 (2H, t, J= 6.2 Hz). Ή-NMR (200MHz, CDC1 3 ): 51.33 (2H, m), 1.64 (2H, brd, J = 14.0 Hz), 1.60 (1H, m), 1.70 (2H, brdd, J = 7.0, 4.4 Hz), 3.02 (3H, s), 3.99 (2H, dt, J = 1.8, 11.9 Hz), 3.96 (2H, brdd, J = 11.6, 4.4 Hz), 4.29 (2H, t, J = 6.2 Hz).
参考例 88 Reference Example 88
3 -ブロモ -2_(4-フルオロフェニル) プロペン 3-bromo-2_ (4-fluorophenyl) propene
2-(4_フルオロフェニル)プロペン (9.94 g, 71.5 mfflol)および N-ブロモスクシ ミド (13.6 g, 75.1 mmol)の酢酸ェチル(100 ml)懸濁液に、加熱還流しながら AIBN (120 mg, 0.175 mmol)を 10分毎に 12回加え、 さらに反応液を 4時間加熱還流し た。 反応液を濃縮し、 へキサン (60 ml)を加えて希釈した。不溶物を濾別してへキ サン-ジイソプロピルエーテル (3:1, 60 ml)で洗浄した。 母液および洗浄液を合わ せて濃縮乾固して 3-ブロモ -2- (4-フルオロフェニル) プロペン(純度 70%, 11.7g, 約 35 mmol)を淡黄色油状物として得た。 AIBN (120 mg, 0.175 g) was added to a suspension of 2- (4_fluorophenyl) propene (9.94 g, 71.5 mfflol) and N-bromosuccinimide (13.6 g, 75.1 mmol) in ethyl acetate (100 ml) while heating under reflux. mmol) was added 12 times every 10 minutes, and the reaction solution was further heated to reflux for 4 hours. The reaction solution was concentrated, and diluted with hexane (60 ml). The insoluble material was separated by filtration and washed with hexane-diisopropyl ether (3: 1, 60 ml). The mother liquor and the washings were combined and concentrated to dryness to give 3-bromo-2- (4-fluorophenyl) propene (purity 70%, 11.7 g, about 35 mmol) as a pale yellow oil.
Ή- MR (200MHz, CDC13): 54.35 (2H, d, J= 0.6 Hz), 5.47 (1H, brs), 5.50 (1H, brs), 7.06 (2H, t, J= 8.8 Hz), 7.46 (2H, dd, J= 9.0, 5.4 Hz). Ή- MR (200MHz, CDC1 3) : 54.35 (2H, d, J = 0.6 Hz), 5.47 (1H, brs), 5.50 (1H, brs), 7.06 (2H, t, J = 8.8 Hz), 7.46 ( 2H, dd, J = 9.0, 5.4 Hz).
参考例 89 Reference example 89
メタンスルホン酸 2-プロピルペンチル 2-propylpentyl methanesulfonate
2-プロピルペンタン酸 (8.50 g, 57.8丽 ol) と 濃塩酸 (9.6 ml, 0.116 mmol) のメタノール (140 ml) 溶液を室温で 20 時間攪拌した。 酢酸ェチルにより抽出 で精製し、 2-プロピルペンタン酸メチルと酢酸ェチル混合物(2:1, 6.38 g, 31.8 mmol)を無色油状物として得た。 A solution of 2-propylpentanoic acid (8.50 g, 57.8 mol) and concentrated hydrochloric acid (9.6 ml, 0.116 mmol) in methanol (140 ml) was stirred at room temperature for 20 hours. Purification by extraction with ethyl acetate gave a mixture of methyl 2-propylpentanoate and ethyl acetate (2: 1, 6.38 g, 31.8 mmol) as a colorless oil.
Ή-NMR (200MHz, CDC13): (50.89 (6H, t, J= 7.0 Hz), 1.31 (4H, m), 1.45 (2H, m), 1.57 (2H, m), 2.37 (1H, m), 3.67 (3H, s). Ή-NMR (200MHz, CDC1 3 ): (50.89 (6H, t, J = 7.0 Hz), 1.31 (4H, m), 1.45 (2H, m), 1.57 (2H, m), 2.37 (1H, m) , 3.67 (3H, s).
この油状物(6.38 g)を THF (100 ml)に溶解し、 0°Cで攪拌しながら水素化リチ ゥムアルミニウム (純度 80%, 1.51 g, 31.8匪01) の粉末を数回に分けて加え、 0°Cで 2時間攪拌した。 反応液を参考例 31と同様に後処理し、 2-プロピルペン夕 ノール (純度 90%, 4.2 g, 29.0蘭 ol)を無色油状物として得た。 Dissolve the oil (6.38 g) in THF (100 ml) and stir at 0 ° C Pum aluminum (purity 80%, 1.51 g, 31.8 band01) powder was added in several portions and stirred at 0 ° C for 2 hours. The reaction solution was post-treated in the same manner as in Reference Example 31 to give 2-propylphenol (purity 90%, 4.2 g, 29.0 ol) as a colorless oil.
'H-NMR (200MHz, CDC13): <50.91 (6H, t, J= 6.5 Hz), 1.30 (8H, m), 1.49 (1H, m), 3.54 (2H, d, J= 5.2 Hz). 'H-NMR (200MHz, CDC1 3): <50.91 (6H, t, J = 6.5 Hz), 1.30 (8H, m), 1.49 (1H, m), 3.54 (2H, d, J = 5.2 Hz).
この 2-プロピルペンタノ一ル(純度 90%, 4.2g, 29.0腿 ol)をクロ口ホルム (60 ml)に溶解し、 0°Cで攪拌しながらメタンスルホニルクロリド(2.39ml, 30.5匪 ol) を加え、 次いでトリェチルァミン(4.62 ml, 33.4腿 ol)のクロ口ホルム(10 ml) 溶液を 30分かけて滴下し、 反応液を (TCで 3時間攪拌した。 この間反応液にメタ ンスルホニルクロリド (0.69 ml, 8.70 mmol) およびトリェチルァミン(1· 21 ml, 8.70 mmol)を加えた。 反応液を参考例 31 と同様に後処理し、 メタンスルホン酸 2 -プロピルペンチル(5.57 g, 26.7 mmol)を無色油状物として得た。 This 2-propylpentanol (purity 90%, 4.2 g, 29.0 t ol) was dissolved in chloroform (60 ml), and methanesulfonyl chloride (2.39 ml, 30.5 ol) was stirred at 0 ° C while stirring. Then, a solution of triethylamine (4.62 ml, 33.4 tmol) in chloroform (10 ml) was added dropwise over 30 minutes, and the reaction mixture was stirred for 3 hours with TC. During this time, methanesulfonyl chloride ( 0.69 ml, 8.70 mmol) and triethylamine (1.21 ml, 8.70 mmol) were added, and the reaction mixture was worked up in the same manner as in Reference Example 31 to give 2-propylpentyl methanesulfonate (5.57 g, 26.7 mmol) as a colorless product. Obtained as an oil.
Ή-NMR (200MHz, CDC13): 50.91 (6H, m), 1.34 (8H, m), 1.74 (1H, m), 3.00 (3H, s), 4.13 (2H, d, J= 5.0 Hz). Ή-NMR (200MHz, CDC1 3 ): 50.91 (6H, m), 1.34 (8H, m), 1.74 (1H, m), 3.00 (3H, s), 4.13 (2H, d, J = 5.0 Hz).
参考例 90 Reference Example 90
aゥ化 2, 6-ジメチルヘプ夕ン- 4-ィル a ゥ 2,6-Dimethylheptan-4-yl
2, 6-ジメチル _4 -へプタノール(4.02 g, 23.7匪 ol)をクロ口ホルム (60 ml)に 溶解し、 で攪拌しながらメタンスルホニルクロリド (2.22 ml, 28.4腿 ol) を 加え、 次いでトリェチルァミン(4.27 ml, 30.8腿 ol)のクロ口ホルム(10 ml)溶液 を 60分かけて滴下し、 反応液を 0°Cで 20分間攙拌した。反応液を参考例 31と同 様に後処理し、メタンスルホン酸 2, 6-ジメチルヘプタン- 4 -ィル (純度 83%, 6.32 g, 23.7顧 ol)を無色油状物として得た。 Dissolve 2,6-dimethyl_4-heptanol (4.02 g, 23.7 ol) in chloroform (60 ml), add methanesulfonyl chloride (2.22 ml, 28.4 t) with stirring with, and then triethylamine ( A solution of 4.27 ml (30.8 tmol) in black form (10 ml) was added dropwise over 60 minutes, and the reaction solution was stirred at 0 ° C for 20 minutes. The reaction solution was post-treated in the same manner as in Reference Example 31 to obtain 2,6-dimethylheptane-4-yl methanesulfonate (purity 83%, 6.32 g, 23.7 mol) as a colorless oil.
ΐ-NMR (200MHz, CDC13): (50.94 (6H, d, J= 6.2 Hz), 1.45 (2H, m), 1.69 (4H, m), 3.00 (3H, s), 4.87 (1H, d, m). ΐ-NMR (200MHz, CDC1 3 ): (50.94 (6H, d, J = 6.2 Hz), 1.45 (2H, m), 1.69 (4H, m), 3.00 (3H, s), 4.87 (1H, d, m).
得られたメタンスルホン酸 2, 6 -ジメチルヘプタン - 4 -ィル (純度 83 , 6.32 g, 23.7腿 ol)およびヨウ化ナトリウム(7.10 g, 47.4 mmol)の 2-ブ夕ノン(140 ml) 懸濁液を室温で 14時間攪拌した後、 2時間加熱還流した。 酢酸ェチル -へキサン (1:1)により抽出で精製し、 粗製のヨウ化 2, 6-ジメチルヘプタン- 4-ィル(純度 50%, 7.2 g)を黄色油状物として得た。 参考例 91 The obtained 2,6-dimethylheptane-4-yl methanesulfonate (purity 83, 6.32 g, 23.7 tmol) and sodium iodide (7.10 g, 47.4 mmol) were suspended in 2-butane nonone (140 ml). The suspension was stirred at room temperature for 14 hours and then heated to reflux for 2 hours. Purification by extraction with ethyl acetate-hexane (1: 1) gave crude 2,6-dimethylheptane-4-yl iodide (purity 50%, 7.2 g) as a yellow oil. Reference Example 91
メ夕ンスルホン テトラヒドロフラン- 3-メチル Mesulfone tetrahydrofuran-3-methyl
テトラヒドロフラン- 3 -メタノール(2.75 g, 26.7 mmol)およびメタンスルホ二 ルクロリド (3.23 g, 28.0 mmol) のクロ口ホルム(30 ml)溶液に、 0でで攪拌しな がらトリェチルァミン(4.06 ml, 29.3匪 ol)のクロ口ホルム(5 ml)溶液を 60分間 かけて滴下し、 反応液を 0°Cで 60分間攪拌した。 反応液を酢酸ェチル抽出により 精製し、 メタンスルホン酸テトラヒドロフラン- 3-メチル (4.20 g, 23.3 mmol)を 無色油状物として得た。 To a solution of tetrahydrofuran-3-methanol (2.75 g, 26.7 mmol) and methanesulfonyl chloride (3.23 g, 28.0 mmol) in chloroform (30 ml), stir at 0, and triethylamine (4.06 ml, 29.3 bandol). Was added dropwise over 60 minutes, and the reaction solution was stirred at 0 ° C for 60 minutes. The reaction solution was purified by extraction with ethyl acetate to give tetrahydrofuran-3-methyl methanesulfonate (4.20 g, 23.3 mmol) as a colorless oil.
Ή-NMR (200MHz, CDC13): 51.66 (1H, m), 2.10 (1H, m), 2.69 (1H, brquint, J= 6.9 Hz), 3.03 (3H, s), 3.65 (1H, dd, J= 9.0, 5.2 Hz), 3.74 (1H, dd, J- 8.4, 7.4 Hz), 3.84 (1H, dd, J= 9.2, 6.6 Hz), 3.87 (1H, m), 4.14 (1H, dd, J= 9.8, 8.0 Hz), 4.19 (1H, dd, J= 9.8, 7.0 Hz). Ή-NMR (200MHz, CDC1 3 ): 51.66 (1H, m), 2.10 (1H, m), 2.69 (1H, brquint, J = 6.9 Hz), 3.03 (3H, s), 3.65 (1H, dd, J = 9.0, 5.2 Hz), 3.74 (1H, dd, J-8.4, 7.4 Hz), 3.84 (1H, dd, J = 9.2, 6.6 Hz), 3.87 (1H, m), 4.14 (1H, dd, J = 9.8, 8.0 Hz), 4.19 (1H, dd, J = 9.8, 7.0 Hz).
参考例 92 Reference Example 92
メタンスルホン酸 2- (テトラヒドロフラン- 3 -ィル)ェチル Methanesulfonic acid 2- (tetrahydrofuran-3-yl) ethyl
参考例 91で得たメタンスルホン酸テトラヒドロフラン -3-メチル(3.16 g, 17.5 醒 ol)およびシアン化ナトリウム (1.77 g, 35.1腿 ol)の DMSO (15 ml) 懸濁液を 50°C で 14時間攪拌した。 酢酸ェチルによる抽出で得た粗油状物をシリカゲル (40 g)のクロマトグラフィー (酢酸ェチルーへキサン、 3:7-5:5) で精製し、 テト ラヒドロフラン- 3-ァセトニトリルと酢酸ェチルの混合物(2:1, 2.83 g, 14.5 mmol)を無色油状物として得た。 A suspension of tetrahydrofuran-3-methyl methanesulfonate (3.16 g, 17.5 ol) and sodium cyanide (1.77 g, 35.1 ol) in DMSO (15 ml) obtained in Reference Example 91 at 50 ° C. for 14 hours. Stirred. The crude oil obtained by extraction with ethyl acetate was purified by chromatography on silica gel (40 g) (ethyl acetate-hexane, 3: 7-5: 5), and a mixture of tetrahydrofuran-3-acetonitrile and ethyl acetate (2 : 1, 2.83 g, 14.5 mmol) as a colorless oil.
'H-NMR (300MHz, CDC13): (51.73 (1H, brsixtet, J= 7.5 Hz), 2.20 (1H, m), 2.42 (1H, dd, J= 16.7, 7.7 Hz), 2.45 (1H, dd, J= 16.7, 6.6 Hz), 2.61 (1H, brseptet, J= 6.5 Hz), 3.56 (1H, dd, J= 9.0, 5.2 Hz), 3.81 (1H, q, J= 7.0 Hz), 3.92 (2H, m). 'H-NMR (300MHz, CDC1 3): (51.73 (1H, brsixtet, J = 7.5 Hz), 2.20 (1H, m), 2.42 (1H, dd, J = 16.7, 7.7 Hz), 2.45 (1H, dd , J = 16.7, 6.6 Hz), 2.61 (1H, brseptet, J = 6.5 Hz), 3.56 (1H, dd, J = 9.0, 5.2 Hz), 3.81 (1H, q, J = 7.0 Hz), 3.92 (2H , m).
この無色油状物(2.80 g, 14.3匪 ol) を 3N 7_Κ酸化ナトリウム(60 ml)に溶解し、 13 時間加熱還流した。反応液を酸性に調整し、酢酸ェチルで抽出して精製し、テ トラヒドロフラン- 3-酢酸(1.80 g, 13.8 mmol)を淡褐色油状物として得た。 This colorless oil (2.80 g, 14.3 ol) was dissolved in 3N sodium 7-dioxide (60 ml) and heated under reflux for 13 hours. The reaction solution was adjusted to be acidic, extracted and purified with ethyl acetate, and tetrahydrofuran-3-acetic acid (1.80 g, 13.8 mmol) was obtained as a pale brown oil.
¾一 NMR (300MHz, CDC13): (51.60 (1H, m), 2.12 (1H, m), 2.44 (1H, dd, J= 13.0, 1.0 Hz), 2.43 (1H, d, J= 13.0 Hz), 2.65 (1H, brseptet, J= 7.3 Hz), 3.45 (1H, dd, J= 8.8, 6.2 Hz), 3.79 (1H, q, J= 8.4 Hz), 3.90 (2H, m). テトラヒドロフラン - 3 -酢酸(1.80 g, 13.8匪01)と 濃塩酸 (2.3ml, 27.6imol) のエタノール (36 ml)'溶液を室温で 13時間攪拌した。 酢酸ェチルにより抽出で 精製し、 テトラヒドロフラン -3-酢酸ェチル (1.70 g, lO.Onraiol)を淡黄色油状物 として得た。 ¾ one NMR (300MHz, CDC1 3): (51.60 (1H, m), 2.12 (1H, m), 2.44 (1H, dd, J = 13.0, 1.0 Hz), 2.43 (1H, d, J = 13.0 Hz) , 2.65 (1H, brseptet, J = 7.3 Hz), 3.45 (1H, dd, J = 8.8, 6.2 Hz), 3.79 (1H, q, J = 8.4 Hz), 3.90 (2H, m). Tetrahydrofuran-3-acetic acid (1.80 g, 13.8 banded 01) and concentrated hydrochloric acid (2.3ml, 27.6) A solution of imol) in ethanol (36 ml) was stirred at room temperature for 13 hours. Purification by extraction with ethyl acetate gave tetrahydrofuran-3-ethyl acetate (1.70 g, 10 Onraiol) as a pale yellow oil.
'H-NMR (200MHz, CDCI3): 51.26 (3H, t, J= 7.1 Hz), 1.60 (1H, m), 2.13 (1H, m), 2.41 (2H, d, J= 8.0 Hz), 2.63 (1H, brsixtet, J= 7.1 Hz), 3.42 (1H, dd, J= 8.4, 6.6 Hz), 3.76 (1H, dd, J- 8.4, 7.2 Hz), 3.85 (1H, m), 3.95 (1H, dd, J= 8.6, 7.0 Hz), 4.14 (2H, q, J= 7.1 Hz). 'H-NMR (200 MHz, CDCI 3 ): 51.26 (3H, t, J = 7.1 Hz), 1.60 (1H, m), 2.13 (1H, m), 2.41 (2H, d, J = 8.0 Hz), 2.63 (1H, brsixtet, J = 7.1 Hz), 3.42 (1H, dd, J = 8.4, 6.6 Hz), 3.76 (1H, dd, J-8.4, 7.2 Hz), 3.85 (1H, m), 3.95 (1H, dd, J = 8.6, 7.0 Hz), 4.14 (2H, q, J = 7.1 Hz).
このテトラヒドロフラン- 3-酢酸ェチル (1.70 g, 10.0誦 ol)を THF (35 ml)に 溶解し、 0°Cで攪拌しながら水素化リチウムアルミニウム (純度 80¾, 0.47 g, 10.0丽 ol) の粉末を数回に分けて加え、 0°Cで 1時間攪拌した。 反応液を参考例 31と同様に後処理し、 2- (テトラヒドロフラン- 3-ィル) エタノールと酢酸ェチル の混合物(1.43 g, 9.55 mmol)を褐色油状物として得た。 This tetrahydrofuran-3-ethyl acetate (1.70 g, 10.0 ol) was dissolved in THF (35 ml), and lithium aluminum hydride (purity 80¾, 0.47 g, 10.0 olol) was stirred at 0 ° C to obtain a powder. The mixture was added in several portions and stirred at 0 ° C for 1 hour. The reaction solution was post-treated in the same manner as in Reference Example 31 to obtain a mixture of 2- (tetrahydrofuran-3-yl) ethanol and ethyl acetate (1.43 g, 9.55 mmol) as a brown oil.
'H-NMR (200MHz, CDC13): (51.56 (1H, m), 1.68 (2H, q, J= 6.7 Hz), 2.09 (1H, m), 2.31 (1H, sixtet, J= 7.4 Hz), 3.39 (1H, dd, J= 8.4, 7.4 Hz), 3.69 (2H, dd, J= 1.4, 6.6 Hz), 3.74 (1H, brt, J= 7.4 Hz), 3.86 (1H, m), 3.93 (1H, brt, J= 8.2 Hz). 'H-NMR (200MHz, CDC1 3): (51.56 (1H, m), 1.68 (2H, q, J = 6.7 Hz), 2.09 (1H, m), 2.31 (1H, sixtet, J = 7.4 Hz), 3.39 (1H, dd, J = 8.4, 7.4 Hz), 3.69 (2H, dd, J = 1.4, 6.6 Hz), 3.74 (1H, brt, J = 7.4 Hz), 3.86 (1H, m), 3.93 (1H , Brt, J = 8.2 Hz).
この褐色油状物(1.43 g, 9.55 mmol)を THF (20 ml)に溶解し、 Ot:で攪拌しな がらメタンスルホニルクロリド (0.783 ml, 10.0 mmol) を加え、 次いでトリェチ ルァミン(1.52 ml, 11.0 mmol)の THF (5 ml)溶液を 60分かけて滴下し、 反応液 を 0 で 1.5時間攪拌した。反応液を参考例 31と同様に後処理して得た油状物を シリカゲル(10g)のクロマトグラフィー (酢酸ェチレーへキサン、 2:8-5:5) で精 製し、 メタンスルホン酸 2- (テトラヒドロフラン- 3-ィル)ェチル(1.10 g, 5.66 mmol)を無色油状物として得た。 This brown oil (1.43 g, 9.55 mmol) was dissolved in THF (20 ml), methanesulfonyl chloride (0.783 ml, 10.0 mmol) was added while stirring with Ot :, and then triethylamine (1.52 ml, 11.0 mmol). ) In THF (5 ml) was added dropwise over 60 minutes, and the reaction solution was stirred at 0 for 1.5 hours. The reaction solution was post-treated in the same manner as in Reference Example 31 to obtain an oily product, which was purified by chromatography on silica gel (10 g) (ethyl hexane acetate, 2: 8-5: 5) to give methanesulfonic acid 2- ( Tetrahydrofuran-3-yl) ethyl (1.10 g, 5.66 mmol) was obtained as a colorless oil.
Ή-NMR (200MHz, CDC13): 51.57 (1H, m), 1.83 (1H, dd, J= 6.2, 2.8 Hz), 1.88 (1H, dd, J= 6.6, 2.6 Hz), 2.11 (1H, m), 2.33 (1H, brsixtet, J= 7.1 Hz), 3.03 (3H, s), 3.39 (1H, brt, J= 7.7 Hz), 3.77 (1H, brq, J= 7.9 Hz), 3.87 (1H, in), 3.93 (1H, brt, J= 8.0 Hz), 4.26 (2H, t, J= 6.4 Hz). 参考例 93 Ή-NMR (200MHz, CDC1 3 ): 51.57 (1H, m), 1.83 (1H, dd, J = 6.2, 2.8 Hz), 1.88 (1H, dd, J = 6.6, 2.6 Hz), 2.11 (1H, m ), 2.33 (1H, brsixtet, J = 7.1 Hz), 3.03 (3H, s), 3.39 (1H, brt, J = 7.7 Hz), 3.77 (1H, brq, J = 7.9 Hz), 3.87 (1H, in ), 3.93 (1H, brt, J = 8.0 Hz), 4.26 (2H, t, J = 6.4 Hz). Reference 93
メ夕ンスルホン酸(S) -2-フエニルプロピル Maleic sulfonic acid (S) -2-phenylpropyl
(S) - 2 -フエニルプロピオン酸 (1.0 g, 6.66讓 ol)と 濃塩酸 (1.11 ml, 13.3 mmol) のメタノール (20ml) 溶液を室温で 18 時間攪拌した。 酢酸ェチル抽出に より精製し、 (S)-2 -フエニルプロピオン酸メチル (1.20 g, 6.66匪 ol)を無色油 状物として得た。 A solution of (S) -2-phenylpropionic acid (1.0 g, 6.66 benzyl) and concentrated hydrochloric acid (1.11 ml, 13.3 mmol) in methanol (20 ml) was stirred at room temperature for 18 hours. Purification by ethyl acetate extraction gave methyl (S) -2-phenylpropionate (1.20 g, 6.66 ol) as a colorless oil.
Ή-NMR (200MHz, CDC13): δΐ.50 (3Η, d, J= 7.0 Hz), 3.66 (3H, s), 3.73 (1H, q, J= 7.3 Hz), 7.30 (5H, m) . Ή-NMR (200MHz, CDC1 3 ): δΐ.50 (3Η, d, J = 7.0 Hz), 3.66 (3H, s), 3.73 (1H, q, J = 7.3 Hz), 7.30 (5H, m).
この(S)-2-フエニルプロピオン酸メチル (1.20 g, 6.66 mmol)を THF (25 ml) に溶解し、 0°Cで攪拌しながら水素化リチウムアルミニウム (純度 80 , 316 mg, 6.66腿01) の粉末を数回に分けて加え、 0°Cで 80分間攪拌した。 反応液を酢酸ェ チル抽出により精製し、(S)- 2 -フエニルプロパノールと酢酸ェチルの混合物 (3:2, 1.24 g, 6.48腿 ol)を無色油状物として得た。 This methyl (S) -2-phenylpropionate (1.20 g, 6.66 mmol) was dissolved in THF (25 ml), and lithium aluminum hydride (purity 80, 316 mg, 6.66 t ) Was added in several portions, and the mixture was stirred at 0 ° C for 80 minutes. The reaction solution was purified by extraction with ethyl acetate to obtain a mixture of (S) -2-phenylpropanol and ethyl acetate (3: 2, 1.24 g, 6.48 mol) as a colorless oil.
'H-NM (200MHz, CDC13): δ 1.28 (3H, d, J= 7.0 Hz), 2.96 (1H, sixtet, J= 7.3 Hz), 3.71 (2H, brd, J= 7.0 Hz), 7.25 (3H, m), 7.33 (2H, m). 'H-NM (200MHz, CDC1 3): δ 1.28 (3H, d, J = 7.0 Hz), 2.96 (1H, sixtet, J = 7.3 Hz), 3.71 (2H, brd, J = 7.0 Hz), 7.25 ( 3H, m), 7.33 (2H, m).
この(S)- 2-フエニルプロパノール(1.24 g, 6.48讓 ol)をクロ口ホルム (20 ml) に溶解し、 0 で攪拌しながらメタンスルホニルクロリド (0.533 ml, 6.80 mmol) を加え、 次いでトリェチルァミン(1.03 ml, 7.45 mmol)のクロ口ホルム(5 ml)溶 液を 30分かけて滴下し、 反応液を 0 で 80分間攪拌した。 反応液を酢酸ェチル 抽出により精製し、 メタンスルホン酸(S)-2-フエニルプロピル(1.29 g, 6.02 mmol)を無色油状物として得た。 This (S) -2-phenylpropanol (1.24 g, 6.48 alcohol) was dissolved in chloroform (20 ml), methanesulfonyl chloride (0.533 ml, 6.80 mmol) was added while stirring at 0, and then triethylamine was added. A solution of (1.03 ml, 7.45 mmol) in chloroform (5 ml) was added dropwise over 30 minutes, and the reaction solution was stirred at 0 for 80 minutes. The reaction solution was purified by extraction with ethyl acetate to obtain (S) -2-phenylpropyl methanesulfonate (1.29 g, 6.02 mmol) as a colorless oil.
^- MR (200MHz, CDC13): <51.37 (3H, d, J= 7.0 Hz), 2.80 (3H, s), 3.19 (1H, sixtet, J= 7.0 Hz) , 4.25 (1H, dd, J= 9.8, 7.0 Hz), 4.30 (1H, dd, J= 10.0, 7.0 Hz), 7.20-7.33 (5H, m). ^ - MR (200MHz, CDC1 3 ): <51.37 (3H, d, J = 7.0 Hz), 2.80 (3H, s), 3.19 (1H, sixtet, J = 7.0 Hz), 4.25 (1H, dd, J = 9.8, 7.0 Hz), 4.30 (1H, dd, J = 10.0, 7.0 Hz), 7.20-7.33 (5H, m).
参考例 94 Reference Example 94
メタンスルホン酸 (R)_2-フエニルプロピル Methanesulfonic acid (R) _2-phenylpropyl
(R)- 2-フエニルプロピオン酸(1.0 g, 6.66 mmol)から参考例 93の方法に従って (R)- 2-フエニルプロパノール (1.30 g, 6.66 mmoK 酢酸ェチル含有)を無色油状 物として得た。 lH-N R (200MHz, CDC13): 51.28 (3H, d, J= 6.8 Hz), 2.96 (1H, sixtet, ]= 6.8 Hz), 3.71 (2H, d, J= 7.0 Hz), 7.23 (3H, m), 7.32 (2H, d, J= 5.4 Hz). According to the method of Reference Example 93, (R) -2-phenylpropanol (1.30 g, 6.66 mmoK containing ethyl acetate) was obtained as a colorless oil from (R) -2-phenylpropionic acid (1.0 g, 6.66 mmol). . l HN R (200MHz, CDC1 3 ): 51.28 (3H, d, J = 6.8 Hz), 2.96 (1H, sixtet,] = 6.8 Hz), 3.71 (2H, d, J = 7.0 Hz), 7.23 (3H, m), 7.32 (2H, d, J = 5.4 Hz).
この無色油状物から参考例 93 の方法に従ってメタンスルホン酸 (R)- 2 -フエ二 ルプロピル (純度 90%, 1.55 g, 6.51 腿 ol)を無色油状物として得た。 From this colorless oil, methanesulfonic acid (R) -2-phenylpropyl (purity 90%, 1.55 g, 6.51 mol) was obtained as a colorless oil according to the method of Reference Example 93.
¾-NMR (200MHz, CDC13): 51.37 (3H, d, J= 7.2 Hz), 2.81 (3H, s), 3.19 (1H, sixtet, J= 7.0 Hz), 4.25 (1H, dd, J= 9.8, 7.0 Hz), 4.31 (1H, dd, J= 10.0, 7.0 Hz), 7.20-7.39 (5H, m). ¾-NMR (200MHz, CDC1 3 ): 51.37 (3H, d, J = 7.2 Hz), 2.81 (3H, s), 3.19 (1H, sixtet, J = 7.0 Hz), 4.25 (1H, dd, J = 9.8 , 7.0 Hz), 4.31 (1H, dd, J = 10.0, 7.0 Hz), 7.20-7.39 (5H, m).
参考例 95 . Reference example 95.
メ夕ンスルホン酸 2- (4-フルオロフェニル)プロピル 2- (4-fluorophenyl) propyl sulfonic acid
参考例 88で得た 3-ブロモ -2- (4-フルオロフェニル)プロペン (純度 70 , 3.31 g, 10.7 mmol), 酢酸(0.93 ml, 16.2匪 ol)および炭酸カリウム(2· 33 g, 16.2 mmol) のァセトニトリル (20 ml)溶液を室温で 20時間、 ついで 7(T で 6時間攪拌した。 反応液を濃縮後、酢酸ェチル -へキサン(1:1, 40 ml)で希釈した。析出物を除去後、 酢酸ェチル -へキサン( 1, 40 ml)で洗浄した。 母液および洗浄液を濃縮乾固して 酢酸 2_ (4 -フルオロフェニル )—2—プロぺニル (純度 70 2.33 g, 8.4 mmol)を褐色 油状物として得た。 3-bromo-2- (4-fluorophenyl) propene (purity 70, 3.31 g, 10.7 mmol), acetic acid (0.93 ml, 16.2 ol) and potassium carbonate (2.33 g, 16.2 mmol) obtained in Reference Example 88 ) In acetonitrile (20 ml) was stirred at room temperature for 20 hours and then at 7 (6 hours at T). The reaction mixture was concentrated and diluted with ethyl acetate-hexane (1: 1, 40 ml). After removal, the residue was washed with ethyl acetate-hexane (1, 40 ml) and the mother liquor and the washings were concentrated to dryness to give 2_ (4-fluorophenyl) -2-propenyl acetate (purity 70 2.33 g, 8.4 mmol). Was obtained as a brown oil.
この無色油状物 (2.33 g, 8.4 mmol) を酢酸ェチル(75 ml)に溶解し、 10%パラ ジゥム炭素 (417 mg, 52%含水) 存在下、 水素雰囲気下に室温で 1時間攪拌した。 反応液を濾過して触媒を除去し、 濃縮乾固して粗製の酢酸 2- (4-フルオロフェニ ル)プロピル (純度 78%, 2.10 g, 8.4讓 ol)を淡黄色油状物として得た。 This colorless oil (2.33 g, 8.4 mmol) was dissolved in ethyl acetate (75 ml), and the mixture was stirred at room temperature for 1 hour under a hydrogen atmosphere in the presence of 10% palladium carbon (417 mg, containing 52% water). The reaction solution was filtered to remove the catalyst, and concentrated to dryness to obtain crude 2- (4-fluorophenyl) propyl acetate (78% purity, 2.10 g, 8.4 benzyl) as a pale yellow oil.
Ή-NMR (200MHz, CDC13): 51.28 (3H, d, J= 6.8 Hz), 2.01 (3H, s), 3.08 (1H, sixtet, J二 7.0 Hz), 4.10 (1H, dd, J= 10.6, 7.0 Hz), 4.14 (1H, dd, J= 11.0, 7.4 Hz), 7.00 (2H, i, J- 8.8 Hz), 7.18 (2H, dd, J= 8.8, 5.6 Hz). Ή-NMR (200MHz, CDC1 3 ): 51.28 (3H, d, J = 6.8 Hz), 2.01 (3H, s), 3.08 (1H, sixtet, J two 7.0 Hz), 4.10 (1H, dd, J = 10.6 , 7.0 Hz), 4.14 (1H, dd, J = 11.0, 7.4 Hz), 7.00 (2H, i, J- 8.8 Hz), 7.18 (2H, dd, J = 8.8, 5.6 Hz).
この淡黄色油状物 (純度 78 , 2.10 g, 8.411111101)をメタノ一ル(401111)に溶解し、 水酸化ナトリウム(0.71 g, 16.8 腿 ol)と室温で 1.5 時間攪拌した。 反応液を中 和後、 酢酸ェチルで抽出して脱水乾燥、 濃縮乾固して粗製の 2- (4-フルオロフェ ニル)プロパノール(1.98 g)を淡褐色油状物として得た。 This pale yellow oil (purity 78, 2.10 g, 8.411111101) was dissolved in methanol (401111), and the mixture was stirred with sodium hydroxide (0.71 g, 16.8 t) at room temperature for 1.5 hours. The reaction solution was neutralized, extracted with ethyl acetate, dehydrated and dried, and concentrated to dryness to obtain crude 2- (4-fluorophenyl) propanol (1.98 g) as a pale brown oil.
この油状物 α.98 g)をクロ口ホルム(20 ml)に溶解し、 0 で攪拌しながらメタ ンスルホニルクロリド (0.72 ml, 9.22 mmol) を加え、 次いでトリェチルァミン (1.39 ml, 10.1 mmol)のクロ口ホルム(5 ml)溶液を 40分かけて滴下し、 反応液を 0°Cで 60分間攪拌した。 反応液を濃縮して酢酸ェチル抽出して得た油状物をシリ 力ゲル (10 g)のクロマトグラフィー(酢酸ェチル—へキサン、 1:9-2: 8)で精製し、 メタンスルホン酸 2- (4-フルオロフェニル)プロピル(1.33 g, 5.68 mmol)を淡黄 色油状物として得た。 This oily substance α.98 g) was dissolved in chloroform (20 ml), methanesulfonyl chloride (0.72 ml, 9.22 mmol) was added while stirring at 0, and then triethylamine was added. A solution of (1.39 ml, 10.1 mmol) in chloroform (5 ml) was added dropwise over 40 minutes, and the reaction solution was stirred at 0 ° C for 60 minutes. The reaction solution was concentrated and extracted with ethyl acetate. The oil obtained was purified by chromatography on silica gel (10 g) (ethyl acetate-hexane, 1: 9-2: 8) to give methanesulfonic acid 2- (4-Fluorophenyl) propyl (1.33 g, 5.68 mmol) was obtained as a pale yellow oil.
¾-NMR (200MHz, CDC13): 51.35 (3H, d, J= 7.4 Hz), 2.86 (3H, s), 3.18 (1H, sixtet, J= 6.9 Hz), 4.22 (1H, dd, J= 9.6, 7.0 Hz) , 4.26 (1H, dd, J= 10.0, 7.0 Hz), 7.03 (2H, t, J= 8.6 Hz), 7.20 (2H, dd, J= 8.8, 3.0 Hz). ¾-NMR (200MHz, CDC1 3 ): 51.35 (3H, d, J = 7.4 Hz), 2.86 (3H, s), 3.18 (1H, sixtet, J = 6.9 Hz), 4.22 (1H, dd, J = 9.6 , 7.0 Hz), 4.26 (1H, dd, J = 10.0, 7.0 Hz), 7.03 (2H, t, J = 8.6 Hz), 7.20 (2H, dd, J = 8.8, 3.0 Hz).
参考例 96 Reference Example 96
メタンスルホン酸 2 -(2, 3, 4, 5, 6-ペンタメチルフエニル)ェチル Methanesulfonic acid 2- (2,3,4,5,6-pentamethylphenyl) ethyl
塩化 2, 3, 4, 5, 6 -ペンタメチルベンジル(4.01 g, 20.0 mmol)およびシアン化ナ 卜リゥム (2.02 g, 40.0醒 ol)の DMS0 (20 ml) 懸濁液を室温で 3日間攪拌した。 酢酸ェチルによる抽出で得た淡黄色固形物をメタノール中で粉砕し、 2, 3, 4, 5, 6 - ペンタメチルフエ二ルァセトニトリル(1.75 g, 9.36腿 ol)を白色粉末として得た。 'H-NMR (200MHz, CDC13): 52.24 (9H, s), 2.32 (6H, s), 3.70 (2H, s) . A suspension of 2,3,4,5,6-pentamethylbenzyl chloride (4.01 g, 20.0 mmol) and sodium cyanide (2.02 g, 40.0 mmol) in DMS0 (20 ml) was stirred at room temperature for 3 days. did. The pale yellow solid obtained by extraction with ethyl acetate was pulverized in methanol to give 2,3,4,5,6-pentamethylphenylacetonitrile (1.75 g, 9.36 t ol) as a white powder. 'H-NMR (200MHz, CDC1 3): 52.24 (9H, s), 2.32 (6H, s), 3.70 (2H, s).
粗製の 2, 3, 4, 5, 6 -ペンタメチルフエ二ルァセトニトリル (純度 90%, 2.04 g, 9.8 讓 ol)を酢酸(13.6ml)および濃塩酸 (6.8ml)に溶解し、 24時間加熱還流した。 反 応液を冷やして水(30 ml)希釈した。 析出物を濾取し、 水で洗浄、 乾燥して 2, 3, 4, 5, 6-ペンタメチルフエニル酢酸(2.08 g, 10.1腿 ol)を白色粉末として得た。 Ή-NMR (200MHz, CDC13): δ 2.23 (9Η, s), 2.24 (6H, s), 3.80 (2H, s). Dissolve the crude 2,3,4,5,6-pentamethylphenylacetonitrile (purity 90%, 2.04 g, 9.8 benzene) in acetic acid (13.6 ml) and concentrated hydrochloric acid (6.8 ml) and heat to reflux for 24 hours did. The reaction solution was cooled and diluted with water (30 ml). The precipitate was collected by filtration, washed with water, and dried to give 2,3,4,5,6-pentamethylphenylacetic acid (2.08 g, 10.1 liter) as a white powder. Ή-NMR (200MHz, CDC1 3 ): δ 2.23 (9Η, s), 2.24 (6H, s), 3.80 (2H, s).
2, 3, 4, 5, 6 -ペンタメチルフエニル酢酸(1.55 g, 7.49 mmol)から参考例 93のェ ステル化の方法に従って 2, 3, 4, 5, 6-ペンタメチルフエニル酢酸メチル(1.51 g, 6.86腿 ol)を白色粉末として得た。 Methyl 2,3,4,5,6-pentamethylphenylacetate (2,3,4,5,6-pentamethylphenylacetate (1.55 g, 7.49 mmol) according to the esterification method of Reference Example 93 1.51 g, 6.86 t) was obtained as a white powder.
Ή-NMR (200MHz, CDC13): (52.23 (9H, s), 2.24 (6H, s), 3.68 (3H, s), 3.77 (2H, s). Ή-NMR (200MHz, CDC1 3 ): (52.23 (9H, s), 2.24 (6H, s), 3.68 (3H, s), 3.77 (2H, s).
2, 3, 4, 5, 6 -ペン夕メチルフエニル酢酸メチル(1.51 g, 6.86 mmol)から参考例 93の水素化リチウムアルミニウムを用いた還元の方法に従って 2-(2, 3, 4, 5, 6 -ぺ ンタメチルフエニル)エタノール (1.34 g, 6.86 mmol)を白色粉末として得た。 Ή-NMR (200MHz, CDC13): 52.23 (9H, s), 2.29 (6H, s), 3.04 (2H, t, J= 7.7 Hz), 3.75 (2H, t, J= 7.7 Hz). According to the reduction method using lithium aluminum hydride of Reference Example 93 from methyl 2,3,4,5,6-pentanomethylphenylacetate (1.51 g, 6.86 mmol), 2- (2,3,4,5,6 -Phenylmethylphenyl) ethanol (1.34 g, 6.86 mmol) was obtained as a white powder. Ή-NMR (200MHz, CDC1 3 ): 52.23 (9H, s), 2.29 (6H, s), 3.04 (2H, t, J = 7.7 Hz), 3.75 (2H, t, J = 7.7 Hz).
2 -(2, 3,4, 5, 6 -ペン夕メチルフエニル)エタノール(1.34 g, 6.86 mmol)から参考 例 93 のメタンスルホン酸エステル化の方法に従ってメタンスルホン酸 2- (2, 3, 4, 5, 6-ペンタメチルフエニル)ェチル(1.85 g, 6.84 mmol)を白色粉末とし て得た。 . According to the method of methanesulfonic acid esterification of Reference Example 93 from 2- (2,3,4,5,6-pentanoylmethylphenyl) ethanol (1.34 g, 6.86 mmol), methanesulfonic acid 2- (2,3,4, 5,6-Pentamethylphenyl) ethyl (1.85 g, 6.84 mmol) was obtained as a white powder. .
Ή-NMR (200MHz, CDC13): 62.22 (6H, s), 2.23 (3H, s), 2.29 (6H, s), 2.95 (3H, s), 3.23 (2H, brdd, J= 8.4, 8.0 Hz), 4.25 (2H, dd, J= 8.4, 8.2 Hz). Ή-NMR (200MHz, CDC1 3 ): 62.22 (6H, s), 2.23 (3H, s), 2.29 (6H, s), 2.95 (3H, s), 3.23 (2H, brdd, J = 8.4, 8.0 Hz ), 4.25 (2H, dd, J = 8.4, 8.2 Hz).
参考例 97 Reference Example 97
メタンスルホン酸 2- (2, 4, 6-トリメチルフエニル)ェチル 2- (2,4,6-trimethylphenyl) ethyl methanesulfonate
塩化 2, 4, 6 -トリメチルベンジル(3.91 g, 22.0匪 ol)から参考例 96のシァノ化 の方法に従って 2, , 6-トリメチルフエ二ルァセトニトリル(3.69 g, 22.0 mmol) を淡黄色粉末として得た。 According to the method of cyanation in Reference Example 96, 2,2,6-trimethylphenylacetonitrile (3.69 g, 22.0 mmol) was obtained from 2,4,6-trimethylbenzyl chloride (3.91 g, 22.0 ol) as a pale yellow powder. .
lH-NMR (200MHz, CDC13): 52.27 (3H, s), 2.36 (6H, s), 3.70 (2H, s), 6.90 (2H, s). lH-NMR (200MHz, CDC1 3 ): 52.27 (3H, s), 2.36 (6H, s), 3.70 (2H, s), 6.90 (2H, s).
2, 4,6-トリメチルフエ二ルァセトニトリル(2.72 g, 16.2腿01) から参考例 96 の酸加水分解の方法に従って 2, 4, 6-トリメチルフエニル酢酸(2.80 g, 15.7腿 ol) を白色粉末として得た。 2,4,6-Trimethylphenylacetic acid (2.80 g, 15.7 t ol) was obtained from 2,4,6-trimethylphenylacetonitrile (2.72 g, 16.2 t) according to the acid hydrolysis method of Reference Example 96 as a white powder. As obtained.
— NMR (200MHz, CDC13): (32.26 (3H, s), 2.29 (6H, s), 3.68 (2H, s), 6.87 (2H, brs) . - NMR (200MHz, CDC1 3) : (32.26 (3H, s), 2.29 (6H, s), 3.68 (2H, s), 6.87 (2H, brs).
2, 4, 6-トリメチルフエニル酢酸(1.84 g, 10.3腿01) から参考例 96のエステル 化の方法に従って 2, 4, 6 -トリメチルフエニル酢酸メチル(1.96 g, 10.2 mmol)を 無色油状物として得た。 Methyl 2,4,6-trimethylphenylacetate (1.96 g, 10.2 mmol) was obtained from 2,4,6-trimethylphenylacetic acid (1.84 g, 10.3 thigh 01) according to the esterification method of Reference Example 96 as a colorless oil. As obtained.
Ή-NMR (200MHz, CDC13): 52.26 (3H, s), 2.29 (6H, s), 3.66 (2H, s), 3.67 (3H, s), 6.87 (2H, brs). Ή-NMR (200MHz, CDC1 3 ): 52.26 (3H, s), 2.29 (6H, s), 3.66 (2H, s), 3.67 (3H, s), 6.87 (2H, brs).
2, 4, 6-トリメチルフエニル酢酸メチル a.96 g, 10.2腿 ol)から参考例 96の水 素化リチウムアルミニウムを用いた還元の方法に従つて 2- (2, 4, 6-トリメチルフ ェニル)エタノール (純度 90%, 1.83 g, 10.0 mmol) を白色ゲル状物質として得 た。 Methyl 2,4,6-trimethylphenylacetate a.96 g, 10.2 mol) according to the method of reduction using lithium aluminum hydride in Reference Example 96 to give 2- (2,4,6-trimethylphenylacetate). Enyl) ethanol (purity 90%, 1.83 g, 10.0 mmol) was obtained as a white gel.
Ή-NMR (200MHz, CDC13): (52.25 (3H, s), 2.32 (6H, s), 2.93 (2H, t, . J= 7.5 Hz), 3.74 (2H, t, J= 7.5 Hz), 6.85 (2H, brs). Ή-NMR (200MHz, CDC1 3 ):. (52.25 (3H, s), 2.32 (6H, s), 2.93 (2H, t, J = 7.5 Hz), 3.74 (2H, t, J = 7.5 Hz), 6.85 (2H, brs).
2_(2, 4, 6 -トリメチルフエニル)エタノール (純度 90¾, 1.83 g, 10.0 mmol)か ら参考例 96 のメタンスルホン酸エステル化の方法に従ってメタンスルホン酸 2-(2, 4, 6-トリメチルフエニル)ェチル(2.46 g, 9.64 mmol)を淡黄色油状物として 得た。 From 2_ (2,4,6-trimethylphenyl) ethanol (purity 90 1.8, 1.83 g, 10.0 mmol), 2- (2,4,6-trimethyl methanesulfonate was obtained according to the method for methanesulfonate esterification in Reference Example 96. Phenyl) ethyl (2.46 g, 9.64 mmol) was obtained as a pale yellow oil.
Ή-NMR (200MHz, CDC13): 52.25 (3H, s), 2.32 (6H, s), 2.89 (3H, s), 3.11 (2H, t, J= 7.9 Hz), 4.27 (2H, t, J= 7.7 Hz), 6.86 (2H, brs). Ή-NMR (200MHz, CDC1 3 ): 52.25 (3H, s), 2.32 (6H, s), 2.89 (3H, s), 3.11 (2H, t, J = 7.9 Hz), 4.27 (2H, t, J = 7.7 Hz), 6.86 (2H, brs).
参考例 98 Reference 98
ョゥ化 I- (2-テトラヒドロピラニル)ェチル I- (2-tetrahydropyranyl) ethyl ester
臭化 2-テトラヒドロビラニルメチル(2.98 g, 16.3腿01) およびシアン化ナト リウム(2.02 g, 40.0 mmol)の DMSO (15 ml)懸濁液を室温で 2 日間攪拌した。 酢 酸ェチルで抽出して精製し、 2-テトラヒドロピラニルァセトニトリル (2.81 g, 16.3腿 ol, 酢酸ェチル含有) を黄色油状物として得た。 A suspension of 2-tetrahydroviranylmethyl bromide (2.98 g, 16.3 thigh) and sodium cyanide (2.02 g, 40.0 mmol) in DMSO (15 ml) was stirred at room temperature for 2 days. Extraction and purification were performed with ethyl acetate, and 2-tetrahydropyranyl acetonitrile (2.81 g, 16.3 mol, containing ethyl acetate) was obtained as a yellow oil.
Ή-NMR (200MHz, CDC13): 51.47 (1H, brq, J= 12.4 Hz), 1.54 (3H, m), 1.73 (1H, dm, J= 9.6 Hz), 1.89 (1H, m), 2.50 (2H, d, J- 6.2 Hz), 3.47 (1H, tm, J= 10.6 Ή-NMR (200MHz, CDC1 3 ): 51.47 (1H, brq, J = 12.4 Hz), 1.54 (3H, m), 1.73 (1H, dm, J = 9.6 Hz), 1.89 (1H, m), 2.50 ( 2H, d, J- 6.2 Hz), 3.47 (1H, tm, J = 10.6
Hz), 3.56 (1H, m), 4.01 (1H, dm, J= 11.0 Hz). Hz), 3.56 (1H, m), 4.01 (1H, dm, J = 11.0 Hz).
得られた 2-テトラヒドロピラニルァセトニトリル(2.81 g, 16.3讓 ol)を酢酸 The obtained 2-tetrahydropyranyl acetonitrile (2.81 g, 16.3 acetyl) was converted to acetic acid.
(20ml) および濃塩酸 (10ml)に溶解し、 20 時間加熱還流した。 反応液を濃縮乾 固して無色油状物と塩化アンモニゥムの混合物を得た。 これをエタノール(39 ml) に懸濁し、 濃塩酸(2.72 ml, 32.6 腿 ol)とともに室温で 3日間攪拌した。 酢酸ェ チルで抽出して精製し、 2-テトラヒドロピラニル酢酸ェチル (3.3 g, 13.4匪 ol, 酢酸ェチル含有)を黄色油状物として得た。 (20 ml) and concentrated hydrochloric acid (10 ml) and heated under reflux for 20 hours. The reaction solution was concentrated to dryness to obtain a mixture of a colorless oil and ammonium chloride. This was suspended in ethanol (39 ml) and stirred at room temperature for 3 days with concentrated hydrochloric acid (2.72 ml, 32.6 mol). Extraction and purification with ethyl acetate yielded 2-tetrahydropyranyl ethyl acetate (3.3 g, 13.4 mol ol, containing ethyl acetate) as a yellow oil.
'H-NMR (300MHz, CDC13): 51.26 (3H, t, J= 7.1 Hz), 1.45-1.58 (4H, m), 1.64 (1H, dm, J= 11.0 Hz), 1.83 (1H, m) , 2.38 (1H, dd, J= 15.0, 5.3 Hz), 2.50 (1H, dd, J= 15.0, 7.8 Hz), 3.45 (1H, brt, J= 10.8 Hz), 3.74 (1H, m), 3.96 (1H, brd, J= 10.3 Hz), 4.15 (2H, q, J= 7.2 Hz). 'H-NMR (300MHz, CDC1 3): 51.26 (3H, t, J = 7.1 Hz), 1.45-1.58 (4H, m), 1.64 (1H, dm, J = 11.0 Hz), 1.83 (1H, m) , 2.38 (1H, dd, J = 15.0, 5.3 Hz), 2.50 (1H, dd, J = 15.0, 7.8 Hz), 3.45 (1H, brt, J = 10.8 Hz), 3.74 (1H, m), 3.96 ( 1H, brd, J = 10.3 Hz), 4.15 (2H, q, J = 7.2 Hz).
得られた 2 -テトラヒドロピラニル酢酸ェチル(3.3 g, 13.4 mmol)から参考例 93 の水素化リ.チウムアルミニウムを用いた還元の方法に従って粗製の 2- (2-テトラ ヒドロビラニル) エタノール(1.96 g)を褐色油状物として得た。 この油状物から 参考例 91 のメタンスルホン酸エステル化の方法に従って得た褐色油状物をシリ 力ゲル(25 g)のクロマトグラフィー(酢酸ェチルーへキサン、 3:7-4: 6)で精製し、 メタンスルホン酸 2- (2 -テトラヒドロピラニル)ェチル (純度 95%, 2.08 g, 9.44 mmol)を淡黄色油状物として得た。 From the obtained 2-tetrahydropyranyl acetate ethyl (3.3 g, 13.4 mmol), according to the reduction method using lithium aluminum hydride of Reference Example 93, crude 2- (2-tetrahydroviranyl) ethanol (1.96 g) was used. Was obtained as a brown oil. From this oil The brown oil obtained according to the method of methanesulfonic acid esterification of Reference Example 91 was purified by chromatography on silica gel (25 g) (ethyl acetate-hexane, 3: 7-4: 6) to give methanesulfonic acid 2 -(2-Tetrahydropyranyl) ethyl (purity 95%, 2.08 g, 9.44 mmol) was obtained as a pale yellow oil.
Ή-NMR (200MHz, CDC13): 61.30 (1H, brq, J= 10.6 Hz), 1.54 (3H, m), 1.58 (1H, brd, J= 10 Hz), 1.86 (3H, m), 3.01 (3H, s), 3.40 (2H, m), 3.96 (1H, brd, J= 11 Hz), 4.31 (1H, dd, J= 10.0, 6.6 Hz), 4.38 (1H, dd, J= 10.0, 6.6 Hz). 得られたメタンスルホン酸 2- (2-テトラヒドロピラエル)ェチル (純度 95 , 2.08 g, 9.44讓 ol)およびヨウ化ナトリウム(3.00 g, 20.0腿 ol)のアセトン(40 ml) 懸濁液を室温で 2時間加熱還流した。.酢酸ェチルにより抽出で精製し、 粗製のョ ゥ化 2- (2-テトラヒドロピラニル)ェチル(2.60 g, 9.4廳 ol, 酢酸ェチル含有)を 褐色油状物として得た。 Ή-NMR (200MHz, CDC1 3 ): 61.30 (1H, brq, J = 10.6 Hz), 1.54 (3H, m), 1.58 (1H, brd, J = 10 Hz), 1.86 (3H, m), 3.01 ( 3H, s), 3.40 (2H, m), 3.96 (1H, brd, J = 11 Hz), 4.31 (1H, dd, J = 10.0, 6.6 Hz), 4.38 (1H, dd, J = 10.0, 6.6 Hz) A suspension of the obtained 2- (2-tetrahydropyrael) ethyl methanesulfonate (purity: 95, 2.08 g, 9.44 acetyl) and sodium iodide (3.00 g, 20.0 liter) in acetone (40 ml) Was heated to reflux at room temperature for 2 hours. The extract was purified by extraction with ethyl acetate to obtain crude 2- (2-tetrahydropyranyl) ethyl chloride (2.60 g, 9.4 mmol, containing ethyl acetate) as a brown oil.
Ή-NMR (200MHz, CDC13): 1.32 (1H, brq, J= 11 Hz), 1.53 (3H, m), ca. 1.6 (1H, m), 1.84 (3H, m), 1.93 (2H, m), 3.26 (1H, d, J= 7.8 Hz), 3.30 (1H, d, J= 7.6 Hz), 3.38 (2H, m), 3.97 (1H, dm, J= 11.0 Hz). Ή-NMR (200MHz, CDC1 3 ): 1.32 (1H, brq, J = 11 Hz), 1.53 (3H, m), ca. 1.6 (1H, m), 1.84 (3H, m), 1.93 (2H, m ), 3.26 (1H, d, J = 7.8 Hz), 3.30 (1H, d, J = 7.6 Hz), 3.38 (2H, m), 3.97 (1H, dm, J = 11.0 Hz).
参考例 99 Reference 99
3-ブロモ -2-ォキソ酪酸 tert-ブチル Tert-butyl 3-bromo-2-oxobutyrate
クロログリオキシル酸ェチル(13.9 g, 0.10 mol)および tert-ブタノ一ル(10.3 ml, 0.11 mo 1)のクロロホルム(60 ml)溶液に室温で攪拌しながらピリジン(8.90 ml, 0.11 mol)のクロ口ホルム(10 ml)溶液を 40 分かけて滴下し、 混合物を 20時間攪 拌した。 反応液をジイソプロピルェ一テルおよびへキサン混合液(1:1, 300 ml) で希釈後、 不溶物を除去し、 上清液を濃縮してダリオキシル酸 tert-プチルェチ ル (純度 79%, 21.9 g, ジイソプロピルエーテル含有) を無色油状物として得た。 Ή-NMR (200MHz, CDC13): 1.37 (3H, t, J= 7.1 Hz), 1.56 (9H, s), 4.32 (2H, q, J= 7.1 Hz). A solution of ethyl chloroglyoxylate (13.9 g, 0.10 mol) and tert-butanol (10.3 ml, 0.11 mol) in chloroform (60 ml) was stirred at room temperature at room temperature with pyridine (8.90 ml, 0.11 mol). (10 ml) solution was added dropwise over 40 minutes and the mixture was stirred for 20 hours. After diluting the reaction mixture with a mixture of diisopropyl ether and hexane (1: 1, 300 ml), the insolubles were removed, and the supernatant was concentrated and tert-butylethyl dalioxylate (purity 79%, 21.9 g) , Containing diisopropyl ether) as a colorless oil. Ή-NMR (200MHz, CDC1 3 ): 1.37 (3H, t, J = 7.1 Hz), 1.56 (9H, s), 4.32 (2H, q, J = 7.1 Hz).
得られたグリオキシル酸 tert-ブチルェチル(5.15 g, 23.3匪 ol)の THF-ジェチ ルエーテル (1:2, 90 ml)溶液にアルゴン雰囲気下、 臭化工チルマグネシウム(1.0 M THF溶液, 28.0 ml)を - 78 で 60分間かけて滴下した。 反応液を- 78で で 2 時間攪拌後、 - 60 まで 30分間かけて昇温した。 反応液を 1N塩酸 (28 ml) お よび 4.5 塩化アンモニゥム (120 ml)と混合し、 ジイソプロピルエーテル-へキ サン(1:1, 100 ml)で抽出した。 有機層を水および飽和食塩水で洗浄後、 硫酸ナト リウムで乾燥し、 濃縮して粗製の 2-ォキソ酪酸 tert-ブチル (純度 70%, 3.87 g, 17.1 mmol)を淡黄色油状物として得た。 To a solution of the obtained tert-butylethyl glyoxylate (5.15 g, 23.3 bandol) in THF-ethyl ether (1: 2, 90 ml) was added, under an argon atmosphere, tilmagnesium bromide (1.0 M THF solution, 28.0 ml). The solution was dropped at 78 at 60 minutes. After the reaction solution was stirred at −78 for 2 hours, the temperature was raised to −60 over 30 minutes. Dilute the reaction solution with 1N hydrochloric acid (28 ml) And 4.5 ammonium chloride (120 ml) and extracted with diisopropyl ether-hexane (1: 1, 100 ml). The organic layer was washed with water and saturated saline, dried over sodium sulfate, and concentrated to obtain crude tert-butyl 2-oxobutyrate (purity 70%, 3.87 g, 17.1 mmol) as a pale yellow oil. .
Ή-NMR (200MHz, CDC13): άΐ.11 (3Η, t, J= 7.2 Hz), 1.55 (9H, s), 2.81 (2H, q, J= 7.3 Hz). Ή-NMR (200MHz, CDC1 3 ): άΐ.11 (3Η, t, J = 7.2 Hz), 1.55 (9H, s), 2.81 (2H, q, J = 7.3 Hz).
2 -ォキソ酪酸 tert-ブチル(0.98 g, 4.3腿 ol)のクロ口ホルム(16 ml)溶液に、 0°Cで攪拌しながら三臭化トリメチルフエ二ルアンモニゥム α.66 g, 4.3腿 ol) を加え、 反応液を 0°Cで 90分間攪拌した。 反応液を室温まで戻して 40分間攪拌 した。反応液をジイソプロピルエーテルで抽出し、 粗製の tert-ブチル 3-ブロモ -2 -ォキソ酪酸 (純度 50, 0.94 g, 約 2腿 ol)を淡黄色油状物として得た。 To a solution of tert-butyl 2-oxobutyrate (0.98 g, 4.3 t) in chloroform (16 ml) was added, while stirring at 0 ° C, trimethylphenammonium tribromide α.66 g, 4.3 t). The reaction was stirred at 0 ° C. for 90 minutes. The reaction solution was returned to room temperature and stirred for 40 minutes. The reaction solution was extracted with diisopropyl ether to obtain crude tert-butyl 3-bromo-2-oxobutyric acid (purity: 50, 0.94 g, about 2 t ol) as a pale yellow oil.
参考例 100 Reference example 100
ヨウ化 2-(4-クロ口フエニル)プロピル 2- (4-chlorophenyl) propyl iodide
2- (4-クロ口フエニル)プロピオン酸(6.4 g, 33.6醒 ol)から参考例 31のエステ ル化の方法に従って 2_ (4 -クロ口フエニル)プロピオン酸メチル(6.67 g, 33.6 mmol) の淡黄色油状物を得た。 According to the method of esterification in Reference Example 31 from methyl 2- (4-chlorophenyl) propionate (6.4 g, 33.6 mmol), methyl 2- (4-chlorophenyl) propionate (6.67 g, 33.6 mmol) was obtained. A yellow oil was obtained.
Ή-NMR (200MHz, CDC13): 61.49 (3H, d, J= 7.4 Hz), 3.66 (3H, s), 3.70 (1H, q, J- 7.2 Hz), 7.25 (2H, dt, J= 8.8, 2.6 Hz), 7.28 (2H, dt, J= 8.8, 2.4 Hz). Ή-NMR (200MHz, CDC1 3 ): 61.49 (3H, d, J = 7.4 Hz), 3.66 (3H, s), 3.70 (1H, q, J- 7.2 Hz), 7.25 (2H, dt, J = 8.8 , 2.6 Hz), 7.28 (2H, dt, J = 8.8, 2.4 Hz).
2- (4-クロ口フエニル)プロピオン酸メチル(6.67 g, 33.6 mmol)から参考例 31 の水素化リチウムアルミニウムを用いた還元の方法に従って 2- (4-クロ口フエ二 ル)プロパノール(5.64 g, 33.1 腿 ol) の淡渴色油状物を得た。 According to the method of reduction using lithium aluminum hydride of Reference Example 31 from methyl 2- (4-cyclobutenyl) propionate (6.67 g, 33.6 mmol), 2- (4-chlorobutenyl) propanol (5.64 g) , 33.1 thigh).
lH - NMR (200MHz, CDC13): 51.26 (3H, d, J= 6.8Hz), 2.94 (1H, sixtet, J= 6.8 Hz), 3.69 (2H, d, J= 7.0 Hz), 7.18 (2H, dt, J= 8.4, 2.0 Hz), 7.29 (2H, dt, J= 8.8, 2.2 Hz). lH - NMR (200MHz, CDC1 3 ): 51.26 (3H, d, J = 6.8Hz), 2.94 (1H, sixtet, J = 6.8 Hz), 3.69 (2H, d, J = 7.0 Hz), 7.18 (2H, dt, J = 8.4, 2.0 Hz), 7.29 (2H, dt, J = 8.8, 2.2 Hz).
2- (4-クロ口フエニル)プロパノール(5.64 g, 33.1腿 ol) から参考例 31のメタ ンスルホン酸エステル化の方法に従ってメタンスルホン酸 2-(4-クロ口フエ二 ル)プロピル (純度 95%, 8.54 g, 32.6匪 ol)の淡渴色油状物を得た。 According to the method of methanesulfonic acid esterification of Reference Example 31 from 2- (4-chlorophenyl) propanol (5.64 g, 33.1 tmol), 2- (4-chlorophenyl) propyl methanesulfonate (purity 95%) , 8.54 g, 32.6 marl ol).
Ή— NMR (200MHz, CDC13): 1.35 (3H, d, J= 7.2 Hz), 2.86 (3H, s), 3.18 (1H, sixtet, J= 7.0 Hz), 4.22 (1H, dd, J= 9.8, 7.0 Hz), 4.27 (1H, dd, J= 9.8, 6.8 Hz), 7.19 (2H, dt, J= 8.4, 2.2 Hz), 7.30 (2H, dt, J= 8.8, 2.2 Hz). Ή- NMR (200MHz, CDC1 3) : 1.35 (3H, d, J = 7.2 Hz), 2.86 (3H, s), 3.18 (1H, sixtet, J = 7.0 Hz), 4.22 (1H, dd, J = 9.8 , 7.0 Hz), 4.27 (1H, dd, J = 9.8, 6.8 Hz), 7.19 (2H, dt, J = 8.4, 2.2 Hz), 7.30 (2H, dt, J = 8.8, 2.2 Hz).
メタンスルホン酸 2- (4-クロ口フエニル)プロピル(純度 95%, 8.54 g, 32.6 mmol) から参考例 98のョード化の方法に従ってヨウ化 2- (4-クロ口フエニル)プ 口ピル (純度 95, 9.04 g, 30.6 丽 ol)の淡渴色油状物を得た。 From 2- (4-chlorophenyl) propyl methanesulfonate (purity 95%, 8.54 g, 32.6 mmol) according to the method of iodinating in Reference Example 98, 2- (4-chlorophenyl) propyl pill (purity) 95, 9.04 g, 30.6 mol) of a pale oily substance was obtained.
Ή-NMR (200MHz, CDC13): <51.39 (3H, d, J= 6.8 Hz), 3.02 (1H, sixtet, J= 7.0 Hz), 3.30 (1H, dd, J= 9.4, 7.0 Hz), 3.35 (1H, dd, J= 9.4, 7.0 Hz), 7.14 (2H, dt, J= 8.4, 1.8 Hz), 7.29 (2H, dt, J= 8.4, 1.8 Hz). Ή-NMR (200MHz, CDC1 3 ): <51.39 (3H, d, J = 6.8 Hz), 3.02 (1H, sixtet, J = 7.0 Hz), 3.30 (1H, dd, J = 9.4, 7.0 Hz), 3.35 (1H, dd, J = 9.4, 7.0 Hz), 7.14 (2H, dt, J = 8.4, 1.8 Hz), 7.29 (2H, dt, J = 8.4, 1.8 Hz).
参考例 101 Reference Example 101
トリフルォロメ夕ンスルホン酸 2, 2 -ジフルォ口- 2-フエニルェチル 2,2-Difluoro mouth trifluorosulfonic acid 2-phenylethyl
フエ二ルォキザリル酸ェチル (純度 80¾, 7.35 g, 33.0 mmol) 3フッ化ジェチル アミノィォゥ(以下 DASTと略すことがある, 5.91 g, 33.0 mmol) を室温で 80分 間かけて滴下した後、 混合物を 60°Cで 4時間攪拌した。 反応液を氷 (200 g)に 注いで混和し、 酢酸ェチル(250 ml)で抽出した。 有機層を 2%炭酸水素ナトリウム 水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮して粗製 の 2, 2-ジフルォ口- 2-フエニル酢酸ェチル(8.6 g)の黄色油状物を得た。 Ethyl phenyloxalylate (purity: 80 7., 7.35 g, 33.0 mmol) Diethyl trifluoride (hereinafter sometimes abbreviated as DAST, 5.91 g, 33.0 mmol) was added dropwise over 80 minutes at room temperature. The mixture was stirred at ° C for 4 hours. The reaction solution was poured into ice (200 g), mixed, and extracted with ethyl acetate (250 ml). The organic layer was washed with a 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dried over sodium sulfate, and concentrated to give a crude oily substance of crude 2,2-difluoro-2-ethylphenyl acetate (8.6 g) as a yellow oil. I got
得られた粗製の 2, 2 -ジフルオロ- 2_フエニル酢酸ェチル(8.6 g)から参考例 31 の水素化リチウムアルミニウムを用いた還元の方法に従つて得た褐色油状物 (8.7 g)をシリカゲル(80 g)のクロマトグラフィー (酢酸ェチル—へキサン、 3:97-15:85)で精製し、 2,2-ジフルオロ- 2-フエネチルアルコール(純度 78%, 4.94 g, 24.4腿 ol)を油状物として得た。 The brown oily substance (8.7 g) obtained from the obtained crude 2,2-difluoro-2-ethylphenylacetate (8.6 g) according to the reduction method using lithium aluminum hydride of Reference Example 31 was treated with silica gel ( 80 g) chromatography (ethyl acetate-hexane, 3: 97-15: 85), and 2,2-difluoro-2-phenethyl alcohol (purity 78%, 4.94 g, 24.4 tmol) was purified. Obtained as an oil.
Ή-N R (200MHz, CDC13): 61.94 (1H, t, J- 7.0 Hz), 3.98 (2H, dt, J= 7.2, 13.5 Hz), 7.48 (5H, m). Ή-NR (200MHz, CDC1 3 ): 61.94 (1H, t, J- 7.0 Hz), 3.98 (2H, dt, J = 7.2, 13.5 Hz), 7.48 (5H, m).
2, 2-ジフルォ口- 2-フエニルエタノール(0.30 g, 1.90画 ol)およびトリフルォ ロメタンスルホン酸無水物 (0.665 g, 2.28讓01) のクロ口ホルム(5 ml)溶液に、 0°Cで攪拌しながらトリェチルァミン(0.316 ml, 2.28讓 ol)のクロ口ホルム(2 ml) 溶液を 50分間かけて滴下し、 反応液を 0°Cで 30分間攪拌した。 反応液を酢酸ェ チル(30 ml)およびへキサン(12 ml)で希釈し、 氷水および飽和食塩水で洗浄後、 硫酸ナトリゥムで乾燥し、濃縮してトリフルォロメタンスルホン酸 2, 2-ジフルォ ロ- 2-フエニルェチル (0.50 g, 1.73腿 ol)を淡褐色油状物として得た。 ¾-NMR (200MHz, CDC13): δ4.69 (2H, t, J= 12.1 Hz), 7.52 (5H, s). At 0 ° C, a solution of 2,2-difluoro mouth-2-phenylethanol (0.30 g, 1.90 mmol) and trifluoromethanesulfonic anhydride (0.665 g, 2.28 benzene) in chloroform (5 ml) was added at 0 ° C. While stirring, a solution of triethylamine (0.316 ml, 2.28 alcohol) in chloroform (2 ml) was added dropwise over 50 minutes, and the reaction solution was stirred at 0 ° C for 30 minutes. The reaction mixture was diluted with ethyl acetate (30 ml) and hexane (12 ml), washed with ice water and saturated saline, dried over sodium sulfate, and concentrated to give 2,2-difluorotrifluoromethanesulfonic acid. -2-phenylethyl (0.50 g, 1.73 t) was obtained as a pale brown oil. ¾-NMR (200MHz, CDC1 3 ): δ4.69 (2H, t, J = 12.1 Hz), 7.52 (5H, s).
参考例 102 Reference Example 102
2, 2-ジメチルブタノール 2,2-dimethylbutanol
2, 2 -ジメチル酪酸 (純度 90¾, 10.8 g, 83.7 inmol) 濃塩酸 (13.9 ml, 0.167 mol) のメタノ一ル (90ml) 溶液を室温で 22時間攪拌した。反応液に炭酸水素ナ トリウムを加えて中和後、 酢酸ェチル抽出により精製し、 2, 2-ジメチル酸メチル の淡黄色油状物を得た。 A solution of 2,2-dimethylbutyric acid (purity 90 °, 10.8 g, 83.7 inmol) concentrated hydrochloric acid (13.9 ml, 0.167 mol) in methanol (90 ml) was stirred at room temperature for 22 hours. The reaction solution was neutralized by adding sodium hydrogen carbonate, and purified by extraction with ethyl acetate to obtain a pale yellow oil of methyl 2,2-dimethylate.
Ή-NMR (200MHz, CDC13): 50.83 (3H, t, J= 7.3 Hz), 1.16 (6H, s), 1.56 (2H, q, J= 7.3 Hz), 3.66 (3H, s). Ή-NMR (200MHz, CDC1 3 ): 50.83 (3H, t, J = 7.3 Hz), 1.16 (6H, s), 1.56 (2H, q, J = 7.3 Hz), 3.66 (3H, s).
得られた 2, 2 -ジメチル酪酸メチルの THF (200 ml)溶液に 0°Cで攪拌しながら水 素化リチウムアルミニウム (純度 80%, 3.97 g, 83.7雇01) の粉末を数回に分け て加え、 0°Cで 2.時間攪拌した。 ジェチルエーテルにより抽出し、乾燥、 濃縮して 得た油状物を減圧下(95-100 mmHg)蒸留し、 2, 2-ジメチルブタノ一ル(5.70 g, 55.8 mmol)の無色油状物を得た。 The powder of lithium aluminum hydride (purity 80%, 3.97 g, 83.7 hi) was added to the obtained solution of methyl 2,2-dimethylbutyrate in THF (200 ml) with stirring at 0 ° C in several portions. The mixture was stirred at 0 ° C for 2 hours. Extraction with getyl ether, drying and concentration, the resulting oil was distilled under reduced pressure (95-100 mmHg) to give a colorless oil of 2,2-dimethylbutanol (5.70 g, 55.8 mmol).
¾-NMR (200MHz, CDC13): δ 0.84 (3Η, t, J= 7.5' Hz), 0.86 (6H, s), 1.28 (2H, q, J= 7.5 Hz), 3.32 (2H, s). ¾-NMR (200MHz, CDC1 3 ): δ 0.84 (3Η, t, J = 7.5 'Hz), 0.86 (6H, s), 1.28 (2H, q, J = 7.5 Hz), 3.32 (2H, s).
沸点. 77- 80 Boiling point. 77-80
参考例 103 Reference Example 103
2, 2-ジフルォロイソペン夕ノール 2,2-difluoroisopene
3-メチル -2-ォキソ酪酸ェチル(4.15 g, 27.9 mmol) に室温で攪拌しながら DAST(5.00g, 27.9 mmol)を室温で 60分間かけて滴下した後、 混合物を 60°C で 4 時間攪拌した。 反応液を氷 (200 g)に注いで混和し、 酢酸ェチル (250 ml)で抽出し た。 有機層を 5%炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫酸 ナトリウムで乾燥し、 濃縮して粗製の 2,2-ジフルォ口- 3-メチル酪酸ェチル(5.0 g)の黄色油状物を得た。 DAST (5.00 g, 27.9 mmol) was added dropwise to ethyl 3-methyl-2-oxobutyrate (4.15 g, 27.9 mmol) at room temperature with stirring at room temperature over 60 minutes, and the mixture was stirred at 60 ° C for 4 hours. did. The reaction solution was poured into ice (200 g), mixed, and extracted with ethyl acetate (250 ml). The organic layer was washed with 5% aqueous sodium hydrogencarbonate, water and saturated saline, dried over sodium sulfate, and concentrated to give a crude oily substance of crude ethyl 2,2-difluoro-3-methylbutyrate (5.0 g) as a yellow oil. I got
得られた粗製の 2, 2-ジフルォロ- 3-メチル酪酸ェチル(5.0 g)から参考例 31 の 水素化リチウムアルミニウムを用いた還元の方法に従って得た褐色油状物をシリ 力ゲル(35 g)のクロマトグラフィー (ジェチルエーテル一へキサン、 0:10-2:8) で精製し、 2, 2-ジフルォロイソペン夕ノール (純度 20¾, 0.34 g, 0.55顧 ol、 ジ ェチルエーテル含有)を油状物として得た。 The brown oil obtained from the crude 2,2-difluoro-3-methylbutyrate (5.0 g) according to the reduction method using lithium aluminum hydride of Reference Example 31 was converted to a silica gel (35 g). Purified by chromatography (getyl ether-hexane, 0: 10-2: 8), 2,2-difluoroisopentanol (purity 20 純度, 0.34 g, 0.55 ml) Ethyl ether) as an oil.
'H-NMR (200MHz, CDC13): (51.05 (6H, d, J= 6.8 Hz), 2.25 (1H, septet, J= 7.1 Hz), 3.80 (2H, dt, J= 7.0, 13.4 Hz). 'H-NMR (200MHz, CDC1 3): (51.05 (6H, d, J = 6.8 Hz), 2.25 (1H, septet, J = 7.1 Hz), 3.80 (2H, dt, J = 7.0, 13.4 Hz).
参考例 104 Reference Example 104
トリフルォロメタンスルホン酸 2, 2-ジフルォロイソペンチル 2,2-difluoroisopentyl trifluoromethanesulfonate
参考例 103 と同様にして得た 2,2-ジフルォロイソペンタノール (純度 80%, 1.51 g, 9.73 腿 ol)およびトリフルォロメ夕ンスルホン酸無水物(2.22 ml, 13.3 删 ol)のクロ口ホルム(25 ml)溶液に 0。Cで攪拌しながらトリェチルァミン(2.00 ml, 14.5腿 ol)のクロ口ホルム(5 ml)溶液を 30分間かけて滴下し、 反応液を 0°C で 45分間攪拌した。反応液をジイソプロピルエーテル(160 ml)で希釈し氷水およ び飽和食塩水で洗浄し、 乾燥、 濃縮した。 残渣をシリカゲル (20 g)のクロマトグ ラフィ一(酢酸ェチルーへキサン、 1:19-2:18)で精製して濃縮乾固してトリフル ォロメタンスルホン酸 2, 2-ジフルォロイソペンチル (純度 60¾, 1.24g, 3.4 mmol) を黄色粉末として得た。 Clonal form of 2,2-difluoroisopentanol (purity 80%, 1.51 g, 9.73 tmol) and trifluoromethanesulfonic anhydride (2.22 ml, 13.3 l) obtained in the same manner as in Reference Example 103. (25 ml) 0 in solution. While stirring with C, a solution of triethylamine (2.00 ml, 14.5 tmol) in chloroform (5 ml) was added dropwise over 30 minutes, and the reaction solution was stirred at 0 ° C for 45 minutes. The reaction solution was diluted with diisopropyl ether (160 ml), washed with ice water and saturated saline, dried and concentrated. The residue was purified by chromatography on silica gel (20 g) (ethyl acetate-hexane, 1: 19-2: 18), concentrated to dryness, and 2,2-difluoroisopentyl trifluoromethanesulfonate (purity 60¾, 1.24 g, 3.4 mmol) as a yellow powder.
'H-NMR (300MHz, CDC13): 61.09 (6H, d, J= 6.9 Hz), 2.25 (1H, t of septet, J= 14.4, 7.2 Hz), 4.57 (2H, t, J= 11.7 Hz). 'H-NMR (300MHz, CDC1 3): 61.09 (6H, d, J = 6.9 Hz), 2.25 (1H, t of septet, J = 14.4, 7.2 Hz), 4.57 (2H, t, J = 11.7 Hz) .
実施例 1 - 1 Example 1-1
式 (VI) において R 5が n -ペンチル基、 R6がメチル基、 R10がー ZR15 (Z が S02、 R15が p -トルィル基)、 Rがェチル基である化合物の合成 R 5 is n in formula (VI) - pentyl group, R 6 is a methyl group, R 10 gar ZR 15 (Z is S0 2, R 15 is p - Toruiru group) Synthesis of Compound R is Echiru group
参考例 3-2で合成した化合物 (20.0 g, 46.5 mmol) を THF (200 ml) および DMF (100 ml) に溶解し、 3-ブロモ -2-ォクタノン (17.6 ml, 純度 51%, 51.2 mmol) およびトリェチルァミン (19.4ml, 0.140 mol) を加えて室温で 2時間撹捽した。 反応液を濃縮乾固後、 残渣を酢酸ェチル (700 ml) で希釈し、 水、 3%炭酸水素 ナトリウム水溶液、 水、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウ ムで乾燥した。溶媒を減圧下に溜去し、残渣をシリカゲル 60 (200g、 キ一ゼ ルゲル 60、 70〜230メッシュ、 Art. 7734、 エー'メルク社製、 ドイツ) のカラムクロマトグラフィーに付し、 酢酸ェチル:へキサン (30 : 70から 4 0: 60)の溶出画分を減圧下濃縮乾固し、標題化合物の淡黄色粉末(19.6g, 35.3 mmol) を四種異性体の混合物として得た。 ¾-NMR (300 MHz, 重クロ口ホルム中) : δ 0.89/0.90 (3Η, t, J= 6.5 Hz), 1.27 (3H, t, J= 7.1 Hz), 1.27 (6H, m), 1.57 (1H, m), 1.71/1.80 (3H, s), 2.10 (1H, m), 2.42 (3H, s), 2.69 (2H, t, J= 7.5 Hz) , 3.25 (1H, m), 3.26/3.27 (2H, t, J= 7.5 Hz), 4.16 (2H, q, J= 7.1 Hz), 5.57/5.73 (1H, brs), 7.30 (2H, d, J= 8.2 Hz), 7.87/7.89 (2H, d, J= 8.2 Hz), 9.72 (1H, brs). , 元素分析値: C24H33N306S3 · 0.4H20 として計算値: C, 51.21; H, 6.05; N, 7.46; S, 17.09 (%), 実測値; C, 51.20; H, 5.90; N, 6.92; S, 16.99 (%) . The compound synthesized in Reference Example 3-2 (20.0 g, 46.5 mmol) was dissolved in THF (200 ml) and DMF (100 ml), and 3-bromo-2-octanone (17.6 ml, purity 51%, 51.2 mmol) was dissolved. And triethylamine (19.4 ml, 0.140 mol) were added, and the mixture was stirred at room temperature for 2 hours. After the reaction mixture was concentrated to dryness, the residue was diluted with ethyl acetate (700 ml), washed with water, 3% aqueous sodium hydrogen carbonate, water, 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. . The solvent was distilled off under reduced pressure, and the residue was subjected to column chromatography on silica gel 60 (200 g, Kieselgel 60, 70-230 mesh, Art. 7734, manufactured by A Merck, Germany) to give ethyl acetate: The eluted fraction of hexane (30:70 to 40:60) was concentrated to dryness under reduced pressure to obtain a pale yellow powder (19.6 g, 35.3 mmol) of the title compound as a mixture of four isomers. ¾-NMR (300 MHz, in double-mouthed form): δ 0.89 / 0.90 (3Η, t, J = 6.5 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.27 (6H, m), 1.57 ( 1H, m), 1.71 / 1.80 (3H, s), 2.10 (1H, m), 2.42 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.25 (1H, m), 3.26 / 3.27 (2H, t, J = 7.5 Hz), 4.16 (2H, q, J = 7.1 Hz), 5.57 / 5.73 (1H, brs), 7.30 (2H, d, J = 8.2 Hz), 7.87 / 7.89 (2H, . d, J = 8.2 Hz) , 9.72 (1H, brs), elemental analysis: C 24 H 33 N 3 0 6 S 3 · 0.4H 2 0 calculated: C, 51.21; H, 6.05 ; N, 7.46 S, 17.09 (%), found; C, 51.20; H, 5.90; N, 6.92; S, 16.99 (%).
実施例 1 - 2 Example 1-2
式 (V I) において R 5が n-へキシル基、 R6が水素原子、 R1。が— ZR15 (Z が S〇2、 R 15が p-トルィル基)、 Rがェチル基である化合物の合成 In the formula (VI), R 5 is an n-hexyl group, R 6 is a hydrogen atom, R 1 . Synthesis of compounds with ZR 15 (Z is S〇 2 , R 15 is p-tolyl group) and R is ethyl group
参考例 3-2で合成した化合物 (0.49 g, 1.14讓 ol) を THF (8 ml) および DMF (4 ml) に溶解し、 2 -ブロモォクタナール (0.21 ml, 純度 94%, 1.14腿 ol) お よびトリェチルァミン (0.47ml, 3.42imol) を加えて室温で 90分間撹拌した。 反応液を濃縮後、 残渣を酢酸ェチル (35 ml) で希釈し、 0.1N塩酸、 水および飽 和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去し、 残渣を シリカゲル 60 (10g) のカラムクロマトグラフィーに付し、 酢酸ェチル:へキ サン (30 : 70から 40 : 60) の溶出画分を減圧下濃縮乾固し、 標題化合物 の淡黄色粉末 (0.48 , 0.86腿 ol) を四種異性体の混合物として得た。 The compound synthesized in Reference Example 3-2 (0.49 g, 1.14 alcohol) was dissolved in THF (8 ml) and DMF (4 ml), and 2-bromooctanal (0.21 ml, purity 94%, 1.14 t ol) was dissolved. ) And triethylamine (0.47 ml, 3.42 imol) were added, and the mixture was stirred at room temperature for 90 minutes. After concentrating the reaction solution, the residue was diluted with ethyl acetate (35 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, and the residue was subjected to column chromatography on silica gel 60 (10 g). The fraction eluted with ethyl acetate: hexane (30:70 to 40:60) was concentrated to dryness under reduced pressure. A pale yellow powder (0.48, 0.86 tmol) of the title compound was obtained as a mixture of four isomers.
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 0.87/0.88 (3H, t, J= 6.8 Hz), 1.25/1.26 (3H, t, J= 7.2 Hz), 1.28 (4H, brs), 1.32 (2H, m), 1.45 (1H, m), 1.48 (2H, brquint, J= 5.7 Hz), 1.74/1.85 (1H, m), 2.41 (3H, s), 2.69 (2H, t, J= 7.5 Hz), 3.23 (1H, m), 3.25/3.27 (2H, t, J= 7.5 Hz), 4.15 (2H, q, J= 7.2 Hz), 4.55/4.78 (1H, m), 5.80/5.86 (1H, d/brs, J= 1.9 Hz), 7.28/7.29 (2H, d, J= 7.9 Hz), 7.88/7.89 (2H, d, J= 7.9 Hz), 10.03 (1H, brs). 'H-NMR (300 MHz, in double-mouthed form): δ 0.87 / 0.88 (3H, t, J = 6.8 Hz), 1.25 / 1.26 (3H, t, J = 7.2 Hz), 1.28 (4H, brs) , 1.32 (2H, m), 1.45 (1H, m), 1.48 (2H, brquint, J = 5.7 Hz), 1.74 / 1.85 (1H, m), 2.41 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.23 (1H, m), 3.25 / 3.27 (2H, t, J = 7.5 Hz), 4.15 (2H, q, J = 7.2 Hz), 4.55 / 4.78 (1H, m), 5.80 / 5.86 (1H, d / brs, J = 1.9 Hz), 7.28 / 7.29 (2H, d, J = 7.9 Hz), 7.88 / 7.89 (2H, d, J = 7.9 Hz), 10.03 (1H, brs).
元素分析値: C24H33N306S3 として計算値: C, 51.87; H, 5.99; , 7.56; S, 17.31 (%), 実測値; C, 51.58; H, 6.03; N, 7.59; S, 17.48 (%) . Elemental analysis: C 24 H 33 N 3 0 6 S 3 Calculated: C, 51.87; H, 5.99 ;, 7.56; S, 17.31 (%), Found; C, 51.58; H, 6.03 ; N, 7.59 S, 17.48 (%).
実施例 ί - 3 Example ί-3
式 (V I) において R 5がエトキシカルポニル基、 R 6がメチル基、 R1(^S—ZR 15 (Zが S02、 R15が P-トルィル基)、 Rがェチル基である化合物の合成 参考例 3-2で合成した化合物 (0.51 g, 1.19 mmol) を THF (8 ml) および DMF (4 ml) に溶解し、 2-クロロアセト酢酸ェチル (0.18 ml, 1.31 匪 ol) およびト リエチルァミン (0.49 ml, 3.56 mmol) を加えて室温で 2時間撹拌した。 反応液 を濃縮後、 残渣を酢酸ェチル (35 ml) で希釈し、 0.1N塩酸、 水および飽和食塩 水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去し、 残渣をシリカ ゲル 60 (10g)のカラムクロマトグラフィーに付し、酢酸ェチル:へ サン(4 0 : 60から 50 : 50) の溶出画分を減圧下濃縮乾固し、 標題化合物の淡黄色 粉末 (0.52g, 0.93腿 ol) を四種異性体の混合物として得た。 Formula (VI) in R 5 is ethoxy Cal Poni Le group, R 6 is a methyl group, the synthesis of R 1 (^ S-ZR 15 (Z is S0 2, R 15 is P- Toruiru group), and R is an Echiru group The compound synthesized in Reference Example 3-2 (0.51 g, 1.19 mmol) was dissolved in THF (8 ml) and DMF (4 ml), and ethyl 2-chloroacetoacetate (0.18 ml, 1.31 ol) and triethylamine (0.49 g) were dissolved. ml, 3.56 mmol) and stirred at room temperature for 2 hours. After concentrating the reaction solution, the residue was diluted with ethyl acetate (35 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, and the residue was subjected to column chromatography on silica gel 60 (10 g). The fraction eluted with ethyl acetate: hexane (40:60 to 50:50) was concentrated to dryness under reduced pressure. Then, a pale yellow powder (0.52 g, 0.93 mol) of the title compound was obtained as a mixture of four isomers.
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 1.25 (6Η, t, J= 7.2 Hz), 1.81/2.01 (3H, s), 2.42 (3H, s), 2.64 (2H, m), 3.21 (2H, m), 3.79/3.92 (1H, s), 4.15 (2H, q, J= 7.2 Hz), 4.19 (2H, q, J= 7.2 Hz), 6.02/4.98 (1H, br), 7.31 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.77 (1H, brs). Ή-NMR (300 MHz, in double-mouthed form): δ 1.25 (6Η, t, J = 7.2 Hz), 1.81 / 2.01 (3H, s), 2.42 (3H, s), 2.64 (2H, m), 3.21 (2H, m), 3.79 / 3.92 (1H, s), 4.15 (2H, q, J = 7.2 Hz), 4.19 (2H, q, J = 7.2 Hz), 6.02 / 4.98 (1H, br), 7.31 (2H, d, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.77 (1H, brs).
元素分析値: C22H27N308S3 として計算値: C, 47.38; H, 4.88; N, 7.53; S, 17.25 (%), 実測値; C, 47.18; H, 5.08; N, 7.55; S, 17.28 (%) . Elemental analysis: Calculated as C 22 H 27 N 3 0 8 S 3: C, 47.38; H, 4.88; N, 7.53; S, 17.25 (%), Found; C, 47.18; H, 5.08 ; N, 7.55; S, 17.28 (%).
実施例卜 4 Example 4
式 (VI) において R 5が n-ブチル基、 R6がメチル基、 R1Qがー ZR15 が S02、 R15が p-トルィル基)、 Rがェチル基である化合物の合成 R 5 is n- butyl group in the formula (VI), the synthesis of R 6 is a methyl group, R 1Q gar ZR 15 is S0 2, R 15 is p- Toruiru group), and R is an Echiru group
参考例 3-2で合成した化合物 (697 mg, 1.62匪 ol) を THF (12 ml) および DMF (8 ml) に溶解し、 3 -ブロモ- 2_ヘプ夕ノン (1.01 ml, 純度 77%, 1.64 mmol) およびトリェチルァミン(0.675 ml, 4.87醒 ol)を加えて室温で 2時間撹拌した。 反応液を実施例卜 1と同様に処理し、標題化合物の淡黄色粉末(0.65 g, 1.20 mmol) を四種異性体の混合物として得た。 The compound synthesized in Reference Example 3-2 (697 mg, 1.62 bandol) was dissolved in THF (12 ml) and DMF (8 ml), and 3-bromo-2-hepnonone (1.01 ml, purity 77%, 1.64 mmol) and triethylamine (0.675 ml, 4.87 mmol) were added, and the mixture was stirred at room temperature for 2 hours. The reaction solution was treated in the same manner as in Example 1, to give a pale yellow powder of the title compound (0.65 g, 1.20 mmol) as a mixture of four isomers.
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 0.93/0.91 (3Η, t, J= 6.9 Hz), 1.27 (3H, t, J= 7.1 Hz), 1.32 (2H, m), 1.36 (2H, m), 1.55 (1H, m), 1.72/1.80 (3H, s), 2.10 (1H, m), 2.41 (3H, s), 2.69 (2H, t, J= 7.4 Hz), 3.24 (1H, m), 3.27 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.1 Hz), 5.61/5.77 (1H, br), 7.29 (2H, brd, J= 8.3 Hz), 7.87/7.89 (2H, d, J= 8.3 Hz), 9.83 (1H, br). , 元素分析値: C23H31N306S3 · 0.2H20 として計算値: C, 50.66; H, 5.80; N, 7.71; S, 17.64 (%), 実測値; C, 50.60; H, 5.59; N, 7.72; S, 17.88 (%) . 以下に実施例 1_1〜1-4で合成した化合物および同様にして合成した化合物の 一覧を示す。 Ή-NMR (300 MHz, in double-hole form): δ 0.93 / 0.91 (3Η, t, J = 6.9 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.32 (2H, m), 1.36 ( 2H, m), 1.55 (1H, m), 1.72 / 1.80 (3H, s), 2.10 (1H, m), 2.41 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.24 (1H , m), 3.27 (2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.1 Hz), 5.61 / 5.77 (1H, br), 7.29 (2H, brd, J = 8.3 Hz), 7.87 /7.89 (2H, d, J = 8.3 Hz), 9.83 (1H, br)., Elemental analysis: C 23 H 31 N 3 0 6 S 3 · 0.2H 2 0 Calculated: C, 50.66; H, 5.80; N, 7.71; S, 17.64 (%), found; C, 50.60; H, 5.59; N, 7.72; S, 17.88 (%). The following is a list of compounds synthesized in Examples 1_1 to 1-4 and compounds synthesized in the same manner.
〔表 2〕(Table 2)
差替え用紙 則 〔表 3〕Replacement paper rules (Table 3)
差螯ぇ用紙(規則 26) 〔表 4〕Difference paper (Rule 26) (Table 4)
差替え用紙(規則 ) 〔表 5〕Replacement form (rules) (Table 5)
羞替え用涎(規則 26) 〔表 6〕Swallowing saliva (Rule 26) (Table 6)
差替え用紙 (m ) 以下に実施例 1- 5〜1- 52で合成した化合物のデータを示す。 Replacement paper (m) The data of the compounds synthesized in Examples 1-5 to 1-52 are shown below.
実施例 1-5 Example 1-5
Ή-NMR (300MHz, CDC13) : δ 1.25 (3Η, t, J= 7.2 Hz), 2.44 (3H, s), 2.68 (2H, t, J= 7.2 Hz), 3.17 (1H, d, J= 13.2 Hz), 3.25 (2H, dt, J= 5.8, 7.2 Hz), 3.43 (1H, d, J= 13.2 Hz), 4.14 (2H, q, J= 7.2 Hz), 5.61 (1H, s), 7.28 (2H, d, J= 8.3 Hz), 7.28 (2H, m), 7.40 (3H, m), 7.90 (2H, d, J= 8.3 Hz), 9.70 (1H, brs). 元素分析値: C24H25N306S3として計算値: C, 52.63; H, 4.60; N, 7.67; S, 17.56 Ή-NMR (300MHz, CDC1 3 ): δ 1.25 (3Η, t, J = 7.2 Hz), 2.44 (3H, s), 2.68 (2H, t, J = 7.2 Hz), 3.17 (1H, d, J = 13.2 Hz), 3.25 (2H, dt, J = 5.8, 7.2 Hz), 3.43 (1H, d, J = 13.2 Hz), 4.14 (2H, q, J = 7.2 Hz), 5.61 (1H, s), 7.28 (2H, d, J = 8.3 Hz), 7.28 (2H, m), 7.40 (3H, m), 7.90 (2H, d, J = 8.3 Hz), 9.70 (1H, brs). Elemental analysis: C 24 H 25 N 3 0 6 S 3 calculated: C, 52.63; H, 4.60 ; N, 7.67; S, 17.56
(%), 実測値: C, 52.39; H, 4.61; N, 7.74; S, 17.59 (%) . (%), Found: C, 52.39; H, 4.61; N, 7.74; S, 17.59 (%).
実施例 1 - 6 Example 1-6
Ή-NMR (300MHz, CDC13) : δ 2.44 (3Η, s), 2.69 (2H, t, J= 7.2 Hz), 3.17 (1H, d, J= 13.2 Hz), 3.25 (2H, dt, J= 5.7, 7.2 Hz), 3.42 (1H, d, J= 13.2 Hz), 3.68 (3H, s), 5.61 (1H, s), 7.33 (2H, d, J= 8.3 Hz), 7.33 (2H, m), 7.40 (3H, m), 7.91 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 2.44 (3Η, s), 2.69 (2H, t, J = 7.2 Hz), 3.17 (1H, d, J = 13.2 Hz), 3.25 (2H, dt, J = 5.7, 7.2 Hz), 3.42 (1H, d, J = 13.2 Hz), 3.68 (3H, s), 5.61 (1H, s), 7.33 (2H, d, J = 8.3 Hz), 7.33 (2H, m) , 7.40 (3H, m), 7.91 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C23H23N306S3として計算値: C, 51.77; H, 4.34; N, 7.87; S, 18.03Elemental analysis: C 23 H 23 N 3 0 6 S 3 Calculated: C, 51.77; H, 4.34 ; N, 7.87; S, 18.03
(%)、 実測値: C, 51.86; H, 4.54; N, 7.95; S, 18.08 (%) . (%), Found: C, 51.86; H, 4.54; N, 7.95; S, 18.08 (%).
実施例 1-7 ' 'H-NMR (300MHz, CDC13) : δ 1.24 (3Η, t, J= 7.1 Hz), 2.44 (3H, s), 2.69 (2H, t, J= 7.4 Hz) , 3.20 (1H, d, J= 13.2 Hz), 3.26 (2H, dt, J= 5.4, 7.4 Hz), 3.44 (1H, d, J= 13.2 Hz), 4.14 (2H, q, J= 7.1 Hz), 5.66 (1H, s), 7.33 (2H, d, J= 8.2 Hz), 7.36-44 (5H, m), 7.58 (2H, d, J- 8.9 Hz), 7.61 (2H, d, J= 8.9 Hz), 7.91 (2H, d, J= 8.2 Hz), 9.74 (1H, brs). Example 1-7 '' H-NMR (300MHz , CDC1 3): δ 1.24 (3Η, t, J = 7.1 Hz), 2.44 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.20 (1H, d, J = 13.2 Hz), 3.26 (2H, dt, J = 5.4, 7.4 Hz), 3.44 (1H, d, J = 13.2 Hz), 4.14 (2H, q, J = 7.1 Hz), 5.66 (1H, s), 7.33 (2H, d, J = 8.2 Hz), 7.36-44 (5H, m), 7.58 (2H, d, J- 8.9 Hz), 7.61 (2H, d, J = 8.9 Hz) , 7.91 (2H, d, J = 8.2 Hz), 9.74 (1H, brs).
元素分析値: C30H29N306S3として計算値: C, 57.77; H, 4.69; N, 6.74; S, 15.42 (%) .実測値: C, 57.49; H, 4.80; N, 6.74; S, 15.44 ( ) . Elemental analysis: C 30 H 29 N 3 0 6 S 3 Calculated:. C, 57.77; H, 4.69; N, 6.74; S, 15.42 (%) Found: C, 57.49; H, 4.80 ; N, 6.74; S, 15.44 ().
実施例 1 - 8 Example 1-8
4種異性体混合物 4-isomer mixture
Ή-NMR (300 MHz, 重クロ口ホルム中) : 51.24 (3H, t, J= 7.1 Hz), 2.45 (3H, s), 2.67/2.71 (2H, t, J= 7.3 Hz) , 3.26 (2H, m), 4.14 (2H, q, J- 7.1 Hz) , 4.36/4.72 (1H, s), 5.93/6.08 (1H, s), 6.77/6.93 (2H, brd, J= 7.5 Hz), 7.12-7.40 (10H, m), 7.92 (2H, brd, J= 8.1 Hz), 9.67/9.72 (1H, brs). 元素分析値: C30H29N306S3 として計算値: C, 57.77; H, 4.69; N, 6.74; S, 15.42Ή-NMR (300 MHz, in double-mouthed form): 51.24 (3H, t, J = 7.1 Hz), 2.45 (3H, s), 2.67 / 2.71 (2H, t, J = 7.3 Hz), 3.26 (2H , m), 4.14 (2H, q, J- 7.1 Hz), 4.36 / 4.72 (1H, s), 5.93 / 6.08 (1H, s), 6.77 / 6.93 (2H, brd, J = 7.5 Hz), 7.12- 7.40 (10H, m), 7.92 (2H, brd, J = 8.1 Hz), 9.67 / 9.72 (1H, brs). Elemental analysis: C 30 H 29 N 3 0 6 S 3 Calculated: C, 57.77; H, 4.69 ; N, 6.74; S, 15.42
(%)、 実測値; C, 57.52; H, 4.80; N, 6.78; S, 15.43 (%) . (%), Found; C, 57.52; H, 4.80; N, 6.78; S, 15.43 (%).
実施例 1-9 Example 1-9
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή-NMR (300MHz, CDC13) : δ 0.88 (3Η, brt, J= 7 Hz), 1.27 (3H, t, J= 7.1 Hz), 1.29 (9H, m), 1.32 (6H, m), 1.56 (1H, i), 1.71/1.80 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J= 7.2 Hz), 3.25 (1H, m), 3.26/3.27 (2H, t, J= 7.6 Hz), 4.17 (2H, q, J= 7.1 Hz), 5.57/5.73 (1H, brs), 7.30/7.31 (2H, d, J= 8.2 Hz), 7.87/7.89 (2H, d, J= 8.2 Hz), 9.70 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3Η, brt, J = 7 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.29 (9H, m), 1.32 (6H, m), 1.56 (1H, i), 1.71 / 1.80 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J = 7.2 Hz), 3.25 (1H, m), 3.26 / 3.27 (2H, t, J = 7.6 Hz), 4.17 (2H, q, J = 7.1 Hz), 5.57 / 5.73 (1H, brs), 7.30 / 7.31 (2H, d, J = 8.2 Hz), 7.87 / 7.89 (2H, d, J = 8.2 Hz), 9.70 (1H, brs).
元素分析値: C26H37N306S3*H20として計算値: C, 51.89; H, 6.53; , 6.98; S, 15.98 (%), 実測値: C, 51.93; H, 6.31; N, 6.94; S, 16.21 {%) . Elemental analysis: C 26 H 37 N 3 0 6 S 3 * H 2 0 Calculated: C, 51.89; H, 6.53;, 6.98; S, 15.98 (%), Found: C, 51.93; H, 6.31 ; N, 6.94; S, 16.21 {%).
実施例 1-10 Example 1-10
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή -腿 (300MHz, CDC13) : δ 0.86/0.87 (3Η, brt, J= 7 Hz), 1.24/1.26 (3H, t, J= 7.1 Hz), 1.24 (8H, br), 1.28 (2H, ) , 1.41 (1H, m), 2.17 (1H, m), 2.43 (3H, s), 2.65/2.71 (2H, t, J= 7.5 Hz), 3.17/3.53 (1H, dd, J= 12.1/11.3, 2.3, 2.6 Hz), 3.29 (2H, m), 4.13/4.16 (2H, q, J= 7.1 Hz), 5.59/6.11 (1H, brs), 7.26 (1H, m), 7.34 (6H, m), 7.89/7.92 (2H, d, J= 8.3 Hz), 9.75 (1H, brs). E - thigh (300MHz, CDC1 3): δ 0.86 / 0.87 (3Η, brt, J = 7 Hz), 1.24 / 1.26 (3H, t, J = 7.1 Hz), 1.24 (8H, br), 1.28 (2H, ), 1.41 (1H, m), 2.17 (1H, m), 2.43 (3H, s), 2.65 / 2.71 (2H, t, J = 7.5 Hz), 3.17 / 3.53 (1H, dd, J = 12.1 / 11.3 , 2.3, 2.6 Hz), 3.29 (2H, m), 4.13 / 4.16 (2H, q, J = 7.1 Hz), 5.59 / 6.11 (1H, brs), 7.26 (1H, m), 7.34 (6H, m) , 7.89 / 7.92 (2H, d, J = 8.3 Hz), 9.75 (1H, brs).
元素分析値: C31H39N306S3として計算値: C, 57.65; H, 6.09; N, 6.51; S, 14.89 (%) 、 実測値: C, 57.41; H, 6.00; N, 6.57; S, 15.08 (%) . Elemental analysis: C 31 H 39 N 3 0 6 S 3 Calculated: C, 57.65; H, 6.09 ; N, 6.51; S, 14.89 (%), Found: C, 57.41; H, 6.00 ; N, 6.57; S, 15.08 (%).
実施例ト 11 Example 11
Ή-NMR (300MHz, CDC13) : δ 1.26 (3Η, t, J= 7.2 Hz), 1.28 (3H, t, J= 7.1 Hz), 2.42 (3H, s), 2.70 (2H, t, J= 7.5 Hz), 3.11 (1H, d, J= 13.2 Hz), 3.21 (2H, s), 3.27 (2H, t, J= 7.5 Hz), 3.53 (1H, d, J= 13.2 Hz), 4.16 (2H, q, J= 7.2 Hz), 4.20 (2H, dq, J= 1.5, 7.2 Hz), 5.96 (1H, br), 7.30 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3Η, t, J = 7.2 Hz), 1.28 (3H, t, J = 7.1 Hz), 2.42 (3H, s), 2.70 (2H, t, J = 7.5 Hz), 3.11 (1H, d, J = 13.2 Hz), 3.21 (2H, s), 3.27 (2H, t, J = 7.5 Hz), 3.53 (1H, d, J = 13.2 Hz), 4.16 (2H , q, J = 7.2 Hz), 4.20 (2H, dq, J = 1.5, 7.2 Hz), 5.96 (1H, br), 7.30 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C22H27N308S3として計算値: C, 47.38; H, 4.88; N, 7.53; S, 17.25Elemental analysis: C 22 H 27 N 3 0 8 S 3 Calculated: C, 47.38; H, 4.88 ; N, 7.53; S, 17.25
(%)、 実測値: C, 47.14; H, 5.01; , 7.48; S, 17.11 (%) . (%), Found: C, 47.14; H, 5.01;, 7.48; S, 17.11 (%).
実施例卜 12 異性体混合物 主/副生成物 Example 12 Isomer mixture Main / By-product
'H-NMR (300MHz, CDC13) : δ 0.86/0.87 (3Η, brt, J= 6.8/6.6 Hz), 1.26/1.24 (3H, t, J= 7.2 Hz), 1.27 (4H, brs), 1.30 (2H, i), 1.40 (IH, m), 2.17 (1H, m), 2.43 (3H, s), 2.65/2.71 (2H, t, J= 7.3/7.2 Hz), 3.17/3.53 (1H, dd, J= 10.9/11.3, 2.6/3.0 Hz), 3.29/3.22 (2H, q/m, J- 6.9 Hz), 4.13/4.16 (2H, q, J= 7.2 Hz), 5.59/6.10 (IH, s), 7.27 (IH, m), 7.29-7.39 (4H, m), 7.35/7.34 (2H, d, J= 7.9 Hz), 7.89/7.92 (2H, d, J= 7.9 Hz), 9.74 (IH, br). 'H-NMR (300MHz, CDC1 3): δ 0.86 / 0.87 (3Η, brt, J = 6.8 / 6.6 Hz), 1.26 / 1.24 (3H, t, J = 7.2 Hz), 1.27 (4H, brs), 1.30 (2H, i), 1.40 (IH, m), 2.17 (1H, m), 2.43 (3H, s), 2.65 / 2.71 (2H, t, J = 7.3 / 7.2 Hz), 3.17 / 3.53 (1H, dd , J = 10.9 / 11.3, 2.6 / 3.0 Hz), 3.29 / 3.22 (2H, q / m, J- 6.9 Hz), 4.13 / 4.16 (2H, q, J = 7.2 Hz), 5.59 / 6.10 (IH, s ), 7.27 (IH, m), 7.29-7.39 (4H, m), 7.35 / 7.34 (2H, d, J = 7.9 Hz), 7.89 / 7.92 (2H, d, J = 7.9 Hz), 9.74 (IH, m br).
元素分析値: C29H35N306S3として計算値: C, 56.38; H, 5.71; , 6.80; S, 15.57 (%), 実測値: C, 56.12; H, 5.85; N, 6.81; S, 15.51 (%) . Elemental analysis: Calculated as C 29 H 35 N 3 0 6 S 3: C, 56.38; H, 5.71;, 6.80; S, 15.57 (%), Found: C, 56.12; H, 5.85 ; N, 6.81 ; S, 15.51 (%).
実施例 1-13 Example 1-13
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή-NMR (300MHz, CDC13) : <5 1.24 (3H, t, J= 7.2 Hz), 2.13/2.15 (3H, s), 2.42 (3H, s), 2.66 (2H, t, J= 7.5 Hz), 3.11 (IH, d, J= 13.2 Hz), 3.23 (2H, dt, J= 2.6, 7.5 Hz), 3.32 (IH, d, J= 13.2 Hz), 4.13/4.12 (2H, q, J= 7.2 Hz), 5.86 (1H, brs), 7.21 (2H, d, J= 8.3 Hz), 7.30 (2H, d, J= 8.3 Hz) , 7.49 (2H, d, J= 8.3 Hz), 7.89 (2H, d, J= 8.3 Hz), 8.00 (IH, brs), 9.86 (IH, brs). Ή-NMR (300MHz, CDC1 3 ): <5 1.24 (3H, t, J = 7.2 Hz), 2.13 / 2.15 (3H, s), 2.42 (3H, s), 2.66 (2H, t, J = 7.5 Hz ), 3.11 (IH, d, J = 13.2 Hz), 3.23 (2H, dt, J = 2.6, 7.5 Hz), 3.32 (IH, d, J = 13.2 Hz), 4.13 / 4.12 (2H, q, J = 7.2 Hz), 5.86 (1H, brs), 7.21 (2H, d, J = 8.3 Hz), 7.30 (2H, d, J = 8.3 Hz), 7.49 (2H, d, J = 8.3 Hz), 7.89 (2H , d, J = 8.3 Hz), 8.00 (IH, brs), 9.86 (IH, brs).
元素分析値: C26H28N407S3として計算値: C, 51.64; H, 4.67; N, 9.26; S, 15.91Elemental analysis: C 26 H 28 N 4 0 7 S 3 Calculated: C, 51.64; H, 4.67 ; N, 9.26; S, 15.91
(%) 、 実測値: C, 51.23; H, 4.72; N, 9.33; S, 15.68 (%) . (%), Found: C, 51.23; H, 4.72; N, 9.33; S, 15.68 (%).
実施例 1 - 14 Examples 1-14
異性体混合物 Isomer mixture
Ή-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J= 6.8 Hz), 0.91 (3H, t, J= 6.8 Hz), 1.26/1.27 (3H, t, J- 7.2 Hz), 1.28-1.43 (8H, m), 1.55 (2H, m), 2.00 (2H, m), 2.24/1.89 (2H, m), 2.42 (3H, s), 2.69 (2H, t, J= 7.5 Hz), 3.08/3.27 (2H, dd/m, J= 10.1, 2.3 Hz), 3.26 (2H, dt, J= 6.4, 7.5 Hz), 4.16/4.17 (2H, q, J= 7.2 Hz), 5.80/6.07 (IH, brs/br), 7.31/7.30 (2H, d, J= 8.3 Hz), 7.89/7.88 (2H, d, J= 8.3 Hz), 9.69 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3Η, t, J = 6.8 Hz), 0.91 (3H, t, J = 6.8 Hz), 1.26 / 1.27 (3H, t, J- 7.2 Hz), 1.28 -1.43 (8H, m), 1.55 (2H, m), 2.00 (2H, m), 2.24 / 1.89 (2H, m), 2.42 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.08 / 3.27 (2H, dd / m, J = 10.1, 2.3 Hz), 3.26 (2H, dt, J = 6.4, 7.5 Hz), 4.16 / 4.17 (2H, q, J = 7.2 Hz), 5.80 / 6.07 ( IH, brs / br), 7.31 / 7.30 (2H, d, J = 8.3 Hz), 7.89 / 7.88 (2H, d, J = 8.3 Hz), 9.69 (1H, brs).
実施例 1-15 Example 1-15
1:1 異性体混合物 1: 1 isomer mixture
Ή-NMR (300MHz, CDC13) : 6 0.87/0.88 (3H, t, J= 6.7 Hz), 1.25 (3H, t, J- 7.2 Hz), 1.25 (12H, brs), 1.48 (2H, m), 2.41 (3H, s), 2.69 (2H, t, J= 7.5 Hz), 3.23 (IH, m), 3.27/3.25 (2H, t, J= 7.5 Hz), 4.15 (2H, q, J= 7.2 Hz), 4.6/4.8 (IH, br), 5.80/5.87 (IH, d, J= 2.6/1.1 Hz), 7.29/7.28 (2H, d, J= 8.3 Hz), 7.89/7.88 (2H, d, J= 8.3 Hz), 10.05 (IH, brs). Ή-NMR (300MHz, CDC1 3 ): 6 0.87 / 0.88 (3H, t, J = 6.7 Hz), 1.25 (3H, t, J- 7.2 Hz), 1.25 (12H, brs), 1.48 (2H, m), 2.41 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.23 (IH, m), 3.27 / 3.25 (2H, t , J = 7.5 Hz), 4.15 (2H, q, J = 7.2 Hz), 4.6 / 4.8 (IH, br), 5.80 / 5.87 (IH, d, J = 2.6 / 1.1 Hz), 7.29 / 7.28 (2H, d, J = 8.3 Hz), 7.89 / 7.88 (2H, d, J = 8.3 Hz), 10.05 (IH, brs).
元素分析値: C26H37N306S3 · 0.5H20として計算値: C, 52.68; H, 6.46; N, 7.09; S, 16.22 ( ), 実測値: C, 52.63; H, 6.31; , 7.12; S, 16.56 (%) . Elemental analysis: C 26 H 37 N 3 0 6 S 3 · 0.5 H 2 0 Calculated: C, 52.68; H, 6.46; N, 7.09; S, 16.22 (), Found: C, 52.63; H, 6.31;, 7.12; S, 16.56 (%).
実施例 1-16 Example 1-16
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή-NMR (300MHz, CDC13) : δ 1.23/1.24 (3Η, t, J= 7.2 Hz), 2.41 (3H, s), 2.69 (2H, t, J- 7.5 Hz), 3.28 (2H, t, J= 7.5 Hz), 4.13 (2H, q, J= 7.2 Hz), 4.38/4.54 (IH, d, J= 1.1/2.6 Hz), 4.93/4.57 (IH, br), 6.05 (IH, brs/brd, J= 2.6 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.30 (IH, m) , 7.37 (2H, m), 7.50/7.15 (2H, m), 7.87/7.88 (2H, d, J= 8.3 Hz), 9.95 (IH, br). Ή-NMR (300MHz, CDC1 3 ): δ 1.23 / 1.24 (3Η, t, J = 7.2 Hz), 2.41 (3H, s), 2.69 (2H, t, J- 7.5 Hz), 3.28 (2H, t, J = 7.5 Hz), 4.13 (2H, q, J = 7.2 Hz), 4.38 / 4.54 (IH, d, J = 1.1 / 2.6 Hz), 4.93 / 4.57 (IH, br), 6.05 (IH, brs / brd , J = 2.6 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.30 (IH, m), 7.37 (2H, m), 7.50 / 7.15 (2H, m), 7.87 / 7.88 (2H, d, J = 8.3 Hz), 9.95 (IH, br).
元素分析値: C24H25N306S3 · 0·3Η2Οとして計算値: C, 52.12; Η, 4.67; Ν, 7.60; S, 17.39 (%) 、 実測値: C, 52.17; Η, 4.77; Ν, 7.66; S, 17.54 (%) . Elemental analysis: C 24 H 25 N 3 0 6 S 3 · 0 3Η 2計算 Calculated: C, 52.12; Η, 4.67; Ν, 7.60; S, 17.39 (%), found: C, 52.17; Η, 4.77; Ν, 7.66; S, 17.54 (%).
実施例 1-17 Example 1-17
^-NMR (300MHz, CDC13) : δ 1.26 (3Η, t, J= 7.2 Hz) , 1.32 (3H, t, J= 7.1 Hz), 2.43 (3H, s), 2.71 (2H, dd, J= 7.5, 7.2 Hz), 3.08 (IH, d, J= 12.8 Hz), 3.28 (2H, dt, J= 3.0, 7.5 Hz), 3.31 (IH, d, J= 12.8 Hz), 4.16 (2H, q, J= 7.2 Hz), 4.38 (2H, dq, J= 1.1, 7.2 Hz), 5.24 (1H, br), 7.31 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.73 (IH, brs). ^ -NMR (300MHz, CDC1 3) : δ 1.26 (3Η, t, J = 7.2 Hz), 1.32 (3H, t, J = 7.1 Hz), 2.43 (3H, s), 2.71 (2H, dd, J = 7.5, 7.2 Hz), 3.08 (IH, d, J = 12.8 Hz), 3.28 (2H, dt, J = 3.0, 7.5 Hz), 3.31 (IH, d, J = 12.8 Hz), 4.16 (2H, q, J = 7.2 Hz), 4.38 (2H, dq, J = 1.1, 7.2 Hz), 5.24 (1H, br), 7.31 (2H, d, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz) , 9.73 (IH, brs).
元素分析値: C21H25N308S3として計算値: C, 46.40; H, 4.64; , 7.73; S, 17.70Elemental analysis: C 21 H 25 N 3 0 8 S 3 Calculated: C, 46.40; H, 4.64;, 7.73; S, 17.70
(%)、 実測値: C, 46.49; H, 4.63; N, 7.77; S, 17.74 (%) . (%), Found: C, 46.49; H, 4.63; N, 7.77; S, 17.74 (%).
実施例卜 18 Example 18
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή-NMR (300MHz, CDC13) : δ 0.89/0.88 (3Η, t, J= 6.8 Hz), 1.26 (11H, m), 1.50 (IH, in), 1.60/1.67 (3H, s), 2.41 (3H, s), 3.17/2.90 (1H, brdd, J= 9.9/10.2, 2.4/2.5 Hz), 4.16/4.15 (IH, d, J= 12.8/13.2 Hz) , 4.26/4.25 (IH, d, J= 12.8/13.2 Hz), 5.49/5.69 (IH, br), 7.28 (7H, m), 7.86/7.88 (2H, d, J- 8.3 Hz), 9.73 (IH, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 / 0.88 (3Η, t, J = 6.8 Hz), 1.26 (11H, m), 1.50 (IH, in), 1.60 / 1.67 (3H, s), 2.41 ( 3H, s), 3.17 / 2.90 (1H, brdd, J = 9.9 / 10.2, 2.4 / 2.5 Hz), 4.16 / 4.15 (IH, d, J = 12.8 / 13.2 Hz), 4.26 / 4.25 (IH, d, J = 12.8 / 13.2 Hz), 5.49 / 5.69 (IH, br), 7.28 (7H, m), 7.86 / 7.88 (2H, d, J-8.3 Hz), 9.73 (IH, br).
元素分析値: C28H25N304S3 · 0.5H20として計算値: C, 57.70; H, 6.23; N, 7.21; S, 16.50 (%), 実測値: C, 57.78; H, 6.26; N, 7.37; S, 16.60 (%) . Elemental analysis: C 28 H 25 N 3 0 4 S 3 · 0.5H 2 0 Calculated: C, 57.70; H, 6.23 ; N, 7.21; S, 16.50 (%), Found: C, 57.78; H , 6.26; N, 7.37; S, 16.60 (%).
実施例 1-19 Example 1-19
異性体混合物 主/副生成物 Isomer mixture Main / By-product
¾-NMR (300MHz, CDC13) : δ 0.87/0.88 (3Η, brt, J= 6.8 Hz), 1.25 (1 H, m), 1.45 (9H, s), 1.72 (IH, m), 1.84 (IH, m), 2.40/2.41 (3H, s), 3.19/3.28 (IH, brddd/m, J= 7.9, 6.8, 1.2 Hz), 3.67 (IH, d, J= 15.8 Hz), 3.72 (IH, dd, J= 15.8, 2.3 Hz), 4.30/4.53 (IH, brd/br, J= 5.3 Hz), 5.78/5.84 (IH, brd, J= 1.9/3.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.88/7.90 (2H, d, J= 8.3 Hz), 9.96 (IH, br). 元素分析値: C27H3gN306S3 · 0.5H20として計算値: C, 53.44; H, 6.48; N, 6.92; S, 15.85 (%) 、 実測値: C, 53.40; H, 6.41; , 7.07; S, 16.58 (%) . ¾-NMR (300MHz, CDC1 3 ): δ 0.87 / 0.88 (3Η, brt, J = 6.8 Hz), 1.25 (1 H, m), 1.45 (9H, s), 1.72 (IH, m), 1.84 (IH , m), 2.40 / 2.41 (3H, s), 3.19 / 3.28 (IH, brddd / m, J = 7.9, 6.8, 1.2 Hz), 3.67 (IH, d, J = 15.8 Hz), 3.72 (IH, dd , J = 15.8, 2.3 Hz), 4.30 / 4.53 (IH, brd / br, J = 5.3 Hz), 5.78 / 5.84 (IH, brd, J = 1.9 / 3.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.88 / 7.90 (2H , d, J = 8.3 Hz), 9.96 (IH, br) elemental analysis:. C 27 H 3g N 3 0 6 S 3 · 0.5H 2 0 calculated: C, 53.44 H, 6.48; N, 6.92; S, 15.85 (%), found: C, 53.40; H, 6.41 ;, 7.07; S, 16.58 (%).
実施例 1-20 Example 1-20
1:1 異性体混合物 1: 1 isomer mixture
¾-NMR (300MHz, CDC13) : δ 0.88/0.91 (3H, t, J= 7.2/7.3 Hz), 1.25/1.25 (3H, t, J= 7.2 Hz), 1.25 (2H, m), 1.34 (2H, m), 1.47 (IH, m), 1.75/1.86 (IH, m), 2.41 (3H, s), 2.69 (2H, t, J= 7.2 Hz), 3.23 (1H, m), 3.27 (2H, m), 4.14 (2H, q, J= 7.2 Hz), 4.67/4.91 (IH, m), 5.81/5.87 (IH, brs/brd, J= 4.2 Hz), 7.27/7.29 (2H, d, J= 8.3 Hz), 7.87/7.89 (2H, d, J= 8.3 Hz), 10.05 (1H, br). 実施例 1-21 ¾-NMR (300MHz, CDC1 3 ): δ 0.88 / 0.91 (3H, t, J = 7.2 / 7.3 Hz), 1.25 / 1.25 (3H, t, J = 7.2 Hz), 1.25 (2H, m), 1.34 ( 2H, m), 1.47 (IH, m), 1.75 / 1.86 (IH, m), 2.41 (3H, s), 2.69 (2H, t, J = 7.2 Hz), 3.23 (1H, m), 3.27 (2H , m), 4.14 (2H, q, J = 7.2 Hz), 4.67 / 4.91 (IH, m), 5.81 / 5.87 (IH, brs / brd, J = 4.2 Hz), 7.27 / 7.29 (2H, d, J = 8.3 Hz), 7.87 / 7.89 (2H, d, J = 8.3 Hz), 10.05 (1H, br).
1:1 異性体混合物 1: 1 isomer mixture
LH-NMR (300MHz, CDC13) ; δ 0.88 (3H, t, J= 6.9 Hz), 1.25 (3H, t, J= 7.1 Hz), 1.25 (20H, brs), 1.48 (IH, m), 1.74/1.85 (IH, m), 2.41/2.41 (3H, s), 2.69 (2H, t, J= 7.3 Hz), 3.23 (IH, m), 3.27 (2H, m), 4.15 (2H, q, J= 7.1 Hz), 4. 9/4.74 (IH, brd/brs, J= 5.0 Hz), 5.80/5.86 (IH, brs/brd, J= 3.7 Hz), 7.28/7.29 (2H, d, J- 8.0 Hz), 7.87/7.89 (2H, d, J= 8.0 Hz), 10.02 (1H, br). 実施例 1-22 L H-NMR (300MHz, CDC1 3); δ 0.88 (3H, t, J = 6.9 Hz), 1.25 (3H, t, J = 7.1 Hz), 1.25 (20H, brs), 1.48 (IH, m), 1.74 / 1.85 (IH, m), 2.41 / 2.41 (3H, s), 2.69 (2H, t, J = 7.3 Hz), 3.23 (IH, m), 3.27 (2H, m), 4.15 (2H, q, J = 7.1 Hz), 4.9 / 4.74 (IH, brd / brs, J = 5.0 Hz), 5.80 / 5.86 (IH, brs / brd, J = 3.7 Hz), 7.28 / 7.29 (2H, d, J- 8.0 Hz), 7.87 / 7.89 (2H, d, J = 8.0 Hz), 10.02 (1H, br).
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή—NMR (300MHz, CDC13) ; δ 1.27 (3Η, t, J= 7.1 Hz), 1.72 (IH, m), 1.79/1.81 (3H, s), 2.42 (3H, s), 2.46 (1H, m), 2.56 (2H, i), 2.68 (2H, t, J= 7.3 Hz), 3.11 (1H, dd, J= 10.8, 3.3 Hz), 3.25 (2H, t, J= 7.3 Hz), 3.70/3.68 (3H, s), 4.16 (2H, q, J= 7.1 Hz), 5.42/5.83 (1H, br), 7.30 (2H, d, J= 8.2 Hz), 7.86/7.88 (2H, d, J= 8.2 Hz), 9.74 (1H, br). Ή-NMR (300MHz, CDC1 3 ); δ 1.27 (3Η, t, J = 7.1 Hz), 1.72 (IH, m), 1.79 / 1.81 (3H, s), 2.42 (3H, s), 2.46 (1H, m), 2.56 (2H, i), 2.68 (2H, t, J = 7.3 Hz), 3.11 (1H, dd, J = 10.8, 3.3 Hz), 3.25 (2H, t, J = 7.3 Hz), 3.70 / 3.68 (3H, s), 4.16 (2H, q, J = 7.1 Hz), 5.42 / 5.83 (1H, br), 7.30 (2H, d , J = 8.2 Hz), 7.86 / 7.88 (2H, d, J = 8.2 Hz), 9.74 (1H, br).
元素分析値: C23H29N308S3として計算値: C, 48.32; H, 5.11; N, 7.35; S, 16.83 (%) 、 実測値: C, 48.13; H, 4.94; N, 7.40; S, 17.06 (%) . Elemental analysis: C 23 H 29 N 3 0 8 S 3 Calculated: C, 48.32; H, 5.11 ; N, 7.35; S, 16.83 (%), Found: C, 48.13; H, 4.94 ; N, 7.40; S, 17.06 (%).
実施例 1-23 Example 1-23
異性体混合物 Isomer mixture
'H-NMR (300MHz, CDC13) ; δ 1.26 (3Η, t, J= 7.1 Hz), 1.88 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J= 7.4 Hz), 3.03 (1H, d, J= 13.3 Hz), 3.16 (1H, d, J= 13.3'H-NMR (300MHz, CDC1 3); δ 1.26 (3Η, t, J = 7.1 Hz), 1.88 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.03 (1H, d, J = 13.3 Hz), 3.16 (1H, d, J = 13.3
Hz), 3.26 (2H, t, J- 7.4 Hz), 4.16 (2H, q, J= 7.1 Hz), 5.32 (1H, br), 7.30Hz), 3.26 (2H, t, J- 7.4 Hz), 4.16 (2H, q, J = 7.1 Hz), 5.32 (1H, br), 7.30
(2H, d, J= 8.2 Hz), 7.87 (2H, d, J= 8.2 Hz), 9.76 (1H, br). (2H, d, J = 8.2 Hz), 7.87 (2H, d, J = 8.2 Hz), 9.76 (1H, br).
元素分析値: C19H23N306S3として計算値: C, 46.99; H, 4.77; N, 8.65; S, 19.81 (%)、 実測値: C, 46.71; H, 4.80; N, 8.61; S, 19.88 (%) . Elemental analysis: C 19 H 23 N 3 0 6 S 3 Calculated: C, 46.99; H, 4.77 ; N, 8.65; S, 19.81 (%), Found: C, 46.71; H, 4.80 ; N, 8.61; S, 19.88 (%).
実施例 1-24 Example 1-24
異性体混合物 主/副生成物 Isomer mixture Main / By-product
¾- MR (300MHz, CDC13) ; δ 1.26 (3Η, t, J= 7.1 Hz), 1.42 (1H, m), 1.72 (1H, m), 1.72/1.80 (3H, s), 1.93 (1H, m), 2.15 (1H, m), 2.36 (2H, m), 2.42 (3H, s), 2.68 (2H, t, J= 7.3 Hz), 3.27 (2H, dt, J= 1.2, 7.3 Hz), 3.68/3.66 (3H, s), 4.16 (2H, q, J= 7.0 Hz), 5.59/5.80 (1H, br), 7.30 (2H, brd, J= 8.2 Hz), 7.87/7.88 (2H, d, J= 8.2 Hz), 9.79 (1H, br). ¾- MR (300MHz, CDC1 3) ; δ 1.26 (3Η, t, J = 7.1 Hz), 1.42 (1H, m), 1.72 (1H, m), 1.72 / 1.80 (3H, s), 1.93 (1H, m), 2.15 (1H, m), 2.36 (2H, m), 2.42 (3H, s), 2.68 (2H, t, J = 7.3 Hz), 3.27 (2H, dt, J = 1.2, 7.3 Hz), 3.68 / 3.66 (3H, s), 4.16 (2H, q, J = 7.0 Hz), 5.59 / 5.80 (1H, br), 7.30 (2H, brd, J = 8.2 Hz), 7.87 / 7.88 (2H, d, J = 8.2 Hz), 9.79 (1H, br).
元素分析値: C24H31N308S3 · 0.6H20として計算値: C, 48.32; H, 5.44; N, 7.04; S, 16.12 (%), 実測値: C, 48.00; H, 5.01; N, 7.00; S, 16.71 (%) . Elemental analysis: C 24 H 31 N 3 0 8 S 3 · 0.6 H 2 0 Calculated: C, 48.32; H, 5.44; N, 7.04; S, 16.12 (%), found: C, 48.00; H , 5.01; N, 7.00; S, 16.71 (%).
実施例卜 25 Example 25
異性体混合物のうちの 1異性体のデータ Data for one isomer of the isomer mixture
Ή-NMR (300MHz, CDC13) : δ 1.25 (3Η, t, J= 7.2 Hz), 1.60 (1H, m), 1.90 (1H, m), 2.41 (3H, s), 2.50 (2H, brt, J= 7.5 Hz), 2.68 (2H, brdt, J= 1.5, 7.2 Hz), 3.26 (2H, dt, J= 3.4, 7.2 Hz), 3.47 (1H, m), 3.67 (3H, s), 4.14 (2H, q, J= 7.2 Hz), 4.82 (1H, br), 5.84 (1H, brs), 7.29 (2H, d, J= 8.3 Hz), 7.88 (2H, d, J= 8.3 Hz), 10.04 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 1.25 (3Η, t, J = 7.2 Hz), 1.60 (1H, m), 1.90 (1H, m), 2.41 (3H, s), 2.50 (2H, brt, J = 7.5 Hz), 2.68 (2H, brdt, J = 1.5, 7.2 Hz), 3.26 (2H, dt, J = 3.4, 7.2 Hz), 3.47 (1H, m), 3.67 (3H, s), 4.14 ( 2H, q, J = 7.2 Hz), 4.82 (1H, br), 5.84 (1H, brs), 7.29 (2H, d, J = 8.3 Hz), 7.88 (2H, d, J = 8.3 Hz), 10.04 (1H, br).
元素分析値: C22H27N308S3として計算値: C, 47.38; H, 4.88; N, 7.53; S, 17.25Elemental analysis: C 22 H 27 N 3 0 8 S 3 Calculated: C, 47.38; H, 4.88 ; N, 7.53; S, 17.25
(%) 、 実測値: C, 47.11; H, 4.99; N, 7.52; S, 17.25 .( ) . (%), Found: C, 47.11; H, 4.99; N, 7.52; S, 17.25.
実施例 1-26 Example 1-26
異性体混合物 主/副生成物 Isomer mixture Main / By-product
]H-NMR (300MHz, CDCi3) : δ 1.24/1.25 (3H, t, J= 7.2 Hz), 2.39/2.41 (3H, s), 2.65/2, 66 (2H, brt, J= 7.5 Hz), 2.69/3.02 (IH, dd, J= 13.9/14.0, 8.3/7.2 Hz) , 2.80/3.13 (IH, dd, J= 13.9/14.0, 6.8/6.8 Hz), 3.20/3.23 (2H, t, J= 7.5 Hz), 3.48/3.58 (IH, dt, J= 1.5/2.6, 7.7/7.9 Hz), 4.13 (2H, q, J- 7.2 Hz), 4.70/4.75 (IH, br), 5.72/5.80 (IH, brs/d, J= 2.3 Hz), 7.10/7.21 (2H, d, J= 7.2/7.0 Hz), 7.27 (3H, m), 7.29 (2H, d, J= 8.3 Hz), 7.85/7.90 (2H, d, J= 8.3 Hz), 10.00 (IH, br). ] H-NMR (300MHz, CDCi 3 ): δ 1.24 / 1.25 (3H, t, J = 7.2 Hz), 2.39 / 2.41 (3H, s), 2.65 / 2, 66 (2H, brt, J = 7.5 Hz) , 2.69 / 3.02 (IH, dd, J = 13.9 / 14.0, 8.3 / 7.2 Hz), 2.80 / 3.13 (IH, dd, J = 13.9 / 14.0, 6.8 / 6.8 Hz), 3.20 / 3.23 (2H, t, J = 7.5 Hz), 3.48 / 3.58 (IH, dt, J = 1.5 / 2.6, 7.7 / 7.9 Hz), 4.13 (2H, q, J- 7.2 Hz), 4.70 / 4.75 (IH, br), 5.72 / 5.80 ( IH, brs / d, J = 2.3 Hz), 7.10 / 7.21 (2H, d, J = 7.2 / 7.0 Hz), 7.27 (3H, m), 7.29 (2H, d, J = 8.3 Hz), 7.85 / 7.90 (2H, d, J = 8.3 Hz), 10.00 (IH, br).
元素分析値: C25H27N306S3 · 0.4H20として計算値: C, 52.78; H, 4.93; N, 7.39; S, 16.91 (%) .実測値: C, 52.56; H, 4.89; N, 7.52; S, 17.41 (%) . Elemental analysis: C 25 H 27 N 3 0 6 S 3 · 0.4H 2 0 Calculated: C, 52.78; H, 4.93; N, 7.39; S, 16.91 (%). Observed: C, 52.56; H , 4.89; N, 7.52; S, 17.41 (%).
実施例 1-27 Example 1-27
'H-NMR (300MHz, CDC13) : δ 1.25 (3Η, t, J= 7.1 Hz), 2.40 (3H, s), 2.70 (2H, t, J= 7.3 Hz), 3.03 (IH, dd, J= 13.1, 1.4 Hz), 3.23 (IH, dd, J= 13.1, 2.3 Hz), 3.27 (2H, q, J= 7.3 Hz), 4.14 (2H, q, J= 7.1 Hz), 4.83 (IH, br), 6.10 (IH, brs), 7.28 (2H, d, J= 8.2 Hz), 7.87 (2H, d, J= 8.2 Hz), 10.03 (IH, br). 元素分析値: C18H21N306S3として計算値: C, 45.85; H, 4.49; N, 8.91; S, 20.40 (%)ゝ 実測値: C, 45.76; H, 4.60; N, 8.81; S, 20.52 (%) . 'H-NMR (300MHz, CDC1 3): δ 1.25 (3Η, t, J = 7.1 Hz), 2.40 (3H, s), 2.70 (2H, t, J = 7.3 Hz), 3.03 (IH, dd, J = 13.1, 1.4 Hz), 3.23 (IH, dd, J = 13.1, 2.3 Hz), 3.27 (2H, q, J = 7.3 Hz), 4.14 (2H, q, J = 7.1 Hz), 4.83 (IH, br ), 6.10 (IH, brs), 7.28 (2H, d, J = 8.2 Hz), 7.87 (2H, d, J = 8.2 Hz), 10.03 (IH, br). Elemental analysis: C 18 H 21 N 3 0 6 S 3 calculated: C, 45.85; H, 4.49 ; N, 8.91; S, 20.40 (%)ゝFound: C, 45.76; H, 4.60 ; N, 8.81; S, 20.52 (%).
実施例 1-28 Example 1-28
1:1 異性体混合物 1: 1 isomer mixture
Ή-NMR (300MHz, CDC13) : δ 1.26 (3Η, t, J= 7.2 Hz), 1.71/1.95 (IH, m), 2.15/2.29 (IH, m), 2.43/2.42 (3H, s), 2.71/2.67 (2H, t, J= 7.2 Hz), 3.25 (2H, m), 3.33 (IH, m), 3.66/3.67 (IH, brd, J= 7.2/6.8 Hz), 3.88 (2H, m), 4.15/4.14 (2H, q, J= 7.2 Hz), 5.81/6.04 (IH, dd, J= 7.2/6.8, 3.0/1.5 Hz), 7.31/7.29 (2H, d, J= 8.3 Hz), 7.74 (2H, m), 7.85 (4H, m), 9.68 (IH, brs). Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3Η, t, J = 7.2 Hz), 1.71 / 1.95 (IH, m), 2.15 / 2.29 (IH, m), 2.43 / 2.42 (3H, s), 2.71 / 2.67 (2H, t, J = 7.2 Hz), 3.25 (2H, m), 3.33 (IH, m), 3.66 / 3.67 (IH, brd, J = 7.2 / 6.8 Hz), 3.88 (2H, m) , 4.15 / 4.14 (2H, q, J = 7.2 Hz), 5.81 / 6.04 (IH, dd, J = 7.2 / 6.8, 3.0 / 1.5 Hz), 7.31 / 7.29 (2H, d, J = 8.3 Hz), 7.74 (2H, m), 7.85 (4H, m), 9.68 (IH, brs).
元素分析値: C28H28N408S3として計算値: C, 52.16; H, 4.38; N, 8.69; S, 14.92 (%) 、 実測値: C, 52.01; H, 4.27; , 8.69; S, 14.98 (%) . 実施例 1-29 Elemental analysis: C 28 H 28 N 4 0 8 S 3 Calculated: C, 52.16; H, 4.38 ; N, 8.69; S, 14.92 (%), Measured: C, 52.01; H, 4.27;, 8.69; S, 14.98 (%). Examples 1-29
'H-NMR (300MHz, CDC13) : δ 0.87 (3H, brt, J= 6.8 Hz), 1.26 (6H, br), 1.44 (2H, brquint, J= 7.1 Hz), 2.04 (2H, brt, J= 7.1 Hz), 2.41 (3H, s) , 4.22 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J= 8.3 Hz), 9, 73 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.87 (3H, brt, J = 6.8 Hz), 1.26 (6H, br), 1.44 (2H, brquint, J = 7.1 Hz), 2.04 (2H, brt, J = 7.1 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J = 8.3 Hz), 9, 73 (1H, brs).
元素分析値: C26H31N304S3 · 0.2H2Oとして計算値: C, 56.84; H, 5.76; N, 7.65; S, 17.51 (%), 実測値: C, 56.78; H, 5.66; N, 7.61; S, 17.83 (%) · Elemental analysis: C 26 H 31 N 3 0 4 S 3 · 0.2H 2 O Calculated: C, 56.84; H, 5.76 ; N, 7.65; S, 17.51 (%), Found: C, 56.78; H , 5.66; N, 7.61; S, 17.83 (%)
実施例 1 - 30 Example 1-30
異性体混合物 主/副生成物 Isomer mixture Main / By-product
'H-NMR (300MHz, CDC13) : δ 0.89/0.88 (3Η, t, J= 6.7 Hz), 1.27 (3H, t, J= 7.1 Hz), 1.29 (6H, m), 1.32 (2H, m), 1.55 (1H, m), 1.71/1.80 (3H, s), 2.10 (1H, m), 2.42 (3H, s), 2.69 (2H, t, J= 7.5 Hz), 3.25 (1H, m), 3.26/3.27 (2H, t, J= 7.6/7.5 Hz), 4.16 (2H, q, J= 7.1 Hz), 5.58/5.75 (1H, br), 7.29 (2H, brd, J= 8.2 Hz), 7.87/7.89 (2H, d, J= 8.2 Hz), 9.76 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 0.89 / 0.88 (3Η, t, J = 6.7 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.29 (6H, m), 1.32 (2H, m ), 1.55 (1H, m), 1.71 / 1.80 (3H, s), 2.10 (1H, m), 2.42 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.25 (1H, m) , 3.26 / 3.27 (2H, t, J = 7.6 / 7.5 Hz), 4.16 (2H, q, J = 7.1 Hz), 5.58 / 5.75 (1H, br), 7.29 (2H, brd, J = 8.2 Hz), 7.87 / 7.89 (2H, d, J = 8.2 Hz), 9.76 (1H, br).
元素分析値: C25H35N306S3 · 0.5H20として計算値: C, 51.88; H, 6.27; N, 7.26; S, 16.62 (%), 実測値: C, 51.86; H, 5.97; N, 7.24; S, 16.38 (%) . Elemental analysis: C 25 H 35 N 3 0 6 S 3 · 0.5 H 2 0 Calculated: C, 51.88; H, 6.27; N, 7.26; S, 16.62 (%), Found: C, 51.86; H , 5.97; N, 7.24; S, 16.38 (%).
実施例 1-31 Example 1-31
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή-NMR (300MHz, CDC13) : δ 0.87/0.88 (3Η, ί, J= 6.8 Hz), 1.25 (3H, t, J= 7.2 Hz), 1.28 (2H, m), 1.31 (2H, m), 1.35 (1H, m), 1.48 (2H, m), 1.74/1.85 (1H, m), 2.40/2.41 (3H, s), 2.69 (2H, t, J= 7.4 Hz), 3.20/3.31 (1H, m), 3.25/3.27 (2H, brt, J= 7.4 Hz) , 4.14/4.15 (2H, q, J= 7.2 Hz), 4.69/4.91 (1H, brd/br, J= 5.3 Hz), 5.80/5.86 (1H, brd, J= 2.3/3.1 Hz), 7.28/7.29 (2H, brd, J= 8.2 Hz), 7.87/7.89 (2H, d, J= 8.2 Hz), 10.06 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.87 / 0.88 (3Η, ί, J = 6.8 Hz), 1.25 (3H, t, J = 7.2 Hz), 1.28 (2H, m), 1.31 (2H, m) , 1.35 (1H, m), 1.48 (2H, m), 1.74 / 1.85 (1H, m), 2.40 / 2.41 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.20 / 3.31 (1H , m), 3.25 / 3.27 (2H, brt, J = 7.4 Hz), 4.14 / 4.15 (2H, q, J = 7.2 Hz), 4.69 / 4.91 (1H, brd / br, J = 5.3 Hz), 5.80 / 5.86 (1H, brd, J = 2.3 / 3.1 Hz), 7.28 / 7.29 (2H, brd, J = 8.2 Hz), 7.87 / 7.89 (2H, d, J = 8.2 Hz), 10.06 (1H, br).
元素分析値: C23H31N306S3として計算値: C, 51.00; H, 5.77; N, 7.76; S, 17.76Elemental analysis: C 23 H 31 N 3 0 6 S 3 Calculated: C, 51.00; H, 5.77 ; N, 7.76; S, 17.76
(%) 、 実測値: C, 50.70; H, 5.54; N, 7.87; S, 17.53 (%) . (%), Found: C, 50.70; H, 5.54; N, 7.87; S, 17.53 (%).
実施例 1 - 32 Example 1-32
異性体混合物 主/副生成物 '(in 'ΗΖ) '(in 'ΜΙ) SS — ΟΓΙ ' (m ¾S) 6·0 - 09 : (ε10α3)丽- Isomer mixture Main / By-product '(in' ΗΖ) '(in' ΜΙ) SS — ΟΓΙ '( m ¾S) 60-09: ( ε 10α3) 丽-
"(in Έ ΓΟΙ-ΟΖ'β '(in 'ΗΖ) Γ8 - 6·Ζ ' ¾S) Z9'L-9fL '(ω ¾ΐ) Ζ8-9- Ζ·9 '(ιπ Έΐ) S H0 ' ¾S) 8Γδ-29 "S '(m Ή8) OS—SO '(in ΈΖ) S8 ー OS '(ω 'ΗΟΙ) 01 'Ζ-ΟΟ '(ω Ήε) 26'0-28"05 : 13(13)丽 -E ^ "(in Έ ΓΟΙ-ΟΖ'β '(in' ΗΖ) Γ8-6Ζ '¾S) Z9'L-9fL' (ω ¾ΐ) Ζ8-9- Ζ9 '(ιπ Έΐ) S H0' ¾S) 8Γδ-29 "S '(m Ή8) OS—SO' (in ΈΖ) S8 ー OS '(ω' ΗΟΙ) 01 'Ζ-ΟΟ' (ω Ήε) 26'0-28" 05 : 13 (13) 丽-E ^
9S-T画第 9S-T drawing
• ( ) 9 ·9 '13 ·' 8.ί 'S -ΌΓ8 'Ν : ¾ :60·,, '3 :摩腿牽 (%) Ζ8'9 '13 :03·81 'S :80 ·8 'Ν :9Ζ 'Η :88·^ '3:掛翥 +ェっマ 13sS9o¥¾6,3 :軍 ί ^挲 • () 9 · 9 '13 · '8.ί' S -ΌΓ8 'Ν: ¾: 60 ·,' 3: Thigh traction (%) Ζ8'9 '13: 030381 'S: 80 · 8 'Ν: 9Ζ' Η: 88 · ^ '3: Kake + Emma 13 s S 9 o ¥ ¾ 6, 3: Army ί ^ 挲
'(s-rq ¾t) ΟΑ'6 '(ΖΗ Γ8 =Γ 'Ρ 'ΕΖ)'(s-rq ¾t) ΟΑ'6' ( Ζ Η Γ8 = Γ 'Ρ' ΕΖ)
98· '(ΖΗ 6· =r 'ρ 'κ) τε'Α '(jq 'HI) 's ' l '1\ =ί 'Ρ Ήΐ) 91 ' £(ΖΗ ΟΖ98 '(ΖΗ 6ΖΗ = r' ρ 'κ) τε'Α' (jq 'HI)' s 'l' 1 \ = ί 'Ρ Ήΐ) 91' £ (ΖΗ ΟΖ
1·ί =ί 'Μ) l\ 'f '(ΖΗ 0Ί 'Γ2ΐ =ί 'ΡΡ 'Ηΐ) 96 "S '(ΖΗ 9"8ΐ =f 'Ρ Ήΐ) 09 "8 1ί = ί 'Μ) l \' f '(ΖΗ 0Ί' Γ2ΐ = ί 'ΡΡ' Ηΐ) 96 "S '(ΖΗ 9" 8ΐ = f' Ρ Ήΐ) 09 "8
'(ΖΗ 9 'Ζ'ί =1 ΊΡ 'ΗΖ) ΙΙ'Ζ '(ΖΗ 0Ί '9 · =ί 'ΡΡ ¾ΐ) 80*8 '(ΖΗ S'Z =f Ί 'ΗΖ) Wl '(S 'Η2) '(ZH 0*Α =ί 'Η2) 8Γΐ 9 : (ε1303 'ZHWOOS) 扁- '(ΖΗ 9'Ζ'ί = 1 ΊΡ 'ΗΖ) ΙΙ'Ζ' (ΖΗ 0Ί '9 · = ί' ΡΡ ¾ΐ) 80 * 8 '(ΖΗ S'Z = f Ί' ΗΖ) Wl '(S' Η2) '(ZH 0 * Α = ί' Η2) 8Γΐ 9: ( ε 1303 'ZHWOOS) flat-
•(s Jq 'Ηΐ) 9·0Ϊ '(in QI ¾ΐ) 68·9-9 '5 '(ιπ ·ト 0Γ '(s 'Η8) Zf '(ω ¾8) fS-irg '(ZH9-Z=f(S Jq 'Ηΐ) 9 90' (in QI ¾ΐ) 689-9 '5' (ιπ '0 0' '(s' Η8) Zf '(ω ¾8) fS-irg' (ZH9-Z = f
'1 ( ) U'Z '(in 'Η8ΐ) 96Ί-0 Ϊ '(ω ¾S) S6 Ό P : md) ¾WN-H, tS-ΐ '1 ( ) U'Z' (in 'Η8ΐ) 96Ί-0 Ϊ' (ω ¾S) S6 Ό P: md) ¾WN-H, tS-ΐ
• (%) Si '91 'S :6n 'N : S0'9 'H : 8S '3 :扉}腿牽 、(%) •91 'S -n-l 'Μ :91'9 'Η :I9'8S '3 :掛翥 1つ θ¥¾8 : ΟΤ • (%) Si '91 'S: 6n' N: S0'9 'H: 8S' 3: Door} Thigh pull, (%) • 91 'S -nl' Μ: 91'9 'Η: I9'8S '3: One hook θ ¥ ¾ 8 : ΟΤ
SC-I MSi牽 SC-I MSi
• (%) 81 '91 'S - fL 'N : i0'9 Ή :6Ϊ·Ζ3 '3 :掛腿牽 、(%) 2Χ*9ΐ 'S : ΐΓ 'Ν 0¾S ·0 · SS90¾9¾SZ3 :華 挲 • (%) 81 '91 'S-fL' N: i0'9 Ή: 6Ϊ · 3 '3: Thigh traction, (%) 2Χ * 9ΐ' S: ΐΓ 'Ν 0¾S · 0 · S S 9 0¾ 9 ¾ SZ 3 : Hana
•( q ¾ΐ) 20*01 '(ΖΗ VI =f 'Ρ ' Ζ) 68 Ί/1 Ί '(ΖΗ Ζ"Ζ =ί 'P-iq ΈΖ) QZ'WL '(ΖΗ Ί =ί q 'HI) 98*9/08 "9 '(ZH f S • (q ¾ΐ) 20 * 01 '(ΖΗ VI = f' Ρ 'Ζ) 68 Ί / 1 Ί' (ΖΗ Ζ "Ζ = ί 'P-iq ΈΖ) QZ'WL' (ΖΗ Ί = ί q 'HI ) 98 * 9/08 "9 '(ZH f S
=r 'jq/p-iq Ήΐ) ε8· /09· '(ΖΗ Ι - =r 'f> sr / ' q = r 'jq / p-iq Ήΐ) ε8 / 09' '(ΖΗ Ι-= r' f> sr / 'q
'ΗΖ) LZ'WZ '(ω ¾l) Og-g/ZZ'S '(ΖΗ Γ丄 =ί '1 ¾) 69 '(s 'Η8) / 0 Z '(∞ ¾Ι) 8 Hi '(«ι ) 8f "ΐ '( 'HI) S8't '(sjq ¾8) 6Π '(ΖΗ t -i =Γ 'ϊ 'Η8) 9Ζ"ΐ/92·ΐ '(ΖΗ ΓΑ/8'9 =f Ί ¾S) 88·0/Α8·0 Q : (81303 '聊 0S) 誦- 'ΗΖ) LZ'WZ' (ω ¾l) Og-g / ZZ'S '(ΖΗ Γ 丄 = ί' 1 ¾) 69 '(s' Η8) / 0 Z '(∞ ¾Ι) 8 Hi' («ι) 8f "ΐ '(' HI) S8't '(sjq ¾8) 6Π' (ΖΗ t -i = Γ 'ϊ' Η8) 9Ζ" ΐ / 92ΐ ΐ '(ΓΑ ΓΑ / 8'9 = f Ί ¾S) 88 · 0 / Α8 · 0 Q: ( 8 1303 'lia 0S) recitation-
0t9爾 Odf/ェ:) d 909Z60/Z0 OAV 3.21-3.30 (2H, m), 3.70 (3H, s), 5.48-5.80 (1H, m), 7.46-7.61 (3H, m), 7.96-8.03 (2H, m), 9.70 (1H, br s). 0t9 爾 Odf / ェ :) d 909Z60 / Z0 OAV 3.21-3.30 (2H, m), 3.70 (3H, s), 5.48-5.80 (1H, m), 7.46-7.61 (3H, m), 7.96-8.03 (2H, m), 9.70 (1H, br s) .
実施例 1-38 Example 1-38
Ή-NMR (300MHz, CDC13) : δ 1.25 (3H, t, J= 7.2 Hz), 2.44 (3H, s), 2.68 (2H, t, J= 7.2 Hz), 3.15 (1H, d, J= 13.6 Hz), 3.24 (1H, dt, J= 6.8, 6.8 Hz), 3.27 (1H, quint, J= 7.2 Hz), 3.42 (1H, d, J= 13.2 Hz), 4.14 (2H, q, J= 7.2 Hz), 5.60 (1H, s), 6.99 (2H, d, J 8.7 Hz), 7.04 (2H, brd, J= 8.3 Hz), 7.15 (1H, t, J= 7.5 Hz), 7.27 (2H, m), 7.32 (2H, d, J= 7.9 Hz), 7.36 (2H, brt, J= 7.5 Hz), 7.89 (2H, d, J= 8.3 Hz), 9.68 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 1.25 (3H, t, J = 7.2 Hz), 2.44 (3H, s), 2.68 (2H, t, J = 7.2 Hz), 3.15 (1H, d, J = 13.6 Hz), 3.24 (1H, dt, J = 6.8, 6.8 Hz), 3.27 (1H, quint, J = 7.2 Hz), 3.42 (1H, d, J = 13.2 Hz), 4.14 (2H, q, J = 7.2 Hz), 5.60 (1H, s), 6.99 (2H, d, J 8.7 Hz), 7.04 (2H, brd, J = 8.3 Hz), 7.15 (1H, t, J = 7.5 Hz), 7.27 (2H, m), 7.32 (2H, d, J = 7.9 Hz), 7.36 (2H, brt, J = 7.5 Hz), 7.89 (2H, d, J = 8.3 Hz), 9.68 (1H, brs).
元素分析値: C3。H29N307S3として計算値: C, 56.32; H, 4.57; N, 6.57; S, 15.04 (%), 実測値: C, 56.33; H, 4.56; N, 6.30; S, 14.86 (%) . Elemental analysis: C 3. H 29 N 3 0 7 S 3 Calculated: C, 56.32; H, 4.57; N, 6.57; S, 15.04 (%), Found: C, 56.33; H, 4.56; N, 6.30; S, 14.86 ( %).
実施例 1-39 Example 1-39
1:1異性体混合物 主/副生成物 1: 1 isomer mixture main / by-product
Ή— NMR (300MHz, DMS0-d6) : δ 2.35/2.38 (3Η, s), 3.60 (1H, brd, J= 12.1 Hz), 3.99 (1H, d, J= 12.1 Hz), 7.03 (2H, brd, J= 8.7 Hz), 7.06 (2H, brd, J= 9.0 Hz), 7.18 (1H, t, J= 7.3 Hz), 7, 35/7.39 (2H, d, J= 8.0 Hz), 7.42 (2H, brt, J= 7.8 Hz), 7.72 (2H, brd, J= 7.7 Hz), 7.81/7.86 (2H, d, J= 8.3/7.5 Hz), 10.59/10.74 (1H, br/s), 11.24/11.40 (1H, s/br) . Ή— NMR (300MHz, DMS0-d 6 ): δ 2.35 / 2.38 (3Η, s), 3.60 (1H, brd, J = 12.1 Hz), 3.99 (1H, d, J = 12.1 Hz), 7.03 (2H, brd, J = 8.7 Hz), 7.06 (2H, brd, J = 9.0 Hz), 7.18 (1H, t, J = 7.3 Hz), 7, 35 / 7.39 (2H, d, J = 8.0 Hz), 7.42 ( 2H, brt, J = 7.8 Hz), 7.72 (2H, brd, J = 7.7 Hz), 7.81 / 7.86 (2H, d, J = 8.3 / 7.5 Hz), 10.59 / 10.74 (1H, br / s), 11.24 /11.40 (1H, s / br).
元素分析値: C25H21N305S3 · 0.3H20として計算値: C, 55.09; H, 3.99; N, 7.71; S, 17.65 (%), 実測値: C, 55.16; H, 4.03; N, 7.65; S, 17.42 ( ) . Elemental analysis: C 25 H 21 N 3 0 5 S 3 · 0.3 H 2 0 Calculated: C, 55.09; H, 3.99; N, 7.71; S, 17.65 (%), found: C, 55.16; H , 4.03; N, 7.65; S, 17.42 ().
実施例 1-40 Example 1-40
異性体混合物 Isomer mixture
'H-NMR (300 MHz, 重クロ口ホルム中): <50.88/0.89 (3H, t, J=6.8Hz), 1.26 (3H, t, J=7.2Hz), 1.29 (6H, m), 1.33 (2H, m), 1.75 (1H, m), 1.84 (1H, m), 2.70/2.70 (2H, t, J二 7.2 Hz), 3.27 (3H, m), 4.15 (2H, q, J= 7.2 Hz), 5.74/5.81 (1H, m), 7.02/7.04 (2H, d, J= 9.0 Hz), 7.07 (2H, d, J= 8 Hz), 7.21 (1H, brt, J= 7.9 Hz), 7.41 (2H, brt, J- 7.7 Hz), 7.92/7.93 (2H, dt, J= 8.7, 1.9 Hz), 9.65 (1H, br). 'H-NMR (300 MHz, in double-mouthed form): <50.88 / 0.89 (3H, t, J = 6.8Hz), 1.26 (3H, t, J = 7.2Hz), 1.29 (6H, m), 1.33 (2H, m), 1.75 (1H, m), 1.84 (1H, m), 2.70 / 2.70 (2H, t, J2 7.2 Hz), 3.27 (3H, m), 4.15 (2H, q, J = 7.2 Hz), 5.74 / 5.81 (1H, m), 7.02 / 7.04 (2H, d, J = 9.0 Hz), 7.07 (2H, d, J = 8 Hz), 7.21 (1H, brt, J = 7.9 Hz), 7.41 (2H, brt, J- 7.7 Hz), 7.92 / 7.93 (2H, dt, J = 8.7, 1.9 Hz), 9.65 (1H, br).
元素分析値: C29H35N307S3として計算値: C, 54.96; H, 5.57; N, 6.63; S, 15.18 (%)、 実測値; C, 54.87; H, 5.20; , 6.72; S, 15.20 (%) . 実施例 1-41 Elemental analysis: C 29 H 35 N 3 0 7 S 3 Calculated: C, 54.96; H, 5.57 ; N, 6.63; S, 15.18 C, 54.87; H, 5.20;, 6.72; S, 15.20 (%). Examples 1-41
'Η - NMR(CDC13, 300 MHz): δ 2.01 (3H, s), 2.42 (3H, s), 3.18 (3H, s), 3.68 (3H, s), 3.71 (IH, s), 4.13 (2H, d, J= 12.5 Hz), 4.25(2H, d, J= 12.5 Hz) 7.27 (5H, m), 7.28 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.62 (IH, brs). 元素分析値: C24H25N306S3として計算値: C, 52.63; H, 4.60; N, 7.67; S, 17.56 'Η - NMR (CDC1 3, 300 MHz): δ 2.01 (3H, s), 2.42 (3H, s), 3.18 (3H, s), 3.68 (3H, s), 3.71 (IH, s), 4.13 ( 2H, d, J = 12.5 Hz), 4.25 (2H, d, J = 12.5 Hz) 7.27 (5H, m), 7.28 (2H, d, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz) .), 9.62 (IH, brs ) elemental analysis: C 24 H 25 N 3 0 6 S 3 calculated: C, 52.63; H, 4.60 ; N, 7.67; S, 17.56
(%)、 実測値: C, 52.53; H, 4.61; , 7.65; S, 17.45 (%) . (%), Found: C, 52.53; H, 4.61;, 7.65; S, 17.45 (%).
実施例卜 42 Example 42
Ή-N R (CDC13, 300 MHz): δ 1.03 (6Η, d, J= 6.7 Hz): 1.85 (3H, s), 1.90 (IH, sept, J= 6.8 Hz), 2.42 (3H, s), 2.88 (2H, d, J= 6.8 Hz), 3.00 (IH, d, J= 13.3 Hz), 3.15 (IH, d, J= 13.4 Hz), 5.26 (1H, s), 7.30 (2H, d, J= 8.3 Hz), 7.89 (2H, d, J= 8.3 Hz), 9.61 (IH, brs). Ή-NR (CDC1 3, 300 MHz): δ 1.03 (6Η, d, J = 6.7 Hz): 1.85 (3H, s), 1.90 (IH, sept, J = 6.8 Hz), 2.42 (3H, s), 2.88 (2H, d, J = 6.8 Hz), 3.00 (IH, d, J = 13.3 Hz), 3.15 (IH, d, J = 13.4 Hz), 5.26 (1H, s), 7.30 (2H, d, J = 8.3 Hz), 7.89 (2H, d, J = 8.3 Hz), 9.61 (IH, brs).
元素分析値: C18H23N304S3として計算値: C, 48.96; H, 5.25; N, 9.52; S, 21.78 (%)、 実測値: C, 49.00; H, 5.23; N, 9.55; S, 21.92 (%) . Elemental analysis: C 18 H 23 N 3 0 4 Calculated S 3: C, 48.96; H , 5.25; N, 9.52; S, 21.78 (%), Found: C, 49.00; H, 5.23 ; N, 9.55; S, 21.92 (%).
実施例卜 43 Example 43
異性体混合物 主/副生成物 Isomer mixture Main / By-product
Ή-NMR (CDC13, 300 MHz): δ 1.28/1.32 (3Η, t, J= 7.2 Hz), 1.83/2.02 (3H, s), 2.72/2.74 (2H, t, J= 7.5 Hz), 3.13/3.14 (3H, s), 3.29 (2H, m), 3.72/3.72 (3H, s), 3.82/3.95 (IH, s), 4.22/4.27 (2H, q, J= 7.2 Hz), 4.88/6.01 (IH, brs/s), 9.55 (IH, brs). Ή-NMR (CDC1 3, 300 MHz): δ 1.28 / 1.32 (3Η, t, J = 7.2 Hz), 1.83 / 2.02 (3H, s), 2.72 / 2.74 (2H, t, J = 7.5 Hz), 3.13 /3.14 (3H, s), 3.29 (2H, m), 3.72 / 3.72 (3H, s), 3.82 / 3.95 (IH, s), 4.22 / 4.27 (2H, q, J = 7.2 Hz), 4.88 / 6.01 (IH, brs / s), 9.55 (IH, brs).
元素分析値: C15H2iN308S3として計算値: C, 38.53; H, 4.53; N, 8.99; S, 20.58Elemental analysis: Calculated as C 15 H 2i N 3 0 8 S 3: C, 38.53; H, 4.53; N, 8.99; S, 20.58
(%)、 実測値: C, 38.57; H, 4.52; N, 9.10; S, 20.40 (%) . (%), Found: C, 38.57; H, 4.52; N, 9.10; S, 20.40 (%).
実施例 1 - 44 Example 1-44
酢酸ェチルから再結晶して融点 140- 142 の淡黄色結晶を得た。 Recrystallization from ethyl acetate gave pale yellow crystals with a melting point of 140-142.
赤外吸収スぺクトル(IR) (KBr中) v 3700-3100, 2961, 2934, 2872, 1730, 1624, 1557 cm-Infrared absorption spectrum (IR) (in KBr) v 3700-3100, 2961, 2934, 2872, 1730, 1624, 1557 cm-
Ή-N R (CDC13) : δ 0.974 (3Η, t, J=7.2 Hz), 1.27 (3H, t, J=7.0Hz), 1.20-1.58 (3H, m), 1.71 (3HX0.87, s), 1.80 (3HX0.13, s), 2.00-2.18 (IH, m), 2.42 (3H, s), 2.69 (2H, t, J=7.6 Hz), 3.20-3.34 (1H, m), 3.27 (2H, i, J=7.0 Hz), 4.17 (2H, q, J=7.4 Hz), 5.52 (1HX0.87, bs), 5.70 (1HX0.13, bs), 7.30 (2H, d, J=7.8 Hz), 7.87 (2H, d, J=8.4 Hz), 9.61 (1H, bs). Ή-NR (CDC1 3): δ 0.974 (3Η, t, J = 7.2 Hz), 1.27 (3H, t, J = 7.0Hz), 1.20-1.58 (3H, m), 1.71 (3HX0.87, s) , 1.80 (3HX0.13, s), 2.00-2.18 (IH, m), 2.42 (3H, s), 2.69 (2H, t, J = 7.6 Hz), 3.20-3.34 (1H, m), 3.27 (2H , I, J = 7.0 Hz), 4.17 (2H, q, J = 7.4 Hz), 5.52 (1HX0.87, bs), 5.70 (1HX0.13, bs), 7.30 (2H, d, J = 7.8 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.61 (1H, bs).
元素分析値: C22H29N306S3として Elementary analysis: as C 22 H 29 N 3 0 6 S 3
計算値 (%) : C, 50.07; H, 5.54; N, 7.96; S, 18.23. Calculated value (%): C, 50.07; H, 5.54; N, 7.96; S, 18.23.
実測値 (%) : C, 50.07; H, 5.53; N, 7.93; S, 18.11. Obtained value (%): C, 50.07; H, 5.53; N, 7.93; S, 18.11.
実施例 1 - 45 Example 1-45
酢酸ェチルから再結晶して融点 143- 145 の淡黄色結晶を得た。 Recrystallization from ethyl acetate gave pale yellow crystals having a melting point of 143-145.
赤外吸収スペクトル(IR) (KBr中) リ: 3700-2800, 2957, 1732, 1622, 1557 cm"1.Infrared absorption spectrum (IR) (in KBr): 3700-2800, 2957, 1732, 1622, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 0.934 (3Η, d, J=6.2 Hz), 0.990 (3H, d, J=6.4 Hz), 1.27 (3H, t, J-7.0 Hz), 1.37 (1H, t, J=10.6 Hz), 1.76 (3HX0.90, s), 1.81 (3H Ή-NMR (CDC1 3): δ 0.934 (3Η, d, J = 6.2 Hz), 0.990 (3H, d, J = 6.4 Hz), 1.27 (3H, t, J-7.0 Hz), 1.37 (1H, t , J = 10.6 Hz), 1.76 (3HX0.90, s), 1.81 (3HX
X0.10, s), 1.75-1.95 (2H, m), 2.42 (3H, s), 2.70 (2H, t, J=7.6 Hz), 3.26X0.10, s), 1.75-1.95 (2H, m), 2.42 (3H, s), 2.70 (2H, t, J = 7.6 Hz), 3.26
(2H, t, J=7.4 Hz), 3.30 (1H, t, J=11.4 Hz), 4.17 (2H, q, J=7.0 Hz), 5.54 (1H(2H, t, J = 7.4 Hz), 3.30 (1H, t, J = 11.4 Hz), 4.17 (2H, q, J = 7.0 Hz), 5.54 (1H
X0.81, bs), 5.64 (1HX0.19, bs), 7.30 (2H, d, J=8.0 Hz), 7.87 (2H, d, J=8.6X0.81, bs), 5.64 (1HX0.19, bs), 7.30 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.6
Hz), 9.61 (1H, bs). Hz), 9.61 (1H, bs).
元素分析値: C23H31N306S3として Elementary analysis: as C 23 H 31 N 3 0 6 S 3
計算値 (¾) : C, 51.00; H, 5.77; N, 7.76; S, 17.76. Calculated (¾): C, 51.00; H, 5.77; N, 7.76; S, 17.76.
実測値 (¾) : C, 51.04; H, 5.65; N, 7.89; S, 17.70. Found (¾): C, 51.04; H, 5.65; N, 7.89; S, 17.70.
実施例 1-46 Example 1-46
3:1 異性体混合物 主/副生成物 3: 1 isomer mixture main / by-product
'H-NMR (200MHz, CDC13) : δ 1.00/1.04 (3Η, t, J= 7.4 Hz), 1.27 (3H, t, J= 7.1'H-NMR (200MHz, CDC1 3): δ 1.00 / 1.04 (3Η, t, J = 7.4 Hz), 1.27 (3H, t, J = 7.1
Hz), 1.43/1.41 (3H, d, J= 7.0/6.8 Hz), 2.07 (2H, brq, J= 7.4 Hz), 2.43 (3H, s) , 2.68/3.69 (2H, t, J= 7.4/7.8 Hz) , 3.24 (2H, m), 3.32/3.48 (1H, q, J= 6.8/7.0Hz), 1.43 / 1.41 (3H, d, J = 7.0 / 6.8 Hz), 2.07 (2H, brq, J = 7.4 Hz), 2.43 (3H, s), 2.68 / 3.69 (2H, t, J = 7.4 / 7.8 Hz), 3.24 (2H, m), 3.32 / 3.48 (1H, q, J = 6.8 / 7.0
Hz), 4.16 (2H, q, J= 7.1 Hz), 5.65/5.95 (1H, s/br), 7.31 (2H, d, J= 8.4 Hz),Hz), 4.16 (2H, q, J = 7.1 Hz), 5.65 / 5.95 (1H, s / br), 7.31 (2H, d, J = 8.4 Hz),
7.88/7.87 (2H, d, J- 8.4 Hz), 9.63 (1H, brs). 7.88 / 7.87 (2H, d, J-8.4 Hz), 9.63 (1H, brs).
元素分析値: C21H27N306S3 · 0.25H2Oとして計算値: C, 48.68; H, 5.35; , 8.11; S,Elemental analysis: C 21 H 27 N 3 0 6 S 3 · 0.25H 2 O Calculated: C, 48.68; H, 5.35 ;, 8.11; S,
18.57 (%) 、 実測値: C, 48.63; H, 5.41; , 8.08; S, 18.66 (%) . 18.57 (%), Found: C, 48.63; H, 5.41;, 8.08; S, 18.66 (%).
実施例 1-47 Example 1-47
赤外吸収スぺクトル(IR) (KBr中) v: 3700-2800, 3061, 3031, 2984, 2938, 1732, 1626, 1557, 1495 cm—1. ¾-NMR (CDCI3) : 51.25 (3HX0.58, t, J=7.0 Hz), 1.26 (3HX0.42, t, J=7.0 Hz), 1.84 (3H, s), 2.41 (3HX0.58, s), 2.44 (3HX0.42, s), 2.56 (2HX0.58, t, J=7.2 Hz), 2.63 (2HX0.42, t, J=7.4Hz), 3.12 (2H, t, J=7.2 Hz), 3.16-3.32 (1H, m), 3.45-3.72 (2H, m), 4.13 (2HX0.58, q, J=7.4 Hz), 4.14 (2HX0.42, q, J=7.2 Hz), 5.59 (1HX0.58, bs), 5.95 (1HX0.42, bs), 7.12-7.38 (7H, m), 7.85 (2HX0.58, d, J=8. Hz), 7.90 (2HX0.42, d, J=8.4 Hz), 9.63 (1H, bs). 元素分析値: C26H29N306S3として Infrared absorption spectrum (IR) (in KBr) v: 3700-2800, 3061, 3031, 2984, 2938, 1732, 1626, 1557, 1495 cm- 1 . ¾-NMR (CDCI3): 51.25 (3HX0.58, t, J = 7.0 Hz), 1.26 (3HX0.42, t, J = 7.0 Hz), 1.84 (3H, s), 2.41 (3HX0.58, s) , 2.44 (3HX0.42, s), 2.56 (2HX0.58, t, J = 7.2 Hz), 2.63 (2HX0.42, t, J = 7.4 Hz), 3.12 (2H, t, J = 7.2 Hz), 3.16-3.32 (1H, m), 3.45-3.72 (2H, m), 4.13 (2HX0.58, q, J = 7.4 Hz), 4.14 (2HX0.42, q, J = 7.2 Hz), 5.59 (1HX0. 58, bs), 5.95 (1HX0.42, bs), 7.12-7.38 (7H, m), 7.85 (2HX0.58, d, J = 8.Hz), 7.90 (2HX0.42, d, J = 8.4 Hz ), 9.63 (1H, bs) Elementary analysis:. as C 26 H 29 N 3 0 6 S 3
計算値 (¾) : C, 54.24; H, 5.08; N, 7.30; S, 16.71. Calculated value (¾): C, 54.24; H, 5.08; N, 7.30; S, 16.71.
実測値 (%) : C, 54.18; H, 5.30; N, 7.13; S, 16.71. Found (%): C, 54.18; H, 5.30; N, 7.13; S, 16.71.
実施例 1-48 Example 1-48
8 - [(3-ェトキシ -3 -ォキソプロピル)スルファニル] -5-ヒドロキシ- 6-メチル [(4_メチルフエニル)スルホニル]ィミノ }- 3 -ォキソ -2, 3, 5, 6-テトラヒドロ -1H-イミダゾ [5, 1-c] [1, 4]チアジン- 5-カルボン酸 tert-ブチル 8-[(3-ethoxy-3-oxopropyl) sulfanyl] -5-hydroxy-6-methyl [(4_methylphenyl) sulfonyl] imino} -3-oxo-2,3,5,6-tetrahydro-1H-imidazo [5, 1-c] [1, 4] tert-butyl thiazine-5-carboxylate
参考例 3-2で合成した化合物(純度 80%, 1.07 g, 2.0匪 ol)、 参考例 99で得た 3-ブロモ -2-ォキソ酪酸 tert-ブチル(0.94 g, 約 2 mmol)およびトリェチルァミン (0.83 ml, 6.0 mmol)の DMF- THF (1:4, 20 ml)懸濁液を室温で 1時間攪拌した。 ついで反応液に参考例 3-2で合成した化合物 (純度 80%, 0.55 g, 1.0扁 ol)を 1 時間毎に 2回加えた。 反応液を室温で 3時間攪拌した。 反応液を濃縮し、 酢酸ェ チルで抽出した。 有機層を水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥 し、 濃縮した。 残渣をシリカゲル(15 g)のクロマトグラフィー (酢酸ェチルーへ キサン、 2:8-5:5) で精製し、 標題化合物 (0.97g, 1.66 mmol)を黄色油状物とし て得た。 The compound synthesized in Reference Example 3-2 (purity 80%, 1.07 g, 2.0 ol), tert-butyl 3-bromo-2-oxobutyrate (0.94 g, about 2 mmol) obtained in Reference Example 99, and triethylamine ( A suspension of 0.83 ml (6.0 mmol) in DMF-THF (1: 4, 20 ml) was stirred at room temperature for 1 hour. Then, the compound synthesized in Reference Example 3-2 (purity 80%, 0.55 g, 1.0 mmol) was added to the reaction solution twice every hour. The reaction was stirred at room temperature for 3 hours. The reaction solution was concentrated and extracted with ethyl acetate. The organic layer was washed with water and saturated saline, dried over sodium sulfate, and concentrated. The residue was purified by silica gel (15 g) chromatography (ethyl acetate-hexane, 2: 8-5: 5) to give the title compound (0.97 g, 1.66 mmol) as a yellow oil.
Ή-NMR (200MHz, CDC13) : δ 1.26 (3Η, t, J= 7.1 Hz) , 1.29 (3H, d, J= 7.0 Hz), 1.54 (9H, s), 2.43 (3H, s), 2.71 (2H, t, J= 7.5 Hz), 3.26 (2H, t, J= 7.4 Hz), 3.53 (1H, q, J= 7.0 Hz), 4.16 (2H, q, J= 7.1 Hz), 4.96 (1H, s), 7.31 (2H, d, J= 8.0 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.61 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.26 (3Η, t, J = 7.1 Hz), 1.29 (3H, d, J = 7.0 Hz), 1.54 (9H, s), 2.43 (3H, s), 2.71 (2H, t, J = 7.5 Hz), 3.26 (2H, t, J = 7.4 Hz), 3.53 (1H, q, J = 7.0 Hz), 4.16 (2H, q, J = 7.1 Hz), 4.96 (1H , s), 7.31 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.61 (1H, brs).
元素分析値: CMH31N308S3として計算値: C, 49.21; H, 5.33; N, 7.17; S, 16.42Elemental analysis: C M H 31 N 3 0 8 S 3 Calculated: C, 49.21; H, 5.33 ; N, 7.17; S, 16.42
(%)、 実測値: C, 49.13; H, 5.44; N, 6.94; S, 16.21 (%) · (%), Found: C, 49.13; H, 5.44; N, 6.94; S, 16.21 (%) ·
実施例 1-49 ' 3 - [(5_(1-ブロモェチル )-5-ヒドロキシ- 6-メチル -卜 {[(4-メチルフエニル)スル ホニル]ィミノト 3_ォキソ -2, 3, 5, 6 -テトラヒドロ- 1H-ィミダゾ [5, 1-c] [1, ]チア ジン- 8-ィル)スルファニル]プロピオン酸 ェチル Example 1-49 ' 3-[(5_ (1-bromoethyl) -5-hydroxy-6-methyl-tri {[(4-methylphenyl) sulfonyl] iminoto 3_oxo-2,3,5,6-tetrahydro-1H-imidazo [5 , 1-c] [1,] thiazin-8-yl) sulfanyl] ethyl propionate
参考例 3-2で合成した化合物(純度 80¾, 5.08 g, 9.46匪01) および 2, 4 -ジブ 口モ- 3 -ペン夕ノン(2.90 g, 11.8 mmol)の DMF - THF (1:9, 100 ml)懸濁液にトリ ェチルァミン(3.93 ml, 28.4醒 ol)を室温で 30分間かけて滴下し、 混合物を 30 分間攪拌した。 反応液を濃縮し、 酢酸ェチル (200 ml)で希釈し、 2¾炭酸水素ナト リゥム水溶液、 0.1 塩酸、水および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥 し、 濃縮した。 残渣をシリカゲル (50 g)のクロマトグラフィー (酢酸ェチルーへ キサン、 2:8-4:6)で精製し、 ジェチルエーテルから結晶化して標題化合物 (1.81 g, 3.05 mmol)を黄色粉末として得た。 The compound synthesized in Reference Example 3-2 (purity 80¾, 5.08 g, 9.46 maraudal 01) and 2,4-dibutene-3- (pentamerone) (2.90 g, 11.8 mmol) in DMF-THF (1: 9, To the suspension (100 ml), triethylamine (3.93 ml, 28.4 mmol) was added dropwise at room temperature over 30 minutes, and the mixture was stirred for 30 minutes. The reaction solution was concentrated, diluted with ethyl acetate (200 ml), washed with aqueous sodium hydrogen carbonate, 0.1 hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was purified by silica gel (50 g) chromatography (ethyl acetate-hexane, 2: 8-4: 6) and crystallized from getyl ether to give the title compound (1.81 g, 3.05 mmol) as a yellow powder. .
一 NMR (200MHz, CDC13) : δ 1.27 (3Η, t, J= 7.2 Hz), 1.41 (3H, d, J= 6.6 Hz), 1.76 (3H, d, J= 7.0 Hz), 2.44 (3H, s), 2.70 (2H, t, J= 7.3 Hz), 3.27 (2H, dt, J= 3.2, 7.2 Hz), 4.00 (1H, q, J= 6.9 Hz), 4.17 (2H, q, J= 7.2 Hz), 4.46 (1H, q, J= 6.9 Hz), 6.63 (1H, s), 7.33 (2H, d, J= 8.4 Hz), 7.89 (2H, d, J= 8.0 Hz), 9.73 (1H, br). One NMR (200MHz, CDC1 3): δ 1.27 (3Η, t, J = 7.2 Hz), 1.41 (3H, d, J = 6.6 Hz), 1.76 (3H, d, J = 7.0 Hz), 2.44 (3H, s), 2.70 (2H, t, J = 7.3 Hz), 3.27 (2H, dt, J = 3.2, 7.2 Hz), 4.00 (1H, q, J = 6.9 Hz), 4.17 (2H, q, J = 7.2 Hz), 4.46 (1H, q, J = 6.9 Hz), 6.63 (1H, s), 7.33 (2H, d, J = 8.4 Hz), 7.89 (2H, d, J = 8.0 Hz), 9.73 (1H, br).
元素分析値: C21H26N306S3Brとして計算値: C, 42.75; H, 4.42; N, 7.09; S, 16.23: Br, 13.48 ( ) 、 実測値: C, 42.55; H, 4.19; N, 7.16; S, 15.89: Br, 13.03 (%) . Elemental analysis: C 21 H 26 N 3 0 6 S 3 Br Calculated: C, 42.75; H, 4.42 ; N, 7.09; S, 16.23: Br, 13.48 (), Found: C, 42.55; H, 4.19; N, 7.16; S, 15.89: Br, 13.03 (%).
mp. 95.0 - 99.0°C mp. 95.0-99.0 ° C
実施例 1-50 Example 1-50
3- [(5 -ヒドロキシ- 6-メチル _1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォ キソ- 5-プロピオニル- 2, 3, 5, 6 -テトラヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン -8 -ィル)スルファニル]プロピオン酸ェチル 3-[(5-hydroxy-6-methyl_1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-5-propionyl-2,3,5,6-tetrahydro-1H-imidazo [5, 1-c] [1,4] thiazine-8-yl) sulfanyl] ethyl propionate
実施例 2-48と同様にして得た 2-ブロモ - 3,4-へキサジオン(8. OMIOI)を含む褐 色油状物(8.6 g) および参考例 3-2 で合成した化合物(純度 80%, 12.9 g, 24.0 mmol)の DMF - THF (1:4, 250 ml)懸濁液に室温でトリェチルァミン(10.0 ml, 72.0 mmol)を 10分間かけて加え、 混合物を 1.5時間攪拌した。 反応液を実施例 2 - 48 と同様に後処理して褐色油状物を得た。 この褐色油状物をシリカゲル(120 g)のク 口マトグラフィー (酢酸ェチル—へキサン、 15:85-40:60)で精製し、 酢酸ェチル ―へキサン(30: 70-35: 65)の黄褐色画分をジェチルエーテルから結晶化して標題 化合物 (3.19 g, 5.89 mmol)を黄色粉末として得た。 A brown oil (8.6 g) containing 2-bromo-3,4-hexadione (8. OMIOI) obtained in the same manner as in Example 2-48 and the compound synthesized in Reference Example 3-2 (purity: 80% , 12.9 g, 24.0 mmol) in DMF-THF (1: 4, 250 ml) was added at room temperature over 10 minutes with triethylamine (10.0 ml, 72.0 mmol) and the mixture was stirred for 1.5 hours. The reaction solution was worked up in the same manner as in Example 2-48 to obtain a brown oil. This brown oil is washed with silica gel (120 g). Purification by oral chromatography (ethyl acetate-hexane, 15: 85-40: 60), and the yellow-brown fraction of ethyl acetate-hexane (30: 70-35: 65) was crystallized from getyl ether to give the title. The compound (3.19 g, 5.89 mmol) was obtained as a yellow powder.
Ή-NMR (200MHz, CDC13) : δ 1.18 (3Η, d, J= 7.0 Hz), 1.23 (3H, t, J= 7.1 Hz), 1.27 (3H, t, J= 7.1 Hz), 2.43 (3H, s), 2.43 (IH, m), 2.71 (2H, t, J= 7.2 Hz), 2.72 (1H, m), 3.27 (2H, dt, J= 1.4, 7.0 Hz), 3.56 (IH, q, J= 6.8 Hz), 4.16 (2H, q, J= 7.1 Hz), 5.19 (IH, s), 7.31 (2H, d, J= 8.2 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.65 (1H, br). Ή-NMR (200MHz, CDC1 3 ): δ 1.18 (3Η, d, J = 7.0 Hz), 1.23 (3H, t, J = 7.1 Hz), 1.27 (3H, t, J = 7.1 Hz), 2.43 (3H , s), 2.43 (IH, m), 2.71 (2H, t, J = 7.2 Hz), 2.72 (1H, m), 3.27 (2H, dt, J = 1.4, 7.0 Hz), 3.56 (IH, q, J = 6.8 Hz), 4.16 (2H, q, J = 7.1 Hz), 5.19 (IH, s), 7.31 (2H, d, J = 8.2 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.65 (1H, br).
元素分析値: C22H27N307S3として計算値: C, 48.78; H, 5.02; N, 7.76; S, 17.76 ( ) 、 実測値: C, 48.69; H, 4.97; N, 7.82; S, 17.53 (%) . Elemental analysis: C 22 H 27 N 3 0 7 S 3 Calculated: C, 48.78; H, 5.02 ; N, 7.76; S, 17.76 (), Found: C, 48.69; H, 4.97 ; N, 7.82 ; S, 17.53 (%).
mp. 158.5-159.0°C mp.158.5-159.0 ° C
実施例 1-51 , Examples 1-51,
異性体混合物 主/副生成物 Isomer mixture Main / By-product
'H-NM (300MHz, CDC13) : δ 1.26 (3Η, t, J= 7.1 Hz), 1.54, 1.75, 1.90, 2.17, 2.85 (8H, m), 2.42 (3H, s), 2.69 (2H, t, J= 7.4 Hz), 3.25 (2H, t, J= 7.4 Hz),'H-NM (300MHz, CDC1 3): δ 1.26 (3Η, t, J = 7.1 Hz), 1.54, 1.75, 1.90, 2.17, 2.85 (8H, m), 2.42 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.25 (2H, t, J = 7.4 Hz),
3.30/3.22 (IH, m), 4.16 (2H, q, J= 7.1 Hz), 4.78/3.62 (IH, br), 7.29 (2H, brd,3.30 / 3.22 (IH, m), 4.16 (2H, q, J = 7.1 Hz), 4.78 / 3.62 (IH, br), 7.29 (2H, brd,
J= 8.3 Hz), 7.87/7.86 (2H, d, J= 8.3 Hz), 9.71 (IH, br) . J = 8.3 Hz), 7.87 / 7.86 (2H, d, J = 8.3 Hz), 9.71 (IH, br).
元素分析値: C22H27N306S3として計算値: C, 50.27; H, 5.18; N, 7.99; S, 18.30. (%) 、 実測値: C, 50.07; H, 5.16; N, 7.97; S, 18.40 (%) . Elemental analysis: C 22 H 27 N 3 0 6 S 3 Calculated:. C, 50.27; H, 5.18; N, 7.99; S, 18.30 (%), Found: C, 50.07; H, 5.16 ; N , 7.97; S, 18.40 (%).
実施例ト 52 Example G 52
8 - [ (3-ェトキシ- 3-ォキソプ口ピル)スルファニル] -5-ヒドロキシ- 6- (2-メ卜キシ -2 -ォキソェチル)_1- {[(4-メチルフエニル)スルホニル]イミノ}- 3 -ォキソ - 2, 3, 5, 6 -テトラヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 5 -力ルボン酸メチル 参考例 3-2で合成した化合物 (純度 90%, 9.63 g, 20.2 mmol)およびトリェチル ァミン(8.40 ml, 60.5腿 ol)の THF-DMF (9:1, 160 ml)懸濁液に室温で攪拌しな がら参考例 18で合成した 3 -ブロモ -2 -ケトグルタル酸ジメチル (純度 70%, 7.29 g, 20. l mmol)の THF-DMF (9:1, 20 ml) 溶液を 20分間かけて滴下し、 反応液を室温 で 4分時間攪拌した。 反応液を濃縮後、 酢酸ェチル (300 ml)で希釈し、 水、 7¾炭 酸水素ナトリウムおよび飽和食塩水で洗浄した。 水洗液は酢酸ェチル (200 ml)で 抽出した。 有機層を硫酸ナトリウムで乾燥、 濃縮乾固して橙色固形物を 得た。 これをジェチルエーテル中で粉砕し、 濾取後、 ジェチルエーテル で洗浄し、 乾燥して表題化合物 (6.22 g, 10.3 mmol)を橙色粉末として 得た。 8-[(3-Ethoxy-3-oxop-mouth pill) sulfanyl] -5-Hydroxy-6- (2-methoxy-2- oxoethyl) _1- {[(4-Methylphenyl) sulfonyl] imino} -3- Methyl oxo-2,3,5,6-tetrahydro-1H-imidazo [5,1-c] [1,4] thiazine-5-methylcarboxylate Compound synthesized in Reference Example 3-2 (purity 90%, 9.63 g, 20.2 mmol) and triethylamine (8.40 ml, 60.5 tmol) in THF-DMF (9: 1, 160 ml) while stirring at room temperature while stirring at room temperature. A solution of dimethyl ketoglutarate (purity 70%, 7.29 g, 20.l mmol) in THF-DMF (9: 1, 20 ml) was added dropwise over 20 minutes, and the reaction solution was stirred at room temperature for 4 minutes. The reaction solution was concentrated, diluted with ethyl acetate (300 ml), and washed with water, sodium hydrogencarbonate and saturated saline. Wash solution with ethyl acetate (200 ml) Extracted. The organic layer was dried over sodium sulfate and concentrated to dryness to obtain an orange solid. This was ground in getyl ether, collected by filtration, washed with getyl ether, and dried to give the title compound (6.22 g, 10.3 mmol) as an orange powder.
Ή-NMR (300MHz, CDC13): d 1.20 (3H, t, J- 7.2 Hz), 2.33 (3H, s), 2.49 (1H, dd, J= 17.7, 9.9 Hz), 2.58 (2H, t, J= 7.2 Hz), 2.75 (1H, dd, J= 17.7, 3.6 Hz), 3.11 (2H, m) , 3.67 (6H, s), 3.83 (1H, dd, J= 9.9, 3.6 Hz), 4.08 (2H, q, J= 7.1 Hz), 7.14 (2H, d, J= 7.8 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.81 (1H, br). Ή-NMR (300MHz, CDC1 3 ): d 1.20 (3H, t, J- 7.2 Hz), 2.33 (3H, s), 2.49 (1H, dd, J = 17.7, 9.9 Hz), 2.58 (2H, t, J = 7.2 Hz), 2.75 (1H, dd, J = 17.7, 3.6 Hz), 3.11 (2H, m), 3.67 (6H, s), 3.83 (1H, dd, J = 9.9, 3.6 Hz), 4.08 ( 2H, q, J = 7.1 Hz), 7.14 (2H, d, J = 7.8 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.81 (1H, br).
元素分析値: C23H27N3010S3 ·2Η20 として計算値: C, 43.32; Η, 4.90; Ν, 6.59; S, 15.09 (%) 、 実測値: C, 43.33; H, 4.30; N, 6.76; S, 14.94Elemental analysis: C 23 H 27 N 3 0 10 S 3 · 2Η 20 Calculated: C, 43.32; Η, 4.90; Ν, 6.59; S, 15.09 (%), found: C, 43.33; H, 4.30; N, 6.76; S, 14.94
(%) . (%).
実施例 2-1 Example 2-1
式 (V I I ) において R 5が n-ペンチル基、 R 6がメチル基、 R1 Qが— Z R 15 (∑が302、 R 15が P-トルィル基)、 Rがェチル基である化合 物の合成 R 5 is n- pentyl group in the formula (VII), R 6 is a methyl group, R 1 Q is - ZR 15 (sigma is 30 2, R 15 is P- Toruiru group) of the compound R is an Echiru group Synthesis
実施例 1-1で合成した化合物(19.6 g, 35.3 mmol)をトルエン(600 ml) および無水酢酸(4.08 ml)に溶解し、 D -力ンファ—- 10 -スルホン酸(8.20 g, 35.3 mmol) を加えて 1時間加熱還流した。 反応液を濃縮乾固後、 残 渣を酢酸ェチル (700 ml) で希釈し、 2 %炭酸水素ナトリウム水溶液、 水、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し た。 溶媒を減圧下に溜去し、 残渣にメタノールを加え、 生成した沈澱物 を濾取し、メタノールで洗浄後、乾燥して標題化合物の赤紫色粉末(U.2 g、 20.2 mmol) を得た。 The compound synthesized in Example 1-1 (19.6 g, 35.3 mmol) was dissolved in toluene (600 ml) and acetic anhydride (4.08 ml), and D-methyl-10-sulfonic acid (8.20 g, 35.3 mmol) was dissolved. Was added and heated under reflux for 1 hour. After the reaction solution was concentrated to dryness, the residue was diluted with ethyl acetate (700 ml), washed with a 2% aqueous sodium hydrogen carbonate solution, water, 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, methanol was added to the residue, and the resulting precipitate was collected by filtration, washed with methanol, and dried to obtain a red-purple powder of the title compound (U.2 g, 20.2 mmol). .
Ή-NMR (300 MHz, 重クロ口ホルム中): δ 0.90 (3Η, t, J= 6.9 Hz), 1. 7 (3H, t, J= 7.1 Hz), 1.32 (2H, m) , 1.34 (2H, m) , 1.47 (2H, brquint, J= 7.1 Hz), 2.11 (2H, brt, J= 7.4 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.70 (2H, t, J= 7.4 Hz), 3.21 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 7.28 (2H, d, J= 8.2 Hz), 7.84 (2H, d, J= 8.2 Hz), 9.5 (1H, br). ' Ή-NMR (300 MHz, in double-mouthed form): δ 0.90 (3Η, t, J = 6.9 Hz), 1.7 (3H, t, J = 7.1 Hz), 1.32 (2H, m), 1.34 ( 2H, m), 1.47 (2H, brquint, J = 7.1 Hz), 2.11 (2H, brt, J = 7.4 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 7.28 (2H, d, J = 8.2 Hz), 7.84 (2H, d, J = 8.2) Hz), 9.5 (1H, br). '
元素分析値: C24H31N305S3 として計算値: C, 53.61; H, 5.81; N, 7.81; S, 17.89 ( ), 実測値: C, 53.77; H, 5.80; N, 7.78; S, 17.92 (%) . 実施例 2-2 ' Elemental analysis: C 24 H 31 N 3 0 5 S 3 Calculated: C, 53.61; H, 5.81 ; N, 7.81; S, 17.89 (), Found: C, 53.77; H, 5.80 ; N, 7.78 ; S, 17.92 (%). Example 2-2 '
式 (V I I ) において R 5が n-へキシル基、 R 6が水素原子、 R1 Qが— Z R 15 (Zが S〇2、 R15が p-トルィル基)、 Rがェチル基である化合 物の合成 Formula (VII) in R 5 is hexyl group n-, R 6 is a hydrogen atom, R 1 Q is - compound ZR 15 (Z is S_〇 2, R 15 is p- Toruiru group), R is Echiru group Synthesis of things
実施例 1-2で合成した化合物(325 mg, 0.584 mmol)をトルエン(11 ml) に溶解し、 D-カンファ一- 10 -スルホン酸 (136 mg, 0.584 mmol) を加え て 2時間加熱還流した。反応液を酢酸ェチル(20 ml)で希釈し、水、 2 % 炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリ ゥムで乾燥し、 減圧下濃縮乾固した。 残渣をメタノールに溶解し、 逆相 系分取 HPLC (担体: 0DS、 YMC-Pack, S- 363-1- 15、 内径 30 mm x 長さ 250 mm、 ワイ · ェム · シィ社製、 移動相 : 0.05(v/v)%トリフルォロ酢酸 Z 75 (v/v)%ァセトニトリル水) に付し、 溶出容量 720〜900 ml の画分を集 めて濃縮後、 残水層を酢酸ェチル (45 ml) で抽出した。 有機層を 0.05N 塩酸、 水および飽和食塩水で洗浄し、 硫酸ナトリウムで乾燥後、 減圧下 濃縮乾固した。 残渣にエーテルを加え、 生成した沈澱物を濾取し、 エー テルで洗浄後、 乾燥して標題化合物の赤色粉末 (103 mg、 0.192 mmol) を得た。 The compound synthesized in Example 1-2 (325 mg, 0.584 mmol) was dissolved in toluene (11 ml), D-camphor-10-sulfonic acid (136 mg, 0.584 mmol) was added, and the mixture was heated under reflux for 2 hours. . The reaction solution was diluted with ethyl acetate (20 ml), washed with water, a 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure. The residue is dissolved in methanol, and reversed-phase preparative HPLC (Carrier: 0DS, YMC-Pack, S-363--15, inner diameter 30 mm x length 250 mm, manufactured by YMC Corp., mobile phase : Apply 0.05% (v / v)% trifluoroacetic acid Z 75 (v / v)% acetonitrile water), collect the fractions with elution volume of 720-900 ml, concentrate and concentrate the remaining aqueous layer with ethyl acetate (45 ml). ). The organic layer was washed with 0.05N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure. Ether was added to the residue, and the resulting precipitate was collected by filtration, washed with ether, and dried to give a red powder of the title compound (103 mg, 0.192 mmol).
Ή-NMR (300 MHz, 重クロ口ホルム中): <5 0.89 (3H, t, J= 6.8Hz), 1.27 (3H, t, J= 7.2 Hz), 1.29 (4H, brs), 1.32 (2H, m) , 1.46 (2H, brquint, J= 5.7 Hz), 2.07 (2H, brt, J= 7.5 Hz), 2.42 (3H, s), 2.70 (2H, t, J= 7.5 Hz), 3.23 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 6.50 (1H, s), 7.29 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.79 (1H, br), Ή-NMR (300 MHz, in double-mouthed form): <5 0.89 (3H, t, J = 6.8 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.29 (4H, brs), 1.32 (2H , m), 1.46 (2H, brquint, J = 5.7 Hz), 2.07 (2H, brt, J = 7.5 Hz), 2.42 (3H, s), 2.70 (2H, t, J = 7.5 Hz), 3.23 (2H , t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 6.50 (1H, s), 7.29 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz) ), 9.79 (1H, br),
元素分析値: C24H31N305S3 として計算値: (:, 53.61; H, 5.81; N, 7.81; S, 17.89 (%), 実測値: C, 53.58; H, 5.87; N, 7.89; S, 18.13 (%) . 実施例 2-3 式 (V I I ) において R 5がエトキシカルポニル基、 R 6がメチル基、 R 10がー Z R15 (Zが S 02、 R15が P-トルィル基)、 Rがェチル基であ る化合物の合成 Elemental analysis: C 24 H 31 N 3 0 5 S 3 Calculated: (:, 53.61; H, 5.81; N, 7.81; S, 17.89 (%), Found: C, 53.58; H, 5.87 ; N , 7.89; S, 18.13 (%). Examples 2-3 R 5 is ethoxy Cal Poni Le group in the formula (VII), R 6 is a methyl group, the synthesis of R 10 Gar ZR 15 (Z is S 0 2, R 15 is P- Toruiru group), R is Ru Echiru group Der compound
実施例 1-3で合成した化合物 (236 mg, 0.424 mmol) をトルエン (8 ml) に溶解し、 D-カンファ一- 10-スルホン酸 (98 mg, 0.424 mmol) を加えて 2時間加熱還流した。 反応液を酢酸ェチル (15mU で希釈し、 2 %炭酸 水素ナトリウム水溶液、 Q.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸 ナトリウムで乾燥し、 減圧下濃縮乾固した。 残渣にエーテルを加え、 生 成した沈澱物を濾取し、 エーテルで洗浄後、 乾燥して標題化合物の赤色 粉末 (174 mg、 0.323 mmol) を得た。 The compound (236 mg, 0.424 mmol) synthesized in Example 1-3 was dissolved in toluene (8 ml), D-camphor-10-sulfonic acid (98 mg, 0.424 mmol) was added, and the mixture was heated under reflux for 2 hours. . The reaction solution was diluted with ethyl acetate (15 mU, washed with 2% aqueous sodium hydrogen carbonate, Q.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, concentrated to dryness under reduced pressure, and ether was added to the residue. The resulting precipitate was collected by filtration, washed with ether, and dried to give a red powder of the title compound (174 mg, 0.323 mmol).
-龍 R (300 MHz, 重クロ口ホルム中): <5 1.27 (3H, t, J= 7.2 Hz), 1.32 (3H, t, J= 7.2 Hz), 2.42 (3H, s), 2.75 (3H, s), 2.71 (2H, t, J= 7.2 Hz), 3.26 (2H, t, J= 7.2 Hz), 4.17 (2H, q, J= 7.2 Hz), 4.26 (2H, q, J= 7.2 Hz), 7.30 (2H, d, J= 8.3 Hz) , 7.83 (2H, d, J= 8.3 Hz), 9.78 (1H, brs). -Dragon R (300 MHz, double-holed): <5 1.27 (3H, t, J = 7.2 Hz), 1.32 (3H, t, J = 7.2 Hz), 2.42 (3H, s), 2.75 (3H , s), 2.71 (2H, t, J = 7.2 Hz), 3.26 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J = 7.2 Hz), 4.26 (2H, q, J = 7.2 Hz) ), 7.30 (2H, d, J = 8.3 Hz), 7.83 (2H, d, J = 8.3 Hz), 9.78 (1H, brs).
元素分析値: C22H25N307S3 として計算値: C, 48.97; H, 4.67; N, 7.79; S, 17.83 { % ) , 実測値; 48.78; H, 4.59; N, 7.80; S, 17.91 (%) . 実施例 2-4 Elemental analysis: Calculated as C 22 H 25 N 3 0 7 S 3: C, 48.97; H, 4.67; N, 7.79; S, 17.83 {%), Found; 48.78; H, 4.59; N , 7.80; S, 17.91 (%). Example 2-4
式 (V I I ) において R 5が n-ブチル基、 R 6がメチル基、 R1 (^S— Z R 1 5 (Zが S〇2、 R 15が p-トルィル基)、 Rがェチル基である化合物 の合成 Formula (VII) in R 5 is n- butyl group, R 6 is a methyl group, R 1 (^ S- ZR 1 5 (Z is S_〇 2, R 15 is p- Toruiru group), R is a Echiru group Synthesis of compound
実施例卜 4で合成した化合物(415 mg, 0.766 mmol)をトルエン(14 ml) に溶解し、 D-カンファ一- 10-スルホン酸 (178 mg, 0.766 mmol) を加え て 1時間加熱還流した。 反応液を酢酸ェチル (30ml) で希釈し、 2 %炭 酸水素ナトリウム水溶液、 塩酸、 水および飽和食塩水で洗浄後、 硫 酸ナトリウムで乾燥し、 減圧下濃縮乾固した。 残渣をメタノールに溶解 し、 逆相系分取 HPLC (担体: 0DS、 YMC- Pack、 S- 363-15、 内径 30 mm X 長さ 250 mm、 移動相 : 0.05(v/v) トリフルォロ酢酸/ (v/v ァセトニ トリル水) に付し、 溶出容量 640〜780ml の画分を集めて濃縮後、 残水 層を酢酸ェチル (20ml) で抽出した。 有機層を 0·05Ν塩酸、 水おょぴ飽 和食塩水で洗浄し、 硫酸ナトリウムで乾燥後、 減圧下濃縮乾固した。 残 渣にエーテルを加え、 生成した沈澱物を濾取し、 エーテルで洗浄後、 乾 燥して標題化合物の赤色粉末 (155 mg、 0.296 mmol) を得た。 The compound (415 mg, 0.766 mmol) synthesized in Example 4 was dissolved in toluene (14 ml), D-camphor-10-sulfonic acid (178 mg, 0.766 mmol) was added, and the mixture was heated under reflux for 1 hour. The reaction solution was diluted with ethyl acetate (30 ml), washed with a 2% aqueous sodium hydrogen carbonate solution, hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure. The residue was dissolved in methanol and subjected to reversed-phase preparative HPLC (Carrier: 0DS, YMC-Pack, S-363-15, 30 mm ID x 250 mm length, mobile phase: 0.05 (v / v) trifluoroacetic acid / ( v / v acetonitrile water), collect fractions with an elution volume of 640-780 ml, concentrate, The layer was extracted with ethyl acetate (20ml). The organic layer was washed with 0.05M hydrochloric acid and saturated sodium chloride solution, dried over sodium sulfate, and concentrated to dryness under reduced pressure. Ether was added to the residue, and the resulting precipitate was collected by filtration, washed with ether, and dried to give a red powder of the title compound (155 mg, 0.296 mmol).
Ή-NMR (300 MHz, 重クロ口ホルム中): δ 0.92 (3Η, t, J= 7.1 Hz), 1.26 (3H, t, J= 7.1 Hz), 1.34 (2H, m) , 1.44 (2H, m) , 2.12 (2H, brdd, J = 7.9, 7.0 Hz), 2.26 (3H, s), 2.40 (3H, s), 2.70 (2H, t, J= 7.4 Hz), 3.21 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.1 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.82 (1H, br) . Ή-NMR (300 MHz, in double-mouthed form): δ 0.92 (3Η, t, J = 7.1 Hz), 1.26 (3H, t, J = 7.1 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.12 (2H, brdd, J = 7.9, 7.0 Hz), 2.26 (3H, s), 2.40 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.1 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.82 (1H, br).
元素分析値: C23H29N305S3 として計算値: C, 52.75; H, 5.58; N, 8.02; S, 18.37 (%), 実測値; C, 52.77; H, 5.38; N, 8.21; S, 18.28 (%) . 実施例 2-5 Elemental analysis: C 23 H 29 N 3 0 5 S 3 Calculated: C, 52.75; H, 5.58 ; N, 8.02; S, 18.37 (%), Found; C, 52.77; H, 5.38 ; N, 8.21; S, 18.28 (%). Example 2-5
式 (V I I ) において R5および R6がともにメチル基、 R1 Qがー Z R1 5 (Zが S〇2、 R15が p-トルィル基)、 Rがェチル基である化合物の合 成 Both methyl groups R 5 and R 6 in formula (VII), R 1 Q Gar ZR 1 5 (Z is S_〇 2, R 15 is p- Toruiru group), if the compound R is an Echiru group formed
参考例 3-2で合成した化合物 (40 g, 純度約 50%、 約 43 mmol) を THF (600 ml) に溶解し、 3-クロ口- 2-ブタノン (9.1 ml, 86 mmol) および トリェチルァミン (35.8 ml, 0.258 mol) を加えて室温で 1.5時間撹拌 後、 8時間加熱還流した。 反応液を濃縮後、 残渣を酢酸ェチル (600 ml) で希釈し、 2 %炭酸水素ナトリウム水溶液、 水、 0.1N塩酸、 水および飽 和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 減圧下濃縮乾固して標題 化合物を含む褐色油状物 (29g) を得た。 The compound synthesized in Reference Example 3-2 (40 g, about 50% purity, about 43 mmol) was dissolved in THF (600 ml), and 3-chloro-2-butanone (9.1 ml, 86 mmol) and triethylamine ( 35.8 ml, 0.258 mol) was added, and the mixture was stirred at room temperature for 1.5 hours, and then heated and refluxed for 8 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate (600 ml), washed with 2% aqueous sodium hydrogen carbonate, water, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated under reduced pressure to dryness Solidification afforded a brown oil (29 g) containing the title compound.
上記の褐色油状物をトル ン (580 ml) および無水酢酸 (11.4ml) に 溶解し、 P-トルエンスルホン酸 1水和物 (8.98 g, 47 mmol) を加えて 1 0 0 で 2時間加熱撹拌した。 反応液を濃縮後、 残渣を酢酸ェチル (700 ml) で希釈し、 水で四回および飽和食塩水で洗浄後、 硫酸ナトリウムで 乾燥した。 溶媒を減圧下に溜去し、 残渣にメタノールを加え、 生成した 沈澱物を濾取し、 メタノールで洗浄後、 乾燥して標題化合物の赤紫色粉 末 (9.68 g、 20.1 mmol) を得た。 Ή-NMR (300 MHz, 重クロ口ホルム中): δ 1.27 (3Η, t, J= 7.2 Hz), 1.79 (3H, brq, J= 0.8 Hz), 2.26 (3H, brq, J= 0.8 Hz), 2.42 (3H, s), 2.70 (2H, t, J= 7.5 Hz), 3.20 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.61 (1H, br). 元素分析値: C2。H23N305S3 として計算値: C, 49.88; H, 4.81; N, 8.72; S, 19.97 (%)、 実測値; C, 49.78; H, 5.03; N, 8.68; S, 20.00 (%) . 実施例 2 - 6 The above brown oil was dissolved in toluene (580 ml) and acetic anhydride (11.4 ml), and P-toluenesulfonic acid monohydrate (8.98 g, 47 mmol) was added. The mixture was heated and stirred at 100 for 2 hours. did. After concentration of the reaction solution, the residue was diluted with ethyl acetate (700 ml), washed four times with water and saturated brine, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, and methanol was added to the residue. The resulting precipitate was collected by filtration, washed with methanol, and dried to obtain a red-purple powder of the title compound (9.68 g, 20.1 mmol). Ή-NMR (300 MHz, in double-mouthed form): δ 1.27 (3Η, t, J = 7.2 Hz), 1.79 (3H, brq, J = 0.8 Hz), 2.26 (3H, brq, J = 0.8 Hz) , 2.42 (3H, s), 2.70 (2H, t, J = 7.5 Hz), 3.20 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.84 ( 2H, d, J = 8.3 Hz), 9.61 (1H, br) elemental analysis:. C 2. H 23 N 3 0 5 S 3 Calculated: C, 49.88; H, 4.81 ; N, 8.72; S, 19.97 (%), Found; C, 49.78; H, 5.03 ; N, 8.68; S, 20.00 ( %). Examples 2-6
式 (V I I ) において R 5がェチル基、 R6がメチル基、 R1 (がー Z R 1 5 (∑が3〇2、 R 15が p-トルィル基)、 Rがェチル基である化合物の合 成 Case of compounds wherein R 5 Echiru group, R 6 is a methyl group, R 1 (gar ZR 1 5 (sigma is 3_Rei 2, R 15 is p- Toruiru group), R is Echiru group in the formula (VII) Success
参考例 3- 2で合成した化合物 (8.70 g, 20.3 mmol) を THF (100 ml) および DMF(40ml)に溶解し、 3-ブロモ -2-ペン夕ノン(5.7 g, 純度 60%、 約 20 mmol) およびトリェチルァミン (8.4 ml, 61 mmol) を加えて室温 で 7.5時間撹拌した。 この間、 反応開始より 2、 4、 6時間後に 3 -プロ モ- 2-ペン夕ノン (1.4g, 純度 50%、 約 4mfflol) を加えた。 反応液を濃 縮後、 残渣を酢酸ェチル (300 ml) で希釈し、 水、 2 %炭酸水素ナトリ ゥム水溶液、 水、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリ ゥムで乾燥し、 減圧下濃縮乾固して標題化合物を含む褐色油状物 (8.6 g) を得た。 The compound synthesized in Reference Example 3-2 (8.70 g, 20.3 mmol) was dissolved in THF (100 ml) and DMF (40 ml), and 3-bromo-2-pentanone (5.7 g, purity 60%, approx. mmol) and triethylamine (8.4 ml, 61 mmol) were added, and the mixture was stirred at room temperature for 7.5 hours. During this time, 2, 4-, and 6 hours after the start of the reaction, 3-promo-2-pennone (1.4 g, purity 50%, about 4 mfflol) was added. After concentrating the reaction mixture, the residue was diluted with ethyl acetate (300 ml), washed with water, 2% aqueous sodium hydrogen carbonate, water, 0.1N hydrochloric acid, water and saturated saline, and then washed with sodium sulfate. It was dried and concentrated to dryness under reduced pressure to give a brown oil (8.6 g) containing the title compound.
上記の褐色油状物をトルエン (250 ml) および無水酢酸 (5.86 ml) に 溶解し、 p -トルエンスルホン酸 1水和物 (3.12 g, 16.2 mmol) を加えて 1 0 0 °Cで 2時間加熱撹拌した。反応液を濃縮後、残渣を酢酸ェチル(300 ml) で希釈し、 水で 3回および飽和食塩水で洗浄後、 硫酸ナトリウムで 乾燥した。 溶媒を減圧下に溜去し、 残渣にメタノールを加え、 生成した 沈澱物を濾取し、 メタノールで洗浄後、 乾燥して標題化合物の赤色粉末 (4.71 g、 9.50 mmol) を得た。 The above brown oil was dissolved in toluene (250 ml) and acetic anhydride (5.86 ml), p-toluenesulfonic acid monohydrate (3.12 g, 16.2 mmol) was added, and the mixture was heated at 100 ° C for 2 hours. Stirred. After concentration of the reaction solution, the residue was diluted with ethyl acetate (300 ml), washed three times with water and saturated brine, and dried over sodium sulfate. The solvent was evaporated under reduced pressure, methanol was added to the residue, and the resulting precipitate was collected by filtration, washed with methanol, and dried to give the title compound as a red powder (4.71 g, 9.50 mmol).
'Η-醒 R (300 MHz, 重クロ口ホルム中): δ 1.09 (3Η, t, J= 7.4Hz), 1.27 (3H, t, J= 7. Hz), 2.14 (2H, q, J= 7.0 Hz) , 2.26 (3H, s), 2.42 (3H, s), 2.70 (2H, t, J= 7.6 Hz), 3.22 (2H, t, J= 7.8 Hz), 4.17 (2H, q, J= 7.4 Hz), 7.29 (2H, d, J= 8.2 Hz), 7.84 (2H, d, J= 8,4 Hz), 9.63 (1H, br). 'Η-Awake R (300 MHz, in the double-mouthed form): δ 1.09 (3Η, t, J = 7.4 Hz), 1.27 (3H, t, J = 7. Hz), 2.14 (2H, q, J = 7.0 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.70 (2H, t, J = 7.6 Hz), 3.22 (2H, t, J = 7.8 Hz), 4.17 (2H, q, J = 7.4 Hz), 7.29 (2H, d, J = 8.2 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, br).
元素分析値: C21H25N305S3 として計算値: C, 50.89; H, 5.08; N, 8· 48; S, 19.41 (%), 実測値; C, 50.76; H, 4.84; N, 8.46; S, 19.37 ( ) . 実施例 2 - 7 Elemental analysis: C 21 H 25 N 3 0 5 S 3 Calculated: C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%), found; C, 50.76; H, 4.84; N, 8.46; S, 19.37 (). Examples 2-7
式 (V I I ) において R 5および R 6がともにメチル基、 R1 Qが— Z R1 5 (∑が3〇2、 R 15が p-クロ口フエニル基)、 Rがェチル基である化合 物の合成 Formula (VII) in which R 5 and R 6 are both methyl, R 1 Q is - ZR 1 5 (Σ is 3_Rei 2, R 15 is p- black port phenyl group), the compound wherein R is an Echiru group Synthesis
参考例 3-1で合成した化合物 (7.9 g、 純度約 7 0 %、 12.3 mmol) を THF (130 ml) に溶解し、 3 -クロ口— 2 -ブタノン (1.66 ml, 15.7 mmol) およびトリェチルァミン (5.93 ml, 42.8 mmol) を加えて 7時間加熱還 流した。 さらに 3-クロ口- 2-ブタノン (0.45 ml, 4.28 mmol) およびト リエチルァミン (1.97 ml, 14.3 mmol) を加えてさらに 5時間加熱還流 した。 反応液を濃縮後、 残渣を酢酸ェチル (200 ml) で希釈し、 水、 お よび飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 減圧下濃縮乾固し て褐色油状物 (7.7g) を得た。 The compound synthesized in Reference Example 3-1 (7.9 g, purity of about 70%, 12.3 mmol) was dissolved in THF (130 ml), and 3-chloro-2-butanone (1.66 ml, 15.7 mmol) and triethylamine ( 5.93 ml, 42.8 mmol) was added and the mixture was refluxed for 7 hours. Further, 3-chloro-2-butanone (0.45 ml, 4.28 mmol) and triethylamine (1.97 ml, 14.3 mmol) were added, and the mixture was further refluxed for 5 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate (200 ml), washed with water and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure to give a brown oil (7.7 g). Obtained.
上記の褐色油状物をトルエン(140 ml)および無水酢酸 .12 ml, 42.8 mmol) に溶解し、 P-トルエンスルホン酸 1水和物 (2.17 g, 11.4 mmol) を加えて室温で 6 3時間撹拌した。 反応液を酢酸ェチル (200 ml) で希 釈し、水で 3回および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥した。 溶媒を減圧下に溜去し、 残渣にメタノールを加え、 生成した沈澱物を濾 取し、 メタノールで洗浄後、 乾燥して標題化合物の灰赤色粉末(3.67g、 7.31 mmol) を得た。 The above brown oil was dissolved in toluene (140 ml) and acetic anhydride .12 ml, 42.8 mmol), and P-toluenesulfonic acid monohydrate (2.17 g, 11.4 mmol) was added, followed by stirring at room temperature for 63 hours. did. The reaction solution was diluted with ethyl acetate (200 ml), washed three times with water and saturated saline, and dried over sodium sulfate. The solvent was evaporated under reduced pressure, methanol was added to the residue, and the resulting precipitate was collected by filtration, washed with methanol, and dried to give the title compound as a gray-red powder (3.67 g, 7.31 mmol).
Ή-N R (300 MHz, 重クロ口ホルム中): δ 1.27 (3Η, t, J- 7.1 Hz) ,· 1.80 (3H, brq, J= 1.0 Hz), 2.27 (3H, q, J= 1.2 Hz), 2.70 (2H, t, J= 7.4 Hz), 3.22 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J=- 7.0 Hz), 7.47 (2H, dt, J= 8.8, 1.8 Hz), 7.90 (2H, dt, J= 8.8, 2.2 Hz), 9.60 (1H, brs). 元素分析値: C19H2QN305S3C1 として計算値: C, 45.46; H, 4.02; N, 8.37; S, 19.16; Cl, 7.06 ( ) 実測値; C, 45.54; H, 3.97; N, 8.38; S, 19.07; CI, 6.82 (%) . Ή-NR (300 MHz, in the double-mouthed form): δ 1.27 (3Η, t, J- 7.1 Hz), 1.80 (3H, brq, J = 1.0 Hz), 2.27 (3H, q, J = 1.2 Hz) ), 2.70 (2H, t, J = 7.4 Hz), 3.22 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J =-7.0 Hz), 7.47 (2H, dt, J = 8.8, 1.8 . Hz), 7.90 (2H, dt, J = 8.8, 2.2 Hz), 9.60 (1H, brs) elemental analysis: calculated C 19 H 2Q N 3 0 5 S 3 C1: C, 45.46; H, 4.02 N, 8.37; S, 19.16; Cl, 7.06 () Found; C, 45.54; H, 3.97; N, 8.38; S, 19.07; CI, 6.82 (%).
実施例 2 - 8 Example 2-8
式 (V I I ) において R5および R6がともにメチル基、 R 1 ()が— Z R 1 5 (2が3〇2、 R 15が P-ブロモフエニル基)、 Rがェチル基である化合 物の合成 Formula (VII) in which R 5 and R 6 are both methyl, R 1 () is - ZR 1 5 (2 is 3_Rei 2, R 15 is P- Buromofueniru group) Synthesis of Compound R is Echiru group
参考例 3-4で合成した化合物 (5.36 g、 純度約 9 6 %、 10.5 mmol) を THF (110 ml) に溶解し、 3-クロ口- 2 -ブ夕ノン (1,22 ml, 11.6 mmol) およびトリェチルァミン (4.40 ml, 31.5 mmol) を加えて 2 0時間加熱 還流した。 反応液を酢酸ェチル (300 ml) で希釈し、 水、 2 %炭酸水素 ナトリウム水溶液および飽和食塩水で洗浄後、硫酸ナトリウムで乾燥し、 減圧下濃縮乾固して褐色油状物 (5.87g) を得た。 The compound synthesized in Reference Example 3-4 (5.36 g, purity about 96%, 10.5 mmol) was dissolved in THF (110 ml), and 3-chloro-2--2-butanone (1,22 ml, 11.6 mmol) was dissolved. ) And triethylamine (4.40 ml, 31.5 mmol) were added, and the mixture was refluxed for 20 hours. The reaction solution was diluted with ethyl acetate (300 ml), washed with water, 2% aqueous sodium hydrogen carbonate solution and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure to give a brown oil (5.87 g). Obtained.
上記の褐色油状物をトルエン (120 ml).および無水酢酸(3.03ml, 31.5 mmol) に溶解し、 p-トルエンスルホン酸 1水和物 (1.60 g, 8.40 mmol) を加えて 8 0 °Cで 2時間撹拌した。 反応液を減圧下に濃縮し、 残渣にメ タノールを加え、 生成した沈澱物を濾取し、 メタノールで洗浄後、 乾燥 して標題化合物の灰赤色粉末 (3.46 g、 6.33 mmol) を得た。 The above brown oil was dissolved in toluene (120 ml) and acetic anhydride (3.03 ml, 31.5 mmol), and p-toluenesulfonic acid monohydrate (1.60 g, 8.40 mmol) was added. Stir for 2 hours. The reaction solution was concentrated under reduced pressure, methanol was added to the residue, the resulting precipitate was collected by filtration, washed with methanol, and dried to give the title compound as an off-white powder (3.46 g, 6.33 mmol).
^- R (300 MHz, 重クロ口ホルム中): δ 1.27 (3Η, t, J= 6.9 Hz), 1.80 (3H, brq, J= 1.2 Hz), 2.27 (3H, q, J= 0.9 Hz), 2.71 (2H, t, J= 7.4 Hz), 3.22 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 7.64 (2H, dt, J= 8.7, 2.1 Hz), 7.83 (2H, dt, J= 8.7, 2.1 Hz), 9.60 (1H, brs) . 元素分析値: C19H2。N305S3Br として計算値: C, 41.76; H, 3.69; , 7.69; S, 17.60; Br, 14.62 ( ),実測値; C, 41.80; H, 3.71; N, 7.72; S, 17.61; Br, 14.42 (%) .融点: 165.0-167.0。C . ^ -R (300 MHz, in double-hole form): δ 1.27 (3Η, t, J = 6.9 Hz), 1.80 (3H, brq, J = 1.2 Hz), 2.27 (3H, q, J = 0.9 Hz) , 2.71 (2H, t, J = 7.4 Hz), 3.22 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.64 (2H, dt, J = 8.7, 2.1 Hz) , 7.83 (2H, dt, J = 8.7, 2.1 Hz), 9.60 (1H, brs) elemental analysis:. C 19 H 2. Calculated for N 3 0 5 S 3 Br: C, 41.76; H, 3.69;, 7.69; S, 17.60; Br, 14.62 (), found; C, 41.80; H, 3.71; N, 7.72; S, 17.61. Br, 14.42 (%). Melting point: 165.0-167.0. C.
以下に実施例 2- 1〜2-8 で合成した化合物および同様にして合成した 化合物の一覧を示す。 〔表 7〕The following is a list of compounds synthesized in Examples 2-1 to 2-8 and compounds synthesized in the same manner. (Table 7)
差替え用紙 (M 26) 〔表 8〕Replacement paper (M 26) (Table 8)
差替え用紙 »2β) 〔表 9〕Replacement paper »2β) (Table 9)
差替え用弒 (M026) 〔表 1 0〕'For replacement 弒 (M026) (Table 10) '
差替え用紙 〔表 1 1〕'Replacement paper (Table 11) '
差替え用飆 ( Μ2^ 〔表 1 2〕'Replacement bridal (Μ2 ^ (Table 12) '
差螢え^^; ^m ) 〔表 1 3〕'^^; ^ m) (Table 13) '
差替え用紙(規則 26) 以下に実施例 2- 9〜2- 78で合成した化合物のデ一夕を示す。 Replacement form (Rule 26) The following shows the results of the compounds synthesized in Examples 2-9 to 2-78.
実施例 2 - 9Example 2-9
-匪 R (300MHz, CDC13) : δ 1.27 (3Η, t, J= 7.1 Hz), 2.43 (3H, s),- negation R (300MHz, CDC1 3): δ 1.27 (3Η, t, J = 7.1 Hz), 2.43 (3H, s),
2.73 (2H, t, J= 7.4 Hz), 3.24 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 4.98 (1H, s), 7.20 (2H, dd, J= 8.3, 1.5 Hz), 7.32 (2H, d, J= 8.3 Hz), 7.37 (3H, m), 7.87 (2H, d, J= 8.3 Hz), 9.69 (1H, brs) . 2.73 (2H, t, J = 7.4 Hz), 3.24 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 4.98 (1H, s), 7.20 (2H, dd, J = 8.3, 1.5 Hz), 7.32 (2H, d, J = 8.3 Hz), 7.37 (3H, m), 7.87 (2H, d, J = 8.3 Hz), 9.69 (1H, brs).
元素分析値: C24H23N305S3として計算値: C, 54.42; H, 4.38; N, 7.93; S, 18.16 (%), 実測値: C, 54.23; H, 4.46; N, 8.00; S, 18.45 (%) . 実施例 2- 10 Elemental analysis: C 24 H 23 N 3 0 5 S 3 Calculated: C, 54.42; H, 4.38 ; N, 7.93; S, 18.16 (%), Found: C, 54.23; H, 4.46 ; N, 8.00; S, 18.45 (%). Example 2-10
Ή-NMR (300MHz, CDC13) : δ 2.43 (3Η, s), 2.74 (2H, t, J- 7.4 Hz), Ή-NMR (300MHz, CDC1 3 ): δ 2.43 (3Η, s), 2.74 (2H, t, J- 7.4 Hz),
3.24 (2H, t, J= 7.4 Hz), 3.72 (3H, s), 4.98 (1H, s) , 7.20 (2H, dd, J- 7.9, 1.8 Hz), 7.32 (2H, d, J= 8.2 Hz), 7.36 (3H, m), 7.87 (2H, d, J= 8.2 Hz), 9.68 (1H, brs). 3.24 (2H, t, J = 7.4 Hz), 3.72 (3H, s), 4.98 (1H, s), 7.20 (2H, dd, J-7.9, 1.8 Hz), 7.32 (2H, d, J = 8.2 Hz) ), 7.36 (3H, m), 7.87 (2H, d, J = 8.2 Hz), 9.68 (1H, brs).
元素分析値: C23H2IN305S3として計算値: C, 53.58; H, 4.10; N, 8.15; S, 18.66 ( ), 実測値: C, 53.66; H, 4.08; N, 8.14; S, 18.65 (%) . 実施例 2 - 11 Elemental analysis: C 23 H 2 I N 3 0 5 S 3 Calculated: C, 53.58; H, 4.10; N, 8.15; S, 18.66 (), found: C, 53.66; H, 4.08; N, 8.14 ; S, 18.65 (%). Examples 2-11
'H-NMR (300MHz, CDC13) : δ 1.28 (3Η, t, J= 7.1 Hz), 2.43 (3H, s), 2.73 (2H, t, J= 7.4 Hz), 3.25 (2H, t, J= 7.4 Hz), 4.18 (2H, q, J= 7.1 Hz), 5.04 (1H, s), 7.27 (2H, d, J= 8.2 Hz), 7.32 (2H, d, J= 8.3 Hz), 7.36 (1H, t, J= 7.2 Hz), 7.44 (2H, t, J= 7.8 Hz), 7.58 (2H, d, J= 8.2 Hz), 7.58 (2H, m), 7.88 (2H, d, J= 8.3 Hz), 9.73 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 1.28 (3Η, t, J = 7.1 Hz), 2.43 (3H, s), 2.73 (2H, t, J = 7.4 Hz), 3.25 (2H, t, J = 7.4 Hz), 4.18 (2H, q, J = 7.1 Hz), 5.04 (1H, s), 7.27 (2H, d, J = 8.2 Hz), 7.32 (2H, d, J = 8.3 Hz), 7.36 ( 1H, t, J = 7.2 Hz), 7.44 (2H, t, J = 7.8 Hz), 7.58 (2H, d, J = 8.2 Hz), 7.58 (2H, m), 7.88 (2H, d, J = 8.3) Hz), 9.73 (1H, brs).
元素分析値: C3。H27N305S3として計算値: C, 59.48; H, 4.49; N, 6.94; S, 15.88 ( ) 、 実測値: 59.41; H, 4.31; , 6.68; S, 15.94 ( ) . 実施例 2 - 12 Elemental analysis: C 3. H 27 N 3 0 5 S 3 Calculated: C, 59.48; H, 4.49; N, 6.94; S, 15.88 (), Found: 59.41; H, 4.31;, 6.68; S, 15.94 (). 2-12
Ή-NMR (300MHz, CDC13) : δ 1.25 (3Η, t, J= 7.1 Hz), 2.44 (3H, s), 2.71 (2H, t, J= 7.3 Hz), 3.24 (2H, t, J= 7.3 Hz), 4.15 (2H, q, J= 7.1 Hz), 7.03 (2H, m), 7.05 (2H, dd, J= 7.5, 1.4 Hz), 7.17 (5H, m) , 7.30 (1H, m), 7.32 (2H, d, J= 8.2 Hz), 7.87 (2H, d, J= 8.2 Hz), 9.63 (1H, brs) . 元素分析値: C3。H27N305S3として計算値: 59.48; H, 4.49; N, 6.94; S, Ή-NMR (300MHz, CDC1 3 ): δ 1.25 (3Η, t, J = 7.1 Hz), 2.44 (3H, s), 2.71 (2H, t, J = 7.3 Hz), 3.24 (2H, t, J = 7.3 Hz), 4.15 (2H, q, J = 7.1 Hz), 7.03 (2H, m), 7.05 (2H, dd, J = 7.5, 1.4 Hz), 7.17 (5H, m), 7.30 (1H, m) , 7.32 (2H, d, J = 8.2 Hz), 7.87 (2H, d, J = 8.2 Hz), 9.63 (1H, brs). Elemental analysis: C 3. Calculated for H 27 N 3 0 5 S 3 : 59.48; H, 4.49; N, 6.94; S,
15.88 (%)、 実測値: (:, 59.47; H, 4. 0; N, 7.03; S, 15.90 {%) . 実施例 2-13 15.88 (%), Found: (:, 59.47; H, 4.0; N, 7.03; S, 15.90 (%).
Ή-NMR (300MHz, CDC13) : 6 0.89 (3H, t, J= 6,9 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.29 (6H, brs), 1.31 (2H, br), 1. 6 (2H, brquint, J= 7.2 Hz), 2.11 (2H, dd, J= 7.9, 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.70 (2H, t, J= 7.4 Hz), 3.22 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 7.28 (2H, d, J= 8.2 Hz), 7.85 (2H, d, J= 8.2 Hz), 9.73 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): 6 0.89 (3H, t, J = 6,9 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.29 (6H, brs), 1.31 (2H, br) , 1.6 (2H, brquint, J = 7.2 Hz), 2.11 (2H, dd, J = 7.9, 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.22 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 7.28 (2H, d, J = 8.2 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.73 (1H, brs).
元素分析値: C26H35N305S3として計算値: C, 55.20; H, 6.24; N, 7.43; S, 17.00 (%), 実測値: C, 55.09; H, 6.13; N, 7.43; S, 17.29 (%) . 実施例 2-14 Elemental analysis: C 26 H 35 N 3 0 5 S 3 Calculated: C, 55.20; H, 6.24; N, 7.43; S, 17.00 (%), found: C, 55.09; H, 6.13; N, 7.43; S, 17.29 (%). Example 2-14
^-NMR (300MHz, CDC13) : <5 0.85 (3H, t, J= 6.8 Hz), 1.13 (6H, br), 1.17 (2H, m), 1.28 (3H, t, J= 7.2 Hz), 1.37 (2H, brquint, J= 7.2 Hz), 1.94 (2H, dd, J= 8.3, 7. Hz), 2.42 (3H, s), 2.74 (2H, t, J= 7.5 Hz), 3.28 (2H, t, J= 7.5 Hz), 4.18 (2H, q, J= 7.2 Hz), 7.17 (2H, dd, J = 6.8, 3.1 Hz), 7.30 (2H, d, J= 8.3 Hz), 7.39 (3H, m) , 7.85 (2H, d, J = 8.3 Hz), 9.52 (1H, brs) . ^ -NMR (300MHz, CDC1 3) : <5 0.85 (3H, t, J = 6.8 Hz), 1.13 (6H, br), 1.17 (2H, m), 1.28 (3H, t, J = 7.2 Hz), 1.37 (2H, brquint, J = 7.2 Hz), 1.94 (2H, dd, J = 8.3, 7.Hz), 2.42 (3H, s), 2.74 (2H, t, J = 7.5 Hz), 3.28 (2H, t, J = 7.5 Hz), 4.18 (2H, q, J = 7.2 Hz), 7.17 (2H, dd, J = 6.8, 3.1 Hz), 7.30 (2H, d, J = 8.3 Hz), 7.39 (3H, m), 7.85 (2H, d, J = 8.3 Hz), 9.52 (1H, brs).
元素分析値: C31H37N305S3として計算値: C, 58.47; H, 6.01; , 6.60; S, 15.10 (%) 、 実測値: C, 58.53; H, 5.86; N, 6.53; S, 15.30 ( ) . 実施例 2-15 Elemental analysis: C 31 H 37 N 3 0 5 S 3 Calculated: C, 58.47; H, 6.01;, 6.60; S, 15.10 (%), found: C, 58.53; H, 5.86; N, 6.53 S, 15.30 (). Examples 2-15
Ή-NMR (300MHz, DMS0-'d6) : 6 0.80 (3H, t, J= 7.2 Hz), 1.05-1.20 (8H, m), 1.34 (2H, brquint, J= 6.6 Hz), 1.95 (2H, dd, J= 7.5, 7.2 Hz), 2.39 (3H, s), 2.66 (2H, t, J= 7.1 Hz), 3.20 (2H, t, J= 7.1 Hz), 7.28 (2H, m), 7.35 (3H, m) , 7.39 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 11.62 (1H, br), 12.62 (1H, br). Ή-NMR (300 MHz, DMS0-'d 6 ): 6 0.80 (3H, t, J = 7.2 Hz), 1.05-1.20 (8H, m), 1.34 (2H, brquint, J = 6.6 Hz), 1.95 (2H , Dd, J = 7.5, 7.2 Hz), 2.39 (3H, s), 2.66 (2H, t, J = 7.1 Hz), 3.20 (2H, t, J = 7.1 Hz), 7.28 (2H, m), 7.35 (3H, m), 7.39 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 11.62 (1H, br), 12.62 (1H, br).
元素分析値: C29H33N305S3として計算値: C, 58.07; H, 5.55; N, 7.01; S,Elemental analysis: C 29 H 33 N 3 0 5 S 3 Calculated: C, 58.07; H, 5.55 ; N, 7.01; S,
16.04 (%;)、 実測値: 58.02; H, 5.58; N, 7.02; S, 16.22 ( ) . 実施例 2 - 16 16.04 (%;), Found: 58.02; H, 5.58; N, 7.02; S, 16.22 (). Examples 2-16
lH- NMR (300MHz, CDC13) : δ 0.81 (3Η, t, J= 6.8 Hz), 1.15 (2H, m) , 1.15 (2H, m), 1.28 (3H, t, J- 7.2 Hz), 1.37 (2H, brquint, J= 7.4 Hz), 1.94 (2H, dd, J= 7.9, 7.5 Hz), 2.42 (3H, s), 2.74 (2H, t, J= 7.2 Hz), 3.28 (2H, t, J= 7.2 Hz), 4.18 (2H, q, J= 7.2 Hz), 7.17 (2H, m) , 7.30 (2H, d, J= 8.3 Hz), 7.39 (3H, m) , 7.85 (2H, d, J= 8.3 Hz), 9.50 (1H, brs) . lH- NMR (300MHz, CDC1 3) : δ 0.81 (3Η, t, J = 6.8 Hz), 1.15 (2H, m), 1.15 (2H, m), 1.28 (3H, t, J- 7.2 Hz), 1.37 (2H, brquint, J = 7.4 Hz), 1.94 (2H, dd, J = 7.9, 7.5 Hz), 2.42 (3H, s ), 2.74 (2H, t, J = 7.2 Hz), 3.28 (2H, t, J = 7.2 Hz), 4.18 (2H, q, J = 7.2 Hz), 7.17 (2H, m), 7.30 (2H, d , J = 8.3 Hz), 7.39 (3H, m), 7.85 (2H, d, J = 8.3 Hz), 9.50 (1H, brs).
元素分析値: C29H33N305S3として計算値: C, 58.07; H, 5.55; N, 7.01; S, 16.04 (%), 実測値: C, 57.79; H, 5.39; N, 6.85; S, 16.09 (%) . 実施例 2-17 Elemental analysis: C 29 H 33 N 3 0 5 S 3 Calculated: C, 58.07; H, 5.55 ; N, 7.01; S, 16.04 (%), Found: C, 57.79; H, 5.39 ; N, 6.85; S, 16.09 (%). Example 2-17
Ή-NMR (300MHz, CDC : δ 1.27 (3Η, t, J= 7.2 Hz), 2.15 (3H, s), 2. 3 (3H, s), 2.72 (2H, t, J= 7.5 Hz), 3.24 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 4.96 (1H, s), 7.14 (2H, d, J= 8.3 Hz), 7.32 (2H, d, J= 8.3 Hz), 7.47 (2H, d, J= 8.3 Hz), 7.91 (2H, d, J= 8.3 Hz), 7.33 (1H, brs), 9.68 (1H, brs). Ή-NMR (300 MHz, CDC: δ 1.27 (3Η, t, J = 7.2 Hz), 2.15 (3H, s), 2.3 (3H, s), 2.72 (2H, t, J = 7.5 Hz), 3.24 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 4.96 (1H, s), 7.14 (2H, d, J = 8.3 Hz), 7.32 (2H, d, J = 8.3 Hz), 7.47 (2H, d, J = 8.3 Hz), 7.91 (2H, d, J = 8.3 Hz), 7.33 (1H, brs), 9.68 (1H, brs).
元素分析値: C26H26N406S3として計算値: C, 53.23; H, 4.47; N, 9.55; S, 16.40 ( ) 、 実測値: C, 52.96; H, 4.25; N, 9.55; S, 16.11 (%) . 実施例 2-18 Elemental analysis: C 26 H 26 N 4 0 6 S 3 Calculated: C, 53.23; H, 4.47 ; N, 9.55; S, 16.40 (), Found: C, 52.96; H, 4.25 ; N, 9.55 S, 16.11 (%). Example 2-18
Έ— NMR (300MHz, CDC13) : δ 0.73 (3H, t, J= 7.2 Hz), 1.18 (2H, m) , 1.24 (3H, t, J= 7.2 Hz), 1.33 (2H, m) , 2.43 (3H, s), 2.59 (2H, brt, J= 7.2 Hz), 2.68 (2H, t, J= 7.5 Hz), 3.18 (2H, t, J= 7.5 Hz), 4.14 (2H, q, J= 7.2 Hz), 7.28 (2H, m) , 7.31 (2H, d, J= 8.3 Hz), 7.40 (2H, m), 7. 0 (1H, m), 7.87 (2H, d, J= 8.3 Hz), 9.67 (1H, br). Έ- NMR (300MHz, CDC1 3) : δ 0.73 (3H, t, J = 7.2 Hz), 1.18 (2H, m), 1.24 (3H, t, J = 7.2 Hz), 1.33 (2H, m), 2.43 (3H, s), 2.59 (2H, brt, J = 7.2 Hz), 2.68 (2H, t, J = 7.5 Hz), 3.18 (2H, t, J = 7.5 Hz), 4.14 (2H, q, J = 7.2 Hz), 7.28 (2H, m), 7.31 (2H, d, J = 8.3 Hz), 7.40 (2H, m), 7.0 (1H, m), 7.87 (2H, d, J = 8.3 Hz) , 9.67 (1H, br).
元素分析値: C28H31N305S3 として計算値: C, 57.41; H, 5.33; N, 7.17Elemental analysis: C 28 H 31 N 3 0 5 S 3 Calculated: C, 57.41; H, 5.33; N, 7.17
(%) 実測値: C, 57.67; H, 5.24; , 7.18 (%) . (%) Found: C, 57.67; H, 5.24;, 7.18 (%).
実施例 2-19 Example 2-19
]H-NMR (300MHz, CDC13) : δ 0.89 (3H, brt, J= 6.4 Hz), 0.93 (3H, t, J= 7.2 Hz), 1.27 (3H, t, J= 7.2 Hz), 1.32 (6H, m) , 1.40 (4H, m) , 2.15 (2H, dd, J= 8.7, 6.8 Hz), 2.42 (3H, s), 2.70 (2H, t, J= 7.5 Hz), 2.72 (2H, brt, J- 7.5 Hz), 3.22 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.61 (1H, br). 実施例 2-20 ] H-NMR (300MHz, CDC1 3): δ 0.89 (3H, brt, J = 6.4 Hz), 0.93 (3H, t, J = 7.2 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.32 ( 6H, m), 1.40 (4H, m), 2.15 (2H, dd, J = 8.7, 6.8 Hz), 2.42 (3H, s), 2.70 (2H, t, J = 7.5 Hz), 2.72 (2H, brt , J- 7.5 Hz), 3.22 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.61 (1H, br). Example 2-20
Ή-N R (300MHz, CDC13) : δ 0.88 (3H, t, J= 6.7 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.27 (8H, brs), 1.30 (2H, m) , 1. 6 (2H, brquint, J= 7.5 Hz), 2.06 (2H, brt, J= 7.2 Hz), 2.41 (3H, s), 2.70 (2H, t, J= 7.5 Hz), 3.23 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 6.50 (1H, brt, J= 1.0 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.81 (1H, br) . 元素分析値: C26H35N305S3として計算値: C, 54.33; H, 6.31; , 7.31; S, 16.73 ( ) 、 実測値: C, 54.43; H, 6.15; N, 7.37; S, 17.19 ( ) . 実施例 2-21 Ή-NR (300MHz, CDC1 3 ): δ 0.88 (3H, t, J = 6.7 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.27 (8H, brs), 1.30 (2H, m), 1 6 (2H, brquint, J = 7.5 Hz), 2.06 (2H, brt, J = 7.2 Hz), 2.41 (3H, s), 2.70 (2H, t, J = 7.5 Hz), 3.23 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 6.50 (1H, brt, J = 1.0 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = . 8.3 Hz), 9.81 (1H , br) elemental analysis: calculated C 26 H 35 N 3 0 5 S 3: C, 54.33; H, 6.31;, 7.31; S, 16.73 (), Found: C , 54.43; H, 6.15; N, 7.37; S, 17.19 (). Examples 2-21
Ή-NMR (300MHz, CDC13) : δ 1.27 (3H, t, J= 7.2 Hz), 2.43 (3H, s), 2.74 (2H, t, J= 7.5 Hz), 3.30 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 6.93 (1H, s) , 7.31 (2H, d, J= 8.3 Hz), 7.38 (5H, s) , 7.86 (2H, d, J= 8.3 Hz), 9.82 (1H, br) . Ή-NMR (300MHz, CDC1 3 ): δ 1.27 (3H, t, J = 7.2 Hz), 2.43 (3H, s), 2.74 (2H, t, J = 7.5 Hz), 3.30 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 6.93 (1H, s), 7.31 (2H, d, J = 8.3 Hz), 7.38 (5H, s), 7.86 (2H, d, J = 8.3 Hz), 9.82 (1H, br).
元素分析値: C24H23N305S3として計算値: C, 54.42; H, 4.38; N, 7.93; S, 18.16 (%), 実測値: C, 54.22; H, 4.34; N, 7.97; S, 18.29 (%) . 実施例 2-22 Elemental analysis: C 24 H 23 N 3 0 5 S 3 Calculated: C, 54.42; H, 4.38 ; N, 7.93; S, 18.16 (%), Found: C, 54.22; H, 4.34 ; N, 7.97; S, 18.29 (%). Examples 2-22
8 - [(3-ェトキシ- 3-ォキソプロピル)スルファニル ]-1-{[(4-メチルフエ ニル)スルホニル]ィミノ }- 3 -ォキソ -2, 3 -ジヒ ド口- 1H-ィ ミダゾ [5,卜 c] [1, 4]チアジン- 5-カルボン酸 ェチル 8-[(3-Ethoxy-3-oxopropyl) sulfanyl] -1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrogen-1H-imidazo c] [1,4] thiazine-5-ethyl carboxylate
参考例 3- 2で得た化合物(純度 80%, 10.1 g, 18.8 mmol)、 3-ブロモ- 2 - ォキソプロピオン酸ェチル(3.66 g, 18.8 mmol)およびトリエチルアミン (7.81 ml, 56.3 mmo 1)の DMF- THF (1:4, 200 ml)懸濁液を室温で 50分間 攪拌した。 反応液を濃縮し、 酢酸ェチルで抽出した。 有機層を 5¾炭酸水 素ナトリゥム水溶液および飽和食塩水で洗浄後、 硫酸ナトリゥムで乾燥 し、 濃縮して褐色固形物(11 g)を得た。 Of the compound obtained in Reference Example 3-2 (purity 80%, 10.1 g, 18.8 mmol), ethyl 3-bromo-2-oxopropionate (3.66 g, 18.8 mmol) and triethylamine (7.81 ml, 56.3 mmo 1) The suspension of DMF-THF (1: 4, 200 ml) was stirred at room temperature for 50 minutes. The reaction solution was concentrated and extracted with ethyl acetate. The organic layer was washed with a 5% aqueous solution of sodium hydrogen carbonate and saturated saline, dried over sodium sulfate, and concentrated to obtain a brown solid (11 g).
得られた褐色固形物(11 g)、無水酢酸(5.42 ml, 56.3 mmol)および p- TsOH 水和物(3.58 g, 18.8腿 ol)のトルエン(200 ml)懸濁液を 80 で 3時間 攪拌した。 反応液を濃縮し、 酢酸ェチルで抽出した。 有機層を 5%炭酸水 素ナトリウム水溶液および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥 し、 濃縮した。 残渣にメタノールを加え、 析出物を濾取し、 メタノール で洗浄、 乾燥して標題化合物 (4.86 g, 9.23 mmol)を赤紫色粉末として 得た。 A suspension of the obtained brown solid (11 g), acetic anhydride (5.42 ml, 56.3 mmol) and p-TsOH hydrate (3.58 g, 18.8 tmol) in toluene (200 ml) was stirred at 80 for 3 hours. did. The reaction solution was concentrated and extracted with ethyl acetate. The organic layer is washed with a 5% aqueous sodium hydrogen carbonate solution and saturated saline, and then dried over sodium sulfate. And concentrated. Methanol was added to the residue, and the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (4.86 g, 9.23 mmol) as a red-purple powder.
Ή-N R (300MHz, CDC13) : δ 1.27 (3Η, t, J= 7.2 Hz) , 1.32 (3H, t, J = 7.1 Hz), 2.43 (3H, s), 2.69 (2H, t, J= 7.5 Hz), 3.21 (2H, t, J= 7.5 Hz), 4.16 (2H, q, J= 7.2 Hz), 4.31 (2H, q, J- 7.2 Hz), 5.89 (1H, s), 7.31 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.72 (1H, brs). 元素分析値: C21H23N307S3として計算値: C, 47.99; H, 4.41; N, 7.99; S, 18.30 (%), 実測値: C, 47.87; H, 4.47; N, 8.01; S, 18.10 (%) . mp. 130.0-131.5 Ή-NR (300MHz, CDC1 3 ): δ 1.27 (3Η, t, J = 7.2 Hz), 1.32 (3H, t, J = 7.1 Hz), 2.43 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.21 (2H, t, J = 7.5 Hz), 4.16 (2H, q, J = 7.2 Hz), 4.31 (2H, q, J- 7.2 Hz), 5.89 (1H, s), 7.31 (2H , d, J = 8.3 Hz) , 7.84 (2H, d, J = 8.3 Hz), 9.72 (1H, brs) elemental analysis: calculated C 21 H 23 N 3 0 7 S 3: C, 47.99;. H, 4.41; N, 7.99; S, 18.30 (%), Found: C, 47.87; H, 4.47; N, 8.01; S, 18.10 (%). Mp.130.0-131.5
実施例 2-23 Example 2-23
Ή-NMR (300MHz, CDC13) : δ 0.92 (3H, t, J= 7.2 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.36 (2H, quint, J= 7. Hz), 1.45 (2H, quint, J= 7.2 Hz), 2.07 (2H, brt, J= 7.0 Hz), 2.41 (3H, s), 2.70 (2H, t, J= 7.3 Hz), 3.23 (2H, t, J= 7.3 Hz), 4.17 (2H, q, J= 7.1 Hz), 6.51 (1H, s) , 7.29 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.80 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.92 (3H, t, J = 7.2 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.36 (2H, quint, J = 7. Hz), 1.45 ( 2H, quint, J = 7.2 Hz), 2.07 (2H, brt, J = 7.0 Hz), 2.41 (3H, s), 2.70 (2H, t, J = 7.3 Hz), 3.23 (2H, t, J = 7.3) Hz), 4.17 (2H, q, J = 7.1 Hz), 6.51 (1H, s), 7.29 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.80 (1H, br).
元素分析値: C22H27N305S3 · H20として計算値: C, 50.08; H, 5.54; N, 7.96; S, 18.22 (%) 、 実測値: 9.66; H, 5.22; N, 7.70; S, 18.08 ( ) . 実施例 2-24 Elemental analysis: C 22 H 27 N 3 0 5 S 3 · H 2 0 Calculated: C, 50.08; H, 5.54 ; N, 7.96; S, 18.22 (%), Found: 9.66; H, 5.22; N, 7.70; S, 18.08 (). Examples 2-24
Ή-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J- 6.9 Hz), 1.26 (16H, brs), 1.27 (3H, t, J= 7.1 Hz), 1. 6 (2H, brquint, J= 6.8 Hz), 2.07 (2H, brt, J二 7.2 Hz), 2. 2 (3H, s), 2.70 (2H, t, J= 7.4 Hz), 3.23 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 6.50 (1H, brt, J= 1 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.79 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3Η, t, J- 6.9 Hz), 1.26 (16H, brs), 1.27 (3H, t, J = 7.1 Hz), 1. 6 (2H, brquint, J = 6.8 Hz), 2.07 (2H, brt, J2 7.2 Hz), 2.2 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.23 (2H, t, J = 7.4 Hz) , 4.17 (2H, q, J = 7.1 Hz), 6.50 (1H, brt, J = 1 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.79 (1H, br).
元素分析値: C3。H43N305S3 · 0.4H20として計算値: C, 57.28; H, 6.89; N, 6.68; S, 15.29 (%), 実測値: C, 57.28; H, 7.00; N, 6.64; S, 15.20Elemental analysis: C 3. H 43 N 3 0 5 S 3 · 0.4H 2 0 Calculated: C, 57.28; H, 6.89; N, 6.68; S, 15.29 (%), Found: C, 57.28; H, 7.00; N, 6.64 ; S, 15.20
(%) . (%).
実施例 2-25 Example 2-25
Ή-NMR (300MHz, CDC13) : δ 1.26 (3H, t, J= 7.2 Hz), 2.27 (3H, d, J = 1.1 Hz), 2.42 (3H, s), 2.69 (2H, t, J= 7.5 Hz), 3.18 (2H, t, J= 7.5 Hz), 4.16 (2H, q, J= 7.2 Hz), 4.63 (1H, q, J= 1.1 Hz), 7.29 (2H, d, J= 8.2 Hz), 7.84 (2H, d, J= 8.2 Hz), 9.71 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3H, t, J = 7.2 Hz), 2.27 (3H, d, J = 1.1 Hz), 2.42 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.18 (2H, t, J = 7.5 Hz), 4.16 (2H, q, J = 7.2 Hz), 4.63 (1H , q, J = 1.1 Hz), 7.29 (2H, d, J = 8.2 Hz), 7.84 (2H, d, J = 8.2 Hz), 9.71 (1H, br).
元素分析値: C19H21N305S3として計算値: C, 48.81; H, 4.53; N, 8.99; S, 20.57 ( ), 実測値: C, 48.52; H, 4.51; N, 9.00; S, 20.69 (%) . 実施例 2-26 Elemental analysis: C 19 H 21 N 3 0 5 S 3 Calculated: C, 48.81; H, 4.53; N, 8.99; S, 20.57 (), found: C, 48.52; H, 4.51; N, 9.00 ; S, 20.69 (%). Example 2-26
Ή-NMR (300MHz, CDC13) : δ 1.27 (3Η, t, J- 7.2 Hz), 1.80 (2H, quint, J= 7.2 Hz), 2.19 (2H, brdd, J= 7.9, 7.5 Hz), 2.27 (3H, s), 2.36 (2H, t, J= 7.2 Hz), 2.41 (3H, s), 2.69 (2H, t, J= 7.5 Hz), 3.21 (2H, t, J= 7.5 Hz), 3.68 (3H, s), 4.16 (2H, q, J= 7.2 Hz), 7.28 (2H, brd, J = 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.76 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 1.27 (3Η, t, J- 7.2 Hz), 1.80 (2H, quint, J = 7.2 Hz), 2.19 (2H, brdd, J = 7.9, 7.5 Hz), 2.27 (3H, s), 2.36 (2H, t, J = 7.2 Hz), 2.41 (3H, s), 2.69 (2H, t, J = 7.5 Hz), 3.21 (2H, t, J = 7.5 Hz), 3.68 (3H, s), 4.16 (2H, q, J = 7.2 Hz), 7.28 (2H, brd, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.76 (1H, br).
元素分析値: C24H29N307S3として計算値: C, 50.78 ; H, 5.15; N, 7.40; S, 16.94 (%) 、 実測値: C, 50.75; H, 5.21; , 7.45; S, 17.01 (%) . 実施例 2-27 Elemental analysis: C 24 H 29 N 3 0 7 S 3 Calculated: C, 50.78; H, 5.15 ; N, 7.40; S, 16.94 (%), Found: C, 50.75; H, 5.21 ;, 7.45 ; S, 17.01 (%). Example 2-27
Ή-NMR (300MHz, CDC13) : δ 1.26 (3H, t, J= 7.1 Hz), 2.41 (2H, m) , 2.41 (3H, s), 2.51 (2H, brt, J= 6.4 Hz), 2.70 (2H, t, J= 7.4 Hz), 3.22 (2H, t, J= 7.4 Hz), 3.70 (3H, s), 4.17 (2H, q, J= 7.1 Hz), 6.57 (1H, brs), 7.28 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.89 (1H, br). 元素分析値: C22H25N307S3として計算値: C, 48.97; H, 4.67; N, 7.79; S, 17.83 (%), 実測値: C, 48.67; H, 4.60; N, 7.59; S, 17.94 (%) . 実施例 2-28 Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3H, t, J = 7.1 Hz), 2.41 (2H, m), 2.41 (3H, s), 2.51 (2H, brt, J = 6.4 Hz), 2.70 (2H, t, J = 7.4 Hz), 3.22 (2H, t, J = 7.4 Hz), 3.70 (3H, s), 4.17 (2H, q, J = 7.1 Hz), 6.57 (1H, brs), 7.28 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.89 (1H, br) elemental analysis:. C 22 H 25 N 3 0 7 S 3 calculated: C, 48.97; H, 4.67; N, 7.79; S, 17.83 (%), Found: C, 48.67; H, 4.60; N, 7.59; S, 17.94 (%). Example 2-28
Ή-NMR (300MHz, CDC13) : δ 1.26 (3Η, t, J= 7.1 Hz), 2.41 (2H, m), 2.41 (3H, s), 2.56 (2H, brt, J= 6.6 Hz), 2.70 (2H, t, J= 7.4 Hz), 3.22 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 6.61 (1H, brs), 7.29 (2H, d, ]= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.96 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3Η, t, J = 7.1 Hz), 2.41 (2H, m), 2.41 (3H, s), 2.56 (2H, brt, J = 6.6 Hz), 2.70 (2H, t, J = 7.4 Hz), 3.22 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 6.61 (1H, brs), 7.29 (2H, d,] = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.96 (1H, br).
元素分析値: C21H23N307S3として計算値: C, 47.99; H, 4.41; N, 7.99; S, 18.30 (%)、 実測値: C, 47.83; H, 4.33; N, 8.01; S, 18.55 (%) . 実施例 2 - 29 Elemental analysis: C 21 H 23 N 3 0 7 S 3 Calculated: C, 47.99; H, 4.41; N, 7.99; S, 18.30 (%), found: C, 47.83; H, 4.33; N, 8.01; S, 18.55 (%). Example 2-29
Ή-NMR (300MHz, CDC13) : δ 1.24 (3Η, t, J= 7.1 Hz), 2.40 (3H, s), 2.62 (2H, brt, J= 7.3 Hz), 3.15 (2H, t, J= 7.3 Hz), 3.36 (2H, brs), 4.14 (2H, q, J= 7.1 Hz), 6.58 (1H, brt, J= 1 Hz), 7.20 (2H, bfd, J = 7.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.30 (1H, m) , 7.32 (2H, brt, J= 7.3 Hz), 7.83 (2H, d, J= 8.3 Hz), 9.82 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 1.24 (3Η, t, J = 7.1 Hz), 2.40 (3H, s), 2.62 (2H, brt, J = 7.3 Hz), 3.15 (2H, t, J = 7.3 Hz), 3.36 (2H, brs), 4.14 (2H, q, J = 7.1 Hz), 6.58 (1H, brt, J = 1 Hz), 7.20 (2H, bfd, J = 7.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.30 (1H, m), 7.32 (2H, brt, J = 7.3 Hz), 7.83 ( 2H, d, J = 8.3 Hz), 9.82 (1H, br).
元素分析値: C25H25N305S3として計算値: C, 55.23; H, 4.63; , 7.73; S, 17.69 (%) 、 実測値: C, 55.04; H, 4.73; N, 7.66; S, 17.82 (%) . 実施例 2 - 30 Elemental analysis: C 25 H 25 N 3 0 5 S 3 Calculated: C, 55.23; H, 4.63;, 7.73; S, 17.69 (%), found: C, 55.04; H, 4.73; N, 7.66 ; S, 17.82 (%). Example 2-30
Ή-NMR (300MHz, CDC13) : 6 1.26 (3H, t, J= 7.1 Hz), 2.42 (3H, s) , 2.70 (2H, t, J= 7.4 Hz), 3.20 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.1 Hz) , 5.02 (1H, t, J= 8.0 Hz), 6.66 (1H, d, J= 8.0 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.83 (2H, d, J= 8.3 Hz), 9.82 (1H, br). Ή-NMR (300MHz, CDC1 3 ): 6 1.26 (3H, t, J = 7.1 Hz), 2.42 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.20 (2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.1 Hz), 5.02 (1H, t, J = 8.0 Hz), 6.66 (1H, d, J = 8.0 Hz), 7.29 (2H, d, J = 8.3 Hz) ), 7.83 (2H, d, J = 8.3 Hz), 9.82 (1H, br).
元素分析値: C18H19N303S3として計算値: C, 47.67; H, 4.22; N, 9.26; S, 21.21 (%)、 実測値: C, 47.48; H, 4.16; N, 9.13; S, 21.42 (%) . 実施例 2 - 31 Elemental analysis: C 18 H 19 N 3 0 3 S 3 Calculated: C, 47.67; H, 4.22 ; N, 9.26; S, 21.21 (%), Found: C, 47.48; H, 4.16 ; N, 9.13; S, 21.42 (%). Examples 2-31
Ή-NMR (300MHz, DMS0-d6) : δ 1.18 (3Η, t, J= 7.1 Hz), 2.39 (3H, s), 2.50 (2H, t, J= 6.4 Hz), 2.63 (2H, t, J= 7.0 Hz), 3.09 (2H, t, J= 7.0 Hz), 3.73 (2H, brt, J= 6.4 Hz), 4.08 (2H, q, J- 7.1 Hz), 6.71 (1H, brs), 7.37 (2H, d, J= 8.2 Hz), 7.80-7.90 (6H, m) , 11.80 (1H, br). 元素分析値: C28H26N407S3として計算値: 53.66; H, 4.18; N, 8.94; S, 15.35 (%), 実測値: 53.48; H, 4.21; N, 8.77; S, 15.34 ( ) . 実施例 2-32 Ή-NMR (300 MHz, DMS0-d 6 ): δ 1.18 (3Η, t, J = 7.1 Hz), 2.39 (3H, s), 2.50 (2H, t, J = 6.4 Hz), 2.63 (2H, t, J = 7.0 Hz), 3.09 (2H, t, J = 7.0 Hz), 3.73 (2H, brt, J = 6.4 Hz), 4.08 (2H, q, J- 7.1 Hz), 6.71 (1H, brs), 7.37 . (2H, d, J = 8.2 Hz), 7.80-7.90 (6H, m), 11.80 (1H, br) elemental analysis: C 28 H 26 N 4 0 7 S 3 calculated: 53.66; H, 4.18 N, 8.94; S, 15.35 (%), Found: 53.48; H, 4.21; N, 8.77; S, 15.34 (). Examples 2-32
Ή-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.8 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.29 (2H, brs), 1.31 (2H, m) , 1.47 (2H, brquint, J= 7.4 Hz), 2.07 (2H, brt, J= 7.5 Hz), 2.42 (3H, s), 2.70 (2H, t, J= 7.4 Hz), 3.23 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 6.50 (1H, brt, J= 0.5 Hz) , 7.29 (2H, d, 8.3 Hz), 7.83 (2H, d, J= 8.3 Hz), 9.70 (1H, br) . 元素分析値 : C23H29N305S3 · 0.3H20として計算値: C, 52.21; H, 5.64; N, 7.94; S, 18.18 (%) 、 実測値: C, 52.28 ; H, 5.53; N, 7.98; S, 18.12 (%) . 実施例 2-33 Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 6.8 Hz), 1.27 (3H, t, J = 7.1 Hz), 1.29 (2H, brs), 1.31 (2H, m), 1.47 (2H, brquint, J = 7.4 Hz), 2.07 (2H, brt, J = 7.5 Hz), 2.42 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.23 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 6.50 (1H, brt, J = 0.5 Hz), 7.29 (2H, d, 8.3 Hz), 7.83 (2H, d, J = 8.3 Hz), 9.70 (1H, br). Elemental analysis: C 23 H 29 N 3 0 5 S 3 · 0.3H 2 0 Calculated: C, 52.21; H, 5.64; N, 7.94; S, 18.18 (%), found Values: C, 52.28; H, 5.53; N, 7.98; S, 18.12 (%). Example 2-33
Ή-N R (300MHz, CDC13) : δ 0.89 (3H, brt, J= 6.9 Hz), 1.27 (3H, t, J= 7.2 Hz), 1.28 (8H, brs), 1.46 (2H, brquint, J= 6.8 Hz), 2.06 (2H, brt, J= 7.3 Hz), 2.41 (3H, s), 2.70 (2H, t, J= 7.4 Hz), 3.23 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 6.50 (1H, brs), 7.29 (2H, d, J = 8.3 Hz), 7.83 (2H, d, J= 8.3 Hz), 9.79 (1H, brs). Ή-NR (300MHz, CDC1 3 ): δ 0.89 (3H, brt, J = 6.9 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.28 (8H, brs), 1.46 (2H, brquint, J = 6.8 Hz), 2.06 (2H, brt, J = 7.3 Hz), 2.41 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.23 (2H, t, J = 7.4 Hz), 4.17 (2H , q, J = 7.1 Hz), 6.50 (1H, brs), 7.29 (2H, d, J = 8.3 Hz), 7.83 (2H, d, J = 8.3 Hz), 9.79 (1H, brs).
元素分析値: C25H33N305S3として計算値: C, 54.42; H, 6.03; N, 7.62; S, 17.43 (%), 実測値: 54.14; H, 5.78; N, 7. 4; S, 17.30 (%) . 実施例 2 - 34 Elemental analysis: C 25 H 33 N 3 0 5 S 3 Calculated: C, 54.42; H, 6.03 ; N, 7.62; S, 17.43 (%), Found: 54.14; H, 5.78; N , 7. 4; S, 17.30 (%). Example 2-34
Ή-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.8 Hz), 1.26 (3H, t, J = 7.1 Hz), 1. 9 (6H, m) , 1. 4 (2H, brqiunt, J= 7.2 Hz), 2.11 (2H, brdd, J- 7.9, 7.2 Hz) , 2.26 (3H, s), 2.40 (3H, s), 2.70 (2H, t, J= 7.4 Hz), 3.21 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.1 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.84 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3Η, t, J = 6.8 Hz), 1.26 (3H, t, J = 7.1 Hz), 1. 9 (6H, m), 1. 4 (2H, brqiunt, J = 7.2 Hz), 2.11 (2H, brdd, J-7.9, 7.2 Hz), 2.26 (3H, s), 2.40 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.21 ( 2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.1 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.84 (1H, br).
元素分析値: C25H33N305S3として計算値: C, 54.42; H, 6.03; N, 7.62; S, 17.43 (%), 実測値: C, 54.27; H, 5.96; N, 7.76; S, 17.33 (%) . 実施例 2 - 35 Elemental analysis: Calculated as C 25 H 33 N 3 0 5 S 3: C, 54.42; H, 6.03; N, 7.62; S, 17.43 (%), Found: C, 54.27; H, 5.96 ; N, 7.76; S, 17.33 (%). Examples 2-35
Ή-NMR (300MHz, CDC13) : δ 1.26 (3Η, t, J= 7.2 Hz), 2.42 (3H, s), 2.68 (2H, t, J= 7.1 Hz), 3.26 (2H, t, J= 7.1 Hz), 3.81 (2H, s), 4.16 (2H, q, J= 7.2 Hz), 6.90 (1H, s), 7.31 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.76 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3Η, t, J = 7.2 Hz), 2.42 (3H, s), 2.68 (2H, t, J = 7.1 Hz), 3.26 (2H, t, J = 7.1 Hz), 3.81 (2H, s), 4.16 (2H, q, J = 7.2 Hz), 6.90 (1H, s), 7.31 (2H, d, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.76 (1H, brs).
元素分析値: C19H2。N305S3C1として計算値: C, 45.46; H, 4.02; N, 8.37; S, 19.16 (%) 、 実測値: C, 45.76; H, 3.90; N, 8.28; S, 19.12 (%) . 実施例 2-36 Elemental analysis: C 19 H 2. Calculated for N 3 0 5 S 3 C1: C, 45.46; H, 4.02; N, 8.37; S, 19.16 (%), found: C, 45.76; H, 3.90; N, 8.28; S, 19.12 (% Example 2-36
Ή-NMR (300MHz, CDC13) : δ 1. 7 (3H, t, J= 7.2 Hz), 2.43 (3H, s), 2.72 (2H, t, J= 7.3 Hz), 3.24 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.2 Hz), 4.95 (1H, s), 6.94 (2H, dt, J= 8.7, 2.3 Hz), 7.05 (2H, dq, J = 8.3, 1.1 Hz), 7.09 (2H, d, J= 8.7 Hz), 7.10 (1H, tq, J= 6.0, 0.7 Hz), 7.31 (2H, d, J= 8.3 Hz), 7.36 (2H, brt, J= 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.68 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 1. 7 (3H, t, J = 7.2 Hz), 2.43 (3H, s), 2.72 (2H, t, J = 7.3 Hz), 3.24 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.2 Hz), 4.95 (1H, s), 6.94 (2H, dt, J = 8.7, 2.3 Hz), 7.05 (2H, dq, J = 8.3, 1.1 Hz), 7.09 (2H, d, J = 8.7 Hz), 7.10 (1H, tq, J = 6.0, 0.7 Hz), 7.31 (2H, d, J = 8.3 Hz), 7.36 (2H, brt, J = 8.3) Hz), 7.86 (2H, d, J = 8.3 Hz), 9.68 (1H, brs).
元素分析値: C3。H27N306S3として計算値: C, 57.95; H, 4.38; , 6.76; S, 15. 7 (%), 実測値: C, 58.22; H, 4.34; N, 6.58; S, 15.37 (%) . 実施例 2-37 Elemental analysis: C 3. Calcd H 27 N 3 0 6 S 3 : C, 57.95; H, 4.38;, 6.76; S, 15. 7 (%), Found: C, 58.22; H, 4.34 ; N, 6.58; S, 15.37 Example 2-37
'Η-丽 R (300 MHz, 重クロ口ホルム中): δ 2.27 (3Η, d, J= 1.2 Hz), 2.42 (3H, s), 2.71 (2H, t, J= 7.5 Hz), 3.18 (2H, t, J= 7.6 Hz), 3.71 (3H, s), 4.63 (1H, q, J= 1.3 Hz), 7.30 (2H, d, J= 8.2 Hz), 7.84 (2H, d, 8.3 Hz), 9.67 (1H, brs).元素分析値: C18H19N305S3 として計算値: C, 47.67; H, 4.22; N, 9.26; S, 21.21 (%), 実測値; C, 47.38; H, 4.26; N, 9.09; S, 21.35 ( ) . 'Η- 丽 R (300 MHz, in double-mouthed form): δ 2.27 (3Η, d, J = 1.2 Hz), 2.42 (3H, s), 2.71 (2H, t, J = 7.5 Hz), 3.18 ( 2H, t, J = 7.6 Hz), 3.71 (3H, s), 4.63 (1H, q, J = 1.3 Hz), 7.30 (2H, d, J = 8.2 Hz), 7.84 (2H, d, 8.3 Hz) , 9.67 (1H, brs) elemental analysis: calculated C 18 H 19 N 3 0 5 S 3:. C, 47.67; H, 4.22; N, 9.26; S, 21.21 (%), Found; C, 47.38; H, 4.26; N, 9.09; S, 21.35 ().
実施例 -38 Example -38
Ή-NMR (CDC13, 300 MHz): δ 1.79 (3H, s), 2.26 (3H, d, J= 0.8 Hz), 2.42 (3H, s), 2.71 (2H, t, J= 7.5 Hz), 3.21 (2H, t, J= 7.6 Hz), 3.72 (3H, s), 7.29 (2H, d, J= 8.2 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Ή-NMR (CDC1 3, 300 MHz): δ 1.79 (3H, s), 2.26 (3H, d, J = 0.8 Hz), 2.42 (3H, s), 2.71 (2H, t, J = 7.5 Hz), 3.21 (2H, t, J = 7.6 Hz), 3.72 (3H, s), 7.29 (2H, d, J = 8.2 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C19H21N305S3として計算値: C, 48.80; H, 4.53; N, 8.99; S, 20.57 (%), 実測値: C, 48.55; H, 4. 7; N, 9.09; S, 20.64 (%) . 実施例 2 - 39 Elemental analysis: Calculated as C 19 H 21 N 3 0 5 S 3: C, 48.80; H, 4.53; N, 8.99; S, 20.57 (%), Found: C, 48.55; H, 4. 7; N, 9.09; S, 20.64 (%). Example 2-39
Ή-NMR (CDC13, 300 MHz): δ 1.27 (3Η, ί, J= 7.2 Hz), 2.41 (3H, s), 2.63 (3H, s), 2.72 (2H, t, J= 7.5 Hz), 3.29 (2H, t, J= 7.5 Hz), 4.17 (2H, q, J- 7.2 Hz), 7.30 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.80 (1H, br). Ή-NMR (CDC1 3, 300 MHz): δ 1.27 (3Η, ί, J = 7.2 Hz), 2.41 (3H, s), 2.63 (3H, s), 2.72 (2H, t, J = 7.5 Hz), 3.29 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J-7.2 Hz), 7.30 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.80 ( 1H, br).
元素分析値: C21H23N306S3として計算値: C, 49.49; H, 4.55; N, 8.25; S, 18.88 (%), 実測値: C, 49.17; H, 4.50; N, 8.26; S, 18.78 (%) . 実施例 2-40 Elemental analysis: Calculated for C 21 H 23 N 3 0 6 S 3 : C, 49.49; H, 4.55; N, 8.25; S, 18.88 (%), found: C, 49.17; H, 4.50; N, 8.26; S, 18.78 (%). Examples 2-40
Ή-NMR (CDC13, 200 MHz): δ 1.10 (6Η, d, J= 7.0 Hz), 1.27 (3H, t, J= 7.0 Hz), 2.28 (3H, s) , 2.42 (3H, s) , 2.71 (2H, t, J= 7.2 Hz), 2.96 (1H, quint, J= 6.6 Hz), 3.25 (2H, t, J= 7.2 Hz), 4.17 (2H, q, J= 7.4 Hz), 7.29 (2H, d, J= 7.8 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.64 (1H, br). 元素分析値: C22H27N305S3として計算値: C, 51.84; H, 5.34; N, 8.24; S, 18.87 (%), 実測値: 51.62; H, 5.44; N, 8.26; S, 18.99 (%) . 実施例 -41 Ή-NMR (CDC1 3, 200 MHz): δ 1.10 (6Η, d, J = 7.0 Hz), 1.27 (3H, t, J = 7.0 Hz), 2.28 (3H, s), 2.42 (3H, s), 2.71 (2H, t, J = 7.2 Hz), 2.96 (1H, quint, J = 6.6 Hz), 3.25 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J = 7.4 Hz), 7.29 ( 2H, d, J = 7.8 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.64 (1H, br). Elemental analysis: C 22 H 27 N 3 0 5 S 3 Calculated: C, 51.84; H, 5.34 ; N, 8.24; S, 18.87 (%), Found: 51.62; H, 5.44; N , 8.26; S, 18.99 (%). Example -41
酢酸ェチルから再結晶して融点 138- 140°Cの暗赤色結晶を得た。 Recrystallization from ethyl acetate gave dark red crystals with a melting point of 138-140 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3280, 2961 , 2930, 2872, 1732, 1657, 1559 cm"1. Infrared absorption spectrum (IR) (in KBr): 3280, 2961, 2930, 2872, 1732, 1657, 1559 cm " 1 .
^-NMR (CDClg) : δ 0.950 (3Η, t, J = 7.0 Hz), 1.27 (3H, t, J = 7.0 Hz), 1.44-1.60 (2H, m), 2.11 (2H, t, J = 7.2 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J = 7.6 Hz), 3.21 (2H, t, J = 7.8 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs) . 元素分析値: C22H27N305S3として ^ -NMR (CDClg): δ 0.950 (3Η, t, J = 7.0 Hz), 1.27 (3H, t, J = 7.0 Hz), 1.44-1.60 (2H, m), 2.11 (2H, t, J = 7.2 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J = 7.6 Hz), 3.21 (2H, t, J = 7.8 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.29 (2H, d , J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs) Elementary analysis:. as C 22 H 27 N 3 0 5 S 3
計算値 ) : C, 51.84; H, 5.34; N, 8.24; S, 18.87. Calculated)): C, 51.84; H, 5.34; N, 8.24; S, 18.87.
実測値 (¾) : C, 51.70; H, 5.21; N, 8.12; S, 18.72. Found (¾): C, 51.70; H, 5.21; N, 8.12; S, 18.72.
実施例 2-42 Example 2-42
酢酸ェチル -ジェチルェ一テルから再結晶して融点 139-140°Cの暗赤色 結晶を得た。 Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 139-140 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3248, 2957, 2934, 2870, 1732, 1657, 1615, 1564, 1495 cm"1. Infrared absorption spectrum (IR) (in KBr): 3248, 2957, 2934, 2870, 1732, 1657, 1615, 1564, 1495 cm " 1 .
Ή-NMR (CDC13) : δ 0.946 (6H, d, J = 6.4 Hz), 1.27 (3H, t, J = 7.4 Hz), 1.71-1.90 (1H, m) , 2.03 (2H, d, J = 7.0 Hz), 2.27 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.8 Hz), 4.17 (2H, q, J = 7.0 Hz), 7.29 (2H, d, J = 9.2 Hz), 7.84 (2H, d, J = 8.0 Hz), 9.63 (1H, bs). 元素分析値: C23H29N305S3として Ή-NMR (CDC1 3): δ 0.946 (6H, d, J = 6.4 Hz), 1.27 (3H, t, J = 7.4 Hz), 1.71-1.90 (1H, m), 2.03 (2H, d, J = 7.0 Hz), 2.27 (3H, s), 2.42 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.8 Hz), 4.17 (2H, q, J = 7.0 Hz), 7.29 (2H, d, J = 9.2 Hz), 7.84 (2H, d, J = 8.0 Hz), 9.63 (1H, bs). Elemental analysis: C 23 H 29 N 3 0 5 S 3
計算値 ( : C, 52.75; H, 5.58; N, 8.02; S, 18.37. , 実測値 ) : C, 52.75; H, 5.55; N, 7.87; S, 18.36. Calculated value (: C, 52.75; H, 5.58; N, 8.02; S, 18.37., Measured value): C, 52.75; H, 5.55; N, 7.87; S, 18.36.
実施例 2 - 43 Example 2-43
- NMR (200MHz, CDC13) : δ 1.08 (3Η, t, J- 7.3 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.82 (3H, s), 2. 2 (3H, s), 2.70 (2H, t, J= 7.3 Hz), 2.78 (2H, q, J= 7.3 Hz), 3.21 (2H, t, J二 7.5 Hz), 4.17 (2H, q, J= 7.2 Hz), 7.29 (2H, d, 8.4 Hz), 7.84 (2H, dt, J= 8.4, 1.8 Hz), 9.63 (1H, brs). 元素分析値: C21H25N305S3として計算値: C, 50.89; H, 5.08; N, 8.48; S, 19. 1 (%) 、 実測値: C, 50.62; H, 5.08; N, 8.46; S, 19.40 (%) . 融点. 148.5-150.0°C - NMR (200MHz, CDC1 3) : δ 1.08 (3Η, t, J- 7.3 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.82 (3H, s), 2. 2 (3H, s), 2.70 (2H, t, J = 7.3 Hz), 2.78 (2H, q, J = 7.3 Hz), 3.21 (2H, t, J = 7.5 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.29 (2H, d, 8.4 Hz), 7.84 (2H, dt, J = 8.4, 1.8 Hz), 9.63 (1H, brs). Elemental analysis: C 21 H 25 N 3 0 5 S 3 Calculated: C , 50.89; H, 5.08; N, 8.48; S, 19.1 (%), found: C, 50.62; H, 5.08; N, 8.46; S, 19.40 (%). Melting point. 148.5-150.0 ° C
実施例 2 - 44 Example 2-44
酢酸ェチル -ジェチルェ一テルから再結晶して融点 1-143°Cの暗赤色 結晶を得た。 The crystal was recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 1-143 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3243, 3063, 3029, 2980, 1732, 1657, 1620, 1564, 1495 cm—1. Infrared absorption spectrum (IR) (in KBr) Re: 3243, 3063, 3029, 2980, 1732, 1657, 1620, 1564, 1495 cm- 1 .
Ή-NMR (CDC13) : δ 1.25 (3Η, t, J = 7.0 Hz), 2.37 (3H, s) , 2.42 (3H, s), 2.58 (2H, t, J = 7.8 Hz), 3.12 (2H, t, J = 7.4 Hz), 3.50 (2H, s), 4.13 (2H, q, J = 7.2 Hz), 7.14-7. 4 (7H, m), 7.84 (2H, d, J-8.4 Hz) , 9.67 (1H, bs). Ή-NMR (CDC1 3): δ 1.25 (3Η, t, J = 7.0 Hz), 2.37 (3H, s), 2.42 (3H, s), 2.58 (2H, t, J = 7.8 Hz), 3.12 (2H , t, J = 7.4 Hz), 3.50 (2H, s), 4.13 (2H, q, J = 7.2 Hz), 7.14-7.4 (7H, m), 7.84 (2H, d, J-8.4 Hz) , 9.67 (1H, bs).
元素分析値: C26H27N3O5S3'0.3H2Oとして Elemental analysis: as C 26 H 27 N 3 O 5 S 3 '0.3H 2 O
計算値 (%) : C, 55.46; H, 4.94; N, 7. 6; S, 17.08. Calculated value (%): C, 55.46; H, 4.94; N, 7.6; S, 17.08.
実測値 (%) : C, 55.54; H, 5.07; N, 7.39; S, 17.23. Found (%): C, 55.54; H, 5.07; N, 7.39; S, 17.23.
実施例 2-45 Example 2-45
8 - [(3-ェトキシ- 3-ォキソプロピル)スルファニル ]-6-メチル -1-{ [(4 -メ チルフエニル)スルホニル]イミノ卜 3-ォキソ -2, 3 -ジヒドロ- 1H -ィミダ ゾ [5, 1-c] [1, 4]チアジン- 5-カルボン酸 ェチル 8-[(3-Ethoxy-3-oxopropyl) sulfanyl] -6-methyl-1-{[(4-methylphenyl) sulfonyl] iminoto 3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-5-carboxylate
粗製のェチル 2-ォキソ酪酸 (7.8 g, 35.7 mmol)のクロ口ホルム(100 ml)溶液に、 0°Cで攪拌しながら三臭化トリメチルフエ二ルアンモニゥム (13.7 g, 35.7 mmol).を加えた。 反応液を 20分間かけて室温まで戻して 室温で 70分間攪拌した。反応液をジィソプロピルエーテルで抽出し、 粗 製の 3-プロモ- 2_ォキソ酪酸ェチル(純度 60¾, 6.5 g, 約 18.7 fflfflol)を 黄色油状物として得た。 . To a solution of the crude ethyl 2-oxobutyric acid (7.8 g, 35.7 mmol) in chloroform (100 ml) was added trimethylphenylammonium tribromide (13.7 g, 35.7 mmol) while stirring at 0 ° C. . The reaction solution was returned to room temperature over 20 minutes and stirred at room temperature for 70 minutes. The reaction solution was extracted with diisopropyl ether to obtain crude ethyl 3-bromo-2-oxobutyrate (purity 60¾, 6.5 g, about 18.7 fflfflol) as a yellow oil. .
JH-NMR (200MHz, CDC13): δ 1.40 (3Η, t, J= 7.1 Hz), 1.82 (3H, d, J二 6.8 Hz), 4.39 (2H, q, J= 7.1 Hz), 5.18 (1H, q, J= 6.8 Hz). J H-NMR (200MHz, CDC1 3): δ 1.40 (3Η, t, J = 7.1 Hz), 1.82 (3H, d, J two 6.8 Hz), 4.39 (2H, q, J = 7.1 Hz), 5.18 ( 1H, q, J = 6.8 Hz).
参考例 3- 2で合成した化合物(純度 80 , 10.0 g, 18.6 mmol), 上記 3-ブロモ -2_ォキソ酪酸ェチル(6.5 g, 18.6膽 ol)およびトリェチルアミ ン(7· 75 ml, 55.9 mmol)の DMF- THF (1:4, 200 ml)懸濁液を室温で 1.5 時間攪拌した。 ついで反応液に参考例 3-2で合成した化合物(純度 80%, 1.0 g, 1.86 mmol)を 20分毎に 3回加えた。 反応液を室温で 1.5時間攪 拌した。 反応液を濃縮し、 酢酸ェチルで抽出した。 有機層を 5 炭酸水素 ナトリゥム水溶液および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥し、 濃縮して褐色固形物(10.1 g)を得た。 Compound synthesized in Reference Example 3-2 (purity 80, 10.0 g, 18.6 mmol), A suspension of ethyl 3-bromo-2_oxobutyrate (6.5 g, 18.6 benzyl) and triethylamine (7.75 ml, 55.9 mmol) in DMF-THF (1: 4, 200 ml) was stirred at room temperature for 1.5 hours. did. Then, the compound synthesized in Reference Example 3-2 (purity 80%, 1.0 g, 1.86 mmol) was added to the reaction solution three times every 20 minutes. The reaction solution was stirred at room temperature for 1.5 hours. The reaction solution was concentrated and extracted with ethyl acetate. The organic layer was washed with an aqueous solution of sodium hydrogencarbonate and saturated saline, dried over sodium sulfate, and concentrated to obtain a brown solid (10.1 g).
'Η -丽 R (200MHz, CDC13) : δ 1.27 (3Η, t, J= 7.2 Hz), 1.29 (3H, d, J二 6.6 Hz), 1.34 (3H, t, J= 7.1 Hz), 2. 3 (3H, s) , 2.71 (2H, t, J= 7.4 Hz), 3.26 (2H, t, J= 7.4 Hz), 3.57 (1H, q, J= 6.8 Hz), 4.16 (2H, q, J= 7.1 Hz), 4.40 (2H, q, J= 7.2 Hz), 4.93 (1H, brs), 7.31 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). 'Eta -丽R (200MHz, CDC1 3): δ 1.27 (3Η, t, J = 7.2 Hz), 1.29 (3H, d, J two 6.6 Hz), 1.34 (3H, t, J = 7.1 Hz), 2 .3 (3H, s), 2.71 (2H, t, J = 7.4 Hz), 3.26 (2H, t, J = 7.4 Hz), 3.57 (1H, q, J = 6.8 Hz), 4.16 (2H, q, J = 7.1 Hz), 4.40 (2H, q, J = 7.2 Hz), 4.93 (1H, brs), 7.31 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
得られた褐色固形物(10.1 g)、 無水酢酸(5, 05 ml, 52.5 腿 ol)および p-トルエンスルホン酸水和物(以下 P- TsOH 水和物と略すことがある、 3.33 g, 17.5 mmol)のトルエン(200 ml)懸濁液を 80^ で 2.5時間攪拌 した。 反応液を濃縮し、 酢酸ェチルで抽出した。 有機層を ^炭酸水素ナ トリウム水溶液および飽和食塩水で洗浄後、 硫酸ナトリゥムで乾燥し、 濃縮した。 残渣にメタノールを加え、 析出物を濾取し、 メタノールで洗 浄、乾燥して標題化合物.(5.68g, 10.5 mmol)を赤紫色粉末として得た。 Ή-NMR (200MHz, CDC13) : 6 1. 7 (3H, t, J= 7.2 Hz), 1.33 (3H, t, 7.1 Hz), 1.93 (3H, s), 2.43 (3H, s) , 2.70 (2H, t, J= 7.3 Hz), 3.23 (2H, t, J= 7.3 Hz), 4.17 (2H, q, J= 7.2 Hz), 4.33 (2H, q, J= 7.1 Hz), 7.31 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.71 (1H, brs). 元素分析値: C22H25N307S3として計算値: C, 48.96; H, 4.67; N, 7.79; S, 17.83 (%) 、 実測値: C, 48.86; H, 4.52; N, 7.58; S, 17.80 (%) . 融点. 165.0-165.5で The resulting brown solid (10.1 g), acetic anhydride (5,05 ml, 52.5 t) and p-toluenesulfonic acid hydrate (hereinafter sometimes abbreviated as P-TsOH hydrate, 3.33 g, 17.5 g) mmol) in toluene (200 ml) was stirred at 80 ^ for 2.5 hours. The reaction solution was concentrated and extracted with ethyl acetate. The organic layer was washed with a sodium hydrogencarbonate aqueous solution and saturated saline, dried over sodium sulfate, and concentrated. Methanol was added to the residue, and the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (5.68 g, 10.5 mmol) as a red-purple powder. Ή-NMR (200MHz, CDC1 3 ): 6 1. 7 (3H, t, J = 7.2 Hz), 1.33 (3H, t, 7.1 Hz), 1.93 (3H, s), 2.43 (3H, s), 2.70 (2H, t, J = 7.3 Hz), 3.23 (2H, t, J = 7.3 Hz), 4.17 (2H, q, J = 7.2 Hz), 4.33 (2H, q, J = 7.1 Hz), 7.31 (2H , d, J = 8.4 Hz) , 7.84 (2H, d, J = 8.4 Hz), 9.71 (1H, brs) elemental analysis: calculated C 22 H 25 N 3 0 7 S 3: C, 48.96;. H, 4.67; N, 7.79; S, 17.83 (%), Found: C, 48.86; H, 4.52; N, 7.58; S, 17.80 (%). Melting point.
実施例 2 - 46 Example 2-46
8 - [(3-ェトキシ -3-ォキソプロピル)スルファニル ]-6-メチル -卜 {[(4-メ チルフエニル)スルホニル]ィミノ }- 3 -ォキソ - 2, 3-ジヒドロ- 1H -ィミダ ゾ [5, 1-c] [1,4]チアジン- 5-カルボン酸 8-[(3-Ethoxy-3-oxopropyl) sulfanyl] -6-methyl-tri {[(4-methylphenyl) sulfonyl] imino} -3- 3-oxo-2,3-dihydro-1H-imida Zo [5, 1-c] [1,4] thiazine-5-carboxylic acid
実施例 1-48で得た化合物(0.38 g, 0.649 mmol)、 無水酢酸(0.187 ml, 1.95 mmol)および p-TsOH水和物(123 mg, 0.649 mmol)の卜ルェン(7.6 ml) 懸濁液を 80°Cで 40分間攪拌した。 反応液を濃縮し、 酢酸ェチルで抽出 した。有機層を 5%炭酸水素ナトリゥム水溶液および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣に酢酸ェチルおよびジェチル エーテルを加え、 析出物を濾取し、 ジェチルェ一テルで洗浄、 乾燥して 標題化合物 (274 mg, 0.536 mmol)を赤色粉末として得た。 Suspension of the compound obtained in Examples 1-48 (0.38 g, 0.649 mmol), acetic anhydride (0.187 ml, 1.95 mmol) and p-TsOH hydrate (123 mg, 0.649 mmol) in toluene (7.6 ml) Was stirred at 80 ° C for 40 minutes. The reaction solution was concentrated and extracted with ethyl acetate. The organic layer was washed with a 5% aqueous sodium hydrogen carbonate solution and saturated saline, dried over sodium sulfate, and concentrated. Ethyl acetate and getyl ether were added to the residue, and the precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (274 mg, 0.536 mmol) as a red powder.
!H-醒 R (200MHz, CDC13) ': δ 1.27 (3Η, t, J= 7.1 Hz), 2.00 (3H, brs), 2.41 (3H, s), 2.70 (2H, t, J= 7.3 Hz), 3.23 (2H, t, J= 7.3 Hz), 4.17 (2H, q, J= 7.1 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 10.24 (1H, br). ! H-wake R (200MHz, CDC1 3 ) ': δ 1.27 (3Η, t, J = 7.1 Hz), 2.00 (3H, brs), 2.41 (3H, s), 2.70 (2H, t, J = 7.3 Hz) ), 3.23 (2H, t, J = 7.3 Hz), 4.17 (2H, q, J = 7.1 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 10.24 (1H, br).
元素分析値: C2。H21N307S3として計算値: C, 46.95; H, 4.14; N, 8.21; S, 18.80 (%) 、 実測値: C, 46.91; H, 4.21; N, 8.07; S, 18.72 (%) . 融点. 176.5-178.0°C Elemental analysis: C 2. H 21 N 3 0 7 S 3 Calculated: C, 46.95; H, 4.14; N, 8.21; S, 18.80 (%), found: C, 46.91; H, 4.21; N, 8.07; S, 18.72 ( %) Melting point. 176.5-178.0 ° C
実施例 2 - 47 Example 2-47
3 - { [6-メチル (4-メチルフエニル)スルホニル]ィミノ }-5-(4-モル ホ リ 二ルカルポ二ル)- 3-ォキソ - 1, 3-ジ ヒ ド ロ - 1H-イ ミ ダゾ [5, 1-c] [1,4]チアジン- 8-ィル] スルファニル }プロピオン酸ェチル 実施例 2- 46で得た化合物(1.60 g, 3.1 mmol) および H0BT (0· 42 g, 3.1 mmol)の THF (32 ml)溶液に WSC (0.71 g, 3.7 mmol)を室温で加え、 混合 物を室温で 30時間攪拌した。 反応液を濃縮後、 酢酸ェチルで希釈し、 希 塩酸、 2%炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫 酸ナトリウムで乾燥し、 濃縮した。 残渣にジェチルエーテルを加えて析 出物を濾取して実施例 2- 46で得た化合物の H0BTエステル (1.53 g, 2.43 腿 ol)を赤橙色粉末として得た。 この赤橙色粉末(1, 53 g, 2.43 mmol)の THF (30 ml)溶液にモルホリン(0.648 ml, 7.29 mmo 1)を室温で加えて混 合物を室温で 30分間攪拌した。 反応液を濃縮後、 酢酸ェチルで希釈し、 希塩酸、 2%炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣に酢酸ェチルを加えて析出物 を濾取し、 酢酸ェチルおよぴジェチルエーテルで洗浄後乾燥して標題化 合物 (1.32 g, 2.27 mmol)を赤紫色粉末として得た。 3-{[6-Methyl (4-methylphenyl) sulfonyl] imino} -5- (4-morphoylcarbonyl) -3-oxo-1,3-dihydro-1H-imidazo [ 5, 1-c] [1,4] thiazin-8-yl] sulfanyl} ethyl propionate The compound obtained in Example 2-46 (1.60 g, 3.1 mmol) and H0BT (0.442 g, 3.1 mmol) WSC (0.71 g, 3.7 mmol) was added to a THF (32 ml) solution at room temperature, and the mixture was stirred at room temperature for 30 hours. The reaction solution was concentrated, diluted with ethyl acetate, washed with dilute hydrochloric acid, a 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dried over sodium sulfate, and concentrated. Getyl ether was added to the residue, and the precipitate was collected by filtration to obtain a H0BT ester (1.53 g, 2.43 tmol) of the compound obtained in Example 2-46 as a red-orange powder. Morpholine (0.648 ml, 7.29 mmol) was added to a solution of this red-orange powder (1.53 g, 2.43 mmol) in THF (30 ml) at room temperature, and the mixture was stirred at room temperature for 30 minutes. After concentrating the reaction solution, dilute with ethyl acetate, wash with dilute hydrochloric acid, 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, Dried over sodium sulfate and concentrated. Ethyl acetate was added to the residue, and the precipitate was collected by filtration, washed with ethyl acetate and getyl ether, and dried to give the title compound (1.32 g, 2.27 mmol) as a purple-red powder.
Ή-NMR (200MHz, CDC13) : δ 1.27 (3Η, t, J= 7.2 Hz), 1.80 (3H, s) , 2.43 (3H, s), 2.70 (2H, t, J- 7.3 Hz), 3.22 (2H, t, J= 7.3 Hz), 3.49 (2H, m), 3.64 (4H, m) , 3.80 (2H, m) , 4.17 (2H, q, J= 7.2 Hz), 7.31 (2H, d, J= 8.6 Hz), 7.82 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.27 (3Η, t, J = 7.2 Hz), 1.80 (3H, s), 2.43 (3H, s), 2.70 (2H, t, J- 7.3 Hz), 3.22 (2H, t, J = 7.3 Hz), 3.49 (2H, m), 3.64 (4H, m), 3.80 (2H, m), 4.17 (2H, q, J = 7.2 Hz), 7.31 (2H, d, J = 8.6 Hz), 7.82 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C24H28N407S3として計算値: C, 49.64; H, 4.86; N, 9.65; S, 16.57 (%) 、 実測値: C, 49.50; H, 4.91; N, 9.74; S, 16.24 (%) . 融点. 180.0-181.0°C Elemental analysis: C 24 H 28 N 4 0 7 S 3 Calculated: C, 49.64; H, 4.86 ; N, 9.65; S, 16.57 (%), Found: C, 49.50; H, 4.91 ; N, 9.74; S, 16.24 (%). Melting point. 180.0-181.0 ° C
実施例 2-48 Example 2-48
3 - [(6-メチル -1-{ [(4-メチルフエニル)スルホニル]ィミノ } - 3 -ォキソ -5 -プロピオニル _2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 8- ィル) スルファニル]プロピオン酸ェチル 3-[[6-Methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-5-propionyl_2,3-dihydrido-1H-imidazo [5,1-c] [1,4 ] Thiazine-8-yl) sulfanyl] ethyl propionate
三臭化トリメチルフエ二ルアンモニゥム(28.8 g, 75.0 mmol)のクロ口 ホルム(100 ml)懸濁液に、室温で攪拌しながら 3, 4-へキサジオン (10ml, 82.2 mmol)を加え、混合物を室温で 75分間攪拌した。反応液を濃縮後、 酢酸ェチル -へキサン-ジェチルエーテル(1:1:1, 150 ml)で抽出し、 2 - プロモ -3, 4-へキサジオン(8.9 mmol)を含む褐色油状物(13.4 g)を得た。 参考例 3-2で合成した化合物(純度 80%, 12.5 g, 23.3 mmol)およびトリ ェチルァミン(9.68 ml, 69.8 mmol)の DMF- THF (1:3, 200 ml)懸濁液に 室温で上記褐色油状物(13.4 g)の THF (50 ml) 溶液を加え、混合物を 3.5 時間攪拌した。 反応液を濃縮し、 酢酸ェチル(300 ml)で抽出した。 有機 層を 4%炭酸水素ナトリゥム水溶液および飽和食塩水で洗浄後、硫酸ナト リウムで乾燥し、 濃縮して褐色油状物を得た。 得られた褐色油状物、 無 水酢酸(6.72 ml, 69.8 mmo 1)および p- TsOH水和物(4.43 g, 23.3 mmol) のトルエン(250ml)懸濁液を 80°C で 2時間攪拌した。反応液を濃縮し、 酢酸ェチル(300 ml) および水(150 ml)と混和した。 析出物を濾取し、 水 および酢酸ェチルで洗浄、 乾燥して赤紫色粉末(4.5 g)を得た。 母液およ び洗浄液を合わせ、有機層を ^炭酸水素ナトリウム水溶液、 0.2N 塩酸、 水および飽和食塩水で洗浄後、 乾燥、 濃縮して得た残渣にメタノールを 加えて 50°Cで混和した。 析出物を濾取し、 上記赤紫色粉末(4.5 g)と合 わせ、 THF (200 ml)と 50°Cで混和した。 THF可溶分を濃縮し、 酢酸ェチ ル(100 ml)に懸濁した。 析出物を濾取し、 酢酸ェチルで洗浄、 乾燥して 標題化合物 (3.22 g, 6.15 minol)を赤紫色粉末として得た。 To a suspension of trimethylphenylammonium tribromide (28.8 g, 75.0 mmol) in chloroform (100 ml) was added 3,4-hexadione (10 ml, 82.2 mmol) with stirring at room temperature, and the mixture was cooled to room temperature. For 75 minutes. After concentrating the reaction mixture, it was extracted with ethyl acetate-hexane-ethyl ether (1: 1: 1, 150 ml), and a brown oil (13.4) containing 2-bromo-3,4-hexadione (8.9 mmol) was obtained. g) was obtained. The above brown solution was added to a suspension of the compound synthesized in Reference Example 3-2 (purity 80%, 12.5 g, 23.3 mmol) and triethylamine (9.68 ml, 69.8 mmol) in DMF-THF (1: 3, 200 ml) at room temperature. A solution of the oil (13.4 g) in THF (50 ml) was added and the mixture was stirred for 3.5 hours. The reaction solution was concentrated and extracted with ethyl acetate (300 ml). The organic layer was washed with a 4% aqueous sodium hydrogen carbonate solution and saturated saline, dried over sodium sulfate, and concentrated to give a brown oil. A suspension of the obtained brown oil, anhydrous acetic acid (6.72 ml, 69.8 mmol) 1 and p-TsOH hydrate (4.43 g, 23.3 mmol) in toluene (250 ml) was stirred at 80 ° C for 2 hours. The reaction solution was concentrated and mixed with ethyl acetate (300 ml) and water (150 ml). The precipitate was collected by filtration, washed with water and ethyl acetate, and dried to obtain a reddish purple powder (4.5 g). Mother liquor and The organic layer was washed with an aqueous solution of sodium bicarbonate, 0.2N hydrochloric acid, water and saturated saline, dried and concentrated, and the residue was mixed with methanol at 50 ° C. The precipitate was collected by filtration, combined with the reddish purple powder (4.5 g), and mixed with THF (200 ml) at 50 ° C. The THF soluble matter was concentrated and suspended in ethyl acetate (100 ml). The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (3.22 g, 6.15 minol) as a red-purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.17 (3Η, t, J= 7.2 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.76 (3H, s), 2.43 (3H, s), 2.58 (2H, q, J= 7.2 Hz), 2.70 (2H, t, J= 7.5 Hz), 3.23 (2H, t, J= 7.3 Hz), 4.17 (2H, q, J= 7.2 Hz), 7.31 (2H, d, J= 8.0 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.74 (1H, brs). 元素分析値 : C22H25N306S3 として計算値: C, 50.46; H, 4.81; N, 8.02 Ή-NMR (200MHz, CDC1 3 ): δ 1.17 (3Η, t, J = 7.2 Hz), 1.27 (3H, t, J = 7.2 Hz), 1.76 (3H, s), 2.43 (3H, s), 2.58 (2H, q, J = 7.2 Hz), 2.70 (2H, t, J = 7.5 Hz), 3.23 (2H, t, J = 7.3 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.31 (2H , d, J = 8.0 Hz) , 7.84 (2H, d, J = 8.4 Hz), 9.74 (1H, brs) elemental analysis: calculated C 22 H 25 N 3 0 6 S 3: C, 50.46;. H, 4.81; N, 8.02
(%)、 実測値: C, 50.56 ; H, 4.52; N, 8.04 ( ) . (%), Found: C, 50.56; H, 4.52; N, 8.04 ().
融点. 206.5-209.0°C Melting point. 206.5-209.0 ° C
実施例 2-49 Example 2-49
'H-NMR (CDC13, 200 MHz): δ 1.26 (3Η, t, J= 7.2 Hz), 1.79 (3H, s), 2.26 (3H, s), 2.42 (3H, s) , 2.70 (2H, t, J= 7.4 Hz), 3.21 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.2 Hz), 7.36 (1H, brd, J= 2.0 Hz), 7.38 (1H, brs), 7.77 (2H, m) , 9.63 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1.26 (3Η, t, J = 7.2 Hz), 1.79 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.70 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.2 Hz), 7.36 (1H, brd, J = 2.0 Hz), 7.38 (1H, brs) , 7.77 (2H, m), 9.63 (1H, brs).
元素分析値: C2。H23N305S3として計算値: 9.88; H, 4.81; , 8.72; S, 19.97 (%), 実測値: C, 50.11; H, 4.89; N, 8.67; S, 19.91 (%) . 実施例 2-50 Elemental analysis: C 2. Calculated as H 23 N 3 0 5 S 3 : 9.88; H, 4.81;, 8.72; S, 19.97 (%), Found: C, 50.11; H, 4.89 ; N, 8.67; S, 19.91 (%). Example 2-50
Ή-NMR (CDC13, 200 MHz): δ 1.26 (3Η, t, J= 7.2 Hz), 1.32 (3H, t, J= 7.1 Hz), 2.43 (3H, s), 2.72 (2H, t, J= 7.4 Hz), 2.76 (3H, s), 3.27 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.2 Hz), 4.27 (2H, q, J= 7.4 Hz), 7.38 (2H, m) , 7.76 (2H, m) , 9.79 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.26 (3Η, t, J = 7.2 Hz), 1.32 (3H, t, J = 7.1 Hz), 2.43 (3H, s), 2.72 (2H, t, J = 7.4 Hz), 2.76 (3H, s), 3.27 (2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.2 Hz), 4.27 (2H, q, J = 7.4 Hz), 7.38 ( 2H, m), 7.76 (2H, m), 9.79 (1H, brs).
元素分析値: C22H25N307S3として計算値: C, 48.96; H, 4.67; N, 7.79; S, 17.83 (%), 実測値: C, 48.89; H, 4.56; N, 7.72; S, 17.83 (%) . 実施例 2 - 51 Elemental analysis: C 22 H 25 N 3 0 7 S 3 Calculated: C, 48.96; H, 4.67 ; N, 7.79; S, 17.83 (%), Found: C, 48.89; H, 4.56 ; N, 7.72; S, 17.83 (%). Examples 2-51
Ή-NMR (CDC13) : δ 0.89 (3H, t, J = 7.0Hz), 1.24-1.37 (6H, m) , 1.39-1.52 (2H, m), 2.07 (2H, t, J = 7.0Hz) , 2.72 (2H, t, J = 7.3Hz), 2.77 (3H, s), 3.25 (2H, t, J = 7.3Hz) , 3.71 (3H, s), 6.50 (1H, s), 7.25-7.34 (2H, m), 7.45 (1H, ddd, J=7.5, 7.5, 1.2Hz), 8.01 (1H, dd, J=7.5, 1.5Hz), 9.75 (1H, brs). Ή-NMR (CDC1 3): δ 0.89 (3H, t, J = 7.0Hz), 1.24-1.37 (6H, m), 1.39-1.52 (2H, m), 2.07 (2H, t, J = 7.0Hz), 2.72 (2H, t, J = 7.3Hz), 2.77 (3H, s), 3.25 (2H, t, J = 7.3Hz ), 3.71 (3H, s), 6.50 (1H, s), 7.25-7.34 (2H, m), 7.45 (1H, ddd, J = 7.5, 7.5, 1.2Hz), 8.01 (1H, dd, J = 7.5) , 1.5Hz), 9.75 (1H, brs).
元素分析値: C23H29N305S3 として計算値: C; 52.75, H; 5.58, N; 8.02 ( ), 実測値: C; 52.63, H; 5.59, N; 8.08 (%) . Elemental analysis: Calculated as C 23 H 29 N 3 0 5 S 3: C; 52.75, H; 5.58, N; 8.02 (), Found: C; 52.63, H; 5.59 , N; 8.08 (%).
実施例 2 - 52 Example 2-52
Ή-NMR (CDC13, 200 MHz): δ 1.26 (3H, t, J= 7.2 Hz), 1.79 (3H, s) , 2.26 (3H, s), 2.69 (2H, t, J- 7.3 Hz), 2.77 (3H, s) , 3.22 (2H, t, J= 7.5 Hz), 4.16 (2H, q, J= 7.0 Hz), 4.26 (2H, q, J= 7.4 Hz), 7.31 (2H, m), 7.43 (1H, tm, J= 7 Hz), 8.01 (1H, dm, J= 7.6 Hz), 9.66 (1H, brs) . Ή-NMR (CDC1 3, 200 MHz): δ 1.26 (3H, t, J = 7.2 Hz), 1.79 (3H, s), 2.26 (3H, s), 2.69 (2H, t, J- 7.3 Hz), 2.77 (3H, s), 3.22 (2H, t, J = 7.5 Hz), 4.16 (2H, q, J = 7.0 Hz), 4.26 (2H, q, J = 7.4 Hz), 7.31 (2H, m), 7.43 (1H, tm, J = 7 Hz), 8.01 (1H, dm, J = 7.6 Hz), 9.66 (1H, brs).
元素分析値: C2。H23N305S3として計算値: C, 49.88; H, 4.81; N, 8.72; S, 19.97 (%), 実測値: C, 49.74; H, 4.61; N, 8.86; S, 19.99 (%) . 実施例 2-53 Elemental analysis: C 2. H 23 N 3 0 5 S 3 Calculated: C, 49.88; H, 4.81; N, 8.72; S, 19.97 (%), Found: C, 49.74; H, 4.61; N, 8.86; S, 19.99 ( %). Example 2-53
Ή-NMR (200 MHz, 重クロ口ホルム中): δ 1.26 (3Η, t, J= 7.0Hz), 1.32 (3H, t, J= 7.0 Hz), 2.71 (2H, t, J= 7.2 Hz), 2.75 (3H, s), 2.76 (3H, s), 3.28 (2H, t, J= 7.2 Hz), 4.17 (2H, q, J= 7.2 Hz), 4.26 (2H, q, J= 7.4 Hz), 7.31 (2H, m) , 7.46 (1H, brt, J= 5.4 Hz), 8.01 (1H, dm, J= 8.4 Hz), 9.82 (1H, brs). Ή-NMR (200 MHz, in double-mouthed form): δ 1.26 (3Η, t, J = 7.0 Hz), 1.32 (3H, t, J = 7.0 Hz), 2.71 (2H, t, J = 7.2 Hz) , 2.75 (3H, s), 2.76 (3H, s), 3.28 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J = 7.2 Hz), 4.26 (2H, q, J = 7.4 Hz) , 7.31 (2H, m), 7.46 (1H, brt, J = 5.4 Hz), 8.01 (1H, dm, J = 8.4 Hz), 9.82 (1H, brs).
元素分析値: C22H25N307S3 として計算値: C, 48.96; H, 4.67; N, 7.79; S, 7.83 (%)、 実測値; C, 48.89; H, 4.82; N, 7.95; S, 17.85 (%) . 実施例 2-54 Elemental analysis: C 22 H 25 N 3 0 7 S 3 Calculated: C, 48.96; H, 4.67 ; N, 7.79; S, 7.83 (%), Found; C, 48.89; H, 4.82 ; N, 7.95; S, 17.85 (%). Example 2-54
Ή-NMR (CDC13) : δ 0.89 (3Η, t, J = 6.9Hz), 1.23-1.37 (6H, m) , 1.41-1.52 (2H, m) , 2.01 (2H, t, J = 7.6Hz) , 2.72 (2H, t, J = 7.4Hz), 3.24 (2H, t, J = 7.4Hz), 3.71 (3H, s), 6.51 (1H, br s), 7.52-7.61 (3H, m), 7.94-7.99 (2H, m) , 9.73 (1H, br s). Ή-NMR (CDC1 3): δ 0.89 (3Η, t, J = 6.9Hz), 1.23-1.37 (6H, m), 1.41-1.52 (2H, m), 2.01 (2H, t, J = 7.6Hz) , 2.72 (2H, t, J = 7.4Hz), 3.24 (2H, t, J = 7.4Hz), 3.71 (3H, s), 6.51 (1H, br s), 7.52-7.61 (3H, m), 7.94 -7.99 (2H, m), 9.73 (1H, br s).
元素分析値: C22H27N305S3として計算値: C; 51.85, H; 5.34, N; 8.24(%)、 実測値: C; 51.77, H; 5.37, N; 8.19 ( ) . 実施例 2-55 Elemental analysis: Calculated as C 22 H 27 N 3 0 5 S 3: C; 51.85, H; 5.34, N; 8.24 (%), Found: C; 51.77, H; 5.37 , N; 8.19 (). Example 2-55
'Η-讓 R (CDC13, 200 MHz): δ 1.27 (3H, t, J= 7.4 Hz), 1.32 (3H, t, J= 7.2 Hz), 2.72 (2H, t, J= 7.0 Hz), 2.76 (3H, s), 3.27 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J= 7.0 Hz), 4.27 (2H, q, J= 7.0 Hz), 7.53 (3H, m), 7.96 (2H, dm, J= 8 Hz) , 9.80 (1H, brs). '.Eta. Yuzuru R (CDC1 3, 200 MHz) : δ 1.27 (3H, t, J = 7.4 Hz), 1.32 (3H, t, J = 7.2 Hz), 2.72 (2H, t, J = 7.0 Hz), 2.76 (3H, s), 3.27 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.0 Hz), 4.27 (2H, q, J = 7.0 Hz), 7.53 (3H, m), 7.96 (2H, dm, J = 8 Hz), 9.80 (1H, brs).
元素分析値: C21H23N307S3として計算値: C, 47.99; H, 4.41; N, 7.99; S,Elemental analysis: Calculated for C 21 H 23 N 3 0 7 S 3 : C, 47.99; H, 4.41; N, 7.99; S,
18.30 (%), 実測値: C, 47.89; H, 4.32; N, 7.90; S, 18.45 (%) . 実施例 2-56 18.30 (%), Found: C, 47.89; H, 4.32; N, 7.90; S, 18.45 (%).
lH-匪 R (CDC 13, 200 MHz): δ 1.27 (3H, t, J= 7.1 Hz), 1.79 (3H, brq, J= 0.8 Hz), 2.27 (2H, brq, J= 0.6 Hz), 2.70 (2H, t, J= 7.4 Hz), 3.21 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.2 Hz), 7.52 (3H, m) , 7.96 (2H, dm, J= 7.6 Hz), 9.64 (1H, brs). lH- negation R (CDC 1 3, 200 MHz ): δ 1.27 (3H, t, J = 7.1 Hz), 1.79 (3H, brq, J = 0.8 Hz), 2.27 (2H, brq, J = 0.6 Hz), 2.70 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.2 Hz), 7.52 (3H, m), 7.96 (2H, dm, J = 7.6 Hz), 9.64 (1H, brs).
元素分析値: C19H21N305S3として計算値: C, 48.80; H, 4.53; N, 8.99; S, 20.57 (%), 実測値: C, 48.67; H, 4. 9; N, 8.86; S, 20.60 (%) . 実施例 2-57 Elemental analysis: Calculated as C 19 H 21 N 3 0 5 S 3: C, 48.80; H, 4.53; N, 8.99; S, 20.57 (%), Found: C, 48.67; H, 4. 9; N, 8.86; S, 20.60 (%). Example 2-57
Ή-NMR (CDC13, 200 MHz): δ 1.09 (3Η, t, J= 7.5 Hz), 1.27 (3H, t, J= 7.2 Hz), 2.24 (2H, q, J= 7.6 Hz), 2.27 (3H, s), 2.70' (2H, t, J = 7.4 Hz), 3.23 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.2 Hz), 7.52 (3H, m), 7.96 (2H, m) , 9.65 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.09 (3Η, t, J = 7.5 Hz), 1.27 (3H, t, J = 7.2 Hz), 2.24 (2H, q, J = 7.6 Hz), 2.27 ( 3H, s), 2.70 '(2H, t, J = 7.4 Hz), 3.23 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.52 (3H, m), 7.96 (2H, m), 9.65 (1H, brs).
元素分析値: C2。H23N305S3として計算値: 49.88; H, 4.81; N, 8.72; S,Elemental analysis: C 2. H 23 N 3 0 5 S 3 Calculated: 49.88; H, 4.81; N , 8.72; S,
19.97 (%), 実測値: C, 49.73; H, 4.53; N, 8.67; S, 20.01 (%) . 実施例 2-58 19.97 (%), found: C, 49.73; H, 4.53; N, 8.67; S, 20.01 (%). Example 2-58
LH-NMR (200 MHz, 重クロ口ホルム中): δ 0.89 (3Η, t, J= 7.2Hz), 1.26 (3H, t, J= 7.2 Hz), 1.30 (4H, m) , 1.33 (2H, m) , 1. 7 (2H, brquint, J- 7.0 Hz), 2.07 (2H, t, J= 7.5 Hz), 2.71 (2H, t, J= 7.4 Hz), 3.24 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7. I Hz), 6.50 (1H, s), 7.02 (2H, dt, J= 8.7, 2.3 Hz), 7.07 (2H, brd, J= 8.0 Hz), 7.21 (1H, brt, J = 7.5 Hz), 7.40 (2H, tt, J= 7.9, 2.3 Hz), 7.89 (2H, dt, J= 9.0, 2.3 Hz), 9.70 (1H, brs). 元素分析値: C29H33N306S3 として計算値: C, 56.56; H, 5.40; N, 6.82 L H-NMR (200 MHz, in double-mouthed form): δ 0.89 (3Η, t, J = 7.2 Hz), 1.26 (3H, t, J = 7.2 Hz), 1.30 (4H, m), 1.33 (2H , m), 1.7 (2H, brquint, J- 7.0 Hz), 2.07 (2H, t, J = 7.5 Hz), 2.71 (2H, t, J = 7.4 Hz), 3.24 (2H, t, J = 7.4 Hz), 4.16 (2H, q, J = 7.I Hz), 6.50 (1H, s), 7.02 (2H, dt, J = 8.7, 2.3 Hz), 7.07 (2H, brd, J = 8.0 Hz) , 7.21 (1H, brt, J = 7.5 Hz), 7.40 (2H, tt, J = 7.9, 2.3 Hz), 7.89 (2H, dt, J = 9.0, 2.3 Hz), 9.70 (1H, brs). Elemental analysis: C 29 H 33 N 3 0 6 S 3 Calculated: C, 56.56; H, 5.40 ; N, 6.82
{%), 実測値; C, 56.41; H, 5.33; N, 6.81 (%) . {%), Found; C, 56.41; H, 5.33; N, 6.81 (%).
実施例 2-59 Example 2-59
Ή-NMR (CDC13, 200 MHz): δ 1.26 (3Η, t, J= 7.4 Hz), 1.33 (3H, t, J= 7.4 Hz), 2.72 (2H, t, J= 7.6 Hz), 2.76 (3H, s), 3.27 (2H, t, J = 7.2 Hz), 4.16 (2H, q, J= 7.4 Hz), 4.27 (2H, q, J= 7.0 Hz), 7.03 (2H, d, J= 8.8 Hz), 7.07 (2H, d, J= 7.4 Hz), 7.26 (1H, m) , 7.41 (2H, d, J= 8.0 Hz), 7.89 (2H, d, J= 8.8 Hz), 9.77 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.26 (3Η, t, J = 7.4 Hz), 1.33 (3H, t, J = 7.4 Hz), 2.72 (2H, t, J = 7.6 Hz), 2.76 ( 3H, s), 3.27 (2H, t, J = 7.2 Hz), 4.16 (2H, q, J = 7.4 Hz), 4.27 (2H, q, J = 7.0 Hz), 7.03 (2H, d, J = 8.8 Hz), 7.07 (2H, d, J = 7.4 Hz), 7.26 (1H, m), 7.41 (2H, d, J = 8.0 Hz), 7.89 (2H, d, J = 8.8 Hz), 9.77 (1H, brs).
元素分析値: C27H27N308S3として計算値: C, 52.50; H, 4.41; , 6.80; S, 15.57 (%), 実測値: 52.72; H, 4.27; N, 6.80; S, 15.59 (%) . 実施例 2-60 Elemental analysis: Calculated as C 27 H 27 N 3 0 8 S 3: C, 52.50; H, 4.41;, 6.80; S, 15.57 (%), Found: 52.72; H, 4.27; N , 6.80; S , 15.59 (%). Examples 2-60
'H-NMR (CDC13, 200 MHz): δ 1. 6 (3Η, i, J= 7.4 Hz), 1.80 (3H, brq, J二 0.8 Hz), 2.27 (3H, brq, J= 1.0 Hz), 2.70 (2H, t, J= 7.4 Hz), 3.21 (2H, t, J= 7.4 Hz), 4.15 (2H, q, J= 7.0 Hz), 7.02 (2H, d, J= 8.8 Hz), 7.06 (2H, d, J= 8.0 Hz), 7.21 (1H, t, J= 7.4 Hz), 7.41 (2H, t, J = 8.2 Hz), 7.90 (2H, d, J= 8.8 Hz), 9.61 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1. 6 (3Η, i, J = 7.4 Hz), 1.80 (3H, brq, J two 0.8 Hz), 2.27 (3H, brq, J = 1.0 Hz) , 2.70 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.4 Hz), 4.15 (2H, q, J = 7.0 Hz), 7.02 (2H, d, J = 8.8 Hz), 7.06 (2H, d, J = 8.0 Hz), 7.21 (1H, t, J = 7.4 Hz), 7.41 (2H, t, J = 8.2 Hz), 7.90 (2H, d, J = 8.8 Hz), 9.61 (1H , Brs).
元素分析値: C25H25N306S3として計算値: C, 53.65; H, 4.50; N, 7.51; S, 17.19 (%), 実測値: C, 53.57; H, 4.26; N, 7.76; S, 17.15 (%) . 実施例 2-61 Elemental analysis: C 25 H 25 N 3 0 6 S 3 Calculated: C, 53.65; H, 4.50 ; N, 7.51; S, 17.19 (%), Found: C, 53.57; H, 4.26 ; N, 7.76; S, 17.15 (%). Example 2-61
^-NMR (CDC 13, 200 MHz): δ 1.24 (3Η, t, 1= 7.1 Hz), 1.80 (3H, brs), 2.29 (3H, brs), 2.67 (2H, t, J= 7.3 Hz), 3.19 (2H, t, J= 7.3 Hz), 4.14 (2H, q, J= 7.0 Hz), 7.42 (1H, i) , 7. 9 (2H, m) , 8.18 (1H, d, J= 8.0 Hz), 9.75 (1H, brs). ^ -NMR (CDC 1 3, 200 MHz): δ 1.24 (3Η, t, 1 = 7.1 Hz), 1.80 (3H, brs), 2.29 (3H, brs), 2.67 (2H, t, J = 7.3 Hz) , 3.19 (2H, t, J = 7.3 Hz), 4.14 (2H, q, J = 7.0 Hz), 7.42 (1H, i), 7.9 (2H, m), 8.18 (1H, d, J = 8.0 Hz), 9.75 (1H, brs).
元素分析値: C20H23N305S3C1 として計算値: C, 45.46; H, 4.02; N, 8.37; S, 19.16; CI, 7.06 (%)、 実測値: C, 45.36; H, 3.99; N, 8.12; S, 19.14; CI, 7.09 (%) . Elemental analysis: Calculated as C 20 H 23 N 3 0 5 S 3 C1: C, 45.46; H, 4.02; N, 8.37; S, 19.16; CI, 7.06 (%), Found: C, 45.36; H , 3.99; N, 8.12; S, 19.14; CI, 7.09 (%).
実施例 2-62 Example 2-62
Ή-NMR (CDC13, 200 MHz): δ 1.27 (3H, t, J= 6.8 Hz), 1.33 (3H, t, J= 7.0 Hz), 2.72 (2H, t, J= 7.4 Hz), 2.77 (3H, s), 3.28 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J= 7.0 Hz), 4.27 (2H, q, J= 7.0 Hz), 7.48 (2H, d, J= 8.8 Hz), 7.90 (2H, d, J= 8.8 Hz), 9.78 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.27 (3H, t, J = 6.8 Hz), 1.33 (3H, t, J = 7.0 Hz), 2.72 (2H, t, J = 7.4 Hz), 2.77 ( 3H, s), 3.28 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.0 Hz), 4.27 (2H, q, J = 7.0 Hz), 7.48 (2H, d, J = 8.8 Hz), 7.90 (2H, d, J = 8.8 Hz) ), 9.78 (1H, brs).
元素分析値: C21H22N307S3C1 として計算値: C, 45.03; H, 3.96; N, 7.50; S, 17.18; C1, 6.33(%)、実測値: C, 44.77; H, 3.89; N, 7.52; S, 17.39; C1, 6.24 ( ) . Elemental analysis: C 21 H 22 N 3 0 7 S 3 C1 Calculated: C, 45.03; H, 3.96; N, 7.50; S, 17.18; C1, 6.33 (%), found: C, 44.77; H , 3.89; N, 7.52; S, 17.39; C1, 6.24 ().
実施例 2-63 Example 2-63
Ή-NMR (CDC13, 200 MHz): δ 1.27 (3Η, t, J= 7.2 Hz), 1.33 (3H, t, J= 7.2 Hz), 2.72 (2H, t, J= 7.4 Hz), 2.76 (3H, s), 3.28 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J= 7.4 Hz), 4.27 (2H, q, J= 7.4 Hz), 7.18 (2H, t, J= 8.6 Hz), 7.97 (2H, dd, J= 8.8, 5.0 Hz), 9.77 (1H, brs). 元素分析値: C2iH22N307S3Fとして計算値: C, 46.40;. H, 4.08; N, 7.73; S, 17.70; F, 3.49 (%)、実測値: C, 46.50; H, 4.29; N, 7.64; S, 17.76; F, 3.50 (%) . Ή-NMR (CDC1 3, 200 MHz): δ 1.27 (3Η, t, J = 7.2 Hz), 1.33 (3H, t, J = 7.2 Hz), 2.72 (2H, t, J = 7.4 Hz), 2.76 ( 3H, s), 3.28 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J = 7.4 Hz), 4.27 (2H, q, J = 7.4 Hz), 7.18 (2H, t, J = 8.6) Hz), 7.97 (2H, dd, J = 8.8, 5.0 Hz), 9.77 (1H, brs). Elemental analysis: Calculated as C 2i H 22 N 3 0 7 S 3 F: C, 46.40 ;. H, 4.08; N, 7.73; S, 17.70; F, 3.49 (%), Found: C, 46.50; H, 4.29; N, 7.64; S, 17.76; F, 3.50 (%).
実施例 2-64 ' Example 2-64 '
Ή-NMR (CDCl 3, 200 MHz): 6 1.27 (3H, t, J= 7.1 Hz), 1.80 (3H, brs), 2.28 (3H, brs), 2.70 (2H, t, J= 7.2 Hz), 3.22 (2H, t, J= 7.2 Hz), 4.17 (2H, q, J= 7.4 Hz), 7.17 (2H, t, J= 8.4 Hz), 7.98 (2H, dd, J = 8.4, 5.2 Hz), 9.62 (1H, brs). Ή-NMR (CDCl 3, 200 MHz): 6 1.27 (3H, t, J = 7.1 Hz), 1.80 (3H, brs), 2.28 (3H, brs), 2.70 (2H, t, J = 7.2 Hz), 3.22 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J = 7.4 Hz), 7.17 (2H, t, J = 8.4 Hz), 7.98 (2H, dd, J = 8.4, 5.2 Hz), 9.62 (1H, brs).
元素分析値: C2。H22N305S3F · 0.3H20として計算値: C, 46.48; H, 4.23; N, 8.56; S, 19.59; F, 3.87 (%)、 実測値: C, 46.42; H, 4.20; N, 8.68; S, 19.72; F, 3.96 (%) . Elemental analysis: C 2. H 22 N 3 0 5 S 3 F · Calculated as 0.3H 2 0: C, 46.48; H, 4.23; N, 8.56; S, 19.59; F, 3.87 (%), found: C, 46.42; H, 4.20; N, 8.68; S, 19.72; F, 3.96 (%).
実施例 2-65 Example 2-65
lH-NMR (CDC13, 300 MHz): δ 1.34 (3H, t, J= 7.2 Hz), 2.76 (3H, s), 2.77 (2H, t, J= 7.2 Hz), 3.10 (3H, s), 3.31 (2H, t, J= 7.2 Hz), 3.74 (3H, s), 4.28 (2H, q, J- 7.2 Hz), 9.61 (1H, br). lH-NMR (CDC1 3, 300 MHz): δ 1.34 (3H, t, J = 7.2 Hz), 2.76 (3H, s), 2.77 (2H, t, J = 7.2 Hz), 3.10 (3H, s), 3.31 (2H, t, J = 7.2 Hz), 3.74 (3H, s), 4.28 (2H, q, J- 7.2 Hz), 9.61 (1H, br).
元素分析値: C15H19N307S3として計算値: 0.08; H, 4.26; N, 9.35; S, 21. 0 (%), 実測値: C, 39.85; H, 4.23; N, 9.20; S, 21.36 ( ) . 実施例 2-66 Elemental analysis: C 15 H 19 N 3 0 7 S 3 Calculated: 0.08; H, 4.26; N, 9.35; S, 21.0 (%), found: C, 39.85; H, 4.23; N, 9.20; S, 21.36 (). Example 2-66
Ή-NMR (CDC 13, 300 MHz): δ 1.29 (3Η, t, J= 7.2 Hz), 1.34 (3H, t, J= 7.2 Hz), 2.75 (2H, t, J= 7.5 Hz), 2.76 (3H, s) , 3.10 (3H, s), 3.31 (2H, t, J= 7.2 Hz), 4.19 (2H, q, J= 7.2 Hz), 4.28 (2H, q, J= 7.2 Hz), 9.58 (1H, br) . Ή-NMR (CDC 1 3, 300 MHz): δ 1.29 (3Η, t, J = 7.2 Hz), 1.34 (3H, t, J = 7.2 Hz), 2.75 (2H, t, J = 7.5 Hz), 2.76 (3H, s), 3.10 (3H, s), 3.31 (2H, t, J = 7.2 Hz), 4.19 (2H, q, J = 7.2 Hz), 4.28 (2H, q, J = 7.2 Hz), 9.58 (1H, br).
元素分析値: C16H21N307S3として計算値: C, 41.46; H, 4.57; N, 9.06; S, 20.75 (%), 実測値: C, 41.75; H, 4.77; N, 8.87; S, 20.65 (%) . 実施例 2-67 Elemental analysis: C 16 H 21 N 3 0 7 S 3 Calculated: C, 41.46; H, 4.57; N, 9.06; S, 20.75 (%), found: C, 41.75; H, 4.77; N, 8.87; S, 20.65 (%). Example 2-67
3-[(3-{[(4-メチルフエニル)スルホニル]イミノ} -卜ォキソ - 2,3-ジヒド 口- 1H-ィミダゾ [5,.1- c] [1, 4]ベンゾチアジン- 4-ィル)スルファニル]プ 口ピオン酸ェチル 3-[(3-{[(4-methylphenyl) sulfonyl] imino} -toxo-2,3-dihydrone-1H-imidazo [5, .1-c] [1,4] benzothiazine-4-yl ) Sulfanyl] p Ethyl pionate
参考例 3-2で合成した化合物 (5.70 g, 13.3 mmol) を THF (50.0 ml) および DMF (25.0 ml) に溶解し、 2, 6-ジブ口モシクロへキサノン (4.25 g, 16.6 mmol) およびトリェチルァミン (5.55 ml, 39.8 mmol) を加え て室温で 6.5時間撹拌した。 反応液を濃縮後、 残渣を酢酸ェチルで希釈 し、 水、 および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒 を減圧下に留去して表題化合物の油状物を四種異性体の混合物として得 た。 本品はこれ以上精製することなく次の反応に使用した。 The compound synthesized in Reference Example 3-2 (5.70 g, 13.3 mmol) was dissolved in THF (50.0 ml) and DMF (25.0 ml), and 2,6-dibumocyclohexanone (4.25 g, 16.6 mmol) and triethylamine were dissolved. (5.55 ml, 39.8 mmol) and the mixture was stirred at room temperature for 6.5 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed with water and saturated saline, and then dried over sodium sulfate. The solvent was distilled off under reduced pressure to obtain an oily substance of the title compound as a mixture of four isomers. This product was used for the next reaction without further purification.
上記の油状物をトルエン (50.0 ml) および無水酢酸 (3.76 ml, 39.8 mmol) に溶解し、 P-トルエンスルホン酸 1水和物 (2.13 g, 10.6 mmol) を加えて 100 で 45分加熱撹拌した。 反応液を濃縮後、 残渣を酢酸ェチ ルで希釈し、水で 3回飽和食塩水で洗浄後、硫酸ナトリウムで乾燥した。 溶媒を減圧下に留去して題記化合物 (890 mg, 13¾) を得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 163- 165での橙色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) リ: 3252, 2980, 2926, 1738, 1632, 1586, 1557, 1485 cm"1. The above oil was dissolved in toluene (50.0 ml) and acetic anhydride (3.76 ml, 39.8 mmol), and P-toluenesulfonic acid monohydrate (2.13 g, 10.6 mmol) was added. The mixture was heated and stirred at 100 for 45 minutes. . After concentrating the reaction solution, the residue was diluted with ethyl acetate, washed with water three times with a saturated saline solution, and dried over sodium sulfate. The solvent was distilled off under reduced pressure to obtain the title compound (890 mg, 13¾). Recrystallization from ethyl acetate-getyl ether gave orange crystals with mp 163-165. Infrared absorption spectrum (IR) (in KBr): 3252, 2980, 2926, 1738, 1632, 1586, 1557, 1485 cm " 1 .
Ή-NMR (CDC13) : δ 1.28 (3Η, t, J=7.0 Hz), 2. 3 (3H, s), 2.74 (2H, t, J = 7.2 Hz), 3.29 (2H, t, J = 7.8 Hz), 4.18 (2H, q, J = 7.0 Hz), 6.96 (1H, dd, J = 7.6, 1.4 Hz), 7.08 (1H, dt, J = 6.2, 1.6 Hz), 7.22 (1H, dt, J = 8.4, 1.8 Hz), 7.51 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.0 Hz), 8.45 (1H, dd, J=8.4, 1.0 Hz), 9.79 (1H, bs). 元素分析値: C22H2lN303S3として Ή-NMR (CDC1 3): δ 1.28 (3Η, t, J = 7.0 Hz), 2. 3 (3H, s), 2.74 (2H, t, J = 7.2 Hz), 3.29 (2H, t, J = 7.8 Hz), 4.18 (2H, q, J = 7.0 Hz), 6.96 (1H, dd, J = 7.6, 1.4 Hz), 7.08 (1H, dt, J = 6.2, 1.6 Hz), 7.22 (1H, dt, J = 8.4, 1.8 Hz), 7.51 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.0 Hz), 8.45 (1H, dd, J = 8.4, 1.0 Hz), 9.79 (1H, bs). Elemental analysis: as C 22 H 2l N 3 0 3 S 3
計算値 (%) : C, 52.47; H, 4.20; N, 8.34; S, 19.10. Calculated value (%): C, 52.47; H, 4.20; N, 8.34; S, 19.10.
実測値 (%) : C, 52.33; H, 4.31; , 8. 9; S, 19.03. Observed value (%): C, 52.33; H, 4.31;, 8.9; S, 19.03.
実施例 2-68 Example 2-68
3-[1, 6_ジォキソ -3 - {[(4 -メ チルフ エニル) スルホニル ] ィ ミ ノ卜 2, 3, 6, 7, 8, 9-へキサヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジ ン- 4-ィル)スルファニル]プロピオン酸ェチル 3- [1, 6_dioxo-3-{[(4-methylphenyl) sulfonyl] iminot 2, 3, 6, 7, 8, 9-hexahydro-1H-imidazo [5, 1-c ] [1,4] benzothiazin-4-yl) sulfanyl] ethyl propionate
参考例 3-2で合成した化合物 (10.0 g, 23.2 mmol) を THF (100 ml) および DMF (50.0 ml) に溶解し、 2_ブロモ -1, 3-シクロへキサンジオン (4.44 g, 23.2 mmol) およびトリェチルァミン (9.74 ml, 69.8 mmol) を加えて室温で 5時間撹拌した。 反応液を濃縮後、 残渣を酢酸ェチルで 希釈し、 水、 および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に留去して表題化合物の油状物 (10.8g, 86%) を四種異性 体の混合物として得た。 本品はこれ以上精製することなく次の反応に使 用した。 The compound synthesized in Reference Example 3-2 (10.0 g, 23.2 mmol) was dissolved in THF (100 ml) and DMF (50.0 ml), and 2_bromo-1,3-cyclohexanedione (4.44 g, 23.2 mmol) was dissolved. And triethylamine (9.74 ml, 69.8 mmol) were added, and the mixture was stirred at room temperature for 5 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed with water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure to obtain the title compound as an oil (10.8 g, 86%) as a mixture of four isomers. This product was used for the next reaction without further purification.
上記の油状物 (10.8 g, 20.0 mmol) をトルエン (170 ml) および無水 酢酸 (5.66 ml, 60.0 mmol) に溶解し、 p -トルエンスルホン酸 1水和物 (3.21 g, 16.0 mmol) を加えて 100°Cで 15分加熱撹拌した。 反応液を 濃縮後、 残渣を酢酸ェチルで希釈し、 水で 3回飽和食塩水で洗浄後、 硫 酸ナトリウムで乾燥した。 溶媒を減圧下に留去して題記化合物 (6.77g, 65¾) を得た。 酢酸ェチルから再結晶して融点 176- 178°Cの暗赤色結晶を 得た。 The above oil (10.8 g, 20.0 mmol) was dissolved in toluene (170 ml) and acetic anhydride (5.66 ml, 60.0 mmol), and p-toluenesulfonic acid monohydrate (3.21 g, 16.0 mmol) was added. The mixture was heated and stirred at 100 ° C for 15 minutes. After concentrating the reaction solution, the residue was diluted with ethyl acetate, washed three times with water and a saturated saline solution, and dried over sodium sulfate. The solvent was distilled off under reduced pressure to obtain the title compound (6.77 g, 65%). Recrystallization from ethyl acetate gave dark red crystals with a melting point of 176-178 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3619, 3235, 2980, 2957, 1738, 1626, 1595, 1557, 1495 cnf1. Infrared absorption spectrum (IR) (in KBr): 3619, 3235, 2980, 2957, 1738, 1626, 1595, 1557, 1495 cnf 1 .
Ή-NMR (CDC13) : δ 1.27 (3Η, i, J = 7.0 Hz), 1.97-2.14 (2H, m) , 2.43 (3H, s), 2.51 (2H, t, J = 7.0 Hz), 2.72 (2H, t, J = 7.0 Hz), 3.21 (2H, t, J=6.2 Hz), 3.30 (2H, t, J=7.2 Hz), 4.16 (2H, q, J=7.4 Hz), 7.30 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8. Hz), 9.89 (1H, bs). Ή-NMR (CDC1 3): δ 1.27 (3Η, i, J = 7.0 Hz), 1.97-2.14 (2H, m), 2.43 (3H, s), 2.51 (2H, t, J = 7.0 Hz), 2.72 (2H, t, J = 7.0 Hz), 3.21 (2H, t, J = 6.2 Hz), 3.30 (2H, t, J = 7.2 Hz), 4.16 (2H, q, J = 7.4 Hz), 7.30 (2H , d, J = 8.4 Hz), 7.83 (2H, d, J = 8. Hz), 9.89 (1H, bs).
元素分析値: C22H23N306S3として 計算値 (¾) : C, 50.66; H, 4.44; , 8.06; S, 18.44. Elementary analysis: as C 22 H 23 N 3 0 6 S 3 Calculated value (¾): C, 50.66; H, 4.44;, 8.06; S, 18.44.
実測値 ) : C, 50.44; H, 4.32; Pi, 7.91; S, 18.27. Obtained): C, 50.44; H, 4.32; Pi, 7.91; S, 18.27.
実施例 2 - 69 Example 2-69
3- [(3 - メチルフエニル)スルホニル]ィ ミ ノ }- 1, 6-ジォキソ -2, 3, 7, 8 -テトラヒドロ - 1H, 6H-シク口ペン夕 [b]イミダゾ [1, 5-d] [1,4] チアジン -4-ィル)スルファニル] プロピオン酸ェチル 3-[(3-Methylphenyl) sulfonyl] imino} -1, 6-dioxo-2,3,7,8-tetrahydro-1H, 6H-cyclohexyl [b] imidazo [1,5-d] [1,4] thiazine-4-yl) sulfanyl] ethyl propionate
式 (VII) において 、 R6が縮環したシクロペンテノン環、 R1Qが _ZR15 (Z が S02、 R15が p-トルィル基)、 Rがェチル基である化合物の合成 In formula (VII), the synthesis of cyclopentenone ring, R 1Q is _ZR 15 (Z is S0 2, R 15 is p- Toruiru group), and R is a Echiru group R 6 is condensed
参考例, 3- 2で合成した化合物 (5.00 g, 11.6 mmol) を THF (50.0 ml) および DMF (50.0 ml) に溶解し、 2-ブロモ -1, 3-シクロペンタンジオン (2.47 g, .0 mmol) およびトリェチルァミン (4.87 ml, 34.9 mmol) を加えて室温で 20時間撹拌した。反応液を濃縮後、残渣を酢酸ェチルで 希釈し、 水、 および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に留去して表題化合物の油状物を四種異性体の混合物とし て得た。 本品はこれ以上精製することなく次の反応に使用した。 The compound synthesized in Reference Example 3-2 (5.00 g, 11.6 mmol) was dissolved in THF (50.0 ml) and DMF (50.0 ml), and 2-bromo-1,3-cyclopentanedione (2.47 g, .0 mmol) and triethylamine (4.87 ml, 34.9 mmol), and the mixture was stirred at room temperature for 20 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed with water and saturated saline, and then dried over sodium sulfate. The solvent was distilled off under reduced pressure to obtain an oily substance of the title compound as a mixture of four isomers. This product was used for the next reaction without further purification.
上記の油状物をトルエン (50.0 ml) および無水酢酸 (3.29 ml, 34.9 mmol) に溶解し、 P-トルエンスルホン酸 1水和物 (1.86 g, 9.31 mmol) を加えて lOOtで 4時間加熱撹拌した。 反応液を濃縮後、 残渣を酢酸ェ チルで希釈し、 水で 3回飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し た。 溶媒を減圧下に留去して題記化合物 (1.57 g, 27%) を得た。 酢酸ェ チルから再結晶して融点 207- 208での暗赤色結晶を得た。 The above oil was dissolved in toluene (50.0 ml) and acetic anhydride (3.29 ml, 34.9 mmol), P-toluenesulfonic acid monohydrate (1.86 g, 9.31 mmol) was added, and the mixture was heated and stirred with lOOt for 4 hours. . After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed three times with water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure to obtain the title compound (1.57 g, 27%). Recrystallization from ethyl acetate gave dark red crystals with mp 207-208.
赤外吸収スペクトル(IR) (KBr中) リ: 3202, 2980, 2926, 1769, 1748, 1698, 1626, 1605, 1541, 1530 cm"1. Infrared absorption spectrum (IR) (in KBr): 3202, 2980, 2926, 1769, 1748, 1698, 1626, 1605, 1541, 1530 cm " 1 .
Ή-NMR (CDC13) : δ 1.26 (3Η, t, J=7.0 Hz), 2.43 (3H, s), 2.54-2.62 (2H, m), 2.71 (2H, t, J = 7.4Hz), 3.20-3.28 (2H, m) , 3.26 (2H, t, J = 7.2 Hz), 4.16 (2H, q, J=7.2 Hz), 7.31 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8. Hz), 9.91 (1H, bs). Ή-NMR (CDC1 3): δ 1.26 (3Η, t, J = 7.0 Hz), 2.43 (3H, s), 2.54-2.62 (2H, m), 2.71 (2H, t, J = 7.4Hz), 3.20 -3.28 (2H, m), 3.26 (2H, t, J = 7.2 Hz), 4.16 (2H, q, J = 7.2 Hz), 7.31 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8. Hz), 9.91 (1H, bs).
元素分析値: C21H21N306S3として Elemental analysis: as C 21 H 21 N 3 0 6 S 3
計算値 (%) : C, 49.69; H, 4.17; N, 8.28; S, 18.95. 実測値 (%) : C, 49.69; H, 4.19; N, 7.90; S, 18.57. Calculated value (%): C, 49.69; H, 4.17; N, 8.28; S, 18.95. Found (%): C, 49.69; H, 4.19; N, 7.90; S, 18.57.
実施例 2-70 Example 2-70
-讓 R (300MHz, CDC13) .: δ 1.27 (3Η, t, J= 7.1 Hz), 1.71 (4H, m) , 1.98 (2H, m), 2.42 (3H, s), 2.70 (2H, t, J= 7.5 Hz), 2.75 (2H, m) , 3.20 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.83 (2H, d, J= 8.3 Hz), 9.60 (1H, br). -Lease R (300MHz, CDC1 3 ).: Δ 1.27 (3Η, t, J = 7.1 Hz), 1.71 (4H, m), 1.98 (2H, m), 2.42 (3H, s), 2.70 (2H, t , J = 7.5 Hz), 2.75 (2H, m), 3.20 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.83 (2H, d, J = 8.3 Hz), 9.60 (1H, br).
実施例 2-71 Example 2-71
Ή-NMR (DMSO) : δ 1.80-1.92 (2H, m) , 2.31-2.38 (2H, m) , 2.58 (3H, s), 2.70 (2H, t, J = 7.0Hz), 3.08 (2H, t, J = 6.1Hz), 3.60 (2H, t, J = 7.0Hz), 3.63 (3H, s) , 7.33-7.43 (3H, m) , 7.91-7.96 (1H, m) . 実施例 2 - 72 Ή-NMR (DMSO): δ 1.80-1.92 (2H, m), 2.31-2.38 (2H, m), 2.58 (3H, s), 2.70 (2H, t, J = 7.0Hz), 3.08 (2H, t) , J = 6.1Hz), 3.60 (2H, t, J = 7.0Hz), 3.63 (3H, s), 7.33-7.43 (3H, m), 7.91-7.96 (1H, m).
Ή-NM (DMSO) : 61.73-1.79 (2H, m) , 1.84-1.94 (2H, m) , 2.32-2.40 (2H, m), 3.19 (2H, t, J = 6.1Hz), 3.60 (2H, t, J = 6.4Hz) , 7.40-7.49 (2H, m), 7.56-7.64 (1H, m), 8.11 (1H, br d, J = 8.0Hz) . Ή-NM (DMSO): 61.73-1.79 (2H, m), 1.84-1.94 (2H, m), 2.32-2.40 (2H, m), 3.19 (2H, t, J = 6.1Hz), 3.60 (2H, m t, J = 6.4Hz), 7.40-7.49 (2H, m), 7.56-7.64 (1H, m), 8.11 (1H, br d, J = 8.0Hz).
実施例 2 - 73 Example 2-73
Ή-NMR (DMSO) : δ 1.85-1.96 (2H, m) , 2.35-2.43 (2H, m) , 2.38 (3H, s), 2.72 (2H, t, J-7.0Hz), 3.09 (2H, t, J = 5.9Hz), 3.20 (2H, t, J = 7.0Hz), 3.63 (3H, s), 7.35 (2H, d, J = 8.1Hz), 7.81 (2H, d, J = 8.1Hz) . 実施例 2-74 Ή-NMR (DMSO): δ 1.85-1.96 (2H, m), 2.35-2.43 (2H, m), 2.38 (3H, s), 2.72 (2H, t, J-7.0Hz), 3.09 (2H, t) , J = 5.9Hz), 3.20 (2H, t, J = 7.0Hz), 3.63 (3H, s), 7.35 (2H, d, J = 8.1Hz), 7.81 (2H, d, J = 8.1Hz). Example 2-74
Ή-NMR (300MHz, CDC13) : δ 1.26 (3H, t, J= 7.1 Hz), 1.68 (2H, m) ; Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3H, t, J = 7.1 Hz), 1.68 (2H, m);
1.84 (1H, m), 2.28 (1H, m) , 2.32 (2H, m) , 2. 2 (3H, s), 2.68 (2H, t; 1.84 (1H, m), 2.28 (1H, m), 2.32 (2H, m), 2.2 (3H, s), 2.68 (2H, t ;
J= 7.4 Hz), 3.23 (2H, dt, J= 5.0, 7.0 Hz), 3.71 (1H, brt, J= 6.5 Hz) ; J = 7.4 Hz), 3.23 (2H, dt, J = 5.0, 7.0 Hz), 3.71 (1H, brt, J = 6.5 Hz) ;
4.16 (2H, q, J= 7.1 Hz), 6.36 (1H, brt, J- 4.2 Hz), 7.30 (2H, d, J: 4.16 (2H, q, J = 7.1 Hz), 6.36 (1H, brt, J- 4.2 Hz), 7.30 (2H, d, J :
8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.70 (1H, br). 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.70 (1H, br).
元素分析値: C22H25N305S3として計算値: C, 52.05; H, 4.96; N, 8.28; S,Elemental analysis: C 22 H 25 N 3 0 5 S 3 Calculated: C, 52.05; H, 4.96 ; N, 8.28; S,
18.95 (%) .実測値: 52.01; H, 4.71; N, 8.34; S, 18.71 (%) . 実施例 2-75 18.95 (%). Found: 52.01; H, 4.71; N, 8.34; S, 18.71 (%).
8-[(3-エトキシ- 3-ォキソプロピル)スルファニル ]-6- (2-メトキシ- 2-ォ キソェチル) -1-{ [(4-メチルフエニル)スルホニル]ィミノ卜 3-ォキソ - 2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 5-カルボン酸メチル 実施例卜 52 で合成した化合物(8.40 g, 14.011111101)をトルェン(1601111) に溶解し、 無水酢酸(4.05 ml, 42.1 Miol)および P-トルエンスルホン酸 水和物(2.67 g, 14.0 jnmol) 存在下 80°C で 1時間攪拌した。 反応液を 濃縮後、 酢酸ェチル(300 ml)で希釈し、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥、 濃縮して赤色油状物を得た。 これをシリカゲル (80 g)のクロマトグラフィー (酢酸ェチルーへキサン、 3:7-5:5 および 酢酸ェチルートルエン、 5:5) により精製して表題化合物 (5.16 g, 8.84 龍 ol)を橙色粉末として得た。 8-[(3-ethoxy-3-oxopropyl) sulfanyl] -6- (2-methoxy-2-oxoethyl) -1-{[(4-methylphenyl) sulfonyl] iminoto 3-oxo -Methyl 2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-5-carboxylate Dissolve the compound synthesized in Example 52 (8.40 g, 14.011111101) in toluene (1601111) Then, the mixture was stirred at 80 ° C for 1 hour in the presence of acetic anhydride (4.05 ml, 42.1 Miol) and P-toluenesulfonic acid hydrate (2.67 g, 14.0 jnmol). The reaction mixture was concentrated, diluted with ethyl acetate (300 ml), washed with water and saturated saline, dried over sodium sulfate, and concentrated to obtain a red oil. This was purified by chromatography on silica gel (80 g) (ethyl acetate-hexane, 3: 7-5: 5 and ethyl acetate-toluene, 5: 5) to give the title compound (5.16 g, 8.84 dragonol) as an orange powder As obtained.
Ή-NMR (300MHz, CDC13): δ 1.27 (3Η, t, J= 7.2 Hz), 2.43 (3H, s), 2.69 (2H, t, J= 7.2 Hz), 3.23 (2H, t, J= 7.5 Hz), 3.34 (2H, s), 3.75 (3H, s), 3.85 (3H, s), 4.16 (2H, q, J= 7.0 Hz), 7.31 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.76 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 1.27 (3Η, t, J = 7.2 Hz), 2.43 (3H, s), 2.69 (2H, t, J = 7.2 Hz), 3.23 (2H, t, J = 7.5 Hz), 3.34 (2H, s), 3.75 (3H, s), 3.85 (3H, s), 4.16 (2H, q, J = 7.0 Hz), 7.31 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.76 (1H, br).
元素分析値: C23H25N309S3として計算値: C, 47.33; H, 4.32; N, 7.20; S, 16.48 (%) , 実測値: 47.28; H, 4.11; N, 7.11; S, 16.45 (%) . 実施例 2-76 Elemental analysis: C 23 H 25 N 3 0 9 S 3 Calculated: C, 47.33; H, 4.32 ; N, 7.20; S, 16.48 (%), Found: 47.28; H, 4.11; N , 7.11; S, 16.45 (%). Example 2-76
3 - [(8, 8-ジメチル- 1, 6-ジォキソ -3- {[(4-メチルフエニル)スルホニル] ィミノ}- 2, 3, 6, 7, 8, 9-へキサヒド口- 1 -ィミダゾ [5, 1-c] [1, 4]ベンゾチ 7ジン -4 -ィル)スルファニル]プロピオン酸ェチル 3-[(8,8-dimethyl-1,6-dioxo-3-{[(4-methylphenyl) sulfonyl] imino} -2,3,6,7,8,9-hexahydrid-1-imidazo [ 5, 1-c] [1,4] benzothi 7zin-4-yl) sulfanyl] ethyl ester propionate
参考例 3— 2で合成した化合物 (8.00 g, 18.6 mmol) を THF (80.0 ml) および DMF (80.0 ml) に溶解し、 参考例 42 で得られた 2-ブロモ -5,5- ジメチルシクロへキサン- 1, 3-ジオン (4.90 g, 22.3 mmol) およびトリ ェチルァミン(7.79 ml, 55.9 mmol)を加えて室温で 20.5時間撹拌した。 反応液を濃縮後、 残渣を酢酸ェチルで希釈し、 水、 および飽和食塩水で 洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に留去した。 残渣を トルエン (80.0 ml) および無水酢酸 (5.27 ml, 55.9 mmol) に溶解し、 p -トルエンスルホン酸 1水和物 (2.98 g, 14.9 mmol) を加えて 100°Cで 1 時間加熱撹拌した。 反応液を濃縮後、 残渣を酢酸ェチルで希釈し、 水 で 3回飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下 に留去して題記化合物 (3.13 g, 31¾) を得た。 酢酸ェチルから再結晶し て融点 227- 229°Cの暗赤色結晶を得た。 The compound synthesized in Reference Example 3-2 (8.00 g, 18.6 mmol) was dissolved in THF (80.0 ml) and DMF (80.0 ml), and the 2-bromo-5,5-dimethylcyclo obtained in Reference Example 42 was dissolved. Xan-1,3-dione (4.90 g, 22.3 mmol) and triethylamine (7.79 ml, 55.9 mmol) were added, and the mixture was stirred at room temperature for 20.5 hours. After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed with water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure. The residue was dissolved in toluene (80.0 ml) and acetic anhydride (5.27 ml, 55.9 mmol), p-toluenesulfonic acid monohydrate (2.98 g, 14.9 mmol) was added, and the mixture was heated and stirred at 100 ° C for 1 hour. After concentration of the reaction solution, the residue was diluted with ethyl acetate, washed three times with water and saturated saline, and dried over sodium sulfate. Solvent under reduced pressure The title compound (3.13 g, 31¾) was obtained. Recrystallization from ethyl acetate gave dark red crystals with a melting point of 227-229 ° C.
IR (KBr) : 3221, 2963, 1757, 1738, 1655, 1626, 1557 cm'1. IR (KBr): 3221, 2963 , 1757, 1738, 1655, 1626, 1557 cm '1.
!H-NMR (CDC13) : 61.12 (6H, s), 1.27 (3H, t, J = 7.2 Hz), 2.36 (2H, s), 2. 3 (3H, s), 2.73 (2H, t, J = 7.2 Hz), 3.08 (2H, s), 3.30 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.83 (2H, d, J = 8.7 Hz), 9.86 (1H, bs). ! H-NMR (CDC1 3) : 61.12 (6H, s), 1.27 (3H, t, J = 7.2 Hz), 2.36 (2H, s), 2. 3 (3H, s), 2.73 (2H, t, J = 7.2 Hz), 3.08 (2H, s), 3.30 (2H, t, J = 7.2 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.83 (2H, d, J = 8.7 Hz), 9.86 (1H, bs).
元素分析値: C24H27N306S3として Elementary analysis: as C 24 H 27 N 3 0 6 S 3
計算値 ) : C, 52.44; H, 4.95; N, 7.64; S, 17.50. Calculated values): C, 52.44; H, 4.95; N, 7.64; S, 17.50.
実測値 ) : C, 52.72; H, 4.94; N, 7.34; S, 17.11. Found): C, 52.72; H, 4.94; N, 7.34; S, 17.11.
実施例 2 - 77 Example 2-77
3- [(9, 9-ジメチル -1, 6-ジォキソ- 3- {[(4-メチルフエニル)スルホニル] ィミノ卜 2, 3, 6,7, 8, 9-へキサヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]ベンゾチ ァジン- 4_ィル)スルファニル]プロピオン酸ェチル 3-[(9,9-dimethyl-1,6-dioxo-3-{[(4-methylphenyl) sulfonyl] iminoto 2,3,6,7,8,9-hexahydro-1H-imidazo [5, 1-c] [1,4] Benzothiazin-4-yl) sulfanyl] ethyl propionate
実施例 2- 76と同様の方法により、 2-ブロモ -5, 5-ジメチルシクロへキ サン- 1, 3-ジオンの代わりに参考例 43で得られた 2-ブロモ -4, 4-ジメチ ルシクロへキサン- 1, 3 -ジオン (4.90 g, 22.3 mmol) を用いて、 参考例 3— 2で合成した化合物 (8.00 g, 18.6 mmol) から題記化合物 (4.92 g, 48¾) を合成した。 酢酸ェチルから再結晶して融点 202- 204 の暗赤色結 晶を得た。 In the same manner as in Example 2-76, 2-bromo-4,4-dimethylcycloalkyl obtained in Reference Example 43 was used instead of 2-bromo-5,5-dimethylcyclohexane-1,3-dione. The title compound (4.92 g, 48¾) was synthesized from the compound (8.00 g, 18.6 mmol) synthesized in Reference Example 2-2 using hexane-1,3-dione (4.90 g, 22.3 mmol). Recrystallization from ethyl acetate gave dark red crystals with a melting point of 202-204.
IR (KBr) V: 3227, 2976, 2932, 2870, 1759, 1738, 1626, 1593, 1557 cm-1. IR (KBr) V: 3227, 2976, 2932, 2870, 1759, 1738, 1626, 1593, 1557 cm -1 .
^-NMR (CDCI3) : 51.15 (6H, s), 1.26 (3H, t, J = 7. Hz) , 1.86 (2H, t, J=6.2 Hz), 2.42 (3H, s), 2.72 (2H, t, J=7.4 Hz), 3.22 (2H, t, J=6.4 Hz), 3.30 (2H, t, J = 7.2 Hz), 4.16 (2H, q, J = 6.8 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.86 (1H, bs) . ^ -NMR (CDCI3): 51.15 (6H, s), 1.26 (3H, t, J = 7. Hz), 1.86 (2H, t, J = 6.2 Hz), 2.42 (3H, s), 2.72 (2H, s) t, J = 7.4 Hz), 3.22 (2H, t, J = 6.4 Hz), 3.30 (2H, t, J = 7.2 Hz), 4.16 (2H, q, J = 6.8 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.86 (1H, bs).
元素分析値: C24H27N306S3として Elemental analysis: C 24 H 27 N 3 0 6 S 3
計算値 (¾) : C, 52.44; H, 4.95; N, 7.64; S, 17.50. Calculated value (¾): C, 52.44; H, 4.95; N, 7.64; S, 17.50.
実測値 (¾) : C, 52.32; H, 4.68; N, 7.45; S, 17.43. 実施例 2 - 78 Found (¾): C, 52.32; H, 4.68; N, 7.45; S, 17.43. Example 2-78
3- [(1, 6 -ジォキソ -8-メチル -3- {[(4-メチルフエニル)スルホニル]ィミ ハ- 1, 3, 6, 7, 8, 9-へキサヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジ ン- 4-ィル)スルファニル]プロピオン酸ェチル 3-[(1,6-dioxo-8-methyl-3-{[(4-methylphenyl) sulfonyl] imi--1,3,6,7,8,9-hexahydro-1H-imidazo [5, 1-c] [1,4] benzothiazin-4-yl) sulfanyl] ethyl propionate
実施例 2-76と同様の方法により、 2-ブロモ -5, 5-ジメチルシクロへキ サン- 1, 3-ジオンの代わりに参考例 44で得られた 2-ブロモ -5-メチルシ クロへキサン- 1, 3 -ジオン (6.87 g, 33.5 mmol) を用いて、 参考例 3— 2 で合成した化合物 (12.0 g, 27.9 mmol) から題記化合物 (10.4 g, 69%) を合成した。 酢酸ェチルから再結晶して融点 202- 204°Cの暗赤色結晶を 得た。 In the same manner as in Example 2-76, 2-bromo-5-methylcyclohexane-1,3-dione was replaced with 2-bromo-5-methylcyclohexane obtained in Reference Example 44. Using 1,3-dione (6.87 g, 33.5 mmol), the title compound (10.4 g, 69%) was synthesized from the compound (12.0 g, 27.9 mmol) synthesized in Reference Example 2-2. Recrystallization from ethyl acetate gave dark red crystals with a melting point of 202-204 ° C.
IR (KBr) V: 3250, 2959, 2928, 2907, 2874, 2255, 1742, 1651, 1622, 1595, 1557 cm一1. IR (KBr) V: 3250, 2959, 2928, 2907, 2874, 2255, 1742, 1651, 1622, 1595, 1557 cm- 1 .
Ή-NMR (CDC13) : (51.14 (3H, d, J = 6.3 Hz), 1.26 (3H, t, J = 7.2 Hz), 2.12-2.30 (2H, m) , 2.43 (3H, s) , 2.56 (1H, t, J = 10.5 Hz), 2.62 (1H, d, J = 5.1 Hz), 2.72 (2H, t, J = 6.9 Hz), 3.29 (2H, t, J = 7.2 Hz), 3.58 (1H, dd, J = 19.5, 3.9 Hz), 4.16 (2H, q, J = 7.2 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.83 (2H, d, J=8.4 Hz), 9.87 (1H, bs). Ή-NMR (CDC1 3 ): (51.14 (3H, d, J = 6.3 Hz), 1.26 (3H, t, J = 7.2 Hz), 2.12-2.30 (2H, m), 2.43 (3H, s), 2.56 (1H, t, J = 10.5 Hz), 2.62 (1H, d, J = 5.1 Hz), 2.72 (2H, t, J = 6.9 Hz), 3.29 (2H, t, J = 7.2 Hz), 3.58 (1H , Dd, J = 19.5, 3.9 Hz), 4.16 (2H, q, J = 7.2 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.87 (1H , bs).
元素分析値: C23H25N306S3として Elementary analysis: as C 23 H 25 N 3 0 6 S 3
計算値 (%) : C, 51.57; H, 4.70; N, 7.84; S, 17.96. Calculated value (%): C, 51.57; H, 4.70; N, 7.84; S, 17.96.
実測値 (%) : C, 51.42.; H, 4.60; N, 7.70; S, 17.99. Found (%): C, 51.42 .; H, 4.60; N, 7.70; S, 17.99.
実施例 3 - 1 Example 3-1
式 (V I I I ) において R 5が n-ペンチル基、 R 6がメチル基、 R 1 0が - Z R 1 5 (Zが S 02、 R 1 5が P-トルィル基) である化合物の合成 実施例 2-1 で合成した化合物 (3.37 g, 6.27 mmol) を THF (40 ml) およびメ夕ノール(40 ml)に溶解し、水酸化力リゥム(2.07 g, 31.3 mmol) を加えて室温で 4 5分間撹拌した。 反応液に 1N塩酸 (32 ml) を加えて 混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を濾取し、 5 0 %メ 夕ノール水で洗浄後、乾燥して標題化合物の橙色粉末(2.67 g、 6.10 mmol) を得た。 'Η-醒 R (300 MHz, 重クロ口ホルム中): δ 0.88 (3Η, t, J= 6.8Hz), 1.29 (4H, m), 1.46 (2H, brquint, J= 7.2 Hz), 2.14 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 7.20 (2H, dm, J= 7.2 Hz), 7.83 (2H, d, J= 8.3 Hz), 9.36 (1H, br), 12.65 (1H, brs). R 5 is n- pentyl group in formula (VIII), R 6 is a methyl group, R 1 0 is - Synthesis Example of ZR 1 5 (Z is S 0 2, R 1 5 is P- Toruiru group) compound is The compound synthesized in 2-1 (3.37 g, 6.27 mmol) was dissolved in THF (40 ml) and methanol (40 ml), and hydroxylated water (2.07 g, 31.3 mmol) was added. Stirred for minutes. The reaction mixture was mixed with 1N hydrochloric acid (32 ml) and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with 50% methanol water, and dried to give the title compound as an orange powder (2.67 g, 6.10 mmol). 'Η-wake R (300 MHz, in double-mouthed form): δ 0.88 (3Η, t, J = 6.8 Hz), 1.29 (4H, m), 1.46 (2H, brquint, J = 7.2 Hz), 2.14 ( 2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 7.20 (2H, dm, J = 7.2 Hz), 7.83 (2H, d, J = 8.3 Hz), 9.36 ( 1H, br), 12.65 (1H, brs).
元素分析値: C19H23N303S3 として計算値: C, 52.15; H, 5.30; , 9.60; S, 21.98 (%), 実測値; C, 52.21; H, 5.03; , 9.59; S, 21.84 (%) . 実施例 3 - 2 Elemental analysis: C 19 H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30;, 9.60; S, 21.98 (%), found; C, 52.21; H, 5.03;, 9.59; S, 21.84 (%). Example 3-2
式 (V I I I ) において R 5が n-へキシル基、 R 6が水素原子、 R 1 Qが - Z R 1 5 (2が3〇2、 R 1 5が p -トルィル基) である化合物の合成 実施例 2-2で合成した化合物 (1.44 g, 純度 93%、 2.49 mmol) を THF (15 ml) およびメタノール (15ml) に溶解し、 水酸化カリウム (0.67g, 10.1匪 ol) を加えて室温で 4 0分間撹拌した。反応液に 1 N塩酸(10 ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物の橙色粉末 (1.00 g、 2.29 mmol) を得た。 Cyclohexyl group in formula (VIII) R 5 is n-, R 6 is a hydrogen atom, R 1 Q is - ZR 1 5 (2 is 3_Rei 2, R 1 5 is p - Toruiru group) Synthesis embodiment, compound The compound synthesized in Example 2-2 (1.44 g, purity 93%, 2.49 mmol) was dissolved in THF (15 ml) and methanol (15 ml), and potassium hydroxide (0.67 g, 10.1 bandol) was added thereto. Stirred for 40 minutes. The reaction mixture was mixed with 1N hydrochloric acid (10 ml) and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give the title compound as an orange powder (1.00 g, 2.29 mmol).
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 0.87 (3Η, brt, J= 6.9 Hz), 1.27 (4H, brs), 1.30 (2H, i) , 1.49 (2H, brquint, J= 7.3 Hz), 2.19 (2H, brt, J= 7.0 Hz), 2.45 (3H, s), 6.95 (IE, brt, J= 0.5 Hz), 7.36 (2H, brd, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.50 (1H, br), 12.03 (1H, brs). Ή-NMR (300 MHz, in double-mouthed form): δ 0.87 (3Η, brt, J = 6.9 Hz), 1.27 (4H, brs), 1.30 (2H, i), 1.49 (2H, brquint, J = 7.3) Hz), 2.19 (2H, brt, J = 7.0 Hz), 2.45 (3H, s), 6.95 (IE, brt, J = 0.5 Hz), 7.36 (2H, brd, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.50 (1H, br), 12.03 (1H, brs).
元素分析値: C19H23N303S3 として計算値: C, 52.15; H, 5.30; , 9.60; S, 21.98 ( ), 実測値; C, 52.14; H, 5.13; N, 9.52; S, 21.81 (%) . 実施例 3-3 ' Elemental analysis: C 19 H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30 ;, 9.60; S, 21.98 (), Found; C, 52.14; H, 5.13 ; N, 9.52; S, 21.81 (%). Example 3-3 ′
式 (V I I I ) において R 5がエトキシカルポニル基、 R 6がメチル基、 R 1。がー Z R 1 5 (Zが S〇2、 R 1 5が p-トルィル基) である化合物の 合成 In the formula (VIII), R 5 is an ethoxycarbonyl group, R 6 is a methyl group, and R 1 . Synthesis of a compound of the formula ZR 15 (Z is S〇 2 and R 15 is a p-tolyl group)
実施例 2-3で合成した化合物 (1.02 g, 1.89 mmol) を THF (10 ml) およびエタノール(10 ml)に溶解し、ナ卜リウムェチラ一ト(257 mg, 3.78 mmol) を加えて室温で 8 0分間撹拌した。 反応液に 1 N 塩酸 (4.5 ml) および水 (5.5 ml) を加えて混和し、 室温で 1 0分間撹拌した。 生成し た沈澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物の橙色 粉末 (0.82 g、 1.86 mmol) を得た。 The compound synthesized in Example 2-3 (1.02 g, 1.89 mmol) was dissolved in THF (10 ml) and ethanol (10 ml), and sodium ethylate (257 mg, 3.78 mmol) was added. Stirred for 0 minutes. 1N hydrochloric acid (4.5 ml) And water (5.5 ml) were added and mixed, followed by stirring at room temperature for 10 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give the title compound as an orange powder (0.82 g, 1.86 mmol).
'Η -丽 R (300 MHz,重ジスチルスルホキシド中) : δ 1.20 (3Η, t, J= 7.0 Hz), 2.39 (3H, s), 2.72 (3H, s), 4.13 (2H, q, J= 7.0 Hz), 7.42 (2H, d, J= 8.3 Hz), 7.88 (2H, d, J= 8.3 Hz). 'Η-丽 R (300 MHz, in heavy distyrsulfoxide): δ 1.20 (3Η, t, J = 7.0 Hz), 2.39 (3H, s), 2.72 (3H, s), 4.13 (2H, q, J = 7.0 Hz), 7.42 (2H, d, J = 8.3 Hz), 7.88 (2H, d, J = 8.3 Hz).
元素分析値: C17H17N305S3 として計算値: C, 46.45; H, 3.90; N, 9.56; S, 21.89 ( ), 実測値; C, 46.72; H, 3.93; N, 9.32; S, 20, 98 (%) . 実施例 3 - 4 Elemental analysis: C 17 H 17 N 3 0 5 S 3 Calculated: C, 46.45; H, 3.90 ; N, 9.56; S, 21.89 (), Found; C, 46.72; H, 3.93 ; N, 9.32 ; S, 20, 98 (%). Example 3-4
式 (V I I I ) において R 5および R 6がともにメチル基、 R 1 ()が— Z R 1 5 ifiS O 2、 R 15が P-トルィル基) である化合物の合成 Both methyl groups R 5 and R 6 in formula (VIII), R 1 () is - Synthesis of ZR 1 5 ifiS O 2, compound R 15 is P- Toruiru group)
実施例 2-5 で合成した化合物 (5.00 g, 10.4 mmol) を THF (50 ml) およびメタノール(50 ml)に溶解し、水酸化力リゥム(2.74 g, 41.5 mmol) を加えて室温で 4 5分間撹拌した。 反応液に 1 N塩酸 (42 ml) を加えて 混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を濾取し、 5 0 %メ 夕ノール水で洗浄後、乾燥して標題化合物の赤色粉末(3.47 g、9.10 mmol) を得た。 The compound synthesized in Example 2-5 (5.00 g, 10.4 mmol) was dissolved in THF (50 ml) and methanol (50 ml), and a hydroxylic power (2.74 g, 41.5 mmol) was added. Stirred for minutes. The reaction mixture was mixed with 1N hydrochloric acid (42 ml) and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with 50% methanol water, and dried to give a red powder of the title compound (3.47 g, 9.10 mmol).
lH-NMR (300 MHz, 重クロ口ホルム-重ジメチルスルホキシド == 9 :.1 中) : <5 1.81 (3H, s), 2.42 (3H, d, J= 0.5 Hz), 2.45 (3H, s), 7.35 (2H, d, J= 8.1 Hz), 7.88 (2H, d, J= 8.3 Hz), 12.82 (2H, br). 元素分析値: C15H15N303S3 として計算値: C, 47.22; H, 3.96; , 11.01; S, 25.22 (%)、 実測値; C, 47.20; Η,.3.90; Ν, 10.73; S, 25.19 (%) . 実施例 3-5 lH-NMR (300 MHz, double-mouthed form-deuterated dimethyl sulfoxide == 9: in 0.1): <5 1.81 (3H, s), 2.42 (3H, d, J = 0.5 Hz), 2.45 (3H, s ), 7.35 (2H, d, J = 8.1 Hz), 7.88 (2H, d, J = 8.3 Hz), 12.82 (2H, br) elemental analysis:. calculated C 15 H 15 N 3 0 3 S 3 : C, 47.22; H, 3.96;, 11.01; S, 25.22 (%), measured value; C, 47.20; Η, .3.90; Ν, 10.73; S, 25.19 (%).
式 (V I I I ) において R 5がェチル基、 R 6がメチル基、 R 1。がー Z R l b (Zが S〇2、 R 1 5が P-トルィル基) である化合物の合成 In the formula (VIII), R 5 is an ethyl group, R 6 is a methyl group, and R 1 . Synthesis of a compound of the formula ZR lb (Z is S〇 2 , R 15 is P-tolyl group)
実施例 2-6 で合成した化合物 (4.15 g, 8.37 mmol) を THF (50 ml) およびメタノール(50 ml)に溶解し、水酸化力リゥム(2.21 g, 33.5 mmol) を加えて室温で 4 0分間撹拌した。 反応液に 1 N塩酸 (34 ml) および水 (16 ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を 濾取し、メタノール水で洗浄後、乾燥して標題化合物の橙色粉末(2.86 g、 7.23 mmol) を得た。 The compound (4.15 g, 8.37 mmol) synthesized in Example 2-6 was dissolved in THF (50 ml) and methanol (50 ml), and hydroxylated water (2.21 g, 33.5 mmol) was added. Stirred for minutes. 1N Hydrochloric acid (34 ml) and water (16 ml) were added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The resulting precipitate The crystals were collected by filtration, washed with aqueous methanol, and dried to give the title compound as an orange powder (2.86 g, 7.23 mmol).
lH-匪 R (200 MHz, 重クロ口ホルム中): δ 1.08 (3Η, t, J= 7.2Hz), 2.18 (2H, q, J= 7.6 Hz), 2.45 (6H, s), 7.36 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz) . lH-band R (200 MHz, in double-mouthed form): δ 1.08 (3Η, t, J = 7.2 Hz), 2.18 (2H, q, J = 7.6 Hz), 2.45 (6H, s), 7.36 (2H , d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz).
元素分析値: C16H17N303S3 として計算値: C, 48.59; H, 4.33; , 10.62; S, 24.32 (%), 実測値; C, 48.46; H, 4.29; , 10.58; S, 24.33 (%) . 実施例 3-6 Elemental analysis: Calculated for C 16 H 17 N 3 0 3 S 3 : C, 48.59; H, 4.33;, 10.62; S, 24.32 (%), found; C, 48.46; H, 4.29;, 10.58; S, 24.33 (%). Example 3-6
式 (V I I I ) において R 5および R 6がともにメチル基、 ^が— R 15 (2が302、 R 15が P-クロ口フエ二ル基) である化合物の合成 実施例 2-7で合成した化合物 (3.28 g, 6.53 mmol) を THF (35 ml) およぴメ夕ノ一ル(35 ml)に溶解し、水酸化力リゥム(1.73 g, 26.1 mmol) を加えて室温で 40分間撹拌した。 反応液に 1N塩酸 (26ml) を加えて 混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を濾取し、 メタノー ル水で洗浄後、 乾燥して標題化合物の橙色粉末 (2.56 g、 6.37 mmol) を 得た。 Formula (VIII) in which R 5 and R 6 are both methyl, ^ is - R 15 synthesized in (2 30 2, R 15 is P- black port Hue group) Synthesis Example of compounds wherein 2-7 The obtained compound (3.28 g, 6.53 mmol) was dissolved in THF (35 ml) and methanol (35 ml). Hydroxidizing rim (1.73 g, 26.1 mmol) was added, and the mixture was stirred at room temperature for 40 minutes. did. The reaction mixture was mixed with 1N hydrochloric acid (26 ml) and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with methanol water, and dried to give the title compound as an orange powder (2.56 g, 6.37 mmol).
'H-NMR (200 MHz, 重ジメチルスルホキシド中) : 1.76 (3H, brs), 2.32 (3H, brs), 7.69 (2H, dt, J= 8.4, 1.8 Hz), 7.99 (2H, dt, J= 8.8, 1.8 Hz). 'H-NMR (200 MHz, in heavy dimethyl sulfoxide): 1.76 (3H, brs), 2.32 (3H, brs), 7.69 (2H, dt, J = 8.4, 1.8 Hz), 7.99 (2H, dt, J = (8.8, 1.8 Hz).
元素分析値: C14H12N303S3C1 として計算値: C, 41.84; H, 3.01; N, 10.46; S, 23.93; C1, 8.82 (%),実測値; C, 41.96; H, 2.97; N, 10.40; S, 23.69; C1, 8.59 ( ) . Elemental analysis: C 14 H 12 N 3 0 3 S 3 C1 Calculated: C, 41.84; H, 3.01 ; N, 10.46; S, 23.93; C1, 8.82 (%), Found; C, 41.96; H , 2.97; N, 10.40; S, 23.69; C1, 8.59 ().
融点: 241.5-244.0°C. Melting point: 241.5-244.0 ° C.
実施例 3-7 Example 3-7
式 (V I I I ) において R 5および R 6がともにメチル基、 尺1 ()がー 2 R 15 (2が3〇2、 R 15が p_ブロモフエニル基) である化合物の合成 実施例 2-8で合成した化合物 (3.04 g, 5.56 mmol) を THF (30 ml) およびメタノール(30 ml)に溶解し、水酸化力リウム(し 10 g, 16· 7腿 ol) を加えて室温で 8 0分間撹拌した。 反応液に 1N塩酸 (16.7 ml) および 水 (13ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈澱物 を濾取し、 メタノール水で洗浄後、乾燥して標題化合物の橙色粉末(2.42 g、 5.42 mmol) を得た。 R 5 and R 6 are both methyl groups In the formula (VIII), the at scale 1 () gar 2 R 15 Synthesis Example 2-8 (2 3_Rei 2, R 15 is p_ Buromofueniru group) compound Dissolve the synthesized compound (3.04 g, 5.56 mmol) in THF (30 ml) and methanol (30 ml), add lithium hydroxide (10 g, 16.7 ol) and stir at room temperature for 80 minutes. did. 1N hydrochloric acid (16.7 ml) and Water (13 ml) was added and mixed, and the mixture was stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give the title compound as an orange powder (2.42 g, 5.42 mmol).
-丽 R (200 MHz, 重ジメチルスルホキシド中) : δ 1.75 (3Η, brs), 2.31 (3H, brs), 7.84 (2H, d, J= 8.8 Hz), 7.91 (2H, dt, J= 8.8 Hz). 元素分析値: C14Hl2N303S3Br として計算値: C, 37.67; H, 2.71; , 9.41; S, 21.55; Br, 17.90 (%)、実測値; C, 37.86; H, 2.67; N, 9.17; S, 21.50; Br, 17.63 (%) . -丽 R (200 MHz, in heavy dimethyl sulfoxide): δ 1.75 (3Η, brs), 2.31 (3H, brs), 7.84 (2H, d, J = 8.8 Hz), 7.91 (2H, dt, J = 8.8 Hz) .) elemental analysis: C 14 H l2 N 3 0 3 S 3 Br calculated: C, 37.67; H, 2.71 ;, 9.41; S, 21.55; Br, 17.90 (%), Found; C, 37.86; H, 2.67; N, 9.17; S, 21.50; Br, 17.63 (%).
融点: 242.5-247.5°C. Melting point: 242.5-247.5 ° C.
以下に実施例 3-1〜3- 7 で合成した化合物および同様にして合成した 化合物の一覧を示す。 The following is a list of compounds synthesized in Examples 3-1 to 3-7 and compounds synthesized in the same manner.
〔表 1 4〕(Table 14)
差替え用綁 [jM Q) 〔表 1 5〕For replacement 綁 (jM Q) (Table 15)
差替え用紙(規則 26》 〔表 1 6〕'Replacement Form (Rule 26) (Table 16) '
¾養え用涎(規則 26) 涎 Feeding saliva (Rule 26)
aim t9爾 Odf/ェ:) d 909Z60/Z0 OAV 以下に実施例 3-8〜3- 45で合成した化合物のデータを示す。 aim t9 Odf / e :) d 909Z60 / Z0 OAV The data of the compounds synthesized in Examples 3-8 to 3-45 are shown below.
実施例 3-8 Example 3-8
Ή-NMR (CDC13) : δ 0.87 (3Η, t, J = 6.8Hz), 1.23-1.33 (6H, m), 1.40-1.60 (2H, ηι), 2.19 (2H, br t, J=7.3Hz), 6.96 (1H, br s), 7.54-7.62 (2H, m), 7.64-7.72 (2H, m) , 7.88-7.93 (2H, m) , 12.1 (1H, br . s) . Ή-NMR (CDC1 3): δ 0.87 (3Η, t, J = 6.8Hz), 1.23-1.33 (6H, m), 1.40-1.60 (2H, ηι), 2.19 (2H, br t, J = 7.3Hz ), 6.96 (1H, br s), 7.54-7.62 (2H, m), 7.64-7.72 (2H, m), 7.88-7.93 (2H, m), 12.1 (1H, br.s).
元素分析値: C18H21N303S2 として計算値: C; 51.04, H; 5.00, N; 9.92Elemental analysis: C 18 H 21 N 3 0 3 S 2 Calculated: C; 51.04, H; 5.00 , N; 9.92
(%)、 実測値 C; 51.50, H; 4.87, N; 9.82 (%)' . (%), Found C; 51.50, H; 4.87, N; 9.82 (%) '.
実施例 3-9 Example 3-9
-麵 R (CDC13-DMS0- d62:1 , 200 MHz): δ 1.80 (3Η, d, J= 1.0 Hz), 2.39 (3H, d, J= 0.8 Hz), 7.58-74 (3H, m) , 8.02 (2H, dm, J= 7 Hz). 元素分析値: C14H13N303S3として計算値: C, 45.76; H, 3.57; N, 11.44; S, 26.18 (%), 実測値: C, 45.61; H, 3.56; N, 11.37; S, 26.21 (%) . 実施例 3 - 10 -麵 R (CDC1 3 -DMS0- d 6 2: 1, 200 MHz): δ 1.80 (3Η, d, J = 1.0 Hz), 2.39 (3H, d, J = 0.8 Hz), 7.58-74 (3H, . m), 8.02 (2H, dm, J = 7 Hz) elemental analysis: C 14 H 13 N 3 0 3 S 3 calculated: C, 45.76; H, 3.57 ; N, 11.44; S, 26.18 (% ), Found: C, 45.61; H, 3.56; N, 11.37; S, 26.21 (%). Examples 3-10
lH-NMR (CDC13, 200 MHz): δ 1.08 (3Η, t, J= 7.4 Hz), 2.18 (2H, q, J= 7.2 Hz), 2.45 (3H, s), 7.59 (3H, m) , 7.91 (2H, brd, J= 7.0 Hz). 元素分析値: C15H15N303S3として計算値: C, 47.22; H, 3.96; N, 11.01; S, 25.22 (%), 実測値: C, 47.25; H, 3.94; N, 11.30; S, 25.24 (%) . 実施例 3-11 lH-NMR (CDC1 3, 200 MHz): δ 1.08 (3Η, t, J = 7.4 Hz), 2.18 (2H, q, J = 7.2 Hz), 2.45 (3H, s), 7.59 (3H, m), . 7.91 (2H, brd, J = 7.0 Hz) elemental analysis: C 15 H 15 N 3 0 3 S 3 calculated: C, 47.22; H, 3.96 ; N, 11.01; S, 25.22 (%), measured Values: C, 47.25; H, 3.94; N, 11.30; S, 25.24 (%). Examples 3-11
Ή-NMR (CDCl3-DMS0-d69:1 , 200 MHz): δ 1.30 (3Η, t, J= 7.4 Hz), 2.90 (3H, s), 4.23 (2H, q, J= 6.8 Hz), 7.60 (2H, m) , 7.68 (1H, brt, J = 7.2 Hz), 8.05 (2H, d, J= 7.2 Hz). Ή-NMR (CDCl 3 -DMS0- d 6 9: 1, 200 MHz): δ 1.30 (3Η, t, J = 7.4 Hz), 2.90 (3H, s), 4.23 (2H, q, J = 6.8 Hz) , 7.60 (2H, m), 7.68 (1H, brt, J = 7.2 Hz), 8.05 (2H, d, J = 7.2 Hz).
元素分析値: C16H15N305S3 として計算値: 45.16; H, 3.55; N, 9.88 (%), 実測値: C, 45.26; H, 3.67; N, 9.54 ( ) . Elemental analysis: C 16 H 15 N 3 0 5 S 3 Calculated: 45.16; H, 3.55; N , 9.88 (%), Found: C, 45.26; H, 3.67 ; N, 9.54 ().
実施例 3-12 Example 3-12
Ή-NMR (CDClrDMS0-d6= 9:1, 300 MHz): δ 2.43 (3Η, d, J= 1.1 Hz), 2.45 (3H, s), 5.01 (1H, q, J= 1.5 Hz), 7.36 (2H, d, J= 8.3 Hz), 7.88 (2H, d, J= 8.3 Hz), 12.78, (1H, br). Ή-NMR (CDCl r DMS0-d 6 = 9: 1, 300 MHz): δ 2.43 (3Η, d, J = 1.1 Hz), 2.45 (3H, s), 5.01 (1H, q, J = 1.5 Hz) , 7.36 (2H, d, J = 8.3 Hz), 7.88 (2H, d, J = 8.3 Hz), 12.78, (1H, br).
元素分析値: C14H13N303S3として計算値: C, 45.76; H, 3.57; , 11.44; S, 26.18 (%), 実測値: C, 45.82; H, 3.64; N, 11.48; S, 26.06 ( ) . 実施例 3-13 Elemental analysis: Calculated as C 14 H 13 N 3 0 3 S 3: C, 45.76; H, 3.57;, 11.44; S, 26.18 (%), Found: C, 45.82; H, 3.64; N, 11.48; S, 26.06 ().
酢酸ェチルから再結晶して融点 225- 227°Cの橙色結晶を得た。 Recrystallization from ethyl acetate gave orange crystals with a melting point of 225-227 ° C.
赤外吸収スペクトル(IR) (KBr 中) リ: 3320-2700, 2969, 2930, 2868, 1713, 1634, 1580, 1514 cm— Infrared absorption spectrum (IR) (in KBr): 3320-2700, 2969, 2930, 2868, 1713, 1634, 1580, 1514 cm—
Ή-NMR (CDC13) : δ 0.930 (3Η, t, J = 7.4 Hz), 1.38-1.60 (2H, m) , 2.14 (2H, t, J = 8.2 Hz), 2.46 (6H, s), 7.36 (2H, d, 7=8.0 Hz), 7.79 (2H, d, J = 8.4 Hz), 9.38 (1H, bs) . Ή-NMR (CDC1 3): δ 0.930 (3Η, t, J = 7.4 Hz), 1.38-1.60 (2H, m), 2.14 (2H, t, J = 8.2 Hz), 2.46 (6H, s), 7.36 (2H, d, 7 = 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz), 9.38 (1H, bs).
元素分析値: C17H19N303S3として Elementary analysis: as C 17 H 19 N 3 0 3 S 3
計算値 ( : C, 49.86; H, 4.68; N, 10.26; S, 23.49. Calculated values (: C, 49.86; H, 4.68; N, 10.26; S, 23.49.
実測値 (¾) : C, 49.71; H, 4.57; N, 10.10; S, 23. 6. Found (¾): C, 49.71; H, 4.57; N, 10.10; S, 23. 6.
実施例 3 - 14 Example 3-14
】H -薩 R (CDC13, 200 MHz): δ 1.10 (6H, d, J= 7.0 Hz), 2.45 (3H, s), 2.48 (3H, s), 3.00 (1H, quint, J= 7.0 Hz), 7.36 (2H, d, J= 8.0 Hz), 7.79 (2H, d, J= 8.4 Hz), 9.5 (1H, br). ] H - Hokusatsu R (CDC1 3, 200 MHz) : δ 1.10 (6H, d, J = 7.0 Hz), 2.45 (3H, s), 2.48 (3H, s), 3.00 (1H, quint, J = 7.0 Hz ), 7.36 (2H, d, J = 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz), 9.5 (1H, br).
元素分析値: C17HigN303S3として計算値: C, 49.86; H, 4.68; N, 10.26; S, 23.49 (%) 実測値: C, 49.80; H, 4.54; N, 10.21; S, 23.54 (%) . 実施例 3 - 15 Elemental analysis: C 17 H ig N 3 0 3 S 3 Calculated: C, 49.86; H, 4.68 ; N, 10.26; S, 23.49 (%) Found: C, 49.80; H, 4.54 ; N, 10.21 S, 23.54 (%). Examples 3 to 15
酢酸ェチル-メタノールから再結晶して融点 192- 194°Cの橙色結晶を得 た。 Recrystallization from ethyl acetate-methanol afforded orange crystals with a melting point of 192-194 ° C.
赤外吸収スペクトル(IR) (KBr 中) レ : 3320-2600, 2953, 2928, 2865, 1715, 1638, 1582, 1518 cm—1. Infrared absorption spectrum (IR) (in KBr) Re: 3320-2600, 2953, 2928, 2865, 1715, 1638, 1582, 1518 cm- 1 .
'Η-醒 (CDC13) : δ 0.901 (3Η, t, J = 7.0 Hz), 1.24-1.50 (4H, m) , 2.15 (2H, d, J = 8.0 Hz), 2.45 (6H, s), 7.36 (2H, d, J = 8.0 Hz), 7.78 (2H, d, J = 8. Hz), 9.32 (1H, bs). '.Eta. Awakening: (CDC1 3): δ 0.901 ( 3Η, t, J = 7.0 Hz), 1.24-1.50 (4H, m), 2.15 (2H, d, J = 8.0 Hz), 2.45 (6H, s), 7.36 (2H, d, J = 8.0 Hz), 7.78 (2H, d, J = 8. Hz), 9.32 (1H, bs).
元素分析値: C18H21N303S3として Elemental analysis: as C 18 H 21 N 3 0 3 S 3
計算値 ) : C, 51.04; H, 5.00; N, 9.92; S, 22.71. Calculated)): C, 51.04; H, 5.00; N, 9.92; S, 22.71.
実測値 (¾) : C, 50.98; H, 4.86; N, 9.86; S, 22.77. Found (¾): C, 50.98; H, 4.86; N, 9.86; S, 22.77.
実施例 3 - 16 酢酸ェチル -ジィソプロピルエーテルから再結晶して融点 231-233での 橙色結晶を得た。 Example 3-16 Recrystallization from ethyl acetate-diisopropyl ether gave orange crystals with a melting point of 231-233.
赤外吸収スペクトル(IR) (KBr 中) リ: 3500-2400, 2951 , 1705, 1634, 1574, 1508 cm— Infrared absorption spectrum (IR) (in KBr) Re: 3500-2400, 2951, 1705, 1634, 1574, 1508 cm—
Ή-NMR (CDC13) : δ 0.925 (6Η, d, J = 6.6 Hz), 1.68-1.90 (1H, m) , 2.05 Ή-NMR (CDC1 3): δ 0.925 (6Η, d, J = 6.6 Hz), 1.68-1.90 (1H, m), 2.05
(2H, d, J = 7.4 Hz) , 2. 6 (6H, s), 7.36 (2H, d, J = 8.0 Hz), 7.80 (2H, d, J = 8. Hz), 9.46 (1H, bs) . (2H, d, J = 7.4 Hz), 2.6 (6H, s), 7.36 (2H, d, J = 8.0 Hz), 7.80 (2H, d, J = 8. Hz), 9.46 (1H, bs ).
元素分析値: Cl8H2lN303S3'0.7H20として Elementary analysis: as C l8 H 2l N 3 0 3 S 3 '0.7H 2 0
計算値 ( : C, 49.57; H, 5.18; N, 9.63; S, 22.05. Calculated values (: C, 49.57; H, 5.18; N, 9.63; S, 22.05.
実測値 (¾) : C, 49.67; H, 4.90; N, 9.37; S, 21.70. Found (¾): C, 49.67; H, 4.90; N, 9.37; S, 21.70.
実施例 3-17 Example 3-17
エタノール-ジェチルエーテルから再結晶して融点 211-213 の橙色結 晶を得た。 The crystals were recrystallized from ethanol-ethyl ether to obtain orange crystals having a melting point of 211-213.
赤外吸収スペクトル(IR) (KBr 中) リ: 3400-2700, 2949, 2926, 2860, 1715, 1645, 1595, 1578, 1514 cm—1. Infrared absorption spectrum (IR) (in KBr): 3400-2700, 2949, 2926, 2860, 1715, 1645, 1595, 1578, 1514 cm— 1 .
Ή-NMR (CDC13) : δ 0.875 (3Η, t, J = 6.8 Hz), 1.20-1.52 (8H, m), 2.15 (2H, t, J-8.2 Hz), 2.45 (6H, s), 7.36 (2H, d, J-8.4 Hz), 7.80 (2H, d, J = 8.4 Hz), 9.38 (1H, bs). Ή-NMR (CDC1 3): δ 0.875 (3Η, t, J = 6.8 Hz), 1.20-1.52 (8H, m), 2.15 (2H, t, J-8.2 Hz), 2.45 (6H, s), 7.36 (2H, d, J-8.4 Hz), 7.80 (2H, d, J = 8.4 Hz), 9.38 (1H, bs).
元素分析値: C20H25N3O3S3として Elemental analysis: as C 20 H 25 N 3 O 3 S 3
計算値 ) : C, 53.19; H, 5.58; N, 9.30; S, 21.30. Calculated values): C, 53.19; H, 5.58; N, 9.30; S, 21.30.
実測値 ) : C, 52.99; H, 5.67; N, 9.22; S, 21.22. Obtained): C, 52.99; H, 5.67; N, 9.22; S, 21.22.
実施例 3-18 Example 3-18
Ή-NMR (CDC13 - DMS0 - d6= 19 : 1, 300 MHz): δ 2.36 (3Η, s), 2.46 (3H, s), 2.74 (3H, s), 7.37 (2H, d, J= 8.3 Hz), 7.90 (2H, d, J= 8.3 Hz). 元素分析値: C16H15N304S3として計算値: C, 46.93; H, 3.69; N, 10.26; S, 23.49 (%) 実測値: 46.75; H, 3.83; N, 10.00; S, 23.50 {%) . 実施例 3 - 19 Ή-NMR (CDC1 3 -DMS0-d 6 = 19: 1, 300 MHz): δ 2.36 (3Η, s), 2.46 (3H, s), 2.74 (3H, s), 7.37 (2H, d, J = . 8.3 Hz), 7.90 (2H , d, J = 8.3 Hz) elemental analysis: C 16 H 15 N 3 0 4 S 3 calculated: C, 46.93; H, 3.69 ; N, 10.26; S, 23.49 ( %) Found: 46.75; H, 3.83; N, 10.00; S, 23.50 (%). Examples 3-19
ェタノ一ル-ジェチルエーテルから再結晶して融点 216- 218 の橙色結 晶を得た。 ' 赤外吸収スペクトル(IR) (KBr 中) リ: 3400-2500, 1715, 1634, 1580, 1518, 1495 cm— The crystal was recrystallized from ethanol-ethyl ether to give an orange crystal having a melting point of 216-218. ' Infrared absorption spectrum (IR) (in KBr): 3400-2500, 1715, 1634, 1580, 1518, 1495 cm—
Ή-NMR (CDC13) : δ 2.45 (3Η, s), 2.54 (3H, s), 3.52 (2H, s) , 7.14-7.32 (5Η, m), 7.36 (2Η, d, J = 7.6 Hz), 7.78 (2H, d, J=8. Hz), 9.34 (1H, bs). Ή-NMR (CDC1 3): δ 2.45 (3Η, s), 2.54 (3H, s), 3.52 (2H, s), 7.14-7.32 (5Η, m), 7.36 (2Η, d, J = 7.6 Hz) , 7.78 (2H, d, J = 8.Hz), 9.34 (1H, bs).
元素分析値: C21H19N303S3'0.2H20として Elemental analysis: C 21 H 19 N 3 0 3 S 3 '0.2H 2 0
計算値 ) : C, 54.69; H, 4.24; N, 9.11; S, 20.86. Calculated values): C, 54.69; H, 4.24; N, 9.11; S, 20.86.
実測値 ) : C, 54.66; H, 4.05; N, 8.99; S, 21.09. Obtained): C, 54.66; H, 4.05; N, 8.99; S, 21.09.
実施例 3 - 20 Example 3-20
1-{[ (4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -8-スルファニル -2, 3-ジヒドロ 1H-ィミダゾ [5, 1- c] [1,4]チアジン- 5-カルボン酸 ェチ ル 1-{[(4-Methylphenyl) sulfonyl] imino} -3-oxo-8-sulfanyl-2,3-dihydro 1H-imidazo [5, 1-c] [1,4] thiazine-5-carboxylic acid Le
実施例 2-22 で得た化合物 (998 mg, 1.90 mmol)の THF -エタノール (1:1, 0 ml)の懸濁液に室温で攪拌しながらナトリウムエトキシド(271 mg, 3.99 mmol)を加え、 反応液を室温で 7時間攪拌した。 この間反応液 にナトリゥムェトキシド(65 mg, 0.95 mmol)を 1.5時間毎に 3回加えた。 反応液に 1N 塩酸 (6.8 ml)、 水(10 ml)を加えて 30分間攪拌混和した。 析出物を濾取し、メタノール一水(1:1)で洗浄、乾燥して表題化合物 (700 mg, 1.65 mmol)を橙色粉末として得た。 To a suspension of the compound (998 mg, 1.90 mmol) obtained in Example 2-22 in THF-ethanol (1: 1, 0 ml) was added sodium ethoxide (271 mg, 3.99 mmol) with stirring at room temperature. The reaction solution was stirred at room temperature for 7 hours. During this time, sodium methoxide (65 mg, 0.95 mmol) was added to the reaction solution three times every 1.5 hours. 1N hydrochloric acid (6.8 ml) and water (10 ml) were added to the reaction solution, and the mixture was stirred and mixed for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1), and dried to give the title compound (700 mg, 1.65 mmol) as an orange powder.
Ή-N R (200MHz, CDC13) : δ 1.33 (3H, t, J= 7.2 Hz), 2.47 (3H, s), 4.35 (1H, q, J= 7.2 Hz), 6.14 (1H, s), 7.39 (2H, d, J= 8.2 Hz), 7.82 (2H, d, J= 8.4 Hz), 9.71 (1H, brs). Ή-NR (200MHz, CDC1 3 ): δ 1.33 (3H, t, J = 7.2 Hz), 2.47 (3H, s), 4.35 (1H, q, J = 7.2 Hz), 6.14 (1H, s), 7.39 (2H, d, J = 8.2 Hz), 7.82 (2H, d, J = 8.4 Hz), 9.71 (1H, brs).
元素分析値: C16H15N305S3として計算値: C, 45.16; H, 3.55; N, 9.88; S, 22.61 (%) 、 実測^: C, 45.40; H, 3.56; N, 9.87; S, 22.49 (%) . 融点. 209.5-210.5 Elemental analysis: C 16 H 15 N 3 0 5 S 3 Calculated: C, 45.16; H, 3.55 ; N, 9.88; S, 22.61 (%), Found ^: C, 45.40; H, 3.56; N, 9.87; S, 22.49 (%). Mp. 209.5-210.5
実施例 3-21 Example 3-21
6 -メチル [(4-メチルフエニル)スルホニル]ィミノ}- 3_ォキソ -8 -ス ルファニル -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 5-力ルポ ン酸 ェチル 実施例 2- 45で得た化合物(0.29 g, 0.52 mmol)の THF-エタノール (1:1, 6 ml)の懸濁液に室温で攪拌しながら 20%ナトリウムエトキシド(ェタノ —ル溶液、 0.45 ml, 1.14 mmol)を加え、 反応液を室温で 30分間攪拌し た。 反応液に 1N 塩酸 (1.2 ml)、 水(2 ml)を加えて 60分間攪拌混和し た。 析出物を濾取し、 メタノール一水(1:1)で洗浄、 乾燥して表題化合物 (101 mg, 0.230 匪 ol)を橙色粉末として得た。 6-Methyl [(4-methylphenyl) sulfonyl] imino} -3_oxo-8-sulfanyl-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-5-force Lutyl acid ester A suspension of the compound obtained in Example 2-45 (0.29 g, 0.52 mmol) in THF-ethanol (1: 1, 6 ml) was stirred at room temperature with stirring at room temperature for 20% sodium ethoxide (ethanol solution, 0.45 g). ml, 1.14 mmol), and the reaction solution was stirred at room temperature for 30 minutes. 1N Hydrochloric acid (1.2 ml) and water (2 ml) were added to the reaction solution, and the mixture was stirred and mixed for 60 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1), and dried to give the title compound (101 mg, 0.230 ol) as an orange powder.
Ή-NMR (200MHz, CDCi3) : <5 1.35 (3H, t, J= 7.2 Hz), 1.92 (3H, s), 2.46 (3H, s), 4.39 (2H, q, J= 7.2 Hz), 7.37 (2H, d, J= 8.0 Hz), 7.79 (2H, d, J= 8.4 Hz), 12.17 (1H, brs) . Ή-NMR (200MHz, CDCi 3 ): <5 1.35 (3H, t, J = 7.2 Hz), 1.92 (3H, s), 2.46 (3H, s), 4.39 (2H, q, J = 7.2 Hz), 7.37 (2H, d, J = 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz), 12.17 (1H, brs).
元素分析値: C17H17N305S3として計算値: C, 46.45; H, 3.90; N, 9.56; S, 21.89 (%) 、 実測値: C, 46.42; H, 3.99; N, 9.39; S, 21.83 (%) . 融点. 224.5-226. O : Elemental analysis: C 17 H 17 N 3 0 5 S 3 Calculated: C, 46.45; H, 3.90 ; N, 9.56; S, 21.89 (%), Found: C, 46.42; H, 3.99 ; N, 9.39; S, 21.83 (%). Melting point. 224.5-226. O:
実施例 3-22 Example 3-22
4-メチル -N- (6-メチル -3-ォキソ -5-プロピオニル -8-スルファニル -2, 3- ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン-卜ィリデン)ベンゼンスル ホンアミド 4-methyl-N- (6-methyl-3-oxo-5-propionyl-8-sulfanyl-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-trilidene) benzenesul Honamide
実施例 2-48 で得た化合物 (3.03 g, 5.79 腿 ol)の THF-メタノール (1:1, 60 ml)の懸濁液に室温で攪拌しながら水酸化カリウム(1.15 g, 17.4inmol)を加え、 反応液を室温で 30分間攪拌した。 反応液に 1N 塩酸 (17.4ml)および水(13ml)を加え、 室温で 30分間攪拌した。 析出物を濾 取し、 メタノール一水 (1:1)で洗浄、乾燥して標題化合物(2.24 g, 5.28 mmol)を赤色粉末として得た。 To a suspension of the compound obtained in Example 2-48 (3.03 g, 5.79 tmol) in THF-methanol (1: 1, 60 ml) was added potassium hydroxide (1.15 g, 17.4 inmol) while stirring at room temperature. The reaction was stirred at room temperature for 30 minutes. 1N Hydrochloric acid (17.4 ml) and water (13 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1), and dried to give the title compound (2.24 g, 5.28 mmol) as a red powder.
'Η -匪 R (200MHz, CDC13) : δ 1.15 (3Η, t, J= 7.2 Hz), 1.78 (3H, s), 2.46 (3H, s) , 2.64 (2H, q, J= 7.6 Hz), 7.37 (2H, d, J= 8.4 Hz), 7.89 (2H, d, J= 8.4 Hz). 'Eta - negation R (200MHz, CDC1 3): δ 1.15 (3Η, t, J = 7.2 Hz), 1.78 (3H, s), 2.46 (3H, s), 2.64 (2H, q, J = 7.6 Hz) , 7.37 (2H, d, J = 8.4 Hz), 7.89 (2H, d, J = 8.4 Hz).
元素分析値: C17H17N304S3として計算値: C, 48.21; H, 4.05; N, 9.92(%)、 実測値: C, 48.51; H, 4.09; N, 9.87 (%) . Elemental analysis: Calculated as C 17 H 17 N 3 0 4 S 3: C, 48.21; H, 4.05; N, 9.92 (%), Found: C, 48.51; H, 4.09 ; N, 9.87 (%) .
実施例 3 - 23 Example 3-23
4 -メチル - N- [6-メチル - 5-(4-モルホリ二ルカルポ二ル)- 3-ォキソ -8 -ス ルファニル- 2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデ ン]ベンゼンスルホンアミ ド 4-Methyl-N- [6-methyl-5- (4-morpholinylcarbonyl) -3-oxo-8-s Ruphanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] benzenesulfonamide
実施例 2-47で得た化合物(1.00 g, 1.72 mmol)の THF-メタノール (1:1, 20 ml)の懸濁液に室温で攪拌しながら水酸化力リウム(0.34 g, 5.17 mmol)を加え、 反応液を室温で 30分間攪拌した。 反応液に IN 塩酸 (5.2 ml) を加え、 室温で 60分間攪拌した。 析出物を濾取し、 メタノール一水 (1:1)およびジェチルエーテルで洗浄、'乾燥して標題化合物 (637 mg, 1.33 mmol)を赤色粉末として得た。 To a suspension of the compound obtained in Example 2-47 (1.00 g, 1.72 mmol) in THF-methanol (1: 1, 20 ml) was added lithium hydroxide (0.34 g, 5.17 mmol) with stirring at room temperature. The reaction was stirred at room temperature for 30 minutes. IN hydrochloric acid (5.2 ml) was added to the reaction solution, and the mixture was stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and getyl ether, and dried to give the title compound (637 mg, 1.33 mmol) as a red powder.
顏 R (200MHz, CDC13) : δ 1.87 (3H, s), 2.47 (3H, s), 3.46 (2H, brt, J= 4.6 Hz), 3.60-3.90 (6H, m), 7.38 (2H, d, J= 8.0 Hz), 7.79 (2H, d, J= 8.4 Hz), 12.26 (1H, brs). 顏R (200MHz, CDC1 3): δ 1.87 (3H, s), 2.47 (3H, s), 3.46 (2H, brt, J = 4.6 Hz), 3.60-3.90 (6H, m), 7.38 (2H, d , J = 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz), 12.26 (1H, brs).
元素分析値: C19H2。N405S3として計算値: C, 47.48; H, 4.19; N, 11.66; S, 20.02 (%), 実測値: C, 47.23; H, 4.21; N, 11.58; S, 19.62 (%) . 実施例 3-24 Elemental analysis: C 19 H 2. N 4 0 5 S 3 Calculated: C, 47.48; H, 4.19 ; N, 11.66; S, 20.02 (%), Found: C, 47.23; H, 4.21 ; N, 11.58; S, 19.62 (%) Example 3-24
Ή-NMR (300MHz, CDC13) : δ 2. 6 (3Η, s), 5.31 (1Η, s) , 6.95 (2H, d, J= 8.7 Hz), 7.06 (2H, brd, J= 7.5 Hz), 7.14 (1H, brt, J= 7.3 Hz), 7.20 (2H, d, J= 8.7 Hz), 7.36 (2H, brt, J= 8.3 Hz), 7.38 (2H, d, J= 8.3 Hz), 7.79 (2H, d, J= 8.3 Hz), 9.34 (1H, br), 12.62 (1H, s) . Ή-NMR (300MHz, CDC1 3 ): δ 2. 6 (3Η, s), 5.31 (1Η, s), 6.95 (2H, d, J = 8.7 Hz), 7.06 (2H, brd, J = 7.5 Hz) , 7.14 (1H, brt, J = 7.3 Hz), 7.20 (2H, d, J = 8.7 Hz), 7.36 (2H, brt, J = 8.3 Hz), 7.38 (2H, d, J = 8.3 Hz), 7.79 (2H, d, J = 8.3 Hz), 9.34 (1H, br), 12.62 (1H, s).
元素分析値: C25Hl9N304S3として計算値: C, 57.56; H, 3.67; N, 8.06; S, 18.44 (%)、 実測値: C, 57.62; H, 3.77; N, 7.82; S, 18.43 ( ) . 実施例 3-25 Elemental analysis: C 25 H l9 N 3 0 4 S 3 Calculated: C, 57.56; H, 3.67 ; N, 8.06; S, 18.44 (%), Found: C, 57.62; H, 3.77 ; N, 7.82; S, 18.43 (). Examples 3-25
Ή-NMR (CDCl3-DMS0-d6= 9:1, 00 MHz): δ 1.81 (3Η, s), 2.44 (3H, s) , 7.46 (2H, brs), 7.80 (2H, brs). Ή-NMR (CDCl 3 -DMS0-d 6 = 9: 1, 00 MHz): δ 1.81 (3Η, s), 2.44 (3H, s), 7.46 (2H, brs), 7.80 (2H, brs).
元素分析値.: C15Hl5N303S3として計算値: C, 47.22; H, 3.96; , 11.01; S, 25.22 (%)、 実測値: C, 47.02; H, 4.18; N, 10.92; S, 25.22 (%) . 実施例 3-26 Elemental analysis: Calculated for C 15 H 15 N 3 0 3 S 3 : C, 47.22; H, 3.96;, 11.01; S, 25.22 (%), found: C, 47.02; H, 4.18; N, 10.92; S, 25.22 (%). Example 3-26
'Η -蘭 R (DMS0-d6, 200 MHz): δ 1.21 (3H, ί, J= 6.9 Hz), 2. 0 (3H, s), 2.73 (3H, s), 4.13 (2H, q, J= 7.0 Hz), 7.50 (2H, brd, J= 4.4 Hz), 7.83 (2H, brs). 元素分析値: C17H17N305S3として計算値: C, 46.45; H, 3.90; N, 9.56; S, 21.89 (%), 実測値: C, 46. 7; H, 4.12; N, 9.47; S, 21.76 (%) . 実施例 3-27 'Η-Ran R (DMS0-d 6 , 200 MHz): δ 1.21 (3H, ί, J = 6.9 Hz), 2.0 (3H, s), 2.73 (3H, s), 4.13 (2H, q, J = 7.0 Hz), 7.50 (2H, brd, J = 4.4 Hz), 7.83 (2H, brs). Elemental analysis: Calculated as C 17 H 17 N 3 0 5 S 3: C, 46.45; H, 3.90; N, 9.56; S, 21.89 (%), Found: C, 46. 7; H, 4.12; N, 9.47; S, 21.76 (%). Example 3-27
Ή-NMR (CDC13- MS0- d6 = 9:1, 200 MHz): δ 1.81 (3H, s), 2.41 (3H, brq, J= 0.8 Hz), 2.71 (3H, s), 7.37 (2H, πι) , 7.55 (1H, tm, J= 8.1 Hz), 8.16 (1H, dm, J= 8.6 Hz). NMR-NMR (CDC1 3 -MS0- d 6 = 9: 1, 200 MHz): δ 1.81 (3H, s), 2.41 (3H, brq, J = 0.8 Hz), 2.71 (3H, s), 7.37 (2H , πι), 7.55 (1H, tm, J = 8.1 Hz), 8.16 (1H, dm, J = 8.6 Hz).
