WO2002053548A1 - Benzothiazepine derivatives - Google Patents
Benzothiazepine derivatives Download PDFInfo
- Publication number
- WO2002053548A1 WO2002053548A1 PCT/JP2001/011267 JP0111267W WO02053548A1 WO 2002053548 A1 WO2002053548 A1 WO 2002053548A1 JP 0111267 W JP0111267 W JP 0111267W WO 02053548 A1 WO02053548 A1 WO 02053548A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- alkyl
- alkoxy
- amino
- rubamoyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D281/00—Heterocyclic compounds containing rings of more than six members having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D281/02—Seven-membered rings
- C07D281/04—Seven-membered rings having the hetero atoms in positions 1 and 4
- C07D281/08—Seven-membered rings having the hetero atoms in positions 1 and 4 condensed with carbocyclic rings or ring systems
- C07D281/10—Seven-membered rings having the hetero atoms in positions 1 and 4 condensed with carbocyclic rings or ring systems condensed with one six-membered ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
- the present invention is clearly useful in the field of medicine. More specifically, since the benzothiazepine derivative of the compound of the present invention has an activity of exhibiting a high blood GLP-1 concentration, it is useful as a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes or an antiobesity agent. is there. Background technology
- the blood sugar level of a healthy person is constantly controlled by the action of insulin. Diabetes refers to a chronic hyperglycemic state in which this control is ineffective and a disease caused by it.
- diabetes treatment The basis of diabetes treatment is to correct hyperglycemia, i.e., to return blood glucose levels to normal levels.In recent years, however, it has been particularly important in recent years to prevent a rapid rise in blood glucose after eating without affecting fasting blood glucose. It has been recognized that it is extremely important in terms of treatment to control the amount of the drug.
- the first is a group of drugs called insulin-releasing drugs typified by sulfonylprea drugs, which promote insulin secretion directly from the brain and lower blood sugar levels.
- the second is a drug that has recently been launched, called an insulin sensitizer, that lowers blood glucose by promoting glucose uptake in peripheral tissues without directly promoting insulin release. is there.
- the third monoglucosidase inhibitor is a drug that controls the rapid rise in blood glucose by delaying the digestion and absorption of carbohydrates in the gastrointestinal tract and suppressing the temporary rise in blood glucose after meals. is there. .
- GLP-1 glucagon-like peptide-1
- L cells which are endocrine cells present in the small intestinal intestinal epithelium due to intensification, etc., and acts on ⁇ cells present in the islets of Langerhans to promote the secretion of insulin to lower blood sugar.
- GLP_1 insulin secretion by GLP_1 depends on blood glucose level.Insulin secretion by GLP-1 is not observed in normoglycemia, and it is reported that insulin secretion is enhanced only in hyperglycemia.
- GLP-1 not only enhances insulin secretion but also enhances insulin biosynthesis (Endocri no lgygy, 130, 159, 1992) and promotes the proliferation of] 3 cells (Diabeto 1 ogia, Vol. 42, p. 856, 1999), which is an essential factor for maintaining 8 cells.
- GLP-1 gastrointestinal motility inhibitory effect
- Metatab. D is pos., 27, p. 637 , 1999 (hereinafter referred to as Reference I), Curr. Opin. Cardiova sc., Renal Instst Drugs, 1, p. 276, 1999 (hereinafter referred to as Reference J). ), J. Lipid Res., 40, p. 2158, 1999 (hereinafter referred to as Reference K), Anal. Biochem., 282, p. 94, 2000 (hereinafter referred to as Reference L). ), International Publication No. WOO 0Z38725 (hereinafter referred to as Reference M), International Publication No. WO00 / 38726 (hereinafter referred to as Reference N), and International Publication No.
- WO00 / 38727 (hereinafter referred to as Reference 0). ), International Publication Number WO00 / 3 No. 8728 (hereinafter referred to as Reference P), International Publication No. WO 00/38729 (hereinafter referred to as Reference Q), and International Publication No. WO00 / 55355 (hereinafter referred to as Reference R). ing.
- references A to R describe compounds having a 1,4-benzothiazepine skeleton.
- the 1,4-benzothiazepine 1,1-dioxoside skeleton is common to the compounds of References A to R, the 3-position substituent on the skeleton is a monosubstituted compound.
- References A to R are hypolipidemic compounds caused by the inhibitory action of bile acid uptake, and have the same industrial application fields as the present invention, but are completely different from the uses of the present invention. This is an unrelated application. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
- Reference S discloses a 1,4-benzothiazepine skeleton and its oxidized and reduced forms, specifically, 1,4-benzen. Scaffolds such as 4,5-dihydro, 4,5-dihydro, N-oxidized, and 4,5-year-old oxaziridine derivatives of the zothiazepine skeleton are described.
- the compounds of the present invention have a 1,4-benzothiazepine skeleton in common with the compound of Reference J
- the 4-benzodiazepine skeleton has a 4-dihydro form, a 4-dihydroform, an N-oxidized form, and a 4,5-year-old compound.
- Oxaziridines are compounds with completely different structures outside the scope of the present invention.
- the use of Reference S is a compound having a muscle relaxant and anticonvulsant activity and has the same industrial field of application, but is completely unrelated to the use of the present invention. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
- Reference T discloses a three-ring fused ring containing a 1,4-benzothiazepine skeleton, pyrrole [1,2-d]-. Compounds having a 1,4-benzothiazepine skeleton have been described.
- Reference T is a condensed tricyclic ring, and is a compound having a completely different structure outside the scope of the present invention.
- the use of Reference T is a compound having a locomotor activity-suppressing action, and although it has the same industrial field of application, it has no relation to the use of the present invention. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
- Reference U discloses a cheno [3,2-f] _1,4,1-benzothiazepine containing a 1,4-benzothiazepine skeleton. Compounds having a skeleton are described.
- Reference U is a compound in which the substituent at the 5-position on the skeleton has hydrogen, a straight-chain C 5 alkyl group or a C 3 -C 7 cyclic alkyl group, and the compound of the present invention has a substituent.
- the compound has a completely different structure in that it may have an aromatic ring or a heterocyclic ring. Although it has the same industrial field of application as a drug for suppressing intraocular pressure rise and for treating glaucoma, it is a use that has no relation to the use of the present invention. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
- antidiabetic drugs such as sulfonylprea drugs, insulin resistance Drugs such as sex-improving agents and mono-dalcosidase inhibitors are widely used in the clinic, but have the following problems, and they cannot be said to be sufficient drugs.
- sulfonylurea agents have a slow onset of action and a long duration of action, making it difficult to develop a synergistic effect during postprandial hyperglycemia, and also lower fasting blood sugar and are often life-threatening Such severe hypoglycemic attacks can be caused.
- Insulin sensitizers often have side effects on the liver and require careful use under strict control. It may also cause edema and other side effects. In addition, side effects such as bloating and diarrhea have become a problem with Hi-Dalcosidase inhibitors.
- An object of the present invention is to provide a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes, an anti-obesity drug, and the like, since they exhibit an activity of exhibiting a high blood GLP-1 concentration.
- the present inventors have intensively studied for the purpose of creating a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes, or an anti-obesity agent capable of controlling blood glucose level depending on the blood glucose level. ]
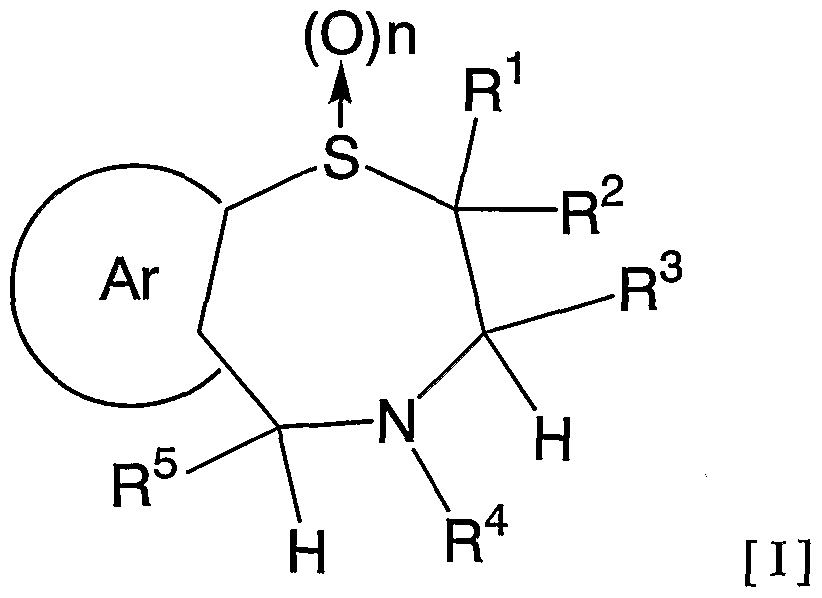
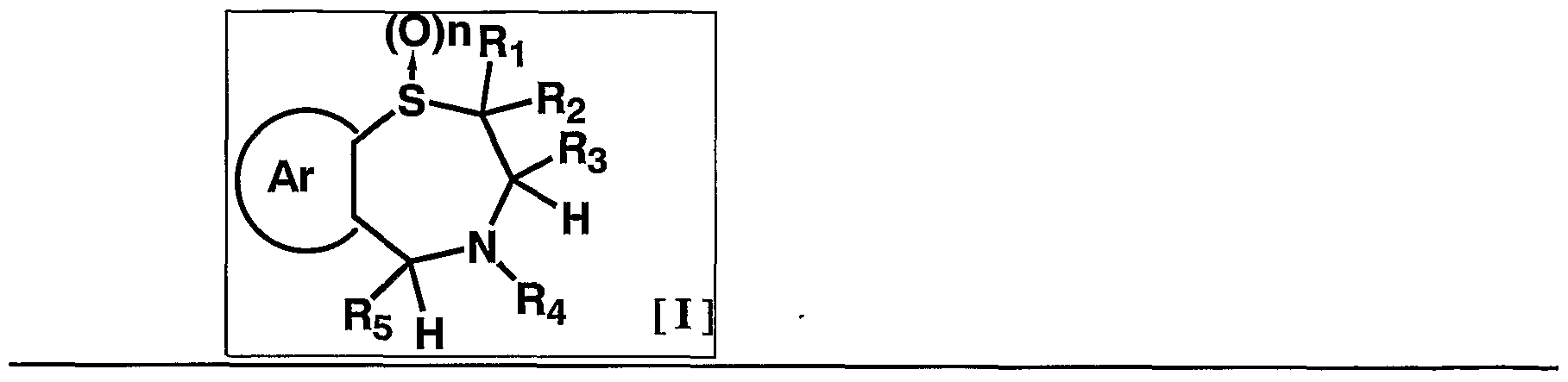
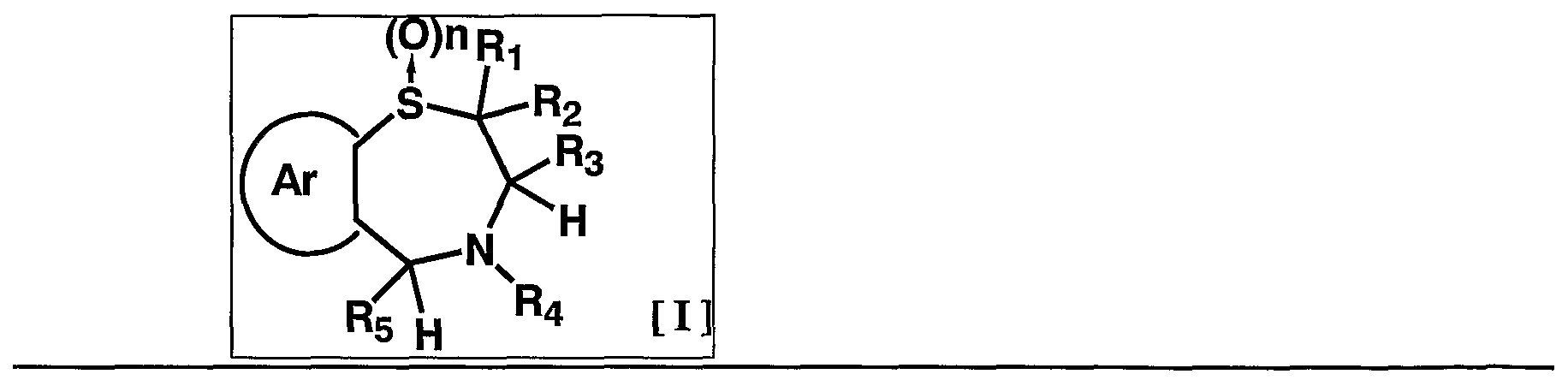
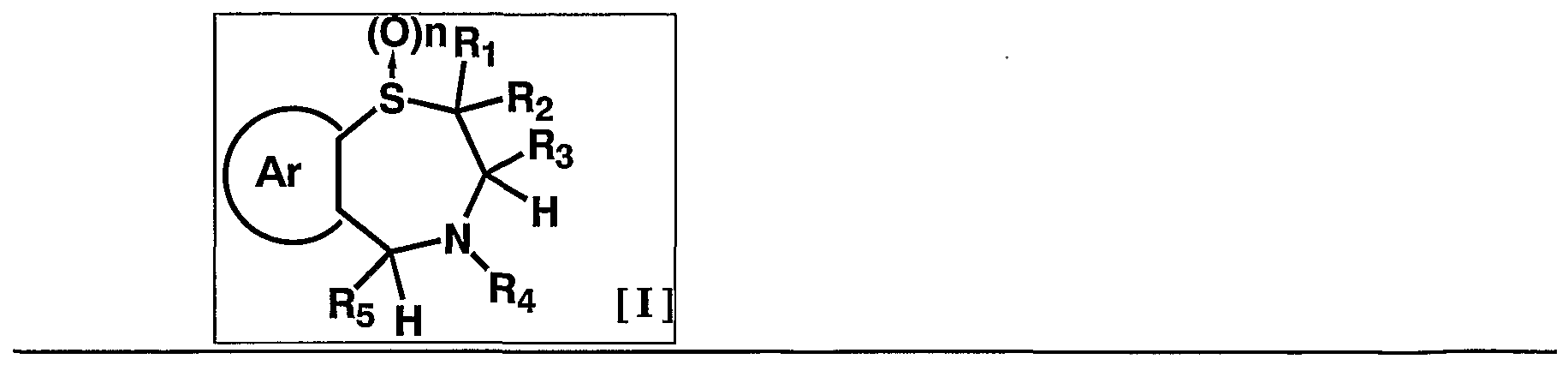
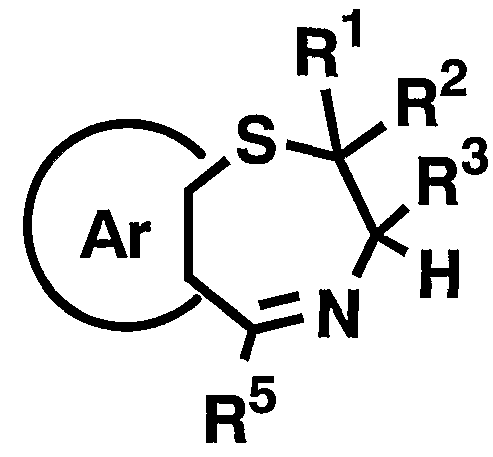
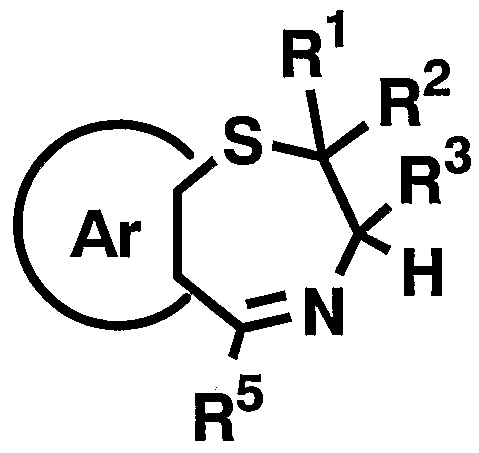
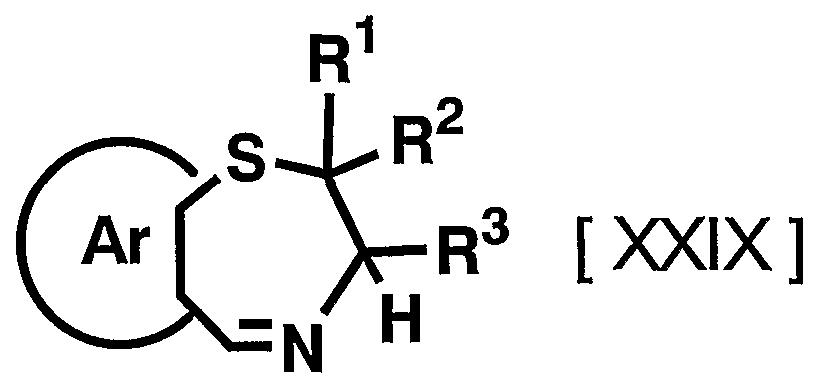
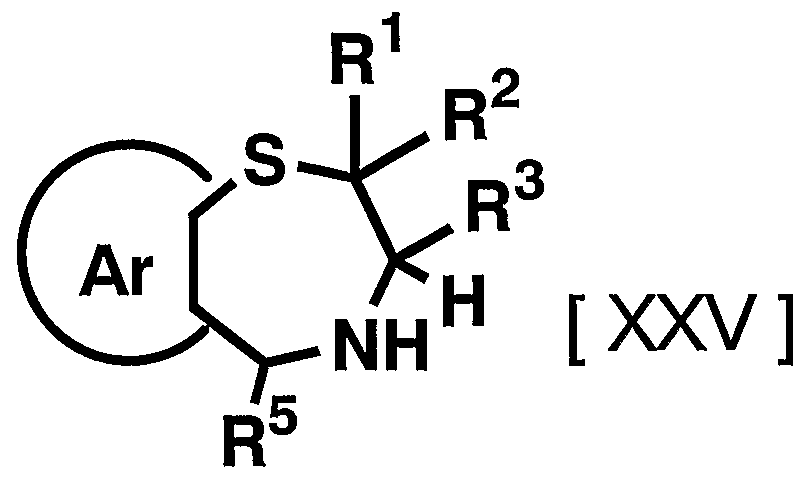
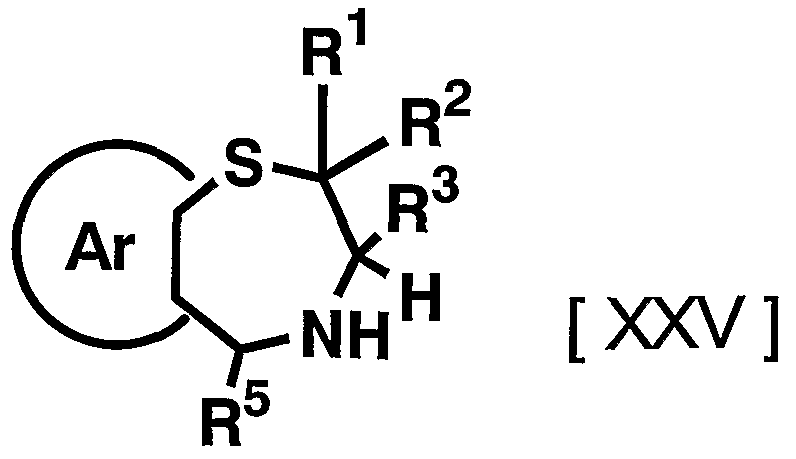
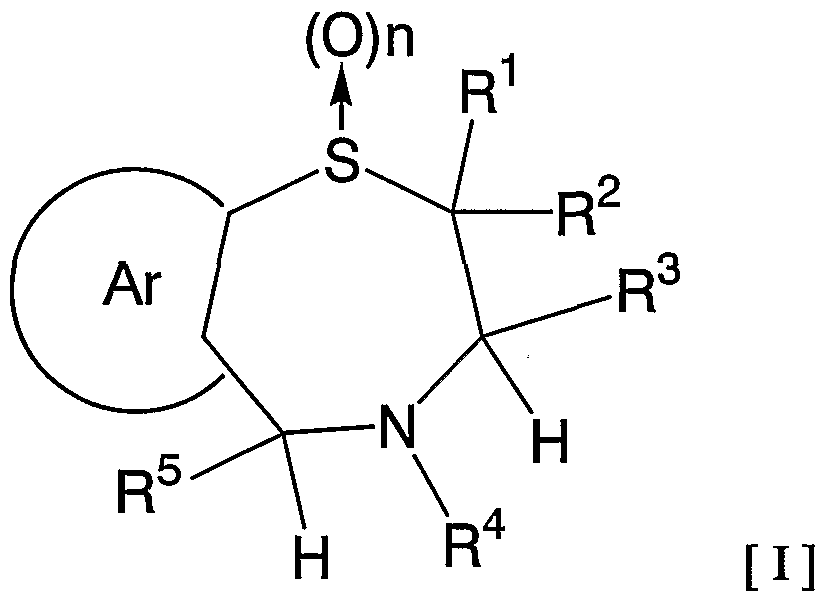
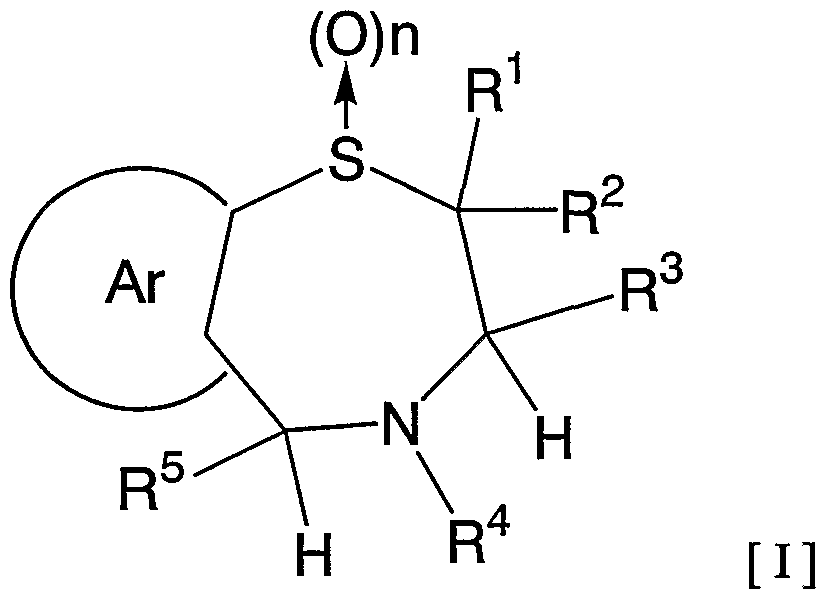
- 1 and R 2 are the same or different and each represents a hydrogen atom, a C—C ⁇ alkyl group,
- R 3 is a hydrogen atom, a C, —C 6 alkyl group (excluding an n-butyl group), R 4 is a hydrogen atom, a hydroxyl group, a C 3 alkyl group,
- R 5 is a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, a carbamoyloxy group, a carboxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, a formyl group Group, formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkylcarbonyl group, N -Aralkyl groups: rubamoyl group, aryl group, N-arylamino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfonyl
- C i one C 6 alkoxy group, one C 6 alkylthio group, Nyu- - C 6 alkylamino group, N-CI- C 6 alkyl force Rubamoiru group, N-Ci-C 6 alkyl
- a carbon atom which may have a substituent selected from the group consisting of thiocarbamoyl groups; Kowamoto, 1 to tricyclic C 7-C 15 carbon ring group or a 5- or 6-membered heterocyclic group or a nitrogen atom, a heteroatom 1 ring system selected from the group consisting of SansoHara bar and a sulfur atom 1 to 5 mono- to 3-cyclic heteroaromatic groups (excluding 5- or 6-membered heterocyclic groups),
- Ar represents a hydrogen atom, an azide group, an amino group, a rubamoyl group, a rubamoylamino group, a rubamoyloxy group, a lipoxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, a formyl group, Formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkylcarbonyl group, N -Aralkyl groups: rubamoyl group, aryl group, N-aryl amino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfonyloxy group,
- Alkyl rubamoyl groups N, N-di-I. Alkyl Scarpa model I le group, N- C 2 - C 6 alkenyl carbamoylmethyl group, N, N- di C 2 -. C 6 alkenyl carbamoylmethyl group, N- amino ( ⁇ primary alkyl force Rubamoiru group, N- one C 6 Alkoxy G 10 Alkyl Lubamoyl group, NC x -C 6 Alkoxy Luponyl C! -1 C.
- Alkyl Lubamoyl group N-C "C 6 Alkoxy Lubamoyl group C 10 Alkyl Lubamoyl group, NC i -Ce alkoxycarbonylamino ( ⁇ — Ce alkoxycarbonyl group, 1 C 6 alkylthio group, NC i 1 C 6 alkylsulfamoyl group, N, N-di C — C 6 alkylsulfamoyl group, C!
- C 6 alkyl force Rubamoiru group has a NC-Ce. alkylthio force Rubamoiru substituents selected from the group ing from the base which may be a carbon aromatic ring group, 1 to 3 rings of the C 7 - C 15 carbon ring group or 5 young Or a 6-membered heterocyclic group, or a 2- or 3-cyclic fused heteroaromatic group having 1 to 5 heteroatoms per ring system selected from the group consisting of nitrogen, oxygen and sulfur , And 5 or 6 membered heterocyclic groups are excluded.), And n is an integer of 0 to 2.] It has been found that the compound represented by the formula (1) achieves a high blood GLP-1 concentration in vivo. Completed the invention.
- the present invention relates to benzothiazepine derivatives and uses thereof, and these inventions are novel ones not described in the literature. Next, various symbols and terms described in this specification will be described.
- the C er C 3 alkyl group such as methyl group, Echiru group, a propyl group can be mentioned, et al are, inter alia methyl, and the like Echiru group.
- a C 6 alkyl group includes, for example, a methyl group, an ethyl group, a propyl group, an isopropyl group, an isobutyl group, a sec-butyl group, a tert-butyl group, a pentyl group, a neopentyl group, a hexyl group, and an isohexyl group.
- isopropyl group, isobutyl group, sec-butyl group, neopentyl group, isohexyl group and the like are preferable.
- halogen atom examples include a fluorine atom, a chlorine atom, a bromine atom and an iodine atom, among which a fluorine atom, a chlorine atom and an iodine atom are preferred, and a fluorine atom and a chlorine atom are more preferred.
- the cyclic saturated or unsaturated C 3 -C 9 aliphatic group includes a cyclic alkyl group or alkenyl group having 3 to 9 carbon atoms, among which a cyclic alkyl group having 3 to 6 carbon atoms. Or an alkenyl group, and more preferably a cyclic alkyl group having 3 to 6 carbon atoms.
- cyclic alkyl group examples include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclooctyl group, and a cyclononyl group.
- a tyl group and a cyclohexyl group are preferred.
- Examples of the cyclic alkenyl group include a cyclopropenyl group, a cyclobutenyl group, a cyclopentenyl group, a cyclohexenyl group, a cycloheptenyl group, a cyclohexenyl group, and a cyclononenyl group.
- a cyclopropenyl group, a cyclobutenyl group and the like are exemplified.
- Group, cyclopentenyl group, cyclohexenyl group and the like are exemplified.
- the aralkyl group means an aralkylyl group having 7 to 15 carbon atoms, specifically, for example, a benzyl group, a monomethylbenzyl group, a phenethyl group, a 3-phenylpropyl group, a 1-naphthylmethyl group. , 2-naphthylmethyl group, ⁇ -methyl (1-naphthyl) methyl group, a-methyl (2-naphthyl) methyl group, ⁇ -ethyl (1naphthyl) methyl group, Q!
- the N-aralkylamino group means a group in which the above-mentioned aralkyl group is substituted on the amino group, and specifically, for example, N-benzylamino group, N- (1-methylbenzyl) amino group, N-phenethylamino group, N- (3-Phenylpropyl) amino group, N— (1-naphthylmethyl) amino group, N— (2-naphthylmethyl) amino group, N— [Hi-methyl (1-naphthyl) methyl] amino group, N— [1-methyl (2-naphthyl) methyl] amino group, N— [1-ethyl (1-naphthyl) methyl] amino group, N — [— ethyl (2-naphthyl) methyl] amino group, diphenylmethyl amino group, Examples include an N- (dinaphthylmethyl) amino group, and among them, an N-benzylamin
- the ⁇ , ⁇ -aralkylamino group means a group obtained by substituting the above aralkyl group for an amino group, and specifically, for example, ⁇ , ⁇ -dibenzylamino group, ⁇ , ⁇ -di (monomethyl) group.
- An aralkyloxy group means a group in which an oxygen atom is substituted by the above aralkyl group.
- the aralkylcarbonyl group means a group in which the aralkyl group is substituted on the carbonyl group, specifically, for example, a benzylcarbonyl group, a monomethylbenzylcarbonyl group, a phenethylcarbonyl group, Phenylpropyl carbonyl group, 1-naphthylmethyl carbonyl group, 2-naphthylmethyl carbonyl group, mono-methyl (1-naphthyl) methyl carbonyl group, ⁇ -methyl (2-naphthyl) methyl carbonyl group, a-ethyl (1-naphthyl) ) Methylcarbonyl group, methylethyl (2-naphthyl) methylcarbonyl group, diphenylmethylcarbonyl group, dinaphthylmethylcarbonyl group, etc., among which benzylcarbonyl group, 1-methylbenzylcarbonyl group,
- the N-aralkyl molybamoyl group means a group obtained by substituting the above-mentioned aralkyl group with a molybmoyl group, and specifically includes, for example, N-benzylcarbamoyl group, N- (monomethylbenzyl) molybmoyl group, Netyl carbamoyl group, N— (3-phenylphenyl) dirubamoyl group, N— (1-naphthylmethyl) dirubamoyl group, N— (2-naphthylmethyl) dirubamoyl group, N- ( ⁇ -methyl (1 Naphthyl) methyl) dirubamoyl group, N— (Hi-methyl (2-naphthyl) methyl) dirubamoyl group, N— (1-ethyl (1-naphthyl) methyl) dirubamoyl group, N- ( ⁇ -ethyl) 2-naph
- the aryl group means an aryl group having 6 to 15 carbon atoms, for example, a naphthyl group, a phenyl group and the like, among which a phenyl group and the like are preferable.
- arylamino means a group obtained by substituting the above-mentioned aryl group with an amino group, specifically, for example, a phenylamino group, a ⁇ (1-naphthyl) amino group, an ⁇ — (2— Naphthyl) amino group and the like, among which a ⁇ -phenylamino group and the like are preferable.
- the ⁇ , ⁇ -diarylamino group means a group obtained by disubstituting the above aryl group with an amino group.
- the aryloxy group means a group in which an oxygen atom is substituted by the above aryl group, and specific examples include a phenoxy group and a naphthyloxy group. Among them, a phenoxy group and the like are preferable.
- the arylsulfonyl group means a group in which the above-mentioned aryl group is substituted on the sulfonyl group, specifically, for example, a phenylsulfonyl group, a naphthylsulfonyl group and the like. Groups and the like are preferred.
- the arylsulfonyloxy group means a group in which a sulfonyloxy group is substituted with the above-mentioned aryl group, and specifically, for example, phenylsulfonyloxy group, naphthylsulfonyloxy group and the like. Among them, a phenylsulfonyloxy group and the like are preferable.
- the N-arylsulfonylamino group means a group in which the above-mentioned arylsulfonyl group is N_substituted for an amino group, specifically, for example, an N-phenylsulfonylamino group, (1_naphthylsulfonyl) amino group, N- (2-naphthylsulfonyl) amino group and the like are preferable, and among them, N-phenylsulfonylamino group and N- (2-naphthylsulfonyl) amino group are preferable.
- N- ⁇ Li one Le sulfonyl ⁇ amino d-C 6 alkylamino group, d-C 6 alkyl groups having on the Symbol of ⁇ Li one Le sulfonyl ⁇ amino group to Amino group means a N- substituted group Specifically, for example, N-phenylsulfonylaminomethylamino group, N- (1-phenylsulfonylaminoethyl) amino group, N— (2-phenylsulfonylaminoethyl) amino group, N-naphthylsulfonylamino Methylamino group, N- (1-naphthylsulfonylaminoethyl) amino group, N- (2-naphthylsulfonylaminoethyl) amino group and the like, among which N-phenylsulfonylaminomethylamino group, N- (2 —Phen
- the C 6 alkoxy Cal Poni group, a group of the above N- ⁇ Li one Le sulfonyl ⁇ amino group is substituted with one C 6 alkoxy Cal Poni Le group was meaning taste, specifically, for example N- phenylalanine sulfonylamino methoxy Cal Poni Le Group, N-naphthylsulfonylaminomethoxycarbonyl group, 1- (N-phenylsulfonylamino) ethoxycarbonyl group, 2- (N-phenylsulfonylamino) ethoxycarbonyl group, etc., among which N-phenylsulfonylaminomethoate Xycarbonyl group, N-naphthylsulfony
- the arylsulfamoyl group means a sulfamoyl group substituted with the above aryl group, and specifically includes, for example, a phenylsulfamoyl group, a naphthylsulfamoyl group and the like, and among them, a phenylsulfamoyl group and the like. preferable.
- the arylsulfamoyloxy group means a sulfamoyloxy group substituted with the above aryl group, specifically, for example, phenylsulfamoyloxy group, naphthylsulfayl group Moyloxy groups and the like can be mentioned, among which phenylsulfamoyloxy groups and the like are preferable.
- the arylsulfamoyl C 11 -C] _ 0 alkyl rubamoyl group means a group in which the above arylsulfamoyl is substituted with a d-C 10 alkyl rubamoyl group, and specifically, for example, phenylsulfamoyl Moylmethylcarbamoyl group, naphthyls And a phenylsulfamoylmethylcarbamoyl group.
- Li one Rusurufamoiru ( ⁇ - The C 6 alkoxy Cal Poni group, means a group above ⁇ Li one Rusurufamoiru has been substituted with d-C 6 alkoxy Cal Poni group, concrete in, for example Hue acylsulfamoyl methoxy Cal Poni Le group And a naphthylsulfamoylmethoxycarbonyl group, among which a phenylsulfamoylmethoxycarbonyl group and the like are preferable.
- the N-arylcarbamoyl group means a group in which the above aryl group is N-substituted with a carbamoyl group, and specifically includes, for example, a phenylcarbamoyl group, a naphthylcarbamoyl group and the like. And the like.
- An aroyl group means a group in which a carbonyl group is substituted with the above-mentioned aryl group, and specific examples thereof include a benzoyl group and a naphthylcarboel group, and among them, a benzoyl group and the like are preferable.
- the alkoxy group means a group in which the above-mentioned aroyl group is substituted by an oxygen atom, and specifically includes, for example, a benzoyloxy group, a naphthylcarponyloxy group and the like, and among them, a benzoyloxy group and the like are preferable.
- the N— (N-aroylamino) d _C 10 alkyl rubamoyl group means a group obtained by substituting a rubamoyl group with the above-mentioned N-aroylamino C 11 -Ci 0 alkyl group.
- N- (N-benzoylaminomethyl) rubamoyl group N- (1- (N-benzoylamino) ethyl) rubamoyl group, N— (2- (N-benzoyl) aminoethyl) rubamoyl group, N— (N-naphthylcarbonylaminomethyl) rubamoyl group, N— (1- (N-naphthylcarbonylamino) ethyl) rubamoyl group, N— (2- (N-naphthylcarbonylamino) ethyl) rubamoyl group Among them, an N- (N-benzoylaminomethyl) carbamoyl group and an N- (2- (N-benzoylamino) ethyl) rubamoyl group are preferable.
- the N-aroylamino d-Ce alkoxycarbonyl group means a group in which the above-mentioned N-aroylamino d-C 6 alkyl group is substituted for an oxycarbonyl group, and specifically, for example, an N-benzoylaminomethoxycarbonyl group , 1- (N-benzoylamino) ethoxycarbonyl group, 2_ (N-benzoylamino) ethoxycarbonyl group, N-naphthylcarbonylaminomethoxycarbonyl group, 1- (N-naphthylcarbonylamino) ethoxycarbonyl group, 2- (N-naphthylcarbonylamino) ethoxycarbonyl group and the like, among which an N-benzoylaminomethoxycarbonyl group and a 2_ (N-benzoylamino) ethoxycarbonyl group are preferable.
- the C 2 -C 6 alkanol group means a carbonyl group substituted with an alkyl group having 1 to 5 carbon atoms, and specifically includes, for example, an acetyl group, a propionyl group, a butyryl group, an isoptyryl group, and a valeryl group.
- acetyl group, propionyl group, pivaloyl group and the like are preferable.
- the C 2 —C 6 alkenyl group refers to a group in which the above-mentioned C 2 -C 6 alkynyl group is substituted for a hydroxyl group, specifically, for example, an acetyloxy group, a propionyloxy group, a butyryloxy group, Examples include an isoptyryloxy group, a valeryloxy group, an isovaleryloxy group, a pivaloyloxy group, and a pentanoyloxy group. Of these, an acetyloxy group, a propionyloxy group, and a vivaloyloxy group are preferable.
- the N—C 2 —C 6 alkanoylamino group refers to a group in which the above C 2 —C 6 alkanoyl group is substituted for an amino group, and specifically, for example, an N—acetylamino group, Mouth pionylamino, N-butyrylamino, N-isobutyrylamino, N-valerylamino, N-isovalerylamino, N-bivaloylamino, N-pentanoylamino, etc.
- N, N - The C 6 alkanoyloxy noisy Rua amino group, the Amino group of the C 2 - - di -C 2 C 6 Arukanoiru group means two substituted groups, and specific examples N, N-Jiasechi A lumino group, an N, N-dipropionylamino group, an N, N-dibutyrylamino group, N, N-diisobutyrylamino group, N, N-divalerylamino group, N, N-diisovalerylamino group, N, N-dipiparylylamino group, N, N-dipentanoylamino group, N-acetyl-N- A propionylamino group, an N-acetyl-N-butylylamino group, an N-acetyl-N-piparylylamino group and the like, among which N, N-diacetylamino, N, N-dipropion
- the N-Ci-C 6 alkylamino group means a group in which an amino group is substituted with an alkyl group having 1 to 6 carbon atoms, specifically, for example, N-methylamino group, N-ethylamino group, N- To propylamino group, N-isopropylamino group, N-butylamino group, N-isobutylamino group, N-sec-butylamino group, N-tert-butylamino group, N-pentylamino group, N-neopentylamino group, N- Xylamino group, N-isohexylamino group and the like.
- N-methylamino group, N-ethylamino group, N-propylamino group, N-isopropylamino group, N-butylamino group, N-isobutylamino group, — Tert-butylamino group and the like are preferable.
- the N, N-di-d-C 6 alkylamino group means a diamino-substituted alkyl group having 1 to 6 carbon atoms, specifically, for example, N, N-dimethylamino group, N, N-diethylamino, N, N-dipropylamino, N, N-diisopropylamino, N, N-dibutylamino, N, N-ditert-butylamino, N, N-dipentylamino, N, N-dihexylamino group, N-ethyl-N-methylamino group, N-methyl-N-propylamino group, N-isopropyl-N-methylamino group, N-tert-butyl-N-methylamino group, N-ethyl N-isopropylamino group and the like, among which N, N-dimethylamino group, N, N-diethylamino
- N—Ci—C 10 alkyl rubamoyl group means that the rubamoyl group does not have 1 carbon atom And a group substituted by 10 alkyl groups, specifically, for example, N-methylcarbamoyl, N-ethylcarbamoyl, N-propylcarbamoyl, N-isopropylcarbamoyl, Butylcarbamoyl, N-isobutyl carbamoyl, N-sec-butylcarbamoyl, N-tert-butylcarbamoyl, N-pentylcarbamoyl, N-neopentylcarbamoyl, N-hexylcarbamoyl, N-iso A xylcarbamoyl group, an N-octylcarbamoyl group, an N-decylcarbamoyl group, and the like.
- an N-methyl rubamoyl group an N-ethyl carbamoyl group, an N-propyl carbamoyl group, an N-isopropyl carbamoyl group, N-isobutylcarbamoyl group, N-sec-butyl carbamoyl group, N-tert-butyl Rubamoiru group, N- old Kuchiruka Rubamoiru group, N- dodecylcarbamoyl group and the like.
- the N, N-di-C i-C ioalkyl rubamoyl group refers to a group obtained by disubstituting a carbamoyl group with an alkyl group having 1 to 10 carbon atoms.
- N - C 2 - A C 6 alkenyl carbamoylmethyl group, having 2 to carbon Karupamoiru based means a group alkenyl group substituted consisting six, specifically for example, N- vinyl carbamoyl group, N- Ariru force Rubamoyl group, N- (1-probenyl) carbamoyl group, N-isoprobenylcarbamoyl group, N- (2-butenyl) carbamoyl group, N-isobutenylcarbamoyl group, N— (2-pentenyl) Cal Bamoyl group, N- (2-hexenyl) carpamoyl group, N- (2-heptenyl) -powered rubamoyl group, N- (2-octenyl) -powered rubamoyl group, etc., among which N-vinylcarbamoyl group, Preferred are an N-arylcarpamoyl group, an N- (1
- the N, N-di-C 2 -C 6 alkenylcarbamoyl group refers to a disubstituted alkamoyl group having 2 to 6 carbon atoms in a carbamoyl group.
- N- The Amino Ci one C 1 0 alkyl force Rubamoiru group, 1 -C force Rubamoiru based means a group aminoalkyl group consisting of 1 0-substituted, specifically, example if N- amino-methylcarbamoyl N-aminoethylcarbamoyl group, N-aminopropylcarbamoyl group, N-aminomethylethylcarbamoyl group, N-aminobutylcarbamoyl group, N-aminopropylcarbamoyl group, N-aminopropylcarbamoyl group, N-aminopentylcarbamoyl group, N— Examples include an aminohexylcarbamoyl group, and among them, an N-aminomethylcarbamoyl group, an N-aminoethylcarbamoyl group, an N-aminopropylcarbamoy
- N—d—C 6 alkoxy d—C 10 alkyl rubamoyl group refers to a group in which an alkoxy group having 1 to 10 carbon atoms is N-substituted to the above-mentioned C 1 -C 1 0 alkyl rubamoyl group.
- N-methoxymethylcarbamoyl, N-methoxyethylcarbamoyl, N-methoxypropylcarbamoyl, N-methoxybutylcarbamoyl, N-ethoxypentylcarbamoyl And N-butoxyhexylcarbamoyl group among which N-methoxymethylcarbamoyl group, N-methoxyethylcarbamoyl group, N-methoxypropylcarbamoyl group, N-methoxybutylcarbamoyl group and the like are preferable.
- N - a Ci o alkyl force Rubamoiru 1 -C based to an alkoxycarbonyl group composed of six N- substituted group - C i - Ce alkoxy Cal The Poni Le Ci _ C 1 0 alkyl force Rubamoiru group, the above d
- C ioalkylcarbamoyl group means a C 1 C] L 0 alkyl group having an alkoxycarbonylamino group having 1 to 6 carbon atoms in addition to an N-alkylcarbamoyl group.
- Means a substituted group specifically, for example, N-methoxycarbonylaminomethylcarbamoyl group, N-methoxycarbonylaminoethylcarbamoyl group, N-methoxycarbonylaminopropyl carbamoyl group, N-methoxycarbonylaminobutylcarbamoyl Group, N-ethoxycarbonylaminopentylcarbamoyl group, N-butoxycarbonylaminohexylcarbamoyl group, N-tert-butoxycarbonylaminoethylcarbamoyl group, etc., among which N-methoxycarbonylaminomethylcarbamoyl Group, N-methoxycarbonylaminoethylcal Moil group, N- methoxy Cal Poni Rua amino propyl force Rubamoiru group, N- methoxycarbonylamino butylcarbamoyl group, N- tert-butoxy-
- the C 6 alkoxy force Ruponiru group, 1 -C on Ci one C 6 alkoxy Cal Poni Le group consisting of six alkoxy It means a N-substituted cycarponylamino group, specifically, for example, N-methoxycarbonylaminomethoxycarbonyl group, N-methoxycarbonylaminoethoxycarbonyl group, N-methoxycarbonylaminopropoxycarbonyl group N-methoxycarbonylaminobutoxycarbonyl group, N-ethoxycarbonylaminopentyloxycarbonyl group, N-butoxycarbonylaminohexyloxycarbonyl group, N-tert-butoxycarbonylaminoethoxycarbonyl group, etc.
- N-methoxycarbonylaminomethoxycarbonyl group N-methoxycarbonylaminoethoxycarbonyl group, N-methoxycarbonylaminopropoxycarbonyl group, N-methoxycarbonylaminobutoxycarbonyl group, N-tert-butoxy Cal Poni Le aminoethoxy Cal Poni Le group and the like.
- the d-Cealkylthio group means a group in which a sulfur atom is substituted with an alkyl group having 1 to 6 carbon atoms, and specifically, for example, a methylthio group, an ethylthio group, a propylthio group, an isopropylthio group, a butylthio group Group, isobutylthio group, sec monobutylthio group, tert-butylthio group, pentylthio group, neopentylthio group, hexylthio group, isohexylthio group, etc., among which methylthio group, ethylthio group, propylthio group, isopropylthio group, A butylthio group, a tert-butylthio group and the like are preferred.
- the N-d-Ce alkylsulfamoyl group means a group in which a sulfamoyl group is substituted by an alkyl group having 1 or 6 carbon atoms, specifically, for example, an N-methylsulfamoyl group, an N-ethyls group.
- N, N-Gee d—C 6 alkylsulfamoyl group means It means a group in which an alkyl group having 1 to 6 carbon atoms is disubstituted.
- the Ci one C 6 alkyl sulfide El group, 1 -C sulfinyl alkyl group composed of six is meant substituted group, specifically, for example Mechirusurufi two group, E chill sulfide El, propyl sulfinyl Group, isopropylsulfinyl group, butylsulfinyl group, isobutylsulfinyl group, sec-butylsulfinyl group, tert-butylsulfinyl group, pentylsulfinyl group, neopentylsulfinyl group, hexylsulfinyl group, isohexylsulfinyl group, etc.
- a methylsulfinyl group, an ethylsulfinyl group, a propylsulfinyl group, an isopropylsulfinyl group, a butylsulfinyl group, a tert-butylsulfinyl group and the like are preferable.
- d—C 6 alkylsulfonyl group means a group in which a sulfonyl group is substituted by an alkyl group having 1 to 6 carbon atoms, specifically, for example, methylsulfonyl group, ethylsulfonyl group, propylsulfonyl group, isopropyl A sulfonyl group, a butylsulfonyl group, an isobutylsulfonyl group, a sec-butylsulfonyl group, a tert-butylsulfonyl group, a pentylsulfonyl group, a neopentylsulfonyl group, a hexylsulfonyl group, and an isohexylsulfonyl group.
- the C i -Ce alkoxy group means a group in which an oxygen atom is substituted by an alkyl group having 1 to 6 carbon atoms, and specifically, for example, a methoxy group, an ethoxy group, a propoxy group, an isopropoxy group, a butoxy group Group, isobutoxy group, sec-butoxy group, tert-butoxy group, pentyloxy group, neopentyloxy group, hexoxy group, isohexyloxy group, etc., among which methoxy group, ethoxy group, propoxy group, isopropoxy group Group, butoxy group, isobutoxy group, tert-butoxy group and the like are preferable.
- the d—C 6 alkoxycarbonyl group means a group in which a carbonyl group is substituted with an alkoxy group having 1 to 5 carbon atoms, specifically, for example, a methoxycarbonyl group, an ethoxycarbonyl group, a propoxycarbonyl group.
- a methoxycarbonyl group, an ethoxycarbonyl group, a propoxycarbonyl group, an isopropoxycarbonyl group, a butoxycarbonyl group, a tert-butoxycarbonyl group and the like are preferable.
- the amino d-C 6 alkoxycarbonyl group means a group in which a carbonyl group is substituted with an aminoalkoxy group having 1 to 6 carbon atoms, specifically, for example, an aminomethoxycarbonyl group, an aminoethoxycarbonyl group, Aminopropoxy carbonyl, 2-amino-2-methylpropoxy carbonyl, 2-aminomethyl ethoxycarbonyl, aminobutoxy carbonyl, 2-amino propoxy Luponyl, aminopentyloxy carbonyl, amino Xyloxycarbonyl group and the like, among which aminomethoxycarbonyl group, aminoethoxycarbonyl group, aminopropoxycarbonyl group, 2-aminomethylethoxycarbonyl group, 2-amino-2-methylpropoxycarbonyl group, etc. Is preferred.
- 6- cycloalkylamino group refers to a group in which a cyclic alkyl group having 3 to 6 carbon atoms is N-substituted on an amino group, and specifically, for example, an N-cyclopropylpyramino group , N-cyclobutylamino group, N-cyclopentylamino group, N-cyclohexylamino group and the like, among which N-cyclopropylamino group , N- cyclopentyl Rua amino group, N- cycloheteroalkyl Kishiruamino group and the Shi-liked ⁇ N, N- di C 3 - and Ji 6 cycloalkylamino group, an annular in Amino group consisting six number 3 of stone carbon It means a group in which an alkyl group is disubstituted, and specifically, for example, N, N-dicyclopropylamino group, N, N-dicyclobutyla
- Cycloalkyl O alkoxy group, C 3 -C oxygen atom 6 becomes ring-shaped alkyl group means a group obtained by substituting, for example, N- cyclopropoxy group, N- sheet Kurobutokishi group, Examples thereof include an N-cyclopentyloxy group and an N-cyclohexyloxy group, among which an N-cyclopropoxy group, an N-cyclopentyloxy group, an N-cyclohexyloxy group and the like are preferable.
- the N, N-di-C 3 -C 6 cycloalkyl forcerubamoyl group means a group in which a forcerubamoyl group is disubstituted with a cyclic alkyl group having 3 to 6 carbon atoms.
- N-dicyclopropyl rubamoyl group N, N-dicyclobutyl carbamoyl group, N, N-dicyclopentylcarbamoyl group, N, N-dicyclohexylcarbamoyl group, N-cyclobutyl-N-cyclopropyl rubamoyl group, N-cyclopentyl -N-cyclopropyl rubamoyl group, N-cyclohexyl N-cyclopropyl rubamoyl group, etc., among which N, N-dicyclopropyl rubamoyl group, N, N-dicyclobutylcarbamoyl group, N, N —Dicyclopentylcarbamoyl group and the like are preferable.
- the 5- or 6-membered heterocyclic group includes, for example, an isoxazolyl group, an isothiazolyl group, an imidazolyl group, an oxazolyl group, an oxaziazolyl group, a thiazolyl group, a thiaziazolyl group, a chenyl group, a triazinyl group, a triazolyl group, a pyridyl group, a pyrazyl group, Pyrazinyl group, pyrimidinyl group, pyridazinyl group, pyrazolyl group, pyrrolyl group, virazyl group, furyl group, furazanyl group, imidazolidinyl group, imidazolinyl group, tetrahydrofuranyl group, pyrazolidinyl group, pyrazolinyl group, piperidinyl group, piperidinyl group Group, pyrrolidinyl group,
- isooxazolyl group isothiazolyl group, imidazolyl group, oxazolyl group, thiazolyl group, chenyl group, pyridyl group, pyrazyl group Piraji group, pyrimidinyl group, pyridazinyl group, pyrazolyl group, pyrrolyl group, pyranyl group, furyl group, tetrahydrofuranyl group, morpholino group and the like are preferable.
- a mono- to tri-cyclic heteroaromatic group having 1 to 5 heteroatoms per ring system selected from the group consisting of nitrogen, oxygen and sulfur is, for example, an acridinyl group, an isoquinolyl group, an isoindolyl group , Indazolyl group, indolyl group, indolizinyl group, ethylenedioxyphenyl group, carbazolyl group, quinazolinyl group, quinoxalinyl group, quinolizinyl group, quinolyl group, coumalonyl group, chromenyl group, fenanslidinyl group, fenansloryl Nyl group, dibenzofuranyl group, dibenzothiophenyl group, cinnolinyl group, thonaphthenyl group, naphthyridinyl group, phenazinyl group, phenaxazinyl group, phenothiazinyl group, phthalazin
- the alkyl group is, for example, a methyl group, an ethyl group, a propyl group, an isopropyl group. Butyl group, isobutyl group, sec-butyl group, tert-butyl group, pentyl group, neopentyl group, hexyl group, isohexyl group, heptyl group, octyl group, nonyl group, etc., among which methyl group, ethyl group Group, propyl group, isopropyl group, isobutyl group, sec-butyl group, tert-butyl group and the like.
- the alkenyl group includes, for example, vinyl group, aryl group, 1-propenyl group, isopropenyl group, 2-butenyl group, isobutenyl group, 2-pentenyl group, 2-hexenyl group, 2-heptenyl group And 2-octenyl group, among which vinyl group, aryl group, 1-propenyl group and the like are preferable.
- the alkynyl group includes, for example, ethynyl group, 1-propenyl group, 1-butenyl group, 11-pentenyl group, 1-hexynyl group, 11-heptynyl group, 1-octynyl group and the like. And a 1-propynyl group.
- the N-Ci-Cjo alkyl rubamoyl group means a carbamoyl group substituted with an alkyl group having 1 to 10 carbon atoms, specifically, for example, an N-methylcarbamoyl group, —Ethylcarbamoyl group, N _propylcarbamoyl group, N-isopropylcalilevamoyl group, N-butylcarbamoyl group, N_isobutylcarbamoyl group, N-sec-butylcarbamoyl group, N-tert-butylcarbamoyl group, N-pentylcarbamoyl group, N-neopentylcarbamoyl group, N-hexylcarbamoyl group, N-isohexylcarbamoyl group, N-butylcarbamoyl group, N-decylcarbamoyl group, etc., among which
- N—C “CJ 0 alkylthioalkavamoyl group means a group in which a thiocarbamoyl group is N-substituted with an alkyl group having 1 to 10 carbon atoms, and specifically, for example, an N-methylthioalkavamoyl group N-ethylthiolrubamoyl group, N-propylthiolrubamoyl group, N-isopropylthiolrubamoyl group, N-butyltylylrubamoyl group, NTsobutylthiolrubamoyl group, N-sec-butylbutylcarbamoyl group, N-tert —Butylthiocarpamoyl group, N—Pentylchi Carbamoyl group, N-neopentylthiolrubamoyl group, N-hexylthiolrubamoyl group, N-isohe
- the monocyclic to tricyclic C 7 -C 15 carbon aromatic ring group excludes the above aryl group, and includes, for example, an acenaphthylenyl group, an adamantyl group, an anthryl group, an indenyl group, a norpolnyl group, a phenanthryl group and the like. Among them, an anthryl group and a phenanthryl group are preferred.
- R 1 and R 2 are the same or different and each represent, for example, a hydrogen atom or a -alkyl group, and among them, a hydrogen atom is preferable.
- R 3 represents, for example, a hydrogen atom or a ⁇ - ⁇ alkyl group (excluding n-butyl group), and among them, a C 2 -C 6 alkyl group (excluding n-butyl group) is preferable. Particularly, for example, a C 3 -C 5 branched alkyl group is suitable.
- R 4 represents, for example, a hydrogen atom, a hydroxyl group or a 1 C 3 alkyl group, among which a hydrogen atom and a —C 3 alkyl group are preferable, and a hydrogen atom is particularly preferable.
- R 5 is, for example, a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, a carbamoyloxy group, a propyloxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, Formyl group, formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkyl Kilcarponyl group, N-aralkyl rubamoyl group, aryl group, N-aryl amino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfony
- Alkylsulfamoyl group, arylsulfamoyl. i one C 6 alkoxycarbonyl group, N- Arirukaru Bamoiru group, Aroiru group, Arokishi group, N- Aroiru group, N- (N-Aroirua Mino) C ⁇ Co alkyl force Rubamoiru group, N- Aroiruamino (: ⁇ one C 6 Alkoxycarbonyl group, C 2 —C 6 alkanoyl group, C 2 —C 6 alkanoyloxy group, N—C 2 _C 6 alkanoylamino group, N, N—di—C 2 _C 6 alkanoylamino group, N—Ci—C 6 alkylamino group, N, N—diCi—C 6 alkylamino group, N——C 1Q alkyl rubamoyl group, N, N—di — ( ⁇ - ⁇ alkircalcarbamoyl group, N,
- C i alkyl L-bamoyl group N — ⁇ -C 6 alkoxycarbonyl C 1 () alkyl rubamoyl group, N—1 C 6 alkoxy ruponylamino Ci—C i Q alkyl rubamoyl group, N _C i—C 6 alkoxy rupponylamino d—C 6 alkoxy carbonyl, Ci— Ce alkylthio group, N eleven C 6 alkylsulfamoyl group, N, N-di-C - Ce alkylsulfamoyl Moil group, C - C 6 alkylsulfinyl group, CI- C 6 alkylsulfonyl group, Ct-C 6 alkoxy group, an alkoxycarbonyl Cal Poni group, an amino one (6 ⁇ Le Koki deer Lupo sulfonyl group, N-C 3 - C 6 cycloalkyl amino group, N, N-
- a linear or branched, saturated or unsaturated C A C 9 aliphatic group which may be substituted with a mono- to tricyclic heteroaromatic ring group having 5 C 6 alkoxy group, C—C 6 alkylthio group, A carbon aromatic ring group which may have a substituent selected from the group consisting of N-Ci-Ce alkylamino group, NC x -C 6 alkyl rubamoyl group, and N-Ci-C 6 alkylthio rubamoyl group , 1 to 3 cyclic C 7 — C] L 5 carbon aromatic ring groups or 5 or 6 membered heterocyclic groups or a heteroatom selected from the group consisting of nitrogen, oxygen and sulfur atoms per ring system
- a 5- to 6-membered heteroaromatic group (excluding a 5- or 6-membered heterocyclic group) having 5 to 5 carbon atoms, and among them, a carbon-aromatic ring group is a phenyl group or a naphth
- the C 7 -C 15 carbon aromatic ring group is an acenaphthylenyl group, an adamantyl group, an anthryl group, an indenyl group, a phenanthryl group, and the 5- or 6-membered heterocyclic group is an isoxazolyl group, an isothiazolyl group, an imidazolyl group, an oxazozolyl group.
- a naphthyl group, and a substituent of the optionally substituted carbon aromatic ring group may be a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carboxy group, a cyano group, a nitro group, Rogen atom, hydroxy group, aralkyl group, N-aralkylamino group, aralkyloxy group, aralkylcarponyl group, N-aralkyl rubamoyl group, aryl group, aryloxy group, arylsulfonyl group, arylsulfonyl Okishi group, ⁇ reel sulfamoyl group, Aroiru group, Arokishi group, N- Aroiru group, C 2 one C 6 Arukanoiru groups, C 2 - C 6 alkanoyloxy noisy Ruo alkoxy group, N- C 2 - C 6 alkanol Iruamino group An N-Cj-Ce
- Alkyl carbamoyl group N—C 2 —C 6 alkenylcarbamoyl group, N, N—di C 2 —C 6 alkenylcarbamoyl group, N—amino C 1 CJ 0 Alkyl rubamoyl group, N—C — Ce alkoxy ( : C 10 alkyl alkyl group, NC x -C 6 alkoxycarbonyl C!
- alkylcarbamoyl group C — C 6 alkylsulfinyl group, C — C 6 alkoxy group, C 1 -C 6 alkoxycarbonyl Group, amino Ci—C 6 alkoxycarbonyl group, N—C 3 — ( 6 cycloalkylamino group, (: 3 _ ( 6 cycloalkyloxy group, isoxoazolyl group, isothiazolyl group, oxazolyl group, thiazolyl group, Nyl, pyridyl, pyrazinyl, pyrimigel, pyridazinyl, furyl, tetrahydrofuranyl, morpholino, isoquinolyl, iso Ndoriru group, ethylenedioxy O carboxymethyl-phenylalanine group, quinazolinyl group, quinoxalinyl group, quinolyl group, a dibenzofuranyl El group, dibenzothiophen
- Ar is, for example, a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, a carbamoyloxy group, a propyloxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, Formyl group, formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkyl Kill carbonyl group, N-aralkyl rubamoyl group, aryl group, N-aryl amino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfonyloxy
- Alkyl Rubamoyl group N, N—Gee Ci—. Alkyl force Rubamoiru group, N- C 2 - C 6 alkenyl carbamoylmethyl group, N, N- di C 2 - C 6 alkenyl carbamoylmethyl group, N- amino ( ⁇ one C 1Q alkyl force Rubamoiru group, N one CI- C 6 alkoxy 1.
- Alkyl group Lbamoyl group, N—C i-C 6 alkoxyl force L-ponylamino C, _ C 6 alkoxycarbonyl group, C 6 alkylthio group, N-C! -C 6 alkylsulfamoyl group, N, N-G C-C 6 alkylsulfamoyl group, C 1 -C 6 alkylsulfinyl group, C i — C 6 alkylsulfonyl group,
- the carbon aromatic ring group is preferably a phenyl group or a naphthyl group.
- Alkylcarbamoyl group C i one C 6 alkylsulfinyl group, (1 -0 6 Arukokishi groups, C one C 6 alkoxy Cal Poni group, an amino C i one C 6 alkoxycarbonyl alkylsulfonyl group, N-C 3 - C 6 cycloalkyl Alkylamino group, C 3 — (: 6 cycloalkyloxy group, dioxoxazolyl group, isothiazolyl group, oxazolyl group, thiazolyl group, cetyl group, pyridyl group, pyrazinyl group, pyrimidinyl group, pyridazinyl group, furyl group, tetrahydrofura Nyl, morpholino, isoquinolyl, isoindolyl, ethylenedioxyphenyl, quinazolinyl, quinoxalinyl, quinolyl, dibenzofurany
- oxazolyl group, thiazolyl group, thienyl group, pyridyl group, pyrazinyl group, pyrimidinyl group, pyridazinyl group, and furyl group, and a heteroatom selected from the group consisting of nitrogen atom, oxygen atom and sulfur atom as one ring 1 to 5 mono- to 3-cyclic heteroaromatic groups (excluding 5- or 6-membered heterocyclic groups) per group include isoquinolyl, isoindolyl, ethylenedioxyphenyl, quinazolinyl, quinoxalinyl, and dibenzofuran.
- a zoxazolyl group, a benzothiazolyl group, a benzofuranyl group, a dihydrobenzofuranyl group or a methylenedioxyphenyl group is more preferred.
- n represents an integer of 0 to 2, and 0 or 2 is preferable, and 2 is more preferable.
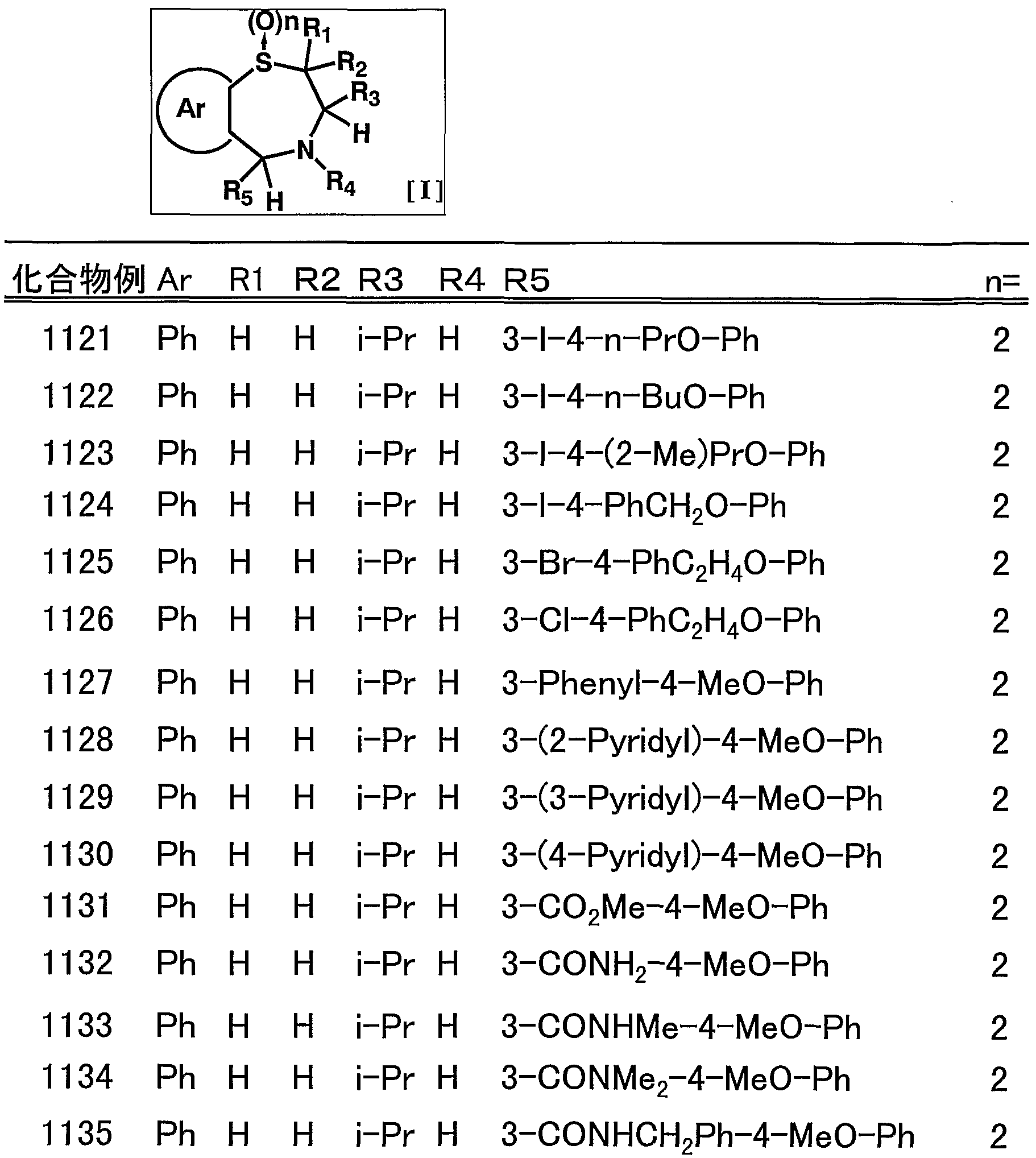
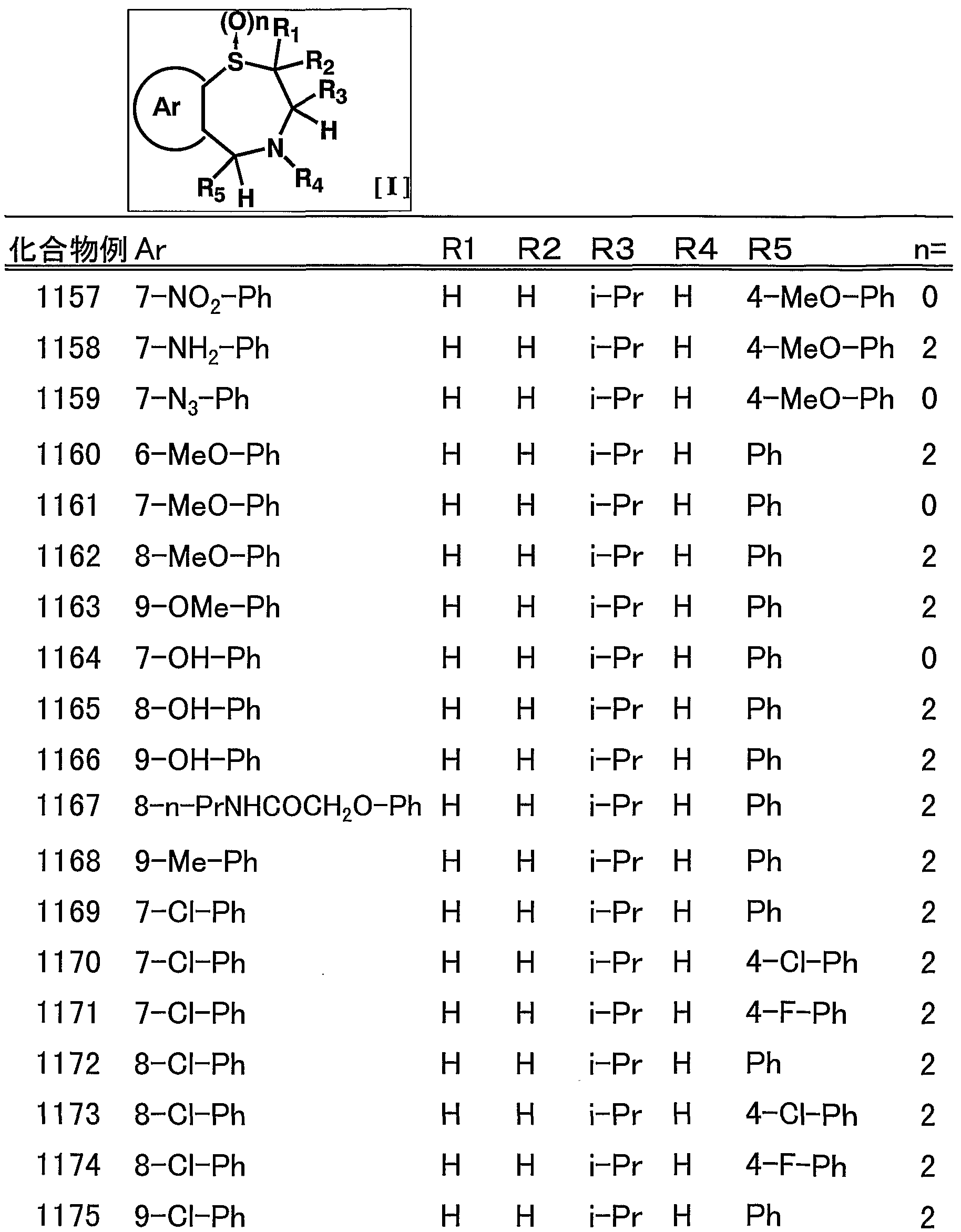
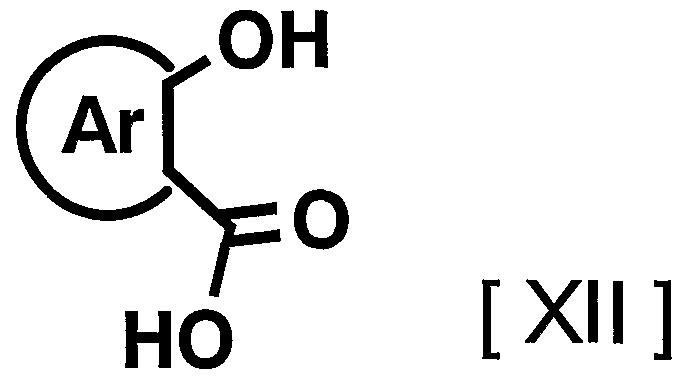
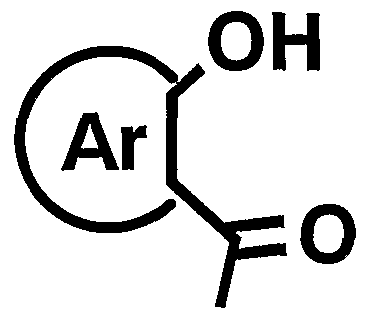
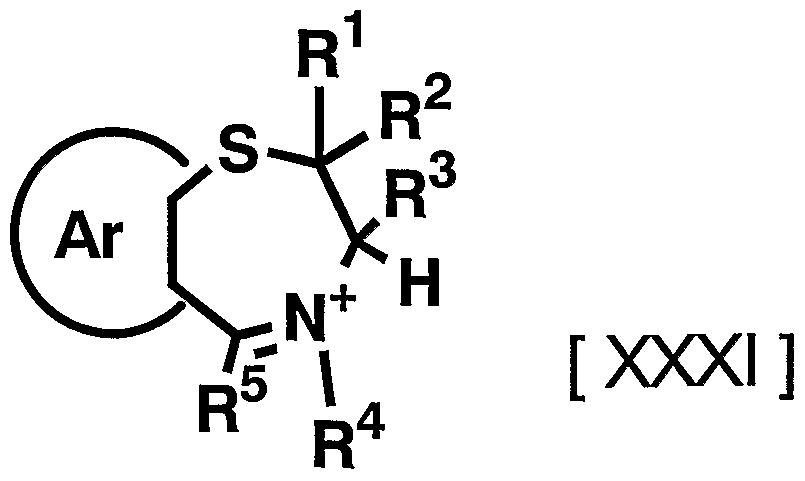
- suitable compounds include, for example, 1009, 1011, 1
- the compound represented by the general formula [I] can be produced by the following production methods AH. Manufacturing method A
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.
- the reaction is carried out by using a base in a dehydrated inert organic solvent in the presence of a suitable additive.
- the compound represented by the general formula [II] can be obtained by reacting the solvent at a temperature of from 0 ° C to the boiling point of the solvent, preferably from 17 to 30 ° C, for from 0.5 to 96 hours, preferably from 12 to 24 hours. Then, a solution of arylnitrile represented by the general formula [III] in an inert solvent is added dropwise at a temperature of 100 ° C. to the boiling point of the solvent, preferably ⁇ 78 to 3O: 0.5.
- the reaction is carried out for up to 96 hours, preferably 12 to 24 hours.
- the inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include hexane, cyclohexane, pentane, ether, 1,4-dioxane , Tetrahydrofuran, tetrahydropyran and the like. Among them, ether, tetrahydrofuran, cyclohexane and the like are preferable.
- Additives used in the reaction include N, N, N ', N' And TMEDA, hexamethylphosphoramine HMPA, lithium chloride and the like. Among them, ⁇ , ⁇ , ⁇ ′, ⁇ ′-tetramethylethylenediamine is preferable.
- Examples of the base used in the reaction include ⁇ -butyllithium, sec-butyllithium, t-butyllithium, phenyllithium, methyllithium and the like. Among them, n-butyllithium and sec-butyllithium are preferable. is there.
- the amount of the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions.
- 1 to 10 equivalents, preferably 1.5 to 2 equivalents, of arylyl nitrile represented by the above general formula [III] is used.
- RR 2 is a hydrogen atom or a C 3 alkyl group
- R 3 is a hydrogen atom or a C 1 C 6 alkyl group (however, excluding n-butyl group)].
- the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions.
- the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [IV].
- the compound represented by the above general formula [V] is reacted at room temperature for 0.5 to 96 hours, preferably 3 to 24 hours.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and tetrachloroethane.
- methyl alcohol, ethyl alcohol, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferable.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 with respect to the compound represented by the above general formula [IV]. 1.2—Use 2 equivalents of aziridin represented by the above general formula [V].
- R 1 R 2 , R 3 , 1 ⁇ 5 and 8 has the above-mentioned meaning] in a suitable solvent such as 2,6-lutidine.
- Azeotropic distillation optionally in the presence of an acid, or heating to reflux in the presence of a desiccant, for example, molecular sieves.
- a desiccant for example, molecular sieves.
- R 1 R 2 , R 3 , R 5 and Ar have the above-mentioned meanings].
- the reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but usually the reaction is carried out in the presence of a desiccant such as molecular sieves, and in an appropriate solvent such as 2,6-lutidine.
- the reaction is completed by refluxing for 5 to 96 hours, preferably 12 to 24 hours, in the presence of a suitable acid such as hydrochloric acid.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and N , N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, n-propanol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran, etc. From the viewpoint of securing a suitable reaction temperature, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are particularly preferable.
- Examples of the acid used in the reaction include inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid, and perchloric acid; for example, Lewis such as trifluoroboric acid, titanium tetrachloride, and zinc chloride.
- inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid, and perchloric acid
- Lewis such as trifluoroboric acid, titanium tetrachloride, and zinc chloride.
- Acids for example, sulfonic acids such as P-toluenesulfonic acid, trifluoromethanesulfonic acid, and methanesulfonic acid; and organic acids such as formic acid, trifluoroacetic acid, and acetic acid, and among them, hydrochloric acid, acetic acid, and P-toluenesulfonic acid
- sulfonic acids such as P-toluenesulfonic acid, trifluoromethanesulfonic acid, and methanesulfonic acid
- organic acids such as formic acid, trifluoroacetic acid, and acetic acid, and among them, hydrochloric acid, acetic acid, and P-toluenesulfonic acid
- Preferred examples of the desiccant used in the reaction include molecular sieves, magnesium sulfate, and sodium sulfate. Among them, molecular sieves are preferred.
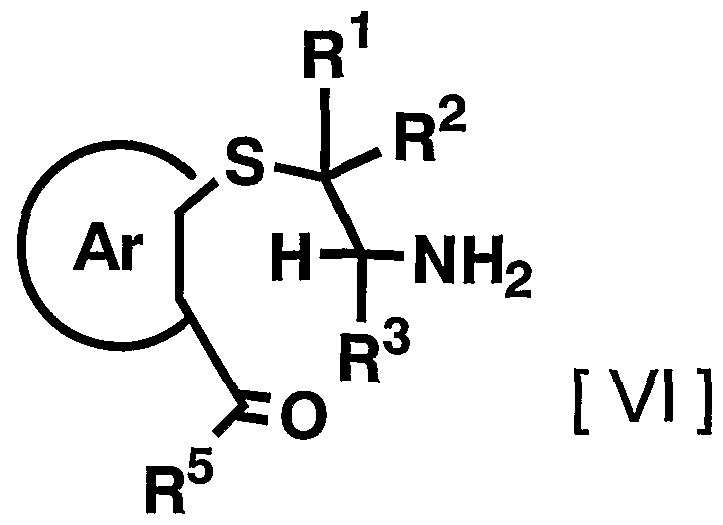
- the amount of the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but usually, 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [VI] is used.
- the acid and the desiccant may be used alone or in appropriate combination of two or more.
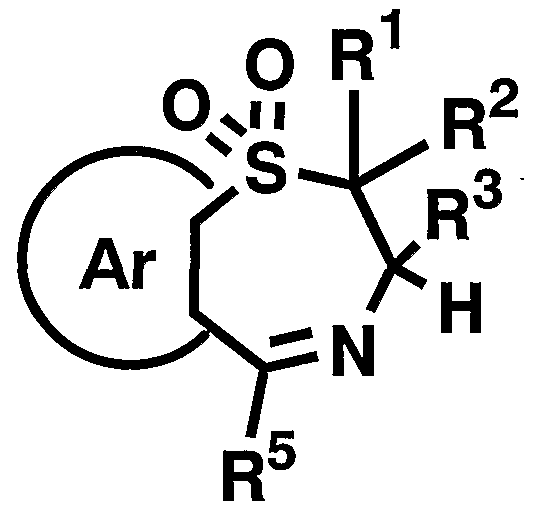
- R 1 R 2 , R 3 , R 5 and Ar have the meaning described above, in an appropriate organic solvent with an oxidizing agent to give a compound of the general formula [VIII ]
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.
- the reaction is carried out in a suitable organic acid solvent such as trifluoroacetic acid, for example, a suitable oxidizing agent such as hydrogen peroxide.
- a suitable organic acid solvent such as trifluoroacetic acid
- a suitable oxidizing agent such as hydrogen peroxide.
- the reaction is completed by stirring for 5 to 96 hours, preferably 12 to 24 hours, in the presence of
- stirring in a suitable inert organic solvent such as, for example, methylene chloride
- a suitable oxidizing agent such as, for example, methacrylic acid mouth perbenzoic acid
- the inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples thereof include methylene chloride, methylene chloride, 1,2-dichloroethane, and trichloroethane.
- Preferred are methylene, chloroform, 1,2-dichloroethane, trichloroethane, toluene, xylene and the like.
- Examples of the acid solvent used in the reaction include organic acids such as formic acid, trifluoroacetic acid and acetic acid, among which trifluoroacetic acid is preferred.
- the oxidizing agent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- hydrogen peroxide, peracetic acid, trifluoroperacetic acid, methachloroperbenzoic acid, oxone examples thereof include chromic acid and permanganese sun.
- hydrogen peroxide, metachloroperbenzoic acid, and the like are preferable.
- the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [VII]. 1.2 Use 2 to 2 equivalents of oxidizing agent.
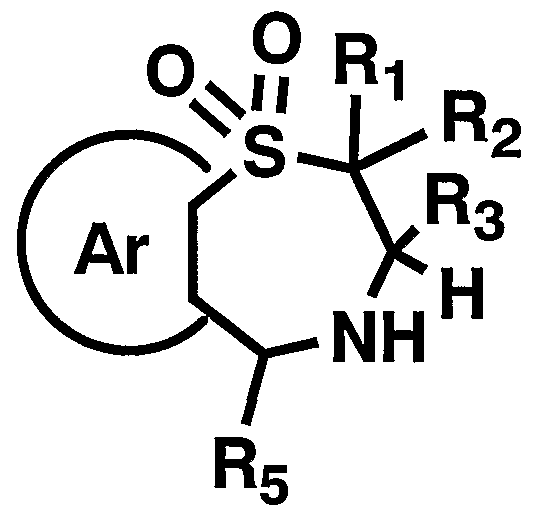
- R 1 , R 2 , R 3 , 5 and 8 have the above-mentioned meanings] in a suitable organic solvent such as tetrahydrofuran (THF).
- a suitable organic solvent such as tetrahydrofuran (THF).
- Reduction using a reducing agent or catalytic hydrogenation using a catalyst such as 10% palladium on carbon, or reaction with zinc chloride in the presence of an acid catalyst such as hydrochloric acid The general formula [I]
- the reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.
- the reaction can be carried out in a suitable organic acid solvent such as THF, for example, using a suitable reducing agent such as porane.
- the reaction is completed by stirring for up to 96 hours, preferably 12 to 24 hours.
- a suitable organic solvent such as methyl alcohol in the presence of a suitable palladium catalyst such as 10% palladium on carbon for 5 to 96 hours, preferably 12 to 24 hours
- the reaction is complete.
- the reaction is completed by appropriately stirring with a metal or a metal salt for 5 to 96 hours, preferably 12 to 24 hours in an acid or alkali solution.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and tetrachloroethane.
- Carbon chloride acetic acid, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, toluene, xylene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran, etc., among which methylene chloride, Preferred are chloroform, 1,2-dichloroethane, trichloroethane, toluene, xylene and the like.
- Examples of the reducing agent used in the reaction include lithium borohydride, sodium borohydride, sodium cyanotrihydroborate, porane methyl sulfide complex, porane THF complex, and lithium aluminum hydride. Among them, a porane methylsulfide complex and a porane THF complex are preferable.
- the amount of the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [VIII]. Use 2 to 2 equivalents of reducing agent.
- Catalysts used in the catalytic hydrogenation reaction include, for example, palladium activated carbon, Raney nickel, platinum oxide, etc., and among them, 10% palladium activated carbon is preferred.
- Metal used in the reaction is, for example, zinc, aluminum, iridium. , Chromium, titanium, tin, cerium, iron, copper, vanadium, magnesium, etc., of which zinc and magnesium are preferred.
- the product represented by the above general formula [I] can be obtained by purifying the product by a conventional method.
- the compound represented by the general formula [I] or a salt thereof can be isolated and purified from the reaction solution by a known separation means such as solvent extraction, recrystallization, and chromatography.