元素分析値: C15H15N303S3として計算値: C, 47.22; H, 3.96; N, 11.01; S, 25.22 (%), 実測値: C, 47.41; H, 3.80; N, 11.01; S, 25.07 (%) . 実施例 3-28 Elemental analysis: C 15 H 15 N 3 0 3 S 3 Calculated: C, 47.22; H, 3.96 ; N, 11.01; S, 25.22 (%), Found: C, 47.41; H, 3.80 ; N, 11.01; S, 25.07 (%). Example 3-28
Ή-NMR (顯 0- d6, 200 MHz): δ 1.20 (3Η, i, J= 7.0 Hz), 2.64 (3H, s), 2.68 (3H, s), 4.12 (2H, q, 7.2 Hz), 7.39 (2H, t, J= 8 Hz), 7.52 (1H, brd, J= 8 Hz), 8.06 (1H, brd, J= 8 Hz), 9.42 (1H, brs). 元素分析値: CnH17N305S3 として計算値: C, 46.45; H, 3.90; N, 9.56Ή-NMR (Akira 0- d 6, 200 MHz): δ 1.20 (3Η, i, J = 7.0 Hz), 2.64 (3H, s), 2.68 (3H, s), 4.12 (2H, q, 7.2 Hz) , 7.39 (2H, t, J = 8 Hz), 7.52 (1H, brd, J = 8 Hz), 8.06 (1H, brd, J = 8 Hz), 9.42 (1H, brs). Elemental analysis: C n Calculated for H 17 N 3 0 5 S 3 : C, 46.45; H, 3.90; N, 9.56
( ), 実測値: C, 46.74; H, 3.73; N, 9.59 (%) . (), Found: C, 46.74; H, 3.73; N, 9.59 (%).
実施例 3-29 Example 3-29
Ή-NMR (200 MHz, 重クロ口ホルム中): δ 0.89 (3Η, t, J= 7.2 Hz), 1.26 (3H, t, J= 7.2 Hz), 1.30 (4H, m), 1.33 (2H, m), 1.47 (2H, brquint, J= 7.0 Hz), 2.07 (2H, t, J= 7.5 Hz), 2.71 (2H, t, J= 7.4 Hz), 3.24 (2H, t, J= 7.4 Hz), 4.16 (2H, q, J= 7.2 Hz), 6.50 (1H, s), 7.02 (2H, dt, J= 8,7, 2.3 Hz), 7.07 (2H, brd, J= 8.0 Hz), 7.21 (1H, brt, J = 7.5 Hz), 7.40 (2H, tt, J= 7.9, 2.3 Hz), 7.89 (2H, dt, J= 9.0, 2.3 Hz), 9.70 (1H, brs). Ή-NMR (200 MHz, in double-mouthed form): δ 0.89 (3Η, t, J = 7.2 Hz), 1.26 (3H, t, J = 7.2 Hz), 1.30 (4H, m), 1.33 (2H, m), 1.47 (2H, brquint, J = 7.0 Hz), 2.07 (2H, t, J = 7.5 Hz), 2.71 (2H, t, J = 7.4 Hz), 3.24 (2H, t, J = 7.4 Hz) , 4.16 (2H, q, J = 7.2 Hz), 6.50 (1H, s), 7.02 (2H, dt, J = 8, 7, 2.3 Hz), 7.07 (2H, brd, J = 8.0 Hz), 7.21 ( 1H, brt, J = 7.5 Hz), 7.40 (2H, tt, J = 7.9, 2.3 Hz), 7.89 (2H, dt, J = 9.0, 2.3 Hz), 9.70 (1H, brs).
元素分析値: C24H25N304.S3 として計算値: 55.90; H, 4.89; N, 8.15; S, 18.65 (%) 実測値; C, 55.67; H, 4.64; N, 8.03; S, 18.52 (%) . 実施例 3-30 Elemental analysis: Calculated as C 24 H 25 N 3 0 4 .S 3: 55.90; H, 4.89; N, 8.15; S, 18.65 (%) Found; C, 55.67; H, 4.64 ; N, 8.03; S, 18.52 (%). Example 3-30
-麵 R (CDC13, 200 MHz): δ 1.84 (3Η, s), 2.46 (3H, brs), 7.06 (2H, d, J= 9.2 Hz), 7.08 (2H, d, J= 7 Hz), 7.27 (1H, t, J= 8 Hz), 7. 3 (2H, t, J= 7.9 Hz), 7.83 (2H, d, J= 8.8 Hz). - noodles R (CDC1 3, 200 MHz) : δ 1.84 (3Η, s), 2.46 (3H, brs), 7.06 (2H, d, J = 9.2 Hz), 7.08 (2H, d, J = 7 Hz), 7.27 (1H, t, J = 8 Hz), 7.3 (2H, t, J = 7.9 Hz), 7.83 (2H, d, J = 8.8 Hz).
元素分析値: C2。H17N304S3として計算値: C, 52.27; H, 3.73; N, 9.14; S, 20.93 (%), 実測値: C, 52.33; H, 3.96; , 9.29; S, 20.73 (%) . 実施例 3 - 31 Elemental analysis: C 2. H 17 N 3 0 4 S 3 Calculated: C, 52.27; H, 3.73 ; N, 9.14; S, 20.93 (%), Found: C, 52.33; H, 3.96;, 9.29; S, 20.73 (%). Examples 3-31
Ή-NMR (CDC13, 200 MHz): δ 1.31 (3H, t, J= 7.0 Hz), 2.93 (3H, s), Ή-NMR (CDC1 3, 200 MHz): δ 1.31 (3H, t, J = 7.0 Hz), 2.93 (3H, s),
4.25 (2H, q, J= 7.2 Hz), 7.06 (2H, d, J= 9.2 Hz), 7.08 (2H, d, J = 8 Hz), 7.27 (1H, t, J= 8 Hz), 7.43 (2H, d, J= 8.0 Hz), 7.84 (2H, d, J= 8.8 Hz). 4.25 (2H, q, J = 7.2 Hz), 7.06 (2H, d, J = 9.2 Hz), 7.08 (2H, d, J = 8 Hz), 7.27 (1H, t, J = 8 Hz), 7.43 ( 2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.8 Hz).
元素分析値: C22H19N306S3として計算値: C, 51.05; H, 3.70; N, 8.12; S, 18.59 (%)、 実測値: 51.12; H, 3.64; N, 8.13; S, 18.51 ( ) . 実施例 3-32 Elemental analysis: C 22 H 19 N 3 0 Calculated 6 S 3: C, 51.05; H, 3.70; N, 8.12; S, 18.59 (%), Found: 51.12; H, 3.64; N , 8.13; S, 18.51 (). Example 3-32
Ή-NMR (DMSO- d6, 200 MHz): δ 1.74 (3H, s), 2.29 (3H, s), 7.60 (1H, m), 7.68 (2H, m) , 1H, d, J= 7.0 Hz). Ή-NMR (DMSO- d 6, 200 MHz): δ 1.74 (3H, s), 2.29 (3H, s), 7.60 (1H, m), 7.68 (2H, m), 1H, d, J = 7.0 Hz ).
実施例 3-33Example 3-33
-匪 R (CDCl3-DMS0-d63:1, 200 MHz): d 1.30 (3H, t, J= 7.0 Hz), 2.88 (3H, s), 4.22 (2H, q, J= 7.0 Hz), 7.60 (2H, d, J= 8.8 Hz), 8.03 (2H, d, J= 8.8 Hz). -Banded R (CDCl 3 -DMS0-d 6 3: 1, 200 MHz): d 1.30 (3H, t, J = 7.0 Hz), 2.88 (3H, s), 4.22 (2H, q, J = 7.0 Hz) , 7.60 (2H, d, J = 8.8 Hz), 8.03 (2H, d, J = 8.8 Hz).
元素分析値: C16H14N305S3C1 として計算値: C, 41.78; H, 3.07; N, 9.14;Elemental analysis: C 16 H 14 N 3 0 5 S 3 C1 Calculated: C, 41.78; H, 3.07 ; N, 9.14;
5, 20.91; C1, 7.71 (%)、 実測値: C, 41.68; H, 2.96; N, 9.13; S, 20.90; C1, 7.54 ( ) . 5, 20.91; C1, 7.71 (%), found: C, 41.68; H, 2.96; N, 9.13; S, 20.90; C1, 7.54 ().
実施例 3-34Example 3-34
- NMR (DMS0-d6, 200 MHz): δ 1.21 (3Η, t, J= 7.2 Hz), 2.73 (3H, s), 4.14 (2H, q, J= 7.2 Hz), 7.45 (2H, t, J= 8.8 Hz), 8.08 (2H, dd, J= 8.8, 5.4 Hz). -NMR (DMS0-d 6 , 200 MHz): δ 1.21 (3Η, t, J = 7.2 Hz), 2.73 (3H, s), 4.14 (2H, q, J = 7.2 Hz), 7.45 (2H, t, J = 8.8 Hz), 8.08 (2H, dd, J = 8.8, 5.4 Hz).
元素分析値: C16HuN305S3Fとして計算値: C, 43.33; H, 3.18; , 9.47; S, 21.69; F, 4.28 (%)、実測値: C, 43.20; H, 3.44; N, 9.32; S, 21.33; F, 4.29 (%) . Elemental analysis: C 16 H u N 3 0 5 S 3 F Calculated: C, 43.33; H, 3.18 ;, 9.47; S, 21.69; F, 4.28 (%), Found: C, 43.20; H, 3.44; N, 9.32; S, 21.33; F, 4.29 (%).
実施例 3 - 35 Example 3-35
Ή-NMR (DMS0-d6, 200 MHz): δ 1.74 (3H, brs), 2.31 (3H, brs), 7.46Ή-NMR (DMS0-d 6 , 200 MHz): δ 1.74 (3H, brs), 2.31 (3H, brs), 7.46
(2H, t, J= 8.7 Hz), 8.06 (2H, dd, J= 9.2, 5.2 Hz) . (2H, t, J = 8.7 Hz), 8.06 (2H, dd, J = 9.2, 5.2 Hz).
元素分析値: C14H12N303S3Fとして計算値: C, 43.62; H, 3.14; N, 10.90; F, 4.93 {%), 実測値: C, 43.38; H, 3.34; N, 10.88; F, 4.93 (%) . 実施例 3 - 36 Elemental analysis: C 14 H 12 N 3 0 3 S 3 F Calculated: C, 43.62; H, 3.14 ; N, 10.90; F, 4.93 (%), Found: C, 43.38; H, 3.34; N, 10.88; F, 4.93 (%).
Ή-NMR (DMS0-d6, 300 MHz): δ 1.23 (3H, t, J= 7.2 Hz), 2.80 (3H, s), 3.45 (3H, s), 4.17 (2H, q, J= 7.2 Hz). Ή-NMR (DMS0-d 6 , 300 MHz): δ 1.23 (3H, t, J = 7.2 Hz), 2.80 (3H, s), 3.45 (3H, s), 4.17 (2H, q, J = 7.2 Hz) ).
元素分析値: CuHi3N305S3として計算値: C, 36.35; H, 3.61; N, 11.56; S, 26.47.(%)、 実測値: 36.45; H, 3.75; , 11.33; S, 26.26 (%) . 実施例 3-37 Elemental analysis: Calculated as C u H i3 N 3 0 5 S 3:. C, 36.35; H, 3.61; N, 11.56; S, 26.47 (%), Found: 36.45; H, 3.75;, 11.33; S, 26.26 (%). Example 3-37
N -(4-スルファニル- 1-ォキソ _1, 2-ジヒドロ- 3H-ィミダゾ [5, 1-c] [1,4] ベンゾチアジン- 3 -ィリデン) -4-メチルベンゼンスルホンアミド N- (4-sulfanyl-1-oxo_1,2-dihydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene) -4-methylbenzenesulfonamide
実施例 2- 67で合成した化合物 (252 mg, 0.501 mmol) の THF (2.50 ml) およびエタノール (2.50 ml) 懸濁液に、 水酸化カリウム (99.1 mg, 1.50 mmol) を加えて、 室温で 30分間撹拌した。 反応液に 1N塩酸 (2.00 ml) を加えて混和し、室温で 1時間撹拌した。生成した沈澱物を濾取し、水、 エタノールで洗浄後、 乾燥して表題化合物の黄色結晶 (183 mg, 9U) を 得た。 本品はこれ以上精製することなく次の反応に使用した。 To a suspension of the compound synthesized in Example 2-67 (252 mg, 0.501 mmol) in THF (2.50 ml) and ethanol (2.50 ml) was added potassium hydroxide (99.1 mg, 1.50 mmol), and the mixture was stirred at room temperature for 30 minutes. Stirred for minutes. 1N Hydrochloric acid (2.00 ml) was added to the reaction solution, mixed, and stirred at room temperature for 1 hour. The resulting precipitate was collected by filtration, washed with water and ethanol, and dried to give the title compound as yellow crystals (183 mg, 9U). This product was used for the next reaction without further purification.
IR (KBr) V: 3300-2400, 2996, 1715, 1588, 1574, 1524 cm"1. IR (KBr) V: 3300-2400, 2996, 1715, 1588, 1574, 1524 cm " 1 .
Ή-N R (DMS0-d6) : δ 2.51 (3Η, s) , 7.04-7.14 (2Η, m) , 7.16-7.28 (1Η, m), 7.44 (2H, d, J=8.4 Hz), 7.94 (2H, d, J = 8.4 Hz), 8.29 (1H, bs), 8.69 (1H, d, J = 8.4 Hz). Ή-NR (DMS0-d 6 ): δ 2.51 (3Η, s), 7.04-7.14 (2Η, m), 7.16-7.28 (1Η, m), 7.44 (2H, d, J = 8.4 Hz), 7.94 ( 2H, d, J = 8.4 Hz), 8.29 (1H, bs), 8.69 (1H, d, J = 8.4 Hz).
実施例 3-38 Example 3-38
N -(1, 6-ジォキソ- 4-スルファニル -1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミ ダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン) -4-メチルベンゼンスル ホンアミ ド N- (1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene ) -4-Methylbenzenesulfonamide
実施例 2-68で合成した化合物 (2.43 g, 4.66 mmol) の THF (24.0 ml) およびエタノール (24.0 ml) 懸濁液に、 水酸化カリウム (980 mg, 14.0 mmol) を加えて、 室温で 1時間撹拌した。 反応液に 1N塩酸 (16.8 ml) を加えて混和し、室温で 1時間撹拌した。生成した沈澱物を濾取し、水、 エタノールで洗浄後、 乾燥して表題化合物の橙色結晶 (1.57 g, 80¾) を 得た。 酢酸ェチルから再結晶して融点 246- 248°Cの橙色結晶を得た。 赤外吸収スペクトル(IR) (KBr 中) リ: 3700-2300, 1721, 1661, 1584, 1501 cm— To a suspension of the compound synthesized in Example 2-68 (2.43 g, 4.66 mmol) in THF (24.0 ml) and ethanol (24.0 ml) was added potassium hydroxide (980 mg, 14.0 mmol), and the mixture was stirred at room temperature for 1 hour. Stirred for hours. The reaction mixture was mixed with 1N hydrochloric acid (16.8 ml) and stirred at room temperature for 1 hour. The resulting precipitate was collected by filtration, washed with water and ethanol, and dried to give the title compound as orange crystals (1.57 g, 80 °). Recrystallization from ethyl acetate gave orange crystals with a melting point of 246-248 ° C. Infrared absorption spectrum (IR) (in KBr): 3700-2300, 1721, 1661, 1584, 1501 cm—
Ή-NMR (CDC13側 SO - d6) : δ 1.98-2.16 (2Η, m) , 2.34-2.62 (2Η, m) , 2.45 (3Η, s), 3.37 (2Η, t, J = 5.8 Hz), 7.36 (2H, d, J = 8.0 Hz), 7.89 (2H, d, J = 8.0 Hz). Ή-NMR (CDC1 3 side SO-d 6 ): δ 1.98-2.16 (2Η, m), 2.34-2.62 (2Η, m), 2.45 (3Η, s), 3.37 (2Η, t, J = 5.8 Hz) , 7.36 (2H, d, J = 8.0 Hz), 7.89 (2H, d, J = 8.0 Hz).
元素分析値: C17H15N304S3'0.4H20として Elementary analysis: as C 17 H 15 N 3 0 4 S 3 '0.4H 2 0
計算値 (¾) : C, 47.63; H, 3.71; N, 9.80. Calculated value (¾): C, 47.63; H, 3.71; N, 9.80.
実測値 (¾) : C, 47.95; H, 3.66; N, 9.72. Found (¾): C, 47.95; H, 3.66; N, 9.72.
実施例 3 - 39Example 3-39
-(l, 6-ジォキソ- 4-スルファニル -1, 2, 7, 8-テトラヒドロ- 3H, 6H-シク口 ペン夕 [b]イミダゾ [1, 5-d] [1,4]チアジン- 3 -ィリデン) -4-メチルベンゼ ンスルホンアミ ド -(l, 6-Dioxo-4-sulfanyl-1,2,7,8-tetrahydro-3H, 6H-cyclopent [b] imidazo [1,5-d] [1,4] thiazine-3- (Ylidene) -4-methylbenzenesulfonamide
実施例 2- 69で合成した化合物 (800 mg, 1.63 mmol) の THF (8.00 ml) およびエタノール (8.00 ml) 懸濁液に、 水酸化カリウム (322 mg, 4.88 imol) を加えて、 室温で 20分間撹拌した。 反応液に 1N塩酸 (6.51 ml) を加えて混和し、室温で 1時間撹拌した。生成した沈澱物を濾取し、水、 エタノールで洗浄後、 乾燥して表題化合物の橙色結晶 (540 mg, 85%) を 得た。 本品はこれ以上精製することなく次の反応に使用した。 To a suspension of the compound (800 mg, 1.63 mmol) synthesized in Example 2-69 in THF (8.00 ml) and ethanol (8.00 ml) was added potassium hydroxide (322 mg, 4.88 imol), and the mixture was added at room temperature for 20 minutes. Stirred for minutes. 1N Hydrochloric acid (6.51 ml) was added to the reaction solution, mixed, and stirred at room temperature for 1 hour. The resulting precipitate was collected by filtration, washed with water and ethanol, and dried to give the title compound as orange crystals (540 mg, 85%). This product was used for the next reaction without further purification.
赤外吸収スペクトル(IR) (KBr 中) リ: 3700-2300, 1732, 1696, 1686, 1607, 1582, 1495 cm一1. Infrared absorption spectrum (IR) (in KBr): 3700-2300, 1732, 1696, 1686, 1607, 1582, 1495 cm- 1 .
Ή-NMR (CDCl3+DMSO-d6) : δ 2.41 (3Η, s), 2.48-2.60 (2H, m) , 3.10-3.24 (2Η, m), 7.41 (2H, d, J = 8.2 Hz), 7.91 (2H, d, J = 8.0 Hz). Ή-NMR (CDCl 3 + DMSO-d 6 ): δ 2.41 (3Η, s), 2.48-2.60 (2H, m), 3.10-3.24 (2Η, m), 7.41 (2H, d, J = 8.2 Hz) , 7.91 (2H, d, J = 8.0 Hz).
実施例 3 - 40 Example 3-40
5-C2- トキシ- 2-ォキソェチル) -卜 {[(4-メチルフエニル)スルホニル] ィ ミ ノ } - 3 -ォキソ -8-スルフ ァニル -2, 3 -ジヒ ドロ - 1H-ィ ミ ダゾ [5, 1-c] [1,4]チアジン _6 -力ルボン酸ェチル 5-C2-Toxy-2-oxoethyl) -tri {[(4-methylphenyl) sulfonyl] imino} -3--3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine _6-ethyl rubonate
3-ケトグル夕ル酸ジェチル (11.0 g, 54.3 mmol)のジクロロメタン (50 ml)の溶液に室温で攪拌しながら塩化スルフリル (7.44 g, 54.3 mmol) のジクロロメタン (5 ml)溶液を 60分間かけて滴下し、 反応液を室温で 1.5 時間攪拌した。 反応液を氷(50 g)および飽和炭酸水素ナトリウム水 溶液(50 ml)の混合液に注ぎ、 激しく攪拌し、 水層をジクロロメタン (80 ml)で抽出した。有機層を 15%食塩水で洗浄後、硫酸ナトリゥムで乾燥後、 濃縮乾固して粗製の 2_クロ口- 3-ケトグルタル酸ジェチル(純度 50%, 13.4 g, 28 mmol, 52%)を無色油状物として得た。 A solution of sulfuryl chloride (7.44 g, 54.3 mmol) in dichloromethane (5 ml) was added dropwise over 60 minutes to a solution of getyl 3-ketoglurate (11.0 g, 54.3 mmol) in dichloromethane (50 ml) with stirring at room temperature. At room temperature Stir for 1.5 hours. The reaction solution was poured into a mixture of ice (50 g) and a saturated aqueous solution of sodium hydrogen carbonate (50 ml), stirred vigorously, and the aqueous layer was extracted with dichloromethane (80 ml). The organic layer was washed with a 15% saline solution, dried over sodium sulfate, and concentrated to dryness to give crude 2_ clomouth-3-ethyl ketoglutarate (purity 50%, 13.4 g, 28 mmol, 52%) colorless. Obtained as an oil.
Ή-N R (300MHz, CDC13, 主ピーク) : 53.72 (1H, d, J= 16.5 Hz), 3.82 (1H, d, J= 16.5 Hz), 5.03 (1H, s) . Ή-NR (300MHz, CDC1 3 , main peak): 53.72 (1H, d, J = 16.5 Hz), 3.82 (1H, d, J = 16.5 Hz), 5.03 (1H, s).
参考例 3- 2で合成した化合物(純度 90%, 8.07 g, 16.9 mmol)およびト リェチルアミン(7.03 ml, 50.7 mmol)の THF-DMF (9:1, 120 ml)懸濁液 に室温で攪拌しながら上記 2-クロ口- 3 -ケトグルタル酸ジェチル (純度 50¾, 8.00 g, 16.9 mmol)の THF- DMF (9:1, 20 ml) 溶液を 30分間かけ て滴下し、 反応液を室温で 3時間攪拌した。 反応液を濃縮後、 酢酸ェチ ル(300 ml)で希釈し、 水、 Π炭酸水素ナトリウム水溶液および飽和食塩 水で洗浄した。 水洗液は酢酸ェチル(200 ml)で抽出した。 有機層を硫酸 ナトリウムで乾燥、 濃縮乾固して褐色油状物 (12.6 g)を得た。 A suspension of the compound synthesized in Reference Example 3-2 (purity 90%, 8.07 g, 16.9 mmol) and triethylamine (7.03 ml, 50.7 mmol) in THF-DMF (9: 1, 120 ml) was stirred at room temperature. A solution of 2-ethyl-3-ethyl ketoglutarate (purity 50¾, 8.00 g, 16.9 mmol) in THF-DMF (9: 1, 20 ml) was added dropwise over 30 minutes, and the reaction solution was allowed to stand at room temperature for 3 hours Stirred. The reaction solution was concentrated, diluted with ethyl acetate (300 ml), and washed with water, an aqueous solution of sodium hydrogen carbonate, and saturated saline. The washing solution was extracted with ethyl acetate (200 ml). The organic layer was dried over sodium sulfate and concentrated to dryness to give a brown oil (12.6 g).
得られた褐色油状物をトルエン(200 ml)に溶解し、 無水酢酸(4.88 ml, 50.7 mmol)および p-トルエンスルホン酸水和物(3.21 g, 16.9 mmol) 存 在下 80°C で 2時間攪拌した。 反応液を濃縮後、 酢酸ェチル(300 ml)で 希釈し、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥、 濃縮し て赤色油状物を得た。 これをシリカゲル(100 g)のクロマトグラフィー (酢酸ェチルーへキサン、 3:7-4:6) により精製し、 得られた橙色油状物 (3.85 g)にジェチルエーテルを加えた。 析出物を濾取してジェチルエー テルで洗浄後、 乾燥して下記に示す化合物 (3.35 g, 5.48 mmol, 32.4%) を橙色粉末として得た。 The resulting brown oil was dissolved in toluene (200 ml) and stirred at 80 ° C for 2 hours in the presence of acetic anhydride (4.88 ml, 50.7 mmol) and p-toluenesulfonic acid hydrate (3.21 g, 16.9 mmol). did. The reaction solution was concentrated, diluted with ethyl acetate (300 ml), washed with water and saturated saline, dried over sodium sulfate, and concentrated to obtain a red oil. This was purified by chromatography on silica gel (100 g) (ethyl acetate-hexane, 3: 7-4: 6), and getyl ether was added to the obtained orange oil (3.85 g). The precipitate was collected by filtration, washed with getyl ether, and dried to obtain the following compound (3.35 g, 5.48 mmol, 32.4%) as an orange powder.
Ή-NMR (200MHz, CDC13) : δ 1.21 (3H, t, J= 7.0 Hz), 1.26 (3H, t, J = 7.2 Hz), 1.31 (3H, t, J= 7.2 Hz), 2.43 (3H, s), 2.69 (2H, dt, J= 2.6, 7.2 Hz), 3.24 (2H, m) , 4.15 (2H, q, J= 7.2 Hz), 4.21 (2H, q, J= 7.2 Hz), 4.15 (2H, q, J- 7.0 Hz), 6.62 (1H, s), 6.64 (1H, s), 7.31 (2H, d, J= 8.0 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.81 (1H, br). Ή-NMR (200MHz, CDC1 3 ): δ 1.21 (3H, t, J = 7.0 Hz), 1.26 (3H, t, J = 7.2 Hz), 1.31 (3H, t, J = 7.2 Hz), 2.43 (3H , s), 2.69 (2H, dt, J = 2.6, 7.2 Hz), 3.24 (2H, m), 4.15 (2H, q, J = 7.2 Hz), 4.21 (2H, q, J = 7.2 Hz), 4.15 (2H, q, J- 7.0 Hz), 6.62 (1H, s), 6.64 (1H, s), 7.31 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.81 (1H, br).
元素分析値: C25H29N309S3として計算値: 9.09; H, 4.78; N, 6.87; S, 15.73. Elemental analysis: Calculated as C 25 H 29 N 3 0 9 S 3: 9.09; H, 4.78; N, 6.87; S, 15.73.
実測値: C, 49.08; H, 4.51; N, 6.99; S, 15.71. Found: C, 49.08; H, 4.51; N, 6.99; S, 15.71.
上記で得られた化合物 (3.39 g, 5.54 mol)の THF -エタノール (1:1, 70 ml)溶液に室温で攪拌しながら 20%ナトリウムエトキシド (ェタノ一 ル溶液、 5.00 ml, 12.7 mmol) を加え、 反応液を 1時間攪拌した。 反応 液に 1N 塩酸 (13.8 ml)および水(75 ml)を加えた。 析出物を濾取し、 メ タノ一ル-水(1:1)で洗浄後、 乾燥して表題化合物(2.18 g, 4.26 mmol, 76.8 )を橙色粉末として得た。 To a solution of the compound obtained above (3.39 g, 5.54 mol) in THF-ethanol (1: 1, 70 ml) was added 20% sodium ethoxide (ethanol solution, 5.00 ml, 12.7 mmol) with stirring at room temperature. The reaction was stirred for 1 hour. 1N Hydrochloric acid (13.8 ml) and water (75 ml) were added to the reaction solution. The precipitate was collected by filtration, washed with methanol-water (1: 1), and dried to give the title compound (2.18 g, 4.26 mmol, 76.8) as an orange powder.
lH-NMR (200MHz, CDC13): δ 1.28 (3H, t, J= 7.1 Hz), 1.30 (3H, t, J = 7.2 Hz), 2.47 (3H, s), 4.20 (2H, q, J= 7.0 Hz), 4.24 (2H, q, J= 7.0 Hz), 4.65 (2H; s), 6.64 (1H, s), 7.39 (2H, d, J= 8.2 Hz), 7.78 (2H, d, J= 8.4 Hz) . lH-NMR (200MHz, CDC1 3 ): δ 1.28 (3H, t, J = 7.1 Hz), 1.30 (3H, t, J = 7.2 Hz), 2.47 (3H, s), 4.20 (2H, q, J = 7.0 Hz), 4.24 (2H, q, J = 7.0 Hz), 4.65 (2H ; s), 6.64 (1H, s), 7.39 (2H, d, J = 8.2 Hz), 7.78 (2H, d, J = 8.4 Hz).
元素分析値: C2。H21N307S3として計算値: 46.95; H, 4.14; N, 8.21; S, 18.80 ( ) 、 実測値: C, 47.08; H, 4.04; N, 8.07; S, 18.74 (%) . mp. 199.0-200.5°C Elemental analysis: C 2. H 21 N 3 0 7 S 3 Calculated: 46.95; H, 4.14; N, 8.21; S, 18.80 (), Found: C, 47.08; H, 4.04; N, 8.07; S, 18.74 (%). mp. 199.0-200.5 ° C
実施例 3-41 Example 3-41
6 -(2-メトキシ- 2-ォキソェチル) -卜 {[(4-メチルフエニル)スルホニル] ィ ミ ノ }-3-ォキソ -8-スルフ ァニル -2, 3-ジヒ ド ロ - 1H-イ ミ ダゾ [5, 1-c] [1, 4]チアジン- 5-カルボン酸メチル 6- (2-Methoxy-2-oxoethyl) -tri {[(4-methylphenyl) sulfonyl] Imino} -3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-5-carboxylate
実施例 2- 75で合成した化合物 (3.74 g, 6.41 mmol)の THF -メタノール (1:1, 70 ml)溶液に室温で攪拌しながら 28%ナトリウムメトキシド (メ 夕ノール溶液、 2.84 g, 14.7 mmol) を加え、 反応液を 45分間攪拌した。 反応液に 1N 塩酸 (14.7 ml)を加え、 析出物を濾取し、 メタノール -水 (1:1)で洗浄後、 乾燥して表題化合物(1.13 g, 2.34 mmol)を橙色粉末と して得た。母液を濃縮して酸性に調整して酢酸ェチル(100 ml)で抽出し、 得られた有機層を酸性に調整した 7 および 15%の食塩水、 ついで飽和食 塩水で洗浄後、 硫酸ナトリウムで乾燥、 濃縮乾固して粗製の表題化合物 (2.28 g, 異性体混合物)を橙褐色粉末として得た。 To a solution of the compound synthesized in Example 2-75 (3.74 g, 6.41 mmol) in THF-methanol (1: 1, 70 ml) was added 28% sodium methoxide (ethanol solution, 2.84 g, 14.7 ml) while stirring at room temperature. mmol) and the reaction was stirred for 45 minutes. 1N Hydrochloric acid (14.7 ml) was added to the reaction solution, and the precipitate was collected by filtration, washed with methanol-water (1: 1), and dried to give the title compound (1.13 g, 2.34 mmol) as an orange powder. Was. The mother liquor was concentrated, adjusted to be acidic, extracted with ethyl acetate (100 ml), and the obtained organic layer was washed with acidified 7 and 15% saline, then with saturated brine, and dried over sodium sulfate. The residue was concentrated to dryness to give the crude title compound (2.28 g, a mixture of isomers) as an orange-brown powder.
Ή-NMR (300MHz, CDC13): 62.47 (3H, s), 3.27 (2H, s), 3.72 (3H, s) , 3.90 (3H, s), 7.38 (2H, d, J= 8.1 Hz), 7.81 (2H, d, J= 8.4 Hz), 12.11 (1H, br). Ή-NMR (300MHz, CDC1 3 ): 62.47 (3H, s), 3.27 (2H, s), 3.72 (3H, s), 3.90 (3H, s), 7.38 (2H, d, J = 8.1 Hz), 7.81 (2H, d, J = 8.4 Hz), 12.11 (1H, br).
元素分析値: C18H17N307S3として計算値: C, 44.71 ; H, 3.54; N, 8.69; S, 19.89 (%) 、 実測値: C, 44.65; H, 3.50; N, 8.40; S, 19.86 (%) . mp. 211.0-214.0°C Elemental analysis: C 18 H 17 N 3 0 7 S 3 Calculated: C, 44.71; H, 3.54 ; N, 8.69; S, 19.89 (%), Found: C, 44.65; H, 3.50 ; N, 8.40; S, 19.86 (%) .mp. 211.0-214.0 ° C
実施例 3-42 Example 3-42
N -(8, 8 -ジメチル- 1, 6 -ジォキソ- 4-スルファニル -1, 2, 6, 7, 8, 9-へキサヒ ドロ- 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン _4 -ィリデン) -4-メチル ベンゼンスルホンアミド N- (8,8-Dimethyl-1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] Benzothiazine_4-ylidene) -4-methylbenzenesulfonamide
実施例 2- 76で合成した 3-[(8,8-ジメチル- 1, 6-ジォキソ- 3- {[(4-メチ ルフエニル)スルホニル]ィミノ }- 2, 3, 6, 7, 8, 9 -へキサヒド口- 1H-ィミダ ゾ [5, 1-c] [1, 4]ベンゾチアジン- 4-ィル)スルファニル]プロピオン酸ェ チル (3.00 g, 5.46 mmol) の THF (30.0 ml) およびエタノール (30.0ml) 懸濁液に、 水酸化カリウム (1.08 g, 16.4 mmol) を加えて、 室温で 15 分間撹拌した。 反応液に 1N塩酸 (21.8ιηυ を加えて混和し、 室温で 30 分間撹拌した。 生成した沈澱物を濾取し、 水、 エタノールで洗浄後、 乾 燥して表題化合物の橙色結晶 (2.30g, 94¾) を得た。 酢酸ェチルから再 結晶して融点 255- 257°Cの橙色結晶を得た。 3-[(8,8-Dimethyl-1,6-dioxo-3-{[(4-methylphenyl) sulfonyl] imino} -2,3,6,7,8,9 synthesized in Example 2-76 -Hexahydrid-1H-imidazo [5,1-c] [1,4] benzothiazin-4-yl) sulfanyl] ethyl propionate (3.00 g, 5.46 mmol) in THF (30.0 ml) and ethanol ( Potassium hydroxide (1.08 g, 16.4 mmol) was added to the suspension, and the mixture was stirred at room temperature for 15 minutes. The reaction mixture was mixed with 1N hydrochloric acid (21.8ιηυ) and stirred at room temperature. The resulting precipitate was collected by filtration, washed with water and ethanol, dried and dried to give orange crystals of the title compound (2.30 g, 94¾) was obtained again from ethyl acetate. Crystallization yielded orange crystals with a melting point of 255-257 ° C.
IR (KBr) V: 3350-2500, 2967, 2878, 2242, 1717, 1655, 1584, 1505 cm"1. LH-NMR (CDC13) : δ 1.13 (6Η, s), 2.37 (2H, s), 2.46 (3H, s), 3.25 (2H, s), 7.39 (2H, d, J = 8.4 Hz), 7.80 (2H, d, J = 8.7 Hz), 9.50 (1H, bs). 元素分析値: C19H19N304S3として IR (KBr) V:. 3350-2500 , 2967, 2878, 2242, 1717, 1655, 1584, 1505 cm "1 L H-NMR (CDC1 3): δ 1.13 (6Η, s), 2.37 (2H, s) , 2.46 (3H, s), 3.25 (2H, s), 7.39 (2H, d, J = 8.4 Hz), 7.80 (2H, d, J = 8.7 Hz), 9.50 (1H, bs). as C 19 H 19 N 3 0 4 S 3
計算値 ( ) : C, 50.76; H, 4.26; N, 9.35; S, 21.40. Calculated (): C, 50.76; H, 4.26; N, 9.35; S, 21.40.
実測値 ) : C, 50.84; H, 4.55; N, 9.21; S, 20.49. Obtained): C, 50.84; H, 4.55; N, 9.21; S, 20.49.
実施例 3-43 Example 3-43
N-(9, 9-ジメチル -1, 6-ジォキソ- 4-スルファニル -1, 2, 6, 7, 8, 9 -へキサヒ ドロ -3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン- 4 -ィリデン) -4-メチル ベンゼンスルホンアミド N- (9,9-dimethyl-1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] Benzothiazine-4-ylidene) -4-methylbenzenesulfonamide
実施例 3-42と同様の方法により、実施例 2-77で合成した 3- [(9, 9-ジ メチル -1, 6-ジォキソ- 3- {[(4-メチルフエニル)スルホニル]イ ミ ノ }- 2, 3, 6, 7, 8, 9-へキサヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]ベンゾチアジ ン- 4-ィル)スルファニル]プロピオン酸ェチル (4.80 g, 8.73 mmol) か ら題記化合物 (4.02 g, ca.100%) を合成した。 酢酸ェチルから再結晶し て融点 256-258 の暗赤色結晶を得た。 3-[(9,9-dimethyl-1,6-dioxo-3-{[((4-methylphenyl) sulfonyl] imino) synthesized in Example 2-77 in the same manner as in Example 3-42. }-2, 3, 6, 7, 8, 9-Hexahydro-1H-imidazo [5,1-c] [1,4] benzothiazin-4-yl) sulfanyl] ethyl ester propionate (4.80 g, 8.73 mmol) to give the title compound (4.02 g, ca. 100%). Recrystallization from ethyl acetate gave dark red crystals with a melting point of 256-258.
IR (KBr) V: 3350-2500, 2973, 2922, 2868, 2255, 1717, 1651, 1586, 1505 cm— IR (KBr) V: 3350-2500, 2973, 2922, 2868, 2255, 1717, 1651, 1586, 1505 cm—
Ή-NMR (CDC13) : 51.15 (6H, s), 1.90 (2H, t, J = 6.0 Hz), 2.46 (3H, s), 3.38 (2H, t, J-6.3 Hz), 7.38 (2H, d, J=8.4 Hz), 7.79 (2H, d, J = 8.4 Hz) , 9.49 (1H, bs). Ή-NMR (CDC1 3): 51.15 (6H, s), 1.90 (2H, t, J = 6.0 Hz), 2.46 (3H, s), 3.38 (2H, t, J-6.3 Hz), 7.38 (2H, d, J = 8.4 Hz), 7.79 (2H, d, J = 8.4 Hz), 9.49 (1H, bs).
元素分析値: C19H19N304S3 ·0· 5H20として Elementary analysis: as C 19 H 19 N 3 0 4 S 3 · 0 · 5H 2 0
計算値 (%) : C, 49.76; H, 4.40; N, 9.16; S, 20.98. Calculated (%): C, 49.76; H, 4.40; N, 9.16; S, 20.98.
実測値 ) : C, 49.84; H, 4.14; N, 9.09; S, 20.78. Obtained): C, 49.84; H, 4.14; N, 9.09; S, 20.78.
実施例 3-44 Example 3-44
N -(1, 6-ジォキソ -8-メチル -4-スルファニル -1, 2, 6, 7, 8, 9-へキサヒド口 - 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 4-ィリデン) -4-メチルベン ゼンスルホンアミ ド 実施例 3-42と同様の方法により、実施例 2- 78で合成した 3- [(1, 6 -ジォ キ ソ - 8-メ チル -3- {[(4-メ チル フ エ ニル) スルホニル ] イ ミ ノ卜 2, 3, 6, 7, 8, 9-へキサヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]ベンゾチアジ ン- 4-ィル)スルファニル]プロピオン酸ェチル (10.2 g, 19.0 匪 ol) か ら題記化合物 (8.49 g, ca.100%) を合成した。 酢酸ェチルから再結晶し て融点 259- 261°Cの暗赤色結晶を得た。 N- (1,6-Dioxo-8-methyl-4-sulfanyl-1,2,6,7,8,9-hexahydride-3H-imidazo [5, 1-c] [1,4] benzothiazine- 4-ylidene) -4-methylbenzenesulfonamide In the same manner as in Example 3-42, 3-[(1,6-dioxo-8-methyl-3-{[((4-methylphenyl)) synthesized in Example 2-78. Sulfonyl] imino 2,3,6,7,8,9-hexahydro-1H-imidazo [5,1-c] [1,4] benzothiazin-4-yl) sulfanyl] ethyl ethyl propionate ( The title compound (8.49 g, ca. 100%) was synthesized from 10.2 g, 19.0 bandages. Recrystallization from ethyl acetate gave dark red crystals with a melting point of 259-261 ° C.
IR (KBr) V 3700-2700, 2955, 2926, 2851, 1717, 1663, 1584, 1493 cm"1. IR (KBr) V 3700-2700, 2955, 2926, 2851, 1717, 1663, 1584, 1493 cm " 1 .
'Η-蘭 R (CDC13) : (51.16 (3H, d, J = 6.0 Hz), 2.13-2.33 (2H, m) , 2.46 (3H, s), 2.59 (1H, d, J = 13.2 Hz), 2.71 (1H, dd, J = 18.9, 9.9 Hz), 3.76 (1H, dd, J = 19.2, 3.0 Hz), 7.88 (2H, d, J = 8.7 Hz), 8.29 (2H, d, J-8.1 Hz), 9.41 (1H, bs). '.Eta. Ran R (CDC1 3): (51.16 (3H, d, J = 6.0 Hz), 2.13-2.33 (2H, m), 2.46 (3H, s), 2.59 (1H, d, J = 13.2 Hz) , 2.71 (1H, dd, J = 18.9, 9.9 Hz), 3.76 (1H, dd, J = 19.2, 3.0 Hz), 7.88 (2H, d, J = 8.7 Hz), 8.29 (2H, d, J-8.1 Hz), 9.41 (1H, bs).
実施例 3-45 Example 3-45
N- (4-スルファニル- 2, 3 -ジヒドロ- 3H -ィミダゾ [5, 1-c] [1, 4]ベンゾチア ジン- 4-ィリデン) -4-メチルベンゼンスルホンアミド N- (4-sulfanyl-2,3-dihydro-3H-imidazo [5,1-c] [1,4] benzothiazine-4-ylidene) -4-methylbenzenesulfonamide
実施例 3-42と同様の方法により、実施例 2- 67で合成した 3-[(3- {[(4 - メチルフエニル)スルホニル]ィミノ } -卜ォキソ - 2, 3-ジヒドロ- 1H -ィミ ダゾ [5, 1-c] [1,4]ベンゾチアジン - 4-ィル)スルファニル]プロピオン酸 ェチル ( 252 mg, 0.501 匪 ol) から題記化合物 (183 mg, 91%) を合成し た。 By the same method as in Example 3-42, 3-[(3-{[(4-methylphenyl) sulfonyl] imino} -toxo-2,3-dihydro-1H-imi synthesized in Example 2-67. The title compound (183 mg, 91%) was synthesized from ethyl ethyl dazo [5,1-c] [1,4] benzothiazine-4-yl) sulfanyl] propionate (252 mg, 0.501 bandol).
IR (KBr) V: 3300-2400, 2996, 1715, 1558, 1574, 1524 cm— IR (KBr) V: 3300-2400, 2996, 1715, 1558, 1574, 1524 cm—
Ή-N R (CDC13) : δ 2.51 (3Η, s), 7.04-7.14 (2H, m) , 7.16-7.28 (2Η, in), 7.44 (2Η, d, J = 8.4 Hz), 7.94 (2H, d, J = 8. Hz), 9.29 (1H, bs), 8.69 (1H, d, J=8.4 Hz). Ή-NR (CDC1 3): δ 2.51 (3Η, s), 7.04-7.14 (2H, m), 7.16-7.28 (2Η, in), 7.44 (2Η, d, J = 8.4 Hz), 7.94 (2H, d, J = 8.Hz), 9.29 (1H, bs), 8.69 (1H, d, J = 8.4 Hz).
実施例 4-1 Example 4-1
式 ( I X) において R2がべンジル基、 R5が n-へキシル基、 R 6が水素 原子、 R 1 Qがー Z R15 (Zが S〇2、 R15が p-トルィル基)、 nが 1で ある化合物の合成 In the formula (IX), R 2 is a benzyl group, R 5 is an n-hexyl group, R 6 is a hydrogen atom, R 1 Q is —ZR 15 (Z is S〇 2 , R 15 is a p-tolyl group), Synthesis of compounds where n is 1
実施例 2-2で合成した化合物 (150 mg, 純度 95.3%、 0.266 mmol) を THF (2 ml) およびメタノール (2 ml) に溶解し、 水酸化カリウム (86mg, 1.30 mmol) を加えて室温で 30 分間撹拌した。 反応液に臭化べンジル (0.035 ml, 0.292 mmol) を加え、 室温で 2時間攪拌した。 反応液に 1 N 塩酸 (1.03ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈 澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物の橙色粉末 (110 mg、 0.208 mmol) を得た。 Compound (150 mg, purity 95.3%, 0.266 mmol) synthesized in Example 2-2 The residue was dissolved in THF (2 ml) and methanol (2 ml), potassium hydroxide (86 mg, 1.30 mmol) was added, and the mixture was stirred at room temperature for 30 minutes. Benzyl bromide (0.035 ml, 0.292 mmol) was added to the reaction solution, and the mixture was stirred at room temperature for 2 hours. 1N Hydrochloric acid (1.03 ml) was added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give the title compound as an orange powder (110 mg, 0.208 mmol).
lH -匪 R (300 MHz, 重クロ口ホルム中) : δ 0.89 (3Η, t, J= 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m) , 1.54 (2H, brquint, J= 6.8 Hz), 2.41 (3H, s), 2.90 (1H, dt, J= 13.1, 1.6 Hz), 3.12 (1H, dt, J= 13.1, 2.7 Hz), 3.53 (1H, dt, J= 9. , 6.8 Hz), 3.60 (1H, dt, J- 9.4, 6.8 Hz), 4.22 (1H, d, J= 12.6 Hz), 4.25 (1H, d, J= 12.6 Hz), 5.71 (1H, brdd, J= 2.7,lH-Maraudal R (300 MHz, in double-mouthed form): δ 0.89 (3Η, t, J = 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m), 1.54 (2H, brquint, J = 6.8 Hz), 2.41 (3H, s), 2.90 (1H, dt, J = 13.1, 1.6 Hz), 3.12 (1H, dt, J = 13.1, 2.7 Hz), 3.53 (1H, dt, J = 9. 6.8 Hz), 3.60 (1H, dt, J-9.4, 6.8 Hz), 4.22 (1H, d, J = 12.6 Hz), 4.25 (1H, d, J = 12.6 Hz), 5.71 (1H, brdd, J = 2.7,
I.6 Hz), 7.26 (2H, d, J- 8.2 Hz), 7.32 (5H, s) , 7.87 (2H, d, J= 8.2 Hz), 9.73 (1H, brs). I.6 Hz), 7.26 (2H, d, J-8.2 Hz), 7.32 (5H, s), 7.87 (2H, d, J = 8.2 Hz), 9.73 (1H, brs).
元素分析値: C26H29N303S3 として計算値: C, 59.18; H, 5.54; N, 7.96; S, 18.23 (%), 実測値; C, 59.07; H, 5.50; N, 7.94; S, 18.52 (%) . 実施例 4-2 Elemental analysis: C 26 H 29 N 3 0 3 S 3 Calculated: C, 59.18; H, 5.54 ; N, 7.96; S, 18.23 (%), Found; C, 59.07; H, 5.50 ; N, 7.94; S, 18.52 (%). Example 4-2
式 ( I X) において R 2がイソブチル基、、 R5が n -ペンチル基、 R6が メチル基、 R 1。がー Z R 1 5 ( Zが S O 2、 R 15が P-卜ルイル基)、 nが 1である化合物の合成 In the formula (IX), R 2 is an isobutyl group, R 5 is an n-pentyl group, R 6 is a methyl group, and R 1 . Synthesis of Gar ZR 1 5 (Z is SO 2, R 15 is P- Bok Ruiru group), compounds wherein n is 1
実施例 3-1で合成した化合物 (5.22 g, 11.9 mmol) および炭酸力リウ ム (2.47 g, 17.9 mmol) を DMF (87 ml) に混和し、 8 0でで 5分間撹 拌した。 反応液によう化イソブチル (1.51 ml, 13.1 mmol) を加えてさ らに 2時間撹拌した。 反応液に 1N塩酸 (30ml) を加えて混和し、 室温 で 3 0分間撹拌した。 生成した沈澱物を濾取し、 5 0 %メタノール水、 ついでメタノールで洗浄後、 乾燥して標題化合物の紫色粉末 (5.54 g、 The compound synthesized in Example 3-1 (5.22 g, 11.9 mmol) and potassium carbonate (2.47 g, 17.9 mmol) were mixed with DMF (87 ml), and the mixture was stirred at 80 for 5 minutes. Isobutyl iodide (1.51 ml, 13.1 mmol) was added to the reaction solution, and the mixture was further stirred for 2 hours. The reaction mixture was mixed with 1N hydrochloric acid (30 ml) and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with 50% aqueous methanol and then with methanol, and dried to give a purple powder of the title compound (5.54 g,
II.2 mmol) を得た。 II.2 mmol).
Ή-NMR (300 MHz, 重クロ口ホルム中): δ 0.90 (3Η, t, J= 6.8Hz), 1.02 (6H, d, J= 6.8 Hz), 1.31 (4H, m) , 1. 7 (2H, brquint, J= 7.5 Hz), 1.90 (1H, septet, J= 6.8 Hz), 2.11 (2H, brdd, J= 7.9, 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, t, J= 7.1 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). Ή-NMR (300 MHz, in double-mouthed form): δ 0.90 (3Η, t, J = 6.8 Hz), 1.02 (6H, d, J = 6.8 Hz), 1.31 (4H, m), 1.7 ( 2H, brquint, J = 7.5 Hz), 1.90 (1H, septet, J = 6.8 Hz), 2.11 (2H, brdd, J = 7.9, 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, t, J = 7.1 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C23H31N303S3 として計算値: C, 55.95; H, 6.33; N, 8.51; S, 19.48 (%), 実測値; C, 55.96; H, 6.06; N, 8.75; S, 19.56 (%) . 融点: 123.5-124. Ot: Elemental analysis: C 23 H 31 N 3 0 3 S 3 Calculated: C, 55.95; H, 6.33 ; N, 8.51; S, 19.48 (%), Found; C, 55.96; H, 6.06 ; N, 8.75; S, 19.56 (%). Melting point: 123.5-124. Ot:
実施例 4 - 3 Example 4-3
式 ( I X) において R 2が 4_ピリジルメチル基、 R 5が n-へキシル基、 R 6が水素原子、 R1 0がー Z R 1 5 i/LifiS O 2、 R 15が p -トルィル基)、 nが 1である化合物の合成 R 2 is 4_ pyridylmethyl group in formula (IX), hexyl radical R 5 is the n-, R 6 is a hydrogen atom, R 1 0 Gar ZR 1 5 i / LifiS O 2 , R 15 is p - Toruiru group ), Synthesis of compounds where n is 1
実施例 3- 2で合成した化合物 (99 mg, 0.226 mmol), 塩化ピコリル塩 酸塩 (57 mg, 0.339 mmol) および炭酸カリウム (63 mg, 0.452 匪 ol) を DMF (1.5 ml) に混和し、 室温で 8 0分間撹拌し、 さらに 8 0°Cで 2 時間半撹拌した。 反応液に 1 N塩酸 (0.68 ml) および 5 0 %メタノール 水 (2 ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈澱物 を濾取し、メ夕ノール水で洗浄後、乾燥して標題化合物の紫色粉末(82 mg、 0.155 mmol) を得た b The compound synthesized in Example 3-2 (99 mg, 0.226 mmol), picolyl chloride hydrochloride (57 mg, 0.339 mmol) and potassium carbonate (63 mg, 0.452 ol) were mixed with DMF (1.5 ml). The mixture was stirred at room temperature for 80 minutes and further at 80 ° C for 2.5 hours. 1N Hydrochloric acid (0.68 ml) and 50% aqueous methanol (2 ml) were added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. It was filtered off and the resulting precipitate was washed with main evening Knoll water, purple powder (82 mg, 0.155 mmol) in dried title compound was obtained b
Ή-N R (300 MHz, 重クロ口ホルム中): d 0.89 (3H, t, J= 6.6Hz), 1.27 (6H, br), 1. 1 (2H, brquint, J= 6.6 Hz), 2.02 (2H, t, J= 7.2 Hz), 2.43 (3H, s), 4.15 (2H, s), 6.52 (1H, brs), 7.26 (2H, d, J= 5.7 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.82, (2H, d, J= 8.3 Hz), 8.57 (2H, d, J = 5.7 Hz). Ή-NR (300 MHz, in double-hole form): d 0.89 (3H, t, J = 6.6 Hz), 1.27 (6H, br), 1.1 (2H, brquint, J = 6.6 Hz), 2.02 ( 2H, t, J = 7.2 Hz), 2.43 (3H, s), 4.15 (2H, s), 6.52 (1H, brs), 7.26 (2H, d, J = 5.7 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.82, (2H, d, J = 8.3 Hz), 8.57 (2H, d, J = 5.7 Hz).
元素分析値: C25H28N403S3 として計算値: C, 56.79; H, 5.34; N, 10.60; S, 18.19 (%) 実測値; C, 56.59; H, 5.23; N, 10.52; S, 18.08 (%) . 実施例 4 - 4 Elemental analysis: Calculated for C 25 H 28 N 4 0 3 S 3 : C, 56.79; H, 5.34; N, 10.60; S, 18.19 (%) found; C, 56.59; H, 5.23; N, 10.52 ; S, 18.08 (%). Examples 4-4
式 ( I X) において R 2が 2-フエネチル基、 R 5がエトキシカルポニル 基、 R6がメチル基、 R1 ()がー Z R1 5 (Zが S〇2、 R 1 5が p-トルィル 基)、 nが 1である化合物の合成 R 2 is 2-phenethyl in formula (IX), R 5 is ethoxy Cal Poni Le group, R 6 is a methyl group, R 1 () gar ZR 1 5 (Z is S_〇 2, R 1 5 is p- Toruiru group ), Synthesis of compounds where n is 1
実施例 3- 3で合成した化合物 (520 mg, 1.18 mmol), 臭化 2-フエネチ ル (202 ml, 1.42 mmol) および炭酸カリウム (245 mg, 1.77 mmol) を DMF (8ml) に混和し、 8 0°Cで 2時間 2 0分間撹拌した。 反応液を濃縮 後、 酢酸ェチル (50 ml) で希釈し、 0.1N 塩酸、 水および飽和食塩水で 洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去し、 残渣にメ 夕ノールを加え、 生成した沈澱物を濾取し、 メタノールで洗浄後、 乾燥 して標題化合物の濃赤色粉末 (609 mg、 1.12 mmol) を得た。 The compound synthesized in Example 3-3 (520 mg, 1.18 mmol), 2-phenyl bromide (202 ml, 1.42 mmol) and potassium carbonate (245 mg, 1.77 mmol) were added. It was mixed with DMF (8 ml) and stirred at 80 ° C for 2 hours and 20 minutes. The reaction mixture was concentrated, diluted with ethyl acetate (50 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, and methanol was added to the residue. The resulting precipitate was collected by filtration, washed with methanol, and dried to obtain a dark red powder of the title compound (609 mg, 1.12 mmol). .
lH-NMR (300 MHz, 重クロ口ホルム中): δ 1.34 (3Η, t, J= 7.1 Hz), 2.43 (3H, s) , 2.76 (3H, s), 2.99 (2H, brdd, J= 8.5, 7.2 Hz), 3.23 (2H, brdd, J= 8.5, 7.0 Hz), 4.26 (2H, q, J= 7.1 Hz), 7.21 (2H, brd, = 7.8 Hz), 7.29 (5H, m), 7.83 (2H, brd, J= 8.3 Hz), 9.80 (1H, brs). 元素分析値: C25H25N305S3 として計算値: C, 55.23; H, 4.63; N, 7.73; S, 17.69 (%)> 実測値; C, 55.10; H, 4.51; N, 7.63; S, 17.55 (%) . 実施例 4-5 lH-NMR (300 MHz, in double-mouthed form): δ 1.34 (3Η, t, J = 7.1 Hz), 2.43 (3H, s), 2.76 (3H, s), 2.99 (2H, brdd, J = 8.5) , 7.2 Hz), 3.23 (2H, brdd, J = 8.5, 7.0 Hz), 4.26 (2H, q, J = 7.1 Hz), 7.21 (2H, brd, = 7.8 Hz), 7.29 (5H, m), 7.83 . (2H, brd, J = 8.3 Hz), 9.80 (1H, brs) elemental analysis: C 25 H 25 N 3 0 5 S 3 calculated: C, 55.23; H, 4.63 ; N, 7.73; S, 17.69 (%)>Found; C, 55.10; H, 4.51; N, 7.63; S, 17.55 (%).
式 ( I X) において R2がイソブチル基、 R5および R6がともにメチル 基、 R1 ()がー Z R15 (Zが S〇2、 R 15が p -トルィル基)、 nが 1であ る化合物の合成 R 2 is an isobutyl group in the formula (IX), R 5 and R 6 are both methyl, R 1 () gar ZR 15 (Z is S_〇 2, R 15 is p - Toruiru group), n is 1 der Compound synthesis
実施例 3-4で合成した化合物 (1.98 g, 5.19 mmol) および炭酸力リウ ム (1.08 g, 7.79 mmol) を DMF (35 ml) に混和し、 8 Ot:で 5分間撹 拌した。 反応液によう イソブチル (0.72 ml, 6.23 mmol) を加えてさ らに 7 0分間撹拌した。 反応液に 1N塩酸 (10.5 ml) および水 (10 ml) を加えて混和し、 室温で 6 0分間撹拌した。 生成した沈澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物の紫色粉末 (1.96 g、 4.48 mmol) を得た。 The compound synthesized in Example 3-4 (1.98 g, 5.19 mmol) and lithium carbonate (1.08 g, 7.79 mmol) were mixed with DMF (35 ml), and the mixture was stirred at 8 Ot: for 5 minutes. Isobutyl (0.72 ml, 6.23 mmol) was added to the reaction solution, and the mixture was further stirred for 70 minutes. 1N Hydrochloric acid (10.5 ml) and water (10 ml) were added to the reaction solution, mixed, and stirred at room temperature for 60 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give a purple powder of the title compound (1.96 g, 4.48 mmol).
Ή-NMR (200 MHz, 重クロ口ホルム中): δ 1.02 (6Η, d, J= 6.6 Hz), 1.78 (3H, brq, J= 1.2 Hz), 1.90 (1H, sept, J= 6.7 Hz), 2.26 (3H, brq, J = 1.2 Hz), 2.41 (3H, s), 2.81 (2H, d, J= 7.0 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J- 8. Hz), 9.65 (1H, brs). Ή-NMR (200 MHz, in double-mouthed form): δ 1.02 (6Η, d, J = 6.6 Hz), 1.78 (3H, brq, J = 1.2 Hz), 1.90 (1H, sept, J = 6.7 Hz) , 2.26 (3H, brq, J = 1.2 Hz), 2.41 (3H, s), 2.81 (2H, d, J = 7.0 Hz), 7.27 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J- 8. Hz), 9.65 (1H, brs).
元素分析値: C19H23N303S3 として計算値: C, 52.15; H, 5.30; N, 9.60; S, 21.98 (%), 実測値; 51.97; H, 5.13; N, 9.69; S, 21.92 ( ) . 実施例 4 - 6 式 ( I X) において R2が 2-フエネチル基、 R 5および R 6がともにメチ ル基、 R 1 Qがー Z R1 5 (Zが S〇2、 R 1 5が p-トルィル基)、 nが 1で ある化合物の合成 Elemental analysis: C 19 H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30 ; N, 9.60; S, 21.98 (%), measured value; 51.97; H, 5.13; N , 9.69; S, 21.92 (). Examples 4-6 Wherein in (IX) R 2 is 2-phenethyl, R 5 and R 6 are both methylation group, R 1 Q Gar ZR 1 5 (Z is S_〇 2, R 1 5 is p- Toruiru group), n Of compounds with
実施例 3- 4で合成した化合物 (1.73 g, 4.53 mmol) および炭酸力リウ ム (0.94 g, 6.80 mmol) を DMF (35 ml) に混和し、 8 0 で 5分間撹 拌した。 反応液によう化 2-フエネチル (1.22 g, 4.99 mmol) を加えて さらに 6 0分間撹拌した。 反応液に 1 N塩酸 (9.1 ml) および水 (10 ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物の紫色粉末 (2.17 g、 4.47 mmol) を得た。 The compound synthesized in Example 3-4 (1.73 g, 4.53 mmol) and lithium carbonate (0.94 g, 6.80 mmol) were mixed with DMF (35 ml), and the mixture was stirred at 80 for 5 minutes. 2-Phenethyl iodide (1.22 g, 4.99 mmol) was added to the reaction solution, and the mixture was further stirred for 60 minutes. 1N Hydrochloric acid (9.1 ml) and water (10 ml) were added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give a purple powder of the title compound (2.17 g, 4.47 mmol).
'Η -匪 R (200 MHz, 重クロ口ホルム中) : δ 1.79 (3Η, brq, J= 1.2 Hz), 2.26 (3H, brq, J= 1.2 Hz), 2.42 (3H, s), 2.97 (2H, brdd, J= 9.2, 5.8 Hz), 3.17 (2H, dd, J= 9.0, 5.4 Hz), 7.16-7.34 (7H, m) , 7.83 (2H, d, J= 8.6 Hz), 9.66 (1H, brs). 'Η-Marauder R (200 MHz, in double-mouthed form): δ 1.79 (3Η, brq, J = 1.2 Hz), 2.26 (3H, brq, J = 1.2 Hz), 2.42 (3H, s), 2.97 ( 2H, brdd, J = 9.2, 5.8 Hz), 3.17 (2H, dd, J = 9.0, 5.4 Hz), 7.16-7.34 (7H, m), 7.83 (2H, d, J = 8.6 Hz), 9.66 (1H , brs).
元素分析値: C23H23N303S3 として計算値: C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%)ゝ 実測値; C, 56.75; H, 4.65; N, 8.71; S, 19.58 (%) . 実施例 4-7 Elemental analysis: Calculated as C 23 H 23 N 3 0 3 S 3: C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%)ゝFound; C, 56.75; H, 4.65 ; N, 8.71; S, 19.58 (%). Examples 4-7
式 ( I X) において R 2がイソブチル基、 R 5がェチル基、 R 6がメチル 基、 1^ °がー 21 1 5 、τΐ^ Ο> R 15が ρ-トルィル基)、 ηが 1であ る化合物の合成 R 2 is an isobutyl group in the formula (IX), R 5 is Echiru group, R 6 is a methyl group, 1 ^ ° gar 21 1 5, τΐ ^ Ο> R 15 is ρ- Toruiru group), eta 1 der Compound synthesis
実施例 3- 5で合成した化合物 (313 mg, 0.791 mmol) および炭酸カリ ゥム (164 mg, 1.19 mmol) を DMF (5 ml) に混和し、 8 0°Cで 5分間撹 拌した。 反応液によう化イソブチル (O. lO mI, 0.870 mmol) を加えてさ らに 1 0 5分間撹拌した。 反応液に 1 N塩酸 (1.6ml) および水 (1.0ml) を加えて混和し、 室温で 6 0分間撹拌した。 生成した沈澱物を濾取し、 メタノ一ル水およびメ夕ノールで洗浄後、 乾燥して標題化合物の赤色粉 末 (274 mg、 0.608 mmol) を得た。 The compound synthesized in Example 3-5 (313 mg, 0.791 mmol) and potassium carbonate (164 mg, 1.19 mmol) were mixed with DMF (5 ml), and the mixture was stirred at 80 ° C for 5 minutes. Isobutyl iodide (O.lO mI, 0.870 mmol) was added to the reaction solution, and the mixture was further stirred for 105 minutes. 1N Hydrochloric acid (1.6 ml) and water (1.0 ml) were added to the reaction solution, mixed, and stirred at room temperature for 60 minutes. The resulting precipitate was collected by filtration, washed with methanol and water, and dried to give a red powder of the title compound (274 mg, 0.608 mmol).
- NMR (200 MHz,. 重クロ口ホルム中): δ 1.03 (6Η, d, J= 7.8Hz), 1.08 (3H, ί, J= 7.8 Hz), 1.91 (1H, sepi, J= 7.0 Hz), 2.14 (2H, q, J= 7.4 Hz), 2.26 (3H, s), 2. 1 (3H, s), 2.83 (2H, d, J= 6.8 Hz), 7.28 (2H, d, J= 8.2 Hz), 7.86 (2H, d, J= 7.8 Hz), 9.65 (1H, brs). -NMR (200 MHz, in double chromate form): δ 1.03 (6Η, d, J = 7.8 Hz), 1.08 (3H, ί, J = 7.8 Hz), 1.91 (1H, sepi, J = 7.0 Hz) , 2.14 (2H, q, J = 7.4 Hz), 2.26 (3H, s), 2.1 (3H, s), 2.83 (2H, d, J = 6.8 Hz), 7.28 (2H, d, J = 8.2 Hz), 7.86 (2H, d, J = 7.8 Hz), 9.65 (1H, brs).
元素分析値: C2QH25N303S3 として計算値: C, 53.19; H, 5.58; , 9.30; S, 21.30 (%)、 実測値; C, 53.11; H, 5.55; N, 9.43; S, 21.15 (%) . 実施例 4-8 Elemental analysis: C 2Q H 25 N 3 0 3 S 3 Calculated: C, 53.19; H, 5.58 ;, 9.30; S, 21.30 (%), Found; C, 53.11; H, 5.55 ; N, 9.43 ; S, 21.15 (%). Example 4-8
式 ( I X) において R2が 2 -フエニルプロピル基、 R5がおよび R6がと もにメチル基、 R1 (^S— Z R15 (∑が302、 R 15が P-トルィル基)、 nが 1である化合物の合成 Wherein R 2 in (IX) is 2 - phenylpropyl group, R 5 is and R 6 Gato Moni methyl, R 1 (^ S- ZR 15 (Σ are 30 2, R 15 is P- Toruiru group) Synthesis of compounds in which n is 1
実施例 3-4で合成した化合物 (2.48 g, 6.49 mmol) および炭酸力リウ ム (1.35 g, 9.74 mmol) を DMF (40 ml) に混和し、 8 0°Cで 5分間撹 拌した。 反応液によう化 2-フエニルプロピル (1.24 ml, 7.14 ramol) を 加えてさらに 6 0分間撹拌した。 反応液に 1N塩酸 (13 ml). 水 (5 ml) およびメタノール (5 ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物 の赤紫色粉末 (3.05 g、 6.10 imol) を得た。 The compound synthesized in Example 3-4 (2.48 g, 6.49 mmol) and lithium carbonate (1.35 g, 9.74 mmol) were mixed with DMF (40 ml), and the mixture was stirred at 80 ° C for 5 minutes. 2-Phenylpropyl iodide (1.24 ml, 7.14 ramol) was added to the reaction solution, and the mixture was further stirred for 60 minutes. 1N Hydrochloric acid (13 ml). Water (5 ml) and methanol (5 ml) were added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give a red-purple powder of the title compound (3.05 g, 6.10 imol).
Ή-NMR (200 MHz, 重クロ口ホルム中): δ 1.38 (3Η, d, J= 6.6Hz), 1.78 (3H, q, J= 0.8 Hz), 2.25 (3H, q, J= 1.2 Hz), 2.42 (3H, s) , 3.03 (1H, dd, J= 13.0, 7.0 Hz), 3.10 (1H, dd, J= 15.0, 8.0 Hz), 3.22 (1H, sept, J= 5.8 Hz), 7.26 (7H, m), 7.79 (2H, d, J= 8.4 Hz) , 9.60 (1H, brs) . 元素分析値: C24H25N303S3 として計算値: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%)、 実測値; C, 57.54; H, 4.77; N, 8.31; S, 19.08 (%) . 融点: 184.0-186. O : NMR-NMR (200 MHz, in double-mouthed form): δ 1.38 (3Η, d, J = 6.6 Hz), 1.78 (3H, q, J = 0.8 Hz), 2.25 (3H, q, J = 1.2 Hz) , 2.42 (3H, s), 3.03 (1H, dd, J = 13.0, 7.0 Hz), 3.10 (1H, dd, J = 15.0, 8.0 Hz), 3.22 (1H, sept, J = 5.8 Hz), 7.26 ( . 7H, m), 7.79 ( 2H, d, J = 8.4 Hz), 9.60 (1H, brs) elemental analysis: calculated C 24 H 25 N 3 0 3 S 3: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%), found; C, 57.54; H, 4.77; N, 8.31; S, 19.08 (%). Melting point: 184.0-186. O:
実施例 4-9 , Example 4-9,
式 ( I X) において R 2がイソペンチル基、 R5がおよび R6がともにメ チル基、 R1 ()が— Z R15 (Zが S〇2、 R15が P -トルィル基)、 nが 1 である化合物の合成 R 2 is an isopentyl group in formula (IX), R 5 is and R 6 are both methylation group, R 1 () is - ZR 15 (Z is S_〇 2, R 15 is P - Toruiru group), n is 1 Of compounds that are
実施例 3-4で合成した化合物 (5.06 g, 13.3 mmol) および炭酸力リウ ム (2.38 g, 17.2 mmol) を DMF (63 ml) に混和し、 6 0°Cで 5分間撹 拌した。 反応液によう化イソペンチル (1.93 ml, 14.6 mmol) を加えて 3時間半撹拌した。 この間、 反応開始より 1および 2時間後によう化ィ ソペンチル(0.20 ml, 1.66 mmol) を加えた。 反応液に 1 N塩酸(21.2 ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成した沈澱物を 5 0 %メ 夕ノール水 (20 ml) に懸濁して濾取し、 メタノール水およびメタノール で洗浄後、 乾燥して標題化合物の赤紫色粉末 (1.96g、 4.48匪01) を得 た。 The compound synthesized in Example 3-4 (5.06 g, 13.3 mmol) and lithium carbonate (2.38 g, 17.2 mmol) were mixed with DMF (63 ml), and the mixture was stirred at 60 ° C for 5 minutes. Isopentyl iodide (1.93 ml, 14.6 mmol) was added to the reaction mixture. Stir for 3.5 hours. During this period, isopentyl iodide (0.20 ml, 1.66 mmol) was added 1 and 2 hours after the start of the reaction. The reaction mixture was mixed with 1N hydrochloric acid (21.2 ml) and stirred at room temperature for 30 minutes. The resulting precipitate was suspended in 50% aqueous methanol (20 ml), collected by filtration, washed with aqueous methanol and methanol, and dried to give a red-purple powder of the title compound (1.96 g, 4.48 band01). Obtained.
Ή-N R (200 MHz, 重クロ口ホルム中): δ 0.93 (6Η, d, J= 6.2 Hz), 1.57 (2H, quint, J= 6.9 Hz), 1.69 (2H, septet, J= 6.5 Hz), 1.79 (3H, brs), 2.26 (3H, q, J= 0.6 Hz), 2.42 (3H, s), 2.94 (2H, d, J- 7.5 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.63 (1H, brs) . 元素分析値: C2。H25N303S3 として計算値: C, 53.19; H, 5.58; N, 9.30; S, 21.30 (%), 実測値; 53.17; H, 5.87; N, 9.24; S, 21.50 (%) . 融点: 175.0-175.5°C Ή-NR (200 MHz, in double-hole form): δ 0.93 (6Η, d, J = 6.2 Hz), 1.57 (2H, quint, J = 6.9 Hz), 1.69 (2H, septet, J = 6.5 Hz) , 1.79 (3H, brs), 2.26 (3H, q, J = 0.6 Hz), 2.42 (3H, s), 2.94 (2H, d, J- 7.5 Hz), 7.28 (2H, d, J = 8.0 Hz) , 7.85 (2H, d, J = 8.0 Hz), 9.63 (1H, brs) elemental analysis:. C 2. Calculated for H 25 N 3 0 3 S 3 : C, 53.19; H, 5.58; N, 9.30; S, 21.30 (%), found; 53.17; H, 5.87; N, 9.24; S, 21.50 (%) Melting point: 175.0-175.5 ° C
実施例 4-10 Example 4-10
式 ( I X) において R 2が 2_(4-メトキシフエニル)ェチル基、 R 5がお よび R 6がともにメチル基、 R 10が— Z R 15 ( Zが S O 2、 R 15が p - 卜ルイル基)、 nが 1である化合物の合成 In the formula (IX), R 2 is a 2_ (4-methoxyphenyl) ethyl group, R 5 and R 6 are both methyl groups, R 10 is —ZR 15 (Z is SO 2 , R 15 is p-toluyl Group), synthesis of a compound in which n is 1
実施例 3- 4で合成した化合物 (240 mg, 0.629 mmol) および炭酸カリ ゥム (130 mg, 0.944 mmol) を DMF (4.0 ml) に混和し、 メタンスルホ ン酸 2- (4 -メトキシフエ二ル)ェチル (0.19 g, 純度 91%、 0.755 mmol) を加えて 8 0°Cで 5 0分間撹拌した。 反応液に 1N塩酸 (1.3ml)、 水 (2 ml) およびメタノール (2 ml) を加えて混和し、 室温で 3 0分間撹拌し た。生成した沈澱物を濾取し、メ夕ノ一ル水およびメ夕ノールで洗浄後、 乾燥して標題化合物の赤紫色粉末 (267 mg、 0.518 mmol) を得た。 The compound synthesized in Example 3-4 (240 mg, 0.629 mmol) and potassium carbonate (130 mg, 0.944 mmol) were mixed with DMF (4.0 ml), and methanesulfonate 2- (4-methoxyphenyl) was mixed. After adding ethyl (0.19 g, purity 91%, 0.755 mmol), the mixture was stirred at 80 ° C for 50 minutes. To the reaction solution, 1N hydrochloric acid (1.3 ml), water (2 ml) and methanol (2 ml) were added and mixed, followed by stirring at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol and methanol, and dried to give a purple-red powder of the title compound (267 mg, 0.518 mmol).
Ή-NMR (200 MHz, 重クロ口ホルム中): δ 1.78 (3Η, q, J= 0.8Hz), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.90 (2H, dd, J= 8.4, 6.6 Hz), 3.15 (2H, dd, J= 8. , 6.6 Hz), 3.80 (3H, s), 6.84 (2H, dt, J= 8.6, 2.5 Hz), 7.11 (2H, d, J= 8.4 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.83 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). 元素分析値: C24H25N304S3 として計算値: C, 55.90; H, 4.89; N, 8.15; S, 18.65 (%) 実測値; C, 55.64; H, 5.16; N, 8.22; S, 18.52 (%) . 融点: 174.5-175.5°C. Ή-NMR (200 MHz, in double-mouthed form): δ 1.78 (3Η, q, J = 0.8 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 2.90 (2H, dd, J = 8.4, 6.6 Hz), 3.15 (2H, dd, J = 8, 6.6 Hz), 3.80 (3H, s), 6.84 (2H, dt, J = 8.6, 2.5 Hz), 7.11 (2H, d, J = 8.4 Hz), 7.27 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.63 (1H, brs). Elemental analysis: Calculated as C 24 H 25 N 3 0 4 S 3: C, 55.90; H, 4.89; N, 8.15; S, 18.65 (%) Found; C, 55.64; H, 5.16 ; N, 8.22 ; S, 18.52 (%). Melting point: 174.5-175.5 ° C.
実施例 4-11 Example 4-11
式 ( I X) において R 2が 2, 2 -ジメチルブチル基、 R 5がおよび R 6がと もにメチル基、 R10がー Z R 1 5 (Zが S 02、 R1 5が p-トルィル基)、 nが 1である化合物の合成 R 2 is 2 in formula (IX), 2 - dimethyl butyl group, R 5 is and R 6 Gato Moni methyl, R 10 Gar ZR 1 5 (Z is S 0 2, R 1 5 is p- Toruiru Group), synthesis of a compound in which n is 1
実施例 3-4で合成した化合物 (327 mg, 0.857 imol), 参考例 102で得 た 2,2-ジメチルブタノ一ル (114 mg, 1.11 mmol)およびトリフエニルホ スフイン (324 mg, 1.20 mmol) を THF (6.5 ml) に混和し、 ァゾジカル ボン酸ジェチル (0.199 ml, 1.20 mmol) を滴下して 2時間撹拌した。 さ らに 5 0 °Cで 2 6時間撹拌した。反応液を酢酸ェチル(30 ml)で希釈し、 2 %炭酸水素ナトリゥム水溶液および飽和食塩水で洗浄後、 硫酸ナトリ ゥムで乾燥した。 溶媒を減圧下に溜去し、 残渣にメタノールを加え、 生 成した沈澱物を濾取し、 メタノールで洗浄後、 乾燥して標題化合物の紫 色粉末 (241 mg、 0.518 mmol) を得た。 Compound (327 mg, 0.857 imol) synthesized in Example 3-4, 2,2-dimethylbutanol (114 mg, 1.11 mmol) obtained in Reference Example 102, and triphenylphosphine (324 mg, 1.20 mmol) were converted into THF ( 6.5 ml), and getyl azodicarbonate (0.199 ml, 1.20 mmol) was added dropwise, followed by stirring for 2 hours. The mixture was further stirred at 50 ° C for 26 hours. The reaction solution was diluted with ethyl acetate (30 ml), washed with a 2% aqueous sodium hydrogen carbonate solution and a saturated saline solution, and dried over sodium sulfate. The solvent was evaporated under reduced pressure, methanol was added to the residue, and the generated precipitate was collected by filtration, washed with methanol, and dried to give a purple powder of the title compound (241 mg, 0.518 mmol).
^-NMR (200 MHz, 重クロ口ホルム中): 0.85 (3H, t, J= 7.5 Hz), 0.97 (6H, s), 1.36 (2H, q, J= 7.5 Hz), 1.79 (3H, q, J= 0.8 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.41 (3H, s) , 2.88 (2H, s), 7.27 (2H, d, J= 8.2 Hz), 7.87 (2H, d, J= 8.0 Hz), 9.60 (1H, brs). ^ -NMR (200 MHz, in double-mouthed form): 0.85 (3H, t, J = 7.5 Hz), 0.97 (6H, s), 1.36 (2H, q, J = 7.5 Hz), 1.79 (3H, q , J = 0.8 Hz), 2.26 (3H, q, J = 0.8 Hz), 2.41 (3H, s), 2.88 (2H, s), 7.27 (2H, d, J = 8.2 Hz), 7.87 (2H, d , J = 8.0 Hz), 9.60 (1H, brs).
元素分析値: C21H N303S3 として計算値: C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), 実測値; 54.26; H, 5.89; N, 9.12; S, 20.61 (%) . 融点: 177.5-178. c Elemental analysis: Calculated for C 21 HN 3 0 3 S 3 : C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), found; 54.26; H, 5.89; N, 9.12; S, 20.61 (%). Melting point: 177.5-178. C
実施例 4- Π Example 4-Π
式 ( I X) において R 2が 4-ェチルフエニル基、 R 5がおよび R 6がとも にメチル基、 R1 Gがー Z R 1 5 (Zが S〇2、 R1 5が P-トルィル基)、 n が 1である化合物の合成 Wherein R 2 is 4-Echirufueniru group in (IX), a methyl group R 5 is and R 6 Gatomo, R 1 G Gar ZR 1 5 (Z is S_〇 2, R 1 5 is P- Toruiru group), Synthesis of compounds where n is 1
実施例 3- 4で合成した化合物 (n2 mg, 0.582 mmol)、 4-ェチルチオフ ェノ一ル(91.2 mg, 0.640 mmol)およびトリフエニルホスフィン(236 mg, 0.873 mmol)を THF (4.5 ml)に混和し、ァゾジ力ルポン酸ジェチル(0.145 ml, 0.873 mmol) を滴下して 2 2時間撹拌した。 さらに 5 0 で 1 8時 間撹拌した。 反応液にトリフエニルホスフィン (157 mg, 0.582 mmol) およびァゾジ力ルポン酸ジェチル (0.097 ml, 0.582 mmol) を滴下して、 さらに 5 0°Cで 3日間撹拌した。反応液を酢酸ェチル(30 ml)で希釈し、The compound synthesized in Example 3-4 (n2 mg, 0.582 mmol), 4-ethylthiophenol (91.2 mg, 0.640 mmol) and triphenylphosphine (236 mg, 0.873 mmol) was mixed with THF (4.5 ml), and azodiphenol getyl sulfonate (0.145 ml, 0.873 mmol) was added dropwise, followed by stirring for 22 hours. The mixture was further stirred at 50 for 18 hours. To the reaction solution were added dropwise triphenylphosphine (157 mg, 0.582 mmol) and getyl azodisulfonic acid (0.097 ml, 0.582 mmol), and the mixture was further stirred at 50 ° C for 3 days. Dilute the reaction with ethyl acetate (30 ml)
2 %炭酸水素ナトリゥム水溶液、 0.1 塩酸および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去後、 残渣をシリカゲルThe extract was washed with a 2% aqueous sodium hydrogen carbonate solution, 0.1 hydrochloric acid and saturated saline, and then dried over sodium sulfate. After evaporating the solvent under reduced pressure, the residue was silica gel
6 0 (10 g) のカラムクロマトグラフィ—に付した。 酢酸ェチル: へキ サン (3 0 : 7 0) の溶出画分を減圧下濃縮乾固し、 残渣にメタノール を加え、 生成した沈澱物を濾取し、 メタノールで洗浄後、 乾燥して標題 化合物の紫色粉末 (36 mg、 0.074 mmol) を得た。 60 (10 g) of column chromatography. Ethyl acetate: The fraction eluted with hexane (30: 70) was concentrated to dryness under reduced pressure, methanol was added to the residue, the resulting precipitate was collected by filtration, washed with methanol, and dried to give the title compound To give a purple powder (36 mg, 0.074 mmol).
LH-NMR (200 MHz, 重クロ口ホルム中): δ 1.26 (3Η, t, J= 7.5 Hz), 1.60 (3H, q, J= 0.9 Hz), 2.23 (3H, q, J= 1.2 Hz), 2.43 (3H, s), 2.70 (2H, q, J= 7.5 Hz), 7.22 (2H, d, J- 7.8 Hz), 7.30 (2H, d, J= 7.8 Hz), 7.45 (2H, d, J= 8.1 Hz), 7.89 (2H, d, J= 8.1 Hz), 9.63 (1H, brs). 元素分析値: C23H23N303S3 として計算値: C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%)ゝ 実測値; C, 56.82; H, 4.52; N, 8.42; S, 19.84 (%) . 融点 : 183.0-184.5 L H-NMR (200 MHz, in double-mouthed form): δ 1.26 (3Η, t, J = 7.5 Hz), 1.60 (3H, q, J = 0.9 Hz), 2.23 (3H, q, J = 1.2 Hz) ), 2.43 (3H, s), 2.70 (2H, q, J = 7.5 Hz), 7.22 (2H, d, J-7.8 Hz), 7.30 (2H, d, J = 7.8 Hz), 7.45 (2H, d , J = 8.1 Hz), 7.89 (2H, d, J = 8.1 Hz), 9.63 (1H, brs) elemental analysis: calculated C 23 H 23 N 3 0 3 S 3:. C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%) ゝ Found; C, 56.82; H, 4.52; N, 8.42; S, 19.84 (%). Melting point: 183.0-184.5
実施例 4-13 Example 4-13
式 ( I X) において R2が 4-メトキシフエ二ル基、 R5がおよび R6がと もにメチル基、 R 1。が— Z R 1 5 (Zが S 02、 R 1 5が P-トルィル基)、 nが 2である化合物の合成 In the formula (IX), R 2 is a 4-methoxyphenyl group, R 5 and R 6 are both a methyl group, R 1 . Is— ZR 15 (Z is S 0 2 , R 15 is a P-tolyl group) and n is 2
実施例 3- 4で合成した化合物 (257 mg, 0.674 mmol), 4-メトキシベン ゼンチォ一ル(0.131 ml, 1.01 mmol)およびトリフエニルホスフィン(729 mg, 2.69 mmol) を THF (5 ml) に混和し、 6 0 °Cで撹拌しながらァゾジ 力ルポン酸ジイソプロピル (0.547 ml, 2.69 mmol) の THF (1.5 ml) 溶 液を 2時間半かけて滴下し、 さらに 6 0 °Cで 1時間撹拌した。 反応液を 酢酸ェチル (30 ml) で希釈し、 2 %炭酸水素ナトリウム水溶液、 0.1N 塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒 を減圧下に溜去後、残渣にメタノールを加え、生成した沈澱物を濾取し、 メタノールで洗浄後、 乾燥して標題化合物の赤紫色粉末 (169 mg、 0.325 mmol) を得た。 The compound synthesized in Example 3-4 (257 mg, 0.674 mmol), 4-methoxybenzene (0.131 ml, 1.01 mmol) and triphenylphosphine (729 mg, 2.69 mmol) were mixed with THF (5 ml). While stirring at 60 ° C, a solution of diisopropyl azodisulfonic acid (0.547 ml, 2.69 mmol) in THF (1.5 ml) was added dropwise over 2.5 hours, and the mixture was further stirred at 60 ° C for 1 hour. The reaction solution was diluted with ethyl acetate (30 ml), washed with a 2% aqueous sodium hydrogen carbonate solution, 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. solvent After distilling off under reduced pressure, methanol was added to the residue. The resulting precipitate was collected by filtration, washed with methanol, and dried to obtain a red-purple powder of the title compound (169 mg, 0.325 mmol).
'Η-雇 R (200 MHz, 重クロ口ホルム中): δ 1.81 (3Η, q, J= 1.2 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.43 (3H, s), 3.82 (3H, s), 6.86 (2H, dt, J= 8.8, 2.0 Hz), 7.28 (2H, d, J= 8.2 Hz), 7.50 (2H, dt, J= 8.8, 2.0 Hz) , 7.82 (2H, d, 】= 8.2 Hz), 9.56 (1H, brs). 'Η-employed R (200 MHz, in double-mouthed form): δ 1.81 (3Η, q, J = 1.2 Hz), 2.26 (3H, q, J = 0.8 Hz), 2.43 (3H, s), 3.82 ( 3H, s), 6.86 (2H, dt, J = 8.8, 2.0 Hz), 7.28 (2H, d, J = 8.2 Hz), 7.50 (2H, dt, J = 8.8, 2.0 Hz), 7.82 (2H, d ,) = 8.2 Hz), 9.56 (1H, brs).
元素分析値: C22H21N304S4 として計算値: C, 50.85; H, 4.07; N, 8.09; S, 24.68 (%)、 実測値; C, 50.87; H, 4.03; N, 8.01; S, 24.15 (%) . 融点 : 197.0-198.5Ό Elemental analysis: C 22 H 21 N 3 0 4 S 4 Calculated: C, 50.85; H, 4.07 ; N, 8.09; S, 24.68 (%), Found; C, 50.87; H, 4.03 ; N, 8.01; S, 24.15 (%). Melting point: 197.0-198.5Ό
実施例 4 - 14 Example 4-14
N-[8- (ィソブチルスルファニル)-5-メチル -3-ォキソ -6-プロピル- 2, 3 - ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン -1-ィリデン] -4-メチルべ ンゼンスルホンアミ ド N- [8- (isobutylsulfanyl) -5-methyl-3-oxo-6-propyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene ] -4-Methylbenzenesulfonamide
実施例 3-13で得られた N- (5-メチル -3-ォキソ -6-プロピル- 8-スルフ ァニル -2, 3-ジヒ ド口- 1H-ィミダゾ [5,卜 c] [1, 4]チアジン-卜ィ リデ ン) -4-メチルベンゼンスルホンアミ ド (l. OOg, 2.44 mmol) と炭酸カリ ゥム (506 mg, 3.66mmol) の N, N-ジメチルホルムアミ ド (15.0 ml) の 懸濁液に、 よう化イソブチル (0, 309 ml, 2.69 mmol) の N,N-ジメチル ホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を 80°Cで 40分加熱 した後、 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (852 mg, 75%) を暗 赤色結晶として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 144- 146 の暗赤色結晶を得た。 N- (5-methyl-3-oxo-6-propyl-8-sulfanyl-2,3-dihydrido-1H-imidazo obtained in Example 3-13 [5, tric] [1, 4 ] Thiazine-trilidene) -4-methylbenzenesulfonamide (l.OOg, 2.44 mmol) and potassium carbonate (506 mg, 3.66 mmol) in N, N-dimethylformamide (15.0 ml) To the suspension was added a solution of isobutyl iodide (0,309 ml, 2.69 mmol) in N, N-dimethylformamide (1.00 ml). After the reaction mixture was heated at 80 ° C for 40 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (852 mg, 75%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether afforded dark red crystals, mp 144-146.
赤外吸収スペクトル(IR) (KBr中) レ : 3218, 2961, 2930, 2872, 1732, 1659, 1615, 1557, 1507, 1495 cm'1 Infrared absorption spectrum (IR) (in KBr) Re: 3218, 2961, 2930, 2872, 1732, 1659, 1615, 1557, 1507, 1495 cm ' 1
- NMR (CDC13) : δ 0.950 (3Η, t, J = 7.2 Hz), 1.02 (6H, d, J = 6.6 Hz), 1.40-1.61 (2H, m), 1.82-1.98 (1H, m), 2.10 (2H, t, J = 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 6.6 Hz), 7.86 (2H, d, J=8.4 Hz), 9.61 (IH, bs). - NMR (CDC1 3): δ 0.950 (3Η, t, J = 7.2 Hz), 1.02 (6H, d, J = 6.6 Hz), 1.40-1.61 (2H, m), 1.82-1.98 (1H, m), 2.10 (2H, t, J = 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 6.6 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.61 (IH, bs).
元素分析値: C21H27N303S3として Elemental analysis: as C 21 H 27 N 3 0 3 S 3
計算値 (%) : C, 54.17; H, 5.84; N, 9.02; S, 20.66. Calculated value (%): C, 54.17; H, 5.84; N, 9.02; S, 20.66.
実測値 ( : C, 54.02; H, 5.84; N, 9.05; S, 20.41. Obtained value (: C, 54.02; H, 5.84; N, 9.05; S, 20.41.
実施例 4-15 Example 4-15
N - [6-ブチル -8- (ィソプチルスルファ二ル)- 5-メチル -3-ォキソ -2, 3 -ジ ヒドロ- 1H-ィミダゾ [5,l-c] [1, 4]チアジン -卜ィリデン ]-4-メチルベン ゼンスルホンアミ ド N- [6-butyl-8- (isoptylsulfanyl) -5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine Ylidene] -4-methylbenzene benzenesulfonamide
実施例 4-14 と同様の方法により、 実施例 3-15で得られた N-(6 -プチ ル- 5-メチル -3-ォキソ -8-スルファニル- 2, 3-ジヒ ド口 -1H-ィミダゾ According to a method similar to that of Example 4-14, N- (6-butyl-5-methyl-3-oxo-8-sulfanyl-2,3-dihydrochloride-1H- obtained in Example 3-15 was obtained. Imidazo
[5, 1-c] [1,4]チアジン- 1-ィリデン)-4_メチルベンゼンスルホンアミド (1.00 g, 2.36 mmol) から題記化合物 (620 mg, 55%) を合成した。 酢 酸ェチル -ジィソプロピルエーテルから再結晶して融点 116-U7°Cの暗 赤色結晶を得た。 ■ 赤外吸収スペクトル(IR) ( r中) レ : 3214, 2957, 2930, 2870, 1732,[5, 1-c] [1,4] thiazine-1-ylidene) -4_methylbenzenesulfonamide (1.00 g, 2.36 mmol) was used to synthesize the title compound (620 mg, 55%). Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals with a melting point of 116-U7 ° C. ■ Infrared absorption spectrum (IR) (in r) Re: 3214, 2957, 2930, 2870, 1732,
1657, 1617, 1563, 1557, 1507, 1495 cm—1. 1657, 1617, 1563, 1557, 1507, 1495 cm- 1 .
^- MR (CDC13) : δ 0.929 (3Η, t, J = 6.6 Hz), 1.03 (6H, d, J = 6.6 Hz), 1.26-1.51 (4H, m), 1.80-1.98 (IH, m), 2.12 (2H, t, J = 8.0 Hz), 2.26 (3H, s), 2. 1 (3H, s) , 2.82 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 7.0 Hz), 7.86 (2H, d, J = 8. Hz), 9.60 (IH, bs). ^ - MR (CDC1 3): δ 0.929 (3Η, t, J = 6.6 Hz), 1.03 (6H, d, J = 6.6 Hz), 1.26-1.51 (4H, m), 1.80-1.98 (IH, m) , 2.12 (2H, t, J = 8.0 Hz), 2.26 (3H, s), 2.1 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 7.0) Hz), 7.86 (2H, d, J = 8. Hz), 9.60 (IH, bs).
元素分析値: C22H29N303S3として Elementary analysis: as C 22 H 29 N 3 0 3 S 3
計算値 ) : C, 55.09; H, 6.09; N, 8.76; S, 20.05. Calculated values): C, 55.09; H, 6.09; N, 8.76; S, 20.05.
実測値 ) : C, 54.99; H, 6.04; N, 8.76; S, 19.98. Obtained): C, 54.99; H, 6.04; N, 8.76; S, 19.98.
実施例 4-16 Example 4-16
N - [6-ィソブチル 8 -(ィソブチルスルファ二ル)- 5-メチル -3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5,卜 c] [1, 4]チアジン-卜ィリデン] -4-メチ 実施例 4-14と同様の方法により、 実施例 3-16で得られた N-(6-ィソ ブチル 5-メチル -3-ォキソ -8-スルファニル -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン) -4-メチルベンゼンスルホンアミ ド (1.00 g, 2.36 mmol) から題記化合物 (924 mg, 82%) を合成した。 酢 酸ェチル -ジィソプロピルエーテルから再結晶して融点 150-152^の暗 赤色結晶を得た。 N- [6-isobutyl 8- (isobutylsulfanyl) -5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-trilidene ] -4-Methyl N- (6-isobutyl 5-methyl-3-oxo-8-sulfanyl-2,3-dihydro obtained in Example 3-16 by the same method as in Example 4-14. -1H-imidazo [5, 1-c] [1,4] thiazine-triylidene) -4-methylbenzenesulfonamide (1.00 g, 2.36 mmol) was used to synthesize the title compound (924 mg, 82%). Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals with a melting point of 150-152 ^.
赤外吸収スペクトル(IR) (KBr中) リ: 3372, 3248, 2959, 2930, 2870, 1740, 1655, 1618, 1563, 1495 cm一 Infrared absorption spectrum (IR) (in KBr): 3372, 3248, 2959, 2930, 2870, 1740, 1655, 1618, 1563, 1495 cm
Ή-NMR (CDC13) : δ 0.946 (6Η, d, J-6.6 Hz), 1.02 (6H, d, J = 7.0 Hz), 1.74-1.98 (2H, m) , 2.03 (2H, d, J = 7.4 Hz), 2.26 (3H, s) , 2. 1 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 7.0 Hz), 7.86 (2H, d, J-8.2 Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3): δ 0.946 (6Η, d, J-6.6 Hz), 1.02 (6H, d, J = 7.0 Hz), 1.74-1.98 (2H, m), 2.03 (2H, d, J = 7.4 Hz), 2.26 (3H, s), 2.1 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 7.0 Hz), 7.86 (2H, d, J-8.2 Hz), 9.62 (1H, bs).
元素分析値: C22H29N303S3として Elementary analysis: as C 22 H 29 N 3 0 3 S 3
計算値 ) : C, 55.09; H, 6.09; N, 8.76; S, 20.05. Calculated values): C, 55.09; H, 6.09; N, 8.76; S, 20.05.
実測値 ) : C, 55.10; H, 6.07; N, 8.09; S, 20.07. Obtained): C, 55.10; H, 6.07; N, 8.09; S, 20.07.
実施例 4- 17 Example 4-17
N_{5, 6 -ジメチル -8- [(2, 2 -ジフエニルェチル)スルファ二ル]- 3-ォキソ - 2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) -4-メチ 実施例 4-14と同様の方法により、よう化イソブチルの代わりにメタン スルホン酸 2, 2-ジフエニルェチル エステル (261 mg, 0.944 mmol) を 用いて、 実施例 3-4で得られた N-(5, 6-ジメチル -3-ォキソ -8-スルファ ニル- 2, 3-ジヒドロ- 1H-ィミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン) - 4- メチルベンゼンスルホンアミ ド (300 mg, 0.786 mmol) から題記化合物 (690 mg, 38%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶 して融点 192- 194^の暗赤色結晶を得た。 N_ {5,6-dimethyl-8-[(2,2-diphenylethyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-Yylidene) -4-methyl Example 3-4 in the same manner as in Example 4-14, except that 2,2-diphenylethyl methanesulfonate (261 mg, 0.944 mmol) was used instead of isobutyl iodide. N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-triylidene) obtained in Step 4 The title compound (690 mg, 38%) was synthesized from methylbenzenesulfonamide (300 mg, 0.786 mmol). Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 192-194 ^.
赤外吸収スペクトル(IR) (KBr中) レ : 3231, 3061, 3029, 2924, 1732, 1665, 1615, 1563, 1495 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3231, 3061, 3029, 2924, 1732, 1665, 1615, 1563, 1495 cm " 1 .
Ή-NMR (CDC13) : δ 1.77 (3Η, d, J = l. Hz), 2.24 (3H, d, J = l. Hz), 2.41 (3H, s), 3.63 (2H, d, J = 8.0 Hz), 4.25 (1H, t, 7.6 Hz), 7.12-7.38 (12H, m) , 7.67 (2H, d, J = 8.4 Hz), 9.52 (1H, bs). 元素分析値: C29H27N303S3として Ή-NMR (CDC1 3): δ 1.77 (. 3Η, d, J = l Hz), 2.24 (. 3H, d, J = l Hz), 2.41 (3H, s), 3.63 (2H, d, J = 8.0 Hz), 4.25 (1H, t, 7.6 Hz), 7.12-7.38 (12H, m), 7.67 (2H, d, J = 8.4 Hz), 9.52 (1H, bs). Elemental analysis: C 29 H 27 N 3 0 3 S 3
計算値 ) : C, 62.01; H, 4.84; N, 7.48; S, 17.12. Calculated)): C, 62.01; H, 4.84; N, 7.48; S, 17.12.
実測値 (%) : C, 61.69; H, 4.71; N, 7.34; S, 16.91. Found (%): C, 61.69; H, 4.71; N, 7.34; S, 16.91.
実施例 4-18 Example 4-18
N- {5,6-ジメチル- 8_[(3, 3-ジフエ二ルプロピル)スルファニル ]-3-ォキ ソ- 2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン) -4 -メ 実施例 4-14と同様の方法により、よう化イソブチルの代わりにメタン スルホン酸 3, 3-ジフエ二ルプロピル エステル (268 mg, 0.944 mmol) を用いて、 実施例 3-4で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルフ ァニル -2, 3 -ジヒ ド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィ リデ ン)- 4-メチルベンゼンスルホンアミド (300 mg, 0.786 mmol) から題記 化合物 (284 mg, 63¾) を合成した。 酢酸ェチル -ジェチルェ一テルから 再結晶して融点 146- 148°Cの暗赤色結晶を得た。 N- {5,6-dimethyl-8 _ [(3,3-diphenylpropyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] Thiazine-1-ylidene) -4-me A method similar to that of Example 4-14, except that methanesulfonic acid 3,3-diphenylpropyl ester (268 mg, 0.944 mmol) was used instead of isobutyl iodide. N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrogen-1H-imidazo [5,1-c] [1,4] thiazine obtained in Example 3-4 -Trilidene)-The title compound (284 mg, 63¾) was synthesized from 4-methylbenzenesulfonamide (300 mg, 0.786 mmol). The crystal was recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 146-148 ° C.
赤外吸収スペクトル(IR) (KBr中) リ ': 3225, 3061, 3027, 2924, 1736, 1665, 1611, 1561, 1493 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3225, 3061, 3027, 2924, 1736, 1665, 1611, 1561, 1493 cm " 1 .
Ή-NMR (CDC13) : δ 1.73 (3Η, s), 2.24 (3H, s), 2.34-2. 8 (2H, m) , 2.41 (3H, s), 2.85 (2H, t, J = 7.0 Hz), 4.01 (1H, t, J = 7.0 Hz), 7.25-7.38 (12H, m) , 7.86 (2H, d, J = 8.0 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): . Δ 1.73 (3Η, s), 2.24 (3H, s), 2.34-2 8 (2H, m), 2.41 (3H, s), 2.85 (2H, t, J = 7.0 Hz), 4.01 (1H, t, J = 7.0 Hz), 7.25-7.38 (12H, m), 7.86 (2H, d, J = 8.0 Hz), 9.60 (1H, bs).
元素分析値: C3。H29N303S3'0.2H20として Elemental analysis: C 3. H 29 N 3 0 3 S 3 '0.2 As H 2 0
計算値 ) : C, 62.19; H, 5.11; N, 7.25; S, 16.60. Calculated)): C, 62.19; H, 5.11; N, 7.25; S, 16.60.
実測値 (%) : C, 62.02; H, 5.02; N, 7.25; S, 16.55. Found (%): C, 62.02; H, 5.02; N, 7.25; S, 16.55.
実施例 4-19 Example 4-19
N-{5, 6-ジメチル- 8- [(4-フエニルブチル)スルファニル ]-3-ォキソ -2, 3 - ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン卜 4-メチルベ ンゼンスルホンアミド N- {5,6-dimethyl-8-[(4-phenylbutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene 4-Methylbenzenesulfonamide
実施例 4-14と同様の方法により、よう化ィソブチルの代わりにメタン スルホン酸 4-フエニルブチル エステル (197 mg, 0.865 mmol) を用い て、 実施例 3-4で得られた N- (5,6-ジメチル- 3-ォキソ -8-スルファニル -I, 3-ジヒド口- IH -ィミダゾ [5, 1- c] [1, 4]チアジン-卜ィリデン) -4 -メチ ルベンゼンスルホンアミド (300 mg, 0.786 mmol) から題記化合物 (283 mg, 70¾) を合成した。 酢酸ェチルから再結晶して融点 158- 159°Cの暗赤 色結晶を得た。 According to a method similar to that of Example 4-14, 4-phenylbutyl methanesulfonate (197 mg, 0.865 mmol) was used in place of isobutyl iodide to obtain N- (5,6) obtained in Example 3-4. -Dimethyl-3-oxo-8-sulfanyl -I, 3-Dihydrido-IH-imidazo [5,1-c] [1,4] thiazine-trilidene) -4-methylbenzenesulfonamide (300 mg, 0.786 mmol) from the title compound (283 mg, 70¾) was synthesized. Recrystallization from ethyl acetate gave dark red crystals with a melting point of 158-159 ° C.
赤外吸収スペクトル(IR) (KBr中) レ: 3196, 3063, 3027, 2930, 1725, 1663, 1613, 1557, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3196, 3063, 3027, 2930, 1725, 1663, 1613, 1557, 1497 cm " 1 .
•H-NMR (CDC13) : δ 1.65-1.82 (4Η, m) , 1.77 (3H, s), 2.26 (3H, s), 2.40 (3H, s), 2.59-2.70 (2H, m) , 2.88-3.02 (2H, m) , 7.12-7.34 (7H, m), 7.84 (2H, d, J = 8.0 Hz), 9.60(1H, bs). • H-NMR (CDC1 3) : δ 1.65-1.82 (4Η, m), 1.77 (3H, s), 2.26 (3H, s), 2.40 (3H, s), 2.59-2.70 (2H, m), 2.88 -3.02 (2H, m), 7.12-7.34 (7H, m), 7.84 (2H, d, J = 8.0 Hz), 9.60 (1H, bs).
元素分析値: C25H27N303S3として Elemental analysis: as C 25 H 27 N 3 0 3 S 3
計算値 ) : C, 58.45; H, 5.30; N, 8.18; S, 18.73. Calculated values): C, 58.45; H, 5.30; N, 8.18; S, 18.73.
実測値 (%) : C, 58.05; H, 5.34; N, 8.08; S, 18.42. Found (%): C, 58.05; H, 5.34; N, 8.08; S, 18.42.
実施例 4-20 Example 4-20
N-{5, 6 -ジメチル -8- (へキシルスルファニル)-3-ォキソ - 2, 3-ジヒドロ -1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }-4-メチルベンゼンス ルホンアミ ド N- {5,6-dimethyl-8- (hexylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene} -4 -Methylbenzenesulfonamide
実施例 4-14 と同様の方法により、 よう化イソブチルの代わりに 卜ブ 口モへキサン (143 mg, 0.865 mmol) を用いて、 実施例 3- 4で得られた N-(5, 6-ジメチル- 3-ォキソ -8-スルファニル -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン) -4-メチルベンゼンスルホンアミ ド (300 mg, 0.786 mmol) から題記化合物 (296 mg, 81¾) を合成した。 酢 酸ェチルから再結晶して融点 157- 158 の暗赤色結晶を得た。 The N- (5,6- Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide (300 mg, 0.786) mmol) to give the title compound (296 mg, 81¾). Recrystallization from ethyl acetate gave dark red crystals with a melting point of 157-158.
赤外吸収スペクトル(IR) (KBr中) リ: 3227, 2955, 2928, 2859, 1748, 1719, 1661, 1613, 1559, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr): 3227, 2955, 2928, 2859, 1748, 1719, 1661, 1613, 1559, 1497 cm " 1 .
Ή-NMR (CDCI3) : δ 0.899 (3Η, t, J = 6.6 Hz), 1. 4-1.48 (6H, m), 1.58-1.75 (2H, m) , 1.79 (3H, d, J = l.2 Hz), 2.26 (3H, d, J = l.0 Hz), 2. 2 (3H, s), 2.93 (2H, t, J = 7.6 Hz), 7.28 (2H, d, J = 7.6 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.61 (1H, bs). Ή-NMR (CDCI3): δ 0.899 (3Η, t, J = 6.6 Hz), 1.4-1.48 (6H, m), 1.58-1.75 (2H, m), 1.79 (3H, d, J = l. 2 Hz), 2.26 (3H, d, J = 1.0 Hz), 2.2 (3H, s), 2.93 (2H, t, J = 7.6 Hz), 7.28 (2H, d, J = 7.6 Hz) , 7.86 (2H, d, J = 8.4 Hz), 9.61 (1H, bs).
元素分析値: C21H27N303S3として 計算値 (%) : C, 54.17; H, 5.84; N, 9.02; S, 20.66. Elemental analysis: as C 21 H 27 N 3 0 3 S 3 Calculated value (%): C, 54.17; H, 5.84; N, 9.02; S, 20.66.
実測値 (%) : C, 54.14; H, 5.69; N, 9.00; S, 20.66. Found (%): C, 54.14; H, 5.69; N, 9.00; S, 20.66.
実施例 4-21 Example 4-21
N-{8- (デシルスルファニル) -5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H - イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホ ンアミ ド N- {8- (decylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene} -4- Methylbenzene sulfonamide
実施例 4-14 と同様の方法により、 よう化イソブチルの代わりに 1 -ブ ロモデカン (0.183 ml, 0.865 miol) を用いて、 実施例 3- 4で得られた N-(5, 6 -ジメチル -3-ォキソ -8-スルファニル- 2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン)-4_メチルベンゼンスルホンアミ ド (300 mg, 0.786 mmol) から題記化合物 (78.4 mg, 19%) を合成した。 酢酸ェチルから再結晶して融点 121-123°Cの暗赤色結晶を得た。 In the same manner as in Example 4-14, using 1-bromodecane (0.183 ml, 0.865 miol) in place of isobutyl iodide, the N- (5,6-dimethyl- From 3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene) -4_methylbenzenesulfonamide (300 mg, 0.786 mmol) The title compound (78.4 mg, 19%) was synthesized. Recrystallization from ethyl acetate gave dark red crystals with a melting point of 121-123 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3229, 2924, 2853, 1748, 1717, 1661 , 1611, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr): 3229, 2924, 2853, 1748, 1717, 1661, 1611, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 0.888 (3Η, i, J = 6.6 Hz), 1.14-1.48 (1 H, m) , 1.61-1.72 (2H, m), 1.79 (3H, s), 2.27 (3H, s), 2.42 (3H, s), 2.93 (2H, t, J = 7.2 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.6 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): δ 0.888 (3Η, i, J = 6.6 Hz), 1.14-1.48 (1 H, m), 1.61-1.72 (2H, m), 1.79 (3H, s), 2.27 (3H , S), 2.42 (3H, s), 2.93 (2H, t, J = 7.2 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.6 Hz), 9.61 (1H , Bs).
元素分析値: C25H35N303S3として Elemental analysis: as C 25 H 35 N 3 0 3 S 3
計算値 ) : C, 57.55; H, 6.76; N, 8.05; S, 18. 4. Calculated)): C, 57.55; H, 6.76; N, 8.05; S, 18.4.
実測値 ) : C, 57.24; H, 6.76; N, 8.18; S, 18.20. Found): C, 57.24; H, 6.76; N, 8.18; S, 18.20.
実施例 4-22Example 4-22
-{5, 6-ジメチル- 8- {[2- (1-ナフチル)ェチル]スルファニル }- 3-ォキソ -2, 3-ジヒドロ -1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン) -4-メチ ルベンゼンスルホンアミ ド -{5,6-Dimethyl-8-{[2- (1-naphthyl) ethyl] sulfanyl} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1 -ylidene) -4-methylbenzenesulfonamide
実施例 4-14と同様の方法により、よう化イソプチルの代わりにメタン スルホン酸 2- (1-ナフチル)ェチル エステル (206 mg, 0.865 mmol) を 用いて、 実施例 3- 4で得られた N- (5, 6-ジメチル -3-ォキソ -8-スルファ ニル- 2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) - 4 - ミ ド (300 mg, 0.786 匪 ol) から題記化合物 (254 mg, 61%) を合成した。 酢酸ェチルから再結晶して融点 203- 204 の暗赤色結晶を得た。 According to a method similar to that of Example 4-14, 2- (1-naphthyl) ethyl methanesulfonate (206 mg, 0.865 mmol) was used instead of isoptyl iodide, to obtain N obtained in Example 3-4. -(5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene)-4- The title compound (254 mg, 61%) was synthesized from mid (300 mg, 0.786 bandol). Recrystallization from ethyl acetate gave dark red crystals with a melting point of 203-204.
赤外吸収スペクトル(IR) (KBr中) レ : 3220, 3052, 2928, 1721, 1663, 1601, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3220, 3052, 2928, 1721, 1663, 1601, 1557 cm " 1 .
'H-NMR (CDC13) : δ 1.74 (3Η, d, J = l.0 Hz), 2.25 (3H, d, J = l.0 Hz), 'H-NMR (CDC1 3) : δ 1.74 (3Η, d, J = l.0 Hz), 2.25 (3H, d, J = l.0 Hz),
2.43 (3H, s), 3.24-3.37 (2H, m) , 3.38-3.50 (2H, m) , 7.20-7.60 (6H, m), 7.74-7.92 (4H, m) , 7.96-8.04 (1H, m), 9.62 (1H, bs). 2.43 (3H, s), 3.24-3.37 (2H, m), 3.38-3.50 (2H, m), 7.20-7.60 (6H, m), 7.74-7.92 (4H, m), 7.96-8.04 (1H, m ), 9.62 (1H, bs).
元素分析値: C27H25N303S3として Elementary analysis: as C 27 H 25 N 3 0 3 S 3
計算値 (%) : C, 58.57; H, 4.91; N, 7.59; S, 17.37. Calculated value (%): C, 58.57; H, 4.91; N, 7.59; S, 17.37.
実測値 ) : C, 58.21; H, 4.78; N, 7.59; S, 17.60. Found): C, 58.21; H, 4.78; N, 7.59; S, 17.60.
実施例 4 - 23 Example 4-23
N- {5, 6 -ジメチル -8- {[2_(2-ナフチル)ェチル]スルファニル }-3-ォキソ_2,3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン) -4-メチ 実施例 4-14と同様の方法により、よう化ィソブチルの代わりにメタン スルホン酸 2- (2-ナフチル)ェチル エステル (206 mg, 0.865 mmol) を 用いて、 実施例 3-4で得られた N- (5, 6-ジメチル -3-ォキソ -8-スルファ ニル- 2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン -1-ィリデン)-4 - メチルベンゼンスルホンアミ ド, (300 mg, 0.786 mmol) から題記化合物 (253 mg, 60¾) を合成した。 酢酸ェチルから再結晶して融点 189- 191 の暗赤色結晶を得た。 N- {5,6-dimethyl-8-{[2_ (2-naphthyl) ethyl] sulfanyl} -3-oxo_2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -Trilydene) -4-methyl The procedure of Example 4-14 was repeated, except that 2- (2-naphthyl) ethyl methanesulfonate (206 mg, 0.865 mmol) was used instead of isobutyl iodide. N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene obtained in 3-4 The title compound (253 mg, 60¾) was synthesized from) -4-methylbenzenesulfonamide (300 mg, 0.786 mmol). Recrystallization from ethyl acetate gave dark red crystals with a melting point of 189-191.
赤外吸収スペクトル(IR) (KBr中) リ: 3220, 3052, 2921 , 1723, 1667, 1601, 1553 cm"1. Infrared absorption spectrum (IR) (in KBr): 3220, 3052, 2921, 1723, 1667, 1601, 1553 cm " 1 .
Ή-NMR (CDCI3) : δ 1.77 (3Η, s), 2.22 (3H, s), 2.37 (3H, s),Ή-NMR (CDCI3): δ 1.77 (3Η, s), 2.22 (3H, s), 2.37 (3H, s),
3.08-3.22 (2H, m), 3.22-3.37 (2H, m) , 7.13 (2H, d, J-7.8 Hz), 7.22-7.36 (2H, m) , 7.42-7.52 (2H, m) , 7.66 (1H, s), 7.73-7.88 (4H, m), 9.58 (1H, bs) . 3.08-3.22 (2H, m), 3.22-3.37 (2H, m), 7.13 (2H, d, J-7.8 Hz), 7.22-7.36 (2H, m), 7.42-7.52 (2H, m), 7.66 ( 1H, s), 7.73-7.88 (4H, m), 9.58 (1H, bs).
元素分析値: C27H25N303S3'0.8H20として 計算値 (%) : C, 58.95; H, 4.87; N, 7.64; S, 17.49. Elemental analysis: C 27 H 25 N 3 0 3 S 3 '0.8H 2 0 Calculated value (%): C, 58.95; H, 4.87; N, 7.64; S, 17.49.
実測値 ) : C, 58.93; H, 4.83; N, 7.32; S, 17.18. Found): C, 58.93; H, 4.83; N, 7.32; S, 17.18.
実施例 4 - 24 Example 4-24
N - [5, 6-ジメチル -3-ォキソ -8- (ペンチルスルファニル) - 2, 3-ジヒド口 - 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン] -4-メチルベンゼンス ルホンアミド N-[5,6-Dimethyl-3-oxo-8- (pentylsulfanyl)-2,3-dihydrido-1H-imidazo [5, 1-c] [1,4] thiazine-triylidene] -4- Methylbenzenesulfonamide
実施例 4-1'4 と同様の方法により、 よう化イソブチルの代わりに n -ァ ミル ブロミ ド (0.0907 ml, 0.721 mmol) を用いて、 実施例 3- 4で得ら れた N -(5, 6-ジメチル -3-ォキソ -8-スルファニル- 2, 3-ジヒドロ- 1H-ィ ミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン) -4-メチルベンゼンスルホン アミ ド (250 mg, 0.655 mmol) から題記化合物 (232 mg, 78%) を合成し た。酢酸ェチル -ジェチルエーテルから再結晶して融点 151- 153°Cの暗赤 色結晶を得た。 N- (5) obtained in Example 3-4 was obtained in the same manner as in Example 4-1'4, except that n-amyl bromide (0.0907 ml, 0.721 mmol) was used instead of isobutyl iodide. , 6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazin-1-ylidene) -4-methylbenzenesulfonamide ( The title compound (232 mg, 78%) was synthesized from 250 mg (0.655 mmol). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 151-153 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3250, 2957, 2926, 2863, 1748, 1717, 1661, 1611, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3250, 2957, 2926, 2863, 1748, 1717, 1661, 1611, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 0.910 (3Η, t, J = 7.0 Hz), 1.24-1.44 (4H, m), 1.64-1.76 (2H, ι) , 1.79 (3H, d, J=0.8 Hz), 2.26 (3H, d, J = l.0 Hz), 2. 2 (3H, s), 2.93 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.60 (1H, bs). ' Ή-NMR (CDC1 3): δ 0.910 (3Η, t, J = 7.0 Hz), 1.24-1.44 (4H, m), 1.64-1.76 (2H, ι), 1.79 (3H, d, J = 0.8 Hz) , 2.26 (3H, d, J = 1.0 Hz), 2.2 (3H, s), 2.93 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 ( 2H, d, J = 8.2 Hz), 9.60 (1H, bs).
実施例 4-25 Example 4-25
N-[5, 6-ジメチル- 3-ォキソ -8- (へプチルスルファニル)-2, 3-ジヒド口 - 1 H-イミダゾ [5, 1 -C] [1, 4]チアジン- 1 -イリデン] -4-メチルベンゼンス ルホンアミド N- [5,6-Dimethyl-3-oxo-8- (heptylsulfanyl) -2,3-dihydrido-1H-imidazo [5,1-C] [1,4] thiazine-1-ylidene] -4-methylbenzenesulfonamide
実施例 4-14 と同様の方法により、 よう化イソブチルの代わりに 1 -ブ ロモヘプタン (0.113 ml, 0.721 mmol) を用いて、 実施例 3 - 4で得られ た N- (5, 6-ジメチル -3-ォキソ -8-スルファニル -2,3-ジヒドロ- 1H-ィミ ダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン) -4-メチルベンゼンスルホンァ ミ ド (250 mg, 0.655腿 ol) から題記化合物 (257 mg, 82¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 156-158 の暗赤色 結晶を得た。 In the same manner as in Example 4-14, 1-bromoheptane (0.113 ml, 0.721 mmol) was used in place of isobutyl iodide, and the N- (5,6-dimethyl- 3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide (250 mg, The title compound (257 mg, 82¾) was synthesized from 0.655 t-ol). Recrystallization from ethyl acetate-getyl ether, dark red with melting point 156-158 Crystals were obtained.
赤外吸収スペクトル(IR) (KBr中) リ: 3233, 2955, 2928, 2857, 1748, 1717, 1663, 1613, 1557 cm—1. Infrared absorption spectrum (IR) (in KBr): 3233, 2955, 2928, 2857, 1748, 1717, 1663, 1613, 1557 cm- 1 .
Ή-NMR (CDC13) : δ 0.897 (3Η, t, J = 6.8 Hz), 1.22-1.49 (8H, m) , 1.62-1.74 (2H, m) , 1.79 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.93 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 0.897 (3Η, t, J = 6.8 Hz), 1.22-1.49 (8H, m), 1.62-1.74 (2H, m), 1.79 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.93 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.60 (1H, bs).
元素分析値: C22H29N303S3として Elementary analysis: as C 22 H 29 N 3 0 3 S 3
計算値 (%) : C, 55.09; H, 6.09; N, 8.76; S, 20.05. Calculated value (%): C, 55.09; H, 6.09; N, 8.76; S, 20.05.
実測値 ) : C, 54.91; H, 6.22; N, 8.68; S, 20.04. Obtained value): C, 54.91; H, 6.22; N, 8.68; S, 20.04.
実施例 -26 Example -26
N- [5, 6 -ジメチル- 8- (2-ィンダニルスルファ二ル)- 3-ォキソ -2, 3-ジヒド 口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン] -4-メチルベンゼン スルホンアミ ド N- [5,6-dimethyl-8- (2-indanylsulfanyl) -3-oxo-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine- Trilidene] -4-methylbenzene sulfonamide
実施例 3- で得られた N- (5, 6 -ジメチル- 3 -ォキソ -8-スルファニル -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) -4-メチ ルベンゼンスルホンアミ ド (250 mg, 0.655 mmol) と炭酸カリウム (136 ig, 0.983 mmol) の N, N-ジメチルホルムアミ ド (4.00 ml) の懸濁液に、 参考例 4で得られたメタンスルホン酸 インダン- 2 -ィル エステル (153 mg, 0.721 mmol) の N, N-ジメチルホルムアミ ド (1.00 ml) 溶液を加え た。反応混合物を 80°Cで 1時間加熱した後、 1N塩酸を加えて酢酸ェチル で抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して 題記化合物 (129 mg, 40¾) を暗赤色結晶として得た。 酢酸ェチル-ジェ チルエーテルから再結晶して融点 210- 212°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- obtained in Example 3- Reference Example 4 was prepared by adding a suspension of (ylidene) -4-methylbenzenesulfonamide (250 mg, 0.655 mmol) and potassium carbonate (136 ig, 0.983 mmol) in N, N-dimethylformamide (4.00 ml). A solution of N, N-dimethylformamide (1.00 ml) of indan-2-yl methanesulfonate (153 mg, 0.721 mmol) obtained in the above was added. After heating the reaction mixture at 80 ° C. for 1 hour, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (129 mg, 40¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 210-212 ° C.
赤外吸収スペクトル(IR) (KBr 中) レ : 3229, 1732, 1665, 1609, 1547 • cm一1. Infrared absorption spectrum (IR) (in KBr) Re: 3229, 1732, 1665, 1609, 1547 • cm- 1 .
Ή-NMR (CDC13) : δ 1.81 (3Η, d, J = l.0 Hz), 2.28 (3H, d, J = l.2 Hz), 2.39 (3H, s), 3.06 (2H, dd, J = 16.0, 6.0 Hz), 3.48 (2H, dd, J = 16.6, 7.8 Hz), 4.13-4.28 (1H, m) , 7.16—7.32 (,6H, m) , 7.78 (2H, d, J = 8.6 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 1.81 (3Η, d, J = l.0 Hz), 2.28 (3H, d, J = l.2 Hz), 2.39 (3H, s), 3.06 (2H, dd, J = 16.0, 6.0 Hz), 3.48 (2H, dd, J = 16.6, 7.8 Hz), 4.13-4.28 (1H, m), 7.16-7.32 (, 6H, m), 7.78 (2H, d, J = 8.6 Hz), 9.60 (1H, bs).
実施例 4-27 Example 4-27
N-(8-{[3, 5-ビス(卜リフルォロメチル)ベンジル]スルファニル }_5, 6-ジ メチル -3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1- c] [1,4]チアジン - 1- 実施例 4-14と同様の方法により、よう化イソブチルの代わりにメタン スルホン酸 3, 5-ビス(卜リフルォロメチル)ベンジル エステル(253 mg, 0.786匪 ol) を用いて、 実施例 3- 4で得られた N- (5, 6-ジメチル -3-ォキ ソ- 8-スルファニル -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン -卜ィ リデン) -4-メチルベンゼンスルホンア ミ ド (250 mg, 0.655 匪 ol) から題記化合物 (291 mg, 73%) を合成した。 酢酸エヂル-ジェチルエー テルから再結晶して融点 211- 213 の暗赤色結晶を得た。 N- (8-{[3,5-bis (trifluoromethyl) benzyl] sulfanyl} _5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] Thiazine-1-In the same manner as in Example 4-14, except that isobutyl iodide was replaced with 3,5-bis (trifluoromethyl) benzyl methanesulfonate (253 mg, 0.786 bandol). N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrido-1H-imidazo obtained in Example 3-4 [5,1-c] [1,4] thiazine The title compound (291 mg, 73%) was synthesized from 4-tribenzene) -4-methylbenzenesulfonamide (250 mg, 0.655 bandol) and recrystallized from ethyl acetate-ethyl ether to obtain a melting point of 211-213. Of dark red crystals were obtained.
赤外吸収スペクトル(IR) (KBr中) リ : 3260, 3206, 3061, 2930, 1732, 1665, 1624, 1564, 1497 cm—1. Infrared absorption spectrum (IR) (in KBr): 3260, 3206, 3061, 2930, 1732, 1665, 1624, 1564, 1497 cm- 1 .
^- MR (CDC13) : δ 1.76 (3Η, d, J = l.2 Hz), 2.24 (3H, d, J = l.2 Hz), 2. 2 (3H, s), 4.26 (2H, s), 7.27 (2H, d, J = 8.4 Hz), 7.79 (2H, s), 7.81 (2H, d, J = 8.2 Hz), 7.82 (1H, s) , 9.62 (1H, bs). ^-MR (CDC1 3 ): δ 1.76 (3Η, d, J = l.2 Hz), 2.24 (3H, d, J = l.2 Hz), 2.2 (3H, s), 4.26 (2H, s), 7.27 (2H, d, J = 8.4 Hz), 7.79 (2H, s), 7.81 (2H, d, J = 8.2 Hz), 7.82 (1H, s), 9.62 (1H, bs).
元素分析値: C24H19N303S3F6として Elemental analysis: C 24 H 19 N 3 0 3 S 3 F 6
計算値 (%) : C, 47. 4; H, 3.15; N, 6.92; S, 15.83. Calculated value (%): C, 47.4; H, 3.15; N, 6.92; S, 15.83.
実測値 (¾) : C, 47.55; H, 2.96; N, 6.96; S, 16.07. Found (¾): C, 47.55; H, 2.96; N, 6.96; S, 16.07.
実施例 4 - 28 Example 4-28
N-[8-({2-[3, 5-ビス(トリフルォロメチル)フエニル]ェチル }スルファ二 ル) - 5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4] チアジン-卜ィリデン ]-4-メチルベンゼンスルホンアミド N- [8-({2- [3,5-Bis (trifluoromethyl) phenyl] ethyl] sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [ 5, 1-c] [1,4] thiazine-triylidene] -4-methylbenzenesulfonamide
実施例 4- Uと同様の方法により、よう化ィソブチルの代わりにメタン スルホン酸 2-[3, 5-ビス(トリフルォロメチル)フエニル]ェチル エステ ル (242 mg, 0.721 mmol) を用いて、 実施例 3-4で得られた N -(5, 6 -ジ メチル- 3-ォキソ -8-スルフ ァニル -2, 3-ジヒ ド ロ -1H-イ ミ ダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン) -4-メチルベンゼンスルホンア ミ ド (250 fflg, 0.655 mmol) から題記化合物 (169 mg, 42¾) を合成した。 酢 酸ェチル -ジェチルエーテルから再結晶して融点 198_200°Cの暗赤色結 晶を得た。 . In the same manner as in Example 4-U, 2- [3,5-bis (trifluoromethyl) phenyl] ethyl methanesulfonate (242 mg, 0.721 mmol) was used instead of isobutyl iodide. N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo obtained in Example 3-4 [5,1-c] [1 , 4] thiazine-triylidene) -4-methylbenzenesulfonamide The title compound (169 mg, 42 mmol) was synthesized from (250 fflg, 0.655 mmol). Recrystallization from ethyl acetate-getyl ether gave a dark red crystal with a melting point of 198-200 ° C. .
赤外吸収スペクトル(IR) (KBr 中) リ: 3221, 1721, 1665, 1609, 1564 cm一 Infrared absorption spectrum (IR) (in KBr): 3221, 1721, 1665, 1609, 1564 cm
Ή-N R (CDC13) : δ 1.78 (3Η, s), 2.26 (3H, s), 2. 2 (3H, s), 3.05-3.16 (2H, m) , 3.16-3.27 (2H, m), 7.28 (2H, d, J = 8.0 Hz), 7.65 (2H, s), 7.79 (1H, s), 7.82 (2H, d, J = 8.4 Hz), 9.62 (1H, bs). 元素分析値: C25H21N303S3F6として Ή-NR (CDC1 3): δ 1.78 (3Η, s), 2.26 (3H, s), 2. 2 (3H, s), 3.05-3.16 (2H, m), 3.16-3.27 (2H, m), 7.28 (2H, d, J = 8.0 Hz), 7.65 (2H, s), 7.79 (1H, s), 7.82 (2H, d, J = 8.4 Hz), 9.62 (1H, bs). Elemental analysis: C 25 H 21 N 3 0 3 As S 3 F 6
計算値 (%) : C, 48.30; H, 3.40; N, 6.76; S, 15.47. Calculated value (%): C, 48.30; H, 3.40; N, 6.76; S, 15.47.
実測値 (¾) : C, 48.10; H, 3.35; N, 6.63; S, 15.89. Found (¾): C, 48.10; H, 3.35; N, 6.63; S, 15.89.
実施例 -29 Example -29
[(5,6-ジメチル- 1-{[(4-メチルフエニル)スルホニル]ィミノ }_3-ォキ ソ- 2, 3-ジヒドロ -1H-ィミダゾ [5, 1-c] [1, 4]チアジン -8-ィル)スルファ ニル]酢酸 メチルエステル [(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} _3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 8-yl) sulfanyl] acetic acid methyl ester
実施例 4-14と同様の方法により、よう化イソブチルの代わりにブロモ 酢酸メチル (0.0858 ml, 0.577 mmol) を用いて、 実施例 3 - 4で得られた N- (5, 6-ジメチル- 3 -ォキソ -8-スルファニル -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン)-4-メチルベンゼンスルホンアミ ド In the same manner as in Example 4-14, using methyl bromoacetate (0.0858 ml, 0.577 mmol) in place of isobutyl iodide, the N- (5,6-dimethyl-3) obtained in Example 3-4 was used. -Oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-triylidene) -4-methylbenzenesulfonamide
(200 mg, 0.524 mmol) から題記化合物 (155 mg, 65¾) を合成した。 酢 酸ェチル -ジェチルエーテルから再結晶して融点 192_194°Cの暗赤色結 晶を得た。 (200 mg, 0.524 mmol) to give the title compound (155 mg, 65¾). Recrystallization from ethyl acetate-getyl ether yielded dark red crystals with a melting point of 192_194 ° C.
赤外吸収スペクトル(IR) (KBr中) レ: 3177, 2957, 1757, 1740, 1663, 1632, 1570 cm'1. Infrared absorption spectrum (IR) (in KBr) L: 3177, 2957, 1757, 1740, 1663, 1632, 1570 cm ' 1 .
Ή-NMR (CDC13) : δ 1.78 (3Η, d, J = l.0 Hz), 2.25 (3H, d, J = l.2 Hz), 2.42 (3H, s), 3.73 (2H, s) , 3.78 (3H, s), 7.30 (2H, d, J-8.0 Hz), 7.85 (2H, d, J = 8. Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 1.78 (3Η, d, J = l.0 Hz), 2.25 (3H, d, J = l.2 Hz), 2.42 (3H, s), 3.73 (2H, s) , 3.78 (3H, s), 7.30 (2H, d, J-8.0 Hz), 7.85 (2H, d, J = 8. Hz), 9.60 (1H, bs).
元素分析値: C18H19N305S3として Elementary analysis: as C 18 H 19 N 3 0 5 S 3
計算値 (%) : C, 47.67; H, 4.22; N, 9.26; S, 21.21. 実測値 ) : C, 47.31; H, 4.02; N, 8.91; S, 20.87. Calculated value (%): C, 47.67; H, 4.22; N, 9.26; S, 21.21. Found): C, 47.31; H, 4.02; N, 8.91; S, 20.87.
実施例 4-30 Example 4-30
〔(5,6-ジメチル- 1-{[ (4-メチルフエニル)スルホニル]ィミノ卜 3-ォキ ソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル)スルファ ニル]酢酸 · [(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 8-yl) sulfanyl] acetic acid
実施例 4-29で得られた [(5, 6-ジメチル- 1- {[(4-メチルフエニル)スル ホニル]イミノ}-3-ォキソ - 2,3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チア ジン- 8-ィル)スルファニル]酢酸 メチルエステル (155 mg, 0.342 mmol) のメタノール (5.00 ml) 及びテトラヒドロフラン (5.00 ml) 溶液に、 1N 水酸化ナトリウム水溶液 (1.03 ml) を加えた。 反応混合物を室温で 30分かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水 洗、 乾燥(MgS04) 後、 溶媒を減圧下に留去して題記化合物 (136iag, 91%) を暗赤色結晶として得た。メタノ一ル-ジェチルエーテルから再結晶して 融点 198-200°Cの暗赤色結晶を得た。 [(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo obtained in Example 4-29 [5,1- c] To a solution of [1,4] thiazin-8-yl) sulfanyl] acetic acid methyl ester (155 mg, 0.342 mmol) in methanol (5.00 ml) and tetrahydrofuran (5.00 ml) was added a 1N aqueous sodium hydroxide solution (1.03 ml). ) Was added. After stirring the reaction mixture at room temperature for 30 minutes, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was water washed, dried (MgSO 4), and the solvent was evaporated under reduced pressure to give the title compound (136iag, 91%) as a dark red crystals. Recrystallization from methanol-getyl ether gave dark red crystals with a melting point of 198-200 ° C.
赤外吸収スペクトル(IR) (KBr 中) レ : 3700-2750, 1732, 1661, 1620, 1559 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3700-2750, 1732, 1661, 1620, 1559 cm " 1 .
Ή-NMR (CDCl3+D SO-d6) : δ 1.78 (3Η, s), 2.24 (3H, s), 2.42 (3H, s), 3.74 (2H, s), 7.30 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 10.18 (1H, bs). Ή-NMR (CDCl 3 + D SO-d 6 ): δ 1.78 (3Η, s), 2.24 (3H, s), 2.42 (3H, s), 3.74 (2H, s), 7.30 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 10.18 (1H, bs).
元素分析値: C17H17N305S3として Elementary analysis: as C 17 H 17 N 3 0 5 S 3
計算値 (¾) : C, 44.63; H, 4.19; N, 9.18; S, 21.02. Calculated (¾): C, 44.63; H, 4.19; N, 9.18; S, 21.02.
実測値 (¾) : C, 44.73; H, 4.1.2; N, 9.14; S, 20.74. Found (¾): C, 44.73; H, 4.1.2; N, 9.14; S, 20.74.
実施例 4 - 31 Example 4-31
N- [5, 6-ジメチル- 8- [(5, 5, 5-トリフルォロペンチル)スルファニル] - 3 - ォキソ -2, 3-ジヒ ド口- 1H-ィミダゾ [5,1- c] [1,4]チアジン-卜ィ リデ 実施例 4-14と同様の方法により、よう化ィソプチルの代わりにメタン スルホン酸 5,5,5-トリフルォロペンチルエステル (790 mg, 3.59 mmol) を用いて、 実施例 3-4で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルフ ァニル -2, 3-ジヒ ドロ -1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィ リデ ン)- 4-メチルベンゼンスルホンアミ ド (1.14 g, 2.99 mmol) から題記化 合物 (1.26 g, 84%) を合成した。 酢酸ェチル -ジェチルェ一テルから再 結晶して融点 177- 179°Cの暗赤色結晶を得た。 N- [5,6-Dimethyl-8-[(5,5,5-trifluoropentyl) sulfanyl] -3-oxo--2,3-dihydrido-1H-imidazo [5,1-c] [ 1,4] Thiazine trilide In the same manner as in Example 4-14, using methanesulfonic acid 5,5,5-trifluoropentyl ester (790 mg, 3.59 mmol) instead of isoptyl iodide. The N- (5,6-dimethyl-3-oxo-8-sulfone obtained in Example 3-4 Title compound from phenyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-trilidene) -4-methylbenzenesulfonamide (1.14 g, 2.99 mmol) Was synthesized (1.26 g, 84%). Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 177-179 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3245, 3069, 2946, 2878, 1748, 1732, 1663, 1622, 1557, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3245, 3069, 2946, 2878, 1748, 1732, 1663, 1622, 1557, 1497 cm " 1 .
Ή-NMR (CDC13) : δ 1.61-1.83 (4Η, m) , 1.79 (3H, d, J = l.2 Hz), 2.02-2.20 (2H, m) , 2.27 (3H, d, J = 0.6 Hz), 2.42 (3H, s), 2.95 (2H, t, J = 6.9 Hz), 7.29 (2H, dt, J = 8.7, 0.6 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, bs). Ή-NMR (CDC1 3): δ 1.61-1.83 (4Η, m), 1.79 (3H, d, J = l.2 Hz), 2.02-2.20 (2H, m), 2.27 (3H, d, J = 0.6 Hz), 2.42 (3H, s), 2.95 (2H, t, J = 6.9 Hz), 7.29 (2H, dt, J = 8.7, 0.6 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 ( 1H, bs).
元素分析値: C20H22N303S3F3として Elemental analysis: as C 20 H 22 N 3 0 3 S 3 F 3
計算値 (%) : C, 47.51; H, 4.39; N, 8.31; S, 19.03. Calculated value (%): C, 47.51; H, 4.39; N, 8.31; S, 19.03.
実測値 (« : C, 47. 3; H, 4.30; N, 8.41; S, 19.10. Actual values («: C, 47.3; H, 4.30; N, 8.41; S, 19.10.
実施例 4 - 32 Example 4-32
N-[5, 6-ジメチル -8- [(2, 2, 3, 3-テトラフルォロプロピル)スルファニ ル] - 3_ォキソ -2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリ 実施例 4-14 と同様の方法により、 よう化イソブチルの代わりに 3-ョ ード -1, 1, 2,2-テトラフルォロプロパン (76.1 mg, 0.315 mmol) を用い て、 実施例 3- 4で得られた N_(5, 6-ジメチル -3-ォキソ -8-スルファニル -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン) -4-メチ ルベンゼンスルホンアミド (100 mg, 0.262 mmol) から題記化合物 (51.6 mg, 40¾) を合成した。 酢酸ェチル -ジイソプロピルエーテルから再結晶 して融点 182- 184°Cの暗赤色結晶を得た。 N- [5,6-dimethyl-8-[(2,2,3,3-tetrafluoropropyl) sulfanyl] -3_oxo-2,3-dihydro-1H-imidazo [5,1-c [1,4] Thiazine-trily In the same manner as in Example 4-14, 3-ode-1,1,2,2-tetrafluoropropane (76.1 mg) was used instead of isobutyl iodide. , 0.315 mmol) to obtain N_ (5,6-dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] obtained in Example 3-4. The title compound (51.6 mg, 40%) was synthesized from 1,4] thiazine-triylidene) -4-methylbenzenesulfonamide (100 mg, 0.262 mmol). Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals having a melting point of 182-184 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3221, 2990, 2930, 1748, 1667, 1626, 168 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3221, 2990, 2930, 1748, 1667, 1626, 168 cm " 1 .
Ή-NMR (CDC13) : δ 1.80 (3Η, d, J = 1.2 Hz), 2.25 (3H, d, J = l.2 Hz), 2.43 (3H, s), 3.49 (2H, t, J = 15.6 Hz), 5.84 (1H, tt, J = 53.4, 2.7 Hz), 7.31 (2H, d, J = 7.8 Hz), 7.85 (2H, d, J = 8. Hz), 9.63 (1H, bs) . 元素分析値: C18H17N303S3F4として Ή-NMR (CDC1 3): δ 1.80 (3Η, d, J = 1.2 Hz), 2.25 (3H, d, J = l.2 Hz), 2.43 (3H, s), 3.49 (2H, t, J = 15.6 Hz), 5.84 (1H, tt, J = 53.4, 2.7 Hz), 7.31 (2H, d, J = 7.8 Hz), 7.85 (2H, d, J = 8. Hz), 9.63 (1H, bs). Elementary analysis: as C 18 H 17 N 3 0 3 S 3 F 4
計算値 (%) : C, 43.63; H, 3.46; N, 8.48; S, 19.41. Calculated value (%): C, 43.63; H, 3.46; N, 8.48; S, 19.41.
実測値 (%) : C, 43.56; H, 3.39; N, 8.33; S, 19.22. Found (%): C, 43.56; H, 3.39; N, 8.33; S, 19.22.
実施例 4 - 33 Example 4-33
N - [5, 6 -ジメチル- 8-[(4-フルォロブチル)スルファニル ]-3-ォキソ -2, 3- ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン] -4-メチルベ ンゼンスルホンアミ ド N- [5,6-Dimethyl-8-[(4-fluorobutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene] -4-methylbenzenesulfonamide
実施例 4-14 と同様の方法により、 よう化イソプチルの代わりに 1 -ブ ロモ- 4-フルォロブタン (149 mg, 0.944 mmol) を用いて、 実施例 3 - 4 で得られた N -(5, 6-ジメチル -3-ォキソ -8-スルファニル- 2, 3-ジヒド口 - 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) -4-メチルベンゼンス ルホンアミド (300 mg, 0.786 mmol) から題記化合物 (211 mg, 59¾) を 合成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 156- 158で の暗赤色結晶を得た。 According to a method similar to that of Example 4-14, 1-bromo-4-fluorobutane (149 mg, 0.944 mmol) was used in place of isoptyl iodide, and N- (5,5) obtained in Example 3-4 was used. 6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydric port-1H-imidazo [5, 1-c] [1, 4] thiazine-1-ylidene) -4-methylbenzenesulfonamide (300 mg) , 0.786 mmol) to give the title compound (211 mg, 59%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with melting point 156-158.
赤外吸収スペクトル(IR) (KBr中) : 3227, 3067, 2965, 2922, 1748, 1725, 1663, 1615, 1563 cm"1. Infrared absorption spectrum (IR) (in KBr): 3227, 3067, 2965, 2922, 1748, 1725, 1663, 1615, 1563 cm " 1 .
lH - NMR (CDC13) : δ 1.70-1.90 (4Η, ) , 1. 9 (3Η, d, J = l.2 Hz), 2.26 (3H, d, J = 0.9 Hz), 2. 2 (3H, s)., 2.99 (2H, t, J = 7.2 Hz), 4.47 (2H, dt, J=47.1, 5.7 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.7 Hz), 9.62 (1H, bs). lH - NMR (CDC1 3): δ 1.70-1.90 (4Η,), 1. 9 (3Η, d, J = l.2 Hz), 2.26 (3H, d, J = 0.9 Hz), 2. 2 (3H , s)., 2.99 (2H, t, J = 7.2 Hz), 4.47 (2H, dt, J = 47.1, 5.7 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.7 Hz), 9.62 (1H, bs).
元素分析値: C19H22N303S3Fとして Elemental analysis: as C 19 H 22 N 3 0 3 S 3 F
計算値 ( : C, 50.09; H, 4.87; N, 9.22; S, 21.11. Calculated value (: C, 50.09; H, 4.87; N, 9.22; S, 21.11.
実測値 ) : C, 49.89; Η,.4.77; Ν, 9.21; S, 21.24. Obtained value): C, 49.89; Η, 4.77; Ν, 9.21; S, 21.24.
実施例 4 - 34 Example 4-34
N-{5, 6-ジメチル- 3-ォキソ -8- [(4, 4, 5, 5, 5 -ペンタフルォロペンチル)ス ルファニル] -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリ 実施例 3- で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルファニル -2,3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン) -4-メチ ルベンゼンスルホンアミ ド (29.8 g, 78.1 mmol) と炭酸カリウム (16.2 g, 0.117 mol) の N,N-ジメチルホルムアミド (400 ml) の懸濁液に、 参 考例 5 で得られたメタンスルホン酸 4,4,5,5,5-ペンタフルォロペンチ ル エステル(24.0 g, 93.7 mmo 1 ) の N, N-ジメチルホルムアミ ド (100 ml) 溶液を加えた。反応混合物を 80°Cで 2時間加熱した後、 1N塩酸を加えて 酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去して題記化合物 (41.5 g, 98%) を暗赤色結晶として得た。 酢酸ェ チル-ジェチルェ一テルから再結晶して融点 190_192°Cの暗赤色結晶を 得た。 N- {5,6-dimethyl-3-oxo-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1 -c] [1,4] thiazine-trily N- (5,6-dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo obtained in Example 3- [5, 1-c] [1,4] thiazine-1-ylidene) -4-methyl Methanesulfonic acid obtained in Reference Example 5 was added to a suspension of benzenesulfonic acid amide (29.8 g, 78.1 mmol) and potassium carbonate (16.2 g, 0.117 mol) in N, N-dimethylformamide (400 ml). A solution of 4,4,5,5,5-pentafluoropentyl ester (24.0 g, 93.7 mmol) in N, N-dimethylformamide (100 ml) was added. After heating the reaction mixture at 80 ° C. for 2 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (41.5 g, 98%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 190-192 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3208, 2934, 1753, 1663, 1624, 1566, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr): 3208, 2934, 1753, 1663, 1624, 1566, 1497 cm " 1 .
iH-NMR (CDC13) : δ 1.79 (3Η, d, J = l.0 Hz), 1.90-2.22 (4H, m) , 2.27 (3H, d, J = l.2 Hz), 2.42 (3H, s), 3.01 (2H, t, J = 6.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs). iH-NMR (CDC1 3): δ 1.79 (3Η, d, J = l.0 Hz), 1.90-2.22 (4H, m), 2.27 (3H, d, J = l.2 Hz), 2.42 (3H, s), 3.01 (2H, t, J = 6.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C2。H2。N303S3F5として Elemental analysis: C 2. H 2. N 3 0 3 S 3 F 5
計算値 (¾) : C, 44.35; H, 3.72; N, 7.76; S, 17.76. Calculated value (¾): C, 44.35; H, 3.72; N, 7.76; S, 17.76.
実測値 ) : C, 44.32; H, 3.69; N, 7. 4; S, 17.67. Found): C, 44.32; H, 3.69; N, 7.4; S, 17.67.
実施例 4 - 35 Example 4-35
N-{5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)ス ルファニル ]-2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリ 実施例 3-4 で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルファニル -2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン) -4-メチ ルベンゼンスルホンアミド (2.00 g, 5.24 mmol) と炭酸カリウム (1.09 g, 7.86 mmol) の N, N-ジメチルホルムアミ ド (30.0 ml) の懸濁液に、 参考例 9 で得られたメタンスルホン酸 5, 5, 6, 6, 6-ペンタフルォ口へキ シル エステル (1.70 g, 6.29 mmol) の N, N-ジメチルホルムアミ ド (10.0 ml) 溶液を加えた。 反応混合物を 80 で 1時間加熱した後、 1N塩酸を加 えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (2.38g, 82¾) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルエーテルから再結晶して融点 173- 175°Cの暗赤色結 晶を得た。 N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-trily N- (5,6-dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo obtained in Example 3-4 [ 5, 1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide (2.00 g, 5.24 mmol) and potassium carbonate (1.09 g, 7.86 mmol) in N, N-dimethylformamide Of methanesulfonic acid 5,5,6,6,6-pentafluorohexyl ester (1.70 g, 6.29 mmol) obtained in Reference Example 9 in N, N-dimethyl suspension (30.0 ml). A solution of formamide (10.0 ml) was added. After heating the reaction mixture at 80 for 1 hour, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after reduction the solvent Evaporation under pressure gave the title compound (2.38 g, 82 °) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 173-175 ° C.
赤外吸収スペク トル(IR) (KBr中) リ : 3202, 3054, 2924, 2855, 1732, 1717, 1661, 1615, 1559, 1497 cm'1. Infrared absorption spectrum (IR) (in KBr): 3202, 3054, 2924, 2855, 1732, 1717, 1661, 1615, 1559, 1497 cm ' 1 .
Ή-NMR (CDC13) : δ 1.64-1.84 (4Η , m) , 1.79 (3H, d, J=0.6 Hz), 1.90-2.20 (2H, m), 2.27 (3H, s) , 2. 2 (3H, s), 2.97 (2H, t, J = 7.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.6 Hz), 9.63 (1H, bs). 元素分析値: C21H22N303S3F5として Ή-NMR (CDC1 3): δ 1.64-1.84 (4Η, m), 1.79 (3H, d, J = 0.6 Hz), 1.90-2.20 (2H, m), 2.27 (3H, s), 2. 2 ( 3H, s), 2.97 (2H, t, J = 7.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.6 Hz), 9.63 (1H, bs). Elemental analysis Value: as C 21 H 22 N 3 0 3 S 3 F 5
計算値 (%) : C, 45.40; H, 3.99; N, 7.56; S, 17.31. Calculated value (%): C, 45.40; H, 3.99; N, 7.56; S, 17.31.
実測値 ( ) : C, 45.30; Η, 3.87; Ν, 7.42; S, 17.29. Observed (): C, 45.30; Η, 3.87; Ν, 7.42; S, 17.29.
実施例 4-36 Example 4-36
ェチル 6-[(5, 6-ジメチル- 1-{[(4-メチルフエニル)スルホニル]イミ ノ卜 3 -ォキソ -2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィ ル)スルファニル]へキサノエ一ト Ethyl 6-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] iminot 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-8-yl) sulfanyl] hexanoate
実施例 4-14と同様の方法により、よう化ィソブチルの代わりにェチル 6-ブロモへキサノエート (527 ml, 2.88 mmol) を用いて、 実施例 3-4 で得られた N- (5, 6-ジメチル -3-ォキソ -8-スルファニル -2, 3-ジヒドロ -1H -ィミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン) -4-メチルベンゼンス ルホンアミ ド (1.00 g, 2.62 mmol) から題記化合物 (1.19 g, 87¾) を 合成した。酢酸ェチル-ジェチルェ一テルから再結晶して融点 122-124 の暗赤色結晶を得た。 In the same manner as in Example 4-14, using ethyl 6-bromohexanoate (527 ml, 2.88 mmol) instead of isobutyl iodide, the N- (5,6- Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide (1.00 g, 2.62 mmol) to give the title compound (1.19 g, 87¾). Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 122-124.
赤外吸収スペクトル(IR) (KBr中) レ : 3304, 3137, 2980, 2934, 1744, 1728, 1661, 1615, 1557, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3304, 3137, 2980, 2934, 1744, 1728, 1661, 1615, 1557, 1497 cm " 1 .
Ή-NMR (CDCI3) : δ 1.26 (3H, t, J=7.0 Hz), 1.34-1.52 (2H, in), 1.52-1.76 (4H, m) , 1.73 (3H, s), 2.27 (3H, s) , 2.31 (2H, t, J = 7.0 Hz), 2.42 (3H, s), 2.93 (2H, t, J = 7.2 Hz), 4.13 (2H, q, J = 7.0 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8. Hz), 9.61 (1H, bs). 元素分析値: C23H29N305S3として 計算値 ) : C, 52.75; H, 5.58; N, 8.02; S, 18.37. Ή-NMR (CDCI3): δ 1.26 (3H, t, J = 7.0 Hz), 1.34-1.52 (2H, in), 1.52-1.76 (4H, m), 1.73 (3H, s), 2.27 (3H, s ), 2.31 (2H, t, J = 7.0 Hz), 2.42 (3H, s), 2.93 (2H, t, J = 7.2 Hz), 4.13 (2H, q, J = 7.0 Hz), 7.29 (2H, d , J = 8.4 Hz), 7.85 (2H, d, J = 8. Hz), 9.61 (1H, bs). Elemental analysis: As C 23 H 29 N 3 0 5 S 3 Calculated values): C, 52.75; H, 5.58; N, 8.02; S, 18.37.
実測値 、%) : C, 52.66; H, 5.32; N, 7.95; S, 18.28. Found,%): C, 52.66; H, 5.32; N, 7.95; S, 18.28.
実施例 4-37 Example 4-37
6_[ (5, 6-ジメチル- 1- (4-メチルフエニル)スルホニル]イミノ}- 3-ォキ ソ- 2, 3-ジヒドロ- 1H -ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファ ニル]へキサン酸 6 _ [(5,6-Dimethyl-1- (4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 8-yl) sulfanyl] hexanoic acid
実施例 4-36で得られたェチル 6- [(5, 6 -ジメチル- 1- {[(4 メチルフエ ニル)スルホニル]ィ ミ ノ卜 3-ォキソ -2, 3-ジヒ ド口 - 1H -ィ ミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィル)スルファニル]へキサノエ一ト (1.00 g, 1.91 imol) をテトラヒドロフラン (10.0 ml) とエタノール (10.0 ml) の混合物に溶かし、 1N水酸化ナトリウム水溶液 (5.73 ml) を加えて 2.5 時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(822 mg, 87¾) を得た。 メタノール-ジェチルエーテルから再結晶して融点 153-155°Cの暗赤色結晶を得た。 Ethyl 6-[(5,6-Dimethyl-1-{[(4 methylphenyl) sulfonyl] imino obtained in Example 4-36 3-oxo-2,3-dihydrogen port-1H Midazo [5,1-c] [1,4] thiazine-8-yl) sulfanyl] hexanoate (1.00 g, 1.91 imol) was dissolved in a mixture of tetrahydrofuran (10.0 ml) and ethanol (10.0 ml), A 1N aqueous sodium hydroxide solution (5.73 ml) was added and the mixture was stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain the solvent was distilled off under reduced pressure the title compound (822 mg, 87¾). Recrystallization from methanol-getyl ether gave dark red crystals with a melting point of 153-155 ° C.
赤外吸収スペクトル(IR) ( Br 中) リ: 3700-2760, 2934, 2861, 1728, 1661 , 1615, 1557, 1495 cm"1. Infrared absorption spectrum (IR) (in Br): 3700-2760, 2934, 2861, 1728, 1661, 1615, 1557, 1495 cm " 1 .
Ή-NMR (CDC13) : δ 1.38-1.82 (6Η, m) , 1.79 (3H, d, J = 1.2 Hz), 2.26 (3H, d, J = l.2 Hz), 2.38 (2H, t, J = 7.2 Hz), 2.42 (3H, s), 2.95 (2H, t, J = 7.0 Hz), 7.31 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.65 (1H, bs). Ή-NMR (CDC1 3): δ 1.38-1.82 (6Η, m), 1.79 (3H, d, J = 1.2 Hz), 2.26 (3H, d, J = l.2 Hz), 2.38 (2H, t, J = 7.2 Hz), 2.42 (3H, s), 2.95 (2H, t, J = 7.0 Hz), 7.31 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.65 (1H, bs).
元素分析値: C21H25N305S3として Elemental analysis: as C 21 H 25 N 3 0 5 S 3
計算値 、%) : C, 50.89; H, 5.08; N, 8. 8; S, 19.41. (Calculated,%): C, 50.89; H, 5.08; N, 8.8; S, 19.41.
実測値 ) : C, 50.82; H, 5.14; N, 8.22; S, 19.32. Found): C, 50.82; H, 5.14; N, 8.22; S, 19.32.
実施例 4-38 Example 4-38
N - [8- [(5 -クロロペンチル)スルファニル ]-5, 6-ジメチル- 3-ォキソ - 2, 3- ジヒドロ- 1H-ィミダゾ [5,1- c] [1, 4]チアジン -1-ィリデン ]-4-メチルベ ンゼンスルホンアミ ド N- [8-[(5-chloropentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene] -4-methylbenzenesulfonamide
実施例 4-14 と同様の方法により、 よう化イソブチルの代わりに 1 -ブ ロモ- 5-クロ口ペンタン (0.380 ml, 2.88 mmol) を用いて、 実施例 3 - 4 で得られた N -(5, 6-ジメチル- 3-ォキソ _8-スルファニル -2, 3-ジヒドロ -1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) -4-メチルベンゼンス ルホンアミ ド (1.00 g, 2.62 mmol) から題記化合物 (1.18 g, 92¾) を 合成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 167_169Τ の暗赤色結晶を得た。 In the same manner as in Example 4-14, 1-butyl was used in place of isobutyl iodide. N- (5,6-Dimethyl-3-oxo_8-sulfanyl-2,3-dihydro-1H obtained in Example 3-4 was obtained using lomo-5-chloromouth pentane (0.380 ml, 2.88 mmol). The title compound (1.18 g, 92¾) was synthesized from -imidazo [5,1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide (1.00 g, 2.62 mmol). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 167-169 °.
赤外吸収スペクトル(IR) (KBr中) リ: 3250, 3065, 2928, 2867, 1746, 1728, 1661, 1615, 1557, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr): 3250, 3065, 2928, 2867, 1746, 1728, 1661, 1615, 1557, 1497 cm " 1 .
Ή-NMR (CDC13) : δ 1.50-1.88 (6Η, m) , 1.79 (3H, s), 2.26 (3H, s) , 2. 2 (3H, s), 2.95 (2H, t, J = 7.4 Hz), 3.54 (2H, t, J = 6.6 Hz), 7.31 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3): δ 1.50-1.88 (6Η, m), 1.79 (3H, s), 2.26 (3H, s), 2. 2 (3H, s), 2.95 (2H, t, J = 7.4 Hz), 3.54 (2H, t, J = 6.6 Hz), 7.31 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.62 (1H, bs).
元素分析値: C2。H24N305S3として Elemental analysis: C 2. H 24 N 3 0 5 S 3
計算値 (%) : C, 49.42; H, 4.98; N, 8.64; S, 19.79. Calculated value (%): C, 49.42; H, 4.98; N, 8.64; S, 19.79.
実測値 (%) : C, 49.07; H, 4.95; N, 8.46; S, 19.68. Found (%): C, 49.07; H, 4.95; N, 8.46; S, 19.68.
実施例 4 - 39 Example 4-39
N-[5, 6-ジメチル- 8_[(2, 2, 3, 3, 3-ペン夕フルォロプロピル)スルファニ ル]— 3-ォキソ -2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリ 実施例 4- 14 と同様の方法により、 よう化イソブチルの代わりに 1 -ョ —ド- 2, 2, 3, 3, 3-ペンタフルォロプロパン (0.238 ml, 1.26 mmol) を用 いて、 実施例 3-4で得られた -(5, 6-ジメチル- 3-ォキソ -8-スルファニ ル- 2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) -4 -メ チルベンゼンスルホンアミド (400 mg, 1.05 mmol) から題記化合物(56.5 mg, 11%) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融 点 176- 178°Cの暗赤色結晶を得た。 N- [5,6-dimethyl-8 _ [(2,2,3,3,3-pentafluorofluoropropyl) sulfanyl] —3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-yl In the same manner as in Example 4-14, 1-iodo-2,2,3,3,3-pentafluoropropane (instead of isobutyl iodide) Using 0.238 ml, 1.26 mmol) of-(5,6-dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo obtained in Example 3-4 [5,1- c] The title compound (56.5 mg, 11%) was synthesized from [1,4] thiazine-1-ylidene) -4-methylbutylsulfonamide (400 mg, 1.05 mmol). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 176-178 ° C.
赤外吸収スペクトル(IR) (KBr中) レ: 3240, 2990, 2936, 1748, 1667, 1626, 1574, 1495 cm—1. Infrared absorption spectrum (IR) (in KBr) d: 3240, 2990, 2936, 1748, 1667, 1626, 1574, 1495 cm— 1 .
Ή-NMR (CDCI3) : δ 1.81 (3Η, d, J = l.2 Hz), 2.25 (3H, d, J = l.2 Hz), 2.43 (3H, s), 3.53 (2H, t, J = 16.2 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J=8.4 Hz), 9.63 (1H, bs). Ή-NMR (CDCI3): δ 1.81 (3Η, d, J = 1.2 Hz), 2.25 (3H, d, J = 1.2 Hz), 2.43 (3H, s), 3.53 (2H, t, J) = 16.2 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, bs).
実施例 4-40 Example 4-40
N-[5, 6-ジメチル- 8- [(3-フルォロ -3_メチルプチル)スルファニル ]-3-ォ キソ- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン] - 4 - 実施例 4-14と同様の方法により、よう化ィソブチルの代わりにメタン スルホン酸 3-フルォ口- 3-メチルブタン エステル (380 mg, 2.06 nimol) を用いて、 実施例 3-4で得られた N- (5, 6-ジメチル -3-ォキソ -8-スルフ ァニル _2, 3 -ジヒ ド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィ リデ ン)- 4-メチルベンゼンスルホンアミ ド (393 mg, 1.03 mmol) から題記化 合物 (111 mg, 23¾) を合成した。 酢酸ェチル -ジェチルエーテルから再 結晶して融点 173-175t:の暗赤色結晶を得た。 N- [5, 6-dimethyl-8-[(3-fluoro-3_methylbutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1, 4] Thiazine-triylidene]-4-In the same manner as in Example 4-14, using methanesulfonic acid 3-fluoro-3-methylbutane ester (380 mg, 2.06 nimol) instead of isobutyl iodide. N- (5,6-Dimethyl-3-oxo-8-sulfanyl_2,3-dihydrogen-1H-imidazo obtained in 3-4 [5,1-c] [1,4] thiazine The title compound (111 mg, 23¾) was synthesized from (ylidene) -4-methylbenzenesulfonamide (393 mg, 1.03 mmol). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 173-175t :.
赤外吸収スペクトル(IR) (KBr中) リ: 3241, 2976, 2934, 1752, 1717, 1659, 1613, 1561, 1497 cm—1. Infrared absorption spectrum (IR) (in KBr): 3241, 2976, 2934, 1752, 1717, 1659, 1613, 1561, 1497 cm- 1 .
Ή-NMR (CDC13) δ 1.39 (6Η, d, J = 21.2 Hz), 1.79 (3H, s) , 1.87-2.08 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 3.04 (2H, t, J = 8.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3) δ 1.39 (6Η, d, J = 21.2 Hz), 1.79 (3H, s), 1.87-2.08 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 3.04 (2H, t, J = 8.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C2。H24N303S3Fとして Elemental analysis: C 2. H 24 N 3 0 3 As S 3 F
計算値 (%) : C, 51.15; H, 5.15; N, 8.95; S, 20.48. Calculated value (%): C, 51.15; H, 5.15; N, 8.95; S, 20.48.
実測値 (%) : C, 51.40; H, 5.02; N, 8.98; S, 20.34. Found (%): C, 51.40; H, 5.02; N, 8.98; S, 20.34.
実施例 4 - 41 Example 4-41
N - [5, 6 -ジメチル -8- [(4, 4, 4 -トリフルォロブチル)スルファニル ]-3 -才 キソ- 2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン] - 4 - 実施例 4- 14 と同様の方法により、 よう化イソブチルの代わりに 4-ブ ロモ- 1, 1, 1-トリフルォロブタン (123 mg, 0.629 mmol) を用いて、 実施 例 3-4で得られた N-(5, 6-ジメチル -3-ォキソ -8-スルファニル -2, 3-ジヒ ドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン) _4_メチルベンゼ ンスルホンアミド ( 200 mg, 0.524 mmol) から題記化合物 (172 mg, 67¾) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 178-180°Cの暗赤色結晶を得た。 N- [5,6-dimethyl-8-[(4,4,4-trifluorobutyl) sulfanyl] -3-year-old oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-triylidene]-4-In the same manner as in Example 4-14, 4-bromo-1,1,1-trifluorotrifluorobutane (123 mg, 0.629 mmol) was used instead of isobutyl iodide. The N- (5,6-dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, 1-c] [1, 4] Thiazine-triylidene) _4_Methylbenzenesulfonamide (200 mg , 0.524 mmol) from the title compound (172 mg, 67¾) Was synthesized. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 178-180 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3225, 2932, 1755, 1725, 1663, 1624, 1566 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3225, 2932, 1755, 1725, 1663, 1624, 1566 cm " 1 .
醒 R (CDC13) : δ 1.79 (3H, d, J = l.2 Hz), 1.90-2.02 (2H, m) , 2.11-2.26 (2H, m) , 2.27 (3H, d, J = 0.9 Hz), 2.42 (3H, s), 3.00 (2H, t, J = 7.5 Hz), 7.29 (2H, d, J = 7.8 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.62 (1H, bs). Awakening: R (CDC1 3): δ 1.79 (3H, d, J = l.2 Hz), 1.90-2.02 (2H, m), 2.11-2.26 (2H, m), 2.27 (3H, d, J = 0.9 Hz ), 2.42 (3H, s), 3.00 (2H, t, J = 7.5 Hz), 7.29 (2H, d, J = 7.8 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.62 (1H, bs ).
元素分析値: C19H2。N303S3F3として . Elemental analysis: C 19 H 2. As N 3 0 3 S 3 F 3 .
計算値 ) : C, 46.42; H, 4.10; N, 8.55; S, 19.57. Calculated)): C, 46.42; H, 4.10; N, 8.55; S, 19.57.
実測値 ( ) : C, 46.56; H, 4.31; N, 8.70; S, 19.41. Found (): C, 46.56; H, 4.31; N, 8.70; S, 19.41.
実施例 4-42 Example 4-42
N - [1, 6-ジォキソ- 4- (ィソペンチルスルファ二ル)- 1, 2, 6, 1, 8, 9-へキサ ヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4-メチ 実施例 3-38 で得 られた N- (1 , 6-ジォキソ -4-スルフ ァ ニル -1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン -3 -イリデン) -4-メチルベンゼンスルホンアミド (2.00 g, 4.74 mmol) と炭酸力リゥム(984 mg, 7.12 mmol)の N, N-ジメチルホルムアミ ド(40.0 ml) の懸濁液に、 よう化イソアミル (0.691 ml, 5.22 mmol) の Ν,Ν -ジ メチルホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を室温で 19 時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水 洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (2.19 g, 94%) を暗赤色結晶として得た。 酢酸ェチル -メタノール-テトラヒドロフラン から再結晶して融点 234- 236°Cの暗赤色結晶を得た。 N- [1,6-Dioxo-4- (isopentylsulfanyl) -1, 2,6,1,8,9-hexahydro-3H-imidazo [5,1-c] [1, 4] Benzothiazine-3-ylidene] -4-methyl N- (1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro obtained in Example 3-38 -3H-imidazo [5, 1-c] [1, 4] benzothiazine-3-ylidene) -4-methylbenzenesulfonamide (2.00 g, 4.74 mmol) and carbon dioxide (984 mg, 7.12 mmol) N, To a suspension of N-dimethylformamide (40.0 ml) was added a solution of isoamyl iodide (0.691 ml, 5.22 mmol) in Ν, Ν-dimethylformamide (1.00 ml). After stirring the reaction mixture at room temperature for 19 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was water washed, dried (MgSO 4), and the solvent was evaporated under reduced pressure to give the title compound (2.19 g, 94%) as a dark red crystals. Recrystallization from ethyl acetate-methanol-tetrahydrofuran yielded dark red crystals with a melting point of 234-236 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3183, 2957, 2928, 1872, 1742, 1653, 1611, 1586, 1549, 1495 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3183, 2957, 2928, 1872, 1742, 1653, 1611, 1586, 1549, 1495 cm " 1 .
Ή-NMR (CDC13) : (5 0.934 (6H, d, J = 6.2 Hz), 1.50-1.63 (2H, m) , 1.63-1.78 (1H, m), 1.96-2.12 (2H, m) , 2.43 (3H, s), 2.50 (2H, t, J = 7.0 Hz), 3.04 (2H, t, J = 7.6 Hz), 3.21 (2H, t, J = 6.2 Hz), 7.29 (2H, d, J = 8.8 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.85 (1H, bs). Ή-NMR (CDC1 3): (5 0.934 (6H, d, J = 6.2 Hz), 1.50-1.63 (2H, m), 1.63-1.78 (1H, m), 1.96-2.12 (2H, m), 2.43 (3H, s), 2.50 (2H, t, J = 7.0 Hz), 3.04 (2H, t, J = 7.6 Hz), 3.21 (2H, t, J = 6.2 Hz), 7.29 (2H, d, J = 8.8 Hz), 7.84 (2H, d, J = 8.4 Hz) , 9.85 (1H, bs).
元素分析値: C22H25N304S3として Elementary analysis: as C 22 H 25 N 3 0 4 S 3
計算値 (%) : C, 53.74; H, 5.13; N, 8.55; S, 19: 57. Calculated value (%): C, 53.74; H, 5.13; N, 8.55; S, 19: 57.
実測値 ) : C, 53.68; H, 5.08; N, 8.27; S, 19.31. Found): C, 53.68; H, 5.08; N, 8.27; S, 19.31.
実施例 4-43 Example 4-43
N - [6 -ヒ ド ロ キ シ - 4 -(イ ソ ペ ンチルス レフ ァ ニル ) -トォキソ -1,2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン 水素化リチウムアルミニウム (281 mg, 5.92 mmol) のテトラヒドロフ ラン (10.0 ml) の懸濁液に、 実施例 4- 42で得られた N_[l, 6-ジォキソ - 4 -(ィソペンチルスルファ二ル)- 1, 2, 6, 7, 8, 9-へキサヒドロ- 3H -ィミダ ゾ [5 ,卜 c] [1, 4]ベンゾチアジン- 3-イリデン] -4-メチルベンゼンスルホ ンアミ ド (1.94 g, 3.95 匪 oU のテトラヒドロフラン (90.0 ml) 溶液 を氷冷下加えた。 反応混合物を室温で 5分かき混ぜた後、 過剰の水素化 リチウムアルミニウムを水で分解し、 1N塩酸を加えて酢酸ェチルで抽出 した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留 物をシリカゲル力ラムクロマトグラフィ一に付し、 n -へキサン-酢酸ェチ レ (10:1, v/v) で溶出して題記化合物 (1.18 g, 61¾) を暗赤色結晶と して得た。酢酸ェチル-ジェチルエーテルから再結晶して融点 162- 164°C の暗赤色結晶を得た。 N- [6-Hydroxy-4- (isopentyls phenyl) -toxo-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine A suspension of lithium aluminum hydride (281 mg, 5.92 mmol) in tetrahydrofuran (10.0 ml) was added to the N_ [l, 6-dioxo obtained in Example 4-42. (Isopentylsulfanyl) -1,2,6,7,8,9-hexahydro-3H-imidazo [5, tric] [1,4] benzothiazine-3-ylidene] -4-methyl A solution of benzenesulfonamide (1.94 g, 3.95 marudium oU in tetrahydrofuran (90.0 ml) was added under ice cooling. The reaction mixture was stirred at room temperature for 5 minutes, and excess lithium aluminum hydride was decomposed with water and 1N hydrochloric acid was added. was extracted with acetic acid Echiru added. the extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. the residue was silica gel force ram chromatograph The title compound (1.18 g, 61¾) was obtained as dark red crystals by elution with n-hexane-ethyl acetate (10: 1, v / v). Recrystallization from tyl ether gave dark red crystals with a melting point of 162-164 ° C.
赤外吸収スペクトル(IR) (KBr 中) レ : 3700-3000, 2955, 2870, 1732, 1655, 1615, 1557, 1495 cm—1. Infrared absorption spectrum (IR) (in KBr) Re: 3700-3000, 2955, 2870, 1732, 1655, 1615, 1557, 1495 cm- 1 .
Ή-N R (CDC13) : δ 0.928 (6H, d, J = 6.2 Hz), 1.44-2.02 (7H, m) , 2.42 (3H, s), 2.48-2.66 (1H, m) , 2.90-3.12 (1H, m), 2.98 (2H, t, J = 8.0 Hz), 4.04-4.16 (1H, 1) , 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J = 8. Hz), 9.66 (1H, bs). Ή-NR (CDC1 3): δ 0.928 (6H, d, J = 6.2 Hz), 1.44-2.02 (7H, m), 2.42 (3H, s), 2.48-2.66 (1H, m), 2.90-3.12 ( 1H, m), 2.98 (2H, t, J = 8.0 Hz), 4.04-4.16 (1H, 1), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8. Hz) , 9.66 (1H, bs).
元素分析値: C22H27N304S3 · 0.3H20として Elementary analysis: as C 22 H 27 N 3 0 4 S 3 · 0.3H 2 0
計算値 (%) : C, 52.95; H, 5.57; N, 8.42. 実測値 (¾) : C, 52.95; H, 5.47; N, 8.19. Calculated value (%): C, 52.95; H, 5.57; N, 8.42. Found (¾): C, 52.95; H, 5.47; N, 8.19.
実施例 4 - 44 Example 4-44
N - [4- (ィソペンチルスルファニル) -卜ォキソ -1, 2-ジヒド口- 3H-ィミダ ゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4-メチルベンゼンスルホ ンアミ ド N- [4- (Isopentylsulfanyl) -toxo-1,2-dihydrido-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzenesulfo Medium
実施例 4-43で得られた N- [6-ヒドロキシ- 4 -(ィソペンチルスルファ二 ル)-卜ォキソ -1, 2, 6, 7, 8, 9-へキサヒドロ- 3H -ィミダゾ [5, 1-c] [1,4]ベ ンゾチアジン- 3-ィリデン] -4-メチルベンゼンスルホンアミ ド (980 rag, 1.99 iiol) と p-トルエンスルホン酸 1水和物 (318 mg, 1.59 mmol) の トルエン (20.0 ml) 溶液に、 無水酢酸 (0.562 ml, 5.96 mmol) を加え た。 反応混合物を 100°Cで 10分かき混ぜた後、 溶媒を減圧下に留去し、 残留物に水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化 合物 (Π8 mg, 15¾) を橙色結晶として得た。 酢酸ェチル -メタノール - テトラヒドロフランから再結晶して融点 207-209°Cの橙色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) リ: 3250, 1744, 1626, 1586, 1557, 1485 cm—1. N- [6-hydroxy-4- (isopentylsulfanyl) -toxo-1,2,6,7,8,9-hexahydro-3H-imidazo obtained in Example 4-43 [ 5, 1-c] [1,4] Benzothiazine-3-ylidene] -4-methylbenzenesulfonamide (980 rag, 1.99 iiol) and p-toluenesulfonic acid monohydrate (318 mg, 1.59 mmol) Acetic anhydride (0.562 ml, 5.96 mmol) was added to a toluene (20.0 ml) solution of. After stirring the reaction mixture at 100 ° C for 10 minutes, the solvent was distilled off under reduced pressure, water was added to the residue, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound ({8 mg, 15}) as orange crystals. Recrystallization from ethyl acetate-methanol-tetrahydrofuran gave orange crystals having a melting point of 207-209 ° C. Infrared absorption spectrum (IR) (in KBr): 3250, 1744, 1626, 1586, 1557, 1485 cm— 1 .
Ή-NMR (CDC13) : δ 0.952 (6Η, d, J = 6.2 Hz), 1.54-1.80 (3H, 1) , 2.42 (3H, s), 3.02 (2H, d, J = 7.6 Hz), 6.95 (1H, dd, J = 7.6, 1.4 Hz), 7.07 Ή-NMR (CDC1 3): δ 0.952 (6Η, d, J = 6.2 Hz), 1.54-1.80 (3H, 1), 2.42 (3H, s), 3.02 (2H, d, J = 7.6 Hz), 6.95 (1H, dd, J = 7.6, 1.4 Hz), 7.07
(1H, dt, J = 7.2, 1.0 Hz), 7.21 (1H, ddd, J = 8.4, 4.2, 1.8 Hz), 7.30(1H, dt, J = 7.2, 1.0 Hz), 7.21 (1H, ddd, J = 8.4, 4.2, 1.8 Hz), 7.30
(2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8. Hz), 8.46 (1H, dd, J = 8. , 1.0(2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8. Hz), 8.46 (1H, dd, J = 8., 1.0
Hz), 9.80 (1H, bs). Hz), 9.80 (1H, bs).
元素分析値: C22H23N303S3として Elementary analysis: as C 22 H 23 N 3 0 3 S 3
計算値 (%) : C, 55.79; H, 4.89; N, 8.87; S, 20.31. Calculated value (%): C, 55.79; H, 4.89; N, 8.87; S, 20.31.
実測値 (¾) : C, 55.89; H, 4.65; N, 8.85; S, 20.27. Found (¾): C, 55.89; H, 4.65; N, 8.85; S, 20.27.
実施例 4 - 45 Example 4-45
N-[4- (イソペンチルスルファ二ル)- 1-ォキソ _1, 2, 7, 8-テトラヒ ドロ - 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4-メチルベン ゼンスルホンアミ ド N- [4- (Isopentylsulfanyl) -1-oxo-1,2,7,8-tetrahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene]- 4-methylben Zensulfonamide
実施例 4-43で得られた N_[6-ヒドロキシ- 4- (ィソペンチルスルファ二 ル)-卜ォキソ -1, 2, 6, 7, 8, 9-へキサヒドロ- 3H -ィミダゾ [5,卜 c] [1,4]ベ ンゾチアジン- 3-ィリデン ]-4-メチルベンゼンスルホンアミド (200 nig, 0.405 mmol) と p-トルエンスルホン酸 1水和物 (64.9 mg, 0.324 mmol) のトルエン (4.00 ml) 溶液に、 無水酢酸 (0.115 ml, 1.22 mmol) を加 えた。反応混合物を室温で 15分かき混ぜた後、 反応混合物に水を加えて 酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去して題記化合物 (108mg, 57¾) を橙色結晶として得た。 酢酸ェチ ル-ジェチルエーテルから再結晶して融点 154- 156での橙色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) リ: 3230, 2957, 2934, 2870, 1732, 1622, 1584, 1553 cm"1. N_ [6-hydroxy-4- (isopentylsulfanyl) -toxo-1,2,6,7,8,9-hexahydro-3H-imidazo obtained in Example 4-43 [5 C, [1,4] benzothiazine-3-ylidene] -4-methylbenzenesulfonamide (200 nig, 0.405 mmol) and p-toluenesulfonic acid monohydrate (64.9 mg, 0.324 mmol) in toluene ( Acetic anhydride (0.115 ml, 1.22 mmol) was added to the 4.00 ml) solution. After stirring the reaction mixture at room temperature for 15 minutes, water was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (108mg, 57¾) as orange crystals. Recrystallization from ethyl acetate-ethyl ether gave an orange crystal with a melting point of 154-156. Infrared absorption spectrum (IR) (in KBr): 3230, 2957, 2934, 2870, 1732, 1622, 1584, 1553 cm " 1 .
Ή-NMR (CDC13) : (50.926 (6H, d, J = 6.2 Hz), 1.46-1.78 (3H, 1) , 2.27-2.38 (4Η, m) , 2.42 (3Η, s) , 2.96 (2Η, t, J = 8.0 Hz), 6.12 (1H, t, J=4.0 Hz), 6.94 (1H, t, J = 5.0 Hz), 7.29 (1H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.6 Hz), 9.68 (1H, bs). Ή-NMR (CDC1 3): (50.926 (6H, d, J = 6.2 Hz), 1.46-1.78 (3H, 1), 2.27-2.38 (4Η, m), 2.42 (3Η, s), 2.96 (2Η, t, J = 8.0 Hz), 6.12 (1H, t, J = 4.0 Hz), 6.94 (1H, t, J = 5.0 Hz), 7.29 (1H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.6 Hz), 9.68 (1H, bs).
実施例 4 - 46 Example 4-46
N- [1, 6-ジォキソ- 4- (ィソブチルスルファ二ル)- 1, 2, 6, 7, 8, 9-へキサヒ ド口- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4-メチル ベンゼンスルホンアミ ド N- [1,6-dioxo-4- (isobutylsulfanyl) -1,2,6,7,8,9-hexahydrid-3H-imidazo [5,1-c] [1 , 4] Benzothiazine-3-ylidene] -4-methylbenzenesulfonamide
実施例 4-42 と同様の方法により、よう化ィソァミルの代わりにょう化 イソブチル (0.546 ml, 4.74 mmol) を用いて、 実施例 3- 38で得られた N -(1, 6-ジォキソ- 4-スルファニル - 1, 2, 6, 7, 8, 9_へキサヒドロ- 3H-ィミ ダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン) -4-メチルベンゼンスル ホンアミ ド (2.00 g, 4.74 mmol) から題記化合物 (1.82 g, 80%) を合 成した。酢酸ェチルから再結晶して融点 223-225°Cの暗赤色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) レ : 3218, 2961, 2928, 2872, 1759, 1653, 1624, 1595, 1549, 1495 cm"1. In the same manner as in Example 4-42, using isobutyl sulphide (0.546 ml, 4.74 mmol) in place of isisoamyl iodide, the N- (1,6-dioxo-4) obtained in Example 3-38 was obtained. -Sulfanyl-1,2,6,7,8,9_Hexahydro-3H-imidazo [5, 1-c] [1,4] benzothiazine-3-ylidene) -4-methylbenzenesulfonamide ( The title compound (1.82 g, 80%) was synthesized from 2.00 g, 4.74 mmol). Recrystallization from ethyl acetate gave dark red crystals with a melting point of 223-225 ° C. Infrared absorption spectrum (IR) (in KBr) Re: 3218, 2961, 2928, 2872, 1759, 1653, 1624, 1595, 1549, 1495 cm " 1 .
Ή-NMR (CDCI3) : δ 1.04 (6Η, d, J = 7.0 Hz), 1.86-2.16 (3H, m), 2.42 (3H, s), 2.50 (2H, t, J = 5. Hz), 2.92 (2H, d, J = 6.6 Hz), 3.21 (2H, t, J = 6.2 Hz), 7.29 (2H, d, J = 8.0 Hz); 7.85 (2H, d, J = 8.0 Hz), 9.84 (1H, bs). Ή-NMR (CDCI3): δ 1.04 (6Η, d, J = 7.0 Hz), 1.86-2.16 (3H, m), 2.42 (3H, s), 2.50 (2H, t, J = 5. Hz), 2.92 (2H, d, J = 6.6 Hz), 3.21 (2H, t, J = 6.2 Hz), 7.29 (2H, d, J = 8.0 Hz); 7.85 (2H, d, J = 8.0 Hz), 9.84 (1H, bs).
元素分析値: C21H23N304S3として Elementary analysis: as C 21 H 23 N 3 0 4 S 3
計算値 (%) : C, 52.81; H, 4.85; N, 8.80; S, 20.14. Calculated value (%): C, 52.81; H, 4.85; N, 8.80; S, 20.14.
実測値 ( ) : C, 52.54; H, 4.59; N, 8.73; S, 19.96. Found (): C, 52.54; H, 4.59; N, 8.73; S, 19.96.
実施例 4 - 47 Example 4-47
N_[6-ヒドロキシ- 4- (ィソブチルスルファニル) -卜ォキソ -1, 2, 6, 7, 8, 9- へキサヒ ドロ - 3H -イミダゾ [5, 1-c] [1, 4]ベンゾチアジン -3-ィ リデ 実施例 4-43 と同様の方法により、 実施例 4-46で得られた N-[l, 6 -ジ ォキソ -4- (ィソブチルスルファニル) - 1, 2, 6, 7, 8, 9_へキサヒドロ- 3H-ィ ミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3 -ィ Vデン] -4-メチルベンゼンス ルホンアミド (1.82 g, 3.81 mmol) から題記化合物 (690 mg, 38%) を 合成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 143- 145°C の暗赤色結晶を得た。 N_ [6-Hydroxy-4- (isobutylsulfanyl) -toxo-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine -3-ylide N- [l, 6-dioxo-4- (isobutylsulfanyl) -l, 2,6, obtained in Example 4-46 in the same manner as in Example 4-43. 7,8,9_Hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-yVden] -4-methylbenzenesulfonamide (1.82 g, 3.81 mmol) from the title compound (690 mg, 38%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 143-145 ° C.
赤外吸収スペクトル(IR) ( Br 中) レ : 3600-3000, 2959, 2872, 1742, 1653, 1617, 1557 cm—Infrared absorption spectrum (IR) (in Br) Re: 3600-3000, 2959, 2872, 1742, 1653, 1617, 1557 cm—
-丽 R (CDC13) : δ 1.03 (6Η, d, J = 7.0 Hz), 1.65-2.08 (5H, m) , 2.42 (3H, s), 2.46-2.66 (1H, m) , 2.86 (2H, d, J = 6.6 Hz), 2.80-3.08 (1H, m), 4.04-4.1.5 (1H, m) , 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4-丽R (CDC1 3): δ 1.03 (6Η, d, J = 7.0 Hz), 1.65-2.08 (5H, m), 2.42 (3H, s), 2.46-2.66 (1H, m), 2.86 (2H, d, J = 6.6 Hz), 2.80-3.08 (1H, m), 4.04-4.1.5 (1H, m), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4)
Hz), 9.65 (1H, bs) . Hz), 9.65 (1H, bs).
元素分析値: C21H25N304S3 · 0.2H20として Elementary analysis: as C 21 H 25 N 3 0 4 S 3 · 0.2H 2 0
計算値 ( ) : C, 52.20; H, 5.30; , 8.70; S, 19.91. Calculated (): C, 52.20; H, 5.30;, 8.70; S, 19.91.
実測値 ) : C, 52.25; H, 5.14; N, 8.65; S, 19.58. Observed): C, 52.25; H, 5.14; N, 8.65; S, 19.58.
実施例 4-48 Example 4-48
N-[4- (ィソブチルスルファニル) -卜ォキソ _1, 2-ジヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4-メチルベンゼンスルホン アミ ド 実施例 4-44と同様の方法により、 実施例 4-47で得られた N_[6-ヒド 口キシ- 4- (ィソブチルスルファ二ル)-卜ォキソ -1, 2, 6, 7, 8, 9-へキサヒ ドロ- 3H -ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4-メチル ベンゼンスルホンアミ ド(150 mg, 0.313 mmol)から題記化合物(43.1 mg, 30%) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 233- 235°Cの橙色結晶を得た。 N- [4- (Isobutylsulfanyl) -toxo_1,2-dihydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzenesulfonamide In the same manner as in Example 4-44, the N_ [6-hydroxy-4- (isobutylsulfanyl) -toxo-1,2,6,7 obtained in Example 4-47 was obtained. , 8,9-Hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzenesulfonamide (150 mg, 0.313 mmol) from the title compound (43.1 mg, 30%). Recrystallization from ethyl acetate-getyl ether gave orange crystals with a melting point of 233-235 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3237, 2961, 1744, 1628, 1586; 1555, 1485 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3237, 2961, 1744, 1628, 1586; 1555, 1485 cm " 1 .
Ή-NMR (CDC") : δ 1.05 (6Η, d, J = 6.6 Hz), 1.88-2.08 (1H, m) , 2.42 (3H, s), 2.89 (2H, d, J = 7.0 Hz), 6.95 (1H, d, J = 8.2 Hz), 7.06 (1H, t, J = 6.2 Hz), 7.21 (1H, t, J-7.0 Hz), 7.29 (2H, d, 8.4 Hz), 7.88 (2H, d, J = 8.4 Hz), 8. 6 (1H, d, J = 6.8 Hz), 9.79 (1H, bs). Ή-NMR (CDC "): δ 1.05 (6Η, d, J = 6.6 Hz), 1.88-2.08 (1H, m), 2.42 (3H, s), 2.89 (2H, d, J = 7.0 Hz), 6.95 (1H, d, J = 8.2 Hz), 7.06 (1H, t, J = 6.2 Hz), 7.21 (1H, t, J-7.0 Hz), 7.29 (2H, d, 8.4 Hz), 7.88 (2H, d , J = 8.4 Hz), 8.6 (1H, d, J = 6.8 Hz), 9.79 (1H, bs).
実施例 4-49 Example 4-49
N-[l, 6-ジォキソ- 4- (ペンチルスルファニル) -1, 2, 6, 7, 8, 9 -へキサヒド 口- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3 -ィリデン ]- 4_メチルベ ンゼンスルホンアミド N- [l, 6-Dioxo-4- (pentylsulfanyl) -1,2,6,7,8,9-hexahydride-3H-imidazo [5,1-c] [1,4] benzothiazine-3 -Ylidene]-4_methylbenzenesulfonamide
実施例 4-42と同様の方法により、よう化イソァミルの代わりにョ一ド ペンタン (0.911 ml, 6.99 mmol) を用いて、 実施例 3-38 で得られた Obtained in Example 3-38 by a method similar to that in Example 4-42, except that iodopentane (0.911 ml, 6.99 mmol) was used instead of isoamyl iodide.
N-(l, 6 -ジォキソ- 4-スルファニル- 1, 2, 6, 7, 8, 9 -へキサヒドロ- 3H -ィミ ダゾ [5, 1-c] [1, 4]ベンゾチアジン- 3-ィリデン) -4-メチルベンゼンスル ホンアミ ド (2.68 g, 6.36 mmol) から題記化合物 (2.72 g, 87%) を合 成した。エタソ一ル-ジェチルェ一テルから再結晶して融点 218- 220 の 暗赤色結晶を得た。 N- (l, 6-Dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene ) The title compound (2.72 g, 87%) was synthesized from -4-methylbenzenesulfonamide (2.68 g, 6.36 mmol). Recrystallization from ethasol-jetilether gave dark red crystals with a melting point of 218-220.
赤外吸収スペクトル(IR) (KBr中) リ: 3187, 2955, 2928, 2870, 2857, 1738, 1651, 1609 cm-1. Ή-NMR (CDC13) : δ 0.908 (3Η, t, J = 6.6 Hz), 1.24-1.46 (4H, m) , 1.60-1.78 (2H, m) , 1.96-2.12 (2H, m) , 2.42 (3H, s), 2.51 (2H, t, 7.2 Hz), 3.03 (2H, t, J-7.6 Hz), 3.21 (2H, t, J = 6.0 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.86 (1H, bs) . 実施例 4-50 N_[6-ヒドロキシ- 1-ォキソ -4- (ペンチルスルファニル) -1, 2, 6, 7, 8, 9 -へ キサヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] - 4 - 実施例 4-43 と同様の方法により、 実施例 4 - 49で得られた N-[l,6 -ジ ォキソ -4- (ペンチルスルファ二ル)- 1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミ ダゾ [5,1- c] [1, 4]ベンゾチアジン- 3-ィリデン] -4-メチルベンゼンスル ホンアミ ド (1.82 g, 3.81 mmol) から題記化合物 (690 mg, 38%) を合 成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 143- 145^の 暗赤色結晶を得た。 Infrared absorption spectrum (IR) (in KBr) Re:. 3187, 2955, 2928, 2870, 2857, 1738, 1651, 1609 cm -1 Ή-NMR (CDC1 3): δ 0.908 (3Η, t, J = 6.6 Hz), 1.24-1.46 (4H, m), 1.60-1.78 (2H, m), 1.96-2.12 (2H, m), 2.42 (3H, s), 2.51 (2H, t, 7.2 Hz), 3.03 (2H , t, J-7.6 Hz), 3.21 (2H, t, J = 6.0 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.86 (1H, bs Example 4-50 N_ [6-hydroxy-1-oxo-4- (pentylsulfanyl) -1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3 -Ylidene]-4-In the same manner as in Example 4-43, N- [l, 6-dioxo-4- (pentylsulfanyl) -1,2,2 obtained in Example 4-49. 6, 7, 8, 9-Hexahydro-3H-imidazo [5,1-c] [1, 4] benzothiazine-3-ylidene] -4-methylbenzenesulfonamide (1.82 g, 3.81 mmol) The title compound (690 mg, 38%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals, mp 143-145 ^.
赤外吸収スペクトル(IR) (KBr 中) リ: 3600-3000, 2959, 2872, 1742, 1653, 1617, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr) Re: 3600-3000, 2959, 2872, 1742, 1653, 1617, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 1.03 (6Η, d, J = 7.0 Hz), 1.65-2.08 (5H, m) , 2.42 (3H, s), 2.46-2.66 (1H, m), 2.86 (2H, d, J = 6.6 Hz), 2.80-3.08 (1H, m), 4.04-4.15 (1H, m) , 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.65 (1H, bs). Ή-NMR (CDC1 3): δ 1.03 (6Η, d, J = 7.0 Hz), 1.65-2.08 (5H, m), 2.42 (3H, s), 2.46-2.66 (1H, m), 2.86 (2H, d, J = 6.6 Hz), 2.80-3.08 (1H, m), 4.04-4.15 (1H, m), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.65 (1H, bs).
元素分析値: C21H25N304S3 ' 0.2H20として Elementary analysis: as C 21 H 25 N 3 0 4 S 3 '0.2H 2 0
計算値 (¾) : C, 52.20; H, 5.30; , 8.70; S, 19.91. Calculated (¾): C, 52.20; H, 5.30;, 8.70; S, 19.91.
実測値 ) : C, 52.25; H, 5.14; N, 8.65; S, 19.58. Obtained): C, 52.25; H, 5.14; N, 8.65; S, 19.58.
実施例 4-51 Example 4-51
N- [卜ォキソ -4- (ペンチルスルファニル)-1, 2-ジヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4-メチルベンゼンスルホン アミ ド N- [Toxo-4- (pentylsulfanyl) -1,2-dihydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzenesulfonamide
実施例 4-44 と同様の方法により、 実施例 4-50で得られた N_[6-ヒド ロキシ -1-ォキソ -4- (ペンチルスルファニル) - 1, 2, 6, 7, 8, 9-へキサヒド 口- 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン- 3-ィリデン ]-4-メチルベ ンゼンスルホンアミ ド (200 mg, 0.405匪 ol) から題記化合物 (53.8mg, 28%) を合成した。 酢酸ェチルから再結晶して融点 176-178°Cの橙色結晶 を得た。 N_ [6-hydroxy-1-oxo-4- (pentylsulfanyl) -1, 2, 6, 7, 8, 9- obtained in Example 4-50 by the same method as in Example 4-44. Hexahydrid-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzenebenzenesulfonamide (200 mg, 0.405 bandol) to give the title compound (53.8 mg, 28% ) Was synthesized. Recrystallization from ethyl acetate gave orange crystals with a melting point of 176-178 ° C.
赤外吸収スペクトル(IR) (KBr中) ソ : 3225, 2957, 2930, 2859, 1744, 1626, 1586, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr) So: 3225, 2957, 2930, 2859, 1744, 1626, 1586, 1557 cm " 1 .
Ή-N R (CDClg) : δ 0.923 (3H, t, J = 6.6 Hz), 1.32-1.48 (4H, m) , 1.64-1.82 (2H, m) , 2.42 (3H, s), 3.01 (2H, d, J = 7.6 Hz), 6.95 (1H, d, J = 7.4 Hz), 7.07 (1H, t, J = 7.4 Hz), 7.14-7.26 (1H, i) , 7.30 (2H, d, J = 8.0 Hz), 7.88 (2H, d, J = 8.2 Hz), 8.46 (1H, d, J = 8.0 Hz), 9.78 (1H, bs) . Ή-NR (CDClg): δ 0.923 (3H, t, J = 6.6 Hz), 1.32-1.48 (4H, m), 1.64-1.82 (2H, m), 2.42 (3H, s), 3.01 (2H, d , J = 7.6 Hz), 6.95 (1H, d, J = 7.4 Hz), 7.07 (1H, t, J = 7.4 Hz), 7.14-7.26 (1H, i), 7.30 (2H, d, J = 8.0 Hz) ), 7.88 (2H, d, J = 8.2 Hz), 8.46 (1H, d, J = 8.0 Hz), 9.78 (1H, bs).
元素分析値: C22H23N303S3 · 0.5C4H802として ' 計算値 ) : C, 55.68; H, 5.26; N, 8.12; S, 18.58. Elemental analysis: 'Calculated C 22 H 23 N 3 0 3 S 3 · 0.5C 4 H 8 0 2): C, 55.68; H, 5.26; N, 8.12; S, 18.58.
実測値 (%) : C, 55.55; H, 5.23; N, 7.88; S, 18.62. Found (%): C, 55.55; H, 5.23; N, 7.88; S, 18.62.
実施例 4-52 Example 4-52
N-[4- (シク口へプチルスルファニル)-1, 6-ジォキソ- 1, 2, 6, 7, 8, 9-へキ サヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン] -4 -メ 実施例 4-42と同様の方法により、よう化イソァミルの代わりによう化 シクロへプチル (0.877 ml, 3.64 mmol) を用いて、 実施例 3-38で得ら れた N- (1, 6-ジォキソ -4-スルファニル -1, 2, 6, 7, 8, 9-へキサヒドロ- 3H- ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン) -4-メチルベンゼン スルホンアミ ド (1.28 g, 3.04 mmol) から題記化合物 (1.12 g, 71%) を合成した。 酢酸ェチル-メタノ一ル-テトラヒドロフランから再結晶し て融点 229- 23 Cの暗赤色結晶を得た。 N- [4- (cyclohexylheptylsulfanyl) -1,6-dioxo-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] [Benzothiazine-3-ylidene] -4-me obtained in the same manner as in Example 4-42 except that cycloheptyl iodide (0.877 ml, 3.64 mmol) was used instead of isoamyl iodide. The N- (1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3- The title compound (1.12 g, 71%) was synthesized from (ylidene) -4-methylbenzene sulfonamide (1.28 g, 3.04 mmol). Recrystallization from ethyl acetate-methanol-tetrahydrofuran gave dark red crystals of melting point 229-23C.
赤外吸収スペクトル(IR) (KBr中) レ: 3191, 2930, 2853, 1746, 1645, 1609, 1541 cm—1. Infrared absorption spectrum (IR) (in KBr) d: 3191, 2930, 2853, 1746, 1645, 1609, 1541 cm— 1 .
Ή-NMR (CDC13) : δ 1.42-1.90 (10H, m) , 1.96-2.16 (4H, m) , 2.43 (3H, s), 2.51 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 6.2 Hz), 3.60-3.80 (1H, m), 7.30 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J-8.4 Hz), 9.86 (1H, bs). 元素分析値: C22H25N304S3として Ή-NMR (CDC1 3): δ 1.42-1.90 (10H, m), 1.96-2.16 (4H, m), 2.43 (3H, s), 2.51 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 6.2 Hz), 3.60-3.80 (1H, m), 7.30 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J-8.4 Hz), 9.86 (1H, bs). Elemental analysis value: as C 22 H 25 N 3 0 4 S 3
計算値 ) : C, 53.74; H, 5.13; N, 8.55; S, 19.57. Calculated)): C, 53.74; H, 5.13; N, 8.55; S, 19.57.
実測値 (%) : C, 53.68; H, 5.08; N, 8.27; S, 19.31. Found (%): C, 53.68; H, 5.08; N, 8.27; S, 19.31.
実施例 4 - 53 N- [4- (シク 口へプチルスルフ ァニル) -6-ヒ ド 口キシ- 1-ォキソ - 1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミダゾ [5, 1- c] [1,4]ベンゾチアジン - 3-ィリデン] -4-メチルベンゼンスルホンアミ ド Example 4-53 N- [4- (cyclohexylsulfanyl) -6-hydroxy-1-oxo-1,2,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c] [ 1,4] Benzothiazine-3-ylidene] -4-methylbenzenesulfonamide
実施例 4-43と同様の方法により、実施例 4-52で得られた N- [4- (シク 口へプチルスルファ二ル)- 1,6-ジォキソ -1,2, 6, 7, 8, 9-へキサヒドロ - 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン- 3-ィリデン] -4-メチルベン ゼンスルホンアミ ド (1.56 g, 3.01 mmol) から題記化合物 (657 mg, 42¾) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 156- 158°Cの暗赤色結晶を得た。 In the same manner as in Example 4-43, N- [4- (cyclohexylsulfanyl) -1,6-dioxo-1,2,6,7,8, 9-Hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzenesulfonamide (1.56 g, 3.01 mmol) from the title compound (657 mg, 42¾) Was synthesized. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 156-158 ° C.
赤外吸収スペクトル(IR) (KBr 中) リ: 3700-3000, 2932, 2855, 1740, 1657, 1615, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr): 3700-3000, 2932, 2855, 1740, 1657, 1615, 1557 cm " 1 .
-腿 R (CDC13) : δ 1. 4-2.16 (16H, m) , 2.42 (3H, s) , 2.46-2.66 (1Η, m), 2.90-3.10 (1H, m) , 3.52-3.70 (1H, m), 4.04-4.18 (1H, m), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.66 (1H, bs). - thigh R (CDC1 3): δ 1. 4-2.16 (16H, m), 2.42 (3H, s), 2.46-2.66 (1Η, m), 2.90-3.10 (1H, m), 3.52-3.70 (1H , m), 4.04-4.18 (1H, m), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.66 (1H, bs).
元素分析値: C24H2gN304S3として Elementary analysis: as C 24 H 2g N 3 0 4 S 3
計算値 ) : C, 55.47; H, 5.62; N, 8.09; S, 18.51. Calculated values): C, 55.47; H, 5.62; N, 8.09; S, 18.51.
実測値 (%) : C, 55.33; H, 5.66; N, 7.82; S, 18.28. Found (%): C, 55.33; H, 5.66; N, 7.82; S, 18.28.
実施例 4 - 54 Example 4-54
N - [4- (シク口へプチルスルファニル) -卜ォキソ -1,2-ジヒドロ - 3H-ィミ ダゾ [5,1- c] [1, 4]ベンゾチアジン- 3 -ィリデン] -4-メチルベンゼンスル ホンアミド N- [4- (Cyctoheptylsulfanyl) -toxo-1,2-dihydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzene Sulfonamide
実施例 4-44と同様の方法により、実施例 4-53で得られた N-[4- (シク 口へプチルスルファニル)_6-ヒドロキシ- 1-ォキソ -1, 2, 6, 7, 8, 9-へキサ ヒド口- 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン- 3-ィリデン] -4-メチ ルベンゼンスルホンアミ ド (456 mg, 0.879 mmol) から題記化合物 (51.8 mg, 12%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融 点 218- の橙色結晶を得た。 In the same manner as in Example 4-44, N- [4- (cyclohexylsulfanyl) _6-hydroxy-1-oxo-1,2,6,7,8, obtained in Example 4-53. 9-Hexahydrid-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene] -4-methylbenzenesulfonamide (456 mg, 0.879 mmol) from the title compound (51.8 mg , 12%). The crystals were recrystallized from ethyl acetate-ethyl ether to give orange crystals having a melting point of 218-.
赤外吸収スペクトル(IR) (KBr中) リ: 3239, 2928, 2855, 1746, 1626, 1588, 1557, 1483 cm"1. Ή-NMR (CDCI3) : δ 1.40-2.20 (12H, m) , 2.42 (3Η, s), 3.48-3.68 (1Η, m), 6.95 (1H, dd, J=8.0, 1.4 Hz), 7.06 (1H, dt, J = 7.4, 1. Hz), 7.20 (1H, dt, 8.0, 1.4 Hz), 7.30 (2H, d, 8.0 Hz), 7.88 (2H, d, J=8.0 Hz), 8. 5 (1H, dd, J = 8. , 1.0 Hz), 9.79 (1H, bs). Infrared absorption spectrum (IR) (in KBr): 3239, 2928, 2855, 1746, 1626, 1588, 1557, 1483 cm " 1 . Ή-NMR (CDCI3): δ 1.40-2.20 (12H, m), 2.42 (3Η, s), 3.48-3.68 (1Η, m), 6.95 (1H, dd, J = 8.0, 1.4 Hz), 7.06 (1H , dt, J = 7.4, 1.Hz), 7.20 (1H, dt, 8.0, 1.4 Hz), 7.30 (2H, d, 8.0 Hz), 7.88 (2H, d, J = 8.0 Hz), 8.5 ( 1H, dd, J = 8., 1.0 Hz), 9.79 (1H, bs).
実施例 4-55 Example 4-55
N - {1, 6-ジォキソ- 4- [(4, 4, 5, 5, 5 -ペン夕フルォロペンチル)スルファニ ル]- 1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミダゾ [5,卜 c] [1, 4]ベンゾチア 実施例 4-42と同様の方法により、よう化イソァミルの代わりにメタン スルホン酸 4,4,5,5,5_ペンタフルォロペンチル エステル (201 mg, 0.783 mmol) を用いて、 実施例 3 - 38で得られた N- (1, 6-ジォキソ- 4-ス ルファニル -1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミダゾ [5,1- c] [1,4]ベン ゾチアジン- 3-ィリデン)-4-メチルベンゼンスルホンアミ ド (300 mg, 0.712 mmol) から題記化合物 (65.4 mg, 16%) を合成した。 酢酸ェチル- ジェチルェ一テルから再結晶して融点 205- 207°Cの暗赤色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) リ: 3200, 3061, 2959, 1744, 1653, 1613, 1553 cm-1. N- {1,6-dioxo-4-[(4,4,5,5,5-pentafluorofluorpentyl) sulfanyl]-1,2,6,7,8,9-hexahydro-3H-imidazo [ 5, tric] [1,4] benzothia In the same manner as in Example 4-42, methanesulfonic acid 4,4,5,5,5_pentafluoropentyl ester (201 mg) was used instead of isoamyl iodide. , 0.783 mmol) using the N- (1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo obtained in Example 3-38 [ The title compound (65.4 mg, 16%) was synthesized from 5,1-c] [1,4] benzothiazine-3-ylidene) -4-methylbenzenesulfonamide (300 mg, 0.712 mmol). Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 205-207 ° C. Infrared absorption spectrum (IR) (in KBr): 3200, 3061, 2959, 1744, 1653, 1613, 1553 cm- 1 .
Ή-NMR (CDCI3) : δ 1.95-2.24 (6Η, m) , 2.43 (3H, s) , 2.51 (2H, t, J = 7.2 Hz), 3.10 (2H, t, J = 6.9 Hz), 3.21 (2H, t, J = 6.3 Hz), 7.30 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.87 (1H, bs) . Ή-NMR (CDCI3): δ 1.95-2.24 (6Η, m), 2.43 (3H, s), 2.51 (2H, t, J = 7.2 Hz), 3.10 (2H, t, J = 6.9 Hz), 3.21 ( 2H, t, J = 6.3 Hz), 7.30 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.87 (1H, bs).
元素分析値: C22H2。N304S3F5として Elemental analysis: C 22 H 2. N 3 0 4 S 3 F 5
計算値 ) : C, 45. 3; H, 3.47; N, 7.22; S, 16.54. Calculated values): C, 45.3; H, 3.47; N, 7.22; S, 16.54.
実測値 (¾) : C, 45.24; H, 3.22; N, 7.32; S, 16.45. Found (¾): C, 45.24; H, 3.22; N, 7.32; S, 16.45.
実施例 -56 Example -56
N - {1,6- ジ ォ キ ソ -4-[(3, 3- ジ メ チ ル ブ チ ル ) ス ル フ ァ ニ ル]- 1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジ ン- 3-ィリデン }- 4-メチルベンゼンスルホンアミド N- {1,6-Dioxo-4-[(3,3-dimethylbutyl) sulfanyl]-1,2,6,7,8,9-hexahydro -3H-imidazo [5, 1-c] [1, 4] benzothiazin-3-ylidene} -4-methylbenzenesulfonamide
実施例 4-42と同様の方法により、よう化イソァミルの代わりにメタン スルホン酸 3,3-ジメチルブチル エステル (70.6 mg, 0.391 ramol) を用 いて、 実施例 3-38 で得られた N -(1, 6-ジォキソ- 4-スルファニル - 1, 2, 6, 7, 8, 9-へキサヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン - 3-ィリデン) -4-メチルベンゼンスルホンアミ ド (150 mg, 0.356 mniol) から題記化合物 (8.8mg, 4.9%) を合成した。 酢酸ェチル-ジェチルェ一 テルから再結晶して融点 223- 225°Cの暗赤色結晶を得た。 In the same manner as in Example 4-42, methanesulfonic acid 3,3-dimethylbutyl ester (70.6 mg, 0.391 ramol) was used in place of isoamyl iodide. And N- (1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo obtained in Example 3-38 [5,1-c] [ The title compound (8.8 mg, 4.9%) was synthesized from 1,4] benzothiazine-3-ylidene) -4-methylbenzenesulfonamide (150 mg, 0.356 mniol). Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 223-225 ° C.
赤外吸収スペクトル(IR) (KBr中) リ : 3187, 3073, 2957, 2868, 1742, 1651, 1609, 1584, 1547 cm—1. Infrared absorption spectrum (IR) (in KBr): 3187, 3073, 2957, 2868, 1742, 1651, 1609, 1584, 1547 cm- 1 .
lH-NMR (CDC13) : δ 0.949 (9H, s), 1.50-1.64 (2H, m) , 1.96-2.12 (2H, m), 2. 3 (3H, s), 2.51 (2H, t, J = 6.2 Hz), 3.00 (2H, t, J = 9.2 Hz), 3.21 (2H, t, J = 6.2 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.88 (1H, bs). lH-NMR (CDC1 3): δ 0.949 (9H, s), 1.50-1.64 (2H, m), 1.96-2.12 (2H, m), 2. 3 (3H, s), 2.51 (2H, t, J = 6.2 Hz), 3.00 (2H, t, J = 9.2 Hz), 3.21 (2H, t, J = 6.2 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4) Hz), 9.88 (1H, bs).
実施例 4 - 57 Example 4-57
N - {1-ォキソ -4- [(4, 4, 5, 5, 5-ペンタフルォロペンチル)スルファニ ル ]-1, 2 -ジヒドロ- 3H-ィミダゾ [5, 1-c] [1,4;]ベンゾチアジン- 3-ィリデ 実施例 4-42と同様の方法により、よう化イソァミルの代わりにメタン スルホン酸 4, 4, 5, 5, 5-ペンタフルォロペンチル エステル (76.2 mg, 0.297 mmol) を用いて、 実施例 3-37で得られた N- (卜ォキソ -4-スルフ ァニル - 1, 2-ジヒドロ- 3H-ィ 'ミダゾ [5, 1-c] [1,4]ベンゾチアジン -3-ィリ デン) -4-メチルベンゼンスルホンアミ ド (100 mg, 0.248 mmol) から題 記化合物 (100 mg, 72¾) を合成した。 酢酸ェチル-ジェチルエーテルか ら再結晶して融点 210-212^:の暗赤色結晶を得た。 N- {1-oxo-4-[(4,4,5,5,5-pentafluoropentyl) sulfanyl] -1,2-dihydro-3H-imidazo [5,1-c] [1, 4;] Benzothiazine-3-ylide In the same manner as in Example 4-42, methanesulfonic acid 4,4,5,5,5-pentafluoropentyl ester (76.2 mg, 0.297 mmol) was used instead of isoamyl iodide. ) To give N- (toxo-4-sulfanyl-1,2-dihydro-3H-'midazo [5,1-c] [1,4] benzothiazine- obtained in Example 3-37- The title compound (100 mg, 72 mg) was synthesized from 3-ylidene) -4-methylbenzenesulfonamide (100 mg, 0.248 mmol). Recrystallization from ethyl acetate-getyl ether yielded dark red crystals with a melting point of 210-212 ^ :.
赤外吸収スペクトル(IR) (KBr中) リ : 3170, 3071, 1742, 1630, 1590, 1559, 1485 cm—1. Infrared absorption spectrum (IR) (in KBr) Re: 3170, 3071, 1742, 1630, 1590, 1559, 1485 cm— 1 .
Ή-NMR (CDCI3) : δ 2.09-2.34 (4Η, m) , 2.43 (3Η, s), 3.09 (2H, t, J = 6.6 Hz), 6.95 (1H, dd, J = 7.6, 1.4 Hz), 7.09 (1H, dt, J = 7.2, 0.8 Hz), 7.15—7.38 (3H, m) , 7.86 (2H, d, J = 8.4 Hz), 8.45 (1H, dd, J = 7.8, 0.8 Hz), 9.81 (1H, bs). Ή-NMR (CDCI3): δ 2.09-2.34 (4Η, m), 2.43 (3Η, s), 3.09 (2H, t, J = 6.6 Hz), 6.95 (1H, dd, J = 7.6, 1.4 Hz), 7.09 (1H, dt, J = 7.2, 0.8 Hz), 7.15-7.38 (3H, m), 7.86 (2H, d, J = 8.4 Hz), 8.45 (1H, dd, J = 7.8, 0.8 Hz), 9.81 (1H, bs).
元素分析値: C22H18N303S3F5として 計算値 (%) : C, 46.88; H, 3.22; N, 7.46; S, 17.07. Elementary analysis: as C 22 H 18 N 3 0 3 S 3 F 5 Calculated value (%): C, 46.88; H, 3.22; N, 7.46; S, 17.07.
実測値 ) : C, 46.92; H, 3.39; N, 7.33; S, 17.20. Found): C, 46.92; H, 3.39; N, 7.33; S, 17.20.
実施例 4-58 Example 4-58
N-U-ォキソ - 4-[(4,4,4-トリフルォロ- 3-トリフルォロメチルブチル)ス ルファニル ]-1, 2-ジヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]ベンゾチアジン 実施例 4-42 と同様の方法により、よう化イソァミルの代わりにメタン スルホン酸 4, 4, 4-トリフルォロ- 3-トリフルォロメチルブチル エステ ル (81.5 mg, 0.297 muiol) を用いて、 実施例 3 - 37で得られた N- U-ォ キソ -4-スルファニル _1, 2-ジヒド口- 3H -ィミダゾ [5, 1-c] [1, 4]ベンゾチ アジン- 3-ィリデン) -4-メチルベンゼンスルホンアミ ド (100 mg, 0.248 mmol) から題記化合物 (76.1 mg, 53%) を合成した。 酢酸:!;チル-ジェチ ルェ一テルから再結晶して融点 207-209°Cの暗赤色結晶を得た。 NU-oxo-4-[(4,4,4-trifluoro-3-trifluoromethylbutyl) sulfanyl] -1,2-dihydro-3H-imidazo [5, 1-c] [1,4] benzothiazine Example 4 In the same manner as in Example 4-42, 4,4,4-trifluoro-3-trifluoromethylbutyl methanesulfonate (81.5 mg, 0.297 muiol) was used instead of isoamyl iodide. N-U-oxo-4-sulfanyl_1,2-dihydropene-3H-imidazo [5,1-c] [1,4] benzothiazin-3-ylidene) -4-methyl obtained in 3-37 The title compound (76.1 mg, 53%) was synthesized from benzenesulfonamide (100 mg, 0.248 mmol). Acetic acid:!; Recrystallization from chill-ethyl ether gave dark red crystals with a melting point of 207-209 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3247, 1750, 1732, 1647, 1590, 1559, 1487 cm"1. Infrared absorption spectrum (IR) (in KBr): 3247, 1750, 1732, 1647, 1590, 1559, 1487 cm " 1 .
•H-NMR (CDC13) : <5 2.17-2.35 (2H, m) , 2.43 (3H, s), 2.95-3.22 (1H, m), 3.16 (2H, t, J = 7.6 Hz), 6.95 (1H, dd, J = 7.8, 1.6 Hz), 7.10 (1H, dt, J = 7.4, 1.4 Hz), 7.16-7.38 (3H, m) , 7.86 (2H, d, J = 8.0 Hz), 8.45 (1H, dd, J=8.8, 1.4 Hz), 9.78 (1H, bs). • H-NMR (CDC1 3) : <5 2.17-2.35 (2H, m), 2.43 (3H, s), 2.95-3.22 (1H, m), 3.16 (2H, t, J = 7.6 Hz), 6.95 ( 1H, dd, J = 7.8, 1.6 Hz), 7.10 (1H, dt, J = 7.4, 1.4 Hz), 7.16-7.38 (3H, m), 7.86 (2H, d, J = 8.0 Hz), 8.45 (1H , dd, J = 8.8, 1.4 Hz), 9.78 (1H, bs).
元素分析値: C22H17N303S3F6として Elementary analysis: as C 22 H 17 N 3 0 3 S 3 F 6
計算値 (%) : C, 45.43; H, 2.95; N, 7.23; S, 16.54. Calculated value (%): C, 45.43; H, 2.95; N, 7.23; S, 16.54.
実測値 (¾) : C, 45.80; H, 3.22; N, 7.26; S, 16.73. Found (¾): C, 45.80; H, 3.22; N, 7.26; S, 16.73.
実施例 4-59 Example 4-59
N-{4-[(3, 3-ジメチルブチル)スルファニル] -卜ォキソ -1, 2-ジヒ ド口 -3H-ィミダゾ [5, 1-c] [1,4〕ベンゾチアジン- 3-ィリデン }- 4-メチルベン ゼンスルホンアミド N- {4-[(3,3-Dimethylbutyl) sulfanyl] -toxo-1,2-dihydrido-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene}- 4-methylbenzenesulfonamide
実施例 4-42と同様の方法により、よう化ィソァミルの代わりにメタン スルホン酸 3, 3-ジメチルプチル エステル (536 mg, 2.97 mmol) を用い て、実施例 3-37で得られた N -(卜ォキソ -4-スルファニル -1, 2 -ジヒド口 _3H -ィミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3 -ィリデン) -4-メチルベン ゼンスルホンアミ ド(1.00g, 2.4811111101)から題記化合物(27.21^, 2.2%) を合成した。酢酸ェチル -n-へキサンから再結晶して融点 205- 207 の暗 赤色結晶を得た。 In the same manner as in Example 4-42, methanesulfonic acid 3,3-dimethylbutyl ester (536 mg, 2.97 mmol) was used in place of isosamil iodide to obtain the N-( Toloxo-4-sulfanyl-1,2-dihydric mouth The title compound (27.21 ^, 2.2%) was synthesized from _3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene) -4-methylbenzenesulfonamide (1.00 g, 2.4811111101). Recrystallization from ethyl acetate-n-hexane gave dark red crystals with a melting point of 205-207.
赤外吸収スペクトル(IR) (KBr中) リ: 3250, 2957, 2905; 1740, 1626, 1586, 1557, 1485 cm—1. Infrared absorption spectrum (IR) (in KBr): 3250, 2957, 2905; 1740, 1626, 1586, 1557, 1485 cm- 1 .
Ή-NMR (CDC13) : δ 0.965 (9H, s) , 1.52-1.66 (2Η, m) , 2.43 (3Η, s) , 2.93-3.04 (2Η, m) , 6.96 (1Η, dd. J = 7.8, 1.8 Hz) , 7.07 (1H, dt, J = 8.8, 1.4 Hz), 7.22 (1H, dt, J = 8.4, 1.8 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.0 Hz), 8.46 (1H, dt, J = 8.4, 1.0 Hz), 9.81 (1H, bs). 元素分析値: C23H25N303S3として Ή-NMR (CDC1 3): . Δ 0.965 (9H, s), 1.52-1.66 (2Η, m), 2.43 (3Η, s), 2.93-3.04 (2Η, m), 6.96 (1Η, dd J = 7.8 , 1.8 Hz), 7.07 (1H, dt, J = 8.8, 1.4 Hz), 7.22 (1H, dt, J = 8.4, 1.8 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.87 (2H, d , J = 8.0 Hz), 8.46 (1H, dt, J = 8.4, 1.0 Hz), 9.81 (1H, bs). Elemental analysis: As C 23 H 25 N 3 0 3 S 3
計算値 (%) : C, 56.65; H, 5.17; N, 8.62; S, 19.73. Calculated value (%): C, 56.65; H, 5.17; N, 8.62; S, 19.73.
実測値 ) : C, 56.59; H, 5.23; N, 8.26; S, 19.47. Obtained): C, 56.59; H, 5.23; N, 8.26; S, 19.47.
実施例 4 - 60 Example 4-60
N- [1, 6-ジォキソ- 4- (ィソペンチルスルファニル) - 1, 2, 7, 8-テトラヒド ロ- 3H, 6H-シクロペン夕 [b]イミダゾ [1,5- d] [1 , 4]チアジン -3-ィリデ 実施例 4- 42 と同様の方法により、 実施例 3- 39で得られた N-(l, 6 -ジ ォキソ _4_スルファニル - 1, 2, 7, 8-テトラヒドロ- 3H, 6H-シクロペン夕 [b] イミダゾ [1, 5-d] [1,4]チアジン- 3 -ィリデン) -4-メチルベンゼンスルホ ンアミド (540 mg, 1.38 mraol) から題記化合物 (527 mg, 80%) を合成 した。 酢酸ェチルから再結晶して融点 229- 23 Cの暗赤色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) レ: 3175, 2955, 2932, 2874, 1773, 1748, 1696, 1622, 1603, 1530 cm"1. N- [1,6-Dioxo-4- (isopentylsulfanyl)-1,2,7,8-tetrahydro-3H, 6H-cyclopentene [b] Imidazo [1,5-d] [1,4 ] Thiazine-3-ylide N- (l, 6-dioxo_4_sulfanyl- 1,2,7,8-tetrahydro-3H obtained in Example 3-39 by a method similar to that in Example 4-42. [B] Imidazo [1,5-d] [1,4] thiazine-3-ylidene) -4-methylbenzenesulfonamide (540 mg, 1.38 mraol) from the title compound (527 mg, 80% ) Was synthesized. Recrystallization from ethyl acetate gave dark red crystals of melting point 229-23C. Infrared absorption spectrum (IR) (in KBr) Re: 3175, 2955, 2932, 2874, 1773, 1748, 1696, 1622, 1603, 1530 cm " 1 .
Ή-NMR (CDCI3) : δ 0.928 (6Η, d, J = 6.2 Hz), 1.50-1.64 (2H, m) , 1.64-1.77 (1H, m), 2.43 (3H, s), 2.52-2.62 (2H, m) , 3.00 (2H, t, J = 7.6 Hz), 3.20-3.28 (2H, m) , 7.31 (2H, d, J = 7.6 Hz), 7.85 (2H, d, J = 8. Hz), 9.91 (1H, bs). Ή-NMR (CDCI3): δ 0.928 (6Η, d, J = 6.2 Hz), 1.50-1.64 (2H, m), 1.64-1.77 (1H, m), 2.43 (3H, s), 2.52-2.62 (2H , m), 3.00 (2H, t, J = 7.6 Hz), 3.20-3.28 (2H, m), 7.31 (2H, d, J = 7.6 Hz), 7.85 (2H, d, J = 8. Hz), 9.91 (1H, bs).
実施例 4 - 61 (a) -[6-ヒ ド ロキシ _4- (ィ ソペンチルスルフ ァニル) -卜ォキソ -1,2,7, 8 -テトラヒドロ- 3H, 6H-シクロペン夕 [b]ィミダゾ [l,5-d] [1,4] チアジン- 3-ィリデン] -4-メチルベンゼンスルホンアミド Example 4-61 (a)-[6-Hydroxy_4- (bisopentylsulfanyl) -toxo-1,2,7,8-tetrahydro-3H, 6H-cyclopentyl [b] imidazo [l, 5-d] [1 , 4] Thiazine-3-ylidene] -4-methylbenzenesulfonamide
(b) N-[l, 6-ジォキソ- 1, 2, 6, 7, 8, 8a-へキサヒドロ- 4- (ィソペンチルスル ファニル)- 3H, 5aH-シクロペン夕 [b]イミダゾ [1, 5- d] [1,4]チアジン - 3 - 水素化リチウムアルミニウム (264 mg, 5.56 mmol) のテトラヒドロフ ラン (10.0 ml) の懸濁液に、 実施例 4 - 60で得られた N- [1,6-ジォキソ -4 -(ィソペンチルスルファニル)-1, 2, 7, 8-テトラヒドロ - 3H, 6H-シク口 ペンタ [b]イミダゾ [1, 5-d] [1, 4]チアジン- 3-ィリデン ]_4-メチルベンゼ ンスルホンアミド (1.57 g, 3.29 mmol) のテトラヒドロフラン (90.0 ml) 溶液を氷冷下加えた。 反応混合物を室温で 5分かき混ぜた後、 過剰の水 素化リチウムアルミニウムを水で分解し、 1N塩酸を加えて酢酸ェチルで 抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィ一に付し、n-へキサン-酢酸 ェチル (10:1, v/v) で溶出して題記化合物(a) (24.0 mg, 1.5¾) を暗赤 色結晶として、 題記化合物(b) (429 mg, 27¾) を黄色結晶として得た。 (b) N- [l, 6-dioxo-1,2,6,7,8,8a-hexahydro-4- (isopentylsulfanyl) -3H, 5aH-cyclopentyl [b] imidazo [1,5-d ] A suspension of [1,4] thiazine-3- lithium aluminum hydride (264 mg, 5.56 mmol) in tetrahydrofuran (10.0 ml) was added to the suspension of N- [1,6] obtained in Example 4-60. -Dioxo-4-(isopentylsulfanyl) -1,2,7,8-tetrahydro-3H, 6H-cyclopenta [b] imidazo [1,5-d] [1,4] thiazine-3-ylidene ] _4-Methylbenzenesulfonamide (1.57 g, 3.29 mmol) in tetrahydrofuran (90.0 ml) was added under ice cooling. After stirring the reaction mixture at room temperature for 5 minutes, excess lithium aluminum hydride was decomposed with water, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (a) (24.0 mg, 1.5¾) as dark red crystals. The title compound (b) (429 mg, 27%) was obtained as yellow crystals.
題記化合物(a):酢酸ェチル -ジェチルエーテルから再結晶して融 点 156- 158°Cの暗赤色結晶を得た。 The title compound (a) was recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 156-158 ° C.
赤外吸収スペクトル (IR) (KBr 中) レ : 3700-2800, 3449, 2957, 2932, 2870, 1744, 1671, 1611, 1549, 1497 cm— Infrared absorption spectrum (IR) (in KBr) Re: 3700-2800, 3449, 2957, 2932, 2870, 1744, 1671, 1611, 1549, 1497 cm—
Ή-NMR (CDC13) : δ 0.927 (6Η, d, J = 6.6 Hz), 1.50-1.77 (3H, m) , 1.77-1.95 (1H, m), 2.42 (3H, s), 2.38-2.56 (1H, m) , 2.80-3.02 (1H, m), 2.97 (2H, t, J = 7.2 Hz), 3.07-3.26 (1H, m) , 4.62-4.76 (1H, m), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J-8.4 Hz), 9.70 (1H, bs). 元素分析値: C21H23N304S3'0.4H20として Ή-NMR (CDC1 3): δ 0.927 (6Η, d, J = 6.6 Hz), 1.50-1.77 (3H, m), 1.77-1.95 (1H, m), 2.42 (3H, s), 2.38-2.56 ( 1H, m), 2.80-3.02 (1H, m), 2.97 (2H, t, J = 7.2 Hz), 3.07-3.26 (1H, m), 4.62-4.76 (1H, m), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J-8.4 Hz), 9.70 (1H, bs). Elemental analysis: C 21 H 23 N 3 0 4 S 3 '0.4H 2 0
計算値 (%) : C, 51.81; Η, 5.34; Ν, 8.63; S, 19.76. Calculated value (%): C, 51.81; Η, 5.34; Ν, 8.63; S, 19.76.
実測値 ) : C, 51.72; Η, 5.19; Ν, 8.63; S, 19.68. Found): C, 51.72;;, 5.19; Ν, 8.63; S, 19.68.
題記化合物(b):酢酸ェチル -ジェチルエーテルから再結晶して融 点 172- 174°Cの黄色結晶を得た。 Title compound (b): Recrystallized from ethyl acetate-getyl ether and melted Yellow crystals at points 172-174 ° C were obtained.
赤外吸収スペク トル(IR) (KBr中) V: 3266, 2959, 2928, 2870, 1746, 1626, 1568 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3266, 2959, 2928, 2870, 1746, 1626, 1568 cm " 1 .
'Η -丽 R (CDC13) : (5 0.906 (6H, d, J = 6.2 Hz), 1. 8-1.75 (3H, m), 2.05-2.30 (1H, m) , 2.42 (3H, s), 2.30-2.62 (2H, m), 2.62-2.80 (1H, m), 3.00 (2H, t, J = 7.2 Hz), 3.70 (1H, d, J = 5.4 Hz), 4.90 (1H, dt, J = 9.6, 5.8 Hz), 7.31 (2H, d, J = 8.2 Hz), 7.88 (2H, d, J = 8.4 Hz), 9.67 (1H, bs). 'Eta -丽R (CDC1 3): (5 0.906 (6H, d, J = 6.2 Hz), 1. 8-1.75 (3H, m), 2.05-2.30 (1H, m), 2.42 (3H, s) , 2.30-2.62 (2H, m), 2.62-2.80 (1H, m), 3.00 (2H, t, J = 7.2 Hz), 3.70 (1H, d, J = 5.4 Hz), 4.90 (1H, dt, J = 9.6, 5.8 Hz), 7.31 (2H, d, J = 8.2 Hz), 7.88 (2H, d, J = 8.4 Hz), 9.67 (1H, bs).
元素分析値: C21H23N304S3として Elementary analysis: as C 21 H 23 N 3 0 4 S 3
計算値 (%) : C, 52.59; H, 5.25; N, 8.76; S, 20.06. Calculated value (%): C, 52.59; H, 5.25; N, 8.76; S, 20.06.
実測値 ) : C, 52.63; H, 5.38; N, 8.63; S, 20.03. Obtained): C, 52.63; H, 5.38; N, 8.63; S, 20.03.
実施例 4-62 Example 4-62
式 ( I X) において R2がイソペンチル基、、 R5および R6がともにメチ ル基、 R1 (^f— Z R15 (2が 02、 R 15が p-クロ口フエニル基)、 n が 1である化合物の合成 R 2 is an isopentyl group ,, R 5 and R 6 are both methylation group in formula (IX), R 1 (^ f- ZR 15 (2 is 0 2, R 15 is p- black port phenyl group), n is Synthesis of compound 1
実施例 3- 6で合成した化合物 (301 mg, 0.749 mmol) および炭酸カリ ゥム (155 mg, 1.12 mmol) を DMF (5 ml) に混和し、 6 0°Cで 5分間撹 拌した。 反応液によう化イソペンチル (0.109 ml, 0.824 mmol) を加え て約 3時間撹拌した。 反応液に 1N 塩酸 (1.5 ml) および 5 0 %メタノ —ル水 (10 ml) を加えて混和し、 室温で 3 0分間撹拌した。 生成レた沈 澱物を濾取し、 メタノール水で洗浄後、 乾燥して標題化合物の紫褐色粉 末 (318 mg、 0.674 mmol) を得た。 The compound synthesized in Example 3-6 (301 mg, 0.749 mmol) and potassium carbonate (155 mg, 1.12 mmol) were mixed with DMF (5 ml), and the mixture was stirred at 60 ° C for 5 minutes. Isopentyl iodide (0.109 ml, 0.824 mmol) was added to the reaction solution, and the mixture was stirred for about 3 hours. 1N Hydrochloric acid (1.5 ml) and 50% aqueous methanol (10 ml) were added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The resulting precipitate was collected by filtration, washed with aqueous methanol, and dried to give a purple-brown powder of the title compound (318 mg, 0.674 mmol).
匪 R (200 MHz, 重クロ口ホルム中) : δ 0.93 (6Η, d, J- 6.2 Hz), 1.56 (2H, q, J= 7.0 Hz), 1.67 (1H, sept, J= 6.6 Hz), 1.80 (3H, brs), 2.28 (3H, brs), 2.95 (2H, t, 7.6 Hz), 7. 6 (2H, d, J= 8.4 Hz), 7.91 (2H, d, J= 8. Hz), 9.65 (1H, brs). Marauder R (200 MHz, in double-mouthed holm): δ 0.93 (6Η, d, J- 6.2 Hz), 1.56 (2H, q, J = 7.0 Hz), 1.67 (1H, sept, J = 6.6 Hz), 1.80 (3H, brs), 2.28 (3H, brs), 2.95 (2H, t, 7.6 Hz), 7.6 (2H, d, J = 8.4 Hz), 7.91 (2H, d, J = 8 Hz) , 9.65 (1H, brs).
元素分析値: C19H22N303S3C1 として計算値: C, 48.34; H, 4.70; N, 8.90; S, 20.38; Cl, 7.51 (%), 実測値; C, 48.38; H, 4.56; N, 8.82; S, 20.35; C1,, 7.44 ( ) . 融点: 177. 5-178. 0°C Elemental analysis: Calculated as C 19 H 22 N 3 0 3 S 3 C1: C, 48.34; H, 4.70; N, 8.90; S, 20.38; Cl, 7.51 (%), Found; C, 48.38; H , 4.56; N, 8.82; S, 20.35; C1 ,, 7.44 (). Melting point: 177.5-178.0 ° C
以下に実施例 4_1〜4- 62で合成した化合物および同様にして合成した 化合物の一覧を示す。 The following is a list of compounds synthesized in Examples 4_1 to 4-62 and compounds synthesized in the same manner.
〔表 1 8〕(Table 18)
差替え用紙(規則 26) 〔表 1 9〕'Replacement form (Rule 26) (Table 19) '
差替え用紙 (M 26) 〔表 2 0〕'Replacement paper (M 26) [Table 20] '
差替え用紙 ( ) 〔表 2 1〕Replacement paper () (Table 21)
差替え ^ ¾ 26) 〔表 2 2〕Replacement ^ ¾ 26) (Table 22)
差替え用弒 (m ) 〔表 2 3〕For replacement 弒 (m) (Table 23)
差替え用鉞(規則 26) 〔表 2 4〕'Acet for replacement (Rule 26) (Table 24) '
差替え用鉞(細 ΰ2δ) 〔表 2 5〕'Acetate for replacement (fine ΰ2δ) (Table 25) '
差瞽ぇ用鉞(規則 26) 〔表 2 6〕Azeze for Acet (Rule 26) (Table 26)
轡ぇ ϋ欽 (β26^ 〔表 2 7〕ぇ ぇ ϋ Kin (β 26 ^ (Table 27)
差替え用紙(規則 26) 〔表 2 8〕Replacement form (Rule 26) (Table 28)
差替え用紙(規則 26) 〔表 2 9〕'Replacement form (Rule 26) (Table 29) '
差替え用鈸(規則 26) 〔表 3 0〕 For replacement (Rule 26) (Table 30)
差替え用紙 ) 〔表 3 1〕(Replacement paper) (Table 31)
差替え用弒(規則 2 〔表 3 2〕 For replacement (Rule 2 (Table 3 2)
羞替え用紙 wm) 〔表 3 3〕Shuffle paper wm) (Table 33)
差香え Λ 〔表 34〕差 (Table 34)
差替え用紙 (»J26) 〔表 3 5〕'Replacement paper (»J26) (Table 35) '
差簪ぇ用紙(規則 26) 〔表 3 6〕 Hairpin paper (Rule 26) (Table 36)
差替え用弒(規則 26) 〔表 3 7〕' For replacement (Rule 26) (Table 37) '
差替え用紙(規則 26) 〔表 3 8〕Replacement form (Rule 26) (Table 38)
盖替え用紙(ffi則 26) 〔表 3 9〕' Lid replacement paper (ffi-rule 26) (Table 39) '
差替え用觝 〔表 40〕' Replacement (Table 40) '
差替え用 # & 〔表 4 1〕Replacement # & (Table 41)
蓬替え用弒(規則 ) 〔表 4 2〕 (Rule) (Table 4 2)
差替え用殺 (¾ i]26) 〔表 4 3〕'Replacement killing (¾ i) 26 (Table 43) '
替 用 i¾II"J26) 〔表 4 4〕'Replacement i¾II "J26) (Table 4 4) '
差替え用飆(S126) 〔表 4 5〕'Replacement british (S126) (Table 45) '
差替え用弒(細 U26) 〔表 4 6〕For replacement 弒 (Thin U26) (Table 46)
差替え用紙(S¾26》 〔表 4 7〕Replacement paper (S¾26) (Table 47)
齄替え用紙 m ) 〔表 4 8〕(Replacement paper m) (Table 48)
差替え用鉞(|| (|26:) 〔表 4 9〕'Acet for replacement (|| (| 26 :) (Table 49) '
辇瞽ぇ用紙(a則 2β) 〔表 5 0〕'辇 Goze ぇ paper (a rule 2β) [Table 50] '
羞替え用弒 mm26) 〔表 5 1〕 Mm26) (Table 51)
差替え用弒 (M J26) 〔表 5 2〕For replacement 弒 (M J26) (Table 52)
差替え用鉞(S則 ) 〔表 5 3〕Acet for replacement (S-law) (Table 53)
差替え用弒 m 〔表 5 4〕'弒 m for replacement (Table 54) '
差替え用弒(規則 ) 〔表 5 5〕For replacement (rule) (Table 55)
差替え用弒 (¾IJ26) 〔表 5 6〕 For replacement 弒 (¾IJ26) (Table 56)
羞瞽ぇ用弒 (MM2Q) 〔表 5 7〕'For Goze (MM2Q) (Table 5 7) '
差替え用紙(規則 26〉 〔表 5 8〕Replacement paper (Rule 26) (Table 58)
差替え用飆 mu ) 〔表 5 9〕'(Replacement bri mu) (Table 59) '
差 え用紙(!·則 26) 〔表 6 0〕'Difference paper (! Rule 26) (Table 60) '
鍫替え角鉞(規則 26 〔表 6 1 )'鍫替example corner鉞(Rule 2 6 (Table 6 1) '
差簪ぇ用弒 (i¾|iJ2s; 〔表 6 2〕For hairpins (i | iJ2s; (Table 62)
差替え用紙 〔表 6 3〕Replacement paper (Table 6-3)
羞替え用弒 (MM26) 〔表 6 4〕'Replacement 弒 (MM26) (Table 6 4) '
蘧きえ用弒(規則 2 〔表 6 5〕蘧 for 弒 (Rule 2 (Table 65)
差替え用弒(鬼則 26: 〔表 6 6〕 For replacement 鬼 (Kiyoshi 26: (Table 66)
以下に実施例 4-63〜4-580で合成した化合物のデータを示す。 The data of the compounds synthesized in Examples 4-63 to 4-580 are shown below.
実施例 4 - 63 Example 4-63
Ή-NMR (CDC13, 200 MHz): δ 1.75 (3Η, d, J= 1.0 Hz), 2.15 (2H, quint, J= 7.0 Hz), 2.25 (3H, d, J- 1.2 Hz), 2.38 (3H, s), 3.08 (2H, t, J= 7.0 Hz), 3.13 (2H, t, J= 7.0 Hz), 7.25 (2H, d, J= 8.4 Hz), 7.49 (2H, tm, J= 7.8 Hz), 7.59 (1H, tm, J- 7 Hz), 7.84 (2H, d, J= 8.4 Hz), 7.97 (2H, dm, J= 8 Hz), 9.64 (1H, brs) . Ή-NMR (CDC1 3, 200 MHz): δ 1.75 (3Η, d, J = 1.0 Hz), 2.15 (2H, quint, J = 7.0 Hz), 2.25 (3H, d, J- 1.2 Hz), 2.38 ( 3H, s), 3.08 (2H, t, J = 7.0 Hz), 3.13 (2H, t, J = 7.0 Hz), 7.25 (2H, d, J = 8.4 Hz), 7.49 (2H, tm, J = 7.8) Hz), 7.59 (1H, tm, J-7 Hz), 7.84 (2H, d, J = 8.4 Hz), 7.97 (2H, dm, J = 8 Hz), 9.64 (1H, brs).
元素分析値: C25H25N304S3として計算値: C, 56.90; H, 4.78; N, 7.96; S, 18.23 (%)、 実測値: C, 56.68; H, 4.90; N, 7.95; S, 18.02 (%) . Elemental analysis: C 25 H 25 N 3 0 4 S 3 Calculated: C, 56.90; H, 4.78 ; N, 7.96; S, 18.23 (%), Found: C, 56.68; H, 4.90 ; N, 7.95; S, 18.02 (%).
実施例 4-64 Example 4-64
lE-MR (CDC13, 200 MHz): δ 1.43 (4Η, m), 1.5-1.75 (4H, m), 1.79 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.94 (2H, i, J= 7.0 Hz), 3.66 (2H, t, J= 6.3 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). lE-MR (CDC1 3, 200 MHz): δ 1.43 (4Η, m), 1.5-1.75 (4H, m), 1.79 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 1.2 Hz ), 2.42 (3H, s), 2.94 (2H, i, J = 7.0 Hz), 3.66 (2H, t, J = 6.3 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d , J = 8.0 Hz), 9.64 (1H, brs).
元素分析値: C2!H27N304S3として計算値: C, 52.37; H, 5.65; N, 8.72; S, 19.97 (%)、 実測値: C, 52.18; H, 5.62; N, 8.83; S, 20.05 (%) . Elemental analysis: Calculated as C 2 H 27 N 3 0 4 S 3:! C, 52.37; H, 5.65; N, 8.72; S, 19.97 (%), Found: C, 52.18; H, 5.62 ; N , 8.83; S, 20.05 (%).
実施例 4-65 Example 4-65
'H-NMR (CDC13, 200 MHz): δ 1.77 (3H, q, J= 1.2 Hz), 2.25 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 4.19 (2H, s), 7.24 (2H, d, J= 8.0 Hz), 7.32 (5H, s), 7.81 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1.77 (3H, q, J = 1.2 Hz), 2.25 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 4.19 (2H, s) , 7.24 (2H, d, J = 8.0 Hz), 7.32 (5H, s), 7.81 (2H, d, J = 8.0 Hz), 9.63 (1H, brs).
元素分析値: C22H21N303S3として計算値: C, 56.03; H, 4.49; N, 8.91; S, 20.40Elemental analysis: C 22 H 21 N 3 0 3 S 3 Calculated: C, 56.03; H, 4.49 ; N, 8.91; S, 20.40
(%)、 実測値: C, 55.90; H, 4.50; N, 8.82; S, 20.19 (%) . (%), Found: C, 55.90; H, 4.50; N, 8.82; S, 20.19 (%).
実施例 4-66 Example 4-66
'H-NMR (CDC13, 200 MHz): δ 1.75 (3Η, q, J= 1.0 Hz), 2.00 (2H, quint, J= 7.4 Hz), 2.25 (3H, q, J= 0.6 Hz), 2.39 (3H, s), 2.71 (2H, t, J= 7.4 Hz), 2.92 (2H, t, J= 7.3 Hz), 7.16 (2H, d, J= 8 Hz), 7.27 (5H, m), 7.85 (2H, d, J= 8.2 Hz), 9.64 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1.75 (3Η, q, J = 1.0 Hz), 2.00 (2H, quint, J = 7.4 Hz), 2.25 (3H, q, J = 0.6 Hz), 2.39 (3H, s), 2.71 (2H, t, J = 7.4 Hz), 2.92 (2H, t, J = 7.3 Hz), 7.16 (2H, d, J = 8 Hz), 7.27 (5H, m), 7.85 (2H, d, J = 8.2 Hz), 9.64 (1H, brs).
元素分析値: C24H25N303S3として計算値: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%)、 実測値: C, 57.63; H, 5.31; N, 8.11; S, 19.09 (%) . 実施例 4 - 67 Elemental analysis: C 24 H 25 N 3 0 3 S 3 Calculated: C, 57.69; H, 5.04 ; N, 8.41; S, 19.25 (%), Found: C, 57.63; H, 5.31 ; N, 8.11; S, 19.09 (%). Example 4-67
Ή-N R (CDC13, 200 MHz): δ 1.79 (3Η, brs), 2.25 (3Η, brs), 2.43 (3H, s), 2.73 (1H, d, J= 3.6Hz), 3.19 (1H, dd, J-13.6, 8.4 Hz), 3.32 (1H, dd, J=13.9, 4.0 Hz), 4.90 (1H, ddd, J= 8.8, 4.0, 3.6 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.36 (5H, s), 7.84 (2H, d, J= 8.4 Hz), 9.67 (1H, brs). Ή-NR (CDC1 3, 200 MHz): δ 1.79 (3Η, brs), 2.25 (3Η, brs), 2.43 (3H, s), 2.73 (1H, d, J = 3.6Hz), 3.19 (1H, dd , J-13.6, 8.4 Hz), 3.32 (1H, dd, J = 13.9, 4.0 Hz), 4.90 (1H, ddd, J = 8.8, 4.0, 3.6 Hz), 7.29 (2H, d, J = 8.4 Hz) , 7.36 (5H, s), 7.84 (2H, d, J = 8.4 Hz), 9.67 (1H, brs).
元素分析値: C23H23N304S3*H20として計算値: C, 53.16; H, 4.85; M, 8.09; S, 18.51Elemental analysis: C 23 H 23 N 3 0 4 S 3 * H 2 0 Calculated: C, 53.16; H, 4.85 ; M, 8.09; S, 18.51
(%), 実測値: C, 53.02; H, 4.79; N, 8.18; S, 18.53 (%) . (%), Found: C, 53.02; H, 4.79; N, 8.18; S, 18.53 (%).
実施例 4-68 Example 4-68
N- {8- [(シクロへキシルメチル)スルファニル ]-5, 6-ジメチル- 3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホ ン了ミド N- {8-[(Cyclohexylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene }-4-Methylbenzenesulfonamide
実施例 3- 4で合成した化合物 (3.28 g, 8.60mmol) および炭酸カリウム (1.78 g, 12.9 mmol) を DMF (50 ml) に混和し、 よう化シクロへキシルメチル (2.30g, 9.46mmol) を加えて 60°Cで 80分間撹拌した。 反応液に IN塩酸 (17.2ml) を加 えて混和し、室温で 30分間撹拌した。生成した沈澱物を 50%メタノール水(20 ml) に懸濁して濾取し、 メタノール水およびメタノールで洗浄後、 乾燥して標題 化合物の赤紫色粉末 (4.24 g、 8.60 mmol) を得た。 The compound synthesized in Example 3-4 (3.28 g, 8.60 mmol) and potassium carbonate (1.78 g, 12.9 mmol) were mixed with DMF (50 ml), and cyclohexylmethyl iodide (2.30 g, 9.46 mmol) was added. And stirred at 60 ° C. for 80 minutes. IN hydrochloric acid (17.2 ml) was added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The resulting precipitate was suspended in 50% aqueous methanol (20 ml), collected by filtration, washed with aqueous methanol and methanol, and dried to give a red-purple powder of the title compound (4.24 g, 8.60 mmol).
Ή-NMR (CDC13) 200 MHz): 6 0.97 (2H, brt, J= 11.5 Hz), 1.22 (3H, m), 1.60 (1H, m), 1.70 (3H, brs), 1.79 (3H, brq, J- 0.8 Hz), 1.87 (2H, m), 2.26 (3H, q, J= 0.8 Hz), 2.41 (3H, s), 2.81 (2H, t, J= 7.0 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.61 (1H, brs). Ή-NMR (CDC1 3) 200 MHz): 6 0.97 (2H, brt, J = 11.5 Hz), 1.22 (3H, m), 1.60 (1H, m), 1.70 (3H, brs), 1.79 (3H, brq , J- 0.8 Hz), 1.87 (2H, m), 2.26 (3H, q, J = 0.8 Hz), 2.41 (3H, s), 2.81 (2H, t, J = 7.0 Hz), 7.27 (2H, d , J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.61 (1H, brs).
元素分析値: C22H27N303S3として計算値: C, 55.32; H, 5.70; N, 8.80; S, 20.14Elemental analysis: C 22 H 27 N 3 0 3 S 3 Calculated: C, 55.32; H, 5.70 ; N, 8.80; S, 20.14
(%), 実測値: C, 55.21; H, 5.71; , 8.71; S, 20.10 (%) . (%), Found: C, 55.21; H, 5.71;, 8.71; S, 20.10 (%).
融点: 188.0-190.0°C Melting point: 188.0-190.0 ° C
実施例 4-69 Example 4-69
'H-NMR (CDC13, 200 MHz): d 0.30 (2H, brq, J= 5.6 Hz), 0.66 (2H, brq, J= 6.6 Hz), 1.04 (1H, m), 1.77 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.90 (2H, d, J= 7.4 Hz), 7.28 (2H, d, J= 8.4 Hz) , 7.87 (2H, d, J= 8.0 Hz), 9.64 (1H, brs) . 元素分析値: C19H21N303S3として計算値: C, 52.39; H, 4.86; N, 9.65; S, 22.08 (%)、 実測値: C, 52.72; H, 4.86; , 9.56; S, 21.91 (%) . 実施例 4 - 70 'H-NMR (CDC1 3, 200 MHz): d 0.30 (2H, brq, J = 5.6 Hz), 0.66 (2H, brq, J = 6.6 Hz), 1.04 (1H, m), 1.77 (3H, s) , 2.26 (3H, s), 2.42 (3H, s), 2.90 (2H, d, J = 7.4 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.87 (2H, d, J = 8.0 Hz) , 9.64 (1H, brs) elemental analysis: C 19 H 21 N 3 0 3 S 3 calculated:. C, 52.39; H, 4.86; N, 9.65; S, 22.08 (%), Found: C, 52.72; H, 4.86;, 9.56; S, 21.91 (%).
Ή-NMR (CDC13, 200 MHz): δ 1.60 (4H, m), 1.78 (3H, q, J= 1.2 Hz), 1.84 (2H, m), 2.12 (1H, quint, J= 7.3 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 2.94 (2H, t, J= 7.0 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.61 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.60 (4H, m), 1.78 (3H, q, J = 1.2 Hz), 1.84 (2H, m), 2.12 (1H, quint, J = 7.3 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 2.94 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.61 (1H, brs).
元素分析値: C21 5N303S3として計算値: C, 54.40; H, 5.43; N, 9.06; S, 20.75Elemental analysis: Calculated as C 2 15 N 3 0 3 S 3 : C, 54.40; H, 5.43; N, 9.06; S, 20.75
(%), 実測値: C, 54.23; H, 5.48; N, 8.95; S, 20.56 (%) . (%), Found: C, 54.23; H, 5.48; N, 8.95; S, 20.56 (%).
実施例 4-71 Example 4-71
-腿 (CDC13, 200 MHz): δ 1.79 (3H, brs), 2.26 (3H, brs), 2.43 (3H, s), 2.93 (2H, brdd, J= 8.0, 7.0 Hz), 3.15 (2H, dd, J= 8.0, 6.6 Hz), 6.98 (2H, t, J= 8.8 Hz), 7.15 (2H, dd, J- 8.0, 5.4 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.82 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). - thigh (CDC1 3, 200 MHz): δ 1.79 (3H, brs), 2.26 (3H, brs), 2.43 (3H, s), 2.93 (2H, brdd, J = 8.0, 7.0 Hz), 3.15 (2H, dd, J = 8.0, 6.6 Hz), 6.98 (2H, t, J = 8.8 Hz), 7.15 (2H, dd, J- 8.0, 5.4 Hz), 7.27 (2H, d, J = 8.0 Hz), 7.82 ( 2H, d, J = 8.0 Hz), 9.63 (1H, brs).
元素分析値: C23H22N303S3Fとして計算値: C, 54.85; H, 4.40; , 8.34; S, 19.10; F, 3.77 (%)、 実測値: C, 54.72; H, 4.30; N, 8.49; S, 18.92; F, 3.94 (%) . 実施例 4-72 Elemental analysis: Calculated as C 23 H 22 N 3 0 3 S 3 F: C, 54.85; H, 4.40;, 8.34; S, 19.10; F, 3.77 (%), found: C, 54.72; H, 4.30; N, 8.49; S, 18.92; F, 3.94 (%).
Ή-NMR (CDC13, 200 MHz): δ 1.67-1.94 (4H, i), 1.78 (3H, q, J= 0.8 Hz), 2.15 (2H, m), 2.26 (3H, q, J= 0.8 Hz), 2.41 (3H, s), 2.56 (1H, quint, J= 7.7 Hz), 3.02 (2H, d, J= 7.8 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.67-1.94 (4H, i), 1.78 (3H, q, J = 0.8 Hz), 2.15 (2H, m), 2.26 (3H, q, J = 0.8 Hz ), 2.41 (3H, s), 2.56 (1H, quint, J = 7.7 Hz), 3.02 (2H, d, J = 7.8 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.85 (2H, d , J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C2DH23N303S3として計算値: C, 53.43; H, 5.16; N, 9.35; S, 21.40Elemental analysis: C 2D H 23 N 3 0 3 S 3 Calculated: C, 53.43; H, 5.16 ; N, 9.35; S, 21.40
(%), 実測値: C, 53.25; H, 5.31; , 9.31; S, 21.32 (%) . (%), Found: C, 53.25; H, 5.31;, 9.31; S, 21.32 (%).
実施例 4 - 73 Example 4-73
Ή-NMR (CDC13, 200 MHz): 6 0.94 (3H, t, J= 7.2 Hz), 1.43 (2H, sixtet, J= 7.2 Hz), 1.59 (2H, sixtet, J= 7.0 Hz), 1.79 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.94 (2H, d, J= 7.3 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): 6 0.94 (3H, t, J = 7.2 Hz), 1.43 (2H, sixtet, J = 7.2 Hz), 1.59 (2H, sixtet, J = 7.0 Hz), 1.79 ( 3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 2.94 (2H, d, J = 7.3 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C19H23N303S3として計算値: C, 52.15; H, 5.30; N, 9.60; S, 21.98 (%)、 実測値: C, 52.22; H, 5.14; N, 9.58; S, 21.82 (%) · 実施例 4-74 Elemental analysis: C 19 H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30; N, 9.60; S, 21.98 (%), found: C, 52.22; H, 5.14; N, 9.58; S, 21.82 (%) Example 4-74
Ή-NMR (200 MHz, 重クロ口ホルム中) : δ 1.36 (3Η, t, J- 7.6 Hz), 1.78 (3H, brs), 2.27 (3H, brs), 2.42 (3H, s), 2.98 (2H, q, J= 7.5 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή-NMR (200 MHz, in double-hole form): δ 1.36 (3Η, t, J- 7.6 Hz), 1.78 (3H, brs), 2.27 (3H, brs), 2.42 (3H, s), 2.98 ( 2H, q, J = 7.5 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
実施例 4 - 75 Example 4-75
¾-NMR (CDCI3+TFA, 200 MHz): δ 1.76 (3H, brs), 2.26 (3H, brs), 2.41 (3H, s), 4.51 (2H, s), 7.29 (2H, d, J= 8.0 Hz), 7.53 (2H, brt, J= 7.6 Hz), 7.67 (1H, brt, J= 7.6 Hz), 7.88 (2H, d, J= 8.4 Hz), 7.96 (2H, dm, J= 7.8 Hz). 元素分析値: C23H21N304S3として計算値: C, 55.29; H, 4.24; N, 8.41; S, 19.25 (%), 実測値: C, 55.37; H, 4.21; , 8.26; S, 19.07 (%) . ¾-NMR (CDCI3 + TFA, 200 MHz): δ 1.76 (3H, brs), 2.26 (3H, brs), 2.41 (3H, s), 4.51 (2H, s), 7.29 (2H, d, J = 8.0 Hz), 7.53 (2H, brt, J = 7.6 Hz), 7.67 (1H, brt, J = 7.6 Hz), 7.88 (2H, d, J = 8.4 Hz), 7.96 (2H, dm, J = 7.8 Hz) . elemental analysis: calculated C 23 H 21 N 3 0 4 S 3: C, 55.29; H, 4.24; N, 8.41; S, 19.25 (%), Found: C, 55.37; H, 4.21 ;, 8.26; S, 19.07 (%).
実施例 4-76 Example 4-76
'H-NMR (CDC", 200 MHz): δ 1.75 (3Η, brq. J= 1.0 Hz), 2.24 (3H, brq. J- 1.0 Hz), 2.39 (3H, s), 2.72 (1H, brd, J= 6.0 Hz), 3.23 (2H, dd, J= 9.2, 7.0 Hz), 3.80 (3H, s), 4.02 (2H, d, J= 4.8 Hz), 4..17 (1H, m), 6.47 (1H, brs), 6.49 (1H, dm, J= 7.6 Hz), 6.55 (1H, dd, J= 9, 2 Hz), 7.20 (1H, t, J= 8.0 Hz), 7.25 (2H, d, J= 8.0 Hz), 7.83 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). 'H-NMR (CDC ", 200 MHz): δ 1.75 (3Η, brq. J = 1.0 Hz), 2.24 (3H, brq. J- 1.0 Hz), 2.39 (3H, s), 2.72 (1H, brd, J = 6.0 Hz), 3.23 (2H, dd, J = 9.2, 7.0 Hz), 3.80 (3H, s), 4.02 (2H, d, J = 4.8 Hz), 4..17 (1H, m), 6.47 (1H, brs), 6.49 (1H, dm, J = 7.6 Hz), 6.55 (1H, dd, J = 9, 2 Hz), 7.20 (1H, t, J = 8.0 Hz), 7.25 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.64 (1H, brs).
元素分析値: C25H27N306S3として計算値: C, 53.46; H, 4.85; N, 7.48; S, 17.13Elemental analysis: C 25 H 27 N 3 0 6 S 3 Calculated: C, 53.46; H, 4.85 ; N, 7.48; S, 17.13
(%)、 実測値: C, 53.41; H, 4.91; N, 7.28; S, 17.02 (%) . (%), Found: C, 53.41; H, 4.91; N, 7.28; S, 17.02 (%).
実施例 4-77 Example 4-77
¾-腿 (CDC13, 200 MHz): δ 1.79 (3Η, brs), 2.27 (3H, brs), 2.42 (3H, s), 2.42 (2H, brq, J= 7.1 Hz), 3.00 (2H, t, J- 7.5 Hz), 5.07 (1H, brs), 5.14 (1H, brd, J= 8.0 Hz), 5.79 (1H, ddt, J= 17.0, 10.4, 6.6 Hz), 7.28 (2H, d, J- 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). ¾- thigh (CDC1 3, 200 MHz): δ 1.79 (3Η, brs), 2.27 (3H, brs), 2.42 (3H, s), 2.42 (2H, brq, J = 7.1 Hz), 3.00 (2H, t , J- 7.5 Hz), 5.07 (1H, brs), 5.14 (1H, brd, J = 8.0 Hz), 5.79 (1H, ddt, J = 17.0, 10.4, 6.6 Hz), 7.28 (2H, d, J- 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C19H2iN303S3として計算値: C, 52.39; H, 4.86; N, 9.65; S, 22.08 (%) 実測値: C, 52.58; H, 4.83; N, 9.75; S, 22.11 (%) . Elemental analysis: C 19 H 2i N 3 0 3 S 3 Calculated: C, 52.39; H, 4.86 ; N, 9.65; S, 22.08 (%) Found: C, 52.58; H, 4.83 ; N, 9.75 ; S, 22.11 (%).
実施例 4-78 Example 4-78
Ή-NMR (CDC13, 200 MHz): δ 1.80 (3Η, brs), 2.26 (3H, brs), 2.43 (3H, s), 2.75 (2H, t, J- 7.3 Hz), 3.20 (2H, t, J= 7.3 Hz), 7.31 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). 元素分析値: C18H18N403S3として計算値: C, 49.75; H, 4.18; N, 12.89; S, 22.14 (%)、 実測値: C, 49.73; H, 4.00; N, 13.06; S, 22.14 (%) . Ή-NMR (CDC1 3, 200 MHz): δ 1.80 (3Η, brs), 2.26 (3H, brs), 2.43 (3H, s), 2.75 (2H, t, J- 7.3 Hz), 3.20 (2H, t , J = 7.3 Hz), 7.31 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.0 Hz), 9.64 (1H, brs). Elemental analysis: Calculated as C 18 H 18 N 4 0 3 S 3: C, 49.75; H, 4.18; N, 12.89; S, 22.14 (%), Found: C, 49.73; H, 4.00 ; N, 13.06; S, 22.14 (%).
実施例 4-79 Example 4-79
'H-NMR (CDC13, 200 MHz): δ 1.78 (3Η, s), 2.25 (3H, s), 2.41 (3H, s), 3.59 (2H, brs), 4.99 (1H, brs), 5.04 (1H, brs), 7.28 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.65 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1.78 (3Η, s), 2.25 (3H, s), 2.41 (3H, s), 3.59 (2H, brs), 4.99 (1H, brs), 5.04 ( 1H, brs), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.65 (1H, brs).
元素分析値: CigH21N303S3として計算値: C, 52.39; H, 4.86; , 9.65; S, 22.08Elemental analysis: Calculated C ig H 21 N 3 0 3 S 3: C, 52.39; H, 4.86;, 9.65; S, 22.08
(%), 実測値: C, 52.24; H, 4.88; , 9.71; S, 22.19 (%) . (%), Found: C, 52.24; H, 4.88;, 9.71; S, 22.19 (%).
実施例 4-80 Example 4-80
Ή-NM (CDC13, 200 MHz): δ 1.77 (3H, brq, J= 1.0 Hz), 2.25 (3H, brq, J= 1.2 Hz), 2.39 (3H, s), 4.05 (2H, s), 5.43 (1H, brs), 5.57 (1H, brs), 7.19 (2H, d, J= 8.0 Hz), 7.36 (5H, m), 7.76 (2H, d, J= 8.4 Hz), 9.60 (1H, brs). 元素分析値: C24H23N303S3として計算値: C, 57.92; H, 4.66; N, 8.44; S, 19.33 Ή-NM (CDC1 3, 200 MHz): δ 1.77 (3H, brq, J = 1.0 Hz), 2.25 (3H, brq, J = 1.2 Hz), 2.39 (3H, s), 4.05 (2H, s), 5.43 (1H, brs), 5.57 (1H, brs), 7.19 (2H, d, J = 8.0 Hz), 7.36 (5H, m), 7.76 (2H, d, J = 8.4 Hz), 9.60 (1H, brs .) elemental analysis: C 24 H 23 N 3 0 3 S 3 calculated: C, 57.92; H, 4.66 ; N, 8.44; S, 19.33
(%)、 実測値: C, 57.58; H, 4.61; N, 8.42; S, 19.37 (%) . (%), Found: C, 57.58; H, 4.61; N, 8.42; S, 19.37 (%).
実施例 4-81 Example 4-81
^-NMR (200MHz, CDC13) : δ 0.98 (4Η, ,brd, J= 13.2 Hz), 1.73 (3H, q, J= 0.8 Hz), 2.22 (3H, q, J= 0.8 Hz), 2.43 (3H, s), 3.26 (2H, s), 7.26 (7H, m), 7.82 (2H, d, J= 8.4 Hz), 9.56 (1H, brs). ^ -NMR (200MHz, CDC1 3) : δ 0.98 (4Η,, brd, J = 13.2 Hz), 1.73 (3H, q, J = 0.8 Hz), 2.22 (3H, q, J = 0.8 Hz), 2.43 ( 3H, s), 3.26 (2H, s), 7.26 (7H, m), 7.82 (2H, d, J = 8.4 Hz), 9.56 (1H, brs).
元素分析値: C25H25N303S3として計算値: C, 58.68; H, 4.92; N, 8.21; S, 18.80Elemental analysis: Calculated for C 25 H 25 N 3 0 3 S 3 : C, 58.68; H, 4.92; N, 8.21; S, 18.80
( ) 、 実測値: C, 58.74; H, 5.01; N, 7.97; S, 18.54 (%) . . (), Found: C, 58.74; H, 5.01; N, 7.97; S, 18.54 (%).
融点. 141.5-142.5°C 141.5-142.5 ° C
実施例 4-82 Example 4-82
N -(5, 6 -ジメチル- 3-ォキソ -8- {[3- (4-ピリジニル)プロピル]スルファニル }- 2, 3 - ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン)-4-メチルベンゼンス ルホンアミド N- (5,6-dimethyl-3-oxo-8-{[3- (4-pyridinyl) propyl] sulfanyl} -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine -Trilidene) -4-methylbenzenesulfonamide
(方法 1) 実施例 3-4で得た化合物 (300 mg, 0.786匪 ol)、 3 - (4-ピリジル) -1 - プロパノール (132 mg, 0.944讓 ol)、 CSA (228 rag, 0.983ιηπιο1)およびトリフエ ニルホスフィン(298 mg, 1.10 mmol)の THF (6.0 ml)懸濁液に室温で攪拌しなが ら DEAD (0.182 ml, 1.10腿 ol)を滴下し、 反応液を室温で 7.5時間、 50°C で 60 時間攪拌した。 この間反応液にトリフエニルホスフィン(213 mg, 0.786 mmol)お よび DEAD (0.130 ml, 0.786 mmol)を加えた。 酢酸ェチル(30 ml)を加えて希釈し、 2%炭酸ナトリゥム水溶液、 2%炭酸水素ナトリゥム水溶液および飽和食塩水で洗浄 し、乾燥、濃縮した。残渣をメタノールから結晶化して標題化合物 a25mg, 0.250 mmol)を紫色粉末として得た。 (Method 1) Compound obtained in Example 3-4 (300 mg, 0.786 marl ol), 3- (4-pyridyl) -1-propanol (132 mg, 0.944 sol ol), CSA (228 rag, 0.983ιηπιο1) DEAD (0.182 ml, 1.10 mL) was added dropwise to a suspension of THF and 6.0 ml of triphenylphosphine (298 mg, 1.10 mmol) with stirring at room temperature, and the reaction solution was allowed to stand at room temperature for 7.5 hours. 60 at ° C Stirred for hours. During this time, triphenylphosphine (213 mg, 0.786 mmol) and DEAD (0.130 ml, 0.786 mmol) were added to the reaction solution. The mixture was diluted with ethyl acetate (30 ml), washed with a 2% aqueous sodium carbonate solution, a 2% aqueous sodium hydrogen carbonate solution and saturated saline, dried and concentrated. The residue was crystallized from methanol to give the title compound (a25 mg, 0.250 mmol) as a purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.76 (3Η, brq, J= 0.8 Hz), 2.02 (2H, quint, J= 7.3 Hz), 2.26 (3H, brq, J= 0.8 Hz), 2.40 (3H, s), 2.73 (2H, t, J= 7.5 Hz), 2.92 (2H, t, J= 7.1 Hz), 7.10 (2H, d, J= 5.8 Hz), 7.28 (2H, d, J= 8.6 Hz), 7.85 (2H, d, J= 8.6 Hz), 8.52 (2H, dt, J= 6.2, 1.8 Hz). Ή-NMR (200MHz, CDC1 3 ): δ 1.76 (3Η, brq, J = 0.8 Hz), 2.02 (2H, quint, J = 7.3 Hz), 2.26 (3H, brq, J = 0.8 Hz), 2.40 (3H , s), 2.73 (2H, t, J = 7.5 Hz), 2.92 (2H, t, J = 7.1 Hz), 7.10 (2H, d, J = 5.8 Hz), 7.28 (2H, d, J = 8.6 Hz) ), 7.85 (2H, d, J = 8.6 Hz), 8.52 (2H, dt, J = 6.2, 1.8 Hz).
元素分析値: C23H24N403S3として計算値: C, 55.18; H, 4.83; N, 11.19; S, 19.21 ( ) 、 実測値: C, 55.15; H, 4.89; N, 11.12; S, 19.09 (%) . Elemental analysis: C 23 H 24 N 4 0 3 S 3 Calculated: C, 55.18; H, 4.83; N, 11.19; S, 19.21 (), found: C, 55.15; H, 4.89; N, 11.12 ; S, 19.09 (%).
(方法 ) 3 -(4 -ピリジル) -卜プロパノール(3.47 g, 24.8腿 ol)のクロ口ホルム (60 ml)溶液に室温で攪拌しながら -TsCl (4.87 g, 24.8 mmol)を数回に分けて 加え、反応液を室温で 4時間攪拌した。反応液にトリェチルァミン(1.03 ml, 7.43 匪 ol)を加え、反応液を室温で 1時間攪拌した。反応液を濃縮し、酢酸ェチル、 THF およびァセトニトリルで希釈した。 析出物を除去後、 母液を酢酸ェチルを加えて 希釈し、 飽和炭酸水素ナトリウム水溶液および飽和食塩水で洗浄し、 乾燥、 濃縮 した。 残渣をシリカゲル(50 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:1, 3:1 および 4:1) に付した。酢酸ェチルーへキサン('3:1 および 4:1)の画分 を濃縮乾固して P-トシル酸 3- (4-ビリジル)- 1-プロピル (純度 8 3.53 g, 10.2 mmol)を淡黄色粉末として得た。 (Method) To a solution of 3- (4-pyridyl) -topropanol (3.47 g, 24.8 tmol) in chloroform (60 ml) at room temperature while stirring -TsCl (4.87 g, 24.8 mmol) was divided into several portions. The reaction was stirred at room temperature for 4 hours. Triethylamine (1.03 ml, 7.43 ol) was added to the reaction solution, and the reaction solution was stirred at room temperature for 1 hour. The reaction was concentrated and diluted with ethyl acetate, THF and acetonitrile. After removing the precipitate, the mother liquor was diluted by adding ethyl acetate, washed with a saturated aqueous solution of sodium hydrogencarbonate and brine, dried and concentrated. The residue was chromatographed on silica gel (50 g) (ethyl acetate-hexane, 1: 1, 3: 1 and 4: 1). The fractions of ethyl acetate-hexane ('3: 1 and 4: 1) were concentrated to dryness to give 3- (4-viridyl) -1-propyl P-tosylate (purity 8 3.53 g, 10.2 mmol) as pale yellow. Obtained as a powder.
'H-N R (300MHz, CDC13): δ 1.99 (2H, m), 2.47 (3H, s), 2.67 (2H, t, J= 7.7 Hz), 4.03 (2H, t, J= 7.1 Hz), 7.01 (2H, d, J= 6.2 Hz), 7.36 (2H, d, J= 8.0 Hz), 7.79 (2H, d, J= 8.4 Hz), 8.46 (2H, dt, J= 6.2, 1.8 Hz). 'HN R (300MHz, CDC1 3 ): δ 1.99 (2H, m), 2.47 (3H, s), 2.67 (2H, t, J = 7.7 Hz), 4.03 (2H, t, J = 7.1 Hz), 7.01 (2H, d, J = 6.2 Hz), 7.36 (2H, d, J = 8.0 Hz), 7.79 (2H, d, J = 8.4 Hz), 8.46 (2H, dt, J = 6.2, 1.8 Hz).
実施例 3- 4で得た化合物 (260 mg, 0.682 mmol)および炭酸カリウム (141 mg, 1.02 mmol) の DMF (4.0 ml) の懸濁液に攪拌しながら上記 p-トシル酸 3- (4-ピリ ジル) -卜プロピル(純度 84%, 308 mg, 0.886 mmol)を加え、 反応液を 40°Cで 12 時間、 さらに 80でで 2.5時間攪拌した。 反応液に酢酸ェチルー THF (2:1, 0 ml) を加え、 炭酸水素ナトリウム水溶液および飽和食塩水で洗浄した。 水層を酢酸ェ チル (50ml)で抽出した。 有機層を合わせて、 乾燥、 濃縮し、 残渣をメタノールで 希釈した。 析出物を濾取し、 メタノールで洗浄、 乾燥して標題化合物 (216 mg, 0.431 歷 ol)を濃紫色粉末として得た。 While stirring a suspension of the compound obtained in Example 3-4 (260 mg, 0.682 mmol) and potassium carbonate (141 mg, 1.02 mmol) in DMF (4.0 ml), the above p-tosylate 3- (4- Pyridyl) -propyl (purity 84%, 308 mg, 0.886 mmol) was added, and the reaction mixture was stirred at 40 ° C for 12 hours and further at 80 at 2.5 hours. Ethyl acetate-THF (2: 1, 0 ml) was added to the reaction mixture, and the mixture was washed with an aqueous sodium hydrogen carbonate solution and saturated saline. Acetic acid Extracted with chill (50 ml). The organic layers were combined, dried and concentrated, and the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (216 mg, 0.431 mol) as a dark purple powder.
元素分析: C23H24N403S3として計算値: C, 55.18; H, 4.83; N, 11.19; S, 19.21»)· 実測値: C, 55.10; H, 4.71; N, 11.01; S, 19.13(%). Elemental analysis: C 23 H 24 N 4 0 3 S 3 Calculated: C, 55.18; H, 4.83 ; N, 11.19; S, 19.21 ») · Found: C, 55.10; H, 4.71 ; N, 11.01; S, 19.13 (%).
融点. 185.0- 186.0°C Melting point.185.0- 186.0 ° C
実施例 4-83 Example 4-83
¾-NMR (200MHz, CDC13) : δ 1.76 (3Η, q, J= 1.2 Hz), 2.01 (2H, quint, J= 7.5 Hz), 2.26 (3H, q, J= 1.0 Hz), 2.40 (3H, s), 2.73 (2H, t, J= 7.5 Hz), 2.93 (2H, t, J= 7.3 Hz), 7.23 (1H, m), 7.28 (2H, d, J= 8.4 Hz), 7.49 (1H, brd, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 8.47 (2H, m). ¾-NMR (200MHz, CDC1 3 ): δ 1.76 (3Η, q, J = 1.2 Hz), 2.01 (2H, quint, J = 7.5 Hz), 2.26 (3H, q, J = 1.0 Hz), 2.40 (3H , s), 2.73 (2H, t, J = 7.5 Hz), 2.93 (2H, t, J = 7.3 Hz), 7.23 (1H, m), 7.28 (2H, d, J = 8.4 Hz), 7.49 (1H , brd, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 8.47 (2H, m).
元素分析値: C23H24N403S3として計算値: C, 55.18; H, 4.83; N, 11.19; S, 19.21Elemental analysis: C 23 H 24 N 4 0 3 S 3 Calculated: C, 55.18; H, 4.83 ; N, 11.19; S, 19.21
(%) 、 実測値: C, 55.16; H, 4.85; N, 11.06; S, 18.99 (%) . (%), Found: C, 55.16; H, 4.85; N, 11.06; S, 18.99 (%).
融点. 191.5-193.0°C Melting point.191.5-193.0 ° C
実施例 4-84 Example 4-84
N- {5, 6-ジメチル- 8- [ (4-メチルペンチル)スルファニル] -3-ォキソ -2, 3-ジヒドロ - 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホンァ ミド . N- {5,6-dimethyl-8-[(4-methylpentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene }-4-methylbenzenesulfonamide.
実施例 3 - 4で合成した化合物 (2.50g, 6.56讓01) および炭酸カリウム (1.36 g, 9.84 mmol) を DMF (40 ml) に混和し、 80°Cで 5分間撹拌した。 反応液によう 化イソへキシル (1.53 ml, 7.21 mmol) を加えて 50分間撹拌した。 反応液に 1N 塩酸 (13.1ml) を加えて混和し、 室温で 30分間撹拌した。 生成した沈澱物を 5 0%メタノ一ル水 (20ml) に懸濁して濾取し、 メタノール水およびメタノールで 洗浄後、 乾燥して表題化合物の赤紫色粉末 (2.94 g、 6.31 腿 ol) を得た。 The compound synthesized in Example 3-4 (2.50 g, 6.56 amino acid) and potassium carbonate (1.36 g, 9.84 mmol) were mixed with DMF (40 ml), and the mixture was stirred at 80 ° C for 5 minutes. Isohexyl iodide (1.53 ml, 7.21 mmol) was added to the reaction solution, and the mixture was stirred for 50 minutes. 1N Hydrochloric acid (13.1 ml) was added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The resulting precipitate was suspended in 50% aqueous methanol (20 ml), collected by filtration, washed with methanol water and methanol, and dried to obtain a red-purple powder of the title compound (2.94 g, 6.31 tmol). Was.
Ή-NMR (200MHz, CDC13) : δ 0.89 (6Η, d, J= 6.6 Hz), 1.27 (2H, dt, J= 9.2, 6.2 Hz), 1.56 (1H, quint, J= 6.6 Hz), 1.67 (2H, m), 1.79 (3H, q, J= 0.6 Hz), 2.26 (3H, q, J= 1.0 Hz), 2.41 (3H, s) , 2.91 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.89 (6Η, d, J = 6.6 Hz), 1.27 (2H, dt, J = 9.2, 6.2 Hz), 1.56 (1H, quint, J = 6.6 Hz), 1.67 (2H, m), 1.79 (3H, q, J = 0.6 Hz), 2.26 (3H, q, J = 1.0 Hz), 2.41 (3H, s), 2.91 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C21H27N303S3として計算値: C, 54.17; H, 5.84; N, 9.02; S, 20.66 {%) 、 実測値: C, 54.25; H, 5.71; N, 8.90; S, 20.43 ( ) . 融点. 166.0-168.5 : Elemental analysis: Calculated for C 21 H 27 N 3 0 3 S 3 : C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), Found: C, 54.25; H, 5.71; N, 8.90; S, 20.43 (). Melting point. 166.0-168.5:
実施例 4-85 Example 4-85
'H-NMR (200MHz, CDC13) : δ 1.27 (2H, m), 1.4-1.7 (10H, m), 1.79 (3H, brs), 1.8 (3H, m), 2.27 (3H, brs), 2.42 (3H, s), 2.73 (2H, d, J= 6.2 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.2 Hz), 9.63 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 1.27 (2H, m), 1.4-1.7 (10H, m), 1.79 (3H, brs), 1.8 (3H, m), 2.27 (3H, brs), 2.42 (3H, s), 2.73 (2H, d, J = 6.2 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.2 Hz), 9.63 (1H, brs).
元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56Elemental analysis: C 23 H 29 N 3 0 3 S 3 Calculated: C, 56.18; H, 5.94 ; N, 8.55; S, 19.56
(%) 、 実測値: C, 55.96; H, 5.83; N, 8.51; S, 19.54 (%) . (%), Found: C, 55.96; H, 5.83; N, 8.51; S, 19.54 (%).
融点. 179.5-180.0 Melting point.179.5-180.0
実施例 4-86 Example 4-86
Ή-NMR (200MHz, CDC13) : δ 1.74 (3Η, brs), 2.24 (3H, brs), 2.43 (3H, s), 4.13 (2H, s), 7.28 (2H, d, J= 8.4 Hz), 7.28 (2H, d, J= 6.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 8.58 (2H, d, J= 6.4 Hz). Ή-NMR (200MHz, CDC1 3 ): δ 1.74 (3Η, brs), 2.24 (3H, brs), 2.43 (3H, s), 4.13 (2H, s), 7.28 (2H, d, J = 8.4 Hz) , 7.28 (2H, d, J = 6.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 8.58 (2H, d, J = 6.4 Hz).
元素分析値: C21H2。N403S3として計算値: C, 53.37; H, 4.27; N, 11.85; S, 20.35 (%) 、 実測値: C, 53.16; H, 4.22; N, 11.81; S, 20.29 (%) . Elemental analysis: C 21 H 2. Calculated for N 4 0 3 S 3 : C, 53.37; H, 4.27; N, 11.85; S, 20.35 (%), found: C, 53.16; H, 4.22; N, 11.81; S, 20.29 (%) .
融点. 189.4- 189.7°C Melting point.189.4- 189.7 ° C
実施例 4-87 Example 4-87
Ή-NMR (200MHz, CDC13) : δ 1.80 (3Η, q, J= 1.2 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.43 (3H, s), 3.07 (1H, brdd, J= 7.2, 2. Hz), 3.11 (1H, brd, J- 7.2 Hz), .3.19 (1H, dd, J= 6.2, 1.4 Hz), 3.22 (1H, dd, J= 6.2, 2.6 Hz), 7.28 (2H, d, J= 8.2 Hz), 7.36 (2H, d, J= 8.8 Hz), 7.81 (2H, d, J= 8.3 Hz), 8.16 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.80 (3Η, q, J = 1.2 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.43 (3H, s), 3.07 (1H, brdd, J = 7.2, 2.Hz), 3.11 (1H, brd, J- 7.2 Hz), 3.19 (1H, dd, J = 6.2, 1.4 Hz), 3.22 (1H, dd, J = 6.2, 2.6 Hz), 7.28 ( 2H, d, J = 8.2 Hz), 7.36 (2H, d, J = 8.8 Hz), 7.81 (2H, d, J = 8.3 Hz), 8.16 (2H, d, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C23 2N405S3として計算値: C, 52.06; H, 4.18; N, 10.56; S, 18.13 (%) 、 実測値: C, 51.88; H, 4.24; N, 10.53; S, 17.87 (%) . Elemental analysis: C 23 2 N 4 0 5 S 3 Calculated: C, 52.06; H, 4.18 ; N, 10.56; S, 18.13 (%), Found: C, 51.88; H, 4.24 ; N, 10.53 ; S, 17.87 (%).
融点. 192.0- 192.4。C Melting point. 192.0-192.4. C
実施例 4 - 88 Example 4-88
Ή-NMR (200MHz, CDC13) : δ 0.88 (6Η, d, J= 6.6 Hz), 1.20 (2H, brquint, J= 6.8 Hz), 1.39 (2H, m), 1.50 (1H, sept, J= 7.2 Hz), 1.60 (2H, quint, J= 10.6 Hz), 1.79 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.41 (3H, s), 2.93 (2H, t, J= 7.4 Hz), 7.28 (2H, d, J- 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). 元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05 Ή-NMR (200MHz, CDC1 3 ): δ 0.88 (6Η, d, J = 6.6 Hz), 1.20 (2H, brquint, J = 6.8 Hz), 1.39 (2H, m), 1.50 (1H, sept, J = 7.2 Hz), 1.60 (2H, quint, J = 10.6 Hz), 1.79 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 0.8 Hz), 2.41 (3H, s), 2.93 (2H , t, J = 7.4 Hz), 7.28 ( 2H, d, J- 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs) Elemental analysis:. C 22 H 29 N 3 0 3 S Calculated as 3 : C, 55.09; H, 6.09; N, 8.76; S, 20.05
(%) 、 実測値: C, 55.00; H, 6.10; N, 8.76; S, 20.11 (%) . (%), Found: C, 55.00; H, 6.10; N, 8.76; S, 20.11 (%).
融点. 150.5-150.7°C Melting point.150.5-150.7 ° C
実施例 4 - 89 Example 4-89
Ή-NMR (200MHz, CDC13) : δ 1.79 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 3.02 (2H, dd, J- 7.9, 7.1 Hz), 3.20 (2H, dd, J= 8.0, 7.0 Hz), 3.92 (3H, s), 7.27 (4H, brs), 7.81 (2H, d, J- 8.0 Hz), 7.98 (2H, d, J= 7.9 Hz), 9.62 (1H, brs). 元素分析値: C25H25N305S3として計算値: C, 55.23; H, 4.63; N, 7.73; S, 17.69 (%) 、 実測値: C, 55.08; H, 4.79; N, 7.69; S, 17.47 (%) . Ή-NMR (200MHz, CDC1 3 ): δ 1.79 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 3.02 (2H, dd, J- 7.9, 7.1 Hz), 3.20 (2H, dd, J = 8.0, 7.0 Hz), 3.92 (3H, s), 7.27 (4H, brs), 7.81 (2H, d, J- 8.0 Hz), 7.98 (2H, d, J = 7.9 Hz), 9.62 ( 1H, brs). Elemental analysis: C 25 H 25 N 3 0 5 S 3 Calculated: C, 55.23; H, 4.63; N, 7.73; S, 17.69 (%), found: C, 55.08; H , 4.79; N, 7.69; S, 17.47 (%).
融点. 191.0-191.5°C Melting point.191.0-191.5 ° C
実施例 4-90 Example 4-90
Ή-N R (200MHz, CDC13) : δ 1.78 (3Η, brs), 2.26 (3H, brs), 2.42 (3H, s), 2.94 (2H, dd, J= 7.8, 7.1 Hz), 3.16 (2H, dd, J- 7.9, 7.4 Hz), 7.12. (2H, d, J= 8.3 Hz), 7.26 (2H, d, J二 8.3 Hz), 7.26 (2H, d, J= 8.2 Hz), 7.81 (2H, d, J= 8.2 Hz), 9.63 (1H, brs). Ή-NR (200MHz, CDC1 3 ): δ 1.78 (3Η, brs), 2.26 (3H, brs), 2.42 (3H, s), 2.94 (2H, dd, J = 7.8, 7.1 Hz), 3.16 (2H, dd, J- 7.9, 7.4 Hz), 7.12. (2H, d, J = 8.3 Hz), 7.26 (2H, d, J 8.3 Hz), 7.26 (2H, d, J = 8.2 Hz), 7.81 (2H , d, J = 8.2 Hz), 9.63 (1H, brs).
元素分析値: C23H22N303S3C1として計算値: C, 53.12; H, 4.26; , 8.08; S, 18.50; CI, 6.82 (%) 、 実測値: C, 52.83; H, 4.55; N, 8.21; S, 18.29; CI, 6.82 (%) . 融点. 189.0-190.5°C Elemental analysis: Calculated as C 23 H 22 N 3 0 3 S 3 C1: C, 53.12; H, 4.26;, 8.08; S, 18.50; CI, 6.82 (%), Found: C, 52.83; H, 4.55; N, 8.21; S, 18.29; CI, 6.82 (%). Melting point. 189.0-190.5 ° C
実施例 4-91 Example 4-91
Ή-NMR (200MHz, CDCl3-DMSO-d64:1) : δ 1.80 (3H, s), 2.25 (3H, s), 2.43 (3H, s), 3.03 (2H, dd, J= 7.8, 7.2 Hz), 3.22 (2H, dd, J= 7.7, 7.2 Hz), 7.27 (2H, d, J= 7.8 Hz), 7.28 (2H, d, J= 7.7 Hz), 7.82 (2H, d, J= 8.1 Hz), 7.96 (2H, d, J= 7.9 Hz), 10.84 (1H, br). Ή-NMR (200 MHz, CDCl 3 -DMSO-d 6 4: 1): δ 1.80 (3H, s), 2.25 (3H, s), 2.43 (3H, s), 3.03 (2H, dd, J = 7.8, 7.2 Hz), 3.22 (2H, dd, J = 7.7, 7.2 Hz), 7.27 (2H, d, J = 7.8 Hz), 7.28 (2H, d, J = 7.7 Hz), 7.82 (2H, d, J = 8.1 Hz), 7.96 (2H, d, J = 7.9 Hz), 10.84 (1H, br).
元素分析値: C24H23N305S3として計算値: C, 54.42; H, 4.38; N, 7.93; S, 18.16 (%) 、 実測値: C, 54.65; H, 4.42; N, 7.71; S, 18.03 ( ) . Elemental analysis: C 24 H 23 N 3 0 5 S 3 Calculated: C, 54.42; H, 4.38 ; N, 7.93; S, 18.16 (%), Found: C, 54.65; H, 4.42 ; N, 7.71; S, 18.03 ().
融点. 236.0-236.5°C Melting point.236.0-236.5 ° C
実施例 4-92 Example 4-92
Ή-NMR (200MHz, CDC13) : <3 1.78 (3H, brq, J= 0.8 Hz), 2.26 (3H, brq, J= 1.2 Hz), 2.34 (3H, s), 2.43 (3H, s), 2.93 (2H, dd, J- 8.2, 6.8 Hz), 3.15 (2H, dd, J= 8.8, 6.2 Hz), 7.09 (2H, d, J= 8.8 Hz), 7.11 (2H, d, J= 8.8 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.64 (1H, brs) . Ή-NMR (200MHz, CDC1 3 ): <3 1.78 (3H, brq, J = 0.8 Hz), 2.26 (3H, brq, J = 1.2 Hz), 2.34 (3H, s), 2.43 (3H, s), 2.93 (2H, dd, J- 8.2, 6.8 Hz), 3.15 (2H, dd, J = 8.8, 6.2 Hz), 7.09 (2H, d , J = 8.8 Hz), 7.11 (2H, d, J = 8.8 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C24H25N303S3として計算値: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%) 、 実測値: C, 57.54; H, 4.93; N, 8.32; S, 19.09 (%) . Elemental analysis: C 24 H 25 N 3 0 3 S 3 Calculated: C, 57.69; H, 5.04 ; N, 8.41; S, 19.25 (%), Found: C, 57.54; H, 4.93 ; N, 8.32; S, 19.09 (%).
融点. 179.0-180.0°C Melting point.179.0-180.0 ° C
実施例 4-93 Example 4-93
Ή-NMR (200MHz, CDC13) : δ 1.79 (3Η, brq, J= 0.8 Hz), 2.26 (3H, brq, J- 1.2 Hz), 2.42 (3H, s), 3.03 (2H, dd, J= 7.2, 6.6 Hz), 3.19 (2H, dd, J= 7.0, 6.6 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.31 (2H, d, J= 8.8 Hz), 7.56 (2H, d, J= 8.6 Hz), 7.82 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.79 (3Η, brq, J = 0.8 Hz), 2.26 (3H, brq, J- 1.2 Hz), 2.42 (3H, s), 3.03 (2H, dd, J = 7.2, 6.6 Hz), 3.19 (2H, dd, J = 7.0, 6.6 Hz), 7.27 (2H, d, J = 8.0 Hz), 7.31 (2H, d, J = 8.8 Hz), 7.56 (2H, d, J = 8.6 Hz), 7.82 (2H, d, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C24H22N303S3F3として計算値: C, 52.07; H, 4.01; , 7.59; S, 17.38; F, 10.29 (%) 、 実測値: C, 51.79; H, 4.05; N, 7.39; S, 17.35; F, 10.56 (%) . 融点. 193.0- 194.0°C Elemental analysis: Calculated for C 24 H 22 N 3 0 3 S 3 F 3 : C, 52.07; H, 4.01;, 7.59; S, 17.38; F, 10.29 (%), found: C, 51.79; H , 4.05; N, 7.39; S, 17.35; F, 10.56 (%). Melting point. 193.0-194.0 ° C
実施例 4-94 Example 4-94
N - {8 - [ (シクロォクチルメチル)スルファニル] -5, 6-ジメチル- 3-ォキソ- 2, 3-ジヒ ドロ- 1H-イミダゾ [5,1-c] [1,4]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホ ンアミド N- {8-[(cyclooctylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1 -Yylidene}-4-methylbenzenesulfonamide
実施例 3- 4で得た化合物 (1.85g, 4.85腿 ol)、 ョードメチルシクロオクタン(純 度 90%, 1.63 g, 5.82匪 ol)および炭酸カリウム (1.01 g, 7.27 mmol) の DMF (30 ml) の懸濁液を室温で 12時間攪拌した。 反応液に 1N塩酸 (9.7 ml)を加え、 水 (10ml)およびメタノール(10ml)で希釈し、 室温で 60分間攪拌した。 析出物を濾 取し、 メ夕ノ一ルー水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物 (2.27 g, 4.49画 ol, 92.5%)を赤紫色粉末として得た。 DMF (1.05 g, 7.27 mmol) of the compound obtained in Example 3-4 (1.85 g, 4.85 t ol), eodomethyl cyclooctane (purity 90%, 1.63 g, 5.82 ol) and potassium carbonate (1.01 g, 7.27 mmol) 30 ml) was stirred at room temperature for 12 hours. The reaction mixture was diluted with 1N hydrochloric acid (9.7 ml), diluted with water (10 ml) and methanol (10 ml), and stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol (1: 1) and methanol, and dried to give the title compound (2.27 g, 4.49 fraction, 92.5%) as a red-purple powder.
Ή-NMR (300MHz, CDC13): δ 1.38 (2Η, m), 1.52 (6H, m), 1.60-1.80 (7H, m), 1.78 (3H, q, J= 0.9 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 2.81 (2H, d, J= 6.6 Hz), 7.26 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8^ Hz), 9.58 (1H, brs). 元素分析値: C24H31N303S3として計算値: C, 57.00; H, 6.18; N, 8.31; S, 19.02 (%) 、 実測値: C, 56.89; H, 6.26; N, 8.17; S, 18.88 (%) · 融点. 176.0- 178.0°C Ή-NMR (300MHz, CDC1 3 ): δ 1.38 (2Η, m), 1.52 (6H, m), 1.60-1.80 (7H, m), 1.78 (3H, q, J = 0.9 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 2.81 (2H, d, J = 6.6 Hz), 7.26 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8 ^ Hz) .), 9.58 (1H, brs ) elemental analysis: calculated C 24 H 31 N 3 0 3 S 3: C, 57.00; H, 6.18; N, 8.31; S, 19.02 (%), Found: C , 56.89; H, 6.26; N, 8.17; S, 18.88 (%) Melting point.176.0- 178.0 ° C
実施例 4-95 ' Example 4-95 '
融点 168- 170T: EtOAc- Et20. 赤外吸収スペクトル(IR) (KBr中) ソ: 3247, 2975, 2930, 2868, 1748, 1721, 1663, 1613, 1557 cnf1. MMR (CDC13) : δ 1.21 (3H, t, J-7.0 Hz), 1.79 (3H, s), 2.26 (3H, d, J=1.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J=6.6 Hz), 3.52 (2H, q, J=7.0 Hz), 3.64 (2H, q, J=6.6 Hz), 7.28 (2H, d, J-8.4 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs) . Mp 168- 170T: EtOAc- Et 2 0. The infrared absorption spectrum (IR) (in KBr) Seo:. 3247, 2975, 2930, 2868, 1748, 1721, 1663, 1613, 1557 cnf 1 MMR (CDC1 3): δ 1.21 (3H, t, J-7.0 Hz), 1.79 (3H, s), 2.26 (3H, d, J = 1.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J = 6.6 Hz) , 3.52 (2H, q, J = 7.0 Hz), 3.64 (2H, q, J = 6.6 Hz), 7.28 (2H, d, J-8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: CI9H23N304S3として計算値: C, 50.31; H, 5.11; N, 9.26; S, 21.21 ( ), 実測値: C, 50.19; H, 4.95; N, 9.23; S, 21.23 (%) . Elemental analysis: C I9 H 23 N 3 0 4 S 3 Calculated: C, 50.31; H, 5.11 ; N, 9.26; S, 21.21 (), Found: C, 50.19; H, 4.95 ; N, 9.23 ; S, 21.23 (%).
実施例 4 - 96 Example 4-96
Ή-NMR (200MHz, CDC13) : δ 1.78 (3Η, q, J= 1.2 Hz), 2.27 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.46 (3H, s), 7.29 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.63 (1H, br). Ή-NMR (200MHz, CDC1 3 ): δ 1.78 (3Η, q, J = 1.2 Hz), 2.27 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 2.46 (3H, s), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, br).
元素分析値: C16H17N303S3として計算値: C, 48.59; H, 4.33; , 10.62; S, 24.32 (%) 、 実測値: C, 48.50; H, 4.30; N, 10.52; S, 24.24 (%) . Elemental analysis: Calculated as C 16 H 17 N 3 0 3 S 3: C, 48.59; H, 4.33;, 10.62; S, 24.32 (%), Found: C, 48.50; H, 4.30 ; N, 10.52 ; S, 24.24 (%).
融点. 218.0-218.5 Melting point.218.0-218.5
実施例 4-97 Example 4-97
Ή-NMR (300MHz, CDC13) : δ 1.12 (2H, m), 1.56 (4H, m), 1.67 (2H, brq, J= 7.8 Ή-NMR (300MHz, CDC1 3 ): δ 1.12 (2H, m), 1.56 (4H, m), 1.67 (2H, brq, J = 7.8
Hz), 1.79 (3H, brq, J= 0.9 Hz), 1.79 (2H, m), 1.84 (1H, quint, J= 7.0 Hz), 2.26 (3H, brq, J= 0.9 Hz), 2.41 (3H, s), 2.94 (2H, t, J= 7.6 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.61 (1H, brs). Hz), 1.79 (3H, brq, J = 0.9 Hz), 1.79 (2H, m), 1.84 (1H, quint, J = 7.0 Hz), 2.26 (3H, brq, J = 0.9 Hz), 2.41 (3H, s), 2.94 (2H, t, J = 7.6 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.61 (1H, brs).
元素分析値: C22H27N303S3として計算値: C, 55.32; H, 5.70; N, 8.80; S, 20.14 (%) 、 実測値: C, 55.21; H, 5.77; N, 8.85; S, 20.25 (%) . Elemental analysis: C 22 H 27 N 3 0 3 S 3 Calculated: C, 55.32; H, 5.70 ; N, 8.80; S, 20.14 (%), Found: C, 55.21; H, 5.77 ; N, 8.85; S, 20.25 (%).
融点. 166.0-166.5 : Melting point: 166.0-166.5:
実施例 4 - 98 Example 4-98
Ή-NMR (200MHz, CDC13) : δ 1.06 (2Η, m), 1.42 (2H, m), 1.50-1.80 (9H, m), 1.78 (3H, brs), 2.26 (3H, brs), 2.41 (3H, s), 2.93 (2H, t, J- 7.5 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.06 (2Η, m), 1.42 (2H, m), 1.50-1.80 (9H, m), 1.78 (3H, brs), 2.26 (3H, brs), 2.41 ( 3H, s), 2.93 (2H, t, J-7.5 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56 (%) 、 実測値: C, 56.24; H, 5.93; N, 8.71; S, 19.61 (%) . 融点. 180.0-181.0°C Elemental analysis: C 23 H 29 N 3 0 3 S 3 Calculated: C, 56.18; H, 5.94 ; N, 8.55; S, 19.56 (%), Found: C, 56.24; H, 5.93; N, 8.71; S, 19.61 (%). Melting point. 180.0-181.0 ° C
実施例 4 - 99 Example 4-99
酢酸ェチル -ジェチルェ一テルから再結晶し、濃赤色結晶を得た。 融点: 112- 114°C. 赤外吸収スペクトル(IR) (KBr中) V: 3210, 2934, 2880, 1732, 1615, 1557 cm"1. Ή-NMR (CDC13) : δ 1.50-1.82 (6Η, m), 1.78 (3H, d, J=0.8 Hz), 2.26 (3H, s), 2.32 (2H, t, J=7.0 Hz), 2.42 (3H, s), 2.95 (2H, t, J=8.4 Hz), 2.95 (3H, s), 3.01 (3H, s), 7.31 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.61 (1H, bs). 元素分析値: C23 。N404S3'0.2 0として計算値: C, 52.49; H, 5.82; N, 10.64; S, 18.27 (%), 実測値: C, 52.41; H, 5.71; N, 10.67; S, 18.34 ( ) . The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals. Mp:. 112- 114 ° C Infrared absorption spectrum (IR) (in KBr) V:. 3210, 2934 , 2880, 1732, 1615, 1557 cm "1 Ή-NMR (CDC1 3): δ 1.50-1.82 (6Η , m), 1.78 (3H, d, J = 0.8 Hz), 2.26 (3H, s), 2.32 (2H, t, J = 7.0 Hz), 2.42 (3H, s), 2.95 (2H, t, J = 8.4 Hz), 2.95 (3H, s), 3.01 (3H, s), 7.31 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.61 (1H, bs). Analytical value: C 23 , Calculated as N 4 0 4 S 3 '0.20: C, 52.49; H, 5.82; N, 10.64; S, 18.27 (%), found: C, 52.41; H, 5.71; N , 10.67; S, 18.34 ().
実施例 4-100 Example 4-100
酢酸ェチル -ジェチルエーテルから再結晶し、 濃赤色結晶を得た。 融点: 99.0-10rC. 赤外吸収スペクトル(IR) (KBr 中) ソ: 3129, 2926, 2861, 1736, 1686, 1659, 1593, 1545, 1508 cm"1. Ή-NMR (CDC13) : δ 1.34-1.50 (2Η, m), 1.62-1.86 (4H, m), 1.78 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.91 (2H, t, J=7.6 Hz), 7.07 (1H, s), 7.31 (2H, d, J=8.0 Hz), 7.47 (1H, s), 7.84 (2H, d, J=8.2 Hz), 9.63 (1H, bs) . The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals. Mp:. 99.0-10rC infrared absorption spectrum (IR) (in KBr) Seo:. 3129, 2926, 2861, 1736, 1686, 1659, 1593, 1545, 1508 cm "1 Ή-NMR (CDC1 3): δ 1.34 -1.50 (2Η, m), 1.62-1.86 (4H, m), 1.78 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.91 (2H, t, J = 7.6 Hz), 7.07 (1H, s), 7.31 (2H, d, J = 8.0 Hz), 7.47 (1H, s), 7.84 (2H, d, J = 8.2 Hz), 9.63 (1H, bs).
元素分析値: C23H27N503S3 · 0.8H20として計算値: C, 51.92; H, 5.42; N, 13.16; S, 18.08 (%), 実測値: C, 52.06; H, 5.36; N, 12.70; S, 18.09 ( ) . Elemental analysis: C 23 H 27 N 5 0 3 S 3 · 0.8 H 2 0 Calculated: C, 51.92; H, 5.42; N, 13.16; S, 18.08 (%), found: C, 52.06; H , 5.36; N, 12.70; S, 18.09 ().
実施例 4-101 Example 4-101
酢酸ェチル -メタノール -テトラヒドロフランから再結晶し、濃赤色結晶を得た。: 融点 174 - 176°C. 赤外吸収スペクトル(IR) (KBr中) v: 3300, 3061, 2938, 2863, 2247, 1732, 1663, 1615, 1557, 1495cm—1. Ή-NMR (CDC13): <5 1.50-1.82 (6H, m), 1.79 (3H, s), 2.27 (3H, s), 2.37 (2H, t, J=6.6 Hz), 2.42 (3H, s), 2.95 (2H, t, J=7.4 Hz), 7.29 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.0 Hz), 9.62 (1H, bs). 実施例 4-102 Recrystallization from ethyl acetate-methanol-tetrahydrofuran yielded dark red crystals. :.. Mp 174 - 176 ° C Infrared absorption spectrum (IR) (in KBr) v: 3300, 3061, 2938, 2863, 2247, 1732, 1663, 1615, 1557, 1495cm- 1 Ή-NMR (CDC1 3) : <5 1.50-1.82 (6H, m), 1.79 (3H, s), 2.27 (3H, s), 2.37 (2H, t, J = 6.6 Hz), 2.42 (3H, s), 2.95 (2H, t) , J = 7.4 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.0 Hz), 9.62 (1H, bs).
N - {5, 6-ジメチル _3-ォキソ -8- [(テトラヒド口- 2-フラニルメチル)スルファニ ル] -2, 3-ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, ] チアジン- 1-ィリデン }- 4-メチルベ ンゼンスルホンアミド 実施例 3- 4で得た化合物 (281 mg, 0.737匪 ol)および炭酸カリウム (153 mg, 1.10匪 ol) の DMF (4.5 ml)の懸濁液に 80 で攪拌しながらヨウ化テドラヒドロ フルフリル (0.120 ml, 0.958imnol)を加え、 反応液を 2.5時間攪拌した。 反応液 に ίΝ塩酸 (1.47 ml)を加え、 メタノール—水(8:3, 11 ml)で希釈し、 室温で 30 分間攪拌した。析出物を濾取し、 メタノール一水 (1:1 および 2:1)で洗浄、 乾燥 して標題化合物 (205 mg, 0.440画 ol)を灰赤色粉末として得た。 N- {5,6-dimethyl_3-oxo-8-[(tetrahydrid-2-furanylmethyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,] thiazine-1 -Ylidene}-4-methylbenzenesulfonamide Tedrahydrofurfuryl iodide (0.120 mg) was added to a suspension of the compound obtained in Example 3-4 (281 mg, 0.737 bandol) and potassium carbonate (153 mg, 1.10 bandol) in DMF (4.5 ml) while stirring at 80. ml, 0.958imnol), and the reaction solution was stirred for 2.5 hours. Hydrochloric acid (1.47 ml) was added to the reaction solution, which was diluted with methanol-water (8: 3, 11 ml) and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1 and 2: 1), and dried to give the title compound (205 mg, 0.440 mmol) as a gray-red powder.
Ή-NMR (200MHz, CDC13) : δ 1.73 (1Η, m), 1.78 (3H, brs), 1.91 (2H, quint, J= 7.0 Hz), 2.07 (1H, m), 2.26 (3H, brs), 2.42 (3H, s), 3.03 (1H, dd, J= 13.6, 6.2 Hz), 3.16 (1H, dd, J= 13.2, 5.4 Hz), 3.79 (1H, dt, J= 15.0, 9.0 Hz), 3.86 (1H, dt, J= 15.0, 7.0 Hz), 4.11 (1H, quint, J= 6.0 Hz), 7.28 (2H, d, J= 8.2 Hz), 7.85 (2H, d, J= 8.2 Hz), 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.73 (1Η, m), 1.78 (3H, brs), 1.91 (2H, quint, J = 7.0 Hz), 2.07 (1H, m), 2.26 (3H, brs) , 2.42 (3H, s), 3.03 (1H, dd, J = 13.6, 6.2 Hz), 3.16 (1H, dd, J = 13.2, 5.4 Hz), 3.79 (1H, dt, J = 15.0, 9.0 Hz), 3.86 (1H, dt, J = 15.0, 7.0 Hz), 4.11 (1H, quint, J = 6.0 Hz), 7.28 (2H, d, J = 8.2 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.64 (1H, brs).
元素分析値: C2。H23N304S3として計算値: C, 51.59; H, 4.98; N, 9.02; S, 20.66Elemental analysis: C 2. H 23 N 3 0 4 S 3 Calculated: C, 51.59; H, 4.98 ; N, 9.02; S, 20.66
(%) 、 実測値: C, 51.34; H, 4.90; N, 8.96; S, 20.71 (%) . (%), Found: C, 51.34; H, 4.90; N, 8.96; S, 20.71 (%).
融点. 147.5- 148.0°C 147.5- 148.0 ° C
実施例 4-103 Example 4-103
N - [8- (シクロへプチルスルファニル) - 5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H- イミダゾ [5,1- c][l,4〕 チアジン-卜イリデン] -4-メチルベンゼンスルホンアミド 実施例 3-4で得た化合物 (260 mg, 0.682腿 ol)および炭酸カリウム(HI mg, 1.02 mmol)の DMF (4.3 ml)の懸濁液に 80°Cで攪拌しながらヨウ化シクロへプチ ル (0.197 ml, 0.818匪 ol)を加え、 反応液を 2.5時間攪拌した。 反応液に 1N塩 酸 (1.36 ml)を加え、 メタノール一水(5:3, 8 ml)で希釈し、 室温で 30分間攪拌 した。 析出物を濾取し、 メタノール一水 (1:1)、 水、 メタノール一水 (1:1)およ びジェチルエーテルで洗浄、 乾燥して標題化合物 (214 mg, 0.448 mmol)を灰赤色 粉末として得た。 N- [8- (cycloheptylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [l, 4] thiazine-triylidene] -4 -Methylbenzenesulfonamide A suspension of the compound obtained in Example 3-4 (260 mg, 0.682 mol) and potassium carbonate (HI mg, 1.02 mmol) in DMF (4.3 ml) was stirred at 80 ° C while stirring. Cycloheptyl iodide (0.197 ml, 0.818 bandol) was added, and the reaction solution was stirred for 2.5 hours. 1N Hydrochloric acid (1.36 ml) was added to the reaction solution, diluted with methanol / water (5: 3.8 ml) and stirred at room temperature for 30 minutes. The precipitate is collected by filtration, washed with methanol-water (1: 1), water, methanol-water (1: 1) and getyl ether, and dried to give the title compound (214 mg, 0.448 mmol) as a gray-red color Obtained as a powder.
Ή-NMR (200MHz, CDC13) : 6 1.55 (6H, brs), 1.70 (4H, brs), 1.78 (3H, q, J= 0.8 Hz), 2.06 (2H, m), 2.26 (3H, q, J= 0.8 Hz), 2.42 (3H, s), 3.52 (1H, sept, J= 4.4 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). 元素分析値: C22H27N304S3として計算値: C, 55.32; H, 5.70; N, 8.80; S, 20.14 (%) 、 実測値: C, 55.32; H, 5.79; N, 8.82; S, 20.03 (%) · 融点. 167.5-168.5°C Ή-NMR (200MHz, CDC1 3 ): 6 1.55 (6H, brs), 1.70 (4H, brs), 1.78 (3H, q, J = 0.8 Hz), 2.06 (2H, m), 2.26 (3H, q, J = 0.8 Hz), 2.42 (3H, s), 3.52 (1H, sept, J = 4.4 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.64 . (1H, brs) elemental analysis: calculated C 22 H 27 N 3 0 4 S 3: C, 55.32; H, 5.70; N, 8.80; S, 20.14 (%), Found: C, 55.32; H, 5.79; N, 8.82; S, 20.03 (%) Melting point.167.5-168.5 ° C
実施例 4-104 Example 4-104
N - (8- {[2-(1, 3-ジォキソラン- 2 -ィル)ェチル]スルファエル) -5, 6 -ジメチル- 3 -才 キソ- 2, 3_ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン ) -4-メチルベ ンゼンスルホンアミド N- (8-{[2- (1,3-Dioxolan-2-yl) ethyl] sulfael) -5,6-dimethyl-3-year-old oxo-2,3_dihydro-1H-imidazo [5,1 -c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物(262 mg, 0.687雇 ol)および炭酸カリウム(142 mg, 1.03 mmol)の DMF (4.4 ml)の懸濁液に 80 で攪拌しながら 2 -(2-ブロモェチル )-1, 3- ジォキソラン (0.109 ml, 0.893 mmol)を加え、 反応液を 100分間攪拌した。 反応 液に 1N塩酸 (1.37 ml)を加え、 メタノール一水(1:2, 3 ml)で希釈し、 室温で 30 分間攪拌した。析出物を濾取し、メタノール一水 (1:1)、水、メタノール一水 (1:1) およびジェチルエーテルで洗浄、 乾燥して標題化合物 (234 mg, 0.486 mfflol)を灰 赤色粉末として得た。 A suspension of the compound obtained in Example 3-4 (262 mg, 0.687 mol) and potassium carbonate (142 mg, 1.03 mmol) in DMF (4.4 ml) was stirred at 80 with 2- (2-bromoethyl). -1,3-Doxolan (0.109 ml, 0.893 mmol) was added, and the reaction solution was stirred for 100 minutes. The reaction mixture was diluted with 1N hydrochloric acid (1.37 ml), diluted with methanol / water (1: 2, 3 ml), and stirred at room temperature for 30 minutes. The precipitate is collected by filtration, washed with methanol-water (1: 1), water, methanol-water (1: 1) and getyl ether, and dried to give the title compound (234 mg, 0.486 mfflol) as a gray-red powder Obtained.
Ή-NMR (200MHz, CDC13) : δ 1.79 (3Η, s), 2.03 (2H, dt, J= 4.4, 7.7 Hz), 2.26 (3H, brs), 2.42 (3H, s), 3.05 (2H, t, J= 7.7 Hz), 3.88 (2H, m), 3.97 (2H, m), 4.96 (1H, t, J= 4.2 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.79 (3Η, s), 2.03 (2H, dt, J = 4.4, 7.7 Hz), 2.26 (3H, brs), 2.42 (3H, s), 3.05 (2H, t, J = 7.7 Hz), 3.88 (2H, m), 3.97 (2H, m), 4.96 (1H, t, J = 4.2 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C2。H23N305S3として計算値: C, 49.88; H, 4.81; N, 8.72; S, 19.97Elemental analysis: C 2. H 23 N 3 0 5 S 3 Calculated: C, 49.88; H, 4.81 ; N, 8.72; S, 19.97
(%) 、 実測値: G, 49.53; H, 4.74; N, 8.56; S, 19.81 (%) . (%), Found: G, 49.53; H, 4.74; N, 8.56; S, 19.81 (%).
融点. 185.0-188.0°C Melting point.185.0-188.0 ° C
実施例 4-105 Example 4-105
N- (8- {[2-(1, 3-ジォキサン _2-ィル)ェチル] スルファ二ル} -5, 6-ジメチル- 3-ォ キソ- 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン )-4-メチルベ ンゼンスルホンアミド , 実施例 3-4 で得た化合物(234 mg, 0.613 mmol)および炭酸カリウム(127 mg, 0.920匪 ol)の DMF (4.0 ml)の懸濁液に 80 で攪拌しながら 2- (2-プロモェチ ル)- 1,3-ジォキサン (0.103 ml, 0.736 mmol)を加え、 反応液を 2時間攪拌した。 反応液に 1N塩酸 (1.23 ml)を加え、 メタノール—水(1:2, 3 ml)で希釈し、 室温 で 30分間攪拌した。析出物を濾取し、 メタノール—水 (1:1)、 水、 メ夕ノ一ルー 水 (1:1)およびジェチルェ一テルで洗浄、 乾燥して標題化合物 (248 mg, 0.500 醒 ol)を灰赤色粉末として得た。 N- (8-{[2- (1,3-Dioxane_2-yl) ethyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide, the compound obtained in Example 3-4 (234 mg, 0.613 mmol) and potassium carbonate (127 mg, 0.920 bandol) 2- (2-Promoethyl) -1,3-dioxane (0.103 ml, 0.736 mmol) was added to a suspension of DMF (4.0 ml) with stirring at 80, and the reaction solution was stirred for 2 hours. The reaction mixture was diluted with 1N hydrochloric acid (1.23 ml), diluted with methanol-water (1: 2, 3 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1), water, methanol (1: 1) and water, dried and dried to give the title compound (248 mg, 0.500 Ol) was obtained as a gray-red powder.
Ή-N R (200MHz, CDC13) : <5 1.36 (1H, brd, J= 13.4 Hz), 1.78 (3H, s), 1.95 (2H, dt, J= 5.0, 7.0 Hz), 2.07 (1H, m), 2.26 (3H, brq, J= 1.2 Hz), 2.42 (3H, s), 3.04 (2H, t, J= 7.5 Hz), 3.75 (2H, dt, J= 2.2, 11.2 Hz), 4.09 (2H, dd, J= 11.0, 4.8 Hz) , 4.63 (1H, t, J= 4.8 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή-NR (200MHz, CDC1 3 ): <5 1.36 (1H, brd, J = 13.4 Hz), 1.78 (3H, s), 1.95 (2H, dt, J = 5.0, 7.0 Hz), 2.07 (1H, m ), 2.26 (3H, brq, J = 1.2 Hz), 2.42 (3H, s), 3.04 (2H, t, J = 7.5 Hz), 3.75 (2H, dt, J = 2.2, 11.2 Hz), 4.09 (2H , dd, J = 11.0, 4.8 Hz), 4.63 (1H, t, J = 4.8 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H , brs).
元素分析値: C2lH25N305S3 ·0.3 0として計算値: C, 50.34; Η, 5.15; Ν, 8.39; S, 19.20 (%) 、 実測値: C, 50.60; Η, 5.13; Ν, 8.42; S, 18.86 (%) . Elemental analysis: C 2l H 25 N 3 0 5 S 3 · 0.30 Calculated: C, 50.34; Η, 5.15; Ν, 8.39; S, 19.20 (%), found: C, 50.60; Η, 5.13 ; Ν, 8.42; S, 18.86 (%).
融点. 196.5-199.0°C Melting point.196.5-199.0 ° C
実施例 4-106 Example 4-106
iert -プチル 3_[(5,6-ジメチル-卜 {[(4_メチルフエニル)スルホニル]イミ ノ }-3 -ォキソ - 2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, ]チアジン- 8-ィル)スルフ ァニル]プロピル力一バメー卜 iert-butyl 3 _ [(5,6-dimethyl-tri {[(4_methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,] Thiazine-8-yl) sulfanyl] propyl
実施例 3-4で得た化合物(263 mg, 0.689 mmol)および炭酸カリウム(143 mg, 1.03 職 ol)の DMF (4.0 ml)の懸濁液に 80°Cで攪拌しながら、 1-ァミノ- 3-ブロモプロ パン臭化水素酸塩、 重炭酸ジ tert-ブチルおよびトリェチルァミンから調製した N - Boc-卜ァミノ- 3-ブロモプロパン (純度 88%, 0.22 ml, 0.758匪 ol)を加え、 反 応液を 1.5時間攪拌した。 反応液に 1N塩酸 (1.38 ml)を加え、 メタノール一水 (1:1, 4 ml)で希釈し、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール— 水 (1:1)、 水、 メタノール一水 (1:1)およびメタノールで洗浄、 乾燥して標題化 合物 (323 mg, 0.600匪 ol)を灰赤色粉末として得た。 To a suspension of the compound (263 mg, 0.689 mmol) obtained in Example 3-4 and potassium carbonate (143 mg, 1.03 mmol) in DMF (4.0 ml) was added 1-amino-amine while stirring at 80 ° C. N-Boc-Tamino-3-bromopropane (purity 88%, 0.22 ml, 0.758 bandol) prepared from 3-bromopropane hydrobromide, ditert-butyl bicarbonate and triethylamine was added, and the reaction solution was added. Was stirred for 1.5 hours. 1N Hydrochloric acid (1.38 ml) was added to the reaction solution, diluted with methanol / water (1: 1, 4 ml), and stirred at room temperature for 30 minutes. The precipitate is collected by filtration, washed with methanol-water (1: 1), water, methanol-aqueous solution (1: 1) and methanol, and dried to give the title compound (323 mg, 0.600 bandol ol) as a gray-red powder As obtained.
Ή-NMR (200MHz, CDC13) δ 1.45 (9Η, s), 1.78 (3H, brs), 1.86 (2H, quint, J= 7.3 Hz), 2.26 (3H, brs), 2.42 (3H, s), 2.96 (2H, t, J= 7.3 Hz), 3.20 (2H, q, J= 6.5 Hz), 4.64 (1H, m), 7.29 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.66 (1H, brs). Ή-NMR (200MHz, CDC1 3 ) δ 1.45 (9Η, s), 1.78 (3H, brs), 1.86 (2H, quint, J = 7.3 Hz), 2.26 (3H, brs), 2.42 (3H, s), 2.96 (2H, t, J = 7.3 Hz), 3.20 (2H, q, J = 6.5 Hz), 4.64 (1H, m), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.66 (1H, brs).
元素分析値: C23H3。N405S3 ·0.2 0として計算値: C, 50.94; Η, 5.65; Ν, 10.33; S, 17.74 (%) 、 実測値: C, 51.22; Η, 5.66; Ν, 10.39; S, 17.43 (%) . Elemental analysis: C 23 H 3. Calculated as N 4 0 5 S 3 · 0.20: C, 50.94; Η, 5.65; Ν, 10.33; S, 17.74 (%), found: C, 51.22; Η, 5.66; Ν, 10.39; S, 17.43 (%).
融点. 166.0-168.0°C Melting point.166.0-168.0 ° C
実施例 4- 107 酢酸ェチル-ジェチルエーテルから再結晶し、濃赤色結晶を得た。 融点: 204- 206 :. 赤外吸収スペクトル(IR) (KBr中) V: 3167, 2922, 1734, 1663, 1618, 1561 cm"1. Ή-NMR (CDC") : δ 1.78 (3H, d, J=0.6 Hz), 2.26 (3H, d, J=l.2 Hz), 2.43 (3H, s), 2.66-2.82 (3H, m), 3.04-3.24 (4H, m), 7.22-7.45 (4H, m), 7.28 (2H, d, J=8.0 Hz), 7.87 (2H, d, J=8. Hz), 9.61 (1H, bs). Example 4-107 The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals. Melting point: 204-206:. Infrared absorption spectrum (IR) (in KBr) V: 3167, 2922, 1734, 1663, 1618, 1561 cm " 1. Ή-NMR (CDC"): δ 1.78 (3H, d, J = 0.6 Hz), 2.26 (3H, d, J = l.2 Hz), 2.43 (3H, s), 2.66-2.82 (3H, m), 3.04-3.24 (4H, m), 7.22-7.45 (4H , m), 7.28 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.Hz), 9.61 (1H, bs).
元素分析値: C25H25N303S3 · 0.5H20 として計算値: C, 57.67; H, 5.03; N, 8.07; S, 18.48 (%)、 実測値: C, 57.52; H, 5.02; N, 8.24; S, 18.76 (%) . Elemental analysis: C 25 H 25 N 3 0 3 S 3 · 0.5 H 2 0 Calculated: C, 57.67; H, 5.03; N, 8.07; S, 18.48 (%), found: C, 57.52; H , 5.02; N, 8.24; S, 18.76 (%).
実施例 4-108 Example 4-108
酢酸ェチル -ジェチルェ一テルから再結晶し、濃赤色結晶を得た。 融点 200- 202°C. 赤外吸収スペクトル(IR) (KBr中) リ: 3231, 2976, 2932, 1761, 1694, 1622, 1559 cm"1. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals. Melting point 200-202 ° C. Infrared absorption spectrum (IR) (in KBr): 3231, 2976, 2932, 1761, 1694, 1622, 1559 cm " 1 .
¾-NMR (CDC13) : δ 1.02-1.28 (2Η, m), 1.46 (9H, s), 1.50-1.86 (3H, m), 1.79 (3H,s), 2.26 (3H, s), 2.42 (3H, s), 2.67 (2H, t, J=11.4 Hz), 2.84 (2H, d, J=7.0 Hz), 3.98-4.20 (2H, m), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8. Hz), 9.62 (1H, bs). ¾-NMR (CDC1 3): δ 1.02-1.28 (2Η, m), 1.46 (9H, s), 1.50-1.86 (3H, m), 1.79 (3H, s), 2.26 (3H, s), 2.42 ( 3H, s), 2.67 (2H, t, J = 11.4 Hz), 2.84 (2H, d, J = 7.0 Hz), 3.98-4.20 (2H, m), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.Hz), 9.62 (1H, bs).
元素分析値: C26H34N405S3'0.2 0として計算値: C, 53.62; H, 5.95; N, 9.62; S, 16.51 (%)> 実測値: C, 53.53; H, 6.08; N, 9.48; S, 16.31 ( ) . Elemental analysis: C 26 H 34 N 4 0 5 S 3 '0.20 Calculated: C, 53.62; H, 5.95; N, 9.62; S, 16.51 (%)> Found: C, 53.53; H, 6.08 ; N, 9.48; S, 16.31 ().
実施例 4-109 Example 4-109
酢酸ェチル -メタノールから再結晶し、無色結晶を得た。 融点 210-212°C. 赤外 吸収スペクトル(IR) (KBr 中) リ: 3700-2200, 3376, 3247, 2978, 2930, 2556, 2461, 2433, 1740, 1717, 1659, 1613, 1557 cm— Recrystallization from ethyl acetate-methanol gave colorless crystals. Melting point 210-212 ° C. Infrared absorption spectrum (IR) (in KBr): 3700-2200, 3376, 3247, 2978, 2930, 2556, 2461, 2433, 1740, 1717, 1659, 1613, 1557 cm—
'H-NMR (DMS0-d6) : δ 1.82 (3Η, s), 2.18 (3H, s), 2.40 (3H, s), 2.80 (6H, s), 3.12-3.48 (4H, m), 7.38 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz). 'H-NMR (DMS0-d 6 ): δ 1.82 (3Η, s), 2.18 (3H, s), 2.40 (3H, s), 2.80 (6H, s), 3.12-3.48 (4H, m), 7.38 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz).
元素分析値: CigH24N403Ss'HCl'0.4H20として計算値: C, 45.98; H, 5.24; N, 11.29; S, 19.38 (%), 実測値: C, 46.13; H, 5.07; N, 11.07; S, 19.02 {%) . Elemental analysis: C ig H 24 N 4 0 3 S s'HCl'0.4H 2 0 Calculated: C, 45.98; H, 5.24 ; N, 11.29; S, 19.38 (%), Found: C, 46.13 ; H, 5.07; N, 11.07; S, 19.02 (%).
実施例 4-110 Example 4-110
酢酸ェチル-メタノールから再結晶し、無色結晶を得た。 融点 206- 208°C. 赤外 吸収スペクトル(IR) (KBr 中) V: 3700-2200, 3248, 2949, 2647, 2469, 1728, 1661, 1611, 1557 cm—1. Ή-NMR (DMS0-d6) : δ 1.80 (3H, s), 1.93-2.08 (2H, HI), 2.18 (3H, s), 2.39 (3H, s), 2.76 (6H, s), 2.97-3.18 (4H, m), 7.38 (2H, d, J=8.0 Hz), 7.86 (2H, d, J=8.4 Hz). Recrystallization from ethyl acetate-methanol gave colorless crystals. Melting point 206-208 ° C. Infrared absorption spectrum (IR) (in KBr) V: 3700-2200, 3248, 2949, 2647, 2469, 1728, 1661, 1611, 1557 cm— 1 . Ή-NMR (DMS0-d 6 ): δ 1.80 (3H, s), 1.93-2.08 (2H, HI), 2.18 (3H, s), 2.39 (3H, s), 2.76 (6H, s), 2.97- 3.18 (4H, m), 7.38 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz).
実施例 4- 111 Example 4-111
酢酸ェチル -ジェチルェ一テルから再結晶し、濃赤色結晶を得た。融点: 173-175°C. 赤外吸収スぺクトル(IR) ( Br中) V: 3221, 2934, 2249, 1732, 1663, 1615, 1563, 1495 cm-1. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals. Melting point: 173-175 ° C. Infrared absorption spectrum (IR) (in Br) V: 3221, 2934, 2249, 1732, 1663, 1615, 1563, 1495 cm- 1 .
Ή-NMR (CDC13) : δ 1.79 (3Η, d, J=1.2 Hz), 1.96-2.12 (2H, m), 2.26 (3H, d, J=1.2 Hz), 2.43 (3H, s), 2.48 (2H, t, J-7.2 Hz), 3.06 (2H, t, J=7.0 Hz), 7.31 (2H, d, J=8.4 Hz), 7.84 (2H, d, J=8. Hz), 9.64 (1H, bs) . Ή-NMR (CDC1 3): δ 1.79 (3Η, d, J = 1.2 Hz), 1.96-2.12 (2H, m), 2.26 (3H, d, J = 1.2 Hz), 2.43 (3H, s), 2.48 (2H, t, J-7.2 Hz), 3.06 (2H, t, J = 7.0 Hz), 7.31 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.Hz), 9.64 ( 1H, bs).
元素分析値: C19 。N403S3として計算値: C, 50.87; H, 4.49; N, 12.49; S, 21.44Elemental analysis: C 19. Calculated for N 4 0 3 S 3 : C, 50.87; H, 4.49; N, 12.49; S, 21.44
(%), 実測値: C, 50:95; H, 4.46; N, 12.18; S, 21.15 ( ) . (%), Found: C, 50:95; H, 4.46; N, 12.18; S, 21.15 ().
実施例 4-112 Example 4-112
酢酸ェチル -ジェチルェ一テルから再結晶し、 濃赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals.
融点 166-168°C. 166-168 ° C.
赤外吸収スペクトル aR) (KBr中) : 3291, 2928, 2245, 1748, 1717, 1661, 1611, 1555, 1497 cm"1. Infrared absorption spectrum aR) (in KBr): 3291, 2928, 2245, 1748, 1717, 1661, 1611, 1555, 1497 cm " 1 .
Ή-NMR (CDC13) : δ 1.72-1.92 (4Η, ι), 1.79 (3H, d, J=0.8 Hz), 2.26 (3H, s), 2.39 (2H, t, J-6.6 Hz), 2.43 (3H, s), 2.97 (2H, t, J=6.8 Hz), 7.30 (2H, d, J=8.2 Hz), 7.84 (2H, d, J=8.6 Hz), 9.63 (1H, bs) . Ή-NMR (CDC1 3): δ 1.72-1.92 (4Η, ι), 1.79 (3H, d, J = 0.8 Hz), 2.26 (3H, s), 2.39 (2H, t, J-6.6 Hz), 2.43 (3H, s), 2.97 (2H, t, J = 6.8 Hz), 7.30 (2H, d, J = 8.2 Hz), 7.84 (2H, d, J = 8.6 Hz), 9.63 (1H, bs).
元素分析値: C2。H22N403S3として計算値: C, 51.93; H, 4.79; N, 12.11 (%)、 実 測値: C, 51.87; H, 4.61; , 12.10 ( ) · Elemental analysis: C 2. Calculated as H 22 N 4 0 3 S 3 : C, 51.93; H, 4.79; N, 12.11 (%), actual Hakachi: C, 51.87; H, 4.61 ;, 12.10 () ·
実施例 4-113 Example 4-113
N - [5, 6-ジメチル- 3-ォキソ -8- (ビニルスルファニル) -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 1-ィリデン] - 4 -メチルベンゼンスルホンアミド N- [5,6-Dimethyl-3-oxo-8- (vinylsulfanyl) -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene]-4- Methylbenzenesulfonamide
実施例 3-4 で得た化合物 (503 g, 1.3 雇 ol)および 1,2-ジブロモェタン (0.121 ml, 1.38腿 ol)の DMF (10 ml)の懸濁液に 80°Cで攪拌しながら炭酸力リウ ム (146 mg, 1.06匪 ol)を加え、 反応液を 80分間攪拌した。 反応液に 1 塩酸 (0.79 ml)を加え、 メタノール一水(1:1, 10 ml)で希釈し、 室温で 30分間攪拌し た。析出物を濾取し、 メタノール—水 (1:1)、 水およびメタノールで洗浄後、 THF に懸濁した。不溶物を濾取し、 THF-メ夕ノール(1: 1)およびメ夕ノールで洗浄した。 母液および洗浄液をすベてあわせて酢酸ェチルで抽出し、 0.1N塩酸、水および飽 和食塩水で洗浄後、硫酸ナトリウムで乾燥し、濃縮して標題化合物 (49mg, 0.120 腿 ol)を赤紫色粉末として得た。 Carbon dioxide was added to a suspension of the compound obtained in Example 3-4 (503 g, 1.3 1.3ol) and 1,2-dibromoethane (0.121 ml, 1.38 olol) in DMF (10 ml) while stirring at 80 ° C. Potassium (146 mg, 1.06 marl) was added and the reaction was stirred for 80 minutes. Hydrochloric acid (0.79 ml) was added to the reaction solution, diluted with methanol / water (1: 1, 10 ml), and stirred at room temperature for 30 minutes. Was. The precipitate was collected by filtration, washed with methanol-water (1: 1), water and methanol, and suspended in THF. The insoluble material was collected by filtration, and washed with THF-methanol (1: 1) and methanol. The mother liquor and the washings were combined, extracted with ethyl acetate, washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to give the title compound (49 mg, 0.120 t ol) as a red-purple powder As obtained.
Ή-NMR (200MHz, CDC13) : δ 1.77 (3Η, q, J= 0.6 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.43 (3H, s), 5.75 (1H, d, J= 9.4 Hz), 5.79 (1H, d, J= 16.6 Hz), 6.61 (1H, dd, J= 16.6, 9.2 Hz), 7.30 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, br). Ή-NMR (200MHz, CDC1 3 ): δ 1.77 (3Η, q, J = 0.6 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.43 (3H, s), 5.75 (1H, d, J = 9.4 Hz), 5.79 (1H, d, J = 16.6 Hz), 6.61 (1H, dd, J = 16.6, 9.2 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, br).
元素分析値: C17H17N303S3 · 0.25 0として計算値: C, 49.56; H, 4.28; N, 10.20; S, 23.35 ( ) 、 実測値: C, 49.48; H, 4.23; N, 10.09; S, 22.98 (%) . Elemental analysis: C 17 H 17 N 3 0 3 S 3 · 0.25 0 Calculated: C, 49.56; H, 4.28; N, 10.20; S, 23.35 (), found: C, 49.48; H, 4.23; N, 10.09; S, 22.98 (%).
融点. 212.5-213.5°C Melting point.212.5-213.5 ° C
実施例 4-114 Example 4-114
tert-ブチル 2- [(5, 6-ジメチル-卜 {[(4 -メチルフエニル)スルホニル]イミノ} - 3 - ォキソ - 2,3-ジヒドロ- 1H -イミダゾ [5,l_c] [1, 4]チアジン- 8-ィル)スルファニル] ェチルカーバメート tert-butyl 2-[(5,6-dimethyl-tolu [[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine -8-yl) sulfanyl] ethyl carbamate
実施例 3- 4で得た化合物(310 mg, 0.813匪 ol)および炭酸カリウム(169 mg, 1.22 蘭 ol)の DMF (5.0 ml)の懸濁液に 80°Cで攪拌しながら、 卜ァミノ- 2-クロ口エタ ン塩酸塩、 重炭酸ジ tert -プチルおよびトリェチルァミンから調製した N - Boc -卜 ァミノ _3_クロロェタン (純度 86%, 0.25 ml, 1.22顧 ol)を加え、 反応液を 1.5 時間攪拌した。 反応液に N-Boc-卜ァミノ- 3-クロロェタン(純度 86%, 0.08 ml, 0. 07 mmol)を加え、 反応液を 0.5時間攪拌した。 反応液に 1N塩酸 (1.63 ml)を 加え、 メタノール一水(3:2, 5 ml)で希釈し、 室温で 30分間攪拌した。 析出物を 濾取し、 メタノール一水 (1:1)、 水、 メタノール一水 (1:1)およびメタノールで 洗浄、 乾燥して標題化合物 (237 mg, 0.452 mraol)を灰赤色粉末として得た。 To a suspension of the compound obtained in Example 3-4 (310 mg, 0.813 ol) and potassium carbonate (169 mg, 1.22 ol) in DMF (5.0 ml) was added triamino- N-Boc-triamino_3_chloroethane (purity 86%, 0.25 ml, 1.22 ml) prepared from 2-chloro mouth ethane hydrochloride, ditert-butyl bicarbonate and triethylamine was added, and the reaction solution was stirred for 1.5 hours. did. N-Boc-Tamino-3-chloroethane (purity 86%, 0.08 ml, 0.07 mmol) was added to the reaction solution, and the reaction solution was stirred for 0.5 hour. 1N Hydrochloric acid (1.63 ml) was added to the reaction solution, diluted with methanol / water (3: 2.5 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1), water, methanol / water (1: 1) and methanol, and dried to give the title compound (237 mg, 0.452 mraol) as a gray-red powder .
Ή-NM (200MHz, CDC13) : δ 1.45 (9H, s), 1.78 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 3.09 (2H, t, J= 6.4 Hz), 3.39 (2H, q, J= 6.2 Hz), 4.93 (1H, m), 7.30 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.66 (1H, brs). Ή-NM (200MHz, CDC1 3 ): δ 1.45 (9H, s), 1.78 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 3.09 (2H, t, J = 6.4 Hz) , 3.39 (2H, q, J = 6.2 Hz), 4.93 (1H, m), 7.30 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.66 (1H, brs) .
元素分析値: C22H28N405S3として計算値: C, 50.36; H, 5.38; N, 10.68; S, 18.33 (%) 、 実測値: C, 50.18; H, 5.32; N, 10.70; S, 18.05 (%) . 融点. 200.0-201.0°C Elemental analysis: C 22 H 28 N 4 0 5 S 3 Calculated: C, 50.36; H, 5.38 ; N, 10.68; S, 18.33 (%), Found: C, 50.18; H, 5.32; N, 10.70; S, 18.05 (%). Melting point. 200.0-201.0 ° C
実施例 4-115 Example 4-115
N- (5, 6 -ジメチル- 3-ォキソ -8- { [2- (テトラヒドロ -2-ィル)ェチル] スルファニ リレ }- 2,3-ジヒドロ -1H-イミダゾ [5, 1- c] [1,4] チアジン -1-イリデン) -4-メチルベ ンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[2- (tetrahydro-2-yl) ethyl] sulfanylyl} -2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
テトラヒドロフラン -2-酢酸ェチル(4.82 g, 29.9匪01)の11? (96 ml)溶液に 0°C で攪拌しながら水素化リチウムアルミニウム(純度 80 1.06 g, 22.4腿01) の 粉末を数回に分けて加え、 0°Cで 80分間攪拌した。 反応液に 0.5M クェン酸水溶 液 (451111)を加ぇ、 1^(2001111)で3回抽出した。《1?を留去し、 クロロホルム(100 ml)で希釈後、乾燥、濃縮して淡黄色油状物を得た。この油状物をクロ口ホルム (60 ml)に溶解し、 0 で攪拌しながらメタンスルホニルクロリド(2.81ml, 35.9腿01) を加え、 次いでトリェチルァミン(5.38 ml, 38.8 mmol)のクロ口ホルム(10 ml) 溶液を 40分間かけて滴下し、 反応液を 0 で 40分間攪拌した。 反応液を濃縮し て酢酸ェチル(100 ml)で希釈し、 水、 2 炭酸水素ナトリウム水溶液および飽和食 塩水で洗浄し、乾燥、濃縮してメタンスルホン酸 2- (テトラヒドロフラン -2-ィル) ェチル (純度 90%, 4.58 g, 21.2 mmol)を淡褐色油状物として得た。 11? Of tetrahydrofuran-2-ethyl acetate (4.82 g, 29.9 bandages 01) (96 ml) The powder of lithium aluminum hydride (purity 80 1.06 g, 22.4 thigh 01) was added in several portions to the solution while stirring at 0 ° C, and the mixture was stirred at 0 ° C for 80 minutes. A 0.5 M aqueous solution of citrate (451111) was added to the reaction solution, and extracted three times with 1 ^ (2001111). << 1 was distilled off, diluted with chloroform (100 ml), dried and concentrated to obtain a pale yellow oil. This oil was dissolved in chloroform (60 ml), methanesulfonyl chloride (2.81 ml, 35.9 t01) was added while stirring at 0, and then triethylamine (5.38 ml, 38.8 mmol) in chloroform (10 ml). ) The solution was added dropwise over 40 minutes and the reaction was stirred at 0 for 40 minutes. The reaction mixture is concentrated, diluted with ethyl acetate (100 ml), washed with water, aqueous sodium bicarbonate solution and saturated brine, dried and concentrated to give 2- (tetrahydrofuran-2-yl) ethyl methanesulfonate (Purity 90%, 4.58 g, 21.2 mmol) was obtained as a pale brown oil.
Ή-NMR (200MHz, CDC13): (51.70-2.20 (6H, m), 3.02 (3H, s), 3.74 (2H, dt, J= 14.0, 7.0 Hz), 3.85 (1H, dt, J= 14.0, 6.8 Hz), 3.95 (1H, m), 4.07 (2H, dd, J= 7.1, 5.7 Hz). Ή-NMR (200MHz, CDC1 3 ): (51.70-2.20 (6H, m), 3.02 (3H, s), 3.74 (2H, dt, J = 14.0, 7.0 Hz), 3.85 (1H, dt, J = 14.0 , 6.8 Hz), 3.95 (1H, m), 4.07 (2H, dd, J = 7.1, 5.7 Hz).
実施例 3- 4で得た化合物(264 mg, 0.692腿 ol)および炭酸カリウム(143 mg, 1.04 mmol)の DMF (4.4 ml)の懸濁液に 80°Cで攪拌しながら上記メタンスルホン 酸 2- (テトラヒドロフラン -2-ィル)ェチル (161 mg, 0.830 mmol)を加え、 反応液 を 2時間攪拌した。さらに反応液にメタンスルホン酸 2- (テトラヒドロフラン- 2 - ィル)ェチル (30 mg, 0.140 mmol)を加え、 反応液を 1 時間攪拌した。 反応液に 1N塩酸 (1.38 ml)を加え、 メタノール—水(1:1, 6ml)で希釈し、 室温で 30分間 攪拌した。析出物を母液から分離し、メタノールから結晶化して標題化合物 (181 mg, 0.377匪 ol)を濃紫色粉末として得た。 The above methanesulfonic acid 2 was added to a suspension of the compound obtained in Example 3-4 (264 mg, 0.692 mol) and potassium carbonate (143 mg, 1.04 mmol) in DMF (4.4 ml) while stirring at 80 ° C. -(Tetrahydrofuran-2-yl) ethyl (161 mg, 0.830 mmol) was added, and the reaction solution was stirred for 2 hours. Further, 2- (tetrahydrofuran-2-yl) ethyl methanesulfonate (30 mg, 0.140 mmol) was added to the reaction solution, and the reaction solution was stirred for 1 hour. 1N Hydrochloric acid (1.38 ml) was added to the reaction solution, diluted with methanol-water (1: 1, 6 ml), and stirred at room temperature for 30 minutes. The precipitate was separated from the mother liquor and crystallized from methanol to give the title compound (181 mg, 0.377 ol) as a dark purple powder.
¾ - NMR (200MHz, CDC13) 'δ 1.78 (3Η, brs), 1.84 (2H, q, J= 6.6 Hz), 1.90 (2H, quint, J= 7.2 Hz), 1.93 (2H, m), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 3.05 (2H, m), 3.75 (1H, t, J= 6.6 Hz), 3.84 (1H, t, J= 6.6 Hz), 3.89 (1H, m), 7.28 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). ¾ - NMR (200MHz, CDC1 3 ) 'δ 1.78 (3Η, brs), 1.84 (2H, q, J = 6.6 Hz), 1.90 (2H, quint, J = 7.2 Hz), 1.93 (2H, m), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 3.05 (2H, m), 3.75 (1H, t, J = 6.6 Hz), 3.84 (1H, t, J = 6.6 Hz), 3.89 (1H, m), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.63 (1H, brs).
元素分析値: C21 5N304S3として計算値: C, 52.59; H, 5.25; N, 8.76; S, 20.06 (%) 、 実測値: C, 52.22; H, 5.23; N, 8.77; S, 20.11 (%) . Elemental analysis: Calculated as C 21 5 N 3 0 4 S 3: C, 52.59; H, 5.25; N, 8.76; S, 20.06 (%), Found: C, 52.22; H, 5.23 ; N, 8.77 ; S, 20.11 (%).
融点. 159.0-160.0°C Melting point.159.0-160.0 ° C
実施例 4-116 Example 4-116
エタノールから再結晶し、 橙色結晶を得た。 Recrystallization from ethanol gave orange crystals.
融点 :196- 198°C. Melting point: 196-198 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3700-2300, 2953, 2795, 2726, 1730, 1717, 1663, 1607, 1557, 1495 cm1. Infrared absorption spectrum (IR) (in KBr) V: 3700-2300, 2953, 2795, 2726, 1730, 1717, 1663, 1607, 1557, 1495 cm 1 .
'Η-賺 (CDCl3+DMS0-d6) : δ 1.64-1.84 (3Η, m), 1.79 (3H, s), 1.90-2.08 (2H, m), 2.26 (3H, s), 2.44 (3H, s), 2.64-2.96 (2H, m), 2.90 (2H, d, J=5.8 Hz), 3.32-3.50 (2H, m), 7.30 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.62 (1H, bs). 'Η-new (CDCl 3 + DMS0-d 6 ): δ 1.64-1.84 (3Η, m), 1.79 (3H, s), 1.90-2.08 (2H, m), 2.26 (3H, s), 2.44 (3H , s), 2.64-2.96 (2H, m), 2.90 (2H, d, J = 5.8 Hz), 3.32-3.50 (2H, m), 7.30 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.62 (1H, bs).
実施例 4-117 Example 4-117
N-{5, 6-ジメチル- 3-ォキソ- 8- [(テトラヒド口 -2H-ピラン- 4-ィルメチル)スルフ ァニル ] -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン卜 4-メチ 実施例 3-4で得た化合物 (305 ig, 0.799 mmol)および炭酸カリウム(166 mg, N- {5,6-dimethyl-3-oxo-8-[(tetrahydric-2H-pyran-4-ylmethyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-yldent 4-methyl The compound obtained in Example 3-4 (305 ig, 0.799 mmol) and potassium carbonate (166 mg,
I.20 miiiol)の DMF (5.0 ml)の懸濁液に 80 で攪拌しながら参考例 86で得たメタ ンスルホン酸テトラヒドロピラン- 4-メチル(288 mg, 1.04 mmol)を加え、 反応液 を 2.5時間攪拌した。 反応液に 1N塩酸 (1.60 および水 (40 ml)を加え、 酢酸 ェチル(50ml)で抽出した。 有機層を水および飽和食塩水で洗浄し、 乾燥、 濃縮し た。 残渣にメタノールを加え、 析出物を集めてメタノールで洗浄、 乾燥して標題 化合物 (124 mg, 0.259 腿 ol)を赤色粉末として得た。 To a suspension of I.20 miiiol) in DMF (5.0 ml) was added, while stirring at 80, the tetrahydropyran-4-methyl methanesulfonate (288 mg, 1.04 mmol) obtained in Reference Example 86, and the reaction mixture was stirred for 2.5 hours. Stirred for hours. 1N Hydrochloric acid (1.60 and water (40 ml) was added to the reaction solution, and the mixture was extracted with ethyl acetate (50 ml) .The organic layer was washed with water and saturated saline, dried and concentrated. The collected substance was collected, washed with methanol and dried to obtain the title compound (124 mg, 0.259 mol) as a red powder.
Ή-NMR (200MHz, CDC ) : 6 1.32 (2H, dq, J= 11.6, 4.0 Hz), 1.74 (2H, brd, J= Ή-NMR (200MHz, CDC): 6 1.32 (2H, dq, J = 11.6, 4.0 Hz), 1.74 (2H, brd, J =
II.6 Hz), 1.79 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 2.86 (2H, d, J= 6.6 Hz), 2.98 (1H, m), 3.36 (2H, td, J= 11.6, 0.8 Hz), 3.97 (2H, dd, J= 11.8, 4.0 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). 元素分析値: CZIH25N304S3 · 0.2¾0として計算値: C, 52.20; H, 5.30; N, 8.70; S, 19.91 (%), 実測値: C, 52.21; H, 5.34; N, 8.68; S, 20.05 (%) . II.6 Hz), 1.79 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 2.86 (2H, d, J = 6.6 Hz), 2.98 (1H, m), 3.36 (2H, td, J = 11.6, 0.8 Hz), 3.97 (2H, dd, J = 11.8, 4.0 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.65 (1H, brs). Elemental analysis: C ZI H 25 N 3 0 4 S 3 · 0.2 Calculated as ¾0: C, 52.20; H, 5.30; N, 8.70; S, 19.91 (%), Found: C, 52.21; H, 5.34; N, 8.68; S, 20.05 (%).
実施例 4-118 Example 4-118
N- {8- [(3-ァミノプロピル)スルファニル ]-5, 6-ジメチル -3-ォキソ -2, 3-ジヒド口 -1H-イミダゾ [5,1- c] [1,4]チアジン-卜ィリデン }-4-メチルベンゼンスルホンァ ミド トリフルォロ酢酸塩 N- {8-[(3-Aminopropyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-triylidene } -4-Methylbenzenesulfonamide trifluoroacetate
実施例 4- 106で得た化合物 (327 mg, 0.607匪 ol) を TFA (0.5 ml)に溶解し、 室温で 60分間放置した。 反応液をトルエン(10 ml)で希釈して濃縮乾固し、 残 渣に酢酸ェチルを加えて赤色懸濁液を得た。 析出物を濾取し、 酢酸ェチルで洗浄 後、 乾燥して標題化合物 (334 rag, 0.604 mmol)を赤紫色粉末として得た。 The compound (327 mg, 0.607 bandol) obtained in Example 4-106 was dissolved in TFA (0.5 ml) and left at room temperature for 60 minutes. The reaction solution was diluted with toluene (10 ml) and concentrated to dryness. Ethyl acetate was added to the residue to obtain a red suspension. The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (334 rag, 0.604 mmol) as a red-purple powder.
Ή-NMR (200 MHz, 重クロ口ホルム中): δ 1.80 (3Η, q, J- 0.6 Hz), 2.04 (2H, quint, J= 6.9 Hz), 2.25 (3H, brs), 2.43 (3H, s), 2.97 (2H, t, J= 5.6 Hz), 3.06 (2H, t, J= 7.1 Hz), 7.32 (2H, d, J= 8.0 Hz), 7.87 (2H, d, J= 8.4 Hz), 8.33 (3H, brs). Ή-NMR (200 MHz, in double-mouthed form): δ 1.80 (3Η, q, J-0.6 Hz), 2.04 (2H, quint, J = 6.9 Hz), 2.25 (3H, brs), 2.43 (3H, s), 2.97 (2H, t, J = 5.6 Hz), 3.06 (2H, t, J = 7.1 Hz), 7.32 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.4 Hz) , 8.33 (3H, brs).
元素分析値: C2。H23N305S3F3 として計算値; C, 43.47; H, 4.20; N, 10.14; S, 17.41Elemental analysis: C 2. Calculated as H 23 N 3 0 5 S 3 F 3; C, 43.47; H, 4.20; N, 10.14; S, 17.41
(%)、 実測値; C, 43.32; H, 4.21; , 9.84; S, 17.26 (%) . (%), Found; C, 43.32; H, 4.21 ;, 9.84; S, 17.26 (%).
実施例 4-119 Example 4-119
N - {8- [(4 -ヒドロキシブチル)スルファニル] - 5, 6 -ジメチル- 3-ォキソ - 2, 3_ジヒド 口- 1H -イミダゾ [5,1- c] [1,4]チアジン -1-ィリデン }_4-メチルベンゼンスルホン アミド N- {8-[(4-Hydroxybutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3_dihydrone-1H-imidazo [5,1-c] [1,4] thiazine-1 -Ylidene} _4-methylbenzenesulfonamide
実施例 3- で得た化合物(1.26 g, 3.31醒 ol)および炭酸力リゥム (686 mg, 4.97 mmol)の DMF (20 ml)の懸濁液に 80°Cで ¾拌しながら 4-ブロモ- 1-プ夕ノール (608 mg, 3.97 minol)を加え、 反応液を 6時間攪拌した。 この間反応液に 4 -プロ モ- 1-ブタノール (506 ig, 3.31腿 ol)を 2時間毎に 2回加えた。 反応液に 1N塩 酸 (3.4 ml)、 水(6.4 ml) およびメタノール一水(1 :1, 10 ml)を加え、 室温で 60 分間攪拌した。析出物を濾取し、メタノール—水(1:1)、水、メタノール一水(1:1) およびメタノールで洗浄後、 THF および酢酸ェチルに懸濁し、 60°Cに加温して不 溶物を除去した。 可溶分を濃縮し、 メタノールで希釈後、 析出物を濾取し、 メタ ノールで洗浄、 乾燥して標題化合物 (277 mg, 0.611 mmol)を赤橙色粉末として得 た。 A suspension of the compound obtained in Example 3- (1.26 g, 3.31 mmol) and carbonated rim (686 mg, 4.97 mmol) in DMF (20 ml) was stirred at 80 ° C with 4-bromo- 1-Pynool (608 mg, 3.97 minol) was added, and the reaction solution was stirred for 6 hours. During this time, 4-promo-1-butanol (506 ig, 3.31 tmol) was added to the reaction solution twice every two hours. To the reaction solution were added 1N hydrochloric acid (3.4 ml), water (6.4 ml) and methanol / water (1: 1, 10 ml), and the mixture was stirred at room temperature for 60 minutes. The precipitate is collected by filtration, washed with methanol-water (1: 1), water, methanol-water (1: 1) and methanol, suspended in THF and ethyl acetate, and heated to 60 ° C to make it insoluble Things were removed. After concentrating the soluble matter and diluting with methanol, the precipitate was collected by filtration and meta The residue was washed with ethanol and dried to give the title compound (277 mg, 0.611 mmol) as a red-orange powder.
Ή-NMR (200MHz, CDC13) : δ 1.72 (4Η, m), 1.78 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.42 (3H, s), 2.99 (2H, t, J= 7.1 Hz), 3.68 (2H, t, J= 5.9 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.72 (4Η, m), 1.78 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 0.8 Hz), 2.42 (3H, s), 2.99 (2H, t, J = 7.1 Hz), 3.68 (2H, t, J = 5.9 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.64 (1H , brs).
元素分析値: C19H23N304S3 · 0.25H20として計算値: C, 49.81; H, 5.17; N, 9.17; S, 21.00 (%) 、 実測値: C, 49.85; H, 5.09; N, 9.13; S, 21.13 (%) . Elemental analysis: C 19 H 23 N 3 0 4 S 3 · 0.25H 2 0 Calculated: C, 49.81; H, 5.17 ; N, 9.17; S, 21.00 (%), Found: C, 49.85; H , 5.09; N, 9.13; S, 21.13 (%).
融点. 120.5-124.0°C Melting point.120.5-124.0 ° C
実施例 4-120 Example 4-120
N- (5, 6-ジメチル- 3-ォキソ -8- {[4- (テトラヒドロ- 2H-ピラン -2_ィルォキシ)プチ ル]スルファニル } - 2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデ ン)- 4-メチルベンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[4- (tetrahydro-2H-pyran-2_yloxy) butyl] sulfanyl}}-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物(280 mg, 0.734 mmol)および炭酸カリウム(152 mg, 1.10 mmol)の DMF (4.5 ml)の懸濁液に 80°Cで攪拌しながら、 4-ブロモ -1-ブ夕ノール、 3, 4 -ジヒドロ- 2H -ピランおよび CSAから調製した 4-ブロモ- 1-ブチル 2 -テトラヒド ロビラニル エーテル (0.30 ml)を加え、 反応液を 70分間攪拌した。 反応液に 1N塩 酸 (3.4ml) および水(30 ml)を加え、 酢酸ェチル (40.ml)で抽出した。 有機層を水 および飽和食塩水で洗浄し、 乾燥、濃縮した。残渣に酢酸ェチルおよびジェチル 一テルを加え、析出物を集めてジェチルエーテルで洗浄、乾燥して標題化合物 (161 g, 0.299 mmoU 3: 1ァノマー混合物)を赤色粉末として得た。 To a suspension of the compound obtained in Example 3-4 (280 mg, 0.734 mmol) and potassium carbonate (152 mg, 1.10 mmol) in DMF (4.5 ml) was added 4-bromo-1 while stirring at 80 ° C. 4-Bromo-1-butyl 2-tetrahydrobiranyl ether (0.30 ml) prepared from -butanol, 3,4-dihydro-2H-pyran and CSA was added, and the reaction solution was stirred for 70 minutes. 1N Hydrochloric acid (3.4 ml) and water (30 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (40. ml). The organic layer was washed with water and saturated saline, dried and concentrated. Ethyl acetate and getyl ether were added to the residue, and the precipitate was collected, washed with getyl ether, and dried to give the title compound (161 g, 0.299 mmoU 3-1 anomeric mixture) as a red powder.
Ή-NMR (200MHz, CDC13) : δ 1.55 (4Η, ra), 1.74 (4H, m), 1.79 (3H, brs), 2.26 (3H, q, ]= 1.2 Hz), 2.42 (3H, s), 2.99 (2H, t, J= 7.1 Hz), 3.47 (2H, m), 3.78 (2H, m), 4.23 (1/4H, m, ひ—ァノマー), 4.57 (3/4H, brt, J= 3.4 Hz, β -ァノ マ一), 7.29 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). 融点. 105.0- 108. (T Ή-NMR (200MHz, CDC1 3 ): δ 1.55 (4Η, ra), 1.74 (4H, m), 1.79 (3H, brs), 2.26 (3H, q,] = 1.2 Hz), 2.42 (3H, s) , 2.99 (2H, t, J = 7.1 Hz), 3.47 (2H, m), 3.78 (2H, m), 4.23 (1 / 4H, m, hyanoomer), 4.57 (3 / 4H, brt, J = 3.4 Hz, β-anomer), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.64 (1H, brs). Melting point. 105.0- 108. (T
実施例 4-121 Example 4-121
Ή-NMR (200MHz, CDC13) : δ 1.78 ('3Η, s), 2.06 (3H, s), 2.26 (3H, s), 2.41 (3H, s), 2.97 (2H, m), 3.43 (m), 3.78 (m), 4.08 (m), 5,01 (brs), 5.56 (brs), 7.29 (2H, i), 7.84 (2H, m), 9.64 (1H, br). 元素分析値: C19H23N304S3 · 0.25H2Oとして計算値: C, 49.81; H, 5.17; N, 9.17; S, 21.00 ( ) 、 実測値: C, 49.85; H, 5.09; N, 9.13; S, 21.13 (%) . Ή-NMR (200MHz, CDC1 3 ): δ 1.78 ( '3Η, s), 2.06 (3H, s), 2.26 (3H, s), 2.41 (3H, s), 2.97 (2H, m), 3.43 (m ), 3.78 (m), 4.08 (m), 5,01 (brs), 5.56 (brs), 7.29 (2H, i), 7.84 (2H, m), 9.64 (1H, br). Elemental analysis: C 19 H 23 N 3 0 4 S 3 · 0.25H 2 O Calculated: C, 49.81; H, 5.17 ; N, 9.17; S, 21.00 (), Found: C, 49.85; H, 5.09; N, 9.13; S, 21.13 (%).
融点. 120.5-124.0°C Melting point.120.5-124.0 ° C
実施例 4-122 Example 4-122
N_{8-[(4-ク口ロブチル)スルファニル ]-5, 6-ジメチル -3-ォキソ -2, 3-ジヒド口 -1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン卜 4-メチルベンゼンスルホンアミ F N_ {8-[(4-Culobutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrido-1H-imidazo [5, tric] [1,4] thiazine-1- Silent 4-methylbenzenesulfonami F
実施例 3- 4で得た化合物(498 mg, 1.30匪 ol)および炭酸カリウム(180 mg, 1.30 irnnol)の DMF (8 ml)の懸濁液に 80°Cで攪拌しながら卜ブロモ -4-クロロブタン (0.182 ml, 1.57匪 ol)を加え、反応液を 1時間攪拌した。反応液に 1N塩酸 (1.3ml) およびメタノール—水(1:1, 10ml)を加え、 室温で 30分間攪拌した。 析出物を濾取 し、 メタノール一水(1:1)、 水、 メタノール一水(1:1)およびメタノールで洗浄後、 酢酸ェチルに懸濁して不溶物を除去した。 可溶分を濃縮し、 メ夕ノ一ルで希釈後、 析出物を濾取し、 メタノールで洗浄、 乾燥して標題化合物 (307 mg, 0.650 mmol) を灰紫色粉末として得た。 To a suspension of the compound (498 mg, 1.30 bandol) obtained in Example 3-4 and potassium carbonate (180 mg, 1.30 irnnol) in DMF (8 ml) was stirred at 80 ° C at 80 ° C while tribromo-4-. Chlorobutane (0.182 ml, 1.57 ol) was added and the reaction solution was stirred for 1 hour. 1N Hydrochloric acid (1.3 ml) and methanol-water (1: 1, 10 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1), water, methanol-water (1: 1) and methanol, and suspended in ethyl acetate to remove insolubles. The soluble matter was concentrated, diluted with methanol, and the precipitate was collected by filtration, washed with methanol, and dried to obtain the title compound (307 mg, 0.650 mmol) as a gray purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.79 (3Η, brs), 1.87 (4H, m), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.97 (2H, brt, J= 7.0 Hz), 3.55 (2H, brt, J= 5.8 Hz), 7.29 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J- 8.0 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.79 (3Η, brs), 1.87 (4H, m), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 2.97 (2H, brt, J = 7.0 Hz), 3.55 (2H, brt, J = 5.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J- 8.0 Hz), 9.63 (1H, brs).
元素分析値: C19H22N303S3C1 ·0.2Η20として計算値: C, 47.98; Η, 4.75; Ν, 8.83; S, 20.22; Cl, 7.45 (%) 、 実測値: C, 47.99; H, 4.66; N, 8.69; S, 20.36; C1, 7.10 (%) . Elemental analysis: C 19 H 22 N 3 0 3 S 3 C1 · 0.2Η 2 0 Calculated: C, 47.98; Η, 4.75 ; Ν, 8.83; S, 20.22; Cl, 7.45 (%), Found: C, 47.99; H, 4.66; N, 8.69; S, 20.36; C1, 7.10 (%).
実施例 4-123 Example 4-123
N - {8_ [ (4-ョ―ドブチル)スルファニル] -5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ - 1H-イミダゾ [5,1 - c] [1,4]チアジン- 1 -ィリデン }- 4-メチルベンゼンスルホンァ Sド N-{8_ [(4-odobutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene}-4-Methylbenzenesulfone S
実施例 4- 122 で得た化合物(0.73 g, 1.55腿 ol)およびヨウ化ナトリウム(464 mg, 3.09 mmol)の 2-ブタノン(20 ml)の溶液を 80 で 3時間攪拌した。 反応液に 酢酸ェチル (60 ml)を加えて混和し、水および飽和食塩水で洗浄し、乾燥、濃縮後、 残渣にメタノールを加えた。 析出物を濾取し、 メタノールで洗浄、 乾燥して粗製 の標題化合物 (純度 89 , 712 g, 1.12 mmol)を赤紫色粉末として得た。 A solution of the compound obtained in Example 4-122 (0.73 g, 1.55 mol) and sodium iodide (464 mg, 3.09 mmol) in 2-butanone (20 ml) was stirred at 80 for 3 hours. Ethyl acetate (60 ml) was added to the reaction mixture, mixed, washed with water and saturated saline, dried and concentrated, and methanol was added to the residue. The precipitate is collected by filtration, washed with methanol, dried and crude The title compound (purity 89, 712 g, 1.12 mmol) was obtained as a red-purple powder.
¾一 NMR (200MHz, CDC13) : δ 1.79 (3Η, q, J= 1.2 Hz), 1.87 (4H, ), 2.26 (3H, q, J= 0.8 Hz), 2.42 (3H, s), 2.95 (2H, brt, J= 7.2 Hz), 3.18 (2H, brt, J= 6.4 Hz), 7.30 (2H, d, J= 8.8 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). 実施例 4-124 ¾ one NMR (200MHz, CDC1 3): δ 1.79 (3Η, q, J = 1.2 Hz), 1.87 (4H,), 2.26 (3H, q, J = 0.8 Hz), 2.42 (3H, s), 2.95 ( 2H, brt, J = 7.2 Hz), 3.18 (2H, brt, J = 6.4 Hz), 7.30 (2H, d, J = 8.8 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs). Example 4-124
N- (5, 6-ジメチル- 3-ォキソ -8- { [2- (1H-ピロール-卜ィル)ェチル]スルファニ ル}- 2, 3 -ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン - 1-ィリデン)-4-メチルベ ンゼンスルホンアミド N- (5,6-Dimethyl-3-oxo-8-{[2- (1H-pyrrole-tolyl) ethyl] sulfanyl} -2,3-dihydro-1H-imidazo [5, tric] [ [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物(276 mg, 0.723 mmol)および炭酸カリウム(150 mg, 1.09 腿 ol)の DMF (4.5 ml)の懸濁液に 60°Cで攪拌しながら卜(2-ブロモェチル)ピロ一 ル (151 mg, 0.868 mmol)を加え、 反応液を 60°Cで 3時間攪拌した。 この間反応 液に 1- (2-プロモェチル)ピロール (30 mg, 0.174 mmol)を加えた。反応液に 1 塩 酸 (1.5 ml)およびメタノール一水(1:1, 3 ml)を加え、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール一水 α:1 および 2:1)で洗浄後、 乾燥して標題化合 物 (247 mg, 0.520匪 ol)を紫色粉末として得た。 ' A suspension of the compound obtained in Example 3-4 (276 mg, 0.723 mmol) and potassium carbonate (150 mg, 1.09 tmol) in DMF (4.5 ml) was stirred at 60 ° C at 60 ° C to give 2-bromoethyl. ) Pyrrol (151 mg, 0.868 mmol) was added, and the reaction solution was stirred at 60 ° C for 3 hours. During this time, 1- (2-bromoethyl) pyrrole (30 mg, 0.174 mmol) was added to the reaction solution. Monohydrochloric acid (1.5 ml) and methanol-water (1: 1, 3 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water α: 1 and 2: 1), and dried to give the title compound (247 mg, 0.520 bandol) as a purple powder. '
'H-NMR (200MHz, CDC13) : δ 1.79 (3Η, q, J= 1.0 Hz), 2.26 (3H, q, J= 1.0 Hz), 2.43 (3H, s), 3.26 (2H, t, J= 7.2 Hz), 4.14 (2H, i, J= 7.1 Hz), 6.15 (2H, t, J= 2.1 Hz), 6.63 (2H, t, J= 2.0 Hz), 7.29 (2H, d, J= 8.0 Hz), 7.83 (2H, d, J二 8.0 Hz), 9.64 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 1.79 (3Η, q, J = 1.0 Hz), 2.26 (3H, q, J = 1.0 Hz), 2.43 (3H, s), 3.26 (2H, t, J = 7.2 Hz), 4.14 (2H, i, J = 7.1 Hz), 6.15 (2H, t, J = 2.1 Hz), 6.63 (2H, t, J = 2.0 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J2 8.0 Hz), 9.64 (1H, brs).
元素分析値: C21H22N403S3として計算値: C, 53.14;. H, 4.67; N, 11.80; S, 20.27 (%) 、 実測値: C, 53.06; H, 4.65; N, 11.56; S, 20.29 (%) . Elemental analysis: Calculated for C 21 H 22 N 4 0 3 S 3 : C, 53.14 ;. H, 4.67; N, 11.80; S, 20.27 (%), found: C, 53.06; H, 4.65; N , 11.56; S, 20.29 (%).
融点. 168.0-169.5°C Melting point.168.0-169.5 ° C
実施例 4-125 Example 4-125
N -(5, 6-ジメチル- 3-ォキソ - 8 - { [3_ (1H -ピロール-卜ィル)プロピル]スルファニ ル } -2, 3-ジヒドロ- 1H -イミダゾ [5, 1- c] [1, 4]チアジン -卜ィリデン) - 4_メチルベ ンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[3_ (1H-pyrrole-tolyl) propyl] sulfanyl} -2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-triylidene)-4_methylbenzenesulfonamide
実施例 3-4で得た化合物(261 mg, 0.684腿 )および炭酸カリウム(142 mg, 1.03 mmol)の DMF (4.0 ml)の懸濁液に 60°Cで攪拌しながら卜(3-ブロモプロピル)ピロ ール (154 mg, 0.821 画 ol)を加え、 反応液を 60 で 3.5時間攪拌した。 この間 反応液に 1 -(3 -ブロモプロピル)ピロール (27 rag, 0.144 mmol)を加えた。 反応液 に 1N塩酸 (1.4 ml)およびメタノール一水(1:1, 3 ml)を加え、 室温で 30分間攪 拌した。 析出物を濾取し、 メタノール一水(1:1 および 2:1)で洗浄後、 メタノー ルから結晶化して標題化合物 (283 mg, 0.579匪 ol)を紫色粉末として得た。 To a suspension of the compound obtained in Example 3-4 (261 mg, 0.684 thigh) and potassium carbonate (142 mg, 1.03 mmol) in DMF (4.0 ml) was stirred at 60 ° C while adding tri (3-bromopropyl). ) Pyrrol (154 mg, 0.821 ol) was added, and the reaction solution was stirred at 60 for 3.5 hours. During this time 1- (3-Bromopropyl) pyrrole (27 rag, 0.144 mmol) was added to the reaction solution. 1N Hydrochloric acid (1.4 ml) and methanol-water (1: 1, 3 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1 and 2: 1), and then crystallized from methanol to give the title compound (283 mg, 0.579 bandol) as a purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.76 (3Η, q, J= 0.8 Hz), 2.11 (3H, quint, J= 6.7 Hz), 2.25 (3H, q, J= 0.6 Hz), 2.41 (3H, s), 2.83 (2H, t, J= 7.1 Hz), 3.97 (2H, t, J= 6.4 Hz), 6.15 (2H, t, J= 2.0 Hz), 6.60 (2H, t, J= 2.0 Hz), 7.29 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.65 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.76 (3Η, q, J = 0.8 Hz), 2.11 (3H, quint, J = 6.7 Hz), 2.25 (3H, q, J = 0.6 Hz), 2.41 (3H , s), 2.83 (2H, t, J = 7.1 Hz), 3.97 (2H, t, J = 6.4 Hz), 6.15 (2H, t, J = 2.0 Hz), 6.60 (2H, t, J = 2.0 Hz) ), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.65 (1H, brs).
元素分析値: C22H24N403S3として計算値: C, 54.07; H, 4.95; N, 11.47; S, 19.69Elemental analysis: C 22 H 24 N 4 0 3 S 3 Calculated: C, 54.07; H, 4.95 ; N, 11.47; S, 19.69
( ) 、 実測値: C, 54.01; H, 4.94; N, 11.09; S, 19.64 (%) . (), Found: C, 54.01; H, 4.94; N, 11.09; S, 19.64 (%).
融点. 194.5-195.0°C Melting point.194.5-195.0 ° C
実施例 4-126 Example 4-126
N - {8- [ (2-シク口へキシルェチル)スルファニル] -5, 6-ジメチル -3-ォキソ -2, 3-ジ ヒドロ -1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン卜 4-メチルベンゼンスル ホンアミド N- {8-[(2-cyclohexylethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine- 1-ylidene 4-methylbenzenesulfonamide
実施例 3-4で得た化合物(275 mg, 0.721 mmol)および炭酸カリウム(149 mg, 1.08 龍 ol)の DMF (4.5 ml)の懸濁液に室温で攪拌しながら臭化 2 -シクロへキシルェチ ル(155 mg, 0.792 mmol)を加え、 反応液を 80 で 1.5時間攪拌した。 反応液に 1N 塩酸 (1.44 ml)およびメタノール一水(1:1, 5 ml)を加え、 室温で 30分間攪拌し た。 析出物を濾取し、 メタノール—水(1:1) およびメタノールで洗浄後、 乾燥し て標題化合物 (225 mg, 0.458删 ol)を紫褐色粉末として得た。 A suspension of the compound obtained in Example 3-4 (275 mg, 0.721 mmol) and potassium carbonate (149 mg, 1.08 dragonol) in DMF (4.5 ml) was stirred at room temperature with stirring at room temperature for 2-cyclohexylethyl bromide. (155 mg, 0.792 mmol) was added, and the reaction solution was stirred at 80 for 1.5 hours. 1N Hydrochloric acid (1.44 ml) and methanol / water (1: 1, 5 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (225 mg, 0.458 mol) as a purple-brown powder.
Ή-NMR (200MHz, CDC13) : δ 0.93 (2Η, brq, J= 10.6 Hz), 1.18 (2H, brd, J= 8.4 Hz), 1.27 (3H, m), 1.56 (2H, q, J= 8.0 Hz), 1.71 (4H, brd, J= 9.8 Hz), 1.78 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 2.95 (2H, t, J= 7.7 Hz), 7.28 (2H, d, J= 8.6 Hz), 7.85 (2H, d, J= 8.6 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.93 (2Η, brq, J = 10.6 Hz), 1.18 (2H, brd, J = 8.4 Hz), 1.27 (3H, m), 1.56 (2H, q, J = 8.0 Hz), 1.71 (4H, brd, J = 9.8 Hz), 1.78 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 2.95 (2H, t, J = 7.7 Hz), 7.28 (2H, d, J = 8.6 Hz), 7.85 (2H, d, J = 8.6 Hz), 9.63 (1H, brs).
元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56Elemental analysis: C 23 H 29 N 3 0 3 S 3 Calculated: C, 56.18; H, 5.94 ; N, 8.55; S, 19.56
(%) 、 実測値: C, 56.14; H, 5.90; N, 8.45; S, 19.70 (%) . (%), Found: C, 56.14; H, 5.90; N, 8.45; S, 19.70 (%).
融点. 166.2-167.2°C Melting point. 166.2-167.2 ° C
実施例 4-127 N - (5, 6-ジメチル- 3-ォキソ -8- { [ (3-テトラヒドロフラニル)メチル]スルファニ ル卜 2,3-ジヒドロ _m-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン)-4-メチルベ ンゼンスルホンアミド Example 4-127 N- (5,6-dimethyl-3-oxo-8-{[(3-tetrahydrofuranyl) methyl] sulfanyl 2,3-dihydro_m-imidazo [5,1-c] [1,4] thiazine- Trilidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物(300 mg, 0.786 mmol)および炭酸カリウム(163 mg, 1.18 mmol)の DMF (5 ml)の懸濁液に 80°Cで攪拌しながら参考例 91で得たメタンスル ホン酸テトラヒドロフラン- 3-メチル(170 mg, 0.944 mmol)を加え、 反応液を 80°C で 4.5時間攪拌した。 この間反応液にヨウ化ナトリウム(118 mg, 0.786 匪 ol)お よびメタンスルホン酸テトラヒドロフラン- 3-メチル(34 mg, 0.157醒 ol)を 2回 加えた。 反応液に 1N塩酸 (1.60 ml)および水(3 ml)を加え、 室温で 30分間攪拌 した。 析出物を濾取し、 メタノール一水(1:1) およびメタノールで洗浄後、 乾燥 して標題化合物 (224 rag, 0.481 mmol)を紫色粉末として得た。 A suspension of the compound obtained in Example 3-4 (300 mg, 0.786 mmol) and potassium carbonate (163 mg, 1.18 mmol) in DMF (5 ml) was obtained in Reference Example 91 while stirring at 80 ° C. Tetrahydrofuran-3-methyl methanesulfonate (170 mg, 0.944 mmol) was added, and the reaction solution was stirred at 80 ° C for 4.5 hours. During this time, sodium iodide (118 mg, 0.786 ol) and tetrahydrofuran-3-methyl methanesulfonate (34 mg, 0.157 ol) were added twice to the reaction mixture. 1N Hydrochloric acid (1.60 ml) and water (3 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (224 rag, 0.481 mmol) as a purple powder.
!H-NMR (200MHz, CDC13) : d 1.66 (1H, m), 1.79 (3H, q, J= 1.0 Hz), 2.14 (1H, ddt, J= 13.0, 7.6, 5.6 Hz), 2.26 (3H, q, J- 1.0 Hz), 2.42 (3H, s), 2.52 (1H, brquint, J= 6.9 Hz), 2.99 (2H, d, J= 7.2 Hz), 3.53 (1H, dd, J= 8.8, 5.6 Hz), 3.77 (1H, brq, J= 7.8 Hz), 3.86 (1H, dd, J= 8.8, 6.8 Hz), 3.89 (1H, dt, J= 5.5, 8.6 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). 元素分析値: C2。H23N304S3として計算値: C, 51.59; H, 4.98; N, 9.02; S, 20.66! H-NMR (200MHz, CDC1 3): d 1.66 (1H, m), 1.79 (3H, q, J = 1.0 Hz), 2.14 (1H, ddt, J = 13.0, 7.6, 5.6 Hz), 2.26 (3H , q, J- 1.0 Hz), 2.42 (3H, s), 2.52 (1H, brquint, J = 6.9 Hz), 2.99 (2H, d, J = 7.2 Hz), 3.53 (1H, dd, J = 8.8, 5.6 Hz), 3.77 (1H, brq, J = 7.8 Hz), 3.86 (1H, dd, J = 8.8, 6.8 Hz), 3.89 (1H, dt, J = 5.5, 8.6 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 ( 2H, d, J = 8.4 Hz), 9.65 (1H, brs) elemental analysis:. C 2. H 23 N 3 0 4 S 3 Calculated: C, 51.59; H, 4.98 ; N, 9.02; S, 20.66
(%) 、 実測値: C, 51.37; H, 5.17; , 9.04; S, 20.69 (%) . (%), Found: C, 51.37; H, 5.17;, 9.04; S, 20.69 (%).
融点. 199.0- 201. O : Melting point. 199.0- 201. O:
実施例 4-128 Example 4-128
N-{5, 6-ジメチル- 3 -ォキソ -8 - [ (2 -テトラヒドロ -2H-ピラン _4 -ィルェチル)スル ファニル] -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }-4 -メ 実施例 3-4で得た化合物(284 mg, 0.744 mmol), 参考例 87で得たメタンスルホ ン酸 2- (テトラヒドロピラン- 4-ィル)ェチル(186 mg, 0.893 mmol)および炭酸力 リウム(154mg, 1.128 mmol)の DMF (4.5 ml)の懸濁液を 80°Cで 3時間攪拌した。 この間反応液にメ夕ンスルホン酸 2- (テトラヒドロビラン- 4-ィル)ェチル(31 mg, 0.148膽 ol)を 2回加えた。反応液に IN塩酸 (2.0ml)およびメタノ一ル—水(1:1, 5 ml)を加え、室温で 30分間攪拌した。析出物を濾取し、メタノール—水(1:1) お よびメタノールで洗浄後、 乾燥し ΊΓ標題化合物 (226 Dig, 0.458膽 ol)を灰赤色粉 末として得た。 N- {5,6-dimethyl-3-oxo-8-[(2-tetrahydro-2H-pyran_4-ylethyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazin-1-ylidene} -4-mer The compound obtained in Example 3-4 (284 mg, 0.744 mmol), 2- (tetrahydropyran-4-yl) methanesulfonate obtained in Reference Example 87 A suspension of ethyl ester (186 mg, 0.893 mmol) and potassium carbonate (154 mg, 1.128 mmol) in DMF (4.5 ml) was stirred at 80 ° C for 3 hours. During this time, 2- (tetrahydrobilan-4-yl) ethyl methyl sulfonate (31 mg, 0.148 bunol) was added twice to the reaction solution. IN hydrochloric acid (2.0 ml) and methanol-water (1: 1, 5 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate is collected by filtration, and methanol-water (1: 1) and After washing with methanol and drying, the title compound (226 Dig, 0.458 benzyl) was obtained as a gray-red powder.
'H-NMR (200MHz, CDC13) : δ 1.32 (2Η, m), 1.61 (5H, m), 1.79 (3H, q, J= 1.2 Hz), 2.27 (3H, q, J= 0.6 Hz), 2.42 (3H, s), 2.96 (2H, t, J= 7.3 Hz) , 3.36 (1H, dt, J= 1.8, 11.7 Hz), 3.97 (2H, brdd, J= 11.4, 4.0 Hz), 7.29 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.67 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 1.32 (2Η, m), 1.61 (5H, m), 1.79 (3H, q, J = 1.2 Hz), 2.27 (3H, q, J = 0.6 Hz), 2.42 (3H, s), 2.96 (2H, t, J = 7.3 Hz), 3.36 (1H, dt, J = 1.8, 11.7 Hz), 3.97 (2H, brdd, J = 11.4, 4.0 Hz), 7.29 (2H , d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.67 (1H, brs).
元素分析値: C22H27N304S3として計算値: C, 53.53; H, 5.51; N, 8.51; S, 19.49Elemental analysis: C 22 H 27 N 3 0 4 S 3 Calculated: C, 53.53; H, 5.51 ; N, 8.51; S, 19.49
(%) 、 実測値: C, 53.16; H, 5.63; N, 8.40; S, 19.47 (%) . (%), Found: C, 53.16; H, 5.63; N, 8.40; S, 19.47 (%).
融点. 166.0- 167. (TC Melting point. 166.0- 167. (TC
実施例 4-129 Example 4-129
N- [5, 6-ジメチル -3-ォキソ -8- (テトラヒドロ -2H-ピラン- 4-ィルスルファ二 ル) -2, 3-ジヒド口- 1H-ィミダゾ [5,卜 c] [1, 4]チ Tジン-トイリデン] -4-メチルべ ンゼンスルホンアミド N- [5,6-Dimethyl-3-oxo-8- (tetrahydro-2H-pyran-4-ylsulfanyl) -2,3-dihydrido-1H-imidazo [5, tric] [1,4] Thizine-tolylidene] -4-methylbenzenesulfonamide
実施例 3 - 4で得た化合物(337 mg, 0.883 mmol)、 ヨウ化ナトリウム(265 mg, 1.77 mmol)および tert-ブトキシカリウム(175 mg, 1.33腿 ol)の N-メチルピロリドン (5.0 ml)の懸濁液を室温で攪拌しながら 4 -クロロテトラヒドロピラン(0.12 ml, 1.06 mmol)を加えた。 反応液を 100°C で 1.5 時間、 140°C で 3 時間攪拌した。 反応液に 1N塩酸 (1.77 ml)および水(35 ml)を加えて酢酸ェチル -ジイソプロピ ルエーテル(30:5, 35 ml)で抽出した。 有機層を水および飽和食塩水で洗浄し、 乾 燥、 濃縮した。 残渣をシリカゲル(10 g)のクロマトグラフィー (酢酸ェチル—へ キサン、 3:7-5:5)に付し、酢酸ェチルーへキサン(35:65- 50:50)の画分を濃縮後、 メタノールから結晶化して標題化合物 (19 mg, 0.041 腿 ol)を赤紫色粉末として 得た。 Compound (337 mg, 0.883 mmol) obtained in Example 3-4, sodium iodide (265 mg, 1.77 mmol) and potassium tert-butoxide (175 mg, 1.33 mol) of N-methylpyrrolidone (5.0 ml) While stirring the suspension at room temperature, 4-chlorotetrahydropyran (0.12 ml, 1.06 mmol) was added. The reaction solution was stirred at 100 ° C for 1.5 hours and at 140 ° C for 3 hours. 1N Hydrochloric acid (1.77 ml) and water (35 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate-diisopropyl ether (30: 5, 35 ml). The organic layer was washed with water and saturated saline, dried and concentrated. The residue was subjected to silica gel (10 g) chromatography (ethyl acetate-hexane, 3: 7-5: 5), and the ethyl acetate-hexane (35: 65-50: 50) fraction was concentrated. The title compound (19 mg, 0.041 tmol) was obtained as red-purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.75 (2Η, brq, J= 12 Hz), 1.79 (3H, q, J= 1.0 Hz), 2.00 (2H, brq, J= 12 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.43 (3H, s), 3.47 (2H, dt, J= 2.2, 11.2 Hz), 3.53 (1H, m), 3.95 (2H, dt, J= 11.4, 7.6 Hz), 7.30 (2H, d, J= 8.2 Hz), 7.86 (2H, d, J= 8.2 Hz), 9.66 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.75 (2Η, brq, J = 12 Hz), 1.79 (3H, q, J = 1.0 Hz), 2.00 (2H, brq, J = 12 Hz), 2.26 (3H , q, J = 0.8 Hz), 2.43 (3H, s), 3.47 (2H, dt, J = 2.2, 11.2 Hz), 3.53 (1H, m), 3.95 (2H, dt, J = 11.4, 7.6 Hz) , 7.30 (2H, d, J = 8.2 Hz), 7.86 (2H, d, J = 8.2 Hz), 9.66 (1H, brs).
元素分析値: C2。H23N304S3として計算値: C, 51.59; H, 4.98; N, 9.02; S, 20.66 (%) 、 実測値: C, 51.29; H, 4.98; N, 8.98; S, 20.79 (%) · 融点. 195.5-201.0°C Elemental analysis: C 2. H 23 N 3 0 4 S 3 Calculated: C, 51.59; H, 4.98 ; N, 9.02; S, 20.66 (%), Found: C, 51.29; H, 4.98 ; N, 8.98; S, 20.79 ( %) · Melting point.195.5-201.0 ° C
実施例 4- 130 Example 4-130
N - (8- {[2- (4-フルオロフェニル) -2-プロぺニル]スルファニルト 5, 6_ジメチル- 3 - ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン )-4-メチル ベンゼンスルホンアミド N- (8-{[2- (4-fluorophenyl) -2-propenyl) sulfanylto 5,6_dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [ 1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3- 4で得た化合物(329 mg, 0.862 mmol) およびトリェチルァミン(0· 30 ml, 2.16腿01)の 1:1^ (6.6 ml)懸濁液に、 室温で攪拌しながら参考例 88で得た 3 -ブロモ -2- (4-フルオロフェニル) プロペン(純度 70%, 342 mg, 1.03腿 ol)を加 えた。この間反応液に 3-ブロモ- 2_(4-フルオロフェニル) プロペン(純度 70%, 165 mg, 0.52醒 ol)を加えた。 反応液を室温で 7.5 時間攪拌した。 反応液に 1N塩酸 (1.3 ml)およびメタノール—水(1:1, 6 ml)を加え、 室温で 30分間攪拌した。 析 出物を濾取し、 メタノール一水(1:1) およびメタノールで洗浄後、 乾燥して標題 化合物 (288 mg, 0.559匪 ol)を赤紫色粉末として得た。 Reference Example 88 was added to a suspension of the compound (329 mg, 0.862 mmol) obtained in Example 3-4 and 1: 1 ^ (6.6 ml) of triethylamine (0.330 ml, 2.16 thigh 01) at room temperature while stirring. The 3-bromo-2- (4-fluorophenyl) propene (purity 70%, 342 mg, 1.03 tmol) obtained in Step 1 was added. During this time, 3-bromo-2_ (4-fluorophenyl) propene (purity 70%, 165 mg, 0.52 mmol) was added to the reaction solution. The reaction was stirred at room temperature for 7.5 hours. 1N Hydrochloric acid (1.3 ml) and methanol-water (1: 1, 6 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-one water (1: 1) and methanol, and dried to give the title compound (288 mg, 0.559 bandol) as a red-purple powder.
^-NMR (200MHz, CDC13) : δ 1.77 (3Η, brs), 2.25 (3H, brs), 2.40 (3H, s), 4.02 (2H, s), 5.41 (1H, brs), 5.51 (1H, brs), 7.03 (2H, t, J= 8.6 Hz), 7.19 (2H, d, J= 8.4 Hz), 7.37 (2H, dd, J= 8.8, 5.4 Hz), 7.75 (2H, d, J= 8.0 Hz) , 9.59 (1H, brs). ^ -NMR (200MHz, CDC1 3) : δ 1.77 (3Η, brs), 2.25 (3H, brs), 2.40 (3H, s), 4.02 (2H, s), 5.41 (1H, brs), 5.51 (1H, brs), 7.03 (2H, t, J = 8.6 Hz), 7.19 (2H, d, J = 8.4 Hz), 7.37 (2H, dd, J = 8.8, 5.4 Hz), 7.75 (2H, d, J = 8.0 Hz), 9.59 (1H, brs).
元素分析値: C24H22N303S3Fとして計算値: C, 55.90; H, 4.30; , 8.15; S, 18.66; F, 3.68 (%) 、 実測値: C, 55.51; H, 4.15; N, 7.96; S, 19.02; F, 3.52 (%) . 融点. 154.5-156. Elemental analysis: C 24 H 22 N 3 0 3 S 3 F Calculated: C, 55.90; H, 4.30;, 8.15; S, 18.66; F, 3.68 (%), found: C, 55.51; H, 4.15; N, 7.96; S, 19.02; F, 3.52 (%). Melting point. 154.5-156.
実施例 4-131 Example 4-131
N- { 5, 6-ジメチル- 3-ォキソ _8- [ (1 _プロピルプチル)スルファニル ] -2, 3 -ジヒドロ - 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン }-4-メチルベンゼンスルホンァ ミド N- {5,6-dimethyl-3-oxo_8-[(1-propylbutyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene } -4-Methylbenzenesulfonamide
実施例 3-4で得た化合物 (310 mg, 0.813匪 ol)および炭酸カリウム(168 mg, 1.22 mmol)の DMF (5 ml)の懸濁液に 80°Cで攪拌しながら 4-ョードヘプタン(純度 83¾, 0.104 ml, 0.49匪 ol)を 30分毎に 2回加えた。 さらに 4 -ョ一ドヘプタン(純 度 83%, 0.20 ml, 0.094匪 ol)を加えて反応液を 30分間攪拌した。 反応液に 1N塩 酸 (1.63ml)およびメタノール一水 (2:1, 4 ml)を加え、室温で 14時間攪拌した。 析出物を濾取し、 メタノール一水 (1:1)'およびメタノールで洗浄後、 乾燥して標 題化合物 (128 mg, 0.267 匪 ol)を紫色粉末として得た。 A suspension of the compound obtained in Example 3-4 (310 mg, 0.813 bandol) and potassium carbonate (168 mg, 1.22 mmol) in DMF (5 ml) was stirred at 80 ° C while stirring at a temperature of 80 ° C. 83¾, 0.104 ml, 0.49 marl ol) were added twice every 30 minutes. 4-Heptoheptane (purity 83%, 0.20 ml, 0.094 ol) was further added, and the reaction solution was stirred for 30 minutes. 1N Hydrochloric acid (1.63 ml) and methanol-water (2: 1, 4 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 14 hours. The precipitate was collected by filtration, washed with methanol / water (1: 1) 'and methanol, and dried to give the title compound (128 mg, 0.267 ol) as a purple powder.
Ή-NMR (200MHz, CDC13) : δ 0.92 (6Η, ί, J= 7.2 Hz), 1.42 (4H, brq, J= 7.4 Hz), 1.62 (4H, dt, J= 8.4, 3.8 Hz), 1.79 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 3.39 (1H, quint, J= 6.4 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.86 (2H, d, = 8.4 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.92 (6Η, ί, J = 7.2 Hz), 1.42 (4H, brq, J = 7.4 Hz), 1.62 (4H, dt, J = 8.4, 3.8 Hz), 1.79 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 3.39 (1H, quint, J = 6.4 Hz), 7.27 (2H, d, J = 8.0 Hz), 7.86 (2H, d, = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05Elemental analysis: C 22 H 29 N 3 0 3 S 3 Calculated: C, 55.09; H, 6.09 ; N, 8.76; S, 20.05
(%) 、 実測値: C, 55.16; H, 6.06; N, 8.73; S, 20.08 (%) . (%), Found: C, 55.16; H, 6.06; N, 8.73; S, 20.08 (%).
融点. 128.5-129.5°C Melting point.128.5-129.5 ° C
実施例 4-132 Example 4-132
N - {5, 6-ジメチル- 8- [(卜メチルへキシル)スルファニル ]-3-ォキソ -2, 3-ジヒドロ -1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン }-4-メチルベンゼンスルホンァ ミド N- {5,6-dimethyl-8-[(trimethylhexyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene } -4-Methylbenzenesulfonamide
実施例 3-4で得た化合物 (267 mg, 0.697 mmol)および炭酸カリウム 45 mg, 1.05匪 ol)の DMF (4 ml)の懸濁液に 80 で攪拌しながら 2-ョ一ドヘプタン(純度 87¾, 109 mg, 0.42 顏 ol)を 90分毎に 2回加えた後、 反応液を 3時間攪拌した。 反応液に 1N塩酸 (1.4 ml)およびメタノール一水(2:1, 5 ml)を加え、.室温で 30 分間攪拌した。析出物を濾取し、 メタノール一水(2:1 および 1:1) およびメ夕ノ —ルで洗浄後、乾燥して標題化合物 (195 mg, 0.407mmol)を紫色粉末として得た。 Ή-NMR (200MHz, CDC13) : δ 0.90 (3Η, t, J= 6.6 Hz), 1.25-1.42 (6H, m), 1.37 (3H, d, J= 6.6 Hz), 1.63 (2H, quint, J= 7.2 Hz), 1.79 (3H, q, J= 0.8 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H5 s), 3.45 (1H, q, J= 6.6 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). To a suspension of the compound obtained in Example 3-4 (267 mg, 0.697 mmol) and potassium carbonate (45 mg, 1.05 marl) in DMF (4 ml) was stirred at 80 at 80 ° C. while stirring at 2-hydroxyheptane (purity 87¾). , 109 mg, 0.42 mol) was added twice every 90 minutes, and the reaction solution was stirred for 3 hours. 1N Hydrochloric acid (1.4 ml) and methanol / water (2: 1, 5 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (2: 1 and 1: 1) and methanol, and dried to give the title compound (195 mg, 0.407 mmol) as a purple powder. Ή-NMR (200MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 6.6 Hz), 1.25-1.42 (6H, m), 1.37 (3H, d, J = 6.6 Hz), 1.63 (2H, quint, J = 7.2 Hz), 1.79 (3H, q, J = 0.8 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H 5 s), 3.45 (1H, q, J = 6.6 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05 (%) 、 実測値: C, 54.96; H, 5.82; N, 8.55; S, 20.14 (%) . Elemental analysis: C 22 H 29 N 3 0 3 S 3 Calculated: C, 55.09; H, 6.09 ; N, 8.76; S, 20.05 (%), Found: C, 54.96; H, 5.82 ; N, 8.55; S, 20.14 (%).
融点. 140.5-141.5°C 140.5-141.5 ° C
実施例 4-133 Example 4-133
N- {8- [ (1-ェチルペンチル)スルファニル] -5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ - 1H-イミダゾ [5, l-c] [1, 4]チアジン- 1 -ィリデン }-4-' ミド N- {8-[(1-Ethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-1-ylidene}- Four-' Mid
実施例 3-4で得た化合物 (285 mg, 0.747腿 ol)および炭酸カリウム(155 mg, 1.12 mmol)の DMF (4.5 ml)の懸濁液に 80°Cで攪拌しながら 3-ョードヘプタン(純 度 90%, 113 mg, 0.45龍 ol)を 30分毎に 2回加えた後、反応液を 2時間攪拌した。 反応液に 1N塩酸 (1.5 ml)およびメタノール—水(2:1, 5 ml)を加え、 室温で 30 分間攪拌した。析出物を濾取し、 メタノール—水(2:1 および 1:1) およびメタノ ールで洗浄後、 乾燥して標題化合物 (190 mg, 0.382雇 ol)を紫色粉末として得た Ή-NMR (200MHz, CDC13) : 6 0.90 (3H, t, J= 7.0 Hz), 0.97 (3H, t, J= 7.5 Hz), 1.35 (4H, m), 1.68 (4H, m), 1.79 (3H, q, J= 1.0 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 3.32 (1H, quint, J= 6.4 Hz), 7.27 (2H, d, J= 7.8 Hz), 7.86 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). A suspension of the compound obtained in Example 3-4 (285 mg, 0.747 mol) and potassium carbonate (155 mg, 1.12 mmol) in DMF (4.5 ml) was stirred at 80 ° C at 80 ° C with stirring for 3-heptoheptane (pure). 90%, 113 mg, 0.45 dragonol) was added twice every 30 minutes, and the reaction solution was stirred for 2 hours. 1N Hydrochloric acid (1.5 ml) and methanol-water (2: 1, 5 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (2: 1 and 1: 1) and methanol, and dried to give the title compound (190 mg, 0.382 雇 ol) as a purple powder, Ή-NMR ( 200MHz, CDC1 3): 6 0.90 (3H, t, J = 7.0 Hz), 0.97 (3H, t, J = 7.5 Hz), 1.35 (4H, m), 1.68 (4H, m), 1.79 (3H, q , J = 1.0 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 3.32 (1H, quint, J = 6.4 Hz), 7.27 (2H, d, J = 7.8 Hz), 7.86 (2H, d, J = 8.0 Hz), 9.64 (1H, brs).
元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05Elemental analysis: C 22 H 29 N 3 0 3 S 3 Calculated: C, 55.09; H, 6.09 ; N, 8.76; S, 20.05
(%) 、 実測値: C, 55.02; H, 5.88; N, 8.55; S, 20.19 (%) . (%), Found: C, 55.02; H, 5.88; N, 8.55; S, 20.19 (%).
融点. 0.0- 140.5°C Melting point.0.0-140.5 ° C
実施例 4-134 Example 4-134
N- { 5, 6-ジメチル -3-ォキソ -8- [ (2-プロピルべンチル)スルファニル ]-2, 3-ジヒド ロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン卜 4-メチルベンゼンスルホン アミド N- {5,6-dimethyl-3-oxo-8-[(2-propylbenzyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1 -Ilident 4-methylbenzenesulfonamide
実施例 3-4で得た化合物 (287 mg, 0.752 mmol)および炭酸カリウム(156 mg, 1.13 mmol)の DMF (4.5 ml)の懸濁液に 80°Cで攪拌しながら参考例 89で得たメタ ンスルホン酸 2-プロピルペンチル (63 mg, 0.301 mmol)を 30分毎に 7回加えて 80°Cで 5時間攪拌した。 この間炭酸カリウム(120 mg, 0.868讓 ol) を加えた。 反 応液に 1N塩酸 (3.2 ml)および水(10 ml)を加え、 室温で 30分間攪拌した。 析出 物を濾取し、 THF-酢酸ェチル -メタノールから結晶化して標題化合物 (182 mg, 0.369 '腿 ol)を濃赤色粉末として得た。 A suspension of the compound obtained in Example 3-4 (287 mg, 0.752 mmol) and potassium carbonate (156 mg, 1.13 mmol) in DMF (4.5 ml) was obtained in Reference Example 89 with stirring at 80 ° C. 2-Propylpentyl methanesulfonate (63 mg, 0.301 mmol) was added 7 times every 30 minutes, and the mixture was stirred at 80 ° C for 5 hours. During this time, potassium carbonate (120 mg, 0.868 alcohol) was added. 1N Hydrochloric acid (3.2 ml) and water (10 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration and crystallized from THF-ethyl acetate-methanol to give the title compound (182 mg, 0.369 '-mol) as a dark red powder.
Ή-NMR (200MHz, CDC13) : δ 0.89 (6Η, brt, J= 7.0 Hz), 1.33 (8H, i), 1.69 (1H, m), 1.78 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.41 (3H, s), 2.91 (2H, d, J= 6.6 Hz), 7.27 (2H, d, J= 7.6 Hz), 7.86 (2H, dt, J= 8.6, 2.0 Hz), 9.60 (1H, brs). 元素分析値: C23H31N303S3として計算値: C, 55.95; H, 6.33; N, 8.51; S, 19.48 Ή-NMR (200MHz, CDC1 3 ): δ 0.89 (6Η, brt, J = 7.0 Hz), 1.33 (8H, i), 1.69 (1H, m), 1.78 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 0.8 Hz), 2.41 (3H, s), 2.91 (2H, d, J = 6.6 Hz), 7.27 (2H, d, J = 7.6 Hz), 7.86 (2H, dt, J = 8.6, 2.0 Hz), 9.60 (1H, brs). Elemental analysis: Calculated as C 23 H 31 N 3 0 3 S 3: C, 55.95; H, 6.33; N, 8.51; S, 19.48
(%) 、 実測値: C, 55.97; H, 6.25; N, 8.55; S, 19.58 (%) . (%), Found: C, 55.97; H, 6.25; N, 8.55; S, 19.58 (%).
融点. 134.5-144.0°C 134.5-144.0 ° C
実施例 4-135 Example 4-135
N- {8- [ (1-ィソブチル -3-メチルブチル)スルファニル] -5, 6-ジメチル -3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1 , 4]チアジン-トイリデン } -4-メチルベンゼ ンスルホンアミド ' N- {8-[(1-Isobutyl-3-methylbutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -Tolylidene} -4-methylbenzenesulfonamide ''
実施例 3-4で得た化合物 (298 mg, 0.781 mmol)および炭酸カリウム(162 mg, 1.17 ramol)の DMF (5.0 ml)の懸濁液に 80°Cで攪拌しながら参考例 90で得たヨウ 化 2, 6-ジメチルヘプチル(120 mg, 0.312 mmol)を 30分毎に 7回加えて 80 で 5.5 時間攪拌した。 この間炭酸カリウム(120 mg, 0.868 mmol) を加えた。 反応液に 1N塩酸(3.3 ml)および 0.05N塩酸(20 ml)を加えて酢酸ェチルで抽出した。 有機 層を ^炭酸ナトリゥム水溶液、 2%炭酸水素ナトリゥム水溶液、 水および飽和食塩 水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣をシリカゲル(10 g)のク 口マトグラフィー (酢酸ェチルーへキサン、 2:8) で精製し、 ジェチルエーテル - ジイソプロピルエーテルから結晶化して標題化合物 (57 mg, 0.112删 ol)を濃赤 紫色粉末として得た。 A suspension of the compound obtained in Example 3-4 (298 mg, 0.781 mmol) and potassium carbonate (162 mg, 1.17 ramol) in DMF (5.0 ml) was obtained in Reference Example 90 while stirring at 80 ° C at 80 ° C. 2,6-Dimethylheptyl iodide (120 mg, 0.312 mmol) was added 7 times every 30 minutes, and the mixture was stirred at 80 for 5.5 hours. During this time, potassium carbonate (120 mg, 0.868 mmol) was added. 1N hydrochloric acid (3.3 ml) and 0.05N hydrochloric acid (20 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate. The organic layer was washed with aqueous sodium carbonate solution, 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was purified by silica gel (10 g) chromatography (ethyl acetate-hexane, 2: 8) and crystallized from getyl ether-diisopropyl ether to give the title compound (57 mg, 0.112 mol) in dark red purple Obtained as a powder.
Ή-NMR (200MHz, CDC13) : δ 0.91 (6Η, d, J= 6.6 Hz), 0.92 (6H, d, J= 6.6 Hz), 1.38 (1H, t, J= 6.4 Hz), 1.50 (3H, t, J= 7.0 Hz), 1.74 (2H, m), 1.80 (3H, brs), 2.27 (3H, brs), 2.42 (3H, s), 3.46 (1H, t, J= 7.3 Hz), 7.27 (2H, d, J= 8.2 Hz), 7.86 (2H, d, J= 8.2 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.91 (6Η, d, J = 6.6 Hz), 0.92 (6H, d, J = 6.6 Hz), 1.38 (1H, t, J = 6.4 Hz), 1.50 (3H , t, J = 7.0 Hz), 1.74 (2H, m), 1.80 (3H, brs), 2.27 (3H, brs), 2.42 (3H, s), 3.46 (1H, t, J = 7.3 Hz), 7.27 (2H, d, J = 8.2 Hz), 7.86 (2H, d, J = 8.2 Hz), 9.63 (1H, brs).
元素分析値: C24H33N303S3として計算値: C, 56.77; H, 6.55; , 8.28; S, 18.95Elemental analysis: Calculated as C 24 H 33 N 3 0 3 S 3: C, 56.77; H, 6.55;, 8.28; S, 18.95
(%) 実測値: C, 56.69; H, 6.70; N, 8.20; S, 18.92 ( ) . (%) Found: C, 56.69; H, 6.70; N, 8.20; S, 18.92 ().
実施例 4-136 Example 4-136
N-{5, 6-ジメチル- 3-ォキソ -8- [(2-テトラヒド口- 3-フラエルェチル)スルファニ ル]- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデン }- 4-メチルベ ンゼンスルホンアミド N- {5,6-dimethyl-3-oxo-8-[(2-tetrahydrido-3-furerethyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene} -4-Methylbenzenesulfonamide
実施例 3-4で得た化合物 (299 mg, 0.784 mmol)および炭酸カリウム mg, 1.18匪 ol)の DMF (5 ml)の懸濁液に 80°Cで攪拌しながら参考例 92で得たメタン スルホン酸 2- (テトラヒドロフラン- 3-ィル)ェチル (183 mg, 0.940 mmol)を加え、 反応液を 80°Cで Ί時間攪拌した。この間反応液にヨウ化ナトリウム(118 mg, 0.786 mmol)およびメタンスルホン酸 2- (テトラヒドロフラン- 3-ィル)ェチル(30 mg, 0.154 mmol)を 1時間毎に 5回加えた。 反応液に 1N塩酸 (1.57 ml), 水(5 ml)お よびメタノール(1 ml)を加えて混和した。 析出物を母液と分離し、 THFおよびァ セトニトリルに溶解して濃縮後、 メタノールで希釈した。 析出物を濾取し、 メタ ノールで洗浄、 乾燥して標題化合物 (152 mg, 0.317 mmol)を赤褐色粉末として得 た。 The methane obtained in Reference Example 92 was stirred in a suspension of the compound obtained in Example 3-4 (299 mg, 0.784 mmol) and potassium carbonate mg, 1.18 ol) in DMF (5 ml) at 80 ° C. 2- (Tetrahydrofuran-3-yl) ethyl sulphonate (183 mg, 0.940 mmol) was added, and the reaction solution was stirred at 80 ° C for 2 hours. During this time, sodium iodide (118 mg, 0.786 mmol) and 2- (tetrahydrofuran-3-yl) ethyl (30 mg, 0.154 mmol) were added to the reaction solution five times every hour. To the reaction solution, 1N hydrochloric acid (1.57 ml), water (5 ml) and methanol (1 ml) were added and mixed. The precipitate was separated from the mother liquor, dissolved in THF and acetonitrile, concentrated, and diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (152 mg, 0.317 mmol) as a red-brown powder.
lH-NMR (200MHz, CDC13) : δ 1.53 (IH, dq, J= 11.6, 7.2 Hz), 1.76 (1H, m), 1.79 (3H, q, J= 1.0 Hz), 2.07 (IH, m), 2.26 (3H, q, J= 1.2 Hz), 2.27 (IH, m), 2.42 lH-NMR (200MHz, CDC1 3 ): δ 1.53 (IH, dq, J = 11.6, 7.2 Hz), 1.76 (1H, m), 1.79 (3H, q, J = 1.0 Hz), 2.07 (IH, m) , 2.26 (3H, q, J = 1.2 Hz), 2.27 (IH, m), 2.42
(3H, s), 2.94 (2H, dt, J= 1.8, 7.6 Hz), 3.38 (IH, dd, J= 8.4, 6.6 Hz), 3.74(3H, s), 2.94 (2H, dt, J = 1.8, 7.6 Hz), 3.38 (IH, dd, J = 8.4, 6.6 Hz), 3.74
(IH, dd, J= 8.6, 7.2 Hz), 3.85 (IH, dt, J= 2.2, 7.0 Hz), 3.89 (IH, dd, J= 8.4,(IH, dd, J = 8.6, 7.2 Hz), 3.85 (IH, dt, J = 2.2, 7.0 Hz), 3.89 (IH, dd, J = 8.4,
7.0 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.65 (IH, brs). 元素分析値: C22H27N304S3として計算値: C, 52.59; H, 5.25; N, 8.76; S, 20.06 (%) 、 実測値: C, 52.50; H, 5.23; N, 8.73; S, 19.93 (%) . 7.0 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.65 (IH, brs) Elemental analysis:. As C 22 H 27 N 3 0 4 S 3 Calculated: C, 52.59; H, 5.25; N, 8.76; S, 20.06 (%), Found: C, 52.50; H, 5.23; N, 8.73; S, 19.93 (%).
融点. 139.0-140.0°C 139.0-140.0 ° C
実施例 4-137 Example 4-137
N- (5, 6 -ジメチル- 3 -ォキソ -8 - { [2- (トリフルォ口メチル)ベンジル]スルファニ ル卜 2, 3-ジヒドロ- 1H-イミダゾ [5,l-c] [1,4]チアジン -1-ィリデン)-4-メチルベ ンゼンスルホンアミド N- (5,6-Dimethyl-3-oxo-8-{[2- (trifluoromethyl) benzyl] sulfanyl 2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine- 1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4 で得た化合物 (275 mg, 0.721 讓 ol)および炭酸カリウム(149 mg, 1.08 iol)の DMF (4.5 ml)の懸濁液に室温で攪拌しながら臭化 2-トリフルォロ メチルベンジル(193 mg, 0.793 mmol)を加え、 反応液を室温で 2 時間攪拌した。 反応液に 1N塩酸 (1.44 ml)および水 (5 ml)を加え、 室温で 30分間攪拌した。 析 出物を濾取し、 メタノール一水(1:1) およびメタノールで洗浄、 乾燥して標題化 合物 (369 mg, 0.663 mmol)を赤紫色粉末として得た。 2-Trifluoromethylbenzyl bromide was added to a suspension of the compound obtained in Example 3-4 (275 mg, 0.721 alcohol) and potassium carbonate (149 mg, 1.08 iol) in DMF (4.5 ml) with stirring at room temperature. (193 mg, 0.793 mmol) was added, and the reaction solution was stirred at room temperature for 2 hours. 1N Hydrochloric acid (1.44 ml) and water (5 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (369 mg, 0.663 mmol) as a red-purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.77 (3Η, q, J= 0.6 Hz), 2.26 (3H, q, J= 0.6 Hz), 2.41 (3H, s), 4.36 (2H, s), 7.24 (2H, brt, J= 8.4 Hz), 7.44 (IH, brt, J= 7.1 Hz), 7.57 (2H, brt, J= 7.1 Hz), 7.69 (IH, brd, J= 7.8 Hz), 7.80 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.77 (3Η, q, J = 0.6 Hz), 2.26 (3H, q, J = 0.6 Hz), 2.41 (3H, s), 4.36 (2H, s), 7.24 (2H, brt, J = 8.4 Hz), 7.44 (IH, brt, J = 7.1 Hz), 7.57 (2H, brt, J = 7.1 Hz), 7.69 (IH, brd, J = 7.8 Hz), 7.80 (2H , d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C23H2。N303S3F3として計算値: C, 51.19; H, 3.74; N, 7.79; S, 17.83; F, 10.56 .(%) 、 実測値: C, 51.02; H, 3.45; N, 7.86; S, 17.93; F, 10.48 (%) . 融点. 206.5-207.5°C Elemental analysis: C 23 H 2. Calculated for N 3 0 3 S 3 F 3 : C, 51.19; H, 3.74; N, 7.79; S, 17.83; F, 10.56. (%), Found: C, 51.02; H, 3.45; N, 7.86 ; S, 17.93; F, 10.48 (%). Melting point. 206.5-207.5 ° C
実施例 4-138 Example 4-138
N - [8 -(シクロへキシルスルファ二ル)- 5,6-ジメチル -3-ォキソ _2, 3 -ジヒドロ- 1H- イミダゾ [5,l-c] [1, 4]チアジン-卜イリデン] -4-メチルベンゼンスルホンアミド 実施例 3-4で得た化合物 (277 mg, 0.726匪 ol)および炭酸カリウム(151 mg, 1.09 iranol)の DMF (4.5 ml)の懸濁液に 80°Cで攪拌しながらョードシクロへキサ ン(0.105 ml, 0.799匪 ol)を加え、 反応液を 80°Cで 8時間攪拌した。 この間反応 液にョ一ドシクロへキサン(0.105 ml, 0.799 rafflol)を 2時間毎に 3回、 および炭 酸カリウム(50 mg, 0.363 mmol)を加えた。 反応液に 1N塩酸 (1.45 ml), メタノ —ルー水(2:1, 10 ml)を加えて 17時間混和した。 析出物を母液と分離し、 THFお よびメタノ一ルに溶解して濃縮後、 メタノールで希釈した。 析出物を濾取し、 メ タノ一ルで洗浄、 乾燥して標題化合物 (180 mg, 0.388 mmol)を灰赤色粉末として 得た。 N- [8- (Cyclohexylsulfanyl) -5,6-dimethyl-3-oxo_2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-triylidene] -4-methyl Benzenesulfonamide A suspension of the compound obtained in Example 3-4 (277 mg, 0.726 bandol) and potassium carbonate (151 mg, 1.09 iranol) in DMF (4.5 ml) was stirred at 80 ° C while stirring at 80 ° C. Xanthane (0.105 ml, 0.799 ol) was added, and the reaction solution was stirred at 80 ° C for 8 hours. During this time, cyclohexane (0.105 ml, 0.799 rafflol) was added to the reaction solution three times every two hours, and potassium carbonate (50 mg, 0.363 mmol) was added. To the reaction solution were added 1N hydrochloric acid (1.45 ml) and methanol-luu water (2: 1, 10 ml) and mixed for 17 hours. The precipitate was separated from the mother liquor, dissolved in THF and methanol, concentrated, and diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (180 mg, 0.388 mmol) as a gray-red powder.