- R represents an alkyl group such as a methyl group
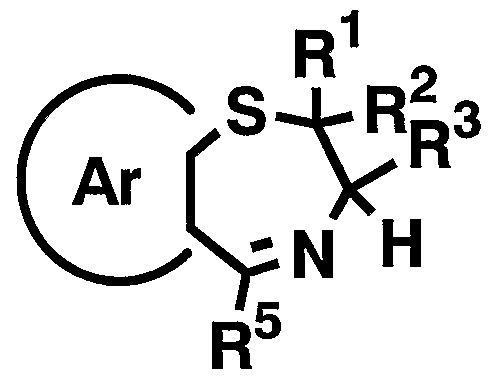
- R 5 and Ar have the above-mentioned meanings] by heating the compound represented by the formula [XI]
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but usually the reaction is carried out in a suitable organic acid solvent such as N, N-dimethylformamide, for example, sodium hydride.
- a suitable organic acid solvent such as N, N-dimethylformamide, for example, sodium hydride.
- the reaction is completed by reacting the mixture with a suitable base at 0 ° C. to the boiling point of the solvent, preferably 0 to 150, 0.5 to 96 hours, preferably 12 to 24 hours. Then, for example, in an inert organic solvent or neat, from 0 ° C. to the boiling point of the solvent, preferably 100 to 300 ° C., 0.1 to 96 hours, preferably 0.5 to 2 hours.
- the reaction is completed by stirring for an hour.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, but specific examples thereof include, for example, Shiridani methylene, chloroform, 1,2-dichloroethane, trichloroethane, Carbon tetrachloride, N, N-dimethylformamide, acetonitrile, acetone, benzene, toluene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran, etc., among which N, N-dimethylformamide, toluene, xylene, acetone , Ether, tetrahydrofuran and the like are preferred.
- Examples of the base used in the reaction include trimethylamine, triethylamine, N, N-diisopropylethylamine, N-methylmorpholine, N-methylpiperidine, N-methylpiperidine, N, N-dimethylaniline, Tertiary aliphatics such as 1, 8-diazabicyclo [5.4.0] pendant force _7-en (DBU), 1,5-azabicyclo [4.3.0] nona5 -en (DBN)
- Amines for example, alkali metal hydrides such as sodium hydride and potassium hydride, for example, alkali metal alkoxides such as potassium tert-butylate, sodium ethylate, and sodium methylate; for example, potassium hydroxide, sodium hydroxide, and the like Alkali metal hydroxides; for example, alkali metal carbonates such as carbonated lime; and the like, among which triethylamine, N, N-diisopropylethylamine, Sodium
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 relative to the compound represented by the above general formula [IX].
- inert solvent used in the rearrangement reaction for example, toluene, xylene, diphenyl ether, dipara-tolyl ether and the like can be mentioned, and among them, diphenyl ether is preferable.
- the compound represented by the general formula [XI] is hydrolyzed with a base such as sodium hydroxide in a suitable organic solvent, or with a reducing agent such as lithium aluminum hydride in a non-polar solvent such as THF.
- a base such as sodium hydroxide in a suitable organic solvent
- a reducing agent such as lithium aluminum hydride in a non-polar solvent such as THF.
- the compound represented by the above general formula [IV] can be prepared by reduction.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, benzene examples thereof include silene, toluene, xylene, ether, 1,4-dioxane, tetrahydrofuran, and tetrahydropyran.
- methyl alcohol, ethyl alcohol, ether, 1,4-dioxane, and tetrahydrofuran are preferable.
- Examples of the base used in the reaction include alkali metal alkoxides such as potassium tert-butylate, sodium ethylate and sodium methylate; for example, alkali metal hydroxides such as lithium hydroxide and sodium hydroxide.
- alkali metal carbonates such as potassium carbonate and the like, among which potassium tert-butylate, sodium ethylate, sodium methylate, potassium hydroxide and sodium hydroxide are preferred.
- the inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples thereof include hexane, cyclohexane, pentane, ether, 1,4-dioxane And tetrahydrofuran, tetrahydropyran and the like, among which ether, tetrahydrofuran, cyclohexane and the like are preferable.
- Lithium aluminum hydride is suitable as the reducing agent used in the reaction.
- the compound represented by the above general formula [IV] thus obtained is prepared in the same manner as in Step 2-5 of Production Method A.
- the compound represented by the above general formula [I] can be obtained by reacting
- Ar has the above-mentioned meaning
- salicylic acid an aryl compound represented by R 5 —H (where R 5 has the above-mentioned meaning); It can be synthesized by a Friedel-Crafts reaction using lumimium.
- Ar has the above-mentioned meaning] by converting an aryl halide compound and metallic magnesium into a suitable ether-based dehydrating solvent such as, for example, ethyl ether, tetrahydrofuran, etc., at a low temperature.
- a suitable ether-based dehydrating solvent such as, for example, ethyl ether, tetrahydrofuran, etc.
- the Grignard reagent prepared by reacting at the boiling point of the solvent is reacted with the above-mentioned dehydrated inert organic solvent at a low to room temperature in the above-mentioned dehydrated inert organic solvent, and the resulting product is subsequently dissolved in a suitable solvent such as dichloromethane.
- a suitable solvent such as dichloromethane.
- it can be produced by reacting with an oxidizing agent such as manganese dioxide.
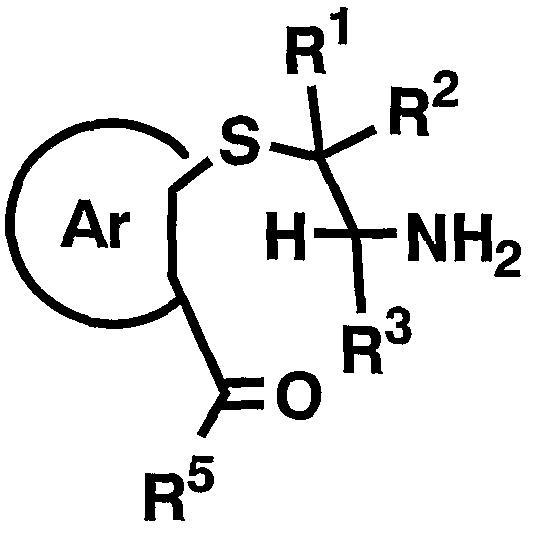
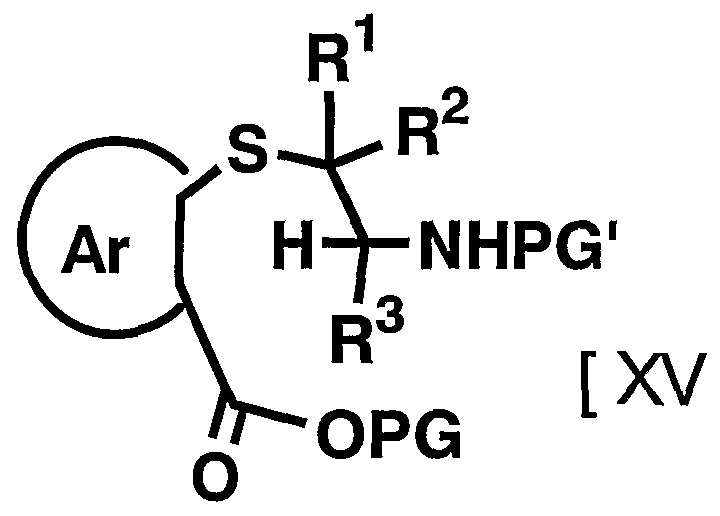
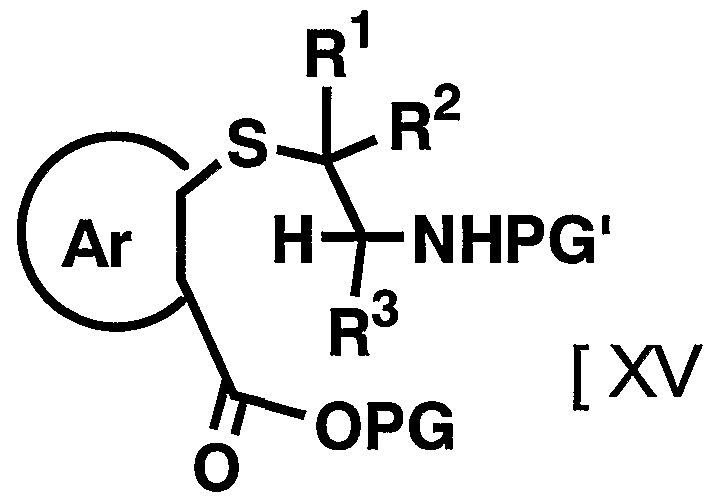
- R 2 and 1 3 are as defined above] is reacted with a compound represented by then formula by the resulting amino group apply a protecting group suitable Amino group [XV]
- PG 1 represents a protecting group for an amino group
- R 1 R 2 , R Ar and PG have the above-mentioned meanings.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.
- the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [XIV] and the above general formula
- the compound [V] is reacted at room temperature for 0.5 to 96 hours, preferably 3 to 24 hours.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, and specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and tetrachloroethane.
- methyl alcohol, ethyl alcohol, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferable.
- the amount of the reagent used in the reaction can be appropriately increased or decreased depending on the starting material and the reaction conditions. Usually, 1.2 to 10 equivalents, preferably 1 Use 2 to 2 equivalents of the aziridine represented by the above general formula [V].
- Examples of the protective group for the carbonyl group include lower alkyl groups such as methyl group, ethyl group and tert-butyl group, and aralkyl groups such as benzyl group and p-methoxybenzyl group. , An ethyl group, a tert-butyl group, a benzyl group and the like.
- Examples of the protecting group for the amino group include benzyl, p-nitrobenzyl and the like.
- An aralkyl group for example, an acyl group such as a formyl group or an acetyl group; a lower alkoxy group such as an ethoxycarbonyl group, an aryloxycarbonyl group, or a tert-butoxycarbonyl group; for example, a benzyloxycarbonyl group, p-nitro Aralkyloxycarbonyl groups such as benzyloxycarbonyl group, triphenylmethyl group, paramethoxyphenyldiphenylmethyl group, diphenylphosphonyl group, and benzenesulfenyl group; among them, aryloxycarbonyl group Tert-butoxycarbonyl, benzyloxycarbonyl, triphenylmethyl and the like are preferred.
- the removal of the protecting group of the hydroxyl group depends on the type and stability of the compound, but the method described in the literature [Protective Groups 'In' Organic Synthesis (Organic Synthesis), TW Greene, John John & Sons (1981)] or a method analogous thereto, for example, solvolysis using an acid or base, chemical reduction using a metal hydride complex, or the like. It can be carried out by corrosion reduction using a palladium carbon catalyst, Raney nickel catalyst or the like.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but usually, the reaction is carried out by reacting the deprotected carboxylic acid with N, 1-dimethylhydroxylamine in a dehydrated inert organic solvent. , In the presence of a base, a condensation aid and Z or a condensing agent as appropriate — from 100 ° C. to the boiling point of the solvent, preferably from 0 to 30 ° C., from 0.5 to 96 hours, preferably from 3 to 24 hours. Let it.
- the inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, and specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, and trichloroethane. , N, N-dimethylformamide, ethyl acetate, methyl acetate, acetonitrile, acetic anhydride, methyl alcohol, ethyl alcohol, benzene, xylene, water, acetic acid, toluene, 1,4-dioxane, tetrahydrofuran and the like.
- Bases used in the reaction include, for example, trimethylamine, triethyl ⁇ / ami, N, N-diisopropylethylamine, N-methylmorpholine, N-methylvinyl lysine, N-methylpiperidine, N, N-dimethyl Anilin, 1,8-diazabicyclo [5.4.0] pendant 7-ene (DBU), 1,5-azabicyclo [4.3.0] nona-5-ene (DBN), etc.
- Tertiary aliphatic amines for example, aromatic amines such as pyridine, 4-dimethylaminopyridine, picoline, lutidine, quinoline, and isoquinoline; metal salts such as potassium metal, sodium metal, and lithium metal; Metal hydrides such as sodium hydride and hydrogen hydride; alkyl metal hydrides such as butyl lithium; potassium lithium-tert-butylate, sodium ethylate Alkali metal alkoxides such as sodium methylate and the like; alkali metal hydroxides such as hydroxylated sodium hydroxide and sodium hydroxide; alkali metal alkoxides such as carbonated lithium and the like.
- Tertiary aliphatic amines and the like are preferred, and in particular, triethylamine, N, N-diisopropylethylamine and the like are more preferred.
- condensation auxiliary used in the reaction examples include N-hydroxybenzotriazole hydrate, N-hydroxysuccinimide, N-hydroxy-15-norpolene-2,3-dicarboximide, and 3-hydroxy-3 , 4-dihydro-4-oxo-1,2,3-benzotriazol and the like. Among them, N-hydroxybenzotriazole and the like are preferable.
- Examples of the condensing agent used in the reaction include thionyl chloride, N, N-dicyclohexylcarbodiimide, 1-methyl-2-bromopyridinium iodide, and N, N'-capillonyldiimidazole , Diphenylphosphoryl chloride, diphenylphosphoryl azide, N, N, disuccinimidyl carbone, N, N'-disuccinimidyl oxalate, 1-ethyl-3- (3-dimethylaminopropyl) carbodiimide Hydrochloride, ethyl ethyl chloroformate, isoptyl chloroformate, benzotriazo-1-lyloxysheetris (dimethylamino) phosphonium hexafluorophosphate, and the like, among which N, N-dicyclohexylcarposimide, 1-ethyl-3- ( 3-dimethylaminopropyl) carposi
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but is usually 1 to 50 equivalents, preferably 1 to 2 equivalents of N, di-dimethylhydroxyl hydroxylase relative to the carboxylic acid.
- Min 1 to 50 equivalents, preferably 3 to 5 equivalents of base, 1 to 50 equivalents, preferably 1 to 5 equivalents of condensation aid and / or 1 to 50 equivalents, preferably 1 to 5 equivalents
- the base, the condensation aid, and the condensing agent can be used alone or in appropriate combination of two or more.
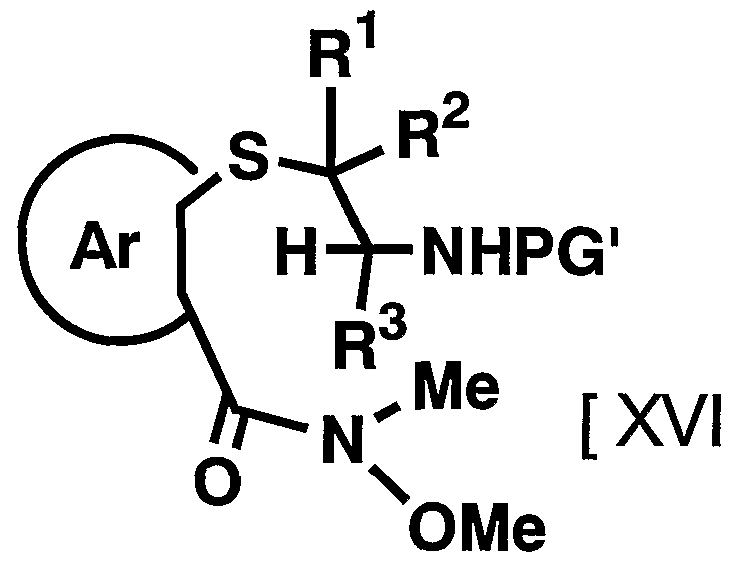
- RR 2 , R 3 , Ar, PG 1 and Me have the above-mentioned meaning
- a halogenated aryl compound and metal magnesium to a suitable ether-based compound such as, for example, methyl ether, tetrahydrofuran, etc.
- a general formula [XVI I] is obtained by reacting in a dehydrated inert organic solvent at a low temperature to room temperature with a Grignard reagent prepared by reacting at a low temperature to the boiling point of the solvent in a dehydrated solvent.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.
- the reaction is carried out in an inert organic solvent that has been dehydrated by reacting the compound represented by the above general formula [XVI] with the Grignard reagent.
- the reaction is carried out at a temperature of 100 to the boiling point of the solvent, preferably -78 to 30 ⁇ , for 0.5 to 96 hours, preferably for 12 to 24 hours.
- the inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and reaction conditions, but is usually 2 to 10 equivalents, preferably 2 to 10 equivalents to the compound represented by the above general formula [XVI]. Use 3 equivalents of Grignard reagent.
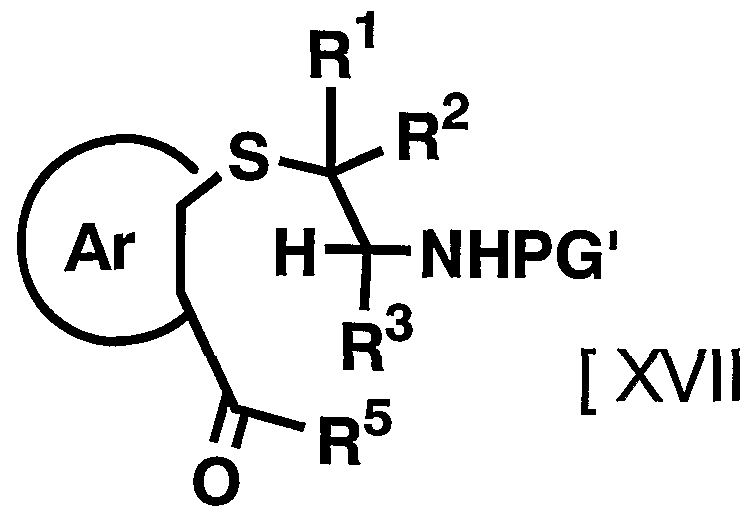
- the removal of the protecting group depends on the type and stability of the compound, but the method described in the literature [Protective Groups in Organic Synthesis, T.W. Green (TW Greene, John Wiley & Sons (1981)) or a method analogous thereto, for example, solvolysis using an acid or a base, chemical reduction using a metal hydride complex, or the like. Alternatively, it can be carried out by catalytic reduction using a palladium carbon catalyst, a Raney nickel catalyst or the like.
- the compound represented by the above general formula [VI] can be synthesized by subsequently reacting the compound represented by the above general formula [VI] by the method of Step 3-5 of Production method A. . Manufacturing method D
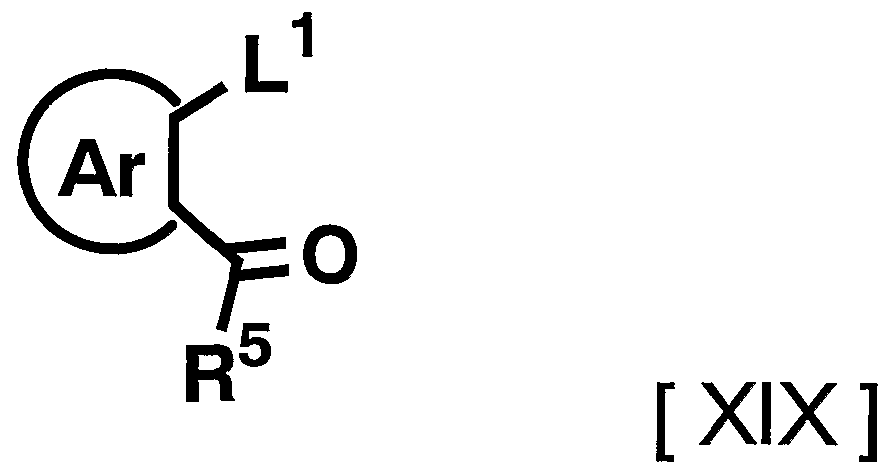
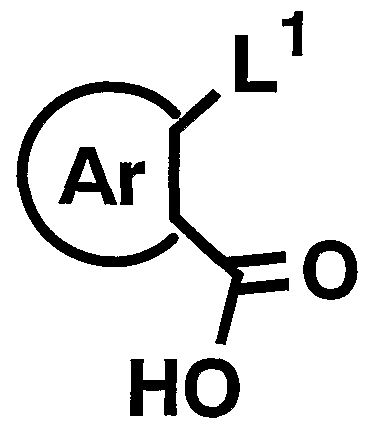
- L 1 represents a leaving group such as halogen, and R 5 and Ar have the above-mentioned meanings] can be synthesized by a known method.
- R 5 is The compound can be synthesized by reacting with an aryl compound represented by the following formula) by, for example, a Friedel-Crafts reaction using aluminum chloride.
- the reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.
- the reaction is carried out in a suitable organic acid solvent such as N, N-dimethylformamide, for example, potassium carbonate.
- the reaction is completed by reacting with a suitable base at o: from the boiling point of the solvent, preferably 50 to 120 ° C, for 0.5 to 96 hours, preferably 2 to 12 hours.
- any organic solvent that does not adversely affect the reaction can be used.
- Examples of the base used in the reaction include trimethylamine, triethylamine, N, N-diisopropylethylamine, N-methylmorpholine, N-methylpiperidine, N-methylpiperidine, N, N-dimethylaniline, Tertiary fats such as 1,8-diazabicyclo [5.4.0] pendant 7-ene (DBU) and 1,5-azabicyclo [4.3.0] noner 5-ene (DBN)
- Aliphatic amines for example, alkali metal hydrides such as sodium hydride and potassium hydride, for example, alkali metal alkoxides such as potassium tert-butylate, sodium ethylate, sodium methylate; potassium hydroxide, 7jc oxidation Alkali metal hydroxides such as sodium; for example, alkali metal carbonates such as carbon dioxide, etc., among which triethylamine, N, N-diisopropylethyl Amin, sodium hydride, potassium
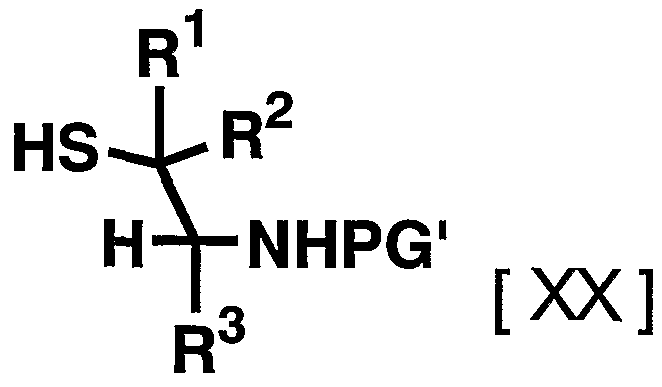
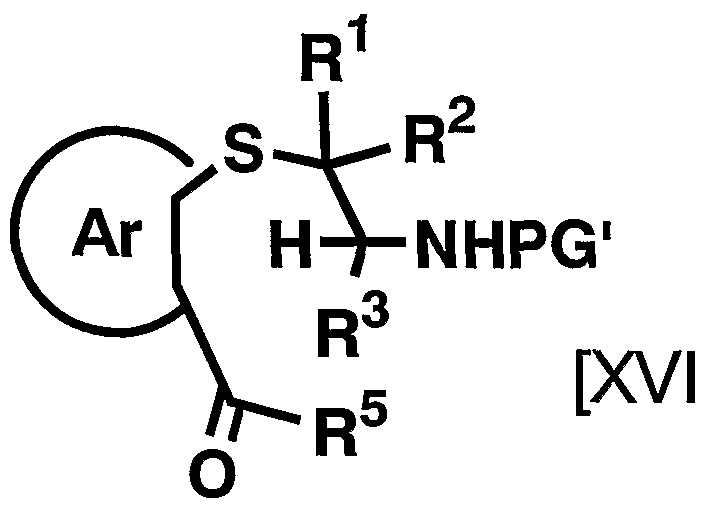
- the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compounds and reaction conditions, but is usually 1.2 to 20 equivalents, preferably 1 to 20 equivalents to the compound represented by the above general formula [XIX]. 1. Use 2 to 5 equivalents of the compound represented by the general formula [XX].
- the compound represented by the above general formula [XV II] is synthesized into the compound represented by the above general formula [I] by the method of the fourth step of the production method C and the third to fifth steps of the production method A. can do.
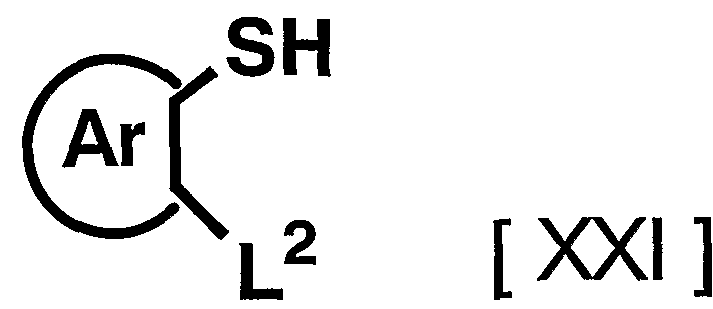
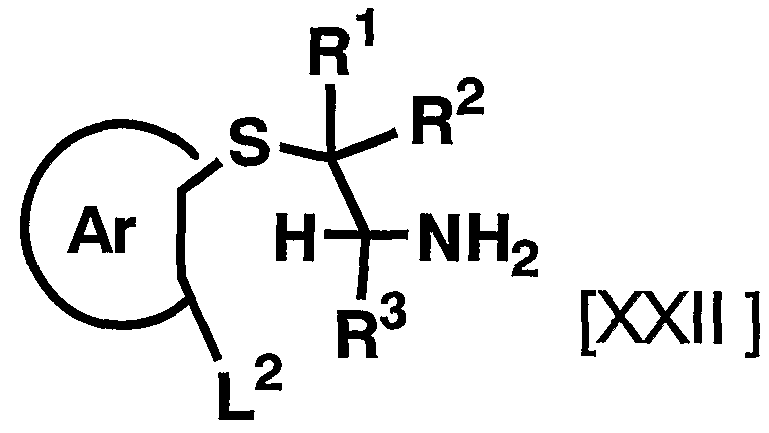
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.
- the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [XXI] and the above general compound.
- the compound represented by the formula [V] is dissolved at room temperature in 0.
- the reaction is performed for 5 to 96 hours, preferably 3 to 24 hours.
- any organic solvent that does not adversely affect the reaction can be used.
- the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions, but usually, the amount of the compound represented by the above general formula [XXI] is 1.
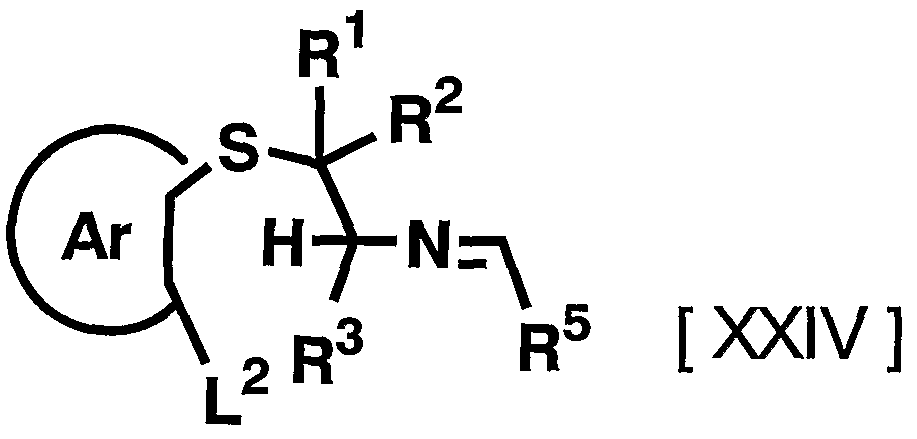
- R 5 has the meaning described above, and azeotropic distillation in a suitable solvent such as 2,6-lutidine in the presence of an appropriate acid, or for example, By heating to reflux in the presence of a desiccant such as Ura sieves, the general formula [XXIV]
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but usually the reaction is carried out in the presence of a desiccant such as molecular sieves, for example, using a suitable agent such as 2,6-lutidine.
- the reaction is completed by refluxing in a solvent, for example for 5 to 96 hours, preferably 12 to 24 hours, in the presence of a suitable acid such as hydrochloric acid.
- any organic solvent that does not adversely affect the reaction can be used.
- specific examples include, but are not limited to, methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, N, N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, and methyl alcohol.
- pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferred.
- Examples of the acid used in the reaction include inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid, and perchloric acid; for example, Lewis acids such as trifluoroboric acid, titanium tetrachloride, and zinc chloride; !) Sulfonic acids such as mono-toluenesulfonic acid, trifluoromethanesulfonic acid, and methanesulfonic acid; organic acids such as formic acid, trifluoroacetic acid, and acetic acid; and hydrochloric acid, acetic acid, and p-toluenesulfonic acid are preferable.
- inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid, and perchloric acid
- Lewis acids such as trifluoroboric acid, titanium tetrachloride, and zinc chloride
- Sulfonic acids such as mono-tolu
- the desiccant used in the reaction includes, for example, molecular sieves, magnesium sulfate, sodium sulfate, etc. Among them, the molecular sieves are preferable. Although it can be increased or decreased as appropriate, it is usually 1.2 to 10 equivalents to the compound represented by the above general formula [XXII]. Preferably 1.2 to 2 equivalents of the aldehyde represented by the above general formula [XXIII], 1.2 to 10 equivalents, preferably 1.2 to 2 equivalents of the desiccant, 1 to 50 equivalents Preferably, 3 to 5 equivalents of acid are used. The acid and the desiccant may be used alone or in combination of two or more.
- R 1 , R 2 , R 3 , R 5 , Ar, and L 2 have the above-mentioned meanings.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.
- the reaction is carried out in a dehydrated inert organic solvent using a suitable base in the presence of an appropriate additive at a temperature of 100 ° C.
- an appropriate additive at a temperature of 100 ° C.
- To the boiling point of the solvent preferably 1 to 78 to 30 ° (: 0.5 to 96 hours, preferably 12 to 24 hours to complete the intramolecular ring closure reaction.
- the inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples thereof include hexane, cyclohexane, pentane, ether, and 1,4-dioxane.
- tetrahydrofuran, tetrahydropyran and the like, and particularly, for example, ether, tetrahydrofuran, cyclohexane and the like are preferable.
- additives used in the reaction include ⁇ , ⁇ , ⁇ ′, ⁇ ′-tetramethylethylenediamine TMEDA, hexamethylphosphoramine HMPA, lithium salt, and the like.
- ', ⁇ '-Tetramethylethylenediamine is preferred.
- Examples of the base used in the reaction include ⁇ -butyllithium, sec-butyllithium, t-butyllithium, phenyllithium, methyllithium and the like. Among them, n-butyllithium and sec-butyllithium are preferred. is there.
- the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions. However, usually, the amount of the reagent represented by the above general formula [XXIV] is 2%. ⁇ 10 equivalents, preferably 2-3 equivalents of base are used.
- the obtained compound represented by the general formula [XXV] can be synthesized by the method of the fourth step of the production method A to produce the compound represented by the general formula [I].
- Manufacturing method F
- R 1 R 2 and R 3 have the above-mentioned meanings in a polar solvent such as lutidine, and the compound represented by the general formula [XXVI I]
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.
- the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [XXVI] and the compound represented by the above general formula.
- the compound represented by [V] is reacted at room temperature for 0.5 to 96 hours, preferably 3 to 24 hours.
- any organic solvent that does not adversely affect the reaction can be used.
- specific examples include, but are not limited to, methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, carbon tetrachloride, N, N-dimethylformamide, ethyl acetate, methyl acetate, and acetate nitrite.
- Ryl methyl alcohol, ethyl alcohol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran, etc., among which methyl alcohol, ethyl alcohol, pyridine, lutidine, N, N —Dimethylformamide, toluene, xylene and the like are preferred.
- the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compounds and reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [XXV II]. 1. Use 2 to 2 equivalents of the aziridine represented by the above general formula [V].
- the reagent used in the oxidation reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.
- the reaction is carried out, for example, in a suitable solvent with a suitable oxidizing agent such as manganese dioxide at room temperature for 5 to 96 hours.
- the reaction is completed for a period of time, preferably 12 to 24 hours.
- the oxidizing agent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, but specific examples thereof include manganese dioxide, chromic acid, t-butyl chromate, lead tetraacetate, and tetroxide. Ruthenium, selenium dioxide, halogen, oodosil compounds, oxygen, organic peroxides, silver peroxosulfate, nitric oxide, dimethyl sulfoxide, o-chloranil, silver oxide, etc., among which manganese dioxide, chromic acid, selenium dioxide And dimethyl sulfoxide.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but is usually preferably from 0.22 to 50 equivalents, preferably from the compound represented by the above general formula [VII]. Use 1.2 to 2 equivalents of oxidizing agent.
- the reagent used in the ring closure reaction can be appropriately increased or decreased depending on the starting compound and reaction conditions, but usually the reaction is carried out in the presence of a desiccant such as molecular sieves, for example, 2,6-lutidine.
- the reaction is completed by refluxing in a suitable solvent, for example in the presence of a suitable acid such as hydrochloric acid for 5 to 96 hours, preferably 12 to 24 hours.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and N , N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, n-propanol, benzene, xylene, pyridine , Lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran and the like.
- pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferable.
- Examples of the acid used in the reaction include inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid and perchloric acid; for example, Lewis acids such as trifluoroboric acid, titanium tetrachloride, zinc chloride and the like. Sulfonic acids such as p-toluenesulfonic acid, trifluoromethanesulfonic acid, and methanesulfonic acid; organic acids such as formic acid, trifluoroacetic acid, and acetic acid; and hydrochloric acid, acetic acid, and p-toluenesulfonic acid.
- inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid and perchloric acid
- Lewis acids such as trifluoroboric acid, titanium tetrachloride, zinc chloride and the like.
- Sulfonic acids such as p-tolu
- Suitable desiccants used in the reaction include, for example, molecular sieves, magnesium sulfate, and sodium sulfate, with molecular sieves being preferred.
- the amount of the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but is usually 1.2 to 10 equivalents to the compound represented by the above general formula [XXVIII].
- 1.2 to 2 equivalents of desiccant, 1 to 50 equivalents, preferably 3 to 5 equivalents of acid are used.
- the acid and the desiccant may be used alone or in appropriate combination of two or more.
- R 1 R 2 , R 3 and Ar have the above-mentioned meanings] and a compound obtained by activating a halogenated aryl compound with a metal such as magnesium, lithium, copper or zinc. Is reacted in a suitable ether-based dehydrating solvent at a low temperature to the boiling point of the solvent to obtain the general formula [XXV]
- This production method is a method for producing a compound represented by the general formula [I] wherein R 4 is 1 C 3 alkyl.
- R 4 represents one C 3 alkyl and L 3 represents a leaving group such as halogen] by reacting an alkyl octride represented by the general formula [XXX I]
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.
- the reaction is carried out by reacting the compound represented by the above general formula [VII] with the alkyl halide represented by the above general formula [XX].
- the reaction is completed by heating to reflux in an inert organic solvent for 5 to 96 hours, preferably 12 to 24 hours.
- the organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- Specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and N , N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, n-propanol, benzene, xylene, toluene, xylene, 1,4-dioxane, tetrahydrofuran, and the like. Of these, acetonitrile, toluene, xylene and the like are preferred.
- the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and reaction conditions, but is usually 1 to 50 equivalents, preferably 10 to 10 equivalents to the compound represented by the above general formula [VII]. Use 50 equivalents of the compound represented by the above general formula [XXX].
- This production method is a method for producing a compound represented by the general formula [I] wherein R 4 is a hydroxyl group.
- the reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.
- the reaction is carried out in a suitable inert organic solvent such as methylene chloride, for example,
- the reaction is completed by stirring at room temperature for 5 to 96 hours, preferably 12 to 24 hours, in the presence of a suitable oxidizing agent.
- the inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, and specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, and trichloroethane. , Carbon tetrachloride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, toluene, xylene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran and the like. Preferred are methylene, chloroform, 1,2-dichloroethane, trichloroethane, ethyl acetate and the like.
- the oxidizing agent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.
- hydrogen peroxide, peracetic acid, trifluoroperacetic acid, methachloroperbenzoic acid, oxone examples thereof include chromic acid and permanganese sun.
- hydrogen peroxide, metachloroperbenzoic acid, and the like are particularly preferable.
- the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1.2 to 10 equivalents to the compound represented by the above general formula [XXV]. Use 1.2 to 2 equivalents of oxidizing agent.
- the compound of Compound Example 116 was used as a representative compound, and the effect on GLP-1 concentration in plasma after administration of the compound was examined. .
- Control group 111 610 mg / kg group Based on the above results, 30 minutes after administration, plasma GLP_1 was significantly higher in the plasma of the group to which 1 Omg / kg of the compound was administered compared to the control group. Was. From these results, it was shown that the compound of the present invention has an activity capable of exhibiting a high blood GLP-1 concentration in rats.
- the compound of the present invention is useful as a therapeutic agent for diabetes mellitus, a preventive for chronic complications of diabetes or an antiobesity drug, since it exhibits an activity of exhibiting a high blood GLP-1 concentration.
- the compound of the general formula [I] of the present invention can be used as a medicament containing the same as an active ingredient, particularly as an agent for treating diabetes, an agent for preventing chronic complications of diabetes or an antiobesity agent.
- the compound of the present invention in a therapeutic agent, a preventive agent for chronic complications of diabetes or an anti-obesity agent means a pharmaceutically acceptable conventional one,
- RR 2 , R 3 , R 4 , R 5 , Ar and n have the above-mentioned meanings.
- Compounds represented by, E ester or a pharmaceutically acceptable salt in the force Rupokishiru group on R 5 or A r, salts in hydroxyl groups on R 5 or A r, or at the amino group on R 5 or A r Salts are included.
- Examples of the salt in the carboxyl group or the hydroxy group include alkali metal salts such as sodium salt and potassium salt; and alkaline earth metal salts such as calcium salt and magnesium salt.
- Examples of the acid addition salt for the amino group include inorganic salts such as hydrochloride, sulfate, nitrate, phosphate, carbonate, hydrogencarbonate and perchlorate; for example, acetate, propionate and lactic acid Organic salts such as salt, maleate, fumarate, tartrate, malate, citrate, and ascorbate; for example, methanesulfonate, isethionate, benzenesulfonate, toluenesulfonate, etc.
- Sulfonic acid salts for example, acidic amino acid salts such as aspartate and glutamate.
- the compound of the present invention when used as a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes, or an antiobesity agent, it can also be used as a pharmaceutically acceptable salt thereof.
- Typical examples of the pharmaceutically acceptable salts include, for example, salts with alkali metals such as sodium and potassium.
- the method for producing a pharmaceutically acceptable salt of the compound of the present invention can be carried out by appropriately combining methods usually used in the field of synthetic organic chemistry. Specific examples include neutralization titration of a free solution of the compound of the present invention with an alkaline solution.
- compositions of the present invention for treating diabetes, preventing chronic complications of diabetes or treating obesity Various forms can be selected as the dosage form for use as a tablet, for example, oral preparations such as tablets, capsules, powders, granules, and liquid preparations, and sterilized liquid parenteral preparations such as solutions and suspensions. Is mentioned.
- Solid preparations can be produced as they are in the form of tablets, capsules, granules or powders, or they can be produced using appropriate additives.
- the additive include sugars such as lactose and glucose; starches such as corn, wheat and rice; fatty acids such as stearic acid; inorganic salts such as sodium metasilicate, magnesium aluminate and anhydrous calcium phosphate; Synthetic polymers such as polyvinylpyrrolidone and polyalkylene glycol; fatty acids such as calcium stearate and magnesium stearate; alcohols such as stearyl alcohol and benzyl alcohol; methyl cellulose, carboxymethyl cellulose, and ethyl cellulose And synthetic cellulose derivatives such as hydroxypropylmethylcellulose, and other commonly used additives, such as water, gelatin, talc, vegetable oil, and gum arabic.
- These tablets, capsules, granules, powders and other solid preparations can generally contain 0.1 to 100% by weight, preferably 5 to 100% by weight, of the active ingredient.
- appropriate additives normally used in liquid preparations such as water, alcohols or plant-derived oils such as soybean oil, peanut oil, sesame oil, etc. It can be manufactured as a form.
- Particularly suitable solvents for parenteral administration include, for example, distilled water for injection, aqueous lidocaine hydrochloride (for intramuscular injection), physiological saline, aqueous dextrose, ethanol, and liquid for intravenous injection (for example, quinic acid). , Sodium citrate, etc.), an electrolyte solution (for example, intravenous drip, intravenous injection) and the like, or a mixed solution thereof.
- Liquid preparations such as suspensions or syrups for oral administration may contain 0.5 to 10% by weight of active ingredients.
- the practically preferred dosage of the compounds of the present invention may be adjusted as appropriate depending on the type of compound used, the type of composition formulated, the frequency of application and the particular site to be treated and the condition of the patient. .
- the daily dose per adult per day is 0.1 to 100 mg for oral administration and 100 mg / day for parenteral administration. It is 0.01 to 50 Omg.
- the frequency of administration varies depending on the administration method and symptoms, but it can be administered once or in 2 to 5 doses.
- the thin-layer chromatograph of the example has a plate of Si 1 i c ag e 160 F
- the silica gel for the column is Wakoge 1 TM C-300 (Wako Pure Chemical Industries), and the silica gel for the reversed-phase column is LC-SORB TM S PB-ODS (Chemco) or Y
- the solution was boiled for 90 minutes, cooled to room temperature, and acidified to pH ⁇ 2 with concentrated hydrochloric acid.
- the acidic solution was extracted with dichloromethane, and the combined extracts were dried and concentrated to give a red oil.
- This oil was treated with a 1 M aqueous sodium hydroxide solution, extracted with dichloromethane, the water tank was separated, and treated with concentrated hydrochloric acid to obtain an oil.
- the oil was extracted into dichloromethane, the extracts were combined, dried and concentrated under reduced pressure to give the desired orange-red oily product phenyl (2-sulfanylphenyl) methanone (36.4 g, 17 Ommo). 1) was obtained (80% yield).
- Step 1 The compound obtained in Step 1 (0.3 ml) was combined with the product of Step 2 (300 mg, 1.
- step 3 To a solution of the product of step 3 (180 mg, 0.64 Ommo 1) in trifluoroacetic acid (3.0 ml) was added 30% aqueous hydrogen peroxide (1.0 ml) at a temperature of about 0. After the addition was completed, the mixture was stirred at room temperature overnight, treated with a 10% aqueous sodium thiosulfate solution and a saturated aqueous sodium hydrogen carbonate solution, and extracted twice with ethyl acetate. And concentrated under reduced pressure. The residue and 4-hydrogen chloride 1,4-dioxane solution (3.0 ml) were mixed, and zinc powder (200 mg, 3.06 mmol 1) was gradually added.
- Diastereomer a (A r: Ph; R 1 : H; R 2 : H; 3 : i-Pr; R 4 : H
- Diastereomer b (Ar: Ph j R ⁇ H .- R 'r H j R 3 : i -P r; R 4 : H
- Diastereomer a (Ar i Ph j R ⁇ H j R ⁇ H i R 3 : i- 1 Pr; R 4 : H
- Diastereomer b (Ar i P j R ⁇ H j R ⁇ H i R 3 : i-P r; 4 : H
Landscapes
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Diabetes (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Obesity (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Veterinary Medicine (AREA)
- Hematology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Child & Adolescent Psychology (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
技 術 分 野 Technical field
本発明は医薬の分野において明有用である。 更に詳しくは、 本発明化合物のベン ゾチアゼピン誘導体は、 高い血中 G L P— 1濃度を呈する活性を示していること から、 糖尿病治療剤、 糖尿病の慢性合併田症の予防剤又は抗肥満薬として有用であ る。 背 景 技 術 The present invention is clearly useful in the field of medicine. More specifically, since the benzothiazepine derivative of the compound of the present invention has an activity of exhibiting a high blood GLP-1 concentration, it is useful as a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes or an antiobesity agent. is there. Background technology
健常人の血糖値は、 インスリンの作用により、 一定にコントロールされている 。 糖尿病とは、 このコントロールが効かなくなり慢性的に高血糖状態を呈するこ と及びそのことによって引き起こされる疾患をいう。 The blood sugar level of a healthy person is constantly controlled by the action of insulin. Diabetes refers to a chronic hyperglycemic state in which this control is ineffective and a disease caused by it.
糖尿病治療の根本は高血糖状態の是正、すなわち血中グルコース濃度を正常値 に戻すことにあるが、 近年では特に、 空腹時血糖に影響を与えることなく、 食後 の急激な血糖値の上昇を如何に抑えるかという点が治療上極めて重要であると 認識されてきている。 The basis of diabetes treatment is to correct hyperglycemia, i.e., to return blood glucose levels to normal levels.In recent years, however, it has been particularly important in recent years to prevent a rapid rise in blood glucose after eating without affecting fasting blood glucose. It has been recognized that it is extremely important in terms of treatment to control the amount of the drug.
現在、 臨床現場において使用されている主な糖尿病治療薬は、 各種インスリン 製剤の他に大別して以下の三種類の治療薬が存在する。第 1はスルホニルゥレア 剤に代表されるインスリン放出薬と呼ばれる薬剤群で、 この薬剤は滕臓から直接 インスリン分泌を促進し血糖値を下げるものである。第 2は近年上市されたイン スリン抵抗性改善剤と呼ばれるもので、 この薬剤は直接ィンスリン放出を助長す ることなく、末梢組織での糖の取り込みを促進することにより血糖値を下げる薬 剤である。 第 3の 一グルコシダ一ゼ阻害剤は、 消化管における糖質の消化、 吸 収を遅延し、一時的な食後の血糖値の上昇を抑えることにより、 血糖値の急激な 上昇を制御する薬である。 . At present, there are three main types of diabetes therapeutic agents used in clinical practice, in addition to various insulin preparations, as follows. The first is a group of drugs called insulin-releasing drugs typified by sulfonylprea drugs, which promote insulin secretion directly from the brain and lower blood sugar levels. The second is a drug that has recently been launched, called an insulin sensitizer, that lowers blood glucose by promoting glucose uptake in peripheral tissues without directly promoting insulin release. is there. The third monoglucosidase inhibitor is a drug that controls the rapid rise in blood glucose by delaying the digestion and absorption of carbohydrates in the gastrointestinal tract and suppressing the temporary rise in blood glucose after meals. is there. .
一方、 グルカゴン様ペプチド— 1 (以下、 G L P— 1という。 ) は、 食事の剌 激等によって、 小腸腸管上皮に存在する内分泌細胞である L細胞から分泌される ホルモンであり、 勝臓ランゲルハンス島に存在する β細胞に作用してィンスリン 分泌を促進することにより血糖を降下させることがわかっている (Eu r. J. C l i n. I n v e s t、 第 22巻、 第 154頁、 1992年) 。 GL P _ 1に よるインスリン分泌作用は、 血糖値に依存しており、 正常血糖時では GLP— 1 によるィンスリン分泌は認められず、 高血糖時にのみィンスリン分泌の亢進が見 られることが報告されている (Lanc e t、 第 2巻、 第 1300頁、 1987 年) 。 GLP— 1はインスリン分泌を亢進するだけでなく、 インスリンの生合成 を高め (End o c r i no l ogy, 第 130巻、 第 159頁、 1992年) 、 また、 ]3細胞の増殖を促す (D i a b e t o 1 o g i a, 第 42巻、 第 856 頁、 1999年) ことから、 8細胞の維持にとっても欠かすことの出来ない因子 である。 On the other hand, glucagon-like peptide-1 (hereinafter referred to as GLP-1) It is a hormone secreted by L cells, which are endocrine cells present in the small intestinal intestinal epithelium due to intensification, etc., and acts on β cells present in the islets of Langerhans to promote the secretion of insulin to lower blood sugar. I know (Eur. J. Clin. Invest, Vol. 22, p. 154, 1992). It has been reported that insulin secretion by GLP_1 depends on blood glucose level.Insulin secretion by GLP-1 is not observed in normoglycemia, and it is reported that insulin secretion is enhanced only in hyperglycemia. (Lanc et, Vol. 2, p. 1300, 1987). GLP-1 not only enhances insulin secretion but also enhances insulin biosynthesis (Endocri no lgygy, 130, 159, 1992) and promotes the proliferation of] 3 cells (Diabeto 1 ogia, Vol. 42, p. 856, 1999), which is an essential factor for maintaining 8 cells.
一般の I I型糖尿病患者への G LP— 1投与によって血中 GLP— 1濃度を 高値に維持した結果、 高血糖状態が有意に改善されることが示されており、 医療 現場においてもその糖尿病に対しての有効性が確認されている(D i a b e t o 1 o g i a、 第 36巻、 第 741頁、 1994年又は同、 第 39巻、 第 1546 頁、 1996年) It has been shown that maintaining GLP-1 in blood at a high level by administering GLP-1 to general type 2 diabetic patients significantly improves hyperglycemic status. (Diabeto 1 ogia, 36, 741, 1994 or 39, 1546, 1996)
さらには、 GLP— 1の作用点は i3細胞だけにとどまらず、 末梢組織において 糖の利用を亢進することが認められ (Endo c r i no l ogy、 第 135巻 、 第 2070頁、 1994年又は D i a b e t o 1 o g i a、 第 37巻、 第 11 63頁、 1994年) 、 また、 GLP— 1の脳室内投与により摂食抑制作用を示 すとの報告がなされている (D i ge s t i on, 第 54巻、 第 392頁、 19 93年) 。 さらに、 GLP— 1投与により消化管運動抑制作用があることも報告 されている (D i g. D i s. S c i. 、 第 43巻、 第 1113頁、 1998年 ) 。 Furthermore, the action point of GLP-1 was not limited to i3 cells, but was also found to enhance sugar utilization in peripheral tissues (Endocrinology, 135, 2070, 1994 or Diabeto 1 ogia, 37, 1163, 1994), and it has been reported that intracerebroventricular administration of GLP-1 exerts an antifeeding effect (Digestion, Vol. 54). , 392, 1993). It has also been reported that GLP-1 administration has a gastrointestinal motility inhibitory effect (Dig. Dis. Sci., 43, 1113, 1998).
本発明の化合物と構造が最も近似する化合物は、特表平 7— 503724号公 報 (以下、 引例 Aという。 ) 、 特表平 8— 506376号公報 (以下、 引例 Bと いう。 ) 、 特表平 8— 507049号公報 (以下、 引例 Cという。 ) 、 特表平 1 0- 504035号公報 (以下、 引例 Dという。 ) 、 特表平 10— 279568 号公報 (以下、 引例 Eという。 ) 、 国際公開番号 WOO 0 61568号公報 ( 以下、 引例 Fという。 ;) 、 J. Li p i d Re s. , 36、 第 1106頁、 1 995年 (以下、 引例 Gという。 ) 、 J. Li p i d Re s. 、 36、 第 1 098頁、 1995年 (以下、 引例 Hという。 ;) 、 D r ug Me t a b. D i s po s. 、 27、 第 637頁、 1999年 (以下、 引例 Iという。 ) 、 C u r r. Op i n. Ca r d i ova s c. , Rena l I nve s t D r ug s、 1、 第 276頁、 1999年 (以下、 引例 Jという。 ) 、 J . L i p i d Re s. , 40, 第 2158頁, 1999年 (以下、 引例 Kという。 ) 、 Ana l. B i o c hem. , 282, 第 94頁, 2000年 (以下、 引例 L という。 ) 、 国際公開番号 WOO 0Z38725号公報 (以下、 引例 Mという。 ) 、 国際公開番号 WO00/38726号公報 (以下、 引例 Nという。 ) 、 国際 公開番号 WO00/38727号公報 (以下、 引例 0という。 ) 、 国際公開番号 WO00/38728号公報 (以下、 引例 Pという。 ) 、 国際公開番号 WO 00 /38729号公報 (以下、 引例 Qという。 ) 及び国際公開番号 WO00/55 355号公報 (以下、 引例 Rという。 ) に記載されている。 The compounds whose structures are most similar to the compounds of the present invention are described in JP-A-7-503724 (hereinafter referred to as Reference A), JP-A-8-506376 (hereinafter referred to as Reference B), and the like. Japanese Patent Publication No. 8-5050749 (hereinafter referred to as Reference C), Japanese Patent Publication No. 10-504035 (hereinafter referred to as Reference D), Japanese Patent Publication No. 10-279568. Publication No. WOO 0 61568 (hereinafter referred to as Reference F.), J. Lipid Res., 36, 1106, 1995 (hereinafter referred to as Reference). G.), J. Lipid Res., 36, p. 1098, 1995 (hereinafter referred to as Reference H;), Dr. Metatab. D is pos., 27, p. 637 , 1999 (hereinafter referred to as Reference I), Curr. Opin. Cardiova sc., Renal Instst Drugs, 1, p. 276, 1999 (hereinafter referred to as Reference J). ), J. Lipid Res., 40, p. 2158, 1999 (hereinafter referred to as Reference K), Anal. Biochem., 282, p. 94, 2000 (hereinafter referred to as Reference L). ), International Publication No. WOO 0Z38725 (hereinafter referred to as Reference M), International Publication No. WO00 / 38726 (hereinafter referred to as Reference N), and International Publication No. WO00 / 38727 (hereinafter referred to as Reference 0). ), International Publication Number WO00 / 3 No. 8728 (hereinafter referred to as Reference P), International Publication No. WO 00/38729 (hereinafter referred to as Reference Q), and International Publication No. WO00 / 55355 (hereinafter referred to as Reference R). ing.
引例 Aから Rには、 1、 4一べンゾチアゼピン骨格を有している化合物が記載 されている。 References A to R describe compounds having a 1,4-benzothiazepine skeleton.
しかしながら、 本発明の化合物は、 1、 4—ベンゾチアゼピン 1、 1—ジォキ シド骨格は引例 Aから Rの化合物と共通するものの、該骨格上 3位置換基部分が 1置換化合物であり、 直鎖状 — C6、 2置換アルキル基を有する 、 具体的に は例えばェチル基、 ブチル基などの 2置換を有する引例 Aから Rの化合物とは、 全く構造が異なる化合物である。 However, in the compound of the present invention, although the 1,4-benzothiazepine 1,1-dioxoside skeleton is common to the compounds of References A to R, the 3-position substituent on the skeleton is a monosubstituted compound. Linear — A compound having a C 6 , disubstituted alkyl group, specifically a compound having a completely different structure from the compounds of References A to R having disubstitution, for example, an ethyl group or a butyl group.
また、 引例 Aから Rの用途は、 胆汁酸取込阻害作用に起因する血脂減少化合物 であり、 本発明とは、 産業上の利用分野は同じであるが、 本発明の用途とは異な る全く関連性のない用途である。 また該特許に開示されている化合物が、糖尿病 治療薬、糖尿病の慢性合併症の予防剤として有用であることは該特許明細中には 開示されていない。 The uses of References A to R are hypolipidemic compounds caused by the inhibitory action of bile acid uptake, and have the same industrial application fields as the present invention, but are completely different from the uses of the present invention. This is an unrelated application. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
さらに、 米国特許第 3362962号公報 (以下、 引例 Sという。 ) には、 1 、 4一べンゾチアゼピン骨格およびその酸化体、 還元体具体的には 1、 4一ベン ゾチアゼピン骨格の 4、 5—ジヒドロ体、 4、 5—ジヒドロ, N-酸化体および 4 、 5一才キサジリジン体などの化合物などの骨格が記載されている。 Further, US Pat. No. 3,362,962 (hereinafter referred to as Reference S) discloses a 1,4-benzothiazepine skeleton and its oxidized and reduced forms, specifically, 1,4-benzen. Scaffolds such as 4,5-dihydro, 4,5-dihydro, N-oxidized, and 4,5-year-old oxaziridine derivatives of the zothiazepine skeleton are described.
しかしながら、 本発明の化合物は、 1、 4一べンゾチアゼピン骨格は引例 Jの 化合物と共通するものの、 ベンゾチアゼピン骨格の 4—ジヒドロ体、 4一ジヒド 口, N-酸化体および 4、 5—才キサジリジン体は、 本発明の範囲外で全く構造が 異なる化合物である。 また、 引例 Sの用途は、 筋弛緩および抗痙攣活性を有する 化合物で、産業上の利用分野は同じであるが、本発明の用途とは全く関連性のな い用途である。 また該特許に開示されている化合物が、 糖尿病治療薬、 糖尿病の 慢性合併症の予防剤として有用であることは該特許明細中には開示されていな い。 However, although the compounds of the present invention have a 1,4-benzothiazepine skeleton in common with the compound of Reference J, the 4-benzodiazepine skeleton has a 4-dihydro form, a 4-dihydroform, an N-oxidized form, and a 4,5-year-old compound. Oxaziridines are compounds with completely different structures outside the scope of the present invention. The use of Reference S is a compound having a muscle relaxant and anticonvulsant activity and has the same industrial field of application, but is completely unrelated to the use of the present invention. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
さらにまた、 特開昭 5 3— 6 5 8 9 5号公報 (以下、 引例 Tという。 ) には、 1, 4一べンゾチアゼピン骨格を含む 3環縮合環、 ピロール [ 1, 2— d ] - 1 、 4一べンゾチアゼピン骨格を有する化合物が記載されている。 Furthermore, Japanese Patent Application Laid-Open No. 53-65895 (hereinafter referred to as Reference T) discloses a three-ring fused ring containing a 1,4-benzothiazepine skeleton, pyrrole [1,2-d]-. Compounds having a 1,4-benzothiazepine skeleton have been described.
しかしながら、 引例 Tは 3環縮合環であり、 本発明の範囲外で、 全く構造が異 なる化合物である。 また、 引例 Tの用途は、 自発運動抑制作用を有する化合物で 、産業上の利用分野は同じであるが、 本発明の用途とは全く関連性のない用途で ある。 また該特許に開示されている化合物が、 糖尿病治療薬、 糖尿病の慢性合併 症の予防剤として有用であることは該特許明細中には開示されていない。 However, Reference T is a condensed tricyclic ring, and is a compound having a completely different structure outside the scope of the present invention. The use of Reference T is a compound having a locomotor activity-suppressing action, and although it has the same industrial field of application, it has no relation to the use of the present invention. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
また、 米国特許第 5 2 7 6 0 2 5号公報 (以下、 引例 Uという。 ) には、 1, 4一べンゾチアゼピン骨格を含むチェノ [ 3 , 2 - f ] _ 1、 4一べンゾチアゼ ピン骨格を有する化合物が記載されている。 U.S. Pat. No. 5,276,025 (hereinafter referred to as Reference U) discloses a cheno [3,2-f] _1,4,1-benzothiazepine containing a 1,4-benzothiazepine skeleton. Compounds having a skeleton are described.
しかしながら、 引例 Uは該骨格上 5位置換基部分が、 水素、 直鎖状じェ一 C 5 アルキル基あるいは C 3— C 7環状アルキル基を有する化合物で 、 本発明の化合 物が置換基を有してもよい芳香環あるいは複素環である点で、全く構造が異なる 化合物である。 眼圧上昇抑制および緑内障治療薬で、産業上の利用分野は同じで あるが、 本発明の用途とは全く関連性のない用途である。 また該特許に開示され ている化合物が、糖尿病治療薬、糖尿病の慢性合併症の予防剤として有用である ことは該特許明細中には開示されていない。 However, Reference U is a compound in which the substituent at the 5-position on the skeleton has hydrogen, a straight-chain C 5 alkyl group or a C 3 -C 7 cyclic alkyl group, and the compound of the present invention has a substituent. The compound has a completely different structure in that it may have an aromatic ring or a heterocyclic ring. Although it has the same industrial field of application as a drug for suppressing intraocular pressure rise and for treating glaucoma, it is a use that has no relation to the use of the present invention. Further, it is not disclosed in the patent specification that the compound disclosed in the patent is useful as a therapeutic agent for diabetes or an agent for preventing chronic complications of diabetes.
現在では、 糖尿病治療薬として、 例えばスルホニルゥレア剤、 インスリン抵抗 性改善剤、 一ダルコシダーゼ阻害剤等の薬剤が臨床で多く用いられているが、 以下のような問題点が挙げられ、 これらは十分な薬剤であるといえない。 すなわ ち、 スルホニルゥレア剤は、 効果の発現が遅くしかも作用持続時間が長いため、 食後の高血糖時に合わせ作用を発現させるのが難しく、 さらには空腹時の血糖を 降下させしばしば生命にかかわるような重篤な低血糖発作を引き起こす場合もあ る。 インスリン抵抗性改善剤は、 肝臓に対する副作用がしばしば問題となってお り、 厳密な管理下における慎重な使用が必要となっている。 また浮腫等の副作用 を引き起こす場合もある。 また、 ひ一ダルコシダーゼ阻害剤には、 膨満感ゃ下痢 などの副作用が問題となっている。 At present, antidiabetic drugs such as sulfonylprea drugs, insulin resistance Drugs such as sex-improving agents and mono-dalcosidase inhibitors are widely used in the clinic, but have the following problems, and they cannot be said to be sufficient drugs. In other words, sulfonylurea agents have a slow onset of action and a long duration of action, making it difficult to develop a synergistic effect during postprandial hyperglycemia, and also lower fasting blood sugar and are often life-threatening Such severe hypoglycemic attacks can be caused. Insulin sensitizers often have side effects on the liver and require careful use under strict control. It may also cause edema and other side effects. In addition, side effects such as bloating and diarrhea have become a problem with Hi-Dalcosidase inhibitors.
したがって、 昨今の糖尿病患者の増加に伴い、 現在、 副作用がなく、 血糖値に 依存して血糖値を制御できる一層有益な糖尿病治療薬の開発が臨床の現場から求 められている。 発 明 の 開 示 Therefore, with the recent increase in the number of diabetic patients, clinical sites are now demanding the development of more beneficial antidiabetic drugs that have no side effects and can control blood glucose levels in dependence on blood glucose levels. Disclosure of the invention
本発明の目的は、 高い血中 G L P— 1濃度を呈する活性を示していることから 、 糖尿病治療剤、 糖尿病の慢性合併症の予防剤又は抗肥満薬等を提供することで あ 。 An object of the present invention is to provide a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes, an anti-obesity drug, and the like, since they exhibit an activity of exhibiting a high blood GLP-1 concentration.
本発明者らは、 血糖値に依存して血糖値を制御できる糖尿病治療薬、 糖尿病の 慢性合併症の予防剤又は抗肥満薬の創製を目的に鋭意研究を進めてきたところ、 一般式 [ I ] The present inventors have intensively studied for the purpose of creating a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes, or an anti-obesity agent capable of controlling blood glucose level depending on the blood glucose level. ]
[式中、 ; 1及び R 2は同一又は異なって、 水素原子、 C — C^アルキル基、 Wherein, 1 and R 2 are the same or different and each represents a hydrogen atom, a C—C ^ alkyl group,
R 3は水素原子、 C,— C 6アルキル基 (但し、 n—ブチル基は除く) 、 R 4は水素原子、 水酸基、 一 C3アルキル基、 R 3 is a hydrogen atom, a C, —C 6 alkyl group (excluding an n-butyl group), R 4 is a hydrogen atom, a hydroxyl group, a C 3 alkyl group,
R5は水素原子、 アジド基、 アミノ基、 力ルバモイル基、 力ルバモイルァミノ 基、 カルパモイルォキシ基、 力ルポキシル基、 シァノ基、 スルファモイル基、 ス ルホ基、 ニトロ基、 ハロゲン原子、 ヒドロキシ基、 ホルミル基、 ホルミルアミノ 基、 環状の飽和若しくは不飽和の C3— C9脂肪族基、 ァラルキル基、 N—ァラル キルアミノ基、 N, N—ジァラルキルアミノ基、 ァラルキルォキシ基、 ァラルキ ルカルポニル基、 N -ァラルキル力ルバモイル基、 ァリール基、 N—ァリ一ルァ ミノ基、 N, N—ジァリールアミノ基、 ァリールォキシ基、 ァリ一ルスルホニル 基、 ァリ一ルスルホニルォキシ基、 N—ァリ一ルスルホニルァミノ基、 N—ァリ 一ルスルホニルァミノ Ci— Ceアルキルアミノ基、 N—ァリ一ルスルホニルアミ ノ 一 C10アルキル力ルバモイル基、 N—ァリ一ルスルホニルァミノ C i一 C 6 アルコキシカルポニル基、 ァリールスルファモイル基、 ァリールスルファモイル ォキシ基、 7リ一ルスルファモイル C x― C 0アルキルカルパモイル基、 ァリ一 ルスルファモイル〇1_(:6アルコキシカルポニル基、 N—ァリ一ルカルバモイル 基、 ァロイル基、 ァロキシ基、 N—ァロイル基、 N— ( N—ァロイルァミノ) C !-C10アルキル力ルバモイル基、 N—ァロイルァミノ C — C 6アルコキシカル ポニル基、 C2_C6アルカノィル基、 C2-C 6ァルカノィルォキシ基、 N-C2 一 C6アルカノィルァミノ基、 N, N—ジ— C2_C6アルカノィルァミノ基、 N — Ci— C6アルキルアミノ基、 N, N—ジ— 一 C6アルキルアミノ基、 N-C !-C10アルキル力ルバモイル基、 N, N—ジー Ci-CiQアルキル力ルバモイ ル基、 N— C 2 _ C 6アルケニルカルバモイル基、 N, N—ジー C2— C6アルケニ ルカルバモイル基、 N—ァミノ ^— C^アルキル力ルバモイル基、 N-Cx-C 6アルコキシ C ,~010アルキル力ルバモイル基、 N— C C6アルコキシカルボ ニル 一 C10アルキル力ルバモイル基、 N— C i _ C 6アルコキシカルポニルァ ミノ 一 C10アルキル力ルバモイル基、 N - C「C6アルコキシカルポニルァ ミノ C — Ceアルコキシカルポニル基、 C — Ceアルキルチオ基、 N— — C6 アルキルスルファモイル基、 N, N—ジー C 一 C 6アルキルスルファモイル基、 C 一 C 6アルキルスルフィニル基、 d— C6アルキルスルホニル基、 c:-c6 アルコキシ基、 Ci— C 6アルコキシカルポニル基、 アミノじ丄一 Cfiアルコキシ カルポニル基、 N_C3— < 6シクロアルキルアミノ基、 N, N—ジー C3— C6 シクロアルキルアミノ基、 C3—じ6シクロアルキルォキシ基、 N— C3— C6シ クロアルキル力ルバモイル基、 N, N—ジ— C3— ( 6シクロアルキル力ルバモイ ル基からなる群から選択される置換基、 5又は 6員の複素環基、 窒素原子、 酸素 原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1ないし 5個 有する 1ないし 3環性の複素芳香環基並びに該置換基で置換されていてもよい、 直鎖若しくは分枝鎖の飽和若しくは不飽和の C!― C 9脂肪族基、 C i一 C 6アルコ キシ基、 一 C6アルキルチオ基、 Ν— — C6アルキルアミノ基、 N— Ci— C 6アルキル力ルバモイル基、 N-Ci-C 6アルキルチォカルバモイル基からな る群より選ばれる置換基を有してしてもよい炭素芳香環基、 1ないし 3環性の C 7-C15炭素芳香環基又は 5若しくは 6員の複素環基若しくは窒素原子、 酸素原 子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1ないし 5個有 する 1ないし 3環性の複素芳香環基 (5又は 6員の複素環基を除く。 ) 、 R 5 is a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, a carbamoyloxy group, a carboxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, a formyl group Group, formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkylcarbonyl group, N -Aralkyl groups: rubamoyl group, aryl group, N-arylamino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfonyloxy group, N-aryl Suruhoniruamino group, N- § Li one Le sulfonyl § amino CI- Ce alkylamino group, N- § Li one Rusuruhoniruami Roh one C 10 Al Le force Rubamoiru group, N- § Li one Le sulfonyl § amino C i one C 6 alkoxy Cal Poni group, § reel sulfamoyl group, § reel sulfamoyl Okishi group, 7 Li one Rusurufamoiru C x - C 0 alkyl Scarpa moil group, Arylsulfamoyl〇 1 _ (: 6 alkoxycarbonyl group, N-arylcarbamoyl group, aroyl group, alkoxy group, N-aroyl group, N- (N-aroylamino) C! -C 10 alkyl rubamoyl group, N- Aroiruamino C - C 6 Arukokishikaru Poniru group, C 2 _C 6 Arukanoiru group, C 2 -C 6 § Luke Noi Ruo alkoxy group, NC 2 one C 6 alkanoyloxy Noi Rua amino group, N, N- di - C 2 _C 6 alkanoylamino group, N — Ci—C 6 alkylamino group, N, N—di-C 6 alkylamino group, NC! -C 10 alkyl rubamoyl group, N, N—Gi Ci-CiQ alkyl Lubamoyl group, NC 2 _ C 6 alkenyl carbamoylmethyl group, N, N- di C 2 - C 6 alkenyl carbamoyl group, N- Amino ^ - C ^ alkyl force Rubamoiru group, NC x -C 6 alkoxy C, ~ 0 10 alkyl force Rubamoyl group, N—CC 6 alkoxycarbonyl-C 10 alkyl group Rubamoyl group, N—C i _C 6 alkoxycarbonylamino—C 10 alkyl group Lubamoyl group, N—C “C 6 alkoxycarbonylamino C — Ce Alkoxycarbonyl group, C — Ce alkylthio group, N— — C 6 alkylsulfamoyl group, N, N—G C 1 C 6 alkylsulfamoyl group, C 1 C 6 alkylsulfinyl group, d—C 6 alkylsulfonyl Group, c : -c 6 alkoxy group, Ci—C 6 alkoxy carbonyl group, amino di-C fi alkoxy Carbonyl group, N_C 3 — < 6 cycloalkylamino group, N, N-di C 3 — C 6 cycloalkylamino group, C 3 — 6 cycloalkyloxy group, N—C 3 — C 6 cycloalkyl force A group selected from the group consisting of a rubamoyl group, a N, N-di-C 3 — ( 6- cycloalkyl group and a 5- or 6-membered heterocyclic group, a nitrogen atom, an oxygen atom, and a sulfur atom A mono- to tri-cyclic heteroaromatic group having 1 to 5 heteroatoms per ring system selected from the group consisting of a straight or branched saturated or unsaturated C ! - C 9 aliphatic groups, C i one C 6 alkoxy group, one C 6 alkylthio group, Nyu- - C 6 alkylamino group, N-CI- C 6 alkyl force Rubamoiru group, N-Ci-C 6 alkyl A carbon atom which may have a substituent selected from the group consisting of thiocarbamoyl groups; Kowamoto, 1 to tricyclic C 7-C 15 carbon ring group or a 5- or 6-membered heterocyclic group or a nitrogen atom, a heteroatom 1 ring system selected from the group consisting of SansoHara bar and a sulfur atom 1 to 5 mono- to 3-cyclic heteroaromatic groups (excluding 5- or 6-membered heterocyclic groups),
Arは、 水素原子、 アジド基、 アミノ基、 力ルバモイル基、 力ルバモイルアミ ノ基、 力ルバモイルォキシ基、 力ルポキシル基、 シァノ基、 スルファモイル基、 スルホ基、 ニトロ基、 ハロゲン原子、 ヒドロキシ基、 ホルミル基、 ホルミルアミ ノ基、 環状の飽和若しくは不飽和の C 3— C 9脂肪族基、 ァラルキル基、 N—ァラ ルキルアミノ基、 N, N—ジァラルキルアミノ基、 ァラルキルォキシ基、 ァラル キルカルポニル基、 N -ァラルキル力ルバモイル基、 ァリール基、 N—ァリール アミノ基、 N, N—ジァリールアミノ基、 ァリールォキシ基、 ァリールスルホニ ル基、 ァリ一ルスルホニルォキシ基、 N—ァリ一ルスルホニルァミノ基、 N—ァ リ一ルスルホニルァミノ 一 C6アルキルァミノ基、 N—ァリ一ルスルホニルァ ミノ C〖一 C10アルキル力ルバモイル基、 N—ァリ一ルスルホニルァミノ Cェ一 C 6アルコキシカルポニル基、 ァリ一ルスルファモイル基、 ァリ一ルスルファモイ ルォキシ基、 7リ一ルスルファモイル C x - C 0アルキル力ルバモイル基、 ァリ 一ルスルファモイル。 _ C 6アルコキシカルポニル基、 N—ァリ一ルカルバモイ ル基、 ァロイル基、 ァロキシ基、 N—ァロイル基、 N— ( N—ァ口ィルァミノ) C「C10アルキル力ルバモイル基、 N—ァロイルァミノ 一 C6アルコキシ力 ルポニル基、 C2— C6アルカノィル基、 C2— C6アルカノィルォキシ基、 N— C 2— C6アルカノィルァミノ基、 N, N—ジー C2— C6アルカノィルァミノ基、 N— C^— C6アルキルアミノ基、 N, N—ジー 一 C6アルキルアミノ基、 N— Ci— 。アルキル力ルバモイル基、 N, N -ジ— 一 。アルキルカルパモ ィル基、 N— C 2— C 6アルケニルカルバモイル基、 N, N—ジー C2— C6アルケ 二ルカルバモイル基、 N—アミノ(^一 。アルキル力ルバモイル基、 N— 一 C 6アルコキシ 一 G10アルキル力ルバモイル基、 N-C x-C6アルコキシ力 ルポニル C!一 C 。アルキル力ルバモイル基、 N - C「 C6アルコキシ力ルポ二 ルァミノじェ一 C10アルキル力ルバモイル基、 N-C i-Ceアルコキシカルボ二 ルァミノ(^— Ceアルコキシカルポニル基、 一 C6アルキルチオ基、 N-C i 一 C 6アルキルスルファモイル基、 N, N—ジー C — C 6アルキルスルファモイ ル基、 C!― C 6アルキルスルフィニル基、 C.-Ceアルキルスルホニル基、 C1 —C6アルコキシ基、 C 1― C 6アルコキシカルボニル基、 アミ 一 C6アルコ キシカルポニル基、 N— C 3— C 6シクロアルキルァミノ基、 N, N—ジ— C3— C6シクロアルキルアミノ基、 C 3— C 6シク口アルキルォキシ基、 N-C3-C6 シクロアルキル力ルバモイル基、 N, N—ジー C3— ( 6シクロアルキルカルバモ ィル基からなる群から選択される置換基、 5又は 6員の複素環基、 窒素原子、 酸 素原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1ないし 5 個有する 1ないし 3環性の複素芳香環基又は該置換基で置換されていてもよい、 ' 直鎖若しくは分枝鎖の飽和若しくは不飽和の C丄— C 9脂肪族基、 C ,一 C 6アルコ キシ基、 Ci— Ceアルキルチオ基、 N— 一 C6アルキルアミノ基、 N—。ェ— C 6アルキル力ルバモイル基、 N-C .-Ceアルキルチオ力ルバモイル基からな る群より選ばれる置換基を有していてもよい炭素芳香環基、 1ないし 3環性の C 7— C15炭素芳香環基又は 5若しくは 6員の複素環基、 若しくは窒素原子、 酸素 原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1ないし 5個 有する 2ないし 3環性の縮合複素芳香環基 (伹し、 5若しくは 6員の複素環基を 除く。 ) 、 nは 0— 2の整数を示す。 ] で表される化合物が生体内において高い 血中 GL P— 1濃度を達成することを見出し、 本発明を完成した。 Ar represents a hydrogen atom, an azide group, an amino group, a rubamoyl group, a rubamoylamino group, a rubamoyloxy group, a lipoxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, a formyl group, Formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkylcarbonyl group, N -Aralkyl groups: rubamoyl group, aryl group, N-aryl amino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfonyloxy group, N-arylsulfonylamino group, N - § Li one Le sulfonyl § Mino one C 6 Arukiruamino group, N- § Li one Rusuruhonirua amino C 〖one C 10 Alkyl force Rubamoiru group, N- § Li one Le sulfonyl § amino C E one C 6 alkoxy Cal Poni group, § Li one Rusurufamoiru group, § Li one Rusurufamoi Ruokishi group, 7 Li one Rusurufamoiru C x - C 0 alkyl force Rubamoiru group, Alisulfamoyl. _ C 6 alkoxycarbonyl group, N-aryl carbamoyl group, aroyl group, alkoxy group, N-aroyl group, N— (N-arylamino) C “C 10 alkyl carbamoyl group, N-aroylamino 1 C 6 Alkoxy force Luponyl group, C 2 —C 6 alkanoyl group, C 2 —C 6 alkanoyloxy group, N—C 2 — C 6 alkanoylamino group, N, N—di C 2 — C 6 alkanoylamino group, N—C ^ —C 6 alkylamino group, N, N—di-C 6 alkylamino group, N — Ci—. Alkyl rubamoyl groups, N, N-di-I. Alkyl Scarpa model I le group, N- C 2 - C 6 alkenyl carbamoylmethyl group, N, N- di C 2 -. C 6 alkenyl carbamoylmethyl group, N- amino (^ primary alkyl force Rubamoiru group, N- one C 6 Alkoxy G 10 Alkyl Lubamoyl group, NC x -C 6 Alkoxy Luponyl C! -1 C. Alkyl Lubamoyl group, N-C "C 6 Alkoxy Lubamoyl group C 10 Alkyl Lubamoyl group, NC i -Ce alkoxycarbonylamino (^ — Ce alkoxycarbonyl group, 1 C 6 alkylthio group, NC i 1 C 6 alkylsulfamoyl group, N, N-di C — C 6 alkylsulfamoyl group, C! — C 6 alkylsulfinyl group, C.-Ce alkylsulfonyl group, C 1 -C 6 alkoxy group, C 1 - C 6 alkoxycarbonyl group, amino one C 6 alkoxy Kishikaruponiru group, N-C 3 - C 6 cycloalkyl § Mino group N, N-di - C 3 - C 6 cycloalkylamino group, C 3 - C 6 consequent opening Arukiruokishi group, NC 3 -C 6 cycloalkyl force Rubamoiru group, N, N-di-C 3 - (6 cycloalkyl carba A substituent selected from the group consisting of a moly group, a 5- or 6-membered heterocyclic group, and 1 to 5 hetero atoms per ring system selected from the group consisting of a nitrogen atom, an oxygen atom and a sulfur atom A linear or branched saturated or unsaturated C 丄 -C 9 aliphatic group, which may be substituted with a 1 to 3 cyclic heteroaromatic group or the substituent, a C 1, 1 C 6 alcohol alkoxy group, CI- Ce alkylthio group, N- one C 6 alkylamino group, N- E -. C 6 alkyl force Rubamoiru group has a NC-Ce. alkylthio force Rubamoiru substituents selected from the group ing from the base which may be a carbon aromatic ring group, 1 to 3 rings of the C 7 - C 15 carbon ring group or 5 young Or a 6-membered heterocyclic group, or a 2- or 3-cyclic fused heteroaromatic group having 1 to 5 heteroatoms per ring system selected from the group consisting of nitrogen, oxygen and sulfur , And 5 or 6 membered heterocyclic groups are excluded.), And n is an integer of 0 to 2.] It has been found that the compound represented by the formula (1) achieves a high blood GLP-1 concentration in vivo. Completed the invention.
本発明は、 ベンゾチアゼピン誘導体及びその用途に関するものであり、 これら の発明は、 文献に未記載の新規なものである。 次に本明細書中に記載された各種記号及び用語について説明する。 The present invention relates to benzothiazepine derivatives and uses thereof, and these inventions are novel ones not described in the literature. Next, various symbols and terms described in this specification will be described.
Cエー C 3アルキル基とは、 例えばメチル基、 ェチル基、 プロピル基等が挙げら れ、 中でもメチル基、 ェチル基等が好ましい。 The C er C 3 alkyl group such as methyl group, Echiru group, a propyl group can be mentioned, et al are, inter alia methyl, and the like Echiru group.
一 C 6アルキル基とは、 例えばメチル基、 ェチル基、 プロピル基、 イソプロ ピル基、 イソブチル基、 s e c—ブチル基、 t e r t 一ブチル基、 ペンチル基、 ネオペンチル基、 へキシル基、 イソへキシル基が挙げられ、 中でもイソプロピル 基、 イソブチル基、 s e c—ブチル基、 ネオペンチル基、 イソへキシル基等が好 ましい。 A C 6 alkyl group includes, for example, a methyl group, an ethyl group, a propyl group, an isopropyl group, an isobutyl group, a sec-butyl group, a tert-butyl group, a pentyl group, a neopentyl group, a hexyl group, and an isohexyl group. Among them, isopropyl group, isobutyl group, sec-butyl group, neopentyl group, isohexyl group and the like are preferable.
ハロゲン原子とは、 例えばフッ素原子、 塩素原子、 臭素原子又はヨウ素原子等 が挙げられ、 中でもフッ素原子、 塩素原子、 ヨウ素原子等が好ましく、 特にフッ 素原子、 塩素原子等がより好ましい。 Examples of the halogen atom include a fluorine atom, a chlorine atom, a bromine atom and an iodine atom, among which a fluorine atom, a chlorine atom and an iodine atom are preferred, and a fluorine atom and a chlorine atom are more preferred.
環状の飽和又は不飽和の C 3 - C 9脂肪族基とは、 炭素数 3ないし 9個よりなる 環状のアルキル基又はアルケニル基等が挙げられ、 中でも炭素数 3ないし 6個な る環状のアルキル基又はアルケニル基等が好ましく、 特に炭素数 3ないし 6個な る環状のアルキル基等がより好ましい。 The cyclic saturated or unsaturated C 3 -C 9 aliphatic group includes a cyclic alkyl group or alkenyl group having 3 to 9 carbon atoms, among which a cyclic alkyl group having 3 to 6 carbon atoms. Or an alkenyl group, and more preferably a cyclic alkyl group having 3 to 6 carbon atoms.
該環状のアルキル基とは、 例えばシクロプロピル基、 シクロブチル基、 シクロ ペンチル基、 シクロへキシル基、 シクロへプチル基、 シクロォクチル基、 シクロ ノニル基等が挙げられ、 中でもシクロプロピル基、 シクロプチル基、 シクロペン チル基、 シクロへキシル基等が好ましい。 Examples of the cyclic alkyl group include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclooctyl group, and a cyclononyl group. A tyl group and a cyclohexyl group are preferred.
該環状のアルケニル基とは、 例えばシクロプロぺニル基、 シクロブテニル基、 シクロペンテニル基、 シクロへキセニル基、 シクロへプテニルル基、 シグロォク テニル基、 シクロノネニル基等が挙げられ、 中でもシクロプロぺニル基、 シクロ ブテニル基、 シクロペンテニル基、 シクロへキセニル基等が好ましい。 Examples of the cyclic alkenyl group include a cyclopropenyl group, a cyclobutenyl group, a cyclopentenyl group, a cyclohexenyl group, a cycloheptenyl group, a cyclohexenyl group, and a cyclononenyl group. Among them, a cyclopropenyl group, a cyclobutenyl group and the like are exemplified. Group, cyclopentenyl group, cyclohexenyl group and the like.
ァラルキル基とは、 炭素数 7〜1 5個よりなるァラルキリレ基を意味し、 具体的 には例えばべンジル基、 ひ一メチルベンジル基、 フエネチル基、 3—フエニルプ 口ピル基、 1—ナフチルメチル基、 2—ナフチルメチル基、 α—メチル (1ーナ フチル) メチル基、 a—メチル ( 2—ナフチル) メチル基、 α—ェチル (1ーナ フチル) メチル基、 Q!—ェチル (2—ナフチル) メチル基、 ジブェ ルメチル基 等、 ジナフチルメチル基等が挙げられ、 中でもべンジル基、 1一ナフチルメチル 基、 2—ナフチルメチル基、 一メチルベンジル基、 フエネチル基等が好ましい The aralkyl group means an aralkylyl group having 7 to 15 carbon atoms, specifically, for example, a benzyl group, a monomethylbenzyl group, a phenethyl group, a 3-phenylpropyl group, a 1-naphthylmethyl group. , 2-naphthylmethyl group, α-methyl (1-naphthyl) methyl group, a-methyl (2-naphthyl) methyl group, α-ethyl (1naphthyl) methyl group, Q! -Ethyl (2-naphthyl) ) Methyl group, dibenzylmethyl group, etc., and dinaphthylmethyl group. Among them, benzyl group, 11-naphthylmethyl group Group, 2-naphthylmethyl group, monomethylbenzyl group, phenethyl group, etc.
N—ァラルキルァミノ基とは、 ァミノ基に上記のァラルキル基が置換した基を 意味し、 具体的には例えば N—ベンジルァミノ基、 N— (ひ一メチルベンジル) アミノ基、 N—フエネチルァミノ基、 N— ( 3—フエニルプロピル) アミノ基、 N— (1—ナフチルメチル) アミノ基、 N— (2—ナフチルメチル) アミノ基、 N— [ひ一メチル ( 1一ナフチル) メチル] アミノ基、 N— [ひ一メチル ( 2 - ナフチル) メチル] アミノ基、 N— [ひ一ェチル ( 1 _ナフチル) メチル] アミ ノ基、 N— [ —ェチル (2—ナフチル) メチル] アミノ基、 ジフエニルメチル アミノ基、 N— (ジナフチルメチル) アミノ基等が挙げられ、 中でも N—ベンジ ルァミノ基、 N - ( α—メチルベンジル) アミノ基、 Ν—フエネチルァミノ基 等が好ましい。 The N-aralkylamino group means a group in which the above-mentioned aralkyl group is substituted on the amino group, and specifically, for example, N-benzylamino group, N- (1-methylbenzyl) amino group, N-phenethylamino group, N- (3-Phenylpropyl) amino group, N— (1-naphthylmethyl) amino group, N— (2-naphthylmethyl) amino group, N— [Hi-methyl (1-naphthyl) methyl] amino group, N— [1-methyl (2-naphthyl) methyl] amino group, N— [1-ethyl (1-naphthyl) methyl] amino group, N — [— ethyl (2-naphthyl) methyl] amino group, diphenylmethyl amino group, Examples include an N- (dinaphthylmethyl) amino group, and among them, an N-benzylamino group, an N- (α-methylbenzyl) amino group, a Ν-phenethylamino group and the like are preferable.
Ν, Ν—ジーァラルキルアミノ基とは、 ァミノ基に上記のァラルキル基が二置 換した基を意味し、 具体的には例えば Ν, Ν—ジベンジルァミノ基、 Ν, Ν—ジ ( 一メチルベンジル) アミノ基、 Ν, Ν—ジフエネチルァミノ基、 Ν, Ν—ジ ( 3—フエニルプロピル) アミノ基、 Ν, Ν—ジ (1一ナフチルメチル) ァミノ 基、 Ν, Ν—ジ (2—ナフチルメチル) アミノ基、 Ν, Ν—ジ [ひ一メチル ( 1 一ナフチル) メチル] アミノ基、 Ν, Ν—ジ [ 一メチル ( 2—ナフチル) メチ ル] アミノ基、 Ν—ベンジルー Ν— (α—メチルベンジル) アミノ基、 Ν—ベン ジルー Ν—フエネチルァミノ基、 Ν—べンジルー Ν— ( 3—フエニルプロピル) アミノ基等が挙げられ、 中でも Ν, Ν—ジベンジルァミノ基、 Ν, Ν—ジ (《_ メチルベンジル) アミノ基、 Ν, Ν—ジフエネチルァミノ基等が好ましい。 ァラルキルォキシ基とは、 酸素原子に上記のァラルキル基が置換した基を意味 し、 具体的には例えばべンジルォキシ基、 α—メチルベンジルォキシ基、 フエネ チルォキシ基、 3—フエニルプロポキシ基、 1一ナフチルメトキシ基、 2—ナフ チルメトキシ基、 α—メチル (1一ナフチル) メトキシ基、 a—メチル (2—ナ フチル) メトキシ基、 α—ェチル (1—ナフチル) メトキシ基、 α—ェチル (2 一ナフチル) メトキシ基、 ジフエニルメトキシ基、 ジナフチルメトキシ基等が挙 げられ、 中でもべンジルォキシ基、 ひ _メチルベンジルォキシ基、 フエネチルォ キシ基等が好ましい。 The Ν, Ν-aralkylamino group means a group obtained by substituting the above aralkyl group for an amino group, and specifically, for example, Ν, Ν-dibenzylamino group, Ν, Ν-di (monomethyl) group. Benzyl) amino group, Ν, Ν-diphenethylamino group, Ν, Ν-di (3-phenylpropyl) amino group, Ν, Ν-di (1-naphthylmethyl) amino group, Ν, Ν-di (2-naphthylmethyl) amino group, Ν, Ν-di [1-methyl (1-naphthyl) methyl] amino group, Ν, ジ -di [monomethyl (2-naphthyl) methyl] amino group, Ν-benzyl- Ν— (α-methylbenzyl) amino group, Ν-benzyl-Ν-phenethylamino group, べ -benzyl-Ν— (3-phenylpropyl) amino group and the like, among which Ν, Ν-dibenzylamino group, Ν,ジ -di (<< _ methylbenzyl) amino group, Ν, Ν-diphen Chiruamino group, and the like are preferable. An aralkyloxy group means a group in which an oxygen atom is substituted by the above aralkyl group. Specifically, for example, a benzyloxy group, an α-methylbenzyloxy group, a phenethyloxy group, a 3-phenylpropoxy group, an Naphthylmethoxy group, 2-naphthylmethoxy group, α-methyl (1-naphthyl) methoxy group, a-methyl (2-naphthyl) methoxy group, α-ethyl (1-naphthyl) methoxy group, α-ethyl (21-naphthyl) methoxy group Naphthyl) methoxy group, diphenylmethoxy group, dinaphthylmethoxy group, etc., among which benzyloxy group, phenylmethylbenzyloxy group, phenethylo group Xyl groups and the like are preferred.
ァラルキルカルポニル基とは、 カルポニル基に上記のァラルキル基が置換した 基を意味し、 具体的には例えばべンジルカルポニル基、 ひ一メチルベンジルカル ポニル基、 フエネチルカルポニル基、 3—フエニルプロピルカルポニル基、 1一 ナフチルメチルカルポニル基、 2—ナフチルメチルカルポニル基、 一メチル ( 1一ナフチル) メチルカルポニル基、 α—メチル (2—ナフチル) メチルカルポ ニル基、 a—ェチル (1一ナフチル) メチルカルポニル基、 ひ一ェチル (2—ナ フチル) メチルカルポニル基、 ジフエ二ルメチルカルポニル基、 ジナフチルメチ ルカルポニル基等が挙げられ、 中でもべンジルカルポニル基、 ひ一メチルベンジ ルカルポニル基、 フエネチルカルポニル基等が好ましい。 The aralkylcarbonyl group means a group in which the aralkyl group is substituted on the carbonyl group, specifically, for example, a benzylcarbonyl group, a monomethylbenzylcarbonyl group, a phenethylcarbonyl group, Phenylpropyl carbonyl group, 1-naphthylmethyl carbonyl group, 2-naphthylmethyl carbonyl group, mono-methyl (1-naphthyl) methyl carbonyl group, α-methyl (2-naphthyl) methyl carbonyl group, a-ethyl (1-naphthyl) ) Methylcarbonyl group, methylethyl (2-naphthyl) methylcarbonyl group, diphenylmethylcarbonyl group, dinaphthylmethylcarbonyl group, etc., among which benzylcarbonyl group, 1-methylbenzylcarbonyl group, phenethylcarbonyl group And the like.
N―ァラルキル力ルバモイル基とは、 力ルバモイル基に上記のァラルキル基が 置換した基を意味し、 具体的には例えば N—ベンジルカルバモイル基、 N— ( 一メチルベンジル) 力ルバモイル基、 N—フエネチルカルバモイル基、 N— ( 3 一フエニルプロピル) 力ルバモイル基、 N— ( 1一ナフチルメチル) 力ルバモイ ル基、 N— ( 2—ナフチルメチル) 力ルバモイル基、 N - ( α—メチル (1ーナ フチル) メチル) 力ルバモイル基、 N— (ひ一メチル (2—ナフチル) メチル) 力ルバモイル基、 N— ( 一ェチル (1—ナフチル) メチル) 力ルバモイル基、 N - ( α—ェチル (2—ナフチル) メチル) 力ルバモイル基、 N— (ジフエニル メチル) 力ルバモイル基、 N— (ジナフチルメチル) 力ルバモイル基等が挙げら れ、 特に例えば N—ベンジルカルバモイル基、 N— (α—メチルベンジル) カル バモイル基、 Ν—フエネチルカルバモイル基等が好ましい。 The N-aralkyl molybamoyl group means a group obtained by substituting the above-mentioned aralkyl group with a molybmoyl group, and specifically includes, for example, N-benzylcarbamoyl group, N- (monomethylbenzyl) molybmoyl group, Netyl carbamoyl group, N— (3-phenylphenyl) dirubamoyl group, N— (1-naphthylmethyl) dirubamoyl group, N— (2-naphthylmethyl) dirubamoyl group, N- (α-methyl (1 Naphthyl) methyl) dirubamoyl group, N— (Hi-methyl (2-naphthyl) methyl) dirubamoyl group, N— (1-ethyl (1-naphthyl) methyl) dirubamoyl group, N- (α-ethyl) 2-naphthyl) methyl) rubamoyl group, N- (diphenylmethyl) rubamoyl group, N- (dinaphthylmethyl) rubamoyl group and the like. Yl group, N-(alpha-methylbenzyl) Cal Bamoiru group, Nyu- Hue phenethyl carbamoyl group and the like.
ァリール基としては、 炭素数 6ないし 1 5個よりなるァリール基を意味し、 例 えばナフチル基、 フエニル基等が挙げられ、 中でもフエニル基等が好ましい。 The aryl group means an aryl group having 6 to 15 carbon atoms, for example, a naphthyl group, a phenyl group and the like, among which a phenyl group and the like are preferable.
Ν—ァリールァミノ基とは、 ァミノ基に上記のァリ一ル基が置換した基を意味 し、 具体的には例えば Ν—フエニルァミノ基、 Ν— ( 1—ナフチル) アミノ基、 Ν— (2—ナフチル) アミノ基等が挙げられ、 中でも Ν—フエニルァミノ基等が 好ましい。 The term "arylamino" means a group obtained by substituting the above-mentioned aryl group with an amino group, specifically, for example, a phenylamino group, a Ν (1-naphthyl) amino group, an Ν— (2— Naphthyl) amino group and the like, among which a Ν-phenylamino group and the like are preferable.
Ν, Ν—ジァリールァミノ基とは、 ァミノ基に上記のァリール基が二置換した 基を意味し、 具体的には例えば Ν, Ν—ジフエニルァミノ基、 Ν, Ν—ジ (1 一 ナフチル) アミノ基、 N, N—ジ (2—ナフチル) アミノ基、 N—フエ二ルー N 一 (1一ナフチル) アミノ基、 N—フエニル— N— ( 2—ナフチル) アミノ基、 N— ( 1—ナフチル) 一 N— ( 2—ナフチル) アミノ基等が挙げられ、 中でも N , N—ジフエニルァミノ基、 N, N—ジ (1一ナフチル) アミノ基、 N, N—ジ ( 2—ナフチル) アミノ基等が好ましい。 The Ν, Ν-diarylamino group means a group obtained by disubstituting the above aryl group with an amino group. Specifically, for example, 例 え ば, Ν-diphenylamino group, Ν, Ν- Naphthyl) amino group, N, N-di (2-naphthyl) amino group, N-phenyl N- (1-naphthyl) amino group, N-phenyl-N— (2-naphthyl) amino group, N— ( 1-naphthyl) 1 N- (2-naphthyl) amino group, among which N, N-diphenylamino group, N, N-di (1-naphthyl) amino group, N, N-di (2-naphthyl) Amino groups and the like are preferred.
ァリールォキシ基とは、 酸素原子に上記のァリール基が置換した基を意味し、 具体的には例えばフエノキシ基、 ナフチルォキシ基等が挙げられ、 中でもフエノ キシ基等が好ましい。 The aryloxy group means a group in which an oxygen atom is substituted by the above aryl group, and specific examples include a phenoxy group and a naphthyloxy group. Among them, a phenoxy group and the like are preferable.
ァリ一ルスルホニル基とは、 スルホニル基に上記のァリ一ル基が置換した基を 意味し、 具体的には例えばフエニルスルホニル基、 ナフチルスルホニル基等が挙 げられ、 中でもフエニルスルホニル基等が好ましい。 The arylsulfonyl group means a group in which the above-mentioned aryl group is substituted on the sulfonyl group, specifically, for example, a phenylsulfonyl group, a naphthylsulfonyl group and the like. Groups and the like are preferred.
ァリ—ルスルホニルォキシ基とは、 スルホニルォキシ基に上記のァリ一ル基が 置換した基を意味し、 具体的には例えばフエニルスルホニルォキシ基、 ナフチル スルホニルォキシ基等が挙げられ、 中でもフエニルスルホニルォキシ基等が好ま しい。 The arylsulfonyloxy group means a group in which a sulfonyloxy group is substituted with the above-mentioned aryl group, and specifically, for example, phenylsulfonyloxy group, naphthylsulfonyloxy group and the like. Among them, a phenylsulfonyloxy group and the like are preferable.
N—ァリ一ルスルホニルァミノ基とは、 アミノ基に上記のァリ一ルスルホニル 基が N _置換した基を意味し、 具体的には例えば N—フエニルスルホニルァミノ 基、 N— ( 1 _ナフチルスルホニル) アミノ基、 N— ( 2—ナフチルスルホニル ) アミノ基等が挙げられ、 中でも N—フエニルスルホニルァミノ基、 N— ( 2 - ナフチルスルホニル) アミノ基等が好ましい。 The N-arylsulfonylamino group means a group in which the above-mentioned arylsulfonyl group is N_substituted for an amino group, specifically, for example, an N-phenylsulfonylamino group, (1_naphthylsulfonyl) amino group, N- (2-naphthylsulfonyl) amino group and the like are preferable, and among them, N-phenylsulfonylamino group and N- (2-naphthylsulfonyl) amino group are preferable.
N—ァリ一ルスルホニルァミノ d— C6アルキルアミノ基とは、 ァミノ基に上 記のァリ一ルスルホニルァミノ基を有する d— C6アルキル基が N—置換した 基を意味し、 具体的には例えば N—フエニルスルホニルアミノメチルァミノ基、 N - ( 1—フエニルスルホニルアミノエチル) アミノ基、 N— (2—フエニルス ルホニルアミノエチル) アミノ基、 N—ナフチルスルホニルアミノメチルアミノ 基、 N— ( 1—ナフチルスルホニルアミノエチル) アミノ基、 N— (2—ナフチ ルスルホニルアミノエチル) アミノ基等が挙げられ、 中でも N—フエニルスルホ ニルアミノメチルァミノ基、 N— (2—フエニルスルホニルアミノエチル)アミ ノ基等が好ましい。 N—ァリ一ルスルホニルァミノ d - C1 0アルキル力ルバモイル基とは、 カル バモイル基に上記のァリ一ルスルホニルァミノ基を有する C i一 C6アルキル基 が N—置換した基を意味し、 具体的には例えば N—フエニルスルホニルアミノメ チルカルバモイル基、 N— ( 1—フエニルスルホニルアミノエチル) 力ルバモイ ル基、 N— ( 2—フエニルスルホニルアミノエチル) 力ルバモイル基、 N—ナフ チルスルホニルアミノメチルカルバモイル基、 N _ ( 1—ナフチルスルホニルァ ミノェチル) 力ルバモイル基、 N— ( 2—ナフチルスルホニルアミノエチル) 力 ルバモイル基等が挙げられ、 中でも N—フエニルスルホニルァミノメチルカルバ モイル基、 N— (2—フエニルスルホニルアミノエチル) 力ルバモイル基、 N— ( 2—ナフチルスルホニルアミノエチル) 力ルバモイル基等が好ましい。 The N- § Li one Le sulfonyl § amino d-C 6 alkylamino group, d-C 6 alkyl groups having on the Symbol of § Li one Le sulfonyl § amino group to Amino group means a N- substituted group Specifically, for example, N-phenylsulfonylaminomethylamino group, N- (1-phenylsulfonylaminoethyl) amino group, N— (2-phenylsulfonylaminoethyl) amino group, N-naphthylsulfonylamino Methylamino group, N- (1-naphthylsulfonylaminoethyl) amino group, N- (2-naphthylsulfonylaminoethyl) amino group and the like, among which N-phenylsulfonylaminomethylamino group, N- (2 —Phenylsulfonylaminoethyl) amino group and the like are preferred. N- § Li one Le sulfonyl § amino d - A C 1 0 alkyl force Rubamoiru group, C i one C 6 alkyl group having the above-mentioned § Li one Le sulfonyl § amino group to cull Bamoiru group N- substituted group And specifically, for example, an N-phenylsulfonylaminomethylcarbamoyl group, an N- (1-phenylsulfonylaminoethyl) caproluvyl group, an N- (2-phenylsulfonylaminoethyl) caproluvyl group , N-naphthylsulfonylaminomethylcarbamoyl group, N_ (1-naphthylsulfonylamino) ethyl group, a rubamoyl group, N— (2-naphthylsulfonylaminoethyl) group, and a rubamoyl group Minomethylcarbamoyl group, N- (2-phenylsulfonylaminoethyl) carbamoyl group, N— (2-naphthylsulfonylaminoethyl group Le) force Rubamoiru group are preferable.
N—ァリールスルホニルァミノ d— C6アルコキシカルポニル基とは、 N—ァ リ一ルスルホニルァミノ C! - C 6アルコキシカルポニル基とは、 上記の N—ァリ 一ルスルホニルァミノ基が 一 C 6アルコキシカルポニル基に置換した基を意 味し、 具体的には例えば N—フエニルスルホニルアミノメトキシカルポニル基、 N—ナフチルスルホニルアミノメトキシカルポニル基、 1— ( N—フエニルスル ホニルァミノ) エトキシカルポニル基、 2— ( N—フエニルスルホニルァミノ) エトキシカルポニル基等が挙げられ、 中でも N—フエニルスルホニルアミノメト キシカルポニル基、 N—ナフチルスルホニルアミノメトキシカルポニル基等が好 ましい。 The N- § reel sulfonyl § amino d-C 6 alkoxy Cal Poni Le group, N- § Li one Le sulfonyl § Mino C! - The C 6 alkoxy Cal Poni group, a group of the above N- § Li one Le sulfonyl § amino group is substituted with one C 6 alkoxy Cal Poni Le group was meaning taste, specifically, for example N- phenylalanine sulfonylamino methoxy Cal Poni Le Group, N-naphthylsulfonylaminomethoxycarbonyl group, 1- (N-phenylsulfonylamino) ethoxycarbonyl group, 2- (N-phenylsulfonylamino) ethoxycarbonyl group, etc., among which N-phenylsulfonylaminomethoate Xycarbonyl group, N-naphthylsulfonylaminomethoxycarbonyl group and the like are preferable.
ァリールスルファモイル基とは、 スルファモイル基に上記のァリール基が置換 した基を意味し、 具体的には例えばフエニルスルファモイル基、 ナフチルスルフ ァモイル基等が挙げられ、 中でもフエニルスルファモイル基等が好ましい。 The arylsulfamoyl group means a sulfamoyl group substituted with the above aryl group, and specifically includes, for example, a phenylsulfamoyl group, a naphthylsulfamoyl group and the like, and among them, a phenylsulfamoyl group and the like. preferable.
ァリ一ルスルファモイルォキシ基とは、 スルファモイルォキシ基に上記のァリ ール基が置換した基を意味し、 具体的には例えばフエニルスルファモイルォキシ 基、 ナフチルスルファモイルォキシ基等が挙げられ、 中でもフエニルスルファモ ィルォキシ基等が好ましい。 The arylsulfamoyloxy group means a sulfamoyloxy group substituted with the above aryl group, specifically, for example, phenylsulfamoyloxy group, naphthylsulfayl group Moyloxy groups and the like can be mentioned, among which phenylsulfamoyloxy groups and the like are preferable.
ァリ一ルスルファモイル C 1一 C ]_ 0アルキル力ルバモイル基とは、 上記のァリ 一ルスルファモイルが d— C1 0アルキル力ルバモイル基に置換した基を意味し 、 具体的には例えばフエニルスルファモイルメチルカルバモイル基、 ナフチルス ルファモイルメチルカルバモイル基等が挙げられ、 中でもフエニルスルファモイ ルメチルカルバモイル基等が好ましい。 The arylsulfamoyl C 11 -C] _ 0 alkyl rubamoyl group means a group in which the above arylsulfamoyl is substituted with a d-C 10 alkyl rubamoyl group, and specifically, for example, phenylsulfamoyl Moylmethylcarbamoyl group, naphthyls And a phenylsulfamoylmethylcarbamoyl group.
ァリ一ルスルファモイル(^― C6アルコキシカルポニル基とは、 上記のァリ一 ルスルファモイルが d—C6アルコキシカルポニル基に置換した基を意味し、具 体的には例えばフエニルスルファモイルメトキシカルポニル基、 ナフチルスルフ ァモイルメトキシカルポニル基等が挙げられ、 中でもフエニルスルファモイルメ トキシカルポニル基等が好ましい。 § Li one Rusurufamoiru (^ - The C 6 alkoxy Cal Poni group, means a group above § Li one Rusurufamoiru has been substituted with d-C 6 alkoxy Cal Poni group, concrete in, for example Hue acylsulfamoyl methoxy Cal Poni Le group And a naphthylsulfamoylmethoxycarbonyl group, among which a phenylsulfamoylmethoxycarbonyl group and the like are preferable.
N—ァリ一ルカルバモイル基とは、 上記のァリール基が力ルバモイル基に N— 置換した基を意味し、 具体的には例えばフエ二ルカルバモイル基、 ナフチルカル バモイル基等が挙げられ、 中でもフエ二ルカルバモイル基等が好ましい。 The N-arylcarbamoyl group means a group in which the above aryl group is N-substituted with a carbamoyl group, and specifically includes, for example, a phenylcarbamoyl group, a naphthylcarbamoyl group and the like. And the like.
ァロイル基とは、 カルポニル基に上記のァリール基が置換した基を意味し、 具 体的には例えばベンゾィル基、 ナフチルカルボエル基等が挙げられ、 中でもベン ゾィル基等が好ましい。 An aroyl group means a group in which a carbonyl group is substituted with the above-mentioned aryl group, and specific examples thereof include a benzoyl group and a naphthylcarboel group, and among them, a benzoyl group and the like are preferable.
ァロキシ基とは、 上記のァロイル基が酸素原子に置換した基を意味し、 具体的 には例えばベンゾィルォキシ基、 ナフチルカルポニルォキシ基等が挙げられ、 中 でもべンゾィルォキシ基等が好ましい。 The alkoxy group means a group in which the above-mentioned aroyl group is substituted by an oxygen atom, and specifically includes, for example, a benzoyloxy group, a naphthylcarponyloxy group and the like, and among them, a benzoyloxy group and the like are preferable.
N—ァロイル基とは、 上記のァロイル基が窒素原子に置換した基を意味し、 具 体的には例えばべンゾィルァミノ基、 ナフチルカルポニルァミノ基等が挙げられ 、 中でもべンゾィルァミノ基等が好ましい。 The N-aroyl group means a group in which the above-mentioned aroyl group is substituted by a nitrogen atom, and specific examples include a benzoylamino group and a naphthylcarponylamino group, and among them, a benzoylamino group and the like are preferable.
N— (N—ァロイルァミノ) d _ C 1 0アルキル力ルバモイル基とは、 力ルバ モイル基に上記の N—ァロイルァミノ C 1一 C i 0アルキル基が置換した基を意味 し、 具体的には例えば N— (N—ベンゾィルアミノメチル) 力ルバモイル基、 N 一 (1一 (N—ベンゾィルァミノ) ェチル) 力ルバモイル基、 N— (2— (N - ベンゾィル) アミノエチル) 力ルバモイル基、 N— (N—ナフチルカルポニルァ ミノメチル) 力ルバモイル基、 N— ( 1— (N—ナフチルカルポニルァミノ) ェ チル) 力ルバモイル基、 N— ( 2— (N—ナフチルカルポニルァミノ) ェチル) 力ルバモイル基等が挙げられ、 中でも N— (N—ベンゾィルアミノメチル) カル パモイル基、 N - (2— (N—ベンゾィルァミノ) ェチル) 力ルバモイル基等が 好ましい。 N—ァロイルァミノ d— Ceアルコキシカルポニル基とは、 ォキシカルポニル 基に上記の N—ァロイルァミノ d— C6アルキル基が置換した基を意味し、 具体 的には例えば N—ベンゾィルアミノメトキシカルボ二ル基、 1一 (N—べンゾィ ルァミノ) エトキシカルポニル基、 2 _ (N—ベンゾィルァミノ) エトキシカル ポニル基、 N—ナフチルカルポニルアミノメトキシカルポニル基、 1一 (N—ナ フチルカルポニルァミノ) エトキシカルポニル基、 2— (N—ナフチルカルボ二 ルァミノ) エトキシカルボニル基等が挙げられ、 中でも N—ベンゾィルアミノメ トキシカルポニル基、 2 _ (N—ベンゾィルァミノ) エトキシカルポニル基等が 好ましい。 The N— (N-aroylamino) d _C 10 alkyl rubamoyl group means a group obtained by substituting a rubamoyl group with the above-mentioned N-aroylamino C 11 -Ci 0 alkyl group. N- (N-benzoylaminomethyl) rubamoyl group, N- (1- (N-benzoylamino) ethyl) rubamoyl group, N— (2- (N-benzoyl) aminoethyl) rubamoyl group, N— (N-naphthylcarbonylaminomethyl) rubamoyl group, N— (1- (N-naphthylcarbonylamino) ethyl) rubamoyl group, N— (2- (N-naphthylcarbonylamino) ethyl) rubamoyl group Among them, an N- (N-benzoylaminomethyl) carbamoyl group and an N- (2- (N-benzoylamino) ethyl) rubamoyl group are preferable. The N-aroylamino d-Ce alkoxycarbonyl group means a group in which the above-mentioned N-aroylamino d-C 6 alkyl group is substituted for an oxycarbonyl group, and specifically, for example, an N-benzoylaminomethoxycarbonyl group , 1- (N-benzoylamino) ethoxycarbonyl group, 2_ (N-benzoylamino) ethoxycarbonyl group, N-naphthylcarbonylaminomethoxycarbonyl group, 1- (N-naphthylcarbonylamino) ethoxycarbonyl group, 2- (N-naphthylcarbonylamino) ethoxycarbonyl group and the like, among which an N-benzoylaminomethoxycarbonyl group and a 2_ (N-benzoylamino) ethoxycarbonyl group are preferable.
C2— C6アルカノィル基とは、 カルポニル基に炭素数 1ないし 5個よりなるァ ルキル基が置換した基を意味し、 具体的には例えばァセチル基、 プロピオニル基 、 プチリル基、 イソプチリル基、 バレリル基、 イソバレリル基、 ピバロイル基、 ペンタノィル基等が挙げられ、 中でもァセチル基、 プロピオニル基、 ピバロィル 基等が好ましい。 The C 2 -C 6 alkanol group means a carbonyl group substituted with an alkyl group having 1 to 5 carbon atoms, and specifically includes, for example, an acetyl group, a propionyl group, a butyryl group, an isoptyryl group, and a valeryl group. Group, isovaleryl group, pivaloyl group, pentanoyl group and the like. Among them, acetyl group, propionyl group, pivaloyl group and the like are preferable.
C 2— C 6アル力ノィルォキシ基とは、水酸基に上記の C2 - C6アル力ノィル基 が置換した基を意味し、 具体的には例えばァセチルォキシ基、 プロピオ二ルォキ シ基、 プチリルォキシ基、 イソプチリルォキシ基、 バレリルォキシ基、 イソバ レリルォキシ基、 ピバロィルォキシ基、 ペンタノィルォキシ基等が挙げられ、 中 でもァセチルォキシ基、 プロピオニルォキシ基、 ビバロイルォキシ基等が好まし い。 The C 2 —C 6 alkenyl group refers to a group in which the above-mentioned C 2 -C 6 alkynyl group is substituted for a hydroxyl group, specifically, for example, an acetyloxy group, a propionyloxy group, a butyryloxy group, Examples include an isoptyryloxy group, a valeryloxy group, an isovaleryloxy group, a pivaloyloxy group, and a pentanoyloxy group. Of these, an acetyloxy group, a propionyloxy group, and a vivaloyloxy group are preferable.
N— C2— C6アルカノィルァミノ基とは、ァミノ基に上記の C2— C6アルカノ ィル基が置換した基を意味し、 具体的には例えば N—ァセチルァミノ基、 N—プ 口ピオニルァミノ基、 N—プチリルアミノ基、 N—イソプチリルアミノ基、 N 一バレリルアミノ基、 N—イソバレリルアミノ基、 N—ビバロイルァミノ基、 N—ペンタノィルァミノ基等が挙げられ、 中でも N—ァセチルァミノ基、 N—プ 口ピオニルァミノ基、 N—ピバロィルァミノ基等が好ましい。 The N—C 2 —C 6 alkanoylamino group refers to a group in which the above C 2 —C 6 alkanoyl group is substituted for an amino group, and specifically, for example, an N—acetylamino group, Mouth pionylamino, N-butyrylamino, N-isobutyrylamino, N-valerylamino, N-isovalerylamino, N-bivaloylamino, N-pentanoylamino, etc. Preferred are an acetylamino group, an N-piopenylamino group, an N-pivaloylamino group and the like.
N, N - ジ—C2— C6アルカノィルァミノ基とは、 ァミノ基に上記の C2— C 6アルカノィル基が二置換した基を意味し、 具体的には例えば N, N—ジァセチ ルァミノ基、 N, N—ジプロピオニルァミノ基、 N, N—ジブチリルァミノ基、 N, N—ジイソプチリルアミノ基、 N, N—ジバレリルアミノ基、 N, N—ジィ ソバレリルアミノ基、 N, N—ジピパロィルァミノ基、 N, N—ジペンタノィル アミノ基、 N—ァセチルー N—プロピオニルァミノ基、 N—ァセチルー N—プチ リルアミノ基、 N—ァセチルー N—ピパロィルァミノ基等が挙げられ、 中でも N , N—ジァセチルァミノ基、 N, N—ジプロピオニルァミノ基、 N, N—ジイソ プロピルアミノ基、 N, N—ジブチルァミノ基、 N, N—ジピバロィルァミノ基 等力好ましい。 N, N - The C 6 alkanoyloxy Noi Rua amino group, the Amino group of the C 2 - - di -C 2 C 6 Arukanoiru group means two substituted groups, and specific examples N, N-Jiasechi A lumino group, an N, N-dipropionylamino group, an N, N-dibutyrylamino group, N, N-diisobutyrylamino group, N, N-divalerylamino group, N, N-diisovalerylamino group, N, N-dipiparylylamino group, N, N-dipentanoylamino group, N-acetyl-N- A propionylamino group, an N-acetyl-N-butylylamino group, an N-acetyl-N-piparylylamino group and the like, among which N, N-diacetylamino, N, N-dipropionylamino, N, N-diisopropyl Amino group, N, N-dibutylamino group, N, N-dipivaloylamino group and the like are preferable.
N - Ci ~ C6アルキルアミノ基とは、 ァミノ基に炭素数 1ないし 6個よりなる アルキル基が置換した基を意味し、 具体的には例えば N—メチルァミノ基、 N— ェチルァミノ基、 N—プロピルアミノ基、 N—イソプロピルアミノ基、 N—プチ ルァミノ基、 N—イソプチルァミノ基、 N— s e c—プチルァミノ基、 N— t e r tーブチルァミノ基、 N—ペンチルァミノ基、 N—ネオペンチルァミノ基、 N —へキシルァミノ基、 N—イソへキシルァミノ基等が挙げられ、 中でも N—メチ ルァミノ基、 N—ェチルァミノ基、 N—プロピルアミノ基、 N—イソプロピルァ ミノ基、 N—プチルァミノ基、 N—イソプチルァミノ基、 N— t e r t—ブチル アミノ基等が好ましい。 The N-Ci-C 6 alkylamino group means a group in which an amino group is substituted with an alkyl group having 1 to 6 carbon atoms, specifically, for example, N-methylamino group, N-ethylamino group, N- To propylamino group, N-isopropylamino group, N-butylamino group, N-isobutylamino group, N-sec-butylamino group, N-tert-butylamino group, N-pentylamino group, N-neopentylamino group, N- Xylamino group, N-isohexylamino group and the like. Among them, N-methylamino group, N-ethylamino group, N-propylamino group, N-isopropylamino group, N-butylamino group, N-isobutylamino group, — Tert-butylamino group and the like are preferable.
N, N—ジー d— C6アルキルアミノ基とは、 ァミノ基に炭素数 1ないし 6 個よりなるアルキル基が二置換した基を意味し、 具体的には例えば N, N—ジメ チルァミノ基、 N, N—ジェチルァミノ基、 N, N—ジプロピルアミノ基、 N, N—ジイソプロピルアミノ基、 N, N—ジブチルァミノ基、 N, N—ジ t e r t 一プチルァミノ基、 N, N—ジペンチルァミノ基、 N, N—ジへキシルァミノ基 、 N—ェチル一 N—メチルァミノ基、 N—メチルー N—プロピルアミノ基、 N— イソプロピル— N—メチルァミノ基、 N— t e r t —プチルー N—メチルァミノ 基、 N—ェチル—N—イソプロピルアミノ基等が挙げられ、 中でも N, N—ジメ チルァミノ基、 N, N—ジェチルァミノ基、 N, N—ジイソプロピルアミノ基、 N, N—ジブチルァミノ基、 N, N—ジ t e r t—ブチルァミノ基、 N _ェチル 一 N—メチルァミノ基、 N—メチルー N—プロピルアミソ基、 N—イソプロピル 一 N—メチルァミノ基、 N—ェチルー N—イソプロピルアミノ基等が好ましい。 The N, N-di-d-C 6 alkylamino group means a diamino-substituted alkyl group having 1 to 6 carbon atoms, specifically, for example, N, N-dimethylamino group, N, N-diethylamino, N, N-dipropylamino, N, N-diisopropylamino, N, N-dibutylamino, N, N-ditert-butylamino, N, N-dipentylamino, N, N-dihexylamino group, N-ethyl-N-methylamino group, N-methyl-N-propylamino group, N-isopropyl-N-methylamino group, N-tert-butyl-N-methylamino group, N-ethyl N-isopropylamino group and the like, among which N, N-dimethylamino group, N, N-diethylamino group, N, N-diisopropylamino group, N, N-dibutylamino group, N, N-ditert-butylamino group The N_ethyl N-methylamino, N-methyl-N-propylamiso, N-isopropyl N-methylamino, N-ethyl-N-isopropylamino and the like are preferred.
N— Ci— C 1 0アルキル力ルバモイル基とは、 力ルバモイル基に炭素数 1ない し 1 0個よりなるアルキル基が置換した基を意味し、 具体的には例えば N—メチ ルカルバモイル基、 N—ェチルカルバモイル基、 N—プロピル力ルバモイル基、 N—イソプロピルカルパモイル基、 N—プチルカルバモイル基、 N—イソブチル 力ルバモイル基、 N— s e c—ブチルカルバモイル基、 N - t e r tーブチルカ ルパモイル基、 N—ペンチルカルバモイル基、 N—ネオペンチルカルバモイル基 、 N—へキシルカルバモイル基、 N—イソへキシルカルバモイル基、 N—ォクチ ルカルバモイル基、 N—デシルカルバオイル基等が挙げられ、 中でも N—メチル 力ルバモイル基、 N—ェチルカルバモイル基、 N—プロピル力ルバモイル基、 N —イソプロピル力ルバモイル基、 N—イソブチルカルバモイル基、 N— s e c— プチルカルバモイル基、 N— t e r t 一プチルカルバモイル基、 N—才クチルカ ルバモイル基、 N—デシルカルバモイル基等が好ましい。 N—Ci—C 10 alkyl rubamoyl group means that the rubamoyl group does not have 1 carbon atom And a group substituted by 10 alkyl groups, specifically, for example, N-methylcarbamoyl, N-ethylcarbamoyl, N-propylcarbamoyl, N-isopropylcarbamoyl, Butylcarbamoyl, N-isobutyl carbamoyl, N-sec-butylcarbamoyl, N-tert-butylcarbamoyl, N-pentylcarbamoyl, N-neopentylcarbamoyl, N-hexylcarbamoyl, N-iso A xylcarbamoyl group, an N-octylcarbamoyl group, an N-decylcarbamoyl group, and the like. Among them, an N-methyl rubamoyl group, an N-ethyl carbamoyl group, an N-propyl carbamoyl group, an N-isopropyl carbamoyl group, N-isobutylcarbamoyl group, N-sec-butyl carbamoyl group, N-tert-butyl Rubamoiru group, N- old Kuchiruka Rubamoiru group, N- dodecylcarbamoyl group and the like.
N, N—ジー C i一 C i oアルキル力ルバモイル基とは、 力ルバモイル基に炭素 数 1ないし 1 0個よりなるアルキル基が二置換した基を意味し、 具体的には例え ば N, N—ジメチルカルバモイル基、 N, N—ジェチリレカルバモイル基、 N, N —ジプロピル力ルバモイル基、 N, N—ジイソプロピル力ルバモイル基、 N, N 一ジブチルカルバモイル基、 N, N—ジ t e r t —ブチルカルバモイル基、 N, N—ジペンチルカルバモイル基、 N, N—ジへキシルカルバモイル基、 N—ェチ ルー N—メチルカルバモイル基、 N—イソプロピル— N—メチルカルバモイル基 、 N - t e r tーブチルー N—メチルカルバモイル基、 N—ェチルー N—イソプ 口ピル力ルバモイル基等が挙げられ、 中でも N, N—ジメチルカルバモイル基、 N, N—ジェチルカルバモイル基、 N, N—ジイソプロピル力ルバモイル基、 N , N—ジブチルカルバモイル基、 N, N—ジ t e r t —ブチルカルバモイル基、 N—ェチル一 N—メチルカルバモイル基、 N—イソプロピル— N—メチルカルバ モイル基、 N—ェチル—N—イソプロピル力ルバモイル基等が好ましい。 The N, N-di-C i-C ioalkyl rubamoyl group refers to a group obtained by disubstituting a carbamoyl group with an alkyl group having 1 to 10 carbon atoms. Specifically, for example, N, N —Dimethylcarbamoyl group, N, N-Getylilecarbamoyl group, N, N —Dipropyl rubamoyl group, N, N-Diisopropyl rubamoyl group, N, N-dibutylcarbamoyl group, N, N-ditert-butylcarbamoyl N, N-dipentylcarbamoyl, N, N-dihexylcarbamoyl, N-ethyl N-methylcarbamoyl, N-isopropyl-N-methylcarbamoyl, N-tert-butyl-N-methylcarbamoyl , N-ethyl-N-isopropyl pyrumomoyl group and the like, among which N, N-dimethylcarbamoyl group, N, N-getylcarbamoyl group, N, N-diisopropyl N-N-dibutylcarbamoyl, N, N-ditert-butylcarbamoyl, N-ethyl-N-methylcarbamoyl, N-isopropyl-N-methylcarbamoyl, N-ethyl-N— Isopropyl rubamoyl groups and the like are preferred.
N - C2 - C 6アルケニルカルバモイル基とは、 カルパモイル基に炭素数 2ない し 6個よりなるアルケニル基が置換した基を意味し、 具体的には例えば N—ビニ ルカルバモイル基、 N—ァリル力ルバモイル基、 N— (1 一プロべニル) カル バモイル基、 N—イソプロべ二ルカルバモイル基、 N— ( 2—ブテニル) カル パモイル基、 N—イソブテニルカルバモイル基、 N— (2—ペンテニル) カル バモイル基、 N— ( 2—へキセニル) カルパモイル基、 N— ( 2—ヘプテニル ) 力ルバモイル基、 N— (2—ォクテニル) 力ルバモイル基等が挙げられ、 中で も N—ビニルカルパモイル基、 N—ァリルカルパモイル基、 N— (1 一プロべ ニル) 力ルバモイル基等が好ましい。 N - C 2 - A C 6 alkenyl carbamoylmethyl group, having 2 to carbon Karupamoiru based means a group alkenyl group substituted consisting six, specifically for example, N- vinyl carbamoyl group, N- Ariru force Rubamoyl group, N- (1-probenyl) carbamoyl group, N-isoprobenylcarbamoyl group, N- (2-butenyl) carbamoyl group, N-isobutenylcarbamoyl group, N— (2-pentenyl) Cal Bamoyl group, N- (2-hexenyl) carpamoyl group, N- (2-heptenyl) -powered rubamoyl group, N- (2-octenyl) -powered rubamoyl group, etc., among which N-vinylcarbamoyl group, Preferred are an N-arylcarpamoyl group, an N- (1-proenyl) capilluvyl group and the like.
N, N—ジ— C2— C6アルケニルカルバモイル基とは、 力ルバモイル基に炭素 数 2ないし 6個よりなるアルケニル基が二置換した基を意味し、 具体的には例え ば N, N—ジピニルカルバモイル基、 N, N—ジァリル力ルバモイル基、 N, N—ジ (1—プロぺニル) 力ルバモイル基、 N, N—ジイソプロぺニルカルバモ ィル基、 N—ァリル一 N—ビニルカルバモイル基、 N—ァリル—N—イソブテ 二ルカルバモイル基、 N—ァリル—N— ( 2—ペンテニル) 力ルバモイル基、 N—ァリル— N— ( 2—へキセニル) 力ルバモイル基、 N—ァリルー N - ( 2 —ヘプテニル) 力ルバモイル基、 N—ァリル—N— (2—ォクテニル) カルパモ ィル基等が挙げられ、 中でも N—ァリル— N—ビニルカルバモイル基、 N, N —ジァリル力ルバモイル基、 N—ァリル— N— ( 1—プロぺニル)力ルバモイル 基等が好ましい。 The N, N-di-C 2 -C 6 alkenylcarbamoyl group refers to a disubstituted alkamoyl group having 2 to 6 carbon atoms in a carbamoyl group. Specifically, for example, N, N— Dipinylcarbamoyl group, N, N-diallyl rubamoyl group, N, N-di (1-propenyl) rubamoyl group, N, N-diisopropenyl carbamoyl group, N-aryl-1-N-vinylcarbamoyl group , N-aryl-N-isobutenyl carbamoyl group, N-aryl-N- (2-pentenyl) -powered rubamoyl group, N-aryl-N- (2-hexenyl) -powered rubamoyl group, N-aryl-N- (2 —Heptenyl) carbamoyl group, N-aryl-N— (2-octenyl) carbamoyl group, etc., among which N-aryl-N-vinylcarbamoyl group, N, N—diaryl lvamoyl group, N-aryl — N— (1—Pro Yl) force Rubamoiru group are preferable.
N—ァミノ Ci 一 C 1 0アルキル力ルバモイル基とは、 力ルバモイル基に炭素数 1ないし 1 0個よりなるアミノアルキル基が置換した基を意味し、 具体的には例 えば N—アミノメチルカルバモイル基、 N—ァミノェチルカルバモイル基、 N— ァミノプロピル力ルバモイル基、 N—アミノメチルェチルカルバモイル基、 N— アミノブチルカルバモイル基、 N—ァミノプロピル力ルバモイル基、 N—ァミノ ペンチルカルバモイル基、 N—ァミノへキシルカルバモイル基等が挙げられ、 中 でも N—アミノメチルカルバモイル基、 N—ァミノェチルカルバモイル基、 N— ァミノプロピル力ルバモイル基、 N _アミノメチルェチルカルバモイル基等が好 ましい。 N- The Amino Ci one C 1 0 alkyl force Rubamoiru group, 1 -C force Rubamoiru based means a group aminoalkyl group consisting of 1 0-substituted, specifically, example if N- amino-methylcarbamoyl N-aminoethylcarbamoyl group, N-aminopropylcarbamoyl group, N-aminomethylethylcarbamoyl group, N-aminobutylcarbamoyl group, N-aminopropylcarbamoyl group, N-aminopropylcarbamoyl group, N-aminopentylcarbamoyl group, N— Examples include an aminohexylcarbamoyl group, and among them, an N-aminomethylcarbamoyl group, an N-aminoethylcarbamoyl group, an N-aminopropylcarbamoyl group, and an N_aminomethylethylcarbamoyl group are preferable.
N— d— C6アルコキシ d— C 1 0アルキル力ルバモイル基とは、 上記の C i 一 C i 0アルキル力ルバモイル基に炭素数 1ないし 1 0個よりなるアルコキシ基 が N—置換した基を意味し、 具体的には例えば N—メトキシメチルカルバモイル 基、 N—メトキシェチルカルバモイル基、 N—メトキシプロピル力ルバモイル基 、 N—メトキシプチルカルバモイル基、 N—ェトキシペンチルカルバモイル基 、 N—ブトキシへキシルカルバモイル基等が挙げられ、 中でも N—メトキシメチ ルカルバモイル基、 N—メトキシェチルカルバモイル基、 N—メトキシプロピル カルパモイル基、 N—メトキシブチルカルバモイル基等が好ましい。 N—d—C 6 alkoxy d—C 10 alkyl rubamoyl group refers to a group in which an alkoxy group having 1 to 10 carbon atoms is N-substituted to the above-mentioned C 1 -C 1 0 alkyl rubamoyl group. And specifically, for example, N-methoxymethylcarbamoyl, N-methoxyethylcarbamoyl, N-methoxypropylcarbamoyl, N-methoxybutylcarbamoyl, N-ethoxypentylcarbamoyl And N-butoxyhexylcarbamoyl group, among which N-methoxymethylcarbamoyl group, N-methoxyethylcarbamoyl group, N-methoxypropylcarbamoyl group, N-methoxybutylcarbamoyl group and the like are preferable.
N - C i - Ceアルコキシカルポニル Ci _ C 1 0アルキル力ルバモイル基とは、 上記の d - Ci oアルキル力ルバモイル基に炭素数 1ないし 6個よりなるアルコ キシカルボニル基が N—置換した基を意味し、 具体的には例えば N—メトキシカ ルポ二ルメチルカルバモイル基、 N—メトキシカルポ二ルェチルカルバモイル基 、 N—メトキシカルボニルプロピル力ルバモイル基、 N—メトキシカルポニルブ チルカルバモイル基、 N—エトキシカルボニルペンチルカルバモイル基、 N—ブ トキシカルポニルへキシルカルバモイル基、 N - t e r t 一ブトキシカルボニル ェチルカルバモイル基等が挙げられ、 中でも N—メトキシカルポニルメチルカル バモイル基、 N—メトキシカルポ二ルェチルカルバモイル基、 N—メトキシカル ポニルプロピル力ルバモイル基、 N—メトキシカルポ二ルブチルカルバモイル基 、 N - t e r t 一ブトキシカルボ二ルェチルカルバモイル基等が好ましい。 N - a Ci o alkyl force Rubamoiru 1 -C based to an alkoxycarbonyl group composed of six N- substituted group - C i - Ce alkoxy Cal The Poni Le Ci _ C 1 0 alkyl force Rubamoiru group, the above d And specifically, for example, N-methoxycarbonylmethylcarbamoyl group, N-methoxycarbonylethylcarbamoyl group, N-methoxycarbonylpropyl carbamoyl group, N-methoxycarbonylbutylcarbamoyl group, N-ethoxycarbonyl A pentylcarbamoyl group, an N-butoxycarbonylhexylcarbamoyl group, an N-tert-butoxycarbonylethylcarbamoyl group, etc., among which an N-methoxycarbonylmethylcarbamoyl group, an N-methoxycarbonylethylcarbamoyl group, —Methoxycarbonylpropyl group rubamoyl group, N-meth A xycarbonylbutylcarbamoyl group, an N-tert-butoxycarbonylethylcarbamoyl group and the like are preferable.
N— C 1 一 C 6アルコキシカルボニルァミノ d — C i oアルキルカルパモイル基 とは、 C 一 C ]L 0アルキル力ルバモイル基に炭素数 1ないし 6個よりなるアルコ キシカルポニルァミノ基が N—置換した基を意味し、 具体的には例えば N—メト キシカルポニルアミノメチルカルバモイル基、 N—メトキシカルポニルアミノエ チルカルバモイル基、 N—メトキシカルポニルァミノプロピル力ルバモイル基、 N—メトキシカルボニルアミノブチルカルバモイル基、 N—エトキシカルボニル アミノぺンチルカルバモイル基、 N—ブ卜キシカルポニルァミノへキシルカルバ モイル基、 N - t e r tーブトキシカルポニルァミノェチルカルバモイル基等が 挙げられ、 中でも N—メトキシカルボニルアミノメチルカルバモイル基、 N—メ トキシカルボニルァミノェチルカルバモイル基、 N—メトキシカルポニルァミノ プロピル力ルバモイル基、 N—メトキシカルボニルアミノブチルカルバモイル基 、 N— t e r t―ブトキシカルボエルアミノエヂルカルバモイル基等が好ましい N—C 1 -C 6 alkoxycarbonylamino d — C ioalkylcarbamoyl group means a C 1 C] L 0 alkyl group having an alkoxycarbonylamino group having 1 to 6 carbon atoms in addition to an N-alkylcarbamoyl group. Means a substituted group, specifically, for example, N-methoxycarbonylaminomethylcarbamoyl group, N-methoxycarbonylaminoethylcarbamoyl group, N-methoxycarbonylaminopropyl carbamoyl group, N-methoxycarbonylaminobutylcarbamoyl Group, N-ethoxycarbonylaminopentylcarbamoyl group, N-butoxycarbonylaminohexylcarbamoyl group, N-tert-butoxycarbonylaminoethylcarbamoyl group, etc., among which N-methoxycarbonylaminomethylcarbamoyl Group, N-methoxycarbonylaminoethylcal Moil group, N- methoxy Cal Poni Rua amino propyl force Rubamoiru group, N- methoxycarbonylamino butylcarbamoyl group, N- tert-butoxy-carbonitrile El amino Edji carbamoyl group and the like
N - d - Ceアルコキシ力ルポニルァミノ C i - C6アルコキシ力ルポニル基 とは、 Ci一 C6アルコキシカルポニル基に炭素数 1ないし 6個よりなるアルコキ シカルポニルァミノ基が N—置換した基を意味し、 具体的には例えば N—メトキ シカルポニルアミノメトキシカルポニル基、 N—メトキシカルポニルァミノエト キシカルポニル基、 N—メトキシカルポニルァミノプロポキシカルポニル基、 N—メトキシカルポニルアミノブトキシカルボ二ル基、 N—エトキシカルポニル ァミノペンチルォキシカルポニル基、 N—ブトキシカルポニルァミノへキシルォ キシカルポニル基、 N - t e r t 一ブトキシカルボニルアミノエトキシカルポ二 ル基等が挙げられ、 中でも N—メトキシカルポニルアミノメトキシカルポニル基 、 N—メトキシカルポニルアミノエトキシカルポニル基、 N—メトキシカルポ二 ルァミノプロポキシカルポニル基、 N—メトキシカルポニルアミノブトキシカル ポニル基、 N— t e r t —ブトキシカルポニルアミノエトキシカルポニル基等が 好ましい。 N - d - Ce alkoxy force Ruponiruamino C i - The C 6 alkoxy force Ruponiru group, 1 -C on Ci one C 6 alkoxy Cal Poni Le group consisting of six alkoxy It means a N-substituted cycarponylamino group, specifically, for example, N-methoxycarbonylaminomethoxycarbonyl group, N-methoxycarbonylaminoethoxycarbonyl group, N-methoxycarbonylaminopropoxycarbonyl group N-methoxycarbonylaminobutoxycarbonyl group, N-ethoxycarbonylaminopentyloxycarbonyl group, N-butoxycarbonylaminohexyloxycarbonyl group, N-tert-butoxycarbonylaminoethoxycarbonyl group, etc. N-methoxycarbonylaminomethoxycarbonyl group, N-methoxycarbonylaminoethoxycarbonyl group, N-methoxycarbonylaminopropoxycarbonyl group, N-methoxycarbonylaminobutoxycarbonyl group, N-tert-butoxy Cal Poni Le aminoethoxy Cal Poni Le group and the like.
d - Ceアルキルチオ基とは、硫黄原子に炭素数 1ないし 6個よりなるアルキ ル基が置換した基を意味し、 具体的には例えばメチルチオ基、 ェチルチオ基、 プ ロピルチオ基、 イソプロピルチオ基、 プチルチオ基、 イソプチルチオ基、 s e c 一プチルチオ基、 t e r t—プチルチオ基、 ペンチルチオ基、 ネオペンチルチオ 基、 へキシルチオ基、 イソへキシルチオ基等が挙げられ、 中でもメチルチオ基、 ェチルチオ基、 プロピルチオ基、 イソプロピルチオ基、 プチルチオ基、 t e r t -プチルチオ基等が好ましい。 The d-Cealkylthio group means a group in which a sulfur atom is substituted with an alkyl group having 1 to 6 carbon atoms, and specifically, for example, a methylthio group, an ethylthio group, a propylthio group, an isopropylthio group, a butylthio group Group, isobutylthio group, sec monobutylthio group, tert-butylthio group, pentylthio group, neopentylthio group, hexylthio group, isohexylthio group, etc., among which methylthio group, ethylthio group, propylthio group, isopropylthio group, A butylthio group, a tert-butylthio group and the like are preferred.
N - d - Ceアルキルスルファモイル基とは、 スルファモイル基に炭素数 1な いし 6個よりなるアルキル基が置換した基を意味し、 具体的には例えば N—メチ ルスルファモイル基、 N—ェチルスルファモイル基、 N _プロピルスルファモイ ル基、 N _イソプロピルスルファモイル基、 N—プチルスルファモイル基、 N— イソプチルスルファモイル基、 N - s e c 一プチルスルファモイル基、 N— t e r tーブチルスルファモイル基、 N—ペンチルスルファモイル基、 N—ネオペン チルスルファモイル基、 N—へキシルスルファモイル基、 N—イソへキシルスル ファモイル基等が挙げられ、 中でも N—メチルスルファモイル基、 N -ェチルス ルファモイル基、 N—イソプロピルスルファモイル基、 N— t e r t—ブチルス ルファモイル基等が好ましい。 The N-d-Ce alkylsulfamoyl group means a group in which a sulfamoyl group is substituted by an alkyl group having 1 or 6 carbon atoms, specifically, for example, an N-methylsulfamoyl group, an N-ethyls group. Rufamoyl group, N_propylsulfamoyl group, N_isopropylsulfamoyl group, N-butylsulfamoyl group, N-isobutylsulfamoyl group, N-sec monobutylsulfamoyl group, N— tert-butylsulfamoyl group, N-pentylsulfamoyl group, N-neopentylsulfamoyl group, N-hexylsulfamoyl group, N-isohexylsulfamoyl group, and the like. Famoyl, N-ethylsulfamoyl, N-isopropylsulfamoyl, N-tert-butylsulfamoyl and the like are preferred.
N, N—ジー d—C6アルキルスルファモイル基とは、 スルファモイル基に 炭素数 1ないし 6個よりなるアルキル基が二置換した基を意味し、 具体的には例 えば N, N—ジメチルスルファモイル基、 N, N—ジェチルスルファモイル基、 N, N—ジプロピルスルファモイル基、 N, N—ジイソプロピルスルファモイル 基、 N, N—ジブチルスルファモイル基、 N, N—ジ t e r t—ブチルスルファ モイル基、 N, N -ジぺンチルスルファモイル基、 N, N—ジへキシルスルファ モイル基、 N—ェチルー N—メチルスルファモイル基、 N—イソプロピル一 N— メチルスルファモイル基、 N - t e r t 一ブチル—N—メチルスルファモイル基 、 N—ェチルー N—イソプロピルスルファモイル基等が挙げられ、 中でも N, N 一ジメチルスルファモイル基、 N, N—ジェチルスルファモイル基、 N, N—ジ イソプロピルスルファモイル基、 N, N—ジブチルスルファモイル基、 N, N— ジ t e r t—ブチルスルファモイル基、 N—ェチル— N—メチルスルファモイル 基、 N—イソプロピル— N—メチルスルファモイル基、 N—ェチル— N— ^ Γソプ 口ピルスルファモイル基等が好ましい。 N, N-Gee d—C 6 alkylsulfamoyl group means It means a group in which an alkyl group having 1 to 6 carbon atoms is disubstituted. For example, N, N-dimethylsulfamoyl, N, N-getylsulfamoyl, N, N-dipro Pyrsulfamoyl group, N, N-diisopropylsulfamoyl group, N, N-dibutylsulfamoyl group, N, N-ditert-butylsulfamoyl group, N, N-dimethylsulfamoyl group, N, N-dihexylsulfamoyl, N-ethyl-N-methylsulfamoyl, N-isopropyl-N-methylsulfamoyl, N-tert-butyl-N-methylsulfamoyl, N-ethyl-N- Isopropylsulfamoyl group; among them, N, N-dimethylsulfamoyl group, N, N-diethylsulfamoyl group, N, N-diisopropylsulfamoyl group, N, N-dibutylsulfamoyl group Group, N, N-ditert-butylsulfamoyl group, N-ethyl-N-methylsulfamoyl group, N-isopropyl-N-methylsulfamoyl group, N-ethyl-N- ^ Rufamoyl and the like are preferred.
Ci一 C6アルキルスルフィエル基とは、 スルフィニル基に炭素数 1ないし 6個 よりなるアルキル基が置換した基を意味し、 具体的には例えばメチルスルフィ二 ル基、 ェチルスルフィエル基、 プロピルスルフィニル基、 イソプロピルスルフィ ニル基、 ブチルスルフィニル基、 イソブチルスルフィニル基、 s e cーブチルス ルフィニル基、 t e r tーブチルスルフィニル基、 ペンチルスルフィニル基、 ネ ォペンチルスルフィニル基、 へキシルスルフィニル基、 イソへキシルスルフィ二 ル基等が挙げられ、 中でもメチルスルフィニル基、 ェチルスルフィニル基、 プロ ピルスルフィニル基、 イソプロピルスルフィニル基、 プチルスルフィニル基、 t e r t 一プチルスルフィニル基等が好ましい。 The Ci one C 6 alkyl sulfide El group, 1 -C sulfinyl alkyl group composed of six is meant substituted group, specifically, for example Mechirusurufi two group, E chill sulfide El, propyl sulfinyl Group, isopropylsulfinyl group, butylsulfinyl group, isobutylsulfinyl group, sec-butylsulfinyl group, tert-butylsulfinyl group, pentylsulfinyl group, neopentylsulfinyl group, hexylsulfinyl group, isohexylsulfinyl group, etc. Among them, a methylsulfinyl group, an ethylsulfinyl group, a propylsulfinyl group, an isopropylsulfinyl group, a butylsulfinyl group, a tert-butylsulfinyl group and the like are preferable.
d—C6アルキルスルホニル基とは、 スルホニル基に炭素数 1ないし 6個より なるアルキル基が置換した基を意味し、 具体的には例えばメチルスルホニル基、 ェチルスルホニル基、 プロピルスルホニル基、 イソプロピルスルホニル基、 ブチ ルスルホニル基、 イソプチルスルホニル基、 s e c—プチルスルホニル基、 t e r t 一プチルスルホニル基、 ペンチルスルホニル基、 ネオペンチルスルホニル基 、 へキシルスルホニル基、 イソへキシルスルホニル基等が挙げられ、 中でもメチ ルスルホニル基、 ェチルスルホニル基、 プロピルスルホニル基、 プチルスルホニ ル基、 t e r t —プチルスルホニル基等が好ましい。 d—C 6 alkylsulfonyl group means a group in which a sulfonyl group is substituted by an alkyl group having 1 to 6 carbon atoms, specifically, for example, methylsulfonyl group, ethylsulfonyl group, propylsulfonyl group, isopropyl A sulfonyl group, a butylsulfonyl group, an isobutylsulfonyl group, a sec-butylsulfonyl group, a tert-butylsulfonyl group, a pentylsulfonyl group, a neopentylsulfonyl group, a hexylsulfonyl group, and an isohexylsulfonyl group. Methylsulfonyl, ethylsulfonyl, propylsulfonyl, butylsulfonyl And a tert-butylsulfonyl group and the like.
C i - Ceアルコキシ基とは、酸素原子に炭素数 1ないし 6個よりなるアルキル 基が置換した基を意味し、 具体的には例えばメトキシ基、 エトキシ基、 プロポキ シ基、 イソプロポキシ基、 ブトキシ基、 イソブトキシ基、 s e c —ブトキシ基、 t e r t—ブトキシ基、 ペンチルォキシ基、 ネオペンチルォキシ基、 へキシルォ キシ基、 イソへキシルォキシ基等が挙げられ、 中でもメトキシ基、 エトキシ基、 プロポキシ基、 イソプロポキシ基、 ブトキシ基、 イソブトキシ基、 t e r t—ブ トキシ基等が好ましい。 The C i -Ce alkoxy group means a group in which an oxygen atom is substituted by an alkyl group having 1 to 6 carbon atoms, and specifically, for example, a methoxy group, an ethoxy group, a propoxy group, an isopropoxy group, a butoxy group Group, isobutoxy group, sec-butoxy group, tert-butoxy group, pentyloxy group, neopentyloxy group, hexoxy group, isohexyloxy group, etc., among which methoxy group, ethoxy group, propoxy group, isopropoxy group Group, butoxy group, isobutoxy group, tert-butoxy group and the like are preferable.
d— C6アルコキシカルポニル基とは、 カルポニル基に炭素数 1ないし 5個よ りなるアルコキシ基が置換した基を意味し、 具体的には例えばメトキシカルボ二 ル基、 エトキシカルポニル基、 プロポキシカルボ二ル基、 イソプロポキシ力ルポ ニル基、 ブトキシカルポニル基、 イソブトキシカルボ二ル基、 s e c—ブトキシ カルポニル基、 t e r t 一ブトキシカルポニル基、 ペンチルォキシカルポニル基 、 ネオペンチルォキシカルポニル基等が挙げられ、 中でもメトキシカルポニル基 、 エトキシカルボニル基、 プロポキシカルポニル基、 イソプロポキシカルポニル 基、 ブトキシカルボ二ル基、 t e r t—ブトキシカルポニル基等が好ましい。 アミノ d— C6アルコキシカルポニル基とは、 カルボニル基に炭素数 1ないし 6個よりなるアミノアルコキシ基が置換した基を意味し、 具体的には例えばアミ ノメトキシカルポニル基、 ァミノエトキシカルポニル基、 ァミノプロポキシカル ポニル基、 2—ァミノ— 2—メチルプロポキシカルポニル基、 2ーァミノメチル エトキシカルボニル基、 アミノブトキシカルポニル基、 2—ァミノプロポキシ力 ルポニル基、ァミノペンチルォキシカルボ二ル基、 ァミノへキシルォキシカルボ ニル基等が挙げられ、 中でもアミノメトキシカルボニル基、 アミノエトキシカル ポニル基、ァミノプロポキシカルポニル基、 2—アミノメチルエトキシカルボ二 ル基、 2—アミノー 2—メチルプロポキシ力ルポニル基等が好ましい。 The d—C 6 alkoxycarbonyl group means a group in which a carbonyl group is substituted with an alkoxy group having 1 to 5 carbon atoms, specifically, for example, a methoxycarbonyl group, an ethoxycarbonyl group, a propoxycarbonyl group. Group, isopropoxyl propyl group, butoxycarbonyl group, isobutoxycarbonyl group, sec-butoxycarbonyl group, tert-butoxycarbonyl group, pentyloxycarbonyl group, neopentyloxycarbonyl group, and the like. Among them, a methoxycarbonyl group, an ethoxycarbonyl group, a propoxycarbonyl group, an isopropoxycarbonyl group, a butoxycarbonyl group, a tert-butoxycarbonyl group and the like are preferable. The amino d-C 6 alkoxycarbonyl group means a group in which a carbonyl group is substituted with an aminoalkoxy group having 1 to 6 carbon atoms, specifically, for example, an aminomethoxycarbonyl group, an aminoethoxycarbonyl group, Aminopropoxy carbonyl, 2-amino-2-methylpropoxy carbonyl, 2-aminomethyl ethoxycarbonyl, aminobutoxy carbonyl, 2-amino propoxy Luponyl, aminopentyloxy carbonyl, amino Xyloxycarbonyl group and the like, among which aminomethoxycarbonyl group, aminoethoxycarbonyl group, aminopropoxycarbonyl group, 2-aminomethylethoxycarbonyl group, 2-amino-2-methylpropoxycarbonyl group, etc. Is preferred.
N— C3— ( 6シクロアルキルアミノ基とは、 ァミノ基に炭素数 3ないし 6個な る環状のアルキル基が N—置換した基を意味し、 具体的には例えば N—シクロプ 口ピルアミノ基、 N—シクロブチルァミノ基、 N—シクロペンチルァミノ基、 N—シクロへキシルァミノ基等が挙げられ、 中でも N—シクロプロピルァミノ基 、 N—シクロペンチルァミノ基、 N—シクロへキシルァミノ基等が好ましレ^ N, N—ジー C3—じ6シクロアルキルアミノ基とは、 ァミノ基に炭素数 3な いし 6個なる環状のアルキル基が二置換した基を意味し、 具体的には例えば N, N—ジシクロプロピルアミノ基、 N, N—ジシクロブチルァミノ基、 N, N—ジ シクロペンチルァミノ基、 N, N—ジシクロへキシルァミノ基、 N—シクロプチ ル— N—シクロプロピルアミノ基、 N—シクロペンチル— N—シクロプロピルァ ミノ基、 N—シクロへキシルー N—シクロプロピルアミノ基等が挙げられ、 中で も N, N—ジシクロプロピルアミノ基、 N, N—ジシクロブチルァミノ基、 N, N—ジシクロペンチルァミノ基等が好ましい。 N—C 3 — ( 6- cycloalkylamino group refers to a group in which a cyclic alkyl group having 3 to 6 carbon atoms is N-substituted on an amino group, and specifically, for example, an N-cyclopropylpyramino group , N-cyclobutylamino group, N-cyclopentylamino group, N-cyclohexylamino group and the like, among which N-cyclopropylamino group , N- cyclopentyl Rua amino group, N- cycloheteroalkyl Kishiruamino group and the Shi-liked ^ N, N- di C 3 - and Ji 6 cycloalkylamino group, an annular in Amino group consisting six number 3 of stone carbon It means a group in which an alkyl group is disubstituted, and specifically, for example, N, N-dicyclopropylamino group, N, N-dicyclobutylamino group, N, N-dicyclopentylamino group, N, N -Dicyclohexylamino group, N-cyclobutyl-N-cyclopropylamino group, N-cyclopentyl-N-cyclopropylamino group, N-cyclohexyl N-cyclopropylamino group and the like. , N-dicyclopropylamino, N, N-dicyclobutylamino, N, N-dicyclopentylamino and the like.
じ3 _ (:6シクロアルキルォキシ基とは、酸素原子に炭素数 3ないし 6個なる環 状のアルキル基が置換した基を意味し、例えば N—シクロプロポキシ基、 N—シ クロブトキシ基、 N—シクロペンチルォキシ基、 N—シクロへキシルォキシ基 等が挙げられ、 中でも N—シクロプロポキシ基、 N—シクロペンチルォキシ基、 N—シクロへキシルォキシ基等が好ましい。 Flip 3 _ (6 Cycloalkyl O alkoxy group, C 3 -C oxygen atom 6 becomes ring-shaped alkyl group means a group obtained by substituting, for example, N- cyclopropoxy group, N- sheet Kurobutokishi group, Examples thereof include an N-cyclopentyloxy group and an N-cyclohexyloxy group, among which an N-cyclopropoxy group, an N-cyclopentyloxy group, an N-cyclohexyloxy group and the like are preferable.
N - C3 - C6シクロアルキル力ルバモイル基とは、 力ルバモイル基に炭素数 3 ないし 6個なる環状のアルキル基が置換した基を意味し、 例えば N—シクロプロ ピル力ルバモイル基、 N—シクロプチルカルバモイル基、 N—シクロペンチル 力ルバモイル基、 N—シクロへキシルカルバモイル基等が挙げられ、中でも N— シクロプロピル力ルバモイル基、 N—シクロペンチルカルバモイル基、 N—シ クロへキシルカルバモイル基等が好ましい。 N - C 3 - The C 6 cycloalkyl force Rubamoiru group, 3 -C force Rubamoiru based means 6 comprising a cyclic group in which the alkyl group is substituted, such as N- Shikuropuro pills force Rubamoiru group, N- cyclo Examples include a butylcarbamoyl group, an N-cyclopentylcarbamoyl group, an N-cyclohexylcarbamoyl group, and among them, an N-cyclopropylcarbamoyl group, an N-cyclopentylcarbamoyl group, and an N-cyclohexylcarbamoyl group are preferable.
N, N -ジ— C3 - C6シクロアルキル力ルバモイル基とは、 力ルバモイル基 に炭素数 3ないし 6個なる環状のアルキル基が二置換した基を意味し、 具体的に は例えば N, N—ジシクロプロピル力ルバモイル基、 N, N—ジシクロプチルカ ルバモイル基、 N, N—ジシクロペンチルカルバモイル基、 N, N—ジシクロへ キシルカルバモイル基、 N—シクロプチルー N—シクロプロピル力ルバモイル基 、 N—シクロペンチルー N—シクロプロピル力ルバモイル基、 N—シクロへキシ ルー N—シクロプロピル力ルバモイル基等が挙げられ、 中でも N, N—ジシクロ プロピル力ルバモイル基、 N, N—ジシクロブチルカルバモイル基、 N, N—ジ シクロペンチルカルバモイル基等が好ましい。 5又は 6員の複素環基とは、 例えばイソォキサゾリル基、 イソチアゾリル基、 イミダゾリル基、 ォキサゾリル基、 ォキサジァゾリル基、 チアゾリル基、 チアジ ァゾリル基、 チェニル基、 トリアジニル基、 トリァゾリル基、 ピリジル基、 ピラ ジル基、 ピラジニル基、 ピリミジニル基、 ピリダジニル基、 ピラゾリル基、 ピロ リル基、 ビラ二ル基、 フリル基、 フラザニル基、 イミダゾリジニル基、 イミダゾ リニル基、 テトラヒドロフラニル基、 ピラゾリジニル基、 ピラゾリニル基、 ピぺ ラジェル基、 ピペリジニル基、 ピロリジニル基、 ピロリニル基、 モルホリノ基等 が挙げられ、 中でもイソォキサゾリル基、 イソチアゾリル基、 イミダゾリル基、 ォキサゾリル基、 チアゾリル基、 チェニル基、 ピリジル基、 ピラジル基、 ピラジ ニル基、 ピリミジニル基、 ピリダジニル基、 ピラゾリル基、 ピロリル基、 ピラニ ル基、 フリル基、 テトラヒドロフラニル基、 モルホリノ基等が好ましい。 The N, N-di-C 3 -C 6 cycloalkyl forcerubamoyl group means a group in which a forcerubamoyl group is disubstituted with a cyclic alkyl group having 3 to 6 carbon atoms. N-dicyclopropyl rubamoyl group, N, N-dicyclobutyl carbamoyl group, N, N-dicyclopentylcarbamoyl group, N, N-dicyclohexylcarbamoyl group, N-cyclobutyl-N-cyclopropyl rubamoyl group, N-cyclopentyl -N-cyclopropyl rubamoyl group, N-cyclohexyl N-cyclopropyl rubamoyl group, etc., among which N, N-dicyclopropyl rubamoyl group, N, N-dicyclobutylcarbamoyl group, N, N —Dicyclopentylcarbamoyl group and the like are preferable. The 5- or 6-membered heterocyclic group includes, for example, an isoxazolyl group, an isothiazolyl group, an imidazolyl group, an oxazolyl group, an oxaziazolyl group, a thiazolyl group, a thiaziazolyl group, a chenyl group, a triazinyl group, a triazolyl group, a pyridyl group, a pyrazyl group, Pyrazinyl group, pyrimidinyl group, pyridazinyl group, pyrazolyl group, pyrrolyl group, virazyl group, furyl group, furazanyl group, imidazolidinyl group, imidazolinyl group, tetrahydrofuranyl group, pyrazolidinyl group, pyrazolinyl group, piperidinyl group, piperidinyl group Group, pyrrolidinyl group, pyrrolinyl group, morpholino group and the like. Among them, isooxazolyl group, isothiazolyl group, imidazolyl group, oxazolyl group, thiazolyl group, chenyl group, pyridyl group, pyrazyl group Piraji group, pyrimidinyl group, pyridazinyl group, pyrazolyl group, pyrrolyl group, pyranyl group, furyl group, tetrahydrofuranyl group, morpholino group and the like are preferable.
窒素原子、 酸素原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当 たり 1ないし 5個有する 1ないし 3環性の複素芳香環基とは、 例えばァクリジニ ル基、 イソキノリル基、 イソインドリル基、 インダゾリル基、 インドリル基、 ィ ンドリジニル基、 エチレンジォキシフエニル基、 カルバゾリル基、 キナゾリニル 基、 キノキサリニル基、 キノリジニル基、 キノリル基、 クマロニル基、 クロメニ ル基、 フエナンスリジニル基、 フエナンスロリニル基、 ジベンゾフラニル基、 ジ ベンゾチオフェニル基、 シンノリニル基、 チォナフテニル基、 ナフチリジニル基 、 フエナジニル基、 フエナキサジニル基、 フエノチアジニル基、 フタラジニル基 、 プテリジニル基、 プリニル基、 ベンゾイミダゾリル基、 ベンゾォキサゾリル基 、 ベンゾチアゾリル基、 ベンゾトリアゾリル基、 ベンゾフラニル基、 メチレンジ ォキシフエニル基等が挙げられ、 中でもエチレンジォキシフエニル基、 ジべンゾ フラニル基、 ジベンゾチオフェニル基、 メチレンジォキシフエニル基等が好まし い。 A mono- to tri-cyclic heteroaromatic group having 1 to 5 heteroatoms per ring system selected from the group consisting of nitrogen, oxygen and sulfur is, for example, an acridinyl group, an isoquinolyl group, an isoindolyl group , Indazolyl group, indolyl group, indolizinyl group, ethylenedioxyphenyl group, carbazolyl group, quinazolinyl group, quinoxalinyl group, quinolizinyl group, quinolyl group, coumalonyl group, chromenyl group, fenanslidinyl group, fenansloryl Nyl group, dibenzofuranyl group, dibenzothiophenyl group, cinnolinyl group, thonaphthenyl group, naphthyridinyl group, phenazinyl group, phenaxazinyl group, phenothiazinyl group, phthalazinyl group, pteridinyl group, purinyl group, benzoimidazolyl group, benzoxazolyl group Le Benzothiazolyl group, benzotriazolyl group, benzofuranyl group, methylenedioxyphenyl group, etc., among which ethylenedioxyphenyl group, dibenzofuranyl group, dibenzothiophenyl group, methylenedioxyphenyl group, etc. Is preferred.
直鎖若しくは分枝鎖の飽和又は不飽和の d— C9脂肪族基とは、 炭素数 1ない し 9個よりなる直鎖状又は分枝状のアルキル基、 アルケニル基又はアルキニル基 が挙げられ、 中でも炭素数 1ないし 6個なる直鎖状又は分枝状のアルキル基、 ァ ルケニル基又はアルキニル基が好ましい。 The linear or branched, saturated or unsaturated d-C 9 aliphatic groups, straight-chain or branched alkyl group 1 -C consisting nine, alkenyl or include alkynyl groups Among them, a linear or branched alkyl group having 1 to 6 carbon atoms, an alkenyl group or an alkynyl group is preferred.
該アルキル基とは、 例えばメチル基、 ェチル基、 プロピル基、 イソプロピル基 、 ブチル基、 イソブチル基、 s e c—ブチル基、 t e r t—プチル基、 ペンチル 基、 ネオペンチル基、 へキシル基、 イソへキシル基、 ヘプチル基、 ォクチル基、 ノニル基等が挙げられ、 中でもメチル基、 ェチル基、 プロピル基、 イソプロピル 基、 イソブチル基、 s e c一プチル基、 t e r t—ブチル基等が好ましい。 該ァルケニル基とは、 例えばビニル基、 ァリル基、 1一プロぺニル基、 イソプ 口ぺニル基、 2—ブテニル基、 イソブテニル基、 2—ペンテニル基、 2—へキセ ニル基、 2—ヘプテニル基、 2—ォクテニル基等が挙げられ、 中でもビニル基、 ァリル基、 1—プロぺニル基等が好ましい。 The alkyl group is, for example, a methyl group, an ethyl group, a propyl group, an isopropyl group. Butyl group, isobutyl group, sec-butyl group, tert-butyl group, pentyl group, neopentyl group, hexyl group, isohexyl group, heptyl group, octyl group, nonyl group, etc., among which methyl group, ethyl group Group, propyl group, isopropyl group, isobutyl group, sec-butyl group, tert-butyl group and the like. The alkenyl group includes, for example, vinyl group, aryl group, 1-propenyl group, isopropenyl group, 2-butenyl group, isobutenyl group, 2-pentenyl group, 2-hexenyl group, 2-heptenyl group And 2-octenyl group, among which vinyl group, aryl group, 1-propenyl group and the like are preferable.
該アルキニル基とは、 例えばェチニル基、 1一プロピエル基、 1ーブテニル基 、 1一ペンテニル基、 1—へキシニル基、 1一へプチニル基、 1—ォクチ二ル基 等が挙げられ、 中でもェチェル基、 1—プロピニル基等が好ましい。 The alkynyl group includes, for example, ethynyl group, 1-propenyl group, 1-butenyl group, 11-pentenyl group, 1-hexynyl group, 11-heptynyl group, 1-octynyl group and the like. And a 1-propynyl group.
N - C i - C j oアルキル力ルバモイル基とは、 力ルバモイル基に炭素数 1ない し 1 0個よりなるアルキル基が置換した基を意味し、 具体的には例えば N—メチ ルカルバモイル基、 N—ェチルカルバモイル基、 N _プロピル力ルバモイル基、 N—イソプロピルカリレバモイル基、 N—プチルカルバモイル基、 N _イソブチル 力ルバモイル基、 N— s e c—ブチルカルバモイル基、 N - t e r t一プチルカ ルバモイル基、 N -ペンチルカルバモイル基、 N—ネオペンチルカルバモイル基 、 N―へキシルカルバモイル基、 N—イソへキシルカルバモイル基、 N—才クチ ルカルバモイル基、 N—デシルカルバオイル基等が挙げられ、 中でも N—メチル 力ルバモイル基、 N -ェチルカルバモイル基、 N—プロピル力ルバモイル基、 N 一イソプロピル力ルバモイル基、 N—イソブチルカルバモイル基、 N— s e c— プチルカルバモイル基、 N - t e r t一プチルカルバモイル基、 N—ォクチルカ ルバモイル基、 N—デシルカルバモイル基等が好ましい。 The N-Ci-Cjo alkyl rubamoyl group means a carbamoyl group substituted with an alkyl group having 1 to 10 carbon atoms, specifically, for example, an N-methylcarbamoyl group, —Ethylcarbamoyl group, N _propylcarbamoyl group, N-isopropylcalilevamoyl group, N-butylcarbamoyl group, N_isobutylcarbamoyl group, N-sec-butylcarbamoyl group, N-tert-butylcarbamoyl group, N-pentylcarbamoyl group, N-neopentylcarbamoyl group, N-hexylcarbamoyl group, N-isohexylcarbamoyl group, N-butylcarbamoyl group, N-decylcarbamoyl group, etc., among which N-methyl L-bamoyl group, N-ethylcarbamoyl group, N-propyl-lubamoyl group, N-isopropyl l-rubamoyl group, N-iso A butylcarbamoyl group, N-sec-butylcarbamoyl group, N-tert-butylcarbamoyl group, N-octylcarbamoyl group, N-decylcarbamoyl group and the like are preferable.
N - C「 C J 0アルキルチオ力ルバモイル基とは、 チォカルバモイル基に炭素 数 1ないし 1 0個よりなるアルキル基が N—置換した基を意味し、 具体的には例 えば N—メチルチオ力ルバモイル基、 N—ェチルチオ力ルバモイル基、 N—プロ ピルチオ力ルバモイル基、 N—イソプロピルチオ力ルバモイル基、 N—プチルチ 才力ルバモイル基、 N Tソブチルチオ力ルバモイル基、 N— s e c—プチルチ ォカルバモイル基、 N— t e r t —プチルチオカルパモイル基、 N—ペンチルチ ォカルバモイル基、 N—ネオペンチルチオ力ルバモイル基、 N—へキシルチオ力 ルバモイル基、 N—イソへキシルチオ力ルバモイル基、 N—才クチルチオ力ルバ モイル基、 N—デシルチオ力ルバモイル基等が挙げられ、 中でも N—メチルチオ 力ルバモイル基、 N—ェチルチオ力ルバモイル基、 N—プロピルチオ力ルバモイ ル基、 N—イソプロピルチオ力ルバモイル基、 N—イソプチルチオ力ルバモイル 基、 N— s e c—プチルチオ力ルバモイル基、 N— t e r t—プチルチオカルパ モイル基、 N—才クチルチオ力ルバモイル基、 N—デシルチオ力ルバモイル基等 が好ましい。 N—C “CJ 0 alkylthioalkavamoyl group means a group in which a thiocarbamoyl group is N-substituted with an alkyl group having 1 to 10 carbon atoms, and specifically, for example, an N-methylthioalkavamoyl group N-ethylthiolrubamoyl group, N-propylthiolrubamoyl group, N-isopropylthiolrubamoyl group, N-butyltylylrubamoyl group, NTsobutylthiolrubamoyl group, N-sec-butylbutylcarbamoyl group, N-tert —Butylthiocarpamoyl group, N—Pentylchi Carbamoyl group, N-neopentylthiolrubamoyl group, N-hexylthiolrubamoyl group, N-isohexylthiolrubamoyl group, N-octylthiolrubamoyl group, N-decylthiolrubamoyl group, and the like. Among them, N-methylthiol-rubamoyl, N-ethylthiol-rubamoyl, N-propylthio-lumbamoyl, N-isopropylthio-lumbamoyl, N-isobutylthiol-luvamoyl, N-sec-butylthiol-luvamoyl, N-tert -A butylthiocarbamoyl group, an N-octylthiolrubamoyl group, an N-decylthiolrubamoyl group and the like are preferable.
1ないし 3環性の C7— C 1 5炭素芳香環基とは、 上記のァリール基を除く、 例 えばァセナフチレニル基、 ァダマンチル基、 アンスリル基、 インデニル基、 ノル ポルニル基、 フエナンスリル基等が挙げられ、 中でもアンスリル基、 フエナンス リル基等が好ましい。 The monocyclic to tricyclic C 7 -C 15 carbon aromatic ring group excludes the above aryl group, and includes, for example, an acenaphthylenyl group, an adamantyl group, an anthryl group, an indenyl group, a norpolnyl group, a phenanthryl group and the like. Among them, an anthryl group and a phenanthryl group are preferred.