Ή—NMR (200MHz, CDC13) : δ 1.30 (1Η, m), 1.41 (2H, brq, J= 9.7 Hz), 1.60 (1H, m), 1.79 (3H, q, J= 0.8 Hz), 1.79 (2H, m), 2.02 (2H, m), 2.26 (3H, q, J= 1.0 Hz), 2.42 (3H, s), 3.36 (1H, m), 7.28 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.30 (1Η, m), 1.41 (2H, brq, J = 9.7 Hz), 1.60 (1H, m), 1.79 (3H, q, J = 0.8 Hz), 1.79 (2H, m), 2.02 (2H, m), 2.26 (3H, q, J = 1.0 Hz), 2.42 (3H, s), 3.36 (1H, m), 7.28 (2H, d, J = 8.0 Hz) , 7.86 (2H, d, J = 8.0 Hz), 9.63 (1H, brs).
元素分析値: C21H25N303S3として計算値: C, 54.40; H, 5.43; N, 9.06; S, 20.75Elemental analysis: C 21 H 25 N 3 0 3 S 3 Calculated: C, 54.40; H, 5.43 ; N, 9.06; S, 20.75
(%) 、 実測値: C, 54.19; H, 5.38; N, 8.90; S, 20.37 (%) . (%), Found: C, 54.19; H, 5.38; N, 8.90; S, 20.37 (%).
融点. 190.0-191.5°C Melting point.190.0-191.5 ° C
実施例 4-139 Example 4-139
N-(5, 6 -ジメチル- 3-ォキソ -8- {[(2S)-2-フエニルプロピル]スルファニル卜 2, 3- ジヒド口- 1H-ィミダゾ [5, 1-c] [1 , 4]チアジン- 1 ィリデン) -4-メチルべンゼンス ルホンアミド N- (5,6-dimethyl-3-oxo-8-{[((2S) -2-phenylpropyl] sulfanyl chloride 2,3-dihydrido-1H-imidazo [5,1-c] [1,4] ] Thiazine-1 ylidene) -4-methylbenzenesulfonamide
実施例 3- 4で得た化合物 (1.57g, 4.11讓 ol)および炭酸カリウム(852 mg, 6.17 mmol)の DMF (20 ml)の懸濁液に 80°Cで攪拌しながら参考例 93で得たメタンスル ホン酸(S)- 2-フエニルプロピル (0.90 g, 4.20 mmol)の DMF (5 ml)溶液を 1時間 カ^て滴下し、 反応液を 80°Cで 8時間攪拌した。 この間反応液にメタンスルホン 酸(S)- 2-フエニルプロピル (0.18g, 0.82 mmol.)を加えた。反応液に IN塩酸 (8.2 ml)、メタノール一水(1:2, 15 ml)を加えて 1時間攪拌混和した。析出物を濾取し、 メタノール一水(1:1)およびメタノールで洗浄、 乾燥して標題化合物 .57 g, 3.17 mmol)を紫色粉末として得た。 A suspension of the compound obtained in Example 3-4 (1.57 g , 4.11 alcohol) and potassium carbonate (852 mg, 6.17 mmol) in DMF (20 ml) was obtained in Reference Example 93 while stirring at 80 ° C at 80 ° C. Methanesul A solution of (S) -2-phenylpropyl phonate (0.90 g, 4.20 mmol) in DMF (5 ml) was added dropwise over 1 hour, and the reaction solution was stirred at 80 ° C for 8 hours. During this time, (S) -2-phenylpropyl methanesulfonate (0.18 g, 0.82 mmol.) Was added to the reaction solution. IN hydrochloric acid (8.2 ml) and methanol / water (1: 2, 15 ml) were added to the reaction solution, and the mixture was stirred and mixed for 1 hour. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (0.57 g, 3.17 mmol) as a purple powder.
'H-NMR (200MHz, CDC13) : δ 1.38 (3Η, d, J= 6.6 Hz), 1.78 (3H, q, J= 1.0 Hz), 2.25 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 3.03 (1H, m), 3.09 (1H, m), 3.22 (1H, quint, J= 5.7 Hz), 7.17-7.37 (7H, m), 7.79 (2H, d, J= 8.0 Hz), 9.60 (1H, brs). 元素分析値: C24H25N303S3として計算値: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%) 、 実測値: C, 57.36; H, 5.13; N, 8.33; S, 19.27 (%) . 'H-NMR (200MHz, CDC1 3): δ 1.38 (3Η, d, J = 6.6 Hz), 1.78 (3H, q, J = 1.0 Hz), 2.25 (3H, q, J = 1.2 Hz), 2.42 ( 3H, s), 3.03 (1H, m), 3.09 (1H, m), 3.22 (1H, quint, J = 5.7 Hz), 7.17-7.37 (7H, m), 7.79 (2H, d, J = 8.0 Hz .), 9.60 (1H, brs ) elemental analysis: calculated C 24 H 25 N 3 0 3 S 3: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%), Found: C , 57.36; H, 5.13; N, 8.33; S, 19.27 (%).
融点. 161.5-162. Ot: Melting point. 161.5-162. Ot:
実施例 4-140 Example 4-140
N - (5, 6-ジメチル- 3-ォキソ -8- { [ (2R) -2-フェニルプ口ピル]スルファ二ル} -2, 3- ジヒドロ- 1H -ィミダゾ [5, 1- c] [1 , 4]チアジン-卜ィリデン) -4-メチルベンゼンス ルホンアミド N- (5,6-Dimethyl-3-oxo-8-{[(2R) -2-phenylpyropenpyr] sulfanyl} -2,3-Dihydro-1H-imidazo [5,1-c] [1 , 4] thiazine-triylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物 (1.58g, 4.13匪 ol)および炭酸カリウム(857 mg, 6.20 匪 ol)の DMF (20 ml)の懸濁液に 80でで攪拌しながら参考例 94で得たメタンスル ホン酸 00- 2_フエニルプロピル (1.02 g, 4.28讓 ol)の DMF (5 ml)溶液を 1時間 かけて滴下し、 反応液を 80°Cで 6時間攪拌した。 この間反応液にメタンスルホン 酸 00- 2-フエニルプロピル (0.14g, 0.60 mmol)を加えた。反応液に IN塩酸 (8.3 ml)、メタノ一ルー水(1: 2, 15 ml)を加えて 1時間攪拌混和した。析出物を濾取し、 メタノール一水(1:1)およびメタノールで洗浄、 乾燥して標題化合物 (1.56 g, 3.12 膽 ol)を紫色粉末として得た。 A suspension of the compound obtained in Example 3-4 (1.58 g, 4.13 marl ol) and potassium carbonate (857 mg, 6.20 marl ol) in DMF (20 ml) was obtained in Reference Example 94 while stirring at 80 at 80 ° C. A solution of 00-2-phenylphenyl methanesulfonate (1.02 g, 4.28 benzyl) in DMF (5 ml) was added dropwise over 1 hour, and the reaction solution was stirred at 80 ° C for 6 hours. During this time, 00-2-phenylpropyl methanesulfonate (0.14 g, 0.60 mmol) was added to the reaction solution. To the reaction solution were added IN hydrochloric acid (8.3 ml) and methanolic water (1: 2, 15 ml), and the mixture was stirred and mixed for 1 hour. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (1.56 g, 3.12 benzyl) as a purple powder.
Ή-N R (200MHz, CDC13) : δ 1.38 (3Η, d, J= 6.2 Hz), 1.78 (3H, brs), 2.25 (3H, q, J= 0.8 Hz), 2.42 (3H, s), 3.03 (1H, m), 3.09 (1H, m), 3.22 (1H, quini, J= 5.6 Hz) , 7.17-7.36 (7H, m), 7.79 (2H, d, J= 8.0 Hz), 9.60 (1H, brs). Ή-NR (200MHz, CDC1 3 ): δ 1.38 (3Η, d, J = 6.2 Hz), 1.78 (3H, brs), 2.25 (3H, q, J = 0.8 Hz), 2.42 (3H, s), 3.03 (1H, m), 3.09 (1H, m), 3.22 (1H, quini, J = 5.6 Hz), 7.17-7.36 (7H, m), 7.79 (2H, d, J = 8.0 Hz), 9.60 (1H, brs).
元素分析値: C24¾5Ns03S3として計算値: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%) 、 実測値: C, 57.55; H, 5.12; N, 8.38; S, 19.08 (%) · 融点. 162.0-163. Ot: Elemental analysis: C 24 ¾ 5 N s 0 3 S 3 Calculated: C, 57.69; H, 5.04 ; N, 8.41; S, 19.25 (%), Found: C, 57.55; H, 5.12 ; N, 8.38; S, 19.08 (%) Melting point. 162.0-163. Ot:
実施例 4-141 Example 4-141
N- (8- {[2- (4 -フルオロフェニル) プロピル]スルファ二ル} -5, 6-ジメチル -3-ォキ ソ- 2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1 , ]チアジン-トイリデン) -4-メチルべン ゼンスルホンアミド N- (8-{[2- (4-fluorophenyl) propyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,] thiazine-toylidene) -4-methylbenzene benzenesulfonamide
実施例 3-4 で得た化合物 (295 mg, 0.773 mmol)および炭酸カリウム(160 mg, 1.16腿 ol)の DMF (5.0 ml)の懸濁液に 80DCで攪拌しながら参考例 95で得たメタ ンスルホン酸 2- (4-フルオロフェニル)プロピル (181 mg, 0.773腿 ol)を加え、 反応液を 80°Cで 7時間攪拌した。 この間反応液にメタンスルホン酸 2- (4-フルォ 口フエニル)プロピル(36 mg, 0.15mmol)を加えた。反応液に 1N塩酸 (1.55 ml), メタノ一ルー水(1: 1, 4 ml)を加えて 1時間攪拌混和した。析出物を母液と分離し、 THF およびメタノールに溶解して濃縮後、 メタノールで希釈した。 析出物を濾取 し、 メタノールで洗浄、 乾燥して標題化合物 (235 mg, 0.455 mmol)を紫色粉末と して得た。 Of the compound obtained in Example 3-4 (295 mg, 0.773 mmol) and potassium carbonate (160 mg, 1.16 thigh ol) obtained in the stirring Reference Example 95 in suspension in 80 D C of DMF (5.0 ml) of 2- (4-Fluorophenyl) propyl methanesulfonate (181 mg, 0.773 tmol) was added, and the reaction solution was stirred at 80 ° C for 7 hours. During this period, 2- (4-fluorophenyl) propyl methanesulfonate (36 mg, 0.15 mmol) was added to the reaction solution. To the reaction solution were added 1N hydrochloric acid (1.55 ml) and methanolic water (1: 1, 4 ml), and the mixture was stirred and mixed for 1 hour. The precipitate was separated from the mother liquor, dissolved in THF and methanol, concentrated, and then diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (235 mg, 0.455 mmol) as a purple powder.
Ή-N R (200MHz, CDC13) : δ 1.36 (3H, d, J= 6.6 Hz), 1.78 (3H, brs), 2.25 (3H, brs), 2.43 (3H, s), 3.03 (1H, m), 3.12 (2H, m), 6.99 (2H, t, J= 8.6 Hz), 7.15 (2H, dd, J= 8.8, 5.6 Hz), 7.25 (2H, d, J= 8.4 Hz), 7.78 (2H, d, J= 8.4 Hz), 9.61 (1H, brs). Ή-NR (200MHz, CDC1 3 ): δ 1.36 (3H, d, J = 6.6 Hz), 1.78 (3H, brs), 2.25 (3H, brs), 2.43 (3H, s), 3.03 (1H, m) , 3.12 (2H, m), 6.99 (2H, t, J = 8.6 Hz), 7.15 (2H, dd, J = 8.8, 5.6 Hz), 7.25 (2H, d, J = 8.4 Hz), 7.78 (2H, d, J = 8.4 Hz), 9.61 (1H, brs).
元素分析値: C24H24N303S3Fとして計算値: C, 55.68; H, 4.67; N, 8.12; S, 18.58; F, 3.67 (%) 、 実測値: C, 55.33; H, 4.67; N, 7.98; S, 18.56; F, 3.49 (%) . 融点. 183.5- 185.5 Elemental analysis: C 24 H 24 N 3 0 3 S 3 F Calculated: C, 55.68; H, 4.67 ; N, 8.12; S, 18.58; F, 3.67 (%), Found: C, 55.33; H , 4.67; N, 7.98; S, 18.56; F, 3.49 (%).
実施例 4-142 Example 4-142
N - {5, 6 -ジメチル -3-ォキソ -8- [(2, 3, 4, 5, 6 -ペンタメチルベンジル)スルファニ ル]- 2,3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン }- 4-メチルベ ンゼンスルホンアミド , N- {5,6-dimethyl-3-oxo-8-[(2,3,4,5,6-pentamethylbenzyl) sulfanyl]-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene} -4-methylbenzenesulfonamide,
実施例 3-4 で得た化合物 (268 mg, 0.702 mmol)および炭酸カリウム(146 mg, 1.05 mmol)の DMF (4.5 ml)の懸濁液に 80°Cで攪拌しながら塩化 2, 3, 4, 5, 6-ペン 夕メチルベンジルおよびョゥ化ナトリゥムから調製したヨウ化 2, 3, 4, 5, 6 -ペン タメチルベンジル(176 mg, 0.773讓 ol)を加え、 反応液を 80°Cで 2.5時間攪拌し た。 この間反応液に同ヨウ化物 (18 mg, 0.08匪 ol)を加えた。 反応液に 1N塩酸 (1.4 ml), メタノール一水(1:1, 4 mDを加えて 30分間攪拌混和した。 析出物を 濾取し、メタノール一水( 1: 1 )およびメタノールで洗浄、乾燥して標題化合物 (360 ig, 0.665腦 ol)を赤紫色粉末として得た。 To a suspension of the compound obtained in Example 3-4 (268 mg, 0.702 mmol) and potassium carbonate (146 mg, 1.05 mmol) in DMF (4.5 ml) was added 2,3,4 chloride with stirring at 80 ° C. 2,5,6-Pentamethylbenzyl and 2,3,4,5,6-pentamethylbenzyl iodide prepared from sodium iodide (176 mg, 0.773 mL) were added, and the reaction mixture was heated to 80 ° C. And stir for 2.5 hours Was. During this time, the same iodide (18 mg, 0.08 ol) was added to the reaction mixture. 1N Hydrochloric acid (1.4 ml) and methanol / water (1: 1, 4 mD) were added to the reaction solution, and the mixture was stirred for 30 minutes, and the precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried. Thus, the title compound (360 ig, 0.665 mol) was obtained as a red-purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.82 (3Η, q, J= 1.0 Hz), 2.24 (9H, s), 2.26 (3H, brs), 2.28 (6H, s), 2.40 (3H, s), 4.29 (2H, s), 7.19 (2H, d, J= 8.0 Hz), 7.76 (2H, d, J= 8.0 Hz), 9.65 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.82 (3Η, q, J = 1.0 Hz), 2.24 (9H, s), 2.26 (3H, brs), 2.28 (6H, s), 2.40 (3H, s) , 4.29 (2H, s), 7.19 (2H, d, J = 8.0 Hz), 7.76 (2H, d, J = 8.0 Hz), 9.65 (1H, brs).
元素分析値: C27H31N303S3 · 0· 5H20として計算値: C, 58.88; H, 5.86; N, 7.63; S, 17.47 (%) 、 実測値: C, 58.93; H, 5.69; N, 7.65; S, 17.32 ( ) . Elemental analysis: C 27 H 31 N 3 0 3 S 3 .5 · 5H 2 0 Calculated: C, 58.88; H, 5.86; N, 7.63; S, 17.47 (%), found: C, 58.93; H, 5.69; N, 7.65; S, 17.32 ().
融点. 213.0-215.0°C Melting point.213.0-215.0 ° C
実施例 4-143 Example 4-143
酢酸ェチル -ジェチルェ一テルから再結晶し、 濃赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals.
融点: 175-177°C. Melting point: 175-177 ° C.
赤外吸収スぺクトル(IR) (KBr中) V 3272, 2946, 2907, 2868, 1748, 1715, 1659, 1613, 1557 cm—1. Infrared absorption spectrum (IR) (in KBr) V 3272, 2946, 2907, 2868, 1748, 1715, 1659, 1613, 1557 cm- 1 .
Ή-NMR (CDC") : δ 0.936 (9Η, s), 1.50-1.62 (2H, m), 1.79 (3H, d, J=1.2 Hz), 2.27 (3H, d, J=0.6 Hz), 2.42 (3H, s), 2.85-2.96 (2H, m), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). Ή-NMR (CDC "): δ 0.936 (9Η, s), 1.50-1.62 (2H, m), 1.79 (3H, d, J = 1.2 Hz), 2.27 (3H, d, J = 0.6 Hz), 2.42 (3H, s), 2.85-2.96 (2H, m), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C21H27N303S3として計算値: C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), 実測値: C, 53.81; H, 5.72; N, 8.94; S, 20.42 (%) . Elemental analysis: Calculated for C 21 H 27 N 3 0 3 S 3 : C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), found: C, 53.81; H, 5.72; N, 8.94; S, 20.42 (%).
実施例 4-144 Example 4-144
N- {8- [(メシチルメチル)スルファニル] -5, 6-ジメチル- 3-ォキソ- 2, 3-ジヒドロ - 1H-イミダゾ [5,1 - c] [1,4]チアジン - 1-ィリデン }- 4-メチルベンゼンスルホンァ ミド N- {8-[(mesitylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene}- 4-methylbenzenesulfonamide
実施例 3- 4で得た化合物 (287 mg, 0.752 mmol)および炭酸カリウム(156 mg, 1.13 mmol)の DMF (4.5 ml)の懸濁液に 6(TCで攪拌しながら塩化 2, 4, 6-トリメチ ルペンジル(147 mg, 0.828 mmol)を加え、 反応液を 60°Cで 13時間攪拌した。 反 応液に 1N塩酸 (1.5 ml), メタノール一水(3:2, 5 ml)を加えて 60分間攪拌混和 した。析出物を濾取し、 メタノール一水(1:1)およびメタノールで洗浄、乾燥して 標題化合物 (381 mg, 0.741 mmol)を赤紫色粉末として得た。 To a suspension of the compound (287 mg, 0.752 mmol) obtained in Example 3-4 and potassium carbonate (156 mg, 1.13 mmol) in DMF (4.5 ml) was added 6 (2, 4, 6 -Trimethylpentyl (147 mg, 0.828 mmol) was added, and the reaction solution was stirred for 13 hours at 60 ° C. To the reaction solution was added 1N hydrochloric acid (1.5 ml), methanol / water (3: 2, 5 ml). The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, dried, and dried. The title compound (381 mg, 0.741 mmol) was obtained as a purplish red powder.
Ή-NMR (200MHz, CDC13) : δ 1.82 (3Η, q, J= 1.0 Hz), 2.28 (3H, q, J= 1.2 Hz), 2.29 (3H, s), 2.33 (6H, s), 2.40 (3H, s), 4.22 (2H, s), 6.88 (2H, brs), 7.20 (2H, d, J= 7.6 Hz), 7.75 (2H, d, J= 8.0 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.82 (3Η, q, J = 1.0 Hz), 2.28 (3H, q, J = 1.2 Hz), 2.29 (3H, s), 2.33 (6H, s), 2.40 (3H, s), 4.22 (2H, s), 6.88 (2H, brs), 7.20 (2H, d, J = 7.6 Hz), 7.75 (2H, d, J = 8.0 Hz), 9.62 (1H, brs) .
融点. 200.5- 202.5°C Melting point. 200.5-202.5 ° C
実施例 4-145 Example 4-145
N-(5, 6-ジメチル- 3-ォキソ -8- {[2- (2, 3, 4, 5, 6-ペンタメチルフエニル)ェチル]ス ルファ二ル} - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン)-4- 実施例 3-4 で得た化合物 (280 mg, 0.734 imnol)および炭酸カリウム(152 rag, 1.10丽 ol)の DMF (4.5 ml)の懸濁液に 6(ΤΌで攪拌しながら参考例 96で得たメタ ンスルホン酸 2- (2, 3, 4, 5, 6-ペン夕メチルフエニル)ェチル(238 mg, 0.881 mmol) を加え、 反応液を 60°Cで 20時間攪拌した。 反応液に IN塩酸 (1.47ml)および水 (20 ml)を加えて酢酸ェチルで抽出した。有機層を 2%炭酸水素ナトリゥム水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮後、 残渣にメタノ —ルを加えた。析出物を濾取し、メタノールで洗浄、乾燥して標題化合物 (52 mg, 0.094匪 ol)を赤紫色粉末として得た。 N- (5,6-dimethyl-3-oxo-8-{[2- (2,3,4,5,6-pentamethylphenyl) ethyl] sulfanyl} -2,3-dihydro-1H -Imidazo [5,1-c] [1,4] thiazine-1-ylidene) -4- The compound obtained in Example 3-4 (280 mg, 0.734 imnol) and potassium carbonate (152 rag, 1.10 mol) To a suspension of DMF (4.5 ml) in 2- (2,3,4,5,6-pentanomethylphenyl) ethyl methanesulfonate (238 mg, 0.881 The reaction mixture was stirred at 60 ° C. for 20 hours, IN hydrochloric acid (1.47 ml) and water (20 ml) were added to the reaction mixture, and the mixture was extracted with ethyl acetate.The organic layer was extracted with a 2% aqueous sodium hydrogen carbonate solution. After washing with water and saturated saline, drying over sodium sulfate, concentrating and adding methanol to the residue, the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (52 mg, 0.094 ol) as a red-purple powder.
Ή-NMR (200MHz, CDCI3) : δ 1.77 (3Η, brs), 2.23 (6H, s), 2.24 (3H, s), 2.26 (9H, s), 2.43 (3H, s), 3.03 (4H, s), 7.30 (2H, d, J= 8.6 Hz), 7.87 (2H, d, J= 8.6 Hz), 9.66 (1H, brs). Ή-NMR (200 MHz, CDCI 3 ): δ 1.77 (3Η, brs), 2.23 (6H, s), 2.24 (3H, s), 2.26 (9H, s), 2.43 (3H, s), 3.03 (4H, s) s), 7.30 (2H, d, J = 8.6 Hz), 7.87 (2H, d, J = 8.6 Hz), 9.66 (1H, brs).
元素分析値: C28H33N303S3として計算値: C, 60.51; H, 5.98; N, 7.56; S, 17.31Elemental analysis: C 28 H 33 N 3 0 3 S 3 Calculated: C, 60.51; H, 5.98 ; N, 7.56; S, 17.31
(%) 、 実測値: C, 60.48; H, 6.01; , 7.34; S, 17.01 ( ) · (%), Found: C, 60.48; H, 6.01;, 7.34; S, 17.01 () ·
融点. 200.7-202.3°C Melting point.200.7-202.3 ° C
実施例 4-146 Example 4-146
N- {8- [(2-メシチルメチル)スルファニル ]-5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ -1H-イミダゾ [5,卜 c] [ 4]チアジン-卜ィリデン }-4-メチルベンゼンスルホンァ ^ド N- {8-[(2-mesitylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [4] thiazine-trilidene} -4- Methylbenzene sulfone
実施例 3-4で得た化合物 (291 mg, 0.763 mmol)および炭酸カリウム(158 mg, 1.14 mmol)の DMF (4.5 ml)の懸濁液に 6CTCで攪拌しながら参考例 97で得たメタ ンスルホン酸 2- (2, 4, 6-トリメチルフエニル)ェチル(232 mg, 0.915顧 ol)を加え、 反応液を 60°Cで 24時間攪拌した。 この間反応液にメタンスルホン酸 2- (2,4, 6 - ト リメチルフエニル)ェチル(97 mg, 0.38 匪 ol)を加えた。 反応液に 1N塩酸 (1.53 ml) 、 メタノール一水(2:5, 7 ml)を加えて混和した。 析出物を母液と分離 し、 THF およびメタノールに溶解して濃縮後、 メタノールで希釈した。 析出物を 濾取し、 メタノールで洗浄、 乾燥して標題化合物 (84 mg, 0.159翻 ol)を赤紫色 粉末として得た。 To a suspension of the compound (291 mg, 0.763 mmol) obtained in Example 3-4 and potassium carbonate (158 mg, 1.14 mmol) in DMF (4.5 ml) was added the meta 2- (2,4,6-Trimethylphenyl) ethyl sulfonic acid (232 mg, 0.915) was added, and the reaction solution was stirred at 60 ° C for 24 hours. During this time, 2- (2,4,6-trimethylphenyl) ethyl methanesulfonate (97 mg, 0.38 ol) was added to the reaction mixture. The reaction mixture was mixed with 1N hydrochloric acid (1.53 ml) and methanol / water (2: 5.7 ml). The precipitate was separated from the mother liquor, dissolved in THF and methanol, concentrated, and then diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (84 mg, 0.159 mmol) as a red-purple powder.
lH-NMR (200MHz, CDC13) : δ 1.77 (3Η, brs), 2.26 (6H, s), 2.30 (6H, s), 2.43 (3H, s), 2.98 (4H, m), 6.86 (2H, brs), 7.30 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.67 (1H, brs). lH-NMR (200MHz, CDC1 3 ): δ 1.77 (3Η, brs), 2.26 (6H, s), 2.30 (6H, s), 2.43 (3H, s), 2.98 (4H, m), 6.86 (2H, brs), 7.30 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.67 (1H, brs).
元素分析値: C26H29N303S3として計算値: C, 59.17; H, 5.54; N, 7.96; S, 18.23Elemental analysis: C 26 H 29 N 3 0 3 S 3 Calculated: C, 59.17; H, 5.54 ; N, 7.96; S, 18.23
(%) 、 実測値: C, 58.90; H, 5.61; , 7.83; S, 17.96 (%) . (%), Found: C, 58.90; H, 5.61;, 7.83; S, 17.96 (%).
融点. 193.0-195.5°C Melting point.193.0-195.5 ° C
実施例 4-147 Example 4-147
N-{5, 6-ジメチル- 3-ォキソ -8- [(テトラヒドロ- 2H-ピラン- 2-ィルメチル)スルフ ァニル ] -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン } -4-メチ 実施例 3- 4で得た化合物 (243 ig, 0.637讓 ol)、ヨウ化ナトリウム(19 mg, 0.127 mmol)および炭酸カリウム(132 mg, 0.955膽 ol)の DMF (4.0 ml)の懸濁液に 60°C で攪拌しながら臭ィヒ 2-テトラヒドロピラニルメチル(232 mg, 0.915 mmol)を加え、 反応液を 60 で 20時間攪拌した。 反応液に 1N塩酸 (1.27ml), 水(5 ml)を加え て混和した。 析出物を母液と分離し、 THF に溶解して濃縮後、 メタノールで希釈 した。 析出物を濾取し、 メタノールで洗浄、 乾燥して標題化合物 (207 mg, 0.432 匪 ol)を赤紫色粉末として得た。 N- {5,6-Dimethyl-3-oxo-8-[(tetrahydro-2H-pyran-2-ylmethyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1, 4] thiazin-1-ylidene} -4-methyl compound obtained in Example 3-4 (243 ig, 0.637 benzene), sodium iodide (19 mg, 0.127 mmol) and potassium carbonate (132 mg, 0.955 benzene) ) Was added to a suspension of DMF (4.0 ml) with stirring at 60 ° C, and the reaction mixture was stirred at 60 for 20 hours. The reaction mixture was mixed with 1N hydrochloric acid (1.27 ml) and water (5 ml). The precipitate was separated from the mother liquor, dissolved in THF, concentrated, and diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (207 mg, 0.432 ol) as a red-purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.30 (2Η, brq, J= 13.0 Hz), 1.53 (2H, m), 1.73 (2H, brd, J= 13.0 Hz), 1.79 (3H, brs), 2.26 (3H, brs), 2.41 (3H, s), 2.99 (1H, dd, J= 13.0, 5.2 Hz), 3.06 (1H, dd, J= 13.2, 6.8 Hz), 3.44 (2H, m), 3.99 (1H, brd, J= 11.4 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). 元素分析値: C21H25N304S3として計算値: C, 52.59; H, 5.25; N, 8.76; S, 20.06 (%) 、 実測値: C, 52.68; H, 5.32; , 8.64; S, 19.68 (%) . 融点. 187.0- 189.0°C Ή-NMR (200MHz, CDC1 3 ): δ 1.30 (2Η, brq, J = 13.0 Hz), 1.53 (2H, m), 1.73 (2H, brd, J = 13.0 Hz), 1.79 (3H, brs), 2.26 (3H, brs), 2.41 (3H, s), 2.99 (1H, dd, J = 13.0, 5.2 Hz), 3.06 (1H, dd, J = 13.2, 6.8 Hz), 3.44 (2H, m), 3.99 ( 1H, brd, J = 11.4 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.0 Hz), 9.63 (1H, brs). Elemental analysis: C 21 H 25 N 3 0 4 S 3 calculated: C, 52.59; H, 5.25 ; N, 8.76; S, 20.06 (%), Found: C, 52.68; H, 5.32;, 8.64; S, 19.68 (%). Melting point. 187.0-189.0 ° C
実施例 4-148 Example 4-148
N- {5, 6-ジメチル- 3-ォキソ -8- [ (2-テトラヒド口- 2H-ピラン- 2-ィルェチル)スル ファニル]- 2,3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン卜 4 -メ 実施例 3-4で得た化合物 (289 mg, 0.758 mmol)および炭酸カリウム(157 mg, 1.14 mmol)の DMF (4.8 ml)の懸濁液に 60°Cで攪拌しながら参考例 98で得たヨウ 化 2- (2 -テトラヒドロピラエル)ェチル(251 mg, 0.909 mmol)を加え、反応液を 60°C で 1.5時間攪拌した。反応液に IN塩酸 (1.5ml) およびメタノール一水(1 :1, 10 ml)を加え、室温で 30分間攪拌した。析出物を濾取し、 メタノール—水(2:1 およ び 1:1)で洗浄、 乾燥して標題化合物 (336 mg, 0.681 讓 ol)を紫色粉末として得 た。 N- {5,6-dimethyl-3-oxo-8-[(2-tetrahydrido-2H-pyran-2-ylethyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [ 1,4] Thiazine-1-yrident 4-me A suspension of the compound obtained in Example 3-4 (289 mg, 0.758 mmol) and potassium carbonate (157 mg, 1.14 mmol) in DMF (4.8 ml). While stirring at 60 ° C, 2- (2-tetrahydropyrael) ethyl iodide (251 mg, 0.909 mmol) obtained in Reference Example 98 was added, and the reaction solution was stirred at 60 ° C for 1.5 hours. IN hydrochloric acid (1.5 ml) and methanol / water (1: 1, 10 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (2: 1 and 1: 1), and dried to give the title compound (336 mg, 0.681 benzyl) as a purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.28 (1Η, brq, J= 10.8 Hz), 1.52 (3H, br), ca. 1.6 (1H, m),. 1.78 (2H, brq, J= 1.2 Hz), 1.79 (3H, m), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 3.02 (1H, ddd, J= 13.0, 8.8, 7.5 Hz), 3.08 (1H, ddd, J= 12.8, 8.4, 6.0 Hz), 3.32 (1H, dm, J= 8 Hz), 3.39 (1H, dm, J= 8 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.28 (1Η, brq, J = 10.8 Hz), 1.52 (3H, br), ca. 1.6 (1H, m) ,. 1.78 (2H, brq, J = 1.2 Hz ), 1.79 (3H, m), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 3.02 (1H, ddd, J = 13.0, 8.8, 7.5 Hz), 3.08 (1H, ddd, J = 12.8, 8.4, 6.0 Hz), 3.32 (1H, dm, J = 8 Hz), 3.39 (1H, dm, J = 8 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C22H21N304S3 - 0.25 Oとして計算値: C, 53.04; H, 5.56; N, 8.43; S, 19.31 (%) 、 実測値: C, 53.05; H, 5.59; N, 8.46; S, 19.24 (%) . Elemental analysis: C 22 H 21 N 3 0 4 S 3 - 0.25 O Calculated: C, 53.04; H, 5.56 ; N, 8.43; S, 19.31 (%), Found: C, 53.05; H, 5.59 ; N, 8.46; S, 19.24 (%).
融点. 169.5-171.0°C Melting point.169.5-171.0 ° C
実施例 4- 149 Example 4-149
2- [ (5, 6-ジメチル-卜 { [ (4-メチルフエニル)スルホニル]イミノ } -3-ォキソ -2, 3- ジヒドロ- 1H-イミダゾ [5, c] [1, 4]チアジン- 8-ィル)スルファニル]ヘプタン酸 ェチル 2-[(5,6-Dimethyl-toluene [[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, c] [1,4] thiazine-8- Yl) sulfanyl] ethyl heptanoate
実施例 3- 4で得た化合物 (349 mg, 0.915 mmol)およびトリェチルァミン(0.380 ml, 2.74 mmol)の THF (7.0 ml)懸濁液に 2-ブロモヘプタン酸ェチル(0.201 ml, 1.01腿 ol)を室温で加えた。 混合物を室温で 1時間攪拌した後、 24時間加熱還流 した。 この間反応液にェチル 2_ブロモヘプタン酸(0.055 ml, 0.275 mmol)を 1 回加えた。 反応液を濃縮し、 酢酸ェチルで抽出した。 有機層を水および飽和食塩 水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣をシリカゲル (10 g)のク 口マトグラフィ一 (酢酸ェチルーへキサン、 1:4-1:3) に付した。酢酸ェチルーへ キサン(1:3)の画分を濃縮し、 ジェチルェ一テルから結晶化して標題化合物 (142 mg, 0.264腿 ol)を紫色粉末として得た。 To a suspension of the compound obtained in Example 3-4 (349 mg, 0.915 mmol) and triethylamine (0.380 ml, 2.74 mmol) in THF (7.0 ml) was added ethyl 2-bromoheptanoate (0.201 ml, 1.01 mol). Added at room temperature. The mixture was stirred at room temperature for 1 hour and then heated to reflux for 24 hours. During this period, 1 ml of ethyl 2-bromoheptanoic acid (0.055 ml, 0.275 mmol) was added to the reaction mixture. Added twice. The reaction solution was concentrated and extracted with ethyl acetate. The organic layer was washed with water and saturated saline, dried over sodium sulfate, and concentrated. The residue was subjected to chromatography on silica gel (10 g) (ethyl acetate-hexane, 1: 4-1: 3). The fraction of ethyl acetate-hexane (1: 3) was concentrated and crystallized from getyl ether to obtain the title compound (142 mg, 0.264 tmol) as a purple powder.
¾-NMR (200MHz, CDC13) : δ 0.89 (3Η, brt, J= 6.2 Hz), 1.28 (3H, t, J= 7.1 Hz), ca. 1.31 (4H, brs), 1.38 (2H, m), 1.78 (2H, brq, J= 1.0 Hz), 1.88 (2H, m), 2.24 (3H, q, J= 0.6 Hz), 2.42 (3H, s), 3.99 (1H, t, J= 7. l Hz), 4.21 (2H, dq, J= 0.8, 7.1 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.86 (2H, dt, J= 8.0, 1.4 Hz), 9.65 (1H, brs). ¾-NMR (200MHz, CDC1 3 ): δ 0.89 (3Η, brt, J = 6.2 Hz), 1.28 (3H, t, J = 7.1 Hz), ca. 1.31 (4H, brs), 1.38 (2H, m) , 1.78 (2H, brq, J = 1.0 Hz), 1.88 (2H, m), 2.24 (3H, q, J = 0.6 Hz), 2.42 (3H, s), 3.99 (1H, t, J = 7. l Hz), 4.21 (2H, dq, J = 0.8, 7.1 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, dt, J = 8.0, 1.4 Hz), 9.65 (1H, brs).
元素分析値: C24H31N305S3として計算値: (:, 53.61; H, 5.81; N, 7.81; S, 17.89Elemental analysis: C 24 H 31 N 3 0 5 S 3 Calculated: (:, 53.61; H, 5.81; N, 7.81; S, 17.89
(%) 、 実測値: C, 53.81; H, 5.81; N, 8.07; S, 17.82 (%) . (%), Found: C, 53.81; H, 5.81; N, 8.07; S, 17.82 (%).
融点. 82.5- 87.5°C Melting point.82.5-87.5 ° C
実施例 4-150 Example 4-150
酢酸ェチル -ジェチルエーテルから再結晶し、 濃赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals.
融点 137- 139°C. 137-139 ° C.
赤外吸収スペクトル(IR) (KBr中) : 3254, 2924, 1748, 1719, 1663, 1609, 1557, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr): 3254, 2924, 1748, 1719, 1663, 1609, 1557, 1497 cm " 1 .
Ή-NMR (CDC") : δ 1.70-1.86 (2Η, m), 1.78 (3H, d, J=0.8 Hz), 2.10-2.24 (2H, m), 2.26 (3H, d, J=0.8 Hz), 2.42 (3H, s), 2.94 (2H, d, J=7.8 Hz), 5.01 (1H, s), 5.04-5.11 (1H, m), 5.64-5.87 (1H, m), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). Ή-NMR (CDC "): δ 1.70-1.86 (2Η, m), 1.78 (3H, d, J = 0.8 Hz), 2.10-2.24 (2H, m), 2.26 (3H, d, J = 0.8 Hz) , 2.42 (3H, s), 2.94 (2H, d, J = 7.8 Hz), 5.01 (1H, s), 5.04-5.11 (1H, m), 5.64-5.87 (1H, m), 7.28 (2H, d , J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C2。H23N303S3として計算値: C, 53.43; H, 5.16; N, 9.35; S, 21.40 (%)、 実測値: C, 53.43; H, 5.16; N, 9.33; S, 21.26 (%) · Elemental analysis: C 2. H 23 N 3 0 3 S 3 Calculated: C, 53.43; H, 5.16; N, 9.35; S, 21.40 (%), Found: C, 53.43; H, 5.16; N, 9.33; S, 21.26 ( %) ·
実施例 4-151 Example 4-151
酢酸ェチル-ジェチルエーテルから再結晶し、 濃赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals.
融点 184- 186 . Melting point 184-186.
赤外吸収スペクトル(IR) (KBr中) V: 3285, 2946, 1753, 1717 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3285, 2946, 1753, 1717 cm " 1 .
Ή-N R (CDC13) : δ 1.79 (3Η, d, J=1.2 Hz), 2.14-2.30 (2H, m), 2.27 (3H, d, J=1.2 Hz), 2.42 (3H, s), 2.95-3.14 (1H, m), 3.08 (2H, t, J=7.8Hz), 7.30 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.4 Hz), 9.62 (1H, bs). Ή-NR (CDC1 3): δ 1.79 (3Η, d, J = 1.2 Hz), 2.14-2.30 (2H, m), 2.27 (3H, d, J = 1.2 Hz), 2.42 (3H, s), 2.95-3.14 (1H, m), 3.08 (2H, t, J = 7.8Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C2。HigN303S3F6として計算値: C, 42.93; H, 3.42; N, 7.51; S, 17.19Elemental analysis: C 2. H ig N 3 0 3 S 3 F 6 Calculated: C, 42.93; H, 3.42 ; N, 7.51; S, 17.19
(%)、 実測値: C, 43.17; H, 3.52; N, 7.49; S, 17.38 (%) . (%), Found: C, 43.17; H, 3.52; N, 7.49; S, 17.38 (%).
実施例 4-152 Example 4-152
酢酸ェチル-ジイソプロピルエーテルから再結晶し、 濃赤色結晶を得た。 Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals.
融点 195-197°C. Mp 195-197 ° C.
赤外吸収スぺクトル(IR) (KBr中) ソ : 3250, 2924, 1732, 1667, 1615, 1557, 1495 cm-1. Infrared absorption spectrum (IR) (in KBr) So: 3250, 2924, 1732, 1667, 1615, 1557, 1495 cm -1 .
Ή-NMR (CDC13) : δ 1.6-1.78 (2Η, m) , 1.82 (3H, d, J=1.2 Hz), 2.12-2.42 (2H, m), 2.87 (3H, d, J=0.8 Hz), 2.35 (3H, s), 2.69-2.98 (4H, m), 3.64-3.80 (1H, 1) , 7.06-7.18 (4H, in), 7.21 (2H, d, J=8.0 Hz), 7.82 (2H, d, J=8.4 Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3): δ 1.6-1.78 (2Η, m), 1.82 (3H, d, J = 1.2 Hz), 2.12-2.42 (2H, m), 2.87 (3H, d, J = 0.8 Hz) , 2.35 (3H, s), 2.69-2.98 (4H, m), 3.64-3.80 (1H, 1), 7.06-7.18 (4H, in), 7.21 (2H, d, J = 8.0 Hz), 7.82 (2H , D, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C26HZ7N303S3 · 0.3H20として計算値: C, 58.79; H, 5.24; N, 7.91; S, 18.11 (%), 実測値: C, 58.71; H, 5.37; N, 7.92; S, 17.81 (%) . Elemental analysis: C 26 H Z7 N 3 0 3 S 3 · 0.3 H 2 0 Calculated: C, 58.79; H, 5.24; N, 7.91; S, 18.11 (%), found: C, 58.71; H , 5.37; N, 7.92; S, 17.81 (%).
実施例 4-153 Example 4-153
N - {5, 6-ジメチル- 8-[(2-メチルプチル)スルファニル ]-3-ォキソ -2, 3-ジヒドロ - 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }-4-メチルベンゼンスルホンァ ミド N- {5,6-dimethyl-8-[(2-methylbutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene } -4-Methylbenzenesulfonamide
実施例 3-4 で得た化合物 (256 mg, 0.671 mmol)および炭酸カリウム(139 mg, 1.01 mmol)の DMF (4.0 ml) の懸濁液に 80°Cで攪拌しながら 1_ョード -2-メチル ブタン (155 mg, 0.738腿 ol)を加え、 混合物を 50分間攪拌した。 反応液に 1N塩 酸 (1.34ml) およびメタノール—水(1:1, 4ml)を加え、室温で 60分間攪拌した。 析出物を濾取し、メタノール一水 (1:1)およびメタノールで洗浄、乾燥して標題 化合物 (275 mg, 0.609匪 ol)を赤色粉末として得た。 To a suspension of the compound obtained in Example 3-4 (256 mg, 0.671 mmol) and potassium carbonate (139 mg, 1.01 mmol) in DMF (4.0 ml) was stirred at 80 ° C at 80 ° C for 1-do-2- Methyl butane (155 mg, 0.738 tmol) was added and the mixture was stirred for 50 minutes. 1N Hydrochloric acid (1.34 ml) and methanol-water (1: 1, 4 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (275 mg, 0.609 bandol) as a red powder.
Ή-NMR (200MHz, CDC13) : δ 0.91 (3Η, t, J= 7.4 Hz), 1.00 (3H, d, J= 6.6 Hz), 1.25 (1H, dt, J= 13.6, 7.4 Hz), 1.48 (1H, sixtet, J= 6.6 Hz), 1.68 (1H, m), 1.78 (3H, s), 2.26 (3¾ s), 2.43 (3H, s), 2.75 (1H, dd, J= 12.6, 7.4 Hz), 2.94 (1H, dd, J= 12.4, 6.2 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.91 (3Η, t, J = 7.4 Hz), 1.00 (3H, d, J = 6.6 Hz), 1.25 (1H, dt, J = 13.6, 7.4 Hz), 1.48 (1H, sixtet, J = 6.6 Hz), 1.68 (1H, m), 1.78 (3H, s), 2.26 (3¾s), 2.43 (3H, s), 2.75 (1H, dd, J = 12.6, 7.4 Hz ), 2.94 (1H, dd, J = 12.4, 6.2 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C2。H25N303S3として計算値: C, 53.19; H, 5.58; N, 9.30; S, 21.30Elemental analysis: C 2. H 25 N 3 0 3 S 3 Calculated: C, 53.19; H, 5.58 ; N, 9.30; S, 21.30
(%), 実測値: C, 53.02; H, 5.60; N, 9.40; S, 21.20 (%) . (%), Found: C, 53.02; H, 5.60; N, 9.40; S, 21.20 (%).
融点 165.0-166.2°C Melting point 165.0-166.2 ° C
実施例 4-154 Example 4-154
N -(8- {[2- (4-ク口口フエニル)プロピル]スルファニル } - 5, 6-ジメチル -3-ォキソ - 2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデン)-4-メチルベンゼ ンスルホンアミド N- (8-{[2- (4-Mouth-phenyl) propyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3- 4で得た化合物 (1.59g, 4.1711111101)ぉょび炭酸カリゥム(8651^, 6.26 膽 ol)の DMF (20 ml)の懸濁液に 60°Cで攪拌しながら参考例 100 で得たヨウ化 2 -(4 -クロロフヱニル)プロピル(純度 9 1.35 g, 4.59讓 ol)を 2回に分けて 30 分間隔で加え、 反応液を 1時間攪拌した。 反応液に 1N塩酸 (8.4ml)を加え、 メ タノ一ルー水(1:1, 20 ml)で希釈し、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール一水(1:1) およびメタノールで洗浄、 乾燥して標題化合物 (2.08 g, 3.89 mmol)を赤紫色粉末として得た。 In Reference Example 100, a suspension of the compound (1.59 g, 4.1711111101) obtained in Example 3-4 in DMF (20 ml) of potassium carbonate (8651 ^, 6.26 mmol) was stirred at 60 ° C. The obtained 2- (4-chlorophenyl) propyl iodide (purity 9 1.35 g, 4.59 benzyl) was added in two portions at 30 minute intervals, and the reaction solution was stirred for 1 hour. 1N Hydrochloric acid (8.4 ml) was added to the reaction solution, and the mixture was diluted with methanolic water (1: 1, 20 ml) and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (2.08 g, 3.89 mmol) as a red-purple powder.
'H-NMR (200MHz, CDC13) : δ 1.36 (3Η, d, J= 6.6 Hz), 1.78 (3H, q, J= 0.8 Hz), 2.25 (3H, q, J= 0.8 Hz), 2.43 (3H, s), 3.03 (1H, m), 3.12 (2H, m), 7.14 (2H, brd, J= 8.8 Hz), 7.26 (4H, m), 7.77 (2H, d, J= 8.6 Hz), 9.62 (1H, brs). 元素分析値: C24H24N303S3C1 · 0.5H20として計算値: C, 53.08; H, 4.64; N, 7.74; S, 17.71: CI, 6.53 ( ), 実測値: C, 53.27; H, 4.60; N, 7.85; S, 17.44: Cl, 6.83 ( ) . 'H-NMR (200MHz, CDC1 3): δ 1.36 (3Η, d, J = 6.6 Hz), 1.78 (3H, q, J = 0.8 Hz), 2.25 (3H, q, J = 0.8 Hz), 2.43 ( 3H, s), 3.03 (1H, m), 3.12 (2H, m), 7.14 (2H, brd, J = 8.8 Hz), 7.26 (4H, m), 7.77 (2H, d, J = 8.6 Hz), . 9.62 (1H, brs) elemental analysis: C 24 H 24 N 3 0 3 S 3 C1 · 0.5H 2 0 calculated: C, 53.08; H, 4.64 ; N, 7.74; S, 17.71: CI, 6.53 (), Found: C, 53.27; H, 4.60; N, 7.85; S, 17.44: Cl, 6.83 ().
実施例 4-155 Example 4-155
N— [8— ({ [1— (4—クロ口フエニル)シクロプロピル]メチル }スルファニル) -5, 6-ジメ チル- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン-卜ィリデ ン]- 4-メチルベンゼンスルホンアミド N— [8 — ({[1— (4-chlorophenyl) cyclopropyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-trilidene] -4-methylbenzenesulfonamide
卜 (4 -クロ口フエニル)シクロプロパンカルボン酸(7.07 g, 35.6 mmol)から参考 例 31 のエステル化の方法に従って 1- (4-クロ口フエニル)シクロプロパンカルボ ン酸メチル (純度約 80%, 7.3 g)の淡黄色油状物を得た。 According to the esterification method of Reference Example 31, methyl 4- (4-chlorophenyl) cyclopropanecarboxylate (7.07 g, 35.6 mmol) was obtained according to the esterification method of Reference Example 31 (purity about 80%, 7.3 g) of a pale yellow oil was obtained.
Ή-NMR (200MHz, CDC13): 51.16 (2H, q, J= 3.5 Hz), 1.61 (2H, q, J= 3.7 Hz), 3.63 (3H, s), 7.28 (4H, s). Ή-NMR (200MHz, CDC1 3 ): 51.16 (2H, q, J = 3.5 Hz), 1.61 (2H, q, J = 3.7 Hz), 3.63 (3H, s), 7.28 (4H, s).
1 -(4-クロ口フエニル)シクロプロパンカルボン酸メチル (純度約 80%, 7.3 g)か ら参考例 31の水素化リチウムアルミニウムを用いた還元の方法に従って 1_(4 -ク ロロフエニル)シクロプロパンメタノール(純度 93%, 5.6 g, 29 fflmol) の無色油 状物を得た。 Methyl 1- (4-chlorophenyl) cyclopropanecarboxylate (purity: about 80%, 7.3 g) was prepared according to the method of reduction using lithium aluminum hydride in Reference Example 31 to give 1_ (4-chlorophenyl) cyclopropanemethanol. A colorless oil (purity 93%, 5.6 g, 29 fflmol) was obtained.
Ή-NMR (200MHz, CDC13): (50.86 (4H, s), 3.65 (2H, s), 7.29 (4H, s). Ή-NMR (200MHz, CDC1 3 ): (50.86 (4H, s), 3.65 (2H, s), 7.29 (4H, s).
2 -(4-クロ口フエニル)プロパノール(0.24 g, 1.30讓 ol)およびメタンスルホ二 ルクロリド (0.108 ml, 1.38廳 ol) の THF (2.0 ml)溶液に、 0°Cで攪拌しながら トリェチルァミン(0.209 ml, 1.51 匪 ol)の THF (0.4 ml)溶液を 5分間かけて滴 下し、 反応液を 0°Cで 25分間攪拌した。 この反応液を、 実施例 3- 4で得た化合物 (300 mg, 0.786腿 ol)および炭酸カリウム(163 ig, 1.18 mmol)の DMF (3.5 ml) の懸濁液に 60°Cで攪拌しながら加え、混合物を 80 で 16時間攪拌した。反応液 に 1N塩酸 (2.4 ml)を加え、 メタノール一水(1:1, 10 ml)で希釈し、 室温で 30 分間攪拌した。析出物を濾取し、 メタノール一水 α:ι)で洗浄後、 メタノールから 結晶化した。 得られた濃紫色粉末(212 mg)を THFおよび酢酸ェチルから、 さらに 酢酸ェチルおよびメタノールから再結晶して標題'化合物 (134 mg, 0.245 匪 ol) を濃紫色粉末として得た。 Triethylamine (0.209 ml) was added to a solution of 2- (4-chlorophenyl) propanol (0.24 g, 1.30 benzyl) and methanesulfonyl chloride (0.108 ml, 1.38 ml) in THF (2.0 ml) while stirring at 0 ° C. , 1.51 ol) in THF (0.4 ml) was added dropwise over 5 minutes, and the reaction solution was stirred at 0 ° C for 25 minutes. This reaction solution was added to a suspension of the compound obtained in Example 3-4 (300 mg, 0.786 mol) and potassium carbonate (163 ig, 1.18 mmol) in DMF (3.5 ml) while stirring at 60 ° C. The mixture was stirred at 80 for 16 hours. 1N Hydrochloric acid (2.4 ml) was added to the reaction solution, diluted with methanol / water (1: 1, 10 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (α: ι), and then crystallized from methanol. The resulting dark purple powder (212 mg) was recrystallized from THF and ethyl acetate, and further from ethyl acetate and methanol to give the title compound (134 mg, 0.245 ol) as dark purple powder.
¾-NMR (200MHz, CDC13) : δ 0.97 (4Η, brd, J= 3.6 Hz), 1.73 (3H, brq, J= 1.2 Hz), 2.23 (3H, brq, J= 1.2 Hz), 2.44 (3H, s), 3.21 (2H, s), 7.23 (4H, m), 7.26 (2H, d, J= 8.4 Hz), 7.79 (2H, dt, J= 8.4, 1.8 Hz), 9.60 (1H, brs). ¾-NMR (200MHz, CDC1 3 ): δ 0.97 (4Η, brd, J = 3.6 Hz), 1.73 (3H, brq, J = 1.2 Hz), 2.23 (3H, brq, J = 1.2 Hz), 2.44 (3H , s), 3.21 (2H, s), 7.23 (4H, m), 7.26 (2H, d, J = 8.4 Hz), 7.79 (2H, dt, J = 8.4, 1.8 Hz), 9.60 (1H, brs) .
元素分析値: C25H24N303S3C1として計算値: C, 54.98; H, 4.43; N, 7.69; S, 17.61Elemental analysis: C 25 H 24 N 3 0 3 S 3 C1 Calculated: C, 54.98; H, 4.43 ; N, 7.69; S, 17.61
(%) 、 実測値: C, 54.76; H, 4.44; N, 7.83; S, 17.57 (%) . (%), Found: C, 54.76; H, 4.44; N, 7.83; S, 17.57 (%).
実施例 4-156 Example 4-156
N-{8- [ (2, 2 -ジフルォロ- 2-フエニルェチル)スルファニル] -5, 6-ジメチル -3-ォキ ソ- 2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン -1-イリデン }- 4-メチルベン ゼンスルホンアミド N- {8-[(2,2-difluoro-2-phenylethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene} -4-Methylbenzenezensulfonamide
実施例 3-4で得た化合物 (283 mg, 0.742 mmol)および炭酸カリウム(154 mg, 1.11 ranol)の DMF (4.5 nil)懸濁液に 60 で攪拌しながら参考例 101で得たトリ フルォロメタンスルホン酸 2, 2-ジフルォロ- 2-フエニルェチル(237 mg, 0.816 mmol)を加え、 混合物を 60°C で 4時間攪拌した。 反応液に 1N塩酸 (1.50 ml)を 加え、 室温で 30分間攪拌後、 メタノール一水(1:1, 5 ml)で希釈し、 30分間攪拌 した。析出物を濾取し、 メタノール—水(1:1)およびメタノールで洗浄後、乾燥し て標題化合物 (302 mg, 0.580靈 ol)を濃紫色粉末として得た。 To a suspension of the compound (283 mg, 0.742 mmol) obtained in Example 3-4 and potassium carbonate (154 mg, 1.11 ranol) in DMF (4.5 nil) was stirred at 60 at 60.degree. 2,2-difluoro-2-phenylethyl methanesulfonate (237 mg, 0.816 mmol) and the mixture was stirred at 60 ° C. for 4 hours. 1N Hydrochloric acid (1.50 ml) was added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes, diluted with methanol / water (1: 1, 5 ml), and stirred for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (302 mg, 0.580 eol) as a dark purple powder.
'H-NMR (200MHz, CDC13) : δ 1.77 (3Η, brs), 2.21 (3H, brs), 2.43 (3H, s), 3.61 (2H, t, J- 13.7 Hz), 7.30 (2H, d, J= 8.0 Hz), 7.41 (5H, m), 7.85 (2H, d, J= 8.0 Hz), 9.62 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 1.77 (3Η, brs), 2.21 (3H, brs), 2.43 (3H, s), 3.61 (2H, t, J- 13.7 Hz), 7.30 (2H, d , J = 8.0 Hz), 7.41 (5H, m), 7.85 (2H, d, J = 8.0 Hz), 9.62 (1H, brs).
元素分析値: C23H21N303S3F2として計算値: C, 52.96; H, 4.06; N, 8.06; S, 18.44; F, 7.28 (%), 実測値: C, 52.93; H, 4.11; N, 8.05; S, 18.50; F, 7.33 (%) . mp. 180.0-181.5°C Elemental analysis: C 23 H 21 N 3 0 3 S 3 F 2 Calculated: C, 52.96; H, 4.06 ; N, 8.06; S, 18.44; F, 7.28 (%), Found: C, 52.93; H, 4.11; N, 8.05; S, 18.50; F, 7.33 (%). Mp.180.0-181.5 ° C
実施例 4-157 Example 4-157
酢酸ェチル -ジェチルエーテルから再結晶し濃赤色結晶を得た。 Recrystallization from ethyl acetate-getyl ether gave dark red crystals.
融点 167-169°C. Melting point 167-169 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3243, 2928, 2868, 1748, 1721, 1661, 1611, 1557, 1497 cm一1. Infrared absorption spectrum (IR) (in KBr) V: 3243, 2928, 2868, 1748, 1721, 1661, 1611, 1557, 1497 cm- 1 .
Ή-NMR (CDC13) : (5 1.61-1.84 (4H, in), 1.78 (3H, d, J=0.8 Hz), 2.26 (3H, d, J=0.8 Hz), 2.42 (3H, s), 2.97 (2H, t, J=6.4 Hz), 3.33 (3H, s), 3.39 (2H, i, J-5.8 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). 元素分析値: C2()H25N304S3'0.3H20として計算値: C, 50.78; H, 5.45; N, 8.88; S, 20.34 (%), 実測値: C, 50.85; H, 5.31; N, 8.99; S, 20.04 ( ) · Ή-NMR (CDC1 3): (5 1.61-1.84 (4H, in), 1.78 (3H, d, J = 0.8 Hz), 2.26 (3H, d, J = 0.8 Hz), 2.42 (3H, s), 2.97 (2H, t, J = 6.4 Hz), 3.33 (3H, s), 3.39 (2H, i, J-5.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H , bs) elemental analysis: C 2 () H 25 N 3 0 4 S 3 '0.3H 2 0 calculated:. C, 50.78; H, 5.45; N, 8.88; S , 20.34 (%), Found: C, 50.85; H, 5.31; N, 8.99; S, 20.04 () ·
実施例 4-158 Example 4-158
N-{5,6-ジメチル - 8_ [ (2-メチル- 2-フェニルプ口ピル)スルファニル] - 3 -ォキソ - 2, 3_ジヒドロ- 1H-イミダゾ [5, 1 - c] [1,4]チアジン- 1 -ィリデン卜 4-メチルベンゼ ンスルホンアミド N- {5,6-dimethyl-8 _ [(2-methyl-2-phenylpyrpyr) sulfanyl] -3-oxo-2,3_dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene 4-methylbenzenesulfonamide
塩ィ匕 2-フエニル- 2-メチルプロピル(8.1 g, 48mmol)、 水酸化カリウム(12.7 g, 0.192 mol)および酢酸 (5.51 ml, 96腿 ol) の DMS0 (40 ml)溶液を 120 で 24 時間攪拌した。 反応液をジイソプロピルエーテル(150 ml)および水(100 ml)と混 和し、有機層を希塩酸、水および飽和食塩水で洗浄後、硫酸ナトリウムで乾燥し、 濃縮して淡褐色油状物を得た。 この油状物をシリカゲル (50 g)のクロマトグラフ ィ一 (ジェチルェ一テル—へキサン、 5:95-25:75) で精製し、 2-フエニル- 2 -メチ ルプロパノール(1.29 g, 8.59 mmol)を無色油状物として得た。 A solution of 2-phenyl-2-methylpropyl (8.1 g, 48 mmol), potassium hydroxide (12.7 g, 0.192 mol), and acetic acid (5.51 ml, 96 t ol) in DMS0 (40 ml) at 120 for 24 hours. Stirred. The reaction solution was mixed with diisopropyl ether (150 ml) and water (100 ml), and the organic layer was washed with dilute hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to obtain a pale brown oil. . The oil was chromatographed on silica gel (50 g). Purification with ethyl acetate (5: 95-25: 75) gave 2-phenyl-2-methylpropanol (1.29 g, 8.59 mmol) as a colorless oil.
]H-N R (200MHz, CDC13): (51.20 (1H, br), 1.35 (6H, s), 3.63 (2H, brs), 7.27 (1H, m), 7.34 (1H, m), 7.38 (3H, m). ] HN R (200MHz, CDC1 3 ): (51.20 (1H, br), 1.35 (6H, s), 3.63 (2H, brs), 7.27 (1H, m), 7.34 (1H, m), 7.38 (3H, m).
実施例 3- 4で得た化合物 (335 mg, 0.878 mmol), 上記 2-フエニル- 2-メチルプ ロパノール(176 mg, 1.17腿 ol) およびトリフエニルホスフィン(332 mg, 1.23 mmol)の THF (7 ml)の懸濁液に室温で攪拌しながらァゾジカルボン酸ジェチル (以下 DEAD と略すことがある、 0.204 ml, 1.23 mmol)を滴下し、 反応液を室温で 15時間、 ついで 6(TC で 7時間攪拌した。 反応液を酢酸ェチルで希釈し、 2¾炭酸 水素ナトリゥム、 0.1 塩酸、水および飽和食塩水で洗浄後、硫酸ナトリゥムで乾 燥し、 濃縮した。 残渣をメタノールから結晶化して標題化合物 (253 mg, 0.493 mmol)を濃紫色粉末として得た。 The compound (335 mg, 0.878 mmol) obtained in Example 3-4, 2-phenyl-2-methylpropanol (176 mg, 1.17 mmol) and triphenylphosphine (332 mg, 1.23 mmol) in THF (7 ml) were used. ) Was added dropwise to the suspension at room temperature with stirring at room temperature, and the reaction solution was stirred at room temperature for 15 hours, and then at 6 (TC for 7 hours). The reaction mixture was diluted with ethyl acetate, washed with sodium bicarbonate, 0.1 hydrochloric acid, water and saturated saline, dried over sodium sulfate and concentrated, and the residue was crystallized from methanol to give the title compound (253 mg, 0.493 mmol) as a dark purple powder.
^- R (200MHz, CDC13) : δ 1.44 (6Η, s), 1.76 (3H, q, J= 1.2 Hz), 2.22 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 3.24 (2H, s), 7.24 (2H, d, J= 8 Hz), 7.29 (3H, m), 7.30 (2H, d, J= 8.4 Hz), 7.76 (2H, d, J= 8.4 Hz), 9.55 (1H, brs). 元素分析値: C25H27N303S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73 ^ - R (200MHz, CDC1 3 ): δ 1.44 (6Η, s), 1.76 (3H, q, J = 1.2 Hz), 2.22 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 3.24 (2H, s), 7.24 (2H, d, J = 8 Hz), 7.29 (3H, m), 7.30 (2H, d, J = 8.4 Hz), 7.76 (2H, d, J = 8.4 Hz), 9.55 . (1H, brs) elemental analysis: C 25 H 27 N 3 0 3 S 3 calculated: C, 58.45; H, 5.30 ; N, 8.18; S, 18.73
(%), 実測値: C, 58.23; H, 5.19; N, 8.28; S, 18.68 (%) . (%), Found: C, 58.23; H, 5.19; N, 8.28; S, 18.68 (%).
mp. 183.5-185.2°C mp. 183.5-185.2 ° C
実施例 4-159 Example 4-159
N-(5,6-ジメチル- 3-ォキソ - 8 - {[2-(3-チェニル)ェチル]スルファニル }-2,3-ジヒ ド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン) -4-メチルベンゼンスルホ ンアミド N- (5,6-Dimethyl-3-oxo-8-{[2- (3-Chenyl) ethyl] sulfanyl} -2,3-dihydrido-1H-imidazo [5,1-c] [1, 4] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
(方法 1) 2- (3-チェニル)エタノール (0.21 g, 1.61 mmol)およびメタンスルホ ニルクロリド (0.138 ml, 1.77顧 ol) の THF(4.2 ml)溶液に、 0°Cで攪拌しなが らトリェチルァミン(0.267 ml, 1.93 mmol)を加え、 反応液を 0°Cで 60分間攪拌 した。 この反応液を酢酸ェチルで抽出し、 水洗、 乾燥、 濃縮乾固して粗製のメタ ンスルホン酸 2- (3-チェニル)ェチル (純度 60%, 104 mg, 0.30 mmol)を無色油状 物として得た。 (Method 1) To a solution of 2- (3-thenyl) ethanol (0.21 g, 1.61 mmol) and methanesulfonyl chloride (0.138 ml, 1.77) in THF (4.2 ml) while stirring at 0 ° C was added triethylamine (0.1 ml). (0.267 ml, 1.93 mmol) was added, and the reaction solution was stirred at 0 ° C for 60 minutes. The reaction solution was extracted with ethyl acetate, washed with water, dried and concentrated to dryness to obtain crude 2- (3-thenyl) ethyl methanesulfonate (purity 60%, 104 mg, 0.30 mmol) as a colorless oil. .
'H-NMR (200MHz, CDC13): 52.90 (3H, s), 3.10 (2H, t, J= 6.8 Hz), 4.42 (2H, t, J= 6.8 Hz), 6.99 (1H, dd, J= 4.8, 1.3 Hz), 7.10 (1H, m), 7.31 (1H, dd, J二 5.0, 2.9 Hz). 'H-NMR (200MHz, CDC1 3): 52.90 (3H, s), 3.10 (2H, t, J = 6.8 Hz), 4.42 (2H, t, J = 6.8 Hz), 6.99 (1H, dd, J = 4.8, 1.3 Hz), 7.10 (1H, m), 7.31 (1H, dd, J2 5.0, 2.9 Hz).
実施例 3-4 で得た化合物 (110 mg, 0.288腿 ol)および炭酸カリウム(60 mg, 0.433 mmol)の DMF (2.0 ml)の懸濁液に 80°Cで攪拌しながら上記メタンスルホン 酸 2- (3-チェニル)ェチル(純度 60 , 102 mg, 0.297腿 ol) の DMF (1.0 ml)溶液 を加え、混合物を 80°C で 1時間、 ついで 60°C で 12時間攪拌した。反応液に 1N 塩酸 (0.58 ml)および水(20 ml)を加え、酢酸ェチルで抽出した。有機層を 0.1N塩 酸、 水および飽和食塩水で洗净後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣を メタノールから結晶化して標題化合物 (62 mg, 0.126匪 ol)を紫色粉末として得 た。 The above methanesulfonic acid 2 was added to a suspension of the compound obtained in Example 3-4 (110 mg, 0.288 mol) and potassium carbonate (60 mg, 0.433 mmol) in DMF (2.0 ml) while stirring at 80 ° C. -A solution of (3-Chenyl) ethyl (purity 60, 102 mg, 0.297 t) in DMF (1.0 ml) was added and the mixture was stirred at 80 ° C for 1 hour and then at 60 ° C for 12 hours. 1N Hydrochloric acid (0.58 ml) and water (20 ml) were added to the reaction solution, and extracted with ethyl acetate. The organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was crystallized from methanol to give the title compound (62 mg, 0.126 ol) as a purple powder.
¾— NMR (200MHz, CDC13) : δ 1.79 (3Η, q, J= 0.8 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.43 (3H, s), 3.00 (2H, brt, J= 7.7 Hz), 3.18 (2H, brt, J= 7.4 Hz), 6.95 (1H, dd, J= 4.8, 1.6 Hz), 7.01 (1H, m), 7.27 (2H, m), 7.28 (2H, d, J= 8.4 Hz), 7.83 (2H, d, J- 8.4 Hz), 9.63 (1H, brs). ¾- NMR (200MHz, CDC1 3) : δ 1.79 (3Η, q, J = 0.8 Hz), 2.26 (3H, q, J = 0.8 Hz), 2.43 (3H, s), 3.00 (2H, brt, J = 7.7 Hz), 3.18 (2H, brt, J = 7.4 Hz), 6.95 (1H, dd, J = 4.8, 1.6 Hz), 7.01 (1H, m), 7.27 (2H, m), 7.28 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J-8.4 Hz), 9.63 (1H, brs).
元素分析値: C21H21N303S4 · 0.2H20として計算値: C, 50.93; H, 4.35; N, 8.48; S, 25.90 (%)、 実測値: C, 50.74; H, 4.29; N, 8.81; S, 25.86 (%) · Elemental analysis: C 21 H 21 N 3 0 3 S 4 · 0.2H 2 0 Calculated: C, 50.93; H, 4.35; N, 8.48; S, 25.90 (%), Observed: C, 50.74; H , 4.29; N, 8.81; S, 25.86 (%)
mp. 182.0- 183.0 mp. 182.0- 183.0
(方法 2) 実施例 3- 4で得た化合物 (240 mg, 0.629 mmol)、 2-(3-チェニル)エタ ノール(82mg, 0.629 mmol) およびトリフエニルホスフィン(179 mg, 0.661 mmol) の THF (5 ml)の懸濁液に室温で攪拌しながら DEAD (0.109 ml, 0.661 腿 ol)を滴 下し、 反応液を室温で 3時間攪拌した。 反応液を酢酸ェチルで希釈し、 2%炭酸水 素ナトリゥム、 0.1 塩酸、水および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥 し、 濃縮した。 残渣を酢酸ェチルから結晶化し、 さらに再度 THFから結晶化して 標題化合物(166 mg, 0.338 mmol)を紫色粉末として得た。 (Method 2) THF (179 mg, 0.661 mmol) of the compound (240 mg, 0.629 mmol), 2- (3-thenyl) ethanol (82 mg, 0.629 mmol) and triphenylphosphine (179 mg, 0.661 mmol) obtained in Example 3-4 was used. DEAD (0.109 ml, 0.661 t) was added dropwise to the 5 ml) suspension while stirring at room temperature, and the reaction solution was stirred at room temperature for 3 hours. The reaction solution was diluted with ethyl acetate, washed with 2% sodium hydrogen carbonate, 0.1 hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was crystallized from ethyl acetate and again from THF to give the title compound (166 mg, 0.338 mmol) as a purple powder.
実施例 4 - 160 Example 4-160
N- (5, 6-ジメチル- 3-ォキソ -8- { [2- (2-チェニル)ェチル]スルファ二ル} -2, 3-ジヒ ド口- 1H-ィミダゾ [5, l-c] [1, ]チアジン- 1-ィリデン) -4-メチルベンゼンスルホ ンアミド N- (5,6-Dimethyl-3-oxo-8-{[2- (2-Chenyl) ethyl] sulfanyl} -2,3-dihydrogen-1H-imidazo [5, lc] [1, ] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
2- (2-チェニル)エタノ一ル(5.78 g, 44.2删 ol)およびメ夕ンスルホニルク口リ ド (3.63 ml, 46. 腿 ol) の THF(90 ml)溶液に、 0°Cで攪拌しながらトリェチル ァミン(7.04 ml, 50.8腿 ol)を 40分間かけて加え、 反応液を 0°Cで 60分間攪拌 した。 この反応液を酢酸ェチル -へキサン (1:1) で抽出し、 水洗、 乾燥、 濃縮乾 固してメタンスルホン酸 2- (2-チェニル)ェチル (純度 94%, 9.7 g, 44.2腿 ol)を 無色油状物として得た。 2- (2-Chenyl) ethanol (5.78 g, 44.2 olol) Triethylamine (7.04 ml, 50.8 t) was added to THF (90 ml) solution of the compound (3.63 ml, 46.t) at 40 ° C with stirring at 0 ° C for 40 minutes, and the reaction solution was added at 0 ° C. Stirred for 60 minutes. The reaction mixture was extracted with ethyl acetate-hexane (1: 1), washed with water, dried and concentrated to dryness. 2- (2-Chenyl) ethyl methanesulfonate (purity 94%, 9.7 g, 44.2 t ol) Was obtained as a colorless oil.
Ή-NMR (200MHz, CDC13): 52.93 (3H, s), 3.28 (2H, dt, J= 0.6, 6.6 Hz), 4.42 (2H, t, J= 6.6 Hz), 6.91 (1H, m), 6.96 (1H, dd, J= 5.2, 3.4 Hz), 7.20 (1H, dd, J= 5.0, 1.0 Hz). Ή-NMR (200MHz, CDC1 3 ): 52.93 (3H, s), 3.28 (2H, dt, J = 0.6, 6.6 Hz), 4.42 (2H, t, J = 6.6 Hz), 6.91 (1H, m), 6.96 (1H, dd, J = 5.2, 3.4 Hz), 7.20 (1H, dd, J = 5.0, 1.0 Hz).
実施例 3-4で得た化合物 (403 mg, 1.06 mmol)および炭酸カリウム(219 mg, 1.58 膽 ol)の DMF (6.0 ml)の懸濁液に 80°Cで攪拌しながら上記メタンスルホン酸 2 -(2-チェニル)ェチル(純度 94%, 278 mg, 1.27 mmol)の DMF (1.0 ml)溶液を加 え、 混合物を 80 で 4時間攪拌した。 反応液に 1N塩酸 (2.1 ml)およびメタノ 一ルー水(1:1, 10ml)を加え、 室温で 30分間攪拌した。 析出物を濾取し、 メタノ 一ルー水(1:1)およびメタノールで洗浄後、 乾燥して標題化合物 (430 mg, 0.875 mmol)を紫色粉末として得た。 The above methanesulfonic acid 2 was added to a suspension of the compound obtained in Example 3-4 (403 mg, 1.06 mmol) and potassium carbonate (219 mg, 1.58 toluene) in DMF (6.0 ml) while stirring at 80 ° C. A solution of-(2-Chenyl) ethyl (purity 94%, 278 mg, 1.27 mmol) in DMF (1.0 ml) was added, and the mixture was stirred at 80 for 4 hours. To the reaction solution were added 1N hydrochloric acid (2.1 ml) and methanolic water (1: 1, 10 ml), and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol (1: 1) and methanol, and dried to give the title compound (430 mg, 0.875 mmol) as a purple powder.
'H-NMR (200MHz, CDC13) : 6 1.79 (3H, q, J= 0.8 Hz), 2.27 (3H, q, J= 0.6 Hz), 2.43 (3H, s), 3.20 (4H, s), 6.85 (1H, dd, J= 3.6, 1.0 Hz), 6.94 (1H, dd, J= 5.1, 3.3 Hz), 7.18 (1H, dd, J= 5.1, 1.0 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). 'H-NMR (200MHz, CDC1 3): 6 1.79 (3H, q, J = 0.8 Hz), 2.27 (3H, q, J = 0.6 Hz), 2.43 (3H, s), 3.20 (4H, s), 6.85 (1H, dd, J = 3.6, 1.0 Hz), 6.94 (1H, dd, J = 5.1, 3.3 Hz), 7.18 (1H, dd, J = 5.1, 1.0 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C21H21N303S4として計算値: C, 51.30; H, 4.31; , 8.55; S, 26.09 (%), 実測値: C, 51.25; H, 4.34; N, 8.77; S, 25.88 (%) . Elemental analysis: Calculated for C 21 H 21 N 3 0 3 S 4 : C, 51.30; H, 4.31;, 8.55; S, 26.09 (%), found: C, 51.25; H, 4.34; N, 8.77 ; S, 25.88 (%).
mp. 172.0-173.5°C mp.172.0-173.5 ° C
実施例 4-161 Example 4-161
酢酸ェチル-ジイソプロピルエーテルから再結晶し濃赤色結晶を得た。 Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals.
融点 118-120°C. Melting point 118-120 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3250, 2936, 2863, 1728, 1661, 1615, 1557, 1495 cnr1. Infrared absorption spectrum (IR) (in KBr): 3250, 2936, 2863, 1728, 1661, 1615, 1557, 1495 cnr 1 .
'H-NMR (CDC13) : δ 1.37-1.77 (6Η, m), 1.78 (3H, d, J=0.8 Hz), 2.26 (3H, s), 2.42 (3H, d, J=0.8 Hz), 2.94 (2H, t, J=7.8 Hz), 3.34 (3H, s), 3.37 (2H, t, J=5.8 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.61 (1H, bs). 元素分析値: C21H27N304S3として計算値: C, 52.37; H, 5.65; N, 8.72; S, 19.97 (%), 実測値: C, 52.03; H, 5.69; N, 8.98; S, 20.15 (%) . 'H-NMR (CDC1 3) : δ 1.37-1.77 (6Η, m), 1.78 (3H, d, J = 0.8 Hz), 2.26 (3H, s), 2.42 (3H, d, J = 0.8 Hz), 2.94 (2H, t, J = 7.8 Hz), 3.34 (3H, s), 3.37 (2H, t, J = 5.8 Hz), 7.29 ( 2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.61 (1H, bs) Elemental analysis:. C 21 H 27 N 3 0 4 S Calculated as 3 : C, 52.37; H, 5.65; N, 8.72; S, 19.97 (%), Found: C, 52.03; H, 5.69; N, 8.98; S, 20.15 (%).
実施例 4-162 Example 4-162
酢酸ェチル -ジェチルェ一テルから再結晶し赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain red crystals.
融点 190- 192°C. 190-192 ° C.
赤外吸収スペクトル(IR) (KBr中) V 3243, 2973, 2932, 2826, 1748, 1732, 1717, 1661 cnr1. Infrared absorption spectrum (IR) (in KBr) V 3243, 2973, 2932, 2826, 1748, 1732, 1717, 1661 cnr 1 .
Ή-NMR (CDC13) : δ 1.19 (6Η, s), 1.79 (3H, s), 1.75-1.88 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, t, J=8.4 Hz), 3.18 (3H, s), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.6 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 1.19 (6Η, s), 1.79 (3H, s), 1.75-1.88 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, t, J = 8.4 Hz), 3.18 (3H, s), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.6 Hz), 9.60 (1H, bs).
元素分析値: C21H27N304S3として計算値: C, 52.37; H, 5.65; N, 8.72; S, 19.97Elemental analysis: C 21 H 27 N 3 0 4 S 3 Calculated: C, 52.37; H, 5.65 ; N, 8.72; S, 19.97
(%), 実測値: C, 52.03; H, 5.65; N, 8.79; S, 19.65 (%) . (%), Found: C, 52.03; H, 5.65; N, 8.79; S, 19.65 (%).
実施例 4-163 Example 4-163
酢酸ェチル -ジェチルェ一テルから再結晶し赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain red crystals.
融点: 162- 164°C. Melting point: 162-164 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3700-3100, 2696, 1732, 1663, 1613, 1557 cm"1. ¾—應 R (CDCI3) : δ 1.27 (6Η, s), 1.79 (3H, s), 1.82 (2H, t, J=8.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 3.06 (2H, t, J=8.8 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.2 Hz), 9.63 (1H, bs). Infrared absorption spectrum (IR) (in KBr) Re: 3700-3100, 2696, 1732, 1663, 1613, 1557 cm " 1. ¾-R (CDCI 3 ): δ 1.27 (6Η, s), 1.79 (3H , s), 1.82 (2H, t, J = 8.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 3.06 (2H, t, J = 8.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.63 (1H, bs).
元素分析値: C20H25N304S3 · 0.3H20として計算値: C, 50.78; H, 5.45; N, 8.88; S, 20.34 (%), 実測値: C, 50.87; H, 5.44; N, 8.50; S, 19.97 (%) . Elemental analysis: C 20 H 25 N 3 0 4 S 3 · 0.3H 2 0 Calculated: C, 50.78; H, 5.45 ; N, 8.88; S, 20.34 (%), Found: C, 50.87; H , 5.44; N, 8.50; S, 19.97 (%).
実施例 4-164 Example 4-164
N- [8- (シクロォクチルスルファニル) -5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H- イミダゾ [5, 1-c] [1, 4]チアジン- 1-イリデン ]_4-メチルベンゼンスルホンアミド 実施例 3-4 で得た化合物 (1.85 g, 4.85 mmol)および炭酸カリウム (2.82 g, 20.4腿01) の DMF (80 ml) の懸濁液に、 60°Cで攪拌しながらョ一ドシクロォク タン(純度 75%, 4.34 g, 13.6 mmol)の DMF (10 ml)溶液を 2時間かけて滴下した。 反応液を 60°Cで 4時間攪拌した。反応中にョードシクロオクタン (純度 75%, 0.43 g, 1.36 iMiol)を 3回加えた。 反応液に IN塩酸 (30 ml)を加え、 水 (50 ml)で希 釈し、室温で 60分間攪拌した。析出物を濾取し、 メタノール一水 (1:1)およびメ 夕ノールで洗浄後、 乾燥して粗製の化合物 4-164 (純度 80%、 5.45g, 8.87mmol) を赤紫色粉末として得た。 この粉末 (1.40 g) を THF-酢酸ェチルから結晶化し、 表題化合物 (837 nig, 1.70蘭 ol)を赤紫色粉末として得た。 N- [8- (Cyclooctylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] _4 -Methylbenzenesulfonamide A suspension of the compound obtained in Example 3-4 (1.85 g, 4.85 mmol) and potassium carbonate (2.82 g, 20.4 thigh 01) in DMF (80 ml) was stirred at 60 ° C. Meanwhile, a solution of o-cyclocyclotanne (purity 75%, 4.34 g, 13.6 mmol) in DMF (10 ml) was added dropwise over 2 hours. The reaction solution was stirred at 60 ° C for 4 hours. During the reaction, eodocyclooctane (purity 75%, 0.43 g, 1.36 iMiol) was added three times. IN hydrochloric acid (30 ml) was added to the reaction solution, diluted with water (50 ml), and stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give crude compound 4-164 (purity 80%, 5.45 g, 8.87 mmol) as a magenta powder . This powder (1.40 g) was crystallized from THF-ethyl acetate to give the title compound (837 nig, 1.70 ol) as a purple-red powder.
Ή-NMR (200MHz, CDC13) : δ 1.50-1.80 (10H, m), 1.79 (3H, brs), 1.98 (4H, m), 2.26 (3H, brs), 2.42 (3H, s), 3.59 (1H, m), 7.28 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.0 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.50-1.80 (10H, m), 1.79 (3H, brs), 1.98 (4H, m), 2.26 (3H, brs), 2.42 (3H, s), 3.59 ( 1H, m), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.0 Hz), 9.62 (1H, brs).
元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56 ( ) 、 実測値: C, 55.81; H, 6.04; N, 8.54; S, 19.66 (%) . Elemental analysis: C 23 H 29 N 3 0 3 S 3 Calculated: C, 56.18; H, 5.94 ; N, 8.55; S, 19.56 (), Found: C, 55.81; H, 6.04 ; N, 8.54 ; S, 19.66 (%).
nip. 166.5-169.0°C nip.166.5-169.0 ° C
実施例 4-165 Example 4-165
酢酸ェチル -ジェチルェ一テルから再結晶し赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain red crystals.
融点 195-197°C. Mp 195-197 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3220, 2959, 1740, 1661, 1622, 1564 cm"1. Ή-NMR (CDC13) : δ 1.03 (9Η, s), 1.79 (3H, s), 2.26 (3H, s), 2.41 (3H, s), 2.89 (2H, s), 7.27 (2H, d, J=8.0 Hz), 7.87 (2H, d, J=8.4 Hz), 9.58 (1H, bs). 実施例 4-166 Infrared absorption spectrum (IR) (in KBr) Re:. 3220, 2959, 1740, 1661, 1622, 1564 cm "1 Ή-NMR (CDC1 3): δ 1.03 (9Η, s), 1.79 (3H, s) , 2.26 (3H, s), 2.41 (3H, s), 2.89 (2H, s), 7.27 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.58 (1H, bs). Example 4-166
N-{5, 6-ジメチル -8- [ (4-メチルフエニル)スルファニル] -3-ォキソ -2, 3-ジヒドロ - 1H-イミダゾ [5,1 - c] [1,4]チアジン-卜ィリデン }- 4-メチルベンゼンスルホンァ ^ド N- {5,6-dimethyl-8-[(4-methylphenyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene} -4-methylbenzenesulfone
実施例 3- 4で得た化合物 (259 mg, 0.679匪 ol)、 p-トルエンチオール (103 mg, 0.815腿 ol) およびトリフエニルホスフィン(275 mg, 1.02腿 ol)の THF (5 ml) の懸濁液に室温で攪拌しながらァゾジカルボン酸ジイソプロピル (以下 DIAD と 略すことがある、 0.20.7 ml, 1.02 mmol)を滴下し、 反応液を室温で 3日間、 さら に 60°Cで 2日間攪拌した。 この間トリフエニルホスフィン(184 mg, 0.679腿 ol) および DIAD(0.138 ml, 0.679腿 ol)を反応液に加えた。 反応液を酢酸ェチルで希 釈し、 2 炭酸水素ナトリウム、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナ トリウムで乾燥し、濃縮した。残渣をシリカゲル(10g)のクロマトグラフィー(酢 酸ェチルーへキサン、 2:8-3:7) で精製し、 酢酸ェチルーへキサン(3:7)の赤色画 分をメタノールから結晶化して標題化合物 (112 mg, 0.237顏 ol)を赤紫色粉末と して得た。 A suspension of the compound obtained in Example 3-4 (259 mg, 0.679 ol), p-toluenethiol (103 mg, 0.815 ol) and triphenylphosphine (275 mg, 1.02 ol) in THF (5 ml) was used. Diisopropyl azodicarboxylate (hereinafter sometimes referred to as DIAD, 0.20.7 ml, 1.02 mmol) was added dropwise to the suspension while stirring at room temperature, and the reaction solution was stirred at room temperature for 3 days and further at 60 ° C for 2 days. . During this time, triphenylphosphine (184 mg, 0.679 t ol) and DIAD (0.138 ml, 0.679 t ol) were added to the reaction solution. The reaction solution was diluted with ethyl acetate, washed with sodium bicarbonate, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was chromatographed on silica gel (10 g) (vinegar Purification with ethyl acetate-hexane (2: 8-3: 7), and the red fraction of ethyl acetate-hexane (3: 7) was crystallized from methanol to give the title compound (112 mg, 0.237 mol) as a purple-red powder. I got it.
Ή-NMR (200MHz, CDC13) : δ 1.60 (3Η, brs), 2.23 (3H, brs), 2.41 (3H, s), 2.43 (3H, s), 7.20 (2H, brd, J- 8.0 Hz), 7.31 (2H, d, J= 8.4 Hz), 7.42 (2H, d, J = 8.0 Hz), 7.89 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.60 (3Η, brs), 2.23 (3H, brs), 2.41 (3H, s), 2.43 (3H, s), 7.20 (2H, brd, J- 8.0 Hz) , 7.31 (2H, d, J = 8.4 Hz), 7.42 (2H, d, J = 8.0 Hz), 7.89 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C22H21N303S3として計算値: C, 56.03; H, 4.49; N, 8.91; S, 20.40Elemental analysis: C 22 H 21 N 3 0 3 S 3 Calculated: C, 56.03; H, 4.49 ; N, 8.91; S, 20.40
(%), 実測値: C, 55.97; H, 4.45; N, 8.76; S, 20.22 ( ) . (%), Found: C, 55.97; H, 4.45; N, 8.76; S, 20.22 ().
mp. 190.5-191.51: mp. 190.5-191.51:
実施例 4-167 Example 4-167
N- {8- [ (2, 2-ジフルォロ- 3-メチルプチル)スルファニル] -5, 6-ジメチル- 3-ォキソ - 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン }- 4 -メチルベンゼ ンスルホンアミド N- {8-[(2,2-difluoro-3-methylbutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene} -4-Methylbenzenesulfonamide
(方法 1)実施例 3- で得た化合物 (195 mg, 0.511讓 ol)、参考例 101で得た 2, 2 - ジフルォロイソペンタノール (純度 20%, 0.34 g, 0.55 mmol) およびトリフエ二 ルホスフィン(553 mg, 2.04匪01)の:[11? (4 ml)の懸濁液に 60 で攪拌しながら DIAD (0.415 ml, 2.04腿01)の1^ (1 ml)溶液を 3時間かけて滴下し、 反応液を 3時間攪拌した。 反応液を酢酸ェチルで希釈し、 2%炭酸水素ナトリウム、 0.1N塩 酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣を シリカゲル(10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 15:85-30:70) で精製し、酢酸ェチルーへキサン(20: 80-25 :75)の赤色画分をメタノールから結晶 化して標題化合物 (22 rag, 0.045匪 ol)を濃紫色粉末として得た。 (Method 1) Compound (195 mg, 0.511 alcohol) obtained in Example 3-, 2,2-difluoroisopentanol (purity 20%, 0.34 g, 0.55 mmol) obtained in Reference Example 101 and trifluorophenol Nilphosphine (553 mg, 2.04 bandit 01): [11? A 1 ^ (1 ml) solution of DIAD (0.415 ml, 2.04 thigh 01) was added dropwise to the (4 ml) suspension with stirring at 60 over 3 hours, and the reaction solution was stirred for 3 hours. The reaction solution was diluted with ethyl acetate, washed with 2% sodium hydrogencarbonate, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was purified by chromatography on silica gel (10 g) (ethyl acetate-hexane, 15: 85-30: 70), and the red fraction of ethyl acetate-hexane (20: 80-25: 75) was crystallized from methanol. The title compound (22 rag, 0.045 ol) was obtained as a deep purple powder.
Ή— NMR (200MHz, CDC13) : δ 1.03 (6H, d, J= 7.0 Hz), 1.80 (3H, brs), 2.19 (1H, tq, J= 14.8, 7.0 Hz), 2.25 (3H, brs), 2.42 (2H, s), 3.37 (2H, t, J= 14.8 Hz) , 7.29 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.0 Hz) , 9.63 (1H, brs). Ή- NMR (200MHz, CDC1 3) : δ 1.03 (6H, d, J = 7.0 Hz), 1.80 (3H, brs), 2.19 (1H, tq, J = 14.8, 7.0 Hz), 2.25 (3H, brs) , 2.42 (2H, s), 3.37 (2H, t, J = 14.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.0 Hz), 9.63 (1H, brs) .
元素分析値: C2。H23N303S3F2として計算値: C, 49.26; H, 4.75; N, 8.62; S, 19.73 (%)、 実測値: C, 49.59; H, 4.71; , 8.61; S, 19.93 (%) . Elemental analysis: C 2. Calculated as H 23 N 3 0 3 S 3 F 2 : C, 49.26; H, 4.75; N, 8.62; S, 19.73 (%), found: C, 49.59; H, 4.71;, 8.61; S, 19.93 (%).
(方法 2)実施例 3-4で得た化合物 (591 mg, 1.55 mmol)および炭酸力リゥム (321 mg, 2.32腳01) の DMF (10 ml) の懸濁液に 60でで攪拌しながら参考例 102で得 たトリフルォロメタンスルホン酸 2, 2-ジフルォロイソペンチル (純度 60%, 187 mg, 0.517IMO1)を 1時間毎に 3回加え、 さらに反応液を 4時間攪拌した。 反応液 に 1N塩酸 (3. l ml)を加え、 メタノール一水(1:1, 5 ml)で希釈し、 室温で 30分 間攪拌した。 析出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄、 乾燥して標題化合物 (487 mg, 0.999匪 ol)を紫色粉末として得た。 (Method 2) A suspension of the compound (591 mg, 1.55 mmol) obtained in Example 3-4 and DMF (10 ml) of carbonated carbonate (321 mg, 2.32 2.301) was stirred at 60 with reference to Example 102 2,2-Difluoroisopentyl trifluoromethanesulfonate (purity 60%, 187 mg, 0.517IMO1) was added three times every hour, and the reaction solution was further stirred for 4 hours. 1N Hydrochloric acid (3.1 ml) was added to the reaction solution, diluted with methanol / water (1: 1, 5 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (487 mg, 0.999 bandol) as a purple powder.
即. 168.5-169.5t Immediately. 168.5-169.5t
実施例 4-168 Example 4-168
N- (5, 6-ジメチル _3 -ォキソ - 8 - {[2- (4-ピリジニル)ェチル]スルファニル }_2, 3 -ジ ヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン)-4-メチルベンゼンスル ホンアミド N- (5,6-dimethyl_3-oxo-8-{[2- (4-pyridinyl) ethyl] sulfanyl} _2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物 (276 mg, 0.723 mmol), 2 - (4-ピリジル)エタノール塩 酸塩(173 mg, 1.09 mmol) およびトリフエニルホスフィン(783 mg, 2.89 mmol) の THF (5 ml)の懸濁液に 60°Cで攪拌しながら DIAD (0.587 ml, 2.89腿 ol)の THF (1 mi)溶液を 40分間かけて滴下後、 反応液を 2時間攪拌した。反応液を酢酸ェチ ル(20 ml)で希釈し、析出物を濾取後酢酸ェチルおよびジェチルエーテルで洗浄し、 メタノールおよび THFに懸濁した。 懸濁液を濃縮後ァセトニトリルで希釈し、 不 溶物を濾取してァセトニトリルで洗浄、 乾燥して標題化合物 (47 mg, 0.097 mmol) を灰紫色粉末として得た。 The compound (276 mg, 0.723 mmol) obtained in Example 3-4, 2- (4-pyridyl) ethanol hydrochloride (173 mg, 1.09 mmol) and triphenylphosphine (783 mg, 2.89 mmol) in THF (5 A suspension of DIAD (0.587 ml, 2.89 fold) in THF (1 mi) was added dropwise to the suspension of the resulting mixture at 60 ° C. over 40 minutes, and the reaction solution was stirred for 2 hours. The reaction solution was diluted with ethyl acetate (20 ml), and the precipitate was collected by filtration, washed with ethyl acetate and getyl ether, and suspended in methanol and THF. The suspension was concentrated and diluted with acetonitrile. The insolubles were collected by filtration, washed with acetonitrile, and dried to obtain the title compound (47 mg, 0.097 mmol) as a gray-purple powder.
Ή - NMR (200MHz, CDC13+TFA) : δ 1.82 (3H, br), 2.27 (3H, brs), 2.43 (3H, s), 3.30 (4H, s), 7.30 (2H, d, J= 8.0 Hz), 7.81 (2H, d, J= 8.0 Hz), 7.84 (2H, d, J= 5.2 Hz), 8.91 (2H, d,'J= 6.2 Hz), 9.96 (1H, br). Ή - NMR (200MHz, CDC1 3 + TFA): δ 1.82 (3H, br), 2.27 (3H, brs), 2.43 (3H, s), 3.30 (4H, s), 7.30 (2H, d, J = 8.0 Hz), 7.81 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 5.2 Hz), 8.91 (2H, d, 'J = 6.2 Hz), 9.96 (1H, br).
元素分析値: C22H22N403S3 ·Η20として計算値: C, 52.36; Η, 4.79; Ν, 11.10 ( ), 実測値: C, 52.60; H, 4.91; , 11.02 (%) . Elemental analysis: C 22 H 22 N 4 0 3 S 3 · Η 20 Calculated: C, 52.36; Η, 4.79; Ν, 11.10 (), found: C, 52.60; H, 4.91;, 11.02 ( %).
実施例 4-169 Example 4-169
酢酸ェチル-ジェチルエーテルから再結晶し赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-getyl ether to obtain red crystals.
融点 157-159°C. 157-159 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3241, 1746, 1725, 1663, 1618, 1561 cm一1. 'H-NMR (CDC13) : δ 1.75 (3H, s), 1.79 (3H, d, J-0.8 Hz), 2.27 (3H, d, J=0.8 Hz), 2.37 (2H, t, J=7.8 Hz), 2.42 (3H, s), 3.05 (2H, t, J=8.4 Hz), 4.76 (IH, s), 4.83 (1H, s), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.60 (1H, bs). Infrared absorption spectrum (IR) (in KBr) V:. 3241, 1746 , 1725, 1663, 1618, 1561 cm one 1 'H-NMR (CDC1 3 ): δ 1.75 (3H, s), 1.79 (3H, d , J-0.8 Hz), 2.27 (3H, d, J = 0.8 Hz), 2.37 (2H, t, J = 7.8 Hz), 2.42 (3H, s), 3.05 (2H, t, J = 8.4 Hz), 4.76 (IH, s), 4.83 (1H, s), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.60 (1H, bs).
元素分析値: C2。H23N303S3として計算値: C, 53.43; H, 5.16; N, 9.35; S, 21.40 (%)、 実測値: C, 53.40; H, 5.26; N, 9.06; S, 21.06 (%) . Elemental analysis: C 2. H 23 N 3 0 3 S 3 Calculated: C, 53.43; H, 5.16; N, 9.35; S, 21.40 (%); Found: C, 53.40; H, 5.26; N, 9.06; S, 21.06 ( %).
実施例 4-170 Example 4-170
N_(5,6-ジメチル - 3_ォキソ - 8- {[4- (トリフルォロメチル)フエニル]スルファニ ル卜 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン)-4-メチルベ ンゼンスルホンアミド N_ (5,6-Dimethyl-3_oxo-8-{[4- (trifluoromethyl) phenyl] sulfanyl 2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物 (259 ing, 0·679 ΜΙΟ1)、 4-トリフルォロメチルベンゼ ンチォ一ル (234 mg, 1.31 画 ol) およびトリフエニルホスフィン(734 mg, 2.72 匪 ol)の THF (5 ml)の懸濁液に室温で攪拌しながら DEAD (0.450 ml, 2.72 mmol) を加え、 反応液を 6日間攪拌した。 反応液を酢酸ェチルで希釈し、 2%炭酸水素ナ トリウム水溶液、 0.1N塩酸、水および飽和食塩水で洗浄後、硫酸ナトリウムで乾 燥し、 濃縮した。 残渣をシリカゲル (10 g)のクロマトグラフィー (酢酸ェチルー へキサン、 1:9-5:5) に付した。 得られた粗画分を再度シリカゲル(10 g).のクロマ トグラフィー (酢酸ェチルーへキサン、 1:9-3:7) に付し、 酢酸ェチルーへキサン (25:75)の赤色画分をメタノールから結晶化して標題化合物 (115 mg, 0.219 腿 ol)を赤橙色粉末として得た。 Of the compound (259 ing, 0 · 679-1) obtained in Example 3-4, 4-trifluoromethylbenzenthiol (234 mg, 1.31 ol) and triphenylphosphine (734 mg, 2.72 ol) To a suspension of THF (5 ml) was added DEAD (0.450 ml, 2.72 mmol) while stirring at room temperature, and the reaction solution was stirred for 6 days. The reaction solution was diluted with ethyl acetate, washed with a 2% aqueous sodium hydrogen carbonate solution, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was chromatographed on silica gel (10 g) (ethyl acetate-hexane, 1: 9-5: 5). The obtained crude fraction was again subjected to chromatography on silica gel (10 g) (ethyl acetate-hexane, 1: 9-3: 7), and a red fraction of ethyl acetate-hexane (25:75) was obtained. Crystallization from methanol gave the title compound (115 mg, 0.219 tmol) as a red-orange powder.
Ή— NMR (200MHz, CDC13) : δ 1.61 (3Η, brs), 2.24 (3H, brs), 2.43 (3H, s), 7.31 (2H, d, J= 8.2 Hz), 7.65 (2H, d, J= 8.4 Hz), 7.67 (2H, d, J= 8.8 Hz), 7.88 (2H, d, J= 8.2 Hz), 9.66 (1H, brs). Ή- NMR (200MHz, CDC1 3) : δ 1.61 (3Η, brs), 2.24 (3H, brs), 2.43 (3H, s), 7.31 (2H, d, J = 8.2 Hz), 7.65 (2H, d, J = 8.4 Hz), 7.67 (2H, d, J = 8.8 Hz), 7.88 (2H, d, J = 8.2 Hz), 9.66 (1H, brs).
元素分析値: C22H18N303S3F3として計算値: C, 50.27; H, 3.45; N, 7.99; S, 18.30; F, 10.84 (%) 、 実測値: C, 50.24; H, 3.15; N, 8.12; S, 18.39; F, 10.89 (%) . 即. 197.0-199.0°C Elemental analysis: C 22 H 18 N 3 0 3 Calculated S 3 F 3: C, 50.27 ; H, 3.45; N, 7.99; S, 18.30; F, 10.84 (%), Found: C, 50.24; H, 3.15; N, 8.12; S, 18.39; F, 10.89 (%). Immediate. 197.0-199.0 ° C
実施例 4-171 Example 4-171
N - {8- [(4-フルオロフェニル)スルファニル ]-5, 6-ジメチル- 3-ォキソ -2, 3-ジヒド ロ- 1H-イミダゾ [5, i-c] [1,4]チアジン -1 -ィリデン } -4-メチルべンゼンスルホン アミド N- {8-[(4-Fluorophenyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, ic] [1,4] thiazine-1-ylidene } -4-Methylbenzenesulfonamide
実施例 3- 4で得た化合物 (219 mg, 0.574腿 ol)、 4-フルォロベンゼンチオール (115 mg, 0.861 mmol) およびトリフエニルホスフィン(621 mg, 2.30膽01)の!1? (4.5 ml)の懸濁液に室温で攪拌しながら DEAD (0.381 ml, 2.30 mmol)を滴下後、 反応液を室温で 5日間、 6(TCで 2日間攪拌した。 反応液を酢酸ェチルで希釈し、 2%炭酸水素ナトリウム水溶液、 0, 1N塩酸、水および飽和食塩水で洗浄後、硫酸ナ トリウムで乾燥し、濃縮した。残渣をシリカゲル(10g)のクロマトグラフィー(酢 酸ェチルーへキサン、 1:9-3:7) に付し、 酢酸ェチルーへキサン(25:75- 30:70)の 赤色画分をメタノールから結晶化して標題化合物 (82 mg, 0.172删 ol)を赤紫色 粉末として得た。 Compound obtained in Example 3-4 (219 mg, 0.574 tmol), 4-fluorobenzenethiol (115 mg, 0.861 mmol) and triphenylphosphine (621 mg, 2.30 brutally 01)! 1? DEAD (0.381 ml, 2.30 mmol) was added dropwise to the suspension (4.5 ml) with stirring at room temperature, and the reaction solution was stirred at room temperature for 5 days and 6 (TC for 2 days. The reaction solution was diluted with ethyl acetate. The residue was washed with 2% aqueous sodium hydrogen carbonate solution, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate and concentrated, and the residue was chromatographed on silica gel (10 g) (ethyl acetate hexane, 1 g). : 9-3: 7), and the red fraction of ethyl acetate-hexane (25: 75-30: 70) was crystallized from methanol to give the title compound (82 mg, 0.172 mol) as a red-purple powder. Was.
'H-NMR (200MHz, CDC13) : δ 1.60 (3Η, brq, J= 1.0 Hz), 2.24 (3H, brq, J= 1.0 Hz), 2.43 (3H, s), 7.09 (2H, tt, J= 8.6, 2.2 Hz), 7.31 (2H, d, J= 8.2 Hz),'H-NMR (200MHz, CDC1 3): δ 1.60 (3Η, brq, J = 1.0 Hz), 2.24 (3H, brq, J = 1.0 Hz), 2.43 (3H, s), 7.09 (2H, tt, J = 8.6, 2.2 Hz), 7.31 (2H, d, J = 8.2 Hz),
7.54 (2H, ddd, J= 8.6, 5.5, 2.2 Hz), 7.89 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). 元素分析値: C2IH18N303S3Fとして計算値: C, 53.03; H, 3.81; N, 8.84; S, 20.23;7.54 (2H, ddd, J = 8.6, 5.5, 2.2 Hz), 7.89 (2H, d, J = 8.0 Hz), 9.64 (1H, brs). Elemental analysis: C 2I H 18 N 3 0 3 S 3 F Calculated as: C, 53.03; H, 3.81; N, 8.84; S, 20.23;
F, 3.99 (%), 実測値: C, 52.98; H, 3.77; N, 9.04; S, 20.55; F, 3.80 (%) . mp. 195.0-196. Ot: F, 3.99 (%), Found: C, 52.98; H, 3.77; N, 9.04; S, 20.55; F, 3.80 (%). Mp. 195.0-196. Ot:
実施例 4-172 Example 4-172
N- { 8- [ (4-ブロモフエニル)スルファニル ] -5 , 6 -ジメチル -3-ォキソ -2 , 3-ジヒドロ - 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン }-4-メチルベンゼンスルホンァ ミ H N- {8-[(4-bromophenyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene} -4-methylbenzenesulfonami H
実施例 3- 4で得た化合物 (208 mg, 0.545 mmol)、4-ブロモベンゼンチオール(159 mg, 0.818腿 ol) およぴトリフエニルホスフィン(590 mg, 2.18 mmol)の THF (4.5 ml)の懸濁液に室温で攪拌しながら DEAD (0.361 ml, 2.18 mmol)を滴下後、 反応 液を室温で 6 日間、 60°Cで 20時間攪拌した。 反応液を酢酸ェチルで希釈し、 2% 炭酸水素ナトリウム水溶液、 (UN塩酸、水および飽和食塩水で洗浄後、硫酸ナト リウムで乾燥し、 濃縮した。 残渣をシリカゲル(10 g)のクロマトグラフィー (酢 酸ェチルーへキサン、' 1:9-3:7) に付し、 酢酸ェチルーへキサン(25 :75)の赤色画 分をメタノールから結晶ィ匕して標題化合物(59 mg, 0.110醒 01 )を赤紫色粉末とし て得た。 The compound (208 mg, 0.545 mmol) obtained in Example 3-4, 4-bromobenzenethiol (159 mg, 0.818 mol) and triphenylphosphine (590 mg, 2.18 mmol) in THF (4.5 ml) were added. After DEAD (0.361 ml, 2.18 mmol) was added dropwise to the suspension while stirring at room temperature, the reaction solution was stirred at room temperature for 6 days and at 60 ° C for 20 hours. The reaction mixture was diluted with ethyl acetate, washed with 2% aqueous sodium hydrogen carbonate solution, (UN hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was chromatographed on silica gel (10 g). Ethyl acetate-hexane ('1: 9-3: 7), and the red fraction of ethyl acetate-hexane (25:75) was crystallized from methanol to give the title compound (59 mg, 0.110 mg 01). Was obtained as a red-purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.62 (3Η, brs), 2.24 (3H, brq, J= 1.0 Hz), 2.43 (3H, s), 7.31 (2H, d, J= 8.0 Hz), 7.41 (2H, dt, J= 8.4, 1.8 Hz), 7.52 (2H, dt, J= 8.4, 1.8 Hz), 7.88 (2H, d, J= 8.0 Hz), 9.65 (1H, brs). ' 元素分析値: C21H18N303S3Brとして計算値: C, 47.01; H, 3.38; N, 7.83; S, 17.93 Ή-NMR (200MHz, CDC1 3 ): δ 1.62 (3Η, brs), 2.24 (3H, brq, J = 1.0 Hz), 2.43 (3H, s), 7.31 (2H, d, J = 8.0 Hz), 7.41 (2H, dt, J = 8.4, 1.8 Hz), 7.52 (2H, dt, J = 8.4, 1.8 Hz), 7.88 (2H, d, J = 8.0 Hz), 9.65 (1H, brs). 'Elemental analysis: C 21 H 18 N 3 0 3 S 3 Br Calculated: C, 47.01; H , 3.38; N, 7.83; S, 17.93
(%)、 実測値: C, 47.14; H, 3.14; N, 8.01; S, 17.76 ( ) . (%), Found: C, 47.14; H, 3.14; N, 8.01; S, 17.76 ().
mp. 193.5-195.0°C mp. 193.5-195.0 ° C
実施例 4-173 Example 4-173
N - {8 - [(4- tert-ブチルフエニル)スルファニル ]-5, 6-ジメチル -3-ォキソ -2, 3 -ジ ヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン }-4-メチルベンゼンスル ホンアミド N- {8-[(4-tert-butylphenyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1 -Ylidene} -4-methylbenzenesulfonamide
(方法 1) 実施例 3- で得た化合物 (203 mg,. 0.532漏 1)、 4- tert-ブチルベン ゼンチオール(135 mg, 0.798画 ol) およびトリフエニルホスフィン(576 mg, 2.13 腿 ol)の THF (4.5 ml)の懸濁液に室温で攪拌しながら DEAD (0.353 ml, 2.13 mmol) を滴下後、反応液を室温で 5日間、 60°Cで 28時間攪拌した。反応液を酢酸ェチル で希釈し、 2%炭酸水素ナトリウム水溶液、 0.1N塩酸、水および飽和食塩水で洗浄 後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣をシリカゲル (10 g)のクロマトグ ラフィー (酢酸ェチルーへキサン、 10:90-25:75) に付し、 酢酸ェチルーへキサン (15:85-20:80)の赤橙色画分をメタノールから結晶化して標題化合物 (17 mg, 0.033 mmol)を赤紫色粉末として得た。 (Method 1) THF of the compound obtained in Example 3- (203 mg, 0.532 leak 1), 4-tert-butyl benzene thiol (135 mg, 0.798 ol) and triphenylphosphine (576 mg, 2.13 ol) DEAD (0.353 ml, 2.13 mmol) was added dropwise to the suspension (4.5 ml) while stirring at room temperature, and the reaction solution was stirred at room temperature for 5 days and at 60 ° C for 28 hours. The reaction solution was diluted with ethyl acetate, washed with a 2% aqueous sodium hydrogen carbonate solution, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was chromatographed on silica gel (10 g) (ethyl acetate-hexane, 10: 90-25: 75), and the red-orange fraction of ethyl acetate-hexane (15: 85-20: 80) was crystallized from methanol. To give the title compound (17 mg, 0.033 mmol) as a purplish red powder.
'H-NMR (200MHz, CDC13) : δ 1.34 (9Η, s), 1.61 (3H, q, J= 0.8 Hz), 2.24 (3H, brq, J= 0.8 Hz), 2.42 (3H, s), 7.30 (2H, d, J= 8.4 Hz), 7.40 (2H, d, J= 8.8 Hz), 7.45 (2H, d, J= 8.8 Hz), 7.89 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). 元素分析値: C25H27N303S3 · 0.3 0として計算値: C, 57.84; H, 5.36; N, 8.09; S, 18.53 (%) 、 実測値: C, 57.86; H, 5.30; N, 8.01; S, 18.17 ( ) . 'H-NMR (200MHz, CDC1 3): δ 1.34 (9Η, s), 1.61 (3H, q, J = 0.8 Hz), 2.24 (3H, brq, J = 0.8 Hz), 2.42 (3H, s), 7.30 (2H, d, J = 8.4 Hz), 7.40 (2H, d, J = 8.8 Hz), 7.45 (2H, d, J = 8.8 Hz), 7.89 (2H, d, J = 8.4 Hz), 9.63 ( 1H, brs). Elemental analysis: Calculated as C 25 H 27 N 3 0 3 S 3 · 0.30: C, 57.84; H, 5.36; N, 8.09; S, 18.53 (%), found: C, 57.86; H, 5.30; N, 8.01; S, 18.17 ().
mp. 194.0-197.0°C mp. 194.0-197.0 ° C
(方法 2) 実施例 3-4で得た化合物 (229 mg, 0.600 mmol), 4 - tert-ブチルベン ゼンチオール(152 mg, 0.900讓 ol) およびト リプチルホスフィン(131 mg, 0.600 mmol)の THF (5 ml)の懸濁液に室温で攪拌しながら 1, 1 ' -(ァゾジカルポニル)ジ ピぺリジン (以下 ADDPと略すことがある、 170 mg, 0.660讓 ol)を加え、 反応液 を室温で 19時間、 60°Cで 4日間攪拌した。 この間反応液にトリブチルホスフィン (131 mg, 0.600腿 0 および ADDP (170 mg, 0.660腿 ol)をそれぞれ 2回加えた。 反応液を同様に後処理し、 化合物 (40 mg, 0.078 腿 ol)を赤紫色粉末として得た。 実施例 4-174 (Method 2) THF (131 mg, 0.600 mmol) of the compound obtained in Example 3-4 (229 mg, 0.600 mmol), 4-tert-butylbenzenthiol (152 mg, 0.900 mL) and triptylphosphine (131 mg, 0.600 mmol) To this suspension, 1,1 '-(azodicarbonyl) dipiperidine (hereinafter abbreviated as ADDP, 170 mg, 0.660 mL) was added with stirring at room temperature. The mixture was stirred at 60 ° C for 4 days. During this time, tributylphosphine (131 mg, 0.600 thigh 0) and ADDP (170 mg, 0.660 thigh ol) were added twice each to the reaction solution. The reaction solution was post-treated in the same manner to obtain a compound (40 mg, 0.078 tmol) as a red-purple powder. Example 4-174
酢酸ェチル-ジェチルエーテルから再結晶し赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-getyl ether to obtain red crystals.
融点 184-186T . Melting point: 184-186T.
赤外吸収スペクトル(IR) (KBr中) V : 3243, 2969, 2922, 1746, 1721, 1661, 1615, 1559 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3243, 2969, 2922, 1746, 1721, 1661, 1615, 1559 cm " 1 .
iH-NMR (CDC13) : 6 1.69 (3H, s), 1.76 (3H, s), 1.78 (3H, d, J=0.8 Hz), 2.26 (3H, d, J-1.2 Hz), 2.42 (3H, s), 3.61 (3H, d, J=7.6 Hz), 5.23 (1H, t, J=7.6 Hz), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.61 (1H, bs) . iH-NMR (CDC1 3): 6 1.69 (3H, s), 1.76 (3H, s), 1.78 (3H, d, J = 0.8 Hz), 2.26 (3H, d, J-1.2 Hz), 2.42 (3H , s), 3.61 (3H, d, J = 7.6 Hz), 5.23 (1H, t, J = 7.6 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz) ), 9.61 (1H, bs).
実施例 4-175 . Example 4-175.
N-{8 - [(4 -ク口口フエニル)スルファニル] - 5, 6-ジメチル- 3-ォキソ -2, 3 -ジヒド口 - 1H-イミダゾ [5,卜 c] [1, 4]チアジン-卜イリデン }- 4-メチルベンゼンスルホンァ $ド N- {8-[(4-Couguchiphenyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrido-1H-imidazo [5, tric] [1,4] thiazine- Triylidene}-4-methylbenzenesulfone
実施例 3-4で得た化合物 (198 mg, 0.519醒 ol)、4-クロ口ベンゼンチオール(113 mg, 0.779讓 ol) およびトリフエニルホスフィン(561 mg, 2.08腿01)の1^ (3.3 ml)の懸濁液に室温で攪拌しながら DEAD (0.172 ml, 1.04匪 ol)を滴下後、 反応 液を室温で 24時間攪拌した。 反応液に DEAD (0.172 ml, 1.04腿 ol)を追加し、 室温で 3日間攪拌した。 この反応液を、 実施例 3- 4で得た化合物 (200 mg, 0.524 mmol)から同様にして得た反応液とあわせて酢酸ェチルで希釈し、 2%炭酸水素ナト リウム水溶液、 0.1N塩酸、水および飽和食塩水で洗浄後、硫酸ナトリウムで乾燥 し、 濃縮した。 残渣をシリカゲル(10 g)のクロマトグラフィー (酢酸ェチルーへ キサン、 1:9-3:7) に付し、 酢酸ェチルーへキサン(25 :75- 30 :70)の赤橙色画分を メタノールから結晶化して標題化合物 (69 mg, 0.140腿 ol)を紫色粉末として得 た。 1 ^ (3.3 ml) of the compound obtained in Example 3-4 (198 mg, 0.519 violated ol), 4-chloro benzenethiol (113 mg, 0.779 benzene) and triphenylphosphine (561 mg, 2.08 thigh 01) ) Was added dropwise to the suspension with stirring at room temperature, and the reaction solution was stirred at room temperature for 24 hours. DEAD (0.172 ml, 1.04 tmol) was added to the reaction solution, and the mixture was stirred at room temperature for 3 days. This reaction mixture was diluted with ethyl acetate together with the reaction mixture similarly obtained from the compound (200 mg, 0.524 mmol) obtained in Example 3-4, and 2% aqueous sodium hydrogen carbonate, 0.1N hydrochloric acid, After washing with water and saturated saline, it was dried over sodium sulfate and concentrated. The residue was subjected to silica gel (10 g) chromatography (ethyl acetate-hexane, 1: 9-3: 7), and the red-orange fraction of ethyl acetate-hexane (25: 75-30: 70) was crystallized from methanol. To give the title compound (69 mg, 0.140 t ol) as a purple powder.
^-NMR (200MHz, CDC13) : <5 1.61 (3H, q, J= 0.8 Hz), 2.23 (3H, q, J- 0.6 Hz), 2.43 (3H, s), 7.31 (2H, d, J= 8.2 Hz), 7.37 (2H, dt, J= 8.8, 2.2 Hz), 7.47 (2H, dt, J= 8.8, 2.2 Hz), 7.88 (2H, d, J= 8.6 Hz), 9.65 (1H, brs). ^ -NMR (200MHz, CDC1 3) : <5 1.61 (3H, q, J = 0.8 Hz), 2.23 (3H, q, J- 0.6 Hz), 2.43 (3H, s), 7.31 (2H, d, J = 8.2 Hz), 7.37 (2H, dt, J = 8.8, 2.2 Hz), 7.47 (2H, dt, J = 8.8, 2.2 Hz), 7.88 (2H, d, J = 8.6 Hz), 9.65 (1H, brs ).
元素分析値: C21H18N303S3C1として計算値: C, 51.26; H, 3.69; N, 8.54; S, 19.55; C1, 7.21 (%), 実測値: C, 51.27; H, 3.54; N, 8.77; S, 19.52; C1, 7.05 (%) · mp. 201.5-204. Elemental analysis: Calculated for C 21 H 18 N 3 0 3 S 3 C1: C, 51.26; H, 3.69; N, 8.54; S, 19.55; C1, 7.21 (%), found: C, 51.27; H , 3.54; N, 8.77; S, 19.52; C1, 7.05 (%) mp. 201.5-204.
実施例 4- 176 Example 4-176
Ν-{8-[(1, 1 -ジメチルブチル)スルファニル] -5, 6-ジメチル -3-ォキソ -2, 3-ジヒド ロ- 1H -イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホン アミド Ν- {8-[(1,1-Dimethylbutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine- 1-ylidene}-4-methylbenzenesulfonamide
実施例 3- 4で得た化合物 (163 mg, 0.427 mmol)と 2-メチル -卜ペンテン (0.54 ml, 4.27 mmol)のジクロロメタン (3.3 ml) 溶液を 3フッ化ホウ素ェ一テル錯体 (0.11 ml, 0.854 mmol) とともに室温で 23時間攪拌した。 反応液を濃縮後、 酢酸 ェチルで希釈し、 希塩酸、 炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗 浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣をシリカゲル (7 g)のクロマトグ ラフィ一 (酢酸ェチルーへキサン、 1:9-3:7) に付した。 酢酸ェチルーへキサン (2:8- 3:7)の画分を濃縮乾固後メタノールから結晶化して標題化合物 (48 mg, 0.103删 ol)を濃赤色粉末として得た。 A solution of the compound obtained in Example 3-4 (163 mg, 0.427 mmol) and 2-methyl-topentene (0.54 ml, 4.27 mmol) in dichloromethane (3.3 ml) was mixed with a boron trifluoride ether complex (0.11 ml, 0.854 mmol) at room temperature for 23 hours. The reaction mixture was concentrated, diluted with ethyl acetate, washed with dilute hydrochloric acid, aqueous sodium hydrogen carbonate, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was subjected to chromatography on silica gel (7 g) (ethyl acetate-hexane, 1: 9-3: 7). The fraction of ethyl acetate-hexane (2: 8-3: 7) was concentrated to dryness and crystallized from methanol to give the title compound (48 mg, 0.103 mol) as a dark red powder.
'H-NMR (200MHz, CDC13) : δ 0.88 (3Η, t, J= 7.2 Hz), 1.36 (2H, m), 1.41 (6H, s), 1.60 (2H, m), 1.75 (3H, q, J= 1.2 Hz), 2.22 (3H, q, J= 1.0 Hz), 2.42 (3H, s), 7.29 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.69 (1H, brs). 元素分析値: C21H27N303S3として計算値: C, 54.17; H, 5.84; N, 9.02 (%) 、 実測 値: C, 54.06; H, 5.73; N, 9.09 (%) . 'H-NMR (200MHz, CDC1 3): δ 0.88 (3Η, t, J = 7.2 Hz), 1.36 (2H, m), 1.41 (6H, s), 1.60 (2H, m), 1.75 (3H, q , J = 1.2 Hz), 2.22 (3H, q, J = 1.0 Hz), 2.42 (3H, s), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.69 (1H, brs). Elemental analysis: Calculated for C 21 H 27 N 3 0 3 S 3 : C, 54.17; H, 5.84; N, 9.02 (%), found: C, 54.06; H, 5.73 ; N, 9.09 (%).
即. 169.0- 170.5 Immediately. 169.0- 170.5
実施例 4-177 Example 4-177
N- {5, 6-ジメチル- 8- [ (3-メチル- 2-ォキソプチル)スルファニル] -3-ォキソ- 2, 3- ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン } -4-メチルベンゼンス ルホンアミド N- {5,6-dimethyl-8-[(3-methyl-2-oxobutyl) sulfanyl] -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] Thiazine-triylidene} -4-methylbenzenesulfonamide
3 -メチル- 2-ブ夕ノン (8.50 g, 95.7 mmol)の無水メタノ一ル (60 ml) 溶液に - 7°Cで臭素(5.5 ml, 0.106 mol) を 30秒かけて加え、 混合液を 0°Cで 30分間さ らに 10 で 1時間攪拌した。 反応液に水 (30ml) を加え、 さらに室温で 16時間 攪拌した。 反応液に水 (90 ml) を加えてジェチルエーテル(80 ml)およびジェチ ルエーテル一へキサン(1:1, 80 nil)で 2回抽出した。 有機層を飽和炭酸水素ナト リウム水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮し た。 残渣油状物を減圧下 (14腿 Hg)蒸留し、 1-ブロモ -3-メチル -2-ブ夕ノン (純度 90¾, 12.4 g, 67.6 mmol)を無色油状物として得た。 To a solution of 3-methyl-2-butanone (8.50 g, 95.7 mmol) in anhydrous methanol (60 ml) was added bromine (5.5 ml, 0.106 mol) at -7 ° C over 30 seconds, and the mixture was mixed. The mixture was stirred at 0 ° C for 30 minutes and further at 10 for 1 hour. Water (30 ml) was added to the reaction solution, and the mixture was further stirred at room temperature for 16 hours. Water (90 ml) was added to the reaction solution, and the mixture was extracted twice with dimethyl ether (80 ml) and dimethyl ether monohexane (1: 1, 80 nil). The organic layer was washed with a saturated aqueous solution of sodium hydrogen carbonate, water and saturated saline, dried over sodium sulfate, and concentrated. Was. The residual oil was distilled under reduced pressure (14 g Hg) to give 1-bromo-3-methyl-2-butanone (purity 90 °, 12.4 g, 67.6 mmol) as a colorless oil.
!H-NMR (200MHz, CDC13): (51.17 (6H, d, J= 7.0 Hz), 2.99 (1H, septet, J= 7.0 Hz), 3.99 (2H, s). ! H-NMR (200MHz, CDC1 3): (51.17 (6H, d, J = 7.0 Hz), 2.99 (1H, septet, J = 7.0 Hz), 3.99 (2H, s).
沸点. 58-63°C (14 mmHg) Boiling point.58-63 ° C (14 mmHg)
実施例 3- 4で得た化合物 (172 mg, 0.451 mmol)と卜ブロモ -3-メチル -2-ブ 夕ノン (91 mg, 0.496 匪 ol)の THF (3.4 ml)溶液に室温でト リェチルァミン (0.125 ml, 0.902腿 ol) を加え、 反応液を 2.5時間攪拌した。 反応液に 1 N塩 酸 (0.91 ml)を加えて 10分攪拌し、 メタノール一水 (1:1, 5 ml)で希釈した。 析 出物をメタノール一水(1:1)およびメタノールで洗浄後、 乾燥して標題化合物 (192 mg, 0.412 fflnol)を赤褐色粉末として得た。 To a solution of the compound obtained in Example 3-4 (172 mg, 0.451 mmol) and tribromo-3-methyl-2-butanone (91 mg, 0.496 ol) in THF (3.4 ml) at room temperature was added triethylamine (RT). 0.125 ml, 0.902 tmol) was added, and the reaction solution was stirred for 2.5 hours. 1N Hydrochloric acid (0.91 ml) was added to the reaction solution, stirred for 10 minutes, and diluted with methanol / water (1: 1, 5 ml). The precipitate was washed with methanol-water (1: 1) and methanol, and dried to give the title compound (192 mg, 0.412 fflnol) as a red-brown powder.
Ή— NMR (200MHz, CDC13) : δ 1.15 (6Η, d, J= 6.6 Hz), 1.75 (3H, q, J= 1.2 Hz), 2.24 (3H, q, J= 0.8 Hz), 2.42 (3H, s), 2.82 (1H, septet, J= 6.8 Hz), 3.90 (2H, s), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). 元素分析値: C19H17N303S3として計算値: C, 51.59; H, 4.98; N, 9.02; S, 20.66 (%) 、 実測値: C, 51.40; H, 4.94; N, 9.13; S, 20.61 ( ) . Ή- NMR (200MHz, CDC1 3) : δ 1.15 (6Η, d, J = 6.6 Hz), 1.75 (3H, q, J = 1.2 Hz), 2.24 (3H, q, J = 0.8 Hz), 2.42 (3H , s), 2.82 (1H, septet, J = 6.8 Hz), 3.90 (2H, s), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.64 (1H , brs). Elemental analysis: Calculated for C 19 H 17 N 3 0 3 S 3 : C, 51.59; H, 4.98; N, 9.02; S, 20.66 (%), found: C, 51.40; H, 4.94; N, 9.13; S, 20.61 ().
融点. 205.0-206.5°C Melting point.205.0-206.5 ° C
実施例 4-178 Example 4-178
酢酸ェチル -ジィソプロピルエーテルから再結晶し赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-disopropyl ether to obtain red crystals.
融点: 186- 188°C. Melting point: 186-188 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3281, 2976, 2928, 1750, 1717, 1661, 1615, 1563, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr): 3281, 2976, 2928, 1750, 1717, 1661, 1615, 1563, 1497 cm " 1 .
Ή-NMR (CDC13) : δ 1.61 (6Η, s), 1.80 (3H, d, J=1.0 Hz), 2.01-2.13 (2H, m), 2.27 (3H, d, J=1.0 Hz), 2.42 (3H, s), 3.07-3.19 (2H, m), 7.30 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.2 Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3): δ 1.61 (6Η, s), 1.80 (3H, d, J = 1.0 Hz), 2.01-2.13 (2H, m), 2.27 (3H, d, J = 1.0 Hz), 2.42 (3H, s), 3.07-3.19 (2H, m), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.2 Hz), 9.62 (1H, bs).
実施例 4-179 Example 4-179
Ή一 NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 7.2 Hz), 1.33 (2H, m), 1.42 (2H, m), 2.05 (2H, brdd, J= 7.2, 6.8 Hz), 2.42 (3H, s), 4.16 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.22 (1H, q, J= 7.6 Hz), 7.26 (1H, brd, J= 7.6 Hz), 7.28 (2H, brd, J= 8.3 Hz), 7.45 (1H, dt, J= 7.7, 1.6 Hz), 7.47 (1H, brs), 7.81 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). Ή one NMR (300MHz, CDC1 3): δ 0.91 (3H, t, J = 7.2 Hz), 1.33 (2H, m), 1.42 (2H, m), 2.05 (2H, brdd, J = 7.2, 6.8 Hz) , 2.42 (3H, s), 4.16 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.22 (1H, q, J = 7.6 Hz), 7.26 (1H, brd, J = 7.6 Hz) , 7.28 (2H, brd, J = 8.3 Hz), 7.45 (1H, dt, J = 7.7, 1.6 Hz), 7.47 (1H, brs), 7.81 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C24H24N303S3Br'0.6H20として計算値: C, 48.91; H, 4.31; N, 7.13(%)、 実測値: C, 48.61; H, 4.08; 7.42 (%) . Elemental analysis: C 24 H 24 N 3 0 3 S 3 Br'0.6H 2 0 Calculated: C, 48.91; H, 4.31 ; N, 7.13 (%), Found: C, 48.61; H, 4.08 ; 7.42 (%).
実施例 4-180 Example 4-180
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 7.2 Hz), 1.32 (2H, brquint, J= 7.4 Hz), 1.42 (2H, m), 2.05 (2H, brdd, J= 7.1, 6.7 Hz), 2.41 (3H, s), 3.93 (3H, s), 4.25 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.42 (1H, t, J= 7.7 Hz), 7.53 (1H, dt, J= 7.7, 1.1 Hz), 7.80 (2H, d, J= 8.3 Hz), 7.99 (1H, brdt, J= 7.7, 1.1 Hz), 8.02 (1H, brt, J= 1 Hz), 9.74 (1H, brs). 元素分析値: C26H27N305S3として計算値: C, 55.99; H, 4.88; N, 7.53; S, 17.25 Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 7.2 Hz), 1.32 (2H, brquint, J = 7.4 Hz), 1.42 (2H, m), 2.05 (2H, brdd, J = 7.1, 6.7 Hz), 2.41 (3H, s), 3.93 (3H, s), 4.25 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.26 (2H, d, J = 8.3 Hz) , 7.42 (1H, t, J = 7.7 Hz), 7.53 (1H, dt, J = 7.7, 1.1 Hz), 7.80 (2H, d, J = 8.3 Hz), 7.99 (1H, brdt, J = 7.7, 1.1 Hz), 8.02 (1H, brt, J = 1 Hz), 9.74 (1H, brs). Elemental analysis: Calculated as C 26 H 27 N 3 0 5 S 3 : C, 55.99; H, 4.88; N, 7.53; S, 17.25
(%), 実測値: C, 55.83; H, 4.66; N, 7.57; S, 17.26 (%) . (%), Found: C, 55.83; H, 4.66; N, 7.57; S, 17.26 (%).
実施例 4-181 Example 4-181
'H-NMR (300MHz, CDC13) : δ 0.92 (3H, t, J= 7.2 Hz), 1.36 (2H, brquint, J= 7.4 Hz), 1.46 (2H, m), 2.08 (2H, dd, J- 7.1, 6.8 Hz), 2.36 (3H, s), 2.40 (3H, s), 4.23 (2H, s), 6.49 (1H, brt, = 0.5 Hz), 7.15-7.28 (6H, m), 7.77 (2H, d, J= 8.3 Hz), 9.71 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 0.92 (3H, t, J = 7.2 Hz), 1.36 (2H, brquint, J = 7.4 Hz), 1.46 (2H, m), 2.08 (2H, dd, J -7.1, 6.8 Hz), 2.36 (3H, s), 2.40 (3H, s), 4.23 (2H, s), 6.49 (1H, brt, = 0.5 Hz), 7.15-7.28 (6H, m), 7.77 ( 2H, d, J = 8.3 Hz), 9.71 (1H, br).
元素分析値: C25H27N303S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73 (%), 実測値: C, 58.35; H, 5.31; N, 8.14; S, 18.57 (%) . Elemental analysis: C 25 H 27 N 3 0 3 S 3 Calculated: C, 58.45; H, 5.30 ; N, 8.18; S, 18.73 (%), Found: C, 58.35; H, 5.31 ; N, 8.14; S, 18.57 (%).
実施例 4-182 Example 4-182
'H-NMR (300MHz, CDC13) : δ 0.92 (3H, t, J= 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.06 (2H, brdd, J= 7.0, 6.8 Hz), 2.41 (3H, s), 4.15 (2H, s), 5.99 (2H, s), 6.48 (1H, brt, J= 0.5 Hz), 6.76 (1H, s), 6.78 (2H, brd, J= 8 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.92 (3H, t, J = 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.06 (2H, brdd, J = 7.0, 6.8 Hz ), 2.41 (3H, s), 4.15 (2H, s), 5.99 (2H, s), 6.48 (1H, brt, J = 0.5 Hz), 6.76 (1H, s), 6.78 (2H, brd, J = 8 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C25H25N305S3として計算値: C, 55.23; H, 4.63; N, 7.73; S, 17.69 (%) 、 実測値: C, 55.11; H, 4.59; N, 7.69; S, 17.79 ( ) . Elemental analysis: Calculated as C 25 H 25 N 3 0 5 S 3: C, 55.23; H, 4.63; N, 7.73; S, 17.69 (%), Found: C, 55.11; H, 4.59 ; N, 7.69; S, 17.79 ().
実施例 4-183 Example 4-183
Ή- MR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 7.2 Hz), 1.33 (2H, m), 1.43 (2H, m), 2.05 (2H, brdd, J= 7.1, 6.8 Hz), 2.41 (3H, s), 3.67 (2H, s), 4.20 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.27 (4H, m), 7.27 (2H, d, J= 8.3 Hz), 7.80 (2H, brd, J= 8.3 Hz), 9.74 (1H, brs). Ή- MR (300MHz, CDC1 3) : δ 0.91 (3Η, t, J = 7.2 Hz), 1.33 (2H, m), 1.43 (2H, m), 2.05 (2H, brdd, J = 7.1, 6.8 Hz) , 2.41 (3H, s), 3.67 (2H, s), 4.20 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.27 (4H, m), 7.27 (2H, d, J = 8.3 Hz), 7.80 (2H, brd, J = 8.3 Hz), 9.74 (1H, brs).
実施例 4-184 Example 4-184
Ή一 NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 7.2 Hz), 1.34 (2H, brquint, J= 7.3 Hz), 1.44 (2H, m), 2.05 (2H, brdd, J= 7.2, 6.9 Hz), 2.41 (3H, s), 3.63 (2H, s), 3.71 (3H, s), 4.20 (2H, s), 6.48 (1H, brs), 7.27 (6H, m), 7.80 (2H, brd, J= 8.2 Hz), 9.73 (1H, brs). Ή one NMR (300MHz, CDC1 3): δ 0.91 (3H, t, J = 7.2 Hz), 1.34 (2H, brquint, J = 7.3 Hz), 1.44 (2H, m), 2.05 (2H, brdd, J = 7.2, 6.9 Hz), 2.41 (3H, s), 3.63 (2H, s), 3.71 (3H, s), 4.20 (2H, s), 6.48 (1H, brs), 7.27 (6H, m), 7.80 ( 2H, brd, J = 8.2 Hz), 9.73 (1H, brs).
実施例 4-185 Example 4-185
¾-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J- 7.1 Hz), 1.32 (2H, m), 1.39 (2H, m), 2.03 (2H, brdd, J= 7.1, 6.6 Hz), 2.43 (3H, s), 4.22 (2H, s), 6.45 (1H, brt, J= 0.5 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.45 (2H, d, J= 8.3 Hz), 7.62 (2H, d, J- 8.3 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.60 (1H, br). ¾-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J- 7.1 Hz), 1.32 (2H, m), 1.39 (2H, m), 2.03 (2H, brdd, J = 7.1, 6.6 Hz) , 2.43 (3H, s), 4.22 (2H, s), 6.45 (1H, brt, J = 0.5 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.45 (2H, d, J = 8.3 Hz) , 7.62 (2H, d, J- 8.3 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.60 (1H, br).
元素分析値: C25H24N403S3として計算値: C, 57.23; H, 4.61; N, 10.68; S, 18.33 (%) 、 実測値: C, 56.85; H, 4.60; N, 10.59; S, 18.29 (%) . Elemental analysis: C 25 H 24 N 4 0 3 S 3 Calculated: C, 57.23; H, 4.61 ; N, 10.68; S, 18.33 (%), Found: C, 56.85; H, 4.60 ; N, 10.59; S, 18.29 (%).
実施例 4-186 Example 4-186
Ή-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 7.1 Hz), 1.32 (2H, m), 1.40 (3H, t, J= 7.1 Hz), 1.41 (2H, m), 2.04 (2H, brdd, J= 7.3, 6.6 Hz), 2.42 (3H, s), 4.24 (2H, s), 4.39 (2H, q, J= 7.1 Hz), 6.46 (1H, brs), 7.27 (2H, d, J= 8.2 Hz), 7.39 (2H, d, J= 7.1 Hz), 7.81 (2H, d, J= 8.2 Hz), 8.00 (2H, d, J= 8.2 Hz), 9.73 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 7.1 Hz), 1.32 (2H, m), 1.40 (3H, t, J = 7.1 Hz), 1.41 (2H, m), 2.04 (2H, brdd, J = 7.3, 6.6 Hz), 2.42 (3H, s), 4.24 (2H, s), 4.39 (2H, q, J = 7.1 Hz), 6.46 (1H, brs), 7.27 (2H, d, J = 8.2 Hz), 7.39 (2H, d, J = 7.1 Hz), 7.81 (2H, d, J = 8.2 Hz), 8.00 (2H, d, J = 8.2 Hz), 9.73 (1H, brs) .
元素分析値: C27H2gN305S3として計算値: C, 56.72; H, 5.11; N, 7.35; S, 16.83Elemental analysis: C 27 H 2g N 3 0 5 S 3 Calculated: C, 56.72; H, 5.11 ; N, 7.35; S, 16.83
(%), 実測値: C, 56.54; H, 4.98; N, 7.22; S, 16.95 (%) . (%), Found: C, 56.54; H, 4.98; N, 7.22; S, 16.95 (%).
実施例 4-187 Example 4-187
Ή一蘭 R (300MHz, DMS0-d6) : δ 0.85 (3H, t, J- 7.1 Hz), 1.26 (2H, m), 1.32 (2H, m), 2.11 (2H, brdd, J= 7.0, 6.6 Hz), 2.37 (3H, s), 4.38 (2H, s), 6.60 (1H, brs), 7.33 (2H, d, J= 8.2 Hz), 7.48 (2H, d, J= 7.1 Hz), 7.80 (2H, d, J= 8.2 Hz), 7.90 (2H, d, J= 8.2 Hz), 11.80 (1H, br), 12.95 (1H, brs). ΉIchiran R (300MHz, DMS0-d 6 ): δ 0.85 (3H, t, J- 7.1 Hz), 1.26 (2H, m), 1.32 (2H, m), 2.11 (2H, brdd, J = 7.0, 6.6 Hz), 2.37 (3H, s), 4.38 (2H, s), 6.60 (1H, brs), 7.33 (2H, d, J = 8.2 Hz), 7.48 (2H, d, J = 7.1 Hz), 7.80 (2H, d, J = 8.2 Hz), 7.90 (2H, d, J = 8.2 Hz), 11.80 (1H, br), 12.95 (1H, brs).
元素分析値: C25H25N305S3として計算値: C, 55.23; H, 4.63; N, 7.73; S, 17.69 実測値: C, 55.12; H, 4.65; N, 7.68; S, 17.58 (%) . 実施例 4-188 Elemental analysis: C 25 H 25 N 3 0 5 S 3 Calculated: C, 55.23; H, 4.63 ; N, 7.73; S, 17.69 Found: C, 55.12; H, 4.65 ; N, 7.68; S, 17.58 (%). Example 4-188
Ή-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.06 (2H, brt, J= 7.0 Hz), 2.41 (3H, s), 3.82 (3H, s), 4.18 (2H, s), 6.48 (1H, brt, J= 1 Hz), 6.86 (2H, dt, J= 8.6, 1.8 Hz), 7.23 (2H, d, J= 8.6 Hz), 7.25 (2H, d, J= 7.3 Hz), 7.80 (2H, d, J= 8.2 Hz), 9.72 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.06 (2H, brt, J = 7.0 Hz), 2.41 (3H, s), 3.82 (3H, s), 4.18 (2H, s), 6.48 (1H, brt, J = 1 Hz), 6.86 (2H, dt, J = 8.6, 1.8 Hz), 7.23 (2H, d, J = 8.6 Hz), 7.25 (2H, d, J = 7.3 Hz), 7.80 (2H, d, J = 8.2 Hz), 9.72 (1H, brs).
元素分析値: C25H27N304S3として計算値: C, 56.69; H, 5.14; , 7.92; S, 18.16Elemental analysis: Calculated as C 25 H 27 N 3 0 4 S 3: C, 56.69; H, 5.14;, 7.92; S, 18.16
(%) .実測値: C, 56.53; H, 5.14; N, 7.93; S, 18.25 (%) . Found: C, 56.53; H, 5.14; N, 7.93; S, 18.25 (%).
実施例 4-189 Example 4-189
Ή-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 7.2 Hz), 1.33 (2H, m), 1.43 (2H, m), 2.05 (2H, brdd, J= 7.1, 6.8 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.10 (1H, d, J= 16.5 Hz), 7.11 (1H, d, J= 16.5 Hz), 7.26 (2H, brd, J= 8.3 Hz), 7.28 (1H, m), 7.31 (2H, d, J= 8.1 Hz), 7.37 (2H, brt, J= 7.7 Hz), 7.47 (2H, d, J- 8.2 Hz), 7.52 (2H, brd, J= 7.3 Hz), 7.81 (2H, d, J= 8.2 Hz), 9.72 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 7.2 Hz), 1.33 (2H, m), 1.43 (2H, m), 2.05 (2H, brdd, J = 7.1, 6.8 Hz) , 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.10 (1H, d, J = 16.5 Hz), 7.11 (1H, d, J = 16.5 Hz) , 7.26 (2H, brd, J = 8.3 Hz), 7.28 (1H, m), 7.31 (2H, d, J = 8.1 Hz), 7.37 (2H, brt, J = 7.7 Hz), 7.47 (2H, d, J- 8.2 Hz), 7.52 (2H, brd, J = 7.3 Hz), 7.81 (2H, d, J = 8.2 Hz), 9.72 (1H, br).
元素分析値: C32H3lN303S3として計算値: C, 63.87; H, 5.19; N, 6.98; S, 15.98 (%)、 実測値: C, 63.77; H, 5.07; N, 6.83; S, 15.77 (%) . Elemental analysis: C 32 H 3l N 3 0 3 S 3 Calculated: C, 63.87; H, 5.19 ; N, 6.98; S, 15.98 (%), Found: C, 63.77; H, 5.07 ; N, 6.83; S, 15.77 (%).
実施例 4-190 Example 4-190
Ή-NMR (300MHz, CDC13) : δ 0.91 (3Η, ί, J= 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.05 (2H, brt, J= 7 Hz), 2.40 (3H, s), 4.18 (2H, s), 5.07 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 6.93 (2H, dt, J= 8.6, 1.7 Hz), 7.23 (2H, d, J= 8.6 Hz), 7.25 (2H, brd, J= 8.3 Hz), 7.36 (1H, m), 7.41 (2H, d, J= 7.6 Hz), 7.43 (2H, brt,. J= 7.6 Hz), 7.80 (2H, d, J- 8.3 Hz), 9.70 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3Η, ί, J = 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.05 (2H, brt, J = 7 Hz), 2.40 (3H, s), 4.18 (2H, s), 5.07 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 6.93 (2H, dt, J = 8.6, 1.7 Hz), 7.23 (2H, s) d, J = 8.6 Hz), 7.25 (2H, brd, J = 8.3 Hz), 7.36 (1H, m), 7.41 (2H, d, J = 7.6 Hz), 7.43 (2H, brt, .J = 7.6 Hz) ), 7.80 (2H, d, J- 8.3 Hz), 9.70 (1H, brs).
元素分析値: C31H31N304S3として計算値: C, 61.46; H, 5.16; , 6.94; S, 15.88 (%), 実測値: C, 61.13; H, 5.07; N, 6.96; S, 15.90 (%) . Elemental analysis: C 31 H 31 N 3 0 4 S 3 Calculated: C, 61.46; H, 5.16 ;, 6.94; S, 15.88 (%), Found: C, 61.13; H, 5.07 ; N, 6.96 ; S, 15.90 (%).
実施例 4-191 Example 4-191
Ή一 NMR (300MHz, CDC13) : δ 0.92 (3H, t, J= 7.2 Hz), 0.97 (2H, brddt, J= 12.6, 2.5, 12.1 Hz), 1.19 (1H, m), 1.22 (1H, m), 1.35 (2H, m), 1.45 (2H, m), 1.57 (1H, m), 1.69 (1H, m), 1.74 (2H, dquint, J= 12, 3.0 Hz), 1.85 (2H, brd, J= 12.6 Hz), 2.07 (2H, brt, J= 7.1 Hz), 2.42 (3H, s), 2.84 (2H, d, J= 6.8 Hz), 6.50 (1H, brt, J= 0.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.69 (1H, brs). Ή one NMR (300MHz, CDC1 3): δ 0.92 (3H, t, J = 7.2 Hz), 0.97 (2H, brddt, J = 12.6, 2.5, 12.1 Hz), 1.19 (1H, m), 1.22 (1H, m), 1.35 (2H, m), 1.45 (2H, m), 1.57 (1H, m), 1.69 (1H, m), 1.74 (2H, dquint, J = 12, 3.0 Hz), 1.85 (2H, brd , J = 12.6 Hz), 2.07 (2H, brt, J = 7.1 Hz), 2.42 (3H, s), 2.84 (2H, d, J = 6.8 Hz), 6.50 (1H, brt, J = 0.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.69 (1H, brs).
元素分析値: C24H31N303S3として計算値: C, 57.00; H, 6.18; N, 8.31; S, 19.02 (%) 、 実測値: C, 56.76; H, 6.16; N, 8.35; S, 19.09 ( ) . Elemental analysis: C 24 H 31 N 3 0 3 S 3 Calculated: C, 57.00; H, 6.18 ; N, 8.31; S, 19.02 (%), Found: C, 56.76; H, 6.16 ; N, 8.35; S, 19.09 ().
実施例 4-192 Example 4-192
'H-NMR (300MHz, CDC13) : δ 0.92 (3H, t, J= 7.2 Hz), 1.35 (2H, m), 1.46 (2H, m), 2.07 (2H, t, J= 7.2 Hz), 2.42 (3H, s), 2.96 (2H, dd, J= 8.4, 7.2 Hz), 3.20 (2H, dd, J- 8.4, 7.2 Hz), 6.50 (1H, s), 7.19 (2H, brd, J= 7.2 Hz), 7.27 (5H, m), 7.83 (2H, d, J= 8.2 Hz), 9.78 (1H, br) . 'H-NMR (300MHz, CDC1 3): δ 0.92 (3H, t, J = 7.2 Hz), 1.35 (2H, m), 1.46 (2H, m), 2.07 (2H, t, J = 7.2 Hz), 2.42 (3H, s), 2.96 (2H, dd, J = 8.4, 7.2 Hz), 3.20 (2H, dd, J-8.4, 7.2 Hz), 6.50 (1H, s), 7.19 (2H, brd, J = 7.2 Hz), 7.27 (5H, m), 7.83 (2H, d, J = 8.2 Hz), 9.78 (1H, br).
実施例 4-193 Example 4-193
Ή-NMR (300MHz, CDC13) : δ 0.92 (3H, ί, J= 7.0 Hz), 1.35 (2H, m), 1.42 (2H, m), 2.02 (2H, quint, J= 7.3 Hz), 2.04 (2H, bri, J= 6.8 Hz), 2.40 (3H, s), 2.72 (2H, brt, J= 7.3 Hz), 2.95 (2H, dd, J= 7.6, 7.2 Hz), 6.49 (1H, brs), 7.15 (2Η,' brd, J= 7.0 Hz), 7.24 (1H, m), 7.27 (2H, d, J= 8.2 Hz), 7.29 (2H, brt, J= 7.0 Hz), 7.84 (2H, d, J= 8.2 Hz), 9.71 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.92 (3H, ί, J = 7.0 Hz), 1.35 (2H, m), 1.42 (2H, m), 2.02 (2H, quint, J = 7.3 Hz), 2.04 (2H, bri, J = 6.8 Hz), 2.40 (3H, s), 2.72 (2H, brt, J = 7.3 Hz), 2.95 (2H, dd, J = 7.6, 7.2 Hz), 6.49 (1H, brs) , 7.15 (2Η, 'brd, J = 7.0 Hz), 7.24 (1H, m), 7.27 (2H, d, J = 8.2 Hz), 7.29 (2H, brt, J = 7.0 Hz), 7.84 (2H, d , J = 8.2 Hz), 9.71 (1H, br).
元素分析値: C26H2 03S3として計算値: C, 59.18; H, 5.54; N, 7.96; S, 18.23Elemental analysis: C 26 H 2 0 3 S 3 Calculated: C, 59.18; H, 5.54 ; N, 7.96; S, 18.23
(%)、 実測値: C, 58.92; H, 5.53; N, 7.95: S, 18.37 (%) . (%), Found: C, 58.92; H, 5.53; N, 7.95: S, 18.37 (%).
実施例 4-194 Example 4-194
¾-NMR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 7.2 Hz), 1.34 (2H, quint, J= 7.3 Hz), 1.44 (2H, brquint, J= 7.4 Hz), 2.05 (2H, brt, J= 7.1 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J= 1 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J= 8.3 Hz), 9.72 (1H, br). ¾-NMR (300MHz, CDC1 3 ): δ 0.91 (3Η, t, J = 7.2 Hz), 1.34 (2H, quint, J = 7.3 Hz), 1.44 (2H, brquint, J = 7.4 Hz), 2.05 (2H , brt, J = 7.1 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J = 1 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.32 (5H , s), 7.80 (2H, d, J = 8.3 Hz), 9.72 (1H, br).
元素分析値: C24H25N303S3 · 0.5H2Oとして計算値: C, 56.67; H, 5.15; N, 8.26; S, 18.91 ( ) .実測値: C, 56.71; H, 5.12; N, 8.36; S, 19.12 (%) . Elemental analysis: C 24 H 25 N 3 0 3 S 3 · 0.5H 2 O Calculated:. C, 56.67; H, 5.15; N, 8.26; S, 18.91 () Found: C, 56.71; H, 5.12; N, 8.36; S, 19.12 (%).
実施例 4-195 Example 4-195
Ή-NMR (300MHz, CDC13) : <5 0.91 (3H, t, J= 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.06 (2H, brt, J= 6.9 Hz), 2.40 (3H, s), 4.63 (2H, s), 5.04 (2H, s), 6.49 (1H, brt, J= 0.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.33 (5H, m), 7.86 (2H, brd, J= 8.3 Hz), 9.83 (1H, brs). 元素分析値: C25H27N304S3として計算値: C, 56.69; H, 5.14; N, 7.93; S, 18.16 Ή-NMR (300MHz, CDC1 3 ): <5 0.91 (3H, t, J = 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.06 (2H, brt, J = 6.9 Hz), 2.40 (3H, s), 4.63 (2H, s), 5.04 (2H, s), 6.49 (1H, brt, J = 0.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.33 (5H, m ), 7.86 (2H, brd, J = 8.3 Hz), 9.83 (1H, brs). Elemental analysis: Calculated as C 25 H 27 N 3 0 4 S 3: C, 56.69; H, 5.14; N, 7.93; S, 18.16
(%)、 実測値: C, 56.49; H, 5.06; N, 7.91; S, 18.23 (%) . (%), Found: C, 56.49; H, 5.06; N, 7.91; S, 18.23 (%).
実施例 4- 196 Example 4-196
'H-NMR (300MHz, CDC13) δ 0.92 (3Η, t, J= 7,1 Hz), 1.33 (2H, m), 1.41 (2H, m), 2.09 (2H, brdd, J= 8.0, 6.8 Hz), 2.24 (3H, s), 2.40 (3H, s), 4.20 (2H, s), 7.24 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J= 8.3 Hz), 9.62 (1H, br). 'H-NMR (300MHz, CDC1 3) δ 0.92 (3Η, t, J = 7,1 Hz), 1.33 (2H, m), 1.41 (2H, m), 2.09 (2H, brdd, J = 8.0, 6.8 Hz), 2.24 (3H, s), 2.40 (3H, s), 4.20 (2H, s), 7.24 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J = 8.3 Hz), 9.62 (1H, br).
元素分析値: C25H27N303S3 · 0.6H20として計算値: C, 57.25; H, 5.42; N, 8.01; S, 18.34 (%), 実測値: C, 57.35; H, 5.12; N, 7.89; S, 18.13 (%) Elemental analysis: C 25 H 27 N 3 0 3 S 3 · 0.6H 2 0 Calculated: C, 57.25; H, 5.42; N, 8.01; S, 18.34 (%), Observed: C, 57.35; H , 5.12; N, 7.89; S, 18.13 (%)
実施例 4-197 Example 4-197
融点 159- 161°C Melting point 159-161 ° C
赤外吸収スぺクトル(IR) (KBr中) V: 3202, 2926, 2853, 1732, 1657, 1615, 1559, 1497 cm— Infrared absorption spectrum (IR) (in KBr) V: 3202, 2926, 2853, 1732, 1657, 1615, 1559, 1497 cm—
'H-NMR (CDC13) : δ 0.929 (3Η, t, J=6.8 Hz), 0.86-1.92 (15H, m), 2.12 (2H, t, J=6.6 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, d, J=7.0 Hz), 7.27 (2H, d, J=8.0 Hz), 7.86 (2H, d, J-8. Hz), 9.61 (1H, bs). 'H-NMR (CDC1 3) : δ 0.929 (3Η, t, J = 6.8 Hz), 0.86-1.92 (15H, m), 2.12 (2H, t, J = 6.6 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, d, J = 7.0 Hz), 7.27 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J-8.Hz), 9.61 (1H, bs) .
元素分析値: C25H33N303S3として計算値: C, 57.77; H, 6.40; N, 8.08; S, 18.51 (%), 実測値: C, 57.66 ; H, 6.36; N, 7.90; S, 18.58 (%) . Elemental analysis: C 25 H 33 N 3 0 3 S 3 Calculated: C, 57.77; H, 6.40 ; N, 8.08; S, 18.51 (%), Found: C, 57.66; H, 6.36 ; N, 7.90; S, 18.58 (%).
実施例 4-198 Example 4-198
融点 138- 140°C 138-140 ° C
赤外吸収スペクトル(IR) (KBr中) リ: 3199, 2957, 2930, 2870, 1732, 1659, 1615,Infrared absorption spectrum (IR) (in KBr): 3199, 2957, 2930, 2870, 1732, 1659, 1615,
1559, 1495 cm"1. 1559, 1495 cm " 1 .
Ή-NMR (CDC13) : δ 0.927 (3Η, t, J=7.0 Hz), 1.20-1.50 (4H, m), 1.37 (3H, t, J=7.2 Hz), 2.11 (2H, t, J=6.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.99 (2H, q, J=7.6 Hz), 7.31 (2H, d, J=8.0 Hz), 7.86 (2H, d, J=8. Hz), 9.63 (1H, bs). 元素分析値: C2。H25N303S3として計算値: C, 53.19; H, 5.58; N, 9.30; S, 21.30 (%) 実測値: C, 53.28; H, 5.52; N, 9.50; S, 21.47 (%) . Ή-NMR (CDC1 3): δ 0.927 (3Η, t, J = 7.0 Hz), 1.20-1.50 (4H, m), 1.37 (3H, t, J = 7.2 Hz), 2.11 (2H, t, J = 6.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.99 (2H, q, J = 7.6 Hz), 7.31 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8. Hz), 9.63 (1H, bs) elemental analysis:. C 2. Calculated for H 25 N 3 0 3 S 3 : C, 53.19; H, 5.58; N, 9.30; S, 21.30 (%) Found: C, 53.28; H, 5.52; N, 9.50; S, 21.47 (% ).
実施例 4-199 Example 4-199
融点 139- 141で 赤外吸収スぺクトル(IR) (KBr中) V: 3202, 2957, 1932, 1870, 1740, 1657, 1617, 1563, 1497 cm"1. With melting point 139-141 Infrared absorption spectrum (IR) (in KBr) V: 3202, 2957, 1932, 1870, 1740, 1657, 1617, 1563, 1497 cm " 1 .
Ή-NMR (CDC13) : δ 0.930 (3Η, t, J=6.6 Hz), 0.932 (6H, d, J=6.2Hz), 1.24-1.74 (7H, m), 2.12 (2H, t, ]=7.2 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J=7.6 Hz), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). 元素分析値: C23H31N303S3として計算値: C, 55.95; H, 6.33; N, 8.51; S, 19.48 Ή-NMR (CDC1 3): δ 0.930 (3Η, t, J = 6.6 Hz), 0.932 (6H, d, J = 6.2Hz), 1.24-1.74 (7H, m), 2.12 (2H, t,] = 7.2 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.6 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = . 8.4 Hz), 9.62 (1H , bs) elemental analysis: C 23 H 31 N 3 0 3 S 3 calculated: C, 55.95; H, 6.33 ; N, 8.51; S, 19.48
(%), 実測値: C, 55.89; H, 6.33; N, 8.68; S, 19.57 (%) . (%), Found: C, 55.89; H, 6.33; N, 8.68; S, 19.57 (%).
実施例 4-200 Example 4-200
融点 170- 172 Melting point 170-172
赤外吸収スペクトル(IR) (KBr中) V : 3180, 2955, 2928, 2867, 1728, 1655, 1609, 1561 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3180, 2955, 2928, 2867, 1728, 1655, 1609, 1561 cm " 1 .
¾-NMR (CDC13) : δ 0.912 (3Η, t, J=7.0Hz), 0.927 (3H, t, J=7.0Hz), 1.24-1.52 (8H, m), 1.60-1.78 (2H, m), 2.11 (3H, t, J=7.8 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J=7.8 Hz), 7.30 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.61 (1H, bs). ¾-NMR (CDC1 3): δ 0.912 (3Η, t, J = 7.0Hz), 0.927 (3H, t, J = 7.0Hz), 1.24-1.52 (8H, m), 1.60-1.78 (2H, m) , 2.11 (3H, t, J = 7.8 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J = 7.8 Hz), 7.30 (2H, d, J = 8.0 Hz) , 7.85 (2H, d, J = 8.4 Hz), 9.61 (1H, bs).
元素分析値: C23H31N303S3として計算値: C, 55.95; H, 6.33; N, 8.51; S, 19.48Elemental analysis: Calculated as C 23 H 31 N 3 0 3 S 3: C, 55.95; H, 6.33; N, 8.51; S, 19.48
(%;)、 実測値: C, 55.73; H, 6.37; N, 8.22; S, 19.45 (%) . (%;), Found: C, 55.73; H, 6.37; N, 8.22; S, 19.45 (%).
実施例 4-201 Example 4-201
酢酸ェチル-メ夕ノ一ル-テトラヒドロフランから再結晶し、 濃赤色結晶を得た。 融点 155- 157°C. Recrystallization from ethyl acetate-methyl-tetrahydrofuran gave dark red crystals. 155-157 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3229, 2957, 2932, 2861, 1732, 1657, 1617, 1559, 1508 cm-1. Infrared absorption spectrum (IR) (in KBr) V: 3229, 2957, 2932, 2861, 1732, 1657, 1617, 1559, 1508 cm- 1 .
一 MR (CDCI3) : δ 0.925 (3Η, t, J=7.0 Hz), 1.24-1.52 (4H, m), 2.12 (2H, t, J-8.0 Hz), 2.26 (3H, s), 2.43 (3H, s), 2.94 (2H, t, J=7.6 Hz), 3.17 (2H, t, J=8.6Hz), 6.99 (2H, t, J=8. Hz), 7.09-7.20 (2H, m), 7.28 (2H, d, J=8.0 Hz), 7.82 (2H, d, J=8.4 Hz), 9.63 (1H, bs). One MR (CDCI3): δ 0.925 (3Η, t, J = 7.0 Hz), 1.24-1.52 (4H, m), 2.12 (2H, t, J-8.0 Hz), 2.26 (3H, s), 2.43 (3H , s), 2.94 (2H, t, J = 7.6 Hz), 3.17 (2H, t, J = 8.6 Hz), 6.99 (2H, t, J = 8 Hz), 7.09-7.20 (2H, m), 7.28 (2H, d, J = 8.0 Hz), 7.82 (2H, d, J = 8.4 Hz), 9.63 (1H, bs).
元素分析値: C26H28N303S3Fとして計算値: C, 57.22; H, 5.17; N, 7.70; S, 17.63Elemental analysis: C 26 H 28 N 3 0 3 S 3 F Calculated: C, 57.22; H, 5.17 ; N, 7.70; S, 17.63
(%)、 実測値: C, 57.24; H, 5.31; , 7.65; S, 17.55 (%) . (%), Found: C, 57.24; H, 5.31 ;, 7.65; S, 17.55 (%).
実施例 4-202 酢酸ェチル -テトラヒドロフランから再結晶し濃赤色結晶を得た。 Example 4-202 Recrystallization from ethyl acetate-tetrahydrofuran gave dark red crystals.
融点 167- 168°C. 167-168 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3220, 2955, 2936, 2863, 1732, 1659, 1615, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr): 3220, 2955, 2936, 2863, 1732, 1659, 1615, 1557 cm " 1 .
'H-NMR (CDC13) : δ 0.930 (3Η, t, J=6.6 Hz), 1.20-1.90 (10H, m), 2.03-2.18 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 2.96 (2H, t, J-7.4 Hz), 3.54 (2H, t, J=6.2 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.63 (1H, bs). 元素分析値: C23 。N303S3CIとして計算値: C, 52.30; H, 5.73; N, 7.96; S, 18.21 'H-NMR (CDC1 3) : δ 0.930 (3Η, t, J = 6.6 Hz), 1.20-1.90 (10H, m), 2.03-2.18 (2H, m), 2.26 (3H, s), 2.42 (3H , s), 2.96 (2H, t, J-7.4 Hz), 3.54 (2H, t, J = 6.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz) ), 9.63 (1H, bs) elemental analysis:. C 23. Calculated for N 3 0 3 S 3 CI: C, 52.30; H, 5.73; N, 7.96; S, 18.21
(%), 実測値: C, 52.24; H, 5.54; N, 8.05; S, 17.97 (%) . (%), Found: C, 52.24; H, 5.54; N, 8.05; S, 17.97 (%).
実施例 4-203 Example 4-203
酢酸ェチル -ジイソプロピルェ一テルから再結晶し濃赤色結晶を得た。 Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals.
融点: 154-156^ Melting point: 154-156 ^
赤外吸収スペクトル(IR) (KBr中) V: 3214, 2957, 2863, 1732, 1661, 1617, 1559 cm— Infrared absorption spectrum (IR) (in KBr) V: 3214, 2957, 2863, 1732, 1661, 1617, 1559 cm—
Ή-NMR (CDC") : δ 0.927 (3Η, t, J=7.0Hz), 1.24-1.51 (4H, m), 1.92-2.22 (6H, m), 2.26 (3H, s), 2.42 (3H, s), 3.02 (2H, t, J=7.0 Hz), 7.29 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.4 Hz), 9.62 (1H, bs) . Ή-NMR (CDC "): δ 0.927 (3Η, t, J = 7.0Hz), 1.24-1.51 (4H, m), 1.92-2.22 (6H, m), 2.26 (3H, s), 2.42 (3H, s), 3.02 (2H, t, J = 7.0 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C23H26N303S3F5として計算値: C, 47.33; H, 4.49; N, 7.20; S, 16.48 (%), 実測値: C, 47.51; H, 4.25; N, 7.30; S, 16.46 (%) . Elemental analysis: C 23 H 26 N 3 0 3 S 3 F 5 Calculated: C, 47.33; H, 4.49 ; N, 7.20; S, 16.48 (%), Found: C, 47.51; H, 4.25 ; N, 7.30; S, 16.46 (%).
実施例 4-204 Example 4-204
ィソプロピルエーテル- n-へキサンから再結晶し濃赤色結晶を得た。 Recrystallization from isopropyl ether-n-hexane gave dark red crystals.
融点 111-113^. Melting point 111-113 ^.
赤外吸収スペクトル(IR) (KBr中) V: 3235, 2961, 1934, 2865, 1742, 1728, 1659, 1615, 1564, 1555 cnf1. Infrared absorption spectrum (IR) (in KBr) V: 3235, 2961, 1934, 2865, 1742, 1728, 1659, 1615, 1564, 1555 cnf 1 .
Ή-NMR (CDCI3) : δ 0.925 (3Η, t, J=6.6 Hz), 1.24-1.55 (4H, m), 2.12 (3H, t, J=7.8 Hz), 2.18-2.34 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 2.95-3.16 (3H, m), 7.30 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=8. Hz), 9.66 (1H, bs). Ή-NMR (CDCI3): δ 0.925 (3Η, t, J = 6.6 Hz), 1.24-1.55 (4H, m), 2.12 (3H, t, J = 7.8 Hz), 2.18-2.34 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 2.95-3.16 (3H, m), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.Hz), 9.66 ( 1H, bs).
実施例 4-205 Example 4-205
酢酸ェチル -ジェチルエーテルから再結晶し濃赤色結晶を得た。 融点: 162-164°C. Recrystallization from ethyl acetate-getyl ether gave dark red crystals. Melting point: 162-164 ° C.
赤外吸収スペクトル(IR) (KBr中) ν 3230, 2957, 2934, 2870, 1732, 1659, 1615, 1559 cm—1. Infrared absorption spectrum (IR) (in KBr) ν 3230, 2957, 2934, 2870, 1732, 1659, 1615, 1559 cm- 1 .
¾-NMR (CDC13) : a 0.930 (3H, t, J=6.6 Hz), 0.943 (9H, s), 1.24-1.64 (6H, m), 2.12 (2H, t, J=7. Hz), 2.26 (3H, s), 2.42 (3H, s), 2.85-2.97 (2H, m), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.63 (1H, bs). ¾-NMR (CDC1 3): a 0.930 (3H, t, J = 6.6 Hz), 0.943 (9H, s), 1.24-1.64 (6H, m), 2.12 (. 2H, t, J = 7 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.85-2.97 (2H, m), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H , Bs).
元素分析値: C24H33N303S3として計算値: C, 56.77; H, 6.55; N, 8.28; S, 18.95Elemental analysis: Calculated as C 24 H 33 N 3 0 3 S 3: C, 56.77; H, 6.55; N, 8.28; S, 18.95
(%)、 実測値: C, 56.40; H, 6.62; N, 8.02; S, 18.71 (%) . (%), Found: C, 56.40; H, 6.62; N, 8.02; S, 18.71 (%).
実施例 4-206 Example 4-206
Ή-NM (300MHz, CDC : δ 0.90 (3Η, t, J= 6.9 Hz), 1.29 (2H, m), 1.31 (2H, m), 1.45 (2H, brquint, J= 7.3 Hz), 2.04 (2H, brt, J= 7.1 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J= 8.3 Hz), 9.74 (1H, br). Ή-NM (300MHz, CDC: δ 0.90 (3Η, t, J = 6.9 Hz), 1.29 (2H, m), 1.31 (2H, m), 1.45 (2H, brquint, J = 7.3 Hz), 2.04 (2H , brt, J = 7.1 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.32 (5H , s), 7.80 (2H, d, J = 8.3 Hz), 9.74 (1H, br).
元素分析値: C25H27N303S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73Elemental analysis: Calculated as C 25 H 27 N 3 0 3 S 3: C, 58.45; H, 5.30; N, 8.18; S, 18.73
(%) 、 実測値: C, 58.39; H, 5.23; N, 8.07; S, 18.86 (%) . (%), Found: C, 58.39; H, 5.23; N, 8.07; S, 18.86 (%).
実施例 4-207 Example 4-207
Ή-NMR (300MHz, CDC13) : δ 0.91 (3Η, i, J= 6.8 Hz), 1.32 (4H, m), 1.48 (2H, brquint, J= 7.2 Hz), 2.12 (2H, t, J= 7.7 Hz), 2.27 (3H, s), 2.35 (3H, s), 4.67 (2H, s), 7.06 (2H, d, J= 7.9 Hz), 7.43 (1H, dd, J= 7.9, 7.2 Hz), 7.53 (2H, ffl), 7.54 (1H, dd, J= 6.4, 3.4 Hz), 7.68 (2H, d, J= 8.33 Hz), 7.85 (1H, brd, J- 8.3 Hz), 7.91 (1H, dt, J= 9.4, 3.4 Hz), 8.01 (1H, dt, J= 9.8, 3.4 Hz), 9.59 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3Η, i, J = 6.8 Hz), 1.32 (4H, m), 1.48 (2H, brquint, J = 7.2 Hz), 2.12 (2H, t, J = 7.7 Hz), 2.27 (3H, s), 2.35 (3H, s), 4.67 (2H, s), 7.06 (2H, d, J = 7.9 Hz), 7.43 (1H, dd, J = 7.9, 7.2 Hz) , 7.53 (2H, ffl), 7.54 (1H, dd, J = 6.4, 3.4 Hz), 7.68 (2H, d, J = 8.33 Hz), 7.85 (1H, brd, J- 8.3 Hz), 7.91 (1H, dt, J = 9.4, 3.4 Hz), 8.01 (1H, dt, J = 9.8, 3.4 Hz), 9.59 (1H, brs).
元素分析値: C3。H31N303S3として計算値: C, 62.36; H, 5.41; N, 7.27; S, 16.65Elemental analysis: C 3. Calculated for H 31 N 3 0 3 S 3 : C, 62.36; H, 5.41; N, 7.27; S, 16.65
(%) .実測値: C, 62.44; H, 5.52; N, 7.26; S, 16.82 (%) . Found: C, 62.44; H, 5.52; N, 7.26; S, 16.82 (%).
実施例 4-208 Example 4-208
¾-N R (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J= 7.3 Hz), 2.10 (2H, t, J= 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.32 (2H, s), 7.18 (1H, dt, J= 1.5, 7.5 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.30 (1H, dt, J= l.l, 7.5 Hz), 7.43 (1H, dd, J= 7.5, 1.5 Hz), 7.58 (1H, dd, J= 7.9, 1.1 Hz), 7.82 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). ¾-NR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J = 7.3 Hz), 2.10 (2H, t, J = 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.32 (2H, s), 7.18 (1H, dt, J = 1.5, 7.5 Hz), 7.25 (2H, d, J = 8.3 Hz) , 7.30 (1H, dt, J = ll, 7.5 Hz), 7.43 (1H, dd, J = 7.5, 1.5 Hz), 7.58 (1H, dd, J = 7.9, 1.1 Hz), 7.82 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C26H28N303S3Brとして計算値: C, 51.48; H, 4.65; N, 6.93; S, 15.86; Br, 13.17 ( ), 実測値: C, 51.48; H, 4.62; N, 6.95; S, 15.83; Br, 13.20 (%) . Elemental analysis: C 26 H 28 N 3 0 3 S 3 Br Calculated: C, 51.48; H, 4.65 ; N, 6.93; S, 15.86; Br, 13.17 (), Found: C, 51.48; H, 4.62; N, 6.95; S, 15.83; Br, 13.20 (%).
実施例 4-209 Example 4-209
Ή-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.6 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J= 7.3 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.31 (2H, s), 7.26 (4H, m) , 7.41 (2H, m), 7.82 (2H, d, J= 8.7 Hz), 9.60 (1H, brs). 元素分析値: C26H28N303S3C1として計算値: C, 55.55; H, 5.02; N, 7.47; S, 17.11; C1, 6.31 (%) 、 実測値: C, 55.48; H, 4.95; N, 7.51; S, 17.23; C1, 6.28 (%) . 実施例 4-210 Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 6.6 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J = 7.3 Hz), 2.10 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.31 (2H, s), 7.26 (4H, m), 7.41 (2H, m), 7.82 (2H, d, J = 8.7 Hz) , 9.60 (1H, brs). Elemental analysis: Calculated for C 26 H 28 N 3 0 3 S 3 C1: C, 55.55; H, 5.02; N, 7.47; S, 17.11; C1, 6.31 (%), Found: C, 55.48; H, 4.95; N, 7.51; S, 17.23; C1, 6.28 (%).
一 NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.8 Hz), 1.30 (4H, m), 1.43 (2H, brquint, J= 7.3 Hz), 2.09 (2H, t, J= 7.5 Hz), 2.24 (3H, s), 2.41 (3H, s), 4.23 (2H, s), 7.07 (1H, dt, J- 1.0, 8.7 Hz), 7.12 (1H, dt, J= l.l, 7.5 Hz), 7.25 (2H, d, Jこ 8.3 Hz), 7.30 (1H, m), 7.37 (1H, dt, J= 1.5, 7.5 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). One NMR (300MHz, CDC1 3): δ 0.90 (3Η, t, J = 6.8 Hz), 1.30 (4H, m), 1.43 (2H, brquint, J = 7.3 Hz), 2.09 (2H, t, J = 7.5 Hz), 2.24 (3H, s), 2.41 (3H, s), 4.23 (2H, s), 7.07 (1H, dt, J-1.0, 8.7 Hz), 7.12 (1H, dt, J = ll, 7.5 Hz ), 7.25 (2H, d, J 8.3 Hz), 7.30 (1H, m), 7.37 (1H, dt, J = 1.5, 7.5 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.62 (1H , brs).
元素分析値: C26¾8N303S3Fとして計算値: C, 57.22; H, 5.17; N, 7.70; S, 17.63; F, 3.48 (%), 実測値: C, 57.28; H, 5.15; N, 7.74; S, 17.71; F, 3.33 (%) . 実施例 4- 211 Elemental analysis: C 26 ¾ 8 N 3 0 3 S 3 F Calculated: C, 57.22; H, 5.17; N, 7.70; S, 17.63; F, 3.48 (%), found: C, 57.28; H , 5.15; N, 7.74; S, 17.71; F, 3.33 (%).
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.31 (4H, m), 1.45 (2H, brquint, J= 7.3 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.30 (2H, s), 7.01 (1H, dt, J= 1.5, 7.5 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.33 (1H, dt, J= 1.1, 7.5 Hz) , 7.43 (1H, dd, J= 7.9, 1.5 Hz), 7.82 (2H, d, J= 8.3 Hz), 7.86 (1H, dd, J= 7.9, 1.5 Hz), 9.6,0 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 1.31 (4H, m), 1.45 (2H, brquint, J = 7.3 Hz), 2.10 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.30 (2H, s), 7.01 (1H, dt, J = 1.5, 7.5 Hz), 7.25 (2H, d, J = 8.3 Hz) , 7.33 (1H, dt, J = 1.1, 7.5 Hz), 7.43 (1H, dd, J = 7.9, 1.5 Hz), 7.82 (2H, d, J = 8.3 Hz), 7.86 (1H, dd, J = 7.9) , 1.5 Hz), 9.6,0 (1H, brs).
元素分析値: C26H28N303S3Iとして計算値: C, 47.78; H, 4.32; N, 6.43; S, 14.72; I, 19.42 (%), 実測値: C, 47.79; H, 4.29; N, 6.40; S, 14.88; I, 19.45 (%) . 実施例 4-212 Elemental analysis: C 26 H 28 N 3 0 3 S 3 I Calculated: C, 47.78; H, 4.32 ; N, 6.43; S, 14.72; I, 19.42 (%), Found: C, 47.79; H , 4.29; N, 6.40; S, 14.88; I, 19.45 (%).
Ή-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.6 Hz) , 1.32 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 2.12 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.36 (3H, s), 2.40 (3H, s), 4.20 (2H5 s), 7.15-7.28 (6H, m) , 7.78 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 6.6 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 2.12 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.36 (3H, s), 2.40 (3H, s), 4.20 (2H 5 s), 7.15-7.28 (6H, m), 7.78 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C27H31N303S3として計算値: C, 59.86; H, 5.77; N, 7.76; S, 17.76 (%) 、 実測値: C, 59.81; H, 5.72; N, 7.85; S, 17.67 (%) . Elemental analysis: C 27 H 31 N 3 0 3 S 3 Calculated: C, 59.86; H, 5.77 ; N, 7.76; S, 17.76 (%), Found: C, 59.81; H, 5.72 ; N, 7.85; S, 17.67 (%).
実施例 4-213 Example 4-213
Ή-NMR (300MHz, CDC13) : δ 0.88 (3H, t, J= 6.8 Hz), 1.27 (4H, m), 1.41 (2H, brquint, J= 7.3 Hz), 2.08 (2H, t, J= 7.5 Hz), 2.23 (3H, s), 2.38 (3H, s), 4.36 (2H, s), 7.21 (2H, d, J= 7.9 Hz), 7.42 (1H, dd, J= 8.3, 1.9 Hz), 7.50 (2H, m), 7.77-7.84 (6H, m), 9.62 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3H, t, J = 6.8 Hz), 1.27 (4H, m), 1.41 (2H, brquint, J = 7.3 Hz), 2.08 (2H, t, J = 7.5 Hz), 2.23 (3H, s), 2.38 (3H, s), 4.36 (2H, s), 7.21 (2H, d, J = 7.9 Hz), 7.42 (1H, dd, J = 8.3, 1.9 Hz) , 7.50 (2H, m), 7.77-7.84 (6H, m), 9.62 (1H, brs).
元素分析値: C3。H31N303S3として計算値: C, 62.36; H, 5.41; N, 7.27; S, 16.65 (%)、 実測値: C, 62.00; H, 5.35; N, 7.32; S, 16.40 (%) . Elemental analysis: C 3. Calculated for H 31 N 3 0 3 S 3 : C, 62.36; H, 5.41; N, 7.27; S, 16.65 (%), found: C, 62.00; H, 5.35; N, 7.32; S, 16.40 ( %).
実施例 4-214 Example 4-214
Ή-NMR (300MHz, CDC13) : 6 0.89 (3H, t, J= 7.0 Hz), 1.27 (4H, m), 1.37 (2H, brquint, J= 7.5 Hz), 2.01 (2H, brt, J- 7.5 Hz), 2.22 (3H, s), 2.41 (3H, s), 4.14 (2H, s), 7.25 (2H, d, J= 8.3 Hz), 7.26-7.41 (8H, m), 7.81 (2H, d, J= 8.3 Hz), 9.59 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): 6 0.89 (3H, t, J = 7.0 Hz), 1.27 (4H, m), 1.37 (2H, brquint, J = 7.5 Hz), 2.01 (2H, brt, J- 7.5 Hz), 2.22 (3H, s), 2.41 (3H, s), 4.14 (2H, s), 7.25 (2H, d, J = 8.3 Hz), 7.26-7.41 (8H, m), 7.81 (2H, d, J = 8.3 Hz), 9.59 (1H, brs).
元素分析値: C32H33N303S3 · 0.3H20として計算値: C, 63.09; H, 5.56; N, 6.90; S, 15.79 ( ), 実測値: C, 62.97; H, 5.34; N, 6.95; S, 15.60 (%) . Elemental analysis: C 32 H 33 N 3 0 3 S 3 · 0.3H 2 0 Calculated: C, 63.09; H, 5.56 ; N, 6.90; S, 15.79 (), Found: C, 62.97; H, 5.34; N, 6.95; S, 15.60 (%).
実施例 4-215 Example 4-215
Ή-N R (300MHz, CDC13) : δ 0.90 (3Η, t, J= 7.0 Hz), 1.30 (4H, m), 1.43 (2H, brquint, J= 7.3 Hz), 2.08 (2H, t, J= 7.5 Hz), 2.24 (3H, s), 2.42 (3H, s), 4.15 (2H, s), 7.22 (1H, m), 7.27 (2H, d, J= 8.3 Hz), 7.27 (2H, m) , 7.31 (2H, brd, J= 8 Hz), 7.82 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). Ή-NR (300MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 7.0 Hz), 1.30 (4H, m), 1.43 (2H, brquint, J = 7.3 Hz), 2.08 (2H, t, J = 7.5 Hz), 2.24 (3H, s), 2.42 (3H, s), 4.15 (2H, s), 7.22 (1H, m), 7.27 (2H, d, J = 8.3 Hz), 7.27 (2H, m) , 7.31 (2H, brd, J = 8 Hz), 7.82 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C26 8N303S3C1として計算値: C, C, 55.55; H, 5.02; N, 7.47; S, 17.11; CI, 6.31 (%) 、 実測値: C, 55.37; H, 4.91; N, 7.52; S, 17.30; CI, 6.31 (%) . 実施例 4-216 ' Elemental analysis: C 26 8 N 3 0 3 S 3 C1 Calculated: C, C, 55.55; H , 5.02; N, 7.47; S, 17.11; CI, 6.31 (%), Found: C, 55.37; H, 4.91; N, 7.52; S, 17.30; CI, 6.31 (%).
lH- NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.2 Hz), 2.11 (2H, brt, J= 7.5 Hz) , 2.24 (3H, s), 2.38 (3H, s), 4.24 (2H, s), 7.25 (2H, d, J= 8.3 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.33 (2H, d, J= 8.3 Hz), 7.36 (1H, dd, J= 7.2, 0.8 Hz), 7.45 (1H, dt, J= 1.5, 7.5 Hz), 7.58 (1H, dt, J= 1.5, 7.5 Hz), 7.81 (2H, d, J= 7.9 Hz), 7.95 (1H, dd, J= 7.5, 1.1 Hz), 9.64 (1H, brs). lH- NMR (300MHz, CDC1 3) : δ 0.90 (3H, t, J = 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.2 Hz), 2.11 (2H, brt, J = 7.5 Hz), 2.24 (3H, s), 2.38 (3H, s), 4.24 (2H, s), 7.25 (2H, d, J = 8.3 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.33 (2H, d, J = 8.3 Hz), 7.36 (1H, dd, J = 7.2, 0.8 Hz), 7.45 (1H, dt, J = 1.5, 7.5 Hz), 7.58 (1H, dt, J = 1.5, 7.5 Hz), 7.81 ( 2H, d, J = 7.9 Hz), 7.95 (1H, dd, J = 7.5, 1.1 Hz), 9.64 (1H, brs).
元素分析値: C33H29N305S3 · 0.9H20として計算値: C, 60.05; H, 4.70; N, 6.37 (%)、 実測値: C, 60.11; H, 4.99; N, 6.51 (%) . Elemental analysis: C 33 H 29 N 3 0 5 S 3 · 0.9H 2 0 Calculated: C, 60.05; H, 4.70 ; N, 6.37 (%), Found: C, 60.11; H, 4.99 ; N , 6.51 (%).
実施例 4-217 Example 4-217
Ή-NMR (300MHz, CDC13) : δ .0.91 (3Η, t, J= 6.8 Hz), 1.24 (9H, s), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.40 (3H, s), 4.26 (2H, s), 7.25 (2H, d, J= 7.9 Hz), 7.28 (2H, d, J- 8.3 Hz), 7.30 (1H, m), 7.34 (1H, m), 7.35 (2H, d, J= 8.3 Hz), 7.41 (1H, dt, J= 1.5, 7.5 Hz), 7.50 (1H, dt, J= 1.5, 7.5 Hz), 7.80 (2H, d, J= 7.9 Hz), 9.65 (1H, brs). 元素分析値: C37H37N305S3 · 0.3¾0として計算値: C, 63.00; H, 5.37; N, 5.96; S, 13.64 (%), 実測値: C, 63.05; H, 5.43; N, 6.07; S, 13.67 (%) . Ή-NMR (300MHz, CDC1 3 ): δ .0.91 (3Η, t, J = 6.8 Hz), 1.24 (9H, s), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.40 (3H, s), 4.26 (2H, s), 7.25 (2H, d, J = 7.9 Hz), 7.28 (2H, d , J- 8.3 Hz), 7.30 (1H, m), 7.34 (1H, m), 7.35 (2H, d, J = 8.3 Hz), 7.41 (1H, dt, J = 1.5, 7.5 Hz), 7.50 (1H , Dt, J = 1.5, 7.5 Hz), 7.80 (2H, d, J = 7.9 Hz), 9.65 (1H, brs). Elemental analysis: Calculated as C 37 H 37 N 3 0 5 S 3 · 0.3¾0 : C, 63.00; H, 5.37; N, 5.96; S, 13.64 (%), found: C, 63.05; H, 5.43; N, 6.07; S, 13.67 (%).
実施例 4-218 Example 4-218
'H-NMR (300MHz, CDC13) : <5 0.90 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J= 7.3 Hz), 2.09 (2H, t, J= 7.5 Hz), 2.24 (3H, s), 2.31 (3H, s), 2.41 (3H, s), 4.18 (2H, s), 7.06 (2H, d, J= 8.7 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.33 (2H, d, ]= 8.3 Hz), 7.81 (2H, d, J- 8.3 Hz), 9.63 (1H, brs). 'H-NMR (300MHz, CDC1 3): <5 0.90 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J = 7.3 Hz), 2.09 (2H, t, J = 7.5 Hz), 2.24 (3H, s), 2.31 (3H, s), 2.41 (3H, s), 4.18 (2H, s), 7.06 (2H, d, J = 8.7 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.33 (2H, d,] = 8.3 Hz), 7.81 (2H, d, J- 8.3 Hz), 9.63 (1H, brs).
元素分析値: C28H31N305S3として計算値: C, 57.41; H, 5.33; N, 7.17; S, 16.42 (%) 、 実測値: C, 57.27; H, 5.03; N, 7.09; S, 16.49 (%) . Elemental analysis: C 28 H 31 N 3 0 5 S 3 Calculated: C, 57.41; H, 5.33; N, 7.17; S, 16.42 (%), found: C, 57.27; H, 5.03; N, 7.09; S, 16.49 (%).
実施例 4-219 Example 4-219
Ή-NMR (300MHz, CDC13) : <5 0.90 (3H, t, J= 6.8 Hz), 1.29 (4H, m), 1.42 (2H, brquint, J= 7.3 Hz), 2.08 (2H, t, J= 7.5 Hz), 2.24 (3H, s), 2.42 (3H, s), 4.15 (2H, s), 7.26 (4H, m), 7.29 (2H, m), 7.81 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). 元素分析値: CZ6H28N303S3C1として計算値: C, 55.55; H, 5.02; N, 7.47; S, 17.11; Cl, 6.31 (%), 実測値: C, 55.47; H, 4.94; N, 7.49; S, 17.22; C1, 6.52 (%) · 実施例 4-220 Ή-NMR (300MHz, CDC1 3 ): <5 0.90 (3H, t, J = 6.8 Hz), 1.29 (4H, m), 1.42 (2H, brquint, J = 7.3 Hz), 2.08 (2H, t, J = 7.5 Hz), 2.24 (3H, s), 2.42 (3H, s), 4.15 (2H, s), 7.26 (4H, m), 7.29 (2H, m), 7.81 (2H, d, J = 8.3 Hz ), 9.62 (1H, brs) elemental analysis: C Z6 H 28 N 3 0 3 S 3 C1 calculated:. C, 55.55; H, 5.02; N, 7.47; S, 17.11; Cl, 6.31 (%) , Found: C, 55.47; H, 4.94; N, 7.49; S, 17.22; C1, 6.52 (%) · Example 4-220
Ή-NM (300MHz, CDC : <5 0.89 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J= 7.2 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.39 (3H, s), 4.24 (2H, s), 7.24 (2H, d, J= 8.3 Hz), 7.36 (1H, t, J= 7.2 Hz), 7.39 (2H, d, J= 8.3 Hz), 7.45 (2H, i, J- 7.4 Hz), 7.56 (2H, d, J= 8.7 Hz), 7.58 (2H, dm, J= 8 Hz), 7.82 (2H, d, J= 8.3 Hz), 9.65 (1H, brs). Ή-NM (300MHz, CDC: <5 0.89 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.44 (2H, brquint, J = 7.2 Hz), 2.10 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.39 (3H, s), 4.24 (2H, s), 7.24 (2H, d, J = 8.3 Hz), 7.36 (1H, t, J = 7.2 Hz), 7.39 (2H, d, J = 8.3 Hz), 7.45 (2H, i, J- 7.4 Hz), 7.56 (2H, d, J = 8.7 Hz), 7.58 (2H, dm, J = 8 Hz), 7.82 (2H, d, J = 8.3 Hz), 9.65 (1H, brs).
元素分析値: C32H33N303S3として計算値: C, 63.65; H, 5.51; N, 6.96; S, 15.93 (%)、 実測値: C, 63.91; H, 5.39; N, 6.67; S, 15.78 (%) Elemental analysis: C 32 H 33 N 3 0 3 S 3 Calculated: C, 63.65; H, 5.51 ; N, 6.96; S, 15.93 (%), Found: C, 63.91; H, 5.39 ; N, 6.67; S, 15.78 (%)
実施例 4-221 Example 4-221
Ή-NMR (300MHz, CDC13) : d 0.91 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.33 (9H, s), 1.45 (2H, brquint, J= 7.3 Hz), 2.10 (2H, brt, J= 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.18 (2H, s), 7.25 (4H, d, J= 8.3 Hz), 7.35 (2H, d, J= 8.3 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.61 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): d 0.91 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.33 (9H, s), 1.45 (2H, brquint, J = 7.3 Hz), 2.10 (2H, brt, J = 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.18 (2H, s), 7.25 (4H, d, J = 8.3 Hz), 7.35 (2H, d, J = 8.3 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.61 (1H, brs).
元素分析値: C3。H37N303S3として計算値: C, 61.72; H, 6.39; N, 7.20; S, 16.48Elemental analysis: C 3. H 37 N 3 0 3 S 3 Calculated: C, 61.72; H, 6.39 ; N, 7.20; S, 16.48
(%) 、 実測値: C, 61.55; H, 6.34; N, 7.23; S, 16.55 (%) . (%), Found: C, 61.55; H, 6.34; N, 7.23; S, 16.55 (%).
実施例 4-222 Example 4-222
'H-NMR (300MHz, CDC13) : δ 0.84 (3Η, t, J= 6.8 Hz), 1.21 (4H, m), 1.32 (2H, brquint, J= 7.3 Hz), 1.99 (2H, t, J= 7.5 Hz), 2.17 (3H, s), 2.42 (3H, s), 4.53 (2H, s), 7.29 (2H, m), 7.30 (2H, d, J= 8.3 Hz), 7.64 (2H, dt, J= 9.4, 3. Hz) , 7.83 (2H, d, J= 8.3 Hz). 'H-NMR (300MHz, CDC1 3): δ 0.84 (3Η, t, J = 6.8 Hz), 1.21 (4H, m), 1.32 (2H, brquint, J = 7.3 Hz), 1.99 (2H, t, J = 7.5 Hz), 2.17 (3H, s), 2.42 (3H, s), 4.53 (2H, s), 7.29 (2H, m), 7.30 (2H, d, J = 8.3 Hz), 7.64 (2H, dt) , J = 9.4, 3.Hz), 7.83 (2H, d, J = 8.3 Hz).
元素分析値: C27H29N503S3 · 0.2H20として計算値: C, 56.76; H, 5.19; N, 12.26; S, 16.84 (%) 、 実測値: C, 56.71; H, 5.33; N, 12.35; S, 17.03 (%) . Elemental analysis: C 27 H 29 N 5 0 3 S 3 · 0.2H 2 0 Calculated: C, 56.76; H, 5.19 ; N, 12.26; S, 16.84 (%), Found: C, 56.71; H , 5.33; N, 12.35; S, 17.03 (%).
実施例 4-223 Example 4-223
'H-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.0 Hz), 1.61 (3H, s), 1.69 (3H, s), 1.69 (3H, s)., 2.06 (4H, m), 2.10 (2H, brt, J= 7.3 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.63 (2H, d, J= 7.6 Hz), 5.07 (1H, brt, J= 6 Hz), 5.24 (1H, brt, J= 7.6 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.90 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.0 Hz), 1.61 (3H, s), 1.69 (3H, s), 1.69 (3H, s)., 2.06 (4H, m), 2.10 (2H, brt, J = 7.3 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.63 ( 2H, d, J = 7.6 Hz), 5.07 (1H, brt, J = 6 Hz), 5.24 (1H, brt, J = 7.6 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C29H29N303S3 · 0.2H20として計算値: C, 60.32; H, 6.88; N, 7.28; S, 16.66 (%)、 実測値: C, 60.25; H, 6.54; N, 7.27; S, 16.61 (%) . Elemental analysis: C 29 H 29 N 3 0 3 S 3 · 0.2H 2 0 Calculated: C, 60.32; H, 6.88; N, 7.28; S, 16.66 (%), Observed: C, 60.25; H , 6.54; N, 7.27; S, 16.61 (%).
実施例 4-224 Example 4-224
Ή-NMR (300MHz, CDC13) : <5 0.88 (6H, d, J= 6.4 Hz), 0.90 (3H, t, J= 6.8 Hz), 1.20 (2H, quint, J= 7.3 Hz), 1.31 (4H, m), 1.37 (2H, m), 1.45 (2H, brquint, J= 7.2 Hz), 1.54 (1H, quint, J= 6.4 Hz), 1.66 (2H, quint, J= 7.5 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J= 7.4 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): <5 0.88 (6H, d, J = 6.4 Hz), 0.90 (3H, t, J = 6.8 Hz), 1.20 (2H, quint, J = 7.3 Hz), 1.31 (4H, m), 1.37 (2H, m), 1.45 (2H, brquint, J = 7.2 Hz), 1.54 (1H, quint, J = 6.4 Hz), 1.66 (2H, quint, J = 7.5 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.64 (1H, brs).
元素分析値: C26 7N303S3として計算値: C, 58.28; H, 6.96; , 7.84; S, 17.95 (%) 、 実測値: C, 58.26; H, 6.85; , 7.82; S, 18.19 (%) . Elemental analysis: Calculated as C 26 7 N 3 0 3 S 3: C, 58.28; H, 6.96;, 7.84; S, 17.95 (%), Found: C, 58.26; H, 6.85 ;, 7.82; S , 18.19 (%).
実施例 4-225 Example 4-225
'H-NMR (300MHz, CDC13) : δ 0.88 (6H, d, J= 6.4 Hz), 0.90 (3H, t, J= 6.8 Hz), 1.18 (2H, brt, J= 7.2 Hz), 1.31 (8H, m), 1.38 (2H, m), 1.47 (2H, m), 1.53 (1H, quint, J= 6.4 Hz), 1.68 (2H, quint, J= 7.3 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J二 8.3 Hz), 9.63 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.88 (6H, d, J = 6.4 Hz), 0.90 (3H, t, J = 6.8 Hz), 1.18 (2H, brt, J = 7.2 Hz), 1.31 ( 8H, m), 1.38 (2H, m), 1.47 (2H, m), 1.53 (1H, quint, J = 6.4 Hz), 1.68 (2H, quint, J = 7.3 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J (8.3 Hz), 9.63 (1H, brs).
元素分析値: C27H3gN303S3として計算値: C, 58.98; H, 7.15; N, 7.64; S, 17.50 (%), 実測値: C, 59.03; H, 7.05; N, 7.52; S, 17.67 (%) . Elemental analysis: Calculated as C 27 H 3g N 3 0 3 S 3: C, 58.98; H, 7.15; N, 7.64; S, 17.50 (%), Found: C, 59.03; H, 7.05 ; N, 7.52; S, 17.67 (%).
実施例 4-226 Example 4-226
'H-NMR (300MHz, CDC13) : δ 0.90 (6Η, brt, J= 6.4 Hz), 1.31 (8H, m), 1.40 (2H, m), 1.46 (2H, brquint, J= 7.4 Hz), 1.67 (2H, quint, J= 7.3 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J= 7.3 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). ' 元素分析値: C25H35N303S3として計算値: C, 57.55; H, 6.76; N, 8.05; S, 18.44 (%)、 実測値: C, 57.35; H, 6.53; N, 8.11; S, 18.41 ( ) . 'H-NMR (300MHz, CDC1 3): δ 0.90 (6Η, brt, J = 6.4 Hz), 1.31 (8H, m), 1.40 (2H, m), 1.46 (2H, brquint, J = 7.4 Hz), 1.67 (2H, quint, J = 7.3 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J = 7.3 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs). 'Elemental analysis: Calculated as C 25 H 35 N 3 0 3 S 3 : C, 57.55; H, 6.76; N, 8.05; S, 18.44 (%), found: C, 57.35; H, 6.53; N, 8.11; S, 18.41 ().
実施例 4-227 Example 4-227
Ή- R (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.6 Hz), 0.90 (3H, t, J- 6.6 Hz), 1.30 (12H, m), 1.47 (2H, brquint, J= 7.2 Hz), 1.68 (2H, quint, J= 7.5 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.93 (2H, t, J= 7.3 Hz), 7.28 (2H, d: J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Ή- R (300MHz, CDC1 3) : δ 0.90 (3Η, t, J = 6.6 Hz), 0.90 (3H, t, J- 6.6 Hz), 1.30 (12H, m), 1.47 (2H, brquint, J = 7.2 Hz), 1.68 (2H, quint, J = 7.5 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.93 (2H, t, J = 7.3 Hz), 7.28 (2H, d: J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C26H37N303S3として計算値: C, 58.28; H, 6.96; N, 7.84; S, 17.95Elemental analysis: C 26 H 37 N 3 0 3 S 3 Calculated: C, 58.28; H, 6.96 ; N, 7.84; S, 17.95
(%) 、 実測値: C, 58.21; H, 6.84; , 7.89; S, 17.73 (%) . (%), Found: C, 58.21; H, 6.84;, 7.89; S, 17.73 (%).
実施例 4-228 Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 0.90 (3H, i, J= 6.8 Hz), 1.29 (14H, m), 1.47 (2H, brquint, J= 7.2 Hz), 1.67 (2H, quint, J- 7.5 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.93 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Example 4-228 Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 0.90 (3H, i, J = 6.8 Hz), 1.29 (14H, m), 1.47 (2H, brquint, J = 7.2 Hz), 1.67 (2H, quint, J- 7.5 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.93 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C„H3gN303S3として計算値: C, 58.98; H, 7.15; N, 7.64; S, 17.50 (%), 実測値: C, 58.86; H, 7.18; N, 7.64; S, 17.31 ( ) . Elemental analysis: C „H 3g Calculated as N 3 0 3 S 3 : C, 58.98; H, 7.15; N, 7.64; S, 17.50 (%), found: C, 58.86; H, 7.18; N, 7.64; S, 17.31 ().
実施例 4-229 Example 4-229
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.97 (2H, dd, J= 8.7, 7.2 Hz), 3.17 (2H, dd, J= 8.7, 7.5 Hz), 7.20 (2H, dm, J= 7.2 Hz), 7.26-7.35 (5H, m), 7.83 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.97 (2H, dd, J = 8.7, 7.2 Hz), 3.17 (2H, dd, J = 8.7, 7.5 Hz), 7.20 (2H, dm, J = 7.2 Hz), 7.26-7.35 (5H, m), 7.83 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C27H31N303S3として計算値: C, 59.86; H, 5.77; N, 7.76; S, 17.76Elemental analysis: C 27 H 31 N 3 0 3 S 3 Calculated: C, 59.86; H, 5.77 ; N, 7.76; S, 17.76
(%)、 実測値: C, 59.51; H, 5.77; N, 7.85; S, 17.77 (%) . (%), Found: C, 59.51; H, 5.77; N, 7.85; S, 17.77 (%).
実施例 4-230 Example 4-230
¾-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.31 (4H, m), 1.44 (2H, brquint, J= 7.2 Hz), 2.01 (2H, quint, J= 7.5 Hz), 2.08 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.39 (3H, s), 2.72 (2H, t, J= 7.3 Hz), 2.92 (2H, t, J= 7.3 Hz), 7.15 (2H, d, J= 7.5 Hz), 7.21 (1H, tt, J= 7.2, 2.6 Hz), 7.28 (4H, m), 7.85 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). ¾-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 1.31 (4H, m), 1.44 (2H, brquint, J = 7.2 Hz), 2.01 (2H, quint, J = 7.5 Hz), 2.08 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.39 (3H, s), 2.72 (2H, t, J = 7.3 Hz), 2.92 (2H, t, J = 7.3 Hz), 7.15 (2H, d, J = 7.5 Hz), 7.21 (1H, tt, J = 7.2, 2.6 Hz), 7.28 (4H, m), 7.85 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C28H33N303S3として計算値: C, 60.51; H, 5.98; N, 7.56; S, 17.31 (%) 、 実測値: C, 60.46; H, 5.86; N, 7.64; S, 17.57 (%) · Elemental analysis: C 28 H 33 N 3 0 3 S 3 Calculated: C, 60.51; H, 5.98 ; N, 7.56; S, 17.31 (%), Found: C, 60.46; H, 5.86 ; N, 7.64; S, 17.57 (%)
実施例 4-231 Example 4-231
Ή-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J= 6.7 Hz), 1.24 (4H, m), 1.26 (2H, brquint, J= 6.8 Hz), 1.97 (2H, t, J= 7.2 Hz), 2.17 (3H, s), 2.42 (3H, s), 5.86 (1H, s), 7.24 (2H, d, J= 8.3 Hz), 7.32 (10H, m), 7.84 (2H, d, J- 8.3 Hz), 9.61 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3Η, t, J = 6.7 Hz), 1.24 (4H, m), 1.26 (2H, brquint, J = 6.8 Hz), 1.97 (2H, t, J = 7.2 Hz), 2.17 (3H, s), 2.42 (3H, s), 5.86 (1H, s), 7.24 (2H, d, J = 8.3 Hz), 7.32 (10H, m), 7.84 (2H, d, J- 8.3 Hz), 9.61 (1H, brs).
元素分析値: C32H33N303S3として計算値: C, 63.65; H, 5.51; N, 6.96; S, 15.93Elemental analysis: C 32 H 33 N 3 0 3 S 3 Calculated: C, 63.65; H, 5.51 ; N, 6.96; S, 15.93
(%), 実測値: C, 63.64; H, 5.48; N, 6.93; S, 15.96 (%) . (%), Found: C, 63.64; H, 5.48; N, 6.93; S, 15.96 (%).
実施例 4-232 'H-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J- 6.6 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J= 7.3 Hz), 2.11 (2H, t, J= 7.7 Hz), 2.25 (3H, s), 2.39 (3H, s), 3.80 (2H, dd, J= 7.2, 1.1 Hz), 6.18 (1H, dt, J= 15.5, 7.2 Hz), 6.61 (1H, d, J= 15.8 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.28 (1H, m), 7.34 (4H, m), 7.84 (2H, d, J= 8.3 Hz), 9.65 (1H, brs). Example 4-232 'H-NMR (300MHz, CDC1 3): δ 0.89 (3H, t, J- 6.6 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J = 7.3 Hz), 2.11 (2H, t, J = 7.7 Hz), 2.25 (3H, s), 2.39 (3H, s), 3.80 (2H, dd, J = 7.2, 1.1 Hz), 6.18 (1H, dt, J = 15.5, 7.2 Hz), 6.61 (1H , d, J = 15.8 Hz), 7.26 (2H, d, J = 8.3 Hz), 7.28 (1H, m), 7.34 (4H, m), 7.84 (2H, d, J = 8.3 Hz), 9.65 (1H , brs).
元素分析値: C28H31N303S3として計算値: C, 60.73; H, 5.64; N, 7.59; S, 17.37 (%), 実測値: C, 60.71; H, 5.46; N, 7.77; S, 17.23 (%) . Elemental analysis: C 28 H 31 N 3 0 3 S 3 Calculated: C, 60.73; H, 5.64 ; N, 7.59; S, 17.37 (%), Found: C, 60.71; H, 5.46 ; N, 7.77; S, 17.23 (%).
実施例 4- 233 Example 4-233
Ή—NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.9 Hz) , 1.30 (4H, m), 1.44 (2H, quint, J= 7.3 Hz), 2.09 (2H, dd, J= 7.8, 7.3 Hz), 2.24 (3H, s), 2.42 (3H, s), 4.20 (2H, s), 7.25 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J二 8.3 Hz) , 9.59 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 6.9 Hz), 1.30 (4H, m), 1.44 (2H, quint, J = 7.3 Hz), 2.09 (2H, dd, J = 7.8, 7.3 Hz), 2.24 (3H, s), 2.42 (3H, s), 4.20 (2H, s), 7.25 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.81 (2H, s) d, J2 8.3 Hz), 9.59 (1H, brs).
元素分析値: C26H29N303S3 · 0.2H2Oとして計算値: C, 58.77; H, 5.58; N, 7.91; S, 18.11 (%) 、 実測値: C, 58.75; H, 5.19; N, 8.03; S, 18.47 (%) . Elemental analysis: C 26 H 29 N 3 0 3 S 3 · 0.2H 2 O Calculated: C, 58.77; H, 5.58 ; N, 7.91; S, 18.11 (%), Found: C, 58.75; H , 5.19; N, 8.03; S, 18.47 (%).
実施例 4-234 Example 4-234
'H-NMR (300MHz, CDC13) : <5 0.91 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.50 (2H, brquint, J= 7.3 Hz), 2.13 (2H, t, J= 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.25 (2H, dd, J= 7.2, 6.8 Hz), 3.99 (2H, dd, J= 7.2, 6.8 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.75 (2H, dd, J= 5.3, 3.0 Hz), 7.81 (2H, d, J= 8.3 Hz), 7.86 (2H, dd, J= 5.3, 3.0 Hz), 9.60 (1H, brs). 'H-NMR (300MHz, CDC1 3): <5 0.91 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.50 (2H, brquint, J = 7.3 Hz), 2.13 (2H, t, J = 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.25 (2H, dd, J = 7.2, 6.8 Hz), 3.99 (2H, dd, J = 7.2, 6.8 Hz), 7.26 ( 2H, d, J = 8.3 Hz), 7.75 (2H, dd, J = 5.3, 3.0 Hz), 7.81 (2H, d, J = 8.3 Hz), 7.86 (2H, dd, J = 5.3, 3.0 Hz), 9.60 (1H, brs).
元素分析値: C29H3。N405S3として計算値: C, 57.03; H, 4.95; N, 9.17; S, 15.75 (%), 実測値: C, 56.76; H, 4.81; , 9.31; S, 15.94 (%) . Elemental analysis: C 29 H 3. N 4 0 5 S 3 Calculated: C, 57.03; H, 4.95 ; N, 9.17; S, 15.75 (%), Found: C, 56.76; H, 4.81 ;, 9.31; S, 15.94 (%).
実施例 4-235 Example 4-235
■H-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.8 Hz), 1.28 (4H, i), 1.41 (2H, brquint, J= 7.4 Hz) , 2.04 (2H, t, J= 7.5 Hz), 2.07 (2H, quint, J= 6.8 Hz), 2.24 (3H, s), 2.39 (3H, s), 2.97 (2H, dd, 1= 7.9, 7.5 Hz), 3.77 (2H, t, J= 6.8 Hz), 7.28 (2H, brd, J- 8 Hz), 7.74 (2H, dd, J= 5.7, 3.0 Hz), 7.84 (2H, d, J= 8 Hz), 7.86 (2H, dd, J= 5.3, 3.4 Hz), 9.61 (1H, brs). ■ H-NMR (300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.8 Hz), 1.28 (4H, i), 1.41 (2H, brquint, J = 7.4 Hz), 2.04 (2H, t, J = 7.5 Hz), 2.07 (2H, quint, J = 6.8 Hz), 2.24 (3H, s), 2.39 (3H, s), 2.97 (2H, dd, 1 = 7.9, 7.5 Hz), 3.77 (2H, t , J = 6.8 Hz), 7.28 (2H, brd, J-8 Hz), 7.74 (2H, dd, J = 5.7, 3.0 Hz), 7.84 (2H, d, J = 8 Hz), 7.86 (2H, dd , J = 5.3, 3.4 Hz), 9.61 (1H, brs).
元素分析値: C3。H32N405S3として計算値: C, 57.37; H, 5.16; N, 8.97; S, 15.40 (%), 実測値: C, 57.63; H, 5.14; , 9.06; S, 15.44 (%) . Elemental analysis: C 3. H 32 N 4 0 5 S 3 Calculated: C, 57.37; H, 5.16 ; N, 8.97; S, 15.40 (%), Found: C, 57.63; H, 5.14;, 9.06; S, 15.44 (%).
実施例 4-236 Example 4-236
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.28 (4H, m), 1.39 (2H, brquint, J= 7.3 Hz), 2.06 (2H, t, J= 7.5 Hz), 2.23 (3H, s), 2.43 (3H, s), 4.14 (2H, s), 7.26 (2H, m), 7.28 (2H, d, J= 8.3 Hz), 7.82 (2H, d, J= 8.3 Hz), 8.57 (2H, d, J= 6.0 Hz), 9.62· (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.28 (4H, m), 1.39 (2H, brquint, J = 7.3 Hz), 2.06 (2H, t, J = 7.5 Hz), 2.23 (3H, s), 2.43 (3H, s), 4.14 (2H, s), 7.26 (2H, m), 7.28 (2H, d, J = 8.3 Hz), 7.82 (2H, d, J = 8.3 Hz), 8.57 (2H, d, J = 6.0 Hz), 9.62 (1H, brs).
元素分析値: C25H28N403S3として計算値: C, 56.79; H, 5.34; N, 10.60; S, 18.19Elemental analysis: Calculated for C 25 H 28 N 4 0 3 S 3 : C, 56.79; H, 5.34; N, 10.60; S, 18.19
(%) 、 実測値: C, 56.51; H> 5.52; N, 10.63; S, 18.36 (%) . (%), Found: C, 56.51; H> 5.52; N, 10.63; S, 18.36 (%).
実施例 4-237 Example 4-237
Ή-NMR (300MHz, CDC13) : δ 0.85 (3H, t, J= 6.8 Hz), 1.22 (4Η,' m), 1.35 (2H, brquint, J= 7.2 Hz), 2.04 (2H, t, J= 7.5 Hz), 2.22 (3H, s), 2.39 (3H, s), 4.52 (2H, s), 7.23 (2H, d, J= 8.3 Hz), 7.53 (1H, d, J= 8.7 Hz), 7.57 (1H, dt, J= 1.0, 7.2 Hz), 7.74 (1H, ddd, J= 8.3, 7.2, 1.5 Hz), 7.83 (1H, brd, J= 8.3 Hz), 7.83 (2H, d, J= 8.3 Hz), 8.05 (1H, brd, J= 8.3 Hz), 8.15 (1H, d, J= 8.3 Hz), 9.65 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.85 (3H, t, J = 6.8 Hz), 1.22 (4Η, 'm), 1.35 (2H, brquint, J = 7.2 Hz), 2.04 (2H, t, J = 7.5 Hz), 2.22 (3H, s), 2.39 (3H, s), 4.52 (2H, s), 7.23 (2H, d, J = 8.3 Hz), 7.53 (1H, d, J = 8.7 Hz), 7.57 (1H, dt, J = 1.0, 7.2 Hz), 7.74 (1H, ddd, J = 8.3, 7.2, 1.5 Hz), 7.83 (1H, brd, J = 8.3 Hz), 7.83 (2H, d, J = 8.3 Hz), 8.05 (1H, brd, J = 8.3 Hz), 8.15 (1H, d, J = 8.3 Hz), 9.65 (1H, brs).
元素分析値: C29H3。N403S3 · 0.5H20として計算値: C, 59.26; H, 5.32; N, 9.53; S, 16.36 (%), 実測値: C, 59.28; H, 5.30; N, 9.59; S, 16.49 (%) . Elemental analysis: C 29 H 3. N 4 0 3 S 3 · 0.5H 2 0 Calculated: C, 59.26; H, 5.32; N, 9.53; S, 16.36 (%), Found: C, 59.28; H, 5.30; N, 9.59; S , 16.49 (%).
実施例 4-238 Example 4-238
Ή-NMR (300MHz, CDC13) : 6 0.90 (3H, t, J= 6.8 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 1.62 (3H, s), 1.71 (3H, s), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.36 (2H, q, J- 7.5 Hz), 2.41 (3H, s), 2.93 (2H, t, J= 7.5 Hz), 5.11 (1H, tt, J= 7.2, 1.2 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, .brs). Ή-NMR (300MHz, CDC1 3 ): 6 0.90 (3H, t, J = 6.8 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 1.62 (3H, s), 1.71 (3H, s), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.36 (2H, q, J-7.5 Hz), 2.41 (3H, s), 2.93 (2H, t, J = 7.5 Hz), 5.11 (1H, tt, J = 7.2, 1.2 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, .brs ).
元素分析値: C25H33N303S3として計算値: C, 57.77; H, 6.40; , 8.08; S, 18.51 (%), 実測値: C, 57.65; H, 6.29; N, 8.15; S, 18.70 (%) Elemental analysis: C 25 H 33 N 3 0 3 S 3 Calculated: C, 57.77; H, 6.40 ;, 8.08; S, 18.51 (%), Found: C, 57.65; H, 6.29 ; N, 8.15 ; S, 18.70 (%)
実施例 -239 Example -239
'H-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.6 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 1.69 (3H, s), 1.76 (3H, s), 2.10 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.61 (2H, d, J= 7.5 Hz), 5.23 (1H, tt, J= 7.7, 1.5 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.91 (3H, t, J = 6.6 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 1.69 (3H, s), 1.76 (3H, s), 2.10 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.61 (2H, d, J = 7.5 Hz), 5.23 (1H, tt , J = 7.7, 1.5 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C24H31N303S3として計算値: C, 57.00; H, 6.18; N, 8.31; S, 19.02Elemental analysis: Calculated as C 24 H 31 N 3 0 3 S 3: C, 57.00; H, 6.18; N, 8.31; S, 19.02
(%) 、 実測値: C, 57.06; H, 6.02; N, 8.27; S, 18.72 (%) . (%), Found: C, 57.06; H, 6.02; N, 8.27; S, 18.72 (%).
実施例 4-240 Example 4-240
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 7.5 Hz), 0.90 (6H, t, J= 6.8 Hz), 1.30 (10H, m), 1.40 (2H, quint, J= 7.2 Hz), 1.47 (2H, brquint, J= 7.2 Hz), Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 7.5 Hz), 0.90 (6H, t, J = 6.8 Hz), 1.30 (10H, m), 1.40 (2H, quint, J = 7.2 Hz), 1.47 (2H, brquint, J = 7.2 Hz),
1.61 (1H, quint, J= 6.0 Hz), 2.11 (2H, t, J= 7.7 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.91 (2H, t, J= 6.4 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). 1.61 (1H, quint, J = 6.0 Hz), 2.11 (2H, t, J = 7.7 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.91 (2H, t, J = 6.4 Hz), 7.26 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C27H39N303S3として計算値: C, 58.98; H, 7.15; N, 7.64; S, 17.50Elemental analysis: Calculated as C 27 H 39 N 3 0 3 S 3: C, 58.98; H, 7.15; N, 7.64; S, 17.50
(%)、 実測値: C, 59.06; H, 7.04; N> 7.75; S, 17.54 (%) . (%), Found: C, 59.06; H, 7.04; N> 7.75; S, 17.54 (%).
実施例 4-241 Example 4-241
Ή-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.8 Hz), 1.21 (2H, m), 1.31 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz) , 1.57 (1H, q, J= 7.9 Hz), 1.67 (1H, m), 1.71 (4H, brd, J= 10.6 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.96 (2H, t, J= 7.9 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 6.8 Hz), 1.21 (2H, m), 1.31 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 1.57 (1H, q, J = 7.9 Hz), 1.67 (1H, m), 1.71 (4H, brd, J = 10.6 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.96 (2H, t, J = 7.9 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz),
9.62 (1H, brs). 9.62 (1H, brs).
実施例 4-242 Example 4-242
Ή-NMR (300MHz, CDC13) : δ 0.88 (2H, brq, J= 12 Hz), 0.91 (3H, t, J= 6.6 Hz), 1.18 (2H, m), 1.26 (4H, m), 1.31 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 1.68 (7H, m), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s) , 2.41 (3H, s), 2.91 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). 元素分析値: C28H39N303S3として計算値: C, 59.86; H, 7.00; N, 7.48; S, 17.12 Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (2H, brq, J = 12 Hz), 0.91 (3H, t, J = 6.6 Hz), 1.18 (2H, m), 1.26 (4H, m), 1.31 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 1.68 (7H, m), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s) , 2.91 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs). Elemental analysis: C 28 H 39 N 3 0 3 S 3 calculated: C, 59.86; H, 7.00 ; N, 7.48; S, 17.12
(%) 、 実測値: C, 60.00; H, 6.99; N, 7.48; S, 17.02 (%) . (%), Found: C, 60.00; H, 6.99; N, 7.48; S, 17.02 (%).
実施例 4-243 Example 4-243
Ή—NMR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 6.6 Hz), 0.98 (2H, brq, J- 11.7 Hz), 1.20 (2H, brt, J= 10.2 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 1.67 (1H, m), 1.73 (2H, brd, J= 13.9 Hz), 1.85 (2H, brd, J= 12.0 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, t, J= 6.8 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.61 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3Η, t, J = 6.6 Hz), 0.98 (2H, brq, J- 11.7 Hz), 1.20 (2H, brt, J = 10.2 Hz), 1.32 (4H , m), 1.47 (2H, brquint, J = 7.3 Hz), 1.67 (1H, m), 1.73 (2H, brd, J = 13.9 Hz), 1.85 (2H, brd, J = 12.0 Hz), 2.11 (2H , t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, t, J = 6.8 Hz), 7.27 (2H, s) d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.61 (1H, brs).
元素分析値: C26H35N303S3として計算値: C, 58.50; H, 6.61; , 7.87; S, 18.02 (%), 実測値: C, 58.27; H, 6.80; N, 7.93; S, 18.05 ( ) . Elemental analysis: C 26 H 35 N 3 0 3 S 3 Calculated: C, 58.50; H, 6.61 ;, 7.87; S, 18.02 (%), Found: C, 58.27; H, 6.80 ; N, 7.93 ; S, 18.05 ().
実施例 4-244 Example 4-244
Ή-NMR (300MHz, CDC : δ 0.91 (3Η, t, J= 6.8 Hz), 1.12 (2H, m), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.5 Hz), 1.50-1.65 (4H, m), 1.68 (2H, q, J= 7.9 Hz), 1.79 (2H, m), 1.87 (2H, quint, J= 7.5 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, d, J= 7.7 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Ή-NMR (300MHz, CDC: δ 0.91 (3Η, t, J = 6.8 Hz), 1.12 (2H, m), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.5 Hz), 1.50-1.65 (4H, m), 1.68 (2H, q, J = 7.9 Hz), 1.79 (2H, m), 1.87 (2H, quint, J = 7.5 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, d, J = 7.7 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: CMH35N303S3として計算値: C, 58.50; H, 6.61; N, 7.87; S, 18.02Elemental analysis: C M H 35 N 3 0 3 S 3 Calculated: C, 58.50; H, 6.61 ; N, 7.87; S, 18.02
(%)、 実測値: C, 58.48; H, 6.47; N, 7.84; S, 18.05 (%) . (%), Found: C, 58.48; H, 6.47; N, 7.84; S, 18.05 (%).
実施例 4-245 Example 4-245
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.08 (2H, in), 1.31 (6H, m), 1.43 (2H, m), 1.45-1.55 (4H, m), 1.59 (2H, m), 1.68 (2H, q, J= 7.8 Hz), 1.75 (2H, m), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.93 (2H, d, J= 7.3 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs) . Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 1.08 (2H, in), 1.31 (6H, m), 1.43 (2H, m), 1.45-1.55 (4H, m), 1.59 (2H, m), 1.68 (2H, q, J = 7.8 Hz), 1.75 (2H, m), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 ( 3H, s), 2.93 (2H, d, J = 7.3 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C27H37N303S3として計算値: C, 59.20; H, 6.81; , 7.67; S, 17.56Elemental analysis: Calculated as C 27 H 37 N 3 0 3 S 3: C, 59.20; H, 6.81;, 7.67; S, 17.56
( ) 、 実測値: C, 59.07; H, 6.81; N, 7.62; S, 17.61 {%) . (), Found: C, 59.07; H, 6.81; N, 7.62; S, 17.61 (%).
実施例 4-246 Example 4-246
Ή— NMR (300MHz, CDC13) : 6 0.90 (3H, t, J= 6.8 Hz), 1.26 (2H, m), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.3 Hz), 1.61 (4H, m), 1.86 (2H, m), 2.10 (2H, t, J= 7.9 Hz), 2.12 (1H, quint, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, d, J= 7.7 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs) . Ή- NMR (300MHz, CDC1 3) : 6 0.90 (3H, t, J = 6.8 Hz), 1.26 (2H, m), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.3 Hz), 1.61 (4H, m), 1.86 (2H, m), 2.10 (2H, t, J = 7.9 Hz), 2.12 (1H, quint, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s) , 2.95 (2H, d, J = 7.7 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C25H33N303S3として計算値: C, 57.77; H, 6.40; N, 8.08; S, 18.51Elemental analysis: Calculated as C 25 H 33 N 3 0 3 S 3: C, 57.77; H, 6.40; N, 8.08; S, 18.51
(%), 実測値: C, 57.59; H, 6.47; N, 8.08; S, 18.46 (%) . (%), Found: C, 57.59; H, 6.47; N, 8.08; S, 18.46 (%).
実施例 4-247 Example 4-247
Ή-NMR (300MHz, CDC13) : δ 0.32 (2H, brq, J= 5.7 Hz), 0.67 (2H, brq, J= 6.8 Hz), 0.90 (3H, t, J= 6.8 Hz), 1.07 (1H, m), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.4 Hz), 2.10 (2H, t, J= 7.7 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.91 (2H, t, J= 7.2 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.64 (1H, brs) . Ή-NMR (300MHz, CDC1 3 ): δ 0.32 (2H, brq, J = 5.7 Hz), 0.67 (2H, brq, J = 6.8 Hz), 0.90 (3H, t, J = 6.8 Hz), 1.07 (1H, m), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.4 Hz), 2.10 (2H, t, J = 7.7 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.91 (2H, t, J = 7.2 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.87 (2H, d, J = 8.3) Hz), 9.64 (1H, brs).
元素分析値: C23H29N303S3 'として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56 (%)、 実測値: C, 56.39; H, 5.91; N, 8.50; S, 19.56 (%) . Elemental analysis: C 23 H 29 N 3 0 3 S 3 Calculated ': C, 56.18; H, 5.94; N, 8.55; S, 19.56 (%), Found: C, 56.39; H, 5.91 ; N , 8.50; S, 19.56 (%).
実施例 4-248 Example 4-248
Ή-N R (300MHz, CDC13) : δ 0.91 (3Η, t, J= 6.8 Hz), 1.32 (4H, m), 1.48 (2H, m), 1.50 (2H, brquini, J= 7.5 Hz), 1.70 (2H, quint, J= 7.4 Hz), 2.07 (2H, m), 2.11 (2H, t, J= 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, t, J= 7.3 Hz), 4.99 (1H, brd, J= 10.2 Hz), 5.03 (1H, dq, J= 17.0, 1.9 Hz), 5.78 (1H, ddt, J- 17.0, 10.2, 6.8 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Ή-NR (300MHz, CDC1 3 ): δ 0.91 (3Η, t, J = 6.8 Hz), 1.32 (4H, m), 1.48 (2H, m), 1.50 (2H, brquini, J = 7.5 Hz), 1.70 (2H, quint, J = 7.4 Hz), 2.07 (2H, m), 2.11 (2H, t, J = 7.2 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, t, J = 7.3 Hz), 4.99 (1H, brd, J = 10.2 Hz), 5.03 (1H, dq, J = 17.0, 1.9 Hz), 5.78 (1H, ddt, J- 17.0, 10.2, 6.8 Hz), 7.28 ( 2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C25H33N303S3として計算値: C, 57.77; H, 6.40; N, 8.08; S, 18.51 (%) 、 実測値: C, 57.72; H, 6.36; N, 8.12; S, 18.59 (%) · Elemental analysis: C 25 H 33 N 3 0 3 S 3 Calculated: C, 57.77; H, 6.40 ; N, 8.08; S, 18.51 (%), Found: C, 57.72; H, 6.36 ; N, 8.12; S, 18.59 (%)
実施例 4-249 Example 4-249
Ή-NMR' (300MHz, CDCI3) : δ 0.90 (3Η, t, J= 6.8 Hz), 1.32 (4H, m), 1.36 (4H, m), 1.42 (2H, m), 1.47 (2H, brquint, J= 7.7 Hz), 1.68 (2H, quint, J= 7.3 Hz), 2.05 (2H, brq, J= 6.8 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.93 (2H, t, J= 7.3 Hz), 4.96 (1H, brd, J= 10.2 Hz), 5.01 (1H, dq, J= 17.0, 1.5 Hz), 5.80 (1H, ddt, J= 17.0, 10.2, 6.8 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). Ή-NMR '(300 MHz, CDCI 3 ): δ 0.90 (3Η, t, J = 6.8 Hz), 1.32 (4H, m), 1.36 (4H, m), 1.42 (2H, m), 1.47 (2H, brquint , J = 7.7 Hz), 1.68 (2H, quint, J = 7.3 Hz), 2.05 (2H, brq, J = 6.8 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.93 (2H, t, J = 7.3 Hz), 4.96 (1H, brd, J = 10.2 Hz), 5.01 (1H, dq, J = 17.0, 1.5 Hz), 5.80 (1H, ddt , J = 17.0, 10.2, 6.8 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.64 (1H, brs).
元素分析値: C27H37N303S3として計算値: C, 59.20; H, 6.81; , 7.67; S, 17.56 (%), 実測値: C, 59.28; H, 6.51; N, 7.74; S, 17.70 (%) . Elemental analysis: Calculated as C 27 H 37 N 3 0 3 S 3: C, 59.20; H, 6.81;, 7.67; S, 17.56 (%), Found: C, 59.28; H, 6.51 ; N, 7.74 ; S, 17.70 (%).
実施例 4-250 Example 4-250
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.3 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.62 (2H, d, J= 6.8 Hz), 5.25 (1H, dd, J= 10.2, 1.1 Hz), 5.32 (1H, dq, J= 17.0, 1.1 Hz), 5.85 (1H, ddt, J= 17.0, 10.2, 6.8 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.3 Hz), 2.10 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.62 (2H, d, J = 6.8 Hz), 5.25 (1H, dd, J = 10.2, 1.1 Hz), 5.32 (1H, dq, J = 17.0, 1.1 Hz), 5.85 (1H, ddt, J = 17.0, 10.2, 6.8 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.64 (1H, brs).
元素分析値: C22H27N303S3として計算値: C, 55.32; H, 5.70; N, 8.80; S, 20.14Elemental analysis: C 22 H 27 N 3 0 3 S 3 Calculated: C, 55.32; H, 5.70 ; N, 8.80; S, 20.14
(%), 実測値: C, 55.08; H, 5.62; N, 9.01; S, 20.16 ( ) . (%), Found: C, 55.08; H, 5.62; N, 9.01; S, 20.16 ().
実施例 4-251 Example 4-251
'Η -臓 (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.6 Hz), 1.32 (4H, m), 1.46 (6H, m), 1.70 (2H, quint, J= 6.9 Hz), 1.78 (2H, quint, J= 7.0 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J= 7.5 Hz), 3.54 (2H, t, J= 6.6 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs) . 'Eta - organs (300MHz, CDC1 3): δ 0.90 (3Η, t, J = 6.6 Hz), 1.32 (4H, m), 1.46 (6H, m), 1.70 (2H, quint, J = 6.9 Hz), 1.78 (2H, quint, J = 7.0 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.5 Hz), 3.54 (2H, t, J = 6.6 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C25H34N303S3C1 · 0.4H20として計算値: C, 53.30; H, 6.23; N, 7.46; S, 17.07; Cl, 6.29 (%) 、 実測値: C, 53.32; H, 6.06; N, 7.53; S, 17.08; C1, 6.46 (%) . Elemental analysis: C 25 H 34 N 3 0 3 S 3 C1 · 0.4H 2 0 Calculated: C, 53.30; H, 6.23 ; N, 7.46; S, 17.07; Cl, 6.29 (%), Found: C, 53.32; H, 6.06; N, 7.53; S, 17.08; C1, 6.46 (%).
実施例 4-252 Example 4-252
Ή-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.6 Hz), 1.32 (6H, m), 1.50 (4H, m), 1.70 (4H, m), 2.05 (2H, m), 2.12 (2H, t, J= 7.4 Hz), 2.25 (3H, s), 2.42 (3H, s), 3.53 (1H, septet, J= 4.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J- 8.3 Hz), 9.63 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 6.6 Hz), 1.32 (6H, m), 1.50 (4H, m), 1.70 (4H, m), 2.05 (2H, m) , 2.12 (2H, t, J = 7.4 Hz), 2.25 (3H, s), 2.42 (3H, s), 3.53 (1H, septet, J = 4.5 Hz), 7.28 (2H, d, J = 8.3 Hz) , 7.86 (2H, d, J- 8.3 Hz), 9.63 (1H, brs).
元素分析値: C26H35N303S3として計算値: C, 58.50; H, 6.61; N, 7.87; S, 18.02 (%), 実測値: C, 58.43; H, 6.57; N, 7.89; S, 17.86 (%) . Elemental analysis: Calculated for C 26 H 35 N 3 0 3 S 3 : C, 58.50; H, 6.61; N, 7.87; S, 18.02 (%), found: C, 58.43; H, 6.57; N, 7.89; S, 17.86 (%).
実施例 4-253 Example 4-253
Ή-N R (300MHz, CDC13) : 6 0.90 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.5 Hz), 1.62 (1H, in), 1.82 (1H, m), 1.89 (1H, m), 2.00 (1H, m), 2.05 (2H, m), 2.12 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 4.13 (1H, m), 5.68 (1H, dm, J= 9.8 Hz), 5.94 (1H, dm, J= 9.8 Hz), 7.27 (2H, d, J= 7.9 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). Ή-NR (300MHz, CDC1 3 ): 6 0.90 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.5 Hz), 1.62 (1H, in), 1.82 (1H, m), 1.89 (1H, m), 2.00 (1H, m), 2.05 (2H, m), 2.12 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.41 (3H, m s), 4.13 (1H, m), 5.68 (1H, dm, J = 9.8 Hz), 5.94 (1H, dm, J = 9.8 Hz), 7.27 (2H, d, J = 7.9 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.64 (1H, brs).
元素分析値: C25H31N303S3として計算値: C, 58.00; H, 6.04; N, 8.12; S, 18.58Elemental analysis: Calculated as C 25 H 31 N 3 0 3 S 3: C, 58.00; H, 6.04; N, 8.12; S, 18.58
(%) 実測値: C, 57.94; H, 5.94; N, 8.23; S, 18. 3 (% . (%) Found: C, 57.94; H, 5.94; N, 8.23; S, 18.3 (%.
実施例 4-254 Example 4-254
Ή-NMR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 6.6 Hz), 1.30 (2H, m), 1.31 (6H, m), 1.43 (2H, m), 1.46 (2H, brd, }= 9.0 Hz), 1.77 (2H, brd, J= 8.7 Hz), 2.02 (2H, brd, J= 11.3 Hz), 2.11 (2H, t, J= 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.37 (IH, m), 7.28 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.63 (IH, brs). 元素分析値: C25¾3N303S3 · 0.1H20として計算値: C, 57.57; H, 6.42; N, 8.06; S, 18.44 (%) 、 実測値: C, 57.25; H, 6.27; N, 8.09; S, 18.43 (%) . Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3Η, t, J = 6.6 Hz), 1.30 (2H, m), 1.31 (6H, m), 1.43 (2H, m), 1.46 (2H, brd,} = 9.0 Hz), 1.77 (2H, brd, J = 8.7 Hz), 2.02 (2H, brd, J = 11.3 Hz), 2.11 (2H, t, J = 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.37 (IH, m), 7.28 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3) Hz), 9.63 (IH, brs). Elemental analysis: Calculated as C 25 ¾ 3 N 3 0 3 S 3 · 0.1H 20 : C, 57.57; H, 6.42; N, 8.06; S, 18.44 (% ), Found: C, 57.25; H, 6.27; N, 8.09; S, 18.43 (%).
実施例 4-255 Example 4-255
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.28 (3H, t, J= 7.2 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J= 7.5 Hz), 2.00 (2H, quint, J= 7.4 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.43 (2H, t, J= 7.0 Hz), 3.01 (2H, t, J= 7.2 Hz), 4.15 (2H, Q, J= 7.2 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (IH, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 1.28 (3H, t, J = 7.2 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J = 7.5 Hz), 2.00 (2H, quint, J = 7.4 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.43 (2H, t, J = 7.0 Hz), 3.01 (2H, t, J = 7.2 Hz), 4.15 (2H, Q, J = 7.2 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz) ), 9.63 (IH, brs).
元素分析値: C25 N305S3として計算値: C, 54.42; H, 6.03; N, 7.62; S, 17.43Elemental analysis: C 25 N 3 0 5 S 3 Calculated: C, 54.42; H, 6.03 ; N, 7.62; S, 17.43
(%) .実測値: C, 54.32; H, 5.91; N, 7.70; S, 17.59 (%) . Found: C, 54.32; H, 5.91; N, 7.70; S, 17.59 (%).
実施例 4^256 Example 4 ^ 256
Ή一 NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.8 Hz), 1.26 (3H, t, J= 7.0 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 1.73 (4H, in), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.33 (2H, t, J= 6.8 Hz), 2.42 (3H, s), 2.95 (2H, brt, J= 7.0 Hz), 4.14 (2H, q, J- 7.2 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (IH, brs). Ή one NMR (300MHz, CDC1 3): δ 0.91 (3H, t, J = 6.8 Hz), 1.26 (3H, t, J = 7.0 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 1.73 (4H, in), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.33 (2H, t, J = 6.8 Hz), 2.42 (3H, s), 2.95 (2H, brt, J = 7.0 Hz), 4.14 (2H, q, J- 7.2 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.64 (IH , brs).
元素分析値: C26¾5N305S3として計算値: C, 55.20; H, 6.24; N, 7.43; S, 17.00 (%) ,実測値: C, 55.10; H, 6.43; N, 7.51; S, 16.94 ( ) . Elemental analysis: C 26と し て5 N 3 0 5 S 3 Calculated: C, 55.20; H, 6.24; N, 7.43; S, 17.00 (%), found: C, 55.10; H, 6.43; N, 7.51; S, 16.94 ().
実施例 4-257 Example 4-257
Ή-NM (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 0.90 (3H, t, J= 6.6 Hz), 1.28 (3H, t, J= 7.0 Hz), 1.29 (12H, m), 1.46 (2H, brquint, J= 7.3 Hz), 1.80 (IH, brquint, J= 7.0 Hz), 1.90 (IH, quint, J= 7.7 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.24 (3H, s), 2.42 (3H, s), 4.00 (IH, t, J= 7.2 Hz), 4.20 (2H, q, J= 7.0 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.61 (1H, brs). 実施例 4 - 258 Ή-NM (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 0.90 (3H, t, J = 6.6 Hz), 1.28 (3H, t, J = 7.0 Hz), 1.29 (12H , m), 1.46 (2H, brquint, J = 7.3 Hz), 1.80 (IH, brquint, J = 7.0 Hz), 1.90 (IH, quint, J = 7.7 Hz), 2.10 (2H, t, J = 7.5 Hz) ), 2.24 (3H, s), 2.42 (3H, s), 4.00 (IH, t, J = 7.2 Hz), 4.20 (2H, q, J = 7.0 Hz), 7.28 (2H, d, J = 8.3 Hz) ), 7.85 (2H, d, J = 8.3 Hz), 9.61 (1H, brs). Example 4-258
lH_NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 1.31 (4H, m), 1.42 (4H, m), 1.47 (2H, brquint, J= 6.8 Hz), 1.58 (2H, quint, J= 6.7 Hz), 1.70 (2H, quint, J= 7.3 Hz), 2.11 (2H, t, J= 7.7 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J= 7.7 Hz), 3.66 (2H, dt, J= 4.5, 6.4 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). lH_NMR (300MHz, CDC1 3): δ 0.90 (3H, t, J = 6.8 Hz), 1.31 (4H, m), 1.42 (4H, m), 1.47 (2H, brquint, J = 6.8 Hz), 1.58 (2H, quint, J = 6.7 Hz), 1.70 (2H, quint, J = 7.3 Hz), 2.11 (2H, t, J = 7.7 Hz) , 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.7 Hz), 3.66 (2H, dt, J = 4.5, 6.4 Hz), 7.28 (2H, d, J = 8.3) Hz), 7.85 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C25H35N304S3として計算値: C, 55.84; H, 6,56; N, 7.81; S, 17.89 (%)、 実測値: C, 55.73; H, 6.49; N, 7.84; S, 17.87 (%) · Elemental analysis: Calculated as C 25 H 35 N 3 0 4 S 3: C, 55.84; H, 6,56; N, 7.81; S, 17.89 (%), Found: C, 55.73; H, 6.49 ; N, 7.84; S, 17.87 (%)
実施例 4-259 Example 4-259
Ή-NMR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 6.8 Hz), 1.31 (4H, m), 1.37 (6H, m), 1.45 (2H, brquint, J= 7.0 Hz), 1.69 (2H, brquint, J= 7.5 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J= 7.5 Hz), 3.65 (2H, brq, J= 5.9 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs) . Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3Η, t, J = 6.8 Hz), 1.31 (4H, m), 1.37 (6H, m), 1.45 (2H, brquint, J = 7.0 Hz), 1.69 (2H, brquint, J = 7.5 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J = 7.5 Hz), 3.65 (2H, brq, J = 5.9 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C26H37N304S3として計算値: C, 56.59; H, 6.76; N, 7.62; S, 17.43 (%), 実測値: C, 56.52; H, 6.62; N, 7.77; S, 17.37 (%) · Elemental analysis: Calculated as C 26 H 37 N 3 0 4 S 3: C, 56.59; H, 6.76; N, 7.62; S, 17.43 (%), Found: C, 56.52; H, 6.62 ; N, 7.77; S, 17.37 (%)
実施例 4-260 Example 4-260
'H-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.7 Hz), 0.90 (3H, t, J= 6.8 Hz), 1.29 (6H, m), 1.45 (2H, brquint, J= 7.1 Hz), 1.84 (1H, m), 1.96 (1H, m), 2.10 (2H, t, J= 7.5 Hz), 2.23 (3H, s), 2.42 (3H, s), 4.01 (1H, t, J= 4.7 Hz), 7.29 (2H, d, J= 7.9 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.7 Hz), 0.90 (3H, t, J = 6.8 Hz), 1.29 (6H, m), 1.45 (2H, brquint, J = 7.1 Hz), 1.84 (1H, m), 1.96 (1H, m), 2.10 (2H, t, J = 7.5 Hz), 2.23 (3H, s), 2.42 (3H, s), 4.01 (1H, t) , J = 4.7 Hz), 7.29 (2H, d, J = 7.9 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.64 (1H, br).
実施例 4-261 Example 4-261
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 7.0 Hz), 1.28 (4H, i), 1.41 (2H, brquint, J= 7.3 Hz), 2.06 (2H, t, J= 7.5 Hz), 2.21 (3H, s), 2.40 (3H, s), 5.29 (1H, s), 7.25 (2H, d, J- 8.3 Hz), 7.37 (3H, i), 7.42 (2H, dd, J= 7.5, 3.8 Hz), 7.83 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 7.0 Hz), 1.28 (4H, i), 1.41 (2H, brquint, J = 7.3 Hz), 2.06 (2H, t, J = 7.5 Hz), 2.21 (3H, s), 2.40 (3H, s), 5.29 (1H, s), 7.25 (2H, d, J- 8.3 Hz), 7.37 (3H, i), 7.42 (2H, dd, J = 7.5, 3.8 Hz), 7.83 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C27H29N305S3として計算値: C, 56.72; H, 5.11; , 7.35; S, 16.83 (%)、 実測値: C, 56.52; H, 4.96; N, 7.29; S, 16.73 (%) · Elemental analysis: Calculated as C 27 H 29 N 3 0 5 S 3: C, 56.72; H, 5.11;, 7.35; S, 16.83 (%), Found: C, 56.52; H, 4.96 ; N, 7.29 ; S, 16.73 (%)
実施例 4-262 Example 4-262
Ή—NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.45 (2H, brquint, J= 7.3 Hz), 2.09 (2H, t, J= 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.14 (2H, dd, J= 7.5, 6.8 Hz), 3.27 (2H, dd, J= 7.5, 7.3 Hz), 7.06 (1H, d, J= 1.9 Hz), 7.13 (1H, brdt, J= 1.1, 7.5 Hz), 7.22 (1H, dt, J= 1.1, 7.5 Hz), 7.24 (2H, d, J= 8.3 Hz), 7.38 (1H, d, J= 7.9 Hz), 7.59 (1H, d, J= 7.9 Hz), 7.82 (2H, d, J= 8.3 Hz), 8.10 (1H, brs), 9.63 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.45 (2H, brquint, J = 7.3 Hz), 2.09 (2H, t, J = 7.7 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.14 (2H, dd, J = 7.5, 7.3 Hz), 3.27 (2H, dd, J = 7.5, 7.3 Hz), 7.06 (1H, d, J = 1.9 Hz), 7.13 (1H, brdt, J = 1.1, 7.5 Hz), 7.22 (1H, dt, J = 1.1, 7.5 Hz), 7.24 (2H, d, J = 8.3 Hz), 7.38 (1H, d, J = 7.9 Hz), 7.59 (1H, d, J = 7.9) Hz), 7.82 (2H, d, J = 8.3 Hz), 8.10 (1H, brs), 9.63 (1H, brs).
元素分析値: C29H32N403S3として計算値: C, 59.97; H, 5.55; N, 9.65; S, 16.56 (%)、 実測値: C, 59.96; H, 5.78; N, 9.68; S, 16.53 (%) . Elemental analysis: C 29 H 32 N 4 0 3 S 3 Calculated: C, 59.97; H, 5.55; N, 9.65; S, 16.56 (%), found: C, 59.96; H, 5.78; N, 9.68; S, 16.53 (%).
実施例 4-263 Example 4-263
NMR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 6.8 Hz), 0.93 (6H, d, J= 6.4 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.2 Hz), 1.57 (2H, m), 1.69 (1H, septet, J= 6.6 Hz), 2.11 (2H, t, J- 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J= 7.7 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). NMR (300MHz, CDC1 3): δ 0.91 (3Η, t, J = 6.8 Hz), 0.93 (6H, d, J = 6.4 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.2 Hz ), 1.57 (2H, m), 1.69 (1H, septet, J = 6.6 Hz), 2.11 (2H, t, J-7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H , t, J = 7.7 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C24H33N303S3として計算値: C, 56.77; H, 6.55; N, 8.28; S, 18.95 (%), 実測値: C, 56.76; H, 6.47; N, 8.13; S, 18.89 (%) . Elemental analysis: C 24 H 33 N 3 0 3 S 3 Calculated: C, 56.77; H, 6.55 ; N, 8.28; S, 18.95 (%), Found: C, 56.76; H, 6.47 ; N, 8.13; S, 18.89 (%).
実施例 4-264 Example 4-264
lH-NMR (300MHz, CDC13) : δ 0.89 (6Η, d, J= 6.4 Hz), 0.90 (3H, t, J= 6.4 Hz), 1.28 (2H, DI), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.2 Hz), 1.57 (1H, septet, J= 6.8 Hz), 1.68 (2H, m), 2.11 (2H, t, J= 7.4 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.92 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 7.9 Hz), 7.85 (2H, d, J= 7.9 Hz),. 9.63 (1H, brs). lH-NMR (300MHz, CDC1 3 ): δ 0.89 (6Η, d, J = 6.4 Hz), 0.90 (3H, t, J = 6.4 Hz), 1.28 (2H, DI), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.2 Hz), 1.57 (1H, septet, J = 6.8 Hz), 1.68 (2H, m), 2.11 (2H, t, J = 7.4 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.92 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 7.9 Hz), 7.85 (2H, d, J = 7.9 Hz), 9.63 (1H, brs).
元素分析値: C25H35N303S3として計算値: C, 57.55; H, 6.76; N, 8.05; S, 18.44Elemental analysis: Calculated as C 25 H 35 N 3 0 3 S 3: C, 57.55; H, 6.76; N, 8.05; S, 18.44
(%), 実測値: C, 57.46; H, 6.66; N, 7.95; S, 18.59 (%) . (%), Found: C, 57.46; H, 6.66; N, 7.95; S, 18.59 (%).
実施例 4-265 Example 4-265
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.2 Hz), 1.84 (2H, quint, J= 7.2 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.25 (9H, s), 2.40 (2H, t, J= 7.0 Hz), 2.41 (3H, s), 2.99 (2H, t, J= 7.4 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.2 Hz), 1.84 (2H, quint, J = 7.2 Hz), 2.10 (2H, t, J = 7.5 Hz), 2.25 (9H, s), 2.40 (2H, t, J = 7.0 Hz), 2.41 (3H, s), 2.99 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz).
元素分析値: C24H34N403S3として計算値: C, 55.14; H, 6.56; N, 10.72; S, 18.40 (%), 実測値: C, 54.87; H, 6.37; N, 10.80; S, 18.29 (%) . 実施例 4-266 Elemental analysis: C 24 H 34 N 4 0 3 S 3 Calculated: C, 55.14; H, 6.56 ; N, 10.72; S, 18.40 (%), Found: C, 54.87; H, 6.37 ; N, 10.80; S, 18.29 (%). Example 4-266
Ή-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.42 (2H, brquint, J= 7.4 Hz)', 2.07 (2H, t, J= 7.5 Hz), 2.21 (3H, s), 2.41 (3H, s), 3.75 (3H, s), 5.26 (1H, s), 7.26 (2H, d, J- 8.3 Hz), 7.36 (5H, m), 7.83 (2H, d, J= 8.3 Hz), 9.58 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.42 (2H, brquint, J = 7.4 Hz) ', 2.07 (2H, t, J = 7.5 Hz), 2.21 (3H, s), 2.41 (3H, s), 3.75 (3H, s), 5.26 (1H, s), 7.26 (2H, d, J-8.3 Hz), 7.36 (5H, m ), 7.83 (2H, d, J = 8.3 Hz), 9.58 (1H, brs).
元素分析値: C28H31N305S3として計算値: C, 57.41; H, 5.33; N, 7.17; S, 16.42Elemental analysis: C 28 H 31 N 3 0 5 S 3 Calculated: C, 57.41; H, 5.33 ; N, 7.17; S, 16.42
(%) 、 実測値: C, 57.22; H, 5.23; N, 7.39; S, 16.42 ( ) . (%), Found: C, 57.22; H, 5.23; N, 7.39; S, 16.42 ().
実施例 4-267 Example 4-267
'H-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.8 Hz), 0.94 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.45 (2H, brquint, J= 7.3 Hz), 1.66 (2H, quint, J= 7.4 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H; s), 2.95 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.90 (3Η, t, J = 6.8 Hz), 0.94 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.45 (2H, brquint, J = 7.3 Hz), 1.66 (2H, quint, J = 7.4 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H; s), 2.95 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C23H31N303S3として計算値: C, 55.95; H, 6.33; N, 8.51; S, 19.48 (%), 実測値: C, 55.79; H, 6.62; N, 8.62; S, 19.50 (%) . Elemental analysis: C 23 H 31 N 3 0 3 S 3 Calculated: C, 55.95; H, 6.33 ; N, 8.51; S, 19.48 (%), Found: C, 55.79; H, 6.62 ; N, 8.62; S, 19.50 (%).
実施例 4- 268 Example 4-268
【H - NMR (300MHz, CDC13) δ 0.90 (3Η, t, J= 6.8 Hz), 0.91 (3H, t, J= 6.8 Hz), 1.31 (4H, m), 1.36 (4H, m), 1.47 (2H, brquint, J= 7.4 Hz), 1.68 (2H, quint, J= 7.3 Hz), 2.11 (2H, t, J= 7.7 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J- 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). [H - NMR (300MHz, CDC1 3 ) δ 0.90 (3Η, t, J = 6.8 Hz), 0.91 (3H, t, J = 6.8 Hz), 1.31 (4H, m), 1.36 (4H, m), 1.47 (2H, brquint, J = 7.4 Hz), 1.68 (2H, quint, J = 7.3 Hz), 2.11 (2H, t, J = 7.7 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J- 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C24H33N303S3として計算値: C, 56.77; H, 6.55; N, 8.28; S, 18.95Elemental analysis: Calculated as C 24 H 33 N 3 0 3 S 3: C, 56.77; H, 6.55; N, 8.28; S, 18.95
( ), 実測値: C, 56.63; H, 6.65; N, 8.39; S, 18.92 ( ) . (), Found: C, 56.63; H, 6.65; N, 8.39; S, 18.92 ().
実施例 4-269 Example 4-269
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.8 Hz), 0.90 (3H, t, J= 6.8 Hz), 1.31 (10H, m), 1.37 (3H, d, J= 6.8 Hz), 1.47 (2H, brquint, J- 7.3 Hz), 1.65 (2H, m), 2.12 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.46 (1H, tq, J= 6.8, 6.8 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.63 (1H, brs) . Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J = 6.8 Hz), 0.90 (3H, t, J = 6.8 Hz), 1.31 (10H, m), 1.37 (3H, d, J = 6.8 Hz), 1.47 (2H, brquint, J- 7.3 Hz), 1.65 (2H, m), 2.12 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.46 (1H, tq, J = 6.8, 6.8 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C26H37N303S3として計算値: C, 58.28; H, 6.96; N, 7.84; S, 17.95 (%) 、 実測値: C, 58.43; H, 6.96; N, 7.64; S, 17.79 (%) . 実施例 4-270 Elemental analysis: C 26 H 37 N 3 0 3 S 3 Calculated: C, 58.28; H, 6.96 ; N, 7.84; S, 17.95 (%), Found: C, 58.43; H, 6.96; N, 7.64; S, 17.79 (%).
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 0.90 (3Η, t, J= 7.2 Hz), 0.97 (6H, t, J= 7.5 Hz), 1.33 (8H, m), 1.47 (2H, brquint, J= 7.2 Hz), 1.60 (2H, m), 1.67 (2H, m), 2.12 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.33 (1H, quint, J= 6.4 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J- 8.3 Hz), 9.63 (1H, brs). 'H-NMR (300 MHz, in double-mouthed form): δ 0.90 (3Η, t, J = 7.2 Hz), 0.97 (6H, t, J = 7.5 Hz), 1.33 (8H, m), 1.47 (2H , brquint, J = 7.2 Hz), 1.60 (2H, m), 1.67 (2H, m), 2.12 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.33 (1H, quint, J = 6.4 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J-8.3 Hz), 9.63 (1H, brs).
元素分析値: C26H37N303S3 として計算値; C, 58.28; H, 6.96; N, 7.84; S, 17.95Elemental analysis: Calculated as C 26 H 37 N 3 0 3 S 3; C, 58.28; H, 6.96; N, 7.84; S, 17.95
(%)、 実測値; C, 58.04; H, 7.12; N, 7.80; S, 17.94 (%) . (%), Found; C, 58.04; H, 7.12; N, 7.80; S, 17.94 (%).
実施例 4-271 Example 4-271
Ή-N R (300MHz, CDC13) : 6 0.90 (3H, t, J= 6.8 Hz), 0.92 (6H, t, J= 7.3 Hz), 1.31 (4H, m), 1.44 (6H, m), 1.61 (4H, m), 2.12 (2H, t, J- 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.40 (1H, quint, J= 6.4 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). ■ 元素分析値: C26H37N303S3として計算値: C, 58.28; H, 6.96; N, 7.84; S, 17.95 Ή-NR (300MHz, CDC1 3 ): 6 0.90 (3H, t, J = 6.8 Hz), 0.92 (6H, t, J = 7.3 Hz), 1.31 (4H, m), 1.44 (6H, m), 1.61 (4H, m), 2.12 (2H, t, J- 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 3.40 (1H, quint, J = 6.4 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.63 (1H, brs). ■ Elemental analysis: Calculated as C 26 H 37 N 3 0 3 S 3 : C, 58.28; H, 6.96; N, 7.84; S, 17.95
(%)、 実測値: C, 58.26; H, 6.88; N, 7.58; S, 17.78 (%) . (%), Found: C, 58.26; H, 6.88; N, 7.58; S, 17.78 (%).
実施例 4-272 Example 4-272
LH-NMR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 6.6 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 2.04 (2H, quint, J= 7.0 Hz), 2.12 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.43 (3H, s), 2.48 (2H, t, J= 7.0 Hz), 3.07 (2H, t, J= 7.0 Hz), 7.30 (2H, d, J= 7.9 Hz), 7.84 (2H, d, J= 7.9 Hz), 9.67 (1H, brs). L H-NMR (300MHz, CDC1 3): δ 0.91 (3Η, t, J = 6.6 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 2.04 (2H, quint, J = 7.0 Hz), 2.12 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.43 (3H, s), 2.48 (2H, t, J = 7.0 Hz), 3.07 (2H, t, J = 7.0 Hz), 7.30 (2H, d, J = 7.9 Hz), 7.84 (2H, d, J = 7.9 Hz), 9.67 (1H, brs).
元素分析値: C23H28N403S3として計算値: C, 54.74; H, 5.59; N, 11.10; S, 19.06Elemental analysis: Calculated for C 23 H 28 N 4 0 3 S 3 : C, 54.74; H, 5.59; N, 11.10; S, 19.06
( ) 、 実測値: C, 54.78; H, 5.60; N, 11.15; S, 19.21 (%) . (), Found: C, 54.78; H, 5.60; N, 11.15; S, 19.21 (%).
実施例 4-273 Example 4-273
Ή-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.3 Hz), 1.82 (4H, m), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.38 (2H, t, J= 6.6 Hz), 2.42 (3H, s), 2.98 (2H, t, J= 7.0 Hz), 7.30 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.3 Hz), 1.82 (4H, m), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.38 (2H, t, J = 6.6 Hz), 2.42 (3H, s), 2.98 (2H, t, J = 7.0 Hz), 7.30 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.64 (1H, brs).
元素分析値: C24H3。N403S3として計算値: C, 55.57; H, 5.83; N, 10.80; S, 18.55 実測値: C, 55.37; H, 5.70; N, 10.74; S, 18.45 (%) . 実施例 4-274 Elemental analysis: C 24 H 3. Calculated for N 4 0 3 S 3 : C, 55.57; H, 5.83; N, 10.80; S, 18.55 Found: C, 55.37; H, 5.70; N, 10.74; S, 18.45 (%).
'H-NMR (300MHz, CDC13) : <5 0.91 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.2 Hz), 1.60 (2H, m), 1.70 (4H, m), 2.11 (2H, brt, J= 7.5 Hz), 2.26 (3H, s), 2.37 (2H, t, J= 6.8 Hz), 2.42 (3H, s), 2.96 (2H, t, J= 7.2 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.65 (1H, brs). 'H-NMR (300MHz, CDC1 3): <5 0.91 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.2 Hz), 1.60 (2H, m) , 1.70 (4H, m), 2.11 (2H, brt, J = 7.5 Hz), 2.26 (3H, s), 2.37 (2H, t, J = 6.8 Hz), 2.42 (3H, s), 2.96 (2H, t, J = 7.2 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.65 (1H, brs).
元素分析値: C25H32N403S3として計算値: C, 56.36; H, 6.05; N, 10.52; S, 18.06Elemental analysis: C 25 H 32 N 4 0 3 S 3 Calculated: C, 56.36; H, 6.05 ; N, 10.52; S, 18.06
(%)、 実測値: C, 56.24; H, 5.91; , 10.53; S, 18.06 (%) . (%), Found: C, 56.24; H, 5.91;, 10.53; S, 18.06 (%).
実施例 4-275 Example 4-275
lH-NMR (300MHz, CDC13) : δ 0.91 (3H, t, J= 6.6 Hz), 1.32 (4H, m), 1.47 (6H, m), 1.69 (4H, m), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.36 (2H, t, J= 7.0 Hz), 2.42 (3H, s), 2.95 (2H, t, J= 7.4 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). lH-NMR (300MHz, CDC1 3 ): δ 0.91 (3H, t, J = 6.6 Hz), 1.32 (4H, m), 1.47 (6H, m), 1.69 (4H, m), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.36 (2H, t, J = 7.0 Hz), 2.42 (3H, s), 2.95 (2H, t, J = 7.4 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.64 (1H, brs).
元素分析値: C26H34N403S3として計算値: C, 57.11; H, 6.27; N, 10.25; S, 17.59 {%) 、 実測値: C, 57.13; H, 6.13; N, 10.28; S, 17.71 ( ) . Elemental analysis: C 26 H 34 N 4 0 3 S 3 Calculated: C, 57.11; H, 6.27; N, 10.25; S, 17.59 (%), found: C, 57.13; H, 6.13; N, 10.28; S, 17.71 ().
実施例 4-276 Example 4-276
Ή- NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J- 6.8 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J= 7.5 Hz), 2.12 (2H, t, J= 7.7 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.67 (1H, dd, J= 4.9, 1.9 Hz), 2.85 (1H, t, J= 4.3 Hz), 3.06 (1H, dd, J= 13.2, 4.5 Hz), 3.19 (1H, m), 3.24 (1H, dd, J= 13.2, 4.9 Hz) , 7.30 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.60 (1H, brs). Ή- NMR (300MHz, CDC1 3) : δ 0.90 (3Η, t, J- 6.8 Hz), 1.31 (4H, m), 1.47 (2H, brquint, J = 7.5 Hz), 2.12 (2H, t, J = 7.7 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.67 (1H, dd, J = 4.9, 1.9 Hz), 2.85 (1H, t, J = 4.3 Hz), 3.06 (1H, dd, J = 13.2, 4.5 Hz), 3.19 (1H, m), 3.24 (1H, dd, J = 13.2, 4.9 Hz), 7.30 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.60 (1H, brs).
元素分析値: C22H27N304S3として計算値: C, 53.53; H, 5.51; , 8.51; S, 19.49Elemental analysis: Calculated as C 22 H 27 N 3 0 4 S 3: C, 53.53; H, 5.51;, 8.51; S, 19.49
(%)ゝ 実測値: C, 53.28; H, 5.37; N, 8.39; S, 19.34 (%) . (%) ゝ Found: C, 53.28; H, 5.37; N, 8.39; S, 19.34 (%).
実施例 4-277 Example 4-277
Ή-NMR (300MHz, CDC13) :- δ 0.9'1 (3Η, t, J- 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.2 Hz), 1.73 (4H, m), 2.09 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.39 (3H, s), 2.64 (2H, t, J= 7.0 Hz), 2.95 (2H, t, J= 7.0 Hz), 7.16 (2H, d, J= 7.5 Hz), 7.20 (1H, t, J- 7.5 Hz), 7.24 (2H, d, J= 8.3 Hz), 7.29 (2H, t, J= 7.5 Hz), 7.84 (2H, dt, J= 8.3, 2.2 Hz), 9.62 (1H, brs). 元素分析値: C29H35N303S3として計算値: C, 61.13; H, 6.19; N, 7.37 (%) 、 実測 値: C, 60.83; H, 5.99; N, 7.27 (%) . Ή-NMR (300MHz, CDC1 3 ): - δ 0.9'1 (3Η, t, J- 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.2 Hz), 1.73 (4H, m ), 2.09 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.39 (3H, s), 2.64 (2H, t, J = 7.0 Hz), 2.95 (2H, t, J = 7.0 Hz) ), 7.16 (2H, d, J = 7.5 Hz), 7.20 (1H, t, J- 7.5 Hz), 7.24 (2H, d, J = 8.3 Hz), 7.29 (2H, t, J = 7.5 Hz), 7.84 (2H, dt, J = 8.3, 2.2 Hz), 9.62 (1H, brs). Elemental analysis: C 29 H 35 N 3 0 3 S 3 Calculated: C, 61.13; H, 6.19; N, 7.37 (%), found: C, 60.83; H, 5.99; N, 7.27 (%) .
実施例 4-278 Example 4-278
Ή-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.8 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J= 7.3 Hz), 2.11 (2H, t, J= 7.7 Hz), 2.26 (3H, s), 2.40 (3H, s), 3.34 (2H, t, J= 6.6 Hz), 4.18 (2H, t, J= 6.8 Hz), 6.89 (2H, d, J= 7.9 Hz), 6.99 (1H, t, J= 7.3 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.30 (2H, i, J= 8.0 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3Η, t, J = 6.8 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J = 7.3 Hz), 2.11 (2H, t, J = 7.7 Hz), 2.26 (3H, s), 2.40 (3H, s), 3.34 (2H, t, J = 6.6 Hz), 4.18 (2H, t, J = 6.8 Hz), 6.89 (2H, d, J = 7.9 Hz), 6.99 (1H, t, J = 7.3 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.30 (2H, i, J = 8.0 Hz), 7.85 (2H, d, J = 8.3 Hz) ), 9.64 (1H, brs).
元素分析値: C27H31N304S3として計算値: C, 58.14; H, 5.60; N, 7.53; S, 17.25 (%) 、 実測値: C, 57.99; H, 5.58; N, 7.60; S, 17.37 (%) . Elemental analysis: C 27 H 31 N 3 0 4 S 3 Calculated: C, 58.14; H, 5.60 ; N, 7.53; S, 17.25 (%), Found: C, 57.99; H, 5.58 ; N, 7.60; S, 17.37 (%).
実施例 4-279 Example 4-279
¾-NMR (300MHz, CDC13) : δ 0.88 (3H, t, J= 6.8 Hz), 1.28 (4H, i), 1.45 (2H, brquint, J= 7.2 Hz) , 2.10 (2H, t, J= 7.2 Hz), 2.26 (3H, s), 2.40 (3H, s), 3.38 (4H, s), 7.26 (2H, d, J= 7.9 Hz), 7.50 (2H, t, J= 7.5 Hz), 7.61 (1H, t, J= 7.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 7.96 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). 元素分析値: C28H31N304S3 · 0.2 0として計算値: C, 58.65; H, 5.52; N, 7.32 ( )、 実測値: C, 58.64; H, 5.47; N, 7.06 (%) · ¾-NMR (300MHz, CDC1 3 ): δ 0.88 (3H, t, J = 6.8 Hz), 1.28 (4H, i), 1.45 (2H, brquint, J = 7.2 Hz), 2.10 (2H, t, J = 7.2 Hz), 2.26 (3H, s), 2.40 (3H, s), 3.38 (4H, s), 7.26 (2H, d, J = 7.9 Hz), 7.50 (2H, t, J = 7.5 Hz), 7.61 (1H, t, J = 7.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 7.96 (2H, d, J = 8.3 Hz), 9.63 (1H, brs). Elemental analysis: C 28 H 31 N 3 0 4 S 3 · 0.2 0 calculated: C, 58.65; H, 5.52 ; N, 7.32 (), Found: C, 58.64; H, 5.47 ; N, 7.06 (%) ·
実施例 4-280 Example 4-280
¾-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.8 Hz), 1.30 (4H, m), 1.45 (2H, brquint, J= 7.4 Hz), 2.09 (2H, t, J= 7.5 Hz), 2.15 (2H, quint, J= 7.0 Hz), 2.25 (3H, s), 2.38 (3H, s), 3.09 (2H, t, J= 7.2 Hz), 3.14 (2H, t, J= 6.8 Hz), 7.24 (2H, d, J= 8.3 Hz), 7.48 (2H, i, J= 7.5 Hz), 7.59 (1H, tt, J= 7.3, 2.0 Hz), 7.84 (2H, d, J= 8.3 Hz), 7.96 (2H, d, J= 7.9 Hz), 9.64 (1H, brs). 元素分析値: C29¾3N304S3として計算値: C, 59.66; H, 5.70; N, 7.20; S, 16.48 (%), 実測値: C, 59.81; H, 5.70; N, 7.15; S, 16.21 (%) . ¾-NMR (300MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 6.8 Hz), 1.30 (4H, m), 1.45 (2H, brquint, J = 7.4 Hz), 2.09 (2H, t, J = 7.5 Hz), 2.15 (2H, quint, J = 7.0 Hz), 2.25 (3H, s), 2.38 (3H, s), 3.09 (2H, t, J = 7.2 Hz), 3.14 (2H, t, J = 6.8 Hz), 7.24 (2H, d, J = 8.3 Hz), 7.48 (2H, i, J = 7.5 Hz), 7.59 (1H, tt, J = 7.3, 2.0 Hz), 7.84 (2H, d, J = . 8.3 Hz), 7.96 (2H , d, J = 7.9 Hz), 9.64 (1H, brs) elemental analysis: C 29 calculated ¾ 3 N 3 0 4 S 3 : C, 59.66; H, 5.70; N , 7.20; S, 16.48 (%), found: C, 59.81; H, 5.70; N, 7.15; S, 16.21 (%).
実施例 4-281 Example 4-281
'H-NMR (300MHz, CDC13) : δ 0.86 (3Η, t, J= 6.8 Hz), 1.25 (4H, m), 1.38 (2H, brquint, J- 7.2 Hz), 2.03 (2H, t, J= 7.5 Hz), 2.23 (3H, s), 2.40 (3H, s), 4.46 (2H, s), 7.26 (2H, d, J= 8.3 Hz), 7.51 (2H, t, J= 7.5 Hz), 7.64 (1H, tt, J= 7.5, 2.0 Hz), 7.85 (2H, d, J= 8.3 Hz), 7.96 (2H, d, J= 7.9 Hz), 9.63 (1H, brs). 元素分析値: C27H29N304S3として計算値: C, 58.35; H, 5.26; N, 7.56; S, 17.31'H-NMR (300MHz, CDC1 3): δ 0.86 (3Η, t, J = 6.8 Hz), 1.25 (4H, m), 1.38 (2H, brquint, J- 7.2 Hz), 2.03 (2H, t, J = 7.5 Hz), 2.23 (3H, s), 2.40 (3H, s), 4.46 (2H, s), 7.26 (2H, d, J = 8.3 Hz), 7.51 (2H, t, J = 7.5 Hz), 7.64 (1H, tt, J = 7.5, 2.0 Hz), 7.85 ( 2H, d, J = 8.3 Hz), 7.96 (2H, d, J = 7.9 Hz), 9.63 (1H, brs) Elemental analysis:. C 27 H 29 N 3 0 4 S Calculated as 3 : C, 58.35; H, 5.26; N, 7.56; S, 17.31
(%) 、 実測値: C, 58.45; H, 5.26; N, 7.54; S, 17.11 (%) . (%), Found: C, 58.45; H, 5.26; N, 7.54; S, 17.11 (%).
実施例 4-282 Example 4-282
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J= 7.3 Hz), 2.11 (2H, i, J= 7.7 Hz), 2.26 (3H, s), 2.40 (3H, s), 3.34 (2H, t, J= 6.6 Hz), 4.18 (2H, t, J= 6.8 Hz), 6.89 (2H, d, J= 7.9 Hz), 6.99 (1H, t, J= 7.3 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.30 (2H, t, J= 8.0 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.46 (2H, brquint, J = 7.3 Hz), 2.11 (2H, i, J = 7.7 Hz), 2.26 (3H, s), 2.40 (3H, s), 3.34 (2H, t, J = 6.6 Hz), 4.18 (2H, t, J = 6.8 Hz), 6.89 (2H, d, J = 7.9 Hz), 6.99 (1H, t, J = 7.3 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.30 (2H, t, J = 8.0 Hz), 7.85 (2H, d, J = 8.3 Hz) ), 9.64 (1H, brs).
元素分析値: C27H31N304S3として計算値: C, 58.14; H, 5.60; N, 7.53; S, 17.25 {%), 実測値: C, 57.99; H, 5.58; N, 7.60; S, 17.37 (%) . Elemental analysis: C 27 H 31 N 3 0 Calculated 4 S 3: C, 58.14; H, 5.60; N, 7.53; S, 17.25 {%), Found: C, 57.99; H, 5.58 ; N, 7.60; S, 17.37 (%).
実施例 4-283 Example 4-283
¾-NMR (300MHz, CDC13) : <5 0.91 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.42 (2H, brquint, J= 7.2 Hz), 2.05 (2H, t, J= 7.5 Hz), 2.24 (3H, s), 2.41 (3H, s), 3.12 (4H, s), 7.26-7.35 (5H, m), 7.41 (2H, brdt, J= 6.8, 1.5 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). ¾-NMR (300MHz, CDC1 3 ): <5 0.91 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.42 (2H, brquint, J = 7.2 Hz), 2.05 (2H, t, J = 7.5 Hz), 2.24 (3H, s), 2.41 (3H, s), 3.12 (4H, s), 7.26-7.35 (5H, m), 7.41 (2H, brdt, J = 6.8, 1.5 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C27H31N303S4として計算値: C, 56.51; H, 5.45; , 7.32; S, 22.35Elemental analysis: Calculated as C 27 H 31 N 3 0 3 S 4: C, 56.51; H, 5.45;, 7.32; S, 22.35
(%), 実測値: C, 56.58; H, 5.47; N, 7.25; S, 22.40 ( ) . (%), Found: C, 56.58; H, 5.47; N, 7.25; S, 22.40 ().
実施例 4-284 Example 4-284
'H-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J= 7.3 Hz), 1.66 (1H, m), 1.91 (2H, quint, J= 7.0 Hz), 2.05 (1H, m), 2.10 (2H, t, J= 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.02 (1H, dd, J= 13.2, 6.0 Hz), 3.18 (1H, dd, J- 13.2, 5.7 Hz), 3.78 (1H, dt, J= 8.3, 6.8 Hz), 3.87 (1H, dt, J= 8.3, 6.8 Hz), 4.10 (1H, quint, J= 6.4 Hz), 7.28 (2H, d, J- 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz) , 9.63 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.90 (3H, t, J = 6.6 Hz), 1.31 (4H, m), 1.46 (2H, brquint, J = 7.3 Hz), 1.66 (1H, m), 1.91 (2H, quint, J = 7.0 Hz), 2.05 (1H, m), 2.10 (2H, t, J = 7.5 Hz), 2.25 (3H, s), 2.41 (3H, s), 3.02 (1H, dd , J = 13.2, 6.0 Hz), 3.18 (1H, dd, J- 13.2, 5.7 Hz), 3.78 (1H, dt, J = 8.3, 6.8 Hz), 3.87 (1H, dt, J = 8.3, 6.8 Hz) , 4.10 (1H, quint, J = 6.4 Hz), 7.28 (2H, d, J- 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C24H31N304S3として計算値: C, 55.25; H, 5.99; , 8.05; S, 18.44Elemental analysis: Calculated as C 24 H 31 N 3 0 4 S 3: C, 55.25; H, 5.99;, 8.05; S, 18.44
(%) 、 実測値: C, 55.12; H, 5.91; , 8.13; S, 18.70 (%) . (%), Found: C, 55.12; H, 5.91;, 8.13; S, 18.70 (%).
実施例 4-285 Example 4-285
Ή -麵 R (300MHz, CDCi3) : δ 0.91 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J= 7.2 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.41 (3H, s), 2.46 (3H, s), 7.28Ή-麵 R (300MHz, CDCi 3 ): δ 0.91 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.47 (2H, brquint, J = 7.2 Hz), 2.10 (2H, t, J = 7.5 Hz), 2.41 (3H, s), 2.46 (3H, s), 7.28
(2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). (2H, d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C2。H25N303S3として計算値: C, 53.19; H, 5.58; N, 9.30; S, 21.30Elemental analysis: C 2. Calculated for H 25 N 3 0 3 S 3 : C, 53.19; H, 5.58; N, 9.30; S, 21.30
(%), 実測値: C, 53.20; H, 5.47; N, 9.24; S, 21.27 ( ) . (%), Found: C, 53.20; H, 5.47; N, 9.24; S, 21.27 ().
実施例 4- 286 t Example 4-286 t
¾-NMR (300 MHz, 重クロ口ホルム中): δ 0.90 (3Η, t, J= 6.8 Hz), 1.31 (4H, ' m), 1.36 (3H, t, J= 7.4 Hz), 1.47 (2H, brquint, J= 7.2 Hz), 2.11 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, q, J= 7.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.64 (1H, brs). ¾-NMR (300 MHz, in double-mouthed form): δ 0.90 (3Η, t, J = 6.8 Hz), 1.31 (4H, 'm), 1.36 (3H, t, J = 7.4 Hz), 1.47 (2H , brquint, J = 7.2 Hz), 2.11 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, q, J = 7.5 Hz), 7.28 (2H , d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.64 (1H, brs).
元素分析値: C21H27N303S3 として計算値; C, 54.17; H, 5.84; N, 9.02; S, 20.66Elemental analysis: Calculated as C 21 H 27 N 3 0 3 S 3; C, 54.17; H, 5.84; N, 9.02; S, 20.66
(%)、 実測値; C, 54.12; H, 5.55; N, 8.87; S, 20.64 (%) . (%), Found; C, 54.12; H, 5.55; N, 8.87; S, 20.64 (%).
実施例 4-287 Example 4-287
Ή-NMR (300MHz, CDC13) : 6 0.90 (3H, t, J= 6.8 Hz), 1.32 (4H, m), 1.39 (6H, d, J= 6.8 Hz), 1.48 (2H, brquint, J= 7.4 Hz), 2.12 (2H, t, J= 7.7 Hz), 2.26 (3H, s), 2.42 (3H, s), 3.62 (1H, sept, J= 6.8 Hz), 7.28 (2H, d, ]= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.66 (1H, brs). - 元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05 Ή-NMR (300MHz, CDC1 3 ): 6 0.90 (3H, t, J = 6.8 Hz), 1.32 (4H, m), 1.39 (6H, d, J = 6.8 Hz), 1.48 (2H, brquint, J = 7.4 Hz), 2.12 (2H, t, J = 7.7 Hz), 2.26 (3H, s), 2.42 (3H, s), 3.62 (1H, sept, J = 6.8 Hz), 7.28 (2H, d,] = . 8.3 Hz), 7.86 (2H , d, J = 8.3 Hz), 9.66 (1H, brs) - elemental analysis: calculated C 22 H 29 N 3 0 3 S 3: C, 55.09; H, 6.09; N, 8.76; S, 20.05
(%) 、 実測値: C, 55.09; H, 6.03; N, 8.71; S, 19.92 (%) . (%), Found: C, 55.09; H, 6.03; N, 8.71; S, 19.92 (%).
実施例 4-288 Example 4-288
'H-NMR (300MHz, CDC13) : 6 0.91 (3H, t, J= 7.0 Hz), 1.03 (3H, t, J= 7.4 Hz), 1.32 (4H, in), 1.47 (2H, brquint, J= 7.2 Hz), 1.79 (3H, tq, J= 7.3, 7.5 Hz), 2.11 (2H, t, J- 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.92 (2H, t, J= 7.3 Hz), 7.28 (2H, d, J- 8.3 Hz), 7.85 (2H, dt, J= 8.3, 1.5 Hz), 9.62 (1H, brs). 元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05 (%), 実測値: (;, 55.10; H, 6.01; , 8.76; S, 20.04 (%) . 'H-NMR (300MHz, CDC1 3): 6 0.91 (3H, t, J = 7.0 Hz), 1.03 (3H, t, J = 7.4 Hz), 1.32 (4H, in), 1.47 (2H, brquint, J = 7.2 Hz), 1.79 (3H, tq, J = 7.3, 7.5 Hz), 2.11 (2H, t, J-7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.92 (2H, t , J = 7.3 Hz), 7.28 (2H, d, J- 8.3 Hz), 7.85 (2H, dt, J = 8.3, 1.5 Hz), 9.62 (1H, brs). Elemental analysis: C 22 H 29 N 3 0 3 S 3 calculated: C, 55.09; H, 6.09 ; N, 8.76; S, 20.05 (%), Found: (;, 55.10; H, 6.01;, 8.76; S, 20.04 (%).
実施例 4-289 Example 4-289
Ή-NMR (300MHz, CDC13) : δ 0.91 (3Η, t, J= 6.8 Hz), 1.31 (4H, m), 1.45 (2H, brquint, J= 7.3 Hz), 1.72 (1H, m), 1.84 (1H, m), 1.87 (1H, brt, J= 3.4 Hz), 1.91 (2H, m), 2.09 (2H, brt, J= 7.8 Hz), 2.25 (3H, s), 2.39 (3H, s), 2.97 (2H, dt, J= 2.3, 7.0 Hz) , 4.70 (1H, brt, J= 4.0 Hz) , 7.24 (2H, d, J= 8.3 Hz) , 7.34 (5H, m), 7.83 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.91 (3Η, t, J = 6.8 Hz), 1.31 (4H, m), 1.45 (2H, brquint, J = 7.3 Hz), 1.72 (1H, m), 1.84 (1H, m), 1.87 (1H, brt, J = 3.4 Hz), 1.91 (2H, m), 2.09 (2H, brt, J = 7.8 Hz), 2.25 (3H, s), 2.39 (3H, s) , 2.97 (2H, dt, J = 2.3, 7.0 Hz), 4.70 (1H, brt, J = 4.0 Hz), 7.24 (2H, d, J = 8.3 Hz), 7.34 (5H, m), 7.83 (2H, d, J = 8.3) Hz), 9.63 (1H, brs).
元素分析値: C29H35N304S3として計算値: C, 59.46; H, 6.02; , 7.17; S, 16.42 (%)、 実測値: C, 59.42; H, 6.16; N, 7.33; S, 16.41 (%) . Elemental analysis: Calculated as C 29 H 35 N 3 0 4 S 3: C, 59.46; H, 6.02;, 7.17; S, 16.42 (%), Found: C, 59.42; H, 6.16 ; N, 7.33 ; S, 16.41 (%).
実施例 4-290 Example 4-290
Ή-NMR (300MHz, CDC13) : d 0.81 (3H, t, J= 7.0 Hz), 1.13 (4H, m), 1.34 (2H, quint, J= 7.3 Hz), 1.92 (2H, dd, J= 8.0, 7.3 Hz), 2.42 (3H, s), 4.26 (2H, s), 7.15 (2H, m), 7.26 (2H, d, J= 8.3 Hz), 7.35 (5H, s), 7.38 (3H, m), 7.83 (2H, d, J= 8.3 Hz), 9.49 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): d 0.81 (3H, t, J = 7.0 Hz), 1.13 (4H, m), 1.34 (2H, quint, J = 7.3 Hz), 1.92 (2H, dd, J = 8.0, 7.3 Hz), 2.42 (3H, s), 4.26 (2H, s), 7.15 (2H, m), 7.26 (2H, d, J = 8.3 Hz), 7.35 (5H, s), 7.38 (3H, s) m), 7.83 (2H, d, J = 8.3 Hz), 9.49 (1H, brs).
元素分析値: C31H31N303S3として計算値: C, 63.13; H, 5.30; N, 7.12; S, 16.31 (%) 、 実測値: C, 63.03; H, 5.19; N, 7.00; S, 16.13 (%) . Elemental analysis: C 31 H 31 N 3 0 3 S 3 Calculated: C, 63.13; H, 5.30; N, 7.12; S, 16.31 (%), found: C, 63.03; H, 5.19; N, 7.00; S, 16.13 (%).
実施例 4-291 Example 4-291
¾-NMR (300MHz, CDC13) : 6 0.89 (3H, t, J= 6.8 Hz), 1.28 (6H, brs), 1.46 (2H, brquint, J= 7.2 Hz), 2.07 (2H, brt, J= 7.2 Hz), 2.42 (3H, s), 2.78 (2H, t, J= 7.2 Hz), 3.24 (2H, t, J= 7.2 Hz), 6.50 (1H, brt, J= 1 Hz), 7.30 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.7 (1H, br). ¾-NMR (300MHz, CDC1 3 ): 6 0.89 (3H, t, J = 6.8 Hz), 1.28 (6H, brs), 1.46 (2H, brquint, J = 7.2 Hz), 2.07 (2H, brt, J = 7.2 Hz), 2.42 (3H, s), 2.78 (2H, t, J = 7.2 Hz), 3.24 (2H, t, J = 7.2 Hz), 6.50 (1H, brt, J = 1 Hz), 7.30 (2H , d, J = 8.3 Hz), 7.84 (2H, d, J = 8.3 Hz), 9.7 (1H, br).
実施例 4-292 Example 4-292
Ή-NMR (300MHz, CDC13) : 6 0.89 (3H, t, J= 6.8 Hz), 1.28 (6H, brs), 1.45 (2H, brquint, J= 7.5 Hz), 2.04 (2H, brt, J= 7.2 Hz), 2.39 (3H, s), 2.59 (2H, t, J= 7.5 Hz), 3.30 (2H, t, J= 7.5 Hz), 6.15 (1H, brt, J= 5.7 Hz), 6.46 (1H, s), 7.27 (7H, m), 7.80 (2H, d, J= 8.3 Hz). Ή-NMR (300MHz, CDC1 3 ): 6 0.89 (3H, t, J = 6.8 Hz), 1.28 (6H, brs), 1.45 (2H, brquint, J = 7.5 Hz), 2.04 (2H, brt, J = 7.2 Hz), 2.39 (3H, s), 2.59 (2H, t, J = 7.5 Hz), 3.30 (2H, t, J = 7.5 Hz), 6.15 (1H, brt, J = 5.7 Hz), 6.46 (1H , s), 7.27 (7H, m), 7.80 (2H, d, J = 8.3 Hz).
実施例 4-293 Example 4-293
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, brt, J= 6.9 Hz), 1.28 (4H, m), 1.31 (2H, in), 1.46 (2H, brquint, J= 7.1 Hz), 1.61 (3H, s), 1.69 (3H, s), 1.69 (3H, s), 2.06 (4H, brs), 2.06 (2H, brt, J= 7.1 Hz), 2.41 (3H, s), 3.66 (2H, d, J= 7.6 Hz), 5.07 (1H, brt, J= 5.9 Hz), 5.24 (1H, brt, J= 7.6 Hz), 6.50 (1H, brs), 7.28 (2H, d, J= 8.2 Hz), 7.83 (2H, d, J= 8.2 Hz), 9.73 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, brt, J = 6.9 Hz), 1.28 (4H, m), 1.31 (2H, in), 1.46 (2H, brquint, J = 7.1 Hz), 1.61 (3H, s), 1.69 (3H, s), 1.69 (3H, s), 2.06 (4H, brs), 2.06 (2H, brt, J = 7.1 Hz), 2.41 (3H, s), 3.66 (2H, d, J = 7.6 Hz), 5.07 (1H, brt, J = 5.9 Hz), 5.24 (1H, brt, J = 7.6 Hz), 6.50 (1H, brs), 7.28 (2H, d, J = 8.2 Hz) , 7.83 (2H, d, J = 8.2 Hz), 9.73 (1H, brs).
元素分析値: C29H29N303S3 · 0.2¾0として計算値: C, 60.32; H, 6.88; N, 7.28; S, 16.66 (%) 、 実測値: C, 60.27; H, 6.59; N, 7.26; S, 16.67 (%) . 実施例 4-294 , Elemental analysis: C 29 H 29 N 3 0 3 S 3 · 0.2 計算 0 Calculated: C, 60.32; H, 6.88; N, 7.28; S, 16.66 (%), found: C, 60.27; H, 6.59 ; N, 7.26; S, 16.67 (%). Example 4-294,
¾-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m), 1.44 (2H, brquint, J- 6.9 Hz), 2.04 (2H, brt, J= 7.1 Hz), 2.42 (3H, s), 4.17 (2H, s), 6.47 (1H, brs), 7.21 (1H, m), 7.27 (2H, ), 7.27 (2H, d, J= 8.3 Hz), 7.31 (1H, m), 7.81 (2H, d, J= 8.3 Hz), 9.72 (1H, brs). ¾-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m), 1.44 (2H, brquint, J- 6.9 Hz), 2.04 (2H, brt, J = 7.1 Hz), 2.42 (3H, s), 4.17 (2H, s), 6.47 (1H, brs), 7.21 (1H, m), 7.27 (2H,), 7.27 (2H, d , J = 8.3 Hz), 7.31 (1H, m), 7.81 (2H, d, J = 8.3 Hz), 9.72 (1H, brs).
元素分析値: C26H28N303S3C1として計算値: C, 55.55; H, 5.02; N, 7.47; S, 17.11; C1, 6.31 (%)、 実測値: C, 55.44; H, 4.86; N, 7.45; S, 17.2,8; C1, 6.24 (%) . 実施例 4-295 Elemental analysis: Calculated for C 26 H 28 N 3 0 3 S 3 C1: C, 55.55; H, 5.02; N, 7.47; S, 17.11; C1, 6.31 (%), found: C, 55.44; H , 4.86; N, 7.45; S, 17.2,8; C1, 6.24 (%).
Ή-NMR (300MHz, CDC13) : <5 0.89 (3H, t, J= 6.9 Hz), 1.28 (4H, m), 1.31 (2H, m), 1.43 (2H, brquint, J= 7.0 Hz), 2.04 (2H, brt, J= 7.0 Hz), 2.43 (3H, s), 4.15 (2H, s), 6.46 (1H, brt, J= 0.5 Hz), 7.19 (2H, d, J= 8.4 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.45 (2H, dt, J= 8.4, 1.8 Hz), 7.80 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): <5 0.89 (3H, t, J = 6.9 Hz), 1.28 (4H, m), 1.31 (2H, m), 1.43 (2H, brquint, J = 7.0 Hz), 2.04 (2H, brt, J = 7.0 Hz), 2.43 (3H, s), 4.15 (2H, s), 6.46 (1H, brt, J = 0.5 Hz), 7.19 (2H, d, J = 8.4 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.45 (2H, dt, J = 8.4, 1.8 Hz), 7.80 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C26H28N303S3Brとして計算値: C, 51.48; H, 4.65; N, 6.93; S, 15.86; Br, 13.17 (%), 実測値: C, 51.29; H, 4.43; N, 6.74; S, 15.48; Br, 13.01 (%) . Elemental analysis: C 26 H 28 N 3 0 3 S 3 Br Calculated: C, 51.48; H, 4.65 ; N, 6.93; S, 15.86; Br, 13.17 (%), Found: C, 51.29; H , 4.43; N, 6.74; S, 15.48; Br, 13.01 (%).
実施例 4-296 Example 4-296
Ή-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.9 Hz), 1.27 (4H, m), 1.31 (2H, m), 1.43 (2H, brquint, J= 7.0 Hz), 2.03 (2H, brt, J= 7.0 Hz), 2.42 (3H, s), 4.24 (2H, s), 6.46 (1H, brt, J= 0.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.44 (2H, brd, J= 8.2 Hz), 7.58 (2H, brd, J= 8.2 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.72 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3Η, t, J = 6.9 Hz), 1.27 (4H, m), 1.31 (2H, m), 1.43 (2H, brquint, J = 7.0 Hz), 2.03 (2H, brt, J = 7.0 Hz), 2.42 (3H, s), 4.24 (2H, s), 6.46 (1H, brt, J = 0.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.44 (2H, brd, J = 8.2 Hz), 7.58 (2H, brd, J = 8.2 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.72 (1H, br).
元素分析値: C27H28N303S3F3として計算値: C, 54.44; H, 4.74; N, 7.05; S, 16.15 ( ) 、 実測値: C, 54.33; H, 4.75; .N, 6.94; S, 15.99 (%) · Elemental analysis: C 27 H 28 N 3 0 3 Calculated S 3 F 3: C, 54.44 ; H, 4.74; N, 7.05; S, 16.15 (), Found: C, 54.33; H, 4.75 ;. N, 6.94; S, 15.99 (%)
実施例 4-297 Example 4-297
'H-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.9 Hz), 1.27 (4H, m), 1.31 (2H, m), 1.43 (2H, brquint, J= 6.8 Hz), 2.04 (2H, brt, J= 7.0 Hz), 2.42 (3H, s), 4.24 (2H, s), 6.46 (1H, brt, J= 0.5 Hz), 7.27 (2H, brd, J= 8.3 Hz), 7.47 (1H, q, J= 7.8 Hz), 7.57 (2H, brs), 7.81 (2H, d, J= 8.3 Hz), 9.72 (1H, br). 元素分析値: C„H28N303S3F3として計算値: C, 54.44; H, 4.74; N, 7.05; S, 16.15'H-NMR (300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.9 Hz), 1.27 (4H, m), 1.31 (2H, m), 1.43 (2H, brquint, J = 6.8 Hz), 2.04 (2H, brt, J = 7.0 Hz), 2.42 (3H, s), 4.24 (2H, s), 6.46 (1H, brt, J = 0.5 Hz), 7.27 (2H, brd, J = 8.3 Hz), 7.47 (1H, q, J = 7.8 Hz), 7.57 (2H, brs), 7.81 (2H, d, J = 8.3 Hz), 9.72 (1H, br). Elemental analysis: C „H 28 N 3 0 3 S 3 F 3 Calculated: C, 54.44; H, 4.74; N, 7.05; S, 16.15
(%) 、 実測値: C, 54.28; H, 4.48; N, 7.10; S, 16.12 (%) (%), Found: C, 54.28; H, 4.48; N, 7.10; S, 16.12 (%)
実施例 4-298 Example 4-298
'H-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J= 7.0 Hz), 1.27 (4H, brs), 1.30 (2H, m), 1.43 (2H, brquint, J= 6.9 Hz), 2.04 (2H, brt, J= 7.1 Hz), 2.39 (3H, s), 4.38 (2H, s), 6.46 (1H, brt, J= 0.5 Hz), 7.22 (2H, brd, J= 8.3 Hz), 7.42 (1H, dd, J= 8.4, 1.7 Hz), 7.51 (2H, m), 7.80 (4H, m), 7.81 (2H, d, J= 8.3 Hz), 9.73 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.88 (3Η, t, J = 7.0 Hz), 1.27 (4H, brs), 1.30 (2H, m), 1.43 (2H, brquint, J = 6.9 Hz), 2.04 (2H, brt, J = 7.1 Hz), 2.39 (3H, s), 4.38 (2H, s), 6.46 (1H, brt, J = 0.5 Hz), 7.22 (2H, brd, J = 8.3 Hz), 7.42 (1H, dd, J = 8.4, 1.7 Hz), 7.51 (2H, m), 7.80 (4H, m), 7.81 (2H, d, J = 8.3 Hz), 9.73 (1H, brs).
元素分析値: C3。H31N303S3として計算値: C, 62.36; H, 5.41; , 7.27; S, 16.65 (%), 実測値: C, 62.29; H, 5.21; N, 7.32; S, 16.60 (%) Elemental analysis: C 3. H 31 N 3 0 3 S 3 Calculated: C, 62.36; H, 5.41;, 7.27; S, 16.65 (%), Found: C, 62.29; H, 5.21; N, 7.32; S, 16.60 (% )
実施例 4-299 Example 4-299
Ή-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.8 Hz), 1.30 (4H, brs), 1.32 (2H, m), 1.47 (2H, brquint, J- 7.3 Hz), 2.08 (2H, brt, J= 7.0 Hz), 2.38 (3H, s), 4.70 (2H, s), 6.50 (1H, brt, J= 0.5 Hz), 7.10 (2H, brd, J= 8.3 Hz), 7.44 (1H, dd, J= 8.0, 7.3 Hz), 7.52 (1H, brd, J= 7 Hz), 7.55 (1H, m), 7.55 (1H, d, J= 8.6 Hz) , 7.69 (2H, d, J= 8.3 Hz), 7.86 (1H, d, J= 8.1 Hz), 7.91 (1H, m), 8.02 (1H, m), 9.69 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3Η, t, J = 6.8 Hz), 1.30 (4H, brs), 1.32 (2H, m), 1.47 (2H, brquint, J- 7.3 Hz), 2.08 (2H, brt, J = 7.0 Hz), 2.38 (3H, s), 4.70 (2H, s), 6.50 (1H, brt, J = 0.5 Hz), 7.10 (2H, brd, J = 8.3 Hz), 7.44 (1H, dd, J = 8.0, 7.3 Hz), 7.52 (1H, brd, J = 7 Hz), 7.55 (1H, m), 7.55 (1H, d, J = 8.6 Hz), 7.69 (2H, d, J = 8.3 Hz), 7.86 (1H, d, J = 8.1 Hz), 7.91 (1H, m), 8.02 (1H, m), 9.69 (1H, brs).
実施例 4-300 Example 4-300
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.9 Hz), 1.27 (4H, m), 1.29 (2H, m), 1.43 (2H, brquint, J= 7.3 Hz), 2.04 (2H, brt, J= 7.2 Hz), 2.42 (3H, s), 4.40 (2H, s), 6.45 (1H, brt, J= 0.5 Hz), 7.29 (2H, brd, J= 8.3 Hz), 7.43 (1H, ddd, J= 7.7, 6.6, 2.2 Hz), 7.58 (1H, d, J= 7.4 Hz), 7.58 (1H, d, J= 7.4 Hz), 7.59 (1H, m), 7.67 (1H, brd, J= 7.7 Hz), 7.82 (2H, d, J= 8.3 Hz), 9.72 (1H, brs) . Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.9 Hz), 1.27 (4H, m), 1.29 (2H, m), 1.43 (2H, brquint, J = 7.3 Hz), 2.04 (2H, brt, J = 7.2 Hz), 2.42 (3H, s), 4.40 (2H, s), 6.45 (1H, brt, J = 0.5 Hz), 7.29 (2H, brd, J = 8.3 Hz), 7.43 (1H, ddd, J = 7.7, 6.6, 2.2 Hz), 7.58 (1H, d, J = 7.4 Hz), 7.58 (1H, d, J = 7.4 Hz), 7.59 (1H, m), 7.67 (1H, brd, J = 7.7 Hz), 7.82 (2H, d, J = 8.3 Hz), 9.72 (1H, brs).
元素分析値: C27H28N403S3 · 0.5H20として計算値: C, 57.73; H, 5.20; N, 9.97; S, 17.12 (%), 実測値: C, 57.69; H, 5.11; , 10.14; S, 17.06 (%) . Elemental analysis: C 27 H 28 N 4 0 3 S 3 · 0.5 H 2 0 Calculated: C, 57.73; H, 5.20; N, 9.97; S, 17.12 (%), found: C, 57.69; H , 5.11;, 10.14; S, 17.06 (%).
実施例 4-301 Example 4-301
'H-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.9 Hz), 1.24 (9H, s), 1.29 (4H, m), 1.32 (2H, m), 1.47 (2H, brquint, J= 7.3 Hz), 2.07 (2H, brt, J= 7.2 Hz), 2.40 (3H, s), 4.28 (2H, s), 6.49 (1H, brt, J= 0.5 Hz), 7.26 (2H, brd, J= 8.1 Hz), 7.28 (2H, di, . J= 8.2 Hz), 7.34 (2H, dm, J= 8.2 Hz), 7.35 (1H, brd, J= 8.0 Hz), 7.41 (1H, dt, J= 7.6, 1.3 Hz), 7.50 (1H, dt, J= 7.8, 1.5 Hz), 7.80 (2H, d, J= 8.1 Hz), 7.80 (1H, d, J= 8 Hz), 9.75 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.89 (3H, t, J = 6.9 Hz), 1.24 (9H, s), 1.29 (4H, m), 1.32 (2H, m), 1.47 (2H, brquint , J = 7.3 Hz), 2.07 (2H, brt, J = 7.2 Hz), 2.40 (3H, s), 4.28 (2H, s), 6.49 (1H, brt, J = 0.5 Hz), 7.26 (2H, brd, J = 8.1 Hz), 7.28 (2H, di,. J = 8.2 Hz) , 7.34 (2H, dm, J = 8.2 Hz), 7.35 (1H, brd, J = 8.0 Hz), 7.41 (1H, dt, J = 7.6, 1.3 Hz), 7.50 (1H, dt, J = 7.8, 1.5 Hz), 7.80 (2H, d, J = 8.1 Hz), 7.80 (1H, d, J = 8 Hz), 9.75 (1H, brs).
元素分析値: C37H37N305S3 · 0.6H20として計算値: C, 62.53; H, 5.42; N, 5.91; S, 13.53 (%), 実測値: C, 62.56; H, 5.55; N, 5.93; S, 13.50 (%) . Elemental analysis: C 37 H 37 N 3 0 5 S 3 · 0.6H 2 0 Calculated: C, 62.53; H, 5.42 ; N, 5.91; S, 13.53 (%), Found: C, 62.56; H , 5.55; N, 5.93; S, 13.50 (%).
実施例 4-302 Example 4-302
Ή -賺 (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m), 1.45 (2H, brquint, J= 7.1 Hz), 2.05 (2H, brt, J= 7.1 Hz), 2.41 (3H, s), 4.25 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.07 (1H, ddd, J= 9.8, 8.5, 1.0 Hz), 7.13 (1H, dt, J= 7.5, 1.0 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.30 (1H, m), 7.37 (1H, dt, J= 7.7, 1.6 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). E -賺(300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m), 1.45 (2H, brquint, J = 7.1 Hz), 2.05 (2H, brt, J = 7.1 Hz), 2.41 (3H, s), 4.25 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.07 (1H, ddd, J = 9.8, 8.5, 1.0 Hz), 7.13 (1H, dt, J = 7.5, 1.0 Hz), 7.26 (2H, d, J = 8.3 Hz), 7.30 (1H, m), 7.37 (1H, dt, J = 7.7, 1.6 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C26H2SN303S3Fとして計算値: C, 57.22; H, 5.17; N, 7.70; S, 17.63; F, 3.48 (%) 、 実測値: C, 57.18; H, 5.18; N, 7.90; S, 17.62; F, 3.19 ( ) . 実施例 4-303 Elemental analysis: C 26 H 2S N 3 0 3 S 3 F Calculated: C, 57.22; H, 5.17 ; N, 7.70; S, 17.63; F, 3.48 (%), Found: C, 57.18; H , 5.18; N, 7.90; S, 17.62; F, 3.19 ().
Ή-N R (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m), 1.43 (2H, brquint, J= 6.8 Hz), 2.04 (2H, bri, J= 7.1 Hz), 2.42 (3H, s), 4.19 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.02 (2H, m) , 7.10 (1H, brd, J= 7.8 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.31 (1H, bri, J= 7.8 Hz), 7.81 (2H, d, J= 8.3 Hz) . Ή-NR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.9 Hz), 1.28 (4H, m), 1.30 (2H, m), 1.43 (2H, brquint, J = 6.8 Hz), 2.04 (2H, bri, J = 7.1 Hz), 2.42 (3H, s), 4.19 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.02 (2H, m), 7.10 (1H, brd, J = 7.8 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.31 (1H, bri, J = 7.8 Hz), 7.81 (2H, d, J = 8.3 Hz).
元素分析値: C26H28N303S3Fとして計算値: C, 57.22; H, 5.17; N, 7.70; S, 17.63; F, 3.48 (%), 実測値: C, 57.10; H, 5.04; N, 7.69; S, 17.79; F, 3.31 (%) . 実施例 4-304 Elemental analysis: C 26 H 28 N 3 0 3 S 3 F Calculated: C, 57.22; H, 5.17 ; N, 7.70; S, 17.63; F, 3.48 (%), Found: C, 57.10; H , 5.04; N, 7.69; S, 17.79; F, 3.31 (%).
Ή-NMR (300MHz, CDC13) δ 0.89 (3Η, t, J= 6.9 Hz), 1.29 (4H, m), 1.30 (2H, m), 1.33 (9H, s), 1.45 (2H, brquint, J= 6.8 Hz), 2.06 (2H, brt, J= 7.2 Hz), Ή-NMR (300MHz, CDC1 3 ) δ 0.89 (3Η, t, J = 6.9 Hz), 1.29 (4H, m), 1.30 (2H, m), 1.33 (9H, s), 1.45 (2H, brquint, J = 6.8 Hz), 2.06 (2H, brt, J = 7.2 Hz),
2.41 (3H, s), 4.20 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.35 (2H, d, J= 8.3 Hz), 7.80 (2H, d, J= 8.3 Hz), 9.70 (1H, br). 2.41 (3H, s), 4.20 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.35 (2H, d, J = 8.3 Hz), 7.80 (2H, d, J = 8.3 Hz), 9.70 (1H, br).
元素分析値: C3。H37N303S3として計算値: C, 61.77; H, 6.39; N, 7.20; S, 16.48 (%), 実測値: C, 61.62; H, 6.24; , 7.36; S, 16.63 (%) · 実施例 4-305 Elemental analysis: C 3. H 37 N 3 0 3 S 3 Calculated: C, 61.77; H, 6.39 ; N, 7.20; S, 16.48 (%), Measured: C, 61.62; H, 6.24;, 7.36; S, 16.63 (%) · Example 4-305
'H-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.9 Hz), 1.26 (4H, brs), 1.29 (2H, m), 1.38 (2H, brquint, J= 6.6 Hz), 1.98 (2H, brt, J= 7.1 Hz), .2.41 (3H, s), 4.17 (2H, s), 6.43 (1H, brt, J= 0.5 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.32 (3H, m), 7.37 (5H, m), 7.47 (1H, m), 7.80 (2H, d, J= 8.3 Hz), 9.67 (1H, brs). 元素分析値: C32 N303S3として計算値: C, 63.65; H, 5.51; , 6.96; S, 15.93'H-NMR (300MHz, CDC1 3): δ 0.89 (3H, t, J = 6.9 Hz), 1.26 (4H, brs), 1.29 (2H, m), 1.38 (2H, brquint, J = 6.6 Hz), 1.98 (2H, brt, J = 7.1 Hz), .2.41 (3H, s), 4.17 (2H, s), 6.43 (1H, brt, J = 0.5 Hz), 7.26 (2H, d, J = 8.3 Hz) , 7.32 (3H, m), 7.37 (5H, m), 7.47 (1H, m), 7.80 (2H, d, J = 8.3 Hz), 9.67 (1H, brs) elemental analysis:. C 32 N 3 0 Calculated for 3 S 3 : C, 63.65; H, 5.51;, 6.96; S, 15.93
(%) 、 実測値: C, 63.61; H, 5.39; M, 6.72; S, 15.88 (%) . (%), Found: C, 63.61; H, 5.39; M, 6.72; S, 15.88 (%).
実施例 4-306 Example 4-306
Ή-NMR (300MHz, CDC13) : δ 0.88 (6H, d, J= 6.8 Hz), 0.89 (3H, t, J= 6.9 Hz), 1.18 (2H, brquint, J= 7.2 Hz), 1.29 (6H, m), 1.33 (2H, m), 1.38 (2H, m), 1.47 (2H, brquint, J= 7.1 Hz), 1.53 (1H, quint, J= 7.1 Hz), 1.69 (2H, quint, J= 7.2 Hz), 2.07 (2H, brt, J= 7.3 Hz), 2.42 (3H, s), 2.96 (2H, t, J= 7.6 Hz), 6.51 (1H, brs), 7.28 (2H, d, J= 7.9 Hz), 7.85 (2H, d, J= 7.9 Hz), 9.71 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (6H, d, J = 6.8 Hz), 0.89 (3H, t, J = 6.9 Hz), 1.18 (2H, brquint, J = 7.2 Hz), 1.29 (6H , M), 1.33 (2H, m), 1.38 (2H, m), 1.47 (2H, brquint, J = 7.1 Hz), 1.53 (1H, quint, J = 7.1 Hz), 1.69 (2H, quint, J = 7.2 Hz), 2.07 (2H, brt, J = 7.3 Hz), 2.42 (3H, s), 2.96 (2H, t, J = 7.6 Hz), 6.51 (1H, brs), 7.28 (2H, d, J = 7.9 Hz), 7.85 (2H, d, J = 7.9 Hz), 9.71 (1H, brs).
元素分析値: C27H39N303S3 · 0.2H2Oとして計算値: C, 58.60; H, 7.18; N, 7.59; S, 17.38 ( ) 、 実測値: C, 58.55; H, 6.87; N, 7.82; S, 17.49 (%) . Elemental analysis: C 27 H 39 N 3 0 3 S 3 · 0.2H 2 O Calculated: C, 58.60; H, 7.18; N, 7.59; S, 17.38 (), found: C, 58.55; H, 6.87; N, 7.82; S, 17.49 (%).
実施例 4-307 Example 4-307
'H-NMR (300MHz, CDC13) : δ 0.88 (6H, d, J= 6.8 Hz), 0.89 (3H, t, J= 6.9 Hz), 1.20 (2H, m), 1.29 (4H, m), 1.31 (2H, m) , 1.38 (2H, in), 1.46 (2H, brquint, J= 7.2 Hz), 1.54 (1H, quint, J= 6.8 Hz), 1.67 (2H, quini, J= 7.5 Hz), 2.07 (2H, brt, J= 7.2 Hz), 2.42 (3H, s), 2.96 (2H, t, J= 7.3 Hz), 6.51 (1H, brt, J= 0.5 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.84 (2H, d, J= 8.3 Hz), 9.72 (1H, brs). 元素分析値: C26H37N303S3として計算値: C, 58.28; H, 6.96; N, 7.84; S, 17.95 (%), 実測値: C, 58.17; H, 6.97; N, 7.94; S, 17.94 {%) . 'H-NMR (300MHz, CDC1 3): δ 0.88 (6H, d, J = 6.8 Hz), 0.89 (3H, t, J = 6.9 Hz), 1.20 (2H, m), 1.29 (4H, m), 1.31 (2H, m), 1.38 (2H, in), 1.46 (2H, brquint, J = 7.2 Hz), 1.54 (1H, quint, J = 6.8 Hz), 1.67 (2H, quini, J = 7.5 Hz), 2.07 (2H, brt, J = 7.2 Hz), 2.42 (3H, s), 2.96 (2H, t, J = 7.3 Hz), 6.51 (1H, brt, J = 0.5 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.84 (2H , d, J = 8.3 Hz), 9.72 (1H, brs) elemental analysis: C 26 H 37 N 3 0 3 S 3 calculated:. C, 58.28; H, 6.96; N, 7.84; S, 17.95 (%), Found: C, 58.17; H, 6.97; N, 7.94; S, 17.94 (%).
実施例 4-308 Example 4-308
Ή -腿 (300MHz, CDC13) : δ 0.89 (3Η, t, J- 7.2 Hz), 1.29 (4H, br), 1.32 (2H, m), 1.49 (2H, brquint, J= 6.9 Hz), 2.07 (2H, brt, J= 6.8 Hz), 2.08 (1H, br), 2.09 (2H, s), 2.42 (3H, s), 6.45 (1H, brt, J= 0.5 Hz), 7.30 (2H, d, J= 8.3 Hz), 7.82 (2H, d, J= 8.3 Hz), 9.63 (1H, brs). E - thigh (300MHz, CDC1 3): δ 0.89 (3Η, t, J- 7.2 Hz), 1.29 (4H, br), 1.32 (2H, m), 1.49 (2H, brquint, J = 6.9 Hz), 2.07 (2H, brt, J = 6.8 Hz), 2.08 (1H, br), 2.09 (2H, s), 2.42 (3H, s), 6.45 (1H, brt, J = 0.5 Hz), 7.30 (2H, d, J = 8.3 Hz), 7.82 (2H, d, J = 8.3 Hz), 9.63 (1H, brs).
元素分析値: C22¾5N303S3として計算値: C, 55.55; H, 5.30; N, 8.83; S, 20.22Elemental analysis: C 22 ¾ 5 N 3 0 3 S 3 Calculated: C, 55.55; H, 5.30 ; N, 8.83; S, 20.22
(%) 、 実測値: C, 55.27; H, 5.09; N, 8.97; S, 20.38 (%) . (%), Found: C, 55.27; H, 5.09; N, 8.97; S, 20.38 (%).
実施例 4-309 Example 4-309
ΐ-NM (300MHz, DMSO— d6) δ 0.85 (3Η, t, J= 6.8 Hz), 1.26 (4H, br), 1.28 (2H, m), 1.41 (2H, brquint, J= 6.4 Hz), 2.14 (2H, brt, J= 7.0 Hz), 2.35 (3H, s), 4.37 (2H, s), 6.63 (1H, brs), 7.32 (4H, m), 7.39 (1H, dd, J= 7.5, 1.1 Hz), 7.41 (2H, brt, J= 7.9 Hz), 7.46 (1H, dt, J= 1.1, 7.5 Hz), '7.57 (1H, dt, J= 1.1, 7.5 Hz), 7.73 (1H, dd, J= 7.5, 1.1 Hz), 7.81 (2H, d, J= 8.3 Hz), 11.77 (1H, br), 12.76 (1H, br) . ΐ-NM (300MHz, DMSO- d 6 ) δ 0.85 (3Η, t, J = 6.8 Hz), 1.26 (4H, br), 1.28 (2H, m), 1.41 (2H, brquint, J = 6.4 Hz), 2.14 (2H, brt, J = 7.0 Hz), 2.35 (3H, s), 4.37 (2H, s), 6.63 (1H, brs), 7.32 (4H, m), 7.39 (1H, dd, J = 7.5, 1.11), 7.41 (2H, brt, J = 7.9 Hz), 7.46 (1H, dt, J = 1.1, 7.5 Hz), '7.57 (1H, dt, J = 1.1, 7.5 Hz), 7.73 (1H, dd , J = 7.5, 1.1 Hz), 7.81 (2H, d, J = 8.3 Hz), 11.77 (1H, br), 12.76 (1H, br).
実施例 4-310 Example 4-310
Ή-NMR (300MHz, CDC13) : δ 0.88 (3H, t, J= 6.8 Hz), 1.28 (4H, brs), 1.31 (2H, m), 1.46 (2H, brquint, J= 7.3 Hz), 2.06 (2H, brt, J= 7.0 Hz), 2.40 (3H, s), 3.82 (2H, dd, J= 7.2, 1.1 Hz), 6.18 (1H, dt, J= 15.5, 7.2 Hz), 6.49 (1H, brt, J= 0.5 Hz), 6.61 (1H, d, J= 15.8 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.30 (1H, m), 7.35 (4H, m), 7.84 (2H, d, J= 8.3 Hz), 9.75 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3H, t, J = 6.8 Hz), 1.28 (4H, brs), 1.31 (2H, m), 1.46 (2H, brquint, J = 7.3 Hz), 2.06 (2H, brt, J = 7.0 Hz), 2.40 (3H, s), 3.82 (2H, dd, J = 7.2, 1.1 Hz), 6.18 (1H, dt, J = 15.5, 7.2 Hz), 6.49 (1H, brt, J = 0.5 Hz), 6.61 (1H, d, J = 15.8 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.30 (1H, m), 7.35 (4H, m), 7.84 (2H, d, J = 8.3 Hz), 9.75 (1H, brs).
元素分析値: C28H31N303S3として計算値: C, 60.73; H, 5.64; N, 7.59; S, 17.37Elemental analysis: C 28 H 31 N 3 0 3 S 3 Calculated: C, 60.73; H, 5.64 ; N, 7.59; S, 17.37
(%)、 実測値: C, 60.68; H, 5.53; N, 7.74; S, 17.39 (%) . (%), Found: C, 60.68; H, 5.53; N, 7.74; S, 17.39 (%).
実施例 4-311 Example 4-311
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.14 (2H, brdq, J= 4.4, Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.14 (2H, brdq, J = 4.4,
12.1 Hz), 1.28 (6H, m), 1.46 (2H, m), 1.46 (9H, s), 1.69 (1H, m), 1.79 (2H, brd, J= 13.6 Hz), 2.06 (2H, brt, J= 7.2 Hz), 2.41 (3H, s), 2.68 (2H, brt, J=12.1 Hz), 1.28 (6H, m), 1.46 (2H, m), 1.46 (9H, s), 1.69 (1H, m), 1.79 (2H, brd, J = 13.6 Hz), 2.06 (2H, brt, J = 7.2 Hz), 2.41 (3H, s), 2.68 (2H, brt, J =
12.2 Hz), 2.87 (2H, d, J= 6.8 Hz), 4.11 (2H, brd, J= 14 Hz), 6.51 (1H, brt, J= 0.5 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz). 12.2 Hz), 2.87 (2H, d, J = 6.8 Hz), 4.11 (2H, brd, J = 14 Hz), 6.51 (1H, brt, J = 0.5 Hz), 7.27 (2H, d, J = 8.3 Hz) ), 7.85 (2H, d, J = 8.3 Hz).
元素分析値: C3。H42N405S3 · 0.7H20として計算値: C, 55.65; H, 6.76; , 8.65; S, 14.85 (%) 、 実測値: C, 55.67; H, 6.47; N, 8.58; S, 14.84 ( ) . Elemental analysis: C 3. H 42 N 4 0 5 S 3 · 0.7H 2 0 Calculated: C, 55.65; H, 6.76 ;, 8.65; S, 14.85 (%), Found: C, 55.67; H, 6.47 ; N, 8.58; S, 14.84 ().
実施例 4-312 Example 4-312
Ή-NMR (300MHz, DMSO-d6) : δ 0.86 (3H, t, J= 6.6 Hz), 1.26 (4H, m), 1.30 (2H, m), 1.38 (2H,'m), 1.81 (1H, i), 1.87 (2H, brd, J= 15.4 Hz), 2.13 (2H, brt, J= 7.0 Hz), 2.38 (3H, s), 2.86 (2H, brq, J= 11.3 Hz), 2.97 (2H, d, J= 6.4 Hz), 3.30 (2H, brd, J= 14 Hz), 6.63 (1H, brs), 7.37 (2H, d, J= 7.9 Hz), 7.83 (2H, d, J= 7.9 Hz), 8.19 (1H, brs), 8.50 (1H, br), 11.80 (1H, br). Ή-NMR (300 MHz, DMSO-d 6 ): δ 0.86 (3H, t, J = 6.6 Hz), 1.26 (4H, m), 1.30 (2H, m), 1.38 (2H, 'm), 1.81 (1H , i), 1.87 (2H, brd, J = 15.4 Hz), 2.13 (2H, brt, J = 7.0 Hz), 2.38 (3H, s), 2.86 (2H, brq, J = 11.3 Hz), 2.97 (2H, d, J = 6.4 Hz), 3.30 (2H, brd, J = 14 Hz), 6.63 (1H, brs), 7.37 (2H, d, J = 7.9 Hz), 7.83 (2H, d, J = 7.9 Hz), 8.19 (1H, brs), 8.50 (1H, br), 11.80 (1H, br) .
元素分析値: C27H35N405S3F3として計算値: C, 49.98; H, 5.44; N, 8.64; S, 14.83; F, 8.78 (%), 実測値: C, 49.72; H, 5.33; N, 8.70; S, 14.86 (%) . Elemental analysis: Calculated as C 27 H 35 N 4 0 5 S 3 F 3: C, 49.98; H, 5.44; N, 8.64; S, 14.83; F, 8.78 (%), Found: C, 49.72; H, 5.33; N, 8.70; S, 14.86 (%).
実施例 4-313 Example 4-313
Ή-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.6 Hz), 1.29 (6H, br), 1.39 (2H, m), 1.43 (2H, brquint, J= 7.2 Hz), 1.56 (1H, m), 1.81 (2H, brd, J= 13.8 Hz), 2.02 (2H, brt, J= 11.7 Hz), 2.06 (2H, t, J= 7.3 Hz), 2.39 (3H, s), 2.87 (2H, d, J= 6.8 Hz), 2.97 (2H, brt, J= 11.3 Hz), 3.57 (2H, s), 6.49 (1H, brt, J= 0.5 Hz), 7.26 (2H, m), 7.32 (5H, s), 7.85 (2H, d, J- 8.3 Hz). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3Η, t, J = 6.6 Hz), 1.29 (6H, br), 1.39 (2H, m), 1.43 (2H, brquint, J = 7.2 Hz), 1.56 (1H, m), 1.81 (2H, brd, J = 13.8 Hz), 2.02 (2H, brt, J = 11.7 Hz), 2.06 (2H, t, J = 7.3 Hz), 2.39 (3H, s), 2.87 (2H, d, J = 6.8 Hz), 2.97 (2H, brt, J = 11.3 Hz), 3.57 (2H, s), 6.49 (1H, brt, J = 0.5 Hz), 7.26 (2H, m), 7.32 (5H, s), 7.85 (2H, d, J- 8.3 Hz).
実施例 4-314 Example 4-314
¾-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.28 (6H, m), 1.44 (2H, brquint, J= 6.8 Hz), 2.04 (2H, t, J= 7.0 Hz), 2.31 (3H, s), 2.41 (3H, s), 4.20 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.06 (2H, dt, J= 8.7, 1.9 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.33 (2H, dt, J= 8.7, 2.6 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.72 (1H, brs). ¾-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.28 (6H, m), 1.44 (2H, brquint, J = 6.8 Hz), 2.04 (2H, t, J = 7.0 Hz), 2.31 (3H, s), 2.41 (3H, s), 4.20 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.06 (2H, dt, J = 8.7, 1.9 Hz) , 7.27 (2H, d, J = 8.3 Hz), 7.33 (2H, dt, J = 8.7, 2.6 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.72 (1H, brs).
元素分析値: C28H31N305S3として計算値: C, 57.41; H, 5.33; N, 7.17; S, 16.42 (%) 、 実測値: C, 57.35; H, 5.06; N, 7.21; S, 16.37 ( ) . Elemental analysis: C 28 H 31 N 3 0 5 S 3 Calculated: C, 57.41; H, 5.33; N, 7.17; S, 16.42 (%), found: C, 57.35; H, 5.06; N, 7.21; S, 16.37 ().
実施例 4-315 Example 4-315
'H-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.6 Hz), 1.29 (6H, m), 1.48 (2H, brquint, J= 7.3 Hz), 2.06 (2H, t, J= 7.0 Hz), 2.41 (3H, s), 3.27 (2H, t, J= 7.0 Hz), 3.99 (2H, t, J= 7.0 Hz), 6.47 (1H, brt, J= 0.5 Hz), 7.26 (2H, d, J= 7.9 Hz), 7.75 (2H, dd, J= 5.3, 3.0 Hz), 7.81 (2H, d, J= 8.3 Hz), 7.86 (2H, dd, J= 5.3, 3.0 Hz), 9.69 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.6 Hz), 1.29 (6H, m), 1.48 (2H, brquint, J = 7.3 Hz), 2.06 (2H, t, J = 7.0 Hz), 2.41 (3H, s), 3.27 (2H, t, J = 7.0 Hz), 3.99 (2H, t, J = 7.0 Hz), 6.47 (1H, brt, J = 0.5 Hz), 7.26 ( 2H, d, J = 7.9 Hz), 7.75 (2H, dd, J = 5.3, 3.0 Hz), 7.81 (2H, d, J = 8.3 Hz), 7.86 (2H, dd, J = 5.3, 3.0 Hz), 9.69 (1H, brs).
元素分析値: C29H30N405S3 · 0.5H20として計算値: C, 56.20; H, 5.04; N, 9.04; S, 15.52 (%), 実測値: C, 56.11; H, 5.01; N, 9.15; S, 15.65 (%) . Elemental analysis: C 29 H 30 N 4 0 5 S 3 · 0.5 H 2 0 Calculated: C, 56.20; H, 5.04; N, 9.04; S, 15.52 (%), found: C, 56.11; H , 5.01; N, 9.15; S, 15.65 (%).
実施例 4-316 Example 4-316
'H-NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.8 Hz), 1.27 (6H, m), 1.40 (2H, brquint, J= 7.0 Hz), 1.99 (2H, t, J= 7.2 Hz), 2.07 (2H, quint, J= 6.8 Hz), 2.39 (3H, s), 2.99 (2H, t, J= 7.5 Hz), 3.76 (2H, t, J= 6.8 Hz), 6.47 (1H, brt, J= 0.5 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.74 (2H, dd, J= 5.3, 3.0 Hz), 7.83 (2H, d, J= 8.3 Hz), 7.86 (2H, dd, J= 5.7, 3.0 Hz), 9.72 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.8 Hz), 1.27 (6H, m), 1.40 (2H, brquint, J = 7.0 Hz), 1.99 (2H, t, J = 7.2 Hz), 2.07 (2H, quint, J = 6.8 Hz), 2.39 (3H, s), 2.99 (2H, t, J = 7.5 Hz) , 3.76 (2H, t, J = 6.8 Hz), 6.47 (1H, brt, J = 0.5 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.74 (2H, dd, J = 5.3, 3.0 Hz) , 7.83 (2H, d, J = 8.3 Hz), 7.86 (2H, dd, J = 5.7, 3.0 Hz), 9.72 (1H, brs).
元素分析値: C3。H32N405S3として計算値: C, 57.37; H, 5.16; N, 8.9-7; S, 15.40 (%)、 実測値: C, 57.64; H, 5.11; , 9.13; S, 15.45 (%) . Elemental analysis: C 3. H 32 N 4 0 5 S 3 Calculated: C, 57.37; H, 5.16 ; N, 8.9-7; S, 15.40 (%), Found: C, 57.64; H, 5.11 ;, 9.13; S, 15.45 (%).
実施例 4-317 . ' Ή-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J= 6.8 Hz), 1.23 (4H, m), 1.27 (2H, m), 1.32 (2H, m), 1.93 (2H, t, J= 7.2 Hz), 2.42 (3H, s), 5.90 (1H, s), 6.35 (1H, brt, J= 0.5 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.30 (2H, m), 7.33 (8H, m), 7.83 (2H, d, J= 8.3 Hz), 9.70 (1H, brs). . Example 4-317 'Ή-NMR (300MHz, CDC1 3): δ 0.88 (3Η, t, J = 6.8 Hz), 1.23 (4H, m), 1.27 (2H, m), 1.32 (2H, m) , 1.93 (2H, t, J = 7.2 Hz), 2.42 (3H, s), 5.90 (1H, s), 6.35 (1H, brt, J = 0.5 Hz), 7.26 (2H, d, J = 8.3 Hz) , 7.30 (2H, m), 7.33 (8H, m), 7.83 (2H, d, J = 8.3 Hz), 9.70 (1H, brs).
元素分析値: C32 N303S3として計算値: C, 63.65; H, 5.51; N, 6.96; S, 15.93Elemental analysis: Calculated as C 32 N 3 0 3 S 3 : C, 63.65; H, 5.51; N, 6.96; S, 15.93
(%) 、 実測値: C, 63.61; H, 5.43; N, 7.05; S, 16.08 (%) . (%), Found: C, 63.61; H, 5.43; N, 7.05; S, 16.08 (%).
実施例 4-318 Example 4-318
^- MR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.28 (6H, m), 1.45 (2H, brquint, J= 7.0 Hz), 2.06 (2H, t, J= 7.0 Hz), 2.41 (3H, s), 4.33 (2H, s), 6.48 (1H, brt, J= 0.5 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.27 (2H, m), 7.41 (2H, m), 7.81 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). ^ - MR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.28 (6H, m), 1.45 (2H, brquint, J = 7.0 Hz), 2.06 (2H, t, J = 7.0 Hz), 2.41 (3H, s), 4.33 (2H, s), 6.48 (1H, brt, J = 0.5 Hz), 7.26 (2H, d, J = 8.3 Hz), 7.27 (2H, m), 7.41 (2H, m), 7.81 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C26H28N303S3C1 · 0.5H2O: C, 54.67; H, 5.12; N, 7.36; S, 16.84; CI, 6.21 (%)、 実測値: C, 54.63; H, 5.24; N, 7.39; S, 16.82; CI, 6.29 (%) . 実施例 4-319 Elemental analysis: C 26 H 28 N 3 0 3 S 3 C1 · 0.5H 2 O: C, 54.67; H, 5.12; N, 7.36; S, 16.84; CI, 6.21 (%), Found: C, 54.63 ; H, 5.24; N, 7.39; S, 16.82; CI, 6.29 (%). Example 4-319
'H-NMR (300MHz, CDC13) : δ 0.86 (3H, t, J= 6.6 Hz), 1.23 (6H, m), 1.37 (2H, brquint, J= 7.2 Hz), 1.97 (2H, t, J= 7.0 Hz), 2.43 (3H, s), 4.57 (2H, s), 6.34 (1H, brt, J= 0.5 Hz), 7.29 (2H, m), 7.30 (2H, d, J= 8.3 Hz), 7.68 (2H, dd, J= 6.0, 3.0 Hz), 7.82 (2H, d, J= 8.3 Hz). 'H-NMR (300MHz, CDC1 3): δ 0.86 (3H, t, J = 6.6 Hz), 1.23 (6H, m), 1.37 (2H, brquint, J = 7.2 Hz), 1.97 (2H, t, J = 7.0 Hz), 2.43 (3H, s), 4.57 (2H, s), 6.34 (1H, brt, J = 0.5 Hz), 7.29 (2H, m), 7.30 (2H, d, J = 8.3 Hz), 7.68 (2H, dd, J = 6.0, 3.0 Hz), 7.82 (2H, d, J = 8.3 Hz).
元素分析値: C27H29N503S3 · 0.2H20として計算値: C, 56.76; H, 5.19; , 12.26; S, 16.83 (%) .実測値: C, 56.68; H, 5.20; N, 12.33; S, 16.87 (%) . Elemental analysis: C 27 H 29 N 5 0 3 S 3 · 0.2H 2 0 Calculated:. C, 56.76; H, 5.19;, 12.26; S, 16.83 (%) Found: C, 56.68; H, 5.20; N, 12.33; S, 16.87 (%).
実施例 4-320 Example 4-320
Ή-NMR (300MHz, CDC13) : δ 0.86 (3H, i, J= 6.8 Hz), 1.22 (4H, m), 1.25 (2H, m), 1.38 (2H, brquint, J- 7.0 Hz), 2.01 (2H, t, J= 7.5 Hz), 2.40 (3H, s), 4.55 (2H, s), 6.45 (1H, brs), 7.24 (2H, d, J= 8.3 Hz), 7.54 (1H, d, J= 8.7 Hz), 7.58 (1H, brt, J= 7.9 Hz), 7.75 (1H, dt, J= 7.7, 1.0 Hz), 7.83 (2H, d, J= 8.3 Hz), 7.84 (1H, brd, J= 8.3 Hz), 8.07 (1H, brd, J= 8.7 Hz), 8.17 (1H, d, J= 8.3 Hz), 9.73 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.86 (3H, i, J = 6.8 Hz), 1.22 (4H, m), 1.25 (2H, m), 1.38 (2H, brquint, J- 7.0 Hz), 2.01 (2H, t, J = 7.5 Hz), 2.40 (3H, s), 4.55 (2H, s), 6.45 (1H, brs), 7.24 ( 2H, d, J = 8.3 Hz), 7.54 (1H, d, J = 8.7 Hz), 7.58 (1H, brt, J = 7.9 Hz), 7.75 (1H, dt, J = 7.7, 1.0 Hz), 7.83 ( 2H, d, J = 8.3 Hz), 7.84 (1H, brd, J = 8.3 Hz), 8.07 (1H, brd, J = 8.7 Hz), 8.17 (1H, d, J = 8.3 Hz), 9.73 (1H, brs).
元素分析値: C29 。N403S3として計算値: C, 60.18; H, 5.22; N, 9.68; S, 16.62 (%) 、 実測値: C, 59.87; H, 5.23; N, 9.72; S, 16.51 ( ) . Elemental analysis: C 29. Calculated for N 4 0 3 S 3 : C, 60.18; H, 5.22; N, 9.68; S, 16.62 (%), found: C, 59.87; H, 5.23; N, 9.72; S, 16.51 ().
実施例 4- 321 Example 4-321
ー丽 R (300MHz, CDC13) : 6 0.89 (3H, t, J= 6.4 Hz), 1.28 (6H, br), 1.43 (2H, brquint, J= 6.8 Hz), 2.04 (2H, t, J= 7.0 Hz), 2.42 (3H, s), 4.17 (2H, s), 6.46Over丽R (300MHz, CDC1 3): 6 0.89 (3H, t, J = 6.4 Hz), 1.28 (6H, br), 1.43 (2H, brquint, J = 6.8 Hz), 2.04 (2H, t, J = 7.0 Hz), 2.42 (3H, s), 4.17 (2H, s), 6.46
(1H, brs), 7.27 (6H, m), 7.80 (2H, d, J= 7.8 Hz), 9.72 (1H, brs). (1H, brs), 7.27 (6H, m), 7.80 (2H, d, J = 7.8 Hz), 9.72 (1H, brs).
元素分析値: C26H28N303S3C1 · 0.3H20として計算値: C, 55.02; H, 5.08; N, 7.40;Elemental analysis: C 26 H 28 N 3 0 3 S 3 C 1 · 0.3 H 2 0, calculated: C, 55.02; H, 5.08; N, 7.40;
S, 16.95; CI, 6.25 (%), 実測値: C, 55.02; H, 5.10; N, 7.40; S, 17.15; Cl,S, 16.95; CI, 6.25 (%), Found: C, 55.02; H, 5.10; N, 7.40; S, 17.15; Cl,
6.19 (%) . ' 6.19 (%). '
実施例 4-322 Example 4-322
Ή一 NMR (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.6 Hz), 1.27 (6H, br), 1.41 (2H, brquint, J= 6.6 Hz), 2.02 (2H, t, J= 7.2 Hz), 2.43 (3H, s), 4.15 (2H, s), 6.52 (1H, brs), 7.26 (2H, d, J= 5.7 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.82 (2H, d, J= 8.3 Hz), 8.57 (2H, d, J= 5.7 Hz). Ή one NMR (300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.6 Hz), 1.27 (6H, br), 1.41 (2H, brquint, J = 6.6 Hz), 2.02 (2H, t, J = 7.2 Hz), 2.43 (3H, s), 4.15 (2H, s), 6.52 (1H, brs), 7.26 (2H, d, J = 5.7 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.82 (2H, d, J = 8.3 Hz), 8.57 (2H, d, J = 5.7 Hz).
元素分析値: C25H28N403S3として計算値: C, 56.79; H, 5.34; N, 10.60; S, 18.19 (%) .実測値: C, 56.59; H, 5.23; N, 10.52; S, 18.08 (%) . Elemental analysis: C 25 H 28 N 4 0 3 S 3 Calculated:. C, 56.79; H, 5.34; N, 10.60; S, 18.19 (%) Found: C, 56.59; H, 5.23 ; N, 10.52; S, 18.08 (%).
実施例 4-323 Example 4-323
Ή-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J= 6.8 Hz), 1.28 (6H, m), 1.45 (2H, brquint, J= 6.7 Hz), 2.06 (2H, t, J= 7.2 Hz), 2.40 (3H, s), 4.26 (2H, s), 6.48 (1H, brs), 7.26 (2H, d, J= 7.9 Hz), 7.37 (1H, i), 7.39 (2H, d, J= 8.3 Hz), 7.46 (2H, t, J= 7.4 Hz), 7.56 (2H, d, J= 8.7 Hz), 7.59 (2H, dm, J= 8 Hz), 7.81 (2H, d, J= 8.3 Hz), 9.74 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3Η, t, J = 6.8 Hz), 1.28 (6H, m), 1.45 (2H, brquint, J = 6.7 Hz), 2.06 (2H, t, J = 7.2 Hz), 2.40 (3H, s), 4.26 (2H, s), 6.48 (1H, brs), 7.26 (2H, d, J = 7.9 Hz), 7.37 (1H, i), 7.39 (2H, d, J = 8.3 Hz), 7.46 (2H, t, J = 7.4 Hz), 7.56 (2H, d, J = 8.7 Hz), 7.59 (2H, dm, J = 8 Hz), 7.81 (2H, d, J = 8.3 Hz), 9.74 (1H, brs).
元素分析値: C32H33N303S3として計算値: C, 63.65; H, 5.51; N, 6.96; S, 15.93 ( ) 、 実測値: C, 63.49; H, 5.28; N, 6.85; S, 15.90 (%) 実施例 4-324 Elemental analysis: C 32 H 33 N 3 0 3 S 3 Calculated: C, 63.65; H, 5.51 ; N, 6.96; S, 15.93 (), Found: C, 63.49; H, 5.28 ; N, 6.85 ; S, 15.90 (%) Example 4-324
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.28 (6H, m), 1.45 (2H, brqoint, J= 6.8 Hz), 2.06 (2H, t, J= 7.2 Hz), 2.41 (3H, s), 4.34 (2H, s), 6.48 (IH, brt, J= 0.5 Hz), 7.19 (IH, dt, J= 1.5, 7.5 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.30 (IH, dt, J= 1.1, 7.5 Hz), 7.43 (IH, dd, J= 7.5, 1.5 Hz), 7.58 (IH, dd, J= 7.9, 1.1 Hz), 7.82 (2H, d, J= 8.3 Hz), 9.70 (IH, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.28 (6H, m), 1.45 (2H, brqoint, J = 6.8 Hz), 2.06 (2H, t, J = 7.2 Hz), 2.41 (3H, s), 4.34 (2H, s), 6.48 (IH, brt, J = 0.5 Hz), 7.19 (IH, dt, J = 1.5, 7.5 Hz), 7.26 (2H, d, J = 8.3 Hz), 7.30 (IH, dt, J = 1.1, 7.5 Hz), 7.43 (IH, dd, J = 7.5, 1.5 Hz), 7.58 (IH, dd, J = 7.9, 1.1 Hz), 7.82 ( 2H, d, J = 8.3 Hz), 9.70 (IH, brs).
実施例 4-325Example 4-325
-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.29 (6H, m), 1.46 (2H, brquint, J= 6.8 Hz), 2.06 (2H, t, J= 7.0 Hz), 2.41 (3H, s), 4.32 (2H, s), 6.48 (IH, brt, J= 0.5 Hz), 7.01 (IH, dt, J= 1.5, 7.5 Hz), 7.26 (2H, d, J= 8.3 Hz), 7.34 (1H, dt, J= 1.1, 7.2 Hz), 7.43 (IH, dd, J= 7.5, 1.5 Hz), 7.82 (2H, d, J= 8.3 Hz), 7.86 (IH, dd, J= 7.5, 1.5 Hz), 9.70 (IH, brs). -NMR (300MHz, CDC1 3): δ 0.89 (3H, t, J = 6.8 Hz), 1.29 (6H, m), 1.46 (2H, brquint, J = 6.8 Hz), 2.06 (2H, t, J = 7.0 Hz), 2.41 (3H, s), 4.32 (2H, s), 6.48 (IH, brt, J = 0.5 Hz), 7.01 (IH, dt, J = 1.5, 7.5 Hz), 7.26 (2H, d, J = 8.3 Hz), 7.34 (1H, dt, J = 1.1, 7.2 Hz), 7.43 (IH, dd, J = 7.5, 1.5 Hz), 7.82 (2H, d, J = 8.3 Hz), 7.86 (IH, dd , J = 7.5, 1.5 Hz), 9.70 (IH, brs).
元素分析値: C26H28N303S3Iとして計算値: C, 47.78; H, 4.32; N, 6.43; S, 14.72; I, 19.42 (%), 実測値: C, 47.89; H, 4.29; N, 6.38; S, 14.73; I, 19.20 ( ) . 実施例 4-326 Elemental analysis: C 26 H 28 N 3 0 3 S 3 I Calculated: C, 47.78; H, 4.32 ; N, 6.43; S, 14.72; I, 19.42 (%), Found: C, 47.89; H , 4.29; N, 6.38; S, 14.73; I, 19.20 ().
'H-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J= 6.6 Hz), 1.28 (6H, br), 1.44 (2H, brquint, J= 6.7 Hz), 2.04 (2H, t, J= 7.0 Hz), 2.41 (3H, s), 3.15 (2H, dd, J= 7.5, 7.2 Hz), 3.30 (2H, brt, J= 7.5 Hz), 6.47 (IH, brt, J= 0.5 Hz), 7.07 (1H, d, J= 2.3 Hz), 7.13 (IH, dt, J= 0.8, 7.4 Hz), 7.22 (1H, dt, J= 1.1, 7.5 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.38 (IH, d, J= 7.9 Hz), 7.58 (IH, d, J= 7.5 Hz), 7.81 (2H, d, J= 8.3 Hz), 8.07 (IH, brs), 9.71 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.88 (3Η, t, J = 6.6 Hz), 1.28 (6H, br), 1.44 (2H, brquint, J = 6.7 Hz), 2.04 (2H, t, J = 7.0 Hz), 2.41 (3H, s), 3.15 (2H, dd, J = 7.5, 7.2 Hz), 3.30 (2H, brt, J = 7.5 Hz), 6.47 (IH, brt, J = 0.5 Hz), 7.07 (1H, d, J = 2.3 Hz), 7.13 (IH, dt, J = 0.8, 7.4 Hz), 7.22 (1H, dt, J = 1.1, 7.5 Hz), 7.25 (2H, d, J = 8.3 Hz) ), 7.38 (IH, d, J = 7.9 Hz), 7.58 (IH, d, J = 7.5 Hz), 7.81 (2H, d, J = 8.3 Hz), 8.07 (IH, brs), 9.71 (1H, brs) ).
元素分析値: C29H32N403S3として計算値: C, 59.97; H, 5.55; N, 9.65; S, 16.56Elemental analysis: C 29 H 32 N 4 0 3 S 3 Calculated: C, 59.97; H, 5.55 ; N, 9.65; S, 16.56
(%) 、 実測値: C, 59.88; H, 5.60; N, 9.62; S, 16.66 (%) (%), Found: C, 59.88; H, 5.60; N, 9.62; S, 16.66 (%)
実施例 4-327 Example 4-327
Ή-NMR (300MHz, CDC13) : 6 0.89 (3H, t, J= 6.6 Hz), 1.28 (6H, br), 1.43 (2H, brquint, J= 7.3 Hz), 2.03 (2H, t, J= 7.0 Hz), 2.42 (3H, s), 4.56 (2H, s), 6.45 (IH, brs), 7.29 (2H, d, J= 8.3 Hz), 7.50 (IH, m), 7.61 (2H, m), 7.81 (2H, d, J= 8.3 Hz), 8.05 (IH, d, J= 7.9 Hz), 9.70 (IH, brs). Ή-NMR (300MHz, CDC1 3 ): 6 0.89 (3H, t, J = 6.6 Hz), 1.28 (6H, br), 1.43 (2H, brquint, J = 7.3 Hz), 2.03 (2H, t, J = 7.0 Hz), 2.42 (3H, s), 4.56 (2H, s), 6.45 (IH, brs), 7.29 (2H, d, J = 8.3 Hz), 7.50 (IH, m), 7.61 (2H, m) , 7.81 (2H, d, J = 8.3 Hz), 8.05 (IH, d, J = 7.9 Hz), 9.70 (IH, brs).
元素分析値: C26H28N405S3 · 0.2H20として計算値: C, 54.18; H, 4.97; , 9.72; S, 16.69 ( ) 、 実測値: C, 54.06; H, 4.86; N, 9.73; S, 16.65 (%) . Elemental analysis: C 26 H 28 N 4 0 5 S 3 · 0.2 H 2 0 Calculated: C, 54.18; H, 4.97;, 9.72; S, 16.69 (), Found: C, 54.06; H, 4.86; N, 9.73; S, 16.65 (%).
実施例 4-328 Example 4-328
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.27 (6H, m), 1.43 (2H, brquint, J= 7.0 Hz), 2.04 (2H, t, J= 7.2 Hz), 2.41 (3H, s), 4.38 (2H, s), 6.48 (1H, brs), 7.25 (2H, d, J= 8.3 Hz), 7.44 (1H, brt, J= 7.5 Hz), 7.55 (1H, brt, J= 7.5 Hz), 7.61 (1H, brd, J= 7.5 Hz), 7.69 (1H, brd, J= 7.9 Hz), 7.80 (2H, d, J= 8.3 Hz), 9.72 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.27 (6H, m), 1.43 (2H, brquint, J = 7.0 Hz), 2.04 (2H, t, J = 7.2 Hz), 2.41 (3H, s), 4.38 (2H, s), 6.48 (1H, brs), 7.25 (2H, d, J = 8.3 Hz), 7.44 (1H, brt, J = 7.5 Hz), 7.55 (1H, brt, J = 7.5 Hz), 7.61 (1H, brd, J = 7.5 Hz), 7.69 (1H, brd, J = 7.9 Hz), 7.80 (2H, d, J = 8.3 Hz), 9.72 (1H , Brs).
元素分析値: C27 N303S3F3として計算値: C, 54.44; H, 4.74; N, 7.05; S, 16.15; F, 9.57 (%)、 実測値: C, 54.48; H, 4.70; N, 7.08; S, 16.36; F, 9.48 (%) . 実施例 4-329 Elemental analysis: Calculated as C 27 N 3 0 3 S 3 F 3: C, 54.44; H, 4.74; N, 7.05; S, 16.15; F, 9.57 (%), Found: C, 54.48; H, 4.70; N, 7.08; S, 16.36; F, 9.48 (%). Example 4-329
Ή-NMR (300MHz, CDC13) : δ 0.90 (3Η, , J= 7.1 Hz), 1.29 (6H, m), 1.47 (2H, brquint, J= 7.2 Hz), 2.07 (2H, t, J= 7.4 Hz), 2.41 (3H, s), 4.28 (2H, s), 6.48 (1H, brs), 6.93 (2H, t, J= 7.9 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.34 (1H, tt, J= 8.7, 6.8 Hz), 7.80 (2H, d, J= 8.3 Hz), 9.72 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3Η,, J = 7.1 Hz), 1.29 (6H, m), 1.47 (2H, brquint, J = 7.2 Hz), 2.07 (2H, t, J = 7.4 Hz), 2.41 (3H, s), 4.28 (2H, s), 6.48 (1H, brs), 6.93 (2H, t, J = 7.9 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.34 ( 1H, tt, J = 8.7, 6.8 Hz), 7.80 (2H, d, J = 8.3 Hz), 9.72 (1H, brs).
元素分析値: C26H27N303S3F2として計算値: C, 55.40; H, 4.83; N, 7.45; S, 17.07; F, 6.74 (%) 、 実測値: C, 55.24; H, 4.97; N, 7.45; S, 17.14; F, 6.72 ( ) . 実施例 4-330 Elemental analysis: C 26 H 27 N 3 0 3 S 3 F 2 Calculated: C, 55.40; H, 4.83 ; N, 7.45; S, 17.07; F, 6.74 (%), Found: C, 55.24; H, 4.97; N, 7.45; S, 17.14; F, 6.72 () .Example 4-330
'H-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, J= 6.6 Hz), 1.30 (6H, m), 1.48 (2H, brquint, J= 7.2 Hz), 2.08 (2H, t, J= 7.2 Hz), 2.40 (3H, s), 4.55 (2H, s), 6.50 (1H, brs), 7.23 (1H, dd, J= 9.0, 7.2 Hz), 7.24 (2H, d, J= 7.9 Hz), 7.36 (2H, dd, 1= 8.3, 0.8 Hz), 7.79 (2H, d, J= 8.3 Hz), 9.70 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.90 (3Η, t, J = 6.6 Hz), 1.30 (6H, m), 1.48 (2H, brquint, J = 7.2 Hz), 2.08 (2H, t, J = 7.2 Hz), 2.40 (3H, s), 4.55 (2H, s), 6.50 (1H, brs), 7.23 (1H, dd, J = 9.0, 7.2 Hz), 7.24 (2H, d, J = 7.9 Hz) ), 7.36 (2H, dd, 1 = 8.3, 0.8 Hz), 7.79 (2H, d, J = 8.3 Hz), 9.70 (1H, brs).
元素分析値: C26H27N303S3C12 · 0.3H20として計算値: C, 51.87; H, 4.62; , 6.98;Elemental analysis: C 26 H 27 N 3 0 3 S 3 C 12 · 0.3 H 2 0 Calculated: C, 51.87; H, 4.62;, 6.98;
S, 15.98; CI, 11.78 (%), 実測値: C, 51.85; H, 4.69; N, 7.02; S, 15.97; CI,S, 15.98; CI, 11.78 (%), Found: C, 51.85; H, 4.69; N, 7.02; S, 15.97; CI,
11.65 (%) . 11.65 (%).
実施例 4-331 Example 4-331
Ή - NMR(CDC13, 300 MHz): δ 0.89 (3H, brt, J= 7.0 Hz), 1.28 (6H, brs), 1.44 (2H, brquint, 6.9 Hz), 2.03 (2H, brt, J= 7.2 Hz), 2.16 (2H, quint, J= 7.0 Hz), 2.39 (3H, s), 2.75 (3H, s), 3.11 (2H, t, J= 7.1 Hz), 3.14 (2H, t, J= 6.8 Hz), 6.48 (1H, brt, J= 1 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.49 (2H, t, J= •(s J 'Ηΐ) ΐΖ'6 '(in Ή2) 56 -06 '(πι ¾ΐ) 09 -83 '(ω 'ΕΖ) IS' - 'A '(ω ) '(ω 'ΗΖ) Ή - NMR (CDC1 3, 300 MHz): δ 0.89 (3H, brt, J = 7.0 Hz), 1.28 (6H, brs), 1.44 (2H, brquint, 6.9 Hz), 2.03 (2H, brt, J = 7.2 Hz), 2.16 (2H, quint, J = 7.0 Hz), 2.39 (3H, s), 2.75 (3H, s), 3.11 (2H, t, J = 7.1 Hz), 3.14 (2H, t, J = 6.8) Hz), 6.48 (1H, brt, J = 1 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.49 (2H, t, J = • (s J 'Ηΐ) ΐΖ'6' (in Ή2) 56 -06 '(πι ¾ΐ) 09 -83' (ω 'ΕΖ) IS'-'A' (ω) '(ω' ΗΖ)
90· _86'9 '(s Jq 'Ηΐ) 8 9 £(s 'ΗΖ) OZ'f '(ZH0"A=f Ί ュ q 'Μ) 90 *Ζ '(ω ΊΚ) 8f ·ΐ-8Π '(ω Ή9) ^ -Η '(ΖΗ0' =1 '1 'HS) 06·0ί> : (ε13α3 'ZHW 00S) ^ΝΝ-Η, 90_ _86'9 '(s Jq' Ηΐ) 8 9 £ (s 'ΗΖ) OZ'f' (ZH0 "A = fΊ q 'Μ) 90 * Ζ' (ω ΊΚ) 8f (ω Ή9) ^ -Η '(ΖΗ0' = 1 '1' HS) 060ί>: ( ε 13α3 'ZHW 00S) ^ ΝΝ-Η,
'(m 'Η ΐ) 09· 9ΖΊ '(s Jq 'HI) t '9 '(s ) 'Μ) 86 "ΐ ' 'Η8) ΐ-ΟΓΐ '( 6·9=ί ¾S) 68Ό : (εΙ003 £ZHW 008) N-Ht '(m' Η ΐ) 09 · 9ΖΊ '(s Jq' HI) t '9' (s) 'Μ) 86 "ΐ''Η8)ΐ-ΟΓΐ' (6.9 = ί ¾S) 68Ό: ( ε Ι003 £ ZHW 008) NH t
'(ω ¾1) 80 -8-80 "8 '(in 'Η2) OS 96·Ζ- 68 ·Ζ ' 'Η9) ·Α - Sf ·Ζ '(s ¾t) Α 9 '(s Έ2) AS * 'WZ-f '1 Jq 2) 0 '(in 'Η8) 8f 1-0Π '(ΖΗ0 =Γ Ί 'HS) 68 ·0 ρ : ', 00S) 翻 - '(ω ¾1) 80 -8-80 "8' (in 'Η2) OS 96Ζ-68Ζ Ζ' 'Η9) Α S-SfΖ Ζ' (s ¾t) Α 9 '(s Έ2) AS * 'WZ-f' 1 Jq 2) 0 '(in' Η8) 8f 1-0Π '(ΖΗ0 = Γ Ί' HS) 68
•(in 'Ηΐ) AZ-6-t9"6 '(in Z) S6 · ー 68 · '(m ¾S) Id'L-ZfL '(m £H9) • (in 'Ηΐ) AZ-6-t9 "6' (in Z) S6 · ー 68 · '(m¾S) Id'L-ZfL' (m £ H9)
'(s Jq 'Ηΐ) 8f 9 '(s 'ΗΖ) ΖΙ '(¾0·Α=ί Ί Ή2) SO '(ω 'HZ) ST 6fl-88-I '(ω 'Η9) ζξ - Ζ'\ '(ΖΗ8·9=ί Ί ¾8) 68·09 : ( 3(D 'ζ删 OS) ¾ -Η, '(s Jq' Ηΐ) 8f 9 '(s' ΗΖ) ΖΙ '(¾0Α = ί Ή Ή2) SO' (ω 'HZ) ST 6fl-88-I' (ω 'Η9) ζξ-Ζ' \ '(ΖΗ8 · 9 = ί ¾ ¾8) 68 · 09: (3 (D' ζ 删 OS) ¾ -Η,
• ( ) ·9Ι 'S :9ΐ· 'Ν 'Η 69 Ό :華腿牽 (%) 91 'S : ΖΓ 'Ν :Ζ0·9 'Η :9 ·69 '3:ΐ!}鴛 +ェっ? θ¥¾6 :軍½ 挲 • () · 9Ι 'S: 9ΐ ·' Ν 'Η 69 Ό: Thigh pull (%) 91' S: ΖΓ 'Ν: Ζ0 · 9' Η: 9 · 69 '3: ΐ!} ? θ ¥ ¾ 6 : Army 挲
•(s 'HI) '( 8"8 =Γ 'P ¾) Ζ8· '(ω ¾S) 'Ζ OX • (s' HI) '(8 "8 = Γ' P ¾) Ζ8 · '(ω ¾S)' Ζ OX
'(ZH 8*8 =1 'Ρ 'Μ) SZ' '(sjq ¾t) 6 9 '(zH 9'S =i 'Ηΐ) U ' '(ΖΗ 8'9 'Ζ'Ζ =1 'IP ¾2) 66 '(s ¾8) Of Z '(ZH Z Ί =i '1 'HZ) SO '(ZH 9 'Ζ=ί ' ^Q Ήΐ) 86 '(m 'HI) 06 '(m 'ΕΖ) 98 '(m Ήΐ) ΓΙ '(ZH 8*9 =ί '?uini)jq ¾) S I '(SJq ¾9) 62Ί '(ΖΗ 6·9 =ί 'HS) 68 ·0 Q : (zffl 008 <εΐθα3)漏- Ht '(ZH 8 * 8 = 1' Ρ 'Μ) SZ''(sjq ¾t) 6 9' (zH 9'S = i 'Ηΐ) U''(ΖΗ8'9'Ζ'Ζ = 1 'IP ¾2) 66 '(s ¾8) Of Z' (ZH Z Ί = i '1' HZ) SO '(ZH 9' Ζ = ί '^ Q Ήΐ) 86' (m 'HI) 06' (m 'ΕΖ) 98' ( m Ήΐ) ΓΙ '(ZH 8 * 9 = ί'? uini) jq ¾) SI '(SJq ¾9) 62Ί' (ΖΗ 6.9 = ί 'HS) 68 680 Q: (zffl 008 <ε ΐθα3) -H t
" (%) 'S :6Ζ·λ 'N :69 'S ¾ :99'69 Ό :蘄嗨牽 (%)"(%) 'S: 6Ζ · λ' N: 69 'S ¾: 99'69 Ό: 蘄 嗨 traction (%)
8 ·9ΐ 'S 'ΟΖ'ί 'Ν :0i S Ή :99·63 '3 : Ϊ^Ί-Ι Ί ? ss ¾6Z3 : 挲 8 · 9ΐ 'S'ΟΖ'ί'Ν: 0i S Ή: 99 · 63' 3: Ϊ ^ Ί-Ι Ί? S s ¾ 6Z 3: 挲
•(SJq 'Ηΐ) '6 '(ZH ΐ 'I'L =ί 'IP 'ΗΖ) 96' '(ΖΗ g'8 =f 'Ρ 'ΕΖ) 88 "A '^Η Γΐ ' =ί '11 'Ηΐ) 09 ·Ζ '(ZH S "i- • (SJq 'Ηΐ)' 6 '(ZH ΐ'I'L = ί 'IP' ΗΖ) 96 '' ( Ζ Η g'8 = f 'Ρ' ΕΖ) 88 "A '^ Η Γΐ' = ί ' 11 'Ηΐ) 09 · Ζ' (ZH S "i-
0t9t0/Z0dr/lDd 909Ζ60/Ζ0 OAV 実施例 4-337 0t9t0 / Z0dr / lDd 909Ζ60 / Ζ0 OAV Example 4-337
Ή-NMR (300 MHz, CDC13) : 50.89 (3H, t, J=7.0Hz), 1.24-1.37 (6H, m), 1.40-1.51 (2H, m), 2.05 (2H, br t, J=7.1Hz), 3.82 (3H, s), 4.19 (2H, s), 6.49 (IH, br s), 6.83-6.89 (2H, m) , 7.21-7.26 (2H, m), 7.43-7.50 (2H, m), 7.52-7.59 (1H, m), 7.90-7.95 (2H, m), 9.71 (IH, br s). Ή-NMR (300 MHz, CDC1 3): 50.89 (3H, t, J = 7.0Hz), 1.24-1.37 (6H, m), 1.40-1.51 (2H, m), 2.05 (2H, br t, J = 7.1Hz), 3.82 (3H, s), 4.19 (2H, s), 6.49 (IH, br s), 6.83-6.89 (2H, m), 7.21-7.26 (2H, m), 7.43-7.50 (2H, m), 7.52-7.59 (1H, m), 7.90-7.95 (2H, m), 9.71 (IH, br s).
元素分析値: C26H29N304S3として計算値: C; 57.43, H; 5.37, N; 7.72 (%)、 実 測値 C; 57.58, H; 5.30, N; 7.70 (%) . Elemental analysis: Calculated as C 26 H 29 N 3 0 4 S 3: C; 57.43, H; 5.37, N; 7.72 (%), actual Hakachi C; 57.58, H; 5.30, N; 7.70 (%) .
実施例 4-338 Example 4-338
Ή-N R (300 MHz, CDC13) : 50.89 (3H, t, J=7.0Hz), 1.24-1.35 (6H, m), 1.38-1.51 (2H, m), 2.05 (2H, br t, J=6.9Hz), 2.31 (3H, s), 4.21 (2H, s), 6.48 (IH, br s), 7.03-7.09 (2H, m), 7.30-7.36 (2H, m), 7.43-7.59 (3H, m), 7.90-7.96 (2H, m), 9.60-9.85 (IH, m). Ή-NR (300 MHz, CDC1 3): 50.89 (3H, t, J = 7.0Hz), 1.24-1.35 (6H, m), 1.38-1.51 (2H, m), 2.05 (2H, br t, J = 6.9Hz), 2.31 (3H, s), 4.21 (2H, s), 6.48 (IH, br s), 7.03-7.09 (2H, m), 7.30-7.36 (2H, m), 7.43-7.59 (3H, m), 7.90-7.96 (2H, m), 9.60-9.85 (IH, m).
元素分析値: C„H29N305S3として計算値: C; 56.72, H; 5.11, N; 7.34 (%), 実 測値 C; 56.61, H; 5.08, N; 7.42 (%) . Elemental analysis: C „H 29 N 3 0 5 S 3 Calculated: C; 56.72, H; 5.11, N; 7.34 (%), found C; 56.61, H; 5.08, N; 7.42 (%) .
実施例 4-339 Example 4-339
'H-NMR (300 MHz, CDC13) : 50.89 (3H, t, J=7.0Hz), 1.23-1.48 (8H, m), 2.03 (2H, br t, J=7.0Hz), 4.23 (2H, s), 6.46 (IH, br s), 7.42-7.53 (4H, m), 7.56-7.65 (3H, m), 7.91-7.96 (2H, m), 9.65-9.86 (IH, m). 'H-NMR (300 MHz, CDC1 3): 50.89 (3H, t, J = 7.0Hz), 1.23-1.48 (8H, m), 2.03 (2H, br t, J = 7.0Hz), 4.23 (2H, s), 6.46 (IH, br s), 7.42-7.53 (4H, m), 7.56-7.65 (3H, m), 7.91-7.96 (2H, m), 9.65-9.86 (IH, m).
元素分析値: CMH26N403S3として計算値: C; 57.96, H; 4.86, N; 10.40 ( ), 実 測値 C; 57.84, H; 4.98, N; 10.35 (%) . Elemental analysis: Calculated for C M H 26 N 4 0 3 S 3 : C; 57.96, H; 4.86, N; 10.40 (), found C; 57.84, H; 4.98, N; 10.35 (%).
実施例 4-340 Example 4-340
'H-NMR (300 MHz, CDC13) : 50.90 (3H, t, J=7.0Hz), 1.23-1.37 (6H, m), 1.40-1.56 (2H, m), 2.09 (2H, t, J=7.3Hz), 4.71 (2H, s), 6.52 (IH, s), 7.23-7.32 (2H, m), 7.40-7.59 (5H, m), 7.78-7.95 (4H, m), 7.99-8.03 (IH, m), 9.65-9.77 (IH, m). 'H-NMR (300 MHz, CDC1 3): 50.90 (3H, t, J = 7.0Hz), 1.23-1.37 (6H, m), 1.40-1.56 (2H, m), 2.09 (2H, t, J = 7.3Hz), 4.71 (2H, s), 6.52 (IH, s), 7.23-7.32 (2H, m), 7.40-7.59 (5H, m), 7.78-7.95 (4H, m), 7.99-8.03 (IH , m), 9.65-9.77 (IH, m).
元素分析値: C29H29N303S2として計算値: C; 61.78, H; 5.18, N; 7.45 (%), 実 測値 C; 61.39, H; 4.95, N; 7.47 (%) . Elemental analysis: C 29 H 29 N 3 0 3 S 2 Calculated: C; 61.78, H; 5.18 , N; 7.45 (%), actual Hakachi C; 61.39, H; 4.95, N; 7.47 (%) .
実施例 4-341 Example 4-341
'H-NMR (300 MHz, CDC13) : δ 0.88 (3Η, t, J=7.0Hz), 1.22-1.34 (6H, m), 1.37-1.49 (2H, m), 2.04 (2H, brt, J=7.0Hz), 4.39 (2H, s), 6.47 (1H, s), 7.39-7.57 (6H, m), 7.77-7.86 (4H, m), 7.90-7.95 (2H, m), 9.74 (1H, brs). 'H-NMR (300 MHz, CDC1 3): δ 0.88 (3Η, t, J = 7.0Hz), 1.22-1.34 (6H, m), 1.37-1.49 (2H, m), 2.04 (2H, brt, J = 7.0Hz), 4.39 (2H, s), 6.47 (1H, s), 7.39-7.57 (6H, m), 7.77-7.86 (4H, m), 7.90-7.95 (2H, m), 9.74 (1H, brs).
元素分析値: C29H29N303S3として計算値: C; 61.78, H; 5.18, N; 7.45 (%), 実 測値 C; 61.92, H; 5.25, N; 7.47 (%) . Elemental analysis: Calculated as C 29 H 29 N 3 0 3 S 3: C; 61.78, H; 5.18, N; 7.45 (%), actual Hakachi C; 61.92, H; 5.25, N; 7.47 (%) .
実施例 4-342 Example 4-342
Ή-NMR (300 MHz, CDC13) : ά 0.89 (3Η, t, J=7.0Hz), 1.24-1.51 (8H, m), 2.06 (2H, brt, J=7.2Hz), 3.17 (2H, t, J=6.0Hz), 3.85 (2H, t, J=6.0Hz), 6.50 (1H, brs), 7.46-7.61 (3H, m), 7.94-7.99 (2H, m), 9.50-10.0 (1H, m) . Ή-NMR (300 MHz, CDC1 3): ά 0.89 (3Η, t, J = 7.0Hz), 1.24-1.51 (8H, m), 2.06 (2H, brt, J = 7.2Hz), 3.17 (2H, t , J = 6.0Hz), 3.85 (2H, t, J = 6.0Hz), 6.50 (1H, brs), 7.46-7.61 (3H, m), 7.94-7.99 (2H, m), 9.50-10.0 (1H, m).
実施例 4-343 Example 4-343
Ή-NMR (300 MHz, CDC13) : δ 0.90 (3H, t, J=6.9Hz), 1.22-1.38 (6H, m), 1.41-1.53 (2H, m), 2.09 (3H, brt, J=7.3Hz), 2.34 (3H, s), 4.71 (2H, s), 6.51 (1H, brs), 7.08 (1H, brd, J=7.4Hz), 7.20 (1H, brt, J=7.7Hz), 7.34-7.59 (5H, m), 7.82-8.01 (4H, m). Ή-NMR (300 MHz, CDC1 3): δ 0.90 (3H, t, J = 6.9Hz), 1.22-1.38 (6H, m), 1.41-1.53 (2H, m), 2.09 (3H, brt, J = (7.3Hz), 2.34 (3H, s), 4.71 (2H, s), 6.51 (1H, brs), 7.08 (1H, brd, J = 7.4Hz), 7.20 (1H, brt, J = 7.7Hz), 7.34 -7.59 (5H, m), 7.82-8.01 (4H, m).
実施例 4-344 Example 4-344
Ή-NMR (300 MHz, CDC13) : 60.89 (3H, t, J=7.0Hz), 1.22-1.53 (8H, m), 2.06 (2H, t, J=7.1Hz), 2.47 (6H, s), 2.84 (2H, t, J=7.9Hz), 3.23 (2H, t, J=7.9Hz), 6.48 (1H, br s), 7.25-7.34 (2H, m), 7.45 (1H, ddd, J=7.5, 7.5, 1.2Hz), 8.01 (1H, dd, J=7.6, 1.6Hz). Ή-NMR (300 MHz, CDC1 3): 60.89 (3H, t, J = 7.0Hz), 1.22-1.53 (8H, m), 2.06 (2H, t, J = 7.1Hz), 2.47 (6H, s) , 2.84 (2H, t, J = 7.9Hz), 3.23 (2H, t, J = 7.9Hz), 6.48 (1H, br s), 7.25-7.34 (2H, m), 7.45 (1H, ddd, J = 7.5, 7.5, 1.2Hz), 8.01 (1H, dd, J = 7.6, 1.6Hz).
実施例 4-345 Example 4-345
Ή-NMR (300 MHz, DMSO) : 50.86 (3H, t, J=6.8Hz), 1.20-1.47 (8H, m), 2.15 (2H, t, J=6.8Hz), 2.64-2.71 (2H, m), 2.66 (3H, s), 3.16-3.23 (2H, m), 6.62 (1H, s), 7.34-7.42 (2H, m), 7.48-7.56 (1H, in), 8.04-8.11 (1H, m), 11.5-11.7 (1H, m). NMR-NMR (300 MHz, DMSO): 50.86 (3H, t, J = 6.8Hz), 1.20-1.47 (8H, m), 2.15 (2H, t, J = 6.8Hz), 2.64-2.71 (2H, m ), 2.66 (3H, s), 3.16-3.23 (2H, m), 6.62 (1H, s), 7.34-7.42 (2H, m), 7.48-7.56 (1H, in), 8.04-8.11 (1H, m ), 11.5-11.7 (1H, m).
実施例 4-346 Example 4-346
Ή-NMR (300 MHz, CDC13) : 50.89 (3H, t, J=6.8Hz), 1.23-1.50 (8H, m), 2.05 (2H, brt, J=7.3Hz), 2.66 (3H, s), 4.23 (2H, s), 6.48 (1H, brs), 7.22-7.37 (7H, m), 7.44 (1H, br t, J=7.4Hz), 7.99 (1H, brd, J=7.9Hz), 9.71-9.76 (1H, m). Ή-NMR (300 MHz, CDC1 3): 50.89 (3H, t, J = 6.8Hz), 1.23-1.50 (8H, m), 2.05 (2H, brt, J = 7.3Hz), 2.66 (3H, s) , 4.23 (2H, s), 6.48 (1H, brs), 7.22-7.37 (7H, m), 7.44 (1H, brt, J = 7.4Hz), 7.99 (1H, brd, J = 7.9Hz), 9.71 -9.76 (1H, m).
元素分析値: C26H29N3O3S3'0.25¾Oとして計算値: C; 58.67, H; 5.59, N; 7.90(%)> 実測値 C; 58.66, H; 5.54, N; 7.97 (%) . Elemental analysis: Calculated as C 26 H 29 N 3 O 3 S 3 '0.25¾O: C; 58.67, H; 5.59, N; 7.90 (%)> Found C; 58.66, H; 5.54, N; 7.97 (%).
実施例 4-347 Example 4-347
Ή-NMR (300 MHz, CDC13) : 50.89 (3H, t, J=6.9Hz), 1.22-1.50 (8H, m), 2.05 (2H, br t, J=7.1Hz), 2.69 (3H, s), 4.18 (2H, s), 6.47 (IH, brs), 7.18-7.33 (6H, m), 7.45 (1H, ddd, J=7.4, 7.4, 1.1Hz), 8.00 (IH, dd, J=7.4, 1.4Hz), 9.74 (IH, brs). Ή-NMR (300 MHz, CDC1 3): 50.89 (3H, t, J = 6.9Hz), 1.22-1.50 (8H, m), 2.05 (2H, br t, J = 7.1Hz), 2.69 (3H, s ), 4.18 (2H, s), 6.47 (IH, brs), 7.18-7.33 (6H, m), 7.45 (1H, ddd, J = 7.4, 7.4, 1.1Hz), 8.00 (IH, dd, J = 7.4) , 1.4Hz), 9.74 (IH, brs).
元素分析値: C26H26N303S3C1 として計算値: C; 55.75, H; 4.68, N; 7.50 (%), 実測値 C; 55.70, H; 4.87, N; 7.49 (%) . Elemental analysis: C 26 H 26 N 3 0 3 S 3 C1 Calculated: C; 55.75, H; 4.68 , N; 7.50 (%), Found C; 55.70, H; 4.87, N; 7.49 (%) .
実施例 4-348 Example 4-348
Ή-NMR (CDC13) : 50.88 (3H, t, J=7.0Hz), 1.20-1.50 (8H, m), 2.05 (2H, br t, J=7.1Hz), 2.65 (3H, s), 4.40 (2H, s), 6.47 (IH, brs), 7.20-7.31 (2H, m), 7.38-7.46 (2H, m), 7.48-7.55 (2H, m), 7.77-7.86 (4H, in), 7.99 (IH, dd, J=8.9, 1.0Hz) , 9.74 (IH, brs). Ή-NMR (CDC1 3): 50.88 (3H, t, J = 7.0Hz), 1.20-1.50 (8H, m), 2.05 (2H, br t, J = 7.1Hz), 2.65 (3H, s), 4.40 (2H, s), 6.47 (IH, brs), 7.20-7.31 (2H, m), 7.38-7.46 (2H, m), 7.48-7.55 (2H, m), 7.77-7.86 (4H, in), 7.99 (IH, dd, J = 8.9, 1.0Hz), 9.74 (IH, brs).
元素分析値: C3。H31N303S3として計算値: C; 62.36, H; 5.41, ; 7.27 (%), 実 測値 C; 62.30, H; 5.38, N; 7.26 (%) . Elemental analysis: C 3. Calculated as H 31 N 3 0 3 S 3 : C; 62.36, H; 5.41,; 7.27 (%), actual Hakachi C; 62.30, H; 5.38, N; 7.26 (%).
実施例 4-349 Example 4-349
'H-NMR (CDC13) :50.89 (3H, t, J=7.0Hz), 1.20-1.44 (8H, in), 1.97 (2H, br t, J=7.0Hz), 2.64 (3H, s), 4.18 (2H, s), 6.43 (IH, brs), 7.22-7.48 (12H, m), 8.00 (IH, brd, J=7.7Hz), 9.63-9.77 (IH, m). 'H-NMR (CDC1 3) : 50.89 (3H, t, J = 7.0Hz), 1.20-1.44 (8H, in), 1.97 (2H, br t, J = 7.0Hz), 2.64 (3H, s), 4.18 (2H, s), 6.43 (IH, brs), 7.22-7.48 (12H, m), 8.00 (IH, brd, J = 7.7Hz), 9.63-9.77 (IH, m).
実施例 4-350 Example 4-350
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 0.89 (3Η, t, J= 7.2 Hz), 1.03 (3H, t, J= 7.2 Hz), 1.29 (4H, brs), 1.32 (2H, m), 1.47 (2H, brquint, J= 7.2 Hz), 'H-NMR (300 MHz, in double-mouthed form): δ 0.89 (3Η, t, J = 7.2 Hz), 1.03 (3H, t, J = 7.2 Hz), 1.29 (4H, brs), 1.32 (2H , m), 1.47 (2H, brquint, J = 7.2 Hz),
1.73 (2H, sixtet, J= ,7.5 Hz), 2.07 (2H, t, J= 7.4 Hz) , 2.96 (2H, t, J= 7.3 Hz), 6.51 (1H, brs), 7.02 (2H, d, ]= 8.7 Hz), 7.06 (2H, brd, J= 7.9 Hz), 7.21 (IH, t, J= 7.2 Hz), 7.40 (2H, t, J= 8.3 Hz), 7.91 (2H, dt, J= 8.7, 1.5 Hz),1.73 (2H, sixtet, J =, 7.5 Hz), 2.07 (2H, t, J = 7.4 Hz), 2.96 (2H, t, J = 7.3 Hz), 6.51 (1H, brs), 7.02 (2H, d, ] = 8.7 Hz), 7.06 (2H, brd, J = 7.9 Hz), 7.21 (IH, t, J = 7.2 Hz), 7.40 (2H, t, J = 8.3 Hz), 7.91 (2H, dt, J = 8.7, 1.5 Hz),
9.74 (IH, brs). 9.74 (IH, brs).
実施例 4-351 Example 4-351
Ή-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J- 7.1 Hz), 1.29 (2H, m), 1.30 (2H, m), 1.31 (2H,' m), 1.43 (2H, brquint, J= 6.8 Hz), 2.09 (2H, brdd, J= 7.9, 7.2 Hz), 2.24 (3H, s), 2.40 (3H, s), 4.20 (2H, s) , 7.24 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J= 8.3 Hz), 9.62 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.90 (3H, t, J- 7.1 Hz), 1.29 (2H, m), 1.30 (2H, m), 1.31 (2H, 'm), 1.43 (2H, brquint , J = 6.8 Hz), 2.09 (2H, brdd, J = 7.9, 7.2 Hz), 2.24 (3H, s), 2.40 (3H, s), 4.20 (2H, s), 7.24 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J = 8.3 Hz), 9.62 (1H, br).
元素分析値: C27H31N303S3として計算値: C, 59.86; H, 5.77; N, 7.76; S, 17.76 (%)、 実測値: C, 59.99; H, 5.67; N, 7.73; S, 17.95 ( ) . Elemental analysis: C 27 H 31 N 3 0 3 S 3 Calculated: C, 59.86; H, 5.77; N, 7.76; S, 17.76 (%), found: C, 59.99; H, 5.67; N, 7.73; S, 17.95 ().
実施例 4-352 Example 4-352
酢酸ェチル -ジィソプロピルエーテルから再結晶した。 Recrystallized from ethyl acetate-disopropyl ether.
融点 112-114°C . 赤外吸収スぺクトル(IR) (KBr中) リ: 3240, 2957, 2930, 2870,Melting point 112-114 ° C. Infrared absorption spectrum (IR) (in KBr) Re: 3240, 2957, 2930, 2870,
2857, 1732, 1657, 1615, 1557, 1495 cm"1. !H-NMR (CDC13) : δ 0.892 (3Η, t, J=6.6 .! 2857, 1732, 1657, 1615, 1557, 1495 cm "1 H-NMR (CDC1 3): δ 0.892 (3Η, t, J = 6.6
Hz), 1.03 (6H, d, J-6.6 Hz), 1.20-1.54 (8H, m), 1.82-1.98 (1H, m), 2.11 (2H, t, J=8.0 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, t, J=7.0 Hz), 7.28 (2H, d, J=8.0 Hz), 7.86 (2H, d, J=8.4 Hz), 9.62 (1H, bs). Hz), 1.03 (6H, d, J-6.6 Hz), 1.20-1.54 (8H, m), 1.82-1.98 (1H, m), 2.11 (2H, t, J = 8.0 Hz), 2.26 (3H, s ), 2.41 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.62 (1H, bs ).
元素分析値: C24H33N303S3として計算値: C, 56.77; H, 6.55; N, 8.28; S, 18.95 (%)、 実測値: C, 56.88; H, 6.42; N, 8.44; S, 19.03 (%) . Elemental analysis: C 24 H 33 N 3 0 3 S 3 Calculated: C, 56.77; H, 6.55 ; N, 8.28; S, 18.95 (%), Found: C, 56.88; H, 6.42 ; N, 8.44; S, 19.03 (%).
実施例 4-353 Example 4-353
融点 144- 6°C Melting point 144-6 ° C
赤外吸収スペクトル(IR) (KBr中) V 3240, 2957, 2930, 2857, 1734, 1694, 1659,Infrared absorption spectrum (IR) (in KBr) V 3240, 2957, 2930, 2857, 1734, 1694, 1659,
1611, 1563, 1495 cm"1. 1611, 1563, 1495 cm " 1 .
'H-NMR (CDC13) : δ 0.892 (3Η, t, J=6.2Hz), 0.912 (3H, t, J=7.0Hz), 1.24-1.78 (14H, m), 2.11 (2H, t, J=8.4 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J=7.8 Hz), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.2 Hz), 9.62 (1H, bs). 元素分析値: C25H35N303S3として計算値: C, 57.55; H, 6.70; N, 8.05; S, 18.44 . (%)、 実測値: C, 57.55; H, 6.58; N, 8.17; S, 18.38 (%) . 'H-NMR (CDC1 3) : δ 0.892 (3Η, t, J = 6.2Hz), 0.912 (3H, t, J = 7.0Hz), 1.24-1.78 (14H, m), 2.11 (2H, t, J = 8.4 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J = 7.8 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.62 (1H , bs) elemental analysis: C 25 H 35 N 3 0 3 S calculated 3:.. C, 57.55; H, 6.70; N, 8.05; S, 18.44 (%), Found: C, 57.55; H, 6.58; N, 8.17; S, 18.38 (%).
実施例 4-354 Example 4-354
Ή-腿 (CDC13, 200 MHz): δ 1.02 (6H, d, J= 6.6 Hz), 1.09 (3H, t, J= 7.4 Hz), 1.91 (1H, quint, J= 6.6 Hz), 2.18 (2H, q, J= 7.4 Hz), 2.27 (3H, s), 2.83 (2H, d, J= 7.0 Hz), 2.95 (1H, quint, J= 7.0 Hz), 7.50 (3H, m), 7.99 (2H, dm, J= 8.4 Hz), 9.65 (1H, brs). Ή- thigh (CDC1 3, 200 MHz): δ 1.02 (6H, d, J = 6.6 Hz), 1.09 (3H, t, J = 7.4 Hz), 1.91 (1H, quint, J = 6.6 Hz), 2.18 ( 2H, q, J = 7.4 Hz), 2.27 (3H, s), 2.83 (2H, d, J = 7.0 Hz), 2.95 (1H, quint, J = 7.0 Hz), 7.50 (3H, m), 7.99 ( 2H, dm, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C19H23N303S3として計算値: C, 52.15; H, 5.30; N, 9.60; S, 21.98 (%), 実測値: C, 52.04; H, 5.17; N, 9.61; S, 22.01 (%) · 実施例 4-355 Elemental analysis: C 19 H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30; N, 9.60; S, 21.98 (%), found: C, 52.04; H, 5.17; N, 9.61; S, 22.01 (%) Example 4-355
¾-NMR (CDC13, 200 MHz): δ 1.09 (3H, t, J= 7.4 Hz), 2.14 (2H, q, J= 7.2 Hz), 2.27 (3H, s), 2.98 (2H, dd, J= 8.4, 5.6 Hz), 3.20 (2H, dd, J= 8.8, 6.2 Hz), 7.17-34 (5H, m), 7.50 (3H, m), 7.95 (2H, dm, J= 8.6 Hz), 9.66 (1H, brs). 元素分析値: C23 N303S3として計算値: C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%), 実測値: C, 56.74; H, 4.72; N, 8.58; S, 19.72 (%) . ¾-NMR (CDC1 3, 200 MHz): δ 1.09 (3H, t, J = 7.4 Hz), 2.14 (2H, q, J = 7.2 Hz), 2.27 (3H, s), 2.98 (2H, dd, J = 8.4, 5.6 Hz), 3.20 (2H, dd, J = 8.8, 6.2 Hz), 7.17-34 (5H, m), 7.50 (3H, m), 7.95 (2H, dm, J = 8.6 Hz), 9.66 (1H, brs). Elemental analysis: Calculated for C 23 N 3 0 3 S 3 : C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%), found: C, 56.74; H, 4.72; N, 8.58; S, 19.72 (%).
実施例 4-356 Example 4-356
-醒 (CDC13, 200 MHz): δ 1.08 (3Η, t, J= 7.5 Hz), 1.43 (4H, m), 1.50-1.75 (4H, m), 2.13 (2H, q, J= 7.8 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, brt, J= 7.3 Hz), 3.66 (2H, brt, J= 6.2 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). - Awakening: (CDC1 3, 200 MHz): δ 1.08 (3Η, t, J = 7.5 Hz), 1.43 (4H, m), 1.50-1.75 (4H, m), 2.13 (2H, q, J = 7.8 Hz) , 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, brt, J = 7.3 Hz), 3.66 (2H, brt, J = 6.2 Hz), 7.28 (2H, d, J = 8.0 Hz) , 7.85 (2H, d, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C22H29N304S3として計算値: C, 53.31; H, 5.90; , 8.48; S, 19.41Elemental analysis: Calculated as C 22 H 29 N 3 0 4 S 3: C, 53.31; H, 5.90;, 8.48; S, 19.41
(%)、 実測値: C, 53.16; H, 5.87; , 8.60; S, 19.28 ( ) . (%), Found: C, 53.16; H, 5.87;, 8.60; S, 19.28 ().
実施例 4-357 Example 4-357
^-NMR (CDCI3, 200 MHz): δ 1.09 (3H, t, J= 7.2 Hz), 2.14 (2H, q, J= 7.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, brdd, J= 8.8, 5.4 Hz), 3.18 (2H, brdd, J= 8.8, 5.4 Hz), 7.17-35 (7H, m), 7.83 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). ^ -NMR (CDCI3, 200 MHz): δ 1.09 (3H, t, J = 7.2 Hz), 2.14 (2H, q, J = 7.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, brdd, J = 8.8, 5.4 Hz), 3.18 (2H, brdd, J = 8.8, 5.4 Hz), 7.17-35 (7H, m), 7.83 (2H, d, J = 8.4 Hz), 9.63 ( 1H, brs).
元素分析値: C24H25N303S3として計算値: C, 57.69; H, 5.04; N, 8.41; S, 19.25 (%)、 実測値: C, 57.80; H, 5.07; N, 8.52; S, 19.09 (%) . Elemental analysis: C 24 H 25 N 3 0 3 S 3 Calculated: C, 57.69; H, 5.04 ; N, 8.41; S, 19.25 (%), Found: C, 57.80; H, 5.07 ; N, 8.52; S, 19.09 (%).
実施例 4-358 Example 4-358
lH-NMR (CDCI3, 200 MHz): d 1.07 (3H, t, J= 7.4 Hz), 2.11 (2H, q, J= 7.4 Hz), 2.15 (2H, quint, J= 7.0 Hz), 2.25 (3H, s), 2.38 (3H, s), 3.09 (2H, t, J= 7.0 Hz), 3.14 (2H, t, J= 7.0 Hz), 7.24 (2H, d, J= 8.4 Hz), 7.49 (2H, brt, J= 7.8 Hz), 7.59 (1H,. brt, J= 7.4 Hz), 7.84 (2H, d, J= 8.0 Hz), 7.96 (2H, dm,; J= 8.0 Hz), 9.65 (1H, brs). lH-NMR (CDCI3, 200 MHz): d 1.07 (3H, t, J = 7.4 Hz), 2.11 (2H, q, J = 7.4 Hz), 2.15 (2H, quint, J = 7.0 Hz), 2.25 (3H , S), 2.38 (3H, s), 3.09 (2H, t, J = 7.0 Hz), 3.14 (2H, t, J = 7.0 Hz), 7.24 (2H, d, J = 8.4 Hz), 7.49 (2H , brt, J = 7.8 Hz), 7.59 (1H, .brt, J = 7.4 Hz), 7.84 (2H, d, J = 8.0 Hz), 7.96 (2H, dm ,; J = 8.0 Hz), 9.65 (1H , brs).
元素分析値: C26H27N304S3として計算値: C, 57.65; H, 5.02; N, 7.76; S, 17.76Elemental analysis: C 26 H 27 N 3 0 4 S 3 Calculated: C, 57.65; H, 5.02 ; N, 7.76; S, 17.76
(%)、 実測値: C, 57.49; H, 5.10; N, 7.93; S, 17.61 (%) . (%), Found: C, 57.49; H, 5.10; N, 7.93; S, 17.61 (%).
実施例 4-359 一 NMR (200MHz, CDC13) : δ 1.09 (3H, t, J= 7.5 Hz), 2.13 (2H, q, J= 7.5 Hz), 2.26 (3H, s), 2.34 (3H, s), 2.42 (3H, s), 2.94 (2H, dd , J= 8.8, 7.4 Hz), 3.16 (2H, dd, J= 8.8, 6.2 Hz), 7.10 (2H, d, J二 8.6 Hz), 7.11 (2H, d, J= 8.8 Hz) , 7.28 (2H, d, J= 8.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Example 4-359 One NMR (200MHz, CDC1 3): δ 1.09 (3H, t, J = 7.5 Hz), 2.13 (2H, q, J = 7.5 Hz), 2.26 (3H, s), 2.34 (3H, s), 2.42 ( 3H, s), 2.94 (2H, dd, J = 8.8, 7.4 Hz), 3.16 (2H, dd, J = 8.8, 6.2 Hz), 7.10 (2H, d, J2 8.6 Hz), 7.11 (2H, d , J = 8.8 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C25H27N303S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73 ( ) 、 実測値: C, 58.44; H, 5.34; N, 8.12; S, 18.71 ( ) . Elemental analysis: C 25 H 27 N 3 0 3 S 3 Calculated: C, 58.45; H, 5.30 ; N, 8.18; S, 18.73 (), Found: C, 58.44; H, 5.34 ; N, 8.12 S, 18.71 ().
融点: 133.5-135.0°C Melting point: 133.5-135.0 ° C
実施例 4-360 Example 4-360
¾-NMR (200MHz, CDC13) : 6 1.09 (3H, t, J= 7.5 Hz), 2.13 (2H, q, J= 7.5 Hz), 2.26 (3H, s), 2.43 (3H, s), 2.95 (2H, dd, J= 7.6, 7.0 Hz), 3.17 (2H, dd, J= 8.3, 6.8 Hz), 7.12 (2H, d, J= 8.1 Hz), 7.27 (4H, m), 7.81 (2H, d, J= 8.1 Hz), 9.63 (1H, brs). ¾-NMR (200MHz, CDC1 3 ): 6 1.09 (3H, t, J = 7.5 Hz), 2.13 (2H, q, J = 7.5 Hz), 2.26 (3H, s), 2.43 (3H, s), 2.95 (2H, dd, J = 8.3, 6.8 Hz), 7.12 (2H, d, J = 8.1 Hz), 7.27 (4H, m), 7.81 (2H, dd, J = 7.6, 7.0 Hz) d, J = 8.1 Hz), 9.63 (1H, brs).
元素分析値: C24H24N303S3C1として計算値: C, 53.97; H, 4.53; N, 7.87; S, 18.01; CI, 6.64 (%) 、 実測値: C, 53.84; H, 4.46; N, 7.88; S, 17.84; CI, 6.84 (%) . 融点. 174.5-176.0°C Elemental analysis: Calculated as C 24 H 24 N 3 0 3 S 3 C1: C, 53.97; H, 4.53; N, 7.87; S, 18.01; CI, 6.64 (%), Found: C, 53.84; H , 4.46; N, 7.88; S, 17.84; CI, 6.84 (%). Melting point. 174.5-176.0 ° C
実施例 4-361 Example 4-361
Ή— NMR (200MHz, CDC13) : 6 1.09 (3H, t, J= 7.5 Hz), 2.14 (3H, q, J= 7.4 Hz), 2.26 (3H, s), 2.43 (3H, s), 2.94 (1H, brdd, J= 8.0, 6.8 Hz), 3.16 (2H, dd, J= 8.2, 6.4 Hz), 6.98 (2H, d, J= 8.5 Hz), 7.14 (2H, brt, J= 8.5 Hz), 7.27 (2H, d, J= 8.4 Hz), 7.82 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή- NMR (200MHz, CDC1 3) : 6 1.09 (3H, t, J = 7.5 Hz), 2.14 (3H, q, J = 7.4 Hz), 2.26 (3H, s), 2.43 (3H, s), 2.94 (1H, brdd, J = 8.0, 6.8 Hz), 3.16 (2H, dd, J = 8.2, 6.4 Hz), 6.98 (2H, d, J = 8.5 Hz), 7.14 (2H, brt, J = 8.5 Hz) , 7.27 (2H, d, J = 8.4 Hz), 7.82 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C24H24N303S3Fとして計算値: C, 55.68; H, 4.67; N, 8.12; S, 18.58; F, 3.67 (%) 、 実測値: C, 55.73; H, 4.40; N, 8.03; S, 18.33; F, 3.68 ( ) · 融点. 164.5-165.5t: Elemental analysis: C 24 H 24 N 3 0 3 S 3 F Calculated: C, 55.68; H, 4.67 ; N, 8.12; S, 18.58; F, 3.67 (%), Found: C, 55.73; H , 4.40; N, 8.03; S, 18.33; F, 3.68 ()
実施例 4-362 Example 4-362
'H-NMR (200MHz, CDC13) : δ 1.09 (3H, t, J= 7.4 Hz), 2.13 (2H, q, J= 7.4 Hz),'H-NMR (200MHz, CDC1 3): δ 1.09 (3H, t, J = 7.4 Hz), 2.13 (2H, q, J = 7.4 Hz),
2.26 (3H, s), 2.42 (3H, s), 2.91 (2H, dd, J= 8.6, 7.2 Hz), 3.16 (2H, dd, J= 8.8, 6.6 Hz), 3.80 (3H, s), 6.85 (2H, d, J= 8.4 Hz), 7.12 (2H, d, J= 8.8 Hz),2.26 (3H, s), 2.42 (3H, s), 2.91 (2H, dd, J = 8.6, 7.2 Hz), 3.16 (2H, dd, J = 8.8, 6.6 Hz), 3.80 (3H, s), 6.85 (2H, d, J = 8.4 Hz), 7.12 (2H, d, J = 8.8 Hz),
7.27 (2H, d, J= 8.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). 7.27 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C25H27N304S3として計算値: C, 56.69; H, 5.14; N, 7.93; S, 18.16 ( ) 、 実測値: C, 56.59; H, 5.27; N, 7.88; S, 18.14 (%) . 融点. 163.5-174.0°C Elemental analysis: Calculated as C 25 H 27 N 3 0 4 S 3: C, 56.69; H, 5.14; N, 7.93; S, 18.16 (), Found: C, 56.59; H, 5.27; N, 7.88; S, 18.14 (%). Melting point. 163.5-174.0 ° C
実施例 4-363 Example 4-363
Ή-NMR (200MHz, CDC13) : δ 1.09 (3H, t, J= 7.4 Hz), 1.75 (2H, brq, J= 9.0 Hz), 1.88 (2H, ni), 2.13 (2H, brq, J= 7.3 Hz), 2.15 (2H, m), 2.26 (3H, s), 2.41 (3H, s), 2.57 (1H, quint, J= 7.7 Hz), 3.04 (2H, d, J= 7.8 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J- 8.0 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.09 (3H, t, J = 7.4 Hz), 1.75 (2H, brq, J = 9.0 Hz), 1.88 (2H, ni), 2.13 (2H, brq, J = 7.3 Hz), 2.15 (2H, m), 2.26 (3H, s), 2.41 (3H, s), 2.57 (1H, quint, J = 7.7 Hz), 3.04 (2H, d, J = 7.8 Hz), 7.27 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J- 8.0 Hz), 9.62 (1H, brs).
元素分析値: C21 N303S3として計算値: C, 54.40; H, 5.43; N, 9.06; S, 20.75 (%) 、 実測値: C, 54.17; H, 5.56; N, 9.12; S, 20.48 (%) . Elemental analysis: Calculated for C 21 N 3 0 3 S 3 : C, 54.40; H, 5.43; N, 9.06; S, 20.75 (%), found: C, 54.17; H, 5.56; N, 9.12; S, 20.48 (%).
融点. 156.5-157.0°C Melting point.156.5-157.0 ° C
実施例 4-364 Example 4-364
Ή一 NMR (200MHz, CDC13) : δ 0.95 (2Η, brq, J= 12.0 Hz), 1.08 (3H, t, J= 7.3 Hz), 1.19 (2H, m), 1.56 (1H, m), 1.61 (2H, m) , 1.71 (2H, m), 2.13 (2H, q, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, d, J= 7.0 Hz), 7.27 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). Ή one NMR (200MHz, CDC1 3): δ 0.95 (2Η, brq, J = 12.0 Hz), 1.08 (3H, t, J = 7.3 Hz), 1.19 (2H, m), 1.56 (1H, m), 1.61 (2H, m), 1.71 (2H, m), 2.13 (2H, q, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.82 (2H, d, J = 7.0 Hz) , 7.27 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.0 Hz), 9.63 (1H, brs).
元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56Elemental analysis: C 23 H 29 N 3 0 3 S 3 Calculated: C, 56.18; H, 5.94 ; N, 8.55; S, 19.56
(%) 、 実測値: C, 56.10; H, 5.93; N, 8.56; S, 19.59 (%) . (%), Found: C, 56.10; H, 5.93; N, 8.56; S, 19.59 (%).
融点. 161.0-161.5°C Melting point.161.0-161.5 ° C
実施例 4-365 Example 4-365
Ή-NMR (200MHz, CDC13) : δ 1.09 (3H, t, J= 7.4 Hz), 1.10 (2H, m), 1.50-1.90 (9H, m), 2.14 (2H, brq, J= 7.4 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.65 (IH, brs). 元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; , 8.55; S, 19.56 (%) 、 実測値: C, 56.19; H, 5.94; N, 8.62; S, 19.64 (%) . Ή-NMR (200MHz, CDC1 3 ): δ 1.09 (3H, t, J = 7.4 Hz), 1.10 (2H, m), 1.50-1.90 (9H, m), 2.14 (2H, brq, J = 7.4 Hz) , 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz) , 9.65 (IH, brs) elemental analysis: C 23 H 29 N 3 0 3 S 3 calculated:. C, 56.18; H, 5.94;, 8.55; S, 19.56 (%), Found: C, 56.19 H, 5.94; N, 8.62; S, 19.64 (%).
融点. 161.0- 161.5°C Melting point.161.0- 161.5 ° C
実施例 4-366 Example 4-366
Ή-NMR (200MHz, CDC13) : δ 1.08 (2H, m), 1.08 (3H, t, J= 7.5 Hz), 1.42 (2H, m), 1.50-1.80 (9H, m), 2.13 (2H, brq, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J= 7.4 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.08 (2H, m), 1.08 (3H, t, J = 7.5 Hz), 1.42 (2H, m), 1.50-1.80 (9H, m), 2.13 (2H, brq, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.94 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C24H31N303S3として計算値: C, 57.00; H, 6.18; N, 8.31; S, 19.02Elemental analysis: Calculated as C 24 H 31 N 3 0 3 S 3: C, 57.00; H, 6.18; N, 8.31; S, 19.02
(%) 、 実測値: C, 56.90; H, 6.11; N, 8.07; S, 19.09 (%) . (%), Found: C, 56.90; H, 6.11; N, 8.07; S, 19.09 (%).
融点. - 158.5-159. O Melting point.- 158.5-159.O
実施例 4-367 Example 4-367
¾-NMR (200MHz, CDC13) : δ 1.09 (3H, t, J= 7.5 Hz), 1.25 (2H, m), 1.59 (4H, m), 1.85 (2H, m), 2.12 (1H, quint, J= 7.3 Hz), 2.13 (2H, brq, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, d, J= 7.3 Hz), 7.27 (2H, d, J= 8.2 Hz), 7.86 (2H, d, J= 8.2 Hz), 9.60 (1H, brs). ¾-NMR (200MHz, CDC1 3 ): δ 1.09 (3H, t, J = 7.5 Hz), 1.25 (2H, m), 1.59 (4H, m), 1.85 (2H, m), 2.12 (1H, quint, J = 7.3 Hz), 2.13 (2H, brq, J = 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.95 (2H, d, J = 7.3 Hz), 7.27 (2H, d, J = 8.2 Hz), 7.86 (2H, d, J = 8.2 Hz), 9.60 (1H, brs).
元素分析値: C22H27N303S3として計算値: C, 55.32; H, 5.70; , 8.80; S, 20.14 (%) 、 実測値: C, 55.18; H, 5.70; N, 8.76; S, 20.15 ( ) . Elemental analysis: C 22 H 27 N 3 0 3 S 3 Calculated: C, 55.32; H, 5.70 ;, 8.80; S, 20.14 (%), Found: C, 55.18; H, 5.70 ; N, 8.76 ; S, 20.15 ().
融点. 168.0-169.5°C Melting point.168.0-169.5 ° C
実施例 4-368 Example 4-368
Ή-NMR (200MHz, CDC13) : δ 1.09 (3H, t, J= 7.4 Hz), 2.13 (2H, q, J= 7.5 Hz), 2.27 (3H, s), 2.42 (3H, s), 2.47 (3H, s), 7.29 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.63 (1H, br). Ή-NMR (200MHz, CDC1 3 ): δ 1.09 (3H, t, J = 7.4 Hz), 2.13 (2H, q, J = 7.5 Hz), 2.27 (3H, s), 2.42 (3H, s), 2.47 (3H, s), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, br).
元素分析値: C17H19N303S3として計算値: C, 49.86; H, 4.86; N, 10.26; S, 23.49Elemental analysis: C 17 H 19 N 3 0 3 S 3 Calculated: C, 49.86; H, 4.86 ; N, 10.26; S, 23.49
(%) 、 実測値: C, 50.02; H, 4.82; N, 10.02; S, 23.42 {%) . (%), Found: C, 50.02; H, 4.82; N, 10.02; S, 23.42 (%).
融点. 205.0- 206.0°C Melting point.205.0-206.0 ° C
実施例 4-369 Example 4-369
'H-NMR (200MHz, CDC13) : δ 0.93 (6H, d, J= 6.2 Hz), 1.09 (3H, t, J= 7.5 Hz), 1.58 (2H, quint, J= 7.0 Hz), 1.70 (1H, quint, J= 6.6 Hz), 2.14 (2H, q, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J= 7.7 Hz), 7.28 (2H, d, J- 8. Hz), 7.85 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 0.93 (6H, d, J = 6.2 Hz), 1.09 (3H, t, J = 7.5 Hz), 1.58 (2H, quint, J = 7.0 Hz), 1.70 ( 1H, quint, J = 6.6 Hz), 2.14 (2H, q, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.7 Hz), 7.28 ( 2H, d, J- 8. Hz), 7.85 (2H, d, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C21H27N303S3として計算値: C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%) 、 実測値: C, 54.07; H, 5.87; N, 9.03; S, 20.50 ( ) . Elemental analysis: Calculated for C 21 H 27 N 3 0 3 S 3 : C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), found: C, 54.07; H, 5.87; N, 9.03; S, 20.50 ().
融点. 146.0-146.5°C . 146.0-146.5 ° C.
実施例 4-370 Example 4-370
Ή一 NMR (200MHz, CDCL) : <5 0.89 (6H, d, J= 6.6 Hz), 1.08 (3H, t, J= 7.3 Hz), 1.28 (2H, brq, J= 7.5 Hz), 1.57 (1H, quint, J= 6.6 Hz), 1.68 (2H, quint, J= 7.8 Hz), 2.13 (2H, q, J= 7.5 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.92 (2H, t, J= 7.4 Hz), 7.28 (2H, d, J= 8.2 Hz), 7.85 (2H, d, J= 8.2 Hz), 9.63 (1H, brs). 元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05 (%) 実測値: C, 54.83; H, 6.00; N, 8.71; S, 19.89 (%) . Ή-1 NMR (200MHz, CDCL): <5 0.89 (6H, d, J = 6.6 Hz), 1.08 (3H, t, J = 7.3 Hz), 1.28 (2H, brq, J = 7.5 Hz), 1.57 (1H, quint, J = 6.6 Hz), 1.68 (2H, quint, J = 7.8 Hz), 2.13 (2H, q, J = 7.5 Hz), 2.26 ( 3H, s), 2.41 (3H, s), 2.92 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.2 Hz), 7.85 (2H, d, J = 8.2 Hz), 9.63 ( . 1H, brs) elemental analysis: C 22 H 29 N 3 0 3 S 3 calculated: C, 55.09; H, 6.09 ; N, 8.76; S, 20.05 (%) Found: C, 54.83; H, 6.00; N, 8.71; S, 19.89 (%).
融点. 143.5- 144. St 143.5- 144. St
実施例 4-371 Example 4-371
融点 155-156°C Melting point 155-156 ° C
赤外吸収スペクトル(IR) (KBr中) リ: 3214, 2963, 2932, 2872, 1732, 1659, 1615, 1557, 1495 cm-Infrared absorption spectrum (IR) (in KBr): 3214, 2963, 2932, 2872, 1732, 1659, 1615, 1557, 1495 cm-
¾-NMR (CDC13) : δ 0.941 (3Η, t, J=7.4Hz), 1.09 (3H, t, J=7.2 Hz), 1.37-1.54 (2H, m), 1.54-1.76 (2H, m), 2.14 (2H, q, J=7.4 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J=7.6 Hz), 7.30 (2H, d, J=8.4 Hz), 7.85 (2H, d, J=8.6 Hz), 9.63 (1H, bs). ¾-NMR (CDC1 3): δ 0.941 (3Η, t, J = 7.4Hz), 1.09 (3H, t, J = 7.2 Hz), 1.37-1.54 (2H, m), 1.54-1.76 (2H, m) , 2.14 (2H, q, J = 7.4 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.6 Hz), 7.30 (2H, d, J = 8.4 Hz) , 7.85 (2H, d, J = 8.6 Hz), 9.63 (1H, bs).
元素分析値: C2。H25N303S3として計算値: C, 53.19; H, 5.58; N, 9.30; S, 21.30 (%), 実測値:, (;, 53.45; H, 5.56; N, 9.52; S, 21.48 (%) · Elemental analysis: C 2. H 25 N 3 0 3 S 3 Calculated: C, 53.19; H, 5.58; N, 9.30; S, 21.30 (%), Found:, (;, 53.45; H, 5.56; N, 9.52; S, 21.48 (%)
実施例 4-372 Example 4-372
融点 157-159°C Melting point 157-159 ° C
赤外吸収スぺクトル(IR) (KBr中) V: 3158, 2953, 2928, 2865, 1725, 1655, 1599, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3158, 2953, 2928, 2865, 1725, 1655, 1599, 1557 cm " 1 .
Ή-NMR (CDCI3) : δ 0.912 (3Η, t, J=6.6 Hz), 1.09 (3H, t, J=7. Hz), 1.31-1.45 (4H, m), 1.60-1.76 (2H, m), 2.13 (2H, q, J=7.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J=7.4 Hz), 7.28 (2H, d, J=8.4 Hz) , ' 7.86 (2H, d, 1=8: 4 Hz), 9.62 (1H, bs). Ή-NMR (CDCI3): δ 0.912 (3Η, t, J = 6.6 Hz), 1.09 (3H, t, J = 7. Hz), 1.31-1.45 (4H, m), 1.60-1.76 (2H, m) , 2.13 (2H, q, J = 7.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J = 7.4 Hz), 7.28 (2H, d, J = 8.4 Hz) , '7.86 (2H, d, 1 = 8: 4 Hz), 9.62 (1H, bs).
元素分析値: C21H27N303S3として計算値: C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), 実測値: C, 54.04; H, 6.10; N, 8.99; S, 20.60 ( ) . Elemental analysis: Calculated for C 21 H 27 N 3 0 3 S 3 : C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), found: C, 54.04; H, 6.10; N, 8.99; S, 20.60 ().
実施例 4-373 Example 4-373
融点 160- 162°C 160-162 ° C
赤外吸収スペクトル(IR) (KBr中) V: 3156, 2953, 2932, 1726, 1657, 1603, 1559 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3156, 2953, 2932, 1726, 1657, 1603, 1559 cm " 1 .
Ή-NMR (CDC13) : 6 0.901 (3H, t, J=6.6Hz), 1.09 (3H, t, J=7.4 Hz), 1.25-1.50 (6H, 111), 1.58-1.75 (2H, m), 2.14 (2H, q, J=7.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, ]=7.6 Hz), 7.28 (2H, d, J=8.4 Hz), 7.86 (2H, d, J=8.4Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3): 6 0.901 (3H, t, J = 6.6Hz), 1.09 (3H, t, J = 7.4 Hz), 1.25-1.50 (6H, 111), 1.58-1.75 (2H, m) , 2.14 (2H, q, J = 7.8 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t,] = 7.6 Hz), 7.28 (2H, d, J = 8.4 Hz) , 7.86 (2H, d, J = 8.4Hz), 9.62 (1H, bs).
元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05Elemental analysis: C 22 H 29 N 3 0 3 S 3 Calculated: C, 55.09; H, 6.09 ; N, 8.76; S, 20.05
(%)、 実測値: C, 54.95; H, 6.00; , '8.61; S, 19.98 (%) . (%), Found: C, 54.95; H, 6.00;, '8.61; S, 19.98 (%).
実施例 4-374 Example 4-374
Ή-NMR (200MHz, CDC13) : δ 1.06 (3H, t, J= 7.5 Hz), 2.01 (2H, quint, J= 7.5 Hz), 2.10 (2H, q, J= 7· 5 Hz), 2.26 (3H, s), 2.39 (3H, s), 2.72 (2H, t, J= 7.3 Hz), 2.93 (2H, t, J= 7.5 Hz), 7.16 (2H, brd, J= 7.2 Hz), 7.24-7.30 (5H, m), 7.86 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.06 (3H, t, J = 7.5 Hz), 2.01 (2H, quint, J = 7.5 Hz), 2.10 (2H, q, J = 7 · 5 Hz), 2.26 (3H, s), 2.39 (3H, s), 2.72 (2H, t, J = 7.3 Hz), 2.93 (2H, t, J = 7.5 Hz), 7.16 (2H, brd, J = 7.2 Hz), 7.24 -7.30 (5H, m), 7.86 (2H, d, J = 8.0 Hz), 9.64 (1H, brs).
元素分析値: C25H„N303S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73Elemental analysis: Calculated as C 25 H „N 3 0 3 S 3 : C, 58.45; H, 5.30; N, 8.18; S, 18.73
(%) 、 実測値: C, 58.55; H, 5.28; N, 7.95; S, 18.73 (%) · (%), Found: C, 58.55; H, 5.28; N, 7.95; S, 18.73 (%)
融点. 168.5-169.5°C Melting point.168.5-169.5 ° C
実施例 4-375 Example 4-375
NMR (200MHz, CDC13) : δ 1.08 (3H, t, J= 7.5 Hz), 1.39 (3H, d, J= 6.2 Hz), 2.13 (2H, q, J= 7.5 Hz), 2.25 (3H, s), 2.42 (3H, s), 3.04 (1H, m), 3.10 (1H, m), 3.23 (1H, quint, J= 5.6 Hz), 7.23 (3H, n , 7.28 (2H, d, J= 8.4 Hz), 7.29 (2H, m), 7.79 (2H, d, J= 8.4 Hz), 9.61 (1H, brs). NMR (200MHz, CDC1 3): δ 1.08 (3H, t, J = 7.5 Hz), 1.39 (3H, d, J = 6.2 Hz), 2.13 (2H, q, J = 7.5 Hz), 2.25 (3H, s ), 2.42 (3H, s), 3.04 (1H, m), 3.10 (1H, m), 3.23 (1H, quint, J = 5.6 Hz), 7.23 (3H, n, 7.28 (2H, d, J = 8.4) Hz), 7.29 (2H, m), 7.79 (2H, d, J = 8.4 Hz), 9.61 (1H, brs).
元素分析値: C25H27N303S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73Elemental analysis: Calculated as C 25 H 27 N 3 0 3 S 3: C, 58.45; H, 5.30; N, 8.18; S, 18.73
(%) 、 実測値: C, 58.29; H, 5.25; N, 8.16; S, 18.66 (%) . (%), Found: C, 58.29; H, 5.25; N, 8.16; S, 18.66 (%).
融点. 171.5- 172. O Melting point. 171.5- 172. O
実施例 4-376 Example 4-376
'H-NMR (CDClg, 200 MHz): δ 1.01 (3H, t, J= 7.5 Hz), 2.07 (2H, q, J= 7.3 Hz), 2.24 (3H, s), 2.41 (3H, s), 4.47 (2H, s), 7.27 (2H, d, J= 8.2 Hz), 7.51 (2H, brt, J= 7.3 Hz), 7.65 (1H, brt, J= 7.2 Hz) , 7.85 (2H, d, J= 8.4 Hz), 7.97 (2H, d, J= 8 Hz). 'H-NMR (CDClg, 200 MHz): δ 1.01 (3H, t, J = 7.5 Hz), 2.07 (2H, q, J = 7.3 Hz), 2.24 (3H, s), 2.41 (3H, s), 4.47 (2H, s), 7.27 (2H, d, J = 8.2 Hz), 7.51 (2H, brt, J = 7.3 Hz), 7.65 (1H, brt, J = 7.2 Hz), 7.85 (2H, d, J = 8.4 Hz), 7.97 (2H, d, J = 8 Hz).
元素分析値: C24H23N304S3として計算値: C, 56.12; H, 4.51; , 8.18; S, 18.73 (%), 実測値: C, 56.07; H, 4.61; N, 8.12; S, 18.64 (%) . Elemental analysis: Calculated as C 24 H 23 N 3 0 4 S 3: C, 56.12; H, 4.51;, 8.18; S, 18.73 (%), Found: C, 56.07; H, 4.61; N, 8.12; S, 18.64 (%).
実施例 4-377 Example 4-377
N- [8- (シク口へプチルスルファニル) -6-ェチル -5-メチル -3-ォキソ- 2, 3-ジヒド ロ- 1H-ィミダゾ [5, 1-c] [1 , 4]チアジン-卜ィリデン] -4-メチルベンゼンスルホン アミド N- [8- (cyclohexylsulfanyl) -6-ethyl-5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- Ylidene] -4-methylbenzenesulfonamide
実施例 3-5で得た化合物(499 mg, 1.26 mmol)および炭酸力リゥム (261 mg, 1.89 mmpl)の DMF (8.0 ml)の懸濁液に 35 で攪拌しながらヨウ化シクロへプチル (純 度 70%, 484 mg, 1.51 匪 ol)を加え、 反応液を 16時間攪拌した。 反応液に 1N塩 酸 (2.52 ml)を加え、 メタノール一水(1:1, 6 ml)で希釈し、 室温で 3時間攪拌し た。 析出物を濾取し、 メタノール一水 (1:1 および 2:1)で洗浄、 乾燥して標題化 合物 (484 mg, 0.984腿 ol)を赤紫色粉末として得た。 To a suspension of the compound obtained in Example 3-5 (499 mg, 1.26 mmol) and carbon dioxide (261 mg, 1.89 mmpl) in DMF (8.0 ml) was added cycloheptyl iodide (pure) while stirring at 35. 70%, 484 mg, 1.51 ol) and the reaction mixture was stirred for 16 hours. 1N Hydrochloric acid (2.52 ml) was added to the reaction solution, diluted with methanol / water (1: 1, 6 ml), and stirred at room temperature for 3 hours. The precipitate was collected by filtration, washed with methanol / water (1: 1 and 2: 1), and dried to give the title compound (484 mg, 0.984 tmol) as a red-purple powder.
lH— NMR (200MHz, CDC13) : δ 1.09 (3H, t, J= 7.5 Hz), 1.5-1.8 (10H, m), 2.06 (2H, m), 2.14 (2H, q, J= 7.5 Hz) , 2.26 (3H, s), 2.42 (3H, s), 3.54 (1H, septet, J= 4.4 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.66 (1H, brs). 元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56 (%) 、 実測値: C, 56.48; H, 5.92; N, 8.36; S, 19.29 ( ) . lH- NMR (200MHz, CDC1 3) : δ 1.09 (3H, t, J = 7.5 Hz), 1.5-1.8 (10H, m), 2.06 (2H, m), 2.14 (2H, q, J = 7.5 Hz) , 2.26 (3H, s), 2.42 (3H, s), 3.54 (1H, septet, J = 4.4 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz) , 9.66 (1H, brs) elemental analysis: calculated C 23 H 29 N 3 0 3 S 3:. C, 56.18; H, 5.94; N, 8.55; S, 19.56 (%), Found: C, 56.48; H, 5.92; N, 8.36; S, 19.29 ().
mp. 144.5-146.0°C mp.144.5-146.0 ° C
実施例 4-378 Example 4-378
N- {8- [ (2 -シク口へキシルェチル)スルファニル] -6-ェチル -5-メチル -3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン } -4-メチルベンゼ ンスルホンアミド ' N- {8- [(2-cyclohexylethyl) sulfanyl] -6-ethyl-5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-triylidene} -4-methylbenzenesulfonamide ''
実施例 3-5で得た化合物(292 mg, 0.738丽 ol)および炭酸カリウム(153 mg, 1.11 讓 ol)の DMF (5 ml)の懸濁液に室温で攪拌しながら臭化 2-シクロへキシルェチル To a suspension of the compound obtained in Example 3-5 (292 mg, 0.738 mol) and potassium carbonate (153 mg, 1.11 alcohol) in DMF (5 ml) was stirred at room temperature while stirring at room temperature to give 2-cyclohexane bromide. Kicilletil
(159 mg, 0.812扇 ol)を加え、 反応液を 80 で 1.5時間攪拌した。 反応液に 1N塩 酸 (1.5 ml)およびメタノール〜水(1:1, 5 ml)を加え、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール一水(1:1) およびメタノールで洗浄後、 乾燥して標 題化合物(366 mg, 0.724 mmol)を赤色粉末として得た。' (159 mg, 0.812 finol) was added, and the reaction solution was stirred at 80 for 1.5 hours. 1N Hydrochloric acid (1.5 ml) and methanol-water (1: 1, 5 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (366 mg, 0.724 mmol) as a red powder. '
Ή-NMR (200MHz, CDC13) : δ 0.94 (2Η, brquint, J= 11.0 Hz), 1.09 (3H, t, J= 7.4 Hz), 1.19 (2H, brd, J= 9.2 Hz), 1.21 (1H, m), 1.31 (2H, m), 1.57 (2H, q, J= 7.5 Hz), 1.71 (4H, brd, J= 10.6 Hz), 2.13 (2H, q, J= 7.4 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.96 (2H, i, J= 7.7 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz) 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.94 (2Η, brquint, J = 11.0 Hz), 1.09 (3H, t, J = 7.4 Hz), 1.19 (2H, brd, J = 9.2 Hz), 1.21 (1H , m), 1.31 (2H, m), 1.57 (2H, q, J = 7.5 Hz), 1.71 (4H, brd, J = 10.6 Hz), 2.13 (2H, q, J = 7.4 Hz), 2.26 (3H, s), 2.41 (3H, s), 2.96 (2H, i, J = 7.7 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz) 9.64 (1H, brs).
元素分析値: C24H3lN303S3として計算値: C, 57.00; H, 6.18; N, 8.31; S, 19.02 (%), 実測値: C, 56.86; H, 6.11; N, 8.32; S, 18.71 (%) . Elemental analysis: C 24 H 3l N 3 0 3 S 3 Calculated: C, 57.00; H, 6.18 ; N, 8.31; S, 19.02 (%), Found: C, 56.86; H, 6.11 ; N, 8.32; S, 18.71 (%).
mp. 185.5-186.5°C mp. 185.5-186.5 ° C
実施例 4-379 Example 4-379
Ή-NMR (CDC13, 200 MHz): δ 1.03 (6Η, d, J= 7.0 Hz), 1.32 (3H, t, J= 7.2 Hz), 1.93 (1H, sept, J= 6.6 Hz), 2.76 (3H, s), 2.88 (2H, d, J= 7.0 Hz), 4.26 (2H, q, J= 7.2 Hz), 7.52 (3H, m), 7.98 (2H, dm, J= 7.6 Hz), 9.80 (1H, brs). 元素分析値: C2。H23N305S3として計算値: C, 49.88; H, 4.81; N, 8.72; S, 19.97 Ή-NMR (CDC1 3, 200 MHz): δ 1.03 (6Η, d, J = 7.0 Hz), 1.32 (3H, t, J = 7.2 Hz), 1.93 (1H, sept, J = 6.6 Hz), 2.76 ( 3H, s), 2.88 (2H, d, J = 7.0 Hz), 4.26 (2H, q, J = 7.2 Hz), 7.52 (3H, m), 7.98 (2H, dm, J = 7.6 Hz), 9.80 ( IH, brs) elemental analysis:. C 2. H 23 N 3 0 5 S 3 Calculated: C, 49.88; H, 4.81 ; N, 8.72; S, 19.97
(%)、 実測値: C, 49.77; H, 4.75; N, 8.76; S, 19.96 (%) . (%), Found: C, 49.77; H, 4.75; N, 8.76; S, 19.96 (%).
実施例 4-380 Example 4-380
'H-NMR (CDC13, 200 MHz): δ 1.34 (3Η, t, J= 7.4 Hz), 2.76 (3H, s), 2.99 (2H, dd, J= 8.6, 6.2 Hz), 3.23 (2H, dd, J= 8.6, 6.2 Hz), 4.27 (2H, q, J= 7.4 Hz), 7.18 - 33 (5H, m), 7.52 (2H, m), 7.94 (2H, dm, J= 7.6 Hz), 9.79 (1H, brs). 元素分析値: C24H23N305S3として計算値: C, 54.42; H, 4.38; N, 7.93; S, 18.16 'H-NMR (CDC1 3, 200 MHz): δ 1.34 (3Η, t, J = 7.4 Hz), 2.76 (3H, s), 2.99 (2H, dd, J = 8.6, 6.2 Hz), 3.23 (2H, dd, J = 8.6, 6.2 Hz), 4.27 (2H, q, J = 7.4 Hz), 7.18-33 (5H, m), 7.52 (2H, m), 7.94 (2H, dm, J = 7.6 Hz), . 9.79 (1H, brs) elemental analysis: C 24 H 23 N 3 0 5 S 3 calculated: C, 54.42; H, 4.38 ; N, 7.93; S, 18.16
(%), 実測値: C, 54.19; H, 4.33; N, 8.10; S, 18.05 (%) . (%), Found: C, 54.19; H, 4.33; N, 8.10; S, 18.05 (%).
実施例 4 - 381 Example 4-381
'H-NMR (CDCl3-DMS0-d6= 19:1, 300 MHz): 6 2.43 (3H, s), 2.77 (3H, s), 2.97 (2H, brdd, J= 8.3, 7.2 Hz), 3.25 (2H, brdd, J= 8.3, 7.2 Hz), 7.20 (2H, brd, J= 8 Hz), 7.25-31 (5H, i), 7.83 (2H, d, J= 8.3 Hz), 10.43 (1H, brs). 'H-NMR (CDCl 3 -DMS0-d 6 = 19: 1, 300 MHz): 6 2.43 (3H, s), 2.77 (3H, s), 2.97 (2H, brdd, J = 8.3, 7.2 Hz), 3.25 (2H, brdd, J = 8.3, 7.2 Hz), 7.20 (2H, brd, J = 8 Hz), 7.25-31 (5H, i), 7.83 (2H, d, J = 8.3 Hz), 10.43 (1H , Brs).
元素分析値: C21 N305S3として計算値: C, 53.57; H, 4.11; , 8.15; S, 18.66 (%)、 実測値: C, 53.50; H, 4.03; N, 8.25; S, 18.67 (%) . Elemental analysis: Calculated for C 21 N 3 0 5 S 3 : C, 53.57; H, 4.11;, 8.15; S, 18.66 (%), found: C, 53.50; H, 4.03; N, 8.25; S , 18.67 (%).
実施例 4-382 Example 4-382
lH-NMR(CDCl3, 300 MHz): δ 2.41 (3Η, s), 2.75 (3H, s), 3.80 (3H, s), 4.25 (2H, s), 7.25 (2H, d, J= 8.3 Hz), 7.33 (5H, m), 7.79 (2H, d, J= 8.3 Hz), 9.81 (1H, brs). lH-NMR (CDCl 3 , 300 MHz): δ 2.41 (3Η, s), 2.75 (3H, s), 3.80 (3H, s), 4.25 (2H, s), 7.25 (2H, d, J = 8.3 Hz ), 7.33 (5H, m), 7.79 (2H, d, J = 8.3 Hz), 9.81 (1H, brs).
実施例 4-383 Ή-NMR (300MHz, CDC13) : δ 1.32 (3H, t, J= 7.1 Hz), 2.41 (3H, s), 2.74 (3H, s), 4.25 (2H, s), 4.26 (2H, q, J= 7.1 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.33 (5H, s), 7.80 (2H, d, J= 8.3 Hz), 9.77 (1H, br). Example 4-383 Ή-NMR (300MHz, CDC1 3 ): δ 1.32 (3H, t, J = 7.1 Hz), 2.41 (3H, s), 2.74 (3H, s), 4.25 (2H, s), 4.26 (2H, q, J = 7.1 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.33 (5H, s), 7.80 (2H, d, J = 8.3 Hz), 9.77 (1H, br).
実施例 4-384 Example 4-384
Ή-NMR (CDC13, 200 MHz): δ 1.32 (3H, t, J= 7.1 Hz), 1.43 (4H, ), 1.54-1.75 (4H, m), 2.42 (3H, s), 2.75 (3H, s) , 3.00 (2H, brt, J= 7.5 Hz), 3.65 (2H, t, J= 6.2 Hz), 4.26 (2H, q, J= 7.0 Hz), 7.29 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.83 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.32 (3H, t, J = 7.1 Hz), 1.43 (4H,), 1.54-1.75 (4H, m), 2.42 (3H, s), 2.75 (3H, s), 3.00 (2H, brt, J = 7.5 Hz), 3.65 (2H, t, J = 6.2 Hz), 4.26 (2H, q, J = 7.0 Hz), 7.29 (2H, d, J = 8.0 Hz) , 7.85 (2H, d, J = 8.4 Hz), 9.83 (1H, brs).
元素分析値: C23H29N306S3 · 0.2H20として計算値: C, 50.85; H, 5.45; N, 7.73; S, 17.71 (%)、 実測値: C, 50.68; H, 5.33; N, 7.96; S, 17.82 (%) . Elemental analysis: C 23 H 29 N 3 0 6 S 3 · 0.2H 2 0 Calculated: C, 50.85; H, 5.45; N, 7.73; S, 17.71 (%), Observed: C, 50.68; H , 5.33; N, 7.96; S, 17.82 (%).
実施例 4-385 Example 4-385
Ή-NMR (CDC13, 300 MHz): δ 1.03 (6Η, d, J= 6.7 Hz), 1.33 (3H, t, J= 7.1 Hz), 1.93 (1H, tq, J= 6.8, 6.7 Hz), 2.42 (3H, s), 2.75 (3H, s), 2.87 (2H, d, J= 6.8 Hz), 4.26 (2H, q, J= 7.1 Hz), 7.29 (2H, d, J= 8.1 Hz), 7.83 (2H, brd, J= 8.3 Hz), 9.79 (1H, brs). Ή-NMR (CDC1 3, 300 MHz): δ 1.03 (6Η, d, J = 6.7 Hz), 1.33 (3H, t, J = 7.1 Hz), 1.93 (1H, tq, J = 6.8, 6.7 Hz), 2.42 (3H, s), 2.75 (3H, s), 2.87 (2H, d, J = 6.8 Hz), 4.26 (2H, q, J = 7.1 Hz), 7.29 (2H, d, J = 8.1 Hz), 7.83 (2H, brd, J = 8.3 Hz), 9.79 (1H, brs).
元素分析値: C21H25N305S3として計算値: C, 50.89; H, 5.08; N, 8.48; S, 19.41Elemental analysis: Calculated for C 21 H 25 N 3 0 5 S 3 : C, 50.89; H, 5.08; N, 8.48; S, 19.41
(%), 実測値: C, 50.80; H, 5.20; N, 8.54; S, 19.36 ( ) . (%), Found: C, 50.80; H, 5.20; N, 8.54; S, 19.36 ().
実施例 4-386 Example 4-386
酢酸ェチル-メ夕ノ一ル -テ卜ラヒドロフランから再結晶し、 濃赤色結晶を得た。 融点: 142- 144°C. Recrystallization from ethyl acetate-methyl-tetrahydrofuran yielded dark red crystals. Melting point: 142-144 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3219, 2957, 2928, 2855, 1761, 1717, 1674, 1638, 1563, 1497 cnf1. -Infrared absorption spectrum (IR) (in KBr): 3219, 2957, 2928, 2855, 1761, 1717, 1674, 1638, 1563, 1497 cnf 1 .
Ή-NMR (CDC13) : 6 0.910 (3H, t, J=7. Hz), 1.22-1.44 (4H, m), 1.32 (3H, t, /=6.6 Hz), 1.63-1.76 (2H, m), 2.42 (3H, s), 2.75 (3H, s), 2.99 (2H, t, J=7.4 Hz), 4.26 (2H, q, J=7.2 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.77 (m, bs). Ή-NMR (CDC1 3): 6 0.910 (. 3H, t, J = 7 Hz), 1.22-1.44 (4H, m), 1.32 (3H, t, /=6.6 Hz), 1.63-1.76 (2H, m ), 2.42 (3H, s), 2.75 (3H, s), 2.99 (2H, t, J = 7.4 Hz), 4.26 (2H, q, J = 7.2 Hz), 7.29 (2H, d, J = 8.0 Hz) ), 7.85 (2H, d, J = 8.0 Hz), 9.77 (m, bs).
元素分析値: C22H27N305S3として計算値: C, 51.84; H, 5.34; N, 8.24; S, 18.87Elemental analysis: C 22 H 27 N 3 0 5 S 3 Calculated: C, 51.84; H, 5.34 ; N, 8.24; S, 18.87
(%), 実測値: C, 51.69; H, 5.41;凡 8.17; S, 18.85 (%) . (%), Found: C, 51.69; H, 5.41; about 8.17; S, 18.85 (%).
実施例 4-387 ¾-NMR (CDClg, 300 MHz): δ 1.32 (3H, t, J= 7.1 Hz), 1.70-1.95 (4H, m), 1.94 (1H, br), 2.40 (3H, s), 2.75 (3H, s), 3.03 (2H, brt, J= 6.3 Hz), 4.26 (2H, q, J= 7.1 Hz), 4.71 (1H, br), 7.25 (2H, d, J= 8.3 Hz), 7.35 (5H, m), 7.83 (2H, d, J= 8.3 Hz), 9.73 (1H, s). Example 4-387 ¾-NMR (CDClg, 300 MHz): δ 1.32 (3H, t, J = 7.1 Hz), 1.70-1.95 (4H, m), 1.94 (1H, br), 2.40 (3H, s), 2.75 (3H, s), 3.03 (2H, brt, J = 6.3 Hz), 4.26 (2H, q, J = 7.1 Hz), 4.71 (1H, br), 7.25 (2H, d, J = 8.3 Hz), 7.35 (5H, m), 7.83 (2H, d, J = 8.3 Hz), 9.73 (1H, s).
元素分析値: C27H29N306S3として計算値: C, 55.18; H, 4.97; N, 7.15; S, 16.37 (%)、 実測値: C, 55.12; H, 4.81; , 7.05; S, 16.40 ( ) . Elemental analysis: Calculated as C 27 H 29 N 3 0 6 S 3: C, 55.18; H, 4.97; N, 7.15; S, 16.37 (%), Found: C, 55.12; H, 4.81 ;, 7.05 ; S, 16.40 ().
実施例 4-388 Example 4-388
'H-NMRCCDClg, 300 MHz): δ 1.32 (3H, t, J= 7.1 Hz), 2.16 (2H, quint, J= 7.0 Hz), 2.38 (3H, s), 2.75 (3H, s), 3.14 (4H, t, J= 7.0 Hz), 4.25 (2H, q, J= 7.1 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.49 (2H, t, J= 7.7 Hz), 7.59 (1H, tt, J= 7.3, 1.2 Hz), 7.83 (2H, d, J= 8.3 Hz), 7.96 (2H, d, J= 7.2. Hz), 9.84 (1H, brs) . 'H-NMRCCDClg, 300 MHz): δ 1.32 (3H, t, J = 7.1 Hz), 2.16 (2H, quint, J = 7.0 Hz), 2.38 (3H, s), 2.75 (3H, s), 3.14 ( 4H, t, J = 7.0 Hz), 4.25 (2H, q, J = 7.1 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.49 (2H, t, J = 7.7 Hz), 7.59 (1H, tt, J = 7.3, 1.2 Hz), 7.83 (2H, d, J = 8.3 Hz), 7.96 (2H, d, J = 7.2. Hz), 9.84 (1H, brs).
元素分析値: C27H27N306S3として計算値: C, 54.53; H, 4.74; N, 7.07; S, 16.18 (%), 実測値: C, 54.46; H, 4.58; N, 7.18; S, 16.27 (%) . Elemental analysis: Calculated as C 27 H 27 N 3 0 6 S 3: C, 54.53; H, 4.74; N, 7.07; S, 16.18 (%), Found: C, 54.46; H, 4.58 ; N, 7.18; S, 16.27 (%).
実施例 4-389 Example 4-389
^-NMR (CDC13, 200 MHz): 6 1.03 (6H, d, J= 6.6 Hz), 1.33 (3H, t, J= 7.2 Hz), 1.93 (1H, quint, J= 6.7 Hz), 2.42 (3H, s), 2.76 (3H, s), 2.88 (2H, d, J= 6.6 Hz), 4.26 (2H, q, J= 7.0 Hz), 7.38 (2H, brd, J= 5.2 Hz), 7.76 (2H, brs), 9.79 (1H, brs). ^ -NMR (CDC1 3, 200 MHz ): 6 1.03 (6H, d, J = 6.6 Hz), 1.33 (3H, t, J = 7.2 Hz), 1.93 (1H, quint, J = 6.7 Hz), 2.42 ( 3H, s), 2.76 (3H, s), 2.88 (2H, d, J = 6.6 Hz), 4.26 (2H, q, J = 7.0 Hz), 7.38 (2H, brd, J = 5.2 Hz), 7.76 ( 2H, brs), 9.79 (1H, brs).
元素分析値: C21H25N305S3として計算値: C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%)、 実測値: C, 50.78; H, 5.34; N, 8.50; S, 19.46 (%) . Elemental analysis: C 21 H 25 N 3 0 5 S 3 Calculated: C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%), found: C, 50.78; H, 5.34; N, 8.50; S, 19.46 (%).
実施例 4-390 Example 4-390
Ή-NMR (CDC13, 200 MHz): δ 1.34 (3Η, t, J- 7.0 Hz), 2.43 (3H, s), 2.76 (3H, s), 2.99 (2H, dd, ]= 9.2, 6.6 Hz), 3.23 (2H, dd, J= 8.4, 5.8 Hz), 4.27 (2H, q, J= 6.6 Hz), 7.28 (5H, ), 7.38 (2H, brd, J= 5.2 Hz), 7.76 (2H, brs), 9.79 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.34 (3Η, t, J- 7.0 Hz), 2.43 (3H, s), 2.76 (3H, s), 2.99 (2H, dd,] = 9.2, 6.6 Hz ), 3.23 (2H, dd, J = 8.4, 5.8 Hz), 4.27 (2H, q, J = 6.6 Hz), 7.28 (5H,), 7.38 (2H, brd, J = 5.2 Hz), 7.76 (2H, brs), 9.79 (1H, brs).
元素分析値: C25H25N305S3として計算値: C, 55.23; H, 4.63; N, 7.73; S, 17.69Elemental analysis: C 25 H 25 N 3 0 5 S 3 Calculated: C, 55.23; H, 4.63 ; N, 7.73; S, 17.69
(%)、 実測値: C, 54.92; H, 4.76; N, 7.74; S, 17.71 (%) . (%), Found: C, 54.92; H, 4.76; N, 7.74; S, 17.71 (%).
実施例 4-391 'H-NMR (CDCI3, 200 MHz): <5 1.03 (6H, d, J= 6.6 Hz), 1.32 (3H, t, J= 7.0 Hz), 1.93 (1H, sept, J= 6.8 Hz), 2.75 (3H, s), 2.80 (3H, s), 2.90 (2H, d, J= 6.6 Hz), 4.26 (2H, q, J= 7.2 Hz), 7.31 (2H, brd, J= 7 Hz), 7.44 (1H, tm, J= 7.4 Hz), 8.02 (1H, dm, J= 8.6 Hz), 9.84 (1H, brs). Example 4-391 'H-NMR (CDCI3, 200 MHz): <5 1.03 (6H, d, J = 6.6 Hz), 1.32 (3H, t, J = 7.0 Hz), 1.93 (1H, sept, J = 6.8 Hz), 2.75 (3H, s), 2.80 (3H, s), 2.90 (2H, d, J = 6.6 Hz), 4.26 (2H, q, J = 7.2 Hz), 7.31 (2H, brd, J = 7 Hz), 7.44 (1H, tm, J = 7.4 Hz), 8.02 (1H, dm, J = 8.6 Hz), 9.84 (1H, brs).
元素分析値: C21H25N305S3として計算値: C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%)、 実測値: C, 55.69; H, 5.00; N, 8.66; S, 19.36 {%) . Elemental analysis: Calculated for C 21 H 25 N 3 0 5 S 3 : C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%), found: C, 55.69; H, 5.00; N, 8.66; S, 19.36 {%).
実施例 4- 392 Example 4-392
¾ -腿 (CDC13, 200 MHz): δ 1.33 (3H, t, J= 7.4 Hz), 2.72 (3H, s), 2.75 (3H, s), 2.98 (2H, brdd, J= 8.0, 7.0 Hz), 3.26 (2H, dd, J= 8.0, 7.0 Hz), 4.27 (2H, q, J= 7.0 Hz), 7.20 (2H, m), 7.28 (5H, m), 7.46 (1H, brt, J= 7.4 Hz), 8.01 (1H, dm, J= 7 Hz), 9.82 (1H, brs). ¾ - thigh (CDC1 3, 200 MHz): δ 1.33 (3H, t, J = 7.4 Hz), 2.72 (3H, s), 2.75 (3H, s), 2.98 (2H, brdd, J = 8.0, 7.0 Hz ), 3.26 (2H, dd, J = 8.0, 7.0 Hz), 4.27 (2H, q, J = 7.0 Hz), 7.20 (2H, m), 7.28 (5H, m), 7.46 (1H, brt, J = 7.4 Hz), 8.01 (1H, dm, J = 7 Hz), 9.82 (1H, brs).
元素分析値: C25H25N305S3として計算値: C, 55.23; H, 4.63; N, 7.73; S, 17.69Elemental analysis: C 25 H 25 N 3 0 5 S 3 Calculated: C, 55.23; H, 4.63 ; N, 7.73; S, 17.69
(%), 実測値: C, 55.06; H, 4.51; , 7.90; S, 17.75 (%) . (%), Found: C, 55.06; H, 4.51;, 7.90; S, 17.75 (%).
実施例 4-393 Example 4-393
LH-NMR (CDCI3, 200 MHz): δ 1.04 (6Η, d, J= 7.0 Hz), 1.33 (3H, t, J= 7.0 Hz), 1.93 (1H, sept, J= 6.7 Hz), 2.76 (3H, s), 2.89 (2H, d, J= 6.6 Hz), 4.27 (2H, q, J= 7.2 Hz), 7.46 (2H, dt, J= 8.8, 2.0 Hz), 7.91 (2H, dt, J= 8.8, 2.0 Hz)., 9.77 (1H, brs). LH-NMR (CDCI3, 200 MHz): δ 1.04 (6Η, d, J = 7.0 Hz), 1.33 (3H, t, J = 7.0 Hz), 1.93 (1H, sept, J = 6.7 Hz), 2.76 (3H , s), 2.89 (2H, d, J = 6.6 Hz), 4.27 (2H, q, J = 7.2 Hz), 7.46 (2H, dt, J = 8.8, 2.0 Hz), 7.91 (2H, dt, J = 8.8, 2.0 Hz)., 9.77 (1H, brs).
元素分析値: C20H22N305S3C1として計算値: C, 46.55; H, 4.30; N, 8.14; S, 18.64; C1, 6.87 (%)、 実測値: C, 46.26; H, 4.07; N, 8.14; S, 18.69; C1, 6.74 (%) . 実施例 4-394 Elemental analysis: Calculated as C 20 H 22 N 3 0 5 S 3 C1: C, 46.55; H, 4.30; N, 8.14; S, 18.64; C1, 6.87 (%), Found: C, 46.26; H , 4.07; N, 8.14; S, 18.69; C1, 6.74 (%).
Ή-NMR (CDC13, 200 MHz): δ 1.34 (3Η, t, J= 7.0 Hz), 2.77 (3H, s), 2.99 (2H, dd, J= 8.0, 7.0 Hz), 3.26 (2H, dd, J= 8.4, 6.6 Hz), 4.27 (2H, q, J= 6.8 Hz), 7.17-33 (5H, m), 7.45 (2H, d, J= 8.8 Hz), 7.86 (2H, d, J- 8.4 Hz)., 9.78 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.34 (3Η, t, J = 7.0 Hz), 2.77 (3H, s), 2.99 (2H, dd, J = 8.0, 7.0 Hz), 3.26 (2H, dd , J = 8.4, 6.6 Hz), 4.27 (2H, q, J = 6.8 Hz), 7.17-33 (5H, m), 7.45 (2H, d, J = 8.8 Hz), 7.86 (2H, d, J- 8.4 Hz)., 9.78 (1H, brs).
元素分析値: C24H22N305S3C1として計算値: C, 51.10; H, 3.93; N, 7.45; S, 17.05; C1, 6.28 (%), 実測値: C, 50.96; H, 3.83; N, 7.47; S, 17.06; CI, 6.10 (%) . 実施例 4-395 Elemental analysis: C 24 H 22 N 3 0 5 S 3 C1 Calculated: C, 51.10; H, 3.93 ; N, 7.45; S, 17.05; C1, 6.28 (%), Found: C, 50.96; H , 3.83; N, 7.47; S, 17.06; CI, 6.10 (%).
Ή-NMR (CDCK, 200 MHz): δ 1.03 (6H, d, J= 6.6 Hz), 1.33 (3H, t, J= 7.2 Hz), 1.93 (1H, quint, J= 6.4 Hz), 2.76 (3H, s), 2.89 (2H, d, J= 7.0 Hz), 4.27 (2H, q, J= 7.2 Hz), 7.16 (2H, t, J= 7.0 Hz), 7.99 (2H, dd, J= 7.4, 5.2 Hz), 9.77 (1H, brs). Ή-NMR (CDCK, 200 MHz): δ 1.03 (6H, d, J = 6.6 Hz), 1.33 (3H, t, J = 7.2 Hz), 1.93 (1H, quint, J = 6.4 Hz), 2.76 (3H, s), 2.89 (2H, d, J = 7.0 Hz), 4.27 (2H, q, J = 7.2 Hz), 7.16 (2H, t, J = 7.0 Hz), 7.99 (2H, dd, J = 7.4, 5.2 Hz), 9.77 (1H, brs).
元素分析値: C20H22N3O5S3Fとして計算値: C, 48.08; H, 4.44; N, 8.41; S, 19.25; F, 3.80 (%), 実測値: C, 47.75; H, 4.45; N, 8.41; S, 19.26; F, 3.94 (%) . 実施例 4-396 Elemental analysis: C 20 H 22 N 3 O 5 S 3 F Calculated: C, 48.08; H, 4.44 ; N, 8.41; S, 19.25; F, 3.80 (%), Found: C, 47.75; H , 4.45; N, 8.41; S, 19.26; F, 3.94 (%).
¾-NMR (CDC13, 200 MHz): δ 1.34 (3Η, t, J= 7.0 Hz), 2.77 (3H, s), 2.99 (2H, dd, J= 8.6, 7.0 Hz), 3.25 (2H, dd, J= 8.0, 7.8 Hz), 4.27 (2H, q, J= 7.2 Hz), 7.15 (2H, t, J= 8.8 Hz), 7.26 (5H, m), 7.94 (2H, dd, J= 8.8, 5.0 Hz), 9.75 (1H, brs). ' 元素分析値: C24H22N305S3Fとして計算値: C, 52.64; H, 4.05; N, 7.67; S, 17.57; F, 3.47 (%), 実測値: C, 52.45; H, 4.08; N, 7.64; S, 17.60; F, 3.68 ( ) . 実施例 4-397 ¾-NMR (CDC1 3, 200 MHz): δ 1.34 (3Η, t, J = 7.0 Hz), 2.77 (3H, s), 2.99 (2H, dd, J = 8.6, 7.0 Hz), 3.25 (2H, dd , J = 8.0, 7.8 Hz), 4.27 (2H, q, J = 7.2 Hz), 7.15 (2H, t, J = 8.8 Hz), 7.26 (5H, m), 7.94 (2H, dd, J = 8.8, . 5.0 Hz), 9.75 (1H , brs) ' elemental analysis: C 24 H 22 N 3 0 5 S 3 F calculated: C, 52.64; H, 4.05 ; N, 7.67; S, 17.57; F, 3.47 (%), Found: C, 52.45; H, 4.08; N, 7.64; S, 17.60; F, 3.68 (). Examples 4-397
Ή-NMR (CDC13, 200 MHz): δ 1.03 (6Η, d, J= 6.6 Hz), 1.33 (3H, t, J= 7.4 Hz), 1.93 (IH, quint, J= 6.6 Hz), 2.76 (3H, s), 2.88 (2H, d, J= 6.6 Hz), 4.27 (2H, q, J= 7.4 Hz), 7.02 (2H, d, J= 8.4 Hz), 7.06 (2H, d, J= 7.4 Hz), 7.21 (IH, t, J= 7.0 Hz), 7.40 (2H, t, J= 8.0 Hz), 7.91 (2H, d, J= 8.8 Hz), 9.78 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.03 (6Η, d, J = 6.6 Hz), 1.33 (3H, t, J = 7.4 Hz), 1.93 (IH, quint, J = 6.6 Hz), 2.76 ( 3H, s), 2.88 (2H, d, J = 6.6 Hz), 4.27 (2H, q, J = 7.4 Hz), 7.02 (2H, d, J = 8.4 Hz), 7.06 (2H, d, J = 7.4 Hz) Hz), 7.21 (IH, t, J = 7.0 Hz), 7.40 (2H, t, J = 8.0 Hz), 7.91 (2H, d, J = 8.8 Hz), 9.78 (1H, brs).
元素分析値: C26H27N306S3として計算値: C, 54.43; H, 4.74; , 7.32; S, 16.77 (%), 実測値: C, 54.34; H, 4.75; N, 7.64; S, 16.77 (%) . Elemental analysis: Calculated as C 26 H 27 N 3 0 6 S 3: C, 54.43; H, 4.74;, 7.32; S, 16.77 (%), Found: C, 54.34; H, 4.75 ; N, 7.64 ; S, 16.77 (%).
実施例 4-398 Example 4-398
Ή-NMR (CDC13, 200 MHz): δ 1.34 (3H, t, J= 7.2 Hz), 2.76 (3H, s), 2.99 (2H, dd, J= 8.4, 6.6 Hz), 3.25 (2H, dd, J= 8.6, 6.6 Hz), 4.27 (2H, q, J= 7.2 Hz), 7.00 (2H, d, J= 9.2 Hz), 7.07 (2H, d, J= 7.8 Hz), 7.23 (6H, m), 7.41 (2H, bri, J= 8.0 Hz), 7.87 (2H, d, J= 8.8 Hz), 9.70 (IH, brs). Ή-NMR (CDC1 3, 200 MHz): δ 1.34 (3H, t, J = 7.2 Hz), 2.76 (3H, s), 2.99 (2H, dd, J = 8.4, 6.6 Hz), 3.25 (2H, dd , J = 8.6, 6.6 Hz), 4.27 (2H, q, J = 7.2 Hz), 7.00 (2H, d, J = 9.2 Hz), 7.07 (2H, d, J = 7.8 Hz), 7.23 (6H, m ), 7.41 (2H, bri, J = 8.0 Hz), 7.87 (2H, d, J = 8.8 Hz), 9.70 (IH, brs).
元素分析値: C3。H„N306S3 · 0.5 ¾0として計算値: C, 57.13; H, 4.47; N, 6.66; S, 15.25 (%), 実測値: C, 56.96; H, 4.47; N, 6.89; S, 15.29 (%) . Elemental analysis: C 3. H „N 3 0 6 S 3 · 0.5 ¾0, Calculated: C, 57.13; H, 4.47; N, 6.66; S, 15.25 (%), Found: C, 56.96; H, 4.47; N, 6.89; S , 15.29 (%).
実施例 4-399 Example 4-399
Ή-NMR (CDC13, 300 MHz): <5 1.07 (6H, d, J= 6.4 Hz), 1.34 (3H, t, J= 7.2 Hz), 1.96 (1H, sept, J= 6.8 Hz), 2.76 (3H, s), 2.91 (2H, d, J= 7.2 Hz), 3.11 (3H, s), 4.28 (2H, q, J= 7.2 Hz), 9.64 (1H, brs). Ή-NMR (CDC1 3, 300 MHz): <5 1.07 (6H, d, J = 6.4 Hz), 1.34 (3H, t, J = 7.2 Hz), 1.96 (1H, sept, J = 6.8 Hz), 2.76 (3H, s), 2.91 (2H, d, J = 7.2 Hz), 3.11 (3H, s), 4.28 (2H, q, J = 7.2 Hz), 9.64 (1H, brs).
元素分析値: C15H21N305S3として計算値: C, 42.94; H, 5.05; N, 10.02; S, 22.93Elemental analysis: C 15 H 21 N 3 0 5 S 3 Calculated: C, 42.94; H, 5.05 ; N, 10.02; S, 22.93
(%), 実測値: C, 42.89; H, 5.14; N, 10.06; S, 22.91 (%) . (%), Found: C, 42.89; H, 5.14; N, 10.06; S, 22.91 (%).
実施例 4-400 Example 4-400
lH-丽 (CDC13, 300 MHz): δ 1.35 (3H, t, J= 7.2 Hz), 2.77 (3H, s), 3.02 (2H, dd, J= 8.7, 7.2 Hz) , 3.09 (3H, s), 3.26 (2H, dd, J= 8.7, 7.2 Hz), 4.28 (2H, q, J= 7.2 Hz), 7.24 (2H, brd, J= 7.5 Hz), 7.27-7.36 (3H, m), 9.60 (1H, brs). 元素分析値: C19H21N305S3として計算値: C, 48.80; H, 4.53; N, 8.99; S, 20.57lH-丽(CDC1 3, 300 MHz): δ 1.35 (3H, t, J = 7.2 Hz), 2.77 (3H, s), 3.02 (2H, dd, J = 8.7, 7.2 Hz), 3.09 (3H, s ), 3.26 (2H, dd, J = 8.7, 7.2 Hz), 4.28 (2H, q, J = 7.2 Hz), 7.24 (2H, brd, J = 7.5 Hz), 7.27-7.36 (3H, m), 9.60 . (1H, brs) elemental analysis: C 19 H 21 N 3 0 5 S 3 calculated: C, 48.80; H, 4.53 ; N, 8.99; S, 20.57
(%), 実測値: C, 48.78; H, 4.65; N, 9.03; S, 20.59 (%) . (%), Found: C, 48.78; H, 4.65; N, 9.03; S, 20.59 (%).
実施例 4-401 Example 4-401
Ή-NMR (CDC13, 300 MHz): δ 1.33 (3Η, t, J= 7.2 Hz), 2.20 (2H, quint, J= 7.0 Hz), 2.76 (3H, s) , 3.09 (3H, s), 3.17 (3H, s), 3.14 (2H, brt, J= 6.6 Hz), 3.18 (2H, t, J= 7.7 Hz) , 4.27 (2H, q, J= 7.2 Hz) , 7.48 (2H, brt, J= 7.9 Hz) , 7.59 (1H, t, J= 7.5 Hz), 7.97 (2H, brd, J= 7.9 Hz), 9.63 (1H, brs). Ή-NMR (CDC1 3, 300 MHz): δ 1.33 (3Η, t, J = 7.2 Hz), 2.20 (2H, quint, J = 7.0 Hz), 2.76 (3H, s), 3.09 (3H, s), 3.17 (3H, s), 3.14 (2H, brt, J = 6.6 Hz), 3.18 (2H, t, J = 7.7 Hz), 4.27 (2H, q, J = 7.2 Hz), 7.48 (2H, brt, J = 7.9 Hz), 7.59 (1H, t, J = 7.5 Hz), 7.97 (2H, brd, J = 7.9 Hz), 9.63 (1H, brs).
元素分析値: C21H23N306S3として計算値: C, 49.49; H, 4.55; N, 8.25; S, 18.88Elemental analysis: C 21 H 23 N 3 0 6 S 3 Calculated: C, 49.49; H, 4.55 ; N, 8.25; S, 18.88
(%)、 実測値: C, 49.49; H, 4.67; N, 8.29; S, 18.92 (%) . (%), Found: C, 49.49; H, 4.67; N, 8.29; S, 18.92 (%).
実施例 -402 Example -402
Ή-NM (CDC13, 300 MHz): δ 1.04 (6Η, d, J= 6.4 Hz), 1.92 (1H, sept, J= 6.8 Hz), 2.41 (3H, s), 2.42 (3H, s), 2.63 (3H, s), 2.90 (2H, d, J= 7.2 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.81 (1H, brs). Ή-NM (CDC1 3, 300 MHz): δ 1.04 (6Η, d, J = 6.4 Hz), 1.92 (1H, sept, J = 6.8 Hz), 2.41 (3H, s), 2.42 (3H, s), 2.63 (3H, s), 2.90 (2H, d, J = 7.2 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.81 (1H, brs).
元素分析値: C2。H23N304S3として計算値: C, 51.59; H, 4.98; N, 9.02; S, 20.66Elemental analysis: C 2. H 23 N30 4 S 3 Calculated: C, 51.59; H, 4.98 ; N, 9.02; S, 20.66
(%)、 実測値: C, 51.69; H, 5.05; , 8.93; S, 20.64 ( ) . (%), Found: C, 51.69; H, 5.05;, 8.93; S, 20.64 ().
実施例 -403 Example -403
Ή-NMR (CDC13, 300 MHz): 6 2.41 (3H, s), 2.43 (3H, s), 2.63 (3H, s), 2.99 (2H, brdd, J= 8.3, 7.5 Hz), 3.27 (2H, brdd, J= 8.3, 7.2 Hz), 7.20 (2H, brd, J= 8 Hz), 7.26-7.32 (5H, m), 7.83 (2H, d, J= 8.3 Hz), 9.80 (1H, brs). 元素分析値: C24H23N304S3 · 0.2H20として計算値: C, 55.73; H, 4.56; N, 8.12; S, 18.60 (%), 実測値: C, 55.85; H, 4.52; N, 7.97; S, 18.26 ( ) · 実施例 4-404 Ή-NMR (CDC1 3, 300 MHz): 6 2.41 (3H, s), 2.43 (3H, s), 2.63 (3H, s), 2.99 (2H, brdd, J = 8.3, 7.5 Hz), 3.27 (2H , brdd, J = 8.3, 7.2 Hz), 7.20 (2H, brd, J = 8 Hz), 7.26-7.32 (5H, m), 7.83 (2H, d, J = 8.3 Hz), 9.80 (1H, brs) . elemental analysis: C 24 H 23 N 3 0 4 S 3 · 0.2H 2 0 calculated: C, 55.73; H, 4.56 ; N, 8.12; S, 18.60 (%), Found: C, 55.85; H, 4.52; N, 7.97; S, 18.26 () Example 4-404
¾-NMR (CDC13, 300 MHz): δ 2.17 (2H, quint, J= 7.2 Hz), 2.39 (3H, s), 2.40 (3H, s), 2.62 (3H, s), 3.14 (2H, t, J= 6.8 Hz), 3.15 (2H, t, J= 7.2 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.48 (2H, t, J= 7.5 Hz), 7.59 (1H, tt, J= 7.2, 1.8 Hz), 7.84 (2H, d, J= 8.3 Hz), 7.95 (2H, brd, J= 7.5 Hz), 9.81 (1H, brs). ¾-NMR (CDC1 3, 300 MHz): δ 2.17 (2H, quint, J = 7.2 Hz), 2.39 (3H, s), 2.40 (3H, s), 2.62 (3H, s), 3.14 (2H, t , J = 6.8 Hz), 3.15 (2H, t, J = 7.2 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.48 (2H, t, J = 7.5 Hz), 7.59 (1H, tt, J = 7.2, 1.8 Hz), 7.84 (2H, d, J = 8.3 Hz), 7.95 (2H, brd, J = 7.5 Hz), 9.81 (1H, brs).
元素分析値: C26H25N305S3 · 0.25H2Oとして計算値: C, 55.75; H, 4.59; N, 7.50; S, 17.17 (%) 実測値: C, 55.76; H, 4.52; N, 7.55; S, 17.11 (%) . Elemental analysis: C 26 H 25 N 3 0 5 S 3 · 0.25 H 2 O Calculated: C, 55.75; H, 4.59; N, 7.50; S, 17.17 (%) Found: C, 55.76; H, 4.52; N, 7.55; S, 17.11 (%).
実施例 4-405 Example 4-405
lH- NMR (CDC13, 300 MHz): δ 1.19 (3H, t, J= 7.2 Hz), 2.43 (3H, s), 2.48 (3H, s), 2.96 (2H, brdd, J= 8.3, 7.5 Hz), 3.22 (2H, brdd, J= 8.3, 7.4 Hz), 3.36 lH- NMR (CDC1 3, 300 MHz ): δ 1.19 (3H, t, J = 7.2 Hz), 2.43 (3H, s), 2.48 (3H, s), 2.96 (2H, brdd, J = 8.3, 7.5 Hz ), 3.22 (2H, brdd, J = 8.3, 7.4 Hz), 3.36
(2H, dq, J= 5.7, 7.2 Hz), 5.86 (1H, brt, J= 6 Hz), 7.19 (2H, brd, J- 8 Hz),(2H, dq, J = 5.7, 7.2 Hz), 5.86 (1H, brt, J = 6 Hz), 7.19 (2H, brd, J-8 Hz),
7.26-32 (5H, m), 7.83 (2H, d, J= 8.3 Hz), 9.71 (1H, brs). 7.26-32 (5H, m), 7.83 (2H, d, J = 8.3 Hz), 9.71 (1H, brs).
元素分析値: C25H26N404S3として計算値: C, 55.33; H, 4.83; N, 10.32; S, 17.73 (%) 実測値: C, 55.13; H, 4.84; N, 10.16; S, 17.74 (%) . Elemental analysis: Calculated as C 25 H 26 N 4 0 4 S 3: C, 55.33; H, 4.83; N, 10.32; S, 17.73 (%) Found: C, 55.13; H, 4.84 ; N, 10.16 ; S, 17.74 (%).
実施例 4-406 Example 4-406
酢酸ェチルから再結晶し、 橙色結晶として得た。 The crystals were recrystallized from ethyl acetate to obtain orange crystals.
融点 165- 167°C. Melting point 165-167 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3190, 2957, 2932, 2210, 1742, 1622, 1597, 1551 cm—1. Ή-NMR (CDC13) : δ 0.910 (3Η, t, J=7.0 Hz), 1.24-1.80 (6H, m), 2.43 (3H, s), 2.58 (3H, s), 2.94 (2H, t, 1=1.2 Hz), 7.30 (2H, d, J=8.0 Hz), 7.83 (2H, d, J=8.0 Hz), 9.69 (1H, bs). Infrared absorption spectrum (IR) (in KBr) V:. 3190, 2957 , 2932, 2210, 1742, 1622, 1597, 1551 cm- 1 Ή-NMR (CDC1 3): δ 0.910 (3Η, t, J = 7.0 Hz), 1.24-1.80 (6H, m), 2.43 (3H, s), 2.58 (3H, s), 2.94 (2H, t, 1 = 1.2 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.69 (1H, bs).
元素分析値: C2。H22N403S3 · 0.7H20として計算値: C, 50.55; H, 4.96; N, 11.79; S, 20.24 (%), 実測値: C, 50.51; H, 4.83; N, 11.63; S, 20.41 (%) . Elemental analysis: C 2. H 22 N 4 0 3 S 3 · 0.7H 2 0 Calculated: C, 50.55; H, 4.96; N, 11.79; S, 20.24 (%), Found: C, 50.51; H, 4.83; N, 11.63 ; S, 20.41 (%).
実施例 4-407 Example 4-407
Ή-NMR (300MHz, CDC13+TFA) : <5 2.42 (3H, s), 2.45 (2H, brt, J= 7.3 Hz), 2.61 (2H, brt, J= 7.3 Hz), 4.24 (2H, s), 6.64 (1H, s), 7.26 (2H, d, J= 8.2 Hz), 7.34 (5H, s), 7.82 (2H, d, J- 8.2 Hz), 10.22 (1H, brs). Ή-NMR (300MHz, CDC1 3 + TFA): <5 2.42 (3H, s), 2.45 (2H, brt, J = 7.3 Hz), 2.61 (2H, brt, J = 7.3 Hz), 4.24 (2H, s ), 6.64 (1H, s), 7.26 (2H, d, J = 8.2 Hz), 7.34 (5H, s), 7.82 (2H, d, J- 8.2 Hz), 10.22 (1H, brs).
元素分析値: C23H21N305S3として計算値: C, 53.58; H, 4.10; N, 8.15; S, 18.66 ( ) 、 実測値: C, 53.26; H, 4.14; N, 7.92; S, 18.58 (%) · 実施例 4-408 Elemental analysis: C 23 H 21 N 3 0 5 S 3 Calculated: C, 53.58; H, 4.10; N, 8.15; S, 18.66 (), found: C, 53.26; H, 4.14; N, 7.92 ; S, 18.58 (%) Example 4-408
■ ¾-NMR (300MHz, CDC13) : δ 2.39 (2H, m), 2.41 (3H, s), 2.48 (2H, brt, J= 6.5 Hz), 3.70 (3H, s), 4.21 (2H, s), 6.53 (1H, brs), 7.26 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J= 8.3 Hz), 9.74 (1H, brs). ■ ¾-NMR (300MHz, CDC1 3): δ 2.39 (2H, m), 2.41 (3H, s), 2.48 (2H, brt, J = 6.5 Hz), 3.70 (3H, s), 4.21 (2H, s ), 6.53 (1H, brs), 7.26 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J = 8.3 Hz), 9.74 (1H, brs).
元素分析値: C24H23N305S3として計算値: C, 54.23; H, 4.40; , 7.91; S, 18.10 (%) 、 実測値: C, 53.91; H, 4.18; N, 7.91; S, 18.43 (%) . Elemental analysis: C 24 H 23 N 3 0 5 S 3 Calculated: C, 54.23; H, 4.40;, 7.91; S, 18.10 (%), found: C, 53.91; H, 4.18; N, 7.91 ; S, 18.43 (%).
実施例 4-409 Example 4-409
¾ -墜 (300MHz, CDC13+TFA) : δ 1.80 (2H, quint, J= 7.4 Hz), 2.21 (2H, dd, J= 8.1, 7.4 Hz), 2.27 (3H, s), 2.41 (3H, s), 2.45 (2H, t, J- 7.1 Hz), 4.21 (2H, s), 7.25 (2H, d, J= 8.3 Hz), 7.33 (5H, s), 7.82 (2H, d, J= 8.3 Hz), 10.06 (1H, br). ¾ -Crash (300MHz, CDC1 3 + TFA): δ 1.80 (2H, quint, J = 7.4 Hz), 2.21 (2H, dd, J = 8.1, 7.4 Hz), 2.27 (3H, s), 2.41 (3H, s), 2.45 (2H, t, J- 7.1 Hz), 4.21 (2H, s), 7.25 (2H, d, J = 8.3 Hz), 7.33 (5H, s), 7.82 (2H, d, J = 8.3) Hz), 10.06 (1H, br).
元素分析値: C25H25N305S3 · 0.4Η2 として計算値: C, 54.51; H, 4.72; N, 7.63; S, 17.46 (%) 、 実測値: C, 54.50; H, 4.33; N, 7.63; S, 17.51 (%) . Elemental analysis: C 25 H 25 N 3 0 5 S 3 · 0.4Η 2 Calculated: C, 54.51; H, 4.72 ; N, 7.63; S, 17.46 (%), Found: C, 54.50; H, 4.33; N, 7.63; S, 17.51 (%).
実施例 4-410 Example 4-410
一 NMR (300MHz, CDC13) : δ 1.77 (2Η, quint, J= 7. Hz), 2.16 (2H, brdd, J= 8.0, 7.3 Hz), 2.25 (3H, s), 2.34 (2H, t, J= 7.1 Hz), 2.40 (3H, s), 3.69 (3H, s), 4.19 (2H, s), 7.24 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J= 8.3 Hz), 9.65 (1H, br). One NMR (300MHz, CDC1 3): δ 1.77 (2Η, quint, J = 7. Hz), 2.16 (2H, brdd, J = 8.0, 7.3 Hz), 2.25 (3H, s), 2.34 (2H, t, J = 7.1 Hz), 2.40 (3H, s), 3.69 (3H, s), 4.19 (2H, s), 7.24 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J = 8.3 Hz), 9.65 (1H, br).
元素分析値: C26H27N305S3として計算値: C, 55.99; H, 4.88; N, 7.53; S, 17.25 ( ) 、 実測値: C, 55.74; H, 4.90; N, 7.37; S, 17.41 (%) . Elemental analysis: C 26 H 27 N 3 0 5 S 3 Calculated: C, 55.99; H, 4.88; N, 7.53; S, 17.25 (), found: C, 55.74; H, 4.90; N, 7.37 ; S, 17.41 (%).
実施例 4-411 Example 4-411
Ή一 NMR (300MHz, DMS0 - d6) : δ 2.36 (3Η, s), 4.29 (2H, s), 5.44 (1H, t, J= 7.9 Hz), 6.68 (1H, d, J= 7.9 Hz), 7.32 (2H, d, J= 8.2 Hz), 7.33 (1H, m), 7.36 (4H, m), 7.79 (2H, d, J= 8.2 Hz), 11.78 (1H, br). Ή one NMR (300MHz, DMS0 - d 6 ): δ 2.36 (3Η, s), 4.29 (2H, s), 5.44 (1H, t, J = 7.9 Hz), 6.68 (1H, d, J = 7.9 Hz) , 7.32 (2H, d, J = 8.2 Hz), 7.33 (1H, m), 7.36 (4H, m), 7.79 (2H, d, J = 8.2 Hz), 11.78 (1H, br).
元素分析値: C2。H17N303S3 · 0.5H20として計算値: C, 53.08; H, 4.01; , 9.28; S, 21.25 (%), 実測値: C, 53.10; H, 3.86; N, 9.26; S, 21.41 (%) . Elemental analysis: C 2. H 17 N 3 0 3 S 3 · 0.5H 2 0 Calculated: C, 53.08; H, 4.01;, 9.28; S, 21.25 (%), Found: C, 53.10; H, 3.86; N, 9.26; S, 21.41 (%).
実施例 4-412 Example 4-412
Ή - NMR (300MHz, DMSO- d6) : δ 2.15 (3H, d, J= 1.1 Hz), 2.36 (3H, s), 4.27 (2H, s), 5.13 (1H, q J= 1.1 Hz), 7.31 (2H, d, J= 8.3 Hz), 7.32 (2H, m), 7.36 (3H, m), 7.79 (2H, d, J= 8.3 Hz), 11.70 (1H, br). Ή-NMR (300 MHz, DMSO-d 6 ): δ 2.15 (3H, d, J = 1.1 Hz), 2.36 (3H, s), 4.27 (2H, s), 5.13 (1H, q J = 1.1 Hz), 7.31 (2H, d, J = 8.3 Hz), 7.32 (2H, m), 7.36 (3H, m), 7.79 (2H, d, J = 8.3 Hz), 11.70 (1H, br).
元素分析値: C21H19N303S3 · 0.2¾0として計算値: C, 54.69; H, 4.24; N, 9.11; S, 20.85 (%) .実測値: (;, 54.69; H, 4.12; N, 9.17; S, 21.08 (%) . Elemental analysis: C 21 H 19 N 3 0 3 Calculated S 3 · 0.2¾0:. C, 54.69; H, 4.24; N, 9.11; S, 20.85 (%) Found: (;, 54.69; H, 4.12; N, 9.17; S, 21.08 (%).
実施例 4-413 Example 4-413
Ή-NMR (CDC13, 300 MHz): δ 1.01 (6Η, d, J= 6.6 Hz), 1.90 (1H, sept, J= 6.7 Hz), 2.27 (3H, d, J= 1.0 Hz), 2.42 (3H, s), 2.78 (2H, d, J= 6.9 Hz), 4.63 (1H, q, J= 1.1 Hz), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.68 (1H, brs). Ή-NMR (CDC1 3, 300 MHz): δ 1.01 (6Η, d, J = 6.6 Hz), 1.90 (1H, sept, J = 6.7 Hz), 2.27 (3H, d, J = 1.0 Hz), 2.42 ( 3H, s), 2.78 (2H, d, J = 6.9 Hz), 4.63 (1H, q, J = 1.1 Hz), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz) Hz), 9.68 (1H, brs).
元素分析値: Cl8H21N303S3として計算値: C, 51.04; H, 5.00; N, 9.92; S, 22.71 (%)、 実測値: C, 50.83; H, 4.89; N, 10.10; S, 22.74 (%) . Elemental analysis: C l8 H 21 N 3 0 3 S 3 Calculated: C, 51.04; H, 5.00 ; N, 9.92; S, 22.71 (%), Found: C, 50.83; H, 4.89 ; N, 10.10; S, 22.74 (%).
実施例 4-414 Example 4-414
Ή-NMR (CDC13, 300 MHz): 6 2.27 (3H, d, J= 1.1 Hz), 2.43 (3H, s), 2.97 (2H, brdd, J= 8.7, 7.1 Hz), 3.15 (2H, brdd, J= 9.1, 6.4 Hz), 4.63 (1H, brq, J= 1.1 Hz), 7.18 (2H, brd, J= 8 Hz) , 7.25-7.32 (5H, m), 7.82 (2H, d, J= 8.3 Hz), 9.67 (1H, brs). Ή-NMR (CDC1 3, 300 MHz): 6 2.27 (3H, d, J = 1.1 Hz), 2.43 (3H, s), 2.97 (2H, brdd, J = 8.7, 7.1 Hz), 3.15 (2H, brdd , J = 9.1, 6.4 Hz), 4.63 (1H, brq, J = 1.1 Hz), 7.18 (2H, brd, J = 8 Hz), 7.25-7.32 (5H, m), 7.82 (2H, d, J = 8.3 Hz), 9.67 (1H, brs).
元素分析値: C22H2lN303S3として計算値: C, 56.03; H, 4.49; N, 8.91; S, 20.40Elemental analysis: C 22 H 2l N 3 0 3 S 3 Calculated: C, 56.03; H, 4.49 ; N, 8.91; S, 20.40
(%)、 実測値: C, 56.12; H, 4.52; N, 8.83; S, 20.28 (%) . ■ (%), Found: C, 56.12; H, 4.52; N, 8.83; S, 20.28 (%).
実施例 4-415 Example 4-415
'H-NMR (CDC13, 300 MHz): δ 2.14 (2Η, quint, J= 7.2 Hz), 2.26 (3H, d, J= 1.5 Hz), 2.38 (3H, s), 3.05 (2H, t, J= 7.2 Hz), 3.12 (2H, t, J= 7.2 Hz) , 4.61 (1H, brq, J= 1.1 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.48 (2H, t, J= 7.5 Hz), 7.59 (1H, tt, J= 7.2, 1.9 Hz), 7.83 (2H, d, J= 8.3 Hz), 7.95 (2H, brd, J= 8 Hz), 9.68 (1H, brs). 'H-NMR (CDC1 3, 300 MHz): δ 2.14 (2Η, quint, J = 7.2 Hz), 2.26 (3H, d, J = 1.5 Hz), 2.38 (3H, s), 3.05 (2H, t, J = 7.2 Hz), 3.12 (2H, t, J = 7.2 Hz), 4.61 (1H, brq, J = 1.1 Hz), 7.25 (2H, d, J = 8.3 Hz), 7.48 (2H, t, J = 7.5 Hz), 7.59 (1H, tt, J = 7.2, 1.9 Hz), 7.83 (2H, d, J = 8.3 Hz), 7.95 (2H, brd, J = 8 Hz), 9.68 (1H, brs).
元素分析値: C24H23N304S3として計算値: C, 56.12; H, 4.51; N, 8.18; S, 18.73 ( )> 実測値: C, 55.85; H, 4.48; N, 8.25; S, 18.65 (%) . Elemental analysis: C 24 H 23 N 3 0 4 S 3 Calculated: C, 56.12; H, 4.51 ; N, 8.18; S, 18.73 ()> Found: C, 55.85; H, 4.48 ; N, 8.25 ; S, 18.65 (%).
実施例 4-416 Example 4-416
8 - (ィソぺンチルスルファニル) - 1 - {[ (4 -メチルフエニル)スルホニル]イミノト 3 - ォキソ - 2,3-ジヒドロ- 1H-イミダゾ [5,1- c] [1, 4]チアジン- 5-カルボン酸 ェチル 実施例 3-20で得た化合物 (400 mg, 0.940 mmol)および炭酸力リゥム (195 mg, 1.41 腿 ol) の DMF (6.5 ml) の懸濁液に 60°Cで攪拌しながらヨウ化イソペンチ ル (0.137 il, 1.03 腿 ol)を加え、 混合物を 3時間攪拌した。 反応液に 1N塩酸 (1.9ml)およびメタノール一水 a:l, 10ml)を加え、 室温で 30分間 M拌した。 析 出物を濾取し、 メタノール一水 (2:1 および 1:1)で洗浄、 乾燥して標題化合物 (439 mg, 0.886 腿 ol)を赤色粉末として得た。 8- (Isopentylsulfanyl) -1-([(4-methylphenyl) sulfonyl] iminoto 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 5-Carboxylate The compound obtained in Example 3-20 (400 mg, 0.940 mmol) and carbonated lime (195 mg, Isopentyl iodide (0.137 il, 1.03 t) was added to a suspension of 1.41 t) in DMF (6.5 ml) while stirring at 60 ° C, and the mixture was stirred for 3 hours. 1N Hydrochloric acid (1.9 ml) and methanol / aqueous a: l (10 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (2: 1 and 1: 1), and dried to give the title compound (439 mg, 0.886 mol) as a red powder.
Ή-NMR (200MHz, CDC13) : δ 0.92 (6H, d, J= 6.2 Hz), 1.32 (3H, t, J= 7.1 Hz), 1.55 (2H, q, J= 7.6 Hz), 1.64 (1H, sept, J= 6.6 Hz), 2.43 (3H, s), 2.95 (2H, t, J= 7.5 Hz) , 4.32 (1H, q, J= 7.2 Hz), 5.89 (1H, s), 7.31 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.2 Hz), 9.76 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.92 (6H, d, J = 6.2 Hz), 1.32 (3H, t, J = 7.1 Hz), 1.55 (2H, q, J = 7.6 Hz), 1.64 (1H , sept, J = 6.6 Hz), 2.43 (3H, s), 2.95 (2H, t, J = 7.5 Hz), 4.32 (1H, q, J = 7.2 Hz), 5.89 (1H, s), 7.31 (2H , D, J = 8.4 Hz), 7.86 (2H, d, J = 8.2 Hz), 9.76 (1H, brs).
元素分析値: C21H25N305S3として計算値: C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%) 、 実測値: C, 50.85; H, 5.20; N, 8.34; S, 19.29 (%) . Elemental analysis: Calculated for C 21 H 25 N 3 0 5 S 3 : C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%), found: C, 50.85; H, 5.20; N, 8.34; S, 19.29 (%).
融点. 158.5-158.8°C Melting point.158.5-158.8 ° C
実施例 4-420 Example 4-420
!H -蘭 R (300MHz, CDC13) : δ 0.89 (3Η, t, J= 6.9 Hz), 1.28 (6H, s), 1.31 (2H, m), 1.40 (2H, m), 1.70 (2H, brquint, J= 7.4 Hz), 2.43 (3H, s), 2.97 (2H, dd,! H - Ran R (300MHz, CDC1 3): δ 0.89 (3Η, t, J = 6.9 Hz), 1.28 (6H, s), 1.31 (2H, m), 1.40 (2H, m), 1.70 (2H, brquint, J = 7.4 Hz), 2.43 (3H, s), 2.97 (2H, dd,
J= 7.6, 7.3 Hz), 4.98 (1H, s), 7.21 (2H, dd, J= 7.9, 2.0 Hz), 7.31 (2H, d,J = 7.6, 7.3 Hz), 4.98 (1H, s), 7.21 (2H, dd, J = 7.9, 2.0 Hz), 7.31 (2H, d,
J= 8.3 Hz), 7.37 (3H, m), 7.88 (2H, d, J= 8.3 Hz), 9.68 (1H, brs). J = 8.3 Hz), 7.37 (3H, m), 7.88 (2H, d, J = 8.3 Hz), 9.68 (1H, brs).
元素分析値: C27H31N303S3として計算値: C, 59.86; H, 5.77; N, 7.76; S, 17.76 (%), 実測値: C, 59.65; H, 5.73; N, 7.75; S, 17.95 ( ) . Elemental analysis: C 27 H 31 N 3 0 3 S 3 Calculated: C, 59.86; H, 5.77 ; N, 7.76; S, 17.76 (%), Found: C, 59.65; H, 5.73 ; N, 7.75; S, 17.95 ().
実施例 4-421 Example 4-421
'H-NMR (300MHz, CDC13) : δ 0.90 (3Η, t, ]= 6.9 Hz), 1.29 (6H, brs), 1.32 (2H, m), 1.41 (2H, brquint, J= 7.2 Hz), 1.71 (2H, brquint, J= 7.4 Hz), 2.43 (3H, s), 2.97 (2H, t, J= 7.4 Hz), 5.04 (1H, s), 7.27 (2H, d, J= 8.2 Hz), 7.31 (2H, d, J= 8.2 Hz), 7.36 (1H, brt, J= 7.2 Hz), 7.44 (2H, brt, J= 7.7 Hz), 7.58 (2H, d, J= 8.2 Hz), 7.59 (2H, brd, J= 7.2 Hz), 7.89 (2H, d, J= 8.2 Hz), 9.72 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.90 (3Η, t,] = 6.9 Hz), 1.29 (6H, brs), 1.32 (2H, m), 1.41 (2H, brquint, J = 7.2 Hz), 1.71 (2H, brquint, J = 7.4 Hz), 2.43 (3H, s), 2.97 (2H, t, J = 7.4 Hz), 5.04 (1H, s), 7.27 (2H, d, J = 8.2 Hz), 7.31 (2H, d, J = 8.2 Hz), 7.36 (1H, brt, J = 7.2 Hz), 7.44 (2H, brt, J = 7.7 Hz), 7.58 (2H, d, J = 8.2 Hz), 7.59 ( 2H, brd, J = 7.2 Hz), 7.89 (2H, d, J = 8.2 Hz), 9.72 (1H, brs).
実施例 4-422 Example 4-422
'H-NMR (300MHz, CDC13) : δ 1.30 (3H, t, J= 7.2 Hz), 2.43 (3H, s), 3.74 (2H, s), 4.24 (2H, q, J= 7.2 Hz), 4.94 (1H, s), 6.94 (2H, dt, J= 7.7, 1.9 Hz), 7.05 (2H, dq, J= 8.3, 0.8 Hz), 7.14 (2H, d, J= 8.7 Hz), 7.15 (1H, m), 7.31 (2H, d, J= 8.3 Hz), 7.36 (2H, brt, J= 7.5 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.67 (1H, brs) . 'H-NMR (300MHz, CDC1 3): δ 1.30 (3H, t, J = 7.2 Hz), 2.43 (3H, s), 3.74 (2H, s), 4.24 (2H, q, J = 7.2 Hz), 4.94 (1H, s), 6.94 (2H, dt, J = 7.7, 1.9 Hz), 7.05 (2H, dq, J = 8.3, 0.8 Hz), 7.14 (2H, d, J = 8.7 Hz), 7.15 (1H, m), 7.31 (2H, d, J = 8.3 Hz), 7.36 (2H, brt, J = 7.5 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.67 (1H, brs).
元素分析値: C29H25N306S3として計算値: C, 57.31; H, 4.15; N, 6.91; S, 15.83 ( ) 、 実測値: C, 57.35; H, 4.12; N, 7.31; S, 15.88 (%) . Elemental analysis: C 29 H 25 N 3 0 6 S 3 Calculated: C, 57.31; H, 4.15 ; N, 6.91; S, 15.83 (), Found: C, 57.35; H, 4.12 ; N, 7.31 ; S, 15.88 (%).
実施例 4-423 Example 4-423
lH— NMR (300MHz, CDC13) : δ 2.38 (3H, s), 3.81 (2H, s), 4.90 (1H, s), 6.88 (2H, brd, J= 7. 9 Hz), 7.02 (2H, brd, J= 7.9 Hz), 7.08 (2H, brd, J= 8.3 Hz), 7.13 (1H, brt, J= 7.2 Hz) , 7.29 (2H, d, J= 8.3 Hz) , 7.32 (2H, t, J= 7.8 Hz), 7.86 (2H, d, J= 7.9 Hz). lH- NMR (300MHz, CDC1 3) : δ 2.38 (3H, s), 3.81 (2H, s), 4.90 (1H, s), 6.88 (2H, brd, J = 7. 9 Hz), 7.02 (2H, brd, J = 7.9 Hz), 7.08 (2H, brd, J = 8.3 Hz), 7.13 (1H, brt, J = 7.2 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.32 (2H, t, J = 7.8 Hz), 7.86 (2H, d, J = 7.9 Hz).
実施例 4-424 Example 4-424
酢酸ェチル -n-へキサンから再結晶して融点 155-157°Cの暗赤色結晶を得た。 Recrystallization from ethyl acetate-n-hexane gave dark red crystals with a melting point of 155-157 ° C.
赤外吸収スぺクトル(IR) (KBr中) ソ: 3550-3000, 2928, 2905, 2857, 1732, 1651, 1611, 1557 cm"1.Infrared absorption spectrum (IR) (in KBr) So: 3550-3000, 2928, 2905, 2857, 1732, 1651, 1611, 1557 cm " 1 .
— NMR (CDC13) : δ 0.909 (3Η, t, J=7.0 Hz), 1.20-1.48 (4H, m), 1.56-1.76 (2H, m), 2.34 (3H, s), 2.42 (3H, s), 2.97 (2H, t, J=7.6 Hz), 4.25 (2H, d, J=6.0 Hz), 7.29 (2H, d, J=8.0 Hz), 7.86 (2H, d, J=8.4 Hz), 9.67 (1H, bs). - NMR (CDC1 3): δ 0.909 (3Η, t, J = 7.0 Hz), 1.20-1.48 (4H, m), 1.56-1.76 (2H, m), 2.34 (3H, s), 2.42 (3H, s ), 2.97 (2H, t, J = 7.6 Hz), 4.25 (2H, d, J = 6.0 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.67 (1H, bs).
元素分析値 C2。H25N304S3として Elemental analysis value C 2. As H 25 N 3 0 4 S 3
計算値 (¾) : C, 51.37; H, 5.39; N, 8.99; S, 20.57. Calculated (¾): C, 51.37; H, 5.39; N, 8.99; S, 20.57.
実測値 (¾) : C, 51.23; H, 5.48; N, 8.77; S, 20.59. Found (¾): C, 51.23; H, 5.48; N, 8.77; S, 20.59.
実施例 4-425 Example 4-425
Ή-NMR (300MHz, CDC13) : δ 2.41 (3Η, s), 2.48 (2H, brt, J= 6.6 Hz), 3.81 (2H, i, J= 6.6 Hz), 4.22 (2H, s), 6.62 (1H, brs), 7.25 (2H, brd, J= 8.3 Hz), 7.32 (5H, s), 7.73 (1H, dt, J= 1.1, 7.5 Hz), 7.79 (2H, d, J= 8.2 Hz), 7.80 (1H, dt, J= 1.6, 7.5 Hz), 7.92 (1H, d, J= 7.6 Hz), 7.95 (1H, d, J= 7.5 Hz), 9.69 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 2.41 (3Η, s), 2.48 (2H, brt, J = 6.6 Hz), 3.81 (2H, i, J = 6.6 Hz), 4.22 (2H, s), 6.62 (1H, brs), 7.25 (2H, brd, J = 8.3 Hz), 7.32 (5H, s), 7.73 (1H, dt, J = 1.1, 7.5 Hz), 7.79 (2H, d, J = 8.2 Hz) , 7.80 (1H, dt, J = 1.6, 7.5 Hz), 7.92 (1H, d, J = 7.6 Hz), 7.95 (1H, d, J = 7.5 Hz), 9.69 (1H, brs).
元素分析値: C30¾4N405S3 · 0.2H20として計算値: C, 58.08; H, 3.96; N, 9.03; S, 15.51 (%) 、 実測値: C, 57.97; H, 3.96; N, 8.92; S, 15.58 (%) , 実施例 4-426 Ή-N R (300MHz, DMS0-d6) : δ 2.33 (2H, brt, J= 6.3 Hz), 2.36 (3H, s), ca. 2.5 (2H, brt, J= 6.3 Hz), 4.32 (2H, s), 6.68 (1H, brs), 7.31 (2H, brd, J= 8.2 Hz), 7.34 (5H, m), 7.39 (1H, dd, J- 7.6, 1.6 Hz), 7.52 (2H, dquint, J= 2.0, 7.6 Hz), 7.77 (1H, dd, J= 7.6, 1.6 Hz), 7.79 (2H, d, J= 8.2 Hz), 8.41 (1H, brt, J= 5.7 Hz), 11.81 (1H, br), 12.92 (1H, brs). Elemental analysis: C 30 ¾ 4 N 4 0 5 S 3 · 0.2H 2 0 Calculated: C, 58.08; H, 3.96; N, 9.03; S, 15.51 (%), found: C, 57.97; H , 3.96; N, 8.92; S, 15.58 (%), Example 4-426 Ή-NR (300 MHz, DMS0-d 6 ): δ 2.33 (2H, brt, J = 6.3 Hz), 2.36 (3H, s), ca.2.5 (2H, brt, J = 6.3 Hz), 4.32 (2H, s), 6.68 (1H, brs), 7.31 (2H, brd, J = 8.2 Hz), 7.34 (5H, m), 7.39 (1H, dd, J-7.6, 1.6 Hz), 7.52 (2H, dquint, J = 2.0, 7.6 Hz), 7.77 (1H, dd, J = 7.6, 1.6 Hz), 7.79 (2H, d, J = 8.2 Hz), 8.41 (1H, brt, J = 5.7 Hz), 11.81 (1H, br ), 12.92 (1H, brs).
元素分析値: C30H26N406S3 · 0.5¾0として計算値: C, 55.97; H, 4.23; N, 8.70; S, 14.94 (%) .実測値: C, 56.09; H, 4.29; N, 8.76; S, 15.05 {%) · Elemental analysis: C 30 H 26 N 4 0 6 S Calculated 3 · 0.5¾0:. C, 55.97 ; H, 4.23; N, 8.70; S, 14.94 (%) Found: C, 56.09; H, 4.29 ; N, 8.76; S, 15.05 {%)
実施例 4-427 Example 4-427
'H-N R (CDClg, 200 MHz): δ 1.10 (6H, d, J= 6.8 Hz), 1.44 (4H, m), 1.5-1.7 (4H, m), 2.28 (3H, s), 2.41 (3H, s), 2.95 (1H, quint, J= 7.2 Hz), 2.98 (2H, i, J- 7.2 Hz), 3.66 (2H, t, J= 6.2 Hz), 7.28 (2H, d, J= 7.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.67 (1H, brs). 'HNR (CDClg, 200 MHz): δ 1.10 (6H, d, J = 6.8 Hz), 1.44 (4H, m), 1.5-1.7 (4H, m), 2.28 (3H, s), 2.41 (3H, s), 2.95 (1H, quint, J = 7.2 Hz), 2.98 (2H, i, J- 7.2 Hz), 3.66 (2H, t, J = 6.2 Hz), 7.28 (2H, d, J = 7.0 Hz) , 7.85 (2H, d, J = 8.4 Hz), 9.67 (1H, brs).
元素分析値: C23H31N304S3として計算値: C, 54.20; H, 6.13; N, 8.24; S, 18.87 (%)、 実測値: C, 54.19; H, 6.18; N, 8.37; S, 18.59 (%) · Elemental analysis: C 23 H 31 N 3 0 4 S 3 Calculated: C, 54.20; H, 6.13 ; N, 8.24; S, 18.87 (%), Found: C, 54.19; H, 6.18 ; N, 8.37; S, 18.59 (%)
実施例 4-428 Example 4-428
Ή -賺 (CDC13, 200 MHz): <5 1.03 (6H, d, J= 6.8 Hz), 1.10 (6H, d, J= 7.0 Hz), 1.91 (1H, sept, J= 6.6 Hz), 2.28 (3H, s), 2.41 (3H, s), 2.86 (2H, d, J= 7.0 Hz), 2.95 (1H, quint, J= 7.0 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). E -賺(CDC1 3, 200 MHz): <5 1.03 (6H, d, J = 6.8 Hz), 1.10 (6H, d, J = 7.0 Hz), 1.91 (1H, sept, J = 6.6 Hz), 2.28 (3H, s), 2.41 (3H, s), 2.86 (2H, d, J = 7.0 Hz), 2.95 (1H, quint, J = 7.0 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C21H27N303S3として計算値: C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%)、 実測値: C, 53.96; H, 5.80; N, 9.14; S, 20.63 (%) . Elemental analysis: Calculated for C 21 H 27 N 3 0 3 S 3 : C, 54.17; H, 5.84; N, 9.02; S, 20.66 (%), found: C, 53.96; H, 5.80; N, 9.14; S, 20.63 (%).
実施例 4-429 Example 4-429
Ή-NMR (CDC13, 200 MHz): δ 1.11 (6Η, d, J= 6.8 Hz), 2.28 (3H, s), 2.42 (3H, s), 2.96 (1H, quint, J= 7.0 Hz), 2.99 (2H, brdd, J= 8.8, 5.2 Hz), 3.21 (2H, dd, J= 9.2, 5.6 Hz), 7.18-34 (7H, m), 7.84 (2H, d, J= 8. Hz), 9.66 (1H, brs). 元素分析値: C25H27N303S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73 Ή-NMR (CDC1 3, 200 MHz): δ 1.11 (6Η, d, J = 6.8 Hz), 2.28 (3H, s), 2.42 (3H, s), 2.96 (1H, quint, J = 7.0 Hz), 2.99 (2H, brdd, J = 8.8, 5.2 Hz), 3.21 (2H, dd, J = 9.2, 5.6 Hz), 7.18-34 (7H, m), 7.84 (2H, d, J = 8. Hz), . 9.66 (1H, brs) elemental analysis: C 25 H 27 N 3 0 3 S 3 calculated: C, 58.45; H, 5.30 ; N, 8.18; S, 18.73
(%), 実測値: C, 58.55; H, 5.24; N, 8.21; S, 18.62 (%) . (%), Found: C, 58.55; H, 5.24; N, 8.21; S, 18.62 (%).
実施例 4-430 Example 4-430
Ή -丽 (CDC13, 200 MHz): δ 1.08 (6Η, d, J= 7.0 Hz), 2.16 (2H, quint, J= 6.9 Hz), 2.27 (3H, s), 2.38 (3H, s), 2.94 (1H, quint, J= 7.0 Hz), 3.13 (2H, t, J= 7.0 Hz), 3.15 (2H, t, J= 7.0 Hz), 7.24 (2H, d, J= 8.0 Hz), 7.48 (2H, t, J= 7.4 Hz), 7.59 (1H, t, J- 7.2 Hz), 7.84 (2H, d, J= 8.4 Hz), 7.96 (2H, dm, J= 7.8 Hz), 9.66 (1H, brs). E -丽(CDC1 3, 200 MHz): δ 1.08 (6Η, d, J = 7.0 Hz), 2.16 (2H, quint, J = 6.9 Hz), 2.27 (3H, s), 2.38 (3H, s), 2.94 (1H, quint, J = 7.0 Hz), 3.13 (2H, t, J = 7.0 Hz), 3.15 (2H, t, J = 7.0 Hz), 7.24 (2H, d, J = 8.0 Hz), 7.48 (2H, t, J = 7.4 Hz), 7.59 (1H, t, J- 7.2 Hz), 7.84 (2H, d, J = 8.4 Hz) ), 7.96 (2H, dm, J = 7.8 Hz), 9.66 (1H, brs).
元素分析値: C27H29N304S3として計算値: C, 58.35; H, 5.26; N, 7.56; S, 17.31 (%) 実測値: C, 58.19; H, 5.24; N, 7.61; S, 17.29 ( ) . Elemental analysis: C 27 H 29 N 3 0 4 S 3 Calculated: C, 58.35; H, 5.26 ; N, 7.56; S, 17.31 (%) Found: C, 58.19; H, 5.24 ; N, 7.61 S, 17.29 ().
実施例 4-431 Example 4-431
Ή-NMR (300MHz, DMSO— d6) : δ 0.85 (3Η, t, J= 6.8 Hz), 1.25 (6H, brs), 1.30 (2H, m), 1.42 (2H, brquint, J= 6.5 Hz), 2.17 (3H, s), 2.38 (3H, s), 2.62 (2H, t, J= 7.2 Hz), 3.14 (2H, t, J= 7.2 Hz), 7.37 (2H, d, J= 8.3 Hz), 7.83 (2H, d, J= 8.3 Hz), 11.67 (1H, br), 12.50 (1H, br). Ή-NMR (300MHz, DMSO— d 6 ): δ 0.85 (3Η, t, J = 6.8 Hz), 1.25 (6H, brs), 1.30 (2H, m), 1.42 (2H, brquint, J = 6.5 Hz) , 2.17 (3H, s), 2.38 (3H, s), 2.62 (2H, t, J = 7.2 Hz), 3.14 (2H, t, J = 7.2 Hz), 7.37 (2H, d, J = 8.3 Hz) , 7.83 (2H, d, J = 8.3 Hz), 11.67 (1H, br), 12.50 (1H, br).
元素分析値: C24H31N305S3として計算値: C, 53.61; H, 5.81; N, 7.81; S, 17.89 (%) 、 実測値: C, 53.31; H, 5.73; N, 7.82; S, 17.85 (%) . Elemental analysis: C 24 H 31 N 3 0 5 S 3 Calculated: C, 53.61; H, 5.81 ; N, 7.81; S, 17.89 (%), Found: C, 53.31; H, 5.73 ; N, 7.82; S, 17.85 (%).
実施例 4-432 Example 4-432
Ή-NMR (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.28 (8H, brs), 1.43 (2H, brquint, J= 7 Hz), 2.09 (2H, dd, J= 7.9, 7.2 Hz), 2.24 (3H, s), 2.40 (3H, s)., 4.20 (2H, s), 7.24 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.81 (2H, d, J= 8.3 Hz), 9.64 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.28 (8H, brs), 1.43 (2H, brquint, J = 7 Hz), 2.09 (2H, dd, J = 7.9, 7.2 Hz), 2.24 (3H, s), 2.40 (3H, s)., 4.20 (2H, s), 7.24 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.81 (2H , d, J = 8.3 Hz), 9.64 (1H, br).
元素分析値: C28H33N303S3として計算値: C, 60.51; H, 5.98; N, 7.56; S, 17.31 (%), 実測値: C, 60.22; H, 5.78; N, 7.75; S, 17.61 (%) . Elemental analysis: C 28 H 33 N 3 0 3 S 3 Calculated: C, 60.51; H, 5.98; N, 7.56; S, 17.31 (%), found: C, 60.22; H, 5.78; N, 7.75; S, 17.61 (%).
実施例 4-433 Example 4-433
Ή-NMR (300MHz, CDCI3) : δ 0.89 (3Η, t, J= 6.9 Hz), 1.28 (6H, brs), 1.31 (2H, m), 1.44 (2H, brquint, J= 6.6 Hz), 2.04 (2H, brdd, J= 7.4, 7.1 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J= 0.5 Hz), 7.25 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J= 8.3 Hz), 9.72 (1H, br). Ή-NMR (300 MHz, CDCI 3 ): δ 0.89 (3Η, t, J = 6.9 Hz), 1.28 (6H, brs), 1.31 (2H, m), 1.44 (2H, brquint, J = 6.6 Hz), 2.04 (2H, brdd, J = 7.4, 7.1 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, brt, J = 0.5 Hz), 7.25 (2H, d, J = 8.3 Hz) , 7.32 (5H, s), 7.80 (2H, d, J = 8.3 Hz), 9.72 (1H, br).
元素分析値: C27H31N303S3として計算値: C, 59.86; H, 5.77; N, 7.76; S, 17.76Elemental analysis: C 27 H 31 N 3 0 3 S 3 Calculated: C, 59.86; H, 5.77 ; N, 7.76; S, 17.76
(%) .実測値: C, 59.87; H, 5.70; N, 7.74; S, 17.63 (%) . Found: C, 59.87; H, 5.70; N, 7.74; S, 17.63 (%).
実施例 4-434 Example 4-434
Ή-N R (300MHz, CDC13) : <5 0.85 (3H, t, J= 7.2 Hz), 1.19 (6H, brs), 1.25 (2H, m), 1.43 (2H, brquint, J= 7.5 Hz), 2.39 (2H, brdd, J= 8.3, 6.5 Hz), 2.42 (3H, s), 2.59 (2H, dd, J= 7.9, 7.2 Hz), 3.55 (2H, dd, J= 7.9, 7.2 Hz), 4.48 (2H, d, J= 6.0 Hz), 6.57 (1H, brt, J= 5.7 Hz), 7.25 (2H, m), 7.30 (2H, d, J= 8.3 Hz), 7.33 (3H, m), 7.45 (5H, s), 7.71 (2H, d, J= 8.3 Hz), 9.52 (1H, brs). 実施例 -435 Ή-NR (300MHz, CDC1 3 ): <5 0.85 (3H, t, J = 7.2 Hz), 1.19 (6H, brs), 1.25 (2H, m), 1.43 (2H, brquint, J = 7.5 Hz), 2.39 (2H, brdd, J = 8.3, 6.5 Hz), 2.42 (3H, s), 2.59 (2H, dd, J = 7.9, 7.2 Hz), 3.55 (2H, dd, J = 7.9, 7.2 Hz), 4.48 (2H, d, J = 6.0 Hz), 6.57 (1H, brt, J = 5.7 Hz), 7.25 (2H, m), 7.30 (2H, d , J = 8.3 Hz), 7.33 (3H, m), 7.45 (5H, s), 7.71 (2H, d, J = 8.3 Hz), 9.52 (1H, brs).
Ή-NMR (300MHz, CDC13) : δ 0.88 (3H, t, J= 7.2 Hz), 1.27 (8H, brs), 1.30 (3H, t, J= 7.2 Hz), 1.44 (2H, brquint, J= 7.2 Hz), 2.05 (2H, brdd, J= 7.5, 6.5 Hz), 2.42 (3H, s), 3.74 (2H, s), 4.23 (2H, q, J= 7.2 Hz), 6.49 (1H, brt, J= 1 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.80 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3H, t, J = 7.2 Hz), 1.27 (8H, brs), 1.30 (3H, t, J = 7.2 Hz), 1.44 (2H, brquint, J = 7.2 Hz), 2.05 (2H, brdd, J = 7.5, 6.5 Hz), 2.42 (3H, s), 3.74 (2H, s), 4.23 (2H, q, J = 7.2 Hz), 6.49 (1H, brt, J = 1 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.80 (1H, br).
元素分析値: C25H33N305S3として計算値: C, 54.42; H, 6.03; N, 7.62; S, 17.43Elemental analysis: C 25 H 33 N 3 0 5 S 3 Calculated: C, 54.42; H, 6.03 ; N, 7.62; S, 17.43
(%)、 実測値: C, 54.42; H, 6.06; N, 7.46; S, 17.46 ( ) . (%), Found: C, 54.42; H, 6.06; N, 7.46; S, 17.46 ().
実施例 4-436 Example 4-436
Ή-NMR (300MHz, CDC13) : δ 0.88 (3Η, brt, J= 7.2 Hz), 1.26 (10H, brs), 1.44 (2H, brquint, J= 6.8 Hz), 2.05 (2H, brt, J= 7.2 Hz), 2.40 (3H, s), 3.81 (2H, s), 6.49 (1H, brt, J= 1 Hz), 6.91 (1H, s), 7.29 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.98 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3Η, brt, J = 7.2 Hz), 1.26 (10H, brs), 1.44 (2H, brquint, J = 6.8 Hz), 2.05 (2H, brt, J = 7.2 Hz), 2.40 (3H, s), 3.81 (2H, s), 6.49 (1H, brt, J = 1 Hz), 6.91 (1H, s), 7.29 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.98 (1H, br).
元素分析値: C23H29N305S3: C, 52.75; H, 5.58; N, 8.02 (%) . Elemental analysis: C 23 H 29 N 3 0 5 S 3: C, 52.75; H, 5.58; N, 8.02 (%).
実測値: C, 52.51; H, 5.58; N, 8.06 (%) . Found: C, 52.51; H, 5.58; N, 8.06 (%).
実施例 4-437 Example 4-437
Ή-N R (300MHz, CDC13) : δ 0.89 (3H, t, J= 6.8 Hz), 1.27 (10H, brs), 1.44 (2H, brquint, J= 7 Hz), 2.04 (2H, brdd, J= 7.2, 6.8 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, s), 7.25 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J= 8.3 Hz), 9.72 (1H, br). Ή-NR (300MHz, CDC1 3 ): δ 0.89 (3H, t, J = 6.8 Hz), 1.27 (10H, brs), 1.44 (2H, brquint, J = 7 Hz), 2.04 (2H, brdd, J = 7.2, 6.8 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (1H, s), 7.25 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.80 (2H, d, J = 8.3 Hz), 9.72 (1H, br).
実施例 4-438 Example 4-438
Ή-NMR (300MHz, CDC13) : δ 0.88 (3H, brt, ]= 6.8 Hz), 0.95 (3H, brt, J= 7.5 Hz), 1.26 (10H, brs), 1.44 (2H, m), 1.68 (2H, q, J= 7.2 Hz), 2.41 (3H, s), 2.44 (2H, brdd, J= 7.5, 7.2 Hz), 3.75 (2H, s), 4.13 (2H, t, J= 6.8 Hz), 6.49 (1H, s), 7.28 (2H, d, J= 8.3 Hz), 7.85 (2H, d, J= 8.3 Hz). Ή-NMR (300MHz, CDC1 3 ): δ 0.88 (3H, brt,] = 6.8 Hz), 0.95 (3H, brt, J = 7.5 Hz), 1.26 (10H, brs), 1.44 (2H, m), 1.68 (2H, q, J = 7.2 Hz), 2.41 (3H, s), 2.44 (2H, brdd, J = 7.5, 7.2 Hz), 3.75 (2H, s), 4.13 (2H, t, J = 6.8 Hz) , 6.49 (1H, s), 7.28 (2H, d, J = 8.3 Hz), 7.85 (2H, d, J = 8.3 Hz).
元素分析値: C26H35N305S3として計算値: C, 55.20; H, 6.24; , 7.43 ( ), 実 測値: C, 55.01; H, 6.13; , 7.52 (%) . Elemental analysis: Calculated as C 26 H 35 N 3 0 5 S 3: C, 55.20; H, 6.24;, 7.43 (), the real Found: C, 55.01; H, 6.13;, 7.52 (%).
実施例 -439 Example -439
¾-NMR (300MHz, CDC13) : 6 0.88 (3H, t, J= 7.2 Hz), 1.26 (18H, brs), 1.44 (2H, brquint, J= 7 Hz), 2.04 (2H, t, J= 7.0 Hz) , 2.41 (3H, s), 4.22 (2H, s), 6.47 (m, s), 7.25 (2H, d, J= 8.2 Hz), 7.32 (5H, s), 7.80 (2H, d, J= 8.2 Hz), 9.71 (1H, br). ¾-NMR (300MHz, CDC1 3 ): 6 0.88 (3H, t, J = 7.2 Hz), 1.26 (18H, brs), 1.44 (2H, brquint, J = 7 Hz), 2.04 (2H, t, J = 7.0 Hz), 2.41 (3H, s), 4.22 (2H, s), 6.47 (m, s), 7.25 (2H, d, J = 8.2 Hz), 7.32 (5H, s), 7.80 (2H, d, J = 8.2 Hz), 9.71 (1H, br).
元素分析値: C32H41N303S3として計算値: C, 62.81; H, 6.75; , 6.87; S, 15.72Elemental analysis: Calculated as C 32 H 41 N 3 0 3 S 3: C, 62.81; H, 6.75;, 6.87; S, 15.72
(%)、 実測値: C, 62.42; H, 6.64; N, 6.94; S, 15.76 (%) . (%), Found: C, 62.42; H, 6.64; N, 6.94; S, 15.76 (%).
実施例 4-440 Example 4-440
Ή-N R (300MHz, CDC13) : δ 2.42 (3H, s), 4.29 (2H, s), 6.90 (1H, s), 7.28 (2H, d, J= 8.3 Hz), 7.34 (5H, s), 7.37 (5H, s), 7.82 (2H, d, J= 8.3 Hz), 9.77 (1H, br). Ή-NR (300MHz, CDC1 3 ): δ 2.42 (3H, s), 4.29 (2H, s), 6.90 (1H, s), 7.28 (2H, d, J = 8.3 Hz), 7.34 (5H, s) , 7.37 (5H, s), 7.82 (2H, d, J = 8.3 Hz), 9.77 (1H, br).
元素分析値: C26H21N303S3として計算値: C, 60.09; H, 4.07; N, 8.09; S, 18.51Elemental analysis: Calculated for C 26 H 21 N 3 0 3 S 3 : C, 60.09; H, 4.07; N, 8.09; S, 18.51
(%) 、 実測値: C, 60.03; H, 4.11; N, 8.07; S, 18.59 (%) · (%), Found: C, 60.03; H, 4.11; N, 8.07; S, 18.59 (%) ·
実施例 4-441 Example 4-441
Ή一 NMR (300MHz, CDC13) : δ 0.72 (3Η, t, J= 7.2 Hz), 1.15 (2H, dq, J= 14.8, 7.2 Hz), 1.31 (2H, m), 2.42 (3H, s), 2.59 (2H, brdd, J= 7.7, 7.4 Hz), 4.17 (2H, s), 7.25 (2H, m), 7.26 (2H, d, J= 8.3 Hz) , 7.31 (5H, m), 7.39 (1H, m), 7.39 (2H, m), 7.83 (2H, d, J= 8.3 Hz), 9.67 (1H, br). Ή one NMR (300MHz, CDC1 3): δ 0.72 (3Η, t, J = 7.2 Hz), 1.15 (2H, dq, J = 14.8, 7.2 Hz), 1.31 (2H, m), 2.42 (3H, s) , 2.59 (2H, brdd, J = 7.7, 7.4 Hz), 4.17 (2H, s), 7.25 (2H, m), 7.26 (2H, d, J = 8.3 Hz), 7.31 (5H, m), 7.39 ( 1H, m), 7.39 (2H, m), 7.83 (2H, d, J = 8.3 Hz), 9.67 (1H, br).
元素分析値: C3。H29N303S3として計算値: C, 62.58; H, 5.08; N, 7.30; S, 16.71Elemental analysis: C 3. Calculated for H 29 N 3 0 3 S 3 : C, 62.58; H, 5.08; N, 7.30; S, 16.71
(%), 実測値: C, 62.53; H, 5.03; N, 7.33; S, 16.83 (%) . (%), Found: C, 62.53; H, 5.03; N, 7.33; S, 16.83 (%).
実施例 4-442 Example 4-442
'H-NMR (300MHz, CDC13) : δ 0.88 (3Η, t, J= 6.9 Hz), 1.26 (6H, brs), 1.30 (2H, m), 1.38 (2H, brquint, J= 7.2 Hz), ,1.68 (2H, brquint, J= 7.4 Hz), 2.44 (3H, s), 2.96 (2H, t, J= 7.5 Hz), 7.04 (4H, i), 7.17 (6H, m), 7.31 (2H, d, J= 8.3 Hz), 7.89 (2H, d, J= 8.3 Hz), 9.62 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.88 (3Η, t, J = 6.9 Hz), 1.26 (6H, brs), 1.30 (2H, m), 1.38 (2H, brquint, J = 7.2 Hz), , 1.68 (2H, brquint, J = 7.4 Hz), 2.44 (3H, s), 2.96 (2H, t, J = 7.5 Hz), 7.04 (4H, i), 7.17 (6H, m), 7.31 (2H, d, J = 8.3 Hz), 7.89 (2H, d, J = 8.3 Hz), 9.62 (1H, brs).
元素分析値: C33H35N303S3として計算値: C, 64.15; H, 5.71; N, 6.80; S, 15.57 (%) .実測値: C, 64.15; H, 5.71; N, 6.77; S, 15.72 ( ) . Elemental analysis: C 33 H 35 N 3 0 3 S 3 Calculated:. C, 64.15; H, 5.71; N, 6.80; S, 15.57 (%) Found: C, 64.15; H, 5.71 ; N, 6.77; S, 15.72 ().
実施例 4-443 Ή-NMR (300MHz, CDC13) : δ 2.42 (3H, s), 4.22 (2H, s), 6.99 (2H, dd, J= 7.5, 2.1 Hz), 7.04 (2H, dd, J= 8.0, 1.5 Hz), 7.16 (6H, m), 7.27 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.84 (2H, d, J= 8.3 Hz), 9.59 (1H, br). Example 4-443 Ή-NMR (300MHz, CDC1 3 ): δ 2.42 (3H, s), 4.22 (2H, s), 6.99 (2H, dd, J = 7.5, 2.1 Hz), 7.04 (2H, dd, J = 8.0, 1.5 Hz), 7.16 (6H, m), 7.27 (2H, d, J = 8.3 Hz), 7.32 (5H, s), 7.84 (2H, d, J = 8.3 Hz), 9.59 (1H, br).
元素分析値: C32H25N303S3 · 0.3H20として計算値: C, 63.93; H, 4.29; N, 6.99 (%)、 実測値: C, 63.90; H, 4.14; N, 7.38 (%) . Elemental analysis: C 32 H 25 N 3 0 3 S 3 · 0.3 H 2 0 Calculated: C, 63.93; H, 4.29; N, 6.99 (%), found: C, 63.90; H, 4.14; N , 7.38 (%).
実施例 4-444 Example 4-444
Ή-NMR (300MHz, CDC13) : δ 2.40 (3H, s), 3.35 (2H, s), 4.13 (2H, s), 6.55 (1H, brt, J= 1 Hz), 7.18-7.35 (12H, m), 7.79 (2H, d, J= 8.3 Hz), 9.72 (1H, br) . 元素分析値: C27H23N303S3として計算値: C, 60.76; H, 4.34; N, 7.87; S, 18.02 (%)、 実測値: C, 60.74; H, 4.46; N, 7.73; S, 18.18 (%) . Ή-NMR (300MHz, CDC1 3 ): δ 2.40 (3H, s), 3.35 (2H, s), 4.13 (2H, s), 6.55 (1H, brt, J = 1 Hz), 7.18-7.35 (12H, . m), 7.79 (2H, d, J = 8.3 Hz), 9.72 (1H, br) elemental analysis: calculated C 27 H 23 N 3 0 3 S 3: C, 60.76; H, 4.34; N, 7.87; S, 18.02 (%), Found: C, 60.74; H, 4.46; N, 7.73; S, 18.18 (%).
実施例 4-445 Example 4-445
融点 160-162°C 160-162 ° C
赤外吸収スペクトル(IR) (KBr中) v: 3260, 3162, 2961, 2928, 1738, 1657, 1615, 1559, 1495 cm— Infrared absorption spectrum (IR) (in KBr) v: 3260, 3162, 2961, 2928, 1738, 1657, 1615, 1559, 1495 cm—
¾-NMR (CDC13) :50.953 (6H, d, J=6.6 Hz), 1.72-1.88 (1H, m), 2.36 (3H, s), 2.41 (3H, s), 2.73 (2H, d, J=6.4 Hz), 3.50 (2H, s), 7.17-7.40 (7H, i), 7.86 (2H, d, J=8.4 Hz), 9.66 (IH, bs). ¾-NMR (CDC1 3): 50.953 (6H, d, J = 6.6 Hz), 1.72-1.88 (1H, m), 2.36 (3H, s), 2.41 (3H, s), 2.73 (2H, d, J = 6.4 Hz), 3.50 (2H, s), 7.17-7.40 (7H, i), 7.86 (2H, d, J = 8.4 Hz), 9.66 (IH, bs).
元素分析値: C25H27N303S3として計算値: C, 58.45; H, 5.30; , 8.18; S, 18.73 (%), 実測値: (:, 58.27; H, 5.27; N, 8.08; S, 18.66 (%) . Elemental analysis: Calculated as C 25 H 27 N 3 0 3 S 3: C, 58.45; H, 5.30;, 8.18; S, 18.73 (%), Found: (:, 58.27; H, 5.27; N, 8.08; S, 18.66 (%).
実施例 4-446 Example 4-446
酢酸ェチル -ジェチルェ一テルから再結晶し、 濃赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals.
融点: 147- 149で. Melting point: 147-149.
赤外吸収スぺクトル(IR) (KBr中) V: 3250, 2926, 1732, 1659, 1615, 1559, 1495 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3250, 2926, 1732, 1659, 1615, 1559, 1495 cm " 1 .
Ή-NMR (CDC13) : δ 0.820-0.942 (3Η, m), 1.20-1.38 (4H, m), 1.48-1.70 (2H, m), 2.42 (3H, s), 2.85 (2H, t, J-7.2 Hz), 3.50 (2H, s), 7.15-7.42 (7H, m), 7.86 (2H, d, J=8.4 Hz), 9.65 (IH, bs) . Ή-NMR (CDC1 3): δ 0.820-0.942 (3Η, m), 1.20-1.38 (4H, m), 1.48-1.70 (2H, m), 2.42 (3H, s), 2.85 (2H, t, J -7.2 Hz), 3.50 (2H, s), 7.15-7.42 (7H, m), 7.86 (2H, d, J = 8.4 Hz), 9.65 (IH, bs).
元素分析値: C26H29N303S3として計算値: C, 59.18; H, 5.54; N, 7.96; S, 18.23 (%)、 実測値: C, 59.02; H, 5.47; N, 7.89; S, 18.37 (%) · 実施例 4-448 Elemental analysis: C 26 H 29 N 3 0 3 S 3 Calculated: C, 59.18; H, 5.54 ; N, 7.96; S, 18.23 (%), Found: C, 59.02; H, 5.47 ; N, 7.89; S, 18.37 (%) Example 4-448
Ή-NMR (300MHz, CDC13) : δ 0.92 (3H, t, J- 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.05 (2H, brt, J= 7.1 Hz), 2.41 (3H, s), 4.51 (2H, s), 4.76 (2H, s), 4.82 (2H, s), 5.78 (2H, s), 6.44 (1H, brt, J= 0.5 Hz), 7.24 (2H, m), 7.28 (2H, brd, J= 8.3 Hz), 7.29 (1H, m), 7.33 (5H, m) , 7.40 (2H, brd, J= 6.8 Hz), 7.94 (2H, d, J= 8.3 Hz). Ή-NMR (300MHz, CDC1 3 ): δ 0.92 (3H, t, J- 7.2 Hz), 1.34 (2H, m), 1.44 (2H, m), 2.05 (2H, brt, J = 7.1 Hz), 2.41 (3H, s), 4.51 (2H, s), 4.76 (2H, s), 4.82 (2H, s), 5.78 (2H, s), 6.44 (1H, brt, J = 0.5 Hz), 7.24 (2H, s) m), 7.28 (2H, brd, J = 8.3 Hz), 7.29 (1H, m), 7.33 (5H, m), 7.40 (2H, brd, J = 6.8 Hz), 7.94 (2H, d, J = 8.3 Hz) Hz).
元素分析値: C33 05S3として計算値: C, 60.99; H, 5.43; N, 6.47; S, 14.80Elemental analysis: C 33 0 5 S 3 Calculated: C, 60.99; H, 5.43 ; N, 6.47; S, 14.80
(%), 実測値: C, 60.85; H, 5.45; N, 6.47; S, 14.53 (%) . (%), Found: C, 60.85; H, 5.45; N, 6.47; S, 14.53 (%).
実施例 4-449 Example 4-449
Ή-NMR (300MHz, CDC13) : d 0.90 (3H, t, J= 6.8 Hz), 1.30 (4H, m), 1.47 (2H, brquint, J= 7.2 Hz), 2.10 (2H, t, J= 7.5 Hz), 2.23 (3H, s),, 2.42 (3H, s), 4.51 (2H, s), 4.78 (2H, s), 4.83 (2H, s), 5.75 (2H, s), 7.22-7.31 (6H, m), 7.33 (4H, m), 7.42 (2H,. brd, J= 7.2 Hz), 7.95 (2H, d, J= 8.3 Hz). Ή-NMR (300MHz, CDC1 3 ): d 0.90 (3H, t, J = 6.8 Hz), 1.30 (4H, m), 1.47 (2H, brquint, J = 7.2 Hz), 2.10 (2H, t, J = 7.5 Hz), 2.23 (3H, s), 2.42 (3H, s), 4.51 (2H, s), 4.78 (2H, s), 4.83 (2H, s), 5.75 (2H, s), 7.22-7.31 (6H, m), 7.33 (4H, m), 7.42 (2H, .brd, J = 7.2 Hz), 7.95 (2H, d, J = 8.3 Hz).
元素分析値: C35H39N305S3として計算値: C, 62.01; H, 5.80; N, 6.20; S, 14.19Elemental analysis: C 35 H 39 N 3 0 5 S 3 Calculated: C, 62.01; H, 5.80 ; N, 6.20; S, 14.19
(%) .実測値: C, 61.85; H, 5.90; N, 6.26; S, 14.37 (%) . Found: C, 61.85; H, 5.90; N, 6.26; S, 14.37 (%).
実施例 4-450 Example 4-450
^-NMR (CDC13, 300 MHz): δ 0.86 (6Η, d, J= 6.8 Hz), 0.90 (3H, brt, J= 7 Hz), 1.30 (4H, m), 1.47 (2H, brquint, J= 7.2 Hz), 1.70 (1H, brsept, J= 6.8 Hz), 2.11 (2H, brt, J= 7.7 Hz), 2.29 (3H, s), 2.40 (3H, s), 2.70 (2H, d, J= 6.4 Hz), 3.70 (3H, s), 7.25 (2H, d, J= 8.3 Hz), 7.92. (2H, d, J- 8.3 Hz). 元素分析値: C24H33N303S3として計算値: C, 56.77; H, 6..55; N, 8.28; S, 18.95 ^ -NMR (CDC1 3, 300 MHz ): δ 0.86 (6Η, d, J = 6.8 Hz), 0.90 (3H, brt, J = 7 Hz), 1.30 (4H, m), 1.47 (2H, brquint, J = 7.2 Hz), 1.70 (1H, brsept, J = 6.8 Hz), 2.11 (2H, brt, J = 7.7 Hz), 2.29 (3H, s), 2.40 (3H, s), 2.70 (2H, d, J = 6.4 Hz), 3.70 (3H , s), 7.25 (2H, d, J = 8.3 Hz), 7.92 (2H, d, J- 8.3 Hz) elemental analysis:.. C 24 H 33 N 3 0 3 S Calculated as 3 : C, 56.77; H, 6.55; N, 8.28; S, 18.95
(%)、 実測値: C, 56.65; H, 6.36; N, 8.34; S, 19.02 (%) . (%), Found: C, 56.65; H, 6.36; N, 8.34; S, 19.02 (%).
実施例 4-451 Example 4-451
Ή-NMR (200MHz, CDCI3) : δ 1.26 (3Η, t, J= 7.2 Hz), 1.80 (3H, brq, J= 1.2 Hz), 2.29 (3H, brq, J= 0.8 Hz), 2.43 (3H, s), 2.49 (2H, t, J= 7.4 Hz), 3.05 (2H, t, J= 7.3 Hz), 3.51 (3H, s), 4.15 (2H, q, J= 7.2 Hz), 5.60 (2H, s), 7.28 (2H, d, J= 8.4 Hz), 7.89 (2H, d, J= 8.4 Hz). Ή-NMR (200 MHz, CDCI 3 ): δ 1.26 (3Η, t, J = 7.2 Hz), 1.80 (3H, brq, J = 1.2 Hz), 2.29 (3H, brq, J = 0.8 Hz), 2.43 (3H , s), 2.49 (2H, t, J = 7.4 Hz), 3.05 (2H, t, J = 7.3 Hz), 3.51 (3H, s), 4.15 (2H, q, J = 7.2 Hz), 5.60 (2H , s), 7.28 (2H, d, J = 8.4 Hz), 7.89 (2H, d, J = 8.4 Hz).
元素分析値: C22H27N306S3として計算値: C, 50.27; H, 5.18; N, 7.99; S, 18.30 (%) 、 実測値: C, 50.32; H, 5.22; N, 8.03; S, 18.31 (%) . 融点. 89.5- 92.0 Elemental analysis: Calculated as C 22 H 27 N 3 0 6 S 3: C, 50.27; H, 5.18; N, 7.99; S, 18.30 (%), Found: C, 50.32; H, 5.22 ; N, 8.03; S, 18.31 (%). Melting point.89.5- 92.0
実施例 4-452 Example 4-452
'H-NMR (200MHz, CDC13) : <5 1.09 (3H, t, J= 7.5 Hz), 2.14 (2H, q, J= 7.5 Hz),'H-NMR (200MHz, CDC1 3): <5 1.09 (3H, t, J = 7.5 Hz), 2.14 (2H, q, J = 7.5 Hz),
2.30 (3H, s), 2.32 (3H, s), 2.42 (3H, s), 3.50 (3H, s), 5.61 (2H, s), 7.27 (2H, d, J= 8.2 Hz), 7.90 (2H, d, = 8.4 Hz). , 元素分析値: C19H23N304S3として計算値: C, 50.31; H, 5.11; N, 9.26; S, 21.212.30 (3H, s), 2.32 (3H, s), 2.42 (3H, s), 3.50 (3H, s), 5.61 (2H, s), 7.27 (2H, d, J = 8.2 Hz), 7.90 (2H , d, = 8.4 Hz), elemental analysis: calculated C 19 H 23 N 3 0 4 S 3:. C, 50.31; H, 5.11; N, 9.26; S, 21.21
(%) 、 実測値: C, 50.16; H, 5.13; N, 9.27; S, 21.25 ( ) . (%), Found: C, 50.16; H, 5.13; N, 9.27; S, 21.25 ().
融点. 142.5- 143.0°C 142.5- 143.0 ° C
実施例 4-453 Example 4-453
Ή— NMR (200MHz, CDC13) : δ 1.80 (3H, q, J= 1.0 Hz) , 2.30 (3H, q, J= 1.0 Hz), 2.32 (3H, s), 2.42 (3H, s), 3.71 (3H, s), 7.28 (2H, d, J= 8.0 Hz), 7.90 (2H, d, J= 8.4 Hz). Ή- NMR (200MHz, CDC1 3) : δ 1.80 (3H, q, J = 1.0 Hz), 2.30 (3H, q, J = 1.0 Hz), 2.32 (3H, s), 2.42 (3H, s), 3.71 (3H, s), 7.28 (2H, d, J = 8.0 Hz), 7.90 (2H, d, J = 8.4 Hz).
元素分析値: C17Hl9N303S3として計算値: C, 49.86; H, 4.68; , 10.26; S, 23.49 ( ) 、 実測値: C, 49.88; H, 4.66; N, 9.98; S, 23.52 ( ) . Elemental analysis: Calculated as C 17 H l9 N 3 0 3 S 3: C, 49.86; H, 4.68;, 10.26; S, 23.49 (), Found: C, 49.88; H, 4.66 ; N, 9.98; S, 23.52 ().
融点. 203.2- 203.7°C Melting point. 203.2-203.7 ° C
実施例 4-454 Example 4-454
Ή-NMR (200MHz, CDC13) : δ 1.79 (3H, q, J= 1.2 Hz), 2.30 (3H, q, J= 1.2 Hz), Ή-NMR (200MHz, CDC1 3 ): δ 1.79 (3H, q, J = 1.2 Hz), 2.30 (3H, q, J = 1.2 Hz),
2.31 (3H, s), 2.42 (3H, s), 3.50 (3H, s), 5.61 (2H, s), 7.27 (2H, d, J= 8.0 Hz), 7.90 (2H, d, J= 8.4 Hz). 2.31 (3H, s), 2.42 (3H, s), 3.50 (3H, s), 5.61 (2H, s), 7.27 (2H, d, J = 8.0 Hz), 7.90 (2H, d, J = 8.4 Hz) ).
元素分析値: C18H21N304S3として計算値: C, 49.18; H, 4.82; N, 9.56; S, 21.88 (%) 、 実測値: C, 49.19; H, 5.04; N, 9.52; S, 21.95 (%) . Elemental analysis: C 18 H 21 N 3 0 4 S 3 Calculated: C, 49.18; H, 4.82 ; N, 9.56; S, 21.88 (%), Found: C, 49.19; H, 5.04 ; N, 9.52; S, 21.95 (%).
融点. 153.0-153.5t: Melting point.153.0-153.5t:
実施例 4-455 Example 4-455
Ή-NMR (200MHz, CDC13) : δ 1.75 (3H, q, J= 1.2 Hz), 2.28 (3H, q, J= 1.2 Hz), 2.39 (3H, s), 3.52 (3H, s), 4.31 (2H, s), 5. '62 (2H, s), 7.21 (2H, d, J= 8.0 Hz), 7.50 (2H, t, J= 7.5Hz), 7.64 (1H, t, J= 7.4 Hz), 7.89 (4H, m) . Ή-NMR (200MHz, CDC1 3 ): δ 1.75 (3H, q, J = 1.2 Hz), 2.28 (3H, q, J = 1.2 Hz), 2.39 (3H, s), 3.52 (3H, s), 4.31 (2H, s), 5.'62 (2H, s), 7.21 (2H, d, J = 8.0 Hz), 7.50 (2H, t, J = 7.5 Hz), 7.64 (1H, t, J = 7.4 Hz) ), 7.89 (4H, m).
元素分析値: C25H25N305S3 · 0.5 0として計算値: C, 54.33; H, 4.74; N, 7.60; S, 17.41 (%) 、 実測値: C, 54.39; H, 4.59; N, 7.58; S, 17.40 (%) . Elemental analysis: C 25 H 25 N 3 0 5 S 3 · 0.50 Calculated: C, 54.33; H, 4.74; N, 7.60; S, 17.41 (%), found: C, 54.39; H, 4.59 ; N, 7.58; S, 17.40 (%).
融点. 186.5-187.0°C 実施例 4-456 Melting point. 186.5-187.0 ° C Example 4-456
N- (5, 6-ジメチル- 3-ォキリ- 1- [2- (3-チェニル)ェチル] -8- { [2- (3-チェニル)ェチ ル]スルファニル }-2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデ トリフエニルホスフィン(565 mg, 1.57腿01)の1^ (5 ml)の溶液に 0°Cで攪拌 しながら DIAD (0.319 ml, 1.57匪 ol)を滴下し、 30分間攪拌した。 反応液に実施 例 3-4で得た化合物 (30 mg, 0.797 mmol)および 2- (3 -チェニル)ェタノ一ル. (123 mg, 0.944 匪 ol)を加え、 反応液を室温に戻し、 27 時間攪拌した。 この間反応液' にトリフエニルホスフィン(108 mg, 0.400 mmol)および DIAD (83 mg, 0.400 mmol) を加えた。反応液を酢酸ェチルで希釈し、 2 炭酸水素ナトリゥム水溶液、 0.1N塩 酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣を シリカゲル(10g)のクロマトグラフィー (酢酸ェチル一へキサン、 1:9-4:6) で精 製し、 化合物 (85 mg, 0.141 匪 ol)を赤紫色固形物として得た。 N- (5,6-Dimethyl-3-oxo-1--1- [2- (3-Chenyl) ethyl] -8-{[2- (3-Chenyl) ethyl] sulfanyl} -2,3-dihydro- While stirring at 0 ° C DIAD ( 0.319 ml, 1.57 marl ol) was added dropwise, and the mixture was stirred for 30 minutes.The compound (30 mg, 0.797 mmol) obtained in Example 3-4 and 2- (3-Chenyl) ethanol (123 mg , 0.944 ol) and the reaction solution was cooled to room temperature and stirred for 27 hours, during which time triphenylphosphine (108 mg, 0.400 mmol) and DIAD (83 mg, 0.400 mmol) were added. The residue was diluted with ethyl acetate, washed with aqueous sodium hydrogen carbonate, 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate and concentrated.The residue was chromatographed on silica gel (10 g) (ethyl acetate). Hexane, to 1: 9-4: 6) in the paper seminal to give compound (85 mg, and 0.141 negation ol) as a purple-red solid.
-題 R (200MHz, CDC13) : δ 1.79 (3Η, q, J= 1.2 Hz), 2.28 (3H, q, J= 1.0 Hz), 2.41 (3H, s), 2.79 (2H, t, J= 7.3 Hz), 3.05 (2H, t, J= 7.3 Hz), 3.18 (2H, brt, J= 8.5 Hz), 4.42 (2H, brt, J= 8.1 Hz), 6.83 (2H, d, J= 4.0 Hz), 7.17-7.28 (2H, m), 7.92 (2H, d, J= 8.4 Hz). - title R (200MHz, CDC1 3): δ 1.79 (3Η, q, J = 1.2 Hz), 2.28 (3H, q, J = 1.0 Hz), 2.41 (3H, s), 2.79 (2H, t, J = 7.3 Hz), 3.05 (2H, t, J = 7.3 Hz), 3.18 (2H, brt, J = 8.5 Hz), 4.42 (2H, brt, J = 8.1 Hz), 6.83 (2H, d, J = 4.0 Hz) ), 7.17-7.28 (2H, m), 7.92 (2H, d, J = 8.4 Hz).
元素分析値: C27H27N303S5として計算値: C, 53.88; H, 4.52; N, 6.98; S, 26.64 (%), 実測値: C, 54.13; H, 4.45; N, 7.01; S, 26.71 (%) . Elemental analysis: C 27 H 27 N 3 0 3 S 5 Calculated: C, 53.88; H, 4.52 ; N, 6.98; S, 26.64 (%), Found: C, 54.13; H, 4.45 ; N, 7.01; S, 26.71 (%).
実施例 4-457 Example 4-457
N-(5, 6 -ジメチル -3-ォキソ -Ϊ - [3- (3-ピリジニル)プロピル]- 8- {[3- (3-ピリジニ ル)プロピル]スルファニル }_2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1,4]チアジン -卜ィリデン )-4_メチルベンゼンスルホンアミド N- (5,6-Dimethyl-3-oxo-Ϊ- [3- (3-pyridinyl) propyl] -8-{[3- (3-pyridinyl) propyl] sulfanyl} _2,3-dihydro-1H- Imidazo [5, 1-c] [1,4] thiazine-triylidene) -4_methylbenzenesulfonamide
実施例 3 - 4で得た化合物 (163 mg, 0.427 mmol)、 3 -(3-ピリジル) -卜プロパノ ール (90 mg, 0.641 麵 ol) およびトリフエニルホスフィン(231 mg, 0.855腿 ol) の THF (3.3 ml)懸濁液に室温で攪拌しながら DEAD (0.142 ml, 0.855 mmol)を滴 下し、反応液を室温で 19時間攪拌した。反応液にトリフエニルホスフィン α 16 mg, 0.427 顧 ol)および DEAD (0.071 ml, 0.427 腿 ol)を加え、 反応液を室温で 19時 間攪拌した。 酢酸ェチル (25 ml)を加えて希釈し、 2%炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄し、 乾燥、 濃縮した。 残渣をシリカゲル(10 g)のクロ マトグラフィ一 [酢酸ェチル—へキサン (1:1) 、 酢酸ェチルおよびメタノール— 酢酸ェチル(1:19 および 1:9)]に付した。 メタノール—酢酸ェチルの画分を濃縮 乾固後酢酸ェチルから結晶化して標題化合物 (115 mg, 0.186腿 ol)を紫色粉末と して得た。Of the compound (163 mg, 0.427 mmol) obtained in Example 3-4, 3- (3-pyridyl) -topropanol (90 mg, 0.641 mol) and triphenylphosphine (231 mg, 0.855 mol). DEAD (0.142 ml, 0.855 mmol) was added dropwise to the THF (3.3 ml) suspension while stirring at room temperature, and the reaction solution was stirred at room temperature for 19 hours. To the reaction solution, triphenylphosphine α (16 mg, 0.427) and DEAD (0.071 ml, 0.427) were added, and the reaction solution was stirred at room temperature for 19 hours. Dilute with ethyl acetate (25 ml), add 2% aqueous sodium bicarbonate, The extract was washed with water and saturated saline, dried and concentrated. The residue was chromatographed on silica gel (10 g) [ethyl acetate-hexane (1: 1), ethyl acetate and methanol-ethyl acetate (1:19 and 1: 9)]. The methanol-ethyl acetate fraction was concentrated and dried, and then crystallized from ethyl acetate to give the title compound (115 mg, 0.186 mol) as a purple powder.
— NMR (300MHz, CDC13+TFA) : <5 1.78 (3H, q, J= 1.2 Hz), 1.90 (2H, quint, J= 7.2 Hz), 2.28 (3H, q, J= 1.2 Hz), 2.29 (2H, m), 2.35 (3H, s), 2.73 (2H, t, J- 7.8 Hz), 2.82 (2H, t, J= 6.9 Hz), 2.99 (2H, t, J= 7.8 Hz), 4.29 (2H, t, J= 7.6 Hz), 7.27 (2H, d, J= 8.4 Hz), 7.77 (1H, t, J= 5.7 Hz), 7.80 (1H, t, J= 5.1 Hz), 7.88 (2H, d, J= 8.1 Hz), 8.02 (1H, d, J= 8.1 Hz), 8.31 (1H, d, J= 7.5 Hz), 8.72 (3H, m), 8.81 (1H, brs). - NMR (300MHz, CDC1 3 + TFA): <5 1.78 (3H, q, J = 1.2 Hz), 1.90 (2H, quint, J = 7.2 Hz), 2.28 (3H, q, J = 1.2 Hz), 2.29 (2H, m), 2.35 (3H, s), 2.73 (2H, t, J- 7.8 Hz), 2.82 (2H, t, J = 6.9 Hz), 2.99 (2H, t, J = 7.8 Hz), 4.29 (2H, t, J = 7.6 Hz), 7.27 (2H, d, J = 8.4 Hz), 7.77 (1H, t, J = 5.7 Hz), 7.80 (1H, t, J = 5.1 Hz), 7.88 (2H , D, J = 8.1 Hz), 8.02 (1H, d, J = 8.1 Hz), 8.31 (1H, d, J = 7.5 Hz), 8.72 (3H, m), 8.81 (1H, brs).
元素分析値: C31H33N503S3 · 0·3 0として計算値: C, 59.55; Η, 5.42; Ν, 11.20; S, 15.39 (%) 、 実測値: C, 59.38; Η, 5.47; Ν, 11.00; S, 15.41 (%) . Elemental analysis: C 31 H 33 N 5 0 3 S 3 · 0 · 30 Calculated: C, 59.55; Η, 5.42; Ν, 11.20; S, 15.39 (%), found: C, 59.38; Η , 5.47; Ν, 11.00; S, 15.41 (%).
融点. 150.5-152. O 150.5-152. O
実施例 4-458 Example 4-458
N -(5, 6-ジメチル -3-ォキソ -2- [3- (4-ピリジニル)プロピル]- 8- {[3- (4-ピリジニ ル)プロピル]スルファニル卜 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン 実施例 3-4で得た化合物 (219 mg, 0.574腿 ol)、 3 - (4-ピリジル) -卜プロパノ ール (121 mg, 0.861 mmol) およびト リフエニルホスフィン(310 mg, 1.15匪 ol) の THF (4. ml)懸濁液に室温で攪拌しながら DEAD (0.190 ml, 1.15 mmol)を滴下 し、 反応液を室温で 19時間攪拌した。反応液にトリフエニルホスフィン(155 mg, 0.574 mmol)および DEAD (0.095 ml, 0.574 mmol)を加え、 反応液を室温で 4時間 攪拌した。 酢酸ェチル (30 ml)を加えて希釈し、 2%炭酸ナトリウム水溶液、 2%炭酸 水素ナトリウム水溶液および飽和食塩水で洗浄し、 乾燥、 濃縮した。 残渣をシリ 力ゲル(10 g)のクロマトグラフィー [酢酸ェチルーへキサン (1:1)、 酢酸ェチルお よびメ夕ノ一ルー酢酸ェチル(1:19 および 1:9)]に付した。 メタノール一酢酸ェ チルの画分を濃縮乾固後メタノールから結晶化して標題化合物 (178 mg, 0.287 mmo 1 )を紫色粉末として得た。 Ή-NMR (300MHz, CDC13+TFA) : δ 1.77 (3H, s), 1.93 (2H, quint, J= 6.6 Hz), 2.27 (3H, s), 2.36 (2H, m), 2.37 (3H, s), 2.78 (2H, m), 2.82 (2H, t, J= 7.2 Hz), 3.05 (2H, t, J= 6.9 Hz), 4.33 (2H, t, J= 7.5 Hz), 7.29 (2H, d, J= 8.1 Hz), 7.56 (2H, brs), 7.81 (2H, d, J= 5.4 Hz), 7.89 (2H, d, J= 8.1 Hz), 8.76 (4H, brd, J= 5.4 Hz), 8.31 (1H, d, J= 7.5 Hz), 8.72 (3H, m), 8.81 (1H, brs). 元素分析値: C31 N503S3.0.3¾0として計算値: C, 59.55; H, 5.42; N, 11.20; S, 15.39 (%) 、 実測値: C, 59.63; H, 5.39; N, 11.09; S, 15.60 (%) . N- (5,6-Dimethyl-3-oxo-2- [3- (4-pyridinyl) propyl] -8-{[3- (4-pyridinyl) propyl] sulfanyl 2,3-dihydro-1H- Imidazo [5,1-c] [1,4] thiazine The compound obtained in Example 3-4 (219 mg, 0.574 tmol), 3- (4-pyridyl) -tripropanol (121 mg, 0.861 mmol) ) And triphenylphosphine (310 mg, 1.15 bandol) in THF (4. ml) were added dropwise with stirring at room temperature while DEAD (0.190 ml, 1.15 mmol) was added dropwise, and the reaction solution was stirred at room temperature for 19 hours. Triphenylphosphine (155 mg, 0.574 mmol) and DEAD (0.095 ml, 0.574 mmol) were added to the reaction solution, and the reaction solution was stirred at room temperature for 4 hours, and diluted with ethyl acetate (30 ml). The residue was washed with a 2% aqueous sodium carbonate solution, a 2% aqueous sodium hydrogen carbonate solution and saturated saline, dried and concentrated.The residue was chromatographed on silica gel (10 g) [ethyl acetate-hexane (1: 1), ethyl acetate and methyl ethyl acetate (1:19 and 1: 9)] The fraction of methanol-ethyl acetate was concentrated to dryness and then crystallized from methanol to give the title compound (178). mg, 0.287 mmo 1) as a purple powder. Ή-NMR (300MHz, CDC1 3 + TFA): δ 1.77 (3H, s), 1.93 (2H, quint, J = 6.6 Hz), 2.27 (3H, s), 2.36 (2H, m), 2.37 (3H, s), 2.78 (2H, m), 2.82 (2H, t, J = 7.2 Hz), 3.05 (2H, t, J = 6.9 Hz), 4.33 (2H, t, J = 7.5 Hz), 7.29 (2H, d, J = 8.1 Hz), 7.56 (2H, brs), 7.81 (2H, d, J = 5.4 Hz), 7.89 (2H, d, J = 8.1 Hz), 8.76 (4H, brd, J = 5.4 Hz) , 8.31 (1H, d, J = 7.5 Hz), 8.72 (3H, m), 8.81 (1H, brs) elemental analysis: C 31 N 5 0 3 S 3 .0.3¾0 calculated: C, 59.55;. H, 5.42; N, 11.20; S, 15.39 (%), found: C, 59.63; H, 5.39; N, 11.09; S, 15.60 (%).
融点. 143.0- 143.5で Melting point.
実施例 4-459 Example 4-459
酢酸ェチル-ジイソプロピルェ一テルで再結晶し、 暗赤色結晶を得た。 Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals.
融点 178- 180 . Melting point 178-180.
IR (KBr) V: 2957, 2926, 2859, 1738, 1713, 1644, 1624, 1609, 1561, 1535, 1507 cur1. IR (KBr) V: 2957, 2926, 2859, 1738, 1713, 1644, 1624, 1609, 1561, 1535, 1507 cur 1 .
Ή -丽 R (CDC13) : 50.932 (3H, t, J=6.6 Hz), 1.34-1.50 (2H, m), 1.66-1.80 (2H, Hi), 1.84 (3H, s), 2.23 (3H, s), 2.39 (6H, s), 3.01 (2H, t, J=7.8 Hz), 7.20 (2H, d, J=8.4 Hz), 7.25 (2H, d, J=8.4 Hz), 7.80 (2H, d, J=8.0Hz), 8.02 (2H, d, J-8.0 Hz). E -丽R (CDC1 3): 50.932 ( 3H, t, J = 6.6 Hz), 1.34-1.50 (2H, m), 1.66-1.80 (2H, Hi), 1.84 (3H, s), 2.23 (3H, s), 2.39 (6H, s), 3.01 (2H, t, J = 7.8 Hz), 7.20 (2H, d, J = 8.4 Hz), 7.25 (2H, d, J = 8.4 Hz), 7.80 (2H, d, J = 8.0Hz), 8.02 (2H, d, J-8.0 Hz).
元素分析値: C29H3IN303S2 · 0.3¾0として計算値 (%) : C, 64.61; H, 5.91; N, 7.79; S, 11.90. 実測値 (¾) : C, 64.62; H, 5.88; N, 7.67; S, 12.09. Elemental analysis: C 29 H 3 I N 3 0 3 S 2 · Calculated as 0.3¾0 (%): C, 64.61; H, 5.91; N, 7.79; S, 11.90. Observed (¾): C, 64.62; H, 5.88; N, 7.67; S, 12.09.
実施例 4-460 Example 4-460
8- (ィソペンチルスルファニル) - 6-メチル - 1- { [ (4-メチルフェニル)スルホニル] ィミノ卜 3-ォキソ -2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1- c] [1, 4]チアジン- 5 -力ルボン 酸ェチル 8- (isopentylsulfanyl) -6-methyl-1-([(4-methylphenyl) sulfonyl] iminoto 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] Thiazine-5-potassium rubonate
実施例 3-21 で得た化合物(57 mg, 0.130 mmol)および炭酸カリウム (27 mg, 0.195 mmol) の F (1.0 ml) の懸濁液に 60 で攪拌しながらヨウ化イソペンチ ル (0.021 ml, 0.156 mmol)を加え、 混合物を 40分間攪拌した。 反応液に 1N塩 酸 (0.26 ml)およびメタノール一水(1:1, 3 ml)を加え、室温で 30分間攪拌した。 析出物を濾取し、メタノール一水 (1:1)およびメタノールで洗浄、乾燥して標題 化合物 (53 mg, 0.104腿 ol)を赤紫色粉末として得た。 Ή-N R (200MHz, CDC13) : δ 0.93 (6H, d, J= 6.4 Hz), 1.33 (3H, t, J= 7.1 Hz), 1.56 (2H, brq, J= 7.5 Hz), 1.67 (1H, sixtet, J= 6.2 Hz), 1.92 (3H, s), 2.43 (3H, s), 2.96 (2H, t, J= 7.7 Hz), 4.33 (2H, q, J= 7.2 Hz), 7.30 (2H, d, J二 8.6 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.69 (1H, br). To a suspension of the compound obtained in Example 3-21 (57 mg, 0.130 mmol) and potassium carbonate (27 mg, 0.195 mmol) in F (1.0 ml) was stirred at 60 with isopentyl iodide (0.021 ml, 0.156 mmol) was added and the mixture was stirred for 40 minutes. 1N Hydrochloric acid (0.26 ml) and methanol / water (1: 1, 3 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (53 mg, 0.104 tmol) as a red-purple powder. Ή-NR (200MHz, CDC1 3 ): δ 0.93 (6H, d, J = 6.4 Hz), 1.33 (3H, t, J = 7.1 Hz), 1.56 (2H, brq, J = 7.5 Hz), 1.67 (1H , sixtet, J = 6.2 Hz), 1.92 (3H, s), 2.43 (3H, s), 2.96 (2H, t, J = 7.7 Hz), 4.33 (2H, q, J = 7.2 Hz), 7.30 (2H , d, J2 8.6 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.69 (1H, br).
元素分析値: C22H27N305S3として計算値: C, 51.84; H, 5.34; N, 8.24; S, 18.87 (%) 、 実測値: C, 51.60; H, 5.15; N, 8.13; S, 18.84 (%) . Elemental analysis: C 22 H 27 N 3 0 5 S 3 Calculated: C, 51.84; H, 5.34 ; N, 8.24; S, 18.87 (%), Found: C, 51.60; H, 5.15 ; N, 8.13; S, 18.84 (%).
融点: 150.0-150.5°C Melting point: 150.0-150.5 ° C
実施例 4-461 Example 4-461
6 -メチル-卜 {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -8- [(2-フエ二 ルプロピル)スルファニル] -2, 3 -ジヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン- 5- カルボン酸ェチル 6-methyl-{{((4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(2-phenylpropyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [ 1,4] thiazine-5-carboxylate
実施例 3-21で得た化合物(0.59 g, 1.2删 ol)および炭酸力リゥム (0.25 g, 1.8 匪 ol) の DMF (10 ml) の懸濁液に 60 で攪拌しながらヨウ化 2-フエニルプロピ ル(0.208 ml, 1.2 mmol)を加え、 混合物を 4時間攪拌した。 この間反応液にヨウ ィ匕 2 -フエニルプロピル(0.021 ml, 0.12 mmol)を加えた。 反応液に 1N塩酸 (2.4 ml)およびおよびメタノール—水(1:1, 10 ml)を加え、 室温で 60分間攪拌した。 析出物を濾取し、メタノール—水 (1:1)およびメタノールで洗浄、乾燥して標題 化合物 (570 mg, 1.02 mmol)を紫色粉末として得た。 To a suspension of the compound obtained in Example 3-21 (0.59 g, 1.2 mol) and carbonated lime (0.25 g, 1.8 mol ol) in DMF (10 ml) was added 2-phenylphenyl iodide while stirring at 60. (0.208 ml, 1.2 mmol) was added and the mixture was stirred for 4 hours. During this time, iodoani 2-phenylpropyl (0.021 ml, 0.12 mmol) was added to the reaction solution. 1N Hydrochloric acid (2.4 ml) and methanol-water (1: 1, 10 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (570 mg, 1.02 mmol) as a purple powder.
腿 (200MHz, 重クロ口ホルム): 6 1.33 (3Η, t, J= 7.2 Hz), 1.37 (3H, d, J= 6.4 Hz), 1.91 (3H, s), 2.43 (3H, s), 3.03 (1H, m), 3.11 (1H, m), 3.24 (1H, quint, J= 6.2 Hz), 4.33 (2H, q, J= 7.1 Hz), 7.19 (2H, m), 7.29 (5H, m), 7.80 (2H, d, J= 8.4 Hz), 9.67 (1H, brs). Thigh (200MHz, double-mouthed holm): 6 1.33 (3Η, t, J = 7.2 Hz), 1.37 (3H, d, J = 6.4 Hz), 1.91 (3H, s), 2.43 (3H, s), 3.03 (1H, m), 3.11 (1H, m), 3.24 (1H, quint, J = 6.2 Hz), 4.33 (2H, q, J = 7.1 Hz), 7.19 (2H, m), 7.29 (5H, m) , 7.80 (2H, d, J = 8.4 Hz), 9.67 (1H, brs).
元素分析値: C26H27N305S3 として計算値; C, 55.99; H, 4.88; N, 7.53; S, 17.25 (%),実測値; C, 56.16; H, 5.08; N, 7.65; S, 16.95 (%) . 融点 144.5-145.5T: 実施例 4-462 Elemental analysis: Calculated as C 26 H 27 N 3 0 5 S 3; C, 55.99; H, 4.88; N, 7.53; S, 17.25 (%), Found; C, 56.16; H, 5.08 ; N, 7.65; S, 16.95 (%). Melting point: 144.5-145.5T: Example 4-462
8 -(イソペンチルスルファ二ル)- 6-メチル -1-{[(4_メチルフエニル)スルホニル] イミノ}-3-ォキソ - 2,3-ジヒドロ- -ィミダゾ[5,卜(;] [1, 4]チアジン- 5-カルボン 酸 8- (Isopentylsulfanyl) -6-methyl-1-{[(4_methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-imidazo [5, tri (;) [1 , 4] thiazine-5-carboxylic acid
実施例 1 - 48 で得た化合物(1.10 g, 1.88 mmol)の THF -メタノール (1:1, 20 ml) の懸濁液に室温で攪拌しながら水酸化カリウム(0.37 g, 5.64腿 ol)を加え、 反応 液を室温で 40分間攪拌した。 反応液に 1N塩酸 (5.64 ml)、 水(10 ml)を加えて 酢酸ェチルで抽出した。 有機層を 2%および飽和の食塩水で洗浄後、 硫酸ナトリウ ムで乾燥し、 濃縮して黄色油状物 (0.99 g)を得た。 この黄色油状物 (0.99 g)お よび炭酸カリウム (0.39 g, 2.82匪 ol) の DMF (15 ml) の懸濁液に 60°Cで攪拌 しながらヨウ化イソペンチル (0.30ml, 2.26mmol)を加え、 混合物を 2.5時間攪 拌した。 反応液を同様に抽出精製し、 黄褐色油状物 (1.06 g)を得た。 Compound (1.10 g, 1.88 mmol) obtained in Example 1-48 in THF-methanol (1: 1, 20 ml) To the suspension was added potassium hydroxide (0.37 g, 5.64 liters) while stirring at room temperature, and the reaction solution was stirred at room temperature for 40 minutes. 1N Hydrochloric acid (5.64 ml) and water (10 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate. The organic layer was washed with 2% and saturated saline, dried over sodium sulfate, and concentrated to give a yellow oil (0.99 g). To a suspension of this yellow oil (0.99 g) and potassium carbonate (0.39 g, 2.82 bandol) in DMF (15 ml) was added isopentyl iodide (0.30 ml, 2.26 mmol) with stirring at 60 ° C. The mixture was stirred for 2.5 hours. The reaction solution was extracted and purified in the same manner to obtain a tan oil (1.06 g).
¾-NMR (200MHz, CDC13): δθ.92 (6Η, d, J= 6.2 Hz), 1.29 (3H, d, J- 6.6 Hz), 1.53 (9H, s), 2.43 (3H, s), 2.99 (2H, m), 3.51 (1H, q, J= 7.0 Hz), 4.95 (1H, s), 7.30 (2H, d, J= 8.2 Hz), 7.88 (2H, d, J= 8.4 Hz), 9.59 (1H, brs). 得られた黄褐色油状物(1.06 g)、 無水酢酸(0.543 ml, 5.64 mmol)および p - TsOH 水和物(358mg, 1.88腿 oi)のトルエン(20 ml)懸濁液を 80°C で 50分間攪拌した。 反応液を同様に抽出精製して得られた油状物にジェチルェ一テルを加え、 析出物 を濾取し、 ジェチルエーテルで洗浄、 乾燥して標題化合物 (333 mg, 0.693 mmol) を赤橙色粉末として得た。 ¾-NMR (200MHz, CDC1 3 ): δθ.92 (6Η, d, J = 6.2 Hz), 1.29 (3H, d, J- 6.6 Hz), 1.53 (9H, s), 2.43 (3H, s), 2.99 (2H, m), 3.51 (1H, q, J = 7.0 Hz), 4.95 (1H, s), 7.30 (2H, d, J = 8.2 Hz), 7.88 (2H, d, J = 8.4 Hz), 9.59 (1H, brs). Suspension of the obtained tan oil (1.06 g), acetic anhydride (0.543 ml, 5.64 mmol) and p-TsOH hydrate (358 mg, 1.88 oi) in toluene (20 ml). The solution was stirred at 80 ° C for 50 minutes. The reaction mixture was extracted and purified in the same manner, and oil was obtained. Jethyl ether was added, and the precipitate was collected by filtration, washed with getyl ether and dried to give the title compound (333 mg, 0.693 mmol) as a red-orange powder As obtained.
Ή-N R (200MHz, 重クロ口ホルム) : δ 0.93 (6Η, d, J= 6.2 Hz), 1.56 (2H, brq, J= 6.7 Hz), 1.66 (1H, sixtet, J= 6.4 Hz), 2.00 (3H, s), 2.41 (3H, s), 2.97 (2H, t, J= 7.6 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.87 (2H, d, J= 8.4 Hz), 10.19 (1H, brs). Ή-NR (200 MHz, double-mouthed form): δ 0.93 (6Η, d, J = 6.2 Hz), 1.56 (2H, brq, J = 6.7 Hz), 1.66 (1H, sixtet, J = 6.4 Hz), 2.00 (3H, s), 2.41 (3H, s), 2.97 (2H, t, J = 7.6 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.87 (2H, d, J = 8.4 Hz), 10.19 (1H, brs).
元素分析値: C20H23N3O5S3 - 0.2Et20 として計算値: C, 50.32; H, 5.08; N, 8.46; S, 19.38 (¾), 実測値; C, 50.35; H, 5.15; , 8.47; S, 19.33 (%) . 融点: 193.0-195.0°C Elemental analysis: C 20 H 23 N 3 O 5 S 3 - 0.2Et 2 0 Calculated: C, 50.32; H, 5.08 ; N, 8.46; S, 19.38 (¾), Found; C, 50.35; H , 5.15;, 8.47; S, 19.33 (%). Melting point: 193.0-195.0 ° C
実施例 4-465 Example 4-465
N- [8- (ィソペンチルスルファニル) - 6 -メチル- 5 - (4-モルホリ二ルカルポニル) -3- ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン ]-4-メチル ベンゼンスルホンアミド N- [8- (Isopentylsulfanyl) -6-methyl-5- (4-morpholinylcarbonyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene] -4-methylbenzenesulfonamide
(方法 1) 実施例 3- 23で得た化合物 (純度 80¾, 0.26 g, 0.45 mmol)および炭酸 カリウム (93 mg, 0.68 mmol) の DMF (4.0 ml) の懸濁液に 70°Cで攪拌しながら ヨウ化イソペンチル (0.072 ml, 0.54 iiol)を加え、 混合物を 40分間攬拌した。 反応液に IN塩酸 (0.90 ml)および 0.05N塩酸(20 ml)を加えて酢酸ェチルで抽出 した。 有機層を水、 ^および飽和の食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣固形物にジェチルエーテルを加えて粉碎し、 析出物を濾取し、 ジ ェチルェ一テルで洗浄、 乾燥して標題化合物(235 mg, 0.427膽 ol)を赤色粉末と して得た。 (Method 1) A suspension of the compound obtained in Example 3-23 (purity 80¾, 0.26 g, 0.45 mmol) and potassium carbonate (93 mg, 0.68 mmol) in DMF (4.0 ml) was stirred at 70 ° C. While adding isopentyl iodide (0.072 ml, 0.54 iiol), the mixture was stirred for 40 minutes. IN hydrochloric acid (0.90 ml) and 0.05N hydrochloric acid (20 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate. The organic layer was washed with water, ^ and saturated saline, dried over sodium sulfate and concentrated. Getyl ether was added to the residual solid, which was then pulverized. The precipitate was collected by filtration, washed with diethyl ether, and dried to obtain the title compound (235 mg, 0.427 benzyl) as a red powder.
(方法 2) 実施例 4- 462で得た化合物 (0.60 g, 1.25匪 ol)、 H0BT (169 mg, 1.25 mmol)および WSC (249 mg, 1.30醒 ol)の THF (15 ml)溶液を室温で 4時間攪拌し た。 この間反応液に WSC (48 mg, 0.25匪 ol)を 1時間毎に 2回加えた。 反応液を 濃縮後、 酢酸ェチルで希釈し、 希塩酸、 2%炭酸水素ナトリウム、 水および飽和食 塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣にジェチルエーテルを 加えて析出物を濾取して実施例 4-462 で得た化合物の H0BTエステル (399 mg, 0.666腿 ol)を濃赤色固形物として得た。 この濃赤色固形物(399 mg, 0.666 mmol) の THF (8 ml)溶液にモルホリン(0.178 ml, 2.00匪 ol)を室温で加えて混合物を 室温で 24時間攪拌した。 反応液を濃縮後、 酢酸ェチルで希釈し、 希塩酸、 2%炭酸 水素ナトリゥム、 5%食塩水および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥し、 濃縮した。 残渣をシリカゲル(10 g)のクロマトグラフィ一 (酢酸ェチル—へキサ ン、 2:8-6:4) に付した。 酢酸ェチルーへキサン(6:4)の画分を濃縮し、 ジェチル エーテルから結晶化して標題化合物 (47 mg, 0.085 mmol)を赤色粉末として得た。 Ή-NMR (200MHz, CDC13) : δ 0.93 (6Η, d, J= 6.2 Hz), 1.56 (2H, brd, J= 7.2 Hz), 1.67 (1H, quint, J= 6.4 Hz), 1.80 (3H, s), 2.43 (3H, s), 2.95 (2H, t, J= 7.7 Hz), 3.49 (2H, m), 3.66 (4H, m), 3.80 (2H, m), 7.30 (2H, d, J= 8.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.73 (1H, brs). (Method 2) A solution of the compound obtained in Example 4-462 (0.60 g, 1.25 bandol), H0BT (169 mg, 1.25 mmol) and WSC (249 mg, 1.30 mmol) in THF (15 ml) was added at room temperature. Stirred for 4 hours. During this time, WSC (48 mg, 0.25 bandol) was added to the reaction solution twice every hour. The reaction mixture was concentrated, diluted with ethyl acetate, washed with dilute hydrochloric acid, 2% sodium bicarbonate, water and saturated saline, dried over sodium sulfate, and concentrated. Getyl ether was added to the residue, and the precipitate was collected by filtration to obtain a H0BT ester (399 mg, 0.666 tmol) of the compound obtained in Example 4-462 as a dark red solid. To a solution of this dark red solid (399 mg, 0.666 mmol) in THF (8 ml) was added morpholine (0.178 ml, 2.00 ol) at room temperature, and the mixture was stirred at room temperature for 24 hours. The reaction mixture was concentrated, diluted with ethyl acetate, washed with dilute hydrochloric acid, 2% sodium bicarbonate, 5% saline and saturated saline, dried over sodium sulfate, and concentrated. The residue was chromatographed on silica gel (10 g) (ethyl acetate-hexane, 2: 8-6: 4). The ethyl acetate-hexane (6: 4) fraction was concentrated and crystallized from getyl ether to give the title compound (47 mg, 0.085 mmol) as a red powder. Ή-NMR (200MHz, CDC1 3 ): δ 0.93 (6Η, d, J = 6.2 Hz), 1.56 (2H, brd, J = 7.2 Hz), 1.67 (1H, quint, J = 6.4 Hz), 1.80 (3H , s), 2.43 (3H, s), 2.95 (2H, t, J = 7.7 Hz), 3.49 (2H, m), 3.66 (4H, m), 3.80 (2H, m), 7.30 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.73 (1H, brs).
元素分析値: C24H3。N405S3として計算値: C, 52.34; H, 5.49; , 10.17; S, 17.47 (%) 、 実測値: C, 52.30; H, 5.46; N, 10.29; S, 17.35 (%) . Elemental analysis: C 24 H 3. Calculated N 4 0 5 S 3: C , 52.34; H, 5.49;, 10.17; S, 17.47 (%), Found: C, 52.30; H, 5.46 ; N, 10.29; S, 17.35 (%).
融点: 199.5- 201.5°C . Melting point: 199.5-201.5 ° C.
実施例 4-466 Example 4-466
Ή-NMR (200MHz, CDC13) : δ 0.93 (6H, d, J= 6.2 Hz), 1.08 (3H, t, J= 7.5 Hz), 1.61 (3H, m), 1.82 (3H, s), 2.42 (3H, s), 2.78 (2H, q, J= 7.5 Hz), 2.94 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.63 (1H, brs) . Ή-NMR (200MHz, CDC1 3 ): δ 0.93 (6H, d, J = 6.2 Hz), 1.08 (3H, t, J = 7.5 Hz), 1.61 (3H, m), 1.82 (3H, s), 2.42 (3H, s), 2.78 (2H, q, J = 7.5 Hz), 2.94 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
融点: 142.5-143.5°C Melting point: 142.5-143.5 ° C
実施例 4-468 Example 4-468
4 -メチル -N- {6-メチル- 8 - [ (2-メチルブチル)スルファニル] -3-ォキソ -5-プロピ ォニル -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜イリデン }ベンゼン スルホンアミド 4-Methyl-N- {6-methyl-8-[(2-methylbutyl) sulfanyl] -3-oxo-5-propionyl-2,3-dihydro-1H-imidazo [5, tric] [1,4 ] Thiazine-triylidene} benzene sulfonamide
実施例 3-22 で得た化合物(171 mg, 0.404 mmol)および炭酸カリウム(84 mg, 0.606删 ol)の DMF (3.0 ml) の懸濁液に 80°Cで攪拌しながら卜ョ一ド- 2-メチル ブタン (93 mg, 0.444匪 ol)を加え、 混合物を 40分間攪拌した。 反応液に 1N塩 酸 (0.81ml) およびメタノール一水(1:1, 4ml)を加え、室温で 30分間攪拌した。 析出物を濾取し、メタノール—水 (1:1)およびメタノールで洗浄、乾燥して標題 化合物 (63 mg, 0.128匪 ol)を赤褐色粉末として得た。 A suspension of the compound obtained in Example 3-22 (171 mg, 0.404 mmol) and potassium carbonate (84 mg, 0.606 mol) in DMF (3.0 ml) was stirred at 80 ° C. with stirring. 2-Methylbutane (93 mg, 0.444 ol) was added and the mixture was stirred for 40 minutes. 1N Hydrochloric acid (0.81 ml) and methanol / water (1: 1, 4 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (63 mg, 0.128 bandol) as a red-brown powder.
Ή-NMR (200MHz, CDC13) δ 0.92 (3Η, t, J= 7.3 Hz), 1.00 (3H, d, J= 6.6 Hz), 1.17 (3H, t, J= 7.3 Hz), 1.27 (1H, sixtet, J= 7.1 Hz), 1.50 (1H, m), 1.67 (1H, m), 1.76 (3H, s), 2.43 (3H, s), 2.57 (2H, q, J= 7.2 Hz), 2.79 (1H, dd, J= 12.4, 7.4 Hz), 2.97 (1H, dd, J= 12.6, 5.8 Hz), 7.30 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.74 (1H, brs). Ή-NMR (200MHz, CDC1 3 ) δ 0.92 (3Η, t, J = 7.3 Hz), 1.00 (3H, d, J = 6.6 Hz), 1.17 (3H, t, J = 7.3 Hz), 1.27 (1H, sixtet, J = 7.1 Hz), 1.50 (1H, m), 1.67 (1H, m), 1.76 (3H, s), 2.43 (3H, s), 2.57 (2H, q, J = 7.2 Hz), 2.79 ( 1H, dd, J = 12.4, 7.4 Hz), 2.97 (1H, dd, J = 12.6, 5.8 Hz), 7.30 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.74 (1H, brs).
元素分析値: C22H27N304S3として計算値: C, 53.53; H, 5.51; N, 8.51 (%), 実 測値: C, 53.48; H, 5.53; N, 8.24 (%) . Elemental analysis: C 22 H 27 N 3 0 4 S 3 Calculated: C, 53.53; H, 5.51 ; N, 8.51 (%), actual Hakachi: C, 53.48; H, 5.53 ; N, 8.24 (% ).
実施例 4-469Example 4-469
- [8- (ィソペンチルスルファニル) -6-メチル -3-ォキソ- 5-プロピオ二ル- 2, 3 ジ ヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン ]-4-メチルベンゼンスル ホンアミド -[8- (Isopentylsulfanyl) -6-methyl-3-oxo-5-propionyl-2,3 dihydro-1H-imidazo [5, tric] [1,4] thiazine-triylidene] -4-methylbenzenesulfonamide
実施例 3-22 で得た化合物(1.83 g, 4.33讓 ol)および炭酸カリウム (897 mg, 6.49 mmol) の DMF (30 ml) の懸濁液に 70°Cで攪拌しながらヨウ化イソペンチル (0.95 g, 4.76 mmol)を加え、 混合物を 40分間攪拌した。 反応液にヨウ化イソべ ンチル (95 mg, 0.433醒 ol)を加え、 混合物を 30分間攪拌した。 反応液に 1N塩 酸 (8.7ml) およびメタノール一水(1:1, 20ffll)を加え、室温で 60分間攪拌した。 析出物を濾取し、メタノ一ルー水 (1:1)およびメタノールで洗浄、乾燥して標題 化合物 (2.04 g, 4.13 mmol)を赤色粉末として得た。 Isopentyl iodide (0.95 g) was added to a suspension of the compound (1.83 g, 4.33 alcohol) obtained in Example 3-22 and potassium carbonate (897 mg, 6.49 mmol) in DMF (30 ml) while stirring at 70 ° C. g, 4.76 mmol) and the mixture was stirred for 40 minutes. Isobentyl iodide (95 mg, 0.433 mmol) was added to the reaction solution, and the mixture was stirred for 30 minutes. 1N Hydrochloric acid (8.7 ml) and methanol / water (1: 1, 20ffll) were added to the reaction solution, and the mixture was stirred at room temperature for 60 minutes. The precipitate is collected by filtration, washed with methanol (1: 1) and methanol, dried and dried The compound (2.04 g, 4.13 mmol) was obtained as a red powder.
lH-NMR (200MHz, CDC13) : 6 0.93 (6H, d, J- 6.2 Hz), 1.17 (3H, t, J= 7.3 Hz), 1.57 (2H, brq, J= 7.5 Hz), 1.69 (1H, quint, J= 6.7 Hz), 1.76 (3H, s), 2.43 (3H, s), 2.58 (2H, q, J= 7.2 Hz), 2.97 (2H, t, J= 7.7 Hz), 7.30 (2H, d, J= 8.0 Hz), 7.85 (2H, dt, J= 8.4, 1.8 Hz), 9.74 (1H, brs). lH-NMR (200MHz, CDC1 3 ): 6 0.93 (6H, d, J- 6.2 Hz), 1.17 (3H, t, J = 7.3 Hz), 1.57 (2H, brq, J = 7.5 Hz), 1.69 (1H , quint, J = 6.7 Hz), 1.76 (3H, s), 2.43 (3H, s), 2.58 (2H, q, J = 7.2 Hz), 2.97 (2H, t, J = 7.7 Hz), 7.30 (2H , d, J = 8.0 Hz), 7.85 (2H, dt, J = 8.4, 1.8 Hz), 9.74 (1H, brs).
元素分析値: C22H27N304S3: C, 53.53; H, 5.51; N, 8.51 (%) 実測値: C, 53.60;Elemental analysis: C 22 H 27 N 3 0 4 S 3: C, 53.53; H, 5.51; N, 8.51 (%) Found: C, 53.60;
H, 5.67; N, 8.51 (%) . H, 5.67; N, 8.51 (%).
実施例 4-472 Example 4-472
Ή-NMR (CDC13, 200 MHz): 6 1.02 (6H, d, J= 6.6 Hz), 1.79 (3H, brs), 1.90 (1H, sept, J= 6.7 Hz), 2.27 (3H, brs), 2.82 (2H, d, J= 7.0 Hz), 7.50 (3H, m), 7.99 (2H, brd, J= 6.2 Hz), 9.64 (1H, m). Ή-NMR (CDC1 3, 200 MHz): 6 1.02 (6H, d, J = 6.6 Hz), 1.79 (3H, brs), 1.90 (1H, sept, J = 6.7 Hz), 2.27 (3H, brs), 2.82 (2H, d, J = 7.0 Hz), 7.50 (3H, m), 7.99 (2H, brd, J = 6.2 Hz), 9.64 (1H, m).
元素分析値: C18H2lN303S3として計算値: C, 51.04; H, 5.00; N, 9.92; S, 22.71 (%)、 実測値: C, 50.88; H, 5.16; N, 10.10; S, 22.67 (%) . Elemental analysis: Calculated for C 18 H 2l N 3 0 3 S 3 : C, 51.04; H, 5.00; N, 9.92; S, 22.71 (%), found: C, 50.88; H, 5.16; N, 10.10; S, 22.67 (%).
実施例 4-473 Example 4-473
N-[8 -(イソペンチルスルファ二ル)- 5,6-ジメチル -3-ォキソ - 2,3-ジヒドロ- 1H-ィ ミダゾ [5,1- c] [1,4]チアジン- 1-イリデン]ベンゼンスルホンアミド N- [8- (Isopentylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ilidene] benzenesulfonamide
実施例 3-9 で得た化合物(243 mg, 0.661 mmol)および炭酸カリウム (137 mg, 0.992删 ol) の DMF (4.0 ml) の懸濁液に 60°Cで攪拌しながらヨウ化イソペンチ ル (159mg, 0.794腿 ol)を加え、 1.5時間攪拌した。 反応液に 1N塩酸 (1.32ml) を加え、 室温で 30分間攪拌した後、 メタノール一水(1:1, 10mi)で希釈した。 析 出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄、 乾燥して標題化 合物 (265 mg, 0.606 mmol)を灰濃赤色粉末として得た。 A suspension of the compound (243 mg, 0.661 mmol) obtained in Example 3-9 and potassium carbonate (137 mg, 0.992 mol) in DMF (4.0 ml) was stirred at 60 ° C while isopentyl iodide ( 159 mg, 0.794 thigh) and stirred for 1.5 hours. To the reaction solution was added 1N hydrochloric acid (1.32 ml), and the mixture was stirred at room temperature for 30 minutes, and then diluted with methanol-water (1: 1, 10 mi). The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (265 mg, 0.606 mmol) as a grey-red powder.
Ή-NMR (200MHz, CDC13) : δ 0.93 (6Η, d, J= 6.2 Hz), 1.56 (1H, q, J= 7.1 Hz), Ή-NMR (200MHz, CDC1 3 ): δ 0.93 (6Η, d, J = 6.2 Hz), 1.56 (1H, q, J = 7.1 Hz),
I.60 (1H, brq, J= 6.0 Hz), 1.69 (1H, m), 1.79 (3H, q, J= 1.2 Hz), 2.27 (3H, q, J- 0.8 Hz), 2.95 (2H, brq, J= 7.7 Hz), 7.51 (3H, m), 7.98 (2H, dd, J= 8.4,I.60 (1H, brq, J = 6.0 Hz), 1.69 (1H, m), 1.79 (3H, q, J = 1.2 Hz), 2.27 (3H, q, J- 0.8 Hz), 2.95 (2H, brq , J = 7.7 Hz), 7.51 (3H, m), 7.98 (2H, dd, J = 8.4,
1.8 Hz), 9.67 (1H, brs). 1.8 Hz), 9.67 (1H, brs).
元素分析値: C19H23N303S3として計算値: C, 52.15; H, 5.30; N, 9.60; S, 21.98Elemental analysis: C 19 H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30 ; N, 9.60; S, 21.98
(%) 、 実測値: C, 51.99; H, 5.06; N, 9.52; S, 21.97 (%) . (%), Found: C, 51.99; H, 5.06; N, 9.52; S, 21.97 (%).
融点. 170.5-172.5t: 実施例 4-474 170.5-172.5t: Example 4-474
'H-N R (CDC 13, 200 MHz): d 1.79 (3H, brs), 2.27 (3H, brs), 2.96 (2H, dd, J= 9.2, 5.6 Hz), 3.19 (2H, dd, J= 8.4, 6.6 Hz), 7.17-32 (5H, m), 7.50 (2H, m), 7.95 (2H, brd, J= 6.6 Hz), 9.65 (1H, brs). 'HNR (CDC 13, 200 MHz): d 1.79 (3H, brs), 2.27 (3H, brs), 2.96 (2H, dd, J = 9.2, 5.6 Hz), 3.19 (2H, dd, J = 8.4, 6.6 Hz), 7.17-32 (5H, m), 7.50 (2H, m), 7.95 (2H, brd, J = 6.6 Hz), 9.65 (1H, brs).
元素分析値: C22H21N303S3として計算値: C, 56.03; H, 4.49; N, 8.91; S, 20.40 (%), 実測値: C, 55.84; H, 4.65; N, 8.99; S, 20.30 (%) . Elemental analysis: Calculated for C 22 H 21 N 3 0 3 S 3 : C, 56.03; H, 4.49; N, 8.91; S, 20.40 (%), found: C, 55.84; H, 4.65; N, 8.99; S, 20.30 (%).
実施例 4-475 Example 4-475
lH-腿 (CDC13, 200 MHz): δ 1.02 (6Η, d, J= 6.6 Hz), 1.79 (3H, brs), 1.91 (1H, sept, J= 6.7 Hz), 2.26 (3H, s), 2.81 (3H, s), 2.84 (2H, d, J= 7.0 Hz), 7.30 (2H, m), 7.44 (1H, tm, J= 7.2 Hz), 8.03 (1H, dm, J= 8 Hz), 9.68 (1H, brs). lH- thigh (CDC1 3, 200 MHz): δ 1.02 (6Η, d, J = 6.6 Hz), 1.79 (3H, brs), 1.91 (1H, sept, J = 6.7 Hz), 2.26 (3H, s), 2.81 (3H, s), 2.84 (2H, d, J = 7.0 Hz), 7.30 (2H, m), 7.44 (1H, tm, J = 7.2 Hz), 8.03 (1H, dm, J = 8 Hz), 9.68 (1H, brs).
元素分析値: C19H23N303S3として計算値: C, 52.15; H, 5.30; N, 9.60; S, 21.98 (%), 実測値: C, 51.99; H, 5.09; N, 9.76; S,21.98 ( ) . Elemental analysis: C 19 H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30 ; N, 9.60; S, 21.98 (%), Found: C, 51.99; H, 5.09 ; N, 9.76; S, 21.98 ().
実施例 4 - 476 Example 4-476
丽 (CDC13, 200 MHz): 6 1.78 (3H, s), 2.26 (3H, brq, J= 0.6 Hz), 2.72 (3H, s), 2.96 (2H, brdd, J= 8.0, 7.0 Hz), 3.21 (2H, dd, J= 8.0, 6.6 Hz), 7.15—34 (7H, m), 7.45 (1H, tm, J= 7.2 Hz), 8.02 (1H, dm, J= 7.4 Hz), 9.66 (1H, brs). 元素分析値: C23 N303S3として計算値: C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%), 実測値: C, 56.93; H, 4.75; N, 8.91; S, 19.72 (%) . 丽(CDC1 3, 200 MHz): 6 1.78 (3H, s), 2.26 (3H, brq, J = 0.6 Hz), 2.72 (3H, s), 2.96 (2H, brdd, J = 8.0, 7.0 Hz), 3.21 (2H, dd, J = 8.0, 6.6 Hz), 7.15-34 (7H, m), 7.45 (1H, tm, J = 7.2 Hz), 8.02 (1H, dm, J = 7.4 Hz), 9.66 (1H , brs). Elemental analysis: Calculated for C 23 N 3 0 3 S 3 : C, 56.88; H, 4.77; N, 8.65; S, 19.81 (%), found: C, 56.93; H, 4.75; N, 8.91; S, 19.72 (%).
実施例 4-477 Example 4-477
'H-NMR (CDC13, 200 MHz): δ 1.02 (6H, d, J= 6.6 Hz), 1.79 (3H, s), 1.91 (1H, sept, J- 6.8 Hz), 2.27 (3H, s), 2.41 (3H, s), 2.82 (2H, d, J= 7.0 Hz), 7.36 (2H, m), 7.79 (2H, m), 9.63 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1.02 (6H, d, J = 6.6 Hz), 1.79 (3H, s), 1.91 (1H, sept, J- 6.8 Hz), 2.27 (3H, s) , 2.41 (3H, s), 2.82 (2H, d, J = 7.0 Hz), 7.36 (2H, m), 7.79 (2H, m), 9.63 (1H, brs).
元素分析値: CigH23N303S3として計算値: C, 52.15; H, 5.30; N, 9.60; S, 21.98 (%), 実測値: C, 52.02; H, 5.37; N, 9.61; S, 21.93 (%) . Elemental analysis: C ig H 23 N 3 0 3 S 3 Calculated: C, 52.15; H, 5.30 ; N, 9.60; S, 21.98 (%), Found: C, 52.02; H, 5.37 ; N, 9.61; S, 21.93 (%).
実施例 4-478 Example 4-478
N- [8- (ィソぺンチルスルファニル) -5, 6-ジメチル- 3-ォキソ -2, 3-ジヒド口- 1H -ィ ミダゾ [5,卜 c] [1,4]チアジン-卜イリデン] -3-メチルベンゼンスルホンアミド 実施例 3-25で得た化合物(258 mg, 0.676 mmol)および炭酸カリウム Q40 mg, 1.01 mmol) の DMF (4.0 ml) の懸濁液に 60°Cで攪拌しながらヨウ化ィ N- [8- (Isopentylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydrido-1H-imidazo [5, tric] [1,4] thiazine-triylidene ] -3-Methylbenzenesulfonamide The compound obtained in Example 3-25 (258 mg, 0.676 mmol) and potassium carbonate Q40 mg, 1.01 mmol) in DMF (4.0 ml) while stirring at 60 ° C.
ル (163 rag, 0.812讓 ol)を加え、 1.5時間攪拌した。 反応液に 1N塩酸 (1.35 ml) を加え、 室温で 30分間攪拌した後、 メタノール—水(1:1, 10ml)で希釈した。 析 出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄、 乾燥して標題化 合物 (282 mg, 0.624匪 ol)を灰濃赤色粉末として得た。 (163 rag, 0.812 benzene) was added thereto, followed by stirring for 1.5 hours. 1N Hydrochloric acid (1.35 ml) was added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes and diluted with methanol-water (1: 1, 10 ml). The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (282 mg, 0.624 bandol) as a dark red powder.
'H-NMR (200MHz, CDC13) : δ 0.93 (6H, d, J= 6.2 Hz), 1.56 (1H, q, J= 7.0 Hz), 1.60 (1H, brq, J= 6.6 Hz), 1.67 (1H, in), 1.79 (3H, q, J= 0.8 Hz), 2.27 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.95 (2H, brt, J= 7.7 Hz), 7.37 (2H, dd, J= 5.4, 1.4 Hz), 7.77 (2H, m), 9.65 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 0.93 (6H, d, J = 6.2 Hz), 1.56 (1H, q, J = 7.0 Hz), 1.60 (1H, brq, J = 6.6 Hz), 1.67 ( 1H, in), 1.79 (3H, q, J = 0.8 Hz), 2.27 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 2.95 (2H, brt, J = 7.7 Hz), 7.37 ( 2H, dd, J = 5.4, 1.4 Hz), 7.77 (2H, m), 9.65 (1H, brs).
元素分析値: CM 5N303S3として計算値: C, 53.09; H, 5.58; N, 9.30; S, 21.30 (%) 、 実測値: C, 53.16; H, 5.67; N, 9.42; S, 21.35 (%) 、 . Elemental analysis: C M 5 N 3 0 3 S 3 Calculated: C, 53.09; H, 5.58; N, 9.30; S, 21.30 (%), found: C, 53.16; H, 5.67; N, 9.42 ; S, 21.35 (%),.
融点. 150.0- 150.8°C Melting point.150.0- 150.8 ° C
実施例 4-479 Example 4-479
'H-NMR (CDC13, 200 MHz): δ 1.79 (3Η, q, J= 1.0 Hz), 2.27 (3H, q, J= 0.6 Hz), 2.42 (3H, s), 2.97 (2H, dd, J= 9.2, 5.6 Hz), 3.18 (2H, dd, J= 9.2, 5.8 Hz), 7.26 (5H, m), 7.36 (2H, m), 7.76 (2H, brs), 9.65 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1.79 (3Η, q, J = 1.0 Hz), 2.27 (3H, q, J = 0.6 Hz), 2.42 (3H, s), 2.97 (2H, dd, J = 9.2, 5.6 Hz), 3.18 (2H, dd, J = 9.2, 5.8 Hz), 7.26 (5H, m), 7.36 (2H, m), 7.76 (2H, brs), 9.65 (1H, brs).
元素分析値: C23H23N303S3として計算値: C, 56.88; H, 4.77; N, 8.65; S, 19.81Elemental analysis: C 23 H 23 N 3 0 3 S 3 Calculated: C, 56.88; H, 4.77 ; N, 8.65; S, 19.81
(%), 実測値: C, 56.97; H, 4.99; N, 8.56; S, 19.59 (%) . (%), Found: C, 56.97; H, 4.99; N, 8.56; S, 19.59 (%).
実施例 4-480 Example 4-480
¾-NMR (CDC13, 200 MHz): δ 1.02 (6H, d, J= 6.6 Hz), 1.79 (3H, q, J= 1.2 Hz), 1.91 (1H, quint, J= 6.7 Hz), 2.27 (3H, q, J= 1.2 Hz), 2.83 (2H, d, J= 6.6 Hz), 7.15 (2H, t, J= 8.8 Hz), 8.00 (2H, dd, J= 8.8, 4.8 Hz), 9.65 (1H, brs) . ¾-NMR (CDC1 3, 200 MHz): δ 1.02 (6H, d, J = 6.6 Hz), 1.79 (3H, q, J = 1.2 Hz), 1.91 (1H, quint, J = 6.7 Hz), 2.27 ( 3H, q, J = 1.2 Hz), 2.83 (2H, d, J = 6.6 Hz), 7.15 (2H, t, J = 8.8 Hz), 8.00 (2H, dd, J = 8.8, 4.8 Hz), 9.65 ( 1H, brs).
元素分析値: C18H2。N303S3F - 1/3H20として計算値: C, 48.30; H, 4.58; N, 9.39; S, 21.49; F, 4.24 (%), 実測値: C, 48.24; H, 4.56; N, 9.45; S, 21.65; F, 4.44 (%) . Elemental analysis: C 18 H 2. Calculated as N 3 0 3 S 3 F-1 / 3H 2 0: C, 48.30; H, 4.58; N, 9.39; S, 21.49; F, 4.24 (%), found: C, 48.24; H, 4.56 ; N, 9.45; S, 21.65; F, 4.44 (%).
実施例 4-481 ' Example 4-481 '
4-フルォ口- N- [8- (ィソペンチルスルファニル) -5, 6 -ジメチル- 3-ォキソ- 2, 3 -ジ ヒドロ -1H-イミダゾ [5, l-c][l, 4]チアジン-卜イリデン]ベンゼンスルホンアミド 実施例 3-35で得た化合物 (340 mg, 0.882 mmol)および炭酸力リゥム Q83 mg, 1.32膽 ol) の DMF (6.0 ml) の懸濁液に 60°Cで攪拌しながらヨウ化イソペンチ ル (211 mg, 1.06腿 οθを加え、 1.5時間攪拌した。 反応液に 1N塩酸 (1.76 ml) を加え、 室温で 30分間攪拌した後、 メタノール一水(1:1, 10ml)で希釈した。 析 出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄、 乾燥して標題化 合物 (373 mg, 0.819 mmol)を赤褐色粉末として得た。 4-Fluoro-N- [8- (isopentylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, lc] [l, 4] thiazine Ilidene] benzenesulfonamide To a suspension of the compound obtained in Example 3-35 (340 mg, 0.882 mmol) and carbon dioxide lime Q83 mg (1.32 bunol) in DMF (6.0 ml) was stirred at 60 ° C while isopentyl iodide (60 mg). To the reaction mixture was added 1N hydrochloric acid (1.76 ml), the mixture was stirred at room temperature for 30 minutes, and diluted with methanol / water (1: 1, 10 ml). The product was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (373 mg, 0.819 mmol) as a red-brown powder.
'H-NMR (200MHz, CDC13) : δ 0.93 (6Η, d, J= 6.2 Hz), 1.56 (1H, q, J= 7.0 Hz), 1.59 (1H, q, J= 7.0 Hz), 1.66 (1H, m), 1.80 (3H, q, J= 1.2 Hz), 2.28 (3H, q, J= 1.0 Hz), 2.95 (2H, brt, J= 7.7 Hz), 7.16 (2H, tt, J= 8.8, 2.2 Hz), 7.98 (2H, ddt, J= 8.8, 4.8, 2.6 Hz), 9.64 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 0.93 (6Η, d, J = 6.2 Hz), 1.56 (1H, q, J = 7.0 Hz), 1.59 (1H, q, J = 7.0 Hz), 1.66 ( 1H, m), 1.80 (3H, q, J = 1.2 Hz), 2.28 (3H, q, J = 1.0 Hz), 2.95 (2H, brt, J = 7.7 Hz), 7.16 (2H, tt, J = 8.8 , 2.2 Hz), 7.98 (2H, ddt, J = 8.8, 4.8, 2.6 Hz), 9.64 (1H, brs).
元素分析値: C19H22N303S3Fとして計算値: C, 50.09; H, 4.87; N, 9.22; S, 21.11; F, 4.17 (%) 、 実測値: C, 50.03; H, 4.76; N, 9.21; S, 21.17; F, 4.14 (%) · 融点. 167.0- 168.0°C Elemental analysis: Calculated for C 19 H 22 N 3 0 3 S 3 F: C, 50.09; H, 4.87; N, 9.22; S, 21.11; F, 4.17 (%), found: C, 50.03; H , 4.76; N, 9.21; S, 21.17; F, 4.14 (%)
実施例 4-482 Example 4-482
'H-NMR (CDC13, 200 MHz): δ 1.80 (3H, brq, J= 0.6 Hz), 2.27 (3H, brq, J= 1.2 Hz), 2.97 (2H, brdd, J= 8.0, 6.2 Hz), 3.20 (2H, dd, J= 8.4, 6.6 Hz), 7.14 (2H, t, J= 8.4 Hz), 7.20 (2H, d, J= 6.2 Hz), 7.29 (3H, m), 7.95 (2H, dd, J= 9.2, 5.2 Hz), 9.60 (1H, brs). 'H-NMR (CDC1 3, 200 MHz): δ 1.80 (3H, brq, J = 0.6 Hz), 2.27 (3H, brq, J = 1.2 Hz), 2.97 (2H, brdd, J = 8.0, 6.2 Hz) , 3.20 (2H, dd, J = 8.4, 6.6 Hz), 7.14 (2H, t, J = 8.4 Hz), 7.20 (2H, d, J = 6.2 Hz), 7.29 (3H, m), 7.95 (2H, dd, J = 9.2, 5.2 Hz), 9.60 (1H, brs).
元素分析値: C22H20N303S3Fとして計算値: C, 53.97; H, 4.12; N, 8.58; S, 19.65; F, 3.88 (%). 実測値: C, 53.93; H, 3.87; N, 8.49; S, 19.68; F, 3.75 (%) . 実施例 4-483 Elemental analysis: C 22 H 20 N 3 0 3 S 3 F Calculated:. C, 53.97; H, 4.12; N, 8.58; S, 19.65; F, 3.88 (%) Found: C, 53.93; H , 3.87; N, 8.49; S, 19.68; F, 3.75 (%).
Ή-NMR (CDC13, 200 MHz): δ 0.98 (6H, d, J= 6.6 Hz), 1.80 (3H, brs), 1.88 (m, sept, J= 6.6 Hz), 2.30 (3H, s), 2.80 (2H, d, J= 7.0 Hz), 7.41 (1H, m), 7.48 (2H, m), 8.19 (1H, d, J=7.0 Hz), 9.73 (1H, brs). Ή-NMR (CDC1 3, 200 MHz): δ 0.98 (6H, d, J = 6.6 Hz), 1.80 (3H, brs), 1.88 (m, sept, J = 6.6 Hz), 2.30 (3H, s), 2.80 (2H, d, J = 7.0 Hz), 7.41 (1H, m), 7.48 (2H, m), 8.19 (1H, d, J = 7.0 Hz), 9.73 (1H, brs).
元素分析値: C18H20N303S3C1として計算値: C, 47.20; H, 4.40; N, 9.17; S, 21.00; Cl, 7.74 (%), 実測値: C, 46.99; H, 4.25; N, 9.17; S, 20.86; C1, 7.71 (%) . 実施例 4-486 Elemental analysis: Calculated as C 18 H 20 N 3 0 3 S 3 C1: C, 47.20; H, 4.40; N, 9.17; S, 21.00; Cl, 7.74 (%), Found: C, 46.99; H , 4.25; N, 9.17; S, 20.86; C1, 7.71 (%).
4一クロ口— N - {5, 6-ジメチル- 3-ォキソ -8- [ (4, 4, 5, 5, 5-ペンタフルォロペンチル) スルファニル ]_2, 3-ジヒドロ- 1H-イミダゾ [5, 1- c] [1,4]チアジン- 1-ィリデン }ベ ンゼンスルホンアミド 4 Monochrome—N- {5,6-dimethyl-3-oxo-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl] _2,3-dihydro-1H-imidazo [ 5, 1-c] [1,4] thiazine-1-ylidene Nzensulfonamide
実施例 3-6で得た化合物 (364 nig, 0.906 腿 ol)および炭酸カリウム(188 mg, 1.36 IMOI)の DMF (6 ml)の懸濁液に 60°Cで攪拌しながら参考例 5で得たメタン スルホン酸 4, 4, 5, 5, 5-ペン夕フルォロペンチル(278 mg, 1.09匪 ol)を加え、 反 応液を60°〇で5.5時間、 室温で 3日間攪拌した。 この間反応液にメタンスルホン 酸 4, 4, 5, 5, 5-ペン夕フルォロペンチル(70 mg, 0.27腿 ol)を加えた。 反応液に 1N 塩酸 (1.81 ml)を加え、 メタノール一水(1:1, 5 ml)で希釈し、 室温で 10分間攪 拌した。 析出物を濾取し、 メタノール一水(1:1) およびメタノールで洗浄後、 メ 夕ノール— THFから結晶化して標題化合物 (315 mg, 0.560 mmol)を赤紫色粉末と して得た。 A suspension of the compound obtained in Example 3-6 (364 nig, 0.906 thigh) and potassium carbonate (188 mg, 1.36 IMOI) in DMF (6 ml) was obtained in Reference Example 5 with stirring at 60 ° C. 4,4,5,5,5-Pentafluoropentyl methanesulfonate (278 mg, 1.09 marl) was added, and the reaction solution was stirred at 60 ° C for 5.5 hours and at room temperature for 3 days. During this time, 4,4,5,5,5-pentanofluorenepentyl methanesulfonate (70 mg, 0.27 mol) was added to the reaction mixture. To the reaction solution was added 1N hydrochloric acid (1.81 ml), diluted with methanol / water (1: 1, 5 ml), and stirred at room temperature for 10 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and crystallized from methanol-THF to give the title compound (315 mg, 0.560 mmol) as a red-purple powder.
H - NMR (200MHz, CDC13) : δ 1.80 (3H, q, J= 0.8 Hz), 2.05 (2H, m), 2.14 (2H, m), 2.28 (3H, q, J= 0.6 Hz), 3.03 (2H, t, J= 7.1 Hz), 7.47 (2H, d, J= 8.8 Hz), 7.90 (2H, dt, J= 8.8, 1.8 Hz), 9.62 (1H, brs). H - NMR (200MHz, CDC1 3 ): δ 1.80 (3H, q, J = 0.8 Hz), 2.05 (2H, m), 2.14 (2H, m), 2.28 (3H, q, J = 0.6 Hz), 3.03 (2H, t, J = 7.1 Hz), 7.47 (2H, d, J = 8.8 Hz), 7.90 (2H, dt, J = 8.8, 1.8 Hz), 9.62 (1H, brs).
元素分析値: C19H17N303S3C1F7として計算値: C, 40.61; H, 3.05; N, 7.48; S, 17.12; C1, 6.31; F, 16.90 (%) 、 実測値: C, 40.49; H, 2.78; N, 7.49; S, 17.19; CI, 6.32; F, 17.09 '(%) · Elemental analysis: Calculated as C 19 H 17 N 3 0 3 S 3 C1F 7: C, 40.61; H, 3.05; N, 7.48; S, 17.12; C1, 6.31; F, 16.90 (%), Found: C, 40.49; H, 2.78; N, 7.49; S, 17.19; CI, 6.32; F, 17.09 '(%)
融点. 173.5-175.0°C Melting point. 173.5-175.0 ° C
実施例 4-487 Example 4-487
'H-NMR (CDC13, 200 MHz):, δ 1.43 (4H, m), 1.55-1.70 (4H, m), 1.79 (3H, q, J= 0.8 Hz), 2.28 (3H, brq, J= 0.8 Hz), 2.96 (2H, t, J= 7.0 Hz), 3.66 (2H, t, J= 6.2 Hz), 7.46 (2H, d, J= 8.8 Hz) , 7.91 (2H, d, J= 8.8 Hz), 9.66 (1H, brs). 'H-NMR (CDC1 3, 200 MHz) :, δ 1.43 (4H, m), 1.55-1.70 (4H, m), 1.79 (3H, q, J = 0.8 Hz), 2.28 (3H, brq, J = 0.8 Hz), 2.96 (2H, t, J = 7.0 Hz), 3.66 (2H, t, J = 6.2 Hz), 7.46 (2H, d, J = 8.8 Hz), 7.91 (2H, d, J = 8.8 Hz) ), 9.66 (1H, brs).
元素分析値: C21H27N304S3C1として計算値: C, 47.84; H, 4.82; N, 8.37; S, 19.16; - C1, 7.06 (%)、 実測値: C, 47.51; H, 4.67; N, 8.52; S, 19.23; C1, 6.87 (%) . 実施例 4-488 Elemental analysis: C 21 H 27 N 3 0 4 S 3 C1 Calculated: C, 47.84; H, 4.82 ; N, 8.37; S, 19.16; - C1, 7.06 (%), Found: C, 47.51; H, 4.67; N, 8.52; S, 19.23; C1, 6.87 (%). Example 4-488
Ή-證 (CDC13, 200 MHz): δ 1.03 (6Η, d, J= 6.6 Hz), 1.79 (3H, q, J= 1.2 Hz), 1.91 (l.H, sept, J= 6.6 Hz), 2.27 (3H, q, J= 0.8 Hz), 2.83 (2H, d, J= 7.0 Hz), 7.45 (2H, d, J= 8.8 Hz), 7.92 (2H, d, J= 8.8 Hz), 9.62 (1H, brs). 元素分析値: C18H20N303S3C1として計算値: C, 47.20; H, 4.40; N, 9.17; S, 21.00; CI, 7.74 (%), 実測値: C, 47.02; H, 4.29; N, 9.18; S, 20.98; CI, 7.69 (%) . 実施例 4-490 Ή- testimony (CDC1 3, 200 MHz): δ 1.03 (6Η, d, J = 6.6 Hz), 1.79 (3H, q, J = 1.2 Hz), 1.91 (lH, sept, J = 6.6 Hz), 2.27 ( 3H, q, J = 0.8 Hz), 2.83 (2H, d, J = 7.0 Hz), 7.45 (2H, d, J = 8.8 Hz), 7.92 (2H, d, J = 8.8 Hz), 9.62 (1H, brs). Elemental analysis: Calculated for C 18 H 20 N 3 0 3 S 3 C1: C, 47.20; H, 4.40; N, 9.17; S, 21.00; CI, 7.74 (%), Found: C, 47.02; H, 4.29; N, 9.18; S, 20.98; CI, 7.69 (%).
Ή-NMR (CDC13, 200 MHz): d 1.80 (3H, brs), 2.27 (3H, brs), 2.97 (2H, dd, J= 8.0, 7.0 Hz), 3.20 (2H, dd, J= 8.0, 6.6 Hz), 7.19 (2H, brd, J- 7.6 Hz), 7.29 (3H, brt, J= 7.0 Hz), 7.44 (2H, d, J= 8.8 Hz), 7.87 (2H, d, J= 8.8 Hz), 9.62 (m, brs). Ή-NMR (CDC1 3, 200 MHz): d 1.80 (3H, brs), 2.27 (3H, brs), 2.97 (2H, dd, J = 8.0, 7.0 Hz), 3.20 (2H, dd, J = 8.0, 6.6 Hz), 7.19 (2H, brd, J- 7.6 Hz), 7.29 (3H, brt, J = 7.0 Hz), 7.44 (2H, d, J = 8.8 Hz), 7.87 (2H, d, J = 8.8 Hz) ), 9.62 (m, brs).
元素分析値: C20H20N303S3C1として計算値: C, 52.21; H, 3.98; N, 8.30; S, 19.01; C1, 7.01 (%), 実測値: C, 52.25; H, 4.10; N, 8.30; S, 18.87; C1, 6.77 (%) · 実施例 4-493 Elemental analysis: Calculated for C 20 H 20 N 3 0 3 S 3 C1: C, 52.21; H, 3.98; N, 8.30; S, 19.01; C1, 7.01 (%), found: C, 52.25; H , 4.10; N, 8.30; S, 18.87; C1, 6.77 (%)
'H-NMR (CDC13, 200 MHz): d 1.02 (6H, d, J= 6.6 Hz), 1.79 (3H, brq, J= 0.8 Hz), 1.91 (1H, sept, J- 6.7 Hz), 2.27 (3H, q, J= 1.0 Hz), 2.82 (2H, d, J= 7.0 Hz), 7.01 (2H, d, J= 8.8 Hz), 7.06 (2H, dm, J= 8 Hz), 7.20 (1H, t, J= 7.2 Hz), 7.40 (2H, t, J= 8.0 Hz), 7.92 (2H, dm, J= 9.2 Hz), 9.60 (1H, brs). 元素分析値: C24H25N304S3として計算値: C, 55.90; H, 4.89; N, 8.15; S, 18.65 'H-NMR (CDC1 3, 200 MHz): d 1.02 (6H, d, J = 6.6 Hz), 1.79 (3H, brq, J = 0.8 Hz), 1.91 (1H, sept, J- 6.7 Hz), 2.27 (3H, q, J = 1.0 Hz), 2.82 (2H, d, J = 7.0 Hz), 7.01 (2H, d, J = 8.8 Hz), 7.06 (2H, dm, J = 8 Hz), 7.20 (1H , t, J = 7.2 Hz), 7.40 (2H, t, J = 8.0 Hz), 7.92 (2H, dm, J = 9.2 Hz), 9.60 (1H, brs). Elemental analysis: C 24 H 25 N 3 0 4 S 3 calculated: C, 55.90; H, 4.89 ; N, 8.15; S, 18.65
(%), 実測値: C, 55.78; H, 4.91; , 8.38; S, 18.56 (%) . (%), Found: C, 55.78; H, 4.91;, 8.38; S, 18.56 (%).
実施例 4-494 Example 4-494
Ή-腿 (CDC13, 200 MHz): δ 1.80 (3Η, brs), 2.27 (3H, brs), 2.97 (2H, dd, J= 8.4, 7.8 Hz) , 3.20 (2H, dd, J= 8.2, 7.0 Hz) , 7.00 (2H, d, J= 8.8 Hz), 7.07 (2H, d, J= 7.4 Hz), 7.23 (6H, m), 7.41 (2H, t, J= 8.2 Hz), 7.88 (2H, d, J= 9.2 Hz), 9.61 (1H, brs). Ή- thigh (CDC1 3, 200 MHz): δ 1.80 (3Η, brs), 2.27 (3H, brs), 2.97 (2H, dd, J = 8.4, 7.8 Hz), 3.20 (2H, dd, J = 8.2, 7.0 Hz), 7.00 (2H, d, J = 8.8 Hz), 7.07 (2H, d, J = 7.4 Hz), 7.23 (6H, m), 7.41 (2H, t, J = 8.2 Hz), 7.88 (2H , d, J = 9.2 Hz), 9.61 (1H, brs).
元素分析値: C2SH25N304S3として計算値: C, 59.66; H, 4.47; N, 7.45; S, 17.07Elemental analysis: C 2S H 25 N 3 0 4 S 3 Calculated: C, 59.66; H, 4.47 ; N, 7.45; S, 17.07
{%), 実測値: C, 59.43; H, 4.39; N, 7.56; S, 17.05 (%) . (%), Found: C, 59.43; H, 4.39; N, 7.56; S, 17.05 (%).
実施例 4- 495 Example 4-495
4 -ク口口 _N- [8- (シクロォクチルスルファ二ル)- 5, 6 -ジメチル- 3-ォキソ -2, 3 -ジヒ ド口- 1H-ィミダゾ [5,卜 c] [1, ]チアジン-卜ィリデン]ベンゼンスルホンアミド 実施例 3-6で得た化合物 (279 mg, 0.694 mmol)および炭酸カリウム (144 mg, 1.04 mmol) の DMF (4.5 ml) の懸濁液に 50°Cで攪拌しながらブロモシクロォク 夕ン (178 mg, 0.902廳 ol)を加え、 44時間攪拌した。 反応中、 ブロモシクロォ クタン (178 mg, 0.902 mmol)を 3回、 および炭酸カリウム(48 mg, 0.347 mmol) を 2回、 反応液に追加した。 反応液を実施例 4-164と同様に後処理して表題化合 物 (130 mg, 0.254 mmol)を紫色粉末として得た。 4-N- [8- (cyclooctylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro- 1H-imidazo [5, tric] [1, [Thiazine-triylidene] benzenesulfonamide A suspension of the compound obtained in Example 3-6 (279 mg, 0.694 mmol) and potassium carbonate (144 mg , 1.04 mmol) in DMF (4.5 ml) was added at 50 ° C. While stirring with bromocyclooctane (178 mg, 0.902 cafe), the mixture was stirred for 44 hours. During the reaction, bromocyclooctane (178 mg, 0.902 mmol) was added three times and potassium carbonate (48 mg, 0.347 mmol) Was added to the reaction twice. The reaction solution was worked up as in Example 4-164 to give the title compound (130 mg, 0.254 mmol) as a purple powder.
¾ー丽 R (200MHz, CDC13) : δ 1.50-1.80 (10H, m), 1.80 (3H, q, J= 0.8 Hz), 2.00 (4H, 111), 2.27 (3H, q, J= 1.2 Hz), 3.60 (1H, sept, J= 4.7 Hz), 7.46 (2H, dt, J= 8.8, 2.0 Hz), 7.92 (2H, dt, J= 8.8, 2.0 Hz), 9.60 (1H, brs). ¾ over丽R (200MHz, CDC1 3): δ 1.50-1.80 (10H, m), 1.80 (3H, q, J = 0.8 Hz), 2.00 (4H, 111), 2.27 (3H, q, J = 1.2 Hz ), 3.60 (1H, sept, J = 4.7 Hz), 7.46 (2H, dt, J = 8.8, 2.0 Hz), 7.92 (2H, dt, J = 8.8, 2.0 Hz), 9.60 (1H, brs).
元素分析値 C22 6N303S3C1として計算値: C, 51.60; H, 5.12; N, 8.21; S, 18.78; CI, 6.92 (%) 、 Elemental analysis: Calculated for C 2 2 6 N 3 0 3 S 3 C1: C, 51.60; H, 5.12; N, 8.21; S, 18.78; CI, 6.92 (%),
実測値: C, 51.36; H, 4.93; N, 8.27; S, 18.88; C1, 7.03 (%) . Found: C, 51.36; H, 4.93; N, 8.27; S, 18.88; C1, 7.03 (%).
即. 177.5-179.5°C. Immediate. 177.5-179.5 ° C.
実施例 4-496 Example 4-496
4 -クロ口 -N-{8- [(2, 2-ジメチルブチル)スルファニル] -5, 6 -ジメチル -3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン }ベンゼンスルホン アミド 4-chloro-N- {8-[(2,2-dimethylbutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1, 4] thiazine-1-ylidene} benzenesulfonamide
実施例 3-6で得た化合物(3Q8 mg, 0.766 mmol)、 , 2-ジメチルブタノ一ル (144 mg, 0.996 mmol) およびトリフエニルホスフィン(290 mg, 1.07歷 ol)の THF (6.0 ml)懸濁液に室温で攪拌しながらジェチルァゾジカルポキシレート (以下、 DEAD と略記することがある。 ) (0.178 ml, 1.07讓 ol)を滴下し、 反応液を室温で 3 時間、 50°C で 23時間攪拌した。反応液に酢酸ェチル(35 ml)を加えて希釈し、 0.1N 塩酸、 水および飽和食塩水で洗浄し、 乾燥、 濃縮後、 メタノールから結晶化して 表題化合物(198 mg, 0.407讓 ol)を紫色粉末として得た。 A suspension of the compound obtained in Example 3-6 (3Q8 mg, 0.766 mmol), 2,2-dimethylbutanol (144 mg, 0.996 mmol) and triphenylphosphine (290 mg, 1.07 mol) in THF (6.0 ml). While stirring at room temperature, getyl azodicarboxylate (hereinafter may be abbreviated as DEAD) (0.178 ml, 1.07 mL) was added dropwise to the solution, and the reaction solution was added at room temperature for 3 hours at 50 ° C. Stirred for 23 hours. The reaction mixture was diluted with ethyl acetate (35 ml), washed with 0.1N hydrochloric acid, water and saturated saline, dried, concentrated and crystallized from methanol to give the title compound (198 mg, 0.407 benzyl) in purple Obtained as a powder.
Ή-NMR (200MHz, CDC13) : (50.85 (3H, t, J= 7.5 Hz), 0.97 (6H, s), 1.35 (2H, q, J= 7.5 Hz), 1.79 (3H, brs), 2.27 (3H, brs), 2.89 (2H, s), 7.44 (2H, brd, J= 8.6 Hz), 7.93 (2H, brd, J= 8.4 Hz), 9.58 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): (50.85 (3H, t, J = 7.5 Hz), 0.97 (6H, s), 1.35 (2H, q, J = 7.5 Hz), 1.79 (3H, brs), 2.27 (3H, brs), 2.89 (2H, s), 7.44 (2H, brd, J = 8.6 Hz), 7.93 (2H, brd, J = 8.4 Hz), 9.58 (1H, brs).
元素分析値: C20H24N303S3C1として計算値: C, 49.42; H, 4.98; N, 8.64; S, 19.79; C1, 7.29. Elemental analysis: C 20 H 24 N 3 0 3 S 3 C1 Calculated: C, 49.42; H, 4.98 ; N, 8.64; S, 19.79; C1, 7.29.
実測値: C, 49.20; H, 4.85.; N, 8.71; S, 19.76; CI, 7.28. Found: C, 49.20; H, 4.85 .; N, 8.71; S, 19.76; CI, 7.28.
mp. 191.0-193.0°C mp. 191.0-193.0 ° C
実施例 4- 497 Example 4-497
4 -ブロモ -N- {8- [(2, 2 -ジメチルブチル)スルファニル ]-5, 6-ジメチル -3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン -1-ィリデン }ベンゼンスルホン アミド 4-bromo-N- {8-[(2,2-dimethylbutyl) sulfanyl] -5,6-dimethyl-3-oxo -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene} benzenesulfonamide
実施例 3- 7で得た化合物 (299 mg, 0.670 mmol)、 2, 2-ジメチルブ夕ノール (75 mg, 0.737 腿 ol) およびトリフエニルホスフィン(254 mg, 0.938丽 ol)の THF (6.5 ml)懸濁液に室温で攪拌しながら DEAD (0.155 ml, 0.938腿 ol)を滴下し、 反応液を室温で 4時間、 50°C で 46時間攪拌した。 反応液に酢酸ェチル (35 ml) を加えて希釈し、 0.1N塩酸、 水および飽和食塩水で洗浄し、 乾燥、 濃縮後、 メタ ノールから結晶化して表題化合物(232 mg, 0.438 mmol)を紫色粉末として得た。 Ή-丽 (200MHz, CDC13) : 60.85 (3H, t, J= 7.3 Hz), 0.98 (6H, s), 1.35 (2H, q, J= 7.5 Hz), 1.80 (3H, s), 2.27 (3H, s), 2.89 (2H, s), 7.61 (2H, d, J= 8.8 Hz), 7.85 (2H, d, J= 8.8 Hz), 9.57 (1H, brs). THF (6.5 ml) of the compound obtained in Example 3-7 (299 mg, 0.670 mmol), 2,2-dimethylbutanol (75 mg, 0.737 tmol) and triphenylphosphine (254 mg, 0.938 mol) DEAD (0.155 ml, 0.938 liter) was added dropwise to the suspension while stirring at room temperature, and the reaction solution was stirred at room temperature for 4 hours and at 50 ° C for 46 hours. The reaction mixture was diluted with ethyl acetate (35 ml), washed with 0.1N hydrochloric acid, water and saturated saline, dried, concentrated and crystallized from methanol to give the title compound (232 mg, 0.438 mmol) as a purple color Obtained as a powder. Ή-丽(200MHz, CDC1 3): 60.85 (3H, t, J = 7.3 Hz), 0.98 (6H, s), 1.35 (2H, q, J = 7.5 Hz), 1.80 (3H, s), 2.27 ( 3H, s), 2.89 (2H, s), 7.61 (2H, d, J = 8.8 Hz), 7.85 (2H, d, J = 8.8 Hz), 9.57 (1H, brs).
元素分析値: C2。HMN303S3Brとして計算値: C, 45.28; H, 4.56; N, 7.92; S, 18.13; Br, 15.06. Elemental analysis: C 2. Calculated for H M N 3 0 3 S 3 Br: C, 45.28; H, 4.56; N, 7.92; S, 18.13; Br, 15.06.
実測値: C, 45.22; H, 4.65; N, 7.94; S, 18.15; Br, 14.84. Found: C, 45.22; H, 4.65; N, 7.94; S, 18.15; Br, 14.84.
mp. 198.5-199.5t: mp. 198.5-199.5t:
実施例 4- 498 Example 4-498
4-ブロモ -N:- [8- (シクロォクチルスルファニル) -5, 6-ジメチル -3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン]ベンゼンスルホンアミド 実施例 3-7 で得た化合物(273 mg, 0.612醒 ol)および炭酸カリウム (169 mg, 1.22腿 ol) の DMF (4.5 ml) の懸濁液に 50°Cで攪拌しながらブロモシクロォク タン (120 mg, 0.612 mmol)を加え、 29 時間攪拌した。 反応中、 ブロモシクロォ クタン (120 mg, 0.612 mmol)を 2 回反応液に追加した。 反応液を実施例 4-164 と同様に後処理して表題化合物 (133 mg, 0.239 mmol)を紫色粉末として得た。 'H-NMR (300MHz, CDC13) : δ 1.50-1.70 (10Η, m), 1.74 (2H, m), 1.81 (3H, q, J= 0.9 Hz), 1.98 (2H, m), 2.27 (3H, q, J= 0.9 Hz), 3.60 (1H, sept, J= 3.6 Hz), 7.62 (2H, dt, J= 8.4, 1.5 Hz), 7.84 (2H, dt, J= 9.0, 2.3 Hz), 9.60 (1H, brs). 元素分析値: C22H26N303S3Brとして計算値: C, 47.48; H, 4.71; N, 7.55; S, 17.28; Br, 14.36. 4-bromo-N : -[8- (cyclooctylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -Tylidene] benzenesulfonamide A suspension of the compound obtained in Example 3-7 (273 mg, 0.612 ol) and potassium carbonate (169 mg, 1.22 ol) in DMF (4.5 ml) at 50 ° C. While stirring, bromocyclooctane (120 mg, 0.612 mmol) was added, and the mixture was stirred for 29 hours. During the reaction, bromocyclooctane (120 mg, 0.612 mmol) was added twice to the reaction solution. The reaction solution was worked up in the same manner as in Example 4-164 to give the title compound (133 mg, 0.239 mmol) as a purple powder. 'H-NMR (300MHz, CDC1 3): δ 1.50-1.70 (10Η, m), 1.74 (2H, m), 1.81 (3H, q, J = 0.9 Hz), 1.98 (2H, m), 2.27 (3H , q, J = 0.9 Hz), 3.60 (1H, sept, J = 3.6 Hz), 7.62 (2H, dt, J = 8.4, 1.5 Hz), 7.84 (2H, dt, J = 9.0, 2.3 Hz), 9.60 . (1H, brs) elemental analysis: C 22 H 26 N 3 0 3 S 3 Br calculated: C, 47.48; H, 4.71 ; N, 7.55; S, 17.28; Br, 14.36.
実測値: C, 47.33; H, 4.57; N, 7.66;. S, 17.16; Br, 14.26. mp. 176.7-181.0°C Found: C, 47.33; H, 4.57; N, 7.66 ;. S, 17.16; Br, 14.26. mp.176.7-181.0 ° C
実施例 4-499 Example 4-499
4_クロ口- N-{8- [ (2, 2 -ジフルォ口 -3-メチルブチル)スルファニル] -5, 6 -ジメチル -3-ォキソ -2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }べンゼ ンスルホンアミド 4_Chloro-N- {8-[(2,2-difluoro-3-methylbutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihid-1H-imidazo [5,1 -c] [1,4] thiazine-1-ylidene} benzenesulfonamide
実施例 3- 6で得た化合物 (298 mg, 0.741龍 ol)および炭酸カリウム (154 mg, 1.11 ol) の DMF (5.0 ml) の懸濁液に 50 で攪拌しながら参考例 104で得た トリフルォロメ夕ンスルホン酸 2, 2-ジフルォロイソペンチル (純度 60%, 270 mg, 0.741讓 ol)を 2回に分けて加え、 12時間攪拌した。 反応液に 1N塩酸 (1.48 ml) を加え、 メタノール—水(1:1, 3 ml)で希釈し、 室温で 20分間攪拌した。 析出物 を濾取し、 メタノール—水 (1:1)およびメタノールで洗浄、 乾燥して表題化合物 (258 mg, 0.508匪 ol)を紫色粉末として得た。 The trifluorome obtained in Reference Example 104 was added to a suspension of the compound obtained in Example 3-6 (298 mg, 0.741 ol) and potassium carbonate (154 mg, 1.11 ol) in DMF (5.0 ml) while stirring at 50. 2,2-Difluoroisopentyl sulfonic acid (purity 60%, 270 mg, 0.741 acetyl) was added in two portions and stirred for 12 hours. 1N Hydrochloric acid (1.48 ml) was added to the reaction solution, diluted with methanol-water (1: 1, 3 ml), and stirred at room temperature for 20 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (258 mg, 0.508 bandol) as a purple powder.
Ή-NMR (200MHz, CDC13) :51.05 (6H, d, J= 6.8 Hz), 1.81 (3H, q, J= 0.8 Hz), 2.18 (2H, sept, J= 6.9 Hz), 2.26 (3H, q, J= 0.8 Hz), 3.39 (2H, t, J= 14.8 Hz), 7.47 (2H, dt, J= 8.4, 1.8 Hz), 7.92 (2H, di, J= 8.8, 2.1 Hz), 9.60 (1H, brs) . Ή-NMR (200MHz, CDC1 3 ): 51.05 (6H, d, J = 6.8 Hz), 1.81 (3H, q, J = 0.8 Hz), 2.18 (2H, sept, J = 6.9 Hz), 2.26 (3H, q, J = 0.8 Hz), 3.39 (2H, t, J = 14.8 Hz), 7.47 (2H, dt, J = 8.4, 1.8 Hz), 7.92 (2H, di, J = 8.8, 2.1 Hz), 9.60 ( 1H, brs).
元素分析値: C19H2。N303S3C1F2として計算値: C, 44.92; H, 3.97; N, 8.27; S, 18.94; CI, 6.98; F, 7.48. Elemental analysis: C 19 H 2. N 3 0 3 S 3 C1F 2 Calculated: C, 44.92; H, 3.97 ; N, 8.27; S, 18.94; CI, 6.98; F, 7.48.
実測値: C, 45.03; H, 3.96; N, 8.27; S, 18.70; CI, 6.93; F, 7.27. Found: C, 45.03; H, 3.96; N, 8.27; S, 18.70; CI, 6.93; F, 7.27.
mp. 165.7-167.5°C mp.165.7-167.5 ° C
実施例 4- 500 Example 4-500
4 -ブロモ -N - {8- [(2, 2-ジフルォロ- 3-メチルブチル)スルファニル] - 5, 6-ジメチル -3 -ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }べンゼ ンスルホンアミド 4-bromo-N- {8-[(2,2-difluoro-3-methylbutyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene} benzenesulfonamide
実施例 3-7 で得た化合物(253 rag, 0.567龍 ol)および炭酸カリウム (118 mg, 0.850誦 ol) の DMF (4.0 ml) の懸濁液に 50。Cで攪拌しながら参考例 104で得た トリフルォロメタンスルホン酸 2, 2-ジフルォロイソペンチル (純度 60%, 206 mg, 0.567mmol)を 2回に分けて加え、 12時間攪拌した。 反応液に 1N塩酸 (1.13ml) を加え、 メタノール一水(1:1, 3 ml)で希釈し、 室温で 20分間攪拌した。 析出物 を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄、 乾燥して表題化合物 (210 mg, 0.380腿 ol)を紫色粉末として得た。 50 in a suspension of the compound obtained in Example 3-7 (253 rag, 0.567 ol) and potassium carbonate (118 mg, 0.850 ol) in DMF (4.0 ml). While stirring with C, 2,2-difluoroisopentyl trifluoromethanesulfonate obtained in Reference Example 104 (purity 60%, 206 mg, 0.567 mmol) was added in two portions, and the mixture was stirred for 12 hours. 1N Hydrochloric acid (1.13 ml) was added to the reaction solution, diluted with methanol / water (1: 1, 3 ml), and stirred at room temperature for 20 minutes. Precipitate Was collected by filtration, washed with methanol-one water (1: 1) and methanol, and dried to give the title compound (210 mg, 0.380 mol) as a purple powder.
Ή-NMR (200MHz, CDC13) : δ 1.05 (6H, d, J= 6.6 Hz), 1.81 (3H, q, J= 0.8 Hz), 2.18 (2H, sept, J= 6.9 Hz), 2.26 (3H, q, J= 0.8 Hz), 3.39 (2H, t, J= 15.0 Hz), 7.63 (2H, d, J= 8.8 Hz), 7.85 (2H, dt, J= 8.4, 1.8 Hz), 9.66 (1H, brs). 元素分析値: Cl9¾。N303S3BrF2として計算値: C, 41.31; H, 3.65; N, 7.61; S, 17.41; Br, 14.46; F, 6.88. Ή-NMR (200MHz, CDC1 3 ): δ 1.05 (6H, d, J = 6.6 Hz), 1.81 (3H, q, J = 0.8 Hz), 2.18 (2H, sept, J = 6.9 Hz), 2.26 (3H , q, J = 0.8 Hz), 3.39 (2H, t, J = 15.0 Hz), 7.63 (2H, d, J = 8.8 Hz), 7.85 (2H, dt, J = 8.4, 1.8 Hz), 9.66 (1H , brs). Elemental analysis: Cl 9 ¾. Calculated for N 3 0 3 S 3 BrF 2 : C, 41.31; H, 3.65; N, 7.61; S, 17.41; Br, 14.46; F, 6.88.
実測値: C, 41.43; H, 3.47; N, 7.49; S, 17.26; Br, 14.02; F, 6.82. Found: C, 41.43; H, 3.47; N, 7.49; S, 17.26; Br, 14.02; F, 6.82.
mp. 164.5-165.0°C mp. 164.5-165.0 ° C
実施例 4-501 Example 4-501
N - [5, 6 -ジメチル- 3-ォキソ -8- [(6, 6, 7, 7, 7-ペンタフルォ口へプチル)スルファニ ル]- 2,3-ジヒドロ- 1H-イミダゾ [5,l-c] [1,4]チアジン -1-ィリデン ]-4-メチルベ ンゼンスルホンアミド N- [5,6-dimethyl-3-oxo-8-[(6,6,7,7,7-pentafluoroheptyl) sulfanyl]-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-1-ylidene] -4-methylbenzenesulfonamide
実施例 3-4で得られた N- (5, 6-ジメチル -3-ォキソ -8-スルファニル -2, 3-ジヒド 口- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン)-4-メチルベンゼンスルホン アミド (300 mg, 0.786匪 ol) と炭酸カリウム (163 mg, 1.18 ramol) の N,N -ジ メチルホルムアミド (5.00 ml) の懸濁液に、 参考例 52で得られたメタンスルホ ン酸 6, 6, 7, 7,7-ペン夕フルォ口へプチルエステル (268 mg, 0.944腿 ol) の N,N - ジメチルホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を 80°Cで 2.5時間 加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去して題記化合物 (88.7 mg, 20%) を暗赤色結晶として得 た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 149- 151°Cの暗赤色結晶を 得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 A suspension of 1-ylidene) -4-methylbenzenesulfonamide (300 mg, 0.786 marl ol) and potassium carbonate (163 mg, 1.18 ramol) in N, N-dimethylformamide (5.00 ml) was prepared according to Example 52. A solution of 6,6,7,7,7-pentafluorofuran heptyl ester (268 mg, 0.944 tmol) of methanesulfonate obtained in the above in N, N-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 ° C for 2.5 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (88.7 mg, 20%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 149-151 ° C.
IR (KBr) V: 3067, 2948, 1742, 1723, 1661, 1618, 1561 cm—1. IR (KBr) V: 3067, 2948, 1742, 1723, 1661, 1618, 1561 cm— 1 .
Ή-NMR (CDC13) : 51.43-1.67 (4H, m), 1.67-1.80 (2H, m), 1.79 (3H, d, 1=0.9 Ή-NMR (CDC1 3): 51.43-1.67 (4H, m), 1.67-1.80 (2H, m), 1.79 (3H, d, 1 = 0.9
Hz), 2.03 (2H, heptet, J=8.4 Hz), 2.27 (3H, d, J-0.9 Hz), 2.42 (3H, s), 2.95Hz), 2.03 (2H, heptet, J = 8.4 Hz), 2.27 (3H, d, J-0.9 Hz), 2.42 (3H, s), 2.95
(2H, t, J=7.5 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). (2H, t, J = 7.5 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C22H24N303S3F3として 計算値 (¾) : C, 46.39; H, 4.25; , 7.38; S, 16.89. Elementary analysis: as C 22 H 24 N 3 0 3 S 3 F 3 Calculated value (¾): C, 46.39; H, 4.25;, 7.38; S, 16.89.
実測値 (¾) : C, 46.33; H, 4.18; N, 7.56; S, 16.94. Found (値): C, 46.33; H, 4.18; N, 7.56; S, 16.94.
実施例 4-5Q2 ' N - [5, 6-ジメチル- 3-ォキソ - 8 - [ (3, 3, 4, 4,4-ペン夕フルォロブチル)スルファニ ル]- 2, 3-ジヒドロ -1Η-イミダゾ [5,卜 c] [1,4]チアジン -1-ィリデン ]-4-メチルベ ンゼンスルホンアミド Example 4-5Q2'N- [5,6-Dimethyl-3-oxo-8-[(3,3,4,4,4-pentafluorofluorobutyl) sulfanyl] -2,3-dihydro-1Η-imidazo [5, tric] [1,4] thiazine-1-ylidene] -4-methylbenzenesulfonamide
実施例 3-4で得られた N- (5, 6-ジメチル -3-ォキソ - 8-スルファニル -2, 3 -ジヒド 口- 1H-イミダゾ [5,1 - c] [1,4]チアジン - 1-ィリデン)-4-メチルベンゼンスルホン アミド (300 mg, 0.786匪 ol) と炭酸カリウム (163 mg, 1.18 mmol) の Ν,Ν-ジ メチルホルムアミド (5.00 ml) の懸濁液に'、 4-ョ一ド- 1,1,2,2,2_ペンタフルォ ロブタン (267 mg, 0.944 mmol) の N, N-ジメチルホルムアミド (1.00 ml) 溶液 を加えた。反応混合物を 80°Cで 1時間加熱した後、 1N塩酸を加えて酢酸ェチルで 抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (305 mg, 74¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 182_184°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihidido-1H-imidazo [5,1-c] [1,4] thiazine obtained in Example 3-4- To a suspension of 1-ylidene) -4-methylbenzenesulfonamide (300 mg, 0.786 marl) and potassium carbonate (163 mg, 1.18 mmol) in Ν, Ν-dimethylformamide (5.00 ml) A solution of 1,1,2,2,2-pentafluorobutane (267 mg, 0.944 mmol) in N, N-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 ° C. for 1 hour, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (305 mg, 74¾) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 182-184 ° C.
IR (KBr) V 3235, 2982, 2930, 1759, 1665, 1626, 1566 cm-1. IR (KBr) V 3235, 2982, 2930, 1759, 1665, 1626, 1566 cm- 1 .
Ή-NMR (CDC13) : 51.81 (3H, d, J=l.2 Hz), 2.27 (3H, d, J=1.2 Hz), 2.34-2.60 (2H, m), 2.43 (3H, s), 3.14 (2H, t, J=8.6 Hz), 7.30 (2H, d, J=8.0 Hz), 7.83 (2H, d, J=8.0 Hz), 9.63 (1H, bs). Ή-NMR (CDC1 3): 51.81 (3H, d, J = l.2 Hz), 2.27 (3H, d, J = 1.2 Hz), 2.34-2.60 (2H, m), 2.43 (3H, s), 3.14 (2H, t, J = 8.6 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.63 (1H, bs).
元素分析値: C19H16N303S3F3として Elemental analysis: as C 19 H 16 N 3 0 3 S 3 F 3
計算値 (¾) : C, 43.42; H, 3.07; N, 8.00; S, 18.30. Calculated value (¾): C, 43.42; H, 3.07; N, 8.00; S, 18.30.
実測値 (¾) : C, 43.16; H, 3.37; N, 7.83; S, 18.35. Found (値): C, 43.16; H, 3.37; N, 7.83; S, 18.35.
実施例 4- 503 Example 4-503
N - {8- [(2, 2-ジメチルペンチル)スルファニル ]-5, 6 -ジメチル -3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン } -4-メチルべンゼンスルホン アミド ' N- {8-[(2,2-dimethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1-ylidene} -4-methylbenzenesulfonamide ''
2, 2-ジメチルバレリル酸 (純度 95%, 7.50 g, 54.7 mmol)と 濃塩酸 (9.12 ml, 0.109mol) のメタノール (80ml) 溶液を室温で 27時間攪拌した。 反応液に炭酸 水素ナトリウムを加えて中和後、 酢酸ェチル抽出により精製し、 2, 2-ジメチルバ レリル酸メチルの淡黄色油状物を得た。得られた 2, 2-ジメチルバレリル酸メチル の ジェチルエーテル a50 ml)溶液に (TCで攪拌しながら水素化リチウムアルミ二 ゥム(純度 80, 2.59 g, 54.7匪01) の粉末を数回に分けて加え、 0°Cで 4時間攪 拌した。 ジェチルェ一テルにより抽出し、 乾燥、 濃縮乾固して 2, 2-ジメチルペン 夕ノール (純度 87, 89¾, 4.85 g, 37.1 mmol)の無色油状物を得た。 A solution of 2,2-dimethylvaleric acid (purity 95%, 7.50 g, 54.7 mmol) and concentrated hydrochloric acid (9.12 ml, 0.109 mol) in methanol (80 ml) was stirred at room temperature for 27 hours. The reaction mixture was neutralized with sodium bicarbonate, and purified by extraction with ethyl acetate. A pale yellow oil of methyl rerylate was obtained. A powder of lithium aluminum hydride (purity 80, 2.59 g, 54.7 bandages 01) was added several times to the obtained solution of methyl 2,2-dimethylvalerate in getyl ether a50 ml while stirring with TC. The mixture was stirred for 4 hours at 0 ° C. Extracted with getyl ether, dried and concentrated to dryness to give 2,2-dimethylpentanol (purity 87, 89¾, 4.85 g, 37.1 mmol). A colorless oil was obtained.
Ή-NMR (200MHz, CDC13) : δ 0.87 (6H, s), 0.91 (3H, t, J= 7.5 Hz), 1.23 (2H, m), 1.28 (2H, m), 3.32 (2H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.87 (6H, s), 0.91 (3H, t, J = 7.5 Hz), 1.23 (2H, m), 1.28 (2H, m), 3.32 (2H, brs) .
実施例 3- 4で得た化合物 (461 mg, 1.21 mmol)、 上記で得た 2, 2-ジメチルぺ ン夕ノール (189 mg, 1.45 mmol) およびトリフエニルホスフィン(457 mg, 1.69 腿 ol)の THF (9.0 ml)懸濁液に室温で攪拌しながら DEAD (0.280 ml, 1.69 mmol) を滴下し、反応液を室温で 12時間、 5(TC で 33時間攪拌した。反応液に酢酸ェチ ル(35 ml)を加えて希釈し、 0.2N塩酸、 0.1 塩酸、水および飽和食塩水で洗浄し、 乾燥、 濃縮後、 メタノールで希釈した。 析出物を濾取し、 メタノール洗浄後、 乾 燥して表題化合物(313.mg, 0.652腿 ol)を紫色粉末として得た。 Of the compound (461 mg, 1.21 mmol) obtained in Example 3-4, 2,2-dimethylphenol (189 mg, 1.45 mmol) and triphenylphosphine (457 mg, 1.69 tmol) obtained above. DEAD (0.280 ml, 1.69 mmol) was added dropwise to the suspension of THF (9.0 ml) with stirring at room temperature, and the reaction mixture was stirred at room temperature for 12 hours and 5 (TC for 33 hours. (35 ml), diluted with 0.2 N hydrochloric acid, 0.1 hydrochloric acid, water and saturated saline, dried, concentrated, and diluted with methanol The precipitate was collected by filtration, washed with methanol, and dried. The title compound (313.mg, 0.652 t) was obtained as a purple powder.
'H-NMR (200MHz, CDC13) : (50.90 (3H, brt, J= 6. Hz), 0.98 (6H, s), 1.27 (4H, brd, J= 3.4 Hz), 1.78 (3H, brs), 2.26 (3H, s), 2.41 (3H, s), 2.88 (2H, s), 7.27 (2H, d, J= 8.6 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.59 (1H, brs). 'H-NMR (200MHz, CDC1 3): (50.90 (3H, brt, J = 6. Hz), 0.98 (6H, s), 1.27 (4H, brd, J = 3.4 Hz), 1.78 (3H, brs) , 2.26 (3H, s), 2.41 (3H, s), 2.88 (2H, s), 7.27 (2H, d, J = 8.6 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.59 (1H, brs).
元素分析値: C22H29N303S3として計算値: C, 55.09; H, 6.09; N, 8.76; S, 20.05. 実測値: C, 55.10; H, 6.20; N, 8.65; S, 19.86. Elemental analysis: C 22 H 29 N 3 0 3 S 3 Calculated:. C, 55.09; H, 6.09; N, 8.76; S, 20.05 Found: C, 55.10; H, 6.20 ; N, 8.65; S , 19.86.
mp. 172.0-172.5°C mp. 172.0-172.5 ° C
実施例 4- 504 Example 4-504
4-ブ口モ- N- {8- [ (2, 2-ジメチルぺンチル)スルファニル] -5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [ 4]チアジン-卜ィリデン }ベンゼンスルホン アミド 4-Buguchi-N- {8-[(2,2-dimethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [4] thiazine-triylidene} benzenesulfonamide
実施例 3-7 で得た化合物 (435 mg, 0.975 mmol), 2, 2-ジメチルペン夕ノール (140 rag, 1.07 mmol) およびトリフエニルホスフィン(343 mg, 1.27麵 ol)の THF (8.7 ml)の懸濁液に室温で攪拌しながら DEAD (0.210 ml, 1.27 iraiol)を滴下し、 反応液を 50°C で 35時間攪拌した。反応液に酢酸ェチル(35 ml)を加えて希釈し、 0.1 塩酸、水および飽和食塩水で洗浄し、乾燥、濃縮後、メタノールで希釈した。 析出物を濾取し、 メタノール洗浄後、 酢酸ェチルおよびジェチルエーテルから結 晶化して表題化合物 (249 mg, 0.457匪 ol)を濃紫色粉末として得た。 THF (8.7 ml) of the compound obtained in Example 3-7 (435 mg, 0.975 mmol), 2,2-dimethylpentanol (140 rag, 1.07 mmol) and triphenylphosphine (343 mg, 1.27 mol) DEAD (0.210 ml, 1.27 iraiol) was added dropwise to the suspension at room temperature with stirring, and the reaction solution was stirred at 50 ° C for 35 hours. The reaction mixture was diluted with ethyl acetate (35 ml), washed with 0.1 hydrochloric acid, water and saturated saline, dried, concentrated and diluted with methanol. The precipitate was collected by filtration, washed with methanol, and crystallized from ethyl acetate and getyl ether to give the title compound (249 mg, 0.457 methylol) as a dark purple powder.
Ή-NMR (300MHz, CDC13) : 50.91 (3H, m), 0.99 (6H, s), 1.27 (4H, brd, J= 3.6 Hz), 1.79 (3H, q, J= 0.9 Hz), 2.27 (3H, q, J= 0.9 Hz), 2.89 (2H, s), 7.61 (2H, dt, J= 8.7, 1.8 Hz), 7.85 (2H, dt, J- 9.0, 2.1 Hz), 9.56 (1H, brs). 元素分析値: C21H26N303S3Brとして計算値: C, 46.32; H, 4.81; , 7.72; S, 17.67; Br, 14.67. Ή-NMR (300MHz, CDC1 3 ): 50.91 (3H, m), 0.99 (6H, s), 1.27 (4H, brd, J = 3.6 Hz), 1.79 (3H, q, J = 0.9 Hz), 2.27 ( 3H, q, J = 0.9 Hz), 2.89 (2H, s), 7.61 (2H, dt, J = 8.7, 1.8 Hz), 7.85 (2H, dt, J- 9.0, 2.1 Hz), 9.56 (1H, brs .) elemental analysis: C 21 H 26 N 3 0 3 S 3 Br calculated: C, 46.32; H, 4.81 ;, 7.72; S, 17.67; Br, 14.67.
実測値: C, 46.41; H, 4.75; N, 7.71; S, 17.63; Br, 14.57. Found: C, 46.41; H, 4.75; N, 7.71; S, 17.63; Br, 14.57.
mp. 182.0-184.5 mp. 182.0-184.5
実施例 4- 505 Example 4-505
4 -ブロモ -Ν- {8- [(6 -ヒドロキシへキシル)スルファニル] -5, 6-ジメチル- 3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン }ベンゼンスルホン アミド 4-bromo-Ν- {8-[(6-hydroxyhexyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] Thiazine-1-ylidene} benzenesulfonamide
実施例 3- 7で得た化合物 (1.09g, 2.43 mmol)、 6-ョ一ド- 1-へキサノール(671 mg, 2.68顏 ol)および炭酸カリウム (504 mg, 3.65 mmol) の DMF (18 ml) の懸 濁液を 60°C で 100分間攪拌した。反応液に 1N塩酸 (4.9 ml)を加え、水 (10 ml) で希釈し、 室温で攪拌した。 析出物を濾取し、 メタノール—水 (1:1)およびメタ ノールで洗浄後、 乾燥して表題化合物 α.15 g, 2.10 mmol)を赤紫色粉末として得 た。 DMF (18 ml) of the compound obtained in Example 3-7 (1.09 g, 2.43 mmol), 6-node-1-hexanol (671 mg, 2.68 mmol) and potassium carbonate (504 mg, 3.65 mmol) ) Was stirred at 60 ° C for 100 minutes. 1N Hydrochloric acid (4.9 ml) was added to the reaction solution, diluted with water (10 ml), and stirred at room temperature. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (α.15 g, 2.10 mmol) as a red-purple powder.
Ή-NMR (300MHz, CDCI3) : δ 1.43 (4Η, m), 1.57 (2H, m), 1.70 (2H, brquint, J= 7.5 Hz), 1.80 (3H, q, J- 0.9 Hz), 2.28 (3H, q, J= 0.9 Hz), 2.95 (2H, t, J= 7.5 Hz), 3.66 (2H, t, J= 6.3 Hz), 7.63 (2H, dt, J= 8.7, 2.0 Hz), 7.83 (2H, dt, J= 9.0, 2.3 Hz), 9.61 (1H, brs). Ή-NMR (300 MHz, CDCI 3 ): δ 1.43 (4Η, m), 1.57 (2H, m), 1.70 (2H, brquint, J = 7.5 Hz), 1.80 (3H, q, J- 0.9 Hz), 2.28 (3H, q, J = 0.9 Hz), 2.95 (2H, t, J = 7.5 Hz), 3.66 (2H, t, J = 6.3 Hz), 7.63 (2H, dt, J = 8.7, 2.0 Hz), 7.83 (2H, dt, J = 9.0, 2.3 Hz), 9.61 (1H, brs).
元素分析値: C20H24N304S3Brとして計算値: C, 43.95; H, 4.43; N, 7.69; S, 17.60; Br, 14.62 (%) · 実測値: C, 43.90; H, 4.32; N, 7.71; S, 17.54; Br, 14.68 (%) . Elemental analysis: C 20 H 24 N 3 0 4 S 3 Br Calculated: C, 43.95; H, 4.43 ; N, 7.69; S, 17.60; Br, 14.62 (%) · Found: C, 43.90; H , 4.32; N, 7.71; S, 17.54; Br, 14.68 (%).
融点 132.5-134.5°C 132.5-134.5 ° C
実施例 4-506 Example 4-506
N - [5 , 6-ジメチル -3-ォキソ -8- [ (6, 6, 6-トリフルォ口へキシル)スルファニ ル]- 2,3-ジヒドロ _1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1 -ィリデン ]_4-メチルベ ンゼンスルホンアミド N- [5,6-dimethyl-3-oxo-8-[(6,6,6-trifluorohexyl) sulfani ]-2,3-Dihydro_1H-imidazo [5, tric] [1, 4] thiazine-1-ylidene] _4-methylbenzenesulfonamide
実施例 3-4で得られた N- (5, 6-ジメチル- 3 -ォキソ -8-スルファ二ル- 2, 3-ジヒド 口- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン)-4-メチルベンゼンスルホン アミ ド (1.00 g, 2.62腿01) と炭酸カリウム (543 mg, 3.93匪01) の Ν,Ν-ジメ チルホルムアミド (15.0 ml) の懸濁液に、 参考例 56で得られたメタンスルホン 酸 6,6, 6 -トリフルォ口へキシルエステル (737 mg, 3.15 mmol) の N,N-ジメチル ホルムアミド (5.00 ml)溶液を加えた。反応混合物を 80°Cで 4時間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (958 mg, 70%) を暗赤色結晶として得た。 酢酸ェ チル-ジェチルエーテルから再結晶して融点 140- 142°Cの暗赤色結晶を得た。 IR (KBr) ソ : 3139, 3063, 2944, 1744, 1728, 1661, 1617, 1559 cm"1. N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrone-1H-imidazo obtained in Example 3-4 [5,1-c] [1,4] To a suspension of thiazine-triylidene) -4-methylbenzenesulfonamide (1.00 g, 2.62 thigh 01) and potassium carbonate (543 mg, 3.93 marauder 01) in Ν, Ν-dimethylformamide (15.0 ml), A solution of methanesulfonic acid 6,6,6-trifluorohexyl ester (737 mg, 3.15 mmol) obtained in Reference Example 56 in N, N-dimethylformamide (5.00 ml) was added. After heating the reaction mixture at 80 ° C for 4 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (958 mg, 70%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 140-142 ° C. IR (KBr) Seo: 3139, 3063, 2944, 1744, 1728, 1661, 1617, 1559 cm " 1 .
Ή一 NMR (CDC13) : ά 1.40-1.84 (6H, m), 1.78 (3H, s), 1.98-2.18 (2H, ra), 2.26 (3H, s), 2.42 (3H, s), 2.94 (2H, t, J=7.4 Hz), 7.29.(2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.4 Hz), 9.62 (1H, bs). Ή one NMR (CDC1 3): ά 1.40-1.84 (6H, m), 1.78 (3H, s), 1.98-2.18 (2H, ra), 2.26 (3H, s), 2.42 (3H, s), 2.94 ( 2H, t, J = 7.4 Hz), 7.29. (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C21H24N303S3F3として Elementary analysis: as C 21 H 24 N 3 0 3 S 3 F 3
計算値 ) : C, 48.54; H, 4.66; N, 8.09; S, 18.51. Calculated)): C, 48.54; H, 4.66; N, 8.09; S, 18.51.
実測値 (%) : C, 48.35; H, 4.64; N, 8.37; S, 18.36. Found (%): C, 48.35; H, 4.64; N, 8.37; S, 18.36.
実施例 4- 507 Example 4-507
N - {8-[ (7-ヒドロキシへプチル)スルファニル ]-5, 6-ジメチル- 3-ォキソ - 2, 3-ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィ Vデン } -4-メチルベンゼンスルホン アミド N- {8-[(7-hydroxyheptyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1 -Vene} -4-Methylbenzenesulfonamide
実施例 3-4で得た化合物(1.11 g, 2.91匪 ol)および炭酸カリウム (603 mg, 4.36 mmol) の DMF (16ml) の懸濁液に 60°Cで攪拌しながら 7 -ブロモ -1 -ヘプ夕ノール (681 rag, 3.49 mmol)を 3回に分けて 30分間隔で加え、 反応液を 100分間攪拌し た。 反応液に 1N塩酸 (6.4 ml)を加え、 水 (10 ml)で希釈し、 室温で 30分間攪 拌した。 析出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄後、 酢 酸ェチルおよびジェチルェ一テルから結晶化して表題化合物 (998 mg, 2.01 腿 ol)を赤色粉末として得た。 Ή-NMR (300MHz, CDC13) : 51.37 (6H, m), 1.60 (2H, m), 1.65 (2H, m), 1.78 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s), 2.93 (2H, t, J= 7.5 Hz), 3.66 (2H, brt, J= 7.0 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). 元素分析値: C22H29N304S3 -0.2H20として To a suspension of the compound obtained in Example 3-4 (1.11 g, 2.91 marl ol) and potassium carbonate (603 mg, 4.36 mmol) in DMF (16 ml) was added 7-bromo-1- while stirring at 60 ° C. Heptanol (681 rag, 3.49 mmol) was added in three portions at 30 minute intervals, and the reaction solution was stirred for 100 minutes. 1N Hydrochloric acid (6.4 ml) was added to the reaction solution, diluted with water (10 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and crystallized from ethyl acetate and getyl ether to give the title compound (998 mg, 2.01 mol) as a red powder. Ή-NMR (300MHz, CDC1 3 ): 51.37 (6H, m), 1.60 (2H, m), 1.65 (2H, m), 1.78 (3H, brs), 2.26 (3H, brs), 2.42 (3H, s ), 2.93 (2H, t, J = 7.5 Hz), 3.66 (2H, brt, J = 7.0 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, brs) elemental analysis:. as C 22 H 29 N 3 0 4 S 3 -0.2H 2 0
計算値: C, 52.92; H, 5.94; N, 8.42; S, 19.27 (%) . Calculated: C, 52.92; H, 5.94; N, 8.42; S, 19.27 (%).
実測値: C, 52.89; H, 5.87; N, 8.33; S, 19.53 (%) . Found: C, 52.89; H, 5.87; N, 8.33; S, 19.53 (%).
即. 118.0-119.0°C Immediate. 118.0-119.0 ° C
実施例 4- 508 Example 4-508
N- [5, 6 -ジメチル- 3-ォキソ -8- [ (5_フルォロペンチル)スルファニル] -2, 3-ジヒド 口- 1H -イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン ]-4-メチルベンゼンスルホン アミド N- [5,6-Dimethyl-3-oxo-8-[(5_fluoropentyl) sulfanyl] -2,3-dihydrone-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene ] -4-Methylbenzenesulfone amide
例 1 Example 1
実施例 3- で得られた N- (5, 6-ジメチル -3-ォキソ -8 -スルファニル- 2, 3-ジヒドロN- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro obtained in Example 3-
- 1H-イミダゾ [5, 1-c] [1,4]チアジン-トイリデン) - 4 -メチルベンゼンスルホンァ ミド (1.00 g, 2.62匪01) と炭酸カリウム (543 mg, 3.93 mmol) の N,N_ジメチ ルホルムアミド(15.0 ml)の懸濁液に、参考例 60で得られたメタンスルホン酸 5 - フルォロペンチルエステル (579 mg, 3.15 mmol) の N, N-ジメチルホルムアミド (5.00 ml) 溶液を加えた。 反応混合物を 80°Cで 2時間加熱した後、 1N塩酸を加 えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留 去して題記化合物 (1.60 g, 65%) を暗赤色結晶として得た。 酢酸ェチル-ジェチ ルェ一テルから再結晶して融点 167- 169°Cの暗赤色結晶を得た。 -1H-imidazo [5, 1-c] [1,4] thiazine-toylidene)-N, N of 4-methylbenzenesulfonamide (1.00 g, 2.62 band01) and potassium carbonate (543 mg, 3.93 mmol) N-N-dimethylformamide (5.00 ml) solution of methanesulfonic acid 5-fluoropentyl ester (579 mg, 3.15 mmol) obtained in Reference Example 60 in a suspension of _dimethylformamide (15.0 ml) Was added. After heating the reaction mixture at 80 ° C for 2 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was distilled off under reduced pressure to give the title compound (1.60 g, 65%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl acetate gave dark red crystals with a melting point of 167-169 ° C.
例 2 Example 2
実施例 3- 4で得られた N- (2, 3-ジヒドロ- 5, 6-ジメチル- 3-ォキソ -8-スルファニ ル- 1H-イミダゾ [5,l-c] [1,4]チアジン- 1-イリデン) -4-メチルベンゼンスルホン アミ ド (4.00 g, 10.5廳 ol) と炭酸カリウム (2.17 g, 15.7腿 ol) の Ν,Ν-ジメ チルホルムアミド (75.0 ml) の懸濁液に、 参考例 60で得られたメタンスルホン 酸 5-フルォロペンチルエステル (2.32 g, 12.6 mmol) の N, N-ジメチルホルムァ ミド (5.00 ml) 溶液を加えた。 反応混合物を 80°Cで 2.5時間加熱した後、 1N塩 酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧 下に留去して題記化合物 (3.44 g, 70¾) を暗赤色結晶として得た。 N- (2,3-dihydro-5,6-dimethyl-3-oxo-8-sulfanyl-1H-imidazo [5, lc] [1,4] thiazine-1-) obtained in Example 3-4 To a suspension of (ylidene) -4-methylbenzenesulfonamide (4.00 g, 10.5 ol) and potassium carbonate (2.17 g, 15.7 t ol) in Ν, 2.1-dimethylformamide (75.0 ml), refer to Example 60. A solution of the methanesulfonic acid 5-fluoropentyl ester (2.32 g, 12.6 mmol) obtained in the above in N, N-dimethylformamide (5.00 ml) was added. After heating the reaction mixture at 80 ° C. for 2.5 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent under reduced The residue was evaporated under reduced pressure to give the title compound (3.44 g, 70%) as dark red crystals.
IR (KBr) V: 3243, 3054, 2924, 1728, 1661, 1615, 1557 cm"1. IR (KBr) V: 3243, 3054, 2924, 1728, 1661, 1615, 1557 cm " 1 .
Ή-NMR (CDC13) : (51.81 (3H, d, J=1.2 Hz), 2.27 (3H, d, J=1.2 Hz), 2.34-2.60 (2H, m), 2.43 (3H, s), 3.14 (2H, t, J=8.6 Hz), 7.30 (2H, d, J=8.0 Hz), 7.83 (2H, d, J=8.0 Hz), 9.63 (1H, bs). Ή-NMR (CDC1 3): (51.81 (3H, d, J = 1.2 Hz), 2.27 (3H, d, J = 1.2 Hz), 2.34-2.60 (2H, m), 2.43 (3H, s), 3.14 (2H, t, J = 8.6 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.63 (1H, bs).
元素分析値: C19H16N303S3F3として Elemental analysis: as C 19 H 16 N 3 0 3 S 3 F 3
計算値 (¾) : C, 43.42; H, 3.07; N, 8.00; S, 18.30. Calculated value (¾): C, 43.42; H, 3.07; N, 8.00; S, 18.30.
実測値 (%) : C, 43.16; H, 3.37; N, 7.83; S, 18.35. Found (%): C, 43.16; H, 3.37; N, 7.83; S, 18.35.
別方法 Another way
実施例 4- 520で得られた N- [5, 6-ジメチル- 3-ォキソ -8- [(5-ヒドロキシペンチル)ス ルファニル] -2, 3-ジヒド口- 1H -ィミダゾ [5, 1-C] [1 , ]チアジン-トイリデン] -4 -メ チルベンゼンスルホンアミド (3.00 g, 6.41 mmol) のジクロロメタン (150 ml) 溶液に、 ジェチルァミノ硫黄トリフルオリド (1.33ml, 9.62腿01) のジクロロメ タン (30.0ml) 溶液を- 70°Cでゆっくりと滴下した。 反応混合物を - 7(T で 3.5時間 かき混ぜた後、氷冷下飽和炭酸水素ナトリゥム水溶液を加えてジクロロメタンで抽 出した。抽出液を水洗、乾燥 (MgS04)後、溶媒を減圧下に留去して題記化合物(3.22 g, ca.100¾) を暗赤色結晶として得た。 N- [5,6-Dimethyl-3-oxo-8-[(5-hydroxypentyl) sulfanyl] -2,3-dihydrido-1H-imidazo [5,1-] obtained in Example 4-520 C] [1,] Thiazine-toylidene] -4-methylbutylsulfonamide (3.00 g, 6.41 mmol) in dichloromethane (150 ml) was mixed with dimethylaminosulfur trifluoride (1.33 ml, 9.62 t01) in dichloromethane ( 30.0 ml) The solution was slowly added dropwise at -70 ° C. The reaction mixture - 7 (After stirring 3.5 hours at T, distillation was added under ice cooling saturated sodium hydrogen Natoriumu aqueous issued extracted with dichloromethane extract was washed with water, dried (MgSO 4), the solvent under reduced pressure. Thus, the title compound (3.22 g, ca. 100 °) was obtained as dark red crystals.
実施例 4-509 Example 4-509
N - {8- [(4-フルォロ- 4-メチルペンチル)スルファニル ]-5, 6-ジメチル- 3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン-卜イリデン }- 4-メチルベンゼ ンスルホンアミド N- {8-[(4-Fluoro-4-methylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine -Triylidene}-4-methylbenzenesulfonamide
実施例 3-4で得た化合物(601 mg, 1.58腿 ol)および炭酸カリウム (327 mg, 2.36 mmol) の DMF (lOml) の懸濁液に 60°Cで攪拌しながら参考例 37で得た 4-フルォ ロ- 4-メチルペンチルメシレート (344 mg, 1.73 mmol)を加え、 反応液を 10時間 攪拌した。 反応液に 1N塩酸 (3.5ml)を加え、 水 (5 ml)で希釈し、 室温で 30分 間攪拌した。析出物を濾取し、メタノール一水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物(593 mg, 1.23 mmol)を紫色粉末として得た。 A suspension of the compound obtained in Example 3-4 (601 mg, 1.58 mol) and potassium carbonate (327 mg, 2.36 mmol) in DMF (10 ml) was obtained in Reference Example 37 while stirring at 60 ° C. 4-Fluoro-4-methylpentyl mesylate (344 mg, 1.73 mmol) was added, and the reaction solution was stirred for 10 hours. 1N Hydrochloric acid (3.5 ml) was added to the reaction solution, diluted with water (5 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (593 mg, 1.23 mmol) as a purple powder.
Ή-NMR (300MHz, CDC13) : 51.32 (3H, s), 1.39 (3H, s), 1.69 (2H, m), 1.76 (2H, m), 1.79 (3H, q, J= 1.2 Hz), 2.27 (3H, q, J= 0.9 Hz), 2.42 (3H, s), 2.96 (2H, ί, J= 7.1 Hz), 7.29 (2H, d, J= 8.1 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.65 (1H, brs) . Ή-NMR (300MHz, CDC1 3 ): 51.32 (3H, s), 1.39 (3H, s), 1.69 (2H, m), 1.76 (2H, m), 1.79 (3H, q, J = 1.2 Hz), 2.27 (3H, q, J = 0.9 Hz), 2.42 (3H, s), 2.96 (2H, ί, J = 7.1 Hz), 7.29 (2H, d, J = 8.1 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C21H26N303S3F ·0.2¾0として Elemental analysis: C 21 H 26 N 3 0 3 S 3 F · 0.2¾0
計算値: C, 51.77; H, 5.46; N, 8.62; S, 19.74; F, 3.90 (%) . Calculated: C, 51.77; H, 5.46; N, 8.62; S, 19.74; F, 3.90 (%).
実測値: C, 51.79; H, 5.31; , 8.59; S, 19.65; F, 3.57 (%) . Found: C, 51.79; H, 5.31 ;, 8.59; S, 19.65; F, 3.57 (%).
mp. 173.0-174.0°C mp. 173.0-174.0 ° C
実施例 4- 510 Example 4-510
4- kブロモ - N- {8- [ (4-フルォロ _4 -メチルぺンチル)スルファニル] -5, 6-ジメチル- 3 - ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン }ベンゼンス ルホンアミド 4-k-bromo - N- {8- [(4- Furuoro _4 - methyl-pentyl) sulfanyl] -5, 6-dimethyl - 3 - Okiso -2, 3-dihydro - 1H-imidazo [5, Bok c] [ 1,4] thiazine-triylidene} benzenesulfonamide
実施例 3-7で得た化合物(502 mg, 1.12 mmol)および炭酸カリウム (233 mg, 1.69 mmol) の DMF (8 ml) の懸濁液に 60°Cで攪拌しながら参考例 37で得た 4- フルォロ- 4-メチルペンチルメシレ一ト(245 mg, 1.24 mmol)を加え、 反応液を 7.5 時間攪拌した。 反応液に 1N塩酸 (2.5 ml)を加え、 水(5 ml)およびメタノール(5 ml)で希釈し、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール—水 (1:1) およびメタノールで洗浄後、 乾燥して表題化合物(391 mg, 0.713 mmol)を紫色粉 末として得た。 A suspension of the compound (502 mg, 1.12 mmol) obtained in Example 3-7 and potassium carbonate (233 mg, 1.69 mmol) in DMF (8 ml) was obtained in Reference Example 37 while stirring at 60 ° C at 60 ° C. 4-Fluoro-4-methylpentyl mesylate (245 mg, 1.24 mmol) was added, and the reaction solution was stirred for 7.5 hours. 1N Hydrochloric acid (2.5 ml) was added to the reaction solution, diluted with water (5 ml) and methanol (5 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (391 mg, 0.713 mmol) as a purple powder.
^-NMR (300MHz, CDC13) : 51.33 (3H, s), 1.40 (3H, s), 1.69 (2H, m), 1.77 (2H, m), 1.80 (3H, q, J= 1.2 Hz), 2.28 (3H, q, J= 0.9 Hz), 2.98 (2H, t, J= 7.2 Hz), 7.63 (2H, dt, J= 8.7, 2.0 Hz), 7.84 (2H, dt, J= 8.7, 2.1 Hz), 9.64 (1H, brs). ^ -NMR (300MHz, CDC1 3) : 51.33 (3H, s), 1.40 (3H, s), 1.69 (2H, m), 1.77 (2H, m), 1.80 (3H, q, J = 1.2 Hz), 2.28 (3H, q, J = 0.9 Hz), 2.98 (2H, t, J = 7.2 Hz), 7.63 (2H, dt, J = 8.7, 2.0 Hz), 7.84 (2H, dt, J = 8.7, 2.1 Hz) ), 9.64 (1H, brs).
元素分析値: C2。H23N303S3BrFとして Elemental analysis: C 2. H 23 N 3 0 3 S 3 As BrF
計算値: C, 43.79; H, 4.23; N, 7.66; S, 17.54; Br, 14.57; F, 3.46 (%) . 実測値: C, 43.68; H, 4.15; N, 7.79; S, 17.41; Br, 14.30; F, 3.39 (%) · mp. 181.0-181.5t: Calculated: C, 43.79; H, 4.23; N, 7.66; S, 17.54; Br, 14.57; F, 3.46 (%). Found: C, 43.68; H, 4.15; N, 7.79; S, 17.41; Br. , 14.30; F, 3.39 (%) mp.181.0-181.5t:
実施例 4- 511 Example 4-511
N-{5, 6 -ジメチル- 8 - [ (4-メチル -3-ペンテニル)スルファニル] -3 -ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン } - 4 - アミド 実施例 3-4で得た化合物(483 mg, 1.27匪 ol)および炭酸カリウム (262 mg, 1.90 腿 ol) の DMF (8 ml) の懸濁液に 60°Cで攪拌しながら 4-メチル -3-ペンテニルメ シレ一ト(0.27 g, 1.52腦 ol)を加え、反応液を 7.5時間攪拌した。反応液に 1N塩 酸 (2.8 ml)を加え、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物 (412 mg, 0.889匪 ol)を 紫色粉末として得た。 N- {5,6-Dimethyl-8-[(4-methyl-3-pentenyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene}-4-amide A suspension of the compound obtained in Example 3-4 (483 mg, 1.27 ol) and potassium carbonate (262 mg, 1.90 ol) in DMF (8 ml) was stirred at 60 ° C with 4-methyl- 3-Pentenyl mesylate (0.27 g, 1.52 mol) was added, and the reaction solution was stirred for 7.5 hours. 1N Hydrochloric acid (2.8 ml) was added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (412 mg, 0.889 bandol) as a purple powder.
Ή-NMR (300MHz, CDC13) : (51.62 (3H, s), 1.71 (3H, s), 1.78 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 0.9 Hz), 2.35 (2H, q, J= 7.1 Hz), 2.42 (3H, s), 2.93 (2H, t, J= 7.2 Hz), 5.10 (1H, tt, J= 7.2, 1.4 Hz), 7.25 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.1 Hz), 9.63 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): (51.62 (3H, s), 1.71 (3H, s), 1.78 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 0.9 Hz), 2.35 (2H, q, J = 7.1 Hz), 2.42 (3H, s), 2.93 (2H, t, J = 7.2 Hz), 5.10 (1H, tt, J = 7.2, 1.4 Hz), 7.25 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.1 Hz), 9.63 (1H, brs).
元素分析値: C2lH25N303S3として計算値: C, 54.40; H, 5.43; N, 9.06; S, 20.75Elemental analysis: C 2l H 25 N 3 0 3 S 3 Calculated: C, 54.40; H, 5.43 ; N, 9.06; S, 20.75
(%) 、 実測値: C, 54.15; H, 5.43; N, 9.24; S, 20.76 (%) . (%), Found: C, 54.15; H, 5.43; N, 9.24; S, 20.76 (%).
実施例 4-512 Example 4-512
tert-ブチル 5-[(5, 6 -ジメチル- 1 - {[(4-メチルフエニル)スルホ二ル]イミノ} - 3- ォキソ -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィル)スルファニル〕 ペンチルカーバメート tert-butyl 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-8-yl) sulfanyl] pentyl carbamate
実施例 3- 4で得た化合物(2.03 g, 5.32聊 ol)および炭酸カリウム (1.10g, 7.98 Hffliol) の DMF (29 ml) の懸濁液に 60°Cで攪拌しながら参考例 19で得た N_Boc- 5 - ァミノ-卜ペンチル メシレ一ト (1.90 g, 5.85 Miol)を 2回に分けて加え、 反応 液を 22時間攪拌した。 反応液に 1N塩酸 (11.7ml)を加え、 酢酸ェチル (200 nil) で希釈後、 0.1N塩酸、 水および飽和食塩水で洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して濃赤色油状物として得た。 これをシリカゲル (20 g)のクロマトグラフィ ― (酢酸ェチルーへキサン、 3:7-4:6) により精製し、 メタノールから結晶化して 表題化合物(1.29 g, 2.28腿 ol)を赤褐色粉末として得た。 A suspension of the compound obtained in Example 3-4 (2.03 g, 5.32 liolol) and potassium carbonate (1.10 g, 7.98 Hffliol) in DMF (29 ml) was obtained in Reference Example 19 while stirring at 60 ° C. N_Boc-5-aminopentyl mesylate (1.90 g, 5.85 Miol) was added in two portions, and the reaction solution was stirred for 22 hours. 1N Hydrochloric acid (11.7ml) was added to the reaction mixture, diluted with ethyl acetate (200nil), washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to obtain a dark red oil. Was. This was purified by chromatography on silica gel (20 g)-(ethyl acetate-hexane, 3: 7-4: 6), and crystallized from methanol to give the title compound (1.29 g, 2.28 mol) as a red-brown powder.
'H-NMR (300MHz, CDC13) : (51.45 (9H, s), 1.47 (2H, m), 1.70 (2H, quint, J= 7.5 Hz), 1.79 (3H, q, J= 0.9 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.93 (2H, t, J= 7.5 Hz), 3.11 (2H, brq, J= 6.9 Hz), 4.54 (1H, m), 7.29 (2H, d, J= 8.1 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). 'H-NMR (300MHz, CDC1 3): (51.45 (9H, s), 1.47 (2H, m), 1.70 (2H, quint, J = 7.5 Hz), 1.79 (3H, q, J = 0.9 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 2.93 (2H, t, J = 7.5 Hz), 3.11 (2H, brq, J = 6.9 Hz), 4.54 (1H, m), 7.29 (2H, d, J = 8.1 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C25H34N405S3として 計算値: C, 52.98; H, 6.05; N, 9.89; S, 16.97 ( ) . Elementary analysis: as C 25 H 34 N 4 0 5 S 3 Calculated: C, 52.98; H, 6.05; N, 9.89; S, 16.97 ().
実測値: C, 53.09; H, 6.13; N, 10.10; S, 16.78 (%) . Found: C, 53.09; H, 6.13; N, 10.10; S, 16.78 (%).
mp. 144.0-145.0^: mp. 144.0-145.0 ^:
実施例 4- 513 Example 4-513
tert -ブチル 6-[(5,6-ジメチル -1- {[(4 -メチルフエニル)スルホニル]イミノ} - 3_ ォキソ -2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル] へキシルカ一バメ一ト tert-Butyl 6-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1, 4] thiazine-8-yl) sulfanyl] hexylkabamate
実施例 3-4で得た化合物 (2.06 g, 5.39腿 ol)および炭酸カリウム (1.12 g, 8.09 mmol) の DMF (30 ml) の懸濁液に 60°Cで攪拌しながら参考例 20で得たョ ゥ化 N- Boc - 6-ァミノ-卜べキシル (1.88 g, 5.39讓 ol)を 2回に分けて加え、 反 応液を 3時間攪拌した。反応液に 1N塩酸 ( 8ml)を加え、メタノール一水 (1:1、 10 ml)で希釈し、室温で 30分間攪拌した。析出物を濾取し、メタノール—水 (1:1) およびメタノールで洗浄後、 乾燥して表題化合物 (2,60 g, 4.48腿 ol)を赤色粉 末として得た。 A suspension of the compound obtained in Example 3-4 (2.06 g, 5.39 mol) and potassium carbonate (1.12 g, 8.09 mmol) in DMF (30 ml) was obtained in Reference Example 20 with stirring at 60 ° C. N-Boc-6-amino-to-bexyl bromide (1.88 g, 5.39 benzyl) was added in two portions, and the reaction solution was stirred for 3 hours. 1N Hydrochloric acid (8 ml) was added to the reaction solution, diluted with methanol / water (1: 1, 10 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (2,60 g, 4.48 pt) as a red powder.
Ή-NMR (300MHz, CDC13) : 51.37 (2H, m), 1.45 (9H, s), 1.47 (2H, ) , 1.68 (2H, quint, J= 7.2 Hz), 1.79 (3H, brs), 2.26 (3H, q, J= 0.9 Hz), 2.42 (3H, s), 2.92 (2H, t, J= 7.5 Hz), 3.11 (2H, brq, J= 6.3 Hz), 4.53 (1H, m), 7.29 (2H, d, J= 8.7 Hz), 7.85 (2H, d, J= 8.1 Hz), 9.63 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): 51.37 (2H, m), 1.45 (9H, s), 1.47 (2H,), 1.68 (2H, quint, J = 7.2 Hz), 1.79 (3H, brs), 2.26 (3H, q, J = 0.9 Hz), 2.42 (3H, s), 2.92 (2H, t, J = 7.5 Hz), 3.11 (2H, brq, J = 6.3 Hz), 4.53 (1H, m), 7.29 (2H, d, J = 8.7 Hz), 7.85 (2H, d, J = 8.1 Hz), 9.63 (1H, brs).
元素分析値: C26H36N405S3として Elementary analysis: as C 26 H 36 N 4 0 5 S 3
計算値: C, 53.77; H, 6.25; N, 9.65; S, 16.56 ( ) . Calculated: C, 53.77; H, 6.25; N, 9.65; S, 16.56 ().
実測値: C, 53.69; H, 6.17; N, 9.85; S, 16.26 ( ) . Found: C, 53.69; H, 6.17; N, 9.85; S, 16.26 ().
mp. 136.0-136.5°C mp.136.0-136.5 ° C
実施例 4- 514Example 4-514
-(5, 6 -ジメチル- 8-{ [(卜へキサノィル- 4 -ピぺリジニル)メチル]スルファニ ル} - 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン) - 4- 実施例 4- U6で得られた N- (5, 6 -ジメチル- 8- {[(4 -ピベリジニル)メチル]スルフ ァニル }- 3-ォキソ -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデ ン)- 4-メチルベンゼンスルホンアミド塩酸塩 (152 mg, 0.295 mmol) と n-へキサ ン酸 (0.0493 ml, 0.354匪 ol) の Ν,Ν-ジメチルホルムアミド (5.00 ml) 溶液に、 DEPC (0.0572 ml, 0.354腿 ol) 続いてトリェチルァミン (0.0987 ml, 0.708 ηιπιο1) を加えた。反応混合物を室温で 15時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥 (MgS04)後、溶媒を減圧下に留去して題記化合物(74.9 rag, 44¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶し て融点 183- 185°Cの暗赤色結晶を得た。 -(5,6-Dimethyl-8-{[(trihexanoyl-4-piridinyl) methyl] sulfanyl} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-triylidene) -4-Example 4-N- (5,6-dimethyl-8-{[(4-piberidinyl) methyl] sulfanyl} -3-oxo-2 obtained in U6 , 3-Dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-trilidene) -4-methylbenzenesulfonamide hydrochloride (152 mg, 0.295 mmol) and n-hexane To a solution of acetic acid (0.0493 ml, 0.354 bandol) in Ν, Ν-dimethylformamide (5.00 ml) was added DEPC (0.0572 ml, 0.354 ml) followed by triethylamine (0.0987 ml, 0.708 ηιπιο1). After stirring the reaction mixture at room temperature for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (74.9 rag, 44¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 183-185 ° C.
IR (KBr) V: 3233, 2930, 2870, 2859, 1759, 1622, 1557 cm'1. IR (KBr) V: 3233, 2930, 2870, 2859, 1759, 1622, 1557 cm ' 1 .
Ή-NMR (CDC13) : 50.905 (3H, t, J=6.6 Hz), 0.980-1.45 (6H, m), 1.45-2.00 Ή-NMR (CDC1 3): 50.905 (3H, t, J = 6.6 Hz), 0.980-1.45 (6H, m), 1.45-2.00
(5H, m), 1.79 (3H, d, J=1.0 Hz), 2.27 (3H, d, J=0.8 Hz), 2.32 (2H, t, J=7.4 Hz), 2.43 (3H, s), 2.43 (1H, bt, J=14.6 Hz), 2.85 (2H, t, J=6.2 Hz), 2.99(5H, m), 1.79 (3H, d, J = 1.0 Hz), 2.27 (3H, d, J = 0.8 Hz), 2.32 (2H, t, J = 7.4 Hz), 2.43 (3H, s), 2.43 (1H, bt, J = 14.6 Hz), 2.85 (2H, t, J = 6.2 Hz), 2.99
(1H, bt, J=12.6 Hz), 3.87 (1H, bd, J=13.2 Hz), 4.66 (1H, bd. J=12.8 Hz), 7.29(1H, bt, J = 12.6 Hz), 3.87 (1H, bd, J = 13.2 Hz), 4.66 (1H, bd.J = 12.8 Hz), 7.29
(2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.4 Hz), 9.63 (1H, bs). (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, bs).
元素分析値: C27¾6N404S30.3H20として Elemental analysis: C 27 ¾ 6 N 4 0 4 S 3 0.3H 2 0
計算値 (%) : C, 55.70; H, 6.34; N, 9.62; S, 16.52. Calculated value (%): C, 55.70; H, 6.34; N, 9.62; S, 16.52.
実測値 (%) : C, 55.67; H, 6.15; N, 9.62;' S, 16.56. Found (%): C, 55.67; H, 6.15; N, 9.62; 'S, 16.56.
実施例 4- 515 Example 4-515
N-U- [(5,6-ジメチル- 1-{[(4-メチルフエニル)スルホニル]イミノ}-3-ォキソ -2, 3 -ジヒド口- 1H -ィミダゾ [5, 1- c] [1,4]チアジン- 8-ィル)スルファニル]プチ ル}へキサンアミド NU-[(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] Thiazine-8-yl) sulfanyl] butyl} hexaneamide
実施例 3 - 4で得られた N- (5,6-ジメチル -3-ォキソ -8-スルファニル -2, 3 -ジヒド 口- 1H-イミダゾ [5,l_c] [1,4]チアジン - 1 -ィリデン)-4-メチルベンゼンスルホン アミド (200 mg, 0.524匪 ol) と炭酸カリウム (109 mg, 0.786 mmol) の Ν,Ν -ジ メチルホルムアミド (3.00 ml) の懸濁液に、 参考例 63で得られたメタンスルホ ン酸 4 -へキサノィルアミノブチルエステル (209 mg, 0.786 mmol) の N,N-ジメ チルホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を 80°Cで 3時間加熱し た後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥(MgS04)後、 溶媒を減圧下に留去した。残留物をシリカゲル力ラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (110 mg, 38¾) を合 成した。酢酸ェチル-ジェチルエーテルから再結晶して融点 118- 120°Cの暗赤色結 晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihidido-1H-imidazo [5, l_c] [1,4] thiazine obtained in Example 3-4 A suspension of (ylidene) -4-methylbenzenesulfonamide (200 mg, 0.524 ol) and potassium carbonate (109 mg, 0.786 mmol) in Ν, Ν-dimethylformamide (3.00 ml) was obtained in Reference Example 63. A solution of the obtained methanesulfonic acid 4-hexanoylaminobutyl ester (209 mg, 0.786 mmol) in N, N-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 ° C. for 3 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to synthesize the title compound (110 mg, 38¾). Recrystallized from ethyl acetate-getyl ether to give a dark red color with a melting point of 118-120 ° C A crystal was obtained.
IR (KBr) v: 3250, 2928, 2859, 1748, 1645, 1615, 1559 cm"1. IR (KBr) v: 3250, 2928, 2859, 1748, 1645, 1615, 1559 cm " 1 .
Ή-NMR (CDClg) : δ 0.894 (3H, t, J=6.9Hz), 1.22-1.40 (4H, m), 1.50-1.84 (6H, m), 1.78 (3H, d, J=0.9 Hz), 2.16 (2H, t, J=7.2 Hz), 2.26 (3H, d, J=0.9 Hz), 2.42 (3H, s), 2.96 (2H, t, J=6.9 Hz), 3.28 (2H, q, J-6.3 Hz), 5.52 (1H, bs), 7.29 (2H, d, J=7.8 Hz), 7.84 (2H, d, J=8.4 Hz), 9.63 (1H, bs). Ή-NMR (CDClg): δ 0.894 (3H, t, J = 6.9Hz), 1.22-1.40 (4H, m), 1.50-1.84 (6H, m), 1.78 (3H, d, J = 0.9 Hz), 2.16 (2H, t, J = 7.2 Hz), 2.26 (3H, d, J = 0.9 Hz), 2.42 (3H, s), 2.96 (2H, t, J = 6.9 Hz), 3.28 (2H, q, J -6.3 Hz), 5.52 (1H, bs), 7.29 (2H, d, J = 7.8 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, bs).
元素分析値: C25H35N404S3 ·0.4H20として Elemental analysis: C 25 H 35 N 4 0 4 S 3 · 0.4H 2 0
計算値 (%) : C, 53.72; H, 6.46; N, 10.02; S, 17.21. Calculated value (%): C, 53.72; H, 6.46; N, 10.02; S, 17.21.
実測値 (¾) : C, 53.75; H, 6.03; N, 10.19; S, 17.22. Found (¾): C, 53.75; H, 6.03; N, 10.19; S, 17.22.
実施例 4- 516 Example 4-516
N - (4- {(5, 6-ジメチル-卜 {[(4-メチルフエニル)スルホニル]ィミノ卜 3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 8 -ィル)スルファニル]ェチ ル}フエニル)へキサンアミド N- (4-{(5,6-dimethyl-tolu [[(4-methylphenyl) sulfonyl] iminoto 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-8-yl) sulfanyl] ethyl} phenyl) hexaneamide
実施例 3-4で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルファニル -2, 3-ジヒド ロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン)-4-メチルベンゼンスルホン アミド (200 mg, 0.524 mmol) と炭酸カリウム (109 mg, 0.786 mmol) の N,N-ジ メチルホルムアミド (3.00 ml) の懸濁液に、 参考例 65で得られたメタンスルホ ン酸 2- (4 -へキサノィルァミノフエニル)ェチルエステル (197 mg, 0.786 mmol) の N,N -ジメチルホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を 80°Cで 3 時間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 ( gS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル(10:1, v/v)で溶出して題記化合'物 (48.4 mg, 15¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 180-182°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 To a suspension of 1-ylidene) -4-methylbenzenesulfonamide (200 mg, 0.524 mmol) and potassium carbonate (109 mg, 0.786 mmol) in N, N-dimethylformamide (3.00 ml), refer to Reference Example 65. A solution of the obtained methanesulfonate 2- (4-hexanoylaminophenyl) ethyl ester (197 mg, 0.786 mmol) in N, N-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 ° C for 3 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (GS0 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to synthesize the title compound (48.4 mg, 15¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 180-182 ° C.
IR (KBr) リ: 3300, 2955, 2928, 2868, 1732, 1663, 1607, 1559 cm—1. IR (KBr): 3300, 2955, 2928, 2868, 1732, 1663, 1607, 1559 cm— 1 .
Ή-NMR (CDC13) : 0.910 (3H, i, J=6.6Hz), 1.28-1.43 (4H, m) , 1.65-1.82 (2Η, m), 1.78 (3Η, s), 2.26 (3H, s), 2.35 (2H, t, J=7.2 Hz), 2.42 (3H, s), 2.92 (2H, t, J=8.8 Hz), 3.15 (2H, t, J=8.8 Hz), 7.09 (1H, bs), 7.14 (2H, d, J=8.4 Hz), 7.28 (2H, d, J=8.0 Hz), 7.47 (2H, d, J=8,8 Hz), 7.82 (2H, d, J=8.4 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): 0.910 (3H, i, J = 6.6Hz), 1.28-1.43 (4H, m), 1.65-1.82 (2Η, m), 1.78 (3Η, s), 2.26 (3H, s ), 2.35 (2H, t, J = 7.2 Hz), 2.42 (3H, s), 2.92 (2H, t, J = 8.8 Hz), 3.15 (2H, t, J = 8.8 Hz), 7.09 (1H, bs ), 7.14 (2H, d, J = 8.4 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.47 (2H, d, J = 8,8 Hz), 7.82 (2H, d, J = 8.4 Hz) ), 9.60 (1H, bs).
元素分析値: C29H34N404S30.3H20として Elemental analysis: C 29 H 34 N 4 0 4 S 3 0.3 H 2 0
計算値 (%) : C, 57.65; H, 5.77; N, 9.27; S, 15.92. Calculated value (%): C, 57.65; H, 5.77; N, 9.27; S, 15.92.
実測値 (¾) : C, 57.61; H, 5.62; N, 9.30; S, 15.55. Found (値): C, 57.61; H, 5.62; N, 9.30; S, 15.55.
実施例 4- 518 Example 4-518
N - [2, 3-ジヒドロ _5, 6-ジメチル -8- (イソペンチルスルファニル) -3-ォキソ -1H -ィ ミダゾ [5, 1-c] [1, 4]チアジン -1-ィリデン] -4-フルォ口メチルべンゼンスルホン アミド N- [2,3-dihydro_5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] -4 -Fluoro mouth methylbenzene sulfonamide
実施例 7 - 8で得られた N-[2, 3-ジヒド口- 5, 6-ジメチル- 8- (ィソペンチルスルフ ァニル ) -3-ォキソ -1H -イミダゾ [5,卜 c] [1, 4]チアジン -1-ィリデン ]-4-ヒドロキ シメチルベンゼンスルホンアミド (259 mg, 0.554mmol) のジクロロメタン (3.00 ml) 溶液に、 ジェチルァミノ硫黄トリフルオリド (0.114 ml, 0.830 mmol) のジ クロロメタン (2.00ml) 溶液を- 70°Cでゆっくりと滴下した。 反応混合物を- 70°C で 20分間かき混ぜた後、氷冷下飽和炭酸水素ナトリゥム水溶液を加えてジク口口 メタンで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題 記化合物 (30.5mg, 12%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテ ルから再結晶して融点 178- 180DCの暗赤色結晶を得た。 N- [2,3-Dihydrido-5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-1H-imidazo [5, tric] obtained in Examples 7-8 1,4] Thiazine-1-ylidene] -4-hydroxymethylbenzenesulfonamide (259 mg, 0.554 mmol) in dichloromethane (3.00 ml) solution, dimethylaminosulfur trifluoride (0.114 ml, 0.830 mmol) in dichloromethane (2.00 ml) The solution was slowly added dropwise at -70 ° C. After stirring the reaction mixture at -70 ° C for 20 minutes, a saturated aqueous solution of sodium hydrogen carbonate was added thereto under ice-cooling, and the mixture was extracted with methane. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (30.5 mg, 12%) as a dark red crystals. Acetate Echiru - was recrystallized from Jechiruete Le give a dark red crystals, mp 178- 180 D C.
IR (KBr)ソ: 3221, 2959, 2934, 2851, 1753, 1663, 1609, 1559 cm— IR (KBr): 3221, 2959, 2934, 2851, 1753, 1663, 1609, 1559 cm—
¾-NMR (CDC13) : 50.931 (6H, d, J=6.3Hz), 1.50-1.62 (2H, m), 1.69 (1H, septet, J=6.9 Hz), 1.79 (3H, d, J=0.9Hz), 2.27 (3H, d, J-0.9 Hz), 2.95 (2H, t, J=7.8 Hz), 4.62 (2HX0.331, s, CI), 5.46 (2HX0.669, d, J=47.1 Hz, F), 7.48 (2H X0.669, d, J=8.4Hz, F), 7.51 (2HX0.331, d, J=8.7 Hz, CI), 7.96 (2HX0.331, d, J=8.7 Hz, CI), 8.00 (2HX0.661, d, J=7.8 Hz), 9.61 (1H, bs). ¾-NMR (CDC1 3): 50.931 (6H, d, J = 6.3Hz), 1.50-1.62 (2H, m), 1.69 (1H, septet, J = 6.9 Hz), 1.79 (3H, d, J = 0.9 Hz), 2.27 (3H, d, J-0.9 Hz), 2.95 (2H, t, J = 7.8 Hz), 4.62 (2HX0.331, s, CI), 5.46 (2HX0.669, d, J = 47.1 Hz) , F), 7.48 (2H X0.669, d, J = 8.4Hz, F), 7.51 (2HX0.331, d, J = 8.7 Hz, CI), 7.96 (2HX0.331, d, J = 8.7 Hz, CI), 8.00 (2HX0.661, d, J = 7.8 Hz), 9.61 (1H, bs).
元素分析値: C2。H24N303S3F (67¾), CI (33Dとして Elemental analysis: C 2. H 24 N 3 0 3 S 3 F (67¾), CI (as 33D
計算値 (¾) : C, 50.56; H, 5.09; N, 8.85; S, 20.25. Calculated value (¾): C, 50.56; H, 5.09; N, 8.85; S, 20.25.
実測値 (¾) : C, 50.47; H, 4.85; N, 8.78; S, 20.11. Observed value (¾): C, 50.47; H, 4.85; N, 8.78; S, 20.11.
実施例 4- 519 Example 4- 519
6-メチル - 1-{[(4-メチルフエニル)スルホ二ル]イミノ}-3-ォキソ - 8-[(5, 5, 6, 6, 6- ペンタフルォ口)スルファニル ]-2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン -5_カルボン酸ェチル 6-Methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentafluoro) sulfanyl] -2,3-dihydro- 1H-imidazo [5, 1-c] [1,4] thiazine -5_Ethyl carboxylate
実施例 3-21で得た化合物 α.68 g, 3.82薩 ol)および炭酸カリウム (0.792 g, 5.73 mmol) の DMF (25 ml) の懸濁液に 70°Cで攪拌しながらヨウ化 5,5,6,6,6- ペン夕フルォ口へキシル (純度 87 , 1.33 g, 3.82 mmol)を 3回に分けて加え、 反応液を 6 時間攪拌した。 反応液に 1N塩酸 (8.4 ml)を加え、 メタノール—水 (2:3、 25 ml)で希釈し、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール一 水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物(2.00 g, 3.26 mmol) を橙色粉末として得た。 A suspension of the compound obtained in Example 3-21, α.68 g, 3.82 mol) and potassium carbonate (0.792 g, 5.73 mmol) in DMF (25 ml) was stirred at 70 ° C. with iodide 5, 5,6,6,6-Pentafluorohexyl (purity 87, 1.33 g, 3.82 mmol) was added in three portions, and the reaction solution was stirred for 6 hours. 1N Hydrochloric acid (8.4 ml) was added to the reaction solution, diluted with methanol-water (2: 3, 25 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (2.00 g, 3.26 mmol) as an orange powder.
Ή-NMR (200MHz, CDC13) : 51.33 (3H, t, J= 7.1 Hz), 1.72 (4H, m), 1.92 (3H, s), 2.05 (2H, m), 2.43 (3H, s), 2.99 (2H, t, J= 7.0 Hz), 4.33 (2H, q, J= 7.1 Hz), 7.30 (2H, d, J= 8.2 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.70 (1H, brs). 元素分析値: C23H24N305S3F5として計算値: C, 45.02; H, 3.94; N, 6.85; S, 15.68; F, 15.48. Ή-NMR (200MHz, CDC1 3 ): 51.33 (3H, t, J = 7.1 Hz), 1.72 (4H, m), 1.92 (3H, s), 2.05 (2H, m), 2.43 (3H, s), 2.99 (2H, t, J = 7.0 Hz), 4.33 (2H, q, J = 7.1 Hz), 7.30 (2H, d, J = 8.2 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.70 ( . 1H, brs) elemental analysis: C 23 H 24 N 3 0 5 S 3 F 5 calculated: C, 45.02; H, 3.94 ; N, 6.85; S, 15.68; F, 15.48.
実測値: C, 45.13; H, 4.11; N, 6.83; S, 15.74; F, 15.27. Found: C, 45.13; H, 4.11; N, 6.83; S, 15.74; F, 15.27.
mp. 195.0-196.5°C mp. 195.0-196.5 ° C
実施例 4- 520 Example 4-520
N- [5, 6-ジメチル- 3-ォキソ -8- [(5-ヒドロキシペンチル)スルファニル ]-2, 3-ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-トイリデン] -4 -メチルベンゼンスルホ ンアミド N- [5,6-dimethyl-3-oxo-8-[(5-hydroxypentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-toylidene ] -4-Methylbenzenesulfonamide
実施例 3-4で得られた N- (5,6-ジメチル- 3-ォキソ -8-スルファニル -2, 3 -ジヒド ロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 1-ィリデン)-4-メチルベンゼンスルホン アミド (3.00 g, 7.86腿 ol) と炭酸カリウム (1.63 g, 11.8 mmol) の N,N-ジメ チルホルムアミド(55.0 ml)の懸濁液に、 5-ブロモ -卜ペンタノール(1.58 g, 9.44 mmol) の N,N-ジメチルホルムアミド(5.00ml)溶液を加えた。反応混合物を 80 で 1.5時間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (3.04 g, 83%) を暗赤色 結晶として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 182_184°Cの 暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine obtained in Example 3-4 To a suspension of 1-ylidene) -4-methylbenzenesulfonamide (3.00 g, 7.86 tmol) and potassium carbonate (1.63 g, 11.8 mmol) in N, N-dimethylformamide (55.0 ml) was added 5-bromobenzene. A solution of -topentanol (1.58 g, 9.44 mmol) in N, N-dimethylformamide (5.00 ml) was added. After heating the reaction mixture at 80 for 1.5 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (3.04 g, 83%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 182-184 ° C.
IR (KBr)レ : 3700-2700, 2932, 2863, 1730, 1661, 1615, 1557 cm一1. lH - NMR (CDClg) : (51.44-1.64 (4H, m), 1.67-1.77 (2H, m), 1.79 (3H, d, J=0.9 Hz), 2.26 (3H, d, J-0.6 Hz), 2.42 (3H, s), 2.95 (2H, t, J=7.5 Hz), 3.66 (2H, t, J=6.3 Hz), 7.29 (2H, d, J=8.1 Hz), 7.85 (2H, d, J=8.4 Hz), 9.63 (1H, bs). 元素分析値: C2DH25N304S3として IR (KBr): 3700-2700, 2932, 2863, 1730, 1661, 1615, 1557 cm- 1 . l H - NMR (CDClg): (51.44-1.64 (4H, m), 1.67-1.77 (2H, m), 1.79 (3H, d, J = 0.9 Hz), 2.26 (3H, d, J-0.6 Hz) , 2.42 (3H, s), 2.95 (2H, t, J = 7.5 Hz), 3.66 (2H, t, J = 6.3 Hz), 7.29 (2H, d, J = 8.1 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 ( 1H, bs) Elementary analysis:. a C 2D H 25 N 3 0 4 S 3
計算値 ( ) : C, 51.37; H, 5.39; N, 8.99. Calculated (): C, 51.37; H, 5.39; N, 8.99.
実測値 (¾) : C, 51.33; H, 5.39; N, 8.92. Found (¾): C, 51.33; H, 5.39; N, 8.92.
実施例 4-521 Example 4-521
ェチル 5-メチル -卜 {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -8-[(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニル] - 2, 3 -ジヒド口- 1H -ィ ミダゾ [5,1 - c] [1,4]チアジン- 6-カルポキシレート Ethyl 5-methyl-tri {[(4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydric -1H-Midazo [5,1-c] [1,4] thiazine-6-carboxylate
実施例 3-3 で得られたェチル 5-メチル _1-{[ (4-メチルフエニル)スルホニル] ィミノ卜 3_ォキソ -8-スルファニル _2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チア ジン -6-カルポキシレート(6.98 g, 16.8 n脚 1)と炭酸カリウム(3.48 g, 25.2腿01) の Ν,Ν-ジメチルホルムアミド (100ml) の懸濁液に、 参考例 9で得られたメタン スルホン酸 5, 5, 6, 6, 6-ペンタフルォ口へキシルエステル (5.45 g, 20.2 mmol) の N,N-ジメチルホルムアミド (40.0 ml) 溶液を加えた。 反応混合物を 80°Cで 1 時間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (9.00 g, 87¾) を暗赤色結晶 として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 134- 136°Cの暗赤 色結晶を得た。 Ethyl 5-methyl_1-{[((4-methylphenyl) sulfonyl] iminoto 3_oxo-8-sulfanyl_2,3-dihydro-1H-imidazo [5,1-c] obtained in Example 3-3 Example of a suspension of 1,4] thiazine-6-carboxylate (6.98 g, 16.8 n legs 1) and potassium carbonate (3.48 g, 25.2 thigh 01) in Ν, (-dimethylformamide (100 ml) A solution of methanesulfonic acid 5,5,6,6,6-pentafluorohexyl ester (5.45 g, 20.2 mmol) obtained in 9 in N, N-dimethylformamide (40.0 ml) was added. After heating the reaction mixture at 80 ° C. for 1 hour, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (9.00 g, 87¾) and the obtained as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 134-136 ° C.
IR (KBr)リ: 3220, 2984, 2944, 2884, 1748, 1713, 1678, 1622, 1557 cm"1. Ή -腿 (CDC13) : 51.32 (3H, t, J=7. Hz), 1.64-1.80 (4H, m), 1.80-2.20 (2H, m), 2.42 (3H, s), 2.75 (3H, s), 3.02 (2H, t, J=7.0 Hz), 4.26 (2H, q, J=7. Hz), 7.29 (2H, d, J=8.4 Hz), 7.84 (2H, d, J=8.4 Hz), 9.78 (1H, bs). IR (KBr) Re:. 3220, 2984, 2944, 2884, 1748, 1713, 1678, 1622, 1557 cm "1 Ή - thigh (CDC1 3): 51.32 (. 3H, t, J = 7 Hz), 1.64- 1.80 (4H, m), 1.80-2.20 (2H, m), 2.42 (3H, s), 2.75 (3H, s), 3.02 (2H, t, J = 7.0 Hz), 4.26 (2H, q, J = 7.Hz), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.78 (1H, bs).
元素分析値: C23H24N305S3F5として Elemental analysis: as C 23 H 24 N 3 0 5 S 3 F 5
計算値 (¾) : C, 45.02; H, 3.94; N, 6.85; S, 15.68. Calculated value (¾): C, 45.02; H, 3.94; N, 6.85; S, 15.68.
実測値 (¾) : C, 45.07; H, 4.05; N, 6.99; S, 15.89. Found (値): C, 45.07; H, 4.05; N, 6.99; S, 15.89.
実施例 4-522 Example 4-522
ェチル 5- [ (5, 6-ジメチル- 1- { [ (4 -メチルフエニル)スルホニル]ィミノ }-3-ォキ ソ- 2, 3-ジヒドロ _1H-イミダゾ [5, 1-c] [1,4]チアジン- 8 -ィル)スルファニル]ペン タノエート Ethyl 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oki So-2,3-dihydro_1H-imidazo [5, 1-c] [1,4] thiazine-8-yl) sulfanyl] pentanoate
実施例 3-4で得られた N- (5,6-ジメチル -3-ォキソ -8-スルファニル -2, 3-ジヒド 口 -1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン)-4-メチルベンゼンスルホン アミ ド (500 mg, 1.31 腿 ol) と炭酸カリウム (272 mg, 1.97 腿 ol) の N,N-ジメ チルホルムアミド (5.00ml) の懸濁液に、 5-ブロモ吉草酸 ェチルエステル (259 l, 1.57 腿 ol) の N,N -ジメチルホルムアミ ド (5.00 ml) 溶液を加えた。 反応 混合物を 80°Cで 30分間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(459 rag, 69¾) を合成した。酢酸ェチル-ジェチルエーテルから再結晶して融点 162- 164T:の暗赤 色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 A suspension of 1-ylidene) -4-methylbenzenesulfonamide (500 mg, 1.31 thigh) and potassium carbonate (272 mg, 1.97 thigh) in N, N-dimethylformamide (5.00 ml) was added to the suspension. A solution of N-N-dimethylformamide (5.00 ml) in -ethyl bromovalerate (259 l, 1.57 t) was added. After heating the reaction mixture at 80 ° C. for 30 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (459 rag, 69¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 162-164T :.
IR (KBr)リ : 3221, 2980, 2936, 1732, 1663, 1564 cm—1. IR (KBr): 3221, 2980, 2936, 1732, 1663, 1564 cm— 1 .
Ή-NMR (CDC13) : 51.27 (3H, t, J=7.2 Hz) , 1.67-1.82 (4Η, m), 1.79 (3Η, d, Ή-NMR (CDC1 3): 51.27 (3H, t, J = 7.2 Hz), 1.67-1.82 (4Η, m), 1.79 (3Η, d,
J=1.0Hz), 2.26 (3H, d, J=1.2Hz), 2.28-2.38 (2H, m), 2.42 (3H, s), 2.88-2.98 (2H, m), 4.14 (2H, q, J=7.2 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4J = 1.0Hz), 2.26 (3H, d, J = 1.2Hz), 2.28-2.38 (2H, m), 2.42 (3H, s), 2.88-2.98 (2H, m), 4.14 (2H, q, J = 7.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4
Hz), 9.61 (1H, bs). Hz), 9.61 (1H, bs).
元素分析値: C22H27N305S3として Elementary analysis: as C 22 H 27 N 3 0 5 S 3
計算値 (¾) : C, 51.85; H, 5.34; N, 8.24. Calculated value (¾): C, 51.85; H, 5.34; N, 8.24.
実測値 (¾) : C, 51.68; H, 5.29; N, 8.16. Found (¾): C, 51.68; H, 5.29; N, 8.16.
実施例 4-523 Example 4-523
ェチル 4 - [ (5, 6 -ジメチル -1 - { [ (4 -メチルフエニル)スルホニル]イミノ } - 3-ォキ リ- 1, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 8 -ィル)スルファニル]ブタ ノエ一卜 Ethyl 4-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-1,3-dihydro-1H-imidazo [5,1-c] [1, 4] Thiazine-8-yl) sulfanyl] butane
実施例 3-4で得られた N-(5, 6-ジメチル -3-ォキソ -8-スルファニル -2, 3-ジヒド 口- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン)-4-メチルベンゼンスルホン アミド (500 mg, 1.31 mmol) と炭酸カリウム (272 mg, 1.97匪 ol) の Ν,Ν-ジメ チルホルムアミド (5.00ml) の懸濁液に、 4-ブロモ酪酸 ェチルエステル (0.234 ml, 1.57匪 ol) の Ν,Ν-ジメチルホルムアミド (5.00 ml) 溶液を加えた。 反応混 合物を 80°Cで 30分間加熱した後、 塩酸を加えて酢酸ェチルで抽出した。 抽出 液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (396 mg, 61¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 1 0-142での暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 To a suspension of 1-ylidene) -4-methylbenzenesulfonamide (500 mg, 1.31 mmol) and potassium carbonate (272 mg, 1.97 marl) in Ν, Ν-dimethylformamide (5.00 ml) was added 4-bromobenzene. A solution of ethyl butyrate (0.234 ml, 1.57 bandol) in Ν, Ν-dimethylformamide (5.00 ml) was added. After heating the reaction mixture at 80 ° C for 30 minutes, hydrochloric acid was added and extracted with ethyl acetate. Extraction Washing liquid, dried (MgSO 4), the solvent was evaporated under reduced pressure to give the title compound (396 mg, 61¾) was obtained as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with mp 10-142.
IR (KBr) V: 3200, 2976, 2924, 1728, 1661, 1611, 1563 cm"1. IR (KBr) V: 3200, 2976, 2924, 1728, 1661, 1611, 1563 cm " 1 .
Ή-NMR (CDC13) : 51.28 (3H, t, J=7.2 Hz), 1.79 (3H, d, J=0.6 Hz), 2.00 (2H, quint, J=7.2 Hz), 2.26 (3H, d, J=l.2 Hz), 2.42 (3H, s), 2.43 (2H, t, J=6.9 Hz), 3.00 (2H, t, J=7.5 Hz), 4.15 (2H, q, J-6.9 Hz), 7.30 (2H, d, J=7.8 Hz), 7.85 (2H, d, J=8.1 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): 51.28 (3H, t, J = 7.2 Hz), 1.79 (3H, d, J = 0.6 Hz), 2.00 (2H, quint, J = 7.2 Hz), 2.26 (3H, d, J = l.2 Hz), 2.42 (3H, s), 2.43 (2H, t, J = 6.9 Hz), 3.00 (2H, t, J = 7.5 Hz), 4.15 (2H, q, J-6.9 Hz) , 7.30 (2H, d, J = 7.8 Hz), 7.85 (2H, d, J = 8.1 Hz), 9.61 (1H, bs).
元素分析値: C21H25N305S3として Elemental analysis: C 21 H 25 N 3 0 5 S 3
計算値 ) : C, 50.89; H, 5.08; N, 8.48; S, 19.71. Calculated values): C, 50.89; H, 5.08; N, 8.48; S, 19.71.
実測値 (¾) : C, 50.77; H, 4.93; N, 8.53; S, 19.32. Found (¾): C, 50.77; H, 4.93; N, 8.53; S, 19.32.
実施例 4- 524 Example 4-524
N- {8- [(2-ヒド口キシェチル)スルファニル] - 5, 6 -ジメチル -3-ォキソ - 2, 3-ジヒド ロ- 1H-イミダゾ [5,l-c] [1,4]チアジン- 1-イリデン }- 4-メチルベンゼンスルホンァ ミド N- {8- [(2-Hydroxixethyl) sulfanyl]-5, 6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-1- Ilidene}-4-methylbenzenesulfonamide
実施例 3-4で得た化合物(1.06 g, 2.79腿 ol) および炭酸カリウム (558 mg, 4.18 mmol) の DMF (16ml) の懸濁液に 50°Cで攪拌しながら 2-ョ一ドエタノール (0.265 ml, 3.34 mmol) の DMF (2 ml)溶液を 30分間かけて滴下し、 反応液をさ らに 5時間攪拌した。反応中に 2 -ョードエタノール(0.200 ml, 2.51腿01) の DMF (2 ml)溶液を加えた。 反応液に 1N塩酸 (5.6 ml) を加え、 メタノ一ルー水(1:3, 20 ml)で希釈した。析出物を濾取し、 メタノール一水(1:1)およびメタノールで洗 浄後、 THF (70 ml)に懸濁した。 析出物を濾取し、 酢酸ェチルで洗浄後、 乾燥して 表題化合物 (286 mg, 0.672 mmol)を灰紫色粉末として得た。 母液をメタノールお よび酢酸ェチルから結晶化し表題化合物(695 mg, 1.63讓 ol)を灰紫色粉末として 得た。 To a suspension of the compound obtained in Example 3-4 (1.06 g, 2.79 tmol) and potassium carbonate (558 mg, 4.18 mmol) in DMF (16 ml) was added 2-fluoroethanol while stirring at 50 ° C. (0.265 ml, 3.34 mmol) in DMF (2 ml) was added dropwise over 30 minutes, and the reaction solution was further stirred for 5 hours. During the reaction, 2-MF ethanol (0.200 ml, 2.51 thigh 01) in DMF (2 ml) was added. 1N Hydrochloric acid (5.6 ml) was added to the reaction mixture, and the mixture was diluted with methanolic water (1: 3, 20 ml). The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and suspended in THF (70 ml). The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (286 mg, 0.672 mmol) as a gray-violet powder. The mother liquor was crystallized from methanol and ethyl acetate to give the title compound (695 mg, 1.63 alcohol) as a grey-violet powder.
'H-NMR (300MHz, DMS0 - d6) : δ 1.79 (3Η, s), 2.17 (3H, s), 2.38 (3H, s), 3.07 (2H, t, J= 6.5 Hz), 3.60 (2H, brq, J= 4.2 Hz), 5.11 (1H, brt, J= 3 Hz), 7.37 (2H, d, J= 7.1 Hz), 7.85 (2H, d, J= 8.1 Hz), 11.63 (1H, brs). 'H-NMR (300MHz, DMS0-d 6 ): δ 1.79 (3Η, s), 2.17 (3H, s), 2.38 (3H, s), 3.07 (2H, t, J = 6.5 Hz), 3.60 (2H , brq, J = 4.2 Hz), 5.11 (1H, brt, J = 3 Hz), 7.37 (2H, d, J = 7.1 Hz), 7.85 (2H, d, J = 8.1 Hz), 11.63 (1H, brs ).
元素分析値: C17H19N304S3として 計算値: C, 47.98; H, 4.50; , 9.87; S, 22.61 (%) . Elementary analysis: as C 17 H 19 N 3 0 4 S 3 Calculated: C, 47.98; H, 4.50;, 9.87; S, 22.61 (%).
実測値: C, 47.95; H, 4.41; N, 9.77; S, 22.52 (¾) · Found: C, 47.95; H, 4.41; N, 9.77; S, 22.52 (¾) ·
mp. 214.0-216.0°C mp. 214.0-216.0 ° C
実施例 4-525 Example 4-525
4-クロ口- N- {8- [(2 -ヒドロキシェチル)スルファニル ]-5, 6-ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン }ベンゼンスルホン アミド 4-chloro-N- {8-[(2-hydroxyethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4 ] Thiazine-1-ylidene} benzenesulfonamide
実施例 3 - 6 で得た化合物(1.02 g, 2.54 mmol) および炭酸カリウム (702 mg, 5.08 mmol) の DMF (15ml) の懸濁液に 50°Cで攪拌しながら 2_ョ一ドエタノール (0.403 ml, 5.08匪 ol) の DMF (2 ml)溶液を 30分間かけて滴下し、 反応液をさ らに 50分間攪拌した。反応液に 1N塩酸 (8.0 ml) を加え、メタノール一水(1 :2, 15 ml)で希釈して 2時間攪拌した。析出物を濾取し、 メタノール一水(1:1)および メタノールで洗浄後、 THF (70 ml)に懸濁した。 析出物を濾取し、 酢酸ェチルで洗 浄後、 乾燥して表題化合物(778 mg, 1.74腿 ol, 68.7%)を紫色粉末として得た。 Ή一 NMR (300MHz, DMS0-d6) : δ 1.80 (3Η, s), 2.18 (3H, s), 3.08 (2H, t, J= 6.5 Hz), 3.61 (2H, t, J= 6.3 Hz), 5.12 (1H, br), 7.65 (2H, d, J= 9.0 Hz), 7.97 (2H, d, J= 9.0 Hz), 11.78 (1H, brs). To a suspension of the compound (1.02 g, 2.54 mmol) obtained in Example 3-6 and potassium carbonate (702 mg, 5.08 mmol) in DMF (15 ml) was added 2_ ethanol (50 ml) while stirring at 50 ° C. A solution of 0.403 ml, 5.08 bandol) in DMF (2 ml) was added dropwise over 30 minutes, and the reaction solution was further stirred for 50 minutes. 1N Hydrochloric acid (8.0 ml) was added to the reaction solution, diluted with methanol / water (1: 2, 15 ml) and stirred for 2 hours. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and suspended in THF (70 ml). The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (778 mg, 1.74 mol, 68.7%) as a purple powder. Ή one NMR (300MHz, DMS0-d 6 ): δ 1.80 (3Η, s), 2.18 (3H, s), 3.08 (2H, t, J = 6.5 Hz), 3.61 (2H, t, J = 6.3 Hz) , 5.12 (1H, br), 7.65 (2H, d, J = 9.0 Hz), 7.97 (2H, d, J = 9.0 Hz), 11.78 (1H, brs).
元素分析値: C16H16N304S3C1として ' 計算値: C, 43.09; H, 3.62; N, 9.42; S, 21.57; C1, 7.95 (¾) . Elemental analysis: C 16 H 16 N 3 0 4 as S 3 C1 'Calculated: C, 43.09; H, 3.62 ; N, 9.42; S, 21.57; C1, 7.95 (¾).
実測値: C, 42.78; H, 3.55; N, 9.40; S, 21.67; C1, 7.98 (¾) · Found: C, 42.78; H, 3.55; N, 9.40; S, 21.67; C1, 7.98 (¾)
即. 207.5-209.0 Immediately. 207.5-209.0
実施例 4- 526 Example 4-526
N- (5,6-ジメチル- 3-ォキソ -8- {[2- (メトキシェトキシ)メチル]スルファニ ル}- 2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデン)-4-メチルベ ンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[2- (methoxyethoxy) methyl] sulfanyl} -2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3- 4で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルファニル -2, 3-ジヒド ロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン)-4-メチルベンゼンスルホン アミ H (200 mg, 0.524 mmol)と Ν,Ν -ジイソプロピルェチルァミン(0.113 ml, 0.629 mmol) の N,N-ジメチルホルムアミド (4.00ml) の懸濁液に、 2- (メトキシェトキ シ)メチル クロリド (75.6 mg, 0.577腿 ol) を加えた。 反応混合物を室温で 20 分間かき混ぜた後、 酢酸ェチルで希釈し、 水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去して題記化合物 (72.7 mg, 30¾) を暗赤色結晶として得た。 酢酸ェチル- ジェチルェ一テルから再結晶して融点 91.0-93. (TCの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 Suspension of 1-ylidene) -4-methylbenzenesulfonamide H (200 mg, 0.524 mmol) and Ν, Ν-diisopropylethylamine (0.113 ml, 0.629 mmol) in N, N-dimethylformamide (4.00 ml) Add 2- (methoxyethoxy) to the solution B) Methyl chloride (75.6 mg, 0.577 t ol) was added. After stirring 20 minutes the reaction mixture at room temperature, diluted with acetic acid Echiru, washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (72.7 mg, 30¾) as a dark red crystals . The crystal was recrystallized from ethyl acetate-ethyl ether to give a dark red crystal having a melting point of 91.0-93. (TC.
IR (KBr) V: 3250, 2922, 1744, 1665, 1615, 1564 cm一 IR (KBr) V: 3250, 2922, 1744, 1665, 1615, 1564 cm
Ή-NMR (CDC13) : 61.79 (3H, s), 2.25 (3H, s), 2.42 (3Ή, s), 3.38 (3H, s), 3.54 (2H, t, J=4.8 Hz), 3.72 (2H, t, J=4.8 Hz), 5.09 (2H, s), 7.29 (2H, d, J=7.8 Hz), 7.84 (2H, d, J=8.4 Hz), 9.64 (1H, bs). , 元素分析値: C19H23N305S3として Ή-NMR (CDC1 3): 61.79 (3H, s), 2.25 (3H, s), 2.42 (3Ή, s), 3.38 (3H, s), 3.54 (2H, t, J = 4.8 Hz), 3.72 ( 2H, t, J = 4.8 Hz), 5.09 (2H, s), 7.29 (2H, d, J = 7.8 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.64 (1H, bs). as C 19 H 23 N 3 0 5 S 3: analysis
計算値 (¾) : C, 48.60; H, 4.94; N, 8.95; S, 20.48. Calculated value (¾): C, 48.60; H, 4.94; N, 8.95; S, 20.48.
実測値 (¾) : C, 48.24; H, 5.03; N, 8.95; S, 20.38. Found (¾): C, 48.24; H, 5.03; N, 8.95; S, 20.38.
実施例 4- 527 Example 4-527
N- (5, 6-ジメチル- 8-{3- [(2 -メトキシェトキシ)プロピル]スルファニル }- 3-ォキ ソ- 2, 3 -ジヒド口 1H-ィミダゾ [5, 1 - c] [1,4]チアジン -1-ィリデン) - 4-メチルベン ゼンスルホンアミド N- (5,6-Dimethyl-8- {3-[(2-methoxyethoxy) propyl] sulfanyl} -3-oxo-2,3-dihydrogen 1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene)-4-methylbenzene benzenesulfonamide
実施例 3-4で得られた - (5, 6-ジメチル- 3-ォキソ -8-スルファニル -2, 3-ジヒド 口- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン)-4-メチルベンゼンスルホン アミド (200 mg, 0.524 mmol) と炭酸カリウム (109 mg, 0.786 mmol) の N,N-ジ メチルホルムアミド(3.00ml)の懸濁液に、 3- (2-メトキシエトキシ)プロピル ブ ロミド (124 mg, 0.629匪 ol) の N, N-ジメチルホルムアミド (1.00 ml) 溶液を 加えた。 反応混合物を 80°Cで 15分間加熱した後、 1N塩酸を加えて酢酸ェチルで 抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (171 mg, 66¾) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 117- 119°Cの暗赤色結晶を得た。 -(5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrone-1H-imidazo [5, 1-c] [1,4] thiazine obtained in Example 3-4 To a suspension of (ylidene) -4-methylbenzenesulfonamide (200 mg, 0.524 mmol) and potassium carbonate (109 mg, 0.786 mmol) in N, N-dimethylformamide (3.00 ml) was added 3- (2-methoxy). A solution of ethoxy) propyl bromide (124 mg, 0.629 bandol) in N, N-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 ° C. for 15 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (171 mg, 66¾) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 117-119 ° C.
IR (KBr)ソ : 3227, 2921, 2878, 1738, 1663, 1615, 1559 cm"1. IR (KBr): 3227, 2921, 2878, 1738, 1663, 1615, 1559 cm " 1 .
¾-NM (CDC13) : <51.78 (3H, d, J-1.2 Hz), 1.91-2.03 (2H, m), 2.26 (3H, d, J=0.9 Hz), 2.42 (3H, s), 3.05 (2H, t, J=6.9 Hz), 3.39 (3H, s), 3.52-3.60 (6H, m), 7.29 (2H, d, J=8.1 Hz), 7.85 (2H, d, J=8. Hz), 9.59 (1H, bs). ¾-NM (CDC1 3): <51.78 (3H, d, J-1.2 Hz), 1.91-2.03 (2H, m), 2.26 (3H, d, J = 0.9 Hz), 2.42 (3H, s), 3.05 (2H, t, J = 6.9 Hz), 3.39 (3H, s), 3.52-3.60 (6H, m), 7.29 (2H, d, J = 8.1 Hz), 7.85 (2H, d, J = 8. Hz) ), 9.59 (1H, bs).
元素分析値: C21H27N305S3として 計算値 (%) : C, 50.68; H, 5.47; N, 8.44; S, 19.33. Elemental analysis: C 21 H 27 N 3 0 5 S 3 Calculated value (%): C, 50.68; H, 5.47; N, 8.44; S, 19.33.
実測値 (¾) : C, 50.57; H, 5.61; , 8.56; S, 19.28. Found (¾): C, 50.57; H, 5.61;, 8.56; S, 19.28.
実施例 4- 528 Example 4-528
N - (5, 6-ジメチル- 8- { [2- (4-フルォロブトキシ)ェチル]スルファ二ル} - 3 -ォキソ _2,3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン-卜イリデン )-4_メチルベンゼ ンスルホンアミド N- (5,6-dimethyl-8-{[2- (4-fluorobutoxy) ethyl] sulfanyl} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-triylidene) -4_methylbenzenesulfonamide
実施例 3- で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルファニル- 2, 3-ジヒド ロ- 1H-ィミダゾ [5, 1- c] [1,4]チアジン -1-ィリデン) -4-メチルべンゼンスルホン アミド (200 mg, 0.524 mmol) と炭酸カリウム (109 mg, 0.786腿 ol) の Ν,Ν-ジ メチルホルムアミド (3.00 ml) の懸濁液に、 参考例 81で得られたメタンスルホ ン酸 2- (4-フルォロブトキシ)ェチルエステル (135 ig, 0.629 mmol) の N,N -ジ メチルホルムアミド (l.OOml) 溶液を加えた。 反応混合物を 80°Cで 30分間加熱 した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (59.7 mg, 23¾) を暗赤色結晶として得 た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 107- 109 の暗赤色結晶を 得た。 , N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1 obtained in Example 3- Reference Example 81 was added to a suspension of メ チ ル, Ν-dimethylformamide (3.00 ml) of -ylidene) -4-methylbenzenesulfonamide (200 mg, 0.524 mmol) and potassium carbonate (109 mg, 0.786 tmol). A solution of 2- (4-fluorobutoxy) ethyl methanesulfonate (135 ig, 0.629 mmol) obtained in the above in N, N-dimethylformamide (l.OOml) was added. After the reaction mixture was heated at 80 ° C for 30 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (59.7 mg, 23¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 107-109. ,
IR (KBr) V: 3231, 2950, 2874, 1728, 1661, 1616, 1559 cm1. IR (KBr) V: 3231, 2950, 2874, 1728, 1661, 1616, 1559 cm 1 .
¾— NMR (CDC13) : 51.66-1.86 (4H, m), 1.79 (3H, d, J=0.9 Hz), 2.26 (3H, d, J=0.9 Hz), 2.42 (3H, s), 3.15 (2H, t, J=6.6Hz), 3.50 (2H, t, J=6.3Hz), 3.65 (2H, t, J=6.6 Hz), 4.46 (2H, dt, J=47.4, 6.0 Hz), 7.29 (2H, d, J=8.7 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). ¾- NMR (CDC1 3): 51.66-1.86 (4H, m), 1.79 (3H, d, J = 0.9 Hz), 2.26 (3H, d, J = 0.9 Hz), 2.42 (3H, s), 3.15 ( 2H, t, J = 6.6Hz), 3.50 (2H, t, J = 6.3Hz), 3.65 (2H, t, J = 6.6Hz), 4.46 (2H, dt, J = 47.4, 6.0Hz), 7.29 ( 2H, d, J = 8.7 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C21H26N304S3Fとして Elementary analysis: as C 21 H 26 N 3 0 4 S 3 F
計算値 (%) : C, 50.48; H, 5.25; N, 8.41; S, 19.25. Calculated (%): C, 50.48; H, 5.25; N, 8.41; S, 19.25.
実測値 (¾) : C, 50.22; H, 4.81; N, 8.80; S, 19.39. Found (¾): C, 50.22; H, 4.81; N, 8.80; S, 19.39.
実施例 4- 529 Example 4- 529
N- {6-ェチル- 8- [ (5-フルォ口ぺンチル)スルファニル] -5 -メチル- 3-ォキ V-2, 3 - ジヒドロ- 1H-イミダゾ [5, 1 - c] [1,4]チアジン-卜ィリデン }- 4_メチルベンゼンス ルホンアミド N- {6-ethyl-8-[(5-fluoropentyl) sulfanyl] -5-methyl-3-oxoV-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] thiazine-triylidene} -4_methylbenzenesulfonamide
実施例 3-5で得られた N- (6-ェチル -5-メチル- 3-ォキソ -8-スルファニル -2, 3 - ジヒドロ- 1H-イミダゾ [5,l_c] [1,4]チアジン -1-ィリデン)-4-メチルベンゼンス ルホンアミド (770 mg, 1.96讓 ol) と炭酸カリウム (406 mg, 2.93蘭01) の N,N - ジメチルホルムアミド (15.0ml) の懸濁液に、参考例 61で得られた 1 -プロモ- 5 - フルォロペンタン (364 mg, 2.15匪 ol) の N, N-ジメチルホルムアミド (5.00 ml) 溶液を加えた。反応混合物を 80t:で 1時間加熱した後、 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (967 mg, ca. 100%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 156- 158°Cの暗赤色結晶を得た。 N- (6-Ethyl-5-methyl-3-oxo-8-sulfanyl -2,3-obtained in Example 3-5 Dihydro-1H-imidazo [5, l_c] [1,4] thiazin-1-ylidene) -4-methylbenzenesulfonamide (770 mg, 1.96 alcohol) and potassium carbonate (406 mg, 2.93 orchid 01) N, To a suspension of N-dimethylformamide (15.0 ml) was added a solution of 1-promo-5-fluoropentane (364 mg, 2.15 bandol) obtained in Reference Example 61 in N, N-dimethylformamide (5.00 ml). Was. After heating the reaction mixture at 80 t: for 1 hour, 1N hydrochloric acid was added and extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was distilled off under reduced pressure the title of compound (967 mg, ca. 100%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 156-158 ° C.
IR (KBr) : 3218, 2967, 2934, 2868, 1738, 1659, 1617, 1557 cm— IR (KBr): 3218, 2967, 2934, 2868, 1738, 1659, 1617, 1557 cm—
¾一 NMR (CDC13) : 61.09 (3H, d, J=7.6 Hz), 1.50-1.88 (6H, m), 2.14 (2H, q, J=7.4Hz), 2.26 (3H, s), 2.42 (3H, s), 2.96 (2H, i, J=7.4 Hz), 4.45 (2H, dt, J=47.2, 6.0 Hz), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.62 (1H, bs). ¾ one NMR (CDC1 3): 61.09 ( 3H, d, J = 7.6 Hz), 1.50-1.88 (6H, m), 2.14 (2H, q, J = 7.4Hz), 2.26 (3H, s), 2.42 ( 3H, s), 2.96 (2H, i, J = 7.4 Hz), 4.45 (2H, dt, J = 47.2, 6.0 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.62 (1H, bs).
元素分析値: C21H26N303S3Fとして Elementary analysis: as C 21 H 26 N 3 0 3 S 3 F
計算値 (¾) : C, 52.15; H, 5.42; N, 8.69; S, 19.89. Calculated value (¾): C, 52.15; H, 5.42; N, 8.69; S, 19.89.
実測値 (¾) : C, 51.89; H, 5.41; N, 8.50; S,.19.74. Found (¾): C, 51.89; H, 5.41; N, 8.50; S, .19.74.
実施例 4-530 Example 4-530
N- {6-プチル- 8- [ (5-フルォロぺンチル)スルファニル] -5 -メチル- 3-ォキソ- 2, 3- ジヒドロ - 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン メチルベンゼンス ルホンアミド N- {6-butyl-8-[(5-fluoropentyl) sulfanyl] -5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1-ylidene methylbenzenesulfonamide
実施例 3-15で得られた N - (6-ェチル- 5-メチル- 3-ォキソ- 8-スルファニル _2, 3- ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン ) -4-メチルベンゼンス ルホンアミ ド (2.00g, 4.72mmol) と炭酸カリウム (979 mg, 5.19匪01) の Ν,Ν- ジメチルホルムアミド (40.0ml)の懸濁液に、参考例 61で得られた卜ブロモ -5- フルォロペンタン (878 mg, 5.19匪 ol) の N, N-ジメチルホルムアミド (10.0 ml) 溶液を加えた。反応混合物を 80°Cで 1時間加熱した後、 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (2.39 g, 99¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して 融点 170- 172°Cの暗赤色結晶を得た。 IR (KBr) v: 3262, 2957, 2934, 2868, 1734, 1657, 1617, 1561 cm一1. N- (6-Ethyl-5-methyl-3-oxo-8-sulfanyl_2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-15 A suspension of 1-ylidene) -4-methylbenzenesulfonamide (2.00 g, 4.72 mmol) and potassium carbonate (979 mg, 5.19 band01) in Ν, Ν-dimethylformamide (40.0 ml) was added to Reference Example 61. A solution of tribromo-5-fluoropentane (878 mg, 5.19 bandol) obtained in the above in N, N-dimethylformamide (10.0 ml) was added. After heating the reaction mixture at 80 ° C for 1 hour, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title of compound a (2.39 g, 99¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 170-172 ° C. IR (KBr) v: 3262, 2957, 2934, 2868, 1734, 1657, 1617, 1561 cm one 1.
'H-NMR (CDC13) : δ 0.929 (3H, t, J=6.9Hz), 1.30-1.62 (6H, m), 1.62-1.72 (4H, m), 2.12 (2H, t, J=8.1 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.96 (2H, t, J=7.5 Hz), 4.45 (2H, dt, J=47.21, 5.7 Hz), 7.29 (2H, d, J=8.7 Hz), 7.84 (2H, d, J=8.1 Hz), 9.62 (1H, bs) . 'H-NMR (CDC1 3) : δ 0.929 (3H, t, J = 6.9Hz), 1.30-1.62 (6H, m), 1.62-1.72 (4H, m), 2.12 (2H, t, J = 8.1 Hz ), 2.26 (3H, s), 2.42 (3H, s), 2.96 (2H, t, J = 7.5 Hz), 4.45 (2H, dt, J = 47.21, 5.7 Hz), 7.29 (2H, d, J = 8.7 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.62 (1H, bs).
元素分析値: C23H3。N303S3Fとして Elemental analysis: C 23 H 3. N 3 0 3 S 3 F
計算値 (%) : C, 53.99; H, 5.91; N, 8.21; S, 18.80. ' 実測値 (¾) : C, 53.85; H, 5.85; N, 8.01; S, 18.71. Calculated value (%): C, 53.99; H, 5.91; N, 8.21; S, 18.80. 'Actual value (¾): C, 53.85; H, 5.85; N, 8.01; S, 18.71.
実施例 4-531 Example 4-531
N-(5, 6-ジメチル - 3_ォキソ -8-{[5_(2, 2, 2-トリフルォロエトキシ)ペンチル]スル ファニル}-2,3-ジヒドロ- m-イミダゾ [5, 1-c] [1,4]チアジン -1-ィリデン)-4-メ 実施例 3-4で得られた N -(2, 3-ジヒドロ- 5, 6 -ジメチル- 3-ォキソ -8-スルファ二 ル- 1H-イミダゾ [5,1- c] [1,4]チアジン-卜ィリデン)-4-メチルベンゼンスルホン アミド 960 mg, 2.52匪01) と炭酸カリウム (522 mg, 3.77 mmol) の N,N-ジメチ ルホルムアミド(15.0 ml)の懸濁液に、参考例 73で得られた 1_ブロモ - 5-(2, 2, 2_ 卜リフルォロェ卜キシ)ペン夕ン (1.57 g, 6.30 mmol) の N, N -ジメチルホルムァ ミド (5.00 ml) 溶液を加えた。 反応混合物を 80°Cで 20分間加熱した後、 1N塩酸 を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去して題記化合物 (1.01 g, 73%) を暗赤色結晶として得た。 酢酸ェチル-ジ ェチルエーテルから再結晶して融点 153_155°Cの暗赤色結晶を得た。 N- (5,6-dimethyl-3_oxo-8-{[5_ (2,2,2-trifluoroethoxy) pentyl] sulfanyl} -2,3-dihydro-m-imidazo [5,1- c] [1,4] Thiazine-1-ylidene) -4-me N- (2,3-dihydro-5,6-dimethyl-3-oxo-8-sulfanyl obtained in Example 3-4 -1H-imidazo [5,1-c] [1,4] thiazine-triylidene) -4-methylbenzenesulfonamide 960 mg, 2.52 maraudal 01) and potassium carbonate (522 mg, 3.77 mmol) N, N- To a suspension of dimethylformamide (15.0 ml) was added 1_bromo-5- (2,2,2_trifluoroethoxy) pentyne (1.57 g, 6.30 mmol) obtained in Reference Example 73 as N, An N-dimethylformamide (5.00 ml) solution was added. After the reaction mixture was heated at 80 ° C for 20 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (1.01 g, 73%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 153-155 ° C.
IR (KBr) V: 3133, 3061, 2936, 1744, 1728, 1661, 1615, 1557 cm— IR (KBr) V: 3133, 3061, 2936, 1744, 1728, 1661, 1615, 1557 cm—
Ή—NMR (CDC13) : 51.44-1.84 (6H, m), 1.79 (3H, s), 2.26 (3H, d, J=0.8 Hz), 2.42 (3H, s), 2.94 (2H, t, J=7.6 Hz), 3.61 (2H, t, J=6.2 Hz) , 3.81 (2H, q, J=8.8 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.62 (1H, bs). 元素分析値: C22H26N304S3F3として Ή-NMR (CDC1 3): 51.44-1.84 (6H, m), 1.79 (3H, s), 2.26 (3H, d, J = 0.8 Hz), 2.42 (3H, s), 2.94 (2H, t, J = 7.6 Hz), 3.61 (2H, t, J = 6.2 Hz), 3.81 (2H, q, J = 8.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz) Hz), 9.62 (1H, bs ) Elementary analysis:. as C 22 H 26 N 3 0 4 S 3 F 3
計算値 ) : C, 48.07; H, 4.77; N, 7.64; S, 17.50. Calculated)): C, 48.07; H, 4.77; N, 7.64; S, 17.50.
実測値 (%) : C, 48.08; H, 4.53; N, 7.59; S, 17.53. Obtained value (%): C, 48.08; H, 4.53; N, 7.59; S, 17.53.
実施例 4- 532 N-(5, 6-ジメチル- 3-ォキソ -8- {[4- (2, 2, 2-トリフルォロエトキシ)プチル]スルフ ァニル }- 2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン) - 4-メチ 実施例 3-4で得られた N- (2, 3 -ジヒドロ _5, 6-ジメチル- 3-ォキソ -8-スルファニ ル- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン)-4_メチルベンゼンスルホン アミ ド (1.00 g, 2.62 匪 ol) と炭酸カリウム (543 mg, 3.92 匪 ol) の N,N -ジメ チルホルムアミド (15.0 ml) の懸濁液に、 参考例 ΊΪ で得られた 1-ブロモ -4- (2, 2, 2-トリフルォロエトキシ)ブ夕ン (1.54 g, 6.55 腿 ol) の N,N-ジメチル ホルムアミド(5.00 ml)溶液を加えた。反応混合物を で 20分間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (1.34g, 96¾) を暗赤色結晶として得た。 酢酸ェ チル-ジェチルエーテルから再結晶して融点 144-146°Cの暗赤色結晶を得た。 IR (KBr) V: 3229, 2922, 1750, 1719, 1661, 1619, 1561 cm1. Example 4-532 N- (5,6-dimethyl-3-oxo-8-{[4- (2,2,2-trifluoroethoxy) butyl] sulfanyl} -2,3-dihydro-1H-imidazo [5, c] [1,4] thiazine-1-ylidene) -4-methyl N- (2,3-dihydro_5,6-dimethyl-3-oxo-8-sulfanyl-1H obtained in Example 3-4. Of N-imidazo [5,1-c] [1,4] thiazine-1-ylidene) -4_methylbenzenesulfonamide (1.00 g, 2.62 bandol ol) and potassium carbonate (543 mg, 3.92 bandol ol) To a suspension of N-dimethylformamide (15.0 ml) was added 1-bromo-4- (2,2,2-trifluoroethoxy) butane (1.54 g, 6.55 t ) In N, N-dimethylformamide (5.00 ml) was added. After heating the reaction mixture with for 20 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (1.34g, 96¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 144-146 ° C. IR (KBr) V: 3229, 2922, 1750, 1719, 1661, 1619, 1561 cm 1 .
Ή-NMR (CDC13) : 51.62-1.90 (4H, m), 1.78 (3H, d, J=1.2 Hz), 2.26 (3H, d, J=0.8 Hz), 2.42 (3H, s), 2.97 (2H, t, J=6.8Hz), 3.63 (2H, t, J-5.4Hz), 3.81 (2H, q, J=8.8 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.0 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): 51.62-1.90 (4H, m), 1.78 (3H, d, J = 1.2 Hz), 2.26 (3H, d, J = 0.8 Hz), 2.42 (3H, s), 2.97 ( 2H, t, J = 6.8Hz), 3.63 (2H, t, J-5.4Hz), 3.81 (2H, q, J = 8.8 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.61 (1H, bs).
元素分析値: C2IH24N304S3F3として Elementary analysis: as C 2I H 24 N 3 0 4 S 3 F 3
計算値 (%) : C, 47.09; H, 4.52; N, 7.85; S, 17.96. Calculated value (%): C, 47.09; H, 4.52; N, 7.85; S, 17.96.
実測値 (%) : C, 47.29; H, 4.33; N, 8.06; S, 18.32. Found (%): C, 47.29; H, 4.33; N, 8.06; S, 18.32.
実施例 4- 533 Example 4-533
N- (5, 6-ジメチル -3 -ォキソ -8- { [2- (3 -フルォロプロポキシ)ェチル]スルファニ ル}_2,3-ジヒドロ- 1H-イミダゾ [5,1-c] [1, 4]チアジン - 1 -ィリデン)-4-メチルベ ンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[2- (3-fluoropropoxy) ethyl] sulfanyl} _2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3 - 4で得られた N- (5, 6-ジメチル- 3-ォキソ - 8_スルファニル -2, 3 -ジヒド 口- 1H -イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン)-4-メチルベンゼンスルホン アミ ド (269 mg, 0.706 匪 ol) と炭酸カリウム (146 mg, 1.06 mmol) の N,N -ジ メチルホルムアミド (4.00 ml) の懸濁液に、 参考例 80で得られたメタンスルホ ン酸 2- (3-フルォロプロポキシ)ェチルエステル (320 mg, 1.77讓 ol) の N,N -ジ メチルホルムアミド (1.00ml) 溶液を加えた。 反応混合物を 80 で 10分間加熱 した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n -へキサン-酢酸ェチル (10:1, v/v)で溶出して題記化合物 (34.7 mg, 10%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 144- 6°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8_sulfanyl-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine obtained in Example 3-4 A reference example was prepared using a suspension of 1-ylidene) -4-methylbenzenesulfonamide (269 mg, 0.706 ol) and potassium carbonate (146 mg, 1.06 mmol) in N, N-dimethylformamide (4.00 ml). N, N-Dimethylmethane-2- (3-fluoropropoxy) ethyl ester (320 mg, 1.77 acetyl) obtained in Step 80 A solution of methylformamide (1.00 ml) was added. After the reaction mixture was heated at 80 for 10 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (34.7 mg, 10%) as dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 144-6 ° C.
IR (KBr) V: 3243, 2969, 2928, 2905, 2872, 1748, 1721, 1661, 1615, 1557 cm— Ή-NMR (CDC13) : 51.79 (3H, d, J=1.0 Hz), 1.93 (2H, dt, J=25.6, 6.2 Hz), 2.26 (3H, d, J=1.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J-6.2 Hz), 3.57 (2H, t, J=5.8 Hz), 3.66 (2H, t, J=6.2Hz), 4.53 (2H, dt, J=47.4, 6.0 Hz), 7.28 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). IR (KBr) V: 3243, 2969, 2928, 2905, 2872, 1748, 1721, 1661, 1615, 1557 cm- Ή-NMR (CDC1 3): 51.79 (3H, d, J = 1.0 Hz), 1.93 (2H , dt, J = 25.6, 6.2 Hz), 2.26 (3H, d, J = 1.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J-6.2 Hz), 3.57 (2H, t, J = 5.8 Hz), 3.66 (2H, t, J = 6.2 Hz), 4.53 (2H, dt, J = 47.4, 6.0 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
元素分析値: C2。H24N304S3Fとして Elemental analysis: C 2. As H 24 N 3 0 4 S 3 F
計算値 (¾) : C, 49.47; H, 4.98; N, 8.65; S, 19.81. Calculated (¾): C, 49.47; H, 4.98; N, 8.65; S, 19.81.
実測値 (¾) : C, 49.34; H, 4.92; N, 8.63; S, 19.98. Found (¾): C, 49.34; H, 4.92; N, 8.63; S, 19.98.
実施例 4- 534 Example 4-534
N-(5, 6-ジメチル -3-ォキソ _8-{[2-(5-フルォロペンチルォキシ)ェチル]スルファ 二ル 2, 3-ジヒドロ -1H-イミダゾ [5, 1 - c] [1,4]チアジン- 1 -ィリデン )-4 -メチル ベンゼンスルホンアミド N- (5,6-dimethyl-3-oxo_8-{[2- (5-fluoropentyloxy) ethyl] sulfanyl 2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得られた N-(5, 6-ジメチル- 3-ォキソ -8-スルファニル -2, 3 -ジヒド 口- 1H-イミダゾ [5,1- c] [1, 4]チアジン- 1-ィリデン)-4-メチルベンゼンスルホン アミド (1.00 g, 2.62讓 ol) と炭酸カリウム (543 mg, 3.93匪 ol) の N,N -ジメ チルホルムアミド (15.0 ml) の懸濁液に、 参考例 82で得られたメタンスルホン 酸 2-(5-フルォロペンチルォキシ)ェチルエステル (898 mg, 3.93匪01) の N,N- ジメチルホルムアミド (5.00 ml) 溶液を加えた。 反応混合物を 80°Cで 1.5時間 加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (1.23g, 94%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 119- 12ΐ の暗赤色結晶を得た。 IR (KBr) リ : 3241, 2940, 2865, 1748, 1721, 1663, 1615, 1559 cm"1. N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 Example of a suspension of 1-ylidene) -4-methylbenzenesulfonamide (1.00 g, 2.62 benzylol) and potassium carbonate (543 mg, 3.93 melamineol) in N, N-dimethylformamide (15.0 ml). A solution of 2- (5-fluoropentyloxy) ethyl methanesulfonate (898 mg, 3.93 band01) obtained in 82 in N, N-dimethylformamide (5.00 ml) was added. After heating the reaction mixture at 80 ° C. for 1.5 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (1.23 g, 94%) as dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 119-12 °. IR (KBr): 3241, 2940, 2865, 1748, 1721, 1663, 1615, 1559 cm " 1 .
'H-NMR (CDC13) : 61.40-1.80 (6H, m), 1.79 (3H, d, J=1.2 Hz), 2.26 (3H, d, J=0.9 Hz), 2.42 (3H, s), 3.14 (2H, t, J=6.9 Hz), 3.46 (2H, t, J=6.3Hz), 3.64 (2H, t, J=6.6 Hz), 4.45 (2H, dt, J=47.1, 6.0 Hz), 7.29 (2H, d, J=7.8 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). 'H-NMR (CDC1 3) : 61.40-1.80 (6H, m), 1.79 (3H, d, J = 1.2 Hz), 2.26 (3H, d, J = 0.9 Hz), 2.42 (3H, s), 3.14 (2H, t, J = 6.9 Hz), 3.46 (2H, t, J = 6.3 Hz), 3.64 (2H, t, J = 6.6 Hz), 4.45 (2H, dt, J = 47.1, 6.0 Hz), 7.29 (2H, d, J = 7.8 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, bs).
兀素分析値: C22H28N304S3Fとして 兀素analysis: as C 22 H 28 N 3 0 4 S 3 F
計算値 (¾) C, 51.44; H, 5.49; N, 8.18; S, 18.73. Calculated (¾) C, 51.44; H, 5.49; N, 8.18; S, 18.73.
実測値 (¾) C, 51.30; H, 5.35; N, 8.18; S, 18.93. Found (¾) C, 51.30; H, 5.35; N, 8.18; S, 18.93.
実施例 4-535 Example 4-535
N-{8-[(4, 4-ジフルォロシクロへキシル)スルファニル ]-5, 6-ジメチル -3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン卜 4-メチルベンゼ ンスルホンアミド , N- {8-[(4,4-difluorocyclohexyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -Trident 4-methylbenzenesulfonamide,
実施例 3-4で得た化合物(2.49 g, 6.53讓 ol)および炭酸カリウム (1.35g, 9.79 腿 ol) の DMF (50 ml) 懸濁液に参考例 21.得られた 4, 4-ジフルォ口-卜ョードシ クロへキサン(純度 45%, 4.9 g, 9.0藤 ol) の DMF (50 ml) 溶液を 60t:で 2時 間かけて滴下し、反応液を 11時間攪拌した。反応液に 1N塩酸 (13.1ml)、水 (15 ml)およびメタノール(15 ml)を加えた。 析出物を濾取してメタノール—水(1:1) およびメタノールで洗浄後、 乾燥して得た粉末 (2.14 g)を酢酸ェチルから結晶化 し、 酢酸ェチルおよびジェチルェ一テルで洗浄後、 乾燥して赤紫色粉末(1.35 g) を得た。 これを酢酸ェチル -THFから結晶化して表題化合物(469 mg, 0.939匪 ol) を紫色粉末として得た。 Reference Example 21. A suspension of the compound (2.49 g, 6.53 alcohol) obtained in Example 3-4 and potassium carbonate (1.35 g, 9.79 thigh) in DMF (50 ml) was prepared. A solution of mouth cyclohexane (purity 45%, 4.9 g, 9.0 tol) in DMF (50 ml) was added dropwise at 60 t: over 2 hours, and the reaction solution was stirred for 11 hours. To the reaction solution, 1N hydrochloric acid (13.1 ml), water (15 ml) and methanol (15 ml) were added. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried. The powder (2.14 g) obtained was crystallized from ethyl acetate, washed with ethyl acetate and geethyl ether, and dried. This gave a reddish purple powder (1.35 g). This was crystallized from ethyl acetate-THF to give the title compound (469 mg, 0.939 ol) as a purple powder.
Ή-NMR (300MHz, CDC13) : (51.79 (3H, s), 1.84 (2H, m), 2.10 (4H, brq, J= 9.3 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.43 (3H, s), 3.47 (1H, m), 7.30 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.7 Hz), 9.65 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): (51.79 (3H, s), 1.84 (2H, m), 2.10 (4H, brq, J = 9.3 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.43 (3H, s), 3.47 (1H, m), 7.30 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.7 Hz), 9.65 (1H, brs).
元素分析値: C21H23N303S3F2として計算値: C, 50.48; H, 4.61; , 8.41; S, 19.25; F, 7.61 (%) 、 実測値: C, 50.55; H, 4.63; N, 8.41; S, 19.34; F, 7.13 (%) . 即. 201.0-201.5で Elemental analysis: C 21 H 23 N 3 0 3 S 3 F 2 Calculated: C, 50.48; H, 4.61;, 8.41; S, 19.25; F, 7.61 (%), found: C, 50.55; H , 4.63; N, 8.41; S, 19.34; F, 7.13 (%). Immediately.
実施例 4-536 Example 4-536
N-{8-[ (4, 4 -ジフルォロシク口へキシル)メチル]スルファ二ル} -5, 6」ジメチル -3- ォキソ -2,3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン- 1-ィリデン)-4-メチル ベンゼンスルホンアミド N- {8-[(4,4-Difluorocyclohexyl) methyl] sulfanyl} -5,6 "dimethyl-3- Oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物(3.31 g, 8.68 IMOI)および炭酸カリウム (1.80 g, 13.0 mmol) の DMF (60 ml) 懸濁液に参考例 22で得られた 4, 4-ジフルォロ- 1-ョ一ド メチルシクロへキサン(51%酢酸ェチル溶液, 5.6 g, 11 龍 ol) の DMF (5 ml) 溶 液を 60°Cで 1.5時間かけて滴下し、 反応液を 12時間攪拌した。 反応液に 1 N塩 酸 (19ml)、 水 (26 ml)およびメタノール(20 ml)を加えて 1時間攪拌した。 析出 物を濾取してメタノ一ルー水(1: 1)およびメ夕ノールで洗浄後、乾燥して赤紫色粉 末(4.00 g)を得た。 これを酢酸ェチル -THF から結晶化して表題化合物(0.81 g, 1.58 imnol)を紫色粉末として得た。 4,4-Difluoro-1 obtained in Reference Example 22 was added to a suspension of the compound obtained in Example 3-4 (3.31 g, 8.68 IMOI) and potassium carbonate (1.80 g, 13.0 mmol) in DMF (60 ml). A solution of -methylcyclohexane (51% ethyl acetate solution, 5.6 g, 11 R ol) in DMF (5 ml) was added dropwise at 60 ° C over 1.5 hours, and the reaction solution was stirred for 12 hours. To the reaction solution were added 1N hydrochloric acid (19 ml), water (26 ml) and methanol (20 ml), and the mixture was stirred for 1 hour. The precipitate was collected by filtration, washed with methanolic water (1: 1) and methanol, and dried to obtain a magenta powder (4.00 g). This was crystallized from ethyl acetate-THF to give the title compound (0.81 g, 1.58 imnol) as a purple powder.
'H-NM (300MHz, CDC13) : 51.33 (2H, brq, J= 12.3 Hz), 1.62 (2H, m), 1.74 (1H, m), 1.79 (3H, q, J= 1.2 Hz), 1.92 (2H, brd, J= 13.8 Hz), 2.10 (2H, m), 2.26 (3H, q, J= 0.9 Hz), 2.42 (3H, s), 2.86 (2H, d, J= 6.9 Hz), 7.29 (2H, d, J= 8.7 Hz), 7.85 (2H, d, J= 8.7 Hz), 9.63 (1H, brs). 'H-NM (300MHz, CDC1 3): 51.33 (2H, brq, J = 12.3 Hz), 1.62 (2H, m), 1.74 (1H, m), 1.79 (3H, q, J = 1.2 Hz), 1.92 (2H, brd, J = 13.8 Hz), 2.10 (2H, m), 2.26 (3H, q, J = 0.9 Hz), 2.42 (3H, s), 2.86 (2H, d, J = 6.9 Hz), 7.29 (2H, d, J = 8.7 Hz), 7.85 (2H, d, J = 8.7 Hz), 9.63 (1H, brs).
元素分析値: C22H25N303S3F2として計算値: C, 51.44; H, 4.91; N, 8.18; S, 18.73; F, 7.40 (%) 、 実測値: C, 51.63; H, 4.88; N, 8.22; S, 18.77; F, 6.91 (%) . 即. 207.0- 207.5°C Elemental analysis: C 22 H 25 N 3 0 3 S 3 F 2 Calculated: C, 51.44; H, 4.91 ; N, 8.18; S, 18.73; F, 7.40 (%), Found: C, 51.63; H, 4.88; N, 8.22; S, 18.77; F, 6.91 (%). Immediately. 207.0-207.5 ° C
実施例 4-537 ' Example 4-537 '
N_(5, 6-ジメチル- 3_ォキソ -8- {[2- (プロボキシ)ェチル]スルファニル卜 2, 3-ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン)_4-メチルベンゼンスルホ ンアミド , 実施例 4-533と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 147- 149 の暗赤色結晶を得た。 N_ (5,6-Dimethyl-3_oxo-8-{[2- (propoxy) ethyl] sulfanyl chloride 2,3-dihydro-1H-imidazo [5, tri c] [1,4] thiazine-1- (Ylidene) _4-methylbenzenesulfonamide, synthesized in the same manner as in Example 4-533, and recrystallized from ethyl acetate-dimethyl ether to obtain dark red crystals having a melting point of 147-149.
IR (KBr) V: 3258, 2963, 2932, 2870, 1748, 1721,, 1663, 1611, 1557 cm"1. ¾-NMR (CDC13) : 50.921 (3H, t , ]=7.2 Hz), 1.53-1.66 (2H, m), 1.79 (3H, d, J=0.9 Hz), 2.26 (3H, d, J=l.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J=6.6 Hz), 3.41 (2H, t, J=6.6Hz), 3.64 (2H, t, J=6.3 Hz), 7.27 (2H, d, J=8.1 Hz), 7.85 (2H, d, J=8.1 Hz), 9.62 (1H, bs). IR (KBr) V:. 3258 , 2963, 2932, 2870, 1748, 1721 ,, 1663, 1611, 1557 cm "1 ¾-NMR (CDC1 3): 50.921 (3H, t,] = 7.2 Hz), 1.53- 1.66 (2H, m), 1.79 (3H, d, J = 0.9 Hz), 2.26 (3H, d, J = l.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J = 6.6 Hz) ), 3.41 (2H, t, J = 6.6 Hz), 3.64 (2H, t, J = 6.3 Hz), 7.27 (2H, d, J = 8.1 Hz), 7.85 (2H, d, J = 8.1 Hz), 9.62 (1H, bs).
元素分析値: C2QH25N304S3として 計算値 (¾) : C, 51.37; H, 5.39; N, 8.94; S, 20.62. Elementary analysis: as C 2Q H 25 N 3 0 4 S 3 Calculated value (¾): C, 51.37; H, 5.39; N, 8.94; S, 20.62.
実測値 (¾) : C, 51.09; H, 5.39; , 8.94; S, 20.62. Found (¾): C, 51.09; H, 5.39;, 8.94; S, 20.62.
実施例 4- 538 Example 4-538
N-[5, 6-ジメチル - 3_ォキソ - 8_({[4- (テトラヒドロ - 2H-ピラン - 2_ィルォキシ)シク 口へキシル]メチル }スルファニル )-2, 3-ジヒドロ- 1H -イミダゾ [5, 1- c] [1,4]チア ジン- 1-ィリデン ]-4-メチルベンゼンスルホンアミド N- [5,6-dimethyl-3_oxo-8 _ ({[4- (tetrahydro-2H-pyran-2_yloxy) cyclohexyl] methyl] sulfanyl) -2,3-dihydro-1H-imidazo [ 5, 1-c] [1,4] thiazin-1-ylidene] -4-methylbenzenesulfonamide
実施例 3-4 で得た化合物(3.02 g, 7.92 讓 ol) および炭酸カリウム (1.64 g, 11.9 mmol) の DMF (45 ml) の懸濁液に攪拌しながら参考例 23 で得たヨウ化 cis- 4- (2-テトラヒドロビラニル)ォキシシクロへキシルメチル(2.70 g, 8.33 腿 ol) の DMF (5 ml)溶液を 90分間で滴下し、反応液を 60°Cで 3.5 時間攪拌した。 反応中にヨウ化物 (0.257 g, 0.792 mmol) を 2回加え、 さらに 40°Cで 60時間攪 拌した。 反応液に 1N塩酸 (15.8 ml), 水(200 ml)および飽和食塩水 (30 ml)を加 え、酢酸ェチル(200 ml)で 1回抽出した。有機層を水および飽和食塩水で洗浄し、 硫酸ナトリウムで乾燥後、 濃縮した。 残渣の油状物(5.0 g)をシリカゲル(50 g) のクロマトグラフィー (酢酸ェチル—へキサン、 2:8-4:6) に付し、 酢酸ェチルー へキサン (4: 6) の画分を濃縮乾固後、 ジェチルエーテルで希釈した。 析出物を濾 取し、ジェチルェ一テルで洗浄後、乾燥して表題化合物(2.18 g, 3.77腿 ol, 7.6%) を赤色粉末として得た。 母液および洗液を濃縮乾固して表題化合物 (1.15 g, 1.99 讓 ol, 25. \%)を赤色固形物として得た。 The cis iodide obtained in Reference Example 23 was stirred in a suspension of the compound obtained in Example 3-4 (3.02 g, 7.92 alcohol) and potassium carbonate (1.64 g, 11.9 mmol) in DMF (45 ml) while stirring. -A solution of 4- (2-tetrahydroviranyl) oxycyclohexylmethyl (2.70 g, 8.33 tmol) in DMF (5 ml) was added dropwise over 90 minutes, and the reaction solution was stirred at 60 ° C for 3.5 hours. During the reaction, iodide (0.257 g, 0.792 mmol) was added twice, and the mixture was further stirred at 40 ° C for 60 hours. 1N Hydrochloric acid (15.8 ml), water (200 ml) and saturated saline (30 ml) were added to the reaction solution, and the mixture was extracted once with ethyl acetate (200 ml). The organic layer was washed with water and saturated saline, dried over sodium sulfate, and concentrated. The residue oil (5.0 g) was chromatographed on silica gel (50 g) (ethyl acetate-hexane, 2: 8-4: 6), and the ethyl acetate-hexane (4: 6) fraction was concentrated. After drying, it was diluted with getyl ether. The precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (2.18 g, 3.77 mol, 7.6%) as a red powder. The mother liquor and the washings were concentrated to dryness to give the title compound (1.15 g, 1.99 mL, 25. \%) as a red solid.
—NMR (200MHz, CDC13) : 51.41 (2H, brq, J= 8.0 Hz), 1.5-1.7 (10H, m), 1.70 (1H, m), 1.79 (3H, brq, J= 0.6 Hz), 1.84 (2H, m), 2.26 (3H, s), 2.41 (3H, s), 2.86 (2H, d, J= 5.8 Hz), 3.49 (1H, dt, J= 11.0, 4.7 Hz) , 3.88 (2H, m), 4.64 (1H, brs), 7.28 (2H, d, J- 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). -NMR (200MHz, CDC1 3): 51.41 (2H, brq, J = 8.0 Hz), 1.5-1.7 (10H, m), 1.70 (1H, m), 1.79 (3H, brq, J = 0.6 Hz), 1.84 (2H, m), 2.26 (3H, s), 2.41 (3H, s), 2.86 (2H, d, J = 5.8 Hz), 3.49 (1H, dt, J = 11.0, 4.7 Hz), 3.88 (2H, m), 4.64 (1H, brs), 7.28 (2H, d, J-8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C27¾5N305S3として計算値: C, 56.13; H, 6.11; N, 7.27; S, 16.65 (%) 、 実測値 (%) : C, 56.15; H, 6.12; N, 7.41; S, 16.64. Elemental analysis: C 27と し て5 N 3 0 5 S 3 Calculated: C, 56.13; H, 6.11; N, 7.27; S, 16.65 (%), found (%): C, 56.15; H, 6.12 ; N, 7.41; S, 16.64.
mp. 144.5-146.5°C mp.144.5-146.5 ° C
実施例 4-540 Example 4-540
酢酸 5- [(5, 6 -ジメチル -1 - {[(4 -メチルフエニル)スルホニル]イミノ}-3 -ォキソ -I, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1, ]チアジン- 8-ィル)スルファニル]シクロ ォクチル Acetic acid 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo -I, 3-Dihydrido-1H-imidazo [5, 1-c] [1,] thiazin-8-yl) sulfanyl] cyclooctyl
実施例 3-4で得た化合物(566 mg, 1.48腿 ol) および炭酸カリウム (0.41 g, 2.97讓01) の DMF (9 ml) の懸濁液に 80°Cで攪拌しながら参考例 24で得たヨウ 化 cis_5-ァセトキシシクロォクチル(0.81 g, 1.6腿 ol) の DMF (5 ml)溶液を 90 分間かけて滴下し、反応液をさらに 1.5時間攪拌した。反応液に 1N塩酸 (3.5ml), 0. IN塩酸(15 ml)、 水(10 ml)および飽和食塩水(5ml)を加え、 酢酸ェチル (40 ml) で抽出した。 有機層を 5%、 15%および飽和の食塩水で洗浄し、 硫酸ナトリウムで 乾燥後、 濃縮した。 残渣の濃赤色油状物をシリカゲル (10 g)のクロマトグラフィ ― (酢酸ェチルーへキサン、 2:8-4:6) に付し、 酢酸ェチルーへキサン(4: 6) の画 分を濃縮乾固後、メタノールで希釈した。析出物を濾取し、メタノールで洗浄後、 乾燥して表題化合物(457 mg, 0.831 皿 ol, 56.2%)を赤褐色粉末として得た。 In Reference Example 24, a suspension of the compound obtained in Example 3-4 (566 mg, 1.48 tmol) and potassium carbonate (0.41 g, 2.97 benzene 01) in DMF (9 ml) was stirred at 80 ° C. A solution of the obtained cis_5-acetoxycyclooctyl iodide (0.81 g, 1.6 mol) in DMF (5 ml) was added dropwise over 90 minutes, and the reaction solution was further stirred for 1.5 hours. To the reaction solution were added 1N hydrochloric acid (3.5 ml), 0.1 IN hydrochloric acid (15 ml), water (10 ml) and saturated saline (5 ml), and the mixture was extracted with ethyl acetate (40 ml). The organic layer was washed with 5%, 15% and saturated saline, dried over sodium sulfate, and concentrated. The residue was subjected to chromatography on silica gel (10 g)-(ethyl acetate-hexane, 2: 8-4: 6), and the ethyl acetate-hexane (4: 6) fraction was concentrated to dryness. And diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (457 mg, 0.831 ol, 56.2%) as a red-brown powder.
Ή-NMR (300MHz, CDC13) : ά 1.65-1.90 (10Η, m), 1.80 (3H, s), 2.03 (3H, s), 2.03 (2H, m), 2.26 (3H, q, J= 0.8 Hz), 2.42 (3H, s), 3.53 (1H, m), 4.88 (1H, m), 7.28 (2H, dd, J= 8.0, 1.8 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.63 (1H, brs). 元素分析値: C25H31N305S3として: C, 54.62; H, 5.68; N, 7.64; S, 17.50 (%) 、 計算値: C, 54.55; H, 5.86; N, 7.60; S, 17.59 (%) . Ή-NMR (300MHz, CDC1 3 ): ά 1.65-1.90 (10Η, m), 1.80 (3H, s), 2.03 (3H, s), 2.03 (2H, m), 2.26 (3H, q, J = 0.8 Hz), 2.42 (3H, s), 3.53 (1H, m), 4.88 (1H, m), 7.28 (2H, dd, J = 8.0, 1.8 Hz), 7.85 (2H, d, J = 8.0 Hz), . 9.63 (1H, brs) elemental analysis: as C 25 H 31 N 3 0 5 S 3: C, 54.62; H, 5.68; N, 7.64; S, 17.50 (%), calculated: C, 54.55; H , 5.86; N, 7.60; S, 17.59 (%).
mp. 158.0-159.5t mp. 158.0-159.5t
実施例 4-541 Example 4-541
N-(5, 6-ジメチル -3-ォキソ -8- { [2- (プロボキシ)ェチル]スルファニル卜 2, 3-ジヒ ドロ- 1H -ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン) -4 -メチルベンゼンスルホ ンアミド N- (5,6-dimethyl-3-oxo-8-{[2- (propoxy) ethyl] sulfanyl chloride 2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene) -4-methylbenzenesulfonamide
実施例 4-533と同様にして合成した。 Synthesized in the same manner as in Example 4-533.
赤外吸収スぺクトル(IR) (KBr中) V: 3228, 2961, 2928, 2870, 1721, 1661, 1611, 1557 cnr1. Infrared absorption spectrum (IR) (in KBr) V: 3228, 2961, 2928, 2870, 1721, 1661, 1611, 1557 cnr 1 .
Ή-NMR (CDC13) : δ 0.923.(3Η, d, J=7.5 Hz), 1.30-1.44 (2H, m), 1.50-1.62 (2H, m), 1.79 (3H, d, J=0.9 Hz), 2.26 (3H, d, J=1.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J=6.6 Hz), 3.45 (2H, t, J=6.6 Hz), 3.63. (2H, t, J=6.9 Hz), 7.28 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=7.8 Hz), 9.61 (1H, bs). 実施例 4-542 Ή-NMR (CDC1 3): . Δ 0.923 (3Η, d, J = 7.5 Hz), 1.30-1.44 (2H, m), 1.50-1.62 (2H, m), 1.79 (3H, d, J = 0.9 Hz ), 2.26 (3H, d, J = 1.2 Hz), 2.42 (3H, s), 3.15 (2H, t, J = 6.6 Hz), 3.45 (2H, t, J = 6.6 Hz), 3.63. (2H, t, J = 6.9 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 7.8 Hz), 9.61 (1H, bs). Example 4-542
ェチル 5- [ (6-へキシル -5-メチル -1- {[(4 -メチルフエニル)スルホニル]イミ ノ } -3 -ォキソ -3H-ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル]ペン夕 ノエート Ethyl 5-[(6-hexyl-5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5,1-c] [1,4] thiazine- 8-yl) sulfanyl] pen evening noate
実施例 3-17 で得られた N-(6-へキシル -5-メチル -3-ォキソ -8-スルファニル - 2, 3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1, 4]チアジン-卜イリデン )-4_メチルベンゼ ンスルホンアミド (600 mg, 1.33腿 ol) と炭酸カリウム (275 mg, 1.99 mmol) の N,N-ジメチルホルムアミド (12.0ml) の懸濁液に、 5_ブロモ吉草酸ェチルェ ステル (0.257 ml, 1.59匪 ol) を加えた。 反応混合物を 80 で 1.5時間加熱し た後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥(MgS04) 後、 溶媒を減圧下に留去して題記化合物 (639 mg, 83%) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルエーテルから再結晶して融点 110-11 °Cの暗赤色結晶を得た。 IR (KBr) V: 3212, 2957, 2928, 2857, 1732, 1657, 1615, 1563 cm"1. N- (6-Hexyl-5-methyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, tric] [1, 4,] thiazine obtained in Example 3-17 -Tolylidene) -4_methylbenzenebenzenesulfonamide (600 mg, 1.33 liters) and potassium carbonate (275 mg, 1.99 mmol) in N, N-dimethylformamide (12.0 ml) were suspended in Ethylester sterate (0.257 ml, 1.59 ol) was added. After heating the reaction mixture at 80 for 1.5 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (639 mg, 83%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 110-11 ° C. IR (KBr) V: 3212, 2957, 2928, 2857, 1732, 1657, 1615, 1563 cm " 1 .
Ή-NMR (CDC13) : δ 0.892 (3Η, t, J=7.2Hz), 1.27 (3H, t, J=7. Hz), 1.20-1.38 (6H, m), 1.38-1.54 (2H, m), 1.65-1.82 (4H, m), 2.11 (2H, t, J=8.2 Hz), 2.26 (3H, s), 2.33 (2H, t, J=6.8 Hz), 2.41 (3H, s), 2.95 (2H, t, J=6.8 Hz), 4.14 (2H, q, J=7.2 Hz), 7.29 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.0 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 0.892 (3Η, t, J = 7.2Hz), 1.27 (. 3H, t, J = 7 Hz), 1.20-1.38 (6H, m), 1.38-1.54 (2H, m ), 1.65-1.82 (4H, m), 2.11 (2H, t, J = 8.2 Hz), 2.26 (3H, s), 2.33 (2H, t, J = 6.8 Hz), 2.41 (3H, s), 2.95 (2H, t, J = 6.8 Hz), 4.14 (2H, q, J = 7.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.0 Hz), 9.60 (1H , Bs).
元素分析値: C27H37N305S3として Elementary analysis: as C 27 H 37 N 3 0 5 S 3
計算値 (%) : C, 55.93; H, 6.43; N, 7.25; S, 16.59. Calculated value (%): C, 55.93; H, 6.43; N, 7.25; S, 16.59.
実測値 (%) : C, 55.82; H, 6.70; N, 7.28; S, 16.71. Found (%): C, 55.82; H, 6.70; N, 7.28; S, 16.71.
実施例 4-543 Example 4-543
ェチル 5-[(6-ベンジル- 5-メチル -1- {[(4-メチルフエニル)スルホニル]イミ ノ } -3 -ォキソ -3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィル)スルファニル]ペン夕 ノエート Ethyl 5-[(6-benzyl-5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5,1-c] [1,4] thiazine-8 -Ill) Sulfanyl] Pen Evening Noate
実施例 3-19 で得られた N- (6-ベンジル- 5-メチル -3-ォキソ -8-スルファニル - 2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン)- 4_メチルベンゼ ンスルホンアミド (600 mg, 1.31 讓 ol) と炭酸カリウム (272 mg, 1.97腿 ol) の N,N-ジメチルホルムアミド (12.0ml) の懸濁液に、 5-プロモ吉草酸 ェチルェ ステル (0.254 ml, 1.57 mmol) を加えた。 反応混合物を 80°Cで 1.5時間加熱し た後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (567 mg, 74%) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルエーテルから再結晶して融点 108- 110°Cの暗赤色結晶を得た。 IR (KBr) V: 3270, 2930, 1732, 1657, 1617, 1559 cm"1. N- (6-benzyl-5-methyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine obtained in Example 3-19 A suspension of 1-ylidene) -4_methylbenzenebenzenesulfonamide (600 mg, 1.31 acetylol) and potassium carbonate (272 mg, 1.97 thiol) in N, N-dimethylformamide (12.0 ml) was added to the suspension. Promoter valeric acid Stell (0.254 ml, 1.57 mmol) was added. After heating the reaction mixture at 80 ° C for 1.5 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (567 mg, 74%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 108-110 ° C. IR (KBr) V: 3270, 2930, 1732, 1657, 1617, 1559 cm " 1 .
Ή-NMR (CDC13) : δ 1.25 (3Η, t, J=7.4Hz), 1.59-1.72 (4H, m), 2.22-2.30 (2H, m), 2.37 (3H, s), 2.42 (3H, s), 2.79-2.90 (2H, m), 3.50 (2H, s), 4.12 (2H, q, J=7.4 Hz), 7.18-7.38 (7H, m), 7.84 (2H, d, J=8.0 Hz), 9.65 (1H, bs). 元素分析値: C28H31N305S3として Ή-NMR (CDC1 3): δ 1.25 (3Η, t, J = 7.4Hz), 1.59-1.72 (4H, m), 2.22-2.30 (2H, m), 2.37 (3H, s), 2.42 (3H, s), 2.79-2.90 (2H, m), 3.50 (2H, s), 4.12 (2H, q, J = 7.4 Hz), 7.18-7.38 (7H, m), 7.84 (2H, d, J = 8.0 Hz ), 9.65 (1H, bs). Elemental analysis: C 28 H 31 N 3 0 5 S 3
計算値 (%) : C, 57.41; H, 5.33; N, 7.17; S, 16.42. Calculated (%): C, 57.41; H, 5.33; N, 7.17; S, 16.42.
実測値 (¾) : C, 57.20; H, 5.18; N, 7.31; S, 16.45. Found (¾): C, 57.20; H, 5.18; N, 7.31; S, 16.45.
実施例 4- 545 Example 4-545
(2S)-4-[(5, 6_ジメチル- 1 - {[(4 -メチルフエニル)スルホニル]ィミノ } - 3-ォキソ -2, 3-ジヒド口- 1H -ィミダゾ [5, 1 - c] [1 , 4]チアジン- 8-ィル)スルファエル] - 1 , 2 -ピ 口リジンジカルボン酸 1- tert-ブチル 2-メチル (2S) -4-[(5,6_Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [ 1, 4] thiazin-8-yl) sulfael]-1-tert-butyl 2-methyl 1,2-piperidinedicarboxylate
実施例 3- 4で得た化合物(1.00 g, 2.62匪 ol)および炭酸カリウム (543 mg, 3.93 画 ol) の DMF (17 ml) の懸濁液に 60°Cで攪拌しながら参考例 35 で得た (2S)-N-Boc- 4-ョ一ド- L-プロリンメチルエステル (純度 80%, 1.29 g, 2.90mmol) を加え、 反応液を 4時間攪拌した。 反応液に 1N塩酸 (5.3 ml), 水(15 ml)およ び 5%食塩水(100 ml)を加え、 酢酸ェチル(70 ml)で抽出した。 有機層を 5%および 飽和の食塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して濃赤色油状物を 得た。 これをシリカゲル(20 g)のクロマトグラフィー (酢酸ェチルーへキサン、 2:8-5:5) に付し、酢酸ェチルーへキサン(4:6-5:5) の画分を濃縮乾固後、 ジェチ ルエーテルで希釈した。 析出物を濾取し、 ジェチルエーテルで洗浄後、 乾燥して 表題化合物(947 mg, 1.56 mmol, 59.3%)を赤色粉末として得た。 In Reference Example 35, a suspension of the compound obtained in Example 3-4 (1.00 g, 2.62 ol) and potassium carbonate (543 mg, 3.93 ol) in DMF (17 ml) was stirred at 60 ° C. The obtained (2S) -N-Boc-4-ode-L-proline methyl ester (purity 80%, 1.29 g, 2.90 mmol) was added, and the reaction solution was stirred for 4 hours. 1N Hydrochloric acid (5.3 ml), water (15 ml) and 5% saline (100 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (70 ml). The organic layer was washed successively with 5% and saturated saline, dried over sodium sulfate, and concentrated to give a dark red oil. This was subjected to silica gel (20 g) chromatography (ethyl acetate-hexane, 2: 8-5: 5), and the ethyl acetate-hexane (4: 6-5: 5) fraction was concentrated to dryness. Diluted with ethyl ether. The precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (947 mg, 1.56 mmol, 59.3%) as a red powder.
Ή-NMR (300MHz, CDC13) : δ 1.42 (6Η, s), 1.48 (3H, s), 1.78/1.80 (3H, s), 2.09 (1H, dt, J= 13.5, 7.2 Hz), 2.26 (3H, s), 2.37 (1H, brdd, J= 15.6, 7.5 Hz), 2.43 (3H, s), 2.74 (1H, dt, J= 13.2, 7.8 Hz), 3.41 (1H, dd, J= 11.1, 7.5 Hz), 3.75/3.76 (3H, s), 3.85/3.95 (1H, q, J= 7.9/9.6 Hz), 4.09/4.01 (1H, dd, J= 12.6, 8.7 Hz), 4.30/4.38 (1H, t/m, J= 7.7 Hz), 7.31 (2H, d, J= 8.1 Hz), 7.83 (2H, d, J= 8.1 Hz), 9.64 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δ 1.42 (6Η, s), 1.48 (3H, s), 1.78 / 1.80 (3H, s), 2.09 (1H, dt, J = 13.5, 7.2 Hz), 2.26 ( 3H, s), 2.37 (1H, brdd, J = 15.6, 7.5 Hz), 2.43 (3H, s), 2.74 (1H, dt, J = 13.2, 7.8 Hz), 3.41 (1H, dd, J = 11.1, 7.5 Hz), 3.75 / 3.76 (3H, s), 3.85 / 3.95 (1H, q, J = 7.9 / 9.6 Hz), 4.09 / 4.01 (1H, dd, J = 12.6, 8.7 Hz), 4.30 / 4.38 (1H, t / m, J = 7.7 Hz), 7.31 (2H, d, J = 8.1 Hz), 7.83 (2H, d, J = 8.1 Hz), 9.64 (1H, brs).
元素分析値: C26H32N407S3として計算値: C, 51.30; H, 5.30; N, 9.20; S, 15.80 (%) 、 実測値: C, 51.21; H, 5.19; , 9.06; S, 15.51 ( ) . Elemental analysis: Calculated as C 26 H 32 N 4 0 7 S 3: C, 51.30; H, 5.30; N, 9.20; S, 15.80 (%), Found: C, 51.21; H, 5.19 ;, 9.06 S, 15.51 ().
mp. 140.0-140.5°C mp.140.0-140.5 ° C
実施例 4-546 Example 4-546
(2S,4S) - 2- {[(5,6-ジメチル- 1-{ [(4 -メチルフエニル)スルホニル]イミノ} - 3-ォキ ソ- 2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル]メチ ル}- 4_フルォ口- 1-ピロリジンカルポン酸 tert-ブチル (2S, 4S)-2-{[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1 -c] [1,4] thiazin-8-yl) sulfanyl] methyl} -4_fluoro mouth tert-butyl 1-pyrrolidinecarbonate
実施例 3-4 で得た化合物 (0.89 g, 2.33 mmol)、 参考例 25 で得た Compound obtained in Example 3-4 (0.89 g, 2.33 mmol), obtained in Reference Example 25
(2S,4S)- N-Boc- 4-フルォロ- 2-ョードメチルピロリジンの粗物質(1.00 g)および 炭酸カリウム (0.48 g, 3.5 mmol) の DMF (15 ml) の懸濁液を 60°Cで 6 日間攪 拌した。反応液に 1N塩酸 (4.7 ml),水(60 ml)および飽和食塩水(20 ml)を加え、 酢酸ェチル(100 ml および 70 ml)で抽出した。 有機層を 2%炭酸ナトリウム、 2% 炭酸水素ナトリウム水溶液、 5¾および飽和の食塩水で順次洗浄し、 硫酸ナトリウ ムで乾燥後、 濃縮して濃赤色油状物を得た。 これをシリカゲル (20 g)のクロマト グラフィ一 (酢酸ェチルーへキサン、 2:8-4:6) で精製し、 得られた赤色油状物を 酢酸ェチルージェチルェ一テルージイソプロピルエーテルから結晶化し、 表題化 合物(177 mg, 0.303匪 ol, 9.5%)を赤色粉末として得た。 A suspension of the crude material (1.00 g) of (2S, 4S) -N-Boc-4-fluoro-2-iodomethylpyrrolidine and potassium carbonate (0.48 g, 3.5 mmol) in DMF (15 ml) was prepared. Stirred at ° C for 6 days. 1N Hydrochloric acid (4.7 ml), water (60 ml) and saturated saline (20 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (100 ml and 70 ml). The organic layer was washed successively with 2% sodium carbonate, 2% aqueous sodium hydrogen carbonate solution, 5¾ and saturated brine, dried over sodium sulfate, and concentrated to obtain a dark red oil. This was purified by chromatography on silica gel (20 g) (ethyl acetate-hexane, 2: 8-4: 6), and the resulting red oil was crystallized from ethyl acetate-ethyl acetate-terdiisopropyl ether. The title compound (177 mg, 0.303 bandol, 9.5%) was obtained as a red powder.
'H-NMR (200MHz, CDC13) : δΐ.48 (9Η, s), 1.79 (3H, s), 2.20-2.50 (2H, m), 2.25 (3H, s), 2.42 (3H, s), 2.92 (1H, dd, J= 13.0, 10.2 Hz), 3.50 (1H, in), 3.62 (1H, m), 3,68 (1H, brd, J= 27.8 Hz), 4.10 (1H, m), 5.20 (1H, brd, J= 52.0 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). 元素分析値: C25H31N405S3Fとして計算値: C, 51.53; H, 5.36; N, 9.61; S, 16.51; F, 3.26 (%) 、 実測値: C, 51.52; H, 5.40; N, 9.75; S, 16.26; F, 3.12 (%) . 即. 132.5-135.5°C 'H-NMR (200MHz, CDC1 3): δΐ.48 (9Η, s), 1.79 (3H, s), 2.20-2.50 (2H, m), 2.25 (3H, s), 2.42 (3H, s), 2.92 (1H, dd, J = 13.0, 10.2 Hz), 3.50 (1H, in), 3.62 (1H, m), 3,68 (1H, brd, J = 27.8 Hz), 4.10 (1H, m), 5.20 (1H, brd, J = 52.0 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs). Elemental analysis: C 25 H 31 N 4 0 5 S 3 F calculated: C, 51.53; H, 5.36 ; N, 9.61; S, 16.51; F, 3.26 (%), Found: C, 51.52; H, 5.40 ; N, 9.75; S , 16.26; F, 3.12 (%). Immediately. 132.5-135.5 ° C
実施例 4-547 Example 4-547
N - {8- [(5, 5-ジフルォロシクロォクチル)スルファニル ]-5, 6-ジメチル -3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン卜 4 -メチルベンゼ ンスルホンアミド N- {8-[(5,5-difluorocyclooctyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4 ] Thiazine-1-yl 4-methylbenze Sulfonic acid
参考例 26で得た 5, 5-ジフルォロシクロォクチルトシレート (純度 85%, 0.48g, 1.28 腿 ol)およびヨウ化ナトリウム(576 mg, 3.84画 ol) の アセトン (10 ml) 懸濁液を 3時間加熱還流した。 反応液をジイソプロピルエーテル—へキサンによ り抽出して 5,5-ジフルォ口- 1-ョ一ドシクロオクタンの粗物質を褐色油状物とし て得た。 得られた油状物、 実施例 3-4で得た化合物 (488 mg, 1.28 mmol)および炭 酸カリウム (265 mg, 1.92 匪 ol) の DMF (8 ml) の懸濁液を室温で 30分間、 次 いで 6(TCで 6時間攪拌した。 反応液に 10¾食塩水を加え、 酢酸ェチル(50 miおよ び 20 ml)で抽出した。 有機層を 2%炭酸ナトリウムで 2回、 0.1N塩酸、 ^および 飽和の食塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して濃赤色油状物を 得た。 これをメタノールから結晶化し、 表題化合物(236 mg, 0.447 mmol, 34.9%) を濃紫色粉末として得た。 Suspension of 5,5-difluorocyclooctyl tosylate (purity 85%, 0.48 g, 1.28 tmol) and sodium iodide (576 mg, 3.84 l) obtained in Reference Example 26 in acetone (10 ml) The liquid was heated to reflux for 3 hours. The reaction solution was extracted with diisopropyl ether-hexane to obtain a crude substance of 5,5-difluoro-1-hydroxycyclooctane as a brown oil. A suspension of the obtained oil, the compound obtained in Example 3-4 (488 mg, 1.28 mmol) and potassium carbonate (265 mg, 1.92 ol) in DMF (8 ml) was stirred at room temperature for 30 minutes. Then, the mixture was stirred for 6 hours with TC. 10TC saline was added to the reaction solution, and extracted with ethyl acetate (50 mi and 20 ml) .The organic layer was washed twice with 2% sodium carbonate, 0.1N hydrochloric acid, Washed sequentially with ^ and saturated brine, dried over sodium sulfate and concentrated to give a dark red oil which was crystallized from methanol to give the title compound (236 mg, 0.447 mmol, 34.9%) as a dark purple color Obtained as a powder.
Ή—匪 R (200MHz, CDCI3): (51.75 (6H, m), 1.80 (3H, q, J= 0.8 Hz), 2.00-2.20 (6H, m), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 3.54 (1H, ), 7.29 (2H, d, J= 8.6 Hz), 7.84 (2H, d, J= 8.6 Hz), 9.64 (1H, brs). Ή—band R (200MHz, CDCI 3 ): (51.75 (6H, m), 1.80 (3H, q, J = 0.8 Hz), 2.00-2.20 (6H, m), 2.26 (3H, q, J = 1.2 Hz) ), 2.42 (3H, s), 3.54 (1H,), 7.29 (2H, d, J = 8.6 Hz), 7.84 (2H, d, J = 8.6 Hz), 9.64 (1H, brs).
元素分析値 C23H27N303S3F2として計算値: C, 52.35; H, 5.16; N, 7.96; S, 18.23; F, 7.20 (%) 、 実測値: C, 52.43; H, 5.17; N, 8.01; S, 18.28; F, 7.03 ( ) . 即. 169.0-170.5°C Elemental analysis: calculated as C 23 H 27 N 3 0 3 S 3 F 2 : C, 52.35; H, 5.16; N, 7.96; S, 18.23; F, 7.20 (%), found: C, 52.43; H , 5.17; N, 8.01; S, 18.28; F, 7.03 (). Immediately. 169.0-170.5 ° C
実施例 4- 548 . Example 4-548.
N- {5, 6 -ジメチル -3-ォキソ -8- [(5-ォキソシク口才クチル)スルファニル ]-2, 3-ジ ヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン } -4-メチルベンゼンスルホ ンアミド N- {5,6-Dimethyl-3-oxo-8-[(5-oxo-cyclohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene} -4-methylbenzenesulfonamide
参考例 26で得た 5-ォキソシクロォクチルトシレート (純度 70%, 0.48 g, 1.13 mmol)およびヨウ化ナトリウム(0.51 g, 3.40 mmol) の アセトン (10 ml)懸濁液 を 5.5時間加熱還流した。 反応液をジイソプロピルエーテルにより抽出して粗製 の 5-ォキソ -1-ョ一ドシクロオクタンの粗物質を褐色油状物として得た。 得られ た油状物、実施例 3-4で得た化合物(0.43 g, 1.13匪 ol)および炭酸力リゥム (234 mg, 1.70 mmol) の DMF (7 ml) の懸濁液を室温で 10分間、 次いで 50 で 12時 間、 さらに 60°Cで 24時間攪拌した。 反応液に 1N塩酸 (2.3 ml)およびメタノー ルー水 (1:1, 20 ml)を加えて室温で攪拌し、析出した沈殿物を上澄液から分離し、 メタノール一水 (1:1)で洗浄した。 得られた油状物を THF -ァセトニトリル -酢酸 ェチルに溶解して濃縮し、 残渣にメタノ一ルを加えた。 析出物を濾取し、 メタノ ールで洗浄後、 乾燥して表題化合物(230 mg, 0.455蘭 ol, 40.3%)を赤紫色粉末と して得た。 , A suspension of 5-oxocyclooctyl tosylate (purity 70%, 0.48 g, 1.13 mmol) and sodium iodide (0.51 g, 3.40 mmol) in acetone (10 ml) obtained in Reference Example 26 was heated for 5.5 hours. Refluxed. The reaction solution was extracted with diisopropyl ether to obtain a crude 5-oxo-1-hydroxycyclooctane as a brown oil. A suspension of the obtained oil, the compound obtained in Example 3-4 (0.43 g, 1.13 bandol) and carbonated rim (234 mg, 1.70 mmol) in DMF (7 ml) was added at room temperature for 10 minutes. Then, the mixture was stirred at 50 for 12 hours and further at 60 ° C for 24 hours. Add 1N hydrochloric acid (2.3 ml) and methanol Lou water (1: 1, 20 ml) was added and the mixture was stirred at room temperature, and the deposited precipitate was separated from the supernatant and washed with methanol-water (1: 1). The obtained oil was dissolved in THF-acetonitrile-ethyl acetate and concentrated, and methanol was added to the residue. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (230 mg, 0.455 orchid ol, 40.3%) as a red-purple powder. ,
Ή-NMR (200MHz, CDC13) : δ 1.80 (3Η, q, J= 1.0 Hz), 1.85-2.10 (8H, m), 2.25 (3H, q, J= 1.0 Hz), 2.32 (2H, dd, J= 13.2, 8.8 Hz), 2.42 (3H, s), 2.61 (2H, ddd, J= 13.2, 8.4, 3.0 Hz), 3.13 (1H, tin, J= 8.6 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.83 (2H, d, J= 8.0 Hz), 9.61 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.80 (3Η, q, J = 1.0 Hz), 1.85-2.10 (8H, m), 2.25 (3H, q, J = 1.0 Hz), 2.32 (2H, dd, J = 13.2, 8.8 Hz), 2.42 (3H, s), 2.61 (2H, ddd, J = 13.2, 8.4, 3.0 Hz), 3.13 (1H, tin, J = 8.6 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.61 (1H, brs).
元素分析値: C23H27N304S3. H20として計算値: G, 52.75; H, 5.58; N, 8.02; S, 18.37 (%) 、 実測値: C, 52.73; H, 5.61; N, 8.17; S, 18.76 (%) · . Elemental analysis: C 23 H 27 N 3 0 4 S 3 H 2 0 Calculated: G, 52.75; H, 5.58 ; N, 8.02; S, 18.37 (%), Found: C, 52.73; H, 5.61; N, 8.17; S, 18.76 (%)
実施例 4- 549 Example 4-549
酢酸 ((5R,7S)- 3-{[(5,6-ジメチル- 1-{[(4-メチルフエニル)スルホニル]イミ ノ}-3-ォキソ-2,3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン- 8 -ィル)スルファ ニル]メチル }トリシク口 [3.3.1.1—3, 7Ίデシ _1 -ィル)メチル Acetic acid ((5R, 7S) -3-{[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazin-8-yl) sulfanyl] methyl} tris [[3.1.1.1.1-3,7Ίdec_1-yl) methyl
実施例 3-4で得た化合物(3.02 g, 7.92丽 ol)、 参考例 27で得た 3-ョ一ドメ チルァダマンタン- 1-メチルアセテート(762 nig, 2.19 mmol)および炭酸カリウム (434 mg, 3.14腿 ol) の DMF (13 ml) の懸濁液を室温で 30分間、 次いで 70°Cで 24時間攪拌した。反応液に 1N塩酸 (4.1ml)およびメタノール一水 (1:1, 12ml) を加えて室温で 30分間攪拌した。析出物を濾取し、メタノール—水 (1:1)および メタノールで洗浄後、 乾燥して表題化合物(647 mg, 1.08匪 ol, 51.4«を赤紫色 粉末として得た。 The compound obtained in Example 3-4 (3.02 g, 7.92 mol), 3-iodomethyl adamantan-1-methyl acetate obtained in Reference Example 27 (762 nig, 2.19 mmol) and potassium carbonate (434 mg, 3.14 mmol) A suspension of TMF (13 ml) in DMF (13 ml) was stirred at room temperature for 30 minutes and then at 70 ° C for 24 hours. 1N Hydrochloric acid (4.1 ml) and methanol / water (1: 1, 12 ml) were added to the reaction solution, and the mixture was stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (647 mg, 1.08 bandol, 51.4 «) as a red-purple powder.
¾-NMR (300MHz, CDC13) : (51.35 (2H, brs), 1.46 (2H, brs), 1.50 (2H, brs), 1.56 (4H, brs), 1.58 (2H, m), 1.78 (3H, s), 2.06 (3H, s), 2.10 (2H, m), 2.26 (3H, q, J- 0.9 Hz), 2.41 (3H, s), 2.80 (2H, s), 3.70 (2H, s), 7.26 (2H, d, J= 8.1 Hz), 7.86 (2H, d, J= 8.1 Hz), 9.56 (1H, brs). ¾-NMR (300MHz, CDC1 3 ): (51.35 (2H, brs), 1.46 (2H, brs), 1.50 (2H, brs), 1.56 (4H, brs), 1.58 (2H, m), 1.78 (3H, s), 2.06 (3H, s), 2.10 (2H, m), 2.26 (3H, q, J- 0.9 Hz), 2.41 (3H, s), 2.80 (2H, s), 3.70 (2H, s), 7.26 (2H, d, J = 8.1 Hz), 7.86 (2H, d, J = 8.1 Hz), 9.56 (1H, brs).
元素分析値: C29H35N305S3.0.5 0として計算値: C, 57.02; H, 5.94; , 6.88; S, 15.75' (%) 、 実測値: C, 56.84; H, 5.95; N, 6.88; S, 15.66 ( ) . Elemental analysis: C 29 H 35 N 3 0 5 S 3 .0.5 0 Calculated: C, 57.02; H, 5.94 ;, 6.88; S, 15.75 '(%), Found: C, 56.84; H, 5.95 ; N, 6.88; S, 15.66 ().
即. 171.0- 172.0°C 実施例 4-550 Immediate. 171.0- 172.0 ° C Example 4-550
酢酸 (3 - {[(5, 6-ジメチル-ト {[(4-メチルフエニル)スルホニル]イミノ卜 3-ォキソ -2,3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 8-ィル)スルファニル]メチル } 実施例 3- 4で得た化合物(1.45 g, 3.80匪 ol)および炭酸カリウム (788 mg, 5.70 mmol) の DMF (20 ml) 懸濁液に 70°Cで参考例 28で得た 3-ョ一ドメチルシク口へ キサン-卜メチルアセテート(1.24 g, 4.18 mmol) DMF (5 ml)溶液を 20分間かけ て滴下し、 反応液を 70 で 3時間攪拌した。反応液に 1N塩酸 (7.6 ml)およびメ 夕ノール一水 (1:1, 20 ml)を加えた。 析出物を濾取し、 メタノール一水 (1:1) およびメタノールで洗浄後、 乾燥して表題化合物(1.71 g, 3.11 mmol, 81.9%)を 赤紫色粉末として得た。 Acetic acid (3-{[(5,6-dimethyl-to {[(4-methylphenyl) sulfonyl] iminoto 3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine -8-yl) sulfanyl] methyl} A suspension of the compound obtained in Example 3-4 (1.45 g, 3.80 ol) and potassium carbonate (788 mg, 5.70 mmol) in DMF (20 ml) was added at 70 ° C. Then, a solution of xanthromethyl acetate (1.24 g, 4.18 mmol) in DMF (5 ml) was added dropwise to the mouth of 3-hydroxymethylcycline obtained in Reference Example 28 over 20 minutes, and the reaction solution was stirred at 70 for 3 hours. 1N Hydrochloric acid (7.6 ml) and methanol / water (1: 1, 20 ml) were added to the reaction mixture, and the precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried. The title compound (1.71 g, 3.11 mmol, 81.9%) was obtained as a red-purple powder.
'H-NMR (300MHz, CDC13) : (50.90/0.71 (2H, brq/q, J= 12.6/12.3 Hz), 1.29/1.49 (2H, brq/m, J= 12.6Hz), 1.63 (2H, m), 1.75/1.86 (2H, m), 1.79 (3H, s), 1.92/1.88 (2H, m), 2.06/2.05 (3H, s), 2.26 (3H, s), 2.41 (3H, s), 2.83/2.89 (2H, d, J= 6.9 Hz), 3.89/3.96 (2H, d, J= 6.3/6.9 Hz), 7.26 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.60 (1H, brs). 'H-NMR (300MHz, CDC1 3): (50.90 / 0.71 (2H, brq / q, J = 12.6 / 12.3 Hz), 1.29 / 1.49 (2H, brq / m, J = 12.6Hz), 1.63 (2H, m), 1.75 / 1.86 (2H, m), 1.79 (3H, s), 1.92 / 1.88 (2H, m), 2.06 / 2.05 (3H, s), 2.26 (3H, s), 2.41 (3H, s) , 2.83 / 2.89 (2H, d, J = 6.9 Hz), 3.89 / 3.96 (2H, d, J = 6.3 / 6.9 Hz), 7.26 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.60 (1H, brs).
元素分析値: C25H31N305S3として計算値: C, 54.62; H, 5.68; N, 7.64; S, 17.50Elemental analysis: C 25 H 31 N 3 0 5 S 3 Calculated: C, 54.62; H, 5.68 ; N, 7.64; S, 17.50
(%) 、 実測値: C, 54.51; H, 5.54; N, 7.60; S, 17.43 (%) . (%), Found: C, 54.51; H, 5.54; N, 7.60; S, 17.43 (%).
即. 163.5-166.0°C Immediately. 163.5-166.0 ° C
実施例 4-551 Example 4-551
N- [8- ({[3- (フルォロメチル)シクロへキシル]メチル }スルファニル )-5, 6-ジメチ ル -3-ォキソ - 2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン ]_4 -メ チルベンゼンスルホンアミド N- [8-({[3- (Fluoromethyl) cyclohexyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1, 4] thiazine-1-ylidene] _4-methylbenzenesulfonamide
参考例 29 で得た 3 -フルォロメチルシク口へキサン- 1-メチルトシレート(170 mg, 0.566 mmol)およびヨウ化ナトリウム(254 mg, 1.70 mmol) の アセトン (4ml) 懸濁液を 17時間加熱還流した。 反応液をジイソプロピルエーテルにより抽出、 濃縮乾固、 乾燥してヨウ化 3-フルォロメチルシクロへキサン- 1 -メチル (約 140 mg)を無色油状物として得た。 この油状物、 実施例 3-4 で得た化合物 (200 mg, 0.524醒 ol)および炭酸カリウム (109 mg, 0.786匪 ol) の DMF (4 ml) の懸濁液 を室温で 10分間、 次ぃで40でで3.5日間攪拌した。反応液に 1N塩酸 (1.05ml) を加え、 次いでメタノール一水 (1:1, 5 ml) を加えて希釈し、 室温で 60分間攪 拌した。 析出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄後、 乾 燥して表題化合物(230 ing, 0.451 匪 ol, 86.1%)を赤色粉末として得た。 A suspension of acetone (4 ml) of 3-fluoromethylcyclohexane-1-methyltosylate (170 mg, 0.566 mmol) and sodium iodide (254 mg, 1.70 mmol) obtained in Reference Example 29 was added to 17 Heated to reflux for an hour. The reaction solution was extracted with diisopropyl ether, concentrated to dryness, and dried to obtain 3-fluoromethylcyclohexane-1-methyl iodide (about 140 mg) as a colorless oil. A suspension of this oil, the compound obtained in Example 3-4 (200 mg, 0.524 ol) and potassium carbonate (109 mg, 0.786 ol) in DMF (4 ml) Was stirred at room temperature for 10 minutes and then at 40 at 3.5 days. The reaction solution was diluted with 1N hydrochloric acid (1.05 ml), and then added with methanol / water (1: 1, 5 ml) and stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (230 ing, 0.451 ol, 86.1%) as a red powder.
Ή-NMR (300MHz, CDC13) : 50.96/0.75 (2H, qm/q, J= 12.8/12.6 Hz), 1.34 (2H, m), 1.62 (2H, m), 1.75-2.00 (4H, m), 1.79 (3H, s), 2.26 (3H, s), 2.41 (3H, s), 2.84/2.91 (2H, d, J= 6.6/7.0 Hz), 4.24/4.28 (2H, ddd/dd, 1= 47.6, 5.8, 2.6/47.2, 7.4 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.0 Hz), 9.61 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): 50.96 / 0.75 (2H, qm / q, J = 12.8 / 12.6 Hz), 1.34 (2H, m), 1.62 (2H, m), 1.75-2.00 (4H, m) , 1.79 (3H, s), 2.26 (3H, s), 2.41 (3H, s), 2.84 / 2.91 (2H, d, J = 6.6 / 7.0 Hz), 4.24 / 4.28 (2H, ddd / dd, 1 = 47.6, 5.8, 2.6 / 47.2, 7.4 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.0 Hz), 9.61 (1H, brs).
元素分析値: C23H28N303S3Fとして計算値: C, 54.20; H, 5.54; N, 8.24; S, 18.87; F, 3.73 {%) 、 実測値: C, 54.16; H, 5.66; N, 8.39; S, 18.79; F, 3.95 (%) mp. 172.5-173.5°C Elemental analysis: C 23 H 28 N 3 0 3 S 3 F Calculated: C, 54.20; H, 5.54 ; N, 8.24; S, 18.87; F, 3.73 {%), Found: C, 54.16; H , 5.66; N, 8.39; S, 18.79; F, 3.95 (%) mp. 172.5-173.5 ° C
実施例 4- 552 Example 4-552
酢酸 5- [(5,6-ジメチル- 1_{[(4-メチルフエニル)スルホ二ル]イミノ} - 3-ォキソ -2, 3-ジヒド口- 1H-イミダゾ [5, 1-c] [1, ]チアジン- 8-ィル)スルファニル] - 3, 3-ジ メチルペンチル Acetic acid 5-[(5,6-dimethyl-1 _ {[(4-methylphenyl) sulfonyl] imino}-3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1, [Thiazine-8-yl) sulfanyl]-3,3-Dimethylpentyl
実施例 3-4で得た化合物(1.81 g, 4.75腿 ol)、 参考例 30で得た酢酸 5-ョード - 3, 3-ジメチルペンチル (純度 90%, 1.50 g, 4.75匪 ol)および炭酸カリウム (985 mg, 7.13 mmol) の DMF (30 ml) の懸濁液を室温で 30分間、 次いで 30°Cで 2.5 日間攪拌した。 反応液に 1N塩酸 (9.5 ml)を加え、 .水 (10 ml)およびメタノール (15 ml)で希釈し、室温で 60分間攪拌した。析出物を濾取し、メタノール一水 (1:1) およびメタノールで洗浄後、 乾燥して表題化合物 (2.19 g, 4.07 mmol, 85.7¾) を赤褐色粉末として得た。 Compound obtained in Example 3-4 (1.81 g, 4.75 g ol), 5-Eodo-3,3-dimethylpentyl acetate obtained in Reference Example 30 (purity 90%, 1.50 g, 4.75 gall ol) and potassium carbonate A suspension of (985 mg, 7.13 mmol) in DMF (30 ml) was stirred at room temperature for 30 minutes and then at 30 ° C. for 2.5 days. The reaction mixture was diluted with 1N hydrochloric acid (9.5 ml), diluted with water (10 ml) and methanol (15 ml), and stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (2.19 g, 4.07 mmol, 85.7¾) as a red-brown powder.
Ή-N R (300MHz, CDC13) : «50.96 (6H, s), 1.34 (2H, m), 1.59 (2H, t, J= 7.5 Hz), 1.60 (2H, m), 1.79 (3H, q, J= 0.9 Hz), 2.04 (3H, s), 2.26 (3H, q, J= 0.9 Hz), 2:42 (3H, s), 2.90 (2H, m) , 4.11 (2H, t, J= 7.5 Hz), 7.28 (2H, d, J= 8.1 Hz), 7.86 (2H, d, J= 8.1 Hz), 9.62 (1H, brs). Ή-NR (300MHz, CDC1 3 ): «50.96 (6H, s), 1.34 (2H, m), 1.59 (2H, t, J = 7.5 Hz), 1.60 (2H, m), 1.79 (3H, q, J = 0.9 Hz), 2.04 (3H, s), 2.26 (3H, q, J = 0.9 Hz), 2:42 (3H, s), 2.90 (2H, m), 4.11 (2H, t, J = 7.5 Hz), 7.28 (2H, d, J = 8.1 Hz), 7.86 (2H, d, J = 8.1 Hz), 9.62 (1H, brs).
元素分析値 C24H31N305S3として計算値: C, 53.61; H, 5.81; , 7.81; S, 17.89 (%) , 実測値: C, 53.47; H, 5.67; N, 7.84; S, 18.03 (%) · mp. 141.5-143.0°C Calculated elemental analysis C 24 H 31 N 3 0 5 S 3: C, 53.61; H, 5.81;, 7.81; S, 17.89 (%), Found: C, 53.47; H, 5.67 ; N, 7.84; S, 18.03 (%) mp.141.5-143.0 ° C
実施例 4- 553 Example 4-553
N -(8- {[2- (1-ァダマンチル)ェチル]スルファニル } - 5, 6-ジメチル- 3-ォキソ - 2, 3- ジヒドロ _m-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン )_4-メチルベンゼンスル ホンアミド N- (8-{[2- (1-adamantyl) ethyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro_m-imidazo [5,1-c] [1,4] thiazine -1-ylidene) _4-methylbenzenesulfonamide
実施例 3-4で得た化合物 (1.87 g, 4.90 mmol)、 参考例 31で得た卜(2-ョ一 ドエチル)ァダマンタン(純度 86%、 1.53g, 5.27 mmol)および炭酸カリウム (1.02 g, 7.35腿 ol) の DMF (32 ml) の懸濁液を 40°Cで 18時間ついで 60 で 1.5時間 攪拌した。 反応中に上記ヨウ化物(284 mg, 0.98固 ol)を加えた。 反応液に 1N塩 酸 (9.8 ml)を加え、 水 (10 ml)およびメタノール(10 ml)で希釈し、 室温で 60 分間攪拌した。 析出物を濾取し、 メタノール—水 (1:1)およびメタノールで洗浄 後、 THF-ァセトニトリルに溶解して濃縮し、 残渣にメタノールを加えた。 析出物 を濾取し、 メタノールで洗浄後、 乾燥して表題化合物(2.28 g, 4.20匪 ol, 85.7%) を赤紫色粉末として得た。 Compound (1.87 g, 4.90 mmol) obtained in Example 3-4, tri (2-hydroethyl) adamantane (purity 86%, 1.53 g, 5.27 mmol) obtained in Reference Example 31 and potassium carbonate (1.02 g, A suspension of 7.35 tmol) of DMF (32 ml) was stirred at 40 ° C for 18 hours and then at 60 for 1.5 hours. During the reaction, the above iodide (284 mg, 0.98 ol) was added. The reaction mixture was diluted with 1N hydrochloric acid (9.8 ml), diluted with water (10 ml) and methanol (10 ml), and stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, dissolved in THF-acetonitrile and concentrated, and methanol was added to the residue. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (2.28 g, 4.20 bandol, 85.7%) as a red-purple powder.
Ή-NMR (300MHz, CDC13): (51.43 (2H, m), 1.50 (6H, d, J= 2.1 Hz), 1.63 (3H, brd, J= 12.0 Hz), 1.71 (3H, brd, J= 12.3 Hz), 1.79 (3H, q, J= 0.9 Hz), 1.97 (3H, brs), 2.26 (3H, q, J= 0.9 Hz), 2.41 (3H, s), 2.91 (2H, i), 7.27 (2H, d, J= 8.1 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.61 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): (51.43 (2H, m), 1.50 (6H, d, J = 2.1 Hz), 1.63 (3H, brd, J = 12.0 Hz), 1.71 (3H, brd, J = 12.3 Hz), 1.79 (3H, q, J = 0.9 Hz), 1.97 (3H, brs), 2.26 (3H, q, J = 0.9 Hz), 2.41 (3H, s), 2.91 (2H, i), 7.27 (2H, d, J = 8.1 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.61 (1H, brs).
元素分析値: C27H33N303S3: C, 59.64; H, 6.12;. , 7.73; S, 17.69 (%) 、 実測 値: C, 59.66; H, 6.02; N, 7.89; S, 17.83 (%) Elemental analysis: C 27 H 33 N 3 0 3 S 3: C, 59.64; H, 6.12 ;., 7.73; S, 17.69 (%), Found: C, 59.66; H, 6.02 ; N, 7.89; S , 17.83 (%)
即. 189.5-190.5°C Immediately. 189.5-190.5 ° C
実施例 4- 554 Example 4-554
N - {8- [(1-ァダマンチルメチル)スルファニル ]-5, 6-ジメチル -3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン -1-ィリデン卜 4-メチルベンゼンスルホン アミド N- {8-[(1-adamantylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine- 1-ylidene 4-methylbenzenesulfonamide
実施例 3-4で得た化合物(1.85 g, 4.84 mmol), 参考例 32で得た 1-ョードメチ ルァダマンタン(1.43 g, 5.18 mmol)および炭酸カリウム (1.01 g, 7.27 mmol) の DMF (30 ml) の懸濁液を室温で 30分間ついで 60°Cで 42時間攪拌した。 反応液に 1N塩酸 (9.7 mi)を加え、 水 (10 ml)およびメタノール(10 ml)で希釈し、 室温で 30分間攪拌した。析出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗 浄後、 乾燥して表題化合物(1.16 g, 2.19腿 ol, 45.2 )を赤褐色粉末として得た。 Ή-NMR (300MHz, CDC13) : 51.55 (6H, d, J= 2.4 Hz), 1.60 (3H, brd, J= 12.3 Hz), 1.71 (3H, brd, J= 12.3 Hz), 1.78 (3H, q, J= 0.9 Hz), 2.00 (3H, brs), 2.26 (3H, q, J= 0.9 Hz), 2.40 (3H, s), 2.75 (2H, s), 7.25 (2H, d, J= 7.8 Hz), 7.87 (2H, d, J= 8.1 Hz), 9.55 (1H, brs). DMF (30 ml) of the compound obtained in Example 3-4 (1.85 g, 4.84 mmol), 1-odomethyladamantane obtained in Reference Example 32 (1.43 g, 5.18 mmol) and potassium carbonate (1.01 g, 7.27 mmol) Was stirred at room temperature for 30 minutes and then at 60 ° C. for 42 hours. To the reaction mixture was added 1N hydrochloric acid (9.7 mi), diluted with water (10 ml) and methanol (10 ml). Stir for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (1.16 g, 2.19 pt, 45.2) as a red-brown powder. Ή-NMR (300MHz, CDC1 3 ): 51.55 (6H, d, J = 2.4 Hz), 1.60 (3H, brd, J = 12.3 Hz), 1.71 (3H, brd, J = 12.3 Hz), 1.78 (3H, q, J = 0.9 Hz), 2.00 (3H, brs), 2.26 (3H, q, J = 0.9 Hz), 2.40 (3H, s), 2.75 (2H, s), 7.25 (2H, d, J = 7.8 Hz), 7.87 (2H, d, J = 8.1 Hz), 9.55 (1H, brs).
元素分析値: C26H31N303S3として計算値: C, 58.95; H, 5.90; N, 7.93; S, 18.16Elemental analysis: C 26 H 31 N 3 0 3 S 3 Calculated: C, 58.95; H, 5.90 ; N, 7.93; S, 18.16
(%) 、 実測値: C, 58.59; H, 5.95; N, 7.96; S, 18.09 ( ) . (%), Found: C, 58.59; H, 5.95; N, 7.96; S, 18.09 ().
mp. 207.0-209.0°C mp. 207.0-209.0 ° C
実施例 4- 555 Example 4-555
(2S, 4R) -2- { [ (5, 6-ジメチル-卜 { [ (4-メチルフエニル)スルホニル]イミノ } -3-ォキ ソ -2,,3 -ジヒドロ- 1H-ィミダゾ [5, 1- c] [1,4]チアジン- 8-ィル)スルファニル]メチ ル}- 4-ヒドロキシ -1-ピロリジンカルボン酸 tert-ブチル (2S, 4R) -2-{[(5,6-dimethyl-tolu [[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3, -dihydro-1H-imidazo [5,1 -c] [1,4] thiazin-8-yl) sulfanyl] methyl} -tert-butyl 4-hydroxy-1-pyrrolidinecarboxylate
(方法 1) 実施例 3-4で得た化合物(1.35 g, 3.54謹 ol)、 ヨウ化ナトリウム(0.53 g, 3.54腿01) および炭酸カリウム (734 mg, 5.31 mmol) の DMF (25 ml) の懸 濁液に 60°Cで攪拌しながら参考例 33-1で得た (2S,4R)- N- Boc- 4- (2-テトラヒド ロビラニルォキシ)ピロリジン- 2 -メチルメシレート(1.35 g, 3.55漏 1) の DMF (5 ml)溶液を 90分間かけて滴下し、 反応液をさらに 20時間攪拌した。 反応液に 1N塩酸 (7. lml)、水(70 ml)および飽和食塩水(15 l)を加え、酢酸ェチル(140 ml) で抽出した。 有機層を ^炭酸ナトリウム、 2%炭酸水素ナトリウム水溶液、 0.05N 塩酸、 5%および飽和の食塩水で順次洗浄した。水洗層を酢酸ェチル (100 ml)で抽 出した。 有機層を硫酸ナトリウムで乾燥後、 濃縮して濃赤色油状物 (1.39 g)を得 た。 これをメタノール (25 ml) に溶解し、 CSA (95 mg, 0.41顧 ol)と室温で 1.5 時間攪拌した。 反応液を濃縮し、 酢酸ェチル (100 ml)で希釈後、 0.05N塩酸、 5¾ および飽和の食塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して濃赤色油 状物(1.39 g)を得た。 これをシリカゲル(10 g)のクロマトグラフィー (酢酸ェチ ルーへキサン、 1:卜7:3) に付し、酢酸ェチルーへキサン(7 :3) の画分を濃縮乾固 後、 酢酸ェチルおよびジェチルエーテルで希釈した。 析出物を濾取し、 ジェチル エーテルで洗浄後、 乾燥して表題化合物(592 mg, 1.02 mmol, 28.8 )を赤色粉末 として得た。 (Method 1) DMF (25 ml) of the compound obtained in Example 3-4 (1.35 g, 3.54 ol), sodium iodide (0.53 g, 3.54 thigh 01) and potassium carbonate (734 mg, 5.31 mmol) The (2S, 4R) -N-Boc-4- (2-tetrahydrobilanyloxy) pyrrolidine-2-methyl mesylate (1.35 g, 3.55 leaks) obtained in Reference Example 33-1 was stirred into the suspension at 60 ° C. A solution of 1) in DMF (5 ml) was added dropwise over 90 minutes, and the reaction solution was further stirred for 20 hours. To the reaction solution were added 1N hydrochloric acid (7.1 ml), water (70 ml) and saturated saline (15 l), and the mixture was extracted with ethyl acetate (140 ml). The organic layer was washed successively with sodium carbonate, 2% aqueous sodium bicarbonate, 0.05N hydrochloric acid, 5% and saturated saline. The washed layer was extracted with ethyl acetate (100 ml). The organic layer was dried over sodium sulfate and concentrated to give a dark red oil (1.39 g). This was dissolved in methanol (25 ml) and stirred with CSA (95 mg, 0.41 ol) at room temperature for 1.5 hours. The reaction mixture was concentrated, diluted with ethyl acetate (100 ml), washed successively with 0.05N hydrochloric acid, 5¾ and saturated saline, dried over sodium sulfate, and concentrated to give a dark red oil (1.39 g). Obtained. This was subjected to chromatography on silica gel (10 g) (ethyl acetate, 1: 7: 3 ), and the ethyl acetate-hexane (7: 3) fraction was concentrated to dryness. Diluted with getyl ether. The precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (592 mg, 1.02 mmol, 28.8) as a red powder As obtained.
Ή-NMR (200MHz, CDC13) : (51.48 (9H, s), 1.70 (1H, m), 1.78 (3H, q, J= 1.2 Hz), 2.01 (1H, ddd, J= 13.2, 8.0, 4.8 Hz) , 2. ίθ (1H, m), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 3.15 (1H, m), 3.36 (1H, dd, J= 11.6, 3.6 Hz), 3.40 (1H, brd, J= 12 Hz), 3.60 (1H, m), 4.25 (1H, m), 4.41 (1H, m), 7.30 (2H, d, J= 8.2 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): (51.48 (9H, s), 1.70 (1H, m), 1.78 (3H, q, J = 1.2 Hz), 2.01 (1H, ddd, J = 13.2, 8.0, 4.8 Hz), 2.ίθ (1H, m), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 3.15 (1H, m), 3.36 (1H, dd, J = 11.6, 3.6 Hz ), 3.40 (1H, brd, J = 12 Hz), 3.60 (1H, m), 4.25 (1H, m), 4.41 (1H, m), 7.30 (2H, d, J = 8.2 Hz), 7.83 (2H , d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C25H32N406S3として計算値: C, 51.70; H, 5.55; , 9.65; S, 16.56Elemental analysis: Calculated as C 25 H 32 N 4 0 6 S 3: C, 51.70; H, 5.55;, 9.65; S, 16.56
(%) 、 実測値: C, 51.59; H, 5.34; , 9.71; S, 16.55 ( ) . (%), Found: C, 51.59; H, 5.34;, 9.71; S, 16.55 ().
mp. 166.5-168.5 mp. 166.5-168.5
(方法 2) 実施例 3- 4で得た化合物(473 mg, 1.24匪 ol)および炭酸カリウム (257 mg, 1.86画 ol) の DMF (8 ml) の懸濁液にしながら参考例 33-2 で得た ヨウ化 (2S,4R)-N- Boc-4-ヒドロキシピロリジン- 2-メチル (406 mg, 1.24腿 ol)を加え、 反応液を 50DCで 15時間攪拌した。 反応液に 1N塩酸 (2.5il), 水 (20ml)および 飽和食塩水(5 ml)を加え、 酢酸ェチル(50 ml および 20 ml)で抽出した。 有機層 を 5%および飽和の食塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮し、 残 渣に酢酸ェチルおよびジェチルエーテルを加えた。 析出物を濾取し、 ジェチルェ —テルで洗浄後、 乾燥して表題化合物(508 mg, 0.875 mmol, 70.5%)を赤色粉末と して得た。 (Method 2) In Reference Example 33-2, a suspension of the compound obtained in Example 3-4 (473 mg, 1.24 ol) and potassium carbonate (257 mg, 1.86 ol) in DMF (8 ml) was prepared. The obtained (2S, 4R) -N-Boc-4-hydroxypyrrolidine-2-methyl iodide (406 mg, 1.24 liter) was added, and the reaction solution was stirred at 50 DC for 15 hours. 1N Hydrochloric acid (2.5 il), water (20 ml) and saturated saline (5 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (50 ml and 20 ml). The organic layer was washed sequentially with 5% and saturated saline, dried over sodium sulfate, concentrated, and ethyl acetate and getyl ether were added to the residue. The precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (508 mg, 0.875 mmol, 70.5%) as a red powder.
(方法 3)参考例 33-1 で得た(2S, 4R)- N- Boc- 2-ヒドロキシメチル -4- (2-テトラピ ラニルォキシ)ピロリジン (5.50 g, 18.2腿 ol)およびメタンスルホニルクロリ ド (2.22 g, 19.2腿 ol)の THF (100 ml)溶液に 0°Cで攪拌しながらトリェチルアミ ン(3.04 ml, 21.9 mmol) の THF (10 ml)溶液を 40分間かけて滴下し、 反応液を 0°Cで 1時間攪拌した。 酢酸ェチル抽出により精製し、 (2S,4R)_N- Boc-4_(2-テト ラビラニルォキシ)ピロリジン- 2-メチルメシレート(6.74 g, 17.7匪。1, 97.6%) の無色油状物を得た。 この油状物およびヨウ化ナトリウム(5.32 g, 35.5匪 ol) の アセトン (130 ml)懸濁液を 20時間加熱還流した。 反応液を酢酸ェチルーへキサ ン(2:1)により抽出、脱水乾燥、濃縮乾固して粗製の(2S,4R)-N-Boc- 2-ョードメチ ル- 4- (2-テトラピラニルォキシ)ピロリジン(純度 70%, 3.92g, 6.67 mmol, 37.7%) を褐色油状物として得た。 実施例 3- 4で得た化合物(2.55 g, 6.68腿 ol)、 上記(2S, 4R)- N-Boc- 2-ョード メチル -4- (2-テトラピラニルォキシ)ピロリジン(純度 70 3.92 g, 6.67 mmol) および炭酸カリウム (1.39 g, 10.0匪 ol) の DMF (43 ml)の懸濁液を室温で 10 分間ついで 60°Cで 14時間攪拌した。反応液に 1N塩酸 (13.4ml)を加えて、 酢酸 ェチルで抽出し、 赤色油状物(4.64 g)を得た。 これをメタノール(100 ml)に溶解 し濃塩酸 (0.166 ml, 2.01匪 ol)を加えて、 室温で 18時間攪拌した。 反応液に p- トルエンスルホン酸水和物 (255 mg, 1.34 mmol) を加えて、 60°Cで 1時間攪拌 した。 反応液を中和後、 酢酸ェチルで抽出し、 濃赤色油状物を得、 これをシリカ ゲル (40 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:卜7:3) で精製し、 酢酸ェチルージェチルエーテルから結晶化して表題化合物(1.31 g, 2.26讓 ol, 33.8¾)を赤色粉末として得た。 (Method 3) The (2S, 4R) -N-Boc-2-hydroxymethyl-4- (2-tetrapyranyloxy) pyrrolidine (5.50 g, 18.2 t) obtained in Reference Example 33-1 and methanesulfonyl chloride ( A solution of triethylamine (3.04 ml, 21.9 mmol) in THF (10 ml) was added dropwise to a solution of 2.22 g, 19.2 mol) in THF (100 ml) with stirring at 0 ° C over 40 minutes. The mixture was stirred at ° C for 1 hour. Purification by ethyl acetate extraction gave a colorless oil of (2S, 4R) _N-Boc-4_ (2-tetrabiranyloxy) pyrrolidine-2-methyl mesylate (6.74 g, 17.7 marshal, 1, 97.6%). A suspension of this oil and sodium iodide (5.32 g, 35.5 marol) in acetone (130 ml) was heated under reflux for 20 hours. The reaction mixture was extracted with ethyl acetate-hexane (2: 1), dehydrated, dried and concentrated to dryness to give crude (2S, 4R) -N-Boc-2-odomethyl-4- (2-tetrapyranyloxy) ) Pyrrolidine (purity 70%, 3.92 g, 6.67 mmol, 37.7%) was obtained as a brown oil. The compound obtained in Example 3-4 (2.55 g, 6.68 t ol), the above (2S, 4R) -N-Boc-2-odomethyl-4- (2-tetrapyranyloxy) pyrrolidine (purity 70 3.92 g) , 6.67 mmol) and potassium carbonate (1.39 g, 10.0 bandol) in DMF (43 ml) was stirred at room temperature for 10 minutes and then at 60 ° C for 14 hours. 1N Hydrochloric acid (13.4 ml) was added to the reaction solution, and the mixture was extracted with ethyl acetate to obtain a red oil (4.64 g). This was dissolved in methanol (100 ml), concentrated hydrochloric acid (0.166 ml, 2.01 marl) was added, and the mixture was stirred at room temperature for 18 hours. To the reaction solution was added p-toluenesulfonic acid hydrate (255 mg, 1.34 mmol), and the mixture was stirred at 60 ° C for 1 hour. The reaction solution was neutralized and extracted with ethyl acetate to obtain a dark red oily substance, which was purified by chromatography on silica gel (40 g) (ethyl acetate-hexane, 1: 7: 3). The title compound (1.31 g, 2.26 mL, 33.8%) was obtained as a red powder by crystallizing from chillue tyl ether.
実施例 4-556 Example 4-556
酢酸 (2 - {[(5, 6 -ジメチル-卜 {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2,3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン- 8_ィル)スルファニル]メチル } シクロへキシルメチル)メチル Acetic acid (2-{[(5,6-dimethyl-tolu [[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-8_yl) sulfanyl] methyl} cyclohexylmethyl) methyl
実施例 3-4で得た化合物(1.85 g, 4.85 mmol), 参考例 34で得た trans- 2-ョー ドメチルシクロへキサン- 1_メチルアセテート(1.48 g, 5.00 mmol)および炭酸力 リウム (1.01 g, 7.27 mmol) の DMF (30 ml) の懸濁液を室温で 30分間ついで 60°Cで 41時間攪拌した。 反応液に 1N塩酸 (9.7 ml)を加え、 水 (10 ml)および メタノール(10 ml)で希釈し、 室温で 60分間攪拌した。 析出物を濾取し、 メタノ 一ルー水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物(2.41 g, 4.39 蘭 ol, 90.5%)を赤色粉末として得た。 ' The compound obtained in Example 3-4 (1.85 g, 4.85 mmol), trans-2-odomethylcyclohexane-1_methyl acetate obtained in Reference Example 34 (1.48 g, 5.00 mmol) and lithium carbonate (1.01 g) , 7.27 mmol) in DMF (30 ml) was stirred at room temperature for 30 minutes and then at 60 ° C for 41 hours. 1N Hydrochloric acid (9.7 ml) was added to the reaction solution, diluted with water (10 ml) and methanol (10 ml), and stirred at room temperature for 60 minutes. The precipitate was collected by filtration, washed with methanol (1: 1) and methanol, and dried to give the title compound (2.41 g, 4.39 orchid ol, 90.5%) as a red powder. '
Ή— NMR (300MHz, CDC13) : (51.07 (1H, brq, J= 12.6 Hz), 1.16 (1H, brq, J= 13.2 Hz), 1.22 (2H, brq, J= 7.5 Hz), 1.54 (2H, m), 1.75 (3H, brs), 1.78 (3H, q, J= 0.9 Hz), 1.95 (1H, brd, J= 14.1 Hz), 2.06 (3H, s), 2.26 (3H, q, J= 0.9 Hz), 2.41 (3H, s), 2.75 (1H, dd, J= 12.6, 8.1 Hz), 3.29 (1H, dd, J= 12.6, 3.0 Hz), 4.03 (1H, dd, J= 11.1, .5 Hz), 4.05 (1H, dd, J= 11.4, 4.2 Hz) , 7.27 (2H, dt, J= 8.4, 0.6 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.58 (1H, brs). Ή- NMR (300MHz, CDC1 3) : (51.07 (1H, brq, J = 12.6 Hz), 1.16 (1H, brq, J = 13.2 Hz), 1.22 (2H, brq, J = 7.5 Hz), 1.54 (2H , m), 1.75 (3H, brs), 1.78 (3H, q, J = 0.9 Hz), 1.95 (1H, brd, J = 14.1 Hz), 2.06 (3H, s), 2.26 (3H, q, J = 0.9 Hz), 2.41 (3H, s), 2.75 (1H, dd, J = 12.6, 8.1 Hz), 3.29 (1H, dd, J = 12.6, 3.0 Hz), 4.03 (1H, dd, J = 11.1,. 5 Hz), 4.05 (1H, dd, J = 11.4, 4.2 Hz), 7.27 (2H, dt, J = 8.4, 0.6 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.58 (1H, brs) .
元素分析値: C25H31N305S3として計算値: C, 54.62; H, 5.68; N, 7.64; S, 17.50 (%) 、 実測値: C, 54.48; H, 5.78; , 7.52; S, 17.44 (%) . mp. 147.5-148.0°C Elemental analysis: C 25 H 31 N 3 0 5 S 3 Calculated: C, 54.62; H, 5.68 ; N, 7.64; S, 17.50 (%), Found: C, 54.48; H, 5.78;, 7.52; S, 17.44 (%) .mp. 147.5-148.0 ° C
実施例 4- 557 Example 4-557
N - [5, 6 -ジメチル- 3-ォキソ -8- ({[ (2S) -5 -ォキソピ口リジニル]メチル }スルファ二 ル) - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1 - c] [1, 4]チアジン- 1 -ィリデン ]-4-メチルベン ゼンスルホンアミド N- [5,6-dimethyl-3-oxo-8-({[(2S) -5-oxopiperidinyl] methyl} sulfanyl)-2,3-dihydro-1H-imidazo [5,1-c ] [1, 4] thiazine-1-ylidene] -4-methylbenzenesulfonamide
実施例 3-4で得た化合物(1.81 g, 4.74 mmol), (2S)- 5-ヒドロキシメチル- 2 - ピロリドン(619 mg, 5.21 mmol) およびトリフエニルホスフィン(1.67 g, 6.16 腿 ol)の THF (36 ml)の懸濁液に室温で攪拌しながら DEAD (1.02 ml, 6.16 mmol) を滴下し、反応液を室温で 19時間攪拌した。反応液を酢酸ェチルで抽出し、脱水 乾燥、 濃縮して得られた赤色油状物にメタノールを加えた。 析出物を濾取し、 メ 夕ノールおよびジェチルエーテルで洗浄後、 乾燥して表題化合物(1.26 g, 2.63 mmol, 55.4 )を赤紫色粉末として得た。 THF of the compound obtained in Example 3-4 (1.81 g, 4.74 mmol), (2S) -5-hydroxymethyl-2-pyrrolidone (619 mg, 5.21 mmol) and triphenylphosphine (1.67 g, 6.16 mol) DEAD (1.02 ml, 6.16 mmol) was added dropwise to the suspension (36 ml) while stirring at room temperature, and the reaction solution was stirred at room temperature for 19 hours. The reaction solution was extracted with ethyl acetate, dehydrated, dried and concentrated, and methanol was added to the obtained red oil. The precipitate was collected by filtration, washed with methanol and getyl ether, and dried to give the title compound (1.26 g, 2.63 mmol, 55.4) as a red-purple powder.
一 NMR (200MHz, CDC13) : 51.79 (3H, q, J= 0.6 Hz), 1.82 (1H, m), 2.26 (3H, q, J= 1.0 Hz), 2.30 (1H, m), 2.36 (2H, m), 2.43 (3H, s), 2.96 (1H, dd, J= 14.0, 7.8 Hz), 3.12 (1H, dd, J= 13.8, 5.0 Hz), 3.88 (1H, brquint, J= 6.4 Hz), 5.90 (1H, brs), 7.32 (2H, d, J= 8.0 Hz), 7.84 (2H, d, J= 8.6 Hz), 9.69 (1H, brs) . One NMR (200MHz, CDC1 3): 51.79 (3H, q, J = 0.6 Hz), 1.82 (1H, m), 2.26 (3H, q, J = 1.0 Hz), 2.30 (1H, m), 2.36 (2H , m), 2.43 (3H, s), 2.96 (1H, dd, J = 14.0, 7.8 Hz), 3.12 (1H, dd, J = 13.8, 5.0 Hz), 3.88 (1H, brquint, J = 6.4 Hz) , 5.90 (1H, brs), 7.32 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.6 Hz), 9.69 (1H, brs).
元素分析値: C20H22N4O4S3 O.5H20: C, 49.26; H, 4.75; N, 11.49; S, 19.73 (%) 、 実測値: C, 48.99; H, 4.63; N, 11.30; S, 19.34 (%) . Elemental analysis: C 20 H 22 N 4 O 4 S 3 O.5H 2 0: C, 49.26; H, 4.75; N, 11.49; S, 19.73 (%), Found: C, 48.99; H, 4.63 ; N, 11.30; S, 19.34 (%).
実施例 4-558 Example 4-558
3-{[(5, 6-ジメチル-卜 {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 3- ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン - 8-ィル)スルファニル]メチル } -卜ピ ペリジンカルボン酸 tert-ブチル 3-{[(5,6-dimethyl-tolu [[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -8-yl) sulfanyl] methyl} -tert-butyl topiperidinecarboxylate
実施例 3- 4で得た化合物(1.82 g, 4.77腿 ol)、 3-ヒドロキシメチルピベリジ ンおよび重炭酸ジ tert-ブチルから調製した N-Boc-3-ヒドロキシメチルピベリジ ン(1.13 g, 5.24 mmol) およびトリフエニルホスフィン(1· 68 g, 6.20 mmol)の THF (36 ml)の懸濁液に室温で攪拌しながら DEAD (1.03 ml, 6.20 mmol)を滴下し、 反応液を室温で 24時間攪拌した。反応液を酢酸ェチルで抽出し、 脱水乾燥、濃縮 して得られた赤色油状物をシリカゲル(30 g)のクロマトグラフィー (酢酸ェチル —へキサン、 2:8-5.5)で精製し、酢酸ェチルージェチルェ一テルから結晶化して 表題化合物(587 mg, 1.01 mmol, 21.2%)を赤紫色粉末として得た。 N-Boc-3-hydroxymethylpiberidine (1.13 g) prepared from the compound obtained in Example 3-4 (1.82 g, 4.77 tmol), 3-hydroxymethylpiberidine and ditert-butyl bicarbonate. g, 5.24 mmol) and triphenylphosphine (1.68 g, 6.20 mmol) in THF (36 ml) were added dropwise with stirring at room temperature while DEAD (1.03 ml, 6.20 mmol) was added dropwise. For 24 hours. The reaction solution was extracted with ethyl acetate, dehydrated, dried and concentrated The resulting red oil was purified by chromatography on silica gel (30 g) (ethyl acetate-hexane, 2: 8-5.5) and crystallized from ethyl acetate-ethyl ether to give the title compound (587 mg, 1.01 mmol, 21.2%) as a red-purple powder.
Ή一 NMR (300MHz, CDC13) : 51.26 (2H, m), 1.45 (9H, s), 1.50 (2H, m), 1.63 (1H, m), 1.78 (3H, q, J= 1.2 Hz), 1.92 (1H, brd, J= 11.7 Hz), 2.26 (3H, q, J= 0.9 Hz), 2.41 (3H, s), 2.78 (1H, dd, J= 11.7, 7.5 Hz), 2.90 (1H, dd, J= 11.7, 6.3 Hz), 2.86 (2H, br), 3.75 (IH, m), 3.90 (1H, br), 7.28 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.1 Hz), 9.61 (IH, brs). Ή one NMR (300MHz, CDC1 3): 51.26 (2H, m), 1.45 (9H, s), 1.50 (2H, m), 1.63 (1H, m), 1.78 (3H, q, J = 1.2 Hz), 1.92 (1H, brd, J = 11.7 Hz), 2.26 (3H, q, J = 0.9 Hz), 2.41 (3H, s), 2.78 (1H, dd, J = 11.7, 7.5 Hz), 2.90 (1H, dd , J = 11.7, 6.3 Hz), 2.86 (2H, br), 3.75 (IH, m), 3.90 (1H, br), 7.28 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.61 (IH, brs).
元素分析値: C26H34N405S3 O.2Et20: C, 54.23; H, 6.11; N, 9.44; S, 16.21 (%) 、 実測値: C, 54.28; H, 6.29; N, 9.08; S, 16.25.(%) . Elemental analysis: C 26 H 34 N 4 0 5 S 3 O.2Et 2 0: C, 54.23; H, 6.11; N, 9.44; S, 16.21 (%), Found: C, 54.28; H, 6.29 ; N, 9.08; S, 16.25. (%).
即. 154.0-155.0DC Immediately. 154.0-155.0 D C
実施例 4- 559 ' Example 4-559 '
(2S)- 2- {[(5,6-ジメチル -1- {[(4_メチルフエニル)スルホニル]イミノ}- 3-ォキソ - 2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィル)スルファニル]メチ ル} -卜ピロリジンカルボン酸 tert-ブチル (2S)-2-{[(5,6-dimethyl-1-{[(4_methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-8-yl) sulfanyl] methyl} -tert-butyl topyrrolidinecarboxylate
(方法 1) 実施例 3- で得た化合物 (1.90 g, 4.98 mmol)、 (S) - N - Boc-2 -ヒド口 キシメチルピロリジン(純度 94.7%, 1.18 g, 5.55腿01) およびトリフエニルホ スフイン(1.75 g, 6.47廳01)の [ (40 ml)の懸濁液に室温で攪拌しながら DEAD (1.07 ml, 6.47 mmol)を滴下し、 反応液を室温で 63時間攪拌した。 反応液を酢酸 ェチルで抽出し、 脱水乾燥、 濃縮して赤色油状物 (5.6 g)を得た。 これをシリカゲ ル(40 g)のクロマトグラフィー (酢酸ェチル一へキサン、 3:7-4:6) で精製し、 酢 酸ェチルージェチルエーテル、 次いでメタノ一ルから結晶化して表題化合物(964 mg, 1.71 mmol, 34.3»を紫色粉末として得た。 (Method 1) The compound obtained in Example 3- (1.90 g, 4.98 mmol), (S) -N-Boc-2-hydridoxymethylpyrrolidine (purity 94.7%, 1.18 g, 5.55 thigh 01) and triphenylphosphine DEAD (1.07 ml, 6.47 mmol) was added dropwise to a suspension of [(40 ml) in (1.75 g, 6.47 Cafe 01) with stirring at room temperature, and the reaction solution was stirred at room temperature for 63 hours. The reaction solution was extracted with ethyl acetate, dehydrated and dried, and concentrated to obtain a red oil (5.6 g). This was purified by chromatography on silica gel (40 g) (ethyl acetate-hexane, 3: 7-4: 6) and crystallized from ethyl acetate-ethyl ether and then methanol to give the title compound (964 mg, 1.71 mmol, 34.3 »as a purple powder.