窒素原子、 酸素原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当 たり 1ないし 5個有する 1ないし 3環性の縮合複素芳香環基 (但し、 5若しくは '6員の複素環基を除く。 ) とは、 例えばァクリジニル基、 イソキノリル基、 イソ インドリル基、 インダゾリル基、 インドリル基、 インドリジニル基、 エチレンジ ォキシフエニル基、 カルバゾリル基、 キナゾリニル基、 キノキサリニル基、 キノ リル基、 クマロニル基、 イソクロマニル基、 クロメニル基、 フエナンスリジニル 基、 フエナンスロリジル基、 ジベンゾフラニル基、 ジベンゾチオフェニル基、 チ ォナフテニル基、 ナフチリジニル基、 フエナジニル基、 フエナキサジニル基、 フ エノチアジニル基、 フ夕ラジニル基、 プテリジニル基、 プリニル基、 ベンゾイミ ダゾリル基、 ベンゾォキサゾリル基、 ベンゾチアゾリル基、 ベンゾト,リアゾリル 基、 ベンゾフラエル基、 ジヒドロべンゾフラニル基、 メチレンジォキシフエニル 基等が挙げられ、 中でもエチレンジォキシフエニル基、 ジベンゾフラニル基、 ジ ベンゾチオフェニル基、 メチレンジォキシフエニル基等が好ましい。 A mono- to tricyclic fused heteroaromatic group having 1 to 5 heteroatoms per ring system selected from the group consisting of nitrogen, oxygen and sulfur atoms (however, a 5- or 6-membered heterocyclic group ) Means, for example, an acridinyl group, an isoquinolyl group, an isoindolyl group, an indazolyl group, an indolyl group, an indolizinyl group, an ethylenedioxyphenyl group, a carbazolyl group, a quinazolinyl group, a quinoxalinyl group, a quinolyl group, a cumaronyl group, an isochromanyl group; Chromenyl group, phenanthridinyl group, phenanthrolidyl group, dibenzofuranyl group, dibenzothiophenyl group, thionaphthenyl group, naphthyridinyl group, fenadinyl group, phenaxazinyl group, phenothiazinyl group, furazinyl group, pteridinyl group, Purinyl group, benzimi Dazolyl group, benzoxazolyl group, benzothiazolyl group, benzoto, liazolyl group, benzofurayl group, dihydrobenzozofuranyl group, methylenedioxyphenyl group, etc., among which ethylenedioxyphenyl group, dibenzofuranyl Group, dibenzothiophenyl group, methylenedioxyphenyl group and the like.
R 1及び R 2は、 同一又は異なって、 例えば水素原子、 ー アルキル基を示 し、 中でも水素原子が好適である。 R 1 and R 2 are the same or different and each represent, for example, a hydrogen atom or a -alkyl group, and among them, a hydrogen atom is preferable.
R 3は、 例えば水素原子、 ^— ^ァルキル基 (伹し、 n—ブチル基は除く) を示し、 中でも C 2— C 6アルキル基 (但し、 n—ブチル基は除く) が好適であり 、 特に例えば C 3— C 5の分岐したアルキル基が好適である。 R 3 represents, for example, a hydrogen atom or a ^-^ alkyl group (excluding n-butyl group), and among them, a C 2 -C 6 alkyl group (excluding n-butyl group) is preferable. Particularly, for example, a C 3 -C 5 branched alkyl group is suitable.
R4は、 例えば水素原子、 水酸基、 一 C3アルキル基を示し、 中でも水素原 子、 — C3アルキル基が好適であり、 特に例えば水素原子が好適である。 R 4 represents, for example, a hydrogen atom, a hydroxyl group or a 1 C 3 alkyl group, among which a hydrogen atom and a —C 3 alkyl group are preferable, and a hydrogen atom is particularly preferable.
R5は、 例えば水素原子、 アジド基、 アミノ基、 力ルバモイル基、 力ルバモイ ルァミノ基、 力ルバモイルォキシ基、 力ルポキシル基、 シァノ基、 スルファモイ ル基、 スルホ基、 ニトロ基、 ハロゲン原子、 ヒドロキシ基、 ホルミル基、 ホルミ ルァミノ基、 環状の飽和若しくは不飽和の C3— C9脂肪族基、 ァラルキル基、 N ーァラルキルアミノ基、 N, N—ジァラルキルアミノ基、 ァラルキルォキシ基、 ァラルキルカルポニル基、 N―ァラルキル力ルバモイル基、 ァリール基、 N—ァ リールアミノ基、 N, N—ジァリールアミノ基、 ァリールォキシ基、 ァリ一ルス ルホニル基、 ァリールスルホニルォキシ基、 N—ァリールスルホニルァミノ基、 N—ァリ一ルスルホニルァミノ C C 6アルキルァミノ基、 N—ァリ—ルスルホ エルアミノ 一 C10アルキル力ルバモイル基、 N—ァリ—ルスルホニルァミノ Ci— C6アルコキシカルポニル基、 ァリ一ルスルファモイル基、 ァリ一ルスルフ ァモイルォキシ基、 ァリールスルファモイル。 一 。アルキル力ルバモイル基 、 ァリ一ルスルファモイル。 i一 C 6アルコキシカルボニル基、 N—ァリールカル バモイル基、 ァロイル基、 ァロキシ基、 N—ァロイル基、 N— ( N—ァロイルァ ミノ) C^C.oアルキル力ルバモイル基、 N—ァロイルァミノ(:丄一 C6アルコ キシカルポニル基、 C2— C 6アルカノィル基、 C2— C6アルカノィルォキシ基、 N— C2_C6アルカノィルァミノ基、 N, N—ジ— C2_C6アルカノィルアミ ノ基、 N—Ci— C6アルキルアミノ基、 N, N—ジー Ci— C6アルキルアミノ基 、 N— — C1Qアルキル力ルバモイル基、 N, N—ジ— (^ーじ^ァルキルカ ルバモイル基、 N— C2— C6アルケニルカルバモイソレ基、 N, N—ジー C2— C6 アルケニルカルバモイル基、 N_アミ 一 C10アルキル力ルバモイル基、 N - ,- C 6アルコキシ C! - C i。アルキル力ルバモイル基、 N— ^ - C6アルコ キシカルポ二ル 一 C1()アルキル力ルバモイル基、 N— 一 C6アルコキシ力 ルポニルァミノ C i— C i Qアルキル力ルバモイル基、 N _ C i— C 6アルコキシ力 ルポニルァミノ d— C 6アルコキシカルボ二ル基、 Ci— Ceアルキルチオ基、 N 一 一 C6アルキルスルファモイル基、 N, N—ジー C — Ceアルキルスルファ モイル基、 C — C6アルキルスルフィニル基、 Ci— C6アルキルスルホニル基、 Ct— C6アルコキシ基、 アルコキシカルポニル基、 アミノ 一(:6ァ ルコキシカルポニル基、 N— C3— C6シクロアルキルアミノ基、 N, N—ジ—C 3_C6シクロアルキルアミノ基、 C3—〇6シクロアルキルォキシ基、 N— C3 一 C 6シクロアルキル力ルバモイル基、 N, N—ジー C3— ( 6シクロアルキル力 ルバモイル基からなる群から選択される置換基、 5又は 6員の複素環基、 窒素原 子、 酸素原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1な いし 5個有する 1ないし 3環性の複素芳香環基並びに該置換基で置換されていて もよい、 直鎖若しくは分枝鎖の飽和若しくは不飽和の Cエー C9脂肪族基、 (:丄一 C6アルコキシ基、 C — C6ァルキルチオ基、 N— Ci— Ceアルキルアミノ基、 N-Cx-C6アルキル力ルバモイル基、 N— C i— C 6アルキルチオ力ルバモイル 基からなる群より選ばれる置換基を有してしてもよい炭素芳香環基、 1ないし 3 環性の C 7— C ]L 5炭素芳香環基又は 5若しくは 6員の複素環基若しくは窒素原子 、 酸素原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1ない し 5個有する 1ないし 3環性の複素芳香環基 (5又は 6員の複素環基を除く。 ) を示し、 中でも炭素芳香環基が、 フエニル基、 ナフチル基であり、 置換基を有し ていてもよい炭素芳香環基の置換基が、 水素原子、 アジド基、 ァ.ミノ基、 力ルバ モイル基、 力ルバモイルァミノ基、 力ルバモイルォキシ基、 力ルポキシル基、 シ ァノ基、 ニトロ基、 ハロゲン原子、 ヒドロキシ基、 環状の飽和若しくは不飽和の C3— C9脂肪族基、 ァラルキル基、 N—ァラルキルアミノ基、 ァラルキルォキシ 基、 ァラルキルカルポニル基、 N—ァラルキル力ルバモイル基、 ァリール基、 N 一ァリ一ルァミノ基、 ァリールォキシ基、 ァリールスルホニル基、 ァリールスル ホニルォキシ基、 N—ァリ一ルスルホニルァミノ C i— C 6アルコキシカルポニル 基、 ァリールスルファモイル基、 ァリ一ルスルファモイルォキシ基、 ァリールス ルファモイル Cェ _ C i。アルキル力ルバモイル基、 ァリールスルファモイル〇1 _C 6アルコキシカルボ二ル基、 N—ァリ一ルカルバモイル基、 ァロイル基、 ァ ロキシ基、 N—ァロイル基、 C2_C6アルカノィル基、 C2— C6アルカノィルォ キシ基、 N— C2_C6アルカノィルァミノ基、 N, N—ジー C2— C6アルカノ ィルァミノ基、 N— 一 C6アルキルアミノ基、 N— 一 C10アルキル力ルバ モイル基、 N, N—ジ— C i— C 1 0アルキル力ルバモイル基、 N _ C 2— C 6アル ケニルカルバモイル基、 N, N—ジ一 C 2— C 6アルケニルカルバモイル基、 N— アミノじ 一 C 1 0アルキル力ルバモイル基、 N—C — C 6アルコキシ 一 C 1 0 アルキル力ルバモイル基、 N— C「 C 6アルコキシカルポニル C!一 C 1 Qアルキ ルカルバモイル基、 C^— C 6アルキルスルフィニル基、 じェ一 C 6アルコキシ基、 C 1― C 6アルコキシカルポニル基、 ァミノ C「 C 6アルコキシカルポニル基、 N— C 3—(:6シクロアルキルアミノ基、 (:3—〇6シクロアルキルォキシ基、 N 一 C 3— C 6シクロアルキル力ルバモイル基、 イミダゾリジニル基、 イミダゾリニ ル基、 テトラヒドロフラニレ基、 ピラゾリジニル基、 ピラゾリニル基、 ピペラジ ニル基、 ピペリジニル基、 ピロリジニル基、 ピロリニル基、 モルホリノ基、 イソ ォキサゾリル基、 イソチアゾリル基、 イミダゾリル基、 ォキサゾリル基、 ォキサ ジァゾリル基、 チアゾリル基、 チアジアゾリル基、 チェニル基、 トリァゾリル基 、 ピリジル基、 ピラジニル基、 ピリミジニル基、 ピリダジニル基、 ピラゾリル基 、 ピロリル基、 ピラニル基、 フリル基、 ァクリジニル基、 イソキノリル基、 イソ インドリル基、 インダゾリル基、 インドリル基、 エチレンジォキシフエニル基、 カルバゾリル基、 キナゾリニル基、 キノキサリニル基、 キノリル基、 クロマニル 基、 クロメニル基、 フエナンスリジニル基、 フエナンスロリニル基、 ジベンゾフ ラニル基、 ジベンゾチオフェニル基、 シンノリニル基、 チォナフテニル基、 ナフ チリジニル基、 フエナジニル基、 フエナキサジニル基、 ベンゾイミダゾリル基、 ベンゾォキサゾリル基、 ベンゾチアゾリル基、 ベンゾトリアゾリル基、 ベンゾフ ラニル基、 ジヒドロべンゾフラニル基、 メチレンジォキシフエニル基であり、 1 ないし 3環性の C 7— C 1 5炭素芳香環基が、 ァセナフチレニル基、 ァダマンチル 基、 アンスリル基、 インデニル基、 フエナンスリル基であり、 5若しくは 6員の 複素環基が、 イソォキサゾリル基、 イソチアゾリル基、 イミダゾリル基、 ォキサ ゾリル基、 チアゾリル基、 チアジアゾリル基、 チェニル基、 トリァゾリル基、 ピ リジル基、 ピラジニル基、 ピリミジェル基、 ピリダジニル基、 ピラゾリル基、 ピ 口リル基、 ピラニル基、 フリル基であり、 窒素原子、 酸素原子及び硫黄原子から なる群より選ばれる複素原子を 1環系当たり 1ないし 5個有する 1ないし 3環性 の複素芳香環基 (5又は 6員の複素環基を除く。 ) が、 ァクリジニル基、 イソキ ノリル基、 イソインドリル基、 インダゾリル基、 インドリル基、 インドリジニル 基、 エチレンジォキシフエニル基、 カルバゾリル基、 キナゾリニル基、 キノキサ リニル基、 キノリル基、 クロマニル基、 イソクロマニル基、 クロメニル基、 フエ ナンスリジニル基、 フエナンスロリジル基、 ジベンゾフラニル基、 ジベンゾチォ フエニル基、 チォナフテニル基、 ナフチジニル基、 フエナジニル基、 フエナキサ ジニル基、 フエノチアジニル基、 フタラジェル基、 プテリジニル基、 プリニル基 、 ベンゾイミダゾリル基、 ベンゾォキサゾリル基、 ベンゾチアゾリル基、 ベンゾ トリァゾリル基、 ベンゾフラエル基、 ジヒドロベンゾフラニル基若しくはメチレ ンジォキシフエニル基が好適であり、 特に炭素芳香環基が、 フエニル基、 ナフチ ル基であり、 置換基を有していてもよい炭素芳香環基の置換基が、 水素原子、 ァ ジド基、 アミノ基、 力ルバモイル基、 力ルポキシル基、 シァノ基、 ニトロ基、 ハ ロゲン原子、 ヒドロキシ基、 ァラルキル基、 N—ァラルキルアミノ基、 ァラルキ ルォキシ基、 ァラルキルカルポニル基、 N—ァラルキル力ルバモイル基、 ァリ一 ル基、 ァリールォキシ基、 ァリールスルホニル基、 ァリ一ルスルホニルォキシ基 、 ァリールスルファモイル基、 ァロイル基、 ァロキシ基、 N—ァロイル基、 C2 一 C6アルカノィル基、 C 2— C 6アルカノィルォキシ基、 N— C2— C6アルカノ ィルァミノ基、 N-Cj-Ceアルキルァミノ基、 N— 一 C10アルキル力ルバ モイル基、 N, N—ジ— C1-C1。アルキル力ルバモイル基、 N— C2— C6アル ケニルカルバモイル基、 N, N—ジー C 2— C 6アルケニルカルバモイル基、 N— アミノ C 一 C J 0アルキル力ルバモイル基、 N— C — Ceアルコキシ (:ュ— C10 アルキル力ルバモイル基、 N-Cx- C6アルコキシカルポニル C !-010アルキ ルカルバモイル基、 C — C 6アルキルスルフィニル基、 C — C6アルコキシ基、 C 1一 C 6アルコキシカルボ二ル基、 アミノ C i— C 6アルコキシカルポニル基、 N— C3—( 6シクロアルキルアミノ基、 (:3_( 6シクロアルキルォキシ基、 ィ ソォキサゾリル基、 イソチアゾリル基、 ォキサゾリル基、 チアゾリル基、 チェ二 ル基、 ピリジル基、 ピラジニル基、 ピリミジェル基、 ピリダジニル基、 フリル基 、 テトラヒドロフラニル基、 モルホリノ基、 イソキノリル基、 イソインドリル基 、 エチレンジォキシフエニル基、 キナゾリニル基、 キノキサリニル基、 キノリル 基、 ジベンゾフラエル基、 ジベンゾチオフェニル基、 ベンゾイミダゾリル基、 ベ ンゾォキサゾリル基、 ベンゾチアゾリル基、 ベンゾフラニル基、 メチレンジォキ シフエエル基であり、 1ないし 3環性の C 7— C 1 5炭素芳香環基が、 ァセナフチ レニル基、 インデニル基、 フエナンスリル基であり、 5若しくは 6員の複素環基 が、 イソォキサゾリル基、 イソチアゾリル基、 ォキサゾリル基、 チアゾリル基、 チェニル基、 ピリジル基、 ビラジニル基、 ピリミジェル基、 ピリダジニル基、 フ リル基であり、 窒素原子、 酸素原子及び硫黄原子からなる群より選ばれる複素原 子を 1環系当たり 1ないし 5個有する 1ないし 3環性の複素芳香環基 ( 5又は 6 員の複素環基を除く。 ) が、 イソキノリル基、 イソインドリル基、 エチレンジォ キシフエニル基、 キナゾリニル基、 キノキサリニル基、 ジベンゾフラニル基、 ジ ベンゾチオフェニル基、 フエナキサジニル基、 ベンゾイミダゾリル基、 ベンゾォ キサゾリル基、 ベンゾチアゾリル基、 ベンゾフラニル基、 ジヒドロベンゾフラ二 ル基若しくはメチレンジォキシフエニル基がより好適である。 R 5 is, for example, a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, a carbamoyloxy group, a propyloxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, Formyl group, formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkyl Kilcarponyl group, N-aralkyl rubamoyl group, aryl group, N-aryl amino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfonyloxy group, N-arylsulfonyl Amino group, N-arylsulfonylamino CC 6 alkylamino group, N-arylsulfonylamino C 1 0 alkyl force Rubamoiru group, N- § Li - Le sulfonyl § amino CI- C 6 alkoxy Cal Poni group, § Li one Rusurufamoiru group, § Li one Rusurufu Amoiruokishi group, § reel sulfamoyl. One. Alkylsulfamoyl group, arylsulfamoyl. i one C 6 alkoxycarbonyl group, N- Arirukaru Bamoiru group, Aroiru group, Arokishi group, N- Aroiru group, N- (N-Aroirua Mino) C ^ Co alkyl force Rubamoiru group, N- Aroiruamino (:丄one C 6 Alkoxycarbonyl group, C 2 —C 6 alkanoyl group, C 2 —C 6 alkanoyloxy group, N—C 2 _C 6 alkanoylamino group, N, N—di—C 2 _C 6 alkanoylamino group, N—Ci—C 6 alkylamino group, N, N—diCi—C 6 alkylamino group, N——C 1Q alkyl rubamoyl group, N, N—di — (^-^^ alkircalcarbamoyl group, N— C 2 - C 6 alkenyl carbamoylthiopheno Sole group, N, N-di-C 2 - C 6 alkenyl carbamoylmethyl group, N_ Ami one C 10 alkyl force Rubamoiru group, N -, - C 6 alkoxy C -!. C i alkyl L-bamoyl group, N — ^-C 6 alkoxycarbonyl C 1 () alkyl rubamoyl group, N—1 C 6 alkoxy ruponylamino Ci—C i Q alkyl rubamoyl group, N _C i—C 6 alkoxy rupponylamino d—C 6 alkoxy carbonyl, Ci— Ce alkylthio group, N eleven C 6 alkylsulfamoyl group, N, N-di-C - Ce alkylsulfamoyl Moil group, C - C 6 alkylsulfinyl group, CI- C 6 alkylsulfonyl group, Ct-C 6 alkoxy group, an alkoxycarbonyl Cal Poni group, an amino one (6 § Le Koki deer Lupo sulfonyl group, N-C 3 - C 6 cycloalkyl amino group, N, N-di--C 3 _C 6 cycloalkylamino group, C 3 -〇 6 cycloalkyl O alkoxy group, N-C 3 one C 6 cycloalkyl force Rubamoiru group, N, N-di-C 3 - ( 6 Cycloalkyl group) A substituent selected from the group consisting of a rubamoyl group, a 5- or 6-membered heterocyclic group, a heteroatom selected from the group consisting of a nitrogen atom, an oxygen atom, and a sulfur atom is contained in one ring system. A linear or branched, saturated or unsaturated C A C 9 aliphatic group which may be substituted with a mono- to tricyclic heteroaromatic ring group having 5 C 6 alkoxy group, C—C 6 alkylthio group, A carbon aromatic ring group which may have a substituent selected from the group consisting of N-Ci-Ce alkylamino group, NC x -C 6 alkyl rubamoyl group, and N-Ci-C 6 alkylthio rubamoyl group , 1 to 3 cyclic C 7 — C] L 5 carbon aromatic ring groups or 5 or 6 membered heterocyclic groups or a heteroatom selected from the group consisting of nitrogen, oxygen and sulfur atoms per ring system A 5- to 6-membered heteroaromatic group (excluding a 5- or 6-membered heterocyclic group) having 5 to 5 carbon atoms, and among them, a carbon-aromatic ring group is a phenyl group or a naphthyl group; The substituent of the carbon-aromatic ring group which may be substituted is a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, a carbamoyloxy group, a propyloxyl group, a cyano group, a nitro group, Halogen atom, hydroxy group, cyclic saturation Or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, aralkyloxy group, aralkylcarbonyl group, N-aralkyl carbamoyl group, aryl group, N-arylalkylamino group, araryloxy group, § reel sulfonyl group, Arirusuru Honiruokishi group, N- § Li one Le sulfonyl § amino C i-C 6 alkoxy Cal Poni group, § reel sulfamoyl group, § Li one Rusuru sulfamoyl O alkoxy group, Arirusu Rufamoiru C E _ C i. Alkyl carbamoyl group, arylsulfamoyl 〇 1 _C 6 alkoxycarbonyl group, N-arylcarbamoyl group, aroyl group, alkoxy group, N-aroyl group, C 2 _C 6 alkanol group, C 2 — C 6 alkanoyloxy, N—C 2 _C 6 alkanoylamino, N, N—di C 2 —C 6 alkanoylamino, N—C 6 alkylamino, N—C 10 alkyl Moil group, N, N- di - C i- C 1 0 alkyl force Rubamoiru group, N _ C 2 - C 6 Al en-carbamoyl, N, N- di one C 2 - C 6 alkenyl carbamoylmethyl group, N- amino C 1 Alkyl carbamoyl group, N—C—C 6 alkoxyl C 10 Alkyl rubamoyl group, N—C “C 6 alkoxycarbonyl C! -C 1 Q alkylcarbamoyl group, C ^ —C 6 alkyl sulfinyl group, Ji E one C 6 alkoxy group, C 1 - C 6 alkoxy Cal Poni group, amino C "C 6 alkoxy Cal Poni group, N-C 3 - (- 6 cycloalkylamino group, (: 3 -〇 6 cycloalkyl Okishi group, N one C 3 - C 6 cycloalkyl force Rubamoiru group, imidazolidinyl group, imidazolin group, tetrahydrofuranyl elm group, pyrazolidinyl group, pyrazolinyl group, piperazine group, a piperidinyl group, pyrosulfate Dinyl group, pyrrolinyl group, morpholino group, isooxazolyl group, isothiazolyl group, imidazolyl group, oxazolyl group, oxadiazolyl group, thiazolyl group, thiadiazolyl group, chenyl group, triazolyl group, pyridyl group, pyridinyl group, pyrimidinyl group Pyrazolyl, pyrrolyl, pyranyl, furyl, acridinyl, isoquinolyl, isoindolyl, indazolyl, indolyl, ethylenedioxyphenyl, carbazolyl, quinazolinyl, quinoxalinyl, quinolyl, chromanyl Chromenyl group, phenanthridinyl group, phenanthrolinyl group, dibenzofuranyl group, dibenzothiophenyl group, cinnolinyl group, thonaphthenyl group, naphthyridinyl group, Enazinyl group, phenaxazinyl group, benzimidazolyl group, benzoxazolyl group, benzothiazolyl group, benzotriazolyl group, benzofuranyl group, dihydrobenzozofuranyl group, methylenedioxyphenyl group. The C 7 -C 15 carbon aromatic ring group is an acenaphthylenyl group, an adamantyl group, an anthryl group, an indenyl group, a phenanthryl group, and the 5- or 6-membered heterocyclic group is an isoxazolyl group, an isothiazolyl group, an imidazolyl group, an oxazozolyl group. Group, thiazolyl group, thiadiazolyl group, chenyl group, triazolyl group, pyridyl group, pyrazinyl group, pyrimigel group, pyridazinyl group, pyrazolyl group, pyrrolyl group, pyranyl group, and furyl group, and nitrogen, oxygen, and sulfur Selected from the group consisting of atoms 1- to 3-cyclic heteroaromatic groups having 1 to 5 heteroatoms per ring system (excluding 5- or 6-membered heterocyclic groups. ) Is an acridinyl group, Noryl group, isoindolyl group, indazolyl group, indolyl group, indolizinyl group, ethylenedioxyphenyl group, carbazolyl group, quinazolinyl group, quinoxalinyl group, quinolyl group, chromanyl group, isochromanyl group, chromenyl group, phenanthridinyl group, phenyl Nanthrolidyl group, dibenzofuranyl group, dibenzothiophenyl group, thonaphthenyl group, naphthidinyl group, phenazinyl group, phenaxazinyl group, phenothiazinyl group, phthalagel group, pteridinyl group, purinyl group, benzimidazolyl group, benzoxazolyl group , A benzothiazolyl group, a benzotriazolyl group, a benzofurayl group, a dihydrobenzofuranyl group or a methylenedioxyphenyl group, and particularly a carbon aromatic ring group is a phenyl group. , A naphthyl group, and a substituent of the optionally substituted carbon aromatic ring group may be a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carboxy group, a cyano group, a nitro group, Rogen atom, hydroxy group, aralkyl group, N-aralkylamino group, aralkyloxy group, aralkylcarponyl group, N-aralkyl rubamoyl group, aryl group, aryloxy group, arylsulfonyl group, arylsulfonyl Okishi group, § reel sulfamoyl group, Aroiru group, Arokishi group, N- Aroiru group, C 2 one C 6 Arukanoiru groups, C 2 - C 6 alkanoyloxy Noi Ruo alkoxy group, N- C 2 - C 6 alkanol Iruamino group An N-Cj-Ce alkylamino group, an N-C 10 alkyl radical, a N-N-di-C 1 -C 1 alkyl group. Alkyl carbamoyl group, N—C 2 —C 6 alkenylcarbamoyl group, N, N—di C 2 —C 6 alkenylcarbamoyl group, N—amino C 1 CJ 0 Alkyl rubamoyl group, N—C — Ce alkoxy ( : C 10 alkyl alkyl group, NC x -C 6 alkoxycarbonyl C! -0 10 alkylcarbamoyl group, C — C 6 alkylsulfinyl group, C — C 6 alkoxy group, C 1 -C 6 alkoxycarbonyl Group, amino Ci—C 6 alkoxycarbonyl group, N—C 3 — ( 6 cycloalkylamino group, (: 3 _ ( 6 cycloalkyloxy group, isoxoazolyl group, isothiazolyl group, oxazolyl group, thiazolyl group, Nyl, pyridyl, pyrazinyl, pyrimigel, pyridazinyl, furyl, tetrahydrofuranyl, morpholino, isoquinolyl, iso Ndoriru group, ethylenedioxy O carboxymethyl-phenylalanine group, quinazolinyl group, quinoxalinyl group, quinolyl group, a dibenzofuranyl El group, dibenzothiophenyl group, benzimidazolyl group, Baie Nzookisazoriru group, benzothiazolyl group, benzofuranyl group, a Mechirenjioki Shifueeru group, 1 to 3 rings of the C 7 - C 1 5 carbon aromatic ring group, Asenafuchi Reniru group, indenyl group, a Fuenansuriru group, 5- or 6-membered The heterocyclic group is an isoxazolyl group, an isothiazolyl group, an oxazolyl group, a thiazolyl group, a chenyl group, a pyridyl group, a virazinyl group, a pyrimigel group, a pyridazinyl group, or a furyl group; and the group consisting of a nitrogen atom, an oxygen atom, and a sulfur atom A mono- to tricyclic heteroaromatic group having 1 to 5 heteroatoms selected per ring system (excluding a 5- or 6-membered heterocyclic group) is an isoquinolyl group, an isoindolyl group, an ethylenedioxyphenyl group, Quinazolinyl group, quinoxalinyl group, dibenzofuranyl group, Zochiofeniru group, Fuenakisajiniru group, benzimidazolyl group, Benzoo Kisazoriru group, benzothiazolyl group, benzofuranyl group, dihydrobenzofuranyl two Le group or methylenedioxy O carboxymethyl phenylalanine group is more preferable.
A rは、 例えば水素原子、 アジド基、 アミノ基、 力ルバモイル基、 力ルバモイ ルァミノ基、 力ルバモイルォキシ基、 力ルポキシル基、 シァノ基、 スルファモイ ル基、 スルホ基、 ニトロ基、 ハロゲン原子、 ヒドロキシ基、 ホルミル基、 ホルミ ルァミノ基、 環状の飽和若しくは不飽和の C 3— C 9脂肪族基、 ァラルキル基、 N ーァラルキルアミノ基、 N, N—ジァラルキルアミノ基、 ァラルキルォキシ基、 ァラルキルカルボニル基、 N -ァラルキル力ルバモイル基、 ァリール基、 N—ァ リールアミノ基、 N, N—ジァリールアミノ基、 ァリールォキシ基、 ァリ一ルス ルホニル基、 ァリ一ルスルホニルォキシ基、 N—ァリ一ルスルホニルァミノ基、 N—ァリ—ルスルホニルァミノ C「 C 6アルキルァミノ基、 N—ァリ一ルスルホ ニルァミノ C「 C 1 0アルキル力ルバモイル基、 N—ァリ一ルスルホニルアミノ C!一 C 6アルコキシカルボ二ル基、 ァリールスルファモイル基、 ァリ一ルスルフ ァモイルォキシ基、 ァリ一ルスルファモイル C _ C i。アルキルカルパモイル基 、 ァリールスルファモイルじェ一 C 6アルコキシカルポニル基、 N—ァリールカル バモイル基、 ァロイル基、 ァロキシ基、 N-ァロイル基、 N— ( N—ァロイルァ ミノ) C x - C 1 0アルキル力ルバモイル基、 N—ァロイルァミノ 一 C 6アルコ キシカルポニル基、 C 2— C 6アルカノィル基、 C 2— C 6アルカノィルォキシ基、 N— C 2 _ C 6アルカノィルァミノ基、 N, N—ジー C 2— C 6アルカノィルアミ ノ基、 N— C^— C6アルキルアミノ基、 N, N—ジー Ci— Ceアルキルアミノ基 、 N— — 。アルキル力ルバモイル基、 N, N—ジー Ci— 。アルキル力 ルバモイル基、 N— C2— C6アルケニルカルバモイル基、 N, N—ジー C2— C6 アルケニルカルバモイル基、 N—アミノ(^一 C1Qアルキル力ルバモイル基、 N 一 Ci— C6アルコキシ 一 。アルキルカルパモイル基、 N—C「 C6アルコ キシカルボ二ル(31 -C10アルキル力ルバモイル基、 N-C,- C 6アルコキシ力 ルポニルァミノ Cェ— C i。アルキル力ルバモイル基、 N— C i一 C 6アルコキシ力 ルポニルァミノ C , _ C6アルコキシカルポニル基、 一 C6アルキルチオ基、 N - C! - C 6アルキルスルファモイル基、 N, N—ジー C 一 C 6アルキルスルファ モイル基、 C 1一 C 6アルキルスルフィニル基、 C i— C 6アルキルスルホニル基、Ar is, for example, a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, a carbamoyloxy group, a propyloxyl group, a cyano group, a sulfamoyl group, a sulfo group, a nitro group, a halogen atom, a hydroxy group, Formyl group, formylamino group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, N, N-diaralkylamino group, aralkyloxy group, aralkyl Kill carbonyl group, N-aralkyl rubamoyl group, aryl group, N-aryl amino group, N, N-diarylamino group, aryloxy group, arylsulfonyl group, arylsulfonyloxy group, N-alpha Li one Le sulfonyl § amino group, N- § Li - Le sulfonyl § amino C "C 6 Arukiruamino group, N- § Li one Rusuruho Niruamino C" C 10 alkyl alkyl group, N-arylsulfonylamino C! C 6 alkoxycarbonyl group, arylsulfamoyl group, arylsulfamoyloxy group, arylsulfamoyl C_Ci.alkyl Carpamoyl, arylsulfamoyl C 6 alkoxycarbonyl, N-arylcarbamoyl, aroyl, alkoxy, N-aroyl, N— (N-aroylamino) C x -C 10 Alkyl force Rubamoiru group, N- Aroiruamino one C 6 alkoxy Kishikaruponiru groups, C 2 - C 6 Arukanoiru groups, C 2 - C 6 alkanoyloxy Noi Ruo alkoxy group, N- C 2 _ C 6 alkanoyloxy Noi Rua amino group, N, N- Gee C 2 — C 6 alkanoylami Group, N—C ^ —C 6 alkylamino group, N, N—diCi—Ce alkylamino group, N——. Alkyl Rubamoyl group, N, N—Gee Ci—. Alkyl force Rubamoiru group, N- C 2 - C 6 alkenyl carbamoylmethyl group, N, N- di C 2 - C 6 alkenyl carbamoylmethyl group, N- amino (^ one C 1Q alkyl force Rubamoiru group, N one CI- C 6 alkoxy 1. Alkyl carpamoyl group, N—C “C 6 alkoxycarbyl (31-C 10 alkyl group Lubamoyl group, NC, —C 6 alkoxy group Luponylamino C—C i. Alkyl group Lbamoyl group, N—C i-C 6 alkoxyl force L-ponylamino C, _ C 6 alkoxycarbonyl group, C 6 alkylthio group, N-C! -C 6 alkylsulfamoyl group, N, N-G C-C 6 alkylsulfamoyl group, C 1 -C 6 alkylsulfinyl group, C i — C 6 alkylsulfonyl group,
Cj— c6アルコキシ基、 c! - C 6アルコキシカルボニル基、 アミノ 一 c6ァ ルコキシカルポニル基、 N_C3—〇6シクロアルキルァミノ基、 N, N—ジ一 C 3 _ C 6シクロアルキルァミノ基、 C 3— C 6シクロアルキルォキシ基、 N-C3 _ C 6シクロアルキル力ルバモイル基、 N, N—ジ— C3— ( 6シクロアルキル力 ルバモイル基からなる群から選択される置換基、 5又は 6員の複素環基、 窒素原 子、 酸素原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1な いし 5個有する 1ないし 3環性の複素芳香環基又は該置換基で置換されていても よレ 直鎖若しくは分枝鎖の飽和若しくは不飽和の Ci— Cg脂肪族基、 C — C6 アルコキシ基、 一 C6アルキルチオ基、 N— 一 C6アルキルアミノ基、 N— Cx-C6アルキル力ルバモイル基、 N-Cx-C6アルキルチオ力ルバモイル基か らなる群より選ばれる置換基を有していてもよい炭素芳香環基、 1ないし 3環性 の C 7— C i 5炭素芳香環基又は 5若しくは 6員の複素環基、 若しくは窒素原子、 酸素原子及び硫黄原子からなる群より選ばれる複素原子を 1環系当たり 1ないし 5個有する 2ないし 3環性の縮合複素芳香環基 (但し、 5若しくは 6員の複素環 基を除く。 ) を示し、 中でも炭素芳香環基が、 フエニル基、 ナフチル基であり、 置換基を有していてもよい炭素芳香環基の置換基が、 水素原子、 アジド基、 アミ ノ基、 力ルバモイル基、 力ルバモイルァミノ基、 力ルバモイルォキシ基、 力ルポ キシル基、 シァノ基、 ニトロ基、 ハロゲン原子、 ヒドロキシ基、 環状の飽和若し くは不飽和の C3— C9脂肪族基、 ァラルキル基、 N—ァラルキルアミノ基、 ァラ ルキルォキシ基、 ァラルキルカルポニル基、 N—ァラルキルカルバモイル基、 ァ リール基、 N—ァリ一ルァミノ基、 ァリールォキシ基、 ァリ一ルスルホニル基、 ァリ一ルスルホニルォキシ基、 N—ァリ一ルスルホニルァミノ Cェ一 C 6アルコキ シカルボニル基、 ァリールスルファモイル基、 ァリ一ルスルフ 7モイルォキシ基 、 ァリ一ルスルファモイル(^一 C10アルキル力ルバモイル基、 ァリールスルフ ァモイルじェ一 C6アルコキシカルポニル基、 N—ァリ一ルカルバモイル基、 ァロ ィル基、 ァロキシ基、 N—ァロイル基、 C2— C6アルカノィル基、 C2— C6アル カノィルォキシ基、 N-C2-C6アル力ノィルァミノ基、 N, N—ジー C2— C 6アルカノィルァミノ基、 N— Ci— C6アルキルアミノ基、 N— C1ーC10ァルキ ルカルバモイル基、 N, N—ジ— 一 C1Qアルキル力ルバモイル基、 N— C2 一 C6アルケニルカルバモイル基、 N, N—ジ _ C 2— C 6アルケニルカルバモイ ル基、 N—アミ 一 C10アルキル力ルバモイル基、 N—Ci— C6アルコキシ 一 G10アルキル力ルバモイル基、 N— C i一 C 6アルコキシ力ルポ二ルじ1一 C 10アルキル力ルバモイル基、 C丄一 C 6アルキルスルフィニル基、 C「C6アルコ キシ基、 C 1― C 6アルコキシカルポニル基、 アミノ C i _ C 6アルコキシ力ルポ ニル基、 N— C3— (36シクロアルキルアミノ基、 C3— C 6シクロアルキルォキ シ基、 N— C3—じ6シクロアルキル力ルバモイル基、 イミダゾリジニル基、 イミ ダゾリニル基、 テトラヒドロフラエル基、 ビラゾリジニル基、 ピラゾリニル基、 ピペラジニル基、 ピペリジニル基、 ピロリジニル基、 ピロリニル基、 モルホリノ 基、 イソォキサゾリル基、 イソチアゾリル基、 イミダゾリル基、 ォキサゾリル基 、 ォキサジァゾリル基、 チアゾリル基、 チアジアゾリル基、 チェニル基、 トリア ゾリル基、 ピリジル基、 ビラジニル基、 ピリミジェル基、 ピリダジニル基、 ビラ ゾリル基、 ピロリル基、 ピラエル基、 フリル基、 ァクリジニル基、 イソキノ.リル 基、 イソインドリル基、 インダゾリル基、 インドリル基、 エチレンジォキシフエ ニル基、 カルバゾリル基、 キナゾリニル基、 キノキサリニル基、 キノリル基、 ク ロマニル基、 クロメニル基、 フエナンスリジニル基、 フエナンスロリニル基、 ジ ベンゾフラニル基、 ジベンゾチオフェニル基、 シンノリニル基、 チォナフテニル 基、 ナフチリジニル基、 フエナジニル基、 フエナキサジニル基、 ベンゾイミダゾ リル基、 ベンゾォキサゾリル某、 ベンゾチアゾリル基、 ベンゾトリアゾリル基、 ベンゾフラニル基、 ジヒドロべンゾフラエル基、 メチレンジォキシフエニル基で あり、 1ないし 3環性の — 5炭素芳香環基が、 ァセナフチレニル基、 ァダ マンチル基、 アンスリル基、 インデニル基、 フエナンスリル基であり、 5若しく は 6員の複素環基が、 イソォキサゾリル基、 イソチアゾリル基、 イミダゾリル基 、 ォキサゾリル基、 チアゾリル基、 チアジアゾリル基、 チェニル基、 ピリジル基 、 ビラジニル基、 ピリミジェル基、 ピリダジニル基、 ピラゾリル基、 ピロリル基 、 ピラニル基、 フリル基であり、 窒素原子、 酸素原子及び硫黄原子からなる群よ り選ばれる複素原子を 1環系当たり 1ないし 5個有する 1ないし 3環性の縮合複 素芳香環基 (5若しくは 6員の複素環基を除く。 ) が、 ァクリジニル基、 イソキ ノリル基、 イソインドリル基、 インダゾリル基、 インドリル基、 インドリジニル 基、 エチレンジォキシフエニル基、 カルバゾリル基、 キナゾリニル基、 キノキサ リニル基、 キノリル基、 クロマニル基、 イソクロマニル基、 クロメニル基、 フエ ナンスリジニル基、 フエナンスロリジル基、 ジベンゾフラニル基、 ジベンゾチォ フエニル基、 チォナフテニル基、 ナフチジニル基、 フエナジニル基、 フエナキサ ジニル基、 フエノチアジニル基、 フタラジェル基、 プテリジニル基、 プリニル基 、 ベンゾイミダゾリル基、 ベンゾォキサゾリル基、 ベンゾチアゾリル基、 ベンゾ トリァゾリル基、 ベンゾフラニル基、 ジヒドロベンゾフラニル基若しくはメチレ ンジォキシフエニル基が好適であり、 特に炭素芳香環基が、 フエニル基、 ナフチ ル基であり、 置換基を有していてもよい炭素芳香環基の置換基が、 水素原子、 ァ ジド基、 アミノ基、 力ルバモイル基、 力ルポキシル基、 シァノ基、 ニトロ基、 ノ\ ロゲン原子、 ヒドロキシ基、 ァラルキル基、 N—ァラルキルアミノ基、 ァラルキ ルォキシ基、 ァラルキルカルポニル基、 N—ァラルキル力ルバモイル基、 ァリー ル基、 ァリールォキシ基、 ァリールスルホニル基、 ァリールスルホニルォキシ基 、 ァリールスルファモイル基、 ァロイル基、 ァロキシ基、 N—ァロイル基、 C 2 一 C 6アルカノィル基、 C 2— C 6アルカノィルォキシ基、 N— C 2— C 6アルカノ ィルァミノ基、 N— 一 C 6アルキルアミノ基、 N—C i— C 1 0アルキル力ルバ モイル基、 N, N—ジ— C i - C ^アルキル力ルバモイル基、 N— C 2— C 6アル ケニルカルバモイル基、 N, N—ジー C 2— C 6アルケニルカルバモイル基、 N— アミノ C i— C 1 0アルキル力ルバモイル基、 N— — C 6アルコキシ d— C 1 0 アルキル力ルバモイル基、 N _ C 一 C 6アルコキシカルポニル C i— C 。アルキ ルカルバモイル基、 C i一 C 6アルキルスルフィニル基、 ( 1—0 6ァルコキシ基、 C!一 C 6アルコキシカルポニル基、 アミノ C i一 C 6アルコキシカルボ二ル基、 N— C 3— C 6シクロアルキルアミノ基、 C 3— (:6シクロアルキルォキシ基、 ィ ソォキサゾリル基、 イソチアゾリル基、 ォキサゾリル基、 チアゾリル基、 チェ二 ル基、 ピリジル基、 ピラジニル基、 ピリミジニル基、 ピリダジニル基、 フリル基 、 テトラヒドロフラニル基、 モルホリノ基、 イソキノリル基、 イソインドリル基 、 エチレンジォキシフエニル基、 キナゾリニル基、 キノキサリニル基、 キノリル 基、 ジベンゾフラニル基、 ジベンゾチオフェニル基、 ベンゾイミダゾリル基、 ベ ンゾォキサゾリル基、 ベンゾチアゾリル基、 ベンゾフラニル基、 メチレンジォキ シフエニル基であり、 1ないし 3環性の C 7— C 1 5炭素芳香環基が、 ァセナフチ レニル基、 インデニル基、 フエナンスリル基であり、 5若しくは 6員の複素環基 が、 イソォキサゾリル基、 イソチアゾリル基、 ォキサゾリル基、 チアゾリル基、 チェニル基、 ピリジル基、 ピラジニル基、 ピリミジニル基、 ピリダジニル基、 フ リル基であり、 窒素原子、 酸素原子及び硫黄原子からなる群より選ばれる複素原 子を 1環系当たり 1ないし 5個有する 1ないし 3環性の複素芳香環基 ( 5.又は 6 員の複素環基を除く。 ) が、 イソキノリル基、 イソインドリル基、 エチレンジォ キシフエニル基、 キナゾリニル基、 キノキサリニル基、 ジベンゾフラエル基、 ジ ベンゾチオフェニル基、 フエナキサジニル基、 ベンゾイミダゾリル基、 ベンゾォ キサゾリル基、 ベンゾチアゾリル基、 ベンゾフラニル基、 ジヒドロベンゾフラ二 ル基若しくはメチレンジォキシフエニル基がより好適である。 Cj—c 6 alkoxy group, c! -C 6 alkoxycarbonyl group, amino-c 6 alkoxycarbonyl group, N_C 3 —〇 6 cycloalkylamino group, N, N-di-C 3 _ C 6 cycloalkylamino group, C 3 — C 6 A cycloalkyloxy group, a substituent selected from the group consisting of NC 3 _ C 6 cycloalkyl group rubamoyl group, N, N-di-C 3 — ( 6 cycloalkyl group rubamoyl group, 5- or 6-membered heterocyclic ring Group, a nitrogen atom, an oxygen atom, and a sulfur atom, a mono- to tricyclic heteroaromatic group having 1 to 5 heteroatoms per ring system selected from the group consisting of an oxygen atom and a sulfur atom, Linear or branched saturated or unsaturated Ci—Cg aliphatic group, C—C 6 alkoxy group, C 1 C 6 alkylthio group, N—C 1 C 6 alkylamino group, N—C x -C 6 alkyl force Rubamoiru group, Ranaru group or NC x -C 6 alkylthio force Rubamoiru group Good carbon aromatic ring group optionally having a substituent selected from 1 to 3 rings of the C 7 - C i 5 carbon aromatic ring group or a 5- or 6-membered heterocyclic group, or a nitrogen atom, oxygen atom and sulfur A bicyclic to tricyclic fused heteroaromatic ring group having 1 to 5 heteroatoms per ring system selected from the group consisting of atoms (excluding a 5- or 6-membered heterocyclic group); The aromatic ring group is a phenyl group or a naphthyl group, and the optionally substituted carbon aromatic ring group is a hydrogen atom, an azide group, an amino group, a carbamoyl group, a carbamoylamino group, Rubamoyloxy group, hydroxyl group, cyano group, nitro group, halogen atom, hydroxy group, cyclic saturated or unsaturated C 3 -C 9 aliphatic group, aralkyl group, N-aralkylamino group, ara Ruxyoxy group, aralkylcarbonyl group, N-aralkylcarbamoyl group, aryl group, N-arylamino group, aryloxy group, arylsulfonyl group, arylsulfonyloxy group, N— Arylsulfonylamino C 1 C 6 alkoxycarbonyl group, arylsulfamoyl group, arylsulfo 7 moyloxy group, arylsulfamoyl (^ C 10 alkyl alkyl group, arylsulfamoyl group, arylsulfamoyl group) C 6 alkoxy Cal Poni Le group, N- § Li one carbamoyl group, § b I group, Arokishi group, N- Aroiru group, C 2 - C 6 Arukanoiru groups, C 2 - C 6 Al Kanoiruokishi group, NC 2 -C 6 Al force Noiruamino group, N, N-di-C 2 - C 6 alkanoyloxy Noi Rua amino group, N- Ci- C 6 alkylamino group, N-C 1 over C 10 Aruki carbamoyl group, N, N-di One C 1Q alkyl force Rubamoiru group, N- C 2 one C 6 alkenyl carbamoylmethyl group, N, N- di _ C 2 - C 6 alkenyl carbamoylthiopheno Le group, N- Ami one C 10 alkyl force Rubamoiru group, N-Ci - C 6 alkoxy one G 10 alkyl force Rubamoiru group, N-C i one C 6 alkoxy force Lupo sulfonyl Ji 1 one C 10 alkyl force Rubamoiru groups, C丄one C 6 alkylsulfinyl groups, C "C 6 alkoxy group , C 1 - C 6 alkoxy Cal Poni group, an amino C i _ C 6 alkoxy force Lupo sulfonyl group, N- C 3 - (3 6 cycloalkylamino group, C 3 - C 6 cycloalkyl O key sheet group, N-C 3 - Ji 6 cycloalkyl force Rubamoiru group, imidazolidinyl group, imino Dazoriniru group, tetrahydrofuranyl El group, Birazorijiniru group, pyrazolinyl group, a piperazinyl group, a piperidinyl group, pyrrolidinyl group, pyrrolinyl group, Moruhori Group, isoxazolyl group, isothiazolyl group, imidazolyl group, oxazolyl group, oxdiazolyl group, thiazolyl group, thiadiazolyl group, chenyl group, triazolyl group, pyridyl group, virazinyl group, pyrimigel group, pyridazyl group, pirazolyl group Group, furyl group, acridinyl group, isoquinolyl group, isoindolyl group, indazolyl group, indolyl group, ethylenedioxyphenyl group, carbazolyl group, quinazolinyl group, quinoxalinyl group, quinolyl group, chromanyl group, chromenyl group, phenyl Nanthridinyl group, phenanthrolinyl group, dibenzofuranyl group, dibenzothiophenyl group, cinnolinyl group, thonaphthenyl group, naphthyridinyl group, phenazinyl group, phenaxazinyl group, Benzoimidazolyl group, Benzoxazolyl group, Benzothiazolyl group, Benzotriazolyl group, Benzofuranyl group, dihydro base Nzofuraeru group, a methylenedioxy O carboxymethyl-phenylalanine group, 1 to 3 rings of - 5 carbon aromatic ring group, Asenafuchireniru group, § Da Manchiru group, anthryl group, indenyl group, a Fuenansuriru group , A 5- or 6-membered heterocyclic group is an isooxazolyl group, an isothiazolyl group, an imidazolyl group, an oxazolyl group, a thiazolyl group, a thiadiazolyl group, a phenyl group, a pyridyl group, a pyrazinyl group, a pyrazolyl group A pyranyl group or a furyl group, a 1 to 3 ring condensed compound aromatic ring group having 1 to 5 hetero atoms per ring system selected from the group consisting of a nitrogen atom, an oxygen atom and a sulfur atom ( Excluding a 5- or 6-membered heterocyclic group.) Is an acridinyl group, isoquinolyl , Isoindolyl group, indazolyl group, indolyl group, indolizinyl group, ethylenedioxyphenyl group, carbazolyl group, quinazolinyl group, quinoxalinyl group, quinolyl group, chromanyl group, isochromanyl group, chromenyl group, fenansuridinyl group, fenansloryl Zyl group, dibenzofuranyl group, dibenzothiophenyl group, tonaphthenyl group, naphthidinyl group, phenazinyl group, phenaxinyl group, phenothiazinyl group, phthalagel group, pteridinyl group, purinyl group, benzoimidazolyl group, benzothiazolyl group, benzothiazolyl group Group, a benzotriazolyl group, a benzofuranyl group, a dihydrobenzofuranyl group or a methylenedioxyphenyl group. Particularly, the carbon aromatic ring group is preferably a phenyl group or a naphthyl group. A hydrogen atom, an azide group, an amino group, a carbamoyl group, a propyloxyl group, a cyano group, a nitro group, or a nitrogen atom, which may have a substituent. Atom, hydroxy group, aralkyl group, N-aralkylamino group, aralkyloxy group, aralkylcarbonyl group, N-aralkyl rubamoyl group, aryl group, aryloxy group, arylsulfonyl group, arylsulfonyloxy group, aryl group reel sulfamoyl group, Aroiru group, Arokishi group, N- Aroiru group, C 2 one C 6 Arukanoiru groups, C 2 - C 6 alkanoyloxy Noi Ruo alkoxy group, N- C 2 - C 6 alkano Iruamino group, N- one C 6 alkylamino group, N-C i- C 1 0 alkyl force Luba moil group, N, N-di - C i - C ^ alkyl force Rubamoiru group, N- C 2 - C 6 Al Kenirukarubamo Le group, N, N- di C 2 - C 6 alkenyl carbamoylmethyl group, N- amino C i-C 1 0 alkyl force Rubamoiru group, N- - C 6 alkoxy d-C 1 0 Alkyl force Rubamoiru group, N _ C one C 6 alkoxy Cal Poni Le C i-C. ! Alkylcarbamoyl group, C i one C 6 alkylsulfinyl group, (1 -0 6 Arukokishi groups, C one C 6 alkoxy Cal Poni group, an amino C i one C 6 alkoxycarbonyl alkylsulfonyl group, N-C 3 - C 6 cycloalkyl Alkylamino group, C 3 — (: 6 cycloalkyloxy group, dioxoxazolyl group, isothiazolyl group, oxazolyl group, thiazolyl group, cetyl group, pyridyl group, pyrazinyl group, pyrimidinyl group, pyridazinyl group, furyl group, tetrahydrofura Nyl, morpholino, isoquinolyl, isoindolyl, ethylenedioxyphenyl, quinazolinyl, quinoxalinyl, quinolyl, dibenzofuranyl, dibenzothiophenyl, benzimidazolyl, benzoxazolyl, benzothiazolyl, benzofuranyl Group, methylene A dioxy phenyl group, a 1 to 3 cyclic C 7 to C 15 carbon aromatic ring group is an acenaphthylenyl group, an indenyl group, or a phenanthryl group; and a 5- or 6-membered heterocyclic group is an isoxazolyl group or an isothiazolyl group. Group, oxazolyl group, thiazolyl group, thienyl group, pyridyl group, pyrazinyl group, pyrimidinyl group, pyridazinyl group, and furyl group, and a heteroatom selected from the group consisting of nitrogen atom, oxygen atom and sulfur atom as one ring 1 to 5 mono- to 3-cyclic heteroaromatic groups (excluding 5- or 6-membered heterocyclic groups) per group include isoquinolyl, isoindolyl, ethylenedioxyphenyl, quinazolinyl, quinoxalinyl, and dibenzofuran. L group, dibenzothiophenyl group, phenaxazinyl group, benzimidazolyl group, ben A zoxazolyl group, a benzothiazolyl group, a benzofuranyl group, a dihydrobenzofuranyl group or a methylenedioxyphenyl group is more preferred.
nは、 0— 2の整数を示し、 中でも 0又は 2が好適であり、 特に、 2がより好 適でめる。 n represents an integer of 0 to 2, and 0 or 2 is preferable, and 2 is more preferable.
本発明の一般式 [ I ] General formula [I] of the present invention
[式中、 R R2、 R3、 R4、 R 5及び A rは前記の意味を有する。 ]で表される 化合物の代表例を表 1力、ら表 15に示す。 [Wherein, RR 2 , R 3 , R 4 , R 5 and Ar have the above-mentioned meanings. ] Are shown in Table 1 and Table 15.
z H Jd- ! H H Md 0201- z Md-9IAI-8 H Jd -! H H Md 6UU z Md-9I^I-i7 H Jd -! H H Md 8L0Lz H Jd-! HH Md 0201- z Md- 9 IAI-8 H J d-! HH Md 6UU z Md- 9 I ^ I-i7 H Jd-! HH Md 8L0L
0 Md 」d -! H H Md LlOi z d H 」d -! H H Md 9L0L0 Md "d-! H H Md LlOi z d H "d-! H H Md 9L0L
0 Md H 」d -! H H Md 1 10 Md H "d-! H H Md 1 1
0 | jnj - 2 H 」d -! H H Md OL0 | jnj-2H "d-! H H Md OL
0 |Au9jm-2 H 」d -! H H Md 0 | Au9jm-2 H "d-! H H Md
0 1八 ρμ八 d - 2 H Jd-! H H Md ZlOl z ιΑρμ八 d - ε H 」d -! H H Md not 0 1 8 ρμ 8 d-2 H Jd-! H H Md ZlOl z ιΑρμ 8 d-ε H H H Md not
0 | 八 d - ε H Jd-! H H Md OiOl0 | Eight d-ε H J d-! HH Md OiOl
2 |Λρμ八 d - H ^d-! H H Md 6002 | Λρμ8 d-H ^ d-! H H Md 600
0 |Λρμ八 d- H 」d-! H H Md 8001 z Md H H H 」d-! Hd Loov0 | Λρμ8 d-H "d-! H H Md 8001 z Md H H H" d-! Hd Loov
0 Md H H H Jd -! Hd 900 L u 1 M Jd」 1 I 0 Md HHH J d-! Hd 900 Lu 1 M Jd '' 1 I
H H H 1 H 1d」 H H H 1 H 1d "
U Md u Π 1コ u H u Π Md 1700LU Md u Π 1 u u u Π Md 1700L
2 Md H 9IAI H H Hd εοοΐ·2 Md H 9 IAI HH Hd εοοΐ
0 Md H 9ΙΛΙ H H Md 200 L0 Md H 9 ΙΛΙ HH Md 200 L
0 Md H H H H Md 瞧0 Md H H H H Md 瞧
-u 9id l7d Z m -u 9id l7d Z m
Z,9Zll/lOdf/13d 表 2 Z, 9Zll / lOdf / 13d Table 2
化合物例 Ar R1 R2 R3 R4 R5 n= Compound example Ar R1 R2 R3 R4 R5 n =
1021 Ph H H i-Pr H 3-CF3-Ph 21021 Ph HH i-Pr H 3-CF 3 -Ph 2
1022 Ph H H 卜 Pr H 4-GF3-Ph 21022 Ph HH Pr H 4-GF 3 -Ph 2
1023 Ph H H 卜 Pr H 4-Ph-Ph 21023 Ph H H Pr Pr H 4-Ph-Ph 2
1024 Ph H H i-Pr H 3-Ph-Ph 21024 Ph H H i-Pr H 3-Ph-Ph 2
1025 Ph H H i-Pr H 2 - Ph - Ph 21025 Ph H H i-Pr H 2-Ph-Ph 2
1026 Ph H H i-Pr H 4 - Cl-Ph 21026 Ph H H i-Pr H 4-Cl-Ph 2
1027 Ph H H 卜 Pr H 3 - Cl-Ph 21027 Ph H H b Pr H 3-Cl-Ph 2
1028 Ph H H 卜 Pr H 2 - G卜 Ph 21028 Ph H H Pr Pr H 2-G Ph Ph 2
1029 Ph H H 卜 Pr H 3-F-Ph 21029 Ph H H Pr Pr H 3-F-Ph 2
1030 Ph H H i-Pr H 4-F-Ph 21030 Ph H H i-Pr H 4-F-Ph 2
1031 Ph H H i-Pr H 4 - OH - Ph 01031 Ph H H i-Pr H 4-OH-Ph 0
1032 Ph H H 卜 Pr H 4 - OH - Ph 21032 Ph H H Pr H 4-OH-Ph 2
1033 Ph H H i-Pr H 3 - OH - Ph 21033 Ph H H i-Pr H 3-OH-Ph 2
1034 Ph H H 卜 Pr H 2 - OH - Ph 21034 Ph H Ht Pr H 2-OH-Ph 2
1035 Ph H H 卜 Pr H 4-MeO-Ph 21035 Ph H H Pr Pr H 4-MeO-Ph 2
1036 Ph H H 卜 Pr H 3-MeO-Ph 21036 Ph H H r Pr H 3-MeO-Ph 2
1037 Ph H H 卜 Pr H 2-MeO-Ph 21037 Ph H H r Pr H 2-MeO-Ph 2
1038 Ph H H 卜 Pr H 4-PhO-Ph 21038 Ph H H Pr Pr H 4-PhO-Ph 2
1039 Ph H H i-Pr H 4-PhCOO-Ph 21039 Ph H H i-Pr H 4-PhCOO-Ph 2
1040 Ph H H i-Pr H 4-PhCH O-Ph 0 1040 Ph HH i-Pr H 4-PhCH O-Ph 0
22()h HCp o〇H0- σ> 22 () h HCp o〇H0- σ>
o CO o CO
"D "D
了 End
ェ E
工 Engineering
1 1
■D ■ D
, ,
ェ E
l\3l \ 3
/ OョAV - / Oyo AV-
CM CM CM CM CM CM CM CM CM CsJ Csl CM M CM CM C CM CM CvJ CM CM CM CM CM CM CM CM CM CM CsJ Csl CM M CM CM C CM CM CvJ CM
x: x:
Q. Q.
Q. Q.
CL I CL I
CL CL CL CL
〇 〇
ェ , ェ Eh eh
〇 ェ 〇 〇 〇 〇
z z
LO dd _〕〇 M 」.l LO dd_] 〇 M '' .l
寸 寸 d δο干寸干 」- 寸 Dimensions d δο Dimensions-Dimensions
01 ェ ェ 工干 」 ェ ェ ェ 工 ェ ェ ェ ェ ェ ェ X X X ェ ェ 01 X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X X
CO ο寸 Md Ηοώ- HG丄寸d ldOdM 」」-l CO ο Dimension Md Ηοώ- HG 丄 Dimension d ldOdM ”” -l
(Nl (Nl
ェ ェ ェ ェ (0Η寸dェΟΙΟ ΤΗ 」ω—- ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ 工 ェ ェ d 」- ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ ェ 工 ェ ェ ェ ェ d 」- (Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η — — ω Η Η — Η Η Η D)
X: JZ ぷ z X: JZ ぷ z
< CL Q_ Q_ 0- Q_ Q_ CL Q_ HH寸干 4d Q0N, 」」_ Q_ Q_ CL £L GL G_ CL CL CL Q. 寸 CM CO 寸 LO O CO σ> 0寸 o 4dH」 - CM 00 寸 CO 00 σ> Ο o O CD CD CD CD O O O CO N <CL Q_ Q_ 0- Q_ Q_ CL Q_ HH dimension 4d Q0N, '' '' _ Q_ Q_ CL £ L GL G_ CL CL CL Q. dimension CM CO dimension LO O CO σ> 0 dimension o 4dH ''-CM 00 dimension CO 00 σ> Ο o O CD CD CD CD OOO CO N
卜 00 o O O O O o o o o o o o o o o o o o ο ο 干 00 00 O O O O o o o o o o o o o o o o o ο ο
T〇寸C MC¾HT 」I T〇Dimension C MC¾HT '' I
£01寸d-/ 」ω-ιl £ 01 inch d-/ "ω-ιl
Md23寸 dll- Md23 dll-
Mdo¾H寸d 」l-I Mdo¾H dimension d '' l-I
〕〇HN 1Λ寸Ι £Ι1 ) 〇HN 1ΛsizeΙ £ Ι1
£1 £ 1
干 ぶ d- 表 5 Dry d- Table 5
合物例 Ar R1 R2 R3 R4 R5 n= Compound example Ar R1 R2 R3 R4 R5 n =
1081 Ph H H 卜 Pr H MeCONH - Ph 21081 Ph H H Pr Pr H MeCONH-Ph 2
1082 Ph H H i-Pr H PhCONH - Ph 21082 Ph H H i-Pr H PhCONH-Ph 2
1083 Ph H H i-Pr H 2 - Me-3-Me-Ph 21083 Ph H H i-Pr H 2-Me-3-Me-Ph 2
1084 Ph H H i-Pr H 2 - Me - 4- Me - Ph 21084 Ph H H i-Pr H 2-Me-4- Me-Ph 2
1085 Ph H H i-Pr H 2-Me-5-Me-Ph 21085 Ph H H i-Pr H 2-Me-5-Me-Ph 2
1086 Ph H H 卜 Pr H 2 - Me+Me-Ph 21086 Ph H H Pr Pr H 2-Me + Me-Ph 2
1087 Ph H H i-Pr H 3 - Me - 4-Me - Ph 21087 Ph H H i-Pr H 3-Me-4-Me-Ph 2
1088 Ph H H 卜 Pr H 3 - Me-5-Me-Ph 21088 Ph H H Pr H 3-Me-5-Me-Ph 2
1089 Ph H H 卜 Pr H 2,4,6-triMe-Ph 21089 Ph H H Pr H 2,4,6-triMe-Ph 2
1090 Ph H H 卜 Pr H 3 - F-4-Me - Ph 21090 Ph H H Pr H 3-F-4-Me-Ph 2
1091 Ph H H i-Pr H 3-CI-4-Me-Ph 21091 Ph H Hi-Pr H 3-CI-4-Me-Ph 2
1092 Ph H H 卜 Pr H 3-Me-4-F-Ph 21092 Ph H H Pr Pr H 3-Me-4-F-Ph 2
1093 Ph H H i-Pr H 3-Me-4-C卜 Ph 21093 Ph H H i-Pr H 3-Me-4-C Ph 2
1094 Ph H H 卜 Pr H 3 - F-4-F - Ph 21094 Ph H H b Pr H 3-F-4-F-Ph 2
1095 Ph H H 卜 Pr H 3-F- 4-Cト Ph 21095 Ph H H to Pr H 3-F-4-C to Ph 2
1096 Ph H H 卜 Pr H 3-CI-4-CI-Ph 21096 Ph H H H Pr H 3-CI-4-CI-Ph 2
1097 Ph H H 卜 Pr H 3-OH-4 - Me-Ph 21097 Ph H H Pr H 3-OH-4-Me-Ph 2
1098 Ph H H i-Pr H 3-PhCH20-4-Me-Ph 21098 Ph HH i-Pr H 3-PhCH 2 0-4-Me-Ph 2
1099 Ph H H i-Pr H 3- MeO - 4 - Me - Ph 21099 Ph H H i-Pr H 3-MeO-4-Me-Ph 2
1 100 Ph H H 卜 Pr H 3-Me—4— MeO-Ph 2 表 6 1 100 Ph HH Pr H 3-Me—4—MeO-Ph 2 Table 6
合物例 Ar R1 R2 R3 R4 R5 n= Compound example Ar R1 R2 R3 R4 R5 n =
1101 Ph H H 卜 Pr H 3-Vinyl-4-MeO-Ph 21101 Ph H H Pr Pr H 3-Vinyl-4-MeO-Ph 2
1102 Ph H H 卜 Pr H 3+PrO - 4一 Me - Ph 21102 Ph H Ht Pr H 3 + PrO-4 Me-Ph 2
1103 Ph H H 卜 Pr H 3 - MeO - 4一 MeO - Ph 21103 Ph H Ht Pr H 3-MeO-4 1 MeO-Ph 2
1104 Ph H H 卜 Pr H 3,4 - Methylendioxy-Ph 21104 Ph H H p Pr H 3,4-Methylendioxy-Ph 2
1 105 Ph H H i-Pr H 3,4-Ethylendioxy-Ph 21 105 Ph H H i-Pr H 3,4-Ethylendioxy-Ph 2
1 106 Ph H H i-Pr H 3 - OH - 4一 MeO - Ph 21 106 Ph H H i-Pr H 3-OH-4 MeO-Ph 2
1107 Ph H H i-Pr H 3 -ト 4一 OH - Ph 01107 Ph H H i-Pr H 3-to 4 OH-Ph 0
1 108 Ph H H i-Pr H 3 -ト 4一 OH - Ph 21 108 Ph H H i-Pr H 3-to 4 OH-Ph 2
1109 Ph H H i-Pr H 2 - OH - 5 -ト Ph 21109 Ph H H i-Pr H 2-OH-5-G Ph 2
1110 Ph H H i-Pr H 2-1-5-OH-Ph 21110 Ph H H i-Pr H 2-1-5-OH-Ph 2
1 1 1 1 Ph Π u u 1 1 1 1 Ph Π u u
Π D u Π ゥ一 I 1 "T一一■!— Μ Me ΛU— D r Un> ά Π D u Π ゥ 一 I 1 "T 一一 ■! — Μ Me ΛU— D r Un> ά
1112 Ph H H 卜 Pr H 3 - C卜 4-MeO-Ph 21112 Ph H H Pr Pr H 3-C 4- 4-MeO-Ph 2
11 13 Ph H H i-Pr H 3 - Br - 4 - MeO- Ph 211 13 Ph H H i-Pr H 3-Br-4-MeO- Ph 2
1114 Ph H H 卜 Pr H 3-1-4-MeO-Ph 01114 Ph H H Pr Pr H 3-1-4-MeO-Ph 0
1115 Ph H H i-Pr Me 3+4 - MeO - Ph 01115 Ph H H i-Pr Me 3 + 4-MeO-Ph 0
11 16 Ph H H 卜 Pr H 3+4 - MeO - Ph 211 16 Ph H Ht Pr H 3 + 4-MeO-Ph 2
11 17 Ph H H 卜 Pr H 2+5 - MeO - Ph 211 17 Ph H H Pr Pr H 2 + 5-MeO-Ph 2
1118 Ph H H 卜 Pr H 2-MeO- 5-卜 Ph 21118 Ph H H Pr Pr H 2-MeO- 5- 卜 Ph 2
1 119 Ph H H 卜 Pr H 3-卜 4- EtO - Ph 21 119 Ph H H Pr Pr H 3- 卜 4-EtO-Ph 2
1120 Ph H H 卜 Pr H 3-1-4-i-PrO-Ph 2 3丫人lphco 44N〇3-l-l CO CO CO CO 3>(hG 4NpO 3丁4〇-1-- u "0 ■ϋ "D1120 Ph HH Pr H 3-1-4-i-PrO-Ph 2 3 丫 lphco 44N〇3-ll CO CO CO CO 3> (hG 4NpO 3 丁 4〇-1-- u "0 ■ ϋ" D
3)(hco 34Np〇l4lll-- 3" ェ ェ ェ ェoph 34o丄phc-- 1 ェ ェ ェ 工 ェ yyェoindol4Me〇 3〇〇DdrihIll 3) (hco 34Np〇l4lll-- 3 "
1 1 1 1 11 1 1 1 1
"D "D "D "0 "D ェ ェ ェ ェ ェ "D" D "D" 0 "D
ID -aID -a
r 了 r r
■ϋ 了 ■ ϋ End
表 8 Table 8
合物例 Ar 1 R2 R3 R誦、4 R5 pi二 Compound example Ar 1 R2 R3 R recitation, 4 R5 pi
1141 Ph H H 卜 Pr H 4 - (4- N3-3-卜 PhCOO)-Ph 21141 Ph HH Pr Pr H 4-(4-N 3 -3- Ph PhCOO) -Ph 2
1142 Ph H H i-Pr H 3+4-MeNHCOCH20-Ph 21142 Ph HH i-Pr H 3 + 4-MeNHCOCH 20 -Ph 2
1143 Ph H H 卜 Pr H 3-|-4-EtNHCOCH20-Ph 21143 Ph HH Pr H 3- | -4-EtNHCOCH 2 0-Ph 2
1144 Ph H H i-Pr H 3—卜 4-卜 prNHG〇GH2〇-Ph 21144 Ph HH i-Pr H 3— 4-p r NHG〇GH 2 〇-Ph 2
1145 Ph H H i-Pr H 3+4 - n - BuNHG〇GH20-Ph 21145 Ph HH i-Pr H 3 + 4-n-BuNHG〇GH 2 0-Ph 2
1146 Ph H H i-Pr H 3+4— n— PrNHGOGH20-Ph 01146 Ph HH i-Pr H 3 + 4— n— PrNHGOGH 20 -Ph 0
1147 Ph n μ i 1-p「ri H o 1 11 「【i>in リリ η^·リ 「ιι ί 0.1147 Ph n μ i 1-p “ r i H o 1 11“ [i> in lily η ^ · li “ιι ί 0.
1148 Ph H H 卜 Pr H 2+5 - n - PrNHCOCH20 - Ph 21148 Ph HH Pr Pr H 2 + 5-n-PrNHCOCH 20- Ph 2
1149 Ph H H i-Pr H 2-n - PrNHGOCH20- 5+Ph 21149 Ph HH i-Pr H 2-n-PrNHGOCH 2 0-5 + Ph 2
1150 Ph H H 卜 Pr H 3-CH2OH- 4- MeO-Ph 21150 Ph HH Pr H 3-CH 2 OH- 4-MeO-Ph 2
1151 Ph H H 卜 Pr H 3-(1,2— CH(OH)CH2OH)-4_MeO - Ph 21151 Ph HH Pr H 3- (1,2-CH (OH) CH 2 OH) -4_MeO-Ph 2
1152 Ph H H 卜 Pr H 3-MeOCH2-4-MeO-Ph 21152 Ph HH Pr H 3-MeOCH 2 -4-MeO-Ph 2
1153 Ph H H i-Pr H 3 - MeS02NCH2— 4-MeO-Ph 21153 Ph HH i-Pr H 3 - MeS0 2 NCH 2 - 4-MeO-Ph 2
1154 Ph H H 卜 Pr H 3-MeS02N(Me)CH2 - 4- MeO-Ph 21154 Ph HH Bok Pr H 3-MeS0 2 N ( Me) CH 2 - 4- MeO-Ph 2
1155 Ph H H i-Pr H 3 - (4- CH2OH)pneny卜 4- MeO-Ph 21155 Ph HH i-Pr H 3-(4- CH 2 OH) pneny 4-MeO-Ph 2
1156 Ph H H 卜 Pr H 3-MeS02N(Me)CH2 - 4— Me - Ph 2 Ph 9丁〇 H HrP- - Ph 9κCT H Hpr 〇--1156 Ph HH Bok Pr H 3-MeS0 2 N ( Me) CH 2 - 4- Me - Ph 2 Ph 9cho H HrP--Ph 9κCT H Hpr 〇--
9PhICT P H〒 HPr力--9PhICT P H〒 HPr force--
-J -J »J -J -J »J
CD CXJ ~«J 0> CD CD CXJ ~ «J 0> CD
9PhhCTl He Hpr Mu--- 9PhhCTl He Hpr Mu ---
ェ ェ ェ ェ co co Co co
3" 3 3 "3
3" 3 "
Ks ) > Ks)>
O CD CO ~~JO CD CO ~~ J
CO OCO O
Meh4pFI-- 1 - 1Meh4pFI-- 1-1
〇 〇〇 〇
1 4Mph Mee 1Ill3 了 工 ェ ェ ェ 1 4Mph Mee 1Ill3 Completed
1 1 J ェ 工 1 1 J
C0 C C0 C
) > )>
表 1 table 1
合物例 A r R1 2 3 R 1、4~r 5 ri Compound example A r R1 2 3 R 1, 4 ~ r 5 ri
1 199 9- -p μ Π Π i 1-p「r 1 μ Γ 1 Ji *τ ΙΥΙσ r 1 1 0.1 199 9- -p μ Π Π i 1-p `` r 1 μ Γ 1 Ji * τ ΙΥΙσ r 1 1 0.
1200 9- -P 「h 1 1 μ Η 1 1 1 「 I μ π V,t5 * ϊ - £ 0 -1200 9- -P “h 1 1 μ Η 1 1 1” I μ π V, t5 * ϊ-£ 0-
1201 9- - P μ μ π i_pr ί U π ο m 0 l hi t π ί.1201 9--P μ μ π i_p r ί U π ο m 0 l hi t π ί.
1202 9- ト -P 「h【, μ π π i 1一 P「r 01202 9- G -P "h [, μπππi 1 P" r 0
Γ μ l * \J\ 「Π Γ μ l * \ J \ Π
1203 9- -ci- -Ph Η Η i-Pr Η 3— PhCH 0一 4一 Me一 Ph 2 1203 9- -ci- -Ph Η Η i-Pr Η 3— PhCH 0-1 4 1 Me-1 Ph 2
1204 9- _D rn U π U π 1 Γ u π MeU 4 Me rn ο1204 9- _D rn U π U π 1 Γ u π MeU 4 Me rn ο
1205 9- U U 1205 9- U U
""し rn π Π 1一 u π Θ Ulvie rrl "" Then rn π Π 1 1 u π Θ Ulvie rrl
1 206 9- U 1 206 9- U
""し rn π U "" Then rn π U
Π i r u π o Vinyl 4 βυ rn Π i r u π o Vinyl 4 βυ rn
1207 9- 1 1207 9- 1
-し ι- - nrn π 1 1 -Sh ι--nrn π 1 1
Π i-Hr 1 π 3 - 1一 PrO - 4一 Me— Ph 2 Π i-Hr 1 π 3-1-PrO-4-Me-Ph 2
1208 9- -ci- -Ph Η Η i-Pr Η 3 - F - 4一 OMe - Ph 21208 9- -ci- -Ph Η Η i-Pr Η 3-F-4 OMe-Ph 2
1209 9- -ci- -Ph Η Η i-Pr Η 3-C卜 4-OMe-Ph 21209 9- -ci- -Ph Η Η i-Pr Η 3-C 4-OMe-Ph 2
1210 9- -ci- -Ph Η Η i-Pr Η 3+4-OMe - Ph 21210 9- -ci- -Ph Η Η i-Pr Η 3 + 4-OMe-Ph 2
121 1 9- -Cl- -Ph Η Η 卜 Pr Η 3+4 - EtO-Ph 2121 1 9- -Cl- -Ph Η 卜 Pr Pr 3 + 4-EtO-Ph 2
1212 9- -ci- -Ph Η Η i-Pr Η 3-1-4-i-PrO-Ph 21212 9- -ci- -Ph Η Η i-Pr Η 3-1-4-i-PrO-Ph 2
1213 9- -ci- -Ph Η Η i-Pr Η 3 -卜 4-n- BuO- Ph 21213 9- -ci- -Ph Η Η i-Pr Η 3-Tri 4-n- BuO- Ph 2
1214 9- -Cl- -Ph Η Η i-Pr Η 3-l-4-PhCH20-Ph 21214 9- -Cl- -Ph Η Η i-Pr Η 3-l-4-PhCH 2 0-Ph 2
1215 9- -ci- -Ph Η Η 卜 Pr Η 3-Ph - 4-MeO - Ph 21215 9- -ci- -Ph Η 卜 Pr Pr Η 3-Ph-4-MeO-Ph 2
1216 9- -ci- -Ph Η Η 卜 Pr Η 3-C02Me-4-MeO-Ph 21216 9- -ci- -Ph Η 卜 Pr Pr Η 3-C0 2 Me-4-MeO-Ph 2
1217 9- -ci- -Ph Η Η i-Pr Η 3 - CONMe2- 4-MeO-Ph 2 表 1 2 1217 9- -ci- -Ph Η Η i-Pr Η 3-CONMe 2-4 -MeO-Ph 2 Table 1 2
化合物例 Ar R1 R2 R3 R4 R5 n=Compound example Ar R1 R2 R3 R4 R5 n =
1218 9-CI-Ph H H i-Pr H 3 - CONHCH2Ph - 4 - MeO - Ph 21218 9-CI-Ph HH i-Pr H 3-CONHCH 2 Ph-4-MeO-Ph 2
1219 9-C卜 Ph H H i-Pr H 3-l-4-n-PrNHCOCH20-Ph 21219 9-C Ph HH i-Pr H 3-l-4-n-PrNHCOCH 2 0-Ph 2
1220 9-CI-Ph H H i-Pr H 3+4-MeO - Ph 21220 9-CI-Ph H Hi i-Pr H 3 + 4-MeO-Ph 2
1221 9-CI-Ph H H i-Pr H 3-卜 4-PhCOO-Ph 21221 9-CI-Ph H Hi i-Pr H 3-pot 4-PhCOO-Ph 2
1222 9-CI-Ph H H i-Pr H 3-l-4-MeNHCOCH20-Ph 21222 9-CI-Ph HH i-Pr H 3-l-4-MeNHCOCH 2 0-Ph 2
1223 9-CI-Ph H H 卜 Pr H 3 - CH2OH- 4-MeO-Ph 21223 9-CI-Ph HH Pr H 3-CH 2 OH-4- MeO-Ph 2
1224 9-CI-Ph H H i-Pr H 3-MeOCH2-4-MeO-Ph 21224 9-CI-Ph HH i-Pr H 3-MeOCH 2 -4-MeO-Ph 2
1225 9-CI-Ph H H i-Pr H 3-C卜 4 - Cト Ph 21225 9-CI-Ph H Hi i-Pr H 3-C 4-C Ph 2
1226 Q_Q|_p H H i-Pr H 3 - F - 4 - OMe-Ph 21226 Q_Q | _p H H i-Pr H 3-F-4-OMe-Ph 2
1227 9-F-Ph H H 卜 Pr H Ph 21227 9-F-Ph H H Pr Pr H Ph 2
1228 9-F-Ph H H i-Pr H 4-Me-Ph 21228 9-F-Ph H Hi i-Pr H 4-Me-Ph 2
1229 9-F-Ph H H i-Pr H 3 - Ph - Ph 21229 9-F-Ph H Hi i-Pr H 3-Ph-Ph 2
1230 9-F-Ph H H i-Pr H 4 - C卜 Ph 21230 9-F-Ph H Hi-Pr H 4-C Ph 2
1231 9-F-Ph H H i-Pr H 3-F-Ph 21231 9-F-Ph H Hi i-Pr H 3-F-Ph 2
1232 9-F-Ph H H 卜 Pr H 4 - MeO - Ph 21232 9-F-Ph H H H Pr H 4-MeO-Ph 2
1233 9-F-Ph H H i-Pr H 4 - PhC2H40 - Ph 21233 9-F-Ph HH i-Pr H 4-PhC 2 H 40- Ph 2
1234 9-F-Ph H H 卜 Pr H 4-EtNHCOCH20-Ph 21234 9-F-Ph HH Pr H 4-EtNHCOCH 2 0-Ph 2
1235 9-F-Ph H H 卜 Pr H 3-n-PrNHCOCH20-Ph 21235 9-F-Ph HH Pr H 3-n-PrNHCOCH 20 -Ph 2
1236 9-F-Ph H H i-Pr H 4一 n - PrNHCOCH(Me)0 - Ph 2 表 1 3 1236 9-F-Ph HH i-Pr H 4 1 n-PrNHCOCH (Me) 0-Ph 2 Table 13
物例 Ar P?ク ΓνΟ n— Object example Ar P? ク ΓνΟ n—
1237 9-F- -Ph H H i一 Pr H 4 *r-M viec?2NCOCHo 2O^-^- Ph 2 1237 9-F- -Ph H Hi i Pr H 4 * r-M viec? 2NCOCHo 2O ^-^-Ph 2
1238 9-F- -Ph H H i-Pr H 4十 BuO GCH 0 - -Ph 21238 9-F- -Ph H Hi-Pr H 40 BuO GCH 0--Ph 2
1239 9-F- -Ph H H i-Pr H 3— MeOCH Ph 21239 9-F- -Ph H H i-Pr H 3— MeOCH Ph 2
1240 9-F- -Ph H H i-Pr H 4-MeOCH -Ph 21240 9-F- -Ph H H i-Pr H 4-MeOCH -Ph 2
1241 9-F- -Ph H H i-Pr H 4-i-PrOCH -Ph 21241 9-F- -Ph H H i-Pr H 4-i-PrOCH -Ph 2
1242 9-F- -Ph H H i-Pr H 4— EtG G— Ph 21242 9-F- -Ph H Hi i-Pr H 4— EtG G— Ph 2
1243 9 - F- -Ph H H i-Pr H 4-HoNOC-Ph 21243 9-F- -Ph H H i-Pr H 4-HoNOC-Ph 2
1244 9-F- -rh Π Π r Π 4一 Me! Hりし一 Hi I1244 9-F- -rh Π Π r Π 4 One Me! H Rishi one Hi I
1245 9-F- rn Π u Π 1 rr Π 4"MenN"rh L1245 9-F- rn Π u Π 1 rr Π 4 "MenN" rh L
1246 9-F- -Ph H H 1一 Pr H 4一 Me2N - Ph 21246 9-F- -Ph HH 1-1 Pr H 4-1 Me 2 N-Ph 2
1247 9-F- -Ph H H i-Pr H 4-PhCONH-Ph 21247 9-F- -Ph H H i-Pr H 4-PhCONH-Ph 2
1248 9-F- -Ph H H i-Pr H 3 - Me - 4 - Me-Ph 21248 9-F- -Ph H H i-Pr H 3-Me-4-Me-Ph 2
1249 9-F- -Ph H H i-Pr H 3 - F - 4-Me - Ph 21249 9-F- -Ph H H i-Pr H 3-F-4-Me-Ph 2
1250 9 - F - -Ph H H 卜 Pr H 3-Cト 4 - Me-Ph 21250 9-F--Ph H H Pr Pr H 3-C 4 4-Me-Ph 2
1251 9-F- -Ph H H i-Pr H 3-Me - 4 - C卜 Ph 21251 9-F- -Ph H Hi-Pr H 3-Me-4-Ct Ph 2
1252 9 - F - -Ph H H i-Pr H 3 - F - 4 - Cト Ph 21252 9-F--Ph H H i-Pr H 3-F-4-C G Ph 2
1253 9-F- -Ph H H i-Pr H 3-CM-C卜 Ph 21253 9-F- -Ph H Hi-Pr H 3-CM-C Ph 2
1254 9 - F- ■Ph H H 卜 Pr H 3-PhCH20-4-Me- -Ph 21254 9-F- Ph HH Pr H 3-PhCH 2 0-4-Me- -Ph 2
1255 9-F- ■Ph H H i-Pr H 3-MeO— 4-Me-Ph 2 表 1255 9-F- ■ Ph HH i-Pr H 3-MeO— 4-Me-Ph 2 table
合物例 A f D ΓΛ11 Γ *τ 1 Compound example A f D ΓΛ11 Γ * τ 1
1256 9- Γ -p「 U u1256 9- Γ -p `` U u
π 1 1 1 I— P ΙΓ r U π 1 1 1 I— P ΙΓ r U
Π IVI6 UIVI6 ΓΠ Π IVI6 UIVI6 ΓΠ
1 2 9- Γ u 1 2 9- Γ u
57 1 1 u Π p U 57 1 1 u Π p U
1 _ Γ^ Π v inyl ^ ινιβυ rn L 1 _ Γ ^ Π v inyl ^ ινιβυ rn L
1258 C" u 1258 C "u
9- Γ • D rn Π Π 1 Mr Π 3- PrO-4-Me-Ph L 9- Γ • D rn Π Π 1 Mr Π 3- PrO-4-Me-Ph L
1259 9- u LI 1259 9- u LI
1 ^ rn Π U Π 1 T Π 3 - F-4-OMe - Ph 1 ^ rn Π U Π 1 T Π 3-F-4-OMe-Ph
1260 9- Γ -P 「h u U1260 9- Γ -P `` hu u
n 1 1 Π 1 「 U n 1 1 Π 1 “U
i 1 1 3 - C卜 4-OMe-Ph L i 1 1 3-C 4-OMe-Ph L
1261 9- - - _p u u u 1261 9---_p u u u
ir i i Π Π 1 T Π 3-1-4-MeO-Ph Δ ir i i Π Π 1 T Π 3-1-4-MeO-Ph Δ
1262 9- 1 ^ n u ■ D 1262 9- 1 ^ n u ■ D
Π u « Π u «
Π 1 Kr Π 3-1-4-EtO-Ph Π 1 Kr Π 3-1-4-EtO-Ph
1263 9- 1 ^ n u Π u Π 1 D K^r- Π 3- 4- i-PrO-Ph L 1263 9- 1 ^ n u Π u Π 1 D K ^ r- Π 3- 4- i-PrO-Ph L
1264 9-十■rh H H i-Pr Π 3-1-4-n-BuO-Ph 21264 9-10 ■ rh H Hi-Pr Π 3-1-4-n-BuO-Ph 2
1265 9- u Π 1 Mr n 3-l-4-PhCH20-Ph 1265 9- u Π 1 Mr n 3-l-4-PhCH 2 0-Ph
1266 9- -F- -Ph H H i-Pr H 3 - Ph—4 - MeO - Ph 2 1266 9- -F- -Ph H H i-Pr H 3-Ph—4-MeO-Ph 2
1267 9- -F- -Ph H H i-Pr H 3 - C02Me - 4—MeO - Ph 21267 9- -F- -Ph HH i-Pr H 3-C0 2 Me-4—MeO-Ph 2
1268 9- -F- -Ph H H i-Pr H 3 - CONMe2 - 4 - MeO - Ph 21268 9- -F- -Ph HH i-Pr H 3-CONMe 2-4- MeO-Ph 2
1269 9- -F- •Ph H H Pr H 3-CONHCH2Ph-4-MeO -Ph 21269 9- -F-Ph HH Pr H 3-CONHCH 2 Ph-4-MeO -Ph 2
1270 9- -F- -Ph H H i-Pr H 3-1-4-PhCOO-Ph 21270 9- -F- -Ph H H i-Pr H 3-1-4-PhCOO-Ph 2
1271 9- ■F- -Ph H H i-Pr H 3-l-4-MeNHCOCH20- - Ph 21271 9- ■ F- -Ph HH i-Pr H 3-l-4-MeNHCOCH 20 --Ph 2
1272 9- -Ph H H i-Pr H 3+4 - n - PrNHCOGH20 -Ph 21272 9- -Ph HH i-Pr H 3 + 4-n-PrNHCOGH 20 -Ph 2
1273 9- ■F- -Ph H H Pr H 3 - CH2OH - 4- MeO - Ph 21273 9- ■ F- -Ph HH Pr H 3-CH 2 OH-4-MeO-Ph 2
1274 9- ■Ph H H Pr H 3-MeOCH2-4-MeO- Ph 2 表 1 5 1274 9- ■ Ph HH Pr H 3-MeOCH 2 -4-MeO- Ph 2 Table 15
化合物例 Ar R1 R2 R3 R4 R5 n=Compound example Ar R1 R2 R3 R4 R5 n =
1275 7-l-8-n-PrNHCOCH20-Ph H H i-Pr H Ph 2 1275 7-l-8-n-PrNHCOCH 2 0-Ph HH i-Pr H Ph 2
1277 6一 MeO一 9 Ph H H i-Pr H Ph 2 1277 6 1 MeO 1 9 Ph H H i-Pr H Ph 2
1278 6-1-9-MeO-Ph H H i-Pr H Ph 21278 6-1-9-MeO-Ph H H i-Pr H Ph 2
1279 3 4-Pvridvl- H H 卜 Pr H Ph 01279 3 4-Pvridvl- H H Pr Pr H Ph 0
1280 34-Pvridvl- H H i-Pr H Ph 21280 34-Pvridvl- H H i-Pr H Ph 2
1281 3,4-Pyridyl- H H 卜 Pr H 4-PhCH20- -Ph 01281 3,4-Pyridyl- HH Pr H 4-PhCH 2 0- -Ph 0
1282 o, rynoyi U 1282 o, rynoyi U
Π H i-Pr H 3-F - 4 - Me. -Ph 2 Π H i-Pr H 3-F-4-Me.-Ph 2
1283 3,4-Pyridyl- H H 卜 Pr H 3-CI-4-Me -Ph 21283 3,4-Pyridyl- H H pr Pr H 3-CI-4-Me -Ph 2
1284 3,4-Pyridyl- H H i-Pr H 3-Me- 4-CI -Ph 21284 3,4-Pyridyl- H Hi i-Pr H 3-Me- 4-CI -Ph 2
1285 3,4-Pyridyl- H H 卜 Pr H 3-F-4-CI- Ph 21285 3,4-Pyridyl- H H Pr H 3-F-4-CI- Ph 2
1286 3,4-Pyridyl- H H i-Pr H 3-CI-4-CI- -Ph 21286 3,4-Pyridyl- H Hi-Pr H 3-CI-4-CI- -Ph 2
1287 3,4-Pyridyl- H H i-Pr H 4-P CH20- -Ph 21287 3,4-Pyridyl- HH i-Pr H 4-P CH 2 0- -Ph 2
1288 2,3-Pyridyl- H H i-Pr H Ph 01288 2,3-Pyridyl- H H i-Pr H Ph 0
1289 2,3-Pyridyl- H H 卜 Pr H Ph 21289 2,3-Pyridyl- H H pr Pr H Ph 2
1290 2,3-Pyridyl- H H i-Pr H 4-CI-Ph 21290 2,3-Pyridyl- H Hi i-Pr H 4-CI-Ph 2
1291 45— Pyridyl— H H 卜 Pr H Ph 01291 45— Pyridyl— H H Pr Pr H Ph 0
1292 45— Pyridyl— H H 卜 Pr H Ph 21292 45— Pyridyl— H H Pr Pr H Ph 2
1293 4,5-Pyridyl- H H i-Pr H 4-F-Ph 2 これら化合物の内、 好適な化合物としては、 例えば 1009, 1011, 11293 4,5-Pyridyl- HH i-Pr H 4-F-Ph 2 Among these compounds, suitable compounds include, for example, 1009, 1011, 1
016, 1018, 1023, 1026, 1027, 1029, 1030, 10 32, 1035, 1038, 1040, 1043, 1044, 1046, 104 8, 1049, 1051, 1052, 1054, 1056, 1063, 1072 , 1073, 1084, 1087, 1090, 1091, 1092, 1093,016, 1018, 1023, 1026, 1027, 1029, 1030, 1032, 1035, 1038, 1040, 1043, 1044, 1046, 1048, 1049, 1051, 1052, 1054, 1056, 1063, 1072, 1073, 1084, 1087, 1090, 1091, 1092, 1093,
1094, 1095, 1096, 1100, 1101, 1104, 1108, 1 111, 1112, 1113, 1116, 1119, 1120, 1121, 11 22, 1124, 1126, 1 127, 1131, 1135, 1 136, 114 2, 1143, 1147, 1158, 1159, 1162, 1170, 1171 , 1173, 1174, 1175, 1178, 1180, 1182, 1184,1094, 1095, 1096, 1100, 1101, 1104, 1108, 1 111, 1112, 1113, 1116, 1119, 1120, 1121, 11 22, 1124, 1126, 1 127, 1131, 1135, 1 136, 114 2, 1143 , 1147, 1158, 1159, 1162, 1170, 1171, 1173, 1174, 1175, 1178, 1180, 1182, 1184,
1190, 1197, 1199, 1200, 1201, 1202, 1208, 11190, 1197, 1199, 1200, 1201, 1202, 1208, 1
210, 1213, 1219, 1220, 1225, 1227, 1230, 12 32, 1237, 1241, 1242, 1248, 1251, 1253, 125 9, 1260, 1261, 1266, 1282, 1283, 1286, 1290 , 1293等が挙げられ、 中でも 1011, 1016, 1018, 1023, 1210, 1213, 1219, 1220, 1225, 1227, 1230, 1232, 1237, 1241, 1242, 1248, 1251, 1253, 125 9, 1260, 1261, 1266, 1282, 1283, 1286, 1290, 1293, etc. Of which 1011, 1016, 1018, 1023, 1
026, 1027, 1029, 1030, 1032, 1035, 1038, 10 40, 1043, 1044, 1046, 1048, 1049, 1051, 105 2, 1054, 1063, 1072, 1073, 1084, 1087, 1090 , 1091, 1092, 1093, 1094, 1095, 1096, 1100, 1101, 1104, 1108, 1111, 1112, 11 13, 1116, 1026, 1027, 1029, 1030, 1032, 1035, 1038, 1040, 1043, 1044, 1046, 1048, 1049, 1051, 105 2, 1054, 1063, 1072, 1073, 1084, 1087, 1090, 1091, 1092, 1093, 1094, 1095, 1096, 1100, 1101, 1104, 1108, 1111, 1112, 11 13, 1116, 1
119, 1 120, 1 121, 1 122, 1124, 1126, 1127, 11 31, 1135, 1136, 1 147, 1158, 1 159, 1 162, 117 5, 1178, 1180, 1 199, 1200, 1201, 1202, 1208 , 1210, 1219, 1220, 1227, 1230, 1232, 1248, 1251, 1253, 1261, 1266, 1282, 1283, 1286, 1119, 1 120, 1 121, 1 122, 1124, 1126, 1127, 11 31, 1135, 1136, 1 147, 1158, 1 159, 1 162, 117 5, 1178, 1180, 1 199, 1200, 1201, 1202 , 1208, 1210, 1219, 1220, 1227, 1230, 1232, 1248, 1251, 1253, 1261, 1266, 1282, 1283, 1286, 1
290, 1293の化合物等が好ましい。 Compounds 290 and 1293 are preferred.
次に本発明の一般式 [I]で表される化合物の製造方法について説明する 一般式 [I] で表される化合物は、 以下の製造法 A— Hで製造することができ る。 製造法 A Next, a method for producing the compound represented by the general formula [I] of the present invention will be described. The compound represented by the general formula [I] can be produced by the following production methods AH. Manufacturing method A
(第 1工程) (First step)
一般式 [Π] General formula [Π]
[式中、 A rは、 前記の意味を有する] で表される化合物を一般式 [III] [Wherein, Ar has the meaning described above], a compound represented by the general formula [III]
R 5— C N [III] R 5 — CN [III]
[式中、 R 5は、 前記の意味を有する] で表される化合物と反応させることによ り一般式 [ I V] [Wherein R 5 has the meaning described above] to obtain a compound represented by the general formula [IV]
[式中、 R 5及び A rは、 前記の意味を有する]で表される化合物を合成すること ができる。 [Wherein, R 5 and Ar have the same meanings as described above] can be synthesized.
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は脱水した不活性有機溶媒中、 適宜添加剤の存在下で適宜 塩基を用いて、 一 1 0 0 °Cから溶媒の沸点温度、 好ましくは一 7 8〜3 0 °C、 0 . 5〜9 6時間、 好ましくは 1 2〜2 4時間反応させることにより一般式 [II] で表される化合物をリチォ化して、 次いで一般式 [III] で表されるァリールニト リルの不活性溶媒溶液を一 1 0 0 °Cから溶媒の沸点温度、 好ましくは— 7 8〜 3 O :で滴下し、 0 . 5〜9 6時間、 好ましくは 1 2〜2 4時間反応させる。 反応で使用される不活性有機溶媒としては、 反応に悪影響を及ぼさないもので あれば、 特に限定はされないが、 具体的には例えばへキサン、 シクロへキサン、 ペンタン、 エーテル、 1, 4—ジォキサン、 テトラヒドロフラン、 テトラヒドロ ピラン等が挙げられる、 中でもエーテル、 テトラヒドロフラン、 シクロへキサン 等が好適である。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions. Usually, the reaction is carried out by using a base in a dehydrated inert organic solvent in the presence of a suitable additive. The compound represented by the general formula [II] can be obtained by reacting the solvent at a temperature of from 0 ° C to the boiling point of the solvent, preferably from 17 to 30 ° C, for from 0.5 to 96 hours, preferably from 12 to 24 hours. Then, a solution of arylnitrile represented by the general formula [III] in an inert solvent is added dropwise at a temperature of 100 ° C. to the boiling point of the solvent, preferably −78 to 3O: 0.5. The reaction is carried out for up to 96 hours, preferably 12 to 24 hours. The inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include hexane, cyclohexane, pentane, ether, 1,4-dioxane , Tetrahydrofuran, tetrahydropyran and the like. Among them, ether, tetrahydrofuran, cyclohexane and the like are preferable.
反応で使用される添加剤としては、 N,N,N',N '— ン TMEDA、 へキサメチルホスホラミン HMPA、 塩化リチウム等が挙げられ、 中でも Ν,Ν,Ν',Ν'—テトラメチルエチレンジァミンが好適である。 Additives used in the reaction include N, N, N ', N' And TMEDA, hexamethylphosphoramine HMPA, lithium chloride and the like. Among them, Ν, Ν, Ν ′, Ν′-tetramethylethylenediamine is preferable.
反応で使用される塩基としては、 例えば η—ブチルリチウム、 s e c—ブチル リチウム、 t一ブチルリチウム、 フエニルリチウム、 メチルリチウム等が挙げら れ、 中でも n—ブチルリチウム、 s e c—ブチルリチウムが好適である。 Examples of the base used in the reaction include η-butyllithium, sec-butyllithium, t-butyllithium, phenyllithium, methyllithium and the like. Among them, n-butyllithium and sec-butyllithium are preferable. is there.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常一般式 [I I] で表される化合物に対して 2〜10等量 、 好ましくは 2〜3等量の塩基、 1〜10等量、 好ましくは 1. 5〜2等量の上 記一般式 [III] で表されるァリール二トリルを使用する。 The amount of the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions. And 1 to 10 equivalents, preferably 1.5 to 2 equivalents, of arylyl nitrile represented by the above general formula [III] is used.
(第 2工程) (2nd step)
次に一般式 [ I V] Next, the general formula [IV]
[式中、 R5、 A rは前記の意味を有する] で表される化合物と一般式 [V] [Wherein R 5 and Ar have the above-mentioned meanings] and a compound represented by the general formula [V]
[式中、 R R2は、 水素原子又は 一 C3アルキル基、 R3は水素原子又 C 一 C6アルキル基 (但し、 n—ブチル基除く) ] で表される化合物を例えば 2, 6ールチジンのような有機溶媒中でを反応させることにより一般式 [VI] [Wherein, RR 2 is a hydrogen atom or a C 3 alkyl group, and R 3 is a hydrogen atom or a C 1 C 6 alkyl group (however, excluding n-butyl group)]. By reacting in an organic solvent such as
[式中、 R R 2、 R 3、 R 5及び A rは前記の意味を有する] で表される化合物 を合成できる。 [Wherein, RR 2 , R 3 , R 5 and Ar have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によつて適宜増減することが できるが、 通常、 反応には、 例えばルチジンのような有機溶媒中、 上記一般式 [ I V] で表される化合物と上記一般式 [V] で表される化合物とを、 室温で 0 . 5〜9 6時間、 好ましくは 3〜 2 4時間反応させる。 The reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions. Usually, for example, the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [IV]. The compound represented by the above general formula [V] is reacted at room temperature for 0.5 to 96 hours, preferably 3 to 24 hours.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1 , 2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 N, N—ジメチルホルム アミド、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニトリル、 メチルァ ルコール、 エチルアルコール、 ベンゼン、 キシレン、 ピリジン、 ルチジン、 トル ェン、 キシレン、 1 > 4一ジォキサン、 テトラヒドロフラン等が挙げられ、 中で もメチルアルコール、 エチルアルコール、 ピリジン、 ルチジン、 N, N—ジメチ ルホルムアミド、 トルエン、 キシレン等が好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and tetrachloroethane. Carbon chloride, N, N-dimethylformamide, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1> 4-dioxane, tetrahydrofuran, etc. Among them, methyl alcohol, ethyl alcohol, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferable.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [ I V] で表される化合物に対して 1 . 2 一 1 0等量、 好ましくは 1 . 2— 2等量の上記一般式 [V] で表されるアジリジ ンを使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 with respect to the compound represented by the above general formula [IV]. 1.2—Use 2 equivalents of aziridin represented by the above general formula [V].
(第 3工程) (3rd step)
次に、 一般式 [V I ] Next, the general formula [V I]
[式中、 R 1 R 2、 R 3、 1^ 5及び八]:は、 前記の意味を有する] で表される化 合物を、 例えば 2、 6 -ルチジンの様な適当な溶媒中で、 適宜酸の存在下で共沸 蒸留、 または例えばモレキュラーシ一ブスのような乾燥剤の存在下、 加熱還流さ せることにより、 一般式 [V I I ] [Wherein, R 1 R 2 , R 3 , 1 ^ 5 and 8] has the above-mentioned meaning] in a suitable solvent such as 2,6-lutidine. Azeotropic distillation, optionally in the presence of an acid, or heating to reflux in the presence of a desiccant, for example, molecular sieves. The general formula [VII]
[式中、 R 1 R 2、 R 3、 R 5及び A rは、 前記の意味を有する] で表される化合 物を合成できる。 [Wherein, R 1 R 2 , R 3 , R 5 and Ar have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常反応は、 例えばモレキュラーシーブスのような乾燥剤の存在下、 例えば 2, 6—ルチジンの様な適当な溶媒中で、 例えば塩酸の様な適当な酸の存 在下で 5〜9 6時間、 好ましくは 1 2〜2 4時間還流させることにより、 反応は 完結する。 The reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but usually the reaction is carried out in the presence of a desiccant such as molecular sieves, and in an appropriate solvent such as 2,6-lutidine. The reaction is completed by refluxing for 5 to 96 hours, preferably 12 to 24 hours, in the presence of a suitable acid such as hydrochloric acid.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1 , 2—ジクロロェタン、 トリクロロェタン、 N, N—ジメチルホルムアミド、 無水 酢酸、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニトリル、 メチルアル コール、 エチルアルコール、 n—プロパノール、 ベンゼン、 キシレン、 ピリジン 、 ルチジン、 トルエン、 キシレン、 1, 4—ジォキサン、 テトラヒドロフラン等 が挙げられ、 好適な反応温度の確保の点から、 特にピリジン、 ルチジン、 N, N —ジメチルホルムアミド、 トルエン、 キシレン等が好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and N , N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, n-propanol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran, etc. From the viewpoint of securing a suitable reaction temperature, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are particularly preferable.
反応で使用される酸としては、 例えば塩酸、 硝酸、 臭ィ匕水素酸、 硫酸、 フッ化 水素酸、 過塩素酸等の無機酸;例えばトリフルォロホウ酸、 四塩化チタン、 塩ィ匕 亜鉛等のルイス酸;例えば P—トルエンスルホン酸、 トリフルォロメタンスルホ ン酸、 メタンスルホン酸等のスルホン酸;蟻酸、 トリフルォロ酢酸、 酢酸等の有 機酸等が挙げられ、 中でも塩酸、 酢酸又は P—トルエンスルホン酸が好適である 反応で使用される乾燥剤しては、 例えばモレキュラーシーブス、 硫酸マグネシ ゥム、 硫酸ナトリウムが挙げられ、 中でもモレキュラーシーブスが好適である。 反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [V I] で表される化合物に対して 1. 2 一 10等量、 好ましくは 1. 2〜2等量の乾燥剤、 1〜50等量、 好ましくは 3 〜 5等量の酸を使用する。 該酸、 該乾燥剤剤は、 一種又はそれ以上適宜組合せて 使用することもできる。 Examples of the acid used in the reaction include inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid, and perchloric acid; for example, Lewis such as trifluoroboric acid, titanium tetrachloride, and zinc chloride. Acids; for example, sulfonic acids such as P-toluenesulfonic acid, trifluoromethanesulfonic acid, and methanesulfonic acid; and organic acids such as formic acid, trifluoroacetic acid, and acetic acid, and among them, hydrochloric acid, acetic acid, and P-toluenesulfonic acid Preferred examples of the desiccant used in the reaction include molecular sieves, magnesium sulfate, and sodium sulfate. Among them, molecular sieves are preferred. The amount of the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but usually, 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [VI] is used. Use 2 to 2 equivalents of desiccant, 1 to 50 equivalents, preferably 3 to 5 equivalents of acid. The acid and the desiccant may be used alone or in appropriate combination of two or more.
(第 4工程) (4th step)
次に一般式 [V I I] Next, the general formula [V I I]
[式中、 R1 R2、 R3、 R5及び Arは、 前記の意味を有する] で表される化合 物を、 適当な有機溶媒中で、 酸化剤と反応させることにより一般式 [V I I I] Wherein R 1 R 2 , R 3 , R 5 and Ar have the meaning described above, in an appropriate organic solvent with an oxidizing agent to give a compound of the general formula [VIII ]
[式中、 R R2、 R3、 R5及び Arは、 前記の意味を有する] で表される化合 物を合成できる。 [Wherein, RR 2 , R 3 , R 5 and Ar have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は例えばトリフルォロ酢酸の様な適当な有機酸溶媒中、 例 えば過酸化水素のような適当な酸化剤の存在下で 5〜 96時間、 好ましくは 12 〜24時間攪拌させることにより、 反応は完結する。 または、 例えば塩化メチレ ンの様な適当な不活性有機溶媒中、 例えばメタク口口過安息香酸のような適当な 酸化剤の存在下で 5〜 96時間、 好ましくは 12〜 24時間攪拌させることによ り、 反応は完結する。 反応で使用される不活性有機溶媒としては、 反応に悪影響を及ぼさないもので あれば、 特に限定はされないが、 具体的には例えば塩ィ匕メチレン、 クロ口ホルム 、 1, 2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 酢酸ェチルエステ ル、 酢酸メチルエステル、 ァセトニトリル、 メチルアルコール、 ェチルアルコー ル、 ベンゼン、 キシレン、 トルエン、 キシレン、 エーテル、 1, 4一ジォキサン 、 テトラヒドロフラン、 テトラヒドロピラン等が挙げられ、 中でも塩化メチレン 、 クロ口ホルム、 1 , 2ージクロロェタン、 トリクロロェタン、 トルエン、 キシ レン等が好適である。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.In general, the reaction is carried out in a suitable organic acid solvent such as trifluoroacetic acid, for example, a suitable oxidizing agent such as hydrogen peroxide. The reaction is completed by stirring for 5 to 96 hours, preferably 12 to 24 hours, in the presence of Alternatively, stirring in a suitable inert organic solvent, such as, for example, methylene chloride, in the presence of a suitable oxidizing agent, such as, for example, methacrylic acid mouth perbenzoic acid, for 5 to 96 hours, preferably 12 to 24 hours. Thus, the reaction is completed. The inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include methylene chloride, methylene chloride, 1,2-dichloroethane, and trichloroethane. Ethane, carbon tetrachloride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, toluene, xylene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran, etc. Preferred are methylene, chloroform, 1,2-dichloroethane, trichloroethane, toluene, xylene and the like.
反応で使用される酸溶媒としては、 例えば蟻酸、 トリフルォロ酢酸、 酢酸等の 有機酸等が挙げられ、 中でもトリフルォロ酢酸が好適である。 Examples of the acid solvent used in the reaction include organic acids such as formic acid, trifluoroacetic acid and acetic acid, among which trifluoroacetic acid is preferred.
反応で使用される酸化剤としては、 反応に悪影響を及ぼさないものであれば、 特に限定はされないが、 具体的には例えば過酸化水素、 過酢酸、 トリフルォロ過 酢酸、 メタクロル過安息香酸、 ォキソン、 クロム酸、 過マンガンサン等が挙げら れ、 中でも過酸化水素、 メタクロル過安息香酸、 等が好適である。 The oxidizing agent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.Specifically, for example, hydrogen peroxide, peracetic acid, trifluoroperacetic acid, methachloroperbenzoic acid, oxone, Examples thereof include chromic acid and permanganese sun. Among them, hydrogen peroxide, metachloroperbenzoic acid, and the like are preferable.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [V I I ] で表される化合物に対して 1 . 2〜1 0等量、 好ましくは 1 . 2〜 2等量の酸化剤を使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [VII]. 1.2 Use 2 to 2 equivalents of oxidizing agent.
(第 5工程) (Fifth step)
次に一般式 [V I I I ] Next, the general formula [V I I I]
[式中、 R 1 , R 2、 R 3、 5及び八]:は、 前記の意味を有する] で表される化合 物を、 例えばテトラヒドロフラン (TH F ) のような適当な有機溶媒中、 適宜還 元剤を用いて還元するか、 または例えば 1 0 %パラジウム炭素のような触媒を用 いた接触水素化、 あるいは例えば塩酸などの酸触媒の存在下、 塩化亜鉛と反応さ せることにより、 一般式 [ I ] [Wherein, R 1 , R 2 , R 3 , 5 and 8] have the above-mentioned meanings] in a suitable organic solvent such as tetrahydrofuran (THF). Reduction using a reducing agent or catalytic hydrogenation using a catalyst such as 10% palladium on carbon, or reaction with zinc chloride in the presence of an acid catalyst such as hydrochloric acid The general formula [I]
[式中、 R 1 , R 2、 R 3、 R 5及び A rは、 前記の意味を有する] で表される化合 物が合成できる。 [Wherein, R 1 , R 2 , R 3 , R 5 and Ar have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 応は、 例えば TH Fの様な適当な有機酸溶媒中、 例えばポランのよう な適当な還元剤を用いて 5〜 9 6時間、 好ましくは 1 2〜2 4時間攪拌させるこ とにより、 反応は完結する。 または、 例えばメチルアルコールの様な適当な有機 溶媒中、 例えば 1 0 %パラジウム炭素のような適当なパラジウム触媒の存在下で 5〜9 6時間、 好ましくは 1 2〜2 4時間攪拌させることにより、 反応は完結す る。 または、 適宜酸あるいはアルカリ溶液中で金属あるいは金属塩と 5〜 9 6時 間、 好ましくは 1 2〜2 4時間攪拌させることにより、 反応は完結する。 The reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions. However, the reaction can be carried out in a suitable organic acid solvent such as THF, for example, using a suitable reducing agent such as porane. The reaction is completed by stirring for up to 96 hours, preferably 12 to 24 hours. Alternatively, by stirring in a suitable organic solvent such as methyl alcohol in the presence of a suitable palladium catalyst such as 10% palladium on carbon for 5 to 96 hours, preferably 12 to 24 hours, The reaction is complete. Alternatively, the reaction is completed by appropriately stirring with a metal or a metal salt for 5 to 96 hours, preferably 12 to 24 hours in an acid or alkali solution.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1, 2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 酢酸、 酢酸ェチルエステ ル、 酢酸メチルエステル、 ァセトニトリル、 メチルアルコール、 ェチルアルコー ル、 ベンゼン、 キシレン、 トルエン、 キシレン、 エーテル、 1 , 4一ジォキサン 、 テトラヒドロフラン、 テトラヒドロピラン等が挙げられ、 中でも塩化メチレン 、 クロ口ホルム、 1, 2—ジクロロェタン、 トリクロロェタン、 トルエン、 キシ レン等が好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and tetrachloroethane. Carbon chloride, acetic acid, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, toluene, xylene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran, etc., among which methylene chloride, Preferred are chloroform, 1,2-dichloroethane, trichloroethane, toluene, xylene and the like.
反応で使用される還元剤としては、 例えば水素化ホウ素リチウム、 水素化ホウ 素ナトリウム、 シァノトリヒドロホウ酸ナトリウム、 ポランメチルスルフイド錯 体、 ポラン TH F錯体、 水素化アルミニウムリチウムが挙げられ、 中でもポラン メチルスルフィド錯体、 ポラン TH F錯体が好適である。 反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [V I I I] で表される化合物に対して 1 . 2〜10等量、 好ましくは 1. 2〜 2等量の還元剤を使用する。 Examples of the reducing agent used in the reaction include lithium borohydride, sodium borohydride, sodium cyanotrihydroborate, porane methyl sulfide complex, porane THF complex, and lithium aluminum hydride. Among them, a porane methylsulfide complex and a porane THF complex are preferable. The amount of the reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [VIII]. Use 2 to 2 equivalents of reducing agent.
接触水素化反応で使用される触媒としては、 例えばパラジウム活性炭、 ラネー ニッケル、 酸化白金等が挙げられ、 中でも 10%パラジウム活性炭が好適である 反応で使用される金属としては、 例えば亜鉛、 アルミニウム、 イリジウム、 ク ロム、 チタン、 スズ、 セリウム、 鉄、 銅、 バナジウム、 マグネシウム等が挙げら れ、 中でも亜鉛、 マグネシウムが好適である。 Catalysts used in the catalytic hydrogenation reaction include, for example, palladium activated carbon, Raney nickel, platinum oxide, etc., and among them, 10% palladium activated carbon is preferred.Metal used in the reaction is, for example, zinc, aluminum, iridium. , Chromium, titanium, tin, cerium, iron, copper, vanadium, magnesium, etc., of which zinc and magnesium are preferred.
なお、 「第 4工程」 と 「第 5工程」 の製造工程の順番を逆にしても同様に上記 一般式 [I] で表される化合物に変換可能である。 It should be noted that even if the order of the production steps of “fourth step” and “fifth step” is reversed, the compound can be similarly converted to the compound represented by the above general formula [I].
反応終了後、 常法で生成物を精製することにより、 上記一般式 [I] で表され る化合物を得ることができる。 反応液から一般式 [I] で表される化合物又はそ の塩の単離精製は、 溶媒抽出、 再結晶、 クロマトグラフィー等公知の分離手段に より行うことができる。 製造法 B After completion of the reaction, the product represented by the above general formula [I] can be obtained by purifying the product by a conventional method. The compound represented by the general formula [I] or a salt thereof can be isolated and purified from the reaction solution by a known separation means such as solvent extraction, recrystallization, and chromatography. Manufacturing method B
一般式 [IV] で表される化合物は、 一般式 [I X] The compound represented by the general formula [IV] is represented by the general formula [IX]
[式中、 5及び八1"は、 前記の意味を有する] で表される化合物からも合成する ことができる。 例えば上記一般式 [I X] で表される化合物と適切な不活性有機 溶媒中、 塩化ジメチルチオ力ルバモイルを適宜塩基で反応し調整した一般式 [X[Wherein 5 and 8 1 "have the meanings described above." For example, the compound represented by the above general formula [IX] and a suitable inert organic solvent may be used. General formula [X
] [ X ] ] [X]
[式中、 Rはメチル基などのアルキル基を示し、 R 5及び A rは、 前記の意味を有 する]で表される化合物を非極性溶媒中で加熱することにより、 一般式 [X I ] [Wherein, R represents an alkyl group such as a methyl group, and R 5 and Ar have the above-mentioned meanings] by heating the compound represented by the formula [XI]
[式中、 R 5、 1"及び尺は、 前期の意味を有する]で表される化合物を合成する ことができる。 [Wherein, R 5 , 1 ″ and the scale have the same meanings as in the above formula).
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は、 例えば N, N—ジメチルホルムアミドの様な適当な有 機酸溶媒中、 例えば水素化ナトリウムのような適当な塩基を用いて 0 °Cから溶媒 の沸点温度、 好ましくは 0〜 1 5 0 、 0 . 5〜9 6時間、 好ましくは 1 2〜2 4時間反応させることにより、 反応は完結する。 次いで、 例えば不活性な有機溶 媒中、 あるいはニートで 0 °Cから溶媒の沸点温度、 好ましくは 1 0 0〜3 0 0 °C 、 0 . 1〜9 6時間、 好ましくは 0 . 5〜 2時間攪拌させることにより、 反応は 完結する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but usually the reaction is carried out in a suitable organic acid solvent such as N, N-dimethylformamide, for example, sodium hydride. The reaction is completed by reacting the mixture with a suitable base at 0 ° C. to the boiling point of the solvent, preferably 0 to 150, 0.5 to 96 hours, preferably 12 to 24 hours. Then, for example, in an inert organic solvent or neat, from 0 ° C. to the boiling point of the solvent, preferably 100 to 300 ° C., 0.1 to 96 hours, preferably 0.5 to 2 hours. The reaction is completed by stirring for an hour.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれ 、 特に限定はされないが、 具体的には例えば塩ィ匕メチレン、 クロ口ホルム、 1 , 2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 N, N—ジメチルホルム アミド、 ァセトニトリル、 アセトン、 ベンゼン、 トルエン、 エーテル、 1, 4— ジォキサン、 テトラヒドロフラン、 テトラヒドロピラン等が挙げられ、 中でも N , N—ジメチルホルムアミド、 トルエン、 キシレン、 アセトン、 エーテル、 テト ラヒドロフラン等が好適である。 反応で使用される塩基としては、 例えばトリメチルァミン、 トリェチルァミン 、 N, N—ジイソプロピルェチルァミン、 N—メチルモルホリン、 N—メチルピ 口リジン、 N—メチルピペリジン、 N, N—ジメチルァニリン、 1, 8—ジァザ ビシクロ [5. 4. 0] ゥンデ力 _7—ェン (DBU) 、 1, 5—ァザビシクロ [4. 3. 0] ノナー 5—ェン (DBN) 等の第 3級脂肪族ァミン、 例えば水素 化ナトリウム、 水素化カリウム等のアルカリ金属水素化物、 例えばカリウム一 t e r t—プチラート、 ナトリウムェチラート、 ナトリウムメチラート等のアル力 リ金属アルコキシド;例えば水酸化カリウム、 水酸化ナトリウム等のアルカリ金 属水酸化物;例えば炭酸力リゥム等のアル力リ金属炭酸塩等が挙げられ、 中でも トリェチルァミン、 N, N—ジィソプロピルェチルァミン、 水素化ナトリウム、 水素化カリウム、 カリウム— t e r t—プチラート、 ナトリウムェチラート、 ナ トリウムメチラート、 炭酸カリウムが好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, but specific examples thereof include, for example, Shiridani methylene, chloroform, 1,2-dichloroethane, trichloroethane, Carbon tetrachloride, N, N-dimethylformamide, acetonitrile, acetone, benzene, toluene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran, etc., among which N, N-dimethylformamide, toluene, xylene, acetone , Ether, tetrahydrofuran and the like are preferred. Examples of the base used in the reaction include trimethylamine, triethylamine, N, N-diisopropylethylamine, N-methylmorpholine, N-methylpiperidine, N-methylpiperidine, N, N-dimethylaniline, Tertiary aliphatics such as 1, 8-diazabicyclo [5.4.0] pendant force _7-en (DBU), 1,5-azabicyclo [4.3.0] nona5 -en (DBN) Amines, for example, alkali metal hydrides such as sodium hydride and potassium hydride, for example, alkali metal alkoxides such as potassium tert-butylate, sodium ethylate, and sodium methylate; for example, potassium hydroxide, sodium hydroxide, and the like Alkali metal hydroxides; for example, alkali metal carbonates such as carbonated lime; and the like, among which triethylamine, N, N-diisopropylethylamine, Sodium hydride, potassium hydride, potassium tert-butylate, sodium ethylate, sodium methylate and potassium carbonate are preferred.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [I X] で表される化合物に対して 1. 2 〜10等量、 好ましくは 1. 2〜2等量の塩基を使用し、 1. 2〜10等量、 好 ましくは 1. 2〜2等量の塩化ジメチルチオ力ルバモイルを使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 relative to the compound represented by the above general formula [IX]. Use 2 to 2 equivalents of base, use 1.2 to 10 equivalents, preferably 1.2 to 2 equivalents of dimethylthiopotassium chloride.
転位反応において使用する不活性溶媒としては、 例えばトルエン、 キシレン、 ジフエニルエーテル、 ジーパラ—トリルエーテル等が挙げられ、 中でもジフエ二 ルエーテルが好適である。 As the inert solvent used in the rearrangement reaction, for example, toluene, xylene, diphenyl ether, dipara-tolyl ether and the like can be mentioned, and among them, diphenyl ether is preferable.
上記一般式 [X I] で表される化合物を適切な有機溶媒中において、 例えば水 酸化ナトリウムのような塩基で加水分解、 あるいは THFのような非極性溶媒中 で水素化リチウムアルミニウムなどの還元剤で還元することにより上記一般式 [ I V] で表される化合物に調整することができる。 The compound represented by the general formula [XI] is hydrolyzed with a base such as sodium hydroxide in a suitable organic solvent, or with a reducing agent such as lithium aluminum hydride in a non-polar solvent such as THF. The compound represented by the above general formula [IV] can be prepared by reduction.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば酢酸ェチルエステル、 酢酸メチルェ ステル、 ァセトニトリル、 メチルアルコール、 エチルアルコール、 ベンゼン、 キ シレン、 トルエン、 キシレン、 エーテル、 1, 4一ジォキサン、 テトラヒドロフ ラン、 テトラヒドロピラン等が挙げられ、 中でもメチルアルコール、 ェチルアル コール、 エーテル、 1, 4一ジォキサン、 テトラヒドロフラン等が好適である。 反応で使用される塩基としては、 例えばカリウム一 t e r t一プチラート、 ナ トリウムェチラート、 ナトリウムメチラート等のアルカリ金属アルコキシド;例 えば水酸化力リウム、 水酸化ナトリゥム等のアル力リ金属水酸化物;例えば炭酸 カリウム等のアルカリ金属炭酸塩等が挙げられ、 中でもカリウム一 t e r t—ブ チラ一ト、 ナトリウムェチラ一ト、 ナトリウムメチラート、 水酸化カリウム、 水 酸化ナトリゥムが好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specifically, for example, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, benzene Examples thereof include silene, toluene, xylene, ether, 1,4-dioxane, tetrahydrofuran, and tetrahydropyran. Among them, methyl alcohol, ethyl alcohol, ether, 1,4-dioxane, and tetrahydrofuran are preferable. Examples of the base used in the reaction include alkali metal alkoxides such as potassium tert-butylate, sodium ethylate and sodium methylate; for example, alkali metal hydroxides such as lithium hydroxide and sodium hydroxide. For example, alkali metal carbonates such as potassium carbonate and the like, among which potassium tert-butylate, sodium ethylate, sodium methylate, potassium hydroxide and sodium hydroxide are preferred.
反応で使用される不活性有機溶媒としては、 反応に悪影響を及ぼさないもので あれば、 特に限定はされないが、 具体的には例えばへキサン、 シクロへキサン、 ペンタン、 エーテル、 1, 4—ジォキサン、 テ卜ラヒドロフラン、 テトラヒドロ ピラン等が挙げられ、 中でもェ一テル、 テトラヒドロフラン、 シクロへキサン等 が好適である。 The inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include hexane, cyclohexane, pentane, ether, 1,4-dioxane And tetrahydrofuran, tetrahydropyran and the like, among which ether, tetrahydrofuran, cyclohexane and the like are preferable.
反応で使用される還元剤としては、 水素化アルミニウムリチウムが好適である このようにして得られた上記一般式 [ I V] で表される化合物を製造法 Aの第 2— 5工程と同様の方法で反応させることにより、 上記一般式 [ I ] で表される 化合物を得ることができる。 Lithium aluminum hydride is suitable as the reducing agent used in the reaction. The compound represented by the above general formula [IV] thus obtained is prepared in the same manner as in Step 2-5 of Production Method A. The compound represented by the above general formula [I] can be obtained by reacting
なお、 上記一般式 [ I X] で表される化合物は、 公知の方法により合成するこ とができる。 例えば一般式 [X I I ] The compound represented by the above general formula [IX] can be synthesized by a known method. For example, the general formula [X I I]
[式中、 A rは、 前記の意味を有する]で表されるサリチル酸と、 R 5—H (ここに おいて R 5は前記の意味を有する) で表されるァリール化合物と、 例えば塩化ァ ルミユウムを用いたフリーデルクラフツ反応により反応させることにより合成す ることができる。 [Wherein, Ar has the above-mentioned meaning] salicylic acid; an aryl compound represented by R 5 —H (where R 5 has the above-mentioned meaning); It can be synthesized by a Friedel-Crafts reaction using lumimium.
あるいは一般式 [ X I I I ] Or the general formula [XIII]
[式中、 A rは、 前記の意味を有する] で表される化合物をハロゲン化ァリ一ル 化合物と金属マグネシウムとを、 例えばジェチルェ一テル、 テトラヒドロフラン 等適当なエーテル系脱水溶媒中、 低温から溶媒の沸点温度で反応させることによ り調製されるグリニアル試薬とを、 前記の脱水した不活性有機溶媒中、 低温から 室温で反応させ、 生成したものを、 引き続き例えばジクロロメタンなどの適当な 溶媒中、 例えば二酸化マンガンなどの酸化剤と反応させることにより製造するこ とができる。 製造法 C [Wherein, Ar has the above-mentioned meaning] by converting an aryl halide compound and metallic magnesium into a suitable ether-based dehydrating solvent such as, for example, ethyl ether, tetrahydrofuran, etc., at a low temperature. The Grignard reagent prepared by reacting at the boiling point of the solvent is reacted with the above-mentioned dehydrated inert organic solvent at a low to room temperature in the above-mentioned dehydrated inert organic solvent, and the resulting product is subsequently dissolved in a suitable solvent such as dichloromethane. For example, it can be produced by reacting with an oxidizing agent such as manganese dioxide. Manufacturing method C
(第 1工程) (First step)
一般式 [X I V] General formula [XIV]
[式中、 P Gは、 カルボキシル基の保護基を示し、 A rは、 前記の意味を有する ] で表される化合物を一般式 [V] [Wherein, PG represents a carboxyl-protecting group, and Ar has the above-mentioned meaning] The compound represented by the general formula [V]
[式中、 R 2及び1 3は、 前記の意味を有する] で表される化合物と反応さ せ、 次いで生成したアミノ基を適当なァミノ基の保護基をかけることにより一般 式 [XV] Wherein, R 2 and 1 3 are as defined above] is reacted with a compound represented by then formula by the resulting amino group apply a protecting group suitable Amino group [XV]
[式中、 PG1は、 ァミノ基の保護基を示し、 R1 R2、 R Ar及び PGは前 記の意味を有する]で表される化合物を合成することができる。 [Wherein, PG 1 represents a protecting group for an amino group, and R 1 R 2 , R Ar and PG have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は、 例えばルチジンのような有機溶媒中、 上記一般式 [X I V] で表される化合物と上記一般式 [V] の化合物とを、 室温で 0. 5〜96 時間、 好ましくは 3〜 24時間反応させる。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions. Usually, for example, the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [XIV] and the above general formula The compound [V] is reacted at room temperature for 0.5 to 96 hours, preferably 3 to 24 hours.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1, 2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 N, N—ジメチルホルム アミド、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニトリル、 メチルァ ルコール、 エチルアルコール、 ベンゼン、 キシレン、 ピリジン、 ルチジン、 トル ェン、 キシレン、 1, 4一ジォキサン、 テトラヒドロフラン等が挙げられ、 中で もメチルアルコール、 エチルアルコール、 ピリジン、 ルチジン、 N, N—ジメチ ルホルムアミド、 トルエン、 キシレン等が好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, and specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and tetrachloroethane. Carbon chloride, N, N-dimethylformamide, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran, etc. Among them, methyl alcohol, ethyl alcohol, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferable.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [X I V] で表される化合物に対して 1. 2〜10等量、 好ましくは 1. 2〜2等量の上記一般式 [V] で表されるアジリ ジンを使用する。 The amount of the reagent used in the reaction can be appropriately increased or decreased depending on the starting material and the reaction conditions. Usually, 1.2 to 10 equivalents, preferably 1 Use 2 to 2 equivalents of the aziridine represented by the above general formula [V].
力ルポキシル基の保護基としては、 例えばメチル基、 ェチル基、 t e r t—ブ チル基等の低級アルキル基、 例えばべンジル基、 p—メトキシベンジル基等のァ ラルキル基等が挙げられ、 中でもメチル基、 ェチル基、 t e r t—ブチル基、 ベ ンジル基等が好ましい。 Examples of the protective group for the carbonyl group include lower alkyl groups such as methyl group, ethyl group and tert-butyl group, and aralkyl groups such as benzyl group and p-methoxybenzyl group. , An ethyl group, a tert-butyl group, a benzyl group and the like.
ァミノ基の保護基としては、 例えばべンジル基、 p—ニトロベンジル基等のァ ラルキル基;例えばホルミル基、 ァセチル基等のァシル基;例えばェトキシカル ポニル基、 ァリルォキシカルポニル基、 t e r t —ブトキシカルポニル基等の 低級アルコキシ力ルポニル基;例えばべンジルォキシカルポニル基、 p—ニトロ ベンジルォキシカルポニル基等のァラルキルォキシ力ルポニル基等、 トリフエ二 ルメチル基、 パラメトキシフエ二ルジフエニルメチル基、 ジフエニルホスホニル 基、 ベンゼンスルフエニル基が挙げられ、 中でもァリルォキシカルポニル基、 t e r t 一ブトキシカルポニル基、ベンジルォキシカルポニル基、 トリフエニルメ チル基等が好ましい。 Examples of the protecting group for the amino group include benzyl, p-nitrobenzyl and the like. An aralkyl group; for example, an acyl group such as a formyl group or an acetyl group; a lower alkoxy group such as an ethoxycarbonyl group, an aryloxycarbonyl group, or a tert-butoxycarbonyl group; for example, a benzyloxycarbonyl group, p-nitro Aralkyloxycarbonyl groups such as benzyloxycarbonyl group, triphenylmethyl group, paramethoxyphenyldiphenylmethyl group, diphenylphosphonyl group, and benzenesulfenyl group; among them, aryloxycarbonyl group Tert-butoxycarbonyl, benzyloxycarbonyl, triphenylmethyl and the like are preferred.
保護基の導入はその種類及びィ匕合物の安定性により異なるが、 文献記載の方法 [プロテクティブ ·グループス ·イン ·オーガニック ·シンセシス (P r o t e c t i ve Gr oup s i n Or g an i c Syn t he s i s) , T . W. グリーン (T. W. Gr e ene) 著、 J ohn Wi l ey & S o n s社 (1981) 年参照 ] 又はそれに準ずる方法に従って行うことができる。 (第 2工程) The introduction of the protecting group depends on the type and the stability of the compound, but the method described in the literature [Protective Groups in Organic Synthesis (Protective Groups in Organic Synthesis)] , T. W. Green (Tw Greene, John John & Sons, 1981)] or a method analogous thereto. (2nd step)
一般式 [XV] General formula [XV]
[式中、 R R2、 R3、 Ar、 ? 及び 01は、 前記の意味を有する] で表さ れる化合物のカルボキシル基の保護基を除去し、 次いで、 N, O—ジメチルヒド ロキシルァミンと例えば塩ィ匕メチレン中適宜塩基、 縮合補助剤及びまたは縮合剤 の存在下で反応させることにより一般式 [XV I] [XVI] [Where RR 2 , R 3 , Ar,? And 0 1, the carboxyl-protecting group is removed in the compound represented by have the meanings mentioned above, then, N, O-Jimechiruhido Rokishiruamin and example Shioi匕in methylene appropriate base, condensation assistant and or condensation Reacting in the presence of an agent to form a general formula [XVI] [XVI]
[式中、 Meは、 メチル基を示し、 R R2、 R3、 Ar及び PG1は、 前記の意 味を有する]で表される化合物を合成することができる。 [Wherein, Me represents a methyl group, and RR 2 , R 3 , Ar, and PG 1 have the above-mentioned meaning].
力ルポキシル基の保護基の除去はその種類及び化合物の安定性により異なるが 、 文献記載の方法 [プロテクティブ ·グループス 'イン'オーガニック ·シンセ シス (Pro t ec t i ve Groups i n Organ i c S y n t h e s i s) , T. W. グリーン (T. W. G r e e n e) 著、 John Wi l ey & Sons社 (1981) 年参照 ] 又はそれに準ずる方法に従って、 例えば酸又は塩基を用いる加溶媒分解、 水素化金属錯体等を用いる化学的還元又 はパラジウム炭素触媒、 ラネーニッケル触媒等を用いる接蝕還元等により行うこ とができる。 The removal of the protecting group of the hydroxyl group depends on the type and stability of the compound, but the method described in the literature [Protective Groups 'In' Organic Synthesis (Organic Synthesis), TW Greene, John John & Sons (1981)] or a method analogous thereto, for example, solvolysis using an acid or base, chemical reduction using a metal hydride complex, or the like. It can be carried out by corrosion reduction using a palladium carbon catalyst, Raney nickel catalyst or the like.
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は、 脱水した不活性有機溶媒中、 脱保護したカルボン酸と N, 〇一ジメチルヒドロキシルァミンとを、 適宜塩基、 縮合補助剤及び Z又は縮 合剤の存在下に、 — 100 °Cから溶媒の沸点温度、 好ましくは 0〜 30 °C、 0. 5〜96時間、 好ましくは 3〜24時間反応させる。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but usually, the reaction is carried out by reacting the deprotected carboxylic acid with N, 1-dimethylhydroxylamine in a dehydrated inert organic solvent. , In the presence of a base, a condensation aid and Z or a condensing agent as appropriate — from 100 ° C. to the boiling point of the solvent, preferably from 0 to 30 ° C., from 0.5 to 96 hours, preferably from 3 to 24 hours. Let it.
反応で使用される不活性有機溶媒としては、 反応に悪影響を及ぼさないもので あれば、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム 、 1, 2—ジクロロェタン、 トリクロロェタン、 N, N—ジメチルホルムアミド 、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニトリル、 無水酢酸、 メチ ルアルコ一ル、 エチルアルコール、 ベンゼン、 キシレン、 水、 酢酸、 トルエン、 1, 4—ジォキサン、 テトラヒドロフラン等が挙げられ、 好適な反応温度の確保 の点から、 中でも塩化メチレン、 クロ口ホルム、 1, 2—ジクロロェタン、 ァセ トニトリル、 N, N—ジメチルホルムアミド、 1, 4—ジォキサン、 トルエン等 が好適である。 The inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, and specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, and trichloroethane. , N, N-dimethylformamide, ethyl acetate, methyl acetate, acetonitrile, acetic anhydride, methyl alcohol, ethyl alcohol, benzene, xylene, water, acetic acid, toluene, 1,4-dioxane, tetrahydrofuran and the like. From the viewpoint of securing a suitable reaction temperature, among them, methylene chloride, chloroform, 1,2-dichloroethane, acetonitrile, N, N-dimethylformamide, 1,4-dioxane, toluene, etc. Is preferred.
反応で使用される塩基としては、 例えばトリメチルァミン、 トリエチ^/アミ 、 N, N—ジイソプロピルェチルァミン、 N—メチルモルホリン、 N—メチルビ 口リジン、 N—メチルピペリジン、 N, N—ジメチルァニリン、 1, 8—ジァザ ビシクロ [ 5 . 4. 0 ] ゥンデ力一 7—ェン (D B U) 、 1 , 5—ァザビシクロ [ 4. 3 . 0 ] ノナ— 5—ェン (D B N) 等の第 3級脂肪族ァミン;例えばピリ ジン、 4—ジメチルァミノピリジン、 ピコリン、 ルチジン、 キノリン、 イソキノ リン等の芳香族ァミン;例えば金属カリウム、 金属ナトリウム、 金属リチウム等 のアル力リ金属;例えば水素化ナトリウム、 水素化力リゥム等のァレ力リ金属水 素化物;例えばプチルリチウム等のアル力リ金属アルキル化物;例えば力リウム - t e r tーブチラ一ト、 ナトリウムェチラ一ト、 ナトリウムメチラート等のァ ルカリ金属アルコキシド;例えば水酸化力リゥム、 水酸化ナトリゥム等のアル力 リ金属水酸化物;例えば炭酸力リゥム等のアル力リ金属炭酸塩等が挙げられ、 中 でも第 3級脂肪族ァミン等が好適であり、 特にトリェチルァミン、 N, N—ジィ ソプロピルェチルアミン等がより好適である。 Bases used in the reaction include, for example, trimethylamine, triethyl ^ / ami, N, N-diisopropylethylamine, N-methylmorpholine, N-methylvinyl lysine, N-methylpiperidine, N, N-dimethyl Anilin, 1,8-diazabicyclo [5.4.0] pendant 7-ene (DBU), 1,5-azabicyclo [4.3.0] nona-5-ene (DBN), etc. Tertiary aliphatic amines; for example, aromatic amines such as pyridine, 4-dimethylaminopyridine, picoline, lutidine, quinoline, and isoquinoline; metal salts such as potassium metal, sodium metal, and lithium metal; Metal hydrides such as sodium hydride and hydrogen hydride; alkyl metal hydrides such as butyl lithium; potassium lithium-tert-butylate, sodium ethylate Alkali metal alkoxides such as sodium methylate and the like; alkali metal hydroxides such as hydroxylated sodium hydroxide and sodium hydroxide; alkali metal alkoxides such as carbonated lithium and the like. Tertiary aliphatic amines and the like are preferred, and in particular, triethylamine, N, N-diisopropylethylamine and the like are more preferred.
反応で使用される縮合補助剤としては、 例えば N—ヒドロキシベンゾトリァゾ —ル水和物、 N—ヒドロキシスクシンイミド、 N—ヒドロキシ一 5—ノルポルネ ンー 2, 3—ジカルボキシイミド、 3—ヒドロキシ— 3 , 4—ジヒドロー 4ーォ キソ— 1 , 2 , 3—ベンゾトリアゾ一ル等が挙げられ、 中でも N—ヒドロキシべ ンゾ卜リアゾール等が好適である。 Examples of the condensation auxiliary used in the reaction include N-hydroxybenzotriazole hydrate, N-hydroxysuccinimide, N-hydroxy-15-norpolene-2,3-dicarboximide, and 3-hydroxy-3 , 4-dihydro-4-oxo-1,2,3-benzotriazol and the like. Among them, N-hydroxybenzotriazole and the like are preferable.
反応で使用される縮合剤としては、 例えば塩化チォニル、 N, N—ジシクロへ キシルカルポジイミド、 1一メチル—2—ブロモピリジニゥムアイオダイド、 N , N ' —力ルポニルジイミダゾ一ル、 ジフエニルフォスフォリルクロリド、 ジフ ェニルフォスフォリルアジド、 N, N, 一ジスクシ二ミジルカルポネ一ト、 N, N ' —ジスクシ二ミジルォキザレート、 1ーェチルー 3— ( 3—ジメチルァミノ プロピル) カルポジイミド塩酸塩、 クロ口ギ酸ェチル、 クロロギ酸イソプチル、 ベンゾトリアゾ— 1—リル—ォキシートリス (ジメチルァミノ) フォスフォニゥ ムへキサフルオロフォスフェイト等が挙げられ、 中でも N, N—ジシクロへキシ ルカルポジィミド、 1—ェチルー 3— ( 3—ジメチルアミノプロピル) カルポジ イミド塩酸塩、 クロ口ギ酸ェチル、 クロロギ酸イソブチル等が好適である。 反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常カルボン酸に対して 1〜50等量、 好ましくは 1〜2等 量の N, 〇ージメチルヒドロキシルァミン、 1〜50等量、 好ましくは 3〜5等 量の塩基、 1〜 50等量、 好ましくは 1〜 5等量の縮合補助剤及び/又は 1〜 5 0等量、 好ましくは 1〜5等量の縮合剤を使用する。 該塩基、 該縮合補助剤及び 該縮合剤は、 一種又はそれ以上適宜組合せて使用することができる。 Examples of the condensing agent used in the reaction include thionyl chloride, N, N-dicyclohexylcarbodiimide, 1-methyl-2-bromopyridinium iodide, and N, N'-capillonyldiimidazole , Diphenylphosphoryl chloride, diphenylphosphoryl azide, N, N, disuccinimidyl carbone, N, N'-disuccinimidyl oxalate, 1-ethyl-3- (3-dimethylaminopropyl) carbodiimide Hydrochloride, ethyl ethyl chloroformate, isoptyl chloroformate, benzotriazo-1-lyloxysheetris (dimethylamino) phosphonium hexafluorophosphate, and the like, among which N, N-dicyclohexylcarposimide, 1-ethyl-3- ( 3-dimethylaminopropyl) carposi Preference is given to imide hydrochloride, ethyl ethyl chloroformate, isobutyl chloroformate and the like. The reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions, but is usually 1 to 50 equivalents, preferably 1 to 2 equivalents of N, di-dimethylhydroxyl hydroxylase relative to the carboxylic acid. Min, 1 to 50 equivalents, preferably 3 to 5 equivalents of base, 1 to 50 equivalents, preferably 1 to 5 equivalents of condensation aid and / or 1 to 50 equivalents, preferably 1 to 5 equivalents Use an equal amount of condensing agent. The base, the condensation aid, and the condensing agent can be used alone or in appropriate combination of two or more.
(第 3工程) (3rd step)
一般式 [XV I] General formula [XV I]
[式中、 R R2、 R3、 Ar、 PG1及び Meは、 前記の意味を有する] で表さ れる化合物をハロゲン化ァリール化合物と金属マグネシウムとを、 例えばジェチ ルエーテル、 テトラヒドロフラン等適当なエーテル系脱水溶媒中、 低温から溶媒 の沸点温度で反応させることにより調製されるグリ二アル試薬とを、 前記の脱水 した不活性有機溶媒中、 低温から室温で反応させることにより一般式 [XVI I ] [Wherein, RR 2 , R 3 , Ar, PG 1 and Me have the above-mentioned meaning], by converting a halogenated aryl compound and metal magnesium to a suitable ether-based compound such as, for example, methyl ether, tetrahydrofuran, etc. A general formula [XVI I] is obtained by reacting in a dehydrated inert organic solvent at a low temperature to room temperature with a Grignard reagent prepared by reacting at a low temperature to the boiling point of the solvent in a dehydrated solvent.
[式中、 R1, R2、 R3、 R5、 Ar及び PG1は、 前記の意味を有する]で表され る化合物を合成することができる。 反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は脱水した不活性有機溶媒中、 上記一般式 [XV I] で表 される化合物とグリニアル試薬とを一 100 から溶媒の沸点温度、 好ましくは — 78〜30^、 0. 5〜96時間、 好ましくは 12〜24時間反応させる。 反応で使用される不活性有機溶媒としては、 反応に悪影響を及ぼさないもので あれば、 特に限定はされないが、 具体的には例えばへキサン、 シクロへキサン、 ペンタン、 エーテル、 1, 4—ジォキサン、 テトラヒドロフラン、 テトラヒドロ ピラン等が挙げられ、 中でもエーテル、 テトラヒドロフラン、 シクロへキサン等 が好適である。 [Wherein, R 1 , R 2 , R 3 , R 5 , Ar and PG 1 have the above-mentioned meaning]. The reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions. Usually, the reaction is carried out in an inert organic solvent that has been dehydrated by reacting the compound represented by the above general formula [XVI] with the Grignard reagent. The reaction is carried out at a temperature of 100 to the boiling point of the solvent, preferably -78 to 30 ^, for 0.5 to 96 hours, preferably for 12 to 24 hours. The inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include hexane, cyclohexane, pentane, ether, 1,4-dioxane , Tetrahydrofuran, tetrahydropyran and the like, among which ether, tetrahydrofuran, cyclohexane and the like are preferable.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [XV I] で表される化合物に対して 2〜 10等量、 好ましくは 2〜 3等量のグリニアル試薬を使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and reaction conditions, but is usually 2 to 10 equivalents, preferably 2 to 10 equivalents to the compound represented by the above general formula [XVI]. Use 3 equivalents of Grignard reagent.
(第 4工程) (4th step)
一般式 [XV I I] General formula [XV I I]
[式中、 R1 R2、 R3、 R5、 Ar及び PG1は、 前記の意味を有する] で表さ れる化合物のァミノ基の保護基を適宜除去し一般式 [V I ] [Wherein, R 1 R 2 , R 3 , R 5 , Ar and PG 1 have the above-mentioned meaning] The protecting group for the amino group of the compound represented by the general formula [VI]
[式中、 R1 R2、 R3、 1 5及び八1"は、 前記の意味を有する]で表される化合物 を合成することができる。 [Wherein, R 1 R 2 , R 3 , 15 and 18 "have the meanings described above] Can be synthesized.
保護基の除去はその種類及び化合物の安定性により異なるが、 文献記載の方法 [プロテクティブ ·グループス ·イン ·オーガニック ·シンセシス (P r o t e c t i ve Gr ou s i n Or g an i c Syn t h e s i s) , T . W. グリーン (T. W. G r e e n e) 著、 J o hn Wi l ey & S o n s社(1981)年参照 ] 又はそれに準ずる方法に従って、 例えば酸又は塩基 を用いる加溶媒分解、 水素化金属錯体等を用いる化学的還元又はパラジウム炭素 触媒、 ラネーニッケル触媒等を用いる接触還元等により行うことができる。 得られた上記一般式 [VI] で表される化合物は引き続き製造法 Aの第 3— 5 工程の方法で反応させることにより上記一般式 [I] で表される化合物を合成す ることができる。 製造法 D The removal of the protecting group depends on the type and stability of the compound, but the method described in the literature [Protective Groups in Organic Synthesis, T.W. Green (TW Greene, John Wiley & Sons (1981)) or a method analogous thereto, for example, solvolysis using an acid or a base, chemical reduction using a metal hydride complex, or the like. Alternatively, it can be carried out by catalytic reduction using a palladium carbon catalyst, a Raney nickel catalyst or the like. The compound represented by the above general formula [VI] can be synthesized by subsequently reacting the compound represented by the above general formula [VI] by the method of Step 3-5 of Production method A. . Manufacturing method D
(第 1工程) (First step)
なお、 一般式 [X I X] The general formula [X I X]
[式中、 L1は、 ハロゲン等の脱離基を示し、 R5及び Arは、 前記の意味を有す る] で表される化合物は、 公知の方法により合成することができる。 [Wherein L 1 represents a leaving group such as halogen, and R 5 and Ar have the above-mentioned meanings] can be synthesized by a known method.
例えば式一般式 [XV I I I] For example, the general formula [XV I I I]
[式中、 八!:及び!^は、 前記の意味を有すると R 5— H (ここにおいて、 R5は 前記の意味を有する) で表されるァリール化合物と、 例えば塩化アルミニウムを 用いたフリーデルクラフツ反応により反応させることにより合成することができ る。 [In the formula, eight! :as well as! ^ Has the meaning R 5 — H (where R 5 is The compound can be synthesized by reacting with an aryl compound represented by the following formula) by, for example, a Friedel-Crafts reaction using aluminum chloride.
(第 2工程) (2nd step)
上記一般式 [X I X] と、 一般式 [XX] The general formula [X I X] and the general formula [XX]
[式中、 R R2、 3及び?01は、 前記の意味を有する] で表されるチオール 化合物と反応させることによりに一般式 [XV I I] [Where RR 2 , 3 and? 0 1 of the general formula in by reaction with a thiol compound represented by have the meanings of the [XV II]
I]I]
[式中、 R R2、 R3、 R5、 八1"及び? 1は、 前記の意味を有する]で表され る化合物が合成できる。 [Wherein, RR 2 , R 3 , R 5 , 8 1 "and? 1 have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は、 例えば N, N—ジメチルホルムアミドの様な適当な有 機酸溶媒中、 例えば炭酸カリウムのような適当な塩基を用いて o :から溶媒の沸 点温度、 好ましくは 50~120°C、 0. 5〜96時間、 好ましくは 2〜12時 間反応させることにより、 反応は完結する。 The reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions. Usually, the reaction is carried out in a suitable organic acid solvent such as N, N-dimethylformamide, for example, potassium carbonate. The reaction is completed by reacting with a suitable base at o: from the boiling point of the solvent, preferably 50 to 120 ° C, for 0.5 to 96 hours, preferably 2 to 12 hours.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば As the organic solvent used in the reaction, any organic solvent that does not adversely affect the reaction can be used.
、 特に限定はされないが、 具体的には例えば塩ィヒメチレン、 クロ口ホルム、 1,Although not particularly limited, specifically, for example, salt dimethylene, black form,
2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 N, N—ジメチルホルム アミド、 ァセトニトリル、 アセトン、 ベンゼン、 トルエン、 エーテル、 1, 4一 ジォキサン、 テトラヒドロフラン、 テトラヒドロピラン等が挙げられ、 中でも N , N—ジメチルホルムアミド、 トルエン、 キシレン、 アセトン、 エーテル、 テト ラヒドロフラン等が好適である。 2-dichloroethane, trichloroethane, carbon tetrachloride, N, N-dimethylformamide, acetonitrile, acetone, benzene, toluene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran, etc., among which N , N-dimethylformamide, toluene, xylene, acetone, ether, tetrahydrofuran and the like are preferred.
反応で使用される塩基としては、 例えばトリメチルァミン、 トリェチルァミン 、 N, N—ジイソプロピルェチルァミン、 N—メチルモルホリン、 N—メチルピ 口リジン、 N—メチルピペリジン、 N, N—ジメチルァニリン、 1, 8—ジァザ ビシクロ [5. 4. 0] ゥンデ力一 7—ェン (DBU) 、 1, 5—ァザビシクロ [4. 3. 0] ノナー 5—ェン (DBN) 等の第 3級脂肪族ァミン、 例えば水素 化ナトリウム、 水素化カリウム等のアルカリ金属水素化物、 例えばカリウム一 t e r t—プチラート、 ナトリウムェチラート、 ナトリウムメチラ一ト等のアル力 リ金属アルコキシド;例えば水酸化カリウム、 7jc酸化ナトリウム等のアルカリ金 属水酸化物;例えば炭酸力リゥム等のアル力リ金属炭酸塩等が挙げられ、 中でも トリェチルァミン、 N, N—ジイソプロピルェチルァミン、 水素化ナトリウム、 水素化カリウム、 カリウム一 t e r t—プチラート、 ナトリウムェチラート、 ナ トリウムメチラート、 炭酸カリウムが好適である。 Examples of the base used in the reaction include trimethylamine, triethylamine, N, N-diisopropylethylamine, N-methylmorpholine, N-methylpiperidine, N-methylpiperidine, N, N-dimethylaniline, Tertiary fats such as 1,8-diazabicyclo [5.4.0] pendant 7-ene (DBU) and 1,5-azabicyclo [4.3.0] noner 5-ene (DBN) Aliphatic amines, for example, alkali metal hydrides such as sodium hydride and potassium hydride, for example, alkali metal alkoxides such as potassium tert-butylate, sodium ethylate, sodium methylate; potassium hydroxide, 7jc oxidation Alkali metal hydroxides such as sodium; for example, alkali metal carbonates such as carbon dioxide, etc., among which triethylamine, N, N-diisopropylethyl Amin, sodium hydride, potassium hydride, potassium one t e r t-Puchirato, sodium E Ji methoxide, sodium methylate, potassium carbonate is preferred.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [XI X] で表される化合物に対して 1. 2〜20等量、 好ましくは 1. 2〜 5等量の上記一般式 [XX] で表される化合 物を使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the raw material compounds and reaction conditions, but is usually 1.2 to 20 equivalents, preferably 1 to 20 equivalents to the compound represented by the above general formula [XIX]. 1. Use 2 to 5 equivalents of the compound represented by the general formula [XX].
得られた上記一般式 [XV I I] で表される化合物は製造法 Cの第 4工程、 製 造法 Aの第 3— 5工程の方法で上記一般式 [I] で表される化合物に合成するこ とができる。 The compound represented by the above general formula [XV II] is synthesized into the compound represented by the above general formula [I] by the method of the fourth step of the production method C and the third to fifth steps of the production method A. can do.
製造法 E Manufacturing method E
(第 1工程) (First step)
一般式 [XX I] General formula [XX I]
[式中、 L2は、 ハロゲン等の脱離基を示し、 Arは、 前記の意味を有する] で 表される化合物と一般式 [V] [Wherein L 2 represents a leaving group such as halogen, and Ar has the above-mentioned meaning] Compound represented by general formula [V]
[式中、 R R2、 R3は、 前記の意味を有する] で表される化合物と例えばル チジンのような極性溶媒中でを反応させ、 一般式 [XX I I] [Wherein RR 2 and R 3 have the above-mentioned meanings] and a polar solvent such as lutidine to react with a compound represented by the general formula [XXII]
[式中、 R1, R2、 R3、 R5、 Ar、 L2は、 前記の意味を有する] で表される 化合物に変換できる。 [Wherein, R 1 , R 2 , R 3 , R 5 , Ar, and L 2 have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は、 例えばルチジンのような有機溶媒中、 上記一般式 [X XI] で表される化合物と上記一般式 [V] で表される化合物とを、 室温で 0. The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions. Usually, for example, the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [XXI] and the above general compound. The compound represented by the formula [V] is dissolved at room temperature in 0.
5〜96時間、 好ましくは 3〜 24時間反応させる。 The reaction is performed for 5 to 96 hours, preferably 3 to 24 hours.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば As the organic solvent used in the reaction, any organic solvent that does not adversely affect the reaction can be used.
、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1,Although not particularly limited, specifically, for example, methylene chloride, chloroform,
2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 N, N—ジメチルホルム アミド、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニトリル、 メチルァ ルコール、 エチルアルコール、 ベンゼン、 キシレン、 ピリジン、 ルチジン、 トル ェン、 キシレン、 1, 4一ジォキサン、 テトラヒドロフラン等が挙げられ、 中で もメチルアルコール、 エチルアルコール、 ピリジン、 ルチジン、 N, N—ジメチ ルホルムアミド、 トルエン、 キシレン等が好適である。 2-dichloroethane, trichloroethane, carbon tetrachloride, N, N-dimethylformamide, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1 , 4-dioxane, tetrahydrofuran and the like, among which methyl alcohol, ethyl alcohol, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferable.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [XXI] で表される化合物に対して 1. The reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions, but usually, the amount of the compound represented by the above general formula [XXI] is 1.
2〜10等量、 好ましくは 1. 2〜 2等量の上記一般式 [V] で表されるアジリ ジンを使用する。 2 to 10 equivalents, preferably 1.2 to 2 equivalents of the aziridyl represented by the above general formula [V] Use gin.
(第 2工程) (2nd step)
一般式 [XXI I] General formula [XXI I]
[式中、 R1 R2, R3、 Ar、 L2は、 前記の意味を有する] で表される化合物 と、 一般式 [X I I I] [Wherein, R 1 R 2 , R 3 , Ar and L 2 have the above-mentioned meanings] and a general formula [XIII]
R5CH0 [XXI 111 R 5 CH0 [XXI 111
[式中、 R5は、 前記の意味を有する] で表される化合物と、 例えば 2、 6—ル チジンの様な適当な溶媒中で、 適宜酸の存在下で共沸蒸留、 または例えばモレキ ユラーシーブスのような乾燥剤の存在下、 加熱還流させることにより、 一般式 [ XXIV] Wherein R 5 has the meaning described above, and azeotropic distillation in a suitable solvent such as 2,6-lutidine in the presence of an appropriate acid, or for example, By heating to reflux in the presence of a desiccant such as Ura sieves, the general formula [XXIV]
[式中、 R1 R2、 R3、 R5、 Ar、 L2は、 前記の意味を有する] で表され る化合物を合成することができる。 [Wherein, R 1 R 2 , R 3 , R 5 , Ar, and L 2 have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によつて適宜増減することが できるが、 通常反応は、 例えばモレキュラーシーブスのような乾燥剤の存在下、 例えば 2, 6—ルチジンの様な適当な溶媒中で、 例えば塩酸の様な適当な酸の存 在下で 5〜96時間、 好ましくは 12〜24時間還流させることにより、 反応は 完結する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but usually the reaction is carried out in the presence of a desiccant such as molecular sieves, for example, using a suitable agent such as 2,6-lutidine. The reaction is completed by refluxing in a solvent, for example for 5 to 96 hours, preferably 12 to 24 hours, in the presence of a suitable acid such as hydrochloric acid.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1, 2—ジクロロェタン、 トリクロロェタン、 N, N—ジメチルホルムアミド、 無水 酢酸、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニトリル、 メチルアル コール、 エチルアルコール、 n—プロパノール、 ベンゼン、 キシレン、 ピリジン 、 ルチジン、 トルエン、 キシレン、 1, 4一ジォキサン、 テトラヒドロフラン等 が挙げられるが、 好適な反応温度の確保の点から、 中でもピリジン、 ルチジン、 N, N—ジメチルホルムアミド、 トルエン、 キシレン等が好適である。 As the organic solvent used in the reaction, any organic solvent that does not adversely affect the reaction can be used. Specific examples include, but are not limited to, methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, N, N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, and methyl alcohol. , Ethyl alcohol, n-propanol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran, and the like. Among them, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferred.
反応で使用される酸としては、 例えば塩酸、 硝酸、 臭化水素酸、 硫酸、 フッ化 水素酸、 過塩素酸等の無機酸;例えばトリフルォロホウ酸、 四塩化チタン、 塩化 亜鉛等のルイス酸;例えば!)一トルエンスルホン酸、 トリフルォロメタンスルホ ン酸、 メタンスルホン酸等のスルホン酸;蟻酸、 トリフルォロ酢酸、 酢酸等の有 機酸等が挙げられ、 中でも塩酸、 酢酸又は p—トルエンスルホン酸が好適である 反応で使用される乾燥剤しては、 例えばモレキュラーシーブス、 硫酸マグネシ ゥム、 硫酸ナトリウム等が挙げられ、 中でもモレキュラーシーブスが好適である 反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [XX I I ] で表される化合物に対して 1 . 2〜1 0等量、 好ましくは 1 . 2〜2等量の上記一般式 [X X I I I ] で表さ れるアルデヒド、 1 . 2〜1 0等量、 好ましくは 1 . 2〜2等量の乾燥剤、 1〜 5 0等量、 好ましくは 3〜 5等量の酸を使用する。 該酸、 該乾燥剤剤は、 一種又 はそれ以上適宜組み合わせて使用することもできる。 Examples of the acid used in the reaction include inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid, and perchloric acid; for example, Lewis acids such as trifluoroboric acid, titanium tetrachloride, and zinc chloride; !) Sulfonic acids such as mono-toluenesulfonic acid, trifluoromethanesulfonic acid, and methanesulfonic acid; organic acids such as formic acid, trifluoroacetic acid, and acetic acid; and hydrochloric acid, acetic acid, and p-toluenesulfonic acid are preferable. The desiccant used in the reaction includes, for example, molecular sieves, magnesium sulfate, sodium sulfate, etc. Among them, the molecular sieves are preferable. Although it can be increased or decreased as appropriate, it is usually 1.2 to 10 equivalents to the compound represented by the above general formula [XXII]. Preferably 1.2 to 2 equivalents of the aldehyde represented by the above general formula [XXIII], 1.2 to 10 equivalents, preferably 1.2 to 2 equivalents of the desiccant, 1 to 50 equivalents Preferably, 3 to 5 equivalents of acid are used. The acid and the desiccant may be used alone or in combination of two or more.
(第 3工程) (3rd step)
一般式 [XX I V] General formula [XX I V]
[ XXIV ][XXIV]
[式中、 R1, R2、 R3、 R5、 Ar、 L2は、 前記の意味を有する] で表される 化合物を適宜塩基によって、 不活性な有機溶媒中で例えば一 78°Cの様な低温で 処理することにより閉環させ一般式 [XXV] Wherein R 1 , R 2 , R 3 , R 5 , Ar, and L 2 have the above-mentioned meanings. The compound represented by the formula: The ring is closed by treating at a low temperature, such as the general formula [XXV]
[式中、 R R R3、 R5、 Arは、 前記の意味を有する] で表される化合物 が合成できる。 [Wherein, RRR 3 , R 5 , and Ar have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は脱水した不活性有機溶媒中、 適宜添加剤の存在下で適宜 塩基を用いて、 一 100°Cから溶媒の沸点温度、 好ましくは一 78〜 30° (:、 0 . 5〜96時間、 好ましくは 12〜24時間反応させることで分子内閉環反応に より完結する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.Normally, the reaction is carried out in a dehydrated inert organic solvent using a suitable base in the presence of an appropriate additive at a temperature of 100 ° C. To the boiling point of the solvent, preferably 1 to 78 to 30 ° (: 0.5 to 96 hours, preferably 12 to 24 hours to complete the intramolecular ring closure reaction.
反応で使用される不活性有機溶媒としては、 反応に悪影響を及ぼさないもので あれば、 特に限定されないが、 具体的には例えばへキサン、 シクロへキサン、 ぺ ンタン、 エーテル、 1, 4一ジォキサン、 テトラヒドロフラン、 テトラヒドロピ ラン等が挙げられるが、 特に例えばェ一テル、 テトラヒドロフラン、 シクロへキ サン等が好適である。 The inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include hexane, cyclohexane, pentane, ether, and 1,4-dioxane. And tetrahydrofuran, tetrahydropyran and the like, and particularly, for example, ether, tetrahydrofuran, cyclohexane and the like are preferable.
反応で使用される添加剤としては、 Ν,Ν,Ν',Ν'—テトラメチルエチレンジアミ ン TMEDA、 へキサメチルホスホラミン HMPA、 塩ィ匕リチウム等が挙げられ、 中でも Ν,Ν,Ν',Ν'—テトラメチルェチレンジアミンが好適である。 Examples of the additives used in the reaction include Ν, Ν, Ν ′, Ν′-tetramethylethylenediamine TMEDA, hexamethylphosphoramine HMPA, lithium salt, and the like. ', Ν'-Tetramethylethylenediamine is preferred.
反応で使用される塩基しては、 例えば η—ブチルリチウム、 s e c—ブチルリ チウム、 t一ブチルリチウム、 フエニルリチウム、 メチルリチウム等が挙げられ 、 中でも n—ブチルリチウム、 s e c—ブチルリチウムが好適である。 Examples of the base used in the reaction include η-butyllithium, sec-butyllithium, t-butyllithium, phenyllithium, methyllithium and the like. Among them, n-butyllithium and sec-butyllithium are preferred. is there.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [XX I V] で表される化合物に対して 2 〜10等量、 好ましくは 2〜 3等量の塩基を使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions. However, usually, the amount of the reagent represented by the above general formula [XXIV] is 2%. ~ 10 equivalents, preferably 2-3 equivalents of base are used.
得られた上記一般式 [XXV] で表される化合物は製造法 Aの第 4工程の方法 で上記一般式 [I] で表される化合物が合成できる。 製造法 F The obtained compound represented by the general formula [XXV] can be synthesized by the method of the fourth step of the production method A to produce the compound represented by the general formula [I]. Manufacturing method F
(第 1工程) (First step)
一般式 [XXV I] General formula [XXV I]
[式中、 Arは、 前記の意味を有する] で表される化合物と一般式 [V] [Wherein, Ar has the meaning described above] and a general formula [V]
[式中、 R1 R2、 R3は、 前記の意味を有する] で表される化合物を例えばル チジンのような極性溶媒中でを反応させ、 一般式 [XXVI I] Wherein R 1 R 2 and R 3 have the above-mentioned meanings in a polar solvent such as lutidine, and the compound represented by the general formula [XXVI I]
[XXVII][XXVII]
[式中、 R1 R2、 R3、 Arは、 前記の意味を有する] で表される化合物に変 換できる。 [Wherein, R 1 R 2 , R 3 , and Ar have the above-mentioned meanings].
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常反応は、 例えばルチジンのような有機溶媒中、 上記一般式 [XX V I] で表される化合物と上記一般式 [V] で表される化合物とを、 室温で 0. 5〜96時間、 好ましくは 3〜 24時間反応させる。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions. Usually, for example, the reaction is carried out in an organic solvent such as lutidine with the compound represented by the above general formula [XXVI] and the compound represented by the above general formula. The compound represented by [V] is reacted at room temperature for 0.5 to 96 hours, preferably 3 to 24 hours.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1, 2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 N, N—ジメチルホルム アミド、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニ卜リル、 メチルァ ルコール、 エチルアルコール、 ベンゼン、 キシレン、 ピリジン、 ルチジン、 トル ェン、 キシレン、 1, 4—ジォキサン、 テトラヒドロフラン等が挙げられ、 中で もメチルアルコール、 エチルアルコール、 ピリジン、 ルチジン、 N, N—ジメチ ルホルムアミド、 トルエン、 キシレン等が好適である。 As the organic solvent used in the reaction, any organic solvent that does not adversely affect the reaction can be used. Specific examples include, but are not limited to, methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, carbon tetrachloride, N, N-dimethylformamide, ethyl acetate, methyl acetate, and acetate nitrite. Ryl, methyl alcohol, ethyl alcohol, benzene, xylene, pyridine, lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran, etc., among which methyl alcohol, ethyl alcohol, pyridine, lutidine, N, N —Dimethylformamide, toluene, xylene and the like are preferred.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [XXV I I] で表される化合物に対して 1. 2〜10等量、 好ましくは 1. 2〜2等量の上記一般式 [V] で表されるァ ジリジンを使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the raw material compounds and reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1 to 10 equivalents to the compound represented by the above general formula [XXV II]. 1. Use 2 to 2 equivalents of the aziridine represented by the above general formula [V].