Ή-NMR (200MHz, CDC13) : δΐ.47 (9Η, s), 1.78 (3H, q, J= 0.8 Hz), 1.83 (3H, m), 1.98 (IH, m), 2.26 (3H, s), 2.42 (3H, s), 2.90-3.10 (IH, m), 3.33 (2H, m), 3.40 (IH, m), 4.01 (IH, m), 7.28 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.61 (IH, brs). Ή-NMR (200MHz, CDC1 3 ): δΐ.47 (9Η, s), 1.78 (3H, q, J = 0.8 Hz), 1.83 (3H, m), 1.98 (IH, m), 2.26 (3H, s ), 2.42 (3H, s), 2.90-3.10 (IH, m), 3.33 (2H, m), 3.40 (IH, m), 4.01 (IH, m), 7.28 (2H, d, J = 8.4 Hz) , 7.84 (2H, d, J = 8.4 Hz), 9.61 (IH, brs).
元素分析値: C25H32N405S3として計算値: C, 53.17; H, 5.71; N, 9.92; S, 17.03 (%) 、 実測値: C, 53.06; H, 5.74; N, 9.70; S, 17.06 ( ) . mp. 168.0-168.5t: Elemental analysis: C 25 H 32 N 4 0 5 S 3 Calculated: C, 53.17; H, 5.71 ; N, 9.92; S, 17.03 (%), Found: C, 53.06; H, 5.74 ; N, 9.70; S, 17.06 (). mp. 168.0-168.5t:
(方法 2) (S)- N- Boc- 2-ヒドロキシメチルピロリジン(純度 94.7 , 1.05 g, 4.94 mmol)およびメタンスルホニルクロリド (0.406 ml, 5.19龍 ol)の DMF (10 ml)溶液に 0°Cで攪拌しながらトリェチルァミン(0.822 ml, 5.93讓 ol)の DMF (1 ml)溶液を 30分間かけて滴下し、 反応液を Ot:で 1時間攪拌した。 反応液を実施 例 3-4で得た化合物(1.45 g, 3.79 mmol)および炭酸カリウム (1.05g, 7.58讓01) の DMF (15 ml) の懸濁液に 40°Cで 40分間かけて滴下し、 反応液を 40°Cで 18時 間、 ついで 80°Cで 2時間攪拌した。 反応液に 1N塩酸 (12 ml)を加えて酢酸ェチ ルで抽出し、脱水乾燥、濃縮して赤色油状物 (5.6 g)を得た。 これをシリカゲル (40 g)のクロマトグラフィー (酢酸ェチルーへキサン、 3:7-4:6) で精製し、 メタノ一 ルから結晶化して表題化合物(650 mg, 1.15匪 ol, 30.4%)を紫色粉末として得た。 実施例 4 - 560 (Method 2) To a solution of (S) -N-Boc-2-hydroxymethylpyrrolidine (purity 94.7, 1.05 g, 4.94 mmol) and methanesulfonyl chloride (0.406 ml, 5.19 r.l.ol) in DMF (10 ml) at 0 ° C While stirring with, a solution of triethylamine (0.822 ml, 5.93 mL) in DMF (1 ml) was added dropwise over 30 minutes, and the reaction solution was stirred with Ot: for 1 hour. The reaction solution was added dropwise at 40 ° C over 40 minutes to a suspension of the compound obtained in Example 3-4 (1.45 g, 3.79 mmol) and potassium carbonate (1.05 g, 7.58 substitution 01) in DMF (15 ml). Then, the reaction solution was stirred at 40 ° C. for 18 hours, and then at 80 ° C. for 2 hours. The reaction mixture was added with 1N hydrochloric acid (12 ml), extracted with ethyl acetate, dehydrated and dried, and concentrated to give a red oil (5.6 g). This was purified by chromatography on silica gel (40 g) (ethyl acetate-hexane, 3: 7-4: 6) and crystallized from methanol to give the title compound (650 mg, 1.15 marl, 30.4%) in purple Obtained as a powder. Example 4-560
N- {1, 6-ジォキソ -4 - [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル] - 1, 2, 6, 7, 8, 9-へキサヒドロ- 3H -イミダゾ [5, 1 - c] [1, 4]ベンゾチアジン- 3 -ィ 実施例 3-38で得られた N -(1, 6-ジォキソ- 4-スルファニル- 1, 2, 6, 7, 8, 9 -へキサ ヒドロ- 3H-イミダゾ [5, 1-c] [1,4]ベンゾチアジン- 3-ィリデン )-4-メチルベンゼ ンスルホンアミド (300 mg, 0.712匪 ol) と炭酸カリウム (148 mg, 1.07 mmol) の N,N-ジメチルホルムアミド (5.00 ml) の懸濁液に、 参考例 9で得られたメタ ンスルホン酸 5, 5, 6, 6, 6-ペンタフルォ口へキシルエステル(231 mg, 0.854腿 ol) の Ν,Ν-ジメチルホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を 80 で 2 時間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04)後、 溶媒を減圧下に留去した。残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル(10:1, v/v)で溶出して題記化合物(L49 g, 89%) を暗赤色結晶として得た。酢酸ェチル -ジェチルエーテルから再結晶して 融点 194-196°Cの暗赤色結晶を得た。 N- {1,6-dioxo-4-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]-1,2,6,7,8,9-hexahydro-3H -Imidazo [5,1-c] [1,4] benzothiazine-3-y N- (1,6-dioxo-4-sulfanyl-1,2,6,7,8 obtained in Example 3-38 , 9-Hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-3-ylidene) -4-methylbenzensulfonamide (300 mg, 0.712 bandol) and potassium carbonate (148 mg, 1.07 mmol) of N, N-dimethylformamide (5.00 ml) was added to a suspension of methanesulfonic acid 5,5,6,6,6-pentafluoromethylhexyl ester (231 mg, 0.854) obtained in Reference Example 9. A solution of t-ol) in Ν, Ν-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 for 2 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (L49 g, 89%) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 194-196 ° C.
IR (KBr) V: 3220, 2938, 1759, 1651, 1624, 1595, 1553 cm"1.IR (KBr) V: 3220, 2938, 1759, 1651, 1624, 1595, 1553 cm " 1 .
-醒 R (CDC13) : (51.66-1.86 (4H, m), 1.96-2.15 (4H, m), 2.42 (3H, s), 2.51- Awakening: R (CDC1 3): (51.66-1.86 (4H, m), 1.96-2.15 (4H, m), 2.42 (3H, s), 2.51
(2H, t, J=6.3 Hz), 3.07 (2H, t, J=7.5 Hz), 3.22 (2H, i, J=6.0Hz), 7.30 (2H, d, J=7.5 Hz), 7.84 (2H, d, J=8.7 Hz), 9.87 (1H, bs). (2H, t, J = 6.3 Hz), 3.07 (2H, t, J = 7.5 Hz), 3.22 (2H, i, J = 6.0Hz), 7.30 (2H, d, J = 7.5 Hz), 7.84 (2H, d, J = 8.7 Hz), 9.87 (1H, bs).
元素分析値: C23H22N304S3F5 -0.4H20として Elementary analysis: as C 23 H 22 N 3 0 4 S 3 F 5 -0.4H 2 0
計算値 ) : C, 45.83; H, 3.81; , 6.97; S, 15.96. Calculated)): C, 45.83; H, 3.81;, 6.97; S, 15.96.
実測値 (¾) : C, 45.80; H, 3.76; N, 7.06; S, 15.84. Found (¾): C, 45.80; H, 3.76; N, 7.06; S, 15.84.
実施例 4-561 Example 4-561
N-{1, 6 -ジォキソ- 4- [(5_フルォロペンチル)スルファニル] - 1, 2, 6, 7, 8, 9-へキサ ヒドロ- 3H-イミダゾ [5,卜 c] [1,4]ベンゾチアジン- 4 -ィリデン }_4-メチルベンゼ ンスルホンアミド N- {1,6-Dioxo-4-[(5_fluoropentyl) sulfanyl] -1,2,6,7,8,9-hexahydro-3H-imidazo [5, tric] [1,4] Benzothiazine-4-ylidene} _4-methylbenzenesulfonamide
実施例 3 - 38で得られた N- (1, 6 -ジォキソ- 4-スルファニル- 1, 2, 6, 7, 8, 9-へキサヒ ドロ- 3H -イミダゾ [5,1- c] [1,4]ベンゾチアジン - 4 -ィリデン)_4-メチルベンゼン スルホンアミド (2.00 g, 4.74 mmol) と炭酸カリウム (984 mg, 7.12顏 ol) の Ν,Ν -ジメチルホルムアミド (50.0ml) の懸濁液に、 参考例 61で得られた卜プロ モ _5_フルォロペンタン (962 mg, 5.69匪01) を加えた。 反応混合物を 80 で 30 分間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (2.07 g, 86¾) を暗赤色結晶 として得た。 酢酸ェチル-テトラヒドロフラン-メ夕ノ一ルから再結晶して融点 235- 237 の暗赤色結晶を得た。 N- (1,6-Dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo obtained in Example 3-38 [5,1-c] [1 [, 4] Benzothiazine-4-ylidene) _4-methylbenzenesulfonamide (2.00 g, 4.74 mmol) and potassium carbonate (984 mg, 7.12 faceol) in Ν, Ν-dimethylformamide (50.0 ml) Topromo_5_fluoropentane obtained in Reference Example 61 (962 mg, 5.69 bandages 01) was added. After heating the reaction mixture at 80 for 30 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (2.07 g, 86¾) and the obtained as dark red crystals. The crystals were recrystallized from ethyl acetate-tetrahydrofuran-methyl alcohol to obtain dark red crystals having a melting point of 235-237.
IR (KBr) V: 3169, 3059, 2961, 2942, 2909, 2253, 1740, 1649, 1609, 1584, 1547 cm一1. IR (KBr) V: 3169, 3059, 2961, 2942, 2909, 2253, 1740, 1649, 1609, 1584, 1547 cm- 1 .
Ή-NMR (CDC13) : «51.50-1.90 (6H, s), 1.98-2.14 (2H, m), 2.43 (3H, s), 2.51 (2H, t, J=7.0 Hz), 3.05 (2H, t, J=7.0Hz), 3.21 (2H, t, J=6.0 Hz), 4.45 (2H, dt, J=47.2, 6.0 Hz), 7.30 (2H, d, J=8. Hz), 7.84 (2H, d, J=8. Hz), 9.85 (1H, bs). Ή-NMR (CDC1 3): «51.50-1.90 (6H, s), 1.98-2.14 (2H, m), 2.43 (3H, s), 2.51 (2H, t, J = 7.0 Hz), 3.05 (2H, t, J = 7.0Hz), 3.21 (2H, t, J = 6.0 Hz), 4.45 (2H, dt, J = 47.2, 6.0 Hz), 7.30 (2H, d, J = 8.Hz), 7.84 (2H , d, J = 8.Hz), 9.85 (1H, bs).
元素分析値: C22H24N304S3Fとして Elementary analysis: as C 22 H 24 N 3 0 4 S 3 F
計算値 (¾) : C, 51.85; H, 4.75; N, 8.25; S, 18.88. Calculated value (¾): C, 51.85; H, 4.75; N, 8.25; S, 18.88.
実測値 (¾) : C, 51.84; H, 4.62; N, 8.18; S, 18.64. Found (¾): C, 51.84; H, 4.62; N, 8.18; S, 18.64.
実施例 4- 562 Example 4-562
N- {1,6-ジォキソ- 4- [(5, 5, 5-ト リ フルォロペンチル)スルファニル] - 1,2,6, 7, 8, 9 -へキサヒドロ- 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン- 4 -ィリデ 実施例 3-38で得られた N -(1, 6 -ジォキソ -4-スルファニル- 1, 2, 6, 7, 8, 9 -へキサヒ ドロ- 3H-イミダゾ [5,卜 c] [1,4]ベンゾチアジン- 4-ィリデン)_4-メチルベンゼン スルホンアミド (200 mg, 0.443匪 ol) と炭酸カリウム (91.8 mg, 0.664 mmol) の Ν,Ν-ジメチルホルムアミド (50.0ml) の懸濁液に、 参考例 53-4で得られたメ タンスルホン酸 5, 5, 5-トリフルォロペンチルエステル (117 mg, 0.532 mmol) の N,N-ジメチルホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を 80°Cで 1時 間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(121 ig, 50%)を暗赤色結晶として得た。 酢酸ェチル -テトラヒドロフラン-メ夕ノ一ルから再結晶して融点 235- 237 の暗 赤色結晶を得た。 N- {1,6-dioxo-4-[(5,5,5-trifluoropentyl) sulfanyl]-1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c ] [1, 4] Benzothiazine-4-ylide N- (1,6-Dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo [5, tric] [1, obtained in Example 3-38 4] Benzothiazine-4-ylidene) _4-methylbenzenesulfonamide (200 mg, 0.443 ol) and potassium carbonate (91.8 mg, 0.664 mmol) in Ν, Ν-dimethylformamide (50.0 ml) suspension A solution of methanesulfonic acid 5,5,5-trifluoropentyl ester (117 mg, 0.532 mmol) obtained in Example 53-4 in N, N-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 ° C for 1 hour, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (121 ig, 50%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-tetrahydrofuran-methyl to obtain dark red crystals having a melting point of 235-237.
IR (KBr) レ : 3177, 3071, 2965, 2882, 2259, 1740, 1655, 1609, 1586, 1549 cm—1. Ή-NMR (CDC13) : δ 1.60-1.90 (4Η, s), 1.97-2.21 (4H, m), 2.42 (3H, s), 2.51 (2H, t, J=7.0 Hz), 3.05 (2H, t, J=7.0Hz), 3.21 (2H, t, J=6.2 Hz), 7.30 (2H, d, J=7.6 Hz), 7.84 (2H, d, J=8.4 Hz), 9.88 (1H, bs). IR (KBr) Les:. 3177, 3071, 2965, 2882, 2259, 1740, 1655, 1609, 1586, 1549 cm- 1 Ή-NMR (CDC1 3): δ 1.60-1.90 (4Η, s), 1.97-2.21 (4H, m), 2.42 (3H, s), 2.51 (2H, t, J = 7.0 Hz), 3.05 (2H, t, J = 7.0 Hz), 3.21 (2H, t, J = 6.2 Hz), 7.30 (2H, d, J = 7.6 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.88 (1H, bs).
元素分析値: C22H22N304S3F3として Elementary analysis: as C 22 H 22 N 3 0 4 S 3 F 3
計算値 (%) : C, 48.43; H, 4.06; N, 7.70; S, 17.63. Calculated value (%): C, 48.43; H, 4.06; N, 7.70; S, 17.63.
― 実測値 (¾) : C, 48.50; H, 4.09; N, 7.73; S, 17.62. ― Actual value (¾): C, 48.50; H, 4.09; N, 7.73; S, 17.62.
実施例 4- 563 Example 4-563
N-{8, 8-ジメチル -1, 6 -ジォキソ - 4_[(5, 5, 6, 6, 6 -ペンタフルォ口へキシル)スルフ ァニル] - 1,2,6,7,8, 9 -へキサヒド口- 3H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン 実施例 3-42 で得られた N- (8,8-ジメチル _1,6-ジォキソ- 4-スルファニル - 1, 2, 6, 7, 8, 9 -へキサヒドロ- 3H-イミダゾ [5, 1-c] [1, 4]ベンゾチアジン - 4 -ィリデ ン)- 4-メチルベンゼンスルホンアミド(200 mg, 0.445讓 ol)と炭酸カリウム(92.2 mg, 0.667 mmol) の N, N -ジメチルホルムアミド (4.00 ml) の懸濁液に、 参考例 9 で得られたメタンスルホン酸 5, 5, 6, 6, 6 -ペン夕フルォ口へキシルエステル (144 mg, 0.534画 ol) の N, N-ジメチルホルムアミド (1.00 ml) 溶液を加えた。 反応 混合物を 80°Cで 30分間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(227 mg, 82¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 234- 236 の暗赤色結晶を得た。 N- {8,8-dimethyl-1,6-dioxo-4 _ [(5,5,6,6,6-pentafluorohexyl) sulfanyl]-1,2,6,7,8,9- Xahydrido-3H-imidazo [5,1-c] [1,4] benzothiazine N- (8,8-dimethyl_1,6-dioxo-4-sulfanyl-1,2,2 obtained in Example 3-42 6,7,8,9-Hexahydro-3H-imidazo [5,1-c] [1,4] benzothiazine-4-ylidene) -4-methylbenzenesulfonamide (200 mg, 0.445 alcohol) and carbonic acid To a suspension of potassium (92.2 mg, 0.667 mmol) in N, N-dimethylformamide (4.00 ml) was added to the methanesulfonic acid 5,5,6,6,6-phenol obtained in Reference Example 9 A solution of xyl ester (144 mg, 0.534 ol) in N, N-dimethylformamide (1.00 ml) was added. After heating the reaction mixture at 80 ° C. for 30 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. Lottery Washed with water exudates, dried (MgSO 4), the solvent was evaporated under reduced pressure to give the title compound (227 mg, 82¾) was obtained as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 234-236.
IR (KBr) V: 3177, 3065, 2957, 2872, 1738, 1661, 1615, 1591, 1549 cm"1. lH - NMR (CDC13) : (51.12 (6H, s), 1.67-1.86 (4H, m), 1.96-2.14 (2H, m), 2.36 (2H, s), 2.42 (3H, s), 3.06 (2H, t, J=7.2 Hz), 3.08 (2H, s), 7.30 (2H, d, J=8.1 Hz), 7.84 (2H, d, J=8.1 Hz), 9.87 (1H, bs). IR (KBr) V:. 3177 , 3065, 2957, 2872, 1738, 1661, 1615, 1591, 1549 cm "1 lH - NMR (CDC1 3): (51.12 (6H, s), 1.67-1.86 (4H, m ), 1.96-2.14 (2H, m), 2.36 (2H, s), 2.42 (3H, s), 3.06 (2H, t, J = 7.2 Hz), 3.08 (2H, s), 7.30 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.87 (1H, bs).
元素分析値: C25H26N304S3F5として Elementary analysis: as C 25 H 26 N 3 0 4 S 3 F 5
計算値 (¾) : C, 48.14; H, 4.20; N, 6.74; S, 15.42. Calculated (¾): C, 48.14; H, 4.20; N, 6.74; S, 15.42.
実測値 (¾) : C, 48.30; H, 4.03; N, 6.70; S, 15.63. Found (¾): C, 48.30; H, 4.03; N, 6.70; S, 15.63.
実施例 4-564 Example 4-564
N-{9, 9 -ジメチル -1, 6-ジォキソ -4-[(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルフ ァニル] - 1,2, 6, 7, 8, 9-へキサヒドロ- 3H-イミダゾ [5, 1-c] [1,4]ベンゾチアジン - 4-ィリデン }- 4-メチルベンゼンスルホンアミ H N- {9,9-dimethyl-1,6-dioxo-4-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -1,2,6,7,8, 9-Hexahydro-3H-imidazo [5, 1-c] [1,4] benzothiazine-4-ylidene} -4-methylbenzenesulfonami H
実施例 4-563と同様の方法により、実施例 3- 43で得られた N- (9, 9-ジメチル -1, 6- ジォキソ -4-スルファニル -1, 2, 6, 7, 8, 9-へキサヒド口- 3H-ィミダゾ [5, 1-c] [1, 4] ベンゾチアジン- 4-ィリデン)-4-メチルベンゼンスルホンアミド (200 mg, 0.445 醒 ol) から題記化合物 (160 mg, 58%) を合成した。 酢酸ェチル -テトラヒドロフ ラン-ジェチルエーテルから再結晶して融点 216- 218 の暗赤色結晶を得た。 IR (KBr) v: 3177, 3073, 2970, 2932, 2865, 2255, 1742, 1649, 1611, 1588, 1549 , cm一1. In the same manner as in Example 4-563, N- (9,9-dimethyl-1,6-dioxo-4-sulfanyl-1,2,6,7,8,9 obtained in Example 3-43 was obtained. -Hexahydrid- 3H-imidazo [5, 1-c] [1, 4] benzothiazine-4-ylidene) -4-methylbenzenesulfonamide (200 mg, 0.445 awake ol) from the title compound (160 mg, 58% ) Was synthesized. The crystals were recrystallized from ethyl acetate-tetrahydrofuran-getyl ether to obtain dark red crystals having a melting point of 216-218. IR (KBr) v: 3177, 3073, 2970, 2932, 2865, 2255, 1742, 1649, 1611, 1588, 1549, cm- 1 .
Ή-NMR (CDC13) : δΐ.15 (6Η, s), 1.67-1.90 (4H, m), 1.86 (2H, t, J=6.0 Hz), 1.95-2.14 (2H, m), 2.42 (3H, s), 3.06 (2H, t, J=7.2 Hz), 3.22 (2H, t, J=6.3 Hz), 7.29 (2H, d, J=8.4 Hz), 7.83 (2H, d, J=8.4 Hz), 9.86 (1H, bs). Ή-NMR (CDC1 3): δΐ.15 (6Η, s), 1.67-1.90 (4H, m), 1.86 (2H, t, J = 6.0 Hz), 1.95-2.14 (2H, m), 2.42 (3H , s), 3.06 (2H, t, J = 7.2 Hz), 3.22 (2H, t, J = 6.3 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz) ), 9.86 (1H, bs).
元素分析値: C25H26N304S3F5として Elementary analysis: as C 25 H 26 N 3 0 4 S 3 F 5
計算値 (%) : C, 48.14; H, 4.20; N, 6.74; S, 15.42. Calculated (%): C, 48.14; H, 4.20; N, 6.74; S, 15.42.
実測値 (%) : C, 48.24; H, 4.14; N, 6.80; S, 15.70. Found (%): C, 48.24; H, 4.14; N, 6.80; S, 15.70.
実施例 4-565 Example 4-565
N-{1, 6 -ジォキソ- 8-メチル -4- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 1,2, 6, 7, 8, 9 -へキサヒドロ- 3H -イミダゾ [5,l-c] [1, 4]ベンゾチアジン- 4-ィ 実施例 4-563と同様の方法により、 実施例 3-44で得られた N- (1, 6_ジォキソ- 8 - メチル -4 -スルファニル -1, 2, 6, 7, 8, 9 -へキサヒド口署ィミダゾ [5,卜 c] [1, 4]ベ ンゾチアジン- 4-ィリデン)-4-メチルベンゼンスルホンアミド (3.51 g, 8.06 mmol) から題記化合物 (4.55 g, 93%) を合成した。 酢酸ェチルテトラヒドロフ ラン-ジェチルエーテルから再結晶して融点 214- 216 の暗赤色結晶を得た。 IR ( Br) リ: 3167, 2957, 2876, 1742, 1655, 1613, 1590, 1549 cm"1. N- {1,6-dioxo-8-methyl-4-[(5,5,6,6,6-pentafluorohexyl) sulfani L] -1,2,6,7,8,9-Hexahydro-3H-imidazo [5, lc] [1,4] benzothiazine-4-y Example 3 was prepared in the same manner as in Example 4-563. N- (1,6_dioxo-8-methyl-4-sulfanyl-1,2,6,7,8,9-hexatide obtained imidazo [5, tric] [1,4] The title compound (4.55 g, 93%) was synthesized from [benzothiazine-4-ylidene) -4-methylbenzenesulfonamide (3.51 g, 8.06 mmol). Recrystallization from ethyl acetate tetrahydrofuran-getyl ether gave dark red crystals with a melting point of 214-216. IR (Br) Li: 3167, 2957, 2876, 1742, 1655, 1613, 1590, 1549 cm " 1 .
¾-NMR (CDC13) : άΐ.14 (3Η, d, J-6.2 Hz), 1.62-1.88 (4H, m), 1.88-2.30 (4H, m), 2.42 (3H, s), 2.80-2.96 (2H, m), 3.06 (2H, i, J=6.8 Hz), 3.57 (1H, dd, J=18.8, 3.0 Hz), 7.30 (2H, d, J=8.0 Hz), 7.83 (2H, d, J=8.4 Hz), 9.86 (1H, bs). ' 元素分析値: C24H24N304S3F5として ¾-NMR (CDC1 3): άΐ.14 (3Η, d, J-6.2 Hz), 1.62-1.88 (4H, m), 1.88-2.30 (4H, m), 2.42 (3H, s), 2.80-2.96 (2H, m), 3.06 (2H, i, J = 6.8 Hz), 3.57 (1H, dd, J = 18.8, 3.0 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.86 ( 1H, bs) ' elemental analysis:. as C 24 H 24 N 3 0 4 S 3 F 5
計算値 (¾) : C, 47.28; H, 3.97; N, 6.89; S, 15.78. Calculated (¾): C, 47.28; H, 3.97; N, 6.89; S, 15.78.
実測値 (¾) : C, 47.58; H, 4.01; N, 6.98; S, 15.99. Found (値): C, 47.58; H, 4.01; N, 6.98; S, 15.99.
実施例 4- 566 Example 4-566
ェチル 5- [(1,6-ジォキソ- 3-{ [(4-メチルフエニル)スルホニル]イミ ノト 2, 3, 6, 7, 8, 9_へキサヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン _4 -ィ ル)スルファニル]ペン夕ノエー卜 Ethyl 5-[(1,6-dioxo-3-{[(4-methylphenyl) sulfonyl] iminoto 2,3,6,7,8,9_hexahydro-1H-imidazo [5,1-c] [ 1, 4] Benzothiazine _4 -yl) sulfanyl] pennoate
実施例 3- 38で得られた N- (1,6-ジォキソ -4-スルファニル -1, 2, 6, 7, 8, 9-へキサ ヒドロ- 3H-イミダゾ [5,卜 c] [1, 4]ベンゾチアジン- 4-ィリデン)-4-メチルベンゼ ンスルホンアミド (600 mg, 1.42 mmol) と炭酸カリウム (295 mg, 0.2.14膽 ol) の N, N -ジメチルホルムアミド(12.0 ml)の懸濁液に、 5 -プロモ吉草酸ェチル (0.276 ml, 1.71龍 ol) を加えた。 反応混合物を 80 で 20分間加熱した後、 1N塩酸を加 えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留 去して題記化合物 (521 mg, 67%) を暗赤色結晶として得た。 酢酸ェチルから再結 晶して融点 190-192°Cの暗赤色結晶を得た。 _ N- (1,6-dioxo-4-sulfanyl-1,2,6,7,8,9-hexahydro-3H-imidazo obtained in Example 3-38 [5, tric] [1, 4] Benzothiazine-4-ylidene) -4-methylbenzenesulfonamide (600 mg, 1.42 mmol) and potassium carbonate (295 mg, 0.2.14 phenol) in N, N-dimethylformamide (12.0 ml) To the solution was added 5-ethyl ethyl valerate (0.276 ml, 1.71 r. Ol). After heating the reaction mixture at 80 for 20 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was distilled off under reduced pressure to give the title compound (521 mg, 67%) as a dark red crystals. Recrystallization from ethyl acetate gave dark red crystals with a melting point of 190-192 ° C. _
赤外吸収スぺクトル(IR) (KBr中) V: 3177, 3065, 2926, 1740, 1657,1607, 1584, 1545 cm1. ¾-NMR (CDCI3) : δΐ.27 (3H, t, J=7.2 Hz), 1.72-1.82 (4H, m), 2.00-2.10 (2H, m), 2.30-2.39 (2H, m), 2.43 (3H, s), 2.51 (2H, t, J=6.9 Hz), 3.00-3.09 (2H, m), 3.21 (2H, t, J-6.0 Hz), 4.15 (2H, q, J=7.2 Hz), 7.31 (2H, d, 1=7.8 Hz), 7.84 (2H, d, J=8.1 Hz), 9.86 (1H, bs). Infrared absorption spectrum (IR) (in KBr) V: 3177, 3065, 2926, 1740, 1657,1607, 1584, 1545 cm 1 . ¾-NMR (CDCI3): δΐ.27 (3H, t, J = 7.2 Hz), 1.72-1.82 (4H, m), 2.00-2.10 (2H, m), 2.30-2.39 (2H, m), 2.43 ( 3H, s), 2.51 (2H, t, J = 6.9 Hz), 3.00-3.09 (2H, m), 3.21 (2H, t, J-6.0 Hz), 4.15 (2H, q, J = 7.2 Hz), 7.31 (2H, d, 1 = 7.8 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.86 (1H, bs).
元素分析値: C24H27N306S3として Elementary analysis: as C 24 H 27 N 3 0 6 S 3
計算値 (¾) : C, 52.44; H, 4.95; N, 7.64; S, 17.50. Calculated (¾): C, 52.44; H, 4.95; N, 7.64; S, 17.50.
実測値 (%) : C, 52.34; H, 4.81; N, 7.794; S, 17.41. Found (%): C, 52.34; H, 4.81; N, 7.794; S, 17.41.
実施例 4- 567 Example 4-567
5-[(1, 6 -ジォキソ -3- {[(4-メチルフエニル)スルホニル]ィミノト 2, 3, 6, 7, 8, 9-へ キサヒドロ- 1H-イミダゾ [5,卜 c] [1,4]ベンゾチアジン- 4-ィル)スルファニル] - N- へキシルペンタンアミド 5-[(1,6-dioxo-3-{[(4-methylphenyl) sulfonyl] iminoto 2,3,6,7,8,9-hexahydro-1H-imidazo [5, tric] [1,4 [Benzothiazine-4-yl) sulfanyl] -N-hexylpentanamide
実施例 4- 566と同様にして合成し、酢酸ェチルから再結晶して融点 112- 114°Cの暗 赤色結晶を得た。 The compound was synthesized in the same manner as in Example 4-566, and recrystallized from ethyl acetate to obtain dark red crystals having a melting point of 112 to 114 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3277, 2930, 2859, 1748, 1645, 1595, 1557, 1522 cm— Infrared absorption spectrum (IR) (in KBr) V: 3277, 2930, 2859, 1748, 1645, 1595, 1557, 1522 cm—
Ή-NMR (CDC13) : 50.800-0.920 (3H, m), 1.10-1.90 (14H, m), 1.98-2.14 (2H, m), 2.14-2.28 (2H, m), 2.43 (3H, s), 2.40-2.56 (2H, m), 2.96-3.08 (2H, m), 3.14-3.30 (2H, m), 5.52 (1H, bs), 7.30 (2H, d, J=8.0 Hz), 7.83 (2H, d, J=8. Hz), 9.86 (1H, bs). Ή-NMR (CDC1 3): 50.800-0.920 (3H, m), 1.10-1.90 (14H, m), 1.98-2.14 (2H, m), 2.14-2.28 (2H, m), 2.43 (3H, s) , 2.40-2.56 (2H, m), 2.96-3.08 (2H, m), 3.14-3.30 (2H, m), 5.52 (1H, bs), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H , D, J = 8. Hz), 9.86 (1H, bs).
実施例 4- 568 Example 4-568
N-{1-ォキソ - 4-[(5, 5, 6, 6, 6 -ペン夕フルォロペンチル)スルファニル ]_1, 2 -ジヒ ドロ- 3H-イミダゾ [5, 1-c] [1, 4]ベンゾチアジン- 3-ィリデン卜 4-メチルベンゼン スルホンアミド N- {1-oxo- 4-[(5,5,6,6,6-pentafluorofluoropentyl) sulfanyl] _1,2-dihydro-3H-imidazo [5,1-c] [1,4] benzothiazine -3-ylidene 4-methylbenzene sulfonamide
実施例 3-37で得られた Ν'-α-ォキソ -4-スルファニル -1, 2-ジヒドロ - 3Η-ィミダゾ [5,卜 c] [1,4]ベンゾチアジン- 3-イリデン) -4-メチルベンゼンスルホンアミド (150 mg, 0.372 mmol) と炭酸カリウム (77.1 mg, 0.558匪 ol) の Ν,Ν-ジメチ ルホルムアミド (30.0 ml) の懸濁液に、 参考例 9 で得られたメタンスルホン酸 5, 5, 6, 6, 6-ペン夕フルォロペンチルエステル (121 mg, 0.446丽 ol) の N,N -ジメ チルホルムアミド (1.00 ml) 溶液を加えた。 反応混合物を 80°Cで 1時間加熱し た後、 IN塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥(MgS04)後、 溶媒を減圧下に留去した。残留物をシリカゲル力ラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (1.49g, 89 ) を暗 赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 189- 191°Cの暗赤色結晶を得た。 Ν'-α-oxo-4-sulfanyl-1,2-dihydro-3Η-imidazo [5, tric] [1,4] benzothiazine-3-ylidene) -4-methyl obtained in Example 3-37 To a suspension of benzene, sulfonamide (150 mg, 0.372 mmol) and potassium carbonate (77.1 mg, 0.558 marl) in Ν, Ν-dimethylformamide (30.0 ml) was added methanesulfonic acid 5 obtained in Reference Example 9. , 5,6,6,6-Pentate fluoropentyl ester (121 mg, 0.446 mol) in N, N-dimethylformamide (1.00 ml) was added. Heat the reaction mixture at 80 ° C for 1 hour After that, IN hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (1.49 g, 89) as dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 189-191 ° C.
IR (KBr) V: 3216, 3063, 2934, 1728, 1630, 1588, 1557, 1485 cm"1. IR (KBr) V: 3216, 3063, 2934, 1728, 1630, 1588, 1557, 1485 cm " 1 .
Ή-NMR (CDC13) : 51.64-1.90 (4H, m), 1.90-2.24 (2H, in), 2.42 (3H, s), 3.04 (2H, t, J=6.8 Hz), 6.95 (1H, dd, J=7.8, 1.6 Hz), 7.08 (1H, dt, J-7.4, 1.2 Hz), 7.22 (1H, dt, J=8.4, 1.8 Hz), 7.30 (2H, d, J=8.2 Hz), 7.86 (2H, d, J=8.4 Hz), 8.46 (1H, dd, J=8.2, 1.2 Hz), 9.79 (1H, bs). Ή-NMR (CDC1 3): 51.64-1.90 (4H, m), 1.90-2.24 (2H, in), 2.42 (3H, s), 3.04 (2H, t, J = 6.8 Hz), 6.95 (1H, dd , J = 7.8, 1.6 Hz), 7.08 (1H, dt, J-7.4, 1.2 Hz), 7.22 (1H, dt, J = 8.4, 1.8 Hz), 7.30 (2H, d, J = 8.2 Hz), 7.86 (2H, d, J = 8.4 Hz), 8.46 (1H, dd, J = 8.2, 1.2 Hz), 9.79 (1H, bs).
元素分析値: C23H2。N303S3F6として Elemental analysis: C 23 H 2. N 3 0 3 S 3 F 6
計算値 (¾) : C, 47.83; H, 3.49; N, 7.27; S, 16.65. Calculated (¾): C, 47.83; H, 3.49; N, 7.27; S, 16.65.
実測値 (%) : C, 47.91; H, 3.42; N, 7.30; S, 16.38. Found (%): C, 47.91; H, 3.42; N, 7.30; S, 16.38.
実施例 4- 569 Example 4- 569
4 ク口口- N- {2_ (2, 2-ジメチルぺンチル) -8 - [ (2, 2-ジメチルペンチル)スルファ二 ル]- 5, 6-ジメチル -3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン - 1- 実施例 7-64 における結晶母液および洗浄液を併せて得た粗物質をシリカゲル (10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:9-3:7) に付した。 酢酸 ェチルーへキサン(1: 9)の画分を濃縮乾固後メ夕ノールから結晶化して表題化合 物(66 mg, 0.110腿 ol)を濃赤色粉末として得た。 4 口 口 -N- {2_ (2,2-dimethylpentyl) -8-[(2,2-dimethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro -1H -Imidazo [5, 1-c] [1, 4] thiazine-1- Chromatography of silica gel (10 g) on crude material obtained by combining the crystal mother liquor and washing solution in Example 7-64 (to ethyl acetate) Xan, 1: 9-3: 7). The ethyl acetate-hexane (1: 9) fraction was concentrated to dryness and then crystallized from methanol to give the title compound (66 mg, 0.110 mol) as a dark red powder.
Ή-NMR (300MHz, CDC13) : δθ.76 (6Η, s), 0.87 (3H, t, J= 6.6 Hz), 0.91 (3H, m), 1.01 (6H, s), 1.01 (2H, t, J= 8.2 Hz), 1.15 (2H, m), 1.35 (4H, brs), 1.80 (3H, q, J= 1.2 Hz), 2.28 (3H, q, J= 1.2 Hz), 2.73 (2H, s), 4.17 (2H, s), 7.40 (2H, dt, J= 8.7, 1.8 Hz), 7.92 (2H, dt, J= 8.2, 1.8 Hz). Ή-NMR (300MHz, CDC1 3 ): δθ.76 (6Η, s), 0.87 (3H, t, J = 6.6 Hz), 0.91 (3H, m), 1.01 (6H, s), 1.01 (2H, t , J = 8.2 Hz), 1.15 (2H, m), 1.35 (4H, brs), 1.80 (3H, q, J = 1.2 Hz), 2.28 (3H, q, J = 1.2 Hz), 2.73 (2H, s ), 4.17 (2H, s), 7.40 (2H, dt, J = 8.7, 1.8 Hz), 7.92 (2H, dt, J = 8.2, 1.8 Hz).
実施例 4-570 Example 4-570
4-ブロモ - N-{2-(2,2-ジメチルペンチル) -8-[(2,2-ジメチルペンチル)スルファニ ル]- 5, 6-ジメチル _3 -ォキソ - 2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] , 4]チアジン - 1- 実施例 4-504における結晶母液および洗浄液を併せて得た粗物質をシリカゲル (10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:9-3:7) に付した。 酢酸 ェチルーへキサン(1: 9)の画分を濃縮乾固後メ夕ノールから結晶化して表題化合 物 (72 mg, 0.112 rnmol)を濃赤色粉末として得た。 4-bromo-N- {2- (2,2-dimethylpentyl) -8-[(2,2-dimethylpentyl) sulfanyl] -5,6-dimethyl_3-oxo-2,3-dihydro-1H- Imidazo [5, 1-c], 4] thiazine-1- The crude material obtained by combining the crystal mother liquor and the washing solution in Example 4-504 was subjected to silica gel (10 g) chromatography (ethyl acetate-hexane, 1: 9-3: 7). The ethyl acetate-hexane (1: 9) fraction was concentrated to dryness and crystallized from methanol to give the title compound (72 mg, 0.112 rnmol) as a dark red powder.
lH - NMR (300MHz, CDC13) : ά 0.76 (6Η, s), 0.87 (3H, t, J= 7.1 Hz), 0.91 (3H, m), 1.01 (6H, s), 1.01 (2H, m), 1.17 (2H, m), 1.35 (4H, m), 1.80 (3H, q, J= 0.6 Hz), 2.28 (3H, q, J= 0.9 Hz), 2.73 (2H, s), 4.16 (2H, s), 7.57 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.7 Hz). lH - NMR (300MHz, CDC1 3 ): ά 0.76 (6Η, s), 0.87 (3H, t, J = 7.1 Hz), 0.91 (3H, m), 1.01 (6H, s), 1.01 (2H, m) , 1.17 (2H, m), 1.35 (4H, m), 1.80 (3H, q, J = 0.6 Hz), 2.28 (3H, q, J = 0.9 Hz), 2.73 (2H, s), 4.16 (2H, s), 7.57 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.7 Hz).
実施例 4-571 Example 4-571
6_[(Z)-2-メトキシ- 2-ォキソェチリデン] -1- {[(4-メチルフエニル)スルホニル]ィ ミ ノ }_3-ォキソ -8 - [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニ ル]- 2, 3, 5, 6 -テトラヒドロ _ -イミダゾ [5, 1-c] [1, 4]チアジン- 5-カルボン酸メチ ル 6 _ [(Z) -2-Methoxy-2-oxoethylidene] -1-{[(4-methylphenyl) sulfonyl] imino} _3-oxo-8-[(5,5,6,6,6-penn Fluorohexyl) sulfanyl] -2,3,5,6-tetrahydro_-imidazo [5,1-c] [1,4] Methyl thiazine-5-carboxylate
実施例 3 - 41 で得られた粗製の化合物 (2.28 g)および炭酸カリウム (0.98 g, 7.07 rnmol) の DMF (30 ml) の懸濁液にヨウ化 5, 5, 6, 6, 6-ペンタフルォ口へキシ ル (純度 87 , 1.64 g, 4.71顧 ol)を 6(TCで滴下し、 1.5時間攪拌した。 反応液に 1N塩酸 (9.4 ml)を加え、 酢酸ェチル(120 ml)で抽出した。 得られた有機層を酸 性に調整した 7%および 15%の食塩水、 ついで飽和食塩水で洗浄後、 硫酸ナトリウ ムで乾燥、 濃縮乾固した。 残渣をシリカゲル (20 g)のクロマトグラフィー (酢酸 ェチルーへキサン、 3:7-4:6) に付し、酢酸ェチルーへキサン(3:7-4: 6) の黄色画 分を濃縮乾固して、 表題化合物 (0.81 g, 1.23腿 ol, 30.2»を黄橙色粉末として 得た。 酢酸ェチル-へキサン (4: 6) の橙色画分を濃縮乾固して、 粗製の表題化合 物(1.13 g, 1.72腿 ol, 2.2%, 異性体混合物)を橙色粉末として得た。 5,5,6,6,6-pentafluoroiodide was added to a suspension of the crude compound (2.28 g) obtained in Example 3-41 and potassium carbonate (0.98 g, 7.07 rnmol) in DMF (30 ml). Hexyl (purity: 87, 1.64 g, 4.71 mmol) was added dropwise to 6 (TC) and the mixture was stirred for 1.5 hours. 1N hydrochloric acid (9.4 ml) was added to the reaction solution, and the mixture was extracted with ethyl acetate (120 ml). The obtained organic layer was washed with a 7% and 15% saline solution adjusted to be acidic, then with a saturated saline solution, dried over sodium sulfate and concentrated to dryness, and the residue was subjected to chromatography on silica gel (20 g). The residue was subjected to ethyl acetate-hexane (3: 7-4: 6), and the yellow fraction of ethyl acetate-hexane (3: 7-4: 6) was concentrated to dryness to give the title compound (0.81 g, 1.23 mmol). , 30.2 »was obtained as a yellow-orange powder The orange fraction of ethyl acetate-hexane (4: 6) was concentrated to dryness to give the crude title compound (1.13 g, 1.72 mol, 2.2%, isomer) Mixture) orange It was obtained as an end.
Ή-NMR (300MHz, CDC13) : (51.67 (4H, m), 1.99 (2H, m), 2.43 (3H, s), 2.94 (1H, ), 2.99 (2H, q, J= 7.2 Hz), 3.77 (3H, s), 3.81 (3H, s), 5.52 (1H, s), 6.33 (1H, s), 7.32 (2H, d, J- 8.1 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.83 (1H, br). 元素分析値: C24H24N307S3F5として計算値: C, 43.83; H, 3.68; N, 6.39; S, 14.63; F, 14.44 (%) 、 実測値: C, 43.70; H, 3.62; N, 6.41; S, 14.57 (%) . Ή-NMR (300MHz, CDC1 3 ): (51.67 (4H, m), 1.99 (2H, m), 2.43 (3H, s), 2.94 (1H,), 2.99 (2H, q, J = 7.2 Hz), 3.77 (3H, s), 3.81 (3H, s), 5.52 (1H, s), 6.33 (1H, s), 7.32 (2H, d, J- 8.1 Hz), 7.87 (2H, d, J = 8.4 Hz) .), 9.83 (1H, br ) elemental analysis: C 24 H 24 N 3 0 7 S 3 F 5 calculated: C, 43.83; H, 3.68 ; N, 6.39; S, 14.63; F, 14.44 (% ), Found: C, 43.70; H, 3.62; N, 6.41; S, 14.57 (%).
実施例 4- 572 ' N - [5, 6-ジメチル _3-ォキソ -8- [ (5-ブロモぺンチル)スルファニル] -2, 3-ジヒドロ - 1H -イミダゾ [5, i-c] [1,4]チアジン-卜ィリデン ]_4-メチルベンゼンスルホンァ ミド Example 4-572 ' N- [5,6-dimethyl_3-oxo-8-[(5-bromopentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, ic] [1,4] thiazine-triylidene] _4 -Methylbenzenesulfonamide
参考例 3-4で得られた N- (5, 6-ジメチル- 3-ォキソ -8-スルファニル -2, 3-ジヒド口 - 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-イリデン) -4-メチルベンゼンスルホンアミ ド (300 mg, 0.786 匪 ol) と炭酸カリウム (163 mg, 1.18腿 ol) の N,N -ジメチル ホルムアミド (4.00ral)の懸濁液に、 1,5-ジブロモペンタン(0.168m 1 , 1.18mmol) を加えた。 反応混合物を 60°Cで 4時間加熱した後、 1N塩酸を加えて酢酸ェチルで抽 出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシ リカゲルカラムクロマトグラフィーに付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (234 mg, 56%) を暗赤色結晶として得た。 酢酸ェチル -ジェ チルェ一テルから再結晶して融点 158-160°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrido obtained in Reference Example 3-4-1H-imidazo [5,1-c] [1,4] thiazine- To a suspension of 1-ylidene) -4-methylbenzenesulfonamide (300 mg, 0.786 ol) and potassium carbonate (163 mg, 1.18 ol) in N, N-dimethylformamide (4.00ral), 5-Dibromopentane (0.168 ml, 1.18 mmol) was added. After heating the reaction mixture at 60 ° C. for 4 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (234 mg, 56%) as dark red crystals. Recrystallization from ethyl acetate-ethyl acetate gave dark red crystals with a melting point of 158-160 ° C.
IR (KBr)リ : 3250, 3044, 2926, 2857, 1728, 1661, 1615, 1557 cm—1. IR (KBr): 3250, 3044, 2926, 2857, 1728, 1661, 1615, 1557 cm— 1 .
¾-NMR (CDC13) : 51.47- 1.84 (4H, m), 1.79 (3H, d, J=1.0 Hz), 1.84- 1.96 (2H, m), 2.26 (3H, d, J=1.0 Hz), 2.42 (3H, s), 2.95 (2H, i, J=7.4 Hz), 3.41 (2H, t, J=6.6 Hz), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.62 (1H, bs). 元素分析値: C2。H24N303S3Brとして ¾-NMR (CDC1 3): 51.47- 1.84 (4H, m), 1.79 (3H, d, J = 1.0 Hz), 1.84- 1.96 (2H, m), 2.26 (3H, d, J = 1.0 Hz), 2.42 (3H, s), 2.95 (2H, i, J = 7.4 Hz), 3.41 (2H, t, J = 6.6 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H , bs) elemental analysis:. C 2. As H 24 N 3 0 3 S 3 Br
計算値 (¾) : C, 45.28; H, 4.56; N, 7.92; S, 18.13. Calculated value (¾): C, 45.28; H, 4.56; N, 7.92; S, 18.13.
実測値 (%) : C, 45.46; H, 4.48; N, 7.81; S, 18.12. Found (%): C, 45.46; H, 4.48; N, 7.81; S, 18.12.
実施例 4- 573 Example 4-573
N_[8- ({[1,4- cis- 4 -(ヒドロキシメチル)シクロへキシル]メチル }スルファ二 ル)-5, 6-ジメチル- 3 -ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5,1- c] [1, 4]チアジン- 1 - 実施例 3-4で得た化合物 (2.50 g, 6.55 mmol)、 参考例 36で得た 4-ョードメ チルシクロへキサン- 1 -メタノール(1.83 g, 7.20 mmol, cis/ trans= 92:8混合 物)および炭酸カリウム (1.36 g, 9.83 mmol) の DMF (40 ml) の懸濁液も室温で 4日間攪拌した。 反応液に 1N塩酸 (13. lml)を加え、 水 (20ml)で希釈し、 室温 で 2時間攪拌した。 析出物を濾取し、 メタノ一ルー水 (1:1)およびメタノールで 洗浄して得た赤紫色粉末(2.96 g)を THF-酢酸ェチルから結晶化して表題化合物 (2.34 g, 4.61 mmol, 70.4%, cis/ trans= 92:8混合物)を赤紫色粉末として得た。 'H-NMR (200MHz, CDC13) : 51.28 (1H, br), 1.43 (4H, m), 1.58 (4H, m), 1.66 (1H, m), 1.79 (3H, q, J= 0.9 Hz), 1.82 (1H, m), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 2.91 (2H, d, J 7.5 Hz), 3.55 (2H, d, J= 6.6 Hz), 7.26 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.7 Hz), 9.61 (1H, brs). N_ [8-({[1,4-cis-4- (hydroxymethyl) cyclohexyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [ 5,1-c] [1,4] thiazine-1- The compound (2.50 g, 6.55 mmol) obtained in Example 3-4, 4-odomethylcyclohexane-1-methanol (1.83 g) obtained in Reference Example 36. g, 7.20 mmol, cis / trans = 92: 8 mixture) and potassium carbonate (1.36 g, 9.83 mmol) in DMF (40 ml) were also stirred at room temperature for 4 days. 1N Hydrochloric acid (13.lml) was added to the reaction solution, diluted with water (20ml), and stirred at room temperature for 2 hours. The precipitate was collected by filtration, washed with methanolic water (1: 1) and methanol, and the red-purple powder (2.96 g) obtained was crystallized from THF-ethyl acetate to give the title compound. (2.34 g, 4.61 mmol, 70.4%, cis / trans = 92: 8 mixture) was obtained as a purplish red powder. 'H-NMR (200MHz, CDC1 3): 51.28 (1H, br), 1.43 (4H, m), 1.58 (4H, m), 1.66 (1H, m), 1.79 (3H, q, J = 0.9 Hz) , 1.82 (1H, m), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 2.91 (2H, d, J 7.5 Hz), 3.55 (2H, d, J = 6.6 Hz), 7.26 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.7 Hz), 9.61 (1H, brs).
元素分析値: C23 04S3 ·0.3Η20として計算値: C, 53.84; Η, 5.81; , 8.19; S, 18.75 (%) 、 実測値: C, 53.80; Η, 5.81; Ν, 8.27; S, 19.06 (%) . Elemental analysis: C 23 0 4 S 3 · 0.3Η 20 Calculated: C, 53.84; Η, 5.81;, 8.19; S, 18.75 (%), found: C, 53.80; Η, 5.81; Ν, 8.27; S, 19.06 (%).
融点: 170.5-172.0°C Melting point: 170.5-172.0 ° C
実施例 4-574 Example 4-574
N-[8-({[l,4-trans-4-(hドロキシメチル)シクロへキシル]メチル }スルファ二 ル) -5, 6-ジメチル -3-ォキソ -2,3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン - 1- 実施例 3-4で得た化合物(1.27 g, 3.32腿 ol)、 参考例 38で得た trans-4-ョ一 ドメチルシクロへキサン- 1_メタノール (0.92 g, 3.62 腿 ol)および炭酸カリウム (688 mg, 4.98 mmol) の DMF (20 ml) の懸濁液を室温で 15 分間、 ついで 40°C で 15時間攪拌した。 反応液に 1N塩酸 (6.64 ml)を加え、 水 (14 ml)およびメ夕ノ —ル (2ml)で希釈し、 室温で 30分間攪拌した。 析出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物 (1.53 g, 3.01 匪 ol, 90.8¾)を赤紫色粉末として得た。 N- [8-({[l, 4-trans-4- (hdroxymethyl) cyclohexyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1- The compound obtained in Example 3-4 (1.27 g, 3.32 mol), the trans-4-hydroxymethylcyclohexane obtained in Reference Example 38 A suspension of 1_ methanol (0.92 g, 3.62 pt) and potassium carbonate (688 mg, 4.98 mmol) in DMF (20 ml) was stirred at room temperature for 15 minutes, then at 40 ° C for 15 hours. The reaction mixture was diluted with 1N hydrochloric acid (6.64 ml), diluted with water (14 ml) and methanol (2 ml), and stirred at room temperature for 30 minutes. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (1.53 g, 3.01 ol, 90.8¾) as a red-purple powder.
-脆 (300MHz, CDC13) : 50.99 (4H, brt, J= 10.8 Hz), 1.31 (1H, brs), 1.45 (1H, m), 1.53 (1H, m), 1.79 (3H, q, J= 1.2 Hz), 1.84 (2H, in), 1.93 (2H, m), 2.26 (3H, q, J= 0.9 Hz), 2.41 (3H, s), 2.83 (2H, d, J= 6.9 Hz), 3.47 (2H, m), 7.26 (2H, dt, J= 7.6, 1.8 Hz), 7.84 (2H, dt, J= 7.6, 1.8 Hz), 9.61 (1H, brs) . - brittleness (300MHz, CDC1 3): 50.99 (4H, brt, J = 10.8 Hz), 1.31 (1H, brs), 1.45 (1H, m), 1.53 (1H, m), 1.79 (3H, q, J = 1.2 Hz), 1.84 (2H, in), 1.93 (2H, m), 2.26 (3H, q, J = 0.9 Hz), 2.41 (3H, s), 2.83 (2H, d, J = 6.9 Hz), 3.47 (2H, m), 7.26 (2H, dt, J = 7.6, 1.8 Hz), 7.84 (2H, dt, J = 7.6, 1.8 Hz), 9.61 (1H, brs).
元素分析値: C23H29N304S3として計算値: C, 54.41; H, 5.76; N, 8.28; S, 18.95 (%) 、 実測値: C, 54.19; H, 5.77; N, 8.40; S, 18.79 (%) . Elemental analysis: C 23 H 29 N 3 0 4 S 3 Calculated: C, 54.41; H, 5.76 ; N, 8.28; S, 18.95 (%), Found: C, 54.19; H, 5.77 ; N, 8.40; S, 18.79 (%).
実施例 4-575 Example 4-575
4 -メチル安息香酸 5- [(5, 6_ジメチル-卜 {[(4_メチルフエニル)スルホ二ル]イミ ノ }- 3-ォキソ _2, 3_ジヒドロ- 1H-ィミダゾ [5,l-c][l,4]チアジン- 8-ィル) スルファ 二ル]- 4, 4-ジメチルペンチル 4-Methylbenzoic acid 5-[(5,6_dimethyl-{{((4_methylphenyl) sulfonyl] imino} -3-oxo-2,3_dihydro-1H-imidazo [5, lc] [l , 4] thiazine-8-yl) sulfa Nil] -4,4-dimethylpentyl
実施例 3-4で得た化合物(639 mg, 1.67 mmol)、 参考例 39で得た p-トルィル酸 5 -ヒドロキシ- 4, 4-ジメチルペンチル(476 mg, 1.90龍 ol) およびト リフエニルホ スフイン(634 mg, 2.34 mmol)の THF (13 ml)の懸濁液に室温で攪拌しながら DEAD (0.389 ml, 2.34腿 ol)を滴下し、 反応液を室温で 2.5時間、 次いで 50°Cで 20 時 間攪拌した。反応液に酢酸ェチルを加えて希釈し、 0.1N塩酸、 5%および飽和の食 塩水で洗浄した後、脱水乾燥、濃縮して粗油状物を得た。 これをシリカゲル (20 g) のクロマトグラフィー (酢酸ェチルーへキサン、 2:8-7:3) に付し、 赤色画分を濃 縮後、 メダノールを加えた。 析出物を濾取し、 メタノールで洗浄後、 酢酸ェチル ージェチルエーテルから結晶化して表題化合物(588 mg, 0.958 mmol, 57.4%)を赤 紫色粉末として得た。 The compound obtained in Example 3-4 (639 mg, 1.67 mmol), 5-hydroxy-4,4-dimethylpentyl p-toluate obtained in Reference Example 39 (476 mg, 1.90 dragonol) and triphenylphosphine ( DEAD (0.389 ml, 2.34 mol) was added dropwise to a suspension of 634 mg (2.34 mmol) in THF (13 ml) with stirring at room temperature, and the reaction mixture was allowed to stand at room temperature for 2.5 hours and then at 50 ° C for 20 hours. While stirring. The reaction mixture was diluted with ethyl acetate, washed with 0.1N hydrochloric acid, 5% and saturated brine, dried and concentrated to obtain a crude oil. This was subjected to chromatography on silica gel (20 g) (ethyl acetate-hexane, 2: 8-7: 3), and after concentration of the red fraction, medanol was added. The precipitate was collected by filtration, washed with methanol, and crystallized from ethyl acetate-ethyl ether to give the title compound (588 mg, 0.958 mmol, 57.4%) as a red-purple powder.
^-NMR (300MHz, CDC13) : 51.03 (6H, s), 1.45 (2H, m), 1.72 (2H, m), 1.77 (3H, q, J= 0.9 Hz), 2.25 (3H, q, J= 0.9 Hz), 2.39 (3H, s), 2.40 (3H, s), 2.90 (2H, s), 4.26 (2H, t, J= 6.6 Hz), 7.22 (2H, d, J= 8.1 Hz), 7.25 (2H, d, J= 8.7 Hz), 7.85 (2H, d, J= 8.1 Hz), 7.91 (2H, d, J= 8.1 Hz), 9.56 (1H, brs). 実施例 4- 576 ^ -NMR (300MHz, CDC1 3) : 51.03 (6H, s), 1.45 (2H, m), 1.72 (2H, m), 1.77 (3H, q, J = 0.9 Hz), 2.25 (3H, q, J = 0.9 Hz), 2.39 (3H, s), 2.40 (3H, s), 2.90 (2H, s), 4.26 (2H, t, J = 6.6 Hz), 7.22 (2H, d, J = 8.1 Hz), 7.25 (2H, d, J = 8.7 Hz), 7.85 (2H, d, J = 8.1 Hz), 7.91 (2H, d, J = 8.1 Hz), 9.56 (1H, brs).
N - (5, 6-ジメチル -8- { [ (1-メチルシク口へキシル)メチル]スルファ二ル} -3-ォキソ -2, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) -4-メチルベンゼ ンスルホンアミド N- (5,6-dimethyl-8-{[(1-methylcyclohexyl) methyl] sulfanyl} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得た化合物(639 mg, 1.67 mmol)、 参考例 40で得た卜メチルシク 口へキサン -卜メタノール(純度 91.8%, 1.04 g, 7.45 mmol)およびトリフエニル ホスフィン(2.25 g, 8.3311111101)の^? (45 ml)の懸濁液に室温で攪拌しながら DEAD (1.38 ml, 8.33龍 ol)を滴下し、 反応液を室温で 2.5時間、 次いで 50°Cで 2.5日 間攪拌した。反応液に酢酸ェチル(150 ml)を加えて希釈し、 0.1 塩酸で洗浄した 後、 2¾炭酸ナトリウム(100 ml)と混和した。 析出物を濾取してメタノールで洗浄 後、 酢酸ェチルー THF (2:1, 200 ml)に溶解し、 0.1N塩酸おょぴ飽和食塩水で洗 浄した後、 乾燥、 濃縮した。 残渣にメタノールを加え、 析出物を濾取してメタノ ールで洗浄後、 乾燥して表題化合物 (1.00 g, 2.03醒01, 34.2%)を赤紫色粉末と して得た。 Ή-N R (200MHz, CDC13) : δΐ.00 (3H, s), 1.38 (4H, m), 1.45 (6H, m), 1.79 (3H, q, J= 0.6 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 2.92 (2H, s), 7.26 (2H, d, J= 7.6 Hz), 7.87 (2H, d, J= 8.0 Hz), 9.57 (1H, brs). The compound obtained in Example 3-4 (639 mg, 1.67 mmol), the trimethylcycline hexane-trimethanol obtained in Reference Example 40 (purity 91.8%, 1.04 g, 7.45 mmol) and triphenylphosphine (2.25 g, 8.3311111101) ) ^? DEAD (1.38 ml, 8.33 liters) was added dropwise to the suspension (45 ml) at room temperature with stirring, and the reaction solution was stirred at room temperature for 2.5 hours and then at 50 ° C for 2.5 days. The reaction mixture was diluted with ethyl acetate (150 ml), washed with 0.1 hydrochloric acid, and then mixed with sodium bicarbonate (100 ml). The precipitate was collected by filtration, washed with methanol, dissolved in ethyl acetate-THF (2: 1, 200 ml), washed with 0.1N hydrochloric acid and saturated saline, dried and concentrated. Methanol was added to the residue, the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (1.00 g, 2.03, 01, 34.2%) as a red-purple powder. Ή-NR (200MHz, CDC1 3 ): δΐ.00 (3H, s), 1.38 (4H, m), 1.45 (6H, m), 1.79 (3H, q, J = 0.6 Hz), 2.26 (3H, q , J = 1.2 Hz), 2.41 (3H, s), 2.92 (2H, s), 7.26 (2H, d, J = 7.6 Hz), 7.87 (2H, d, J = 8.0 Hz), 9.57 (1H, brs ).
元素分析値: C23H29N303S3として計算値: C, 56.18; H, 5.94; N, 8.55; S, 19.56. 実測値: C, 56.21; H, 5.95; N, 8.38; S, 19.54. Elemental analysis: C 23 H 29 N 3 0 3 S 3 Calculated:. C, 56.18; H, 5.94; N, 8.55; S, 19.56 Found: C, 56.21; H, 5.95 ; N, 8.38; S , 19.54.
融点: 180.0-181.0°C Melting point: 180.0-181.0 ° C
実施例 4- 577 Example 4-577
N - {8- [(5-フルォロ- 2, 2-ジメチルペンチル) スルファニル] -5, 6-ジメチル- 3-ォキ ソ- 2 , 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -イリデン } -4-メチルベンゼ ンスルホンアミド N- {8-[(5-Fluoro-2,2-dimethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene} -4-methylbenzensulfonamide
実施例 3-4で得た化合物 (1.71 g, 4.48龍 ol)、 参考例 41で得た 5-フルォ ロ- 2, 2-ジメチルペン夕ノール(1.40 g, 1:1 酢酸ェチル混合物) およびトリフエ ニルホスフィン(1.57 g, 5.82 mmol)の THF (35 ml)の懸濁液に室温で攪拌しなが ら DEAD (0.965 ml, 5.82匪 ol)を滴下し、 反応液を室温で 11時間、 次いで 50°C で 26 時間攪捽した。 反応液を濃縮後メタノールを加えた。 析出物を濾取し、 メ 夕ノールで洗浄後、 酢酸- THF から結晶化して表題化合物(874 mg, 1.76 mmol)を 赤色粉末として得た。 The compound obtained in Example 3-4 (1.71 g, 4.48 dragonol), 5-fluoro-2,2-dimethylpentanol obtained in Reference Example 41 (1.40 g, a 1: 1 mixture of ethyl acetate) and triphenyl To a suspension of nilphosphine (1.57 g, 5.82 mmol) in THF (35 ml) was added dropwise DEAD (0.965 ml, 5.82 bandol) while stirring at room temperature, and the reaction was allowed to stand at room temperature for 11 hours, Stirred at ° C for 26 hours. After concentrating the reaction solution, methanol was added. The precipitate was collected by filtration, washed with methanol, and crystallized from acetic acid-THF to give the title compound (874 mg, 1.76 mmol) as a red powder.
Ή-NMR (300MHz, CDC13): δΐ.01 (6Η, s), 1.41 (2H, m), 1.68 (2H, m), 1.78 (3H, s), 2.26 (3H, s), 2.41 (3H, s), 2.89 (2H, s), 4.41 (2H, dt, J= 47.4, 5.9 Hz), 7.27 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.58 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): δΐ.01 (6Η, s), 1.41 (2H, m), 1.68 (2H, m), 1.78 (3H, s), 2.26 (3H, s), 2.41 (3H , s), 2.89 (2H, s), 4.41 (2H, dt, J = 47.4, 5.9 Hz), 7.27 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.58 (1H, brs).
元素分析値: C22H28N303S3F 0.4H20として計算値: C, 52.34; H, 5.75; , 8.32; S, 19.05(%). 実測値: C, 52.57; H, 5.68; N, 8.29; S, 18.76 ). Elemental analysis: C 22 H 28 N 3 0 3 S 3 F 0.4H 2 0 Calculated:. C, 52.34; H, 5.75;, 8.32; S, 19.05 (%) Found: C, 52.57; H, 5.68; N, 8.29; S, 18.76).
融点: 179.0 180: 5 : Melting point: 179.0 180: 5:
実施例 4-578 Example 4-578
4-ク口ロ- N- {8- [ (2, 2-ジメチルぺンチル)スルファニル] -5, 6 -ジメチル -3-ォキソ - 2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, ]チアジン- 1-ィリデン }ベンゼンスルホ ンアミド 4-N- {8-[(2,2-Dimethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydric-1H-imidazo [5,1-c ] [1,] thiazine-1-ylidene} benzenesulfonamide
実施例 3- 6で得た化合物(427 mg, 1.06卿 ol)、 2, 2-ジメチルペンタノール (153 mg, 1.17 mmol) およびトリフエニルホスフィン(373 mg, 1.38 ranol)の THF (8.5 ml)懸濁液に室温で攪拌しながら DEAD (0.229 ml, 1.38讓 ol)を滴下し、 反応液 を 50°C で 35時間攪拌した。反応液に酢酸ェチル (35 ml)を加えて希釈し、 0. IN塩 酸、 水および飽和食塩水で洗浄し、 乾燥、 濃縮後、 メタノールで希釈した。 析出 物を濾取し、 メタノール洗浄後、 酢酸ェチルおよびジェチルェ一テルから結晶化 して表題化合物 (224 n)g, 0.448腳 ol)を紫色粉末として得た。 The compound obtained in Example 3-6 (427 mg, 1.06 lol), 2,2-dimethylpentanol (153 mg, 1.17 mmol) and triphenylphosphine (373 mg, 1.38 ranol) in THF (8.5 DEAD (0.229 ml, 1.38 mL) was added dropwise to the suspension at room temperature while stirring at room temperature, and the reaction solution was stirred at 50 ° C for 35 hours. The reaction mixture was diluted with ethyl acetate (35 ml), washed with 0.1 IN hydrochloric acid, water and saturated saline, dried, concentrated and diluted with methanol. The precipitate was collected by filtration, washed with methanol, and crystallized from ethyl acetate and diethyl ether to give the title compound (224 n) g, 0.448 mmol) as a purple powder.
Ή-NMR (300MHz, CDC13): (50.92 (3H, m), 0.98 (6H, s), 1.27 (4H, brd, J= 3.6 Hz), 1.79 (3H, q, J= 0.9 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.89 (2H, s), 7.44 (2H, dt, J= 8.7, 1.8 Hz), 7.87 (2H, dt, J= 8.7, 2.1 Hz), 9.56 (1H, brs). 元素分析値: C21H26N303S3C1として計算値: C, 50.44; H, 5.24; N, 8.40; S, 19.24; C1, 7.09 ( ) 、 実測値 C, 50.43; H, 5.18; N, 8.38; S, 19.20; C1, 7.12 ( ) . mp. 185.5-186.5 Ή-NMR (300MHz, CDC1 3 ): (50.92 (3H, m), 0.98 (6H, s), 1.27 (4H, brd, J = 3.6 Hz), 1.79 (3H, q, J = 0.9 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.89 (2H, s), 7.44 (2H, dt, J = 8.7, 1.8 Hz), 7.87 (2H, dt, J = 8.7, 2.1 Hz), 9.56 (1H, brs). Elemental analysis: Calculated for C 21 H 26 N 3 0 3 S 3 C1: C, 50.44; H, 5.24; N, 8.40; S, 19.24; C1, 7.09 (), found C, 50.43; H, 5.18; N, 8.38; S, 19.20; C1, 7.12 () .mp.185.5-186.5
実施例 4-579 Example 4-579
N-(5, 6 -ジメチル- 3-ォキソ -8 - {[2 -(2, 2, 2 -トリフルォロエトキシ)エヂル]スルフ ァニルト 2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン) -4-メチ ルベンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[2- (2,2,2-trifluoroethoxy) edyl] sulfanylto 2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-triylidene) -4-methylbenzenesulfonamide
実施例 3- 4で得られた N - (5, 6 -ジメチル- 3 -ォキソ -8-スルファニル -2, 3 -ジヒド ロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン) -4-メチルベンゼンスルホン アミド (200 mg, 0.524讓 ol) と炭酸カリウム (109 mg, 0.786 mmol) の Ν,Ν-ジ メチルホルムアミド (3.00 ml) の懸濁液に、 参考例 71で得られたメタンスルホ ン酸 2 -(2, 2,2-トリフルォロエトキシ)ェチルエステル (140 mg, 0.629匪 ol) の N,N-ジメチルホルムアミド (l.OOml) 溶液を加えた。 反応混合物を 8(TCで 30分 間加熱した後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (87.9 mg, 33¾) を暗赤色結晶として得 た。酢酸ェチル-ジェチルエーテルから再結晶して融点 167- 168°Cの暗赤色結晶を 得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 To a suspension of 1-ylidene) -4-methylbenzenesulfonamide (200 mg, 0.524 acetyl) and potassium carbonate (109 mg, 0.786 mmol) in Ν, Ν-dimethylformamide (3.00 ml) was added Reference Example 71. A solution of 2- (2,2,2-trifluoroethoxy) ethyl methanesulfonate (140 mg, 0.629 ol) in N, N-dimethylformamide (l.OOml) obtained in the above was added. After heating for 30 minutes at the reaction mixture 8 (TC, and extracted with acetic acid Echiru addition of 1N hydrochloric acid. The extract was washed with water, dried (MgSO 4) After the title compound was evaporated under reduced pressure (87.9 mg, 33¾) as dark red crystals, which were recrystallized from ethyl acetate-ethyl ether to give dark red crystals having a melting point of 167-168 ° C.
IR (KBr) V 3243, 2932, 1755, 1742, 1665, 1624, 1561 cm"1. IR (KBr) V 3243, 2932, 1755, 1742, 1665, 1624, 1561 cm " 1 .
Ή-NMR (CDC13) : δ 1.79 (3Η, d, J=1.2 Hz), 2.26 (3H, d, J=0.9 Hz), 2.42 (3H, s), 3.17 (2H, t, J=6.6 Hz), 3.83 (2H, t, J=8.4 Hz), 3.84 (2H, q, J=8.7 Hz), 7.29 (2H, d, J=8.1 Hz), 7.84 (2H, d, J=8. Hz), 9.63 (1H, bs). 元素分析値: C19¾。N304S3F3として Ή-NMR (CDC1 3): δ 1.79 (3Η, d, J = 1.2 Hz), 2.26 (3H, d, J = 0.9 Hz), 2.42 (3H, s), 3.17 (2H, t, J = 6.6 Hz ), 3.83 (2H, t, J = 8.4 Hz), 3.84 (2H, q, J = 8.7 Hz), 7.29 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.Hz) , 9.63 (1H, bs). Elemental analysis: C 19 ¾. N 3 0 4 S 3 F 3
計算値 (%) : C, 44.96; H, 3.97; N, 8.28; S, 18.95. Calculated (%): C, 44.96; H, 3.97; N, 8.28; S, 18.95.
実測値 (%) : C, 45.37; H, 3.99; N, 8.41; S, 19.32. Found (%): C, 45.37; H, 3.99; N, 8.41; S, 19.32.
実施例 4-580 Example 4-580
N- (5, 6-ジメチル- 3-ォキソ -8- { [2- (2-フルォロェトキシ)ェチル]スルファニ ル}-2,3_ジヒドロ -1H-イミダゾ [5,1 - c] [1,4]チアジン - 1-ィリデン)-4-メチルベ ンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[2- (2-fluoroethoxy) ethyl] sulfanyl} -2,3_dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 3-4で得られた N-(5, 6-ジメチル -3-ォキソ -8-スルファニル -2, 3-ジヒド ロ- 1H-イミダゾ [5, c] [1,4]チアジン- 1-ィリデン)-4-メチルベンゼンスルホン アミド (1.50 g, 3.93腿01) と炭酸カリウム (815 mg, 5.90 mmol) の N,N -ジメ チルホルムアミド (15.0 ml) の懸濁液に、 参考例 85で得られたメタンスルホン 酸 2- (2-フルォロエトキシ)ェチルエステル (879 mg, 4.72 mmol) の Ν,Ν-ジメチ ルホルムアミド (5.00 ml) 溶液を加えた。 反応混合物を 80 で 2時間加熱した 後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。残留物をシリカゲルカラムクロマトグラフィーに付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (34.7 mg, 10%) を 暗赤色結晶として得た。 酢酸ェチル-ジェチルェ一テルから再結晶して融点 156- 158°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydro-1H-imidazo [5, c] [1,4] thiazine-1- obtained in Example 3-4 A suspension of N, N-dimethylformamide (15.0 ml) of (ylidene) -4-methylbenzenesulfonamide (1.50 g, 3.93 t01) and potassium carbonate (815 mg, 5.90 mmol) was obtained in Reference Example 85. A solution of the obtained methanesulfonic acid 2- (2-fluoroethoxy) ethyl ester (879 mg, 4.72 mmol) in Ν, Ν-dimethylformamide (5.00 ml) was added. After the reaction mixture was heated at 80 for 2 hours, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (34.7 mg, 10%) as dark red crystals. Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 156-158 ° C.
IR (KBr) V: 3241, 2951, 1742, 1663, 1620, 1561 cm"1. IR (KBr) V: 3241, 2951, 1742, 1663, 1620, 1561 cm " 1 .
Ή-N R (CDC13): δ 1.79 (3Η, d, J=1.0 Hz), 2.26 (3H, d, J=1.2 Hz), 2.42 (3H, s), 3.17 (2H, t, J=6.6Hz), 3.72 (2H, dt, J=29.8, 4.0 Hz) , 3.74 (2H, t, J=6.6 Ή-NR (CDC1 3): δ 1.79 (3Η, d, J = 1.0 Hz), 2.26 (3H, d, J = 1.2 Hz), 2.42 (3H, s), 3.17 (2H, t, J = 6.6Hz ), 3.72 (2H, dt, J = 29.8, 4.0 Hz), 3.74 (2H, t, J = 6.6
Hz), 4.56 (2H, dt, J=47.6, 4. Hz), 7.29 (2H, d, J=8.0Hz), 7.85 (2H, d, J=8.Hz), 4.56 (2H, dt, J = 47.6, 4.Hz), 7.29 (2H, d, J = 8.0Hz), 7.85 (2H, d, J = 8.
Hz), 9.59 (1H, bs). Hz), 9.59 (1H, bs).
元素分析値: C19H22N304S3Fとして Elementary analysis: as C 19 H 22 N 3 0 4 S 3 F
計算値 (%) : C, 48.39; H, 4.70; N, 8.91; S, 20.40. Calculated (%): C, 48.39; H, 4.70; N, 8.91; S, 20.40.
実測値 (%) : C, 48.48; H, 4.79; N, 8.88; S, 20.40. 実施例 5-1 Found (%): C, 48.48; H, 4.79; N, 8.88; S, 20.40. Example 5-1
N- {2,3-ジヒドロ- 5, 6-ジメチル -3-ォキソ -8- (4-トルイジノ) -1H-ィミダゾ [5, 1-c] [1, 4]チアジン - 1_イリデン }-4-メチルベンゼンスルホンアミド N- {2,3-dihydro-5,6-dimethyl-3-oxo-8- (4-toluidino) -1H-imidazo [5,1-c] [1,4] thiazine-1_ylidene} -4 -Methylbenzenesulfonamide
実施例 -24で得られた N - [2, 3 -ジヒドロ- 5, 6 -ジメチル -8- (ペンチルスルファ ニル) -3-ォキソ -1H-イミダゾ [5,1- c] [1, 4]チアジン -1-ィリデン ]-4-メチルベン ゼンスルホンアミド (200 mg, 0.443腿 ol) と p-トルイジン (102 mg, 0.930 mmol) の混合物を 120^で 3時間かき混ぜた。 反応混合物に水を加えて酢酸ェチルで抽 出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシ リカゲルカラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル(10:1, v/v) で溶出して題記化合物 (74.2mg, 37¾) を橙色結晶として得た。 酢酸ェチル -ジェ チルエーテルから再結晶して融点 237-239T:の橙色結晶を得た。 N- [2,3-dihydro-5,6-dimethyl-8- (pentylsulfanyl) -3-oxo-1H-imidazo [5,1-c] [1,4] obtained in Example-24 A mixture of] thiazine-1-ylidene] -4-methylbenzenesulfonamide (200 mg, 0.443 tmol) and p-toluidine (102 mg, 0.930 mmol) was stirred with 120 ^ for 3 hours. Water was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (74.2 mg, 37 °) as orange crystals. Recrystallization from ethyl acetate-ethyl ether gave orange crystals of melting point 237-239T :.
赤外吸収スペクトル(IR) (KBr中) V: 3164, 3031, 2924, 1717, 1653, 1607, 1588, 1557, 1514, 1485 cm"1. , Infrared absorption spectrum (IR) (in KBr) V: 3164, 3031, 2924, 1717, 1653, 1607, 1588, 1557, 1514, 1485 cm " 1 .
•H-NMR (CDC13) : δ 1.77 (3Η, d, J=0.6 Hz), 2.37 (6H, s), 2.41 (3H, s), 7.05 (2H, d, J=8.6Hz), 7.19 (2H, d, J=8.2 Hz), 7.28 (2H, d, J=8.0 Hz), 7.80 (2H, d, J=8.2 Hz), 9.58 (1H, bs), 10.45 (1H, bs). • H-NMR (CDC1 3) : δ 1.77 (3Η, d, J = 0.6 Hz), 2.37 (6H, s), 2.41 (3H, s), 7.05 (2H, d, J = 8.6Hz), 7.19 ( 2H, d, J = 8.2 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.80 (2H, d, J = 8.2 Hz), 9.58 (1H, bs), 10.45 (1H, bs).
元素分析値: C22H22N403S2 · 0.2H20として Elemental analysis: C 22 H 22 N 4 0 3 S 2 · 0.2 H 2 0
計算値 (%) : C, 57.67; H, 4.93; N, 12.23; S, 14.00. Calculated value (%): C, 57.67; H, 4.93; N, 12.23; S, 14.00.
実測値 (%) : C, 57.62; H, 4.90; N, 12.02; S, 14.24. Found (%): C, 57.62; H, 4.90; N, 12.02; S, 14.24.
実施例 5-2 Example 5-2
N- {5, 6 -ジメチル- 8- [メチル(2-フエニルェチル)ァミノ] - 3 -ォキソ -2, 3_ジヒドロ -1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }-4-メチルベンゼンスルホンァ ミド N- {5,6-Dimethyl-8- [methyl (2-phenylethyl) amino] -3-oxo-2,3_dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene} -4-methylbenzenesulfonamide
実施例 -38で得られた N- [8- [(5 -クロ口ペンチル)スルファニル ]-5, 6 -ジメチ ル -3-ォキソ - 2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデント 4- メチルベンゼンスルホンアミド (200 mg, 0.411 nimo'l) と N-メチルフエネチルァ ミン (0.179 ml, 1.23 mmol) のテトラヒドロフラン (3.00 ml) 溶液を 16時間加 熱環流した。 反応混合物に水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾 燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグ ラフィ一に付し、 n-へキサン-酢酸ェチル(10:1, v/v)で溶出して題記化合物(7.2 mg, 3.9¾) を橙色不定形晶として得た。 N- [8-[(5-Phenylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrido-1H-imidazo [5,1- c] A solution of [1,4] thiazine-1-ylident 4-methylbenzenesulfonamide (200 mg, 0.411 nimo'l) and N-methylphenethylamine (0.179 ml, 1.23 mmol) in tetrahydrofuran (3.00 ml) was prepared. Heated reflux for 16 hours. Water was added to the reaction mixture, which was extracted with ethyl acetate. The extract was washed with water, after Drying (MgSO 4), the solvent was distilled off under reduced pressure. The residue is purified by silica gel column chromatography. The title compound (7.2 mg, 3.9¾) was obtained as orange amorphous crystals by elution with n-hexane-ethyl acetate (10: 1, v / v).
赤外吸収スペクトル(IR) (KBr中) V: 3368, 3260, 3061, 3027, 2926, 1728, 1663, 1601, 1537, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3368, 3260, 3061, 3027, 2926, 1728, 1663, 1601, 1537, 1497 cm " 1 .
'H-NMR (CDC13) : δ 1.83 (3H, d, J=1.0 Hz), 2.26 (3H, d, J=0.6 Hz), 2.38 (3H, s), 2.82 (2H, t, J=7.0 Hz), 3.28 (3H, s), 3.7 (2H, t, J=7.4 Hz), 7.02-7.10 'H-NMR (CDC1 3) : δ 1.83 (3H, d, J = 1.0 Hz), 2.26 (3H, d, J = 0.6 Hz), 2.38 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 3.28 (3H, s), 3.7 (2H, t, J = 7.4 Hz), 7.02-7.10
(2H, m), 7.14-7.24 (5H, m), 7.79 (2H, d, J=8.0 Hz), 9.67 (1H, bs) . (2H, m), 7.14-7.24 (5H, m), 7.79 (2H, d, J = 8.0 Hz), 9.67 (1H, bs).
元素分析値: C24H26N403S2 · 0.6 0として Elemental analysis: as C 24 H 26 N 4 0 3 S 2 · 0.60
計算値 ( ) : C, 58.42; H, 5.56; N, 11.35; S, 13.00. Calculated (): C, 58.42; H, 5.56; N, 11.35; S, 13.00.
実測値 (%) : C, 58.06; H, 5.36; N, 11.11; S, 13.52. Found (%): C, 58.06; H, 5.36; N, 11.11; S, 13.52.
以下に実施例 5-1、 5- 2で合成した化合物および同様にして合成した化合物の一 覧を示す。 , The following is a list of the compounds synthesized in Examples 5-1 and 5-2 and the compounds synthesized in the same manner. ,
〔表 6 7〕(Table 67)
差替え用弒 ®則 26) 以下に実施例 5- 3〜5 - 8で合成した化合物のデータを示す。 Replacement rules 26) The data of the compounds synthesized in Examples 5-3 to 5-8 are shown below.
実施例 5-3 Example 5-3
酢酸ェチル -ジェチルエーテルで再結晶し、 橙色結晶を得た。 The crystals were recrystallized from ethyl acetate-getyl ether to give orange crystals.
融点 216-218^. ' 216-218 ^. '
赤外吸収スペクトル(IR) (KBr中) リ: 3358, 3235, 2921, 1732, 1661, 1605, 1539 cur1. Infrared absorption spectrum (IR) (in KBr): 3358, 3235, 2921, 1732, 1661, 1605, 1539 cur 1 .
Ή-NMR (CDC13) : δ 1.89 (3Η, d, J-1.0 Hz), 2.31 (3H, d, J=1.0 Hz), 2.40 (3H, s), 3.17 (6H, s), 7.26 (2H, d, J=8.2 Hz), 7.79 (2H, d, J=8.4 Hz), 9.69 (1H, bs). Ή-NMR (CDC1 3): δ 1.89 (3Η, d, J-1.0 Hz), 2.31 (3H, d, J = 1.0 Hz), 2.40 (3H, s), 3.17 (6H, s), 7.26 (2H , d, J = 8.2 Hz), 7.79 (2H, d, J = 8.4 Hz), 9.69 (1H, bs).
元素分析値: C17H2。N403S2として計算値: C, 52.02; H, 5.14; N, 14.27; S, 16.34 (%)、 実測値: C, 52.09; H, 5.12; N, 14.21; S, 16.60 (%) . Elemental analysis: C 17 H 2. Calculated for N 4 0 3 S 2 : C, 52.02; H, 5.14; N, 14.27; S, 16.34 (%), Found: C, 52.09; H, 5.12; N, 14.21; S, 16.60 (%) .
実施例 5-4 Example 5-4
赤外吸収スペクトル(IR) (KBr中) v 3353, 2930, 2859, 1728, 1661, 1607, 1537 cm"1. Infrared absorption spectrum (IR) (in KBr) v 3353, 2930, 2859, 1728, 1661, 1607, 1537 cm " 1 .
Ή-NMR (CDCI3) : δ 0.855 (3Η, i, J=6.6Hz), 1.10-1.34 (6H, m), 1.44-1.64 (2H, m), 1.90 (3H, s), 2.31 (3H, s), 2.39 (3H, s), 3.25 (3H, s), 3.39 (2H, t, J=7.2 Hz), 7.24 (2H, d, J=8.0 Hz), 7.77 (2H, d, J=8.2 Hz), 9.75 (1H, bs). Ή-NMR (CDCI3): δ 0.855 (3Η, i, J = 6.6Hz), 1.10-1.34 (6H, m), 1.44-1.64 (2H, m), 1.90 (3H, s), 2.31 (3H, s ), 2.39 (3H, s), 3.25 (3H, s), 3.39 (2H, t, J = 7.2 Hz), 7.24 (2H, d, J = 8.0 Hz), 7.77 (2H, d, J = 8.2 Hz) ), 9.75 (1H, bs).
実施例 5-5 Example 5-5
酢酸ェチル -ジェチルェ一テルで再結晶し、 橙色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain orange crystals.
融点 207-209°C. 207-209 ° C.
赤外吸収スペクトル(IR) (KBr中) : 3252, 2971, 2924, 2859, 1732, 1661, 1611, 1539, 1497 cm1. Infrared absorption spectrum (IR) (in KBr): 3252, 2971, 2924, 2859, 1732, 1661, 1611, 1539, 1497 cm 1 .
Ή-NMR (CDCI3) : (51.88 (3H, d, J=0.8 Hz), 2.31 (3H, d, J=1.0 Hz), 2.41 (3H, s), 3.37 (2H, t, J=4.8 Hz), 3.72 (2H, t, J=5.2 Hz), 7.29 (2H, d, J=8.0 Hz), 7.78 (2H, d, J=8.4 Hz), 9.67 (1H, bs). Ή-NMR (CDCI3): (51.88 (3H, d, J = 0.8 Hz), 2.31 (3H, d, J = 1.0 Hz), 2.41 (3H, s), 3.37 (2H, t, J = 4.8 Hz) , 3.72 (2H, t, J = 5.2 Hz), 7.29 (2H, d, J = 8.0 Hz), 7.78 (2H, d, J = 8.4 Hz), 9.67 (1H, bs).
元素分析値: C19H22N404S2'0.2 0として計算値: C, 52.08; H, 5.15; , 12.79; S, 14.64 (%), 実測値: C, 52.07; H, 5.23; N, 12.74; S, 14.84 (%) . Elemental analysis: C 19 H 22 N 4 0 4 S 2 '0.20 Calculated: C, 52.08; H, 5.15;, 12.79; S, 14.64 (%), found: C, 52.07; H, 5.23; N, 12.74; S, 14.84 (%).
実施例 5-6 Example 5-6
酢酸ェチル -ジェチルェ一テルで再結晶し、 橙色結晶を得た。 融点 21卜 213°C. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain orange crystals. Mp 21 ° C 213 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3287, 2944, 2880, 1728, 1655, 1607, 1534, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr): 3287, 2944, 2880, 1728, 1655, 1607, 1534, 1497 cm " 1 .
】H-腿 (CDC13) : δ 1.58-1.76 (6Η, m), 1.89 (3H, d, J=1.2 Hz), 2.31 (3H, d, J=1.2 Hz), 2.40 (3H, s), 3.35-3.44 (4H, m), 7.25 (2H, d, J=8.4Hz), 7.78 (2H, d, J=8.4 Hz), 9.66 (1H, bs). ] H- thigh (CDC1 3): δ 1.58-1.76 ( 6Η, m), 1.89 (3H, d, J = 1.2 Hz), 2.31 (3H, d, J = 1.2 Hz), 2.40 (3H, s), 3.35-3.44 (4H, m), 7.25 (2H, d, J = 8.4Hz), 7.78 (2H, d, J = 8.4Hz), 9.66 (1H, bs).
元素分析値: C2。H24N403S2として計算値: C, 55.53; H, 5.59; N, 12.95; S, 14.83 (%)、 実測値: C, 55.14; H, 5.50; , 12.75; S, 14.57 ( ) . Elemental analysis: C 2. H 24 N 4 0 3 S 2 Calculated: C, 55.53; H, 5.59; N, 12.95; S, 14.83 (%), Found: C, 55.14; H, 5.50;, 12.75; S, 14.57 () .
実施例 5 - 7 Example 5-7
酢酸ェチル -ジェチルェ一テルで再結晶し、 橙色結晶を得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain orange crystals.
融点 192- 194°C. 192-194 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 2922, 1726, 1663, 1605, 1537, 1495 cm—1. Ή-NMR (CDC13) : δ 1.20-1.82 (5Η, m), 1.88 (3H, d, J=1.0 Hz), 2.30 (3H, d, J=0.8Hz), 2.39 (3H, s), 2.53 (2H, d, J=7.4 Hz), 2.98-3.18 (2H, m), 3.56-3.72 (2H, m), 7.12 (2H, d, J=6.6 Hz), 7.20-7.38 (5H, m), 7.77 (2H, d, J=8.4 Hz), 9.65 (1H, bs). Infrared absorption spectrum (IR) (in KBr) Re:. 2922, 1726, 1663, 1605, 1537, 1495 cm- 1 Ή-NMR (CDC1 3): δ 1.20-1.82 (5Η, m), 1.88 (3H, d, J = 1.0 Hz), 2.30 (3H, d, J = 0.8Hz), 2.39 (3H, s), 2.53 (2H, d, J = 7.4 Hz), 2.98-3.18 (2H, m), 3.56- 3.72 (2H, m), 7.12 (2H, d, J = 6.6 Hz), 7.20-7.38 (5H, m), 7.77 (2H, d, J = 8.4 Hz), 9.65 (1H, bs).
実施例 5-8 Example 5-8
酢酸ェチル -ジィソプロピルエーテルで再結晶し、 橙色結晶を得た。 The crystals were recrystallized from ethyl acetate-disopropyl ether to give orange crystals.
融点: 194- 196°C. Melting point: 194-196 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3362, 3027, 2926, 1732, 1663, 1601, 1539, 1495 cm— Infrared absorption spectrum (IR) (in KBr): 3362, 3027, 2926, 1732, 1663, 1601, 1539, 1495 cm—
Ή一 NMR (CDC13) : δ 1.89 (3Η, d, J=0.6 Hz), 2.30 (3H, d, J=1.0 Hz), 2.41 (3H, s), 2.96-3.06 (4H, m), 3.56-3.66 (4H, m), 7.06-7.22 (4H, m), 7.25 (2H, d, J=8.4 Hz), 7.77 (2H, d, J=8. Hz), 9.71 (1H, bs). Ή one NMR (CDC1 3): δ 1.89 (3Η, d, J = 0.6 Hz), 2.30 (3H, d, J = 1.0 Hz), 2.41 (3H, s), 2.96-3.06 (4H, m), 3.56 -3.66 (4H, m), 7.06-7.22 (4H, m), 7.25 (2H, d, J = 8.4 Hz), 7.77 (2H, d, J = 8.Hz), 9.71 (1H, bs).
元素分析値: C25H26N403S2-0.2H20として計算値 (¾) : C, 60.27; H, 5.34; N, 11.24; S, 12.87. 実測値 (¾) : C, 60.18; H, 5.42; N, 10.97; S, 12.82. Elemental analysis: Calculated as C 25 H 26 N 4 0 3 S 2 -0.2H 2 0 (¾): C, 60.27; H, 5.34; N, 11.24; S, 12.87. Observed (¾): C, 60.18; H, 5.42; N, 10.97; S, 12.82.
実施例 6-1 Example 6-1
1 -ァミノ -5, 6 -ジメチル -8- (ペンチルスルファニル )-3H -ィミダゾ [5, 1-c] [1, 4]チ ァジン- 3-オン 方法 a 1-Amino-5,6-dimethyl-8- (pentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one Method a
実施例 -24 で得られた N-[5, 6-ジメチル- 3 -ォキソ -8- (ペンチルスルファ二 ル)- 2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン ]-4-メチルベ ンゼンスルホンアミド (220 mg, 0.487 mmol) をテトラヒドロフラン (4.00 ml) に溶かし、 25%アンモニア水 (500 ml) を加えた。 反応混合物を 80°Cで 17時間封 管中加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n -へキサン -酢酸ェチル(10:1, v/v)で溶出して題記化合物(62.4mg, 43%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 127-129°Cの暗赤色結晶を得た。 N- [5,6-Dimethyl-3-oxo-8- (pentylsulfanyl) -2,3-dihydro-1H-imidazo [5, tric] [1,4] obtained in Example-24 [Thiazine-1-ylidene] -4-methylbenzenesulfonamide (220 mg, 0.487 mmol) was dissolved in tetrahydrofuran (4.00 ml), and 25% aqueous ammonia (500 ml) was added. After the reaction mixture was heated in a sealed tube at 80 ° C. for 17 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (62.4 mg, 43%) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 127-129 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3397, 3279, 3104, 2957, 2932, 2861, 1715, 1636, 1605, 1545 cm— Infrared absorption spectrum (IR) (in KBr): 3397, 3279, 3104, 2957, 2932, 2861, 1715, 1636, 1605, 1545 cm—
¾-NMR (CDC13) : δ 0.919 (3Η, t, J=6.6Hz), 1.33-1.46 (4H, m), 1.66-1.77 (2H, m), 1.74 (3H, d, J=0.8 Hz), 2.27 (3H, d, J-0.8 Hz), 2.92 (2H, t, J=7.8 Hz), 7.36 (2H, bs). ' ¾-NMR (CDC1 3): δ 0.919 (3Η, t, J = 6.6Hz), 1.33-1.46 (4H, m), 1.66-1.77 (2H, m), 1.74 (3H, d, J = 0.8 Hz) , 2.27 (3H, d, J-0.8 Hz), 2.92 (2H, t, J = 7.8 Hz), 7.36 (2H, bs).
元素分析値: C13H19N30S2として Elementary analysis: as C 13 H 19 N 3 0S 2
計算値 (%) : C, 51.56; H, 6.52; N, 13.88; S, 21.18. Calculated value (%): C, 51.56; H, 6.52; N, 13.88; S, 21.18.
実測値 (%) : C, 51.37; H, 6.27; N, 13.81; S, 21.15. Found (%): C, 51.37; H, 6.27; N, 13.81; S, 21.15.
実施例 6-2 Example 6-2
卜アミノ- 5,6-ジメチル- 8- (ィソペンチルスルファニル) - 3H-イミダゾ [5, 1 - c] [1,4]チアジン- 3 -オン Triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one
方法 a Method a
実施例 6-1と同様の方法により、実施例 4-9で得られた N- [5, 6-ジメチル- 8- (ィ ソペンチルスルファニル) -3-ォキソ - 2, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1,4]チ ァジン-卜イリデン] -4-メチルベンゼンスルホンアミド (2.00 g, 4.43 mmol) か ら題記化合物 (1.14 g, 86%) を合成した。 酢酸ェチル -ジェチルェ一テルから再 結晶して融点 174- 176°Cの暗赤色結晶を得た。 In the same manner as in Example 6-1, the N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydrido-1H obtained in Example 4-9 was obtained. The title compound (1.14 g, 86%) was synthesized from -imidazo [5,1-c] [1,4] thiazine-triylidene] -4-methylbenzenesulfonamide (2.00 g, 4.43 mmol). Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 174-176 ° C.
赤外吸収スぺクトル(IR) (KBr中) ソ: 3403, 3115, 2957, 2870, 1715, 1634, 1605, 1543 cm—1. ¾一画 R (CDCI3) : δ 0.946 (6Η, d, J=6.2 Hz)., 1.50-1.70 (3H, in), 1.74 (3H, d, J=1.2 Hz), 2.27 (3H, d, J=1.2 Hz), 2.93 (2H, t, J=8.0 Hz), 7.05 (1H, bs). 元素分析値: C13H19N30S2として Infrared absorption spectrum (IR) (in KBr) So: 3403, 3115, 2957, 2870, 1715, 1634, 1605, 1543 cm- 1 . ¾ Single stroke R (CDCI3): δ 0.946 (6Η, d, J = 6.2 Hz)., 1.50-1.70 (3H, in), 1.74 (3H, d, J = 1.2 Hz), 2.27 (3H, d, J) = 1.2 Hz), 2.93 (2H, t, J = 8.0 Hz), 7.05 (1H, bs). Elemental analysis: C 13 H 19 N 3 0S 2
計算値 (%) : C, 52.49; H, 6.44; N, 14.13; S, 21.56. Calculated value (%): C, 52.49; H, 6.44; N, 14.13; S, 21.56.
実測値 (%) : C, 52.50; H, 6.34; N, 14.11; S, 21.47. Found (%): C, 52.50; H, 6.34; N, 14.11; S, 21.47.
実施例 6 - 3 Example 6-3
1 -ァミノ- 5, 6 -ジメチル- 8 - [(4, 4, 5, 5, 5-ペンタフルォ口べンチル)スルファニ ル] - 3H -イミダゾ [5,1 - c] [1,4]チアジン- 3-オン 1-Amino-5,6-dimethyl-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine- 3-on
実施例 6-1 と同様の方法により、 実施例 4-34で得られた N- [5, 6-ジメチル -3 - ォキソ -8- [(4, 4, 5, 5, 5-ペン夕フルォロペンチル)スルファニル ]-2, 3-ジヒドロ - 1H -イミダゾ [5,l_c] [1,4]チアジン-卜イリ.デン ]_4-メチルベンゼンスルホンァ ミド (2.00 g, 4.43 mmol) から題記化合物 (1.34 g, 94¾) を合成した。 酢酸ェ チル-ジェチルエーテルから再結晶して融点 173- 175 の暗赤色結晶を得た。 In the same manner as in Example 6-1, the N- [5,6-dimethyl-3-oxo--8-[(4,4,5,5,5-pentanofluoropentyl) obtained in Example 4-34 was obtained. ) Sulfanyl] -2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine-triy.den] _4-methylbenzenesulfonamide (2.00 g, 4.43 mmol) from the title compound (1.34 g) , 94¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 173-175.
赤外吸収スペクトル(IR) (KBr中) V: 3387, 2940, 1713, 1669, 1605, 1539 cm"1. Ή-NMR (CDC13) : δ 1.74 (3Η, d, J=1.0 Hz), 2.00-2.24 (4H, m), 2.26 (3H, d, J=1.0 Hz), 3.01 (2H, t, J=7.2 Hz), 7.27 (2H, bs). Infrared absorption spectrum (IR) (in KBr) V:. 3387, 2940 , 1713, 1669, 1605, 1539 cm "1 Ή-NMR (CDC1 3): δ 1.74 (3Η, d, J = 1.0 Hz), 2.00 -2.24 (4H, m), 2.26 (3H, d, J = 1.0 Hz), 3.01 (2H, t, J = 7.2 Hz), 7.27 (2H, bs).
元素分析値: C13H14N30S2F5として Elementary analysis: as C 13 H 14 N 3 0S 2 F 5
計算値 (¾) : C, 40.30; H, 3.64; N, 10.85; S, 16.55. Calculated (¾): C, 40.30; H, 3.64; N, 10.85; S, 16.55.
実測値 (%) : C, 40.34; H, 3.46; N, 10.93; S, 16.72. Found (%): C, 40.34; H, 3.46; N, 10.93; S, 16.72.
実施例 6-4 Example 6-4
1 -ァミノ -5, 6 -ジメチル- 8- [ (3-フルオロ- 3 -メチルブチル)スルファニル] - 3H -ィ ミダゾ [5, 1-c] [1, 4]チアジン- 3 -オン 1-Amino-5,6-dimethyl-8-[(3-fluoro-3-methylbutyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 6-1 と同様の方法により、 実施例 4-40 で得られた N- [5, 6-ジメチル -8- [ (3 -フルォロ- 3-メチルプチル)スルファニル] - 3 -ォキソ - 2, 3-ジヒド口- 1H -ィ ミダゾ [5,卜 c][l, 4]チアジン-卜イリデン] -4-メチルベンゼンスルホンアミド (526 mg, 1.12雇 ol) から題記化合物 (320 mg, 91%) を合成した。 酢酸ェチル- ジェチルェ一テルから再結晶して融点 188-190°Cの暗赤色結晶を得た。 In the same manner as in Example 6-1, N- [5,6-dimethyl-8-[(3-fluoro-3-methylbutyl) sulfanyl] -3-oxo-2, obtained in Example 4-40. 3-Dihydrid-1H-dimidazo [5, tric] [l, 4] thiazine-triylidene] -4-methylbenzenesulfonamide (526 mg, 1.12 hid ol) from the title compound (320 mg, 91%) Was synthesized. Recrystallization from ethyl acetate-ethyl ether gave a dark red crystal with a melting point of 188-190 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3387, 2978, 2936, 1717, 1655, 1636, 1605, 1541 cm-1. lH - NMR (CDCI3) : δ 1.41 (6H, d, J=21.2 Hz), 1.74 (3H, d, J=1.0 Hz), 1.90-2.10 (2H, m), 2.28 (3H, d, J=1.2 Hz), 2.98-3.08 (2H, m), 7.03 (2H, bs). Infrared absorption spectrum (IR) (in KBr) V: 3387, 2978, 2936, 1717, 1655, 1636, 1605, 1541 cm- 1 . l H - NMR (CDCI3): δ 1.41 (6H, d, J = 21.2 Hz), 1.74 (3H, d, J = 1.0 Hz), 1.90-2.10 (2H, m), 2.28 (3H, d, J = 1.2 Hz), 2.98-3.08 (2H, m), 7.03 (2H, bs).
元素分析値: C13H18N30S2Fとして Elementary analysis: as C 13 H 18 N 3 0S 2 F
計算値 (¾) : C, 49.50; H, 5.75; N, 13.32; S, 20.33. Calculated (¾): C, 49.50; H, 5.75; N, 13.32; S, 20.33.
実測値 (%) : C, 49.54; H, 5.72; N, 13.39; S, 20.00. Found (%): C, 49.54; H, 5.72; N, 13.39; S, 20.00.
実施例 6 - 5 Example 6-5
トァミノ -5, 6-ジメチル- 8 - [ (3, 3 -ジメチルブチル)スルファニル] -3H-ィミダゾToamino-5,6-dimethyl-8-[(3,3-dimethylbutyl) sulfanyl] -3H-imidazo
[5, 1-c] [1, 4]チアジン- 3 -オン [5, 1-c] [1, 4] thiazin-3-one
実施例 6-1 と同様の方法により、 実施例 4-143 で得られた N- [5, 6-ジメチル -8- [(3,3-ジメチルブチル)スルファニル ]-3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ In the same manner as in Example 6-1, the N- [5,6-dimethyl-8-[(3,3-dimethylbutyl) sulfanyl] -3-oxo-2,3 obtained in Example 4-143 was obtained. -Dihydro-1H-imidazo
[5,卜 c] [1,4]チアジン-卜イリデン ]-4_メチルベンゼンスルホンアミド (1.82 g,[5, tric] [1,4] thiazine-triylidene] -4_methylbenzenesulfonamide (1.82 g,
3.91 mmol) から題記化合物 (665 mg, 55%) を合成した。 酢酸ェチル -ジェチルェ 一テルから再結晶して融点 191_193°Cの暗赤色結晶を得た。 The title compound (665 mg, 55%) was synthesized from 3.91 mmol). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 191 ° -193 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3385, 3287, 2957, 2863, 1717, 1628, 1605, 1543 cm-1. Infrared absorption spectrum (IR) (in KBr) V: 3385, 3287, 2957, 2863, 1717, 1628, 1605, 1543 cm- 1 .
Ή-NMR (CDCI3) : δ 0.949 (9Η, s), 1.52-1.70 (2H, m), 1.75 (3H, s), 2.27 (3H, s), 2.83-2.95- (2H, m), 7.34 (2H, bs). Ή-NMR (CDCI3): δ 0.949 (9Η, s), 1.52-1.70 (2H, m), 1.75 (3H, s), 2.27 (3H, s), 2.83-2.95- (2H, m), 7.34 ( 2H, bs).
実施例 6-6 Example 6-6
卜ァミノ- 5, 6-ジメチル -8- [ (5, 5, 6, 6, 6 -ぺンタフルォ口へキシル)スルファニ ル] - 3H-ィミダゾ [5, 1-c] [1,4]チアジン -3-オン Triamino-5,6-dimethyl-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine- 3-on
実施例 6-1 と同様の方法により、 実施例 4-35で得られた N- [5, 6-ジメチル- 3- ォキソ -8- [(5, 5,6,6, 6-ペンタフルォ口へキシル)スルファニル 3-ジヒドロ - 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン] -4-メチルベンゼンスルホンァ ミド (2.00 g, 3.60匪 ol) から題記化合物 (781 g, 54%) を合成した。 酢酸ェ チル-ジェチルェ一テルから再結晶して融点 155_157°Cの暗赤色結晶を得た。 In the same manner as in Example 6-1, the N- [5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluoride) obtained in Example 4-35 was obtained. Xyl) sulfanyl 3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] -4-methylbenzenesulfonamide (2.00 g, 3.60 bandol) from the title compound (781 g) , 54%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 155 to 157 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3397, 3283, 2944, 1715, 1626, 1605, 1551 cm"1. ' Infrared absorption spectrum (IR) (in KBr): 3397, 3283, 2944, 1715, 1626, 1605, 1551 cm " 1. '
一腿 R (CDC13) : δ 1.66-1.89 (4Η, m), 1.74 (3H, d, J=1.2 Hz), 1.94-2.24 (2H, m), 2.27 (3H, d, J=1.0 Hz), 2.95 (2H, t, J-7.2 Hz), 7.29 (2H, bs). 元素分析値: C14Hl6N3OS2F5として One thigh R (CDC1 3): δ 1.66-1.89 (4Η, m), 1.74 (3H, d, J = 1.2 Hz), 1.94-2.24 (2H, m), 2.27 (3H, d, J = 1.0 Hz) , 2.95 (2H, t, J-7.2 Hz), 7.29 (2H, bs). Elemental analysis: as C 14 H 16 N 3 OS 2 F 5
計算値 (%) : C, 41.89; H, 4.02; N, 10.47; S, 15.98. Calculated value (%): C, 41.89; H, 4.02; N, 10.47; S, 15.98.
実測値 (%) : C, 41.83; H, 3.89; N, 10.55; S, 15.93. Found (%): C, 41.83; H, 3.89; N, 10.55; S, 15.93.
実施例 6 - 7 Example 6-7
トァミノ- 5, 6 -ジメチル- 8- [(5, 5, 5 -トリフルォロペンチル)スルファニル ]-3H -ィ ミダゾ [5, 1 - c] [1,4]チアジン- 3-オン Tomino-5,6-dimethyl-8-[(5,5,5-trifluoropentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one
方法 b Method b
実施例 4-31で得られた N- {5, 6-ジメチル -3-ォキソ -8- [(5, 5, 5-トリフルォロぺ ンチル)スルファニル] - 2, 3-ジヒドロ- 1 H-イミダゾ [5,卜 c] [1, 4]チアジン- 1 -ィリ デン }- 4-メチルベンゼンスルホンアミド (800 mg, 1.58mmol) とギ酸アンモニゥ ム (1.05 g, 15.8靈 ol) のエタノール (30.0 ml) の懸濁液を 80°Cで 15時間加 熱した後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。残留物をシリカゲルカラムクロマトグラフィ一に付し、 n -へキサン-酢酸ェチル (10:l,'v/v) で溶出して題記化合物 (354 mg, 64¾) を暗 赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 150- 152°Cの暗赤色結晶を得た。 N- {5,6-dimethyl-3-oxo-8-[(5,5,5-trifluoropentyl) sulfanyl] obtained in Example 4-31] -2,3-dihydro-1H-imidazo [ 5, tri c] [1,4] thiazine-1-ylidene} -4-methylbenzenesulfonamide (800 mg, 1.58 mmol) and ammonium formate (1.05 g, 15.8 eol) in ethanol (30.0 ml) The suspension was heated at 80 ° C. for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: l, 'v / v) to give the title compound (354 mg, 64¾) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 150-152 ° C.
赤外吸収スぺクトル(IR) (KBr中) V 3397, 3279, 2940, 1715, 1628, 1605, 1545 cm"1. Infrared absorption spectrum (IR) (in KBr) V 3397, 3279, 2940, 1715, 1628, 1605, 1545 cm " 1 .
'H-NMR (CDC13) : δ 1.50-1.86 (4Η, m), 1.74 (3H, s), 2.00-2.32 (2H, m), 2.26 (3H, s), 2.94 (2H, t, J-7.4 Hz), 7.30 (2H, bs). 'H-NMR (CDC1 3) : δ 1.50-1.86 (4Η, m), 1.74 (3H, s), 2.00-2.32 (2H, m), 2.26 (3H, s), 2.94 (2H, t, J- 7.4 Hz), 7.30 (2H, bs).
元素分析値: C13H16N30S2F3として Elementary analysis: as C 13 H 16 N 3 0S 2 F 3
計算値 (%) : C, 44.43; H, 4.59; N, 11.96; S, 18.25. Calculated (%): C, 44.43; H, 4.59; N, 11.96; S, 18.25.
実測値 ) : C, 44.40; H, 4.68; N, 11.89; S, 18.50. Obtained): C, 44.40; H, 4.68; N, 11.89; S, 18.50.
実施例 6-8 Example 6-8
卜ァミノ- 5, 6-ジメチル- 8- [ (4, 4, 4-トリフルォ口プチル)スルファニル] -3H-ィミ ダゾ [5, 1 - c] [1 , 4]チアジン -3-ォン Triamino-5,6-dimethyl-8-[(4,4,4-trifluoromethyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine-3-one
方法 b Method b
実施例 6-7と同様の方法により、 実施例 4-41で得られた N- [5, 6-ジメチル- 3 - ォキソ -8- [(4, 4,4-トリフルォロブチル)スルファニル ]-2, 3-ジヒドロ- 1H-イミダ ゾ [5,1- c] [1,4]チアジン-卜イリデン] -4-メチルベンゼンスルホンアミド(830 mg, 1.69腿 ol) から題記化合物 (475 mg, 84¾) を合成した。 酢酸ェチル -ジェチルェ 一テルから再結晶して融点 170-172 の暗赤色結晶を得た。 N- [5,6-dimethyl-3-oxo-8-[(4,4,4-trifluorofluoro) sulfanyl] obtained in Example 4-41 by a method similar to that of Example 6-7. -2,3-dihydro-1H-imida The title compound (475 mg, 84¾) was synthesized from azo [5,1-c] [1,4] thiazine-triylidene] -4-methylbenzenesulfonamide (830 mg, 1.69 tmol). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 170-172.
赤外吸収スぺクトル(IR) (KBr中) V: 3409, 3289, 3096, 3042, 1715, 1634, 1605, 1539 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3409, 3289, 3096, 3042, 1715, 1634, 1605, 1539 cm- 1 .
Ή-NMR (CDC13) : δ 1.74 (3Η, d, J=1.2 Hz), 2.01 (2H, quintet, J=8.1 Hz), 2.16-2.36 (2H, m), 2.26 (3H, d, J=1.2 Hz), 2.99 (2H, t, J=7.8 Hz), 7.61 (2H, bs). Ή-NMR (CDC1 3): δ 1.74 (3Η, d, J = 1.2 Hz), 2.01 (2H, quintet, J = 8.1 Hz), 2.16-2.36 (2H, m), 2.26 (3H, d, J = 1.2 Hz), 2.99 (2H, t, J = 7.8 Hz), 7.61 (2H, bs).
元素分析値: C12H14N30S2F3として Elementary analysis: as C 12 H 14 N 3 0S 2 F 3
計算値 (%) : C, 42.72; H, 4.18; N, 12.45; S, 19.01. Calculated value (%): C, 42.72; H, 4.18; N, 12.45; S, 19.01.
実測値 (%) : C, 42.48; H, 4.12; N, 12.34; S, 19.10. Found (%): C, 42.48; H, 4.12; N, 12.34; S, 19.10.
以下に実施例 6-1〜6- 8 で合成した化合物および同様にして合成した化合物の 一覧を示す。 The following is a list of compounds synthesized in Examples 6-1 to 6-8 and compounds synthesized in the same manner.
〔表 6 8〕(Table 68)
差替え用弒(¾(ί2φ 〔表 6 9〕 (弒 (ί2φ (Table 69)
差替え 則 26) 〔表 70〕Substitution rules 26) (Table 70)
差替え用弒 mm 〔表 7 1〕弒 mm for replacement (Table 7 1)
差替え用弒 (m 以下に実施例 6-9〜6- 51で合成した化合物のデータを示す。 弒 (m The data of the compounds synthesized in Examples 6-9 to 6-51 are shown below.
実施例 6-9 Example 6-9
酢酸ェチル -ジィソプロピルェ一テルで再結晶し、 赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-diisopropyl ether to obtain red crystals.
融点: 116- 118°C. Melting point: 116-118 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3318, 3025, 2924, 2835, 1713, 1655, 1590, 1518, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3318, 3025, 2924, 2835, 1713, 1655, 1590, 1518, 1497 cm " 1 .
ΐ-NMR (CDC13) : δ 1.72 (3Η, s), 2.26 (3H, s), 2.28 (3H, s), 2.96 (2H, t, ΐ-NMR (CDC1 3): δ 1.72 (3Η, s), 2.26 (3H, s), 2.28 (3H, s), 2.96 (2H, t,
J=6.6 Hz), 3.80 (2H, dt, J=6.6, 6.2 Hz), 7.18-7.42 (6H, m) . J = 6.6 Hz), 3.80 (2H, dt, J = 6.6, 6.2 Hz), 7.18-7.42 (6H, m).
元素分析値: C17H19N30S2として計算値: C, 59.10; H, 5.54; N, 12.16; S, 18.56Elemental analysis: C 17 H 19 N 3 0S 2 Calculated: C, 59.10; H, 5.54 ; N, 12.16; S, 18.56
(%)、 実測値: C, 59.10; H, 5.51; N, 12.03; S, 18.01 (%) . (%), Found: C, 59.10; H, 5.51; N, 12.03; S, 18.01 (%).
実施例 6 - 10 Example 6-10
酢酸ェチル -ジィソプロピルエーテルで再結晶し、 赤色結晶を得た。 The crystals were recrystallized from ethyl acetate-diisopropyl ether to obtain red crystals.
融点: 117- 119°C. Melting point: 117-119 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3310, 2959, 2932, 2857, 1701, 1661, 1582 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3310, 2959, 2932, 2857, 1701, 1661, 1582 cm " 1 .
NMR (CDCI3) : δ 0.910 (3Η, t, J=6.2 Hz), 1.24-1.44 (4H, m), 1.60-1.72 (2H, m), 1.74 (3H, d, J=1.2 Hz), 2.26 (3H, d, J=1.0 Hz), 2.48 (3H, s), 3.50 (2H, dt, J=7.0, 5.8 Hz), 7.41 (1H, bs). NMR (CDCI3): δ 0.910 (3Η, t, J = 6.2 Hz), 1.24-1.44 (4H, m), 1.60-1.72 (2H, m), 1.74 (3H, d, J = 1.2 Hz), 2.26 ( 3H, d, J = 1.0 Hz), 2.48 (3H, s), 3.50 (2H, dt, J = 7.0, 5.8 Hz), 7.41 (1H, bs).
元素分析値: C14H21N30S2として計算値: C, 53.99; H, 6.80; N, 13.49; S, 20.59Elemental analysis: C 14 H 21 N 3 0S 2 Calculated: C, 53.99; H, 6.80 ; N, 13.49; S, 20.59
(%), 実測値: C, 53.83; H, 6.65; N, 13.40; S, 20.52 (%) . (%), Found: C, 53.83; H, 6.65; N, 13.40; S, 20.52 (%).
実施例 6 - 11 Examples 6-11
酢酸ェチル-ジイソプロピルエーテルで再結晶し、 赤色結晶を得た。 Recrystallization from ethyl acetate-diisopropyl ether gave red crystals.
融点: 130- 132 . Melting point: 130-132.
赤外吸収スペクトル(IR) (KBr中) V: 3320, 2957, 2932, 2859, 1715, 1591, 1516 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3320, 2957, 2932, 2859, 1715, 1591, 1516 cm " 1 .
'H-NMR (CDC13) : δ 0.866 (3Η, t, J=7.0Hz), 1.22-1.36 (4H, m), 1.52-1.66 (2H, m), 1.73 (3H, s), 2.28 (3H, s) , 2.35 (3H, s), 2.84 (2H, t, J=7.8 Hz), 4.65 (2H, d, J-5.4 Hz), 7.12-7.30 (4H, m), 7.71 (1H, bs). 'H-NMR (CDC1 3) : δ 0.866 (3Η, t, J = 7.0Hz), 1.22-1.36 (4H, m), 1.52-1.66 (2H, m), 1.73 (3H, s), 2.28 (3H , s), 2.35 (3H, s), 2.84 (2H, t, J = 7.8 Hz), 4.65 (2H, d, J-5.4 Hz), 7.12-7.30 (4H, m), 7.71 (1H, bs) .
元素分析値: C21H27N30S2として計算値: C, 62.81; H, 6.78; N, 10.46; S, 15.97 (%)、 実測値: C, 62.58; H, 6.65; , 10.29; S, 16.02 {%) . 実施例 6-12 Elemental analysis: Calculated as C 21 H 27 N 30 S 2 : C, 62.81; H, 6.78; N, 10.46; S, 15.97 (%), Found: C, 62.58; H, 6.65;, 10.29; S, 16.02 (%).
濃赤色油状物. Dark red oil.
赤外吸収スペクトル(IR) (KBr中) V: 3230, 3027, 2955, 2930, 1717, 1655, 1590, 1518, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3230, 3027, 2955, 2930, 1717, 1655, 1590, 1518, 1497 cm " 1 .
Έ-NMR (CDC13) : 50.903 (3H, t, J=6.6 Hz), 1.16-1.40 (4H, m), 1.40-1.64 (2H, m), 1.71 (3H, s), 2.26 (3H, d, J=0.8 Hz), 2.71 (2H, t, J=7.8 Hz), 2.96 (2H, t, J=6.6 Hz), 3.80 (2H, q, J=6.2 Hz), 7.15-7.37 (5H, m), 7.43 (1H, bs). 実施例 6-13 Έ-NMR (CDC1 3): 50.903 (3H, t, J = 6.6 Hz), 1.16-1.40 (4H, m), 1.40-1.64 (2H, m), 1.71 (3H, s), 2.26 (3H, d , J = 0.8 Hz), 2.71 (2H, t, J = 7.8 Hz), 2.96 (2H, t, J = 6.6 Hz), 3.80 (2H, q, J = 6.2 Hz), 7.15-7.37 (5H, m ), 7.43 (1H, bs).
NMR (DMSO— d3, 300 MHz): 6 2.45 (3H, s), 5.19 (1H, d, J= 7.9 Hz), 6.54 (1H, d, J= 7.9 Hz), 7.46 (1H, br), 8.69 (1H, br). NMR (DMSO- d 3, 300 MHz ): 6 2.45 (3H, s), 5.19 (1H, d, J = 7.9 Hz), 6.54 (1H, d, J = 7.9 Hz), 7.46 (1H, br), 8.69 (1H, br).
元素分析値: C7H7N3OS2 · 0.15 0として計算値: C, 38.93; H, 3.41; N, 19.46; S, 29.69 (%), 実測値: C, 39.15; H, 3.40; N, 19.18; S, 29.78 (%) . Elemental analysis: C 7 H 7 N 3 OS 2 · 0.15 0 Calculated: C, 38.93; H, 3.41; N, 19.46; S, 29.69 (%), found: C, 39.15; H, 3.40; N , 19.18; S, 29.78 (%).
実施例 6 - 14 Example 6-14
3-[(1 -ィミノ- 5, 6_ジメチル- 3_ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, ] チアジン- 8-ィル)スルファエル]プロピオン酸ェチル 3-[(1-imino-5,6_dimethyl-3_oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,] thiazin-8-yl) sulfael] ethyl propionate
実施例 2- 5で得た化合物(705 mg, 1.46 mmol)とギ酸アンモニゥム (486 mg, 7.32 腿 ol) のエタノール (24 ml) 懸濁液を 15時間加熱還流した。 反応液を酢酸ェチ ル(50 ml)で希釈し、 10%塩化アンモニゥム水溶液および飽和食塩水で洗浄後、 硫 酸ナトリウムで乾燥し、 濃縮乾固後、 残渣を酢酸ェチルおよびジェチルエーテル から結晶化して標題化合物 (264 mg, 0.808 mmol)を濃紫色粉末として得た。 A suspension of the compound (705 mg, 1.46 mmol) obtained in Example 2-5 and ammonium formate (486 mg, 7.32 tmol) in ethanol (24 ml) was heated under reflux for 15 hours. The reaction mixture was diluted with ethyl acetate (50 ml), washed with 10% aqueous ammonium chloride solution and saturated saline, dried over sodium sulfate, concentrated to dryness, and the residue was crystallized from ethyl acetate and getyl ether. To give the title compound (264 mg, 0.808 mmol) as a dark purple powder.
'H-NMR (300MHz, CDC13) : δ 1.28 (3Η, t, J= 7.2 Hz), 1.74 (3H, s), 2.26 (3H, s), 2.75 (2H, t, J= 6.9 Hz), 3.19 (2H, t, J= 6.9 Hz), 4.18 (2H, q, J= 7.2 Hz), 7.3 (1H, br), 8.61 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 1.28 (3Η, t, J = 7.2 Hz), 1.74 (3H, s), 2.26 (3H, s), 2.75 (2H, t, J = 6.9 Hz), 3.19 (2H, t, J = 6.9 Hz), 4.18 (2H, q, J = 7.2 Hz), 7.3 (1H, br), 8.61 (1H, br).
元素分析値: C13H17N303S2 · 0.1¾0.として計算値: C, 47.43; H, 5.20; N, 12.76; S, 19.47 ( ) 、 実測値: C, 47.55; H, 5.26; N, 12.47; S, 19.23 (%) . . Elemental analysis: C 13 H 17 N 3 0 3 S 2 · 0.1¾0 Calculated: C, 47.43; H, 5.20 ; N, 12.76; S, 19.47 (), Found: C, 47.55; H, 5.26 ; N, 12.47; S, 19.23 (%).
融点. 159.5-161.0°C Melting point.159.5-161.0 ° C
実施例 6 - 15 Example 6-15
8- (シクロへプチルスルファ二ル)- 1-ィミノ- 5, 6-ジメチル -1, 2-ジヒド口 -3H -ィ ミダゾ [5,1 - c] [1,4]チアジン- 3-オン 8- (cycloheptylsulfanyl) -1-imino-5,6-dimethyl-1,2-dihydric -3H-di Midazo [5,1-c] [1,4] thiazin-3-one
実施例 4-103で得た化合物 (603 mg, 1.26 mmol)とギ酸アンモニゥム (419 mg, 6.31匪 ol) のエタノール (20 ml) 懸濁液を 22時間加熱還流した。 反応液を濃縮 後、 酢酸ェチル(40 ml)で希釈し、 10%塩ィ匕アンモニゥム水溶液および飽和食塩水 で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固した。 残渣を,酢酸ェチルおよびジ ェチルエーテルから結晶化して標題化合物 (303 mg, 0·937ΙΜΟ1)を赤褐色粉末と して得た。 A suspension of the compound (603 mg, 1.26 mmol) obtained in Example 4-103 and ammonium formate (419 mg, 6.31 bandol) in ethanol (20 ml) was heated under reflux for 22 hours. The reaction solution was concentrated, diluted with ethyl acetate (40 ml), washed with a 10% aqueous solution of sodium chloride and saturated brine, dried over sodium sulfate, and concentrated to dryness. The residue was crystallized from ethyl acetate and dimethyl ether to give the title compound (303 mg, 0.937-1) as a red-brown powder.
lH- NMR (300MHz, CDC13) : d 1.50-1.80 (10H, m), 1.73 (3H, q, J= 0.6 Hz), 2.11 (2H, in), 2.26 (3H, q, J= 0.6 Hz), 3.46 (1H, sept, J= 4.5 Hz), 7.30 (1H, br), 約 8.2 (1H, br). lH- NMR (300MHz, CDC1 3) : d 1.50-1.80 (10H, m), 1.73 (3H, q, J = 0.6 Hz), 2.11 (2H, in), 2.26 (3H, q, J = 0.6 Hz) , 3.46 (1H, sept, J = 4.5 Hz), 7.30 (1H, br), about 8.2 (1H, br).
元素分析値: C15H21N30S2として計算値: (:, 55.69; H, 6.54; N, 12.99; S, 19.83Elemental analysis: C 15 H 21 N 3 0S 2 Calculated: (:, 55.69; H, 6.54; N, 12.99; S, 19.83
(%) 、 実測値: C, 55.58; H, 6.49; N, 12.86; S, 19.83 (%) . (%), Found: C, 55.58; H, 6.49; N, 12.86; S, 19.83 (%).
融点. 202.5- 203.5=0 , 202.5- 203.5 = 0,
実施例 6 - 16 Example 6-16
卜ィミノ -5,6-ジメチル- 8-[(4 -メチルペンチル) スルファニル] - 1, 2-ジヒドロ - 3H -ィミダゾ [5, 1- c] [1, 4]チアジン - 3-オン Trimino-5,6-dimethyl-8-[(4-methylpentyl) sulfanyl] -1,2-dihydro-3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4-84で得た化合物 (495 mg, 1.06 誦 ol)とギ酸アンモニゥム (353 mg, 5.32腿01) のエタノール (20ml) 懸濁液を 70 で 3日間、 80°Cで 6.5時間攪拌 した。 反応液を濃縮後、 酢酸ェチル (50 ml)で希釈し、 10%塩化アンモニゥム水溶 液および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固した。 残渣を シリカゲル(10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:卜 8:2、 0.1¾ ギ酸含有) で精製してジェチルエーテルから結晶化して標題化合物 (238 mg, 0.764體 ol)を紫色粉末として得た。 A suspension of the compound obtained in Example 4-84 (495 mg, 1.06 ol) and ammonium formate (353 mg, 5.32, 01) in ethanol (20 ml) was stirred at 70 for 3 days and at 80 ° C for 6.5 hours. . The reaction solution was concentrated, diluted with ethyl acetate (50 ml), washed with a 10% aqueous solution of ammonium chloride and saturated saline, dried over sodium sulfate, and concentrated to dryness. The residue was purified by silica gel (10 g) chromatography (ethyl acetate-hexane, 1: 8: 2, containing 0.1¾formic acid) and crystallized from getyl ether to give the title compound (238 mg, 0.764 vol). Obtained as a purple powder.
Ή-NM (300MHz, CDC13) : δ 0.90 (6H, d, J= 6.6 Hz), 1.29 (2H, brq, J= 8.7 Hz), 1.59 (1H, sept, J= 6.5 Hz ), 1.71 (2H, m), 1.74 (3H, q, J= 1.0 Hz), 2.26 (3H, q, J- 0.6 Hz), 2.92 (2H, t, J= 7.5 Hz), 7.26 (1H, s), 8.12 (1H, s). Ή-NM (300MHz, CDC1 3 ): δ 0.90 (6H, d, J = 6.6 Hz), 1.29 (2H, brq, J = 8.7 Hz), 1.59 (1H, sept, J = 6.5 Hz), 1.71 (2H , m), 1.74 (3H, q, J = 1.0 Hz), 2.26 (3H, q, J- 0.6 Hz), 2.92 (2H, t, J = 7.5 Hz), 7.26 (1H, s), 8.12 (1H , s).
元素分析値: C14 iN30S2 · 0.4 0として計算値: C, 52.77; H, 6.90; N, '13.18; S, 20.12 (%) 、 実測値: C, 52.85; H, 6.80; N, 13.20; S, 19.83 (%) . Elemental analysis: C 14 iN 3 0S 2 · 0.40 Calculated: C, 52.77; H, 6.90; N, '13 .18; S, 20.12 (%), found: C, 52.85; H, 6.80; N, 13.20; S, 19.83 (%).
融点. 136.8-137. Ot 実施例 6-17 136.8-137. Ot Example 6-17
8 - [(シク口へキシルメチル)スルファニル] -卜ィミノ- 5, 6-ジメチル- 1, 2 -ジヒド ロ- -イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン 8-[(cyclohexylmethyl) sulfanyl] -trimino-5,6-dimethyl-1,2-dihydro- -imidazo [5, tric] [1,4] thiazin-3-one
実施例 4-68で得た化合物 (559 mg, 1.17腿 ol)とギ酸アンモニゥム (388 mg, 5.85 mmol) のエタノール (19 ml) 懸濁液を 40時間加熱還流した。 この間ギ酸ァ ンモニゥム (230 mg, 3.51 mmol)を 2回反応液に加えた。 反応液を濃縮後、 酢酸 ェチル(40 ml)で希釈し、 10%塩化アンモニゥム水溶液および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固した。 残渣を酢酸ェチルから結晶化して標題 化合物 (258 mg, 0.798 mmol)を赤紫色粉末として椿た。 A suspension of the compound (559 mg, 1.17 mol) obtained in Example 4-68 and ammonium formate (388 mg, 5.85 mmol) in ethanol (19 ml) was heated under reflux for 40 hours. During this time, ammonium formate (230 mg, 3.51 mmol) was added twice to the reaction solution. The reaction solution was concentrated, diluted with ethyl acetate (40 ml), washed with a 10% aqueous solution of ammonium chloride and saturated saline, dried over sodium sulfate, and concentrated to dryness. The residue was crystallized from ethyl acetate to give the title compound (258 mg, 0.798 mmol) as a reddish purple powder.
一 NMR (200MHz, CDC13) : δ 0.98 (2H, brq, J= 12 Hz), 1.25 (3H, m), 1.61 (1H, m), 1.70-1.90 (5H, m), 1.74 (3H, brs), 2.26 (3H, brs), 2.83 (2H, d, J= 7.0 Hz), 約 7.5 (1H, br), 約 8.2 (1H, br). One NMR (200MHz, CDC1 3): δ 0.98 (2H, brq, J = 12 Hz), 1.25 (3H, m), 1.61 (1H, m), 1.70-1.90 (5H, m), 1.74 (3H, brs ), 2.26 (3H, brs), 2.83 (2H, d, J = 7.0 Hz), about 7.5 (1H, br), about 8.2 (1H, br).
元素分析値: C15H21N3OS2として計算値: C, 55.69; H, 6.54; N, 12.99; S, 19.83 (%) 、 実測値: C, 55.56; H, 6.51; , 12.83; S, 19.83 (%) . Elemental analysis: C 15 H 21 N 3 OS 2 Calculated: C, 55.69; H, 6.54 ; N, 12.99; S, 19.83 (%), Found: C, 55.56; H, 6.51 ;, 12.83; S , 19.83 (%).
融点. 190.0- 192.0°C Melting point.190.0- 192.0 ° C
実施例 6 - 18 Examples 6-18
トァミノ- 5, 6 -ジメチル -8- [(4-フルォロブチル)スルファニル ]_3H-ィミダゾ [5,1-c] [1,4]チアジン -3-オン Tomino-5,6-dimethyl-8-[(4-fluorobutyl) sulfanyl] _3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 -33 で得られた N-{5, 6-ジメチル -8- [ (4-フルォロブチル)スルファニ ル]- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン }-4- メチルベンゼンスルホンアミド (204 mg, 0.448腿 ol) とギ酸アンモニゥム (297 mg, 4.48 mmol) のエタノール (8.00ml) の懸濁液を 80°Cで 20時間加熱した後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧 下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに付し、 n-へキサ ン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (116 mg, 86¾) を暗赤色結晶 として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 118- 120°Cの暗赤 色結晶を得た。 N- {5,6-dimethyl-8-[(4-fluorobutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] obtained in Example -33 A suspension of 1,4] thiazine-1-ylidene} -4-methylbenzenesulfonamide (204 mg, 0.448 mol) and ammonium formate (297 mg, 4.48 mmol) in ethanol (8.00 ml) was added at 80 ° C. After heating for 20 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (116 mg, 86¾) as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 118-120 ° C.
IR (KBr)ソ : 3393, 3283, 2965, 2938, 1713, 1628, 1625, 1548 cm"1. IR (KBr): 3393, 3283, 2965, 2938, 1713, 1628, 1625, 1548 cm " 1 .
'H-NMR (CDC13) : (51.74 (3H, d, J=1.2 Hz), 1.62-1.96 (4H, m), 2.26 (3H, d, J=1.0 Hz), 2.98 (2H, t, J=7.4 Hz), 4.50 (2H, dt, J=47.2, 5.4 Hz), 7.64 (2H, bs). 'H-NMR (CDC1 3) : (51.74 (3H, d, J = 1.2 Hz), 1.62-1.96 (4H, m), 2.26 (3H, d, J = 1.0 Hz), 2.98 (2H, t, J = 7.4 Hz), 4.50 (2H, dt, J = 47.2, 5.4 Hz), 7.64 (2H, bs).
元素分析値: C12H1()N30S2Fとして Elemental analysis: as C 12 H 1 () N 3 0S 2 F
計算値 (%) : C, 47.82; H, 5.35; N, 13.94; S, 21.28. Calculated (%): C, 47.82; H, 5.35; N, 13.94; S, 21.28.
実測値 (%) : C, 47.62; H, 5.39; N, 13.80; S, 21.24. Found (%): C, 47.62; H, 5.39; N, 13.80; S, 21.24.
実施例 6-19 Example 6-19
8- [ (2, 2 -ジメチルぺンチル) スルファニル] - 1 -ィミノ -5, 6 -ジメチル -1 , 2-ジヒド ロ- 3H-イミダゾ [5,卜 c] [1,4]チアジン -3-オン 8-[(2,2-dimethylpentyl) sulfanyl] -1-imino-5,6-dimethyl-1,2, -dihydro-3H-imidazo [5, tric] [1,4] thiazine-3- On
実施例 4-503で得た化合物(1.04 g, 2.17 mmol)とギ酸アンモニゥム (720 mg, 10.9匪01) のエタノール (20ml) 懸濁液を 20時間加熱還流した。 反応液を濃縮 後、 酢酸ェチルで抽出した。 有機層を 10%塩化アンモニゥム水溶液および飽和食 塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固後、 残渣をシリカゲル (10 g) のクロマトグラフィー (酢酸ェチル一へキサン、 3:7-7:3) に付した。酢酸ェチル —へキサン(6: 4-7: 3)の画分を濃縮乾固後酢酸ェチルおよびジェチルエーテルか ら結晶化して表題化合物(338 mg, 1.04 mmol)を紫色粉末として得た。 A suspension of the compound (1.04 g, 2.17 mmol) obtained in Example 4-503 and ammonium formate (720 mg, 10.9 band01) in ethanol (20 ml) was heated under reflux for 20 hours. After the reaction solution was concentrated, it was extracted with ethyl acetate. The organic layer was washed with 10% aqueous ammonium chloride solution and saturated saline, dried over sodium sulfate, concentrated to dryness, and the residue was chromatographed on silica gel (10 g) (ethyl acetate, hexane, 3: 7-7: 3) The fraction of ethyl acetate-hexane (6: 4-7: 3) was concentrated to dryness and crystallized from ethyl acetate and getyl ether to give the title compound (338 mg, 1.04 mmol) as a purple powder.
Ή-NMR (300MHz, CDC13) : 50.92 (3H, dt, J= 1.8, 4.5 Hz), 1.01 (6H, s), 1.29 (4H, brd, J= 3.6 Hz), 1.75 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.92 (2H, s). Ή-NMR (300MHz, CDC1 3 ): 50.92 (3H, dt, J = 1.8, 4.5 Hz), 1.01 (6H, s), 1.29 (4H, brd, J = 3.6 Hz), 1.75 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.92 (2H, s).
元素分析値: C15H23N3OS2 O.2H20として計算値: C, 54.74; H, 7.17; N, 12.77; S, 19.49 (%) 、 実測値: C, 54.78; H, 7.09; N, 12.99; S, 19.51 ( ) . Elemental analysis: C 15 H 23 N 3 OS 2 O.2H 2 0 Calculated: C, 54.74; H, 7.17 ; N, 12.77; S, 19.49 (%), Found: C, 54.78; H, 7.09 N, 12.99; S, 19.51 ().
nip. 139.0-140.3°C nip.139.0-140.3 ° C
実施例 6-20 Example 6-20
卜ァミノ- 5, 6-ジメチル- 8- [(3, 3, 4, 4, 4-ペンタフルォロブチル)スルファニ ル] - 3H-ィミダゾ [5, 1 - c] [1, 4]チアジン- 3 -才ン Triamino-5,6-dimethyl-8-[(3,3,4,4,4-pentafluorobutyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine- 3-year-old
実施例 4-502で得られた N-{5, 6-ジメチル- 3-ォキソ -8- [(3,3,4,4,4-ペンタフ ルォ口プチル)スルファエル] -2, 3-ジヒド口- 1H-ィミダゾ [5,卜 c] [1, 4]チアジン - 1 -イリデン }-4-メチルベンゼンスルホンアミド (311 mg, 0.593 mmol) とギ酸ァ ンモニゥム (393 mg, 5.93mmol) のエタノール (12.0 ml) の懸濁液を 80°Cで 20.5 時間加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (256 mg, 89¾) 'を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 172-174°Cの暗赤色結晶を得た。 N- {5,6-Dimethyl-3-oxo-8-[(3,3,4,4,4-pentafluoromethylbutyl) sulfael] obtained in Example 4-502 -2,3-Dihydrido -1H-imidazo [5, tric] [1, 4] thiazine-1-ylidene} -4-methylbenzenesulfonamide (311 mg, 0.593 mmol) and ammonium formate (393 mg, 5.93 mmol) in ethanol (12.0 was heated at 80 ° C for 20.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) Thereafter, the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (256 mg, 89¾) ′ as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 172-174 ° C.
IR (KBr) V: 3397, 2940, 1732, 1715, 1701, 1655, 1607, 1535 cm"1. IR (KBr) V: 3397, 2940, 1732, 1715, 1701, 1655, 1607, 1535 cm " 1 .
Ή-NMR (CDC13) : (51.75 (3H, s), 2.27 (3H, s), 2.38-2.58 (2H, m), 3.15 (2H, t, J=8.1 Hz), 7.34 (2H, bs). Ή-NMR (CDC1 3): (51.75 (3H, s), 2.27 (3H, s), 2.38-2.58 (2H, m), 3.15 (2H, t, J = 8.1 Hz), 7.34 (2H, bs) .
元素分析値: C12H12N3OS2F5 -0.3H20として Elemental analysis: as C 12 H 12 N 3 OS 2 F 5 -0.3H 20
計算値 ( ) : C, 38.05; H, 3.35; N, 11.09; S, 16.93. Calculated (): C, 38.05; H, 3.35; N, 11.09; S, 16.93.
実測値 (¾) : C, 38.09; H, 3.09; N, 11.14; S, 17.27. Found (¾): C, 38.09; H, 3.09; N, 11.14; S, 17.27.
実施例 6-21 Example 6-21
卜ァミノ- 5, 6 -ジメチル -8- [(6, 6, 7, 7, 7-ペンタフルォ口へプチル)スルファニ ル]- 3Hイミダゾ [5,卜 c] [1,4]チアジン -3-オン · Triamino-5,6-dimethyl-8-[(6,6,7,7,7-pentafluoroheptyl) sulfanyl] -3H imidazo [5, tric] [1,4] thiazin-3-one ·
実施例 6-7と同様の方法により、実施例 4-501で得られた N - [5, 6-ジメチル -3 - ォキソ- 8- [ (3, 3, 4, 4, 4 -ペン夕フルォ口ブチル)スルファニル] -2, 3-ジヒドロ- 1H - イミダゾ [5, l-c] [1,4]チアジン- 1-ィリデン ]-4-メチルベンゼンスルホンアミド (750 mg, 1.32 mmol) から題記化合物 (250 mg, 46%) を合成した。 酢酸ェチル- ジェチルエーテルから再結晶して融点 1U- 116°Cの暗赤色結晶を得た。 According to a method similar to that of Example 6-7, N- [5,6-dimethyl-3-oxo- 8-[(3,3,4,4,4-pentenefluoro) obtained in Example 4-501 was obtained. Butyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazin-1-ylidene] -4-methylbenzenesulfonamide (750 mg, 1.32 mmol) from the title compound (250 mg mg, 46%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 1U-116 ° C.
IR (KBr) V : 3399, 3289, 3104, 3032, 2946, 1868, 1715, 1632, 1605, 1541 cm"'.. ¾-NMR (CDCI3) : (51.40-1.86 (6H, m), 1.74 (3H, d, J=1.2 Hz), 1.90-2.20 (2H, m), 2.26 (3H, s), 2.93 (2H, t, J=7.6 Hz), 7.78 ,.(2H, bs). IR (KBr) V: 3399, 3289, 3104, 3032, 2946, 1868, 1715, 1632, 1605, 1541 cm "'.. ¾-NMR (CDCI3): (51.40-1.86 (6H, m), 1.74 (3H , d, J = 1.2 Hz), 1.90-2.20 (2H, m), 2.26 (3H, s), 2.93 (2H, t, J = 7.6 Hz), 7.78,. (2H, bs).
元素分析値: C15H18N30S2F5として Elementary analysis: as C 15 H 18 N 3 0S 2 F 5
計算値 ( ) : C, 43.37; H, 4.37; N, 10.11; S, 15.44. Calculated (): C, 43.37; H, 4.37; N, 10.11; S, 15.44.
実測値 (%) : C, 43.55; H, 4.57; N, 10.09; S, 15.18. Found (%): C, 43.55; H, 4.57; N, 10.09; S, 15.18.
実施例 6-Example 6-
1-アミノ- 5, 6 -ジメチル- 8 - [(6, 6, 6-トリフルォ口へキシル)スルファニル] - 3H -ィ ミダゾ [5, I - c] [1, 4]チアジン -3-オン 1-Amino-5,6-dimethyl-8-[(6,6,6-trifluorohexyl) sulfanyl] -3H-imidazo [5, I-c] [1,4] thiazin-3-one
実施例 6 - 7と同様の方法により、実施例 4-506で得られた N_[5, 6 -ジメチル- 3 - ォキソ - 8 (6, 6, 6-トリフルォ口へキシル)スルファニル ]-2, 3 -ジヒドロ- 1H-イミ ダゾ [5,卜 c] [1,4]チアジン-卜イリデン] -4-メチルベンゼンスルホンアミド (658 mg, 1.27 mrnol) から題記化合物 (290 mg, 63%) を合成した。 酢酸ェチル-ジェチ ルエーテルから再結晶して融点 114- 116°Cの暗赤色結晶を得た。 In the same manner as in Examples 6-7, N_ [5,6-dimethyl-3-oxo-8 (6,6,6-trifluorohexyl) sulfanyl] -2, obtained in Example 4-506. 3-dihydro-1H-imi The title compound (290 mg, 63%) was synthesized from dazo [5, tric] [1,4] thiazine-triylidene] -4-methylbenzenesulfonamide (658 mg, 1.27 mrnol). Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals with a melting point of 114-116 ° C.
IR (KBr) V: 3399, 3285, 3108, 3021, 2942, 1715, 1626, 1605, 1541 cm-1. ¾-NMR (CDC13) : δ 1.40—1.86 (6Η, m), 1.74 (3H, d, J=0.8 Hz), 1.98-2.22 (2H, m), 2.26 (3H, s), 2.93 (2H, t, J=7.8 Hz), 7.50 (2H, bs) . IR (KBr) V:. 3399 , 3285, 3108, 3021, 2942, 1715, 1626, 1605, 1541 cm- 1 ¾-NMR (CDC1 3): δ 1.40-1.86 (6Η, m), 1.74 (3H, d , J = 0.8 Hz), 1.98-2.22 (2H, m), 2.26 (3H, s), 2.93 (2H, t, J = 7.8 Hz), 7.50 (2H, bs).
元素分析値: OS2F5として Elemental analysis: as OS 2 F 5
計算値 (%) : C, 46.01; H, 4.96; N, 11.50; S, 17.55. Calculated value (%): C, 46.01; H, 4.96; N, 11.50; S, 17.55.
実測値 (%) : C, 45.79; H, 5.13; N, 11.67; S, 17.68. Found (%): C, 45.79; H, 5.13; N, 11.67; S, 17.68.
実施例 6-23 Example 6-23
1 -ァミノ- 5, 6-ジメチル- 8- [ (5-フルォ口ぺンチル)スルファニル] - 3H-ィミダゾ [5, 1-c] [1, 4]チアジン- 3 -オン 1-Amino-5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4-508で得られた {5,6-ジメチル-3-ォキソ-8-[(3,3,4,4,4-ぺン夕フ ルォロブチル)スルファニル ]-2, 3-ジヒドロ- m -イミダゾ [5, 1-c] [1,4]チアジン -卜イリデン }- 4-メチルベンゼンスルホンアミド (900 mg, 1.92 mmol) とギ酸ァ ンモニゥム (1.27 g, 19.2 mmol) のエタノール (38.0 ml) の懸濁液を 80°Cで 18 時間加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (583 mg, 96%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 140- 142°Cの暗赤色結晶を得た。 {5,6-Dimethyl-3-oxo-8-[(3,3,4,4,4-benzoylfluorobutyl) sulfanyl] -2,3-dihydro-m obtained in Example 4-508 -Imidazo [5,1-c] [1,4] thiazine-triylidene}-Ethanol (38.0 ml) of 4-methylbenzenesulfonamide (900 mg, 1.92 mmol) and ammonium formate (1.27 g, 19.2 mmol) The suspension was heated at 80 ° C for 18 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (583 mg, 96%) as dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 140-142 ° C.
IR (KBr) リ: 3403, 3285, 3112, 2936, 2863, 1715, 1634, 1605, 1541 cm— Ή一 NMR (CDC13) : 51.50-1.64 (2H, m), 1.64-1.86 (4H, m), 1.74 (3H, d, J=0.9 •Hz), 2.27 (3H, d, J=1.2 Hz), 2.94 (2H, t, J=7, 5 Hz) , 4.47 (2H, dt, J=47.1, 5.4 Hz), 7.43 (2H, bs) . IR (KBr) Re: 3403, 3285, 3112, 2936 , 2863, 1715, 1634, 1605, 1541 cm- Ή one NMR (CDC1 3): 51.50-1.64 ( 2H, m), 1.64-1.86 (4H, m) , 1.74 (3H, d, J = 0.9 Hz), 2.27 (3H, d, J = 1.2 Hz), 2.94 (2H, t, J = 7, 5 Hz), 4.47 (2H, dt, J = 47.1, 5.4 Hz), 7.43 (2H, bs).
元素分析値: C13H18N30S2Fとして Elementary analysis: as C 13 H 18 N 3 0S 2 F
計算値 (¾) : C, 49.50; H, 5.75;'! N, 13.32; S, 20.33. Calculated value (¾): C, 49.50; H, 5.75; ' ! N, 13.32; S, 20.33.
実測値 (¾) : C, 49.48; H, 5.60; N, 13.35; S, 20.32. Found (¾): C, 49.48; H, 5.60; N, 13.35; S, 20.32.
実施例 6 - 24 ェチル 5 - [卜ァミノ- 5,6-ジメチル- 3-ォキソ - 3H-イミダゾ [5,卜 c] [1,4]チアジン - 8 -ィル]スルファニル]ペン夕ノエ一卜 Examples 6-24 Ethyl 5- [triamino-5,6-dimethyl-3-oxo-3H-imidazo [5, tric] [1,4] thiazine-8-yl] sulfanyl] pen
実施例 4-522で得られたェチル 5 - [(5, 6-ジメチル-卜 {[(4 -メチルフエニル)ス ルホニル]ィミノ }- 3-ォキソ - 2, 3 -ジヒドロ -1H-ィミダゾ [5, 1-c] [1, 4]チアジン -8 -ィル)スルファニル]ペンタノェ一ト (1.34 g, 2.63匪 ol) とギ酸アンモニゥ ム (1.75 g, 26.3歷 ol) のエタノール (26.0 ml) の懸濁液を 80°Cで 17時間加 熱した後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。残留物をシリカゲルカラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (695 mg, 74%) を暗 赤色結晶として得た。 酢酸ェチル-ジェチルェ一テルから再結晶して融点 92.0-94.0°Cの暗赤色結晶を得た。 Ethyl obtained in Example 4-522 5-[(5,6-Dimethyl-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, Suspension of 1-c] [1,4] thiazine-8-yl) sulfanyl] pentanoate (1.34 g, 2.63 ol) and ammonium formate (1.75 g, 26.3 ol) in ethanol (26.0 ml) After the solution was heated at 80 ° C for 17 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (695 mg, 74%) as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 92.0-94.0 ° C.
IR (KBr) V: 3405, 3283, 3115, 2978, 2936, 1723, 1634, 1665, 1537 cm"1. Ή-NMR (CDC13) : 51.26 (3H, d, J=6.9 Hz), 1.70-1.84 (4H, m), 1.74 (3H, s), 2.26 (3H, s), 2.32-2.40 (2H, m), 2.88-2.98 (2H, m), 4.14 (2H, q, J=6.9 Hz), 7.51 (2H, bs). IR (KBr) V:. 3405 , 3283, 3115, 2978, 2936, 1723, 1634, 1665, 1537 cm "1 Ή-NMR (CDC1 3): 51.26 (3H, d, J = 6.9 Hz), 1.70-1.84 (4H, m), 1.74 (3H, s), 2.26 (3H, s), 2.32-2.40 (2H, m), 2.88-2.98 (2H, m), 4.14 (2H, q, J = 6.9 Hz), 7.51 (2H, bs).
元素分析値: C15H21N303S2として Elemental analysis: as C 15 H 21 N 3 0 3 S 2
計算値 (¾) : C, 49.92; H, 5.87; N, 11.64; S, 17.77. Calculated (¾): C, 49.92; H, 5.87; N, 11.64; S, 17.77.
実測値 (%) : C, 49.94; H, 5.79; N, 11.41; S, 17.72. Found (%): C, 49.94; H, 5.79; N, 11.41; S, 17.72.
実施例 6 - 25 Example 6-25
8- (シクロォクチルスルファ二ル)- 1-ィミノ- 5, 6-ジメチル- 1, 2-ジヒドロ- 3H -ィ ミダゾ [5, 1-c] [1, 4]チアジン - 3 -オン 8- (cyclooctylsulfanyl) -1-imino-5,6-dimethyl-1,2-dihydro-3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4-164で得た粗製の化合物 (純度 80%, 4.00 g, 6.5腿 ol)とギ酸アンモ ニゥム (1.73 g, 26腿 ol) のエタノール (100 ml) 懸濁液を 20時間加熱還流し た。 反応中にギ酸アンモニゥム(2.26 g, 33靈 ol)を 5回に分けて加えた。 反応液 を濃縮後酢酸ェチル (180 ml)および水(100 ml)で希釈した。 水層を酢酸ェチル -THF (4:1, 50 ml)で抽出した。 有機層を 10塩化アンモニゥム水溶液および飽和 食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固後、 残渣をメタノールで希 釈した。 析出物を濾取し、 メタノ一ルおよびジェチルエーテルで洗浄後、 メタノ 一ルー酢酸ェチルから結晶化して表題化合物 (1.46 g, 4.32腿。1, 66.3%)を濃紫 色粉末として得た。 A suspension of the crude compound obtained in Example 4-164 (purity 80%, 4.00 g, 6.5 t ol) and ammonium formate (1.73 g, 26 t ol) in ethanol (100 ml) was heated under reflux for 20 hours. Was. During the reaction, ammonium formate (2.26 g, 33 ol) was added in five portions. The reaction solution was concentrated and diluted with ethyl acetate (180 ml) and water (100 ml). The aqueous layer was extracted with ethyl acetate-THF (4: 1, 50 ml). The organic layer was washed with aqueous ammonium chloride solution and saturated saline, dried over sodium sulfate, concentrated to dryness, and the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol and getyl ether, and then crystallized from methyl ethyl acetate to give the title compound (1.46 g, 4.32 g, 1, 66.3%) in dark purple. Obtained as a colored powder.
Ή-NMR (300MHz, CDC13) : (51.60 (8H, brs), 1.73 (3H, s), 1.75 (4H, m), 2.01 (2H, m), 2.26 (3H, q, J= 0.9 Hz), 3.54 (1H, it, J= 9.0, 4.1 Hz), 7.35 (1H, br), 8.43 (1H, br). Ή-NMR (300MHz, CDC1 3 ): (51.60 (8H, brs), 1.73 (3H, s), 1.75 (4H, m), 2.01 (2H, m), 2.26 (3H, q, J = 0.9 Hz) , 3.54 (1H, it, J = 9.0, 4.1 Hz), 7.35 (1H, br), 8.43 (1H, br).
元素分析値: C16H23N30S2として計算値: C, 56.94; H, 6.87; N, 12.45; S, 19.00 {%) 、 実測値: C, 56.66; H, 6.67; N, 12.36; S, 19.03 (%) . Elemental analysis: C 16 H 23 N 30 S 2 Calculated: C, 56.94; H, 6.87; N, 12.45; S, 19.00 (%), found: C, 56.66; H, 6.67; N, 12.36; S, 19.03 (%).
mp. 179.5-181.0°C mp.179.5-181.0 ° C
実施例 6-26 Example 6-26
トァミノ- 5, 6 -ジメチル- 8 - {[2- (2, 2, 2-トリフルォロェトキシ)ェチル]スルファ 二ル}-311-イミダゾ [5,1 - c] [1,4]チアジン - 3-オン Tamino-5,6-dimethyl-8-{[2- (2,2,2-trifluoroethoxy) ethyl] sulfanyl} -311-imidazo [5,1-c] [1,4] thiazine- 3-on
実施例 4-579で得られた N -(5, 6-ジメチル- 3-ォキソ -8- {[2- (2, 2, 2-トリフルォ 口エトキシ)ェチル]スルファニル } - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1- c] [1,4]チア ジン-卜イリデン)-4_メチルベンゼンスルホンアミド (1.08 g, 2.13匪01) とギ 酸アンモニゥム (1.41 g, 21.3腿01) のエタノール (20.0 ml) の懸濁液を 80°C で 16.5時間加熱した後、 水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04)後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (144 mg, 19¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶し て融点 143_145°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-{[2- (2,2,2-trifluoro mouth ethoxy) ethyl] sulfanyl} obtained in Example 4-579} -2,3-dihydro- Ethanol of 1H-imidazo [5,1-c] [1,4] thiazine-triylidene) -4_methylbenzenesulfonamide (1.08 g, 2.13 band01) and ammonium formate (1.41 g, 21.3 thigh01) (20.0 ml) was heated at 80 ° C for 16.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (144 mg, 19¾) as dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 143 to 145 ° C.
IR (KBr) V: 3393, 3289, 3011, 1717, 1667, 1659, 1605, 1539 cm一1. IR (KBr) V: 3393, 3289, 3011, 1717, 1667, 1659, 1605, 1539 cm- 1 .
'H-NMR (CDC13) : 51.73 (3H, d, J=1.2 Hz), 2.26 (3H, d, J=1.2 Hz), 3.12 (2H, t, J=5.7 Hz), 3.85 (2H, t, J=5.1 Hz), 3.88 (2H, q, J=8.4 Hz), 7.16 (2H, bs). 元素分析値: C12H14N302S2F3として 'H-NMR (CDC1 3) : 51.73 (3H, d, J = 1.2 Hz), 2.26 (3H, d, J = 1.2 Hz), 3.12 (2H, t, J = 5.7 Hz), 3.85 (2H, t , J = 5.1 Hz), 3.88 (2H, q, J = 8.4 Hz), 7.16 (2H, bs). Elemental analysis: As C 12 H 14 N 3 0 2 S 2 F 3
計算値 (¾) : C, 40.79; H, 3.99; N, 11.89; S, 18.15. Calculated value (¾): C, 40.79; H, 3.99; N, 11.89; S, 18.15.
実測値 (D : C, 41.07; H, 3.88; N, 11.86; S, 18.01. ' 実施例 6-27 Measured value (D: C, 41.07; H, 3.88; N, 11.86; S, 18.01.'Example 6-27
1-ァミノ - 6 -ェチル- 8 - [ (5-フルォ口ぺンチル)スルファニル] -5-メチル- 3H-ィミ ダゾ [5,1- c] [1,4]チアジン- 3-オン 1-amino-6-ethyl-8-[(5-fluoropentyl) sulfanyl] -5-methyl-3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4-529 で得られた N- {6-ェチル -8- [(5-フルォロペンチル)スルファニ ル] -5-メチル -3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1 , 4]チアジン -トイ リデン }- 4-メチルベンゼンスルホンアミド (810mg, 1.67腿 ol) とギ酸アンモニ ゥム (1.11 g, 16.7匪01) のエタノール (17.0 ml) の懸濁液を 80°Cで 15時間 加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去した。残留物をシリカゲルカラムクロマトグラフィーに付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (316 mg, 58%) を暗 赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 115- 117°Cの暗赤色結晶を得た。 N- {6-ethyl-8-[(5-fluoropentyl) sulfani obtained in Example 4-529 ] -5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-tolylidene} -4-methylbenzenesulfonamide (810 mg, 1.67 t )) And a suspension of ammonium formate (1.11 g, 16.7 band01) in ethanol (17.0 ml) were heated at 80 ° C for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (316 mg, 58%) as dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 115-117 ° C.
IR (KBr) リ: 3403, 3293, 3100, 2965, 2934, 2868, 1715, 1634, 1603, 1539 era—1. Ή-NMR (CDC13) : δ 1.08 (3Η, d, J=7.5 Hz), 1.50-1.62 (4H, m), 1.66-1.84 (4H, m), 2.11 (2H, q, J=7.2 Hz), 2.27 (3H, s), 2.95 (2H, t, J=7.5 Hz), 4.46 (2H, dt, J=47.4, 5.7 Hz), 7.37 (2H, bs). IR (KBr) Re:. 3403, 3293, 3100, 2965, 2934, 2868, 1715, 1634, 1603, 1539 era- 1 Ή-NMR (CDC1 3): δ 1.08 (3Η, d, J = 7.5 Hz), 1.50-1.62 (4H, m), 1.66-1.84 (4H, m), 2.11 (2H, q, J = 7.2 Hz), 2.27 (3H, s), 2.95 (2H, t, J = 7.5 Hz), 4.46 (2H, dt, J = 47.4, 5.7 Hz), 7.37 (2H, bs).
元素分析値: C14H2。N30S2Fとして Elemental analysis: C 14 H 2. N 3 0S 2 F
計算値 (%) : C, 51.04; H, 6.12; N, 12.75; S, 19.47. Calculated value (%): C, 51.04; H, 6.12; N, 12.75; S, 19.47.
実測値 (¾) : C, 50.92; H, 6.05; N, 12.66; S, 19.52. Found (¾): C, 50.92; H, 6.05; N, 12.66; S, 19.52.
実施例 6 - 28 Example 6-28
1 -ァミノ -6 -ブチル 8- [ (5-フルォ口ペンチル)スルファニル] -5 -メチル -3H-ィミ ダゾ [5,1- c] [1,4]チアジン- 3_オン 1-Amino-6-butyl 8-[(5-fluoropentyl) sulfanyl] -5-methyl-3H-imidazo [5,1-c] [1,4] thiazin-3_one
実施例 4-530 で得られた N- {6-ブチル -8- [(5-フルォロペンチル)スルファニ ル]- 5-メチル -3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1 -ィ リデン }-4-メチルベンゼンスルホンアミド (2.19g, 4.28mmol) とギ酸アンモニ ゥム (2.84 g, 4.28 mmol) のエタノール (43.0 ml) の懸濁液を 80 で 15時間 加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去した。残留物をシリカゲルカラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル (10:1, v/y) で溶出して題記化合物 (1.12 g, 74¾) を合 成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 124- 126 の暗赤色結 晶を得た。 N- {6-butyl-8-[(5-fluoropentyl) sulfanyl] -5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] obtained in Example 4-530 ] A suspension of [1,4] thiazine-1-ylidene} -4-methylbenzenesulfonamide (2.19 g, 4.28 mmol) and ammonium formate (2.84 g, 4.28 mmol) in ethanol (43.0 ml) was prepared. After heating at 80 for 15 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / y) to synthesize the title compound (1.12 g, 74¾). Recrystallization from ethyl acetate-getyl ether gave a dark red crystal with a melting point of 124-126.
IR (KBr) V: 3389, 3287, 2963, 2934, 2861, 1717, 1688, 1661, 1603, 1541 cm—1. Ή-NMR (CDCI3) : (50.928 (3H, t, J=7.2 Hz), 1.30-1.64 (6H, m), 1.65-1.84 (4H, m), 2.07 (2H, t, J=6.6 Hz), 2.26 (3H, s), 2.94 (2H, t, J=7.5 Hz), 4.46 (2H, dt, J=47. , 5.7 Hz), 7.48 (2H, bs). IR (KBr) V: 3389, 3287, 2963, 2934, 2861, 1717, 1688, 1661, 1603, 1541 cm— 1. .-NMR (CDCI3): (50.928 (3H, t, J = 7.2 Hz), 1.30 -1.64 (6H, m), 1.65-1.84 (4H, m), 2.07 (2H, t, J = 6.6 Hz), 2.26 (3H, s), 2.94 (2H, t, J = 7.5 Hz), 4.46 (2H, dt, J = 47., 5.7 Hz), 7.48 (2H, bs).
元素分析値: C16H24N30S2Fとして Elemental analysis: as C 16 H 24 N 30 S 2 F
計算値 (¾) : C, 53.75; H, 6.77; N, 11.75; S, 17.94. Calculated (¾): C, 53.75; H, 6.77; N, 11.75; S, 17.94.
実測値 (¾) : C, 53.80; H, 6.70; N, 11.78; S, 17.95. Found (¾): C, 53.80; H, 6.70; N, 11.78; S, 17.95.
実施例 6-29 Example 6-29
トァミノ- 5, 6 -ジメチル- 8- {[5- (2, 2, 2 -トリフルォロエトキシ)ペンチル]スルフ ァニル }- 3H-イミダゾ [5,1-c] [1,4]チアジン -3-オン Toamino-5,6-dimethyl-8-{[5- (2,2,2-trifluoroethoxy) pentyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazine-3 -ON
実施例 4-531で得られた N- (5, 6 -ジメチル -3-ォキソ -8- { [5- (2, 2, 2-トリフルォ 口エトキシ)ペンチル]スルファニル卜 2, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1,4]チ ァジン- 1-ィリデン)-4-メチルベンゼンスルホンアミド (1.01 g, 1.84難 ol) と ギ酸アンモニゥム (1.22g, 18.4匪01) のエタノール (18.0ml) の懸濁液を 80°C で 15.5時間加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (526 mg, 72%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶し て融点 110-112°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-{[5- (2,2,2-trifluoro-mouth ethoxy) pentyl] sulfanyl 2-, 3-dihydroxy- obtained in Example 4-531 Ethanol of 1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene) -4-methylbenzenesulfonamide (1.01 g, 1.84 difficult ol) and ammonium formate (1.22 g, 18.4 bandan 01) (18.0 ml) was heated at 80 ° C. for 15.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (526 mg, 72%) as dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 110-112 ° C.
IR (KBr) ソ : 3407, 3279, 3110, 3048, 2936, 2867, 1713, 1632, 1605, 1547 cm—1.IR (KBr) S: 3407, 3279, 3110, 3048, 2936, 2867, 1713, 1632, 1605, 1547 cm— 1 .
Ή-NMR (CDC13) : 51.40-1.84 (6H, m), 1.74 (3H, s), 2.26 (3H, s), 2.93 (2H, t, J=7.6 Hz), 3.62 (2H, t, J=6.2 Hz), 3.81 (2H, q, J=8.8 Hz), 7.41 (2H, bs). 元素分析値: C15H2flN302S2F3して Ή-NMR (CDC1 3): 51.40-1.84 (6H, m), 1.74 (3H, s), 2.26 (3H, s), 2.93 (2H, t, J = 7.6 Hz), 3.62 (2H, t, J = 6.2 Hz), 3.81 (2H, q, J = 8.8 Hz), 7.41 (2H, bs). Elemental analysis: C 15 H 2fl N 3 0 2 S 2 F 3
計算値 (%) : C, 45.56; H, 5.10; N, 10.63; S, 16.22. Calculated value (%): C, 45.56; H, 5.10; N, 10.63; S, 16.22.
実測値 (¾) : C, 45.68; H, 5.00; N, 10.60; S, 16.43. Found (¾): C, 45.68; H, 5.00; N, 10.60; S, 16.43.
実施例 6 - 30 Example 6-30
トァミノ- 5, 6 -ジメチル -8- {[4 -(2, 2, 2-トリフルォロエトキシ)プチル]スルファ 二ル} - 3H-ィミダゾ [5, 1-c] [1,4]チアジン- 3-ォン Toamino-5,6-dimethyl-8-{[4- (2,2,2-trifluoroethoxy) butyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazine- 3-one
実施例 4-532で得られた N- (5, 6 -ジメチル -3-ォキソ -8 - {[4 -(2, 2, 2-トリフルォ 口エトキシ)プチル]スルファニル }- 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チア ジン-卜イリデン) -4 -メチルベンゼンスルホンアミド (1.12 g, 2.09 mmol) とギ 酸アンモニゥム (1.39 g, 20.9腿01) のエタノール (20.0 ml) の懸濁液を 80°C で 15· 5時間加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04)後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (352 mg, 44%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 108-110°Cの暗赤色結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-{-[4- (2,2,2-trifluoro-mouth ethoxy) butyl] sulfanyl} -2,3-dihydro- obtained in Example 4-532 1H-imidazo [5,1-c] [1,4] thiazine-triylidene) -4-methylbenzenesulfonamide (1.12 g, 2.09 mmol) A suspension of ammonium acid (1.39 g, 20.9 thigh 01) in ethanol (20.0 ml) was heated at 80 ° C for 15.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (352 mg, 44%) as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 108-110 ° C.
IR (KBr) リ: 3387, 2944, 115, 1628, 1605, 541 cm"1. IR (KBr): 3387, 2944, 115, 1628, 1605, 541 cm " 1 .
Ή-NMR (CDC13) : (51.66-1.90 (6H, m), 1.74 (3H, d, J=0.9 Hz), 2.27 (3H, d, J=1.2Hz), 2.97 (2H, t, J=7.5 Hz), 3.65 (2H, t, J=6.0 Hz), 3.82 (2H, q, J=8.4 Hz), 6.94 (2H, bs). Ή-NMR (CDC1 3): (51.66-1.90 (6H, m), 1.74 (3H, d, J = 0.9 Hz), 2.27 (3H, d, J = 1.2Hz), 2.97 (2H, t, J = 7.5 Hz), 3.65 (2H, t, J = 6.0 Hz), 3.82 (2H, q, J = 8.4 Hz), 6.94 (2H, bs).
元素分析値: C14H18N302S2F3して Elemental analysis: C 14 H 18 N 3 0 2 S 2 F 3 to
計算値 (¾) : C, 44.08; H, 4.76; N, 11.02; S, 16.81. Calculated value (¾): C, 44.08; H, 4.76; N, 11.02; S, 16.81.
実測値 (¾) : C, 44.10; H, 4.74; N, 10.88; S, 16.88. Found (¾): C, 44.10; H, 4.74; N, 10.88; S, 16.88.
実施例 6 - 31 Example 6-31
8 - [ (4, 4-ジフルォロシク口へキシル)スルファニル] -卜ィミノ -5, 6-ジメチル -1 , 2 -ジヒドロ- 3H-ィミダゾ [5, 1-c] [1, 4]チアジン- 3 -才ン 8-[(4,4-Difluorocyclohexyl) sulfanyl] -trimino-5,6-dimethyl-1,2, -dihydro-3H-imidazo [5, 1-c] [1,4] thiazine-3- Talent
実施例 4-535で得た化合物 (212 mg, 0.424匪 ol) とギ酸アンモニゥム Q41 mg, 2.12 mmol) のエタノール (7 ml) 溶液を 12 時間加熱還流した。 反応液を濃縮 乾固後、 酢酸ェチル (25 ml)で希釈し、 水および飽和食塩水で洗浄後、 硫酸ナト リウムで乾燥した。 濃縮乾固後、 残渣をシリカゲルクロマトグラフィー (溶出溶 媒、酢酸ェチルーへキサン、 5:5-8:2)に付し、酢酸ェチル—へキサン(7:3- 8:2) の 画分を濃縮乾固後、 酢酸ェチルおよびジェチルェ一テルで希釈した。 析出物を濾 取し、 ジェチルエーテルで洗浄後、 乾燥して表題化合物 (115 mg, 0.333匪 ol, 78.5%) を赤褐色の粉末として得た。 A solution of the compound obtained in Example 4-535 (212 mg, 0.424 bandol) and ammonium formate Q41 mg, 2.12 mmol) in ethanol (7 ml) was heated under reflux for 12 hours. The reaction solution was concentrated to dryness, diluted with ethyl acetate (25 ml), washed with water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was subjected to silica gel chromatography (eluent, ethyl acetate-hexane, 5: 5-8: 2), and the ethyl acetate-hexane (7: 3-8: 2) fraction was obtained. After concentration to dryness, the mixture was diluted with ethyl acetate and ethyl ether. The precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (115 mg, 0.333 ol, 78.5%) as a red-brown powder.
¾ - NMR (300MHz, CDC13) : (51.73 (3H, s), 1.82 (4H, m), 2.19 (4H, brd, J= 9.2 ¾ - NMR (300MHz, CDC1 3 ): (51.73 (3H, s), 1.82 (4H, m), 2.19 (4H, brd, J = 9.2
Hz), 2.26 (3H, brq, J= 0.8 Hz), 3.37 (1H, m), 7.30 (2H, br). Hz), 2.26 (3H, brq, J = 0.8 Hz), 3.37 (1H, m), 7.30 (2H, br).
元素分析値: C14H17N30S2F2として計算値: C, 48.68; H, 4.96; N, 12.16; S, 18.57Elemental analysis: C 14 H 17 N 3 0S 2 F 2 Calculated: C, 48.68; H, 4.96 ; N, 12.16; S, 18.57
(%) 、 実測値: C, 48.95; H, 4.66; N, 12.12; S, 18.82 (%) . (%), Found: C, 48.95; H, 4.66; N, 12.12; S, 18.82 (%).
mp. 203.0-204. O : 実施例 6-32 mp. 203.0-204.O: Example 6-32
8- {[(4, 4-ジフルォロシク口へキシル)メチル]スルファニル } -卜ィミノ- 5, 6-ジメ チル- 1,2-ジヒドロ- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3 -オン 8-{[(4,4-difluorocyclohexyl) methyl] sulfanyl} -trimino-5,6-dimethyl-1,2-dihydro-3H-imidazo [5, 1-c] [1,4] Thiazine-3-one
実施例 4- 536で得た化合物 (468 mg, 0.911 mmol) とギ酸アンモニゥム (302 mg, 4.56顧 ol) のエタノール (12ml) 溶液を 48 時間加熱還流した。反応中に THF (3 ml)およびギ酸アンモニゥム(270 mg, 4.07 mmol)を加えた。反応液を濃縮乾固後、 酢酸ェチル (40 ml)で希釈し、 水 および飽和食塩水で洗浄後、 硫酸ナトリウムで 乾燥した。 濃縮乾固後、 残渣をメタノールで希釈した。 析出物を濾取し、 メタノ ールで洗浄した。 濾液および洗液合わせてシリカゲルクロマトグラフィー (溶出 溶媒、酢酸ェチルーへキサン、 5:5-9:1)に付し、酢酸ェチルーへキサン(8:2- 9:1) の画分を濃縮乾固後、 ジェチル: ίΐ一テルで希釈した。 析出物を濾取し、 ジェチル エーテルで洗浄後、 乾燥して表題化合物 α 05 mg, 0.2'92 mmol, 32.1 )を灰赤色の 粉末として得た。 A solution of the compound obtained in Example 4-536 (468 mg, 0.911 mmol) and ammonium formate (302 mg, 4.56 mmol) in ethanol (12 ml) was heated under reflux for 48 hours. During the reaction, THF (3 ml) and ammonium formate (270 mg, 4.07 mmol) were added. The reaction mixture was concentrated to dryness, diluted with ethyl acetate (40 ml), washed with water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration and washed with methanol. The combined filtrate and washings were subjected to silica gel chromatography (elution solvent, ethyl acetate-hexane, 5: 5-9: 1), and the ethyl acetate-hexane (8: 2-9: 1) fraction was concentrated to dryness. Later, Jechil: diluted with 1 liter. The precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (α05 mg, 0.2'92 mmol, 32.1) as a gray-red powder.
¾— NMR (300MHz, CDC13) : 51.38 (2H, brq, J= 13.2 Hz), 1.71 (2H, m), 1.74 (3H, s), 1.82 (4H, m), 1.95 (2H, brd, J= 13.2 Hz), 2.13 (2H, m), 2.26 (3H, s), ¾- NMR (300MHz, CDC1 3) : 51.38 (2H, brq, J = 13.2 Hz), 1.71 (2H, m), 1.74 (3H, s), 1.82 (4H, m), 1.95 (2H, brd, J = 13.2 Hz), 2.13 (2H, m), 2.26 (3H, s),
2.88 (2H, d, J= 7.2 Hz), 7.30 (1H, br), 8.88 (1H, br) . 2.88 (2H, d, J = 7.2 Hz), 7.30 (1H, br), 8.88 (1H, br).
元素分析値: C15H19N30S2F2として計算値: C, 50.12; H, 5.33; N, 11.69; S, 17.84 (%) 、 実測値: C, 50.31; H, 5.19; N, 11.70; S, 17.74 (%) . Elemental analysis: C 15 H 19 N 3 0S 2 F 2 Calculated: C, 50.12; H, 5.33 ; N, 11.69; S, 17.84 (%), Found: C, 50.31; H, 5.19 ; N, 11.70; S, 17.74 (%).
mp. 175.5-177.0°C mp. 175.5-177.0 ° C
実施例 6-33 Example 6-33
卜ァミノ- 5, 6-ジメチル- 8- { [2- (4-フルォロブトキシ)ェチル]スルファ二ル} -3H- イミダゾ [5,1 - c] [1,4]チアジン - 3 -オン Triamino-5,6-dimethyl-8-{[2- (4-fluorobutoxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazine-3-one
実施例 18-23 で得られた N- (5,6-ジメチル -8-{[2- (4-フルォロブトキシ)ェチ ル]スルファニル }- 3-ォキソ -2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン -卜イリデン) -4-メチルベンゼンスルホンアミド (290 mg, 0.580 mmol) とギ酸ァ ンモニゥム (385 mg, 5.80腿01) のエタノール (5.80 ml) の懸濁液を 80°Cで 16 時間加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (100 mg, 50¾) を不定形晶として得た。 N- (5,6-Dimethyl-8-{[2- (4-fluorobutoxy) ethyl] sulfanyl) obtained in Example 18-23} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-triylidene) -4-methylbenzenesulfonamide (290 mg, 0.580 mmol) and ammonium formate (385 mg, 5.80 thigh 01) in ethanol (5.80 ml) After heating the suspension at 80 ° C for 16 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (100 mg, 50¾) Was obtained as amorphous crystals.
IR (KBr) リ: 3403, 3287, 3106, 2965, 2868, 1715, 1636, 1607, 1545 cm"1. 'Η—醒 R (CDC13) : (51.67-1.92 (4H, m), 1.72 (3H, d, J=1.2 Hz), 2.25 (3H, d, J=l.2 Hz), 3.08 (2H, t, J=5.7Hz), 3.51 (2H, t, J=6.3 Hz), 3.66 (2H, t, J=5.7 Hz), 4.47 (2H, dt, J=47. , 5.7 Hz), 7.50 (2H, bs). IR (KBr) Re:. 3403, 3287, 3106, 2965, 2868, 1715, 1636, 1607, 1545 cm "1 'Η- Awakening: R (CDC1 3): (51.67-1.92 (4H, m), 1.72 (3H , d, J = 1.2 Hz), 2.25 (3H, d, J = 1.2 Hz), 3.08 (2H, t, J = 5.7 Hz), 3.51 (2H, t, J = 6.3 Hz), 3.66 (2H , t, J = 5.7 Hz), 4.47 (2H, dt, J = 47., 5.7 Hz), 7.50 (2H, bs).
実施例 6 - 34 Example 6-34
卜ァミノ -5,6-ジメチル -8- {[2- (5 -フルォロペンチルォキシ)ェチル]スルファニ ル }-3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3 -オン Triamino-5,6-dimethyl-8-{[2- (5-fluoropentyloxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazine-3- On
実施例 4-534で得られた N- (5, 6-ジメチル -8- { [2- (5-フルォロペンチルォキシ) ェチル]スルファニル卜 3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジ ン-卜イリデン) -4-メチルベンゼンスルホンアミド (1.03 g, 2.01 腿 ol) とギ酸 アンモニゥム (1.36 g, 20.6讓01) のエタノール (20.0 ml) の懸濁液を 80 で 16.5 時間加熱した後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトダラ フィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (427 mg, 55¾) を不定形晶として得た。 N- (5,6-dimethyl-8-{[2- (5-fluoropentyloxy) ethyl] sulfanilto 3-oxo-2,3-dihydro-1H- obtained in Example 4-534 Imidazo [5,1-c] [1,4] thiazin-triylidene) -4-methylbenzenesulfonamide (1.03 g, 2.01 thigh ol) and ammonium formate (1.36 g, 20.6 seq.) In ethanol (20.0 ml) ) Was heated at 80 for 16.5 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (427 mg, 55¾) as amorphous crystals.
IR (KBr) リ: 3389, 3285, 2942, 2865, 1715, 1651, 1605, 1543 cm— IR (KBr): 3389, 3285, 2942, 2865, 1715, 1651, 1605, 1543 cm—
'H-NMR (CDC13) : 51.34-1.85 (6H, m), 1.72 (3H, d, J=0.8 Hz), 2.25 (3H, d, J=1.0 Hz), 3.07 (2H, t, J=5.8 Hz), 3.47 (2H, t, J=6.6 Hz), 3.65 (2H, t, J=5.8 Hz), 4.45 (2H, dt, J=47.2, 5.8 Hz), 7.94 (2H, bs). 'H-NMR (CDC1 3) : 51.34-1.85 (6H, m), 1.72 (3H, d, J = 0.8 Hz), 2.25 (3H, d, J = 1.0 Hz), 3.07 (2H, t, J = 5.8 Hz), 3.47 (2H, t, J = 6.6 Hz), 3.65 (2H, t, J = 5.8 Hz), 4.45 (2H, dt, J = 47.2, 5.8 Hz), 7.94 (2H, bs).
実施例 6-35 Example 6-35
5 - [卜ァミノ- 5,6-ジメチル- 3-ォキソ -3H-イミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィ ル]スルファニル] -N-へキシルペンタンアミド 5-[Toramino-5,6-dimethyl-3-oxo-3H-imidazo [5,1-c] [1,4] thiazine-8-yl] sulfanyl] -N-hexylpentanamide
実施例 4-528で得られた 5_[(5,6_ジメチル- 1-{[(4 -メチルフエニル)スルホ二 ル]イミノ}- 3-ォキソ - 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィル) スルファニル]- N-へキシルペンタンアミド (520 mg, 0.921 匪 ol) とギ酸アンモ ニゥム (611 mg, 9.21 匪 ol) のエタノール (9.20 ml) の懸濁液を 80°Cで 21時 間加熱した後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (201 nig, 53¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルェ一テルから再結晶して融点 142- 144°Cの暗赤色結晶を得た。 5 _ [(5,6_dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, obtained in Example 4-528 1-c] [1,4] thiazin-8-yl) sulfanyl] -N-hexylpentanamide (520 mg, 0.921 bandol) and ammonium formate (611 mg, 9.21 bandol) in ethanol (9.20 ml) ) Was heated at 80 ° C for 21 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography. After elution with n-hexane-ethyl acetate (10: 1, v / v), the title compound (201 nig, 53) was obtained as dark red crystals. Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 142-144 ° C.
IR (KBr) V 3385, 3287, 3058, 2928, 2859, 1705, 1642, 1605, 1545 cm"1.IR (KBr) V 3385, 3287, 3058, 2928, 2859, 1705, 1642, 1605, 1545 cm " 1 .
Ή-NMR (CDC13) : 50.885 (3H, t, J=6.9Hz), 1.22-1.36 (6H, m), 1.43-1.56 (2H, m), 1.70-1.85 (4H, m), 1.73 (3H, d, J=0.9 Hz), 2.20 (2H, t, J=6.6 Hz), 2.26 Ή-NMR (CDC1 3): 50.885 (3H, t, J = 6.9Hz), 1.22-1.36 (6H, m), 1.43-1.56 (2H, m), 1.70-1.85 (4H, m), 1.73 (3H , d, J = 0.9 Hz), 2.20 (2H, t, J = 6.6 Hz), 2.26
(3H, d, J=0.9 Hz), 2.94 (2H, t, J=6.0 Hz), 3.24 (2H, q, J=6.0 Hz), 5.56 (1H, bs), 7.46 (2H, bs). (3H, d, J = 0.9 Hz), 2.94 (2H, t, J = 6.0 Hz), 3.24 (2H, q, J = 6.0 Hz), 5.56 (1H, bs), 7.46 (2H, bs).
元素分析値: 。 N402S2 ·0.2Η20として Elemental analysis:. N 4 0 2 S 2 0.2Η 20
計算値 (%) : C, 55.10; H, 7.40; N, 13.53; S, 15.48. Calculated (%): C, 55.10; H, 7.40; N, 13.53; S, 15.48.
実測値 (¾) : C, 55.06; H, 7.27; N, 13.70; S, 15.46. Found (値): C, 55.06; H, 7.27; N, 13.70; S, 15.46.
実施例 6 - 36 Example 6-36
1 -アミノ- 5, 6-ジメチル- 8- { [2- (3-フルォロプロポキシ)ェチル]スルファニ M - 3H-ィミダゾ [5 ,卜 c] [1 , 4]チアジン- 3 -才ン 1-amino-5,6-dimethyl-8-{[2- (3-fluoropropoxy) ethyl] sulfani M-3H-imidazo [5, tric] [1,4] thiazine-3-
実施例 4-533で得られた N-(5, 6-ジメチル -8- {[2- (3-フルォロプロボキシ)ェチ ル]スルファニル }_3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン -卜イリデン) -4-メチルベンゼンスルホンアミド (370 mg, 0.762 mmol) とギ酸ァ ンモニゥム (505 mg, 7.62腿01) のエタノール (7.50 ml) の懸濁液を 80°Cで 15 時間加熱した後、水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (181 mg, 72%) を不定形晶として得た。 N- (5,6-Dimethyl-8-{[2- (3-fluoropropoxy) ethyl] sulfanyl} _3-oxo-2,3-dihydro-1H obtained in Example 4-533 -Imidazo [5,1-c] [1,4] thiazine-triylidene) -4-methylbenzenesulfonamide (370 mg, 0.762 mmol) and ammonium formate (505 mg, 7.62 thigh 01) in ethanol (7.50 ml) ) Was heated at 80 ° C for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (181 mg, 72%) as amorphous crystals.
IR (KBr) V: 3391, 3289, 3113, 3038, 2969, 2932, 2870, 1715, 1651, 1634, 1605, 1543 cm"1. IR (KBr) V: 3391, 3289, 3113, 3038, 2969, 2932, 2870, 1715, 1651, 1634, 1605, 1543 cm " 1 .
Ή-NMR (CDCI3) : 51.73 (3H, d, J=0.9 Hz), 1.97 (2H, dt, J=27.0, 6.0 Hz), 2.25 (3H, d, J=0.9 Hz), 3.08 (2H, t, J=6.0 Hz), 3.60 (2H, t, J=6.0 Hz), 4.45 (2H, dt, J=47.4, 6.0 Hz), 7.50 (2H, bs). Ή-NMR (CDCI3): 51.73 (3H, d, J = 0.9 Hz), 1.97 (2H, dt, J = 27.0, 6.0 Hz), 2.25 (3H, d, J = 0.9 Hz), 3.08 (2H, t , J = 6.0 Hz), 3.60 (2H, t, J = 6.0 Hz), 4.45 (2H, dt, J = 47.4, 6.0 Hz), 7.50 (2H, bs).
実施例 6_38 Example 6_38
tert -ブチル 3 - [(卜アミノ- 5, 6 -ジメチル _3-ォキソ - 3H-イミダゾ [5,1- c] [1,4] チアジン -8-ィル)スルファニル]プロピル力一バメート tert-Butyl 3-[(triamino-5,6-dimethyl_3-oxo-3H-imidazo [5,1-c] [1,4] Thiazine-8-yl) sulfanyl] propyl
実施例 4-106と同様にして得た化合物 (1.22 g, 2.26腿01) とギ酸アンモニゥ ム (752 mg, 11.3mmol) のエタノール (25 ml) 懸濁液を 23時間加熱還流した。 反応中にギ酸アンモニゥム(300 mg, 4.52匪 ol)を加えた。 反応液を濾過し、 エタ ノールで洗浄後、 乾燥して表題化合物(318 mg, 0.827腿 ol, 36.6%)を紫色粉末と して得た。 母液を濃縮後、 酢酸ェチル (30ml)および水(30ml)で希釈した。 析出 物を濾取し、 メタノール一水 (1:1)、 ジェチルェ一テルおよび酢酸ェチルで洗浄 後、 乾燥して表題化合物(287 mg, 0.746匪 ol, 33.0%)を灰紫色粉末として得た。 'H-NMR (200MHz, CDC13) : 51.45 (9H, s), 1.73 (3H, brs), 1.90 (2H, quint, J= 7.0 Hz), 2.26 (3H, s), 2.96 (2H, t, J= 7.3 Hz), 3.26 (2H, q, J= 6.4 Hz), 4.69 (1H, m), 7.42 (2H, br). A suspension of the compound (1.22 g, 2.26 thigh 01) obtained in the same manner as in Example 4-106 and ammonium formate (752 mg, 11.3 mmol) in ethanol (25 ml) was heated under reflux for 23 hours. During the reaction, ammonium formate (300 mg, 4.52 ol) was added. The reaction solution was filtered, washed with ethanol, and dried to give the title compound (318 mg, 0.827 mol, 36.6%) as a purple powder. After concentration, the mother liquor was diluted with ethyl acetate (30 ml) and water (30 ml). The precipitate was collected by filtration, washed with methanol / water (1: 1), getyl ether, and ethyl acetate, and dried to give the title compound (287 mg, 0.746 bandol, 33.0%) as a gray-violet powder. 'H-NMR (200MHz, CDC1 3): 51.45 (9H, s), 1.73 (3H, brs), 1.90 (2H, quint, J = 7.0 Hz), 2.26 (3H, s), 2.96 (2H, t, J = 7.3 Hz), 3.26 (2H, q, J = 6.4 Hz), 4.69 (1H, m), 7.42 (2H, br).
元素分析値: C16H24N403S2として計算値: C, 49.98; H, 6.29; N, 14.57; S, 16.68Elemental analysis: C 16 H 24 N 4 0 3 S 2 Calculated: C, 49.98; H, 6.29 ; N, 14.57; S, 16.68
(%) 、 実測値: C, 49.89; H, 6.19; N, 14.59; S, 16.57 ( ) . (%), Found: C, 49.89; H, 6.19; N, 14.59; S, 16.57 ().
即. 197.5-198.5°C Immediately. 197.5-198.5 ° C
実施例 6-39 Example 6-39
N- {3- [(卜ァミノ- 5, 6-ジメチル -3-ォキソ - 3H-イミダゾ [5, 1-c] [1,4] チアジン -8-ィル)スルファニル] プロピル }へキサンアミド N- {3-[(Tamino-3,6-dimethyl-3-oxo-3H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] propyl} hexanamide
実施例 18- 13で得た化合物 (520 mg, 0.969 mmol)とギ酸アンモニゥム(322 mg, 4.84腿 ol)のエタノール (10 ml) 懸濁液を 15時間加熱還流した。 反応中にギ酸 アンモニゥム(95mg, 1.43mmol)を加えた。 反応液を濃縮後、 酢酸ェチル (40 ml) で希釈し、 5%炭酸水素ナトリウム、 5%食塩水、 および飽和食塩水で洗浄した。 析 出物を濾取し、 水、 メタノール水および酢酸ェチルで洗浄後、 乾燥して表題化合 物(88mg, 0.230腿 ol, 23.7%)を灰紫色粉末として得た。 濾液の有機層を分離し、 硫酸ナトリウムで乾燥後、 濃縮し、 残渣を酢酸ェチルで希釈した。 析出物を濾取 し、 酢酸ェチルおよびジェチルエーテルで洗浄後、 乾燥して表題化合物(153 mg, 0.400腿 ol, 41.3%)を灰紫色粉末として得た。 A suspension of the compound obtained in Examples 18 to 13 (520 mg, 0.969 mmol) and ammonium formate (322 mg, 4.84 mol) in ethanol (10 ml) was heated under reflux for 15 hours. During the reaction, ammonium formate (95 mg, 1.43 mmol) was added. The reaction solution was concentrated, diluted with ethyl acetate (40 ml), and washed with 5% sodium hydrogen carbonate, 5% saline, and saturated saline. The precipitate was collected by filtration, washed with water, aqueous methanol and ethyl acetate, and dried to give the title compound (88 mg, 0.230 mol, 23.7%) as a gray-violet powder. The organic layer of the filtrate was separated, dried over sodium sulfate and concentrated, and the residue was diluted with ethyl acetate. The precipitate was collected by filtration, washed with ethyl acetate and getyl ether, and dried to give the title compound (153 mg, 0.400 ml, 41.3%) as a gray-violet powder.
¾-NMR (200MHz, CDC13) : (50.89 (3H, t, J= 6.6 Hz), 1.30 (4H, ), 1.63 (2H, brquint, 1= 7.7 Hz), 1.73 (3H, q, J= 1.2 Hz), 2.18 (2H, t, J= 7.5 Hz), 2.25 (3H, q, J= 1.0 Hz), 2.95 (2H, i, J= 7.1 Hz), 3.38 (2H, q, J= 6.5 Hz), 6.02 (1H, m), 7.76 (2H, br). ¾-NMR (200MHz, CDC1 3 ): (50.89 (3H, t, J = 6.6 Hz), 1.30 (4H,), 1.63 (2H, brquint, 1 = 7.7 Hz), 1.73 (3H, q, J = 1.2 Hz), 2.18 (2H, t, J = 7.5 Hz), 2.25 (3H, q, J = 1.0 Hz), 2.95 (2H, i, J = 7.1 Hz), 3.38 (2H, q, J = 6.5 Hz) , 6.02 (1H, m), 7.76 (2H, br).
元素分析値: C17H26N402S2として計算値: C, 53.37; H, 6.85; N, 14.65; S, 16.76Elemental analysis: C 17 H 26 N 4 0 2 S 2 Calculated: C, 53.37; H, 6.85 ; N, 14.65; S, 16.76
(%) 、 実測値 (%) : C, 53.25; H, 6.99; N, 14.71; S, 16.68. (%), Found (%): C, 53.25; H, 6.99; N, 14.71; S, 16.68.
mp. 162.0-163.5°C mp.162.0-163.5 ° C
実施例 6 - 40 Example 6-40
8- {[2- (1 -ァダマンチル)ェチル]スルファ二ル}- 1 -ィミノ -5, 6-ジメチル -1, 2 -ジ ヒドロ- 3H-ィミダゾ [5, 1-c] [1,4]チアジン -3-オン 8-{[2- (1-Adamantyl) ethyl] sulfanyl} -1-imino-5,6-dimethyl-1,2-dihydro-3H-imidazo [5,1-c] [1,4] Thiazine-3-one
実施例 4- 553で得た化合物 (1.96g, 3.60mmol)とギ酸アンモニゥム (1.20g, 18.0mmol) のエタノール (50ml) 懸濁液を 26時間加熱還流した。 反応中にギ酸 アンモニゥム(1.20 g, 18,0 mmol)を加えた。 反応液を濃縮後、 酢酸ェチル - THF (5:1)で抽出した。有機層を 10塩化アンモニゥム水溶液および飽和食塩水で洗浄 後、 硫酸ナトリウムで乾燥し、 濃縮乾固後、 残渣をメタノールで希釈した。 析出 物を濾取し、 メタノールで洗浄後、 メタノール—酢酸ェチルから結晶化して表題 化合物(983 mg, 2.52讓01,' 70.1»を濃紫色粉末として得た。 A suspension of the compound (1.96 g, 3.60 mmol) obtained in Example 4-553 and ammonium formate (1.20 g, 18.0 mmol) in ethanol (50 ml) was heated under reflux for 26 hours. During the reaction, ammonium formate (1.20 g, 18,0 mmol) was added. After the reaction solution was concentrated, it was extracted with ethyl acetate-THF (5: 1). The organic layer was washed with aqueous ammonium chloride solution and saturated saline, dried over sodium sulfate, concentrated to dryness, and the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and then crystallized from methanol-ethyl acetate to give the title compound (983 mg, 2.52 alcohol 01, '70 .1 ») as a dark purple powder.
— NMR (300MHz, CDC13): (51.45 (2H, m), 1.51 (6H, d, J= 2.4 Hz), 1.63 (3H, brd, J= 11.7 Hz), 1.72 (3H, brd, J= 12 Hz), 1.75 (3H, q, J= 0.9 Hz), 1.98 (3H, brs), 2.27 (3H, q, J= 0.9 Hz), 2.88 (2H, m), 7.56 (2H, br). - NMR (300MHz, CDC1 3) : (51.45 (2H, m), 1.51 (6H, d, J = 2.4 Hz), 1.63 (3H, brd, J = 11.7 Hz), 1.72 (3H, brd, J = 12 Hz), 1.75 (3H, q, J = 0.9 Hz), 1.98 (3H, brs), 2.27 (3H, q, J = 0.9 Hz), 2.88 (2H, m), 7.56 (2H, br).
元素分析値: C2。H27N30S2 ·0.2Η20として計算値: C, 60.82; Η, 7.04; Ν, 10.64; S, 16.24 (%) 、 Elemental analysis: C 2. H 27 N 3 0S 2 · 0.2Η 2 0 Calculated: C, 60.82; Η, 7.04 ; Ν, 10.64; S, 16.24 (%),
元素分析値 C20H27N3OS2 ·0.漏 Acとして計算値: C, 61.35; H, 7.08; N, 10.32; S, 15.75 (%) 、 Elemental analysis: C 20 H 27 N 3 OS 2 · 0. Calculated as leak Ac: C, 61.35; H, 7.08; N, 10.32; S, 15.75 (%),
実測値: C, 61.03; H, 6.89; N, 10.29; S, 15.58 (%) . Found: C, 61.03; H, 6.89; N, 10.29; S, 15.58 (%).
mp. 184.0- 185.5 mp. 184.0- 185.5
実施例 6-41 Example 6-41
8- [ ( アダマンチルメチル)スルファニル] -トイミノ -5, 6-ジメチル- 1, 2-ジヒド ロ- 3H-イミダゾ [5,1- c] [1,4]チアジン- 3-オン 8-[(adamantylmethyl) sulfanyl] -tomino-5,6-dimethyl-1,2-dihydro-3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4- 554で得た化合物(875 mg, 1.65 mmol)とギ酸アンモニゥム (548 mg, 8.26 mmol) のエタノール (20 ml) 懸濁液を 35時間加熱還流した。 反応中にギ酸 アンモニゥム(0.88 g, 13.2醒 ol)を 2回に分けて加えた。 反応液を濃縮後、 酢酸 ェチル - THF (5:1)で抽出した。 有機層を 10塩化アンモニゥム水溶液および飽和 食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固して濃紫色油状物を得た。 これをシリカゲル(10 g)のクロマトグラフィ一(酢酸ェチル一へキサン、 7:3-8: 2) で精製し、 THF—酢酸ェチルから結晶化して表題化合物 (240 mg, 0.639 廳 ol, 38.7%)を濃紫色粉末として得た。 A suspension of the compound (875 mg, 1.65 mmol) obtained in Example 4-554 and ammonium formate (548 mg, 8.26 mmol) in ethanol (20 ml) was heated under reflux for 35 hours. During the reaction, ammonium formate (0.88 g, 13.2 ol) was added in two portions. After concentrating the reaction solution, acetic acid Extracted with ethyl-THF (5: 1). The organic layer was washed with an aqueous solution of ammonium chloride 10 and saturated saline, dried over sodium sulfate, and concentrated to dryness to obtain a dark purple oil. This was purified by chromatography on silica gel (10 g) (ethyl acetate, hexane, 7: 3-8: 2) and crystallized from THF-ethyl acetate to give the title compound (240 mg, 0.639 ol, 38.7%) Was obtained as a deep purple powder.
'H-NMR (200MHz, CDC13) : (51.60 (6H, brs), 1.63 (3H, brd, J= 12 Hz), 1.72 (3H, brd, J= 12 Hz), 1.75 (3H, q, J= 1.0 Hz), 2.02 (3H, brs), 2.27 (3H, q, J= 0.6 Hz), 2.79 (2H, s), 7.38 (2H, br). 'H-NMR (200MHz, CDC1 3): (51.60 (6H, brs), 1.63 (3H, brd, J = 12 Hz), 1.72 (3H, brd, J = 12 Hz), 1.75 (3H, q, J = 1.0 Hz), 2.02 (3H, brs), 2.27 (3H, q, J = 0.6 Hz), 2.79 (2H, s), 7.38 (2H, br).
元素分析値: C19H25N30S2として計算値: C, 60.76; H, 6.71; , 11.19; S, 17.08 (%) 、 実測値: C, 60.58; H, 6.76; N, 10.84; S, 16.83 (%) . Elemental analysis: C 19 H 25 N 30 S 2 Calculated: C, 60.76; H, 6.71;, 11.19; S, 17.08 (%), found: C, 60.58; H, 6.76; N, 10.84; S , 16.83 (%).
即. 199.0- 201. (T Immediately. 199.0- 201. (T
実施例 6-42 Example 6-42
1-Tミノ -8- [(シク口才クチルメチル)スルファニル] -5, 6-ジメチル- 3H-ィミダゾ 1-T-mino-8-[(Six-octylmethyl) sulfanyl] -5,6-dimethyl-3H-imidazo
[5, 1-c] [1, 4]チアジン - 3-オン [5, 1-c] [1, 4] thiazine-3-one
実施例 4- 94 で得た化合物(1.95 g, 3.86腿 ol)とギ酸アンモニゥム (1.28 g, The compound obtained in Example 4-94 (1.95 g, 3.86 tmol) and ammonium formate (1.28 g,
19.3mmol) のエタノール,(40 ml) 懸濁液を 22時間加熱還流した。 反応中にギ酸 アンモニゥム(1.28 g, 19.3 IM01)を加えた。 反応液を濃縮後、 残渣をメタノール で希釈し、 析出物を濾取し、 メタノールで洗浄後、 乾燥して表題化合物(936 mg,A suspension of 19.3 mmol) in ethanol (40 ml) was heated under reflux for 22 hours. During the reaction, ammonium formate (1.28 g, 19.3 IM01) was added. After concentrating the reaction solution, the residue was diluted with methanol, and the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (936 mg,
2.66 mmol, 69.0%)を紫色粉末として得た。 2.66 mmol, 69.0%) as a purple powder.
'H-NMR (200MHz, CDC13) : δ 1.42 (2Η, brq, J= 10.4 Hz), 1.40-1.80 (13H, m),'H-NMR (200MHz, CDC1 3): δ 1.42 (2Η, brq, J = 10.4 Hz), 1.40-1.80 (13H, m),
1.74 (3H, q, J= 1.0 Hz), 2.27 (3H, q, J= 1.0 Hz), 2.84 (2H, d, J= 6.6 Hz),1.74 (3H, q, J = 1.0 Hz), 2.27 (3H, q, J = 1.0 Hz), 2.84 (2H, d, J = 6.6 Hz),
7.54 (2H, br). 7.54 (2H, br).
元素分析値: C17H25N3OS2として計算値: C, 58.08; H, 7.17; N, 11.95; S, 18.24 (%) 、 実測値: C, 57.80; H, 7.47; N, 11.74; S, 18.18 ( ) · Elemental analysis: C 17 H 25 N 3 OS 2 Calculated: C, 58.08; H, 7.17 ; N, 11.95; S, 18.24 (%), Found: C, 57.80; H, 7.47 ; N, 11.74; S, 18.18 ()
即. 190.0-191.5°C Immediate. 190.0-191.5 ° C
実施例 6-43 Example 6-43
1 -ァミノ- 5,6-ジメチル- 8_{[2- (2 -フルォロエトキシ)ェチル]スルファニル }-3H- イミダゾ [5, 1-c] [1,4]チアジン- 3-オン 1-Amino-5,6-dimethyl-8 _ {[2- (2-fluoroethoxy) ethyl] sulfanyl} -3H-imidazo [5, 1-c] [1,4] thiazin-3-one
実施例 4-580で得られた N- (5,6-ジメチル- 3-ォキソ -8- {[2- (2-フルォロェトキ シ)ェチル]スルファニル } - 2, 3-ジヒド口 1H-ィミダゾ [5, 1丄 c] [1, ]チアジン- 1- イリデン) -4-メチルベンゼンスルホンアミド (770 mg, 1.63 mmol) とギ酸アンモ ニゥム (l,08 g, 16.3誦 ol) のエタノール (16.0 ml) の懸濁液を 80°Cで U時 間加熱した後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (313 mg, 61%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 120-122 :の暗赤色結晶を得た。 N- (5,6-dimethyl-3-oxo-8-{[2- (2-fluoroethoxy) obtained in Example 4-580 C) Ethyl] sulfanyl}-2,3-dihydric 1H-imidazo [5,1 丄 c] [1,] thiazine-1-ylidene) -4-methylbenzenesulfonamide (770 mg, 1.63 mmol) and ammonium formate A suspension of ethanol (l, 08 g, 16.3 ol) in ethanol (16.0 ml) was heated at 80 ° C for U hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (313 mg, 61%) as dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 120-122.
IR (KBr) V: 3385, 3283, 3121, 2976, 2932, 1715, 1634, 1607 cm"1. IR (KBr) V: 3385, 3283, 3121, 2976, 2932, 1715, 1634, 1607 cm " 1 .
¾-NMR (CDC13) : (51.72 (3H, s), 2.26 (3H, s), 3.09 (2H, t, J=5.8 Hz), 3.73 ¾-NMR (CDC1 3): (51.72 (3H, s), 2.26 (3H, s), 3.09 (2H, t, J = 5.8 Hz), 3.73
(2H, dt, J=31.6, 3.6 Hz), 3.74 (2H, t, J=5.8 Hz), 4.57 (2H, dt, J=47.6, 4.0(2H, dt, J = 31.6, 3.6 Hz), 3.74 (2H, t, J = 5.8 Hz), 4.57 (2H, dt, J = 47.6, 4.0
Hz), 7.40 (2H, bs). Hz), 7.40 (2H, bs).
元素分析値: C12H16N302S2Fとして Elementary analysis: as C 12 H 16 N 3 0 2 S 2 F
実施例 6-44 Example 6-44
卜ァミノ -5, 6-ジメチル -8- ({ [ (2S) -5 -ォキソピ口リジニル]メチル }スルファニ ル) - 3H-イミダゾ [5, 1-c] [1,4]チアジン - 3 -オン Triamino-5,6-dimethyl-8-({[(2S) -5-oxopiridinyl] methyl} sulfanyl) -3H-imidazo [5,1-c] [1,4] thiazine-3-one
実施例 4- 557で得た化合物 (660 mg, 1.38歷 ol)とギ酸アンモニゥム (458 mg, 6.89 mmol) のエタノール (14ml) 懸濁液を 22時間加熱還流した。 反応中にギ酸 アンモニゥム (275 mg, 4.14 mmol)を加えた。 反応液を濃縮後、 残渣をメタノール で希釈した。析出物を濾取し、 メタノール一水(1:1)、 メタノールおよびジェチル エーテルで洗浄後、 乾燥して表題化合物(184 mg, 0.567 imol, 41.1%)を紫色粉末 として得た。 A suspension of the compound (660 mg, 1.38 mol) obtained in Example 4-557 and ammonium formate (458 mg, 6.89 mmol) in ethanol (14 ml) was heated under reflux for 22 hours. During the reaction, ammonium formate (275 mg, 4.14 mmol) was added. After concentration of the reaction solution, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol / water (1: 1), methanol and getyl ether, and dried to give the title compound (184 mg, 0.567 imol, 41.1%) as a purple powder.
Ή— NMR (200MHz, CDC13) : 51.71 (3H, q, J= 1.0 Hz), 1.79 (1H, m), 2.25 (3H, q, J= 1.2 Hz), 2.31 (1H, m), 2.40 (2H, m), 3.07 (1H, dd, J= 13.8, 5.4 Hz), 3.31 (1H, dd, J= 13.4, 4.0 Hz), 4.09 (1H, m), 7.2-7.8 (2H, br), 8.33 (1H, brs) . Ή- NMR (200MHz, CDC1 3) : 51.71 (3H, q, J = 1.0 Hz), 1.79 (1H, m), 2.25 (3H, q, J = 1.2 Hz), 2.31 (1H, m), 2.40 ( 2H, m), 3.07 (1H, dd, J = 13.8, 5.4 Hz), 3.31 (1H, dd, J = 13.4, 4.0 Hz), 4.09 (1H, m), 7.2-7.8 (2H, br), 8.33 (1H, brs).
元素分析値: C)3H16N402S2として計算値: C, 48.13; H, 4.97; N, 17.27; S, 19.77Elemental analysis: C) 3 H 16 N 4 0 2 S 2 Calculated: C, 48.13; H, 4.97 ; N, 17.27; S, 19.77
(%) 、 実測値: C, 48.15; H, 4.73; N, 17.13; S, 19.95 (%) . (%), Found: C, 48.15; H, 4.73; N, 17.13; S, 19.95 (%).
即. 197.0-200.0°C 実施例 6 - 45 Immediate. 197.0-200.0 ° C Example 6-45
3 - { [(卜ァミノ- 5, 6-ジメチル- 3-ォキソ -3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィ ル) スルファニル]メチル }-1_ピぺリジンカルボン酸 tert-ブチル 3-{[(Tamino-5,6-dimethyl-3-oxo-3H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] methyl} -1_piperidine Tert-butyl carboxylate
実施例 4-558と同様にして得た粗製の 3- {[(5, 6-ジメチル- 1- {[(4-メチルフエ ニル)スルホニル]イミノ}-3_ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チ ァジン- 8-ィル)スルファニル]メチル } -卜ピペリジンカルボン酸 tert-ブチル (純度 50¾, 2.0 g, 1.7 mmol)とギ酸アンモニゥム (0.57 g, 8.6匪 ol) のェタノ ール (40ml) 懸濁液を 23時間加熱還流した。反応中にギ酸アンモニゥム(0.34 g, 5.2 inmol)を加えた。 反応液を濃縮後、 酢酸ェチルで抽出した。 有機層を ^およ び飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固して濃紫色油状物を 得た。 これをシリカゲル(12 g)のクロマトグラフィ一 (酢酸ェチルーへキサン、 4:6-8:2)で精製し、酢酸ェチルおよびジェチルェ一テルから結晶化して表題化合 物(201 mg, 0.473 腿 ol, 27.4%)を濃紫色粉末として得た。 The crude 3-{[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3_oxo-2,3-dihydro-1H obtained in the same manner as in Example 4-558 -Imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] methyl} -tert-butyl topiperidinecarboxylate (purity 50¾, 2.0 g, 1.7 mmol) and ammonium formate (0.57 g) , 8.6 bandol) in ethanol (40 ml) was heated to reflux for 23 hours. During the reaction, ammonium formate (0.34 g, 5.2 inmol) was added. After the reaction solution was concentrated, it was extracted with ethyl acetate. The organic layer was washed with ^ and saturated saline, dried over sodium sulfate, and concentrated to dryness to obtain a dark purple oil. This was purified by chromatography on silica gel (12 g) (ethyl acetate-hexane, 4: 6-8: 2) and crystallized from ethyl acetate and geethyl ether to give the title compound (201 mg, 0.473 t, 27.4). %) As a dark purple powder.
Ή—NMR (200MHz, CDC13) : δ 1.35 (2Η, m), 1.46 (9H, s), 1.66 (1H, m), 1.74 (3H, s), 1.89 (2H, ), 2.26 (3H, s), 2.81 (1H, m), 2.86 (2H, m), 2.98 (1H, brt, J= 9.8 Hz), 3.74 (1H, brd, J= 13.8 Hz) , 3.90 (1H, brd, J= 14.4 Hz) , 7.55 (2H, br), Ή-NMR (200MHz, CDC1 3 ): δ 1.35 (2Η, m), 1.46 (9H, s), 1.66 (1H, m), 1.74 (3H, s), 1.89 (2H,), 2.26 (3H, s ), 2.81 (1H, m), 2.86 (2H, m), 2.98 (1H, brt, J = 9.8 Hz), 3.74 (1H, brd, J = 13.8 Hz), 3.90 (1H, brd, J = 14.4 Hz) ), 7.55 (2H, br),
元素分析値: C19H28N403S2として計算値: C, 53.75; H, 6.65; N, 13.20; S, 15.10 (%) 、 実測値: C, 53.72; H, 6.66; N, 13.05; S, 14.97 (%) · Elemental analysis: C 19 H 28 N 4 0 3 S 2 Calculated: C, 53.75; H, 6.65; N, 13.20; S, 15.10 (%), found: C, 53.72; H, 6.66; N, 13.05; S, 14.97 (%)
mp. 163.0-164.5°C mp.163.0-164.5 ° C
実施例 6-46 Example 6-46
(2S, 4R) -2-{[ (卜ァミノ - 5, 6-ジメチル- 3-ォキソ -3Η-ィミダゾ [5, 1-c] [1,4]チア ジン- 8-ィル)スルファニル]メチル }_4-ヒドロキシ -1-ピロリジンカルボン酸 tert-ブチル (2S, 4R) -2-{[(Tamino-5,6-dimethyl-3-oxo-3Η-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] methyl } _Tert-butyl 4-hydroxy-1-pyrrolidinecarboxylate
実施例 4- 555と同様にして得た粗製の(2S)- 4-[(5,6- )- 1,2_ (2S, 4R)-2-{ [(5, 6- ジメチル -1 - { [ (4-メチルフェニル)スルホニル]イミノ } -3-ォキソ -1, 3-ジヒドロ - 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル)スルファニル]メチル } -4-ヒドロキ シ- 1-ピロリジンカルボン酸 ter卜ブチル (純度 70%, 1.6 g, 1.9 醒 ol)とギ酸ァ ンモニゥム (0.64 g, 9.7 讓01) のエタノール (30 ml) 懸濁液を 18時間加熱還 流した。 反応中にギ酸アンモニゥム(0.51 g, 7.7 mmol)を加えた。 反応液を濃縮 後、 酢酸ェチルで抽出した。 有機層を 5%および飽和食塩水で洗浄後、 硫酸ナトリ ゥムで乾燥し、 濃縮乾固して濃紫色油状物を得た。 これをシリカゲル (15 g)のク 口マトグラフィー(メタノール一酢酸ェチル、 1:99-7:93) で精製し、酢酸ェチル およびジェチルエーテルから結晶化して表題化合物(198 mg, 0.464匪 ol, 23.9%) を濃紫色粉末として得た。 Crude (2S) -4-[(5,6-)-1,2_ (2S, 4R) -2-{[(5,6-dimethyl-1-{obtained in the same manner as in Example 4-555 [(4-Methylphenyl) sulfonyl] imino} -3-oxo-1,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] methyl} -4 A suspension of tert-butyl-1-hydroxy-1-pyrrolidinecarboxylate (purity 70%, 1.6 g, 1.9 mmol) and ammonium formate (0.64 g, 9.7 benzene) in ethanol (30 ml) was heated and returned for 18 hours. Shed. During the reaction, ammonium formate (0.51 g, 7.7 mmol) was added. After the reaction solution was concentrated, it was extracted with ethyl acetate. The organic layer was washed with 5% and saturated saline, dried over sodium sulfate, and concentrated to dryness to obtain a dark purple oil. This was purified by chromatography on silica gel (15 g) (ethyl acetate-methanol, 1: 99-7: 93) and crystallized from ethyl acetate and getyl ether to give the title compound (198 mg, 0.464 ol, 23.9%) as a dark purple powder.
¾-NMR (300MHz, CDC13) : 51.48 (9H, s), 1.72 (3H, q, J= 0.6 Hz), 1.99 (1H, ddd, J= 13.5, 7.8, 4.8 Hz), 2.24 (3H, q, J= 0.9 Hz), 2.97 (1H, m), 3.27 (1H, m), 3.40 (1H, brs), 3.45 (1H, brs), 3.50 (1H, m), 3.64(1H, m), 4.25 (1H, m), 4.45 (1H, brs), 7.49 (2H, br). ¾-NMR (300MHz, CDC1 3 ): 51.48 (9H, s), 1.72 (3H, q, J = 0.6 Hz), 1.99 (1H, ddd, J = 13.5, 7.8, 4.8 Hz), 2.24 (3H, q , J = 0.9 Hz), 2.97 (1H, m), 3.27 (1H, m), 3.40 (1H, brs), 3.45 (1H, brs), 3.50 (1H, m), 3.64 (1H, m), 4.25 (1H, m), 4.45 (1H, brs), 7.49 (2H, br).
元素分析値: C18H26N404S2 Ό.2酢酸ェチルとして Elemental analysis: C 18 H 26 N 4 0 4 S 2 Ό.2 As ethyl acetate
計算値: C, 50.84; Η, 6.26; Ν, 12.61; S, 14.44 (%) . Calculated: C, 50.84; Η, 6.26; Ν, 12.61; S, 14.44 (%).
実測値: C, 50.66; Η, 6.23; Ν, 12.62; S, 14.38 (%) . Found: C, 50.66; Η, 6.23; Ν, 12.62; S, 14.38 (%).
実施例 6-47 Example 6-47
3-ァミノ- 4- [(5-フルォロペンチル)スルファニル ]_8, 9-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チァジン-1,6(711)-ジォン 3-Amino-4-[(5-fluoropentyl) sulfanyl] _8,9-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1,6 (711) -dione
実施例 4-561で得られた N-{1, 6-ジォキソ- 4- [(5-フルォロペンチル)スルファ 二ル]- 1, 2, 6, 7, 8, 9 -へキサヒド口- 3H -ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン- 4- イリデン }- 4-メチルベンゼンスルホンアミド (1.50g, 2.94 mmol) とギ酸アンモ ニゥム (1.95 g, 29.4 mmol) のエタノール (30.0 ml) の懸濁液を 80°Cで 15時 間加熱した後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに 付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (185 mg, 18¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 191- 193°Cの暗赤色結晶を得た。 N- {1,6-Dioxo-4-[(5-fluoropentyl) sulfanyl]-1,2,6,7,8,9-hexahydrid-3H-imidazo obtained in Example 4-561 [5,1-c] [1,4] benzothiazine-4-ylidene} -4-methylbenzenesulfonamide (1.50 g, 2.94 mmol) and ammonium formate (1.95 g, 29.4 mmol) in ethanol (30.0 ml) After heating the suspension at 80 ° C for 15 hours, water was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (185 mg, 18¾) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals, mp 191-193 ° C.
IR (KBr)リ: 3387, 3287, 2948, 2240, 1721, 1653, 1642, 1563 cm"1. IR (KBr): 3387, 3287, 2948, 2240, 1721, 1653, 1642, 1563 cm " 1 .
Ή—NMR (CDC13) : δ 1.50-1.70 (2Η, m), 1.70-1.90 (4 m), 1.92-2.10 (2H, m), 2.48 (2H, t, J=7.2 Hz), 3.04 (2H, t, J=7.2 Hz), 3.30 (2H, t, J=6.4 Hz), 4.46 (2H, dt, J=47.2, 5.6 Hz), 7.31 (2H, bs). 元素分析値: C15H18N302S2Fとして Ή-NMR (CDC1 3): δ 1.50-1.70 (2Η, m), 1.70-1.90 (4 m), 1.92-2.10 (2H, m), 2.48 (2H, t, J = 7.2 Hz), 3.04 (2H , t, J = 7.2 Hz), 3.30 (2H, t, J = 6.4 Hz), 4.46 (2H, dt, J = 47.2, 5.6 Hz), 7.31 (2H, bs). Elementary analysis: as C 15 H 18 N 3 0 2 S 2 F
計算値 (¾) : C, 50.69; H, 5.10; N, 11.82; S, 18.04. Calculated value (¾): C, 50.69; H, 5.10; N, 11.82; S, 18.04.
実測値 (¾) : C, 50.71; H, 5.02; N, 11.55; S, 17.82. Found (¾): C, 50.71; H, 5.02; N, 11.55; S, 17.82.
実施例 6-48 Example 6-48
(2S)-2- {[ (卜ァミノ- 5,6-ジメチル- 3-ォキソ - 3H-イミダゾ [5,卜 c] [1, 4]チアジン -8 -ィル)スルファニル]メチル }_1-ピロリジンカルボン酸 tert-ブチル (2S) -2-{[(Tamino-5,6-dimethyl-3-oxo-3H-imidazo [5, tric] [1,4] thiazin-8-yl) sulfanyl] methyl} _1-pyrrolidine Tert-butyl carboxylate
実施例 4- 559で得た化合物 (885 mg, 1.57歷 ol)とギ酸アンモニゥム (520 mg, 7.84匪 ol) のエタノール (20ml) 懸濁液を 17時間加熱還流した。 反応中にギ酸 アンモニゥム(313 mg, 4.71扁 ol)を加えた。 反 液を濃縮後、 酢酸ェチルで抽出 して得た有機層を 2 炭酸水素ナトリゥムおよび飽和食塩水で洗浄後、硫酸ナトリ ゥムで乾燥し、 濃縮乾固して濃紫色油状物を得た。 これをシリカゲル (10 g)のク 口マトグラフィ一 (酢酸ェチルーへキサン、 4:6-8:2) で精製し、 ジェチルェ一テ ルから結晶化して表題化合物(467 mg, l.Hmmol, 72.5%)を赤色粉末として得た。 Ή- NMR (200MHz, CDC13) : (51.48 (9H, s), 1.73 (3H, s), 1.89 (3H, m), 2.07 (1H, m), 2.26 (3H, q, J= 1.0 Hz), 2.94/3.06 (1H, br), 3.29 (1H, d, J= 4.0 Hz), 3.35 (1H, dd, J= 4.0 Hz), 3.40 (1H, m), 4.05 (1H, m), 7.19 (2H, br). A suspension of the compound (885 mg, 1.57 mol) obtained in Example 4-559 and ammonium formate (520 mg, 7.84 bandol) in ethanol (20 ml) was heated under reflux for 17 hours. During the reaction, ammonium formate (313 mg, 4.71 mmol) was added. After concentrating the reaction solution, the organic layer obtained by extraction with ethyl acetate was washed with sodium bicarbonate and saturated saline, dried over sodium sulfate, and concentrated to dryness to obtain a dark purple oil. This was purified by chromatography on silica gel (10 g) (ethyl acetate-hexane, 4: 6-8: 2) and crystallized from getyl ether to give the title compound (467 mg, l.Hmmol, 72.5% ) Was obtained as a red powder. Ή- NMR (200MHz, CDC1 3) : (51.48 (9H, s), 1.73 (3H, s), 1.89 (3H, m), 2.07 (1H, m), 2.26 (3H, q, J = 1.0 Hz) , 2.94 / 3.06 (1H, br), 3.29 (1H, d, J = 4.0 Hz), 3.35 (1H, dd, J = 4.0 Hz), 3.40 (1H, m), 4.05 (1H, m), 7.19 ( 2H, br).
元素分析値: C18H26N403S2 O.5H20として: C, 51.53; H, 6.49; N, 13.35; S, 15.29Elementary analysis: as C 18 H 26 N 4 0 3 S 2 O.5H 2 0: C, 51.53; H, 6.49; N, 13.35; S, 15.29
(%) 、 実測値: C, 51.62; H, 6.47; N, 13.30; S, 15.18 (%) . (%), Found: C, 51.62; H, 6.47; N, 13.30; S, 15.18 (%).
融点: 175.0-176.0°C Melting point: 175.0-176.0 ° C
実施例 6-49 Example 6-49
1 -ァミノ- 8- [(5, 5-ジフルォロシクロォクチル)スルファニル ]- 5,6-ジメチル -3H_ ィミダゾ [5, 1-c] [1, 4]チアジン- 3 -オン 1-Amino-8-[(5,5-difluorocyclooctyl) sulfanyl] -5,6-dimethyl-3H_ imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4-547で得た化合物(653 mg, 1.24讓 ol)とギ酸アンモニゥム (0.41 g, 6.19匪01) のエタノール a5ml) 懸濁液を 16時間加熱還流した。 反応中にギ酸 アンモニゥム(247 mg, 3.72腿 ol)を加えた。 反応液を濃縮後、 酢酸ェチルで抽出 して得た有機層を 2%炭酸水素ナトリゥムおよび飽和食塩水で洗浄後、硫酸ナトリ ゥムで乾燥し、 濃縮して得た濃紫色油状物にメタノールを加えた。 析出物を濾取 し、 メタノールで洗浄、 乾燥して表題化合物 (245 mg, 0.656腿 ol, 52.9 )を紫 褐色粉末として得た。 ¾-NMR (300MHz, CDC13) : <51.72 (4H, m), 1.74 (3H, q, J= 0.9 Hz), 1.91 (2H, m), 2.10 (6H, m), 2.26 (3H, q, J= 0.9 Hz), 3.46 (1H, bri, J= 8 Hz), 7.25 (1H, br), 8.60 (1H, br). A suspension of the compound (653 mg, 1.24 benzyl) obtained in Example 4-547 and ammonium formate (0.41 g, 6.19 maraudal 01) in ethanol a5 ml) was heated under reflux for 16 hours. During the reaction, ammonium formate (247 mg, 3.72 tmol) was added. After concentrating the reaction solution, the organic layer obtained by extraction with ethyl acetate was washed with 2% sodium bicarbonate and saturated saline, dried over sodium sulfate, and concentrated. added. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (245 mg, 0.656 mol, 52.9) as a purple-brown powder. ¾-NMR (300MHz, CDC1 3 ): <51.72 (4H, m), 1.74 (3H, q, J = 0.9 Hz), 1.91 (2H, m), 2.10 (6H, m), 2.26 (3H, q, J = 0.9 Hz), 3.46 (1H, bri, J = 8 Hz), 7.25 (1H, br), 8.60 (1H, br).
元素分析値: C16H21N3OS2F2として計算値: C, 51.45; H, 5.67; , 11.25; S, 17.17; F, 10.17 (%) 、 実測値: C, 51.54; H, 5.61; , 11.22; S, 16.90; F, 10.08 (%) · Elemental analysis: C 16 H 21 N 3 OS 2 F 2 Calculated: C, 51.45; H, 5.67;, 11.25; S, 17.17; F, 10.17 (%), found: C, 51.54; H, 5.61 ;, 11.22; S, 16.90; F, 10.08 (%)
融点: 182.5-183.0°C Melting point: 182.5-183.0 ° C
実施例 6 - 50 Example 6-50
1 -ァミノ- 8 -({[3- (フルォロメチル)シク口へキシル]メチル }スルファニル )-5, 6- ジメチル- 3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3-オン 1-Amino-8-({[3- (fluoromethyl) cyclohexyl] methyl} sulfanyl) -5,6-dimethyl-3H-imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4-551で得た化合物 (1.55 g, 3.04腿 ol)とギ酸アンモニゥム (1.01 g, 15.2腿01) のエタノール (31ml) 懸濁液を 23時間加熱還流した。 反応中にギ酸 アンモニゥム(605 mg, 9.12imol)を 2回加えた。反応液を濃縮後、 酢酸ェチル(80 ml)で抽出した。 有機層を水、 2%炭酸水素ナトリウムおよび飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固して濃紫色油状物を得た。 これをシリカゲル (10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 4:6-8:2) で精製し、 メタ ノールから結晶化して表題化合物(448 Dig, 1.26腿 ol, 41.5 )を紫色粉末として 得た。 . A suspension of the compound (1.55 g, 3.04 mol) obtained in Example 4-551 and ammonium formate (1.01 g, 15.2 mol) in ethanol (31 ml) was heated under reflux for 23 hours. During the reaction, ammonium formate (605 mg, 9.12 imol) was added twice. After the reaction solution was concentrated, it was extracted with ethyl acetate (80 ml). The organic layer was washed with water, 2% sodium hydrogen carbonate and saturated saline, dried over sodium sulfate, and concentrated to dryness to obtain a dark purple oil. This was purified by chromatography on silica gel (10 g) (ethyl acetate-hexane, 4: 6-8: 2) and crystallized from methanol to give the title compound (448 Dig, 1.26 mol, 41.5) as a purple powder. Obtained. .
Ή-證 (200MHz, CDC13, 1:1 c is/trans 混合物) : ά 0.96/0.80 (2H, m/q, J= 12.4 Hz), 1.35 (2H, m), 1.50-1.80 (3H, m), 1.75 (3H, s), 1.93 (3H, m), 2.26 (3H, s), 2.86/2.94 (2H, d, J= 6.0/7.2 Hz), 4.25/4.32 (2H, ddd/dd, J= 47.4, 5.3, 1.7/47.4, 6.6 Hz), 約 7.6 (2H, br). Ή- testimony (200MHz, CDC1 3, 1: 1 c is / trans mixture): ά 0.96 / 0.80 (2H , m / q, J = 12.4 Hz), 1.35 (2H, m), 1.50-1.80 (3H, m ), 1.75 (3H, s), 1.93 (3H, m), 2.26 (3H, s), 2.86 / 2.94 (2H, d, J = 6.0 / 7.2 Hz), 4.25 / 4.32 (2H, ddd / dd, J = 47.4, 5.3, 1.7 / 47.4, 6.6 Hz), about 7.6 (2H, br).
元素分析値: C16H22N3OS2Fとして計算値: C, 54.06; H, 6.24; Ν,.11.82; S, 18.04; F, 5.34 (%) 、 実測値: C, 53.99; Η, 6.40; Ν, 11.61; S, 18.17; F, 5.39 (%) · 融点: 168.5-169.5°C Elemental analysis: Calculated for C 16 H 22 N 3 OS 2 F: C, 54.06; H, 6.24; Ν, .11.82; S, 18.04; F, 5.34 (%), found: C, 53.99; Η, 6.40; Ν, 11.61; S, 18.17; F, 5.39 (%) · Melting point: 168.5-169.5 ° C
実施例 6-51 Example 6-51
1 -アミノ -5 , 6-ジメチル- 8- {[( 1 -メチルシクロへキシル)メチル]スルファニ ル}- 3Hイミダゾ [5, 1-c] [1, 4]チアジン- 3-オン 1-amino-5,6-dimethyl-8-{[(1-methylcyclohexyl) methyl] sulfanyl} -3H imidazo [5,1-c] [1,4] thiazin-3-one
実施例 4- 576で得た化合物 (427 mg, 0.868 mmol)とギ酸アンモニゥム (288 mg, 4.34廳01) のエタノール (10 ml) 懸濁液を 22時間加熱還流した。 反応中に ギ酸アンモニゥム(28 mg, 4.34 mmol, および 170 mg, 2.60匪 ol)を加えた。 反応液を濃縮後、 酢酸ェチル (40 ml)で抽出した。 有機層を水、 2%炭酸水素ナトリ ゥムおよび飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮して濃紫色油状 物を得た。 これをメタノールから結晶化して表題化合物 (168 mg, 0.498腿 ol, 57.3%)を灰紫色粉末として得た。 The compound obtained in Example 4-576 (427 mg, 0.868 mmol) and ammonium formate (288 A suspension of mg, 4.34 hall 01) in ethanol (10 ml) was heated under reflux for 22 hours. Ammonium formate (28 mg, 4.34 mmol, and 170 mg, 2.60 bandol) was added during the reaction. After the reaction solution was concentrated, it was extracted with ethyl acetate (40 ml). The organic layer was washed with water, 2% sodium hydrogen carbonate and saturated saline, dried over sodium sulfate, and concentrated to obtain a dark purple oil. This was crystallized from methanol to give the title compound (168 mg, 0.498 mol, 57.3%) as a gray purple powder.
Ή-NMR (300MHz, CDC13) : (51.02 (3H, s), 1.37 (5H, m), 1.48 (5H, m), 1.75 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 0.9 Hz), 2.95 (2H, s), 約 7.4 (1H, br), 約 7.8 (1H, br). Ή-NMR (300MHz, CDC1 3 ): (51.02 (3H, s), 1.37 (5H, m), 1.48 (5H, m), 1.75 (3H, q, J = 1.2 Hz), 2.26 (3H, q, J = 0.9 Hz), 2.95 (2H, s), about 7.4 (1H, br), about 7.8 (1H, br).
元素分析値 Cl6H23N30S2として計算値: C, 56.94; H, 6.87; N, 12.45; S, 19.00 (%) 、 実測値: C, 56.88; H, 7.02; , 12.36; S, 19.05 (%) . Elemental analysis C l6 H 23 N 3 0S 2 Calculated: C, 56.94; H, 6.87 ; N, 12.45; S, 19.00 (%), Found: C, 56.88; H, 7.02 ;, 12.36; S, 19.05 (%).
融点: 186.0-188.5°C Melting point: 186.0-188.5 ° C
実施例 7-1 Example 7-1
2, 4-ジク口口- N- [5, 6-ジメチル- 3-ォキソ -8- (ペンチルスルファ二ル)- 2, 3-ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン]ベンゼンスルホンアミド 実施例 6-1で得られた卜アミノ- 5, 6-ジメチル- 8_ (ぺンチルスルファニル) -3H- イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (200 mg, 0.672 mmol) をピリジン (2.00 ml) に溶かし、 2, 4-ジクロロベンゼンスルホニル クロリド (330 mg, 1.34腿 ol) を加えた。 反応混合物を室温で 24時間かき混ぜた後、 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留 物をシリカゲルカラムクロマトグラフィーに付し、 n-へキサン-酢酸ェチル(10:1, v/v) で溶出して題記化合物 (15.7 mg, 4.6%) を暗赤色結晶として得た。 酢酸ェ チル-ジィソプロピルエーテルから再結晶して融点 168-170°Cの暗赤色結晶を得 た。 2,4-Dichloro-N- [5,6-dimethyl-3-oxo-8- (pentylsulfanyl) -2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene] benzenesulfonamide triamino-5,6-dimethyl-8_ (pentylsulfanyl) -3H-imidazo obtained in Example 6-1 [5, tric] [1, 4] Thiazin-3-one (200 mg, 0.672 mmol) was dissolved in pyridine (2.00 ml), and 2,4-dichlorobenzenesulfonyl chloride (330 mg, 1.34 t ol) was added. After stirring the reaction mixture at room temperature for 24 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (15.7 mg, 4.6%) as dark red crystals. Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals with a melting point of 168-170 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3264, 2961, 2930, 2859, 1748, 1725, 1663, 1615, 1557 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3264, 2961, 2930, 2859, 1748, 1725, 1663, 1615, 1557 cm- 1 .
¾-NMR (CDC13) : δ 0.894 (3Η, t, J=7.0Hz), 1.23-1.40 (4H, m), 1.57-1.75 (2H, m), 1.81 (3H, s), 2.30 (3H, s), 2.92 (2H, t, J=7.2 Hz), 7.38 (2H, dd, J=8.8, 2.2 Hz), 7.51 (1H, d, J=2.2 Hz), 8.12 (1H, d, J=8.4 Hz), 9.69 (1H, bs) . 元素分析値: C19H21N303S3C 12として ¾-NMR (CDC1 3): δ 0.894 (3Η, t, J = 7.0Hz), 1.23-1.40 (4H, m), 1.57-1.75 (2H, m), 1.81 (3H, s), 2.30 (3H, s), 2.92 (2H, t, J = 7.2 Hz), 7.38 (2H, dd, J = 8.8, 2.2 Hz), 7.51 (1H, d, J = 2.2 Hz), 8.12 (1H, d, J = 8.4 Hz), 9.69 (1H, bs ) Elementary analysis:. as C 19 H 21 N 3 0 3 S 3 C 1 2
計算値 (¾) : C, 45.06; H, 4.18; N, 8.30; S, 18.99. Calculated (¾): C, 45.06; H, 4.18; N, 8.30; S, 18.99.
実測値 (%) : C, 44.98; H, 3.88; N, 8.18; S, 18.85. Found (%): C, 44.98; H, 3.88; N, 8.18; S, 18.85.
実施例 7 - 2 Example 7-2
N - [5, 6-ジメチル -3-ォキソ -8- (ペンチルスルファ二ル)- 2, 3-ジヒドロ- 1H-イミダ ゾ [5,1- c] [1, 4]チアジン-卜イリデン] -4-トリフルォロメチルベンゼンスルホン アミド N- [5,6-dimethyl-3-oxo-8- (pentylsulfanyl) -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene] -4-trifluoromethylbenzenesulfonamide
方法 A Method A
実施例 7-1と同様の方法により、 2, 4-ジクロロベンゼンスルホニル クロリドの 代わりに 4 -トリフルォロメチルベンゼンスルホニル クロリド (220 mg, 0.883 讓 ol) を用いて、 実施例 6-1で得られた卜ァミノ- 5,6-ジメチル- 8- (ペンチルス ルファ二ル)- 3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 3-オン (200 mg, 0.67 匪 ol) から題記化合物 (29.5 mg, 8.7¾) を合成した。 酢酸ェチル-ジェチルエーテルか ら再結晶して暗赤色結晶を得た。 In the same manner as in Example 7-1, the compound of Example 6-1 was obtained using 4-trifluoromethylbenzenesulfonyl chloride (220 mg, 0.883 benzyl) instead of 2,4-dichlorobenzenesulfonyl chloride. Triamino-5,6-dimethyl-8- (pentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.67 bandol ol) From there, the title compound (29.5 mg, 8.7 合成) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals.
方法 B Method B
実施例 6-1で得られた 1-ァミノ- 5, 6-ジメチル -8- (ペンチルスルファニル) -3H- イミダゾ [5, 1-c] [1, 4]チアジン -3-オン (175 mg, 0.588 mmol) をテトラヒドロフ ラン (2.00 ml) に溶かし、 4-卜リフルォロメチルベンゼンスルホニル クロリド (220 mg, 0.883 mmol) 及びト リェチルァミン (0.164 ml, 1.18腿。1) を順次加 えた。 反応混合物を室温で 16時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで 抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (114 mg, 39¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再 結晶して融点 162- 164°Cの喑赤色結晶を得た。 1-amino-5,6-dimethyl-8- (pentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-1 (175 mg, 0.588 mmol) was dissolved in tetrahydrofuran (2.00 ml), and 4-trifluoromethylbenzenesulfonyl chloride (220 mg, 0.883 mmol) and triethylamine (0.164 ml, 1.18 thigh) were sequentially added. After stirring the reaction mixture at room temperature for 16 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (114 mg, 39¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-jetil ether to give reddish crystals having a melting point of 162-164 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3231, 2961, 2932, 2861, 1761, 1748, 1663, 1620, 1557 cm—1. Infrared absorption spectrum (IR) (in KBr): 3231, 2961, 2932, 2861, 1761, 1748, 1663, 1620, 1557 cm- 1 .
Ή-NMR (CDC13) : δ 0.910 (3Η, t, J=7.0 Hz), 1.31-1.47 (4H, m), 1.60-1.75 (2H, m), 1.80 (3H, d, J-1.0 Hz), 2.28 (3H, d, J=l.2 Hz), 2.96 (2H, t, J=7.4 Hz), 7.75 (2H, d, J=8.0 Hz), 8.11 (2H, d, J=8.0 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 0.910 (3Η, t, J = 7.0 Hz), 1.31-1.47 (4H, m), 1.60-1.75 (2H, m), 1.80 (3H, d, J-1.0 Hz) , 2.28 (3H, d, J = 1.2 Hz), 2.96 (2H, t, J = 7.4 Hz), 7.75 (2H, d, J = 8.0 Hz), 8.11 (2H, d, J = 8.0 Hz) , 9.60 (1H, bs).
元素分析値: C20H22N303S3として Elemental analysis: as C 20 H 22 N 3 0 3 S 3
計算値 ) : C, 47.51; H, 4.19; N, 8.31; S, 19.03. Calculated)): C, 47.51; H, 4.19; N, 8.31; S, 19.03.
実測値 (¾) : C, 47.20; H, 4.32; N, 8.14; S, 18.94. Found (¾): C, 47.20; H, 4.32; N, 8.14; S, 18.94.
実施例 7-3 Example 7-3
N - [5, 6-ジメチル- 3-ォキソ- 8- (ぺンチルスルファエル) -2, 3 -ジヒドロ- 1H -ィミダ ゾ [5, 1-c] [1,4]チアジン- 1-イリデン] -4-ニトロベンゼンスルホンアミド N- [5,6-Dimethyl-3-oxo-8- (pentylsulfael) -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene ] -4-nitrobenzenesulfonamide
実施例 7-1と同様の方法により、 2, 4-ジクロロベンゼンスルホニル クロリドの 代わりに 4-ニトロベンゼンスルホニル クロリド (183 mg, 0.808匪 ol) を用い て、 実施例 6-1 で得られた 1 -ァミノ- 5, 6-ジメチル -8 -(ペンチルスルファ二 ル) -3H-イミダゾ [5, 1-c] [1,4]チアジン- 3 -オン (160 mg, 0.539腿 ol) から題記 化合物 (4.3mg, 1.7¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶し て融点 183-185°Cの暗赤色結晶を得た。 According to a method similar to that of Example 7-1, 4-nitrobenzenesulfonyl chloride (183 mg, 0.808 ol) was used in place of 2,4-dichlorobenzenesulfonyl chloride to obtain 1- The title compound (160 mg, 0.539 t ol) was obtained from amino-3,6-dimethyl-8- (pentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one. 4.3 mg, 1.7¾) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 183-185 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3108, 2957, 2928, 2868, 1750, 1723, 1661, 1603, 1557, 1532 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3108, 2957, 2928, 2868, 1750, 1723, 1661, 1603, 1557, 1532 cm " 1 .
'Η—丽 R (CDC13) : δ 0.916 (3Η, t, J=6.8Hz), 1.31-1.44 (4H, m), 1.64-1.76 (2H, m), 1.81 (3H, s), 2.29 (3H, d, J=0.8 Hz), 2.97 (2H, t, J=7.2 Hz), 8.16 (2H, d, J=9.2 Hz), 8.34 (2H, d, J=8.8 Hz), 9.59 (1H, bs). '.Eta.丽R (CDC1 3): δ 0.916 (3Η, t, J = 6.8Hz), 1.31-1.44 (4H, m), 1.64-1.76 (2H, m), 1.81 (3H, s), 2.29 ( 3H, d, J = 0.8 Hz), 2.97 (2H, t, J = 7.2 Hz), 8.16 (2H, d, J = 9.2 Hz), 8.34 (2H, d, J = 8.8 Hz), 9.59 (1H, bs).
実施例 7-4 , Example 7-4,
4- ({[5,6-ジメチル -8- (イソペンチルスルファニル) -3-ォキソ -2,3-ジヒドロ- 1H- イミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン]アミノ}スルファニル)安息香酸 実施例 7-2方法 Bと同様の方法により、 4-トリフルォロメチルベンゼンスルホ ニル クロリドの代わりに 4- (クロロスルファニル)安息香酸(174mg, 0.756匪 ol) を用いて、 実施例 6-2で得られた 1-ァミノ- 5, 6-ジメチル- 8 -(イソペンチルスル ファニル) -3H-イミダゾ [5, 1-c] [1,4]チアジン- 3 -オン (350 mg, 1.18腿 ol) から 題記化合物 (258 mg, 46%) を合成した。 酢酸ェチル -ジェチルエーテルから再結 晶して融点 178-180°Cの暗赤色結晶を得た。 4-({[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene] amino } Sulfanyl) benzoic acid Example 7-2 By a method similar to that of Method B, using 4- (chlorosulfanyl) benzoic acid (174 mg, 0.756 bandol) instead of 4-trifluoromethylbenzenesulfonyl chloride, 1-Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-2 (350 The compound (258 mg, 46%) was synthesized from the compound (258 mg, 1.18 t). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 178-180 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3700-2800, 3275, 2957, 2928, 2872, 1730, 1659, 1624, 1599, 1572, 1553, 1539 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3700-2800, 3275, 2957, 2928, 2872, 1730, 1659, 1624, 1599, 1572, 1553, 1539 cm " 1 .
'H-NMR (CDCI3) : δ 1.01 (6Η, d, J=6.4 Hz), 1.50-1.82 (3H, m), 1.87 (3H, d, J=1.2 Hz), 2.38 (3H, d, J=0.6 Hz), 3.11 (2H, t, J-7.8 Hz), 8.11 (2H, d, J=8.4 Hz), 8.55 (2H, d, J=8.8 Hz), 11.13 (1H, bs). 'H-NMR (CDCI3): δ 1.01 (6Η, d, J = 6.4 Hz), 1.50-1.82 (3H, m), 1.87 (3H, d, J = 1.2 Hz), 2.38 (3H, d, J = 0.6 Hz), 3.11 (2H, t, J-7.8 Hz), 8.11 (2H, d, J = 8.4 Hz), 8.55 (2H, d, J = 8.8 Hz), 11.13 (1H, bs).
実施例 7-5 Example 7-5
4- (ブロモメチル) -N- [5, 6-ジメチル- 8 - (ィソぺンチルスルフ 7ニル) -3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1- c] [1,4]チアジン- 1-ィリデン]ベンゼンスルホ ンアミド 4- (bromomethyl) -N- [5,6-dimethyl-8- (isopentylsulfinyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 [Thiazine-1-ylidene] benzenesulfonamide
実施例 7- 2方法 Bと同様の方法により、 4-トリフルォロメチルベンゼンスルホ ニル クロリドの代わりに 4-ブロモメチルベンゼンスルホニル クロリド (204 mg, 0.756腿 ol) を用いて、 実施例 6 - 2で得られた卜ァミノ- 5, 6 -ジメチル -8- (イソ ペンチルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン(350 mg, 1.18 mmol) から題記化合物 (20.0 mg, 7.5%) を合成した。 酢酸ェチル-ジェチルエー テルから再結晶して暗赤色結晶を得た。 ' Example 7-2 In the same manner as in Method B, 4-bromomethylbenzenesulfonyl chloride (204 mg, 0.756 t) was used in place of 4-trifluoromethylbenzenesulfonyl chloride, and Example 6-2 was used. -5,6-Dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (350 mg, 1.18 mmol) )) To give the title compound (20.0 mg, 7.5%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals. '
方法 C 水素化ナトリウム (20.2 mg, 0.504 mmol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-2で得られた卜ァミノ- 5, 6 -ジメチル -8- (イソペンチ ルスルファ二ル)- 3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (100 nig, 0.336 腿 ol) のテトラヒドロフラン(1.00 ml)溶液を加え、氷冷下 10分間かき混ぜた。 これに 4-ブロモメチルベンゼンスルホニル クロリド (136 mg, 0.504 mmol) の テトラヒドロフラン (1.00 ml) 獰液を加え、 さらに 1時間かき混ぜた。 反応混合 物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (139 mg, 78%) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルェ一テルから再結晶して融点 187-188°Cの喑赤色結晶を得た。 赤外吸収スぺクトル(IR) (KBr,中) : 3120, 2959, 2926, 2870, 1742, 1728, 1663, 1605, 1553 cm一1. Method C To a suspension of sodium hydride (20.2 mg, 0.504 mmol) (60% oil) in tetrahydrofuran (1.00 ml) was added the triamino-5,6-dimethyl-8- (isopentylsulfurate) obtained in Example 6-2. Nil) -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 nig, 0.336 t ol) in tetrahydrofuran (1.00 ml) was added thereto, and the mixture was stirred under ice cooling for 10 minutes. To this was added a solution of 4-bromomethylbenzenesulfonyl chloride (136 mg, 0.504 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (139 mg, 78%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain red-red crystals having a melting point of 187-188 ° C. Infrared absorption spectrum (IR) (KBr, medium): 3120, 2959, 2926, 2870, 1742, 1728, 1663, 1605, 1553 cm- 1 .
'H-NMR (CDC13) : δ 0.935 (6Η, d, J=6.2 Hz), 1.50-1.76 (3H, m), 1.80 (3H, s), 2.28 (3H, d, J=0.6 Hz), 2.96 (2H, t, J=8.0 Hz), 4.49 (2H, s, 75% Br), 4.61 (2H, s, 25¾ CI), 7.50 (2H, d, J=8.6 Hz), 7.94 (2H, d, J=8.0 Hz), 9.60 (1H, bs). 'H-NMR (CDC1 3) : δ 0.935 (6Η, d, J = 6.2 Hz), 1.50-1.76 (3H, m), 1.80 (3H, s), 2.28 (3H, d, J = 0.6 Hz), 2.96 (2H, t, J = 8.0 Hz), 4.49 (2H, s, 75% Br), 4.61 (2H, s, 25¾ CI), 7.50 (2H, d, J = 8.6 Hz), 7.94 (2H, d , J = 8.0 Hz), 9.60 (1H, bs).
元素分析値: C20H24N303S3Br 75.2¾ C20HMN303S3C1 24· 8%として Elementary analysis: as C 20 H 24 N 3 0 3 S 3 Br 75.2¾ C 20 H M N 3 0 3 S 3 C1 24 · 8%
計算値 (%) : C, 46.28; H, 4.66; , 8.09; S, 18.51. Calculated value (%): C, 46.28; H, 4.66;, 8.09; S, 18.51.
実測値 (¾) : C, 45.91; H, 4.67; N, 8.13; S, 18.32. Found (¾): C, 45.91; H, 4.67; N, 8.13; S, 18.32.
実施例 7 - 6 Example 7-6
N- [5, 6-ジメチル- 8- (ィソペンチルスルファニル) -3-ォキソ -2, 3-ジヒド口- 1H-ィ ミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン ]-4_エトキシメチルベンゼンスルホン アミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydrido-1H-dimidazo [5,1-c] [1,4] thiazine-1-ylidene ] -4_Ethoxymethylbenzenesulfonamide
実施例 7-5で得られた 4- (プロモメチル) [5, 6-ジメチル -8- (ィソペンチルス ルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン - 1 -ィリ デン]ベンゼンスルホンアミド(259 mg, 0.489 mmol)と酢酸カリウム(145 mg, 1.47 mmol) と水酸化カリウム (96.8mg, 1.47 mmol) のエタノール (5.00 ml) 懸濁液 を 1時間加熱還流した。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(82.4mg, 34%) を得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 116- 118°Cの暗赤色結 晶を得た。 4- (Promomethyl) [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] obtained in Example 7-5 [1,4] Thiazine-1-ylidene] benzenesulfonamide (259 mg, 0.489 mmol), potassium acetate (145 mg, 1.47 mmol) and potassium hydroxide (96.8 mg, 1.47 mmol) in ethanol (5.00 ml) The suspension was heated at reflux for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), to obtain the solvent was distilled off under reduced pressure the title compound (82.4mg, 34%). Recrystallized from Ethyl acetate-Jetil ether to form a dark red color with a melting point of 116-118 ° C A crystal was obtained.
赤外吸収スペクトル(IR) (KBr中) リ: 3223, 2957, 2932, 2870, 1748, 1663, 1618, 1565 cm"1. Infrared absorption spectrum (IR) (in KBr): 3223, 2957, 2932, 2870, 1748, 1663, 1618, 1565 cm " 1 .
Ή-NMR (CDC13) : δ 0.929 (6Η, d, J=6.6 Hz), 1.26 (3H, t, J=7.0 Hz), 1.48-1.78 (3H, m), 1.79 (3H, s), 2.27 (3H, s), 2.94 (2H, t, J=7.6 Hz), 3.57 (2H, q, J=7.0 Hz), 4.56 (2H, s), 7.46 (2H, d, J=8.2 Hz), 7.94 (2H, d, J=8.0 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): δ 0.929 (6Η, d, J = 6.6 Hz), 1.26 (3H, t, J = 7.0 Hz), 1.48-1.78 (3H, m), 1.79 (3H, s), 2.27 (3H, s), 2.94 (2H, t, J = 7.6 Hz), 3.57 (2H, q, J = 7.0 Hz), 4.56 (2H, s), 7.46 (2H, d, J = 8.2 Hz), 7.94 (2H, d, J = 8.0 Hz), 9.61 (1H, bs).
元素分析値: C22H29N304S3として Elementary analysis: as C 22 H 29 N 3 0 4 S 3
計算値 (¾) : C, 53.31; H, 5.90; N, 8.48; S, 19.41. Calculated (¾): C, 53.31; H, 5.90; N, 8.48; S, 19.41.
実測値 (¾) : C, 53.28; H, 5.79; N, 8.42; S, 19.80. Found (¾): C, 53.28; H, 5.79; N, 8.42; S, 19.80.
実施例 7-7 Example 7-7
酢酸 4-{[5, 6-ジメチル- 8- (ィソペンチルスルファニル) - 3-ォキソ -2, 3 -ジヒドロ - 1H -イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン]スルフィンナモイルォキシ}ベ ンジルエステル Acetic acid 4-{[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene ] Sulfin namoyoxy benzyl ester
実施例 7-5で得られた 4- (プロモメチル) -N- [5, 6-ジメチル- 8- (ィソぺンチルス ルファニル) -3-ォキソ - 2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン -トイリ デン]ベンゼンスルホンアミド(315 mg, 0.595 mmol)と酢酸カリウム(177mg, 1.78 腿 ol) と水酸化カリウム (118 mg, 1.78讓 ol) のァセトニトリル (5.00 ml) 懸 濁液を 3時間加熱還流した。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲル カラムクロマトグラフィーに付し、 n-へキサン -酢酸ェチル (5:1, v/v) で溶出し て題記化合物 (110mg, 36%) を得た。 本品はこれ以上精製することなく次の反応 に使用した。 4- (Promomethyl) -N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydrido-1H-imidazo obtained in Example 7-5 [ Acetonitrile of 5, 1-c] [1,4] thiazine-tolylidene] benzenesulfonamide (315 mg, 0.595 mmol), potassium acetate (177 mg, 1.78 pt. Ol) and potassium hydroxide (118 mg, 1.78 dl) (5.00 ml) The suspension was heated to reflux for 3 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (5: 1, v / v) to give the title compound (110 mg, 36%). This product was used for the next reaction without further purification.
赤外吸収スぺクトル(IR) (KBr中) ソ : 3250, 2957, 2934, 2872, 1744, 1663, 1615, 1559 cm"1. Infrared absorption spectrum (IR) (in KBr) So: 3250, 2957, 2934, 2872, 1744, 1663, 1615, 1559 cm " 1 .
^-N R (CDCI3) : δ 0.934 (6Η, d, J=6.2 Hz), 1.48-1.77 (3H, m), 1.79 (3H, d, J=1.2 Hz), 2.13 (3H, s), 2.27 (3H, d, J=1.2 Hz), 2.95 (2H, t, J=7.4Hz), 5.16 (2H, s), 7.46 (2H, d, J=8.4 Hz), 7.96 (2H, d, J=8.4 Hz), 9.60 (1H, bs). 実施例 7-8 N-[5, 6-ジメチル- 8- (ィソペンチルスルファ二ル)- 3-ォキソ -2, 3 -ジヒドロ- 1H -ィ ミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン ]-4-ヒドロキシメチルベンゼンスルホ ンアミド ^ -NR (CDCI3): δ 0.934 (6Η, d, J = 6.2 Hz), 1.48-1.77 (3H, m), 1.79 (3H, d, J = 1.2 Hz), 2.13 (3H, s), 2.27 ( 3H, d, J = 1.2 Hz), 2.95 (2H, t, J = 7.4 Hz), 5.16 (2H, s), 7.46 (2H, d, J = 8.4 Hz), 7.96 (2H, d, J = 8.4) Hz), 9.60 (1H, bs). N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-dimidazo [5,1-c] [1,4] thiazine-1 -Ylidene] -4-hydroxymethylbenzenesulfonamide
実施例 7 - 7 で得られた酢酸 4- {[5, 6-ジメチル -8 -(イソペンチルスルファ二 ル)- 3-ォキソ -2, 3-ジヒドロ -1Η-イミダゾ [5, 1- c] [1,4]チアジン-卜ィリデン]ス ルフィンナモイルォキシ}ベンジル エステル (110 mg) をテトラヒドロフラン (1.00 ml) とエタノール (1.00 ml) の混合物に溶かし、 1N水酸化ナトリウム水 溶液 (0.647 ml) を加えて 10分間かき混ぜた。 反応混合物に 1N塩酸を加えて酢 酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して 題記化合物 (58.2 ig, 58%) を得た。 酢酸ェチル -ジィソプロピルエーテルから再 結晶して融点 165-167 の暗赤色結晶を得た。 Acetic acid 4-{[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1Η-imidazo [5,1-c] obtained in Examples 7-7 [1,4] Thiazine-triylidene] sulfinnamoyloxy} benzyl ester (110 mg) was dissolved in a mixture of tetrahydrofuran (1.00 ml) and ethanol (1.00 ml), and 1N aqueous sodium hydroxide solution (0.647 ml) And stirred for 10 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain the solvent was distilled off under reduced pressure the title compound (58.2 ig, 58%). Recrystallization from ethyl acetate-disopropyl ether gave dark red crystals with a melting point of 165-167.
赤外吸収スぺクトル(IR) (KBr中) V 3449, 3380-2720, 2959, 2934, 2872, 1748, 1665, 1611, 1553 cm"1. , Infrared absorption spectrum (IR) (in KBr) V 3449, 3380-2720, 2959, 2934, 2872, 1748, 1665, 1611, 1553 cm " 1 .
Ή一 NMR (CDC13) : (50.931 (6H, d, J=6.6 Hz), 1.49-1.78 (3H, m), 1.79 (3H, s), 2.27 (3H, d, J=1.2 Hz), 2.95 (2H, t, J=7.6 Hz), 4.79 (2H, d, J=5.0 Hz), 7.49 (2H, d, J=8.4 Hz), 7.96 (2H, d, J=8.4 Hz), 9.61 (1H, bs). Ή one NMR (CDC1 3): (50.931 (6H, d, J = 6.6 Hz), 1.49-1.78 (3H, m), 1.79 (3H, s), 2.27 (3H, d, J = 1.2 Hz), 2.95 (2H, t, J = 7.6 Hz), 4.79 (2H, d, J = 5.0 Hz), 7.49 (2H, d, J = 8.4 Hz), 7.96 (2H, d, J = 8.4 Hz), 9.61 (1H , bs).
元素分析値 C2。H25N304S3として Elemental analysis value C 2. As H 25 N 3 0 4 S 3
計算値 (¾) : C, 51.37; H, 5.39; N, 8.99; S, 20.57. Calculated (¾): C, 51.37; H, 5.39; N, 8.99; S, 20.57.
実測値 (¾) : C, 51.18; H, 5.41; , 9.08; S, 20.56. Found (¾): C, 51.18; H, 5.41;, 9.08; S, 20.56.
実施例 7 - 9 Example 7-9
N - [5, 6-ジメチル -8- (イソペンチルスルファ二ル) - 3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン ]-4-メトキシベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4-メトキシベンゼンスルホニル クロリド (208 mg, 1.01 mraol) を用いて、 実施例 6-2で得られた卜ァミノ- 5,6-ジメチル- 8- (イソペンチルスル ファニル) -3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3-オン (350 mg, 1.18画 1) から 題記化合物 (222 mg, 95%) を合成した。 酢酸ェチル -ジェチルエーテルから再結 晶して融点 181-182 の暗赤色結晶を得た。 N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene ] -4-Methoxybenzenesulfonamide Example 7-5 Method was performed in the same manner as in Method C, except that 4-methoxybenzenesulfonyl chloride (208 mg, 1.01 mraol) was used instead of 4-bromomethylbenzenesulfonyl chloride. Triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-2 (350 mg, The title compound (222 mg, 95%) was synthesized from 1.18. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 181-182.
赤外吸収スペクトル(IR) (KBr中) V: 3235, 2955, 2840, 1736, 1663, 1613, 1597, 1563, 1499 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3235, 2955, 2840, 1736, 1663, 1613, 1597, 1563, 1499 cm " 1 .
¾— NMR (CDC13) : δ 0.927 (6H, d, J=6.6 Hz), 1.46-1.74 (3H, m), 1.79 (3H, s), 2.24 (3H, d, J=1.0 Hz), 2.94 (2H, t, J=7.6 Hz), 3.87 (3H, s), 6.96 (2H, d, J=8.8 Hz), 7.90 (2H, d, J-8.8 Hz), 9.61 (1H, bs). ¾- NMR (CDC1 3): δ 0.927 (6H, d, J = 6.6 Hz), 1.46-1.74 (3H, m), 1.79 (3H, s), 2.24 (3H, d, J = 1.0 Hz), 2.94 (2H, t, J = 7.6 Hz), 3.87 (3H, s), 6.96 (2H, d, J = 8.8 Hz), 7.90 (2H, d, J-8.8 Hz), 9.61 (1H, bs).
元素分析値: C2QH25N304S3として . Elementary analysis: as C 2Q H 25 N 3 0 4 S 3.
計算値 ) : C, 51.37; H, 5.39; N, 8.99; S, 20.57. Calculated)): C, 51.37; H, 5.39; N, 8.99; S, 20.57.
実測値 ( ) : C, 51.23; H, 5.40; N, 9.09; S, 20.48. Found (): C, 51.23; H, 5.40; N, 9.09; S, 20.48.
実施例 7-10 Example 7-10
N- [5, 6-ジメチル- 8- (ィソぺンチルスルファニル) -3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, 1-c] [1,4]チアジン-卜イリデン] -4-ェチルベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4-ェチルベンゼンスルホニル クロリド (115 mg, 0.504 mmol) を用いで、 実施例 6-2で得られた 1 -ァミノ- 5, 6-ジメチル- 8- (イソペンチルスル ファニル) - 3H -イミダゾ [5,卜 c] [1, 4]チアジン- 3 -オン (100 mg, 0.336匪 ol) か ら題記化合物 (92.8mg, 59¾) を合成した。 酢酸ェチル-ジェチルエーテルから再 結晶して融点 169- 171°Cの暗赤色結晶を得た。 N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene ] -4-Ethylbenzenesulfonamide Example 7-5 In the same manner as in Method C, 4-ethylmethylsulfonyl chloride (115 mg, 0.504 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. The 1-amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one obtained in Example 6-2 (100 mg, 0.336 bandol ol) to give the title compound (92.8 mg, 59 mg). The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals having a melting point of 169-171 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3208, 2955, 2934, 1870, 1748, 1717, 1661, 1615, 1559 cm"'. Infrared absorption spectrum (IR) (in KBr) V: 3208, 2955, 2934, 1870, 1748, 1717, 1661, 1615, 1559 cm "'.
¾-NMR (CDCI3) : δ 0.927 (6Η, d, J=6.6 Hz), 1.25 (3H, t, J=7.6 Hz), 1.48-1.75 (3H, m), 1.79 (3H, d, J=0.8 Hz), 2.26 (3H, d, J=0.8 Hz), 2.71 (2H, q, J=7.4 Hz), 2.94 (2H, t, J=7.6 Hz), 7.30 (2H, d, J=8.4 Hz), 7.87 (2H, d, J=8.4 Hz), 9.60 (1H, bs). ¾-NMR (CDCI3): δ 0.927 (6Η, d, J = 6.6 Hz), 1.25 (3H, t, J = 7.6 Hz), 1.48-1.75 (3H, m), 1.79 (3H, d, J = 0.8 Hz), 2.26 (3H, d, J = 0.8 Hz), 2.71 (2H, q, J = 7.4 Hz), 2.94 (2H, t, J = 7.6 Hz), 7.30 (2H, d, J = 8.4 Hz) , 7.87 (2H, d, J = 8.4 Hz), 9.60 (1H, bs).
元素分析値: C21H27N303S3として Elemental analysis: as C 21 H 27 N 3 0 3 S 3
計算値 (%) : C, 54.17; H, 5.84; N, 9.02; S, 20.66. Calculated value (%): C, 54.17; H, 5.84; N, 9.02; S, 20.66.
実測値 (%) : C, 54.09; H, 5.96; N, 8.99; S, 20.69. Found (%): C, 54.09; H, 5.96; N, 8.99; S, 20.69.
実施例 7- 11 Example 7-11
N - [5, 6-ジメチル- 8- (イソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン ]-4-イソプロピルベンゼンスルホンァ ミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 4-イソプロピルベンゼンスルホニル クロリド (113mg> 0.504 mmol) を用いて、 実施例 6- 2で得られた 1-ァミノ- 5, 6 -ジメチル- 8- (イソペンチ ルスルファニル) - 3H -イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (100 mg, 0.336 腿 ol) から題記化合物 (109 mg, 68¾) を合成した。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 198- 200°Cの暗赤色結晶を得た。 N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene] -4-isopropylbenzenesulfonamide Example 7-5 was obtained in the same manner as in Method C, except that 4-isopropylbenzenesulfonyl chloride (113 mg> 0.504 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. The title compound (109 mg) was obtained from 1-amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.336 t). mg, 68¾). Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals with a melting point of 198-200 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 2959, 2928, 2892, 1744, 1728, 1663, 1607, 1551 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 2959, 2928, 2892, 1744, 1728, 1663, 1607, 1551 cm— 1 .
¾-NMR (CDC13) : δ 0.932 (6Η, d, J-6.2 Hz), 1.26 (6H, d, J=7.0 Hz), 1.50-1.76 (3H, m), 1.79 (3H, d, J=1.0 Hz), 2.27 (3H, d, J=1.0 Hz), 2.95 (2H, t, J=7.8 Hz), 2.89-3.03 (1H, m), 7.33 (2H, d, J=8.2 Hz), 7.88 (2H, d, J=8.4 Hz), 9.61 (1H, bs). ¾-NMR (CDC1 3): δ 0.932 (6Η, d, J-6.2 Hz), 1.26 (6H, d, J = 7.0 Hz), 1.50-1.76 (3H, m), 1.79 (3H, d, J = 1.0 Hz), 2.27 (3H, d, J = 1.0 Hz), 2.95 (2H, t, J = 7.8 Hz), 2.89-3.03 (1H, m), 7.33 (2H, d, J = 8.2 Hz), 7.88 (2H, d, J = 8.4 Hz), 9.61 (1H, bs).
元素分析値: C22H29N303S3として Elementary analysis: as C 22 H 29 N 3 0 3 S 3
計算値 (%) : C, 55.09; H, 6.09; N, 8.76; S, 20.05. Calculated value (%): C, 55.09; H, 6.09; N, 8.76; S, 20.05.
実測値 (¾) : C, 55.11; H, 6.14; N, 8.71; S, 19.98. Found (¾): C, 55.11; H, 6.14; N, 8.71; S, 19.98.
実施例 7-12 , Example 7-12,
3-クロ口- N- [5, 6-ジメチル- 8- (ィソペンチルスルファニル) -3-ォキソ -2, 3-ジヒ ドロ - 1H-イミダゾ [5,l_c] [1,4]チアジン- 1 -ィリデン]ベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 3-クロ口ベンゼンスルホニル クロリド (145 mg, 0.672顧 ol ) を用いて、 実施例 6 - 2で得られた 1 -アミノ- 5, 6-ジメチル- 8- (イソペンチルスル ファニル) - 3H -イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (100 mg, 0.336 mmol) か ら題記化合物 (116 mg, 74¾) を合成した。 酢酸ェチル-ジェチルエーテルから再 結晶して融点 154-156 の暗赤色結晶を得た。 3-chloro-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine-1 -Ylidene] benzenesulfonamide Example 7-5 Method was performed in the same manner as in Method C, except that benzenesulfonyl chloride (145 mg, 0.672) was used instead of 4-bromomethylbenzenesulfonyl chloride. 1-Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one obtained in Example 6-2 (100 mg, 0.336 mmol) to give the title compound (116 mg, 74%). The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals having a melting point of 154-156.
赤外吸収スペクトル(IR) (KBr中) V 3229, 2955, 2907, 2870, 1755, 1661, 1620, 1557 cm— Ή- R (CDC13) : δ 0.932 (6Η, d, J=6.6Hz), 1.46-1.76 (3H, m), 1.80 (3H, d, J=1.0 Hz), 2.28 (3H, d, J-1.2 Hz), 2.97 (2H, t, J=7.8 Hz), 7.38-7.56 (2H, m), 7.85 (1H, dt, J=7.8, 2.0 Hz), 7.99 (1H, t, J=2.0Hz), 9.58 (1H, bs). 元素分析値: C19H22N303S3C1として 計算値 (%) : C, 48.34; H, 4.70; N, 8.90; S, 20.38. Infrared absorption spectrum (IR) (in KBr) V 3229, 2955, 2907 , 2870, 1755, 1661, 1620, 1557 cm- Ή- R (CDC1 3): δ 0.932 (6Η, d, J = 6.6Hz), 1.46-1.76 (3H, m), 1.80 (3H, d, J = 1.0 Hz), 2.28 (3H, d, J-1.2 Hz), 2.97 (2H, t, J = 7.8 Hz), 7.38-7.56 (2H , m), 7.85 (1H, dt, J = 7.8, 2.0 Hz), 7.99 (1H, t, J = 2.0 Hz), 9.58 (1H, bs). Elemental analysis: C 19 H 22 N 3 0 3 S 3 As C1 Calculated value (%): C, 48.34; H, 4.70; N, 8.90; S, 20.38.
実測値 (%) : C, 48.39; H, 4.46; N, 8.86; S, 20.37. Found (%): C, 48.39; H, 4.46; N, 8.86; S, 20.37.
実施例 7 - 13 Example 7-13
2 -クロ口 -N- [5, 6 -ジメチル- 8- (ィソぺンチルスルファニル) -3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜イリデン]ベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 2_クロ口ベンゼンスルホニル クロリド (145 mg, 0.672匪 ol) を用いて、 実施例 6-2で得られた 1 -ァミノ _5, 6 -ジメチル- 8- (イソペンチルスル ファニル) - 3H -イミダゾ [5,卜 c] [1, 4]チアジン -3-オン (100 mg, 0.336丽 ol) か ら題記化合物 (113 mg, 72¾) を合成した。 酢酸ェチル-ジェチルェ一テルから再 結晶して融点 178-180°Cの暗赤色結晶を得た。 2-chloro-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] Thiazine-triylidene] benzenesulfonamide Example 7-5 Method 2-5 was used in the same manner as in Method C, except that 4-bromomethylbenzenesulfonyl chloride was used instead of 2-bromobenzenebenzenesulfonyl chloride (145 mg, 0.672 bandol). Thus, 1-amino_5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (100) obtained in Example 6-2 mg, 0.336 mol) to give the title compound (113 mg, 72 mol). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 178-180 ° C.
赤外吸収スペクトル(IR) (KBr中) ソ : 3245, 2957, 2909, 2872, 1728, 1661, 1607, 1549 cm—1. Infrared absorption spectrum (IR) (in KBr) So: 3245, 2957, 2909, 2872, 1728, 1661, 1607, 1549 cm- 1 .
Ή-NMR (CDC13) : δ 0.894 (6Η, d, J=6.2 Hz), 1.46-1.73 (3H, m), 1.81 (3H, s), 2.30 (3H, d, J=1.0Hz), 2.92 (2H, t, J=7.6 Hz), 7.36-7.54 (3H, m), 8.20 (1H, d, J=6.6 Hz), 9.74 (1H, bs). Ή-NMR (CDC1 3): δ 0.894 (6Η, d, J = 6.2 Hz), 1.46-1.73 (3H, m), 1.81 (3H, s), 2.30 (3H, d, J = 1.0Hz), 2.92 (2H, t, J = 7.6 Hz), 7.36-7.54 (3H, m), 8.20 (1H, d, J = 6.6 Hz), 9.74 (1H, bs).
元素分析値: C19H22N303S3CIとして Elementary analysis: as C 19 H 22 N 3 0 3 S 3 CI
計算値 (¾) : C, 48.34; H, 4.70; N, 8.90; S, 20.38. Calculated (¾): C, 48.34; H, 4.70; N, 8.90; S, 20.38.
実測値 (%) : C, 48.16; H, 4.66; N, 8.92; S, 20.34. Found (%): C, 48.16; H, 4.66; N, 8.92; S, 20.34.
実施例 7-14 Example 7-14
N- [5, 6 -ジメチル- 8- (ィソぺンチルスルファニル) -3-ォキソ -2, 3 -ジヒド口- 1H -ィ ミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン ] -4- (トリフルォロメトキシ)ベンゼン スルホンアミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydrido-1H-dimidazo [5,1-c] [1,4] thiazine-1 -Ylidene] -4- (trifluoromethoxy) benzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4-トリフルォロメトキシベンゼンスルホニル クロリド(0.116 ml, 0.672 mmol) を用いて、 実施例 6-2で得られた卜アミノ- 5, 6-ジメチル -8 -(ィ ソペンチルスルファ二ル)- 3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (100 mg, 0.336腿 ol) から題記化合物 (152 mg, 87%) を合成した。 酢酸ェチル -ジェチル エーテルから再結晶して融点 200-201°Cの暗赤色結晶を得た。 赤外吸収スぺクトル(IR) (KBr中) V: 3227, 2959, 2932, 2913, 2872, 1759, 1748, 1663, 1620, 1559, 1495 cm一1. Example 7-5 Method In the same manner as in Method C, but using 4-trifluoromethoxybenzenesulfonyl chloride (0.116 ml, 0.672 mmol) instead of 4-bromomethylbenzenesulfonyl chloride, the procedure of Example 6-2 was repeated. The resulting triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.336 t ol )) To give the title compound (152 mg, 87%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 200-201 ° C. Infrared absorption spectrum (IR) (in KBr) V: 3227, 2959, 2932, 2913, 2872, 1759, 1748, 1663, 1620, 1559, 1495 cm- 1 .
¾一 NMR (CDC13) : 6 0.933 (6H, d, J-6.2 Hz), 1.50-1.78 (3H, m), 1.80 (3H, d, J=1.0 Hz), 2.28 (3H, d, J=1.0 Hz), 2.96 (2H, t, J=7.6 Hz), 7.31 (2H, d, J=8.0 Hz), 8.02 (2H, d, J=8.8 Hz), 9.60 (1H, bs). ¾ one NMR (CDC1 3): 6 0.933 (6H, d, J-6.2 Hz), 1.50-1.78 (3H, m), 1.80 (3H, d, J = 1.0 Hz), 2.28 (3H, d, J = 1.0 Hz), 2.96 (2H, t, J = 7.6 Hz), 7.31 (2H, d, J = 8.0 Hz), 8.02 (2H, d, J = 8.8 Hz), 9.60 (1H, bs).
元素分析値: C2。H22N304S として Elemental analysis: C 2. As H 22 N 3 0 4 S
計算値 (%) : C, 46.05; H, 4.25; N, 8.06; S, 18.44. Calculated value (%): C, 46.05; H, 4.25; N, 8.06; S, 18.44.
実測値 (%) : C, 45.78; H, 4.43; N, 8.16; S, 18.44. Found (%): C, 45.78; H, 4.43; N, 8.16; S, 18.44.
実施例 7-15 Example 7-15
N- [4- ({[5,6-ジメチル- 8- (イソペンチルスルファ二ル) -3-ォキソ -2, 3-ジヒドロ - 1H -ィミダゾ [5, 1 - c] [1,4]チアジン- 1-ィリデン]ァミノ }スルホニル)フエニル] ァセトアミド N- [4-({[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene] amino} sulfonyl) phenyl] acetamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 4-ァセトアミドベンゼンスルホニル クロリド (157 mg, 0.672 mmol) を用いて、 実施例 6-2で得られた卜アミノ- 5, 6-ジメチル _8 - (イソペンチ ルスルファ二ル) - 3H -イミダゾ [5,卜 c] [1,4]チアジン _3 -オン (100 mg, 0.336 mmol) から題記化合物 (154 mg, 93¾) を合成した。 酢酸ェチル -ジェチルェ一テ ル-テトラヒドロフランから再結晶して融点 213- 215°Cの暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, but using 4-acetamidobenzenesulfonyl chloride (157 mg, 0.672 mmol) instead of 4-bromomethylbenzenesulfonyl chloride, the procedure of Example 6-2 was repeated. The title compound (154 mg) was obtained from the obtained triamino-5,6-dimethyl_8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin_3-one (100 mg, 0.336 mmol). mg, 93¾). Recrystallization from ethyl acetate-ethyl ether-tetrahydrofuran gave dark red crystals with a melting point of 213-215 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3343, 3279, 3189, 2955, 2930, 2915, 2872, 1752, 1738, 1692, 1667, 1615, 1591, 1559, 1537, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3343, 3279, 3189, 2955, 2930, 2915, 2872, 1752, 1738, 1692, 1667, 1615, 1591, 1559, 1537, 1497 cm " 1 .
'H-NMR (CDC13) : δ 0.934 (6Η, d, J=6.2 Hz), 1.50—1.78 (3H, m), 1.79 (3H, d, J=0.8 Hz), 2.22 (3H, s), 2.27 (3H, d, J=1.2 Hz), 2.94 (2H, t, J=7.2 Hz), 7.34 (1H, bs), 7.63 (2H, d, J=9.2 Hz), 7.91 (2H, d, J=8.8 Hz), 9.59 (1H, bs). 実施例 7-16 'H-NMR (CDC1 3) : δ 0.934 (6Η, d, J = 6.2 Hz), 1.50-1.78 (3H, m), 1.79 (3H, d, J = 0.8 Hz), 2.22 (3H, s), 2.27 (3H, d, J = 1.2 Hz), 2.94 (2H, t, J = 7.2 Hz), 7.34 (1H, bs), 7.63 (2H, d, J = 9.2 Hz), 7.91 (2H, d, J = 8.8 Hz), 9.59 (1H, bs).
、 N- [5,6-ジメチル -8_ (イソペンチルスルファニル) -3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5,卜 c] [1, 4]チアジン - 1 -ィリデン ]-3, 4 -ジメトキシベンゼンスルホンァ ミド , N- [5,6-dimethyl-8_ (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene] -3 , 4-Dimethoxybenzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 3, 4-ジメトキシベンゼンスルホニル クロリド (130 mg, 0.538 IMOI) を用いて、 実施例 6- 2で得られた卜アミノ- 5, 6-ジメチル- 8- (イソペンチ ルスルファニル) -3H-イミダゾ [5,1- c] [1,4]チアジン- 3-オン (80.0 mg, 0.269 mmol) から題記化合物 Q23 mg, 92%) を合成した。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 160-162°Cの暗赤色結晶を得た。 Example 7-5 By the same method as in Method C, 3,4-dimethoxybenzenesulfonyl chloride (130 mg, 0.538 mg) was used instead of 4-bromomethylbenzenesulfonyl chloride. Using the IMOI), the triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazine-3 obtained in Example 6-2 The title compound Q23 mg, 92%) was synthesized from -one (80.0 mg, 0.269 mmol). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 160-162 ° C.
赤外吸収スペクトル(IR) (KBr中) V : 3362, 2953, 1740, 1665, 1620, 1563, 1508 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3362, 2953, 1740, 1665, 1620, 1563, 1508 cm " 1 .
Ή-NMR (CDC13) : δ 0.924 (6Η, d, J-6.4 Hz), 1.48-1.78 (3H, m), 1.79 (3H, d, J=0.8 Hz), 2.27 (3H, d, J=1.0 Hz), 2.95 (2H, t, J=7.8 Hz), 3.93 (3H, s), 3.94 (3H, s), 6.91 (1H, d, J=8. Hz), 7.45 (1H, d, J=1.8Hz), 7.57 (2H, dd, J=8.4, 2.2 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 0.924 (6Η, d, J-6.4 Hz), 1.48-1.78 (3H, m), 1.79 (3H, d, J = 0.8 Hz), 2.27 (3H, d, J = 1.0 Hz), 2.95 (2H, t, J = 7.8 Hz), 3.93 (3H, s), 3.94 (3H, s), 6.91 (1H, d, J = 8.Hz), 7.45 (1H, d, J = 1.8Hz), 7.57 (2H, dd, J = 8.4, 2.2 Hz), 9.60 (1H, bs).
元素分析値: C21H„N305S3'0.4¾0として Elemental analysis: C 21 H „N 3 0 5 S 3 '0.4¾0
計算値 (¾) : C, 49.96; H, 5.55; N, 8.32; S, 19.05. Calculated (¾): C, 49.96; H, 5.55; N, 8.32; S, 19.05.
実測値 (¾) : C, 50.17; H, 5.44; N, 8.10; S, 18.71. Found (¾): C, 50.17; H, 5.44; N, 8.10; S, 18.71.
実施例 7-17 Example 7-17
4-ブ口モ- N- [5, 6 -ジメチル- 8- (ィソペンチルスルファニル) -3-ォキソ- 2, 3-ジヒ ド口- m-ィミダゾ [5,卜 c] [1 , 4]チアジン -卜ィリデン]ベンゼンスルホンアミド 水素化ナトリウム(269 mg, 6.72醒01) (60¾油性)のテトラヒドロフラン(10.0 ml) の懸濁液に、 実施例 6-2で得られた卜アミノ- 5, 6-ジメチル- 8- (イソペンチ ルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン(1.00 g, 3.36匪 ol) のテトラヒドロフラン (5.00ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (1.75 g, 6.72腿01) のテトラヒドロフ ラン (5.00 ml) 溶液を加え、 さらに 3時間かき混ぜた。 反応混合物に 1N塩酸を 加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に 留去して題記化合物 (1.61 g, 93¾) を合成した。 酢酸エヂル-ジェチルエーテル から再結晶して融点 178- 180°Cの暗赤色結晶を得た。 4-Bumo-N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-m-imidazo [5, tric] [1,4] Thiazine-triylidene] benzenesulfonamide To a suspension of sodium hydride (269 mg, 6.72 Aging 01) (60¾ oily) in tetrahydrofuran (10.0 ml) was added triamino-5,6 obtained in Example 6-2. A solution of -dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (1.00 g, 3.36 bandol) in tetrahydrofuran (5.00 ml) was added. The mixture was stirred for 5 minutes under ice cooling. To this was added a solution of 4-bromobenzenesulfonyl chloride (1.75 g, 6.72 / 1) in tetrahydrofuran (5.00 ml), and the mixture was further stirred for 3 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (1.61 g, 93¾). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 178-180 ° C.
赤外吸収スぺクトル(IR) (KBr中) y: 3229, 2955, 2926, 1759, 1663, 1653, 1622, 1559 cm一1. Infrared absorption spectrum (IR) (in KBr) y: 3229, 2955, 2926, 1759, 1663, 1653, 1622, 1559 cm- 1 .
Ή-NMR (CDC13) : δ 0.935 (6Η, d, J=6.4 Hz), 1.46-1.78 (3H, m), 1.80 (3H, d, J=1.0 Hz), 2.28 (3H, d, J=0.6 Hz), 2.95 (2H, t, J=7.2 Hz), 7.63 (2H, d, J=8.8 Hz), 7.84 (2H, d, J=8.8 Hz), 9.59 (1H, bs). Ή-NMR (CDC1 3): δ 0.935 (6Η, d, J = 6.4 Hz), 1.46-1.78 (3H, m), 1.80 (3H, d, J = 1.0 Hz), 2.28 (3H, d, J = 0.6 Hz), 2.95 (2H, t, J = 7.2 Hz), 7.63 (2H, d, J = 8.8 Hz), 7.84 (2H, d, J = 8.8 Hz), 9.59 (1H, bs).
元素分析値: C19H22N303S3Brとして Elementary analysis: as C 19 H 22 N 3 0 3 S 3 Br
計算値 (%) : C, 44.18; H, 4.29; N, 8.14; S, 18.63. Calculated (%): C, 44.18; H, 4.29; N, 8.14; S, 18.63.
実測値 (¾) : C, 44.12; H, 4.23; N, 8.21; S, 18.56. Found (¾): C, 44.12; H, 4.23; N, 8.21; S, 18.56.
実施例 7 - 18 Examples 7-18
N - [5, 6-ジメチル- 8- (イソペンチルスルファニル) - 3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン] -4-ビニルベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに P-スチレンスルホニル クロリド (88.7 mg, 0.538廳 ol) を用 いて、実施例 6- 2で得られた卜ァミノ- 5, 6 -ジメチル 8- (イソペンチルスルファ二 ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3 -オン (80.0 mg, 0.269匪 ol) から題記 化合物 (25.5mg, 20¾) を合成した。 酢酸ェチル-ジェチルェ一テルから再結晶し て融点 173- 175°Cの暗赤色結晶を得た。 N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene]- 4-Vinylbenzenesulfonamide Example 7-5 In the same manner as in Method C, except that P-styrenesulfonyl chloride (88.7 mg, 0.538 ol) was used instead of 4-bromomethylbenzenesulfonyl chloride. Triamino-5,6-dimethyl 8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in 6-2 (80.0 mg, The title compound (25.5 mg, 20 mg) was synthesized from 0.269 bandol ol). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 173-175 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3218, 2953, 2903, 2872, 1746, 1661, 1620, 1559 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3218, 2953, 2903, 2872, 1746, 1661, 1620, 1559 cm- 1 .
'Η -腿 (CDC") : δ 0.928 (6Η, d, J=6.2 Hz), 1.50-1.78 (3H, m), 1.79 (3H, d, J=1.0Hz), 2.27 (3H, d, J=1.2Hz), 2.95 (2H, t, J=7.6 Hz), 5.41 (1H, d, J=11.0 Hz), 5.87 (1H, d, J=17.6 Hz), 6.75 (1H, dd, J=18.0, 10.5 Hz), 7.50 (2H, d, J=8.4 Hz), 7.92 (2H, d, J=8.4 Hz), 9.63 (1H, bs). 'Η-thigh (CDC "): δ 0.928 (6Η, d, J = 6.2 Hz), 1.50-1.78 (3H, m), 1.79 (3H, d, J = 1.0 Hz), 2.27 (3H, d, J = 1.2Hz), 2.95 (2H, t, J = 7.6 Hz), 5.41 (1H, d, J = 11.0 Hz), 5.87 (1H, d, J = 17.6 Hz), 6.75 (1H, dd, J = 18.0) , 10.5 Hz), 7.50 (2H, d, J = 8.4 Hz), 7.92 (2H, d, J = 8.4 Hz), 9.63 (1H, bs).
元素分析値: C21H25N303S3として Elemental analysis: as C 21 H 25 N 3 0 3 S 3
計算値 %) : C, 54.40; H, 5.43; N, 9.06; S, 20.75. (Calculated%): C, 54.40; H, 5.43; N, 9.06; S, 20.75.
実測値 (¾) : C, 54.13; H, 5.62; N, 8.92; S, 20.65. Found (¾): C, 54.13; H, 5.62; N, 8.92; S, 20.65.
実施例 7 - 19 Examples 7 to 19
N - [5, 6-ジメチル -8- (イソペンチルスルファニル) -3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5,1-c] [1,4]チアジン -卜ィリデン] [1, 1'-ビフエ二ル] -4-スルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4-ビフエニルスルホニル クロリド (156 mg, 0.555 mmol) を 用いて、実施例 6-2で得られた 1 -ァミノ- 5, 6-ジメチル 8- (ィソペンチルスルファ 二ル)- 3H-イミダゾ [5,1- c] [1,4]チアジン -3-オン (82.5 mg, 0.277 mmol) から題 記化合物 (110 mg, 78%) を合成した。 酢酸ェチル -ジェチルェ一テルから'再結晶 して融点 183- 185Cの暗赤色結晶を得た。 N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-dimidazo [5,1-c] [1,4] thiazine-triylidene] [1 , 1'-biphenyl] -4-sulfonamide Example 7-5 Method 4-Biphenylsulfonyl chloride (156 mg, 0.555 mmol) in place of 4-bromomethylbenzenesulfonyl chloride in the same manner as in Method C Was used to obtain 1-amino-5,6-dimethyl 8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazine obtained in Example 6-2. Title from -3-one (82.5 mg, 0.277 mmol) The title compound (110 mg, 78%) was synthesized. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals, mp 183-185C.
赤外吸収スペクトル(IR) (KBr中) リ: 3239, 2955, 2828, 2868, 1738, 1663, 1615, 1559 cm"1. Infrared absorption spectrum (IR) (in KBr): 3239, 2955, 2828, 2868, 1738, 1663, 1615, 1559 cm " 1 .
Ή-NMR (CDC13) : δ 0.928 (6Η, d, J=6.2 Hz), 1.50-1.76 (3H, m), 1.79 (3H, s), Ή-NMR (CDC1 3): δ 0.928 (6Η, d, J = 6.2 Hz), 1.50-1.76 (3H, m), 1.79 (3H, s),
2.28 (3H, s), 2.96 (2H, t, J=7.6 Hz), 7.39-7.54 (3H, m), 7.56-7.64 (2H, m),2.28 (3H, s), 2.96 (2H, t, J = 7.6 Hz), 7.39-7.54 (3H, m), 7.56-7.64 (2H, m),
7.69 (2H, d, J=8.4 Hz), 8.03 (2H, d, J=8.4 Hz), 9.65 (1H, bs). 7.69 (2H, d, J = 8.4 Hz), 8.03 (2H, d, J = 8.4 Hz), 9.65 (1H, bs).
元素分析値: C25H27N303S3として Elemental analysis: as C 25 H 27 N 3 0 3 S 3
計算値 (¾) : C, 57.84; H, 5.36; N, .8.09. Calculated value (¾): C, 57.84; H, 5.36; N, .8.09.
実測値 (¾) : C, 58.15; H, 5.66; N, 7.85. Found (¾): C, 58.15; H, 5.66; N, 7.85.
実施例 7-20 Example 7-20
N - [5, 6-ジメチル -8 (イソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H-ィ ミダゾ [5,1 c] [1, 4]チアジン - イリデン] (フエニル)メタンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりにベンジルスルホニル クロリド(128 nig, 0.672 mmol)を用いて、 実施例 6-2 で得られた 1-ァミノ- 5, 6 -ジメチル 8 -(イソペンチルスルファ二 ル)- 3H-イミダゾ [5, 1- c] [1,¾チアジン- 3-オン (100 mg, 0.336 mmol) から題記 化合物 (104mg, 69¾) を合成した。 酢酸ェチル -ジェチルェ一テル-テトラヒドロ フランから再結晶して融点 188- 190°Cの暗赤色結晶を得た。 N- [5,6-Dimethyl-8 (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1c] [1,4] thiazine-ylidene] (phenyl ) Methanesulfonamide Example 7-5 was obtained in the same manner as in Method C, except that benzylsulfonyl chloride (128 nig, 0.672 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. 1-Amino-5,6-dimethyl 8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,1¾thiazin-3-one (100 mg, 0.336 mmol) to give the title compound (104 mg, 69 mg) was synthesized. Recrystallization from ethyl acetate-ethyl ether-tetrahydrofuran gave dark red crystals with a melting point of 188-190 ° C.
赤外吸収スペクトル(IR) (KBr中) レ: 3139, 2957, 2928, 1732, 1663, 1615, 1561, 1541 cm-1. Infrared absorption spectrum (IR) (in KBr) d: 3139, 2957, 2928, 1732, 1663, 1615, 1561, 1541 cm- 1 .
Ή-NMR (CDCI3) : δ 0.974 (6Η, d, J=6.2 Hz), 1.50-1.78 (3H, m), 1.80 (3H, d, J=0.8 Hz), 2.21 (3H, d, ]= 2 Hz), 3.00 (2H, i, J=8.0 Hz), 4.35 (2H, s), 7.30-7.47 (5H, m), 9.11 (1H, bs). Ή-NMR (CDCI3): δ 0.974 (6Η, d, J = 6.2 Hz), 1.50-1.78 (3H, m), 1.80 (3H, d, J = 0.8 Hz), 2.21 (3H, d,] = 2 Hz), 3.00 (2H, i, J = 8.0 Hz), 4.35 (2H, s), 7.30-7.47 (5H, m), 9.11 (1H, bs).
元素分析値: C2。 3N303S3として Elemental analysis: C 2. 3 N 3 0 3 S 3
計算値 (%) : C, 53.43; H, 5.16; N, 9.35; S, 21.40. Calculated value (%): C, 53.43; H, 5.16; N, 9.35; S, 21.40.
実測値 (%) : C, 53.07; H, 5.50; N, 9.24; S, 21.23. Found (%): C, 53.07; H, 5.50; N, 9.24; S, 21.23.
実施例 7 - 21 Example 7-21
N - [5, 6-ジメチル- 8- (イソペンチルスルファニル) -3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, 1-c] [1, 4]チアジン - 1 ィリデン] -卜ブタンスルホンアミド 実施例 7-2方法 Bと同様の方法により、 4_トリフルォロメチルベンゼンスルホ ニル クロリドの代わりに卜ブタンスルホニル クロリド(0.0872 ml, 0.672 mmol) を用いて、 実施例 6-2で得られた卜アミノ- 5, 6-ジメチル- 8 - (イソペンチルスル ファニル)- 3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (100 mg, 0.336 mmol ) か ら題記化合物 (96.0mg, 68%) を合成した。 酢酸ェチル-ジェチルェ一テルから再 結晶して融点 180- 182°Cの暗赤色結晶を得た。 N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-di Midazo [5,1-c] [1,4] thiazine-1-ylidene] -tobutanesulfonamide Example 7-2 Method 4-Trifluoromethylbenzenesulfonyl chloride instead of 4_trifluoromethylbenzenesulfonyl chloride Using butanesulfonyl chloride (0.0872 ml, 0.672 mmol), triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo obtained in Example 6-2 [5, tric] The title compound (96.0 mg, 68%) was synthesized from [1,4] thiazin-3-one (100 mg, 0.336 mmol). Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 180-182 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3156, 2963, 2922, 2872, 1753, 1736, 1663, 1622, 1572 cm"1. Infrared absorption spectrum (IR) (in KBr): 3156, 2963, 2922, 2872, 1753, 1736, 1663, 1622, 1572 cm " 1 .
¾ -雇 R (CDC13) : δ 0.945 (6H, d, J=6.2Hz), 0.951 (3H, t, J=7.0Hz), 1.40-1.78 (5H, m), 1.81 (3H, d, J-1.0 Hz), 1.78-1.96 (2H, m), 2.28 (3H, d, J=0.6 Hz), 2.98 (2H, t, J=7.8 Hz), 3.12 (2H, t, J=8.0 Hz), 9.45 (1H, bs). ¾ - employment R (CDC1 3): δ 0.945 (6H, d, J = 6.2Hz), 0.951 (3H, t, J = 7.0Hz), 1.40-1.78 (5H, m), 1.81 (3H, d, J -1.0 Hz), 1.78-1.96 (2H, m), 2.28 (3H, d, J = 0.6 Hz), 2.98 (2H, t, J = 7.8 Hz), 3.12 (2H, t, J = 8.0 Hz), 9.45 (1H, bs).
元素分析値: C17H27N303S3として Elementary analysis: as C 17 H 27 N 3 0 3 S 3
計算値 (¾) : C, 48.89; H, 6.52; N, 10.06; S, 23.04. Calculated value (¾): C, 48.89; H, 6.52; N, 10.06; S, 23.04.
実測値 (¾) : C, 48.81; H, 6.24; N, 10.33; S, 22.99. Found (¾): C, 48.81; H, 6.24; N, 10.33; S, 22.99.
実施例 7 - 22 Examples 7 to 22
N- [5,6-ジメチル- 8- (イソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, 1-c] [1,4]チアジン -卜ィリデン ]-2, 2, 2-トリフルォロエタンスルホン アミド N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-trilidene ] -2,2,2-Trifluoroethanesulfonamide
実施例 7-5方法 Cと同様の方法により、 4 -プロモメチルべンゼンスルホニル ク 口リ ドの代わりに 2, 2, 2-トリフルォロェ夕ンスルホニル クロリド (129 mg, 0.672 mmol) を用いて、 実施例 6-2で得られた卜アミノ- 5, 6 -ジメチル- 8- (イソ ペンチルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3-オン (100 mg, 0.336腿 ol) から題記化合物 (5.8 mg, 3.9%) を合成した。 n -へキサンから再結 晶して融点 161- 163°Cの暗赤色結晶を得た。 Example 7-5 Method was carried out in the same manner as in Method C, except that 2,2,2-trifluorobenzenesulfonyl chloride (129 mg, 0.672 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. Triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 The title compound (5.8 mg, 3.9%) was synthesized from the compound (0.3 mg, 0.336 t). Recrystallization from n-hexane gave dark red crystals with a melting point of 161-163 ° C.
赤外吸収スペクトル(IR) (KBr中) ソ : 3202, 2959, 2928, 2874, 1750, 1661, 1597, 1557 cnf1. Infrared absorption spectrum (IR) (in KBr) So: 3202, 2959, 2928, 2874, 1750, 1661, 1597, 1557 cnf 1 .
lH - NMR (CDC13) : δ 0.950 (6Η, d, J=6.2 Hz), 1.44-1.78 (3H, m), 1.82 (3H, d, J=1.0 Hz), 2.31 (3H, d, J=1.2 Hz), 3.00 (2H, t, J-7.6 Hz), 3.91 (2H, q, J=9.2 Hz), 9.51 (1H, bs). ' lH - NMR (CDC1 3): δ 0.950 (6Η, d, J = 6.2 Hz), 1.44-1.78 (3H, m), 1.82 (3H, d, J = 1.0 Hz), 2.31 (3H, d, J = 1.2 Hz), 3.00 (2H, t, J-7.6 Hz), 3.91 (2H, q, J = 9.2 Hz), 9.51 (1H, bs).
実施例 7-23 Example 7-23
N - [5, 6-ジメチル- 8- (イソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, l_c] [1,4]チアジン- 1-イリデン] -卜ナフタレンスルホンアミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine-1-ylidene] -Tonaphthalene sulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 卜ナフタレンスルホニル クロリド (122 mg, 0.538 mmol) を 用いて、 実施例 6-2で得られた卜ァミノ- 5,6-ジメチル -8 - (イソペンチルスルフ ァニル) - 3H-イミダゾ [5,卜 c] [1,4]チアジン _3 -オン (80.0 mg, 0.269 mmol) から 題記化合物 (114mg, 87%) を合成した。 酢酸ェチル -ジェチルエーテル-から再結 晶して融点 190- 192°Cの暗赤色結晶を得た。 Example 7-5 Triamino obtained in Example 6-2 was prepared in the same manner as in Method C, except that tronaphthalenesulfonyl chloride (122 mg, 0.538 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. -5,6-Dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin_3-one (80.0 mg, 0.269 mmol) from the title compound (114 mg, 87% ) Was synthesized. Recrystallization from ethyl acetate-getyl ether-gave dark red crystals with a melting point of 190-192 ° C.
赤外吸収スペクトル(IR) (KBr中) レ : 3260, 2957, 1732, 1661, 1613, 1553, 1507 cm—1. Infrared absorption spectrum (IR) (in KBr) Re: 3260, 2957, 1732, 1661, 1613, 1553, 1507 cm- 1 .
Ή-NMR (CDC13) : δ 0.905 (6Η, d, J=6.6 Hz), 1.42-1.74 (3H, in), 1.75 (3H, d, Ή-NMR (CDC1 3): δ 0.905 (6Η, d, J = 6.6 Hz), 1.42-1.74 (3H, in), 1.75 (3H, d,
J=1.2 Hz), 2.25 (3H, d, J=0.6 Hz), 2.90 (2H, t, J=7.8 Hz), 7.50-7.68 (3H, m), 7.88-7.95 (1H, m), 8.05 (1H, dd, J=8.0, 1.2 Hz), 8.29 (1H, dd, J=7.4,J = 1.2 Hz), 2.25 (3H, d, J = 0.6 Hz), 2.90 (2H, t, J = 7.8 Hz), 7.50-7.68 (3H, m), 7.88-7.95 (1H, m), 8.05 ( 1H, dd, J = 8.0, 1.2 Hz), 8.29 (1H, dd, J = 7.4,
1.0 Hz), 8.88-8.98 (1H, m), 9.67 (1H, bs). 1.0 Hz), 8.88-8.98 (1H, m), 9.67 (1H, bs).
元素分析値: C23H25N303S3: として Elemental analysis: C 23 H 25 N 3 0 3 S 3: as
計算値 (%) : C, 56.65; H, 5.17; N, 8.62; S, 19.73. Calculated value (%): C, 56.65; H, 5.17; N, 8.62; S, 19.73.
実測値 (%) : C, 56.52; H, 4.99; N, 8.82; S, 19.62. Found (%): C, 56.52; H, 4.99; N, 8.82; S, 19.62.
実施例 7 - 24 Examples 7-24
N - [5, 6-ジメチル -8- (ィソペンチルスルファニル) -3-ォキソ -2, 3-ジヒド口- 1H-ィ ミダゾ [5, 1- c] [1, ]チアジン- 1-ィリデン] -2 -ナフタレンスルホンアミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,] thiazine-1-ylidene] -2-Naphthalenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 2-ナフタレンスルホニル クロリド (91.4 mg, 0.403腿 ol) を 用いて、 実施例 6-2で得られた卜ァミノ- 5, 6-ジメチル -8- (イソペンチルスルフ ァニル) - 3H-イミダゾ [5, 1 - c]〔1,4]チアジン- 3 -オン (80.0 mg, 0.269 mmol) から 題記化合物 (85.4mg, 87¾) を合成した。 酢酸ェチル-ジェチルエーテルから再結 晶して融点 172- 174°Cの暗赤色結晶を得た。 Example 7-5 According to the method similar to that of Method C, 2-naphthalenesulfonyl chloride (91.4 mg, 0.403 tmol) was used in place of 4-bromomethylbenzenesulfonyl chloride to obtain Example 6-2. Title compound from triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (80.0 mg, 0.269 mmol) (85.4 mg, 87 mg) was synthesized. The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals having a melting point of 172-174 ° C.
赤外吸収スペクトル(IR) (KBr中) リ : 3231, 2957, 2915, 2867, 1736, 1663, 1615, 1557, 1505 cm"1. Infrared absorption spectrum (IR) (in KBr): 3231, 2957, 2915, 2867, 1736, 1663, 1615, 1557, 1505 cm " 1 .
Ή-NMR (CDC13) : δ 0.909 (6H, d, J=6.6 Hz), 1.48-1.76 (3H, m), 1.79 (3H, s), 2.27 (3H, d, J=1.2 Hz), 2.94 (2H, t, J=7.4 Hz), 7.58-7.70 (2H, m), 7.86-8.04 (4H, m), 8.52 (1H, s), 9:69 (1H, bs). Ή-NMR (CDC1 3): δ 0.909 (6H, d, J = 6.6 Hz), 1.48-1.76 (3H, m), 1.79 (3H, s), 2.27 (3H, d, J = 1.2 Hz), 2.94 (2H, t, J = 7.4 Hz), 7.58-7.70 (2H, m), 7.86-8.04 (4H, m), 8.52 (1H, s), 9:69 (1H, bs).
元素分析値: C23¾5N303S3として Elemental analysis: C 23 ¾ 5 N 3 0 3 S 3
計算値 (%) : C, 56.65; H, 5.17; N, 8.62; S, 19.73. Calculated value (%): C, 56.65; H, 5.17; N, 8.62; S, 19.73.
実測値 ( ) : C, 56.63; H, 5.27; N, 8.56; S, 19.67. Observed value (): C, 56.63; H, 5.27; N, 8.56; S, 19.67.
実施例 7-25 Example 7-25
N - [5, 6-ジメチル -8- (イソペンチルスルファニル) _3_ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, l-c] [1,4]チアジン - 1_イリデン] - 8_キノリンスルホンアミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) _3_oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-1_ylidene] -8_quinoline Sulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 8_キノリンスルホニル クロリド (91.4 mg, 0.403腿 ol) を用 いて、 実施例 6 - 2で得られた卜アミノ- 5, 6-ジメチル- 8- (イソペンチルスルファ ニル) - 3H-イミダゾ [5,1- c] [1,4]チアジン - 3_オン (70.0 mg, 0.235匪 ol) から題 記化合物 (74.3mg, 65%) を合成した。 酢酸ェチル -ジイソプロピルエーテルから 再結晶して融点 194- 195°Cの暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, except that 4-bromomethylbenzenesulfonyl chloride was replaced with 8_quinoline sulfonyl chloride (91.4 mg, 0.403 t ol), the product was obtained in Example 6-2. Title from triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3_one (70.0 mg, 0.235 bandol ol) The title compound (74.3 mg, 65%) was synthesized. Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals with a melting point of 194-195 ° C.
赤外吸収スペクトル(IR) (KBr中) ソ : 3364, 2957, 2930, 2909, 2870, 1738, 1663, 1615, 1557, 1495 cm— Infrared absorption spectrum (IR) (in KBr) So: 3364, 2957, 2930, 2909, 2870, 1738, 1663, 1615, 1557, 1495 cm—
¾-NMR (CDC13) : δ 0.853 (6Η, d, J=6.2 Hz), 1.40-1.70 (3H, m), 1.79 (3H, s), 2.32 (3H, s), 2.86 (2H, t, J=7.6 Hz), 7.51 (1H, dd, J=8.8, 4.8 Hz), 7.66 (1H, t, J=8.0 Hz), 8.03 (1H, dd, J=8.0, 1.4 Hz), 8.25 (1H, dd, J=8.0, 1.4 Hz), 8.64 (1H, dd, J=7.4, 1.4 Hz), 8.97 (1H, dd, J=4.4, 1.4 Hz), 11.19 (1H, bs). 元素分析値: C22H24N40sS3として ¾-NMR (CDC1 3): δ 0.853 (6Η, d, J = 6.2 Hz), 1.40-1.70 (3H, m), 1.79 (3H, s), 2.32 (3H, s), 2.86 (2H, t, J = 7.6 Hz), 7.51 (1H, dd, J = 8.8, 4.8 Hz), 7.66 (1H, t, J = 8.0 Hz), 8.03 (1H, dd, J = 8.0, 1.4 Hz), 8.25 (1H, dd, J = 8.0, 1.4 Hz), 8.64 (1H, dd, J = 7.4, 1.4 Hz), 8.97 (1H, dd, J = 4.4, 1.4 Hz), 11.19 (1H, bs). Elemental analysis: C 22 H 24 N 40 s As S 3
計算値 (%) : C, 54.07; H, 4.95; N, 11.47; S, 19.69. Calculated value (%): C, 54.07; H, 4.95; N, 11.47; S, 19.69.
実測値 (¾) : C, 54.09; H, 4.72; N, 11.35; S, 19.60. Observed value (¾): C, 54.09; H, 4.72; N, 11.35; S, 19.60.
実施例 7-26 Example 7-26
5-ク口ロ- N-[5, 6 -ジメチル- 8 -(ィソペンチルスルファニル) -3-ォキソ -2, 3 -ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン ]-2-チオフェンスルホンァ 5ド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 5-クロロチォフェン- 2-スルホニル クロリド (120 nig, 0.538 mmol) を用いて、 実施例 6 - 2で得られた 1-ァミノ- 5,6-ジメチル -8- (イソペンチ ルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (80.0 nig, 0.269 顧 ol) から題記化合物 (90.5mg, 70¾) を合成した。 酢酸ェチル-ジェチルェ一テ ルから再結晶して融点 154-156 の暗赤色結晶を得た。 5-cyclo-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene] -2-thiophenesulfonic acid Example 7-5 According to a method similar to that of Method C, 5-chlorothiophene-2-sulfonyl chloride (120 nig, 0.538 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride, and Example 6-2 was used. 1-Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (80.0 nig, 0.269 )) To give the title compound (90.5 mg, 70%). The crystals were recrystallized from ethyl acetate-ethyl ether to give dark red crystals having a melting point of 154-156.
赤外吸収スペクトル(IR) (KBr中) V : 3231, 2957, 1757, 1663, 1617, 1555, 1520 cm— Infrared absorption spectrum (IR) (in KBr) V: 3231, 2957, 1757, 1663, 1617, 1555, 1520 cm—
'H-NMR (CDC13) : δ 0.946 (6Η, d, J=6.2 Hz), 1.52-1.78 (3H, m), 1.81 (3H, d, J=0.6 Hz), 2.29 (3H, d, J=1.0 Hz), 2.98 (2H, t, J=7. Hz), 6.85 (1H, d, J=4.0 'H-NMR (CDC1 3) : δ 0.946 (6Η, d, J = 6.2 Hz), 1.52-1.78 (3H, m), 1.81 (3H, d, J = 0.6 Hz), 2.29 (3H, d, J = 1.0 Hz), 2.98 (2H, t, J = 7.Hz), 6.85 (1H, d, J = 4.0
Hz), 7.45 (1H, d, J-4.0 Hz), 9.51 (1H, bs). Hz), 7.45 (1H, d, J-4.0 Hz), 9.51 (1H, bs).
元素分析値: C17H20N303S4C1として Elementary analysis: as C 17 H 20 N 3 0 3 S 4 C1
計算値 (%) : C, 42.71; H, 4.22; N, 8.79; S, 26.83. Calculated value (%): C, 42.71; H, 4.22; N, 8.79; S, 26.83.
実測値 (¾) : C, 42.53; H, 4.04; N, 8.81; S, 26.91. Found (¾): C, 42.53; H, 4.04; N, 8.81; S, 26.91.
実施例 7 - 27 Example 7-27
2, 5-ジク口口- N - [5, 6-ジメチル- 8- (ィソペンチルスルファ二ル)- 3-ォキソ -2, 3- ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1 -ィリデン ]-3-チオフェンスルホ ンアミド 2,5-dic-mouth-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene] -3-thiophenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 2,5-ジクロロチォフェン- 3-スルホニル クロリド (144 mg, 0.535腿 ol) を用いて、 実施例 6-2で得られた 1-ァミノ- 5, 6 -ジメチル -8- (イソ ペンチルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (82.5 mg, 0.277讓 ol) から題記化合物 (104 mg, 73%) を合成した。 酢酸ェチル -ジェチル エーテルから再結晶して融点 168- 170°Cの暗赤色結晶を得た。 Example 7-5 Method was performed in the same manner as in Method C, except that 2,5-dichlorothiophene-3-sulfonyl chloride (144 mg, 0.535 t) was used instead of 4-bromomethylbenzenesulfonyl chloride. 1-Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-2 ( The title compound (104 mg, 73%) was synthesized from 82.5 mg (0.277 benzyl). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 168-170 ° C.
赤外吸収スぺクトル(IR) (KBr中) レ : 3337, 2961, 2926, 2868, 1744, 1661, 1611, 1559, 1510 cur1. Infrared absorption spectrum (IR) (in KBr) Re: 3337, 2961, 2926, 2868, 1744, 1661, 1611, 1559, 1510 cur 1 .
lH - NMR (CDC13) : δ 0.936 (6H, d, J=6.6 Hz), 1.50-1.78 (3H, m), 1.81 (3H, d, J=1.0 Hz), 2.30 (3H, d, J=0.8 Hz), 2.98 (2H, t, J=7.8Hz), 7.20 (1H, s), 9.63 (1H, bs). 元素分析値: C17Hl9N303S4C "として lH - NMR (CDC1 3): δ 0.936 (6H, d, J = 6.6 Hz), 1.50-1.78 (3H, m), 1.81 (3H, d, J = 1.0 Hz), 2.30 (3H, d, J = 0.8 Hz), 2.98 (2H, t, J = 7.8Hz), 7.20 (1H, s), 9.63 (1H, bs). Elementary analysis: as C 17 H l9 N 3 0 3 S 4 C "
計算値 (%) : C, 39.84; H, 3.74; N, 8.20; S, 25.03. Calculated value (%): C, 39.84; H, 3.74; N, 8.20; S, 25.03.
実測値 (¾) : C, 39.62; H, 3.65; N, 8.28; S, 25.02. Found (¾): C, 39.62; H, 3.65; N, 8.28; S, 25.02.
実施例 7 - 28 Examples 7-28
N_[5, 6-ジメチル -8- (イソペンチルスルファニル) -3-ォキソ -2, 3-ジヒドロ- 1H-ィ ミダゾ [5, 1 - c] [1, 4]チアジン-卜ィリデン] - 2, 1, 3-ベンゾォキサゾ一ル- 4-スルホ ンアミド N_ [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene] -2 1,3-benzoxazol-4-sulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりにベンゾフラザン- 4-スルホニル クロリド (103mg, 0.471匪 ol) を用いて、 実施例 6-2で得られた 1-ァミノ- 5,6-ジメチル- 8 - (イソペンチルスル ファニル)- 3H -イミダゾ [5, 1 - c] [1, 4]チアジン- 3-オン (70.0 mg, 0.235腿 ol) か ら題記化合物 (46.8mg, 42%) を合成した。 酢酸ェチル -ジェチルエーテルから再 結晶して融点 197- 198°Cの暗赤色結晶を得た。 Example 7-5 Method was performed in the same manner as in Method C, except that benzofurazan-4-sulfonyl chloride (103 mg, 0.471 marl) was used instead of 4-bromomethylbenzenesulfonyl chloride to obtain Example 6-2. 1-Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (70.0 mg, 0.235 t ol) The title compound (46.8 mg, 42%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 197-198 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3202, 2148, 2961, 2932, 1725, 1657, 1615, 1584 cur1. Infrared absorption spectrum (IR) (in KBr) V: 3202, 2148, 2961, 2932, 1725, 1657, 1615, 1584 cur 1 .
^-NMR (CDC13) : δ 0.876 (6H, d, J=6.6 Hz), 1.42-1.68 (3H, m), 1.80 (3H, d, J=0.8Hz), 2.33 (3H, d, J=1.0Hz), 2.89 (2H, t, J=7.8Hz), 7.54 (1H, dd, J=9.0, 6.6 Hz), 8.06 (1H, dd, J=9.2, 0.8 Hz), 8.13 (1H, dd, J=7.0, 0.6 Hz), 9.87 (1H, bs). ^ -NMR (CDC1 3): δ 0.876 (6H, d, J = 6.6 Hz), 1.42-1.68 (3H, m), 1.80 (3H, d, J = 0.8Hz), 2.33 (3H, d, J = 1.0Hz), 2.89 (2H, t, J = 7.8Hz), 7.54 (1H, dd, J = 9.0, 6.6 Hz), 8.06 (1H, dd, J = 9.2, 0.8 Hz), 8.13 (1H, dd, J = 7.0, 0.6 Hz), 9.87 (1H, bs).
元素分析値: C19H21N504S3として Elementary analysis: as C 19 H 21 N 5 0 4 S 3
計算値 (%) : C, 47.58; H, 4.41; , 14.60; S, 20.06. Calculated (%): C, 47.58; H, 4.41;, 14.60; S, 20.06.
実測値 (%) : C, 47.49; H, 4.34; N, 14.41; S, 19.91. Found (%): C, 47.49; H, 4.34; N, 14.41; S, 19.91.
実施例 7-29 Example 7-29
N- [5, 6 -ジメチル- 8- (ィソペンチルスルファニル) - 3-ォキソ -2 , 3-ジヒドロ - 1H -ィ ミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデント 2, 4, 6-トリメチルベンゼンスルホン アミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-lirident 2 , 4, 6-trimethylbenzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 2,4, 6-卜リメチルベンゼンスルホニル クロリド (150 mg, 0.672匪 ol) を用いて、 実施例 6- 2で得られた卜ァミノ- 5,6-ジメチル -8- (イソ ペンチルスルファ二ル)- 3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (100 mg, 0.336 腿 ol) から題記化合物 (102 mg, 63¾) を合成した。 酢酸ェチル-ジェチル ェ一テルから再結晶して融点 161-163 の暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, except that 4-bromomethylbenzenesulfonyl chloride was replaced with 2,4,6-trimethylbenzenesulfonyl chloride (150 mg, 0.672 bandol), the procedure of Example 7-5 was repeated. 6-Diamino-5,6-dimethyl-8- (iso The title compound (102 mg, 63 mg) was synthesized from (pentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.336 t ol). Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 161-163.
赤外吸収スぺクトル(IR) ( Br中) レ : 3378, 3245, 2957, 2936, 2870, 1744, 1726, 1661, 1615, 1557 cm"1. 'Infrared absorption spectrum (IR) (in Br) Re: 3378, 3245, 2957, 2936, 2870, 1744, 1726, 1661, 1615, 1557 cm " 1 .
-醒 R (CDC13) : δ 0.913 (6Η, d, J=6.6 Hz), 1.45-1.59 (2H, m), 1.59-1.72 (IH, m), 1.78 (3H, s), 2.26 (3H, s), 2.28 (3H, s), 2.71 (6H, s), 2.93 (2H, t, J=6.6 Hz), 6.92 (2H, s), 9.66 (1H, bs) . - Awakening: R (CDC1 3): δ 0.913 (6Η, d, J = 6.6 Hz), 1.45-1.59 (2H, m), 1.59-1.72 (IH, m), 1.78 (3H, s), 2.26 (3H, s), 2.28 (3H, s), 2.71 (6H, s), 2.93 (2H, t, J = 6.6 Hz), 6.92 (2H, s), 9.66 (1H, bs).
実施例 7-30 Example 7-30
4, 5 -ジク口 P-N-C5, 6-ジメチル- 8- (ィソペンチルスルファ二ル)- 3-ォキソ -2, 3- ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン-卜ィリデン ]-2-チオフェンスルホ ンアミド 4,5-Dimethyl PN-C5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1, 4,] Thiazine-triylidene] -2-thiophenesulfonamide
実施例 7- 5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4, 5-ジクロロチォフェン -2-スルホニル クロリド (174 mg, 0.672 匪 ol) を用いて、 実施例 6-2で得られた卜ァミノ- 5, 6-ジメチル- 8- (イソ ペンチルスルファ二ル)- 3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (100 mg, 0.336讓 ol) から題記化合物 (104 mg, 60%) を合成した。 酢酸ェチル -ジェチル ェ一テルから再結晶して融点 203- 205 :の暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, except that 4,5-dichlorothiophene-2-sulfonyl chloride (174 mg, 0.672 bandol) was used instead of 4-bromomethylbenzenesulfonyl chloride. Triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one obtained in 6-2 (100 mg, The title compound (104 mg, 60%) was synthesized from 0.336 benzyl). Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 203-205.
赤外吸収スペクトル(IR) (KBr中) レ : 3451, 3214, 3079, 2955, 1757, 1742, 1661, 1615, 1553 cm""1. Infrared absorption spectrum (IR) (in KBr) Re: 3451, 3214, 3079, 2955, 1757, 1742, 1661, 1615, 1553 cm "" 1 .
Ή-NMR (CDC13) : 6 0.951 (6H, d, J=6.6Hz), 1.56-1.68 (2H, m), 1.68-1.77 (IH, m), 1.82 (3H, d, J=1.2 Hz), 2.30 (3H, d, J=1.2 Hz), 3.00 (2H, t, J=7.8 Hz), 7.44 (1H, s), 9.50 (IH, bs). Ή-NMR (CDC1 3): 6 0.951 (6H, d, J = 6.6Hz), 1.56-1.68 (2H, m), 1.68-1.77 (IH, m), 1.82 (3H, d, J = 1.2 Hz) , 2.30 (3H, d, J = 1.2 Hz), 3.00 (2H, t, J = 7.8 Hz), 7.44 (1H, s), 9.50 (IH, bs).
元素分析値: CnH19N303S4C として Elementary analysis: as C n H 19 N 3 0 3 S 4 C
計算値 (¾) : C, 39.84; H, 3.74; N, 8.20; S, 25.03. Calculated value (¾): C, 39.84; H, 3.74; N, 8.20; S, 25.03.
実測値 ( ) : C, 39.66; H, 3.70; N, 8.08; S, 24.93. Found (): C, 39.66; H, 3.70; N, 8.08; S, 24.93.
実施例 7 - 31 Example 7-31
3, 4-ジク口口- N- [5, 6-ジメチル- 8- (ィソペンチルスルファニル) -3-ォキソ -2, 3- ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 1ーィリデン]ベンゼンスルホンアミ ド 3,4-Dichloro-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 [Thiazine-1-ylidene] benzenesulfone Do
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 3, 4 -ジクロ口ベンゼンスルホニル クロリド (168 mg, 0.672 miol) を用いて、 実施例 6-2で得られた卜ァミノ- 5, 6 -ジメチル- 8_ (イソペンチ ルスルファニル) - 3H -イミダゾ [5,卜 c] [1,4]チアジン - 3-オン (100 mg, 0.336 画 ol) から題記化合物 (112 mg, 66%) を合成した。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 162_164°Cの暗赤色結晶を得た。 Example 7-5 Method 6-2 was repeated in the same manner as in Method C, except that benzenesulfonyl chloride (168 mg, 0.672 miol) was used instead of 4-bromomethylbenzenesulfonyl chloride. -5,6-Dimethyl-8_ (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.336 fraction ol) (112 mg, 66%) was synthesized. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 162 to 164 ° C.
赤外吸収スぺクトル(IR) (KBr中) リ: 3227, 3092, 2957, 2930, 2872, 1759, 1661, 1620, 1557 cur1. Infrared absorption spectrum (IR) (in KBr): 3227, 3092, 2957, 2930, 2872, 1759, 1661, 1620, 1557 cur 1 .
Ή-NMR (CDC13) : δ 0.938 (6Η, d, J=6.6Hz), 1.51-1.63 (2H, m), 1.63-1.78 (1H, m), 1.80 (3H, d, J-0.8 Hz), 2.29 (3H, d, J=1.2 Hz), 2.97 (2H, t, J=7.8 Hz), Ή-NMR (CDC1 3): δ 0.938 (6Η, d, J = 6.6Hz), 1.51-1.63 (2H, m), 1.63-1.78 (1H, m), 1.80 (3H, d, J-0.8 Hz) , 2.29 (3H, d, J = 1.2 Hz), 2.97 (2H, t, J = 7.8 Hz),
7.56 (1H, d, J=8. Hz), 7.79 (1H, dd, J=8.0, 1.8 Hz), 8.08 (1H, d, J=2.2 Hz),7.56 (1H, d, J = 8.Hz), 7.79 (1H, dd, J = 8.0, 1.8 Hz), 8.08 (1H, d, J = 2.2 Hz),
9.57 (1H, bs). 9.57 (1H, bs).
元素分析値: C19H21N303S3C12として Elemental analysis: as C 19 H 21 N 3 0 3 S 3 C 12
計算値 (%) : C, 45.06; H, 4.18; N, 8.30; S, 18.99. Calculated value (%): C, 45.06; H, 4.18; N, 8.30; S, 18.99.
実測値 (¾) : C, 45.27; H, 4.18; N, 8.04; S, 18.65. Found (¾): C, 45.27; H, 4.18; N, 8.04; S, 18.65.
実施例 7-32 Example 7-32
2, 5-ジク口ロ- N- [5, 6 -ジメチル -8- (ィソペンチルスルファ二ル)- 3-ォキソ -2, 3- ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン]ベンゼンスルホンアミ ド 2,5-Dicyclo-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene] benzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 2,5-ジクロロベンゼンスルホニル クロリド (168 mg, 0.672 mmol) を用いて、 実施例 6 - 2で得られた卜ァミノ- 5, 6-ジメチル -8- (イソペンチ ルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.336 mmol) から題記化合物 (146 mg, 85¾) を合成した。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 180- 182°Cの喑赤色結晶を得た。 Example 7-5 In the same manner as in Method C, except that 4-bromomethylbenzenesulfonyl chloride was replaced with 2,5-dichlorobenzenesulfonyl chloride (168 mg, 0.672 mmol), the procedure of Example 6-2 was repeated. Title obtained from the obtained triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 mg, 0.336 mmol) Compound (146 mg, 85¾) was synthesized. The crystals were recrystallized from ethyl acetate-getyl ether to obtain reddish crystals having a melting point of 180-182 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3245, 3090, 2961, 2928, 1725, 1663, 1605, 1543 cm-1. Infrared absorption spectrum (IR) (in KBr) V: 3245, 3090, 2961, 2928, 1725, 1663, 1605, 1543 cm- 1 .
Ή-NMR (CDCI3) : δ 0.906 (6Η, d, J=6.2 Hz), 1.47-1.62 (2H, m), 1.62-1.74 (1H, m), 1.81 (3H, d, J=1.0 Hz), 2.31 (3H, d, J=0.8 Hz), 2.94 (2H, t, J=7.6 Hz), 7.43 (IH, s), 7.44 (IH, d, J=0.8 Hz), 8.20 (IH, t, J=1.0 Hz), 9.71 (1H, bs). 元素分析値: C19H21N303S3C12として Ή-NMR (CDCI3): δ 0.906 (6Η, d, J = 6.2 Hz), 1.47-1.62 (2H, m), 1.62-1.74 (1H, m), 1.81 (3H, d, J = 1.0 Hz), 2.31 (3H, d, J = 0.8 Hz), 2.94 (2H, t, J = 7.6 Hz), 7.43 (IH, s), 7.44 (IH, d, J = 0.8 Hz), 8.20 (IH, t, J = 1.0 Hz), 9.71 (1H, bs) Elementary analysis:. as C 19 H 21 N 3 0 3 S 3 C1 2
計算値 : C, 45.06; H, 4.18; N, 8.30; S, 18.99. Calculated: C, 45.06; H, 4.18; N, 8.30; S, 18.99.
実測値 (¾) : C, 44.96; H, 4.45; N, 8.40; S, 18.86. Found (¾): C, 44.96; H, 4.45; N, 8.40; S, 18.86.
実施例 7-33 Example 7-33
2, 5-ジフルォロ - N- [5, 6-ジメチル- 8- (イソペンチルスルファニル) -3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン]ベンゼンスルホ ンアミド 2,5-difluoro-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene] benzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-プロモメチルベンゼンスルホニル ク ロリドの代わりに 2,4-ジフルォロベンゼンスルホニル クロリド (143 mg, 0.672 匪 ol) を用いて、 実施例 6- 2で得られた 1-ァミノ- 5, 6-ジメチル- 8 -(イソペンチ ルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3-オン (100 mg, 0.336 mmol) から題記化合物 (85.1mg, 54%) を合成した。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 187- 189°Cの暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, except that 2,4-difluorobenzenesulfonyl chloride (143 mg, 0.672 ol) was used instead of 4-bromomethylbenzenesulfonyl chloride. 1-Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Step 2 (100 mg, 0.336 mmol )) To give the title compound (85.1 mg, 54%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 187-189 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3384, 3248, 3102, 2955, 2930, 2870, 1728, 1661, 1603, 1557, 1481 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3384, 3248, 3102, 2955, 2930, 2870, 1728, 1661, 1603, 1557, 1481 cm " 1 .
Ή-NMR (CDC13) : δ 0.954 (6Η, d, J=6.6 Hz), 1.46-1.62 (2H, m), 1.62-1.75 (1H, m), 1.81 (3H, d, J=0.8 Hz), 2.30 (3H, d, J=1.0 Hz), 2.93 (2H, t, J=7.6 Hz), 6.86-7.05 (2H, m), 7.94-8.08 (IH, m), 9,69 (IH, bs). Ή-NMR (CDC1 3): δ 0.954 (6Η, d, J = 6.6 Hz), 1.46-1.62 (2H, m), 1.62-1.75 (1H, m), 1.81 (3H, d, J = 0.8 Hz) , 2.30 (3H, d, J = 1.0 Hz), 2.93 (2H, t, J = 7.6 Hz), 6.86-7.05 (2H, m), 7.94-8.08 (IH, m), 9,69 (IH, bs ).
元素分析値: C19H21N303S3F2として Elemental analysis: as C 19 H 21 N 3 0 3 S 3 F 2
計算値 (%) : C, 48.19; H, 4.47; N, 8.87; S, 20.31. Calculated value (%): C, 48.19; H, 4.47; N, 8.87; S, 20.31.
実測値 (%) : C, 47.96; H, 4.39; N, 8.92; S, 20.21. Found (%): C, 47.96; H, 4.39; N, 8.92; S, 20.21.
実施例 7-34 Example 7-34
2-ク口口- 4-フルォロ- N- [5, 6-ジメチル- 8- (ィソペンチルスルファニル) -3-ォキ ソ- 2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン]ベンゼンスル ホンアミド 2-c-mouth- 4-fluoro-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro- mouth-1H-imidazo [5,1-c ] [1,4] thiazine-1-ylidene] benzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 2-クロ口- 4-フルォロベンゼンスルホニル クロリド (154 mg, 0.672腿 ol) を用いて、 実施例 6-2で得られた卜アミノ- 5, 6 -ジメチル- 8- (イソ ペンチルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.336腿 ol) から題記化合物 (107 mg, 65¾) を合成した。'酢酸ェチル -ジェチル エーテルから再結晶して融点 204- 206°Cの暗赤色結晶を得た。 Example 7-5 According to a method similar to that of Method C, 4-bromomethylbenzenesulfonyl chloride was used instead of 4-bromomethylbenzenesulfonyl chloride (154 mg, Using the 0.672 thigh ol), the triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,1) obtained in Example 6-2 was obtained. 4] The title compound (107 mg, 65%) was synthesized from thiazin-3-one (100 mg, 0.336 tmol). 'Recrystallization from ethyl acetate-getyl ether yielded dark red crystals with a melting point of 204-206 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3210, 2957, 2934, 2872, 1734, 1715, 1661, 1609, 1588, 1555 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3210, 2957, 2934, 2872, 1734, 1715, 1661, 1609, 1588, 1555 cm- 1 .
Ή-NMR (CDC13) : δ 0.904 (6Η, d, J=6.2Hz), 1.45-1.61 (2H, m), 1.61-1.74 (1H, m), 1.81 (3H, s), 2.30 (3H, s), 2.93 (2H, t, J=7.2 Hz), 7.04-7.16 (1H, m), Ή-NMR (CDC1 3): δ 0.904 (6Η, d, J = 6.2Hz), 1.45-1.61 (2H, m), 1.61-1.74 (1H, m), 1.81 (3H, s), 2.30 (3H, s), 2.93 (2H, t, J = 7.2 Hz), 7.04-7.16 (1H, m),
7.22-7.32 (1H, m), 8.20 (1H, dd, J=8.8, 5.8 Hz), 9.71 (1H, bs). 7.22-7.32 (1H, m), 8.20 (1H, dd, J = 8.8, 5.8 Hz), 9.71 (1H, bs).
元素分析値: C19H21N303S3C1Fとして Elemental analysis: as C 19 H 21 N 3 0 3 S 3 C1F
計算値 (%) : C, 46.57; H, 4.32; N, 8.57; S, 19.63. Calculated (%): C, 46.57; H, 4.32; N, 8.57; S, 19.63.
実測値 (¾) : C, 46.50; H, 4.26; N, 8.59; S, 19.43. Found (¾): C, 46.50; H, 4.26; N, 8.59; S, 19.43.
実施例 7-35 Example 7-35
3, 4-ジフルォロ -N- [5, 6-ジメチル -8- (イソペンチルスルファニル) -3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン]ベンゼンスルホ ンアミド 3,4-Difluoro-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene] benzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 3, 4-ジフルォロベンゼンスルホニル クロリド (143 mg, 0.672 mmol) を用いて、 実施例 6-2で得られた 1 -アミノ- 5, 6-ジメチル- 8- (イソペンチ ルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.336 腿 ol) から題記化合物 (80.5mg, 51%) を合成した。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 153- 155°Cの暗赤色結晶を得た。 Example 7-5 Method 6-2 was repeated in the same manner as in Method C, except that 3,4-difluorobenzenesulfonyl chloride (143 mg, 0.672 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. 1-amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 mg, 0.336 t )) To give the title compound (80.5 mg, 51%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 153-155 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3206, 3069, 2963, 2909, 2872, 1757, 1744, 1744, 1663 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3206, 3069, 2963, 2909, 2872, 1757, 1744, 1744, 1663 cm- 1 .
Ή- NMR (CDCI3) : δ 0.939 (6Η, d, J=6.2 Hz), 1.50-1.63 (2H, m), 1.63-1.78 (1H, m), 1.81 (3H, d, J=1.2 Hz), 2.28 (3H, d, J-0.8 Hz), 2.97 (2H, t, J=7.6 Hz), 7.18-7.35 (1H, m), 7.70-7.79 (1H, m), 7.80-7.88 (1H, m), 9.57 (1H, bs). 元素分析値: Cl9H21N303S3F2として Ή-NMR (CDCI3): δ 0.939 (6Η, d, J = 6.2 Hz), 1.50-1.63 (2H, m), 1.63-1.78 (1H, m), 1.81 (3H, d, J = 1.2 Hz), 2.28 (3H, d, J-0.8 Hz), 2.97 (2H, t, J = 7.6 Hz), 7.18-7.35 (1H, m), 7.70-7.79 (1H, m), 7.80-7.88 (1H, m) , 9.57 (1H, bs). Elemental analysis: Cl 9 H 21 N 3 0 3 S 3 F 2
計算値 (%) : C, 48.19; H, 4.47; N, 8.87; S, 20.31. 実測値 (%) : C, 48.26; H, 4.28; N, 8.88; S, 20.39. Calculated (%): C, 48.19; H, 4.47; N, 8.87; S, 20.31. Found (%): C, 48.26; H, 4.28; N, 8.88; S, 20.39.
実施例 7 - 36 Example 7-36
N - [5, 6-ジメチル- 8- (ィソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5,1 - c] [1,4]チアジン-卜イリデン]オクタンスルホンアミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine Ilidene] octanesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりにオクタンスルホニル クロリド(129mg, 0.605腿 ol)を用いて、 実施例 6-2 で得られた 1-ァミノ- 5, 6-ジメチル -8- (ィソペンチルスルファ二 ル)- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.336 mmol) から題記 化合物 (24.9mg, 17%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 153_155°Cの暗赤色結晶を得た。 Example 7-5 Method 1-Amino obtained in Example 6-2 was obtained in the same manner as in Method C, except that octanesulfonyl chloride (129 mg, 0.605 mol) was used instead of 4-bromomethylbenzenesulfonyl chloride. -5,6-Dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 mg, 0.336 mmol) from the title compound ( 24.9 mg, 17%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 153 to 155 ° C.
赤外吸収スペクトル(IR) (KBr中) V 3210, 2955, 2924, 2857, 1755, 1738, 1663, 1622, 1570 cm"1. Infrared absorption spectrum (IR) (in KBr) V 3210, 2955, 2924, 2857, 1755, 1738, 1663, 1622, 1570 cm " 1 .
'H-NMR (CDC13) : δ 0.881 (3Η, t, J=6.6Hz), 0.945 (6H, d, J=6.2Hz), 1.22-1.64 (12H, m), 1.64-1.78 (1H, m), 1.81 (3H, s), 1.83-1.97 (2H, m), 2.28 (3H, s), 2.98' (2H, t, J=8.0 Hz), 3.11 (2H, t, J=8.0 Hz), 9.45 (1H, bs). 'H-NMR (CDC1 3) : δ 0.881 (3Η, t, J = 6.6Hz), 0.945 (6H, d, J = 6.2Hz), 1.22-1.64 (12H, m), 1.64-1.78 (1H, m ), 1.81 (3H, s), 1.83-1.97 (2H, m), 2.28 (3H, s), 2.98 '(2H, t, J = 8.0 Hz), 3.11 (2H, t, J = 8.0 Hz), 9.45 (1H, bs).
元素分析値: C21H35N303S3として Elemental analysis: as C 21 H 35 N 3 0 3 S 3
計算値 (%) : C, 53.24; H, 7.45; N, 8.87. Calculated value (%): C, 53.24; H, 7.45; N, 8.87.
実測値 (%) : C, 52.95; H, 7.40; N, 8.56. Found (%): C, 52.95; H, 7.40; N, 8.56.
実施例 7-37 Example 7-37
4 -ァセチル -N-[5, 6 -ジメチル -8- (ィソペンチルスルファ二ル)- 3-ォキソ -2, 3 -ジ ヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン]ベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4-ァセチルベンゼンスルホニル クロリド(136mg, 0.605 mmol) を用いて、 実施例 6-2で得られた卜ァミノ _5, 6 -ジメチル -8- (イソペンチルスル ファニル) -3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (90.0 mg, 0.303誦 ol) か ら題記化合物 (85.9mg, 59%) を合成した。 酢酸ェチル-ジェチルェ一テルから再 結晶して融点 176-178°Cの暗赤色結晶を得た。 4-Acetyl-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene] benzenesulfonamide Example 7-5 Method similar to Method C, using 4-acetylbenzenesulfonyl chloride (136 mg, 0.605 mmol) instead of 4-bromomethylbenzenesulfonyl chloride. The triamino_5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-2 (90.0 mg The title compound (85.9 mg, 59%) was synthesized from the following compounds. The crystals were recrystallized from ethyl acetate-jetil ether to obtain dark red crystals having a melting point of 176-178 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3231, 2957, 2934, 2870, 1759, 1694, 1663, 1620, 1557 cm—1. Ή-NMR (CDCI3) : δ 0.935 (6Η, d, J=6.4 Hz), 1.44-1.77 (3H, m), 1.80 (3H, s), 2.28 (3H, s), 2.65 (3H, s), 2.96 (2H, i, J=7.4 Hz), 8.06 (4H, s), 9.62 (1H, bs). Infrared absorption spectrum (IR) (in KBr): 3231, 2957, 2934, 2870, 1759, 1694, 1663, 1620, 1557 cm- 1 . Ή-NMR (CDCI3): δ 0.935 (6Η, d, J = 6.4 Hz), 1.44-1.77 (3H, m), 1.80 (3H, s), 2.28 (3H, s), 2.65 (3H, s), 2.96 (2H, i, J = 7.4 Hz), 8.06 (4H, s), 9.62 (1H, bs).
元素分析値: C21¾5N304S3として · Elemental analysis: - as C 21 ¾ 5 N 3 0 4 S 3
計算値 (¾) : C, 52.59; H, 5.25; N, 8.76; S, 20.06. Calculated value (¾): C, 52.59; H, 5.25; N, 8.76; S, 20.06.
実測値 (%) : C, 52.23; H, 5.11; N, 8.67; S, 20.01. Found (%): C, 52.23; H, 5.11; N, 8.67; S, 20.01.
実施例 7-38 Example 7-38
N-[5, 6-ジメチル -8- (イソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H-ィ ミダゾ [5,卜 c] [1, 4]チアジン- 1-イリデン] -2-メトキシ- 4-メチルベンゼンスルホ ンアミド N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene ] -2-Methoxy-4-methylbenzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 2-メトキシ -4-メチルベンゼンスルホニル クロリド (134 mg, 0.605廳 ol) を用いて、 実施例 6-2で得られた卜アミノ- 5, 6 -ジメチル -8- (イソ ペンチルスルファ二ル)- 3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3-オン (90.0 nig, 0.303 mmol) から題記化合物 (123 mg, 84%) を合成した。 酢酸ェチル-ジェチル エーテルから再結晶して融点 192-194°Cの暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, using 2-methoxy-4-methylbenzenesulfonyl chloride (134 mg, 0.605 hall) instead of 4-bromomethylbenzenesulfonyl chloride, Triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in 2 (90.0 nig, 0.303 mmol) to give the title compound (123 mg, 84%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 192-194 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3262, 2953, 2926, 2867, 1717, 1663, 1615, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr): 3262, 2953, 2926, 2867, 1717, 1663, 1615, 1557 cm " 1 .
¾-NMR (CDCI3) : δ 0.886 (6Η, d, J-6.2 Hz), 1.45-1.72 (3H, m), 1.80 (3H, s), 2.30 (3H, s), 2.40 (3H, s), 2.89 (2H, t, J=7.6 Hz), 3.87 (3H, s), 6.77 (1H, s), 6.85 (1H , dd, J=7.8, 1.0 Hz), 7.90 (1H, d, J=7.8 Hz), 9.85 (1H, bs). 元素分析値: C21H27N3O4S3'0.3EtOAcとして ¾-NMR (CDCI3): δ 0.886 (6Η, d, J-6.2 Hz), 1.45-1.72 (3H, m), 1.80 (3H, s), 2.30 (3H, s), 2.40 (3H, s), 2.89 (2H, t, J = 7.6 Hz), 3.87 (3H, s), 6.77 (1H, s), 6.85 (1H, dd, J = 7.8, 1.0 Hz), 7.90 (1H, d, J = 7.8 Hz) ), 9.85 (1H, bs). Elemental analysis: C 21 H 27 N 3 O 4 S 3 '0.3EtOAc
計算値 (¾) : C, 52.48; H, 5.83; N, 8.27; S, 18.93. Calculated (¾): C, 52.48; H, 5.83; N, 8.27; S, 18.93.
実測値 (¾) : C, 52.31; H, 5.63; N, 8.22; S, 18.74. Found (¾): C, 52.31; H, 5.63; N, 8.22; S, 18.74.
実施例 7-39 ■Example 7-39 ■
3 -ク口ロ- N- [5, 6-ジメチル- 8- (ィソペンチルスルファ二ル)- 3-ォキソ - 2, 3 -ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン ]-4-メチルベンゼンスルホ ンアミド 3-Cupro-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene] -4-methylbenzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 3-クロ口- 4-メチルベンゼンスルホニル クロリド (136 mg, 0.605 mmol) を用いて、 実施例 6-2で得られた卜ァミノ- 5, 6-ジメチル- 8- (イソ ペンチルスルファニル) -3H-イミダゾ [5, 1- c] [1,4]チアジン- 3-オン (90.0 mg, 0.303 mmol) から題記化合物 (123 mg, 83%) を合成した。 酢酸ェチル-ジェチル ェ一テルから再結晶して融点 166_168°Cの暗赤色結晶を得た。 Example 7-5 Method 4-bromomethylbenzenesulfonyl chloride was obtained in the same manner as in Method C. Using trichloro-4-methylbenzenesulfonyl chloride (136 mg, 0.605 mmol) instead of chloride, triamino-5,6-dimethyl-8- (isopentylsulfanyl) obtained in Example 6-2 was used. ) The title compound (123 mg, 83%) was synthesized from -3H-imidazo [5,1-c] [1,4] thiazin-3-one (90.0 mg, 0.303 mmol). Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 166-168 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3237, 2955, 2926, 2868, 1757, 1663, 1620, 1561 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3237, 2955, 2926, 2868, 1757, 1663, 1620, 1561 cm " 1 .
丽 R (CDC13) : δ 0.934 (6Η, d, J=6.2Hz), 1.51-1.64 (2H, m), 1.64-1.77 (1H, m), 1.80 (3H, d, J-0.8 Hz), 2.28 (3H, s), 2.43 (3H, s), 2.96 (2H, t, J=7.6 Hz), 7.34 (1H, d, J=8.2 Hz), 7.74 (1H , dd, J=8.0, 1.8 Hz), 7.96 (1H, d, J=1.8丽R (CDC1 3): δ 0.934 (6Η, d, J = 6.2Hz), 1.51-1.64 (2H, m), 1.64-1.77 (1H, m), 1.80 (3H, d, J-0.8 Hz), 2.28 (3H, s), 2.43 (3H, s), 2.96 (2H, t, J = 7.6 Hz), 7.34 (1H, d, J = 8.2 Hz), 7.74 (1H, dd, J = 8.0, 1.8 Hz) ), 7.96 (1H, d, J = 1.8
Hz), 9.57 (1H, bs). Hz), 9.57 (1H, bs).
元素分析値: C2。 4N303S3C1として Elemental analysis: C 2. 4 N 3 0 3 S 3 C1
計算値 (%) : C, 49.42; H, 4.98; N, 8.64; S, 19.79. Calculated (%): C, 49.42; H, 4.98; N, 8.64; S, 19.79.
実測値 (%) : C, 49.22; H, 4.82; N, 8.71; S, 19.71. Found (%): C, 49.22; H, 4.82; N, 8.71; S, 19.71.
実施例 7- 40 Example 7-40
N - [5, 6-ジメチル- 8- (イソペンチルスルファ二ル)- 3-ォキソ - 2,3-ジヒドロ- 1H -ィ ミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン] -4-メチル -3-二トロベンゼンスルホン アミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene ] -4-methyl-3-nitrobenzenesulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4-メチル -3-ニトロベンゼンスルホニル クロリド (317 mg, 1.3 mmol) を用いて、 実施例 6- 2で得られた 1-ァミノ- 5, 6-ジメチル- 8- (イソべ ンチルスルファニル) -3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (200 mg, 0.672 mmol) から題記化合物 (296 mg, 89¾) を合成した。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 163- 165°Cの暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, using 4-methyl-3-nitrobenzenesulfonyl chloride (317 mg, 1.3 mmol) instead of 4-bromomethylbenzenesulfonyl chloride, the procedure of Example 6-2 was repeated. From the obtained 1-amino-5,6-dimethyl-8- (isobenzylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.672 mmol) The title compound (296 mg, 89%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 163-165 ° C.
赤外吸収スぺクトル(IR) (KBr中) v: 3220, 2959, 2934, 2870, 1759, 1661, 1618, 1557, 1532 cm— Infrared absorption spectrum (IR) (in KBr) v: 3220, 2959, 2934, 2870, 1759, 1661, 1618, 1557, 1532 cm—
Ή-NMR (CDC13) : δ 0.940 (6Η, d, J=6.6 Hz), 1.52-1.65 (2H, m), 1.65-1.78 (1H, m), 1.81 (3H, d, J=1.2 Hz), 2.29 (3H, d, J=1.0 Hz), 2.67 (3H, s), 2.98 (2H, t, J=7.8 Hz), 7.49 (1H, d, J=8.0 Hz), 8.07 (1H , dd, J=8.0, 2.2 Hz), 8.61 (1H, d, J=1.8 Hz), 9.57 (1H, bs). Ή-NMR (CDC1 3): δ 0.940 (6Η, d, J = 6.6 Hz), 1.52-1.65 (2H, m), 1.65-1.78 (1H, m), 1.81 (3H, d, J = 1.2 Hz) , 2.29 (3H, d, J = 1.0 Hz), 2.67 (3H, s), 2.98 (2H, t, J = 7.8 Hz), 7.49 (1H, d, J = 8.0 Hz), 8.07 (1H, dd, J = 8.0, 2.2 Hz), 8.61 (1H, d, J = 1.8 Hz), 9.57 (1H, bs).
元素分析値: C2。H24N405S3として Elemental analysis: C 2. H 24 N 4 0 5 S 3
計算値 (%) : C, 48.37; H, 4.87; N, 11.28; S, 19.37. Calculated value (%): C, 48.37; H, 4.87; N, 11.28; S, 19.37.
実測値 (%) : C, 48.48; H, 4.80; N, 11.15; S, 19.46. Found (%): C, 48.48; H, 4.80; N, 11.15; S, 19.46.
実施例 7-41 Example 7-41
N - [5, 6-ジメチル- 8 -(イソペンチルスルファ二ル)- 3-ォキソ - 2,3-ジヒドロ- 1H-ィ ミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン ]-1_メチル -4-イミダゾールスルホンァ ミ F N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene ] -1_Methyl-4-imidazolesulfonami F
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 1-メチル -4-イミダゾ一ルスルホニル クロリド(115mg, 0.605 mniol) を用いて、 実施例 6-2で得られた 1 -アミノ- 5, 6-ジメチル -8- (イソペンチ ルスルファニル) - 3H -イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (90.0 mg, 0.303 腿 ol) から題記化合物 (62.5mg, 47¾) を合成した。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 213-215°Cの暗赤色結晶を得た。 Example 7-5 Method A method similar to that of Example C was repeated, except that 1-methyl-4-imidazolsulfonyl chloride (115 mg, 0.605 mniol) was used instead of 4-bromomethylbenzenesulfonyl chloride. From 1-amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (90.0 mg, 0.303 t) obtained in The title compound (62.5 mg, 47¾) was synthesized. Recrystallization from ethyl acetate-getyl ether yielded dark red crystals with a melting point of 213-215 ° C.
赤外吸収スペクトル(IR) (KBr中) ソ : 3248, 3117, 2957, 2930, 2870, 1769, 1732, 1665, 1617, 1559, 1532 cm"1. Infrared absorption spectrum (IR) (in KBr) So: 3248, 3117, 2957, 2930, 2870, 1769, 1732, 1665, 1617, 1559, 1532 cm " 1 .
Ή-NMR (CDC") : δ 0.919 (6Η, d, J-6.2 Hz), 1.42-1.76 (3H, m), 1.80 (3H, d, J=0.8 Hz), 2.28 (3H, d, J=0.6 Hz), 2.94 (2H, t, J=7.8Hz), 3.75 (3H, s), 7.48 (1H, s), 7.51 (1H, s). Ή-NMR (CDC "): δ 0.919 (6Η, d, J-6.2 Hz), 1.42-1.76 (3H, m), 1.80 (3H, d, J = 0.8 Hz), 2.28 (3H, d, J = 0.6 Hz), 2.94 (2H, t, J = 7.8Hz), 3.75 (3H, s), 7.48 (1H, s), 7.51 (1H, s).
元素分析値: C17H23N503S3として Elementary analysis: as C 17 H 23 N 5 0 3 S 3
計算値 (¾) : C, 46.24; H, 5.25; N, 15.86; S, 21.78. Calculated value (¾): C, 46.24; H, 5.25; N, 15.86; S, 21.78.
実測値 (¾) : C, 46.13; H, 5.23; N, 15.98; S, 21.86. Found (¾): C, 46.13; H, 5.23; N, 15.98; S, 21.86.
実施例 7 - 42 Example 7-42
3 -ブロモ -N- [5, 6-ジメチル -8- (ィソペンチルスルファニル) -3-ォキソ - 2, 3-ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜イリデン]ベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 3-ブロモベンゼンスルホニル クロリド (137 mg, 0.538 mmol) を用いて、 実施例 6-2で得られた卜ァミノ- 5, 6-ジメチル -8- (イソペンチルスル ファニル )_3H -イミダゾ [5, 卜 c] [1,4]チアジン- 3 -オン (80.0 mg, 0.269腿01) か ら題記化合物 (114 mg, 75¾) を合成した。 酢酸ェチル -ジェチルエーテルから再 結晶して融点 152_154°Cの暗赤色結晶を得た。 3-bromo-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-tri [Ilidene] benzenesulfonamide Example 7-5 Method 6 was repeated in the same manner as in Method C, except that 3-bromobenzenesulfonyl chloride (137 mg, 0.538 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. -5,6-Dimethyl-8- (isopentylsulfanyl) _3H-imidazo [5, tric] [1,4] thiazin-3-one obtained in -2 (80.0 mg, 0.269 thigh 01) Or The title compound (114 mg, 75%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 152-154 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3231, 3086, 2955, 2932, 2868, 1757, 1663, 1618, 1557 cnf1. Infrared absorption spectrum (IR) (in KBr) V: 3231, 3086, 2955, 2932, 2868, 1757, 1663, 1618, 1557 cnf 1 .
¾-NMR (CDC13) : δ 0.936 (6Η, d, J=6.6 Hz), 1.52-1.78 (3H, m), 1.80 (3H, d, J=1.0 Hz), 2.28 (3H, d, J=1.2Hz), 2.97 (2H, t, J=7.6 Hz), 7.36 (1H, t, J=7.8 Hz), 7.64-7.72 (1H, m), 7.86-7.94 (1H, m), 8.14 (1H, t, J=1.8Hz), 9.58 (1H, bs). ¾-NMR (CDC1 3): δ 0.936 (6Η, d, J = 6.6 Hz), 1.52-1.78 (3H, m), 1.80 (3H, d, J = 1.0 Hz), 2.28 (3H, d, J = 1.2Hz), 2.97 (2H, t, J = 7.6 Hz), 7.36 (1H, t, J = 7.8 Hz), 7.64-7.72 (1H, m), 7.86-7.94 (1H, m), 8.14 (1H, t, J = 1.8Hz), 9.58 (1H, bs).
元素分析値: C19H22N303S3Brとして Elementary analysis: as C 19 H 22 N 3 0 3 S 3 Br
計算値 (%) : C, 44.18; H, 4.29; N, 8.14; S, 18.63. Calculated (%): C, 44.18; H, 4.29; N, 8.14; S, 18.63.
実測値 (¾) : C, 44.04; H, 4.12; N, 8.34; S, 18.62. Found (¾): C, 44.04; H, 4.12; N, 8.34; S, 18.62.
実施例 7-43 Example 7-43
4 -シァノ -N- [5, 6 -ジメチル- 8- (ィソぺンチルスルファニル) _3 -ォキソ- 2, 3-ジヒ ド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン]ベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 4-シァノベンゼンスルホニル クロリド (203 mg, 1.01 腿 ol) を用いて、 実施例 6- で得られた卜アミノ- 5, 6 -ジメチル -8- (イソペンチルスル ファニル) -3H-イミダゾ [5, 卜 c] [1,4]チアジン- 3-オン (150 mg, 0.504龍 ol) か ら題記化合物 (207 mg, 89%) を合成した。 酢酸ェチル-ジェチルエーテルから再 結晶して融点 197_199°Cの暗赤色結晶を得た。 4-Cyano-N- [5,6-dimethyl-8- (isopentylsulfanyl) _3-oxo-2,3-dihydrogen-1H-imidazo [5,1-c] [1,4] Thiazine-triylidene] benzenesulfonamide Example 7-5 In the same manner as in Method C, using 4-cyanobenzenesulfonyl chloride (203 mg, 1.01 mol) instead of 4-bromomethylbenzenesulfonyl chloride. Triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one obtained in Example 6- (150 mg, The title compound (207 mg, 89%) was synthesized from 0.504 Ryuol). The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals with a melting point of 197-199 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3227, 2957, 2932, 2872, 2234, 1750, 1661, 1607, 1553 cm"1. Infrared absorption spectrum (IR) (in KBr): 3227, 2957, 2932, 2872, 2234, 1750, 1661, 1607, 1553 cm " 1 .
¾-NMR (CDCI3) : δ 0.942 (6Η, d, J=6.6 Hz), 1.51-1.78 (3H, m), 1.81 (3H, s), 2.29 (3H, d, J=0.6 Hz), 2.97 (2H, t, J=7.6 Hz), 7.79 (2H, d, J=8.4 Hz), 8.09 (2H, d, J=8.8 Hz), 9.59 (1H, bs) . ¾-NMR (CDCI3): δ 0.942 (6Η, d, J = 6.6 Hz), 1.51-1.78 (3H, m), 1.81 (3H, s), 2.29 (3H, d, J = 0.6 Hz), 2.97 ( 2H, t, J = 7.6 Hz), 7.79 (2H, d, J = 8.4 Hz), 8.09 (2H, d, J = 8.8 Hz), 9.59 (1H, bs).
元素分析値: C2。H22N403S3として Elemental analysis: C 2. H 22 N 4 0 3 As S 3
計算値 (%) : C, 51.93; H, 4.79; N, 12.11; S, 20.79 Calculated value (%): C, 51.93; H, 4.79; N, 12.11; S, 20.79
実測値 ) : C, 51.87; H, 4.76; N, 12.20; S, 20.80. Obtained): C, 51.87; H, 4.76; N, 12.20; S, 20.80.
実施例 7-44 ' 3 -シァノ- N - [5, 6-ジメチル- 8- (ィソペンチルスルファ二ル) - 3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン-卜イリデン]ベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 3-シァノベンゼンスルホニル クロリド (203 ing, 1.01 ininol) を用いて、 実施例 6-2で得られた 1-ァミノ- 5, 6-ジメチル- 8- (イソペンチルスル ファニル) -3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3-オン (150 mg, 0.504 mniol) か ら題記化合物 (216 mg, 93¾) を合成した。 酢酸ェチル -ジェチルエーテルから再 結晶して融点 133-135°Cの暗赤色結晶を得た。 Example 7-44 ' 3-cyano-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] Thiazine-triylidene] benzenesulfonamide Example 7-5 By a method similar to Method C, using 3-cyanobenzenesulfonyl chloride (203 ing, 1.01 ininol) instead of 4-bromomethylbenzenesulfonyl chloride, 1-Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-2 (150 mg, 0.504 mniol) to give the title compound (216 mg, 93¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 133-135 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3247, 3073, 2959, 2930, 2870, 2234, 1748, 1663, 1615, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3247, 3073, 2959, 2930, 2870, 2234, 1748, 1663, 1615, 1557 cm " 1 .
LH-NMR (CDC13) : δ 0.946 (6Η, d, J=6.4 Hz), 1.52-1.78 (3H, m), 1.81 (3H, s), L H-NMR (CDC1 3) : δ 0.946 (6Η, d, J = 6.4 Hz), 1.52-1.78 (3H, m), 1.81 (3H, s),
2.29 (3H, s), 2.99 (2H, t, J=7.6 Hz), 7.63 (1H, t, J=7.8 Hz), 7.80-7.86 (1H, m), 8.19 (1H, dt, J=8.2, 1.8 Hz), 8.29 (1H, t, J=1.8 Hz), 9.57 (1H, bs). 元素分析値: C2QH22N403S3'0.2H20として 2.29 (3H, s), 2.99 (2H, t, J = 7.6 Hz), 7.63 (1H, t, J = 7.8 Hz), 7.80-7.86 (1H, m), 8.19 (1H, dt, J = 8.2, 1.8 Hz), 8.29 (1H, t, J = 1.8 Hz), 9.57 (1H, bs). Elemental analysis: C 2Q H 22 N 4 0 3 S 3 '0.2H 2 0
計算値 (%) : C, 51.53; H, 4.84; N, 12.02; S, 20.63. Calculated (%): C, 51.53; H, 4.84; N, 12.02; S, 20.63.
実測値 (%) : C, 51.49; H, 4.81; N, 11.88; S, 20.46. Found (%): C, 51.49; H, 4.81; N, 11.88; S, 20.46.
実施例 7-45 Example 7-45
N- [5, 6 -ジメチル- 8- (イソペンチルスルファニル) -3 -ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン ]_2-チオフェンスルホンアミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] _2 -Thiophene sulfonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 2 -チオフェンスルホニル クロリド (195 nig, 0.536 ramol) を 用いて、 実施例 6-2で得られた 1-ァミノ- 5, 6 -ジメチル- 8- (イソペンチルスルフ ァニル )-3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (50.0 mg, 0.168匪 ol) から 題記化合物 (46.0mg, 62¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結 晶して融点 182-184°Cの暗赤色結晶を得た。 Example 7-5 According to the method similar to Method C, 2-thiophenesulfonyl chloride (195 nig, 0.536 ramol) was used in place of 4-bromomethylbenzenesulfonyl chloride to obtain Example 1-5. -Amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, 1-c] [1,4] thiazin-3-one (50.0 mg, 0.168 bandol) from the title compound (46.0 mg, 62 mg) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 182-184 ° C.
赤外吸収スペクトル(IR) (KBr中) V 3290, 2955, 2926, 2874, 1736, 1713, 1661, 1607, 1557, 1507 cm-1. Infrared absorption spectrum (IR) (in KBr) V 3290, 2955, 2926, 2874, 1736, 1713, 1661, 1607, 1557, 1507 cm- 1 .
Ή-NMR (CDC13) : δ 0.934 (6Η, d, J=6.2 Hz), 1.50-1.78 (3H, m), 1.80 (3H, d, J=l.0 Hz), 2.28 (3H, d, J=1.0Hz), 2.96 (2H, t, J-7.6Hz), 7.05 (1H, dd, J=4.8, 3.6 Hz), 7.57 (1H, dd, J=5.2, 1.0 Hz), 7.68 (1H, dd, J=3.2, 0.8 Hz), 9.56 (1H, bs). Ή-NMR (CDC1 3): δ 0.934 (6Η, d, J = 6.2 Hz), 1.50-1.78 (3H, m), 1.80 (3H, d, J = l.0 Hz), 2.28 (3H, d, J = 1.0Hz), 2.96 (2H, t, J-7.6Hz), 7.05 (1H, dd, J = 4.8, 3.6 Hz), 7.57 (1H, dd, J = 5.2, 1.0 Hz), 7.68 (1H, dd, J = 3.2, 0.8 Hz), 9.56 (1H, bs).
元素分析値: Cl7H21N303S4として Elementary analysis: as C l7 H 21 N 3 0 3 S 4
計算値 (%) : C, 46.03; H, 4.77; N, 9.47; S, 28.91, Calculated (%): C, 46.03; H, 4.77; N, 9.47; S, 28.91,
実測値 (%) : C, 46.01; H, 4.72; N, 9.59; S, 28.80. Found (%): C, 46.01; H, 4.72; N, 9.59; S, 28.80.
実施例 7 - 46 Example 7-46
5 -ブ口モ _N_ [5, 6-ジメチル -8- (ィソぺンチルスルファニル) -3-ォキソ -2, 3 -ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン ]-2-チオフェンスルホンァ $ド 5 -Buguchi _N_ [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine -1-ylidene] -2-thiophene sulfone
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 5-ブロモ -2-チオフェンスルホニル クロリド (352 mg, 1.34 腿 ol) を用いて、 実施例 6-2で得られた 1 ァミノ- 5, 6-ジメチル -8- (イソペンチ ルスルファニル) -3H -イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (200 mg, 0.672 mmol) から題記化合物 (272 mg, 78¾) を合成した。 酢酸エヂル-ジェチルェ一テ ルから再結晶して融点 163- 165 の暗赤色結晶を得た。 Example 7-5 Method 6-5 was repeated in the same manner as in Method C, except that 5-bromo-2-thiophenesulfonyl chloride (352 mg, 1.34 tmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. From 1-amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.672 mmol) (272 mg, 78 mg) was synthesized. Recrystallization from ethyl acetate-ethyl acetate yielded dark red crystals, mp 163-165.
赤外吸収スペクトル(IR) (KBr中) V: 3227, 2957, 1757, 1663, 1615, 1559, 1516 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3227, 2957, 1757, 1663, 1615, 1559, 1516 cm " 1 .
¾-NMR (CDC13) : δ 0.947 (6Η, d, J=6.6 Hz), 1.46-1.78 (3H, m), 1.81 (3H, d, J=1.2 Hz), 2.28 (3H, s), 2.99 (2H, t, J=8.0 Hz), 7.02 (2H, d, J=4.0 Hz), 7.41 (2H, d, J=4.0 Hz), 9.39 (1H, bs). ¾-NMR (CDC1 3): δ 0.947 (6Η, d, J = 6.6 Hz), 1.46-1.78 (3H, m), 1.81 (3H, d, J = 1.2 Hz), 2.28 (3H, s), 2.99 (2H, t, J = 8.0 Hz), 7.02 (2H, d, J = 4.0 Hz), 7.41 (2H, d, J = 4.0 Hz), 9.39 (1H, bs).
実施例 7-47 Example 7-47
N- [5, 6_、ジメチル- 8- (ィソペンチルスルファニル) -3-ォキソ -2, 3-ジヒド口- 1H -ィ ミダゾ [5,卜 c] [1,4]チアジン-卜イリデン] -4-ョードベンゼンスルホンアミド 実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンズルホニル ク 口リドの代わりに 4 -ョードベンゼンスルホニル クロリド (219 mg, 0.672 mmol) を用いて、 実施例 6 - 2で得られた卜ァミノ- 5, 6 -ジメチル -8- (イソペンチルスル ファニル)- 3H -イミダゾ [5,1- c] [1,4]チアジン- 3-オン (100 mg, 0.336 mmol) か ら題記化合物 (169 mg, 89¾) を合成した。 酢酸ェチル-ジェチルェ一テルから再 結晶して融点 186-188での暗赤色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) リ: 3229, 2953, 2928, 2907, 2868, 1759, 1663, 1620, 1557 cm"1. N- [5,6_, Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydrido-1H-imidazo [5, tric] [1,4] thiazine-triylidene]- 4-Eodobenzenesulfonamide Example 7-5 By the same method as in Method C, 4-bromoodobenzenesulfonyl chloride was substituted for 4-bromomethylbenzenesulfonyl chloride (219 mg, 0.672 mmol). And the triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazine-3- obtained in Example 6-2. The title compound (169 mg, 89%) was synthesized from ON (100 mg, 0.336 mmol). Recrystallization from ethyl acetate-jetil ether gave dark red crystals with melting point 186-188. Infrared absorption spectrum (IR) (in KBr): 3229, 2953, 2928, 2907, 2868, 1759, 1663, 1620, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 0.937 (6Η, d, J=6.2 Hz), 1.50-1.76 (3H, ni), 1.80 (3H, d, J=1.0 Hz), 2.28 (3H, d, J=1.0 Hz), 2.95 (2H, t, J=7.6 Hz), 7.68 (2H, d, J=8.8 Hz), 7; 85 (2H, d, 1=8.8 Hz), 9.58 (1H, bs). Ή-NMR (CDC1 3): δ 0.937 (6Η, d, J = 6.2 Hz), 1.50-1.76 (3H, ni), 1.80 (3H, d, J = 1.0 Hz), 2.28 (3H, d, J = 1.0 Hz), 2.95 (2H, t, J = 7.6 Hz), 7.68 (2H, d, J = 8.8 Hz), 7; 85 (2H, d, 1 = 8.8 Hz), 9.58 (1H, bs).
元素分析値: C19H22N303S3Iとして Elemental analysis: as C 19 H 22 N 3 0 3 S 3 I
計算値 (¾) : C, 40.50; H, 3.94; N, 7.46; S, 17.07. Calculated value (¾): C, 40.50; H, 3.94; N, 7.46; S, 17.07.
実測値 (%) : C, 40.53; H, 3.79; N, 7.60; S, 17.09. Found (%): C, 40.53; H, 3.79; N, 7.60; S, 17.09.
実施例 7 - 48 Example 7-48
2, 6-ジク口口- N- [5, 6 -ジメチル -8- (ィソペンチルスルファ二ル)- 3-ォキソ _2, 3 - ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン]ベンゼンスルホンアミ F 2,6-dic-mouth-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo_2,3-dihydro- 1H-imidazo [5,1-c] [ 1,4] thiazine-triylidene] benzenesulfonami F
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク 口リドの代わりに 2, 6 -ジクロ口ベンゼンスルホニル クロリド (174 mg, 0.672 mmol) を用いて、 実施例 6-2で得られた 1-ァミノ- 5, 6-ジメチル- 8- (イソペンチ ルスルファニル) -3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.336 mmol) から題記化合物 (161 mg, 95%) を合成した。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 198-200°Cの暗赤色結晶を得た。 Example 7-5 Method 6-2 was repeated in the same manner as in Method C, except that benzenesulfonyl chloride (174 mg, 0.672 mmol) was used instead of 4-bromomethylbenzenesulfonyl chloride. From the 1-amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, 1-c] [1,4] thiazin-3-one (100 mg, 0.336 mmol) obtained in The title compound (161 mg, 95%) was synthesized. Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals with a melting point of 198-200 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3250, 2957, 2932, 2868, 1736, 1717, 1661, 1611, 1553 cm"1. Infrared absorption spectrum (IR) (in KBr): 3250, 2957, 2932, 2868, 1736, 1717, 1661, 1611, 1553 cm " 1 .
ΐ-NMR (CDCI3) : δ 0.953 (6Η, d, J=6.2 Hz), 1.46-1.76 (3H, m), 1.81 (3H, s),ΐ-NMR (CDCI3): δ 0.953 (6Η, d, J = 6.2 Hz), 1.46-1.76 (3H, m), 1.81 (3H, s),
2.30 (3H, d, J=0.6 Hz), 2.94 (2H, t, J=7.6 Hz), 7.30 (1H, dd, J=9.2, 7.0 Hz),2.30 (3H, d, J = 0.6 Hz), 2.94 (2H, t, J = 7.6 Hz), 7.30 (1H, dd, J = 9.2, 7.0 Hz),
7.40-7.48 (2H, m) , 9.68 (1H, bs). 7.40-7.48 (2H, m), 9.68 (1H, bs).
元素分析値: C19H21N303S3C 12として Elementary analysis: as C 19 H 21 N 3 0 3 S 3 C 1 2
計算値 (%) : C, 45.06; H, 4.18; N, 8.30; S, 18.99. Calculated value (%): C, 45.06; H, 4.18; N, 8.30; S, 18.99.
実測値 (%) : C, 44.84; H, 4.13; N, 8.22; S, 18.82. Found (%): C, 44.84; H, 4.13; N, 8.22; S, 18.82.
実施例 7-49 Example 7-49
2, 4 -ジク口ロ- N- [5, 6 -ジメチル- 8- (ィソペンチルスルファニル) -3 ォキソ -2, 3- ジヒドロ- 1H-イミダゾ [5,1 - c] [1, 4]チアジン - 1 -ィリ H , 2,4-Dicyclo-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-Iri H,
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりに 2, 4-ジクロロベンゼンスルホニル クロリド (230 mg, 1.34 腿 ol) を用いて、 実施例 6 - 2で得られた卜ァミノ- 5, 6 -ジメチル- 8- (イソペンチ ルスルファニル) -3H -イミダゾ [5, 1- c][l,4]チアジン- 3 -オン (200 mg, 0.672 聽 ol) から題記化合物 (231 mg, 68%) を合成した。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 187-189°Cの暗赤色結晶を得た。 Example 7-5 In the same manner as in Method C, using 2,4-dichlorobenzenesulfonyl chloride (230 mg, 1.34 t ol) instead of 4-bromomethylbenzenesulfonyl chloride, the procedure of Example 6-2 was repeated. Title obtained from the obtained triamino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [l, 4] thiazin-3-one (200 mg, 0.672 listening). Compound (231 mg, 68%) was synthesized. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 187-189 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3281, 2955, 2934, 2468, 1750, 1725, 1663, 1615, 1557 cnf1. Infrared absorption spectrum (IR) (in KBr): 3281, 2955, 2934, 2468, 1750, 1725, 1663, 1615, 1557 cnf 1 .
Ή-NMR (CDC13) : δ 0.906 (6Η, d, J=6.2 Hz), 1.45-1.78 (3H, m), 1.81 (3H, d, J=0.8Hz), 2.30 (3H, d, J=l.0 Hz), 2.93 (2H, t, J=7.6 Hz), 7.38 (1H, dd, J=8.6, 2.0 Hz), 7.51 (1H, d, J=1.8 Hz), 8.12 (1H, d, J=8.4 Hz), 9.69 (1H, bs). 元素分析値: C19II21N303S3C12として Ή-NMR (CDC1 3): δ 0.906 (6Η, d, J = 6.2 Hz), 1.45-1.78 (3H, m), 1.81 (3H, d, J = 0.8Hz), 2.30 (3H, d, J = l.0 Hz), 2.93 (2H, t, J = 7.6 Hz), 7.38 (1H, dd, J = 8.6, 2.0 Hz), 7.51 (1H, d, J = 1.8 Hz), 8.12 (1H, d, J = 8.4 Hz), 9.69 ( 1H, bs) Elementary analysis:. a C 19 II 21 N 3 0 3 S 3 C1 2
計算値 (¾) : C, 45.06; H, 4.18; N, 8.30; S, 18.99. Calculated (¾): C, 45.06; H, 4.18; N, 8.30; S, 18.99.
実測値 (¾) : C, 45.09; H, 4.12; N, 8.39; S, 18.99. Found (¾): C, 45.09; H, 4.12; N, 8.39; S, 18.99.
実施例 7-50Example 7-50
-[5, 6-ジメチル -8- (ィソペンチルスルファニル) _3_ォキソ -2, 3-ジヒド口- 1H-ィ ミダゾ [5, 1-c] [1, 4]チアジン -1-ィリデン] -5-ジメチルァミノ-卜ナフタレンスル ホンアミド -[5, 6-Dimethyl-8- (isopentylsulfanyl) _3_oxo -2,3-dihydrido-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene]- 5-dimethylamino-naphthalenesulphonamide
実施例 7-5方法 Cと同様の方法により、 4-ブロモメチルベンゼンスルホニル ク ロリドの代わりにダニシル クロリド (185 mg, 0.672腿 ol) を用いて、 実施例 6-2で得られた 1-ァミノ- 5, 6-ジメチル -8 -(ィソペンチルスルファ二ル)- 3H-ィミ ダゾ [5, 1-c] [1, 4]チアジン -3-オン (100 mg, 0.336匪 ol)から題記化合物 (164 mg, 92¾) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 186- 188 の暗赤色結晶を得た。 Example 7-5 Method 1-Amino obtained in Example 6-2 was obtained in the same manner as in Method C, except that dansyl chloride (185 mg, 0.672 t ol) was used instead of 4-bromomethylbenzenesulfonyl chloride. -5,6-Dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, 1-c] [1, 4] thiazin-3-one (100 mg, 0.336 ol) The title compound (164 mg, 92%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 186-188.
赤外吸収スぺクトル(IR) (KBr中) V : 3241, 2955, 2868, 2832, 2786, 1732, 1663, 1615, 1557, 1505 cnf1. Infrared absorption spectrum (IR) (in KBr) V: 3241, 2955, 2868, 2832, 2786, 1732, 1663, 1615, 1557, 1505 cnf 1 .
Ή -匿 (CDC13) : δ 0.914 (6Η, d, J=6.2 Hz), 1.42-1.74 (3H, m), 1.75 (3H, d, J=1.2 Hz), 2.25 (3H, d, J=0.8 Hz), 2.87 (6H, s), 2.91 (2H, t, J=7.2 Hz), 7.16 (1H, dd, J=7.8, 0.8 Hz), 7.46-7.58 (3H, m), 8.28 (1H, dd, J=7.4, 1.2 Hz), 8.52 (1H, d, J=8.0 Hz), 8.59 (1H, d, J=8.8 Hz), 9.67 (1H, bs). E - Anonymous (CDC1 3): δ 0.914 ( 6Η, d, J = 6.2 Hz), 1.42-1.74 (3H, m), 1.75 (3H, d, J = 1.2 Hz), 2.25 (3H, d, J = 0.8 Hz), 2.87 (6H, s), 2.91 (2H, t, J = 7.2 Hz), 7.16 (1H, dd, J = 7.8, 0.8 Hz), 7.46-7.58 (3H, m), 8.28 (1H, dd, J = 7.4, 1.2 Hz), 8.52 (1H, d, J = 8.0 Hz), 8.59 ( 1H, d, J = 8.8 Hz), 9.67 (1H, bs).
元素分析値: C25¾。N403S3として Elemental analysis: C 25 ¾. As N 4 0 3 S 3
計算値 (%) : C, 56.58; H, 5.70; N, 10.56; S, 18.13. Calculated (%): C, 56.58; H, 5.70; N, 10.56; S, 18.13.
実測値 (¾) : C, 56.31; H, 5.62; N, 10.46; S, 17.92. Found (¾): C, 56.31; H, 5.62; N, 10.46; S, 17.92.
実施例 7-51 Example 7-51
N-{5, 6 -ジメチル -8- [(4·, 4, 5, 5, 5 -ペンタフルォロペンチル)スルファニル ]-3 -ォ キソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン } (フェニル) メタンスルホンアミド N- {5,6-dimethyl-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-triylidene} (phenyl) methanesulfonamide
方法 C Method C
水素化ナトリウム(41.3mg, 1.03mmol) (60 由性)のテトラヒドロフラン(2.00 ml) の懸濁液に、実施例 6-3で得られた 1-ァミノ- 5,6-ジメチル- 8- [ (4, 4, 5, 5, 5 - ペンタフルォ口ぺンチル)スルファニル] - 3H-ィミダゾ [5, 1 -C] [1, 4]チアジン - 3_ オン (200 mg, 0.516 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これにべンジルスルホニル クロリド (197 mg, 1.03 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 19時間かき混ぜた。 反応 混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (206 mg, 74¾) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 192-19'4°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (41.3 mg, 1.03 mmol) (freedom of 60) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8-[(obtained in Example 6-3. 4,4,5,5,5-pentapentafluoropentyl) sulfanyl] -3H-imidazo [5,1-C] [1,4] thiazine-3-one (200 mg, 0.516 mmol) in tetrahydrofuran (1.00 ml) The solution was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of benzylsulfonyl chloride (197 mg, 1.03 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 19 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (206 mg, 74¾) was synthesized. Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 192-19'4 ° C.
赤外吸収スペクトル(IR) (KBr中) レ: 3150, 3079, 2965, 2942, 2714, 1740, 1661, 1615, 1564, 1497 cm"1. Infrared absorption spectrum (IR) (in KBr) d: 3150, 3079, 2965, 2942, 2714, 1740, 1661, 1615, 1564, 1497 cm " 1 .
Ή一 NMR (CDC13) : δ 1.80 (3Η, d, J = 1.2 Hz), 1.98-2.36 (4H, m), 2.21 (3H, s), 3.06 (2H, t, J=7.4 Hz), 4.36 (2H, s), 7.32-7.47 (5H, m), 9.14 (1H, bs). 元素分析値: C2。 N303S3F5として Ή one NMR (CDC1 3): δ 1.80 (3Η, d, J = 1.2 Hz), 1.98-2.36 (4H, m), 2.21 (3H, s), 3.06 (2H, t, J = 7.4 Hz), 4.36 (2H, s), 7.32-7.47 ( 5H, m), 9.14 (1H, bs) elemental analysis:. C 2. N 3 0 3 S 3 F 5
計算値 (%) : C, 44.35; H, 3.72; N, 7.76; S, 17.76. Calculated value (%): C, 44.35; H, 3.72; N, 7.76; S, 17.76.
実測値 (%) : C, 44.47; H, 3.69; N, 7.60; S, 17.68. Found (%): C, 44.47; H, 3.69; N, 7.60; S, 17.68.
実施例 7 - 52 Example 7-52
4—ブロモ— N一 {5, 6-ジメチル- 3-ォキソ -8- [(4, 4, 5, 5, 5 -ペンタフルォロペンチル) スルファニル] -2, 3-ジヒド口 - 1H -ィミダゾ [5, 1-c] [1 , ]チアジンー 1ーィリデン }ベ ンゼンスルホンアミド 4 - bromo - N one {5, 6-dimethyl - 3- Okiso 8- [(4, 4, 5, 5, 5 - penta Full O b pentyl) sulfanyl] -2, 3-Jihido port - IH - Imidazo [5, 1-c] [1,] thiazin-1-ylidene Nzensulfonamide
方法 C Method C
水素化ナトリウム(82.6 mg, 2.07腿01) (60%油性)のテトラヒドロフラン(4.00 ml) の懸濁液に、 実施例 6-3で得られた卜アミノ- 5, 6-ジメチル -8- [(4, 4, 5,5, 5 - ペン夕フルォロぺンチル)スルファニル] - 3H-ィミダゾ [5,卜 c] [1 , 4]チアジン- 3- オン (400 mg, 1.03匪 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに 4-ブロモベンゼンスルホニル クロリド(538 mg, 2.07 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 さらに 40 かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (602 mg, 96¾) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 189- 191°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (82.6 mg, 2.07 thigh 01) (60% oily) in tetrahydrofuran (4.00 ml) was added the triamino-5,6-dimethyl-8-[( 4,4,5,5,5-Penyu fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (400 mg, 1.03 bandol) in tetrahydrofuran (2.00 ml), and the mixture was stirred under ice-cooling for 5 minutes. To this was added a solution of 4-bromobenzenesulfonyl chloride (538 mg, 2.07 mmol) in tetrahydrofuran (2.00 ml), and the mixture was further stirred for 40 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (602 mg, 96¾) was synthesized. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 189-191 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3237, 3090, 2932, 1759, 1665, 1622, 1563 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3237, 3090, 2932, 1759, 1665, 1622, 1563 cm " 1 .
^- MR (CDC13) : δ 1.80 (3Η, d, J=0.9 Hz), 1.95-2.26 (4H, m), 2.28 (3H, d, J=1.2Hz), 3.03 (2H, t, J=7.2 Hz), 7.63 (2H, d, J=8.7Hz), 7.82 (2H, d, J=9.0 ^ - MR (CDC1 3): δ 1.80 (3Η, d, J = 0.9 Hz), 1.95-2.26 (4H, m), 2.28 (3H, d, J = 1.2Hz), 3.03 (2H, t, J = 7.2 Hz), 7.63 (2H, d, J = 8.7Hz), 7.82 (2H, d, J = 9.0
Hz), 9.60 (1H, bs). Hz), 9.60 (1H, bs).
元素分析値: CigH17N303S3BrF5として Elementary analysis: as C ig H 17 N 3 0 3 S 3 BrF 5
計算値 (¾) : C, 37.63; H, 2.83; N, 6.93; S, 15.86. Calculated (¾): C, 37.63; H, 2.83; N, 6.93; S, 15.86.
実測値 (%) :' C, 37.79; H, 2.84; N, 7.04; S, 15.85. Found (%): 'C, 37.79; H, 2.84; N, 7.04; S, 15.85.
実施例 7-53 Example 7-53
N-{5, 6 -ジメチル- 3-ォキソ - 8-[(4, 4, 5, 5, 5-ペン夕フルォロペンチル)スルファニ ル]- 2,3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン卜 4-フルォロ ベンゼンスルホンアミド N- {5,6-dimethyl-3-oxo-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl]-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-trident 4-fluorobenzenesulfonamide
実施例 7- 51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4- フルォロベンゼンスルホニル クロリド (311 mg, 1.55誦 ol) を用いて、 実施例 6-3 で得られた 1-ァミノ- 5, 6 -ジメチル- 8 - [(4, 4, 5, 5, 5-ペン夕フルォロペンチ ル)スルファニル] - 3H-イミダゾ [5, 1 - c] [1,4]チアジン - 3-オン (312 mg, 74%) か ら題記化合物 (602 mg, 96%) を合成した。 酢酸ェチル-ジェチルエーテルから再 結晶して融点 189- 190°Cの暗赤色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) ν 3227, 3079, 2948, 1759, 1665, 1622, 1593, 1564, 1497 cm"1. According to a method similar to that of Example 7-51, 4-fluorobenzenesulfonyl chloride (311 mg, 1.55 recited ol) was used instead of benzylsulfonyl chloride, and the 1-amino- 5,6-Dimethyl-8-[(4,4,5,5,5-pentafluorofluoropentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (312 mg, 74%) to give the title compound (602 mg, 96%). The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 189-190 ° C. Infrared absorption spectrum (IR) (in KBr) ν 3227, 3079, 2948, 1759, 1665, 1622, 1593, 1564, 1497 cm ” 1 .
Ή -匪 R (CDC13) : δ 1.80 (3Η, d, J=0.9 Hz), 1.95-2.26 (4H, m), 2.28 (3H, d, J=1.2 Hz), 3.02 (2H, t, J=7.2 Hz), 7.17 (2H, d, J=8.4 Hz), 7.94-8.00 (2H, m), 9.60 (1H, bs). Ή-Maraudal R (CDC1 3 ): δ 1.80 (3Η, d, J = 0.9 Hz), 1.95-2.26 (4H, m), 2.28 (3H, d, J = 1.2 Hz), 3.02 (2H, t, J) = 7.2 Hz), 7.17 (2H, d, J = 8.4 Hz), 7.94-8.00 (2H, m), 9.60 (1H, bs).
元素分析値: C19H17N303S3F6として Elementary analysis: as C 19 H 17 N 3 0 3 S 3 F 6
計算値 ( ) : C, 41.83; H, 3.14; N, 7.70; S, 17.63. Calculated (): C, 41.83; H, 3.14; N, 7.70; S, 17.63.
実測値 : C, 41.99; H, 3.19; N, 7.89; S, 17.82. Found: C, 41.99; H, 3.19; N, 7.89; S, 17.82.
実施例 7-54 Example 7-54
5-ク口口- N - {5, 6 -ジメチル -3-ォキソ - 8-[(4, 4, 5, 5, 5 -ペン夕フルォロペンチル) スルファニル ] -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-イリデ ン}-2-チオフェンスルホンアミド 5-kuguchi-N- {5,6-dimethyl-3-oxo- 8-[(4,4,5,5,5-pentylfluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [ 5, tric] [1,4] thiazine-1-ylidene} -2-thiophene sulfonamide
実施例 7- 51と同様の方法により、ベンジルスルホニル クロリドの代わりに 5- クロロチォフェン- 2-スルホニル クロリド (231 mg, 1.03mmol) を用いて、 実施 例 6-3で得られた卜ァミノ- 5, 6 -ジメチル- 8_[(4, 4, 5, 5, 5 -ペンタフルォロペンチ ル)スルファニル ]-3H -イミダゾ [5,卜 c] [1, 4]チアジン- 3 -オン (200 mg, 0.516 蘭 ol) から題記化合物 (242 mg, 83¾) を合成した。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 160-162°Cの暗赤色結晶を得た。 According to a method similar to that of Example 7-51, 5-chlorothiophene-2-sulfonyl chloride (231 mg, 1.03 mmol) was used in place of benzylsulfonyl chloride, and the amino acid obtained in Example 6-3 was used. 5,6-Dimethyl-8 _ [(4,4,5,5,5-pentafluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.516 orchid ol) to give the title compound (242 mg, 83¾). Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 160-162 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3223, 3081, 2926, 1755, 1663, 1617, 1559, 1520 cm-1. Infrared absorption spectrum (IR) (in KBr) V: 3223, 3081, 2926, 1755, 1663, 1617, 1559, 1520 cm- 1 .
'H-NMR (CDCI3) : δ 1.8Γ(3Η, d, J-1.0 Hz), 1.96-2.26 (4H, m), 2.29 (3H, d, J=1.0 Hz), 3.06 (2H, t, J=7.0 Hz), 6.89 (2H, d, J=4.0 Hz), 7.46 (2H, d, J=4.0 Hz), 9.51 (1H, bs). 'H-NMR (CDCI3): δ 1.8Γ (3Η, d, J-1.0 Hz), 1.96-2.26 (4H, m), 2.29 (3H, d, J = 1.0 Hz), 3.06 (2H, t, J = 7.0 Hz), 6.89 (2H, d, J = 4.0 Hz), 7.46 (2H, d, J = 4.0 Hz), 9.51 (1H, bs).
元素分析値: C17H15N303S4CIF5として Elementary analysis: as C 17 H 15 N 3 0 3 S 4 CIF 5
計算値 (%) : C, 35.95; H, 2.66; N, 7.40; S, 22.58. , 実測値 (¾) : C, 35.92; H, 2.73; N, 7.38; S, 22.57. Calculated value (%): C, 35.95; H, 2.66; N, 7.40; S, 22.58., Measured value (¾): C, 35.92; H, 2.73; N, 7.38; S, 22.57.
実施例 7-55 Example 7-55
4 -ブロモ -N-{5, 6-ジメチル- 8- [(3-フルォロ- 3-メチルプチル)スルファニル] - 3- ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }ベンゼン スルホンアミド 4-bromo-N- {5,6-dimethyl-8-[(3-fluoro-3-methylbutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] thiazine-1-ylidene} benzene Sulfonamide
実施例 7-51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4- ブロモベンゼンスルホニル クロリド (230nig, 1.34腿 ol) を用いて、 実施例 6- 4 で得られた 1-ァミノ- 5, 6 -ジメチル -8- [ (3-フルォ口 - 3-メチルプチル)スルファ 二ル]- 3H-イミダゾ [5,1- c] [1, 4]チアジン - 3 -オン (200 mg, 0.634 mmol) から題 記化合物 (284 mg, 84%) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶 して融点 160_162°Cの暗赤色結晶を得た。 According to a method similar to that of Example 7-51, 4-bromobenzenesulfonyl chloride (230 nig, 1.34 mol) was used instead of benzylsulfonyl chloride, and 1-amino-5,6 obtained in Example 6-4 was used. -Dimethyl-8-[(3-Fluoro mouth-3-methylbutyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] Thiazin-3-one (200 mg, 0.634 mmol) The title compound (284 mg, 84%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 160-162 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3231, 2980, 2938, 1759, 1663, 1622, 1559, 1470 cur1. Infrared absorption spectrum (IR) (in KBr): 3231, 2980, 2938, 1759, 1663, 1622, 1559, 1470 cur 1 .
Ή-NMR (CDC13) : δ 1.40 (6Η, d, J=21.4Hz), 1.80 (3H, d, J=0.8 Hz), 1.88 - 2.08 (2H, m), 2.28 (3H, s), 3.01-3.12 (2H, m), 7.63 (2H, d, J=8.4 Hz), 7.83 (2H, d, J=8.4 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 1.40 (6Η, d, J = 21.4Hz), 1.80 (3H, d, J = 0.8 Hz), 1.88 - 2.08 (2H, m), 2.28 (3H, s), 3.01 -3.12 (2H, m), 7.63 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.60 (1H, bs).
実施例 7-56 Example 7-56
5-ブロモ _N - {5, 6 -ジメチル- 8- [(3, 3 -ジメチルブチル)スルファニル ] -3-ォキソ -2, 3-,ジヒドロ- 1H-イミダゾ [5,l-c] [1,4]チアジン- 1 -ィリデン }ベンゼンスルホ ンアミド 5-bromo_N- {5,6-dimethyl-8-[(3,3-dimethylbutyl) sulfanyl] -3-oxo-2,3-, dihydro-1H-imidazo [5, lc] [1,4] Thiazine-1-ylidene} benzenesulfonamide
実施例 7-51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4- ブロモベンゼンスルホニル クロリド (493 mg, 1.89 mmol) を用いて、 実施例 6 - 5 で得られた 卜ァミノ- 5,6-ジメチル- 8-[(3,3-ジメチルブチル)スルファニ ル] -3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (300 mg, 0.945 mmol) から題記 化合物 (379 mg, 76¾) を合成した。 酢酸ェチル-ジェチルェ一テルから再結晶し て融点 210- 212°Cの暗赤色結晶を得た。 According to a method similar to that of Example 7-51, 4-bromobenzenesulfonyl chloride (493 mg, 1.89 mmol) was used in place of benzylsulfonyl chloride, and the triamino-5,6- obtained in Example 6-5 was used. Dimethyl-8-[(3,3-dimethylbutyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (300 mg, 0.945 mmol) from the title compound (379 mg, 76¾) was synthesized. The crystals were recrystallized from ethyl acetate-jetil ether to obtain dark red crystals having a melting point of 210-212 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3285, 2949, 2909, 1750, 1715, 1657, 1611, 1557 cm'1. Infrared absorption spectrum (IR) (in KBr) V: 3285, 2949, 2909, 1750, 1715, 1657, 1611, 1557 cm ' 1 .
Ή-NMR (CDCI3) : δ 0.945 (9Η, s), 1.51-1.63 (2H, m), 1.80 (3H, s), 2.28 (3H, d, J=0.8Hz), 2.84-2.98 (2H, m), 7.63 (2H, d, J=8. Hz), 7.83 (2H, d, J=8.8 Hz), 9.60 (1H, bs). Ή-NMR (CDCI3): δ 0.945 (9Η, s), 1.51-1.63 (2H, m), 1.80 (3H, s), 2.28 (3H, d, J = 0.8 Hz), 2.84-2.98 (2H, m ), 7.63 (2H, d, J = 8 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.60 (1H, bs).
実施例 7-57 Example 7-57
4-ブロモ -N - {5, 6-ジメチル -3-ォキソ - 8 - [(5, 5;6, 6, 6-ぺ スルファニル] -2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, ]チアジン- 1-ィリデン }ベ ンゼンスルホンアミド 4-bromo-N- {5,6-dimethyl-3-oxo-8-[(5,5; 6,6,6- ぺ Sulfanyl] -2,3-dihydrido-1H-imidazo [5, 1-c] [1,] thiazine-1-ylidene} benzenesulfonamide
実施例 7- 51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4 - ブロモベンゼンスルホニル クロリド (493 mg, 1.89腿 ol) を用いて、 実施例 6- 6 で得られた 1-ァミノ- 5, 6 -ジメチル -8- [(5, 5, 6, 6, 6 -ペンタフルォ口へキシル)ス ルファ二ル]- 3H -イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (300 mg, 0.770腿 ol) から題記化合物 (330 mg, 69¾) を合成した。 酢酸ェチル-ジェチルエーテルから 再結晶して融点 166- 168°Cの暗赤色結晶を得た。 According to a method similar to that of Example 7-51, 4-bromobenzenesulfonyl chloride (493 mg, 1.89 mol) was used in place of benzylsulfonyl chloride, and 1-amino-5,5 obtained in Example 6-6 was used. 6-dimethyl-8-[(5,5,6,6,6-pentahexafluorohexyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (300 The title compound (330 mg, 69 mg) was synthesized from the compound (0.7 mg, 0.770 t). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 166-168 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3233, 3090, 2926, 2859, 1759, 1665, 1622, 1561 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3233, 3090, 2926, 2859, 1759, 1665, 1622, 1561 cm- 1 .
Ή-NMR (CDC13) : δ 1.64-1.68 (4Η, ι), 1.80 (3H, s), 1.88-2.22 (2H, 1), 2.28 (3H, s), 2.98 (2H, t, J=7.0 Hz), 7.63 (2H, d, J=8.8 Hz), 7.82 (2H, d, J-8.8 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): δ 1.64-1.68 (4Η, ι), 1.80 (3H, s), 1.88-2.22 (2H, 1), 2.28 (3H, s), 2.98 (2H, t, J = 7.0 Hz), 7.63 (2H, d, J = 8.8 Hz), 7.82 (2H, d, J-8.8 Hz), 9.61 (1H, bs).
元素分析値: C2。H19N303S3BrF5として Elemental analysis: C 2. H 19 N 3 0 3 S 3 As BrF 5
計算値 (%) : C, 38.71; H, 3.09; N, 6.77; S, 15.50. Calculated (%): C, 38.71; H, 3.09; N, 6.77; S, 15.50.
実測値 (¾) : C, 38.74; H, 2.99; N, 6.67; S, 15.40. Found (¾): C, 38.74; H, 2.99; N, 6.67; S, 15.40.
実施例 7-58 Example 7-58
4-ク口ロ- N-{5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシ ル)スルファニル ]-2, 3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1, 4]チアジン- 1-ィリデ ン}ベンゼンスルホンアミド , 4-N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro- 1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene} benzenesulfonamide,
実施例 7-51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4- クロ口ベンゼンスルホニル クロリド (398mg, 1.88匪 ol) を用いて、 実施例 6 - 6 で得られた 1 -ァミノ- 5, 6 -ジメチル -8- [(5, 5, 6, 6, 6 -ペンタフルォ口へキシル)ス ルファニル] -3H -イミダゾ [5, 1-c] [1,4]チアジン - 3 -オン (378 mg, 0.942 mmol) から題記化合物 (319 mg, 59¾) を合成した。 酢酸ェチル-ジェチルエーテルから 再結晶して融点 183- 185°Cの暗赤色結晶を得た。 According to a method similar to that of Example 7-51, benzylsulfonyl chloride was replaced by 4-chlorobenzenebenzenesulfonyl chloride (398 mg, 1.88 ol), to obtain 1-amino-5,5 obtained in Example 6-6. 6-dimethyl-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (378 mg , 0.942 mmol) to give the title compound (319 mg, 59¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 183-185 ° C.
赤外吸収スペクトル(IR) (KBr中) V : 3241, 2948, 1759, 1665, 1622, 1559 era—1. Ή-NMR (CDCI3) : δ 1.65-1.85 (4Η, m), 1.80 (3H, d, J=0.9 Hz), 1.96-2.16 (2H, m), 2.28 (3H, d, J=0.9 Hz), 2.98 (2H, t, J=7.2 Hz), 7.46 (2H, dt, J=9.0, 2.1 Hz), 7.90 (2H, dt, J=8.7, 1.8 Hz), 9.59 (1H, bs). Infrared absorption spectrum (IR) (in KBr) V: 3241, 2948, 1759, 1665, 1622, 1559 era— 1. Ή-NMR (CDCI3): δ 1.65-1.85 (4Η, m), 1.80 (3H, d , J = 0.9 Hz), 1.96-2.16 (2H, m), 2.28 (3H, d, J = 0.9 Hz), 2.98 (2H, t, J = 7.2 Hz), 7.46 (2H, dt, J = 9.0, 2.1 Hz), 7.90 (2H, dt, J = 8.7, 1.8 Hz), 9.59 (1H, bs).
元素分析値: C2QH19N303S3C1F5として Elemental analysis: as C 2Q H 19 N 3 0 3 S 3 C1F 5
計算値 (¾) : C, 41.70; H, 3.32; N, 7.29; S, 16.70. Calculated (¾): C, 41.70; H, 3.32; N, 7.29; S, 16.70.
実測値 (%) : C, 41.55; H, 3.21; , 7.10; S, 16.85. Found (%): C, 41.55; H, 3.21;, 7.10; S, 16.85.
実施例 7 - 59 Example 7-59
4 -プロモ {5, 6 -ジメチル -3 -ォキソ -8- [(5, 5, 5- ァニル] - 2, 3-ジヒドロ- 1H-イミダゾ [5 ,卜 c] [1 , 4]チアジン- 1 -イリデン }ベンゼン スルホンアミド 4-Promo {5,6-dimethyl-3-oxo-8-[(5,5,5-anil) -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1 -Ylidene} benzene sulfonamide
実施例 7-51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4 - ブロモベンゼンスルホニル クロリド (445mg, 1.71 mmol) を用いて、 実施例 6-7 で得られた トァミノ- 5, 6-ジメチル -8 - [(5, 5, 5-トリフルォロペンチル)スルファ ニル] -3H-イミダゾ [5,1- c] [1,4]チアジン- 3-オン (300 mg, 0.854 mmol) から題 記化合物 (356 mg, 73%) を合成した。 酢酸ェチル-ジェチルェ一テルから再結晶 して融点 179-180°Cの暗赤色結晶を得た。 According to a method similar to that of Examples 7-51, 4-aminobenzenesulfonyl chloride (445 mg, 1.71 mmol) was used in place of benzylsulfonyl chloride, and the amino-5,6-dimethyl- obtained in Example 6-7 was used. 8-[(5,5,5-Trifluoropentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (300 mg, 0.854 mmol) from the title compound (356 mg, 73%) was synthesized. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 179-180 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3245, 2932, 1759, 1746, 1665, 1622, 1559 cm-1. Infrared absorption spectrum (IR) (in KBr): 3245, 2932, 1759, 1746, 1665, 1622, 1559 cm -1 .
-腹 (CDC13) : δ 1.62-1.82 (4Η, ), 1.80 (3H, d, J=1.2 Hz), 2.04-2.21 (2H, m), 2.28 (3H, d, J=0.6 Hz), 2.97 (2H, t, J=7.5 Hz), 7.63 (2H, dt, J=8.7, 2.1 Hz), 7.86 (2H, dt, J=9.0, 2.1 Hz), 9.59 (1H, bs). - belly (CDC1 3): δ 1.62-1.82 ( 4Η,), 1.80 (3H, d, J = 1.2 Hz), 2.04-2.21 (2H, m), 2.28 (3H, d, J = 0.6 Hz), 2.97 (2H, t, J = 7.5 Hz), 7.63 (2H, dt, J = 8.7, 2.1 Hz), 7.86 (2H, dt, J = 9.0, 2.1 Hz), 9.59 (1H, bs).
元素分析値: Cl9H19N303S3BrF3として Elementary analysis: as C l9 H 19 N 3 0 3 S 3 BrF 3
計算値 (¾) : C, 40.00; H, 3.36; N, 7.37; S, 16.86. Calculated (¾): C, 40.00; H, 3.36; N, 7.37; S, 16.86.
実測値 (¾) : C, 39.95; H, 3.24; N, 7.39; S, 16.92. Found (¾): C, 39.95; H, 3.24; N, 7.39; S, 16.92.
実施例 7 - 60 Example 7-60
4 -ブロモ -N- { 5, 6 -ジメチル -3-ォキソ -8- [ (4, 4, 4-トリフルォロプチル)スルファ 二ル]- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }ベンゼンス ルホンアミド 4-bromo-N- {5,6-dimethyl-3-oxo-8-[(4,4,4-trifluoropropyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1 -c] [1,4] thiazine-1-ylidene} benzenesulfonamide
実施例 7- 51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4- プロモベンゼンスルホニル クロリド (275 mg, 1.30删 ol) を用いて、 実施例 6-8 で得られた 卜ァミノ- 5, 6 -ジメチル- 8 - [ (4, 4, 4-トリフルォロブチル)スルファニ ル] - 3H -イミダゾ [5, 1- c] [1,4]チアジン- 3-オン (220 mg, 0.652腿 ol) から題記 化合物 (298 mg, 82%) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶し て融点 177- 179°Cの暗赤色結晶を得た。 According to a method similar to that of Example 7-51, 4-bromobenzenesulfonyl chloride (275 mg, 1.30 mol) was used in place of benzylsulfonyl chloride, and the amino and triamino-5, 6 obtained in Example 6-8 were used. -Dimethyl-8-[(4,4,4-trifluorobutyl) sulfani [3] -H-imidazo [5,1-c] [1,4] thiazin-3-one (220 mg, 0.652 t ol) to give the title compound (298 mg, 82%). The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 177-179 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3233, 2928, 1761, 1665, 1622, 1561 cm"1. 'H-NMR (CDC13) : δ 1.80 (3Η, d, J=1.2 Hz), 1.97 (2H, quintet, J=7.2 Hz), 2.13-2.30 (2H, m), 2.28 (3H, d, J=0.9 Hz), 3.01 (2H, t, J-7.2 Hz), 7.64 (2H, dt, J=8.7, 2.1 Hz), 7.82 (2H, dt, J=9.0, 2.4 Hz), 9.60 (1H, bs). Infrared absorption spectrum (IR) (in KBr) Re:. 3233, 2928, 1761, 1665, 1622, 1561 cm "1 'H-NMR (CDC1 3): δ 1.80 (3Η, d, J = 1.2 Hz), 1.97 (2H, quintet, J = 7.2 Hz), 2.13-2.30 (2H, m), 2.28 (3H, d, J = 0.9 Hz), 3.01 (2H, t, J-7.2 Hz), 7.64 (2H, dt , J = 8.7, 2.1 Hz), 7.82 (2H, dt, J = 9.0, 2.4 Hz), 9.60 (1H, bs).
元素分析値: Cl8H17N303S3BrF3として Elementary analysis: as C l8 H 17 N 3 0 3 S 3 BrF 3
計算値 (%) : C, 38.85; H, 3.08; N, 7.55; S, 17.29. Calculated value (%): C, 38.85; H, 3.08; N, 7.55; S, 17.29.
実測値 (¾) : C, 38.89; H, 3.13; N, 7.49; S, 17.25. Found (¾): C, 38.89; H, 3.13; N, 7.49; S, 17.25.
実施例 7 - 61 Example 7-61
4-クロ Π-Ν-{5, 6 -ジメチル- 3-ォキソ -8- [(4, 4, 4-トリフルォロブチル)スルファ 二ル]- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン 1 -ィリデン }ベンゼンス ルホンアミド 4-chloro Π-Ν- {5,6-dimethyl-3-oxo-8-[(4,4,4-trifluorobutyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine 1-ylidene} benzenesulfonamide
実施例 7 - 51と同様の方法により、ベンジルスルホニル クロリドの代わりに 4- クロ口ベンゼンスルホニル クロリ ド (275 mg, 1,30腿 ol) を用いて、 実施例 6-8 で得られた トァミノ- 5, 6 -ジメチル- 8- [ (4, 4, 4 -トリフルォ口ブチル)スルファニ ル] -3H -イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (220 mg, 0.652腿 ol) から題記 化合物 (207 mg, 62%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 171- 173°Cの喑赤色結晶を得た。 According to a method similar to that of Examples 7-51, 4-amino benzenesulfonyl chloride (275 mg, 1,30 t ol) was used in place of benzylsulfonyl chloride, and the amino-amino compound obtained in Example 6-8 was used. 5,6-Dimethyl-8-[(4,4,4-trifluorobutyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (220 mg, 0.652 t )) To give the title compound (207 mg, 62%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain red-red crystals having a melting point of 171-173 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3233, 3094, 2936, 1761, 1665, 1624, 1563 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3233, 3094, 2936, 1761, 1665, 1624, 1563 cm- 1 .
Ή-NMR (CDCI3) : δ 1.80 (3Η, d, J=1.2 Hz), 1.97 (2H, quintet, J=7.5 Hz), 2.13-2.30 (2H, m), 2.28 (3H, d, J=0.9 Hz), 3.01 (2H, t, J=7.5 Hz), 7.47 (2H, dt, J=9.0, 2.1 Hz), 7.90 (2H, dt, J=9.0, 2.4 Hz), 9.61 (1H, bs). Ή-NMR (CDCI3): δ 1.80 (3Η, d, J = 1.2 Hz), 1.97 (2H, quintet, J = 7.5 Hz), 2.13-2.30 (2H, m), 2.28 (3H, d, J = 0.9 Hz), 3.01 (2H, t, J = 7.5 Hz), 7.47 (2H, dt, J = 9.0, 2.1 Hz), 7.90 (2H, dt, J = 9.0, 2.4 Hz), 9.61 (1H, bs).
元素分析値: C18H17N303S3C1F3として Elementary analysis: as C 18 H 17 N 3 0 3 S 3 C1F 3
計算値 (¾) : C, 42.23; H, 3.35; N, .8.21; S, 18.79. Calculated value (¾): C, 42.23; H, 3.35; N, .8.21; S, 18.79.
実測値 (%) : C, 42.24; H, 3.20; N, 8.26; S, 18.85. Found (%): C, 42.24; H, 3.20; N, 8.26; S, 18.85.
実施例 7 - 62 N-{5, 6 -ジメチル- 3-ォキソ -8- [(4, 4, 5, 5, 5-ペン夕フルォロペンチル)スルファニ ル]- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }- 4 -ヒドロキ シメチルベンゼンスルホンアミド Example 7-62 N- {5,6-Dimethyl-3-oxo-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl]-2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-1-ylidene} -4-hydroxymethylbenzenesulfonamide
水素化ナトリウム(145 mg, 3.61匪01) (60%油性)のテトラヒドロフラン(15.0 ml) の懸濁液に、 実施例 6 - 3で得られた 1-ァミノ- 5, 6 -ジメチル -8 - [(4, 4, 5,5,5- ペン夕フルォロペンチル)スルファニル] - 3H-ィミダゾ [5 , 1 -C] [1, 4]チアジン - 3- オン (700 mg, 1.81 mmol) のテトラヒドロフラン (4.00 ml) 溶液を加え、 氷冷 下 10 分間かき混ぜた。 これに 4-ブロモメチルベンゼンスルホニル クロリド (1.00 g, 3.61 匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1 時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を 水洗、 乾燥 (MgS04) '後、 溶媒を減圧下に留去して 4- (プロモメチル) -N_[5,6 -ジ メチル -3-ォキソ -8- [(4, 4, 5, 5, 5-ペンタフルォロペンチル)スルファニル] - 2, 3- ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-イ リデン]ベンゼンスルホンアミ ド (220mg, 20¾) を暗赤色結晶として得た。 本品はこれ以上精製することなく次 の反応に使用した。 To a suspension of sodium hydride (145 mg, 3.61 band01) (60% oil) in tetrahydrofuran (15.0 ml) was added 1-amino-5,6-dimethyl-8- [ (4,4,5,5,5-pentanofluoropentyl) sulfanyl] -3H-imidazo [5,1-C] [1,4] thiazin-3-one (700 mg, 1.81 mmol) in tetrahydrofuran (4.00 ml ) The solution was added, and the mixture was stirred under ice cooling for 10 minutes. To this was added a solution of 4-bromomethylbenzenesulfonyl chloride (1.00 g, 3.61 ol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) 'After the solvent was distilled off under reduced pressure 4- (Promo methyl) -N_ [5,6 - di methyl-3- Okiso 8- [(4, 4, 5,5,5-pentafluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] benzenesulfonamide (220 mg, 20¾) was obtained as dark red crystals. This product was used for the next reaction without further purification.
上記の 4 -(ブロモメチル) -N- [5, 6 -ジメチル -3-ォキソ -8- [(4, 4, 5, 5, 5-ペン夕 フルォロペンチル)スルファニル ]-2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チア ジン-卜イ リデン]ベンゼンスルホンアミ ド (220 mg, 0.355腿 ol) と酢酸力リウ ム (105 mg, 1.06匪 ol) と水酸化カリウム (70.2 mg, 1.06讀 ol) のァセトニト リル (4.00ml) 懸濁液を 50分間加熱還流した。 反応混合物に 1N塩酸を加えて酢 酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去した。 残留物をシリカゲル力ラムクロマトグラフィーに付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して酢酸 4 - {[5, 6 -ジメチル- 3 -ォキソ - 8-[(4, 4, 5, 5,5-ペン 夕フルォロペンチル)スルファニル] -2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1, 4]チ アジン- 1-ィリデン]スルフィンナモイルォキシ}ベンジル エステル (42.0 mg, 20¾) を得た。 本品はこれ以上精製することなく次の反応に使用した。 4- (Bromomethyl) -N- [5,6-dimethyl-3-oxo-8-[(4,4,5,5,5-pentylfluoropentyl) sulfanyl] -2,3-dihydro-1H- Imidazo [5,1-c] [1,4] thiazine-trilidene] benzenesulfonamide (220 mg, 0.355 t ol), potassium acetate (105 mg, 1.06 bandol) and potassium hydroxide ( A suspension of 70.2 mg, 1.06 dl) of acetonitrile (4.00 ml) was heated to reflux for 50 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give acetic acid 4-{[5,6-dimethyl-3-oxo- 8- [ (4,4,5,5,5-Pen fluoropentyl) sulfanyl] -2,3-Dihydrido-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene] sulfinnamoylo Xyl} benzyl ester (42.0 mg, 20%) was obtained. This product was used for the next reaction without further purification.
赤外吸収スペクトル(IR) (KBr中) レ : 3210, 2938, 1748, 1667, 1622, 1559 cm"1. Ή-N R (CDC13) : δ 1.80 (3Η, s), 1.92-2.24 (4H, m), 2.13 (3H, s), 2.27 (3H, s), 3.02 (2H, t, J=6.6 Hz), 5.16 (2H, s), 7.47 (2H, d, J=8.8 Hz), 7.95 (2H, d, J=8.0 Hz), 9.62 (1H, bs). Infrared absorption spectrum (IR) (in KBr) Les:. 3210, 2938, 1748, 1667, 1622, 1559 cm "1 Ή-NR (CDC1 3): δ 1.80 (3Η, s), 1.92-2.24 (4H, m), 2.13 (3H, s), 2.27 (3H, s), 3.02 (2H, t, J = 6.6 Hz), 5.16 (2H, s), 7.47 (2H, d, J = 8.8 Hz), 7.95 ( 2H, d, J = 8.0 Hz), 9.62 (1H, bs).
上記の酢酸 4-{[5, 6-ジメチル- 3 -ォキソ - 8- [(4, 4, 5, 5, 5-ペンタフルォロペン チル)スルファニル ]-2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-イリデ ン]スルフィンナモイルォキシ}ベンジルエステル (42.0mg, 0.0700讓 ol) をテ トラヒドロフラン (1.00 ml) とエタノール (1.00 ml) の混合物に溶かし、 1N水 酸化ナトリウム水溶液 (210 ml) を加えて 10分間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (6.30mg, 16¾) を得た。 酢酸ェチル-ジイソプロピル エーテルから再結晶して融点 144-146°Cの暗赤色結晶を得た。 The above acetic acid 4-{[5,6-dimethyl-3-oxo-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene] sulfinnamoyloxy} benzyl ester (42.0 mg, 0.0700 benzyl) in a mixture of tetrahydrofuran (1.00 ml) and ethanol (1.00 ml) , And a 1N aqueous sodium hydroxide solution (210 ml) was added thereto, followed by stirring for 10 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the title compound and the solvent was removed reduced pressure to (6.30mg, 16¾). Recrystallization from ethyl acetate-diisopropyl ether gave dark red crystals with a melting point of 144-146 ° C.
赤外吸収スペクトル(IR) (KBr中) V 3463, 3350-2800, 1748, 1667, 1622, 1557 cm— Infrared absorption spectrum (IR) (in KBr) V 3463, 3350-2800, 1748, 1667, 1622, 1557 cm—
Ή— NMR (CDC13) δ: 1.80 (3Η, s), 1.90-2.22 (4H, m), 2.27 (3H, s) , 3.02 (2H, t, J=6.4 Hz), 4.79 (2H, s), 7.50 (2H, d, J=8.0 Hz), 7.95 (2H, d, J=8.4 Hz), 9.63 (1H, bs). Ή- NMR (CDC1 3) δ: 1.80 (3Η, s), 1.90-2.22 (4H, m), 2.27 (3H, s), 3.02 (2H, t, J = 6.4 Hz), 4.79 (2H, s) , 7.50 (2H, d, J = 8.0 Hz), 7.95 (2H, d, J = 8.4 Hz), 9.63 (1H, bs).
以下に実施例 7- 1〜7-62で合成した化合物および同様にして合成した化合物の 一覧を示す。 The following is a list of compounds synthesized in Examples 7-1 to 7-62 and compounds synthesized in the same manner.
〔表 7 2〕(Table 7 2)
差眷ぇ用弒 ( Μ2β) 〔表 7 3〕For difference (Μ2β) (Table 7 3)
差替え (規則 26) 〔表 7 4〕Substitution (Rule 26) (Table 74)
羞替え用紙(¾則 2δ》 〔表 7 5〕Shuffle paper (¾ Rule 2δ) (Table 75)
羞替え用紙 (»2β) 〔表 7 6〕Shuffle paper (»2β) (Table 76)
差替え用就 C¾1W 〔表 7 7〕Replacement C 就 1W (Table 7 7)
差替え用弒 則 26) 〔表 7 8〕Replacement rules 26) (Table 78)
差替え用銑 (MMI26) 〔表 79〕Replacement pig (MMI26) (Table 79)
麄替え用弒 m) 〔表 8 0〕(For replacement 弒 m) (Table 80)
差替え用紙(規則 26) 〔表 8 1〕Replacement form (Rule 26) (Table 8 1)
差替え用紙(規則 26) 〔表 8 23Replacement form (Rule 26) (Table 8 23
籙眷ぇ用紙(規則 2 〔表 8 3〕Customs form (Rule 2 (Table 83)
用紙(m 〔表 8 4〕Paper (m (Table 84)
差 ぇ用弒 (MM26) 〔表 8 5〕Difference ぇ (MM26) (Table 85)
替え用紙(規則 2 〔表 8 6〕Replacement paper (Rule 2 (Table 86)
差替え用紙(Si! 〔表 8. 7〕Replacement paper (Si! (Table 8.7)
差替え用弒(親則 26) 〔表 8 8〕For replacement (Chief rule 26) (Table 88)
差替え用紙(規則 2β) 〔表 8 9〕Replacement form (Rule 2β) (Table 89)
差眷ぇ用鈹(規則 26) 〔表 90〕Substitution (Rule 26) (Table 90)
眷 f勰 m 以下に実施例 7- 63〜7- 206で合成した化合物のデ一夕を示す。 F 勰 m The following shows the results of the compounds synthesized in Examples 7-63 to 7-206.
実施例 7-63 Example 7-63
4_クロロ- N - [5, 6-ジメチル- 3-ォキソ -8- [(4, 4, 4-トリフルォロブチル)スルファ 二ル]- 2, 3 -ジヒドロ- 1H -イミダゾ [5,1 - c] [1,4]チアジン - 1 -ィリデン]ベンゼンス ルホンアミド 4_Chloro-N- [5,6-dimethyl-3-oxo-8-[(4,4,4-trifluorobutyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1 -c] [1,4] thiazine-1 -ylidene] benzenesulfonamide
水素化ナトリウム(52.2mg, 1.30薩 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-8で得られた 1 -ァミノ- 5, 6-ジメチル 8-[(4, 4, 4 -トリ フルォロプチル)スルファニル] -31-トイミダゾ [5, 1-c] [1,4]チアジン - 3-オン (220 mg, 0.652 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間か き混ぜた。 これに 4-クロ口ベンゼンスルホニル クロリド (275 mg, 1.30 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (207 mg, 62%) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルエーテルから再結晶して融点 171-173°Cの暗赤色結晶を得た。 IR (KBr)リ : 3233, 3094, 2936, 1761, 1665, 1624, 1563 cm"1. To a suspension of sodium hydride (52.2 mg, 1.30 sol) (60% oil) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl 8-[(obtained in Example 6-8. 4,4,4-Trifluoroptyl) sulfanyl] -31-toimidazo [5,1-c] [1,4] thiazin-3-one (220 mg, 0.652 mmol) in tetrahydrofuran (1.00 ml) was added, and ice was added. Stir for 5 minutes under cooling. To this was added a solution of benzenesulfonyl chloride 4-chloro (275 mg, 1.30 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (207 mg, 62%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 171-173 ° C. IR (KBr): 3233, 3094, 2936, 1761, 1665, 1624, 1563 cm " 1 .
一 NMR (CDC13) : δ 1.80 (3Η, d, J=1.2 Hz), 1.97 (2H, quintet, J=7.5 Hz), 2.13-2.30 (2H, m), 2.28 (3H, d, J-0.9 Hz), 3.01 (2H, t, J=7.5 Hz), 7.47 (2H, dt, J=9.0, 2.1 Hz), 7.90 (2H, dt, J=9.0, 2.4 Hz), 9.61 (1H, bs). One NMR (CDC1 3): δ 1.80 (3Η, d, J = 1.2 Hz), 1.97 (2H, quintet, J = 7.5 Hz), 2.13-2.30 (2H, m), 2.28 (3H, d, J-0.9 Hz), 3.01 (2H, t, J = 7.5 Hz), 7.47 (2H, dt, J = 9.0, 2.1 Hz), 7.90 (2H, dt, J = 9.0, 2.4 Hz), 9.61 (1H, bs).
元素分析値: C18H17N303S3C1F3として Elementary analysis: as C 18 H 17 N 3 0 3 S 3 C1F 3
計算値 (¾) : C, 42.23; H, 3.35; N, 8.21; S, 18.79. Calculated value (¾): C, 42.23; H, 3.35; N, 8.21; S, 18.79.
実測値 (%) : C, 42.24; H, 3.20; N, 8. 6; S, 18.85. Found (%): C, 42.24; H, 3.20; N, 8.6; S, 18.85.
実施例 7 - 65 Example 7-65
4-ク口 Π-Ν-[5, 6-ジメチル- 3-ォキソ -8- [(5, 5, 5-トリフルォロペンチル)スルフ ァニル] -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-トイリデン]ベンゼン スルホンアミド 4- ク mouth Π-Ν- [5,6-dimethyl-3-oxo-8-[(5,5,5-trifluoropentyl) sulfanyl] -2,3-dihydro mouth-1H-imidazo [5 , 1-c] [1,4] thiazine-toylidene] benzenesulfonamide
水素化ナトリウム(101 mg, 2.52 mmol) (60 油性) のテトラヒドロフラン(3.00 ml) の懸濁液に、 実施例 6- 7で得られた 1 -アミノ- 5, 6 -ジメチル 8- [(5,5,5-トリ フルォロペンチル)スルファニル ]-3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (442 mg, 1.26腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4-クロ口ベンゼンスルホニル クロリド (531mg, 2.52 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(191 mg, 29%)を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 166- 168°Cの暗赤色結晶を得 た。 To a suspension of sodium hydride (101 mg, 2.52 mmol) (60 oily) in tetrahydrofuran (3.00 ml) was added 1-amino-5,6-dimethyl 8-[(5,5) obtained in Example 6-7. 5,5-Trifluoropentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (442 mg, 1.26 t ol) in tetrahydrofuran (2.00 ml) and ice-cooling Lower 5 Stir for a minute. To this was added a solution of benzenesulfonyl chloride 4-chloro (531 mg, 2.52 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (191 mg, 29%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 166-168 ° C.
IR (KBr) リ: 3248, 2944, 1759, 1665, 1622, 1561 cm"1. IR (KBr): 3248, 2944, 1759, 1665, 1622, 1561 cm " 1 .
Ή -腿 (CDC13) : δ 1.62—1.93 (4Η, m), 1.80 (3H, d, J=0.9 Hz), 2.04-2.20 (2H, m), 2.27 (3H, d, J=0.9 Hz), 2.96 (2H, t, J=7.2 Hz), 7.46 (2H, di, J=8.7, 2.4 Hz), 7.90 (2H, dt, J=8.4, 1.5 Hz), 9.59 (1H, bs). E - thigh (CDC1 3): δ 1.62-1.93 ( 4Η, m), 1.80 (3H, d, J = 0.9 Hz), 2.04-2.20 (2H, m), 2.27 (3H, d, J = 0.9 Hz) , 2.96 (2H, t, J = 7.2 Hz), 7.46 (2H, di, J = 8.7, 2.4 Hz), 7.90 (2H, dt, J = 8.4, 1.5 Hz), 9.59 (1H, bs).
元素分析値: C19H19N303S3C1F3として Elementary analysis: as C 19 H 19 N 3 0 3 S 3 C1F 3
計算値 (¾) : C, 43.38; H, 3.64; N, 7.99; S, 18.29. Calculated (¾): C, 43.38; H, 3.64; N, 7.99; S, 18.29.
実測値 (¾) : C, 43.51; H, 3.74; N, 8.18; S, 18.58. Found (¾): C, 43.51; H, 3.74; N, 8.18; S, 18.58.
実施例 7-66 Example 7-66
4 -クロ Π-Ν- [5, 6 -ジメチル- 8- [ (3 -フルォ口- 3-メチルブチル)スルファニル] -3- ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン]ベンゼン スルホンアミド 4-Chloro-Π-Ν- [5,6-dimethyl-8-[(3-fluoromethyl-3-methylbutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] benzene sulfonamide
水素化ナトリウム(5.07 mg, 0.127讓 ol) (60%油性)のテトラヒドロフラン(0.50 ml) の懸濁液に、 実施例 6-4で得られた卜ァミノ- 5,6-ジメチル 8-[(3-フルォロ -3-メチルブチル)スルファニル] -3H-イミダゾ [5, 1-c] [1,4]チアジン -3 -オン (20.0 mg, 0.0634 mmol) のテトラヒドロフラン (0.50 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。これに 4-クロ口ベンゼンスルホニル クロリド(39.4mg, 0.127 讓 ol)のテトラヒドロフラン(0.50 ml)溶液を加え、 さらに 30分間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (15.8 mg, 51%) を合成した。 酢酸ェチ ル-ジェチルエーテルから再結晶して融点 163- 165 の暗赤色結晶を得た。 To a suspension of sodium hydride (5.07 mg, 0.127 acetyl) (60% oil) in tetrahydrofuran (0.50 ml) was added the triamino-5,6-dimethyl 8-[(3) obtained in Example 6-4. -Fluoro-3-methylbutyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (20.0 mg, 0.0634 mmol) in tetrahydrofuran (0.50 ml) was added and the mixture was cooled on ice. Stir for 5 minutes. To this was added a solution of benzenesulfonyl chloride (39.4 mg, 0.127 benzyl) in tetrahydrofuran (0.50 ml), and the mixture was further stirred for 30 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (15.8 mg, 51%) was synthesized. Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals, mp 163-165.
IR (KBr) V: 3227, 2984, 2928, 1757, 1663, 1622, 1559 cm"1. IR (KBr) V: 3227, 2984, 2928, 1757, 1663, 1622, 1559 cm " 1 .
Ή -證 (CDC13) : (51.40 (6H, d, J=21.4 Hz), 1.80 (3H, s) , 1.86-2.06 (2H, m), 2.28 (3H, s), 3.06 (2H, t, J=8.6 Hz), 7.47 (2H, d, J=8.8 Hz), 7.90 (2H, d, J=8.8 Hz), 9.58 (1H, bs). E - testimony (CDC1 3): (51.40 ( 6H, d, J = 21.4 Hz), 1.80 (3H, s), 1.86-2.06 (2H, m), 2.28 (3H, s), 3.06 (2H, t, J = 8.6 Hz), 7.47 (2H, d, J = 8.8 Hz), 7.90 (2H, d, J = 8.8 Hz), 9.58 (1H, bs).
元素分析値: C19H21N303S3C1Fとして Elemental analysis: as C 19 H 21 N 3 0 3 S 3 C1F
計算値 (%) : C, 46.57; H, 4.32; N, 8.57; S, 19.63. Calculated (%): C, 46.57; H, 4.32; N, 8.57; S, 19.63.
実測値 (%) : C, 46.28; H, 4.61; N, 8.45; S, 19.28. Found (%): C, 46.28; H, 4.61; N, 8.45; S, 19.28.
実施例 7-67 Example 7-67
4 -ブ口モ- N- [5, 6-ジメチル- 8- [ (4-フルォロプチル)スルファニル] -3-ォキソ -2,3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン]ベンゼンスルホ ンアミド 4-Bumo-N- [5,6-dimethyl-8-[(4-fluorobutyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 [Thiazine-1-ylidene] benzenesulfonamide
水素化ナトリウム(60. 1.51删 ol) (60 由性)のテトラヒドロフラン(3.00 ml)の懸濁液に、実施例 6-18で得られた 1-ァミノ- 5, 6-ジメチル 8- [(4-フルォロ プチル)スルファニル ]-3H-イミダゾ [5, 1-c] [1, 4]チアジン- 3 -オン(227 mg, 0.754 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (393 mg, 1.51腿 ol) のテトラヒ ドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (120 mg, 31%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 168-170°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (60.1.51 mol) (60-free) in tetrahydrofuran (3.00 ml) was added 1-amino-5,6-dimethyl 8-[(4 -Fluorobutyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (227 mg, 0.754 mmol) in tetrahydrofuran (2.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. Was. To this was added a solution of 4-bromobenzenesulfonyl chloride (393 mg, 1.51 mol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was evaporated reduced pressure to give the title compound (120 mg, 31%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 168-170 ° C.
IR (KBr) V: 3233, 2965, 1759, 1663, 1622, 1561 cm"1. IR (KBr) V: 3233, 2965, 1759, 1663, 1622, 1561 cm " 1 .
Ή-NMR (CDC13) : 51.70-1.88 (4H, m), 1.80 (3H, d, J=0.9 Hz), 2.28 (3H, d, J=0.9 Hz), 3.01 (2H, t, J=7.2 Hz), 4.48 (2H, dt, J=47.1, 5.7 Hz), 7.63 (2H, dt, J=8.7, 1.8 Hz), 7.83 (2H, dt, J=8.7, 1.8 Hz), 9.59 (1H, bs). Ή-NMR (CDC1 3): 51.70-1.88 (4H, m), 1.80 (3H, d, J = 0.9 Hz), 2.28 (3H, d, J = 0.9 Hz), 3.01 (2H, t, J = 7.2 Hz), 4.48 (2H, dt, J = 47.1, 5.7 Hz), 7.63 (2H, dt, J = 8.7, 1.8 Hz), 7.83 (2H, dt, J = 8.7, 1.8 Hz), 9.59 (1H, bs ).
元素分析値: C18HigN303S3BrFとして Elementary analysis: as C 18 H ig N 3 0 3 S 3 BrF
計算値 (¾) : C, 41.54; H, 3.68; N, 8.07; S, 18.48. Calculated value (¾): C, 41.54; H, 3.68; N, 8.07; S, 18.48.
実測値 (¾) : C, 41.31; H, 3.67; N, 8.04; S, 18.58. Found (¾): C, 41.31; H, 3.67; N, 8.04; S, 18.58.
実施例 7-68 Example 7-68
4-ブロモ -N - [5, 6-ジメチル -3-ォキソ -8- [ (3, 3, 4, 4, 4 -ペンタフルォロブチル)ス ルファニル] - 2, 3_ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン]ベン ゼンスルホンアミド 4-bromo-N- [5,6-dimethyl-3-oxo-8-[(3,3,4,4,4-pentafluorobutyl) sulfanyl] -2,3_dihydro-1H-imidazo [5, 1-c] [1, 4] thiazine-triylidene] benzenesulfonamide
水素化ナトリウム(49.2mg, 1.23 mmol) (60%油性)のテトラヒドロフラン(2.00 ml)の懸濁液に、実施例 6- 20で得られた卜アミノ- 5, 6-ジメチル 8 - [(3, 3, 4, 4,4- ペン夕フルォロプチル)スルファニル] - 3H -ィミダゾ [5, 1-c] [1, 4]チアジン -3-ォ ン (200 mg, 0.615 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (321 mg, 1.23 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (167 mg, 46¾) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 180-182°Cの暗赤色結晶を得た。 Sodium hydride (49.2 mg, 1.23 mmol) (60% oil) in tetrahydrofuran (2.00 ml) in the suspension of triamino-5,6-dimethyl 8-[(3,3,4,4,4-pentafluorofluortyl) sulfanyl] -3H-imidazo obtained in Example 6-20. A solution of 5, 1-c] [1,4] thiazine-3-one (200 mg, 0.615 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-bromobenzenesulfonyl chloride (321 mg, 1.23 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (167 mg, 46¾) was synthesized. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 180-182 ° C.
IR (KBr) V: 3221, 3088, 2928, 1761, 1665, 1624, 1563 cm"1. IR (KBr) V: 3221, 3088, 2928, 1761, 1665, 1624, 1563 cm " 1 .
Ή—NMR (CDC13) : δ 1.81 (3Η, s), 2.28 (3H, d, J=1.0 Hz), 2.30-2.60 (2H, m), 3.16 (2H, t, J=8.8 Hz), 7.64 (2H, dt, J=8.4, 1.8 Hz), 7.81 (2H, dt, J=8.4, 2.2 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 1.81 (3Η, s), 2.28 (3H, d, J = 1.0 Hz), 2.30-2.60 (2H, m), 3.16 (2H, t, J = 8.8 Hz), 7.64 (2H, dt, J = 8.4, 1.8 Hz), 7.81 (2H, dt, J = 8.4, 2.2 Hz), 9.60 (1H, bs).
元素分析値: C18H15N303S3BrF5として Elementary analysis: as C 18 H 15 N 3 0 3 S 3 BrF 5
計算値 (¾) : C, 36.49; H, 2.55; N, 7.09; S, 16.24. Calculated (¾): C, 36.49; H, 2.55; N, 7.09; S, 16.24.
実測値 (¾) : C, 36.53; H, 2.34; N, 7.07; S, 16.17. Found (¾): C, 36.53; H, 2.34; N, 7.07; S, 16.17.
実施例 7 - 69 Example 7-69
4 -({[5, 6-ジメチル- 8- (ィソペンチルスルファニル) -3-ォキソ -3H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィル]イミノ}スルフォニル)ベンジル アセテート 実施例 7 - 63と同様にして合成した。 4-({[5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-3H-imidazo [5,1-c] [1,4] thiazine-triyl] imino} sulfonyl) benzyl Acetate Synthesized in the same manner as in Examples 7-63.
赤外吸収スぺクトル(IR) (KBr中) v: 3250, 2957, 2934, 2972, 1744, 1663, 1615, 1559 cm— Infrared absorption spectrum (IR) (in KBr) v: 3250, 2957, 2934, 2972, 1744, 1663, 1615, 1559 cm—
'H-NMR (CDC13) : δ 0.934 (6Η, d, J=6.2 Hz), 1.48-1.77 (3H, m), 1.79 (3H, d, J=1. Hz), 2.13 (3H, s), 2.27 (3H, d, J=1.2 Hz), 2.95 (3H, d, J=7.4Hz), 5.16 (2H, s), 7.46 (2H, d, J=8.4 Hz), 7.96 (2H, d, J=8.4 Hz), 9.60 (1H, bs). 実施例 7-70 'H-NMR (CDC1 3) : δ 0.934 (6Η, d, J = 6.2 Hz), 1.48-1.77 (3H, m), 1.79 (. 3H, d, J = 1 Hz), 2.13 (3H, s) , 2.27 (3H, d, J = 1.2 Hz), 2.95 (3H, d, J = 7.4 Hz), 5.16 (2H, s), 7.46 (2H, d, J = 8.4 Hz), 7.96 (2H, d, J = 8.4 Hz), 9.60 (1H, bs).
4_ブロモ 6-ジメチル- 3-ォキソ -8 - [(6, 6, 7, 7, 7 -ペン夕フルォ口へプチル) スルファニル ]-2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン - 1 -ィリデン]ベ ンゼンスルホンアミド 4_Bromo 6-dimethyl-3-oxo-8-[(6,6,7,7,7-pentylfluoroheptyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-1-ylidene] benzenesulfonamide
水素化ナトリウム(38.5 mg, 0.963匪 ol) (60%油性)のテトラヒド Πフラン(2.00 ml)の懸濁液に、実施例 6 - 21で得られた卜アミノ- 5, 6-ジメチル 8-[(6, 6, 7, 7, 7- ペン夕フルォ口へプチル)スルファニル] -3H -ィミダゾ [5, 1- c] [1 , 4]チアジン- 3 - オン (200 mg, 0.481 inraol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷 下 5 分間かき混ぜた。 これに 4 -プロモベンゼンスルホニル クロリド (251 mg, 0.963匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 2.5時間かき 混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (117 mg, 39%) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 160 - 162T:の暗赤色結晶を得 た。 Sodium hydride (38.5 mg, 0.963 marl ol) (60% oil) tetrahydrofuran (2.00% ml) of the suspension of triamino-5,6-dimethyl 8-[(6,6,7,7,7-pentafluoroheptyl) sulfanyl] -3H obtained in Example 6-21. A solution of -imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.481 inraol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice-cooling for 5 minutes. To this was added a solution of 4-bromobenzenesulfonyl chloride (251 mg, 0.963 ol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, after Drying (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (117 mg, 39%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 160-162T :.
IR (KBr) y: 3227,2949, 1759, 1665, 1622, 1559 cm—1. IR (KBr) y: 3227,2949, 1759, 1665, 1622, 1559 cm— 1 .
'H-NMR (CDC13) : (51.40-1.84 (6H, m), 1.80 (3H, s), 1.96-2.24 (2H, m), 2.27 'H-NMR (CDC1 3) : (51.40-1.84 (6H, m), 1.80 (3H, s), 1.96-2.24 (2H, m), 2.27
(3H, s), 2.30-2.60 (2H, m), 2.95 (2H, t, J=7.6 Hz), 7.63 (2H, d, J-8.8 Hz),(3H, s), 2.30-2.60 (2H, m), 2.95 (2H, t, J = 7.6 Hz), 7.63 (2H, d, J-8.8 Hz),
7.83 (2H, d, J=8.4 Hz), 9.59 (1H, bs). 7.83 (2H, d, J = 8.4 Hz), 9.59 (1H, bs).
元素分析値: C21H21N303S3BrF5として Elemental analysis: C 21 H 21 N 3 0 3 S 3 BrF 5
計算値 ) : C, 39.75; H, 3.34; N, 6.62; S, 15.16. Calculated)): C, 39.75; H, 3.34; N, 6.62; S, 15.16.
実測値 (¾) : C, 39.55; H, 3.05; N, 6.53; S, 15.14. Found (¾): C, 39.55; H, 3.05; N, 6.53; S, 15.14.
実施例 7-71 Example 7-71
4-ブロモ -N - [5, 6-ジメチル- 3-ォキソ -8 - [(6, 6, 6-トリフルォ口へキシル)スルフ ァニル ]-2, 3-ジヒドロ- 1H -イミダゾ [5, 1- c] [1, 4]チアジン-卜ィリデン]ベンゼン スルホンアミド 4-bromo-N- [5,6-dimethyl-3-oxo-8-[(6,6,6-trifluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-triylidene] benzenesulfonamide
水素化ナトリウム(43.8mg, 1.09mmol) (60%油性)のテトラヒドロフラン(2.00 ml)の懸濁液に、実施例 6 - 22で得られた卜ァミノ- 5, 6 -ジメチル 8_[(6, 6, 6 -トリ フルォ口へキシル).スルファニル ] - 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (200 mg, 0.547匪 ol) めテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (285 mg, 1.09 腿 ol)のテトラヒドロフラン(1.00ml)溶液を加え、さらに 2.5時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (160 mg, 50%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 156- 158 の暗赤色結晶を得た。 IR (KBr) v: 3239, 3071, 2949, 1759, 1665, 1622, 1559 cm一1. To a suspension of sodium hydride (43.8 mg, 1.09 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl 8 _ [(6,6 , 6-Trifluorohexyl) .sulfanyl]-3H-imidazo [5, 1-c] [1,4] thiazin-3-one (200 mg, 0.547 marl) and a solution of tetrahydrofuran (1.00 ml) The mixture was stirred for 5 minutes under ice cooling. To this was added a solution of 4-bromobenzenesulfonyl chloride (285 mg, 1.09 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (160 mg, 50%) was synthesized. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 156-158. IR (KBr) v: 3239, 3071, 2949, 1759, 1665, 1622, 1559 cm- 1 .
'H-NMR (CDC13) : δ 1.40-1.84 (6H, m), 1.80 (3H, s), 1.87-2.19 (2H, m), 2.28 (3H, d, J=1.0 Hz), 2.96 (2H, t, J=7.8 Hz), 7.63 (2H, d, J=8.8 Hz), 7.83 (2H, d, J=8.8 Hz), 9.59 (1H, bs). 'H-NMR (CDC1 3) : δ 1.40-1.84 (6H, m), 1.80 (3H, s), 1.87-2.19 (2H, m), 2.28 (3H, d, J = 1.0 Hz), 2.96 (2H , t, J = 7.8 Hz), 7.63 (2H, d, J = 8.8 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.59 (1H, bs).
元素分析値: C2。H21N303S3BrF3として Elemental analysis: C 2. H 21 N 3 0 3 S 3 As BrF 3
計算値 (¾) : C, 41.10; H, 3.62; N, 7.19; S, 16.46. Calculated (¾): C, 41.10; H, 3.62; N, 7.19; S, 16.46.
実測値 (%) : C, 40.78; H, 3.34; N, 7.16; S, 16.56. Found (%): C, 40.78; H, 3.34; N, 7.16; S, 16.56.
実施例 7-72 Example 7-72
4-プ口モ- N- [5, 6-ジメチル -8- [ (5-フルォ口ペンチル)スルファニル] -3-ォキソ -2, 3 -ジヒドロ -III-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン]ベンゼンスルホ ンアミド 4-p-mo-N- [5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-III-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene] benzenesulfonamide
例 1 Example 1
水素化ナトリウム(143 mg, 3.58mmol) (60%油性)のテトラヒドロフラン(3· 00 ml)の懸濁液に、実施例 6-23で得られた 1-ァミノ- 5, 6-ジメチル 8- [(4-フルォロ ブチル)スルファニル ]-3H -イミダゾ [5,卜 c] [1,4]チアジン -3-オン (564 mg, 1.79 mmol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 4 -プロモベンゼンスルホニル クロリド (933 mg, 3.58麗 ol) のテトラヒ ドロフラン (2.00 ml) 溶液を加え、 さらに 30分間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (820 mg, 86%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 183- 185°Cの暗赤色結晶を得た。 A suspension of sodium hydride (143 mg, 3.58 mmol) (60% oil) in tetrahydrofuran (3000 ml) was added to the suspension of 1-amino-5,6-dimethyl 8- [obtained in Example 6-23. (4-Fluorobutyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (564 mg, 1.79 mmol) in tetrahydrofuran (5.00 ml) was added, and the mixture was cooled on ice for 5 minutes. Stirred. To this was added a solution of 4-bromobenzenesulfonyl chloride (933 mg, 3.58 ml) in tetrahydrofuran (2.00 ml), and the mixture was further stirred for 30 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was evaporated reduced pressure to give the title compound (820 mg, 86%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 183-185 ° C.
例 2 Example 2
水素化ナトリウム (71.3mg, 1.78匪 ol) (60 油性) のテトラヒドロフラン (2 ml)の懸濁液に、実施例 6-23で得られた 1 -ァミノ- 5, 6-ジメチル 8_[(5_フルォロ ペンチル)スルファニル ]_3H-イミダゾ [5,卜 c] [1,4]チアジン -3-オン (281 mg, 0.891 腿 ol) のテトラヒドロフラン (2 ml) 溶液を加え、 氷冷下 5分間かき混ぜ た。 これに 4-ブロモベンゼンスルホニル クロリド (465 mg, 1.78 mmol) のテト ラヒドロフラン (2ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (260 mg, 55%) を暗赤色結晶として得た。 To a suspension of sodium hydride (71.3 mg, 1.78 marl ol) (60 oily) in tetrahydrofuran (2 ml) was added 1-amino-5,6-dimethyl 8 _ [(5_ Fluoropentyl) sulfanyl] _3H-imidazo [5, tric] [1,4] thiazin-3-one (281 mg, 0.891 tmol) in tetrahydrofuran (2 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-bromobenzenesulfonyl chloride (465 mg, 1.78 mmol) in tetrahydrofuran (2 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after reduction the solvent Evaporation under pressure gave the title compound (260 mg, 55%) as dark red crystals.
IR (KBr) V: 3218, 3092, 2963, 2936, 2867, 1759, 1665, 1622, 1559 cm"1. Ή-NMR (CDC13) : (51.44-1.84 (6H, m), 1.80 (3H, d, J-1.0 Hz), 2.28 (3H, d, J=0.8 Hz), 2.97 (2H, d, J=7.4 Hz), 4.46 (2H, dt, J=47.4, 5.8 Hz), 7.63 (2H, d, J=8.8 Hz), 7.83 (2H, d, J=8.8 Hz), 9.59 (1H, bs). IR (KBr) V:. 3218 , 3092, 2963, 2936, 2867, 1759, 1665, 1622, 1559 cm "1 Ή-NMR (CDC1 3): (51.44-1.84 (6H, m), 1.80 (3H, d , J-1.0 Hz), 2.28 (3H, d, J = 0.8 Hz), 2.97 (2H, d, J = 7.4 Hz), 4.46 (2H, dt, J = 47.4, 5.8 Hz), 7.63 (2H, d , J = 8.8 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.59 (1H, bs).
元素分析値: C19H21N303S3BrFとして Elementary analysis: as C 19 H 21 N 3 0 3 S 3 BrF
計算値 (%) : C, 42.70; H, 3.96; N, 7.86; S, 18.00. Calculated (%): C, 42.70; H, 3.96; N, 7.86; S, 18.00.
実測値 (%) : C, 42.66; H, 3.90; N, 7.86; S, 17.88. Found (%): C, 42.66; H, 3.90; N, 7.86; S, 17.88.
実施例 7 - 73 Example 7-73
N - [5, 6-ジメチル -3-ォキソ -8 [(4, 4, 5, 5, 5 -ペンタフルォロペンチル)スルファニ ル ]-2,3 -ジヒドロ- 1H-イミダゾ [5, 1 - c] [1,4]チアジン- 1 -ィリデン ]-4 -ビエルべ ンゼンスルホンアミド N- [5,6-dimethyl-3-oxo-8 [(4,4,5,5,5-pentafluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene] -4-bierbenzensulfonamide
水素化ナトリウム(62.4mg, 1.56匪01) (60%油性)のテトラヒドロフラン(3.00 ml) の懸濁液に、 実施例 6-3で得られた卜ァミノ- 5, 6-ジメチル 8 - [(4, 4, 5, 5, 5 - ペン夕フルォ口ペンチル)スルファニル] - 3H -ィミダゾ [5, 1-c] [1,4]チアジン- 3- オン (300 mg, 0.780 imol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに P-スチレンスルホニル クロリド(340 mg, 1.56腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (278 mg, 65%) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルエーテルから再結晶して融点 176- 178°Cの暗赤色結晶を得た。 IR (KBr) V: 3229, 2944, 1755, 1665, 1622, 1563 era"1. To a suspension of sodium hydride (62.4 mg, 1.56 marl 01) (60% oil) in tetrahydrofuran (3.00 ml) was added the triamino-5,6-dimethyl 8-[(4 , 4,5,5,5-Pentyl fluorene pentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (300 mg, 0.780 imol) in tetrahydrofuran (2.00 ml) ) The solution was added and stirred for 5 minutes under ice cooling. To this was added a solution of P-styrenesulfonyl chloride (340 mg, 1.56 mol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (278 mg, 65%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether yielded dark red crystals with a melting point of 176-178 ° C. IR (KBr) V: 3229, 2944, 1755, 1665, 1622, 1563 era " 1 .
Ή-NMR (CDCI3) : 51.80 (3H, d, J=0.6 Hz), 1.90-2.30 (4H, m), 2.27 (3H, s), 3.02 (2H, t, J=4.6 Hz), 5.42 (1H, d, J=7.0 Hz), 5.87 (1H, d, J=11.6 Hz), 6.74 (1H, dd, J=12.0, 7.4 Hz), 7.51 (2H, d, J=5.6 Hz), 7.91 (2H, d, J=6.0 Hz), 9.64 (1H, bs). Ή-NMR (CDCI3): 51.80 (3H, d, J = 0.6 Hz), 1.90-2.30 (4H, m), 2.27 (3H, s), 3.02 (2H, t, J = 4.6 Hz), 5.42 (1H , d, J = 7.0 Hz), 5.87 (1H, d, J = 11.6 Hz), 6.74 (1H, dd, J = 12.0, 7.4 Hz), 7.51 (2H, d, J = 5.6 Hz), 7.91 (2H , d, J = 6.0 Hz), 9.64 (1H, bs).
元素分析値: C21H20N303S3F5として Elemental analysis: as C 21 H 20 N 3 0 3 S 3 F 5
計算値 (%) : C, 45.56; H, 3.64; N, 4.59; S, 17.38. Calculated (%): C, 45.56; H, 3.64; N, 4.59; S, 17.38.
実測値 (%) : C, 45.12; H, 3.34; N, 7.37; S, 17.48. 実施例 7 - 74 Found (%): C, 45.12; H, 3.34; N, 7.37; S, 17.48. Example 7-74
N-[5, 6-ジメチル -3 -ォキソ - 8-[(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル] - 2, 3-ジヒドロ -1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン ]-4 -ビエルべ ンゼンスルホンアミド N- [5,6-Dimethyl-3-oxo- 8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene] -4-bierbenzensulfonamide
水素化ナトリウム(59.84 mg, 1.49匪 ol) (60%油性)のテトラヒドロフラン(3.00 ml) の懸濁液に、 実施例 6-6で得られた卜アミノ- 5, 6-ジメチル 8- [(5, 5, 6, 6, 6 - ペンタフルォ口へキシル)スルファニル ]-3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3 - オン (300 mg, 0.747匪 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに P-スチレンスルホニル クロリド(326mg, 1.49mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (278 mg, 65¾) を暗赤色結晶として得た。 酢 酸ェチル-ジェチルエーテルから再結晶して融点 160- 162°Cの暗赤色結晶を得た。 IR (KBr) V: 3237, 3075, 2948, 1755, 1665, 1622, 1561 cm"1. To a suspension of sodium hydride (59.84 mg, 1.49 ol) (60% oily) in tetrahydrofuran (3.00 ml) was added the triamino-5,6-dimethyl 8-[(5 , 5,6,6,6-pentahexafluorohexyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (300 mg, 0.747 bandol) in tetrahydrofuran (2.00 ml) The solution was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of P-styrenesulfonyl chloride (326 mg, 1.49 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (278 mg, 65¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 160-162 ° C. IR (KBr) V: 3237, 3075, 2948, 1755, 1665, 1622, 1561 cm " 1 .
'H-NMR (CDC13) : 1.68 - 1.84 (4H, m), 1.79 (3H, d, J=1.2 Hz), 1.92-2.20 (2H, m), 2.27 (3H, d, J-l. Hz), 2.30-2.60 (2H, m), 2.97 (2H, t, J=6.6 Hz), 5.42 (1H, d, J=11.0 Hz), 5.87 (1H, d, J=17.6Hz), 6.74 (1H, dd, J=17.6, 10.6 Hz), 7.50 (2H, d, J=8.0 Hz), 7.91 (2H, d, J=8. Hz), 9.63 (1H, bs). 'H-NMR (CDC1 3) : 1.68 - 1.84 (4H, m), 1.79 (3H, d, J = 1.2 Hz), 1.92-2.20 (2H, m), 2.27 (. 3H, d, Jl Hz), 2.30-2.60 (2H, m), 2.97 (2H, t, J = 6.6 Hz), 5.42 (1H, d, J = 11.0 Hz), 5.87 (1H, d, J = 17.6Hz), 6.74 (1H, dd , J = 17.6, 10.6 Hz), 7.50 (2H, d, J = 8.0 Hz), 7.91 (2H, d, J = 8.Hz), 9.63 (1H, bs).
元素分析値: C22H22N303S3F5として Elementary analysis: as C 22 H 22 N 3 0 3 S 3 F 5
計算値 () : C, 46.55; H, 3.91; , 7.40; S, 16.95. Calculated (): C, 46.55; H, 3.91;, 7.40; S, 16.95.
実測値 (%) : C, 46.32; H, 4.17; N, 7.43; S, 17.11. Found (%): C, 46.32; H, 4.17; N, 7.43; S, 17.11.
実施例 7-75 Example 7-75
4 -クロロメチル- N- [5, 6-ジメチル -8- (ィソぺンチルスルファニル) -3-ォキソ -2,3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン]ベンゼンスルホ ンアミド 4-Chloromethyl-N- [5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine -1-ylidene] benzenesulfonamide
酢酸ェチル-ジェチルェ一テルから再結晶して融点 185-187^の暗赤色結晶を得 た。 Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 185-187 ^.
赤外吸収スペクトル(IR) (KBr中) V : 3237, 2955, 2928, 2872, 1752, 1663, 1607, 1553 cm"1. LH-丽 R (CDClg) : δ 0.934 (6H, d, J=6.3Hz), 1.54-1.62 (4H, in), 1.69 (2H, septe, J=6.9Hz), 1.80 (3H, d, J=0.9Hz), 2.27 (3H, d, J=0.9 Hz), 2.95 (2H, t, J-7.5 Hz), 4.62 (2H, s), 7.51 (2H, d, J=8.7 Hz), 7.96 (2H, d, J=8.7 Hz), 9.61 (1H, bs). Infrared absorption spectrum (IR) (in KBr) V: 3237, 2955, 2928, 2872, 1752, 1663, 1607, 1553 cm " 1 . L H- 丽 R (CDClg): δ 0.934 (6H, d, J = 6.3Hz), 1.54-1.62 (4H, in), 1.69 (2H, septe, J = 6.9Hz), 1.80 (3H, d, J = 0.9Hz), 2.27 (3H, d, J = 0.9 Hz), 2.95 (2H, t, J-7.5 Hz), 4.62 (2H, s), 7.51 (2H, d, J = 8.7 Hz), 7.96 ( 2H, d, J = 8.7 Hz), 9.61 (1H, bs).
元素分析値: C2。H24N303S3C1として Elemental analysis: C 2. H 24 N 3 0 3 S 3 C1
計算値 (¾) : C, 49.42; H, 4.98; N, 8.64; S, 19.79. Calculated value (¾): C, 49.42; H, 4.98; N, 8.64; S, 19.79.
実測値 (¾) : C, 49.07; H, 4.91; N, 8.38; S, 19.55. Found (¾): C, 49.07; H, 4.91; N, 8.38; S, 19.55.
実施例 7-76 Example 7-76
N-{4-[({5, 6-ジメチル -3-ォキソ -8- [ (5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スル ファニル]- 2,3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン }アミ N- {4-[({5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]-2,3-dihydro-1H -Imidazo [5, tric] [1,4] thiazine-trilidene} ami
N-ベンジルへキサミド (1.12g, 5.45腿 ol)およびクロロスルホン酸 (0.665 g, 5.45匪 ol)の混合物を室温で 7時間、 70°C で 13時間攪拌した。 反応液にクロ口 スルホン酸 (0.665 g, 5.45腿 ol)を加えてさらに室温で 3日攪拌した。 反応液を 実施例 7-122と同様に処理して N- (4--クロロスルホニルベンジル) へキサミドぉ よび原料の混合物(1:4, 0.77g、 塩化物の純度 38 )を白色固形物として得た。 実 施例 6- 6で得た化合物 (230 mg, 0.573醒 ol) の THF (5.0 ml) 懸濁液に水素化 ナトリウム (純度 60%油性, 57.3mg, 1.43mmol) を室温で加え、 20分間攪拌後、 上記白色固形物(純度 38%, 533 mg, 0.573匪 ol)の THF (1 ml)溶液を加え、 さらに 50分間攪拌した。 反応液に 1N塩酸 (1.43 ml)を加え、 酢酸ェチル (30 ml)で希 釈し、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃 縮乾固後、 残渣をシリカゲル(10 g)のクロマトグラフィー (酢酸エヂルーへキサ ン、 3:7-7:3) に付した。 酢酸ェチルーへキサン(5:5-6:4)の画分を濃縮乾固後メ 夕ノールから結晶化して表題化合物(103 mg, 0.154腿 ol)を赤褐色粉末として得 た。 A mixture of N-benzylhexamide (1.12 g, 5.45 ol) and chlorosulfonic acid (0.665 g, 5.45 ol) was stirred at room temperature for 7 hours and at 70 ° C. for 13 hours. To the reaction solution was added sulfonic acid (0.665 g, 5.45 tmol) and the mixture was further stirred at room temperature for 3 days. The reaction mixture was treated in the same manner as in Example 7-122 to give a mixture of N- (4-chlorosulfonylbenzyl) hexamide and the raw materials (1: 4, 0.77 g, chloride purity 38) as a white solid. Obtained. Sodium hydride (purity 60%, 57.3 mg, 1.43 mmol) was added to a suspension of the compound obtained in Example 6-6 (230 mg, 0.573 ol) in THF (5.0 ml) at room temperature, and the mixture was added for 20 minutes. After stirring, a solution of the above white solid (purity 38%, 533 mg, 0.573 bandol) in THF (1 ml) was added, and the mixture was further stirred for 50 minutes. The reaction mixture was diluted with 1N hydrochloric acid (1.43 ml), diluted with ethyl acetate (30 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentrating to dryness, the residue was chromatographed on silica gel (10 g) (Eluoxane acetate, 3: 7-7: 3). The ethyl acetate-hexane (5: 5-6: 4) fraction was concentrated to dryness and crystallized from methanol to give the title compound (103 mg, 0.154 mol) as a red-brown powder.
Ή-NMR (300MHz, CDC13) : 50.90 (3H, t, J= 6.6 Hz), 1.32 (4H, brquint, J= 3.7 Hz), 1.68 (2H, m), 1.75 (4H, m), 1.80 (3H, q, J= 0.9 Hz), 2.06 (2H, m), 2.24 (2H, t, J= 7.8 Hz), 2.27 (3H, q, J= 0.9 Hz), 2.97 (2H, t, J= 7.2 Hz), 4.50 (2H, d, J= 6.0 Hz), 5.89 (1H, br), 7.37 (2H, d, J= 8.4 Hz), 7.90 (2H, d, J= 8.7 Hz), 9.65 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): 50.90 (3H, t, J = 6.6 Hz), 1.32 (4H, brquint, J = 3.7 Hz), 1.68 (2H, m), 1.75 (4H, m), 1.80 ( 3H, q, J = 0.9 Hz), 2.06 (2H, m), 2.24 (2H, t, J = 7.8 Hz), 2.27 (3H, q, J = 0.9 Hz), 2.97 (2H, t, J = 7.2) Hz), 4.50 (2H, d, J = 6.0 Hz), 5.89 (1H, br), 7.37 (2H, d, J = 8.4 Hz), 7.90 (2H, d, J = 8.7 Hz), 9.65 (1H, brs).
元素分析値: C27 N404S3F5として計算値: C, 48.49; H, 4.97; N, 8.38; S, 14.38; F, 14.20 (%) 、 実測値: C, 48.56; H, 5.17; N, 8.59; S, 14.27; F, 14.20 (%) . mp. 154.0-156. O Elemental analysis: C 27 N 4 0 4 S 3 F 5 Calculated: C, 48.49; H, 4.97 ; N, 8.38; S, 14.38; F, 14.20 (%), Found: C, 48.56; H, 5.17; N, 8.59; S, 14.27; F, 14.20 (%) .mp. 154.0-156. O
実施例 7-77 Example 7-77
N-(2-{4-[({5, 6-ジメチル- 3-ォキソ - 8 - [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)ス ルファ二ル]- 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン }アミ ノ)スルホニル]フエ二ル}ェチル)へキサンアミド N- (2- {4-[({5,6-dimethyl-3-oxo--8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -sulfanyl] -2,3- Dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene} amino) sulfonyl] phenyl} ethyl) hexaneamide
N_(2-フエネチル) へキサミド(5· Π g, 23.6 mmol)およびクロロスルホン酸 (4.86 ml, 70.8匪 ol)の混合物を室温で 6時間、 40で で 60時間攪拌した。 反応 液を実施例 7-122と同様に処理して N - [2- (4-ク口ロスルホニルフエニル)ェチル] へキサミド(純度 80%, 5.27 g, 13.3腿 ol)を黄色油状物として得た。 A mixture of N_ (2-phenethyl) hexamide (5.3 g, 23.6 mmol) and chlorosulfonic acid (4.86 ml, 70.8 diol) was stirred at room temperature for 6 hours and at 40 for 60 hours. The reaction solution was treated in the same manner as in Example 7-122 to give N- [2- (4-clothrosulfonylphenyl) ethyl] hexamide (purity 80%, 5.27 g, 13.3 t ol) as a yellow oil. Obtained.
¾一 NMR (200MHz, CDC13) : 50.89 (3H, t, J= 6.6 Hz), 1.28 (4H, m), 1.60 (2H, brquint, J= 7.5 Hz), 2.15 (2H, t, J= 7.5 Hz), 2.97 (2H, t, J= 6.9 Hz), 3.56 (2H, q, J= 6.6 Hz), 5.60 (1H, br), 7.45 (2H, d, J= 8.4 Hz), 7.98 (2H, q, J= 8.4 Hz). ¾ one NMR (200MHz, CDC1 3): 50.89 (3H, t, J = 6.6 Hz), 1.28 (4H, m), 1.60 (2H, brquint, J = 7.5 Hz), 2.15 (2H, t, J = 7.5 Hz), 2.97 (2H, t, J = 6.9 Hz), 3.56 (2H, q, J = 6.6 Hz), 5.60 (1H, br), 7.45 (2H, d, J = 8.4 Hz), 7.98 (2H, q, J = 8.4 Hz).
実施例 6- 6で得た化合物 (268 mg, 0.668讓 ol) の THF (5.5 ml) 懸濁液に水 素化ナトリウム(純度 60% 油性, 74.8 mg, 1.87腿 ol) を室温で加え、 20分間攪 拌後、 上記黄色油状物 (純度 80%, 265 mg, 0.668 mmol)を加え、 さらに 2時間攪 拌した。 反応液を酢酸ェチル (30ml)で希釈し、 0.1N塩酸、 水および飽和食塩水 で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣をジェチルエーテルで 希釈した。析出物を濾取し、ジェチルェ一テルで洗浄後、乾燥して表題化合物 (408 mg, 0.598 mmol)を赤色粉末として得た。 To a suspension of the compound obtained in Example 6-6 (268 mg, 0.668 alcohol) in THF (5.5 ml) was added sodium hydride (purity 60%, oily, 74.8 mg, 1.87 mol) at room temperature. After stirring for 1 minute, the above-mentioned yellow oil (purity 80%, 265 mg, 0.668 mmol) was added, and the mixture was further stirred for 2 hours. The reaction solution was diluted with ethyl acetate (30 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with getyl ether. The precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (408 mg, 0.598 mmol) as a red powder.
Ή-NMR (300MHz, CDC13) : 50.89 (3H, t, J= 6.6 Hz), 1.30 (4H, m), 1.75 (6H, m), 1.80 (3H, s), 2.06 (2H, m), 2.31 (2H, t, J= 7.5 Hz), 2.28 (3H, s), 2.89 (2H, t, J= 6.9 Hz), 2.98 (2H, t, J= 7.0 Hz), 3.52 (2H, brq, J= 6.4 Hz), 5.47 (1H, m), 7.31 (2H, d, J= 8.4 Hz), 7.90 (2H,. d, J= 8.7 Hz), 9.65 (1H, br). 元素分析値: C28H35N404S3F5として計算値: C, 49.25; H, 5.17; N, 8.21; S, 14.09; F, 13.91 (%) 、 実測値: C, 49.15; H, 5.16; N, 8.27; S, 14.08; F, 13.90 ( ) . mp. 146.0-147.5°C Ή-NMR (300MHz, CDC1 3 ): 50.89 (3H, t, J = 6.6 Hz), 1.30 (4H, m), 1.75 (6H, m), 1.80 (3H, s), 2.06 (2H, m), 2.31 (2H, t, J = 7.5 Hz), 2.28 (3H, s), 2.89 (2H, t, J = 6.9 Hz), 2.98 (2H, t, J = 7.0 Hz), 3.52 (2H, brq, J = 6.4 Hz), 5.47 (1H, m), 7.31 (2H, d, J = 8.4 Hz), 7.90 (2H, .d, J = 8.7 Hz), 9.65 (1H, br). Elemental analysis: C 28 Calculated for H 35 N 4 0 4 S 3 F 5 : C, 49.25; H, 5.17; N, 8.21; S, 14.09; F, 13.91 (%), found: C, 49.15; H, 5.16; N, 8.27; S, 14.08; F, 13.90 (). mp.146.0-147.5 ° C
実施例 7 - 78 Example 7-78
N-{5, 6 -ジメチル- 3 -ォキソ -8- [(5, 5, 6, 6, 6 -ペン N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pen
ル ]-2, 3-ジ ヒ ド ロ -1H-イ ミ ダゾ [5, 1-c] [1,4] チアジン - 1-ィ リ デ ン} [(IS, 4R) -7, 7 -ジメチル -2-ォキソビシクロ [2.2.1]ヘプト-トィル]メタンスル ホンアミド ] -2,3-Dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene} [(IS, 4R) -7,7-dimethyl 2-oxobicyclo [2.2.1] hept-tolyl] methanesulfonamide
水素化ナトリウム(49.8mg, 1.25腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-6で得られた 1-アミノ- 5, 6-ジメチル 8-[(5, 5, 6, 6, 6 - ペンタフルォ口へキシル)スルファニル] - 3H-ィミダゾ [5, 1 - c] [1,4]チアジン- 3- オン (250 mg, 0.623蘭 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 10-(+)-カンファースルホニル クロリド (312 mg, 1.25mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 45分間かき混 ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (209 mg, 55%) を暗赤色結晶 として得た。酢酸ェチル-ジェチルエーテルから再結晶して融点 14卜 143 の暗赤 色結晶を得た。 To a suspension of sodium hydride (49.8 mg, 1.25 t ol) (60% oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl 8-[(obtained in Example 6-6. 5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (250 mg, 0.623 ol) in tetrahydrofuran (2.00 ml), and the mixture was stirred under ice-cooling for 5 minutes. To this was added a solution of 10-(+)-camphorsulfonyl chloride (312 mg, 1.25 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 45 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after solvent was obtained as a distilled to give the title compound (209 mg, 55%) dark red crystals under reduced pressure. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 14-143.
IR (KBr) V 3220, 2961, 1744, 1665, 1618, 1566 cm"1. IR (KBr) V 3220, 2961, 1744, 1665, 1618, 1566 cm " 1 .
lH_NMR (CDC13) : δ 0.868 (3Η, s), 1.10 (3H, s), 1.38-1.51 (1H, m), 1.68—1.87 lH_NMR (CDC1 3): δ 0.868 (3Η, s), 1.10 (3H, s), 1.38-1.51 (1H, m), 1.68-1.87
(4H, m), 1.81 (3H, s), 1.93 (2H, d, J=18.0 Hz), 1.99—2.18 (4H, m), 2.29 (3H, s), 2.33-2.45 (1H, m), '2.46-2.59 (1H, m), 3.00 (2H, t, J=7.2 Hz), 3.08 (1H, d, J=15.0 Hz), 3.61 (1H, d, J=14.7 Hz), 9.80 (1H, bs). (4H, m), 1.81 (3H, s), 1.93 (2H, d, J = 18.0 Hz), 1.99-2.18 (4H, m), 2.29 (3H, s), 2.33-2.45 (1H, m), '2.46-2.59 (1H, m), 3.00 (2H, t, J = 7.2 Hz), 3.08 (1H, d, J = 15.0 Hz), 3.61 (1H, d, J = 14.7 Hz), 9.80 (1H, bs).
元素分析値: C24H3QN304S3F5として Elementary analysis: as C 24 H 3Q N 3 0 4 S 3 F 5
計算値 (%) : C, 46.82; H, 4.91; N, 6.82; S, 15.62. Calculated value (%): C, 46.82; H, 4.91; N, 6.82; S, 15.62.
実測値 (¾) : C, 46.56; H, 4.85; N, 6.87; S, 15.72. Found (¾): C, 46.56; H, 4.85; N, 6.87; S, 15.72.
実施例 7-79 Example 7-79
N-{5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル ]-2,3 -ジ ヒ ド ロ -1H-イ ミ ダゾ [5, 1-c] [1,4] チアジン -1-ィ リ デ ン} [ (1 R, 4S) - 7, 7 -ジメチル- 2 -ォキソビシクロ [2 · 2.1]ヘプト- 1 -ィル]メタンスル ホンアミド 水素化ナトリウム(49.8mg, 1.25mniol) (60油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-6で得られた卜ァミノ- 5, 6 -ジメチル 8-[(5, 5,6, 6, 6- ペンタフルォ口へキシル)スルファニル ]-3H-イミダゾ [5,1- c] [1,4]チアジン - 3 - オン (250 mg, 0.623 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 10- (-) -カンファースルホニル クロリド (312 mg, 1.25腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 45分間かき混 ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] Thiazine-1-ylidene} [(1R, 4S) -7,7-dimethyl-2-oxobicyclo [2 · 2.1] hept-1-yl] Methanesulfonamide To a suspension of sodium hydride (49.8 mg, 1.25 mniol) (60 oils) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl 8-[(5,5) obtained in Example 6-6. , 6,6,6-Pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (250 mg, 0.623 mmol) in tetrahydrofuran (2.00 ml) In addition, the mixture was stirred for 5 minutes under ice cooling. To this was added a solution of 10-(-)-camphorsulfonyl chloride (312 mg, 1.25 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 45 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. Wash and dry the extract
(MgS04) 後、 溶媒を減圧下に留去して題記化合物 (256 rag, 67%) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 141_143°Cの暗赤色結晶を得 た。 (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (256 rag, 67%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 141-143 ° C.
IR (KBr) V: 3216, 2959, 1744, 1665, 1618, 1566 cm"1. IR (KBr) V: 3216, 2959, 1744, 1665, 1618, 1566 cm " 1 .
[H-NMR (CDC13) : δ 0.867 (3H, s), 1.10 (3H, s), 1.38-1.48 (1H, m), 1.70-1.86 (4H, m), 1.81 (3H, d, J=1.2 Hz), 1.93 (2H, d, J=18.6 Hz), 1.98-2.17 (4H, m), 2.29 (3H, d, J=1.2 Hz), 2.33-2.44 (1H, m), 2.46-2.58 (1H, m), 3.00 (2H, t, J=6.9 Hz), 3.08 (2H, t, J=15.0Hz), 3.61 (1H, d, J=14.7 Hz), 9.80 (1H, bs). 元素分析値: C24H3。N304S3F5として [H-NMR (CDC1 3) : δ 0.867 (3H, s), 1.10 (3H, s), 1.38-1.48 (1H, m), 1.70-1.86 (4H, m), 1.81 (3H, d, J = 1.2 Hz), 1.93 (2H, d, J = 18.6 Hz), 1.98-2.17 (4H, m), 2.29 (3H, d, J = 1.2 Hz), 2.33-2.44 (1H, m), 2.46-2.58 ( 1H, m), 3.00 (2H, t, J = 6.9 Hz), 3.08 (2H, t, J = 15.0Hz), 3.61 (1H, d, J = 14.7 Hz), 9.80 (1H, bs). value: C 24 H 3. N 3 0 4 S 3 F 5
計算値 (¾) : C, 46.82; H, 4.91; N, 6.82; S, 15.62. Calculated (¾): C, 46.82; H, 4.91; N, 6.82; S, 15.62.
実測値 (%) : C, 46.72; H, 5.01; N, 6.88; S, 15.46. Found (%): C, 46.72; H, 5.01; N, 6.88; S, 15.46.
実施例 7-80 Example 7-80
3- ( { [5, 6-ジメチル- 8- (ィソペンチルスルファニル) -3-ォキソ- 2, 3-ジヒドロ- 1H - ィミダゾ [5, 1-c] [1, 4]チアジン -1-イリデン]アミノ }スルホニル)安息香酸 3-({[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene ] Amino} sulfonyl) benzoic acid
水素化ナトリウム(134 mg, 3.36画01) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 2で得られた 1 -ァミノ- 5,6-ジメチル -8 - (イソペンチ ルスルファ二ル)- 3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (500 mg, 1.68匪 ol) のテトラヒドロフラン (6.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに (3 -クロロスルホニル)安息香酸 (742 mg, 3.36 mmol)のテトラヒドロフラン(2· 00 ml) 溶液を加え、 さらに 28時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸 ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題 記化合物 (243 mg, 30%) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶 して融点 182- 184°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (134 mg, 3.36 fraction 01) (60% oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8- (obtained in Example 6-2. Isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (500 mg, 1.68 bandol) in tetrahydrofuran (6.00 ml) was added and stirred under ice cooling for 5 minutes. . To this was added a solution of (3-chlorosulfonyl) benzoic acid (742 mg, 3.36 mmol) in tetrahydrofuran (2.00 ml), and the mixture was further stirred for 28 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was evaporated under reduced pressure to synthesize the title compound (243 mg, 30%). Recrystallized from ethyl acetate-getyl ether This gave dark red crystals with a melting point of 182-184 ° C.
IR (KBr) ソ: 3400-2800, 2959, 2932, 2872, 1740, 1661, 1622, 1593, 1541 cm"1. Ή-NMR (CDC13) : 50.997 (6H, d, J=6.6Hz), 1.68-1.79 (2H, m), 1.79-1.88 (1H, m), 1.86 (3H, d, J=0.9 Hz), 2.38 (3H, d, J=1.2 Hz), 3.12 (2H, t, J=7.8 Hz), 7.71 (1H, t, J=7.5 Hz), 8.16 (1H, dt, J=9.0, 0.9 Hz), 8.56 (1H, dt, J=7.5, 0.8 Hz), 9.20 (1H, t, J=1.8 Hz), 11.03 (1H, bs). IR (KBr) Seo:. 3400-2800, 2959, 2932, 2872, 1740, 1661, 1622, 1593, 1541 cm "1 Ή-NMR (CDC1 3): 50.997 (6H, d, J = 6.6Hz), 1.68 -1.79 (2H, m), 1.79-1.88 (1H, m), 1.86 (3H, d, J = 0.9 Hz), 2.38 (3H, d, J = 1.2 Hz), 3.12 (2H, t, J = 7.8 Hz), 7.71 (1H, t, J = 7.5 Hz), 8.16 (1H, dt, J = 9.0, 0.9 Hz), 8.56 (1H, dt, J = 7.5, 0.8 Hz), 9.20 (1H, t, J = 1.8 Hz), 11.03 (1H, bs).
実施例 7-81 Example 7-81
4- ({[5, 6 -ジメチル -8_ (イソペンチルスルファニル) -3-ォキソ -2, 3 -ジヒドロ- 1H- イミダゾ [5, 1 - c] [1,4]チアジン - 1 -ィリデン]アミノ}スルホ二ル)- N-へキシルベ ンズアミド 4-({[5,6-Dimethyl-8_ (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] amino } Sulfonyl) -N-hexylbenzamide
実施例 7-4で得られた 4-({[5,6 -ジメチル -8- (イソペンチルスルファニル) - 3- ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン]アミノ}ス ルホニル)安息香酸 (150 mg, 0.311腿 ol) と卜へキシルァミン (0.0454ml, 0.343 mmol) の N,N-ジメチルホルムアミド (6.00 ml) 溶液に、 シァノりん酸ジェチル (0.55 il, 0.343 mmol) 続いてトリェチルァミン (0.0478 ml, 0.343腿 ol) を 加えた。反応混合物を室温で 19.5時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥 (MgS04)後、溶媒を減圧下に留去して題記化合物(20.2 mg, U%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 182-183°Cの暗赤色結晶を得た。 4-({[5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo obtained in Example 7-4 [5,1-c] [1 , 4] Thiazine-1-ylidene] amino} sulfonyl) benzoic acid (150 mg, 0.311 tmol) and trihexylamine (0.0454 ml, 0.343 mmol) in N, N-dimethylformamide (6.00 ml) Getyl phosphate (0.55 il, 0.343 mmol) was added, followed by triethylamine (0.0478 ml, 0.343 t ol). After stirring the reaction mixture at room temperature for 19.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (20.2 mg, U%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 182-183 ° C.
IR (KBr) リ: 3345, 3247, 2957, 2934, 2870, 1740, 1663, 1617, 1597, 1557 cm"1. ]H-NMR (CDCI3) : (50.849 (3H, t, J=7.2 Hz), 1.00 (6H, d, J=6.3Hz), 1.16-1.32 (6H, m), 1.40-1.53 (2H, m), 1.64-1.74 (2H, in), 1.74-1.83 (1H, 1), 1.86 (3H, s), 2.37 (3H, s), 2.97 (2H, q. J=6.6 Hz), 3.09 (2H, t, J=7.5 Hz), 4.33 (1H, bs), 7.92 (2H, d, J=8.7 Hz), 8.45 (2H, d, J=8.4 Hz), 11.11 (1H, bs). 実施例 7 - 82 IR (KBr): 3345, 3247, 2957, 2934, 2870, 1740, 1663, 1617, 1597, 1557 cm " 1. ] H-NMR (CDCI3): (50.849 (3H, t, J = 7.2 Hz), 1.00 (6H, d, J = 6.3Hz), 1.16-1.32 (6H, m), 1.40-1.53 (2H, m), 1.64-1.74 (2H, in), 1.74-1.83 (1H, 1), 1.86 ( 3H, s), 2.37 (3H, s), 2.97 (2H, q. J = 6.6 Hz), 3.09 (2H, t, J = 7.5 Hz), 4.33 (1H, bs), 7.92 (2H, d, J = 8.7 Hz), 8.45 (2H, d, J = 8.4 Hz), 11.11 (1H, bs).
N-[5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン - 1-ィリデン] - 4-フルォロ ベンゼンスルホンアミド N- [5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric ] [1,4] thiazine-1-ylidene]-4-fluorobenzenesulfonamide
水素化ナトリウム(39.9 mg, 0.996 mmol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-6で得られた卜ァミノ- 5, 6-ジメチル 8- [(5, 5, 6, 6, 6- ペン夕フルォ口へキシル)スルファニル] - 3H-ィミダゾ [5, 1-c] [1 , 4]チアジン- 3 - オン (200 mg, 0.498 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 4-フルォロベンゼンスルホニル クロリド (194 mg, 0.996腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 2時間かき混 ぜた。反応混合物に 塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥Sodium hydride (39.9 mg, 0.996 mmol) (60% oily) in tetrahydrofuran (1.00 ml) of the suspension of triamino-5,6-dimethyl 8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H obtained in Example 6-6. A solution of -imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 mmol) in tetrahydrofuran (2.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-fluorobenzenesulfonyl chloride (194 mg, 0.996 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2 hours. Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. Wash and dry the extract
,(MgS04) 後、 溶媒を減圧下に留去して題記化合物 (208 mg, 75%) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 177- 179 の暗赤色結晶を得 た。 It was synthesized (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (208 mg, 75%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 177-179.
IR (KBr) V: 3220, 2946, 2859, 1755, 1665, 1622, 1593, 1559, 1497 cm"1.IR (KBr) V: 3220, 2946, 2859, 1755, 1665, 1622, 1593, 1559, 1497 cm " 1 .
Ή-NMR (CDC13) : 51.66-1.84 (4H, m), 1.80 (3H, d, J=1.2 Hz), 1.96-2.16 (2H, m), 2.27 (3H, d, J=0.9 Hz), 2.98 (2H, t, J=7.2 Hz), 7.16 (2H, dd, J=9.3, 0.6 Ή-NMR (CDC1 3): 51.66-1.84 (4H, m), 1.80 (3H, d, J = 1.2 Hz), 1.96-2.16 (2H, m), 2.27 (3H, d, J = 0.9 Hz), 2.98 (2H, t, J = 7.2 Hz), 7.16 (2H, dd, J = 9.3, 0.6
Hz), 7.97 (2H, dd, J=8.7, 3.6 Hz), 9.61 (1H, bs). Hz), 7.97 (2H, dd, J = 8.7, 3.6 Hz), 9.61 (1H, bs).
元素分析値: C2。H19N303S3F6として Elemental analysis: C 2. H 19 N 3 0 3 As S 3 F 6
計算値 (¾) : C, 42.93; H, 3.42; N, 7.51; S, 17.19. Calculated value (¾): C, 42.93; H, 3.42; N, 7.51; S, 17.19.
実測値 (¾) : C, 42.95; H, 3.34; N, 7.51; S, 17.11. Found (¾): C, 42.95; H, 3.34; N, 7.51; S, 17.11.
実施例 7-83 Example 7-83
N-[2, 3 -ジヒドロ- 5, 6-ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシ ル)スルファニル] - 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン] -4-ョードベ ンゼンスルホンアミド N- [2,3-dihydro-5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -1H-imidazo [5, 1-c] [1, 4] thiazine-triylidene] -4-odobenzenesulfonamide
水素化ナトリウム(i99mg, 4.98歷01) (60%油性)のテトラヒドロフラン(2.50 ml) の懸濁液に、 実施例 6-6で得られた 1 -ァミノ- 5,6-ジメチル 8 - [(5, 5, 6, 6,6 - ペンタフルォ口へキシル)スルファニル] - 3H -ィミダゾ [5, 1-c] [1,4]チアジン- 3- オン (1.00 g, 2.49醒 ol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに 4-ョ一ドベンゼンスルホ ル クロリド(1.51 g, 4.98 腿 ol)のテトラヒドロフラン (2.50ml)溶液を加え、 さらに 19時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物題記化合物 (1.59 g, 96%) を合成した。 酢酸エヂル-ジェチルエーテルから再結晶して融点 Π1- 173 の暗赤色結晶を得 た。 To a suspension of sodium hydride (i99 mg, 4.98 歷 01) (60% oily) in tetrahydrofuran (2.50 ml) was added 1-amino-5,6-dimethyl 8-[(5 , 5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (1.00 g, 2.49 awake ol) in tetrahydrofuran (5.00 ml ) The solution was added and stirred for 5 minutes under ice cooling. To this was added a solution of 4-hydrobenzenesulfonyl chloride (1.51 g, 4.98 tmol) in tetrahydrofuran (2.50 ml), and the mixture was further stirred for 19 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized and the solvent evaporated under reduced pressure to give the title compound the title compound (1.59 g, 96%). Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals with a melting point of 1-173. Was.
IR (KBr) v: 3223, 3086, 2946, 2863, 1759, 1748, 1665, 1622, 1557 cm"1. 'H-NMR (CDC13) : δ 1.66-1.84 (4H, m), 1.80 (3H, d, J=0.9 Hz), 1.96-2.16 (2H, m), 2.28 (3H, d, J=0.6 Hz), 2.98 (2H, t, J=7.2 Hz), 7.67 (2H, d, J=8.7 Hz), 7.85 (2H, dd, J=8.7 Hz), 9.61 (1H, bs). IR (KBr) v:. 3223 , 3086, 2946, 2863, 1759, 1748, 1665, 1622, 1557 cm "1 'H-NMR (CDC1 3): δ 1.66-1.84 (4H, m), 1.80 (3H, d, J = 0.9 Hz), 1.96-2.16 (2H, m), 2.28 (3H, d, J = 0.6 Hz), 2.98 (2H, t, J = 7.2 Hz), 7.67 (2H, d, J = 8.7 Hz), 7.85 (2H, dd, J = 8.7 Hz), 9.61 (1H, bs).
元素分析値: C2。H19N303S3F5Iとして Elemental analysis: C 2. H 19 N 3 0 3 S 3 F 5 I
計算値 (¾) : C, 35.99; H, 2.87; N, 6.30; S, 14.41. Calculated (¾): C, 35.99; H, 2.87; N, 6.30; S, 14.41.
実測値 (¾) : C, 36.08; H, 2.87; N, 6.39; S, 14.57. Found (値): C, 36.08; H, 2.87; N, 6.39; S, 14.57.
実施例 7-84 Example 7-84
4-シァノ -N- [- 5, 6-ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル) スルファニル ]-2, 3-ジヒド口 1H -ィミダゾ [5, 1-c] [1, ]チアジン-卜ィリデン]ベ ンゼンスルホンアミド 4-cyano-N-[-5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydric 1H-imidazo [ 5, 1-c] [1,] thiazine-triylidene] benzenesulfonamide
水素化ナトリウム (341 mg, 8.5 mmol) (60%油性)のテトラヒドロフラン(5.00 ml) の懸濁液に、 実施例 6-6で得られた卜アミノ- 5, 6 -ジメチル 8 - [(5, 5, 6,6,6- ペンタフルォ口へキシル)スルファニル] - 3H-ィミダゾ [5, 1 - c] [1,4]チアジン- 3 - オン (1.71 g, 4.26腿 ol) のテトラヒドロフラン (10.0 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに 4-シァノベンゼンスルホニル クロリド(1.72 g, 8.52 mmol)のテトラヒドロフラン(5.00ml)溶液を加え、 さらに 15分間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物題記化合物 (1.96 g, 81¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 169-171°Cの暗赤色結晶を得 た。 To a suspension of sodium hydride (341 mg, 8.5 mmol) (60% oily) in tetrahydrofuran (5.00 ml) was added triamino-5,6-dimethyl 8-[(5,5) obtained in Example 6-6. 5,6,6,6-Pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (1.71 g, 4.26 t) of tetrahydrofuran (10.0 ml) The solution was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-cyanobenzenesulfonyl chloride (1.72 g, 8.52 mmol) in tetrahydrofuran (5.00 ml), and the mixture was further stirred for 15 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized and the solvent evaporated under reduced pressure to give the title compound the title compound (1.96 g, 81¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 169-171 ° C.
IR (KBr) V : 3229, 2944, 2875, 2234, 1744, 1663, 1607, 1553 cm"1. IR (KBr) V: 3229, 2944, 2875, 2234, 1744, 1663, 1607, 1553 cm " 1 .
Ή-NM (CDC13) : δ 1.68-1.86 (4Η, m), 1.81 (3H, d, J=0.9 Hz), 1.98-2.15 (2H, 1) , 2.29 (3H, d, J=0.9 Hz), 3.00 (2H, t, J=7.2 Hz), 7.79 (2H, d, J=8.7 Hz), 8.08 (2H, dd, J=9.0 Hz), 9.62 (1H, bs) . Ή-NM (CDC1 3): δ 1.68-1.86 (4Η, m), 1.81 (3H, d, J = 0.9 Hz), 1.98-2.15 (2H, 1), 2.29 (3H, d, J = 0.9 Hz) , 3.00 (2H, t, J = 7.2 Hz), 7.79 (2H, d, J = 8.7 Hz), 8.08 (2H, dd, J = 9.0 Hz), 9.62 (1H, bs).
元素分析値: C21H2。N403S3F5として Elemental analysis: C 21 H 2. N 4 0 3 S 3 F 5
計算値 (¾) : C, 44.44; H, 3.55; N, 9.87; S, 16.95. Calculated value (¾): C, 44.44; H, 3.55; N, 9.87; S, 16.95.
実測値 (¾) : C, 44.46; H, 3.48; N, 10.03; S, 17.08. 実施例 7 - 85 Found (¾): C, 44.46; H, 3.48; N, 10.03; S, 17.08. Example 7-85
N-{4-[({5, 6 -ジメチル -3-ォキソ -8- [(4, 4, 5, 5, 5- ファニル]- 2, 3-ジヒドロ -1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン }アミ ノ)スルホニル]フエ二ル}ァセタミド N- {4-[({5,6-dimethyl-3-oxo-8-[(4,4,5,5,5-phanyl] -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene} amino) sulfonyl] phenyl} acetamide
実施例 6-3と同様にして得た粗製の卜ァミノ- 5, 6 -ジメチル _8 - [(4, 4, 5, 5, 5- ペンタフルォロペンチル)スルファニル ]-3H-ィミダゾ [5, 1-c] [1, 4]チアジン - 3 - オン(純度 70%, 553 mg, 1.0匪 ol) の THF (10 ml) 懸濁液に水素化ナトリウム(純 度 60% 油性, 160 mg, 4.0匪 ol) を室温で加え、 20分間攪拌後、 塩ィ匕 4_ァセ夕 ミドベンゼンスルホニル (313 mg, 1.30 mmol)を加え、 さらに 30分間攪拌した。 反応液を酢酸ェチル (30ml)で希釈し、 0.2N塩酸、 0.1N塩酸、 水および飽和食塩 水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣を酢酸ェチルおよび ジェチルエーテルで希釈した。 析出物を濾取し、 酢酸ェチル-ジェチルエーテル (1:1) およびジェチルエーテルで洗浄後、 乾燥して表記化合物(270 mg, 0.462 mmol)を赤紫色粉末として得た。 Crude triamino-5,6-dimethyl-8-[(4,4,5,5,5-pentafluoropentyl) sulfanyl] -3H-imidazo [5, obtained in the same manner as in Example 6-3 1-c] [1,4] Thiazine-3-one (purity 70%, 553 mg, 1.0 bandol) in THF (10 ml) suspension was added to sodium hydride (purity 60% oil, 160 mg, 4.0 mg). Was added at room temperature and stirred for 20 minutes, followed by addition of Shii-Dai 4_asemidobenzenesulfonyl (313 mg, 1.30 mmol) and further stirring for 30 minutes. The reaction solution was diluted with ethyl acetate (30 ml), washed with 0.2N hydrochloric acid, 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with ethyl acetate and getyl ether. The precipitate was collected by filtration, washed with ethyl acetate-ethyl ether (1: 1) and dimethyl ether, and dried to give the title compound (270 mg, 0.462 mmol) as a red-purple powder.
'H-NMR (300MHz, DMS0 - d6) : 51.79 (3H, q, J= 1.0 Hz), 1.84 (2H, quint, J= 7.8 Hz), 2.08 (3H, s), 2.17 (3H, q, J= 0.6 Hz), 2.31 (2H, m), 3.07 (2H, t, J= 7.3 Hz), 7.74 (2H, dt, J= 9.2, 2.2 Hz), 7.86 (2H, d, J= 8.8 Hz), 10.30 (1H, brs), 11.65 (1H, brs). 'H-NMR (300MHz, DMS0 - d 6): 51.79 (3H, q, J = 1.0 Hz), 1.84 (2H, quint, J = 7.8 Hz), 2.08 (3H, s), 2.17 (3H, q, J = 0.6 Hz), 2.31 (2H, m), 3.07 (2H, t, J = 7.3 Hz), 7.74 (2H, dt, J = 9.2, 2.2 Hz), 7.86 (2H, d, J = 8.8 Hz) , 10.30 (1H, brs), 11.65 (1H, brs).
元素分析値: C21H21N404S3F5として計算値: C, 43.14; H, 3.62; N, 9.58; S, 16.45; F, 16.25 ( ) 、 実測値: C, 42.86; H, 3.76; N, 9.53; S, 16.32; F, 16.00 ( ) . mp. 187.0-189.0°C Elemental analysis: Calculated for C 21 H 21 N 4 0 4 S 3 F 5 : C, 43.14; H, 3.62; N, 9.58; S, 16.45; F, 16.25 (), found: C, 42.86; H , 3.76; N, 9.53; S, 16.32; F, 16.00 () .mp.187.0-189.0 ° C
実施例 7-86 Example 7-86
N-{4-[({5, 6 -ジメチル- 3-ォキソ -8- [ (5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スル ファニル]- 2,3-ジヒドロ -1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン }アミ ノ)スルホニル]フエ二ル}ァセタミド N- {4-[({5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]-2,3-dihydro-1H -Imidazo [5, tric] [1,4] thiazine-1-ylidene} amino) sulfonyl] phenyl} acetamide
実施例 6-6と同様にして得た粗製の 1 -ァミノ- 5^ -ジメチル 8— [(5, 5, 6, 6, 6 -ぺ ン夕フルォ口へキシル)スルファニル ]- 3H-ィミダゾ [5, 1-c] [1, 4]チアジン- 3 -才 ン(431 mg, 1.07匪 ol) の THF (8.6 ml) 懸濁液に水素化ナトリウム(純度 60% 油 性, 107 mg, .2.68匪 ol) を室温で加え、 10分間攪拌後、 塩化 4-ァセタミドベン ゼンスルホニル (285 mg, 1.18腿 ol)を加え、 さらに 60分間攪拌した。 反応液に 1N塩酸 (2.68 ml)を加え、 メタノール-水 (1:1, 5 ml)で希釈して 30分間攪拌し た。 析出物を濾取し、 メタノール -水 (1:1) およびメタノールで洗浄後、 乾燥し て表記化合物 (518 mg, 0.865 mmol)を赤紫色粉末として得た。 Crude 1-amino-5 ^ -dimethyl 8-[(5,5,6,6,6-d-fluorohexyl) sulfanyl] -3H-imidazo [obtained in the same manner as in Example 6-6] Sodium hydride (60% pure oily, 107 mg, 2.68) was added to a suspension of 5,1-c] [1,4] thiazine-3-amine (431 mg, 1.07 marl) in THF (8.6 ml). Ol) at room temperature and stir for 10 minutes, then add 4-acetamidoben chloride Zensulfonyl (285 mg, 1.18 t ol) was added, and the mixture was further stirred for 60 minutes. 1N Hydrochloric acid (2.68 ml) was added to the reaction solution, diluted with methanol-water (1: 1, 5 ml) and stirred for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (518 mg, 0.865 mmol) as a red-purple powder.
Ή -醒 R (300MHz, DMS0 - d6): 51.59 (2H, m), 1.70 (2H, m), 1.79 (3H, s), 2.08 (3H, s), 2.17 (3H, s), 2.22 (2H, m), 3.02 (2H, t, J= 7.2 Hz), 7.73 (2H, d, J= 8.7 Hz), 7r86 (2H, d, J= 9.0 Hz), 10.30 (1H, s), 11.62 (1H, brs). 元素分析値: C22H23N404S3F5として計算値: C, 44.14; H, 3.87; N, 9.36; S, 16.07; F, 15.87 (%) 、 実測値: C, 44.20; H, 3.83; N, 9.26; S, 16.05; F, 15.72 (%) . mp. 211.5-212.5°C E - Awakening: R (300MHz, DMS0 - d 6 ): 51.59 (2H, m), 1.70 (2H, m), 1.79 (3H, s), 2.08 (3H, s), 2.17 (3H, s), 2.22 ( 2H, m), 3.02 (2H, t, J = 7.2 Hz), 7.73 (2H, d, J = 8.7 Hz), 7 r 86 (2H, d, J = 9.0 Hz), 10.30 (1H, s), . 11.62 (1H, brs) elemental analysis: C 22 H 23 N 4 0 4 S 3 F 5 calculated: C, 44.14; H, 3.87 ; N, 9.36; S, 16.07; F, 15.87 (%), Found: C, 44.20; H, 3.83; N, 9.26; S, 16.05; F, 15.72 (%). Mp. 211.5-212.5 ° C
実施例 7 - 87 Example 7-87
4 -ク口ロ- N-[5, 6-ジメチル- 8- [ (5-フルォ口ぺンチル)スルファニル] -3-ォキソ -2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-レイリデン]ベンゼンスルホ ンアミド 4-Cu-N- [5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-leileiden] benzenesulfonamide
水素化ナトリウム(131mg, 3.27誦01) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた 1 -ァミノ- 5, 6-ジメチル- 8- [(4-フルォ ロブチル)スルファニル ]-3H -イミダゾ [5, 1-c] [1,4]チアジン - 3-オン (515 mg, 1.63 mmol) のテトラヒドロフラン (6.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 4_クロ口ベンゼンスルホニル クロリド (690 mg, 3.27匪01) のテ トラヒドロフラン (2.00ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物 に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶 媒を減圧下に留去して題記化合物 (168mg, 21%) を暗赤色結晶として得た。 酢酸 ェチル -ジェチルエーテルから再結晶して融点 180- の暗赤色結晶を得た。 IR (KBr) V 3227, 2940, 2861, 1757, 1663, 1621, 1559 cm"1. To a suspension of sodium hydride (131 mg, 3.27 recited 01) (60% oil) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8-[(obtained in Example 6-23. 4-Fluorobutyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (515 mg, 1.63 mmol) in tetrahydrofuran (6.00 ml) was added, and the mixture was cooled on ice for 5 minutes. Stirred. To this was added a solution of benzenesulfonyl chloride (690 mg, 3.27 band01) in tetrahydrofuran (2.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the title compound was evaporated Solvent under reduced pressure (168 mg, 21%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 180-. IR (KBr) V 3227, 2940, 2861, 1757, 1663, 1621, 1559 cm " 1 .
Ή-NMR (CDC13) : (51.52-1.62 (2H, m), 1.64-1.83 (4H, m), 1.79 (3H, d, J=0.9 Hz), 2.27 (3H, d, J=0.6 Hz), 2.97 (2H, i, J=7.5 Hz), 4.46 (2H, dt, J=47.1, 5.7), 7.46 (2H, d, J=9.0 Hz), 7.90 (2H, d, J=8.7 Hz), 9.59 (1H, bs) . 元素分析値: C19H21N303S3C1Fとして Ή-NMR (CDC1 3): (51.52-1.62 (2H, m), 1.64-1.83 (4H, m), 1.79 (3H, d, J = 0.9 Hz), 2.27 (3H, d, J = 0.6 Hz) , 2.97 (2H, i, J = 7.5 Hz), 4.46 (2H, dt, J = 47.1, 5.7), 7.46 (2H, d, J = 9.0 Hz), 7.90 (2H, d, J = 8.7 Hz), 9.59 (1H, bs). Elemental analysis: C 19 H 21 N 3 0 3 S 3 C1F
計算値 (%) : C, 46.57; H, 4.32; N, 8.57; S, 19.63. 実測値 (%) : C, 46.69; H, 4.41; N, 8.45; S, 19.40. Calculated value (%): C, 46.57; H, 4.32; N, 8.57; S, 19.63. Found (%): C, 46.69; H, 4.41; N, 8.45; S, 19.40.
実施例 7 - 88 Example 7-88
メチル 2 -({[5, 6 -ジメチル- 8 -(ィソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5,1-c] [1,4]チアジン- 1-ィリデン]アミノ}スルホニル)ベンゾ ェ一卜 Methyl 2-({[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene] amino} sulfonyl) benzoate
水素化ナトリウム (134 mg, 3.36mmol) (60 油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-2で得られた 1 -ァミノ- 5, 6-ジメチル -8- (イソペンチ ルスルファ二ル)- 3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (500 mg, 1.68mmol) のテトラヒドロフラン (6.00ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに メチル (4-クロロスルホニル)ベンゾェ一ト (789 mg, 3.36腿01) のテトラヒド 口フラン (2.00ml) 溶液を加え、 さらに 24.5時間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (U8mg, 14%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 147- U9°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (134 mg, 3.36 mmol) (60 oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8- (isopentylsulfuric acid) obtained in Example 6-2. A solution of nil) -3H-imidazo [5, tric] [1,4] thiazin-3-one (500 mg, 1.68 mmol) in tetrahydrofuran (6.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of methyl (4-chlorosulfonyl) benzoate (789 mg, 3.36 thigh 01) in tetrahydrofuran (2.00 ml), and the mixture was further stirred for 24.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the solvent was evaporated reduced pressure the title compound (U8mg, 14%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 147-U9 ° C.
IR (KBr) v: 3260, 2955, 2932, 2870, 1732, 1663, 1615, 1557 cr1. IR (KBr) v: 3260, 2955, 2932, 2870, 1732, 1663, 1615, 1557 cr 1 .
¾-NMR (CDC13) : δθ.909 (6Η, d, J=6.6 Hz), 1.49-1.58 (2H, m), 1.60-1.74 (1H, m), 1.79 (3H, d, J=0.9 Hz), 2.29 (3H, d, J-0.9 Hz), 2.93 (2H, t, J=7.5 Hz), 3.95 (3H, s), 7.57-7.62 (3H, m), 8.14-8.20 (1H, m), 9.86 (1H, bs). ¾-NMR (CDC1 3): δθ.909 (6Η, d, J = 6.6 Hz), 1.49-1.58 (2H, m), 1.60-1.74 (1H, m), 1.79 (3H, d, J = 0.9 Hz ), 2.29 (3H, d, J-0.9 Hz), 2.93 (2H, t, J = 7.5 Hz), 3.95 (3H, s), 7.57-7.62 (3H, m), 8.14-8.20 (1H, m) , 9.86 (1H, bs).
元素分析値: C21H25N305S3として Elemental analysis: as C 21 H 25 N 3 0 5 S 3
計算値 (%) : C, 50.89; H, 5.08; N, 8.48. Calculated value (%): C, 50.89; H, 5.08; N, 8.48.
実測値 (%) : C, 50.68; H, 5.09; N, 8.12. Found (%): C, 50.68; H, 5.09; N, 8.12.
実施例 7-89 Example 7-89
2- ( { [5, 6 -ジメチル- 8- (ィソぺンチルスルファ二ノレ) -3-ォキソ- 2, 3-ジヒドロ _ 1 H - イミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン]アミノ}スルホニル安息香酸 2-({[5,6-Dimethyl-8- (isopentylsulfinole) -3-oxo-2,3-dihydro_1H-imidazo [5,1-c] [1,4] thiazine- Trilidene] amino} sulfonylbenzoic acid
実施例 7-88で得られたメチル 2- ( { [5, 6-ジメチル -8- (ィソペンチルスルファ二 ル)- 3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン]ァ ミノ }スルホ二ル)ベンゾェ一ト (89.4mg, 0.180 mmol) のエタノール (1.00 ml) 及びテトラヒドロフラン (1.00ml) 溶液に、 1N水酸化ナトリウム水溶液 (0.541 ml) を加えた。 反応混合物を室温で 3時間かき混ぜた後、 溶媒を減圧下に留去し た。 残渣に IN塩酸を加えて、 生成した沈澱物を濾取し、 水洗後、乾燥して題記化 合物 (70.5mg, 81¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルか ら再結晶して融点 162- 164°Cの暗赤色結晶を得た。 The methyl 2-({[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1 -c] [1,4] Thiazine-1-ylidene] amino} sulfonyl) benzoate (89.4 mg, 0.180 mmol) in ethanol (1.00 ml) and tetrahydrofuran (1.00 ml) was added to a 1N sodium hydroxide solution. An aqueous solution (0.541 ml) was added. After stirring the reaction mixture at room temperature for 3 hours, the solvent was distilled off under reduced pressure. Was. IN hydrochloric acid was added to the residue, and the resulting precipitate was collected by filtration, washed with water, and dried to give the title compound (70.5 mg, 81¾) as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 162-164 ° C.
IR (KBr) V: 3700-2700, 2957, 2930, 2870, 1732, 1661, 1615, 1557 cm"1. Ή-NMR (CDC13) : 60.921 (6H, d, J=6.2 Hz), 1.52-1.78 (3H, m), 1.79 (3H, d, J=l.2 Hz), 2.28 (3H, d, J=1.0 Hz), 2.96 (2H, t, J=7.6 Hz), 7.61-7.70 (2H, m), 7.88-7.96 (1H, m), 8.12-8.18 (1H, m), 9.83 (1H, bs). ' IR (KBr) V:. 3700-2700 , 2957, 2930, 2870, 1732, 1661, 1615, 1557 cm "1 Ή-NMR (CDC1 3): 60.921 (6H, d, J = 6.2 Hz), 1.52-1.78 (3H, m), 1.79 (3H, d, J = l.2 Hz), 2.28 (3H, d, J = 1.0 Hz), 2.96 (2H, t, J = 7.6 Hz), 7.61-7.70 (2H, m), 7.88-7.96 (1H, m), 8.12-8.18 (1H, m), 9.83 (1H, bs).
元素分析値: C2。H23N305S3として Elemental analysis: C 2. H 23 N 3 0 5 S 3
計算値 ) : C, 49.88; H, 4.81; N, 8.72; S, 19.97. Calculated)): C, 49.88; H, 4.81; N, 8.72; S, 19.97.
実測値 (%) : C, 49.90; H, 4.56; N, 8.51; S, 19.72. Found (%): C, 49.90; H, 4.56; N, 8.51; S, 19.72.
実施例 7-90 Example 7-90
N-{5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6 -ペンタフルォ口へキシル)スルファニ ル]- 2, 3 -ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン - 1 -ィリデン } - 3, 4-ジメチ ルベンゼンスルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1 -ylidene} -3,4-dimethylbenzenesulfonamide
水素化ナトリウム (189 mg, 4.72膽 ol) (60%油性) のテトラヒドロフラン(5· 00 ml) の懸濁液に、 実施例 6-6で得られた 1-アミノ- 5, 6 -ジメチル -8-[(5, 5, 6, 6, 6 - ペンタフルォ口へキシル)スルファニル ]-3H-ィミダゾ [5, 1-c] [1,4]チアジン- 3- オン (948 mg, 2.36 誦01) のテトラヒドロフラン (10.0 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに 3, 4-ジメチルベンゼンスルホニル クロリド(910 ig, 4.45 腿 ol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 さらに 2.5時間かき 混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (206 mg, 15%) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 155-157での暗赤色結晶を得 た。 1-amino-5,6-dimethyl-8 obtained in Example 6-6 was added to a suspension of sodium hydride (189 mg, 4.72 bunol) (60% oily) in tetrahydrofuran (5.00 ml). -[(5,5,6,6,6-pentahexafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (948 mg, 2.36 recitation 01) A solution of tetrahydrofuran (10.0 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 3,4-dimethylbenzenesulfonyl chloride (910 ig, 4.45 tmol) in tetrahydrofuran (5.00 ml), and the mixture was further stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, after Drying (MgSO 4), and the solvent was evaporated under reduced pressure to synthesize the title compound (206 mg, 15%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with melting point 155-157.
IR (KBr) リ : 3227, 2932, 1721, 1663, 1618, 1563 cm -】. IR (KBr): 3227, 2932, 1721, 1663, 1618, 1563 cm-】.
Ή-NMR (CDC13) : 51.64-1.84 (4H, Hi), 1.79 (3H, s), 1.95-2.16 (2H, m), 2.26 (3H, d, J=1.2 Hz), 2.36 (3H, s), 2.73 (3H, s), 2.92-3.04 (2H, m), 7.11 (1H, s), 7.30 (1H, t, J-7.5 Hz), 7.88 (1H, t, J=8.7 Hz), 9.65 (1H, bs). Ή-NMR (CDC1 3): 51.64-1.84 (4H, Hi), 1.79 (3H, s), 1.95-2.16 (2H, m), 2.26 (3H, d, J = 1.2 Hz), 2.36 (3H, s ), 2.73 (3H, s), 2.92-3.04 (2H, m), 7.11 (1H, s), 7.30 (1H, t, J-7.5 Hz), 7.88 (1H, t, J = 8.7 Hz), 9.65 (1H, bs).
元素分析値: C22H24N303S3F5として 計算値 (%) : C, 46.39; H, 4.25; N, 7.38; S, 16.89. Elementary analysis: as C 22 H 24 N 3 0 3 S 3 F 5 Calculated value (%): C, 46.39; H, 4.25; N, 7.38; S, 16.89.
実測値 (%) : C, 46.21; H, 4.02; N, 7.50; S, 17.09. Found (%): C, 46.21; H, 4.02; N, 7.50; S, 17.09.
実施例 7-91Example 7-91
-{5, 6-ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニ ル] _2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン -トイリデント 2, 1, 3_ベン ゾチアジアゾ一ル- 4-スルホンアミド -{5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentahexylhexyl) sulfanyl] _2,3-dihydrido-1H-imidazo [5,1 -c] [1,4] thiazine-tolydent 2,1,3_benzothiadiazol-4-sulfonamide
水素化ナトリウム (39.9 mg, 0.996匪 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6 - 6で得られた 1-ァミノ- 5, 6 -ジメチル- 8-[(5, 5, 6, 6, 6- ペンタフルォ口へキシル)スルファニル] -3H -ィミダゾ [5, 1-c] [1,4]チアジン- 3 - オン (200 mg, 0.498讓 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2, 1, 3-ベンゾチアジアゾ一ル- 4-スルホニル クロリ ド (241 mg, 0.996匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液 を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (249 mg, 84%) を合成した。酢酸ェチル -ジェチルェ一テルから再結晶して融点 170- 172°Cの暗赤 色結晶を得た。 A suspension of sodium hydride (39.9 mg, 0.996 ol) (60% oil) in tetrahydrofuran (1.00 ml) was added to a suspension of 1-amino-5,6-dimethyl-8- [obtained in Example 6-6. (5,5,6,6,6-pentahexafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 benzyl) tetrahydrofuran ( 2.00 ml) solution and stirred under ice-cooling for 5 minutes. To this was added a solution of 2,1,3-benzothiadiazol-4-sulfonyl chloride (241 mg, 0.996 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (249 mg, 84%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 170-172 ° C.
IR (KBr) リ : 3245, 2942, 2253, 1740, 1663, 1615, 1557, 1524 cm"1. IR (KBr): 3245, 2942, 2253, 1740, 1663, 1615, 1557, 1524 cm " 1 .
^-NMR (CDC13) : δ 1.48-1.76 (4Η, m), 1.80 (3H, d, J=1.0 Hz), 1.84-2.14 (2H, m), 2.33 (3H, d, J=0.8 Hz), 2.89 (2H, t, J-7.4 Hz), 7.72 (1H, dd, J=8.8, 7.0 Hz), 8.22 (1H, dd, J=8.8, 1.0 Hz), 8.37 (1H, dd, J=7.0, 1.0 Hz), 10.10 (1H, bs). ^ -NMR (CDC1 3): δ 1.48-1.76 (4Η, m), 1.80 (3H, d, J = 1.0 Hz), 1.84-2.14 (2H, m), 2.33 (3H, d, J = 0.8 Hz) , 2.89 (2H, t, J-7.4 Hz), 7.72 (1H, dd, J = 8.8, 7.0 Hz), 8.22 (1H, dd, J = 8.8, 1.0 Hz), 8.37 (1H, dd, J = 7.0 , 1.0 Hz), 10.10 (1H, bs).
元素分析値: C2。H1SN503S4F5として Elemental analysis: C 2. H 1S N 5 0 3 As S 4 F 5
計算値 ) : C, 40.06; H, 3.03; N, 11.68; S, 21.39. Calculated values): C, 40.06; H, 3.03; N, 11.68; S, 21.39.
実測値 (%) : C, 40.06; H, 2.96; N, 11.52; S, 21.5.4. Found (%): C, 40.06; H, 2.96; N, 11.52; S, 21.5.4.
実施例 7-92 Example 7-92
N - {5, 6-ジメチル- 3-ォキソ -8_[(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 2, 3-ジヒドロ - 1H-イミダゾ [5,卜 c] [1, 4]チアジン - 1-ィリデン卜 2, 1,3-ベン ゾチアジアゾ一ル- 5-スルホンアミド N- {5,6-dimethyl-3-oxo-8 _ [(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylident 2,1,3-benzothiadiazol-5-sulfonamide
水素化ナトリウム(39.9 mg, 0.996 mmol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6- 6で得られた 1-ァミノ- 5, 6-ジメチル- 8- [(5, 5, 6, 6,6- ペンタフルォ口へキシル)スルファニル] - 3H -イミダゾ [5 ,卜 c] [1, 4]チアジン- 3 - オン (200 mg, 0.498讓 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2, 1, 3-ベンゾチアジアゾール- 5-スルホニル クロリ ド (234 mg, 0.996腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液 を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (254 mg, 85%) を合成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 123- 125°Cの喑赤 色結晶を得た。 Sodium hydride (39.9 mg, 0.996 mmol) (60% oily) in tetrahydrofuran (1.00 of the 1-amino-5,6-dimethyl-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H obtained in Example 6-6. A solution of -imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.498 acetyl) in tetrahydrofuran (2.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 2,1,3-benzothiadiazole-5-sulfonyl chloride (234 mg, 0.996 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was evaporated under reduced pressure to synthesize the title compound (254 mg, 85%). The crystals were recrystallized from ethyl acetate-getyl ether to give red crystals with a melting point of 123-125 ° C.
IR (KBr) V: 3250, 2944, 2260, 1738, 1663, 1615, 1557, 1518 cm"1. IR (KBr) V: 3250, 2944, 2260, 1738, 1663, 1615, 1557, 1518 cm " 1 .
Ή-NMR (CDC13) : (51.70-1.86 (4H, m), 1.80 (3H, d, J=1.2 Hz), 1.92-2.20 (2H, m), 2.29 (3H, d, J=1.0 Hz), 2.99 (2H, t, J=7.0 Hz), 8.11 (2H, d, J=1.2 Hz), Ή-NMR (CDC1 3): (51.70-1.86 (4H, m), 1.80 (3H, d, J = 1.2 Hz), 1.92-2.20 (2H, m), 2.29 (3H, d, J = 1.0 Hz) , 2.99 (2H, t, J = 7.0 Hz), 8.11 (2H, d, J = 1.2 Hz),
8.69 (1H, t, J=1.2 Hz), 9.68 (1H, bs). 8.69 (1H, t, J = 1.2 Hz), 9.68 (1H, bs).
元素分析値: C2。H18N503S4F5として Elemental analysis: C 2. H 18 N 5 0 3 S 4 F 5
実施例 7-93 Example 7-93
N-{5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル] -2, 3-ジヒドロ- 1H-イミダゾ [5,l_c] [1,4]チアジン- 1 -ィリデン }- 1_ベンゾチ ォフェン- 3-スルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine-1-ylidene} -1_benzothiophene-3-sulfonamide
水素化ナトリウム(39.9 mg, 0.996籠 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-6で得られた 1 -ァミノ- 5, 6 -ジメチル -8- [(5, 5, 6, 6, 6 - ペンタフルォ口へキシル)スルファニル] - 3H-ィミダゾ [5, 1 -c] [1, 4]チアジン- 3- オン (200 mg, 0.498醒 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに 1-ベンゾチォフェン- 3-スルホニル クロリド(239 mg, 0.996 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 2時間かき混 ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (218 mg, 73¾) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 194- 196 の暗赤色結晶を得 た。 A suspension of sodium hydride (39.9 mg, 0.996 ol) (60% oil) in tetrahydrofuran (1.00 ml) was added to a suspension of 1-amino-5,6-dimethyl-8- [obtained in Example 6-6. (5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 ol) in tetrahydrofuran ( 2.00 ml) solution and stirred under ice-cooling for 5 minutes. To this was added a solution of 1-benzothiophene-3-sulfonyl chloride (239 mg, 0.996 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (218 mg, 73¾) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals mp 194-196.
IR (KBr) V: 3220, 3102, 2946, 1748, 1728, 1663, 1615, 1559, 1495 cm一1. 'H-NMR (CDCI3) : δ 1.6卜 1.76 (4Η, m), 1.77 (3H, d, J=0.9 Hz), 1.92-2.12 (2H, m), 2.26 (3H, d, J=0.9 Hz), 2.94 (2H, t, J=6.6 Hz), 7.40-7.50 (2H, m), 7.86-7.90 (1H, m) , 8.25 (1H, s), 8.34-8.40 (1H, m) , 9.66 (1H, bs) . IR (KBr) V: 3220, 3102, 2946, 1748, 1728, 1663, 1615, 1559, 1495 cm one 1. 'H-NMR (CDCI3): δ 1.6 1.76 (4Η, m), 1.77 (3H, d, J = 0.9 Hz), 1.92-2.12 (2H, m), 2.26 (3H, d, J = 0.9 Hz) , 2.94 (2H, t, J = 6.6 Hz), 7.40-7.50 (2H, m), 7.86-7.90 (1H, m), 8.25 (1H, s), 8.34-8.40 (1H, m), 9.66 (1H , Bs).
元素分析値: C22H2。N303S4F5として Elemental analysis: C 22 H 2. N 3 0 3 S 4 F 5
計算値 (%) : C, 44.21; H, 3.37; N, 7.03; S, 21.46. Calculated value (%): C, 44.21; H, 3.37; N, 7.03; S, 21.46.
実測値 (¾) : C, 44.05; H, 3.21; , 6.91; S, 21.63. Found (¾): C, 44.05; H, 3.21;, 6.91; S, 21.63.
実施例 7-94 Example 7-94
N - {5, 6 -ジメチル -3-ォキソ - 8-[(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニ ル] -2, 3 -ジヒド口 -1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデント 3, 5 -ジメチ ル- 4-イソォキサゾ一ルスルホンアミド N- {5,6-dimethyl-3-oxo- 8-[(5,5,6,6,6-pentahexylhexyl) sulfanyl] -2,3-dihidid-1H-imidazo [5 , 1-c] [1,4] thiazine-1-ylidentate 3,5-dimethyl-4-isoxazolylsulfonamide
水素化ナトリウム(39.9 mg, 0.996顧 ol) (60%油性)のテトラヒドロフラン(1.00 ' ml) の懸濁液に、 実施例 6-6で得られた卜ァミノ- 5,6-ジメチル-8-[(5,5,6,6,6- ぺン夕フルォロへキシル)スルファニル] - 3H -ィミダゾ [5 , 1 - c] [1, 4]チアジン- 3- オン (200 mg, 0.498 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 3, 5-ジメチルイソォキサゾール -4-スルホニル ク口 リド (201 mg, 0.996腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さら に 2時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出 液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (109 mg, 39¾) を合成した。酢酸ェチル -ジェチルェ一テルから再結晶して融点 176- 178tの暗赤 色結晶を得た。 To a suspension of sodium hydride (39.9 mg, 0.996 ethanol) (60% oil) in tetrahydrofuran (1.00 ml) was added the triamino-5,6-dimethyl-8- [obtained in Example 6-6. (5,5,6,6,6-difluorofluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 mmol) in tetrahydrofuran (2.00 ml) solution was added and stirred under ice cooling for 5 minutes. To this was added a solution of 3,5-dimethylisoxazole-4-sulfonyl chloride (201 mg, 0.996 mol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (109 mg, 39¾) was synthesized. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 176-178 t.
IR (KBr) V: 3260, 2930, 1732, 1665, 1617, 1595, 1559 cm"1. IR (KBr) V: 3260, 2930, 1732, 1665, 1617, 1595, 1559 cm " 1 .
Ή-NMR (CDCI3) : 51.66-1.84 (4H, m), 1.82 (3H, d, J=1.2 Hz), 1.97-2.17 (2H, m), 2.29 (3H, d, J=1.2 Hz), 2.48 (3H, s), 2.66 (3H, s), 3.01 (2H, t, J=6.9 Hz), 9.55 (1H, bs). Ή-NMR (CDCI3): 51.66-1.84 (4H, m), 1.82 (3H, d, J = 1.2 Hz), 1.97-2.17 (2H, m), 2.29 (3H, d, J = 1.2 Hz), 2.48 (3H, s), 2.66 (3H, s), 3.01 (2H, t, J = 6.9 Hz), 9.55 (1H, bs).
元素分析値: C19H21N404S3F5として Elemental analysis: as C 19 H 21 N 4 0 4 S 3 F 5
計算値 (%) : C, 40.71; H, 3.78; N, 9.99; S, 17.16. Calculated value (%): C, 40.71; H, 3.78; N, 9.99; S, 17.16.
実測値 (%) : C, 40.63; H, 3.94; N, 10.08; S, 17.34. Found (%): C, 40.63; H, 3.94; N, 10.08; S, 17.34.
実施例 7-95 Example 7-95
N-{5, 6-ジメチル- 3 -ォキソ -8 - [(5, 5, 6, 6, 6 -ペンタフルォ口へキシル)スルファニ ル] - 2, 3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン卜 2, 3-ジヒド ロ- 1, 4-ベンゾジォキシン- 6-スルホンアミド N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfani ] -2,3-Dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene 2,3-dihydro-1,4-benzodioxin-6-sulfonamide
水素化ナトリウム(39.9 mg, 0.996匪 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、実施例 6- 6で得られた卜アミノ- 5, 6-ジメチル -8-[(5, 5, 6, 6,6 - ペンタフルォ口へキシル)スルファニル] - 3H-ィミダゾ [5, 1 -C] [1, 4]チアジン - 3_ オン (200 mg, 0.498腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2, 3-ジヒドロ -1, 4-ベンゾジォキシン - 6-スルホニル クロリド (241 mg, 0.996腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 2時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(228 mg, 77%) を合成した。酢酸ェチル -ジェチルェ一テルから再結晶して融点 162-164 の暗赤 色結晶を得た。 To a suspension of sodium hydride (39.9 mg, 0.996 ol) (60% oil) in tetrahydrofuran (1.00 ml) was added triamino-5,6-dimethyl-8-[(obtained in Example 6-6. 5,5,6,6,6-pentafluoromethylhexyl) sulfanyl] -3H-imidazo [5,1-C] [1,4] thiazine-3_one (200 mg, 0.498 tmol) of tetrahydrofuran (2.00 ml) ) The solution was added and stirred for 5 minutes under ice cooling. To this was added a solution of 2,3-dihydro-1,4-benzodioxin-6-sulfonyl chloride (241 mg, 0.996 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (228 mg, 77%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 162-164.
IR (KBr) V : 3230, 2948, 1730, 1665, 1617, 1563, 1497 cm"1. IR (KBr) V: 3230, 2948, 1730, 1665, 1617, 1563, 1497 cm " 1 .
Ή-NMR (CDC13) : δ 1.60-1.85 (4Η, m), 1.79 (3H, s), 1.90-2.22 (2H, m), 2.26 (3H, d, J=1.0 Hz), 2.97 (2H, t, J=7.0 Hz), 4.24-4.36 (4H, m), 6.93 (1H, d, J=8.4 Hz), 7.45 (1H, dd, J=8.4, 2.2' Hz), 7.48 (1H, d, J=1.8 Hz), 9.58 (1H, bs). Ή-NMR (CDC1 3): δ 1.60-1.85 (4Η, m), 1.79 (3H, s), 1.90-2.22 (2H, m), 2.26 (3H, d, J = 1.0 Hz), 2.97 (2H, t, J = 7.0 Hz), 4.24-4.36 (4H, m), 6.93 (1H, d, J = 8.4 Hz), 7.45 (1H, dd, J = 8.4, 2.2 'Hz), 7.48 (1H, d, J = 1.8 Hz), 9.58 (1H, bs).
元素分析値: C22H22N305S3F5として Elementary analysis: as C 22 H 22 N 3 0 5 S 3 F 5
計算値 (%) : C, 44.07; H, 3.70; N, 7.01; S, 16.04. Calculated value (%): C, 44.07; H, 3.70; N, 7.01; S, 16.04.
実測値 (¾) : C, 44.01; H, 3.63; N, 7.05; S, 16.12. Found (¾): C, 44.01; H, 3.63; N, 7.05; S, 16.12.
実施例 7-96 Example 7-96
N-{5, 6 -ジメチル -3 -ォキソ -8- [(5, 5, 6, 6, 6 -ペンタフルォ口へキシル)スルファニ ル]- 2, 3 -ジヒドロ -1H -イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン }- 2, 3 -ジヒド 口- 1-ベンゾフラン- 5-スルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric ] [1,4] Thiazine-1-ylidene} -2,3-Dihydrido-1-benzofuran-5-sulfonamide
水素化ナトリウム(39.9 mg, 0.996塵 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6- 6で得られた卜ァミノ- 5, 6 -ジメチル -8-[(5, 5, 6, 6, 6 - ペン夕フルォ口へキシル)スルファニル ]_3H-ィミダゾ [5, 1-c] [1,4]チアジン - 3 - オン (200 mg, 0.498腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2, 3-ジヒドロ- 1-ベンゾフラン- 5-スルホニル クロ リド (225 mg, 0.996匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さら に 2.5時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(208 mg, 72%) を合成した。酢酸ェチル -ジェチルェ一テルから再結晶して融点 164- 165°Cの暗赤 色結晶を得た。 To a suspension of sodium hydride (39.9 mg, 0.996 dol ol) (60% oily) in tetrahydrofuran (1.00 ml) was added the triamino-5,6-dimethyl-8-[(obtained in Example 6-6. 5,5,6,6,6-pentanofluorhexyl) sulfanyl] _3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 tmol) of tetrahydrofuran ( 2.00 ml) solution and stirred under ice-cooling for 5 minutes. This is followed by 2,3-dihydro-1-benzofuran-5-sulfonyl chloride A solution of lido (225 mg, 0.996 bandol) in tetrahydrofuran (1.00 ml) was added, and the mixture was further stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (208 mg, 72%). Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 164-165 ° C.
IR (KBr) V: 3225, 2948, 1732, 1661, 1615, 1559, 1487 cm"1. IR (KBr) V: 3225, 2948, 1732, 1661, 1615, 1559, 1487 cm " 1 .
Ή-NMR (CDC13) : 51.65-1.85 (4H, m), 1.79 (3H, s), 1.96-2.16 (2H, m), 2.26 (3H, s), 2.96 (2H, t, J=6.9 Hz), 3.25 (2H, t, J-8.7 Hz), 4.66 (2H, t, J=9.0 Hz), 6.82 (1H, d, J=8.4 Hz), 7.75 (1H, d, J=8.1 Hz), 7.76 (1H, s), 9.62 (1H, bs). Ή-NMR (CDC1 3): 51.65-1.85 (4H, m), 1.79 (3H, s), 1.96-2.16 (2H, m), 2.26 (3H, s), 2.96 (2H, t, J = 6.9 Hz ), 3.25 (2H, t, J-8.7 Hz), 4.66 (2H, t, J = 9.0 Hz), 6.82 (1H, d, J = 8.4 Hz), 7.75 (1H, d, J = 8.1 Hz), 7.76 (1H, s), 9.62 (1H, bs).
元素分析値: C22H22N304S3F5として Elementary analysis: as C 22 H 22 N 3 0 4 S 3 F 5
計算値 (%) : C, 45.28; H, 3.80; N, 7.20; S, 16.48. Calculated value (%): C, 45.28; H, 3.80; N, 7.20; S, 16.48.
実測値 (%) : C, 45.41; H, 3.62; N, 7.04; S, 16.68. Found (%): C, 45.41; H, 3.62; N, 7.04; S, 16.68.
実施例 7-97 Example 7-97
N-{5, 6 -ジメチル- 3 -ォキソ -8 - [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル] -2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン-卜ィリデン }ジベンゾ [b,d]フラン- 2-スルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-triylidene} dibenzo [b, d] furan-2-sulfonamide
水素化ナトリウム(53.5mg, 1.34腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、実施例 6-6で得られた卜ァミノ- 5, 6 -ジメチル- 8- [(5, 5, 6,6,6- ペンタフルォ口へキシル)スルファニル] - 3H -イミダゾ [5,卜 c] [1,4]チアジン- 3 - オン (268 mg, 0.668讓 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これにジベンゾ [b,d]フラン- 2-スルホニル クロリド(356 mg, 1.34匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 2.5時間かき 混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥(MgS04)後、溶媒を減圧下に留去して題記化合物(83.6mg, 20¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 176- 178 の暗赤色結晶を得 た。 To a suspension of sodium hydride (53.5 mg, 1.34 tmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8-[(obtained in Example 6-6. 5,5,6,6,6-Pentafluorohexyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (268 mg, 0.668 Substituted ol) in tetrahydrofuran (2.00 ml ) The solution was added and stirred for 5 minutes under ice cooling. To this was added a solution of dibenzo [b, d] furan-2-sulfonyl chloride (356 mg, 1.34 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, Drying (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (83.6mg, 20¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 176-178.
IR (KBr) V 3231, 3069, 3031, 2932, 1744, 1665, 1617, 1559 cm -1. IR (KBr) V 3231, 3069, 3031, 2932, 1744, 1665, 1617, 1559 cm- 1 .
Ή-NMR (CDC13) : (51.62-1.82 (4H, m), 1.78 (3H, d, J=0.9Hz), 1.92-2.12 (2H, m), 2.27 (3H, d, J-1.2 Hz), 2.95 (2H, t, J=6.9 Hz), 7.41 (1H, dt, J=7.8, 1.2 Hz), 7.53 (1H, dt, J=6.9, 1.2 Hz), 7.64 (2H, t, J-8.7 Hz), 8.00 (1H, d, J=7.8 Hz), 8.08 (1H, dd, J=8.7, 1.8 Hz), 8.59 (1H, d, J=1.5 Hz), 9.69 (1H, bs). 元素分析値: C26H22N304S3F5として Ή-NMR (CDC1 3): (51.62-1.82 (4H, m), 1.78 (3H, d, J = 0.9Hz), 1.92-2.12 (2H, m), 2.27 (3H, d, J-1.2 Hz), 2.95 (2H, t, J = 6.9 Hz), 7.41 (1H, dt, J = 7.8, 1.2 Hz), 7.53 (1H, dt, J = 6.9 , 1.2 Hz), 7.64 (2H, t, J-8.7 Hz), 8.00 (1H, d, J = 7.8 Hz), 8.08 (1H, dd, J = 8.7, 1.8 Hz), 8.59 (1H, d, J = 1.5 Hz), 9.69 (1H , bs) Elementary analysis:. as C 26 H 22 N 3 0 4 S 3 F 5
計算値 (¾) : C, 49.44; H, 3.51; , 6.65; S, 15.23. Calculated (¾): C, 49.44; H, 3.51;, 6.65; S, 15.23.
実測値 (¾) : C, 49.65; H, 3.50; N, 6.38; S, 15.51. Found (¾): C, 49.65; H, 3.50; N, 6.38; S, 15.51.
実施例 7-98 Example 7-98
N-{5, 6-ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル] -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン } (フエニル)メ タンスルホンアミド N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene} (phenyl) methansulfonamide
水素化ナトリウム (39.9 mg, 0.996 mmol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6 - 6で得られた 1 -ァミノ- 5,6-ジメチル-8-[(5,5,6,6,6_ ペン夕フルォ口へキシル)スルファニル] -3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 3 - オン (200 mg, 0.498讓 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これにべンジルスルホニル クロリド (190 mg, 0.996 mmol) のテトラヒドロフラン (1.00ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (187 mg, 68 ) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルェ一テルから再結晶して融点 195- 196 の暗赤色結晶を得た。 IR (KBr) v: 3250, 2919, 1740, 1665, 1618, 1561 cm1. To a suspension of sodium hydride (39.9 mg, 0.996 mmol) (60% oily) in tetrahydrofuran (1.00 ml) was added 1-amino-5,6-dimethyl-8-[(obtained in Example 6-6. 5,5,6,6,6_ pentaylfluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 acetylol) of tetrahydrofuran ( 2.00 ml) solution and stirred under ice-cooling for 5 minutes. To this was added a solution of benzylsulfonyl chloride (190 mg, 0.996 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain the solvent was distilled off under reduced pressure the title compound (187 mg, 68) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 195-196. IR (KBr) v: 3250, 2919, 1740, 1665, 1618, 1561 cm 1 .
•H-NMR (CDC13) : δ 1.70-1.94 (4Η, m), 1.80 (3H, d, J=1.0 Hz), 1.94-2.28 (2H, m), 2.21 (3H, d, J=1.2 Hz), 3.02 (2H, t, J=7.0 Hz), 4.35 (2H, s), 7.31-7.46 (5H, m), 9.11 (1H, bs). • H-NMR (CDC1 3) : δ 1.70-1.94 (4Η, m), 1.80 (3H, d, J = 1.0 Hz), 1.94-2.28 (2H, m), 2.21 (3H, d, J = 1.2 Hz ), 3.02 (2H, t, J = 7.0 Hz), 4.35 (2H, s), 7.31-7.46 (5H, m), 9.11 (1H, bs).
元素分析値: C21H22N303S3F5として Elemental analysis: as C 21 H 22 N 3 0 3 S 3 F 5
計算値 (%) : C, 45.40; H, 3.99; N, 7.56; S, 17.31. Calculated value (%): C, 45.40; H, 3.99; N, 7.56; S, 17.31.
実測値 (¾) : C, 45.76; H, 4.09; N, 7.39; S, 17.22. Found (¾): C, 45.76; H, 4.09; N, 7.39; S, 17.22.
実施例 7-99 Example 7-99
N-{5, 6-ジメチル -8- [ (5-フルォ口ペンチル)スルファニル] -3-ォキソ- 2, 3-ジヒド 口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン -1-ィリデン } (フエニル)メタンスルホンァ ミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene} (phenyl) methanesulfone Mid
水素化ナトリウム (50.7mg, 1.27mmol) (60%油性) のテトラヒドロフラン (1.00 ml) の懸濁液に、 実施例 6-23で得られた卜アミノ- 5,6-ジメチル-8-[(5-フルォ 口ペンチル)スルファニル ]-3H -イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (200 mg, 0.634mmol) のテトラヒドロフラン (2.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにべンジルスルホニル クロリド (242 mg, 1.27 mmol) のテトラヒド 口フラン (1.00 ml) 溶液を加え、 さらに 2.5時間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (191 mg, 64%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 196-198°Cの暗赤色結晶を得た。 A suspension of sodium hydride (50.7 mg, 1.27 mmol) (60% oily) in tetrahydrofuran (1.00 ml) was added to the triamino-5,6-dimethyl-8-[(5 -Fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.634 mmol) in tetrahydrofuran (2.00 ml) was added and stirred under ice-cooling for 5 minutes. mixed. To this was added a solution of benzylsulfonyl chloride (242 mg, 1.27 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was evaporated reduced pressure to give the title compound (191 mg, 64%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 196-198 ° C.
IR (KBr) リ : 3140, 3069, 2963, 2940, 2922, 1734, 1663, 1617, 1563 cm—1.IR (KBr): 3140, 3069, 2963, 2940, 2922, 1734, 1663, 1617, 1563 cm— 1 .
Ή-NMR (CDC13) : 51.50-1.92 (6H, m), 1.79 (3H, s) , 2.20 (3Η, s), 3.01 (2H, t, J=7.2 Hz), 4.35 (2H, s), 4.48 (2H, dt, J=47.4, 6.0 Hz), 7.29-7.47 (5H, m), 9.12 (1H, bs). Ή-NMR (CDC1 3): 51.50-1.92 (6H, m), 1.79 (3H, s), 2.20 (3Η, s), 3.01 (2H, t, J = 7.2 Hz), 4.35 (2H, s), 4.48 (2H, dt, J = 47.4, 6.0 Hz), 7.29-7.47 (5H, m), 9.12 (1H, bs).
元素分析値: C2。H24N303S3Fとして Elemental analysis: C 2. H 24 N 3 0 3 As S 3 F
計算値 (¾) : C, 51.15; H, 5.15; N, 8.95; S, 20.48. Calculated (¾): C, 51.15; H, 5.15; N, 8.95; S, 20.48.
実測値 (%) : C, 51.09; H, 5.00; N, 8.76; S, 20.24. Found (%): C, 51.09; H, 5.00; N, 8.76; S, 20.24.
実施例 7-100 Example 7-100
4 -フルォロ- N- {5, 6-ジメチル- 8- [ (5-フルォ口ぺンチル)スルファニル] -3-ォキソ -2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }ベンゼンスルホ ンアミド 4-Fluoro-N- {5,6-dimethyl-8-[(5-fluoroopentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] thiazine-1-ylidene} benzenesulfonamide
水素化ナトリウム(63.4mg, 1.59腿 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5,6-ジメチル -8- [(5-フルォ 口ペンチル)スルファニル ]-3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (250 mg, 0.793 mmol) のテトラヒドロフラン (2.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 4 -フルォロベンゼンスルホニル クロリド (308 mg, 1.59匪 ol) の テトラヒドロフラン (l.OOral) 溶液を加え、 さらに 1時間かき混ぜた。 反応混合 物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (226 mg, 61¾) を合成した。 酢酸ェチル-ジ ェチルエーテルから再結晶して融点 170- 2°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (63.4 mg, 1.59 t ol) (60% oily) in tetrahydrofuran (1.00 ml) was added 1-amino-5,6-dimethyl-8- [obtained in Example 6-23. (5-Fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (250 mg, 0.793 mmol) in tetrahydrofuran (2.00 ml) was added, and the mixture was cooled under ice-cooling. Stir for a minute. To this was added a solution of 4-fluorobenzenesulfonyl chloride (308 mg, 1.59 marl) in tetrahydrofuran (l.OOral), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (226 mg, 61¾) was synthesized. Ethyl acetate Recrystallization from ethyl ether gave dark red crystals with a melting point of 170-2 ° C.
IR (KBr) リ: 3223, 2963, 2940, 2863, 1755, 1663, 1622, 1591, 1563, 1495 cm"1. -匪 R (CDC13) : δ 1.44 - 1.86 (6Η, m), 1.79 (3H, s), 2.27 (3H, s), 2.97 (2H, t, J=7.6Hz), 4.45 (2H, dt, J=47.0, 5.8 Hz), 7.16 (2H, d, J=8.8Hz), 7.94-8.04 (2H, m), 9.59 (1H, bs) . IR (KBr) Re:. 3223, 2963, 2940, 2863, 1755, 1663, 1622, 1591, 1563, 1495 cm "1 - negation R (CDC1 3): δ 1.44 - 1.86 (6Η, m), 1.79 (3H , s), 2.27 (3H, s), 2.97 (2H, t, J = 7.6Hz), 4.45 (2H, dt, J = 47.0, 5.8 Hz), 7.16 (2H, d, J = 8.8Hz), 7.94 -8.04 (2H, m), 9.59 (1H, bs).
元素分析値: C19H21N303S3F2として Elemental analysis: as C 19 H 21 N 3 0 3 S 3 F 2
計算値 (¾) : C, 48.19; H, 4.47; N, 8.87; S, 20.31. Calculated value (¾): C, 48.19; H, 4.47; N, 8.87; S, 20.31.
実測値 (¾) : C, 48.13; H, 4.46; N, 8.71; S, 20.05. Found (¾): C, 48.13; H, 4.46; N, 8.71; S, 20.05.
実施例 7- 102 Example 7-102
N-{5, 6-ジメチル- 3-ォキソ- 8- [ (5, 5, 6, 6, 6 -ペンタフルォ口へキシル)スルファニ ) 1-2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1 ィリデント 2, 1, 3-ベン ゾォキサジァゾ一ル- 4-スルホンアミド N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfani) 1-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylident 2,1,3-benzoxaziazol-4-sulfonamide
水素化ナトリウム(39.9 mg, 0.996匪 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-6で得られた卜アミノ -5, 6 -ジメチル- 8_[(5, 5, 6, 6, 6- ペンタフルォ口へキシル)スルファニル] - 3H -イミダゾ [5, 1-c] [1,4]チアジン- 3 - オン (200 mg, 0.498 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 1, 1, 3_ベンゾォキサジァゾール- 4-スルホニル ク口 リド (218 mg, 0.996 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さら に 1時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出 液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (154 mg, 53%) を合成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 192_194 の暗赤 色結晶を得た。 To a suspension of sodium hydride (39.9 mg, 0.996 ol) (60% oil) in tetrahydrofuran (1.00 ml) was added the triamino-5,6-dimethyl-8 _ [(5 , 5, 6, 6, 6-Pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 mmol) in tetrahydrofuran (2.00 ml) The solution was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 1,1,3_benzoxodiazole-4-sulfonyl chloride (218 mg, 0.996 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (154 mg, 53%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 192_194.
IR (KBr) V: 3202, 2946, 1746, 1725, 1661, 1618, 1557 cm"1. IR (KBr) V: 3202, 2946, 1746, 1725, 1661, 1618, 1557 cm " 1 .
^-NMR (CDC13) : δ 1.50-1.78 (4Η, m), 1.80 (3H, d, J=0.8 Hz), 1.85-2.16 (2H, m), 2.22 (3H, d, J=1.2 Hz), 2.91 (2H, t, J=6.6 Hz), 7.55 (1H, dd, J=8.8, 6.6 Hz), 8.06 (1H, dd, J=9.2, 0.6 Hz), 8.13 (1H, dd, J=6.6, 0.6 Hz), 9.87 (1H, bs). ^ -NMR (CDC1 3): δ 1.50-1.78 (4Η, m), 1.80 (3H, d, J = 0.8 Hz), 1.85-2.16 (2H, m), 2.22 (3H, d, J = 1.2 Hz) , 2.91 (2H, t, J = 6.6 Hz), 7.55 (1H, dd, J = 8.8, 6.6 Hz), 8.06 (1H, dd, J = 9.2, 0.6 Hz), 8.13 (1H, dd, J = 6.6 , 0.6 Hz), 9.87 (1H, bs).
元素分析値: C2。H18N504S3F5として Elemental analysis: C 2. H 18 N 5 0 4 S 3 F 5
計算値 (%) : C, 41.16; H, 3.11; N, 12.00; S, 16.48. 実測値 (%) : C, 41.21; H, 3.02; N, 11.94; S, 16.67. Calculated value (%): C, 41.16; H, 3.11; N, 12.00; S, 16.48. Found (%): C, 41.21; H, 3.02; N, 11.94; S, 16.67.
実施例 7-103 Example 7-103
N' -{5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファ ニル] -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-トイリデン }- Ν,Ν-ジメ チルゥレア N '-{5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5 C] [1,4] thiazine-tolylidene}-Ν, Ν-dimethyl-rea
水素化ナトリウム(39.9 mg, 0.996 mmol) (60%油性)のテトラヒドロフラン(1, 00 ml) の懸濁液に、 実施例 6-6で得られた卜ァミノ- 5, 6 -ジメチル- 8 - [(5, 5, 6, 6, 6 - ペン夕フルォ口へキシル)スルファニル] _3H -ィミダゾ [5, 1-c] [1,4]チアジン- 3- オン (200 mg, 0.498讓 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに参考例 45の方法 Aで得られた溶液 (2.00 ml) を加 え、 さらに室温で 2時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで 抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (142 mg, 60%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルェ一テルから再 結晶して融点 147- 149°Cの喑赤色結晶を得た。 To a suspension of sodium hydride (39.9 mg, 0.996 mmol) (60% oily) in tetrahydrofuran (100 ml) was added the triamino-5,6-dimethyl-8- [obtained in Example 6-6. (5,5,6,6,6-hexylfluorohexyl) sulfanyl] _3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 alcohol) in tetrahydrofuran (2.00 ml) solution was added and stirred under ice cooling for 5 minutes. The solution (2.00 ml) obtained in Method A of Reference Example 45 was added thereto, and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (142 mg, 60%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-jetil ether to obtain red-red crystals having a melting point of 147-149 ° C.
IR (KBr) レ : 3256, 2938, 1732, 1667, 1644, 1613, 1584 cm—1. IR (KBr) level: 3256, 2938, 1732, 1667, 1644, 1613, 1584 cm— 1 .
Ή一 NMR (CDC13) : δ 1.70-1.88 (4Η, m), 1.80 (3H, d, J-0.9 Hz), 1.98-2.17 (2H, m), 2.27 (3H, d, J=0.9 Hz), 3.00 (3H, s), 3.00 (2H, t, J=7.1 Hz), 3.21 (3H, s), 10.73 (1H, bs). Ή one NMR (CDC1 3): δ 1.70-1.88 (4Η, m), 1.80 (3H, d, J-0.9 Hz), 1.98-2.17 (2H, m), 2.27 (3H, d, J = 0.9 Hz) , 3.00 (3H, s), 3.00 (2H, t, J = 7.1 Hz), 3.21 (3H, s), 10.73 (1H, bs).
元素分析値: C17H21N403S2F5として Elemental analysis: as C 17 H 21 N 4 0 3 S 2 F 5
計算値 (%) : C, 43.21; H, 4.48; N, 11.86; S, 13.57. Calculated value (%): C, 43.21; H, 4.48; N, 11.86; S, 13.57.
実測値 (%) : C, 43.05; H, 4.40; N, 11.74; S, 13.70. Found (%): C, 43.05; H, 4.40; N, 11.74; S, 13.70.
実施例 7-104 Example 7-104
N-{5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6_ペンタフルォ口へキシル)スルファニ ル] - 2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }- 4- (1H-ビラ ゾ一ル- 1-ィル)ベンゼンスルホンアミド N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6_pentafluorohexyl) sulfanyl] -2,3-dihydrido-1H-imidazo [5,1 -c] [1,4] thiazine-1-ylidene} -4- (1H-villazol-1-yl) benzenesulfonamide
卜フエ二ルピラゾ一ル (2.11 g, 14.3 mmol)およびクロロスルホン酸 (6.07 g, 50.5腿 ol)の混合物を室温で 6日間、 50°C で 4日間攪拌した。 反応液をトルェ ン(50 ml)で希釈し、 氷 (90 g)に注ぎ、 酢酸ェチル(100 ml)で抽出した。 有機層を 水、 飽和炭酸水素ナトリウム、 飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥後、 濃縮乾固して塩ィ匕 4- (卜ピラゾリル)ベンゼンスルホニルおよび 卜フエ二ルピラ ゾールの混合物(2:3 1.70 g)を白色固形物として得た。 A mixture of triphenylpyrazol (2.11 g, 14.3 mmol) and chlorosulfonic acid (6.07 g, 50.5 t) was stirred at room temperature for 6 days and at 50 ° C for 4 days. The reaction solution was diluted with toluene (50 ml), poured into ice (90 g), and extracted with ethyl acetate (100 ml). The organic layer was washed with water, saturated sodium bicarbonate, and saturated saline, and dried over sodium sulfate. The mixture was concentrated to dryness to give a mixture of Shiridani 4- (topyrazolyl) benzenesulfonyl and tofenylpyrazole (2: 3 1.70 g) as a white solid.
実施例 6 - 6で得た化合物 (209 rag, 0.521 皿 ol) の THF (4.0 ml) 懸濁液に 水素化ナトリゥム (純度 60%油性, 58.3 mg, 1.46讓 ol) を室温で加え、 15分間 攪拌後、上記塩ィ匕 4- (1-ピラゾリル)ベンゼンスルホニル (純度 53%, 217 mg, 0.472 mmol)の THF (2.0 ml)溶液を加え、さらに 60分間攪拌した。反応液に IN塩酸 (4.0 ml)および水(20 ml)を加え、 酢酸ェチル (35 ml)で抽出した。 有機層を水および 飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣をメタノ一 ルで希釈した。析出物を濾取し、 メタノールで洗浄後、乾燥して表記化合物 (22i mg, 0.364 mmol, 69.8%)を灰赤色の粉末として得た。 ' To a suspension of the compound obtained in Example 6-6 (209 rag, 0.521 dish ol) in THF (4.0 ml) was added sodium hydride (purity 60% oily, 58.3 mg, 1.46 ml) at room temperature, and the mixture was added for 15 minutes. After stirring, a solution of 4- (1-pyrazolyl) benzenesulfonyl (purity 53%, 217 mg, 0.472 mmol) in THF (2.0 ml) was added, and the mixture was further stirred for 60 minutes. IN hydrochloric acid (4.0 ml) and water (20 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (35 ml). The organic layer was washed with water and saturated saline, and then dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (22i mg, 0.364 mmol, 69.8%) as a gray-red powder. '
ΐ-NMR (200MHz, CDC13) : δ 1.75 (4Η, m), 1.80 (3H, q, J= 1.0 Hz), 2.05 (2H, m), 2.28 (3H, q, J= 0.6 Hz), 2.97 (2H, t, J= 7.0 Hz), 6.52 (1H, dd, J= 2.6, 1.8 Hz), 7.77 (1H, d, J= 1.4 Hz), 7.84 (2H, d, J= 8.8 Hz), 7.99 (1H, d, J= 2.6 Hz), 8.04 (2H, d, J= 8.8 Hz), 9.64 (1H, br). ΐ-NMR (200MHz, CDC1 3 ): δ 1.75 (4Η, m), 1.80 (3H, q, J = 1.0 Hz), 2.05 (2H, m), 2.28 (3H, q, J = 0.6 Hz), 2.97 (2H, t, J = 7.0 Hz), 6.52 (1H, dd, J = 2.6, 1.8 Hz), 7.77 (1H, d, J = 1.4 Hz), 7.84 (2H, d, J = 8.8 Hz), 7.99 (1H, d, J = 2.6 Hz), 8.04 (2H, d, J = 8.8 Hz), 9.64 (1H, br).
元素分析値: C23H22N503S3F5として計算値: C, 45.46; H, 3.65; N, 11.53; S, 15.83; F, 15.63 (%) 、 実測値: C, 45.49; H, 3.87; N, 11.65; S, 16.02; F, 15.61. mp. 182.5-186. Ot: Elemental analysis: C 23 H 22 N 5 0 3 S 3 F 5 Calculated: C, 45.46; H, 3.65 ; N, 11.53; S, 15.83; F, 15.63 (%), Found: C, 45.49; H, 3.87; N, 11.65; S, 16.02; F, 15.61.mp.182.5-186. Ot:
実施例 7-105 Example 7-105
ェチル 5- {[5,6-ジメチル- 3-ォキソ - 1-({[4- (トリフルォロメチル)フエニル]ス ルホニル}ィミノ) - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 8 -ィル]ス ルファ二ル}ペン夕ノエ一卜 Ethyl 5-{[5,6-dimethyl-3-oxo-1-({[4- (trifluoromethyl) phenyl] sulfonyl} imino) -2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-8-yl] sulfanil} pen
水素化ナトリウム(230 mg, 5.74 mmol) (60%油性)のテトラヒドロフラン(10.0 ml) の懸濁液に、 実施例 6-24で得られたェチル 5- [卜アミノ- 5, 6-ジメチル- 3 - ォキソ -3H -ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル]ペンタノエー ト (1.02 g, 2.87腿 ol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 氷冷下 To a suspension of sodium hydride (230 mg, 5.74 mmol) (60% oil) in tetrahydrofuran (10.0 ml) was added the ethyl 5- [triamino-5,6-dimethyl-3 obtained in Example 6-24. -Oxo-3H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] pentanoate (1.02 g, 2.87 tmol) in tetrahydrofuran (5.00 ml) was added, and the mixture was cooled on ice.
5分間かき混ぜた。これに 4- (トリフルォロメチル)ベンゼンスルホニル クロリド (1.40 g, 5.74匪01) のテトラヒドロフラン (5.00 ml) 溶液を加え、 さらに室 温で 1時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽 出液を水洗、 乾燥 (MgS04)後、溶媒を減圧下に留去して題記化合物(1.15g, 71¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 146- 148°Cの暗赤色結晶を得た。 Stir for 5 minutes. To this was added a solution of 4- (trifluoromethyl) benzenesulfonyl chloride (1.40 g, 5.74 band01) in tetrahydrofuran (5.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. Washed with water extract exudates dried (MgSO 4) After the title compound and the solvent was evaporated under reduced pressure (1.15g, 71¾) Was obtained as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 146-148 ° C.
IR (KBr) V: 3235, 2984, 2936, 1759, 1738, 1663, 1620, 1559 cm—1. IR (KBr) V: 3235, 2984, 2936, 1759, 1738, 1663, 1620, 1559 cm— 1 .
Ή-NM (CDC13) : δΐ.26 (3Η, d, J=7.2 Hz), 1.70-1.78 (4H, m), 1.80 (3H, d, J=1.2Hz), 2.28 (3H, d, J=0.9 Hz), 2.31-2.38 (2H, m), 2.94-3.00 (2H, m), 4.14 (2H, q, J=7.2 Hz), 7.77 (2H, d, J-8.7 Hz), 8.11 (2H, d, J=9.0 Hz), 9.61 (1H, bs). Ή-NM (CDC1 3 ): δΐ.26 (3Η, d, J = 7.2 Hz), 1.70-1.78 (4H, m), 1.80 (3H, d, J = 1.2 Hz), 2.28 (3H, d, J = 0.9 Hz), 2.31-2.38 (2H, m), 2.94-3.00 (2H, m), 4.14 (2H, q, J = 7.2 Hz), 7.77 (2H, d, J-8.7 Hz), 8.11 (2H , d, J = 9.0 Hz), 9.61 (1H, bs).
元素分析値: C22H24N305S3F3として Elementary analysis: as C 22 H 24 N 3 0 5 S 3 F 3
計算値 (¾) : C, 46.88; H, 4.29; N, 7.46; S, 17.07. Calculated value (¾): C, 46.88; H, 4.29; N, 7.46; S, 17.07.
実測値 (%) : C, 46.86; H, 4.17; N, 7.62; S, 17.16. Found (%): C, 46.86; H, 4.17; N, 7.62; S, 17.16.
実施例 7-106 Example 7-106
N-{5, 6 -ジメチル -3-ォキソ -8- [ (5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル]- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン卜 3-ピリジン スルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene 3-pyridinesulfonamide
水素化ナトリウム(64.6mg, 1.61匪 ol) (60%油性)のテトラヒドロフラン(2.50 ml) の懸濁液に、 実施例 6-6で得られた 1-ァミノ- 5, 6 -ジメチル -8_[(5, 5, 6, 6, 6 - ペンタフルォ口へキシル)スルファニル]署ィミダゾ [5, 1-c] [1,4]チアジン -3- オン (324 mg, 0.807 mmol) のテトラヒドロフラン (2.50 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。これに参考例 45の方法 Bで得られた残留物のテトラヒドロ フラン (1.00 ml) 溶液を加え、 さらに室温で 17時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (348 mg, 79%) を暗赤色結晶として得た。 酢酸ェ チル-ジェチルエーテルから再結晶して融点 174-176°Cの暗赤色結晶を得た。 IR (KBr) リ: 2940, 1742, 1732, 1663, 1609, 1557 cm"1. To a suspension of sodium hydride (64.6 mg, 1.61 ol) (60% oil) in tetrahydrofuran (2.50 ml) was added 1-amino-5,6-dimethyl-8 _ [( 5,5,6,6,6-pentafluorohexyl) sulfanyl] imidazo [5,1-c] [1,4] thiazin-3-one (324 mg, 0.807 mmol) in tetrahydrofuran (2.50 ml) And stirred under ice cooling for 5 minutes. A solution of the residue obtained in Method B of Reference Example 45 in tetrahydrofuran (1.00 ml) was added thereto, and the mixture was further stirred at room temperature for 17 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (348 mg, 79%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 174-176 ° C. IR (KBr) Re: 2940, 1742, 1732, 1663, 1609, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 1.68-1.84 (4Η, m), 1.81 (3H, d, J=0.6 Hz), 1.96-2.16 (2H, m), 2.29 (3H, d, J=0.6 Hz), 2.98 (2H, t, J=6.9 Hz), 7.44 (1H, dd, J=7.8, 4.8 Hz), 8.24 (1H, dt, J=8. , 1.5 Hz), 8.78 (1H, dd, J=5.1, 1.2 Hz), 9.19 (1H, d, J-2.1 Hz), 9.64 (1H, bs). Ή-NMR (CDC1 3): δ 1.68-1.84 (4Η, m), 1.81 (3H, d, J = 0.6 Hz), 1.96-2.16 (2H, m), 2.29 (3H, d, J = 0.6 Hz) , 2.98 (2H, t, J = 6.9 Hz), 7.44 (1H, dd, J = 7.8, 4.8 Hz), 8.24 (1H, dt, J = 8., 1.5 Hz), 8.78 (1H, dd, J = 5.1, 1.2 Hz), 9.19 (1H, d, J-2.1 Hz), 9.64 (1H, bs).
元素分析値: C19H19N403S3F5として 計算値 (%) : C, 42.06; H, 3.53; , 10.33; S, 17.73. Elemental analysis: as C 19 H 19 N 4 0 3 S 3 F 5 Calculated value (%): C, 42.06; H, 3.53;, 10.33; S, 17.73.
実測値 (¾) : C, 41.90; H, 3.38; N, 10.23; S, 18.00. Found (¾): C, 41.90; H, 3.38; N, 10.23; S, 18.00.
実施例 7-107 Example 7-107
7 -ク口ロ- N - {5, 6-ジメチル- 3 -ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル) スルファニル ]_2,3-ジヒドロ- 1H-イミダゾ [5, l-c] [1,4]チアジン-卜ィリデ ン} - 2, 1, 3-ベンゾォキサゾール -4 -スルホンアミド 7-Cupro-N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] _2,3-dihydro-1H- Imidazo [5, lc] [1,4] thiazine-trilidene}-2,1,3-benzoxazole-4-sulfonamide
水素化ナトリウム(39.9 mg, 0.996 mmol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-6で得られた卜ァミノ- 5, 6-ジメチル -8 - [(5, 5, 6, 6,6 - ペン夕フルォ口へキシル)スルファニル] - 3H-ィミダゾ [5, 1-c] [1,4]チアジン -3- オン (200 mg, 0.498腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2, 1, 3-ベンゾォキサゾ一ル -4 -スルホニル クロリド (252 mg, 0.996匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室 温で 2時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(143 mg, 47%) を喑赤色結晶として得た。 酢酸ェチル-ジェチルェ一テルから再結晶して融点 198-200°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (39.9 mg, 0.996 mmol) (60% oily) in tetrahydrofuran (1.00 ml) was added triamino-5,6-dimethyl-8-[(5 , 5,6,6,6-Pentate fluoridylhexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 tmol) in tetrahydrofuran ( 2.00 ml) solution and stirred under ice-cooling for 5 minutes. To this was added a solution of 2,1,3-benzoxazol-4-sulfonyl chloride (252 mg, 0.996 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. Washed with water extract exudates, to give after drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (143 mg, 47%) as a喑赤white crystals. Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 198-200 ° C.
IR (KBr) V: 3239, 3081, 2946, 1746, 1663, 1609, 1559, 1520 cm IR (KBr) V: 3239, 3081, 2946, 1746, 1663, 1609, 1559, 1520 cm
¾-NMR (CDC13) : δ 1.60-1.76 (4Η, m), 1.81 (3H, d, J=0.9 Hz), 1.90-2.12 (2H, m), 2.33 (3H, d, J=0.6 Hz), 2.92 (2H, t, J=7.2 Hz), 7.54 (1H, d, J=7.2 Hz), 8.06 (1H, d, J=7.5 Hz), 9.85 (1H, bs). ¾-NMR (CDC1 3): δ 1.60-1.76 (4Η, m), 1.81 (3H, d, J = 0.9 Hz), 1.90-2.12 (2H, m), 2.33 (3H, d, J = 0.6 Hz) , 2.92 (2H, t, J = 7.2 Hz), 7.54 (1H, d, J = 7.2 Hz), 8.06 (1H, d, J = 7.5 Hz), 9.85 (1H, bs).
元素分析値: C2。H17N504S3C1F5として Elemental analysis: C 2. H 17 N 5 0 4 S 3 C1F 5
計算値 (%) : C, 38.87; H, 2.77; N, 11.33; S, 15.57. Calculated (%): C, 38.87; H, 2.77; N, 11.33; S, 15.57.
実測値 (¾) : C, 38.95; H, 2.68; N, 11.41; S, 15.64. Found (¾): C, 38.95; H, 2.68; N, 11.41; S, 15.64.
実施例 7-108 Example 7-108
N-{5, 6-ジメチル -3-ォキソ -8_ [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 2, 3_ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }- 2, 1, 3 -ベン ゾォキサジァゾール -4-スルホンアミド N- {5,6-dimethyl-3-oxo-8 _ [(5,5,6,6,6-pentafluorohexyl) sulfanyl]-2,3_dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-1-ylidene} -2,1,3-benzoxaziazol-4-sulfonamide
水素化ナトリウム(265 mg, 6.64mmol) (60%油性)のテトラヒドロフラン(10.0 ml) の懸濁液に、 実施例 6-6で得られたトァミノ- 5, 6-ジメチル- 8- [(5, 5, 6, 6, 6 - ペンタフルォ口へキシル)スルファニル] -3H -ィミダゾ [5,1-c] [1,4]チアジン- 3 - オン (1.48 g, 3.69皿 ol) のテトラヒドロフラン (15.0 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 4-メトキシベンゼンスルホニル クロリド (1.37g, 6.64 mmol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 さらに室温で 18.5 時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を 水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (1.37 g, 65¾) を 合成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 160_162 の暗赤色 結晶を得た。 To a suspension of sodium hydride (265 mg, 6.64 mmol) (60% oil) in tetrahydrofuran (10.0 ml) was added the toamino-5,6-dimethyl-8-[(5,5) obtained in Example 6-6. 5, 6, 6, 6- Add a solution of pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (1.48 g, 3.69 dish ol) in tetrahydrofuran (15.0 ml), and cool under ice-cooling. Stir for a minute. To this was added a solution of 4-methoxybenzenesulfonyl chloride (1.37 g, 6.64 mmol) in tetrahydrofuran (5.00 ml), and the mixture was further stirred at room temperature for 18.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (1.37 g, 65¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 160-162.
IR (KBr) レ : 3241, 2948, 2845, 1755, 1665, 1624, 1597, 1564, 1501 cnf1.IR (KBr) Re: 3241, 2948, 2845, 1755, 1665, 1624, 1597, 1564, 1501 cnf 1 .
Ή-NMR (CDC13) : 51.66-1.82 (4H, in), 1.79 (3H, d, J=1.2 Hz), 1.96-2.14 (2H, m), 2.26 (3H, d, J=0.9 Hz), 2.96 (2H, t, J=6.9 Hz), 3.86 (3H, s), 6.96 (2H, d, J=9.0 Hz), 7.89 (2H, d, J=9.3 Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3): 51.66-1.82 (4H, in), 1.79 (3H, d, J = 1.2 Hz), 1.96-2.14 (2H, m), 2.26 (3H, d, J = 0.9 Hz), 2.96 (2H, t, J = 6.9 Hz), 3.86 (3H, s), 6.96 (2H, d, J = 9.0 Hz), 7.89 (2H, d, J = 9.3 Hz), 9.62 (1H, bs).
元素分析値: C21H22N304S3F3として Elementary analysis: as C 21 H 22 N 3 0 4 S 3 F 3
計算値 (¾) : C, 44.13; H, 3.88; N, 7.35; S, 16.83. Calculated (¾): C, 44.13; H, 3.88; N, 7.35; S, 16.83.
実測値 (¾) : C, 43.94; H, 3.66; N, 7.48; S, 16.93. Found (¾): C, 43.94; H, 3.66; N, 7.48; S, 16.93.
実施例 7-109 Example 7-109
N-{5, 6-ジメチル- 3-ォキソ -8- [ (5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル]- 2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }-2_メトキシ - 4_メチルベンゼンスルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydrido-1H-imidazo [5 , 1-c] [1, 4] thiazine-1-ylidene} -2_methoxy-4_methylbenzenesulfonamide
水素化ナトリウム(59.8mg, 1.49 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-6で得られた卜ァミノ- 5,6-ジメチル -8- [(5, 5, 6,6, 6- ペンタフルォ口へキシル)スルファニル ]- 3H-ィミダゾ [5, 1-c] [1, ]チアジン- 3- オン (300 mg, 0.747腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2-メトキシ -4-メチルベンゼンスルホニル クロリド (337 mg, 1.49匪 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 さらに室 温で 20時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(332 mg, 76%) を合成した。酢酸ェチル-ジェチルエーテルから再結晶して融点 192-194°Cの暗赤 色結晶を得た。 ' IR (KBr) v 3270, 2946, 1717, 1665, 1613, 1557 cm"1. To a suspension of sodium hydride (59.8 mg, 1.49 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8-[(5 , 5,6,6,6-Pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,] thiazin-3-one (300 mg, 0.747 t) of tetrahydrofuran (2.00 ml) The solution was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 2-methoxy-4-methylbenzenesulfonyl chloride (337 mg, 1.49 marl) in tetrahydrofuran (2.00 ml), and the mixture was further stirred at room temperature for 20 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (332 mg, 76%). The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals with a melting point of 192-194 ° C. ' IR (KBr) v 3270, 2946, 1717, 1665, 1613, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 1.62-1.78 (4H, m), 1.80 (3H, d, J=0.8 Hz), 1.86-2.16 (2H, m), 2.30 (3H, d, J=1.0 Hz), 2.40 (3H, s), 2.91 (2H, t, J=7.4 Hz), 3.87 (3H, s), 6.78 (1H, s), 6.86 (1H, d, J=7.6 Hz), 7.89 (1H, d, J=7.6 Hz), 9.85 (1H, bs). Ή-NMR (CDC1 3): δ 1.62-1.78 (4H, m), 1.80 (3H, d, J = 0.8 Hz), 1.86-2.16 (2H, m), 2.30 (3H, d, J = 1.0 Hz) , 2.40 (3H, s), 2.91 (2H, t, J = 7.4 Hz), 3.87 (3H, s), 6.78 (1H, s), 6.86 (1H, d, J = 7.6 Hz), 7.89 (1H, d, J = 7.6 Hz), 9.85 (1H, bs).
元素分析値: C22H24N304S3F5 -0.5H20として Elementary analysis: as C 22 H 24 N 3 0 4 S 3 F 5 -0.5H 2 0
計算値 (¾) : C, 44.44; H, 4.24; N, 7.07; S, 16.18. Calculated value (¾): C, 44.44; H, 4.24; N, 7.07; S, 16.18.
実測値 (¾) : C, 44.36; H, 4.09; N, 7.11; S, 16.43. Found (¾): C, 44.36; H, 4.09; N, 7.11; S, 16.43.
実施例 7-110 Example 7-110
5-ブロモ -N- {5, 6 -ジメチル- 3-ォキソ - 8_[(5, 5, 6, 6, 6 -ペンタフルォ口へキシル) スルファニル ] _2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1 , 4]チアジン-卜イリデ 水素化ナトリウム(59.8mg, 1.49mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例, 6-6で得られた 1-ァミノ- 5, 6-ジメチル -8 - [(5, 5, 6,6,6- ペンタフルォ口へキシル)スルファニル ]-3H-イミダゾ [5, 1-c] [1,4]チアジン- 3- オン (300 mg, 0.747腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 5-ブロモ -2-メトキシベンゼンスルホニル クロリド (436 mg, 1.49 腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 さらに室 温で 3時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(380 mg, 78%) を合成した。酢酸ェチル-ジェチルエーテルから再結晶して融点 169- 170°Cの暗赤 色結晶を得た。 5-bromo-N- {5,6-dimethyl-3-oxo-8 _ [(5,5,6,6,6-pentafluorohexyl) sulfanyl] _2,3-dihydro-1H-imidazo [5, c] To a suspension of [1,4] thiazine-triide sodium hydride (59.8 mg, 1.49 mmol) (60% oily) in tetrahydrofuran (2.00 ml) was added 1- obtained in Example, 6-6. Amino-5,6-dimethyl-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (300 mg, 0.747 t ol) in tetrahydrofuran (2.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 5-bromo-2-methoxybenzenesulfonyl chloride (436 mg, 1.49 t) in tetrahydrofuran (2.00 ml), and the mixture was further stirred at room temperature for 3 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (380 mg, 78%) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 169-170 ° C.
IR (KBr) V: 3241, 1721, 1663, 1611, 1557, 1481 cm—1. ' Ή -腿 (CDC13) : (51.64-1.86 (4H, m), 1.82 (3H, d, J=1.2 Hz), 1.88-2.18 (2H, m), 2.31 (3H, d, J-1.0 Hz), 2.94 (2H, t, J=6.8 Hz), 3.08 (3H, s), 6.87 (1H, d, J=8.8Hz), 7.60 (1H, dd, J=8.8, 2.6 Hz), 8.17 (1H, d, J=2.2 Hz), 9.81 (1H, bs). IR (KBr) V:. 3241 , 1721, 1663, 1611, 1557, 1481 cm- 1 'Ή - thigh (CDC1 3): (51.64-1.86 ( 4H, m), 1.82 (3H, d, J = 1.2 Hz ), 1.88-2.18 (2H, m), 2.31 (3H, d, J-1.0 Hz), 2.94 (2H, t, J = 6.8 Hz), 3.08 (3H, s), 6.87 (1H, d, J = 8.8 Hz), 7.60 (1H, dd, J = 8.8, 2.6 Hz), 8.17 (1H, d, J = 2.2 Hz), 9.81 (1H, bs).
元素分析値: C21H21N304S3BrFsとして Elementary analysis: as C 21 H 21 N 3 0 4 S 3 BrF s
計算値 (%) : C, 38.77; H, 3.25; N, 6.46; S, 14.79. 実測値 (¾) : C, 38.40; H, 3.19; N, 6.38; S, 14.93. Calculated value (%): C, 38.77; H, 3.25; N, 6.46; S, 14.79. Found (¾): C, 38.40; H, 3.19; N, 6.38; S, 14.93.
実施例 7-112 Example 7-112
2, 4 -ジク口 D-6-[{5, 6-ジメチル- 3-ォキソ - 8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキ シル)スルファニル]- 2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリ デン }ァミノ]スルホニル]フエニル- 3,5-ジクロロベンゼンスルホンアミド 水素化ナトリウム(79.7mg, 1.99丽 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6 - 6で得られた卜アミノ- 5, 6-ジメチル- 8 - [(5, 5, 6,6,6- ペン夕フルォ口へキシル)スルファニル] - 3H-イミダゾ [5,卜 c] [ 1, 4]チアジン- 3 - オン (200 mg, 0.498 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 3,5-ジクロロ- 2-ヒドロキシベンゼンスルホニル ク ロリド (263 mg, 0.996翻 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さ らに室温で 25時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出 した。抽出液を水洗、 乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物(302 mg, 71¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶し て融点 181- 183°Cの暗赤色結晶を得た。 2,4-Dic-mouth D-6-[{5,6-Dimethyl-3-oxo-3--8-[(5,5,6,6,6-Pentafluorohexyl) sulfanyl]-2,3 -Dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene} amino] sulfonyl] phenyl-3,5-dichlorobenzenesulfonamide sodium hydride (79.7 mg, 1.99 mol ) (60% oily) in tetrahydrofuran (2.00 ml) was added to the suspension of triamino-5,6-dimethyl-8-[(5,5,6,6,6- Pentafluorohexyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.498 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled on ice. Stir for a minute. To this was added a solution of 3,5-dichloro-2-hydroxybenzenesulfonyl chloride (263 mg, 0.996 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 25 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (302 mg, 71¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 181-183 ° C.
IR (KBr)レ : 3700-2800, 3083, 2930, 1738, 1661, 1607, 1551 cm"1. IR (KBr): 3700-2800, 3083, 2930, 1738, 1661, 1607, 1551 cm " 1 .
Ή-NMR (CDC") : δ 1.68-1.86 (4Η, m), 1.82 (3H, d, J=1.2 Hz), 1.97-2.16 (2H, m), 2.30 (3H, d, J=0.9 Hz), 3.03 (2H, t, J=7.5 Hz), 7.52 (1H, d, J=2.7 Hz), 7.66 (1H, d, J=3.0 Hz), 7.89 (1H, d, J=2.4 Hz), 8.07 (1H, d, J=2.4 Hz), 8.73 (m, bs), 9.60 (1H, bs). Ή-NMR (CDC "): δ 1.68-1.86 (4Η, m), 1.82 (3H, d, J = 1.2 Hz), 1.97-2.16 (2H, m), 2.30 (3H, d, J = 0.9 Hz) , 3.03 (2H, t, J = 7.5 Hz), 7.52 (1H, d, J = 2.7 Hz), 7.66 (1H, d, J = 3.0 Hz), 7.89 (1H, d, J = 2.4 Hz), 8.07 (1H, d, J = 2.4 Hz), 8.73 (m, bs), 9.60 (1H, bs).
元素分析値: C26H2。N307S4C14F5として Elemental analysis: C 26 H 2. N 3 0 7 S 4 C1 4 F 5
計算値 (¾) : C, 36.67; H, 2.37; N, 4.93; S, 15.06. Calculated (¾): C, 36.67; H, 2.37; N, 4.93; S, 15.06.
実測値 (%) : C, 36.83; H, 2.38; N, 5.01; S, 15.29. Found (%): C, 36.83; H, 2.38; N, 5.01; S, 15.29.
実施例 7-113 Example 7-113
3, 5 -ジクロ口- N-{5, 6-ジメチル- 3 -ォキソ - 8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシ ル)スルファニル] - 2, 3 -ジヒドロ- 1H -イミダゾ [5,1- c] [1,4]チアジン- 1 -ィリデ 実施例 7-112 で得られた 2, 4-ジクロロ- 6- [{5, 6 -ジメチル- 3-ォキソ 一 8 - [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニル]一 2, 3 -ジヒドロ- 1H-ィ ミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン }アミノ]スルホニル]フエニル -3, 5 -ジ クロ口ベンゼンスルホンアミド (250 nig, 0.294醒 ol) のエタノール (4.00 ml) 及びテトラヒドロフラン (4.00 ml) 溶液に、 1N水酸化ナトリウム水溶液 (0.969 ml) を加えた。 反応混合物を室温で 30分間かき混ぜ、 続いて 100°Cで 2時間かき 混ぜた後、溶媒を減圧下に留去した。残渣に 1N塩酸を加えて、生成した沈澱物を 濾取し、 水洗後、 乾燥して題記化合物 (147 mg, 80¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 183- 184°Cの暗赤色結晶を得 た。 3,5-Dichloro-N- {5,6-dimethyl-3-oxo- 8-[[5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H -Imidazo [5,1-c] [1,4] thiazine-1-ylide 2,4-Dichloro-6-[{5,6-dimethyl-3-oxo-1-8- obtained in Example 7-112 [(5,5,6,6,6-pentahexyl hexyl) sulfanyl] -1,2,3-dihydro-1H-di Midazo [5,1-c] [1,4] thiazin-1-ylidene} amino] sulfonyl] phenyl-3,5-dichlorobenzene benzenesulfonamide (250 nig, 0.294 ol) in ethanol (4.00 ml) and To a solution of tetrahydrofuran (4.00 ml) was added a 1N aqueous solution of sodium hydroxide (0.969 ml). The reaction mixture was stirred at room temperature for 30 minutes and subsequently at 100 ° C. for 2 hours, and the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and the resulting precipitate was collected by filtration, washed with water, and dried to give the title compound (147 mg, 80 °) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 183-184 ° C.
IR (KBr) V: 3700-2800, 3073, 2942, 1744, 1661, 1597, 1549 cm"1. IR (KBr) V: 3700-2800, 3073, 2942, 1744, 1661, 1597, 1549 cm " 1 .
¾-NMR (CDC13) : δ 1.69-1.88 (4Η, m), 1.82 (3H, d, J=1.2 Hz), 1.97-2.16 (2H, m), 2.30 (3H, d, J=0.9 Hz), 3.03 (2H, t, 1=Ί Λ Hz), 7.52 (1H, d, J=2.4 Hz), ¾-NMR (CDC1 3): δ 1.69-1.88 (4Η, m), 1.82 (3H, d, J = 1.2 Hz), 1.97-2.16 (2H, m), 2.30 (3H, d, J = 0.9 Hz) , 3.03 (2H, t, 1 = Ί Λ Hz), 7.52 (1H, d, J = 2.4 Hz),
7.66 (1H, d, J=2.4 Hz), 8.75 (1H, bs), 9.58 (1H, bs). 7.66 (1H, d, J = 2.4 Hz), 8.75 (1H, bs), 9.58 (1H, bs).
元素分析値: C2。H1SN304S3C12F5として Elemental analysis: C 2. H 1S N 3 0 4 S 3 C1 2 F 5
計算値 (¾) : C, 38.34; H, 2.90; N, 6.71; S, 15.36. Calculated (¾): C, 38.34; H, 2.90; N, 6.71; S, 15.36.
実測値 (¾) : C, 38.37; H, 2.82; N, 6.56; S, 15.55. Found (¾): C, 38.37; H, 2.82; N, 6.56; S, 15.55.
実施例 7-115 Example 7-115
N - {5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6 -ペンタフルォ口へキシル)スルファニ ル]- 2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }- 3-(4-ピリジ 卜フエニルピリジン (2.16 g, 13.6廳 ol)およびクロロスルホン酸 (4.91 g, 40.9 mmol)の混合物を室温で 16時間、 60で で 3時間攪拌した。 反応液を卜ルェ ン(50 ml)で希釈し、 氷 (80 g)に注ぎ、 酢酸ェチル (50 ml)を加え、 炭酸ナトリウ ムで中和した。 水層を酢酸ェチル (50 ml)で抽出した。 有機層を水、 飽和炭酸水素 ナトリウム、 飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥後、 濃縮乾固して淡紫 色固形物を得た。 これを塩化チォニル(10 ml)に溶解して室温で 7日間攪拌した。 反応液をトルエン(100 ml)で希釈して濃縮し、 再度トルエン(100 ml)で希釈して 濃縮し、 残渣を酢酸ェチル (50 ml)に懸濁した。 析出物を濾取し、 酢酸ェチルで 洗浄後、 乾燥して白色固形物を得た。 N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]-2,3-dihydro-1H-imidazo [5,1- c] A mixture of [1,4] thiazine-1-ylidene} -3- (4-pyriditophenylphenylpyridine (2.16 g, 13.6 ol) and chlorosulfonic acid (4.91 g, 40.9 mmol) at room temperature for 16 hours The mixture was stirred for 3 hours at 60. The reaction solution was diluted with toluene (50 ml), poured into ice (80 g), added with ethyl acetate (50 ml), and neutralized with sodium carbonate. The organic layer was washed with water, saturated sodium bicarbonate and brine, dried over sodium sulfate, and concentrated to dryness to give a pale purple solid. The reaction mixture was dissolved in thionyl chloride (10 ml) and stirred at room temperature for 7 days The reaction mixture was diluted with toluene (100 ml), concentrated, diluted again with toluene (100 ml), concentrated, and the residue was ethyl acetate (50 ml) The precipitate was collected by filtration, washed with ethyl acetate, and dried to give a white solid.
実施例 6 6で得た化合物 (235 mg, 0.585 mmol) の THF (5.0 ml) 懸濁液に 水素化ナトリウム (純度 60% 油性, 82.0 mg, 2.05 mmol) を室温で加え、 15分間 攪拌後、 上記白色固形物(187 mg)を加え、 さらに 4.5時間攪拌した。 反応液にメ 夕ノール(1 ml) および水 1N塩酸 (3.0 ml)を加え、 酢酸ェチル (35 ml)で希釈 し、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮 乾固後、 残渣を酢酸ェチルで希釈した。 析出物を濾取し、 酢酸ェチルおよびジェ チルエーテルで洗浄後、 メタノ一ルー酢酸ェチルから結晶化して表題化合物 (45 mg, 0.073 mmol, 12.4%)を灰赤色粉末として得た。 Example 6 To a suspension of the compound obtained in Step 6 (235 mg, 0.585 mmol) in THF (5.0 ml) was added. Sodium hydride (purity 60% oily, 82.0 mg, 2.05 mmol) was added at room temperature, and the mixture was stirred for 15 minutes. Then, the white solid (187 mg) was added, and the mixture was further stirred for 4.5 hours. The reaction mixture was diluted with methanol (1 ml) and 1N hydrochloric acid (3.0 ml), diluted with ethyl acetate (35 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with ethyl acetate. The precipitate was collected by filtration, washed with ethyl acetate and ethyl ether, and crystallized from methyl ethyl acetate to give the title compound (45 mg, 0.073 mmol, 12.4%) as a gray-red powder.
Ή-NMR (300MHz, CDC13) : 61.72 (4H, m), 1.80 (3H, s), 2.02 (2H, m), 2.28 (3H, s), 2.97 (2H, t, J= 6.9 Hz), 7.53 (2H, m), 7.63 (1H, t, J= 8.0 Hz), 7.82 (1H, d, J= 6.6 Hz), 8.04 (1H, brd, J= 7.5 Hz), 8.25 (1H, brs), 8.71 (2H, m). 元素分析値: C25 N403S3F50.2H20として計算値: C, 48.25; H, 3.79; N, 9.00; S, 15.46 (%) 、 実測値: C, 48.03; H, 3.70; N, 8.93; S, 15.72 (%) . Ή-NMR (300MHz, CDC1 3 ): 61.72 (4H, m), 1.80 (3H, s), 2.02 (2H, m), 2.28 (3H, s), 2.97 (2H, t, J = 6.9 Hz), 7.53 (2H, m), 7.63 (1H, t, J = 8.0 Hz), 7.82 (1H, d, J = 6.6 Hz), 8.04 (1H, brd, J = 7.5 Hz), 8.25 (1H, brs), 8.71 (2H, m). Elemental analysis: Calculated for C 25 N 4 0 3 S 3 F 5 0.2H 2 0: C, 48.25; H, 3.79; N, 9.00; S, 15.46 (%), found : C, 48.03; H, 3.70; N, 8.93; S, 15.72 (%).
mp. 205.0-206.0°C mp. 205.0-206.0 ° C
実施例 7-118 Example 7-118
4-ブロモ- N- (5, 6-ジメチル- 3-ォキソ -8- { [2- (2, 2, 1-トリフルォロェトキシ)ェチ ル]スルファニル } - 2, 3 ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデ 水素化ナトリウム(28.5 mg, 0.711 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-26で得られたトァミノ- 5, 6-ジメチル -8- {[2- (2, 2, 2- トリフルォロェトキシ)ェチル]スルファ二ル}鲁ィミダゾ [5, 1-c] [1 , 4]チアジ ン- 3-オン (120mg, 0.340腿 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混ぜた。これに 4-ブロモベンゼンスルホニル クロリド(185 mg, 0.711 mmol) のテトラヒドロフラン (1.00ml) 溶液を加え、 さらに室温で 1時間 かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (88.6 mg, 46¾) を暗赤色 結晶として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 158- 159 の 暗赤色結晶を得た。 4-bromo-N- (5,6-dimethyl-3-oxo-8-{[2- (2,2,1-trifluoroethoxy) ethyl] sulfanyl} -2,3 dihydro-1H-imidazo [5, 1-c] [1, 4] Thiazine-1-ylide sodium hydride (28.5 mg, 0.711 mmol) (60% oily) in a suspension of tetrahydrofuran (2.00 ml) was prepared in Example 6-26. The resulting toamino-5,6-dimethyl-8-{[2- (2,2,2-trifluoroethoxy) ethyl] sulfanyl} 鲁 imidazo [5,1-c] [1,4] thiazi A solution of 1-3-one (120 mg, 0.340 tmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice-cooling for 5 minutes, and mixed with 4-bromobenzenesulfonyl chloride (185 mg, 0.711 mmol) in tetrahydrofuran (1.00 ml). ) was added, evaporated and further stirred at room temperature for 1 hour. the reaction mixture was extracted with acetic acid Echiru by adding 1N hydrochloric acid. the extract was washed with water, dried (MgSO 4), of the solvent under reduced pressure Title compound Te (88.6 mg, 46¾) was obtained as a dark red crystalline acetate Echiru -. Jechirue give recrystallized to dark red crystals, mp 158- 159 from scratch ether.
IR (KBr) V 3246, 3096, 2928, 1759, 1744, 1663, 1622, 1559 cm—1. IR (KBr) V 3246, 3096, 2928, 1759, 1744, 1663, 1622, 1559 cm- 1 .
Ή - NMR (CDC13) : δ 1.80 (3Η, s), 2.27 (3H, s), 3.19 (2H, t, J=6.2 Hz), 3.83 (2H, t, J=5.8 Hz), 3.85 (2H, q, J-8.8 Hz), 7.63 (2H, d, J=8.4 Hz), 7.83 (2H, d, J=8.8 Hz), 9.60 (1H, bs). Ή - NMR (CDC1 3): δ 1.80 (3Η, s), 2.27 (3H, s), 3.19 (2H, t, J = 6.2 Hz), 3.83 (2H, t, J = 5.8 Hz), 3.85 (2H, q, J-8.8 Hz), 7.63 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.60 (1H , bs).
元素分析値: C18H17N304S3BrF3として Elementary analysis: as C 18 H 17 N 3 0 4 S 3 BrF 3
計算値 (%) : C, 37.77; H, 2.98; N, 7.34; S, 16.80. Calculated (%): C, 37.77; H, 2.98; N, 7.34; S, 16.80.
実測値 (¾) : C, 37.68; H, 2.79; N, 7.30; S, 16.79. Found (¾): C, 37.68; H, 2.79; N, 7.30; S, 16.79.
実施例 7-119 Example 7-119
N - {5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニ ル]- 2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン }-2- (トリフル 水素化ナトリウム(39.9 mg, 0.996丽 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6 - 6で得られた 1-ァミノ- 5, 6-ジメチル -8-[(5, 5, 6, 6,6 - ペン夕フルォ口へキシル)スルファニル]鲁ィミダゾ [5, 1-c] [1,4]チアジン -3- オン (200 mg, 0.498匪 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2- (トリフルォロメチル)ベンゼンスルホニル クロ リド (244 mg, 0.996腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さら に室温で 1.5時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出し た。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(182 mg, 60%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 189-191°C の暗赤色結晶を得た。 N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydrido-1H-imidazo [5 , 1-c] [1,4] thiazine-triylidene} -2- (Trifluodium hydride (39.9 mg, 0.996 丽 ol) (60% oil) in tetrahydrofuran (1.00 ml) suspension 1-Amino-5,6-dimethyl-8-[(5,5,6,6,6-hexylfluorhexyl) sulfanyl] dimidazo obtained in 6-6 [5,1-c] [ A solution of 1,4] thiazin-3-one (200 mg, 0.498 ol) in tetrahydrofuran (2.00 ml) was added, and the mixture was stirred for 5 minutes under ice-cooling, followed by 2- (trifluoromethyl) benzenesulfonyl chloride ( 244 mg, was added tetrahydrofuran (1.00 ml) solution of 0.996 thigh ol), was stirred for 1.5 hours at room temperature further. the reaction mixture was added with 1N hydrochloric acid and extracted with acetic acid Echiru. the extract was washed with water, dried (MgSO 4 )rear The solvent was synthesized the title compound was evaporated under reduced pressure (182 mg, 60%) acetic acid Echiru -. Jechirue was recrystallized from a ether to give a dark red crystals, mp 189-191 ° C.
IR (KBr) リ : 3218, 2953, 1738, 1717, 1661,' 1611, 1557 cm—1. IR (KBr): 3218, 2953, 1738, 1717, 1661, '1611, 1557 cm— 1 .
¾-NMR (CDC13) : δ 1.62-1.76 (4Η, ι), 1.80 (3H, d, J=1.0 Hz), 1.86-2.16 (2H, m), 2.29 (3H, d, J=0.8 Hz), 2.94 (2H, t, J=6.6 Hz), 7.65-7.74 (2H, m), 7.79-7.90 (1H, m), 8.32-8.40 (1H, m), 9.66 (1H, bs). ¾-NMR (CDC1 3): δ 1.62-1.76 (4Η, ι), 1.80 (3H, d, J = 1.0 Hz), 1.86-2.16 (2H, m), 2.29 (3H, d, J = 0.8 Hz) , 2.94 (2H, t, J = 6.6 Hz), 7.65-7.74 (2H, m), 7.79-7.90 (1H, m), 8.32-8.40 (1H, m), 9.66 (1H, bs).
元素分析値: C21H19N303S3F5として Elemental analysis: as C 21 H 19 N 3 0 3 S 3 F 5
計算値 (¾) : C, 41.38; H, 3.14; N, 6.89; S, 15.78. Calculated value (¾): C, 41.38; H, 3.14; N, 6.89; S, 15.78.
実測値 (¾) : C, 41.32; H, 3.06; N, 6.85; S, 16.00. Found (¾): C, 41.32; H, 3.06; N, 6.85; S, 16.00.
実施例 7-120 Example 7-120
N-{5, 6-ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 2, 3 -ジヒドロ- 1H -イミダゾ [5,1 - c] [1,4]チアジン - 1 -ィリデン卜 2- (トリフル 水素化ナトリウム(39.9 mg, 0.996 mmol) (60 油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6- 6で得られた卜ァミノ- 5, 6 -ジメチル- 8-[(5, 5, 6, 6,6- ペンタフルォ口へキシル)スルファニル] -31-トイミダゾ [5, 1-c] [1, 4]チアジン- 3 - オン (200 mg, 0.498腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2- (トリフルォロメトキシ)ベンゼンスルホニル ク ロリド (250 mg, 0.959 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さ らに室温で 1.5時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出 した。 抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物(215 mg, 69%) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 190-192°Cの暗赤色結晶を得た。 N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene 2- (triflu A suspension of sodium hydride (39.9 mg, 0.996 mmol) (60 oily) in tetrahydrofuran (1.00 ml) was added to the triamino-5,6-dimethyl-8-[(5,5) obtained in Example 6-6. 5,6,6,6-Pentafluorohexyl) sulfanyl] -31-toimidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.498 tmol) of tetrahydrofuran (2.00 ml) The solution was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 2- (trifluoromethoxy) benzenesulfonyl chloride (250 mg, 0.959 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (215 mg, 69%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 190-192 ° C.
IR (KBr) V: 3252, 2930, 1746, 1726, 1661, 1615, 1559 cm"1. IR (KBr) V: 3252, 2930, 1746, 1726, 1661, 1615, 1559 cm " 1 .
Ή-NMR (CDC13) : 61.61-1.77 (4H, m), 1.81 (3H, s), 1.90-2.10 (2H, m), 2.30 (3H, s), 2.94 (2H, t, J=7.2 Hz), 7.34-7.44 (2H, m), 7.60 (1H, dt, J=8.1, 1.5 Hz), 8.14 (1H, dd, J=7.8, 1.5 Hz), 9.79 (1H, bs). Ή-NMR (CDC1 3): 61.61-1.77 (4H, m), 1.81 (3H, s), 1.90-2.10 (2H, m), 2.30 (3H, s), 2.94 (2H, t, J = 7.2 Hz ), 7.34-7.44 (2H, m), 7.60 (1H, dt, J = 8.1, 1.5 Hz), 8.14 (1H, dd, J = 7.8, 1.5 Hz), 9.79 (1H, bs).
元素分析値: C21H19N304S3F5として Elementary analysis: as C 21 H 19 N 3 0 4 S 3 F 5
計算値 (¾) : C, 40.32; H, 3.06; N, 6.72; S, 15.38. Calculated value (¾): C, 40.32; H, 3.06; N, 6.72; S, 15.38.
実測値 (%) : C, 40.34; H, 2.91; N, 6.77; S, 15.52. Found (%): C, 40.34; H, 2.91; N, 6.77; S, 15.52.
実施例 7-121 Example 7-121
N - {5, 6-ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 2, 3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1, 4]チアジン- 1-ィリデン }- 2-(卜ナフチ 水素化ナトリウム(39.9 mg, 0.996 mmol) (60%油性)のテトラヒドロフラン(1.00 il) の懸濁液に、 実施例 6-6で得られた 1-ァミノ- 5, 6-ジメチル- 8- [(5, 5, 6, 6, 6 - ペンタフルォ口へキシル)スルファニル ]-3H-ィミダゾ [5, 1-c] [1,4]チアジン- 3- オン (200 mg, 0.498皿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 2- (卜ナフチル)ベンゼンスルホニル クロリド (250 mg, 0.959讓 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 1.5時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出 液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (215 mg, 69%) を合成した。酢酸ェチル -ジェチルエーテルから再結晶して融点 139-U0°Cの暗赤 色結晶を得た。 N-{5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]-2,3-dihydro-1H-imidazo [5,1- c] Example 6 was added to a suspension of [1,4] thiazine-1-ylidene}-2- (tonaphthyl sodium hydride (39.9 mg, 0.996 mmol) (60% oil) in tetrahydrofuran (1.00 il). 1-Amino-5,6-dimethyl-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1, 4] A solution of thiazin-3-one (200 mg, 0.498 dish ol) in tetrahydrofuran (2.00 ml) was added, and the mixture was stirred for 5 minutes under ice-cooling, followed by 2- (tonaphthyl) benzenesulfonyl chloride (250 mg, 0.959 parts). ol) in tetrahydrofuran (1.00 ml), stirred at room temperature for 1.5 hours, added 1N hydrochloric acid to the reaction mixture, and extracted with ethyl acetate. Washing liquid, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (215 mg, 69%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 139-U0 ° C.
IR (KBr) V: 3200, 3075, 2948, 1755, 1665, 1622, 1566 cm一1. IR (KBr) V: 3200, 3075, 2948, 1755, 1665, 1622, 1566 cm one 1.
Ή— NMR (CDC13) : (51.63-1.80 (4H, m), 1.83 (3H, d, J=0.9Hz), 1.90-2.10 (2H, m), 2.30 (3H, d, J=0.9 Hz), 2.94 (2H, t, J=7.2 Hz), 3.50-3.57 (2H, m), 3.64-3.73 (2H, m), 7.35-7.43 (2H, m), 7.46-7.56 (2H, m), 7.78 (1H, dd, J=7.2, 2.4 Hz), 7.86-7.90 (1H, m), 8.04-8.08 (1H, m), 9.50 (1H, bs). Ή- NMR (CDC1 3): ( 51.63-1.80 (4H, m), 1.83 (3H, d, J = 0.9Hz), 1.90-2.10 (2H, m), 2.30 (3H, d, J = 0.9 Hz) , 2.94 (2H, t, J = 7.2 Hz), 3.50-3.57 (2H, m), 3.64-3.73 (2H, m), 7.35-7.43 (2H, m), 7.46-7.56 (2H, m), 7.78 (1H, dd, J = 7.2, 2.4 Hz), 7.86-7.90 (1H, m), 8.04-8.08 (1H, m), 9.50 (1H, bs).
元素分析値: C26H26N303S3F5として Elemental analysis: as C 26 H 26 N 3 0 3 S 3 F 5
計算値 (¾) : C, 50.39; H, 4.23; N, 6.78; S, 15.52. Calculated value (¾): C, 50.39; H, 4.23; N, 6.78; S, 15.52.
実測値 (%) : C, 50.39; H, 4.25; N, 6.80; S, 15.51. Found (%): C, 50.39; H, 4.25; N, 6.80; S, 15.51.
実施例 7-122 Example 7-122
N - {5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6,6, 6-ペン夕フルォ口へキシル)スルファニ ル] - 2, 3 -ジヒドロ- 1H -イミダゾ [5, l_c] [1,4]チアジン-卜イリデン } - 4- (1-ピペリ ジニル)ベンゼンスルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine-triylidene}-4- (1-piperidinyl) benzenesulfonamide
卜フエ二ルビペリジン (2.67 g, 16.2讓 ol)およびクロロスルホン酸 (5.84 g, 48.6匪 ol)の混合物を 6(T で 7日間攪拌した。 反応液をトルエン(50 ml)で希釈 し、 氷 (80 g)に注ぎ、 酢酸ェチル(50 ml)を加え、 炭酸ナトリウムで中和した。 水 層を酢酸ェチル (50 ml)で抽出した。 有機層を水、 飽和炭酸水素ナトリウム、 飽和 食塩水で洗浄後、 硫酸ナトリゥムで乾燥後、 濃縮乾固して塩化 4- (卜ピペリジル) ベンゼンスルホニルおよび原料の 1:4.8混合物(2.50 g, 塩化物の純度 25%, 2.4 mmol, 14.8¾)を褐色油状物として得た。 実施例 6-6で得た化合物(254 mg, 0.633 mmol) の THF (5.0 ml) 懸濁液に水素化ナトリウム(純度 60 油性, 63.3 mg, 1.58 腿 ol) を室温で加え、 20分間攪拌後、 上記褐色油状物 (純度 25%, 0.724 g, 0.696 mmol)の THF (1 ml)溶液を加え、さらに 1時間攪拌した。反応液にメタノール(1 ml) および IN塩酸 (1.6ml)を加え、 酢酸ェチル (30ml)で希釈し、 0.1N塩酸、 水お よび飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣をメタ ノールで希釈した。 析出物を濾取し、 メタノールで洗浄後、 乾燥して表記化合物 (329 mg, 0.527 mmol, 83.2»を褐紫色粉末として得た。 ¾-NMR (300MHz, CDC13): 51.65 (8H, HI), 1.75 (4H, m), 1.78 (3H, q, J= 0.9 Hz), 2.05 (2H, tt, J= 18.2, 7.1 Hz), 2.25 (3H, q, J= 0.6 Hz), 2.95 (2H, t, J= 6.8 Hz), 3.32 (4H, m), 6.86 (2H, d, J= 9.0 Hz), 7.76 (2H, d, J= 9.3Hz), 9.63 (1H, brs). A mixture of triphenylbiperidine (2.67 g, 16.2 alcohol) and chlorosulfonic acid (5.84 g, 48.6 marl) was stirred for 6 days at T. The reaction solution was diluted with toluene (50 ml), and ice ( 80 g), ethyl acetate (50 ml) was added, the mixture was neutralized with sodium carbonate, the aqueous layer was extracted with ethyl acetate (50 ml), and the organic layer was washed with water, saturated sodium bicarbonate, and saturated saline. Then, the mixture was dried over sodium sulfate, concentrated to dryness, and concentrated to dryness to give a 1: 4.8 mixture of 4- (topiperidyl) benzenesulfonyl chloride and the raw material (2.50 g, chloride purity 25%, 2.4 mmol, 14.8¾) as a brown oil. To a suspension of the compound obtained in Example 6-6 (254 mg, 0.633 mmol) in THF (5.0 ml) was added sodium hydride (purity: 60, oily: 63.3 mg, 1.58 mol) at room temperature. After stirring for 20 minutes, a solution of the above brown oil (purity 25%, 0.724 g, 0.696 mmol) in THF (1 ml) was added, and the mixture was further stirred for 1 hour. Methanol (1 ml) and IN hydrochloric acid (1.6 ml) were added to the reaction solution, diluted with ethyl acetate (30 ml), washed with 0.1 N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. Thereafter, the residue was diluted with methanol, and the precipitate was collected by filtration, washed with methanol, and dried to obtain the title compound (329 mg, 0.527 mmol, 83.2 »as a brown-purple powder). ¾-NMR (300MHz, CDC1 3 ): 51.65 (8H, HI), 1.75 (4H, m), 1.78 (3H, q, J = 0.9 Hz), 2.05 (2H, tt, J = 18.2, 7.1 Hz), 2.25 (3H, q, J = 0.6 Hz), 2.95 (2H, t, J = 6.8 Hz), 3.32 (4H, m), 6.86 (2H, d, J = 9.0 Hz), 7.76 (2H, d, J = 9.3Hz), 9.63 (1H, brs).
元素分析値: C25 9N403S3F5として計算値: C, 48.07; H, 4.68; N, 8.97; S, 15.40; F, 15.21 (%) 、 実測値: C, 48.20; H, 4.56; N, 8.95; S, 15.47; F, 15.15 (%) . mp. 175.0-179.0°C Elemental analysis: Calculated for C 25 9 N 4 0 3 S 3 F 5 : C, 48.07; H, 4.68; N, 8.97; S, 15.40; F, 15.21 (%), found: C, 48.20; H , 4.56; N, 8.95; S, 15.47; F, 15.15 (%) .mp. 175.0-179.0 ° C
実施例 7-123 Example 7-123
N - {8- [(5-フルォロペンチル)スルファニル ]_5, 6-ジメチル- 3 -ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン卜 4-(卜ピベリジニル)ベンゼ ンスルホンアミド N- {8-[(5-Fluoropentyl) sulfanyl] _5,6-dimethyl-3-oxo-2,3-dihydrone-1H-imidazo [5, tric] [1,4] thiazine-triident 4 -(Topiberidinyl) benzene sulfonamide
実施例 6- 23で得た化合物 (193 mg, 0.612腿 ol) の THF (4.0 ml) 懸濁液に 水素化ナトリウム (純度 60%油性, 61.2 mg, 1.53腿 ol) を室温で加え、 15分間 攪拌後、 塩化 4- (卜ピペリジル)ベンゼンスルホ二ルの粗物質 (純度 25 , 0.70 g, 0.673讓 ol)の THF(1 ml)溶液を 20分間かけて加え、 さらに 1時間攪拌した。 反 応液にメタノール(1 ml) 、 IN塩酸 (1.53 ml) および水(20 ml)を加え、 酢酸ェ チル (30ml)で抽出した。 有機層を 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫 酸ナトリウムで乾燥した。 濃縮乾固後、 残淹をメタノールで希釈した。 析出物を 濾取し、 メタノールで洗浄後、 乾燥して表記化合物 (281 mg, 0.521 mmol, 85.2¾) を紫色粉末として得た。 To a suspension of the compound obtained in Example 6-23 (193 mg, 0.612 t) in THF (4.0 ml) was added sodium hydride (purity 60% oily, 61.2 mg, 1.53 t) at room temperature, and the mixture was added for 15 minutes. After stirring, a solution of a crude substance of 4- (topiperidyl) benzenesulfonyl chloride (purity: 25, 0.70 g, 0.673 mL) in THF (1 ml) was added over 20 minutes, and the mixture was further stirred for 1 hour. Methanol (1 ml), IN hydrochloric acid (1.53 ml) and water (20 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (30 ml). The organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, and then dried over sodium sulfate. After concentration to dryness, the remaining brew was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (281 mg, 0.521 mmol, 85.2%) as a purple powder.
Ή—NMR (300MHz, CDC13) : (51.54 (2H, m), 1.65 (6H, m), 1.73 (4H, m), 1.78 (3H, q, J= 0.9 Hz), 2.25 (3H, q, J= 0.6 Hz), 2.94 (2H, t, J= 7.2 Hz), 3.30 (4H, m), 4.45 (2H, dt, J= 47.4, 5.9 Hz), 6.86 (2H, d, J= 9.0 Hz), 7.76 (2H, d, J= 9.0 Hz), 9.62 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): (51.54 (2H, m), 1.65 (6H, m), 1.73 (4H, m), 1.78 (3H, q, J = 0.9 Hz), 2.25 (3H, q, J = 0.6 Hz), 2.94 (2H, t, J = 7.2 Hz), 3.30 (4H, m), 4.45 (2H, dt, J = 47.4, 5.9 Hz), 6.86 (2H, d, J = 9.0 Hz) , 7.76 (2H, d, J = 9.0 Hz), 9.62 (1H, brs).
元素分析値: C24H31N403S3Fとして計算値: C, 53.51; H, 5.80; N, 10.40; S, 17.86; F, 3.53 (%) 、 実測値: C, 53.48; H, 5.87; N, 10.28; S, 17.81; F, 3.45 (%) . mp. 170.0-171. o Elemental analysis: C 24 H 31 N 4 0 3 S 3 F Calculated: C, 53.51; H, 5.80 ; N, 10.40; S, 17.86; F, 3.53 (%), Found: C, 53.48; H , 5.87; N, 10.28; S, 17.81; F, 3.45 (%). Mp.170.0-171.o
実施例 7-124 Example 7-124
N- {5, 6 -ジメチル- 3-ォキソ -8- [(4, 4, 5, 5, 5-ペン夕フルォロペンチル)スルファニ ル]- 2 , 3-ジヒドロ- 1Η-イミダゾ [5 ,卜 c] [1, 4]チアジン - 1 -イリデン卜 4- α -ピペリ 実施例 6- 3で得た化合物 (217 mg, 0.560匪 ol) の THF (4.0 ml) 懸濁液に水素 化ナトリウム(純度 60% 油性, 56.0 mg, 1.40 mmol) を室温で加え、 45分間攪拌 後、塩ィヒ 4 -(1-ピペリジル)ベンゼンスルホ二ルの粗物質 (純度 25%, 0.64 g, 0.616 腿 ol)の THF(1 ml)溶液を 30分間かけて加え、 さらに 2時間攪拌した。 反応液に メタノール(1 ml) および 1N塩酸 (1.5 ml)を加え、 酢酸ェチル (30 ml)で希釈 し、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮 乾固後、 残渣をメタノールで希釈した。 析出物を濾取し、 メタノールで洗浄後、 乾燥して表記化合物(280 mg, 0.463腿 ol, 82.8%)を灰赤色粉末として得た。 N- {5,6-Dimethyl-3-oxo-8-[(4,4,5,5,5-pentafluoropentyl) sulfani L] -2,3-Dihydro-1Η-imidazo [5, tric] [1,4] thiazine-1-ylident-4-α-piperi The compound obtained in Example 6-3 (217 mg, 0.560 ol ) In THF (4.0 ml) was added sodium hydride (purity 60%, oily, 56.0 mg, 1.40 mmol) at room temperature, stirred for 45 minutes, and added with salt 4-(1-piperidyl) benzenesulfonyl. A solution of the crude substance (purity 25%, 0.64 g, 0.616 tmol) in THF (1 ml) was added over 30 minutes, and the mixture was further stirred for 2 hours. Methanol (1 ml) and 1N hydrochloric acid (1.5 ml) were added to the reaction solution, diluted with ethyl acetate (30 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (280 mg, 0.463 mol, 82.8%) as a gray-red powder.
^-NMR (300MHz, CDC13) : 51.65 (6H, m), 1.78 (3H, brs), 2.03 (2H, brq, J= 7.5 Hz), 2.16 (2H, m), 2.25 (3H, s), 3.00 (2H, t, J= 7.2 Hz), 3.33 (4H, m) , 6.86 (2H, d, J= 8.7 Hz), 7.75 (2H, d, J= 9.0 Hz), 9.65 (1H, brs). ^ -NMR (300MHz, CDC1 3) : 51.65 (6H, m), 1.78 (3H, brs), 2.03 (2H, brq, J = 7.5 Hz), 2.16 (2H, m), 2.25 (3H, s), 3.00 (2H, t, J = 7.2 Hz), 3.33 (4H, m), 6.86 (2H, d, J = 8.7 Hz), 7.75 (2H, d, J = 9.0 Hz), 9.65 (1H, brs).
元素分析値: C24H27N403S3F5として計算値: C, 47.20; H, 4.46; N, 9.17; S, 15.75; F, 15.56 (%) 、 実測値: C, 47.13; H, 4.32; N, 9.12; S, 15.85; F, 15.57 (%) · 即. 192.0- 194.0°C Elemental analysis: C 24 H 27 N 4 0 3 S 3 F 5 Calculated: C, 47.20; H, 4.46 ; N, 9.17; S, 15.75; F, 15.56 (%), Found: C, 47.13; H, 4.32; N, 9.12; S, 15.85; F, 15.57 (%) Immediate. 192.0-194.0 ° C
実施例 7-125 Example 7-125
メチル 2-[({5, 6-ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル) スルファニル ]_2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }ァ ミノ)スルホニル]ベンゾェ一ト Methyl 2-[({5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] _2,3-dihydrido-1H-imidazo [ 5, 1-c] [1,4] thiazine-1-ylidene} amino) sulfonyl] benzoate
実施例 7-121と同様にして合成した。 Synthesized in the same manner as in Examples 7-121.
テトラヒドロフラン-メ夕ノールから再結晶して融点 148-150°Cの暗赤色結晶を 得た。 The crystals were recrystallized from tetrahydrofuran-methanol to obtain dark red crystals having a melting point of 148-150 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3370, 3250, 3200, 2953, 1728, 1663, 1618, 1557 cm"1. Infrared absorption spectrum (IR) (in KBr): 3370, 3250, 3200, 2953, 1728, 1663, 1618, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 1.62-1.82 (4Η, m), 1.79 (3H, d, J=0.9 Hz), 1.94-2.13 (2H, m), 2.29 (3H, d, J=0.9 Hz), 2.95 (2H, t, J=7.2 Hz), 3.95 (3H, s), 7.56-7.62 (3H, m), 8.12-8.19 (1H, m), 9.87 (1H, bs). Ή-NMR (CDC1 3): δ 1.62-1.82 (4Η, m), 1.79 (3H, d, J = 0.9 Hz), 1.94-2.13 (2H, m), 2.29 (3H, d, J = 0.9 Hz) , 2.95 (2H, t, J = 7.2 Hz), 3.95 (3H, s), 7.56-7.62 (3H, m), 8.12-8.19 (1H, m), 9.87 (1H, bs).
元素分析値: C22H22N305S3F5として 計算値 (%) : C, 44.07; H, 3.70; N, 7.01; S, 16.04. Elementary analysis: as C 22 H 22 N 3 0 5 S 3 F 5 Calculated value (%): C, 44.07; H, 3.70; N, 7.01; S, 16.04.
実測値 (%) : C, 43.97; H, 3.69; N, 7.04; S, 16.20. Found (%): C, 43.97; H, 3.69; N, 7.04; S, 16.20.
実施例 7-126 Example 7-126
4-ブ口モ- N - {6-ェチル 8 - [ (5-フル才口ぺンチル)スルファニル] -5-メチル- 3-ォ, キソ- 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }ベンゼンス ルホンアミド 4-Bumo-N- {6-ethyl-8-[(5-full-aminopentyl) sulfanyl] -5-methyl-3-o, xo-2,3-dihydro-1H-imidazo [5,1 -c] [1,4] thiazine-1-ylidene} benzenesulfonamide
水素化ナトリウム (29.1 mg, 0.728 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6 - 27で得られた 1-アミノ- 6-ェチル -8-[(5 -フルォロぺ ンチル)スルファニル] - 5-メチル -3H-ィミダゾ [5, 1-c] [1,4]チアジン- 3 -才ン(120 mg, 0.364 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間か き混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (190 mg, 0.728 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 1時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(180 mg, 90%)を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 182- 184°Cの暗赤色結晶を得 ' た。 To a suspension of sodium hydride (29.1 mg, 0.728 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added 1-amino-6-ethyl-8-[(5- Fluorintyl) sulfanyl] -5-methyl-3H-imidazo [5,1-c] [1,4] thiazine-3-amine (120 mg, 0.364 mmol) in tetrahydrofuran (1.00 ml) and ice Stir for 5 minutes under cooling. To this was added a solution of 4-bromobenzenesulfonyl chloride (190 mg, 0.728 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (180 mg, 90%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 182-184 ° C.
IR (KBr) V: 3144, 2959, 2936, 2873, 1723, 1657, 1593, 1553 cm—1. IR (KBr) V: 3144, 2959, 2936, 2873, 1723, 1657, 1593, 1553 cm— 1 .
Ή-NMR (CDC13) : (51.09 (3H, t, J=7.8 Hz), 1.48-1.62 (2H, m), 1.64-1.84 (4H, m), 2.15 (2H, q, J=7.2 Hz), 2.27 (3H, s), 2.98 (2H, t, J=7.2 Hz), 4.46 (2H, dt, J=47.4, 5.7 Hz), 7.63 (2H, d, J=8.4 Hz), 7.83 (2H, d, J=8.7 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): (51.09 (3H, t, J = 7.8 Hz), 1.48-1.62 (2H, m), 1.64-1.84 (4H, m), 2.15 (2H, q, J = 7.2 Hz) , 2.27 (3H, s), 2.98 (2H, t, J = 7.2 Hz), 4.46 (2H, dt, J = 47.4, 5.7 Hz), 7.63 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.7 Hz), 9.60 (1H, bs).
実施例 7-127 Example 7-127
4 -クロ口- N- {6-ェチル -8- [ (5-フル才口ぺンチル)スルファニル] -5-メチル- 3-ォ キソ- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 1-ィリデン }ベンゼンス ルホンアミド 4-chloro-N- {6-ethyl-8-[(5-full-pentyl) sulfanyl] -5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene} benzenesulfonamide
水素化ナトリゥム(35.8 mg, 0.895 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 27で得られた 1-ァミノ- 6 -ェチル -8 - [(5-フルォロぺ ンチル)スルファニル] -5-メチル -3H -ィミダゾ [5, 1-c] [1,4]チアジン - 3 -オン(160 mg, 0.448 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間か き混ぜた。 これに 4-クロ口ベンゼンスルホニル クロリド (189 mg, 0.895腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 1時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (139 mg, 62¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 174-175 の暗赤色結晶を得た。 To a suspension of sodium hydride (35.8 mg, 0.895 mmol) (60% oily) in tetrahydrofuran (2.00 ml) was added 1-amino-6-ethyl-8-[(5- Fluorintyl) sulfanyl] -5-methyl-3H-imidazo [5,1-c] [1,4] thiazin-3-one (160 mg, 0.448 mmol) in tetrahydrofuran (1.00 ml) and ice-cooling 5 minutes below I mixed. To this was added a solution of benzenesulfonyl chloride (189 mg, 0.895 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (139 m g, 62¾). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 174-175.
IR (KBr) リ: 3135, 2963, 2936, 2872, 1728, 1657, 1615, 1557 cm"'. IR (KBr): 3135, 2963, 2936, 2872, 1728, 1657, 1615, 1557 cm "'.
Ή-NMR (CDC13) : (51.09 (3H, t, J=7.5 Hz), 1.48-1.62 (2H, m), 1.64-1.84 (4H, m), 2.15 (2H, q, J=7.5 Hz), 2.28 (3H, s), 2.98 (2H, t, J=7.5 Hz), 4.46 (2H, dt, J=47.4, 5.7 Hz), 7.46 (2H, d, J=9.0 Hz), 7.90 (2H, d, J=8.7 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): (51.09 (3H, t, J = 7.5 Hz), 1.48-1.62 (2H, m), 1.64-1.84 (4H, m), 2.15 (2H, q, J = 7.5 Hz) , 2.28 (3H, s), 2.98 (2H, t, J = 7.5 Hz), 4.46 (2H, dt, J = 47.4, 5.7 Hz), 7.46 (2H, d, J = 9.0 Hz), 7.90 (2H, d, J = 8.7 Hz), 9.61 (1H, bs).
元素分析値: C2。H23N303S3C1Fとして Elemental analysis: C 2. H 23 N 3 0 3 S 3 C1F
計算値 (¾) : C, 47.66; H, 4.60; N, 8.34; S, 19.08. Calculated value (¾): C, 47.66; H, 4.60; N, 8.34; S, 19.08.
実測値 : C, 47.53; H, 4.51; , 8.16; S, 18.99. Found: C, 47.53; H, 4.51;, 8.16; S, 18.99.
実施例 7-128 Example 7-128
4-ブロモ -N- {6-ブチル -8- [ (5-フルォ口ペンチル)スルファニル] -5-メチル -3-ォ キソ- 2,3-ジヒドロ- 1H-イミダゾ [5,l_c] [1, 4]チアジン- 1-ィリデン }ベンゼンス ルホンアミド 4-bromo-N- {6-butyl-8-[(5-fluoropentyl) sulfanyl] -5-methyl-3-oxo- 2,3-dihydro-1H-imidazo [5, l_c] [1, 4] thiazine-1-ylidene} benzenesulfonamide
水素化ナトリウム(72.7mg, 1.82mrao) (60%油性) のテトラヒドロフラン(6.00 ml) の懸濁液に、 実施例 6-28で得られた卜ァミノ- 6_ブチル -8- [(5-フルォロぺ ンチル)スルファニル ]-5-メチル -3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン(325 mg, 0.909匪 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5分間か き混ぜた。 これに 4_ブロモベンゼンスルホニル クロリド (474 mg, 1.82 imol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 さらに室温で 2時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(349 mg, 67¾)を暗赤色結晶として得た。 酢酸ェチル-ジェチルェ一テルから再結晶して融点 179- 181°Cの暗赤色結晶を得 た。 To a suspension of sodium hydride (72.7 mg, 1.82 mrao) (60% oil) in tetrahydrofuran (6.00 ml) was added the triamino-6_butyl-8-[(5-fluoro) obtained in Example 6-28.ぺ ntyl) sulfanyl] -5-methyl-3H-imidazo [5, tric] [1,4] thiazin-3-one (325 mg, 0.909 bandol) in tetrahydrofuran (2.00 ml), and add ice-cooling Stir for 5 minutes. To this was added a solution of 4_bromobenzenesulfonyl chloride (474 mg, 1.82 imol) in tetrahydrofuran (2.00 ml), and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (349 mg, 67¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-jetil ether to obtain dark red crystals having a melting point of 179-181 ° C.
IR (KBr) V: 3166, 2955, 2938, 2868, 1725, 1651, 1595, 1557 cm—1. IR (KBr) V: 3166, 2955, 2938, 2868, 1725, 1651, 1595, 1557 cm- 1 .
Ή-NMR (CDCI3) : (50.933 (3H, t, J=7.2Hz), 1.24-1.62 (6H, m), 1.64-1.84 (4H, m), 2.12 (2H, i, J=7.8 Hz), 2.27 (3H, s), 2.97 (2H, t, J=7.2 Hz), 4.45 (2H, dt, J=47.4, 5.7 Hz), 7.63 (2H, d, J=8.1 Hz), 7.83 (2H, d, J=8.7 Hz), 9.61 (1H, bs). Ή-NMR (CDCI3): (50.933 (3H, t, J = 7.2Hz), 1.24-1.62 (6H, m), 1.64-1.84 (4H, m), 2.12 (2H, i, J = 7.8 Hz), 2.27 (3H, s), 2.97 (2H, t, J = 7.2 Hz), 4.45 (2H, dt, J = 47.4, 5.7 Hz), 7.63 ( 2H, d, J = 8.1 Hz), 7.83 (2H, d, J = 8.7 Hz), 9.61 (1H, bs).
元素分析値: C22H27N303S3Brとして Elementary analysis: as C 22 H 27 N 3 0 3 S 3 Br
計算値 (¾) : C, 45.83; H, 4.72; N, 7.29; S, 16.68. Calculated value (¾): C, 45.83; H, 4.72; N, 7.29; S, 16.68.
実測値 (¾) : C, 45.80; H, 4.74; N, 7.16; S, 16.47. Found (¾): C, 45.80; H, 4.74; N, 7.16; S, 16.47.
実施例 7-129 Example 7-129
4_クロ口 -N- {6-ブチル -8- [ (5-フルォ口ぺンチル)スルファニル] -5-メチル -3 -ォ キソ- 2,3-ジヒドロ - 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン }ベンゼンス ルホンアミド 4_ black mouth -N- {6-butyl-8- [(5-fluoropentyl) sulfanyl] -5-methyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene} benzenesulfonamide
水素化ナトリウム (72.7fflg, 1.82mmo) (60油性)のテトラヒドロフラン(6.00 ml) の懸濁液に、 実施例 6 - 28で得られた 1 -ァミノ- 6 -ブチル- 8-[(5 -フルォロぺ ンチル)スルファニル] -5-メチル -3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 3-オン(325 mg, 0.909 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5分間か き混ぜた。 これに 4-クロ口ベンゼンスルホニル クロリド (384 mg, 1.82腿 ol) のテトラヒドロフラン (2.00 mi) 溶液を加え、 さらに室温で 2時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (302 mg, 63%) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 173-175°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (72.7fflg, 1.82mmo) (60 oily) in tetrahydrofuran (6.00ml) was added 1-amino-6-butyl-8-[(5-fluoro) obtained in Example 6-28. [Pentyl) sulfanyl] -5-methyl-3H-imidazo [5,1-c] [1,4] thiazin-3-one (325 mg, 0.909 mmol) in tetrahydrofuran (2.00 ml), and add ice-cooling Stir for 5 minutes. To this was added a solution of benzenesulfonyl chloride 4-chloro (384 mg, 1.82 mol) in tetrahydrofuran (2.00 mi), and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (302 mg, 63%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 173-175 ° C.
IR (KBr) v: 3166, 3090, 2955, 2938, 2863, 1725, 1653, 1597, 1557 cm"1. Ή-NMR (CDC13) : 50.933 (3H, t, J=6.9Hz), 1.25-1.62 (6H, m), 1.64-1.84 (4H, m), 2.12 (2H, ί, J=7.5 Hz), 2.27 (3H, s), 2.97 (2H, t, J=7.2 Hz), 4.45 (2H, dt, J=47.4, 5.7 Hz), 7.46 (2H, d, J=8.7 Hz), 7.91 (2H, d, J=8.4 Hz), 9.60 (1H, bs). IR (KBr) v:. 3166 , 3090, 2955, 2938, 2863, 1725, 1653, 1597, 1557 cm "1 Ή-NMR (CDC1 3): 50.933 (3H, t, J = 6.9Hz), 1.25-1.62 (6H, m), 1.64-1.84 (4H, m), 2.12 (2H, ί, J = 7.5 Hz), 2.27 (3H, s), 2.97 (2H, t, J = 7.2 Hz), 4.45 (2H, dt, J = 47.4, 5.7 Hz), 7.46 (2H, d, J = 8.7 Hz), 7.91 (2H, d, J = 8.4 Hz), 9.60 (1H, bs).
元素分析値: C22H27N303S3C1Fとして Elementary analysis: as C 22 H 27 N 3 0 3 S 3 C1F
計算値 (%) : C, 49.66; H, 5.11; , 7.90. Calculated (%): C, 49.66; H, 5.11;, 7.90.
実測値 (¾) : C, 49.57; H, 5.18; N, 7.77. Found (¾): C, 49.57; H, 5.18; N, 7.77.
実施例 7-130 Example 7-130
4 -クロ口- N- (5, 6-ジメチル- 3-ォキソ -8 - {[2 -(2, 2,2-トリフルォロエトキシ)ェチ ル]スルファニル }- 2,3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデ 水素化ナトリゥム (29.0 mg, 0.724 ramol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 26で得られた卜ァミノ- 5, 6 -ジメチル- 8- {[2 -(2, 2, 2 - トリフルォロエトキシ)ェチル]スルファ二ル} - 3H-ィミダゾ [5, 1-c] [1,4]チアジ ン -3-オン (128mg, 0.362 mmol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混ぜた。これに 4-クロロベンゼンスルホニル クロリド(153 mg, 0.724 mmol) のテトラヒドロフラン (1.00ml) 溶液を加え、 さらに室温で 1時間 かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (148 mg, 11%) を暗赤色 結晶として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 162- 164°Cの 暗赤色結晶を得た。 4-chloro-N- (5,6-dimethyl-3-oxo-8-{[2- (2,2,2-trifluoroethoxy) ethy L] Sulfanyl}-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene Hydrogenated sodium (29.0 mg, 0.724 ramol) (60% oil) in tetrahydrofuran (2.00 ml ), The triamino-5,6-dimethyl-8-{[2- (2,2,2-trifluoroethoxy) ethyl] sulfanyl obtained in Example 6-26}- A solution of 3H-imidazo [5,1-c] [1,4] thiazin-3-one (128 mg, 0.362 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-chlorobenzenesulfonyl chloride (153 mg, 0.724 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (148 mg, 11%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 162-164 ° C.
IR (KBr) V: 3254, 2930, 1759, 1744, 1663, 1622, 1559 cm"1. IR (KBr) V: 3254, 2930, 1759, 1744, 1663, 1622, 1559 cm " 1 .
Ή-NMR (CDC13) : 51.80 (3H, d, J=l.2 Hz), 2.27 (3H, d, J=1.2 Hz), 3.19 (2H, t, J=6.6 Hz), 3.84 (2H, t, J-6.0 Hz), 3.85 (2H, q, J=8.7 Hz), 7.46 (2H, dt, Ή-NMR (CDC1 3): 51.80 (3H, d, J = l.2 Hz), 2.27 (3H, d, J = 1.2 Hz), 3.19 (2H, t, J = 6.6 Hz), 3.84 (2H, t, J-6.0 Hz), 3.85 (2H, q, J = 8.7 Hz), 7.46 (2H, dt,
J=8.7, 2.1 Hz), 7.90 (2H, dt, J=9.0, 2.1 Hz), 9.62 (1H, bs). J = 8.7, 2.1 Hz), 7.90 (2H, dt, J = 9.0, 2.1 Hz), 9.62 (1H, bs).
元素分析値: C18H17N304S3C1F3として Elementary analysis: as C 18 H 17 N 3 0 4 S 3 C1F 3
計算値 (%) : C, 40.95; H, 3.25; N, 7.96; S, 18.22. Calculated (%): C, 40.95; H, 3.25; N, 7.96; S, 18.22.
実測値 (%) : C, 41.15; H, 3.46; N, 7.73; S, 18.16. Found (%): C, 41.15; H, 3.46; N, 7.73; S, 18.16.
実施例 7-131 Example 7-131
4_ブロモ - N -(5, 6 -ジメチル -3-ォキソ -8- { [5- (2, 2, 2-トリフルォロェトキシ)ペン チル]スルファニル }- 2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-イリデ 水素化ナトリウム(40.5mg, 1.01腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6 - 29で得られた卜アミノ- 5, 6 -ジメチル -8-{[5 -(2,2, 2- トリフルォロェトキシ)ペンチル]スルファニル }-3H-イミダゾ [5, 1-c] [1,4]チア ジン- 3-オン (200 mg, 0.506 ιπιοΐ) のテトラヒドロフラン (1.00 ml) 溶液を加 え、氷冷下 5分間かき混ぜた。これに 4-ブロモベンゼンスルホニル クロリド(264 mg, 1.01 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 19 時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を 水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (220 mg, 71¾) を 暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 172-174°Cの暗赤色結晶を得た。 4_bromo-N- (5,6-dimethyl-3-oxo-8-{[5- (2,2,2-trifluoroethoxy) pentyl] sulfanyl} -2,3-dihydro-1H-imidazo Example 6-29 [5, 1-c] [1,4] Thiazine-1-ylide A suspension of sodium hydride (40.5 mg, 1.01 mol) (60% oil) in tetrahydrofuran (2.00 ml) was prepared. -Amino-5,6-dimethyl-8-{[5- (2,2,2-trifluoroethoxy) pentyl] sulfanyl} -3H-imidazo [5,1-c] [1,4 ] A solution of thiazin-3-one (200 mg, 0.506 ιπιοΐ) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred for 5 minutes under ice-cooling. (1.00 ml) solution. Stir for hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (220 mg, 71¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 172-174 ° C.
IR (KBr) V: 3227, 3086, 2930, 2868, 1759, 1665, 1622, 1559 cm"1. IR (KBr) V: 3227, 3086, 2930, 2868, 1759, 1665, 1622, 1559 cm " 1 .
Ή-NMR (CDC13) : 61.44-1.82 (6H, m), 1.79 (3H, d, J=0.6Hz), 2.27 (3H, d, J=0.6 Hz), 2.96 (2H, t, J=7.5 Hz), 3.61 (2H, t, J=6.0Hz), 3.81 (2H, q, J=8.7 Hz), 7.63 (2H, d, J=8.7 Hz), 7.83 (2H, d, J=8.7 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): 61.44-1.82 (6H, m), 1.79 (3H, d, J = 0.6Hz), 2.27 (3H, d, J = 0.6 Hz), 2.96 (2H, t, J = 7.5 Hz), 3.61 (2H, t, J = 6.0Hz), 3.81 (2H, q, J = 8.7 Hz), 7.63 (2H, d, J = 8.7 Hz), 7.83 (2H, d, J = 8.7 Hz) , 9.60 (1H, bs).
元素分析値: C21H24N304S3BrF3して Elemental analysis: C 21 H 24 N 3 0 4 S 3 BrF 3 to
計算値 (%) : C, 40.98; H, 3.93; N, 6.83; S, 15.63; F, 9.26. Calculated (%): C, 40.98; H, 3.93; N, 6.83; S, 15.63; F, 9.26.
実測値 (%) : C, 41.12; H, 3.71; N, 6.92; S, 15.73; F, 8.92. Found (%): C, 41.12; H, 3.71; N, 6.92; S, 15.73; F, 8.92.
実施例 7-132 Example 7-132
4-ク口口- N- (5, 6 -ジメチル -3-ォキソ -8- { [5- (2, 2, 2 -トリフルォロエトキシ)ペン チル]スルファニル }-2, 3 -ジヒドロ- II-トイミダゾ [5, 1- c] [1, 4]チアジン- 1-ィリデ 水素化ナトリウム(36.9mg, 0.923 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 29で得られた卜ァミノ- 5, 6-ジメチル- 8- { [5- (2, 2, 2 - トリフルォロェトキシ)ペンチル]スルファ二ル}臺ィミダゾ [5, 1-c] [1,4]チア ジン- 3-オン (183 mg, 0.462 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加 え、氷冷下 5分間かき混ぜた。これに 4-クロロベンゼンスルホニル クロリド(195 mg, 0.923 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 19時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出 液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (108 mg, 41%) を合成した。酢酸ェチル-ジェチルエーテルから再結晶して融点 155-157 の暗赤 色結晶を得た。 4- ク 口 口 -N- (5,6-dimethyl-3-oxo-8-{[5- (2,2,2-trifluoroethoxy) pentyl] sulfanyl} -2,3-dihydro-II Example 6- Toimidazo [5,1-c] [1,4] thiazine-1-ylide A suspension of sodium hydride (36.9 mg, 0.923 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was prepared. Triamino-5,6-dimethyl-8 {{[5- (2,2,2-trifluoroethyl) pentyl] sulfanyl] obtained in 29} imidazo [5, 1-c] [1, 4] A solution of thiazin-3-one (183 mg, 0.462 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice-cooling for 5 minutes, followed by 4-chlorobenzenesulfonyl chloride (195 mg, 0.923 mmol) in tetrahydrofuran. (1.00 ml) was added, further stirred at room temperature for 19 hours. the reaction mixture was extracted with acetic acid Echiru by adding 1N hydrochloric acid. the extract was washed with water, dried (MgSO 4), the solvent It was evaporated under pressure to synthesize the title compound (108 mg, 41%) acetic acid Echiru -. To give a dark red crystals, mp 155-157 recrystallized from Jefferies chill ether.
IR (KBr) V: 3235, 2940, 1759, 1663, 1622, 1559 cm— IR (KBr) V: 3235, 2940, 1759, 1663, 1622, 1559 cm—
Ή-NMR (CDC13) : 51.40-1.84 (6H, m), 1.79 (3H, d, J=0.6 Hz), 2.27 (3H, d, J=1.0 Hz), 2.96 (2H, t, J=7.8Hz), 3.61 (2H, t, J=5.8Hz), 3.81 (2H, q, J=8.8 Hz), 7.46 (2H, d, J=8.4 Hz), 7.90 (2H, d, J=8.4 Hz), 9.59 (1H, bs). 実施例 7-133 Ή-NMR (CDC1 3): 51.40-1.84 (6H, m), 1.79 (3H, d, J = 0.6 Hz), 2.27 (3H, d, J = 1.0 Hz), 2.96 (2H, t, J = 7.8 Hz), 3.61 (2H, t, J = 5.8Hz), 3.81 (2H, q, J = 8.8 Hz), 7.46 (2H, d, J = 8.4 Hz), 7.90 (2H, d, J = 8.4 Hz) , 9.59 (1H, bs). Example 7-133
4 -ブロモ -N -(5, 6-ジメチル -3-ォキソ -8- { [4- (2, 2, 2-トリフルォロエトキシ)プチ ル]スルファニル }- 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデ 水素化ナトリウム(37.8 mg, 0.944腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-30で得られた卜ァミノ- 5, 6 -ジメチル -8- {[4 -(2,2,2- トリフルォロェ卜キシ)プチル]スルファニル }-3H-ィミダゾ [5, 1-c] [1,4]チアジ ン- 3 -オン (180mg, 0.472匪 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混ぜた。これに 4-ブロモベンゼンスルホニル クロリド(246 mg, 0.944 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 18時 間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水 洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (196 mg, 69%) を暗 赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 154_156°Cの暗赤色結晶を得た。 4-bromo-N- (5,6-dimethyl-3-oxo-8-{[4- (2,2,2-trifluoroethoxy) butyl] sulfanyl} -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylide A suspension of sodium hydride (37.8 mg, 0.944 tmol) (60% oil) in tetrahydrofuran (2.00 ml) was prepared in Example 6-30. -5,6-dimethyl-8- {[4- (2,2,2-trifluoroethoxy) butyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] A solution of thiazin-3-one (180 mg, 0.472 marl) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice-cooling for 5 minutes, and mixed with 4-bromobenzenesulfonyl chloride (246 mg, 0.944 mmol) in tetrahydrofuran (1.00 ml). ml) was added and further extracted with acetic Echiru by adding 1N hydrochloric acid. the reaction mixture was stirred for 18 hours at room temperature. the extract was water washed, dried (MgSO 4), of the solvent under reduced pressure And removed by to give the title compound (196 mg, 69%) as a dark red crystalline acetate Echiru -. Jechirue was recrystallized from a ether to give a dark red crystals, mp 154_156 ° C.
IR (KBr) v: 3183, 3063, 2934, 1757, 1663, 1622, 1559 cm—1. IR (KBr) v: 3183, 3063, 2934, 1757, 1663, 1622, 1559 cm— 1 .
Ή-NMR (CDC13) : δ 1.64-1.90 (4Η, m), 1.79 (3H, s), 2.27 (3H, s), 2.99 (2H, t, J=7.0 Hz) , 3.63 (2H, t, J=5.8 Hz), 3.81 (2H, q, J=8.8 Hz), 7.63 (2H, d, J=8.8 Hz), 7.83 (2H, d, J=8.8 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): δ 1.64-1.90 (4Η, m), 1.79 (3H, s), 2.27 (3H, s), 2.99 (2H, t, J = 7.0 Hz), 3.63 (2H, t, J = 5.8 Hz), 3.81 (2H, q, J = 8.8 Hz), 7.63 (2H, d, J = 8.8 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.60 (1H, bs).
元素分析値: C2。H21N304S3BrF3して Elemental analysis: C 2. H 21 N 3 0 4 S 3 BrF 3 to
計算値 (%) : C, 40.00; H, 3.52; N, 7.00; S, 16.02. Calculated value (%): C, 40.00; H, 3.52; N, 7.00; S, 16.02.
実測値 (%) : C, 40.13; H, 3.53; N, 7.01; S, 16.01. Found (%): C, 40.13; H, 3.53; N, 7.01; S, 16.01.
実施例 7-134 Example 7-134
4 -クロ口- N -(5, 6-ジメチル- 3-ォキソ -8- {[4- (2, 2, 2-トリフルォロェトキシ)ブチ ル]スルファニル卜 2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1 -ィリデ 水素化ナトリゥム(29.8 mg, 0.74 匪 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-30で得られた卜ァミノ -5, 6 -ジメチル -8- {[4 -(2, 2, 2- トリフルォロエトキシ)プチル]スルファニル }-3H-イミダゾ [5,卜 c] [1,4]チアジ ン- 3-オン (141 mg, 0.372腿 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混ぜた。これに 4-ク口口ベンゼンスルホニル クロリド(157 mg, 0.744匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 18時 間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水 洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (38.0 mg, 18¾) を合 成した。酢酸ェチル-ジェチルエーテルから再結晶して融点 131-133°Cの暗赤色結 晶を得た。 4-chloro-N- (5,6-dimethyl-3-oxo-8-{[4- (2,2,2-trifluoroethoxy) butyl] sulfanilto 2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylide sodium hydride (29.8 mg, 0.74 ol) (60% oil) in tetrahydrofuran (2.00 ml) was added to a suspension of Example 6-30. -5,6-Dimethyl-8-{[4- (2,2,2-trifluoroethoxy) butyl] sulfanyl} -3H-imidazo [5, tric] [1,4] A solution of thiazin-3-one (141 mg, 0.372 tmol) in tetrahydrofuran (1.00 ml) was added. The mixture was stirred for 5 minutes under ice cooling. To this was added a solution of benzenesulfonyl chloride (157 mg, 0.744 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 18 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was water washed, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (38.0 mg, 18¾) was form if the. The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals with a melting point of 131-133 ° C.
IR (KBr) リ: 3229, 3092, 2932, 2876, 1759, 1663, 1622, 1559 cm"1. IR (KBr): 3229, 3092, 2932, 2876, 1759, 1663, 1622, 1559 cm " 1 .
ΐ-NMR (CDC13) : 51.62-1.90 (4H, m), 1.79 (3H, d, J=1.0 Hz), 2.27 (3H, d, J=0.6 Hz), 2.99 (2H, t, J=7.0 Hz), 3.63 (2H, t, J=5.8Hz), 3.81 (2H, q, J-8.8 Hz), 7.46 (2H, d, J-8.6 Hz), 7.90 (2H, d, J=8. Hz), 9.59 (1H, bs). ΐ-NMR (CDC1 3): 51.62-1.90 (4H, m), 1.79 (3H, d, J = 1.0 Hz), 2.27 (3H, d, J = 0.6 Hz), 2.99 (2H, t, J = 7.0 Hz), 3.63 (2H, t, J = 5.8Hz), 3.81 (2H, q, J-8.8 Hz), 7.46 (2H, d, J-8.6 Hz), 7.90 (2H, d, J = 8.Hz) ), 9.59 (1H, bs).
元素分析値: C2QH21N304S3C1F3して Elemental analysis: C 2Q H 21 N 3 0 4 S 3 C1F 3 to
計算値 (¾) : C, 43.20; H, 3.81; N, 7.56; S, 17.30. Calculated value (¾): C, 43.20; H, 3.81; N, 7.56; S, 17.30.
実測値 (¾) : C, 43.19; H, 3.51; N, 7.36; S, 17.27. Found (¾): C, 43.19; H, 3.51; N, 7.36; S, 17.27.
実施例 7-135 Example 7-135
4-ブロモ -N- {8- [(4, 4-ジフルォロシクロへキシル) スルファニル ]-5, 6-ジメチル - 3-ォキソ -2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン -1-ィリデン }べンゼ ンスルホンアミド 4-bromo-N- {8-[(4,4-difluorocyclohexyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene} benzenesulfonamide
実施例 6- 31で得た化合物(85 mg, 0.246 mmol) の THF (2.5 ml) 懸濁液に水素 化ナトリウムの粉末 (純度 60%油性, 24.6 mg, 0.615 mmol) を室温で加え、 30 分間攪捽後、塩ィ匕 4_ブロモベンゼンスルホニル (75. '5 mg, 0.295腿 ol) を加え、 さらに 50分間攪拌した。 反応液にメタノール (1 ml) および IN塩酸 (0.7 ml) を加え、酢酸ェチル (25 ml)で抽出した。有機層を水 および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣をメタノールで希釈した。 析出物 を濾取し、メタノールで洗浄後、乾燥して表記化合物(134 mg, 0.237腿 ol, 96.5%) を紫色の粉末として得た。 To a suspension of the compound obtained in Example 6-31 (85 mg, 0.246 mmol) in THF (2.5 ml) was added sodium hydride powder (purity 60%, 24.6 mg, 0.615 mmol) at room temperature for 30 min. After the stirring, Shiojiri 4_bromobenzenesulfonyl (75. '5 mg, 0.295 t ol) was added, and the mixture was further stirred for 50 minutes. Methanol (1 ml) and IN hydrochloric acid (0.7 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (25 ml). The organic layer was washed with water and saturated saline, and then dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (134 mg, 0.237 mol, 96.5%) as a purple powder.
Ή-NMR (300MHz, CDC13) : 31.81 (3H, s), 1.83 (4H, m), 2.13 (4H, brd, J= 9.6 Hz), 2.27 (3H, brs), 3.49 (1H, m), 7.64 (2H, dt, J= 8.4, 1.5 Hz), 7.83 (2H, dt, J= 8.1, 1.2 Hz), 9.64 (1H, brs)'. Ή-NMR (300MHz, CDC1 3 ): 31.81 (3H, s), 1.83 (4H, m), 2.13 (4H, brd, J = 9.6 Hz), 2.27 (3H, brs), 3.49 (1H, m), 7.64 (2H, dt, J = 8.4, 1.5 Hz), 7.83 (2H, dt, J = 8.1, 1.2 Hz), 9.64 (1H, brs) '.
元素分析値: C2。H2。N303S3BrF2として計算値: C, 42.55; H, 3.57; N, 7.44; S, 17.04 (%) 、 実測値: C, 42.72; H, 3.57; N, 7.42; S, 17.17 (%) . 即. 194.0-195. O Elemental analysis: C 2. H 2. Calculated for N 3 0 3 S 3 BrF 2 : C, 42.55; H, 3.57; N, 7.44; S, 17.04 (%), Found: C, 42.72; H, 3.57; N, 7.42; S, 17.17 (%). Immediate. 194.0-195. O
実施例 7-136 Example 7-136
4 -ブ口モ- N - (8- {[ (4, 4-ジフルォロシクロへキシル)メチル]スルファ二ル} -5, 6 - ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデ 実施例 6-32で得た化合物の粗物質 (純度 80%, 123 mg, 0.278 mmol)の THF (2.5 ml) 懸濁液に水素化ナトリウムの粉末 (純度 60%油性, 33mg, 0.834腿 ol) を室温で加え、 10分間攪拌後、 塩化 4-ブロモベンゼンスルホニル (92mg, 0.361 mmol) を加え、 さらに 60分間攪拌した。 反応液にメタノール (1ml) および 1N 塩酸 (1.0ml)を加え、メタノール一水 (1:1, 5ml)で希釈し、 30分間攪拌した。 析出物を濾取し、 メタノール一水 (1:1)およびメタノールで洗浄後、 乾燥して表 記化合物(118 mg, 0.204 mmol, 73.4%)を赤色の粉末として得た。 4-Bumo-N- (8-{[(4,4-difluorocyclohexyl) methyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5 [1,4] thiazine-1-ylide Sodium hydride was added to a suspension of the crude compound (purity 80%, 123 mg, 0.278 mmol) of the compound obtained in Example 6-32 in THF (2.5 ml). Was added at room temperature, stirred for 10 minutes, added with 4-bromobenzenesulfonyl chloride (92 mg, 0.361 mmol), and further stirred for 60 minutes. 1 ml) and 1N hydrochloric acid (1.0 ml), diluted with methanol-water (1: 1, 5 ml) and stirred for 30 minutes The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol After drying, the title compound (118 mg, 0.204 mmol, 73.4%) was obtained as a red powder.
¾-NMR (200MHz, CDC13) : 61.34 (2H, brq, J= 13.6 Hz), 1.60 (2H, m), 1.71 (2H, m), 1.80 (3H, q, J= 1.2 Hz), 1.91 (2H, brd, J= 13.4 Hz), 2.12 (2H, m), 2.27 (3H, q, J= 1.0 Hz), 2.87 (2H, d, J= 6.6 Hz), 7.62 (2H, dt, J= 8.8, 2.0 Hz), 7.83 (2H, d, J= 8.8 Hz), 9.63 (1H, brs). ¾-NMR (200MHz, CDC1 3 ): 61.34 (2H, brq, J = 13.6 Hz), 1.60 (2H, m), 1.71 (2H, m), 1.80 (3H, q, J = 1.2 Hz), 1.91 ( 2H, brd, J = 13.4 Hz), 2.12 (2H, m), 2.27 (3H, q, J = 1.0 Hz), 2.87 (2H, d, J = 6.6 Hz), 7.62 (2H, dt, J = 8.8 , 2.0 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.63 (1H, brs).
元素分析値: C21H22N303S3BrF2として計算値: C, 43.60; H, 3.83; N, 7.26; S, 16.63; Br, 13.81; F, 6.57 (%) 、 実測値: C, 43.82; H, 3.87; N, 7.30; S, 16.66; Br, 15.80; F, 5.91 (%) . Elemental analysis: Calculated for C 21 H 22 N 3 0 3 S 3 BrF 2 : C, 43.60; H, 3.83; N, 7.26; S, 16.63; Br, 13.81; F, 6.57 (%), found: C, 43.82; H, 3.87; N, 7.30; S, 16.66; Br, 15.80; F, 5.91 (%).
mp. 207.5-208.0°C mp. 207.5-208.0 ° C
実施例 7-137 Example 7-137
3-ブロモ -N-{5, 6-ジメチル -3-ォキソ -8- [(5-フルォロペンチル) スルファニル] - 2,3-ジヒドロ- 1H-イミダゾ [5,1-c] [1, 4]チアジン- 1_イリデン }ベンゼンスルホン アミド 3-bromo-N- {5,6-dimethyl-3-oxo-8-[(5-fluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1_ylidene} benzenesulfonamide
水素化ナトリウム(50.7mg, 1.27 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6-ジメチル -8-{ [2- (5-フル ォロぺンチルォキシ)ェチル]スルファ二ル} 3H-ィミダゾ [5, 1 - c] [1 , 4]チアジン -3 -オン (200 mg, 0.634匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。これに 3-ブロモ ンゼンスルホニル クロリド(324 mg, 1.27 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 1時間 かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (329 mg, 97¾) を暗赤色 結晶として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 144- U6°Cの 暗赤色結晶を得た。 To a suspension of sodium hydride (50.7 mg, 1.27 mmol) (60% oily) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8-{[2 -(5-Fluoroyloxy) ethyl] sulfanyl} 3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.634 bandol ol) in tetrahydrofuran (1.00 ml) Add the solution, The mixture was stirred for 5 minutes under ice cooling. To this was added a solution of 3-bromobenzenesulfonyl chloride (324 mg, 1.27 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (329 mg, 97¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave a dark red crystal with a melting point of 144-U6 ° C.
IR (KBr) V: 3225, 3065, 2961, 2932, 2859, 1755, 1663, 1618, 1559 cm'1. Ή-NMR (CDC13) : δ 1.48-1.62 (2Η, m), 1.62-1.84 (4H, m), 1.80 (3H, d, J=1.2 Hz), 2.28 (3H, d, J=1.2 Hz), 2.99 (2H, t, J=7.2 Hz), 4.46 (2H, dt, J=47.1, 6.0 Hz), 7.37 (1H, t, J=7.8 Hz), 7.65-7.70 (1H, m), 7.8-7.92 (1H, m), 8.14 (1H, t, J=1.8 Hz), 9.58 (1H, bs). IR (KBr) V:. 3225 , 3065, 2961, 2932, 2859, 1755, 1663, 1618, 1559 cm '1 Ή-NMR (CDC1 3): δ 1.48-1.62 (2Η, m), 1.62-1.84 (4H , m), 1.80 (3H, d, J = 1.2 Hz), 2.28 (3H, d, J = 1.2 Hz), 2.99 (2H, t, J = 7.2 Hz), 4.46 (2H, dt, J = 47.1, 6.0 Hz), 7.37 (1H, t, J = 7.8 Hz), 7.65-7.70 (1H, m), 7.8-7.92 (1H, m), 8.14 (1H, t, J = 1.8 Hz), 9.58 (1H, bs).
元素分析値: C19H21N303S3BrFとして Elementary analysis: as C 19 H 21 N 3 0 3 S 3 BrF
計算値 (¾) : C, 42.70; H, 3.96; N, 7.86; S, 18.00; Br, 14.95. Calculated (¾): C, 42.70; H, 3.96; N, 7.86; S, 18.00; Br, 14.95.
実測値 (%) : C, 43.10; H, 3.84; N, 7.89; S, 17.86; Br, 14.72. Found (%): C, 43.10; H, 3.84; N, 7.89; S, 17.86; Br, 14.72.
実施例 7-138 " Example 7-138 "
2-ブロモ - N_{5,6-ジメチル -3 ォキソ _8-[(5_フルォロペンチル) スルファニ ル] -2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, ]チアジン- 1 -ィリデン }ベンゼンスル ホンアミド 2-Bromo-N_ {5,6-dimethyl-3oxo_8-[(5_fluoropentyl) sulfanyl] -2,3-dihydrido-1H-imidazo [5,1-c] [1,] thiazine-1 -Ylidene} benzenesulfonamide
水素化ナトリウム(50.7mg, 1.27 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6-ジメチル- 8-{[2-(5-フル ォロペンチルォキシ)ェチル]スルファニル }- 3H-ィミダゾ [5, 1-c] [1, 4〕チアジン -3 -オン (200 mg, 0.63 腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。これに 2-ブロモベンゼンスルホニル クロリド(331 mg, 1.27 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 1時間 かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (258 mg, 76¾) を暗赤色 結晶として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 182- 184 の 暗赤色結晶を得た。 To a suspension of sodium hydride (50.7 mg, 1.27 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8-{[2 -(5-Fluoropentyloxy) ethyl] sulfanyl}-3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.63 thigh) of tetrahydrofuran (1.00 ml) The solution was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 2-bromobenzenesulfonyl chloride (331 mg, 1.27 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (258 mg, 76¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether afforded dark red crystals, mp 182-184.
IR (KBr) レ : 3260, 2959, 2938, 2859, 1744, 1725, 1661, 1609, 1553 cm"1. ¾-NMR (CDCI3) : δ 1.42-1.60 (2H, m), 1.60-1.78 (4H, m), 1.80 (3H, d, J=0.9 Hz), 2.30 (3H, d, J=0.6 Hz), 2.94 (2H, i, J=7.5 Hz), 4.43 (2H, dt, J=47.4, 6.0 Hz), 7.46 (1H, dt, J=7.5, 1.2 Hz), 7.72 (1H, dd, J=7.5, 1.2 Hz), 8.23 (2H, dd, J=7.8, 1.8 Hz), 9.72 (1H, bs). IR (KBr) Re: 3260, 2959, 2938, 2859, 1744, 1725, 1661, 1609, 1553 cm " 1 . ¾-NMR (CDCI3): δ 1.42-1.60 (2H, m), 1.60-1.78 (4H, m), 1.80 (3H, d, J = 0.9 Hz), 2.30 (3H, d, J = 0.6 Hz), 2.94 (2H, i, J = 7.5 Hz), 4.43 (2H, dt, J = 47.4, 6.0 Hz), 7.46 (1H, dt, J = 7.5, 1.2 Hz), 7.72 (1H, dd, J = 7.5, 1.2 Hz), 8.23 (2H, dd, J = 7.8, 1.8 Hz), 9.72 (1H, bs).
実施例 7-139 Example 7-139
4-ブロモ- N - {5, 6-ジメチル -3-ォキソ- 8- [ (5-フルォ口ぺンチル) スルファニル] - 2,3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン - 1 -ィリデン }- 2-フルォロベン ゼンスルホンアミド ' 4-bromo-N- {5,6-dimethyl-3-oxo-8-[(5-fluoroopentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1, 4] Thiazine-1-ylidene}-2-fluorobenzensulfonamide '
水素化ナトリウム(50.7mg, 1.27腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜アミノ- 5, 6-ジメチル -8-{[2-(5-フル ォロペンチルォキシ)ェチル]スルファニル }_3H -ィミダゾ [5, 1-c] [1,4]チアジン - 3 -オン (200 mg, 0.634 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 4-プロモ- 2-フルォロベンゼンスルホニル クロ リ ド (347 mg, 1.27腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さら に室温で 1時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(176 mg, 50¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 158-160°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (50.7 mg, 1.27 t ol) (60% oil) in tetrahydrofuran (2.00 ml) was added triamino-5,6-dimethyl-8-{[obtained in Example 6-23. 2- (5-Fluoropentyloxy) ethyl] sulfanyl} _3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.634 mmol) in tetrahydrofuran (1.00 ml) And stirred under ice cooling for 5 minutes. To this was added a solution of 4-promo-2-fluorobenzenesulfonyl chloride (347 mg, 1.27 mol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (176 mg, 50¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 158-160 ° C.
IR (KBr) リ: 3241, 3096, 2936, 2863, 1728, 1661, 1609, 1590, 1553 cm—1. Ή-N R (CDC") : δ 1.42-1.88 (6Η, m), 1.81 (3H, s), 2.30 (3H, d, J=0.6 Hz), 2.95 (2H, t, J=7.4 Hz), 4.44 (2H, dt, J=47.2, 6.0 Hz), 7.37 (1H, dd, J=9.0, 1.6 Hz), 7.42 (1H, d, J=8.6 Hz), 7.86 (1H, t, J=8.0 Hz), 9.69 (1H, bs). 元素分析値: C19 。N303S3BrF2として IR (KBr) Re: 3241, 3096, 2936, 2863, 1728, 1661, 1609, 1590, 1553 cm— 1. Ή-NR (CDC "): δ 1.42-1.88 (6Η, m), 1.81 (3H, s ), 2.30 (3H, d, J = 0.6 Hz), 2.95 (2H, t, J = 7.4 Hz), 4.44 (2H, dt, J = 47.2, 6.0 Hz), 7.37 (1H, dd, J = 9.0, 1.6 Hz), 7.42 (1H, d, J = 8.6 Hz), 7.86 (1H, t, J = 8.0 Hz), 9.69 (1H, bs). Elemental analysis: C 19 .N 3 0 3 S 3 BrF 2 As
計算値 (¾) : C, 41.31; H, 3.65; N, 7.61; S, 17.41; Br, 14.46. Calculated (¾): C, 41.31; H, 3.65; N, 7.61; S, 17.41; Br, 14.46.
実測値 (¾) : C, 41.41; H, 3.91; , 7.83; S, 17.50; Br, 14.60. Found (¾): C, 41.41; H, 3.91;, 7.83; S, 17.50; Br, 14.60.
実施例 7-140 Example 7-140
5 -ブロモ- N - {5, 6-ジメチル- 3-ォキソ -8- [(5 -フルォロペンチル) スルファニル] - 2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1, 4]チアジン -1-ィリデン卜 2-メトキシベン ゼンスルホンアミド 水素化ナトリウム(50.7mg, 1.27腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例.6 - 23で得られた卜ァミノ- 5,6-ジメチル- 8- {[2- (5-フル ォロペンチルォキシ)ェチル]スルファ二ル} - 3H-ィミダゾ [5 , 1-c] [1 , 4]チアジン -3-オン (200 mg, 0.634 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 5 -プロモ- 2-メトキシベンゼンスルホニル ク口 リド (369 mg, 1.27匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さら に室温で 1時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(289 mg, 81%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 173- 175 の暗赤色結晶を得た。 5-bromo-N- {5,6-dimethyl-3-oxo-8-[(5-fluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1-yridident 2-methoxybenzenesulfonamide To a suspension of sodium hydride (50.7 mg, 1.27 tmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8- {obtained in Examples 6 to 23. [2- (5-Fluoropentyloxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.634 mmol) in tetrahydrofuran ( 1.00 ml) solution and stirred under ice cooling for 5 minutes. To this was added a solution of 5-bromo-2-methoxybenzenesulfonyl chloride (369 mg, 1.27 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (289 mg, 81%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 173-175.
I (KBr) V: 3243, 2942, 2849, 1719, 1661, 1613, 1553 cm一1. I (KBr) V: 3243, 2942, 2849, 1719, 1661, 1613, 1553 cm- 1 .
Ή-NMR (CDC13) : 51.40-1.85 (6H, m), 1.81 (3H, d, J=1.0 Hz), 2.31 (3H, d, J=1.2 Hz), 2.93 (2H, t, J=7.6 Hz), 3.88 (3H, s), 4.43 (2H, dt, J=47.2, 6.0 Hz), 6.87 (1H, d, J=8.8 Hz), 7.60 (1H, dd, J=8.8, 2. Hz), 8.17 (1H, d, J=2.6 Hz), 9.80 (1H, bs). Ή-NMR (CDC1 3): 51.40-1.85 (6H, m), 1.81 (3H, d, J = 1.0 Hz), 2.31 (3H, d, J = 1.2 Hz), 2.93 (2H, t, J = 7.6 Hz), 3.88 (3H, s), 4.43 (2H, dt, J = 47.2, 6.0 Hz), 6.87 (1H, d, J = 8.8 Hz), 7.60 (1H, dd, J = 8.8, 2. Hz) , 8.17 (1H, d, J = 2.6 Hz), 9.80 (1H, bs).
元素分析値: C2。H23N304S3BrFとして Elemental analysis: C 2. As H 23 N 3 0 4 S 3 BrF
計算値 (%) : C, 42.55; H, 4.11; , 7.44; S, 17.04; Br, 14.15. Calculated (%): C, 42.55; H, 4.11;, 7.44; S, 17.04; Br, 14.15.
実測値 (¾) : C, 42.68; H, 4.20; N, 7.57; S, 17.03; Br, 14.35. Found (¾): C, 42.68; H, 4.20; N, 7.57; S, 17.03; Br, 14.35.
実施例 7-141 Example 7-141
ェチル 5_{[1- ({[4- (ァセチルァミノ)フエニル]スルホ二ル}ィミノ) - 5, 6 -ジメ チル- 3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5,1 - c] [1, 4]チアジン- 8-ィル]スルフ ァニル }ペンタノェ一ト 5 _ {[1-({[4- (Acetylamino) phenyl] sulfonyl} imino)-5, 6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazin-8-yl] sulfanyl} pentanoate
水素化ナトリウム(203 mg, 5.06腿01) (60%油性) のテトラヒドロフラン(12.0 ml) の懸濁液に、 実施例 6-24で得られたェチル 5_[1-ァミノ-5,6-ジメチル-3- ォキソ - 3H-ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル]スルファニル }ペンタノエー ト (1.00 g, 2.81 醒 ol) のテトラヒドロフラン (4.00 il) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4- (ァセチルァミノ)ベンゼンスルホニル クロリド (1.18 g, 5.06 mmol) のテトラヒドロフラン (4.00 ml) 溶液を加え、 さらに室 温で 18時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(219 mg, 14%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 178-180°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (203 mg, 5.06 thigh 01) (60% oily) in tetrahydrofuran (12.0 ml) was added the ethyl 5_ [1-amino-5,6-dimethyl- obtained in Example 6-24. Add 3-oxo-3H-imidazo [5,1-c] [1,4] thiazin-8-yl] sulfanyl} pentanoate (1.00 g, 2.81 awake ol) in tetrahydrofuran (4.00 il), and cool with ice. Stir for 5 minutes below. To this was added a solution of 4- (acetylamino) benzenesulfonyl chloride (1.18 g, 5.06 mmol) in tetrahydrofuran (4.00 ml), and the mixture was further stirred at room temperature for 18 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (219 mg, 14%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 178-180 ° C.
IR (KBr) V: 3272, 2984, 2936, 1728, 1661, 1593, 1557, 1532, 1501 cm"1. ^-NMR (CDC13) : 61.27 (3H, t, J=7.0 Hz), 1.66-1.86 (4H, m), 1.79 (3H, d, J=0.8 Hz), 2.21 (3H, s), 2.26 (3H, s), 2.28-2.38 (2H, m), 2.90-3.00 (2H, m), 4.14 (2H, q, J=6.6 Hz), 7.49 (1H, bs), 7.64 (2H, d, J=8.8 Hz), 7.91 (2H, d, J=8.6 Hz), 9.58 (1H, bs) . IR (KBr) V:. 3272 , 2984, 2936, 1728, 1661, 1593, 1557, 1532, 1501 cm "1 ^ -NMR (CDC1 3): 61.27 (3H, t, J = 7.0 Hz), 1.66-1.86 (4H, m), 1.79 (3H, d, J = 0.8 Hz), 2.21 (3H, s), 2.26 (3H, s), 2.28-2.38 (2H, m), 2.90-3.00 (2H, m), 4.14 (2H, q, J = 6.6 Hz), 7.49 (1H, bs), 7.64 (2H, d, J = 8.8 Hz), 7.91 (2H, d, J = 8.6 Hz), 9.58 (1H, bs).
元素分析値: C23H29N406S3として Elementary analysis: as C 23 H 29 N 4 0 6 S 3
計算値 (%) : C, 49.89; H, 5.28; N, 10.12; S, 17.37. Calculated (%): C, 49.89; H, 5.28; N, 10.12; S, 17.37.
実測値 (¾) : C, 49.66; H, 5.13; N, 9.90; S, 17.28. Found (¾): C, 49.66; H, 5.13; N, 9.90; S, 17.28.
実施例 7-142 Example 7-142
4 -ブ口モ- N- {5, 6-ジメチル -8- [ (5-フルォロぺンチル)スルファニル] -3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデン } - 2_トリフルォロ 水素化ナトリウム(50.7mg, 1.27讓 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた アミノ- 5, 6-ジメチル- 8- {[2- (5-フル ォロペンチルォキシ)ェチル]スルファニル }-3H -ィミダゾ [5, 1-c] [1, 4]チアジン -3-オン (200 mg, 0.634 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 4-ブロモ -2-トリフルォロメトキシベンゼンス ルホニル クロリド (431 mg, 1.27匪 ol) のテトラヒドロフラン (1.00 ml) 溶液 を加え、さらに室温で 1時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (306 mg, 78%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルか ら再結晶して融点 139- U1°Cの暗赤色結晶を得た。 4-Bumo-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylidene} -2_Trifluoro sodium hydride (50.7 mg, 1.27 benzyl) (60% oil) in a suspension of tetrahydrofuran (2.00 ml) in Example 6-23. Amino-5,6-dimethyl-8-{[2- (5-fluoropentyloxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazin-3-one ( A solution of 200 mg (0.634 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-bromo-2-trifluoromethoxybenzenesulfonyl chloride (431 mg, 1.27 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was evaporated under reduced pressure to give the title of compound a (306 mg, 78%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 139-U1 ° C.
IR (KBr) V: 3196, 2938, 2863, 1740, 1665, 1611, 1557 cm-1. IR (KBr) V: 3196, 2938, 2863, 1740, 1665, 1611, 1557 cm- 1 .
Ή-NMR (CDCI3): δ 1.46-1.60 (2Η, m), 1.60-1.78 (4H, m), 1.81 (3H, d, J=1.2 Hz), 2.30 (3H, d, J=1.2 Hz), 2.94 (2H, t, J=7.5 Hz), 4.43 (2H, dt, J=47.1, 6.0 Hz), 7,50-7.58 (2H, m), 8.00 (1H, d, J=8.1 Hz), 9.75 (1H, bs) . 元素分析値: C2Q¾。N304S3BrF4として Ή-NMR (CDCI3): δ 1.46-1.60 (2Η, m), 1.60-1.78 (4H, m), 1.81 (3H, d, J = 1.2 Hz), 2.30 (3H, d, J = 1.2 Hz), 2.94 (2H, t, J = 7.5 Hz), 4.43 (2H, dt, J = 47.1, 6.0 Hz), 7,50-7.58 (2H, m), 8.00 (1H, d, J = 8.1 Hz), 9.75 (1H, bs). Elemental analysis: C 2Q II. N 3 0 4 S 3 As BrF 4
計算値 (%) : C, 38.84; H, 3.26; N, 6.79; S, 15.55; Br, 12.92. Calculated (%): C, 38.84; H, 3.26; N, 6.79; S, 15.55; Br, 12.92.
実測値 ( : C, 39.02; H, 3.19; N, 6.77; S, 15.63; Br, 13.08. Observed (: C, 39.02; H, 3.19; N, 6.77; S, 15.63; Br, 13.08.
実施例 7-143 Example 7-143
4 -プロモ -N- [5, 6-ジメチル- 3-ォキソ -8- (ペンチルスルファ二ル) -2, 3-ジヒドロ -1H-イミダゾ [5,1- c] [1,4]チアジン- イリデン]ベンゼンスルホンアミド 4-Promo-N- [5,6-dimethyl-3-oxo-8- (pentylsulfanyl) -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- Ilidene] benzenesulfonamide
水素化ナトリウム(592 mg, 14.8腿01) (60%油性) のテトラヒドロフラン (30.0 ml) の懸濁液に、 実施例 6-1で得られた 1 -ァミノ- 5, 6 -ジメチル- 8 - (ペンチルス ルファニル) - 3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3 -オン (2.20 g, 7.40 mmol) の テトラヒドロフラン (5.00 ml) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (1.28 g, 7.21 腿 ol) のテトラヒドロフ ラン (5.00 ml) 溶液を加え、 さらに室温で 1.5 時間かき混ぜた。 反応混合物に 1 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (3.43g, 90¾) を暗赤色結晶として得た。 酢酸ェ チル-ジェチルェ一テルから再結晶して融点 176-178での暗赤色結晶を得た。 To a suspension of sodium hydride (592 mg, 14.8 thigh 01) (60% oily) in tetrahydrofuran (30.0 ml) was added 1-amino-5,6-dimethyl-8- (obtained in Example 6-1. A solution of (pentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3-one (2.20 g, 7.40 mmol) in tetrahydrofuran (5.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-bromobenzenesulfonyl chloride (1.28 g, 7.21 mol) in tetrahydrofuran (5.00 ml), and the mixture was further stirred at room temperature for 1.5 hours. Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (3.43g, 90¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals, mp 176-178.
I (KBr) V: 3223, 2957, 2928,. 2861, 1759, 1663, 1622, 1559 cm"1. I (KBr) V: 3223, 2957, 2928, 2861, 1759, 1663, 1622, 1559 cm " 1 .
Ή-N R (CDC13) : 30.917 (3H, t, J=6.9Hz), 1.29-1.44 (4H, m), 1.69 (2H, quint, J=8.1 Hz), 1.79 (3H, d, J=0.9Hz), 2.27 (3H, d, J=0.9 Hz), 2.94 (2H, t, J=7.8 Hz), 7.63 (2H, dt, J=9.0, 2.1 Hz), 7.84 (2H, dt, J=8.7, 1.8 Hz), 9.60 (1H, bs). Ή-NR (CDC1 3): 30.917 (3H, t, J = 6.9Hz), 1.29-1.44 (4H, m), 1.69 (2H, quint, J = 8.1 Hz), 1.79 (3H, d, J = 0.9 Hz), 2.27 (3H, d, J = 0.9 Hz), 2.94 (2H, t, J = 7.8 Hz), 7.63 (2H, dt, J = 9.0, 2.1 Hz), 7.84 (2H, dt, J = 8.7 , 1.8 Hz), 9.60 (1H, bs).
元素分析値: C19H22N303S3Brとして Elementary analysis: as C 19 H 22 N 3 0 3 S 3 Br
計算値 (%) : C, 44.18; H, 4.29; N, 8.14; S, 18.62; Br, 15.47. Calculated (%): C, 44.18; H, 4.29; N, 8.14; S, 18.62; Br, 15.47.
実測値 (%) : C, 44.13; H, 4.20; N, 8.12; S, 18.44; Br, 15.41. Found (%): C, 44.13; H, 4.20; N, 8.12; S, 18.44; Br, 15.41.
実施例 7-144 Example 7-144
4 -ク口口- N - [5, 6 -ジメチル -3-ォキソ- 8 -(ぺンチルスルファニル) -2, 3-ジヒドロ - 1H -イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン]ベンゼンスルホンアミド 4-N- [5,6-dimethyl-3-oxo-8- (pentylsulfanyl) -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -Triylidene] benzenesulfonamide
水素化ナトリウム(172 mg, 4.30匪01) (60%油性) のテトラヒドロフラン(8.00 ml) の懸濁液に、 実施例 6-1で得られた卜アミノ- 5, 6 -ジメチル- 8- (ペンチルス ルファニル )-3H-イミダゾ [5,1 - c] [1,4]チアジン- 3 -オン (640 mg, 2.15 mmol) の テトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4_クロ口ベンゼンスルホニル クロリド (908 mg, 4.30匪 ol) のテトラヒドロフ ラン (2.00 ml) 溶液を加え、 さらに室温で 4時間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (396 mg, 39%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 172-174°Cの喑赤色結晶を得た。 To a suspension of sodium hydride (172 mg, 4.30 band01) (60% oil) in tetrahydrofuran (8.00 ml) was added the triamino-5,6-dimethyl-8- (pentylus) obtained in Example 6-1. Ruphanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (640 mg, 2.15 mmol) A solution of tetrahydrofuran (2.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of benzenesulfonyl chloride (908 mg, 4.30 ol) in tetrahydrofuran (2.00 ml), and the mixture was further stirred at room temperature for 4 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was evaporated reduced pressure to give the title compound (396 mg, 39%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to give reddish crystals having a melting point of 172-174 ° C.
IR (KBr) V: 3225, 2959, 2930, 2863, 1759, 1665, 1622, 1561 cm"1. IR (KBr) V: 3225, 2959, 2930, 2863, 1759, 1665, 1622, 1561 cm " 1 .
Ή-NMR (CDC13) : «50.916 (3H, t, J=7.2 Hz), 1.30-1.44 (4H, m), 1.68 (2H, quint, J=7.5 Hz), 1.80 (3H, d, J=l.2 Hz), 2.28 (3H, d, J=0.9 Hz), 2.95 (2H, t, J=7.8 Hz), 7.46 (2H, dt, J=9.0, 2.1 Hz), 7.91 (2H, dt, J=9.0, 2.1 Hz), 9.60 (1H, bs). Ή-NMR (CDC1 3): «50.916 (3H, t, J = 7.2 Hz), 1.30-1.44 (4H, m), 1.68 (2H, quint, J = 7.5 Hz), 1.80 (3H, d, J = l.2 Hz), 2.28 (3H, d, J = 0.9 Hz), 2.95 (2H, t, J = 7.8 Hz), 7.46 (2H, dt, J = 9.0, 2.1 Hz), 7.91 (2H, dt, J = 9.0, 2.1 Hz), 9.60 (1H, bs).
元素分析値: C19H22N303S3C1として Elemental analysis: C 19 H 22 N 3 0 3 S 3 C1
計算値 (%) : C, 48.34; H, 4.70; N, 8.90; S, 20.38. Calculated value (%): C, 48.34; H, 4.70; N, 8.90; S, 20.38.
実測値 (¾) : C, 48.17; H, 4.72;. N, 8.68; S, 20.46. Found (¾): C, 48.17; H, 4.72 ;. N, 8.68; S, 20.46.
実施例 7-145 Example 7-145
4 -ブロモ - N-(5, 6 -ジメチル- 8- {[2 - (5-フルォロペンチルォキシ)ェチル]スルファ 二ル}- 3 -才キソ -2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン) ベンゼンスルホンアミド 4-Bromo-N- (5,6-dimethyl-8-{[2- (5-fluoropentyloxy) ethyl] sulfanyl} -3- 3-year-old oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene) benzenesulfonamide
水素化ナトリゥム(87.7 mg, 2.19mmol) (60 由性)のテトラヒドロフラン(4.00 ml) の懸濁液に、 実施例 6-34で得られた卜ァミノ- 5, 6-ジメチル- 8-{[2 -(5-フル ォロぺンチルォキシ)ェチル]スルファ二ル}署ィミダゾ [5, 1 - c] [1,4]チアジン -3 -オン (427 mg, 1.10腿 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷 冷下 5分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (572 mg, 2.19mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 さらに室温で 18時間 かき混ぜた。反応混合物に IN塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (245 mg, 39%) を暗赤色 結晶として得た。酢酸ェチル-ジェチルエーテルから再結晶して融点 130- 132°Cの 暗赤色結晶を得た。. To a suspension of hydrogenated sodium (87.7 mg, 2.19 mmol) (60 free) in tetrahydrofuran (4.00 ml) was added the triamino-5,6-dimethyl-8-{[2 -(5-Fluorobenzoyloxy) ethyl] sulfanyl} imidazo [5,1-c] [1,4] thiazin-3-one (427 mg, 1.10 t) in tetrahydrofuran (2.00 ml) And stirred under ice-cooling for 5 minutes. To this was added a solution of 4-bromobenzenesulfonyl chloride (572 mg, 2.19 mmol) in tetrahydrofuran (2.00 ml), and the mixture was further stirred at room temperature for 18 hours. IN hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (245 mg, 39%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals with a melting point of 130-132 ° C. .
IR (KBr)レ : 3235, 3088, 2936, 2865, 1759, 1663, 1620, 1557 cm—1. Ή-NMR (CDCI3) : δ 1.40-1.85 (6Η, m), 1.80 (3Η, d, J=0.8 Hz), 2.27 (3H, d, J=1.0Hz), 3.16 (2H, t, J=6.6 Hz), 3.47 (2H, t, J-6.2Hz), 3.64 (2H, t, J=6.6 Hz), 4.45 (2H, dt, J=47.4, 6.0 Hz), 7.62 (2H, d, J=8.8Hz), 7.83 (2H, d, J=8.8 Hz), 9.60 (1H, bs). IR (KBr): 3235, 3088, 2936, 2865, 1759, 1663, 1620, 1557 cm— 1 . Ή-NMR (CDCI3): δ 1.40-1.85 (6Η, m), 1.80 (3Η, d, J = 0.8 Hz), 2.27 (3H, d, J = 1.0 Hz), 3.16 (2H, t, J = 6.6 Hz), 3.47 (2H, t, J-6.2Hz), 3.64 (2H, t, J = 6.6 Hz), 4.45 (2H, dt, J = 47.4, 6.0 Hz), 7.62 (2H, d, J = 8.8 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.60 (1H, bs).
元素分析値: C21H25N304S3BrFとして Elementary analysis: as C 21 H 25 N 3 0 4 S 3 BrF
計算値 (¾) : C, 43.60; H, 4.36; N, 7.26; S, 16.63; Br, 13.81. Calculated (¾): C, 43.60; H, 4.36; N, 7.26; S, 16.63; Br, 13.81.
実測値 (%) : C, 43.34; H, 4.20; N, 7.50; S, 16.76; Br, 13.91. Found (%): C, 43.34; H, 4.20; N, 7.50; S, 16.76; Br, 13.91.
実施例 7-146 Example 7-146
2-[({5, 6-ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファ ニル] -2, 3 -ジヒドロ -1H -イミダゾ [5,1- c] [1,4]チアジン - 1 -ィリデン }ァミノ)ス ルホニル]安息香酸 2-[({5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene} amino) sulfonyl] benzoic acid
実施例 7-145と同様にして合成し、テトラヒドロフラン-メタノールから再結晶 して融点 152-154°Cの暗赤色結晶を得た。 It was synthesized in the same manner as in Example 7-145, and recrystallized from tetrahydrofuran-methanol to obtain dark red crystals having a melting point of 152-154 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3400-2300, 3260, 2922, 1748, 1705, 1667, 1620, 1615, 1551 cm— Infrared absorption spectrum (IR) (in KBr) V: 3400-2300, 3260, 2922, 1748, 1705, 1667, 1620, 1615, 1551 cm—
lH-NMR (CDCI3) : δ 1.65-1.87 (4Η, m), 1.79 (3H, s), 1.97-2.16 (2H, m), 2.28lH-NMR (CDCI3): δ 1.65-1.87 (4Η, m), 1.79 (3H, s), 1.97-2.16 (2H, m), 2.28
(3H, d, J=0.9 Hz), 2.98 (2H, t, J=6.9 Hz), 7.62-7.70 (2H, m), 7.90-7.96 (1H, m), 8.10-8.18 (1H, m), 9.85 (1H, bs). (3H, d, J = 0.9 Hz), 2.98 (2H, t, J = 6.9 Hz), 7.62-7.70 (2H, m), 7.90-7.96 (1H, m), 8.10-8.18 (1H, m), 9.85 (1H, bs).
元素分析値: C21H2DN305S3F5として Elemental analysis: as C 21 H 2D N 3 0 5 S 3 F 5
計算値 (%) : C, 43.07; H, 3.44; N, 7.18; S, 16.43. Calculated value (%): C, 43.07; H, 3.44; N, 7.18; S, 16.43.
実測値 (%) : C, 42.75; H, 3.53; N, 7.08; S, 16.34. Found (%): C, 42.75; H, 3.53; N, 7.08; S, 16.34.
実施例 7-148 Example 7-148
4-ブ口モ- N- (5, 6-ジメチル- 8-{ [2- (4-フルォロブトキシ)ェチル]スルファニ ル }- 3 -ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン - 1-ィリデン)ベ ンゼンスルホンアミド 4-Bumo-N- (5,6-dimethyl-8-{[2- (4-fluorobutoxy) ethyl] sulfanyl} -3-oxo-2,3-dihydro-1H-imidazo [5, tric ] [1,4] thiazine-1-ylidene) benzenesulfonamide
水素化ナトリウム(31.2 mg, 0.779 mmol) (60%油性)のテトラヒドロフラン(2· 00 ml) の懸濁液に、 実施例 6- 33で得られた卜ァミノ- 5, 6 -ジメチル _8 - {[2- (4-フル ォロブトキシ)ェチル]スルファ二ル} - 3H-ィミダゾ [5, 1-c] [1 , 4]チアジン- 3-ォン (135 mg, 0.390 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (223 mg, 0.779 mmol) のテトラヒドロフラン (1.00ml) 溶液を加え、 さらに室温で 5時間かき混 ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (143 mg, 65¾) を暗赤色結晶 として得た。酢酸ェチル-ジェチルエーテルから再結晶して融点 132-134°Cの暗赤 色結晶を得た。 To a suspension of sodium hydride (31.2 mg, 0.779 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl_8-{[obtained in Example 6-33. 2- (4-Fluorobutoxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazine-3-one (135 mg, 0.390 mmol) in tetrahydrofuran (1.00 ml) And cool under ice 5 Stir for a minute. To this was added a solution of 4-bromobenzenesulfonyl chloride (223 mg, 0.779 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), obtained by the solvent evaporated under reduced pressure to give the title compound (143 mg, 65¾) and a dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals with a melting point of 132-134 ° C.
IR (KBr) V: 3245, 3085, 2961, 2863, 1759, 1663, 1622, 1559 cm"1. IR (KBr) V: 3245, 3085, 2961, 2863, 1759, 1663, 1622, 1559 cm " 1 .
[H-NMR (CDC13) : δ 1.66 - 1.78 (4Η, m), 1.80 (3H, d, J=1.2 Hz), 2.27 (3H, d, J=1.2 Hz), 3.16 (2H, t, J=6.3Hz), 3.50 (2H, t, J=5.7 Hz), 3.65 (2H, t, J=6.3 Hz), 4.46 (2H, dt, J=47.4, 6.0 Hz), 7.63 (2H, d, J=8.7 Hz), 7.83 (2H, d, J=8.4 Hz), 9.60 (1H, bs). [H-NMR (CDC1 3) : δ 1.66 - 1.78 (4Η, m), 1.80 (3H, d, J = 1.2 Hz), 2.27 (3H, d, J = 1.2 Hz), 3.16 (2H, t, J = 6.3Hz), 3.50 (2H, t, J = 5.7 Hz), 3.65 (2H, t, J = 6.3 Hz), 4.46 (2H, dt, J = 47.4, 6.0 Hz), 7.63 (2H, d, J = 8.7 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.60 (1H, bs).
実施例 7-149 Example 7-149
N- {5, 6-ジメチル- 8- [ (5_フルォ口ぺンチル)スルファニル] -3-ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5,l-c] [1, 4]チアジン -1-ィリデン } -3-ピリジンスルホンアミド 水素化ナトリウム(288 mg, 7.21匪01) (60%油性)のテトラヒドロフラン(15.0 ml) の懸濁液に、 実施例 6-23で得られたトァミノ- 5, 6 -ジメチル- 8-{[2- (5 -フル ォロペンチルォキシ)ェチル]スルファニル }- 3H -ィミダゾ [5, 1-c] [1,4]チアジン _3 -オン (1.02 g, 3.23 mmol) のテトラヒドロフラン (5.00 ml) 溶液を加え、 氷 冷下 5分間かき混ぜた。 これに参考例 45の方法 Bで得られた 3-ピリジンスルホ ニル クロリド (1.28 g, 7.21 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加 え、 さらに室温で 16時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチル で抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合 物 (1.11 g, 75%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから 再結晶して融点 135- 137 の暗赤色結晶を得た。 N- {5,6-Dimethyl-8-[(5_fluoromethylpentyl) sulfanyl] -3-oxo-2,3-dihidido-1H-imidazo [5, lc] [1,4] thiazine-1 -Ylidene} -3-pyridinesulfonamide To a suspension of sodium hydride (288 mg, 7.21 maraudal 01) (60% oil) in tetrahydrofuran (15.0 ml) was added the toamino-5, obtained in Example 6-23. 6-Dimethyl-8-{[2- (5-fluoropentyloxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazine_3-one (1.02 g, 3.23 mmol ) In tetrahydrofuran (5.00 ml) and stirred for 5 minutes under ice-cooling. A solution of 3-pyridinesulfonyl chloride (1.28 g, 7.21 mmol) obtained in Method B of Reference Example 45 in tetrahydrofuran (2.00 ml) was added thereto, and the mixture was further stirred at room temperature for 16 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (1.11 g, 75%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 135-137.
IR (KBr) v: 2963, 2938, 2863, 1746, 1663, 1607, 1557 cm1. IR (KBr) v: 2963, 2938, 2863, 1746, 1663, 1607, 1557 cm 1 .
Ή-NMR (CDC13) : δ 1.48-1.64 (2H, m), 1.64-1.84 (4Η, m), 1.81 (3H, d, J=0.9 Hz), 2.29 (3H, d, J=1.2 Hz), 2.98 (2H, t, J=7.8 Hz), 4.46 (2H, dt, J=47. , 5.7 Hz), 7.45 (1H, ddd, J=8.1, 4.8, 0.6 Hz), 8.25 (1H, ddd, J=7.8, 2.1, 1.5 Hz), 8.78 (lH, dd, J=5.1, 1.8 Hz), 9.20 (1H, dd, J=2.4, 0.6 Hz), 9.63 (1H, bs). Ή-NMR (CDC1 3): δ 1.48-1.64 (2H, m), 1.64-1.84 (4Η, m), 1.81 (3H, d, J = 0.9 Hz), 2.29 (3H, d, J = 1.2 Hz) , 2.98 (2H, t, J = 7.8 Hz), 4.46 (2H, dt, J = 47., 5.7 Hz), 7.45 (1H, ddd, J = 8.1, 4.8, 0.6 Hz), 8.25 (1H, ddd, J = 7.8, 2.1, 1.5 Hz), 8.78 (lH, dd, J = 5.1, 1.8 Hz), 9.20 (1H, dd, J = 2.4, 0.6 Hz), 9.63 (1H, bs).
元素分析値: C18H21N403S3F4として Elemental analysis: as C 18 H 21 N 4 0 3 S 3 F 4
計算値 (%) : C, 47.35; H, 4.64; N, 12.27; S, 21.07. Calculated value (%): C, 47.35; H, 4.64; N, 12.27; S, 21.07.
実測値 ) : C, 47.51; H, 4.69; N, 12.14; S, 21.25. Obtained): C, 47.51; H, 4.69; N, 12.14; S, 21.25.
実施例 7-150 Example 7-150
4-ブロモ _N - (5, 6-ジメチル- 8- { [2- (3-フルォロプロポキシ)ェチル]スルファニ ル卜 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン)ベ ンゼンスルホンアミド 4-bromo_N- (5,6-dimethyl-8-{[2- (3-fluoropropoxy) ethyl] sulfanilto 3-oxo-2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-1-ylidene) benzenesulfonamide
水素化ナトリウム(43.8mg, 1.09讓 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-36で得られた 1-ァミノ- 5, 6-ジメチル _8 - {[2 - (3-フル ォロプロボキシ)ェチル]スルファ二ル} -3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 3 -才 ン (181 mg, 0.547 mmol) のテトラヒドロフラン (l.OO ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (285 mg, 1.09 腿 ol) のテトラヒドロフラン (l.OOml) 溶液を加え、 さらに室温で 2時間かき混 ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 ( gS04) 後、 溶媒を減圧下に留去して題記化合物 (128 mg, 43%) を暗赤色結晶 として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 122- 124°Cの暗赤 色結晶を得た。 To a suspension of sodium hydride (43.8 mg, 1.09 acetyl) (60% oil) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl_8-{[obtained in Example 6-36. 2- (3-Fluoropropoxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,4] thiazine-3-amine (181 mg, 0.547 mmol) in tetrahydrofuran (l.OO ml ) The solution was added and stirred for 5 minutes under ice cooling. To this was added a solution of 4-bromobenzenesulfonyl chloride (285 mg, 1.09 tmol) in tetrahydrofuran (l.OOml), and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (GS0 4), solvent was obtained as a dark red crystalline title compound was distilled off (128 mg, 43%) under reduced pressure. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 122-124 ° C.
IR (KBr) V : 3223, 3094, 2868, 1759, 1665, 1620, 1561 cm—1. IR (KBr) V: 3223, 3094, 2868, 1759, 1665, 1620, 1561 cm- 1 .
Ή-NMR (CDC13) : (51.80 (3H, d, J=0.9 Hz), 1.95 (2H, dt, J=26.4, 5.7 Hz), Ή-NMR (CDC1 3): (51.80 (3H, d, J = 0.9 Hz), 1.95 (2H, dt, J = 26.4, 5.7 Hz),
2.27 (3H, d, J=0.9 Hz), 3.17 (2H, t, J=6.3 Hz), 3.58 (2H, t, J=6.0 Hz), 3.672.27 (3H, d, J = 0.9 Hz), 3.17 (2H, t, J = 6.3 Hz), 3.58 (2H, t, J = 6.0 Hz), 3.67
(2H, t, J=6.3 Hz), 4.54 (2H, dt, J=47.1, 6.6 Hz), 7.63 (2H. dt, J=8.4, 1.5(2H, t, J = 6.3 Hz), 4.54 (2H, dt, J = 47.1, 6.6 Hz), 7.63 (2H.dt, J = 8.4, 1.5
Hz), 7.83 (2H, dt, J=8.7, 1.8 Hz), 9.60 (1H, bs). Hz), 7.83 (2H, dt, J = 8.7, 1.8 Hz), 9.60 (1H, bs).
元素分析値: C19H2lN304S3BrFとして Elementary analysis: as C 19 H 2l N 3 0 4 S 3 BrF
計算値 (%) : C, 41.45; H, 3.85; N, 7.63; S, 17.47; Br, 14.52. Calculated (%): C, 41.45; H, 3.85; N, 7.63; S, 17.47; Br, 14.52.
実測値 (%) : C, 41.36; H, 3.85; N, 7.55; S, 17.59; Br, 14.44. Found (%): C, 41.36; H, 3.85; N, 7.55; S, 17.59; Br, 14.44.
実施例 7-151 Example 7-151
4-ブ口モ- N- (5, 6-ジメチル- 8- { [2- (2-フルォロェトキシ)ェチル]スルファニ }-3-ォキソ-2,3_ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1-ィリデン)ベ ンゼンスルホンアミド 4-Buguchi-N- (5,6-dimethyl-8-{[2- (2-fluoroethoxy) ethyl] sulfani} -3-oxo-2,3_dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-1-ylidene) be Nzensulfonamide
水素化ナトリウム(63.0mg, 1.58翻 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-43で得られた卜ァミノ- 5, 6 -ジメチル -8 - {[2- (2 -フル ォロエトキシ)ェチル]スルファニル }- 3H-ィミダゾ [5, 1-c] [1, ]チアジン- 3-オン (250 mg, 0.788醒 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (411 mg, 1.58 讓 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 さらに室温で 6時間かき混 ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (131 mg, 31¾) を暗赤色結晶 として得た。酢酸ェチル-ジェチルエーテルから再結晶して融点 159-161°Cの暗赤 色結晶を得た。 To a suspension of sodium hydride (63.0 mg, 1.58 ol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8-{[obtained in Example 6-43. 2- (2-Fluoroethoxy) ethyl] sulfanyl} -3H-imidazo [5,1-c] [1,]] thiazin-3-one (250 mg, 0.788 ol) in tetrahydrofuran (1.00 ml) was added. The mixture was stirred for 5 minutes under ice cooling. To this was added a solution of 4-bromobenzenesulfonyl chloride (411 mg, 1.58 alcohol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 6 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), obtained by the solvent evaporated under reduced pressure to give the title compound (131 mg, 31¾) and a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 159-161 ° C.
IR (KBr) リ: 3218, 2930, 1757, 1663, 1622, 1559 cm"1. IR (KBr): 3218, 2930, 1757, 1663, 1622, 1559 cm " 1 .
Ή— NMR (CDC13) : (51.80 (3H, s), 2.27 (3H, s), 3.19 (2H, t, J=6.6 Hz), 3.73 (2H, dt, J=30.8, 4.4 Hz), 3.75 (2H, t, J=6.6 Hz), 4.56 (2H, dt, J=47.6, 3.6 Hz), 7.63 (2H, d, J=8.8 Hz), 7.84 (2H, dt, J=8. Hz), 9.59 (1H, bs). 元素分析値: C〖8H19N304S3BrFとして Ή- NMR (CDC1 3): ( 51.80 (3H, s), 2.27 (3H, s), 3.19 (2H, t, J = 6.6 Hz), 3.73 (2H, dt, J = 30.8, 4.4 Hz), 3.75 (2H, t, J = 6.6 Hz), 4.56 (2H, dt, J = 47.6, 3.6 Hz), 7.63 (2H, d, J = 8.8 Hz), 7.84 (2H, dt, J = 8.Hz), 9.59 (1H, bs) Elementary analysis:. a C 〖8 H 19 N 3 0 4 S 3 BrF
計算値 (¾) : C, 40.30; H, 3.57; N, 7.83; S, 17.93; Br, 14.89. Calculated (¾): C, 40.30; H, 3.57; N, 7.83; S, 17.93; Br, 14.89.
実測値 (%) : C, 40.42; H, 3.57; N, 8.04; S, 17.88; Br, 14.96. Found (%): C, 40.42; H, 3.57; N, 8.04; S, 17.88; Br, 14.96.
実施例 7-152 Example 7-152
tert -ブチル 3-{[卜({[4 -(ァセチルァミノ)フエニル]スルホ二ル}ィミノ) - 5,6- ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル]ス ルファ二ル}プロピルカーバメート tert-butyl 3-{[(([[4- (acetylamino) phenyl] sulfonyl} imino))-5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-8-yl] sulfanyl} propyl carbamate
実施例 6-38で得た化合物 (337 mg, 0.876删 ol)の THF (8 ml) 懸濁液に水素化 ナトリウム(純度 60%油性, 105 mg, 2.63 mmol) を室温で加え、 10分間攪拌後、 塩化 4 -ァセタミドベンゼンスルホニル (253 mg, 1.05 mmol) を加え、さらに 2.5 時間攪拌した。 反応液にメタノール (1 ml) および 1N塩酸 (3.0 ml)を加えて 10分間攪拌した。 析出物を濾取し、 メタノール—水 (1:1)およびメタノールで洗 浄後、 乾燥して表題化合物(309 mg, 0.531 mmol, 60.6%)を紫色粉末として得た。 Ή -腿 (200MHz, CDCI3): δ 1.45 (9H, s), 1.78 (3H, brs), 1.85 (2H, quint, J= 7.1 Hz), 2.21 (3H, s), 2.26 (3H, s), 2.95 (2H, t, J= 7.4 Hz), 3.18 (2H, q, J= 7.0 Hz), 4.66 (1H, m), 7.59 (1H, brs) 7.64 (2H, d, J- 8.8 Hz), 7.90 (2H, d, J= 8.4 Hz), 9.59 (1H, brs). Sodium hydride (purity 60%, 105 mg, 2.63 mmol) was added to a suspension of the compound obtained in Example 6-38 (337 mg, 0.876 mol) in THF (8 ml) at room temperature, and the mixture was stirred for 10 minutes. Thereafter, 4-acetamidobenzenesulfonyl chloride (253 mg, 1.05 mmol) was added, and the mixture was further stirred for 2.5 hours. Methanol (1 ml) and 1N hydrochloric acid (3.0 ml) were added to the reaction solution, and the mixture was stirred for 10 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (309 mg, 0.531 mmol, 60.6%) as a purple powder. Ή-thigh (200MHz, CDCI3): δ 1.45 (9H, s), 1.78 (3H, brs), 1.85 (2H, quint, J = 7.1 Hz), 2.21 (3H, s), 2.26 (3H, s), 2.95 (2H, t, J = 7.4 Hz), 3.18 (2H, q, J = 7.0 Hz), 4.66 (1H, m), 7.59 (1H, brs) 7.64 (2H, d, J- 8.8 Hz), 7.90 (2H, d, J = 8.4 Hz), 9.59 (1H, brs).
元素分析値: C24H31N506S3として計算値: C, 49.55; H, 5.37; N, 12.04; S, 16.54 (%) 、 実測値: C, 49.55; H, 5.55; N, 11.92; S, 16.44 (%) . Elemental analysis: C 24 H 31 N 5 0 6 S 3 Calculated: C, 49.55; H, 5.37 ; N, 12.04; S, 16.54 (%), Found: C, 49.55; H, 5.55 ; N, 11.92; S, 16.44 (%).
mp. 170.5-173. Ot: mp. 170.5-173. Ot:
実施例 7-153 Example 7-153
N- {5, 6-ジメチル -8 - [ (5 -フルォロぺンチル)スルファニル] -3-ォキソ- 2, 3 -ジヒド ロ- 1H -イミダゾ [5,1-c] [1,4]チアジン- 1-ィリデン } - 4 -ビニルベンゼンスルホン アミド N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene}-4-vinylbenzenesulfonamide
水素化ナトリウム(50.7 rag, 1.27腿01) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた 1 -アミノ- 5, 6-ジメチル- 8-[(5 -フルォ 口ペンチル)スルファニル ]-3H -イミダゾ [5, 1-c] [1,4]チアジン -3-オン (200 mg, 0.634腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 4-ビニルベンゼンスルホニル クロリド (249 mg, 1.14删 ο1)、のテ トラヒドロフラン (1.00ml) 溶液を加え、 さらに室温で 2時間かき混ぜた。 反応 混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(258 mg, 85%)を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 182_184 の暗赤色結晶を得 た。 To a suspension of sodium hydride (50.7 rag, 1.27 thigh 01) (60% oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8- [obtained in Example 6-23. (5-Fluo-pentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.634 tmol) in tetrahydrofuran (1.00 ml) and ice-cooled Stir for 5 minutes below. To this was added a solution of 4-vinylbenzenesulfonyl chloride (249 mg, 1.14 删 ο1) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (258 mg, 85%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 182_184.
IR (KBr) リ: 3235, 2961, 2936, 2861, 1753, 1663, 1620, 1557 cm-1. IR (KBr): 3235, 2961, 2936, 2861, 1753, 1663, 1620, 1557 cm- 1 .
Ή— NMR (CDC13) : δ 1.48-1.62 (2Η, m), 1.62-1.82 (4H, m), 1.79 (3H, d, J=0.9 Hz), 2.27 (3H, d, J=1.2 Hz), 2.96 (2H, t, J=7.5 Hz), 4.45 (2H, dt, J=47. , 6.0 Hz), 5.41 (1H, d, J=10.8Hz), 5.87 (1H, d, J=17. Hz), 6.74 (1H, dd, J=17.4, 10.5 Hz), 7.51 (2H, d, J=8.4 Hz), 7.92 (2H, d, J=8.4 Hz), 9.63 (1H, bs) . 実施例 7-154 Ή- NMR (CDC1 3): δ 1.48-1.62 (2Η, m), 1.62-1.82 (4H, m), 1.79 (3H, d, J = 0.9 Hz), 2.27 (3H, d, J = 1.2 Hz) , 2.96 (2H, t, J = 7.5 Hz), 4.45 (2H, dt, J = 47., 6.0 Hz), 5.41 (1H, d, J = 10.8Hz), 5.87 (1H, d, J = 17. Hz), 6.74 (1H, dd, J = 17.4, 10.5 Hz), 7.51 (2H, d, J = 8.4 Hz), 7.92 (2H, d, J = 8.4 Hz), 9.63 (1H, bs). 7-154
[(1R, 4S)-7, 7 -ジメチル -2-ォキソビシク口 [2, 2, 1]ヘプト- 1-ィル] - N- {5, 6-ジメ チル- 8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3 -ジヒドロ- m-イミ ダゾ [5,1- c] [1,4]チアジン- 1-イリデン }メタンスルホンアミド 水素化ナトリウム (50.7 mg, 1.27漏 1) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6 -ジメチル- 8-[(5-フルォ 口ペンチル)スルファニル〕-3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (200 mg, 0.634删 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 10- (-) -カンファースルホニル クロリド (286 mg, 1.14腿 oi) の テトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 2.5時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(205 mg, 61¾)を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 161-163°Cの暗赤色結晶を得 た。 [(1R, 4S) -7,7-dimethyl-2-oxobisic mouth [2,2,1] hept-1-yl] -N- {5,6-dimethyl-8-[(5-fluoropentyl) Sulfanyl] -3-oxo-2,3-dihydro-m-imidazo [5,1-c] [1,4] thiazine-1-ylidene} methanesulfonamide To a suspension of sodium hydride (50.7 mg, 1.27 leak 1) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8-[(obtained in Example 6-23. 5-Fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.634 mol) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled under ice-cooling. Stir for a minute. To this was added a solution of 10-(-)-camphorsulfonyl chloride (286 mg, 1.14 oi) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (205 mg, 61¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 161-163 ° C.
IR (KBr) レ : 3204, 2957, 1744, 1665, 1617, 1568 cm—'. IR (KBr) Les: 3204, 2957, 1744, 1665, 1617, 1568 cm— '.
Ή-NMR (CDC13) : (50.867 (3H, s), 1.10 (3H, s), 1.35-2.15 (11H, m), 1.80 (3H, s), 2.29 (3H, s), 2.28-2.45 (1H, m), 2.45-2.64 (1H, m), 2.98 (2H, t, J=7.4 Hz), 3.07 (1H, d, J=14.6 Hz), 3.62 (1H, d, J=14.6 Hz), 4.46 (2H, dt, J=47.2, 5.6 Hz), 9.77 (1H, bs). 実施例 7-155 Ή-NMR (CDC1 3): (50.867 (3H, s), 1.10 (3H, s), 1.35-2.15 (11H, m), 1.80 (3H, s), 2.29 (3H, s), 2.28-2.45 ( 1H, m), 2.45-2.64 (1H, m), 2.98 (2H, t, J = 7.4 Hz), 3.07 (1H, d, J = 14.6 Hz), 3.62 (1H, d, J = 14.6 Hz), 4.46 (2H, dt, J = 47.2, 5.6 Hz), 9.77 (1H, bs).
N-{5, 6-ジメチル- 8-[(5 フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド ロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン - 1 -ィリデン } - 2, 1,3-ベンゾチアジアゾー ル- 5-スルホンアミド N- {5,6-dimethyl-8-[(5fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene }-2,1,3-benzothiadiazol-5-sulfonamide
水素化ナトリウム(50.7mg, 1.27醒01) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた ί -アミノ- 5, 6-ジメチル- 8- [(5-フルォ 口ペンチル)スルファニル ]-3Η-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (200 mg, 0.634nimol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。これに 2, 1,3-ベンゾチアジアゾール -5-スルホニル クロリド(268 mg, 1.14 mmol) のテトラヒドロフラン (1.00 ml)溶液を加え、 さらに室温で 2時間かき混 ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (236 mg, 73%) を暗赤色結晶 として得た。酢酸ェチル-ジェチルエーテルから再結晶して融点 165-167°Cの暗赤 色結晶を得た。 To a suspension of sodium hydride (50.7 mg, 1.27 aging 0 1) (60% oil) in tetrahydrofuran (2.00 ml) was added the ί-amino-5,6-dimethyl-8-methyl obtained in Example 6-23. [(5-Fluoropentyl) sulfanyl] -3Η-imidazo [5, tric] A solution of [1,4] thiazin-3-one (200 mg, 0.634nimol) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled on ice. Stir for 5 minutes. To this was added a solution of 2,1,3-benzothiadiazole-5-sulfonyl chloride (268 mg, 1.14 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was obtained as a dark red crystalline title compound was distilled off (236 mg, 73%) under reduced pressure. Recrystallized from ethyl acetate-getyl ether, dark red with melting point 165-167 ° C Color crystals were obtained.
IR (KBr) V: 3223, 3071, 2936, 2861, 1742, 1663, 1615, 1557, 1520 cm"1. 'H-N R (CDC13) : δ 1.44-1.85 (6H, m), 1.80 (3H, s), 2.28 (3H, d, J=0.8 Hz), 2.98 (2H, t, J=7.0 Hz), 4.45 (2H, dt, J=47.2, 5.6 Hz), 8.12 (2H, d, J=0.8 Hz), 8.68 (1H, s), 9.69 (1H, bs). IR (KBr) V:. 3223 , 3071, 2936, 2861, 1742, 1663, 1615, 1557, 1520 cm "1 'HN R (CDC1 3): δ 1.44-1.85 (6H, m), 1.80 (3H, s ), 2.28 (3H, d, J = 0.8 Hz), 2.98 (2H, t, J = 7.0 Hz), 4.45 (2H, dt, J = 47.2, 5.6 Hz), 8.12 (2H, d, J = 0.8 Hz) ), 8.68 (1H, s), 9.69 (1H, bs).
元素分析値: C19H2。N503S4Fとして Elemental analysis: C 19 H 2. N 5 0 3 S 4 F
計算値 (%) : C, 44.43; H, 3.92; N, 13.63; S, 24.97. Calculated value (%): C, 44.43; H, 3.92; N, 13.63; S, 24.97.
実測値 (¾) : C, 44.50; H, 3.74; N, 13.66; S, 24.76. Found (¾): C, 44.50; H, 3.74; N, 13.66; S, 24.76.
実施例 7-156 Example 7-156
N- {5, 6-ジメチル _8_[ (5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口 -1H-ィミダゾ [5, 1-c] [1 , 4]チアジン-卜イリデン } -2, 1, 3 -べンゾォキサジァゾ ール -4-スルホンアミド N- {5,6-dimethyl_8 _ [(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydric mouth-1H-imidazo [5,1-c] [1,4] thiazine-triylidene}- 2,1,3-Benzoxaziazol-4-sulfonamide
水素化ナトリウム(50.7mg, 1.27匪01) (60油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6 - 23で得られた 1-アミノ- 5,6-ジメチル_8_[(5-フルォ 口ぺンチル)スルファニル] -3H -ィミダゾ [5,卜 c] [1, 4]チアジン- 3-ォン (200 mg, 0.634讓 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 2, 1, 3-ベンゾォキサジァゾール -4-スルホニル クロリド (250 mg, 1.14醒 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 18.5 時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を 水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (159 mg, 51¾) を 暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 179_181°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (50.7 mg, 1.27 band01) (60 oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl_8 _ [(5 -Fluoro mouth pentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazine-3-one (200 mg, 0.634 acetyl) in tetrahydrofuran (1.00 ml), and add ice-cooling Stir for 5 minutes. To this was added a solution of 2,1,3-benzoxadiazole-4-sulfonyl chloride (250 mg, 1.14 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 18.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (159 mg, 51¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 179-181 ° C.
IR (KBr) リ: 3260, 3094, 2930, 2861, 1744, 1661, 1617, 1557 cm"1. IR (KBr): 3260, 3094, 2930, 2861, 1744, 1661, 1617, 1557 cm " 1 .
Ή-NMR (CDCI3) : 51.20-1.78 (6H, m), 1.80 (3H, s), 2.33 (3H, s), 2.90 (2H, t, J=7.0 Hz), 4.43 (2H, dt, J=47.2, 6.0 Hz), 7.54 (1H, dd, J-9.2, 6.6 Hz), 8.06 (1H, d, J=9.2 Hz), 8.13 (1H, d, J=6.6 Hz), 9.87 (1H, bs). Ή-NMR (CDCI3): 51.20-1.78 (6H, m), 1.80 (3H, s), 2.33 (3H, s), 2.90 (2H, t, J = 7.0 Hz), 4.43 (2H, dt, J = 47.2, 6.0 Hz), 7.54 (1H, dd, J-9.2, 6.6 Hz), 8.06 (1H, d, J = 9.2 Hz), 8.13 (1H, d, J = 6.6 Hz), 9.87 (1H, bs) .
元素分析値: C19H2QN504S3Fとして Elementary analysis: as C 19 H 2Q N 5 0 4 S 3 F
計算値 (%) : C, 43.86; H, 4.05; N, 14.07; S, 19.33. Calculated value (%): C, 43.86; H, 4.05; N, 14.07; S, 19.33.
実測値 ( ) : C, 45.87; H, 4.13; N, 13.98; S, 19.12. . 実施例 7-157 Found (): C, 45.87; H, 4.13; N, 13.98; S, 19.12. Example 7-157
N-{5, 6 -ジメチル -8 - [ (5-フルォ口ペンチル)スルファニル] -3-ォキソ -2, 3-ジヒド ロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン }[1,1' -ビフエニル ]_4-スル ホンアミド N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-tri Ylidene} [1,1'-biphenyl] _4-sulfonamide
水素化ナトリウム(25.4 mg, 0.63411111101)(60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた 1 -ァミノ- 5,6-ジメチル 8- [5-フルォロ ペンチル]スルファニル ]-3H -イミダゾ [5,卜 c] [1,4]チアジン -3-オン (100 mg, 0.317mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにビフエニルスルホニル クロリド (134 mg, 0.476 mmol) のテトラヒ ドロフラン (1.00ml) 溶液を加え、 さらに 20分間かき混ぜた。 反応混合物に 1N 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減 圧下に留去して題記化合物 (153 mg, 91%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 181-183 の暗赤色結晶を得た。 To a suspension of sodium hydride (25.4 mg, 0.63411111101) (60% oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl 8- [5-fluoro obtained in Example 6-23. A solution of [pentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.317 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice-cooling for 5 minutes. . To this was added a solution of biphenylsulfonyl chloride (134 mg, 0.476 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 20 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was evaporated reduced pressure to give the title compound (153 mg, 91%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 181-183.
IR (KBr) リ: 3210, 3061, 2934, 1740, 1663, 1615, 1557 cm— IR (KBr): 3210, 3061, 2934, 1740, 1663, 1615, 1557 cm—
Ή-NMR (CDC13) : 51. 8-1.62 (4H, m), 1.62-1.82 (2H, m), 1.79 (3H, s), 2.28 (3H, s), 2.97 (2H, t, J=6.9 Hz), 4.44 (2H, dt, J=47.1, 5.7 Hz), 7.37-7.53 (3H, m), 7.60 (2H, d, J-7.2 Hz), 7.70 (2H, d, J=8.7 Hz), 8.03 (2H, d, J=8.1 Hz), 9.66 (1H, bs). Ή-NMR (CDC1 3): 51. 8-1.62 (4H, m), 1.62-1.82 (2H, m), 1.79 (3H, s), 2.28 (3H, s), 2.97 (2H, t, J = 6.9 Hz), 4.44 (2H, dt, J = 47.1, 5.7 Hz), 7.37-7.53 (3H, m), 7.60 (2H, d, J-7.2 Hz), 7.70 (2H, d, J = 8.7 Hz) , 8.03 (2H, d, J = 8.1 Hz), 9.66 (1H, bs).
元素分析値: C25H26N303S3Fとして Elementary analysis: as C 25 H 26 N 3 0 3 S 3 F
計算値 (%) : C, 56.47; H, 4.93; N, 7.90; S, 18.09. Calculated value (%): C, 56.47; H, 4.93; N, 7.90; S, 18.09.
実測値 (¾) : C, 56.70; H, 5.02; N, 7.69; S, 18.39. Found (¾): C, 56.70; H, 5.02; N, 7.69; S, 18.39.
実施例 7-158 Example 7-158
N-{5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口 -1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン }ォク夕ンスルホンアミド 水素化ナトリウム(25.4 mg, 0.634 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5,6-ジメチル 8-[5 -フルォロ ペンチル]スルファニル] - 3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 3-ォ > (100 mg, 0.317 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに n-オクタンスルホニル クロリド (0.0931 ml, 0.476匪 ol) のテト ラヒドロフラン (1.00 ml) 溶液を加え、 さらに 45分間かき混ぜた。 反応混合物 に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶 媒を減圧下に留去して題記化合物 (97.5 mg, 62¾) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルエーテルから再結晶して ¾点 1'21_123°Cの暗赤色結晶を得た。 IR (KBr) V: 3196, 2924, 2857, 1755, 1665, 1626, 1570 cm— N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dialdehyde Mouth-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene} sulfonamide A suspension of sodium hydride (25.4 mg, 0.634 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added to the triamino-5,6 obtained in Example 6-23. -Dimethyl 8- [5-fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-o> (100 mg, 0.317 mmol) in tetrahydrofuran (1.00 ml) was added. The mixture was stirred for 5 minutes under ice cooling. To this was added n-octanesulfonyl chloride (0.0931 ml, 0.476 A solution of lahydrofuran (1.00 ml) was added, and the mixture was further stirred for 45 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the title compound was evaporated Solvent under vacuum (97.5 mg, 62¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a point 1'21_123 ° C. IR (KBr) V: 3196, 2924, 2857, 1755, 1665, 1626, 1570 cm—
'H-NMR (CDC13) : 50.882 (3H, t, J=6.4 Hz), 1.15-2.00 (18H, m), 1.81 (3H, s), 2.28 (3H, s), 2.99 (2H, t, J=7.4 Hz), 3.11 (2H, t, J=7.8 Hz), 4.46 (2H, dt, J=46.8, 6.4 Hz), 9.46 (1H, bs). 'H-NMR (CDC1 3) : 50.882 (3H, t, J = 6.4 Hz), 1.15-2.00 (18H, m), 1.81 (3H, s), 2.28 (3H, s), 2.99 (2H, t, J = 7.4 Hz), 3.11 (2H, t, J = 7.8 Hz), 4.46 (2H, dt, J = 46.8, 6.4 Hz), 9.46 (1H, bs).
元素分析値: C21H34N303S3Fとして Elementary analysis: as C 21 H 34 N 3 0 3 S 3 F
計算値 (%) : C, 51.30; H, 6.97; N, 8.55; S, 19.56. Calculated (%): C, 51.30; H, 6.97; N, 8.55; S, 19.56.
実測値 ( : C, 51.27; H, 7.19; N, 8.31; S, 19.64. Observed (: C, 51.27; H, 7.19; N, 8.31; S, 19.64.
実施例 7-159 Example 7-159
4-ter卜ブチル -N- {5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニル ]-3 -ォ キソ -2 , 3-ジヒドロ _ 1H-イミダゾ [5 ,卜 c] [ 1, 4]チアジン- 1 -イリデン }ベンゼ'ンス ルホンアミド 4-tert-butyl-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro_1H-imidazo [5, tric] [1,4 ] Thiazine-1-ylidene} benzen's sulfonamide
水素化ナトリウム(50.7mg, 1.27腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6-ジメチル 8-[5-フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5, 1- c] [1,4]チアジン- 3-オン (200 mg, 0.634mmol) のテトラヒドロフラン (2.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 4- tert-ブチルベンゼンスルホニル クロリド (226 mg, 0.951 mmoi) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1時間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (289fflg, 89¾) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルェ一テルから再結晶して融点 199- 201 の暗赤色結晶を得た。 IR (KBr) レ : 3260, 2963, 1742, 1728, 1663, 1626, 1561 cm-1. To a suspension of sodium hydride (50.7 mg, 1.27 t ol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl 8- [5- Add a solution of fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (200 mg, 0.634 mmol) in tetrahydrofuran (2.00 ml) and stir under ice-cooling for 5 minutes. I did. To this was added a solution of 4-tert-butylbenzenesulfonyl chloride (226 mg, 0.951 mmoi) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (289fflg, 89¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 199-201. IR (KBr) level: 3260, 2963, 1742, 1728, 1663, 1626, 1561 cm- 1 .
Ή-NMR (CDC13) : δ 1.33 (9Η, s), 1.50-1.62 (2H, m), 1.64-1.84 (4H, m), 1.79 (3H, s), 2.26 (3H, d, J=0.9Hz), 2.96 (2H, t, J=7.5 Hz), 4.45 (2H, dt, J=47.1, 5.7 Hz), 7.49 (2H, d, J=8.1 Hz), 7.87 (2H, d, J=8.4 Hz), 9.62 (1H, bs). 元素分析値: C23H3。N303S3Fとして 計算値 (¾) : C, 53.99; H, 5.91; , 8.21; S, 18.80. Ή-NMR (CDC1 3): δ 1.33 (9Η, s), 1.50-1.62 (2H, m), 1.64-1.84 (4H, m), 1.79 (3H, s), 2.26 (3H, d, J = 0.9 Hz), 2.96 (2H, t, J = 7.5 Hz), 4.45 (2H, dt, J = 47.1, 5.7 Hz), 7.49 (2H, d, J = 8.1 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.62 (1H, bs ) elemental analysis:. C 23 H 3. N 3 0 3 S 3 F Calculated (¾): C, 53.99; H, 5.91;, 8.21; S, 18.80.
実測値 (%) : C, 53.98; H, 5.74; N, 8.23; S, 18.82. Found (%): C, 53.98; H, 5.74; N, 8.23; S, 18.82.
実施例 7-160 Example 7-160
(E)-N-{5, 6-ジメチル- 8-[(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3 -ジ ヒドロ- 1H-イミダゾ [5,1-c] [1, 4]チアジン-卜ィリデン卜 2 -フエニルエタンスル ホンアミド (E) -N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-triiden 2 -phenylethanesulfonamide
水素化ナトリウム(50.7mg, 1.27mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6 -ジメチル 8- [5-フルォロ ペンチル]スルファニル ]-3H -イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (200 mg, 0.634讓 ol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに trans_)3-スチレンスルホンスルホニル クロリド (199 mg, 0.951 翻 ol) のテトラヒドロフラン (1.00 溶液を加え、 さらに 1時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(226 mg, 74%)を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 87.0-89. (TCの暗赤色結晶を 得た。 To a suspension of sodium hydride (50.7 mg, 1.27 mmol) (60% oily) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl 8- [5-fluoro obtained in Example 6-23. Add a solution of [pentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (200 mg, 0.634 benzyl) in tetrahydrofuran (2.00 ml) and stir under ice-cooling for 5 minutes. Was. To this was added a solution of trans_) 3-styrenesulfonesulfonyl chloride (199 mg, 0.951 ol) in tetrahydrofuran (1.00), and the mixture was stirred for 1 hour. 1N hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. , dried (MgSO 4), to give the title compound (226 mg, 74%) as a dark red crystals and the solvent was distilled off under reduced pressure acetate Echiru -. mp recrystallization from Jeffrey chill ether 87.0-89. (A dark red crystal of TC was obtained.
IR (KBr) V 3260, 2932, 1736, 1663, 1611, 1557 cm"1. IR (KBr) V 3260, 2932, 1736, 1663, 1611, 1557 cm " 1 .
Ή-NMR (CDC13) : δ 1.46-1.68 (2Η, m), 1.68 - 1.88 (4H, m), 1.81 (3H, d, J=1.2 Hz), 2.28 (3H, d, J=1.2 Hz), 2.99 (2H, t, J=7.8 Hz), 4.44 (2H, dt, J=47.2, 6.0 Hz), 6.94 (1H, d, J=15.4 Hz), 7.36-7.44 (3H, m), 7.46-7.54 (2H, m), 7.56 (1H, d, J=15.4 Hz), 9.58 (1H, bs). Ή-NMR (CDC1 3): δ 1.46-1.68 (2Η, m), 1.68 - 1.88 (4H, m), 1.81 (3H, d, J = 1.2 Hz), 2.28 (3H, d, J = 1.2 Hz) , 2.99 (2H, t, J = 7.8 Hz), 4.44 (2H, dt, J = 47.2, 6.0 Hz), 6.94 (1H, d, J = 15.4 Hz), 7.36-7.44 (3H, m), 7.46- 7.54 (2H, m), 7.56 (1H, d, J = 15.4 Hz), 9.58 (1H, bs).
元素分析値: C21H24N303S3Fとして Elementary analysis: as C 21 H 24 N 3 0 3 S 3 F
計算値 (¾) : C, 52.37; H, ,5.02; N, 8.72; S, 19.97. Calculated value (¾): C, 52.37; H,, 5.02; N, 8.72; S, 19.97.
実測値 (¾) : C, 52.11; H, 4.74; N, 8.72; S, 20.13. Found (¾): C, 52.11; H, 4.74; N, 8.72; S, 20.13.
実施例 7-161 Example 7-161
N- {5, 6-ジメチル- 8- [ (5-フルォ口ペンチル)スルファニル] -3-ォキソ- 2, 3-ジヒド 口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン }- 4-フエノキシベンゼンスル ホンアミド N- {5, 6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydropenit-1H-imidazo [5,1-c] [1,4] thiazine- Trilidene}-4-phenoxybenzenesulfonamide
水素化ナトリウム(25.4 mg, 0.634匪 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 23で得られた卜ァミノ- 5, 6 -ジメチル 8_[5 -フルォロ ぺンチル]スルファニル] -3H-ィミダゾ [5, 1 - c] [1 , 4]チアジン- 3-ォン (100 Dig, 0.317imol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 4-フエノキシベンゼンスルホニル クロリ ド (130 mg, 0.476腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 20分間かき混ぜた。 反応 混合物に 塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (Mgso4) 後、溶媒を減圧下に留去して題記化合物(124 mg, 72%)を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 152-154°Cの暗赤色結晶を得 た。 Sodium hydride (25.4 mg, 0.634 ol) (60% oily) in tetrahydrofuran (2.00 ml) of the suspension obtained in Example 6-23, the triamino-5,6-dimethyl 8_ [5-fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] ] A solution of thiazine-3-one (100 Dig, 0.317imol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-phenoxybenzenesulfonyl chloride (130 mg, 0.476 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 20 minutes. Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. After the extract was washed with water and dried (Mgso 4 ), the solvent was distilled off under reduced pressure to obtain the title compound (124 mg, 72%) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 152-154 ° C.
IR (KBr) V: 3240, 2936, 1740, 1665, 1615, 1563 cm1. IR (KBr) V: 3240, 2936, 1740, 1665, 1615, 1563 cm 1 .
ΐ-NMR (CDC13) : 51.45-1.85 (6H, m), 1.79 (3H, s), 2.27 (3H, s), 2.96 (2H, t, J=7.4 Hz), 4.44 (2H, dt, J=47.2, 5.8 Hz), 7.02 (2H, d, J=8.8 Hz), 7.04 (2H, dd, J=9.0, 1.0 Hz), 7.21 (1H, t, J=7.6 Hz), 7.40 (2H, t, J=8.0 Hz) , 7.91 (2H, d, J=8.8 Hz), 9.61 (1H, bs). ΐ-NMR (CDC1 3): 51.45-1.85 (6H, m), 1.79 (3H, s), 2.27 (3H, s), 2.96 (2H, t, J = 7.4 Hz), 4.44 (2H, dt, J = 47.2, 5.8 Hz), 7.02 (2H, d, J = 8.8 Hz), 7.04 (2H, dd, J = 9.0, 1.0 Hz), 7.21 (1H, t, J = 7.6 Hz), 7.40 (2H, t , J = 8.0 Hz), 7.91 (2H, d, J = 8.8 Hz), 9.61 (1H, bs).
元素分析値: C25H26N304S3Fとして . Elementary analysis: as C 25 H 26 N 3 0 4 S 3 F.
計算値 (%) : C, 54.82; H, 4.78; N, 7.67; S, 17.56. Calculated (%): C, 54.82; H, 4.78; N, 7.67; S, 17.56.
実測値 (%) : C, 54.92; H, 4.84; N, 7.72; S, 17.60. Found (%): C, 54.92; H, 4.84; N, 7.72; S, 17.60.
実施例 7-162 ' ェチル 4- [({5, 6-ジメチル- 8- [ (5 フルォロぺンチル)スルファニル] -3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン-卜イリデン }ァミノ)スルホ二 ル]ベンゾェ一卜 Example 7-162'ethyl 4-[({5,6-dimethyl-8-[(5fluoropentanyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene} amino) sulfonyl] benzoate
水素化ナトリウム(76.1mg, 1.90匪 ol) (60%油性)のテトラヒドロフラン(4.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5,6-ジメチル 8 - [5-フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (300 mg, O.951mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにェチル (4-クロロスルホニル)ベンゾェート (284 mg, 1.14 mmol) のテトラヒドロフラン (2.00 ml') 溶液を加え、 さらに 2時間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (360 mg, 75¾) を暗赤色結晶として得た。 酢 酸ェチル-ジェチルエーテルから再結晶して融点 4- 146 の暗赤色結晶を得た。 IR (KBr) リ: 3227, 2926, 1759, 1723, 1663, 1622, 1559 cm"1. To a suspension of sodium hydride (76.1 mg, 1.90 ol) (60% oil) in tetrahydrofuran (4.00 ml) was added 1-amino-5,6-dimethyl 8- [5] obtained in Example 6-23. -Fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (300 mg, O.951mmol) in tetrahydrofuran (2.00 ml) was added, and the mixture was cooled on ice for 5 minutes. Stirred. To this was added a solution of ethyl (4-chlorosulfonyl) benzoate (284 mg, 1.14 mmol) in tetrahydrofuran (2.00 ml '), and the mixture was further stirred for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (360 mg, 75¾) as a dark red crystals. vinegar The crystals were recrystallized from acetic acid-ethyl ether to obtain dark red crystals having a melting point of 4-146. IR (KBr): 3227, 2926, 1759, 1723, 1663, 1622, 1559 cm " 1 .
Ή-NMR (CDC13) : 61.41 (3H, t, J=7.0 Hz), 1.48-1.84 (6H, m), 1.80 (3H, d, J=1.0 Hz), 2.28 (3H, d, J=1.0 Hz), 2.97 (2H, t, J=7. Hz), 4.41 (2H, q, J=7.0 Hz), 4.45 (2H, dt, J=47.2, 5.8 Hz), 8.03 (2H, d, J=8.8Hz), 8.15 (2H, d, J=8.8 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): 61.41 (3H, t, J = 7.0 Hz), 1.48-1.84 (6H, m), 1.80 (3H, d, J = 1.0 Hz), 2.28 (3H, d, J = 1.0 Hz), 2.97 (2H, t, J = 7.Hz), 4.41 (2H, q, J = 7.0 Hz), 4.45 (2H, dt, J = 47.2, 5.8 Hz), 8.03 (2H, d, J = 8.8Hz), 8.15 (2H, d, J = 8.8 Hz), 9.61 (1H, bs).
元素分析値: C22H26N305S3Fとして Elementary analysis: as C 22 H 26 N 3 0 5 S 3 F
計算値 (%) : C, 50.08; H, 4.97; N, 7.96; S, 18.23. Calculated value (%): C, 50.08; H, 4.97; N, 7.96; S, 18.23.
実測値 (¾) : C, 50.22; H, 5.10; N, 8.01; S, 18.32. Found (¾): C, 50.22; H, 5.10; N, 8.01; S, 18.32.
実施例 7463 ' Example 7463 '
N-{5, 6-ジメチル- 8 - [(5-フルォロペンチル)スルファニル ]-3 -ォキソ -2, 3-ジヒド ロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1 -ィリデン }- 4-モルホリンスルホンアミ ド、 N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene}-4-morpholine sulfonamide,
水素化ナトリゥム(38.0 mg, 0.951匪 oi) (60 油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6-ジメチル 8-[5-ヅルォ口 ペンチル]スルファニル ]- 3H-ィミダゾ [5, 1-c] [1,4]チアジン- 3-オン (150 mg, 0.476腿 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 4-モルホリンスルホニル クロリド (106 mg, 0.571 匪 ol) のテト ラヒドロフラン (1.00 ml) 溶液を加え、 さらに 25分間かき混ぜた。 反応混合物 に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶 媒を減圧下に留去した。 残留物をシリカゲル力ラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (41.7 mg, 19%) を 暗赤色結晶として得た。 酢酸エヂル-ジェチルエーテルから再結晶して融点 191- 193 の暗赤色結晶を得た。 A suspension of hydrogenated sodium (38.0 mg, 0.951 oi) (60 oily) in tetrahydrofuran (2.00 ml) was added to the triamino-5,6-dimethyl 8- [5-phenol) obtained in Example 6-23. Mouth Pentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (150 mg, 0.476 tmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. mixed. To this was added a solution of 4-morpholine sulfonyl chloride (106 mg, 0.571 ol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 25 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), followed by distilling off the solvent medium under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (41.7 mg, 19%) as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 191-193.
IR (KBr) v: 3250, 2961, 2924, 2863, 1744, 1665, 1624, 1568 cm—1. IR (KBr) v: 3250, 2961, 2924, 2863, 1744, 1665, 1624, 1568 cm— 1 .
'H-NMR (CDCI3) : δ 1.50-1.66 (2Η, m), 1.66-1.85 (4H, m), 1.81 (3H, s), 2.28 (3H, s), 3.00 (2H, t, J=7.5 Hz), 3.10-3.18 (4H, m), 3.75-3.84 (4H, m), 4.46 (2H, dt, J=46.8, 6.0 Hz), 9.23 (1H, bs) . 'H-NMR (CDCI3): δ 1.50-1.66 (2Η, m), 1.66-1.85 (4H, m), 1.81 (3H, s), 2.28 (3H, s), 3.00 (2H, t, J = 7.5 Hz), 3.10-3.18 (4H, m), 3.75-3.84 (4H, m), 4.46 (2H, dt, J = 46.8, 6.0 Hz), 9.23 (1H, bs).
元素分析値: C17H25N40S?Fとして 計算値 (¾) : C, 43.95; H, 5.42; N, 12.06; S, 20.70. Elemental analysis: as C 17 H 25 N 4 0S F ? Calculated value (¾): C, 43.95; H, 5.42; N, 12.06; S, 20.70.
実測値 (%) : C, 43.94; H, 5.49; N, 11.90; S, 20.42. Found (%): C, 43.94; H, 5.49; N, 11.90; S, 20.42.
実施例 7-164 Example 7-164
ェチル 2- [({5, 6-ジメチル- 8 - [ (5-フルォ口ペンチル)スルファニル] - 3 -ォキソ - 2, 3-ジヒドロ -1H-イミダゾ [5,ト c] [1, 4]チアジン-卜ィリデン }ァミノ)スルホ二 ル] -1-シクロへキセン - 1 -力ルポキシレート Ethyl 2-[({5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, toc] [1,4] thiazine -Triylidene} amino) sulfonyl] -1-cyclohexene-1
水素化ナトリウム(52.8mg, 1.32腿01) (60%油性)のテトラヒドロフラン(4.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6-ジメチル 8 - [5-フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5,1- c] [1, 4]チアジン -3-オン (208 mg, 0.951腿 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにェチル (2-クロロスルホニル)シク口へキセン- 1-カルポキシレ一卜 (200 ig, 0.791匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 1 時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を 水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (323 mg, 92¾) を 暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 149-151°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (52.8 mg, 1.32 thigh 01) (60% oily) in tetrahydrofuran (4.00 ml) was added the triamino-5,6-dimethyl 8- [5- [5- Fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (208 mg, 0.951 tmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. mixed. To this was added a solution of ethyl (2-chlorosulfonyl) cyclohexene-1-carboxylate (200 ig, 0.791 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (323 mg, 92¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 149-151 ° C.
IR (KBr) リ: 2938, 2865, 1748, 1663, 1651, 1609, 1559 cm"1. IR (KBr): 2938, 2865, 1748, 1663, 1651, 1609, 1559 cm " 1 .
¾一 NMR (CDC13) : 51.34 (3H, t, J=7.2 Hz), 1.50-1.64 (2H, m), 1.64-1.84 (4H, m), 1.81 (3H, d, J=1.2 Hz), 2.29 (3H, d, J=0.9 Hz), 2.38-2.44 (2H, m), 2.48-2.56 (2H, m) , 2.98 (2H, t, J=7.2 Hz), 4.32 (2H, q, J=6.9 Hz), 4.46 (2H, dt, J=46.8, 5.7 Hz), 9.99 (1H, bs). ¾ one NMR (CDC1 3): 51.34 ( 3H, t, J = 7.2 Hz), 1.50-1.64 (2H, m), 1.64-1.84 (4H, m), 1.81 (3H, d, J = 1.2 Hz), 2.29 (3H, d, J = 0.9 Hz), 2.38-2.44 (2H, m), 2.48-2.56 (2H, m), 2.98 (2H, t, J = 7.2 Hz), 4.32 (2H, q, J = 6.9 Hz), 4.46 (2H, dt, J = 46.8, 5.7 Hz), 9.99 (1H, bs).
元素分析値: C22H3。N305S3Fとして Elemental analysis: C 22 H 3. N 3 0 5 S 3 F
計算値 (¾) : C, 49.70; H, 5.69; N, 7.90; S, 18.09. Calculated value (¾): C, 49.70; H, 5.69; N, 7.90; S, 18.09.
実測値 (¾) : C, 49.69; H, 5.57; N, 7.90; S, 18.30. Found (¾): C, 49.69; H, 5.57; N, 7.90; S, 18.30.
実施例 7- 165 Example 7-165
N - {5,6-ジメチル _8-[(5-フルォロペンチル)スルファニル ]- 3-ォキソ - 2,3-ジヒド ロ- 1H-イミダゾ [5, 1- c] [1,4]チアジン-卜イリデン プロパンスルホンアミド 水素化ナトリゥム (25.4 mg, 0.634画 1) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6 - 23で得られた卜アミノ- 5, 6-ジメチル 8- [5 -フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (100 mg, 0.317腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 2-プロパンスルホニル クロリド (0.0503 ml, 0.476匪 ol) のテト ラヒドロフラン (1.00 ml) 溶液を加え、 さらに 25分間かき混ぜた。 反応混合物 に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶 媒を減圧下に留去して題記化合物 (75.2 mg, 56¾) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルェ一テルから再結晶して融点 5-147°Cの暗赤色結晶を得た。 IR (KBr) V: 3200, 2973, 2938, 1753, 1665, 1624, 1568 cm"1. N- {5,6-dimethyl_8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidenepropane Sulfonamide Hydrogenated sodium (25.4 mg, 0.634 fraction 1) (60% oil) in tetrahydrofuran (2.00 ml) was added to a suspension of triamino-5,6-dimethyl 8- [obtained in Examples 6-23. 5-Fluoro Add a solution of [pentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.317 tmol) in tetrahydrofuran (1.00 ml), and stir under ice-cooling for 5 minutes. Was. To this was added a solution of 2-propanesulfonyl chloride (0.0503 ml, 0.476 ol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 25 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the title compound was evaporated Solvent under vacuum (75.2 mg, 56¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 5-147 ° C. IR (KBr) V: 3200, 2973, 2938, 1753, 1665, 1624, 1568 cm " 1 .
'Η -丽 R (CDCI3) : άΐ.42 (6Η, d, J=6.6 Hz), 1.50-1.64 (2H, m), 1.64-1.84 (4H, m), 1.81 (3H, s), 2.28 (3H, s), 2.99 (2H, t, J=7.2 Hz), 3.22 (1H, quintet, J=6.3 Hz), 4.45 (2H, dt, J=47.1, 5.7 Hz), 9.46 (1H, bs) . 'Η-丽 R (CDCI 3 ): άΐ.42 (6Η, d, J = 6.6 Hz), 1.50-1.64 (2H, m), 1.64-1.84 (4H, m), 1.81 (3H, s), 2.28 (3H, s), 2.99 (2H, t, J = 7.2 Hz), 3.22 (1H, quintet, J = 6.3 Hz), 4.45 (2H, dt, J = 47.1, 5.7 Hz), 9.46 (1H, bs) .
元素分析値: C16H2 03S3Fとして Elemental analysis: as C 16 H 2 0 3 S 3 F
計算値 (%) : C, 45.58; H, 5.74; N, 9.97; S, 22.82. Calculated value (%): C, 45.58; H, 5.74; N, 9.97; S, 22.82.
実測値 (%) : C, 45.37; H, 5.84; N, 10.07; S, 22.58. Found (%): C, 45.37; H, 5.84; N, 10.07; S, 22.58.
実施例 7-166 Example 7-166
4 -ブトキシ- N- {5, 6-ジメチル- 8- [ (5-フルォロぺンチル)スルファエル] -3-ォキソ -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン }ベンゼンスルホ ンアミド 4-butoxy-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfael] -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1, 4] thiazine-triylidene} benzenesulfonamide
水素化ナトリウム(25.4 mg, 0.634匪 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 23で得られた卜ァミノ -5, 6-ジメチル 8_[5-フルォロ ペンチル]スルファニル] -3H-ィミダゾ [5, 1-c] [1 , 4]チアジン -3-ォン (100 rag, 0.317腿 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 4-ブトキシベンゼンスルホニル クロリド (122 mg, 0.476匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 50分間かき混ぜた。 反応 混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(121 mg, 73¾)を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 140- 142での暗赤色結晶を得 た。 To a suspension of sodium hydride (25.4 mg, 0.634 ol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl 8_ [5-fluoro obtained in Example 6-23. Pentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine-3-one (100 rag, 0.317 t ol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice-cooling for 5 minutes. mixed. To this was added a solution of 4-butoxybenzenesulfonyl chloride (122 mg, 0.476 ol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 50 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (121 mg, 73¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 140-142.
IR (KBr) V 3210, 2961, 1752, 1715, 1663, 1622, 1595, 1566 cm—1. 'H-NMR (CDCI3) : (50.978 (3H, t, J=7.5 Hz), 1.-40-1.62 (4H, m), 1.62-1.75 (6H, m), 1.79 (3H, s), 2.26 (3H, d, J-0.9 Hz), 2.94.(2H, t, J=7.5 Hz), 4.00 (2H, t, J=6.3 Hz), 4.44 (2H, dt, J=47.1, 5.7 Hz), 6.93 (2H, d, J=8.7 Hz), 7.86 (2H, d, J=9.3 Hz), 9.59 (1H, bs). IR (KBr) V 3210, 2961, 1752, 1715, 1663, 1622, 1595, 1566 cm- 1 . 'H-NMR (CDCI3): (50.978 (3H, t, J = 7.5 Hz), 1.-40-1.62 (4H, m), 1.62-1.75 (6H, m), 1.79 (3H, s), 2.26 (3H, d, J-0.9 Hz), 2.94. (2H, t, J = 7.5 Hz), 4.00 (2H, t, J = 6.3 Hz), 4.44 (2H, dt, J = 47.1, 5.7 Hz), 6.93 (2H, d, J = 8.7 Hz), 7.86 (2H, d, J = 9.3 Hz), 9.59 (1H, bs).
元素分析値: C23H3QN304S3Fとして Elementary analysis: as C 23 H 3Q N 3 0 4 S 3 F
計算値 (%) : C, 52.35; H, 5.73; N, 7.96; S, 18.23. Calculated (%): C, 52.35; H, 5.73; N, 7.96; S, 18.23.
実測値 (¾) : C, 52.05; H, 5.72; N, 7.79; S, 18.09. Found (¾): C, 52.05; H, 5.72; N, 7.79; S, 18.09.
実施例 7-167 Example 7-167
(4 -プロモフエニル) -N- {5, 6-ジメチル- 8- [ (5-フルォロペンチル)スルファニ ル]- 3-ォキソ -2, 3-ジヒドロ- m-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン }メ 夕ンスルホンアミド (4-Promophenyl) -N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-m-imidazo [5, tric] [1, 4 ] Thiazine-1-ylidene
実施例 7-166と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 185- ISrCの喑赤色結晶を得た。 The compound was synthesized in the same manner as in Example 7-166, and recrystallized from ethyl acetate-getyl ether to obtain Δ red crystals having a melting point of 185-ISrC.
IR (KBr) V : 3225, 2938, 1742, 1665, 1617, 1564, 1487 cm—1. IR (KBr) V: 3225, 2938, 1742, 1665, 1617, 1564, 1487 cm— 1 .
¾— NMR (CDCI3) : 51.48-1.90 (6H, m), 1.80 (3H, d, J=0.8 Hz), 2.24 (3H, d,¾— NMR (CDCI3): 51.48-1.90 (6H, m), 1.80 (3H, d, J = 0.8 Hz), 2.24 (3H, d,
J=1.0 Hz), 3.00 (2H, t, J=7.2 Hz), 4.30 (2H, s), 4.49 (2H, dt, J=47.2, 5.8J = 1.0 Hz), 3.00 (2H, t, J = 7.2 Hz), 4.30 (2H, s), 4.49 (2H, dt, J = 47.2, 5.8
Hz), 7.31 (2H, d, J=8. Hz), 7.48 (2H, d, J=8.6 Hz), 9.21 (1H, bs). Hz), 7.31 (2H, d, J = 8 Hz), 7.48 (2H, d, J = 8.6 Hz), 9.21 (1H, bs).
元素分析値: C2。H23N303S3BrFとして Elemental analysis: C 2. H 23 N 3 0 3 S 3 As BrF
計算値 (¾) : C, 43.79; H, 4.23; N, 7.66; S, 17.54. Calculated value (¾): C, 43.79; H, 4.23; N, 7.66; S, 17.54.
実測値 (%) : C, 43.81; H, 4.19; N, 7.64; S, 17.44. Found (%): C, 43.81; H, 4.19; N, 7.64; S, 17.44.
実施例 7-168 Example 7-168
[ (1R, 3S, 7R)- 3 -ブロモ - 1, 7 -ジメチル- 2 -ォキソビシクロ [2, 2, 1]ヘプト- 7 -ィ ル] - N - {5, 6 -ジメチル- 3-ォキソ- 8 - [ (5 -フル才口ぺンチル)スルファニル] -2, 3-ジ ヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }メタンスルホンアミド 水素化ナトリウム(39.5 mg, 0.987膽 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6 - 23で得られた卜ァミノ- 5, 6 -ジメチル 8- [5-フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5,1- c] [1,4]チアジン- 3-オン (150 mg, 0.49.3腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 とれに参考例 48で得られた D,(+)-3-ブロモ -カンファ一- 8-スルホニル ク ロリドのテトラヒドロフラン(1.00 ml)溶液を加え、さらに 30分間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、溶媒を減圧下に留去て題記化合物(82.8mg, 28%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 161-163°Cの喑赤色結晶を得 た。 [(1R, 3S, 7R) -3-Bromo-1,7-dimethyl-2-oxobicyclo [2,2,1] hept-7-yl] -N- {5,6-dimethyl-3-oxo- 8-[(5-full-age pentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene} methanesulfonamide sodium hydride ( To a suspension of 39.5 mg, 0.987 benzyl) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl 8- [5-fluoropentyl] sulfanyl obtained in Example 6-23. ] -3H-Imidazo [5,1-c] [1,4] thiazin-3-one (150 mg, 0.49.3 tmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice-cooling for 5 minutes. Was. However, D, (+)-3-bromo-campha-1-sulfonylc obtained in Reference Example 48 A solution of chloride in tetrahydrofuran (1.00 ml) was added, and the mixture was further stirred for 30 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the title compound solvent Te distilled off under reduced pressure (82.8 mg, 28%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to give reddish crystals having a melting point of 161-163 ° C.
IR ( Br) リ: 3229, 2965, 2934, 1752, 1665, 1618, 1564 cm一1. IR (Br): 3229, 2965, 2934, 1752, 1665, 1618, 1564 cm- 1 .
Ή-NMR (CDC13) : (50.997 (3H, s), 1.29 (3H, s), 1.46-1.90 (8H, m), 1.82 (3H, s), 2.14-2.26 (2H, m), 2.29 (3H, s), 3.01 (2H, t, J=7.8 Hz), 3.11 (IH, d, J=14.7 Hz), 3.20-3.28 (IH, m), 3.48 (IH, d, J=14. Hz), 4.46 (2H, dt, J=47.1, 5.7 Hz), 4.62 (IH, d, J=3.0 Hz), 9.56 (IH, bs). Ή-NMR (CDC1 3): (50.997 (3H, s), 1.29 (3H, s), 1.46-1.90 (8H, m), 1.82 (3H, s), 2.14-2.26 (2H, m), 2.29 ( 3H, s), 3.01 (2H, t, J = 7.8 Hz), 3.11 (IH, d, J = 14.7 Hz), 3.20-3.28 (IH, m), 3.48 (IH, d, J = 14.Hz) , 4.46 (2H, dt, J = 47.1, 5.7 Hz), 4.62 (IH, d, J = 3.0 Hz), 9.56 (IH, bs).
元素分析値: C23H31N304S3BrFとして Elementary analysis: as C 23 H 31 N 3 0 4 S 3 BrF
計算値 (¾) : C, 45.39; H, 5.13; N, 6.90; S, 15.81. Calculated value (¾): C, 45.39; H, 5.13; N, 6.90; S, 15.81.
実測値 (¾) : C, 45.25; H, 5.04; N, 6.92; S, 16.06. Found (¾): C, 45.25; H, 5.04; N, 6.92; S, 16.06.
実施例 7-169 Example 7-169
N - {5, 6-ジメチル- 8- [ (5-フルォロペンチル)スルファニル] -3-ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5, 1- c] [1, 4]チアジン-卜イリデン卜 2-フエニルエタンスルホン アミド N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine-triylidene 2-phenylethanesulfonamide
実施例 7-168と同様にして合成し、酢酸ェチル-ジェチルエーテルから再結晶し て融点 159-161^の暗赤色結晶を得た。 The compound was synthesized in the same manner as in Example 7-168, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 159-161 ^.
IR (KBr) リ: 3175, 2963, 2942, 1755, 1665, 1622, 1568 cm— IR (KBr): 3175, 2963, 2942, 1755, 1665, 1622, 1568 cm—
Ή-NMR (CDC13) : 51.48-1.88 (6H, m), 1.82 (3H, s), 2.28 (3H, d, 1=1.2 Hz), Ή-NMR (CDC1 3): 51.48-1.88 (6H, m), 1.82 (3H, s), 2.28 (3H, d, 1 = 1.2 Hz),
2.99 (2H, t, J=7.5 Hz), 3.16-3.25 (2H, m), 3.38-3.48 (2H, m), 4.43. (2H, dt,2.99 (2H, t, J = 7.5 Hz), 3.16-3.25 (2H, m), 3.38-3.48 (2H, m), 4.43. (2H, dt,
J=47.1, 6.0 Hz), 7.18-7.35 (5H, m), 9.44 (IH, bs). J = 47.1, 6.0 Hz), 7.18-7.35 (5H, m), 9.44 (IH, bs).
元素分析値: C21H26N303S3F Ο·7Η20として Elemental analysis: C 21 H 26 N 3 0 3 S 3 F Ο · 7Η 20
計算値 : C, 50.83; Η, 5.57; Ν, 8.47. ' Calculated: C, 50.83; Η, 5.57; Ν, 8.47. '
実測値 (¾) : C, 51.14; Η, 5.37; Ν, 8.62. Found (¾): C, 51.14; Η, 5.37; Ν, 8.62.
実施例 7-170 Example 7-170
C(1S, 4R)-7, 7 -ジメチル -2-ォキソビシク口 [2, 2, 1]ヘプト- 1 -ィル] - N-{5, 6 -ジメ チル -8- [ (5-フルォ口ペンチル)スルファニル] - 3 -ォキソ- 2, 3-ジヒド口- 1H-ィミ ダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン }メタンスルホンアミド C (1S, 4R) -7,7-dimethyl-2-oxovisic mouth [2,2,1] hept-1-yl] -N- {5,6-dimethyl-8-[(5-fluoro mouth Pentyl) sulfanyl] -3 -oxo-2,3-dihydrido-1H-imi Dazo [5,1-c] [1,4] thiazine-1-ylidene} methanesulfonamide
水素化ナトリウム(25.4 mg, 0.634 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6-ジメチル 8-[5_フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5, 1-c] [1, 4]チアジン - 3-オン (100 mg, 0.317mmol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに (+)-10-カンファースルホニル クロリド (122 mg, 0.476腿 ol) の テトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 30分間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去て題記化合物 (151 mg, 90%) を暗赤色結晶として得た。 酢酸 ェチル -ジェチルェ一テルから再結晶して融点 157-159°Cの暗赤色結晶を得た。 IR (KBr) V: 3207, 2961, 1744, 1752, 1665, 1617, 1566 cm—1. To a suspension of sodium hydride (25.4 mg, 0.634 mmol) (60% oily) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl 8- [5_fluoro obtained in Example 6-23. Add a solution of [pentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 mg, 0.317 mmol) in tetrahydrofuran (1.00 ml), and stir under ice-cooling for 5 minutes. Was. To this was added a solution of (+)-10-camphorsulfonyl chloride (122 mg, 0.476 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 30 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain a solvent Te distilled off under reduced pressure the title compound (151 mg, 90%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 157-159 ° C. IR (KBr) V: 3207, 2961, 1744, 1752, 1665, 1617, 1566 cm— 1 .
ΐ-NMR (CDC13) : 50.867 (3H, s), 1.10 (3H, s), 1.43 (1H, ddd, J=12.0, 8.7, 3.6 Hz), 1.50-2.14 (10H, m), 1.81 (3H, s), 2.29 (3H, s), 2.38 (1H, dt, J=18.6, 3.6 Hz), 2.53 (1H, ddd, J=14.7, 8.5, 3.9 Hz), 2.98 (2H, t, J=7.5 Hz), 3.07 (1H, d, J=15.0 Hz), 3.61 (1H, d, J=14.7 Hz), 4.46 (2H, dt, J=47.1, 5.7 Hz), 9.77 (1H, bs). ΐ-NMR (CDC1 3): 50.867 (3H, s), 1.10 (3H, s), 1.43 (1H, ddd, J = 12.0, 8.7, 3.6 Hz), 1.50-2.14 (10H, m), 1.81 (3H , s), 2.29 (3H, s), 2.38 (1H, dt, J = 18.6, 3.6 Hz), 2.53 (1H, ddd, J = 14.7, 8.5, 3.9 Hz), 2.98 (2H, t, J = 7.5 Hz), 3.07 (1H, d, J = 15.0 Hz), 3.61 (1H, d, J = 14.7 Hz), 4.46 (2H, dt, J = 47.1, 5.7 Hz), 9.77 (1H, bs).
元素分析値: C23H32N304S3Fとして Elementary analysis: as C 23 H 32 N 3 0 4 S 3 F
計算値 (¾) : C, 52.15; H, 6.09; N, 7.93; S, 18.16. Calculated (¾): C, 52.15; H, 6.09; N, 7.93; S, 18.16.
実測値 (%) : C, 52.06; H, 6.39; N, 7.61; S, 18.51. Found (%): C, 52.06; H, 6.39; N, 7.61; S, 18.51.
実施例 7-171 Example 7-171
N- {5, 6-ジメチル -8- [ (5-フルォ口ぺンチル)スルファニル] - 3 -ォキソ- 2, 3-ジヒド 口- m -イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン } (4 -フルオロフェニル)メタン スルホンアミド ' 実施例 7-170と同様にして合成し、酢酸ェチル -ジェチルェ一テルから再結晶し て融点 184_186°Cの暗赤色結晶を得た。 N- {5,6-dimethyl-8-[(5-fluoromethylpentyl) sulfanyl] -3-oxo-2,3-dihydrido-m-imidazo [5,1-c] [1,4] thiazine -1-ylidene} (4-fluorophenyl) methanesulfonamide 'Synthesized in the same manner as in Example 7-170, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 184_186 ° C.
IR (KBr) V: 3200, 2938, 2865, 1738, 1665, 1617, 1564, 1508 cm-1. IR (KBr) V: 3200, 2938, 2865, 1738, 1665, 1617, 1564, 1508 cm- 1 .
'H-NMR (CDCI3) : 51.52-1.66 (4H, m), 1.66-1.86 (4H, m), 1.80 (3H, s), 2.23 (3H, s), 3.00 (2H, t, J=7.5 Hz), 4.31 (2H, s), 4.48 (2H, dt, J=47.1, 5.4 Hz), 7.03 (2H, t, J-8.7 Hz), 7.40 (2H, dd, J=8.4, 5.4 Hz), 9.19 (1H, bs). 元素分析値: C2。H23N303S3F2として 'H-NMR (CDCI3): 51.52-1.66 (4H, m), 1.66-1.86 (4H, m), 1.80 (3H, s), 2.23 (3H, s), 3.00 (2H, t, J = 7.5 Hz) ), 4.31 (2H, s), 4.48 (2H, dt, J = 47.1, 5.4 Hz), 7.03 (2H, t, J-8.7 Hz), 7.40 (2H, dd, J = 8.4, 5.4 Hz), 9.19 (1H, bs). Elemental analysis: C 2. H 23 N 3 0 3 As S 3 F 2
計算値 (%) : C, 49.26; H, 4.75; N, 8.62; S, 19.73. Calculated (%): C, 49.26; H, 4.75; N, 8.62; S, 19.73.
実測値 (%) : C, 49.03; H, 4.73; N, 8.44; S, 19.92. Found (%): C, 49.03; H, 4.73; N, 8.44; S, 19.92.
実施例 7-172 Example 7-172
(4-クロ口フエニル) -N-{5, 6 -ジメチル -8- [ (5-フルォロペンチル)スルファニ ル]- 3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5,1-c] [1,4]チアジン -1-ィリデン }メ タンスルホンアミド (4-chlorophenyl) -N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene} methansulfonamide
実施例 7-170と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 182-183 の暗赤色結晶を得た。 The compound was synthesized in the same manner as in Example 7-170, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 182-183.
IR (KBr) リ: 3210, 2960, 2940, 2860, 1742, 1665, 1617, 1564 cm—1. IR (KBr): 3210, 2960, 2940, 2860, 1742, 1665, 1617, 1564 cm— 1 .
'H-NMR (CDC13) : 51.50-1.68 (2H, m), 1.68-1.88 (4H, in), 1.80 (3H, s), 2.24 (3H, s), 2.99 (2H, t, J=7.5 Hz), 4.31 (2H, s), 4.48 (2H, dt, J=47.1, 5.7 Hz), 7.31 (2H, d, J=8.7 Hz), 7.37 (2H, d, J=8.4 Hz), 9.21 (1H, bs). 'H-NMR (CDC1 3) : 51.50-1.68 (2H, m), 1.68-1.88 (4H, in), 1.80 (3H, s), 2.24 (3H, s), 2.99 (2H, t, J = 7.5 Hz), 4.31 (2H, s), 4.48 (2H, dt, J = 47.1, 5.7 Hz), 7.31 (2H, d, J = 8.7 Hz), 7.37 (2H, d, J = 8.4 Hz), 9.21 ( 1H, bs).
元素分析値: C2。H23N303S3C1Fとして Elemental analysis: C 2. H 23 N 3 0 3 S 3 C1F
計算値 (%) : C, 47.66; Η, 4.60; Ν, 8.34; S, 19.08. Calculated value (%): C, 47.66; Η, 4.60; Ν, 8.34; S, 19.08.
実測値 (%) : C, 47.52; H, 4.55; N, 8.33; S, 18.83. Found (%): C, 47.52; H, 4.55; N, 8.33; S, 18.83.
実施例 7-173 Example 7-173
N-{5,6-ジメチル- 8-[(5-フルォロペンチル)スルファニル ]-3-ォキソ - 2,3-ジヒド 口- 1H-ィミダゾ [5,卜 c] [1,4]チアジン _1 -ィリデン } (4 -メチルフエニル) -メ夕ン スルホンアミド N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrone-1H-imidazo [5, tric] [1,4] thiazine_1-ylidene} (4-Methylphenyl) -methyl sulfonamide
実施例 7-170と同様にして合成し、酢酸ェチル -ジェチルェ一テルから再結晶し て融点 188- 190 の暗赤色結晶を得た。 The compound was synthesized in the same manner as in Examples 7-170, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 188-190.
IR (KBr) V: 3250, 2936, 2865, 1740, 1665, 1617, 1566 cm"1. IR (KBr) V: 3250, 2936, 2865, 1740, 1665, 1617, 1566 cm " 1 .
'H-NMR (CDCI3) : δ 1.54-1.68 (2Η, m), 1.68-1.86 (4H, m), 1.80 (3H, d, J=1.5 Hz), 2.21 (3H, d, J=0.9 Hz), 2.34 (3H, s), 3.01 (2H, t, J=7.5 Hz), 4.31 (2H, s), 4.48 (2H, dt, J=47.1, 6.0 Hz), 7.14 (2H, d, J=7.8 Hz), 7.29 (2H, d, J=8.1 Hz), 9.08 (1H, bs). 'H-NMR (CDCI3): δ 1.54-1.68 (2Η, m), 1.68-1.86 (4H, m), 1.80 (3H, d, J = 1.5 Hz), 2.21 (3H, d, J = 0.9 Hz) , 2.34 (3H, s), 3.01 (2H, t, J = 7.5 Hz), 4.31 (2H, s), 4.48 (2H, dt, J = 47.1, 6.0 Hz), 7.14 (2H, d, J = 7.8 Hz), 7.29 (2H, d, J = 8.1 Hz), 9.08 (1H, bs).
元素分析値: C21H26N303S3Fとして Elementary analysis: as C 21 H 26 N 3 0 3 S 3 F
計算値 (%) : C, 52.15; H, 5.42; N, 8.69; S, 19.89. 実測値 (%) : C, 52.32; H, 5.38; N, 8.75; S, 20.19. Calculated (%): C, 52.15; H, 5.42; N, 8.69; S, 19.89. Found (%): C, 52.32; H, 5.38; N, 8.75; S, 20.19.
実施例 7-174 Example 7-174
N - {5, 6-ジメチル -8- [ (5-フルォロペンチル)スルファニル] -3 -ォキソ -2, 3-ジヒド ロ- 1H-ィミダゾ [5, 1 - c] [1,4]チアジン- 1-ィリデン } - 2 -ピリジンスルホンアミド 実施例 7 - 170と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 151- 153°Cの暗赤色結晶を得た。 N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene} -2-pyridinesulfonamide Synthesized in the same manner as in Examples 7-170, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 151-153 ° C.
I (KBr) V: 3233, 2913, 1728, 1661, 1607, 1551 cm"1. I (KBr) V: 3233, 2913, 1728, 1661, 1607, 1551 cm " 1 .
Ή-N R (CDC13) : δ 1.46-1.60 (2Η, m), 1.60-1.84 (4H, m), 1.80 (3H, s), 2.30 (3H, s), 2.95 (2H, t, J=7.5 Hz), 4.43 (2H, dt, J=47.1, 6.0 Hz), 7.49 (IH, ddd, J=7.5, 4.5, 0.6 Hz), 7.91 (IH, dt, J=7.8, 1.5 Hz), 8.12 (1H, d, J=7.8 Hz), 8.66 (IH, d, J-4.5 Hz), 10.26 (IH, bs). Ή-NR (CDC1 3): δ 1.46-1.60 (2Η, m), 1.60-1.84 (4H, m), 1.80 (3H, s), 2.30 (3H, s), 2.95 (2H, t, J = 7.5 Hz), 4.43 (2H, dt, J = 47.1, 6.0 Hz), 7.49 (IH, ddd, J = 7.5, 4.5, 0.6 Hz), 7.91 (IH, dt, J = 7.8, 1.5 Hz), 8.12 (1H , D, J = 7.8 Hz), 8.66 (IH, d, J-4.5 Hz), 10.26 (IH, bs).
元素分析値: C18H21N403S3Fとして Elementary analysis: as C 18 H 21 N 4 0 3 S 3 F
計算値 (¾) : C, 47.35; H, 4.64; N, 12.27; S, 21.07. Calculated value (¾): C, 47.35; H, 4.64; N, 12.27; S, 21.07.
実測値 (%) : C, 47.29; H, 4.61; N, 12.30; S, 21.06. Found (%): C, 47.29; H, 4.61; N, 12.30; S, 21.06.
実施例 7- 175 Example 7-175
N-{5, 6-ジメチル- 8- [ (5-フルォロペンチル)スルファニル] - 3-ォキソ _2, 3-ジヒド 口- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィ Vデン } (2 -二トロフエニル)メタンス ルホンアミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo_2,3-dihydrone-1H-imidazo [5, tric] [1,4] thiazine-trivden } (2-Ditrophenyl) methanesulfonamide
実施例 7-170と同様にして合成し、酢酸ェチル -ジェチルェ一テルから再結晶し て融点 143- 1451:の暗赤色結晶を得た。 The compound was synthesized in the same manner as in Example 7-170, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 143-1451 :.
IR (KBr) V: 3200, 2934, 1740, 1663, 1611, 1563, 1530 cm"1. IR (KBr) V: 3200, 2934, 1740, 1663, 1611, 1563, 1530 cm " 1 .
Ή-NMR (CDCI3) : δ 1.48-1.88 (6Η, m), 1.80 (3H, s), 2.25 (3H, s), 2.96 (2H, t, J=7.5Hz), 4.48 (2H, dt, J=47.4, 5.7 Hz), 4.96 (2H, s), 7.51 (IH, dt, J=8.7, 1.4 Hz), 7.63 (IH, dt, J=7.8, 0.9 Hz), 7.67 (IH, dd, J=7.8, 1.5 Hz), 8.04 (IH, d, J=7.8 Hz), 9.29 (1H, bs). Ή-NMR (CDCI3): δ 1.48-1.88 (6Η, m), 1.80 (3H, s), 2.25 (3H, s), 2.96 (2H, t, J = 7.5Hz), 4.48 (2H, dt, J = 47.4, 5.7 Hz), 4.96 (2H, s), 7.51 (IH, dt, J = 8.7, 1.4 Hz), 7.63 (IH, dt, J = 7.8, 0.9 Hz), 7.67 (IH, dd, J = 7.8, 1.5 Hz), 8.04 (IH, d, J = 7.8 Hz), 9.29 (1H, bs).
元素分析値: C2。H23N405S3Fとして Elemental analysis: C 2. H 23 N 4 0 5 S 3 F
計算値 (¾) : C, 46.68; H, 4.50; N, 10.89; S, 18.69. Calculated value (¾): C, 46.68; H, 4.50; N, 10.89; S, 18.69.
実測値 (¾) : C, 46.49; H, 4.56; N, 10.87; S, 18.80. Found (¾): C, 46.49; H, 4.56; N, 10.87; S, 18.80.
実施例 7-176 3 -ク口口- N- {5, 6-ジメチル -8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン } -卜プロパンスル ホンアミド Example 7-176 3-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene} -propanesulfonamide
水素化ナトリウム(25.4 mg, 0.634腿 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5,6-ジメチル 8- [5-フルォロ ぺンチル]スルファニル] - 3H-ィミダゾ [5, 1-c] [1 , 4]チアジン -3-ォン (100 mg, 0.317讓01) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 3-クロ口プロパンスルホニル クロリド (0.0472 ml, 0.380匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 20分間かき混ぜた。 反応 混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(113 mg, 79%)を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 140- 142°Cの暗赤色結晶を得 た。 To a suspension of sodium hydride (25.4 mg, 0.634 tmol) (60% oil) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl 8- [5 -Fluoropentyl] sulfanyl]-3H-imidazo [5, 1-c] [1, 4] thiazine-3-one (100 mg, 0.317 categoriz 01) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled on ice. Stir for 5 minutes. To this was added a solution of 3-chloropropanesulfonyl chloride (0.0472 ml, 0.380 marl) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 20 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (113 mg, 79%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 140-142 ° C.
IR (KBr) V: 3162, 2961, 2934, 1755, 1665, 1622, 1556 cm— IR (KBr) V: 3162, 2961, 2934, 1755, 1665, 1622, 1556 cm—
'H-NMR (CDC13) : δ 1.50-1.84 (6H, m), 1.82 (3H, s), 2.29 (3H, s), 3.11 (2H, t, J=7.6 Hz), 2.35-2.48 (2H, m), 3.01 (2H, t, J=7.5 Hz), 3.30 (2H, t, J=7.2 Hz), 3.72 (2H, t, J=6.3 Hz), 4.47 (2H, dt, J-47. , 5.7 Hz), 9.43 (1H, bs). 元素分析値: C16H23N303S3C1Fとして 'H-NMR (CDC1 3) : δ 1.50-1.84 (6H, m), 1.82 (3H, s), 2.29 (3H, s), 3.11 (2H, t, J = 7.6 Hz), 2.35-2.48 (2H , m), 3.01 (2H, t, J = 7.5 Hz), 3.30 (2H, t, J = 7.2 Hz), 3.72 (2H, t, J = 6.3 Hz), 4.47 (2H, dt, J-47. , 5.7 Hz), 9.43 (1H, bs). Elemental analysis: C 16 H 23 N 3 0 3 S 3 C1F
計算値 (%) : C, 42.14; H, 5.08; N, 9.21; S, 21.09; C1, 7.77. Calculated value (%): C, 42.14; H, 5.08; N, 9.21; S, 21.09; C1, 7.77.
実測値 ( : C, 42.24; H, 4.91; N, 9.33; S, 20.92; Cl, 8.05. Obtained value (: C, 42.24; H, 4.91; N, 9.33; S, 20.92; Cl, 8.05.
実施例 7-177 Example 7-177
N_{5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン _1 -ィリデン }- 4- (メチルスルホニル)ベン ゼンスルホンアミド N_ {5, 6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine_1-ylidene} -4- (methylsulfonyl) benzene sulphonamide
実施例 7- 176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 195- 197^の暗赤色結晶を得た。 The compound was synthesized in the same manner as in Examples 7-176, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 195-197 ^.
IR (KBr) リ : 3088, 3025, 2957, 2940, 2924, 1723, 1659, 1595, 1559 cm一1. Ή-NMR (CDCI3) : 51.48-1.52 (2H, m), 1.52-1.84 (4H, m), 1.81 (3H, s), 2.29 (3H, s), 2.99 (2H, t, J=7.2 Hz), 3.09 (3H, s), 4.47 (2H, dt, J-47.4, 5.7 Hz) , 8.07 (2H, d, J=8.7 Hz), 8.18 (2H, d, J=8.7 Hz), 9.63 (1H, bs). IR (KBr) Re:. 3088, 3025, 2957, 2940, 2924, 1723, 1659, 1595, 1559 cm one 1 Ή-NMR (CDCI 3) : 51.48-1.52 (2H, m), 1.52-1.84 (4H, m), 1.81 (3H, s), 2.29 (3H, s), 2.99 (2H, t, J = 7.2 Hz), 3.09 (3H, s), 4.47 (2H, dt, J-47.4, 5.7 Hz), 8.07 (2H, d, J = 8.7 Hz), 8.18 (2H, d, J = 8.7 Hz), 9.63 (1H, bs).
元素分析値: C20H24N305S4F · 0 · 5E t OAcとして Elementary analysis: as C 20 H 24 N 3 0 5 S 4 F · 0 · 5E t OAc
計算値 (%) : C, 45.74; H, 4.89; N, 7.27; S, 22.20. Calculated value (%): C, 45.74; H, 4.89; N, 7.27; S, 22.20.
実測値 (%) : C, 45.51; H, 4.89; N, 7.47; S, 21 92. Found (%): C, 45.51; H, 4.89; N, 7.47; S, 21 92.
実施例 7-178 Example 7-178
N- {5, 6-ジメチル -8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン卜 3-フエノキシベンゼンスル ホンアミド N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrone-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene 3-Phenoxybenzenesulfonamide
実施例 7 - 176と同様にして合成し、酢酸ェチル -ジェチルェ一テルから再結晶し て融点 178- 180°Cの暗赤色結晶を得た。 The compound was synthesized in the same manner as in Examples 7-176, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 178-180 ° C.
IR (KBr) V: 3250, 2921, 1732, 1663, 1620, 1584, 1559 cm"1. IR (KBr) V: 3250, 2921, 1732, 1663, 1620, 1584, 1559 cm " 1 .
'H-蘭 R (CDC13) : (51.46-1.62 (2H, jn), 1.62-1.84 (4H, πι), 1.80 (3Η, s), 2.27 (3H, d, J=0.9 Hz), 2.94 (2H, t, 1=7.8 Hz), 4.44 (2H, dt, J=47.1, 5.7 Hz), 7.03 (2H, dd, J=7.5, 1.2 Hz), 7.13-7.20 (2H, m), 7.37 (2H, t, J=7.5 Hz), 7.44 (1H, t, J=8.1 Hz), 7.61 (1H, t, J=1.8Hz), 7.68 (1H, dt, J=3.0, 1.2 Hz), 9.57 (1H, bs). 'H- Ran R (CDC1 3): (51.46-1.62 (2H, jn), 1.62-1.84 (4H, πι), 1.80 (3Η, s), 2.27 (3H, d, J = 0.9 Hz), 2.94 ( 2H, t, 1 = 7.8 Hz), 4.44 (2H, dt, J = 47.1, 5.7 Hz), 7.03 (2H, dd, J = 7.5, 1.2 Hz), 7.13-7.20 (2H, m), 7.37 (2H , t, J = 7.5 Hz), 7.44 (1H, t, J = 8.1 Hz), 7.61 (1H, t, J = 1.8 Hz), 7.68 (1H, dt, J = 3.0, 1.2 Hz), 9.57 (1H , bs).
元素分析値: C25H26N304S3Fとして Elementary analysis: as C 25 H 26 N 3 0 4 S 3 F
計算値 (%) : C, 54.82; H, 4.78; N, 7.67; S, 17.56. Calculated value (%): C, 54.82; H, 4.78; N, 7.67; S, 17.56.
実測値 (» : C, 54.55; H, 4.80; N, 7.59; S, 17.31. Actual value (»: C, 54.55; H, 4.80; N, 7.59; S, 17.31.
実施例 7-179 Example 7-179
(3,4-ジクロロフエニル) -N - {5, 6-ジメチル -8- [(5-フルォロペンチル)スルファニ ル] -3-ォキソ - 2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-トイリデン }メ タンスルホンアミド (3,4-dichlorophenyl) -N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-toylidene} methanesulfonamide
実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルェ一テルから再結晶し て融点 175- 177°Cの暗赤色結晶を得た。 The compound was synthesized in the same manner as in Examples 7-176, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 175-177 ° C.
IR (KBr) リ: 3189, 2940, 1736, 1665, 1618, 1561 cm- Ή-NMR (CDC13) : 51.46-1.68 (2H, m), 1.68-1.80 (4H, m), 1.81 (3H, s), 2.25 (3H, s), 3.01 (2H, t, J=7.2 Hz), 4.28 (2H, s), 4.47 (2H, dt, J=47.1, 5.7 Hz), 7.29 (1H, dd, J=8.1, 2.1 Hz), 7.43 (1H, d, J=8. l Hz), 7.54 (1H, d, J=1.8Hz), 9.23 (1H, bs). IR (KBr) Re: 3189, 2940, 1736, 1665 , 1618, 1561 cm- Ή-NMR (CDC1 3): 51.46-1.68 (2H, m), 1.68-1.80 (4H, m), 1.81 (3H, s ), 2.25 (3H, s), 3.01 (2H, t, J = 7.2 Hz), 4.28 (2H, s), 4.47 (2H, dt, J = 47.1, 5.7 Hz), 7.29 (1H, dd, J = 8.1, 2.1 Hz), 7.43 (1H, d, J = 8. L Hz), 7.54 (1H, d, J = 1.8Hz), 9.23 (1H, bs).
実施例 7-180 Example 7-180
N-{5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド ロ- IH-イミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデン } [4- (トリフルォロメチル)フ ェニル]メタンスルホンアミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-IH-imidazo [5,1-c] [1,4] thiazine-1- Ylidene} [4- (trifluoromethyl) phenyl] methanesulfonamide
実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 177-179 の暗赤色結晶を得た。 The compound was synthesized in the same manner as in Examples 7-176, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 177-179.
赤外吸収スペクトル(IR) (KBr中) リ: 3230, 2938, 1744, 1665, 1615, 1564 cm- Ή-NMR (CDC13) : δ 1.52-1.66 (2Η, m), 1.66-1.88 (4H, m), 1.80 (3H, d, J=1.2 Hz), 2.23 (3H, d, J=0.9 Hz), 2.99 (2H, t, J=6.9 Hz), 4.40 (2H, s), 4.48 (2H, dt, 1=47.1, 6.0 Hz), 7.56 (2H, d, J=9.0 Hz), 7.61 (2H, d, J=8.7 Hz), 9.23 (IH, bs). Infrared absorption spectrum (IR) (in KBr) Re: 3230, 2938, 1744, 1665 , 1615, 1564 cm- Ή-NMR (CDC1 3): δ 1.52-1.66 (2Η, m), 1.66-1.88 (4H, m), 1.80 (3H, d, J = 1.2 Hz), 2.23 (3H, d, J = 0.9 Hz), 2.99 (2H, t, J = 6.9 Hz), 4.40 (2H, s), 4.48 (2H, dt, 1 = 47.1, 6.0 Hz), 7.56 (2H, d, J = 9.0 Hz), 7.61 (2H, d, J = 8.7 Hz), 9.23 (IH, bs).
元素分析値: C2lH23N303S3F4として Elemental analysis: as C 2l H 23 N 3 0 3 S 3 F 4
計算値 (%) : C, 46.92; H, 4.31; N, 7.82; S, 17.89. Calculated value (%): C, 46.92; H, 4.31; N, 7.82; S, 17.89.
実測値 (%) : C, 46.53; H, 4.28; N, 7.92; S, 18.31. Found (%): C, 46.53; H, 4.28; N, 7.92; S, 18.31.
実施例 7-181 Example 7-181
[3, 5 -ビス (トリフルォロメチル)フエニル] - N-{5, 6-ジメチル- 8-[(5-フルォロぺ ンチル)スルファニル ]_3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジ 実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 160- 162での暗赤色結晶を得た。 [3,5-bis (trifluoromethyl) phenyl] -N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] _3-oxo-2,3-dihydro-1H-imidazo [ 5,1-c] [1,4] thiazi Synthesized in the same manner as in Example 7-176, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 160-162.
赤外吸収スペクトル(IR) (KBr中) V: 3281, 2934, 1730, 1667, 1617, 1561 cm- 'H-NMR (CDC13) : δ 1.48-1.65 (2Η, m), 1.65-1.85 (4H, m), 1.80 (3H, s), 2.23 (3H, s), 2.99 (2H, t, J=7.5 Hz), 4.45 (2H, s), 2.98 (2H, t, J-7.5 Hz), 4.47 (2H, di, J=47.1, 6.0 Hz), 7.86 (1H, s), 7.92 (2H, s), 9.21 (ΪΗ, bs). 実施例 7-182 Infrared absorption spectrum (IR) (in KBr) V: 3281, 2934, 1730, 1667, 1617, 1561 cm- 'H-NMR (CDC1 3): δ 1.48-1.65 (2Η, m), 1.65-1.85 (4H , m), 1.80 (3H, s), 2.23 (3H, s), 2.99 (2H, t, J = 7.5 Hz), 4.45 (2H, s), 2.98 (2H, t, J-7.5 Hz), 4.47 (2H, di, J = 47.1, 6.0 Hz), 7.86 (1H, s), 7.92 (2H, s), 9.21 (ΪΗ, bs).
N- {5, 6-ジメチル -8- [ (5-フルォ口ぺンチル)スルファニル] -3 -ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン } [3- (トリフルォロメチル)フ ェニル]メタンスルホンアミド 実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 180-182°Cの暗赤色結晶を得た。 N- {5,6-Dimethyl-8-[(5-fluoromethylpentyl) sulfanyl] -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine -Trilidene} [3- (trifluoromethyl) phenyl] methanesulfonamide The compound was synthesized in the same manner as in Example 7-176, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 180-182 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3220, 2942, 1742, 1667, 1615, 1564 cm"1. Ή-NMR (CDC13) : δ 1.40-1.84 (6Η, m), 1.80 (3H, s), 2.21 (3H, s), 3.01 (2H, t, J=7.2 Hz), 4.40 (2H, s), 4.48 (2H, dt, J=47.2, 5.8 Hz), 7.45-7.72 (4H, m), 9.17 (1H, bs). Infrared absorption spectrum (IR) (in KBr) V:. 3220, 2942 , 1742, 1667, 1615, 1564 cm "1 Ή-NMR (CDC1 3): δ 1.40-1.84 (6Η, m), 1.80 (3H, s), 2.21 (3H, s), 3.01 (2H, t, J = 7.2 Hz), 4.40 (2H, s), 4.48 (2H, dt, J = 47.2, 5.8 Hz), 7.45-7.72 (4H, m ), 9.17 (1H, bs).
元素分析値: C21H23N303S3F4として Elemental analysis: as C 21 H 23 N 3 0 3 S 3 F 4
計算値 (%) : C, 46.92; H, 4.31; , 7.82; S, 17.89. Calculated (%): C, 46.92; H, 4.31;, 7.82; S, 17.89.
実測値 (%) : C, 46.70; H, 4.26; N, 7.63; S, 17.84. Found (%): C, 46.70; H, 4.26; N, 7.63; S, 17.84.
実施例 7-183 Example 7-183
N- {5, 6-ジメチル- 8- [ (5 -フルォ口ぺンチル)スルファニル] -3-ォキソ -2, 3 -ジヒド 口- m-イミダゾ [5, l-c] [1,4]チアジン - 1 -ィリデン } [1,1 ' -ビフエ二ル]- 3-スル ホンアミド N- {5,6-Dimethyl-8-[(5-fluoromethylpentyl) sulfanyl] -3-oxo-2,3-dihydric-m-imidazo [5, lc] [1,4] thiazine-1 -Ylidene} [1,1'-biphenyl]-3-sulfonamide
実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 152- 154T:の暗赤色結晶を得た。 The compound was synthesized in the same manner as in Examples 7-176, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 152-154T :.
IR (KBr) V 3202, 3071, 2963, 2942, 1752, 1663, 1620, 1559 cm"1. IR (KBr) V 3202, 3071, 2963, 2942, 1752, 1663, 1620, 1559 cm " 1 .
Ή -腹 (CDC13) : (51.40-1.84 (6H, m), 1.79 (3H, s), 2.27 (3H, s), 2.95 (2H, t, J-7.4 Hz), 4.41 (2H, dt, J=46.8, 6.0 Hz), 7.38-7.54 (3H, m), 7.54-7.66E - belly (CDC1 3): (51.40-1.84 ( 6H, m), 1.79 (3H, s), 2.27 (3H, s), 2.95 (2H, t, J-7.4 Hz), 4.41 (2H, dt, J = 46.8, 6.0 Hz), 7.38-7.54 (3H, m), 7.54-7.66
(3H, in), 7.78 (1H, d, J=7.0 Hz), 7.94 (1H, d, J=8.0 Hz), 8.22 (1H, s), 9.63 (1H, bs). (3H, in), 7.78 (1H, d, J = 7.0 Hz), 7.94 (1H, d, J = 8.0 Hz), 8.22 (1H, s), 9.63 (1H, bs).
元素分析値: C25H26N3O3S3F'0.3H2Oとして Elementary analysis: as C 25 H 26 N 3 O 3 S 3 F'0.3H 2 O
計算値 (¾) : C, 55.91; H, 4.99; N, 7.82; S, 17.91. Calculated (¾): C, 55.91; H, 4.99; N, 7.82; S, 17.91.
実測値 (%) : C, 55.88; H, 4.77; N, 7.70; S, 18.13. Found (%): C, 55.88; H, 4.77; N, 7.70; S, 18.13.
実施例 7-184 Example 7-184
N-{5, 6-ジメチル -8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口 -1H-ィミダゾ [5, l-c] [1, 4]チアジン- 1-ィリデン }- 2, 2-ジフエ二ルエタンスル ホンアミド N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydric mouth-1H-imidazo [5, lc] [1,4] thiazine-1-ylidene} -2,2-diphenylethanesulfonamide
実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 181- 183 の暗赤色結晶を得た。 IR (KBr) v: 3220, 2934, 1740, 1665, 1622, 1566 cm一1. The compound was synthesized in the same manner as in Example 7-176, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 181-183. IR (KBr) v: 3220, 2934, 1740, 1665, 1622, 1566 cm one 1.
Ή-NMR (CDC13) : 01.46-1.88 (6H, m), 1.81 (3H, d, J=1.2 Hz), 2.24 (3H, d, J=1.0Hz), 2.97 (2H, t, J=7.2Hz), 3.97 (2H, d, J=7.4Hz), 4.48 (2H, dt, J=47.2, 5.8 Hz), 4.64 (IH, t, J=8.0 Hz), 7.14-7.36 (10H, m), 9.25 (IH, bs). Ή-NMR (CDC1 3): 01.46-1.88 (6H, m), 1.81 (3H, d, J = 1.2 Hz), 2.24 (3H, d, J = 1.0Hz), 2.97 (2H, t, J = 7.2 Hz), 3.97 (2H, d, J = 7.4Hz), 4.48 (2H, dt, J = 47.2, 5.8 Hz), 4.64 (IH, t, J = 8.0 Hz), 7.14-7.36 (10H, m), 9.25 (IH, bs).
元素分析値: C27 。N303S3Fとして Elemental analysis: C 27. N 3 0 3 S 3 F
計算値 (%) : C, 57.94; H, 5.40; N, 7.51; S, 17.19. Calculated value (%): C, 57.94; H, 5.40; N, 7.51; S, 17.19.
実測値 (%) : C, 57.59; Η, 5.46; Ν, 7.38; S, 17.10. Found (%): C, 57.59; Η, 5.46; Ν, 7.38; S, 17.10.
実施例 7-185 Example 7-185
Ν- (8- {[2- (卜ァダマンチル)ェチル]スルファ二ル} -5, 6-ジメチル -3-ォキソ -2, 3- ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン-卜ィリデン)-4-ブロモベンゼンス ルホンアミド Ν- (8-{[2- (Tudamantyl) ethyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tri c] [1,4] Thiazine-triylidene) -4-bromobenzenesulfonamide
実施例 6-40で得た化合物 (126 mg, 0.323 腿 ol) の THF (3.0 ml) 懸濁液に水 素化ナトリウム (純度 60%油性, 32.3 mg, 0.809 匪 ol) を室温で加え、 15分間 攪拌後、 塩ィ匕 4 -ブロモベンゼンスルホニル(90.9 mg, 0.356匪 ol)を加え、 さら に 2時間攪拌した。 反応液にメタノール(1 ml) および IN塩酸 (0.7 ml)を加え、 メタノール水(2:1, 5 ml)を加えて希釈した。 析出物を濾取し、 メタノール水、 メ 夕ノールおよびジェチルェ一テルで洗浄後、 乾燥して表題化合物(95 mg, 0.156 腿 ol, 48.3%)を赤色粉末として得た。 To a suspension of the compound obtained in Example 6-40 (126 mg, 0.323 t) in THF (3.0 ml) was added sodium hydride (60% oil, 32.3 mg, 0.809 ol) at room temperature. After stirring for 1 minute, Shiojiri 4-bromobenzenesulfonyl (90.9 mg, 0.356 ol) was added, and the mixture was further stirred for 2 hours. Methanol (1 ml) and IN hydrochloric acid (0.7 ml) were added to the reaction solution, and methanol water (2: 1, 5 ml) was added for dilution. The precipitate was collected by filtration, washed with methanol water, methanol and diethyl ether, and dried to give the title compound (95 mg, 0.156 mol, 48.3%) as a red powder.
Ή-NMR (200MHz, CDC13): 51.43 (2H, m), 1.50 (6H, d, J= 2.6 Hz), 1.65 (3H, brd, J= 12 Hz), 1.71 (3H, brd, J= 12 Hz), 1.80 (3H, q, J= 1.2 Hz), 1.98 (3H, brs), 2.28 (3H, q, J= 1.0 Hz), 2.92 (2H, m), 7.63 (2H, dt, J= 8.4, 2.2 Hz), 7.83 (2H, dt, J= 8.8, 2.2 Hz), 9.60 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): 51.43 (2H, m), 1.50 (6H, d, J = 2.6 Hz), 1.65 (3H, brd, J = 12 Hz), 1.71 (3H, brd, J = 12 Hz), 1.80 (3H, q, J = 1.2 Hz), 1.98 (3H, brs), 2.28 (3H, q, J = 1.0 Hz), 2.92 (2H, m), 7.63 (2H, dt, J = 8.4 , 2.2 Hz), 7.83 (2H, dt, J = 8.8, 2.2 Hz), 9.60 (1H, brs).
元素分析値: C26H30N303S3Brとして計算値: C, 51.31; H, 4.97; N, 6.90; S, 15.81; Br, 13.13 (%) 、 実測値: C, 51.38; H, 5.13; N, 6.64; S, 15.72; Br, 13.23 ( ) . Elemental analysis: C 26 H 30 N 3 0 3 S 3 Br Calculated: C, 51.31; H, 4.97 ; N, 6.90; S, 15.81; Br, 13.13 (%), Found: C, 51.38; H , 5.13; N, 6.64; S, 15.72; Br, 13.23 ().
即. 199.5-201.0°C Immediately. 199.5-201.0 ° C
実施例 7-186 Example 7-186
N - {8- [(1-ァダマンチルメチル) スルファニル ]-5, 6 -ジメチル- 3 -ォキソ -2, 3-ジ ヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン }- 4-ブロモベンゼンスル ホンアミド N- {8-[(1-adamantylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine- 1-ylidene}-4-bromobenzenesul Honamide
実施例 6- 41で得た化合物 (98 mg, 0.261腿 ol) の THF (3.0 ml) 懸濁液に水素 化ナトリウム(純度 60% 油性, 26.1 mg, 0.652匪 ol) を室温で加え、 15分間攪拌 後、 塩化 4-ブロモベンゼンスルホニル(73.3 rag, 0.287腿 ol)を加え、 さらに 1 時間攪拌した。 反応液にメタノール (4 ml) および IN塩酸 OL0 ml)を加えた。 析 出物を濾取し、メタノール—水(1:1)、メタノールおよびジェチルエーテルで洗浄 後、 乾燥して表題化合物(124 mg, 0.208匪 ol, 79.9%)を赤色粉末として得た。 Ή-NMR (200MHz, CDC13): 51.55 (6H, d, J= 2.2 Hz), 1.61 (3H, brd, J= 12 Hz), 1.71 (3H, brd, J= 12 Hz), 1.80 (3H, q, J= 1.2 Hz), 2.01 (3H, brs), 2.27 (3H, q, J- 1.2 Hz), 2.77 (2H, s), 7.60 (2H, dt, J= 8.8, 2.2 Hz), 7.87 (2H, dt, J= 8.8, 2.2 Hz), 9.54 (1H, brs). To a suspension of the compound obtained in Example 6-41 (98 mg, 0.261 tmol) in THF (3.0 ml) was added sodium hydride (purity 60% oil, 26.1 mg, 0.652 bandol) at room temperature, and the mixture was added for 15 minutes. After stirring, 4-bromobenzenesulfonyl chloride (73.3 rag, 0.287 mol) was added, and the mixture was further stirred for 1 hour. Methanol (4 ml) and IN hydrochloric acid (OL 0 ml) were added to the reaction solution. The precipitate was collected by filtration, washed with methanol-water (1: 1), methanol and getyl ether, and dried to give the title compound (124 mg, 0.208 bandol, 79.9%) as a red powder. Ή-NMR (200MHz, CDC1 3 ): 51.55 (6H, d, J = 2.2 Hz), 1.61 (3H, brd, J = 12 Hz), 1.71 (3H, brd, J = 12 Hz), 1.80 (3H, q, J = 1.2 Hz), 2.01 (3H, brs), 2.27 (3H, q, J- 1.2 Hz), 2.77 (2H, s), 7.60 (2H, dt, J = 8.8, 2.2 Hz), 7.87 ( 2H, dt, J = 8.8, 2.2 Hz), 9.54 (1H, brs).
元素分析値: C25 8N303S3Brとして計算値: C, 50.50; H, 4.75; N, 7.07; S, 16.18; Br, 13.44 (%) 、 実測値: C, 50.40; H, 4.56; N, 6.79; S, 15.91; Br, 13.56 (%) . Elemental analysis: C 25 8 N 3 0 3 S 3 Br Calculated: C, 50.50; H, 4.75 ; N, 7.07; S, 16.18; Br, 13.44 (%), Found: C, 50.40; H, 4.56; N, 6.79; S, 15.91; Br, 13.56 (%).
mp. 227.0-228. Ot: 1 実施例 7-187 mp. 227.0-228.Ot: 1 Example 7-187
4 -ブロモ Ν - {8- [(シクロォクチルメチル) スルファニル ]-5, 6-ジメチル- 3-ォキ ソ- 2, 3 -ジヒドロ- 1Η-イミダゾ [5,卜 c] [1,4]チアジン - 1 -ィリデン }ベンゼンスル ホンアミド 4-bromoΝ- {8-[(cyclooctylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1Η-imidazo [5, tric] [1,4] Thiazine-1-ylidene} benzenesulfonamide
実施例 6-42で得た化合物 (135 mg, 0.384讓 ol) の THF (3.0 ml) 懸濁液に水 素化ナトリウム (純度 60%油性, 38.4 mg, 0.960 mmol) を室温で加え、 15分間 攪拌後、 塩化 4-ブロモベンゼンスルホニル(108 mg, 0.422匪 ol)を加え、 さらに 1時間攪拌した。 反応液にメタノール(1 ml) および IN塩酸(1.0 ml)を加え、 メ タノール水 (2:1, 5 ml)を加えて希釈した。析出物を濾取し、メタノ—ル—水 (1:1)、 メタノールおよびジェチルェ一テルで洗浄後、乾燥して表題化合物(217mg, 0.380 誦 ol, 99.0 )を赤橙色粉末として得た。 To a suspension of the compound (135 mg, 0.384 alcohol) obtained in Example 6-42 in THF (3.0 ml) was added sodium hydride (purity 60% oily, 38.4 mg, 0.960 mmol) at room temperature, and the mixture was added for 15 minutes. After stirring, 4-bromobenzenesulfonyl chloride (108 mg, 0.422 ol) was added, and the mixture was further stirred for 1 hour. Methanol (1 ml) and IN hydrochloric acid (1.0 ml) were added to the reaction solution, and aqueous methanol (2: 1, 5 ml) was added for dilution. The precipitate was collected by filtration, washed with methanol-water (1: 1), methanol and diethyl ether, and dried to give the title compound (217 mg, 0.380, ol, 99.0) as a red-orange powder.
Ή-NMR (200MHz, CDC13): δ 1.30-1.90 (15H, m), 1.79 (3H, s), 2.27 (3H, s), 2.83 (2H, d, J= 6.6 Hz), 7.61 (2H, d, J= 8.8 Hz), 7.84 (2H, d, J- 8.4 Hz), 9.57 (1H, brs). 元素分析値: C23H28N303S3Brとして計算値: C, 48.41; H, 4.95; N, 7.36; S, 16.86; Br, 14.00 (%) 、 実測値: C, 48.27; H, 4.98; N, 7.12; S, 16.76; Br, 14.14 Ή-NMR (200MHz, CDC1 3 ): δ 1.30-1.90 (15H, m), 1.79 (3H, s), 2.27 (3H, s), 2.83 (2H, d, J = 6.6 Hz), 7.61 (2H, d, J = 8.8 Hz), 7.84 (2H, d, J-8.4 Hz), 9.57 (1H, brs). Elemental analysis: C 23 H 28 N 3 0 3 S 3 Br Calculated: C, 48.41; H, 4.95 ; N, 7.36; S, 16.86; Br, 14.00 (%), Found: C, 48.27; H , 4.98; N, 7.12; S, 16.76; Br, 14.14
(%) . (%).
mp. 199.0-200. O mp. 199.0-200. O
実施例 7-188 Example 7-188
N-{5, 6-ジメチル- 8- [ (5-フルォ口ぺンチル)スルファニル] -3 -ォキソ- 2, 3-ジヒド ロ- 1H-イミダゾ [5,l-c] [1,4]チアジン- 1-ィリデン }シクロプロパンスルホンアミ ド N- {5,6-Dimethyl-8-[(5-fluoroopentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-1 -Ylidene} cyclopropanesulfonamide
実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 160- 162°Cの暗赤色結晶を得た。 It was synthesized in the same manner as in Examples 7-176, and recrystallized from ethyl acetate-getyl ether to give dark red crystals having a melting point of 160-162 ° C.
IR (KBr) V: 3206, 2926, 1748, 1732, 1665, 1630, 1570 cm"1. IR (KBr) V: 3206, 2926, 1748, 1732, 1665, 1630, 1570 cm " 1 .
Ή-NMR (CDC13) : (50.960-1.08 (2H, m), 1.20-1.32 (2H, m), 1.46-1.86 (6H, m), Ή-NMR (CDC1 3): (50.960-1.08 (2H, m), 1.20-1.32 (2H, m), 1.46-1.86 (6H, m),
1.81 (3H, s), 2.28 (3H, d, J=0.8 Hz), 2.54-2.71 (1H, m), 2.99 (2H, t, J=7.21.81 (3H, s), 2.28 (3H, d, J = 0.8 Hz), 2.54-2.71 (1H, m), 2.99 (2H, t, J = 7.2
Hz), 4.46 (2H, dt, J=47.4, 5.8 Hz), 9.43 (1H, bs) . Hz), 4.46 (2H, dt, J = 47.4, 5.8 Hz), 9.43 (1H, bs).
元素分析値: C16H22N303S3F'0.2H20として Elemental analysis: C 16 H 22 N 3 0 3 S 3 F'0.2H 20
計算値 (%) : C, 45.41; H, 5.34; N, 9.93; S, 22.73. Calculated (%): C, 45.41; H, 5.34; N, 9.93; S, 22.73.
実測値 (%) : C, 45.42; H, 5.34; N, 9.80; S, 22.96. Found (%): C, 45.42; H, 5.34; N, 9.80; S, 22.96.
実施例 7-189 Example 7-189
8, 9-ジメチル- 1卜 [(5-フルォロペンチル)スルファニル ]-3, 4-ジヒドロ [1,4]チア ジノ [4, ,3' :3, 4]イミダゾ [5,1 - c] [1, 2, 4]チアジアジン _6 -オン 2,2-ジォキシ ド、 8,9-dimethyl-1 [[5-fluoropentyl) sulfanyl] -3,4-dihydro [1,4] thiazino [4,, 3 ': 3,4] imidazo [5,1-c] [1 , 2,4] thiadiazine _6-one 2,2-dioxide,
水素化ナトリウム(25.4 mg, 0.634匪 ol) (60 油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 23で得られた 1 -ァミノ- 5,6-ジメチル 8- [5-フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (100 mg, 0.317腿01) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 2-クロ口エタンスルホニル クロリドのテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 22時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸 ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去て題記 化合物 (52.0mg, 41¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テル から再結晶して融点 uo-urcの暗赤色結晶を得た。 To a suspension of sodium hydride (25.4 mg, 0.634 ol) (60 oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl 8- [5- Add a solution of fluoropentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.317 thigh 01) in tetrahydrofuran (1.00 ml), and stir under ice-cooling for 5 minutes. I did. To this was added a solution of ethanesulfonyl chloride 2-tetrachloride in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 22 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract washed with water to give after drying (MgSO 4), the solvent the title compound Te evaporated under reduced pressure (52.0mg, 41¾) as a dark red crystals. Ethyl acetate-Jechlether To give dark red crystals with a melting point of uo-urc.
IR (KBr) V: 2940, 2861, 1738, 1663, 1603, 1564 cm1 IR (KBr) V: 2940, 2861, 1738, 1663, 1603, 1564 cm 1
-腿 R (CDC13) : 51.50-1.64 (2H, ) , 1.64-1.86 (4H, m), 1.83 (3H, d, J=0.6 Hz), 2.29 (3H, d, J=1.2 Hz), 3.00 (2H, t, J=7.8 Hz), 3.26 (2H, t, J=6.3 Hz), 4.21 (2H, i, J=6.3 Hz), 4.45 (2H, di, J=47.1, 5.7 Hz). - thigh R (CDC1 3): 51.50-1.64 ( 2H,), 1.64-1.86 (4H, m), 1.83 (3H, d, J = 0.6 Hz), 2.29 (3H, d, J = 1.2 Hz), 3.00 (2H, t, J = 7.8 Hz), 3.26 (2H, t, J = 6.3 Hz), 4.21 (2H, i, J = 6.3 Hz), 4.45 (2H, di, J = 47.1, 5.7 Hz).
元素分析値: C15 0N3O3S3F -0.3¾0として Elementary analysis: as C 15 0 N 3 O 3 S 3 F -0.3¾0
計算値 ) : C, 43.84; H, 5.05; N, 10.23. Calculated values): C, 43.84; H, 5.05; N, 10.23.
実測値 (¾ : C, 43.86; H, 4.75; N, 10.18. Measured value (¾: C, 43.86; H, 4.75; N, 10.18.
実施例 7-190 Example 7-190
4 -ブロモ - -{1, 6-ジォキソ- 4-[(5-フルォロペンチル)スルフ ァニ ル]- 1, 2, 6, 7, 8, 9 -へキサヒドロ- 3H -ィミダゾ [5, 1-c] [1, 4]ベンゾチアジン - 4 -ィ · 水素化ナトリウム(37.7 mg, 0.942 mmol) (60W由性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6- 47で得られた 3-ァミノ- 4-[(5-フルォロペンチル)ス ルファニル] - 8, 9_ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン - 1, 6 (7H) -ジオン (167 mg, 0.471 mmol) のテトラヒドロフラン (2.00 ml) 溶液を加え、 氷冷下 5 分間かき混ぜた。 これに 4-ブロモベンゼンスルホニル クロリド (246 mg, 0.942 mmol)のテトラヒドロフラン(1.00 ral)溶液を加え、さらに 2.5時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgSO 後、溶媒を減圧下に留去して題記化合物(175 mg, 65 )を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 245- 247°Cの暗赤色結晶を得 た。 4-bromo--{1,6-dioxo-4-[(5-fluoropentyl) sulfanyl] -1,2,6,7,8,9-hexahydro-3H-imidazo [5,1-c ] [1,4] Benzothiazine-4-di • To a suspension of sodium hydride (37.7 mg, 0.942 mmol) (60W free) in tetrahydrofuran (1.00 ml) was added the suspension obtained in Example 6-47. Amino-4-[(5-fluoropentyl) sulfanyl]-8,9_dihydric mouth-1H-imidazo [5,1-c] [1,4] thiazine-1,6 (7H) -dione (167 mg, 0.471 mmol) in tetrahydrofuran (2.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. To this was added a solution of 4-bromobenzenesulfonyl chloride (246 mg, 0.942 mmol) in tetrahydrofuran (1.00 ral), and the mixture was further stirred for 2.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water and dried (after MgSO 4), and the solvent was distilled off under reduced pressure to obtain the title compound (175 mg, 65) as dark red crystals, which were recrystallized from ethyl acetate-ethyl ether to give a melting point of 245-247. ° C dark red crystals were obtained.
IR (KBr) V : 3169, 2967, 2940, 1736, 1649; 1605, 1582, 1541 cm—1. IR (KBr) V: 3169, 2967, 2940, 1736, 1649; 1605, 1582, 1541 cm— 1 .
Ή一画 R (CDC13) : ά 1.50-1.90 (6Η, m), 1.98-2.14 (2H, m) , 2.52 (2Η, ί, 】=7.0 Hz), 3.07 (2Η, t, J=7.4 Hz), 3.22 (2H, t, J=6.2 Hz), 4.46 (2H, dt, J=47. , 5.8 Hz), 7.64 (2H, d, J-8.8 Hz), 7.82 (2H, d, J=8. Hz), 9.83 (1H, bs). 元素分析値: C2i N304S3BrFとして Ή Ichikaku R (CDC1 3): ά 1.50-1.90 (6Η, m), 1.98-2.14 (2H, m), 2.52 (2Η, ί, ] = 7.0 Hz), 3.07 (2Η , t, J = 7.4 Hz ), 3.22 (2H, t, J = 6.2 Hz), 4.46 (2H, dt, J = 47., 5.8 Hz), 7.64 (2H, d, J-8.8 Hz), 7.82 (2H, d, J = 8 . Hz), 9.83 (1H, bs) Elementary analysis:. a C 2i N 3 0 4 S 3 BrF
計算値 (%) : C, 43.90; H, 3.68; N, 7.31; S, 16.74. Calculated value (%): C, 43.90; H, 3.68; N, 7.31; S, 16.74.
実測値 (%) : C, 44.06; Η, 3.59; Ν, 7.24; S, 16.59. 実施例 7-191 Found (%): C, 44.06; Η, 3.59; Ν, 7.24; S, 16.59. Example 7-191
4 -シァノ -N- {5, 6-ジメチル -8_[(5-フルォロペンチル)スルファニル ]-3-ォキソ -2,3-ジヒドロ -1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン }ベンゼンスルホ ンアミド 4-Cyano-N- {5,6-dimethyl-8 _ [(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene} benzenesulfonamide
実施例 7-176と同様にして合成し、酢酸ェチル -ジェチルエーテルから再結晶し て融点 171- 173°Cの暗赤色結晶を得た。 The compound was synthesized in the same manner as in Example 7-176, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 171-173 ° C.
IR (KBr) V 3225, 2934, 2232, 1757, 1663, 1620, 1557 cm— IR (KBr) V 3225, 2934, 2232, 1757, 1663, 1620, 1557 cm—
lH-NMR (CDC13) : δ 1.48-1.62 (4Η, m), 1.64-1.84 (2H, m), 1.80 (3H, d, J=1.2 Hz), 2.28 (3H, d, J=0.9 Hz), 2.98 (2H, t, J=7.2 Hz), 4.46 (2H, dt, J=47. , 5.7 Hz), 7.78 (2H, d, J=8.4 Hz), 8.07 (2H, d, J=8. Hz), 9.58 (1H, bs) . 元素分析値: C2。H21N403S3Fとして lH-NMR (CDC1 3): δ 1.48-1.62 (4Η, m), 1.64-1.84 (2H, m), 1.80 (3H, d, J = 1.2 Hz), 2.28 (3H, d, J = 0.9 Hz) , 2.98 (2H, t, J = 7.2 Hz), 4.46 (2H, dt, J = 47., 5.7 Hz), 7.78 (2H, d, J = 8.4 Hz), 8.07 (2H, d, J = 8. Hz), 9.58 (1H, bs ) elemental analysis:. C 2. H 21 N 4 0 3 As S 3 F
計算値 (¾) : C, 49.98; H, 4.40; N, 11.66; S, 20.02. Calculated (¾): C, 49.98; H, 4.40; N, 11.66; S, 20.02.
実測値 (¾) : C, 49.81; H, 4.39; N, 11.54; S, 19.99. Found (¾): C, 49.81; H, 4.39; N, 11.54; S, 19.99.
実施例 7-192 Example 7-192
N- {5, 6 -ジメチル -8- [ (5-フルォ口ぺンチル)スルファニル] -3-ォキソ -2, 3-ジヒド ロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン } (3-メチルフエニル)メタンス ルホンアミド N- {5,6-Dimethyl-8-[(5-fluoroopentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -Trilidene} (3-methylphenyl) methanesulfonamide
実施例 7-176と同様にして合成し、酢酸ェチル-ジェチルェ一テルから再結晶し て融点 151- 153°Cの暗赤色結晶を得た。 It was synthesized in the same manner as in Examples 7-176, and recrystallized from ethyl acetate-jetil ether to obtain dark red crystals having a melting point of 151-153 ° C.
IR (KBr) v: 3204, 2934, 1742, 1663, 1622, 1564 cm"1. IR (KBr) v: 3204, 2934, 1742, 1663, 1622, 1564 cm " 1 .
Ή-NMR (CDC13) : δ 1.50-1.66 (2Η, m), 1.66-1.86 (4H, m), 1.80 (3H, s), 2.20 Ή-NMR (CDC1 3): δ 1.50-1.66 (2Η, m), 1.66-1.86 (4H, m), 1.80 (3H, s), 2.20
(3H, s), 2.35 (3H, s), 3.01 (2H, t, J=7.5 Hz), 4.31 (2H, s), 4.47 (2H, dt,(3H, s), 2.35 (3H, s), 3.01 (2H, t, J = 7.5 Hz), 4.31 (2H, s), 4.47 (2H, dt,
J=47.1, 5.7 Hz), 7.11-7.17 (1H, m), 7.18?7.24 (3H, m), 9.08 (1H, bs). 元素分析値: C21H24N303S3Fとして ? J = 47.1, 5.7 Hz) , 7.11-7.17 (1H, m), 7.18 7.24 (3H, m), 9.08 (1H, bs) Elementary analysis:. As C 21 H 24 N 3 0 3 S 3 F
計算値 (%) : C, 52.37; H, 5.02; N, 8.72; S, 19.97. Calculated value (%): C, 52.37; H, 5.02; N, 8.72; S, 19.97.
実測値 (%) : C, 52.04; H, 5.36; N, 8.75; S, 19.74. Found (%): C, 52.04; H, 5.36; N, 8.75; S, 19.74.
実施例 7-193 Example 7-193
4 -ブロモ [5, 6-ジメチル- 3-ォキソ -8- ({[(2S)- 5-ォキソピロリジニル]メチル } スルファエル) -2, 3-ジヒドロー 1H -ィミダゾ [5, 1-c] [1, ]チアジン—トイリデン]ベ ンゼンスルホンアミド 4-bromo [5,6-dimethyl-3-oxo-8-({[(2S) -5-oxopyrrolidinyl] methyl} sulfael) -2,3-dihydro-1H-imidazo [5,1-c ] [1,] thiazine-toylidene] Nzensulfonamide
実施例 6-44で得た化合物 (117 mg, 0.361 腿 ol) (D THF-DMF (3:1, 4 ml)懸濁 液に水素化ナトリウム (純度 60% 油性, 43.3 mg, 1.08讓 ol) を室温で加え、 15 分間攪拌後、 塩化 4 -ブロモベンゼンスルホニル(111 mg, 0.433 腿 ol)を加え、 さ らに 1.5時間攪拌した。 反応液にメタノール(1 ml) および IN塩酸(1.1 ml)を加 えて酢酸ェチルで抽出し、 脱水乾燥、 濃縮して赤色油状物を得た。 これに酢酸ェ チルおよびジェチルエーテルを加え、 析出物を濾取し、 酢酸ェチルージェチルェ 一テル(1:4)およびジェチルエーテルで洗浄後、 乾燥して表題化合物(134 mg, 0.247 匪 ol, 68.3%)を濃紫色粉末として得た。 Sodium hydride (purity 60% oil, 43.3 mg, 1.08 mL) was added to a suspension of the compound obtained in Example 6-44 (117 mg, 0.361 tmol) (D THF-DMF (3: 1, 4 ml)) Was added at room temperature, and the mixture was stirred for 15 minutes, 4-bromobenzenesulfonyl chloride (111 mg, 0.433 mL) was added, and the mixture was further stirred for 1.5 hours.The reaction solution was methanol (1 ml) and IN hydrochloric acid (1.1 ml). Then, the mixture was extracted with ethyl acetate, dehydrated and dried, and concentrated to obtain a red oily substance.Ethyl acetate and getyl ether were added thereto, and the precipitate was collected by filtration. 1: 4) and washed with getyl ether, and dried to give the title compound (134 mg, 0.247 ol, 68.3%) as a dark purple powder.
Ή一 NMR (300MHz, CDC13): 51.80 (3H, s), 1.84 (1H, dd, J= 14.1, 4.5 Hz), 2.27 (3H, s), 2.35 (1H, m), 2.36 (2H, brs), 2.99 (1H, dd, J= 13.5, 7.5 Hz), 3.13 (1H, dd, J= 13.8, 5.1 Hz), 3.90 (1H, brquint, J= 5.6 Hz), 5.86 (1H, brs), 7.65 (2H, d, J= 8.4 Hz), 7.81 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή one NMR (300MHz, CDC1 3): 51.80 (3H, s), 1.84 (1H, dd, J = 14.1, 4.5 Hz), 2.27 (3H, s), 2.35 (1H, m), 2.36 (2H, brs ), 2.99 (1H, dd, J = 13.5, 7.5 Hz), 3.13 (1H, dd, J = 13.8, 5.1 Hz), 3.90 (1H, brquint, J = 5.6 Hz), 5.86 (1H, brs), 7.65 (2H, d, J = 8.4 Hz), 7.81 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C19H19N404S3Brとして計算値: C, 41.99; H, 3.52; N, 10.31; S, 17.70; Br, 14.70 (%) 、 実測値: C, 42.02; H, 3.60; N, 10.28; S, 17.46; Br, 14.95Elemental analysis: C 19 H 19 N 4 0 4 S 3 Br Calculated: C, 41.99; H, 3.52 ; N, 10.31; S, 17.70; Br, 14.70 (%), Found: C, 42.02; H , 3.60; N, 10.28; S, 17.46; Br, 14.95
(%) · (%)
rap. 182.0-185. Ot: rap. 182.0-185. Ot:
実施例 7-194 Example 7-194
3 - {[(1- {[(4 -ブロモフエニル)スルホニル]イミノ}- 5,6-ジメチル- 3-ォキソ -2,3 - ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 8 -ィル)スルファニル]メチル }-1 - ピぺリジンカルボン酸 tert-ブチル 3-{[(1-{[(4-bromophenyl) sulfonyl] imino}-5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-8-yl) sulfanyl] methyl} -1-butylpiperidinecarboxylate
実施例 6-45で得た化合物(100 ig, 0.235 匪 ol) の THF (3 ml) 懸濁液に水 素化ナトリウム(純度 60% 油性, 23.5 mg, 0.589 mmol) を室温で加え、 30分間攪 拌後、 塩化 4-ブロモベンゼンスルホニル(72.2 mg, 0.283 mmol)を加え、 さらに 24時間攙拌した p 反応液に 1N塩酸(1.0 ml)を加えて酢酸ェチルで抽出し、 脱水 乾燥、 濃縮してメタノールで希釈した。 析出物を濾取し、 メタノールで洗浄後、 乾燥して表題化合物(66 mg, 0.103 腿 ol, 43.6 )を灰紫色粉末として得た。 To a suspension of the compound obtained in Example 6-45 (100 ig, 0.235 bandol) in THF (3 ml) was added sodium hydride (purity 60% oily, 23.5 mg, 0.589 mmol) at room temperature, and the mixture was added for 30 minutes.攪拌後, 4-bromobenzenesulfonyl chloride (72.2 mg, 0.283 mmol) was extracted with added an additional 24 hours攙拌the p reaction solution are added a 1N hydrochloric acid (1.0 ml) acetic Echiru, dehydrated and dried, and concentrated And diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (66 mg, 0.103 t, 43.6) as a gray-violet powder.
'H-NMR (300MHz, CDC13): 51.46 (9H, s), 1.40-1.52 (3H, m), 1.65 (1H, dt, J= 8.4, 4.8 Hz), 1.79 (3H, q, J= 0.9 Hz), 1.91 (1H, brd, J= 9.0 Hz), 2.27 (3H, q, J= 1.2 Hz), 2.60-2.80 (1H, br), 2.81 (1H, dd, J= 13.2, 7.2 Hz), 2.90 (1H, dd, J= 12.9, 6.6 Hz), 2.90 (1H, br), 3.77 (1H, m), 3.80-4.14 (1H, br), 7.64 (2H, d, J= 8.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.55 (1H, brs). 'H-NMR (300MHz, CDC1 3): 51.46 (9H, s), 1.40-1.52 (3H, m), 1.65 (1H, dt, J = 8.4, 4.8 Hz), 1.79 (3H, q, J = 0.9 Hz), 1.91 (1H, brd, J = 9.0 Hz), 2.27 (3H, q, J = 1.2 Hz), 2.60-2.80 (1H, br), 2.81 (1H, dd, J = 13.2, 7.2 Hz), 2.90 (1H, dd, J = 12.9, 6.6 Hz), 2.90 (1H, br ), 3.77 (1H, m), 3.80-4.14 (1H, br), 7.64 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.55 (1H, brs).
元素分析値: C25H31N405S3Brとして計算値: C, 46.65; H, 4.84; N, 8.70; S, 14.95; Br, 12.41 (%) 、 実測値: C, 47.29; H, 4.90; N, 8.57; S, 14.72; Br, 12.78 (%) . Elemental analysis: C 25 H 31 N 4 0 5 S 3 Br Calculated: C, 46.65; H, 4.84 ; N, 8.70; S, 14.95; Br, 12.41 (%), Found: C, 47.29; H , 4.90; N, 8.57; S, 14.72; Br, 12.78 (%).
mp. 158.5-160.0°C mp.158.5-160.0 ° C
実施例 7-195 ' Example 7-195 '
(2S,4R)- 2 {[(1 - {[(4 -ブロモフエニル)スルホニル]イミノ} - 5, 6 -ジメチル -3 -ォ キソ -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, ]チアジン- 8-ィル)スルファニル]メ チリレ} - 4-ヒドロキシ-卜ピロリジンカルボン酸 tert-ブチル (2S, 4R)-2 {[(1-{[(4-bromophenyl) sulfonyl] imino}-5, 6-dimethyl-3-oxo-2,3-dihydrido-1H-imidazo [5,1- c] [1,] thiazin-8-yl) sulfanyl] methylyl} -tert-butyl 4-hydroxy-topyrrolidinecarboxylate
実施例 6-46で得た粗製の化合物 (120 mg, 0.197 mmol) の THF (3 ml) 懸濁液 に水素化ナトリウム (純度 60¾油性, 23.6 mg, 0.591 蘭 ol) を室温で加え、 10 分間攪拌後、 塩ィヒ 4 -ブロモベンゼンスルホニル(65.4 mg, 0.256 mmol)を加え、 さらに 2時間攪拌した。 反応液にメタノール(1 ml) および IN塩酸(1.0 ml)を加 えて酢酸ェチルで抽出し、 脱水乾燥、 濃縮して赤色油状物を得た。 これをシリカ. ゲル(7.5 g)のクロマトグラフィー(酢酸ェチルーへキサン、 3:7-7:3)で精製し、 酢酸ェチルおよびジェチルエーテルから結晶化して表題化合物(30 mg, 0.046 mmol, 23.6 )を紫色粉末として得た。 To a suspension of the crude compound (120 mg, 0.197 mmol) obtained in Example 6-46 in THF (3 ml) was added sodium hydride (purity: 60¾, 23.6 mg, 0.591 orchid ol) at room temperature for 10 minutes. After stirring, 4-chlorobenzenesulfonyl chloride (65.4 mg, 0.256 mmol) was added, and the mixture was further stirred for 2 hours. Methanol (1 ml) and IN hydrochloric acid (1.0 ml) were added to the reaction solution, extracted with ethyl acetate, dried, dried and concentrated to obtain a red oil. This was purified by chromatography on silica gel (7.5 g) (ethyl acetate-hexane, 3: 7-7: 3) and crystallized from ethyl acetate and getyl ether to give the title compound (30 mg, 0.046 mmol, 23.6 mg). ) Was obtained as a purple powder.
!H-NMR (300MHz, CDC13): δ 1.48 (9H, s), 1.74 (1Η, d, J= 3.6 Hz), 1.79 (3H, s), 1.94 (1H, ddd, J= 13.5, 7.5, 4.5 Hz), 2.11 (1H, ra), 2.26 (3H, q, J= 0.6 Hz), 3.14 (1H, br), 3.35 (1H, dd, J= 12.3, 4.2 Hz), 3.42 (1H, dd, J= 12.3, 4.2 Hz), 3.64 (1H, br), 4.25 (1H, m), 4.41 (1H, brs), 7.63 (2H, d, J= 8.1 Hz), 7.81 (2H, d, J= 8.4 Hz), 9.60 (1H, brs).! H-NMR (300MHz, CDC1 3): δ 1.48 (9H, s), 1.74 (1Η, d, J = 3.6 Hz), 1.79 (3H, s), 1.94 (1H, ddd, J = 13.5, 7.5, 4.5 Hz), 2.11 (1H, ra), 2.26 (3H, q, J = 0.6 Hz), 3.14 (1H, br), 3.35 (1H, dd, J = 12.3, 4.2 Hz), 3.42 (1H, dd, J = 12.3, 4.2 Hz), 3.64 (1H, br), 4.25 (1H, m), 4.41 (1H, brs), 7.63 (2H, d, J = 8.1 Hz), 7.81 (2H, d, J = 8.4 Hz), 9.60 (1H, brs).
p. 173.0-174.5°C p. 173.0-174.5 ° C
実施例 7- 196 Example 7-196
N - {5, 6-ジメチル- 8- C (5-フルォ口ぺンチル)スルファニル] -3-ォキソ -2, 3 -ジヒド ロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィ シクロへキシルメルカプタン (167 mg, 1.36 mmol) のァセトニトリル (13.6ml) 溶液に、硝酸カリウム(348 mg, 3.41 mmol)続いて塩ィ匕スルフリル (0.279 ml, 3.41 醒 ol) を加え、 氷冷下 2時間かき混ぜた。 反応混合物に飽和炭酸水素ナトリウム 水溶液を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を 減圧下に留去して、 シクロへキサンスルホニルクロリドを油状物として得た。 水 素化ナトリウム (25.4mg, 0.634腿 ol) (60%油性) のテトラヒドロフラン (2.00 ml) の懸濁液に、 実施例 6 - 23で得られた卜アミノ- 5, 6-ジメチル -8- [(5-フルォ 口ペンチル)スルファニル] - 3H -イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (100 mg, 0.317 mmol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに上記で得られたシクロへキサンスルホニルクロリドのテトラビドロ フラン溶液 (1.00 ml) を加え、 さらに室温で 1 時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (44.7 mg, 31%) を暗赤色結晶として得た。 酢酸 ェチル-ジェチルエーテルから再結晶して融点 138- 140°Cの暗赤色結晶を得た。 赤外吸収スぺクトル(IR) (KBr中) リ: 3158, 2936, 2857, 1755, 1665, 1626, 1572, 1375, 1094 cnr1. N- {5,6-Dimethyl-8-C (5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1- To a solution of cyclohexylmercaptan (167 mg, 1.36 mmol) in acetonitrile (13.6 ml) was added potassium nitrate (348 mg, 3.41 mmol), followed by sodium chloride sulfuryl (0.279 ml, 3.41 wakeup), and the mixture was cooled on ice for 2 hours. Stirred. A saturated aqueous sodium hydrogen carbonate solution was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure to give a hexane sulfonyl chloride cyclohexane as an oil. To a suspension of sodium hydride (25.4 mg, 0.634 tmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8- [obtained in Example 6-23. (5-Fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.317 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled under ice-cooling. Stir for a minute. To this was added a solution of cyclohexanesulfonyl chloride obtained above in tetravidrofuran (1.00 ml), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (44.7 mg, 31%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 138-140 ° C. Infrared absorption spectrum (IR) (in KBr): 3158, 2936, 2857, 1755, 1665, 1626, 1572, 1375, 1094 cnr 1 .
Ή-NMR (CDC13) : δ 1.14-1.38 (4Η, m), 1.48-1.65 (4H, m), 1.65-1.84 (4H, m), 1.81 (3H, d, J=1.2 Hz), 1.86-1.95 (2H, m), 2.18-2.30 (2H, m), 2.28 (3H, d, J=1.2 Hz), 2.92 (1H, dt, J=8.0, 3.3 Hz), 3.00 (2H, i, J=7.5 Hz), 4.46 (2H, dt, J=47.4, 6.0 Hz), 9.50 (1H, bs). Ή-NMR (CDC1 3): δ 1.14-1.38 (4Η, m), 1.48-1.65 (4H, m), 1.65-1.84 (4H, m), 1.81 (3H, d, J = 1.2 Hz), 1.86- 1.95 (2H, m), 2.18-2.30 (2H, m), 2.28 (3H, d, J = 1.2 Hz), 2.92 (1H, dt, J = 8.0, 3.3 Hz), 3.00 (2H, i, J = 7.5 Hz), 4.46 (2H, dt, J = 47.4, 6.0 Hz), 9.50 (1H, bs).
元素分析値: C19H2SN303S3Fとして Elementary analysis: as C 19 H 2S N 3 0 3 S 3 F
計算値 (%) : C, 49.43; H, 6.11; N, 9.10; S, 20.84. Calculated (%): C, 49.43; H, 6.11; N, 9.10; S, 20.84.
実測値 (¾) : C, 49.27; H, 6.16; N, 9.28; S, 21.10. Found (¾): C, 49.27; H, 6.16; N, 9.28; S, 21.10.
実施例 7-197 Example 7-197
(2S) -2-{[(1-{[ (4 -ブロモフエニル)スルホニル]イミノト 5, 6-ジメチル -3-ォキソ - 2, 3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1, 4]チアジン- 8 -ィル)スルファニル]メチ ル} -卜ピロリジンカルボン酸 tert -プチル (2S) -2-{[(1-{[(4-bromophenyl) sulfonyl] iminoto 5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4 ] Thiazine-8-yl) sulfanyl] methyl} -topyrrolidinecarboxylic acid tert-butyl
実施例 6-48で得た粗製の化合物(純度 70%, 140 mg, 0.24 mmol) の THF (3 ml) 懸濁液に水素化ナトリウム(純度 60%油性, 33.4 mg, 0.835 mmol) を室温で加え、 10分間攪拌後、 塩化 4 -ブロモベンゼンスルホニル(91 mg, 0.358 mmol)を加え、 さらに 2時間攪拌した。 反応液に 1N塩酸(1.0 ml)を加えて酢酸ェチルで抽出し、 脱水乾燥、 濃縮して赤色油状物を得た。 これをシリカゲル (7.5 g)のクロマトダラ フィー (酢酸ェチルーへキサン、 2:8-4:6)で精製し、 ジェチルエーテルから結晶 化して表題化合物(92 mg, 0.146 mmol, 60.9%)を赤紫色粉末として得た。 To a suspension of the crude compound obtained in Examples 6-48 (purity 70%, 140 mg, 0.24 mmol) in THF (3 ml) was added sodium hydride (purity 60%, oily, 33.4 mg, 0.835 mmol) at room temperature. In addition, After stirring for 10 minutes, 4-bromobenzenesulfonyl chloride (91 mg, 0.358 mmol) was added, and the mixture was further stirred for 2 hours. 1N Hydrochloric acid (1.0 ml) was added to the reaction solution, extracted with ethyl acetate, dehydrated and dried, and concentrated to obtain a red oil. This was purified by chromatography on silica gel (7.5 g) (ethyl acetate-hexane, 2: 8-4: 6) and crystallized from getyl ether to give the title compound (92 mg, 0.146 mmol, 60.9%) as a purple-red color. Obtained as a powder.
Ή-NMR (300MHz, CDC13): <5 1.47 (9H, s), 1.79 (3H, s), 1.84 (2H, HI), 1.95 (2H, m), 2.27 (3H, s), 2.90-3.15 (1H, br), 3.34 (2H, m), 3.40 (1H, m), 4.04 (1H, m), 7.63 (2¾ d, J= 8.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.59 (1H, brs). 元素分析値: C24H29N405S3Brとして計算値: C, 45.78; H, 4.64; N, 8.90; S, 15.28; Br, 12.69 (%) 、 実測値: C, 45.87; H, 4.69; N, 8.84; S, 15.01; Br, 12.83 (%) . Ή-NMR (300MHz, CDC1 3 ): <5 1.47 (9H, s), 1.79 (3H, s), 1.84 (2H, HI), 1.95 (2H, m), 2.27 (3H, s), 2.90-3.15 (1H, br), 3.34 (2H, m), 3.40 (1H, m), 4.04 (1H, m), 7.63 (2¾ d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), . 9.59 (1H, brs) elemental analysis: C 24 H 29 N 4 0 5 S 3 Br calculated: C, 45.78; H, 4.64 ; N, 8.90; S, 15.28; Br, 12.69 (%), measured Values: C, 45.87; H, 4.69; N, 8.84; S, 15.01; Br, 12.83 (%).
融点: 182.5-184. O Melting point: 182.5-184. O
実施例 7-198 Example 7-198
4-ブロモ -N- { 8- [ (5 , 5-ジフルォロシクロォクチルメチル) スルファニル ] _5, 6 -ジ メチル -3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデン } ベンゼンスルホンアミド 4-bromo-N- {8- [(5,5-difluorocyclooctylmethyl) sulfanyl] _5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1- c] [1, 4] thiazine-triylidene} benzenesulfonamide
実施例 6-49で得た化合物(147 mg, 0.394腿 ol) の THF (3 ml) 懸濁液に水素 化ナトリウム (純度 60%油性, 39.4 mg, 0.984 mmol) を室温で加え、 15分間攪 拌後、塩ィ匕 4-ブロモベンゼンスルホニル(111 mg, 0.433 mmol)を加え、 さらに 3.5 時間攪拌した。 反応液に 1N塩酸(1.0 ml)を加えて酢酸ェチル(30 ml)で抽出し、 脱水乾燥、 濃縮して赤色油状物を得た。 これを酢酸ェチルに溶解し、 析出物を濾 取してジェチルエーテルで洗浄後、 乾燥して表題化合物 (209 mg, 0.353 mmol, Sodium hydride (purity 60% oily, 39.4 mg, 0.984 mmol) was added to a suspension of the compound obtained in Example 6-49 (147 mg, 0.394 tmol) in THF (3 ml) at room temperature, and stirred for 15 minutes. After stirring, 4-bromobenzenesulfonyl (111 mg, 0.433 mmol) was added, and the mixture was further stirred for 3.5 hours. The reaction mixture was added with 1N hydrochloric acid (1.0 ml), extracted with ethyl acetate (30 ml), dried, dried and concentrated to obtain a red oil. This was dissolved in ethyl acetate, and the precipitate was collected by filtration, washed with getyl ether, dried, and dried to give the title compound (209 mg, 0.353 mmol,
89.5¾)を紫色粉末として得た。 89.5%) as a purple powder.
Ή-NMR (200MHz, CDC13): 51.76 (6H, m), 1.82 (3H, s), 2.09 (6H, m), 2.28 (3H, s), 3.54 (1H, m), 7.63 (2H, d, J= 8.8 Hz), 7.83 (2H, d, J= 8.8 Hz), 9.63 (1H, brs) . Ή-NMR (200MHz, CDC1 3 ): 51.76 (6H, m), 1.82 (3H, s), 2.09 (6H, m), 2.28 (3H, s), 3.54 (1H, m), 7.63 (2H, d , J = 8.8 Hz), 7.83 (2H, d, J = 8.8 Hz), 9.63 (1H, brs).
元素分析値: C22H24N303S3BrF2として計算値: C, 44.59; H, 4.08; N, 7.09; S, 16.23; Br, 13.48; F, 6.41 (%) 、 実測値: C, 44.68; H, 4.12; N, 7.06; S, 16.03; Br, 13.24; F, 6.24 (%) . . 融点: 197.0-199.5°C Elemental analysis: C 22 H 24 N 3 0 3 S 3 BrF 2 Calculated: C, 44.59; H, 4.08 ; N, 7.09; S, 16.23; Br, 13.48; F, 6.41 (%), Found: C, 44.68; H, 4.12; N, 7.06; S, 16.03; Br, 13.24; F, 6.24 (%). Melting point: 197.0-199.5 ° C
実施例 7-199 ル) - 5, 6 -ジメチル -3-ォキソ - 2, 3 -ジヒド口 -1H -ィミダゾ [5, 1-c] [1, 4]チアジン 実施例 6- 50で得た粗製の化合物 (純度 70%, 297 mg, 0.58 mmol) の THF (6 ml) 懸濁液に水素化ナトリウム (純度 60%油性, 81.2mg, 2.03匪01) を室温で加え、 15分間攪拌後、 塩化 4-ブロモベンゼンスルホニル(193 mg, 0.754 mmol)を加え、 さらに 2時間攪拌した。 反応液にメタノール( 1 m 1 )および 1 N塩酸(2.0 m 1 )を加え て 1時間攪拌した。 析出物を濾取してメタノール—水 (3:1 および 1:1)およびメ 夕ノールで洗浄後、 乾燥して表題化合物(3:1 異性体混合物, 209 mg, 0.364讓 ol, 62.7%)を赤橙色粉末として得た。 Example 7-199 l) -5,6-Dimethyl-3-oxo-2,3-dihydrogen-1H-imidazo [5,1-c] [1,4] thiazine Crude obtained in Example 6-50 Sodium hydride (purity 60% oily, 81.2 mg, 2.03 band01) was added to a suspension of the above compound (purity 70%, 297 mg, 0.58 mmol) in THF (6 ml) at room temperature. After stirring for 15 minutes, chloride was added. 4-Bromobenzenesulfonyl (193 mg, 0.754 mmol) was added, and the mixture was further stirred for 2 hours. Methanol (1 m 1) and 1 N hydrochloric acid (2.0 m 1) were added to the reaction solution, and the mixture was stirred for 1 hour. The precipitate is collected by filtration, washed with methanol-water (3: 1 and 1: 1) and methanol, dried, and dried to give the title compound (3: 1 isomer mixture, 209 mg, 0.364 cellulose, 62.7%) Was obtained as a red-orange powder.