(第 2工程) (2nd step)
一般式 [XXV I I] General formula [XXV I I]
[XXVII][XXVII]
[式中、 R R2、 R3、 Arは、 前記の意味を有する] 表される化合物を適当 な酸化剤と反応させることによりに一般式 [XXV I I I] [Wherein, RR 2 , R 3 and Ar have the above-mentioned meanings] The compound represented by the general formula [XXV III]
[XXVIII][XXVIII]
〔式中、 R R2、 R3及び Arは、 前記の意味を有する〕 で表される化合物が 合成でき、 引き続き例えばモレキュラーシーブスのような乾燥剤の存在下、 例え ば 2、 6—ルチジンの様な適当な溶媒中で、 例えば塩酸の様な適当な酸の存在下 で加熱還流させることにより、 一般式 [XX I X] [Wherein RR 2 , R 3 and Ar have the above-mentioned meanings] can be synthesized, and subsequently, in the presence of a desiccant such as molecular sieves, for example, 2,6-lutidine By heating to reflux in a suitable solvent in the presence of a suitable acid such as hydrochloric acid, the compound represented by the general formula [XX IX]
〔式中、 R R 2、 尺3及び八1"は、 前記の意味を有する〕 で表される化合物が 合成できる。 [Wherein, RR 2 , shaku 3 and yachi 1 ”have the above-mentioned meanings].
酸化反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減するこ とができるが、 通常反応は、 例えば適当な溶媒中で、 二酸化マンガンの様な適当 な酸化剤と室温で 5〜9 6時間、 好ましくは 1 2〜2 4時間反応させることによ り完結する。 The reagent used in the oxidation reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions.In general, the reaction is carried out, for example, in a suitable solvent with a suitable oxidizing agent such as manganese dioxide at room temperature for 5 to 96 hours. The reaction is completed for a period of time, preferably 12 to 24 hours.
反応で使用される酸化剤としては、 反応に悪影響を及ぼさないものであれば、 特に限定はされないが、 具体的には例えば二酸化マンガン、 クロム酸、 クロム酸 t—プチル、 四酢酸鉛、 四酸化ルテニウム、 二酸化セレン、 ハロゲン、 ョードシ ル化合物、 酸素、 有機過酸化物、 ペルォキソ硫酸銀塩、 四酸化窒素、 ジメチルス ルフォキシド、 o—クロラニル、 酸化銀等が挙げられ、 中でも二酸化マンガン、 クロム酸、 二酸化セレン、 ジメチルスルフォキシド等が好適である。 The oxidizing agent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, but specific examples thereof include manganese dioxide, chromic acid, t-butyl chromate, lead tetraacetate, and tetroxide. Ruthenium, selenium dioxide, halogen, oodosil compounds, oxygen, organic peroxides, silver peroxosulfate, nitric oxide, dimethyl sulfoxide, o-chloranil, silver oxide, etc., among which manganese dioxide, chromic acid, selenium dioxide And dimethyl sulfoxide.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [V I I ] で表される化合物に対して 0 . 0 2〜5 0等量、 好ましくは 1 . 2〜 2等量の酸化剤を使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but is usually preferably from 0.22 to 50 equivalents, preferably from the compound represented by the above general formula [VII]. Use 1.2 to 2 equivalents of oxidizing agent.
閉環反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減するこ とができるが、 通常反応は、 例えばモレキュラーシ一ブスのような乾燥剤の存在 下、 例えば 2, 6—ルチジンの様な適当な溶媒中で、 例えば塩酸の様な適当な酸 の存在下で 5〜 9 6時間、 好ましくは 1 2〜 2 4時間還流させることにより、 反 応は完結する。 The reagent used in the ring closure reaction can be appropriately increased or decreased depending on the starting compound and reaction conditions, but usually the reaction is carried out in the presence of a desiccant such as molecular sieves, for example, 2,6-lutidine. The reaction is completed by refluxing in a suitable solvent, for example in the presence of a suitable acid such as hydrochloric acid for 5 to 96 hours, preferably 12 to 24 hours.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1 , 2—ジクロロェタン、 トリクロロェタン、 N, N—ジメチルホルムアミド、 無水 酢酸、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニ卜リル、 メチルアル コール、 エチルアルコール、 n—プロパノール、 ベンゼン、 キシレン、 ピリジン 、 ルチジン、 トルエン、 キシレン、 1 , 4—ジォキサン、 テトラヒドロフラン等 が挙げられ、 中でもピリジン、 ルチジン、 N, N—ジメチルホルムアミド、 トル ェン、 キシレン等が好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and N , N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, n-propanol, benzene, xylene, pyridine , Lutidine, toluene, xylene, 1,4-dioxane, tetrahydrofuran and the like. Among them, pyridine, lutidine, N, N-dimethylformamide, toluene, xylene and the like are preferable.
反応で使用される酸としては、 例えば塩酸、 硝酸、 臭化水素酸、 硫酸、 フッ化 水素酸、 過塩素酸等の無機酸;例えばトリフルォロホウ酸、 四塩化チタン、 塩ィ匕 亜鉛等のルイス酸;例えば p—トルエンスルホン酸、 トリフルォロメタンスルホ ン酸、 メタンスルホン酸等のスルホン酸;蟻酸、 トリフルォロ酢酸、 酢酸等の有 機酸等が挙げられ、 中でも塩酸、 酢酸又は p—トルエンスルホン酸が好適である 反応で使用される乾燥剤しては、 例えばモレキュラーシーブス、 硫酸マグネシ ゥム、 硫酸ナトリウムが挙げられ、 中でもモレキュラーシーブスが好適である。 反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [X XV I I I ] で表される化合物に対し て、 1 . 2〜1 0等量、 好ましくは 1 . 2〜 2等量の乾燥剤、 1〜5 0等量、 好 ましくは 3〜 5等量の酸を使用する。 該酸、 該乾燥剤剤は、 一種又はそれ以上適 宜組み合わせて使用することもできる。 Examples of the acid used in the reaction include inorganic acids such as hydrochloric acid, nitric acid, hydrobromic acid, sulfuric acid, hydrofluoric acid and perchloric acid; for example, Lewis acids such as trifluoroboric acid, titanium tetrachloride, zinc chloride and the like. Sulfonic acids such as p-toluenesulfonic acid, trifluoromethanesulfonic acid, and methanesulfonic acid; organic acids such as formic acid, trifluoroacetic acid, and acetic acid; and hydrochloric acid, acetic acid, and p-toluenesulfonic acid. Suitable desiccants used in the reaction include, for example, molecular sieves, magnesium sulfate, and sodium sulfate, with molecular sieves being preferred. The amount of the reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and the reaction conditions, but is usually 1.2 to 10 equivalents to the compound represented by the above general formula [XXVIII]. Preferably, 1.2 to 2 equivalents of desiccant, 1 to 50 equivalents, preferably 3 to 5 equivalents of acid are used. The acid and the desiccant may be used alone or in appropriate combination of two or more.
(第 3工程) (3rd step)
一般式 [X X I X] General formula [X X I X]
[式中、 R 1 R 2、 R 3、 A rは、 前記の意味を有する] で表される化合物と八 ロゲン化ァリール化合物をマグネシウム、 リチウム、 銅、 亜鉛の様な金属で活性 化した化合物を、 適当なエーテル系脱水溶媒中、 低温から溶媒の沸点温度で反応 させることにより一般式 [XXV] [Wherein, R 1 R 2 , R 3 and Ar have the above-mentioned meanings] and a compound obtained by activating a halogenated aryl compound with a metal such as magnesium, lithium, copper or zinc. Is reacted in a suitable ether-based dehydrating solvent at a low temperature to the boiling point of the solvent to obtain the general formula [XXV]
〔式中、 R1 R2、 R3、 R5、 Arは、 前記の意味を有する〕 で表される化合物 を合成することができる。 [Wherein, R 1 R 2 , R 3 , R 5 , and Ar have the above-mentioned meanings].
得られた一般式 [XXV] で表される化合物を用いて製造法 Aの第 4工程の方 法により上記一般式 [I] で表される化合物が合成できる。 製造法 G Using the obtained compound represented by the general formula [XXV], the compound represented by the above general formula [I] can be synthesized by the method of the fourth step of the production method A. Manufacturing method G
本製造法は、 一般式 [I] で表される化合物のうち、 R4が 一 C3アルキル のものについての化合物の製造法である。 This production method is a method for producing a compound represented by the general formula [I] wherein R 4 is 1 C 3 alkyl.
一般式 [V I I] General formula [V I I]
[式中、 R1 R2、 R3、 R5、 Arは、 前記の意味を有する] で表される化合物 と一般式 [XXX] [Wherein, R 1 R 2 , R 3 , R 5 , and Ar have the above-mentioned meanings] and a general formula [XXX]
R4L3 [XXX] R 4 L 3 [XXX]
[式中、 R4は 一 C3アルキル、 L3は、 ハロゲン等の脱離基を示す] で表され るアルキル八ライドを反応させることにより一般式 [XXX I] [Wherein, R 4 represents one C 3 alkyl and L 3 represents a leaving group such as halogen] by reacting an alkyl octride represented by the general formula [XXX I]
[式中、 R1 R2、 R3、 R4、 R5、 Arは、 前記の意味を有する] で表される 4級塩を合成できる。 [Wherein, R 1 R 2 , R 3 , R 4 , R 5 , and Ar have the above-mentioned meanings] A quaternary salt can be synthesized.
反応に用いる試薬は、 原料化合物及び反応条件によって適宜増減することがで きるが、 通常反応は、 上記一般式 [V I I] で表される化合物と上記一般式 [X XX] で表されるアルキルハラィドを不活性な有機溶媒中で、 5〜96時間、 好 ましくは 12〜24時間加熱還流させることにより、 反応は完結する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions. Usually, the reaction is carried out by reacting the compound represented by the above general formula [VII] with the alkyl halide represented by the above general formula [XX]. The reaction is completed by heating to reflux in an inert organic solvent for 5 to 96 hours, preferably 12 to 24 hours.
反応で使用される有機溶媒としては、 反応に悪影響を及ぼさないものであれば 、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム、 1, 2—ジクロロェタン、 トリクロロェタン、 N, N—ジメチルホルムアミド、 無水 酢酸、 酢酸ェチルエステル、 酢酸メチルエステル、 ァセトニ卜リル、 メチルアル コール、 エチルアルコール、 n—プロパノール、 ベンゼン、 キシレン、 トルエン 、 キシレン、 1, 4一ジォキサン、 テトラヒドロフラン等が挙げられ、 中でもァ セトニトリル、 トルエン、 キシレン等が好適である。 The organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction. Specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, trichloroethane, and N , N-dimethylformamide, acetic anhydride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, n-propanol, benzene, xylene, toluene, xylene, 1,4-dioxane, tetrahydrofuran, and the like. Of these, acetonitrile, toluene, xylene and the like are preferred.
反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [V I I] で表される化合物に対して、 1 〜50等量、 好ましくは 10〜50等量の上記一般式 [XXX] で表される化合 物を使用する。 The reagent used in the reaction can be appropriately increased or decreased depending on the starting compound and reaction conditions, but is usually 1 to 50 equivalents, preferably 10 to 10 equivalents to the compound represented by the above general formula [VII]. Use 50 equivalents of the compound represented by the above general formula [XXX].
得られた上記一般式 [XXX] で表される化合物を用いて製造法 A第 5工程の 方法により上記一般式 [I] の化合物が合成できる。 製造法 H Using the obtained compound represented by the above general formula [XXX], the compound of the above general formula [I] can be synthesized by the method of the fifth step of Production method A. Manufacturing method H
本製造法は、 一般式 [I] で表される化合物のうち、 R4が水酸基のものにつ いての化合物の製造法である。 This production method is a method for producing a compound represented by the general formula [I] wherein R 4 is a hydroxyl group.
一般式 [XXV] General formula [XXV]
[式中、 R R2、 R3、 R5 A rは前記の意味を有する] で表される化合物を 適宜酸化剤と反応させ上記一般式 [ I ] で表される化合物が合成できる。 [Wherein RR 2 , R 3 , and R 5 Ar have the meaning described above] The compound represented by the above general formula [I] can be synthesized by appropriately reacting with an oxidizing agent.
反応で使用する試薬は、 原料化合物及び反応条件によって適宜増減することが できるが、 通常、 反応は例えば塩化メチレンの様な適当な不活性有機溶媒中、 例 えばメタク口口過安息香酸のような適当な酸化剤の存在下室温でで 5〜 9 6時間 、 好ましくは 1 2〜2 4時間攪拌させることにより、 反応は完結する。 The reagents used in the reaction can be appropriately increased or decreased depending on the starting compounds and reaction conditions.Normally, the reaction is carried out in a suitable inert organic solvent such as methylene chloride, for example, The reaction is completed by stirring at room temperature for 5 to 96 hours, preferably 12 to 24 hours, in the presence of a suitable oxidizing agent.
反応で使用される不活性有機溶媒としては、 反応に悪影響を及ぼさないもので あれば、 特に限定はされないが、 具体的には例えば塩化メチレン、 クロ口ホルム 、 1, 2—ジクロロェタン、 トリクロロェタン、 四塩化炭素、 酢酸ェチルエステ ル、 酢酸メチルエステル、 ァセトニトリル、 メチルアルコール、 ェチルアルコー ル、 ベンゼン、 キシレン、 トルエン、 キシレン、 エーテル、 1, 4一ジォキサン 、 テトラヒドロフラン、 テトラヒドロピラン等が挙げられ、 中でも塩ィ匕メチレン 、 クロ口ホルム、 1, 2—ジクロロェタン、 トリクロロェタン、 酢酸ェチルエス テル等が好適である。 The inert organic solvent used in the reaction is not particularly limited as long as it does not adversely affect the reaction, and specific examples thereof include methylene chloride, chloroform, 1,2-dichloroethane, and trichloroethane. , Carbon tetrachloride, ethyl acetate, methyl acetate, acetonitrile, methyl alcohol, ethyl alcohol, benzene, xylene, toluene, xylene, ether, 1,4-dioxane, tetrahydrofuran, tetrahydropyran and the like. Preferred are methylene, chloroform, 1,2-dichloroethane, trichloroethane, ethyl acetate and the like.
反応で使用される酸化剤としては、 反応に悪影響を及ぼさないものであれば、 特に限定はされないが、 具体的には例えば過酸化水素、 過酢酸、 トリフルォロ過 酢酸、 メタクロル過安息香酸、 ォキソン、 クロム酸、 過マンガンサン等が挙げら れ、 中でも特に例えば過酸化水素、 メタクロル過安息香酸等が好適である。 反応において使用する試薬は、 原料化合物及び反応条件によって、 適宜増減す ることができるが、 通常、 上記一般式 [X XV] で表される化合物に対して 1 . 2〜 1 0等量、 好ましくは 1 . 2〜 2等量の酸化剤を使用する。 The oxidizing agent used in the reaction is not particularly limited as long as it does not adversely affect the reaction.Specifically, for example, hydrogen peroxide, peracetic acid, trifluoroperacetic acid, methachloroperbenzoic acid, oxone, Examples thereof include chromic acid and permanganese sun. Among them, for example, hydrogen peroxide, metachloroperbenzoic acid, and the like are particularly preferable. The reagent used in the reaction can be appropriately increased or decreased depending on the raw material compound and the reaction conditions, but is usually 1.2 to 10 equivalents, preferably 1.2 to 10 equivalents to the compound represented by the above general formula [XXV]. Use 1.2 to 2 equivalents of oxidizing agent.
次に、 本発明の有用性を具体的に示すために、 化合物例 1 1 1 6の化合物を代 表化合物とし、 該化合物投与後の血しょう中の G L P— 1濃度への影響を検討し た。 Next, in order to specifically demonstrate the usefulness of the present invention, the compound of Compound Example 116 was used as a representative compound, and the effect on GLP-1 concentration in plasma after administration of the compound was examined. .
その試験方法及び結果を以下に示す。 The test method and results are shown below.
(試験方法) (Test method)
自由摂食、 摂水条件で飼育した雄性ウィスターラット (第 9週令、 n = 6 ) を 、 試験前夜から絶食し、 1 %カルボキシメチルセルロース溶液に懸濁した化合物 を投与した。 なお、 対照群としては、 1 %カルボキシメチルセルロース溶液をラ ットに経口投与した。 被検薬剤投与 3 0分後に採血を行い、 得られた血液から遠 心分離を行って血漿を分離した。 血漿中の GLP— 1濃度は市販の抗 GLP— 1 抗体 (コスモバイオ (株) )を用い、 ラジオィムノアッセィ法で定量した。 得られ た数値は、 S t ud en t T検定を用いて解析を行い、 その統計学的有意差を 算出した。 その結果を下記の表に示す。 Male Wistar rats (9-week old, n = 6) bred under free-feeding and water-feeding conditions were fasted the night before the test, and administered with a compound suspended in a 1% carboxymethylcellulose solution. As a control, a 1% carboxymethylcellulose solution was orally administered to rats. Blood was collected 30 minutes after administration of the test drug, and was separated from the obtained blood. Plasma was separated by cardiac separation. GLP-1 concentration in plasma was determined by a radioimmunoassay method using a commercially available anti-GLP-1 antibody (Cosmo Bio Inc.). The obtained values were analyzed using the Student T test, and the statistical significance was calculated. The results are shown in the table below.
(試験結果) (Test results)
表 9 Table 9
対照群 111610mg/kg群 以上の結果より、 投与後 30分に於いて、 1 Omg/kgの化合物を投与され た群の血漿では対照群と比較して有意に高値な血中 GLP_ 1が認められた。 こ の結果より、 本発明の化合物はラットにおいて高い血中 G LP— 1濃度を呈する ことの出来る活性を持つことが示された。 Control group 111 610 mg / kg group Based on the above results, 30 minutes after administration, plasma GLP_1 was significantly higher in the plasma of the group to which 1 Omg / kg of the compound was administered compared to the control group. Was. From these results, it was shown that the compound of the present invention has an activity capable of exhibiting a high blood GLP-1 concentration in rats.
本発明化合物は、 高い血中 G L P - 1濃度を呈する活性を示していることから 、 糖尿病治療剤、 糖尿病の慢性合併症の予防剤又は抗肥満薬として有用である。 本発明の一般式 [I]の化合物は、 それを有効成分とする医薬、 特に糖尿病治療 剤、 糖尿病の慢性合併症の予防剤又は抗肥満薬として使用することができるが、 かかる医薬、 特に糖尿病治療剤、 糖尿病の慢性合併症の予防剤又は抗肥満剤にお ける本発明の化合物は、 医薬上許容される慣用的なものを意味し、 例えば一般式 [ I ] The compound of the present invention is useful as a therapeutic agent for diabetes mellitus, a preventive for chronic complications of diabetes or an antiobesity drug, since it exhibits an activity of exhibiting a high blood GLP-1 concentration. The compound of the general formula [I] of the present invention can be used as a medicament containing the same as an active ingredient, particularly as an agent for treating diabetes, an agent for preventing chronic complications of diabetes or an antiobesity agent. The compound of the present invention in a therapeutic agent, a preventive agent for chronic complications of diabetes or an anti-obesity agent means a pharmaceutically acceptable conventional one, For example, the general formula [I]
[式中、 R R 2、 R 3、 R 4、 R 5、 A r及び nは前記の意味を有する。 ]で表さ れる化合物、 R 5又は A r上の力ルポキシル基における医薬として許容されるェ ステル若しくは塩、 R 5又は A r上の水酸基における塩、 又は R 5又は A r上の アミノ基における塩が包含される。 [Wherein, RR 2 , R 3 , R 4 , R 5 , Ar and n have the above-mentioned meanings. Compounds represented by, E ester or a pharmaceutically acceptable salt in the force Rupokishiru group on R 5 or A r, salts in hydroxyl groups on R 5 or A r, or at the amino group on R 5 or A r Salts are included.
該カルポキシル基又はヒドロキシ基における塩としては、 例えばナトリウム塩 、 カリウム塩等のアルカリ金属塩;例えばカルシウム塩、 マグネシウム塩等のァ ルカリ土類金属塩等が挙げられる。 Examples of the salt in the carboxyl group or the hydroxy group include alkali metal salts such as sodium salt and potassium salt; and alkaline earth metal salts such as calcium salt and magnesium salt.
該ァミノ基における酸付加塩としては、 例えば塩酸塩、 硫酸塩、 硝酸塩、 りん 酸塩、 炭酸塩、 炭酸水素塩、 過塩素酸塩等の無機酸塩;例えば酢酸塩、 プロピオ ン酸塩、 乳酸塩、 マレイン酸塩、 フマール酸塩、 酒石酸塩、 りんご酸塩、 くえん 酸塩、 ァスコルビン酸塩等の有機酸塩;例えばメタンスルホン酸塩、 イセチオン 酸塩、 ベンゼンスルホン酸塩、 トルエンスルホン酸塩等のスルホン酸塩;例えば ァスパラギン酸塩、 グルタミン酸塩等の酸性アミノ酸塩等が挙げられる。 Examples of the acid addition salt for the amino group include inorganic salts such as hydrochloride, sulfate, nitrate, phosphate, carbonate, hydrogencarbonate and perchlorate; for example, acetate, propionate and lactic acid Organic salts such as salt, maleate, fumarate, tartrate, malate, citrate, and ascorbate; for example, methanesulfonate, isethionate, benzenesulfonate, toluenesulfonate, etc. Sulfonic acid salts; for example, acidic amino acid salts such as aspartate and glutamate.
本発明化合物は、 糖尿病治療剤、 糖尿病の慢性合併症の予防剤あるいは抗肥満 薬として使用される場合には、 その薬学的に許容しうる塩としても使用すること ができる。 薬学的に許容しうる塩の典型例としては、 例えばナトリウム、 力リウ ム等のアルカリ金属との塩等を挙げることができる。 When the compound of the present invention is used as a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes, or an antiobesity agent, it can also be used as a pharmaceutically acceptable salt thereof. Typical examples of the pharmaceutically acceptable salts include, for example, salts with alkali metals such as sodium and potassium.
本発明の化合物の薬学的に許容しうる塩の製造法は、 有機合成化学分野で通常 用いられる方法を適宜組み合わせて行うことができる。 具体的には、 本発明化合 物の遊離型の溶液をアル力リ溶液で中和滴定すること等が挙げられる。 The method for producing a pharmaceutically acceptable salt of the compound of the present invention can be carried out by appropriately combining methods usually used in the field of synthetic organic chemistry. Specific examples include neutralization titration of a free solution of the compound of the present invention with an alkaline solution.
本発明化合物を糖尿病治療剤、 糖尿病の慢性合併症の予防剤あるいは抗肥満薬 として使用する際の投与形態としては各種の形態を選択でき、 例えば錠剤、 カブ セル剤、 散剤、 顆粒剤、 液剤等の経口剤、 例えば溶液、 懸濁液等の殺菌した液状 の非経口剤等が挙げられる。 Compounds of the present invention for treating diabetes, preventing chronic complications of diabetes or treating obesity Various forms can be selected as the dosage form for use as a tablet, for example, oral preparations such as tablets, capsules, powders, granules, and liquid preparations, and sterilized liquid parenteral preparations such as solutions and suspensions. Is mentioned.
固体の製剤は、 そのまま錠剤、 カプセル剤、 顆粒剤又は粉末の形態として製造 することもできるが、 適当な添加物を使用して製造することもできる。 該添加物 としては、 例えば乳糖、 ブドウ糖等の糖類、 例えばトウモロコシ、 小麦、 米等の 澱粉類、 例えばステアリン酸等の脂肪酸、 例えばメタケイ酸ナトリウム、 アルミ ン酸マグネシウム、 無水リン酸カルシウム等の無機塩、 例えばポリビニルピロリ ドン、 ポリアルキレングリコール等の合成高分子、 例えばステアリン酸カルシゥ ム、 ステアリン酸マグネシウム等の脂肪酸塩、 例えばステアリルアルコール、 ベ ンジルアルコール等のアルコール類、 例えばメチルセルロース、 カルポキシメチ ルセルロース、 ェチルセルロース、 ヒドロキシプロピルメチルセルロース等の合 成セルロース誘導体、 その他、 水、 ゼラチン、 タルク、 植物油、 アラビアゴム等 通常用いられる添加物等が挙げられる。 Solid preparations can be produced as they are in the form of tablets, capsules, granules or powders, or they can be produced using appropriate additives. Examples of the additive include sugars such as lactose and glucose; starches such as corn, wheat and rice; fatty acids such as stearic acid; inorganic salts such as sodium metasilicate, magnesium aluminate and anhydrous calcium phosphate; Synthetic polymers such as polyvinylpyrrolidone and polyalkylene glycol; fatty acids such as calcium stearate and magnesium stearate; alcohols such as stearyl alcohol and benzyl alcohol; methyl cellulose, carboxymethyl cellulose, and ethyl cellulose And synthetic cellulose derivatives such as hydroxypropylmethylcellulose, and other commonly used additives, such as water, gelatin, talc, vegetable oil, and gum arabic.
これらの錠剤、 カプセル剤、 顆粒剤、 粉末等の固形製剤は、 一般的には 0 . 1 〜1 0 0重量%、 好ましくは 5〜1 0 0重量%の有効成分を含むことができる。 液状製剤は、 水、 アルコール類又は例えば大豆油、 ピーナツ油、 ゴマ油等の植物 由来の油等液状製剤において通常用いられる適当な添加物を使用し、 懸濁液、 シ ロップ剤、 注射剤等の形態として製造することができる。 特に、 非経口的に投与 する場合の適当な溶剤としては、 例えば注射用蒸留水、 塩酸リドカイン水溶液 ( 筋肉内注射用) 、 生理食塩水、 ブドウ糖水溶液、 エタノール、 静脈内注射用液体 (例えばクェン酸、 クェン酸ナトリウム等の水溶液) 、 電解質溶液 (例えば点滴 静注、 静脈内注射用) 等又はこれらの混合溶液が挙げられる。 These tablets, capsules, granules, powders and other solid preparations can generally contain 0.1 to 100% by weight, preferably 5 to 100% by weight, of the active ingredient. For liquid preparations, use appropriate additives normally used in liquid preparations such as water, alcohols or plant-derived oils such as soybean oil, peanut oil, sesame oil, etc. It can be manufactured as a form. Particularly suitable solvents for parenteral administration include, for example, distilled water for injection, aqueous lidocaine hydrochloride (for intramuscular injection), physiological saline, aqueous dextrose, ethanol, and liquid for intravenous injection (for example, quinic acid). , Sodium citrate, etc.), an electrolyte solution (for example, intravenous drip, intravenous injection) and the like, or a mixed solution thereof.
又、 経口投与の懸濁剤又はシロップ剤等の液剤は、 0 . 5〜1 0重量%の有効 成分を含むことができる。 Liquid preparations such as suspensions or syrups for oral administration may contain 0.5 to 10% by weight of active ingredients.
本発明の化合物の実際に好ましい投与量は、 使用される化合物の種類、 配合さ れた組成物の種類、 適用頻度および治療すべき特定部位および患者の病状によつ て適宜増減することができる。 例えば、 一日当りの成人一人当りの投与量は、 経 口投与の場合、 0 . 1ないし 1 0 0 O m gであり、 非経口投与の場合、 1日当り 0. 01ないし 50 Omgである。 なお、 投与回数は、 投与方法および症状によ り異なるが、 単回又は 2ないし 5回に分けて投与することができる。 発明を実施するための最良の形態 The practically preferred dosage of the compounds of the present invention may be adjusted as appropriate depending on the type of compound used, the type of composition formulated, the frequency of application and the particular site to be treated and the condition of the patient. . For example, the daily dose per adult per day is 0.1 to 100 mg for oral administration and 100 mg / day for parenteral administration. It is 0.01 to 50 Omg. The frequency of administration varies depending on the administration method and symptoms, but it can be administered once or in 2 to 5 doses. BEST MODE FOR CARRYING OUT THE INVENTION
実施例を挙げて本発明を更に具体的に説明するが、 本発明はこれらによって何 ら限定されるものではない。 The present invention will be described more specifically with reference to examples, but the present invention is not limited thereto.
実施例の薄層クロマトグラフは、 プレートとして S i 1 i c a g e 1 60 F The thin-layer chromatograph of the example has a plate of Si 1 i c ag e 160 F
245 (Me r c k) を、 検出法として UV検出器を用いた。 カラム用シリカゲル としては、 Wa k o g e 1 TM C— 300 (和光純薬) を、 逆相カラム用シリカ ゲルとしては、 LC— SORBTM S P-B-ODS (Chemc o) または Y 245 (Merck) was detected using a UV detector. The silica gel for the column is Wakoge 1 ™ C-300 (Wako Pure Chemical Industries), and the silica gel for the reversed-phase column is LC-SORB ™ S PB-ODS (Chemco) or Y
MC-GEL™ ODS-AQ 120 _ S 50 (山村化学研究所) を用いた。 i -Bu :イソブチル基 MC-GEL ™ ODS-AQ 120 _ S 50 (Yamamura Chemical Laboratory) was used. i-Bu: isobutyl group
n-B u: n—ブチル基 n-Bu: n-butyl group
t -Bu: t一ブチル基 t-Bu: t-butyl group
Me :メチル基 Me: methyl group
E t :ェチル基 E t: ethyl group
Ph:フエニル基 Ph: phenyl group
i— P r :イソプロピル基 i—Pr: isopropyl group
n— P r: n—プロピル基 n—Pr: n—propyl group
CDC 13:重クロ口ホルム CDC 1 3: heavy black hole Holm
me t h ano l— d4 : メ夕ノ一レ me th ano l— d 4
DMSO— d6:重ジメチルスルホキシド 実施例 1 DMSO-d 6: deuterated dimethyl sulfoxide Example 1
化合物例 1016の製造 ( 3一^ Γソプロピル一 5—フエ二ルー 2, 3, 4, 5 - テトラヒドロー Iff一 1 6, 4—ベンゾチアゼピン _ 1, 1ージオン) Preparation of Compound Example 1016 (3 ^^-sopropyl-1-5-phenyl-2,3,4,5-tetrahydro-Iff-1 16 , 4-benzothiazepine _1,1dione)
ジクロロメタン (300ml) および 2—ァミノ一 3—メチル一1—プタノ一 ル (50. 0 g, 485mmo 1) を窒素雰囲気下混合し、 0°Cで塩化スルホン 酸 (35. 0ml , 524mmo 1 ) を滴下した。 添加終了後、 混合物を室温で 2時間攪拌した。 水 (100ml) および 50%τΚ酸化ナトリウム水溶液 (10 O g) を添加し、 混合物を大気圧下で蒸留した。 有機層を蒸留物から分離し、 固 体水酸ィヒカリウムで乾燥し、 所望の生成物 2—イソプロピルアジリジン (36. 3 g、 426mmo 1) を得た (収率 88%) 。 Dichloromethane (300 ml) and 2-amino-3-methyl-1-butanol (50.0 g, 485 mmo 1) are mixed under a nitrogen atmosphere, and sulfonic acid chloride (35.0 ml, 524 mmo 1) is added at 0 ° C. It was dropped. After the addition is complete, mix the mixture at room temperature. Stir for 2 hours. Water (100 ml) and 50% aqueous τΚ sodium oxide solution (10 Og) were added and the mixture was distilled at atmospheric pressure. The organic layer was separated from the distillate and dried over solid potassium hydroxide to give the desired product 2-isopropylaziridine (36.3 g, 426 mmo 1) (88% yield).
^NMR (CDC 13) : 0. 97 (3H, d, J = 6. 2Hz) , 1. 0 ^ NMR (CDC 1 3): 0. 97 (3H, d, J = 6. 2Hz), 1. 0
3 (3H, d, J = 5. 9Hz) , 1. 04— 1. 20 ( 1 H, m) , 1. 35 — 1. 37 (1H, m) , 1. 71-1. 77 (2H, m) 3 (3H, d, J = 5.9Hz), 1.04-1.20 (1H, m), 1.35-1.37 (1H, m), 1.71-1.77 (2H, m)
N, N, N, , N, —テトラメチルエチレンジァミン (TMEDA) (63. 9ml , 424mmo 1 ) のシクロへキサン (300ml) 溶液を冷却し、 n— ブチルリチウムの 2. 5Mシクロへキサン溶液 (170ml) を添加した。 チォ フエノール (23. 3 g, 212mmo 1 ) を徐々に添加し、 反応物をー晚室温 で攪拌した。 ベンゾニトリル (21. 8 g, 212 mm 01 ) を添加するとスラ リーが得られ、 これを室温で一晩攪拌した。 水 (200ml) を添加し、 混合物 を 30分間攪拌した後、 水層を分離し、 固体の水酸化ナトリウムで処理し、 pH を 14にした。 この溶液を 90分間沸縢させ、 室温にまで冷却し濃塩酸により p Hl〜2に酸性化した。 この酸性溶液をジクロロメタンで抽出し、 合わせた抽出 物を乾燥し、 濃縮し、 赤色油を得た。 この油を 1M水酸化ナトリウム水溶液で処 理し、 ジクロロメタンで抽出し、 水槽を分離し、 濃塩酸で処理し、 油を得た。 こ の油をジクロロメタン中に抽出し、 抽出物を集めて乾燥し、 減圧下に濃縮し、 所 望の橙赤色の油状生成物フエニル (2—スルファニルフエニル) メタノン (36 . 4 g, 17 Ommo 1 ) を得た (収率 80 %) 。 Cool a solution of N, N, N,, N, —tetramethylethylenediamine (TMEDA) (63.9 ml, 424 mmo 1) in cyclohexane (300 ml), and cool n-butyllithium to 2.5 M cyclohexane. A solution (170 ml) was added. Thiophanol (23.3 g, 212 mmo 1) was added slowly and the reaction was stirred at room temperature. Addition of benzonitrile (21.8 g, 212 mm 01) gave a slurry which was stirred at room temperature overnight. After adding water (200 ml) and stirring the mixture for 30 minutes, the aqueous layer was separated and treated with solid sodium hydroxide to a pH of 14. The solution was boiled for 90 minutes, cooled to room temperature, and acidified to pH 〜2 with concentrated hydrochloric acid. The acidic solution was extracted with dichloromethane, and the combined extracts were dried and concentrated to give a red oil. This oil was treated with a 1 M aqueous sodium hydroxide solution, extracted with dichloromethane, the water tank was separated, and treated with concentrated hydrochloric acid to obtain an oil. The oil was extracted into dichloromethane, the extracts were combined, dried and concentrated under reduced pressure to give the desired orange-red oily product phenyl (2-sulfanylphenyl) methanone (36.4 g, 17 Ommo). 1) was obtained (80% yield).
^NMR (CDC 13) δ : 4. 21 ( 1 Η, s) , 7. 14-7. 25 (1 H, m) , 7. 31-7. 39 ( 1 H, m) , 7. 40- 7. 63 (5H, m) , 7. 75 - 7. 80 (2H, m) ^ NMR (CDC 1 3) δ :.. 4. 21 (1 Η, s), 7. 14-7 25 (1 H, m), 7. 31-7 39 (1 H, m), 7. 40 -7.63 (5H, m), 7.75-7.80 (2H, m)
FAB-MS (mZe) : 215 [M + H] + FAB-MS (mZe): 215 [M + H] +
工程 1で得られた化合物 (0. 3ml) を工程 2の生成物 (300mg, 1. The compound obtained in Step 1 (0.3 ml) was combined with the product of Step 2 (300 mg, 1.
4 Ommo 1) の 2, 6—ルチジン (3. Oml ) 懸濁液に室温で滴下した。 添 加終了後、 混合物を室温で 3時間攪拌し、 濃塩酸 (1. Oml) を添加した。 添 加終了後、 混合物を 3時間共沸させ、 減圧留去した。 残渣を飽和炭酸水素ナトリ ゥム水溶液で回収し、 溶液を酢酸ェチルで 2回抽出した。 集めた抽出物を飽和食 塩水溶液で洗浄し、 乾燥し、 減圧下に濃縮した。 残渣をシリカゲル上で、 へキサ ン Z酢酸ェチル (90Z10) を溶離剤とするカラムクロマトグラフィーに供し 、 淡黄色の油状物質 3—イソプロピル一5—フエニル一 2, 3—ジヒドロー 1, 4一べンゾチアゼピン (266mg, 0. 947 mm o 1 ) を得た (収率 68 %4 Ommo 1) was added dropwise to a suspension of 2,6-lutidine (3. Oml) at room temperature. After the addition was completed, the mixture was stirred at room temperature for 3 hours, and concentrated hydrochloric acid (1. Oml) was added. After the addition was completed, the mixture was azeotropically distilled for 3 hours and distilled under reduced pressure. The residue was saturated with sodium bicarbonate The reaction solution was collected with an aqueous solution of water, and the solution was extracted twice with ethyl acetate. The combined extracts were washed with saturated brine solution, dried and concentrated under reduced pressure. The residue was subjected to column chromatography on silica gel using hexane Z ethyl acetate (90Z10) as an eluent to give a pale yellow oily substance, 3-isopropyl-15-phenyl-1,2,3-dihydro-1,4-benzothiazepine. (266 mg, 0.947 mmo 1) (yield 68%
) o ) o
:HNMR (CDC 13) <5 : 0. 95 (3H, d, J = 6. 7Hz) , 1. 0 6 (3H, d, J = 6. 7Hz) , 1. 94— 2. 1 1 ( 1 H, m) , 2. 96 (1H, d d d, J = 12. 3, 6. 5, 4. 0Hz) , 3. 27 (1H, d d , J = 12. 3, 11. 3Hz) , 3. 62 ( 1 H, d d, J = 11. 3, 4. 0Hz) , 7. 09-7. 15 (1H, m) , 7. 32-7. 64 (8H, m) FAB-MS (mZe) : 282 [M + H] + : HNMR (CDC 1 3) < 5: 0. 95 (3H, d, J = 6. 7Hz), 1. 0 6 (3H, d, J = 6. 7Hz), 1. 94- 2. 1 1 ( 1H, m), 2.96 (1H, ddd, J = 12.3, 6.5, 4.0Hz), 3.27 (1H, dd, J = 12.3, 11.3Hz), 3. 62 (1H, dd, J = 11.3, 4.0Hz), 7.09-7.15 (1H, m), 7.32-7.64 (8H, m) FAB-MS (mZe): 282 [M + H] +
工程 3の生成物 (180mg, 0. 64 Ommo 1) のトリフルォロ酢酸 (3 . 0 ml) 溶液に、 30 %過酸化水素水 ( 1. 0ml) を約 0 の温度で添加し た。 添加終了後、 混合物を室温で一晩攪拌し、 10%チォ硫酸ナトリウム水溶液 、 飽和炭酸水素ナトリウム水溶液で処理し、 酢酸ェチルで 2回抽出し、 抽出物を 集めて飽和食塩水溶液で洗浄し、 乾燥し、 減圧下に濃縮した。 残渣および 4 Ν塩 化水素 1, 4—ジォキサン溶液 (3. 0ml) を混合し、 亜鉛粉末 (200mg , 3. 06mmo 1) を徐々に添加した。 混合液をー晚室温で攪拌し、 飽和炭酸 水素ナトリウム水溶液で処理し、 セライトで濾過した。 酢酸ェチルで 2回抽出し 、 抽出物を集めて飽和食塩水溶液で洗浄し、 乾燥し、 減圧下に濃縮した。 残渣を シリカゲル上で、 へキサン Z酢酸ェチル (80/20) を溶離剤とするカラムク 口マトグラフィ一に供し、 所望の化合物 3—イソプロピル一 5—フエ二ルー 2, 3, 4, 5—テトラヒドロー Iff— 1 λ 6, 4一べンゾチアゼピン— 1, 1ージ オンのジァステレオマー a (35. 8mg, 0. 114mmo 1、 収率 18 %) および b (62. 6mg, 0. 199 mmo 1、 収率 31 %) を淡黄色の油状生 成物として得た。 To a solution of the product of step 3 (180 mg, 0.64 Ommo 1) in trifluoroacetic acid (3.0 ml) was added 30% aqueous hydrogen peroxide (1.0 ml) at a temperature of about 0. After the addition was completed, the mixture was stirred at room temperature overnight, treated with a 10% aqueous sodium thiosulfate solution and a saturated aqueous sodium hydrogen carbonate solution, and extracted twice with ethyl acetate. And concentrated under reduced pressure. The residue and 4-hydrogen chloride 1,4-dioxane solution (3.0 ml) were mixed, and zinc powder (200 mg, 3.06 mmol 1) was gradually added. The mixture was stirred at room temperature, treated with a saturated aqueous solution of sodium bicarbonate, and filtered through celite. The extract was extracted twice with ethyl acetate, and the extracts were collected, washed with a saturated saline solution, dried, and concentrated under reduced pressure. The residue was subjected to column chromatography on silica gel using hexane Z ethyl acetate (80/20) as eluent to give the desired compound 3-isopropyl-15-phenyl-2,3,4,5-tetrahydro-Iff - 1 lambda 6, 4 one base Nzochiazepin - 1, -1-di-one Jiasutereoma a (35. 8mg, 0. 114mmo 1 , 18% yield) and b (62. 6mg, 0. 199 mmo 1, yield 31 %) As a pale yellow oily product.
ジァステレオマー a Diastereomer a
^NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 75 - 1. 85 (lH, m) , 3. 07 (1H, d d, J = 11. 4, 14. 3Hz) , 3. 44 ( 1 H, dd, J = 2 . 3, 14. 3Hz) , 3. 49— 3. 58 (1H, m) , 5. 99 (1H, s ) , 6. 68— 6. 71 (lH, m) , 7. 33— 7. 43 (7H, m) , 8. 13— 8. 18 (lH, m) ^ NMR (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6.8Hz), 1.75-1.85 (lH, m), 3.07 (1H, dd, J = 11.4, 14.3Hz), 3.44 (1 H, dd, J = 2.3, 14.3 Hz), 3.49—3.58 (1H, m), 5.99 (1H, s), 6.68—6.71 (lH, m), 7.33—7.43 (7H, m), 8.13—8.18 (lH, m)
FAB-MS (m/e) : 316 [M + H] + FAB-MS (m / e): 316 [M + H] +
ジァステレオマー b Geostereomer b
^NM (CDC 13) (5 : 0. 95 (3H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 7Hz) , 1. 90 -2. 05 ( 1 H, m) , 2. 94 -3. 02 (1H, m) , 3. 42- 3. 50 (2H, m) , 5. 85 (1H, s) , 6. 88-6. 95 (lH, m) , 7. 29- 7. 42 (5H, m) , 7 . 44-7. 50 (2H, m) , 8. 12-8. 20 (lH, m) ^ NM (CDC 1 3) ( 5:. 0. 95 (3H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 7Hz), 1. 90 -2 05 (1 H, m), 2.94 -3. 02 (1H, m), 3.42-3.50 (2H, m), 5.85 (1H, s), 6.88-6.95 (lH, m), 7.29-7.42 (5H, m), 7.44-7.50 (2H, m), 8.12-8.20 (lH, m)
FAB-MS (m/e) : 3 1 6 [M + H] + 以下に実施例 1と同様にして得た化合物の物理定数を示す。 FAB-MS (m / e): 316 [M + H] + The physical constants of the compound obtained in the same manner as in Example 1 are shown below.
実施例 2 Example 2
化合物例 1001の製造 (Ar : Ph ; Rx : H ; R2 : H ; R3 : H ; R4 : H ; R5: Ph; n = 0) Preparation of compound examples 1001 (Ar: Ph; R x : H; R 2: H; R 3: H; R 4: H; R 5: Ph; n = 0)
iHNMR (CDC 13) δ : 2. 77 (1H, ddd, J = 3. 0, 10. 4 , 14. 1Hz) , 2. 86 ( 1 H, ddd, J = 2. 9, 4. 0, 14. 1 H z) , 3. 39 (1H, J =d d d, 2. 9, 10. 4, 14. 1Hz) , 3. 58 (1H, ddd, J = 3. 0, 4. 0, 14. 1Hz) , 5. 71 (1H, s) , 6. 62 - 6. 65 (1H, m) , 7. 03-7. 14 (2H, m) , 7 . 24-7. 40 (5H, m) , 7. 60-7. 64 (lH, m) iHNMR (CDC 1 3) δ: 2. 77 (1H, ddd, J = 3. 0, 10. 4, 14. 1Hz), 2. 86 (1 H, ddd, J = 2. 9, 4. 0, 14.1 Hz), 3.39 (1H, J = ddd, 2.9, 10.4, 14.1 Hz), 3.58 (1H, ddd, J = 3.0, 4.0, 14. 1Hz), 5.71 (1H, s), 6.62-6.65 (1H, m), 7.03-7.14 (2H, m), 7.24-7.40 (5H, m) , 7.60-7.64 (lH, m)
FAB-MS (m/e) : 242 [M + H] + 実施例 3 FAB-MS (m / e): 242 [M + H] + Example 3
化合物例 1002の製造 (Ar r Ph R^ H j R^ H ^^- Me ; R4: H ; R5: Ph; n = 0) ^NMR (CDC 13) 6 : 1. 22 (3H, d, J = 6. 5Hz) , 2. 4 6 (1H, d d, J = 10. 4, 14. 1Hz) , 2. 87 (1H, d d, J = 2. 3, 14. 1Hz) , 3. 48 ( 1 H, J =d Q d, 2. 3, 6. 5, 10 . 4Hz) , 5. 67 (IH, s) , 6. 59— 6. 63 (1H, m) , 7. 0 0— 7. 16 (2H, m) , 7. 26-7. 39 (lH, m) , 7. 40-7. 44 (4H, m) , 7. 61-7. 66 (IH, m) Preparation of Compound Example 1002 (Ar r Ph R ^ H j R ^ H ^^-Me; R 4 : H; R 5 : Ph; n = 0) ^ NMR (CDC 1 3) 6 : 1. 22 (3H, d, J = 6. 5Hz), 2. 4 6 (1H, dd, J = 10. 4, 14. 1Hz), 2. 87 (1H, dd, J = 2.3, 14.1 Hz), 3.48 (1 H, J = d Q d, 2.3, 6.5, 10.4 Hz), 5.67 (IH, s), 6. 59-6.63 (1H, m), 7.00-7.16 (2H, m), 7.26-7.39 (lH, m), 7.40-7.44 (4H, m) , 7.61-7.66 (IH, m)
FAB-MS (m/e) : 256 [M + H] + 実施例 4 FAB-MS (m / e): 256 [M + H] + Example 4
化合物例 1006の製造 (Ar : Ph; R1: i— P r ; R2: H; R3: H; R4 : H; R5: Ph; n = 0) Preparation of compound examples 1006 (Ar: Ph; R 1 : i- P r; R 2: H; R 3: H; R 4: H; R 5: Ph; n = 0)
iHNMR (CDC 13) (5 : 1. 04 (3H, d, J = 6. 8Hz) , 1. 0 7 (3H, d, J = 6. 8Hz) , 1. 90 - 2. 02 (lH, m) , 2. 60 一 2. 68 (IH, m) , 3. 26 ( 1 H, d d, J = 9. 9, 14. 1 Hz) , 3. 44 (IH, d d, J = 2. 7 , 14. 1Hz) , 5. 66 (1H, s ) , 6. 59 - 6. 65 (lH, m) , 7. 00- 7. 40 (7H, m) , 7. 6 0-7. 63 (IH, m) iHNMR (CDC 1 3) (5 : 1. 04 (3H, d, J = 6. 8Hz), 1. 0 7 (3H, d, J = 6. 8Hz), 1. 90 - 2. 02 (lH, m), 2.60-2.68 (IH, m), 3.26 (1H, dd, J = 9.9, 14.1 Hz), 3.44 (IH, dd, J = 2.7) , 14.1 Hz), 5.66 (1H, s), 6.59-6.65 (lH, m), 7.00-7.40 (7H, m), 7.60-7.63 ( IH, m)
FAB-MS (m/e) : 283 [M + H] + 実施例 5 FAB-MS (m / e): 283 [M + H] + Example 5
化合物例 1007の製造 (Ar : Ph; R1: i— P r ; R2: H; R3: H; R' : H; R 5: P h; n=2) Preparation of compound examples 1007 (Ar: Ph; R 1 : i- P r; R 2: H; R 3: H; R ': H; R 5: P h; n = 2)
^NMR (CDC 13) 6 : 1. 09 (3H, d, J = 6. 9Hz) , 1. 1 6 (3H, d, J = 6. 9Hz) 2. 85— 2. 95 (IH, m) , 2. 96 - 3. 03 (1H, m) , 3. 50 (IH, d d, J = 2. 9, 14. 8Hz) , 3. 74 (IH, dd, J =l 1. 1, 14. 8Hz) , 5. 92 (IH, s) , 6. 68 (1H, d, J = 8. 9Hz) , 7. 30— 7. 45 (7H, m) , 8. 13-8. 16 (IH, m) ^ NMR (CDC 1 3) 6 : 1. 09 (3H, d, J = 6. 9Hz), 1. 1 6 (3H, d, J = 6. 9Hz) 2. 85- 2. 95 (IH, m ), 2.96-3.03 (1H, m), 3.50 (IH, dd, J = 2.9, 14.8Hz), 3.74 (IH, dd, J = l 1.1, 14 8Hz), 5.92 (IH, s), 6.68 (1H, d, J = 8.9Hz), 7.30—7.45 (7H, m), 8.13-8.16 (IH , m)
FAB-MS (m/e) : 316 [M + H] + 実施例 6 FAB-MS (m / e): 316 [M + H] + Example 6
化合物例 1015の製造 (Ar : Ph; R1: H; R2: H; R3: i— P r ; R4 : H; R5: Ph ; n = 0)Preparation of compound examples 1015 (Ar: Ph; R 1 : H; R 2: H; R 3: i- P r; R 4: H; R 5: Ph; n = 0)
NMR (CDC 13) δ : 0. 95 (3Η, d, J = 6. 8Hz) , 0. 9 5 (3H, d, J =6. 7Hz) , 1. 90- 2. 05 (lH, m) , 2. 75 -2. 85 (1H, m) , 2. 85-2. 94 (2H, m) , 5. 72 (1H, s) , 6. 72 - 6. 79 (1H, m) , 7. 05-7. 16 (2H, m) , 7 . 24-7. 38 (5H, m) , 7. 48-7. 51 (lH, m) NMR (CDC 1 3) δ: 0. 95 (3Η, d, J = 6. 8Hz), 0. 9 5 (. 3H, d, J = 6 7Hz), 1. 90- 2. 05 (lH, m ), 2.75-2.85 (1H, m), 2.85-2.94 (2H, m), 5.72 (1H, s), 6.72-6.79 (1H, m), 7.05-7.16 (2H, m), 7.24-7.38 (5H, m), 7.48-7.51 (lH, m)
FAB-MS (m/e) : 284 [M + H] + 実施例 7 FAB-MS (m / e): 284 [M + H] + Example 7
化合物例 1017の製造 Preparation of Compound Example 1017
ジァステレオマー a (Ar : Ph R1 : H ; R2 : H ; R: -P r ; R4 : M e ; R 5 : P h; n = 0 ) Jiasutereoma a (Ar: Ph R 1: H; R 2: H; R: -P r; R 4: M e; R 5: P h; n = 0)
XHNMR (CDC 13) δ : 0 91 (3H, d, J = 6. 9Hz) , 1. 0 2 (3H, d, J = 6. 9Hz) , 1. 58- 1. 71 (1H, m) , 2. 19 (3H, s) , 2. 47-2. 54 ( 1 H, m) , 2. 70— 3. 00 (2H, m ) , 5. 97 (1H, s) , 6. 76 - 6. 72 ( 1 H, m) , 7. 22-7. 41 (5H, m) , 7. 60 - 7. 65 (3H, m) X HNMR (CDC 1 3) δ : 0 91 (3H, d, J = 6. 9Hz), 1. 0 2 (3H, d, J = 6. 9Hz), 1. 58- 1. 71 (1H, m ), 2.19 (3H, s), 2.47-2.54 (1H, m), 2.70—3.00 (2H, m), 5.97 (1H, s), 6.76 -6.72 (1H, m), 7.22-7.41 (5H, m), 7.60-7.65 (3H, m)
FAB-MS (m/e) : 298 [M + H] + FAB-MS (m / e): 298 [M + H] +
ジァステレオマー b (Ar i P j R^ H j R^ H j R3 : i一 P r ; R4 :M e ; R5 : Ph ; n = 0) Diastereomer b (Ar i P j R ^ H j R ^ H j R 3 : i-1 Pr; R 4 : Me; R 5 : Ph; n = 0)
XHNMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 9Hz) , 1. 1 7 (3H, d, J = 6. 9Hz) , 1. 58— 1. 71 ( 1 H, m) , 1. 78 (3H, s) , 2. 63— 3. 00 (3H, m) , 6. 03 ( 1 H, s) , 6. 7 6-6. 72 (1H, m) , 7. 22-7. 41 (5H, m) , 7. 65-7. 69 (3H, m) X HNMR (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 9Hz), 1. 1 7 (3H, d, J = 6. 9Hz), 1. 58- 1. 71 (1 H , m), 1.78 (3H, s), 2.63—3.00 (3H, m), 6.03 (1 H, s), 6.76-6.72 (1H, m), 7.22-7.41 (5H, m), 7.65-7.69 (3H, m)
FAB-MS (m/e) : 298 [M + H] + 実施例 8 FAB-MS (m / e): 298 [M + H] + Example 8
化合物例 1031の製造 Preparation of Compound Example 1031
ジァステレオマー a (Ar : Ph ; R1: H; R2: H; R3 i -P r ; R4: H ; R5: 4-OH-Ph; n = 0) Jiasutereoma a (Ar: Ph; R 1 : H; R 2: H; R 3 i -P r; R 4: H; R 5: 4-OH-Ph; n = 0)
XHNMR (CDC 13) δ : 0. 93 (3Η, d, J = 6. 9Hz) , 0. 9 5 (3H, d, J = 6. 8Hz) , 1. 65— 1. 75 (lH, m) , 2. 44 (1H, dd, J = 10. 8, 13. 9Hz) , 2. 89 ( 1 H, d d, J = 2 . 2, 13. 9Hz) , 3. 04 (1H, ddd, J = 2. 2, 5. 2, 10. 8Hz) , 5. 07 (1H, s) , 6. 60- 6. 64 ( 1 H, m) , 6. 83 (2H, d, J = 8. 6Hz) , 7. 02-7. 13 (2H, m) , 7. 27 ( 2H, d, J = 8. 6Hz) , 7. 60— 7. 64 ( 1 H, m) X HNMR (CDC 1 3) δ : 0. 93 (3Η, d, J = 6. 9Hz), 0. 9 5 (3H, d, J = 6. 8Hz), 1. 65- 1. 75 (lH, m), 2.44 (1H, dd, J = 10.8, 13.9Hz), 2.89 (1H, dd, J = 2.2, 13.9Hz), 3.04 (1H, ddd, J = 2.2, 5.2, 10.8 Hz), 5.07 (1H, s), 6.60-6.64 (1H, m), 6.83 (2H, d, J = 8. 6Hz), 7.02-7.13 (2H, m), 7.27 (2H, d, J = 8.6Hz), 7.60—7.64 (1H, m)
FAB-MS (m/e) : 300 [M + H] + FAB-MS (m / e): 300 [M + H] +
ジァステレオマー b (Ar i Ph ^^ H j R^ H j 3 : i -P r ; R4 : H ; R6: 4-OH-Ph; n=0) Diastereomer b (Ar i Ph ^^ H j R ^ H j 3 : i -P r; R 4 : H; R 6 : 4-OH-Ph; n = 0)
XHNMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 8Hz) , 0. 9 4 (3H, d, J = 6. 8Hz) , 1. 95— 2. 05 (1H, m) , 2. 76 一 2. 83 (lH, m) , 2. 87 -2. 94 (2H, m) , 5. 65 (1H, s) , 6. 78 - 6. 82 (lH, m) , 6. 80 (2H, d, J = 8. 6Hz ) , 7. 10-7. 14 (2H, m) , 7. 20 (2H, d, J = 8. 6Hz) , 7. 49 - 7. 52 (1H, m) X HNMR (CDC 1 3) δ : 0. 94 (3H, d, J = 6. 8Hz), 0. 9 4 (3H, d, J = 6. 8Hz), 1. 95- 2. 05 (1H, m), 2.76-2.83 (lH, m), 2.87-2.94 (2H, m), 5.65 (1H, s), 6.78-6.82 (lH, m) , 6.80 (2H, d, J = 8.6 Hz), 7.10-7.14 (2H, m), 7.20 (2H, d, J = 8.6 Hz), 7.49-7. 52 (1H, m)
FAB-MS (m/e) : 300 [M + H] + 実施例 9 FAB-MS (m / e): 300 [M + H] + Example 9
化合物例 1032の製造 ( A r : P h; R 1: H; R 2: H; R 3: i— P r ; R4 : H; R5: 4-OH-Ph; n = 2) Preparation of compound examples 1032 (A r: P h; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 4-OH-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 4Hz) , 0. 9 8 (3H, d, J = 6. 4Hz) , 1. 88 -2. 00 ( 1 H, m) , 2. 92 -3. 00 (lH, m) , 3. 35— 3. 52 (2H, m) , 5. 77 (1H, s) , 6. 83 (2H, d, J = 8. 5Hz) , 6. 92 - 6. 97 (1H, m ) , 7. 18 (2H, d, J = 8. 5Hz) , 7. 45 - 7. 48 (2H, m) , 8. 12-8. 16 (1H, m) X HNMR (CDC 1 3) δ :. 0. 94 (3H, d, J = 6. 4Hz), 0. 9 8 (3H, d, J = 6. 4Hz), 1. 88 -2 00 (1 H , M), 2.92 -3.00 (lH, m), 3.35-3.52 (2H, m), 5.77 (1H, s), 6.83 (2H, d, J = 8.5Hz), 6.92-6.97 (1H, m), 7.18 (2H, d, J = 8.5Hz), 7.45- 7.48 (2H, m), 8.12-8.16 (1H, m)
FAB-MS (m/e) : 332 [M + H] + 実施例 10 FAB-MS (m / e): 332 [M + H] + Example 10
化合物例 1033の製造 (Ar : P ; R1: H; R2: H; R3: i一 P r ; R4 : H; R5: 3 -OH-P h; n=2) Preparation of compound examples 1033 (Ar: P; R 1 : H; R 2: H; R 3: i one P r; R 4: H; R 5: 3 -OH-P h; n = 2)
^NMR (CDC 13) δ : 0. 95 (3H, d, J = 7. 0Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 95-2. 10 ( 1 H, m) , 2. 98 —3. 04 (lH, m) , 3. 40-3. 55 (2H, m) , 5. 82 (1H, s) , 6. 80 (IH, d d, J = 2. 9, 7. 8Hz) , 6. 83 (1H, d , J = 2. 9Hz) , 6. 90 (IH, d, J = 7. 6Hz) , 6. 95— 6. 98 (IH, m) , 7. 25 (1H, d d, J = 7. 6, 7. 8Hz) , 7. 4 7-7. 50 (2H, m) , 8. 13— 8. 16 ( 1 H, m) ^ NMR (CDC 1 3) δ :. 0. 95 (3H, d, J = 7. 0Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 95-2 10 (1 H , M), 2.98 — 3.04 (lH, m), 3.40-3.55 (2H, m), 5.82 (1H, s), 6.80 (IH, dd, J = 2 9, 7.8 Hz), 6.83 (1H, d, J = 2.9 Hz), 6.90 (IH, d, J = 7.6 Hz), 6.95—6.98 (IH, m) , 7.25 (1H, dd, J = 7.6, 7.8Hz), 7.4.7-7.50 (2H, m), 8.13—8.16 (1H, m)
FAB-MS (m/e) : 346 [M + H] + 実施例 11 FAB-MS (m / e): 346 [M + H] + Example 11
化合物例 1034の製造 Preparation of Compound Example 1034
ジァステレオマ一 a (A r : P h; R1: H; R2: H; R3: i一 P r ; R4 : HJiasutereoma one a (A r: P h; R 1: H; R 2: H; R 3: i one P r; R 4: H
; R5: 2-OH-Ph; n=2) R 5 : 2-OH-Ph; n = 2)
XHNMR (CDC 13) (5 : 1. 00 (3H, d, J = 7. 5Hz) , 1. 0 0 (3H, d, J = 7. 5Hz) , 1. 82— 1. 94 ( 1 H, m) , 3. 17 X HNMR (CDC 1 3) ( 5: 1. 00 (3H, d, J = 7. 5Hz), 1. 0 0 (3H, d, J = 7. 5Hz), 1. 82- 1. 94 (1 H, m), 3.17
(IH, dd, J = 11. 3, 14, 5Hz) , 3. 47 ( 1 H, dd, J = 2 . 8, 14. 5Hz) , 3. 48- 3. 58 (lH, m) , 6. 07 (IH, s ) , 6. 80- 6. 85 (2H, m) , 6. 94-6. 97 ( 1 H, m) , 7. 06-7. 10 (IH, m) , 7. 21-7. 28 (lH, m) , 7. 49-7 . 53 (2H, m) , 8. 16-8. 20 ( 1 H, m) (IH, dd, J = 11.3, 14, 5Hz), 3.47 (1H, dd, J = 2.8, 14.5Hz), 3.48-3.58 (lH, m), 6 07 (IH, s), 6.80-6.85 (2H, m), 6.94-6.97 (1H, m), 7.06-7.10 (IH, m), 7. 21-7.28 (lH, m), 7.49-7.53 (2H, m), 8.16-8.20 (1H, m)
FAB-MS (m/e) : 332 [M + H] + ジァステレオマ一 b (Ar : Ph ; R1 : H ; R2 : H ; R3: i -P r ; R4 : H ; R5: 2-OH-Ph ; n=2)FAB-MS (m / e): 332 [M + H] + Jiasutereoma one b (Ar: Ph; R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: 2-OH-Ph; n = 2)
NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 9Hz) , 1. 92 - 2. 05 (1H, m) , 2. 92 -3. 02 (1H, m) , 3. 46 ( 1 H, dd, J = 9. 6, 14. 3Hz) , 3. 57 (1H, d d, J = 1. 9, 14. 3Hz) , 6. 13 ( 1 H, s) , 6. 80 - 6. 86 (2H, m) , 6. 96 - 7. 00 (2H, m) , 7. 1 2-7. 17 (1H, m) , 7. 25-7. 32 ( 1 H, m) , 7. 53-7. 61 (2H, m) , 8. 13-8. 17 (lH, m) NMR (CDC 1 3) δ: 0. 98 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 9Hz), 1. 92 - 2. 05 (1H, m ), 2.92 -3. 02 (1H, m), 3.46 (1H, dd, J = 9.6, 14.3Hz), 3.57 (1H, dd, J = 1.9, 14) 3Hz), 6.13 (1H, s), 6.80-6.86 (2H, m), 6.96-7.00 (2H, m), 7.1 2-7.17 (1H , m), 7.25-7.32 (1H, m), 7.53-7.61 (2H, m), 8.13-8.17 (lH, m)
FAB-MS (m/e) : 332 [M + H] + 実施例 12 FAB-MS (m / e): 332 [M + H] + Example 12
化合物例 1035の製造 Preparation of Compound Example 1035
ジァステレオマー a (Ar : P h; R1: H; R2: H; R3 -P r : R4 : H ; R5: 4-MeO-P ; n=0) Jiasutereoma a (Ar: P h; R 1: H; R 2: H; R 3 -P r: R 4: H; R 5: 4-MeO-P; n = 0)
^NMR (CDC 13) δ 0. 97 (3H, d, J = 6. 7Hz) , 0. 9 ^ NMR (CDC 1 3) δ 0. 97 (3H, d, J = 6. 7Hz), 0. 9
7 (3H, d, J = 6. 7Hz) , 1. 75- 1. 85 (1H, m) , 3. 07 (1H, dd, J =l 1. 4, 14. 3Hz) , 3. 43 ( 1 H, d d, J = 27 (3H, d, J = 6.7 Hz), 1.75-1.85 (1H, m), 3.07 (1H, dd, J = l 1.4, 14.3 Hz), 3.43 ( 1 H, dd, J = 2
. 2, 14. 3Hz) , 3. 51 ( 1 H, d d d, J = 2. 2, 4. 9, 11. 4Hz) , 3. 85 (3H, s) , 5. 81 ( 1 H, s) , 6. 72-6. 76 (lH, m) , 6. 95 (2H, d, J = 8. 8Hz) , 7. 32 (2H, d, J = 8. 8Hz) , 7. 34-7. 41 (2H, m) , 8. 12-8. 16 (1 H, m) 2, 14.3Hz), 3.51 (1H, ddd, J = 2.2, 4.9, 11.4Hz), 3.85 (3H, s), 5.81 (1H, s) , 6.72-6.76 (lH, m), 6.95 (2H, d, J = 8.8Hz), 7.32 (2H, d, J = 8.8Hz), 7.34-7. 41 (2H, m), 8.12-8.16 (1 H, m)
FAB-MS (m/e) : 346 [M + H] + FAB-MS (m / e): 346 [M + H] +
ジァステレオマー b (Ar i Ph j R^ H j R' i H j R3 : i—P r ; R4 : H ; R5: 4-MeO-Ph; n = 0) Diastereomer b (Ar i Ph j R ^ H j R 'i H j R 3 : i—P r; R 4 : H; R 5 : 4-MeO-Ph; n = 0)
iHNMR (CDC 13) d : 0. 95 (3H, d, J = 6. 7Hz) , 0. 9 iHNMR (CDC 1 3) d: 0. 95 (3H, d, J = 6. 7Hz), 0. 9
8 (3H, d, J = 6. 7Hz) , 1. 90— 2. 03 (1H, m) , 2. 98 (1 H, d d d, J = 3. 7, 5. 6, 9. 1 Hz) , 3. 43 (1H, d d, J = 9. 1, 14. 6Hz) , 3. 50 ( 1 H, dd, J = 3. 7, 14. 6H z) , 3. 82 (3H, s) , 5. 79 (IH, s) , 6. 88 - 6. 96 (1 H, m) , 6. 91 (2H, d, J = 8. 8Hz) , 7. 24 (2H, d, J = 8. 8Hz) , 7. 43 - 7. 50 (2H, m) , 8. 13-8. 17 (IH, m) 8 (3H, d, J = 6.7 Hz), 1.90— 2.03 (1H, m), 2.98 (1 H, ddd, J = 3. 7, 5.6, 9.1 Hz) , 3.43 (1H, dd, J = 9.1, 14.6Hz), 3.50 (1H, dd, J = 3.7, 14.6Hz), 3.82 (3H, s), 5.79 (IH, s), 6.88-6.96 (1H, m), 6.91 (2H, d, J = 8.8Hz), 7.24 (2H, d, J = 8.8Hz), 7.43-7. 50 (2H, m), 8.13-8.17 (IH, m)
FAB-MS (m/e) : 346 [M + H] + 実施例 13 FAB-MS (m / e): 346 [M + H] + Example 13
化合物例 1036の製造 (Ar : Ph; R1: H; R2: H; R3: i一 P r ; R4 : H; R5: 3-MeO-Ph; n=2) Preparation of compound examples 1036 (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4: H; R 5: 3-MeO-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 95 (3H, d, J = 7. 0Hz) , 0. 9 9 (3H, d, J = 7. 0Hz) , 1. 92— 2. 05 ( 1 H, m) , 2. 95 一 3. 00 (lH, m) , 3. 40-3. 05 (2H, m) , 3. 79 (3H, s) , 5. 82 (1H, s) , 6. 82-6. 98 (4H, m) , 7. 29 (1 H, dd, J = 7. 4, 8. 3Hz) , 7. 43-7. 47 (2H, m) , 8. 12-8. 15 (1H, m) X HNMR (CDC 1 3) δ : 0. 95 (3H, d, J = 7. 0Hz), 0. 9 9 (3H, d, J = 7. 0Hz), 1. 92- 2. 05 (1 H , m), 2.95-3.00 (lH, m), 3.40-3.05 (2H, m), 3.79 (3H, s), 5.82 (1H, s), 6. 82-6. 98 (4H, m), 7.29 (1H, dd, J = 7.4, 8.3Hz), 7.43-7.47 (2H, m), 8.12-8. 15 (1H, m)
FAB-MS (m/e) : 346 [M + H] + 実施例 14 FAB-MS (m / e): 346 [M + H] + Example 14
化合物例 1037の製造 Preparation of Compound Example 1037
ジァステレオマー a (Ar : Ph; R1: H; R2: H; R3: i—P r ; R4: HJiasutereoma a (Ar: Ph; R 1 : H; R 2: H; R 3: i-P r; R 4: H
; R5: 2-MeO-Ph; n=2) R 5 : 2-MeO-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 96 (3Η, d, J = 6. 7Hz) , 0. 9 XHNMR (CDC 1 3) δ: 0. 96 (3Η, d, J = 6. 7Hz), 0. 9
6 (3H, d, J = 6. 7Hz) , 1. 76 - 1. 85 (lH, m) , 3. 06 (IH, d d, J =l 1. 5, 14. 3Hz) , 3. 45 (IH, d d, J = 26 (3H, d, J = 6.7Hz), 1.76-1.85 (lH, m), 3.06 (IH, dd, J = l 1.5, 14.3Hz), 3.45 ( IH, dd, J = 2
. 2, 14. 3Hz) , 3. 54 (IH, ddd, J = 2. 2, 4. 9, 11.2, 14.3 Hz), 3.54 (IH, ddd, J = 2.2, 4.9, 11.
5Hz) , 3. 70 (3H, s) , 6. 12 ( 1 H, s) , 6. 67— 6. 70 (1H, m) , 6. 92 (1H, d, J = 8. 2Hz) , 7. 06 (IH, t,5 Hz), 3.70 (3H, s), 6.12 (1H, s), 6.67—6.70 (1H, m), 6.92 (1H, d, J = 8.2Hz), 7.06 (IH, t,
J = 7. 5Hz) , 7. 30-7. 40 (3H, m) , 7. 50 (1H, dd, J = 1. 5, 7. 5Hz) , 8. 12-8. 16 (IH, m) J = 7.5Hz), 7.30-7.40 (3H, m), 7.50 (1H, dd, J = 1.5, 7.5 Hz), 8.12-8.16 (IH, m)
FAB-MS (m/e) : 346 [M + H] + FAB-MS (m / e): 346 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i - P r ; R4 : H ; R5: 2-MeO-Ph; n = 2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i - P r; R 4: H; R 5: 2-MeO-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 92 (3H, d, J = 6. 8 Hz) , 0. 9 4 (3H, d, J = 6. 8Hz) , 1. 90— 2. 01 (IH, m) , 2, 83 一 2. 93 (1H, m) , 3. 55 (2H, d, J = 6. 0Hz) , 3. 78 ( 3H, s) , 5. 92 (1H, s) , 6. 86-6. 90 (lH, m) , 6. 9 XHNMR (CDC 1 3) δ: 0. 92 (3H, d, J = 6. 8 Hz), 0. 9 4 (3H, d, J = 6. 8Hz), 1. 90- 2. 01 (IH, m), 2,83-1 2.93 (1H, m), 3.55 (2H, d, J = 6.0 Hz), 3.78 (3H, s), 5.92 (1H, s), 6 86-6. 90 (lH, m), 6.9
4 (IH, d, J = 8. 2Hz) , 6. 99 ( 1 H, t, J = 7. 5Hz) , 7 . 26-7. 45 ( H, m) , 8. 14-8. 18 ( 1 H, m) 4 (IH, d, J = 8.2Hz), 6.99 (1H, t, J = 7.5Hz), 7.26-7.45 (H, m), 8.14-8.18 ( 1 H, m)
FAB-MS (m/e) : 346 [M + H] + 実施例 15 FAB-MS (m / e): 346 [M + H] + Example 15
化合物例 1040の製造 Preparation of Compound Example 1040
ジァステレオマー a ( A r : P h; R 1: H; R 2: H; R 3: i一 P r ; R4: H ; R5 : 4-BnO-Ph ; n = 0) Jiasutereoma a (A r: P h; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 4-BnO-Ph; n = 0)
XHNMR (CDC 13) δ : 0. 94 (3Η, d, J = 6. 8Hz) , 0. 9 XHNMR (CDC 1 3) δ: 0. 94 (3Η, d, J = 6. 8Hz), 0. 9
5 (3H, d, J = 6. 8Hz) , 1. 65— 1. 74 (lH, m) , 2. 43 (IH, dd, J = 10. 8, 13. 9Hz) , 2. 89 (1H, d d, J = 2 . 2, 13. 9Hz) , 3. 04 (IH, ddd, J = 2. 2, 5. 2, 10.5 (3H, d, J = 6.8 Hz), 1.65—1.74 (lH, m), 2.43 (IH, dd, J = 10.8, 13.9 Hz), 2.89 (1H , Dd, J = 2.2, 13.9 Hz), 3.04 (IH, ddd, J = 2.2, 5.2, 10.
8Hz) , 5. 10 (2H, s) , 5. 59 ( 1 H, s) , 6. 60-6. 64 (1H, m) , 7. 01 (2H, d, J = 8. 8Hz) , 7. 02-7. 12 (8Hz), 5.10 (2H, s), 5.59 (1H, s), 6.60-6.64 (1H, m), 7.01 (2H, d, J = 8.8Hz), 7. 02-7. 12 (
2H, m) , 7. 35 (2H, d, J = 8. 8Hz) , 7. 33-7. 49 (52H, m), 7.35 (2H, d, J = 8.8 Hz), 7.33-7.49 (5
H, m) , 7. 60-7. 64 (IH, m) H, m), 7.60-7. 64 (IH, m)
FAB— MS (m/e) : 390 [M + H] + FAB—MS (m / e): 390 [M + H] +
ジァステレオマー b (Ar i Ph ! R^ H j R^ H j R3 : i一 P r ; R4 : HR stereo: b (Ar i Ph! R ^ H j R ^ H j R 3 : i-i Pr; R 4 : H
; R5 : 4— BnO— Ph ; n=0) R 5 : 4— BnO— Ph; n = 0)
iHNMR (CDC 13) δ : 0. 94 ( 3 H, d, J = 6. 8Hz) , 0. 9 iHNMR (CDC 1 3) δ: 0. 94 (3 H, d, J = 6. 8Hz), 0. 9
4 (3H, d, J = 6. 8Hz) , 1. 95— 2. 09 ( 1 H, m) , 2. 83 -2. 91 (3H, m) , 5. 07 (2H, s) , 5. 69 (IH, s) , 6. 79 - 6. 82 (1H, m) , 6. 97 (2H, d, J = 8. 8Hz) , 7. 0 9-7. 17 (2H, m) , 7. 26 (2H, d, J = 8. 8Hz) , 7. 30 -7. 49 (5H, m) , 7. 50 - 7. 52 (lH, m) 4 (3H, d, J = 6.8 Hz), 1.95— 2.09 (1H, m), 2.83 -2.91 (3H, m), 5.07 (2H, s), 5.69 (IH, s), 6.79-6.82 (1H, m), 6.97 (2H, d, J = 8.8 Hz), 7.0 9-7.17 (2H, m), 7.26 (2H, d, J = 8.8 Hz), 7.30-7.49 (5H, m), 7. 50-7.52 (lH, m)
FAB-MS (m/e) : 390 [M + H] + 実施例 16 FAB-MS (m / e): 390 [M + H] + Example 16
化合物例 1041の製造 (Ar : Ph; R1: H; R2: H; R3: i -P r ; R4 : H; R5: 4-BnO-Ph; n= 1) Preparation of compound examples 1041 (Ar: Ph; R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: 4-BnO-Ph; n = 1)
XHNMR (CDC 13) δ : 0. 90 (3Η, d, J = 6. 8Hz) , 0. 9 4 (3H, d, J = 6. 8Hz) , 1. 75 - 1. 90 (IH, m) , 2. 58 -2. 62 (IH, m) , 3. 04 ( 1 H, dd, J =4. 8, 13. OHz) , 3. 50 (IH, d d, 1 = 7. 4, 13. OHz) , 5. 09 (2H, s ) , 5. 28 (IH, s) , 6. 84 ( 1 H, d, J = 7. 9Hz) , 6. 99 ( 2H, dd, J = 2. 0, 6. 7Hz) , 7. 25 (2H, dd, J = 2. 0, 6. 7Hz) , 7. 30- 7. 54 (7H, m) , 7. 95 ( 1 H, d d, J = 1. 2, 7. 9Hz) XHNMR (CDC 1 3) δ: 0. 90 (3Η, d, J = 6. 8Hz), 0. 9 4 (3H, d, J = 6. 8Hz), 1. 75 - 1. 90 (IH, m ), 2.58 -2.62 (IH, m), 3.04 (1H, dd, J = 4.8, 13. OHz), 3.50 (IH, dd, 1 = 7.4, 13) OHz), 5.09 (2H, s), 5.28 (IH, s), 6.84 (1H, d, J = 7.9Hz), 6.99 (2H, dd, J = 2. 0, 6.7Hz), 7.25 (2H, dd, J = 2.0, 6.7Hz), 7.30-7.54 (7H, m), 7.95 (1H, dd, J = (1.2, 7.9 Hz)
FAB-MS (m/e) : 406 [M + H] + 実施例 17 FAB-MS (m / e): 406 [M + H] + Example 17
化合物例 1042の製造 (Ar : Ph; R1: H; R2: H; R3: i -P r ; R4 : OH ; R5: 4-BnO-P h ; n= 1) Preparation of compound examples 1042 (Ar: Ph; R 1 : H; R 2: H; R 3: i -P r; R 4: OH; R 5: 4-BnO-P h; n = 1)
XHNM (CDC 13) δ : 1. 03 (3H, d, J = 6. 8Hz) , 1. 1 4 (3H, d, J = 6. 8Hz) , 1. 82 - 1. 96 (1H, m) , 3. 08 一 3. 26 (2H, m) , 3. 44—3. 57 (IH, m) , 4. 21 (IH, s) , 5. 10 (2H, s) , 5. 19 ( 1 H, s) , 6. 84 ( 1 H, d, J =7. 7Hz) , 7. 00 (2H, d, J = 8. 9Hz) , 7. 31 (2H, d , J = 8. 9Hz) , 7. 26 - 7. 56 (7H, m) , 7. 84-7. 94 ( IH, m) FAB-MS (m/e) : 422 [M + H] 実施例 18 X HNM (CDC 1 3) δ : 1. 03 (3H, d, J = 6. 8Hz), 1. 1 4 (3H, d, J = 6. 8Hz), 1. 82 - 1. 96 (1H, m), 3.08-3.26 (2H, m), 3.44-3.57 (IH, m), 4.21 (IH, s), 5.10 (2H, s), 5.19 (1H, s), 6.84 (1H, d, J = 7.7Hz), 7.00 (2H, d, J = 8.9Hz), 7.31 (2H, d, J = 8. 9Hz), 7.26-7.56 (7H, m), 7.84-7.94 (IH, m) FAB-MS (m / e): 422 [M + H] Example 18
化合物例 1043の製造 (Al^ Ph R^ H R^ H R3 P r ; R : H ; R 5 : 4-BnO-Ph ; n=2) Preparation of Compound Example 1043 (Al ^ Ph R ^ HR ^ HR 3 Pr; R: H; R 5 : 4-BnO-Ph; n = 2)
XHNMR (CDC 13) 5 : 0. 95 (3H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 89— 2. 00 (lH, m) , 2. 94 —3. 01 (1H, m) , 3. 41 (1H, d d, J = 9. 1, 14. 3Hz) , 3. 49 (1H, d d, J = 3. 4, 14. 3Hz) , 5. 08 (2H, s) , 5. 79 (1H, s) , 6. 93— 7. 00 (1H, m) , 6. 99 (1H, d, J = 9. 0Hz) , 7. 25 (2H, d, J = 9. 0Hz) , 7. 32— 7 . 50 (7H, m) , 8. 13-8. 17 (lH, m) X HNMR (CDC 1 3) 5 : 0. 95 (3H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 89- 2. 00 (lH, m), 2.94 — 3.01 (1H, m), 3.41 (1H, dd, J = 9.1, 14.3 Hz), 3.49 (1H, dd, J = 3.4, 14 3 Hz), 5.08 (2H, s), 5.79 (1H, s), 6.93—7.00 (1H, m), 6.99 (1H, d, J = 9.0Hz), 7.25 (2H, d, J = 9.0Hz), 7.32—7.50 (7H, m), 8.13-8.17 (lH, m)
FAB-MS (m/e) : 422 [M + H] + 実施例 1 9 FAB-MS (m / e): 422 [M + H] + Example 19
化合物例 1044の製造 Preparation of Compound Example 1044
ジァステレオマ一 a (A r : P h R1 : H ; R2 : H ; R -P r ; R4 : H ; R5 : 3 - Bn - Ph ; n=2) Jiasutereoma one a (A r: P h R 1: H; R 2: H; R -P r; R 4: H; R 5: 3 - Bn - Ph; n = 2)
iHNMR (CDC 13) (5 : 0 . 97 (3H, d, J = 6. 9Hz) , 0. 9 7 (3H, d, J = 6. 9Hz) : , 1. 74- 1. 86 (1 H, m) , 3. 06 (1H, d d, J =l 1. 5, 14. 3Hz) , 3. 43 (1H, dd, J = 2 iHNMR (CDC 1 3) (5 :. 0 97 (3H, d, J = 6. 9Hz), 0. 9 7 (3H, d, J = 6. 9Hz):, 1. 74- 1. 86 (1 H, m), 3.06 (1H, dd, J = l 1.5, 14.3 Hz), 3.43 (1H, dd, J = 2
. 2, 14. 3Hz) , 3. 52 ( 1 H, ddd, 1 = 2. 2, 4. 8 2, 14.3Hz), 3.52 (1H, ddd, 1 = 2.2, 4.8
5Hz) , 5. 08 (2H, s) , 5. 86 ( 1 H, s) , 6. 95 - 7. 00 (2H, m) , 7. 05 (1H, d, J = 1. 7Hz) , 7. 26-7 46 ( 8H, m) , 8. 12-8. 16 (1H, m) 5Hz), 5.08 (2H, s), 5.86 (1H, s), 6.95-7.00 (2H, m), 7.05 (1H, d, J = 1.7Hz), 7.26-7 46 (8H, m), 8.12-8.16 (1H, m)
FAB-MS (m/e) : 422 [M + H] + FAB-MS (m / e): 422 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 -P r ; R4 : H ; R5 : 3-Bn-Ph ; n=2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3 -P r; R 4: H; R 5: 3-Bn-Ph; n = 2)
!HNMR (CDC 13) (5 : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) 1. 90-2. 04 (1 H, m) , 2. 96 (IH, ddd, J = 3. 8, 5 8, 8. 7Hz) , 3. 43 (IH, d d, J = 8. 7, 14. 2Hz) , 3 50 (1H, d d, J = 3. 8, 14. 2H z) , 5. 05 (2H, s) , 5 82 (1H, s) , 6. 89 - 6. 99 (4 H, m) , 7. 25 - 7. 50 (8H, m) , 8. 12-8. 16 ( 1 H, m) FAB-MS (mZe) : 422 [M + H] + 実施例 20 ! HNMR (CDC 1 3) ( 5: 0. 94 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6.8 Hz) 1.90-2.04 (1 H, m), 2.96 (IH, ddd, J = 3. 8, 5 8, 8.7 Hz), 3. 43 (IH, dd, J = 8.7, 14.2Hz), 350 (1H, dd, J = 3.8, 14.2Hz), 5.05 (2H, s), 582 (1H, s), 6.89-6.99 (4 H, m), 7.25-7.50 (8H, m), 8.12-8.16 (1 H, m) FAB-MS (mZe): 422 [M + H] + Example 20
化合物例 1045の製造 Preparation of Compound Example 1045
ジァステレオマ一 a (A r : P h; R1: H; R2: H; R3: i一 P r ; R4 : H ; R5: 2 -BnO-Ph; n=2) Jiasutereoma one a (A r: P h; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 2 -BnO-Ph; n = 2)
XHNMR (CDC ") 5 : 0. 95 (3H, d, J = 7. OHz) , 0. 9 5 (3H, d, J = 7. OHz) , 1. 75 - 1. 86 ( 1 H, m) , 3. 04 (1H, d d, J = 11. 5, 14. 3Hz) , 3. 45 (1H, d d, J = 2 . 2, 14. 3Hz) , 3. 55 ( 1 H, ddd, J = 2. 2, 4. 9, 11. 5Hz) , 4. 95 (1H, d, J = 12. 3Hz) , 5. 03 ( 1 H, d, J = 12. 3Hz) , 6. 25 ( 1 H, s) , 6. 72 - 6. 76 (IH, m) , 6. 96 (1H, dd, J = l. 0, 8. 2Hz) , 7. 05-7. 13 (3 H , m) , 7. 18-7. 29 (3H, m) , 7. 29 - 7. 39 (3H, m) , 7. 57 (1 H, dd, J = 1. 3, 7. 5Hz) , 8. 12— 8. 15 (1 H , m) X HNMR (CDC ") 5: 0.95 (3H, d, J = 7.OHz), 0.95 (3H, d, J = 7.OHz), 1.75-1.86 (1H, m), 3.04 (1H, dd, J = 11.5, 14.3Hz), 3.45 (1H, dd, J = 2.2, 14.3Hz), 3.55 (1H, ddd, J = 2.2, 4. 9, 11.5Hz), 4.95 (1H, d, J = 12.3Hz), 5.03 (1H, d, J = 12.3Hz), 6.25 ( 1H, s), 6.72-6.76 (IH, m), 6.96 (1H, dd, J = l. 0, 8.2Hz), 7.05-7.13 (3H, m ), 7.18-7.29 (3H, m), 7.29-7.39 (3H, m), 7.57 (1H, dd, J = 1.3, 7.5Hz), 8. 12—8.15 (1 H, m)
FAB-MS (mZe) : 422 [M + H] + FAB-MS (mZe): 422 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i— P r ; R4 : H ; R5 : 2-BnO-Ph ; n=2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i- P r; R 4: H; R 5: 2-BnO-Ph; n = 2)
'HNMR (CDC 13) δ : 0. 85 (3H, d, J = 6. 8Hz) , 0. 8 8 (3H, d, J = 6. 8Hz) , 1. 84- 2. 00 (lH, m) , 2. 85 (IH, d d, J = 6. 4, 12. 8Hz) , 3. 52 (2H, d, J = 6. 4 Hz) , 5. 01 (IH, d, J = 12. OHz) , 5. 06 (IH, d, J = 12. 0Hz) , 5. 98 (IH, s) , 6. 87-6. 91 ( 1 H, m) , 6 . 99 -7. 04 (2H, m) , 7. 13-7. 17 (2H, m) , 7. 23— 7. 32 (3H, m) , 7. 32 - 7. 37 (2H, m) , 7. 38— 7. 46 (2H, m) , 8. 13-8. 17 (IH, m) 'HNMR (CDC 1 3) δ : 0. 85 (3H, d, J = 6. 8Hz), 0. 8 8 (3H, d, J = 6. 8Hz), 1. 84- 2. 00 (lH, m), 2.85 (IH, dd, J = 6.4, 12.8 Hz), 3.52 (2H, d, J = 6.4 Hz), 5.01 (IH, d, J = 12. OHz), 5.06 (IH, d, J = 12.0 Hz), 5.98 (IH, s), 6.87-6.91 (1H, m), 6 99 -7. 04 (2H, m), 7.13-7. 17 (2H, m), 7.23—7.32 (3H, m), 7.32-7.37 (2H, m) , 7.38—7.46 (2H, m), 8.13-8.17 (IH, m)
FAB-MS im/e) : 422 [M + H] + 実施例 21 FAB-MS im / e): 422 [M + H] + Example 21
化合物例 1046の製造 (Ar Ph ; !^1 : !^ ; !^2 : !! ; !^3 : i -P r ; R4 Production of Compound Example 1046 (Ar Ph;! ^ 1 :! ^;! ^ 2 : !!;! ^ 3 : i -P r; R 4
^NMR (CDC 13) 6 : 0. 97 (3H, d, J = 6. 9Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 70— 1. 88 (IH, m) , 3. 06 (1H, d d, J = l l. 3, 14. 1Hz) , 3. 13 (2H, t, J = 7. 0Hz) , 3. 42 (1H, dd, J = 2. 2, 14. 1Hz) , 3. 50 (1 H, ddd, J = 2, 2, 4. 9, 11. 3Hz) , 4. 22 (2H, t, J = 7. 0Hz) , 5. 79 (1 H, s) , 6. 71-6. 75 (lH, m) , 6. 94 (1H, d, J = 8. 7Hz) , 7. 20-7. 40 (9H, m) , 8. 1 0-8. 15 (1H, m) ^ NMR (CDC 1 3) 6 : 0. 97 (3H, d, J = 6. 9Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 70- 1. 88 (IH, m), 3.06 (1H, dd, J = l l. 3, 14.1Hz), 3.13 (2H, t, J = 7.0Hz), 3.42 (1H, dd, J = 2. 2, 14.1 Hz), 3.50 (1 H, ddd, J = 2, 2, 4.9, 11.3 Hz), 4.22 (2H, t, J = 7.0 Hz), 5.79 ( 1H, s), 6.71-6.75 (lH, m), 6.94 (1H, d, J = 8.7Hz), 7.20-7.40 (9H, m), 8.1 0-8. 15 (1H, m)
FAB-MS (mZe) : 436 [M + H] + 実施例 22 FAB-MS (mZe): 436 [M + H] + Example 22
化合物例 1047の製造 (A r : P h; R 1 : H; R2: H; R3: i— P r ; R4 : H ; R5 : 3-OH-3-Me-Bu; n=0) Preparation of compound examples 1047 (A r: P h; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 3-OH-3-Me-Bu; n = 0)
iHNMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 8Hz) , 0. 9 iHNMR (CDC 1 3) δ: 0. 94 (3H, d, J = 6. 8Hz), 0. 9
5 (3H, d, J = 6. 8Hz) , 1. 33 (6H, s) , 1. 64-1. 75 (lH, m) , 2. 02 (1H, t, J = 6. 2Hz) , 2. 43 (1H, d d , J = 10. 7, 13. 9Hz) , 2. 89 ( 1 H, d d, J = 2. 2, 13. 9Hz) , 3. 04 (IH, ddd, J = 2. 2, 5. 2, 10. 7Hz) , 4 . 22 (1H, t, J = 6. 2Hz) , 5. 59 ( 1 H, s) , 6. 59-6.5 (3H, d, J = 6.8Hz), 1.33 (6H, s), 1.64-1.75 (lH, m), 2.02 (1H, t, J = 6.2Hz), 2.43 (1H, dd, J = 10.7, 13.9Hz), 2.89 (1H, dd, J = 2.2, 13.9Hz), 3.04 (IH, ddd, J = 2 .2, 5.2, 10.7Hz), 4.22 (1H, t, J = 6.2Hz), 5.59 (1H, s), 6.59-6.