¾-證 (300MHz, CDC13, major/minor): δ 0.94/0.75 (2Η, m/q, J= 12.3Hz), 1.32 (2H, m), 1.48-1.70 (3H, in), 1.80 (3H, q, J= 0.9 Hz), 1.76-1.97 (4H, m), 2.27 (3H, q, J= 1.2 Hz), 2.85/2.95 (2H, d/dd, J= 6.6/7.2, 1.8 Hz), 4.22 (1H, ddd, J= 47.4, 8.7, 6.0 Hz, major), 4.26 (1H, ddd, J= 47.4, 8.7, 5.1 Hz, major), 4.30 (2H, ddd, J= 47.4, 6.3, 0.9 Hz, minor), 7.61 (2H, dt, J= 9.0, 2.2 Hz), 7.83 (2H, dt, J= 8.7, 1.6 Hz), 9.61 (1H, brs). ¾- testimony (300MHz, CDC1 3, major / minor): δ 0.94 / 0.75 (2Η, m / q, J = 12.3Hz), 1.32 (2H, m), 1.48-1.70 (3H, in), 1.80 (3H , q, J = 0.9 Hz), 1.76-1.97 (4H, m), 2.27 (3H, q, J = 1.2 Hz), 2.85 / 2.95 (2H, d / dd, J = 6.6 / 7.2, 1.8 Hz), 4.22 (1H, ddd, J = 47.4, 8.7, 6.0 Hz, major), 4.26 (1H, ddd, J = 47.4, 8.7, 5.1 Hz, major), 4.30 (2H, ddd, J = 47.4, 6.3, 0.9 Hz) , minor), 7.61 (2H, dt, J = 9.0, 2.2 Hz), 7.83 (2H, dt, J = 8.7, 1.6 Hz), 9.61 (1H, brs).
元素分析値: C22H25N303S3BrFとして計算値: C, 45.99; H, 4.39; N, 7.31; S, 16.74; Br, 13.91; F, 3.31 ( ) 、 実測値: C„ 45.96; H, 4.32; N, 7.20; S, 16.61; Br, 13.68; F, 3.46 (%) . Elemental analysis: C 22 H 25 N 3 0 3 S 3 Calculated BrF: C, 45.99; H, 4.39; N, 7.31; S, 16.74; Br, 13.91; F, 3.31 (), Found: C "45.96; H, 4.32; N, 7.20; S, 16.61; Br, 13.68; F, 3.46 (%).
融点: 195.0- 197. Melting point: 195.0- 197.
実施例 7-200 Example 7-200
4-ブロモ -N-(5, 6-ジメチル- 8- {[(卜メチルシクロへキシル)メチル]スルファニ ル}-3_ォキソ_2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン)ベ ンゼンスルホンアミド 4-bromo-N- (5,6-dimethyl-8-{[(trimethylcyclohexyl) methyl] sulfanyl} -3_oxo_2,3-dihydro-1H-imidazo [5,1-c] [ [1, 4] thiazine-1-ylidene) benzenesulfonamide
実施例 6- 51で得た化合物 (120 mg, 0.355匪 ol) の THF (3 ml) 懸濁液に水素 化ナトリウム (純度 60%油性, 35.6 mg, 0.889匪 ol) を室温で加え、 15分間攪 挣後、 塩化 4-ブロモベンゼンスルホニル(100 mg, 0.391 腿 ol)を加え、 さらに 1 時間攪拌した。反応液にメタノール(lml)および IN塩酸(1.0 ml)を加え、水(2 ml) で希釈した。析出物を濾取してメタノ一ルー水(1:1)、メタノールおよびジェチル エーテルで洗浄後、 乾燥して表題化合物 α 80 mg, 0.324 mmol, 91.1%)を紫色粉末 として得た。 To a suspension of the compound obtained in Example 6-51 (120 mg, 0.355 bandol) in THF (3 ml) was added sodium hydride (purity 60% oil, 35.6 mg, 0.889 bandol) at room temperature, and the mixture was added for 15 minutes. After stirring, 4-bromobenzenesulfonyl chloride (100 mg, 0.391 tmol) was added, and one more portion was added. Stirred for hours. Methanol (lml) and IN hydrochloric acid (1.0 ml) were added to the reaction solution, and the mixture was diluted with water (2 ml). The precipitate was collected by filtration, washed with methanol (1: 1), methanol and getyl ether, and dried to give the title compound α (80 mg, 0.324 mmol, 91.1%) as a purple powder.
'H-NMR (300MHz, CDC13): 1.00 (3H, s), 1.38 (5H, br), 1.47 (5H, br), 1.80 (3H, q, J= 0.6 Hz), 2.27 (3H, brs), 2.94 (2H, s), 7.60 (2H, d, J= 8.2 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.58 (1H, brs). 'H-NMR (300MHz, CDC1 3): 1.00 (3H, s), 1.38 (5H, br), 1.47 (5H, br), 1.80 (3H, q, J = 0.6 Hz), 2.27 (3H, brs) , 2.94 (2H, s), 7.60 (2H, d, J = 8.2 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.58 (1H, brs).
元素分析値: C22H26N303S3Brとして計算値: C, 47.48; H, 4.71; N, 7.55; S, 17.28; Br, 14.36 (%) 、 実測値: C, 47.35; H, 4.62; N, 7.46; S, 17.00; Br, 14.63 (%) . Elemental analysis: C 22 H 26 N 3 0 3 S 3 Br Calculated: C, 47.48; H, 4.71 ; N, 7.55; S, 17.28; Br, 14.36 (%), Found: C, 47.35; H , 4.62; N, 7.46; S, 17.00; Br, 14.63 (%).
実施例 7 - 201 Example 7-201
N - (3_{[1- ({[4_ (ァセチルァミノ)フエニル]スルホ二ル}ィミノ) - 5, 6 -ジメチル - 3-ォキソ -2, 3 -ジヒドロ- m-イミダゾ [5, 1-c] [1, 4]チアジン - 8-ィル]スルファニ 実施例 6-39で得た化合物(153 mg, 0.400 mmol)の THF (5 ml) 懸濁液に水素 化ナトリウム(純度 60%油性, 48.0 rag, 1.20匪 ol) を室温で加え、 10分間攪拌 後、 塩ィ匕 4-ァセタミドベンゼンスルホニル (116 mg, 0.480腿 ol)を加え、 さら に 2時間攪拌した。反応液にメタノール (1ml) 、 IN塩酸 (1.5ml) および水 (20 ml)を加え、 酢酸ェチル(30 ml)で抽出し 。 有機層を 0.1N塩酸、 水および飽和 食塩水で洗浄し、 硫酸ナトリウムで乾燥後、 濃縮した。 析出物を濾取し、 酢酸ェ チルおよびジェチルエーテルで洗浄後、乾燥して表題化合物 (161 mg, 0.278腿 ol, 69.4%)を灰赤色粉末として得た。 N- (3 _ {[1-({[4_ (acetylamino) phenyl] sulfonyl} imino) -5,6-dimethyl-3-oxo-2,3-dihydro-m-imidazo [5,1-c] [1,4] Thiazine-8-yl] sulfani Sodium hydride (purity 60% oil, 48.0 rag) was added to a suspension of the compound obtained in Example 6-39 (153 mg, 0.400 mmol) in THF (5 ml). , 1.20 bandol) at room temperature and stirred for 10 minutes, and then Shio-Dai 4-acetamidobenzenesulfonyl (116 mg, 0.480 liter) was added, and the mixture was further stirred for 2 hours. Then, IN hydrochloric acid (1.5 ml) and water (20 ml) were added, and the mixture was extracted with ethyl acetate (30 ml) .The organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The precipitate was collected by filtration, washed with ethyl acetate and getyl ether, and dried to give the title compound (161 mg, 0.278 mol, 69.4%) as a gray-red powder.
'H-NMR (200MHz, CDCl3-DMSO-d6= 9:1): (50.89 (3H, t, J= 6.6 Hz), 1.30 (4H, m), 1.62 (2H, brquint, J= 7.5 Hz), 1.79 (3H, s), 1.86 (2H, quint, J= 6.3 Hz), 2.16 (2H, t, J= 7.7 Hz), 2.17 (3H, s), 2.26 (3H, s), 2.97 (2H, t, J= 7.5 Hz), 3.27 (2H, q, J= 6.2 Hz), 6.84 (1H, brt, J= 6 Hz), 7.74 (2H, d, J= 8.8 Hz), 7.87 (2H, d, J= 9.0 Hz), 9.68 (1H, brs), 10.23 (1H, br). 'H-NMR (200MHz, CDCl 3 -DMSO-d 6 = 9: 1): (50.89 (3H, t, J = 6.6 Hz), 1.30 (4H, m), 1.62 (2H, brquint, J = 7.5 Hz) ), 1.79 (3H, s), 1.86 (2H, quint, J = 6.3 Hz), 2.16 (2H, t, J = 7.7 Hz), 2.17 (3H, s), 2.26 (3H, s), 2.97 (2H , t, J = 7.5 Hz), 3.27 (2H, q, J = 6.2 Hz), 6.84 (1H, brt, J = 6 Hz), 7.74 (2H, d, J = 8.8 Hz), 7.87 (2H, d , J = 9.0 Hz), 9.68 (1H, brs), 10.23 (1H, br).
元素分析値: C25H33N505S3 O.5¾0として計算値: C, 51.00; H, 5.82; N, 11.90; S, 16.33 (%) 、 実測値: C, 50.82; H, 5.90; N, 11.80; S, 16.54 ( ) . mp. 187-189°C Elemental analysis: C 25 H 33 N 5 0 5 S 3 O.5¾0 Calculated: C, 51.00; H, 5.82; N, 11.90; S, 16.33 (%), found: C, 50.82; H, 5.90 ; N, 11.80; S, 16.54 (). mp.187-189 ° C
実施例 7-202 Example 7-202
4-クロロ- N- [8- (シクロへプチルスルファニル) -5, 6-ジメチル- 3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン] ベンゼンスルホ ンアミド 4-chloro-N- [8- (cycloheptylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1 -Ylidene] benzenesulfonamide
実施例 6- 15で得た化合物 (207 mg, 0.640丽 ol) の THF (4 ml) 懸濁液に水素 化ナトリウム(純度 60%油性, 56.3 mg, 1.41 匪 ol) を室温で加え、 10分間攪拌 後、 塩化 4 -クロ口ベンゼンスルホニル(149 mg, 0.704腿 ol)を加え、 さらに 40 分間攙拌した。 反応液に 1N塩酸 (0.77 ml)を加え、 酢酸ェチルを加えて抽出し、 0.1 塩酸および飽和食塩水で洗浄し、乾燥、濃縮した。残渣にジェチルエーテル を加え、析出物を濾取し、 ジェチルエーテルで洗浄後、乾燥して標題化合物 (234 mg, 0.470 mmol)を紫色粉末として得た。 To a suspension of the compound obtained in Example 6-15 (207 mg, 0.640 mol) in THF (4 ml) was added sodium hydride (purity 60% oily, 56.3 mg, 1.41 mol) at room temperature, and the mixture was added for 10 minutes. After stirring, 4-benzene benzenesulfonyl chloride (149 mg, 0.704 mol) was added, and the mixture was further stirred for 40 minutes. 1N Hydrochloric acid (0.77 ml) was added to the reaction solution, and ethyl acetate was added for extraction, washed with 0.1 hydrochloric acid and saturated saline, dried and concentrated. Getyl ether was added to the residue, the precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (234 mg, 0.470 mmol) as a purple powder.
Ή-NMR (200MHz,, CDC13) : (51.50-1.80 (10H, m), 1.80 (3H, q, J= 1.2 Hz), 2.05 (2H, DI), 2.27 (3H, q, J= 1.2 Hz), 3.52 (1H, dt, J= 4.0, 9.2 Hz), 7.46 (2H, dt, J= 8.8, 2.2 Hz), 7.92 (2H, dt, J= 8.8, 2.2 Hz), 9.60 (1H, brs). Ή-NMR (200MHz ,, CDC1 3 ): (51.50-1.80 (10H, m), 1.80 (3H, q, J = 1.2 Hz), 2.05 (2H, DI), 2.27 (3H, q, J = 1.2 Hz ), 3.52 (1H, dt, J = 4.0, 9.2 Hz), 7.46 (2H, dt, J = 8.8, 2.2 Hz), 7.92 (2H, dt, J = 8.8, 2.2 Hz), 9.60 (1H, brs) .
元素分析: C21H24N303S3C1として計算値: C, 50.64; H, 4.86; , 8.44; S, 19.31; CI, 7.12(%), 実測値: C, 50.52; H, 4.76; N, 8.40; S, 19.44; CI, 7.05 (¾) . Elemental analysis: C 21 H 24 N 3 0 3 S 3 C1 Calculated: C, 50.64; H, 4.86 ;, 8.44; S, 19.31; CI, 7.12 (%), Found: C, 50.52; H, 4.76 ; N, 8.40; S, 19.44; CI, 7.05 (¾).
融点: 178.0-179.0°C Melting point: 178.0-179.0 ° C
実施例 7-203 Example 7-203
4-クロ口 - N- {5, 6-ジメチル- 8- [ (4-メチルペンチル)スルファニル] -3-ォキソ - 2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, ]チアジン- 1-ィリデン }ベンゼンスルホ ンアミド 4-Chloro-N- {5,6-dimethyl-8-[(4-methylpentyl) sulfanyl] -3-oxo-2,3-dihydric-1H-imidazo [5,1-c] [1, ] Thiazine-1-ylidene} benzenesulfonamide
実施例 6-17で得た化合物(203 mg, 0.628 mraol) の THF (4.0 ml) 懸濁液に水 素化ナトリウム(純度 60% 油性, 62.8 mg, 1.57讓 ol) を室温で加え、 30分間攪 拌後、塩化 4 -クロ口ベンゼンスルホニル(146 mg, 0.690腿 ol)を加え、 さらに 3.5 時間攪拌した。反応液を酢酸ェチルで希釈し、 0.1N塩酸および飽和食塩水で洗浄 し、 乾燥、 濃縮した。 残渣にメタノールを加え、 析出物を濾取し、 メタノールで 洗浄後、 乾燥して標題化合物 (268 mg, 0.539匪 ol)を赤色粉末として得た。 ^- MR (300MHz, CDC13) : δ 0.96 (2Η, brq, J= 11.9 Hz), 1.22 (3H, m), 1.54 (1H, quint, J- 3.7 Hz), 1.67-1.87 (5H, m), 1.80 (3H, q, J= 0.9 Hz), 2.27 (3H, q, J= 0.9 Hz), 2.83 (2H, d, J= 6.9 Hz), 7.45 (2H, dt, J= 9.0, 2.4 Hz), 7.92 (2H, dt, J= 8.7, 2.1 Hz), 9.60 (1H, brs). To a suspension of the compound obtained in Example 6-17 (203 mg, 0.628 mraol) in THF (4.0 ml) was added sodium hydride (purity 60% oil, 62.8 mg, 1.57 acetyl) at room temperature, and the mixture was added for 30 minutes. After stirring, 4-chlorobenzenebenzenesulfonyl chloride (146 mg, 0.690 liter) was added, and the mixture was further stirred for 3.5 hours. The reaction solution was diluted with ethyl acetate, washed with 0.1N hydrochloric acid and saturated saline, dried and concentrated. Methanol was added to the residue, and the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (268 mg, 0.539 bandol) as a red powder. ^ - MR (300MHz, CDC1 3 ): δ 0.96 (2Η, brq, J = 11.9 Hz), 1.22 (3H, m), 1.54 (1H, quint, J- 3.7 Hz), 1.67-1.87 (5H, m), 1.80 (3H, q, J = 0.9 Hz), 2.27 (3H, q, J = 0.9 Hz), 2.83 (2H, d, J = 6.9) Hz), 7.45 (2H, dt, J = 9.0, 2.4 Hz), 7.92 (2H, dt, J = 8.7, 2.1 Hz), 9.60 (1H, brs).
元素分析値: C21H24N303S3C1として計算値: C, 50.64; H, 4.86; N, 8.44; S, 19.31; CI, 7.12(%)、 実測値: C, 50.60; H, 4.75; N, 8.62; S, 19.18; CI, 7.08(¾). 融点: 198.5-199.5°C Elemental analysis: C 21 H 24 N 3 0 3 S 3 C1 Calculated: C, 50.64; H, 4.86 ; N, 8.44; S, 19.31; CI, 7.12 (%), Found: C, 50.60; H , 4.75; N, 8.62; S, 19.18; CI, 7.08 (¾). Melting point: 198.5-199.5 ° C
実施例 7-204 Example 7-204
4-ク口口- N-{5, 6-ジメチル -8- [(4-メチルペンチル)スルファニル ]-3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン- 1-ィリデン }ベンゼンスルホ ンアミド 4- ク 口 口 -N- {5,6-dimethyl-8-[(4-methylpentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] thiazine-1-ylidene} benzenesulfonamide
実施例 6- 16で得た化合物(165 mg, 0.530腿 ol) の THF (3.5 ml) 懸濁液に水 素化ナトリウム(純度 60% 油性, 46.6 mg, 1.17腿 ol) を室温で加え、 10分間攪 拌後、 塩化 4-クロ口ベンゼンスルホニル(123 mg, 0.583匪 ol)を加え、 さらに 2.5 時間攪拌した。反応液を酢酸ェチルで希釈し、 0.1N塩酸および飽和食塩水で洗浄 し、 乾燥、 濃縮した。 残渣にメタノールを加え、 析出物を濾取し、 メタノールで 洗浄後、 乾燥して標題化合物 (154 mg, 0.317 mmol)を紫色粉末として得た。 'H-NMR (200MHz, CDC13) : 50.90 (6H, d, J= 6.6 Hz), 1.28 (2H, brq, J= 7.7 Hz), 1.59 (1H, quint, J= 6.4 Hz), 1.68 (2H, m), 1.79 (3H, brs), 2.28 (3H, brs), 2.93 (2H, brt, J= 7.5 Hz), 7.46 (2H, d, J= 8.4 Hz), 7.91 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Sodium hydride (purity 60% oily, 46.6 mg, 1.17 t) was added to a suspension of the compound obtained in Example 6-16 (165 mg, 0.530 t) in THF (3.5 ml) at room temperature. After stirring for one minute, 4-chlorobenzenebenzenesulfonyl chloride (123 mg, 0.583 ol) was added and the mixture was further stirred for 2.5 hours. The reaction solution was diluted with ethyl acetate, washed with 0.1N hydrochloric acid and saturated saline, dried and concentrated. Methanol was added to the residue, the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (154 mg, 0.317 mmol) as a purple powder. 'H-NMR (200MHz, CDC1 3): 50.90 (6H, d, J = 6.6 Hz), 1.28 (2H, brq, J = 7.7 Hz), 1.59 (1H, quint, J = 6.4 Hz), 1.68 (2H , m), 1.79 (3H, brs), 2.28 (3H, brs), 2.93 (2H, brt, J = 7.5 Hz), 7.46 (2H, d, J = 8.4 Hz), 7.91 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C2flH24N303S3Clとして計算値: C, 49.42; H, 4.98; N, 8.64; S, 19.79; CI, 7.29(¾), 実測値: C, 49.44; H, 5.03; N, 8.70; S, 19.76; CI, 7.28(¾). 融点: 163.0-163.5°C Elemental analysis: C 2fl H 24 N 3 0 3 S 3 Calculated Cl: C, 49.42; H, 4.98; N, 8.64; S, 19.79; CI, 7.29 (¾), Found: C, 49.44; H , 5.03; N, 8.70; S, 19.76; CI, 7.28 (¾). Melting point: 163.0-163.5 ° C
実施例 7-205 Example 7-205
4 -プロモ -N- [8 -(シクロへプチルスルファ二ノレ) -5, 6-ジメチソレ -3-ォキソ -2, 3-ジ ヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン] ベンゼンスルホンアミ K 4-Promo-N- [8- (cycloheptylsulfaninole) -5,6-dimethisole-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1-ylidene] benzenesulfonami K
実施例 6- 15で得た化合物(175 mg, 0.562讓 ol) の THF (3.5 ml) 懸濁液に水 素化ナトリウム(純度 60%油性, 56.2 mg) を室温で加え、 10 分間攪拌後、 塩化 4 -ブロモベンゼンスルホニル(222 mg, 0.850腿 ol)を加え、 さらに 50分間攪拌し た。 反応液に酢酸ェチルを加えて希釈し、 0.1N塩酸、 水、 および飽和食塩水で洗 浄後、 乾燥、 濃縮した。 残渣にメタノールを加え、 析出物を濾取し、 メタノール で洗浄後、 乾燥して標題化合物 (234 mg, 0.470匪 ol)を紫色粉末として得た。 'H-NMR (200MHz, 重クロ口ホルム) : δ 1.50-1.75 (10H, m), 1.80 (3H, q, J=0.8 Hz), 2.05 (2H, m), 2.27 (3H, q, J= 1.2 Hz), 3.59 (1H, sept, J= 4.4 Hz), 7.62 (2H, dt, J= 8.8, 2.2 Hz), 7.84 (2H, dt, J= 8.8, 2.2 Hz), 9.66 (1H, brs). 元素分析値: C21H24N303S3Br として計算値: C, 46.49; H, 4.46; N, 7.75; S, 17.73; Br, 14.73 ( ), 実測値; C, 46.55; H, 4.57; N, 7.81; S, 17.73; Br, 14.40 (%) . To a suspension of the compound obtained in Example 6-15 (175 mg, 0.562 benzyl) in THF (3.5 ml) was added sodium hydride (purity 60%, oily, 56.2 mg) at room temperature, and the mixture was stirred for 10 minutes. chloride 4-Bromobenzenesulfonyl (222 mg, 0.850 mol) was added, and the mixture was further stirred for 50 minutes. The reaction mixture was diluted with ethyl acetate, washed with 0.1N hydrochloric acid, water, and saturated saline, dried and concentrated. Methanol was added to the residue, and the precipitate was collected by filtration, washed with methanol, and dried to give the title compound (234 mg, 0.470 bandol) as a purple powder. 'H-NMR (200 MHz, double-mouthed form): δ 1.50-1.75 (10H, m), 1.80 (3H, q, J = 0.8 Hz), 2.05 (2H, m), 2.27 (3H, q, J = 1.2 Hz), 3.59 (1H, sept, J = 4.4 Hz), 7.62 (2H, dt, J = 8.8, 2.2 Hz), 7.84 (2H, dt, J = 8.8, 2.2 Hz), 9.66 (1H, brs) . elemental analysis: C 21 H 24 N 3 0 3 S 3 Br calculated: C, 46.49; H, 4.46 ; N, 7.75; S, 17.73; Br, 14.73 (), Found; C, 46.55; H , 4.57; N, 7.81; S, 17.73; Br, 14.40 (%).
融点: 190.0-191.5°C Melting point: 190.0-191.5 ° C
実施例 7-206 Example 7-206
4 -ブロモ -N- {5, 6-ジメチル- 8- [ (4-メチルペンチル)スルファニル] -3-ォキソ - 2, 3-ジヒドロ -1H -イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }ベンゼンスルホ ンアミド , 4-Bromo-N- {5,6-dimethyl-8-[(4-methylpentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene} benzenesulfonamide,
実施例 6- 16で得た化合物(175 mg, 0.562腿 ol) の THF (3.5 ml) 懸濁液に水素 '化ナトリウム (純度 60%油性, 56.2 mg, 1.41腿 ol) を室温で加え、 10分間攪拌 後、 塩化 4 -ブロモベンゼンスルホニル mg, 0.618腿 ol)を加え、 さらに 45 分間攪拌した。 反応液に 1N塩酸 (1.41 ml) およびメタノール -水 (1:1)を加え て 10分間攪拌した。析出物を濾取し、メタノール-水 (1:1) およびメタノールで 洗浄後、 乾燥して標題化合物 (165 mg, 0.311 匪 ol).を赤紫色粉末として得た。 'H-NMR (300MHz, 重クロ口ホルム) : δ 0.90 (6Η, d, J= 6.6 Hz), 1.26 (1H, brt, J= 6.9 Hz), 1.29 (1H, brt, J= 6.6 Hz), 1.56 (1H, m), 1.67 (1H, m), 1.68 (1H, quint, J= 7.7Hz), 1.80 (3H, q, J= 0.9 Hz) , 2.28 (3H, q, J= 0.9 Hz), 2.93 (2H, t, J= 7.5 Hz), 7.63 (2H, dt, J= 9.0, 2.1 Hz), 7.84 (2H, dt, J= 8.7, 2.1 Hz), 9.60 (1H, brs). To a suspension of the compound obtained in Example 6-16 (175 mg, 0.562 t ol) in THF (3.5 ml) was added sodium hydride (purity 60% oily, 56.2 mg, 1.41 t ol) at room temperature. After stirring for 4 minutes, 4-bromobenzenesulfonyl chloride (mg, 0.618 ml) was added, and the mixture was further stirred for 45 minutes. 1N Hydrochloric acid (1.41 ml) and methanol-water (1: 1) were added to the reaction solution, and the mixture was stirred for 10 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (165 mg, 0.311 bandol) as a red-purple powder. 'H-NMR (300 MHz, double-mouthed form): δ 0.90 (6Η, d, J = 6.6 Hz), 1.26 (1H, brt, J = 6.9 Hz), 1.29 (1H, brt, J = 6.6 Hz), 1.56 (1H, m), 1.67 (1H, m), 1.68 (1H, quint, J = 7.7Hz), 1.80 (3H, q, J = 0.9 Hz), 2.28 (3H, q, J = 0.9 Hz), 2.93 (2H, t, J = 7.5 Hz), 7.63 (2H, dt, J = 9.0, 2.1 Hz), 7.84 (2H, dt, J = 8.7, 2.1 Hz), 9.60 (1H, brs).
元素分析値: C20H24N303S3Br として計算値: C, 45.28; H, 4.56; N, 7.92; S, 18.13Elemental analysis: C 20 H 24 N 3 0 3 S 3 Br Calculated: C, 45.28; H, 4.56 ; N, 7.92; S, 18.13
(%), 実測値; C, 45.16; H, 4.42; N, 7.68; S, 18.01 (%) . (%), Found; C, 45.16; H, 4.42; N, 7.68; S, 18.01 (%).
融点: 184.5-185. (TC 実施例 8 - 1 Melting point: 184.5-185. (TC Example 8-1
N - [5, 6-ジメチル- 3-ォキソ -8- (ペンチルスルファ二ル)- 2, 3-ジヒドロ- 1H-イミダ ' ゾ [5,l_c] [1,4]チアジン -1-イリデン] - 4 -メチル安息香酸アミド N- [5,6-Dimethyl-3-oxo-8- (pentylsulfanyl) -2,3-dihydro-1H-imida'azo [5, l_c] [1,4] thiazine-1-ylidene] -4-Methylbenzoic acid amide
方法 A . Method A.
実施例 6-1で得られた卜ァミノ- 5, 6-ジメチル 8- (ペンチルスルファニル) -3H- イミダゾ [5, 卜 c] [1,4]チアジン- 3_オン (150 mg, 0.504匪 ol) をピリジン (3.00 ml) に溶かし、 4-メチルベンゾィル クロリド (0.102 ml, 0.756醒 ol) を加えた。 反応混合物を室温で 18時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出し た。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカ ゲルカラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル (10:1, v/v) で 溶出して題記化合物 (82.9mg, 40%) を暗赤色結晶として得た。 酢酸ェチル -ジェ チルェ一テルから再結晶して暗赤色結晶を得た。 Triamino-5,6-dimethyl 8- (pentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazin-3_one obtained in Example 6-1 (150 mg, 0.504 ol Was dissolved in pyridine (3.00 ml), and 4-methylbenzoyl chloride (0.102 ml, 0.756 ol) was added. After stirring the reaction mixture at room temperature for 18 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (82.9 mg, 40%) as dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals.
方法 B Method B
実施例 6-1で得られた卜ァミノ -5, 6-ジメチル- 8- (ペンチルスルファニル) -3H- イミダゾ [5, 1-c] [1, 4]チアジン- 3-オン (150 mg, 0.504匪 ol) をテトラヒドロフ ラン (3.00ml) に溶かし、 4-メチルベンゾィル クロリド (0.102 ml, 0.756腿 ol) 及びトリェチルァ ン (0.141 ml, 1.01腿 ol) を順次加えた。 反応混合物を室温 で 18時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (74.3mg, 36%) を暗赤色 結晶として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 196- 198°Cの 暗赤色結晶を得た。 Triamino-5,6-dimethyl-8- (pentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-1 (150 mg, 0.504 Ol) was dissolved in tetrahydrofuran (3.00 ml), and 4-methylbenzoyl chloride (0.102 ml, 0.756 t ol) and triethylan (0.141 ml, 1.01 t ol) were sequentially added. After stirring the reaction mixture at room temperature for 18 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (74.3 mg, 36%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 196-198 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3285, 2071, 2955, 2936, 2861, 1736, 1661, 1603, 1549, 1508 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3285, 2071, 2955, 2936, 2861, 1736, 1661, 1603, 1549, 1508 cm " 1 .
Ή-NMR (CDC13) : δ 0.949 (3Η, t, J=7.0Hz), 1.30-1.58 (4H, m), 1.70-1.88 (2H, m), 1.83 (3H, d, J=1.0 Hz), 2.34 (3H, d, J=l. Hz), 2.41 (3H, s), 3.06 (2H, t, J=7.4 Hz), 7.24 (2H, d, J-8.0 Hz), 8.23 (2H, d, J=8.4 Hz), 11.09 (1H, bs). 元素分析値: C21H25N302S2として Ή-NMR (CDC1 3): δ 0.949 (3Η, t, J = 7.0Hz), 1.30-1.58 (4H, m), 1.70-1.88 (2H, m), 1.83 (3H, d, J = 1.0 Hz) , 2.34 (3H, d, J = l. Hz), 2.41 (3H, s), 3.06 (2H, t, J = 7.4 Hz), 7.24 (2H, d, J-8.0 Hz), 8.23 (2H, d , J = 8.4 Hz), 11.09 (1H, bs). Elemental analysis: C 21 H 25 N 3 0 2 S 2
計算値 (%) : C, 60.69; H, 6.06; N, 10.11; S, 15.43. Calculated value (%): C, 60.69; H, 6.06; N, 10.11; S, 15.43.
実測値 (%) : C, 60.64; H, 5.90; N, 10.02; S, 15.50. 実施例 8-2 Found (%): C, 60.64; H, 5.90; N, 10.02; S, 15.50. Example 8-2
N - [5, 6-ジメチル -8- (ィソペンチルスルファニル) -3-ォキソ -2, 3-ジヒドロ- 1H -ィ ミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン ] -2-フエニル酢酸アミド N- [5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] 2-phenylacetic acid amide
実施例 8-1方法 Bと同様の方法により、 4-メチルベンゾィル クロリドの代わり にフエニルァセチル クロリド (0.0504 ml, 0.363腿 ol) を用いて、 実施例 6-2 で得られた 1-7ミノ -5, 6-ジメチル -8 - (ィソぺンチルスルファニル) - 3H-ィミダ ゾ [5, 1-c] [1, 4]チアジン- 3-オン (90.0 mg, 0.303腿。1)から題記化合物 (59.1 rag, 47%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 152- 153°C の暗赤色結晶を得た。 Example 8-1 In the same manner as in Method B, except that phenylacetyl chloride (0.0504 ml, 0.363 t ol) was used instead of 4-methylbenzoyl chloride, 1-7 mino-5 obtained in Example 6-2 was used. 6-Dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5,1-c] [1,4] thiazin-3-one (90.0 mg, 0.303 t.1) from the title compound (59.1 rag, 47%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 152-153 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3196, 2955, 2928, 2868, 1746, 1620, 1599, 1541, 1495 cm—1. Infrared absorption spectrum (IR) (in KBr) V: 3196, 2955, 2928, 2868, 1746, 1620, 1599, 1541, 1495 cm- 1 .
NMR (CDC13) : δ 0.968 (6Η, d, J=6.4 Hz), 1.38-1.74 (3H, m), 1.80 (3H, s), 2.30 (3H, s), 2.88-3.05 (2H, m), 3.83 (2H, s), 7.29-7.40 (5H, m), 10.81 (1H, bs) NMR (CDC1 3): δ 0.968 (6Η, d, J = 6.4 Hz), 1.38-1.74 (3H, m), 1.80 (3H, s), 2.30 (3H, s), 2.88-3.05 (2H, m) , 3.83 (2H, s), 7.29-7.40 (5H, m), 10.81 (1H, bs)
元素分析値: C21H25N302S2として Elementary analysis: as C 21 H 25 N 3 0 2 S 2
計算値 (%) : C, 60.69; H, 6.06; N, 10.11; S, 15.43. Calculated value (%): C, 60.69; H, 6.06; N, 10.11; S, 15.43.
実測値 (%) : C, 60.60; H, 5.99; N, 9.99; S, 15.35. Found (%): C, 60.60; H, 5.99; N, 9.99; S, 15.35.
実施例 8 - 3 Example 8-3
N- [5, 6-ジメチル -3-ォキソ -8 - (ぺンチルスルファニル) -2, 3-ジヒド口- 1H-ィミダ ゾ [5, 1-c] [1, 4]チアジン-卜ィリデン ]-4-トリフルォロメチル安息香酸アミド 実施例 8-1方法 Bと同様の方法により、 4-メチルベンゾィル クロリドの代わり に 4-トリフルォロメチルベンゾィル クロリド (71.6mg, 0.333腿 ol)を用いて、 実施例 6-2 で得られた 1-ァミノ- 5, 6 -ジメチル- 8 -(イソペンチルスルファ二 ル) -3H-イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (90.0 mg, 0.303 mmol) から題記 化合物 (82.0mg, 57%) を合成した。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 186- 188°Cの暗赤色結晶を得た。 N- [5,6-Dimethyl-3-oxo-8- (pentylsulfanyl) -2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-triylidene] -4-Trifluoromethylbenzoylamide Example 8-1 By the same method as in Method B, 4-trifluoromethylbenzoyl chloride (71.6 mg, 0.333 t) was used in place of 4-methylbenzoyl chloride. Thus, 1-amino-5,6-dimethyl-8- (isopentylsulfanyl) -3H-imidazo [5, tric] [1,4] thiazine-3--obtained in Example 6-2. The title compound (82.0 mg, 57%) was synthesized from ON (90.0 mg, 0.303 mmol). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 186-188 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3272, 3071, 2967, 2963, 2921, 2878, 1732 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3272, 3071, 2967, 2963, 2921, 2878, 1732 cm " 1 .
Ή— NMR (CDCL) : δ 1.00 (6Η, d, J=6.4 Hz) , 1.62-1.84 (3H, m), 1.85 (3H, s), 2.36 (3H, s), 3.09 (2H, t, J=7.6 Hz), 7.70 (2H, d, J=8.4 Hz), 8.43 (2H, d,Ή— NMR (CDCL): δ 1.00 (6Η, d, J = 6.4 Hz), 1.62-1.84 (3H, m), 1.85 (3H, s), 2.36 (3H, s), 3.09 (2H, t, J = 7.6 Hz), 7.70 (2H, d, J = 8.4 Hz), 8.43 (2H, d,
J=8.2 Hz), 11.11 (1H, bs). J = 8.2 Hz), 11.11 (1H, bs).
元素分析値: C21H22N302S2F3-0.4¾0として Elemental analysis: C 21 H 22 N 3 0 2 S 2 F 3 -0.4¾0
計算値 (%) : C, 52.91; H, 4.82; N, 8.81; S, 13.45. Calculated (%): C, 52.91; H, 4.82; N, 8.81; S, 13.45.
実測値 (¾) : C, 52.90; H, 4.72; N, 8.60; S, 13.47. Found (¾): C, 52.90; H, 4.72; N, 8.60; S, 13.47.
以下に実施例 8- 1〜8-3 で合成した化合物および同様にして合成した化合物の 一覧を示す。 The following is a list of compounds synthesized in Examples 8-1 to 8-3 and compounds synthesized in the same manner.
〔表 9 1〕(Table 9 1)
差替え用紙(ΜΜ2β) 〔表 9 2〕Replacement paper (ΜΜ2β) (Table 9 2)
差替え用弒(規則 26) 〔表 93〕For replacement (Rule 26) (Table 93)
差替え用紙(規則 ) 以下に実施例 8-4〜8- 31で合成した化合物のデータを示す。 Replacement form (rules) The data of the compounds synthesized in Examples 8-4 to 8-31 are shown below.
実施例 8-4 Example 8-4
¾-NMR (CDC13, 300 MHz): δ 2.84 (2H, t, J= 7.3 Hz), 3.35 (2H, t, J= 7.3 Hz), 3.74 (3H, s), 5.11 (1H, d, J= 8.0 Hz), 6.78 (1H, d, J= 8.0 Hz), 7.46 (2H, brt, J= 7.4 Hz), 7.55 (1H, brt, J= 7.2 Hz), 8.31 (2H, brd, J= 7.2 Hz), 11.22 (1H, brs). ¾-NMR (CDC1 3, 300 MHz): δ 2.84 (2H, t, J = 7.3 Hz), 3.35 (2H, t, J = 7.3 Hz), 3.74 (3H, s), 5.11 (1H, d, J = 8.0 Hz), 6.78 (1H, d, J = 8.0 Hz), 7.46 (2H, brt, J = 7.4 Hz), 7.55 (1H, brt, J = 7.2 Hz), 8.31 (2H, brd, J = 7.2) Hz), 11.22 (1H, brs).
元素分析値: C17H15N304S2として計算値: C, 52.43; H, 3.88; N, 10.79; S, 16.47Elemental analysis: C 17 H 15 N 3 0 4 S 2 Calculated: C, 52.43; H, 3.88 ; N, 10.79; S, 16.47
(%), 実測値: C, 52.51; H, 3.91; , 10.89; S, 16.43 (%) . (%), Found: C, 52.51; H, 3.91;, 10.89; S, 16.43 (%).
実施例 8-5 Example 8-5
ェチル 5 -({5, 6-ジメチル -1- [4 -(メチルペンゾィル)ィミノ ]-3_ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5, l_c] [1,4]チアジン- 8-ィル}スルファニル)ペン夕ノエ一ト 実施例 6 - 24 で得られた 5-[1-アミノ- 5,6-ジメチル-3-ォキソ-311-ィミダゾ [5,1- c][l,4]チアジン - 8_ィル]スルファニル]ペン夕ノエ一ト (600 mg, 1.49 腿 ol) と リエチルァミン (0.312ml, 2.24腿01) のテトラヒドロフラン (6.00 ml) 溶液に、 p -トルィル クロリド (0.224 ml, 1.64 mmol) を氷冷下加え、 室温 で 22時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(430 mg, 61¾) を合成した。酢酸ェチル-ジェチルエーテルから再結晶して融点 163- 165°Cの暗赤 色結晶を得た。 Ethyl 5-({5,6-dimethyl-1- [4- (methylbenzoyl) imino] -3_oxo-2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine-8 {Sulfanyl) pentyl 5- [1-amino-5,6-dimethyl-3-oxo-311-imidazo [5,1-c] [l, 4] obtained in Example 6-24. [Thiazine-8_yl] sulfanyl] pennoate (600 mg, 1.49 t) and riethylamine (0.312 ml, 2.24 t) in tetrahydrofuran (6.00 ml), p-tolyl chloride (0.224 ml, 1.64 mmol) was added under ice-cooling, and the mixture was stirred at room temperature for 22 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), the solvent was evaporated under reduced pressure to give the title compound (430 mg, 61¾) was synthesized. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 163-165 ° C.
IR (KBr) ν: 3275, 306, 2980, 2905, 1738, 1603, 1547, 1508 cm"1. IR (KBr) ν: 3275, 306, 2980, 2905, 1738, 1603, 1547, 1508 cm " 1 .
¾-NMR (CDC13) : (51.27 (3H, d, J-7.2 Hz), 1.76-1.88 (4H, m), 1.84 (3H, s), 2.34 (3H, d, J=1.2 Hz), 2.36-2.44 (2H, m), 2.41 (3H, s), 3.02-3.12 (2H, m), 4.15 (2H, q, J=7.2 Hz), 7.25 (2H, d, J=7.2 Hz), 8.22 (2H, d, J=8.1 Hz), 11.08 (1H, bs). ¾-NMR (CDC1 3): (51.27 (3H, d, J-7.2 Hz), 1.76-1.88 (4H, m), 1.84 (3H, s), 2.34 (3H, d, J = 1.2 Hz), 2.36 -2.44 (2H, m), 2.41 (3H, s), 3.02-3.12 (2H, m), 4.15 (2H, q, J = 7.2 Hz), 7.25 (2H, d, J = 7.2 Hz), 8.22 ( 2H, d, J = 8.1 Hz), 11.08 (1H, bs).
元素分析 f直: C23H27N304S2として Elemental analysis f straight: as C 23 H 27 N 3 0 4 S 2
計算値 (¾) : C, 58.33; H, 5.75; , 8.87; S, 13.54. Calculated value (¾): C, 58.33; H, 5.75;, 8.87; S, 13.54.
実測値 (¾) : C, 57.95; H, 5.86; , 8.75; S, 13.68. Found (¾): C, 57.95; H, 5.86;, 8.75; S, 13.68.
実施例 8 - 6 Example 8-6
5,6-ジメチル _1._[({[(4-メチルフエニル)スルホニル]アミノ}カルボニル)イミ ノ] - 3-ォキソ -8 - [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニル ]-2, 3 -ジヒ ドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン 5,6-dimethyl _1 ._ [({[(4-methylphenyl) sulfonyl] amino} carbonyl) imi [No] -3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [1, 4] thiazine
実施例 6- 6で得た化合物(223 mg, 0.556醒 ol) の THF (4.0 ml) 懸濁液にイソ シアン酸 P-トルエンスルホニル (123 mg, 0.611 mmol) を室温で加え、 20分間攪 拌した。 反応液にジェチルエーテル(10 ml)を加え、 析出物を濾取し、 ジェチルェ 一テルで洗浄後、 乾燥して表題化合物 (227 mg, 0.379匪 ol, 68.2%)を褐紫色粉 末として得た。 , To a suspension of the compound obtained in Example 6-6 (223 mg, 0.556 mmol) in THF (4.0 ml) was added P-toluenesulfonyl isocyanate (123 mg, 0.611 mmol) at room temperature, and the mixture was stirred for 20 minutes. did. To the reaction mixture was added getyl ether (10 ml), and the precipitate was collected by filtration, washed with getyl ether, and dried to obtain the title compound (227 mg, 0.379 marl, 68.2%) as a brown-purple powder. Was. ,
lH— NMR (300MHz, CDC13): δ 1.78 (4Η, m), 1.80 (3H, q, J= 1.2 Hz), 2.08 (2H, m), 2.28 (3H, q, J= 0.9 Hz), 2.44 (3H, s), 3.00 (2H, t, J= 7.1 Hz) , 7.33 (2H, d, J= 8.4 Hz), 7.97 (2H, d, J= 8.1 Hz), 約 10.3 (1H, br). lH- NMR (300MHz, CDC1 3) : δ 1.78 (4Η, m), 1.80 (3H, q, J = 1.2 Hz), 2.08 (2H, m), 2.28 (3H, q, J = 0.9 Hz), 2.44 (3H, s), 3.00 (2H, t, J = 7.1 Hz), 7.33 (2H, d, J = 8.4 Hz), 7.97 (2H, d, J = 8.1 Hz), about 10.3 (1H, br).
元素分析値: C22l N404S3F5として計算値: C, 44.14; H, 3.87; N, 9.36; S, 16.07; F, 15.87 (%) 、 実測値: C, 43.90; H, 3.77; N, 9.38; S, 16.34 (%) . Elemental analysis: C 22 l N 4 0 4 S 3 F 5 Calculated: C, 44.14; H, 3.87 ; N, 9.36; S, 16.07; F, 15.87 (%), Found: C, 43.90; H , 3.77; N, 9.38; S, 16.34 (%).
mp. 132.5-135.5°C mp.132.5-135.5 ° C
実施例 8- 7 Example 8-7
8-[(5-フルォロペンチル) スルファニル ]_5,6-ジメチル -1_[({[(4_メチルフエ二 ル)スルホニル]アミノ }力ルポニル)ィミノ] -3 -ォキソ _2, 3-ジヒドロ- 1H -ィミダ ゾ [5,卜 c] [1,4]チアジン ' 8-[(5-Fluoropentyl) sulfanyl] _5,6-dimethyl -1 _ [({[(4_methylphenyl) sulfonyl] amino} capillonyl) imino] -3-oxo-2,3-dihydro-1H-imida Zo [5, u c] [1,4] thiazine ''
実施例 6-23で得た化合物 (212 mg, 0.625讓 ol) の THF (4.0 ml) 懸濁液にィ ソシアン酸 P-トルエンスルホニル (139 mg, 0.688腿 ol) を室温で加え、 20分間 攪拌した。 反応液にジェチルェ一テル(10 ml)を加え、 析出物を濾取し、 ジェチル エーテルで洗浄後、 乾燥して表題化合物(196 mg, 0.382匪 oI, 61.2%)を紫色粉末 として得た。 To a suspension of the compound (212 mg, 0.625 alcohol) obtained in Example 6-23 in THF (4.0 ml) was added P-toluenesulfonyl isocyanate (139 mg, 0.688 mol) at room temperature, and the mixture was stirred for 20 minutes. did. To the reaction mixture was added getyl ether (10 ml), and the precipitate was collected by filtration, washed with getyl ether, and dried to obtain the title compound (196 mg, 0.382 band-oI, 61.2%) as a purple powder.
NMR (300MHz, CDC13): 51.59 (2H, m), 1.73 (4H, m), 1.79 (3H, s), 2.28 (3H, s), 2.44 (3H, s), 3.00 (2H, t, J= 7.2 Hz), 4.47 (2H, dt, J= 47.4, 5.7 Hz), 7.33 (2H, d, J= 8.4 Hz), 7.97 (2H, d, J= 8.1 Hz), 約 10.3 (1H, br). NMR (300MHz, CDC1 3): 51.59 (2H, m), 1.73 (4H, m), 1.79 (3H, s), 2.28 (3H, s), 2.44 (3H, s), 3.00 (2H, t, J = 7.2 Hz), 4.47 (2H, dt, J = 47.4, 5.7 Hz), 7.33 (2H, d, J = 8.4 Hz), 7.97 (2H, d, J = 8.1 Hz), about 10.3 (1H, br) .
元素分析値: C21H25N404S3Fとして計算値: C, 49.20; H, 4.92; N, 10.93; S, 18.76Elemental analysis: Calculated for C 21 H 25 N 4 0 4 S 3 F: C, 49.20; H, 4.92; N, 10.93; S, 18.76
( ) 、 実測値: C, 48.89; H, 4.93; N, 11.12; S, 18.96 (%) . (), Found: C, 48.89; H, 4.93; N, 11.12; S, 18.96 (%).
即. 116.0-118.0°C . Immediately.116.0-118.0 ° C.
実施例 8 - 8 5, 6 -ジメチル -1- [({[ (4-メチルフ ニル)スルホニル]アミノ }力ルポニル)ィミ ノ ] - 3-ォキソ- 8 - [(4,4, 5, 5, 5-ペン夕フルォロぺンチル)スルファニル] -2, 3-ジヒ ドロ- 1H-イミダゾ [5,l_c] [1,4]チアジン Example 8-8 5, 6-dimethyl-1-[({[(4-methylphenyl) sulfonyl] amino} aminopropionyl) imino] -3-oxo-8-[(4,4,5,5,5- Fluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine
実施例 6-3で得た化合物 (209 mg, 0.540腿 ol) の THF (4.0 ml) 懸濁液にイソ シアン酸 P-トルエンスルホニル (119 mg, 0.593腿 ol) を室温で加え、 40分間攪 拌した。 反応液にジェチルエーテル(10 ml)を加え、 析出物を濾取し、 ジェチルェ —テルで洗浄後、 乾燥して表題化合物(255 mg, 0.436 mmol, 80.8%)を紫色粉末と して得た。 To a suspension of the compound obtained in Example 6-3 (209 mg, 0.540 mol) in THF (4.0 ml) was added P-toluenesulfonyl isocyanate (119 mg, 0.593 mol) at room temperature, and the mixture was stirred for 40 minutes. Stirred. To the reaction solution was added getyl ether (10 ml), and the precipitate was collected by filtration, washed with getyl ether, and dried to obtain the title compound (255 mg, 0.436 mmol, 80.8%) as a purple powder. .
'H-NMR (300MHz, CDC13): δ 1.80 (3Η, s), 2.05 (2H, q, J= 7.2 Hz), 2.20 (2H, m), 2.28 (3H, s), 2.44 (3H, s), 3.06 (2H, t, J= 7.4 Hz), 7.33 (2H, d, J= 8.1 Hz), 7.97 (2H, d, J= 8.1 Hz), 10.27 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 1.80 (3Η, s), 2.05 (2H, q, J = 7.2 Hz), 2.20 (2H, m), 2.28 (3H, s), 2.44 (3H, s ), 3.06 (2H, t, J = 7.4 Hz), 7.33 (2H, d, J = 8.1 Hz), 7.97 (2H, d, J = 8.1 Hz), 10.27 (1H, br).
元素分析値: C21H2lN404S3F5として計算値: C, 43.14; H, 3.62; , 9.58; S, 16.45Elemental analysis: C 21 H 2l N 4 0 4 S 3 F 5 Calculated: C, 43.14; H, 3.62 ;, 9.58; S, 16.45
(%) 、 実測値: C, 43.00; H, 3.51; N, 9.50; S, 16.54 (%) . (%), Found: C, 43.00; H, 3.51; N, 9.50; S, 16.54 (%).
mp. 143.5-145.5°C mp.143.5-145.5 ° C
実施例 8 - 9 Examples 8-9
N-{5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル] -2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1 -ィリデン }-Ν' - (4 -メチ ルフエニル)ゥレア N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene} -Ν '-(4-methylphenyl) ゥ rea
実施例 6- 6で得た化合物 (200 mg, 0.498 mmol) の THF (4.0 ml) 懸濁液に水素 化ナトリウム (純度 60%油性, 49.8mg, 1.25 mmol) を室温で加え、 10分間攪拌 後、 イソシアン酸 P-トリル(77 mg, 0.548 mmol)の THF (1 ml)溶液を 1時間かけ て滴下し、 さらに 30分間攪拌した。 反応液を酢酸ェチル (35 ml)で希釈し、 0.1N 塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣をメタノールで希釈した。 析出物を濾取し、 メタノールで洗浄後、 乾燥して 表題化合物(227 mg, 0.425讓 ol, 85.3%)を濃紫色粉末として得た。 Sodium hydride (purity 60% oily, 49.8 mg, 1.25 mmol) was added to a suspension of the compound (200 mg, 0.498 mmol) obtained in Example 6-6 in THF (4.0 ml) at room temperature, and the mixture was stirred for 10 minutes. A solution of P-tolyl isocyanate (77 mg, 0.548 mmol) in THF (1 ml) was added dropwise over 1 hour, and the mixture was further stirred for 30 minutes. The reaction solution was diluted with ethyl acetate (35 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (227 mg, 0.425%, 85.3%) as a dark purple powder.
'H-NMR (200MHz, CDC13): δ 1.79/1.81 (3H, s), 約 1.8 (4H, m), 2.10 (2H, m), 2.32/2.29 (3H, s), 2.34/2.32 (3H, s), 3.03 (2H, t, J= 7.2 Hz), 7.14 (2H, d, J= 7.8 Hz), 7.43 (2H, d, J= 7.8 Hz), 9.37/7.39 (1H, brs), 10.86/10.69 (1H, brs) . 元素分析値: C22H23N402S2F5として計算値: C, 49.43; H, 4.34; N, 10.48; S, 12.00; F, 17.77 ( ) 、 実測値: C, 49.66; H, 4.10; N, 10.52; S, 11.91; F, 17.56'H-NMR (200MHz, CDC1 3): δ 1.79 / 1.81 (3H, s), about 1.8 (4H, m), 2.10 (2H, m), 2.32 / 2.29 (3H, s), 2.34 / 2.32 (3H , s), 3.03 (2H, t, J = 7.2 Hz), 7.14 (2H, d, J = 7.8 Hz), 7.43 (2H, d, J = 7.8 Hz), 9.37 / 7.39 (1H, brs), 10.86 /10.69 (1H, brs). Elemental analysis: C 22 H 23 N 4 0 2 S 2 F 5 Calculated: C, 49.43; H, 4.34 ; N, 10.48; S, 12.00; F, 17.77 (), Found: C, 49.66; H , 4.10; N, 10.52; S, 11.91; F, 17.56
(%) . ' (%). '
mp. 122.0-123. O : mp. 122.0-123.O:
実施例 8 - 10 Example 8-10
N-{8-[(5-フルォロペンチル)スルファニル ]-5, 6-ジメチル- 3-ォキソ -2, 3-ジヒド ロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン }-Ν'- (4-メチルフエニル)ウレ ァ N- {8-[(5-Fluoropentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene} -Ν '-(4-methylphenyl) urea
実施例 6-23で得た化合物 (210 mg, 0.666顧 ol) の THF (4.0 ml) 懸濁液に水 素化ナトリウム (純度 60% 油性, 66.6mg, 1.66腿 ol) を室温で加え、 10分間攪 拌後、 イソシアン酸 P-トリル(103 mg, 0.732 ranol)の THF (1 ml)溶液を 30分間 かけて滴下し、さらに 30分間攪拌した。反応液に酢酸ェチル (35 ml) および 0.2N 塩酸 (20 ml)を加え、 有機層を 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナ トリウムで乾燥した。 濃縮乾固後、 残渣をメタノールで希釈した。 析出物を濾取 し、 メタノールで洗浄後、 乾燥して表題化合物(239 mg, 0.533腿 ol, 80.0%)を赤 褐色粉末として得た。 To a suspension of the compound obtained in Example 6-23 (210 mg, 0.666 mmol) in THF (4.0 ml) was added sodium hydride (purity 60% oil, 66.6 mg, 1.66 mol) at room temperature. After stirring for 1 minute, a solution of P-tolyl isocyanate (103 mg, 0.732 ranol) in THF (1 ml) was added dropwise over 30 minutes, and the mixture was further stirred for 30 minutes. Ethyl acetate (35 ml) and 0.2N hydrochloric acid (20 ml) were added to the reaction solution, and the organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, and then dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (239 mg, 0.533 mol, 80.0%) as a red-brown powder.
lH-NMR (200MHz, CDC13): δ 1.60 (2Η, m), 1.79 (3H, s), 1.80 (4H, i), 2.32/2.29 lH-NMR (200MHz, CDC1 3 ): δ 1.60 (2Η, m), 1.79 (3H, s), 1.80 (4H, i), 2.32 / 2.29
(3H, s), 2.39/2.43 (3H, s), 3.02 (2H, t, J= 7.4 Hz), 4.47 (2H, dt, J= 47.2,(3H, s), 2.39 / 2.43 (3H, s), 3.02 (2H, t, J = 7.4 Hz), 4.47 (2H, dt, J = 47.2,
5.6 Hz), 7.14 (2H, d, J- 8.0 Hz), 7.44/7.41 (2H, d, J= 8.4/8.0 Hz), 9.41/7.36 (1H, brs), 10.89/10.71 (1H, ¾rs). 5.6 Hz), 7.14 (2H, d, J- 8.0 Hz), 7.44 / 7.41 (2H, d, J = 8.4 / 8.0 Hz), 9.41 / 7.36 (1H, brs), 10.89 / 10.71 (1H, ¾rs).
元素分析値: C21H25N402S2F.0.5H20として計算値: C, 55.12; H, 5.73; , 12.24;Elemental analysis: C 21 H 25 N 4 0 2 S 2 F.0.5H 2 0 Calculated: C, 55.12; H, 5.73 ;, 12.24;
S, 14.02; F, 4.15 (%) 、 実測値: C, 55.12; H, 5.78; N, 12.21; S, 13.97; F,S, 14.02; F, 4.15 (%), Found: C, 55.12; H, 5.78; N, 12.21; S, 13.97; F,
4.03 ( ) . 4.03 ().
即. 99.5- 101.5°C Immediately. 99.5-101.5 ° C
実施例 8 - 11 Examples 8-11
N-{5, 6 -ジメチル- 3-ォキソ _8-[(4, 4, 5, 5, 5 -ペン夕フルォロペンチル)スルファニ ル] - 2, 3 -ジヒドロ -1H -イミダゾ [5,卜 c] [1,4]チアジン - 1-ィリデン }-Ν' _(4-メチ ルフエニル)ゥレア N- {5,6-Dimethyl-3-oxo_8-[(4,4,5,5,5-pentanofluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [ 1,4] thiazine-1-ylidene} -Ν '_ (4-methylphenyl) ゥ rea
実施例 6-3で得た化合物 (210 mg, 0.542讓 ol) の THF (4.0 ml) 懸濁液に水素 化ナトリウム (純度 60% 油性, 54.2 mg, 1.36腿 ol) を室温で加え、 10分間攪拌 後、 イソシアン酸 P-トリル(84 mg, 0.596膽 ol)の THF (1 nil)溶液を 60分間かけ て滴下し、 さらに 30 分間攪拌した。 反応液に酢酸ェチル (35 ml) および 0.2N 塩酸(20 ml)を加え、 有機層を 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナ トリウムで乾燥した。 濃縮乾固後、 残渣をメタノールで希釈した。 析出物を濾取 し、 メタノールで洗浄後、 乾燥して表題化合物(205 mg, 0.394 mmol, 72.7%)を紫 色粉末として得た。 Hydrogen was added to a suspension of the compound obtained in Example 6-3 (210 mg, 0.542 acetyl) in THF (4.0 ml). Add sodium iodide (purity 60% oily, 54.2 mg, 1.36 t) at room temperature, stir for 10 minutes, and add a solution of P-tolyl isocyanate (84 mg, 0.596 b) in THF (1 nil) for 60 minutes. The mixture was added dropwise and stirred for another 30 minutes. Ethyl acetate (35 ml) and 0.2N hydrochloric acid (20 ml) were added to the reaction solution, and the organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, and then dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (205 mg, 0.394 mmol, 72.7%) as a purple powder.
Ή一 NMR (200MHz, CDC13): δ 1.80/1.81 (3Η, . s) , 2.11 (2Η, m), 2.20 (2Η, m), 2.32/2.29 (3H, S), 2.33/2.34 (3H, s), 3.09/3.06 (2H, t, J= 7.0/5.8 Hz), 7.14/7.13 (2H, d, J= 8.0/8.2 Hz) , 7.44/7.41 (2H, d, J= 8.2/8.0 Hz) , 9.30/7.34 (1H, brs), 10.86/10.69 (1H, brs). Ή one NMR (200MHz, CDC1 3): δ 1.80 / 1.81 (. 3Η, s), 2.11 (2Η, m), 2.20 (2Η, m), 2.32 / 2.29 (3H, S), 2.33 / 2.34 (3H, s), 3.09 / 3.06 (2H, t, J = 7.0 / 5.8 Hz), 7.14 / 7.13 (2H, d, J = 8.0 / 8.2 Hz), 7.44 / 7.41 (2H, d, J = 8.2 / 8.0 Hz) , 9.30 / 7.34 (1H, brs), 10.86 / 10.69 (1H, brs).
元素分析値: C21 N402S2F5として計算値: C, 48.45; H, 4.07; N, 10.76; S, 12.32; F, 18.25 (%) 、 実測値: C, 48.57; H, 3.91; N, 10.84; S, 12.36; F, 18.22 (%) . Elemental analysis: C 21 N 4 0 2 S 2 F 5 Calculated: C, 48.45; H, 4.07; N, 10.76; S, 12.32; F, 18.25 (%), found: C, 48.57; H, 3.91; N, 10.84; S, 12.36; F, 18.22 (%).
mp. 127.0-128.5°C mp.127.0-128.5 ° C
実施例 8-12 Example 8-12
N- (4-ブロモフエニル) -N'-{8 - [(5-フルォロペンチル)スルファニル ]-5, 6-ジメチ ル- 3-ォキソ -2, 3 -ジヒド口 -1H-ィミダゾ [5, 1-c] [1, ]チアジン -1-ィリデン }ゥレ ァ N- (4-bromophenyl) -N '-{8-[(5-fluoropentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydric -1H-imidazo [5,1-c ] [1,] thiazine-1-ylidene
実施例 6-23で得た化合物 (176 mg, 0.558 mmol)の THF (3.5 ml) 懸濁液に水素 化ナトリウム(純度 60% 油性, 55.8 mg, 1.39讓 ol) を室温で加え、 10分間攪拌 後、 イソシアン酸 4_ブロモフエニル (125 mg, 0.614 mmol) を加え、 さらに 60 分間攪拌した。 反応液にメタノール (1ml) および 1N塩酸 (2.0ml)を加え、 メ ' タノール一水 (1:1, 15ml)で希釈し、 10分間攪拌した。 析出物を濾取し、 メタ ノール—水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物 (274 mg, 0.534匪 ol, 95.7%)を赤色粉末として得た。 To a suspension of the compound (176 mg, 0.558 mmol) obtained in Example 6-23 in THF (3.5 ml) was added sodium hydride (purity 60% oily, 55.8 mg, 1.39 acetyl) at room temperature, and the mixture was stirred for 10 minutes. Thereafter, 4_bromophenyl isocyanate (125 mg, 0.614 mmol) was added, and the mixture was further stirred for 60 minutes. Methanol (1 ml) and 1N hydrochloric acid (2.0 ml) were added to the reaction solution, and the mixture was diluted with methanol-one water (1: 1, 15 ml) and stirred for 10 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (274 mg, 0.534 bandol, 95.7%) as a red powder.
'H-NMR (200MHz, CDC13): 51.64 (2H, m), 1.80 (3H, s) , 1.80 (4Η, m), 2.34/2.29 (3Η, s), 3.03 (2H, t, J= 7.5 Hz), 4.47 (2H, dt, J= 46.8, 5.4 Hz), 7.45 (4H, m), 9.44 (1H, brs), 11.07/10.66 (1H, brs). mp. 150-152^ 'H-NMR (200MHz, CDC1 3): 51.64 (2H, m), 1.80 (3H, s), 1.80 (4Η, m), 2.34 / 2.29 (3Η, s), 3.03 (2H, t, J = 7.5 Hz), 4.47 (2H, dt, J = 46.8, 5.4 Hz), 7.45 (4H, m), 9.44 (1H, brs), 11.07 / 10.66 (1H, brs). mp. 150-152 ^
実施例 8 - 13 Examples 8-13
N-{5, 6-ジメチル- 8-[(5-フルォロペンチル)スルファニル ]-3-ォキソ - 2, 3-ジヒド 口 _ m -イミダゾ [5,卜 c] [1 , 4]チアジン-卜イリデン卜 4-メチルベンズアミド 水素化ナトリウム(25.4 mg, 0.634匪 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜アミノ- 5, 6-ジメチル 8- [5-フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5,1- c] [1,4]チアジン -3-オン (100 mg, 0.317mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに p—トルィル クロリド (0.0475 ml, 0.349廳 ol) のテトラヒドロ フラン (1.00ml) 溶液を加え、 さらに 30分間かき混ぜた。 反応混合物に 1N塩酸 を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去して題記化合物 (117 mg, 85%) を暗赤色結晶として得た。 酢酸ェチル-ジ ェチルエーテルから再結晶して融点 195- 197°Cの暗赤色結晶を得た。 N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydroxy mouth _ m -imidazo [5, tric] [1,4] thiazine-triidendent 4-Methylbenzamide To a suspension of sodium hydride (25.4 mg, 0.634 ol) (60% oil) in tetrahydrofuran (2.00 ml) was added triamino-5,6-dimethyl 8 obtained in Example 6-23. -[5-Fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 mg, 0.317 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled on ice. Stir for 5 minutes. To this was added a solution of p-toluyl chloride (0.0475 ml, 0.349 hall) in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 30 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (117 mg, 85%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 195-197 ° C.
IR (KBr) V : 3247, 2961, 1732, 1603, 1547, 1507 cm"1. IR (KBr) V: 3247, 2961, 1732, 1603, 1547, 1507 cm " 1 .
Ή-NMR (CDC13) : (51.50-1.92 (6H, m), 1.84 (3H, d, J=0.6 Hz), 2.34 (3H, d, J=0.6 Hz), 2.42 (3H, s), 3.08 (2H, t, J=7.5 Hz), 4.49 (2H, dt, J-47.1, 5.7 Hz), 7.24 (2H, d, J-8.1 Hz), 8.22 (2H, d, J=8.1 Hz), 11.09 (1H, bs). 元素分析値: C21H24N302S2Fとして Ή-NMR (CDC1 3): (51.50-1.92 (6H, m), 1.84 (3H, d, J = 0.6 Hz), 2.34 (3H, d, J = 0.6 Hz), 2.42 (3H, s), 3.08 (2H, t, J = 7.5 Hz), 4.49 (2H, dt, J-47.1, 5.7 Hz), 7.24 (2H, d, J-8.1 Hz), 8.22 (2H, d, J = 8.1 Hz), 11.09 (1H, bs). Elemental analysis: C 21 H 24 N 3 0 2 S 2 F
計算値 (%) : C, 58.17; H, 5.58; N, 9.69; S, 14.79. Calculated (%): C, 58.17; H, 5.58; N, 9.69; S, 14.79.
実測値 (%) : C, 58.30; H, 5.66; N, 9.76; S, 14.80. . Found (%): C, 58.30; H, 5.66; N, 9.76; S, 14.80.
実施例 8- 14 Example 8-14
4 -ブロモ -2 - [ ( {5, 6-ジメチル -8- [ (5-フルォロペンチル)スルファニル] -3-ォキソ - 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チァジン-1-ィリデン }ァミノ)力ルポ二 ル]フエニル アセテート 4-Bromo-2-[({5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thazin-1-ylidene} amino] phenyl] phenyl acetate
実施例 8 - 13と同様にして合成し、 酢酸ェチル -ジェチルエーテルから再結晶し て融点 220- 222°Cの暗赤色結晶を得た。 The compound was synthesized in the same manner as in Examples 8 to 13, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 220 to 222 ° C.
IR (KBr) レ : 3241, 2957, 2938, 2859, 1757, 1736, 1638, 1603, 1557 cm—1. 'H-NMR (CDCI3) : δ 1.62-1.78 (4Η, m), 1.78-1.92 (2H, m), 1.81 (3H, s), 2.35 (3H, s), 2.37 (3H, s), 3.09 (2H, t, J=7.2 Hz), 4.47 (2H, dt, J=47.4, 6.3 Hz)/ 6.95 (1H, d, J=7.8Hz), 7.60 (1H, dd, J=8.1, 1.8 Hz), 8.76 (1H, d, J-2.7 Hz), 10.92 (1H, bs). IR (KBr): 3241, 2957, 2938, 2859, 1757, 1736, 1638, 1603, 1557 cm— 1. 'H-NMR (CDCI3): δ 1.62-1.78 (4Η, m), 1.78-1.92 (2H , m), 1.81 (3H, s), 2.35 (3H, s), 2.37 (3H, s), 3.09 (2H, t, J = 7.2 Hz), 4.47 (2H, dt, J = 47.4, 6.3 Hz) / 6.95 (1H, d, J = 7.8Hz), 7.60 (1H, dd, J = 8.1, 1.8 Hz), 8.76 (1H, d, J-2.7 Hz), 10.92 (1H, bs).
元素分析値: C22H23N304S2BrFとして Elementary analysis: as C 22 H 23 N 3 0 4 S 2 BrF
計算値 (%) : C, 47.48; H, 4.17; N, 7.55; S, 11.52. Calculated value (%): C, 47.48; H, 4.17; N, 7.55; S, 11.52.
実測値 (%) : C, 47.45; H, 4.07; N, 7.52; S, 11.37. Found (%): C, 47.45; H, 4.07; N, 7.52; S, 11.37.
実施例 8-15 Example 8-15
4-ブ口モ- N- {5, 6-ジメチル -8- [ (5-フルォロぺンチル)スルファニル] -3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1 -ィリデン }- 2-ヒドロキシべ ンズアミド 4-Buguchimo-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1, 4] Thiazine-1-ylidene} -2-hydroxybenzamide
実施例 8-13と同様にして合成し、 酢酸ェチル-ジェチルエーテルから再結晶し て融点 224- 226°Cの暗赤色結晶を得た。 The compound was synthesized in the same manner as in Example 8-13, and recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 224-226 ° C.
IR (KBr) V: 3700-2700, 3303, 2922, 1742, 1601, 1541 cm"1. IR (KBr) V: 3700-2700, 3303, 2922, 1742, 1601, 1541 cm " 1 .
Ή-NMR (CDC13) : δΐ.58-1.96 (6Η, m), 1.86 (3H, d, J=0.9 Hz), 2.36 (3H, d, Ή-NMR (CDC1 3): δΐ.58-1.96 (6Η, m), 1.86 (3H, d, J = 0.9 Hz), 2.36 (3H, d,
J=1.2 Hz), 3.14 (2H, t, J=7.2 Hz), 4.48 (2H, dt, J=47.1, 6.0 Hz), 6.83 (1H, d, J=8.7 Hz), 7.47 (1H, dd, J=9.0, 3.0 Hz), 8.37 (1H, s), 12.20 (1H, bs). 元素分析値: C2flH21N303S2BrF 3 0として J = 1.2 Hz), 3.14 (2H, t, J = 7.2 Hz), 4.48 (2H, dt, J = 47.1, 6.0 Hz), 6.83 (1H, d, J = 8.7 Hz), 7.47 (1H, dd, J = 9.0, 3.0 Hz), 8.37 (1H, s), 12.20 (1H, bs). Elemental analysis: C 2fl H 21 N 3 0 3 S 2 BrF 3 0
計算値 (%) : C, 46.21; H, 4.19; N, 8.08; S, 12.34. Calculated value (%): C, 46.21; H, 4.19; N, 8.08; S, 12.34.
実測値 (%) : C, 46.17; H, 4.10; N, 8.07; S, 12.18. Found (%): C, 46.17; H, 4.10; N, 8.07; S, 12.18.
実施例 8-16 Example 8-16
N-{5, 6 -ジメチル- 8- [(5 -フルォロペンチル)スルファニル] - 3 ォキソ -2, 3 -ジヒド 口- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン }ニコチンアミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihido mouth-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene } Nicotinamide
実施例 8 - 13と同様にして合成し、 酢酸ェチル -ジェチルエーテルから再結晶し て融点 187-189°Cの暗赤色結晶を得た。 It was synthesized in the same manner as in Examples 8-13, and recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 187-189 ° C.
IR (KBr) リ: 2936, 1728, 1661, 1622, 1572, 1547 cm一1. IR (KBr) Re: 2936, 1728, 1661, 1622 , 1572, 1547 cm one 1.
!H-NMR (CDCI3) : 51.58-1.94 (6H, m), 1.86 (3H, s), 2.36 (3H, s), 3.09 (2H, t, J=6.9 Hz), 4.48 (2H, dt, J=47.1, 5.7 Hz), 7.38 (1H, dd, J=7.2, 5.4 Hz), 8.49 (1H, dd, J=9.9, 1.2 Hz), 8.73 (1H, d, J=4.8 Hz), 9.56 (1H, s), 11.13 (1H, bs). ! H-NMR (CDCI3): 51.58-1.94 (6H, m), 1.86 (3H, s), 2.36 (3H, s), 3.09 (2H, t, J = 6.9 Hz), 4.48 (2H, dt, J = 47.1, 5.7 Hz), 7.38 (1H, dd, J = 7.2, 5.4 Hz), 8.49 (1H, dd, J = 9.9, 1.2 Hz), 8.73 (1H, d, J = 4.8 Hz), 9.56 (1H , s), 11.13 (1H, bs).
元素分析値: C19H21N402S2FO.4H20として 計算値 ( ) : C, 53.35 ; H, 5.14; N, 13.10; S, 14.99. Elemental analysis: as C 19 H 21 N 40 2 S 2 FO.4H 20 Calculated (): C, 53.35; H, 5.14; N, 13.10; S, 14.99.
実測値 (%) : C, 53.54; H, 4.96; N, 13.17; S, 14.72. Found (%): C, 53.54; H, 4.96; N, 13.17; S, 14.72.
実施例 8-17 Example 8-17
N - {5, 6 -ジメチル -8- [ (5-フルォ口ぺンチル)スルファニル] -3-ォキソ- 2, 3-ジヒド 口- 1H-イミダゾ [5,1- c] [1,4]チアジン-卜イリデン }- 2-ピリジン力ルポキサミド 2 -ピコリル クロリド塩酸塩(67.4 mg, 0.352匪 oUのテトラヒドロフラン(1.00 ml) 溶液にトリェチルァミン (0.0736 ml, 0.528 mmol) を加え、 反応混合物を室 温で 5分間かき混ぜた。 水素化ナトリウム (25.4mg, 0.634 mmol) (60%油性) の テトラヒドロフラン(2.00ml)の懸濁液に、実施例 6-23で得られたトァミノ- 5, 6- ジメチル 8 - [5-フルォロペンチル]スルファニル ]-3H -ィミダゾ [5, 1-c] [1,4]チア ジン- 3-オン (100 mg, 0.317 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加 え、 氷冷下 5分間かき混ぜた。 これに上記で得られた 2 -ピコリル クロリドのテ トラヒドロフラン溶液を加え、さらに 3時間かき混ぜた。反応混合物に 1N塩酸を 加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に 留去して題記化合物 (83.2mg, 56%) を暗赤色結晶として得た。 酢酸ェチル -ジェ チルェ一テルから再結晶して融点 161- 163°Cの暗赤色結晶を得た。 N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] thiazine -Triylidene}-2-Pyridyl lipoxamide 2-picolyl chloride hydrochloride (67.4 mg, 0.352) To a solution of tetrahydrofuran (1.00 ml) in maraudal oU was added triethylamine (0.0736 ml, 0.528 mmol), and the reaction mixture was heated at room temperature for 5 minutes. To a suspension of sodium hydride (25.4 mg, 0.634 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the toamino-5,6-dimethyl 8- [5 obtained in Example 6-23. -Fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 mg, 0.317 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled on ice for 5 minutes. To this was added the 2-picolyl chloride solution obtained above in tetrahydrofuran, and the mixture was further stirred for 3 hours. It was. The reaction mixture 1N hydrochloric acid was added and extracted with acetic acid Echiru in the. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (83.2 mg, 56%) dark red crystals Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals, mp 161-163 ° C.
IR (KBr) V: 2936, 2861, 1715, 1557, 1539, 1505 cm—1. IR (KBr) V: 2936, 2861, 1715, 1557, 1539, 1505 cm— 1 .
^-NMR (CDC13) : δ 1.50-1.70 (2Η, ι), 1.70-1.92 (4H, m), 1.79 (3H, d, J=0.9 Hz), 2.38 (3H, d, 0.9 Hz), 3.08 (2H, t, J=7.5 Hz), 4.37 (2H, dt, J=47.4, 5.4 Hz), 7.54 (1H, dd, J=6.6, 4.8 Hz), 7.94 (1H, dt, J=7.8, 1.8 Hz), 8.34 (1H, d, J=7.8 Hz), 8.59 (1H, d, J=4.5 Hz). ^ -NMR (CDC1 3): δ 1.50-1.70 (2Η, ι), 1.70-1.92 (4H, m), 1.79 (3H, d, J = 0.9 Hz), 2.38 (3H, d, 0.9 Hz), 3.08 (2H, t, J = 7.5 Hz), 4.37 (2H, dt, J = 47.4, 5.4 Hz), 7.54 (1H, dd, J = 6.6, 4.8 Hz), 7.94 (1H, dt, J = 7.8, 1.8 Hz), 8.34 (1H, d, J = 7.8 Hz), 8.59 (1H, d, J = 4.5 Hz).
元素分析値: C19H2lN402S2F'0.3H20として Elemental analysis: C 19 H 2l N 4 0 2 S 2 F'0.3H 20
計算値 (%) : C, 53.58; H, 5.11; , 13.15; S, 15.06. Calculated value (%): C, 53.58; H, 5.11;, 13.15; S, 15.06.
実測値 (%) : C, 53.46; H, 5.04; N, 13.03; S, 15.33. Found (%): C, 53.46; H, 5.04; N, 13.03; S, 15.33.
実施例 8-18 ' Example 8-18 '
N - {5, 6-ジメチル -8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド ロ- 1 H -イミダゾ [5 , 1 -C] [ 1 , 4]チアジン-卜イリデン } -6-メチルニコチンアミド 実施例 6-23で得られた卜ァミノ -5, 6 -ジメチル 8- [5-フルォ口ぺンチル]スルフ ァニル ]- 3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.317讓 ol) と 6 - メチル二チン酸 (43.4mg, 0.349醒 ol) の N, N-ジメチルホルムアミ ド (3.00 ml) 溶液に、 シァノりん酸ジェチル (0.0564 ml, 0.349腿 ol)、 続いてトリェチルァ ミン (0.0486 ml, 0.349匪 ol) を加えた。 反応混合物を室温で 15時間かき混ぜ た後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (72.1 mg, 52¾) を暗赤色結晶として得た。 酢酸 ェチル -ジェチルェ一テルから再結晶して融点 200-202°Cの暗赤色結晶を得た。 IR (KBr) V: 3289, 3071, 2921, 2861, 1736, 1665, 1615, 1599, 1547 cm"1. Ή-NMR (CDC13) : «51.50-1.94 (6H, m), 1.85 (3H, s), 2.35 (3H, d, J=1.0 Hz), 2.63 (3H, s), 3.07 (2H, t, J=7.8 Hz), 4.48 (2H, dt, J-47.2, 5.8 Hz), 7.24 (1H, d, J=8.0Hz), 8.37 (1H, dd, J=8.0, 2.2 Hz), 9.47 (1H, d, J=1.8Hz), 11.08 (1H, bs). N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-C] [1,4] thiazine- Ilidene} -6-methylnicotinamide triamino-5,6-dimethyl 8- [5-fluoroopentyl] sulfanyl] -3H-imidazo [5,1-c] obtained in Example 6-23 1,4] thiazin-3-one (100 mg, 0.317 rol) and 6- To a solution of methylditinic acid (43.4 mg, 0.349 ol) in N, N-dimethylformamide (3.00 ml) was added getyl cyanophosphate (0.0564 ml, 0.349 t), followed by triethylamine (0.0486 ml, 0.349 ml). Ol). After stirring the reaction mixture at room temperature for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (72.1 mg, 52¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 200-202 ° C. IR (KBr) V:. 3289 , 3071, 2921, 2861, 1736, 1665, 1615, 1599, 1547 cm "1 Ή-NMR (CDC1 3):« 51.50-1.94 (6H, m), 1.85 (3H, s ), 2.35 (3H, d, J = 1.0 Hz), 2.63 (3H, s), 3.07 (2H, t, J = 7.8 Hz), 4.48 (2H, dt, J-47.2, 5.8 Hz), 7.24 (1H , d, J = 8.0Hz), 8.37 (1H, dd, J = 8.0, 2.2 Hz), 9.47 (1H, d, J = 1.8Hz), 11.08 (1H, bs).
元素分析値: C2QH23N402S2F · 0.3¾0として Elemental analysis: C 2 Q H 23 N 4 0 2 S 2 F · 0.3¾0
計算値 (¾) : C, 54.60; H, 5.41; N, 12.73; S, 14.58. Calculated (¾): C, 54.60; H, 5.41; N, 12.73; S, 14.58.
実測値 (¾) : C, 54.63; H, 5.31; N, 12.50; S, 14.56. Found (¾): C, 54.63; H, 5.31; N, 12.50; S, 14.56.
実施例 8-19 Example 8-19
N- {5, 6-ジメチル- 8-[(5-フルォロペンチル)スルファニリレ]- 3-ォキソ -2, 3-ジヒド ロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }イソニコチンアミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanylyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1- Ylidene} isonicotinamide
実施例 6-23で得られた卜ァミノ- 5, 6-ジメチル 8- [5-フルォロペンチル]スルフ ァニル ]-3H-イミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.317誦1) とィ ソニコチン酸 (43.4 mg, 0.349 mmol) の N, N-ジメチルホルムアミド (3.00 ml) 溶液に、 シァノりん酸ジェチル (0.0564 ml, 0.349 mmol), 続いてトリェチルァ ミン (0.0486 ml, 0.349匪 ol) を加えた。 反応混合物を室温で 15時間かき混ぜ た後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (8.7 mg, 6.5¾) を暗赤色結晶として得た。 酢酸 ェチル -ジェチルエーテルから再結晶して融点 203-205°Cの暗赤色結晶を得た。 IR (KBr) ソ : 2934, 2863, 1732, 1661, 1626, 1595, 1553 cm"1. Triamino-5,6-dimethyl 8- [5-fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-23 (100 mg, 0.317 recitation1) and isonicotinic acid (43.4 mg, 0.349 mmol) in N, N-dimethylformamide (3.00 ml) solution, getyl cyanophosphate (0.0564 ml, 0.349 mmol), and then triethylamine (0.0486 ml) , 0.349 bandits ol). After stirring the reaction mixture at room temperature for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (8.7 m g, 6.5¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 203-205 ° C. IR (KBr) S: 2934, 2863, 1732, 1661, 1626, 1595, 1553 cm " 1 .
¾-NMR (CDCI3) : ά 1.45-1.94 (6Η, m), 1.86 (3H, s), 2.37 (3H, s), 3.11 (2H, t, J=7.5 Hz), 4.48 (2H, dt, J=47.1, 5.7 Hz), 8.09 (2H, d, J=5.7 Hz), 8.75 (2H, d, J=6.0 Hz), 11.08 (1H, bs). 実施例 8-20 ¾-NMR (CDCI3): ά 1.45-1.94 (6Η, m), 1.86 (3H, s), 2.37 (3H, s), 3.11 (2H, t, J = 7.5 Hz), 4.48 (2H, dt, J = 47.1, 5.7 Hz), 8.09 (2H, d, J = 5.7 Hz), 8.75 (2H, d, J = 6.0 Hz), 11.08 (1H, bs). Example 8-20
N-{5, 6-ジメチル- 8- [ (5 -フルォ口ぺンチル)スルファニル] -3-ォキソ- 2, 3_ジヒド 口- 1H-イミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン卜 6_クロ口ニコチンアミド 実施例 6 - 23で得られた 1 -ァミノ- 5 , 6-ジメチル 8_ [5-フルォロぺンチル]スルフ ァニル ]_3H -イミダゾ [5,卜 c] [1,4]チアジン _3 -オン (100 mg, 0.317 mmol) と 6_ クロ口ニコチン酸(56. lmg, 0.349 mmol)の N, N-ジメチルホルムアミド(3.00ml) 溶液に、 シァノりん酸ジェチル (0.0564 ml, 0.349 mmol), 続いてトリェチルァ ミン (0.0486 ml, 0.349匪 ol) を加えた。 反応混合物を室温で 16時間かき混ぜ た後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (70.5 mg, 49¾) を暗赤色結晶として得た。 酢酸 ェチル-ジェチルエーテルから再結晶して融点 198'- 200°Cの暗赤色結晶を得た。 IR (KBr) V: 3285, 3063, 2940, 2863, 1738, 1601, 1582, 1559, 1539 cnf1. ¾-NMR (CDC13) : δ 1.48-1.68 (2Η, m), 1.68-1.92 (4H, m), 1.85 (3H, d, J=1.2 Hz), 2.36 (3H, d, J=0.9 Hz), 3.07 (2H, t, J-7.2 Hz), 4.48 (2H, dt, J=47.1, 6.0 Hz), 7.39 (1H, d, J=8.4 Hz), 8.42 (1H, dd, J=8.4, 2.4 Hz), 9.34 (1H, dd, J=2.4, 0.3 Hz), 11.04 (1H, bs). N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3_dihidid-1H-imidazo [5, tric] [1,4] thiazine- Trident 6_cloniconicotinamide 1-amino-5,6-dimethyl8_ [5-fluoropentyl] sulfanyl] _3H-imidazo [5, tric] [1, obtained in Example 6-23 4] To a solution of thiazin _3-one (100 mg, 0.317 mmol) and 6_ cyclonicotinic acid (56. lmg, 0.349 mmol) in N, N-dimethylformamide (3.00 ml) was added getyl cyanophosphate (0.0564 ml, 0.349 mmol). mmol), followed by triethylamine (0.0486 ml, 0.349 ol). After stirring the reaction mixture at room temperature for 16 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (70.5 mg, 49¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 198'-200 ° C. IR (KBr) V:. 3285 , 3063, 2940, 2863, 1738, 1601, 1582, 1559, 1539 cnf 1 ¾-NMR (CDC1 3): δ 1.48-1.68 (2Η, m), 1.68-1.92 (4H, m), 1.85 (3H, d, J = 1.2 Hz), 2.36 (3H, d, J = 0.9 Hz), 3.07 (2H, t, J-7.2 Hz), 4.48 (2H, dt, J = 47.1, 6.0 Hz), 7.39 (1H, d, J = 8.4 Hz), 8.42 (1H, dd, J = 8.4, 2.4 Hz), 9.34 (1H, dd, J = 2.4, 0.3 Hz), 11.04 (1H, bs).
元素分析値: C19H20N402S2C1F-0.5H20として Elemental analysis: as C 19 H 20 N 40 2 S 2 C1F-0.5H 20
計算値 (¾) : C, 49.19; H, 4.56; N, 12.08; S, 13.82. Calculated (¾): C, 49.19; H, 4.56; N, 12.08; S, 13.82.
実測値 (¾) : C, 49.20; H, 4.27; N, 11.74; S, 13.62. Found (¾): C, 49.20; H, 4.27; N, 11.74; S, 13.62.
実施例 8 - 21 Examples 8-21
N- {5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニリレ] -3-ォキソ -2, 3-ジヒド 口- 1H-ィミダゾ [5, 1-c] [1, ]チアジン-トイリデン } -6- (トリフルォロメチル)二 コチンアミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanylyl] -3-oxo-2,3-dihydrone-1H-imidazo [5,1-c] [1,] thiazine-toylidene}- 6- (trifluoromethyl) cotinamide
実施例 6- 23で得られた 1 アミノ -5, 6-ジメチル 8- [5-7ルォ口ぺンチル]スルフ ァニル ]- 3H -イミダゾ [5,1- c] [ 1 , 4]チアジン- 3 -オン( 100 mg, 0.317 mmol)と 6- (ト リフルォロメチル)ニコチン酸 (66.6 mg, 0.349 mmol) の N, N-ジメチルホルムァ ミド (3.00 ml) 溶液に、 シァノりん酸ジェチル (0.0564 ml, 0.349 mmol) 続い てトリエチルァミン (0.0486 ml, 0.349 mmol) を加えた。 反応混合物を室温で 18 時間かき混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (51.8mg, 33¾) を暗赤色結晶 として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 183-185°Cの暗赤 色結晶を得た。 1-Amino-5,6-dimethyl 8- [5-7-chloropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine-3 obtained in Example 6-23 To a solution of 1-one (100 mg, 0.317 mmol) and 6- (trifluoromethyl) nicotinic acid (66.6 mg, 0.349 mmol) in N, N-dimethylformamide (3.00 ml) was added getyl cyanophosphate (0.0564 ml, 0.349 mmol). mmol) Then, triethylamine (0.0486 ml, 0.349 mmol) was added. After stirring the reaction mixture at room temperature for 18 hours, water was added, and the mixture was extracted with ethyl acetate. Wash the extract with water and dry (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (51.8mg, 33¾) and the obtained as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 183-185 ° C.
IR (KBr) V: 3262, 2936, 1732, 1661, 1626, 1599, 1549 cm"1. IR (KBr) V: 3262, 2936, 1732, 1661, 1626, 1599, 1549 cm " 1 .
¾-NMR (CDC13) : δ 1.46-1.70 (2Η, m), 1.70 - 1,96 (4H, m), 1.87 (3H, s), 2.37 (3H, s), 3.09 (2H, t, J=7.5 Hz), 4.48 (2H; dt, J=47.1, 5.7 Hz), 7.76 (1H, d, J=8.1 Hz), 8.65 (1H, dd, J=7.5, 0.6 Hz), 9.65 (1H, d, J=0.6 Hz), 11.06 (1H, bs). ¾-NMR (CDC1 3): δ 1.46-1.70 (2Η, m), 1.70 - 1,96 (4H, m), 1.87 (3H, s), 2.37 (3H, s), 3.09 (2H, t, J = 7.5 Hz), 4.48 (2H; dt, J = 47.1, 5.7 Hz), 7.76 (1H, d, J = 8.1 Hz), 8.65 (1H, dd, J = 7.5, 0.6 Hz), 9.65 (1H, d , J = 0.6 Hz), 11.06 (1H, bs).
元素分析値: C2。H2。N402S2F4'0.5H20として Elemental analysis: C 2. H 2. N 4 0 2 S 2 F 4 '0.5H 2 0
計算値 (%) : C, 48.28; H, 4.25; N, 11.26; S, 12.89. Calculated (%): C, 48.28; H, 4.25; N, 11.26; S, 12.89.
実測値 (¾) : C, 48.59; H, 4.14; N, 11.39; S, 12.65. Found (¾): C, 48.59; H, 4.14; N, 11.39; S, 12.65.
実施例 8 - 22 Examples 8 to 22
N- {5, 6-ジメチル -8 - [ (5-フルォ口ぺンチル)スルファニル] - 3-ォキソ- 2, 3-ジヒド ロ- 1H-イミダゾ [5,l-c] [1, 4]チアジン- 1-ィリデン }- 6_(1H ピラゾ一ル-卜ィル) ニコチンアミド N- {5,6-dimethyl-8-[(5-fluoroopentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-1 -Ylidene}-6_ (1H pyrazolyl) nicotinamide
実施例 6- 23で得られた卜ァミノ- 5, 6-ジメチル 8 - [5-フルォロペンチル]スルフ ァニル ]-3H-イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (100 mg, 0.317 mmol) と 6- (1H-ピラゾール-卜ィル)ニコチン酸 (75.0 mg, 0.349 mmol) の N,N-ジメチル ホルムアミド (3.00ml) 溶液に、 シァノりん酸ジェチル(0.0564 ml, 0.349腿 ol) 続いてトリェチルァミン (0.0486 ml, 0.349讓 ol) を加えた。 反応混合物を室温 で 20時間かき混ぜた後、 水を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (20.6 nig, 13%) を暗赤色結晶 として得た。酢酸ェチル-ジェチルエーテルから再結晶して融点 203- 205 の暗赤 色結晶を得た。 Triamino-5,6-dimethyl 8- [5-fluoropentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one obtained in Example 6-23 (100 mg , 0.317 mmol) and 6- (1H-pyrazole-tolyl) nicotinic acid (75.0 mg, 0.349 mmol) in N, N-dimethylformamide (3.00 ml) were mixed with getyl cyanophosphate (0.0564 ml, 0.349 t ) Subsequently, triethylamine (0.0486 ml, 0.349 alcohol) was added. After the reaction mixture was stirred at room temperature for 20 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after solvent was obtained as a distilled to give the title compound (20.6 nig, 13%) dark red crystals under reduced pressure. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 203-205.
IR (KBr) V: 3270, 2924, 1740, 1603, 1591, 1545 cm—1. IR (KBr) V: 3270, 2924, 1740, 1603, 1591, 1545 cm— 1 .
^- M (CDCI3) : δ 1.42-1.98 (6Η, m), 1.86 (3H, d, J=1.0 Hz), 2.36 (3H, d, J=1.2 Hz), 3.11 (2H, t, J=7.6 Hz), 4.50 (2H, dt, J=47.2, 6.2 Hz), 6.50 (1H, dd, J=2.6, 1.6 Hz), 7.78 (1H, d, J=1.8Hz), 8.04 (1H, d, J=8.8 Hz), 8.58-8.67 (2H, m), 9.35 (1H, dd, J=2.2, 0.6 Hz), 11.09 (1H, bs). 元素分析値: C22H23N602S2F · 0.5H20として ^-M (CDCI3): δ 1.42-1.98 (6Η, m), 1.86 (3H, d, J = 1.0 Hz), 2.36 (3H, d, J = 1.2 Hz), 3.11 (2H, t, J = 7.6 Hz), 4.50 (2H, dt, J = 47.2, 6.2 Hz), 6.50 (1H, dd, J = 2.6, 1.6 Hz), 7.78 (1H, d, J = 1.8Hz), 8.04 (1H, d, J = 8.8 Hz), 8.58-8.67 (2H, m), 9.35 (1H, dd, J = 2.2, 0.6 Hz), 11.09 (1H, bs). Elementary analysis: as C 22 H 23 N 6 0 2 S 2 F · 0.5H 2 0
計算値 (¾) : C, 53.32; H, 4.88; N, 16.96; S, 12.94. Calculated (¾): C, 53.32; H, 4.88; N, 16.96; S, 12.94.
実測値 (¾) : C, 53.14; H, 4.60; N, 16.77; S, 13.25. Found (¾): C, 53.14; H, 4.60; N, 16.77; S, 13.25.
実施例 8 - 23 Examples 8-23
N- {5,6-ジメチル _8_[ (5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口- 1H-ィミダゾ [5, l-c] [1, 4]チアジン- 1-ィリデン }ァクリルアミド N- {5,6-dimethyl _8_ [(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrone-1H-imidazo [5, l-c] [1,4] thiazine-1-ylidene} acrylamide
水素化ナトリウム(20.3 mg, 0.507匪 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5, 6-ジメチル -8- [5-フルォロ ペンチル]スルファニル ]-3H -イミダゾ [5,卜 c] [1,4]チアジン- 3 -オン (80.0 mg, 0.254醒 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 3 -クロ口プロピオニル クロリ ド (0.0353 ml, 0.349醒 ol) を加え、 さらに 23時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (84.1mg, 81%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融 点 154- 156T:の暗赤色結晶を得た。 To a suspension of sodium hydride (20.3 mg, 0.507 marl ol) (60% oil) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8- [obtained in Example 6-23. Add a solution of 5-fluoropentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (80.0 mg, 0.254 mmol ol) in tetrahydrofuran (1.00 ml) and cool on ice for 5 minutes Stirred. To this was added 3-pilot propionyl chloride (0.0353 ml, 0.349 awake ol), and the mixture was further stirred for 23 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (84.1 mg, 81%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-getyl ether to give dark red crystals having a melting point of 154-156T :.
赤外吸収スペクトル(IR) (KBr中) リ: 3208, 2917, 1734, 1663, 1634, 1607, 1543 cm-1. Infrared absorption spectrum (IR) (in KBr): 3208, 2917, 1734, 1663, 1634, 1607, 1543 cm -1 .
一 NMR (CDC13) : δ 1.46-1.88 (6Η, m), 1.82 (3H, s), 2.32 (3H, s), 2.59 (3H, s), 3.02 (2H, t, J=7.2Hz), 4.46 (2H, dt, J=47.1, 5.7 Hz), 5.87 (1H, dd, J=9.9, 0.3 Hz), 6.34 (1H, dd, J=17.1, 10.5 Hz), 6.65 (1H, d, J=15.9 Hz), 10.90 (1H, bs). One NMR (CDC1 3): δ 1.46-1.88 (6Η, m), 1.82 (3H, s), 2.32 (3H, s), 2.59 (3H, s), 3.02 (2H, t, J = 7.2Hz), 4.46 (2H, dt, J = 47.1, 5.7 Hz), 5.87 (1H, dd, J = 9.9, 0.3 Hz), 6.34 (1H, dd, J = 17.1, 10.5 Hz), 6.65 (1H, d, J = 15.9 Hz), 10.90 (1H, bs).
元素分析値: C16H20N3O2S2F-0.1H20として Elemental analysis: C 16 H 20 N 3 O 2 S 2 F-0.1H 20
計算値 (%) : C, 51.76; H, 5.48; N, 11.32; S, 17.27. Calculated value (%): C, 51.76; H, 5.48; N, 11.32; S, 17.27.
実測値 (¾) : C, 51.95; H, 5.33; N, 11.29; S, 16.80. Found (¾): C, 51.95; H, 5.33; N, 11.29; S, 16.80.
実施例 8 - 24 Examples 8-24
N - {5, 6-ジメチル- 8-[(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5, l-c] [1,4]チアジン - 1 -ィリデン }-6-メチル -2-ピリジン力ルポ キサミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrone-1H-imidazo [5, lc] [1,4] thiazine-1-ylidene} -6-methyl-2-pyridine
実施例 6-23で得られた卜ァミノ _5, 6-ジメチル- 8 - [5-フルォロペンチル]スル ファニル] - 3H-イミダゾ [5,卜 c] [1, 4]チアジン - 3-オン (100 mg, 0.317 mmol) と 6 -メチル -2 -ピリジンカルボン酸 (66.5 mg, 0.476 mmol) の Ν,Ν-ジメチルホルム アミド (5.00 ml) 溶液に、 シァノりん酸ジェチル (0.0769 ml, 0.476 mmol) 続 いてトリェチルァミン (0.0663 ml, 0.476 mmol) を加えた。 反応混合物を室温で 15.5時間かき混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (120 mg, 87%) 'を暗赤色結晶 として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 160-162°Cの暗赤 色結晶を得た。 Triamino_5,6-dimethyl-8- [5-fluoropentyl] sulfur obtained in Example 6-23 Fanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (100 mg, 0.317 mmol) and 6-methyl-2-pyridinecarboxylic acid (66.5 mg, 0.476 mmol) Ν, Ν To a solution of -dimethylformamide (5.00 ml) was added getyl cyanophosphate (0.0769 ml, 0.476 mmol), followed by triethylamine (0.0663 ml, 0.476 mmol). After stirring the reaction mixture at room temperature for 15.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (120 mg, 87%) 'and the obtained as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 160-162 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3206, 2942, 2911, 2861, 1713, 1655, 1578, 1549, 1514 cm—1. Infrared absorption spectrum (IR) (in KBr): 3206, 2942, 2911, 2861, 1713, 1655, 1578, 1549, 1514 cm- 1 .
Ή— NMR (CDC13) : δ 1.52-1.92 (6H, m), 1.80 (3H, s), 2.38 (3H, d, J=0.6 Hz), Ή- NMR (CDC1 3): δ 1.52-1.92 (6H, m), 1.80 (3H, s), 2.38 (3H, d, J = 0.6 Hz),
2.59 (3H, s), 3.06 (2H, t, J=7.2 Hz), 4.35 (2H, dt, J=47.1, 5.7 Hz), 7.392.59 (3H, s), 3.06 (2H, t, J = 7.2 Hz), 4.35 (2H, dt, J = 47.1, 5.7 Hz), 7.39
(1H, d, J=7.8 Hz), 7.82 (1H, t, J=7.8 Hz), 8.14 (1H, d, J=7.5 Hz). (1H, d, J = 7.8 Hz), 7.82 (1H, t, J = 7.8 Hz), 8.14 (1H, d, J = 7.5 Hz).
元素分析値: C2QH23N402S2Fとして Elementary analysis: as C 2Q H 23 N 4 0 2 S 2 F
計算値 (¾) : C, 55.28; H, 5.33; N, 12.89; S, 14.76. Calculated (¾): C, 55.28; H, 5.33; N, 12.89; S, 14.76.
実測値 (%) : C, 55.42; H, 5.36; N, 12.65; S, 14.49. Found (%): C, 55.42; H, 5.36; N, 12.65; S, 14.49.
実施例 8 - 25 Example 8-25
3 -クロ口- N- {5, 6-ジメチル -8- [ (5-フルォ口ペンチル)スルファニル] -3-ォキソ -2, 3-ジヒド口- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデント 2, 2 -ジメチルプ 口パンアミド 3-chloro-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydric-1H-imidazo [5,1-c] [1 , 4] Thiazine-1-ylident 2,2-dimethylpropaneamide
水素化ナトリウム(38.0 mg, 0.951匪 ol) (60 油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた 1 -ァミノ- 5, 6 -ジメチル -8- [5-フルォロ ペンチル]スルファニル] -3H-ィミダゾ [5, 1 - c] [1, 4]チアジン- 3-ォン (150 mg, 0.476匪 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 3-クロロピバロィル クロリド (0.0766 ml, 0.571 匪 ol) を加え、 さらに 23時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(103 mg, 50%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 137- 139°Cの暗赤色結晶を得た。 赤外吸収スペクトル(IR) (KBr中) ν: 3247, 2967, 2940, 2870, 1750, 1613, 1599, 1537 cm"1. To a suspension of sodium hydride (38.0 mg, 0.951 ol) (60 oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8- [5] obtained in Example 6-23. -Fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine-3-one (150 mg, 0.476 marl) in tetrahydrofuran (1.00 ml) was added and the mixture was cooled under ice. Stir for a minute. To this was added 3-chloropivaloyl chloride (0.0766 ml, 0.571 ol ol) and stirred for an additional 23 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (103 mg, 50%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 137-139 ° C. Infrared absorption spectrum (IR) (in KBr) ν: 3247, 2967, 2940, 2870, 1750, 1613, 1599, 1537 cm " 1 .
Ή-NMR (CDC") : (51.32 (6H, s), 1.50-1.86 (6H, m), 1.81 (3H, d, J=0.9 Hz), 2.31 (3H, d, J=0.9 Hz), 3.01 (2H, t, J=7.5 Hz), 3.80 (2H, s), 4.46 (2H, dt, J=47.1, 6.0 Hz), 10.70 (1H, bs). Ή-NMR (CDC "): (51.32 (6H, s), 1.50-1.86 (6H, m), 1.81 (3H, d, J = 0.9 Hz), 2.31 (3H, d, J = 0.9 Hz), 3.01 (2H, t, J = 7.5 Hz), 3.80 (2H, s), 4.46 (2H, dt, J = 47.1, 6.0 Hz), 10.70 (1H, bs).
元素分析値: C^H^NAS^IFとして Elemental analysis: as C ^ H ^ NAS ^ IF
計算値 (%) : C, 49.81; H, 5.81; , 9.68; S, 14.78. Calculated (%): C, 49.81; H, 5.81;, 9.68; S, 14.78.
実測値 (%) : C, 49.70; H, 5.71; , 9.70; S, 14.67. Found (%): C, 49.70; H, 5.71;, 9.70; S, 14.67.
実施例 8-26 Example 8-26
N - {5, 6-ジメチル- 8-[(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5, 1- c] [1, 4]チアジン- 1-イリデン } - 3 -メチル 2-ピリジン力ルポ キサミド N- {5, 6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine-1- Ilidene} -3-Methyl 2-pyridine lipoxamide
実施例 6-23で得られた 1-ァミノ- 5, 6 -ジメチル- 8-[5-フルォロペンチル]スル ファニル] - 3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (100 mg, 0.317 mmol) と 3-メチル _2_ピリジンカルポン酸 (68.6 mg, 0.476 mmol) の N,N-ジメチルホルム アミド (5.00 ml) 溶液に、 シァノりん酸ジェチル (0.0769 ml, 0.476 mmol) 続 いてトリェチルァミン (0.0663 ml, 0.476 mmol) を加えた。 反応混合物を室温で 15.5時間かき混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (106 mg, 77%) を暗赤色結晶 として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 148-150 の暗赤 色結晶を得た。 1-Amino-5,6-dimethyl-8- [5-fluoropentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one obtained in Example 6-23 ( To a solution of 100 mg, 0.317 mmol) and 3-methyl_2_pyridinecarbonic acid (68.6 mg, 0.476 mmol) in N, N-dimethylformamide (5.00 ml) was added getyl cyanophosphate (0.0769 ml, 0.476 mmol). Then, triethylamine (0.0663 ml, 0.476 mmol) was added. After stirring the reaction mixture at room temperature for 15.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was obtained as a dark red crystalline title compound was distilled off (106 mg, 77%) under reduced pressure. The crystals were recrystallized from ethyl acetate-ethyl ether to give dark red crystals having a melting point of 148-150.
赤外吸収スペクトル(IR) (KBr中) V 3196, 2934, 2861, 1715, 1651, 1549, 1505 cm"1. Infrared absorption spectrum (IR) (in KBr) V 3196, 2934, 2861, 1715, 1651, 1549, 1505 cm " 1 .
Ή-NMR (CDC13) : δ 1.50-1.90 (6L m), 1.79 (3H, s), 2.37 (3H, d, J=1.2 Hz), 2.82 (3H, s), 3.06 (2H, t, 1=1.2 Hz), 4.38 (2H, dt, J-47.7, 5.7 Hz), 7.43 (1H, dd, J-7.5, 4.5 Hz), 7.70 (1H, d, J=8.4 Hz), 8.44 (1H, d, J=3.9 Hz). 元素分析値: C2。H23N402S2Fとして Ή-NMR (CDC1 3): δ 1.50-1.90 (6L m), 1.79 (3H, s), 2.37 (3H, d, J = 1.2 Hz), 2.82 (3H, s), 3.06 (2H, t, 1 = 1.2 Hz), 4.38 (2H, dt, J-47.7, 5.7 Hz), 7.43 (1H, dd, J-7.5, 4.5 Hz), 7.70 (1H, d, J = 8.4 Hz), 8.44 (1H, d , J = 3.9 Hz) elemental analysis:. C 2. H 23 N 4 0 2 S 2 F
計算値 (¾) : C, 55.28; H, 5.33; N, 12.89; S, 14.76. Calculated (¾): C, 55.28; H, 5.33; N, 12.89; S, 14.76.
実測値 (%) : C, 55.12; H, 5.42; N, 12.86; S, 14.49. 実施例 8-27 Found (%): C, 55.12; H, 5.42; N, 12.86; S, 14.49. Example 8-27
4 -クロ口 -N- {5, 6-ジメチル- 8 - [ (5-フルォロペンチル)スルファニル] -3-ォキソ - 2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン ピリジンカル ポキサミド 4-Cross-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] Thiazine-1-ylidene pyridinecarboxamide
実施例 6-23で得られた 1 -ァミノ- 5, 6-ジメチル- 8- [5-フルォロペンチル]スル ファニル]- 3H-イ ミダゾ [5, 1-c] [1,4]チアジン- 3-オン (100 mg, 0.317 匪 ol) と 4-クロ口- 2-ピリジンカルポン酸 (77.2 mg, 0.476 mmol) の Ν,Ν-ジメチルホルム アミド (5.00 ml) 溶液に、 シァノりん酸ジェチル (0.0769 ml, 0.476 mmol)、 続 いてトリェチルァミン (0.0663 ml, 0.476匪 ol) を加えた。 反応混合物を室温で 21 時間かき混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (56.4mg, 39%) を二種異性体 混合物として得た。 1-Amino-5,6-dimethyl-8- [5-fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine-3- obtained in Example 6-23 To a solution of ON (100 mg, 0.317 marl ol) and 4-chloro-2-pyridinepyridine (77.2 mg, 0.476 mmol) in Ν, Ν-dimethylformamide (5.00 ml) was added getyl cyanophosphate (0.0769 ml). , 0.476 mmol) followed by triethylamine (0.0663 ml, 0.476 bandol). After stirring the reaction mixture at room temperature for 21 hours, water was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, a dry (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (56.4 mg, 39%) of two or isomer mixture.
赤外吸収スペクトル(IR) (KBr中) V: 2940, 2867, 1732, 1597, 1549, 1514 cm1.Infrared absorption spectrum (IR) (in KBr) V: 2940, 2867, 1732, 1597, 1549, 1514 cm 1 .
Ή—NMR (CDC13) : δ 1.50-1.94 (6Η, m), 1.80 (3HX0.57, s), 1.86 (3HX0.43, s), 2.38 (3H, s), 3.07 (2HX0.57, t, J=7.5 Hz), 3.13 (2HX0.43, t, J=7.8Hz), Ή-NMR (CDC1 3): δ 1.50-1.94 (6Η, m), 1.80 (3HX0.57, s), 1.86 (3HX0.43, s), 2.38 (3H, s), 3.07 (2HX0.57, t , J = 7.5 Hz), 3.13 (2HX0.43, t, J = 7.8Hz),
4.39 (2HX0.57, dt, J-46.8, 5.7 Hz), 4.48 (2HX0.43, dt, J=47.1, 6.3 Hz),4.39 (2HX0.57, dt, J-46.8, 5.7 Hz), 4.48 (2HX0.43, dt, J = 47.1, 6.3 Hz),
7.41 (1HX0.43, dd, J=5.1, 2.1 Hz), 7.54 (1HX0.57, dd, J=5.4, 2.1 Hz), 8.327.41 (1HX0.43, dd, J = 5.1, 2.1 Hz), 7.54 (1HX0.57, dd, J = 5.4, 2.1 Hz), 8.32
(1HX0.57, t, J=2.1 Hz), 8.50 (1HX0.57, t, J=5.1 Hz), 8.57 (1HX0.43, d,(1HX0.57, t, J = 2.1 Hz), 8.50 (1HX0.57, t, J = 5.1 Hz), 8.57 (1HX0.43, d,
J=2.1 Hz), 8.68 (1HX0.43, d, J=5.4 Hz), 11.12 (1HX0.43, bs), 12.38 (1H X0.57, bs). J = 2.1 Hz), 8.68 (1HX0.43, d, J = 5.4 Hz), 11.12 (1HX0.43, bs), 12.38 (1H X0.57, bs).
元素分析値: C19H2QN402S2C1Fとして Elemental analysis: C 19 H 2 Q N 4 0 2 S 2 C1F
計算値 (%) : C, 50.16; H, 4.43; N, 12.31; S, 14.10. Calculated value (%): C, 50.16; H, 4.43; N, 12.31; S, 14.10.
実測値 (%) : C, 50.24; H, 4.30; N, 12.36; S, 13.87. Found (%): C, 50.24; H, 4.30; N, 12.36; S, 13.87.
実施例 8-28 Example 8-28
6_クロ口- N-{5,6 -ジメチル -8- [(5 -フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 1 -ィリデン } - 2-ピリジンカル ポキサミド 6_Black-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1, 4] Thiazine-1 -ylidene}-2-pyridinecarboxamide
実施例 6-23で得られた 1-ァミノ- 5, 6 -ジメチル -8- [5-フルォロペンチル]スル ファニル]- 3H -イミダゾ [5, 1-c] [1, 4]チアジン- 3 -オン (80.0 mg, 0.254 mmol) と 6 -クロ口- 2-ピリジンカルポン酸 (59.9 mg, 0.380醒 ol) の N,N-ジメチルホルム アミ ド (4.00 ml) 溶液に、 シァノりん酸ジェチル (0.0615 ml, 0.380扁 ol) 続 いてトリェチルァミン (0.0530 ml, 0.380腿 ol) を加えた。 反応混合物を室温で 21 時間かき混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (64.7mg, 56%) を暗赤色結晶 として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 198- 200°Cの暗赤 色結晶を得た。 1-Amino-5,6-dimethyl-8- [5-fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one obtained in Example 6-23 (80.0 mg, 0.254 mmol) and 6-Chloro-2-pyridinepyridine (59.9 mg, 0.380 ol) in N, N-dimethylformamide (4.00 ml), getyl cyanophosphate (0.0615 ml, 0.380 ol), and then triethylamine (0.0530 ml, 0.380 t). After stirring the reaction mixture at room temperature for 21 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (64.7 mg, 56%) was obtained as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 198-200 ° C.
赤外吸収スペクトル(IR) (KBr中) V: 3204, 3127, 2938, 2865, 1717, 1653, 1624, 1595, 1545, 1516 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3204, 3127, 2938, 2865, 1717, 1653, 1624, 1595, 1545, 1516 cm " 1 .
Ή-NMR (CDC13) : δ 1.48-1.96 (6Η, m), 1.80 (3H, s), 2.38 (3H, s), 3.09 (2H, t, J=6.9 Hz), 4.39 (2H, dt, J=46.8, 5.1 Hz), 7.58 (1H, d, J=7.8 Hz), 7.92 Ή-NMR (CDC1 3): δ 1.48-1.96 (6Η, m), 1.80 (3H, s), 2.38 (3H, s), 3.09 (2H, t, J = 6.9 Hz), 4.39 (2H, dt, J = 46.8, 5.1 Hz), 7.58 (1H, d, J = 7.8 Hz), 7.92
(1H, t, J=7. Hz), 8.26 (1H, d, J=4.8 Hz), 12.40 (1H, bs). (1H, t, J = 7 Hz), 8.26 (1H, d, J = 4.8 Hz), 12.40 (1H, bs).
元素分析値: C19 。N402S2C1Fとして Elemental analysis: C 19. N 4 0 2 S 2 C1F
計算値 (%) : C, 50.16; H, 4.43; N, 12.31; S, 14.10. Calculated value (%): C, 50.16; H, 4.43; N, 12.31; S, 14.10.
実測値 (%) : C, 50.30; H, 4.34; N, 12.53; S, 14.02. Found (%): C, 50.30; H, 4.34; N, 12.53; S, 14.02.
実施例 8-29 Example 8-29
N - {5, 6-ジメチル- 8-[(5-フルォロペンチル)スルファニル ]-3 -ォキソ -2, 3 -ジヒド ロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデン卜卜イソキノリンカルポキサ ミド、 N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene Isoquinoline carboxamide,
実施例 6-23で得られた 1-ァミノ- 5, 6 -ジメチル -8 - [5-フルォロペンチル]スルフ ァニル ]_3H-イミダゾ [5, l_c] [1,4]チアジン- 3-オン (80.0 mg, 0.254誦 ol) と 卜イソキノリンカルボン酸 (66.5 mg, 0.380讓 ol) の Ν,Ν -ジメチルホルムアミ ド (4.00 ml) 溶液に、 シァノりん酸ジェチル (0.0615 ral, 0.380 mmol) 続いて トリェチルァミン (0.0530 ml, 0.380 mmol) を加えた。 反応混合物を室温で 22.5 時間かき混ぜた後、水を加えて酢酸ェテルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (19.0 mg, 16¾) を暗赤色結晶として得 た。酢酸ェチル-ジェチルエーテルから再結晶して融点 198- 200°Cの暗赤色結晶を 得た。 1-Amino-5,6-dimethyl-8- [5-fluoropentyl] sulfanyl] _3H-imidazo [5, l_c] [1,4] thiazin-3-one obtained in Example 6-23 (80.0 mg , 0.254 recitation ol) and triisoquinoline carboxylic acid (66.5 mg, 0.380 SUBSTITLE) in Ν, Ν-dimethylformamide (4.00 ml) solution, getyl cyanophosphate (0.0615 ral, 0.380 mmol), followed by triethylamine (0.0530 ml, 0.380 mmol). After stirring the reaction mixture at room temperature for 22.5 hours, water was added and extracted with ether acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (19.0 mg, 16¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 198-200 ° C.
赤外吸収スペクトル(IR) (KBr中) リ: 3169, 2940, 1709, 1653, 1580, 1549, 1507 CUT1. Infrared absorption spectrum (IR) (in KBr): 3169, 2940, 1709, 1653, 1580, 1549, 1507 CUT 1 .
ΐ-NMR (CDClg) : δ 1.50-1.98 (6H, m) , 1.80 (3H, s), 2.39 (3H, d, J=0.8 Hz), 3.10 (2H, t,, J=7.0 Hz), 4.37 (2H, dt, J=47.2, 5.8 Hz), 7.70-7.84 (2H, m), 7.84-7.97 (2H, m), 8.51 (1H, d, J=5.4 Hz), 9.70 (1H, dd, J-6.8, 3.4 Hz). 元素分析値: C23H23N402S2Fとして ΐ-NMR (CDClg): δ 1.50-1.98 (6H, m), 1.80 (3H, s), 2.39 (3H, d, J = 0.8 Hz), 3.10 (2H, t ,, J = 7.0 Hz), 4.37 (2H, dt, J = 47.2, 5.8 Hz), 7.70-7.84 (2H, m), 7.84-7.97 (2H, m), 8.51 (1H, d, J = 5.4 Hz), 9.70 (1H, dd, J -6.8, 3.4 Hz). Elemental analysis: C 23 H 23 N 40 2 S 2 F
計算値 (%) : C, 58.70; H, 4.93; N, 11.91; S, 13.63. Calculated (%): C, 58.70; H, 4.93; N, 11.91; S, 13.63.
実測値 (%) : C, 58.74; H, 4.90; N, 12.09; S, 13.56. Found (%): C, 58.74; H, 4.90; N, 12.09; S, 13.56.
実施例 8-30 Example 8-30
N-{5, 6-ジメチル- 8- [ (5 -フルォ口ぺンチル)スルファニル] -3-ォキソ -2, 3 -ジヒド 口- 1H-イミダゾ [5,卜 c] [1, 4]チアジン-卜イリデン }- 2-フラミド N- {5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihidido-1H-imidazo [5, tric] [1,4] thiazine- Triylidene}-2-furamide
水素化ナトリウム(20.3 mg, 0.507 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ- 5, 6 -ジメチル -8 - [5 -フルォロ ペンチル]スルファニル ]- 3H-ィミダゾ [5, 1-c] [1, ]チアジン- 3-オン (80.0 mg, 0.254匪 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 2-フロイル クロリド (0.0279 mg, 0.279匪 ol) のテトラヒドロフ ラン溶液を加え、 さらに 30分間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸 ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題 記化合物 (84. ling, 81¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 199-201 °Cの暗赤色結晶を得た。 To a suspension of sodium hydride (20.3 mg, 0.507 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added the triamino-5,6-dimethyl-8- [5-] obtained in Example 6-23. Add a solution of fluoropentyl] sulfanyl] -3H-imidazo [5,1-c] [1,] thiazin-3-one (80.0 mg, 0.254 bandol) in tetrahydrofuran (1.00 ml), and stir under ice-cooling for 5 minutes. I did. To this was added a solution of 2-furoyl chloride (0.0279 mg, 0.279 marl) in tetrahydrofuran, and the mixture was further stirred for 30 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (84. ling, 81¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 199-201 ° C.
赤外吸収スペクトル(IR) (KBr中) ソ : 3239, 3119, 2961, 2944, 2868, 1713, 1663, 1628, 1607, 1576, 1541 cm—1. Infrared absorption spectrum (IR) (in KBr) So: 3239, 3119, 2961, 2944, 2868, 1713, 1663, 1628, 1607, 1576, 1541 cm- 1 .
Ή—NMR (CDCI3) : δ 1.56-1.90 (6Η, m), 1.81 (3H, s), 2.33 (3H, d, J=0.9 Hz), 3.06 (2H, t, J=7.5 Hz), 4.48 (2H, dt, J=47.1, 6.0 Hz), 6.51 (1H, dd, J=3.6, 1.8 Hz), 7.34 (1H, d, J=3.3 Hz), 7.61 (1H, d, J=0.9 Hz), 10.88 (1H, bs). 元素分析値: C18H2。N303S2Fとして Ή—NMR (CDCI3): δ 1.56-1.90 (6Η, m), 1.81 (3H, s), 2.33 (3H, d, J = 0.9 Hz), 3.06 (2H, t, J = 7.5 Hz), 4.48 ( 2H, dt, J = 47.1, 6.0 Hz), 6.51 (1H, dd, J = 3.6, 1.8 Hz), 7.34 (1H, d, J = 3.3 Hz), 7.61 (1H, d, J = 0.9 Hz), 10.88 (1H, bs) elemental analysis:. C 18 H 2. N 3 0 3 S 2 F
計算値 (¾) : C, 52.79; H, 4.92; N, 10.26; S, 15.66. Calculated (¾): C, 52.79; H, 4.92; N, 10.26; S, 15.66.
実測値 (%) : C, 52.76; H, 4.88; N, 10.14; S, 15.42. Found (%): C, 52.76; H, 4.88; N, 10.14; S, 15.42.
実施例 8 - 31 Example 8-31
N-{5, 6 -ジメチル- 8 - [ (5-フルォロペンチル)スルファニル] - 3 -ォキソ -2, 3-ジヒド 口 - m-イミダゾ [5, .1 -c] [ 1 , 4]チアジン-卜イリデン卜 2 -チォフェンカルボキサミ ド . N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydride Mouth-m-imidazo [5, .1 -c] [1,4] thiazine-triylidene 2-thiophenecarboxamide.
水素化ナトリウム (20.3 mg, 0.507 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5, 6-ジメチル- 8- [5-フルォロ ペンチル]スルファニル ]-3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3-オン (80.0 mg, 0.254 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに 2-テノィル クロリド (0.0299 mg, 0.279腿 ol) のテトラヒドロフ ラン溶液を加え、 さらに 1.5時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸 ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題 記化合物 (80.6mg, 75¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テ ルから再結晶して融点 193- 195°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (20.3 mg, 0.507 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8- [5] obtained in Example 6-23. -Fluoropentyl] sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (80.0 mg, 0.254 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was stirred under ice cooling for 5 minutes. I did. To this was added a solution of 2-thenoyl chloride (0.0299 mg, 0.279 t ol) in tetrahydrofuran, and the mixture was further stirred for 1.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (80.6mg, 75¾) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 193-195 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3229, 3083, 2965, 2938, 2915, 2863, 1736, 1661, 1609, 1597, 1553, 1520 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3229, 3083, 2965, 2938, 2915, 2863, 1736, 1661, 1609, 1597, 1553, 1520 cm " 1 .
Ή-NMR (CDC13) : δ 1.54-1.90 (6Η, m), 1.83 (3H, d, J=0.9 Hz), 2.33 (3H, d, J=0.9 Hz), 3.07 (2H, t, J=7.5 Hz), 4.48 (2H, dt, J-47.1, 5.4 Hz), 7.11 (1H, dd, J=4.8, 3.9 Hz), 7.54 (1H, dd, J=4.8, 0.9 Hz), 7.91 (1H, dd, J=3.6, 1.2 Hz), 10.91 (1H, bs). Ή-NMR (CDC1 3): δ 1.54-1.90 (6Η, m), 1.83 (3H, d, J = 0.9 Hz), 2.33 (3H, d, J = 0.9 Hz), 3.07 (2H, t, J = 7.5 Hz), 4.48 (2H, dt, J-47.1, 5.4 Hz), 7.11 (1H, dd, J = 4.8, 3.9 Hz), 7.54 (1H, dd, J = 4.8, 0.9 Hz), 7.91 (1H, dd, J = 3.6, 1.2 Hz), 10.91 (1H, bs).
元素分析値: C18H2。N302S3Fとして Elemental analysis: C 18 H 2. N 3 0 2 S 3 F
計算値 (%) : C, 50.80; H, 4.74; N, 9.87; S, 22.60. Calculated value (%): C, 50.80; H, 4.74; N, 9.87; S, 22.60.
実測値 (¾) : C, 50.77; H, 4.74; N, 9.78; S, 22.62. Found (¾): C, 50.77; H, 4.74; N, 9.78; S, 22.62.
実施例 9 - 1 Example 9-1
式 (XX I ) において R 2が 2- (エトキシカルポニル)ェチル基、 R5が n-ブチル 基、 R8および R9のどちらか一方が n-ブチル基、 他方が水素原子、 Zが S02、 R 15が P-トルィル基である化合物の合成 In the formula (XXI), R 2 is a 2- (ethoxycarbonyl) ethyl group, R 5 is an n-butyl group, one of R 8 and R 9 is an n-butyl group, the other is a hydrogen atom, and Z is S 0 2 Of compounds in which R 15 is a P-tolyl group
実施例 1-14で合成された化合物 (305 Big, 0.510 oifflol) をトルエン (10 ml) および無水酢酸 (0.05 ml, 0.51 mmol) に溶解し、 D -力ンファ一- 10-スルホン酸 (119mg, 0.510讓 ol)を加えて 2時間加熱還流した。反応液を酢酸ェチル(20 ml) で希釈し、 水、 2%炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 減圧下濃縮乾固した。 残渣をメタノールに溶解し、 逆 相系分取 HPLC (担体: 0DS、 YMC- Pack、 S- 363- 1- 15、 内径 30 mm, 長さ 250 mm, ワイ 'ェム 'シィ社製、 移動相: 0.05(v/v)%トリフルォロ酢酸 Z77〜90(v/v)%7 セトニトリル水) に付し、 溶出容量 1060〜1180ml の画分を集めて濃縮後、 残水 層を酢酸ェチル (40 ml) で抽出した。 有機層を 0.05N塩酸、 水および 和食塩水 で洗浄し、 硫酸ナトリウムで乾燥後、 減圧下濃縮乾固した。 残渣にエーテルを加 え、 生成した沈澱物を濾取し、 エーテルで洗浄後、 乾燥して標題化合物の黄色粉 末 (176 mg、 0.304 mmol) を得た。 The compound (305 Big, 0.510 oifflol) synthesized in Example 1-14 was dissolved in toluene (10 ml) and acetic anhydride (0.05 ml, 0.51 mmol), and D-potassin-1-10-sulfonic acid (119 mg, (0.510 mL) was added and the mixture was heated under reflux for 2 hours. The reaction solution was diluted with ethyl acetate (20 ml), washed with water, a 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dried over sodium sulfate, and concentrated to dryness under reduced pressure. The residue is dissolved in methanol and subjected to reverse phase preparative HPLC (Carrier: 0DS, YMC-Pack, S-363-1-15, ID 30 mm, length 250 mm, manufactured by Y'M'C, mobile phase : Elute with 0.05 (v / v)% trifluoroacetic acid Z77 ~ 90 (v / v)% 7 acetonitrile water, collect fractions with elution volume of 1060 ~ 1180ml, concentrate and concentrate the remaining aqueous layer to ethyl acetate (40ml) ). The organic layer was washed with 0.05N hydrochloric acid, water and a saline solution, dried over sodium sulfate, and concentrated to dryness under reduced pressure. Ether was added to the residue, and the resulting precipitate was collected by filtration, washed with ether, and dried to give a yellow powder of the title compound (176 mg, 0.304 mmol).
-腿 (300 MHz, 重クロ口ホルム中): δ 0.89 (3Η, t, J= 7.2 Hz), 0.91 (3H, t, J= 7.2 Hz), 1.26 (3H, t, J= 7.2 Hz), 1.33 (4H, m), 1.41 (4H, m), 1.63 (2H, m), 2.17 (2H, brquint, J= 7.3 Hz), 2.42 (3H, s), 2.66 (2H, i, J= 7.5 Hz), 3.22 (2H, di, J二 3.4, 7.5 Hz), 4.15 (2H, q, J= 7.2 Hz), 4.03 (1H, t, J= 7.9 Hz), 5.92 (1H, t, J= 7.9 Hz), 7.02 (1H, s), 7.30 (2H, brd, J- 8.3 Hz), 7.88 (2H, d, J= 8.3 Hz), 9.63 (1H, br). -Thigh (300 MHz, in double-mouthed holm): δ 0.89 (3Η, t, J = 7.2 Hz), 0.91 (3H, t, J = 7.2 Hz), 1.26 (3H, t, J = 7.2 Hz), 1.33 (4H, m), 1.41 (4H, m), 1.63 (2H, m), 2.17 (2H, brquint, J = 7.3 Hz), 2.42 (3H, s), 2.66 (2H, i, J = 7.5 Hz ), 3.22 (2H, di, J2 3.4, 7.5 Hz), 4.15 (2H, q, J = 7.2 Hz), 4.03 (1H, t, J = 7.9 Hz), 5.92 (1H, t, J = 7.9 Hz) ), 7.02 (1H, s), 7.30 (2H, brd, J- 8.3 Hz), 7.88 (2H, d, J = 8.3 Hz), 9.63 (1H, br).
元素分析値: C27H37N305S3 として計算値: C, 55.93; H, 6.43; N, 7.25; S, 16.59 (%), 実測値; C, 56.19; H, 6.40; N, 7.13; S, 16.80 (%) . Elemental analysis: C 27 H 37 N 3 0 5 S 3 Calculated: C, 55.93; H, 6.43 ; N, 7.25; S, 16.59 (%), Found; C, 56.19; H, 6.40 ; N, 7.13; S, 16.80 (%).
また上記逆相系分取 HPLCにおいて溶出容量 1360〜U00ml の画分を集めて同 様に処理し、 実施例 2-19で合成される化合物の赤色粉末 (18 mg、 0.030 mmol) を得た。 Further, fractions having an elution volume of 1360 to U00 ml were collected in the reverse phase preparative HPLC and treated in the same manner to obtain a red powder (18 mg, 0.030 mmol) of the compound synthesized in Example 2-19.
以下に実施例 9-1で合成した化合物および同様にして合成した化合物の一覧を 示す。 〔表 94〕The following is a list of compounds synthesized in Example 9-1 and compounds synthesized in the same manner. (Table 94)
差替え用紙 ( M ; 〔表 9 5〕Replacement paper (M; (Table 95)
差替え用紙(規則 2β) 〔表 96〕Replacement form (Rule 2β) (Table 96)
ぇ用弒 m ) 以下に実施例 9- 2〜9- 27で合成した化合物のデータを示す。 ぇ m) The data of the compounds synthesized in Examples 9-2 to 9-27 are shown below.
実施例 9-2 Example 9-2
^-NM (300MHz, CDC13) : δ 0.86 (3Η, t, J= 6.8 Hz), 1.23 (3H, t, J= 7.1 Hz), 1.26 (2H, brs), 1.30 (2H, m), 1.37 (2H, m), 1.64 (2H, m), 2.38 (3H, s), 2.64 (2H, t, J= 7.3 Hz), 3.19 (2H, m), 3.51 (1H, brt, J= 7.3 Hz), 4.13 (2H, q, J= 7.1 Hz), 4.83 (1H, brs), 5.46 (1H, brs), 7.23 (2H, brd, J= 8.2 Hz), 7.88 (2H, d, J= 8.2 Hz). ^ -NM (300MHz, CDC1 3) : δ 0.86 (3Η, t, J = 6.8 Hz), 1.23 (3H, t, J = 7.1 Hz), 1.26 (2H, brs), 1.30 (2H, m), 1.37 (2H, m), 1.64 (2H, m), 2.38 (3H, s), 2.64 (2H, t, J = 7.3 Hz), 3.19 (2H, m), 3.51 (1H, brt, J = 7.3 Hz) , 4.13 (2H, q, J = 7.1 Hz), 4.83 (1H, brs), 5.46 (1H, brs), 7.23 (2H, brd, J = 8.2 Hz), 7.88 (2H, d, J = 8.2 Hz) .
元素分析値: C24H31N305S3 · 0.6 0として計算値: C, 52.55; H, 5.92; N, 7.66; S, 17.53 (%) .実測値: C, 52.44; H, 5.67; N, 7.63; S, 17.46 (%) . Elemental analysis: C 24 H 31 N 3 0 5 S 3 · 0.60 Calculated: C, 52.55; H, 5.92; N, 7.66; S, 17.53 (%). Found: C, 52.44; H, 5.67 ; N, 7.63; S, 17.46 (%).
実施例 9-3 Example 9-3
'H-NMR (300MHz, CDC13) : <5 0.89 (3H, t, J= 6.9 Hz), 1.26 (3H, t, J= 7.1 Hz), 1.31 (2H, m), 1.31 (2H, m) , 1.45 (2H, brquint, J= 7.0 Hz), 2.18 (2H, brq, J= 7.5 Hz), 2.42 (3H, s), 2.67 (2H, t, J- 7.4 Hz), 3.24 (2H, t, J= 7.3 Hz), 3.60 (2H, s), 4.15 (2H, q, J= 7.1. Hz), 6.08 (1H, t, J= 7.9 Hz), 7.30 (2H, brd, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.68 (1H, brs). , 元素分析値: C24H31N305S3として計算値: C, 53.61; H, 5.81; N, 7.81; S, 17.89'H-NMR (300MHz, CDC1 3): <5 0.89 (3H, t, J = 6.9 Hz), 1.26 (3H, t, J = 7.1 Hz), 1.31 (2H, m), 1.31 (2H, m) , 1.45 (2H, brquint, J = 7.0 Hz), 2.18 (2H, brq, J = 7.5 Hz), 2.42 (3H, s), 2.67 (2H, t, J-7.4 Hz), 3.24 (2H, t, J = 7.3 Hz), 3.60 (2H, s), 4.15 (2H, q, J = 7.1.Hz), 6.08 (1H, t, J = 7.9 Hz), 7.30 (2H, brd, J = 8.3 Hz), . 7.87 (2H, d, J = 8.3 Hz), 9.68 (1H, brs), elemental analysis: calculated C 24 H 31 N 3 0 5 S 3: C, 53.61; H, 5.81; N, 7.81; S, 17.89
(%) .実測値: C, 53.46; H, 5.53; N, 7.83; S, 18.10 (%) . Found: C, 53.46; H, 5.53; N, 7.83; S, 18.10 (%).
実施例 9-4 Example 9-4
¾-匪 R (300MHz, CDC ) : δ 0.87 (3H, t, J= 7.2 Hz), 1.25 (3H, t, J= 7.2 Hz), 1.42 (2H, m), 1.72 (2H, m), 2.43 (3H, s), 2.66 (2H, dt, J= 6.8, 7.9 Hz), 3.23 (2H, dt, J= 2.3, 7.5 Hz), 4.15 (2H, q, J= 7.2 Hz), 4.31 (1H, dd, J- 8.7, 6.8 Hz), 7.02 (1H, s), 7.21 (2H, d, J= 8.3 Hz), 7.31 (1H, m), 7.32 (2H, brd, J= 8.3 Hz), 7.38 (2H, brt, J= 7.5 Hz), 7.90 (2H, d, J- 8.3 Hz), 9.73 (1H, br). 元素分析値: C28H31N305S3として計算値: C, 57.41; H, 5.33; , 7.17; S, 16.42 (%) .実測値: C, 57.31; H, 5.18; , 7.17; S, 16.46 (%) . 匪 -band R (300MHz, CDC): δ 0.87 (3H, t, J = 7.2 Hz), 1.25 (3H, t, J = 7.2 Hz), 1.42 (2H, m), 1.72 (2H, m), 2.43 (3H, s), 2.66 (2H, dt, J = 6.8, 7.9 Hz), 3.23 (2H, dt, J = 2.3, 7.5 Hz), 4.15 (2H, q, J = 7.2 Hz), 4.31 (1H, dd, J- 8.7, 6.8 Hz), 7.02 (1H, s), 7.21 (2H, d, J = 8.3 Hz), 7.31 (1H, m), 7.32 (2H, brd, J = 8.3 Hz), 7.38 ( 2H, brt, J = 7.5 Hz ), 7.90 (2H, d, J- 8.3 Hz), 9.73 (1H, br) elemental analysis:. C 28 H 31 N 3 0 5 S 3 calculated: C, 57.41 H, 5.33;, 7.17; S, 16.42 (%). Found: C, 57.31; H, 5.18;, 7.17; S, 16.46 (%).
実施例 9-5 Example 9-5
Ή-NMR (300MHz, CDC13) : δ 1.27 (3H, t, J= 7.2 Hz), 2.40 (3H, s), 4.16 (2H, q, J= 7.2 Hz), 4.21 (2H, s), 4.35(2H, s), 6.57 (1H, s), 7.23 (2H, d, J= 8.3 Hz), 7.28 (5H, s), 7.89 (2H, d, J= 8.3 Hz). 実施例 9-6 Ή-NMR (300MHz, CDC1 3 ): δ 1.27 (3H, t, J = 7.2 Hz), 2.40 (3H, s), 4.16 (2H, q, J = 7.2 Hz), 4.21 (2H, s), 4.35 (2H, s), 6.57 (1H, s), 7.23 (2H, d, J = 8.3 Hz), 7.28 (5H, s), 7.89 (2H, d, J = 8.3 Hz). Example 9-6
Ή-NMR (300MHz, CDC13) : δ 0.95 (3H, t, J= 7.2 Hz), 1.26 (3H, t, J= 7.2 Hz), 1.52 (2H, dq, J= 3.0, 7.2 Hz), 2.23 (1H, dt, J= 13.2, 7.2 Hz), 2.26 (1H, dt, J= 13.2, 7.2 Hz), 2.40 (3H, s), 2.58 (2H, dt, J= 3.8, 7.9 Hz) , 3.21 (1H, dt, J= 13.6, 7.2 Hz), 3.25 (1H, dt, J= 13.6, 7.2 Hz), 4.15 (2H, q, J= 7.2 Hz), 5.36 (1H, s), 6.16 (1H, t, J= 7.9 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.31 (5H, s), 7.82 (2H, d, J= 8.3 Hz), 9.63 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.95 (3H, t, J = 7.2 Hz), 1.26 (3H, t, J = 7.2 Hz), 1.52 (2H, dq, J = 3.0, 7.2 Hz), 2.23 (1H, dt, J = 13.2, 7.2 Hz), 2.26 (1H, dt, J = 13.2, 7.2 Hz), 2.40 (3H, s), 2.58 (2H, dt, J = 3.8, 7.9 Hz), 3.21 ( 1H, dt, J = 13.6, 7.2 Hz), 3.25 (1H, dt, J = 13.6, 7.2 Hz), 4.15 (2H, q, J = 7.2 Hz), 5.36 (1H, s), 6.16 (1H, t , J = 7.9 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.31 (5H, s), 7.82 (2H, d, J = 8.3 Hz), 9.63 (1H, br).
元素分析値: C28H31N305S3 · 0.4H20として計算値: C, 56.72; H, 5.41; N, 7.09; S, 16.22 ( ) .実測値: C, 56.75; H, 5.28; N, 6.92; S, 16.48 (%) . Elemental analysis: C 28 H 31 N 3 0 5 S 3 · 0.4H 2 0 Calculated: C, 56.72; H, 5.41; N, 7.09; S, 16.22 (). Observed: C, 56.75; H, 5.28; N, 6.92; S, 16.48 (%).
実施例 9 - 7 Examples 9-7
Ή-NMR (300MHz, CDC13) : δ 0.86 (3H, t, J= 7.2 Hz), 1.42 (2H, m), 1.71 (2H, m), 2.42 (3H, s), 2.73 (2H, t, J= 7.5 Hz), 3.22 (2H, q, J- 7.2 Hz), 4.31 (1H, dd, J= 8.3, 6.8 Hz), 7.01 (1H, s), 7.21 (2H, brd, J= 7.2 Hz), 7.31 (1H, m), 7.32 (2H, brd, J= 8.3 Hz), 7.38 (2H, brt, J= 7.5 Hz), 7.90 (2H, d, 1= 8.3 Hz), 9.87 (1H, br). Ή-NMR (300MHz, CDC1 3 ): δ 0.86 (3H, t, J = 7.2 Hz), 1.42 (2H, m), 1.71 (2H, m), 2.42 (3H, s), 2.73 (2H, t, J = 7.5 Hz), 3.22 (2H, q, J- 7.2 Hz), 4.31 (1H, dd, J = 8.3, 6.8 Hz), 7.01 (1H, s), 7.21 (2H, brd, J = 7.2 Hz) , 7.31 (1H, m), 7.32 (2H, brd, J = 8.3 Hz), 7.38 (2H, brt, J = 7.5 Hz), 7.90 (2H, d, 1 = 8.3 Hz), 9.87 (1H, br) .
実施例 9-8Example 9-8
—NMR (300MHz, CDC13) : δ 0.82 (3H, t, J= 7.5 Hz), 1.33 (2H, m), 1.57 (1H, m), 1.62 (1H, m), 2.43 (3H, s), 4.20 (2H, s), 4.23 (1H, dd, J= 8.7, 6.4 Hz), 6.97 (1H, s), 7.19 (2H, d, J= 7.2 Hz), 7.29 (1H, m), 7.29 (5H, s), 7.30 (2H, d, J= 8.3 Hz), 7.36 (2H, t, J= 7.5 Hz), 7.90 (2H, d, J= 8.3 Hz), 9.75 (1H, br). -NMR (300MHz, CDC1 3): δ 0.82 (3H, t, J = 7.5 Hz), 1.33 (2H, m), 1.57 (1H, m), 1.62 (1H, m), 2.43 (3H, s), 4.20 (2H, s), 4.23 (1H, dd, J = 8.7, 6.4 Hz), 6.97 (1H, s), 7.19 (2H, d, J = 7.2 Hz), 7.29 (1H, m), 7.29 (5H , s), 7.30 (2H, d, J = 8.3 Hz), 7.36 (2H, t, J = 7.5 Hz), 7.90 (2H, d, J = 8.3 Hz), 9.75 (1H, br).
元素分析値: C3。H29N303S3として計算値: C, 62.58; H, 5.08; N, 7.30; S, 16.71Elemental analysis: C 3. Calculated for H 29 N 3 0 3 S 3 : C, 62.58; H, 5.08; N, 7.30; S, 16.71
( ) .実測値: C, 62.34; H, 5.12; N, 7.19; S, 16.56 (%) . (). Found: C, 62.34; H, 5.12; N, 7.19; S, 16.56 (%).
実施例 9-9 Example 9-9
Ή-NMR (300MHz, CDC13) : δ 0.85 (3Η, t, J= 7.5 Hz), 1.41 (2H, m), 1.69 (2H, m), 2.42 (3H, s), 2.56 (2H, brt, J= 7.2 Hz), 3.30 (2H, m), 4.29 (1H, dd, J= 8.3, 6.8 Hz), 4.43 (2H, brd, J= 5.7 Hz),' 5.90 (1H, m), 7.00 (1H, s), 7.21 (2H, brd, J= 7.5 Hz), 7.29 (6H, m), 7.36 (2H, brt, J= 7.5 Hz), 7.88 (2H, d, J= 8.3 Hz), 9.7 (1H, br). 元素分析値: C33H34N404S3 · 0.8 0として計算値: C, 59.94; H, 5.43; N, 8.47 (%) . 実測値: C, 60.02; H, 5.42; N, 8.09 (%) . Ή-NMR (300MHz, CDC1 3 ): δ 0.85 (3Η, t, J = 7.5 Hz), 1.41 (2H, m), 1.69 (2H, m), 2.42 (3H, s), 2.56 (2H, brt, J = 7.2 Hz), 3.30 (2H, m), 4.29 (1H, dd, J = 8.3, 6.8 Hz), 4.43 (2H, brd, J = 5.7 Hz), '5.90 (1H, m), 7.00 (1H , S), 7.21 (2H, brd, J = 7.5 Hz), 7.29 (6H, m), 7.36 (2H, brt, J = 7.5 Hz), 7.88 (2H, d, J = 8.3 Hz), 9.7 (1H , br). Elemental analysis: C 33 H 34 N 4 0 4 S 3 · 0.8 0 Calculated:. C, 59.94; H, 5.43; N, 8.47 (%) Found: C, 60.02; H, 5.42 ; N, 8.09 (%).
実施例 9-10 Example 9-10
'H-NMR (300MHz, CDC13) : δ 0.90 (3Η, brt, J= 7.1 Hz), 1.26 (3H, t, J= 7.1 Hz), 1.34 (2H, m), 1.42 (2H, m), 2.19 (2H, brq, J= 7.5 Hz), 2.41 (3H, s), 2.67 (2H, t, J= 7.4 Hz), 3.23 (2H, t, J= 7.4 Hz), 3.60 (2H, s), 4.15 (2H, q, J= 7.1 Hz), 6.08 (1H, t, J= 7.9 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.74 (1H, br) . 'H-NMR (300MHz, CDC1 3): δ 0.90 (3Η, brt, J = 7.1 Hz), 1.26 (3H, t, J = 7.1 Hz), 1.34 (2H, m), 1.42 (2H, m), 2.19 (2H, brq, J = 7.5 Hz), 2.41 (3H, s), 2.67 (2H, t, J = 7.4 Hz), 3.23 (2H, t, J = 7.4 Hz), 3.60 (2H, s), 4.15 (2H, q, J = 7.1 Hz), 6.08 (1H, t, J = 7.9 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.74 ( 1H, br).
実施例 9 - 11 Examples 9 to 11
'H-NMR (300MHz, CDC13) : δ 0.88 (3H, brt, J= 6.9 Hz), 1.26 (3H, t, J= 7.1 Hz), 1.28 (4H, brs), 1.31 (2H, m), 1.44 (2H, brqiuni, J= 7.3 Hz), 2.18 (2H, q, J= 7.4 Hz), 2.41 (3H, s), 2.67 (2H, t, J- 7.4 Hz), 3.23 (2H, t, J= 7.4 Hz), 3.60 (2H, s), 4.15 (2H, q, J= 7.1 Hz), 6.08 (1H, t, J= 7.9 Hz), 7.29 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.74 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 0.88 (3H, brt, J = 6.9 Hz), 1.26 (3H, t, J = 7.1 Hz), 1.28 (4H, brs), 1.31 (2H, m), 1.44 (2H, brqiuni, J = 7.3 Hz), 2.18 (2H, q, J = 7.4 Hz), 2.41 (3H, s), 2.67 (2H, t, J- 7.4 Hz), 3.23 (2H, t, J = 7.4 Hz), 3.60 (2H, s), 4.15 (2H, q, J = 7.1 Hz), 6.08 (1H, t, J = 7.9 Hz), 7.29 (2H, d, J = 8.3 Hz), 7.87 ( 2H, d, J = 8.3 Hz), 9.74 (1H, br).
実施例 9 - 12 Examples 9-12
'H-NMR (300MHz, CDC13) : δ 0.90 (3H, t, J= 7.1 Hz), 1.33 (2H, m), 1.41 (2H, m), 2.16 (2H, q, J= 7.4 Hz), 2.41 (3H, s) , 3.53 (2H, s), 4.20 (2H, s), 6.04 (1H, t, J= 7.9 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.29 (5H, s), 7.86 (2H, d, J= 8.3 Hz), 9.67 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.90 (3H, t, J = 7.1 Hz), 1.33 (2H, m), 1.41 (2H, m), 2.16 (2H, q, J = 7.4 Hz), 2.41 (3H, s), 3.53 (2H, s), 4.20 (2H, s), 6.04 (1H, t, J = 7.9 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.29 (5H, s) ), 7.86 (2H, d, J = 8.3 Hz), 9.67 (1H, brs).
元素分析値: C25H27N305S3として計算値: C, 58.45; H, 5.30; N, 8.18; S, 18.73 (%) .実測値: C, 58.45; H, 5.26; N, 8.20; S, 18.80 (%) . Elemental analysis: C 25 H 27 N 3 0 5 S 3 Calculated:. C, 58.45; H, 5.30; N, 8.18; S, 18.73 (%) Found: C, 58.45; H, 5.26 ; N, 8.20; S, 18.80 (%).
実施例 9-13 . · Example 9-13.
Ή-N R (300MHz, CDC13) : δ 0.88 (3Η, t, J= 6.9 Hz), 1.27 (4H, m), 1.33 (2H, m), 1.43 (2H, brquint, J= 7.1 Hz), 2.15 (2H, q, J- 7.4 Hz), 2.41 (3H, s), 3.53 (2H, s), 4.21 (2H, s)., 6.04 (1H, t, J= 7.9 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.29 (5H, s), 7.86 (2H, d, J= 8.3 Hz), 9.66 (1H, brs). Ή-NR (300MHz, CDC1 3 ): δ 0.88 (3Η, t, J = 6.9 Hz), 1.27 (4H, m), 1.33 (2H, m), 1.43 (2H, brquint, J = 7.1 Hz), 2.15 (2H, q, J- 7.4 Hz), 2.41 (3H, s), 3.53 (2H, s), 4.21 (2H, s)., 6.04 (1H, t, J = 7.9 Hz), 7.27 (2H, d , J = 8.3 Hz), 7.29 (5H, s), 7.86 (2H, d, J = 8.3 Hz), 9.66 (1H, brs).
元素分析値: C27H31N305S3として計算値: C, 59.86; H, 5.77; N, 7.76; S, 17.76 (%) .実測値: C, 59.72; H, 5.70; N, 7.79; S, 17.68 (%) . Elemental analysis: C 27 H 31 N 3 0 5 S 3 Calculated:. C, 59.86; H, 5.77; N, 7.76; S, 17.76 (%) Found: C, 59.72; H, 5.70 ; N, 7.79; S, 17.68 (%).
実施例 9 - 14 'H-NMR (300MHz, CDC13) : δ 0.88 (3H, t, J= 7.0 Hz), 1.26 (4H, m), 1.32 (2H, m), 1.59 (2H, m), 2.42 (3H, s), 3.51 (1H, t, J= 7.5 Hz), 4.20 (2H, s), 4.90 (1H, brs), 5.50 (1H, brs), 7.28 (2H, d, J= 8.3 Hz), 7.29 (5H, s), 7.87 (2H, d, J= 8.3 Hz), 9.69 (1H, brs). Examples 9-14 'H-NMR (300MHz, CDC1 3): δ 0.88 (3H, t, J = 7.0 Hz), 1.26 (4H, m), 1.32 (2H, m), 1.59 (2H, m), 2.42 (3H, s ), 3.51 (1H, t, J = 7.5 Hz), 4.20 (2H, s), 4.90 (1H, brs), 5.50 (1H, brs), 7.28 (2H, d, J = 8.3 Hz), 7.29 (5H , s), 7.87 (2H, d, J = 8.3 Hz), 9.69 (1H, brs).
元素分析値: C26H29N303S3として計算値: C, 59.18; H, 5.54; N, 7.96; S, 18.23Elemental analysis: C 26 H 29 N 3 0 3 S 3 Calculated: C, 59.18; H, 5.54 ; N, 7.96; S, 18.23
(%)、 実測値: C, 59.06; H, 5.31; N, 7.94; S, 18.46 (%) . (%), Found: C, 59.06; H, 5.31; N, 7.94; S, 18.46 (%).
実施例 9-15 Example 9-15
¾-NMR (300MHz, CDC13) : δ 0.88 (3H, t, J= 6.8 Hz), 1.28 (4H, m), 1.43 (2H, m), 2.15 (2H, q, J= 7.5 Hz), 2.41 (3H, s), 3.53 (2H, s), 4.21 (2H, s), 6.04 (1H, t, J- 7.9 Hz), 7.27 (2H, d, J= 8.3 Hz), 7.29 (5H, s), 7.86 (2H, d, J= 8.3 Hz), 9.66 (1H, brs). ¾-NMR (300MHz, CDC1 3 ): δ 0.88 (3H, t, J = 6.8 Hz), 1.28 (4H, m), 1.43 (2H, m), 2.15 (2H, q, J = 7.5 Hz), 2.41 (3H, s), 3.53 (2H, s), 4.21 (2H, s), 6.04 (1H, t, J-7.9 Hz), 7.27 (2H, d, J = 8.3 Hz), 7.29 (5H, s) , 7.86 (2H, d, J = 8.3 Hz), 9.66 (1H, brs).
元素分析値: C26H29N303S3として計算値: C, 59.18; H, 5.54; N, 7.96; S, 18.23Elemental analysis: C 26 H 29 N 3 0 3 S 3 Calculated: C, 59.18; H, 5.54 ; N, 7.96; S, 18.23
(%), 実測値: C, 59.09; H, 5.31; N, 8.07; S, 18.21 (%) . (%), Found: C, 59.09; H, 5.31; N, 8.07; S, 18.21 (%).
実施例 9-16 Example 9-16
Ή-NMR (300MHz, CDC13) : δ 1.26 (3H, t, J= 7.2 Hz), 2.42 (3H, s), 2.68 (2H, t, J= 7.1 Hz), 3.26 (2H, t, J= 7.1 Hz), 3.81 (2H, s), 4.16 (2H, q, J- 7.2 Hz), 6.90 (1H, s), 7.31 (2H, d, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.76 (1H, brs). 元素分析値: C19H2。N305S3C1として計算値: C, 45.46; H, 4.02; N, 8.37; S, 19.16; Cl, 7.06 (%) 、 実測値: C, 45.41; H, 3.81; N, 8.31; S, 19.15; Cl, 7.23 (%) . 実施例 9_17 Ή-NMR (300MHz, CDC1 3 ): δ 1.26 (3H, t, J = 7.2 Hz), 2.42 (3H, s), 2.68 (2H, t, J = 7.1 Hz), 3.26 (2H, t, J = 7.1 Hz), 3.81 (2H, s), 4.16 (2H, q, J- 7.2 Hz), 6.90 (1H, s), 7.31 (2H, d, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.76 (1H, brs) elemental analysis:. C 19 H 2. Calculated for N 3 0 5 S 3 C1: C, 45.46; H, 4.02; N, 8.37; S, 19.16; Cl, 7.06 (%), found: C, 45.41; H, 3.81; N, 8.31; S , 19.15; Cl, 7.23 (%). Example 9_17
NMR (300 MHz, CDC13) : (31.30 (3H, t, J=7.2Hz), 2.70 (2H, t, J=7.3Hz), 3.27 (2H, t, J=7.3Hz), 3.70 (3H, s), 4.19 (2H, q, J=7.2Hz), 4.67 (2H, s), 6.61 (1H, s), 7.48-7.62 (3H, m), 7.96-8.02 (2H, m). NMR (300 MHz, CDC1 3) : (31.30 (3H, t, J = 7.2Hz), 2.70 (2H, t, J = 7.3Hz), 3.27 (2H, t, J = 7.3Hz), 3.70 (3H, s), 4.19 (2H, q, J = 7.2Hz), 4.67 (2H, s), 6.61 (1H, s), 7.48-7.62 (3H, m), 7.96-8.02 (2H, m).
実施例 9 - 18 Examples 9-18
'H-NMR (300 MHz, CDC13) : 61.29 (3H, t, J=6.9Hz), 4.18 (2H, q, J=6.9Hz), 4.25 (2H, s), 4.41 (2H, s), 6.69 (1H, s), 7.27-7.34 (5H, ID), 7.44-7.62 (3H, m), 7.94-8.01 (2H, m), 9.50-10.0 (1H, m). 'H-NMR (300 MHz, CDC1 3): 61.29 (3H, t, J = 6.9Hz), 4.18 (2H, q, J = 6.9Hz), 4.25 (2H, s), 4.41 (2H, s), 6.69 (1H, s), 7.27-7.34 (5H, ID), 7.44-7.62 (3H, m), 7.94-8.01 (2H, m), 9.50-10.0 (1H, m).
実施例 9-19 Example 9-19
Ή-NMR (200MHz, CDCl3-DMS0-d6= 9:1) : δ 2.42 (3H, s), 2.93 (3H, m), 3.22 (2H, m), 4.39 (1H, brs), 5.06 (1H, brs), 5.60 (1H, brs), 7.14-7.29 (7H, m), 7.86 (2H, d, J= 8.4 Hz). Ή-NMR (200 MHz, CDCl 3 -DMS0-d 6 = 9: 1): δ 2.42 (3H, s), 2.93 (3H, m), 3.22 (2H, m), 4.39 (1H, brs), 5.06 (1H, brs), 5.60 (1H, brs), 7.14-7.29 (7H, m), 7.86 (2H, d, J = 8.4 Hz).
元素分析値: C23H21N305S3-H20 として計算値: C, 51.77; H, 4.34; N, 7.87; S, 18.03 (%), 実測値; C, 51.46; H, 4.33; N, 7.87; S, 17.79 ( ) . Elemental analysis: C 23 H 21 N 3 0 5 S 3 -H 2 0 Calculated: C, 51.77; H, 4.34 ; N, 7.87; S, 18.03 (%), Found; C, 51.46; H, 4.33; N, 7.87; S, 17.79 ().
実施例 9-20 Example 9-20
'H-NMR (200MHz, 'CDC13) : δ 1.26 (3H, t, J= 7.1 Hz), 1.43 (3H, d, J= 7.0 Hz), 1.80 (3H, d, J= 7.0 Hz), 2.42 (3H, s), 2.66 (2H, t, J= 7.3 Hz), 3.21 (2H, t, J= 7.3 Hz), 4.15 (2H, q, J= 7.2 Hz), 4.27 (1H, q, J= 6.9 Hz), 5.94 (1H, q, J= 7.2 Hz), 7.31 (2H, d, J= 8.2 Hz), 7.89 (2H, d, J= 8.6 Hz), 9.70 (1H, brs). 元素分析値: C21H25N305S3 として計算値: C, 50.89; H, 5.08; , 8.48; S, 19.41 ( ) 、 実測値: C, 50.74; H, 5.17; N, 8.48; S, 19.39 {%) . 'H-NMR (200MHz,' CDC1 3): δ 1.26 (3H, t, J = 7.1 Hz), 1.43 (3H, d, J = 7.0 Hz), 1.80 (3H, d, J = 7.0 Hz), 2.42 (3H, s), 2.66 (2H, t, J = 7.3 Hz), 3.21 (2H, t, J = 7.3 Hz), 4.15 (2H, q, J = 7.2 Hz), 4.27 (1H, q, J = 6.9 Hz), 5.94 (1H, q, J = 7.2 Hz), 7.31 (2H, d, J = 8.2 Hz), 7.89 (2H, d, J = 8.6 Hz), 9.70 (1H, brs). : C 21 H 25 N 3 0 5 S 3 Calculated: C, 50.89; H, 5.08;, 8.48; S, 19.41 (), Found: C, 50.74; H, 5.17; N, 8.48; S, 19.39 {%).
融点. 141.5- 142.0°C 141.5- 142.0 ° C
実施例 9-21 Example 9-21
Ή— NMR (200MHz, CDC13) : δ 1.03 (3H, t, J= 7.3 Hz), 1.26 (3H, t, J= 7.2 Hz), 1.76 (2H, dquint, J= 7.7, 4.0 Hz), 2.43 (3H, s), 2.67 (2H, t, J= 7.4 Hz), 3.24 (2H, t, J= 7.2 Hz), 3.52 (1H, i, J= 7.6 Hz), 4.16 (2H, q, J= 7.1 Hz), 4.95 (1H, d, J= 1.0 Hz), 5.55 (1H, brs), 7.31 (2H, d, J= 8.2 Hz), 7.88 (2H, d, J= 8.4 Hz), 9.71 (1H, brs). Ή- NMR (200MHz, CDC1 3) : δ 1.03 (3H, t, J = 7.3 Hz), 1.26 (3H, t, J = 7.2 Hz), 1.76 (2H, dquint, J = 7.7, 4.0 Hz), 2.43 (3H, s), 2.67 (2H, t, J = 7.4 Hz), 3.24 (2H, t, J = 7.2 Hz), 3.52 (1H, i, J = 7.6 Hz), 4.16 (2H, q, J = 7.1 Hz), 4.95 (1H, d, J = 1.0 Hz), 5.55 (1H, brs), 7.31 (2H, d, J = 8.2 Hz), 7.88 (2H, d, J = 8.4 Hz), 9.71 (1H , brs).
元素分析値: C21H25N305S3 として計算値: C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%) 、 実測値: C, 50.78; H, 5.01; N, 8.43; S, 19.40 (%) . Elemental analysis: Calculated for C 21 H 25 N 3 0 5 S 3 : C, 50.89; H, 5.08; N, 8.48; S, 19.41 (%), found: C, 50.78; H, 5.01; N, 8.43; S, 19.40 (%).
融点. 122.5-123.0°C Melting point.122.5-123.0 ° C
実施例 9 - 22 Examples 9 to 22
Ή-NMR (200MHz, CDC13) : δ 0.91 (6Η, d, J- 6.2 Hz), 1.43 (3H, d, J= 7.0 Hz), 1.58 (2H, m), 1.63 (1H, quint, J= 6.6 Hz), 1.80 (3H, d, J= 7.2 Hz), 2.43 (3H, s), 2.94 (2H, brt, J= 7.9 Hz), 4.26 (1H, q, J= 7.1 Hz), 5.94 (1H, q, J- 7.1 Hz), 7.30 (2H, d, J= 8.4 Hz), 7.89 (2H, d, J= 8.4 Hz), 9.68 (1H, brs). 元素分析値: C21H27N303S3 として計算値: C, 54.17; H, 5.84; , 9.02; S, 20.66 Ή-NMR (200MHz, CDC1 3 ): δ 0.91 (6Η, d, J- 6.2 Hz), 1.43 (3H, d, J = 7.0 Hz), 1.58 (2H, m), 1.63 (1H, quint, J = 6.6 Hz), 1.80 (3H, d, J = 7.2 Hz), 2.43 (3H, s), 2.94 (2H, brt, J = 7.9 Hz), 4.26 (1H, q, J = 7.1 Hz), 5.94 (1H , q, J- 7.1 Hz), 7.30 (2H, d, J = 8.4 Hz), 7.89 (2H, d, J = 8.4 Hz), 9.68 (1H, brs). Elemental analysis: C 21 H 27 N 3 0 3 S 3 calculated: C, 54.17; H, 5.84 ;, 9.02; S, 20.66
( ) 、 実測値: C, 54.18; H, 5.85; N, 9.03; S, 20.60 (%) . (), Found: C, 54.18; H, 5.85; N, 9.03; S, 20.60 (%).
融点. 168.5-171.0°C 実施例 9 - 23 Melting point.168.5-171.0 ° C Examples 9 to 23
¾-N R (200MHz, CDC13) : δ 1.26 (3H, t, J= 7.1 Hz), 1.34 (3H, d, J= 6.8 Hz), 1.67 (3H, d, J= 7.3 Hz), 2.44 (3H, s), 2.56 (2H, t, J= 7.0 Hz), 3.19 (2H, t, J- 6.8 Hz), 4.14 (2H, q, J= 7.1 Hz), 4.47 (1H, q, J= 7.1 Hz), 5.15 (1H, q, J= 7.3 Hz), 7.34 (2H, d, J= 8.0 Hz), 7.89 (2H, d, J= 8.4 Hz), 9.86 (1H, brs). 元素分析値: C21H25N305S3 · ¾0 として計算値: C, 49.11; H, 5.30; N, 8.18; S, 18.73 ¾-NR (200MHz, CDC1 3 ): δ 1.26 (3H, t, J = 7.1 Hz), 1.34 (3H, d, J = 6.8 Hz), 1.67 (3H, d, J = 7.3 Hz), 2.44 (3H , s), 2.56 (2H, t, J = 7.0 Hz), 3.19 (2H, t, J-6.8 Hz), 4.14 (2H, q, J = 7.1 Hz), 4.47 (1H, q, J = 7.1 Hz) ), 5.15 (1H, q, J = 7.3 Hz), 7.34 (2H, d, J = 8.0 Hz), 7.89 (2H, d, J = 8.4 Hz), 9.86 (1H, brs). Elemental analysis: C 21 H 25 N 3 0 5 S 3 · Calculated as ¾0: C, 49.11; H, 5.30; N, 8.18; S, 18.73
(%) 、 実測値: C, 49.35; H, 4.96; N, 8.18; S, 18.99 (%) . (%), Found: C, 49.35; H, 4.96; N, 8.18; S, 18.99 (%).
融点. 85.5-87.0°C 85.5-87.0 ° C
実施例 9-24 Example 9-24
Ή— NMR (200MHz, CDC13) : δ 1.02 (3Η, t, J- 7.4 Hz), 1.75 (2H, m), 2.43 (3H, s), 2.48 (3H, s), 3.53 (1H, t, J= 7.5 Hz), 4.94 (1H, d, J= 1.2 Hz), 5.56 (1H, d, J= 1.2 Hz), 7.31 (2H, d, J= 8.0 Hz), 7.88 (2H, d, J= 8.4 Hz), 9.64 (1H, br). Ή- NMR (200MHz, CDC1 3) : δ 1.02 (3Η, t, J- 7.4 Hz), 1.75 (2H, m), 2.43 (3H, s), 2.48 (3H, s), 3.53 (1H, t, J = 7.5 Hz), 4.94 (1H, d, J = 1.2 Hz), 5.56 (1H, d, J = 1.2 Hz), 7.31 (2H, d, J = 8.0 Hz), 7.88 (2H, d, J = 8.4 Hz), 9.64 (1H, br).
'元素分析値: C17H19N303S3 として計算値: C, 49.86; H, 4.86; N, 10.26; S, 23.49'Elemental analysis: C 17 H 19 N 3 0 3 S 3 Calculated: C, 49.86; H, 4.86 ; N, 10.26; S, 23.49
(%) 、 実測値: C, 49.68; H, 4.63; N, 10.14; S, 23.41 (%) . (%), Found: C, 49.68; H, 4.63; N, 10.14; S, 23.41 (%).
融点. 163.0-165. Ot: Melting point. 163.0-165. Ot:
実施例 9-25 Example 9-25
酢酸ェチル -ジェチルェ一テルから再結晶し橙色結晶として得た。 The crystals were recrystallized from ethyl acetate-ethyl ether to obtain orange crystals.
融点: 185-187°C. Melting point: 185-187 ° C.
赤外吸収スぺクトル(IR)' (KBr中) リ: 3438, 3310, 3179, 3065, 2959, 2928, 2857, 1732, 1669, 1645, 1620, 1557, 1497 cm-1. Infrared absorption spectrum (IR) '(in KBr): 3438, 3310, 3179, 3065, 2959, 2928, 2857, 1732, 1669, 1645, 1620, 1557, 1497 cm -1 .
- NMR (CDC13) : δ 0.840-0.942 (3Η, m), 1.26-1.42 (4H, m), 1.60-1.76 (2H, m), 2.43 (3H, s), 3.00 (2H, t, J=7.0 Hz), 4.38 (1H, s), 5.15 (1H, d, J=1.4 Hz), 5.53 (1H, bs), 5.80 (1H, d, J=1.6 Hz), 6.04 (1H, bs), 7.31 (2H, d, J=8.8 Hz), 7.88 (2H, d, J=8.0 Hz), 9.74 (1H, bs) . - NMR (CDC1 3): δ 0.840-0.942 (3Η, m), 1.26-1.42 (4H, m), 1.60-1.76 (2H, m), 2.43 (3H, s), 3.00 (2H, t, J = 7.0 Hz), 4.38 (1H, s), 5.15 (1H, d, J = 1.4 Hz), 5.53 (1H, bs), 5.80 (1H, d, J = 1.6 Hz), 6.04 (1H, bs), 7.31 (2H, d, J = 8.8 Hz), 7.88 (2H, d, J = 8.0 Hz), 9.74 (1H, bs).
元素分析値: C2。H24N404S3として計算値: C, 49.98; H, 5.03; N, 11.66; S, 20.00Elemental analysis: C 2. H 24 N 4 0 4 S 3 Calculated: C, 49.98; H, 5.03 ; N, 11.66; S, 20.00
(%), 実測値: C, 49.72; H, 4.98; N, 11.60; S, 19.76 (%) . (%), Found: C, 49.72; H, 4.98; N, 11.60; S, 19.76 (%).
実施例 9-26 Example 9-26
酢酸ェチル -ジェチルェ一テルから再結晶し橙色結晶として得た。 融点: 154- 156°C. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain orange crystals. Melting point: 154-156 ° C.
赤外吸収スぺクトル(IR) (KBr中) V: 3250, 2961, 2930, 2863, 2243, 2209, 1763, 1642, 1628, 1561 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 3250, 2961, 2930, 2863, 2243, 2209, 1763, 1642, 1628, 1561 cm " 1 .
'H-NMR (CDCI3) : δ 0.890 (3Η, t, J=7.0Hz), 1.24-1.44 (4H, m), 1.58-1.74 (2H, m), 2.44 (3H, s), 2.86-3.02 ,(2H, m), 4.53 (1H, s), 5.24 (1H, d, 1=2.2 Hz), 5.84 (1H, d, J=2.2 Hz), 7.33 (2H, d, 7=8. Hz) , 7.89 (2H, d, J=8.0 Hz), 9.82 (1H, bs). 'H-NMR (CDCI 3 ): δ 0.890 (3Η, t, J = 7.0 Hz), 1.24-1.44 (4H, m), 1.58-1.74 (2H, m), 2.44 (3H, s), 2.86-3.02 , (2H, m), 4.53 (1H, s), 5.24 (1H, d, 1 = 2.2 Hz), 5.84 (1H, d, J = 2.2 Hz), 7.33 (2H, d, 7 = 8.Hz) , 7.89 (2H, d, J = 8.0 Hz), 9.82 (1H, bs).
元素分析値: C2。H22N403S3として計算値: C, 51.93; H, 4.79; N, 12.11; S, 20.79 (%)、 実測値: C, 51.63; H, 4.93; N, 11.95; S, 20.50 (%) . Elemental analysis: C 2. H 22 N 4 0 3 S 3 Calculated: C, 51.93; H, 4.79 ; N, 12.11; S, 20.79 (%), Found: C, 51.63; H, 4.93 ; N, 11.95; S, 20.50 ( %).
実施例 9 - 27 Example 9-27
N- {8- (ィソペンチルスルファニル) -6-メチル -3-ォキソ -5- [ (Z) -プロピリデ ン]- 2, 3, 5, 6-テトラヒド口- 1Hィミダゾ [5, 1- c] [1,4]チアジン- 1-ィリデント 4- 実施例 4-467のシリ力ゲルクロマトグラフィーにおいて実施例 -467の化合物 画分に続く赤色画分を濃縮乾固して化合物 (44 mg, 0.092 IMOI)を濃赤色油状物 として得た。 N- {8- (Isopentylsulfanyl) -6-methyl-3-oxo-5-[(Z) -propylidene] -2,3,5,6-tetrahydrid-1H imidazo [5,1-c [1,4] thiazine-1-ylidentate 4- The compound of Example -467 was subjected to the silylation gel chromatography of Example 4-467. The red fraction following the fraction was concentrated to dryness to give the compound (44 mg, 0.092 IMOI) was obtained as a dark red oil.
Ή-NMR (200MHz, CDC13) : δ 0.90 (6H, d, J= 6.6 Hz), 1.03 (3H, t, J= 7.5 Hz), 1.49 (3H, d, J= 7.0 Hz), 1.54 (2H, m), 1.66 (1H, quint, J= 6.2 Hz), 2.10 (2H, btt, J= 7.0 Hz), 2.43 (3H, s), 2.95 (2H, t, J= 7.7 Hz), 3.81 (1H, q, J= 5.8 Hz), 5.30 (1H, t, J- 6.9 Hz), 7.31 (2H, d, J= 8.4 Hz), 7.90 (2H, d, J= 8.0 Hz), 9.65 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 0.90 (6H, d, J = 6.6 Hz), 1.03 (3H, t, J = 7.5 Hz), 1.49 (3H, d, J = 7.0 Hz), 1.54 (2H , m), 1.66 (1H, quint, J = 6.2 Hz), 2.10 (2H, btt, J = 7.0 Hz), 2.43 (3H, s), 2.95 (2H, t, J = 7.7 Hz), 3.81 (1H , q, J = 5.8 Hz), 5.30 (1H, t, J- 6.9 Hz), 7.31 (2H, d, J = 8.4 Hz), 7.90 (2H, d, J = 8.0 Hz), 9.65 (1H, brs ).
実施例 10-1 Example 10-1
式 (XX I V) において R2が 2 -(メトキシカルポニル)ェチル基、 R5が n—へキ シル基、 Zが S〇2、 R15が P-トルィル基である化合物の合成 Synthesis of a compound of the formula (XX IV) wherein R 2 is a 2- (methoxycarbonyl) ethyl group, R 5 is an n-hexyl group, Z is S〇 2 , and R 15 is a P-tolyl group
参考例 3-2で合成した化合物(739 mg, 1.72腿 ol)および炭酸力リゥム(476 mg, 3.4 mmol) を DMF (15ml) に混和し、 室温で 2-ブロモオクタン酸ェチル (0.493 ml, 2.06讓 ol) を加えて 1時間撹拌した。 さらに 80 °Cで 13時間撹拌後再び室 温で 5日間放置した。反応液を濃縮乾固し、酢酸ェチル(25 ml)を加えて混和し、 0.3N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し溶媒を減圧下 に溜去した。 残渣にエーテルを加えて混和し、 生成した沈澱物を濾取し、 エーテ ルで洗浄後、 乾燥して標題化合物の黄色粉末 (484 mg, 0.874 mmol) を得た。 Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 1.27 (3Η, t, J- 7.1 Hz), 2.43 (3H, s), 2.73 (2H, t, J= 7.4 Hz), 3.24 (2H, t, J= 7.4 Hz), 4.17 (2H, q, J= 7.1 Hz), 4.98 (1H, s), 7.20 (2H, dd, J= 8.3, 1.5 Hz), 7.32 (2H, d, J= 8.3 Hz), 7.37 (3H, m), 7.87 (2H, d, J= 8.3 Hz), 9.69 (1H, brs). The compound synthesized in Reference Example 3-2 (739 mg, 1.72 mol) and carbonated carbonate (476 mg, 3.4 mmol) were mixed with DMF (15 ml), and ethyl 2-bromooctanoate (0.493 ml, 2.06 ml) was added at room temperature. And the mixture was stirred for 1 hour. After further stirring at 80 ° C for 13 hours, the mixture was again left at room temperature for 5 days. The reaction mixture is concentrated to dryness, mixed with ethyl acetate (25 ml), washed with 0.3N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and the solvent is evaporated under reduced pressure. Evaporated. Ether was added to the residue and mixed, and the resulting precipitate was collected by filtration, washed with ether, and dried to give a yellow powder of the title compound (484 mg, 0.874 mmol). Ή-NMR (300 MHz, in double-mouthed form): δ 1.27 (3Η, t, J- 7.1 Hz), 2.43 (3H, s), 2.73 (2H, t, J = 7.4 Hz), 3.24 (2H, t, J = 7.4 Hz), 4.17 (2H, q, J = 7.1 Hz), 4.98 (1H, s), 7.20 (2H, dd, J = 8.3, 1.5 Hz), 7.32 (2H, d, J = 8.3) Hz), 7.37 (3H, m), 7.87 (2H, d, J = 8.3 Hz), 9.69 (1H, brs).
元素分析値: C24H23N305S3 として計算値: C, 54.42; H, 4.38; N, 7.93; S, 18.16Elemental analysis: C 24 H 23 N 3 0 5 S 3 Calculated: C, 54.42; H, 4.38 ; N, 7.93; S, 18.16
(%) 、 実測値; C, 54.23; H, 4.46; N, 8.00; S, 18.45 (%) . (%), Found; C, 54.23; H, 4.46; N, 8.00; S, 18.45 (%).
実施例 10-2 Example 10-2
式 (XX I V) において R 2がべンジル基、 R5が 2-ヒドロキシェチル基、 Zが S 02、 R 15が P-卜ルイル基である化合物の合成 R 2 Gabe Njiru group in the formula (XX IV), R 5 is 2-hydroxy E butyl group, the synthesis of Z is S 0 2, compound R 15 is P- Bok Ruiru group
参考例 11で合成した化合物(117mg, 0.279腿 ol)および炭酸カリウム (77 mg, 0.558 imol) を DMF (2 ml) に混和し、室温で 2-ブロモプチルラクトン (0.033 ml, 0.335 mmol) を加えて 2時間撹拌後、 さらに 80 °Cで 4時間撹拌した。 反応液を 酢酸ェチル(25ml) を加えて混和し、 0.3N塩酸、水および飽和食塩水で洗浄後、 硫酸ナトリゥムで乾燥し溶媒を減圧下に溜去した。 残渣に酢酸ェチルおよびェ一 テルを加えて混和し、 生成した沈澱物を濾取し、 エーテルで洗浄後、 乾燥して黄 色粉末 (64mg) を得た。 これをメタノールから再結晶化し、 標題化合物の黄色粉 末 (42 mg, 0.083 mmol) を得た。 The compound synthesized in Reference Example 11 (117 mg, 0.279 tmol) and potassium carbonate (77 mg, 0.558 imol) were mixed with DMF (2 ml), and 2-bromobutyltyl lactone (0.033 ml, 0.335 mmol) was added at room temperature. After stirring for 2 hours, the mixture was further stirred at 80 ° C for 4 hours. The reaction solution was mixed with ethyl acetate (25 ml), washed with 0.3 N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and the solvent was distilled off under reduced pressure. The residue was mixed with ethyl acetate and ether, and the resulting precipitate was collected by filtration, washed with ether, and dried to give a yellow powder (64 mg). This was recrystallized from methanol to give the title compound as a yellow powder (42 mg, 0.083 mmol).
'H-NMR (300 MHz, 重クロ口ホルム-重ジメチルスルホキシド 9 : 1中): δ 1.81 (1H, m), 2.20 (1H, dq, J= 13.6, 6.4 Hz), 2.43 (3H, s), 3.67 (2H, brq, J= 4.9 Hz), 3.92 (1H, t, J= 7.2 Hz), 4.19 (1H, brt, J= 4.9 Hz), 4.26 (2H, brs), 7.28 (2H, d, J= 8.3 Hz), 7.31 (5H, s), 7.87 (2H, d, J= 8.3 Hz), 11.05 (1H, br). 元素分析値: C22H21N305S3 として計算値: C, 52.47; H, 4.20; N, 8.34; S, 19.10 (%), 実測値; C, 52.41; H, 4.19; N, 8.13; S, 19.10 (%) . ' 以下に実施例 10- 1、 10 - 2で合成した化合物および同様にして合成した化合物の 一覧を示す。 〔表 9 7〕'H-NMR (300 MHz, double-mouthed form-deuterated dimethyl sulfoxide in 9: 1): δ 1.81 (1H, m), 2.20 (1H, dq, J = 13.6, 6.4 Hz), 2.43 (3H, s) , 3.67 (2H, brq, J = 4.9 Hz), 3.92 (1H, t, J = 7.2 Hz), 4.19 (1H, brt, J = 4.9 Hz), 4.26 (2H, brs), 7.28 (2H, d, J = 8.3 Hz), 7.31 ( 5H, s), 7.87 (2H, d, J = 8.3 Hz), 11.05 (1H, br) elemental analysis:. C 22 H 21 N 3 0 5 S 3 calculated: C, 52.47; H, 4.20; N, 8.34; S, 19.10 (%), found; C, 52.41; H, 4.19; N, 8.13; S, 19.10 (%). ' A list of compounds synthesized in 10-2 and compounds synthesized in the same manner is shown below. (Table 9 7)
差簪ぇ甩弒 (ΜΜ 〔表 9 8〕ぇ 甩 弒 (Table 98)
差替え^弒(細 (?26) 以下に実施例 10-3〜10- 22で合成した化合物のデータを示す。 Replacement ^ 弒 (fine (? 2 6) The data of the compounds synthesized in Examples 10-3 to 10-22 are shown below.
実施例 10-3 Example 10-3
'H-NMR (300 MHz, 重クロ口ホルム中): δ 1.28 (3Η, t, J= 7.1 Hz), 2.43 (3H, s), 3.60 (2H, s), 3.81 (2H, s), 4.21 (2H, q, J= 7.1 Hz), 7.32 (2H, brd, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.89 (1H, br). 'H-NMR (300 MHz, in double-mouthed form): δ 1.28 (3Η, t, J = 7.1 Hz), 2.43 (3H, s), 3.60 (2H, s), 3.81 (2H, s), 4.21 (2H, q, J = 7.1 Hz), 7.32 (2H, brd, J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.89 (1H, br).
元素分析値: C17H17N306S3 として計算値: C, 44.82; H, 3.76; N, 9.22; S, 21.12Elemental analysis: C 17 H 17 N 3 0 6 S 3 Calculated: C, 44.82; H, 3.76 ; N, 9.22; S, 21.12
(%), 実測値; C, 45.17; H, 4.04; N, 8.97; S, 21.16 (%) . (%), Found; C, 45.17; H, 4.04; N, 8.97; S, 21.16 (%).
実施 10-4 Implementation 10-4
Ή-NMR (300 MHz, 重ジメチルスルホキシド中) : δ 2.39 (3Η, s), 3.75 (2H, s), 3.98 (2H, s), 7.41 (2H, d, J= 8.1 Hz), 7.88 (2H, d, J= 8.1 Hz), 12.01 (1H, br) , 13.07 (1H, br). Ή-NMR (300 MHz, in heavy dimethyl sulfoxide): δ 2.39 (3Η, s), 3.75 (2H, s), 3.98 (2H, s), 7.41 (2H, d, J = 8.1 Hz), 7.88 (2H , d, J = 8.1 Hz), 12.01 (1H, br), 13.07 (1H, br).
元素分析値: C15H13N306S3 として計算値; C, 42.15; H, 3.07; , 9.83; S, 22.50Elemental analysis: Calculated as C 15 H 13 N 3 0 6 S 3; C, 42.15; H, 3.07;, 9.83; S, 22.50
(%)、 実測値; C, 42.19; H, 3.05; N, 9.92; S, 22.47 (%) . (%), Found; C, 42.19; H, 3.05; N, 9.92; S, 22.47 (%).
実施例 10-5 Example 10-5
Ή-NMR (300 MHz, 重クロ口ホルム中): δ 0.88 (3Η, t, J= 6.9 Hz), 1.26 (26H, brs), 1.64 (2H, brquint, J= 7 Hz) , 2.44 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.14 (2H, t, J= 6.8 Hz), 7.33 (2H, brd, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.86 (1H, br). Ή-NMR (300 MHz, in double-mouthed form): δ 0.88 (3Η, t, J = 6.9 Hz), 1.26 (26H, brs), 1.64 (2H, brquint, J = 7 Hz), 2.44 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.14 (2H, t, J = 6.8 Hz), 7.33 (2H, brd, J = 8.3 Hz), 7.87 (2H, d, J = 8.3) Hz), 9.86 (1H, br).
実施例 10-6 Example 10-6
¾-腿 (300 MHz, 重クロ口ホルム中): δ 2.44 (3Η, s), 3.61 (2H, s), 3.82 (2H, s), 3.77 (3H, s), 7.33 (2H, brd, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz) , 9.82 (1H, br). ¾-thigh (300 MHz, in a double-mouthed form): δ 2.44 (3Η, s), 3.61 (2H, s), 3.82 (2H, s), 3.77 (3H, s), 7.33 (2H, brd, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.82 (1H, br).
実施例 10-7 Example 10-7
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 0.88 (3Η, t, J= 6.9 Hz), 1.26 (16H, brs), 1.33 (2H, m), 1.64 (2H, brquint, J= 6.9 Hz), 2.44 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.14 (2H, t, J= 6.8 Hz), 7.33 (2H, brd, J= 8.3 Hz), 7.87 (2H, d, J= 8.3 Hz), 9.77 (1H, br). 'H-NMR (300 MHz, in double-mouthed form): δ 0.88 (3Η, t, J = 6.9 Hz), 1.26 (16H, brs), 1.33 (2H, m), 1.64 (2H, brquint, J = 6.9 Hz), 2.44 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.14 (2H, t, J = 6.8 Hz), 7.33 (2H, brd, J = 8.3 Hz), 7.87 (2H, d, J = 8.3 Hz), 9.77 (1H, br).
実施例 10-8 Example 10-8
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 0.88 (3Η, t, J= 6.9 Hz), 1.26 (12H, brs), 1.33 (2H, m), 1.64 (2H, brquint, J= 6.9 Hz), 2.44 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.14 (2H, i, J= 6.8 Hz), 7.33 (2H, brd, J= 8.2 Hz), 7.87 (2H, d, J= 8.2 Hz), 9.76 (1H, br). 'H-NMR (300 MHz, in double chromatographic form): δ 0.88 (3Η, t, J = 6.9 Hz), 1.26 (12H, brs), 1.33 (2H, m), 1.64 (2H, brquint, J = 6.9 Hz), 2.44 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.14 (2H, i, J = 6.8 Hz), 7.33 (2H, brd, J = 8.2 Hz), 7.87 (2H, d, J = 8.2 Hz), 9.76 (1H, br).
実施例 10-9 Example 10-9
^-NMR (300 MHz, 重クロ口ホルム-重ジメチルスルホキシド 3: 1中): δ 2.41 (3Η, s), 3.55 (2H, s), 3.92 (2H, s), 5.21 (2H, s), 7.31 (2H, d, J= 8.1 Hz), 7.36 (1H, m), 7.44 (2H, d, J= 8.1 Hz), 7.45 (2H, t, J= 7.3 Hz), 7.59 (4H, brd, J= 8.0 Hz), 7.91 (2H, d, J= 8.1 Hz), 11.73 (1H, br). ^ -NMR (300 MHz, double-mouthed form-deuterated dimethyl sulfoxide in 3: 1): δ 2.41 (3Η, s), 3.55 (2H, s), 3.92 (2H, s), 5.21 (2H, s), 7.31 (2H, d, J = 8.1 Hz), 7.36 (1H, m), 7.44 (2H, d, J = 8.1 Hz), 7.45 (2H, t, J = 7.3 Hz), 7.59 (4H, brd, J = 8.0 Hz), 7.91 (2H, d, J = 8.1 Hz), 11.73 (1H, br).
実施例 10-10 Example 10-10
'H-NMR (300 MHz, 重クロ口ホルム中): δ 1.99 (2Η, it, J= 7.6, 6.5 Hz), 2.41 (3H, s), 2.68 (2H, t, J= 7.6 Hz), 3.58 (2H, s), 3.79 (2H, s) , 4.18 (2H, t, J= 6.5 Hz), 7.15 (2H, brd, J= 7.0 Hz), 7.20 (1H, m), 7.29 (2H, t, J= 8.0 Hz), 7.31 (2H, d, J- 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.87 (1H, br). 'H-NMR (300 MHz, in double-mouthed form): δ 1.99 (2Η, it, J = 7.6, 6.5 Hz), 2.41 (3H, s), 2.68 (2H, t, J = 7.6 Hz), 3.58 (2H, s), 3.79 (2H, s), 4.18 (2H, t, J = 6.5 Hz), 7.15 (2H, brd, J = 7.0 Hz), 7.20 (1H, m), 7.29 (2H, t, J = 8.0 Hz), 7.31 (2H, d, J- 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.87 (1H, br).
元素分析値: C24H23N306S3 として計算値: C, 52.83; H, 4.25; N, 7.70; S, 17.63 (%), 実測値; C, 52.67; H, 4.24; N, 7.89; S, 17.94 (%) . Elemental analysis: C 24 H 23 N 3 0 6 S 3 Calculated: C, 52.83; H, 4.25 ; N, 7.70; S, 17.63 (%), Found; C, 52.67; H, 4.24 ; N, 7.89; S, 17.94 (%).
実施例 10-11 Example 10-11
'H-NMR (300 MHz, 重クロ口ホルム中): δ 1.60 (3Η, s), 1.60 (3H, s), 1.68 (3H, s), 1.71 (3H, s), 1.99 (4H, m), 2.07 (6H, m), 2.43 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.67 (2H, d, J= 7.2 Hz), 5.08 (2H, t, J= 5.6 Hz), 5.33 (1H, brt, J= 7.2 Hz), 7.33 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J- 8.3 Hz), 9.89 (1H, br). 'H-NMR (300 MHz, in double-mouthed form): δ 1.60 (3Η, s), 1.60 (3H, s), 1.68 (3H, s), 1.71 (3H, s), 1.99 (4H, m) , 2.07 (6H, m), 2.43 (3H, s), 3.59 (2H, s), 3.81 (2H, s), 4.67 (2H, d, J = 7.2 Hz), 5.08 (2H, t, J = 5.6) Hz), 5.33 (1H, brt, J = 7.2 Hz), 7.33 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J- 8.3 Hz), 9.89 (1H, br).
元素分析値: C3。H37N306S3 · 0.8H20 として計算値: C, 55.76; H, 6.02; N, 6.50; S, 14.88 (%), 実測値; C, 55.50; H, 5.58; N, 6.70; S, 15.14 (%) . Elemental analysis: C 3. H 37 N 3 0 6 S 3 · 0.8H 2 0 Calculated: C, 55.76; H, 6.02; N, 6.50; S, 14.88 (%), found; C, 55.50; H, 5.58; N, 6.70 ; S, 15.14 (%).
実施例 10 - 12 Examples 10-12
Ή-NMR (300 MHz, 重クロ口ホルム中): δ 1.60 (3Η, s), 1.68 (3H, s), 1.71 (3H, s), 2.07 (4H, m), 2.44 (3H, s), 3.58 (2H, s), 3.81 (2H, s), 4.67 (2H, d, J- 7.2 Hz), 5.07 (1H, m), 5.33 (1H, brt, J= 7.2 Hz), 7.33 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.87 (1H, brs). Ή-NMR (300 MHz, in double-mouthed form): δ 1.60 (3Η, s), 1.68 (3H, s), 1.71 (3H, s), 2.07 (4H, m), 2.44 (3H, s), 3.58 (2H, s), 3.81 (2H, s), 4.67 (2H, d, J- 7.2 Hz), 5.07 (1H, m), 5.33 (1H, brt, J = 7.2 Hz), 7.33 (2H, d , J = 8.3 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.87 (1H, brs).
元素分析値: C25H29N3OsS3 として計算値: C, 53.27; H, 5.19; N, 7.45 (%), 実測値; C, 52.88; H, 4.93; N, 7.54 (%) . Elemental analysis: C 25 H 29 N 3 O s S 3 Calculated: C, 53.27; H, 5.19; N, 7.45 (%), Found: C, 52.88; H, 4.93; N, 7.54 (%).
実施例 10-13 Example 10-13
'H-NMR (300 MHz, 重ク P口ホルム中) : δ 1.26 (3Η, t, J- 7.1 Hz), 2.43 (3H, s), 2.70 (2H, t, J= 7.2 Hz), 3.30 (2H, t, J= 7.2 Hz), 3.65 (2H, s), 4.16 (2H, q, J= 7.1 Hz), 7.32 (2H, d, J= 8.3 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.8 (1H, br). 'H-NMR (300 MHz, heavy duty in P-holm): δ 1.26 (3Η, t, J- 7.1 Hz), 2.43 (3H, s), 2.70 (2H, t, J = 7.2 Hz), 3.30 ( 2H, t, J = 7.2 Hz), 3.65 (2H, s), 4.16 (2H, q, J = 7.1 Hz), 7.32 (2H, d, J = 8.3 Hz), 7.86 (2H, d, J = 8.3) Hz), 9.8 (1H, br).
元素分析値: C18H19N306S3 - 0.3H20 として計算値: C, 45.52; H, 4.16; N, 8.85; S, 20.25 (%), 実測値; C, 45.41; H, 3.90; N, 8.94; S, 20.41 ( ) . Elemental analysis: C 18 H 19 N 3 0 6 S 3 - 0.3H 2 0 Calculated: C, 45.52; H, 4.16 ; N, 8.85; S, 20.25 (%), Found; C, 45.41; H , 3.90; N, 8.94; S, 20.41 ().
実施例 10-14 Example 10-14
4種異性体混合物 4-isomer mixture
-纖 (300 MHz, 重クロ口ホルム中) : 6 1.54/1.55 (3H, d, J= 7.2/6.6 Hz), 1.58/1.59 (3H,. d, J- 6.9/7.4 Hz), 2.43 (3H, s), 3.72/3.83 (1H, q, J= 7.0 Hz), 4.16 (1H, q, J= 7.3 Hz), 7.33 (2H, d, J= 8.2 Hz), 7.86/7.87 (2H, d, J= 8.2 Hz). -Fiber (300 MHz, in double-holed holm): 6 1.54 / 1.55 (3H, d, J = 7.2 / 6.6 Hz), 1.58 / 1.59 (3H, .d, J-6.9 / 7.4 Hz), 2.43 (3H , s), 3.72 / 3.83 (1H, q, J = 7.0 Hz), 4.16 (1H, q, J = 7.3 Hz), 7.33 (2H, d, J = 8.2 Hz), 7.86 / 7.87 (2H, d, J = 8.2 Hz).
実施例 10-15 Example 10-15
4種異性体混合物 4-isomer mixture
Ή -画 R (300 MHz, 重クロ口ホルム中) : δ 0.88 (3Η, t, J= 6.9 Hz), 1.26 (24H, brs), 1.29 (2H, m), 1.51/1.58 (3H, d, J= 7.0/7.2 Hz), 1.57 (3H, d, J= 7.1 Hz), 1.62 (2H, m), 2.44 (3H, s), 3.71/3.73 (1H, q, J- 7.0 Hz), 4.11 (2H, dt, J= 5.2, 6.7 Hz), 4.17/4.20 (1H, q, J= 7.3/7.4 Hz), 7.33 (2H, d, J= 8.2 Hz), 7.88 (2H, d, J= 8:2 Hz), 9.6 (1H, br). Ή-Image R (300 MHz, in double-mouthed form): δ 0.88 (3Η, t, J = 6.9 Hz), 1.26 (24H, brs), 1.29 (2H, m), 1.51 / 1.58 (3H, d, J = 7.0 / 7.2 Hz), 1.57 (3H, d, J = 7.1 Hz), 1.62 (2H, m), 2.44 (3H, s), 3.71 / 3.73 (1H, q, J- 7.0 Hz), 4.11 ( 2H, dt, J = 5.2, 6.7 Hz), 4.17 / 4.20 (1H, q, J = 7.3 / 7.4 Hz), 7.33 (2H, d, J = 8.2 Hz), 7.88 (2H, d, J = 8: 2 Hz), 9.6 (1H, br).
実施例 10-16 Example 10-16
'H-NMR (300MHz, CDC13) : δ 0.88 (3H, t, J= 6.4 Hz), 1.26 (6H, br), 1.30 (2H, m), 1.64 (1H, m), 1,95 (1H, m), 2.42 (3H, s), 3.51 (1H, dd, J= 7· 5, 6.4 Hz), 4.25 (2H, s), 7.30 (2H, brd, ,J= 8.3 Hz), 7.31 (5H, s), 7.86 (2H, d, J= 8.3 Hz), 9.86 (1H, brs). 'H-NMR (300MHz, CDC1 3): δ 0.88 (3H, t, J = 6.4 Hz), 1.26 (6H, br), 1.30 (2H, m), 1.64 (1H, m), 1,95 (1H , m), 2.42 (3H, s), 3.51 (1H, dd, J = 7.5, 6.4 Hz), 4.25 (2H, s), 7.30 (2H, brd,, J = 8.3 Hz), 7.31 (5H , s), 7.86 (2H, d, J = 8.3 Hz), 9.86 (1H, brs).
元素分析値: C2SH29N304S3' 0.4H20として計算値: C, 56.68; H, 5.45; N, 7.62 ( )、 実測値: C, 56.51; H, 5.24; , 7.67 (%) . Elemental analysis: C 2S H 29 N 3 0 4 S 3 '0.4H 2 0 Calculated: C, 56.68; H, 5.45 ; N, 7.62 (), Found: C, 56.51; H, 5.24 ;, 7.67 (%).
実施例 10 - 17 ¾-NMR (300MHz, CDC13) : δ 1.27 (3H, t, J= 7.2 Hz), 1.61 (3H, d, J= 6.8 Hz), 2.44 (3H, s), 2.69 (2H, t, J= 7.4 Hz), 3.29 (2H, dt, J二 2.3, 7.2 Hz), 3.76 (1H, q, J= 7.2 Hz), 4.17 (2H, q, J= 7.2 Hz), 7.32 (2H, brd, J= 7.9 Hz), 7.86 (2H, d, J= 8.3 Hz), 9.66 (1H, br). Examples 10-17 ¾-NMR (300MHz, CDC1 3 ): δ 1.27 (3H, t, J = 7.2 Hz), 1.61 (3H, d, J = 6.8 Hz), 2.44 (3H, s), 2.69 (2H, t, J = 7.4 Hz), 3.29 (2H, dt, J-2.3, 7.2 Hz), 3.76 (1H, q, J = 7.2 Hz), 4.17 (2H, q, J = 7.2 Hz), 7.32 (2H, brd, J = 7.9 Hz), 7.86 (2H, d, J = 8.3 Hz), 9.66 (1H, br).
元素分析値: Ci9H21N3OsS3として計算値: C, 47.19; H, 4.38; N, 8.69; S, 19.89 (%) 、 実測値: C, 47.32; H, 4.38; N, 8.73; S, 19.66 (%) . Elemental analysis: C i9 H 21 N 3 O s S 3 Calculated: C, 47.19; H, 4.38 ; N, 8.69; S, 19.89 (%), Found: C, 47.32; H, 4.38 ; N, 8.73; S, 19.66 (%).
実施例 10-18 Example 10-18
¾ー醒 R (300MHz, CDC13) : δ 0.90 (3H, t, J= 7.0 Hz), 1.33 (4H, m), 1.64 (1H, m), 1.95 (1H, m), 2.43 (3H, s), 3.50 (1H, dd, J= 7.9, 6.4 Hz), 4.26 (2H, s), 7.31 (2H, d, J= 8.3 Hz), 7.31 (5H, s), 7.86 (2H, d, J= 8.3 Hz) , 9.81 (1H, brs). 元素分析値: C24H25N304S3として計算値: C, 55.90; H, 4.89; N, 8.15; S, 18.65. (%), 実測値: C, 55.84; H, 4.88; N, 8.02; S, 18.58 (%) . ¾ chromatography Awakening: R (300MHz, CDC1 3): δ 0.90 (3H, t, J = 7.0 Hz), 1.33 (4H, m), 1.64 (1H, m), 1.95 (1H, m), 2.43 (3H, s ), 3.50 (1H, dd, J = 7.9, 6.4 Hz), 4.26 (2H, s), 7.31 (2H, d, J = 8.3 Hz), 7.31 (5H, s), 7.86 (2H, d, J = . 8.3 Hz), 9.81 (1H , brs) elemental analysis: C 24 H 25 N 3 0 4 S 3 calculated:. C, 55.90; H, 4.89; N, 8.15; S, 18.65 (%), measured Values: C, 55.84; H, 4.88; N, 8.02; S, 18.58 (%).
実施例 10-19 ,Example 10-19,
¾-NMR (300MHz, CDC13) : δ 1.23 (3Η, t, ]= 7.0 Hz), 2.43 (3H, s), 4.19 (1H, dq, J= 12.5, 7.2 Hz), 4.24 (1H, dq, J= 12.5, 7.2 Hz), 4.26 (1H, d, J= 13.6 ¾-NMR (300MHz, CDC1 3 ): δ 1.23 (3Η, t,] = 7.0 Hz), 2.43 (3H, s), 4.19 (1H, dq, J = 12.5, 7.2 Hz), 4.24 (1H, dq, J = 12.5, 7.2 Hz), 4.26 (1H, d, J = 13.6
Hz), 4.28 (1H, d, J= 13.6 Hz), 4.31 (1H, s), 7.31 (2H, d, J- 8.3 Hz), 7.32Hz), 4.28 (1H, d, J = 13.6 Hz), 4.31 (1H, s), 7.31 (2H, d, J- 8.3 Hz), 7.32
(5H, s), 7.85 (2H, d,' J= 8.3 Hz), 9.86 (1H, brs). (5H, s), 7.85 (2H, d, 'J = 8.3 Hz), 9.86 (1H, brs).
元素分析値: C23H21N406S3として計算値: C, 51.96; H, 3.98; N, 7.90; S, 18.10 (%)、 実測値: C, 51.74; H, 3.99; N, 7.78; S, 18.07 (%) . Elemental analysis: Calculated as C 23 H 21 N 4 0 6 S 3: C, 51.96; H, 3.98; N, 7.90; S, 18.10 (%), Found: C, 51.74; H, 3.99 ; N, 7.78; S, 18.07 (%).
実施例 10 20 Example 10 20
'H-NMR (300MHz, CDC13) : δ 2.43 (3Η, s), 4.28 (2H, s), 4.77 (1H, s), 7.26 (3H, m), 7.29 (2H, d, J= 8.3 Hz), 7.32 (5H, s), 7.38 (2H, dd, J- 5.3, 2.3 Hz), 7.85 (2H, d, J= 8.3 Hz), 9.80 (1H, br). 'H-NMR (300MHz, CDC1 3): δ 2.43 (3Η, s), 4.28 (2H, s), 4.77 (1H, s), 7.26 (3H, m), 7.29 (2H, d, J = 8.3 Hz ), 7.32 (5H, s), 7.38 (2H, dd, J- 5.3, 2.3 Hz), 7.85 (2H, d, J = 8.3 Hz), 9.80 (1H, br).
元素分析値: C26H21N304S3として計算値: C, 58.30; H, 3.95; N, 7.84; S, 17.96 ( ) 、 実測値: C, 58.26; H, 3.80; N, 8.09; S, 17.83 ( ) . Elemental analysis: C 26 H 21 N 3 0 4 S 3 Calculated: C, 58.30; H, 3.95 ; N, 7.84; S, 17.96 (), Found: C, 58.26; H, 3.80 ; N, 8.09 S, 17.83 ().
実施例 10-21 Example 10-21
Ή-NMR (300MHz, CDC13) : <5 0.88 (3H, t, J= 6.6 Hz), 1.25 (18H, brs), 1.40 (2H, m), 1.64 (1H, m), 1.95 (1H, m), 2.43 (3H, s), 3.50 (1H, dd, J= 7.9, 6.4 Hz), 4.26 (2H, s), 7.30 (2H, d, J= 8.3 Hz), 7.31 (5H, s), 7.86 (2H, d, J= 8.3 Hz) , 9.80 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): <5 0.88 (3H, t, J = 6.6 Hz), 1.25 (18H, brs), 1.40 (2H, m), 1.64 (1H, m), 1.95 (1H, m ), 2.43 (3H, s), 3.50 (1H, dd, J = 7.9, 6.4 Hz), 4.26 (2H, s), 7.30 (2H, d, J = 8.3 Hz), 7.31 (5H, s), 7.86 (2H, d, J = 8.3 Hz), 9.80 (1H, brs).
元素分析値: C32H41N304S3として計算値: C, 61.21; H, 6.58; N, 6.69; S, 15.32Elemental analysis: C 32 H 41 N 3 0 4 S 3 Calculated: C, 61.21; H, 6.58 ; N, 6.69; S, 15.32
(%), 実測値: C, 61.29; H, 6.58; N, 6.83; S, 15.27 (%) . (%), Found: C, 61.29; H, 6.58; N, 6.83; S, 15.27 (%).
実施例 10-22 Example 10-22
N-[3, 5-ジォキソ- 8_ (イソプチルスルファ二ル)- 6-メチル- 2, 3, 5, 6-テトラヒドロ -1H-イミダゾ [5, l-c] [1,4]チアジン-卜ィリデン ]_4-メチルベンゼンスルホンァ ミド N- [3,5-Dioxo-8_ (isobutylsulfanyl) -6-methyl-2,3,5,6-tetrahydro-1H-imidazo [5, lc] [1,4] thiazine-triylidene ] _4-Methylbenzenesulfonamide
水素化ナトリウム(346 mg, 8.65腿01) (60%油性) のテトラヒドロフラン(15.0 ml) の懸濁液に、 参考例 17で得られた 2-{ [(イソブチルスルファ二ル) (5-{[(4- メチルフエニル)スルホニル]イミノ}- 2 ォキソ -4-イミダゾリジニリデン)メチ ル]スルファニル }プロピオン酸ェチル (3.50 g, 7.21 mmol) のテトラヒドロフラ ン (15.0ml) 溶液を加えた。 反応混合物を 30分間加熱還流した後、 1N塩酸を加 えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留 去して題記化合物 (1.66 g, 52%) を暗赤色結晶として得た。 酢酸ェチル-ジェチ ルェ一テルから再結晶して融点 163-165 の暗赤色結晶を得た。 To a suspension of sodium hydride (346 mg, 8.65 thigh 01) (60% oily) in tetrahydrofuran (15.0 ml) was added 2-{[(isobutylsulfanyl) (5- { A solution of [(4-methylphenyl) sulfonyl] imino} -2-oxo-4-imidazolidinylidene) methyl] sulfanyl} ethyl propionate (3.50 g, 7.21 mmol) in tetrahydrofuran (15.0 ml) was added. After the reaction mixture was heated to reflux for 30 minutes, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was distilled off under reduced pressure to give the title compound (1.66 g, 52%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl acetate gave dark red crystals with a melting point of 163-165.
赤外吸収スペクトル(IR) (KBr中) リ: 3260, 2959, 2930, 2872, 1802, 1713, 1632, 1557, 1495 cm"1. Infrared absorption spectrum (IR) (in KBr): 3260, 2959, 2930, 2872, 1802, 1713, 1632, 1557, 1495 cm " 1 .
Ή-NMR (CDC13) : δ 1.03 (6Η, d, J=6.6 Hz), 1.60 (3H, t, J=7.0 Hz), 1.81-1.96 (1H, in), 2.43 (3H, s), 2.90 (2H, d, J=6.6 Hz), 3.73 (1H, q, J-7.0 Hz), 7.32 (2H, d, J=8.0 Hz), 7.88 (2H, d, J=8. Hz), 9.83 (1H, bs). Ή-NMR (CDC1 3): δ 1.03 (6Η, d, J = 6.6 Hz), 1.60 (3H, t, J = 7.0 Hz), 1.81-1.96 (1H, in), 2.43 (3H, s), 2.90 (2H, d, J = 6.6 Hz), 3.73 (1H, q, J-7.0 Hz), 7.32 (2H, d, J = 8.0 Hz), 7.88 (2H, d, J = 8.Hz), 9.83 ( 1H, bs).
元素分析値: C18H21N304S3として Elementary analysis: as C 18 H 21 N 3 0 4 S 3
計算俥 (¾) : C, 49.18; H, 4.82; N, 9.56; S, 21.88. Computing Shari (¾): C, 49.18; H, 4.82; N, 9.56; S, 21.88.
実測値 (¾) : C, 48.98; H, 4.95; N, 9.49; S, 21.71. Found (¾): C, 48.98; H, 4.95; N, 9.49; S, 21.71.
実施例 11-1 Example 11-1
式 (XV I) において R1が n-へキシル基、 R5および R5がともにメチル基、 Z が S 02、 R 15が p-トルイル基である化合物の合成 Formula (XV I) in a hexyl group R 1 is the n-, synthesis of R 5 and R 5 are both methyl groups, Z is S 0 2, R 15 is p- tolyl group compound
実施例 4- 96で合成した化合物 (247 mg, 0.624 mmol) を THF (5 ml) に混和し、 室温で n-ブチルマグネシウムブロミド (1.31 ml, 1.0 M、 THF溶液) を加えて 46 時間撹拌した。 この間、 反応液に上記 n-ブチルマグネシウムプロミド (0.187 ml) を加えた。 反応液に 0. IN塩酸 (15 ml) および酢酸ェチル (15 ml) を加えて混和し た。 有機層を 0.1N塩酸、 水および飽和食塩水で洗浄後、 不溶物を除去し、 硫酸ナト リウムで乾燥し溶媒を減圧下に溜去した。 残渣をシリカゲル 60 (10g) のカラ ムクロマトグラフィーに付し、 酢酸ェチル:へキサン (20 : 80) の溶出画分を 減圧下濃縮乾固し、 標題化合物の橙色油状物 (19 mg, 0.044 mmol) を得た。 'H-NMR (200 MHz, 重クロ口ホルム中) : δ 0.85 (3Η, t, J= 6.7 Hz), 1.17 (6H, m), 1.41 (2H, m), 1.73 (3H, q, J- 0.8 Hz), 2.25 (3H, brs), 2.43 (3H, s), 2.68 (2H, t, J= 7.6 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.79 (2H, d, J= 8.4 Hz), 9.74 (1H, brs). The compound synthesized in Example 4-96 (247 mg, 0.624 mmol) was mixed with THF (5 ml), n-butylmagnesium bromide (1.31 ml, 1.0 M, THF solution) was added at room temperature, and the mixture was stirred for 46 hours. . During this time, add the above n-butylmagnesium bromide (0.187 ml) to the reaction mixture. Was added. 0.1 IN hydrochloric acid (15 ml) and ethyl acetate (15 ml) were added to the reaction solution and mixed. After the organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, insolubles were removed, dried over sodium sulfate, and the solvent was distilled off under reduced pressure. The residue was subjected to column chromatography on silica gel 60 (10 g), and the fraction eluted with ethyl acetate: hexane (20:80) was concentrated to dryness under reduced pressure to give the title compound as an orange oil (19 mg, 0.044 mmol) ). 'H-NMR (200 MHz, in double-mouthed form): δ 0.85 (3Η, t, J = 6.7 Hz), 1.17 (6H, m), 1.41 (2H, m), 1.73 (3H, q, J- 0.8 Hz), 2.25 (3H, brs), 2.43 (3H, s), 2.68 (2H, t, J = 7.6 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.79 (2H, d, J = 8.4 Hz), 9.74 (1H, brs).
元素分析値: C21 7N303S2 として計算値: C, 58.17; H, 6.28; N, 9.69; S, 14.79 (%) 実測値; C, 57.96; H, 6.21; N, 9.86; S, 14.82 ( ) . Elemental analysis: Calculated for C 2 17 N 3 0 3 S 2 : C, 58.17; H, 6.28; N, 9.69; S, 14.79 (%) found; C, 57.96; H, 6.21; N, 9.86; S, 14.82 ().
融点: 171.0- 174.0°C Melting point: 171.0-174.0 ° C
実施例 11 - 2 Example 11-2
式 (XV I) において R1がベンゾィルメチル基、 R 5がェチル基、 R 6がメチル 基、 Zが S02、 R 15が P-トルィル基である化合物の合成 Synthesis of a compound of the formula (XVI) in which R 1 is a benzoylmethyl group, R 5 is an ethyl group, R 6 is a methyl group, Z is S 0 2 , and R 15 is a P-tolyl group
実施例 4-376で合成した化合物 (166 mg, 0.323蘭 oi) を THF (4 ml) に混和 し、 - 78°Cでメチルマグネシウムブロミド (0.237 ml, 3.0 'Μ、 THF溶液) を加え て 1時間撹拌した。 反応液を室温にまで戻した後、 室温で 18時間撹拌した。 反 応液に 0.1N塩酸 (0.81ml) および酢酸ェチル (25 ml) を加えて混和した。 有機 層を 0.1N塩酸、水および飽和食塩水で洗浄後、硫酸ナ卜リゥムで乾燥し溶媒を減 圧下に溜去した。 残渣にメタノールを加え、 析出した粉末を濾取して赤色の粉末 を得た。 これをシリカゲル 60 (5g) のカラムクロマトグラフィーに付し、 酢酸 ェチル:へキサン (3 : 7〜4 : 6) の溶出画分を減圧下濃縮乾固した。 残渣に エーテルを加え、 析出した粉末を濾取、 エーテルで洗浄、 乾燥して標記化合物の 赤色粉末 (69 mg, 0.143腿 oU を得た。 The compound synthesized in Example 4-376 (166 mg, 0.323 orchid oi) was mixed with THF (4 ml), and methylmagnesium bromide (0.237 ml, 3.0′Μ, THF solution) was added at −78 ° C. to give 1 Stirred for hours. After returning the reaction solution to room temperature, it was stirred at room temperature for 18 hours. 0.1N hydrochloric acid (0.81 ml) and ethyl acetate (25 ml) were added to the reaction solution and mixed. The organic layer was washed with 0.1N hydrochloric acid, water and a saturated saline solution, dried over sodium sulfate, and the solvent was distilled off under reduced pressure. Methanol was added to the residue, and the precipitated powder was collected by filtration to obtain a red powder. This was subjected to column chromatography on silica gel 60 (5 g), and the fraction eluted with ethyl acetate: hexane (3: 7 to 4: 6) was concentrated to dryness under reduced pressure. Ether was added to the residue, and the precipitated powder was collected by filtration, washed with ether, and dried to give a red powder of the title compound (69 mg, 0.143 tU).
Ή-NMR (300 MHz, 重クロ口ホルム中) : 6 1.07 (3H, t, J= 7.5 Hz), 2.09 (2H, q, J- 7.6 Hz), 2.28 (3H, s), 2.33 (3H, s), 4.36 (2H, s), 6.98 (2H, d, J= 8.0 Hz), 7.34 (2H, d, J= 8.2 Hz), 7.43 (2H, brt, J= 7.3 Hz), 7.60 (1H, tt, J= 7.4, 1.2 Hz), 7.78 (2H, dm, J= 8.0 Hz), 9.68 (1H, brs). 元素分析値: C24H23N304S2 として計算値: C, 59.86; H, 4.81; N, 8.73; S, 13.32Ή-NMR (300 MHz, in double-mouthed form): 6 1.07 (3H, t, J = 7.5 Hz), 2.09 (2H, q, J-7.6 Hz), 2.28 (3H, s), 2.33 (3H, s), 4.36 (2H, s), 6.98 (2H, d, J = 8.0 Hz), 7.34 (2H, d, J = 8.2 Hz), 7.43 (2H, brt, J = 7.3 Hz), 7.60 (1H, tt, J = 7.4, 1.2 Hz), 7.78 (2H, dm, J = 8.0 Hz), 9.68 (1H, brs). Elemental analysis: C 24 H 23 N 3 0 4 S 2 Calculated: C, 59.86; H, 4.81 ; N, 8.73; S, 13.32
(%), 実測値; C, 59.47; H, 4.82; N, 8.62; S, 13.48 (%) . (%), Found; C, 59.47; H, 4.82; N, 8.62; S, 13.48 (%).
質量分析 (FAB-MS): 482 (M+H) . Mass spec (FAB-MS): 482 (M + H).
実施例 11-3 Example 11-3
式 (XV I) において R1がベンゾィルメチル基、 R5および R6がメチル基、 Z が S〇2、 R15が p-トルィル基、 である化合物の合成 Synthesis of a compound of the formula (XVI), wherein R 1 is a benzoylmethyl group, R 5 and R 6 are methyl groups, Z is S〇 2 , and R 15 is a p-tolyl group.
(方法 1) 実施例 4-75で得られた化合物 (524 mg, 1.05 mmol) を原料に実施例 11-2と同様にして標記化合物の赤色粉末 (39 mg, 0.083 應 ol) を得た。 (Method 1) Using the compound (524 mg, 1.05 mmol) obtained in Example 4-75 as a starting material, a red powder (39 mg, 0.083 ol) of the title compound was obtained in the same manner as in Example 11-2.
Ή -画 R (300 MHz, 重クロ口ホルム中) : δ 1.73 (3Η, q, J= .2 Hz), 2.28 (3H, q, J= 1.0 Hz), 2.32 (3H, s), 4.35 (2H, s), 6.98 (2H, brd, J= 7.6 Hz), 7.36 (2H, d, J= 8.2 Hz), 7.44 (2H, brt, J= 7.2 Hz), 7.60 (1H, brt, J= 7.4 Hz), 7.78 (2H, d, J= 8 Hz), 9.66 (1H, brs). Ή-Image R (300 MHz, in double-mouthed form): δ 1.73 (3Η, q, J = .2 Hz), 2.28 (3H, q, J = 1.0 Hz), 2.32 (3H, s), 4.35 ( 2H, s), 6.98 (2H, brd, J = 7.6 Hz), 7.36 (2H, d, J = 8.2 Hz), 7.44 (2H, brt, J = 7.2 Hz), 7.60 (1H, brt, J = 7.4) Hz), 7.78 (2H, d, J = 8 Hz), 9.66 (1H, brs).
元素分析値: C23H21N304S2 - 0.5H20 として計算値: C, 57.97; H, 4.65; N, 8.82; S, 13.46 (%), 実測値; C, 58.09; H, 4.43; N, 8.81; S, 13.48 (%) . Elemental analysis: C 23 H 21 N 3 0 4 S 2 - 0.5H 2 0 Calculated: C, 57.97; H, 4.65 ; N, 8.82; S, 13.46 (%), Found; C, 58.09; H , 4.43; N, 8.81; S, 13.48 (%).
(方法 2) 実施例 4-75で得られた化合物(262 rag, 0.52 讓 ol)およびトリフエ二 ルホスフィン(567 mg, 2.10 mmol)の THF (5.0 ml)溶液に n-ブチルリチウム(1.6 M へキサン溶液, 0.754 ml, 1.21 mmol) を加えて室温で 21時間攪拌.した。 反応液 に 1N塩酸 (1.21 ml) および水を加え、 酢酸ェチル(30 ml)で抽出した。 有機層 を 0.1N塩酸、 水および飽和食塩水で洗浄し、 乾燥、 濃縮した。 残渣をシリカゲ ル(10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:9-) に付し、 酢酸ェ チル—へキサン (4: 6)の画分を濃縮し、 メタノ一ルから結晶化して標記化合物 (46 rag, 0.098 匪 ol)を赤色粉末として得た。 (Method 2) To a solution of the compound obtained in Example 4-75 (262 rag, 0.52 acetyl) and triphenylphosphine (567 mg, 2.10 mmol) in THF (5.0 ml) was added n-butyllithium (to 1.6 M). A hexane solution (0.754 ml, 1.21 mmol) was added, and the mixture was stirred at room temperature for 21 hours. 1N Hydrochloric acid (1.21 ml) and water were added to the reaction solution, and the mixture was extracted with ethyl acetate (30 ml). The organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, dried and concentrated. The residue was subjected to chromatography on silica gel (10 g) (ethyl acetate-hexane, 1: 9-), and the ethyl acetate-hexane (4: 6) fraction was concentrated and crystallized from methanol. The title compound (46 rag, 0.098 ol) was obtained as a red powder.
融点: 137.0-137.5°C. Melting point: 137.0-137.5 ° C.
以下に実施例 11- 1〜11_3 で合成した化合物および同様にして合成した化合物 の一覧を示す。 〔表 9 9〕The following is a list of compounds synthesized in Examples 11-1 to 11_3 and compounds synthesized in the same manner. (Table 9 9)
麄替え用紙(規則 26) 以下に実施例 11- 4〜11- 8で合成した化合物のデータを示す。 麄 Replacement form (Rule 26) The data of the compounds synthesized in Examples 11-4 to 11-8 are shown below.
実施例 11 - 4 Example 11-4
'H-NMR (200MHz, CDC13) : 6 1.08 (3H, t, J= 7.3 Hz), 1.73 (3H, q, J= 0.6 Hz), 2.25 (3H, q, J= 1.2 Hz), 2.43 (3H, s), 2.74 (2H, q, J= 7.5 Hz), 7.30 (2H, d, J= 8.2 Hz), 7.80 (2H, d, J= 8.2 Hz), 9.75 (1H, brs). 'H-NMR (200MHz, CDC1 3): 6 1.08 (3H, t, J = 7.3 Hz), 1.73 (3H, q, J = 0.6 Hz), 2.25 (3H, q, J = 1.2 Hz), 2.43 ( 3H, s), 2.74 (2H, q, J = 7.5 Hz), 7.30 (2H, d, J = 8.2 Hz), 7.80 (2H, d, J = 8.2 Hz), 9.75 (1H, brs).
元素分析値: C17H19N303S2として計算値: C, 54.09; H, 5.07; , 11.13; S, 16.99Elemental analysis: C 17 H 19 N 3 0 3 S 2 Calculated: C, 54.09; H, 5.07 ;, 11.13; S, 16.99
(%) 、 実測値: C, 54.09; H, 4.91; N, 10.86; S, 16.83 (%) . (%), Found: C, 54.09; H, 4.91; N, 10.86; S, 16.83 (%).
融点. 202.5- 203.5°C Melting point. 202.5-203.5 ° C
実施例 11 - 5 Example 11-5
¾-NMR (200MHz, CDC13) : δ 1.07 (3H, t, J= 7.5 Hz), 1.09 (3H, t, J= 7.5 Hz), 2.09 (2H, q, J= 7.5 Hz), 2.25 (3H, s), 2.43 (3H, s), 2.76 (2H, q, J= 7.5 Hz), 7.30 (2H, d, J= 8.1 Hz), 7.80 (2H, d, J= 7.0 Hz), 9.75 (1H, brs). ¾-NMR (200MHz, CDC1 3 ): δ 1.07 (3H, t, J = 7.5 Hz), 1.09 (3H, t, J = 7.5 Hz), 2.09 (2H, q, J = 7.5 Hz), 2.25 (3H , s), 2.43 (3H, s), 2.76 (2H, q, J = 7.5 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.80 (2H, d, J = 7.0 Hz), 9.75 (1H , Brs).
元素分析値: C18H21N303S2として計算値: C, 55.22; H, 5.41; N, 10.73; S, 16.38 (%) 、 実測値: C, 54.82; H, 5.42; N, 10.62; S, 16.35 (%) . Elemental analysis: C 18 H 21 N 3 0 3 S 2 Calculated: C, 55.22; H, 5.41 ; N, 10.73; S, 16.38 (%), Found: C, 54.82; H, 5.42 ; N, 10.62; S, 16.35 (%).
融点. 184.0- 185.0°C Melting point. 184.0- 185.0 ° C
実施例 11-6 Example 11-6
'H-NMR (300MHz, CDC13) : δ 1.77 (3H, s), 2.08 (3Η, s), 2.24 (3H, s), 2.37 (3H, s), 6.87 (1H, s), 7.11 (2H, d, J= 8.0 Hz), 7.40 (2H, t, J= 7.6 Hz), 7.45 (2H, d, J= 8.4 Hz), 7.59 (1H, t, J= 7.4 Hz), 7.93 (2H, d, J= 7.5 Hz), 9.82 (1H, brs). ' 'H-NMR (300MHz, CDC1 3): δ 1.77 (3H, s), 2.08 (3Η, s), 2.24 (3H, s), 2.37 (3H, s), 6.87 (1H, s), 7.11 (2H , d, J = 8.0 Hz), 7.40 (2H, t, J = 7.6 Hz), 7.45 (2H, d, J = 8.4 Hz), 7.59 (1H, t, J = 7.4 Hz), 7.93 (2H, d , J = 7.5 Hz), 9.82 (1H, brs).
13C-NMR (75MHz, CDC13) : d 14.67 (C¾), 15.69 (CH3), 17.39 (CH3), 21.56 (CH3), 49.79 (CH), 106.47 (Q), 124.00 (Q), 126.46 ( CH) , 128.73 (2CH), 129.00 (2CH), 129.56 (2CH) , 133.78 (CH), 134.58 (Q), 137.44 (Q), 138.23 (Q), 143.91 (Q), 147.18 (Q), 147.92 (Q), 191.97 (Q). 13 C-NMR (75MHz, CDC1 3): d 14.67 (C¾), 15.69 (CH 3), 17.39 (CH 3), 21.56 (CH 3), 49.79 (CH), 106.47 (Q), 124.00 (Q), 126.46 (CH), 128.73 (2CH), 129.00 (2CH), 129.56 (2CH), 133.78 (CH), 134.58 (Q), 137.44 (Q), 138.23 (Q), 143.91 (Q), 147.18 (Q), 147.92 (Q), 191.97 (Q).
質量分析(FAB- MS) : M+H= 514, M+K= 552 Mass spectrometry (FAB-MS): M + H = 514, M + K = 552
融点. 169.0- 170.0°C Melting point.169.0- 170.0 ° C
実施例 11-7 Example 11-7
'H-NMR (200MHz, CDC13) : δ 1.36 (3H, d, J= 6.5 Hz), 1.76 (3H, q, J= 0.9 Hz), 2.25 (3H, q, J= 0, 9 Hz) , 2.43 (3H, s), 3.30 (1H, d, J= 6.1 Hz) , 5.23 (1H, quint, J= 6.4 Hz), 7.32 (2H, d, J= 8.3 Hz), 7.80 (2H, d, J= 8,3 Hz), 9.78 (1H, brs). 【H -醒 R (200MHz, CDC13+重水) <5 1.35 (3H, d, J= 6.5 Hz), 1.76 (3H, brs), 2.25 (3H, brq, J= 0.8 Hz), 2.43 (3H, s), 5.22 (1H, q, J= 6.4 Hz), 7.32 (2H, d, J= 8.1 Hz), 7.80 (2H, d, J= 8.3 Hz). 'H-NMR (200MHz, CDC1 3): δ 1.36 (3H, d, J = 6.5 Hz), 1.76 (3H, q, J = 0.9 Hz), 2.25 (3H, q, J = 0, 9 Hz), 2.43 (3H, s), 3.30 (1H, d, J = 6.1 Hz), 5.23 (1H, quint, J = 6.4 Hz), 7.32 (2H, d, J = 8.3 Hz), 7.80 (2H, d, J = 8.3 Hz), 9.78 (1H, brs). [H-Awake R (200MHz, CDC1 3 + (Heavy water) <5 1.35 (3H, d, J = 6.5 Hz), 1.76 (3H, brs), 2.25 (3H, brq, J = 0.8 Hz), 2.43 (3H, s), 5.22 (1H, q, J = 6.4 Hz), 7.32 (2H, d, J = 8.1 Hz), 7.80 (2H, d, J = 8.3 Hz).
元素分析値: C17H19N304S2として計算値: C, 51.89; H, 4.87; , 10.68 (%) 、 実 測値: C, 51.70; H, 4.96; N, 10.41 (%) .融点. 192.0-193.0°C Elemental analysis: C 17 H 19 N 3 0 4 S 2 Calculated: C, 51.89; H, 4.87 ;, 10.68 (%), actual Hakachi: C, 51.70; H, 4.96 ; N, 10.41 (%) Melting point: 192.0-193.0 ° C
質量分析 (FAB - MS):: M+H= 393, M+K= 432 Mass Spectrometry (FAB-MS) :: M + H = 393, M + K = 432
実施例 11 8 Example 11 8
¾-NMR (200MHz, CDC13) : δ 1.69 (3Η, q, J= 1.2 Hz), 2.26 (3H, q, J= 1.6 Hz), 2.43 (3H, s), 6.70 (1H, s), 7.31 (2H, d, J= 8.8 Hz), 7.80 (2H, d, J= 8.0 Hz), 9.60 (1H, br). ¾-NMR (200MHz, CDC1 3 ): δ 1.69 (3Η, q, J = 1.2 Hz), 2.26 (3H, q, J = 1.6 Hz), 2.43 (3H, s), 6.70 (1H, s), 7.31 (2H, d, J = 8.8 Hz), 7.80 (2H, d, J = 8.0 Hz), 9.60 (1H, br).
元素分析値: C15H15N303S2として計算値: C, 51.56; H, 4.33; N, 12.03; S, 18.35Elemental analysis: C 15 H 15 N 3 0 3 S 2 Calculated: C, 51.56; H, 4.33 ; N, 12.03; S, 18.35
(%) 、 実測値: C, 51.22; H, 4.39; N, 11.73; S, 18.14 ( ) . (%), Found: C, 51.22; H, 4.39; N, 11.73; S, 18.14 ().
実施例 12 1 Example 12 1
式 (XXV) において R 2が 2- (メトキシカルボニル)ェチル基、 R5が n-へキシ ル基、 R 6が水素原子である化合物の合成 Synthesis of a compound of the formula (XXV) wherein R 2 is a 2- (methoxycarbonyl) ethyl group, R 5 is an n-hexyl group, and R 6 is a hydrogen atom
参考例 3-3 で合成した化合物 (3.27g)と 2 -プロモォクタナール(2.5ml)の THF(80ml)と DMF (15ml)の懸濁液に 0°Cにてトリェチルァミン(3.8ml)を滴下し室 温にて 1時間撹拌した。酢酸ェチル(100ml)を加え、 有機層を 0.05M塩酸、 水、 飽 和食塩水で順次洗浄後、 硫酸マグネシウムで乾燥した。 溶媒を留去して得られる 残留物をシリカゲルカラムクロマトグラフィーに付し、 へキサン:酢酸ェチル (1:2)溶出部より式 (XX) において、 R 2が 2- (メトキシカルポニル)ェチル基、 R5が n-へキシル基、 R6が水素原子、 Zが CO、 R 15がフエニル基である化合 物の淡褐色油状物 (3.13g)を得た。 これをトルエン(50ml)に溶解し、 無水酢酸 (0.6ml)とカンファースルホン酸(0.9g)を加え 6(T にて 4時間撹拌した。 室温に 冷却後、反応液に酢酸ェチル(100ml)を加え、有機層を飽和炭酸ナトリウム水溶液 と飽和食塩水で洗浄し、 硫酸マグネシウムで乾燥後、 溶媒を留去して得られる残 留物をシリカゲル力ラムクロマトグラフィーに付し、へキサン:酢酸ェチル (2:1) 溶出部より表題化合物(1.08g, 46%)を得た。 一 NMR (300 MHz, CDC13) : 60.90 (3H, t, J=6.9Hz), 1.25-1.36 (6H, m), 1.43-1.53 (2H, m), 2.05 (2H, t, J=7.1Hz), 2.74 (2H, t, J=7.5Hz), 3.23 (2H, t, J=7.5Hz), 3.73 (3H, s), 6.50 (1H, brs), 8.46 (1H, brs). Triethylamine (3.8 ml) was added to a suspension of the compound synthesized in Reference Example 3-3 (3.27 g), 2-bromoctanal (2.5 ml) in THF (80 ml) and DMF (15 ml) at 0 ° C. The mixture was added dropwise and stirred at room temperature for 1 hour. Ethyl acetate (100 ml) was added, and the organic layer was washed sequentially with 0.05 M hydrochloric acid, water, and saturated saline, and then dried over magnesium sulfate. The residue obtained by distilling off the solvent was subjected to silica gel column chromatography. From the hexane: ethyl acetate (1: 2) elution part, in the formula (XX), R 2 was a 2- (methoxycarbonyl) ethyl group, hexyl group R 5 is the n-, R 6 is a hydrogen atom, Z is give CO, pale brown oil of the compound R 15 is phenyl group and (3.13 g). This was dissolved in toluene (50 ml), acetic anhydride (0.6 ml) and camphorsulfonic acid (0.9 g) were added, and the mixture was stirred for 4 hours at T. After cooling to room temperature, ethyl acetate (100 ml) was added to the reaction solution. In addition, the organic layer was washed with a saturated aqueous solution of sodium carbonate and saturated saline, dried over magnesium sulfate, and the solvent was distilled off.The residue obtained was subjected to silica gel column chromatography, and hexane: ethyl acetate (hexane) was added. 2: 1) The title compound (1.08 g, 46%) was obtained from the eluate. One NMR (300 MHz, CDC1 3) : 60.90 (3H, t, J = 6.9Hz), 1.25-1.36 (6H, m), 1.43-1.53 (2H, m), 2.05 (2H, t, J = 7.1Hz ), 2.74 (2H, t, J = 7.5Hz), 3.23 (2H, t, J = 7.5Hz), 3.73 (3H, s), 6.50 (1H, brs), 8.46 (1H, brs).
実施例 12-2 Example 12-2
式 (XXV) において R2がべンジル基、 R5が n-へキシル基、 R6が水素原子で ある化合物の合成 Synthesis of a compound of the formula (XXV) wherein R 2 is a benzyl group, R 5 is an n-hexyl group, and R 6 is a hydrogen atom
実施例 10-1で合成した化合物(1.53g)の THF (15ml)とメ'タノール(5ml)混合溶液 に水酸化力リウム(1.2g)のメタノ一ル(10ml)溶液を滴下し 30分間撹拌後、ベンジ ルブロミド(0.59ml)を滴下し 3時間撹拌した。反応液に 0.1M塩酸(40ml)を加え酢 酸ェチルで抽出後、 抽出液を水と飽和食塩水で洗浄し硫酸マグネシウムで乾燥し た。 溶媒を留去して表題化合物(1.54g, 99%)を得た。 To a mixed solution of the compound synthesized in Example 10-1 (1.53 g) in THF (15 ml) and methanol (5 ml) was added dropwise a solution of potassium hydroxide (1.2 g) in methanol (10 ml), and the mixture was stirred for 30 minutes. Thereafter, benzyl bromide (0.59 ml) was added dropwise and stirred for 3 hours. After adding 0.1 M hydrochloric acid (40 ml) to the reaction solution and extracting with ethyl acetate, the extract was washed with water and saturated saline, and dried over magnesium sulfate. The solvent was distilled off to obtain the title compound (1.54 g, 99%).
¾-NMR (300 MHz, CDC13) : d 0.90 (3H, t, J=6.9Hz), 1.24-1.51 (8H, m), 2.03 (2H, t, J=7.1Hz), 4.22 (2H, s), 6.47 (1H, s), 7.24-7.40 (5H, m), 8.30 (1H, br s) . ¾-NMR (300 MHz, CDC1 3): d 0.90 (3H, t, J = 6.9Hz), 1.24-1.51 (8H, m), 2.03 (2H, t, J = 7.1Hz), 4.22 (2H, s ), 6.47 (1H, s), 7.24-7.40 (5H, m), 8.30 (1H, br s).
以下に実施例 12-1および 12-2で合成した化合物を示す。 The compounds synthesized in Examples 12-1 and 12-2 are shown below.
〔表 1 0 0〕(Table 100)
差眷ぇ用弒 (規則 26) 実施例 13-1 Substitution (Rule 26) Example 13-1
式(XXV I)において R2がべンジル基、 R5が n-へキシル基、 R6が水素原子、 R 14が t-ブ卜キシカルボニルメチル基である化合物の合成 Synthesis of a compound of the formula (XXVI) wherein R 2 is a benzyl group, R 5 is an n-hexyl group, R 6 is a hydrogen atom, and R 14 is a t-butoxycarbonylmethyl group
実施例 12-2で合成した化合物(51. Omg)の DMF (lml)溶液にブロモ酢酸卜ブチル エステル (0.024ml)と炭酸力リゥム(37.5 mg)を加え 21時間撹拌した。酢酸ェチル で希釈し、 有機溶媒を 0.05M塩酸と飽和食塩水で洗浄後、 硫酸マグネシウムで乾 燥した。 溶媒を留去しで得られる残留物をシリカゲル力ラムクロマトグラフィー に付し、へキサン:酢酸ェチル(2: 1)溶出部より表題化合物(64.2mg, 97%)を得た。 'H-NMR (300 MHz, CDC13) : 50.91 (3H, i, J=7.1Hz), 1.20-1.55 (8H, ) , 1.46 (9H, s), 2.00-2.07 (2H, m), 4.19 (2H, s), 4.22 (2H, s), 6.50 (1H, br s), 7.20-7.40 (5H, m). To a solution of the compound synthesized in Example 12-2 (51. Omg) in DMF (1 ml) was added tributyl bromoacetate (0.024 ml) and carbonated carbonate (37.5 mg), and the mixture was stirred for 21 hours. After dilution with ethyl acetate, the organic solvent was washed with 0.05M hydrochloric acid and saturated saline, and then dried over magnesium sulfate. The residue obtained by distilling off the solvent was subjected to silica gel column chromatography, and the title compound (64.2 mg, 97%) was obtained from a fraction eluted with hexane: ethyl acetate (2: 1). 'H-NMR (300 MHz, CDC1 3): 50.91 (3H, i, J = 7.1Hz), 1.20-1.55 (8H,), 1.46 (9H, s), 2.00-2.07 (2H, m), 4.19 ( 2H, s), 4.22 (2H, s), 6.50 (1H, br s), 7.20-7.40 (5H, m).
実施例 13-2 Example 13-2
式 (XXV I )において R 2がべンジル基、 R 5が n-へキシル基、 R 6が水素原子、 R 14がカルボキシメチル基である化合物の合成 Synthesis of a compound of the formula (XXV I) wherein R 2 is a benzyl group, R 5 is an n-hexyl group, R 6 is a hydrogen atom, and R 14 is a carboxymethyl group
実施例 13-1 で合成した化合物(32. lmg)にトリフルォロ酢酸(0.5ml)を加え 15 分間撹拌した。エーテルを加え 1N水酸化ナトリウム水溶液で pH4とし、有機層を 1N水酸化ナトリゥム水溶液(10ml)で抽出した。合せた水層を 1N塩酸で pH2とし、 酢酸ェチルで抽出した。 抽出液を硫酸マグネシウムで乾燥後、 溶媒を留去して表 題化合物(7.5mg, 26 )を た。 . Trifluoroacetic acid (0.5 ml) was added to the compound (32.1 mg) synthesized in Example 13-1, and the mixture was stirred for 15 minutes. Ether was added to adjust the pH to 4 with a 1N aqueous sodium hydroxide solution, and the organic layer was extracted with a 1N aqueous sodium hydroxide solution (10 ml). The combined aqueous layer was adjusted to pH 2 with 1N hydrochloric acid and extracted with ethyl acetate. After the extract was dried over magnesium sulfate, the solvent was distilled off to obtain the title compound (7.5 mg, 26). .
Ή-NMR (300 MHz, CDC13) : δ 0.91 (3Η, t, J-6.9Hz), 1.00-1.80. (8H, m), 2.00-2.09 (2H, m), 4.23 (2H, s), 4.35 (2H, s), 6.52 (1H, brs), 7.23-7.39 (5H, m) . 実施例 13 - 3 Ή-NMR (300 MHz, CDC1 3):. Δ 0.91 (3Η, t, J-6.9Hz), 1.00-1.80 (8H, m), 2.00-2.09 (2H, m), 4.23 (2H, s), 4.35 (2H, s), 6.52 (1H, brs), 7.23-7.39 (5H, m). Example 13-3
式(XXV I)において R2がべンジル基、 R5が n-へキシル基、 R6が水素原子、 R 14が 4-メトキシベンジル基である化合物の合成 Synthesis of a compound of the formula (XXVI) wherein R 2 is a benzyl group, R 5 is an n-hexyl group, R 6 is a hydrogen atom, and R 14 is a 4-methoxybenzyl group
実施例 12-2で合成した化合物(30.5mg)の DMF (0.6ml)溶液に 4-メトキシベンジ ルクロリド(0.017ml)と炭酸カリウム(22.5mg)を加え 19.5時間撹拌した。 エーテ ルと水 (各 2ml)を加え 1時間撹拌後、 0.05M塩酸(lml)を加え酢酸ェチルで抽出し た。 抽出液を飽和食塩水で洗浄後、 硫酸マグネシウムで乾燥した。 溶媒を留去し て得られる残留物をシリカゲルカラムクロマトグラフィーに付し、 へキサン:酢 酸ェチル(6: 1)溶出部より表題化合物(26.3mg, 67%)を得た。 To a solution of the compound (30.5 mg) synthesized in Example 12-2 in DMF (0.6 ml) was added 4-methoxybenzyl chloride (0.017 ml) and potassium carbonate (22.5 mg), and the mixture was stirred for 19.5 hours. Ether and water (2 ml each) were added, and the mixture was stirred for 1 hour, added with 0.05 M hydrochloric acid (1 ml), and extracted with ethyl acetate. The extract was washed with saturated saline and dried over magnesium sulfate. Evaporate the solvent The resulting residue was subjected to silica gel column chromatography to give the title compound (26.3 mg, 67%) from a fraction eluted with hexane: ethyl acetate (6: 1).
'H-N R (300 MHz, CDC13) : (50.89 (3H, t, J=6.9Hz), 1.20-1.50 (8H, m), 1.96-2.06 (2H, m), 3.78 (3H, s), 4.20 (2H, s), 4.63 (2H, s), 6.46 (1H, br s), 6.82 (2H, d, J=8.4Hz), 7.24-7.38 (7H, m) . 'HN R (300 MHz, CDC1 3): (50.89 (3H, t, J = 6.9Hz), 1.20-1.50 (8H, m), 1.96-2.06 (2H, m), 3.78 (3H, s), 4.20 (2H, s), 4.63 (2H, s), 6.46 (1H, br s), 6.82 (2H, d, J = 8.4Hz), 7.24-7.38 (7H, m).
実施例 13-4 Example 13-4
式(XXV I)において R2がべンジル基、 R5が n-へキシル基、 R6が水素原子、 R 14が P-トルエンスルホニル基である化合物の合成 Synthesis of a compound of the formula (XXVI) wherein R 2 is a benzyl group, R 5 is an n-hexyl group, R 6 is a hydrogen atom, and R 14 is a P-toluenesulfonyl group
実施例 12-2で合成した化合物(31.9mg)のジクロロメタン( 1 1 )溶液に p-トルェ ンスルホニルクロリド(17. lmg)とトリエチルァミン(0.0143ml)を加え 15.5 時間 攪拌した。 水を加え酢酸ェチルで抽出し、 抽出液を飽和食塩水で洗浄し硫酸マグ ネシゥムで乾燥した。 溶媒を留去して得られる残留物をろ取し、 へキサンで洗浄 して表題化合物 (26. Omg, 61%)を得た。 To a solution of the compound (31.9 mg) synthesized in Example 12-2 in dichloromethane (11), p-toluenesulfonyl chloride (17.lmg) and triethylamine (0.0143 ml) were added, and the mixture was stirred for 15.5 hours. Water was added and the mixture was extracted with ethyl acetate. The extract was washed with saturated saline and dried over magnesium sulfate. The residue obtained by distilling off the solvent was collected by filtration and washed with hexane to obtain the title compound (26. Omg, 61%).
¾-NMR (300 MHz, CDC13) : 50.89 (3H, t, J=6.9Hz), 1.22-1.48 (8H, m), 2.00 (2H, t, J-7.2Hz), 2.44 (3H, s), 4.20 (2H, s), 6.42 (1H, s), 7.27-7.37 (7H, m), 8.03 (2H, d, J=8.3Hz) . ¾-NMR (300 MHz, CDC1 3): 50.89 (3H, t, J = 6.9Hz), 1.22-1.48 (8H, m), 2.00 (2H, t, J-7.2Hz), 2.44 (3H, s) , 4.20 (2H, s), 6.42 (1H, s), 7.27-7.37 (7H, m), 8.03 (2H, d, J = 8.3Hz).
実施例 13 - 5 Example 13-5
式(XXV I)において R 2がべンジル基、 R 5が n -へキシル基、 R 6が水素原子、 R 14が n -プチルスルホニル基である化合物の合成 Synthesis of a compound of the formula (XXVI) wherein R 2 is a benzyl group, R 5 is an n-hexyl group, R 6 is a hydrogen atom, and R 14 is an n-butylsulfonyl group
実施例 12-2 で合成した化合物(22.4mg)と 1 -ブ夕ンスルホニルク口リ ド (0.009ml)から実施例 13-4と同様にして表題化合物を得た。 収率 75%。 The title compound was obtained in the same manner as in Example 13-4 from the compound synthesized in Example 12-2 (22.4 mg) and 1-butanesulfonyl chloride (0.009 ml). Yield 75%.
[H-NMR (200 MHz, CDC13) : 50.91 (3H, t, J=7.0Hz), 0.95 (3H, t, J=7.3Hz), 1.25-1.55 (譲, i), 1.79-1.91 (2H, m), 2.01-2.10 (2H, m), 3.46-3.54 (2H, m), 4.24 (2H, s), 6.49 (1H, s), 7.27-7.40 (5H, m). [H-NMR (200 MHz, CDC1 3): 50.91 (3H, t, J = 7.0Hz), 0.95 (3H, t, J = 7.3Hz), 1.25-1.55 ( Yuzuru, i), 1.79-1.91 (2H , m), 2.01-2.10 (2H, m), 3.46-3.54 (2H, m), 4.24 (2H, s), 6.49 (1H, s), 7.27-7.40 (5H, m).
実施例 13 - 6 Example 13-6
式(XXV I)において R 2がべンジル基、 R 5が n-へキシル基、 R 6が水素原子、 R 14がジメチルァミノメチル基である化合物の合成 Synthesis of a compound of the formula (XXVI) wherein R 2 is a benzyl group, R 5 is an n-hexyl group, R 6 is a hydrogen atom, and R 14 is a dimethylaminomethyl group
実施例 12-2で合成した化合物(29.8mg)と 2-クロロェチルジメチルァミン塩酸 塩 (11.5nig)から実施例 13-3と同様にして表題化合物を得た。 収率 53%。 'H-NMR (300 MHz, CDC13) : 50.90 (3H, t, J-6.9Hz), 1.24-1.38 (8H, m), 1.39-1.51 (2H, m), 2.03 (2H, t, J=7.0Hz), 2.26 (6H, s), 2.54 (2H, t, J=6.7Hz), 3.67 (2H, t, J=6.7Hz), 4.21 (2H, s), 6.49 (1H, s), 7.24-7.40 (5H, m). The title compound was obtained from the compound (29.8 mg) synthesized in Example 12-2 and 2-chloroethyldimethylamine hydrochloride (11.5 nig) in the same manner as in Example 13-3. Yield 53%. 'H-NMR (300 MHz, CDC1 3): 50.90 (3H, t, J-6.9Hz), 1.24-1.38 (8H, m), 1.39-1.51 (2H, m), 2.03 (2H, t, J = 7.0Hz), 2.26 (6H, s), 2.54 (2H, t, J = 6.7Hz), 3.67 (2H, t, J = 6.7Hz), 4.21 (2H, s), 6.49 (1H, s), 7.24 -7.40 (5H, m).
以下に実施例 13- 1〜13- 6 で合成した化合物および同様にして合成した化合物 の一覧を示す。 The following is a list of compounds synthesized in Examples 13-1 to 13-6 and compounds synthesized in the same manner.
〔表 1 0 1〕(Table 101)
蘧眷ぇ用紙 »U26) 〔表 1 .0 2〕Custom paper »U26) (Table 1.02)
差替え用紙(規則 26) 以下に実施例 13-7〜13- 22で合成した化合物のデータを示す。 Replacement form (Rule 26) The data of the compounds synthesized in Examples 13-7 to 13-22 are shown below.
実施例 13-7 Example 13-7
Ή-NMR (CDC13) : (50.85-0.93 (6H, m), 1.20-1.70 (14H, m), 2.00-2.07 (2H, m), 3.53 (2H, t, J=7.3Hz), 4.21 (2H, s), 6.50 (1H, br s), 7.22-7.40 (5H, m) . 実施例 13-8 Ή-NMR (CDC1 3): (50.85-0.93 (6H, m), 1.20-1.70 (14H, m), 2.00-2.07 (2H, m), 3.53 (2H, t, J = 7.3Hz), 4.21 ( 2H, s), 6.50 (1H, br s), 7.22-7.40 (5H, m).
-匪 R (CDC13) : 50.87 (3H, t, J=6.8Hz), 1.20-1.47 (8H, m), 2.11 (2H, t, J=6.7Hz), 3.97 (2H, s), 4.29 (2H, s), 6.66 (1H, s), 7.20-7.43 (6H, m), 7.60 (1H, br s). -Marauder R (CDC1 3 ): 50.87 (3H, t, J = 6.8Hz), 1.20-1.47 (8H, m), 2.11 (2H, t, J = 6.7Hz), 3.97 (2H, s), 4.29 ( 2H, s), 6.66 (1H, s), 7.20-7.43 (6H, m), 7.60 (1H, br s).
実施例 13 - 9 Example 13-9
H-NMR (CDC13) : δ 0.90 (3H, t, J=6.8Hz), 1.10-1.50 (8H, m) , 2.03 (2H, t, J=7.0Hz), 4.21 (2H, s), 4.63 (2H, s), 6.48 (1H, s), 7.20-7.50 (9H, m) . 実施例 13-10 H-NMR (CDC1 3): δ 0.90 (3H, t, J = 6.8Hz), 1.10-1.50 (8H, m), 2.03 (2H, t, J = 7.0Hz), 4.21 (2H, s), 4.63 (2H, s), 6.48 (1H, s), 7.20-7.50 (9H, m) .Example 13-10
'Η—醒 R (CDCI3) : 50.90 (3H, t, J-7.0Hz), 1.22-1.35 (6H, m), 1.24 (3H, t, J=7.2Hz), 1.38-1.50 (2H, m), 2.04 (2H, t, J=7.7Hz), 2.65 (2H, t, J=7.2Hz), 3.85 (2H, t, J=7.2Hz), 4.13 (2H, q, J=7.2Hz), 4.21 (2H, s), 6.49 (1H, s), 7.25-7.40 ,(5H, m). 'Η—Awake R (CDCI3): 50.90 (3H, t, J-7.0Hz), 1.22-1.35 (6H, m), 1.24 (3H, t, J = 7.2Hz), 1.38-1.50 (2H, m) , 2.04 (2H, t, J = 7.7Hz), 2.65 (2H, t, J = 7.2Hz), 3.85 (2H, t, J = 7.2Hz), 4.13 (2H, q, J = 7.2Hz), 4.21 (2H, s), 6.49 (1H, s), 7.25-7.40, (5H, m).
実施例 13 - 11 Examples 13-11
¾-NMR (CDCI3) : ά 0.90 (3H, t, J=6.1Hz), 1.38 (3H, t, J=7.1Hz), 1.25-1.50 (8H, m), 2.03 (2H, t, J=7.6Hz), 4.21 (2H, 's), 4.36 (2H, q, J=7.1Hz), 4.74 (2H, s), 6.49 (1H, s), 7.27-7.39 (5H, m), 7.44 (2H, d, J=7.3Hz), 7.98 (2H, d, J=7.3Hz). ¾-NMR (CDCI3): ά 0.90 (3H, t, J = 6.1Hz), 1.38 (3H, t, J = 7.1Hz), 1.25-1.50 (8H, m), 2.03 (2H, t, J = 7.6) Hz), 4.21 (2H, 's), 4.36 (2H, q, J = 7.1Hz), 4.74 (2H, s), 6.49 (1H, s), 7.27-7.39 (5H, m), 7.44 (2H, d, J = 7.3Hz), 7.98 (2H, d, J = 7.3Hz).
実施例 13-12 Example 13-12
'H—NMR (CDCI3) : 50.90 (3H, t, J=6.9Hz), 1.25 (3H, t, J=7.1Hz), 1.27-1.37 (6H, m), 1.41-1.50 (2H, m), 1.93 (1H, tt, J=7.5, 6.8Hz), 1.96 (1H, tt, J=7.5, 6.8Hz), 2.04 (2H, t, J=7.1Hz), 2.31 (2H, t, J=7.5Hz), 3.61 (2H, t, J-6.8Hz), 4.13 (2H, q, J=7.1Hz), 4.22 (2H, s), 6.49 (1H, s), 7.25-7.40 (5H, m) · 実施例 13-13 'H-NMR (CDCI3): 50.90 (3H, t, J = 6.9Hz), 1.25 (3H, t, J = 7.1Hz), 1.27-1.37 (6H, m), 1.41-1.50 (2H, m), 1.93 (1H, tt, J = 7.5, 6.8Hz), 1.96 (1H, tt, J = 7.5, 6.8Hz), 2.04 (2H, t, J = 7.1Hz), 2.31 (2H, t, J = 7.5Hz) ), 3.61 (2H, t, J-6.8Hz), 4.13 (2H, q, J = 7.1Hz), 4.22 (2H, s), 6.49 (1H, s), 7.25-7.40 (5H, m) Example 13-13
'H—NMR (CDCI3) : δ 0.90 (3H, t, J=6.9Hz), 1.24 (3H, t, J=7.1Hz), 1.28-1.37 (6H, m), 1.39-1.49 (2H, m), 1.60-1.70 (4H, m), 2.04 (2H, t, J=7.0Hz), 2.32 '(s 'HI) 6Γ9 '(s 'Η2) Π ^ '(s Ή8) O S '(WZ^f ) 90 "2 ' 'Μ) ·Ι ' (HI 'Η9) 98 -9 ΐ '(¾8·9=ί Ί 'Η2) W09: (sIOaO) ¾Μ-ΗΓ 'H-NMR (CDCI3): δ 0.90 (3H, t, J = 6.9Hz), 1.24 (3H, t, J = 7.1Hz), 1.28-1.37 (6H, m), 1.39-1.49 (2H, m) , 1.60-1.70 (4H, m), 2.04 (2H, t, J = 7.0Hz), 2.32 '(s' HI) 6Γ9' ( s ' Η2) Π ^ '( s Ή8) OS' (WZ ^ f) 90 "2 '' Μ) Μ Ι '(HI' Η9) 98 -9 ΐ '(¾8 9 = ί Ί 'Η2) W09: ( s IOaO) ¾Μ-Η Γ
0Ζ-8Ϊ圏章 •(m ¾9) Of '(s ¾I) IS'9 0Ζ-8ΪChapter • (m ¾9) Of '(s ¾I) IS'9
'(s ) 82 '( W ' (s Jq 'Ηΐ) 89 '( Γ ί Ί 'ΗΖ) 90 '(ra '(s) 82' (W '(s Jq' Ηΐ) 89 '(Γ ί Ί' ΗΖ) 90 '( ra
'ΕΖ) Z9'i-Ifl '(«ι ¾8) 9ΓΪ- Ϊ '(¾ίΓ9=ί '1 'Η8) 06·0ί : (ε1303) 漏-'ΕΖ) Z9'i-Ifl' («ι ¾8) 9ΓΪ- Ϊ '(¾ίΓ9 = ί' 1 'Η8) 06 · 0ί: ( ε 1303) Leak-
6ΐ-π画 6ΐ-π drawing
•(in ) K'S-LZ' '(in ε) S 'L-lfL '(s 'Ηΐ) Z9 ·9 ' (s ¾C) 's ) SI £ (UI(In) K'S-LZ '' (in ε) S 'L-lfL'(s' Ηΐ) Z9 9 '(s ¾C)' s ) SI £ (UI
W L5 -l-5fl '(in ¾9) ト ·ΐ '( 8·9=ί Ί Ήδ) ΐ6·0ί> : (εΙ3αθ) 漏- ΟΖ W L5 -l-5fl '(in ¾9) G ΐ (' (89 = ί Ί Ήδ) ΐ6.0ΐ>: ( ε Ι3αθ) Leakage-ΟΖ
•(in ¾9) Of Z-Sr '(s Ήΐ) 09 -9 '(s ) ZZ ' 'L=i Ί 'HZ),98'S '(ΖΗΓΑ=Γ Ί ' Ζ) li 'l '( 'L=i (In ¾9) Of Z-Sr '(s Ήΐ) 09 -9' (s) ZZ '' L = i Ί 'HZ), 98'S' (ΖΗΓΑ = Γ Ί 'Ζ) li' l '(' L = i
¾z) ·ζ '(in '腿) os'i - on '(zH8-9=r Ή8) ο6·ορ : ('ιοαο) 漏- ¾z) ζ ζ '(in' thigh) os'i-on '(zH8-9 = r Ή8) ο6 ・ ορ: (' ιοαο)
■(in ¾S) QfL-\Z-L '(s 'Ηΐ) 6f '9 '(s Ή ) U 'f '(WL=i 'P ) A8'S '(ZHl 'Z=f '1 'IK) fO ]Z '(m ¾S) 8A't-SS'l '(in ' ) OS Ί - O I '(ΰΐ 'ΗΟΙ) '( ¾S) A6 ·0 - 68 '0 ρ : (SICK ) Ht¾N-H, ■ (in ¾S) QfL- \ ZL '(s' Ηΐ) 6f' 9 '(s Ή) U' f '(WL = i' P) A8'S '(ZHl' Z = f '1' IK) fO] Z '(m ¾S) 8A't-SS'l' (in ') OS Ί-OI' (ΰΐ 'ΗΟΙ)' (¾S) A6 0-68 '0 ρ: ( S ICK) Ht¾N-H,
91 - SI P 敏牽 91-Sip Toshiki
•(in ¾9) QfL-^Z'L '(s ¾l) 09 '9 '(s 'ΕΖ) ΙΖ ' { '9=Γ '1 'ΕΖ) Οΐ ·ε Ί 'ΗΖ) 88 '(ΖΗ「 =ί '1 'ΗΖ) fO'Z '(ω ¾f) Wi_SS'I '(ω 'ΕΖ) ZS't-Of Ί '(πι 'Η9) 68*l- Z'l '(ζΗ8·9=ί Ί 'Η8) 06 Ό Ρ: ('ΙΟΟΟ) Ν-Η, ΐ-2ΐ m • (in ¾9) QfL- ^ Z'L '(s ¾l) 09' 9 '(s' ΕΖ) ΙΖ' ('' 9 = Γ '1' ΕΖ) Οΐ · ε Ί 'ΗΖ) 88' (ΖΗ “= ί '1' ΗΖ) fO'Z '(ω ¾f) Wi_SS'I' (ω 'ΕΖ) ZS't-Of Ί' (πι 'Η9) 68 * l- Z'l' (ζΗ8 · 9 = ί Ί 'Η8) 06 Ό Ρ: (' ΙΟΟΟ) Ν-Η, ΐ-2ΐ m
'(ιπ ) '(ιπ)
^■1-11·1 '(s 'Ηΐ) '(s 'Μ) 8f '(s 'ΗΖ) '(ΖΗΓ ί ' 'ΗΖ) ίΟ '(ω S 'Μ) Ζ9·ΐ-0^*ΐ £(ιπ 'Η9) 6Γΐ - SZ'I '(ΖΗ6·9=ί 'Ηδ) Ϊ6·0Ρ : (εΐ3α3) 画- ^ ■ 1-111 '(s'Ηΐ)'(s' Μ) 8f '(s'ΗΖ)' (ΖΗΓ ί '' ΗΖ) ίΟ '(ω S' Μ) Ζ9ΐ ΐ-0 ^ * ΐ £ (ιπ 'Η9) 6Γΐ-SZ'I' (ΖΗ6 · 9 = ί 'Ηδ) Ϊ6.0 ·: ( ε ΐ3α3) picture-
•(in ¾S) ^·ί-Π-ί '(s 'Ηΐ) 6 9 '(s 'ΗΖ) ΐΖ· '(ΖΗΓΑ=ί 'b 'ΗΖ) Wf '(ΖΗΓ9=1 ) 99 Έ 'W9=r Ί 'ΗΖ) (In ¾S) ^ · ί-Π-ί '(s' Ηΐ) 6 9' (s' ΗΖ) ΐΖ '(ΖΗΓΑ = ί' b 'ΗΖ) Wf' (ΖΗΓ9 = 1) 99 Έ 'W9 = r Ί 'ΗΖ)
68ム 68 mu
0t9t0/Z0dr/lDd 909Ζ60/Ζ0 OAV (5H, m). 0t9t0 / Z0dr / lDd 909Ζ60 / Ζ0 OAV (5H, m).
実施例 13 - 21 Examples 13-21
lH - NMR (CDC13) : δ 0.89 (3Η, t, J=6.9Hz), 1.24-1.48 (8H, m), 2.01 (2H, br t, J=7.5Hz), 2.30 (3H, s), 2.67 (6H, s), 4.20 (2H, s), 6.42 (1H, br s), 6.97 (2H, s), 7.28-7.34 (5H, m). lH - NMR (CDC1 3): δ 0.89 (3Η, t, J = 6.9Hz), 1.24-1.48 (8H, m), 2.01 (2H, br t, J = 7.5Hz), 2.30 (3H, s), 2.67 (6H, s), 4.20 (2H, s), 6.42 (1H, br s), 6.97 (2H, s), 7.28-7.34 (5H, m).
実施例 13-22 Example 13-22
—匪 R (CDCI3) : 50.90 (3H, t, J=6.9Hz), 1.23-1.51 (8H, ), 2.04 (2H, dt, J=7.0, 1.1Hz), 2.64 (1H, dd, J=4.5, 2.5Hz), 2.77 (1H, dd, J=4.5, 4.2Hz) , 3.13-3.20 (1H, m), 3.61 (1H, dd, J=14.3, 5.3Hz), 3.84 (1H, dd, J=14.3, 5.0Hz), 4.22 (2H, s), 6.51 (1H, s), 7.27-7.40 (5H, m). — Bandit R (CDCI3): 50.90 (3H, t, J = 6.9Hz), 1.23-1.51 (8H,), 2.04 (2H, dt, J = 7.0, 1.1Hz), 2.64 (1H, dd, J = 4.5 , 2.5Hz), 2.77 (1H, dd, J = 4.5, 4.2Hz), 3.13-3.20 (1H, m), 3.61 (1H, dd, J = 14.3, 5.3Hz), 3.84 (1H, dd, J = 14.3, 5.0Hz), 4.22 (2H, s), 6.51 (1H, s), 7.27-7.40 (5H, m).
実施例 14-1 Example 14-1
式 (XXX)において R 5がエトキジカルポニル基、 R 6がメチル基、 R14が tert - ブトキシカルボエルメチル基、 Rがメチル基である化合物の合成 In the formula (XXX), synthesis of a compound wherein R 5 is an ethoxydicarbonyl group, R 6 is a methyl group, R 14 is a tert-butoxycarboylmethyl group, and R is a methyl group
参考例 14で合成した化合物 (2.07 g, 5.50 腿 ol) を THF (45 ml) に溶解し、 2-クロロアセト酢酸ェチル(0.84ml, 6.05mmol) およびトリェチルァミン (1.91 ml, 13.7ramol) を加えて室温で 1時間撹拌した。 反応液を濃縮後、 残渣を酢酸ェ チル (130 ml) で希釈し、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥 した。 溶媒を減圧下に溜去し、残渣をシリカゲル 60 (2 5 ) のカラムクロマト グラフィ—に付し、 酢酸ェチル:へキサン (3 0 : 7 0) の溶出画分を減圧下濃 縮乾固し、 標題化合物の黄色油状物 (2.57g, 5.09mmol) を四種異性体の混合物 として得た。 ' The compound synthesized in Reference Example 14 (2.07 g, 5.50 tmol) was dissolved in THF (45 ml), and ethyl 2-chloroacetoacetate (0.84 ml, 6.05 mmol) and triethylamine (1.91 ml, 13.7 ramol) were added. For 1 hour. After concentration of the reaction solution, the residue was diluted with ethyl acetate (130 ml), washed with water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, and the residue was subjected to silica gel 60 (25) column chromatography. The eluted fraction of ethyl acetate: hexane (30:70) was concentrated under reduced pressure to dryness. A yellow oil (2.57 g, 5.09 mmol) of the title compound was obtained as a mixture of four isomers. '
-麵 R (300 MHz, 重クロ口ホルム中) : 6 1.27/1.30 (3Η, t, J= 7.1 Hz), 1.47 (9H, s), 1.84/2.04 (3H, s), 2.69/2.71 (2H, t, J= 7.3 Hz), 3.05-3.35 (2H, m), 3.70/3.70 (3H, s), 3.76/3.90 (1H, s), 4.19/4.19 (2H, s). 4.20/4.24 (2H, q, J= 7.1 Hz), 4.77/6.00 (1H, s/brs). -麵 R (300 MHz, in double-holed form): 6 1.27 / 1.30 (3Η, t, J = 7.1 Hz), 1.47 (9H, s), 1.84 / 2.04 (3H, s), 2.69 / 2.71 (2H , T, J = 7.3 Hz), 3.05-3.35 (2H, m), 3.70 / 3.70 (3H, s), 3.76 / 3.90 (1H, s), 4.19 / 4.19 (2H, s). 4.20 / 4.24 (2H , q, J = 7.1 Hz), 4.77 / 6.00 (1H, s / brs).
元素分析値: C2。H28N209S2 として計算値: C, 47.61; H, 5.59; N, 5.55; S, 12.71 (%)、 実測値; C, 47.65; H, 5.66; N, 5.47; S, 12.61 (%) Elemental analysis: C 2. Calculated for H 28 N 2 0 9 S 2 : C, 47.61; H, 5.59; N, 5.55; S, 12.71 (%), found; C, 47.65; H, 5.66; N, 5.47; S, 12.61 ( %)
以下に実施例 14-1 で合成した化合物および同様にして合成した化合物の一覧 を示す。 〔表 1 0 3〕The following is a list of compounds synthesized in Example 14-1 and compounds synthesized in the same manner. (Table 103)
差替え用紙(規則 26) 以下に実施例 14-2〜14 - 10で合成した化合物のデータを示す。 Replacement form (Rule 26) The data of the compounds synthesized in Examples 14-2 to 14-10 are shown below.
実施例 14-2 Example 14-2
(Η-醒 R (300 MHz, 重クロ口ホルム中) : δ 0.90 (3Η, t, J= 6.9 Hz), 0.93 (3H, ί, J= 7.3 Hz), 1.30 (8H, brs), 1.37 (2H, brq, J= 7.4 Hz), 1.48 (2H, m), 1.62 (2H, quint, J= 7.0 Hz), 1.72 (1H, m), 1.81 (1H, m), 2.73 (2H, t, J= 7.3 Hz), 3.23 (1H, dt, J= 13.8, 7.1 Hz), 3.27 (1H, m), 3.33 (1H, dt, J= 13.7, 7.3 Hz), 3.54 (1H, brd, J= 8.0 Hz), 3.71 (3H, s), 4.16 (2H, t, J= 6.7 Hz), 4.31 (2H, s), 5.85 (1H, brdd, J=7.8, 1.3 Hz). (Η-wake R (300 MHz, in double-mouthed form)): δ 0.90 (3Η, t, J = 6.9 Hz), 0.93 (3H, ί, J = 7.3 Hz), 1.30 (8H, brs), 1.37 ( 2H, brq, J = 7.4 Hz), 1.48 (2H, m), 1.62 (2H, quint, J = 7.0 Hz), 1.72 (1H, m), 1.81 (1H, m), 2.73 (2H, t, J = 7.3 Hz), 3.23 (1H, dt, J = 13.8, 7.1 Hz), 3.27 (1H, m), 3.33 (1H, dt, J = 13.7, 7.3 Hz), 3.54 (1H, brd, J = 8.0 Hz) ), 3.71 (3H, s), 4.16 (2H, t, J = 6.7 Hz), 4.31 (2H, s), 5.85 (1H, brdd, J = 7.8, 1.3 Hz).
元素分析値: C22H34N207S2 として計算値: C, 52.57; H, 6.82; N, 5.57; S, 12.76 (%) 実測値; C, 52.64; H, 6.86; N, 5.63; S, 12.74 (%) . Elemental analysis: C 22 H 34 N 2 0 7 S 2 Calculated: C, 52.57; H, 6.82 ; N, 5.57; S, 12.76 (%) Found; C, 52.64; H, 6.86 ; N, 5.63 ; S, 12.74 (%).
実施例 14-3 Example 14-3
Ή -腿 (300 MHz, 重クロ口ホルム中) : δ 0.93 (3H, t, J- 7.3 Hz), 1.27/1.30 (3H, t, J= 7.1 Hz), 1.37 (2H, sixtet, Jこ 7.5 Hz), 1.63 (2H, quint, J= 7.3 Hz), 1.84/2.05 (3H, s), 2.69/2.72 (2H, t, J= 7.4/7.2 Hz), 3.10-3.35 (2H, m), 3.70/3.71 (3H, s), 3.77/3.89 (1H, s), 4.17 (2H, t, J= 6.7 Hz), 4.24 (2H, q, J= 7.1 Hz), 4. 9/4.30 (2H, s), 4.75/5.96 (1H, s). Ή-Thigh (300 MHz, in double-mouthed form): δ 0.93 (3H, t, J- 7.3 Hz), 1.27 / 1.30 (3H, t, J = 7.1 Hz), 1.37 (2H, sixtet, J 7.5) Hz), 1.63 (2H, quint, J = 7.3 Hz), 1.84 / 2.05 (3H, s), 2.69 / 2.72 (2H, t, J = 7.4 / 7.2 Hz), 3.10-3.35 (2H, m), 3.70 /3.71 (3H, s), 3.77 / 3.89 (1H, s), 4.17 (2H, t, J = 6.7 Hz), 4.24 (2H, q, J = 7.1 Hz), 4.9 / 4.30 (2H, s ), 4.75 / 5.96 (1H, s).
元素分析値: C20H28N2OgS2 として計算値: C, 47.61; H, 5.59; N, 5.55; S, 12.71 (%), 実測値; C, 47.40; H, 5.54; N, 5.62; S, 12.65 (%) . Elemental analysis: C 20 H 28 N 2 O g S 2 Calculated: C, 47.61; H, 5.59 ; N, 5.55; S, 12.71 (%), Found; C, 47.40; H, 5.54 ; N, 5.62; S, 12.65 (%).
実施例 14-4Example 14-4
-NMR (300 MHz, 重クロ口ホルム中) : δ 0.93 (3Η, t, J= 7.4 Hz), 1.37 (2H, sixtet, J= 7.6 Hz), 1.62 (2H, m), 2.74 (2H, t, J= 7.3 Hz), 3.09 (1H, dd, J= 13.2, 1.7 Hz), 3.16 (1H, dd, J= 13.2, 3.0 Hz), 3.24 (1H, dt, J= 13.8, 7.3 Hz), 3.36 (1H, dt, J= 13.8, 7.3 Hz), 3.71 (3H, s), 3.79 (1H, brd, J= 7.1 Hz), 4.17 (2H, 6.7 Hz), 4.32 (2H, s), 6.11 (1H, brddd, J= 7.1, 3.0, 1.7 Hz). 元素分析値: C16H22N207S2 として計算値: C, 45.92; H, 5.30; N, 6.69; S, 15.32 (%)、 実測値; C, 46.02; H, 5.16; N, 7.06; S, 15.12 (%) . -NMR (300 MHz, in heavy-duty form): δ 0.93 (3Η, t, J = 7.4 Hz), 1.37 (2H, sixtet, J = 7.6 Hz), 1.62 (2H, m), 2.74 (2H, t) , J = 7.3 Hz), 3.09 (1H, dd, J = 13.2, 1.7 Hz), 3.16 (1H, dd, J = 13.2, 3.0 Hz), 3.24 (1H, dt, J = 13.8, 7.3 Hz), 3.36 (1H, dt, J = 13.8, 7.3 Hz), 3.71 (3H, s), 3.79 (1H, brd, J = 7.1 Hz), 4.17 (2H, 6.7 Hz), 4.32 (2H, s), 6.11 (1H , brddd, J = 7.1, 3.0, 1.7 Hz). Elemental analysis: Calculated for C 16 H 22 N 2 0 7 S 2 : C, 45.92; H, 5.30; N, 6.69; S, 15.32 (%), Found: C, 46.02; H, 5.16; N, 7.06; S, 15.12 (%).
実施例 14 - 5 Example 14-5
Ή -丽 R (300 MHz, 重クロ口ホルム中) : <5 0.93 (3H, t, J= 7.3 Hz), 1.37 (2H, sixtet, J= 7.6 Hz), 1.63 (2H, quint, J= 7.5 Hz), 1.91 (3H, s), 2.73 (2H, t, J= 7.4 Hz), 3.03 (1H, d, J= 13.3 Hz), 3.17 (1H, d, J= 13.4 Hz), 3.26 (1H, dt, J= 13.6, 7.1 Hz), 3.30 (1H, dt, J= 13.6, 7.2 Hz), 3.71 (3H, s), 4.17 (2H, i, J= 6.7 Hz), 4.28 (2H, s), 5.12 (1H, s). Ή-丽 R (300 MHz, in double-hole form): <5 0.93 (3H, t, J = 7.3 Hz), 1.37 (2H, sixtet, J = 7.6 Hz), 1.63 (2H, quint, J = 7.5) Hz), 1.91 (3H, s), 2.73 (2H, t, J = 7.4 Hz), 3.03 (1H, d, J = 13.3 Hz), 3.17 (1H, d, J = 13.4 Hz), 3.26 (1H, dt, J = 13.6, 7.1 Hz), 3.30 (1H, dt, J = 13.6, 7.2 Hz), 3.71 (3H, s), 4.17 (2H, i, J = 6.7 Hz), 4.28 (2H, s), 5.12 (1H, s).
元素分析値: C17H24N207S2 として計算値: C, 47.21; H, 5.59; N, 6.48; S, 14.83 (%), 実測値; C, 47.08; H, 5.60; N, 6.80; S, 14.47 (%) . Elemental analysis: C 17 H 24 N 2 0 7 S 2 Calculated: C, 47.21; H, 5.59 ; N, 6.48; S, 14.83 (%), Found; C, 47.08; H, 5.60 ; N, 6.80; S, 14.47 (%).
実施例 14 - 6 Example 14-6
¾-NMR (300 MHz, 重クロ口ホルム中) : δ 1.90 (3Η, s), 2.54 (3H, s), 3.03 (1H, d, J= 13.3 Hz), 3.18 (1H, d, J= 13.3 Hz), 3.77 (3H, s), 4.31 (2H, s), 5.08 (1H, s). ¾-NMR (300 MHz, in double-mouthed form): δ 1.90 (3Η, s), 2.54 (3H, s), 3.03 (1H, d, J = 13.3 Hz), 3.18 (1H, d, J = 13.3) Hz), 3.77 (3H, s), 4.31 (2H, s), 5.08 (1H, s).
元素分析値: CHH!2N204S2. H20 として計算値: C, 41.50; H, 4.43; N, 8.80; S, 20.14 (%), 実測値; C, 41.80; H, 4.41; N, 8.72; S, 19.93 (%) . !. Elemental analysis: C H H 2 N 2 0 4 S 2 H 2 0 Calculated: C, 41.50; H, 4.43 ; N, 8.80; S, 20.14 (%), Found; C, 41.80; H , 4.41; N, 8.72; S, 19.93 (%).
実施例 14-7 Example 14-7
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 2.74 (2Η, t, J= 7.3 Hz), 3.05 (1H, dd, J- 13.3, 1.8 Hz), 3.09 (3H, s), 3.15 (1H, dd, J= 13.4, 3.0 Hz), 3.23 (1H, dt, J- 13.9, 7.4 Hz), 3.35 (1H, dt, J= 13.8, 7.3 Hz), 3.66 (1H, brd, J= 7.6 Hz) , 3.71 (3H, s), 6.09 (1H, ddd, J= 7.6, 3.0, 1.9 Hz). Ή-NMR (300 MHz, in double-mouthed form): δ 2.74 (2Η, t, J = 7.3 Hz), 3.05 (1H, dd, J- 13.3, 1.8 Hz), 3.09 (3H, s), 3.15 ( 1H, dd, J = 13.4, 3.0 Hz), 3.23 (1H, dt, J- 13.9, 7.4 Hz), 3.35 (1H, dt, J = 13.8, 7.3 Hz), 3.66 (1H, brd, J = 7.6 Hz) ), 3.71 (3H, s), 6.09 (1H, ddd, J = 7.6, 3.0, 1.9 Hz).
元素分析値: CHH14N205S2 として計算値: C, 41.50; H, 4.43; N, 8.80; S, 20.14Elemental analysis: C H H 14 N 2 0 5 S 2 Calculated: C, 41.50; H, 4.43 ; N, 8.80; S, 20.14
(%), 実測値; C, 41.59; H, 4.30; N, 8.87; S, 20.09 (%) . (%), Found; C, 41.59; H, 4.30; N, 8.87; S, 20.09 (%).
実施例 14 - 8 Example 14-8
Ή-醒 R (300 MHz, 重クロ口ホルム中) : 6 1.29 (3H, t, J= 7.1 Hz), 1.91 (3H, s), 2.73 (2H, t, J= 7.4 Hz), 3.02 (1H, d, J= 13.3 Hz), 3.18 (1H, brd, J- 13.2 Hz), 3.26 (1H, di, J= 13.8, 7.3 Hz), 3.30 (1H, dt, J= 13.8, 7.5 Hz), 3.71 (3H, s), 4.23 (2H, q, J= 7.1 Hz), 4.28 (2H, s), 5.09 (1H, s) . Ή-Awake R (300 MHz, in double-mouthed form): 6 1.29 (3H, t, J = 7.1 Hz), 1.91 (3H, s), 2.73 (2H, t, J = 7.4 Hz), 3.02 (1H) , d, J = 13.3 Hz), 3.18 (1H, brd, J-13.2 Hz), 3.26 (1H, di, J = 13.8, 7.3 Hz), 3.30 (1H, dt, J = 13.8, 7.5 Hz), 3.71 (3H, s), 4.23 (2H, q, J = 7.1 Hz), 4.28 (2H, s), 5.09 (1H, s).
元素分析値: C15H20N207S2 として計算値: C, 44.54; H, 4.98; N, 6.93; S, 15.86 (%), 実測値; C, 44.75; H, 5.07; N, 6.70; S, 15.80 (%) . Elemental analysis: C 15 H 20 N 2 0 7 S 2 Calculated: C, 44.54; H, 4.98 ; N, 6.93; S, 15.86 (%), Found; C, 44.75; H, 5.07 ; N, 6.70; S, 15.80 (%).
実施例 14-9 Example 14-9
lH-NMR (300 MHz, 重クロ口ホルム中) : δ 1.27/1.30 (3Η, t, J= 7.1 Hz), 1.81/2.03 (3H, s), 2.69/2.71 (2H, t, J= 7.5/7.3 Hz), 3.07/3.07 (3H, s), 3.11/3.24 (2H, m), 3.70/3.70 (3H, s), 3.74/3.87 (1H, s), 4.20/4.25 (2H, q, J= 7.1 Hz), 4.66/6.07 (1H, s/brs). lH-NMR (300 MHz, in a double-mouthed form): δ 1.27 / 1.30 (3Η, t, J = 7.1 Hz), 1.81 / 2.03 (3H, s), 2.69 / 2.71 (2H, t, J = 7.5 / 7.3 Hz), 3.07 / 3.07 (3H, s), 3.11 / 3.24 (2H, m), 3.70 / 3.70 (3H, s), 3.74 / 3.87 (1H, s), 4.20 / 4.25 (2H, q, J = 7.1 Hz), 4.66 / 6.07 (1H, s / brs).
実施例 14-10 Example 14-10
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 1.32 (3Η, t, J= 7.1 Hz), 1.47 (9H, s), 2.03 (3H, s), 4.22 (2H, s), 4.29/4.30 (2H, q, J= 7.1 Hz), 4.55 (1H, s), 4.82 (1H, br), 7.64 (1H, s). Ή-NMR (300 MHz, in double-mouthed form): δ 1.32 (3Η, t, J = 7.1 Hz), 1.47 (9H, s), 2.03 (3H, s), 4.22 (2H, s), 4.29 / 4.30 (2H, q, J = 7.1 Hz), 4.55 (1H, s), 4.82 (1H, br), 7.64 (1H, s).
元素分析値: C16H22N207S2 · 0.3H20として計算値: C, 45.34; H, 5.37; N, 6.61; S, 15.13 (%), 実測値; C, 45.35; H, 5.17; , 6.64; S, 15.23 ( ) . Elemental analysis: C 16 H 22 N 2 0 7 S 2 · 0.3 H 2 0 Calculated: C, 45.34; H, 5.37; N, 6.61; S, 15.13 (%), found; C, 45.35; H , 5.17;, 6.64; S, 15.23 ().
実施例 15-1 Example 15-1
式 (XXX I ) において R5がエトキシカルポニル基、 R6がメチル基、 R14が tert-ブ卜キシカルボニルメチル基、 Rがメチル基である化合物の合成 Synthesis of a compound of the formula (XXXI) wherein R 5 is an ethoxycarbonyl group, R 6 is a methyl group, R 14 is a tert-butoxycarbonylmethyl group, and R is a methyl group
実施例 14-1で合成した化合物 (0.75 g, 1.49腿 ol) をトルエン (25 ml) およ び無水酢酸 (0.215ml, 2.23mmol) に溶解し、 D -力ンファ一- 10 -スルホン酸 (173 mg, 0.743腾 ol) を加えて 6 0°Cで 3時間撹拌した。 反応液を酢酸ェチル (80 nil) で希釈し、 水、 2%炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 溶媒を減圧下に溜去し、 残渣をシリカゲル 6 0 (1 Og) のカラムクロマトグラフィ—に付し、 0.1%ギ酸を含む酢酸ェチル:へキサ ン (2 0 : 8 0) の溶出画分を減圧下濃縮乾固し、 標題化合物の赤色油状物 (300 mg, 0.616 画 1) を得た。 The compound synthesized in Example 14-1 (0.75 g, 1.49 t ol) was dissolved in toluene (25 ml) and acetic anhydride (0.215 ml, 2.23 mmol), and D-potassium 10-sulfonic acid ( 173 mg, 0.743 mmol) and stirred at 60 ° C for 3 hours. The reaction solution was diluted with ethyl acetate (80 nil), washed with water, a 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, and dried over sodium sulfate. The solvent was distilled off under reduced pressure, and the residue was subjected to column chromatography on silica gel 60 (1 Og). The eluted fraction of ethyl acetate: hexane (20:80) containing 0.1% formic acid was reduced in pressure. The residue was concentrated to dryness to give a red oily substance (300 mg, 0.616, fraction 1) of the title compound.
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 1.33 (3Η, t, J= 7.1 Hz), 1.47 (9H, s), 2.75 (2H, t, J= 7.3 Hz), 2.77 (3H, s), 3.28 (2H, t, J= 7.3 Hz), 3.72 (3H, s), 4.19 (2H, s). 4.26 (2H, q, J= 7.1 Hz). 'H-NMR (300 MHz, in double-mouthed form): δ 1.33 (3Η, t, J = 7.1 Hz), 1.47 (9H, s), 2.75 (2H, t, J = 7.3 Hz), 2.77 (3H , s), 3.28 (2H, t, J = 7.3 Hz), 3.72 (3H, s), 4.19 (2H, s). 4.26 (2H, q, J = 7.1 Hz).
元素分析値: C2。H26N208S2 として計算値: C, 49.37; H, 5.39; N, 5.76; S, 13.18 (%), 実測値; C, 49.16; H, 5.38; N, 5.47; S, 13.25 (%) Elemental analysis: C 2. H 26 N 2 0 8 S 2 Calculated: C, 49.37; H, 5.39 ; N, 5.76; S, 13.18 (%), Found; C, 49.16; H, 5.38 ; N, 5.47; S, 13.25 ( %)
以下に実施例 15-1 'で合成した化合物および同様にして合成した化合物の一覧 を示す。 〔表 1 0 4〕The following is a list of compounds synthesized in Example 15-1 'and compounds synthesized in the same manner. (Table 104)
麄瞽ぇ用弒 (mm) 以下に実施例 15- 2〜15- 12で合成した化合物のデータを示す。 For Goze (mm) The data of the compounds synthesized in Examples 15-2 to 15-12 are shown below.
実施例 15-2 Example 15-2
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 0.90 (3Η, brt, J= 7.1 Hz), 0.93 (3H, t, J= 7.3 Hz), 1.30 (6H, brs), 1.37 (2H, brq, J- 7.4 Hz), 1.47 (2H, brquint, J- 6.9 Hz), 1.62 (2H, brquint, J= 6.9 Hz), 2.06 (2H, brt, J= 7.1 Hz), 2.74 (2H, t, J= 7.6 Hz), 3.23 (2H, t, J= 7.6 Hz), 3.72 (3H, s), 4.16 (2H, t, J= 6.7 Hz), 4.30 (2H, s), 5.64 (1H, brt, J= 1.0 Hz). Ή-NMR (300 MHz, in double-mouthed form): δ 0.90 (3Η, brt, J = 7.1 Hz), 0.93 (3H, t, J = 7.3 Hz), 1.30 (6H, brs), 1.37 (2H, brq, J- 7.4 Hz), 1.47 (2H, brquint, J- 6.9 Hz), 1.62 (2H, brquint, J = 6.9 Hz), 2.06 (2H, brt, J = 7.1 Hz), 2.74 (2H, t, J = 7.6 Hz), 3.23 (2H, t, J = 7.6 Hz), 3.72 (3H, s), 4.16 (2H, t, J = 6.7 Hz), 4.30 (2H, s), 5.64 (1H, brt, J = 1.0 Hz).
元素分析値: C22H32N206S2,として計算値: C, 54.52; H, 6.66; N, 5.78; S, 13.23 (%)、 実測値; C, 54.65; H, 6.49; N, 5.93; S, 13.20 (%) · Elemental analysis: C 22 H 32 N 2 0 6 S 2, Calculated: C, 54.52; H, 6.66 ; N, 5.78; S, 13.23 (%), Found; C, 54.65; H, 6.49 ; N , 5.93; S, 13.20 (%)
実施例 15 - 3 Example 15-3
Ή-NMR (300 MHz, 重クロ口ホルム中) : 6 0.93 (3H, t, J= 7.4 Hz), 1.33 (3H, t, J= 7.1 Hz), 1.37 (2H, sixtet, J- 7.5 Hz), 1.62 (2H, m), 2.75 (2H, t, J= 7.4 Hz), 2.77 (3H, s), 3.28 (2H, t, J= 7. Hz), 3.72 (3H, s), 4.17 (2H, t, J= 6.7 Hz), 4.27 (2H, q, J- 7.1 Hz), 4.29 (2H, s). Ή-NMR (300 MHz, in double-mouthed form): 6 0.93 (3H, t, J = 7.4 Hz), 1.33 (3H, t, J = 7.1 Hz), 1.37 (2H, sixtet, J- 7.5 Hz) , 1.62 (2H, m), 2.75 (2H, t, J = 7.4 Hz), 2.77 (3H, s), 3.28 (2H, t, J = 7. Hz), 3.72 (3H, s), 4.17 (2H , t, J = 6.7 Hz), 4.27 (2H, q, J- 7.1 Hz), 4.29 (2H, s).
元素分析値: C20H26N208S2 として計算値: C, 49.37; H, 5.39; N, 5.76; S, 13.18 (%)、 実測値; C, 49.45; H, 5.25; N, 5.94; S, 13.19 (%) . Elemental analysis: C 20 H 26 N 2 0 8 S 2 Calculated: C, 49.37; H, 5.39 ; N, 5.76; S, 13.18 (%), Found; C, 49.45; H, 5.25 ; N, 5.94; S, 13.19 (%).
実施例 15-4 Example 15-4
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 0.93 (3Η, ί, J= 7.3 Hz), 1.37 (2H, sixtet, J= 7.6 Hz), 1.63 (2H, quint, J= 7.0 Hz), 2.74 (2H, t, J= 7.4 Hz), 3.21 (2H, t, J= 7.4 Hz), 3.72 (3H, s), 4.16 (2H, t, J- 6.7 Hz), 4.30 (2H, s), 4.98 (1H, d, J= 8.0 Hz), 6.69 (1H, d, J= 8.0 Hz). Ή-NMR (300 MHz, in double-mouthed form): δ 0.93 (3Η, ί, J = 7.3 Hz), 1.37 (2H, sixtet, J = 7.6 Hz), 1.63 (2H, quint, J = 7.0 Hz) , 2.74 (2H, t, J = 7.4 Hz), 3.21 (2H, t, J = 7.4 Hz), 3.72 (3H, s), 4.16 (2H, t, J- 6.7 Hz), 4.30 (2H, s) , 4.98 (1H, d, J = 8.0 Hz), 6.69 (1H, d, J = 8.0 Hz).
元素分析値: C16H20N2O6S2 として計算値: C, 47.99; H, 5.03; N, 7.00; S, 16.01 (%)、 実測値; C, 47.89; H, 5.20; N, 7.00; S, 15.75 (%) . Elemental analysis: C 16 H 20 N 2 O 6 S 2 Calculated: C, 47.99; H, 5.03; N, 7.00; S, 16.01 (%), found; C, 47.89; H, 5.20; N, 7.00; S, 15.75 (%).
実施例 15-5 Example 15-5
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 0.93 (3Η, t, J= 7.3 Hz), 1.37 (2H, sixtet, J= 7.6 Hz), 1.63 (2H, quint, J= 7.1 Hz), 2.28 (3H, d, J= 1.2 Hz), 2.73 (2H, t, J= 7.4 Hz), 3.20 (2H, t, J= 7.4 Hz), 3.72 (3H, s), 4.16 (2H, t, J= 6.7 Hz), 4.28 (2H, s), 4.59 (1H, d, J= 1.2 Hz). Ή-NMR (300 MHz, in double-mouthed form): δ 0.93 (3Η, t, J = 7.3 Hz), 1.37 (2H, sixtet, J = 7.6 Hz), 1.63 (2H, quint, J = 7.1 Hz) , 2.28 (3H, d, J = 1.2 Hz), 2.73 (2H, t, J = 7.4 Hz), 3.20 (2H, t, J = 7.4 Hz), 3.72 (3H, s), 4.16 (2H, t, J = 6.7 Hz), 4.28 (2H, s), 4.59 (1H, d, J = 1.2 Hz).
元素分析値: C17¾2N206S2 として計算値: C, 49.26; H, 5.35; N, 6.76; S, 15.47 (%)、 実測値; C, 49.34; H, 5.25; N, 7.00; S, 15.43 (%) . 実施例 15 - 6 Elemental analysis: C 17と し て2 N 2 0 6 S 2 Calculated: C, 49.26; H, 5.35; N, 6.76; S, 15.47 (%), Found; C, 49.34; H, 5.25; N, 7.00; S, 15.43 (%).
Ή -麵 R (300 MHz, 重クロ口ホルム中) : δ 2.74 (2Η, t, J= 7.3 Hz), 3.08 (3H, s), 3.21 (2H, t, J= 7.3 Hz), 3.72 (3H, s), 4.96 (1H, d, J= 8.0 Hz), 6.70 (1H, d, J= 8.0 Hz). Ή-麵 R (300 MHz, in the double-mouthed form): δ 2.74 (2Η, t, J = 7.3 Hz), 3.08 (3H, s), 3.21 (2H, t, J = 7.3 Hz), 3.72 (3H , s), 4.96 (1H, d, J = 8.0 Hz), 6.70 (1H, d, J = 8.0 Hz).
元素分析値: CnH12N204S2 として計算値: C, 43.99; H, 4.03; N, 9.33; S, 21.35 (%)、 実測値; C, 44.03; H, 4.15; N, 9.41; S, 21.35 (%) . Elemental analysis: C n H 12 N 2 0 4 S 2 Calculated: C, 43.99; H, 4.03 ; N, 9.33; S, 21.35 (%), Found; C, 44.03; H, 4.15 ; N, 9.41; S, 21.35 (%).
実施例 15-7 , Example 15-7,
Ή -腿 (300 MHz, 重クロ口ホルム中) : δ 1.29 (3Η, t, J= 7.1 Hz), 2.28 (3H, s), 2.73 (2H, t, J- 7.4 Hz), 3.20 (2H, t, J= 7.4 Hz), 3.72 (3H, s), 4.22 (2H, q, J= 7.1 Hz), 4.28 (2H, s), 4.59 (1H, brs). Ή-Thigh (300 MHz, in double-mouthed form): δ 1.29 (3Η, t, J = 7.1 Hz), 2.28 (3H, s), 2.73 (2H, t, J-7.4 Hz), 3.20 (2H, t, J = 7.4 Hz), 3.72 (3H, s), 4.22 (2H, q, J = 7.1 Hz), 4.28 (2H, s), 4.59 (1H, brs).
元素分析値: C15H18N206S2 として計算値: C, 46.62; H, 4.69; N, 7.25; S, 16.60 (%)、 実測値; C, 46.38; H, 4.65; N, 7.43; S, 16.60 ( ) . Elemental analysis: C 15 H 18 N 2 0 6 S 2 Calculated: C, 46.62; H, 4.69 ; N, 7.25; S, 16.60 (%), Found; C, 46.38; H, 4.65 ; N, 7.43; S, 16.60 ().
実施例 15 - 8 Examples 15-8
Ή -醒 (300 MHz, 重クロ口ホルム中) : δ 1.33 (3Η, t, J= 7.1 Hz), 2.75 (2H, t, J= 7.3 Hz), 2.78 (3H, s), 3.07 (3H, s), 3.28 (2H, t, J= 7.3 Hz), 3.72 (3H, s), 4.26 (2H, q, J= 7.1 Hz). Ή-Awake (300 MHz, in the middle of the double mouth): δ 1.33 (3Η, t, J = 7.1 Hz), 2.75 (2H, t, J = 7.3 Hz), 2.78 (3H, s), 3.07 (3H, s), 3.28 (2H, t, J = 7.3 Hz), 3.72 (3H, s), 4.26 (2H, q, J = 7.1 Hz).
元素分析値: C15H18N206S2 - 0.5H20 として計算値: C, 45.56; H, 4.84; N, 7.08; S, 16.22 (%), 実測値; C, 45.74; H, 4.78; N, 6.86; S, 16.38 (%) . Elemental analysis: C 15 H 18 N 2 0 6 S 2 - 0.5H 2 0 Calculated: C, 45.56; H, 4.84 ; N, 7.08; S, 16.22 (%), Found; C, 45.74; H , 4.78; N, 6.86; S, 16.38 (%).
実施例 15-9 ' - NMR (300 MHz, 重クロ口ホルム中) : δ 1.33 (3Η, i, J- 7..1 Hz), 2.76 (2H, t, J= 7.4 Hz), 2.77 (3H, s), 3.28 (2H, t, J= 7.3 Hz), 3.72 (3H, s), 4.27 (2H, q, J= 7.1 Hz), 4.35 (2H, s). Example 15-9'-NMR (300 MHz, in double-mouthed form): δ 1.33 (3Η, i, J-7.1.1 Hz), 2.76 (2H, t, J = 7.4 Hz), 2.77 (3H , s), 3.28 (2H, t, J = 7.3 Hz), 3.72 (3H, s), 4.27 (2H, q, J = 7.1 Hz), 4.35 (2H, s).
元素分析値: C16H18N208S2 として計算値: C, 44.64; H, 4.21; N, 6.51; S, 14.90 (%)、 実測値; C, 45.00; H, 4.37; N, 6.44; S, 14.67 (%) . Elemental analysis: C 16 H 18 N 2 0 8 S 2 Calculated: C, 44.64; H, 4.21 ; N, 6.51; S, 14.90 (%), Found; C, 45.00; H, 4.37 ; N, 6.44; S, 14.67 (%).
実施例 15-10 Example 15-10
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 1.29 (3Η, t, J= 7.2 Hz), 1.33 (3H, t, J= 7.1 Hz), 2.75 (2H, t, J= 7.4 Hz), 2.77 (3H, s), 3.28 (2H, t, J= 7.3 Hz), 3.72 (3H, s), 4.22 (2H, q, J= 7.1 Hz), 4.26 (2H, q, J= 7.2 Hz), 4.28 (2H, s). , 'H-NMR (300 MHz, in double-mouthed form): δ 1.29 (3Η, t, J = 7.2 Hz), 1.33 (3H, t, J = 7.1 Hz), 2.75 (2H, t, J = 7.4 Hz) ), 2.77 (3H, s), 3.28 (2H, t, J = 7.3 Hz), 3.72 (3H, s), 4.22 (2H, q, J = 7.1 Hz), 4.26 (2H, q, J = 7.2 Hz) ), 4.28 (2H, s).,
元素分析値: C18H22N208S2 として計算値: C, 47.15; H, 4.84; N, 6.11; S, 13.99Elemental analysis: C 18 H 22 N 2 0 8 S 2 Calculated: C, 47.15; H, 4.84 ; N, 6.11; S, 13.99
(%)、 実測値; C, 47.38; H, 4.77; N, 6.10; S, 13.97 (%) . (%), Found; C, 47.38; H, 4.77; N, 6.10; S, 13.97 (%).
実施例 15-11 ' Example 15-11 '
Ή-NMR (300 MHz, 重クロ口ホルム中) : δ 1.33 (3Η, t, J= 7.1 Hz), 2.75 (2H, t, J= 7.3 Hz), 2.77 (3H, s), 22.84 (3H, d, J= 4.8 Hz), 3.28 (2H, t, J= 7.3 Hz), 3.72 (3H, s), 4.19 (2H, s), 4.27 (2H, q, J= 7.1 Hz), 5.75 (1H, m). 実施例 15 - 12 Ή-NMR (300 MHz, in double-mouthed form): δ 1.33 (3Η, t, J = 7.1 Hz), 2.75 (2H, t, J = 7.3 Hz), 2.77 (3H, s), 22.84 (3H, d, J = 4.8 Hz), 3.28 (2H, t, J = 7.3 Hz), 3.72 (3H, s), 4.19 (2H, s), 4.27 (2H, q, J = 7.1 Hz), 5.75 (1H, m). Examples 15-12
¾-NMR (300 MHz, 重クロ口ホルム中) : δ 1.33 (3Η, t, J= 7.1 Hz), 2.75 (2H, t, J= 7.3 Hz), 2.77 (3H, s), 3.28 (2H, t, J- 7.3 Hz), 3.72 (3H, s), 3.77 (3H, s), 4.27 (2H, q, J= 7.1 Hz), 4.30 (2H, s). ¾-NMR (300 MHz, in double-mouthed form): δ 1.33 (3Η, t, J = 7.1 Hz), 2.75 (2H, t, J = 7.3 Hz), 2.77 (3H, s), 3.28 (2H, t, J- 7.3 Hz), 3.72 (3H, s), 3.77 (3H, s), 4.27 (2H, q, J = 7.1 Hz), 4.30 (2H, s).
元素分析値: C17H20N208S2 として計算値: C, 45.94; H, 4.54; N, 6.30 (%), 実 測値; C, 46.02; H, 4.73; N, 6.16 (%) . Elemental analysis: C 17 H 20 N 2 0 8 S 2 Calculated: C, 45.94; H, 4.54 ; N, 6.30 (%), actual Hakachi; C, 46.02; H, 4.73 ; N, 6.16 (% ).
実施例 16-1 Example 16-1
式 (XXX I I I) において R2がイソブチル基、 R5がエトキシカルポニル基、 R 6がメチル基、 R 14が tert-ブトキシカルポニルメチル基である化合物の合成 実施例 15 - 1で合成した化合物 (430 mg, 0.88 mmol)を tert -ブタノ一ル (8ml) に溶解し、 tert-ブトキシカリウム (140 mg, 1.06 廳 ol) を加えて室温で 1時間 撹拌した。 反応液に 1N塩酸 (1.06 ml) を加えて混和し、 室温で 30分間撹拌し た。 反応液を濃縮後、 酢酸ェチル (30ml) で希釈し、 0.05N塩酸、 水および飽和 食塩水で洗净後、 硫酸ナトリゥムで乾燥し溶媒を減圧下に溜去して褐色の油状物 (380 mg) を得た。 これを THF (8ml) に溶解し、 トリェチルァミン(0.31 ml, 2.21 mniol) および臭化べンジル (0.117 ml, 1.06 mmol) を加えて室温で 30分間撹拌 した。 さらに反応液を 70°Cで 18時間撹拌した。 この間、 反応液に 1,8-ジァザ ビシクロ [5, 4, 0] - 7 -ゥンデセン (O.Uml, 0.884 mmol) およびよう化ナトリウム (133 mg, 0.884腿 ol) を加えた。 反応液を酢酸ェチル (30 ml) で希釈し、 0.1N 塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し溶媒を減圧下に溜 去し、残渣をシリカゲル 60 (1 Og)のカラムクロマトグラフィ—に付し、 0.1% ギ酸を含む酢酸ェチル:へキサン(10 : 90)の溶出画分を減圧下濃縮乾固し、 標題化合物の橙色油状物 (112 mg, 0.245 匪 ol) を得た。 Synthesis of Compound of Formula (XXXIII) wherein R 2 is an isobutyl group, R 5 is an ethoxycarbonyl group, R 6 is a methyl group, and R 14 is a tert-butoxycarbonylmethyl group Compound (430) synthesized in Example 15-1 mg, 0.88 mmol) was dissolved in tert-butanol (8 ml), potassium tert-butoxide (140 mg, 1.06 mmol) was added, and the mixture was stirred at room temperature for 1 hour. 1N Hydrochloric acid (1.06 ml) was added to the reaction solution, mixed, and stirred at room temperature for 30 minutes. The reaction mixture was concentrated, diluted with ethyl acetate (30 ml), washed with 0.05N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and the solvent was distilled off under reduced pressure to give a brown oil (380 mg). ). This was dissolved in THF (8 ml), to which triethylamine (0.31 ml, 2.21 mniol) and benzyl bromide (0.117 ml, 1.06 mmol) were added, followed by stirring at room temperature for 30 minutes. Further, the reaction solution was stirred at 70 ° C for 18 hours. During this time, 1,8-diazabicyclo [5,4,0] -7-indene (O.Uml, 0.884 mmol) and sodium iodide (133 mg, 0.884 t) were added to the reaction mixture. Dilute the reaction mixture with ethyl acetate (30 ml), wash with 0.1N hydrochloric acid, water and saturated saline, dry over sodium sulfate, evaporate the solvent under reduced pressure, and concentrate the residue on a silica gel 60 (1 Og) column. The fraction eluted with ethyl acetate: hexane (10:90) containing 0.1% formic acid was concentrated to dryness under reduced pressure. An orange oil (112 mg, 0.245 ol) of the title compound was obtained.
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 1.05 (6Η, d, J= 6.7 Hz), 1.33 (3H, t, J= 7.1 Hz), 1.47 (9H, s), 1.93 (1H, sept, J= 6.7 Hz), 2.77 (3H, s), 2.89 ' (2H, d, J= 6.8 Hz), 4.20 (2H, s), 4.26 (2H, q, J- 7.1 Hz). 'H-NMR (300 MHz, in double-mouthed form): δ 1.05 (6Η, d, J = 6.7 Hz), 1.33 (3H, t, J = 7.1 Hz), 1.47 (9H, s), 1.93 (1H , Sept, J = 6.7 Hz), 2.77 (3H, s), 2.89 '(2H, d, J = 6.8 Hz), 4.20 (2H, s), 4.26 (2H, q, J- 7.1 Hz).
元素分析値: C2。H2SN206S2 として計算値: C, 52.61; H, 6.18; N, 6.14; S, 14.05 (%), 実測値; C, 52.38; H, 6.12; N, 6.24; S, 13.90 (%) Elemental analysis: C 2. H 2S N 2 0 6 S 2 Calculated: C, 52.61; H, 6.18 ; N, 6.14; S, 14.05 (%), Found; C, 52.38; H, 6.12 ; N, 6.24; S, 13.90 ( %)
実施例 16-2 Example 16-2
式(XXX I I I )において R 2が 2-フエネチル基、 R 5がエトキシカルボ二ル基、 R6がメチル基、 R14が tert-ブトキシカルボニルメチル基である化合物の合成 実施例 15-1で合成した化合物(0.56 g, 1.15匪 ol)を tert-ブタノール(12 ml) に溶解し、 臭化 2 -フエネチル (0,213 ml, 1.50 mmol) および tert -ブトキシカリ ゥム (182 mg, 1.38 mmol) を加えて室温で 80分間撹拌した。 さらに約 7時間加 熱還流した。 反応液に炭酸カリウム (79 mg, 0.58 mmol) を加えてさらに 6 で約 15時間撹拌した。 反応液を酢酸ェチル(40ml) で希釈し、 0.1N塩酸、 2% 炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾 燥し溶媒を減圧下に溜去し、 メタノール (2 ml) を加えて混和した。 生成した沈 澱物を濾取し、 メタノールで洗浄後、 乾燥して標題化合物の橙色粉末 (169 mg, 0.335 mmol) を得た。In the formula (XXXIII), R 2 is a 2-phenethyl group, R 5 is an ethoxycarbonyl group, R 6 is a methyl group, and R 14 is a tert-butoxycarbonylmethyl group. Synthesis in Example 15-1 The resulting compound (0.56 g, 1.15 bandol) was dissolved in tert-butanol (12 ml) and 2-phenethyl bromide (0.213 ml, 1.50 mmol) and tert-butoxycalide (182 mg, 1.38 mmol) were added. Stirred at room temperature for 80 minutes. The mixture was heated under reflux for about 7 hours. Potassium carbonate (79 mg, 0.58 mmol) was added to the reaction solution, and the mixture was further stirred at 6 for about 15 hours. The reaction solution was diluted with ethyl acetate (40 ml), washed with 0.1N hydrochloric acid, 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dried over sodium sulfate, and the solvent was distilled off under reduced pressure. ml) and mixed. The resulting precipitate was collected by filtration, washed with methanol, and dried to give the title compound as an orange powder (169 mg, 0.335 mmol).
- NMR (300 MHz, 重クロ口ホルム中) : 3 1.35 (3H, t, J= 7.1 Hz), 1.47 (9H, s), 2.78 (3H, s), 3.00 (2H, brdd, J= 8.6, 7.2 Hz), 3.24 (2H, brdd, J= 8.6, 7.2 Hz), 4.20 (2H, s), 4.27 (2H, q, J= 7.1 Hz), 7.25 (3H, m), 7.30 (2H, m). 元素分析値: C24H28N206S2 として計算値: C, 57.12; H, 5.59; N, 5.55; S, 12.71-NMR (300 MHz, in double-mouthed form): 3 1.35 (3H, t, J = 7.1 Hz), 1.47 (9H, s), 2.78 (3H, s), 3.00 (2H, brdd, J = 8.6, 7.2 Hz), 3.24 (2H, brdd, J = 8.6, 7.2 Hz), 4.20 (2H, s), 4.27 (2H, q, J = 7.1 Hz), 7.25 (3H, m), 7.30 (2H, m) . elemental analysis: C 24 H 28 N 2 0 6 S 2 calculated: C, 57.12; H, 5.59 ; N, 5.55; S, 12.71
( ), 実測値; C, 57.25; H, 5.54; N, 5.74; S, 12.73 (%) . (), Found; C, 57.25; H, 5.54; N, 5.74; S, 12.73 (%).
実施例 16-3 Example 16-3
式(XXX I I I)において R2が 2-フエネチル基、 R5がエトキシカルポニル基、 R 6がメチル基、 R 14がカルポキシメチル基である化合物の合成 Synthesis of a compound of the formula (XXXIII) wherein R 2 is a 2-phenethyl group, R 5 is an ethoxycarbonyl group, R 6 is a methyl group, and R 14 is a carboxymethyl group
実施例 16-2で合成した化合物 (32 mg, 0.063匪 ol) をトリフルォロ酢酸 (0.5 ml) に溶解し、 室温で 70分間放置した。 反応液をトルエン (15 ml) で希釈して 濃縮乾固し、残渣にエーテル(2ml) を加えて混和し、生成した沈澱物を濾取し、 ェ一テルで洗浄後、乾燥して標題化合物の橙色粉末 (22 mg, 0.049腿 ol)を得た。 lH -醒 R (300 MHz, 重クロ口ホルム中) : 6 1.34 (3Η, t, J= 7.1 Hz), 2.78 (3H, s), 3.00 (2H, brdd, J= 8.5, 7.2 Hz), 3.24 (2H, brdd, J= 9.6, 7.2 Hz), 4.27 (2H, q, J= 7.1 Hz), 4.36 (2H, brs), 7.24 (3H, m), 7.31 (2H, m). The compound (32 mg, 0.063 bandol) synthesized in Example 16-2 was dissolved in trifluoroacetic acid (0.5 ml) and left at room temperature for 70 minutes. The reaction solution was diluted with toluene (15 ml), concentrated to dryness, ether (2 ml) was added to the residue, and the mixture was mixed. The resulting precipitate was collected by filtration. After washing with ether, drying was performed to obtain an orange powder of the title compound (22 mg, 0.049 mol). l H-Awake R (300 MHz, in double-mouthed form): 6 1.34 (3Η, t, J = 7.1 Hz), 2.78 (3H, s), 3.00 (2H, brdd, J = 8.5, 7.2 Hz), 3.24 (2H, brdd, J = 9.6, 7.2 Hz), 4.27 (2H, q, J = 7.1 Hz), 4.36 (2H, brs), 7.24 (3H, m), 7.31 (2H, m).
元素分析値: C2。¾。N206S2 として計算値: C, 53.56; H, 4.49; N, 6.25; S, 14.30 (%), 実測値; C, 53.32; H, 4.26; N, 6.28; S, 14.13 (%) . Elemental analysis: C 2. ¾. N 2 0 6 S 2 Calculated: C, 53.56; H, 4.49 ; N, 6.25; S, 14.30 (%), Found; C, 53.32; H, 4.26 ; N, 6.28; S, 14.13 (%) .
実施例 16 - 4 Example 16-4
式 (XXX I I I ) において R 2がべンジル基、 R 5がェトキシカルポニル基 R 6 がメチル基、 R14が tert-ブトキシカルポニルメチル基である化合物の合成 実施例 15- 1で得た化合物 (420mg, 0.863 mmol) を THF-エタノール (1 : 1、 8 ml) に溶解し、 臭化べンジル (0.126 ml, 1.03 mmol) を加え、 室温で撹拌しな がら 20%ナトリウムエトキシド (エタノール溶液、 0.372 ml, 0.950 mmol) を 滴下後、 室温で 4時間撹拌した。 反応液を酢酸ェチル (30 ml) で希釈し、 0.1N 塩酸、 水、 2 %炭酸水素ナトリウム水溶液、 水および飽和食塩水で洗浄後、 硫酸 ナトリウムで乾燥し溶媒を減圧下に溜去して赤色油状物を得た。 これををシリカ ゲル 60 (1 Og)のカラムクロマトグラフィーに付し、酢酸ェチル:へキサン(1 0 : 90)·の溶出画分を減圧下濃縮乾固し、 残渣をメタノールで希釈した。 析出 物を濾取し、 メタノールで洗浄、乾燥して標記化合物の赤橙色粉末(30mg, 0.061 mmol) を得た。 In the formula (XXXIII), R 2 is a benzyl group, R 5 is an ethoxycarbonyl group, R 6 is a methyl group, and R 14 is a tert-butoxycarbonylmethyl group. Synthesis of the compound obtained in Example 15-1 ( 420 mg, 0.863 mmol) was dissolved in THF-ethanol (1: 1, 8 ml), benzyl bromide (0.126 ml, 1.03 mmol) was added, and 20% sodium ethoxide (ethanol solution, (0.372 ml, 0.950 mmol) was added dropwise, followed by stirring at room temperature for 4 hours. Dilute the reaction solution with ethyl acetate (30 ml), wash with 0.1N hydrochloric acid, water, 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dry over sodium sulfate, and evaporate the solvent under reduced pressure to red. An oil was obtained. This was subjected to column chromatography on silica gel 60 (1 Og), the fraction eluted with ethyl acetate: hexane (10:90) · was concentrated to dryness under reduced pressure, and the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give a red-orange powder (30 mg, 0.061 mmol) of the title compound.
'H-NMR (300 MHz, 重クロ口ホルム中) : (51.33 (3H, t, J= 7.1 Hz), 1.46 (9H, s), 2.76 (3H, s), 4.18 (2H, s). 4.25 (2H, q, J= 7.1 Hz), 4.26 (2H, s), 7.27-7.39 (5H, m). 'H-NMR (300 MHz, in double-mouthed form): (51.33 (3H, t, J = 7.1 Hz), 1.46 (9H, s), 2.76 (3H, s), 4.18 (2H, s). 4.25 (2H, q, J = 7.1 Hz), 4.26 (2H, s), 7.27-7.39 (5H, m).
元素分析値: C23H26N206S2 として計算値: C, 56.31; H, 5.34; N, 5.71; S, 13.07 (%;)、 実測値; C, 56.10; H, 5.15; N, 5.97; S, 13.07 ( ) . Elemental analysis: C 23 H 26 N 2 0 6 S 2 Calculated: C, 56.31; H, 5.34 ; N, 5.71; S, 13.07 (%;), Found; C, 56.10; H, 5.15 ; N , 5.97; S, 13.07 ().
上記結晶母液おょぴメタノール洗浄液を集めて濃縮し、 下記化合物の橙色油状物 (180 mg, 0.335 mmol) を得た。 The methanol wash of the crystal mother solution was collected and concentrated to obtain an orange oily substance (180 mg, 0.335 mmol) of the following compound.
'H-NMR (300 MHz, 重クロ口ホルム中) : 01.10 (3H, t, J= 7.0 Hz), 1.22 (3H, t, J- 7.1 Hz), 1.46 (9H, s), 2.04 (3H, s), 3.25 (1H, dq, J= 8.7, 7.0 Hz) , 3.64 (1H, dq, J= 8.8, 7.0 Hz), 3.66/3.67 (1H, s), 4.12 (2H, q, J=7.1Hz), 4.13/4.19 (2H, s), 4.20 (1H, d, J= 12.9 Hz), 4.29 (1H, d, J= 12.9 Hz), 7.24-7.37 (5H, i). 'H-NMR (300 MHz, in double-mouthed form): 01.10 (3H, t, J = 7.0 Hz), 1.22 (3H, t, J- 7.1 Hz), 1.46 (9H, s), 2.04 (3H, s), 3.25 (1H, dq, J = 8.7, 7.0 Hz), 3.64 (1H, dq, J = 8.8, 7.0 Hz), 3.66 / 3.67 (1H, s), 4.12 (2H, q, J = 7.1Hz) ), 4.13 / 4.19 (2H, s), 4.20 (1H, d, J = 12.9 Hz), 4.29 (1H, d, J = 12.9 Hz), 7.24-7.37 (5H, i).
元素分析値: C25H32N207S2 として計算値: C, 55.95; H, 6.01; N, 5.22; S, 11.95 (%)、 実測値; C, 56.17; H, 6.12; N, 5.30; S, 11.70 (%) . Elemental analysis: C 25 H 32 N 2 0 7 S 2 Calculated: C, 55.95; H, 6.01; N, 5.22; S, 11.95 (%), found; C, 56.17; H, 6.12; N, 5.30; S, 11.70 (%).
以下に実施例 16- 1〜16-4で合成した化合物および同様にして合成した化合物の 一覧を示す。 The following is a list of compounds synthesized in Examples 16-1 to 16-4 and compounds synthesized in the same manner.
〔表 1 0 5〕 (Table 105)
、ノ —个= 人 , ノ —person = person
X Y R I 14 実施例 Ri R5 R6 R14XYRI 14 Example Ri R5 R6 R14
16-1 -SCH2CH(CH3)2 -COsEt -CHs -CH2C02C(CH3)316-1 -SCH 2 CH (CH3) 2 -COsEt -CHs -CH 2 C02C (CH3) 3
16-2 -S(CH2)2Ph -G02Et ■CH3 -CH2C02C(CH3)3 16-2 -S (CH 2) 2 Ph -G0 2 Et ■ CH 3 -CH 2 C0 2 C (CH 3) 3
16-3 -S(CH2)2Ph -C02Et •CH3 -CH2CO2H16-3 -S (CH 2 ) 2 Ph -C0 2 EtCH 3 -CH2CO2H
16-4 -SGH2PI1 •C02Et ■CHa -CH2C02C(CH3)3 16-4 -SGH2PI1C0 2 EtCHa -CH 2 C0 2 C (CH 3 ) 3
16-5 -SCH2C02C(CH3)3 -(CH2)7CH3 -H -CH2PI116-5 -SCH2C0 2 C (CH 3 ) 3-(CH 2 ) 7 CH 3 -H -CH2PI1
16-6 -SCH2PI1 -(CH2)5CH3 -H -CH2C02C(CH3)3 16-6 -SCH2PI1-(CH 2 ) 5 CH3 -H -CH 2 C0 2 C (CH 3 ) 3
16-7 -SCH3 -H ■CHs •CH2CO2H16-7 -SCH3 -HCHsCH2CO2H
16-8 -SCH2PI1 -C02Et ■CHs -CH2CO2H16-8 -SCH2PI1 -C0 2 EtCHs -CH2CO2H
16-9 -SCH2Ph ■COsEt -CH3 -CH2CO2CH2CH316-9 -SCH 2 PhCOsEt -CH 3 -CH2CO2CH2CH3
16-10 -SCH2PI1 -CO2H -CHs -CH2CO2H16-10 -SCH2PI1 -CO2H -CHs -CH2CO2H
16-11 -SCH2CH(CH3)2 -COsEt •CH3 ■GH2CO2H 16-11 -SCH 2 CH (CH 3) 2 -COsEt • CH 3 ■ GH2CO2H
16-12 -S(CH2)3COPli -C02Et •CH3 -CH2C02C(CH3)3 16-12 -S (CH 2 ) 3 COPli -C0 2 EtCH 3 -CH 2 C0 2 C (CH 3 ) 3
16-13 -S(CH2)3COPh ■C02Et -CH3 -CH2CO2H 16-13 -S (CH 2 ) 3COPhC0 2 Et -CH 3 -CH2CO2H
差替え用弒(規則 26) 以下に実施例 16- 5〜16-13で合成した化合物のデータを示す。 For replacement (Rule 26) The data of the compounds synthesized in Examples 16-5 to 16-13 are shown below.
実施例 16-5 Example 16-5
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 0.88 (3Η, brt, J= 6.8 Hz), 1.26 (12H, brs), 1.46 (9H, s), 2.03 (2H, brdd, J= 7.2, 6.8 Hz), 3.64 (2H, s), 4.71 (2H, s), 6.50 (1H, brs), 7.30 (3H, m), 7.41 (2H, dd, J= 7.9, 1.5 Hz). 'H-NMR (300 MHz, in double-mouthed form): δ 0.88 (3Η, brt, J = 6.8 Hz), 1.26 (12H, brs), 1.46 (9H, s), 2.03 (2H, brdd, J = 7.2, 6.8 Hz), 3.64 (2H, s), 4.71 (2H, s), 6.50 (1H, brs), 7.30 (3H, m), 7.41 (2H, dd, J = 7.9, 1.5 Hz).
質量分析 (SIMS) : 517 (M+H) Mass spectrometry (SIMS): 517 (M + H)
実施例 16 6 Example 16 6
Ή-NMR (300 MHz, 重クロ口ホルム中): 50.90 (3H, brt, J= 6.6 Hz), 0.93 (3H, t, J= 7.3 Hz), 1.30 (6H, brs), 1.37 (2H, brq, J= 7.6 Hz), 1.46 (2H, m), 1.62 (2H, quint, J= 6.9 Hz), 2.05 (2H, t, J= 7.6 Hz), 4.16 (2H, t, J= 6.7 Hz), 4.23 (2H, s), 4.29 (2H, s), 6.51 (1H, s), 7.34 (5H, m). Ή-NMR (300 MHz, in double-mouthed form): 50.90 (3H, brt, J = 6.6 Hz), 0.93 (3H, t, J = 7.3 Hz), 1.30 (6H, brs), 1.37 (2H, brq , J = 7.6 Hz), 1.46 (2H, m), 1.62 (2H, quint, J = 6.9 Hz), 2.05 (2H, t, J = 7.6 Hz), 4.16 (2H, t, J = 6.7 Hz), 4.23 (2H, s), 4.29 (2H, s), 6.51 (1H, s), 7.34 (5H, m).
元素分析値: C25H32N204S2 として計算値: C, 61.45; H, 6.60; N, 5.73; S, 13.12Elemental analysis: C 25 H 32 N 2 0 4 S 2 Calculated: C, 61.45; H, 6.60 ; N, 5.73; S, 13.12
(%)、 実測値; C, 61.14; H, 6.45; N, 5.94; S, 13.11 (%) . (%), Found; C, 61.14; H, 6.45; N, 5.94; S, 13.11 (%).
実施例 16-7 Example 16-7
¾-NMR (300 MHz, 重ジメチルスルホキシド中) : 52.18 (3H, d, J= 1.0 Hz), 2.46 (3H, s), 4.12 (2H, s), 5.09 (1H, d, J= 1.0 Hz), 13.1 (1H, br). ¾-NMR (300 MHz, in heavy dimethyl sulfoxide): 52.18 (3H, d, J = 1.0 Hz), 2.46 (3H, s), 4.12 (2H, s), 5.09 (1H, d, J = 1.0 Hz) , 13.1 (1H, br).
13C-NMR (75 MHz, 重ジメチルスルホキシド中): 6 13.03 (CH3), 19.20 (CH3) , 39.14 (CH2) , 92.77 (CH), 116.31 (Q), 132.07 (Q), 132.90 (Q), 148.16 (Q), 155.01 (Q), 168.39 (Q). 1 3 C-NMR (in 75 MHz, deuterated dimethyl sulfoxide): 6 13.03 (CH 3) , 19.20 (CH 3), 39.14 (CH 2), 92.77 (CH), 116.31 (Q), 132.07 (Q), 132.90 (Q), 148.16 (Q), 155.01 (Q), 168.39 (Q).
元素分析値: C10H10N204S2 として計算値: C, 41.95; H, 3.52; N, 9.78; S, 22.40 (%), 実測値; C, 41.90; H, 3.68; N, 9.57; S, 22.35 (%) . Elemental analysis: C 10 H 10 N 2 0 4 S 2 Calculated: C, 41.95; H, 3.52 ; N, 9.78; S, 22.40 (%), Found; C, 41.90; H, 3.68 ; N, 9.57; S, 22.35 (%).
実施例 16-8Example 16-8
-腿 (300 MHz, 重クロ口ホルム中) : (51.34 (3H, t, J= 7.1 Hz), 2.76 (3H, s), 4.26 (2H, q, J= 7.1 Hz), 4.27 (2H, s), 4.34 (2H, s), 7.27-7.38 (5H, m) . 元素分析値: C19H18N206S2 として計算値: C, 52.52; H, 4.18; N, 6.45; S, 14.76 実測値; C, 52.63; H, 4.28; N, 6.41; S, 14.66 (%) . -Thigh (300 MHz, in double-mouthed holm): (51.34 (3H, t, J = 7.1 Hz), 2.76 (3H, s), 4.26 (2H, q, J = 7.1 Hz), 4.27 (2H, s) ), 4.34 (2H, s) , 7.27-7.38 (5H, m) elemental analysis: C 19 H 18 N 2 0 6 S 2 calculated:. C, 52.52; H, 4.18; N, 6.45; S, 14.76 found; C, 52.63; H, 4.28; N, 6.41; S, 14.66 (%).
実施例 16-9 Example 16-9
'H-NMR (300 MHz, 重クロ口ホルム中) : 51.28 (3H, t, J= 7.2 Hz), 1.34 (3H, t, J= 7.1 Hz), 2.76 (3H, s), 4.22 (2H, q, J= 7.2 Hz), 4.26 (2H, q, J= 7.1 Hz), 4.27 (2H, s), 4.28 (2H, s), 7.28-40 (5H, m) . 'H-NMR (300 MHz, in double-mouthed form): 51.28 (3H, t, J = 7.2 Hz), 1.34 (3H, t, J = 7.1 Hz), 2.76 (3H, s), 4.22 (2H, q, J = 7.2 Hz), 4.26 (2H, q, J = 7.1 Hz), 4.27 (2H, s), 4.28 (2H, s), 7.28-40 (5H, m).
元素分析値: C21H22N206S2 として計算値: C, 54.53; H, 4.79; N, 6.06; S, 13.87Elemental analysis: C 21 H 22 N 2 0 6 S 2 Calculated: C, 54.53; H, 4.79 ; N, 6.06; S, 13.87
(%), 実測値; C, 54.65; H, 4.84; N, 6.09; S, 13.85 (%) . (%), Found; C, 54.65; H, 4.84; N, 6.09; S, 13.85 (%).
実施例 16-10 Example 16-10
lH-NMR (300 MHz, 重クロ口ホルム-重ジメチルスルホキシド 9:1中): δ 2.77 (3Η, s), 4.23 (2H, s), 4.28 (2H, s), 7.34 (5H, m). lH-NMR (300 MHz, double-mouthed form-deuterated dimethyl sulfoxide in 9: 1): δ 2.77 (3Η, s), 4.23 (2H, s), 4.28 (2H, s), 7.34 (5H, m).
元素分析値: C17H14N206S2-0.6Et20 として計算値: C, 51.68; H, 4.47; N, 6.21Elemental analysis: C 17 H 14 N 2 0 6 S 2 -0.6Et 2 0 Calculated: C, 51.68; H, 4.47 ; N, 6.21
(%), 実測値; C, 52.05; H, 4.43; N, 6.41 (%) . (%), Found; C, 52.05; H, 4.43; N, 6.41 (%).
実施例 16-11 Example 16-11
^- MR (300 MHz, 重クロ口ホルム中) : δ 1.05 (6Η, d, J= 6.6 Hz), 1.34 (3H, t, J= 7.1 Hz), 1.94 (1H, sept, J= 6.7 Hz), 2.77 (3H, s), 2.90 (2H, d, J= 6.8 Hz), 4.27 (2H, q, J= 7.1 Hz), 4.36 (2H, brs). ^-MR (300 MHz, in double-hole form): δ 1.05 (6Η, d, J = 6.6 Hz), 1.34 (3H, t, J = 7.1 Hz), 1.94 (1H, sept, J = 6.7 Hz) , 2.77 (3H, s), 2.90 (2H, d, J = 6.8 Hz), 4.27 (2H, q, J = 7.1 Hz), 4.36 (2H, brs).
元素分析値: C16H20N206S2 として計算値: C, 47.99; H, 5.03; N, 7.00; S, 16.01 (%)、 実測値; C, 48.20; H, 4.95; N, 7.27; S, 15.77 (%) . Elemental analysis: C 16 H 20 N 2 0 6 S 2 Calculated: C, 47.99; H, 5.03; N, 7.00; S, 16.01 (%), found; C, 48.20; H, 4.95; N, 7.27; S, 15.77 (%).
実施例 16 - 12 Examples 16-12
[H-NMR (300 MHz, 重クロ口ホルム中) : 51.33 (3H, t, J= 7.1 Hz), 1.47 (9H, s), 2.18 (2H, quint, J= 6.9 Hz) , 2.77 (3H, s) , 3.16 (2H, t, J= 6.9 Hz), 3.18 (2H, t, J= 6.9 Hz), 4.20 (2H, s), 4.25 (2H, q, J= 7.1 Hz), 7.47 (2H, brt, J= 7.8 Hz), 7.57 (1H, tt, J= 7.4, 2.1 Hz), 7.96 (2H, d, J= 7.9 Hz). [ H-NMR (300 MHz, in double-mouthed form): 51.33 (3H, t, J = 7.1 Hz), 1.47 (9H, s), 2.18 (2H, quint, J = 6.9 Hz), 2.77 (3H, s), 3.16 (2H, t, J = 6.9 Hz), 3.18 (2H, t, J = 6.9 Hz), 4.20 (2H, s), 4.25 (2H, q, J = 7.1 Hz), 7.47 (2H, brt, J = 7.8 Hz), 7.57 (1H, tt, J = 7.4, 2.1 Hz), 7.96 (2H, d, J = 7.9 Hz).
元素分析値: C26H30N2O7S2 として計算値: C, 57.13; H, 5.53; N, 5.12; S, 11.73Elemental analysis: C 26 H 30 N 2 O 7 S 2 Calculated: C, 57.13; H, 5.53 ; N, 5.12; S, 11.73
(%)、 実測値; C, 57.18; H, 5.52; N, 5.00; S, 11.71 ( ) · (%), Found; C, 57.18; H, 5.52; N, 5.00; S, 11.71 ()
実施例 16-13 Example 16-13
'H-NMR (300 MHz, 重クロロホルム中) : 51.33 (3H, t, J= 7.1 Hz) , 2.18 (2Η, quint, J= 6.8 Hz), 2.76 (3H, s), 3.16 (2H, t, J= 6.9 Hz), 3.18 (2H, t, J= 6.9 Hz), 4.26 (2H, q, J= 7.1 Hz), 4.35 (2H, brs), 7.47 (2H, brt, J= 7.5 Hz), 7.58 (1H, brt, J= 7.1 Hz), 7.96 (2H, brd, J= 7.3 Hz). 'H-NMR (300 MHz, in deuterated chloroform): 51.33 (3H, t, J = 7.1 Hz), 2.18 (2Η, quint, J = 6.8 Hz), 2.76 (3H, s), 3.16 (2H, t, J = 6.9 Hz), 3.18 (2H, t, J = 6.9 Hz), 4.26 (2H, q, J = 7.1 Hz), 4.35 (2H, brs), 7.47 (2H, brt, J = 7.5 Hz), 7.58 (1H, brt, J = 7.1 Hz), 7.96 (2H, brd, J = 7.3 Hz).
元素分析値: C22H22N2O7S2-0.2¾0 として計算値: C, 53.47; H, 4.57; N, 5.67; S, 12.98 (%), 実測値; C, 53.44; H, 4.54; N, 5.78; S, 12.76 ( ) . 実施例 18 - 1 Elemental analysis: C 22 H 22 N 2 O 7 S 2 -0.2 S0 Calculated: C, 53.47; H, 4.57; N, 5.67; S, 12.98 (%), found; C, 53.44; H, 4.54 ; N, 5.78; S, 12.76 (). Example 18-1
N - {8- [(6-フルォ口へキシル) スルファニル] -5, 6-ジメチル -3-ォキソ- 2, 3-ジヒ ド口- 1H-ィミダゾ [5, 1- c] [1, ]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホ ンアミド ' N- {8-[(6-Fluorohexyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,] thiazine -1-ylidene}-4-methylbenzenesulfonamide ''
DAST(0.50 g, 3.10 mmol)のジクロロメタン(10 ml)の溶液に- 78°Cで攪拌しなが ら実施例 4-64で得た化合物 (1.15 g, 2.39匪 ol)のジクロロメタン (10 ml)溶液 を 40分間かけて滴下し、昇温後、反応液を室温で 1.5時間攪拌した。反応液を酢 酸ェチル(100 ml) で希釈後、 0.1N塩酸、 水および飽和食塩水で洗浄し、 硫酸ナ トリゥムで乾燥後、濃縮して濃赤色固形物として得た。実施例 4-64で得た化合物 (207 mg, 0.430廳 ol) から同様にして得た固形物を合わせてシリカゲル(15 g) のクロマトグラフィ一(酢酸ェチルーへキサン系および酢酸ェチル—トルエン系) により精製して表題化合物(516 mg, 1.07 imnol)を紫色粉末として得た。 The compound obtained in Example 4-64 (1.15 g, 2.39 bandol) in dichloromethane (10 ml) was stirred in a solution of DAST (0.50 g, 3.10 mmol) in dichloromethane (10 ml) at -78 ° C. The solution was added dropwise over 40 minutes, and after heating, the reaction solution was stirred at room temperature for 1.5 hours. The reaction solution was diluted with ethyl acetate (100 ml), washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to obtain a dark red solid. The solids obtained in the same manner from the compound obtained in Example 4-64 (207 mg, 0.430 mmol) were combined and subjected to chromatography on silica gel (15 g) (ethyl acetate-hexane and ethyl acetate-toluene). Purification gave the title compound (516 mg, 1.07 imnol) as a purple powder.
ΐ-NMR (300MHz, CDC13): δ 1.45 (4Η, m), 1.70 (4H, m), 1.79 (3H, q, J= 0.8 Hz), 2.26 (3H, q, J= 1.0 Hz), 2.42 (3H, s), 2.94 (2H, t, J= 7.5 Hz), 4.51 (2H, dt, J= 47.2, 5.9 Hz), 7.29 (2H, d, }= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). ΐ-NMR (300MHz, CDC1 3 ): δ 1.45 (4Η, m), 1.70 (4H, m), 1.79 (3H, q, J = 0.8 Hz), 2.26 (3H, q, J = 1.0 Hz), 2.42 (3H, s), 2.94 (2H, t, J = 7.5 Hz), 4.51 (2H, dt, J = 47.2, 5.9 Hz), 7.29 (2H, d,} = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値 C21H26N303S3F として計算値: C, 52.15; H, 5.42; N, 8.69 (%) 、 実 測値: C, 52.02; H, 5.26; N, 8.58 (%) . Elemental analysis: calculated as C 21 H 26 N 3 0 3 S 3 F: C, 52.15; H, 5.42; N, 8.69 (%), found: C, 52.02; H, 5.26; N, 8.58 (% ).
mp. 157.0-159.0°C mp.157.0-159.0 ° C
以下に実施例 18-1 で合成した化合物および同様にして合成した化合物の一覧 を示す。 The following is a list of compounds synthesized in Example 18-1 and compounds synthesized in the same manner.
〔表 1 0 6〕(Table 106)
簪ぇ用紙(規則26 〔表 1 0 7〕Hairpin paper (Rule 26 (Table 107)
差替え用紙(規則 2β) 〔表 1 0 8〕Replacement form (Rule 2β) (Table 108)
差眷ぇ用銑(規則 26) 〔表 1 0 9〕Pig iron (Rule 26) (Table 10)
羞替え用弒(規則 26) 〔表 1 1 0〕羞 (Rule 26) (Table 110)
左赘& (纖 〔表 1 1 1〕Left 赘 & (Fiber (Table 1 1 1)
褰替え用鈹 (i¾l!l26) 〔表 1 1 2〕褰 for replacement 鈹 (i¾l! L26) (Table 1 1 2)
差替え用弒(規則 2 以下に実施例 18-2〜18-80で合成した化合物のデータを示す。 For replacement (Rule 2 The data of the compounds synthesized in Examples 18-2 to 18-80 are shown below.
実施例 18 - 2 Example 18-2
4-ブロモ -N- {8- [ (6-フルォ口へキシル)スルファニル] -5, 6-ジメチル- 3-ォキソ -2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1, ]チアジン- 1-ィリデン }ベンゼンスルホ ンアミド 4-bromo-N- {8-[(6-fluorohexyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1 ,] Thiazine-1-ylidene} benzenesulfonamide
DAST (335 mg, 1.87画 ol)のジクロロメタン(6 ml)の溶液に- 78°Cで攪拌しなが ら実施例 4- 505で得た化合物 (0.85 g, 1.56 mmol)のジクロロメタン (5 ml)溶液 を 10分間かけて滴下し、 昇温後、 反応液を室温で 30分間攪拌した。 反応液を実 施例 18- 1と同様に後処理、 精製、 メタノールから結晶化して表題化合物 (315 mg, 0.57 皿01)を濃紫色粉末として得た。 The compound obtained in Example 4-505 (0.85 g, 1.56 mmol) in dichloromethane (5 ml) was stirred in a solution of DAST (335 mg, 1.87 ol) in dichloromethane (6 ml) at −78 ° C. The solution was added dropwise over 10 minutes, and after heating, the reaction solution was stirred at room temperature for 30 minutes. The reaction solution was worked up, purified and crystallized from methanol in the same manner as in Example 18-1 to give the title compound (315 mg, 0.57 in dish 01) as a dark purple powder.
¾一 NMR (200MHz, CDC13): δ 1.45 (4Η, m) , 1.71 (4Η, m), 1.80 (3Η, q, J= 1.2 Hz), 2.28 (3H, q, J= 0.6 Hz), 2.96 (2H, t, J= 7.3 Hz), 4.46 (2H, dt, J= 47.2, 5.8 Hz), 7.63 (2H, dt, J= 8.8, 2.2 Hz), 7.83 (2H, di, J= 8.8, 2.2 Hz), 9.63 (lH, brs). ¾ one NMR (200MHz, CDC1 3): δ 1.45 (4Η, m), 1.71 (4Η, m), 1.80 (3Η, q, J = 1.2 Hz), 2.28 (3H, q, J = 0.6 Hz), 2.96 (2H, t, J = 7.3 Hz), 4.46 (2H, dt, J = 47.2, 5.8 Hz), 7.63 (2H, dt, J = 8.8, 2.2 Hz), 7.83 (2H, di, J = 8.8, 2.2 Hz), 9.63 (lH, brs).
元素分析値: C2QH23N303S3BrFとして計算値: C, 43.79; H, 4.23; N, 7.66; S, 17.54; Br, 14.57; F, 3.46 (%) 、 実測値: C, 43.86; H, 4.05; N, 7.55; S, 17.28; Br, 14.51; F, 2.98 (%) . Elemental analysis: C 2Q H 23 N 3 0 3 S 3 BrF Calculated: C, 43.79; H, 4.23 ; N, 7.66; S, 17.54; Br, 14.57; F, 3.46 (%), Found: C , 43.86; H, 4.05; N, 7.55; S, 17.28; Br, 14.51; F, 2.98 (%).
mp. 171.0-171.5°C mp.171.0-171.5 ° C
実施例 18-3 Example 18-3
N - {8- [(7-フルォ口へプチル) スルファニル] - 5, 6-ジメチル -3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン }-4-メチルベンゼンスルホ ンアミド N- {8-[(7-Fluoroheptyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1 -ylidene} -4-methylbenzenesulfonamide
実施例 4-507で得た化合物 (807 mg, 1.63 mmol)のジクロロメタン(7 mi)の溶 液に 0°Cで攪拌しながら DAST (583 mg, 3.26讓 ol)のジクロロメタン (3 ml)溶液 を 40分間かけて滴下し、 昇温後、 反応液を室温で 22時間攪拌した。 反応液を実 施例 18- 1と同様に後処理、 精製、 メタノ一ルから結晶化して表題化合物(343 nig, 0.689匪 ol)を赤色粉末として得た。 To a solution of the compound (807 mg, 1.63 mmol) obtained in Example 4-507 in dichloromethane (7 mi) was added a solution of DAST (583 mg, 3.26 alcohol) in dichloromethane (3 ml) while stirring at 0 ° C. The mixture was added dropwise over 40 minutes, and after heating, the reaction solution was stirred at room temperature for 22 hours. The reaction solution was worked up, purified and crystallized from methanol in the same manner as in Example 18-1 to give the title compound (343 nig, 0.689 bandol) as a red powder.
Ή-NMR (200MHz, CDC13): δΐ.38 (6Η, m), 1.68 (4H, m), 1.79 (3H, q, J= 1.0 Hz), 2.26 (3H, q, J= 1.0 Hz), 2.42 (3H, s), 2.94 (2H, t, J= 7.4 Hz), 4.45 (2H, dt, J= 47.4, 6.1 Hz), 7.29 (2H, d, J- 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δΐ.38 (6Η, m), 1.68 (4H, m), 1.79 (3H, q, J = 1.0 Hz), 2.26 (3H, q, J = 1.0 Hz), 2.42 (3H, s), 2.94 (2H, t, J = 7.4 Hz), 4.45 (2H, dt, J = 47.4, 6.1 Hz), 7.29 (2H, d, J-8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C22H28N303S3Fとして計算値: C, 53.09; H, 5.67; N, 8.44; S, 19.33; F, 3.82 ( ) 、 実測値: C, 53.04; H, 5.47; N, 8.40; S, 19.37; F, 3.45 (%) . nip. 169.0-169.5°C Elemental analysis: C 22 H 28 N 3 0 3 S 3 F Calculated: C, 53.09; H, 5.67 ; N, 8.44; S, 19.33; F, 3.82 (), Found: C, 53.04; H, 5.47; N, 8.40; S, 19.37; F, 3.45 (%) .nip. 169.0-169.5 ° C
実施例 18 - 4 Example 18-4
N-{5- [(5,6-ジメチル -卜 {[(4-メチルフエニル)スルホニル]イミノ}-3 -ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 8 -ィル)スルファニル]ペンチ ル}へキサンアミド N- {5-[(5,6-dimethyl-tri {[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, tri c] [1,4] Thiazine-8-yl) sulfanyl] pentyl} hexaneamide
実施例 18-6 におけるシリカゲルクロマトグラフィーで酢酸ェチルートルエン (4: 6-5: 5)の画分を濃縮乾固後ジェチルエーテルから結晶化して表題化合物(158 mg, 0.280 mmol)を赤色粉末として得た。 The fraction of ethyl acetate-toluene (4: 6-5: 5) was concentrated to dryness by silica gel chromatography in Example 18-6 and crystallized from getyl ether to give the title compound (158 mg, 0.280 mmol) as a red powder. As obtained.
Ή-NM (300MHz, CDC13): 50.89 (3H, t, J- 6.9 Hz), 1.31 (4H, m), 1.46 (2H, m), 1.54 (2H, m), 1.60 (2H, m), 1.71 (2H, quint, J= 7.2 Hz), 1.79 (3H, brs), 2.15 (2H, t,' J= 7.6 Hz), 2.26 (3H, brs), 2.42 (3H, s), 2.94 (2H, t, J= 7.5 Hz), 3.25 (2H, brq, J= 6.6 Hz), 5.51 (1H, m), 7.29 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή-NM (300MHz, CDC1 3 ): 50.89 (3H, t, J- 6.9 Hz), 1.31 (4H, m), 1.46 (2H, m), 1.54 (2H, m), 1.60 (2H, m), 1.71 (2H, quint, J = 7.2 Hz), 1.79 (3H, brs), 2.15 (2H, t, 'J = 7.6 Hz), 2.26 (3H, brs), 2.42 (3H, s), 2.94 (2H, t, J = 7.5 Hz), 3.25 (2H, brq, J = 6.6 Hz), 5.51 (1H, m), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz) , 9.64 (1H, brs).
元素分析値: C26H36N404S3として計算値: C, 55.29; H, 6.42; , 9.92; S, 17.03 (%) 、 実測値: C, 55.37; H, 6.56; , 9.90; S, 16.82 (%) .' Elemental analysis: C 26 H 36 N 4 0 4 S 3 Calculated: C, 55.29; H, 6.42 ;, 9.92; S, 17.03 (%), Found: C, 55.37; H, 6.56 ;, 9.90; S, 16.82 (%). '
mp. 154.5-155.5°C mp.154.5-155.5 ° C
実施例 18 - 5 Example 18-5
N-{8 - [(5-ァミノペンチル) スルファニル ]-5, 6 -ジメチル- 3-ォキソ - 2, 3-ジヒド 口- 1H -イミダゾ [5, 1-c] [1, 4]チアジン- ί -ィリデン }-4-メチルベンゼンスルホン アミド トリフルォロ酢酸塩 N- {8-[(5-Aminopentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine-ί- Ylidene} -4-methylbenzenesulfonamide trifluoroacetate
実施例 4-512で得た化合物(1.07 g, 1.89 mmol) を TFA (2.0 ml)に溶解し、 室 温で 40分間放置した。 反応液をトルエン (50 ml)で希釈して濃縮乾固し、 残渣 に酢酸ェチルを加えて赤色懸濁液を得た。析出物を濾取し、酢酸ェチルで洗浄後、 乾燥して表題化合物 (1.01 g, 1.74 mmol)を赤紫色粉末として得た。 The compound (1.07 g, 1.89 mmol) obtained in Example 4-512 was dissolved in TFA (2.0 ml) and left at room temperature for 40 minutes. The reaction solution was diluted with toluene (50 ml) and concentrated to dryness. Ethyl acetate was added to the residue to obtain a red suspension. The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (1.01 g, 1.74 mmol) as a red-purple powder.
¾-NMR (300MHz, CDC13): δ 1.49 (2Η, m), 1.69 (4H, m), 1.76 (3H, q, J- 0.9 Hz), 2.24 (3H, q, J= 0.9 Hz), 2.40 (3H, s), 2.93 (2H, t, J= 6.9 Hz), 2.98 (2H, t, J= 7.2 Hz), 7.29 (2H, d, J= 8.1 Hz), 7.82 (2H, d, J= 8.1 Hz), 8.13 (3H, brs). ¾-NMR (300MHz, CDC1 3 ): δ 1.49 (2Η, m), 1.69 (4H, m), 1.76 (3H, q, J- 0.9 Hz), 2.24 (3H, q, J = 0.9 Hz), 2.40 (3H, s), 2.93 (2H, t, J = 6.9 Hz), 2.98 (2H, t, J = 7.2 Hz), 7.29 (2H, d, J = 8.1 Hz), 7.82 (2H, d, J = 8.1 Hz), 8.13 (3H, brs).
元素分析値: C22H27N405S3F3 -0.3¾0として計算値: C, 45.09; H, 4.75; N, 9.56; S, 16.41; F, 9.73 ( ) 、 実測値: C, 45.14; H, 4.83; N, 9.62; S, 16.38 ( ) . 実施例 18-6 Elemental analysis: C 22 H 27 N 4 0 5 S 3 F 3 -0.3¾0 Calculated: C, 45.09; H, 4.75 ; N, 9.56; S, 16.41; F, 9.73 (), Found: C, 45.14; H, 4.83; N, 9.62; S, 16.38 ().
N-{5-[(5, 6 -ジメチル- 1- {[(4 -メチルフエニル)スルホニル]ィミノ }- 3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィル)スルファニル]ペンチ ル}-2, 2, 2-トリフルォロアセタミド N- {5-[(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] Thiazine-8-yl) sulfanyl] pentyl} -2,2,2-trifluoroacetamide
実施例 18-5で得た化合物 (290 mg, 0.499 mmol) および塩ィ匕へキサノィル(80.7 mg, 0.599腿 ol)の DMF (4.0 ml)懸濁液にトリェチルァミン(0.208 ml, 1.50匪 ol) を室温で加え、反応液を室温で 18時間攪拌した。反応中に塩化へキサノィル (39 mg, 0.289 mmol)を 2時間毎に 3回加えた。 反応液を酢酸ェチルで抽出し、 脱水乾燥、 濃縮して濃赤色固形物を得た。 これと実施例 18-5 で得た化合物 (0.79 g, 1.36 mmol)から同様にして得た濃赤色固形物(0.45 g)を合わせて THFに懸濁して不溶物 を除去し、 可溶分をシリカゲル(15 g)のクロマトグラフィー (酢酸ェチル—トル ェン、 1:9-5:5) に付した。 酢酸ェチル一トルエン(2:8-3:7)の画分を濃縮乾固後 ジェチルエーテルから結晶化して表題化合物(140 mg, 0.248 mmol)を橙色粉末と して得た。 To a suspension of the compound (290 mg, 0.499 mmol) obtained in Example 18-5 and DMF (4.0 ml) of Shii-dani hexanoyl (80.7 mg, 0.599 t) was added triethylamine (0.208 ml, 1.50 ol). At room temperature, the reaction was stirred at room temperature for 18 hours. Hexanoyl chloride (39 mg, 0.289 mmol) was added three times every two hours during the reaction. The reaction solution was extracted with ethyl acetate, dried, dried and concentrated to obtain a dark red solid. This was combined with the dark red solid (0.45 g) similarly obtained from the compound (0.79 g, 1.36 mmol) obtained in Example 18-5, and suspended in THF to remove insolubles. Chromatography on silica gel (15 g) (ethyl acetate-toluene, 1: 9-5: 5). The fraction of ethyl acetate-toluene (2: 8-3: 7) was concentrated to dryness and crystallized from getyl ether to give the title compound (140 mg, 0.248 mmol) as an orange powder.
— NMR (300MHz, CDC13): δ 1.47 (4Η, m), 1.61 (2H, m), 1.73 (2H, quint, J= 7.2 Hz), 1.79 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J= 7.2 Hz), 3.38 (2H, brq, J= 7.2 Hz), 6.44 (1H, m), 7.29 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 7.8 Hz), 9.64 (1H, brs). - NMR (300MHz, CDC1 3) : δ 1.47 (4Η, m), 1.61 (2H, m), 1.73 (2H, quint, J = 7.2 Hz), 1.79 (3H, s), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.2 Hz), 3.38 (2H, brq, J = 7.2 Hz), 6.44 (1H, m), 7.29 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 7.8 Hz), 9.64 (1H, brs).
元素分析値: C22H25N404S3F3•O tolueneとして計算値: C, 49.69; H, 4.74; N, 9.34; S, 16.05; F, 9.51 ( ) 、 実測値: C, 49.60; H, 4.74; N, 9.37; S, 15.94; F, 9.24 (%) . Elemental analysis: C 22 H 25 N 4 0 4 S 3 F 3 • O toluene Calculated: C, 49.69; H, 4.74 ; N, 9.34; S, 16.05; F, 9.51 (), Found: C, 49.60; H, 4.74; N, 9.37; S, 15.94; F, 9.24 (%).
mp. 168.5-169.5 mp. 168.5-169.5
実施例 18-7 Example 18-7
N - {8 - [(6 -ァミノへキシル) スルファニル ]-5, 6-ジメチル -3 -ォキソ -2, 3-ジヒド ロ- lH-イミダゾ [5, l-c] [1,4]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホン アミド トリフルォロ酢酸塩 N- {8-[(6-aminohexyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrido B-lH-imidazo [5, lc] [1,4] thiazine-1-ylidene} -4-methylbenzenesulfonamide trifluoroacetate
実施例 -513で得た化合物 (2.40 g, 4.13匪 ol) を TFA (5.0 ml)に溶解し、 室温で 50分間放置した。 反応液をトルエン(50 ml)で希釈して濃縮乾固し、 残渣 に酢酸ェチルを加えて赤色懸濁液を得た。析出物を濾取し、酢酸ェチルで洗浄後、 乾燥して表題化合物 (2.41 g, 4.05腿 ol)を赤紫色粉末として得た。 The compound obtained in Example -513 (2.40 g, 4.13 bandol) was dissolved in TFA (5.0 ml) and left at room temperature for 50 minutes. The reaction solution was diluted with toluene (50 ml) and concentrated to dryness. Ethyl acetate was added to the residue to obtain a red suspension. The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (2.41 g, 4.05 l) as a red-purple powder.
¾-NMR (300MHz, DMSO— d6): (51.34 (4H, m), 1.52 (2H, brquint, J= 6.9 Hz), 1.59 (2H, brq, J= 8 Hz), 1.76 (3H, brs), 2.17 (3H, q, J= 0.9 Hz), 2.39 (3H, s), 2.78 (2H, brq, J= 7 Hz), 2.97 (2H, t, J= 7.2 Hz), 7.37 (2H, d, J= 7.8 Hz), 7.67 (3H, brs), 7.84 (2H, d, J- 8.1 Hz), 11.70 (1H, brs). ¾-NMR (300MHz, DMSO— d 6 ): (51.34 (4H, m), 1.52 (2H, brquint, J = 6.9 Hz), 1.59 (2H, brq, J = 8 Hz), 1.76 (3H, brs) , 2.17 (3H, q, J = 0.9 Hz), 2.39 (3H, s), 2.78 (2H, brq, J = 7 Hz), 2.97 (2H, t, J = 7.2 Hz), 7.37 (2H, d, J = 7.8 Hz), 7.67 (3H, brs), 7.84 (2H, d, J- 8.1 Hz), 11.70 (1H, brs).
元素分析値: C23H29N405S3F3として計算値: C, 46.45; H, 4.92; N, 9.42; S, 16.18; F, 9.58 (%) 実測値: C, 46.35; H, 4.91; N, 9.41; S, 16.10 ( ) . Elemental analysis: C 23 H 29 N 4 0 5 Calculated S 3 F 3: C, 46.45 ; H, 4.92; N, 9.42; S, 16.18; F, 9.58 (%) Found: C, 46.35; H , 4.91; N, 9.41; S, 16.10 ().
mp. 169.0-171.5°C mp. 169.0-171.5 ° C
実施例 18 - 8 Example 18-8
Ν-{6-[(5, 6-ジメチル-卜 {[(4-メチルフエニル)スルホニル]ィミノ卜 3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, l-c] [1, 4]チアジン- 8-ィル)スルファニル]へキシ ル}- 2, 2, 2-トリフルォロアセ夕ミド Ν- {6-[(5,6-Dimethyl-{[(4-methylphenyl) sulfonyl] imino 3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine- 8-yl) sulfanyl] hexyl}-2,2,2-trifluoroacetamide
実施例 18-7 で得た化合物 (305 mg, 0.513翻 ol) およびト リェチルァミン (0.213 ml, 1.54 mmol)の DMF-THF (1:3, 8.0 ml)懸濁液に塩化へキサノィル(104 mg, 0.769 mmol)の THF (1.0 ml)溶液を室温で滴下し、 反応液を室温で 1時間攪 拌した。 反応液を酢酸ェチルで抽出し、 脱水乾燥、 濃縮して粗物質を得た。 これ をシリカゲル(15 g)のクロマトグラフィー (酢酸ェチルートルエン、 1:9-4:6) に 付した。酢酸ェチル一トルエン(15: 85-2: 8)の画分を濃縮乾固後ジェチルェ一テル から結晶化して表題化合物 (115 mg, 0.199讓 ol)を赤紫色粉末として得た。 'H-NMR (300MHz, CDC13): <51.43 (4H, m), 1.60 (2H, m), 1.70 (2H, quint, J= 7.0 Hz), 1.79 (3H, brs), 2.26 (3H, q, J= 0.9 Hz), 2.42 (3H, s), 2.94 (2H, t, J= 7.4 Hz), 3.37 (2H, q, J= 6.7 Hz), 6.45 (1H, br), 7.29 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.1 Hz), 9.64 (1H, brs). Hexanoyl chloride (104 mg, 0.513 mmol) was added to a suspension of the compound obtained in Example 18-7 (305 mg, 0.513 mmol) and triethylamine (0.213 ml, 1.54 mmol) in DMF-THF (1: 3, 8.0 ml). (0.769 mmol) in THF (1.0 ml) was added dropwise at room temperature, and the reaction solution was stirred at room temperature for 1 hour. The reaction solution was extracted with ethyl acetate, dried, dried and concentrated to obtain a crude substance. This was chromatographed on silica gel (15 g) (ethyl acetate-toluene, 1: 9-4: 6). The fraction of ethyl ethyl acetate (15: 85-2: 8) was concentrated to dryness and crystallized from geethyl ether to give the title compound (115 mg, 0.199 acetyl) as a red-purple powder. 'H-NMR (300MHz, CDC1 3): <51.43 (4H, m), 1.60 (2H, m), 1.70 (2H, quint, J = 7.0 Hz), 1.79 (3H, brs), 2.26 (3H, q , J = 0.9 Hz), 2.42 (3H, s), 2.94 (2H, t, J = 7.4 Hz), 3.37 (2H, q, J = 6.7 Hz), 6.45 (1H, br), 7.29 (2H, d , J = 8.1 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.64 (1H, brs).
元素分析値: C23H27N404S3F3として計算値: C, 47.90; H, 4.72; N, 9.72; S, 16.68; F, 9.88 (%) 、 実測値: C, 47.95; H, 4.65; N, 9.71; S, 16.69; F, 9.71 (%) · nip. 150.0-150.5 : Elemental analysis: C 23 H 27 N 4 0 4 S 3 F 3 Calculated: C, 47.90; H, 4.72 ; N, 9.72; S, 16.68; F, 9.88 (%), Found: C, 47.95; H, 4.65; N, 9.71; S, 16.69; F, 9.71 (%) · nip. 150.0-150.5:
実施例 18-9 Example 18-9
N- [5, 6-ジメチル- 8- ( { [1- (メチルスルホニル) -4-ピぺリジニル]メチル }スルファ 二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, c] [1, 4]チアジン- 1-ィリデ 実施例 4-116で得た化合物(450 rag, 0.874匪 ol)およびメタンスルホニルク ロリド (121 mg, 1.05 IMOI)の DMF (9.0 ml)溶液に 0°Cで攪拌しながらトリェチ ルァミン (0.363 ml, 2.62腿 ol)を滴下した。 反応液を 0°Cで 7時間攪拌した。 反 応液を酢酸ェチルで希釈し、 0.05N塩酸、 水および飽和食塩水で順次洗浄し、 乾 燥後、濃縮した。析出物を濾取して酢酸ェチルで洗浄、乾燥して表題化合物(145 mg, 0.260 minol)を紫色粉末として得た。 N- [5,6-dimethyl-8-({[1- (methylsulfonyl) -4-pyridinyl] methyl} sulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5, c] [1, 4] thiazine-1-ylide 0% was added to a solution of the compound obtained in Example 4-116 (450 rag, 0.874 ol) and methanesulfonyl chloride (121 mg, 1.05 IMOI) in DMF (9.0 ml). While stirring at ° C, triethylamine (0.363 ml, 2.62 ol) was added dropwise. The reaction was stirred at 0 ° C. for 7 hours. The reaction solution was diluted with ethyl acetate, washed successively with 0.05N hydrochloric acid, water and saturated saline, dried and concentrated. The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (145 mg, 0.260 minol) as a purple powder.
Ή-NMR (300MHz, CDC13): 51.36 (2H, dq, J= 3.0, 13.8 Hz), 1.62 (4H, m) , 1.79 (3H, s), 1.95 (2H, brd, J= 13.5 Hz), 2.27 (3H, s), 2.43 (3H, s), 2.64 (2H, brt, J= 11.4 Hz), 2.79 (3H, s), 2.88 (2H, d, J= 6.6 Hz), 3.83 (2H, brd, J= 11.4 Hz), 7.29 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.1 Hz) , 9.66 (1H, brs). 元素分析値: C22H28N405S4として計算値: C, 47.46; H, 5.07; N, 10.06 (%) 、 実測値: C, 47.80; H, 5.14; N, 9.84 (%) . Ή-NMR (300MHz, CDC1 3 ): 51.36 (2H, dq, J = 3.0, 13.8 Hz), 1.62 (4H, m), 1.79 (3H, s), 1.95 (2H, brd, J = 13.5 Hz), 2.27 (3H, s), 2.43 (3H, s), 2.64 (2H, brt, J = 11.4 Hz), 2.79 (3H, s), 2.88 (2H, d, J = 6.6 Hz), 3.83 (2H, brd , J = 11.4 Hz), 7.29 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.66 (1H, brs). Elemental analysis: C 22 H 28 N 4 0 5 calculated S 4: C, 47.46; H , 5.07; N, 10.06 (%), Found: C, 47.80; H, 5.14 ; N, 9.84 (%).
実施例 18-10 Example 18-10
N- (4- {[(5, 6-ジメチル-ト {[(4-メチルフ ニル)スルホニル]イミノ } - 3 -ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル)スルファニル]メチ ル}フエニル)へキサンアミド N- (4-{[(5,6-dimethyl-to {[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-8-yl) sulfanyl] methyl} phenyl) hexaneamide
実施例 3-4で得られた N- (5, 6-ジメチル -3-ォキソ- 8-スルファニル -2, 3-ジヒド 口- 1H -ィミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン) -4-メチルベンゼンスルホン アミド (200 mg, 0.524腿 ol) と炭酸カリウム (109 mg, 0.786讓 ol) の N,N-ジ メチルホルムアミド(2.00ml)の懸濁液に、参考例 68で得られたへキサン酸 (4- クロロメチルフエニル)アミド (188 mg, 0.780 匪 ol) の N,N -ジメチルホルムァ ミド (2.00 ml) 溶液を加えた。 反応混合物を 80°Cで 30分間加熱した後、 1N塩酸 を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下 に留去した。残留物をシリカゲルカラムクロマトグラフィーに付し、 II-へキサン - 酢酸ェチル (10:1, v/v) で溶出して題記化合物 (48.6 mg, 16%) を暗赤色結晶と して得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 212- 214°Cの暗赤色 結晶を得た。 N- (5,6-Dimethyl-3-oxo-8-sulfanyl-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine- obtained in Example 3-4 A 1-ylidene) -4-methylbenzenesulfonamide (200 mg, 0.524 t ol) and potassium carbonate (109 mg, 0.786 benzyl) suspension in N, N-dimethylformamide (2.00 ml) A solution of hexanoic acid (4-chloromethylphenyl) amide (188 mg, 0.780 ol) in N, N-dimethylformamide (2.00 ml) obtained in 68 was added. After heating the reaction mixture at 80 ° C. for 30 minutes, 1N hydrochloric acid was added and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent under reduced pressure Was distilled off. The residue was subjected to silica gel column chromatography, and eluted with II-hexane-ethyl acetate (10: 1, v / v) to give the title compound (48.6 mg, 16%) as dark red crystals. The crystal was recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 212-214 ° C.
IR (KBr) V: 3248, 3046, 2957, 2930, 2859, 1748, 1725, 1663, 1603, 1559, 1539 cm—1. IR (KBr) V: 3248, 3046, 2957, 2930, 2859, 1748, 1725, 1663, 1603, 1559, 1539 cm— 1 .
'H-NMR (CDC13) : 50.916 (3H, t, J=6.6Hz), 1.28-1.44 (4H, m), 1.64-1.82 (2H, m), 1.76 (3H, s), 2.24 (3H, d, J=1.0 Hz), 2.36 (2H, t, J=7.8 Hz), 2.41 (3H, s), 4.15 (2H, s), 7.19 (1H, bs), 7.26 (4H, d, J=8.6 Hz), 7.50 (2H, d, J=8. Hz), 7.80 (2H, d, J=8.2 Hz), 9.59 (1H, bs) . 'H-NMR (CDC1 3) : 50.916 (3H, t, J = 6.6Hz), 1.28-1.44 (4H, m), 1.64-1.82 (2H, m), 1.76 (3H, s), 2.24 (3H, d, J = 1.0 Hz), 2.36 (2H, t, J = 7.8 Hz), 2.41 (3H, s), 4.15 (2H, s), 7.19 (1H, bs), 7.26 (4H, d, J = 8.6 Hz), 7.50 (2H, d, J = 8 Hz), 7.80 (2H, d, J = 8.2 Hz), 9.59 (1H, bs).
元素分析値: C28H32N404S30.4H20として Elemental analysis: C 28 H 32 N 4 0 4 S 3 0.4 H 2 0
計算値 (¾) : C, 56.81; H, 5.58; N, 9.46; S, 16.25. Calculated (¾): C, 56.81; H, 5.58; N, 9.46; S, 16.25.
実測値 (¾) : C, 56.74; H, 5.50; N, 9.39; S, 16.00. Found (¾): C, 56.74; H, 5.50; N, 9.39; S, 16.00.
実施例 18-11 Example 18-11
tert-ブチル 3- { [ (8- (ィソぺンチルスルファニル) -6-メチル -1 - { [ (4-メチルフェ ニル)スルホニル]ィミノ }_3-ォキソ - 2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, 4]チ ァジン- 5-ィル)カルボニル]アミノ}プロピルカーバメート tert-butyl 3-{[(8- (isopentylsulfanyl) -6-methyl-1-{[(4-methylphenyl) sulfonyl] imino} _3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazin-5-yl) carbonyl] amino} propyl carbamate
実施例 4-462で得た化合物(573 mg, 1.19 mmol)、 卜ヒドロキシベンゾトリ ァゾ一ル (以下、 H0BTと略記することがある) (177 mg, 1.31 腿 ol) および卜 ェチル -3- (3 -ジメチルァミノプロピル)カルポジイミド塩酸塩 (WSC) (以下、 WSC と略記することがある) (251 mg, 1.31 匪 ol) の THF (12 id)の懸濁液を室温で 5時間攪拌した。 反応中に WSC(46 mg, 0.238 mmol)を 2回加えた。 反応液を酢酸 ェチルで希釈し、 0.05N塩酸、 水および飽和食塩水で順次洗浄し、 乾燥後、 濃縮 した。 析出物を濾取して酢酸ェチルで洗浄、 乾燥して 8 - (イソペンチルスルファ ニル) - 6-メチル -1- { [ (4-メチルフエニル)スルホニル]ィミノ }-3-ォキソ - 2, 3 -ジ ヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 5-カルボン酸の H0BTエステル(0.68 g)を紫色粉末として得た。 これを THF (12 ml)に溶解し、 N-Boc -プロパンジアミ ン (254 mg, 1.43画 ol)を加えて室温で 20分間攪拌し、 さらに卜リエチルァミン (0.412 ml, 2.98 腿 ol)を加えて室温で 90分間攪拌した。 反応液を酢酸ェチルで 希釈し、 0.05N塩酸、 水および飽和食塩水で順次洗浄し、 乾燥後、 濃縮し、 酢酸 ェチルおよびジェチルェ一テルで希釈した。析出物を濾取して酢酸ェチル-ジェチ ルェ一テル(1:2) およびジェチルエーテルで洗浄、乾燥して表題化合物 (607 mg, 0.952腿 ol)を赤紫色粉末として得た。 The compound obtained in Example 4-462 (573 mg, 1.19 mmol), trihydroxybenzotriazole (hereinafter sometimes abbreviated as H0BT) (177 mg, 1.31 thigh ol) and triethyl-3- A suspension of (3-mg-dimethylaminopropyl) carbodiimide hydrochloride (WSC) (hereinafter sometimes abbreviated as WSC) (251 mg, 1.31 bandol) in THF (12 id) was stirred at room temperature for 5 hours. . During the reaction, WSC (46 mg, 0.238 mmol) was added twice. The reaction solution was diluted with ethyl acetate, washed successively with 0.05N hydrochloric acid, water and saturated saline, dried and concentrated. The precipitate is collected by filtration, washed with ethyl acetate, dried and dried to give 8- (isopentylsulfanyl) -6-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3 The H0BT ester of 0.6-dihydro-1H-imidazo [5, tric] [1,4] thiazine-5-carboxylic acid (0.68 g) was obtained as a purple powder. This was dissolved in THF (12 ml), N-Boc-propanediamine (254 mg, 1.43 ol) was added, the mixture was stirred at room temperature for 20 minutes, and triethylamine (0.412 ml, 2.98 ol) was added. For 90 minutes. Reaction solution with ethyl acetate The mixture was diluted, washed sequentially with 0.05N hydrochloric acid, water and saturated saline, dried, concentrated, and diluted with ethyl acetate and ethyl acetate. The precipitate was collected by filtration, washed with ethyl acetate-ethyl ether (1: 2) and getyl ether, and dried to give the title compound (607 mg, 0.952 mol) as a red-purple powder.
Ή-NMR (300MHz, CDC13): 50.93 (6H, d, J= 6.3 Hz), 1.44 (9H, s), 1.57 (2H, quint, J= 7.4 Hz), 1.66 (1H, m), 1.73 (2H, m), 1.90 (3H, s), 2.42 (3H, s), 2.96 (2H, t, J= 7.7 Hz), 3.25 (2H, brq," J= 6.3 Hz), 3.41 (2H, brq, J= 6.3 Hz), 4.91 (1H, brt, J= 6.3 Hz), 6.99 (1H, brt, J= 5.5 Hz), 7.29 (2H, d, J二 8.4 Hz), 7.83 (2H, d, J= 8.1 Hz), 9.76 (1H, br). Ή-NMR (300MHz, CDC1 3 ): 50.93 (6H, d, J = 6.3 Hz), 1.44 (9H, s), 1.57 (2H, quint, J = 7.4 Hz), 1.66 (1H, m), 1.73 ( 2H, m), 1.90 (3H, s), 2.42 (3H, s), 2.96 (2H, t, J = 7.7 Hz), 3.25 (2H, brq, "J = 6.3 Hz), 3.41 (2H, brq, J = 6.3 Hz), 4.91 (1H, brt, J = 6.3 Hz), 6.99 (1H, brt, J = 5.5 Hz), 7.29 (2H, d, J 8.4 Hz), 7.83 (2H, d, J = 8.1 Hz), 9.76 (1H, br).
元素分析値: C28H39N506S3 -0.2H20として計算値: C, 52.43; H, 6.19; N, 10.92; S, 15.00 (%) 、 実測値: C, 52.33; H, 6.14; N, 10.75; S, 14.99 (%) . Elemental analysis: C 28 H 39 N 5 0 6 S 3 -0.2H 2 0 Calculated: C, 52.43; H, 6.19 ; N, 10.92; S, 15.00 (%), Found: C, 52.33; H , 6.14; N, 10.75; S, 14.99 (%).
mp. 165.0-166.0°C mp.165.0-166.0 ° C
実施例 18- 12 Example 18-12
N - {8- [(3 -ァミノプロピル) スルファニル ]-5, 6-ジメチル -3-ォキソ -2, 3-ジヒド 口- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン }- 4-メチルベンゼンスルホン アミド 塩酸塩 N- {8-[(3-Aminopropyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydrone-1H-imidazo [5,1-c] [1,4] thiazine-1 Ylidene}-4-methylbenzenesulfonamide hydrochloride
実施例 4-106で得た化合物(883 mg, 1.55 mmol) を TFA (2 ml)に溶解し、 室 温で 1時間放置した。 反応液をトルエンで希釈して濃縮乾固後、残渣を THFおよ びメタノール(1:2, 60 ml)に溶解し、 6N塩酸 (0.39 ml)を加えて濃縮した。 残渣 に THFを加えて懸濁し、 析出物を濾取し、 THFおよび酢酸ェチルで洗浄後、 乾燥 ' して表題化合物 (350 mg, 0.737 mmol)を赤紫色粉末として得た。 The compound (883 mg, 1.55 mmol) obtained in Example 4-106 was dissolved in TFA (2 ml) and left at room temperature for 1 hour. The reaction solution was diluted with toluene and concentrated to dryness. The residue was dissolved in THF and methanol (1: 2, 60 ml), and concentrated by adding 6N hydrochloric acid (0.39 ml). The residue was suspended in THF, and the precipitate was collected by filtration, washed with THF and ethyl acetate, and dried to give the title compound (350 mg, 0.737 mmol) as a red-purple powder.
Ή—NMR (300MHz, DMSO— d6): 51.80 (3H, s),' 1.90 (2H, quint, J= 7.5 Hz), 2.17 (3H, s), 2.39 (3H, s), 2.86 (2H, m), 3.06 (2H, i, J= 7.2 Hz), 7.39 (2H, d, J= 8.1 Hz), 7.85 (2H, d, J- 8.1 Hz), 7.92 (3H, brs), 11.71 (1H, brs). 元素分析値: C18H22N403S3 CI Ό.5¾0として計算値: C, 44.66; Η, 5.00; Ν, 11.57; S, 19.87; C1, 7.32 (%) 、 実測値: C, 44.56; Η, 5.00; Ν, 11.66; S, 19.84; C1, 7.18 (%) . Ή-NMR (300MHz, DMSO-d 6 ): 51.80 (3H, s), '1.90 (2H, quint, J = 7.5 Hz), 2.17 (3H, s), 2.39 (3H, s), 2.86 (2H, s) m), 3.06 (2H, i, J = 7.2 Hz), 7.39 (2H, d, J = 8.1 Hz), 7.85 (2H, d, J- 8.1 Hz), 7.92 (3H, brs), 11.71 (1H, brs). Elemental analysis: Calculated for C 18 H 22 N 4 0 3 S 3 CI Ό.5¾0: C, 44.66; Η, 5.00; Ν, 11.57; S, 19.87; C1, 7.32 (%), actual : C, 44.56; Η, 5.00; Ν, 11.66; S, 19.84; C1, 7.18 (%).
実施例 18-13 Example 18-13
Ν - {3- [(5, 6-ジメチル _1 - {[(4-メチルフエニル)スルホニル]イミノ} - 3 -ォキソ - 2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1 - c] [1, 4]チアジン- 8-ィル)スルファニル]プロピ ル}へキサンアミド Ν- {3-[(5,6-dimethyl_1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo -2,3-Dihydro-1H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] propyl} hexaneamide
実施例 18- 12で得た化合物(0.43 g, 0.81龍 ol) およびト リェチルァミン(0.338 ml, 2.44讓01)の1¾«?-11^ (1:2, 9.0 ml)懸濁液に塩化へキサノィル(164 mg, 1.22 fflmol)を室温で滴下し、反応液を室温で 2時間攪拌した。反応液を酢酸ェテルで抽 出し、 脱水乾燥、 濃縮後、 ジェチルエーテルで希釈した。 析出物を濾取し、 ジェ テルエ一テルで洗浄後、 酢酸ェチルおよびジェチルエーテルから結晶化して表題 化合物 (207 mg, 0.386 蘭 ol)を赤紫色粉末として得た。 Hexanoyl chloride was added to a suspension of the compound obtained in Example 18-12 (0.43 g, 0.81 r. Ol) and triethylamine (0.338 ml, 2.44 alcohol01) in 1¾ «?-11 ^ (1: 2, 9.0 ml). (164 mg, 1.22 fflmol) was added dropwise at room temperature, and the reaction solution was stirred at room temperature for 2 hours. The reaction solution was extracted with ether acetate, dehydrated and dried, concentrated, and then diluted with getyl ether. The precipitate was collected by filtration, washed with petroleum ether, and crystallized from ethyl acetate and getyl ether to give the title compound (207 mg, 0.386 orchid ol) as a red-purple powder.
Ή—NMR (300MHz, CDC13): 50.89 (3H, t, J= 6.9 Hz), 1.30 (4H, m), 1.63 (2H, quint, J= 7.8 Hz), 1.79 (3H, q, J= 0.9 Hz), 1.90 (2H, quint, J= 7.4 Hz), 2.17 (2H, t, J= 7.7 Hz), 2.26 (3H, q, J= 0.9 Hz), 2.42 (3H, s), 2.97 (2H, t, J= 7.4 Hz), 3.34 (2H, q, J= 6.5 Hz), 5.77 (1H, brt, J= 5 Hz), 7.30 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.66 (1H, brs). Ή-NMR (300MHz, CDC1 3 ): 50.89 (3H, t, J = 6.9 Hz), 1.30 (4H, m), 1.63 (2H, quint, J = 7.8 Hz), 1.79 (3H, q, J = 0.9 Hz), 1.90 (2H, quint, J = 7.4 Hz), 2.17 (2H, t, J = 7.7 Hz), 2.26 (3H, q, J = 0.9 Hz), 2.42 (3H, s), 2.97 (2H, t, J = 7.4 Hz), 3.34 (2H, q, J = 6.5 Hz), 5.77 (1H, brt, J = 5 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.66 (1H, brs).
元素分析値: C24H32N404S30.5H20として計算値: C, 52.82; H, 6.09; N, 10.27; S, 17.63 {%) 、 実測値: C, 52.62; H, 5.79; N, 10.36; S, 17.59 (%) . Elemental analysis: C 24 H 32 N 4 0 4 S 3 0.5H 20 Calculated: C, 52.82; H, 6.09; N, 10.27; S, 17.63 (%), found: C, 52.62; H, 5.79; N, 10.36; S, 17.59 (%).
mp. 140.0-141.0°C ' 実施例 18 - 14 mp. 140.0-141.0 ° C '' Example 18-14
N - {8- [(2-アミノエチル) スルファニル ]-5, 6 -ジメチル -3-ォキソ -2, 3-ジヒドロ _1Η-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }- 4-メチルベンゼンスルホンァ ミド 塩酸塩 N- {8-[(2-Aminoethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro_1 イ -imidazo [5,1-c] [1,4] thiazine-1-ylidene }-4-methylbenzenesulfonamide hydrochloride
実施例 4-114で得た化合物(1.33 g, 2.53 mmol) を TFA (2 ml)に溶解し、 室 温で 30分間放置した。 反応液をトルエンで希釈して濃縮乾固後、 残渣を THFお よびメタノール(2:3, 50 ml)に溶解し、 6N塩酸 (0.63 ml, 3.80讓 )を加えて 濃縮した。 残渣に THF およびメタノールを加えて懸濁し、 析出物を濾取し、 THF および酢酸ェチルで洗浄後、 乾燥して表題化合物 (0.92 g, 1.99腿 ol)を赤褐色粉 末として得た。 The compound (1.33 g, 2.53 mmol) obtained in Example 4-114 was dissolved in TFA (2 ml) and left at room temperature for 30 minutes. The reaction solution was diluted with toluene and concentrated to dryness. The residue was dissolved in THF and methanol (2: 3, 50 ml), and concentrated by adding 6N hydrochloric acid (0.63 ml, 3.80%). The residue was suspended by adding THF and methanol, and the precipitate was collected by filtration, washed with THF and ethyl acetate, and dried to give the title compound (0.92 g, 1.99 tmol) as a red-brown powder.
Ή— NMR (300MHz, DMS0—d6): δ 1.80 (3H, q, J= 0.9 Hz), 2.17 (3H, q, J= 1.2 Hz), 2.39 (3H, s), 3.06 (2H, m), 3.21 (2H, t, J= 6.9 Hz), 7.39 (2H, d, J= 8.1 Hz), 7.86 (2H, d, J= 8.1 Hz), 8.09 (3H, brs), 11.74 (1H, br). 元素分析値: C17H2QN403S3 iiCl 0として計算値: C, 42.62; H, 4.84; N, 11.70; S, 20.08; Cl, 7.40 ( ) 、 実測値: C, 42.75; H, 4.85; N, 11.66; S, 20.26; C1, 7.32 (%) . Ή- NMR (300MHz, DMS0-d 6): δ 1.80 (3H, q, J = 0.9 Hz), 2.17 (3H, q, J = 1.2 Hz), 2.39 (3H, s), 3.06 (2H, m) , 3.21 (2H, t, J = 6.9 Hz), 7.39 (2H, d, J = 8.1 Hz), 7.86 (2H, d, J = 8.1 Hz), 8.09 (3H, brs), 11.74 (1H, br) . Elemental analysis: Calculated as C 17 H 2Q N 4 0 3 S 3 iiCl 0: C, 42.62; H, 4.84; N, 11.70; S, 20.08; Cl, 7.40 (), Found: C, 42.75; H , 4.85; N, 11.66; S, 20.26; C1, 7.32 (%).
実施例 18 - 15 Examples 18-15
N- [3- (へキサノィルァミノ)プロピル] -8- (ィソペンチルスルファニル) -6-メチル - 1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 3-ジヒドロ- 1H-イミ ダゾ [5,1 - c] [1, 4]チアジン- 5-力ルポキサミド N- [3- (Hexanoylamino) propyl] -8- (isopentylsulfanyl) -6-methyl-1-({[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H -Imidazo [5,1-c] [1,4] Thiazine-5-Dolpoxamide
実施例 18-11で得た化合物(0.23 g, 0.24画 1) から実施例 18-17と同様に して処理して得た N- (3-ァミノプロピル)- 8- (ィソペンチルスルファ二ル)- 6 -メ チル- 1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 3 -ジヒドロ- 1H - イミダゾ [5, 1-c] [1,4]チアジン- 5-カルボキサミド 塩酸塩の赤褐色祖粗粉末を THF (4 ml)およびトリェチルァミン(0.28 ml, 2.0醒01)に懸濁し、 塩化へキサノ ィル(108mg, 0.80mmol)を加え、 反応液を室温で 3時間攪拌した。 反応液を酢酸 ェチルで抽出し、 脱水乾燥、 濃縮後、 ジェチルェ一テルで希釈した。 析出物を濾 取し、 ジェチルエーテルで洗浄後、 乾燥して表題化合物 (59 mg, 0.093腿 ol)を 赤褐色粉末として得た。 N- (3-Aminopropyl) -8- (isopentylsulfane) obtained by treating the compound (0.23 g, 0.24 fraction 1) obtained in Example 18-11 in the same manner as in Example 18-17 ) -6-methyl-1-{[((4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-5 -Carboxamide hydrochloride crude red-brown powder was suspended in THF (4 ml) and triethylamine (0.28 ml, 2.0 Aging 01), hexanoyl chloride (108 mg, 0.80 mmol) was added, and the reaction solution was stirred at room temperature for 3 hours. Stirred. The reaction solution was extracted with ethyl acetate, dehydrated and dried, concentrated, and then diluted with diethyl ether. The precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (59 mg, 0.093 mol) as a red-brown powder.
Ή一 NMR (300MHz, CDC13): 50.89 (3H, t, J= 7.2 Hz) , 0.93 (6H, d, J= 6.6 Hz), 1.31 (4H, in), 1.57 (2H, quint, J= 7.4 Hz), 1.60 (2H, m), 1.75 (2H, m), 1.90 (3H, s), 2.19 (2H, t, J= 7.5 Hz), 2.43 (3H, s), 2.96 (2H, t, J= 7.8 Hz), 3.38 (2H, brq, J= 6.3 Hz), 6.04 (1H, t, J= 5 Hz), 7.14 (1H, brt, J= 5 Hz), 7.29 (2H, , d, J= 7.8 Hz), 7.83 (2H, d, J= 8.1 Hz), 9.77 (1H, br). Ή one NMR (300MHz, CDC1 3): 50.89 (3H, t, J = 7.2 Hz), 0.93 (6H, d, J = 6.6 Hz), 1.31 (4H, in), 1.57 (2H, quint, J = 7.4 Hz), 1.60 (2H, m), 1.75 (2H, m), 1.90 (3H, s), 2.19 (2H, t, J = 7.5 Hz), 2.43 (3H, s), 2.96 (2H, t, J = 7.8 Hz), 3.38 (2H, brq, J = 6.3 Hz), 6.04 (1H, t, J = 5 Hz), 7.14 (1H, brt, J = 5 Hz), 7.29 (2H,, d, J = 7.8 Hz), 7.83 (2H, d, J = 8.1 Hz), 9.77 (1H, br).
元素分析値: C29H41N505S3 -0.6¾0として計算値: C, 53.86; H, 6.58; N, 10.83; S, 14.88 (%) 、 実測値: C, 53.84; H, 6.43; N, 10.92; S, 15.10 ( ) . Elemental analysis: C 29 H 41 N 5 0 5 S 3 -0.6¾0 Calculated: C, 53.86; H, 6.58 ; N, 10.83; S, 14.88 (%), Found: C, 53.84; H, 6.43 ; N, 10.92; S, 15.10 ().
mp. 154.5 - 155.5 mp. 154.5-155.5
実施例 18-16 Example 18-16
N - {3-[(5,6-ジメチル _1-{[(4-メチルフエニル)スルホニル]イミノ}-3-ォキソ -2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル]プロピ ル}-2, 2, 2-トリフルォロアセ夕ミド N- {3-[(5,6-dimethyl_1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-8-yl) sulfanyl] propyl} -2,2,2-trifluoroacetamide
実施例 18-13 における母液および洗浄液を併せて得た粗物質をシリカゲル (lOg)のクロマトグラフィ一 (酢酸ェチルーへキサン、 3:7-8:2) に付した。 酢酸 ェチル—へキサン(5:5)の画分を濃縮乾固後酢酸ェチルから結晶化して表題化合 物(20 mg, 0.037 匪 ol)を赤褐色粉末として得た。 The crude material obtained by combining the mother liquor and the washing solution in Examples 18-13 was treated with silica gel. (lOg) was subjected to chromatography (ethyl acetate-hexane, 3: 7-8: 2). The ethyl acetate-hexane (5: 5) fraction was concentrated to dryness and then crystallized from ethyl acetate to give the title compound (20 mg, 0.037 ol) as a red-brown powder.
Ή-NMR (300MHz, CDC13): δ 1.78 (3Η, s), 2.00 (2H, quint, J= 7.1 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, i, J= 7.4 Hz), 3.47 (2H, brq, J= 6.6 Hz), 6.83 ( , m), 7.30 (2H, d, J= 8.1 Hz), 7.83 (2H, d, J= 8.1 Hz), 9.66 (1H, brs) . Ή-NMR (300MHz, CDC1 3 ): δ 1.78 (3Η, s), 2.00 (2H, quint, J = 7.1 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.98 (2H, i, J = 7.4 Hz), 3.47 (2H, brq, J = 6.6 Hz), 6.83 (, m), 7.30 (2H, d, J = 8.1 Hz), 7.83 (2H, d, J = 8.1 Hz), 9.66 ( 1H, brs).
元素分析値: C2。H21N404S3F3として計算値: C, 44.93; H, 3.69; N, 10.48; S, 17.99 (%) 、 実測値: C, 45.04; H, 4.02; N, 10.42; S, 18.00 {%) · Elemental analysis: C 2. Calculated for H 21 N 4 0 4 S 3 F 3 : C, 44.93; H, 3.69; N, 10.48; S, 17.99 (%), found: C, 45.04; H, 4.02; N, 10.42; S, 18.00 (%)
mp. 167.0-168.0°C mp.167.0-168.0 ° C
実施例 18-17 Example 18-17
N- (3 -ァミノプロピル) -8- (ィソぺンチルスルファニル) -6-メチル -1- { [ (4-メチル フエニル)スルホニル〕ィミノ }- 3-ォキソ -2, 3-ジヒ ド口- 1H-ィミダゾ [5,卜 c][l, 4]チアジン- 5 -カルボキサミド 塩酸塩 N- (3-Aminopropyl) -8- (isopentylsulfanyl) -6-methyl-1-{[((4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrogen- 1H-imidazo [5, tric] [l, 4] thiazine-5-carboxamide hydrochloride
実施例 18-11 で得た化合物(327 mg, 0.513腿 ol) を TFA (0.5 ml)に溶解し、 室温で 30分間放置した。 反応液を実施例 18-11と同様にして処理し、 表題化合 物 (232 mg, 0.404 mmol)を濃赤色粉末として得た。 The compound obtained in Example 18-11 (327 mg, 0.513 tmol) was dissolved in TFA (0.5 ml) and left at room temperature for 30 minutes. The reaction solution was treated in the same manner as in Example 18-11 to give the title compound (232 mg, 0.404 mmol) as a dark red powder.
Ή一 NMR (300MHz, DMS0-d6): δθ.88 (6Η, d, J= 6.6 Hz), 1.49 (2H, brq, J= 7.0 Hz), 1.62 (1H, sept, J= 6.9 Hz), 1.75 (2H, brquint,. J= 7 Hz), 1.83 (3H, s), 2.39 (3H, s), 2.83 (2H, brq, J= 5 Hz), 3.01 (2H, t, J= 7.5 Hz), 3.21 (2H, brq, J= 6.0 Hz), 7.39 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.4 Hz), 7.86 (3H, brs), 11.86 (1H, br). Ή one NMR (300MHz, DMS0-d 6 ): δθ.88 (6Η, d, J = 6.6 Hz), 1.49 (2H, brq, J = 7.0 Hz), 1.62 (1H, sept, J = 6.9 Hz), 1.75 (2H, brquint, J = 7 Hz), 1.83 (3H, s), 2.39 (3H, s), 2.83 (2H, brq, J = 5 Hz), 3.01 (2H, t, J = 7.5 Hz) , 3.21 (2H, brq, J = 6.0 Hz), 7.39 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.4 Hz), 7.86 (3H, brs), 11.86 (1H, br) .
元素分析値: C23H31N504S3 flCl -0.8H20として計算値: C, 46.93; H, 5.75; N, 11.90; S, 16.34; CI, 6.02 (%) 、 実測値: C, 46.98; H, 5.75; N, 11.90; S, 16.40; CI, 5.57 (%) . Elemental analysis: C 23 H 31 N 5 0 4 S 3 flCl -0.8H 2 0 Calculated: C, 46.93; H, 5.75 ; N, 11.90; S, 16.34; CI, 6.02 (%), Found: C, 46.98; H, 5.75; N, 11.90; S, 16.40; CI, 5.57 (%).
mp. 154.5-155.5°C mp.154.5-155.5 ° C
実施例 18 - 18 Examples 18-18
N- {2- [(5, 6-ジメチル- 1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5, 1 - c] [1,4]チアジン- 8-ィル)スルファニル]ェチ ル}へキサンアミド N- {2-[(5,6-dimethyl-1-[[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] thiazine-8-yl) sulfanyl] eti } Hexanamide
実施例 18- 14で得た化合物(300 mg, 0.651 mmol)を THF (6.0 ml)およびトリエ チルァミン(0.271 ml, 1.95 匪 ol)に懸濁し、 塩化へキサノィル(131 mg, 0.976 匪 ol)を加え、 反応液を室温で 2時間攪拌した。反応液を酢酸ェチルで抽出し、脱 水乾燥、 濃縮後、 齚酸ェチルおよびジェチルエーテルで希釈した。 析出物を濾取 し、 ジェチルェ一テルで洗浄後、 乾燥して表題化合物 (188 mg, 0.360 mmol)を赤 褐色粉末として得た。 The compound (300 mg, 0.651 mmol) obtained in Examples 18 to 14 was suspended in THF (6.0 ml) and triethylamine (0.271 ml, 1.95 ol), and hexanoyl chloride (131 mg, 0.976 ol) was added. The reaction solution was stirred at room temperature for 2 hours. The reaction solution was extracted with ethyl acetate, dried with water, concentrated and diluted with ethyl acetate and getyl ether. The precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (188 mg, 0.360 mmol) as a red-brown powder.
'H-NMR (200MHz, CDC13): 50.89 (3H, t, J= 6.6 Hz), 1.30 (4H, brquint, J= 3.7 Hz), 1.63 (2H, quint, J= 7.2 Hz), 1.78 (3H, q, J= 1.2 Hz), 2.19 ,(2H, t, J= 7.7 Hz), 2.26 (3H, q, J= 1.0 Hz), 2.43 (3H, s), 3.12 (2H, t, J= 6.1 Hz), 3.52 (2H, q, J= 6.3 Hz), 6.09 (1H, brt, J; 6 Hz), 7.30 (2H, d, J= 8.0 Hz), 7.84 (2H, d, J- 8.0 Hz), 9.69 (1H, brs). 'H-NMR (200MHz, CDC1 3): 50.89 (3H, t, J = 6.6 Hz), 1.30 (4H, brquint, J = 3.7 Hz), 1.63 (2H, quint, J = 7.2 Hz), 1.78 (3H , q, J = 1.2 Hz), 2.19, (2H, t, J = 7.7 Hz), 2.26 (3H, q, J = 1.0 Hz), 2.43 (3H, s), 3.12 (2H, t, J = 6.1 Hz), 3.52 (2H, q, J = 6.3 Hz), 6.09 (1H, brt, J; 6 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J- 8.0 Hz) , 9.69 (1H, brs).
元素分析値: C23H30N404S30.2¾0として計算値: C, 52.49; H, 5.82; N, 10.65; S, 18.28 (%) 、 実測値: C, 52.50; H, 5.84; N, 10.71; S, 18.35. Elemental analysis: C 23 H 30 N 4 0 4 S 3 0.2¾0 Calculated: C, 52.49; H, 5.82; N, 10.65; S, 18.28 (%), found: C, 52.50; H, 5.84; N, 10.71; S, 18.35.
mp. 137.5-138.5°C mp.137.5-138.5 ° C
実施例 18-19 Example 18-19
2- [5, 6-ジメチル- 1- { [(4-メチルフエニル)スルホニル]ィミノ }- 3-ォキソ - 2, 3-ジ ヒド口- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル]スルファニル]- N-へキシルァ セ卜アミド 2- [5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,4] Thiazine-8-yl] sulfanyl] -N-hexylacetoamide
実施例 4-30で得られた 2- [5, 6-ジメチル- 1 - {[(4-メチルフェニル)スルホニル] ィミノ}- 3-ォキソ - 2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル]ス ルファニル]酢酸 (30.7 mg, 0.0698 mmol) と 1 -へキシルァミン(0· 0102ml, 0.0768 mmol) の N,N-ジメチルホルムアミド (3,00 ml) 溶液に、 シァノりん酸ジェチル (0.0124 ml, 0.0768膽 ol) 続いてト リェチルァミン (0.0107 ml, 0.0768脑 ol) を加えた。反応混合物を室温で 15時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥 (MgS04)後、溶媒を減圧下に留去して題記化合物(18.5 ' mg, 51¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 160- 162°Cの暗赤色結晶を得た。 2- [5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo obtained in Example 4-30 [5,1 -c] [1,4] Thiazin-8-yl] sulfanyl] acetic acid (30.7 mg, 0.0698 mmol) and 1-hexylamine (0.0102 ml, 0.0768 mmol) in N, N-dimethylformamide (3,00 To the solution, getyl cyanophosphate (0.0124 ml, 0.0768 v) and triethylamine (0.0107 ml, 0.0768 v) were added. After stirring the reaction mixture at room temperature for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (18.5 'mg, 51¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 160-162 ° C.
IR (KBr) V: 3254, 2957, 2930, 2859, 1752, 1719, 1661, 1622, 1559 cm—1. Ή-NMR (CDCI3) : δ 0.883 (3Η, t, J=6.3Hz), 1.24-1.38 (6H, m), 1.46-1.66 (2H, m), 1.76 (3H, d, J=0.9 Hz), 2.25 (3H, d, J=1.2 Hz), 2.43 (3H, s), 3.27 (2H, q. J=6.3Hz), 3.62 (2H, s), 6.56 (1H, bs), 7.32 (2H, d, J=8.1 Hz), 7.84 (2H, d, J=8.7 Hz), 9.66 (1H, bs). IR (KBr) V: 3254, 2957, 2930, 2859, 1752, 1719, 1661, 1622, 1559 cm- 1 . Ή-NMR (CDCI3): δ 0.883 (3Η, t, J = 6.3Hz), 1.24-1.38 (6H, m), 1.46-1.66 (2H, m), 1.76 (3H, d, J = 0.9 Hz), 2.25 (3H, d, J = 1.2 Hz), 2.43 (3H, s), 3.27 (2H, q. J = 6.3 Hz), 3.62 (2H, s), 6.56 (1H, bs), 7.32 (2H, d , J = 8.1 Hz), 7.84 (2H, d, J = 8.7 Hz), 9.66 (1H, bs).
元素分析値: C23H3()N404S3 ·0· 4 0として Elemental analysis: C 23 H 3 () N 4 0 4 S 3 · 0 · 40
計算値 (¾) : C, 52.13; Η, 5.86; Ν, 10.57; S, 18.15. Calculated (¾): C, 52.13; Η, 5.86; Ν, 10.57; S, 18.15.
実測値 (%) C, 52.04; Η, 6.03; Ν, 10.64'; S, 18.13. Found (%) C, 52.04; Η, 6.03; Ν, 10.64 '; S, 18.13.
実施例 18-20 Example 18-20
6- [ (5, 6-ジメチル-卜 { [ (4-メチルフエニル)スルホニル]イミノ } -3-ォキソ- 2, 3- ジヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン- 8-ィル)スルファニル]- N-へキシル へキサンアミド 6-[(5,6-Dimethyl-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, tri-c] [1,4] thiazine-8 -Yl) sulfanyl]-N-hexylhexanamide
実施例 4-37で得られた 6- [5, 6-ジメチル-卜 { [ (4 -メチルフエニル)スルホニル] ィミノ卜 3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 8 -ィル]ス ルファニル]へキサン酸 (103 mg, 0.210匪 ol) と卜へキシルァミン (0.0305ml, 0.231 腿 ol) の N,N-ジメチルホルムアミド (10.0 ml) 溶液に、 シァノりん酸ジ ェチル (0.0373 ml, 0.231画 ol)、続いてトリェチルァミン(0.0321 ml, 0.231腿 ol) を加えた。反応混合物を室温で 15時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(78.8 mg, 65¾) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 118-120°Cの暗赤色結晶を得た。 6- [5,6-Dimethyl-tolu [[(4-methylphenyl) sulfonyl] iminoto 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] obtained in Example 4-37 N, N-dimethylformamide (10.0 ml) solution of [1,4] thiazine-8-yl] sulfanyl] hexanoic acid (103 mg, 0.210 ol) and trihexylamine (0.0305 ml, 0.231 ol) To this was added diethyl cyanophosphate (0.0373 ml, 0.231 ol), followed by triethylamine (0.0321 ml, 0.231 t ol). After stirring the reaction mixture at room temperature for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (78.8 mg, 65¾) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 118-120 ° C.
IR (KBr) V: 3260, 2928, 2859, 1723, 1645, 1620, 1559 cm"1. IR (KBr) V: 3260, 2928, 2859, 1723, 1645, 1620, 1559 cm " 1 .
lH-腿 (CDC13) : (50.884 (3H, t, J=6.9Hz), 1.24-1.36 (6H, HI), 1.41-1.76 (8H, m), 1.78 (3H, d, J=0.9 Hz), 2.17 (2H, t, J=7.5 Hz), 2.26 (3H, d, J=0.9 Hz), 2.42 (3H, s), 2.94 (2H, t, J=7.2 Hz), 3.24 (2H, q. J=6.0 Hz), 5.44 (1H, bs), 7.29 (2H, d, J=7.8 Hz), 7.84 (2H, d, J=8. Hz), 9.57 (1H, bs). lH- thigh (CDC1 3): (50.884 ( 3H, t, J = 6.9Hz), 1.24-1.36 (6H, HI), 1.41-1.76 (8H, m), 1.78 (3H, d, J = 0.9 Hz) , 2.17 (2H, t, J = 7.5 Hz), 2.26 (3H, d, J = 0.9 Hz), 2.42 (3H, s), 2.94 (2H, t, J = 7.2 Hz), 3.24 (2H, q. J = 6.0 Hz), 5.44 (1H, bs), 7.29 (2H, d, J = 7.8 Hz), 7.84 (2H, d, J = 8.Hz), 9.57 (1H, bs).
元素分析値: C27H38N404S3 -0.6H20として Elemental analysis: as C 27 H 38 N 4 0 4 S 3 -0.6H 20
計算値 (%) : C, 55.00; H, 6.70; , 9.50. Calculated value (%): C, 55.00; H, 6.70;, 9.50.
実測値 (%) C, 55.00; H, 6.58; N, 9.63. Found (%) C, 55.00; H, 6.58; N, 9.63.
実施例 18-21 5 - [(5, 6-ジメチル- 1- { [(4-メチルフエニル)スルホニル]ィミノ }- 3-ォキソ -2, 3- ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル]吉草酸 実施例 4-522で得られたェチル 5- [(5,6-ジメチル -1-{[ (4-メチルフエニル)ス ルホニル]イミノ }-3 -ォキソ - 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン -8-ィル)スルファニル]ペンタノェ一ト(400 mg, 0.785 mmol)のエタノール(10.0 ml)及びテトラヒドロフラン(10.0 ml)溶液に、 1N水酸化ナトリウム水溶液(2.35 ml) を加えた。 反応混合物を室温で 3時間かき混ぜた後、 溶媒を減圧下に留去し た。 残渣に 1N塩酸を加えて、 生成した沈澱物を濾取し、 水洗後、 乾燥して題記化 合物 (334 mg, 88%) を合成した。 酢酸ェチル -メタノール-テトラヒドロフランか ら再結晶して融点 208- 210°Cの暗赤色結晶を得た。 Example 18-21 5-[(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -8-yl) sulfanyl] valeric acid Ethyl 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2 obtained in Example 4-522 , 3-Dihydro-1H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] pentanoate (400 mg, 0.785 mmol) in ethanol (10.0 ml) and tetrahydrofuran (10.0 ml) 1) To the solution was added a 1N aqueous sodium hydroxide solution (2.35 ml). After stirring the reaction mixture at room temperature for 3 hours, the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and the resulting precipitate was collected by filtration, washed with water, and dried to synthesize the title compound (334 mg, 88%). Recrystallization from ethyl acetate-methanol-tetrahydrofuran gave dark red crystals with a melting point of 208-210 ° C.
IR (KBr) V: 3600-2780, 2951, 1732, 1663, 1611, 1557 cm"1. IR (KBr) V: 3600-2780, 2951, 1732, 1663, 1611, 1557 cm " 1 .
¾-NMR (CDC13) : δ 1.72-1.82 (4Η, m), 1.79 (3H, d, J=0.6 Hz), 2.26 (3H, d, J=1.2 Hz), 2.38-2.46 (2H, m), 2.42 (3H, s), 2.92-3.00 (2H, m), 7.29 (2H, d, J=8.4 Hz), 7.85 (2H, d, J=8.1 Hz), 9.63 (1H, bs). ¾-NMR (CDC1 3): δ 1.72-1.82 (4Η, m), 1.79 (3H, d, J = 0.6 Hz), 2.26 (3H, d, J = 1.2 Hz), 2.38-2.46 (2H, m) , 2.42 (3H, s), 2.92-3.00 (2H, m), 7.29 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.1 Hz), 9.63 (1H, bs).
元素分析値: C2。H23N305S3として Elemental analysis: C 2. H 23 N 3 0 5 S 3
計算値 (%) : C, 49.88; H, 4.81; , 8.72; S, 19.97. Calculated (%): C, 49.88; H, 4.81;, 8.72; S, 19.97.
実測値 (¾) : C, 50.05; H, 4.73; N, 8.35; S, 19.60. Found (¾): C, 50.05; H, 4.73; N, 8.35; S, 19.60.
実施例 18-22 Example 18-22
4- [(5, 6 -ジメチル-卜 {[(4-メチルフエニル)スルホニル]イミノ 3 -ォキソ - 2, 3- ジヒドロ- 1H-イミダゾ [5, 1 - c] [1, 4]チアジン -8-ィル)スルファニル]酪酸 4-[(5,6-dimethyl-tolu [[(4-methylphenyl) sulfonyl] imino 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-8- Yl) sulfanyl] butyric acid
実施例 4-523で得られたェチル 4-[(5,6 -ジメチル- 1- {[(4-メチルフエニル)ス ルホニル]ィミノ } - 3_ォキソ -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン - 8-ィル)スルファニル]ブタノエ一卜 (169 mg, 0.745 mmol) のエタノール (10.0 ml)及びテトラヒドロフラン(10.0 ml)溶液に、 1 水酸化ナトリゥム水溶液(2.23 ml) を加えた。 反応混合物を室温で 2時間かき混ぜた後、 溶媒を減圧下に留去し た。残渣に 1N塩酸を加えて、 生成した沈澱物を濾取し、 水洗後、 乾燥して題記化 合物 (321 mg, 92%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルか ら再結晶して融点 183-185°Cの暗赤色結晶を得た。 Ethyl 4-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} obtained in Example 4-523} -3_oxo-2,3-dihydrido-1H-imidazo [ To a solution of [5, 1-c] [1,4] thiazin-8-yl) sulfanyl] butanoet (169 mg, 0.745 mmol) in ethanol (10.0 ml) and tetrahydrofuran (10.0 ml) was added an aqueous solution of sodium hydroxide. (2.23 ml) was added. After stirring the reaction mixture at room temperature for 2 hours, the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and the resulting precipitate was collected by filtration, washed with water, and dried to give the title compound (321 mg, 92%) as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 183-185 ° C.
IR (KBr) ソ: 3700-2800, 2930, 1726, 1661, 1615, 1557 cm"1. Ή-腿 (CDC13) : (51.78 (3H, d, J=1.2 Hz), 2.03 (2H, quint, J=6.9 Hz), 2.26 (3H, d, J=1.2 Hz), 2.42 (3H, s), 2.50 (2H, t, J=6.9 Hz), 3.03 (2H, t, J=7.2 Hz), 7.30 (2H, d, J=8.1 Hz), 7.85 (2H, d, J=8.1 Hz), 9.67 (1H, bs). IR (KBr) Seo: 3700-2800, 2930, 1726, 1661, 1615, 1557 cm " 1 . Ή- thigh (CDC1 3): (51.78 ( 3H, d, J = 1.2 Hz), 2.03 (2H, quint, J = 6.9 Hz), 2.26 (3H, d, J = 1.2 Hz), 2.42 (3H, s ), 2.50 (2H, t, J = 6.9 Hz), 3.03 (2H, t, J = 7.2 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.85 (2H, d, J = 8.1 Hz), 9.67 (1H, bs).
元素分析値: C19H21N305S3として Elemental analysis: as C 19 H 21 N 3 0 5 S 3
計算値 (¾) : C, 48.80; H, 4.53; N, 8.99; S, 20.57. Calculated (¾): C, 48.80; H, 4.53; N, 8.99; S, 20.57.
実測値 (%) : C, 48.58; H, 4.90; N, 9.11; S, 20.35. Found (%): C, 48.58; H, 4.90; N, 9.11; S, 20.35.
実施例 18-23 Example 18-23
5 - [(5, 6 -ジメチル-卜 {[(4-メチルフエニル)スルホニル]ィミノ卜 3-ォキソ - 2,3- ジヒドロ- 1H-イミダゾ [5, 1- c] [1, 4]チアジン- 8 -ィル)スルファニル]- N-へキシル ペンタンアミド 5-[(5,6-Dimethyl-{[(4-methylphenyl) sulfonyl] imino 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-8 -Yl) sulfanyl]-N-hexyl pentanamide
実施例 18-21で得られた 5 - [(5, 6-ジメチル -1-{ [(4-メチルフエニル)スルホ二 ル]イミノ}_3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル) スルファニル]吉草酸(150 mg, 0.311匪 ol)と卜へキシルァミン(0.0454ml, 0.343 mniol) の N,N-ジメチルホルムアミド (5.00 ml) 溶液に、 シァノりん酸ジェチル (0.0554 ml, 0.343 mmol)、 続いてトリェチルァミン (0.0478 ml, 0.343匪 ol) を加えた。反応混合物を室温で 23時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(101 mg, 57%) を合成した。 酢酸ェチル-ジェチルエーテルから再結晶して融点 133-135°Cの暗赤色結晶を得た。 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} _3-oxo-2,3-dihydro-1H-imidazo [5, obtained in Example 18-21 1-c] [1,4] Thiazin-8-yl) sulfanyl] valeric acid (150 mg, 0.311 bandol) and trihexylamine (0.0454 ml, 0.343 mniol) in N, N-dimethylformamide (5.00 ml) To the solution was added getyl cyanophosphate (0.0554 ml, 0.343 mmol), followed by triethylamine (0.0478 ml, 0.343 bandol). After stirring the reaction mixture at room temperature for 23 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (101 mg, 57%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 133-135 ° C.
IR (KBr) ソ : 3280, 2957, 2928, 2859, 1750, 1717, 1642, 1622, 1557 cm"1. Ή-NMR (CDC13) : 50.880 (3H, t, J=6.6Hz), 1.24-1.36 (6H, m), 1.43-1.56 (2H, m), 1.66-1.82 (4H, m), 1.78 (3H, s), 2.19 (2H, t, J=6.9 Hz), 2.25 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J=7.2 Hz), 3.24 (2H, q, J=6.3Hz), 5.49 (1H, bs), 7.29 (2H, d, J=8.7 Hz), 7.84 (2H, d, J=8.1 Hz). IR (KBr) Seo:. 3280, 2957, 2928, 2859, 1750, 1717, 1642, 1622, 1557 cm "1 Ή-NMR (CDC1 3): 50.880 (3H, t, J = 6.6Hz), 1.24-1.36 (6H, m), 1.43-1.56 (2H, m), 1.66-1.82 (4H, m), 1.78 (3H, s), 2.19 (2H, t, J = 6.9 Hz), 2.25 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.2 Hz), 3.24 (2H, q, J = 6.3 Hz), 5.49 (1H, bs), 7.29 (2H, d, J = 8.7 Hz), 7.84 (2H, d, J = 8.1 Hz).
元素分析値: C26H36N404S3として Elemental analysis: as C 26 H 36 N 4 0 4 S 3
計算値 (¾) : C, 55.29; H, 6.42; N, 9.92; S, 17.03. Calculated value (¾): C, 55.29; H, 6.42; N, 9.92; S, 17.03.
実測値 (%) : C, 54.95; H, 6.42; N, 9.64; S, 16.92. Found (%): C, 54.95; H, 6.42; N, 9.64; S, 16.92.
実施例 18-24 ' Example 18-24 '
4_[(5,6 -ジメチル- 1_{[(4-メチルフエニル)スルホ二ル]イミノ}- 3-ォキソ - 2,3- ジヒドロ- イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル] へキシル ブタンアミド 4 _ [(5,6-Dimethyl-1 _ {[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3- Dihydro-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] hexyl butanamide
実施例 18-22で得られた 4 - [(5, 6-ジメチル- 1-{ [(4-メチルフエニル)スルホ二 ル]イミノ} - 3-ォキソ - 2, 3 -ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル スルファニル)酪酸 (150 mg, 0.321腿 ol) と卜へキシルァミン (0.0467ml, 0.353 腿 ol) の Ν,Ν-ジメチルホルムアミド (5.00 ml) 溶液に、 シァノりん酸ジェチル (0.0571 ml, 0.353 mmol) 続いてトリェチルァミン (0.0492 ml, 0.353 mmol) を加えた。反応混合物を室温で 23時間かき混ぜた後、水を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥 (MgS04)後、溶媒を減圧下に留去して題記化合物(100 ig, 57¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 151- 153°Cの暗赤色結晶を得た。 4-[(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo obtained in Example 18-22 1, Ν-Dimethylformamide (5.00 ml) of 1,1-c] [1,4] thiazin-8-ylsulfanyl) butyric acid (150 mg, 0.321 t ol) and trihexylamine (0.0467 ml, 0.353 t ol) To the solution was added getyl cyanophosphate (0.0571 ml, 0.353 mmol) followed by triethylamine (0.0492 ml, 0.353 mmol). After stirring the reaction mixture at room temperature for 23 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (100 ig, 57¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 151-153 ° C.
IR (KBr) V: 3300, 2955, 2930, 2857, 1748, 1723, 1659, 1644, 1622,' 1564 cm"1. Ή-NMR (CDC13) : 50.882 (3H, t, J=6.9Hz), 1.24-1.38 (6H, m), 1.44-1.60 (2H, m), 1.78 (3H, s), 2.03 (3H, quint, J=6.9Hz), 2.26 (3H, s), 2.32' (2H, t, J=6.6 Hz), 2.43 (3H, s), 3.02 (2H, t, J=6.9 Hz), 3.24 (2H, q. J=6.0 Hz), 5.65 (1H, bs), 7.30 (2H, d, J=8.4 Hz), 7.84 (2H, d, J=8.1 Hz). IR (KBr) V:. 3300 , 2955, 2930, 2857, 1748, 1723, 1659, 1644, 1622, '1564 cm "1 Ή-NMR (CDC1 3): 50.882 (3H, t, J = 6.9Hz), 1.24-1.38 (6H, m), 1.44-1.60 (2H, m), 1.78 (3H, s), 2.03 (3H, quint, J = 6.9Hz), 2.26 (3H, s), 2.32 '(2H, t , J = 6.6 Hz), 2.43 (3H, s), 3.02 (2H, t, J = 6.9 Hz), 3.24 (2H, q.J = 6.0 Hz), 5.65 (1H, bs), 7.30 (2H, d , J = 8.4 Hz), 7.84 (2H, d, J = 8.1 Hz).
実施例 18-25 Example 18-25
5-メ.チル- 1- {[(4-メチルフエニル)スルホニル]ィミ ノ }- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニル] - 2, 3-ジヒドロ -1H -イミ ダゾ [5,卜 c] [1,4]チアジン- 6-カルボン酸 5-methyl-1-[[(4-methylphenyl) sulfonyl] imino}-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]- 2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-6-carboxylic acid
実施例 -521 で得られたェチル 5_メチル -1- {[(4-メチルフエニル)スルホ二 ル]ィミノ }- 3-ォキソ -8-[(5, 5, 6, 6, 6 -ぺンタフルォ口へキシル)スルファニ ル] -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 6-カルポキシレート (500 mg, 0.815 mmol) のエタノール (10.0 ml) 及びテトラヒドロフラン (10.0 ml) 溶液に、 1N水酸化ナトリウム水溶液 (2.44ml) を加えた。 反応混合物を室温で 3 時間かき混ぜた後、溶媒を減圧下に留去した。残渣に 1N塩酸を加えて、 生成した 沈澱物を濾取し、 水洗後、 乾燥して題記化合物 (85.3 mg, 18%) を暗赤色結晶と して得た。酢酸ェチル -ジェテルエ一テルから再結晶して融点 ZZS- Z?^の暗赤色 結晶を得た。 IR (KBr) リ: 3400-2200, 3183, 2926, 2541, 1755, 1705, 1686, 1630, 1557 cm"1. Ή-NMR (CDC13) : (51.66-1.85 (4H, m), 1.96-2.16 (2H, m), 2.42 (3H, s), 2.77 (3H, s), 3.03 (2H, t, J=6.9 Hz), 7.29 (2H, d, J=7.8 Hz), 7.85 (2H, d, J=8.1 Hz), 10.41 (1H, bs). Ethyl 5_methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentanfluo obtained in Example -521 Hexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-6-carboxylate (500 mg, 0.815 mmol) in ethanol (10.0 ml) and tetrahydrofuran To the (10.0 ml) solution was added a 1N aqueous solution of sodium hydroxide (2.44 ml). After stirring the reaction mixture at room temperature for 3 hours, the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and the resulting precipitate was collected by filtration, washed with water, and dried to give the title compound (85.3 mg, 18%) as dark red crystals. Recrystallization from ethyl acetate-jetelether gave dark red crystals with a melting point of ZZS-Z? ^. IR (KBr) Re:. 3400-2200, 3183, 2926, 2541, 1755, 1705, 1686, 1630, 1557 cm "1 Ή-NMR (CDC1 3): (51.66-1.85 (4H, m), 1.96-2.16 (2H, m), 2.42 (3H, s), 2.77 (3H, s), 3.03 (2H, t, J = 6.9 Hz), 7.29 (2H, d, J = 7.8 Hz), 7.85 (2H, d, J = 8.1 Hz), 10.41 (1H, bs).
元素分析値: C2lH2。N305S3F5として Elemental analysis: C 2l H 2. N 3 0 5 S 3 F 5
計算値 ( : C, 43.07; H, 3.44; , 7.18; S, 16.43. Calculated value (: C, 43.07; H, 3.44;, 7.18; S, 16.43.
実測値 : C, 43.05; H, 3. 4; N, 7.40; S, 16.48. Found: C, 43.05; H, 3.4; N, 7.40; S, 16.48.
実施例 18 - 26 Examples 18-26
N-へキシル -5-メチル -1- {[(4-メチルフエニル)スルホニル]イミノ}-3-ォキソ -8-[(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニル ]-2, 3-ジヒドロ- 1H-ィミ ダゾ [5,1 - c] [1, 4]チアジン- 6 -力ルポ酸アミド N-hexyl-5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentahexylhexyl) sulfanyl] -2,3-Dihydro-1H-imidazo [5,1-c] [1,4] thiazine-6-caproleptamide
実施例 18-25 で得られた 5-メチル (4-メチルフエニル)スルホニル]イミ ノ}- 3-ォキソ - 8 - [(5, 5, 6, 6, 6_ペンタフルォ口へキシル)スルファニル ]-2, 3 -ジヒ ドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 6 -力ルボン酸 (50.0 mg, 0.0854讓 ol) と卜へキシルァミン(0.0124 ml, 0.0939 mmo 1 )の N, N-ジメチルホルムアミド(5.00 ml) 溶液に、 シァノりん酸ジェチル (0.0152 ml, 0.0939 mmol), 続いてトリェチ ルァミン (0.0131 ml, 0.0939匪 ol) を加えた。 反応混合物を室温で 15時間かき 混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (12.6 mg, 22¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 160_162°Cの暗赤色結晶を得 た。 5-methyl (4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2 obtained in Example 18-25 , 3-Dihydro-1H-imidazo [5, tric] [1,4] thiazine-6-carburonic acid (50.0 mg, 0.0854 acetylol) and trihexylamine (0.0124 ml, 0.0939 mmo 1) N, To a solution of N-dimethylformamide (5.00 ml) was added getyl cyanophosphate (0.0152 ml, 0.0939 mmol), followed by triethylamine (0.0131 ml, 0.0939 bandol). After stirring the reaction mixture at room temperature for 15 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (12.6 mg, 22¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 160-162 ° C.
IR (KBr) リ: 3382, 3260, 3150, 2934, 2859, 1763, 1742, 1628, 1595, 1539 cm一1. Ή-NMR (CDC13) : 50.885 (3H, t, J=7.0Hz), 1.24-1.43 (6H, m), 1.44-1.66 (2H, HI), 1.66-1.84 (4H, m), 1.88-2.18 (2H, m), 2.42 (3H, s), 2.47 (3H, s), 2.99 (2H, t, J=7.6 Hz), 3.31 (2H, q. J=6.6 Hz), 5.79 (1H, bs), 7.30 (2H, d, J=8.0 Hz), 7.84 (2H, d, J-8. Hz), 9.70 (1H, bs). IR (KBr) Re:. 3382, 3260, 3150, 2934, 2859, 1763, 1742, 1628, 1595, 1539 cm one 1 Ή-NMR (CDC1 3) : 50.885 (3H, t, J = 7.0Hz), 1.24 -1.43 (6H, m), 1.44-1.66 (2H, HI), 1.66-1.84 (4H, m), 1.88-2.18 (2H, m), 2.42 (3H, s), 2.47 (3H, s), 2.99 (2H, t, J = 7.6 Hz), 3.31 (2H, q.J = 6.6 Hz), 5.79 (1H, bs), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J- 8.Hz), 9.70 (1H, bs).
実施例 18 - 27 Examples 18-27
N - {6 -ヒドロキシメチル -5-メチル -3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキ シル)スルファニル ]-2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデ 水素化リチウムアルミニウム(116mg, 2.44 mmol)のテトラヒドロフラン(40.0 ml)懸濁液に、実施例 4-521で得られたェチル 5-メチル -1-{ [(4-メチルフエニル) スルホニル]ィミノ }- 3-ォキソ - 8-[(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルフ ァニル ]-2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 6 -カルボキシレート (l.OOg, 1.63腿 ol) のテトラヒドロフラン (10.0 ml) 溶液を氷冷下を加えた。 反応混合物を室温で 30分間かき混ぜた後、少量の水を加えて過剰の水素化リチウ ムアルミニウムを分解した。 生成した沈殿を濾取し、 濾液を減圧下に留去して題 記化合物 (648 mg,, 70%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 169- 171°Cの暗赤色結晶を得た。 N- {6-Hydroxymethyl-5-methyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydrid-1H- Imidazo [5, 1-c] [1, 4] thiazine-1-ylide In a suspension of lithium aluminum hydride (116 mg, 2.44 mmol) in tetrahydrofuran (40.0 ml) was added ethyl 5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3 obtained in Example 4-521. -Oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine A solution of -6-carboxylate (l.OOg, 1.63 mol) in tetrahydrofuran (10.0 ml) was added under ice cooling. After stirring the reaction mixture at room temperature for 30 minutes, a small amount of water was added to decompose excess lithium aluminum hydride. The resulting precipitate was collected by filtration, and the filtrate was evaporated under reduced pressure to give the title compound (648 mg, 70%) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 169-171 ° C.
I (KBr) V: 3700-2800, 3202, 2928, 1723, 1651, 1607, 1551 cm"1. I (KBr) V: 3700-2800, 3202, 2928, 1723, 1651, 1607, 1551 cm " 1 .
¾一 NMR (CDC13) : δ 1.60-1.68 (1H, m), 1.68-1.84 (4H, m), 1.96-2.16 (4H, m), 2.34 (3H, s), 2.42 (3H, s), 3.01 (2H, t, J=7.2 Hz), 4.26 (2H, d, J=5.4 Hz), 7.29 (2H, d, J=7.8 Hz), 7.84 (2H, d, J=8.4 Hz), 9.68 (1H, bs) . ¾ one NMR (CDC1 3): δ 1.60-1.68 (1H, m), 1.68-1.84 (4H, m), 1.96-2.16 (4H, m), 2.34 (3H, s), 2.42 (3H, s), 3.01 (2H, t, J = 7.2 Hz), 4.26 (2H, d, J = 5.4 Hz), 7.29 (2H, d, J = 7.8 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.68 ( 1H, bs).
元素分析値: C21H22N304S.3F5として Elementary analysis: as C 21 H 22 N 3 0 4 S. 3 F 5
計算値 (%) : C, 44.13; H, 3.88; N, 7.35; S, 16.83. Calculated (%): C, 44.13; H, 3.88; N, 7.35; S, 16.83.
実測値 (%) : C, 44.14; H, 3.66; N, 7.35; S, 16.97. Found (%): C, 44.14; H, 3.66; N, 7.35; S, 16.97.
実施例 18-28 Example 18-28
メタンスルホン酸 2- [ (5, 6-ジメチル -1- { [ (4-メチルフエニル)スルホニル]ィミ ノ}-3_ォキソ-2,3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1,, 4]チアジン- 8-ィル)スルフ ァニル]ェチル Methanesulfonic acid 2-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3_oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1 ,, 4] thiazine-8-yl) sulfanyl] ethyl
実施例 4-524で得た化合物 (5.10 g, 12.0匪 ol)およびメタンスルホニルク ロリド (1.53 g, 13.2匪 ol)の THF (100 ml)溶液に 0°Cで攪拌しながらトリェチ ルァミン(2.16 ml, 15.6 mmol)を加え、 反応液を 0 で 4時間攪拌した。 反応液 にメタンスルホニルクロリド (139 mg, 1.20腿 ol)およびトリェチルァミン(0.83 ml, 6.00 mmol)を加え、 さらに 1.5時間攪拌した。 反応液を濃縮後、 酢酸ェチル (200 ml) および 0.1N塩酸 (200 ml)で希釈した。 析出物を濾取してメタノール —水(1:1)およびメタノールで洗浄後、 乾燥して表題化合物 (3.42 g, 6.80 mmol, 56.6%)を紫色粉末として得た。 Ή-NMR (300MHz, CDC13): 51.80 (3H, s), 2.26 (3H, s), 2.43 (3H, s), 3.03 (3H, s), 3.28 (2H, t, J= 6.8 Hz), 4.38 (2H, t, J= 6.9 Hz), 7.31 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.1 Hz), 9.67 (1H, brs). Triethylamine (2.16 ml) was added to a solution of the compound obtained in Example 4-524 (5.10 g, 12.0 bandol) and methanesulfonyl chloride (1.53 g, 13.2 bandol) in THF (100 ml) while stirring at 0 ° C. , 15.6 mmol) was added and the reaction was stirred at 0 for 4 hours. Methanesulfonyl chloride (139 mg, 1.20 mmol) and triethylamine (0.83 ml, 6.00 mmol) were added to the reaction solution, and the mixture was further stirred for 1.5 hours. After the reaction solution was concentrated, it was diluted with ethyl acetate (200 ml) and 0.1N hydrochloric acid (200 ml). The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (3.42 g, 6.80 mmol, 56.6%) as a purple powder. Ή-NMR (300MHz, CDC1 3 ): 51.80 (3H, s), 2.26 (3H, s), 2.43 (3H, s), 3.03 (3H, s), 3.28 (2H, t, J = 6.8 Hz), 4.38 (2H, t, J = 6.9 Hz), 7.31 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.67 (1H, brs).
元素分析値: C18H21N306S4として計算値: C, 42.93; H, 4.20; , 8.34; S, 25.47 (%) 、 実測値: C, 43.22; H, 4.30; N, 8.53; S, 25.30 (%) . Elemental analysis: C 18 H 21 N 3 0 6 S 4 Calculated: C, 42.93; H, 4.20 ;, 8.34; S, 25.47 (%), Found: C, 43.22; H, 4.30 ; N, 8.53 ; S, 25.30 (%).
mp. 147.5-149.0°C mp.147.5-149.0 ° C
実施例 18 - 29 Examples 18-29
N-(8- {[(4-ヒドロキシシクロへキシル) メチル] スルファニル }- 5, 6 -ジメチル - 3 -ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン- 1-ィリデン)-4 -メ チルベンゼンスルホンアミド N- (8-{[(4-hydroxycyclohexyl) methyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene) -4-methylbutylsulfonamide
実施例 4 - 538で得た化合物 (1.15 g, 1.99 mmol) の メタノール (20 ml) お よび THF (6ml) に懸濁し、 D-カンファ一- 10-スルホン酸(46 mg, 0.199腿 ol) と 室温で 13 時間攪拌した。 反応液を濃縮後、 酢酸ェチル (200 ml)で希釈し、 水お よび飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥して濃縮した。 残渣を酢酸ェチ ルーへキサン、 2:8-4:6) に付し、酢酸ェチルおよびジェチルェ一テルで希釈して 析出物を濾取し、ジェチルェ一テルで洗浄後、乾燥して表題化合物 (747 mg, 1.51 mmol, 76.0»を濃紫色粉末として得た。 Example 4-A suspension of the compound (1.15 g, 1.99 mmol) obtained in 538 in methanol (20 ml) and THF (6 ml) was mixed with D-camphor-10-sulfonic acid (46 mg, 0.199 mol). The mixture was stirred at room temperature for 13 hours. The reaction solution was concentrated, diluted with ethyl acetate (200 ml), washed with water and saturated saline, dried over sodium sulfate and concentrated. The residue was subjected to ethyl hexane acetate, 2: 8-4: 6), diluted with ethyl acetate and ethyl ether, the precipitate was collected by filtration, washed with ethyl ether, dried and dried to give the title compound. (747 mg, 1.51 mmol, 76.0 »was obtained as a dark purple powder.
Ή-NMR (300MHz, CDC13): 61.32 (1H, brs), 1.46 (2H, brq, J= 10.8 Hz), Ή-NMR (300MHz, CDC1 3 ): 61.32 (1H, brs), 1.46 (2H, brq, J = 10.8 Hz),
1.50-1.70 (5H, m), 1.74 (2H, ra), 1.79 (3H, brq, J= 0.9 Hz), 2.26 (3H, brq, J= 0.6 Hz), 2.42 (3H, s), 2.87 (2H, d, J= 6.0 Hz), 4.02 (1H, brs), 7.27 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). 1.50-1.70 (5H, m), 1.74 (2H, ra), 1.79 (3H, brq, J = 0.9 Hz), 2.26 (3H, brq, J = 0.6 Hz), 2.42 (3H, s), 2.87 (2H , d, J = 6.0 Hz), 4.02 (1H, brs), 7.27 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.65 (1H, brs).
元素分析値: C22H27N304S3として計算値: C, 53.53; H, 5.51; , 8.51; S, 19.49 ( ) 、 実測値: C, 53.63; H, 5.84; N, 8.46; S, 18.70. Elemental analysis: Calculated as C 22 H 27 N 3 0 4 S 3: C, 53.53; H, 5.51;, 8.51; S, 19.49 (), Found: C, 53.63; H, 5.84 ; N, 8.46; S, 18.70.
mp. 191.0-193.0°C mp. 191.0-193.0 ° C
実施例 18-30 Example 18-30
N - {8- [ (3-シクロへキセン-トイルメチル) スルファニル] -5, 6-ジメチル -3-ォキ ソ- 2, 3-ジヒドロ- 1H-イミダゾ [5,l-c] [1, 4]チアジン-卜イリデン }_4-メチルベン ゼンスルホンアミド N- {8-[(3-cyclohexene-toylmethyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine -Triylidene} _4-Methylbenzene benzenesulfonamide
実施例 18- 29で得た化合物(191 mg, 0.387誦 ol) の ジクロロメタン (3 ml) 溶液に室温で Deoxo-Fluor™ (121 mg, 0.547腿 ol)を加えて 16 時間攪拌した。 さらに Deoxo- Fluor™ (46 mg, 0.232醒 ol) を加えて 9 時間攪拌した。 反応液に メタノール(1 ml)および飽和炭酸水素ナトリウム(5 ml)を加え、 酢酸ェチル(20 ml)で抽出した。 有機層を 2%炭酸水素ナトリウム水溶液、 0.1N塩酸、 水および 飽和食塩水で洗 ·し、 硫酸ナトリウムで乾燥後、 濃縮した。 残渣をメタノールで 希釈して得られた粗粉末をシリカゲル(10 g)のクロマトグラフィー (酢酸ェチル —へキサン、 2:8-3:7) に付し、酢酸ェチルーへキサン(1:3- 3:7) の画分を濃縮乾 固後、 メタノールで希釈した。 析出物を濾取し、 メタノールで洗浄後、 乾燥して 表題化合物(94 mg, 0.204 mmol, 53.7%)を赤色粉末として得た。 Compound (191 mg, 0.387 ol) of the compound obtained in Examples 18-29 in dichloromethane (3 ml) Deoxo-Fluor ™ (121 mg, 0.547 tmol) was added to the solution at room temperature, and the mixture was stirred for 16 hours. Further, Deoxo-Fluor ™ (46 mg, 0.232 ol) was added and stirred for 9 hours. Methanol (1 ml) and saturated sodium hydrogen carbonate (5 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate (20 ml). The organic layer was washed with a 2% aqueous sodium hydrogen carbonate solution, 0.1 N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue obtained by diluting the residue with methanol was chromatographed on silica gel (10 g) (ethyl acetate-hexane, 2: 8-3: 7) to give ethyl acetate-hexane (1: 3--3). : 7) was concentrated to dryness and then diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (94 mg, 0.204 mmol, 53.7%) as a red powder.
Ή-NMR (200MHz, CDC13): 1.32 (2H, m), 1.79 (3H, brq, J= 1.2 Hz), 1.84 (2H, m), 2.08 (2H, m), 2.18 (1H, m), 2.26 (3H, brq, J= 0.6 Hz), 2.42 (3H, s), 2.90 (2H, dd, J= 6.6, 2.2 Hz), 5.66 (2H, m), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): 1.32 (2H, m), 1.79 (3H, brq, J = 1.2 Hz), 1.84 (2H, m), 2.08 (2H, m), 2.18 (1H, m), 2.26 (3H, brq, J = 0.6 Hz), 2.42 (3H, s), 2.90 (2H, dd, J = 6.6, 2.2 Hz), 5.66 (2H, m), 7.28 (2H, d, J = 8.4 Hz ), 7.86 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C22H25N303S3として計算値: C, 55.55; H, 5.30; N, 8.83; S, 20.22 (%) 、 実測値: C, 55.36; H, 5.34; N, 8.92; S, 20.22 (%) . Elemental analysis: C 22 H 25 N 3 0 3 S 3 Calculated: C, 55.55; H, 5.30 ; N, 8.83; S, 20.22 (%), Found: C, 55.36; H, 5.34 ; N, 8.92; S, 20.22 (%).
mp. 184.0-185.5°C mp. 184.0-185.5 ° C
実施例 18-31 Example 18-31
5- { [5, 6 -ジメチル-ト ( { [4- (ァセチルァミノ)フエニル]スルホ二ル}ィミノ) -3 -ォ キソ -2, 3-ジヒド口- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル]スルファニ ル}- N-へキシルペンタンアミド 5-{[5,6-dimethyl-({[4- (acetylamino) phenyl] sulfonyl} imino) -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1, 4] thiazin-8-yl] sulfanyl} -N-hexylpentanamide
水素化ナトリウム(9.74 mg, 0.243匪 ol) (60%油性)のテトラヒドロフラン(0.50 ml)の懸濁液に、実施例 6-35で得られた 5-[1-ァミノ- 5, 6 -ジメチル- 3-ォキソ -3H - イミダゾ [5,卜 c] [1, 4]チアジン- 8 -ィル]スルファニル] -N-へキシルぺンタンアミ ド (50.0 mg, 0.121 mmol) のテトラヒドロフラン (0.50 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 4_ (ァセチルァミノ)ベンゼンスルホニル クロリド (42.7 mg, 0.183匪 ol) のテトラヒドロフラン (0.50 ml) 溶液を加え、 さらに 室温で 16時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (48.9mg, 66%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融 点 148- 150°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (9.74 mg, 0.243 marl ol) (60% oil) in tetrahydrofuran (0.50 ml) was added 5- [1-amino-5,6-dimethyl-methyl obtained in Example 6-35. A solution of 3-oxo-3H-imidazo [5, tric] [1,4] thiazine-8-yl] sulfanyl] -N-hexylpentanamide (50.0 mg, 0.121 mmol) in tetrahydrofuran (0.50 ml) was prepared. Then, the mixture was stirred for 5 minutes under ice cooling. To this was added a solution of 4_ (acetylamino) benzenesulfonyl chloride (42.7 mg, 0.183 bandol) in tetrahydrofuran (0.50 ml), and the mixture was further stirred at room temperature for 16 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (48.9 mg, 66%) as a dark red crystals. Recrystallized from ethyl acetate-getyl ether and melted Dark red crystals at a point 148-150 ° C were obtained.
IR (KBr) V: 3270, 3108, 2955, 2928, 2863, 1755, 1728, 1645, 1593, 1557, 1539, 1505 cur1. IR (KBr) V: 3270, 3108, 2955, 2928, 2863, 1755, 1728, 1645, 1593, 1557, 1539, 1505 cur 1 .
Ή-NMR (CDC13) : δ 0.882 (3Η, t, J=6.9Hz), 1.24-1.36 (6H, m), 1.44-1.56 (2H, m), 1.65-1.74 (4H, m), 1.78 (3H, d, J=1.2 Hz), 2.14-2.22 (2H, m) , 2.21 (3H, s), 2.26 (3H, d, J=0.9 Hz), 2.94 (2H, t, J-6.3 Hz), 3.24 (2H, q, J=5.7 Hz), 5.66 (1H, bs), 7.67 (2H, d, J=9.0 Hz), 7.90 (2H, d, J=8.7 Hz), 8.00 (1H, bs), 9.54 (1H, bs). Ή-NMR (CDC1 3): δ 0.882 (3Η, t, J = 6.9Hz), 1.24-1.36 (6H, m), 1.44-1.56 (2H, m), 1.65-1.74 (4H, m), 1.78 ( 3H, d, J = 1.2 Hz), 2.14-2.22 (2H, m), 2.21 (3H, s), 2.26 (3H, d, J = 0.9 Hz), 2.94 (2H, t, J-6.3 Hz), 3.24 (2H, q, J = 5.7 Hz), 5.66 (1H, bs), 7.67 (2H, d, J = 9.0 Hz), 7.90 (2H, d, J = 8.7 Hz), 8.00 (1H, bs), 9.54 (1H, bs).
元素分析値: C27H37N505S3として Elementary analysis: as C 27 H 37 N 5 0 5 S 3
計算値 (%) : C, 53.35; H, 6.14; N, 11.52; S, 15.83. Calculated value (%): C, 53.35; H, 6.14; N, 11.52; S, 15.83.
実測値 (%) : C, 53.03; H, 6.17'; N, 11.43; S, 15.58. Found (%): C, 53.03; H, 6.17 '; N, 11.43; S, 15.58.
実施例 18 - 32 Examples 18-32
5- [(6 -へキシル -5-メチル- (4-メチルフエニル)スルホニル]イミノ}_3-ォキ ソ -3H-イミダゾ [5,卜 c] [1,4]チアジン- 8-ィル)スルファニル]吉草酸 5-[(6-hexyl-5-methyl- (4-methylphenyl) sulfonyl] imino} _3-oxo-3H-imidazo [5, tric] [1,4] thiazin-8-yl) sulfanyl ] Valeric acid
実施例 -542で得られたェチル 5- [ (6-へキシル- 5-メチル -1- { [ (4 -メチルフェ ニル)スルホニル]イミノ } -3 -ォキソ署ィミダゾ [5,1-c] [1,4]チアジン- 8 -ィル) スルファニル]ペンタノエート (500 mg, 0.862腿 ol) のエタノール (10.0 ml) 及びテトラヒドロフラン(10.0 ml)溶液に、 1N水酸化ナトリウム水溶液(2.59 ml) を加えた。反応混合物を室温で 2.5時間かき混ぜた後、溶媒を減圧下に留去した。 残渣に IN塩酸を加えて、 酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、溶媒を減圧下に留去して題記化合物(472 mg, 99¾)を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 181- 183°Cの暗赤色結晶を得 た。 Ethyl 5-[(6-hexyl-5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-inosimidazo [5,1-c] [obtained in Example -542 To a solution of 1,4] thiazin-8-yl) sulfanyl] pentanoate (500 mg, 0.862 mol) in ethanol (10.0 ml) and tetrahydrofuran (10.0 ml) was added a 1N aqueous sodium hydroxide solution (2.59 ml). After stirring the reaction mixture at room temperature for 2.5 hours, the solvent was distilled off under reduced pressure. IN hydrochloric acid was added to the residue, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (472 mg, 99¾) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 181-183 ° C.
IR (KBr) V: 3400-2400, 2926, 2859, 1725, 1657, 1603, 1559 cm-1.IR (KBr) V: 3400-2400, 2926, 2859, 1725, 1657, 1603, 1559 cm- 1 .
-謹 (CDC13) : (50.892 (3H, t, J=6.9Hz), 1.24-1.36 (6H, m), 1.38-1.52 (2H, m), 1.72-1.80 (4H, in), 2.11 (2H, t, J=8.1 Hz), 2.26 (3H, s), 2.38-2.44 (2H, m), 2.42 (3H, s), 2.96 (2H, t, J=6.6 Hz), 7.29 (2H, d, J=8.7 Hz), 7.85 (2H, d, J=8.1 Hz), 9.66 (1H, bs). -謹(CDC1 3): (50.892 ( 3H, t, J = 6.9Hz), 1.24-1.36 (6H, m), 1.38-1.52 (2H, m), 1.72-1.80 (4H, in), 2.11 (2H , t, J = 8.1 Hz), 2.26 (3H, s), 2.38-2.44 (2H, m), 2.42 (3H, s), 2.96 (2H, t, J = 6.6 Hz), 7.29 (2H, d, J = 8.7 Hz), 7.85 (2H, d, J = 8.1 Hz), 9.66 (1H, bs).
元素分析値: C25H33N305S3として 計算値 (¾) : C, 54.42; H, 6.03; N, 7.62; S, 17.43. Elemental analysis: as C 25 H 33 N 3 0 5 S 3 Calculated value (¾): C, 54.42; H, 6.03; N, 7.62; S, 17.43.
実測値 (¾) : C, 54.33; H, 5.82; , 7.61; S, 17.57. Found (¾): C, 54.33; H, 5.82;, 7.61; S, 17.57.
実施例 18-33 Example 18-33
5 - [(6-ベンジル- 5-メチル-卜 {[(4-メチルフエニル)スルホニル]イミノ}-3-ォキ ソ - 3H -ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル)スルファニル]吉草酸 5-[(6-benzyl-5-methyl-{{((4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5, 1-c] [1, 4] thiazine-8 Yl) sulfanyl] valeric acid
実施例 4-543で得られたェチル 5- [ (6-ベンジル- 5-メチル-卜 { [ (4-メチルフエ ニル)スルホニル]ィミノ }-3-ォキソ - 3H-ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル) スルファニル]ペンタノエート (500 mg, 0.854 mmol) のエタノール (10.0 ml) 及びテトラヒドロフラン(10.0 ml)溶液に、 1 水酸化ナトリゥム水溶液(2.56ml) を加えた。反応混合物を室温で 2.5時間かき混ぜた後、溶媒を減圧下に留去した。 残渣に 1N塩酸を加えて、 酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (478 mg, ca. 100%) を暗赤色結晶とし て得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 182-184°Cの暗赤色結 晶を得た。 Ethyl 5-[(6-benzyl-5-methyl-{{((4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5,1-c] obtained in Example 4-543 To a solution of [1,4] thiazin-8-yl) sulfanyl] pentanoate (500 mg, 0.854 mmol) in ethanol (10.0 ml) and tetrahydrofuran (10.0 ml) was added aqueous sodium hydroxide solution (2.56 ml). After stirring the reaction mixture at room temperature for 2.5 hours, the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after solvent was obtained as title compound was evaporated under reduced pressure (478 mg, ca. 100%) of dark red crystals. Recrystallization from ethyl acetate-getyl ether gave a dark red crystal with a melting point of 182-184 ° C.
IR (KBr) v: 3400-2800, 2926, 1734, 1655, 1618, 1561, 1493 cm—1. IR (KBr) v: 3400-2800, 2926, 1734, 1655, 1618, 1561, 1493 cm— 1 .
¾-NMR (CDC13) : δ 1.60-1.72 (4Η, m), 2.32 (2H, t, J-6.9 Hz), 2.37 (3H, s), ¾-NMR (CDC1 3): δ 1.60-1.72 (4Η, m), 2.32 (2H, t, J-6.9 Hz), 2.37 (3H, s),
2.42 (3H, s), 2.85 (2H, t, J=6.9 Hz), 3.50 (2H, s), 7.18-7.38 (7H, m), 7.852.42 (3H, s), 2.85 (2H, t, J = 6.9 Hz), 3.50 (2H, s), 7.18-7.38 (7H, m), 7.85
(2H, d, J-8.4 Hz), 9.68 (1H, bs). (2H, d, J-8.4 Hz), 9.68 (1H, bs).
, 元素分析値: C26H27N305S3として , Elemental analysis: as C 26 H 27 N 3 0 5 S 3
計算値 (¾) : C, 55.99; H, 4.88; N, 7.53; S, 17.25. Calculated (¾): C, 55.99; H, 4.88; N, 7.53; S, 17.25.
実測値 (%) : C, 55.91; H, 5.03; N, 7.62; S, 17.36. Found (%): C, 55.91; H, 5.03; N, 7.62; S, 17.36.
実施例 18-34 Example 18-34
N-{8-[(5-bドロキシシクロォクチル) スルファニル] - 5, 6-ジメチル- 3 -ォキソ - 2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, ]チアジン- 1-ィリデン } -4-メチルベンゼ ンスルホンアミド N- {8-[(5-bdroxycyclooctyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,] thiazine -1-ylidene} -4-methylbenzenesulfonamide
実施例 4- 540で得た化合物(291 mg, 0.529删 ol)の THF -メタノール (1:1, .5 ml) 懸濁液に IN水酸化ナトリウム水溶液(2.38 ml, 2.38腿01) を加え、 室温で 4時間攪拌した。 反応液に 1N塩酸 (2.38 ml)を加えて室温で 4時間攪拌した。析 出物を濾取し、 メ夕ソ一ルー水 (1:1) およびメタノールで洗浄後、乾燥して表題 化合物(261 mg, 0.514醒 ol, 97.2%)を灰赤色粉末として得た。 To a suspension of the compound (291 mg, 0.529 mol) obtained in Example 4-540 in THF-methanol (1: 1.5 ml) was added IN aqueous sodium hydroxide solution (2.38 ml, 2.38 thigh 01). The mixture was stirred at room temperature for 4 hours. 1N Hydrochloric acid (2.38 ml) was added to the reaction solution, and the mixture was stirred at room temperature for 4 hours. The precipitate is collected by filtration, washed with methanol (1: 1) and methanol, dried, and dried. The compound (261 mg, 0.514 ol, 97.2%) was obtained as a gray-red powder.
Ή-N R (200MHz, CDC13): δ 1.34 (1Η, brs), 1.60-1.80 (8H, m), 1.80 (3H, s), 1.85 (2H, m), 1.96 (2H, m), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 3.52 (1H, br), 3.82 (1H, br), 7.28 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). Ή-NR (200MHz, CDC1 3 ): δ 1.34 (1Η, brs), 1.60-1.80 (8H, m), 1.80 (3H, s), 1.85 (2H, m), 1.96 (2H, m), 2.26 ( 3H, q, J = 1.2 Hz), 2.42 (3H, s), 3.52 (1H, br), 3.82 (1H, br), 7.28 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C23H29N304S3 ·0.5Η20として計算値: C, 53.46; Η, 5.85; Ν, 8.13; S, 18.62 (%) 、 実測値: C, 53.39; Η, 5.83; Ν, 8.00; S, 18.92 (%) . Elemental analysis: C 23 H 29 N 3 0 4 S 3 · 0.5Η 2 0 Calculated: C, 53.46; Η, 5.85 ; Ν, 8.13; S, 18.62 (%), Found: C, 53.39; Η , 5.83; Ν, 8.00; S, 18.92 (%).
mp. 191.5-192.5°C mp. 191.5-192.5 ° C
実施例 18-35 Example 18-35
5- [(6-へキシル -5-メチル -1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキ ソ -3H-イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル] - N-へキシルペンタ ンアミド 5-[(6-hexyl-5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5,1-c] [1,4] thiazine- 8-yl) sulfanyl] -N-hexylpentanamide
実施例 18-32で得られた 5 - [ (6-へキシル- 5-メチル -1- { [ (4-メチルフエニル)ス ルホニル]イミノ}- 3-ォキソ - 3H-イミダゾ [5,卜 c] [1, 4]チアジン- 8-ィル)スルフ ァニル]吉草酸(100 nig, 0.181 mmol)と卜へキシルァミン(0.0264 ml, 0.199腿 ol) の N,N-ジメチルホルムアミド (5.00 ml)溶液に、 シァノりん酸ジェチル (以下、 DEPCと略記することがある) (0.0322 ml, 0.199 mmol) 続いてトリェチルァミン 5-[(6-Hexyl-5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5, tric] obtained in Examples 18-32. [1,4] Thiazin-8-yl) sulfanyl] valeric acid (100 nig, 0.181 mmol) and trihexylamine (0.0264 ml, 0.199 tmol) in N, N-dimethylformamide (5.00 ml) solution Getyl cyanophosphate (hereinafter sometimes abbreviated as DEPC) (0.0322 ml, 0.199 mmol) followed by triethylamine
(0.0278 ml, 0.199 mmol) を加えた。反応混合物を室温で 18時間かき混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧 下に留去して題記化合物 (54.8mg, 48 ) を暗赤色結晶として得た。 酢酸ェチル- ジェチルェ一テルから再結晶して融点 98.0-100°Cの暗赤色結晶を得た。 (0.0278 ml, 0.199 mmol) was added. After stirring the reaction mixture at room temperature for 18 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the title compound and the solvent was distilled off under reduced pressure (54.8 mg, 48) as a dark red crystals. The crystals were recrystallized from ethyl acetate-deethyl ether to obtain dark red crystals having a melting point of 98.0-100 ° C.
IR (KBr) リ: 3260, 2955, 2928, 2859, 1734, 1653, 1615, 1559 cm"1. IR (KBr): 3260, 2955, 2928, 2859, 1734, 1653, 1615, 1559 cm " 1 .
Ή-NMR (CDC13) : δ 0.840 - 0.940 (6Η, m), 1.-1.38 (14H, m), 1.38-1.56 (4H, m), 1.68-1.84 (4H, m), 2.10 (2H, t, J=8.1 Hz), 2.20 (2H, t, J=7.21 Hz), 2.25 (3H, s), 2.42 (3H, s), 2.96 (2H, t, 1=1.2 Hz), 3.24 (2H, q, J=5.4 Hz), 5.48 (1H, bs), 7.29 (2H, d, J-8. Hz), 7.84 (2H, d, J-8.1 Hz), 9.62 (1H, bs). Ή-NMR (CDC1 3): δ 0.840 - 0.940 (6Η, m), 1.-1.38 (14H, m), 1.38-1.56 (4H, m), 1.68-1.84 (4H, m), 2.10 (2H, t, J = 8.1 Hz), 2.20 (2H, t, J = 7.21 Hz), 2.25 (3H, s), 2.42 (3H, s), 2.96 (2H, t, 1 = 1.2 Hz), 3.24 (2H, q, J = 5.4 Hz), 5.48 (1H, bs), 7.29 (2H, d, J-8.Hz), 7.84 (2H, d, J-8.1 Hz), 9.62 (1H, bs).
元素分析値: C31H46N404S3として Elemental analysis: as C 31 H 46 N 4 0 4 S 3
計算値 (%) : C, 58.64; H, 7.30; N, 8.82; S, 15.15. Calculated value (%): C, 58.64; H, 7.30; N, 8.82; S, 15.15.
実測値 (%) : C, 58.57; H, 7.41; N, 8.81; S, 15.30. 実施例 18 - 36 Found (%): C, 58.57; H, 7.41; N, 8.81; S, 15.30. Examples 18-36
5 - [ (6-ベンジル- 5-メチル - 1 _ { [ (4-メチルフエニル)スルホニル]イミノ } -3-ォキ ソ- 3H -ィミダゾ [5, 1-c] [1,4]チアジン -8 -ィル)スルファニル] -N-へキシルぺンタ ンアミド 5-[(6-benzyl-5-methyl-1 _ {[((4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5,1-c] [1,4] thiazine-8 -Yl) sulfanyl] -N-hexylpentanamide
実施例 18-33で得られた 5- [ (6-ベンジル- 5-メチル -1- { [ (4-メチルフエニル)ス ルホニル]ィミノ }-3-ォキソ - 3H-ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルフ ァニル]吉草酸(100 mg, 0.179腿 ol)と 1-へキシルァミン(0.0261 ml, 0.197匪 ol) の N,N -ジメチルホルムアミド (5.00ml) 溶液に、 シァノりん酸ジェチル (0.0322 ml, 0.199腿 ol) 続いて、 トリェチルァミン (0.0275 ml, 0.197mmol) を加えた。 反応混合物を室温で 18時間かき混ぜた後、水を加えて酢酸ェチルで抽出した。抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(60.2 mg, 53%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 107- 109 の暗赤色結晶を得た。 5-[(6-benzyl-5-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-3H-imidazo [5,1-c] obtained in Example 18-33 N, N-Dimethylformamide (5.00ml) solution of [1,4] thiazin-8-yl) sulfanyl] valeric acid (100 mg, 0.179 t ol) and 1-hexylamine (0.0261 ml, 0.197 marl ol) To the mixture was added getyl cyanophosphate (0.0322 ml, 0.199 t), followed by triethylamine (0.0275 ml, 0.197 mmol). After stirring the reaction mixture at room temperature for 18 hours, water was added and extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), and the solvent was evaporated under reduced pressure to give the title compound (60.2 mg, 53%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 107-109.
IR (KBr) V: 3250, 2928, 2857, 1742, 1655, 1615, 1557, 1495 cnf1. IR (KBr) V: 3250, 2928, 2857, 1742, 1655, 1615, 1557, 1495 cnf 1 .
'H-NMR (CDC13) : (50.879 (3H, t, J=6.2 Hz), 1.24-1.38 (6H, m), 1.38-1.55 (2H, 'H-NMR (CDC1 3) : (50.879 (3H, t, J = 6.2 Hz), 1.24-1.38 (6H, m), 1.38-1.55 (2H,
1) , 1.55-1.80 (4H, m), 2.12 (2H, t, J=6.6 Hz), 2.36 (3H, s), 2.42 (3H, s),1), 1.55-1.80 (4H, m), 2.12 (2H, t, J = 6.6 Hz), 2.36 (3H, s), 2.42 (3H, s),
2.87 (2H, t, J=7.0 Hz), 3.22 (2H, q, J=6.8Hz), 3.49 (2H, s), 5.482 (1H, bs),2.87 (2H, t, J = 7.0 Hz), 3.22 (2H, q, J = 6.8 Hz), 3.49 (2H, s), 5.482 (1H, bs),
7.16-7.40 (7H, m), 7.84 (2H, d, J=8.4 Hz). 7.16-7.40 (7H, m), 7.84 (2H, d, J = 8.4 Hz).
元素分析値: C32H4。N404S3'0.2H20として Elemental analysis: C 32 H 4. N 4 0 4 S 3 '0.2H 20
計算値 (%) : C, 59.63; H, 6.32; N, 8.69. Calculated value (%): C, 59.63; H, 6.32; N, 8.69.
実測値 (¾) : C, 59.53; H, 6.27; N, 8.83. Found (¾): C, 59.53; H, 6.27; N, 8.83.
実施例 18 - 37 Examples 18-37
N- [8-({[ (2S, 4R) -4-ヒドロキシピロリジニル] メチル }スルファニル) -5, 6-ジメ チル- 3-ォキソ -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン-卜ィリデ ン] -4-メチルベンゼンスルホンアミド 塩酸塩 N- [8-({[(2S, 4R) -4-hydroxypyrrolidinyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1 -c] [1,4] thiazine-trilidene] -4-methylbenzenesulfonamide hydrochloride
実施例 4-555で得た化合物の粉末(416 mg, 0.716匪 ol) を TFA (1.0 ml)に 溶解し、 室温で 90分間放置した。 反応液をトルエン(20 ml)で希釈して濃縮乾 固し、 残渣にトルエン(20 ml)を加えて濃縮した。 得られた残渣を THF -エタノー ル(1:1, 20 ml)に溶解し、 1N塩酸 (1.43 ml)を加えて濃縮し、 残渣にァセトニト リルを加えて濃縮して赤色の懸濁液を得た。 これを THFおよび酢酸ェチルで希釈 した。析出物を濾取し、酢酸ェチルで洗浄後、乾燥して表題化合物(298 mg, 0.576 mmol, 80.5%)を赤色粉末として得た。 The powder (416 mg, 0.716 bandol) of the compound obtained in Example 4-555 was dissolved in TFA (1.0 ml) and left at room temperature for 90 minutes. The reaction solution was diluted with toluene (20 ml) and concentrated to dryness. Toluene (20 ml) was added to the residue and concentrated. The obtained residue was dissolved in THF-ethanol (1: 1, 20 ml), 1N hydrochloric acid (1.43 ml) was added, and the mixture was concentrated. Lil was added and concentrated to give a red suspension. This was diluted with THF and ethyl acetate. The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (298 mg, 0.576 mmol, 80.5%) as a red powder.
Ή-NMR (200MHz, DMSO— d6): άΐ.78 (1Η, m), 1.81 (3H, s), 2.06 (1H, m), 2.18 (3H, q, J= 0.8 Hz), 2.39 (3H, s), 3.08 (1H, m), 3.20-3.40 (3H, m), 3.85 (1H, m), 4.4K1H, brs), 5.44 (1H, br), 7.39 (2H, d, J= 8.2 Hz), 7.87 (2H, d, J= 8.0 Hz), 9.24 (1H, m), 9.59 (1H, m), 11.76 (1H, m). Ή-NMR (200 MHz, DMSO— d 6 ): άΐ.78 (1Η, m), 1.81 (3H, s), 2.06 (1H, m), 2.18 (3H, q, J = 0.8 Hz), 2.39 (3H , s), 3.08 (1H, m), 3.20-3.40 (3H, m), 3.85 (1H, m), 4.4K1H, brs), 5.44 (1H, br), 7.39 (2H, d, J = 8.2 Hz ), 7.87 (2H, d, J = 8.0 Hz), 9.24 (1H, m), 9.59 (1H, m), 11.76 (1H, m).
元素分析値: C20H24N404S3.HC1.0.5H20として計算値: C, 45.66; H, 4.98; N, 10.65; S, 18.29; C1, 6.74 (%) 、 実測値: C, 45.66; H, 4.90; N, 10.67; S, 17.90; C1, 6.60 (%) . Elemental analysis: C 20 H 24 N 4 0 4 S 3 .HC1.0.5H 2 0 Calculated: C, 45.66; H, 4.98 ; N, 10.65; S, 18.29; C1, 6.74 (%), Found : C, 45.66; H, 4.90; N, 10.67; S, 17.90; C1, 6.60 (%).
mp. 188.0-191.0°C mp.188.0-191.0 ° C
実施例 18 - 38 Examples 18-38
(2S) -4- [(5, 6 -ジメチル- 1 - {[ (4-メチルフエニル)スルホニル]ィミノ }_3-ォキソ -2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン -8-ィル)スルファニル ]-2 -ピ 口リジンカルボン酸メチル 塩酸塩 (2S) -4-[(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} _3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] Thiazine-8-yl) sulfanyl] -2-pimethyl lysinecarboxylate hydrochloride
実施例 4-545で得た化合物の粉末(776 mg, 1.27 mmol) を TFA (1.0 ml)に溶 解し、室温で 2時間放置した。 反応液をトルエン(20 ml)で希釈して濃縮乾固し、 残渣にトルエン(20 ml)を加えて濃縮した。 得られた残渣を THF -エタノール(1:1, 20 ml)に溶解し、 1 塩酸 (1.5 ml)を加えて濃縮し、 残渣に THFおよび酢酸ェチ ルを加えて濃縮して赤色の懸濁液を得た。析出物を濾取し、 THF—酢酸ェチル(1:1) および酢酸ェチルで洗浄後、 乾燥して表題化合物(518 mg, 0.950 mmol, 74.8%) を赤紫色粉末として得た。 The powder (776 mg, 1.27 mmol) of the compound obtained in Example 4-545 was dissolved in TFA (1.0 ml) and left at room temperature for 2 hours. The reaction solution was diluted with toluene (20 ml), concentrated to dryness, and the residue was concentrated by adding toluene (20 ml). The obtained residue was dissolved in THF-ethanol (1: 1, 20 ml), concentrated by adding 1 hydrochloric acid (1.5 ml), and concentrated by adding THF and ethyl acetate to the residue. A liquid was obtained. The precipitate was collected by filtration, washed with THF-ethyl acetate (1: 1) and ethyl acetate, and dried to give the title compound (518 mg, 0.950 mmol, 74.8%) as a red-purple powder.
Ή-NMR (200MHz, DMSO— d6): (51.80 (3H, s) , 2.10 (1Η, m), 2.17 (3H, s), 2.39 (3H, s), 2.80 (1H, dt, J= 13.6, 6.8 Hz), 3.24 (1H, m), 3.75 (1H, m), 3.76/3.78 (3H, s), 4.13 (1H, brq, J= 7.2 Hz), 4.54 (1H, t, J= 8.2 Hz), 7.39 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 10.41 (3H, br). Ή-NMR (200 MHz, DMSO—d 6 ): (51.80 (3H, s), 2.10 (1Η, m), 2.17 (3H, s), 2.39 (3H, s), 2.80 (1H, dt, J = 13.6) , 6.8 Hz), 3.24 (1H, m), 3.75 (1H, m), 3.76 / 3.78 (3H, s), 4.13 (1H, brq, J = 7.2 Hz), 4.54 (1H, t, J = 8.2 Hz) ), 7.39 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 10.41 (3H, br).
元素分析値: C2IH24N405S3.HC1.0.3H20として計算値: C, 45.82;H, 4.69; N, 10.18; S, 17.47; CI, 6.44 (%) 、 実測値: C, 45.82; H, 4.71; , 10.21; S, 17.46; CI, 6.61 (%) . 即. 188.0-191.0°C Elemental analysis: C 2I H 24 N 4 0 5 S 3 .HC1.0.3H 2 0 Calculated: C, 45.82; H, 4.69 ; N, 10.18; S, 17.47; CI, 6.44 (%), Found : C, 45.82; H, 4.71 ;, 10.21; S, 17.46; CI, 6.61 (%). Immediate. 188.0-191.0 ° C
実施例 18 - 39 Examples 18-39
(2S,4R)- 1- (tert-ブトキシカルポ二ル)- 4-[(5,6 -ジメチル- 1- {[(4-メチルフエ二 ル)スルホニル]ィミノ}-3-ォキソ - 2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1,4]チア ジン- 8-ィル)スルファニル] -2-ピロリジン力ルポン酸 (2S, 4R) -1- (tert-Butoxycarbonyl) -4-[(5,6-dimethyl-1-{{((4-methylphenyl) sulfonyl] imino} -3-oxo-3,2-oxo Dihydro-1H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] -2-pyrrolidine
実施例 4-545で得た化合物(317 mg, 0.52 mmol)の THF -メタノール-水 (1:1:1, 9 ml)懸濁液に室温で攪拌しながら水酸化カリウム (85%, 172 mg, 2.60 mmol) を 加え、 反応液を 1時間攪拌した。 反応液に 1N塩酸 (2.6 ml)を加え、 酢酸ェチル (40ml)で抽出した。有機層を本および飽和食塩水で順次洗浄し、 硫酸ナトリウム で乾燥後、 濃縮して濃赤色油状物を得た。 これをシリカゲル(10 g)のクロマトグ ラフィー(酢酸ェチル—へキサン、 4:6, 5:5)に付し、酢酸ェチルーへキサン(5 :5) の画分を濃縮乾固後、 酢酸ェチルおよびジェチルェ一テルで希釈した。 析出物を 濾取し、 ジェチルェ一テルで洗浄後、 乾燥して表題化合物(108 mg, 0.182 mmol, 35.1¾)を赤色粉末として得た。 Potassium hydroxide (85%, 172 mg) was added to a suspension of the compound obtained in Example 4-545 (317 mg, 0.52 mmol) in THF-methanol-water (1: 1: 1, 9 ml) at room temperature with stirring. , 2.60 mmol) was added and the reaction was stirred for 1 hour. 1N Hydrochloric acid (2.6 ml) was added to the reaction solution, and extracted with ethyl acetate (40 ml). The organic layer was washed successively with a main solution and saturated saline, dried over sodium sulfate, and concentrated to obtain a dark red oil. This was subjected to silica gel (10 g) chromatography (ethyl acetate-hexane, 4: 6, 5: 5), and the ethyl acetate-hexane (5: 5) fraction was concentrated to dryness. Diluted with Jettlether. The precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (108 mg, 0.182 mmol, 35.1¾) as a red powder.
Ή-NMR (200MHz, CDC13): δ 1.47 (6Η, s), 1.50 (3H, s), 1.77 (3H, s), 2.25 (3H, s), 2.43 (3H, s), 3.47 (1H, m), 3,96 (1H, m), 3.98 (1H, m), 4.46 (1H, m), 7.32 (2H, d, J= 8.4 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.69 (1H, br). Ή-NMR (200MHz, CDC1 3 ): δ 1.47 (6Η, s), 1.50 (3H, s), 1.77 (3H, s), 2.25 (3H, s), 2.43 (3H, s), 3.47 (1H, m), 3,96 (1H, m), 3.98 (1H, m), 4.46 (1H, m), 7.32 (2H, d, J = 8.4 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.69 (1H, br).
元素分析値: C25H3。N407S3として計算値: C, 50.49; H, 5.08; N, 9.42; S, 16.17 (%) 、 実測値: C, 50.29; H, 5.31; N, 9.29; S, 16.03 (%) . Elemental analysis: C 25 H 3. N 4 0 7 S 3 Calculated: C, 50.49; H, 5.08 ; N, 9.42; S, 16.17 (%), Found: C, 50.29; H, 5.31 ; N, 9.29; S, 16.03 (%) .
mp. 152.0-155.0°C (分解) mp. 152.0-155.0 ° C (decomposition)
実施例 18 - 40 Examples 18-40
N - [8- ({[(2S,4R)- 1-ァセチル- 4 -ヒドロキシピロリジニル]メチル }スルファ二 ル)- 5, 6 -ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,1- c] [1, 4]チアジン 実施例 18-37 で得た化合物(214 mg, 0.41 匪 ol)および無水酢酸(0.12 ml, N- [8-({[(2S, 4R) -1-acetyl-4-hydroxypyrrolidinyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H- Imidazo [5,1-c] [1,4] thiazine The compound obtained in Example 18-37 (214 mg, 0.41 ol) and acetic anhydride (0.12 ml,
1. 4 mmol) の THF (5 ml)の懸濁液に室温で攪拌しながらトリ工チルァミン(0.115 ml, 0.828 mmol)を加え、 反応液を 3時間攪拌した。 反応液を酢酸ェチル(40 ml) で希釈し、 0.2N塩酸— 5%食塩水および飽和食塩水で順次洗浄し、 硫酸ナトリウム で乾燥後、 濃縮して得られた濃赤色油状物にメタノールを加えた。 析出物を濾取 し、 メタノ一ルで洗浄後、 乾燥して表題化合物 (179 rag, 0.342 mmol, 82.7 )を 赤色粉末として得た。 To a suspension of 1.4 mmol) in THF (5 ml) was added triethylamine (0.115 ml, 0.828 mmol) with stirring at room temperature, and the reaction solution was stirred for 3 hours. The reaction mixture was diluted with ethyl acetate (40 ml), washed successively with 0.2N hydrochloric acid—5% brine and saturated brine, dried over sodium sulfate, concentrated, and methanol was added to the resulting dark red oil. Was. Precipitate is collected by filtration After washing with methanol and drying, the title compound (179 rag, 0.342 mmol, 82.7) was obtained as a red powder.
Ή-NMR (200MHz, CDC13): δ 1.79 (3Η, s), 2.04 (3H, s), 2.12 (2H, m), 2.25 (3H, s), 2.43 (3H, s), 3.32 (1H, dd, J= 13.0, 6.8 Hz), 3.42 (1H, m), 3.46 (1H, m), 3.55 (1H, dd, J= 11.0, 4.4 Hz), 4.47 (1H, dd, J= 6.6, 4.0 Hz), 4.54 (1H, m), 7.30 (2H, d, J= 8.0 Hz), 7.83 (2H, d, J= 8.0 Hz), 9.67 (1H, br). 元素分析値: ' C22H26N405S3として計算値: C, 50.56; H, 5.01; , 10.72; S, 18.40 Ή-NMR (200MHz, CDC1 3 ): δ 1.79 (3Η, s), 2.04 (3H, s), 2.12 (2H, m), 2.25 (3H, s), 2.43 (3H, s), 3.32 (1H, dd, J = 13.0, 6.8 Hz), 3.42 (1H, m), 3.46 (1H, m), 3.55 (1H, dd, J = 11.0, 4.4 Hz), 4.47 (1H, dd, J = 6.6, 4.0 Hz ), 4.54 (1H, m), 7.30 (2H, d, J = 8.0 Hz), 7.83 (2H, d, J = 8.0 Hz), 9.67 (1H, br). Elemental analysis: 'C 22 H 26 N 4 0 5 S 3 calculated: C, 50.56; H, 5.01 ;, 10.72; S, 18.40
(%) 、 実測値: C, 50.61; H, 5.14; N, 10.76; S, 18.45 (%) . (%), Found: C, 50.61; H, 5.14; N, 10.76; S, 18.45 (%).
即. 208.5-209.5°C Immediately. 208.5-209.5 ° C
実施例 18-41 Example 18-41
メ夕ンスルホン酸 5- [ (5, 6 -ジメチル -1 - { [ (4-メチルフエニル)スルホニル]ィミ ノ}- 3-ォキソ - 2,3-ジヒドロ- 1H-イミダゾ [5,1- c] [1, 4]チアジン- 8-ィル)スルフ ァニル]シクロォクチル Maleic sulfonic acid 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] cyclooctyl
実施例 18-34で得た化合物 (286 mg, 0.563 mmol)およびメタンスルホニルク 口リド (72.3 mg, 0.620 mmol)の THF (6 ml)溶液に 0Tで攪拌しながらトリエチ ルァミン(0.204 ml, 1.46讓 ol)の THF (1 ml)溶液を 20分間かけて滴下した。 反 応液を 0°Cで 2.5時間攪拌した。 反応液を酢酸ェチル(30 ml)で希釈し、 0.05N塩 酸、 水および飽和食塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮乾固して 表題化合物 (330 mg, 0.563 mmol, quant. )を紫色粉末として得た。 To a solution of the compound (286 mg, 0.563 mmol) obtained in Example 18-34 and methanesulfonyl chloride (72.3 mg, 0.620 mmol) in THF (6 ml) was stirred at 0T while stirring triethylamine (0.204 ml, 1.46%). ol) in THF (1 ml) was added dropwise over 20 minutes. The reaction solution was stirred at 0 ° C for 2.5 hours. The reaction mixture was diluted with ethyl acetate (30 ml), washed sequentially with 0.05N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to dryness to give the title compound (330 mg, 0.563 mmol, quant. ) Was obtained as a purple powder.
Ή— NMR (200MHz, CDC13): δ 1.12 (4H, m), 1.80 (3H, s), 2.01 (8H, m), 2.26 (3H, s), 2.42 (3H, s), 3.01/3.02 (3H, s), 3.47 (1H, m), 4.86 (1H, m), 7.29 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή- NMR (200MHz, CDC1 3) : δ 1.12 (4H, m), 1.80 (3H, s), 2.01 (8H, m), 2.26 (3H, s), 2.42 (3H, s), 3.01 / 3.02 ( 3H, s), 3.47 (1H, m), 4.86 (1H, m), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C24H31N306S4として計算値: C, 49.21; H, 5.33; N, 7.17; S, 21.90 (%) 、 実測値: C, 49.05; H, 5.31; N, 7.27; S, 22.03 (%) . Elemental analysis: C 24 H 31 N 3 0 6 S 4 Calculated: C, 49.21; H, 5.33 ; N, 7.17; S, 21.90 (%), Found: C, 49.05; H, 5.31 ; N, 7.27; S, 22.03 (%).
mp. 126.0-129.0°C mp.126.0-129.0 ° C
実施例 1842 Example 1842
N-[8-(4 -シクロォクテン - 1 -ィルスルファ二ル)- 5, 6 -ジメチル -3-ォキソ -2, 3 -ジ ヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデン ]_4-メチルベンゼンスル ホンアミド . 実施例 18-41 で得た化合物(168 mg, 0.287 mmol)のァセトニトリル (1 ml) の溶液に室温で攪拌しながらフッ化テトラプチルアンモニゥム水和物 (150 mg, 0.574腿 ol) を加え、反応液を室温で 2時間、 50°C で 16時間さらに 60°C で 7.5 時間攪拌した。 反応液に酢酸ェチル (30 ml)を加え、 0.1N塩酸、 水および飽和食 塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して濃赤色油状物を得た。 こ れをシリカゲル(10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 2:8-3:7) に付し、 酢酸ェチルーへキサン(3 :7) の画分を濃縮乾固後、 メタノールで希釈し た。析出物を濾取し、メタノールで洗浄後、乾燥して表題化合物(68 mg, 0.139匪 ol, 48.4%)を赤色粉末として得た。 N- [8- (4-Cyclooctene-1-ylsulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine- 1-ylidene] _4-methylbenzenesulfonamide. To a solution of the compound obtained in Examples 18-41 (168 mg, 0.287 mmol) in acetonitrile (1 ml) was added tetrabutylammonium fluoride hydrate (150 mg, 0.574 t) while stirring at room temperature. The reaction solution was stirred at room temperature for 2 hours, at 50 ° C for 16 hours, and at 60 ° C for 7.5 hours. Ethyl acetate (30 ml) was added to the reaction solution, which was washed sequentially with 0.1 N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to obtain a dark red oil. This was subjected to silica gel (10 g) chromatography (ethyl acetate-hexane, 2: 8-3: 7), and the ethyl acetate-hexane (3: 7) fraction was concentrated to dryness and diluted with methanol. did. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (68 mg, 0.139 marl, 48.4%) as a red powder.
Ή-NMR (200MHz, CDC13): (51.5-1.8 (6H, m), 1.80 (3H, q, J= 1.2 Hz), 1.93 (2H, m), 2.16 (2H, brq, J= 6.6 Hz), 2.18 (1H, ), 2.26 (3H, q, J= 1.0 Hz), 2.40 (1H, m), 2.41 (3H, s), 3.46 (1H, m), 5.66 (1H, m), 5.71 (1H, m), 7.28 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.0 Hz), 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): (51.5-1.8 (6H, m), 1.80 (3H, q, J = 1.2 Hz), 1.93 (2H, m), 2.16 (2H, brq, J = 6.6 Hz) , 2.18 (1H,), 2.26 (3H, q, J = 1.0 Hz), 2.40 (1H, m), 2.41 (3H, s), 3.46 (1H, m), 5.66 (1H, m), 5.71 (1H , m), 7.28 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.0 Hz), 9.64 (1H, brs).
元素分析値: C23H27N303S3として計算値: C, 56.41; H, 5.56; N, 8.58; S, 19.64 ( ) 、 実測値: C, 56.39; H, 5.48; N, 8.62; S, 19.53 (%) . Elemental analysis: C 23 H 27 N 3 0 3 S 3 Calculated: C, 56.41; H, 5.56 ; N, 8.58; S, 19.64 (), Found: C, 56.39; H, 5.48 ; N, 8.62 ; S, 19.53 (%).
mp. 177.5-178.5°C mp. 177.5-178.5 ° C
実施例 18-43 Example 18-43
(2S, 4R) -2- {[(5, 6-ジメチル- 1- { [ (4 -メチルフェニル)スルホニル]イミノ } -3 -才 キソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 8-ィル)スルファニル]メ チル }- 4-フルォロ-卜ピロリジンカルポン酸 tert-ブチル (2S, 4R) -2-{[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3--year-old oxo-2,3-dihydro-1H-imidazo [5,1 -c] [1,4] thiazine-8-yl) sulfanyl] methyl} -tert-butyl 4-fluoro-topyrrolidinecarbonate
実施例 4-555 で得た化合物(250 mg, 0.430腿 ol)のジクロロメタン (3 ml) の溶液に 0°Cで攪拌しながら Deoxo- Fluor™ (114 mg, 0.517 mmol)のジクロロメ タン (1 ml)溶液を 60分間かけて滴下し、 反応液を室温で 2.5時間攪拌した。 反 応液にメタノール(1 ml)および飽和炭酸水素ナトリゥム aoml)を順次加え、 酢酸 ェチルージェチルエーテル(5:1, 60 ml)で抽出した。 有機層を 0.1N塩酸- 5%食塩 水、 5 食塩水および飽和食塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮し て濃赤色油状物を得た。 これをシリカゲル(10 g)のクロマトグラフィー (酢酸ェ チル-へキサン、 2:8-5:5) に付し、 酢酸ェチルーへキサン (4 :6) の画分を濃縮乾 固後、 ジェチルエーテルで希釈した。 析出物を濾取し、 ジェチルエーテルで洗浄 後、 乾燥して表題化合物(19 mg, 0.033 ramol, 7.6%)を赤色粉末として得た。 Ή-NMR (200MHz, CDC ): 1.48 (9H, s), 1.78 (3H, s), 2.26 (3H, s), 2.43 (3H, s), 3.29 (1H, dd, J= 13.3, 3.1 Hz), 3.41 (2H, m), 3.60-4.20 (2H, m), 4.35 (1H, m), 5.05 (1H, brd, J= 53 Hz), 7.30 (2H, d, J= 8.2 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). To a solution of the compound obtained in Example 4-555 (250 mg, 0.430 mol) in dichloromethane (3 ml) was added Deoxo-Fluor ™ (114 mg, 0.517 mmol) in dichloromethane (1 ml) while stirring at 0 ° C. ) The solution was added dropwise over 60 minutes, and the reaction solution was stirred at room temperature for 2.5 hours. Methanol (1 ml) and saturated sodium bicarbonate (aoml) were sequentially added to the reaction solution, and the mixture was extracted with ethyl acetate-ethyl ether (5: 1, 60 ml). The organic layer was washed successively with 0.1N hydrochloric acid-5% brine, 5 brine and saturated brine, dried over sodium sulfate, and concentrated to give a dark red oil. This was subjected to chromatography on silica gel (10 g) (ethyl acetate-hexane, 2: 8-5: 5), and the ethyl acetate-hexane (4: 6) fraction was concentrated to dryness. Diluted with ether. The precipitate is collected by filtration and washed with getyl ether. After drying, the title compound (19 mg, 0.033 ramol, 7.6%) was obtained as a red powder. Ή-NMR (200MHz, CDC): 1.48 (9H, s), 1.78 (3H, s), 2.26 (3H, s), 2.43 (3H, s), 3.29 (1H, dd, J = 13.3, 3.1 Hz) , 3.41 (2H, m), 3.60-4.20 (2H, m), 4.35 (1H, m), 5.05 (1H, brd, J = 53 Hz), 7.30 (2H, d, J = 8.2 Hz), 7.83 ( 2H, d, J = 8.4 Hz), 9.62 (1H, brs).
実施例 18-44 Example 18-44
N- (tert-ブチル)-5- [(5, 6-ジメチル- 1-{ [(4-メチルフエニル)スルホニル]イミ ノ}-3-ォキソ-2,3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 8-ィル)スルフ ァニル]ペンタンアミド N- (tert-butyl) -5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5, c] [1,4] thiazin-8-yl) sulfanyl] pentanamide
実施例 18-21 'で得られた 5- {[2, 3 -ジヒドロ- 5, 6-ジメチル -3-ォキソ -1-({[4-(トリフルォロメチル)フエニル]スルホ二ル}ィミノ) - 1H-イミダゾ [5,卜 c] [1,4]チアジン- 8-ィル]スルファニル }吉草酸 (100 mg, 0.208 mmol) と tert-ブチルァミン (0.0247 ml, 0.228 mmol) の N, N-ジメチルホルムアミド (4.00 ml) 溶液に、 シァノりん酸ジェチル (0.165 ml, 10.2讓 ol)、 続いてトリェチル ァミン (0.0369 ml, 0.228匪 ol) を加えた。 反応混合物を室温で 16時間かき混 ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶 媒を減圧下に留去して題記化合物 (23.6mg, 4.6%) を暗赤色結晶として得た。 酢 酸ェチル -ジェチルェ一テルから再結晶して融点 187-189°Cの暗赤色結晶を得た。 IR (KBr) リ: 3198, 2967, 2924, 1752, 1721, 1645, 1618, 1561 cm"1. Example 18-21 5-{[2,3-dihydro-5,6-dimethyl-3-oxo-1-({[4- (trifluoromethyl) phenyl] sulfonyl} imino obtained in ' )-1H-imidazo [5, tric] [1,4] thiazin-8-yl] sulfanyl} N, N- of valeric acid (100 mg, 0.208 mmol) and tert-butylamine (0.0247 ml, 0.228 mmol) To a dimethylformamide (4.00 ml) solution was added getyl cyanophosphate (0.165 ml, 10.2 benzyl), followed by triethylamine (0.0369 ml, 0.228 dandelion). After stirring the reaction mixture at room temperature for 16 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to give the title compound was evaporated Solvent under reduced pressure (23.6 mg, 4.6%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 187-189 ° C. IR (KBr): 3198, 2967, 2924, 1752, 1721, 1645, 1618, 1561 cm " 1 .
¾-NMR (CDC13) : 51.35 (9H, s), 1.54-1.82 (4H, m), 1.78 (3H, s), 2.08-2.18 (2H, m), 2.26 (3H, s), 2.42 (3H, s), 2.88-3.02 (2H, m), 5.27 (1H, bs), 7.29 (2H, d, J=8.0 Hz), 7.84 (2H, d, J=8.4 Hz), 9.61 (1H, bs). ¾-NMR (CDC1 3): 51.35 (9H, s), 1.54-1.82 (4H, m), 1.78 (3H, s), 2.08-2.18 (2H, m), 2.26 (3H, s), 2.42 (3H , s), 2.88-3.02 (2H, m), 5.27 (1H, bs), 7.29 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.61 (1H, bs) .
元素分析値: C24H32N404S3として Elemental analysis: as C 24 H 32 N 4 0 4 S 3
計算値 (¾) : C, 53.71; H, 6.01; , 10.44; S, 17.92. Calculated value (¾): C, 53.71; H, 6.01;, 10.44; S, 17.92.
実測値 ( ) : C, 53.50; H, 5.76; N, 10.36; S, 18.05. Obtained value (): C, 53.50; H, 5.76; N, 10.36; S, 18.05.
実施例 18 - 45 Example 18-45
N - {3_[(5,6-ジメチル -1 - {[(4-メチルフエニル)スルホニル]イミノ}- 3 -ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル)スルファニル]プロピ ル}- 2, 2 -ジメチルプロパンアミド 実施例 18-12で得た化合物(0.49 g, 0.79mmol)および塩化ピバロイル 0151 mg, 1.19 IMOI)の THF (10 ml)の懸濁液に室温で攪拌しながらトリェチルァミン(0.33 ml, 2.37 IMOI)を加え、 反応液を 24時間攪拌した。 反応液を酢酸ェチル(50 ml) で希釈し、 2%炭酸水素ナトリウム水溶液、 0.1N塩酸、 水および飽和食塩水で順 次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮して濃赤色油状物を得た。 これをシリ 力ゲル(10 g)のクロマトグラフィー(酢酸ェチルーへキサン、 2:8-6: 4)で精製し、 得られた赤色油状物をジェチルエーテルから結晶化し、表題化合物(269 nig, 0.515 miol, 65.1%)を赤色粉末として得た。 N-{3 _ [(5,6-dimethyl-1--[[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-8-yl) sulfanyl] propyl} -2,2-dimethylpropanamide To a suspension of the compound obtained in Example 18-12 (0.49 g, 0.79 mmol) and pivaloyl chloride (0151 mg, 1.19 IMOI) in THF (10 ml) was added triethylamine (0.33 ml, 2.37 IMOI) with stirring at room temperature. The reaction was stirred for 24 hours. The reaction mixture was diluted with ethyl acetate (50 ml), washed successively with 2% aqueous sodium hydrogen carbonate, 0.1N hydrochloric acid, water and brine, dried over sodium sulfate, and concentrated to give a dark red oil. Was. This was purified by chromatography on silica gel (10 g) (ethyl acetate-hexane, 2: 8-6: 4), and the obtained red oil was crystallized from getyl ether to give the title compound (269 nig, 0.515 miol, 65.1%) as a red powder.
Ή-NMR (200MHz, CDC13): δ 1.20 (9Η, s), 1.78 (3H, s), 1.90 (2H, quint, J= 7.1 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J= 7.5 Hz), 3.34 (2H, q, J= 6.2 Hz), 5.84 (1H, br), 7.29 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). Ή-NMR (200MHz, CDC1 3 ): δ 1.20 (9Η, s), 1.78 (3H, s), 1.90 (2H, quint, J = 7.1 Hz), 2.26 (3H, s), 2.42 (3H, s) , 2.95 (2H, t, J = 7.5 Hz), 3.34 (2H, q, J = 6.2 Hz), 5.84 (1H, br), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C23H30N404S3.0.3H20として計算値: C, 52.49; H, 5.86; N, 10.65; S, 18.28 (%) 、 実測値: C, 52.37; H, 5.84; N, 10.67; S, 18.38 (%) · Elemental analysis: C 23 H 30 N 4 0 4 S 3 .0.3H 2 0 Calculated: C, 52.49; H, 5.86 ; N, 10.65; S, 18.28 (%), Found: C, 52.37; H , 5.84; N, 10.67; S, 18.38 (%)
mp. 172.0-174. Ot mp. 172.0-174. Ot
実施例 18-46 Example 18-46
N- [8-({[ (2S, 4S) - 4 -フルォ口ピロ' Jジニル]メチル }スルファニル) -5, 6-ジメチル - 3 -ォキソ - 2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1, ]チアジン-卜ィリデン] +メ チルベンゼンスルホンアミド 塩酸塩 N- [8-({[(2S, 4S) -4-fluoro-pyro'J-dinyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro- 1H-imidazo [5 , 1-c] [1,] thiazine-triylidene] + methylbenzenesulfonamide hydrochloride
実施例 4-546 で得た化合物の粉末(純度 80'%, 526 mg, 0.722 讓 ol) を TFA The powder of the compound obtained in Example 4-546 (purity 80 '%, 526 mg, 0.722 alcohol) was added to TFA
(1.0ml)に溶解し、 室温で 3 時間放置した。 反応液をトルエン(50 ml)で希釈し て濃縮乾固し、 残渣にトルエン(50ml)を加えて濃縮した。得られた残渣をメタノ —ルーァセトニトリル(1:9, 50 ml)に溶解し、 1N塩酸 (1.08 ml)を加えて濃縮し、 残渣にメタノールーァセトニトリル α:ι)を加えて濃縮し、エタノールで希釈した。 析出物を濾取し、 ァセトニトリルおよび酢酸ェチルで洗浄後、 乾燥して表題化合 物(148 mg, 0.285 藤 ol, 39.5%)を濃紫色粉末として得た。 (1.0 ml) and left at room temperature for 3 hours. The reaction solution was diluted with toluene (50 ml) and concentrated to dryness. Toluene (50 ml) was added to the residue and concentrated. The obtained residue was dissolved in methano-ruacetonitrile (1: 9, 50 ml), concentrated by adding 1N hydrochloric acid (1.08 ml), and concentrated by adding methanol-acetonitrile α: ι) to the residue. And diluted with ethanol. The precipitate was collected by filtration, washed with acetonitrile and ethyl acetate, and dried to give the title compound (148 mg, 0.285 w / w, 39.5%) as a dark purple powder.
Ή-NMR (200MHz, DMS0-d6): <51.81 (3H, s), 2.06 (2H, m), 2.17 (3H, s)', 2.39 (3H, s), 3.30-3.60 (4H, m), 3.88 (1H, m), 5.42 (1H, brd, J= 51.6 Hz), 7.40 (2H, d, J= 8.2 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.54 (1H, br), 9.89 (1H, br), 11.79 (1H, br). Ή-NMR (200MHz, DMS0- d 6): <51.81 (3H, s), 2.06 (2H, m), 2.17 (3H, s) ', 2.39 (3H, s), 3.30-3.60 (4H, m) , 3.88 (1H, m), 5.42 (1H, brd, J = 51.6 Hz), 7.40 (2H, d, J = 8.2 Hz), 7.87 (2H, d, J = 8.4 Hz), 9.54 (1H, br) , 9.89 (1H, br), 11.79 (1H, br).
元素分析値: C2。H23N403S3F.HC1.0.5¾0として計算値: C, 45.49; H, 4.77; N, 10.61; S, 18.22; CI, 6.71 (%) 、 実測値: C, 45.40; H, 4.87; N, 10.42; S, 18.48; CI, 6.53 (%) . Elemental analysis: C 2. H 23 N 4 3 S 3 F. Calculated as HC 1.0.5¾0: C, 45.49; H, 4.77; N, 10.61; S, 18.22; CI, 6.71 (%), found: C, 45.40; H, 4.87; N, 10.42; S, 18.48; CI, 6.53 (%).
実施例 18-47 ' Example 18-47 '
N-[8-({[(5R, 7S)- 3- (ヒドロキシメチル)トリシク口 [3.3.1.1—3, 7—]デシ- 1-ィル] メチル }スルファニル) -5, 6-ジメチル -3-ォキソ _2, 3-ジヒドロ- m-イミダゾ [5,1-c] [1, 4]チアジン-卜ィリデン ]-4-メチルベンゼンスルホンアミド N- [8-({[(5R, 7S) -3- (Hydroxymethyl) tris-capped [3.3.1.1-3,7-] dec-1-yl] methyl} sulfanyl) -5,6-dimethyl- 3-oxo_2,3-dihydro-m-imidazo [5,1-c] [1,4] thiazine-triylidene] -4-methylbenzenesulfonamide
実施例 4-549で得た化合物 (495 mg, 0.822腿 ol)の THF -メタノール (1:1, 6 ml) 懸濁液に IN水酸化ナトリウム水溶液(3.0 ml, 3.0mmol) を加え、 65°C で 30 分間攪拌した。反応液を室温に戻し、 1N塩酸 (3.0ml)を加えて 30分間攪拌した。 析出物を濾取し、 水およびメタノール一水 (1:1)で洗浄後、 乾燥して表題化合物 (432 mg, 0.772匪 ol, 93.9%)を紫色粉末として得た。 To a suspension of the compound obtained in Example 4-549 (495 mg, 0.822 mol) in THF-methanol (1: 1, 6 ml) was added an aqueous IN sodium hydroxide solution (3.0 ml, 3.0 mmol), and the mixture was heated at 65 ° The mixture was stirred at C for 30 minutes. The reaction solution was returned to room temperature, 1N hydrochloric acid (3.0 ml) was added, and the mixture was stirred for 30 minutes. The precipitate was collected by filtration, washed with water and methanol-water (1: 1), and dried to give the title compound (432 mg, 0.772 bandol, 93.9%) as a purple powder.
-NMR (200MHz, CDC13): 51.34 (1H, s), 1.45 (2H, m), 1.50-1.58 (8H, m), 1.61 (2H, m), 1.78 (3H, q, J= 1.2 Hz), 2.12 (2H, m), 2.26 (3H, q, J= 0.8 Hz), 2.41 (3H, s), 2.81 (2H, s), 3.25 (2H, d, J= 5.6 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.88 (2H, d, J= 8.4 Hz), 9.57 (1H, brs). -NMR (200MHz, CDC1 3): 51.34 (1H, s), 1.45 (2H, m), 1.50-1.58 (8H, m), 1.61 (2H, m), 1.78 (3H, q, J = 1.2 Hz) , 2.12 (2H, m), 2.26 (3H, q, J = 0.8 Hz), 2.41 (3H, s), 2.81 (2H, s), 3.25 (2H, d, J = 5.6 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.88 (2H, d, J = 8.4 Hz), 9.57 (1H, brs).
元素分析値: C27H33N304S3として計算値: C, 57.93; H, 5.94; N, 7.51 (%) 、 実 測値: C, 57.94; H, 6.11; , 7.28 (%) . Elemental analysis: Calculated as C 27 H 33 N 3 0 4 S 3: C, 57.93; H, 5.94; N, 7.51 (%), actual Hakachi: C, 57.94; H, 6.11 ;, 7.28 (%) .
即. 197.5-198.0°C Immediately. 197.5-198.0 ° C
実施例 18 - 48 チル- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1 -ィリデ ン]- 4-メチルベンゼンスルホンアミド Example 18-48 tyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene] -4-methylbenzenesulfonamide
実施例 4-550で得た化合物(1.67 g, 3.04腿 ol)の THF -メタノール (1:1, 20 ml) 懸濁液に IN水酸化ナトリウム水溶液(9.12 ml, 9.12删 ol) を加え、 室温で 2.5時間攪拌した。 反応液に戻し、 IN塩酸(10 ml)を加えて 30分間攪拌した。 析 出物を濾取し、 水およびメタノール一水 (1:1)で洗浄後、 乾燥して表題化合物 (1.17 g, 2.30画 ol, 75.8 )を赤紫色粉末として得た。 ¾-N R (200MHz, CDC13): δ 0.89/0.69 (2H, qm/q, J= 13/12.2 Hz), 1.32 (2H, m), 1.40-1.65 (4H, m), 1.79 (3H, s), 1.90 (2H, brq, J= 11.8 Hz), 2.26 (3H, q, J= 0.8 Hz), 2.42 (3H, s), 2.84/2.92 (2H, d, J= 6.6/7.0 Hz), 3.49 (2H, m), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). To a suspension of the compound obtained in Example 4-550 (1.67 g, 3.04 mol) in THF-methanol (1: 1, 20 ml) was added IN aqueous sodium hydroxide solution (9.12 ml, 9.12 mol), and the mixture was stirred at room temperature. For 2.5 hours. The mixture was returned to the reaction solution, IN hydrochloric acid (10 ml) was added, and the mixture was stirred for 30 minutes. The precipitate was collected by filtration, washed with water and methanol / water (1: 1), and dried to give the title compound (1.17 g, 2.30 fraction ol, 75.8) as a red-purple powder. ¾-NR (200MHz, CDC1 3 ): δ 0.89 / 0.69 (2H, qm / q, J = 13 / 12.2 Hz), 1.32 (2H, m), 1.40-1.65 (4H, m), 1.79 (3H, s ), 1.90 (2H, brq, J = 11.8 Hz), 2.26 (3H, q, J = 0.8 Hz), 2.42 (3H, s), 2.84 / 2.92 (2H, d, J = 6.6 / 7.0 Hz), 3.49 (2H, m), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
元素分析値: C23H29N304S3として計算値: C, 54. 1; H, 5.76; N, 8.28; S, 18.95 (%) 、 実測値: C, 54.26; H, 5.50; N, 8.25; S, 18.98 (%) . Elemental analysis: Calculated as C 23 H 29 N 3 0 4 S 3: C, 54. 1; H, 5.76; N, 8.28; S, 18.95 (%), Found: C, 54.26; H, 5.50 ; N, 8.25; S, 18.98 (%).
mp. 156.5-157.0。C mp. 156.5-157.0. C
実施例 18-49 Example 18-49
N-{8_[(5 -ヒドロキシ- 3, 3-ジメチルペンチル) スルファニル ]-5, 6-ジメチル -3 - ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン }- 4-メチル ベンゼンスルホンアミド N- {8 _ [(5-hydroxy-3,3-dimethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene}-4-methylbenzenesulfonamide
実施例 4-552で得た化合物(1.64 3.05mmol)の THF-メタノール (1:1, 24 ml) 懸濁液に IN NaOH (12.2 ml, 12.2讓01) を加え、 室温で 90分間攪拌した。 反応 液に 1N塩酸(12.2 ml)を加えて 30分間攪拌した。析出物を濾取し、 水およびメタ ノール一水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物(1.48 g, 2.98 匪 ol, 97.8%)を赤紫色粉末として得た。 To a suspension of the compound (1.64 3.05 mmol) obtained in Example 4-552 in THF-methanol (1: 1, 24 ml) was added IN NaOH (12.2 ml, 12.2 SUB01), and the mixture was stirred at room temperature for 90 minutes. 1N Hydrochloric acid (12.2 ml) was added to the reaction solution, and the mixture was stirred for 30 minutes. The precipitate was collected by filtration, washed with water, methanol-one water (1: 1) and methanol, and dried to give the title compound (1.48 g, 2.98 ol, 97.8%) as a red-purple powder.
'H-NMR (300MHz, CDC13): 50.96 (6H, s), 1.22 (1H, br), 1.55 (2H, t, J- 7.5 Hz), 1.60 (2H, m), 1.79 (3H, q, J= 0.9 Hz), 2.26 (3H, q, J= 0.9 Hz), 2.42 (3H, s) , 2.92 (2H, m), 3.71 (2H, brt, J= 7.5 Hz), 7.28 (2H, d, J= 7.8 Hz), 7.83 (2H, d, J= 7.8 Hz), 9.61 (1H, brs). 'H-NMR (300MHz, CDC1 3): 50.96 (6H, s), 1.22 (1H, br), 1.55 (2H, t, J- 7.5 Hz), 1.60 (2H, m), 1.79 (3H, q, J = 0.9 Hz), 2.26 (3H, q, J = 0.9 Hz), 2.42 (3H, s), 2.92 (2H, m), 3.71 (2H, brt, J = 7.5 Hz), 7.28 (2H, d, J = 7.8 Hz), 7.83 (2H, d, J = 7.8 Hz), 9.61 (1H, brs).
元素分析値: C22H29N304S3として計算値: C, 53.31; H, 5.90; , 8.48; S, 19.41Elemental analysis: Calculated as C 22 H 29 N 3 0 4 S 3: C, 53.31; H, 5.90;, 8.48; S, 19.41
(%) 、 実測値: C, 53.11; H, 5.79; N, 8.53; S, 19.58 (%) . (%), Found: C, 53.11; H, 5.79; N, 8.53; S, 19.58 (%).
mp. 193.0-195.5°C mp. 193.0-195.5 ° C
実施例 18-50 Example 18-50
N- [8- ( { [l-(2, 2-ジメチルプロパノィル) -4-ピペリジニル]メチル }スルファニ ル)- 5, 6 -ジメチル- 3-ォキソ -2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン 実施例 4-116 で得た化合物(266 mg, 0.516 mmol) およびトリェチルァミン (0.215 ml, 1.55 mmol)の THF (5 ml)懸濁液に塩ィ匕ピバロィル(0.080 ml, 0.620 mmol)を室温で加え、反応液を室温で 12時間攪拌した。反応液に 1N塩酸 (1.6 ml) を加え、 水 aOml)および酢酸ェチル(15ml)で希釈し、 室温で攪拌した。 析出物 を濾取し、 メタノール—水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合 物(251 mg, 0.446 mmol, 86.4%)を橙色粉末として得た。N- [8-({[l- (2,2-Dimethylpropanoyl) -4-piperidinyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine A suspension of the compound obtained in Example 4-116 (266 mg, 0.516 mmol) and triethylamine (0.215 ml, 1.55 mmol) in THF (5 ml) was salted. Dani Pivaloyl (0.080 ml, 0.620 mmol) at room temperature and the reaction was stirred at room temperature for 12 hours. The reaction mixture was diluted with 1N hydrochloric acid (1.6 ml), diluted with water (aOml) and ethyl acetate (15 ml), and stirred at room temperature. The precipitate was collected by filtration, washed with methanol-water (1: 1) and methanol, and dried to give the title compound (251 mg, 0.446 mmol, 86.4%) as an orange powder.
- NMR (200MHz, CDC13): 51.16 (2H, brq, J= 11.2 Hz), 1.28 (9H, s), 1.79 (3H, q, J= 1.0 Hz), 1.85 (1H, m), 1.87 (2H, brd, J- 11.0 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.43 (3H, s), 2.75 (2H, brt, J= 11.0 Hz), 2.85 (2H, d, J= 6.6 Hz), 4.44 (2H, brd, J= 13.2 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.84 (2H, d, J= 8.4 Hz), 9.63 (1H, brs). - NMR (200MHz, CDC1 3) : 51.16 (2H, brq, J = 11.2 Hz), 1.28 (9H, s), 1.79 (3H, q, J = 1.0 Hz), 1.85 (1H, m), 1.87 (2H , brd, J- 11.0 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.43 (3H, s), 2.75 (2H, brt, J = 11.0 Hz), 2.85 (2H, d, J = 6.6 Hz) ), 4.44 (2H, brd, J = 13.2 Hz), 7.29 (2H, d, J = 8.4 Hz), 7.84 (2H, d, J = 8.4 Hz), 9.63 (1H, brs).
元素分析値: C26H34N404S3として計算値: C, 55.49; H, 6.09; N, 9.96; S, 17.09 ( ) 、 実測値: C, 55.47; H, 5.90; N, 9.95; S, 16.98 (%) . Elemental analysis: C 26 H 34 N 4 0 4 S 3 Calculated: C, 55.49; H, 6.09 ; N, 9.96; S, 17.09 (), Found: C, 55.47; H, 5.90 ; N, 9.95 ; S, 16.98 (%).
mp. 221.0-223.0°C mp.221.0-223.0 ° C
実施例 18-51 Example 18-51
N- (8- { [(卜ァセチル -4-ピベリジニル) メチル] スルファニル } -5, 6-ジメチル -3- ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン )-4-メチル ベンゼンスルホンアミド N- (8-{[(Tracetyl-4-piberidinyl) methyl] sulfanyl} -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-triylidene) -4-methylbenzenesulfonamide
実施例 4-116 で得た化合物(303 mg, 0.588 醒 ol) およびトリェチルァミン Compound obtained in Example 4-116 (303 mg, 0.588 awake ol) and triethylamine
(0.244 ml, 1.76 mmol)の THF (6 ml)懸濁液に無水酢酸(0.113 ml, 1.18 mmol) を室温で加え、 反応液を室温で 12時間攪拌した。 反応液に酢酸ェチル(15 ml)、 IN塩酸 (1.8 ml) および水 (10 ml) を加え、 室温で攪拌した。 得られた懸濁液 をメタノールおよび THFを加えて溶解し、 約 15 mlまで濃縮した。'析出物を濾取 し、 メタノール一水 (1:1)およびメタノールで洗浄後、 乾燥して表題化合物(283 mg, 0.543 mmol, 92.4%)を橙色粉末として得た。 Acetic anhydride (0.113 ml, 1.18 mmol) was added to a suspension of (0.244 ml, 1.76 mmol) in THF (6 ml) at room temperature, and the reaction solution was stirred at room temperature for 12 hours. Ethyl acetate (15 ml), IN hydrochloric acid (1.8 ml) and water (10 ml) were added to the reaction solution, and the mixture was stirred at room temperature. The resulting suspension was dissolved by adding methanol and THF, and concentrated to about 15 ml. The precipitate was collected by filtration, washed with methanol / water (1: 1) and methanol, and dried to give the title compound (283 mg, 0.543 mmol, 92.4%) as an orange powder.
Ή-NMR (200MHz, CDC13, 異性体混合物): 51.16 (2H, m), 1.79 (3H, q, J= 1.0 Hz), 1.79 (1H, m), 1.92 (2H, brd, J= 15.0 Hz), 2.10 (3H, s), 2.26 (3H, q, J= 1.2 Hz), 2.43 (3H, s), 2.51 (2/2H, brt, J= 12.8 Hz), 2.82 (2/2H, d, J= 6.2 Hz), 2.87 (2/2H, d, J= 6.6 Hz), 3.02 (2/2H, dt, J= 2.6, 12.4 Hz), 3.83 (2/2H, brd, J= 13.6 Hz), 4.64 (2/2H, brd, J= 13.0 Hz), 7.29 (2H, d, J= 8.2 Hz), 7.84 (2H, d, J= 8.2 Hz), 9.64 (1H, brs). 元素分析値: C23H28N404S3として計算値: C, 53.05; H, 5.42; N, 10.76; S, 18.47 Ή-NMR (200MHz, CDC1 3 , mixture of isomers): 51.16 (2H, m) , 1.79 (3H, q, J = 1.0 Hz), 1.79 (1H, m), 1.92 (2H, brd, J = 15.0 Hz ), 2.10 (3H, s), 2.26 (3H, q, J = 1.2 Hz), 2.43 (3H, s), 2.51 (2 / 2H, brt, J = 12.8 Hz), 2.82 (2 / 2H, d, J = 6.2 Hz), 2.87 (2 / 2H, d, J = 6.6 Hz), 3.02 (2 / 2H, dt, J = 2.6, 12.4 Hz), 3.83 (2 / 2H, brd, J = 13.6 Hz), 4.64 (2 / 2H, brd, J = 13.0 Hz), 7.29 (2H, d, J = 8.2 Hz), 7.84 (2H, d, J = 8.2 Hz), 9.64 (1H, brs). Elemental analysis: C 23 H 28 N 4 0 4 S 3 Calculated: C, 53.05; H, 5.42 ; N, 10.76; S, 18.47
( ) 、 実測値: C, 52.82; H, 5.34; N, 10.75; S, 18.35. (), Found: C, 52.82; H, 5.34; N, 10.75; S, 18.35.
mp. 216.0-217. O : mp. 216.0-217.O:
実施例 18-52 Example 18-52
N- [8- ( {'[1, 2-trans-2- (ヒドロキシメチル)シクロへキシル]メチル }スルファニ ル)- 5, 6-ジメチル- 3-ォキソ - 2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1,4]チアジン 実施例 4-556で得た化合物(1.90 g, 3.46腿 oDの THF-メタノール (1:1, 26 ml) 懸濁液に 1N水酸化ナトリゥム水溶液(13.8 ml, 13.8丽 ol) を加え、 60°C で 30分間攪拌した。反応液を室温に戻し、 1N塩酸(13.8 ml)を加えて 30分間攪拌し た。 析出物を濾取し、 水およびメタノール—水 (1:1)で洗浄後、 乾燥して表題化 合物(1.55 g, 3.05 mmol, 88.1%)を赤紫色粉末として得た。 . N- [8-({'[1,2-trans-2- (hydroxymethyl) cyclohexyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydrido-1H- Imidazo [5,1-c] [1,4] thiazine 1N sodium hydroxide was added to a suspension of the compound obtained in Example 4-556 (1.90 g, 3.46 tD in THF-methanol (1: 1, 26 ml)). An aqueous solution (13.8 ml, 13.8 mol) was added, and the mixture was stirred for 30 minutes at 60 ° C. The reaction solution was returned to room temperature, 1N hydrochloric acid (13.8 ml) was added, and the mixture was stirred for 30 minutes. After washing with water and methanol-water (1: 1), drying was performed to obtain the title compound (1.55 g, 3.05 mmol, 88.1%) as a red-purple powder.
Ή一 NMR (200MHz, CDC13): δ\.18 (2Η, in), 1.25 (2H, m), 1.36 (2H, m), 1.60 (2H, m), 1.74 (2H, brs), 1.78 (3H, q, J= 0.8 Hz), 2.02 (1H, brd, J= 12.0 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.42 (3H, s), 2.84 (1H, dd, J= 12.8, 7.6 Hz), 3.28 (1H, dd, J= 12.6, 4.2 Hz) , 3.64 (2H, brd, J= 3.5 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.60 (1H, brs). Ή one NMR (200MHz, CDC1 3): δ \ .18 (2Η, in), 1.25 (2H, m), 1.36 (2H, m), 1.60 (2H, m), 1.74 (2H, brs), 1.78 ( 3H, q, J = 0.8 Hz), 2.02 (1H, brd, J = 12.0 Hz), 2.26 (3H, q, J = 1.2 Hz), 2.42 (3H, s), 2.84 (1H, dd, J = 12.8 , 7.6 Hz), 3.28 (1H, dd, J = 12.6, 4.2 Hz), 3.64 (2H, brd, J = 3.5 Hz), 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.60 (1H, brs).
元素分析値: C23 N304S3として計算値: C, 54.41; H, 5.76; N, 8.28; S, 18.95' (%) 、 実測値: C, 54.26; H, 5.56; N, 8.03; S, 19.23 (%) · Elemental analysis: C 23 N 3 0 4 S 3 Calculated: C, 54.41; H, 5.76 ; N, 8.28; S, 18.95 '(%), Found: C, 54.26; H, 5.56 ; N, 8.03 ; S, 19.23 (%)
mp. 183.0-183.5°C mp. 183.0-183.5 ° C
実施例 18-53 Example 18-53
メチル (5, 6-ジメチル- 1-{ [(4 -メチルフエニル)スルホニル]ィミノ卜 3—ォキソ - 8 - [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニル] - 1H-ィミダゾ [5, 1-c] [1, 4]チアジン - 2 (3H)-ィル)ァセテ一ト ' Methyl (5, 6-dimethyl - 1- {[(4 - methylphenyl) sulfonyl] Imino Bok 3 - Okiso - 8 - [(5, 5, 6, 6, 6-pen evening cyclohexyl Furuo port) sulfanyl] - IH -Imidazo [5, 1-c] [1, 4] thiazine-2 (3H) -yl) acetate ''
実施例 4 - 35で得られた N - {5, 6 -ジメチル- 3-ォキソ - 8- [(5, 5, 6, 6, 6 -ペン夕フル ォ口へキシル)スルファニル] -2, 3 -ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン -卜イリデン卜 4-メチルベンゼンスルホンアミド (816 mg, 1.47 mmol) と炭酸力 リウム (305 mg, 2.21腿01) の N, N-ジメチルホルムアミド (16.0 ml) の懸濁液 に、 ブロモ酢酸メチル (0.214 ml, 2.21 扁 oi) を加えた。 反応混合物を室温で 15時間かき混ぜた後、 IN塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグ ラフィ一に付し、 n-へキザン -酢酸ェチル(10:1, v/v)で溶出して題記化合物(167 mg, 18%) を暗赤色結晶として得た。 N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] obtained in Example 4-35 -2,3 -Dihide mouth- 1H-imidazo [5, 1-c] [1, 4] thiazine-triidentyl 4-methylbenzenesulfonamide (816 mg, 1.47 mmol) and potassium carbonate (305 mg, 2.21 thigh 01) Methyl bromoacetate (0.214 ml, 2.21 oi) was added to a suspension of N, N-dimethylformamide (16.0 ml). Reaction mixture at room temperature After stirring for 15 hours, IN hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, after Drying (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexan-ethyl acetate (10: 1, v / v) to give the title compound (167 mg, 18%) as dark red crystals.
IR (KBr) V: 3013, 2955, 2863, 1746, 1665, 1617, 1557 cm— IR (KBr) V: 3013, 2955, 2863, 1746, 1665, 1617, 1557 cm—
'H-NMR (CDC13) : 51.54-1.64 (4H, m), 1.80 (3H, d, J=0.6Hz), 1.84-2.14 (2H, m), 2.29 (3H, d, J=1.2 Hz), 2.39 (3H, s), 2.84 (2H, t, J-6.6 Hz), 3.82 (3H, s), 5.08 (2H, s), 7.24 (2H, d, J=8.0 Hz), 7.88 (2H, d, J=8.4 Hz). . 'H-NMR (CDC1 3) : 51.54-1.64 (4H, m), 1.80 (3H, d, J = 0.6Hz), 1.84-2.14 (2H, m), 2.29 (3H, d, J = 1.2 Hz) , 2.39 (3H, s), 2.84 (2H, t, J-6.6 Hz), 3.82 (3H, s), 5.08 (2H, s), 7.24 (2H, d, J = 8.0 Hz), 7.88 (2H, d, J = 8.4 Hz).
元素分析値: C24H26N305S3F5-0.3H20として Elemental analysis: C 24 H 26 N 3 0 5 S 3 F 5 -0.3 H 2 0
計算値 (¾) : C, 45.53; H, 4.24; N, 6.64; S, 15.20. Calculated (¾): C, 45.53; H, 4.24; N, 6.64; S, 15.20.
実測値 (%) : C, 45.53; H, 4.35; Ν,, 6.62; S, 15.13. Found (%): C, 45.53; H, 4.35; Ν ,, 6.62; S, 15.13.
実施例 18-54 Example 18-54
ェチル 5-[(1-{[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 5, 6-トリ メチル -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル] ペンタノエー卜 Ethyl 5-[(1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,5,6-trimethyl-2,3-dihydro-1H-imidazo [5,1-c] [1 , 4] Thiazine-8-yl) sulfanyl] pentanoate
実施例 4-522で得られたェチル 5- [ (5, 6-ジメチル- 1- { [ (4-メチルフエニル)ス ルホニル]イミノ}-3 -ォキソ _2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン -8-ィル)スルファニル]ペンタノェ一ト(l.OOg, 1.96醒 ol) と炭酸カリウム(407 mg, 2.94匪 ol) の Ν,Ν-ジメチルホルムアミド (20.0 ml) の懸濁液に、 ョ一ドメ タン (336 mg, 2.35腿01) を加えた。 反応混合物を室温で 15時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒 を減圧下に留去して題記化合物 (940 mg, 92%) を暗赤色結晶として得た。 酢酸ェ チル-ジェチルェ一テルから再結晶して融点 128- 130°Cの暗赤色結晶を得た。 IR (KBr) リ: 2926, 1738, 1661, 1615, 1557 cm"1. Ethyl 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo obtained in Example 4-522 1-c] [1,4] thiazine-8-yl) sulfanyl] pentanoate (l.OOg, 1.96 ol) and potassium carbonate (407 mg, 2.94 ol)), Ν-dimethylformamide (20.0 ml) of suspension was added to potassium methoxide (336 mg, 2.35 thigh 01). After stirring the reaction mixture at room temperature for 15 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (940 mg, 92%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 128-130 ° C. IR (KBr): 2926, 1738, 1661, 1615, 1557 cm " 1 .
Ή-NMR (CDC13) : (51.27 (3H, t, J=7.2 Hz), 1.44-1.66 (4H, m), 1.79 (3H, d, J=l.2 Hz), 2.26 (2H, t, J=6.8 Hz), 2.29 (3H, d, J=0.8Hz), 2.41 (3H, s), 2.81 (2H, t, J=6.6 Hz), 3.70 (3H, s), 4.14 (2H, q, J=7.2 Hz), 7.27 (2H, d, J=8.2 Hz), 7.91 (2H, d, J=8.0 Hz). Ή-NMR (CDC1 3): (51.27 (3H, t, J = 7.2 Hz), 1.44-1.66 (4H, m), 1.79 (3H, d, J = l.2 Hz), 2.26 (2H, t, J = 6.8 Hz), 2.29 (3H, d, J = 0.8 Hz), 2.41 (3H, s), 2.81 (2H, t, J = 6.6 Hz), 3.70 (3H, s), 4.14 (2H, q, J = 7.2 Hz), 7.27 (2H, d, J = 8.2 Hz), 7.91 (2H, d, J = 8.0 Hz).
元素分析値: C23H29N305S3として 計算値 (¾)': C, 52.75; H, 5.58; N, 8.02; S, 18.37 Elementary analysis: as C 23 H 29 N 3 0 5 S 3 Calculated value (¾) ': C, 52.75; H, 5.58; N, 8.02; S, 18.37
実測値 (%) : C, 52.85; H, 5.67; , 8.01; S, 18.30. Found (%): C, 52.85; H, 5.67;, 8.01; S, 18.30.
実施例 18-55 Example 18-55
tert-ブチル 5-[(l- {[(4-メチルフエニル)スルホニル]イミノ卜 3-ォキソ -2, 5, 6- トリメチル _2, 3-ジヒドロ- 1H-イミダゾ [5, 1- c] [1 , 4]チアジン- 8-ィル)スルファ ニル]ペン夕ノエ一卜 tert-Butyl 5-[(l-{[(4-methylphenyl) sulfonyl] imino 3-oxo-2,5,6-trimethyl_2,3-dihydro-1H-imidazo [5,1-c] [1, 4] Thiazine-8-yl) sulfanyl] pen
実施例 18-72で得られた tert-ブチル 5 - [(5, 6-ジメチル _1-{ [(4-メチルフエ二 ル)スルホニル]ィミノ }- 3-ォキソ - 2, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1, 4]チア ジン- 8-ィル)スルファニル]ペンタノェ一ト (424 mg, 0.789匪 ol) と炭酸力リウ ム (164 mg, 1.18 mmol) の N, N-ジメチルホルムアミド (9.00 ml) の懸濁液に、 ョ一ドメタン (135 mg, 0.947 mmol) を加えた。 反応混合物を室温で 22時間かき 混ぜた後、 水を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。残留物をシリカゲルカラムクロマトグラフィーに付し、 n -へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 (285 mg, 66¾) を暗 赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶して融点 147-149°Cの暗赤色結晶を得た。 Tert-Butyl 5-[(5,6-dimethyl_1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H- obtained in Examples 18-72 N, N of imidazo [5, 1-c] [1,4] thiazin-8-yl) sulfanyl] pentanoate (424 mg, 0.789 marl ol) and carbonated lithium (164 mg, 1.18 mmol) To a suspension of -dimethylformamide (9.00 ml) was added methane (135 mg, 0.947 mmol). After stirring the reaction mixture at room temperature for 22 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (285 mg, 66¾) as dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals, mp 147-149 ° C.
IR (KBr) V: 2978, 2928, 1736, 1663, 1617, 1561 cm"1. IR (KBr) V: 2978, 2928, 1736, 1663, 1617, 1561 cm " 1 .
'H- R (CDC13) : (51.45 (9H, s), 1.51-1.58 (4H, in), 1.79 (3H, d, J=0.9 Hz), 2.18 (2H, t, J=7.2 Hz), 2.29 (3H, d, J-1.2 Hz), 2.42 (3H, s), 2.81 (2H, t, J=6.6 Hz), 3.70 (3H, s), 7.26 (2H, d, J=8.1 Hz), 7.91 (2H, d, J=8.1 Hz). 元素分析値: C25H33N305S3として 'H- R (CDC1 3): (51.45 (9H, s), 1.51-1.58 (4H, in), 1.79 (3H, d, J = 0.9 Hz), 2.18 (2H, t, J = 7.2 Hz), 2.29 (3H, d, J-1.2 Hz), 2.42 (3H, s), 2.81 (2H, t, J = 6.6 Hz), 3.70 (3H, s), 7.26 (2H, d, J = 8.1 Hz), 7.91 (2H, d, J = 8.1 Hz). Elemental analysis: C 25 H 33 N 3 0 5 S 3
計算値 (¾) : C, 54.42; H, 6.03; , 7.62; S, 17.43. Calculated value (¾): C, 54.42; H, 6.03;, 7.62; S, 17.43.
実測値 (¾) : C, 54.33; H, 6.02; N, 7.64; S, 17.48. Found (¾): C, 54.33; H, 6.02; N, 7.64; S, 17.48.
実施例 18 - 56 Example 18-56
tert-ブチル (5, 6-ジメチル- 1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォ キソ- 8 - [(5, 5,6, 6, 6-ペン夕フルォ口へキシル)スルファニル ]-1Η -ィミダゾ [5, 1 - c] [1, 4]チアジン- 2 (3H) -ィル)アセテート tert-butyl (5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentahexylhexyl) Sulfanyl] -1Η-imidazo [5,1-c] [1,4] thiazine-2 (3H) -yl) acetate
実施例 4-35で得られた N- {5, 6-ジメチル- 3 -ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フル ォ口へキシル)スルファニル] -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン -卜イリデン }- 4-メチルベンゼンスルホンアミド (1.00 g, 1.80 mmol) と炭酸力 リウム (373 mg, 2.70腿01) の N, N-ジメチルホルムアミド (20.0 ml) の懸濁液 に、 ブロモ酢酸 tert-ブチルエステル (0.377 ml, 2.70 mmol) を加えた。 反応混 合物を室温で 18時間かき混ぜた後、 1N塩酸を加えて酢酸ェチルで抽出した。 抽 出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲル力 ラムクロマトグラフィーに付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出し て題記化合物 (940 mg, 92%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ 一テルから再結晶して融点 162- 164°Cの暗赤色結晶を得た。 N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] obtained in Example 4-35] -2,3 -Dihide mouth-1H-imidazo [5,1-c] [1,4] thiazine -Triylidene}-To a suspension of 4-methylbenzenesulfonamide (1.00 g, 1.80 mmol) and potassium carbonate (373 mg, 2.70 01) in N, N-dimethylformamide (20.0 ml) was added tert-bromoacetic acid. -Butyl ester (0.377 ml, 2.70 mmol) was added. After stirring the reaction mixture at room temperature for 18 hours, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (940 mg, 92%) as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 162-164 ° C.
IR (KBr) V 2982, 2934, 2876, 1742, 1665, 1617, 1557 cm—1. IR (KBr) V 2982, 2934, 2876, 1742, 1665, 1617, 1557 cm- 1 .
'H-NMR (CDC13) : (51.51 (9H, s), 1.46-1.64 (4H, m), 1.79 (3H, s), 1.84-2.12 'H-NMR (CDC1 3) : (51.51 (9H, s), 1.46-1.64 (4H, m), 1.79 (3H, s), 1.84-2.12
(2H, m), 2.29 (3H, s), 2.39 (3H, s), 2.80-2.88 (2H, m), 3.70 (3H, s), 4.98(2H, m), 2.29 (3H, s), 2.39 (3H, s), 2.80-2.88 (2H, m), 3.70 (3H, s), 4.98
(2H, s), 7.24 (2H, d, J=8.0 Hz), 7.90 (2H, d, J=8.2 Hz). (2H, s), 7.24 (2H, d, J = 8.0 Hz), 7.90 (2H, d, J = 8.2 Hz).
元素分析値: C27H32N305S3F5として Elemental analysis: as C 27 H 32 N 3 0 5 S 3 F 5
計算値 (¾) : C, 48.42; H, 4.82; N, 6.27; S, 14.36. Calculated (¾): C, 48.42; H, 4.82; N, 6.27; S, 14.36.
実測値 (%) : C, 48.36; H, 4.66; N, 6.31; S, 14.18. Found (%): C, 48.36; H, 4.66; N, 6.31; S, 14.18.
実施例 18-57 Example 18-57
5 - [α_{[(4-メチルフエニル)スルホニル]イミノ} - 3 -ォキソ - 2, 5, 6 -トリメチル -2, 3 -ジヒドロ- 1H-ィミダゾ [5, l_c] [1 , 4]チアジン- 8-ィル)スルファニル]吉草酸 実施例 18-55 で得られた tert-ブチル 5- [(1-{[(4-メチルフエニル)スルホ二 ル]イミノ } - 3 -ォキソ -2, 5, 6_トリメチル -2, 3-ジヒ ドロ -1H -イミダゾ [5, l—c] [1,4]チアジン— 8 -ィル)スルファニル]ペンタノエート (177 nig, 0.322 mmol) をトリフルォロ酢酸 (1.00ml) に溶かし、 室温で 1時間放置した。 酢酸ェ チルで希釈した後、 溶媒を減圧下に留去して題記化合物 (132 mg, 83¾) を暗赤色 結晶として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 189- 19 Cの 暗赤色結晶を得た。 5- [α _ {[(4-Methylphenyl) sulfonyl] imino} -3-oxo-2,5,6-trimethyl-2,3-dihydro-1H-imidazo [5, l_c] [1,4] thiazine-8 -Yl) sulfanyl] valeric acid tert-butyl 5-[(1-{[(4-methylphenyl) sulfonyl] imino} obtained in Example 18-55} -3-oxo-2,5,6_ Dissolve trimethyl-2,3-dihydro-1H-imidazo [5, l-c] [1,4] thiazine-8-yl) sulfanyl] pentanoate (177 nig, 0.322 mmol) in trifluoroacetic acid (1.00 ml). It was left at room temperature for 1 hour. After dilution with ethyl acetate, the solvent was distilled off under reduced pressure to obtain the title compound (132 mg, 83%) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 189-19 C.
IR (KBr) V: 3400-2400, 2909, 1736, 1661, 1615, 1559 cm"1. IR (KBr) V: 3400-2400, 2909, 1736, 1661, 1615, 1559 cm " 1 .
Ή-NMR (CDCI3) : 51.40-1.68 (4H, m), 1.80 (3H, s), 2.30 (3H, s), 2.33 (2H, t, J=6.9 Hz), 2.41 (3H, s), 2.82 (2H, t, J=6.6 Hz), 3.70 (3H, s), 7.27 (2H, d, J=8.1 Hz), 7.91 (2H, d, J=8.1 Hz). 元素分析値: C21H25N305S3として Ή-NMR (CDCI3): 51.40-1.68 (4H, m), 1.80 (3H, s), 2.30 (3H, s), 2.33 (2H, t, J = 6.9 Hz), 2.41 (3H, s), 2.82 (2H, t, J = 6.6 Hz), 3.70 (3H, s), 7.27 (2H, d, J = 8.1 Hz), 7.91 (2H, d, J = 8.1 Hz). Elemental analysis: as C 21 H 25 N 3 0 5 S 3
計算値 (¾) : C, 50.89; H, 5.08; N, 8.48; S, 19.41. Calculated value (¾): C, 50.89; H, 5.08; N, 8.48; S, 19.41.
実測値 (¾) : C, 50.90; H, 5.04; N, 8.60; S, 19.14. Found (¾): C, 50.90; H, 5.04; N, 8.60; S, 19.14.
実施例 18-58 Example 18-58
5 - [(1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 5, 6-トリメチル -2, 3 -ジヒド口 -1H -ィミダゾ [5, 1-c] [1 , 4]チアジン- 8 -ィル)スルファニル] -N-へ キシルペンタンアミド 5-[(1-{[(4-Methylphenyl) sulfonyl] imino} -3-oxo-2,5,6-trimethyl-2,3-dihydrido-1H-imidazo [5,1-c] [1, 4] Thiazine-8-yl) sulfanyl] -N-hexylpentanamide
実施例 18-57で得られた 5 - [(1 - {[(4 -メチルフエニル)スルホニル]イミノ} - 3- ォキソ - 2, 5, 6 -トリメチル -2, 3 -ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, ]チアジン- 8 - ィル)スルファニル]吉草酸 (31.5mg, 0.0636 mmol) と卜へキシルァミン(0.0096 ml, 0.0699 mmol) の Ν,Ν-ジメチルホルムアミド (1.00 ml) 溶液に、 シァノりん 酸ジェチル(0.0113 ml, 0.0699匪 ol)続いてトリェチルァミン(0.0097 ml, 0.0699 腿 ol) を加えた。反応混合物を室温で 17時間かき混ぜた後、水を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (16.4mg, 45 ) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルか ら再結晶して融点 115-117°Cの暗赤色結晶を得た。 5-[(1-{[(4-Methylphenyl) sulfonyl] imino} -3-oxo-2,5,6-trimethyl-2,3-dihydro-1H-imidazo obtained in Example 18-57 , 1-c] [1,] Thiazine-8-yl) sulfanyl] valeric acid (31.5 mg, 0.0636 mmol) and trihexylamine (0.0096 ml, 0.0699 mmol) in Ν, Ν-dimethylformamide (1.00 ml) To this was added getyl cyanophosphate (0.0113 ml, 0.0699 ol) and then triethylamine (0.0097 ml, 0.0699 thigh). After stirring the reaction mixture at room temperature for 17 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title of compound a (16.4 mg, 45) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 115-117 ° C.
IR (KBr) リ: 3291, 2955, 2930, 2859, 1738, 1645, 1611, 1557 cm"1. IR (KBr): 3291, 2955, 2930, 2859, 1738, 1645, 1611, 1557 cm " 1 .
Ή-NMR (CDC13) : 50.886 (3H, t, J=6.2 Hz), 1.20-1.40 (6H, m), 1.40-1.72 (6H, m), 1.80 (3H, s), 2.11 (2H, t, J=7.0 Hz), 2.29 (3H, s), 2.42 (3H, s), 2.82 (2H, t, J=7.0 Hz), 3.24 (2H, q, J=7.0 Hz), 3.70 (3H, s), 5.37 (1H, bs), 7.28 (2H, d, J=8.0 Hz), 7.91 (2H, d, J=8.4 Hz). Ή-NMR (CDC1 3): 50.886 (3H, t, J = 6.2 Hz), 1.20-1.40 (6H, m), 1.40-1.72 (6H, m), 1.80 (3H, s), 2.11 (2H, t , J = 7.0 Hz), 2.29 (3H, s), 2.42 (3H, s), 2.82 (2H, t, J = 7.0 Hz), 3.24 (2H, q, J = 7.0 Hz), 3.70 (3H, s) ), 5.37 (1H, bs), 7.28 (2H, d, J = 8.0 Hz), 7.91 (2H, d, J = 8.4 Hz).
実施例 18 - 59 Examples 18-59
5, 6 -ジメチル- 1- {[(4-メチルフエニル)スルホニル]ィミノ }- 3 -ォキソ - 8 - [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニル]- 1H -ィミダゾ [5, 1-c] [1,4]チァジン_2(311)-ィル)酢酸 5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -1H- Imidazo [5, 1-c] [1,4] thiazine_2 (311) -yl) acetic acid
実施例 18-56で得られた tert -プチル (5, 6-ジメチル -1 - { [ (4 -メチルフ工ニル) スルホニル]ィミノ }-3-ォキソ - 8 - [ (5, 5, 6, 6, 6_ペン夕フルォ口へキシル)スルフ ァニル] - 1H-イミダゾ [5, 1-c] [1,4]チアジン- 2 (3H) -ィル)アセテート (665 mg, 0.993腿 ol) をトリフルォロ酢酸 (1.00 ml) に溶かし、 室温で 3時間放置した。 酢酸ェチルで希釈した後、 溶媒を減圧下に留去して題記化合物 (153 mg, 25¾) を 暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 203-205°Cの暗赤色結晶を得た。 Tert-Butyl (5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-ino} -8-oxo- (5,5,6,6 obtained in Examples 18-56 Hexyl) sulfanyl]-1H-imidazo [5,1-c] [1,4] thiazine-2 (3H) -yl) acetate (665 mg, 0.993 t) It was dissolved in acetic acid (1.00 ml) and left at room temperature for 3 hours. After dilution with ethyl acetate, the solvent was distilled off under reduced pressure to obtain the title compound (153 mg, 25%) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 203-205 ° C.
IR (KBr) V: 3400-2400, 2949, 1750, 1725, 1667, 1613, 1551 cm一1. IR (KBr) V: 3400-2400, 2949, 1750, 1725, 1667, 1613, 1551 cm one 1.
¾-NMR (CDC13) : (51.52-1.66 (4H, m), 1.80 (3H, d, J=1.2 Hz), 1.88-2.08 (2H, m), 2.29 (3H, d, J=1.2 Hz), 2.40 (3H, s), 2.85 (2H, t, J=6.6 Hz), 5.15 (2H, s), 7.25 (2H, d, J=8.1 Hz), 7.89 (2H, d, J=8.1 Hz). ¾-NMR (CDC1 3): (51.52-1.66 (4H, m), 1.80 (3H, d, J = 1.2 Hz), 1.88-2.08 (2H, m), 2.29 (3H, d, J = 1.2 Hz) , 2.40 (3H, s), 2.85 (2H, t, J = 6.6 Hz), 5.15 (2H, s), 7.25 (2H, d, J = 8.1 Hz), 7.89 (2H, d, J = 8.1 Hz) .
元素分析値: C23H24N305S3F5として Elemental analysis: as C 23 H 24 N 3 0 5 S 3 F 5
計算値 (¾) : C, 45.02; H, 3.94; Ν,' 6.85; S, 15.68. Calculated (¾): C, 45.02; H, 3.94; Ν, '6.85; S, 15.68.
実測値 (%) : C, 45.02; Η, 4.11; , 6.92; S, 15.61. Observed value (%): C, 45.02; Η, 4.11;, 6.92; S, 15.61.
実施例 18 - 60 Examples 18-60
6-[(Ζ)-力ルポキシメチリデン] -卜 {[(4-メチルフエ二リレ)スルホニル]イミノ}-3 - ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニル ]-2, 3, 5, 6-テトラ ヒドロ- IH-イミダゾ [5, 1-c] [1,4]チアジン - 5-カルボン酸 6-[(Ζ) -Hydroxymethylidene] -tri {[(4-methylphenylenyl) sulfonyl] imino} -3-oxo-8-[(5,5,6,6,6-pentaphenol Methylhexyl) sulfanyl] -2,3,5,6-tetrahydro-IH-imidazo [5,1-c] [1,4] thiazine-5-carboxylic acid
実施例 4-571 で得た粗製の化合物 (914 mg, 1.39廳 ol)のメタノール(10 ml)溶 液に室温で攪拌しながら ^水酸化ナトリウム (10ml) を加え、反応液を 10分間 攪拌した。 反応液に 1N塩酸 (14 ml)を加え、 酢酸ェチル (60 ml)で抽出した。 有 機層を 10¾および飽和食塩水で順次洗浄し、 硫酸ナトリウムで乾燥後、 濃縮し、 残渣にジェチルエーテルを加えた。析出物を濾取し、ジェチルエーテルで洗浄後、 乾燥して表題化合物(390 mg, 0.619匪 ol, 44.6%)を橙色粉末として得た。 To a solution of the crude compound obtained in Example 4-571 (914 mg, 1.39 hall) in methanol (10 ml) was added ^ sodium hydroxide (10 ml) with stirring at room temperature, and the reaction solution was stirred for 10 minutes. . 1N Hydrochloric acid (14 ml) was added to the reaction solution, and the mixture was extracted with ethyl acetate (60 ml). The organic layer was washed successively with 10¾ and saturated saline, dried over sodium sulfate, concentrated, and getyl ether was added to the residue. The precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (390 mg, 0.619 bandol, 44.6%) as an orange powder.
¾-腿 (300MHz, CDC13 - DMSO- d6= 19:1, endo/exo異性体混合物): δ 1.67 (4Η, m), 2.02 (2H, m), 2.42 (3H, s), 2.99 (2H, q, J= 7.2 Hz), 3.42 (2H, s, for endo), 5.47 (1H, s, for exo), 6.34 (IH, s, for exo), 7.31 (2H, d, J= 8.4 Hz), 7.88 (2H, d, J= 8.4 Hz), 10.27 (IH, br). ¾- thigh (300MHz, CDC1 3 - DMSO- d 6 = 19: 1, endo / exo isomeric mixture): δ 1.67 (4Η, m ), 2.02 (2H, m), 2.42 (3H, s), 2.99 ( 2H, q, J = 7.2 Hz), 3.42 (2H, s, for exo), 5.47 (1H, s, for exo), 6.34 (IH, s, for exo), 7.31 (2H, d, J = 8.4 Hz) ), 7.88 (2H, d, J = 8.4 Hz), 10.27 (IH, br).
元素分析値: C22H2DN307S3F5として計算値: C, 41.97; H, 3.20; N, 6.67; S, 15.28; F, 15.09 (%) 、 実測値: C, 41.63; H, 3.29; N, 6.65; S, 15.20 (%) . Elemental analysis: C 22 H 2D N 3 0 7 S 3 F 5 Calculated: C, 41.97; H, 3.20 ; N, 6.67; S, 15.28; F, 15.09 (%), Found: C, 41.63; H, 3.29; N, 6.65; S, 15.20 (%).
実施例 18-61 · Example 18-61
N-{5, 6-ジメチル- 3 -ォキソ - 8- [ (3-ピぺリジニルメチル) スルファニル] - 2, 3 -ジ ヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }- 4-メチルベンゼンスル ホンアミド N- {5,6-Dimethyl-3-oxo-8-[(3-piridinylmethyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene}-4-methylbenzenesul Honamide
実施例 4-558で得た化合物(0.20 g, 0.345 mmol) を TFA (1.0 ml)に溶解し、 室温で 18時間放置した。 反応液をトルエンで希釈して濃縮乾固後、 残渣をメタ ノールに溶解し、 1N塩酸 (1.0 ml)を加えて濃縮した。 残渣に THFを加えて懸濁 し、析出物を濾取し、 THFおよび酢酸ェチルで洗浄後、乾燥して表題化合物(120 mg, 0.233 mmol, 67.0%)を赤紫色粉末として得た。 The compound (0.20 g, 0.345 mmol) obtained in Example 4-558 was dissolved in TFA (1.0 ml) and left at room temperature for 18 hours. The reaction solution was diluted with toluene and concentrated to dryness. The residue was dissolved in methanol, and concentrated by adding 1N hydrochloric acid (1.0 ml). The residue was suspended in THF, and the precipitate was collected by filtration, washed with THF and ethyl acetate, and dried to give the title compound (120 mg, 0.233 mmol, 67.0%) as a red-purple powder.
¾-NMR (200MHz, DMS0 - d6+TFA): δ 1.21 (2Η, brq, J= 13 Hz), 1.60 (2H, brq, J= 10 Hz), 1.80 (3H, s), 1.97 (1H, m), 2.17 (3H, s), 2.39 (3H, s), 2.64 (1H, brd, J= 7.4 Hz), 2.72 (1H, m), 2.96 (2H, d, J= 6.6 Hz), 3.24 (2H, brt, J= 10 Hz), 7.39 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 8.59 (1H, br), 8.87 (1H, m), 1.1.74 (1H, br). NMR-NMR (200 MHz, DMS0-d 6 + TFA): δ 1.21 (2Η, brq, J = 13 Hz), 1.60 (2H, brq, J = 10 Hz), 1.80 (3H, s), 1.97 (1H, m), 2.17 (3H, s), 2.39 (3H, s), 2.64 (1H, brd, J = 7.4 Hz), 2.72 (1H, m), 2.96 (2H, d, J = 6.6 Hz), 3.24 ( 2H, brt, J = 10 Hz), 7.39 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 8.59 (1H, br), 8.87 (1H, m), 1.1. 74 (1H, br).
元素分析値: C21H26N403S3 flCl Ό.5H20として計算値: C, 48.12; H, 5.38; N, 10.69; S, 18.35; C1, 6.76 (%) 、 実測値: C, 48.34; H, 5.33; N, 10.38; S, 18.55; Cl, 6.76 ( ) · Elemental analysis: C 21 H 26 N 4 0 3 S 3 flCl Ό.5H 2 0 Calculated: C, 48.12; H, 5.38; N, 10.69; S, 18.35; C1, 6.76 (%), found: C, 48.34; H, 5.33; N, 10.38; S, 18.55; Cl, 6.76 ()
mp. 211.5-213. O mp. 211.5-213. O
実施例 18-62 Example 18-62
N - (5, 6-ジメチル- 3-ォキソ -8- { [ (2S) - ピロリジニルメチル] スルファニ ル}-2,3_ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン)-4-メチルベ ンゼンスルホンアミド N- (5,6-dimethyl-3-oxo-8-{[(2S) -pyrrolidinylmethyl] sulfanyl} -2,3_dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-1-ylidene) -4-methylbenzenesulfonamide
実施例 4-559と同様にして得た粗製の化合物(0.38 g, 0.54腿 ol) を TFA (1.0 ml)に溶解し、室温で 16時間放置した。 反応液をトルエンで希釈して濃縮乾固後、 残渣をメタノールに溶解し、 1N塩酸 (1.0 ml)を加え、 ァセトニトリルで希釈し て濃縮した。 残渣に THFを加えて懸濁し、 析出物を濾取し、 THFおよび酢酸ェチ ルで洗浄後、 乾燥して表題化合物(152 mg, 0.303 mmol, 56.2%)を灰紫色粉末とし て得た。 The crude compound (0.38 g, 0.54 tmol) obtained in the same manner as in Example 4-559 was dissolved in TFA (1.0 ml) and left at room temperature for 16 hours. The reaction solution was diluted with toluene and concentrated to dryness. The residue was dissolved in methanol, 1N hydrochloric acid (1.0 ml) was added, and the mixture was diluted with acetonitrile and concentrated. The residue was suspended in THF, and the precipitate was collected by filtration, washed with THF and ethyl acetate, and dried to give the title compound (152 mg, 0.303 mmol, 56.2%) as a gray-violet powder.
lH - NMR (300MHz, DMS0 - d6): δΐ.65 (1H, m), 1.81 (3H, s), 1.88 (1H, m), 1.91 (1H, m), 2.11 (1H, ) , 2.17 (3H, s), 2.39 (3H, s), 3.17 (2H, m), 3.66 (1H, m), 7.39 (2H, brd, J- 8.7 Hz), 7.85 (2H, d, J= 7.8 Hz), 9.06 (1H, br), 9.39 (1H, br). Ή-NMR (200MHz, DMS0-d6+TFA): δ 1.66 (1H, m), 1.81 (3H, s), 1.90 (2H, brq, J= 7.6 Hz), 2.10 (1H, m), 2.17 (3H, s), 2.39 (3H, s), 3.17 (2H, m), 3.35 (2H, d, J= 6.8 Hz), 3.67 (1H, brquint, J= 6.8 Hz), 7.39 (2H, d, J= 8.0 Hz), 7.87 (2H, d,' J= 8.0 Hz), 9.12 (1H, br), 9.45 (1H, br), 11.73 (1H, br). lH - NMR (300MHz, DMS0 - d 6): δΐ.65 (1H, m), 1.81 (3H, s), 1.88 (1H, m), 1.91 (1H, m), 2.11 (1H,), 2.17 ( 3H, s), 2.39 (3H, s), 3.17 (2H, m), 3.66 (1H, m), 7.39 (2H, brd, J- 8.7 Hz), 7.85 (2H, d, J = 7.8 Hz), 9.06 (1H, br), 9.39 (1H, br). Ή-NMR (200 MHz, DMS0-d 6 + TFA): δ 1.66 (1H, m), 1.81 (3H, s), 1.90 (2H, brq, J = 7.6 Hz), 2.10 (1H, m), 2.17 ( 3H, s), 2.39 (3H, s), 3.17 (2H, m), 3.35 (2H, d, J = 6.8 Hz), 3.67 (1H, brquint, J = 6.8 Hz), 7.39 (2H, d, J = 8.0 Hz), 7.87 (2H, d, 'J = 8.0 Hz), 9.12 (1H, br), 9.45 (1H, br), 11.73 (1H, br).
元素分析値: C2。H24N403S3 i!Cl 0.5H20として計算値: C, 47.09; H, 5.14; , 10.98; S, 18.86; CI, 6.95 (%) 、 実測値: C, 47.14; H, 5.10; N, 10.86; S, 18.85; CI, 7.01 (%) . Elemental analysis: C 2. H 24 N 4 0 3 S 3 i! Cl 0.5H 2 0 Calculated: C, 47.09; H, 5.14;, 10.98; S, 18.86; CI, 6.95 (%), Found: C, 47.14; H, 5.10; N, 10.86; S, 18.85; CI, 7.01 (%).
実施例 18-63 Example 18-63
N- [8- ({[ (2S, 4R) -1-へキサノィル- 4_ヒドロキシピロリジニル]メチル }スルファ 二ル)- 5,6-ジメチル- 3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン _1 -イリデン] -4-メチルベンゼンスルホンアミド N- [8- ({[(2S, 4R) -1-hexanoyl-4_hydroxypyrrolidinyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H -Imidazo [5, tric] [1,4] thiazine_1-ylidene] -4-methylbenzenesulfonamide
実施例 18 - 37 で得た化合物(184 mg, 0.356腿 ol) および塩化へキサノィル (0.060 ml, 0.427 mmol)の THF (3.7 ml)懸濁液にトリェチルァミン(0.148 ml, 1.07 mmol)を室温で加え、 反応液を室温で 1.5時間攪拌した。 反応液を酢酸ェチ ルで抽出し、 脱水乾燥、 濃縮後、 酢酸ェチルおよびジェチルエーテルから結晶化 して表題化合物(176 mg, 0.304醒 ol, 85.4%)を赤色粉末として得た。 Triethylamine (0.148 ml, 1.07 mmol) was added to a suspension of the compound obtained in Examples 18-37 (184 mg, 0.356 tmol) and hexaneyl chloride (0.060 ml, 0.427 mmol) in THF (3.7 ml) at room temperature. The reaction was stirred at room temperature for 1.5 hours. The reaction solution was extracted with ethyl acetate, dehydrated, dried and concentrated, and crystallized from ethyl acetate and getyl ether to give the title compound (176 mg, 0.304 mmol, 85.4%) as a red powder.
'H-NMR (200MHz, CDC13): 50.89 (3H, brt, J= 6.8 Hz), 1.31 (4H, m), 1.62 (2H, brquint, J= 6.5 Hz), 1.78 (3H, s), 2.02 (1H, di, J= 14.1, 6.0 Hz), 2.12 (1H, m), 2.20 (1H, d, J= 8.1 Hz), 2.24 (3H, s), 2.26 (1H, m), 2.42 (3H, s), 3.33 (1H, dd, J= 13.2, 7.2 Hz), 3.44 (1H, dd, J= 13.2, 2.7 Hz), 3.44 (1H, brd, J= 11 Hz), 3.52 (1H, dd, J= 11.1, 4.5 Hz), 4.47 (1H, dq, J= 3.0, 7.2 Hz), 4.53 (1H, m), 7.28 (2H, d, J= 8.1 Hz), 7.81 (2H, d, J= 8.4 Hz), 9.66 (1H, brs) . 'H-NMR (200MHz, CDC1 3): 50.89 (3H, brt, J = 6.8 Hz), 1.31 (4H, m), 1.62 (2H, brquint, J = 6.5 Hz), 1.78 (3H, s), 2.02 (1H, di, J = 14.1, 6.0 Hz), 2.12 (1H, m), 2.20 (1H, d, J = 8.1 Hz), 2.24 (3H, s), 2.26 (1H, m), 2.42 (3H, s), 3.33 (1H, dd, J = 13.2, 7.2 Hz), 3.44 (1H, dd, J = 13.2, 2.7 Hz), 3.44 (1H, brd, J = 11 Hz), 3.52 (1H, dd, J = 11.1, 4.5 Hz), 4.47 (1H, dq, J = 3.0, 7.2 Hz), 4.53 (1H, m), 7.28 (2H, d, J = 8.1 Hz), 7.81 (2H, d, J = 8.4 Hz) ), 9.66 (1H, brs).
元素分析値: C26H34N405S3として計算値: C, 53.96; H, 5.92; N, 9.68; S, 16.62 ( ) 、 実測値: C, 53.91; H, 5.86; N, 9.86; S, 16.40 (%) . Elemental analysis: C 26 H 34 N 4 0 5 S 3 Calculated: C, 53.96; H, 5.92 ; N, 9.68; S, 16.62 (), Found: C, 53.91; H, 5.86 ; N, 9.86 ; S, 16.40 (%).
mp. 153-159°C mp. 153-159 ° C
実施例 18-64 Example 18-64
N - {6 -ヒドロキシ -8-メチル- 1_ォキソ -4 - [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル) スルファニル ]-1, 2, 6, 7, 8, 9 -へキサヒドロ- 3H -イミダゾ [5, 1-c] [1, 4]ベンゾチア ジン- 3-ィリデン }-4-メチルベンゼンスルホンアミド N- {6-Hydroxy-8-methyl-1_oxo-4-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -1,2,6,7,8,9 -Hexahydro-3H-imidazo [5, 1-c] [1, 4] benzothia Zin-3-ylidene} -4-methylbenzenesulfonamide
水素化リチウムアルミニウム (509 mg, 10.7mmol) のテトラヒドロフラン (100 ml) の懸濁液に、 実施例 4-565 で得られた N- {1, 6-ジォキソ -8-メチル -4- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニル ]-1, 2, 6, 7, 8, 9-へキサヒ ドロ- 3H-イミダゾ [5,卜 c] [1,4]ベンゾチアジン- 4-イリデン }-4 -メチルベンゼン スルホンアミド (4.36 g, 7.15匪 ol) のテトラヒドロフラン (100 ml) 溶液を氷 冷下加えた。 反応混合物を室温で 5分かき混ぜた後、 過剰の水素化リチウムアル ミニゥムを水で分解し、 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (1.32 g, 30¾) を暗赤色 結晶として得た。酢酸ェチル -n -へキサンから再結晶して融点 190-192 の暗赤色 結晶を得た。 A suspension of lithium aluminum hydride (509 mg, 10.7 mmol) in tetrahydrofuran (100 ml) was added to the suspension of N- {1,6-dioxo-8-methyl-4-[(5 , 5,6,6,6-Pentafluorohexyl) sulfanyl] -1,2,6,7,8,9-Hexahydro-3H-imidazo [5, tric] [1,4] benzothiazine A solution of 4-ylidene} -4-methylbenzenesulfonamide (4.36 g, 7.15 bandol) in tetrahydrofuran (100 ml) was added under ice-cooling. After stirring the reaction mixture at room temperature for 5 minutes, excess lithium hydride was decomposed with water, 1N hydrochloric acid was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (1.32 g, 30¾) as a dark red crystals. Recrystallization from ethyl acetate-n-hexane gave dark red crystals with a melting point of 190-192.
IR (KBr) V: 3700-2800, 3440, 3300,2953, 2930, 1750, 1732, 1655, 1617, 1557 cm"1. IR (KBr) V: 3700-2800, 3440, 3300,2953, 2930, 1750, 1732, 1655, 1617, 1557 cm " 1 .
Ή-NMR (CDC13) : 51.06 (3HX0.80, d, J=6.6 Hz), 1.20 (3HX0.2, d, J=6.6Hz), 1.22-1.36 (1H, m), 1.58-1.84 (6H, m), 1.94-2.20 (2H, m), 2.38-2.50 (1H, m), Ή-NMR (CDC1 3): 51.06 (3HX0.80, d, J = 6.6 Hz), 1.20 (3HX0.2, d, J = 6.6Hz), 1.22-1.36 (1H, m), 1.58-1.84 (6H , m), 1.94-2.20 (2H, m), 2.38-2.50 (1H, m),
2.42 (3H, s), 2.71 (1H, dd, J=18.3, 5.4 Hz), 3.02 (2H, dt, J=7. , 1.2 Hz),2.42 (3H, s), 2.71 (1H, dd, J = 18.3, 5.4 Hz), 3.02 (2H, dt, J = 7., 1.2 Hz),
4.14-4.25 (1H, m), 7.29 (2H, d, J=9.0Hz), 7.84 (2HX0.8, d, J=8.4 Hz), 7.874.14-4.25 (1H, m), 7.29 (2H, d, J = 9.0Hz), 7.84 (2HX0.8, d, J = 8.4Hz), 7.87
(2HX0.2, d, J=8.4 Hz), 9.56 (1HX0.2, bs), 9.66 (1HX0.8, bs). (2HX0.2, d, J = 8.4 Hz), 9.56 (1HX0.2, bs), 9.66 (1HX0.8, bs).
元素分析値: C24H26N304S3F3として Elementary analysis: as C 24 H 26 N 3 0 4 S 3 F 3
計算値 (%) : C, 47.13; H, 4.28; N, 6.87; S, 15.73. Calculated (%): C, 47.13; H, 4.28; N, 6.87; S, 15.73.
実測値 (¾) : C, 47.13; H, 4.51; , 6.69; S, 15.95. Found (¾): C, 47.13; H, 4.51;, 6.69; S, 15.95.
実施例 18-65 Example 18-65
N-{8-メチル- 1-ォキソ -4- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル]- 1,2-ジヒドロ- 3H-イミダゾ [5,卜 c] [1,4]ベンゾチアジン - 3-ィリデン }- 4 -メ チルベンゼンスルホンアミド N- {8-methyl-1-oxo-4-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -1,2-dihydro-3H-imidazo [5, tric ] [1,4] Benzothiazine-3-ylidene} -4-methylbenzenesulfonamide
実施例 18-64で得られた N - {6-ヒドロキシ- 8-メチル -1-ォキソ -4- [(5, 5, 6, 6, 6- ペンタフルォ口へキシル)スルファニル] - 1, 2, 6, 7, 8, 9_へキサヒドロ- 3H -ィミダ ゾ [5,卜 c] [1,4]ベンゾチアジン - 3 -ィリデン }- 4-メチルベンゼンスルホンアミド (l.OOg, 1.63腿 ol)をトルエン (10.0ml)および無水酢酸 (0.463 ml, 4.90画01) に溶解し、 p-トルエンスルホン酸 1水和物 (262 mg, 1.31匪 ol) を加えて lOOt: で 10分間加熱撹拌した。 反応液を濃縮後、残渣を酢酸ェチルで希釈し、 水洗、 乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (175 mg, 18%) を暗赤色結 晶として得た。 酢酸ェチルから再結晶して融点 203- 205°Cのオレンジ色結晶を得 た。 N- {6-hydroxy-8-methyl-1-oxo-4-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] obtained in Examples 18-64-1,2 6,7,8,9_Hexahydro-3H-imidazo [5, tric] [1,4] benzothiazine-3-ylidene} -4-methylbenzenesulfonamide (l.OOg, 1.63 t) (10.0 ml) and acetic anhydride (0.463 ml, 4.90 fraction 01) Then, p-toluenesulfonic acid monohydrate (262 mg, 1.31 ol) was added, and the mixture was heated and stirred with lOOt: for 10 minutes. The reaction mixture was concentrated, the residue was diluted with acetic acid Echiru, washed with water, after Drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (175 mg, 18%) and the obtained as dark red crystals . Recrystallization from ethyl acetate gave orange crystals with a melting point of 203-205 ° C.
IR (KBr) リ: 3200, 3050, 2932, 1728, 1626, 1597, 1557, 1493 cm"1. IR (KBr): 3200, 3050, 2932, 1728, 1626, 1597, 1557, 1493 cm " 1 .
Ή— NMR (CDC13) : (51.66-1.88 (4H, m), 1.98-2.16 (2H, m) , 2.31 (3H, s), 2.42 Ή- NMR (CDC1 3): ( 51.66-1.88 (4H, m), 1.98-2.16 (2H, m), 2.31 (3H, s), 2.42
(3H, s), 3.03 (2H, i, J=7.2 Hz), 6.83 (1H, d, J=8.1 Hz), 6.90 (1H, dd, J-8.7,(3H, s), 3.03 (2H, i, J = 7.2 Hz), 6.83 (1H, d, J = 8.1 Hz), 6.90 (1H, dd, J-8.7,
0.6 Hz), 7.30 (2H, d, J=8.1 Hz), 7.86 (2H, d, J=8.4 Hz), 8.30 (1H, d, J=0.60.6 Hz), 7.30 (2H, d, J = 8.1 Hz), 7.86 (2H, d, J = 8.4 Hz), 8.30 (1H, d, J = 0.6
Hz), 9.75 (1H, bs). Hz), 9.75 (1H, bs).
元素分析値: C24H22N303S3F5として Elemental analysis: as C 24 H 22 N 3 0 3 S 3 F 5
計算値 (¾) : C, 48.72; H, 3.75; N, 7.10; S, 16.26. Calculated value (¾): C, 48.72; H, 3.75; N, 7.10; S, 16.26.
実測値 (%) : C, 48.58; H, 3.72; N, 7.12; S, 16.32. Found (%): C, 48.58; H, 3.72; N, 7.12; S, 16.32.
実施例 18-66 Example 18-66
酢酸 (i,4- cis- 4- {[(5,6-ジメチル-1_{[(4-メチルフェニル)スルホニル]ィミ ノ }_3-ォキソ -2, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1,4]チアジン - 8 -ィル)スルフ 7ニル]メチル }シクロへキシルメチル)メチル Acetic acid (i, 4-cis-4-([(5,6-Dimethyl-1 _ {[(4-methylphenyl) sulfonyl] imino)} _ 3-oxo-2,3-dihydrido-1H-imidazo [5 , 1-c] [1,4] thiazine-8-yl) sulfenyl] methyl} cyclohexylmethyl) methyl
実施例 4-573で得た粗製の化合物 (0.62 g, 0.70匪 ol) および無水酢酸(2 ml) のピリジン (4ml)溶液を室温で 16時間攪拌した。 反応液を濃縮乾固し;酢酸ェチ ル (35 ml)で溶解し、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウム で乾燥し、 濃縮して粗油状物を得た。 これをシリカゲル(10 g)のクロマトグラフ ィー (酢酸ェチルーへキサン、 2:8-4:6) に付し、 赤色画分を濃縮後酢酸ェチルお よびメタノールで希釈した。 析出物を濾取し、 メタノールで洗浄後、 乾燥して表 題化合物(86 mg, 0.156腿 ol, 22.3 )を赤色粉末として得た。 A solution of the crude compound (0.62 g, 0.70 ol) obtained in Example 4-573 and acetic anhydride (2 ml) in pyridine (4 ml) was stirred at room temperature for 16 hours. The reaction solution was concentrated to dryness; dissolved in ethyl acetate (35 ml), washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to obtain a crude oil. This was subjected to chromatography on silica gel (10 g) (ethyl acetate-hexane, 2: 8-4: 6), and the red fraction was concentrated and then diluted with ethyl acetate and methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (86 mg, 0.156 mol, 22.3) as a red powder.
^-NMR (300MHz, CDC13): 51.42 (4H, m), 1.54 (2H, m), 1.62 (2H, m), 1.79 (3H, q, J= 1.2 Hz), 1.85 (2H, m), 2.07 (3H, s), 2.26 (3H, q, J- 0.9 Hz), 2.41 (3H, s), 2.89 (2H, d, J= 7.5 Hz), 3.98 (2H, d, J= 6.9 Hz), 7.27 (2H, d, J= 8.1 Hz), 7.84 (2H, d, J= 8.1 Hz), 9.61 (1H, brs)'. ^ -NMR (300MHz, CDC1 3) : 51.42 (4H, m), 1.54 (2H, m), 1.62 (2H, m), 1.79 (3H, q, J = 1.2 Hz), 1.85 (2H, m), 2.07 (3H, s), 2.26 (3H, q, J- 0.9 Hz), 2.41 (3H, s), 2.89 (2H, d, J = 7.5 Hz), 3.98 (2H, d, J = 6.9 Hz), 7.27 (2H, d, J = 8.1 Hz), 7.84 (2H, d, J = 8.1 Hz), 9.61 (1H, brs) '.
元素分析値: C25H31N305S3として計算値: C, 54.62; H, 5.68; N, 7.64; S, 17.50 (%) 、 実測値: C, 54.74; H, 5.67; N, 7.73; S, 17.36 (%) . 実施例 18 - 67 Elemental analysis: C 25 H 31 N 3 0 5 S 3 Calculated: C, 54.62; H, 5.68 ; N, 7.64; S, 17.50 (%), Found: C, 54.74; H, 5.67; N, 7.73; S, 17.36 (%). Examples 18-67
メタンスルホン酸 (4- {[(5,6-ジメチル -1-{ [(4-メチルフエニル)スルホ二ル]ィ ミノ } -3-ォキ V-2, 3-ジヒドロ- 1H -イミダゾ [5, 1 -c] [1 , 4]チアジン- 8 -ィル)スル 実施例 4-573 で得た化合物(660 mg, 1.30画 1, cis/t画: 92:8混合物) およびメタンスルホニルクロリド(0.112 ml, 1.43匪 ol)の THF (13 ml)溶液に 0°C で攪拌しながらトリェチルァミン(0.216 ml, 1.56讓 ol)を加え、 反応液を 0°Cな いし 10°Cで 2時間攪拌した。 反応液を酢酸ェチル (45 m)で希釈し、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮して赤色油状物を 得た。 これを酢酸ェチルで希釈して、 析出物を濾琅してジェチルエーテルで洗浄 後、 乾燥して表題化合物 (355 mg, 0.606匪 ol, 46.6%)を赤褐色粉末として得た。 'H-NMR (300MHz, CDC13): 51.46 (4H, m), 1.63 (4H, m), 1.79 (3H, s), 1.83 (1H, m), 1.96 (1H, m), 2.26 (3H, s), 2.42 (3H, s), 2.90 (2H, d, J= 7.2 Hz), 3.02 (3H, s), 4.13 (2H, d, J= 7.2 Hz), 7.27 (2H, d, J= 8.4 Hz), 7.83 (2H, d, J= 8.4 Hz), 9.62 (1H, brs). Methanesulfonic acid (4-{[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oki) -3-oxo V-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazin-8-yl) sul The compound obtained in Example 4-573 (660 mg, 1.30 fraction 1, cis / t fraction: 92: 8 mixture) and methanesulfonyl chloride (0.112 Triethylamine (0.216 ml, 1.56 mL) was added to a THF (13 ml) solution of 0.1 ml, 1.43 bandol) at 0 ° C with stirring, and the reaction solution was stirred at 0 ° C or 10 ° C for 2 hours. The reaction solution was diluted with ethyl acetate (45 m), washed with 0.1 N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated to obtain a red oily substance. after washing the precipitate was濾琅by Jeffrey chill ether, dried to give the title compound (355 mg, 0.606 negation ol, 46.6%) was obtained as a red-brown powder 'H-NMR (300MHz, CDC1 3):. 51.46 (4H, m), 1.63 (4H, m), 1.79 (3H, s), 1.83 (1H, m), 1.96 (1H, m), 2.26 (3H, s), 2.42 (3H, s), 2.90 (2H, d, J = 7.2 Hz), 3.02 (3H, s), 4.13 (2H , d, J = 7.2 Hz), 7.27 (2H, d, J = 8.4 Hz), 7.83 (2H, d, J = 8.4 Hz), 9.62 (1H, brs).
融点: 177.5-178.5°C Melting point: 177.5-178.5 ° C
実施例 18-68 Example 18-68
酢酸 (l,4_trans-4-{[(5,6_ジメチル-卜 {[(4-メチルフエニル)スルホニル]イミ ノ}_3_ォキソ_2,3-ジヒドロ- 1H-イミダゾ [5,1- c] [1, 4]チアジン- 8-ィル)スルフ ァニル]メチル }シクロへキシルメチル)メチル Acetic acid (l, 4_trans-4-{[(5,6_dimethyltolu [[(4-methylphenyl) sulfonyl] imino} _3_oxo_2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazin-8-yl) sulfanyl] methyl} cyclohexylmethyl) methyl
実施例 4-574で得た化合物(220 mg, 0.433腿 ol) および無水酢酸(0.6 ml)のピ リジン(2 ml)溶液を室温で 2.5日間攪拌した。 反応液を濃縮乾固し、 メタノール で希釈した。析出物を濾取し、メタノールで洗浄後、乾燥して表題化合物 (207 mg, 0.376匪 ol, 87.0»を赤紫色粉末として得た。 A solution of the compound obtained in Example 4-574 (220 mg, 0.433 mol) and acetic anhydride (0.6 ml) in pyridine (2 ml) was stirred at room temperature for 2.5 days. The reaction was concentrated to dryness and diluted with methanol. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (207 mg, 0.376 ol, 87.0 »as a red-purple powder.
Ή-NMR (200MHz, CDC13): δ 1.00 (4Η, brquint, J= 10.2 Hz), 1.59 (2H, m), 1.79 (3H, s), 1.83 (2H, m), 1.93 (2H, brd, J= 7.4 Hz), 2.07 (3H, s), 2.27 (3H, s), 2.42 (3H, s), 2.83 (2H, d, J= 6.8 Hz), 3.90 (2H, d, J= 6.2 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.65 (1H, brs). 元素分析値: C25H31N305S3として計算値: C, 54.62; H, 5.68; N, 7.64; S, 17.50 Ή-NMR (200MHz, CDC1 3 ): δ 1.00 (4Η, brquint, J = 10.2 Hz), 1.59 (2H, m), 1.79 (3H, s), 1.83 (2H, m), 1.93 (2H, brd, J = 7.4 Hz), 2.07 (3H, s), 2.27 (3H, s), 2.42 (3H, s), 2.83 (2H, d, J = 6.8 Hz), 3.90 (2H, d, J = 6.2 Hz) , 7.28 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.65 (1H, brs). Elemental analysis: C 25 H 31 N 3 0 5 S 3 Calculated: C, 54.62; H, 5.68 ; N, 7.64; S, 17.50
(%) 、 実測値: C, 54.46; H, 5.54; N, 7.50; S, 17.28 (%) . (%), Found: C, 54.46; H, 5.54; N, 7.50; S, 17.28 (%).
融点: 183.5-184.0°C Melting point: 183.5-184.0 ° C
実施例 18-69 Example 18-69
N-[8- ({[4- (フルォロメチル)シクロへキシル]メチル }スルファニル )-5, 6-ジメチ ル- 3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン - 1 -ィリデン ]-4 - メチルベンゼンスルホンアミド N- [8-({[4- (Fluoromethyl) cyclohexyl] methyl} sulfanyl) -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene] -4-methylbenzenesulfonamide
実施例 18-67で得た粗製め化合物(純度 80%, 270 mg, 0.370匪。1, cis/trans= 92:8混合物)、 フッ化テトラブチルアンモニゥム (TBAF, 300 mg, 0.926膽 ol)お よび水(0.025 ml, 1.39腿 ol)のァセトニトリル(0.5 ml) 溶液を 80°C で 3時間 攪拌した。 反応液に TBAF (300 mg, 0.926扇 ol)を加えてさらに 8(T で 3.5時 間攪拌した。 反応液を濃縮後、 酢酸ェチル (80 ml)で抽出した。 有機層を水、 0.1N 塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固して濃 紫色油状物を得た。 これをシリカゲル(10 g)のクロマトグラフィー (酢酸ェチル 一へキサン、 2:8-4:6) で精製し、 メタノールから結晶化して表題化合物 (cis/trans= 4:1, 72 mg, 0.141 匪 ol, 38.2%)を灰紫色粉末として得た。 Crude compound obtained in Examples 18-67 (purity 80%, 270 mg, 0.370 bandages, 1, cis / trans = 92: 8 mixture), tetrabutylammonium fluoride (TBAF, 300 mg, 0.926 holol) ) And water (0.025 ml, 1.39 tmol) in acetonitrile (0.5 ml) were stirred at 80 ° C for 3 hours. TBAF (300 mg, 0.926 finol) was added to the reaction mixture, and the mixture was further stirred for 8 hours (3.5 hours at T.) The reaction mixture was concentrated and extracted with ethyl acetate (80 ml). After washing with water and saturated saline, drying over sodium sulfate and concentration to dryness gave a dark purple oil which was chromatographed on silica gel (10 g) (ethyl acetate monohexane, 2: 8- 4: 6) and crystallized from methanol to give the title compound (cis / trans = 4: 1, 72 mg, 0.141 ol, 38.2%) as a grey-purple powder.
Ή-NMR (300MHz, CDC13, major cis-isomer): δ 1.46 (4Η, m), 1.59 (4Η, m), 1.79 (3Η, q, J= 0.9 Hz), 1.84 (1H, m), 1.93 (1H, m), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 2.91 (2H, d, J= 7.5 Hz), 4.33 (2H, dd, J= 47.4, 6.6 Hz), 7.27 (2H, d, J= 8.4 Hz), 7.84 (2H, dt, J= 8.4, 2.0 Hz), 9.61 (1H, brs). Ή-NMR (300MHz, CDC1 3 , major cis-isomer): δ 1.46 (4Η, m), 1.59 (4Η, m), 1.79 (3Η, q, J = 0.9 Hz), 1.84 (1H, m), 1.93 (1H, m), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 2.91 (2H, d, J = 7.5 Hz), 4.33 (2H, dd, J = 47.4, 6.6 Hz) , 7.27 (2H, d, J = 8.4 Hz), 7.84 (2H, dt, J = 8.4, 2.0 Hz), 9.61 (1H, brs).
元素分析値: C23H28N303S3Fとして計算値: C, 54.20; H, 5.54; N, 8.24; S, 18.87; F, 3.73 (%) 、 実測値: C, 54.50; H, 5.47; N, 8.13; S, 18.83; F, 3.54 (%) . 融点: 186.0-187.5°C Elemental analysis: C 23 H 28 N 3 0 3 S 3 F Calculated: C, 54.20; H, 5.54 ; N, 8.24; S, 18.87; F, 3.73 (%), Found: C, 54.50; H , 5.47; N, 8.13; S, 18.83; F, 3.54 (%). Melting point: 186.0-187.5 ° C
実施例 18-70 Example 18-70
N- {8- [ (5 -ヒド口キシ- 2, 2-ジメチルぺンチル)スルファニル] -5, 6-ジメチル- 3-ォ キソ- 2, 3-ジヒドロ- 1H-イミダゾ [5 , 1 -c] [1, 4]チアジン-卜イリデン } -4-メチルべ ンゼンスルホンアミド N- {8-[(5-Hydroxy-2,2-dimethylpentyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c ] [1, 4] thiazine-triylidene} -4-methylbenzenesulfonamide
実施例 4-575で得た化合物(418 mg, 0.681醒 ol) の THF-メタノール(1: 1, 6 ml) 懸濁液に IN水酸化ナトリウム水溶液(3.4 ml, 3.4腿 ol) を室温で加え、 3時間 攪拌した。 反応液に IN塩酸(3.4 ml)を加えて 30分間攪拌した。 析出物を濾取し てメタノール一水(1:1)およびメタノールで洗浄後、乾燥して表題化合物(275 mg, 0.555讓 ol, 81.5%)を紫色粉末として得た。 To a suspension of the compound obtained in Example 4-575 (418 mg, 0.681 mmol) in THF-methanol (1: 1, 6 ml) was added an aqueous solution of sodium hydroxide IN (3.4 ml, 3.4 mol) at room temperature. , 3 hours Stirred. IN hydrochloric acid (3.4 ml) was added to the reaction solution, and the mixture was stirred for 30 minutes. The precipitate was collected by filtration, washed with methanol-one water (1: 1) and methanol, and dried to give the title compound (275 mg, 0.555 mL, 81.5%) as a purple powder.
'H-NMR (200MHz, CDC13): δ 1.01 (6Η, s), 1.38 (2H, m), 1.41 (1H, br), 1.52 (2H, m), 1.79 (3H, q, J= 1.2 Hz), 2.26 (3H, q, J= 1.2 Hz), 2.41 (3H, s), 2.90 (2H, s), 3.63 (2H, t, J= 6.2 Hz), 7.28 (2H, d, J= 8.4 Hz), 7.87 (2H, d, J= 8.4 Hz), 9.60 (1H, brs). 'H-NMR (200MHz, CDC1 3): δ 1.01 (6Η, s), 1.38 (2H, m), 1.41 (1H, br), 1.52 (2H, m), 1.79 (3H, q, J = 1.2 Hz ), 2.26 (3H, q, J = 1.2 Hz), 2.41 (3H, s), 2.90 (2H, s), 3.63 (2H, t, J = 6.2 Hz), 7.28 (2H, d, J = 8.4 Hz) ), 7.87 (2H, d, J = 8.4 Hz), 9.60 (1H, brs).
元素分析値: C22H29N304S3として計算値: C, 53.31; H, 5.90; , 8.48; S, 19.41 (%) 、 実測値: C, 53.43; H, 5.91; N, 8.22; S, 19.13 (%) . Elemental analysis: C 22 H 29 N 3 0 4 Calculated S 3: C, 53.31; H , 5.90;, 8.48; S, 19.41 (%), Found: C, 53.43; H, 5.91 ; N, 8.22 S, 19.13 (%).
融点: 159.0- 159.5°C Melting point: 159.0-159.5 ° C
実施例 18 - 71 Examples 18-71
N - {6- [(5, 6-ジメチル- 1- {[(4-メチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1,4]チアジン- 8-ィル)スルファニル]へキシ ル}へキサンアミド N- {6-[(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1 , 4] Thiazine-8-yl) sulfanyl] hexyl} hexaneamide
実施例 18-8 におけるシリカゲルクロマトグラフィ一で酢酸ェチルートルェ ン (4:6)の画分を濃縮乾固後ジェチルェ一テルから結晶化して表題化合物(149 mg, 0.257腿 ol)を赤色粉末として得た。 The fraction of ethyl acetate (4: 6) was concentrated to dryness by silica gel chromatography in Example 18-8 and crystallized from getyl ether to give the title compound (149 mg, 0.257 mol) as a red powder.
¾-NMR (300MHz, CDC13) : (50.89 (3H, t, J= 6.9 Hz), 1.31 (4H, m), 1.43 (4H, m), 1.45 (2H, m), 1.51 (2H, quint, J= 7.4 Hz), 1.63 (2H, quint, J= 7.1 Hz), 1.79 (3H, q, J= 0.9 Hz), 2.16 (2H, t, J= 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.93 (2H, t, J= 7.4 Hz), 3.25 (2H, q, J- 6.7 Hz), 5.49 (1H, m), 7.29 (2H, d, J= 8.7 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.64 (1H, brs). ¾-NMR (300MHz, CDC1 3 ): (50.89 (3H, t, J = 6.9 Hz), 1.31 (4H, m), 1.43 (4H, m), 1.45 (2H, m), 1.51 (2H, quint, J = 7.4 Hz), 1.63 (2H, quint, J = 7.1 Hz), 1.79 (3H, q, J = 0.9 Hz), 2.16 (2H, t, J = 7.5 Hz), 2.26 (3H, s), 2.42 (3H, s), 2.93 (2H, t, J = 7.4 Hz), 3.25 (2H, q, J-6.7 Hz), 5.49 (1H, m), 7.29 (2H, d, J = 8.7 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.64 (1H, brs).
元素分析値: C27H38N404S3 O.3H20として計算値: C, 55.51; H, 6.66; , 9.59; S, 16.47 (%) 、 実測値: C, 55.49; H, 6.59; N, 9.52; S, 16.49 (%) . Elemental analysis: C 27 H 38 N 4 0 4 S 3 O.3H 2 0 Calculated: C, 55.51; H, 6.66 ;, 9.59; S, 16.47 (%), Found: C, 55.49; H, 6.59; N, 9.52; S, 16.49 (%).
mp. 121.0-122.0°C mp.121.0-122.0 ° C
実施例 18 - 72 Examples 18-72
t er t -ブチル 5- [ (5, 6 -ジメチル- 1 - { [ (4 -メチルフエニル)スルホニル]イミノ } - 3- ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 8 -ィル)スルファニル] ペンタノエー卜 実施例 18-21で得られた 5 - [(5, 6-ジメチル- 1-{ [(4-メチルフエニル)スルホ二 ル]イミノ卜 3_ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,1-c] [1,4]チアジン- 8-ィル) スルファニル]吉草酸 (560 mg, 1.16翻 ol) とトリエチルァミン (0.162 ml, 1.16 画 ol) のテトラヒドロフラン(2.00 ml)溶液に、 2,4,6-トリクロ口べンゾイル ク ロリド (0.187 ml, 1.16匪 ol) を加えた。 反応混合物を室温で 3時間かき混ぜた 後、 トリェチルァミン塩酸塩を濾過により除去した。 瀘液を濃縮し、 残渣のトル ェン (3.00 ml) 溶液に、 ter卜ブタノ一ル (174 mg, 2.33腿 ol) と 4-ジメチル アミノピリジン (287 mg, 2.33顧 ol) のトルエン (2.00 ml) 溶液を加えた。 反 応混合物を室温で 16時間かき混ぜた後、水を加えて酢酸ェチルで抽出した。抽出 液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去した。 残留物をシリカゲルカラ ムクロマトグラフィ一に付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して 題記化合物 (190 mg, 30¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルェ一 テルから再結晶して融点 124- 126°Cの暗赤色結晶を得た。 tert-butyl 5-[(5,6-dimethyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-8-yl) sulfanyl] pentanoate 5-[(5,6-Dimethyl-1-{[(4-methylphenyl) sulfonyl] iminoto 3_oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1,4] Thiazin-8-yl) sulfanyl] valeric acid (560 mg, 1.16 ol) and triethylamine (0.162 ml, 1.16 ol) in tetrahydrofuran (2.00 ml) , 4,6-Triclo benzoyl chloride (0.187 ml, 1.16 marl ol) was added. After stirring the reaction mixture at room temperature for 3 hours, triethylamine hydrochloride was removed by filtration. The filtrate was concentrated, and a solution of the residue in toluene (3.00 ml) was added to a solution of tert-butanol (174 mg, 2.33 mol) and 4-dimethylaminopyridine (287 mg, 2.33 ml) in toluene (2.00 ml). ) Solution was added. After stirring the reaction mixture at room temperature for 16 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (190 mg, 30¾) as dark red crystals. Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 124-126 ° C.
IR (KBr)リ: 3220, 2980, 2934, 1726, 1661, 1620, 1563 cm—1. IR (KBr): 3220, 2980, 2934, 1726, 1661, 1620, 1563 cm— 1 .
Ή-NMR (CDC13) : (51.45 (9H, s), 1.64-1.76 (4H, m), 1.78 (3H, s), 2.20-2.30 (2H, m), 2. 6 (3H, s), 2.42 (3H, s), 2.89-2.98 (2H, ni), 7.29 (2H, d, J=8.0 Hz), 7.85 (2H, d, J=8.4 Hz), 9.61 (1H, bs). Ή-NMR (CDC1 3): (51.45 (9H, s), 1.64-1.76 (4H, m), 1.78 (3H, s), 2.20-2.30 (2H, m), 2. 6 (3H, s), 2.42 (3H, s), 2.89-2.98 (2H, ni), 7.29 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 9.61 (1H, bs).
元素分析値: C24H31N305S3として Elemental analysis: as C 24 H 31 N 3 0 5 S 3
計算値 (%) : C, 53.61; H, 5.81; , 7.81; S, 17.89. Calculated value (%): C, 53.61; H, 5.81;, 7.81; S, 17.89.
実測値 (%) : C, 53.33; H, 5.55; N, 7.86; S, 17.77. Found (%): C, 53.33; H, 5.55; N, 7.86; S, 17.77.
実施例 18-73 Example 18-73
8- (ィソぺンチルスルファニル) -1-{ [ (4-メチルフエニル)スルホニル]イミノト 3- ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン -5-カルボン酸 8- (isopentylsulfanyl) -1-{[(4-methylphenyl) sulfonyl] iminoto 3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 5-carboxylic acid
実施例 4 - 416で得た化合物(169 mg, 0.341腿 ol)の THF-メタノール (1:1, 2.0 ml)の懸濁液に室温で攪拌しながら 1N水酸化ナトリウム (1.02 ml, 1.02匪 ol) を加え、 反応液を室温で 2時間攪拌した。 反応液に 0.1 N塩酸 (20 ml)を加えて 酢酸ェチルで抽出した。 有機層を水および飽和食塩水で洗浄後、 硫酸ナトリウム で乾燥し、 濃縮した。 残渣にジェチルェ一テルを加え、 析出物を濾取し、 ジェチ ルェ一テルで洗浄、 乾燥して標題化合物 (131 mg, 0.281讓 oi)を赤色粉末として 得た。 In a suspension of the compound obtained in Example 4-416 (169 mg, 0.341 mol) in THF-methanol (1: 1, 2.0 ml) was added 1N sodium hydroxide (1.02 ml, 1.02 ml) while stirring at room temperature. ) Was added, and the reaction solution was stirred at room temperature for 2 hours. The reaction mixture was added with 0.1 N hydrochloric acid (20 ml) and extracted with ethyl acetate. The organic layer was washed with water and saturated saline, dried over sodium sulfate, and concentrated. The residue was mixed with diethyl ether, and the precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (131 mg, 0.281 讓) as a red powder. Obtained.
一 NMR (200MHz, CDC13- DMSO- d 19:1) : δ 0.91 (6H, d, J= 6.2 Hz), 1.56 (2H, brquint, J= 7.4 Hz), 1.65 (1H, sixtet, J= 6.6 Hz), 2.43 (3H, s), 2.95 (2H, t, J- 7.7 Hz), 6.02 (1H, s), 7.30 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 10.01 (1H, br). One NMR (200MHz, CDC1 3 - DMSO- d 19: 1): δ 0.91 (6H, d, J = 6.2 Hz), 1.56 (2H, brquint, J = 7.4 Hz), 1.65 (1H, sixtet, J = 6.6 Hz), 2.43 (3H, s), 2.95 (2H, t, J- 7.7 Hz), 6.02 (1H, s), 7.30 (2H, d, J = 8.4 Hz), 7.85 (2H, d, J = 8.4 Hz) Hz), 10.01 (1H, br).
元素分析値: C19H21N305S3として計算値: C, 48.80; H, 4.53; N, 8.99; S, 20.57Elemental analysis: C 19 H 21 N 3 0 5 S 3 Calculated: C, 48.80; H, 4.53 ; N, 8.99; S, 20.57
(%) 、 実測値: C, 48.87; H, 4.57; N, 8.77; S, 20.32 (%) . (%), Found: C, 48.87; H, 4.57; N, 8.77; S, 20.32 (%).
融点. 211- 216°C Melting point. 211- 216 ° C
実施例 18-74 Example 18-74
8- (ィソペンチルスルファニル) -1-{[ (4-メチルフエニル)スルホニル]イミノ }-3 - ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5,1 - c] [1,4]チアジン- 5-力ルポキサミド 実施例 18-73で得た化合物(201 mg, 0.430顧 ol)、 H0BT (70 mg, 0.516画 1) および WSC (99 mg, 0.519匪 ol)の THF (4.0 ml)溶液に 25 アンモニア水 (0.049 ml, 0.645 mmol)を室温で加えた。 混合物を室温で 3時間攪拌した。 反応液を酢酸 ェチルで希釈し、 2%炭酸水素ナトリウム水溶液、 希塩酸、 水および飽和食塩水で 洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣をジェチルェ一テルから結晶 化して得た粉末(173 mg)をメタノールから再結晶して標題化合物(84 mg, 0.180 腿 ol)を赤色粉末として得た。 , . 8- (Isopentylsulfanyl) -1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 5-forceluxamide A solution of the compound obtained in Example 18-73 (201 mg, 0.430 ol), H0BT (70 mg, 0.516 fraction 1) and WSC (99 mg, 0.519 ol) in a THF (4.0 ml) solution was added to the solution. Aqueous ammonia (0.049 ml, 0.645 mmol) was added at room temperature. The mixture was stirred at room temperature for 3 hours. The reaction solution was diluted with ethyl acetate, washed with a 2% aqueous sodium hydrogen carbonate solution, diluted hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. A powder (173 mg) obtained by crystallizing the residue from geethylether was recrystallized from methanol to give the title compound (84 mg, 0.180 mol) as a red powder. ,.
¾-NMR (200MHz, CDC13_DMS0- d6= 9:1) : δ 0.92 (6Η, d, J= 6.2 Hz), 1.56 (2H, brquint, J= 7.6 Hz), 1.65 (1H, sixtet, J= 7 Hz), 2.43 (3H, s), 2.95 (2H, t, J= 7.5 Hz), 5.75 (1H, s), 6.33 (2H, m), 7.30 (2H, d, J= 8.0 Hz), 7.85 (2H, d, J= 8.4 Hz), 10.30 (1H, br). ¾-NMR (200MHz, CDC1 3 _DMS0- d 6 = 9: 1): δ 0.92 (6Η, d, J = 6.2 Hz), 1.56 (2H, brquint, J = 7.6 Hz), 1.65 (1H, sixtet, J = 7 Hz), 2.43 (3H, s), 2.95 (2H, t, J = 7.5 Hz), 5.75 (1H, s), 6.33 (2H, m), 7.30 (2H, d, J = 8.0 Hz), 7.85 (2H, d, J = 8.4 Hz), 10.30 (1H, br).
元素分析値: C19H22N404S3として計算値: C, 48.91; H, 4.75; , 12.01 (%) 、 実 測値: C, 49.02; H, 4.51; N, 12.34 (%) . Elemental analysis: Calculated as C 19 H 22 N 4 0 4 S 3: C, 48.91; H, 4.75;, 12.01 (%), actual Hakachi: C, 49.02; H, 4.51 ; N, 12.34 (%) .
融点. 197.5-204.0 Melting point. 197.5-204.0
実施例 18-75 Example 18-75
N-ェチル -8- (ィソぺンチルスルファニル) -1-{[ (4-メチルフエニル)スルホニル] ィミノ } - 3_ォキソ -2, 3_ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 5-カルポキ サミド 実施例 18-73で得た化合物(248 mg, 0.530蘭 ol)、 H0BT (86 mg, 0.636 mmol) および WSC (122 mg, 0.636 mmol)の THF (5.0 ml)溶液に 70%ジェチルァミン水溶 液(0.052 ml, 0.636 mmol)を室温で加えた。 混合物を室温で 24時間攪拌した。 反 応液を酢酸ェチルで希釈し、 2%炭酸水素ナトリウム水溶液、 希塩酸、 水および飽 和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣をジェチルエーテ ルから結晶化して標題化合物 (182 mg, 0.368匪 ol)を赤色粉末として得た。 N-ethyl-8- (isopentylsulfanyl) -1-{[(4-methylphenyl) sulfonyl] imino} -3_oxo-2,3_dihydro-1H-imidazo [5,1-c] [ 1, 4] thiazine-5-carboxamide A 70% aqueous solution of getylamine (0.052) was added to a solution of the compound obtained in Example 18-73 (248 mg, 0.530 ol), H0BT (86 mg, 0.636 mmol) and WSC (122 mg, 0.636 mmol) in THF (5.0 ml). ml, 0.636 mmol) at room temperature. The mixture was stirred at room temperature for 24 hours. The reaction solution was diluted with ethyl acetate, washed with a 2% aqueous sodium hydrogen carbonate solution, diluted hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was crystallized from getyl ether to give the title compound (182 mg, 0.368 ol) as a red powder.
Ή-NMR (200MHz, CDC13) : δ 0.92 (6Η, d, J= 6.6 Hz), 1.18 (3H, t, J= 7.2 Hz), 1.53 (2H, brq, J= 7.2 Hz), 1.67 (1H, sixtet, J= 6.6 Hz), 2.43 (3H, s), 2.95 (2H, brt, J= 7.7 Hz), 3.35 (2H, brquint, J= 7.2 Hz), 5.56 (1H, s), 6.57 (1H, brt, J= 4.8 Hz), 7.30 (2H, d, J= 8.0 Hz), 7.84 (2H, d, J= 8.0 Hz), 9.70 (1H, brs) . Ή-NMR (200MHz, CDC1 3 ): δ 0.92 (6Η, d, J = 6.6 Hz), 1.18 (3H, t, J = 7.2 Hz), 1.53 (2H, brq, J = 7.2 Hz), 1.67 (1H , sixtet, J = 6.6 Hz), 2.43 (3H, s), 2.95 (2H, brt, J = 7.7 Hz), 3.35 (2H, brquint, J = 7.2 Hz), 5.56 (1H, s), 6.57 (1H , brt, J = 4.8 Hz), 7.30 (2H, d, J = 8.0 Hz), 7.84 (2H, d, J = 8.0 Hz), 9.70 (1H, brs).
元素分析値: C21H26N404S3として計算値: C, 50.99; H, 5.30; N, 11.33; S, 19.45Elemental analysis: Calculated for C 21 H 26 N 4 0 4 S 3 : C, 50.99; H, 5.30; N, 11.33; S, 19.45
(%) 、 実測値: C, 50.90; H, 5.52; N, 11.12; S, 19.24 ( ) . ' 融点. 222.5- 225.0°C (%), Found: C, 50.90; H, 5.52; N, 11.12; S, 19.24 (). 'Melting point. 222.5-225.0 ° C
実施例 18-76 Example 18-76
N - [5- (ヒドロキシメチル) -8- (ィソペンチルスルファニル) -6-メチル -3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデン ] -4-メチルベンゼ. ンスルホンアミド N- [5- (Hydroxymethyl) -8- (isopentylsulfanyl) -6-methyl-3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 1-ylidene] -4-methylbenzensulfonamide
実施例 4-462で得た化合物(300 mg, 0.623腿 ol)および塩化チォニル (0.052 ml, 0.685 匪 oi)のクロ口ホルム(5 ml)溶液にピリジン(0.060 ml, 0.747讓 ol) のク ロロホルム(1 ml)溶液を 0 で 3分間かけて滴下し、 混合物を 3時間攪拌した。 この間反応液に塩化チォニル(0.019 ml, 0.249匪 ol)およびピリジン(0.020 ml, 0.249腿 ol)を加えた。 反応液を 1時間かけて室温まで戻し、 30分間攪拌した。 反応液に酢酸ェチル(38 ml)およびへキサン(12 ml)で希釈し、 2%炭酸水素ナトリ ゥム、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮して実施例 4-462で得た化合物の酸塩化物 (233 mg, 0.466腿 ol)を濃赤色固形物として得た。 Ή-NMR (200MHz, CDC13) : δ 0.93 (6Η, d, J= 6.6 Hz), 1.56 (2H, brq, J= 7.1 Hz), 1.68 (1H, quint, J= 6.6 Hz), 2.00 (3H? s), 2.43 (3H, s) , 2.98 (2H, brt, J= 7.7 Hz), 7.31 (2H, d, J= 8.0 Hz), 7.45 (1H, brt, J- 5.9 Hz), 7.86 (2H, d, J= 8.4 Hz), 8.68 (1H, br). To a solution of the compound obtained in Example 4-462 (300 mg, 0.623 t ol) and thionyl chloride (0.052 ml, 0.685 dandelion oi) in chloroform (5 ml) was added chloroform (0.060 ml, 0.747 rol) in chloroform. (1 ml) solution was added dropwise at 0 over 3 minutes and the mixture was stirred for 3 hours. During this time, thionyl chloride (0.019 ml, 0.249 ol) and pyridine (0.020 ml, 0.249 ol) were added to the reaction solution. The reaction solution was returned to room temperature over 1 hour and stirred for 30 minutes. The reaction mixture was diluted with ethyl acetate (38 ml) and hexane (12 ml), washed with 2% sodium hydrogen carbonate, water and saturated saline, dried over sodium sulfate, and concentrated. The acid chloride of the compound obtained in 462 (233 mg, 0.466 t) was obtained as a dark red solid. Ή-NMR (200MHz, CDC1 3 ): δ 0.93 (6Η, d, J = 6.6 Hz), 1.56 (2H, brq, J = 7.1 Hz), 1.68 (1H, quint, J = 6.6 Hz), 2.00 (3H ? s), 2.43 (3H, s), 2.98 (2H, brt, J = 7.7 Hz), 7.31 (2H, d, J = 8.0 Hz), 7.45 (1H, brt, J- 5.9 Hz), 7.86 (2H , d, J = 8.4 Hz), 8.68 (1H, br).
この濃赤色固形物(230 mg, 0.460 mmol)の THF (5 ml)溶液に水素化ホウ素ナト リウム(19.3mg, 0.460删 ol)を室温で加え、 混合物を 1時間攪拌した。 反応液に 0.1 塩酸 (10 ml)を加え、 酢酸ェチルで抽出した。 有機層を 2%炭酸水素ナトリ ゥム水溶液、水および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥し、濃縮した。 残渣固形物をジェチルエーテルから結晶化して標題化合物 (32 mg, 0.068匪 ol) を赤色粉末として得た。 To a solution of this dark red solid (230 mg, 0.460 mmol) in THF (5 ml) was added sodium borohydride (19.3 mg, 0.460 mmol) at room temperature, and the mixture was stirred for 1 hour. 0.1 hydrochloric acid (10 ml) was added to the reaction solution, and the mixture was extracted with ethyl acetate. The organic layer was washed with a 2% aqueous sodium hydrogen carbonate solution, water and saturated saline, dried over sodium sulfate, and concentrated. The residual solid was crystallized from getyl ether to give the title compound (32 mg, 0.068 ol) as a red powder.
一顧 R (200MHz, CDC13) : δ 0.93 (6Η, d, J= 6.2 Hz), 1.56 (2H, brq, J= 7.3 Hz), 1.69 (m, quint, J= 6.4 Hz), 1.91 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J= 7.7 Hz), 3.97 (1H, m), 4.43 (2H, brd, J= 5.8 Hz), 7.29 (2H, d, J= 8.4 Hz), 7.85 (2H, d, J= 8.4 Hz), 9.86 (1H, br) . Dang R (200MHz, CDC1 3): δ 0.93 (6Η, d, J = 6.2 Hz), 1.56 (2H, brq, J = 7.3 Hz), 1.69 (m, quint, J = 6.4 Hz), 1.91 (3H, s), 2.42 (3H, s), 2.95 (2H, t, J = 7.7 Hz), 3.97 (1H, m), 4.43 (2H, brd, J = 5.8 Hz), 7.29 (2H, d, J = 8.4) Hz), 7.85 (2H, d, J = 8.4 Hz), 9.86 (1H, br).
元素分析値: C2。H25N304S3 · 1/3H20として計算値: C, 50.72;' H, 5.46; N, 8.87; S, 20.31 (%) 、 実測値: C, 50.73; H, 5.62; N, 8.77; S, 20.08 (%) . Elemental analysis: C 2. H 25 N 3 0 4 S 3 · 1 / 3H 2 0 Calculated: C, 50.72; 'H, 5.46; N, 8.87; S, 20.31 (%), Found: C, 50.73; H, 5.62 ; N , 8.77; S, 20.08 (%).
融点. 184- 188。C Melting point. 184-188. C
実施例 18 - 77 Examples 18-77
N -ェチル- 8 - (ィソぺンチルスルファニル) -6 -メチル -1-{ [ (4 -メチルフェニル)ス ルホニル]ィミノ } - 3-ォキソ _2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1,4]チアジン -5 -力ルポキサミド N-Ethyl-8- (isopentylsulfanyl) -6-methyl-1-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydric mouth-1H-imidazo [5 , 1-c] [1,4] thiazine-5-potrolupoxamide
実施例 4-462で得た化合物(585 mg;)、 H0BT (164 mg, 1.22匪 ol)および WSC (233 mg, 1.22腿 ol)の THF (11.5 ml)溶液に 70%ジェチルァミン水溶液(0.099 ml, 1.22 匪 ol)を室温で加えた。 混合物を室温で 2 時間攪拌した。 反応液を実施例 18 - 75 と同様に処理し、 ジェチルエーテルから結晶化して標題化合物 (29 mg, 0.057 mmol)を赤色粉末として得た。 A 70% aqueous getylamine solution (0.099 ml, 70%) was added to a solution of the compound obtained in Example 4-462 (585 mg;), H0BT (164 mg, 1.22 ol) and WSC (233 mg, 1.22 t) in THF (11.5 ml). 1.22 Maraudal ol) was added at room temperature. The mixture was stirred at room temperature for 2 hours. The reaction solution was treated in the same manner as in Examples 18-75, and crystallized from getyl ether to give the title compound (29 mg, 0.057 mmol) as a red powder.
¾一 NMR (200MHz, CDC13) : δ 0.93 (6Η, d, J= 6.6 Hz), 1.23 (3H, t, J= 7.1 Hz), 1.56 (2H, brq, J= 6.2 Hz), 1.65 (1H, septet, J= 6.6 Hz), 1.89 (3H, s), 2.43 (3H, s), 2.96 (2H, brt, J= 7.7 Hz), 3.43 (2H, dq, J= 5.8, 7.2 Hz), 5.99 (1H, brt, J= 5.8 Hz), 7.29 (2H, d, J= 8.2 Hz), 7.82 (2H, d, J= 8.2 Hz), 9.71 (1H, brs) . ¾ one NMR (200MHz, CDC1 3): δ 0.93 (6Η, d, J = 6.6 Hz), 1.23 (3H, t, J = 7.1 Hz), 1.56 (2H, brq, J = 6.2 Hz), 1.65 (1H , septet, J = 6.6 Hz), 1.89 (3H, s), 2.43 (3H, s), 2.96 (2H, brt, J = 7.7 Hz), 3.43 (2H, dq, J = 5.8, 7.2 Hz), 5.99 (1H, brt, J = 5.8 Hz), 7.29 (2H, d, J = 8.2 Hz), 7.82 (2H, d, J = 8.2 Hz), 9.71 (1H, brs).
融点. 190.0-197.0°C 実施例 18-78 Melting point.190.0-197.0 ° C Example 18-78
N- [5- (1-ヒドロキシプロピル) -8- (ィソペンチルスルファニル) - 6-メチル- 3- ォキソ - 2, 3 -ジヒド口- 1H -ィミダゾ [5, 1-c] [1, ]チアジン- 1-ィリデン] -4-メチル ベンゼンスルホンアミド . (方法 1) 実施例 4-469で得た化合物(1.03 g, 2.08腿 ol)および塩化セリウム 7 水和物(514 mg, 2.08扁 ol)の THF(20 ml)懸濁液に水素化ホウ素ナトリゥム (438 mg, 10.4腿 ol)を室温で加えた。 混合物にメタノ一ル(1.0 ml)の THF (5 ml)溶液を 2.5 時間かけて滴下し、 混合物を 1.5時間攪拌した。 反応液に 1 N塩酸 (13.4ml)お よび水(30 ml)を加え、 酢酸ェチルで抽出した。 有機層を 0.1N塩酸、 水および飽 和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮して得た残渣をジェチルエー テルから結晶化して標題化合物 (590 mg, 1.19 mmol)を赤橙色粉末として得た。 ¾一 NMR (200MHz, CDC13) : δ 0.93 (6Η, d, J= 6.2 Hz), 0.95 (3H, t, J= 7 Hz), 1.55 (2H, brq, J= 6.4 Hz), 1.65 (1H, m), 1.82 (2H, m), 1.95 (3H, s), 2.43 (3H, s), 2.96 (2H, brt, J= 7.7 Hz), 4.58 (1H, dt, J= 11.8, 7.6 Hz), 5.28 (1H, d, J= 12.2 Hz), 7.30 (2H, d, J= 8.4 Hz), 7.86 (2H, d, J= 8.4 Hz), 9.84 (1H, brs). 元素分析値: C22 9N304S3として計算値: C, 53.31; H, 5.90; N, 8.48 (%) 、 実測 値: C, 53.34; H, 5.91; N, 8.41 (%) . N- [5- (1-hydroxypropyl) -8- (isopentylsulfanyl) -6-methyl-3-oxo-2,3-dihydrido-1H-imidazo [5,1-c] [1,] Thiazine-1-ylidene] -4-methylbenzenesulfonamide. (Method 1) The compound obtained in Example 4-469 (1.03 g, 2.08 pt) and cerium chloride heptahydrate (514 mg, 2.08 pt) To a suspension of THF (20 ml) was added sodium borohydride (438 mg, 10.4 mol) at room temperature. To the mixture was added dropwise a solution of methanol (1.0 ml) in THF (5 ml) over 2.5 hours, and the mixture was stirred for 1.5 hours. 1N Hydrochloric acid (13.4 ml) and water (30 ml) were added to the reaction solution, and the mixture was extracted with ethyl acetate. The organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate and concentrated.The residue obtained was crystallized from getyl ether to give the title compound (590 mg, 1.19 mmol) as a red-orange powder. Was. ¾ one NMR (200MHz, CDC1 3): δ 0.93 (6Η, d, J = 6.2 Hz), 0.95 (3H, t, J = 7 Hz), 1.55 (2H, brq, J = 6.4 Hz), 1.65 (1H , m), 1.82 (2H, m), 1.95 (3H, s), 2.43 (3H, s), 2.96 (2H, brt, J = 7.7 Hz), 4.58 (1H, dt, J = 11.8, 7.6 Hz) , 5.28 (1H, d, J = 12.2 Hz), 7.30 (2H, d, J = 8.4 Hz), 7.86 (2H, d, J = 8.4 Hz), 9.84 (1H, brs) elemental analysis:. C 22 9 N 3 0 4 S 3 calculated: C, 53.31; H, 5.90 ; N, 8.48 (%), Found: C, 53.34; H, 5.91 ; N, 8.41 (%).
mp. 170.0-171.5 : mp. 170.0-171.5:
(方法 2) 実施例 4-469で得た化合物(216 mg, 0.438腿 ol)の THF (4.3 ml)溶液に 水素化ホウ素ナトリゥム(19.3 mg, 0. 60 mmol)を室温で加え、 混合物を 4時間攪 拌した。 反応液にメタノール(0.19 ml)の THF (1.0 ml)溶液を滴下し、 さらに水素 化ホウ素ナトリゥム(10 mg, 0.24 mmol)を 1時間毎に 3回室温で加え、 混合物を 15時間攪拌した。 反応液に 1N塩酸 (10ml)を加え、 酢酸ェチルで抽出した。 有 機層を 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃 縮した。残渣をシリカゲル(10 g)のクロマトグラフィー(酢酸ェチルーへキサン、 2:8-4:6) に付し、 酢酸ェチルーへキサン(3 :7)の画分をジェチルェ一テルから結 晶化して標題化合物 (20 mg, 0.040腿 ol)を赤橙色粉末として得た。 (Method 2) To a solution of the compound obtained in Example 4-469 (216 mg, 0.438 mol) in THF (4.3 ml) was added sodium borohydride (19.3 mg, 0.60 mmol) at room temperature. Stirred for hours. A solution of methanol (0.19 ml) in THF (1.0 ml) was added dropwise to the reaction solution, and sodium borohydride (10 mg, 0.24 mmol) was added three times every hour at room temperature, and the mixture was stirred for 15 hours. 1N Hydrochloric acid (10 ml) was added to the reaction solution, and extracted with ethyl acetate. The organic layer was washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was chromatographed on silica gel (10 g) (ethyl acetate-hexane, 2: 8-4: 6), and the ethyl acetate-hexane (3: 7) fraction was crystallized from ethyl acetate to give the title. The compound (20 mg, 0.040 t) was obtained as a red-orange powder.
実施例 18 - 79 Examples 18-79
N- [8- (ィソペンチルスルファニル)—6 -メチル- 3-ォキソ+プロピル- 2, 3-ジヒド 口- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィリデン ]-4-メチルベンゼンスルホン アミ ド N- [8- (Isopentylsulfanyl) -6-methyl-3-oxo + propyl-2,3-dihydride Mouth-1H-imidazo [5, 1-c] [1,4] thiazine-triylidene] -4-methylbenzenesulfon amide
実施例 18-78で得た化合物 (146 mg, 0.295 nimol)のクロ口ホルム(1.0 ml)の溶 液に室温で攪拌しながら TFA (0.5 ml)を加え、 ついでトリェチルシラン(42 nig, 0.354匪 ol)を加えて反応液を室温で 15時間攪拌した。 反応液にトルエン(10 ml) 加えて濃縮後、 酢酸ェチルで希釈し、 水および飽和食塩水で洗浄後、 硫酸ナトリ ゥムで乾燥し、 濃縮した。 残渣をシリカゲル (5 g)のクロマトグラフィー (酢酸ェ チルーへキサン、 2:8-5:5) で精製し、 酢酸ェチルーへキサン(2 :8)の赤色画分を ジェチルェ一テルから結晶化して標題化合物 (3.8 mg, 0.008讓 ol)を濃赤色粉末 として得た。 To a solution of the compound obtained in Examples 18-78 (146 mg, 0.295 nimol) in chloroform (1.0 ml) was added TFA (0.5 ml) with stirring at room temperature, and then triethylsilane (42 nig, 0.354 ol). ) Was added and the reaction solution was stirred at room temperature for 15 hours. The reaction solution was added with toluene (10 ml), concentrated, diluted with ethyl acetate, washed with water and saturated saline, dried over sodium sulfate, and concentrated. The residue was purified by chromatography on silica gel (5 g) (ethyl acetate-hexane, 2: 8-5: 5), and the red fraction of ethyl acetate-hexane (2: 8) was crystallized from getyl ether. The title compound (3.8 mg, 0.008 alcohol) was obtained as a dark red powder.
ΐ-NMR (200MHz, CDC13) : <5 0.93 (6H, d, J= 6.2 Hz), 0.94 (3H, t, J= 7.0 Hz), 1.48 (2H, q, J= 7.7 Hz), 1.60 (1H, m), 1.70 (2H, m), 1.82 (3H, s), 2.42 (3H, s), 2.74 (2H, brt, J= 7.7 Hz), 2.94 (2H, t, J= 7.7 Hz), 7.28 (2H, d, J= 8.0 Hz), 7.86 (2H, d, J= 8.0 Hz), 9.60 (1H, brs). ΐ-NMR (200MHz, CDC1 3 ): <5 0.93 (6H, d, J = 6.2 Hz), 0.94 (3H, t, J = 7.0 Hz), 1.48 (2H, q, J = 7.7 Hz), 1.60 ( 1H, m), 1.70 (2H, m), 1.82 (3H, s), 2.42 (3H, s), 2.74 (2H, brt, J = 7.7 Hz), 2.94 (2H, t, J = 7.7 Hz), 7.28 (2H, d, J = 8.0 Hz), 7.86 (2H, d, J = 8.0 Hz), 9.60 (1H, brs).
実施例 18-80 Example 18-80
0-[l- (8- (イソペンチルスルファ二ル)- 6-メチル -l-{ [(4-メチルフエニル)スルホ ニル]イミノ}-3 -ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン- 5 -ィ ル) プロピル] 1H-イミダゾール- 1-カルポチォェート 0- [l- (8- (Isopentylsulfanyl) -6-methyl-l-{[(4-methylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [ 5,1-c] [1,4] Thiazine-5-yl) propyl] 1H-imidazole-1-carpothioate
実施例 18-78で得た化合物(228 mg, 0.460 mmol)および 1, Γ -チォカルポニル ジイミダゾ一ル(109 mg, 0.552腿 ol)のトルエン(10 ml)の溶液を室温で 3時間攪 拌した。反応液を酢酸ェチルで希釈し、 0.1N塩酸、 水、 ¾炭酸水素ナトリウムお よび飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮した。 残渣をシリカゲ ル(10 g)のクロマトグラフィー (酢酸ェチルーへキサン、 1:9-5:5) で精製し、 酢 酸ェチル—へキサン(4: 6)の赤色画分をジェチルェ一テルから結晶化して標題化 合物 (28 mg, 0.046 mmol)を赤橙色粉末として得た。 A solution of the compound obtained in Examples 18-78 (228 mg, 0.460 mmol) and 1, Γ-thiocarbonyl diimidazole (109 mg, 0.552 mol) in toluene (10 ml) was stirred at room temperature for 3 hours. The reaction solution was diluted with ethyl acetate, washed with 0.1N hydrochloric acid, water, sodium bicarbonate, and saturated saline, dried over sodium sulfate, and concentrated. The residue was purified by chromatography on silica gel (10 g) (ethyl acetate-hexane, 1: 9-5: 5), and the red fraction of ethyl acetate-hexane (4: 6) was crystallized from getyl ether. To give the title compound (28 mg, 0.046 mmol) as a red-orange powder.
賺 (200MHz, CDC13) : δ 0.93 (6H, d, J= 6.2 Hz), 1.07 (3H, t, J= 7.3 Hz), 1.55 (2H, brq, J- 7.4 Hz), 1.61 (1H, m)> 2.08 (2H, s), 2.08 (2H, m), 2.42 (3H, s), 2.95 (2H, t, J= 7.7 Hz), 5.17 (1H, m), 7.10 (1H, brs), 7.29 (2H, d, J= 8.4 Hz), 7.49 (1H, brs), 7.86 (2H, d, J= 8.4 Hz), 8.23 (1H, brs), 9.83 (1H, brs) 賺(200MHz, CDC1 3): δ 0.93 (6H, d, J = 6.2 Hz), 1.07 (3H, t, J = 7.3 Hz), 1.55 (2H, brq, J- 7.4 Hz), 1.61 (1H, m )> 2.08 (2H, s), 2.08 (2H, m), 2.42 (3H, s), 2.95 (2H, t, J = 7.7 Hz), 5.17 (1H, m), 7.10 (1H, brs), 7.29 (2H, d, J = 8.4 Hz), 7.49 (1H, brs), 7.86 (2H, d, J = 8.4 Hz), 8.23 (1H, brs), 9.83 (1H, brs)
元素分析値: C26H31N504S4 · 0.3EtOAcとして計算値: C, 51.67; H, 5.32; N, 11.07; S, 20. 8 (%)、 実測値: C, 52.04; H, 5.33; N, 11.11; S, 20.21 (%) · mp. 171.0-172.0°C Elemental analysis: C 26 H 31 N 5 0 4 S 4 · 0.3EtOAc Calculated: C, 51.67; H, 5.32 ; N, 11.07; S, 20. 8 (%), Found: C, 52.04; H , 5.33; N, 11.11; S, 20.21 (%) mp.171.0-172.0 ° C
実施例 19 Example 19
5 - [(E)-力ルポキシメチリデン] -卜 {[(4-メチルフエニル)スルホニル]ィミノ卜 3 - ォキソ - 8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニル] - 2, 3, 5, 6 -テトラ ヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 6-カルボン酸 5-[(E) -Holepoxymethylidene] -buta [[(4-methylphenyl) sulfonyl] iminoto 3-oxo- 8-[(5,5,6,6,6-penoxyfluorhexyl ) Sulfanyl] -2,3,5,6-tetrahydro-1H-imidazo [5, tric] [1,4] thiazine-6-carboxylic acid
実施例 3-40で得られた粗製の化合物(0.74 g, 1.28誦 ol)および炭酸力リゥム (0.30 g, 2.17顧 ol) の DMF (10 ml) の懸濁液にヨウ化 5, 5, 6, 6, 6-ペン夕フル ォ口へキシル(0.44 g, 1.44mmol)を 60°Cで滴下し、 1.5時間攪拌した。 反応液に 1N塩酸 (3.2ml)を加え、 酢酸ェチル(50 ml)で抽出した。 得られた有機層を酸性 に調整した 7%および 15%の食塩水、 ついで飽和食塩水で洗浄後、 硫酸ナトリゥム で乾燥、 濃縮乾固した。 残渣をシリカゲル(10 g)のクロマトグラフィー (酢酸ェ チルーへキサン、 3:7) に付し、 橙色画分を濃縮乾固して、 赤色油状物(0.68 g) を得た。 これをメタノール(8ml)および THF (3 ml)に溶解し、 室温で攪拌しなが ら 1N水酸化ナトリウム (8ml) を加え、 反応液を 30分間攪拌した。 反応液に 1N 塩酸 (10 ml)を加え、 酢酸ェチル (40 ml)で抽出、 精製し、 粗製生物 (375 mg)を得 た。 これをメタノール(8ml)および 1N水酸化ナトリウム (8ml)に溶解し、 室温で 3時間攪拌した。 反応液に 1N塩酸 (9 ml)を加え、 30分間攪拌した。 析出物を濾 取し、 メタノール-水(1:1)で洗浄後、 乾燥して表題化合物(311 mg, 0.494 mmol, 38.6%)を灰橙色粉末として得た。 5,5,6 Iodide was added to a suspension of the crude compound obtained in Examples 3-40 (0.74 g, 1.28 ol) and carbonated lime (0.30 g, 2.17 dl) in DMF (10 ml). Hexyl (0.44 g, 1.44 mmol) was added dropwise at 60 ° C. and stirred for 1.5 hours. 1N Hydrochloric acid (3.2 ml) was added to the reaction solution, and the mixture was extracted with ethyl acetate (50 ml). The obtained organic layer was washed with a 7% and 15% saline solution adjusted to be acidic, then with a saturated saline solution, dried over sodium sulfate, and concentrated to dryness. The residue was chromatographed on silica gel (10 g) (ethyl acetate-hexane, 3: 7), and the orange fraction was concentrated to dryness to give a red oil (0.68 g). This was dissolved in methanol (8 ml) and THF (3 ml), 1N sodium hydroxide (8 ml) was added while stirring at room temperature, and the reaction solution was stirred for 30 minutes. 1N Hydrochloric acid (10 ml) was added to the reaction solution, extracted with ethyl acetate (40 ml), and purified to obtain a crude organism (375 mg). This was dissolved in methanol (8 ml) and 1N sodium hydroxide (8 ml) and stirred at room temperature for 3 hours. 1N Hydrochloric acid (9 ml) was added to the reaction solution, and the mixture was stirred for 30 minutes. The precipitate was collected by filtration, washed with methanol-water (1: 1), and dried to give the title compound (311 mg, 0.494 mmol, 38.6%) as a grey-orange powder.
'H-NMR (200MHz, CDC1, - DMS0 - d6= 2:1, endo/exo 異性体混合物): δ 1.72 (4Η, m), 2.19 (2H, m), 2.36 (3H, s), 2.98/3.06 (2H, t, J= 6.9/7.1 Hz), 6.53 (1H, s), 6.58 (1H, s), 7.33 (2H, d, J= 8.1 Hz), 7.90 (2H, d, J= 8.4 Hz), ca. 12.0 (1H, br). 'H-NMR (200MHz, CDC1,-DMS0-d 6 = 2: 1, mixture of endo / exo isomers): δ 1.72 (4Η, m), 2.19 (2H, m), 2.36 (3H, s), 2.98 / 3.06 (2H, t, J = 6.9 / 7.1 Hz), 6.53 (1H, s), 6.58 (1H, s), 7.33 (2H, d, J = 8.1 Hz), 7.90 (2H, d, J = 8.4 Hz), ca.12.0 (1H, br).
元素分析値 C22H20N307S3F5 ¾0 として計算値: C, 40.80; H, 3.42; N, 6.49; S, 14.85; F, 14.67 (%) 、 実測値: C, 40.69; H, 3.20; N, 6.41; S, 14.85; F, 14.76Elemental analysis: calculated as C 22 H 20 N 3 0 7 S 3 F 5計算 0: C, 40.80; H, 3.42; N, 6.49; S, 14.85; F, 14.67 (%), found: C, 40.69; H, 3.20; N, 6.41; S, 14.85; F, 14.76
(%) . (%).
実施例 20 Example 20
N-{5 -ヒドロキシ -5, 6-ジメチル- 3 -ォキソ - 8-[(5, 5, 6, 6, 6-ペン夕フルォ口へキシ ル)スルファニル ]_2, 3, 5, 6-テトラヒドロ- 1H-ィミダゾ [5,卜 c] [1, 4]チアジン - 1_ ィリデン } -4-メチルベンゼンスルホンアミド N- {5-Hydroxy-5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] _2,3,5,6-tetrahydro -1H-imidazo [5, tric] [1, 4] thiazine-1_ ylidene} -4-methylbenzenesulfonamide
実施例 4-35で得られた N - {5, 6 -ジメチル -3-ォキソ -8 - [(5, 5, 6, 6, 6-ペン夕フル ォ口へキシル)スルファニル ]-2, 3 -ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン -卜イリデン }- 4-メチルベンゼンスルホンアミド (600 mg, 1.08 mmol) と濃塩酸 (0.200 ml) のエタノール (10.0 ml) 懸濁液を 100 で 3時間かき混ぜた。 反応 混合物を室温まで冷やした後、 酢酸ェチルで希釈し、 水洗、 乾燥 (MgS04) 後、 溶 媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに付し、 n -へキサン -酢酸ェチル (10:1, v/v) で溶出して題記化合物 (83.3 mg, 13¾) を 暗赤色結晶として得た。 N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3 obtained in Example 4-35 -Dihydro-1H-imidazo [5, tric] [1,4] thiazine-triylidene} -4-Hydrogenbenzenesulfonamide (600 mg, 1.08 mmol) and concentrated hydrochloric acid (0.200 ml) suspended in ethanol (10.0 ml) The suspension was stirred with 100 for 3 hours. After cooling the reaction mixture to room temperature, diluted with acetic acid Echiru, washed with water, dried (MgSO 4), followed by distilling off the solvent medium under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound (83.3 mg, 13¾) as dark red crystals.
IR (KBr) v: 3600-2800, 2944, 1732, 1622, 1557 cm一1. IR (KBr) v: 3600-2800, 2944, 1732, 1622, 1557 cm- 1 .
Ή-N R (CDC13) : 51.42 (3H, d, J=7.0 Hz), 1.71 (3H, s), 1.60-1.86 (4H, m), 1.90-2.20 (2H, m), 2.42 (3H, s), 3.01 (2H, t, J=7.0 Hz), 3.42 (2H, q, J=7.0 Hz), 5.50 (1H, s), 7.30 (2H, d, J=8.4 Hz), 7.87 (2H, d, J=8.4 Hz), 9.61 (1H, bs). 元素分析値: C21H24N304S3F5として Ή-NR (CDC1 3): 51.42 (3H, d, J = 7.0 Hz), 1.71 (3H, s), 1.60-1.86 (4H, m), 1.90-2.20 (2H, m), 2.42 (3H, s ), 3.01 (2H, t, J = 7.0 Hz), 3.42 (2H, q, J = 7.0 Hz), 5.50 (1H, s), 7.30 (2H, d, J = 8.4 Hz), 7.87 (2H, d , J = 8.4 Hz), 9.61 (1H, bs). Elementary analysis: as C 21 H 24 N 3 0 4 S 3 F 5
計算値 (¾) : C, 43.97; H, 4.22; N, 7.33; S, 16.77. Calculated value (¾): C, 43.97; H, 4.22; N, 7.33; S, 16.77.
実測値 (¾) : C, 44.00; H, 4.34; N, 7.47; S, 16.72. Found (¾): C, 44.00; H, 4.34; N, 7.47; S, 16.72.
実施例 21-1 Example 21-1
3- ({[5, 6-ジメチル -8- (イソペンチルスルファ二ル)- 3-ォキソ -2, 3-ジヒドロ- 1H - ィミダゾ [5, 1 - c] [1, 4]チアジン- 1 -イリデン]アミノ }スルホ二ル)- N-へキシルベ ンズアミド 3-({[5,6-Dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1 -Ylidene] amino} sulfonyl) -N-hexylbenzamide
実施例 7-80で得られた 3 -({[5, 6-ジメチル- 8- (ィソぺンチルスルファニル) -3- ォキソ - 2, 3 -ジヒド口 -1H -ィミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデン]アミノ}ス ルホニル)安息香酸 (150mg, 0.311顧 ol) と卜へキシルァミン (0.0454ml, 0, 343 mmol) の Ν,Ν ジメチルホルムアミド (6.00 ml) 溶液に、 シァノりん酸ジェチル (0.554 ml, 0.343 mmol), 続いてトリェチルァミン (0.0478 ml, 0.343 mmol) を加えた。反応混合物を室温で 19.5時間かき混ぜた後、水を加えて酢酸ェチルで 抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (18. lmg, 10%) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 181- 183°Cの暗赤色結晶を得た。 The 3-({[5,6-dimethyl-8- (isopentylsulfanyl) -3-oxo-2,3-dihydrido-1H-imidazo [5,1--) obtained in Example 7-80 c] 1,, Ν-dimethylformamide (6.00 ml) of [1,4] thiazine-1-ylidene] amino} sulfonyl) benzoic acid (150 mg, 0.311 mol) and trihexylamine (0.0454 ml, 0, 343 mmol) To the solution was added getyl cyanophosphate (0.554 ml, 0.343 mmol), followed by triethylamine (0.0478 ml, 0.343 mmol). After stirring the reaction mixture at room temperature for 19.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), and the solvent was evaporated under reduced pressure to synthesize the title compound (18. lmg, 10%). Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 181-183 ° C.
IR (KBr) V: 3272, 2955, 2930, 2868, 1732, 1715, 1661, 1615, 1605, 1545 cm—1. Ή-NMR (CDC13) : (5 ,0.844 (3H, t, J=7.2Hz), 0.993 (6H, d, J=6.6Hz), 1.14-1.34 (6H, m), 1.38-1.52 (2H, in), 1.68-1.90 (3H, m), 1.85 (3H, s), 2.37 (3H, s), 2.95 (2H, q. J=7.2 Hz), 3.09 (2H, t, J=8.4 Hz), 4.34 (1H, bs), 7.60 (1H, t, J=8.1 Hz), 7.99 (1H, dt, J=7.5, 3.3 Hz), 8.47 (1H, dt, J=6.3, 0.6 Hz), 8.93 (1H, t, J=1.5 Hz), 11.07· (1H, bs). IR (KBr) V:. 3272 , 2955, 2930, 2868, 1732, 1715, 1661, 1615, 1605, 1545 cm- 1 Ή-NMR (CDC1 3): (5, 0.844 (3H, t, J = 7.2Hz ), 0.993 (6H, d, J = 6.6Hz), 1.14-1.34 (6H, m), 1.38-1.52 (2H, in), 1.68-1.90 (3H, m), 1.85 (3H, s), 2.37 ( 3H, s), 2.95 (2H, q.J = 7.2 Hz), 3.09 (2H, t, J = 8.4 Hz), 4.34 (1H, bs), 7.60 (1H, t, J = 8.1 Hz), 7.99 ( 1H, dt, J = 7.5, 3.3 Hz), 8.47 (1H, dt, J = 6.3, 0.6 Hz), 8.93 (1H, t, J = 1.5 Hz), 11.07 · (1H, bs).
元素分析値: C26H36N404S3として Elemental analysis: as C 26 H 36 N 4 0 4 S 3
計算値 (¾) : C, 55.29; H, 6.42; N, 9.92; S, 17.06. Calculated value (¾): C, 55.29; H, 6.42; N, 9.92; S, 17.06.
実測値 (%) : C, 55.15; H, 6.19; N, 9.76; S, 16.99. Found (%): C, 55.15; H, 6.19; N, 9.76; S, 16.99.
以下に実施例 21-1 で合成した化合物および同様にして合成した化合物の一覧 を示す。 〔表 1 1 3〕The following is a list of compounds synthesized in Example 21-1 and compounds synthesized in the same manner. (Table 1 1 3)
差替 用紙(規則 》 〔表 1 14〕Replacement form (rules) (Table 1 14)
螯簪ぇ用紙 OT1 6' 〔表 1 1 5〕Bun hairstyle paper OT 1 6 ' (Table 1 15)
# IS飆 («26) 以下に実施例 21_2〜21-25で合成した化合物のデ一夕を示す。 # IS Britain («26) The following shows the results of the compounds synthesized in Examples 21_2 to 21-25.
実施例 21-2 Example 21-2
4-[({5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファ ニル]一 2, 3 -ジヒドロ- 1H-ィミダゾ [5, 1-c] [1, ]チアジン-トイリデン }ァミノ)ス ルホニル]ベンズアミド 4-[({5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -l, 3-dihydro-1H-imidazo [ 5, 1-c] [1,] thiazine-tolylidene} amino) sulfonyl] benzamide
実施例 7-8 で得られた 4 -シァノ- N - [5, 6_ジメチル- 3 -ォキソ -8-[(5, 5, 6, 6, 6- ペンタフルォ口へキシル)スルファエル]- 2, 3-ジヒド口- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン - 1 -ィリデン]ベンゼンスルホンアミド (515 nig, 1.63mmol) を濃硫酸 (1.50 ml) に溶かし、 室温で 5.5時間かき混ぜた。 反応混合物を氷水に 加えて酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を 圧下に 留去して題記化合物 (12.5 mg, 8.1%) を暗赤色結晶として得た。 酢酸ェチル-ジ ェチルエーテルから再結晶して融点 206-208°Cの喑赤色結晶を得た。 4-cyano-N- [5,6_dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfael] -2, obtained in Example 7-8 3-Dihydrid-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene] benzenesulfonamide (515 nig, 1.63 mmol) is dissolved in concentrated sulfuric acid (1.50 ml) and left at room temperature for 5.5 hours. Stirred. The reaction mixture was added to ice water and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after, to obtain a solvent the title compound was evaporated under pressure (12.5 mg, 8.1%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-diethyl ether to obtain reddish crystals having a melting point of 206-208 ° C.
IR (KBr) V : 3428, 3160, 1736, 1682, 1665, 1613, 1557 cm"1. IR (KBr) V: 3428, 3160, 1736, 1682, 1665, 1613, 1557 cm " 1 .
NMR (CDC13) : δ 1.68-1.84 (4Η, m), 1.80 (3H, s), 1.92-2.22 (2H, m), 2.28 (3H, s), 2.98 (2H, t, J=7.0 Hz), 7.91 (2H, d, J=8.4 Hz), 8.05 (2H, d, J-8. Hz), 9.65 (1H, bs). NMR (CDC1 3): δ 1.68-1.84 (4Η, m), 1.80 (3H, s), 1.92-2.22 (2H, m), 2.28 (3H, s), 2.98 (2H, t, J = 7.0 Hz) , 7.91 (2H, d, J = 8.4 Hz), 8.05 (2H, d, J-8. Hz), 9.65 (1H, bs).
実施例 21-3 Example 21-3
4-ァミノ- N - {5, 6 -ジメチル- 3-ォキソ -8- [ (5, 5, 6, 6, 6-ペンタフルォ口へキシル) スルファニル ]-2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン }ベ ンゼンスルホンアミド 4-amino-N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5 , 1-c] [1,4] thiazine-1-ylidene} benzenesulfonamide
(方法 1)実施例 7- 86で得た化合物 (219mg, 0.329mmol)のジォキサン一水(9:1, 7 ml)懸濁液をトシル酸水和物(75 mg, 0.39511111101)存在下に5.5 時間加熱還流し た。 酢酸ェチルで抽出して水洗、 乾燥、 濃縮後、 ジェチルエーテルで希釈した。 析出物を濾取し、ジェチルエーテルで洗浄後、乾燥して表題化合物 (37mg, 0.066 醒 ol)を赤色粉末として得た。 (Method 1) A suspension of the compound (219 mg, 0.329 mmol) obtained in Example 7-86 in dioxane / aqueous solution (9: 1, 7 ml) was added in the presence of tosylate hydrate (75 mg, 0.39511111101) for 5.5 min. The mixture was heated under reflux for an hour. After extraction with ethyl acetate, washing with water, drying and concentration, the mixture was diluted with getyl ether. The precipitate was collected by filtration, washed with getyl ether, and dried to give the title compound (37 mg, 0.066 ol) as a red powder.
—匪 R (200MHz, CDCI3): δΐ.75 (4Η, m), 1.78 (3H, brs), 2.04 (2H, m), 2.26 (3H, brs), 2.97 (2H, q, J= 8.2 Hz), 4.10 (2H, brs), 6.66 (2H, d, J= 8.8 Hz), 7.72 (2H, d, J= 8.8 Hz), 9.60 (1H, br). — Band R (200MHz, CDCI3): δΐ.75 (4Η, m), 1.78 (3H, brs), 2.04 (2H, m), 2.26 (3H, brs), 2.97 (2H, q, J = 8.2 Hz) , 4.10 (2H, brs), 6.66 (2H, d, J = 8.8 Hz), 7.72 (2H, d, J = 8.8 Hz), 9.60 (1H, br).
元素分析値: C2。H21N403S3F5として計算値: C, 43.16; H, 3.80; N, 10.07; S, 17.28 (%) 、 実測値: C, 42.88; H, 3.55; , 9.94; S, 17.44 (%) . mp. 172.5-176.5°C Elemental analysis: C 2. Calculated for H 21 N 4 0 3 S 3 F 5 : C, 43.16; H, 3.80; N, 10.07; S, 17.28 (%), Found: C, 42.88; H, 3.55;, 9.94; S, 17.44 (%) .mp. 172.5-176.5 ° C
(方法 2) 実施例 7-86で得た化合物(3.36 g, 5.61 mmol) と p-トルエンスルホン 酸水和物 (1.28 g, 6.73删01) のジォキサン—水 (9:1, 70 ml) 溶液を 80 で 4.5 時間攪拌した。 反応液を濃縮乾固後、エタノール (80 ml)で希釈して濃縮し、 ァセトニトリル(80 ml)で希釈して約 30mlまで濃縮した。 析出物を濾取し、 ァセ トニトリルで洗浄後、 乾燥して表題化合物の P-トルエンスルホン酸塩 (905 ing, 1.24 mmol, 22.1 )を赤褐色の粉末として得た。 母液を濃縮乾固後、 トルエン(60 ml)に溶解し、 80DC で 1 時間攪拌した。 反応液を約 30mlまで濃縮してァセトニ トリル (20 ml)で希釈し、 析出物を濾取してァセトニトリルで洗浄後、 乾燥して表 題化合物の P-トルエンスルホン酸塩 (2.27 g, 3.11 匪 ol, 55.5%)を赤褐色の粉 末として得た。 (Method 2) A solution of the compound obtained in Example 7-86 (3.36 g, 5.61 mmol) and p-toluenesulfonic acid hydrate (1.28 g, 6.73 删 01) in dioxane-water (9: 1, 70 ml) Was stirred at 80 for 4.5 hours. The reaction mixture was concentrated to dryness, diluted with ethanol (80 ml), concentrated, diluted with acetonitrile (80 ml), and concentrated to about 30 ml. The precipitate was collected by filtration, washed with acetonitrile, and dried to give the title compound P-toluenesulfonate (905 ing, 1.24 mmol, 22.1) as a red-brown powder. After the mother liquor was concentrated to dryness, it was dissolved in toluene (60 ml) and stirred at 80 DC for 1 hour. The reaction solution was concentrated to about 30 ml, diluted with acetonitrile (20 ml), and the precipitate was collected by filtration, washed with acetonitrile, dried, and dried to give the title compound P-toluenesulfonate (2.27 g, 3.11 ol, 55.5%) as a red-brown powder.
実施例 21_4 Example 21_4
N-{4-[({5, 6 -ジメチル- 3 -ォキソ - 8-[(5, 5, 6, 6, 6-ペンタフルォ口へキシル) スル ファニル]- 2, 3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1, 4]チアジン- 1 -ィリデン }アミ ノ)スルホニル]フエ二ル}へキサンアミド N- {4-[({5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene} amino) sulfonyl] phenyl} hexaneamide
実施例 21-3 (方法 1) と同様にして得た 4 -ァミノ- N- {5,6-ジメチル -3-ォキソ -8 - [(5, 5, 6, 6, 6-ペンタフルォ口へキシル) スルファニル ]-2, 3-ジヒドロ- 1H -ィ ミダゾ [5,卜 c] [1,4]チアジン-卜ィリデン }ベンゼンスルホンアミドの粗物質(純 度 60 , 0.60g, 0.62 mmol、水和物との混合物)および塩化へキサノィル (125mg, 0.93腿 ol) の THF (10 ml)溶液に室温で攪拌しながらト リェチルァミン(0.34 ml, 2.5 蘭 ol)を加えた。 反応液を 1時間攪拌後、 酢酸ェチル(30 ml)で希釈し、 0.2N 塩酸、 水および飽和食塩水で洗浄し、 硫酸ナトリウムで乾燥後、 濃縮した。 残渣 をトルエン(10 ml)に溶解し、 無水酢酸(0.18 ml)および P-トルエンスルホン酸水 和物(118 mg, 0.62 mmol) 存在下 80°C で 1 時間攪拌した。 反応液を酢酸ェチル (30 ml)で希釈し、水および飽和食塩水で洗浄後、硫酸ナトリゥムで乾燥、濃縮し、 残渣にメタノールを加えた。 析出物を濾取してメタノールで洗浄後、 乾燥して表 題化合物 (125 mg, 0.191 mmol, 30.9%)を赤褐色の粉末として得た。 Example 21-3 4-amino-N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) obtained in the same manner as in (Method 1) ) Sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-triylidene} Crude benzenesulfonamide (purity 60, 0.60 g, 0.62 mmol, hydrate To a solution of hexanoyl chloride (125 mg, 0.93 t) in THF (10 ml) while stirring at room temperature was added triethylamine (0.34 ml, 2.5 n-ol). The reaction solution was stirred for 1 hour, diluted with ethyl acetate (30 ml), washed with 0.2N hydrochloric acid, water and saturated saline, dried over sodium sulfate, and concentrated. The residue was dissolved in toluene (10 ml) and stirred at 80 ° C for 1 hour in the presence of acetic anhydride (0.18 ml) and P-toluenesulfonic acid hydrate (118 mg, 0.62 mmol). The reaction solution was diluted with ethyl acetate (30 ml), washed with water and saturated saline, dried over sodium sulfate and concentrated, and methanol was added to the residue. The precipitate was collected by filtration, washed with methanol, and dried to give the title compound (125 mg, 0.191 mmol, 30.9%) as a red-brown powder.
Ή一 NMR (300MHz, CDC13): δθ.91 (3Η, brt, J= 6.6 Hz), 1.36 (4H, m), 1.74 (6H, m), 1.79 (3H, brs), 2.06 (2H, in), 2.27 (3H, brs), 2.38 (2H, t, J= 7.5 Hz), 2.96 (2H, t, J= 6.9 Hz), 7.39 (1H, brs), 7.65 (2H, d, J= 8.7 Hz) , 7.89 (2H, d, J= 8.4 Hz), 9.63 (1H, br). Ή one NMR (300MHz, CDC1 3): δθ.91 (3Η, brt, J = 6.6 Hz), 1.36 (4H, m), 1.74 (6H, m), 1.79 (3H, brs), 2.06 (2H, in), 2.27 (3H, brs), 2.38 (2H, t, J = 7.5 Hz), 2.96 (2H, t, J = 6.9 Hz), 7.39 ( 1H, brs), 7.65 (2H, d, J = 8.7 Hz), 7.89 (2H, d, J = 8.4 Hz), 9.63 (1H, br).
元素分析値: C26H31N404S3F5 -0.4H20として計算値: C, 47.18; H, 4.84; N, 8.46; S, 14.53; F, 14.35 (%) 、 実測値: C, 47.20; H, 4.78; , 8.53; S, 14.74; F, 14.54 ( ) . Elemental analysis: C 26 H 31 N 4 0 4 S 3 F 5 -0.4H 2 0 Calculated: C, 47.18; H, 4.84; N, 8.46; S, 14.53; F, 14.35 (%), actual : C, 47.20; H, 4.78;, 8.53; S, 14.74; F, 14.54 ().
mp. 165.5-170.0°C mp. 165.5-170.0 ° C
■ 実施例 21-5 ■ Example 21-5
4 - [ビス (メチルスルホニル) ァミ ノ ]-N-{5, 6 -ジメチル -3-ォキソ -8-[(5, 5, 6, 6, 6 -ペン夕フルォロへキシル) スルファニル ]-2, 3-ジヒドロ- 1H -ィ ミダゾ [5,1 - c] [1, 4]チアジン- 1-イリデン }ベンゼンスルホンアミド 4- [Bis (methylsulfonyl) amino] -N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentanofluorohexyl) sulfanyl] -2 , 3-Dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidene} benzenesulfonamide
実施例 21-3 (方法 1) で得た化合物(175 mg, 0.31 匪 ol)およびメタンスルホ ニルクロリド (43.6 mg, 0.377 mraol)の THF (3.5 ml)溶液に 0°Cで攪拌しながら トリエチルァミン(0.065 ml, 0.471 mniol)を加え、 反応液を 0 で 4.5時間攪拌 した。 同量のメタンスルホニルク口リドおよびトリェチルァミンを加え、 さらに 3時間攪拌した。 反応液を酢酸ェチル (30 ml)で希釈し、 水および飽和食塩水で洗 浄後、 硫酸ナトリウムで乾燥、 濃縮し、 残渣にジェチルエーテルを加えた。 析出 物を濾取してジェチルェ一テルで洗浄後、 乾燥して表題化合物(188 mg, 0.264 mmol, 84.0%)を紫色の粉末として得た。 To a solution of the compound obtained in Example 21-3 (Method 1) (175 mg, 0.31 bandol) and methanesulfonyl chloride (43.6 mg, 0.377 mraol) in THF (3.5 ml) was stirred triethylamine (0 ° C) at 0 ° C. 0.065 ml, 0.471 mniol) was added and the reaction solution was stirred at 0 for 4.5 hours. The same amounts of methanesulfonyl chloride and triethylamine were added, and the mixture was further stirred for 3 hours. The reaction solution was diluted with ethyl acetate (30 ml), washed with water and saturated saline, dried over sodium sulfate and concentrated, and getyl ether was added to the residue. The precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (188 mg, 0.264 mmol, 84.0%) as a purple powder.
LH-NMR (200MHz, CDC13): 1.74 (4H, m), 1.81 (3H, q, J= 0.8 Hz), 2.06 (2H, m), 2.29 (3H, q, J- 0.8 Hz), 3.00 (2H, t, J= 7.0 Hz), 3.41 (6H, s), 7.48 (2H, d, J= 8.8 Hz), 8.06 (2H, d, J= 8.8 Hz), 9.63 (1H, br) . LH-NMR (200MHz, CDC1 3 ): 1.74 (4H, m), 1.81 (3H, q, J = 0.8 Hz), 2.06 (2H, m), 2.29 (3H, q, J- 0.8 Hz), 3.00 ( 2H, t, J = 7.0 Hz), 3.41 (6H, s), 7.48 (2H, d, J = 8.8 Hz), 8.06 (2H, d, J = 8.8 Hz), 9.63 (1H, br).
元素分析値: C22H25N407S5F5として計算値: C, 37.07; H, 3.54; N, 7.87; F, 13.33 (%) 、 実測値: C, 37.38; H, 3.34; N, 8.02; F, 13.62 (%) . Elemental analysis: C 22 H 25 N 4 0 7 S 5 F 5 Calculated: C, 37.07; H, 3.54 ; N, 7.87; F, 13.33 (%), Found: C, 37.38; H, 3.34 ; N, 8.02; F, 13.62 (%).
mp. 180.5-182.0°C mp.180.5-182.0 ° C
実施例 21-6(1) Example 21-6 (1)
-{5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル ]-2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1-ィリデン } - 4 -ホルミル ド、 実施例 21- 6(2) -{5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-1 -ylidene}-4 -formyld, Example 21-6 (2)
N-{5, 6 -ジメチル -3-ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニ ル] - 2, 3-ジヒドロ- 1H -イミダゾ [5, l-c] [1,4]チアジン- 1-ィリデン }- 4- (ヒドロキ シメチル)ベンゼンスルホンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine-1-ylidene} -4- (hydroxymethyl) benzenesulfonamide
実施例 7 - 84 で得られた 4-シァノ -N-{2, 3-ジヒドロ- 5, 6-ジメチル -3-ォキソ - 8 - [(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニル] -1H -ィミダゾ [5, l-c] [1, 4]チアジン- 1-ィリデン }ベンゼンスルホンアミド (251 mg, 0.442 画 1) のジクロロメタン (200 ml) 溶液に、 水素化ジイソブチルアルミニウムの 1.5 mol/1 トルエン溶液 (0.590 ml, 0.884漏 1) を- 70°Cで加えた。 反応混合物 を _70°Cでアルゴン気流下 7時間かき混ぜた。 飽和塩化アンモニゥム水溶液 .(250 ml) を加え、 反応混合物を室温で 30分間かき混ぜた後、 1N塩酸を加えてジクロ ロメタンで抽出した。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去した。 残留物をシリカゲルカラムクロマトグラフィーに付し、 n-へキサン-酢酸ェチル (10:1, v/v) で溶出して題記化合物 21-6(1) (15.7mg, 6.2¾) と 21 - 6(2) (26.0 mg, 10¾) を暗赤色結晶として得た。 4-Cyano-N- {2,3-dihydro-5,6-dimethyl-3-oxo-3-8-[(5,5,6,6,6-pentenfluorene) obtained in Example 7-84 [Hexyl) sulfanyl] -1H-imidazo [5, lc] [1,4] thiazine-1-ylidene} To a solution of benzenesulfonamide (251 mg, 0.442, fraction 1) in dichloromethane (200 ml) was added diisobutylaluminum hydride. A 1.5 mol / 1 toluene solution (0.590 ml, 0.884 leak 1) was added at -70 ° C. The reaction mixture was stirred at _70 ° C under a stream of argon for 7 hours. A saturated aqueous solution of ammonium chloride (250 ml) was added, the reaction mixture was stirred at room temperature for 30 minutes, and 1N hydrochloric acid was added, followed by extraction with dichloromethane. The extract was washed with water, dried (MgSO 4), the solvent was distilled off under reduced pressure. The residue was subjected to silica gel column chromatography, and eluted with n-hexane-ethyl acetate (10: 1, v / v) to give the title compound 21-6 (1) (15.7 mg, 6.2¾) and 21-6 (2) (26.0 mg, 10%) was obtained as dark red crystals.
題記化合物 21- 6(1)酢酸ェチルから再結晶して融点 174-175 の暗赤色結晶を得 た。 The title compound 21-6 (1) was recrystallized from ethyl acetate to give dark red crystals having a melting point of 174-175.
IR (KBr) ソ : 3240, 2934, 1748, 1711, 1663, 1597, 1557 cm-1. IR (KBr): 3240, 2934, 1748, 1711, 1663, 1597, 1557 cm- 1 .
Ή-NMR (CDC13) : δ 1.67-1.88 (4Η, m), 1.80 (3H, d, J=1.2 Hz), 1.92-2.22 (2H, 111), 2.28 (3H, d, J=l.2 Hz), 2.99 (2H, t, J=6.6 Hz), 8.00 (2H, d, J=8.4 Hz), Ή-NMR (CDC1 3): δ 1.67-1.88 (4Η, m), 1.80 (3H, d, J = 1.2 Hz), 1.92-2.22 (2H, 111), 2.28 (3H, d, J = l.2 Hz), 2.99 (2H, t, J = 6.6 Hz), 8.00 (2H, d, J = 8.4 Hz),
8.14 (2H, d, J=8.6 Hz), 9.58 (1H, bs), 10.09 (1H, s). 8.14 (2H, d, J = 8.6 Hz), 9.58 (1H, bs), 10.09 (1H, s).
元素分析値: C21H2QN304S3F5として Elementary analysis: as C 21 H 2Q N 3 0 4 S 3 F 5
計算値 (%) : C, 44.28; H, 3.54; N, 7.38; S, 16.89. Calculated (%): C, 44.28; H, 3.54; N, 7.38; S, 16.89.
実測値 (¾) : C, 44.39; H, 3.44; N, 7.73; S, 16.94. Found (¾): C, 44.39; H, 3.44; N, 7.73; S, 16.94.
題記化合物 21- 6 (2)酢酸ェチル -ジェテルエーテルから再結晶して融点 180-181で の暗赤色結晶を得た。 The title compound 21-6 (2) was recrystallized from ethyl acetate-jetether to give dark red crystals with a melting point of 180-181.
IR (KBr) V : 3700-2600, 3472, 1748, 1661, 1615, 1557 cm"1. IR (KBr) V: 3700-2600, 3472, 1748, 1661, 1615, 1557 cm " 1 .
—醒 R (CDC13) : 51.64-1.90 (4H, m), 1.79 (3H, d, J=1.2 Hz), 1.90-2.22 (2H, m), 2.27 (3H, d, J=l.2 Hz), 2.97 (2H, t, J=7.4 Hz), 4.78 (2H, d, J=6.2 Hz), 7.49 (2H, d, J=8.4 Hz), 7.95 (2H, d, J=8. Hz), 9.63 (1H, bs). —Awake R (CDC1 3 ): 51.64-1.90 (4H, m), 1.79 (3H, d, J = 1.2 Hz), 1.90-2.22 (2H, m), 2.27 (3H, d, J = l.2 Hz) ), 2.97 (2H, t, J = 7.4 Hz), 4.78 (2H, d, J = 6.2 Hz), 7.49 (2H, d, J = 8.4 Hz), 7.95 (2H, d, J = 8.Hz), 9.63 (1H, bs).
元素分析値: C21H22N304S3F5として Elementary analysis: as C 21 H 22 N 3 0 4 S 3 F 5
計算値 ) : C, 44.13; H, 3.88; N, 7.35; S, 16.83. Calculated)): C, 44.13; H, 3.88; N, 7.35; S, 16.83.
実測値 (¾) : C, 43.75; H, 3.90; N, 7.42; S, 16.95. Found (¾): C, 43.75; H, 3.90; N, 7.42; S, 16.95.
実施例 21 - 7 Example 21-7
4- (ジメチルァミノ) -N- {5, 6 -ジメチル -3-ォキソ -8 - [(5, 5, 6, 6, 6-ペン夕フルォロ へキシル) スルファニル ]-2, 3 -ジヒドロ- 1H-イミダゾ [5, l_c] [1, 4]チアジン - 1- 実施例 2卜 3 (方法 1) で得た化合物(195 mg, 0.350 mmol)のァセトニトリル (4.0 ml)および THF (0.5 ml) 懸濁液にホルマリン (純度 37%水溶液, 0.105 ml, 1.40 龍 ol) を室温で加え、 30分間攪拌後、 水素化シァノホウ素ナトリゥム (23.2mg, 0.350腿 ol)を加え、さらに 30分間攪拌した。反応液にホルマリン (0.053 ml, 0.70 mmol) および水素化シァノホウ素ナトリウム (12mg, 0.18匪 ol)を加え、 さらに 30分間攪拌した。 反応液を酢酸ェチル (30ml)で希釈し、 0.1N塩酸、 水および飽 和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣をメタノール で希釈した。 析出物を濾取し、 メタノールで洗浄後、 酢酸ェチルから結晶化して 表題化合物(100 mg, 0.171 腿 ol, 48.9%)を赤色粉末として得た。 4- (dimethylamino) -N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorofluorohexyl) sulfanyl] -2,3-dihydro-1H- Imidazo [5, l_c] [1, 4] thiazine-1- Example 2 A suspension of the compound obtained in Step 3 (Method 1) (195 mg, 0.350 mmol) in acetonitrile (4.0 ml) and THF (0.5 ml) To the mixture was added formalin (purity 37% aqueous solution, 0.105 ml, 1.40 dragonol) at room temperature, and after stirring for 30 minutes, sodium cyanoborohydride (23.2 mg, 0.350 liter) was added, and the mixture was further stirred for 30 minutes. Formalin (0.053 ml, 0.70 mmol) and sodium cyanoborohydride (12 mg, 0.18 ol) were added to the reaction solution, and the mixture was further stirred for 30 minutes. The reaction solution was diluted with ethyl acetate (30 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, washed with methanol, and crystallized from ethyl acetate to give the title compound (100 mg, 0.171 tmol, 48.9%) as a red powder.
Ή-NMR (300MHz, CDC13): (51.74 (4H, m), 1.78 (3H, brs), 2.04 (2H, m), 2.25 (3H, brs), 2.95 (2H, t, J= 6.8 Hz), 3.04 (6H, s), 6.66 (2H, d, J- 9.0 Hz), 7.77 (2H, d, J= 9.0 Hz), 9.63 (1H, br). Ή-NMR (300MHz, CDC1 3 ): (51.74 (4H, m), 1.78 (3H, brs), 2.04 (2H, m), 2.25 (3H, brs), 2.95 (2H, t, J = 6.8 Hz) , 3.04 (6H, s), 6.66 (2H, d, J- 9.0 Hz), 7.77 (2H, d, J = 9.0 Hz), 9.63 (1H, br).
元素分析値: C22H25N403S3F5として計算値: C, 45.20; H, 4.31; , 9.58; S, 16.45; F, 16.25 ( ) 、 実測値: C, 44.85; H, 4.34; N, 9.66; S, 16.66; F, 16.24 (%) . 即. 197.5-199. O Elemental analysis: C 22 H 25 N 4 0 3 S 3 F 5 Calculated: C, 45.20; H, 4.31 ;, 9.58; S, 16.45; F, 16.25 (), Found: C, 44.85; H, 4.34; N, 9.66; S, 16.66; F, 16.24 (%). Immediately. 197.5-199. O
実施例 21-8 Example 21-8
N - {5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6 -ペン夕フルォ口へキシル)スルファニ ル] -2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン トイリデン } -4- [(メチル 実施例 21- 3 (方法 2)で得た化合物の P-トルエンスルホン酸塩 (487 mg, 0.668 mmol)の THF (3.5 ml)懸濁液に室温で攪拌しながらピリジン(0.132 ml, 1.63 腿 ol)を加え、 20分間攪拌後、 メタンスルホン酸無水物(129 mg, 0.717 匪 ol)を 加え、 さらに 13時間攪拌した。 反応液を酢酸ェチル (30 ml)で希釈し、 0.1N塩酸 で洗浄後、 2%炭酸ナトリウム水溶液で抽出した。 抽出水層を酸性に調整し、 酢酸 ェチルで抽出した。 同様にして原料(254 mg, 0.348腿 ol)から得た有機層を合わ せて 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥、 濃縮し、 残渣にメタノールを加えた。 析出物を除去して母液およびメタノール洗液を濃縮 し、 ジェチルェ一テルで希釈した。 析出物をジェチルェ一テルで洗浄後、 乾燥し て表題化合物(104 mg, 0.164匪 ol, 16.1%)を紫色粉末として得た。 N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentylfluorohexyl) sulfanyl] -2,3-dihydrido-1H-imidazo [5 , 1-c] [1,4] thiazine toylidene} -4-[(methyl) P-toluenesulfonate (487 mg, 0.668 mmol) of the compound obtained in Example 21-3 (Method 2) in THF (3.5 pyridine (0.132 ml, 1.63 ml) at room temperature with stirring. After adding thigh ol) and stirring for 20 minutes, methanesulfonic anhydride (129 mg, 0.717 ol) was added, and the mixture was further stirred for 13 hours. The reaction solution was diluted with ethyl acetate (30 ml), washed with 0.1N hydrochloric acid, and extracted with a 2% aqueous sodium carbonate solution. The aqueous extraction layer was adjusted to be acidic and extracted with ethyl acetate. Similarly, the organic layers obtained from the raw materials (254 mg, 0.348 mol) were combined, washed with 0.1 N hydrochloric acid, water and saturated saline, dried over sodium sulfate and concentrated, and methanol was added to the residue. The precipitate was removed, the mother liquor and the methanol wash were concentrated, and diluted with geethyl ether. The precipitate was washed with geetyl ether and dried to give the title compound (104 mg, 0.164 ol, 16.1%) as a purple powder.
Ή—NMR (300MHz, DMS0- d6): 51.62 (2H, brq, J= 6.9 Hz), 1.70 (2H, brq, J= 7.2 Hz), 1.79 (3H, brs), 2.17 (3H, brs), 2.23 (2H, m), 3.03 (2H, t, J= 7.1 Hz), 3.11 (3H, s), 7.31 (2H, d, J= 9.0 Hz), 7.90 (2H, d, J= 9.0 Hz), 10.36 (1H, s), 11.64 (1H, brs). Ή-NMR (300MHz, DMS0- d 6): 51.62 (2H, brq, J = 6.9 Hz), 1.70 (2H, brq, J = 7.2 Hz), 1.79 (3H, brs), 2.17 (3H, brs), 2.23 (2H, m), 3.03 (2H, t, J = 7.1 Hz), 3.11 (3H, s), 7.31 (2H, d, J = 9.0 Hz), 7.90 (2H, d, J = 9.0 Hz), 10.36 (1H, s), 11.64 (1H, brs).
元素分析値: C21H23N405S4F5として計算値: C, 39.74; H, 3.65; N, 8.83; S, 20.21; F, 14.97 (%) 、 実測値: C, 40.07; H, 3.65; N, 8.85; S, 20.08; F, 14.32 (%) . mp. 186.5-188.0°C Elemental analysis: Calculated for C 21 H 23 N 4 0 5 S 4 F 5 : C, 39.74; H, 3.65; N, 8.83; S, 20.21; F, 14.97 (%), found: C, 40.07; H, 3.65; N, 8.85; S, 20.08; F, 14.32 (%). Mp. 186.5-188.0 ° C
実施例 21-9 Example 21-9
N-{5, 6-ジメチル- 3 -ォキソ - 8-[(5, 5, 6, 6, 6_ペンタフルォ口へキシル)スルファニ ル]- 2, 3-ジヒドロ - 1H-イミダゾ [5, 1-c] [1,4]チアジン _1 -ィリデン }- 4- { [(4-メチ ルフエ二ル) スルホニル] アミノ}ベンゼンスルホンアミド N- {5,6-dimethyl-3-oxo- 8-[(5,5,6,6,6_pentafluorohexyl) sulfanyl]-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine_1-ylidene}-4-{[(4-methylphenyl) sulfonyl] amino} benzenesulfonamide
実施例 21-3 (方法 2)で得た化合物の P-トルエンスルホン酸塩 (340 mg, 0.467 腿 ol)の THF (7 ml)懸濁液に室温で攪拌しながらピリジン(0.092 ml, 1.14顧 ol) を加え、ついで塩化 P-トルエンスルホニル (以下、 p- TsCiと略記することがある、 107 mg, 0.546匪 ol)を加え、 さらに 2時間攪拌した。 反応液にト リェチルァミン (0.063 ml, 0.455 mmol) を加え、 さらに 2時間攪拌した。 反応液に N,N-ジメチ ルァミノピリジン (以下 DMAPと略記することがある、 11 mg, 0.091 腿 ol) を加 え、 さらに 4時間攪拌した。 反応液にトリェチルァミン(0.063 ml, 0.455 mmol) および p- TsCl (53.5 mg, 0.273 mmol)を加え、 さらに 60時間攪拌した。 反応液 に 1N塩酸 (2.0 ml)を加え、 メタノール -水(1:1, 10 ml)で希釈した。 析出物を メタノール-水(1:1)およびメタノールで洗浄後、 乾燥して表題化合物(259 mg, 0.364匪 ol, 78. IDを紫色粉末として得た。 Example 21-3 Pyridine (0.092 ml, 1.14) was added to a suspension of the compound obtained in (Method 2) in P-toluenesulfonic acid salt (340 mg, 0.467 mol) in THF (7 ml) at room temperature with stirring. ol), followed by P-toluenesulfonyl chloride (hereinafter sometimes abbreviated as p-TsCi, 107 mg, 0.546 ol), and the mixture was further stirred for 2 hours. Triethylamine (0.063 ml, 0.455 mmol) was added to the reaction solution, and the mixture was further stirred for 2 hours. N, N-Dimethylaminopyridine (hereinafter abbreviated as DMAP, 11 mg, 0.091 tmol) was added to the reaction solution, and the mixture was further stirred for 4 hours. Triethylamine (0.063 ml, 0.455 mmol) and p-TsCl (53.5 mg, 0.273 mmol) were added to the reaction solution, and the mixture was further stirred for 60 hours. 1N Hydrochloric acid (2.0 ml) was added to the reaction solution, and the mixture was diluted with methanol-water (1: 1, 10 ml). The precipitate was washed with methanol-water (1: 1) and methanol, dried and dried to give the title compound (259 mg, 0.364 bandol ol, 78. ID was obtained as a purple powder.
Ή-NMR (300MHz, DMS0-d6): 51.61 (2H, brq, J= 6.6 Hz), 1.68 (2H, brq, J= 7.2 Hz), 1.77 (3H, brs), 2.16 (3H, brs), 2.22 (2H, m), 2.34 (3H, s), 3.00 (2H, t, J= 7.1 Hz), 7.23 (2H, d, J= 8.8 Hz), 7.36 (2H, d, J= 8.1 Hz), 7.72 (2H, d, J= 7.8 Hz), 7.79 (2H, d, J= 8.7 Hz), 10.84 (1H, s), 11.59 (1H, brs). 元素分析値: C27H27N405S4F5として計算値: C, 45.62; H, 3.83; N, 7.88; S, 18.04; F, 13.36 ( ) 、 実測値: C, 45.58; H, 4.03; N, 7.94; S, 18.19; F, 13.35 (%) . 即. 225.5- 226.5°C , Ή-NMR (300 MHz, DMS0-d 6 ): 51.61 (2H, brq, J = 6.6 Hz), 1.68 (2H, brq, J = 7.2 Hz), 1.77 (3H, brs), 2.16 (3H, brs), 2.22 (2H, m), 2.34 (3H, s), 3.00 (2H, t, J = 7.1 Hz), 7.23 (2H, d, J = 8.8 Hz), 7.36 (2H, d, J = 8.1 Hz), 7.72 (2H, d, J = 7.8 Hz), 7.79 (2H, d, J = 8.7 Hz), 10.84 (1H, s), 11.59 (1H, brs). Elemental analysis: C 27 H 27 N 4 0 5 S 4 F 5 calculated: C, 45.62; H, 3.83 ; N, 7.88; S, 18.04; F, 13.36 (), Found: C, 45.58; H, 4.03 ; N, 7.94; S, 18.19; F , 13.35 (%). Immediately. 225.5-226.5 ° C,
実施例 21 - 10 Example 21-10
5 - {[5, 6-ジメチル -3-ォキソ-卜({[4- (トリフルォロメチル)フエニル]スルホ二 ル}ィミノ) - 2, 3 -ジヒドロ- 1H-イミダゾ [5, 1 - c] [1, 4]チアジン- 8 -ィル]スルファ 二ル}吉草酸 5-{[5,6-Dimethyl-3-oxo-({[4- (trifluoromethyl) phenyl] sulfonyl} imino) -2,3-dihydro-1H-imidazo [5,1-c [1,4] Thiazine-8-yl] sulfanyl} valeric acid
実施例 7-105 で得られたェチル 5-{[5,6-ジメチル- 3-ォキソ - 1_({[4_ (トリフ ルォロメチル)フエニル]スルホ二ル}ィミノ) -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン - 8-ィル]スルファニル }ペンタノェ一ト (1.05 g, 1.86 ■ol) のエタノール (10.0 ml) 及びテトラヒドロフラン (10.0 ml) 溶液に、 1N 水酸化ナトリウム水溶液(5.59ml) を加えた。 反応混合物を室温で 1時間かき混 ぜた後、溶媒を減圧下に留去した。残渣に 1N塩酸を加えて、酢酸ェチルで抽出し た。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(747 mg, 75%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルェ一テルから再結晶して融 点 191_193°Cの暗赤色結晶を得た。 Ethyl 5-{[5,6-dimethyl-3-oxo-1 _ ({[4_ (trifluoromethyl) phenyl] sulfonyl} imino) -2,3-dihydro-1H- obtained in Example 7-105 To a solution of imidazo [5, 1-c] [1,4] thiazine-8-yl] sulfanyl} pentanoate (1.05 g, 1.86 ol) in ethanol (10.0 ml) and tetrahydrofuran (10.0 ml) was added 1N aqueous solution. An aqueous solution of sodium oxide (5.59 ml) was added. After stirring the reaction mixture at room temperature for 1 hour, the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (747 mg, 75%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 191 to 193 ° C.
IR (KBr) V: 3700-2400, 3125, 2928, 1725, 1663, 1599, 1557 cm"1. IR (KBr) V: 3700-2400, 3125, 2928, 1725, 1663, 1599, 1557 cm " 1 .
Ή-NMR (CDC13) : (51.70-1.83 (4H, m), 1.80 (3H, d, J=1.2 Hz), 2.28 (3H, d, J=1.2Hz), 2.37-2.45 (2H, m), 2.94-3.02 (2H, m), 7.77 (2H, d, J=8.1 Hz), 8.11 (2H, d, J=8.4 Hz), 9.69 (1H, bs) . Ή-NMR (CDC1 3): (51.70-1.83 (4H, m), 1.80 (3H, d, J = 1.2 Hz), 2.28 (3H, d, J = 1.2Hz), 2.37-2.45 (2H, m) , 2.94-3.02 (2H, m), 7.77 (2H, d, J = 8.1 Hz), 8.11 (2H, d, J = 8.4 Hz), 9.69 (1H, bs).
元素分析値: C20H20N305S3F3-0.3H2Oとして Elementary analysis: as C 20 H 20 N 3 0 5 S 3 F 3 -0.3H 2 O
計算値 (%) : C, 44.40; H, 3.83; N, 7.77; S, 17.78. Calculated value (%): C, 44.40; H, 3.83; N, 7.77; S, 17.78.
実測値 (¾) : C, 44.39; H, 4.04; N, 7.83; S, 17.66. Found (¾): C, 44.39; H, 4.04; N, 7.83; S, 17.66.
実施例 21-11 5-{[5, 6-ジメチル- 3 -ォキソ -卜( { [4- (トリフルォロメチル)フエニル]スルホ二 ル}ィミノ) -2, 3-ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル]スルファ ニル卜 N-へキシルペンタンアミド Example 21-11 5-{[5,6-dimethyl-3-oxo-butanol ({[4- (trifluoromethyl) phenyl] sulfonyl} imino) -2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazin-8-yl] sulfanyl nitrate N-hexylpentanamide
実施例 2卜 10で得られた 5-{ [5, 6-ジメチル- 3-ォキソ -卜({[4- (トリフルォロメ チル)フエニル]スルホ二ル}ィミノ)- 2, 3-ジヒドロ- 1H -イミダゾ [5,1- c] [1,4]チ ァジン- 8-ィル]スルファニル }吉草酸 (442 mg, 8.27匪 ol) と卜へキシルァミン (0.135 ml, 10.2匪。1) の N, N-ジメチルホルムアミド (5.00 ml) 溶液に、 シァ ノりん酸ジェチル(0.165 ml, 10.2腿 ol)続いてト リェチルァミン(0.142 ml, 10.2 mmol) を加えた。反応混合物を室温で 21.5時間かき混ぜた後、水を加えて酢酸ェ チルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記 化合物 (23.6 mg, 4.6%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテ ルから再結晶して融点 117-119°Cの暗赤色結晶を得た。 Example 2 5-{[5,6-Dimethyl-3-oxo-({[4- (trifluoromethyl) phenyl] sulfonyl} imino) -2,3-dihydro-1H- N, N of imidazo [5,1-c] [1,4] thiazin-8-yl] sulfanyl} valeric acid (442 mg, 8.27 marl ol) and trihexylamine (0.135 ml, 10.2 marl. 1) To a solution of -dimethylformamide (5.00 ml) was added getyl cyanophosphate (0.165 ml, 10.2 mol), followed by triethylamine (0.142 ml, 10.2 mmol). After stirring the reaction mixture at room temperature for 21.5 hours, water was added, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain a solvent the title compound was evaporated under reduced pressure (23.6 mg, 4.6%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether yielded dark red crystals with a melting point of 117-119 ° C.
IR (KBr) V: 3252, 2932, 2860, 1748, 1622, 1557 cm一1. IR (KBr) V: 3252, 2932, 2860, 1748, 1622, 1557 cm- 1 .
Ή-NM (CDC13) : 50.881 (3H, t, J=6.9 Hz), 1.24-1.38 (6H, in), 1.42-1.54 (2H, m), 1.68-1.84 (4H, m) , 1.80 (3H, d, J=0.9 Hz), 2.19 (2H, t, J=6.6 Hz), 2.28 Ή-NM (CDC1 3): 50.881 (3H, t, J = 6.9 Hz), 1.24-1.38 (6H, in), 1.42-1.54 (2H, m), 1.68-1.84 (4H, m), 1.80 (3H , d, J = 0.9 Hz), 2.19 (2H, t, J = 6.6 Hz), 2.28
(3H, d, J=0.9Hz), 2.98 (2H, t, J=6.9 Hz), 3.24 (2H, q, J=7.2 Hz), 5.45 (1H, bs), 7.77 (2H, d, J=8.1 Hz), 8.10 (2H, d, J=8.1 Hz), 9.63 (1H, bs). (3H, d, J = 0.9Hz), 2.98 (2H, t, J = 6.9 Hz), 3.24 (2H, q, J = 7.2 Hz), 5.45 (1H, bs), 7.77 (2H, d, J = 8.1 Hz), 8.10 (2H, d, J = 8.1 Hz), 9.63 (1H, bs).
実施例 21 - 12 Examples 21-12
N-{5, 6-ジメチル _3-ォキソ _8_[(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル] -2,3-ジヒドロ- 1H-イミダゾ [5,1-c] [1, 4]チアジン- 1 -ィリデン卜 4- (2-ォキソ N- {5,6-dimethyl_3-oxo_8 _ [(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c ] [1,4] thiazine-1-yl 4- (2-oxo
-卜ピロリジニル)ベンゼンスルホンアミド -Topyrrolidinyl) benzenesulfonamide
実施例 21- 23で得た化合物 (0.33 g, 0.42腿 ol)の DMF (5 ml)懸濁液に水素 化ナトリウム (純度 60%油性, 68 nig, 1.70歷 ol) を室温で加え、 ついでヨウ化 ナトリウム(10 nig)を加え、 60分間攪拌した。反応液に 1N塩酸 (2.0 ml)を加え、 酢酸ェチル (30 ml)で希釈し、 0.1N塩酸、 水および飽和食塩水で洗 後、 硫酸ナ トリウムで乾燥した。 濃縮乾固後、 残渣を酢酸ェチルおよびジェチルエーテルで 希釈した。析出物を濾取し、ジェチルェ一テルで洗浄後、乾燥して表題化合物 (173 mg, 0.277腿 ol, 65.3%)を赤色粉末として得た。 To a suspension of the compound obtained in Examples 21-23 (0.33 g, 0.42 mol) in DMF (5 ml) was added sodium hydride (purity 60% oily, 68 nig, 1.70 mol) at room temperature. Sodium chloride (10 nig) was added and the mixture was stirred for 60 minutes. The reaction mixture was diluted with 1N hydrochloric acid (2.0 ml), diluted with ethyl acetate (30 ml), washed with 0.1N hydrochloric acid, water and saturated saline, and then dried over sodium sulfate. After concentration to dryness, the residue was diluted with ethyl acetate and getyl ether. The precipitate was collected by filtration, washed with diethyl ether, and dried to give the title compound (173 mg, 0.277 mol, 65.3%) as a red powder.
lH-匪 R (300MHz, CDC13): δΐ.75 (4Η, m), 1.80 (3H, s), 2.05 (2H, tt, J= 17.4, 7.5 Hz), 2.20 (2H, t, J= 7.7 Hz), 2.27 (3H, q, J- 0.6 Hz), 2.65 (2H, t, J= 8.3 Hz), 2.96 (2H, t, J= 6.9 Hz), 3.89 (2H, t, J= 6.9 Hz), 7.78 (2H, d, J= 9.0 Hz), 7.95 (2H, d, J= 8.7 Hz), 9.63 (1H, brs). lH- negation R (300MHz, CDC1 3): δΐ.75 (4Η, m), 1.80 (3H, s), 2.05 (2H, tt, J = 17.4, 7.5 Hz), 2.20 (2H, t, J = 7.7 Hz), 2.27 (3H, q, J- 0.6 Hz), 2.65 (2H, t, J = 8.3 Hz), 2.96 (2H, t, J = 6.9 Hz) ), 3.89 (2H, t, J = 6.9 Hz), 7.78 (2H, d, J = 9.0 Hz), 7.95 (2H, d, J = 8.7 Hz), 9.63 (1H, brs).
元素分析値: C24H24N404S3F5として計算値: C, 46.15; H, 4.03; N, 8.97; S, 15.40; F, 15.21 (%) 、 実測値: C, 45.85; H, 3.99; N, 8.94; S, 15.40; F, 14.99 (%) . mp. 179.0-181.5°C Elemental analysis: C 24 H 24 N 4 0 4 S 3 F 5 Calculated: C, 46.15; H, 4.03 ; N, 8.97; S, 15.40; F, 15.21 (%), Found: C, 45.85; H, 3.99; N, 8.94; S, 15.40; F, 14.99 (%). Mp. 179.0-181.5 ° C
実施例 21 - 13 Examples 21-13
5_{[1-({[4- (ァセチルアミノ)フエニル]スルホ二ル}イミノ) - 5, 6-ジメチル -3- ォキソ -2, 3 ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 8 -ィル]スルファニル } 吉草酸 5 _ {[1-({[4- (Acetylamino) phenyl] sulfonyl} imino) -5,6-dimethyl-3-oxo-2,3 dihydro-1H-imidazo [5,1-c] [1, 4] thiazine-8-yl] sulfanyl} valeric acid
実施例 7-141 で得られたェチル 5-{[1-({ [4- (ァセチルアミノ)フエニル]スル ホニル }ィミノ)- 5, 6_ジメチル- 3-ォキソ- 2, 3-ジヒ ドロ -1H-イミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル]スルファニル }ペンタノェ一ト (200 mg, 0.361 mmol) のエタノール (5.00 ml) 及びテトラヒドロフラン (5.00 ml) 溶液に、 1N 水酸化ナトリゥム水溶液(1.08 ml) を加えた。 反応混合物を室温で 1時間かき混 ぜた後、溶媒を減圧下に留去した。残渣に 1N塩酸を加えて、酢酸ェチルで抽出し た。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(167 mg, 88%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融 点 226- 228°Cの暗赤色結晶を得た。 Ethyl 5-{[1-({[4- (acetylamino) phenyl] sulfonyl} imino) -5,6_dimethyl-3-oxo-2,3-dihydro-1H obtained in Examples 7-141 To a solution of -imidazo [5,1-c] [1,4] thiazin-8-yl] sulfanyl} pentanoate (200 mg, 0.361 mmol) in ethanol (5.00 ml) and tetrahydrofuran (5.00 ml) was added 1N aqueous solution. An aqueous solution of sodium oxide (1.08 ml) was added. After stirring the reaction mixture at room temperature for 1 hour, the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (167 mg, 88%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 226-228 ° C.
IR (KBr) リ: 3700-2700, 3314, 2930, 1742, 1667, 1591, 1549 cm—1. IR (KBr): 3700-2700, 3314, 2930, 1742, 1667, 1591, 1549 cm— 1 .
Ή—NMR (CDC13) : (51.50-1.70 (4H, m), 1.79 (3H, s), 2.08 (3H, s), 2.17 (3H, s), 2.18-2.28 (2H, m), 2.90-3.00 (2H, m), 7.73 (2H, d, J=8.4 Hz), 7.87 (2H, d, J=8.4 Hz), 10.30 (1H, bs), 11.61 (1H, bs). Ή-NMR (CDC1 3): (51.50-1.70 (4H, m), 1.79 (3H, s), 2.08 (3H, s), 2.17 (3H, s), 2.18-2.28 (2H, m), 2.90- 3.00 (2H, m), 7.73 (2H, d, J = 8.4 Hz), 7.87 (2H, d, J = 8.4 Hz), 10.30 (1H, bs), 11.61 (1H, bs).
元素分析値: C21¾4N40sS30.7H20として Elemental analysis: C 21 ¾ 4 N 40 s S 3 0.7H 20
計算値 (%) : C, 46.95; H, 4.77; N, 10.43; S, 17:91. Calculated value (%): C, 46.95; H, 4.77; N, 10.43; S, 17:91.
実測値 (%) : C, 46.87; H, 4.74; N, 10.33; S, 18.23. Found (%): C, 46.87; H, 4.74; N, 10.33; S, 18.23.
実施例 21- 15 Example 21-15
4-ブロモ -N - [5, 6-ジメチル- 8- [ (5-フルォ口ぺンチル)スルファニル] -3-ォキソ - 2, 3-ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, ]チアジン- 1-ィリデン]ベンゼンスルホ ンアミド ナトリウム塩 4-bromo-N- [5,6-dimethyl-8-[(5-fluoroopentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, ] Thiazine-1-ylidene] benzenesulfo Amide sodium salt
実施例 7-72で得た化合物(2.17 g, 4.06 mmol)の THF -メタノール (1:1, 100 ml) 懸濁液に IN水酸化ナトリウム水溶液(4.00 ml, .00 mmol) を加え、約 50°C に加温して溶解した。得られた溶液に水(50 ml)を加え、 減圧下濃縮して THFおよ びメタノールを除去した。 析出物を濾取し、 水およびメタノール—水 (1:1)で洗 浄後、 乾燥して表題化合物(1.92 g, 3.45匪01, 85.0 )を紫褐色粉末として得た。 Ή-NMR (200MHz, CDC13): 51.47 (2H, m), 1.60 - 1.75 (4H, m), 1.65 (3H, s), 2.02 (3H, s), 2.87 (2H, t, J= 7.2 Hz), 4.42 (2H, dt, 1= 47.4, 5.9 Hz), 7.41 (2H, d, J= 8.6 Hz), 7.80 (2H, d, J= 8.0 Hz). To a suspension of the compound obtained in Example 7-72 (2.17 g, 4.06 mmol) in THF-methanol (1: 1, 100 ml) was added IN aqueous sodium hydroxide solution (4.00 ml, .00 mmol), and the mixture was treated with about 50%. Heated to ° C to dissolve. Water (50 ml) was added to the obtained solution, and the mixture was concentrated under reduced pressure to remove THF and methanol. The precipitate was collected by filtration, washed with water and methanol-water (1: 1), and dried to give the title compound (1.92 g, 3.45 band01, 85.0) as a purple-brown powder. Ή-NMR (200MHz, CDC1 3 ): 51.47 (2H, m), 1.60 - 1.75 (4H, m), 1.65 (3H, s), 2.02 (3H, s), 2.87 (2H, t, J = 7.2 Hz ), 4.42 (2H, dt, 1 = 47.4, 5.9 Hz), 7.41 (2H, d, J = 8.6 Hz), 7.80 (2H, d, J = 8.0 Hz).
元素分析値: C19H2。N303S3BrFNa として計算値: C, 41.01; H, 3.62; N, 7.55; S, 17.29; Br, 14.36; F, 3.41 (%) 、 実測値: C, 40.66; H, 3.66; , 7.57; S, 17.41; Br, 14.28; F, 3.50 (%) . Elemental analysis: C 19 H 2. Calculated for N 3 0 3 S 3 BrFNa: C, 41.01; H, 3.62; N, 7.55; S, 17.29; Br, 14.36; F, 3.41 (%), found: C, 40.66; H, 3.66;, 7.57; S, 17.41; Br, 14.28; F, 3.50 (%).
実施例 21-16 Example 21-16
5- [(5,6-ジメチル- 1- {[(4-ァセチルフエニル)スルホニル]イミノ}- 3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5, 1-c] [1 , 4]チアジン - 8-ィル)スルファニル] -N -へ キシルペンタンアミド 5-[(5,6-dimethyl-1-{[(4-acetylphenyl) sulfonyl] imino} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -8-yl) sulfanyl] -N-hexylpentanamide
水素化ナトリウム(9.74 mg, 0.243 mmol) (60%油性)のテトラヒドロフラン(0.50 ml)の懸濁液に、実施例 6-33で得られた 5 - [1-ァミノ -5, 6-ジメチル -3-ォキソ -3H - イミダゾ [5, 1 - c] [1, 4]チアジン- 8-ィル]スルファニル]- N-へキシルペンタンアミ ド (50.0 mg, 0.121 匪 ol) のテトラヒドロフラン (0.50 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに 4-ァセチルベンゼンスルホニル クロリド(41.2mg, 0.183顏 ol) のテトラヒドロフラン (0.50 il) 溶液を加え、 さらに室温で 21時 間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水 洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (23.8mg, 33%) を暗 赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 103-105^の暗赤色結晶を得た。 To a suspension of sodium hydride (9.74 mg, 0.243 mmol) (60% oil) in tetrahydrofuran (0.50 ml) was added the 5- [1-amino-5,6-dimethyl-3 obtained in Example 6-33. -Oxo-3H-imidazo [5,1-c] [1,4] thiazin-8-yl] sulfanyl] -N-hexylpentanamide (50.0 mg, 0.121 bandol) in tetrahydrofuran (0.50 ml) And stirred under ice cooling for 5 minutes. To this was added a solution of 4-acetylbenzenesulfonyl chloride (41.2 mg, 0.183 mol) in tetrahydrofuran (0.50 il), and the mixture was further stirred at room temperature for 21 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was water washed, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (23.8 mg, 33%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 103-105 ^.
IR (KBr) V: 3266, 3073, 2955, 2930, 2859, 1752, 1692, 1645, 1615, 1557 cm"1. ¾-NMR (CDC13) : (50.881 (3H, t, J=7.0 Hz), 1.24-1.38 (6H, m), 1.42-1.56 (2H, m), 1.68-1.84 (4H, m), 1.79 (3H, d, J=1.2 Hz), 2.19 (2H, t, J=6.2 Hz), 2.28 (3H, d, J=1.2 Hz), 2.65 (3H, s), '2.97 (2H, t, J=7.0 Hz), 3.24 (2H, q, J=5.8 Hz), 5.49 (1H, bs), 8.06 (4H, s), 9.63 (1H, bs). IR (KBr) V:. 3266 , 3073, 2955, 2930, 2859, 1752, 1692, 1645, 1615, 1557 cm "1 ¾-NMR (CDC1 3): (50.881 (3H, t, J = 7.0 Hz), 1.24-1.38 (6H, m), 1.42-1.56 (2H, m), 1.68-1.84 (4H, m), 1.79 (3H, d, J = 1.2 Hz), 2.19 (2H, t, J = 6.2 Hz) , 2.28 (3H, d, J = 1.2 Hz), 2.65 (3H, s), '2.97 (2H, t, J = 7.0 Hz), 3.24 (2H, q, J = 5.8 Hz), 5.49 (1H, bs), 8.06 (4H, s), 9.63 (1H, bs).
実施例 21-17 Example 21-17
N-{4- [({8- [(3-ァミノプロピル)スルファニル ]-5, 6-ジメチル- 3-ォキソ -2, 3 -ジ ヒドロ- 1H-イミダゾ [5,l-c] [1,4]チアジン -卜ィリデン }ァミノ)スルホニル]フエ 二ル}ァセタミド 塩酸塩 N- {4-[({8-[(3-Aminopropyl) sulfanyl] -5,6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, lc] [1,4] thiazine -Trilidene} amino) sulfonyl] phenyl} acetamide hydrochloride
実施例 7-152 で得た化合物の粉末(純度 95%, 255 mg, 0.416 mmol) を TFA The compound powder (purity 95%, 255 mg, 0.416 mmol) obtained in Example 7-152 was added to TFA
(1.0 ml)に溶解し、 室温で 80分間放置した。 反応液をトルエン(25 ml)で希釈 して濃縮乾固し、残渣にトルエン(25 ml)を加えて濃縮した。得られた残渣を THF- エタノール(1:1, 50 ml)に溶解し、 1N塩酸 (0.50 ml)を加えて濃縮し、 残渣に(1.0 ml) and left at room temperature for 80 minutes. The reaction solution was diluted with toluene (25 ml), concentrated to dryness, and the residue was concentrated by adding toluene (25 ml). The obtained residue was dissolved in THF-ethanol (1: 1, 50 ml), and concentrated by adding 1N hydrochloric acid (0.50 ml).
THF-メタノール(1: 1)を加えて濃縮して赤色の懸濁液を得た。これを THFおよび酢 酸ェチルで希釈した。 析出物を濾取し、 酢酸ェチルで洗浄後、 乾燥して表題化合 物(200 mg, 0.386 mmol, 92.8%)を灰赤色粉末として得た。 THF-methanol (1: 1) was added and concentrated to give a red suspension. This was diluted with THF and ethyl acetate. The precipitate was collected by filtration, washed with ethyl acetate, and dried to give the title compound (200 mg, 0.386 mmol, 92.8%) as a gray-red powder.
Ή-NMR (200MHz, DMS0-d6): δ 1.80 (3Η, s), 1.89 (2H, quint, J= 7.2 Hz), 2.09 (3H, s), 2.17 (3H, s), 2.86 (2H, brq, J= 7 Hz), 3.05 (2H, t, J= 6.6 Hz), 7.76Ή-NMR (200 MHz, DMS0-d 6 ): δ 1.80 (3Η, s), 1.89 (2H, quint, J = 7.2 Hz), 2.09 (3H, s), 2.17 (3H, s), 2.86 (2H, brq, J = 7 Hz), 3.05 (2H, t, J = 6.6 Hz), 7.76
(2H, d, J= 9.2 Hz), 7.86 (3H, brs), 7.87 (2H, d, J= 8.8 Hz), 10.41 (1H, s),(2H, d, J = 9.2 Hz), 7.86 (3H, brs), 7.87 (2H, d, J = 8.8 Hz), 10.41 (1H, s),
11.65 (1H, brs). 11.65 (1H, brs).
元素分析値: C19H24N504S3C1 ·1.2Η20として計算値: C, 42.28; Η, 4.93; Ν, 12.98; S, 17.82; C1, 6.57 (%) 、 実測値: C, 42.11; Η, 5.01; , 12.90; S, 18.23; C1, 6.47 (%) . Elemental analysis: C 19 H 24 N 5 0 4 S 3 C1 · 1.2Η 2 0 Calculated: C, 42.28; Η, 4.93 ; Ν, 12.98; S, 17.82; C1, 6.57 (%), Found: C, 42.11; Η, 5.01;, 12.90; S, 18.23; C1, 6.47 (%).
mp. 192-195°C (deco即.) mp. 192-195 ° C (deco immediately)
実施例 21 - 18 Examples 21-18
5 -( {5, 6-ジメチル- 1- [ (n -ォクチルスルホニル)ィミノ] - 3-ォキソ -2, 3 -ジヒドロ -1H-イミダゾ [5, 1-c] [1,4]チアジン- 8-ィル }スルファニル) へキシルペンタン アミド 5-({5,6-dimethyl-1-[(n-octylsulfonyl) imino] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine- 8-yl} sulfanyl) hexylpentane amide
水素化ナトリウム(9.74 mg, 0.243腿 ol) (60%油性)のテトラヒドロフラン(0.50 ml)の懸濁液に、実施例 6-33で得られた 5 -[卜ァミノ- 5, 6-ジメチル- 3-ォキソ -3H - イミダゾ [5, 1-c] [1, 4]チアジン -8-ィル]スルファニル] -N-へキシルペン夕ンアミ ド (50.0 mg, 0.121 匪 ol) のテトラヒドロフラン (0.50 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに n-オクタンスルホニル クロリド (0.0337 ml, 0.183 mmol) のテトラヒドロフラン (0.50 ml) 溶液を加え、 さらに室温で 21時間かき 混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (23.4mg, 33%) を暗赤色結 晶として得た。酢酸ェチル -ジェチルエーテルから再結晶して融点 128- 13(TCの暗 赤色結晶を得た。 To a suspension of sodium hydride (9.74 mg, 0.243 tmol) (60% oil) in tetrahydrofuran (0.50 ml) was added the 5- [triamino-5,6-dimethyl-3) obtained in Example 6-33. -Oxo-3H-imidazo [5, 1-c] [1,4] thiazine-8-yl] sulfanyl] -N-hexylpentene amide (50.0 mg, 0.121 bandol) in tetrahydrofuran (0.50 ml) Add ice-cold Stir for 5 minutes below. To this was added a solution of n-octanesulfonyl chloride (0.0337 ml, 0.183 mmol) in tetrahydrofuran (0.50 ml), and the mixture was further stirred at room temperature for 21 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, Drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (23.4 mg, 33%) was obtained as a dark red crystals of. The crystals were recrystallized from ethyl acetate-getyl ether to obtain dark red crystals having a melting point of 128-13 (TC).
IR (KBr) V: 3179, 3086, 2928, 2857, 1755, 1740, 1665, 1640, 1574 cm"1. 一 NMR (CDC13) : (50.884 (6H, t, J=6.9 Hz), 1.20-1.36 (14H, m), 1.36-1.54 (4H, m), 1.67-1.82 (4H, m), 1.81 (3H, d, J=0.6 Hz), 1.82-1.92 (2H, m), 2.20 (2H, t, J=6.9 Hz), 2.27 (3H, d, J=0.6 Hz), 2.98 (2H, t, J=6.9 Hz), 3.11 (2H, t, J=7.8 Hz), 3.24 (2H, q, J=6.0 Hz), 5.47 (1H, bs), 9.48 (1H, bs). IR (KBr) V:. 3179 , 3086, 2928, 2857, 1755, 1740, 1665, 1640, 1574 cm "1 one NMR (CDC1 3): (50.884 (6H, t, J = 6.9 Hz), 1.20-1.36 (14H, m), 1.36-1.54 (4H, m), 1.67-1.82 (4H, m), 1.81 (3H, d, J = 0.6 Hz), 1.82-1.92 (2H, m), 2.20 (2H, t , J = 6.9 Hz), 2.27 (3H, d, J = 0.6 Hz), 2.98 (2H, t, J = 6.9 Hz), 3.11 (2H, t, J = 7.8 Hz), 3.24 (2H, q, J = 6.0 Hz), 5.47 (1H, bs), 9.48 (1H, bs).
実施例 21- 19 Examples 21-19
4 -ブ口モ -N - [5, 6-ジメチル- 8- [ (5-フルォ口ぺンチル)スルファニル] -3-ォキソ -2, 3 -ジヒドロ- 1H-イミダゾ [5,1- c] [1,4]チアジン- 1 -ィリデン]ベンゼンスルホ ンアミド カルシウム塩 4-Bumo-N- [5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-1-ylidene] benzenesulfonamide calcium salt
実施例 7- 72で得た化合物(1.00 g, 1.87 mmol)の THF-メタノール (1:1, 60 ml) 懸濁液に水酸化カルシウム (70.0 mg, 0.898 mmol)の水溶液 (40 ml)を加えた。 混合物を約 5(TCに加温し、 さらに THFおよびメタノールを加えて溶解した。 得ら れた溶液を減圧下約 50mlまで濃縮し、得られた析出物を濾取後、水およびメタノ —ル—水 (1:1)で洗浄し、 乾燥して表題化合物 (813 mg, 1.47 mmol, 78.5%)を紫 褐色粉末として得た。 To a suspension of the compound obtained in Examples 7-72 (1.00 g, 1.87 mmol) in THF-methanol (1: 1, 60 ml) was added an aqueous solution (40 ml) of calcium hydroxide (70.0 mg, 0.898 mmol). Was. The mixture was heated to about 5 (TC and dissolved by further addition of THF and methanol. The obtained solution was concentrated to about 50 ml under reduced pressure, and the obtained precipitate was collected by filtration, and washed with water and methanol. —Washed with water (1: 1) and dried to give the title compound (813 mg, 1.47 mmol, 78.5%) as a purple-brown powder.
Ή—匪 R (200MHz, CDC13): d 1.55 (2H, m), 1.60-1.80 (4H, m), 1.73 (3H, s), 2.11 (3H, s), 2.91 (1H, t, J= 7.4Hz), 4.40 (2H, dt, J- 46.8, 5.7 Hz), 7.48 (2H, brd, J= 7.8 Hz), 7.80 (2H, d, J= 8.0 Hz), 9.62 (1H, brs). Ή- negation R (200MHz, CDC1 3): d 1.55 (2H, m), 1.60-1.80 (4H, m), 1.73 (3H, s), 2.11 (3H, s), 2.91 (1H, t, J = 7.4Hz), 4.40 (2H, dt, J- 46.8, 5.7 Hz), 7.48 (2H, brd, J = 7.8 Hz), 7.80 (2H, d, J = 8.0 Hz), 9.62 (1H, brs).
元素分析値: C19H2QN3O3S3BrF.0.5Ca.0.5¾Oとして計算値: C, 40.57; H, 3.76; N, 7.47; S, 17.10 (%) 、 実測値: C, 40.73; H, 3.80; N, 7.50; S, 17.12 (%) . 実施例 21-20 Elemental analysis: C 19 H 2Q N 3 O 3 S 3 BrF.0.5Ca.0.5¾O Calculated: C, 40.57; H, 3.76 ; N, 7.47; S, 17.10 (%), Found: C, 40.73 ; H, 3.80; N, 7.50; S, 17.12 (%). Examples 21-20
4 - [ ({5, 6-ジメチル- 8- [ (5-フルォ口ぺンチル)スルファニル] -3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5,1 - c] [1,4]チアジン-卜イリデン }ァミノ)スルホニル]安息香 実施例 7-162で得られたェチル 4_[({5, 6 -ジメチル - 3_ォキソ - 8_[(5-フルォロ ペンチル)スルファニル ]-2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン-卜ィ リデン }ァミノ)スルホニル]ベンゾェ一ト (260 mg, 0.514 mmol) のエタノール (5.00 ml) 及びテトラヒドロフラン (5.00 ml) 溶液に IN水酸化ナトリウム水溶 液 (1.54ml) を加えた。 反応混合物を室温で 1.5時間かき混ぜた後、 溶媒を減圧 下に留去した。残渣に 1N塩酸を加えて、 生成した沈殿物を濾取し、 水洗後、 乾燥 して題記化合物 (241 mg, 94%) を暗赤色結晶として得た。 酢酸ェチル -ジェチル エーテルから再結晶して融点 4_146°Cの暗赤色結晶を得た。 4-[({5,6-Dimethyl-8-[(5-fluoroopentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4 ] Thiazine-triylidene} amino) sulfonyl] benzoic Etyl 4 _ [({5,6-dimethyl-3_oxo-8 _ [(5-fluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5,1-c] obtained in Example 7-162 A solution of [1,4] thiazine-trilidene} amino) sulfonyl] benzoate (260 mg, 0.514 mmol) in ethanol (5.00 ml) and tetrahydrofuran (5.00 ml) was added IN aqueous sodium hydroxide solution (1.54 ml). Was added. After stirring the reaction mixture at room temperature for 1.5 hours, the solvent was distilled off under reduced pressure. 1N Hydrochloric acid was added to the residue, and the resulting precipitate was collected by filtration, washed with water, and dried to give the title compound (241 mg, 94%) as dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 4-146 ° C.
IR (KBr) V 3700-2300, 3208, 2936, 1759, 1696, 1663, 1620, 1559 cm1. Ή-NMR (CDC13 + DMS0-d6) : 51.46-1.60 (2H, m), 1:65-1.82 (4H, m), 1.80 (3H, d, J=1.2 Hz), 2.27 (3H, d, J=0.9 Hz), 2.96 (2H, t, J=7.2 Hz), 4.44 (2H, dt, J=47.4, 5.7 Hz), 8.03 (2H, d, J=8.7 Hz), 8.14 (2H, d, J=8.7 Hz), 10.45 (1H, bs). . IR (KBr) V 3700-2300, 3208, 2936, 1759, 1696, 1663, 1620, 1559 cm 1 Ή-NMR (CDC1 3 + DMS0-d 6): 51.46-1.60 (2H, m), 1:65 -1.82 (4H, m), 1.80 (3H, d, J = 1.2 Hz), 2.27 (3H, d, J = 0.9 Hz), 2.96 (2H, t, J = 7.2 Hz), 4.44 (2H, dt, J = 47.4, 5.7 Hz), 8.03 (2H, d, J = 8.7 Hz), 8.14 (2H, d, J = 8.7 Hz), 10.45 (1H, bs).
元素分析値: C2QH22N305S3Fとして Elemental analysis: as C 2Q H 22 N 3 0 5 S 3 F
計算値 (%) : C, 48.08; H, 4.44; N, 8.41; S, 19.25. Calculated (%): C, 48.08; H, 4.44; N, 8.41; S, 19.25.
実測値 (%) : C, 48.00; H, 4.54; N, 8.31; S, 19.31. Found (%): C, 48.00; H, 4.54; N, 8.31; S, 19.31.
実施例 21-21 Example 21-21
N-(tert-ブチル)-4-[({5,6-ジメチル -8- [(5-フルォロペンチル)スルファニ ル]- 3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [ί, 4]チアジン - イリデン }ァ ミノ)スルホニル]ベンズアミド N- (tert-butyl) -4-[({5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c ] [ί, 4] thiazine-ylidene} amino) sulfonyl] benzamide
実施例 21-20で得られた 4-[({5, 6-ジメチル -3-ォキソ -8 - [(5-フルォロペンチ ル)スルファニル ]-2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデ ン}ァミノ)スルホニル]安息香酸 (150 mg, 0.300讓 ol) と tert -プチルァミン (0.0357 ml, 0.330匪 ol) の N, N-ジメチルホルムアミド (5.00 ml) 溶液に、 シ ァノりん酸ジェチル (0.0534 ml, 0.330腿 ol) 続いてトリェチルァミン (0.0460 ml, 0.330mmol) を加え、 室温で 16時間かき混ぜた。 反応混合物に 1N塩酸を加え て酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去 して題記化合物 (73.1mg, 44¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチル エーテルから再結晶して融点 169- 171°Cの暗赤色結晶を得た。 4-[({5,6-dimethyl-3-oxo-8-[(5-fluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, tric] obtained in Example 21-20 ] N, N-dimethylformamide (5.00 ml) of [1,4] thiazine-1-ylidene} amino) sulfonyl] benzoic acid (150 mg, 0.300 benzylol) and tert-butylylamine (0.0357 ml, 0.330 dandelionol) To the solution was added getyl cyanophosphate (0.0534 ml, 0.330 tmol) followed by triethylamine (0.0460 ml, 0.330 mmol), and the mixture was stirred at room temperature for 16 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The obtained extract was washed with water, dried (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (73.1mg, 44¾) as a dark red crystals. Ethyl acetate-Jetil Recrystallization from ether gave dark red crystals, mp 169-171 ° C.
IR (KBr) リ: 2969, 2936, 1742, 1661, 1615, 1559 cm"1. IR (KBr): 2969, 2936, 1742, 1661, 1615, 1559 cm " 1 .
'H-NMR (CDC13) : δ 1.47 (9Η, s), 1.45-1.62 (2H, m), 1.62-1.82 (4H, m), 1.79 (3H, d, J=0.9 Hz), 2.27 (3H, d, J=1.2 Hz), 2.96 (2H, t, =Ί Hz), 4.45 (2H, dt, J=47.1, 5.7 Hz), 5.96 (1H, bs), 7.80 (2H, d, J=8.4Hz), 8.00 (2H, d, J=8.7 Hz), 9.58 (1H, bs). 'H-NMR (CDC1 3) : δ 1.47 (9Η, s), 1.45-1.62 (2H, m), 1.62-1.82 (4H, m), 1.79 (3H, d, J = 0.9 Hz), 2.27 (3H , d, J = 1.2 Hz), 2.96 (2H, t, = Ί Hz), 4.45 (2H, dt, J = 47.1, 5.7 Hz), 5.96 (1H, bs), 7.80 (2H, d, J = 8.4) Hz), 8.00 (2H, d, J = 8.7 Hz), 9.58 (1H, bs).
元素分析値: C24H3IN404S3F 0.5EiOAcとして Elementary analysis: as C 24 H 3I N 4 0 4 S 3 F 0.5EiOAc
計算値 (¾) : C, 52.15; H, 5.89; K, 9.36; S, 16.07. Calculated value (¾): C, 52.15; H, 5.89; K, 9.36; S, 16.07.
実測値 (%) : C, 52.00; H, 5.63; N, 9.40; S, 16.04. Found (%): C, 52.00; H, 5.63; N, 9.40; S, 16.04.
実施例 21 - 22 Examples 21 to 22
9,10-ジメチル- 11_[(5_フルォロペンチル)スルファニル ]-4, 5-ジヒド口 _3H_[1,4]チアジノ [4, ,3, :3, 4]イミダゾ [5,卜 c] [1,2, 4]チアゼピン- 7 -オン 2, 2-ジォキシド 9,10-dimethyl-11 _ [(5_fluoropentyl) sulfanyl] -4,5-dihydrogen _3H_ [1,4] thiazino [4,, 3,: 3,4] imidazo [5, tric] [1, [2,4] thiazepine-7-one 2,2-dioxide
実施例 7- 176で得られた 3 -ク口 D-N-{5, 6 -ジメチル- 3-ォキソ -8- [(5-フルォロ ペンチル)スルファニル ]-2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィ リデン }-卜プロパンスルホンアミド (145 mg, 0.317 mmol) と炭酸カリウム (65.7 mg, 0.476 mmol) の N, N-ジメチルホルムアミド (5.00 ml) 溶液を、 室温で 22時 間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水 洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (28.8mg, 22%) を暗 赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 109-110°Cの暗赤色結晶を得た。 3-mouth DN- {5,6-dimethyl-3-oxo-8-[(5-fluoropentyl) sulfanyl] -2,3-dihydro-1H-imidazo obtained in Example 7-176 [5, 1-c] [1,4] Thiazine-1-ylidene} -topropanesulfonamide (145 mg, 0.317 mmol) and potassium carbonate (65.7 mg, 0.476 mmol) in N, N-dimethylformamide (5.00 ml) Was stirred at room temperature for 22 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was water washed, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (28.8 mg, 22%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 109-110 ° C.
IR (KBr) リ: 2938, 1738, 1663, 1601, 1557 cm"1. IR (KBr): 2938, 1738, 1663, 1601, 1557 cm " 1 .
Ή一 NMR (CDC13) : δ 1.46-1.70 (2Η, m), 1.70-1.88 (4H, in), 1.83 (3H, s), 2.20?2.35 (2H, m), 2.30 (3H, s), 2.99 (2H, t, J-7.8 Hz), 3.63 (2H, t, J=7.0 Hz), 4.16-4.28 (2H, m), 4.46 (2H, dt, J=47.2, 6.0 Hz). Ή one NMR (CDC1 3):? Δ 1.46-1.70 (2Η, m), 1.70-1.88 (4H, in), 1.83 (3H, s), 2.20 2.35 (2H, m), 2.30 (3H, s), 2.99 (2H, t, J-7.8 Hz), 3.63 (2H, t, J = 7.0 Hz), 4.16-4.28 (2H, m), 4.46 (2H, dt, J = 47.2, 6.0 Hz).
元素分析値: C16H22N303S3Fとして Elementary analysis: as C 16 H 22 N 3 0 3 S 3 F
計算値 (¾) : C, 45.80; H, 5.29; N, 10.02; S, 22.91. Calculated value (¾): C, 45.80; H, 5.29; N, 10.02; S, 22.91.
実測値 (%) : C, 45.61; H, 5.07; N, 9.66; S, 23.07. Found (%): C, 45.61; H, 5.07; N, 9.66; S, 23.07.
実施例 21- 23 4-ク口口- N - {4 - [({5, 6_ジメチル- 3 -ォキソ -8- [(5, 5, 6, 6, 6-ペン夕フルォ口へキ シル)スルファニル] - 2, 3-ジヒド口- 1H-ィミダゾ [5, 1-c] [1, 4]チアジン-卜ィリデ ン}ァミノ)スルホニル]フエ二ル}ブタンアミド Examples 21-23 4- ク 口 口-N-{4-[({5,6_dimethyl-3-oxo-8-[(5,5,6,6,6,6-pentylfluorohexyl) sulfanyl] -2 , 3-Dihydrido-1H-imidazo [5,1-c] [1,4] thiazine-trilidene} amino) sulfonyl] phenyl} butanamide
実施例 2卜 3 で得られた化合物の p-トルエンスルホン酸塩(295 mg, 0.405 fflinol)の THF (6 ml)懸濁液に室温で攪拌しながらピリジン(0.080 ml, 0.988 mmol) を加え、 ついで塩化 4-クロロブチリル (67 nig, 0.474 mmol)を加え、 さらに 2時 間攪拌した。 反応液にトリェチルァミン(0.055 ml,' 0.395腿 ol) を加え、 さらに 1時間攪拌した。 反応液に MAP (10 rag, 0.082匪 ol) を加え、 さらに 3時間攪拌 した。 反応液に同量の卜リエチルァミンおよび塩化 4-クロロブチリルを加え、 さ らに 20分間攪拌した。 反応液を酢酸ェチル (30ml)で希釈し、 0.1N塩酸、 水およ び飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥、 濃縮し、 残渣にジェチルエーテ ルを加えた。析出物をジェチルエーテルで洗浄後、乾燥して表題化合物 (227 mg, 0.343 mmol, 84.8%)を紫色粉末として得た。 Example 2 To a suspension of the compound obtained in Part 3 in p-toluenesulfonic acid salt (295 mg, 0.405 fflinol) in THF (6 ml) was added pyridine (0.080 ml, 0.988 mmol) with stirring at room temperature. Then, 4-chlorobutyryl chloride (67 nig, 0.474 mmol) was added, and the mixture was further stirred for 2 hours. Triethylamine (0.055 ml, '0.395 tmol) was added to the reaction solution, and the mixture was further stirred for 1 hour. MAP (10 rag, 0.082 bandol) was added to the reaction solution, and the mixture was further stirred for 3 hours. The same amount of triethylamine and 4-chlorobutyryl chloride were added to the reaction solution, and the mixture was further stirred for 20 minutes. The reaction solution was diluted with ethyl acetate (30 ml), washed with 0.1N hydrochloric acid, water and saturated saline, dried over sodium sulfate and concentrated, and getyl ether was added to the residue. The precipitate was washed with getyl ether and dried to give the title compound (227 mg, 0.343 mmol, 84.8%) as a purple powder.
Ή— NMR (300MHz, CDC13) : 51.74 (4H, m), 1.80 (3H, brs), 2.05 (2H, m), 2.21 (2H, quint, J= 6.5 Hz), 2.27 (3H, s), 2.61 (2H, t, J= 7.1 Hz), 2.96 (2H, t, J= 6.9 Hz), 3.67 (2H, t, J= 6.0 Hz), 7.52 (1H, brs) , 7.64 (2H, d, J= 8.7 Hz), 7.90 (2H, d, J= 9.0 Hz), 9.62 (1H, brs). Ή- NMR (300MHz, CDC1 3) : 51.74 (4H, m), 1.80 (3H, brs), 2.05 (2H, m), 2.21 (2H, quint, J = 6.5 Hz), 2.27 (3H, s), 2.61 (2H, t, J = 7.1 Hz), 2.96 (2H, t, J = 6.9 Hz), 3.67 (2H, t, J = 6.0 Hz), 7.52 (1H, brs), 7.64 (2H, d, J = 8.7 Hz), 7.90 (2H, d, J = 9.0 Hz), 9.62 (1H, brs).
元素分析値: C24H26N404S3C1F5として計算値: C, 43.60; H, 3.96; N, 8.47; S, 14.55; CI, 5.36; F, 14.37 (%) 、 実測値: C, 43.60; H, 3.96; N, 8.47; S, 14.55; CI, 5.36; F, 14.37 (%) . . ' mp. 137-1411: Elemental analysis: C 24 H 26 N 4 0 4 S 3 C1F 5 Calculated: C, 43.60; H, 3.96 ; N, 8.47; S, 14.55; CI, 5.36; F, 14.37 (%), Found: C, 43.60; H, 3.96; N, 8.47; S, 14.55; CI, 5.36; F, 14.37 (%).. 'Mp. 137-1411:
実施例 21-24 Example 21-24
N-{5, 6-ジメチル -3-ォキソ - 8_[ (5, 5, 6, 6, 6 -ペンタフルォ口へキシル)スルファニ ル] -2, 3-ジヒドロ- 1H-ィミダゾ [5, -c] [1 , ]チアジン-トイリデン } - 4- (1-ピロリ 実施例 21-3で得られた化合物 (206 mg, 0.370讓 ol)および 2, 5-ジメトキシ テトラヒドロフラン(0.245 ml, 1.85讓 ol)のァセトニトリル一 THF (1:18ml) 懸 濁液に水素化シァノホウ素ナトリウム (36.7 mg, 0.555 mmol)を加え、 さらに 23 時間攪拌した。 この間反応液に同量の水素化シァノホウ素ナトリウムを 5回およ び 2, 5 -ジメトキシテトラヒドロフラン(0.245 ml, 1.85 mmol)を加えた。 反応液 を酢酸ェチル -THF (2:1, 40 ml)で希釈し、 0.1N塩酸、 10%炭酸水素ナトリウム、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣を 酢酸ェチルで希釈した。 析出物を除去して酢酸ェチルおよびジェチルェ一テルで 洗浄した。 母液および洗液を濃縮し、 メタノールで希釈した。 析出物をメタノ一 ルで洗浄後、乾燥して表題化合物 (51mg, 0.058腿 ol)を赤褐色粉末として得た。 ¾-NMR (300MHz, CDC13) : 61.14: (4H, m), 1.78 (3H, s), 2.03 (4H, m), 2.05 (2H, m), 2.25 (3H, s), 2.61 (2H, t, J= 7.1 Hz), 2.94 (2H, t, J= 6.6 Hz), 3.33 (4H, brt, J= 6.6 Hz), 6.52 (2H, d, J= 8.7 Hz), 7.75 (2H, d, J= 8.7 Hz), 9.63 (1H, brs). N- {5,6-Dimethyl-3-oxo-8 _ [(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, -c] [1,] thiazine-toylidene} -4- (1-pyrroli) Acetonitrile of the compound obtained in Example 21-3 (206 mg, 0.370 alcohol) and 2,5-dimethoxytetrahydrofuran (0.245 ml, 1.85 alcohol) Sodium cyanoborohydride (36.7 mg, 0.555 mmol) was added to the suspension of THF (1:18 ml), and the mixture was stirred for another 23 hours, during which the same amount of sodium cyanoborohydride was added to the suspension five times. And 2,5-dimethoxytetrahydrofuran (0.245 ml, 1.85 mmol) were added. The reaction solution was diluted with ethyl acetate-THF (2: 1, 40 ml), washed with 0.1N hydrochloric acid, 10% sodium bicarbonate, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with ethyl acetate. The precipitate was removed and washed with ethyl acetate and ethyl acetate. The mother liquor and washings were concentrated and diluted with methanol. The precipitate was washed with methanol, and dried to give the title compound (51 mg, 0.058 mol) as a red-brown powder. ¾-NMR (300MHz, CDC1 3 ): 61.14: (4H, m), 1.78 (3H, s), 2.03 (4H, m), 2.05 (2H, m), 2.25 (3H, s), 2.61 (2H, t, J = 7.1 Hz), 2.94 (2H, t, J = 6.6 Hz), 3.33 (4H, brt, J = 6.6 Hz), 6.52 (2H, d, J = 8.7 Hz), 7.75 (2H, d, J = 8.7 Hz), 9.63 (1H, brs).
元素分析値: C24H27N403S3F5として計算値: C, 47.20; H, 4.46; N, 9.17; S, 15.75; F, 15.56 (%) 、 実測値: C, 47.23; H, 4.19; N, 9.15; S, 15.97; F, 15.53 (%) . mp. 190.0-191.5 Elemental analysis: C 24 H 27 N 4 0 3 S 3 F 5 Calculated: C, 47.20; H, 4.46 ; N, 9.17; S, 15.75; F, 15.56 (%), Found: C, 47.23; H, 4.19; N, 9.15; S, 15.97; F, 15.53 (%). Mp.190.0-191.5
実施例 21-25 Example 21-25
N-{5, 6 -ジメチル -3-ォキソ -8- [ (5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 2,3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1,4]チアジン- 1-ィリデン }_4- (1H-ピロ 実施例 2卜 3で得られた化合物の p-トルエンスルホン酸塩 (390 mg, 0.535匪 ol) および 2, 5 -ジメトキシテトラヒドロフラン(0.707 ml, 5.35龍 ol)の THF (10 ml) 懸濁液を室温で 30分間、 50°Cで 10分間攪拌した。 反応液を酢酸ェチル(40 ml) で希釈し、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 濃縮乾固した。 残渣をメタノ一^^で希釈して生成した析出物をメタノールで洗浄 後、 乾燥して表題化合物(Π5 mg, 0.288匪 ol, 53.9»を赤紫色粉末として得た。 Ή-NMR (300MHz, CDC13) : 51.75 (4H, m), 1.80 (3H, s), 2.06 (2H, m), 2.28 (3H, s), 2.98 (2H, t, J= 7.2 Hz), 6.40 (2H, t, J= 2.1 Hz), 7.14 (2H, t, J= 2.3 Hz), 7.49 (2H, d, J= 8.7 Hz), 8.02 (2H, d, J= 8.7 Hz), 9.67 (1H, brs). 元素分析値 CMH23N403S3F5として計算値: C, 47.52; H, 3.82; , 9.24; S, 15.86; F, 15.66 (%) 、 実測値: C, 47.32; H, 4.00; N, 9.18; S, 15.89; F, 15.40 ( ) . mp. 183.0-187.0°C 実施例 22 - 1 N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl]-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene} _4- (1H-pyro p-toluenesulfonate (390 mg, 0.535 methylol) of the compound obtained in Example 3 and 2,5-dimethoxy A suspension of tetrahydrofuran (0.707 ml, 5.35 ml) in THF (10 ml) was stirred at room temperature for 30 minutes and at 50 ° C. for 10 minutes The reaction solution was diluted with ethyl acetate (40 ml), and 0.1N hydrochloric acid was added. The residue was diluted with methanoic acid, washed with methanol, dried, and dried to give the title compound (Π5 mg, 0.288 mg). . negation ol, 53.9 »was obtained as a red-purple powder Ή-NMR (300MHz, CDC1 3 ): 51.75 (4H, m), 1.80 (3H, s), 2.06 (2H, m), 2.28 (3H, s) , 2.98 (2H, t, J = 7.2 Hz), 6.40 (2H, t, J = 2.1 Hz), 7.14 (2H, t, J = 2.3 Hz) Hz), 7.49 (2H, d, J = 8.7 Hz), 8.02 (2H, d, J = 8.7 Hz), 9.67 (1H, brs). Elemental analysis value C M H 23 N 4 0 3 S 3 F 5 Calculated: C, 47.52; H, 3.82;, 9.24; S, 15.86; F, 15.66 (%), found: C, 47.32; H, 4.00; N, 9.18; S, 15.89; F, 15.40 (). mp.183.0-187.0 ° C Example 22-1
5-({5, 6 -ジメチル -卜 [4- (メチルべンゾィル)ィミノ] -3 -ォキソ -2, 3-ジヒドロ - 1H -イミダゾ [5,卜 c] [1,4]チアジン- 8-ィル }スルファニル)吉草酸 5-({5,6-Dimethyl-tri [4- (methylbenzoyl) imino] -3-oxo-2,3-dihydro-1H-imidazo [5, tri-c] [1,4] thiazine-8 Yl} sulfanyl) valeric acid
実施例 8-5 で得られたェチル 5 -({5, 6-ジメチル-卜 [4- (メチルペンゾィル)ィ ミノ]- 3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5,卜 c] [1,4]チアジン -8-ィル }スル ファニル)ペンタノェ一ト (400 mg, 0.845 mmol) のエタノール (10.0 ml) 及び テトラヒドロフラン (10.0 ml) 溶液に、 1N水酸化ナトリウム水溶液 (2.53 ml) を加えた。反応混合物を室温で 5.5時間かき混ぜた後、溶媒を減圧下に留去した。 残渣に IN塩酸を加えて、 酢酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (314 mg, 84¾) を合成した。 酢酸ェチル -ジェチルエーテルから再結晶して融点 239- 241°Cの暗赤色結晶を得た。 Ethyl 5-({5,6-dimethyl- [4- (methylpentzyl) imino] -3-oxo-2,3-dihydro-1H-imidazo [5, tric] obtained in Example 8-5 To a solution of [1,4] thiazine-8-yl} sulfanyl) pentanoate (400 mg, 0.845 mmol) in ethanol (10.0 ml) and tetrahydrofuran (10.0 ml) was added a 1N aqueous sodium hydroxide solution (2.53 ml). added. After stirring the reaction mixture at room temperature for 5.5 hours, the solvent was distilled off under reduced pressure. IN hydrochloric acid was added to the residue, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) After the solvent was distilled off under reduced pressure to give the title compound (314 mg, 84¾) was synthesized. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 239-241 ° C.
IR (KBr) V: 3500-2700, 1732, 1719, 1653, 1597, 1537, 1505 cm—1. IR (KBr) V: 3500-2700, 1732, 1719, 1653, 1597, 1537, 1505 cm- 1 .
Ή— NMR (CDC13) : δ 1.27 (3Η, d, J=7.2 Hz), 1.76-1.88 (4H, 111), 1.84 (3H, s), 2.34 (3H, d, J=1.2 Hz), 2.36-2.44 (2H, m), 2.41 (3H, s), 3.02-3.12 (2H, m), 4.15 (2H, q, J=7.2 Hz), 7.25 (2H, d, J=7.2 Hz), 8.22 (2H, d, J=8.1 Hz), 11.08 Ή- NMR (CDC1 3): δ 1.27 (3Η, d, J = 7.2 Hz), 1.76-1.88 (4H, 111), 1.84 (3H, s), 2.34 (3H, d, J = 1.2 Hz), 2.36 -2.44 (2H, m), 2.41 (3H, s), 3.02-3.12 (2H, m), 4.15 (2H, q, J = 7.2 Hz), 7.25 (2H, d, J = 7.2 Hz), 8.22 ( 2H, d, J = 8.1 Hz), 11.08
(m, bs). (m, bs).
元素分析値: C21 N304S20.3H20として Elementary analysis: as C 21 N 3 0 4 S 2 0.3H 2 0
計算値 : C, 55.93; H, 5.27; N, 9.32; S, 14.22. Calculated: C, 55.93; H, 5.27; N, 9.32; S, 14.22.
実測値 (¾) : C, 55.80; H, 4.97; N, 9.26; S, 14.29. Found (¾): C, 55.80; H, 4.97; N, 9.26; S, 14.29.
以下に実施例 22-1 で合成した化合物および同様にして合成した化合物の一覧 を示す。 The following is a list of compounds synthesized in Example 22-1 and compounds synthesized in the same manner.
〔表 1 1 6〕 (Table 1 16)
摹蒈ぇ靡弒 «§2 実施例 22 - 2 摹 蒈 ぇ § 弒 «§2 Example 22-2
N -(5, 6-ジメチル -8- { [5- (へキシルァミノ) -5-ォキソペンチル]スルファ二ル} -3- ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,1 - c] [1, 4]チアジン- 8-イリデン) -4-メチル ベンズアミド N- (5,6-dimethyl-8-{[5- (hexylamino) -5-oxopentyl] sulfanyl} -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [ 1,4] thiazine-8-ylidene) -4-methylbenzamide
実施例 22-1 で得られた 5- ({5, 6-ジメチル- 1- [4- (メチルペンゾィル)イミ ノ]- 3-ォキソ -2, 3-ジヒドロ- 1H -イミダゾ [5, 1-c] [1, 4]チアジン- 8-ィル }スルフ ァニル)吉草酸(100 mg, 0.224廳 ol)と卜へキシルァミン(0.0327 ml, 0.242匪 ol) の N,N-ジメチルホルムアミド (2.00ml)溶液に、 シァノりん酸ジェチル(0.0399 ml, 0.247扁 ol).続いてトリェチルァミン (0.0344 ml, 0.247 uimol) を加えた。 反応混合物を室温で 21時間かき混ぜた後、水を加えて酢酸ェチルで抽出した。抽 出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(24.0mg, 20%) を合成した。酢酸ェチル -ジェチルェ一テルから再結晶して融点 128- 130°Cの暗赤 色結晶を得た。 5-({5,6-dimethyl-1- [4- (methylbenzoyl) imino] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] obtained in Example 22-1 ] [1,4] thiazin-8-yl} sulfanyl) valeric acid (100 mg, 0.224 ol) and trihexylamine (0.0327 ml, 0.242 marl ol) in N, N-dimethylformamide (2.00 ml) To this was added getyl cyanophosphate (0.0399 ml, 0.247 mmol) and then triethylamine (0.0344 ml, 0.247 uimol). After stirring the reaction mixture at room temperature for 21 hours, water was added, and the mixture was extracted with ethyl acetate. Washed with water extract exudates, dried (MgSO 4), was synthesized solvent was distilled off under reduced pressure the title compound (24.0mg, 20%). The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 128-130 ° C.
IR (KBr)レ : 3268, 2955, 2928, 2861, 1717, 1607, 1549, 1505 cm"1. IR (KBr): 3268, 2955, 2928, 2861, 1717, 1607, 1549, 1505 cm " 1 .
Ή-NMR (CDC13) : 50.873 (3H, t, J=6.9 Hz), 1.24-1.36 (6H, m), 1.42-1.54 (2H, m), 1.78-1.90 (4H, m), 1.83 (3H, d, J=1.2 Hz), 2.20-2.28 (2H, m), 2.33 (3H, d, J=0.9Hz), 2.41 (3H, s), 3.04-3.12 (2H, m), 3.23 (2H, q, J=6.3 Hz), 7.25 Ή-NMR (CDC1 3): 50.873 (3H, t, J = 6.9 Hz), 1.24-1.36 (6H, m), 1.42-1.54 (2H, m), 1.78-1.90 (4H, m), 1.83 (3H , d, J = 1.2 Hz), 2.20-2.28 (2H, m), 2.33 (3H, d, J = 0.9Hz), 2.41 (3H, s), 3.04-3.12 (2H, m), 3.23 (2H, q, J = 6.3 Hz), 7.25
(2H, d, J=8.0 Hz), 8.22 (2H, d, J=8. Hz), 11.09 (1H, bs) · (2H, d, J = 8.0 Hz), 8.22 (2H, d, J = 8 Hz), 11.09 (1H, bs)
元素分析値: C27H36N403S2 ·0.3Η20として Elementary analysis: as C 27 H 36 N 4 0 3 S 2 · 0.3Η 2 0
計算値 (%) : C, 60.71; Η, 6.91; Ν, 10.49; S, 12.01. Calculated value (%): C, 60.71; Η, 6.91; Ν, 10.49; S, 12.01.
実測値 (¾) : C, 60.57; Η, 6.69; Ν, 10.48; S, 12.31. Found (¾): C, 60.57; Η, 6.69; Ν, 10.48; S, 12.31.
実施例 22 - 3Example 22-3
- (4-ブロモフエニル) -N' - {8- [ (5 -フルォ口ペンチル) スルファニル] -5, 6 -ジメ チル -3-ォキソ -2, 3-ジヒドロ- 1H-イミダゾ [5,卜 c] [1,4]チアジン- 1-ィリデ ン}- Ν-メチルゥレア -(4-Bromophenyl) -N '-{8- [(5-fluoropentyl) sulfanyl] -5, 6-dimethyl-3-oxo-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1-ylidene}-{-methyl} rea
実施例 8-12で得た化合物(106 mg, 0.206醒 ol)の THF (3.0 ml) 懸濁液に水 素化ナトリウム(純度 60% 油性, 20.6 mg, 0.516腿 ol) を室温で加え、 10分間 攪拌後、 反応液にヨウ化メチル (0.017 ml, 0.268腿 ol)の THF (0.17 ml)溶液を 加え、 24時間攪拌した。 実施例 8-12で得た化合物(136 mg)から同様にして得た 反応液をあわせ、 酢酸ェチル(30 ml)で希釈し、 0.1規定塩酸、 水および飽和食塩 水で洗浄後、 硫酸ナトリウムで乾燥した。 濃縮乾固後、 残渣をメタノールで希釈 した。 析出物を濾取し、 シリカゲルクロマトグラフィー (溶出溶媒、 酢酸ェチル 一へキサン、 2 :8- 4:6)に付し、酢酸ェチルーへキサン(4: 6) の画分を濃縮乾固後、 酢酸ェチルで希釈した。 析出物を濾取し、 酢酸ェチルおよびジェチルエーテルで 洗浄後、乾燥して表題化合物(127 mg, 0.241腿 oI, 51.1»を赤色粉末として得た。 — NMR (200MHz, CDC13): δ 1.50 (2H, m), 1.74 (3H, s), 1.75 (2H, m), 1.80 (2H, m), 2.25 (3H, s), 2.72/3.03 (2H, brs/m), 3.31/3.56 (3H, s/brs), 4.48 (2H, dt, J= 47.4, 5.7 Hz), 7.21 (2H, brd, J= 7.8 Hz), 7.45 (2H, brd, J= 8.4 Hz), 10.60/10.63 (1H, brs). To a suspension of the compound obtained in Example 8-12 (106 mg, 0.206 ol) in THF (3.0 ml) was added sodium hydride (purity 60% oily, 20.6 mg, 0.516 ol) at room temperature. After stirring for 1 minute, a solution of methyl iodide (0.017 ml, 0.268 mol) in THF (0.17 ml) was added to the reaction solution, and the mixture was stirred for 24 hours. Obtained similarly from compound (136 mg) obtained in Example 8-12 The reaction solutions were combined, diluted with ethyl acetate (30 ml), washed with 0.1 N hydrochloric acid, water and saturated saline, and dried over sodium sulfate. After concentration to dryness, the residue was diluted with methanol. The precipitate was collected by filtration, subjected to silica gel chromatography (elution solvent, ethyl acetate-hexane, 2: 8-4: 6), and the ethyl acetate-hexane (4: 6) fraction was concentrated to dryness. Diluted with ethyl acetate. The precipitates are collected by filtration, washed with acetic acid Echiru and Jefferies chill ether and dried to give the title compound (127 mg, 0.241 thigh OI, 51.1 »was obtained as a red powder - NMR (200MHz, CDC1 3) :. Δ 1.50 (2H, m), 1.74 (3H, s), 1.75 (2H, m), 1.80 (2H, m), 2.25 (3H, s), 2.72 / 3.03 (2H, brs / m), 3.31 / 3.56 (3H , s / brs), 4.48 (2H, dt, J = 47.4, 5.7 Hz), 7.21 (2H, brd, J = 7.8 Hz), 7.45 (2H, brd, J = 8.4 Hz), 10.60 / 10.63 (1H, brs).
元素分析値: C21H24N402S2BrF として計算値: C, 47.82; H, 4.59; N, 10.62; S, 12.16; Br, 15.15; F, 3.60 (%)、 実測値: C, 47.84; H, 4.65; N, 10.51; S, 12.03; Br, 14.93; F, 3.61 ( ) . Elemental analysis: C 21 H 24 N 4 0 2 S 2 BrF Calculated: C, 47.82; H, 4.59 ; N, 10.62; S, 12.16; Br, 15.15; F, 3.60 (%), Found: C , 47.84; H, 4.65; N, 10.51; S, 12.03; Br, 14.93; F, 3.61 ().
mp. 153.0-155.5°C mp. 153.0-155.5 ° C
実施例 23 Example 23
N-{5, 6-ジメチル -3-ォキソ _8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)スルファニ ル]- 2, 3-ジヒドロ- lri-イミダゾ [5, 1-c] [1, 4]チアジン - 1 -ィリデン }-P,P-ジフエ ニルホスフィニックアミ.ド N- {5,6-dimethyl-3-oxo_8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-lri-imidazo [5,1-c ] [1,4] thiazine-1-ylidene} -P, P-diphenylphosphinic amine
水素化ナトリウム(29.9 mg, 0.747匪 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6- 6で得られた 1-ァミノ- 5, 6-ジメチル- 8_[(5, 5, 6,6, 6- ペン夕フルォ口へキシル)スルファニル ]-3H-イミダゾ [5,卜 c] [1,4]チアジン- 3- オン (150 mg, 0.374 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これにジフエ二ルホスフィニック クロリド(108 mg, 0.448 腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 17時間かき 混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (170 mg, 76%) を暗赤色結 晶として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 214- 216°Cの暗 赤色結晶を得た。 To a suspension of sodium hydride (29.9 mg, 0.747 ol) (60% oily) in tetrahydrofuran (1.00 ml) was added 1-amino-5,6-dimethyl-8 _ [(obtained in Example 6-6. 5,5,6,6,6-pentafluorohexyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (150 mg, 0.374 mmol) in tetrahydrofuran (1.00 ml), and the mixture was stirred under ice-cooling for 5 minutes. To this was added a solution of diphenylphosphinic chloride (108 mg, 0.448 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 17 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, Drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (170 mg, 76%) and the obtained as dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 214-216 ° C.
IR (KBr) V : 2917, 2685, 1734, 1672, 1640, 1574 cm—1. lH-NMR (CDCI3) : (51.78 (3H, d, J=0.6 Hz), 1.74-1.92 (4H, m), 2.00-2.20 (2H, m), 2.23 (3H, d, J=0.9 Hz), 3.05 (2H, t, J=6.9 Hz), 7.37-7.50 (6H, m), 7.92 (4H, ddd, J=12.0, 7.8, 1.5 Hz), 9.85 (1H, bs). IR (KBr) V: 2917, 2685, 1734, 1672, 1640, 1574 cm— 1 . l H-NMR (CDCI3): (51.78 (3H, d, J = 0.6 Hz), 1.74-1.92 (4H, m), 2.00-2.20 (2H, m), 2.23 (3H, d, J = 0.9 Hz) , 3.05 (2H, t, J = 6.9 Hz), 7.37-7.50 (6H, m), 7.92 (4H, ddd, J = 12.0, 7.8, 1.5 Hz), 9.85 (1H, bs).
元素分析値: C26H25N302S2F5Pとして Elemental analysis: as C 26 H 25 N 3 0 2 S 2 F 5 P
計算値 (¾) : C, 51.91; H, 4.19; N, 6.98; S, 10.66. Calculated (¾): C, 51.91; H, 4.19; N, 6.98; S, 10.66.
実測値 (%) : C, 51.92; H, 4.20; N, 7.03; S, 10.45. Found (%): C, 51.92; H, 4.20; N, 7.03; S, 10.45.
実施例 24 Example 24
ジフエニル 5, 6 -ジメチル- 3-ォキソ -8- [(5, 5, 6, 6, 6-ペンタフルォ口へキシル)ス ルファニル ]-2,3-ジヒドロ- 1H -イミダゾ [5, c] [1,4]チァジン-1_ィリデンアミ ドホスフェート Diphenyl 5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, c] [1 , 4] Thazine-1_ylideneamide phosphate
水素化ナトリウム(29.9 nig, 0.747mmol) (60 油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-6で得られた 1 -アミノ- 5, 6-ジメチル- 8- [(5, 5, 6, 6,6 - ペン夕フルォ口へキシル)スルファニル ]-3H-イミダゾ [5,卜 c] [1, 4]チアジン- 3 - オン (150 mg, 0.374腿 ol) のテトラヒドロフラン (1.00 mi) 溶液を加え、 氷冷 下 5分間かき混ぜた。これにジフエ二ル クロ口ホスフェート(120mg, 0.448應 ol) のテトラヒドロフラン(1.00ml)溶液を加え、さらに室温で 30分間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04) 後;溶媒を減圧下に留去して題記化合物(185 mg, 78¾)を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルから再結晶して融点 100-102°Cの暗赤色結晶を得 た。 To a suspension of sodium hydride (29.9 nig, 0.747 mmol) (60 oily) in tetrahydrofuran (1.00 ml) was added 1-amino-5,6-dimethyl-8-[(5 , 5,6,6,6-Penyu fluor-hexyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (150 mg, 0.374 tmol) of tetrahydrofuran (1.00 mi) The solution was added, and the mixture was stirred for 5 minutes under ice cooling. To this was added a solution of diphenyl chloroformate phosphate (120 mg, 0.448 ol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 30 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4) after; solvent was evaporated under reduced pressure to give the title compound (185 mg, 78¾) was obtained as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 100-102 ° C.
IR (KBr)ソ : 3071, 2942, 2735, 1746, 1667, 1635, 1572 cm—1. IR (KBr): 3071, 2942, 2735, 1746, 1667, 1635, 1572 cm— 1 .
賺 (CDC13) : «51.68-1.82 (4H, m), 1.77 (3H, d, J=1.2 Hz), 1.92-2.18 (2H, m), 2.25 (3H, d, J=1.0 Hz), 2.94 (2H, t, J=7.0 Hz), 7.10-7.37 (10H, m), 9.60 (1H, bs). (CDC1 3 ): «51.68-1.82 (4H, m), 1.77 (3H, d, J = 1.2 Hz), 1.92-2.18 (2H, m), 2.25 (3H, d, J = 1.0 Hz), 2.94 (2H, t, J = 7.0 Hz), 7.10-7.37 (10H, m), 9.60 (1H, bs).
元素分析値: C26H25N304S2F5Pとして Elementary analysis: as C 26 H 25 N 3 0 4 S 2 F 5 P
計算値 (%) : C, 49.29; H, 3.98; N, 6.63; S, 10.12. Calculated value (%): C, 49.29; H, 3.98; N, 6.63; S, 10.12.
実測値 (¾) : C, 49.20; H, 3.85; N, 6.64; S, 9.97. Found (¾): C, 49.20; H, 3.85; N, 6.64; S, 9.97.
実施例 25 Example 25
ジェチル 5, 6-ジメチル- 3-ォキソ _8 - [(5, 5, 6, 6, 6-ぺ: ファニル] -2, 3-ジヒドロ- 1H-ィミダゾ [5, 1-c] [1,4]チアジン- 1 -ィリデンアミド ホスフエ一ト Getyl 5, 6-dimethyl-3-oxo _8-[(5, 5, 6, 6, 6- ぺ: Fanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidenamide phosphite
水素化ナトリウム(29.9 mg, 0.747腿 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-6で得られた 1-ァミノ- 5, 6 -ジメチル- 8-[(5, 5, 6, 6, 6- ペン夕フルォ口へキシル)スルファニル ]-3H-イミダゾ [5, 1-c] [1, 4]チアジン - 3- オン (150 mg, 0.374腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷 下 5 分間かき混ぜた。 これにジェチル クロ口ホスフェート (0.0835 ml, 0.561 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 19時間かき 混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (138 mg, 69%) を暗赤色結 晶として得た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 154- 156°Cの暗 赤色結晶を得た。 To a suspension of sodium hydride (29.9 mg, 0.747 tmol) (60% oil) in tetrahydrofuran (1.00 ml) was added 1-amino-5,6-dimethyl-8- [ (5,5,6,6,6-Pentafluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (150 mg, 0.374 tmol) A tetrahydrofuran (1.00 ml) solution was added, and the mixture was stirred for 5 minutes under ice cooling. To this was added a solution of getyl chloroformate (0.0835 ml, 0.561 mmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 19 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, Drying (MgSO 4), solvent was evaporated under reduced pressure to give the title compound (138 mg, 69%) and the obtained as dark red crystals. Recrystallization from ethyl acetate-ethyl ether gave dark red crystals with a melting point of 154-156 ° C.
IR (KBr)ソ : 2986, 1750, 1672, 1651, 1576 cm"1. IR (KBr): 2986, 1750, 1672, 1651, 1576 cm " 1 .
lH-NMR (CDC13) : d 1.34 (6H, d, J=6.9 Hz), 1.68-1.88 (4H, in), 1.79 (3H, d, J=1.2Hz), 1.98-2.16 (2H, m), 2.26 (3H, d, J=0.9 Hz), 4.00-4.15 (4H, m), 9.68 (1H, bs). lH-NMR (CDC1 3): d 1.34 (6H, d, J = 6.9 Hz), 1.68-1.88 (4H, in), 1.79 (3H, d, J = 1.2Hz), 1.98-2.16 (2H, m) , 2.26 (3H, d, J = 0.9 Hz), 4.00-4.15 (4H, m), 9.68 (1H, bs).
元素分析値: C18H25N304S2F5Pとして Elementary analysis: as C 18 H 25 N 3 0 4 S 2 F 5 P
計算値 (%) : C, 40.22; H, 4.69; N, 7.82; S, 11.93. Calculated (%): C, 40.22; H, 4.69; N, 7.82; S, 11.93.
実測値 (%) : C, 40.09; H, 4.79; N, 7.58; S, 11.84. Found (%): C, 40.09; H, 4.79; N, 7.58; S, 11.84.
実施例 26 Example 26
N - {5, 6 -ジメチル -3-ォキソ -8-[(5,5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル] - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1-ィリデン }- P-メチル- P - フエニルホスフィニック アミド N- {5,6-dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3-dihydro-1H-imidazo [5, 1-c] [1,4] thiazine-1-ylidene} -P-methyl-P-phenylphosphinic amide
水素化ナトリウム(29.9 mg, 0.747 mmol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-6で得られた 1_アミノ- 5, 6-ジメチル 8- [(5, 5,6, 6, 6- ペンタフルォ口へキシル)スルファニル ]-3H-イミダゾ [5,卜 c] [1, 4]チアジン - 3- オン (150 mg, 0.574 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これにメチルフエニルホスフィニック クロリド (80.7mg, 0.448讓 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さらに 30分間かき 混ぜた。反応混合物に IN塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾 燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (145 mg, 47%) を暗赤色結 晶として得た。酢酸ェチル-ジェチルェ一テルから再結晶して融点 188-190°Cの暗 赤色結晶を得た。 To a suspension of sodium hydride (29.9 mg, 0.747 mmol) (60% oil) in tetrahydrofuran (2.00 ml) was added 1_amino-5,6-dimethyl 8-[(5 , 5,6,6,6-Pentafluorohexyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (150 mg, 0.574 mmol) in tetrahydrofuran (1.00 ml) And stirred under ice cooling for 5 minutes. To this was added a solution of methylphenylphosphinic chloride (80.7 mg, 0.448 acetyl) in tetrahydrofuran (1.00 ml), and the mixture was stirred for another 30 minutes. mixed. IN hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, Drying (MgSO 4), solvent was obtained as a distilled off the dark red crystals of the title compound (145 mg, 47%) and under reduced pressure. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 188-190 ° C.
IR (KBr) レ : 3058, 2915, 2691, 1744, 1671, 1644, 1582 cm—1. IR (KBr): 3058, 2915, 2691, 1744, 1671, 1644, 1582 cm— 1 .
Ή-NMR (CDC13) : 6 1.78 (6H, s), 1.74-1.92 (4H, m), 1.94-2.20 (2H, m), 2.23 (3H, d, J=l.2 Hz), 2.30 (2H, t, J=6.6Hz), 7.40-7.53 (3H, m), 7.84-7.96 (2H, m), 9.84 (1H, bs). Ή-NMR (CDC1 3): 6 1.78 (6H, s), 1.74-1.92 (4H, m), 1.94-2.20 (2H, m), 2.23 (3H, d, J = l.2 Hz), 2.30 ( 2H, t, J = 6.6Hz), 7.40-7.53 (3H, m), 7.84-7.96 (2H, m), 9.84 (1H, bs).
元素分析値: C2iH23N302S2F5Pとして , Elemental analysis: C 2 iH 23 N 3 0 2 S 2 F 5 P,
計算値 (¾) : C, 46.75; Η, 4.30; Ν, 7.79; S, 11.89. Calculated value (¾): C, 46.75; Η, 4.30; Ν, 7.79; S, 11.89.
実測値 (¾) : C, 46.47; H, 4.20; N, 7.84; S, 11.82. Found (¾): C, 46.47; H, 4.20; N, 7.84; S, 11.82.
実施例 27 Example 27
N-{5, 6-ジメチル- 3-ォキソ - 8-[(5, 5, 6, 6, 6-ペン夕フルォ口へキシル)スルファニ ル] - 2, 3_ジヒドロ- 1H -ィミダゾ [5, 1-c] [1, 4]チアジン _1 -ィリデン }- 4_メチルベ ンゼンスルフィンアミド N- {5,6-Dimethyl-3-oxo-8-[(5,5,6,6,6-pentafluorohexyl) sulfanyl] -2,3_dihydro-1H-imidazo [5, 1-c] [1,4] thiazine_1-ylidene}-4_methylbenzenesulfinamide
水素化ナトリウム(39.9 mg, 0.996顧 ol) (60%油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6 - 6で得られた 1_アミノ- 5, 6-ジメチル 8- [(5, 5, 6,6,6- ペン夕フルォ口へキシル)スルファニル] - 3H-ィミダゾ [5, 1-c] [1, 4]チアジン -3 - オン (200 mg, 0.498腿 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷 下 5分間かき混ぜた。 これに参考例 47で得られた p_トルエンスルフィニル ク口 リド (80.7 mg, 0.448 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 さ らに 19時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。 抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(133 mg, 50¾) を暗赤色結晶として得た。 酢酸ェチルから再結晶して融点 147- 149°Cの暗赤色結 晶を得た。 To a suspension of sodium hydride (39.9 mg, 0.996 ethanol) (60% oil) in tetrahydrofuran (2.00 ml) was added 1_amino-5,6-dimethyl 8-[(obtained in Example 6-6. 5,5,6,6,6-pentylfluorohexyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine-3-one (200 mg, 0.498 tmol) of tetrahydrofuran (1.00 ml) solution was added and stirred for 5 minutes under ice-cooling. To this was added a solution of p_toluenesulfinyl chloride (80.7 mg, 0.448 mmol) obtained in Reference Example 47 in tetrahydrofuran (1.00 ml), and the mixture was further stirred for 19 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (133 mg, 50¾) as a dark red crystals. Recrystallization from ethyl acetate gave dark red crystals with a melting point of 147-149 ° C.
IR (KBr) V: 2934, 2689, 1755, 1684, 1597, 1198 cm"1. IR (KBr) V: 2934, 2689, 1755, 1684, 1597, 1198 cm " 1 .
Ή一 NMR (CDC13) : δ1.62?1.85 (4Η, m), 1.76 (3H, d, J=1.2 Hz), 1.92?2.11 (2H, in), 2.24 (3H, d, J=1.2 Hz), 2.36 (3HX0.51, s), 2.40 (3HX0.49, s), 3.00 (2H, t, J=7.8 Hz), 7.19 (2HX0.42, d, J=8.1 Hz)', 7.27 (2HX0.58, d, J=8.4 Hz), 7.61 (2HX0.58, d, J=8.4 Hz), 7.77 (2HX0.42, d, J=8.1 Hz), 8.20 (1H, bs). 元素分析値: C21H22N302S3F5 ·1· 2H20として Ή one NMR (CDC1 3):?? Δ1.62 1.85 (4Η, m), 1.76 (3H, d, J = 1.2 Hz), 1.92 2.11 (2H, in), 2.24 (3H, d, J = 1.2 Hz ), 2.36 (3HX0.51, s), 2.40 (3HX0.49, s), 3.00 (2H, t, J = 7.8 Hz), 7.19 (2HX0.42, d, J = 8.1 Hz) ', 7.27 (2HX0 .58, d, J = 8.4 Hz), 7.61 (2HX0.58, d, J = 8.4 Hz), 7.77 (2HX0.42, d, J = 8.1 Hz), 8.20 (1H, bs). Elemental analysis: C 21 H 22 N 3 0 2 S 3 F 5・ 1 ・ 2H 2 0
計算値 (%) : C, 44.94; H, 4.38; N, 7.49. Calculated value (%): C, 44.94; H, 4.38; N, 7.49.
実測値 (¾) : C, 44.96; H, 4.30; N, 7.56. Found (¾): C, 44.96; H, 4.30; N, 7.56.
実施例 28 Example 28
N-{5, 6-ジメチル -8- [ (5-フルォ口ぺンチル)スルファニル] -3 -ォキソ -2, 3-ジヒド ロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン- 1 -ィリデン }-P-メチル—P -フエニルホス フィニック アミド N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine -1 -ylidene} -P-methyl-P-phenylphosphinic amide
水素化ナトリウム(25.4 mg, 0.654匪 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5,6-ジメチル -8- [(5_フルォ 口ペンチル)スルファニル ]-3H-イミダゾ [5,1- c] [1, 4]チアジン- 3-オン (100 mg, 0.317醒 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにメチルフエニルホスフィニック クロリド (68.5 mg, 0.380腿 ol) のテトラヒドロフラン(1.00ml)溶液を加え、さらに室温で 40分間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (99.4 mg, 69%) を暗赤色結晶として得 た。酢酸ェチルージェチルエーテルから再結晶して融点 158_160°Cの暗赤色結晶を 得た。 A suspension of sodium hydride (25.4 mg, 0.654 ol) (60% oil) in tetrahydrofuran (1.00 ml) was added to the suspension of 1-amino-5,6-dimethyl-8- [obtained in Example 6-23. (5_Fluoro-pentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazin-3-one (100 mg, 0.317 mmol ol) in tetrahydrofuran (1.00 ml) and ice-cooled Stir for 5 minutes below. To this was added a solution of methylphenylphosphinic chloride (68.5 mg, 0.380 tmol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 40 minutes. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (99.4 mg, 69%) as a dark red crystals. Recrystallization from ethyl acetate-ethyl ether yielded dark red crystals with a melting point of 158-160 ° C.
赤外吸収スペクトル(IR) (KBr中) ソ : 2905, 2699, 1740, 1669, 1644, 1580 cm一1. ¾-NMR (CDC13) : δ 1.48-1.89 (6H, m), 1.78 (6H, s), 2.23 (3H, s), 2.99 (3H, s), 2.99 (2H, t, J=6.8 Hz), .4.47 (2H, dt, J=47.0, 5.8 Hz), 7.39-7.56 (3H, m), 7.84-7.98 (2H, m), 9.82 (1H, bs). Infrared absorption spectrum (IR) (in KBr) Seo:. 2905, 2699, 1740, 1669, 1644, 1580 cm one 1 ¾-NMR (CDC1 3) : δ 1.48-1.89 (6H, m), 1.78 (6H, s), 2.23 (3H, s), 2.99 (3H, s), 2.99 (2H, t, J = 6.8 Hz), .4.47 (2H, dt, J = 47.0, 5.8 Hz), 7.39-7.56 (3H, m), 7.84-7.98 (2H, m), 9.82 (1H, bs).
元素分析値: C2。 N302S2FPとして Elemental analysis: C 2. N 3 0 2 S 2 FP
計算値 (%) : C, 52.96; H, 5.56; N, 9.27; S, 14.14. Calculated value (%): C, 52.96; H, 5.56; N, 9.27; S, 14.14.
実測値 (%) : C, 52.68; H, 5.41; N, 9.36; S, 14.23. Found (%): C, 52.68; H, 5.41; N, 9.36; S, 14.23.
実施例 29 Example 29
ジェチル 5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2,3 - ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 1 -ィリデンアミドホスフェート 水素化ナトリウム(25.4 mg, 0.654丽 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5,6-ジメチル-8_[(5-フルォ 口ペンチル)スルファニル ]-3H-イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (100 mg, 0.317丽 oi) のテトラヒドロフラン a.OOml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにジェチル クロ口ホスフェート (0.0436 ml, 0.476匪 ol) のテトラ ヒドロフラン (1.00 ml) 溶液を加え、 さらに室温で 40分間かき混ぜた。 反応混 合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥(MgS04)後、 溶媒を減圧下に留去して題記化合物 (74.3 mg, 56%) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエーテルから再結晶して融点 125-127 の暗赤色結晶を得 た。 Getyl 5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1-ylidenamide Phosphate Sodium hydride (25.4 mg, 0.654 mol) (60% oil) in tetrahydrofuran (1.00 of the 1-amino-5,6-dimethyl-8 _ [(5-fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1 , 4] Thiazin-3-one (100 mg, 0.317-oi) in tetrahydrofuran a.OOml) was added, and the mixture was stirred under ice-cooling for 5 minutes. To this was added a solution of getyl chloride mouth phosphate (0.0436 ml, 0.476 bandol) in tetrahydrofuran (1.00 ml), and the mixture was further stirred at room temperature for 40 minutes. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (74.3 mg, 56%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 125-127.
赤外吸収スペクトル(IR) (KBr中) V: 2978, 2930, 2708, 1740, 1671, 1649, 1582 cm"1. Infrared absorption spectrum (IR) (in KBr) V: 2978, 2930, 2708, 1740, 1671, 1649, 1582 cm " 1 .
醒 R (CDC13) : δ 1.34 (6H, dt, J=7.0, 0.8 Hz), 1.46-1.85 (6H, m), 1.79 (3H, d, J=0.6Hz), 2.26 (3H, d, J=1. Hz), 3.98-4.20 (2H, m), 4.46 (2H, dt, J=47.2, 5.6 Hz),' 9.66 (1H, bs). Awakening: R (CDC1 3): δ 1.34 (6H, dt, J = 7.0, 0.8 Hz), 1.46-1.85 (6H, m), 1.79 (3H, d, J = 0.6Hz), 2.26 (3H, d, J = 1.Hz), 3.98-4.20 (2H, m), 4.46 (2H, dt, J = 47.2, 5.6 Hz), '9.66 (1H, bs).
元素分析値: CI7¾7N304S2FPとして Elementary analysis: as C I7 ¾ 7 N 3 0 4 S 2 FP
計算値 (¾) : C, 45.22; H, 6.03; N, 9.31; S, 14.20. Calculated (¾): C, 45.22; H, 6.03; N, 9.31; S, 14.20.
実測値 (¾) : C, 45.00; H, 5.93; N, 9.08; S, 14.24. Found (¾): C, 45.00; H, 5.93; N, 9.08; S, 14.24.
実施例 30 Example 30
ェチル P -べンジル -N- {5, 6-ジメチル- 8-[(5-フルォロペンチル)スルファニ ル ] -3-ォキソ -2, 3 -ジヒドロ -1H-イミダゾ [5,1- c] [1, 4]チアジン- 1-ィリデン }ホ スホンアミドエ一卜 Ethyl P-benzyl-N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1, 4] thiazine-1-ylidene} phosphonamide ester
水素化ナトリウム (38. Omg, 0.951匪 ol) (60%油性)のテトラヒドロフラン(1.00 ml) の懸濁液に、 実施例 6-23で得られた 1-ァミノ- 5, 6-ジメチル -8- [(5-フルォ 口ペンチル)スルファニル ]-3Hイミダゾ [5,1- c] [1,4]チアジン- 3-オン (150 mg, 0.476匪 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これに参考例で得られたェチルベンジルクロ口ホスホネート (2.00 ml) を加え、さらに室温で 2時間かき混ぜた。反応混合物に 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (25.7mg, 11%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルか ら再結晶して融点 132-134°Cの暗赤色結晶を得た。 A suspension of sodium hydride (38. Omg, 0.951 ol) (60% oil) in tetrahydrofuran (1.00 ml) was added to a suspension of 1-amino-5,6-dimethyl-8- obtained in Example 6-23. Add [(5-fluoropentyl) sulfanyl] -3H imidazo [5,1-c] [1,4] thiazin-3-one (150 mg, 0.476 ol) to tetrahydrofuran (1.00 ml) and add ice Stir for 5 minutes below. To this was added the ethyl benzyl clonal phosphonate (2.00 ml) obtained in Reference Example, and the mixture was further stirred at room temperature for 2 hours. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title of compound a (25.7 mg, 11%) as a dark red crystals. Ethyl acetate Recrystallization from the solution gave dark red crystals with a melting point of 132-134 ° C.
赤外吸収スペクトル(IR) (KBr中) V 2938, 2905, 2691, 1732, 1671, 1638, 1578 cm一1. Infrared absorption spectrum (IR) (in KBr) V 2938, 2905, 2691, 1732, 1671, 1638, 1578 cm- 1 .
Ή-NMR (CDC13) : δ 1.28 (3Η, t, J=6.9 Hz), 1.50-1.86 (6H, m), 1.77 (3H, s), Ή-NMR (CDC1 3): δ 1.28 (3Η, t, J = 6.9 Hz), 1.50-1.86 (6H, m), 1.77 (3H, s),
2.21 (3H, s), 2.95 (2H, t, J=7.5Hz), 3.20 (2H, d, J=20.7Hz), 3.90-4.16 (2H, m), 4.47 (2H, dt, J=47.1, 6.0 Hz), 7.16-7.34 (5H, m), 9.58 (1H, bs). 元素分析値: C22H29N3O3S2FP-0.7H2Oとして 2.21 (3H, s), 2.95 (2H, t, J = 7.5Hz), 3.20 (2H, d, J = 20.7Hz), 3.90-4.16 (2H, m), 4.47 (2H, dt, J = 47.1, 6.0 Hz), 7.16-7.34 (5H, m), 9.58 (1H, bs). Elemental analysis: C 22 H 29 N 3 O 3 S 2 FP-0.7H 2 O.
計算値 (¾) : C, 51.79; H, 6.01; N, 8.24; S, 12.57. Calculated value (¾): C, 51.79; H, 6.01; N, 8.24; S, 12.57.
実測値 (¾) : C, 51.64; H, 5.95; N, 8.36; S, 12.85. 実施例 23〜30で合成した化合物の一覧を以下に示す。 Observed value (C): C, 51.64; H, 5.95; N, 8.36; S, 12.85. A list of compounds synthesized in Examples 23 to 30 is shown below.
〔表 1 1 7 )(Table 11)
麄赘ぇ用紙(siは 実施例 31 麄 赘 ぇ Paper (si Example 31
8-[(5-フルォロペンチル)スルファニル ]-5, 6-ジメチル -1H-ィミダゾ [5, 1 - c] [1, 4]チアジン- 1,3 (2H) -ジオン 8-[(5-Fluoropentyl) sulfanyl] -5,6-dimethyl-1H-imidazo [5,1-c] [1,4] thiazine-1,3 (2H) -dione
実施例 Ί-ΊΪで得た化合物 (純度 90%, 2.70 g, 4.55 mmol)の THF-メタノール (3:5, 80 ml) 懸濁液に IN水酸化ナトリウム水溶液 (4.55 ml, 4.55腿 ol) を加え て溶解した。得られた溶液にエタノール(50ml)を加えて濃縮した。残渣にェタノ ール(50 ml)を加えて濃縮後、 酢酸ェチルおよびジェチルエーテルで稀釈した。析 出物を濾取し、 酢酸ェチルおよびジェチルェ一テルで洗浄後、 乾燥して To a suspension of the compound obtained in Example Ί-ΊΪ (purity 90%, 2.70 g, 4.55 mmol) in THF-methanol (3: 5, 80 ml) was added IN sodium hydroxide aqueous solution (4.55 ml, 4.55 liter). In addition, it dissolved. Ethanol (50 ml) was added to the obtained solution and concentrated. Ethanol (50 ml) was added to the residue, and the mixture was concentrated and diluted with ethyl acetate and getyl ether. The precipitate is collected by filtration, washed with ethyl acetate and ethyl acetate, and dried.
で表される表題化合物(433 mg, ί.37 匪 ol, 30. )を褐色粉末として得た。 The title compound represented by the formula (433 mg, 匪 .37 Marl ol, 30.) was obtained as a brown powder.
- NMR (200MHz, CDC13): δ 1.58 (2Η, m), 1.62 (2H, m), 1.74 (2H, m), 1.78 (3H, q, J= 1.2 Hz), 2.27 (3H, q, J= 1.2 Hz), 2.97 (2H, t, J= 7.2 Hz), 4.45 (2H, di, J= 47.0, 5.7 Hz). - NMR (200MHz, CDC1 3) : δ 1.58 (2Η, m), 1.62 (2H, m), 1.74 (2H, m), 1.78 (3H, q, J = 1.2 Hz), 2.27 (3H, q, J = 1.2 Hz), 2.97 (2H, t, J = 7.2 Hz), 4.45 (2H, di, J = 47.0, 5.7 Hz).
元素分析値: C13H17N202S2Fとして計算値: C, 49.35; H, 5.42; , 8.85; F, 6.00Elemental analysis: C 13 H 17 N 2 0 2 S 2 F Calculated: C, 49.35; H, 5.42 ;, 8.85; F, 6.00
(%) 、 実測値: C, 49.20; H, 5.38; N, 8.76; F, 5.97 (%) . (%), Found: C, 49.20; H, 5.38; N, 8.76; F, 5.97 (%).
mp. 174.0-175.0°C mp. 174.0-175.0 ° C
実施例 32 Example 32
ェチル N_{5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒドロ- m-イミダゾ [5, He] [1, 4]チアジン - 1 -ィリデン卜 P-フエニルホス ホンアミドエ一卜 . 0CH2CH3 Ethyl N_ {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-m-imidazo [5, He] [1,4] thiazine-1-ylidene P -Phenylphosphonamide ester. 0CH 2 CH 3
ジェチル フエニルホスホネート (0.371ini, 1.90mmol) の塩化チォニル(3.00 ml) 溶液に N,N-ジメチルホルムアミドを一滴加え、 3時間加熱還流した。 溶媒を 減圧下に留去して、 残留物をシリカゲルカラムクロマトグラフィーに付し、 n-へ キサン-酢酸ェチル (10:1, v/v) で溶出してェチル クロ口フエニルホスホネート を油状物として得、次の反応にそのまま用いた。水素化ナトリゥム(38.0 rag, 0.951 mmol) (60油性) のテトラヒドロフラン (1.00 ml) の懸濁液に、 実施例 6- 23で 得られた 卜ァミノ- 5, 6-ジメチル- 8- [ (5 -フルォ口ぺンチル)スルファニル] - 3H- イミダゾ [5,卜 c] [1,4]チアジン- 3-オン (150 mg, 0.476 mmol) のテトラヒドロフ ラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに上記で得られたェ チル クロ口フエニルホスホネートを加え、 さらに室温で 19.5時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽出した。抽出液を水洗、乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化合物 (40.2 mg, 18%) を暗赤色結晶として得 た。酢酸ェチル -ジェチルェ一テルから再結晶して融点 153- 155 の暗赤色結晶を 得た。 One drop of N, N-dimethylformamide was added to a solution of getyl phenylphosphonate (0.371ini, 1.90mmol) in thionyl chloride (3.00ml), and the mixture was refluxed for 3 hours. The solvent was distilled off under reduced pressure, and the residue was subjected to silica gel column chromatography, eluting with n-hexane-ethyl acetate (10: 1, v / v) to remove ethyl phenylphosphonate as an oil. And used directly in the next reaction. To a suspension of sodium hydride (38.0 rag, 0.951 mmol) (60 oily) in tetrahydrofuran (1.00 ml) was added the triamino-5,6-dimethyl-8-[(5- Fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (150 mg, 0.476 mmol) in tetrahydrofuran (1.00 ml) was added. Stir for a minute. To the mixture was added the ethyl phenyl phosphonate obtained above, and the mixture was further stirred at room temperature for 19.5 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title compound (40.2 mg, 18%) as a dark red crystals. The crystals were recrystallized from ethyl acetate-ethyl ether to obtain dark red crystals having a melting point of 153-155.
赤外吸収スペクトル(IR) (KBr中) V: 2934, 2697, 1738, 1671, 1644, 1574 cm"1. Ή-NMR (CDC13) : δ 1.30 (3Η, t, J=6.9 Hz), 1.50-1.66 (2H, m), 1.66-1.92 (4H, m), 1.78 (3H, s), 2.25 (3H, s), 2.98 (2H, t, J=7.5 Hz), 3.96-4.10 (2H, m), 4.46 (2H, dt, J=47.1, 6.0 Hz), 7.40-7.54 (3H, m), 7.93 (2H, dd, J=12.6, 7.2 Hz), 9.88 (1H, bs). Infrared absorption spectrum (IR) (in KBr) V:. 2934, 2697 , 1738, 1671, 1644, 1574 cm "1 Ή-NMR (CDC1 3): δ 1.30 (3Η, t, J = 6.9 Hz), 1.50 -1.66 (2H, m), 1.66-1.92 (4H, m), 1.78 (3H, s), 2.25 (3H, s), 2.98 (2H, t, J = 7.5 Hz), 3.96-4.10 (2H, m ), 4.46 (2H, dt, J = 47.1, 6.0 Hz), 7.40-7.54 (3H, m), 7.93 (2H, dd, J = 12.6, 7.2 Hz), 9.88 (1H, bs).
実施例 33 Example 33
ェチル N-{5, 6-ジメチル -8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ - 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン -卜ィリデン卜 P- (4-メトキシ フエニル)ホスホンアミドエ一ト Ethyl N- {5,6-dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-triylidene U-P- (4-methoxyphenyl) phosphonamide
ジェチル (4-メトキシフエ二ル)ホスホネート (479 mg, 1.90 mmol) の塩化チ ォニル (3.00ml) 溶液に N,N-ジメチルホルムアミドを一滴加え、 3時間加熱還流 した。 溶媒を減圧下に留去して、 残留物をシリカゲルカラムクロマトグラフィー に付し、 n -へキサン-酢酸ェチル (10:1, v/v) で溶出してェチル クロ口(4-メト キシフエニル)ホネートを油状物として得、次の反応にそのまま用いた。水素化ナ トリウム (38.0 nig, 0.951 匪 ol) (60%油性) のテトラヒドロフラン (1.00 ml) の懸濁液に、 実施例 6-23で得られた 1 -ァミノ- 5, 6-ジメチル- 8_[ (5-フルォロぺ ンチル)スルファニル] - 3H -イミダゾ [5,卜 c] [1,4]チアジン- 3-オン(150mg, 0.476 mmol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混ぜた。 これに上記で得られたェチル クロ口(4-メトキシフエ二ル)ホスフェートを加え、 さらに室温で 24時間かき混ぜた。 反応混合物に 1N塩酸を加えて酢酸ェチルで抽 出した。抽出液を水洗、乾燥(MgS04)後、溶媒を減圧下に留去して題記化合物(116 mg, 48¾) を暗赤色結晶として得た。 酢酸ェチル -ジェチルェ一テルから再結晶し て融点 173-175での暗赤色結晶を得た。 One drop of N, N-dimethylformamide was added to a solution of getyl (4-methoxyphenyl) phosphonate (479 mg, 1.90 mmol) in thionyl chloride (3.00 ml), and the mixture was heated under reflux for 3 hours. The solvent was distilled off under reduced pressure, and the residue was subjected to silica gel column chromatography, eluted with n-hexane-ethyl acetate (10: 1, v / v), and ethyl chloride (4-methoxyphenyl). The phonate was obtained as an oil and was used directly in the next reaction. A suspension of sodium hydride (38.0 nig, 0.951 ol) (60% oil) in tetrahydrofuran (1.00 ml) was added to the suspension of 1-amino-5,6-dimethyl-8_ [obtained in Example 6-23. (5-Fluoropentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazin-3-one (150 mg, 0.476 mmol) in tetrahydrofuran (1.00 ml) was added, and the mixture was cooled on ice for 5 minutes. Stirred. To this was added the ethyl clo-mouth (4-methoxyphenyl) phosphate obtained above, and the mixture was further stirred at room temperature for 24 hours. 1N Hydrochloric acid was added to the reaction mixture, and extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (116 mg, 48¾) as a dark red crystals. Recrystallization from ethyl acetate-jetil ether gave dark red crystals with a melting point of 173-175.
赤外吸収スペクトル(IR) (KBr中) V: 2940, 2905, 2693, 1734, 1671, 1642, 1601, 1580, 1507 cnr1. Infrared absorption spectrum (IR) (in KBr) V: 2940, 2905, 2693, 1734, 1671, 1642, 1601, 1580, 1507 cnr 1 .
證 (CDC13) : 6 1.29 (3H, t, J=6.9 Hz), 1.54-1.65 (2H, m), 1.65-1.84 (4H, ill), 1.78 (3H, d, J=0.9 Hz), 2.24 (3H, d, J=0.9 Hz), 2.98 (2H, t, J=7.5 Hz), 3.84 (3H, s), 3.95-4.08 (2H, m), 4.47 (2H, dt, J=47.4, 6.0 Hz), 6.95 (2H, dd, J=8.7, 3.0 Hz), 7.85 (2H, dd, J=12.3, 8.7 Hz), 9.88 (1H, bs) . Testimony (CDC1 3): 6 1.29 ( 3H, t, J = 6.9 Hz), 1.54-1.65 (2H, m), 1.65-1.84 (4H, ill), 1.78 (3H, d, J = 0.9 Hz), 2.24 (3H, d, J = 0.9 Hz), 2.98 (2H, t, J = 7.5 Hz), 3.84 (3H, s), 3.95-4.08 (2H, m), 4.47 (2H, dt, J = 47.4, 6.0 Hz), 6.95 (2H, dd, J = 8.7, 3.0 Hz), 7.85 (2H, dd, J = 12.3, 8.7 Hz), 9.88 (1H, bs).
元素分析値: C22H29N304S2FPとして Elementary analysis: as C 22 H 29 N 3 0 4 S 2 FP
計算値 (%) : C, 51.45; H, 5.69; N, 8.18; S, 12.49. Calculated value (%): C, 51.45; H, 5.69; N, 8.18; S, 12.49.
実測値 (%) : C, 51.19; H, 5.66; N, 7.99; S, 12.35. 実施例 34 Found (%): C, 51.19; H, 5.66; N, 7.99; S, 12.35. Example 34
ビス(2 -クロ口フエニル) 5,6-ジメチル- 8- [(5-フルォ 1 ァ ル]. -3-ォキソ -2,3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン - 1 -ィリデンアミ ドホスフエ一卜 Bis (2-chlorophenyl) 5,6-dimethyl-8-[(5-fluoro1Fal) .- 3-oxo-2,3-dihydro-1H-imidazo [5, 1-c] [1, 4] Thiazine-1-ylideneamide phosphate
水素化ナトリゥム(25.3 mg, 0.634 mmol) (60¾油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6-23で得られた卜ァミノ _5, 6-ジメチル- 8_[(5-フルォ 口ぺンチル)スルファニル] -3H-ィミダゾ [5,卜 c] [1, 4]チアジン -3 -才ン (100 mg, 0.317讓 ol) のテトラヒドロフラン (1.00 ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにビス(2-クロ口フエニル) ホスホロクロリデート (165 mg, 0.476 mmol) を加え、 さらに室温で 1時間かき混ぜた。反応混合物に 1N塩酸を加えて酢 酸ェチルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して 題記化合物 (153 mg, 78¾) を暗赤色結晶として得た。 酢酸ェチル-ジェチルエー テルから再結晶して融点 114-116°Cの暗赤色結晶を得た。 To a suspension of hydrogenated sodium (25.3 mg, 0.634 mmol) (60 g oily) in tetrahydrofuran (2.00 ml) was added the triamino_5,6-dimethyl-8 _ [(5-fluorinated) obtained in Example 6-23. [Pentyl) sulfanyl] -3H-imidazo [5, tric] [1,4] thiazine-3-100-mg (0.317 mL) in tetrahydrofuran (1.00 ml), and shake under ice-cooling for 5 minutes. mixed. To this was added bis (2-chlorophenyl) phosphorochloridate (165 mg, 0.476 mmol), and the mixture was further stirred at room temperature for 1 hour. 1N Hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure to give the title compound (153 mg, 78¾) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 114-116 ° C.
赤外吸収スペクトル(IR) (KBr中) v: 3069, 2936, 2865, 1744, 1667, 1634, 1566 cm"1. Infrared absorption spectrum (IR) (in KBr) v: 3069, 2936, 2865, 1744, 1667, 1634, 1566 cm " 1 .
Ή-NMR (CDC13) : δ 1.46-1.58 (2Η, m), 1.60-1.82 (4H, m), 1.77 (3H, d, J=0.9 Hz), 2.26 (3H, d, J=1.2 Hz), 2.89 (2H, t, J=7.5 Hz), 4.47 (2H, dt, J=47.4, 5.7 Hz), 7.10 (2H, tt, J=7.5, 1.5 Hz), 7.22 (2H, dt, J=7> 8, 2.1 Hz), 7.40 (2H, td, J=8.1, 1.2 Hz), 7.57 (2H, td, J=8.1, 1.5 Hz), 9.50 (1H, bs) . 元素分析値: C25H25N304S2C12FPとして Ή-NMR (CDC1 3): δ 1.46-1.58 (2Η, m), 1.60-1.82 (4H, m), 1.77 (3H, d, J = 0.9 Hz), 2.26 (3H, d, J = 1.2 Hz) , 2.89 (2H, t, J = 7.5 Hz), 4.47 (2H, dt, J = 47.4, 5.7 Hz), 7.10 (2H, tt, J = 7.5, 1.5 Hz), 7.22 (2H, dt, J = 7 > 8, 2.1 Hz), 7.40 (2H, td, J = 8.1, 1.2 Hz), 7.57 (2H, td, J = 8.1, 1.5 Hz), 9.50 (1H, bs). Elemental analysis: C 25 H 25 N 3 0 4 S 2 C1 2 FP
計算値 (¾) : C, 48.71; H, 4.09; N, 6.82; S, 10.40. Calculated (¾): C, 48.71; H, 4.09; N, 6.82; S, 10.40.
実測値 (%) : C, 48.67; H, 3.91; N, 6.79; S, 10.42. 実施例 35 Found (%): C, 48.67; H, 3.91; N, 6.79; S, 10.42. Example 35
N, - {5, 6-ジメチル- 8- [(5-フルォロペンチル)スルファニル ]-3-ォキソ -2, 3-ジヒ ドロ- 1H-イミダゾ [5, 1-c] [1,4]チアジン- 1-イリデン卜 Ν,Ν,Ν' ,Ν' -テトラメチ ルホスホリック 卜リアミド N,-{5,6-Dimethyl-8-[(5-fluoropentyl) sulfanyl] -3-oxo-2,3-dihydro-1H-imidazo [5,1-c] [1,4] thiazine-1 -Irident Ν, Ν, Ν ', Ν'-Tetramethylphosphoric triamide
水素化ナトリウム(12.7 mg, 0.317歷01) (60油性)のテトラヒドロフラン(2.00 ml) の懸濁液に、 実施例 6- 23で得られた 1-ァミノ- 5,6-ジメチル-8-[(5-フルォ 口ペンチル)スルファニル] - 3H -イミダゾ [5, 1-c] [1,4]チアジン- 3 オン (50.0 nig, 0.159腿 ol) のテトラヒドロフラン (1.00ml) 溶液を加え、 氷冷下 5分間かき混 ぜた。 これにビス(ジメチルァミノ)ホスホリルクロリド (0.0370 ml, 0.238睡 1) を加え、 さらに室温で 1時間かき混ぜた。'反応混合物に 1N塩酸を加えて酢酸ェチ ルで抽出した。 抽出液を水洗、 乾燥 (MgS04) 後、 溶媒を減圧下に留去して題記化 合物 (27.3mg, 38%) を暗赤色結晶として得た。 酢酸ェチル -ジェチルエーテルか ら再結晶して融点 150-152°Cの暗赤色結晶を得た。 To a suspension of sodium hydride (12.7 mg, 0.317-01) (60 oily) in tetrahydrofuran (2.00 ml) was added 1-amino-5,6-dimethyl-8-[(obtained in Example 6-23. 5-Fluoro-pentyl) sulfanyl] -3H-imidazo [5,1-c] [1,4] thiazine-3-one (50.0 nig, 0.159 tmol) in tetrahydrofuran (1.00 ml) Stir for a minute. Bis (dimethylamino) phosphoryl chloride (0.0370 ml, 0.238 sleep 1) was added thereto, and the mixture was further stirred at room temperature for 1 hour. '1N hydrochloric acid was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The extract was washed with water, dried (MgSO 4), to obtain solvent was distilled off under reduced pressure the title of compound (27.3 mg, 38%) as a dark red crystals. Recrystallization from ethyl acetate-getyl ether gave dark red crystals with a melting point of 150-152 ° C.
赤外吸収スぺクトル(IR) (KBr中) ソ : 2932, 2845, 2807, 1746, 1672, 1645, 1586 cm"1. Infrared absorption spectrum (IR) (in KBr) So: 2932, 2845, 2807, 1746, 1672, 1645, 1586 cm " 1 .
Ή-NMR (CDC13) : δ 1.48-1.84 (6Η, m), 1.78 (3H, d, J=0.9 Hz), 2.24 (3H, d, J=0.9 Hz), 2.63 (6H, s), 2.66 (6H, s), 2.94 (2H, 1, J=7.5 Hz), 4.45 (2H, dt, J=47.1, 6.0 Hz), 9.87 (1H, bs). Ή-NMR (CDC1 3): δ 1.48-1.84 (6Η, m), 1.78 (3H, d, J = 0.9 Hz), 2.24 (3H, d, J = 0.9 Hz), 2.63 (6H, s), 2.66 (6H, s), 2.94 (2H, 1, J = 7.5 Hz), 4.45 (2H, dt, J = 47.1, 6.0 Hz), 9.87 (1H, bs).
元素分析値: C17H29N502S2FPとして Elementary analysis: as C 17 H 29 N 5 0 2 S 2 FP
計算値 (%) : C, 45.42; H, 6.50; N, 15.58; S, 14.27. Calculated (%): C, 45.42; H, 6.50; N, 15.58; S, 14.27.
実測値 (%) : C, 45.13; H, 6.47; N, 15.19; S, 14.51. Found (%): C, 45.13; H, 6.47; N, 15.19; S, 14.51.
実施例 36 - 1 Example 36-1
N - [8- (ベンジルスルフィ二ル)- 5-メチル -7-ォキシド -3-ォキソ -6-ペンチル- 2, 3 - ジヒド口- 1H-ィミダゾ [5, 1-c] [1, ]チアジン- 1 -ィリデン ] -4-メチルベンゼンス ルホンアミド N- [8- (benzylsulfinyl) -5-methyl-7-oxide-3-oxo-6-pentyl-2,3- Dihydrid-1H-imidazo [5, 1-c] [1,] thiazin-1-ylidene] -4-methylbenzenesulfonamide
実施例 4- 233で合成した化合物 (101 mg, 0.191醒 ol) を酢酸一 THF (1:1、 3.0 ml) に溶解し、 30%過酸化水素水 (0.039 ml, 0.383 腿 ol) を加えて室温で 30 分間、さらに 60°Cで 30分間攪拌した。反応液に 30%過酸化水素水 (0.039 ml, 0.383 匪 ol) を加えてさらに 60°Cで 30分間攪拌した。 反応液を酢酸ェチル (20 ml) で 希釈し、水、 2 %炭酸水素ナトリゥム水溶液、 5 %亜硫酸ナトリゥム水溶液、 0.1N 塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 減圧下濃縮乾固 した。残渣をメタノ一ルに溶解し、逆相系分取 HPLC (担体: 0DS、 YMC-Pack、 D- 0DS-5、 内径 20 mm x長さ 250匪、 ワイ ·ェム ·シィ社製、 移動相: 0.05 (v/v)%トリフル ォロ酢酸/ 60(v/v)%ァセトニトリル水) に付し、 溶出容量 205〜240ml の画分を 集め、 濃縮乾固して標題化合物の黄色粉末 (30 mg、 0.054 mmol) を得た。 The compound synthesized in Example 4-233 (101 mg, 0.191 ol) was dissolved in acetic acid-THF (1: 1, 3.0 ml), and 30% hydrogen peroxide solution (0.039 ml, 0.383 t ol) was added. The mixture was stirred at room temperature for 30 minutes and further at 60 ° C for 30 minutes. A 30% aqueous hydrogen peroxide solution (0.039 ml, 0.383 ol) was added to the reaction solution, and the mixture was further stirred at 60 ° C for 30 minutes. Dilute the reaction mixture with ethyl acetate (20 ml), wash with water, 2% aqueous sodium bicarbonate, 5% aqueous sodium sulfite, 0.1N hydrochloric acid, water and saturated saline, dry over sodium sulfate, and concentrate under reduced pressure It was dried. The residue is dissolved in methanol, and reversed-phase preparative HPLC (Carrier: 0DS, YMC-Pack, D-0DS-5, 20 mm ID x 250 mm long, manufactured by YMC Corporation, mobile phase : 0.05 (v / v)% trifluoroacetic acid / 60 (v / v)% acetonitrile water), collect the fractions with an elution volume of 205-240ml, and concentrate to dryness to give a yellow powder of the title compound (30%). mg, 0.054 mmol).
Ή- NMR (300 MHz, 重クロ口ホルム中) : δ 0.90 (3Η, t, J= 6.8 Hz), 1.36 (4H, m), 1.64 (2H, m), 2.44 (3H, s), 2.63 (3H, s), 2.70 (1H, ddd, J= 16.2, 9.0, 7.2 Hz), 2.85 (1H, ddd, J= 15.8, 8,7, 7.1 Hz), 4.47 (1H, d, J= 12.4 Hz), 4.52 (1H, d, J= 12.4 Hz), 7.32 (2H, d, J= 8.7 Hz), 7.35 (5H, s), 7.82 (2H, d, J- 8.2 Hz). Ή-NMR (300 MHz, in double-mouthed form): δ 0.90 (3Η, t, J = 6.8 Hz), 1.36 (4H, m), 1.64 (2H, m), 2.44 (3H, s), 2.63 ( 3H, s), 2.70 (1H, ddd, J = 16.2, 9.0, 7.2 Hz), 2.85 (1H, ddd, J = 15.8, 8, 7, 7.1 Hz), 4.47 (1H, d, J = 12.4 Hz) , 4.52 (1H, d, J = 12.4 Hz), 7.32 (2H, d, J = 8.7 Hz), 7.35 (5H, s), 7.82 (2H, d, J- 8.2 Hz).
元素分析値: C26H29N305S3 O.4H20として計算値: C, 55.08; H, 5.30; N, 7.41; S, 16.97 (%), 実測値: C, 55.13; H, 5.30; N, 7.23; S, 16.8.0 (%) . Elemental analysis: C 26 H 29 N 3 0 5 S 3 O.4H 2 0 Calculated: C, 55.08; H, 5.30; N, 7.41; S, 16.97 (%), Found: C, 55.13; H , 5.30; N, 7.23; S, 16.8.0 (%).
質量分析 (SIMS) : 560(M+H), 512(M+H-SO), 421鎮- SO- PhCH2) Mass spectrometry (SIMS): 560 (M + H), 512 (M + H-SO), 421-SO-PhCH 2 )
実施例 36-2 ' Example 36-2 '
N- [8- (ベンジルスルホニル) -5-メチル -7-ォキシド- 3-ォキソ -6-ペンチル -2, 3 -ジ ヒドロ- 1H-イミダゾ [5,卜 c] [1, 4]チアジン- 1-ィリデン ]-4-メチルベンゼンスル ホンアミド N- [8- (benzylsulfonyl) -5-methyl-7-oxide-3-oxo-6-pentyl-2,3-dihydro-1H-imidazo [5, tric] [1,4] thiazine-1 -Ylidene] -4-methylbenzenesulfonamide
実施例 4-233で合成した化合物(111 mg, 0.210腿 ol) をクロ口ホルム (3.0 ml) に溶解し、 m-ク口口過安息香酸 (130 rag, 0.526讓 ol) を加えて室温で 20分間攪 拌した。 反応液を酢酸ェチル (20 ml) で希釈し、 水、 2%炭酸水素ナトリウム水 溶液、 5%亜硫酸ナトリウム水溶液、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 減圧下濃縮した。 残渣をメタノールに溶解し、 逆相系 分取 HPLC (担体: 0DS、 YMC-Pack, SH- 343-5、 内径 20匪 x長さ 250讓、 ワイ · ェム ·シィ社製、 移動相: 0.05(v/v)%トリフルォロ酢酸/ 60(v/v)%ァセトニトリ ル水) に付した。 溶出容量 245〜290 ml の画分を集めて濃縮し、 残渣に 50%メ タノ一ル水を加えた。析出物を濾取し、 50%メタノール水で洗浄、乾燥して標題化 合物の黄色粉末 (44 mg、 0.076 mmol) を得た。 The compound synthesized in Example 4-233 (111 mg, 0.210 mol) was dissolved in chloroform (3.0 ml), m-mouth perbenzoic acid (130 rag, 0.526 alcohol) was added, and the mixture was added at room temperature. Stirred for 20 minutes. Dilute the reaction mixture with ethyl acetate (20 ml), wash with water, 2% aqueous sodium bicarbonate, 5% aqueous sodium sulfite, 0.1N hydrochloric acid, water and saturated saline, dry over sodium sulfate, and reduce pressure. Concentrated. Dissolve the residue in methanol and reverse phase Preparative HPLC (Carrier: 0DS, YMC-Pack, SH-343-5, Inner diameter 20 marauder x Length 250 cruises, manufactured by YMC Corp., Mobile phase: 0.05 (v / v)% trifluoroacetic acid / 60 (v / v)% acetonitrile water). Fractions with an elution volume of 245 to 290 ml were collected and concentrated, and 50% aqueous methanol was added to the residue. The precipitate was collected by filtration, washed with 50% aqueous methanol and dried to obtain a yellow powder of the title compound (44 mg, 0.076 mmol).
'H-NMR (300 MHz, 重クロ口ホルム中) : δ 0.92 (3Η, t, J= 7.0 Hz), 1.39 (4H, m), 1.65 (2H, brquint, J= 7.2 Hz), 2.45 (3H, s), 2.50 (3H, s), 2.59 (2H, t, J= 8.3 Hz), 4.51 (1H, d, J= 12.1 Hz), 4.89 (1H, d, J= 12.1 Hz), 7.32 (2H, d, J= 8.3 Hz), 7.34 (5H, m), 7.76 (2H, d, J- 8.3 Hz). 'H-NMR (300 MHz, in double-mouthed form): δ 0.92 (3Η, t, J = 7.0 Hz), 1.39 (4H, m), 1.65 (2H, brquint, J = 7.2 Hz), 2.45 (3H , s), 2.50 (3H, s), 2.59 (2H, t, J = 8.3 Hz), 4.51 (1H, d, J = 12.1 Hz), 4.89 (1H, d, J = 12.1 Hz), 7.32 (2H , d, J = 8.3 Hz), 7.34 (5H, m), 7.76 (2H, d, J- 8.3 Hz).
元素分析値: C26H29N306S3として計算値: C, 54.24; H, 5.08; N, 7.30; S, 16.71 ( ), 実測値: C, 54.19; H, 4.97; N, 7.38; S, 16.79 (%) . Elemental analysis: C 26 H 29 N 3 0 6 S 3 Calculated: C, 54.24; H, 5.08 ; N, 7.30; S, 16.71 (), Found: C, 54.19; H, 4.97 ; N, 7.38 ; S, 16.79 (%).
質量分析 (SIMS): 576 (M+H), 598(M+Na). Mass spectrometry (SIMS): 576 (M + H), 598 (M + Na).
実施例 36-3 Example 36-3
4 -メチル _N_{5-メチル -8- [ (5 -メチルへキシル)スルホニル] - 7 -ォキシド- 3-ォキ ソ -6-ペンチル- 2, 3-ジヒドロ- 1H-イミダゾ [5, 1-c] [1, 4]チアジン - 1 -ィリデン }ベ ンゼンスルホンアミド 4-Methyl _N_ {5-methyl-8-[(5-methylhexyl) sulfonyl] -7-oxide-3-oxo-6-pentyl-2,3-dihydro-1H-imidazo [5,1- c] [1,4] thiazine-1-ylidene} benzenesulfonamide
実施例 4- 225で合成した化合物(113 mg, 0.211腿 ol) をクロ口ホルム (3.0ml) に溶解し、 m-クロ口過安息香酸 (156 mg, 0.633 mmol) を加えて室温で 60分間攪 拌した。 反応液を酢酸ェチル (20 ml) で希釈し、 水、 2%炭酸水素ナトリウム水 溶液、 5 %亜硫酸ナトリゥム水溶液、 0.1N塩酸、 水および飽和食塩水で洗浄後、 硫酸ナトリウムで乾燥し、 減圧下濃縮した。 残渣をメタノールに溶解し、 逆相系 分取 HPLC (担体: 0DS、 YMC-Pack, SH- 343-5、 内径 20腿 x長さ 250 mm、 ワイ · ェム ·シィ社製、 移動相: 0.05(v/v)%トリフルォロ酢酸/ 60(v/v)%ァセトニトリ ル水) に付した。 溶出容量 345〜375 ml の画分を集めて濃縮し、 酢酸ェチルで抽 出、水洗、乾燥後、濃縮乾固して標題化合物の黄色粉末 (38 mg、 0.065 mmol) を得 た。 , The compound synthesized in Example 4-225 (113 mg, 0.211 tmol) was dissolved in chloroform (3.0 ml), m-chloroperbenzoic acid (156 mg, 0.633 mmol) was added, and the mixture was added at room temperature for 60 minutes. Stirred. Dilute the reaction mixture with ethyl acetate (20 ml), wash with water, 2% aqueous sodium bicarbonate, 5% aqueous sodium sulfite, 0.1N hydrochloric acid, water and saturated saline, dry over sodium sulfate, and reduce pressure. Concentrated. The residue was dissolved in methanol and subjected to reversed-phase preparative HPLC (Carrier: 0DS, YMC-Pack, SH-343-5, ID: 20 t x 250 mm, manufactured by YMC), mobile phase: 0.05 (v / v)% trifluoroacetic acid / 60 (v / v)% acetonitrile water). Fractions having an elution volume of 345 to 375 ml were collected, concentrated, extracted with ethyl acetate, washed with water, dried, and concentrated to dryness to obtain a yellow powder of the title compound (38 mg, 0.065 mmol). ,
- NMR (300 MHz, 重クロ口ホルム中) : δ 0.87 (6Η, d, J= 6.4 Hz), 0.89 (3H, t, J= 6.8 Hz), 1.15 (2H, brdt, J= 8.7, 6.8 Hz), 1.36 (6H, m), 1.51 (1H, sept, J= 6.7 Hz), 1.62 (3H, m), 1.82 (1H, m), 2.46 (3H, s), 2.50 (3H, s), 2.54 (2H, brt, J= 8.5 Hz), 3.06 (1H, ddd, J= 12.4, 10.2, 6.8 Hz), 3.66 (1H, ddd, J= 12.4, 9.8, 4.5 Hz), 7.36 (2H, d, J= 8.3 Hz), 7.80 (2H, d, J= 8.3 Hz). 元素分析値: C26H37N306S3として計算値: C, 53.49; H, 6.39; N, 7.20; S, 16.48-NMR (300 MHz, in double-mouthed form): δ 0.87 (6Η, d, J = 6.4 Hz), 0.89 (3H, t, J = 6.8 Hz), 1.15 (2H, brdt, J = 8.7, 6.8 Hz) ), 1.36 (6H, m), 1.51 (1H, sept, J = 6.7 Hz), 1.62 (3H, m), 1.82 (1H, m), 2.46 (3H, s), 2.50 (3H, s), 2.54 (2H, brt, J = 8.5 Hz), 3.06 (1H, ddd, J = 12.4, 10.2, 6.8 Hz), 3.66 (1H, ddd, J = 12.4, 9.8, 4.5 Hz), 7.36 (2H, d, J = 8.3 Hz) .), 7.80 (2H, d , J = 8.3 Hz) elemental analysis: C 26 H 37 N 3 0 6 S 3 calculated: C, 53.49; H, 6.39 ; N, 7.20; S, 16.48
(%), 実測値: C, 53.15; H, 6.28; , 7.44; S, 16.12 (%) . (%), Found: C, 53.15; H, 6.28;, 7.44; S, 16.12 (%).
質量分析 (SIMS) : 584 (M+H), 606 (M+Na). Mass spectrometry (SIMS): 584 (M + H), 606 (M + Na).
製剤例 1 - 1 Formulation Example 1-1
下記に示す処方の全成分を均一に混和し、 ゼラチンカプセルに充填し、 カプセ ル 1個当たり 100 mgの実施例 4-2で得られた化合物を含有するカプセル剤を製造 する。 Mix all the components of the following formulation uniformly and fill into gelatin capsules to produce capsules containing 100 mg of the compound obtained in Example 4-2 per capsule.
実施例 4-2で得られた化合物 100 mg 100 mg of the compound obtained in Example 4-2
乳糖 100 mg Lactose 100 mg
コーンスターチ 40 mg Corn starch 40 mg
ステアリン酸マグネシウム 1 Omg Magnesium stearate 1 Omg
1カプセル 25 Omg 1 capsule 25 Omg
製剤例卜 2 Formulation example 2
下記に示す処方の全成分を均一に混和し、 ゼラチンカプセルに充填し、 カプセ ル 1個当たり 100 mgの実施例 4-6で得られた化合物を含有するカプセル剤を製造 する。 All the components of the following formulation are mixed uniformly, filled into a gelatin capsule, and a capsule is prepared containing 100 mg of the compound obtained in Example 4-6 per capsule.
実施例 4 - 6で得られた化合物 100 m g Compound obtained in Examples 4-6 100 mg
乳糖 10 Omg Lactose 10 Omg
コーンスターチ 4 Omg Cornstarch 4 Omg
ステアリン酸マグネシウム 1 Omg Magnesium stearate 1 Omg
1力プセル 250 m g 250 kg of 1-strength pushel
製剤例卜 3 Formulation example 3
下記に示す処方の全成分を均一に混和し、 ゼラチンカプセルに充填し、 カプセ ル 1個当たり 100 mgの実施例 4-9で得られた化合物を含有するカプセル剤を製造 する。 Mix all the components of the following formulation uniformly, fill into gelatin capsules, and produce capsules containing 100 mg of the compound obtained in Example 4-9 per capsule.
実施例 4-9で得られた化合物 100 m g 100 mg of the compound obtained in Example 4-9
乳糖 10 Omg コーンスターチ 40mg Lactose 10 Omg 40mg corn starch
ステアリン酸マグネシウム 10mg Magnesium stearate 10mg
1カプセル 25 Omg 1 capsule 25 Omg
製剤例卜 4 Formulation example 4
下記に示す処方の全成分を均一に混和し、 ゼラチンカプセルに充填し、 カプセ ル 1個当たり 100 mgの実施例 4 - 34で得られた化合物を含有するカプセル剤を製 造する。 All the components of the following formulation are mixed uniformly and filled into gelatin capsules to produce capsules containing 100 mg of the compound obtained in Examples 4-34 per capsule.
実施例 4-34で得られた化合物 100 m g 100 mg of the compound obtained in Example 4-34
乳糖 10 Omg Lactose 10 Omg
コ一ンスターチ 4 Omg Cornstarch 4 Omg
ステアリン酸マグネシウム 10mg Magnesium stearate 10mg
1カプセル 25 Omg 1 capsule 25 Omg
製剤例 1-5 Formulation Example 1-5
下記に示す処方の全成分を均一に混和し、 ゼラチンカプセルに充填し、 カプセ ル 1個当たり 100 mgの実施例 4-35で得られた化合物を含有するカプセル剤を製 造する。 Mix all the components of the following formulation uniformly and fill into gelatin capsules to produce capsules containing 100 mg of the compound obtained in Example 4-35 per capsule.
実施例 4-35で得られた化合物 10 Omg Compound 10 Omg obtained in Example 4-35
乳糖 10 Omg Lactose 10 Omg
コーンスターチ 4 Omg Cornstarch 4 Omg
ステアリン酸マグネシウム 10mg Magnesium stearate 10mg
1力プセル 25 Omg 1 force 25 Omg
製剤例 2-1 Formulation Example 2-1
実施例 4-2で得られた化合物とステアリン酸マグネシウムを可溶性デンプンの 水溶液で顆粒化し、 乾燥後、 乳糖及びコーンスターチと混和し、 混合物を圧縮成 型し、 錠剤 1個当たり 100 mgの化合物を含有する錠剤を製造する。 The compound obtained in Example 4-2 and magnesium stearate are granulated with an aqueous solution of soluble starch, dried, then mixed with lactose and corn starch, and the mixture is compression-molded to contain 100 mg of the compound per tablet. To make tablets.
実施例 4 - 2で得られた化合物 100 m g 100 mg of the compound obtained in Example 4-2
乳糖 65mg Lactose 65mg
コーンスターチ 3 Omg Cornstarch 3 Omg
可溶性デンプン 35mg ステアリン酸マグネシウム 20mg 35mg soluble starch Magnesium stearate 20mg
1錠 25 Omg 1 tablet 25 Omg
製剤例 2_2 Formulation example 2_2
実施例 4-6で得られた化合物とステアリン酸マグネシウムを可溶性デンプンの 水溶液で顆粒化し、 乾燥後、 乳糖及びコーンスターチと混和し、 混合物を圧縮成 型し、 錠剤 1個当たり 100 mgの化合物を含有する錠剤を製造する。 The compound obtained in Example 4-6 and magnesium stearate were granulated with an aqueous solution of soluble starch, dried, mixed with lactose and corn starch, and the mixture was compression-molded to contain 100 mg of the compound per tablet. To make tablets.
実施例 4-6で得られた化合物 100 m g 100 mg of the compound obtained in Example 4-6
乳糖 '65mg Lactose '65mg
コーンス夕一チ 3 Omg Corn Yuichi 3 Omg
可溶性デンプン 35mg 35mg soluble starch
ステアリン酸マグネシウム 2 Omg Magnesium stearate 2 Omg
1錠 25 Omg 1 tablet 25 Omg
製剤例 2-3 Formulation example 2-3
実施例 4-9で得られた化合物とステアリン酸マグネシゥムを可溶性デンプンの 水溶液で顆粒化し、 乾燥後、 乳糖及びコーンスターチと混和し、 混合物を圧縮成 型し、 錠剤 1個当たり 100 ragの化合物を含有する錠剤を製造する。 ' 実施例 4 - 9で得られた化合物 100 m g The compound obtained in Example 4-9 and magnesium stearate were granulated with an aqueous solution of soluble starch, dried, mixed with lactose and corn starch, and the mixture was compression-molded, containing 100 rag of the compound per tablet. To make tablets. '' 100 mg of compound obtained in Examples 4-9
乳糖 65mg Lactose 65mg
コーンスターチ 3 Omg Cornstarch 3 Omg
可溶性デンプン 35mg 35mg soluble starch
ステアリン酸マグネシゥム 2 Omg Magnesium stearate 2 Omg
1錠 25 Omg 1 tablet 25 Omg
製剤例 2 - 4 Formulation Example 2-4
実施例 4-34で得られた化合物とステアリン酸マグネシウムを可溶性デンプン の水溶液で顆粒化し、 乾燥後、 乳糖及びコーンスターチと混和し、 混合物を圧縮 成型し、 錠剤 1個当たり 10 Omgの化合物を含有する錠剤を製造する。 The compound obtained in Example 4-34 and magnesium stearate are granulated with an aqueous solution of soluble starch, dried, then mixed with lactose and corn starch, and the mixture is compression-molded, containing 10 Omg of the compound per tablet. Make tablets.
実施例 4-34で得られた化合物 100 m g 100 mg of the compound obtained in Example 4-34
乳糖 65mg Lactose 65mg
コーンスターチ 3 Omg 可溶性デンプン 3 5mg Cornstarch 3 Omg Soluble starch 3 5mg
ステアリン酸マグネシウム 20mg Magnesium stearate 20mg
1錠 250mg 250mg / tablet
製剤例 2 - 5 Formulation Example 2-5
実施例 4-35 で得られた化合物とステアリン酸マグネシウムを可溶性デンプン の水溶液で顆粒化し、 乾燥後、 乳糖及びコーンスターチと混和し、 混合物を圧縮 成型し、 錠剤 1個当たり 100 mgの化合物を含有する錠剤を製造する。 The compound obtained in Example 4-35 and magnesium stearate are granulated with an aqueous solution of soluble starch, dried, mixed with lactose and corn starch, and the mixture is compression-molded to contain 100 mg of the compound per tablet. Make tablets.
実施例 4-35で得られた化合物 10 Omg Compound 10 Omg obtained in Example 4-35
乳糖 65mg Lactose 65mg
コーンスターチ 3 Omg Cornstarch 3 Omg
可溶性デンプン 35mg 35mg soluble starch
ステアリン酸マグネシウム 2 Omg Magnesium stearate 2 Omg
1錠 25 Omg 1 tablet 25 Omg
製剤例 3-1 Formulation Example 3-1
実施例 4-2で得られた化合物を 30% (w/v)ポリエチレンダリコール 400を含む生 理食塩水に溶解して化合物の 0.05%溶液を調製し、滅菌濾過後バイアルに 30mlず つ分注し、 バイアル 1個当たり 15mgの化合物を含有する注射剤を製造する。 製剤例 3-2 The compound obtained in Example 4-2 was dissolved in physiological saline containing 30% (w / v) polyethylene dalicol 400 to prepare a 0.05% solution of the compound.After sterile filtration, 30 ml of the compound was dispensed into vials. Inject and manufacture injections containing 15 mg of compound per vial. Formulation Example 3-2
実施例 4-6で得られた化合物を 30 (w/v)ポリエチレングリコ一ル 400を含む生 理食塩水に溶解して化合物の 0.05溶液を調製し、滅菌濾過後バイアルに 30mlず つ分注し、 バイアル 1個当たり 15mgの化合物を含有する注射剤を製造する。 製剤例 3-3 The compound obtained in Example 4-6 was dissolved in physiological saline containing 30 (w / v) polyethylene glycol 400 to prepare a 0.05 solution of the compound.After sterile filtration, 30 ml was dispensed into vials. Then, an injection containing 15 mg of the compound per vial is manufactured. Formulation Example 3-3
実施例 4-9で得られた化合物を 30% (w/v)ポリエチレングリコール 400を含む生 理食塩水に溶解して化合物の 0.05%溶液を調製し、滅菌濾過後バイアルに 30mlず つ分注し、 バイアル 1個当たり 15mgの化合物を含有する注射剤を製造する。 製剤例 3 - 4 The compound obtained in Example 4-9 was dissolved in physiological saline containing 30% (w / v) polyethylene glycol 400 to prepare a 0.05% solution of the compound.After sterile filtration, 30 ml was dispensed into vials. Then, an injection containing 15 mg of the compound per vial is manufactured. Formulation Example 3-4
実施例 4- 34で得られた化合物を 30% (w/v)ポリエチレングリコール 400を含む 生理食塩水に溶解して化合物の 0.05%溶液を調製し、 滅菌濾過後バイアルに 30ml ずつ分注し、 バイアル 1個当たり 15mgの化合物を含有する注射剤を製造する。 製剤例 3-5 The compound obtained in Example 4-34 was dissolved in physiological saline containing 30% (w / v) polyethylene glycol 400 to prepare a 0.05% solution of the compound.After sterile filtration, the solution was dispensed into vials at 30 ml each. Injections containing 15 mg of compound per vial are prepared. Formulation Example 3-5
実施例 4-35で得られた化合物を 30% (w/v)ポリエチレンダリコ一ル 400を含む 生理食塩水に溶解して化合物の 0. 05 溶液を調製し、 滅菌濾過後バイアルに 30ml ずつ分注し、 バイアル 1個当たり 15mgの化合物を含有する注射剤を製造する。 製剤例 4-1 The compound obtained in Example 4-35 was dissolved in a physiological saline solution containing 30% (w / v) polyethylene glycol 400 to prepare a 0.05 solution of the compound. Dispense to produce injections containing 15 mg of compound per vial. Formulation Example 4-1
実施例 4-2で得られた化合物と力ルポキシメチルセルロースナトリゥムを乳鉢 中で均一に混和し、 サッカリンナトリウムを精製水に溶かした液を少しずつ加え てよくかき混ぜる。 10%安息香酸ナトリウム水溶液 1ml を加え、 精製水で全量を 100mlとし、 100mlあたり 10mgの化合物を含有する懸濁剤を製造する。 The compound obtained in Example 4-2 and lipoxymethylcellulose sodium are uniformly mixed in a mortar, and a solution prepared by dissolving saccharin sodium in purified water is added little by little and mixed well. Add 1 ml of 10% aqueous sodium benzoate solution, make up to 100 ml with purified water, and prepare a suspension containing 10 mg of compound per 100 ml.
実施例 4-2で得られた化合物 1 O mg Compound obtained in Example 4-2 1 O mg
カルボキシメチルセルロースナトリゥム 2 . 0 g Carboxymethylcellulose sodium 2.0 g
4 O mg 4 O mg
安息香酸ナトリウム 1 0 O mg Sodium benzoate 10 O mg
1 0 0 m l 1 0 0 ml
製剤例 4- 2 Formulation Example 4-2
実施例 4-6で得られた化合物とカルボキシメチルセルロースナトリゥムを乳鉢 中で均一に混和し、 サッカリンナトリウムを精製水に溶かした液を少しずつ加え てよくかき混ぜる。 10 安息香酸ナトリウム水溶液 lml を加え、 精製水で全量を 100mlとし、 100mlあたり 10mgの化合物を含有する懸濁剤を製造する。 The compound obtained in Example 4-6 and carboxymethylcellulose sodium are uniformly mixed in a mortar, and a solution prepared by dissolving saccharin sodium in purified water is added little by little and mixed well. 10 Add 1 ml of an aqueous solution of sodium benzoate, make up to 100 ml with purified water, and prepare a suspension containing 10 mg of compound per 100 ml.
実施例 4-6で得られた化合物 1 0 m g Compound obtained in Example 4-6 10 mg
カルポキシメチルセル口一スナトリウム 2 . 0 g Carboxymethylcell mouth sodium 2.0 g
サッカリンナトリウム 4 O mg Saccharin sodium 4 O mg
安息香酸ナトリウム 1 0 O m g Sodium benzoate 10 O mg
全量 1 0 0 m l Total volume 100 m l
製剤例 4-3 Formulation Example 4-3
実施例 -9で得られた化合物とカルポキシメチルセル口一スナトリゥムを乳鉢 中で均一に混和し、 サッカリンナトリゥムを精製水に溶かした液を少しずつ加え てよくかき混ぜる。 10%安息香酸ナトリウム水溶液 lml を加え、 精製水で全量を 100mlとし、 100mlあたり 10mgの化合物を含有する懸濁剤を製造する。 実施例 4-9で得られた化合物 丄 Omg カルポキシメチルセルロースナトリゥム 2. 0 g The compound obtained in Example 9 and carboxymethylcellulose sodium are uniformly mixed in a mortar, and a solution prepared by dissolving saccharin sodium in purified water is added little by little and mixed well. Add 1 ml of 10% aqueous sodium benzoate solution, make up to 100 ml with purified water, and prepare a suspension containing 10 mg of compound per 100 ml. Compound obtained in Example 4-9 Omg carboxymethylcellulose sodium 2.0 g
サッカリンナトリウム 4 Omg Saccharin sodium 4 Omg
安息香酸ナトリウム 10 Omg Sodium benzoate 10 Omg
100ml 100ml
製剤例 4 - 4 Formulation Example 4-4
実施例 4 - 34.で得られた化合物とカルポキシメチルセルロースナトリウムを乳 鉢中で均一に混和し、 サッカリンナトリゥムを精製水に溶かした液を少しずつ加 えてよくかき混ぜる。 10安息香酸ナトリゥム水溶液 1mlを加え、精製水で全量を 100mlとし、 100mlあたり 10mgの化合物を含有する懸濁剤を製造する。 The compound obtained in Example 4-34 and carboxymethylcellulose sodium are uniformly mixed in a mortar, and a solution prepared by dissolving saccharin sodium in purified water is added little by little and mixed well. 10 ml of an aqueous solution of sodium benzoate is added, and the total volume is made up to 100 ml with purified water to prepare a suspension containing 10 mg of the compound per 100 ml.
実施例 4-34で得られた化合物 1 Omg Compound 1 Omg obtained in Example 4-34
カルポキシメチルセル口一スナトリゥム 2. 0 g Carboxymethylcell mouth-sunium 2.0 g
4 Omg 4 Omg
安息香酸ナトリウム 10 Omg Sodium benzoate 10 Omg
100ml 100ml
製剤例 4 - 5 Formulation Examples 4-5
実施例 4-35 で得られた化合物とカルポキシメチルセルロースナトリウムを乳 鉢中で均一に混和し、 サッカリンナトリゥムを精製水に溶かした液を少しずつ加 えてよくかき混ぜる。 10%安息香酸ナトリウム水溶液 lmlを加え、精製水で全量を 100mlとし、 100mlあたり 10mgの化合物を含有する懸濁剤を製造する。 The compound obtained in Example 4-35 and carboxymethylcellulose sodium are uniformly mixed in a mortar, and a solution obtained by dissolving saccharin sodium in purified water is added little by little and mixed well. Add 1 ml of 10% aqueous sodium benzoate solution, make up to 100 ml with purified water, and prepare a suspending agent containing 10 mg of compound per 100 ml.
実施例 4-35で得られた化合物 1 Omg Compound 1 Omg obtained in Example 4-35
カルポキシメチルセルロースナトリウム 2. 0 g Sodium carboxymethyl cellulose 2.0 g
サッカリンナトリウム 4 Omg Saccharin sodium 4 Omg
安息香酸ナトリウム 10 Omg Sodium benzoate 10 Omg
全量 100m l Total volume 100ml
試験例 1 Test example 1
軟骨プロテオダリ力ン分解抑制活性の測定法 A method for measuring the activity of inhibiting the degradation of cartilage proteodarin
屠殺した牛から鼻軟骨を無菌的に摘出し、 5%熱不活化牛胎児血清、 ペニシリン G 100単位 Zml、 ストレプトマイシン 100単位 Zml、 を含むダルベッコ改変ィー ダル培地/ハム F12培地 1 : 1混合培地を用いて 37°C、 5%炭酸ガス、 95%空気の 条件下で 3日間前培養した。 前培養終了後、重さが約 2 mgの軟骨小片を作製し、 ヒト組替え体インターロイキン -l i3 (IL-1 :ゼンザィム社製) 及び試験化合物 を加えた I OO Uの培養液に移し、 さらに 4日間培養した。 培養終了後、 培養液 に遊離したプロテオダリカンと軟骨小片残ったプロテオダリカンに含まれる硫酸 化グリコサミノダリカンをそれぞれ 1, 9 -ジメチルメチレンブル一を用いるファ ルンデル ·アール■ダブリュらの方法 [フ 7ルンデル 'アール'ダブリュ (Farndal e, R. W. ) ら、 コネクティブ ·ティッシュー ' リサーチ (Connect. Tissue Res. ) , 9 巻、 247- 248頁、 1982年] によって定量し、 プロテオダリカン分解率を求めた。 なお、対照化合物として Z - D B H (デブロモヒメ二アルジシン) を用いた。 試験 化合物および対照化合物のプロテオダリカン分解抑制活性は、 以下の式により求 めた。 プロテオダリカン分解抑制活性 (%) = 100 X (Pi -Ps) / (Pi -Pc) Aseptic removal of nasal cartilage from slaughtered cows, Dulbecco's modified serum containing 5% heat-inactivated fetal calf serum, 100 units of penicillin G Zml, and 100 units of streptomycin Zml Preculture was performed for 3 days in a 1: 1 mixed medium of Dull medium / Ham F12 medium at 37 ° C, 5% carbon dioxide, and 95% air. After completion of the preculture, a cartilage piece weighing about 2 mg was prepared, and transferred to a culture medium of IOOU containing human recombinant interleukin-li3 (IL-1: manufactured by Zenzym) and a test compound. Culture was continued for another 4 days. After completion of the culture, the proteodalican released into the culture medium and the sulfated glycosaminodalican contained in the proteodalican remaining in the cartilage fragments are each treated with 1,9-dimethylmethylene blue using the method of Farundel-Arl-Double et al. [Farndal e, RW, et al., Connective Tissue Research, Connect. Tissue Res., Volume 9, pp. 247-248, 1982] to determine the rate of proteodalican degradation. I asked. In addition, Z-DBH (debromohymenialdicine) was used as a control compound. The proteodalican degradation inhibitory activity of the test compound and the control compound was determined by the following formula. Proteodalican degradation inhibitory activity (%) = 100 X (Pi -Ps) / (Pi -Pc)
Pc: IL-l i3及び試験化合物いずれも含まない時のプロテオダリカン分解率 Pc: Proteodalican degradation rate when neither IL-li3 nor test compound is contained
Pi: IL - 1 )3を加えた時のプロテオダリカン分解率 Pi: Proteodalican degradation rate when IL-1) 3 is added
Ps: IL-l j3及び試験化合物の両方を加えた時のプロテオダリカン分解率 試験例 2 Ps: Proteodalican degradation rate when both IL-l j3 and test compound were added Test Example 2
軟骨コラーゲン分解抑制活性の測定法 Method for measuring cartilage collagen degradation inhibitory activity
屠殺した牛から鼻軟骨を無菌的に摘出し、 5%熱不活化牛胎児血清、 ペニシリン G 100単位 Ziiil、 ストレプトマイシン 100単位 Zml、 を含むダルべッコ改変ィ一 ダル培地 Zハム F12培地 1 : 1混合培地を用いて 37° (:、 5%炭酸ガス、 95%空気の 条件下で 3日間前培養し、 重さが約 2 mgの軟骨片を作製した。 その軟骨片をヒ ト組替え体インタ一ロイキン- 1 )3 (IL-l iS :ゼンザィム社製) 及びヒト組替え体 , オンコスタチン M (0SM:ゼンザィム社製) を加えた 100 1の培養液で 7日間培 養後、培地及び添加物を更新し、 さらに 7日間培養した。 培養終了後、 培養液に 遊離したコラーゲンと軟骨小片残ったコラーゲンに含まれるビドロキシプロリン をそれぞれクロラミン- T及び P-ジメチルァミノべンズアルデヒドを用いるベル ガム ·アイらの方法 [ベルガム ·アイ (Bergam, I.) ら、 アナリティカル ·バイ オケミストリー (Anal. Biochem.) 35卷、 1961- 1965頁、 1963年] によって定 量し、 コラーゲン分解率を求めた。 なお、 対照化合物として Z-DBH (デブロモ ヒメ二アルジシン) を用いた。 被検化合物のコラーゲン分解抑制活性は、 以下の 式により求めた。 コラーゲン分解抑制活性 ( ) =100 X (Ci-Cs)/ (Ci-Cc) Nasal cartilage is aseptically removed from the slaughtered cattle, and a Dulbecco's modified medium containing 5% heat-inactivated fetal bovine serum, 100 units of penicillin G Ziiil, and 100 units of streptomycin Zml, Zham F12 medium 1: (1) Pre-cultured for 3 days in a mixed medium at 37 ° (5% CO 2, 95% air) to produce a cartilage fragment weighing about 2 mg. After culturing for 7 days in a 100 1 culture medium supplemented with interleukin-1) 3 (IL-liS: manufactured by Zenzam) and human recombinant, Oncostatin M (0SM: manufactured by Zenzam), the medium was added to the medium. The material was renewed and cultured for another 7 days. After completion of the culture, the collagen released in the culture solution and the hydroxyloxyproline contained in the collagen remaining in the cartilage fragments were removed using chloramine-T and P-dimethylaminobenzaldehyde, respectively. The method of Gum-Ie et al. [Bergam, I. et al., Analytical Biochemistry (Anal. Biochem.) 35, 1961-1965, 1963] to determine the rate of collagen degradation. I asked. In addition, Z-DBH (debromohymenialdisine) was used as a control compound. The collagen decomposition inhibitory activity of the test compound was determined by the following equation. Collagen degradation inhibitory activity () = 100 X (Ci-Cs) / (Ci-Cc)
Cc: IL - 1 β、 0SM及び試験化合物いずれも含まない時のコラーゲン分解率 Cc: Collagen degradation rate when neither IL-1β, 0SM nor test compound is contained
Ci: IL-1 β及び 0SMを加えた時のコラーゲン分解率 Ci: Collagen degradation rate when IL-1β and 0SM are added
Cs: IL-1 β、 0SM及び試験化合物の両方を加えた時のコラ一ゲン分解率 試験例 3 Cs: Collagen degradation rate when both IL-1β, 0SM and test compound were added Test Example 3
マトリックスメ夕ロプロティナーゼ - 13 (MMP-13) 産生抑制活性の測定法 試験化合物の ΜΜΡ- 13産生抑制活性は正常ヒト膝関節軟骨細胞 (Clonetics社製) を用いて調べた。 ブレツトキット CGM (Clonetics社製)を用いて培養した正常ヒ ト膝関節軟骨細胞を 1X105個 Zmlとなるように 0.1%熱不活化牛胎児血清、ベニ シリン G100単位 Zml、 ストレプトマイシン 100単位 Zmlを含むダルベッコ改変 イーグル培地/ハム F12培地 1 : 1混合培地に懸濁後、 8ゥエルプレートに 0.5 mlZゥエルずつ播種し、 37 、 5%炭酸ガス、 95 空気の条件下で 1 日間前培養した。 前培養終了後、 各ゥエルに 0.5 mlの予め 37°Cに暖めたダルベッコ改変ィ一ダル 培地ノハム F12培地 1: 1混合培地および Ν,Ν-dimethyliormamideに溶解した試 験化合物を l l添加し、 さらに 30分後に IL-liS (終濃度 lOngZml) を加えた。 これを 37°C、 5%炭酸ガス、 95%空気の条件下で 2日間培養後、 培養液中に産生さ れた MMP-13を MMP- 13 ERISAシステム (アマシャムバイオサイエンス社製) を用 いて定量した。 試験化合物の MMP- 13産生抑制活性は、 以下の式により求めた。 Method for Measuring the Activity of Inhibiting the Production of Matrix Medium Proteinase-13 (MMP-13) The activity of the test compound to inhibit the production of ΜΜΡ-13 was examined using normal human knee articular chondrocytes (Clonetics). Dulbecco's Buretsutokitto CGM (Clonetics Corp.) normal human knee articular chondrocytes to 1X10 5 cells ZML become as 0.1% heat inactivated fetal bovine serum and cultured with, Beni cylindrical G100 units ZML, streptomycin 100 units ZML After suspension in a modified Eagle medium / ham F12 medium 1: 1 mixed medium, the cells were inoculated in 0.5 ml Z-wells in an 8-well plate and pre-cultured for 1 day under the conditions of 37, 5% carbon dioxide, and 95 air. After completion of the preculture, add 0.5 ml of the test compound dissolved in 0.5 ml of Dulbecco's modified idal medium, Noham F12 medium 1: 1 mixed medium and Ν, Ν-dimethyliormamide, to each well, pre-warmed to 37 ° C. Thirty minutes later, IL-liS (final concentration lOngZml) was added. After culturing the cells for 2 days at 37 ° C, 5% carbon dioxide, and 95% air, the MMP-13 produced in the culture solution was purified using the MMP-13 ERISA system (Amersham Biosciences). Quantified. The MMP-13 production inhibitory activity of the test compound was determined by the following formula.
MMP- 13産生抑制活性 (%) =100 X (Mi -Ms)/ (Mi-Mc) ' MMP-13 production inhibitory activity (%) = 100 X (Mi -Ms) / (Mi-Mc) ''
Mc: IL-li3及び試験化合物いずれも含まない時の MMP- 13産生量 Mc: MMP-13 production when neither IL-li3 nor test compound is contained
Mi: を加えた時の MMP- 13産生量 Ms : IL- 1 /3及び試験化合物の両方を加えた時の MMP- 13産生量 試験結果 Mi: MMP-13 production when adding Ms: MMP-13 production amount when both IL-1 / 3 and test compound were added Test result
上記試験例 1、 2および 3における成績を%抑制率で示し、以下の表に示す。% 抑制率が高いほどプロテオダリカン分解抑制効果、 コラーゲン分解抑制効果、 M M P— 1 3産生抑制効果が高いことを意味する。 The results in the above Test Examples 1, 2 and 3 are shown in% inhibition rate and are shown in the following table. % The higher the inhibition rate, the higher the proteodalican degradation inhibitory effect, collagen degradation inhibitory effect, and MMP-13 production inhibition effect.
〔表 1 1 8〕 (Table 1 18)
軟骨プロテオダリカン分解抑制活性 (%抑制率) Cartilage proteodalican degradation inhibitory activity (% inhibition rate)
卖 ^ /旆JUi例 1 化 1 Li^ 1 "物 ΎJ 10 // M VI 1 f〃Jj M V 卖 ^ / hiruJUi Example 1 1 Li ^ 1 "thing ΎJ 10 // M VI 1 f〃Jj M V
Λ . Λ Λ.
I Δ I Δ
■¾ 0 i丄 ■ ¾ 0 i 丄
Λ - Λ-
4-7 100 56 4-7 100 56
4-8 99 4-8 99
4-9 96 4-9 96
4-10 98 4-10 98
4-26 81 4-26 81
4-34 97 4-34 97
4-35 98 4-35 98
4-6l(a) 84 4-6l (a) 84
4-62 87 4-62 87
7-58 91 7-58 91
7-72 90 7-72 90
Z-DBH 94 36 0 〔表 1 1 9〕 Z-DBH 94 36 0 (Table 1 1 9)
軟骨コラーゲン分解抑制活性 (%抑制率) Cartilage collagen degradation inhibitory activity (% inhibition rate)
〔表 1 2 0〕 (Table 12)
MMP-13産生抑制活性 (%抑制率) MMP-13 production inhibitory activity (% inhibition rate)
産業上の利用可能性 Industrial applicability
一般式 ( I ) で表される化合物またはその塩、 たとえば上記試験例 1 2およ び 3で例示された化合物などは I L一 1 3によって引き起こされる軟骨基質の分 解を抑制する作用、 MM P— 1 3産生抑制作用を示すことから優れた軟骨破壊抑 制作用を有するため、 軟骨疾患 (例えば、 変形性膝関節炎、 慢性関節リウマチ、 およびそれらの類似疾患における関節軟骨の破壊等) の予防、 治療に用いること ができ、 ひいては関節軟骨の疲弊あるいは破壊による疼痛などにより日常生活に 支障をきたしている患者のクオリティ一ォブライフを改善することが可能である。 The compound represented by the general formula (I) or a salt thereof, for example, the compounds exemplified in the above Test Examples 12 and 3 are used for analyzing cartilage matrix caused by IL-13. Since it has an excellent inhibitory effect on cartilage destruction due to its ability to inhibit solution and inhibit MMP-13 production, it can be used for cartilage disorders (eg, joints in knee osteoarthritis, rheumatoid arthritis, and similar diseases). It can be used for the prevention and treatment of cartilage destruction, etc., and, in turn, can improve the quality of life of patients who are impaired in daily life due to pain caused by fatigue or destruction of articular cartilage.
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2001-144608 | 2001-05-15 | ||
| JP2001144608 | 2001-05-15 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2002092606A1 true WO2002092606A1 (en) | 2002-11-21 |
| WO2002092606A8 WO2002092606A8 (en) | 2002-12-19 |
Family
ID=18990527
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2002/004640 Ceased WO2002092606A1 (en) | 2001-05-15 | 2002-05-14 | Fused imidazolidine derivatives, process for preparation of the same and use thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2002092606A1 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004069999A3 (en) * | 2003-02-04 | 2004-10-07 | Takeda Chemical Industries Ltd | Screening method |
| WO2009084653A1 (en) | 2007-12-28 | 2009-07-09 | Takeda Pharmaceutical Company Limited | Method for screening of cell-protecting agent |
| US7964615B2 (en) | 2005-05-20 | 2011-06-21 | Almirall, S.A. | Derivatives of 4-(2-amino-1-hydroxyethyl)phenol as agonists of the β2 adrenergic receptor |
| US8178679B2 (en) | 2007-11-28 | 2012-05-15 | Almirall, S.A. | Derivatives of 4-(2-amino-1-hydroxyethyl)phenol as agonists of the β2 adrenergic receptors |
| US8524908B2 (en) | 2009-03-12 | 2013-09-03 | Almirall, S.A. | Process for manufacturing 5-(2-{[6-(2,2-difluoro-2-phenylethoxy)hexyl]amino}-1-hydroxyethyl)-8-hydroxyquinolin-2(1H)-one |
| US9067922B2 (en) | 2013-04-19 | 2015-06-30 | Pfizer Limited | Chemical compounds |
| US9108918B2 (en) | 2011-10-07 | 2015-08-18 | Almirall, S.A. | Process for preparing 5-(2-{[6-(2,2-difluoro-2-phenylethoxy)hexyl]amino}-1(R)-hydroxyethyl)-8-hydroxyquinolin-2(1H)-one via a novel intermediate |
| US9346759B2 (en) | 2012-03-20 | 2016-05-24 | Almirall, S.A. | Polymorphic crystal forms of 5-(2-{[6-(2,2-difluoro-2-phenylethoxy)hexyl]amino}-1-(R)-hydroxyethyl)-8-hydroxyquinolin-2(1H)-one, heminapadisytlate as agonist of the β2 adrenergic receptor |
| WO2018208557A1 (en) | 2017-05-10 | 2018-11-15 | Arixa Pharmaceuticals, Inc. | 3-(((((2s,5r)-2-carbamoyl-7-oxo-1,6-diazabicyclo[3.2.1]octan-6-yl)oxy)sulfonyl)oxy)-2,2-dimethylprop noate derivatives and related compounds as perorally administered profrugs of beta-lactamase inhibitors for treating bacterial infections |
| WO2019070595A1 (en) | 2017-10-02 | 2019-04-11 | Arixa Pharmaceuticals, Inc. | Aztreonam derivatives and uses thereof |
| CN110156620A (en) * | 2019-07-02 | 2019-08-23 | 中节能万润股份有限公司 | A kind of preparation method of tranexamic acid |
| WO2020072442A1 (en) | 2018-10-01 | 2020-04-09 | Arixa Pharmaceuticals, Inc. | Derivatives of relebactam and uses thereof |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4179276A (en) * | 1977-02-01 | 1979-12-18 | E. I. Du Pont De Nemours And Company | Novel imidazothiazine-1,3 (2H)-diones |
| WO1993025555A1 (en) * | 1992-06-12 | 1993-12-23 | Farmitalia Carlo Erba S.R.L. | Derivatives of imidazo [5,1-c][1, 4]benzoxazin-1-one as 5 ht3 antagonists |
| WO1995032208A1 (en) * | 1994-05-25 | 1995-11-30 | Pharmacia S.P.A. | IMIDAZOLYLALKYL DERIVATIVES OF IMIDAZOL(5,1-c)(1,4)BENZOXAZIN-1-ONE AND THEIR USE AS THERAPEUTIC AGENTS |
| AU3106997A (en) * | 1996-06-14 | 1998-01-07 | Kaken Pharmaceutical Co., Ltd. | Bicyclic hydantoin derivatives, intermediates for the preparation thereof, process for the preparation of them, and herbicides containing the same as active ingredient |
| WO1998039313A1 (en) * | 1998-03-04 | 1998-09-11 | Monsanto Company | Thioaryl sulfonamide hydroxamic acid compounds |
| US6066658A (en) * | 1996-09-06 | 2000-05-23 | Takeda Chemical Industries, Ltd. | Condensed 4,5,6,7-tetrahydrobenzo[C]thiophenes as enhancer for cell differentiation induction factor action |
| WO2001030781A2 (en) * | 1999-10-20 | 2001-05-03 | Tanabe Seiyaku Co., Ltd. | INHIBITORS OF αLβ2 MEDIATED CELL ADHESION |
-
2002
- 2002-05-14 WO PCT/JP2002/004640 patent/WO2002092606A1/en not_active Ceased
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4179276A (en) * | 1977-02-01 | 1979-12-18 | E. I. Du Pont De Nemours And Company | Novel imidazothiazine-1,3 (2H)-diones |
| WO1993025555A1 (en) * | 1992-06-12 | 1993-12-23 | Farmitalia Carlo Erba S.R.L. | Derivatives of imidazo [5,1-c][1, 4]benzoxazin-1-one as 5 ht3 antagonists |
| WO1995032208A1 (en) * | 1994-05-25 | 1995-11-30 | Pharmacia S.P.A. | IMIDAZOLYLALKYL DERIVATIVES OF IMIDAZOL(5,1-c)(1,4)BENZOXAZIN-1-ONE AND THEIR USE AS THERAPEUTIC AGENTS |
| AU3106997A (en) * | 1996-06-14 | 1998-01-07 | Kaken Pharmaceutical Co., Ltd. | Bicyclic hydantoin derivatives, intermediates for the preparation thereof, process for the preparation of them, and herbicides containing the same as active ingredient |
| US6066658A (en) * | 1996-09-06 | 2000-05-23 | Takeda Chemical Industries, Ltd. | Condensed 4,5,6,7-tetrahydrobenzo[C]thiophenes as enhancer for cell differentiation induction factor action |
| WO1998039313A1 (en) * | 1998-03-04 | 1998-09-11 | Monsanto Company | Thioaryl sulfonamide hydroxamic acid compounds |
| WO2001030781A2 (en) * | 1999-10-20 | 2001-05-03 | Tanabe Seiyaku Co., Ltd. | INHIBITORS OF αLβ2 MEDIATED CELL ADHESION |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004069999A3 (en) * | 2003-02-04 | 2004-10-07 | Takeda Chemical Industries Ltd | Screening method |
| US7964615B2 (en) | 2005-05-20 | 2011-06-21 | Almirall, S.A. | Derivatives of 4-(2-amino-1-hydroxyethyl)phenol as agonists of the β2 adrenergic receptor |
| US8242177B2 (en) | 2005-05-20 | 2012-08-14 | Almirall, S.A. | Derivatives of 4-(2-amino-1-hydroxyethyl)phenol as agonists of the β2 adrenergic receptor |
| US8178679B2 (en) | 2007-11-28 | 2012-05-15 | Almirall, S.A. | Derivatives of 4-(2-amino-1-hydroxyethyl)phenol as agonists of the β2 adrenergic receptors |
| WO2009084653A1 (en) | 2007-12-28 | 2009-07-09 | Takeda Pharmaceutical Company Limited | Method for screening of cell-protecting agent |
| US8450070B2 (en) | 2007-12-28 | 2013-05-28 | Takeda Pharmaceutical Company Limited | Method for screening of cell-protecting agent |
| US8524908B2 (en) | 2009-03-12 | 2013-09-03 | Almirall, S.A. | Process for manufacturing 5-(2-{[6-(2,2-difluoro-2-phenylethoxy)hexyl]amino}-1-hydroxyethyl)-8-hydroxyquinolin-2(1H)-one |
| US9108918B2 (en) | 2011-10-07 | 2015-08-18 | Almirall, S.A. | Process for preparing 5-(2-{[6-(2,2-difluoro-2-phenylethoxy)hexyl]amino}-1(R)-hydroxyethyl)-8-hydroxyquinolin-2(1H)-one via a novel intermediate |
| US9346759B2 (en) | 2012-03-20 | 2016-05-24 | Almirall, S.A. | Polymorphic crystal forms of 5-(2-{[6-(2,2-difluoro-2-phenylethoxy)hexyl]amino}-1-(R)-hydroxyethyl)-8-hydroxyquinolin-2(1H)-one, heminapadisytlate as agonist of the β2 adrenergic receptor |
| US9067922B2 (en) | 2013-04-19 | 2015-06-30 | Pfizer Limited | Chemical compounds |
| WO2018208557A1 (en) | 2017-05-10 | 2018-11-15 | Arixa Pharmaceuticals, Inc. | 3-(((((2s,5r)-2-carbamoyl-7-oxo-1,6-diazabicyclo[3.2.1]octan-6-yl)oxy)sulfonyl)oxy)-2,2-dimethylprop noate derivatives and related compounds as perorally administered profrugs of beta-lactamase inhibitors for treating bacterial infections |
| WO2019070595A1 (en) | 2017-10-02 | 2019-04-11 | Arixa Pharmaceuticals, Inc. | Aztreonam derivatives and uses thereof |
| WO2020072442A1 (en) | 2018-10-01 | 2020-04-09 | Arixa Pharmaceuticals, Inc. | Derivatives of relebactam and uses thereof |
| CN110156620A (en) * | 2019-07-02 | 2019-08-23 | 中节能万润股份有限公司 | A kind of preparation method of tranexamic acid |
| CN110156620B (en) * | 2019-07-02 | 2022-04-15 | 中节能万润股份有限公司 | Preparation method of tranexamic acid |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2002092606A8 (en) | 2002-12-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1277754B1 (en) | Imidazopyridine derivatives | |
| CN112334466B (en) | Compounds | |
| US6403588B1 (en) | Imidazopyridine derivatives | |
| CN104487440B (en) | The substituted Imidazopyridazine of amino | |
| CN104507950B (en) | Thienopyrimidines | |
| CN101686967B (en) | Pyrazolopyridine Derivatives as NADPH Oxidase Inhibitors | |
| CN103717604B (en) | Replace aminooimidazole and pyridazine | |
| JP4456365B2 (en) | Compound | |
| CN105228982B (en) | 3-Acetylamino-1-(phenyl-heteroaryl-aminocarbonyl or phenyl-heteroaryl-carbonylamino)benzene derivatives for use in the treatment of hyperproliferative disorders | |
| CN103189373B (en) | The Triazolopyridine replaced | |
| WO2001056607A1 (en) | Integrin expression inhibitors | |
| CN104470931B (en) | Substituted benzothiophene pyrimidine | |
| JP2015501833A (en) | Sulfonamide derivatives | |
| JPWO2001056607A1 (en) | Integrin expression inhibitors | |
| CN102762563A (en) | Pyrazolo piperidine derivatives as nadph oxidase inhibitors | |
| CN107108554B (en) | 1-Cyclohexyl-2-phenylaminobenzimidazole as a MIDH1 inhibitor for tumor therapy | |
| CN106535891A (en) | Histone deacetylase inhibitors and compositions and methods of use thereof | |
| CN104321326A (en) | Amino-substituted imidazopyridazines | |
| WO2002092606A1 (en) | Fused imidazolidine derivatives, process for preparation of the same and use thereof | |
| WO2006080450A1 (en) | Igf-1r inhibitor | |
| CN104470926A (en) | Substituted pyrrolopyrimidines | |
| CN106458983A (en) | new compound | |
| WO2000023450A1 (en) | Fused pyridazine derivatives, process for the preparation of the same and uses thereof | |
| CN104837841A (en) | Substituted indazol-pyrrolopyrimidines useful in the treatment of hyperproliferative diseases | |
| CN101160125A (en) | Niacin receptor agonists, compositions containing such compounds and methods of treatment |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| AK | Designated states |
Kind code of ref document: C1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: C1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| CFP | Corrected version of a pamphlet front page |
Free format text: UNDER (54) PUBLISHED TITLE IN ENGLISH REPLACED BY CORRECT TITLE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| 122 | Ep: pct application non-entry in european phase | ||
| NENP | Non-entry into the national phase |
Ref country code: JP |