63 (IH, m) , 6. 94 (2H, d, J = 8. 5Hz) , 7. 03-7. 1 2 (2H, m) , 7. 35 (2H, d, J = 8. 5Hz) , 7. 61-7. 64 (1H, m) 63 (IH, m), 6.94 (2H, d, J = 8.5Hz), 7.03-7.12 (2H, m), 7.35 (2H, d, J = 8.5Hz) , 7.61-7.64 (1H, m)
FAB-MS (m/e) : 386 [M + H] + 実施例 23 FAB-MS (m / e): 386 [M + H] + Example 23
化合物例 1050の製造 Preparation of Compound Example 1050
ジァステレオマー a (A r : Ph; R1: H; R2: H; 3: i一 P r ; R4 : HDiastereomer a (A r: Ph; R 1 : H; R 2 : H; 3 : i-Pr; R 4 : H
; R5: 4-nP rNHCOCH20-Ph; n=0) R 5 : 4-nP rNHCOCH20-Ph; n = 0)
^NMR (CDC 13) δ : 0. 91— 0. 97 (9H, m) , 1. 50— 1 . 65 (3H, m) , 2. 42 (1H, d d, J = 10. 8, 13. 9Hz) , 2. 89 (1H, d d, J = 2. 2, 13. 9Hz) , 3. 22 - 3. 36 (1 H, m) , 3. 28 - 3. 38 (2H, m) , 4. 52 (2H, s) , 5. 62 ^ NMR (CDC 1 3) δ :. 0. 91- 0. 97 (9H, m), 1. 50- 1 65 (3H, m), 2. 42 (1H, dd, J = 10. 8, 13 9Hz), 2.89 (1H, dd, J = 2.2, 13.9Hz), 3.22-3.36 (1H, m), 3.28-3.38 (2H, m), 4.52 (2H, s), 5.62
(1H, s) , 6. 52 - 6. 57 ( 1 H, m) , 6. 94 (2H, d, J = 8 . 8Hz) , 7. 00-7. 12 (2H, m) , 7. 38 (2H, d, J = 8. 8Hz) , 7. 62-7. 65 (1H, m) (1H, s), 6.52-6.57 (1H, m), 6.94 (2H, d, J = 8.8Hz), 7.00-7.12 (2H, m), 7. 38 (2H, d, J = 8.8Hz), 7.62-7.65 (1H, m)
FAB-MS (m e) : 399 [M + H] + FAB-MS (m e): 399 [M + H] +
ジァステレオマー b (Ar : Ph j R^ H .- R' r H j R3 : i -P r ; R4 : HDiastereomer b (Ar: Ph j R ^ H .- R 'r H j R 3 : i -P r; R 4 : H
; R5: 4-nP rNHCOCH20-Ph; n=0) R 5 : 4-nP rNHCOCH20-Ph; n = 0)
^NMR (CDC 13) δ : 0. 85— 1. 00 (9H, m) , 1. 50- 1 . 65 (2H, m) , 1. 90 - 2. 00 (lH, m) , 2. 73 -2. 89 ( 3H, m) , 4. 50 (2H, s) , 5. 67 ( 1 H, s) , 6. 72— 6. 7 3 (lH, m) , 6. 90 (2H, d, J = 8. 7Hz) , 7. 10— 7. 15 ^ NMR (CDC 1 3) δ :. 0. 85- 1. 00 (9H, m), 1. 50- 1 65 (2H, m), 1. 90 - 2. 00 (lH, m), 2. 73-2.89 (3H, m), 4.50 (2H, s), 5.67 (1H, s), 6.72—6.73 (lH, m), 6.90 (2H, m) d, J = 8.7 Hz), 7.10—7.15
(2H, m) , 7. 28 (2H, d, J = 8. 7Hz) , 7. 50 - 7. 53 ( 1H, m) (2H, m), 7.28 (2H, d, J = 8.7 Hz), 7.50-7.53 (1H, m)
FAB-MS (mZe) : 399 [M + H] + 実施例 24 FAB-MS (mZe): 399 [M + H] + Example 24
化合物例 1051の製造 Preparation of Compound Example 1051
ジァステレオマー a (Ar : Ph; R1: H; R2: H; R3 i -P r ; R4: H ; R5: 4-n-P rNHCOCH20-Ph ; n=2) iHNMR (CDC 13) δ : 0. 94 (3H, t, J = 7. 5Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 0. 98 (3H, d, J = 6. 7Hz) , 1 . 53- 1. 65 (2H, m) , 1. 74—1. 89 (IH, m) , 3. 07 ( 1H, dd, J = 1 1. 3, 14. lHz) , 3. 33 ( 1 H, t, J = 6. 8 Hz) , 3. 35 (1 H, t, J = 6. 8Hz) , 3. 44 (IH, dd, J =Jiasutereoma a (Ar: Ph; R 1 : H; R 2: H; R 3 i -P r; R 4: H; R 5: 4-nP rNHCOCH 2 0-Ph; n = 2) iHNMR (CDC 1 3) δ: 0. 94 (3H, t, J = 7. 5Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 0. 98 (3H, d, J = 6 .7-Hz), 1.53-1.65 (2H, m), 1.74-1.89 (IH, m), 3.07 (1H, dd, J = 1 1.3, 14. lHz), 3.33 (1 H, t, J = 6.8 Hz), 3.35 (1 H, t, J = 6.8 Hz), 3.44 (IH, dd, J =
2. 3, 14. 1Hz) , 3. 50 (IH, d d d, J一 2. 3, 5. 4, 1 1 . 3Hz) , 4. 53 (2H, s) , 5. 85 ( 1 H, s) , 6. 59 ( 1 H, b r s) , 6. 69 - 6. 72 (1H, m) , 6. 97 (1H, d, J = 8. 6 Hz) , 7. 35 (1 H, d, J = 8. 6Hz) , 7. 35-7. 41 (2H, m) , 8. 13-8. 16 ( 1 H, m) 2.3, 14.1 Hz), 3.50 (IH, ddd, J-2.3, 5.5.4, 11.3 Hz), 4.53 (2H, s), 5.85 (1H, s) ), 6.59 (1H, brs), 6.69-6.72 (1H, m), 6.97 (1H, d, J = 8.6 Hz), 7.35 (1H, d, J = 8.6Hz), 7.35-7.41 (2H, m), 8.13-8.16 (1H, m)
FAB-MS (m/e) : 431 [M + H] + FAB-MS (m / e): 431 [M + H] +
ジァステレオマー b (Ar : P h ; R1 : H ; R2 : H ; R3 : 一 P r ; R4 : H ; R5: 4-n-P rNH CO CH20-Ph ; n=2) Jiasutereoma b (Ar: P h; R 1: H; R 2: H; R 3: one P r; R 4: H; R 5: 4-nP rNH CO CH 2 0-Ph; n = 2)
^NMR (CDC 13) δ : 0. 94 (3H, t, J = 7 6Hz) , 0 9 5 (3H, d, J = 6. 7Hz) , 0. 99 (3H, d, J 6. 7Hz) , ^ NMR (CDC 1 3) δ : 0. 94 (3H, t, J = 7 6Hz), 0 9 5 (3H, d, J = 6. 7Hz), 0. 99 (3H, d, J 6. 7Hz ),
. 51- 1. 71 (2H, m) , 1. 89— 2. 00 ( 1 H, m) , 2. 97-51- 1.71 (2H, m), 1.89—2.00 (1H, m), 2.97-
3. 04 (IH, m) , 3. 31 ( 1 H, t, J = 6. 9Hz) , 3. 33 (1 H, t, J = 6. 9Hz) , 3. 40- 3. 50 (2H, m) , 4. 51 (2H , s) , 5. 81 (1H, s) , 6. 60 ( 1 H, b r s) , 6. 91-6. 9 6 (IH, m) , 6. 93 ( 1 H, d, J = 8. 7Hz) , 7. 27 ( 1 H, d , J = 8. 7Hz) , 7. 45 -7. 52 (2H, m) , 8. 14— 8. 19 ( IH, m) 3.04 (IH, m), 3.31 (1H, t, J = 6.9Hz), 3.33 (1H, t, J = 6.9Hz), 3.40-3.50 (2H , M), 4.51 (2H, s), 5.81 (1H, s), 6.60 (1H, brs), 6.91-6.96 (IH, m), 6.93 ( 1 H, d, J = 8.7 Hz), 7.27 (1 H, d, J = 8.7 Hz), 7.45 -7.52 (2H, m), 8.14—8.19 (IH , m)
FAB-MS (m/e) : 431 [M + H] + 実施例 25 FAB-MS (m / e): 431 [M + H] + Example 25
化合物例 1052の製造 Preparation of Compound Example 1052
ジァステレオマ一 a (Ar : Ph; 1: H; R2: H; R3: i一 P r ; R4 : H ; R5: 3 - n— P rNHCOCH2〇— Ph; n = 2) Jiasutereoma one a (Ar: Ph; 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 3 - n- P rNHCOCH 2 〇- Ph; n = 2)
^NMR (CDC 13) <5 : 0. 93 ( 3 H, t, J = 7. 4Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 0. 99 (3H, d, J = 6. 8Hz) , 1 . 52- 1. 65 (2H, m) , 1. 75 - 1. 89 (lH, m) , 3. 07 ( IH, d d, J = 1 1. 5, 14. 4Hz) , 3. 31 (IH, t, J = 6. 6 Hz) , 3. 33 (1H, t, J = 6. 6Hz) , 3. 46 ( 1 H, d d, J = 2. 2, 14. 4Hz) , 3. 50 (1H, ddd, J = 2. 2, 5. 0, 1 1 . 5Hz) , 4. 51 (3H, s) , 5. 91 ( 1 H, s) , 6. 61 (1H, b r s) , 6. 69 - 6. 73 (lH, m) , 6. 87 -6. 92 ( 1 H, m) , 7. 04 (1H, d, J = 7. 9Hz) , 7. 33 -7. 43 (3H, m) , 8. 13-8. 17 (IH, m) ^ NMR (CDC 1 3) < 5: 0. 93 (3 H, t, J = 7. 4Hz), 0. 9 9 (3H, d, J = 6.8 Hz), 0.99 (3H, d, J = 6.8 Hz), 1.52-1.65 (2H, m), 1.75-1.89 (lH , M), 3.07 (IH, dd, J = 11.5, 14.4Hz), 3.31 (IH, t, J = 6.6Hz), 3.33 (1H, t, J = 6.6Hz), 3.46 (1H, dd, J = 2.2, 14.4Hz), 3.50 (1H, ddd, J = 2.2, 5.0, 11.5Hz), 4 51 (3H, s), 5.91 (1H, s), 6.61 (1H, brs), 6.69-6.73 (lH, m), 6.87-6.92 (1H , m), 7.04 (1H, d, J = 7.9 Hz), 7.33 -7.43 (3H, m), 8.13-8.17 (IH, m)
FAB-MS (m/e) : 431 [M + H] + FAB-MS (m / e): 431 [M + H] +
ジァステレオマー b (Ar i Ph j R^ H i R^ H j R3 : i一 P r ; R4 : H ; R5: 3-n-P rNHCOCH20-Ph; n=2) Diastereomer b (Ar i Ph j R ^ H i R ^ H j R 3 : i- 1 Pr; R 4 : H; R 5 : 3-nP rNHCOCH 20 0-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 92 (3H, t, J = 7. 4Hz) , 0. 9 7 (3H, d, J = 6. 9Hz) , 1. 01 (3H, d, J = 6. 7Hz) , 1 . 50 - 1. 65 (2H, m) , 1. 91— 2. 03 (lH, m) , 3. 01 ( 1H, d d d, J = 3. 6, 5. 5, 8. 9Hz) , 3. 29 ( 1 H, t, J = 6. 4Hz) , 3. 32 (1H, t, J = 6. 4Hz) , 3. 42 ( 1 H, d d , J = 8. 8, 14. 3Hz) , 3. 46 ( 1 H, d d, J = 3. 6, 14. 3 Hz) , 4. 48 (3H, s) , 5. 84 ( 1 H, s) , 6. 59 (IH, b r s) , 6. 85 - 6. 89 (lH, m) , 6. 93 ( 1 H, s) , 6. 94 (1 H, d, J = 7. 8Hz) , 7. 00 ( 1 H, d, J = 7. 8Hz) , 7. 32 (1H, t, J = 7. 8Hz) , 7. 46- 7. 53 (2H, m) , 8. 15 - 8. 19 (IH, m) X HNMR (CDC 1 3) δ : 0. 92 (3H, t, J = 7. 4Hz), 0. 9 7 (3H, d, J = 6. 9Hz), 1. 01 (3H, d, J = 6.7 Hz), 1.50-1.65 (2H, m), 1.91-2.03 (lH, m), 3.01 (1H, ddd, J = 3.6, 5.5, 8) 9Hz), 3.29 (1H, t, J = 6.4Hz), 3.32 (1H, t, J = 6.4Hz), 3.42 (1H, dd, J = 8.8, 14.3 Hz), 3.46 (1H, dd, J = 3.6, 14.3 Hz), 4.48 (3H, s), 5.84 (1H, s), 6.59 (IH , brs), 6.85-6.89 (lH, m), 6.93 (1H, s), 6.94 (1H, d, J = 7.8Hz), 7.00 (1H, d, J = 7.8Hz), 7.32 (1H, t, J = 7.8Hz), 7.46-7.53 (2H, m), 8.15-8.19 (IH, m)
FAB-MS (m/e) : 431 [M + H] + 実施例 26 FAB-MS (m / e): 431 [M + H] + Example 26
化合物例 1053の製造 Preparation of Compound Example 1053
ジァステレオマー a (Ar : Ph; R1: H; R2: H; R3: i - P r ; R4 : H ; R5: 2_n— P rNHCOCH2〇— Ph; n= 2) XHNMR (CDC 13) (5 : 0. 75 (3H, t, J = 7. 4Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 00 (3H, d, J = 6. 7Hz) , 1 . 37- 1. 52 (2H, m) , 1. 77— 1. 85 (IH, m) , 3. 02— 3. 22 (2H, m) , 3. 49 (1H, d d, J = 2. 2, 7. 2Hz) , 3 . 56 (lH, m) , 4. 21 (1H, d, J = 14. 1Hz) , 4. 40 (1 H, d, J = 14. 1Hz) , 6. 38 ( 1 H, s) , 6. 65— 6. 69 (1 H, m) , 6. 69 (IH, b r s) , 6. 83 (1H, d, J = 7. 6Hz) , 7. 17 (IH, t, J = 7. 6Hz) , 7. 34—7. 44 (3H, m) , 7. 64 (1 H, d, J = 7. 6Hz) , 8. 10-8. 14 (lH, m) FAB-MS (m/e) : 431 [M + H] + Jiasutereoma a (Ar: Ph; R 1 : H; R 2: H; R 3: i - P r; R 4: H; R 5: 2_n- P rNHCOCH 2 〇- Ph; n = 2) X HNMR (CDC 1 3) ( 5: 0. 75 (3H, t, J = 7. 4Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 00 (3H, d, J = 6.7 Hz), 1.37-1.52 (2H, m), 1.77—1.85 (IH, m), 3.02—3.22 (2H, m), 3.49 (1H , Dd, J = 2.2, 7.2 Hz), 3.56 (lH, m), 4.21 (1H, d, J = 14.1 Hz), 4.40 (1H, d, J = 14) 1Hz), 6.38 (1 H, s), 6.65— 6.69 (1 H, m), 6.69 (IH, brs), 6.83 (1H, d, J = 7.6 Hz ), 7.17 (IH, t, J = 7.6 Hz), 7.34—7.44 (3H, m), 7.64 (1 H, d, J = 7.6 Hz), 8.10- 8.14 (lH, m) FAB-MS (m / e): 431 [M + H] +
ジァステレオマ一 b (Ar : Ph j R^ H j R' i H i R3 : i - P r ; R4 : H ; R5: 2-n-P rNHCOCH20-Ph; n=2) Jiasutereoma one b (Ar: Ph j R ^ H j R 'i H i R 3: i - P r; R 4: H; R 5: 2-nP rNHCOCH 2 0-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 77 (3H, t, J = 7. 4Hz) , 0. 9 7 (3H, d, J = 6. 9Hz) , 1. 00 (3H, d, J = 6. 8Hz) , 1 . 30— 1. 51 (2H, m) , 2. 03— 2. 13 (IH, m) , 2. 75- 2. 85 (lH, m) , 3. 08— 3. 23 (2H, m) , 3. 59-3. 62 (2H, m) , 4. 21 (1H, d, J = 14. 2Hz) , 4. 40 (1H, d , J = 14. 2Hz) , 6. 27 ( 1 H, s) , 6. 73— 6. 76 ( 1 H, m ) , 6. 88 (IH, d, J = 7. 8Hz) , 6. 90 ( 1 H, b r s) , 7. 17 (1H, t, J = 7. 8Hz) , 7. 40 ( 1 H, t, J = 7. 8Hz) , 7. 42- 7. 49 (2H, m) , 7. 67 (IH, d, J = 7. 8Hz) , 8 . 05 -8. 09 (IH, m) X HNMR (CDC 1 3) δ : 0. 77 (3H, t, J = 7. 4Hz), 0. 9 7 (3H, d, J = 6. 9Hz), 1. 00 (3H, d, J = 6.8 Hz), 1.30—1.51 (2H, m), 2.03—2.13 (IH, m), 2.75-2.85 (lH, m), 3.08—3. 23 (2H, m), 3.59-3.62 (2H, m), 4.21 (1H, d, J = 14.2Hz), 4.40 (1H, d, J = 14.2Hz), 6.27 (1H, s), 6.73—6.76 (1H, m), 6.88 (IH, d, J = 7.8Hz), 6.90 (1H, brs), 7 .17 (1H, t, J = 7.8Hz), 7.40 (1H, t, J = 7.8Hz), 7.42-7.49 (2H, m), 7.67 (IH, d , J = 7.8 Hz), 8.05 -8.09 (IH, m)
FAB-MS (m/e) : 431 [M + H] + 実施例 27 FAB-MS (m / e): 431 [M + H] + Example 27
化合物例 1091の製造 Preparation of Compound Example 1091
ジァステレオマー a (Ar r Ph j R^ H j R^ H j R3 -P r ; R4 : H ; R5: 3— C l, 4-Me-Ph; n = 2) Diastereomer a (Ar r Ph j R ^ H j R ^ H j R 3 -P r; R 4 : H; R 5 : 3—Cl, 4-Me-Ph; n = 2)
XHNMR (CDC 1 J (5 : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 72- 1. 88 (1H, m) , 2. 41 (3H, s) , 3. 04 (1H, d d, J = 1 1. 3, 14. 2Hz) , 3. 4 X HNMR (CDC 1 J (5: 0.98 (3H, d, J = 6.8 Hz)), 0.9 8 (3H, d, J = 6.8 Hz), 1.72-1.88 (1H, m), 2.41 (3H, s), 3.04 (1H, dd, J = 1 1.3, 14.2Hz), 3.4
4 (IH, dd, J = 2. 3, 14. 2Hz) , 3. 45— 3. 55 (1H, m ) , 5. 87 (1H, s) , 6. 7 1 -6. 74 ( 1 H, m) , 7. 1 5 (IH , dd, J = 1. 6, 7. 8Hz) , 7. 26 ( 1 H, d, J = 7. 8Hz) , 7. 35— 7. 45 (3H, m) , 8. 12-8. 15 (IH, m) 4 (IH, dd, J = 2.3, 14.2 Hz), 3.45—3.55 (1H, m), 5.87 (1H, s), 6.7 1 -6.74 (1H , m), 7.15 (IH, dd, J = 1.6, 7.8 Hz), 7.26 (1H, d, J = 7.8 Hz), 7.35—7.45 (3H, m), 8.12-8.15 (IH, m)
FAB-MS (m/e) : 364/366 [M + H] + FAB-MS (m / e): 364/366 [M + H] +
ジァステレオマ一 b (Ar i Ph j R^ H i R^ H j R3 : i— P r ; R4 : H ; R5: 3 - C l, 4-Me-Ph ; n=2) Diastereomer b (Ar i Ph j R ^ H i R ^ H j R 3 : i—P r; R 4 : H; R 5 : 3-Cl, 4-Me-Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 4Hz) , 0. 9 XHNMR (CDC 1 3) (5 : 0. 95 (3H, d, J = 6. 4Hz), 0. 9
9 (3H, d, J = 6. 8Hz) , 1. 90-2. 01 (IH, m) , 2. 38 (3H, s) , 2. 95— 3. 05 (IH, m) , 3. 40- 3. 50 (2H, m) , 5. 99 (1H, s) , 6. 94 ( 1 H, m) , 7. 03 (1H, dd, J = 1. 6, 7. 8Hz) , 7. 20 ( 1 H, d, J = 7. 8Hz) , 7. 36 (IH, d, J = 1. 6Hz) , 7. 47 - 7. 50 (2H, m) , 8. 13— 8. 16 (IH, m) 9 (3H, d, J = 6.8 Hz), 1.90-2.01 (IH, m), 2.38 (3H, s), 2.95—3.05 (IH, m), 3. 40- 3.50 (2H, m), 5.99 (1H, s), 6.94 (1H, m), 7.03 (1H, dd, J = 1.6, 7. 8Hz), 7 20 (1H, d, J = 7.8Hz), 7.36 (IH, d, J = 1.6Hz), 7.47-7.50 (2H, m), 8.13—8.16 (IH, m)
FAB-MS (m/e) : 364/366 [M + H] + 実施例 28 FAB-MS (m / e): 364/366 [M + H] + Example 28
化合物例 1097の製造 Preparation of Compound Example 1097
ジァステレオマ一 a (Ar : Ph ; R1: H; R2: H; R3: i - P r ; R4: HJiasutereoma one a (Ar: Ph; R 1 : H; R 2: H; R 3: i - P r; R 4: H
; R5 : 3— HO— 4一 Me— Ph ; n=2); R 5: 3- HO- 4 one Me- Ph; n = 2)
NMR (CDC 13) (3 : 0. 97 (3H, d, J = 6. 9Hz) , 0. 9 NMR (CDC 1 3) (3 : 0. 97 (3H, d, J = 6. 9Hz), 0. 9
7 (3H, d, J = 6. 9Hz) , 1. 70— 1. 85 (1H, m) , 2. 28 (3H, s) , 3. 07 ( 1 H, dd, J =l 1. 3, 14. 3Hz) , 3. 47 (3H, d, J = 6.9 Hz), 1.70—1.85 (1H, m), 2.28 (3H, s), 3.07 (1H, dd, J = l 1.3 , 14.3Hz), 3.4
2 (1 H, dd, J = 2. 2, 14. 3Hz) , 3. 46- 3. 52 ( 1 H, m2 (1 H, dd, J = 2.2, 14.3 Hz), 3.46-3.52 (1 H, m
) , 5. 77 (1H, s) , 6. 67 - 6. 87 (3H, m) , 7. 16 (1 H), 5.77 (1H, s), 6.67-6.87 (3H, m), 7.16 (1H, s)
, d, J = 7. 6Hz) , 7. 36 - 7. 39 (2H, m) , 8. 1 1—8. 1, D, J = 7.6 Hz), 7.36-7.39 (2H, m), 8.1 1—8.1
4 (IH, m) FAB-MS (m/e) : 346 [M + H] + 4 (IH, m) FAB-MS (m / e): 346 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i - P r ; R4 : H ; R5: 3-HO-4-Me-Ph ; n=2)Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i - P r; R 4: H; R 5: 3-HO-4-Me-Ph; n = 2)
NMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 92— 2. 03 ( 1 H, m) , 2. 25 (3H, s) , 2. 94- 3. 02 (1H, m) , 3. 48- 3. 52 (2H, m) , 5. 75 (1H, s) , 6. 74 ( 1 H, d, J = 1. 5Hz) , 6. 8 0 (1 H, dd, J = l. 5, 7. 6Hz) , 6. 95 - 6. 98 (lH, m) , 7. 12 (1H, d, J = 7. 6Hz) , 7. 45— 7. 48 (2H, m) , 8. 12-8. 1 5 (1H, m) NMR (CDC 1 3) δ: 0. 94 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 92- 2. 03 (1 H, m), 2.25 (3H, s), 2.94-3.02 (1H, m), 3.48-3.52 (2H, m), 5.75 (1H, s), 6.74 (1H, d, J = 1.5Hz), 6.80 (1H, dd, J = l, 5, 7.6Hz), 6.95-6.98 (lH, m), 7.12 (1H, d, J = 7.6Hz), 7.45—7.48 (2H, m), 8.12-8.15 (1H, m)
FAB-MS (m/e) : 346 [M + H] + 実施例 29 FAB-MS (m / e): 346 [M + H] + Example 29
化合物例 1098の製造 Preparation of Compound Example 1098
ジァステレオマー a (Ar i P j R^ H j R^ H j R3 : i—P r ; R4 : H ; R5 : 3-PhCH20-4-Me-Ph ; n=2) Diastereomer a (Ar i P j R ^ H j R ^ H j R 3 : i—P r; R 4 : H; R 5 : 3-PhCH20-4-Me-Ph; n = 2)
^NMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 7Hz) , 0. 9 ^ NMR (CDC 1 3) δ : 0. 96 (3H, d, J = 6. 7Hz), 0. 9
6 (3H, d, J = 6. 7Hz) , 1. 72— 1. 85 (lH, m) , 2. 32 (3H, s) , 3. 07 (1H, d d, J =l 1. 6, 14. 2Hz) , 3. 4 2 (1H, dd, J = 2. 2, 14. 2Hz) , 3. 47-3. 54 (1H, m ) , 5. 06 (1H. d, J = 2. 8Hz) , 5. 06 ( 1 H, d, J = 2. 8 Hz) , 5. 79 (1H, s) , 6. 74 ( 1 H, dd, J = l. 9, 6. 9H z) , 6. 91 -6. 94 (2H, m) , 7. 21 (1H, d, J = 7. 6Hz ) , 7. 30- 7. 43 (7H, m) , 8. 12— 8. 15 (1H, m) 6 (3H, d, J = 6.7 Hz), 1.72-1.85 (lH, m), 2.32 (3H, s), 3.07 (1H, dd, J = l 1.6, 14.2Hz), 3.4.2 (1H, dd, J = 2.2, 14.2Hz), 3.47-3.54 (1H, m), 5.06 (1H.d, J = 2. 8Hz), 5.06 (1H, d, J = 2.8Hz), 5.79 (1H, s), 6.74 (1H, dd, J = l. 9, 6.9Hz), 6.91 -6.94 (2H, m), 7.21 (1H, d, J = 7.6Hz), 7.30-7.43 (7H, m), 8.12—8.15 (1H , m)
FAB-MS (m/e) : 436 [M + H] + FAB-MS (m / e): 436 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i一 P r ; R4 ·· H ; R5 : 3-PhCH20-4-Me-Ph ; n=2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4 ·· H; R 5: 3-PhCH20-4-Me-Ph; n = 2)
XHNMR (CDC 13) d : 0. 93 ( 3 H, d, J = 6. 9Hz) , 0. 9 XHNMR (CDC 1 3) d: 0. 93 (3 H, d, J = 6. 9Hz), 0. 9
7 (3H, d, J = 6. 7Hz) , 1. 92-2. 01 (lH, m) , 2. 30 (3H, s) , 2. 92-2. 98 (lH, m) , 3. 39— 3. 53 (2H, m) , 5. 03 (2H, s) , 5. 78 (IH, s) , 6. 82 - 6. 90 (3 H, m) , 7. 16 (IH, d, J = 7. 6Hz) , 7. 29- 7. 49 (7H , m) , 8. 12-8. 15 (1H, m) 7 (3H, d, J = 6.7Hz), 1.92-2.01 (lH, m), 2.30 (3H, s), 2.92-2.98 (lH, m), 3.39—3.53 (2H, m), 5.03 (2H, s), 5.78 (IH, s), 6.82-6.90 (3 H, m), 7.16 (IH, d, J = 7.6 Hz), 7.29-7.49 (7H, m), 8.12-8.15 ( 1H, m)
FAB-MS (m/e) : 436 [M + H] + 実施例 30 FAB-MS (m / e): 436 [M + H] + Example 30
化合物例 1099の製造 (A r : Ph; R1: H; R2: H; R3: i— P r ; R4 : H; R5: 3 - MeO— 4一 Me— Ph; n = 2) Preparation of compound examples 1099 (A r: Ph; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 3 - MeO- 4 one Me- Ph; n = 2 )
^NMR (CDC 13) 6 : 0. 96 (3H, d, J = 6. 7Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 94-2. 07 (lH, m) , 2. 03 (3H, s) , 2. 95 - 3. 02 ( 1 H, m) , 3. 41— 3. 56 (2H, m) , 3. 77 (3H, s) , 5. 81 (IH, s) , 6. 79— 6. 82 (2 H, m) , 6. 92 - 6. 95 (IH, m) , 7. 13 (IH, d, J = 7. 3 Hz) , 7. 45- 7. 48 (2H, m) , 8. 13— 8. 16 ( 1 H, m) FAB-MS (m/e) : 360 [M + H] + 実施例 31 ^ NMR (CDC 1 3) 6 :. 0. 96 (3H, d, J = 6. 7Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 94-2 07 (lH, m), 2.03 (3H, s), 2.95-3.02 (1H, m), 3.41—3.56 (2H, m), 3.77 (3H, s), 5. 81 (IH, s), 6.79—6.82 (2H, m), 6.92-6.95 (IH, m), 7.13 (IH, d, J = 7.3 Hz), 7.45- 7.48 (2H, m), 8.13—8.16 (1H, m) FAB-MS (m / e): 360 [M + H] + Example 31
化合物例 1101の製造 (Ar Pt^ R H R^ H R3 : i - P r ; R4 : H; R5: 3— v i ny 1— 4 - Me— Ph; n=2) Preparation of Compound Example 1101 (Ar Pt ^ RHR ^ HR 3 : i-P r; R 4 : H; R 5 : 3-viny 1-4-Me-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 7Hz) , 0. 9 X HNMR (CDC 1 3) δ : 0. 95 (3H, d, J = 6. 7Hz), 0. 9
9 (3H, d, J = 6. 7Hz) , 1. 90— 2. 02 (IH, m) , 2. 36 (3H, s) , 2. 95 - 3. 05 (lH, m) , 3. 40- 3. 55 (2H, m) , 5. 28 (IH, d d, J = l. 3, 10. 9Hz) , 5. 59 (1H, d d. J = l. 3, 17. 4Hz) , 5. 81 (1H, s) , 6. 94 (1H, d d, J = 10. 9, 17. 4Hz) , 6. 90- 7. 00 (lH, m) , 7.9 (3H, d, J = 6.7 Hz), 1.90—2.02 (IH, m), 2.36 (3H, s), 2.95-3.05 (lH, m), 3. 40- 3.55 (2H, m), 5.28 (IH, dd, J = l. 3, 10.9Hz), 5.59 (1H, d d. J = l. 3, 17.4Hz), 5.81 (1H, s), 6.94 (1H, dd, J = 10.9, 17.4Hz), 6.90- 7.00 (lH, m), 7.
09 (1H, d d, J = l. 9, 7. 8Hz) , 7. 13 ( 1 H, d, J = 7.09 (1H, d d, J = l. 9, 7.8Hz), 7.13 (1H, d, J = 7.
8Hz) , 7. 45 - 7. 50 (2H, m) , 7. 65 - 7. 75 (lH, m)8Hz), 7.45-7.50 (2H, m), 7.65-7.75 (lH, m)
, 8. 14-8. 17 (IH, m) FAB-MS (m/e) : 356 [M + H] + 実施例 32 , 8. 14-8. 17 (IH, m) FAB-MS (m / e): 356 [M + H] + Example 32
化合物例 1 102の製造 (Ar Ph s R^ H R^ H R3 : i -P r ;_R^ : H; 5: 3- i P rO-4-Me-Ph; n = 2) Preparation of compound examples 1 102 (Ar Ph s R ^ HR ^ HR 3: i -P r; _R ^: H; 5: 3- i P rO-4-Me-Ph; n = 2)
iHNMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 29 (3H, d, J = 6. 0Hz) , 1 . 29 (3H, d, J = 6. 0Hz) , 1. 95— 2. 08 ( 1 H, m) , 2. 21 (3H, s) , 2. 93- 3. 02 (1H, m) , 3. 41-3. 54 (2 H, m) , 4. 40-4. 50 (lH, m) , 5. 75 (1H, s) , 6. 78 -6. 81 (2H, m) , 6. 92 - 6. 94 ( 1 H, m) , 7. 1 (1H, d, J = 7. 9Hz) , 7. 44-7. 47 (2H, m) , 8. 12-8. 15 (1H, m) iHNMR (CDC 1 3) δ: 0. 95 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 29 (3H, d, J = 6 0 Hz), 1.29 (3H, d, J = 6.0Hz), 1.95—2.08 (1H, m), 2.21 (3H, s), 2.93-3.02 ( 1H, m), 3.41-3.54 (2H, m), 4.40-4.50 (lH, m), 5.75 (1H, s), 6.78-6.81 (2H , M), 6.92-6.94 (1H, m), 7.1 (1H, d, J = 7.9Hz), 7.44-7.47 (2H, m), 8.12- 8.15 (1H, m)
FAB-MS (m/e) : 388 [M + H] + 実施例 33 FAB-MS (m / e): 388 [M + H] + Example 33
化合物例 1106の製造 Preparation of Compound Example 1106
ジァステレオマー a (Ar i Ph j R^ H j R^ H i R3 : i一 P r ; R4 : HDiastereomer a (Ar i Ph j R ^ H j R ^ H i R 3 : i- 1 Pr; R 4 : H
; R5: 3 - OH- 4 -Me O-P h ; n = 2) R 5 : 3-OH-4 -Me OP h; n = 2)
iHNMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 9Hz) , 0. 9 iHNMR (CDC 1 3) δ: 0. 97 (3H, d, J = 6. 9Hz), 0. 9
7 (3H, d, J = 6. 9Hz) , 1. 71- 1. 82 (lH, m) , 3. 06 (1H, dd, J = 11. 5, 14. 3Hz) , 3. 41 ( 1 H, d d, J = 27 (3H, d, J = 6.9 Hz), 1.71-1.82 (lH, m), 3.06 (1H, dd, J = 11.5, 14.3 Hz), 3.41 (1 H, dd, J = 2
. 2, 14. 3Hz) , 3. 50 ( 1 H, d d d, J = 2. 2, 4. 8, 11.2, 14.3Hz), 3.50 (1 H, d d d, J = 2. 2, 4. 8, 11.
5Hz) , 3. 93 (3H, s) , 5. 76 ( 1 H, s) , 6. 77 (1H, m ) , 6. 85— 6. 90 (2H, m) , 7. 01 ( 1 H, d, J = l. 9Hz)5Hz), 3.93 (3H, s), 5.76 (1H, s), 6.77 (1H, m), 6.85—6.90 (2H, m), 7.01 (1H , d, J = l. 9Hz)
, 7. 35 - 7. 38 (2H, m) , 8. 11— 8. 14 ( 1 H, m) , 7.35-7.38 (2H, m), 8.11—8.14 (1H, m)
FAB-MS (m/e) : 362 [M + H] + FAB-MS (m / e): 362 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i - P r ; R4: HJiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i - P r; R 4: H
; R5: 3-OH-4-MeO-P ; n=2) iHNMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 90-2. 04 (1H, m) , 2. 93 一 3. 01 (1H, m) , 3. 38 - 3. 52 (2H, m) , 3. 90 (3H, s) , 5. 74 (1H, s) , 6. 73 (1H, dd, J = 2. 6, 8. 2Hz ) , 6. 83 (IH, d, J = 8. 2Hz) , 6. 94-6. 99 (2H, m) , 7. 44- 7. 47 (2H, m) , 8. 11-8. 14 ( 1 H, m) R 5 : 3-OH-4-MeO-P; n = 2) iHNMR (CDC 1 3) (5 :. 0. 95 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 90-2 04 (1H, m), 2.93-3.01 (1H, m), 3.38-3.52 (2H, m), 3.90 (3H, s), 5.74 (1H, s), 6.73 (1H, dd, J = 2.6, 8.2Hz), 6.83 (IH, d, J = 8.2Hz), 6.94-6.99 (2H, m), 7.44- 7. 47 (2H, m), 8.11-8.14 (1H, m)
FAB-MS (m/e) : 362 [M + H] + 実施例 34 FAB-MS (m / e): 362 [M + H] + Example 34
化合物例 1107の製造 Preparation of Compound Example 1107
ジァステレオマ一 a (Ar : Ph; R1: H; R2: H; R3 -P r ; R4: HJiasutereoma one a (Ar: Ph; R 1 : H; R 2: H; R 3 -P r; R 4: H
; R5: 4-OH- 3 - I -Ph; n=0) R 5 : 4-OH-3-I-Ph; n = 0)
XHNMR (CDC 13) δ : 0. 93 (3Η, d, J = 6. 8Hz) , 0. 9 5 (3H, d, J = 6. 8Hz) , 1. 53— 1. 82 (IH, m) , 2. 40 X HNMR (CDC 1 3) δ : 0. 93 (3Η, d, J = 6. 8Hz), 0. 9 5 (3H, d, J = 6. 8Hz), 1. 53- 1. 82 (IH, m), 2.40
(1H, dd, J = 10. 8, 13. 9Hz) , 2. 89 ( 1 H, d d, J = 2 . 2, 13. 9Hz) , 3. 00 (IH, d d d, J = 2. 2, 5. 0, 10. 8Hz) , 5. 60 (1H, s) , 6. 58— 6. 65 (IH, m) , 6. 99(1H, dd, J = 10.8, 13.9Hz), 2.89 (1H, dd, J = 2.2, 13.9Hz), 3.00 (IH, ddd, J = 2.2, 5.0, 10.8 Hz), 5.60 (1H, s), 6.58—6.65 (IH, m), 6.99
(1 H, d, J = 8. 2Hz) , 7. 03-7. 16 (2H, m) , 7. 28 ( 1H, dd, J = l. 7, 8. 2Hz) , 7. 61-7. 67 (IH, m) , 7 . 77 (1H, d, J = 1. 7Hz) (1H, d, J = 8.2Hz), 7.03-7.16 (2H, m), 7.28 (1H, dd, J = l. 7, 8.2Hz), 7.61-7 . 67 (IH, m), 7.77 (1H, d, J = 1.7 Hz)
FAB-MS (m/e) : 426 [M + H] + FAB-MS (m / e): 426 [M + H] +
ジァステレオマー b (Ar i P j R^ H j R^ H i R3 : i - P r ; 4 : HDiastereomer b (Ar i P j R ^ H j R ^ H i R 3 : i-P r; 4 : H
; R5: 4-OH-3- I -Ph ; n=0) R 5 : 4-OH-3- I -Ph; n = 0)
^NMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 5 (3H, d, J = 6. 7Hz) , 1. 93— 2. 04 ( 1 H, m) , 2. 76 — 2. 88 (3H, m) , 5. 65 ( 1 H, s) , 6. 76— 6. 80 ( 1 H, m) , 6. 94 (1H, d, J = 8. 4Hz) , 7. 12— 7. 20 (2H, m ) , 7. 17 (IH, d d, J = 2. 0, 8. 4Hz) , 7. 50— 7. 54 ( IH, m) , 7. 68 ( 1 H, d, J = 2. 0Hz) FAB-MS (m/e) : 426 [M + H] + 実施例 35 ^ NMR (CDC 1 3) δ : 0. 94 (3H, d, J = 6. 7Hz), 0. 9 5 (3H, d, J = 6. 7Hz), 1. 93- 2. 04 (1 H , m), 2.76 — 2.88 (3H, m), 5.65 (1H, s), 6.76 — 6.80 (1H, m), 6.94 (1H, d, J = 8.4Hz), 7.12-7.20 (2H, m), 7.17 (IH, dd, J = 2.0, 8.4Hz), 7.50-7.54 (IH, m) , 7.68 (1 H, d, J = 2.0 Hz) FAB-MS (m / e): 426 [M + H] + Example 35
化合物例 1 108の製造 (A r : Ph ; R1: H; R2: H; R3: i— P r ; R4 : H; R5 : 3- 1 -4- OH-P ; n=2)Preparation of compound examples 1 108 (A r: Ph; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 3- 1 -4- OH-P; n = 2)
NMR (CDC 13) δ : 0. 97 (3Η, d, J = 6. 8Hz) , 1. 0 1 (3H, d, J = 6. 8Hz) , 1. 88— 2. 00 (1H, m) , 2. 92 一 3. 04 (IH, m) , 3. 36 - 3. 50 (2H, m) , 5. 79 (IH, s) , 6. 90 -7. 00 (lH, m) , 6. 95 (IH, d, J = 8. 3Hz ) , 7. 13 (IH, dd, J = l. 8, 8. 3Hz) , 7. 45-7. 52 ( 2H, m) , 7. 67 (IH, d, J = l. 8Hz) , 8. 12-8. 18 (1 H, m) NMR (CDC 1 3) δ: 0. 97 (3Η, d, J = 6. 8Hz), 1. 0 1 (3H, d, J = 6. 8Hz), 1. 88- 2. 00 (1H, m ), 2.92-3.04 (IH, m), 3.36-3.50 (2H, m), 5.79 (IH, s), 6.90-7.00 (lH, m), 6.95 (IH, d, J = 8.3 Hz), 7.13 (IH, dd, J = l, 8, 8.3 Hz), 7.45-7.52 (2H, m), 7.67 (IH, d, J = l. 8Hz), 8.12-8.18 (1 H, m)
FAB-MS (m/e) : 458 [M + H] + 実施例 36 FAB-MS (m / e): 458 [M + H] + Example 36
化合物例 1 109の製造 (Ar : P h ; R 1: H; R 2: H; R 3: i一 P r ; R4 : H; R 5: 5- 1 -2 -OH-Ph; n=2) Preparation of compound examples 1 109 (Ar: P h; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 5- 1 -2 -OH-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 99 (3H, d, J =6. 9Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 82- 1. 94 (IH, m) , 3. 1 3 (IH, dd, J =l 1. 6, 14. 8Hz) , 3. 46 ( 1 H, d d, J = 2 . 0, 14. 8Hz) , 3. 47 - 3. 58 (IH, m) , 6. 08 (1H, s ) , 6. 71 (IH, d, J = 8. 6Hz) , 7. 07— 7. 1 3 (2H, m) , 7. 48 - 7. 59 (3H, m) , 8. 16-8. 20 ( 1 H, m) X HNMR (CDC 1 3) δ : 0. 99 (. 3H, d, J = 6 9Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 82- 1. 94 (IH, m), 3.13 (IH, dd, J = 1.6, 14.8 Hz), 3.46 (1H, dd, J = 2.0, 14.8 Hz), 3.47-3. 58 (IH, m), 6.08 (1H, s), 6.71 (IH, d, J = 8.6 Hz), 7.07—7.13 (2H, m), 7.48-7 . 59 (3H, m), 8.16-8.20 (1 H, m)
FAB-MS (m/e) : 457 [M + H] + 実施例 37 FAB-MS (m / e): 457 [M + H] + Example 37
化合物例 1 1 10の製造 (Ar : Ph ; R1: H; R2: H; R3: i— P r : R4 : H; R5 : 2- 1 -5- OH-Ph ; n=2) Preparation of compound examples 1 1 10 (Ar: Ph; R 1: H; R 2: H; R 3: i- P r: R 4: H; R 5: 2- 1 -5- OH-Ph; n = 2)
XHNMR (CDC ) δ : 0. 95 ( 3 H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 9Hz) , 1. 90— 2. 00 (IH, m) , 2. 95 —3. 04 (1H, m) , 3. 35- 3. 50 (2H, m) , 4. 75 (1H, b s) , 5. 77 (IH, s) , 6. 60 - 6. 65 (IH, m) , 6. 85— 7. 00 (2H, m) , 7. 32 (IH, d, J = 8. 6Hz) , 7. 45-7 . 52 (lH, m) , 7. 81 ( 1 H, d, J = 8. 6Hz) , 8. 13— 8. 16 (1H, m) XHNMR (CDC) δ: 0.95 (3H, d, J = 6.8 Hz), 0.9 9 (3H, d, J = 6.9 Hz), 1.90—2.00 (IH, m), 2.95—3.04 (1H, m), 3.35-3.50 (2H, m ), 4.75 (1H, bs), 5.77 (IH, s), 6.60-6.65 (IH, m), 6.85—7.00 (2H, m), 7.32 ( IH, d, J = 8.6 Hz), 7.45-7.52 (lH, m), 7.81 (1H, d, J = 8.6 Hz), 8.13—8.16 (1H, m)
FAB-MS (mZe) : 458 [M + H] + 実施例 38 FAB-MS (mZe): 458 [M + H] + Example 38
化合物例 1 1 12の製造 Preparation of Compound Example 1 1 12
ジァステレオマ一 a (Ar : Ph ; R1: H; R2: H; R3: i一 P r ; R4 : H ; R5: 3— B r— 4一 OMe - Ph ; n = 2) Jiasutereoma one a (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4: H; R 5: 3- B r- 4 one OMe - Ph; n = 2)
^NMR (CDC 13) <5 : 0. 98 (3H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 75 - 1. 85 (IH, m) , 3. 05 (IH, d d, J =l 1. 3, 14. 1Hz) , 3. 44 (1H, d d, 1= 2 . 3, 14. 1Hz) , 3. 46-3. 53 ( 1 H, m) , 3. 94 (3H, s ) , 5. 85 (IH, s) , 6. 72 -6. 76 (lH, m) , 6. 96 (1 H , d, J = 8. 5Hz) , 7. 23 ( 1 H, dd, J = 2. 1, 8. 5Hz) , 7. 38- 7. 42 (2H, m) , 7. 45 (1H, d, J = 2. 1Hz) , 8 . 12-8. 16 (IH, m) ^ NMR (CDC 1 3) < 5: 0. 98 (3H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 75 - 1. 85 (IH , m), 3.05 (IH, dd, J = l 1.3, 14.1 Hz), 3.44 (1H, dd, 1 = 2.3, 14.1 Hz), 3.46-3.53 (1H, m), 3.94 (3H, s), 5.85 (IH, s), 6.72-6.76 (lH, m), 6.96 (1H, d, J = 8 .5Hz), 7.23 (1H, dd, J = 2.1, 8.5Hz), 7.38-7.42 (2H, m), 7.45 (1H, d, J = 2.1Hz) ), 8.12-8.16 (IH, m)
FAB-MS (m/e) : 380, 382 [M + H] + FAB-MS (m / e): 380, 382 [M + H] +
ジァステレオマ一 b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i -P r ; R4 : H ; R 5 : 3 - C 1 , 4-MeO-Ph ; n=2) Jiasutereoma one b (Ar: Ph; R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: 3 - C 1, 4-MeO-Ph; n = 2)
XHNMR (CDC 13) d : 0. 96 ( 3 H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 90-2. 10 ( 1 H, m) , 3. 00 XHNMR (CDC 1 3) d: . 0. 96 (3 H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 90-2 10 (1 H , m), 3.00
(IH, ddd, J = 3. 4, 5 6, 9.' 0Hz) , 3. 41 (IH, dd, J = 9. 0, 14. 3Hz) , 3 47 (1H, d d, J - 3. 4, 14. 3H z) , 3. 91 (3H, s) , 5 79 (1H, s) , 6. 90 (IH, d, J(IH, ddd, J = 3.4, 56, 9. '0 Hz), 3.41 (IH, dd, J = 9.0, 14.3 Hz), 347 (1H, dd, J-3. 4, 14.3Hz), 3.91 (3H, s), 579 (1H, s), 6.90 (IH, d, J
=8. 7Hz) , 6. 93 - 6. 97 (IH, m) , 7. 08 (1H, d d, J = 1. 6, 8. 7Hz) , 7. 43 ( 1 H, d, J = l. 6Hz) , 7. 48- 7. 51 (2H, m) , 8. 14— 8. 18 (IH, m) = 8.7 Hz), 6.93-6.97 (IH, m), 7.08 (1H, dd, J = 1. 6, 8.7 Hz), 7.43 (1H, d, J = l. 6Hz), 7.48-7.51 (2H, m), 8.14—8.18 (IH, m )
FAB-MS (mZe) : 380/382 [M + H] + 実施例 39 FAB-MS (mZe): 380/382 [M + H] + Example 39
化合物例 1113の製造 Preparation of Compound Example 1113
ジァステレオマ一 a (Ar : Ph ; Rx : H ; R2 : H ; R3 : : P r ; R4 : H ; R5: 3-B r-4-OMe-Ph ; n = 2) Jiasutereoma one a (Ar: Ph; R x : H; R 2: H; R 3:: P r; R 4: H; R 5: 3-B r-4-OMe-Ph; n = 2)
iHNMR (CDC 13) δ : 0. 98 (3Η, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 73— 1. 88 (IH, m) , 3. 05 (IH, d d, J =l 1. 4, 14. 3Hz) , 3. 44 (1H, dd, J = 2 . 3, 14. 3Hz) , 3. 49 ( 1 H, ddd, J = 2. 3, 4. 9, 11. 4Hz) , 3. 94 (3H, s) , 5. 85 (1H, s) , 6. 73-6. 77 (IH, m) , 6. 94 (IH, d, J = 8. 5Hz) , 7. 28 ( 1 H, dd , J = 2. 1, 8. 5Hz) , 7. 38— 7. 42 (2H, m) , 7. 63 (1 H, d, J = 2. 1Hz) , 8. 12-8. 16 (IH, m) iHNMR (CDC 1 3) δ: 0. 98 (3Η, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 73- 1. 88 (IH, m ), 3.05 (IH, dd, J = l 1.4, 14.3Hz), 3.44 (1H, dd, J = 2.3, 14.3Hz), 3.49 (1H, ddd, J = 2.3, 4.9, 11.4 Hz), 3.94 (3H, s), 5.85 (1H, s), 6.73-6.77 (IH, m), 6.94 ( IH, d, J = 8.5 Hz), 7.28 (1H, dd, J = 2.1, 8.5 Hz), 7.38—7.42 (2H, m), 7.63 (1H , D, J = 2.1 Hz), 8.12-8.16 (IH, m)
FAB-MS (m/e) : 424/426 [M + H] + FAB-MS (m / e): 424/426 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i -P r ; R4 : H ; R5 : 3— B r, 4-MeO-Ph ; n=2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: 3- B r, 4-MeO-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 89— 2. 01 (lH, m) , 3. 00 (1H, ddd, J = 3. 5, 5. 7, 9. 0Hz) , 3. 40 (IH, dd, J = 9. 0, 14. 3Hz) , 3. 47 ( 1 H, dd, 1 = 3. 5, 14. 3H z) , 3. 91 (3H, s) , 5. 79 ( 1 H, s) , 6. 87 ( 1 H, d, J =8. 6Hz) , 6. 94- 6. 98 (IH, m) , 7. 12 (1H, d d, J =2. 0, 8. 6Hz) , 7. 46- 7. 54 (2H, m) , 7. 62 (IH, d, J = 2. 0Hz) , 8. 14-8. 18 (IH, m) XHNMR (CDC 1 3) δ: 0. 96 (3H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 89- 2. 01 (lH, m ), 3.00 (1H, ddd, J = 3.5, 5. 7, 9.0 Hz), 3.40 (IH, dd, J = 9.0, 14.3Hz), 3.47 (1H , dd, 1 = 3.5, 14.3Hz), 3.91 (3H, s), 5.79 (1H, s), 6.87 (1H, d, J = 8.6Hz), 6.94- 6.98 (IH, m), 7.12 (1H, dd, J = 2.0, 8.6 Hz), 7.46-7.54 (2H, m), 7.62 (IH , d, J = 2.0 Hz), 8.14-8.18 (IH, m)
FAB-MS (m/e) : 424/426 [M + H] + 実施例 40 FAB-MS (m / e): 424/426 [M + H] + Example 40
化合物例 1 1 4の製造 Preparation of Compound Example 1 1 4
ジァステレオマー a (Ar Ph ! R^ H j R^ H i R3 i -P r : R4 : H ; R5 : 4-OMe-3- I -Ph; n=0) Jiasutereoma a (Ar Ph R ^ H j R ^ H i R 3 i -P r: R 4:! H; R 5: 4-OMe-3- I -Ph; n = 0)
XHNMR (CDC 13) δ : 0. 94 (3Η, d, J = 6. 8Hz) , 0. 9 5 (3H, d, J = 6. 8Hz) , 1. 65— 1. 78 (1H, m) , 2. 40 (1H, dd, J = 10. 8, 13. 9Hz) , 2. 89 (1H, d d, J = 2 . 2, 13. 9Hz) , 3. 01 (1H, ddd, J = 2. 2, 5. 3 , 10. 8Hz) , 3. 91 (3H, s) , 5. 60 (1H, s) , 6. 59 - 6. 63 (1H, m) , 6. 84 (1H, d, J = 8. 5Hz) , 7. 06— 7. 14 ( 2H, m) , 7. 36 (1H, dd, J = 2. 0, 8. 5Hz) , 7. 62-7 . 66 (lH, m) , 7. 87 (1H, d, J = 2. 0Hz) XHNMR (CDC 1 3) δ: 0. 94 (3Η, d, J = 6. 8Hz), 0. 9 5 (3H, d, J = 6. 8Hz), 1. 65- 1. 78 (1H, m ), 2.40 (1H, dd, J = 10.8, 13.9Hz), 2.89 (1H, dd, J = 2.2, 13.9Hz), 3.01 (1H, ddd, J = 2.2, 5.3, 10.8 Hz), 3.91 (3H, s), 5.60 (1H, s), 6.59-6.63 (1H, m), 6.84 (1H, d, J = 8.5 Hz), 7.06-7.14 (2H, m), 7.36 (1H, dd, J = 2.0, 8.5 Hz), 7.62-7.66 (lH , M), 7.87 (1H, d, J = 2.0Hz)
FAB-MS (m/e) : 440 [M + H] + FAB-MS (m / e): 440 [M + H] +
ジァステレオマ一 b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i P r : R4 : H ; R5 : 4-OMe-3- I -Ph; n=0) Jiasutereoma one b (Ar: Ph; R 1 : H; R 2: H; R 3: i P r: R 4: H; R 5: 4-OMe-3- I -Ph; n = 0)
^NMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 5 (3H, d, J = 6. 7Hz) , 1. 92— 2. 04 (1H, m) , 2. 75 —2. 84 (lH, m) , 2. 85— 2. 92 (2H, m) , 3. 89 (3H, s) , 5. 65 (1H, s) , 6. 77 - 6. 80 (lH, m) , 6. 79 (1 H, d, J = 8. 5Hz) , 7. 10-7. 18 (2H, m) , 7. 22 (1 H , dd, J = 2. 3, 8. 5Hz) , 7. 50— 7. 54 (1H, m) , 7. 8 2 (1H, d, J = 2. 3Hz) ^ NMR (CDC 1 3) δ : 0. 94 (3H, d, J = 6. 7Hz), 0. 9 5 (3H, d, J = 6. 7Hz), 1. 92- 2. 04 (1H, m), 2.75 — 2.84 (lH, m), 2.85—2.92 (2H, m), 3.89 (3H, s), 5.65 (1H, s), 6.77 -6.80 (lH, m), 6.79 (1 H, d, J = 8.5 Hz), 7.10-7.18 (2H, m), 7.22 (1 H, dd, J = 2. 3, 8.5 Hz), 7.50—7.54 (1H, m), 7.82 (1H, d, J = 2.3 Hz)
FAB-MS (m/e) : 440 [M + H] + 実施例 41 FAB-MS (m / e): 440 [M + H] + Example 41
化合物例 1116の製造 Preparation of Compound Example 1116
ジァステレオマー a (Ar i P i R^ H j R^ H i R3 一 P r ; R4 : H ; R5 : 4-OMe - 3 - I -P h; n = 2) Diastereomer a (Ar i P i R ^ H j R ^ H i R 3 -1 Pr; R 4 : H; R 5 : 4-OMe-3-I-Ph; n = 2)
iHNMR (CDC 13) 6 : 0. 98 (3H, d, J = 6. 6Hz) , 0. 9 8 (3H, d, J = 6. 6Hz) , 1. 65— 1. 87 (lH, m) , 3. 05 (IH, d d, J = 11. , 14. 5Hz) , 3. 44 ( 1 H, d d, J = 2 . 3, 14. 5Hz) , 3. 44- 3. 52 (IH, m) , 3. 92 ( 3 H, s ) , 5. 83 (IH, s) , 6. 73- 6. 77 (lH, m) , 6. 86 (1 H , d, J = 8. 5Hz) , 7. 32 (1H, dd, 1 = 2. 0, 8. 5Hz) , 7. 38- 7. 42 (2H, m) , 7. 84 ( 1 H, d, J = 2. OHz) , 8 . 12-8. 16 (IH, m) iHNMR (CDC 1 3) 6: 0. 98 (3H, d, J = 6. 6Hz), 0. 9 8 (3H, d, J = 6.6Hz), 1.65-1.87 (lH, m), 3.05 (IH, dd, J = 11., 14.5Hz), 3.44 (1H , dd, J = 2.3, 14.5 Hz), 3.44-3.52 (IH, m), 3.92 (3H, s), 5.83 (IH, s), 6.73- 6.77 (lH, m), 6.86 (1H, d, J = 8.5Hz), 7.32 (1H, dd, 1 = 2.0, 8.5Hz), 7.38- 7. 42 (2H, m), 7.84 (1H, d, J = 2. OHz), 8.12-8.16 (IH, m)
FAB-MS (m/e) : 472 [M + H] + FAB-MS (m / e): 472 [M + H] +
ジァステレオマー b (Ar i Ph j R^ H j R' i H i R3 : i - P r ; R4 : H ; R5 : 3— I, 4-MeO-Ph ; n = 2) Diastereomer b (Ar i Ph j R ^ H j R ′ i H i R 3 : i-P r; R 4 : H; R 5 : 3—I, 4-MeO-Ph; n = 2)
^NMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 88 - 2. 00 (lH, m) , 2. 99 (IH, ddd, J = 3. 4, 5. 7, 9. 1Hz) , 3. 40 (1H, dd, J = 9. 1, 14. 4Hz) , 3. 47 ( 1 H, dd, J = 3. 4, 14. 4H z) , 3. 89 (IH, s) , 5. 77 ( 1 H, s) , 6. 79 ( 1 H, d, J =8. 5Hz) , 6. 94-6. 97 ( 1 H, m) , 7. 14 (IH, d d, J =2. 1, 8. 5Hz) , 7. 48 - 7. 52 (2H, m) , 7. 85 ( 1 H, d, J = 2. 1Hz) , 8. 14-8. 18 ( 1 H, m) ^ NMR (CDC 1 3) δ : 0. 96 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 88 - 2. 00 (lH, m), 2.99 (IH, ddd, J = 3.4, 5. 7, 9.1 Hz), 3.40 (1H, dd, J = 9.1, 14.4 Hz), 3.47 (1 H, dd, J = 3.4, 14.4Hz), 3.89 (IH, s), 5.77 (1H, s), 6.79 (1H, d, J = 8.5Hz) , 6.94-6.97 (1H, m), 7.14 (IH, dd, J = 2.1, 8.5Hz), 7.48-7.52 (2H, m), 7.85 (1 H, d, J = 2.1 Hz), 8.14-8.18 (1 H, m)
FAB-MS (m/e) : 472 [M + H] + 実施例 42 FAB-MS (m / e): 472 [M + H] + Example 42
化合物例 1117の製造 Preparation of Compound Example 1117
ジァステレオマー a (Ar r P j R' i H j R^ H j 3 P r ; R4 : H ; R5 : 2— I, 5-MeO-Ph ; n = 2) Jiasutereoma a (Ar r P j R ' i H j R ^ H j 3 P r; R 4: H; R 5: 2- I, 5-MeO-Ph; n = 2)
^NMR (CDC 13) (5 : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 72 - 1. 85 (IH, m) , 3. 06 (IH, d d, J = 11. 4, 14. 4Hz) , 3. 46 ( 1 H, dd, J = 1 . 7, 14. 4Hz) , 3. 48- 3. 54 (lH, m) , 3. 86 (3H, s ) , 5. 88 (IH, s) , 6. 73 - 6. 76 ( 1 H, m) , 6. 82 (IH , dd, J = 1. 7, 7. 9Hz) , 6. 87 ( 1 H, d, J = 1. 7Hz) , 7. 37- 7. 42 (2H, m) , 7. 8 1 (IH, d, 1=7. 9Hz) , 8 . 1 3-8. 17 (IH, m) ^ NMR (CDC 1 3) ( 5: 0. 98 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 72 - 1. 85 (IH , m), 3.06 (IH, dd, J = 11.4, 14.4Hz), 3.46 (1H, dd, J = 1.7, 14.4Hz), 3.48-3.54 (lH, m), 3.86 (3H, s), 5.88 (IH, s), 6.73-6.76 (1H, m), 6.82 (IH, m) , Dd, J = 1.7, 7.9 Hz), 6.87 (1H, d, J = 1.7Hz), 7.37- 7.42 (2H, m), 7.81 (IH, d, 1 = 7.9Hz), 8.1 3-8.17 (IH, m)
FAB-MS (m/e) : 472 [M + H] + FAB-MS (m / e): 472 [M + H] +
ジァステレオマー b (A r : Ph ; R1 : H ; 2 : H ; R3 : i -P r ; R4 : H ; R5 : 2 - 1 , 5-MeO-P ; n = 2) Jiasutereoma b (A r: Ph; R 1: H; 2: H; R 3: i -P r; R 4: H; R 5: 2 - 1, 5-MeO-P; n = 2)
XHNMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 9Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 90— 2. 05 ( 1 H, m) , 2. 95 —3. 02 (IH, m) , 3. 38-3. 51 (2H, m) , 3. 83 (3H, s) , 5. 84 (1H, s) , 6. 69 ( 1 H, dd, J = l. 9, 8. 2Hz ) , 6. 84 (IH, d, J = 1. 9Hz) , 6. 91— 6. 95 (1H, m) , 7. 46 -7. 53 (2H, m) , 7. 75 ( 1 H, d, J = 8. 2Hz) , 8. 14-8. 18 (IH, m) X HNMR (CDC 1 3) δ : 0. 96 (3H, d, J = 6. 9Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 90- 2. 05 (1 H , m), 2.95 — 3.02 (IH, m), 3.38-3.51 (2H, m), 3.83 (3H, s), 5.84 (1H, s), 6. 69 (1H, dd, J = l. 9, 8.2Hz), 6.84 (IH, d, J = 1.9Hz), 6.91—6.95 (1H, m), 7.46- 7.53 (2H, m), 7.75 (1H, d, J = 8.2Hz), 8.14-8.18 (IH, m)
FAB-MS (m/e) : 472 [M + H] + 実施例 43 FAB-MS (m / e): 472 [M + H] + Example 43
化合物例 1 1 18の製造 Preparation of Compound Example 1 1 18
ジァステレオマー a ( A r : P h; R 1: H; R 2 H; R3: i -P r ; R HJiasutereoma a (A r: P h; R 1: H; R 2 H; R 3: i -P r; RH
; R5: 5- 1 -2 -MeO-Ph; n=2) R 5 : 5-1-2-MeO-Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 97 (3H, d, J = 6. 7Hz) , 0. 9 7 (3H, d, J = 6. 7Hz) , 1. 75 - 1. 89 ( 1 H, m) , 3. 02 X HNMR (CDC 1 3) ( 5: 0. 97 (3H, d, J = 6. 7Hz), 0. 9 7 (3H, d, J = 6. 7Hz), 1. 75 - 1. 89 (1 H, m), 3.02
(1H, dd, J = 1 1. 4, 14. 3Hz) , 3. 46 (IH, d d, J = 2 . 3, 14. 3Hz) , 3. 48- 3. 54 ( 1 H, m) , 3. 65 (3H, s ) , 6. 08 (IH, s) , 6. 65-6. 69 (IH, m) , 6. 67 (1 H , d, J = 8. 6Hz) , 7. 33— 7. 42 (2H, m) , 7. 63 (1 H, dd, J = 2. 3, 8. 6Hz) , 7. 82 (IH, d, J = 2. 3Hz) , 8 . 1 1 -8. 16 (IH, m) (1H, dd, J = 1.1.4, 14.3Hz), 3.46 (IH, dd, J = 2.3, 14.3Hz), 3.48-3.54 (1H, m), 3.65 (3H, s), 6.08 (IH, s), 6.65-6.69 (IH, m), 6.67 (1H, d, J = 8.6 Hz), 7.33 — 7.42 (2H, m), 7.63 (1 H, dd, J = 2.3, 8.6 Hz), 7.82 (IH, d, J = 2.3 Hz), 8.1 1- 8.16 (IH, m)
FAB-MS (m/e) : 472 [M + H] + FAB-MS (m / e): 472 [M + H] +
ジァステレオマー b (Ar ^ j R^ H .- R^ H j R3 : i -P r ; R4: H ; R5: 5- 1 -2 -MeO-Ph; n=2) Diastereomer b (Ar ^ j R ^ H .- R ^ H j R 3 : i -P r; R 4 : H R 5 : 5-1-2-MeO-Ph; n = 2)
iHNMR (CDC 13) δ : 0. 93 (3H, d, J = 6. 8Hz) , 0. 9 6 (3H, d, J = 6. 8Hz) , 1. 88— 2. 02 (IH, m) , 2. 80 -2. 90 (1H, m) , 3. 52 (2H, d, J = 6. 4Hz) , 3. 72 ( 3H, s) , 5. 92 (1H, s) , 6. 70 ( 1 H, d, J = 8. 5Hz) , 6. 79-6. 84 (IH, m) , 7. 39— 7. 50 (2H, m) , 7. 56 -7. 66 (2H, m) , 8. 12-8. 16 (lH, m) iHNMR (CDC 1 3) δ: 0. 93 (3H, d, J = 6. 8Hz), 0. 9 6 (3H, d, J = 6. 8Hz), 1. 88- 2. 02 (IH, m ), 2.80-2.90 (1H, m), 3.52 (2H, d, J = 6.4Hz), 3.72 (3H, s), 5.92 (1H, s), 6. 70 (1H, d, J = 8.5Hz), 6.79-6.84 (IH, m), 7.39—7.50 (2H, m), 7.56-7.66 (2H, m), 8. 12-8. 16 (lH, m)
FAB-MS (mZe) : 471 [M + H] + 実施例 44 FAB-MS (mZe): 471 [M + H] + Example 44
化合物例 1 1 19の製造 (Ai^ Pl^ R^ H R2 : !! ; !^3 : i - P r ; R4 : H; 5 : 3- I -4-E t O-Ph ; n=2) Preparation of compound examples 1 1 19 (Ai ^ Pl ^ R ^ HR 2:! !!; ^ 3: i - P r; R 4: H; 5: 3- I -4-E t O-Ph; n = 2)
^NMR (CDC 13) d : 0. 96 (3H, d, J = 6. 5Hz) , 1. 0 2 (3H, d, J = 6. 5Hz) , 1. 48 (3H, t, J = 6. 9Hz) , 1 . 90-2. 05 (lH, m) , 2. 92— 3. 08 ( 1 H, m) , 3. 35— 3. 50 (2H, m) , 4. 09 (2H, q, J = 6. 9Hz) , 5. 79 (1 H, s) , 6. 76 (IH, d, J = 8. 1Hz) , 6. 94— 6. 97 (IH , m) , 7. 13 (1H, d d, J =4. 9, 8. lHz) , 7. 45-7. 5 5 (2H, m) , 7. 84 (IH, d, J =4. 9Hz) , 8. 13— 8. 1 8 ( 1 H, m) ^ NMR (CDC 1 3) d : 0. 96 (3H, d, J = 6. 5Hz), 1. 0 2 (3H, d, J = 6. 5Hz), 1. 48 (3H, t, J = 6.9Hz), 1.90-2.05 (lH, m), 2.92—3.08 (1H, m), 3.35—3.50 (2H, m), 4.09 (2H , q, J = 6.9 Hz), 5.79 (1 H, s), 6.76 (IH, d, J = 8.1 Hz), 6.94—6.97 (IH, m), 7. 13 (1H, dd, J = 4.9, 8. lHz), 7.45-7.5.5 (2H, m), 7.84 (IH, d, J = 4.9Hz), 8.13 8. 1 8 (1 H, m)
FAB-MS (m/e) : 486 [M + H] + 実施例 45 FAB-MS (m / e): 486 [M + H] + Example 45
化合物例 1 120の製造 ( A r : P h; R 1: H; R 2 H: R i -P r : R : H; R5 : 3- I -4- i P rO-P ; n= 2) Preparation of compound examples 1 120 (A r: P h ; R 1: H; R 2 H: R i -P r: R: H; R 5: 3- I -4- i P rO-P; n = 2 )
XHNMR (CDC 13) δ : 0. 96 (3H, d, J = 7. 0Hz) , 1. 0 0 (3H, d, J = 6. 5Hz) , 1. 40 (3H, d, J = 5. 8Hz) , 1 . 40 (3H, d, J = 5. 8Hz) , 1. 85 -2. 00 ( 1 H, m) , 2. 90 - 3. 05 (IH, m) , 3. 35-3. 51 (2H, m) , 4. 50-4 . 62 (lH, m) , 5. 76 (1H, s) , 6. 78 ( 1 H, d, J = 7. 9 Hz) , 6. 93 -6. 96 (IH, m) , 7. 11 (1H, dd, J = 1. 9 , 7. 9Hz) , 7. 45 - 7. 52 (2H, m) , 7. 84 (IH, d, J = 1. 9Hz) , 8. 13-8. 16 (IH, m) X HNMR (CDC 1 3) δ : 0. 96 (3H, d, J = 7. 0Hz), 1. 0 0 (3H, d, J = 6. 5Hz), 1. 40 (3H, d, J = (5.8 Hz), 1.40 (3H, d, J = 5.8 Hz), 1.85 -2.00 (1 H, m), 2.90-3.05 (IH, m), 3.35 -3.51 (2H, m), 4.50-4 62 (lH, m), 5.76 (1H, s), 6.78 (1H, d, J = 7.9 Hz), 6.93 -6. 96 (IH, m), 7.11 (1H, dd, J = 1.9, 7.9Hz), 7.45-7.52 (2H, m), 7.84 (IH, d, J = 1.9Hz), 8.13-8. 16 (IH, m)
FAB-MS (m/e) : 500 [M + H] + 実施例 46 FAB-MS (m / e): 500 [M + H] + Example 46
化合物例 1121の製造 (Ar : Ph; R1: H; R2: H; R3: i— P r ; R4 : H; : 3- I -4-nP r O-Ph; n= 2) Preparation of compound examples 1121 (Ar: Ph; R 1 : H; R 2: H; R 3: i- P r; R 4: H;: 3- I -4-nP r O-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 6Hz) , 1. 10 (3H, t, J = 7. 4Hz) , 1 . 80-2. 00 (3H, m) , 2. 95 - 3. 04 (IH, m) , 3. 35- 3. 50 (2H, m) , 3. 98 (2H, t, J = 6. 3Hz) , 5. 76 (1 H, s) , 6. 75 (1H, d, J = 8. 5Hz) , 6. 93- 6. 96 (IH , m) , 7. 11 (IH, dd, J = 2. 0, 8. 5Hz) , 7. 47-7. 5 0 (2H, m) , 7. 83 (1 H, d, J = 2. 2Hz) , 8. 13-8. 16 (IH, m) XHNMR (CDC 1 3) δ: 0. 95 (3H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 6Hz), 1. 10 (3H, t, J = 7 4 Hz), 1.80-2.00 (3H, m), 2.95-3.04 (IH, m), 3.35-3.50 (2H, m), 3.98 (2H, t , J = 6.3Hz), 5.76 (1H, s), 6.75 (1H, d, J = 8.5Hz), 6.93-6.96 (IH, m), 7.11 ( IH, dd, J = 2.0, 8.5Hz), 7.47-7.50 (2H, m), 7.83 (1H, d, J = 2.2Hz), 8.13-8 . 16 (IH, m)
FAB-MS (m/e) : 500 [M + H] + 実施例 47 FAB-MS (m / e): 500 [M + H] + Example 47
化合物例 1122の製造 (Ai^ Ph ; !^1 : !! ; !^2 : !^ ; !^3 : i -P r ; R4 : H; R5: 3— I— 4— nBuO— Ph ; n = 2) Preparation of Compound Example 1122 (Ai ^ Ph;! ^ 1 : !!;! ^ 2 :! ^;! ^ 3 : i -P r; R 4 : H; R 5 : 3— I— 4— nBuO— Ph ; n = 2)
XHNMR (CDC 13) δ : 0. 95- 1. 02 (9H, m) , 1. 50-1 . 70 (2H, m) , 1. 90 - 2. 00 (IH, m) , 2. 95— 3. 05 ( IH, m) , 3. 40— 3. 50 (2H, m) , 4. 02 (2H, t, J = 6. 3Hz) , 5. 78 (1H, s) , 6. 73 ( 1 H, d, J = 8. 4Hz) , 6 . 92-6. 99 (IH, m) , 7. 12 ( 1 H, dd, J = 2. 0, 8. 4H z) , 7. 45-7. 52 (2H, m) , 7. 83 (1H, d, J = 2. OHz ) , 8. 14-8. 17 (IH, m) FAB-MS (m/e) : 514 [M + H] + 実施例 48 X HNMR (CDC 1 3) δ :. 0. 95- 1. 02 (9H, m), 1. 50-1 70 (2H, m), 1. 90 - 2. 00 (IH, m), 2. 95—3.05 (IH, m), 3.40—3.50 (2H, m), 4.02 (2H, t, J = 6.3Hz), 5.78 (1H, s), 6. 73 (1H, d, J = 8.4Hz), 6.92-6.99 (IH, m), 7.12 (1H, dd, J = 2.0, 8.4Hz), 7. 45-7. 52 (2H, m), 7.83 (1H, d, J = 2. OHz), 8.14-8.17 (IH, m) FAB-MS (m / e): 514 [M + H] + Example 48
化合物例 1 1 23の製造 (Ar z Pi^ R H R H R3 : i— P r ; R4 : H; R5 : 3- 1 -4- (2 -Me) P rQ-Ph ; n=2) Preparation of Compound Example 1 123 (Ar z Pi ^ RHRHR 3 : i—P r; R 4 : H; R 5 : 3-1-4- (2-Me) PrQ-Ph; n = 2)
XHNMR (CDC 13) d : 0. 95— 1. 02 (9H, m) , 1. 30— 1 . 36 (3H, m) , 1. 75— 1. 85 (2H, m) , 1. 90 - 2. 05 ( lH, m) , 2. 95— 3. 05 (IH, m) , 3. 38— 3. 50 (2H, m ) , 4. 30-4. 40 (IH, m) , 5. 78 (1H, s) , 6. 75 (1 H , d, J = 8. 3Hz) , 6. 97-6. 99 (IH, m) , 7. 12 (IH, d d, J = 2. 0, 8. 3Hz) , 7. 48-7. 53 (2H, m) , 7. 8 3 (1H, d, J = 2. 0Hz) , 8. 14-8. 17 ( 1 H, m) X HNMR (CDC 1 3) d :. 0. 95- 1. 02 (9H, m), 1. 30- 1 36 (3H, m), 1. 75- 1. 85 (2H, m), 1. 90-2.05 (lH, m), 2.95-3.05 (IH, m), 3.38-3.50 (2H, m), 4.30-4.40 (IH, m), 5.78 (1H, s), 6.75 (1H, d, J = 8.3 Hz), 6.97-6.99 (IH, m), 7.12 (IH, dd, J = 2. 0, 8.3Hz), 7.48-7.53 (2H, m), 7.83 (1H, d, J = 2.0Hz), 8.14-8.17 (1H, m)
FAB-MS (m/e) : 514 [M + H] + 実施例 49 FAB-MS (m / e): 514 [M + H] + Example 49
化合物例 1 1 24の製造 (Ar : Ph; R1: H; R2: H; R3: i一 P r ; R4 : H; R5: 3 - 1 , 4-PhCH¾Q-Ph ; n = 2) Preparation of compound examples 1 1 24 (Ar: Ph; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 3 - 1, 4-PhCH ¾ Q-Ph; n = 2)
^NMR (CDC 13) δ : 0. 96 (3Η, d, J = 6. 9Hz) , 1. 0 0 (3H, d, J = 6. 7Hz) , 1. 86-2. 00 ( 1 H, m) , 2. 9 9 (IH, ddd, J = 3. 4, 5. 7, 9. 4Hz) , 3. 39 ( 1 H, dd, J = 9. 4, 14. 3Hz) , 3. 47 ( 1 H, d d, J = 3. 4, 14. 3H z) , 5. 16 (2H, s) , 5. 77 ( 1 H, s) , 6. 82 (1H, d, J =8. 7Hz) , 6. 93 -6. 97 (lH, m) , 7. 12 ( 1 H, d d, J =2. 0, 8. 7Hz) , 7. 30- 7. 44 (3H, m) , 7. 46-7. 5 2 (4H, m) , 7. 87 (1H, d, J = 2. 0Hz) , 8. 13-8. 1 7 (IH, m) ^ NMR (CDC 1 3) δ :. 0. 96 (3Η, d, J = 6. 9Hz), 1. 0 0 (3H, d, J = 6. 7Hz), 1. 86-2 00 (1 H , m), 2.99 (IH, ddd, J = 3.4, 5.7, 9.4 Hz), 3.39 (1 H, dd, J = 9.4, 14.3 Hz), 3. 47 (1H, dd, J = 3.4, 14.3Hz), 5.16 (2H, s), 5.77 (1H, s), 6.82 (1H, d, J = 8. 7Hz), 6.93 -6.97 (lH, m), 7.12 (1H, dd, J = 2.0, 8.7Hz), 7.30-7.44 (3H, m), 7 46-7. 5 2 (4H, m), 7.87 (1H, d, J = 2.0Hz), 8.13-8. 17 (IH, m)
FAB-MS (m/e) : 548 [M + H] + 実施例 50 化合物例 1 125の製造 (Ar : Ph; R1: H; R2: H; R3: i -P r ; R4 : H; R 5: 3 - B r , 4-P CH^CH^O-Ph ; n = 2) FAB-MS (m / e): 548 [M + H] + Example 50 Preparation of compound examples 1 125 (Ar: Ph; R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: 3 - B r, 4-P CH ^ CH ^ O -Ph; n = 2)
XHNMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 8Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 72 - 1. 87 ( 1 H, m) , 3. 04 (1H, d d, J = 1 1. 4, 14. 5Hz) , 3. 41 (2H, t, J = 6. 8Hz) , 3. 43 (1 H, d d, J = 2. 3, 14. 5Hz) , 3. 48 (1 H, d d d, J = 2, 3, 5. 0, 1 1. 4Hz) , 4. 25 (2H, t, J = 6. 8Hz) , 5. 83 (1H, s) , 6. 70 -6. 74 (lH, m) , 6. 88 (1H, d, J = 8. 5Hz) , 7. 20— 7. 43 (8H, m) , 7. 6 1 (IH, d, J = 2. 1Hz) , 8. 11-8. 1 5 ( 1 H, m) X HNMR (CDC 1 3) δ : 0. 97 (3H, d, J = 6. 8Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 72 - 1. 87 (1 H , m), 3.04 (1H, dd, J = 11.4, 14.5Hz), 3.41 (2H, t, J = 6.8Hz), 3.43 (1H, dd, J = 2.3, 14.5 Hz), 3.48 (1 H, ddd, J = 2, 3, 5.0, 11.4 Hz), 4.25 (2H, t, J = 6.8 Hz), 5 83 (1H, s), 6.70 -6.74 (lH, m), 6.88 (1H, d, J = 8.5 Hz), 7.20—7.43 (8H, m), 7 . 6 1 (IH, d, J = 2.1 Hz), 8.11-8. 1 5 (1 H, m)
FAB-MS (mZe) : 514, 516 [M + H] + 実施例 51 FAB-MS (mZe): 514, 516 [M + H] + Example 51
化合物例 1126の製造 (Ai^ Ph s R H R^H R3 : i—P r ; R4 : H; R5: 3-C 1 , 4-P CH^CH^O-Ph; n= 2)Preparation of Compound Example 1126 (Ai ^ Phs RHR ^ HR 3 : i-P r; R 4 : H; R 5 : 3-C 1, 4-P CH ^ CH ^ O-Ph; n = 2)
NMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 8Hz) , 0. 9 NMR (CDC 1 3) δ: 0. 97 (3H, d, J = 6. 8Hz), 0. 9
7 (3H, d, J = 6. 8Hz) , 1. 72- 1. 84 (lH, m) , 3. 04 (1H, dd, J = 1 1. 4, 14. lHz) , 3. 18 (2H, t, J = 6.7 (3H, d, J = 6.8 Hz), 1.72-1.84 (lH, m), 3.04 (1H, dd, J = 1 1.4, 14. lHz), 3.18 ( 2H, t, J = 6.
9Hz) , 3. 43 (1 H, d d, J = 2. 3, 14. 1Hz) , 3. 48 (1 H, ddd, J = 2, 3, 5. 0, 1 1. 4Hz) , 4. 26 (2H, t, J =9 Hz), 3.43 (1 H, dd, J = 2.3, 14.1 Hz), 3.48 (1 H, ddd, J = 2, 3, 5.0, 11.4 Hz), 4. 26 (2H, t, J =
6. 9Hz) , 5. 83 (1H, s) , 6. 70— 6. 74 (lH, m) , 6.6.9 Hz), 5.83 (1H, s), 6.70—6.74 (lH, m), 6.
92 (1H, d, J = 8. 5Hz) , 7. 17 ( 1 H, dd, J = 2. 1, 8.92 (1H, d, J = 8.5Hz), 7.17 (1H, dd, J = 2.1, 8.
5Hz) , 7. 23-7. 43 (7H, m) , 7. 44 ( 1 H, d, J = 2. 15Hz), 7.23-7.43 (7H, m), 7.44 (1H, d, J = 2.1
Hz) , 8. 12-8. 1 5 ( 1 H, m) Hz), 8.12-8. 1 5 (1 H, m)
FAB-MS im/e) : 470/472 [M + H] + 実施例 52 FAB-MS im / e): 470/472 [M + H] + Example 52
化合物例 1127の製造 (Ai^ Ph R^H R H R3 : i -P r ; R4 : H; R5: 4一 OMe— 3— Ph— Ph; n= 2) iHNMR (CDC 13) d : 0 96 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 9Hz) , 1. 90 - 2. 03 (IH, m) , 3. 02 (IH, d d d, J = 3. 3, 5. 7, 9. 2Hz) , 3. 44 (1 H, dd, J = 9. 2, 14. 3Hz) , 3. 49 (1 H, dd, J = 3. 3, 14. 3H zz)) ,, 33.. 8833 ((33HH,, ss)) ,, 55.. 84 ( 1 H, s) , 7. 22 - 7. 26 (1 H, m) , 7. 28-7. 34 (2H, m) , 7. 36- 7. 42 (2H, m) , 7. 46 - 7. 54 (4H, m) , 8. 14— 8. 18 (IH, m) Preparation of Compound Example 1127 (Ai ^ Ph R ^ HRHR 3 : i -P r; R 4 : H; R 5 : 4-OMe— 3— Ph— Ph; n = 2) iHNMR (CDC 1 3) d: 0 96 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 9Hz), 1. 90 - 2. 03 (IH, m) , 3.02 (IH, ddd, J = 3.3, 5.7.2.2 Hz), 3.44 (1 H, dd, J = 9.2, 14.3 Hz), 3.49 (1 H , dd, J = 3.3, 14.3Hzz)), 33 .. 8833 ((33HH ,, ss)), 55..84 (1H, s), 7.22-7.26 ( 1H, m), 7.28-7.34 (2H, m), 7.36-7.42 (2H, m), 7.46-7.54 (4H, m), 8.14—8 . 18 (IH, m)
FAB-MS (m/e) : 422 [M + H] + 実施例 53 FAB-MS (m / e): 422 [M + H] + Example 53
化合物例 1 128の製造 (Ar : Ph; R1: H; R2: H; R3: i— P r ; R4 H; R5: 4-OMe - 3 (2—— py r i dy l) —Ph ; n=2) Preparation of compound examples 1 128 (Ar: Ph; R 1: H; R 2: H; R 3: i- P r; R 4 H; R 5: 4-OMe - 3 (2-- py ri dy l) --Ph; n = 2)
^NMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 9Hz) , 0. 9 7 (3H, d, J = 7. 5Hz) , 1. 90-2. 03 (1 H, m) , 2. 99 (1H, d d d, J = 3. 6, 5. 6, 8. 7Hz) , 3. 45 ( 1 H, dd, J = 8. 7, 14. 2Hz) , 3. 50 (1H, d d, J = 3. 6, 14. 2H z) , 3. 88 (3H, s) , 5. 87 (1H, s) , 6. 99- 7. 04 (1 H, m) , 7. 01 (IH, d, J =8. 5Hz) , 7. 18-7. 23 (1 H ^ NMR (CDC 1 3) δ :. 0. 94 (3H, d, J = 6. 9Hz), 0. 9 7 (3H, d, J = 7. 5Hz), 1. 90-2 03 (1 H , m), 2.99 (1H, ddd, J = 3.6, 5.6, 8.7Hz), 3.45 (1H, dd, J = 8.7, 14.2Hz), 3.50 (1H, dd, J = 3.6, 14.2Hz), 3.88 (3H, s), 5.87 (1H, s), 6.99-7.04 (1H, m), 7 01 (IH, d, J = 8.5 Hz), 7.18-7.23 (1 H
, m) , 7. 24- 7. 29 (IH, m) , 7. 44- 7. 49 (2H, m) , 7. 67 -7. 74 (1H, m) , 7. 81 (IH, d, J =1. 8Hz) , 7 . 83 (IH, d d, J = l. 8, 8. 5Hz) , 8. 12— 8. 16 (IH, m) , 8. 66-8. 69 (IH, m) , m), 7.24-7.29 (IH, m), 7.44-7.49 (2H, m), 7.67 -7. 74 (1H, m), 7.81 (IH, d , J = 1.8 Hz), 7.83 (IH, dd, J = l, 8, 8.5 Hz), 8.12-8.16 (IH, m), 8.66-8.69 (IH, m)
FAB-MS (m/e) : 423 [M + H] + 実施例 54 FAB-MS (m / e): 423 [M + H] + Example 54
化合物例 1129の製造 (Ar r Ph R^ H R H R i— P r ; R : H; R5: 4-OMe - 3 (3— py r i dy l) -Ph; n = 2) Preparation of Compound Example 1129 (Ar r Ph R ^ HRHR i-P r; R: H; R 5 : 4-OMe-3 (3-pyri dyl) -Ph; n = 2)
'HNMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 88— 2. 02 (lH, m) , 3. 05 (1H, ddd, J = 3. 3, 5. 7, 9. 1Hz) , 3. 45 (IH, d d, J = 9. 1, 14. 3Hz) , 3. 48 (IH, dd, J = 3. 3, 14. 3H z) , 3. 84 (3H, s) , 5. 85 (IH, s) , 7. 00 (IH, dd, J = l. 9, 7. 3Hz) , 7. 01-7. 07 ( 1 H, m) , 7. 25-7. 34 (3H, m) , 7. 46-7. 52 (2H, m) , 7. 82— 7. 87 (1 H, m) , 8. 16-8. 20 (IH, m) , 8. 53 ( 1 H, d, J =4. 8 Hz) , 8. 76 (1H, s) 'HNMR (CDC 1 3) δ : 0. 96 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 88- 2. 02 (lH, m), 3.05 (1H, ddd, J = 3.3, 5.7, 9.1Hz), 3.45 (IH, dd, J = 9.1, 14.3Hz), 3.48 (IH, dd, J = 3 3, 14.3Hz), 3.84 (3H, s), 5.85 (IH, s), 7.00 (IH, dd, J = l. 9, 7.3Hz), 7.01- 7.07 (1H, m), 7.25-7.34 (3H, m), 7.46-7.52 (2H, m), 7.82—7.87 (1H, m), 8.16-8.20 (IH, m), 8.53 (1H, d, J = 4.8 Hz), 8.76 (1H, s)
FAB-MS (m/e) : 423 [M + H] + 実施例 55 FAB-MS (m / e): 423 [M + H] + Example 55
化合物例 1130の製造 (Ai^ Ph R H '- R ^ R3 : i— P r ; R4 : H ; R5 : 4-OMe - 3 (4一 py r i dy l) -Ph; n=2) Preparation of compound examples 1130 (Ai ^ Ph RH '- R ^ R 3: i- P r; R 4: H; R 5: 4-OMe - 3 (4 one py ri dy l) -Ph; n = 2)
XHNMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 4Hz) , 1. 87 -2. 00 ( 1 H, m) , 3. 06 (IH, ddd, J = 3. 4, 5. 7, 9. 2Hz) , 3. 40 (IH, dd, J = 9. 2, 14. 3Hz) , 3. 47 ( 1 H, dd, J = 3. 4, 14. 3H z) , 3. 85 (3H, s) , 5. 85 ( 1 H, s) , 6. 98— 7. 06 (2 H, m) , 7. 28 - 7. 33 (2H, m) , 7. 46 (2H, dd, J = 1. 7, 4. 5Hz) , 7. 49 - 7. 52 (2H, m) , 8. 16-8. 21 (1 H, m) , 8. 60 (2H, dd, J = l. 7, 4. 5Hz) X HNMR (CDC 1 3) δ :. 0. 97 (3H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 4Hz), 1. 87 -2 00 (1 H , m), 3.06 (IH, ddd, J = 3.4, 5.7, 9.2 Hz), 3.40 (IH, dd, J = 9.2, 14.3 Hz), 3.47 ( 1H, dd, J = 3.4, 14.3Hz), 3.85 (3H, s), 5.85 (1H, s), 6.98—7.06 (2H, m), 7.28-7.33 (2H, m), 7.46 (2H, dd, J = 1.7, 4.5 Hz), 7.49-7.52 (2H, m), 8.16-8 . 21 (1 H, m), 8.60 (2H, dd, J = l. 7, 4.5 Hz)
FAB-MS (m/e) : 423 [M + H] + 実施例 56 FAB-MS (m / e): 423 [M + H] + Example 56
化合物例 1131の製造 (Ar : Ph; R1: H; R2: H; R3: i -P r ; R4 : H; R5: 3— COOMe— 4— Me O— P h; n= 2) Preparation of compound examples 1131 (Ar: Ph; R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: 3- COOMe- 4- Me O- P h; n = 2)
XHNMR (CDC 13) δ : 0. 95 (3Η, d, J = 6. 7Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 90— 2. 01 (IH, m) , 2. 94 -3. 04 (lH, m) , 3. 40— 3. 50 (2H, m) , 3. 87 (3H, s) , 3. 92 (3H, s) , 5. 82 ( 1 H, s) , 6. 90 - 7. 00 (2 H, m) , 7. 45 - 7. 52 (2H, m) , 7. 65— 7. 80 (2H, m) , 8. 12— 8. 20 (1H, m) X HNMR (CDC 1 3) δ : 0. 95 (3Η, d, J = 6. 7Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 90- 2. 01 (IH, m), 2.94 -3.04 (lH, m), 3.40—3.50 (2H, m), 3.87 (3H, s), 3.92 (3H, s), 5.82 (1 H, s), 6.90-7.00 (2 H, m), 7.45-7.52 (2H, m), 7.65—7.80 (2H, m), 8.12—8.20 (1H, m)
FAB-MS (mZe) : 404 [M + H] + 実施例 57 FAB-MS (mZe): 404 [M + H] + Example 57
化合物例 1 136の製造 (Ai^ Ph ; !^1 : !! ; !^2 : !! ; !^3 : i— P r ; R4 : H; R5: 3-COhyd r oxy i nd o l y 1 -4-MeO-Ph ; n = 2) Preparation of Compound Example 1 136 (Ai ^ Ph;! ^ 1 : !!;! ^ 2 : !!;! ^ 3 : i—P r; R 4 : H; R 5 : 3-COhyd roxy ind oly 1 -4-MeO-Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 95 (3H, d, J = 7. 0Hz) , 1. 0 0 (3H, d, J = 6. 6Hz) , 1. 85— 2. 00 ( 1 H, m) , 3. 05 一 3. 25 (3H, m) , 3. 35 - 3. 50 (4H, m) , 3. 85 (3H, s) , 5. 79 (1H, s) , 6. 90 - 7. 25 ( 7 H, m) , 7. 40— 7 . 55 (2H, m) , 8. 10— 8. 20 (lH, m) , 8. 25 -8. 35 ( 1H, m) X HNMR (CDC 1 3) ( 5: 0. 95 (3H, d, J = 7. 0Hz), 1. 0 0 (3H, d, J = 6. 6Hz), 1. 85- 2. 00 (1 H, m), 3.05-3.25 (3H, m), 3.35-3.50 (4H, m), 3.85 (3H, s), 5.79 (1H, s), 6 90-7.25 (7H, m), 7.40—7.55 (2H, m), 8.10—8.20 (lH, m), 8.25 -8.35 (1H, m )
FAB -MS (m/e) : 491 [M + H] + 実施例 58 FAB-MS (m / e): 491 [M + H] + Example 58
化合物例 1 137の製造 (Ar : Ph ; R1: H; R2: H; R3: i— P r ; R4 : H ; R5 : 4-PhCOO-Ph ; n=2) Preparation of compound examples 1 137 (Ar: Ph; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 4-PhCOO-Ph; n = 2)
iHNMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 5Hz) , 1. 0 1 (3H, d, J = 6. 9Hz) , 1. 90- 2. 05 ( 1 H, m) , 2. 95 —3. 05 (IH, m) , 3. 35— 3. 50 (2H, m) , 5. 88 (IH, s) , 6. 96 - 7. 00 (lH, m) , 7. 20 - 7. 32 (2H, m) , 7 . 45-7. 56 (4H, m) , 7. 62 - 7. 70 (lH, m) , 7. 92 ( IH, s) , 8. 1 5-8. 18 ( 1 H, m) , 8. 27 (2H, d. J = 8. 6Hz) iHNMR (CDC 1 3) δ: 0. 97 (3H, d, J = 6. 5Hz), 1. 0 1 (3H, d, J = 6. 9Hz), 1. 90- 2. 05 (1 H, m), 2.95 — 3.05 (IH, m), 3.35 — 3.50 (2H, m), 5.88 (IH, s), 6.96-7.00 (lH, m) , 7.20-7.32 (2H, m), 7.45-7.56 (4H, m), 7.62-7.70 (lH, m), 7.92 (IH, s), 8 . 15-8.18 (1H, m), 8.27 (2H, d. J = 8.6Hz)
FAB-MS (m/e) : 562 [M + H] + 実施例 59 化合物例 1 139の製造 (Ar : Ph; R1: H; R2: H; R3: i— P r ; R4 : H; R5: 4— (4— N3) — PhCOO— Ph; n=2) FAB-MS (m / e): 562 [M + H] + Example 59 Preparation of compound examples 1 139 (Ar: Ph; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 4- (4- N3) - PhCOO- Ph; n = 2)
HNMR (CDC 1 J δ : 0. 97 (3Η, d, J = 6. 8Hz) 0 HNMR (CDC 1 Jδ: 0.97 (3Η, d, J = 6.8 Hz) 0
1 (3H, d, J = 6. 6Hz) , 1. 85— 2. 00 ( 1 H, m) , 2. 98 一 3. 05 (IH, m) , 3. 35— 3. 50 (2H, m) , 5. 88 ( 1 H, s) , 6. 97 - 7. 00 (1H, m) , 7. 17 (2H, d, J = 8. 3Hz ) , 7. 23 (1H, d, J = 8. 3Hz) , 7. 31 ( 1 H, dd, J = 1. 9, 8. 3Hz) , 7. 48- 7. 55 (2H, m) , 7. 92 (1H, d, J =1. 9Hz) , 8. 15-8. 18 ( 1 H, m) , 8. 26 (2H, d. J = 8. 3Hz) 1 (3H, d, J = 6.6 Hz), 1.85—2.00 (1H, m), 2.98-1.05 (IH, m), 3.35—3.50 (2H, m), 5.88 (1H, s), 6.97-7.00 (1H, m), 7.17 (2H, d, J = 8.3Hz), 7.23 (1H, d, J = 8.3Hz), 7.31 (1H, dd, J = 1.9, 8.3Hz), 7.48-7.55 (2H, m), 7.92 (1H, d, J = 1) 9 Hz), 8.15-8. 18 (1H, m), 8.26 (2H, d. J = 8.3Hz)
FAB-MS (m/e) : 603 [M + H] + 実施例 60 FAB-MS (m / e): 603 [M + H] + Example 60
化合物例 1 146の製造 Preparation of Compound Example 1 146
ジァステレオマー a (Ar : Ph ; Rx : H ; R2 : H ; R3 : i一 P r ; R4 : H ; R5: 3-1, 4-nP rNHCOCH20-Ph; n=0) Jiasutereoma a (Ar: Ph; R x : H; R 2: H; R 3: i one P r; R 4: H; R 5: 3-1, 4-nP rNHCOCH20-Ph; n = 0)
XHNMR (CDC 13) d : 0. 94 (3H, d, J = 6. 6Hz) , 0. 9 5 (3H, d, J = 6. 7Hz) , 1. 00 (3H, t, J = 7. 4Hz) , 1 . 55- 1. 74 (3H, m) , 2. 49 ( 1 H, d d, J = 10. 8, 13. 9Hz) , 2. 89 (IH, dd, J = 2. 2, 13. 9Hz) , 3. 01 (1 H, ddd. J = 2. 2, 5. 1, 10. 8Hz) , 3. 35— 3. 42 (2H , m) , 4. 54 (2H, s) , 5. 64 ( 1 H, s) , 6. 54- 6. 58 ( lH, m) , 6. 80 (1H, d, J = 8. 4Hz) , 7. 00-7. 15 (2 H, m) , 7. 40 (IH, d d, J = 2. 0, 8. 5Hz) , 7. 63-7. 66 (1H, m) , 7. 88 (IH, d, J = 2. 0Hz) X HNMR (CDC 1 3) d : 0. 94 (3H, d, J = 6. 6Hz), 0. 9 5 (3H, d, J = 6. 7Hz), 1. 00 (3H, t, J = 7.4 Hz), 1.55-1.74 (3H, m), 2.49 (1H, dd, J = 10.8, 13.9Hz), 2.89 (IH, dd, J = 2. 2, 13.9 Hz), 3.01 (1 H, ddd. J = 2.2, 5.1, 10.8 Hz), 3.35—3.42 (2H, m), 4.54 (2H, s), 5.64 (1H, s), 6.54-6.58 (lH, m), 6.80 (1H, d, J = 8.4Hz), 7.00-7.15 (2 H, m), 7.40 (IH, dd, J = 2.0, 8.5Hz), 7.63-7.66 (1H, m), 7.88 (IH, d, J = 2.0Hz) )
FAB-MS ( /e) : 525 [M + H] + FAB-MS (/ e): 525 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i— P r ; R4 : H ; R5: 3-1, 4-nP rNHCOCH20-Ph; n=0) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i- P r; R 4: H; R 5: 3-1, 4-nP rNHCOCH20-Ph; n = 0)
iHNMR (CDC 13) δ : 0. 90— 1. 05 (9H, m) , 1. 55- 1 . 70 (2H, m) , 1. 72 - 1. 85 (3H, m) , 1. 90- 2. 00 ( 1H, m) , 3. 35 - 3. 40 (2H, m) , 4. 52 (2H, s) , 5. 6 6 (1H, s) , 6. 70- 6. 80 (2H, m) , 6. 95— 7. 05 (IH , m) , 7. 06-7. 15 (2H, m) , 7. 48— 7. 52 (IH, m) , 7. 83 (IH, d, J = 2. 2Hz) iHNMR (CDC 1 3) δ: 0. 90- 1. 05 (9H, m), 1. 55- 1 70 (2H, m), 1.72-1.85 (3H, m), 1.90-2.00 (1H, m), 3.35-3.40 (2H, m), 4.52 (2H, s), 5.66 (1H, s), 6.70-6.80 (2H, m), 6.95—7.05 (IH, m), 7.06-7.15 ( 2H, m), 7.48-7.52 (IH, m), 7.83 (IH, d, J = 2.2 Hz)
FAB-MS (m/e) : 525 [M + H] + 実施例 61 FAB-MS (m / e): 525 [M + H] + Example 61
化合物例 1147の製造 Preparation of Compound Example 1147
ジァステレオマ一 a (Ar i P i R^ H j R^- H j R3 : i— P r ; R4 : H ; R5 : 3 - I, 4-n-P rNHCOCH20-Ph; n=2)Diastereomer a (Ar i P i R ^ H j R ^-H j R 3 : i— P r; R 4 : H; R 5 : 3-I, 4-nP rNHCOCH 20 -Ph; n = 2)
NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 01 (3H, t, J = 7. 4Hz) , 1 . 50- 1. 70 (2H, m) , 1. 70 - 1. 85 (IH, m) , 3. 05 ( IH, dd, J =l 1. 8, 14. 7Hz) , 3. 38 (IH, t, J = 6. 5 Hz) , 3. 40 (1H, t, J = 6. 5Hz) , 3. 42-3. 52 (2H, m) , 4. 56 (2H, s) , 5. 88 (1H, s) , 6. 70— 6. 73 (1 H, m) , 6. 82 (IH, d, J = 8. 5Hz) , 7. 02 (IH, b r s) , 7. 37 (1 H, dd, J = 2. 1, 8. 5Hz) , 7. 37 - 7. 43 (2 H, m) , 7. 85 (1H, d, J = 2. 1Hz) , 8. 13-8. 17 (1 H , m) NMR (CDC 1 3) δ: 0. 98 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 01 (3H, t, J = 7 .4Hz), 1.50-1.70 (2H, m), 1.70-1.85 (IH, m), 3.05 (IH, dd, J = l1.8,14.7Hz), 3.38 (IH, t, J = 6.5 Hz), 3.40 (1H, t, J = 6.5 Hz), 3.42-3.52 (2H, m), 4.56 (2H, s), 5.88 (1H, s), 6.70—6.73 (1H, m), 6.82 (IH, d, J = 8.5Hz), 7.02 (IH, brs), 7.37 (1 H, dd, J = 2.1, 8.5 Hz), 7.37-7.43 (2 H, m), 7.85 (1H, d, J = 2.1 Hz), 8 . 13-8. 17 (1 H, m)
FAB-MS (m/e) : 557 [M + H] + FAB-MS (m / e): 557 [M + H] +
ジァステレオマー b (Ar ^h j R' i H j R^ H i R3 : i -P r ; R4 : H ; R5 : 3— I, 4-n-P rNHCOCH20-P ; n=2) Jiasutereoma b (Ar ^ hj R 'i H j R ^ H i R 3: i -P r; R 4: H; R 5: 3- I, 4-nP rNHCOCH 2 0-P; n = 2)
^NMR (CDC 13) δ : 0. 96 ( 3 H, d, J = 6. 8Hz) , 1. 0 0 (3H, t, J = 7. 4Hz) , 1. 01 (3H, d, J = 6. 7Hz) , 1 . 53 - 1. 70 (2H, m) , 1. 90 - 1. 94 (IH, m) , 2. 98 - 3. 05 (IH, m) , 3. 30-3. 49 (4H, m) , 4. 52 (1H, s ) , 5. 79 (2H, s) , 6. 75 ( 1 H, d, J = 8. 3Hz) , 6. 93 -7. 00 (IH, m) , 7. 01 (IH, b r s) , 7. 18 (1H, d d, J = 1. 9, 8. 3Hz) , 7. 48- 7. 55 (2H, m) , 7. 83 (1 H , d, J = l. 9Hz) , 8. 15-8. 19 (IH, m) ^ NMR (CDC 1 3) δ : 0. 96 (3 H, d, J = 6. 8Hz), 1. 0 0 (3H, t, J = 7. 4Hz), 1. 01 (3H, d, J = 6.7 Hz), 1.53-1.70 (2H, m), 1.90-1.94 (IH, m), 2.98-3.05 (IH, m), 3.30-3 49 (4H, m), 4.52 (1H, s), 5.79 (2H, s), 6.75 (1H, d, J = 8.3 Hz), 6.93 -7.00 (IH, m), 7.01 (IH, brs), 7.18 (1H, dd, J = 1.9, 8.3 Hz), 7.48-7.55 (2H, m) , 7.83 (1 H, d, J = l. 9Hz), 8.15-8.19 (IH, m)
FAB-MS (m/e) : 557 [M + H] + 実施例 62 FAB-MS (m / e): 557 [M + H] + Example 62
化合物例 1 148の製造 Preparation of Compound Example 1 148
ジァステレオマー a (Ar : Ph ; R1 : H ; R2 : H ; R3 : i P r ; R4 : H ; R5 : 2 - 1 , 5-n-P r HCOCH20-Ph ; n=2) Jiasutereoma a (Ar: Ph; R 1 : H; R 2: H; R 3: i P r; R 4: H; R 5: 2 - 1, 5-nP r HCOCH 2 0-Ph; n = 2)
^NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 01 (3H, t, J = 7. 4Hz) , 1 . 55 - 1. 69 (2H, m) , 1. 71 -1. 86 (IH, m) , 3. 04 ( IH, d d, J =l 1. 8, 14. 7Hz) , 3. 36 ( 1 H, t, J = 6. 9 Hz) , 3. 38 (1H, t, J = 6. 9Hz) , 3. 45 - 3. 50 ( 1 H, m) , 3. 47 (IH, d d, J = 2. 4, 14. 7Hz) , 4. 51 (2H, s) , 5. 94 (IH, s) , 6. 68- 6. 72 ( 1 H, m) , 6. 82 (1 H, dd, J = 1. 8, 8. 0Hz) , 6. 95 ( 1 H, d, J = 1. 8Hz) , 7. 04 (IH, b r s) , 7. 36- 7. 46 (2H, m) , 7. 79 (1 H, d, J = 8. 0Hz) , 8. 1 -8. 18 (IH, m) ^ NMR (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 01 (3H, t, J = (7.4Hz), 1.55-1.69 (2H, m), 1.71-1.86 (IH, m), 3.04 (IH, dd, J = l 1.8, 14.7Hz) , 3.36 (1H, t, J = 6.9Hz), 3.38 (1H, t, J = 6.9Hz), 3.45-3.50 (1H, m), 3.47 (IH, dd, J = 2.4, 14.7 Hz), 4.51 (2H, s), 5.94 (IH, s), 6.68-6.72 (1H, m), 6. 82 (1 H, dd, J = 1.8, 8.0 Hz), 6.95 (1 H, d, J = 1.8 Hz), 7.04 (IH, brs), 7.36-7.46 (2H, m), 7.79 (1 H, d, J = 8.0 Hz), 8.1 -8.18 (IH, m)
FAB-MS (m/e) : 557 [M + H] + FAB-MS (m / e): 557 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i一 P r ; R4 : H ; 5 : 2- 1 , 5-n-P rNHCOCH20-Ph ; n=2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4: H; 5: 2- 1, 5-nP rNHCOCH 2 0-Ph; n = 2)
^NMR (CDC 13) (5 : 0. 96 (3H, d, J = 6. 9Hz) , 1. 0 0 (3H, t, J = 7. 4Hz) , 1. 01 (3H, d, J = 6. 8Hz) , 1 . 55— 1. 65 (2H, m) , 1. 87— 2. 00 ( 1 H, m) , 3. 31 - 3. 45 (3H, m) , 4. 43 ( 1 H, d, J =l 3. 9Hz) , 4. 46 ( 1H, d, J =l 3. 9Hz) , 5. 82 ( 1 H, s) , 6. 70 ( 1 H, dd , J = 1. 3, 8. 2Hz) , 6. 82 ( 1 H, d, J = 1. 3Hz) , 6. 9 5-7. 02 (1H, m) , 7. 03 (IH, b r s) , 7. 49-7. 54 ( 2H, m) , 7. 73 (IH, d, J = 8. 2Hz) , 8. 16-8. 20 (1 H, m) ^ NMR (CDC 1 3) ( 5: 0. 96 (3H, d, J = 6. 9Hz), 1. 0 0 (3H, t, J = 7. 4Hz), 1. 01 (3H, d, J = 6.8Hz), 1.55-1.65 (2H, m), 1.87-2.00 (1H, m), 3.31-3.45 (3H, m), 4.43 ( 1H, d, J = l 3.9Hz), 4.46 (1H, d, J = l 3.9Hz), 5.82 (1H, s), 6.70 (1H, dd, J = 1. 3, 8.2 Hz), 6.82 (1 H, d, J = 1.3 Hz), 6.95-7.02 (1H, m), 7.03 (IH, brs), 7. 49-7. 54 ( 2H, m), 7.73 (IH, d, J = 8.2 Hz), 8.16-8.20 (1 H, m)
FAB-MS (m/e) : 557 [M + H] + 実施例 63 FAB-MS (m / e): 557 [M + H] + Example 63
化合物例 1149の製造 (Ar : Ph; R1: H; R2: H; R3: i一 P r ; R4 : H ; R5 : 5- 1 -2 -n-P r NHCOCH^O-Ph; n = 2) Preparation of compound examples 1149 (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4: H; R 5: 5- 1 -2 -nP r NHCOCH ^ O-Ph; n = 2)
iHNMR (CDC 13) d : 0. 75 (3H, t, J = 7. 4Hz) , 1. 0 0 (3H, d, J = 6. 7Hz) , 1. 00 (3H, d, J = 6. 7Hz) , 1 . 32 - 1. 47 (2H, m) , 1. 77 - 1. 89 (IH, m) , 3. 04 ( 1H, d d, J = 11. 8, 14. 8Hz) , 3. 05 - 3. 20 (2H, m) , 3. 51 (IH, dd, J = 2. 2, 14. 8Hz) , 3. 48-3. 58 ( IH, m) , 4. 16 (1H, d, J = 14. 0Hz) , 4. 34 (IH, d, J = 14. 0Hz) , 6. 35 (1H, s) , 6. 56-6. 64 ( 1 H, m) , 6. 60 (IH, d, J = 8. 5Hz) , 6. 66— 6. 69 ( 1 H, m) , 7. 37 -7. 46 (2H, m) , 7. 66 (IH, dd, J = 1. 9, 8. 5 Hz) , 7. 93 (IH, d, J = 1. 9Hz) , 8. 10-8. 14 ( 1 H, m) iHNMR (CDC 1 3) d: 0. 75 (3H, t, J = 7. 4Hz), 1. 0 0 (3H, d, J = 6. 7Hz), 1. 00 (3H, d, J = 6 7 Hz), 1.32-1.47 (2H, m), 1.77-1.89 (IH, m), 3.04 (1H, dd, J = 11.8, 14.8 Hz), 3 .05-3.20 (2H, m), 3.51 (IH, dd, J = 2.2, 14.8 Hz), 3.48-3.58 (IH, m), 4.16 (1H, m d, J = 14.0 Hz), 4.34 (IH, d, J = 14.0 Hz), 6.35 (1H, s), 6.56-6.64 (1H, m), 6.60 (IH, d, J = 8.5 Hz), 6.66-6.69 (1H, m), 7.37 -7. 46 (2H, m), 7.66 (IH, dd, J = 1 9, 8.5 Hz), 7.93 (IH, d, J = 1.9 Hz), 8.10-8.14 (1 H, m)
FAB-MS (m/e) : 557 [M + H] + 実施例 64 FAB-MS (m / e): 557 [M + H] + Example 64
化合物例 1150の製造 Preparation of Compound Example 1150
ジァステレオマ一 a (A r : Ph; R1: H; R2: H; R3: i一 P r ; R4 : H ; R5: 3-CH20H-4-OMe-Ph ; n = 2) Jiasutereoma one a (A r: Ph; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 3-CH20H-4-OMe-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 97 ( 3 H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 70— 1. 85 (1H, m) , 3. 07 (IH, dd, J = 11. 4, 14. 4Hz) , 3. 43 (IH, d d, J = 2 . 2, 14. 4Hz) , 3. 45 - 3. 49 (lH, m) , 3. 91 (3H, s ) , 4. 71 (2H, s) , 5. 81 ( 1 H, s) , 6. 73— 6. 76 (IH , m) , 6. 93 (IH, d, J = 8. 3Hz) , 7. 27-7. 39 (4H, m) , 8. 12-8. 15 ( 1 H, m) XHNMR (CDC 1 3) δ: 0. 97 (3 H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 70- 1. 85 (1H, m), 3.07 (IH, dd, J = 11.4, 14.4 Hz), 3.43 (IH, dd, J = 2.2, 14.4 Hz), 3.45-3.49 (lH , M), 3.91 (3H, s), 4.71 (2H, s), 5.81 (1H, s), 6.73—6.76 (IH , m), 6.93 (IH, d, J = 8.3 Hz), 7.27-7.39 (4H, m), 8.12-8.15 (1 H, m)
FAB-MS (m/e) : 376 [M + H] + FAB-MS (m / e): 376 [M + H] +
ジァステレオマー b (A r : P h ; R1 : H ; R2 H; R i -P r ; R4 : HDiastereomer b (A r: P h; R 1 : H; R 2 H; R i -P r; R 4 : H
; R5 : 3-CH20H-4-MeO-Ph; n=2) R 5 : 3-CH20H-4-MeO-Ph; n = 2)
^NMR (CDC 13) (5 : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 90— 2. 00 (IH, m) , 2. 95 —3. 02 (lH, m) , 3. 38- 3. 52 (2H, m) , 3. 88 (3H, s) , 4. 68 (2H, s) , 5. 79 ( 1 H, s) , 6. 88 ( 1 H, d, J =8. 3Hz) , 6. 93 - 6. 96 ( 1 H, m) , 7. 18 ( 1 H, dd, J =2. 0, 8. 3Hz) , 7. 29 ( 1 H, d, J = 2. 0Hz) , 7. 45— 7. 48 (2H, m) , 8. 13— 8. 16 ( 1 H, m) ^ NMR (CDC 1 3) ( 5: 0. 94 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 90- 2. 00 (IH , m), 2.95 — 3.02 (lH, m), 3.38-3.52 (2H, m), 3.88 (3H, s), 4.68 (2H, s), 5. 79 (1H, s), 6.88 (1H, d, J = 8.3Hz), 6.93-6.96 (1H, m), 7.18 (1H, dd, J = 2 0, 8.3Hz), 7.29 (1H, d, J = 2.0Hz), 7.45—7.48 (2H, m), 8.13—8.16 (1H, m)
FAB-MS (m/e) : 376 [M + H] + 実施例 65 FAB-MS (m / e): 376 [M + H] + Example 65
化合物例 1151の製造 (Ar : Ph; R1: H; R2: H; R3: i - P r ; R4 : H; R5: 3 - (1, 2-d i hyd r oxye t hy l) -4-Me-Ph ; n=2) Preparation of compound examples 1151 (Ar: Ph; R 1 : H; R 2: H; R 3: i - P r; R 4: H; R 5: 3 - (1, 2-di hyd r oxye t hy l ) -4-Me-Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 92— 0. 98 (6H, m) , 1. 85— 1 . 95 (IH, m) , 3. 00— 3. 10 ( 1 H, m) , 3. 35- 3. 50 ( 2H, m) , 3. 60— 3. 70 (IH, m) , 3. 75 - 3. 85 ( 1 H, m ) , 3. 85 (3H, s) , 5. 00 - 5. 08 (lH, m) , 5. 78 (1 H , s) , 6. 86 (1H, d d, J = l. 5, 9. 1) , 6. 95— 7. 02 ( IH, m) , 7. 12-7. 23 (IH, m) , 7. 45- 7. 55 (3H, m ) , 8. 15-8. 18 (1 H, m) X HNMR (CDC 1 3) ( 5:. 0. 92- 0. 98 (6H, m), 1. 85- 1 95 (IH, m), 3. 00- 3. 10 (1 H, m), 3.35-3.50 (2H, m), 3.60—3.70 (IH, m), 3.75-3.85 (1H, m), 3.85 (3H, s), 5 00-5.08 (lH, m), 5.78 (1 H, s), 6.86 (1H, dd, J = l. 5, 9.1), 6.95—7.02 (IH , m), 7.12-7.23 (IH, m), 7.45-7.55 (3H, m), 8.15-8.18 (1 H, m)
FAB-MS (m/e) : 406 [M+H] + 実施例 66 FAB-MS (m / e): 406 [M + H] + Example 66
化合物例 1152の製 (A r—: Ph; R1: H; R2: H; R3: i -P r ; R4 : H; R5 : 3-CH2QMe-4-MeO-Ph; n=2) Made in Compound Example 1152 (A r-: Ph; R 1: H; R 2: H; R 3: i -P r; R 4 : H; R 5 : 3-CH2QMe-4-MeO-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 6Hz) , 1. 90 - 2. 05 (IH, m) , 2. 95 一 3. 04 (lH, m) , 3. 41 (3H, s) , 3. 45- 3. 52 (2H, m) , 3. 85 (3H, s) , 4. 49 (2H, s) , 5. 79 (1H, s) , 6. 86 (1H, d, J = 8. 5Hz) , 6. 90 - 6. 95 (IH, m) , 7 . 17 (1H, dd, J = 2. 0, 8. 5Hz) , 7. 37 (1H, d, J = 2 • 0Hz) , 7. 44- 7. 47 (2H, m) , 8. 12-8. 15 (lH, m ) X HNMR (CDC 1 3) δ : 0. 95 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 6Hz), 1. 90 - 2. 05 (IH, m), 2.95-3.04 (lH, m), 3.41 (3H, s), 3.45-3.52 (2H, m), 3.85 (3H, s), 4.49 (2H, s), 5.79 (1H, s), 6.86 (1H, d, J = 8.5 Hz), 6.90-6.95 (IH, m), 7.17 (1H, dd , J = 2.0, 8.5Hz), 7.37 (1H, d, J = 2 • 0Hz), 7.44-7.47 (2H, m), 8.12-8.15 (lH, m)
FAB-MS (mZe) : 390 [M + H] + 実施例 67 FAB-MS (mZe): 390 [M + H] + Example 67
化合物例 1153の製造 (Ar Pt^ R^ H R H ; !^3 : i -P r ; R4 Preparation of Compound Example 1153 (Ar Pt ^ R ^ HRH;! ^ 3 : i -P r; R 4
: H; R5: 3-CH2NHSQ2Me-4-MeO-Ph; n = 2) : H; R 5 : 3-CH2NHSQ2Me-4-MeO-Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 95 ( 3 H, d, J = 6. 9Hz) , 1. 0 XHNMR (CDC 1 3) (5 : 0. 95 (3 H, d, J = 6. 9Hz), 1. 0
0 (3H, d, J = 6. 7Hz) , 1. 90— 2. 04 ( 1 H, m) , 2. 79 (3H, s) , 3. 02-3. 12 ( 1 H, m) , 3. 35 - 3. 50 (2H, m) , 3. 88 (3H, s) , 4. 27— 4. 30 (2H, m) , 4. 91一 40 (3H, d, J = 6.7Hz), 1.90—2.44 (1H, m), 2.79 (3H, s), 3.02-3.12 (1H, m), 3.35-3.50 (2H, m), 3.88 (3H, s), 4.27—4.30 (2H, m), 4.91-1 4
. 92 (IH, m) , 5. 77 (IH, s) , 6. 88 (1H, d, J = 8. 5 Hz) , 6. 97- 7. 00 (IH, m) , 7. 16 (1H, dd, J = 2. 092 (IH, m), 5.77 (IH, s), 6.88 (1H, d, J = 8.5 Hz), 6.97-7.00 (IH, m), 7.16 ( 1H, dd, J = 2.0
, 8. 5Hz) , 7. 21-7. 26 (lH, m) , 7. 48-7. 51 (2H, 8.5 Hz), 7.21-7.26 (lH, m), 7.48-7.51 (2H
, m) , 8. 16-8. 19 ( 1 H, m) , M), 8.16-8.19 (1 H, m)
FAB-MS (m/e) : 453 [M + H] + 実施例 68 FAB-MS (m / e): 453 [M + H] + Example 68
化合物例 1154の製造 (Ar : Ph; R1: H; R2: H; R3: i一 P r ; R4 : H; R5: 3-CH2NMe S〇2Me— 4— MeO— Ph ; n=2)Preparation of compound examples 1154 (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4: H; R 5: 3-CH2NMe S_rei_2Me- 4-MeO- Ph; n = 2)
NMR (CDC 13) 5 : 0. 95 (3H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 90-2. 01 ( 1 H, m) , 2. 79 (3H, s) , 2. 81 (3H, s) , 3. 02-3 10 (IH, m) , 3. 32- 3. 49 (2H, m) , 3. 84 (3H, s) 4. 36 (2H, s) , 5. 76 (IH, s) , 6. 86 (IH, d, J = 8 5Hz) , 6. 98-7 . 04 (1H, m) , 7. 12 (IH, d d, J = 2 3, 8. 5Hz) , 7. 35 (IH, d, J = 2. 3Hz) , 7. 47 - 7. 52 (2H, m) , 8 5-8. 1 9 (1H, m) NMR (CDC 1 3) 5: . 0. 95 (3H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 90-2 01 (1 H, m), 2.79 (3H, s), 2.81 (3H, s), 3.02-3 10 (IH, m), 3.32-3.49 (2H, m), 3.84 (3H, s) 4. 36 (2H, s), 5.76 (IH, s), 6.86 (IH, d, J = 85 Hz), 6.98-7.04 (1H, m), 7.12 (IH, dd) , J = 23, 8.5Hz), 7.35 (IH, d, J = 2.3Hz), 7.47-7.52 (2H, m), 85-8.19 (1H, m )
FAB-MS (m/e) : 467 [M + H] + 実施例 69 FAB-MS (m / e): 467 [M + H] + Example 69
化合物例 1 1 55の製造 (Ar ^h ^^- H i R' i H ^3 : i -P r ; R4 : H; R5: 3 - (4-CH2QH) Ph-4-MeQ-Ph; n=2) iHNMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 90-2. 00 (IH, m) , 3. 00 一 3. 08 (lH, m) , 3. 40- 3. 55 (2H, m) , 3. 83 (3H, s) , 4. 72 (2H, s) , 5. 83 (1 H, s) , 6. 95 - 7. 05 (2 H, m) , 7. 22 - 7. 30 (2H, m) , 7. 36 - 7. 60 (6H, m) , 8. 14-8. 17 (1H, m) Preparation of compound examples 1 1 55 (Ar ^ h ^^ - H i R 'i H ^ 3: i -P r; R 4: H; R 5: 3 - (4-CH2QH) Ph-4-MeQ-Ph ; n = 2) iHNMR (CDC 1 3) δ: 0. 96 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 90-2. 00 (IH, m), 3.00-3.08 (lH, m), 3.40-3.55 (2H, m), 3.83 (3H, s), 4.72 (2H, s) , 5.83 (1 H, s), 6.95-7.05 (2 H, m), 7.22-7.30 (2H, m), 7.36-7.60 (6H, m) , 8. 14-8. 17 (1H, m)
FAB -MS (m/e) : 452 [M + H] + 実施例 Ί 0 FAB-MS (m / e): 452 [M + H] + Example Ί 0
化合物例 1 1 56の製造 (Ar : P h ; R1: H; R2: H; R3: i— P r ; R4 : H; R5 : 3-Me SQ2N (Me) CH2 - 4一 Me - Ph ; n=2) R 1 : H; R 2 : H; R 3 : i—Pr; R 4 : H; R 5 : 3-Me SQ2N (Me) CH2-4 Me-Ph; n = 2)
iHNMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 7Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 90— 2. 01 (lH, m) , 2. 39 (3H, s) , 2. 66 (3H, s) , 2. 81 (H, S) , 3. 05 (1H, m) , 3. 40 -3. 55 (2H, m) , 4. 21 (IH, d, J =14. OH z) , 4. 3 1 (1H, d, J = 14. 0Hz) , 5. 79 (1H, s) , 6. 95 - 6. 98 (lH, m) , 7. 10 ( 1 H, s) , 7. 19— 7. 20 (2 H, m) , 7. 48— 7. 51 (2H, m) , 8. 16-8. 19 (lH, m) FAB-MS (mZe) : 451 [M + H] + 実施例 71 iHNMR (CDC 1 3) (5 : 0. 95 (3H, d, J = 6. 7Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 90- 2. 01 (lH, m), 2.39 (3H, s), 2.66 (3H, s), 2.81 (H, S), 3.05 (1H, m), 3.40 -3.55 (2H, m ), 4.21 (IH, d, J = 14.OHz), 4.31 (1H, d, J = 14.0 Hz), 5.79 (1H, s), 6.95-6.98 (lH, m), 7.10 (1H, s), 7.19—7.20 (2H, m), 7.48—7.51 (2H, m), 8.16-8.19 (lH, m) FAB-MS (mZe): 451 [M + H] + Example 71
化合物例 1161の製造 (Ai^ y—HO— Ph -^ R^ H R2 : !! ; !^3 : i -P r ; R4 : H ; R 5: P h; n = 0 ) Preparation of compound examples 1161 (Ai ^ y-HO- Ph - ^ R ^ HR 2:! !!; ^ 3: i -P r; R 4: H; R 5: P h; n = 0)
^NMR (CDC 13) ά : 0. 94 (3H, d, J = 6. 8Hz) , 0. 9 5 (3H, d, J = 6. 8Hz) , 1. 55— 1. 75 ( 1 H, m) , 2. 48 (1H, dd, J = 10. 8, 13. 9Hz) , 2. 83 (1H, d d, J = 2 . 2, 13. 9Hz) , 3. 00 - 3. 08 (1H, m) , 5. 64 ( 1 H, s ) , 6. 03 (1H, d, J = 2. 6Hz) , 6. 56 ( 1 H, dd, J = 2. 6, 8. 3Hz) , 7. 24- 7. 48 (5H, m) , 7. 50 ( 1 H, d, J =8. 3Hz) ^ NMR (CDC 1 3) ά : 0. 94 (3H, d, J = 6. 8Hz), 0. 9 5 (3H, d, J = 6. 8Hz), 1. 55- 1. 75 (1 H , m), 2.48 (1H, dd, J = 10.8, 13.9Hz), 2.83 (1H, dd, J = 2.2, 13.9Hz), 3.00-3.08 ( 1H, m), 5.64 (1H, s), 6.03 (1H, d, J = 2.6Hz), 6.56 (1H, dd, J = 2.6, 8.3Hz), 7.24-7.48 (5H, m), 7.50 (1H, d, J = 8.3Hz)
FAB-MS (m/e) : 300 [M + H] + 実施例 72 FAB-MS (m / e): 300 [M + H] + Example 72
化合物例 1162の製造 (3—イソプロピル— 7—メトキシー 5—フエ二ルー 2 , 3, 4, 5—テトラヒドロ一 Iff— 1 λ 6, 4一べンゾチアゼピン一 1, 1— ジオン) Preparation of compound examples 1162 (3-isopropyl - 7-methoxy-5-phenylene Lou 2, 3, 4, 5-tetrahydro one Iff- 1 λ 6, 4 one base Nzochiazepin one 1, 1-dione)
水素化ナトリウム (1. 75 g, 43. 9mmo 1) を 2—ヒドロキシ— 4一 メ卜キシベンゾフエノン (5. 00 g, 21. 9mmo 1) のジメチルホルムァ ミド (20. 0ml) 溶液にゆっくり加え、 室温で 30分攪拌した。 ジメチルチ 才力ルバモイルク口ライド (3. 13 g, 25. 2 mm o 1 ) を加え、 60 で 一晩攪拌した。 反応混合物を水にあけ、 酢酸ェチルで 2回抽出した。 有機層を 2 N水酸化ナトリウム水溶液、 飽和食塩水で洗浄し、 乾燥し、 減圧下濃縮した。 残 渣をシリカゲル上で、 へキサン Z酢酸ェチル (80Z20) を溶離剤とするカラ ムクロマトグラフィーに供し、 所望の淡黄色固体 0— (2—べンゾィルー 4ーメ トキシフエ二ル) N, N—ジメチルカルバモチォエート (2. 81 g, 8. 90 mmo 1) を得た (収率 41 %) 。 Sodium hydride (1.75 g, 43.9 mmol 1) was added to a solution of 2-hydroxy-4-methoxybenzophenone (5.00 g, 21.9 mmol 1) in dimethylformamide (20.0 ml). It was added slowly and stirred at room temperature for 30 minutes. Dimethylthiol Rubamoiruku Mouth Ride (3.13 g, 25.2 mmo 1) was added, and the mixture was stirred at 60 overnight. The reaction mixture was poured into water and extracted twice with ethyl acetate. The organic layer was washed with a 2N aqueous solution of sodium hydroxide and saturated saline, dried, and concentrated under reduced pressure. The residue was subjected to column chromatography on silica gel, using hexane Z ethyl acetate (80Z20) as the eluent, to give the desired pale yellow solid 0— (2-benzoyl 4-methoxyphenyl) N, N— Dimethylcarbamothioate (2.81 g, 8.90 mmo 1) was obtained (yield 41%).
^NMR (CDC 13) δ : 3. 09 (3Η, s) , 3. 28 (3H, s) , 3. 87 (3H, s) , 6. 73 ( 1 H, d, J = 2. 4Hz) , 6. 82 (1 ^ NMR (CDC 1 3) δ : 3. 09 (3Η, s), 3. 28 (3H, s), 3.87 (3H, s), 6.73 (1H, d, J = 2.4 Hz), 6.82 (1
H, d d, J = 8. 6, 2. 4Hz) , 7. 38 - 7. 56 (4H, m) , 7. 74- 7. 78 (2H, m) H, d d, J = 8.6, 2.4 Hz), 7.38-7.56 (4H, m), 7.74-7.78 (2H, m)
FAB-MS (m/e) : 316 [M + H] + FAB-MS (m / e): 316 [M + H] +
工程 1の生成物 (2. 73 g, 8. 66mmo 1 ) をフエニルエーテル (4. 0 g) に懸濁し、 260°Cで 30分加熱した。 室温まで冷却した後、 反応混合物 をシリカゲル上で、 へキサン、 次いでへキサン/酢酸ェチル (80/20) を溶 離剤とするカラムクロマトグラフィーに供し、 淡黄色固体の S— (2—べンゾィ ルー 4ーメトキシフエニル) N, N—ジメチルカルバモチォエート (2. 11 g , 5. 96mmo 1) を得た (収率 77%) 。 The product of Step 1 (2.73 g, 8.66 mmol) was suspended in phenyl ether (4.0 g) and heated at 260 ° C. for 30 minutes. After cooling to room temperature, the reaction mixture was subjected to column chromatography on silica gel using hexane and then hexane / ethyl acetate (80/20) as a dissolving agent to give a light yellow solid, S- (2-benzoic). L-4-methoxyphenyl) N, N-dimethylcarbamothioate (2.11 g, 5.96 mmol 1) was obtained (yield 77%).
^NMR (CDC 13) <5 : 2. 89 (6H, s) , 3. 88 (3H, s) , 6. 96 (1Η, d d, J = 8. 5, 2. 5Hz) , 7. 20 ( 1 H, d, J = 2. 5Hz) , 7. 38- 7. 45 (3H, m) , 7. 51-7. 57 (1H, m) , 7. 76-7. 80 (2H, m) ^ NMR (CDC 1 3) < 5: 2. 89 (6H, s), 3. 88 (3H, s), 6. 96 (1Η, dd, J = 8. 5, 2. 5Hz), 7. 20 (1H, d, J = 2.5Hz), 7.38-7.45 (3H, m), 7.51-7.57 (1H, m), 7.76-7.80 (2H, m )
FAB-MS (m/e) : 316 [M + H] + FAB-MS (m / e): 316 [M + H] +
工程 2の生成物 (3. 14 g, 9. 96mmo 1 ) をメタノール (40. 0m 1 ) に溶解し、 10 ΝτΚ酸化ナトリゥム水溶液 (10. Oml) を加え、 室温で 3時間攪拌した。 反応混合物を 2 N塩酸で処理し、 酢酸ェチルで 2回抽出した。 有機層を飽和食塩水で洗浄し、 乾燥し、 減圧下濃縮した。 残渣を 2, 6—ルチジ ン (6. Oml) に溶解し、 実施例 1の工程 1で得られた 2_イソプロピルアジリ ジン (1. 5ml) を室温で滴下した。 添加終了後、 混合物を室温で 3時間攪拌 し、 濃塩酸 (2. Oml) を添加した。 添加終了後、 混合物を 3時間共沸させ、 減圧留去した。 残渣を飽和炭酸水素ナトリウム水溶液で回収し、 溶液を酢酸ェチ ルで 2回抽出した。 集めた抽出物を飽和食塩水溶液で洗浄し、 乾燥し、 減圧下に 濃縮した。 残渣をシリカゲル上で、 へキサン/酢酸ェチル (90ノ 10) を溶離 剤とするカラムクロマトグラフィーに供し、 淡黄色の油状物質 3—イソプロピル 一 7—メトキシー 5—フエニル一 2, 3—ジヒドロ 1, 4一べンゾチアゼピン ( The product of Step 2 (3.14 g, 9.96 mmol) was dissolved in methanol (40.0 ml), a 10% aqueous sodium oxide solution (10. Oml) was added, and the mixture was stirred at room temperature for 3 hours. The reaction mixture was treated with 2N hydrochloric acid and extracted twice with ethyl acetate. The organic layer was washed with saturated saline, dried, and concentrated under reduced pressure. The residue was dissolved in 2,6-lutidine (6. Oml), and 2_isopropylaziridine (1.5 ml) obtained in step 1 of Example 1 was added dropwise at room temperature. After the addition was completed, the mixture was stirred at room temperature for 3 hours, and concentrated hydrochloric acid (2. Oml) was added. After the addition was completed, the mixture was azeotroped for 3 hours and distilled under reduced pressure. The residue was collected with a saturated aqueous solution of sodium hydrogen carbonate, and the solution was extracted twice with ethyl acetate. The collected extracts were washed with a saturated saline solution, dried, and concentrated under reduced pressure. The residue was subjected to column chromatography on silica gel using hexane / ethyl acetate (90-10) as an eluent to give a light yellow oily substance, 3-isopropyl-17-methoxy-5-phenyl-1,2,3-dihydro-1,3-dihydro-1,3-dihydro-1,3-dihydro-1,3-dihydro-1,3-dihydro-1,4-dihydro-1,3-dihydro-1,4-dihydro-1,3-dihydro-1,2-dihydro-1,4-dihydro-1,2-dihydro-1,3-dihydro-1,2-dihydro-1,3-dihydro-1,2-dihydro-1,2-dihydro-1,3-dihydro-1,3-dihydro-1,5-diethyl. 4 Benzothiazepine (
I. 42 g, 4. 57mmo 1 ) を得た (収率 46 %) 。 iHNMRは、 提示さ れた構造と一致した。 XHNMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 8Hz) , 1. 0 6 (3H, d, J = 6. 7Hz) , 2. 00— 2. 13 (lH, m) , 3. 01 (1H, ddd, J = 12. 2, 6. 5, 3. 9Hz) , 3. 29 ( 1 H, dd , J = 12. 2, 11. 1Hz) , 3. 64 ( 1 H, dd, J = 11. 1, 3. 9Hz) , 3. 86 (3H, s) , 6. 87 ( 1 H, dd, J = 8. 5, 2. 6 Hz) , 7. 04 (1H, d, J = 8. 5Hz) , 7. 17 ( 1 H, d, J = 2 . 6Hz) , 7. 31-7. 43 (3H, m) , 7. 58- 7. 64 (2H, m ) I. 42 g, 4.57 mmo 1) was obtained (46% yield). iHNMR was consistent with the proposed structure. X HNMR (CDC 1 3) ( 5: 0. 95 (3H, d, J = 6. 8Hz), 1. 0 6 (3H, d, J = 6. 7Hz), 2. 00- 2. 13 (lH , M), 3.01 (1H, ddd, J = 12.2, 6.5, 3.9 Hz), 3.29 (1H, dd, J = 12.2, 11.1 Hz), 3.64 (1H, dd, J = 11.1, 3.9Hz), 3.86 (3H, s), 6.87 (1H, dd, J = 8.5, 2.6Hz), 7.04 (1H, d, J = 8.5Hz), 7.17 (1H, d, J = 2.6Hz), 7.31-7.43 (3H, m), 7.58- 7.64 (2H , m)
FAB-MS (m/e) : 312 [M + H] + FAB-MS (m / e): 312 [M + H] +
工程 3の生成物 (1. 42 g, 4. 57mmo 1) のトリフルォロ酢酸 (15 . Oml) 溶液に、 30%過酸化水素水 (2. 5ml ) を約 0°Cの温度で添加し た。 添加終了後、 混合物を室温で一晩攪拌し、 10%チォ硫酸ナトリウム水溶液 、 飽和炭酸水素ナトリウム水溶液で処理し、 酢酸ェチルで 2回抽出し、 抽出物を 集めて飽和食塩水溶液で洗浄し、 乾燥し、 減圧下に濃縮した。 残渣 (1. 63 g のうち 502mg) および 4N塩化水素 1, 4一ジォキサン溶液 ( 14. 0ml ) を混合し、 亜鉛粉末 (0. 81mg, 12. 4mmo 1) を徐々に添加した。 混合液を一晩室温で攪拌し、 飽和炭酸水素ナトリウム水溶液で処理し、 セライト で濾過した。 酢酸ェチルで 2回抽出し、 抽出物を集めて飽和食塩水溶液で洗浄し 、 乾燥し、 減圧下に濃縮した。 残渣をシリカゲル上で、 へキサン/酢酸ェチル ( 80/20) を溶離剤とするカラムクロマトグラフィーに供し、 3—イソプロピ ルー 7—メトキシ一 5—フエ二ルー 2, 3, 4, 5—テトラヒドロー — 1 λ 6, 4—ベンゾチアゼピン— 1, 1—ジオンのジァステレオマー a (82. 0m g、 0. 237mmo 1、 収率 17 %) および b (141. 9mg、 0. 411 mmo 1、 収率 29%) を淡黄色の油状生成物として得た。 To a solution of the product of Step 3 (1.42 g, 4.57 mmol) in trifluoroacetic acid (15.0 Oml) was added 30% aqueous hydrogen peroxide (2.5 ml) at a temperature of about 0 ° C. After the addition was completed, the mixture was stirred at room temperature overnight, treated with a 10% aqueous sodium thiosulfate solution and a saturated aqueous sodium hydrogen carbonate solution, and extracted twice with ethyl acetate. And concentrated under reduced pressure. The residue (502 mg of 1.63 g) and 4N hydrogen chloride 1,4-dioxane solution (14.0 ml) were mixed, and zinc powder (0.81 mg, 12.4 mmo 1) was gradually added. The mixture was stirred overnight at room temperature, treated with saturated aqueous sodium bicarbonate and filtered through celite. The mixture was extracted twice with ethyl acetate, and the extracts were collected, washed with a saturated saline solution, dried, and concentrated under reduced pressure. The residue was subjected to column chromatography on silica gel using hexane / ethyl acetate (80/20) as an eluent to give 3-isopropyl-7-methoxy-1-5-phenyl 2,3,4,5-tetrahydro- 1 λ 6 , 4-Benzothiazepine—a diastereomer of 1,1-dione a (82.0 mg, 0.237 mmo 1, 17% yield) and b (141.9 mg, 0.411 mmo 1, yield 29) %) As a pale yellow oily product.
ジァステレオマー a Diastereomer a
^NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 73— 1. 88 ( 1 H, m) , 3. 08 (1H, dd, J = 11. 4, 14. 3Hz) , 3. 43 ( 1 H, dd, J = 2 . 2, 14. 3Hz) , 3. 51 ( 1 H, ddd, J = 2. 2, 4. 8, 11. 4Hz) , 3. 83 (3H, s) , 5. 80 ( 1 H, s) , 6. 59 (1H, d , J = 8. 6Hz) , 6. 85 (1H, dd, J = 2. 9, 8. 6Hz) , 7. 33 - 7. 45 (5H, m) , 7. 68 (1H, d, J = 2. 9Hz) ^ NMR (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 73- 1. 88 (1 H , m), 3.08 (1H, dd, J = 11.4, 14.3Hz), 3.43 (1H, dd, J = 2.2, 14.3Hz), 3.51 (1H, ddd, J = 2.2, 4.8, 11. 4Hz), 3.83 (3H, s), 5.80 (1H, s), 6.59 (1H, d, J = 8.6Hz), 6.85 (1H, dd, J = 2.9) , 8.6Hz), 7.33-7.45 (5H, m), 7.68 (1H, d, J = 2.9Hz)
FAB-MS (m/e) : 346 [M + H] + FAB-MS (m / e): 346 [M + H] +
ジァステレオマー b Geostereomer b
XHNMR (CDC 13) 6 : 0 95 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 88-2. 02 (1H, m) , 2. 97 (1H, d d d, J = 3. 5, 5. 5, 9. 1Hz) , 3. 43 ( 1 H, dd, J = 9. 1, 14. 5Hz) , 3. 49 (1H, d d, J = 3. 5, 14. 5H zz)) ,, 33.. 8866 ((33HH,, ss)) ,, 55.. 78 ( 1 H, s) , 6. 80 ( 1 H, d, J =8. 6Hz) , 6. 96 (1 H, dd, J = 2. 8, 8. 6Hz) , 7. 26 -7. 42 (5H, m) , 7. 66 ( 1 H, d, J = 2. 8Hz) X HNMR (CDC 1 3) 6 :. 0 95 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 88-2 02 (1H, m ), 2.97 (1H, ddd, J = 3.5, 5.5, 9.1 Hz), 3.43 (1H, dd, J = 9.1, 14.5 Hz), 3.49 (1H , Dd, J = 3.5, 14.5Hzz)), 33 .. 8866 ((33HH ,, ss)), 55..78 (1H, s), 6.80 (1H, d , J = 8.6 Hz), 6.96 (1 H, dd, J = 2.8, 8.6 Hz), 7.26 -7. 42 (5H, m), 7.66 (1 H, d, J = 2.8Hz)
FAB-MS (m/e) : 346 [M + H] + 以下に実施例 72と同様にして得た化合物の物理定数を示す。 FAB-MS (m / e): 346 [M + H] + The physical constants of the compound obtained in the same manner as in Example 72 are shown below.
実施例 73 Example 73
化合物例 1160の製造 (Ar : e-MeO-Ph j R^ H ^^ H ^3 : i - P r ; R4: H; R5: Ph; n=2) Preparation of compound examples 1160 (Ar: e-MeO- Ph j R ^ H ^^ H ^ 3: i - P r; R 4: H; R 5: Ph; n = 2)
^NMR (CDC 13) δ : 0. 99 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 9Hz) , 1. 80— 1. 98 ( 1 H, m) , 3. 10 —3. 20 ( 1 H, m) , 3. 20- 3. 38 (2H, m) , 3. 74 (3H, s) , 6. 35 (1H, s) , 7. 11-7. 16 (2H, m) , 7. 20-7 . 32 (4H, m) , 7. 49 ( 1 H, t, J = 8. 0Hz) , 7. 88 (1H , dd, J = 1. 1, 8. 0Hz) ^ NMR (CDC 1 3) δ : 0. 99 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 9Hz), 1. 80- 1. 98 (1 H , m), 3.10 — 3.20 (1 H, m), 3.20-3.38 (2H, m), 3.74 (3H, s), 6.35 (1H, s), 7 11-7. 16 (2H, m), 7.20-7. 32 (4H, m), 7.49 (1H, t, J = 8.0Hz), 7.88 (1H, dd, J = 1.1, 8.0Hz)
FAB-MS (m/e) : 346 [M + H] + 実施例 74 FAB-MS (m / e): 346 [M + H] + Example 74
化合物例 1163の製造 Preparation of Compound Example 1163
ジァステレオマー a (Ar : 9一 OMe— Ph; R1: H; R2: H; R3 P r ; R4 : H ; R5 : Ph ; n=2) Diastereomer a (Ar: 9 OMe—Ph; R 1 : H; R 2 : H; R 3 P r; R 4 : H; R 5 : Ph; n = 2)
XHNMR (CDC 13) δ : 0. 97 (3H, d, J = 7. 0Hz) , 0. 9 7 (3H, d, J = 7. 0Hz) , 1. 70— 1. 85 (IH, m) , 3. 25 一 3. 50 (3H, m) , 3. 93 (3H, s) , 6. 01 (1H, s) , 6. 32 (IH, d, J = 7. 9Hz) , 6. 92 (1H, d, J = 8. 1Hz) , 7. 22- 7. 40 (6H, m) X HNMR (CDC 1 3) δ : 0. 97 (3H, d, J = 7. 0Hz), 0. 9 7 (3H, d, J = 7. 0Hz), 1. 70- 1. 85 (IH, m), 3.25-3.50 (3H, m), 3.93 (3H, s), 6.01 (1H, s), 6.32 (IH, d, J = 7.9 Hz), 6 . 92 (1H, d, J = 8.1Hz), 7.22-7.40 (6H, m)
FAB-MS (m/e) : 346 [M + H] + FAB-MS (m / e): 346 [M + H] +
ジァステレオマー b (Ar : e-OMe-Ph j R^ H j R^ H j R3 : i一 P r ; R4 : H ; R5 : Ph ; n=2) Diastereomer b (Ar: e-OMe-Ph j R ^ H j R ^ H j R 3 : i-Pr; R 4 : H; R 5 : Ph; n = 2)
^NMR (CDC 13) (5 : 0. 91 (3H, d, J = 6. 5Hz) , 0. 9 6 (3H, d, J = 6. 5Hz) , 1. 85— 1. 98 ( 1 H, m) , 3. 08 - 3. 1 5 (IH, m) , 3. 60 ( 1 H, dd, J = 3. 7, 14. 8Hz) , 3. 74 (IH, dd, J = 10. 1, 14. 8Hz) , 3. 99 (3H, s ) , 5. 41 (1H, s) , 6. 27 ( 1 H, d, J = 7. 9Hz) , 6. 96 (1H, d, J = 8. 2Hz) , 7. 18— 7. 48 (6H, m) ^ NMR (CDC 1 3) ( 5: 0. 91 (3H, d, J = 6. 5Hz), 0. 9 6 (3H, d, J = 6. 5Hz), 1. 85- 1. 98 (1 H, m), 3.08-3.15 (IH, m), 3.60 (1H, dd, J = 3.7, 14.8 Hz), 3.74 (IH, dd, J = 10 1, 14.8 Hz), 3.99 (3H, s), 5.41 (1H, s), 6.27 (1H, d, J = 7.9 Hz), 6.96 (1H, d, J = 8.2 Hz), 7.18-7.48 (6H, m)
FAB-MS (m/e) : 346 [M + H] + 実施例 Ί 5 FAB-MS (m / e): 346 [M + H] + Example Ί 5
化合物例 1 165の製造 Preparation of Compound Example 1 165
ジァステレオマー a (Ar : 8-OH-Ph ; RX : H ; R2 : H ; R3 : i _P r ; R4 : H ; R5 : Ph ; n=2) Jiasutereoma a (Ar: 8-OH- Ph; R X: H; R 2: H; R 3: i _P r; R 4: H; R 5: Ph; n = 2)
^NMR (CDC 13) δ : 0. 98 ( 3 H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 73 - 1. 88 (lH, m) , 3. 1 1 (IH, dd, J =l 1. 3, 14. 3Hz) , 3. 43 ( 1 H, d d, J = 2 . 2, 14. 3Hz) , 3. .51 ( 1 H, d d d, 1 = 2. 2, 4. 7, 1 1. 3Hz) , 5. 77 (1H, s) , 6. 55 ( 1 H, d, J = 8. 4Hz) , 6 . 81 (IH, dd, J = 2. 7, 8. 4Hz) , 7. 30-7. 45 (5H, m) , 7. 66 (IH, d, J = 2. 7Hz) ^ NMR (CDC 1 3) δ : 0. 98 (3 H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 73 - 1. 88 (lH , M), 3.11 (IH, dd, J = 1.3, 14.3 Hz), 3.43 (1H, dd, J = 2.2, 14.3 Hz), 3.5.51 ( 1 H, ddd, 1 = 2.2, 4. 7, 1 1.3 Hz), 5.77 (1H, s), 6.55 (1 H, d, J = 8.4 Hz), 6.81 ( IH, dd, J = 2.7, 8.4Hz), 7.30-7.45 (5H, m), 7.66 (IH, d, J = 2.7Hz)
FAB-MS (m/e) : 332 [M + H] + ジァステレオマー b (A r : 8— OH— Ph ; R 1 : H ; R 2 : H ; R 3 一 P r ; R4 : H ; R5 : Ph ; n = 2)FAB-MS (m / e): 332 [M + H] + Jiasutereoma b (A r: 8- OH- Ph ; R 1: H; R 2: H; R 3 one P r; R 4: H; R 5: Ph; n = 2)
NMR (CDC 13) δ : 0 95 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 88-2. 03 (1H, m) , 3. 00 (1H, ddd, J = 3. 6, 5. 7, 9. 0Hz) , 3. 45 (1H, d d, J = 9. 0, 14. 3Hz) , 3. 52 (1H, d d, J = 3. 6, 14. 3H z) , 5. 75 (1H, s) , 6. 77 (1H, d, J = 8. 4Hz) , 6. 9 2 (1H, dd, J = 2. 7, 8. 4Hz) , 7. 28-7. 40 (5H, m) , 7. 66 (1H, d, J = 2. 7Hz) NMR (CDC 1 3) δ: . 0 95 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 88-2 03 (1H, m) , 3.00 (1H, ddd, J = 3.6, 5.7, 9.0Hz), 3.45 (1H, dd, J = 9.0, 14.3Hz), 3.52 (1H, dd , J = 3.6, 14.3Hz), 5.75 (1H, s), 6.77 (1H, d, J = 8.4Hz), 6.92 (1H, dd, J = 2. 7, 8.4 Hz), 7.28-7.40 (5H, m), 7.66 (1H, d, J = 2.7Hz)
FAB-MS (m/e) : 332 [M + H] + 実施例 76 FAB-MS (m / e): 332 [M + H] + Example 76
化合物例 1166の製造 Preparation of Compound Example 1166
ジァステレオマー a (Ar : g-OH-P j R^ H j R^ H j R3 : i一 P r ; R4: H; R5 : Ph ; n = 2) Jiasutereoma a (Ar: g-OH- P j R ^ H j R ^ H j R 3: i one P r; R 4: H; R 5: Ph; n = 2)
iHNMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 6Hz) , 0. 9 8 (3H, d, J = 6. 6Hz) , 1. 75 - 1. 85 (1H, m) , 3. 32 (1H, d d, J = 11. 5, 14. 1Hz) , 4. 45 ( 1 H, dd, J = 2 . 1, 14. 1Hz) 3. 45— 3. 55 (1H, m) , 5. 89 (1H, s) , 6. 14 (1H, d, J = 7. 6Hz) , 6. 86 ( 1 H, d, J = 8. 6H z) , 7. 18 (1H, d d, 1 = 7. 6, 8. 6Hz) , 7. 30-7. 41 (5H, m) , 10. 36 ( 1 H, s) iHNMR (CDC 1 3) δ: 0. 98 (3H, d, J = 6. 6Hz), 0. 9 8 (3H, d, J = 6. 6Hz), 1. 75 - 1. 85 (1H, m ), 3.32 (1H, dd, J = 11.5, 14.1Hz), 4.45 (1H, dd, J = 2.1, 14.1Hz) 3.45—3.55 (1H, m), 5.89 (1H, s), 6.14 (1H, d, J = 7.6Hz), 6.86 (1H, d, J = 8.6Hz), 7.18 (1H, dd, 1 = 7.6, 8.6Hz), 7.30-7.41 (5H, m), 10.36 (1H, s)
FAB-MS (m/e) : 332 [M + H] + FAB-MS (m / e): 332 [M + H] +
ジァステレオマー b (Ar : 9 -OH-P ; R1 : H ; R2 : H ; R3 : i— P r ; R4 : H ; R5 : Ph ; n=2) Jiasutereoma b (Ar: 9 -OH-P ; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: Ph; n = 2)
iHNMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 9Hz) , 1. 0 0 (3H, d, J = 6. 9Hz) , 1. 95- 2. 05 ( 1 H, m) , 2. 95 一 3. 05 (lH, m) , 3. 49— 3. 65 (2H, m) , 5. 90 (1H, s) , 6. 31 (1H, d, J = 7. 9Hz) , 6. 94 ( 1 H, d, J = 8. 4Hz) , 7. 25 - 7. 45 (6H, m) , 10. 0 ( 1 H, s) iHNMR (CDC 1 3) δ: 0. 98 (3H, d, J = 6. 9Hz), 1. 0 0 (3H, d, J = 6. 9Hz), 1. 95- 2. 05 (1 H, m), 2.95-3.05 (lH, m), 3.49-3.65 (2H, m), 5.90 (1H, s), 6.31 (1H, d, J = 7. 9 Hz), 6.94 (1H, d, J = 8. 4Hz), 7.25-7.45 (6H, m), 10.0 (1H, s)
FAB-MS (m/e) : 332 [M + H] + 実施例 77 FAB-MS (m / e): 332 [M + H] + Example 77
化合物例 1167の製造 Preparation of Compound Example 1167
ジァステレオマー a (A r : 8— n— P rNHC〇CH2〇一 Ph ; R1 : H; R 2: H; R3: i -P r ; R4: H; R5: Ph ; n=2) Jiasutereoma a (A r: 8- n- P RNHC_〇_CH 2 〇 one Ph; R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: Ph; n = 2 )
^NMR (CDC 13) δ : 0. 94 ( 3 H, t, J = 7. 6Hz) 0. 98 (3H, d, J = 7. 2Hz) , 0. 98 (3H, d, J = 7. 2Hz) , 1. 52- 1. 65 (2H, m) , 1. 75 - 1. 85 (lH, m) , 3. 08 ( 1 H, dd, J =l 1. 4, 14. 3Hz) , 3. 29 ( 1 H, t, J = 6. 3H z) , 3. 38 (1H, t, J = 6. 3Hz) , 3. 44 ( 1 H, d d, J = 2 . 1, 14. 3Hz) , 3. 52 ( 1 H, d d d, J = 2. 1, 4. 8. 11. 4) , 4. 48 (2H, s) , 5. 79 ( 1 H, s) , 6. 52 (1H, b r s ) , 6. 64 (1H, d, J = 8. 6Hz) , 6. 85 (1H, dd, J = 2. 8, 8. 6Hz) , 7. 32-7. 45 (5H, m) , 7. 76 (1H, d, J =2. 8Hz) ^ NMR (CDC 1 3) δ : 0. 94 (3 H, t, J = 7. 6Hz) 0. 98 (3H, d, J = 7. 2Hz), 0. 98 (3H, d, J = 7 2 Hz), 1.52-1.65 (2H, m), 1.75-1.85 (lH, m), 3.08 (1 H, dd, J = l 1.4, 14.3 Hz) , 3.29 (1H, t, J = 6.3Hz), 3.38 (1H, t, J = 6.3Hz), 3.44 (1H, dd, J = 2.1, 14. 3Hz), 3.52 (1H, ddd, J = 2.1, 4.8.11.4), 4.48 (2H, s), 5.79 (1H, s), 6.52 ( 1H, brs), 6.64 (1H, d, J = 8.6Hz), 6.85 (1H, dd, J = 2.8, 8.6Hz), 7.32-7.45 (5H, m ), 7.76 (1H, d, J = 2.8Hz)
FAB-MS (m/e) : 31 [M + H] + FAB-MS (m / e): 31 [M + H] +
ジァステレオマー b (A r : 8-n-P rNHCOCH20-Ph ; R1: H ; R 2 : H ; R3 : i -P r ; R4 : H ; R5 : Ph ; n=2) Jiasutereoma b (A r: 8-nP rNHCOCH 2 0-Ph; R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) δ : 0. 94 (3H, t, 1 =7. 4Hz) , 0. 9 5 (3H, d, J = 6. 7Hz) , 0. 99 (3H, d, J = 6. 7Hz) , 1 . 53 - 1. 68 (2H, m) , 1. 92 -2. 02 (lH, m) , 2. 97 ( 1H, ddd, J = 3. 5, 5. 8, 12. 1Hz) , 3. 3 1 (1H, t, J =6. 8Hz) , 3. 34 ( 1 H, t, J = 6. 8Hz) , 3. 45 ( 1 H, d d, J = 12. 1, 14. 4Hz) , 3. 50 ( 1 H, dd, J = 3. 5, 14 . 4Hz) , 4. 52 (2H, s) , 5. 78 ( 1 H, s) , 6. 54 ( 1 H, b r s) , 6. 85 ( 1 H, d, J = 8. 6Hz) , 6. 97 ( 1 H, d d, J =2. 8, 8. 6Hz) , 7. 29-7. 42 (5H, m) , 7. 74 (1H, d, J = 2. 8Hz) X HNMR (CDC 1 3) δ : 0. 94 (. 3H, t, 1 = 7 4Hz), 0. 9 5 (3H, d, J = 6. 7Hz), 0. 99 (3H, d, J = 6.7 Hz), 1.53-1.68 (2H, m), 1.92-2.02 (lH, m), 2.97 (1H, ddd, J = 3.5, 5. 8, 12 1Hz), 3.31 (1H, t, J = 6.8Hz), 3.34 (1H, t, J = 6.8Hz), 3.45 (1H, dd, J = 12.1) , 14.4Hz), 3.50 (1H, dd, J = 3.5, 14.4Hz), 4.52 (2H, s), 5.78 (1H, s), 6.54 (1 H, brs), 6.85 (1H, d, J = 8.6Hz), 6.97 (1H, dd, J = 2.8, 8.6Hz), 7.29-7.42 (5H , M), 7.74 (1H, d, J = 2.8Hz)
FAB-MS (m/e) : 43 [M + H] + 実施例 78 FAB-MS (m / e): 43 [M + H] + Example 78
化合物例 1 168の製造 Preparation of Compound Example 1 168
ジァステレオマ一 a (A r : 9-Me-Ph ; R^- H J R^ H I R3 i一 P r ; R4: H; R5 : Ph ; n=2) Diastereomer a (A r: 9-Me-Ph; R ^-HJR ^ HIR 3 i-Pr; R 4 : H; R 5 : Ph; n = 2)
XHNM (CDC 13) δ : 0. 98 (3H, d, J = 6. 6Hz) , 0. 9 8 (3H, d, J = 6. 6Hz) , 1. 72- 1. 85 ( 1 H, m) , 2. 79 (3H, s) , 3. 20 (1H, dd, J = 11. 3, 14. 5Hz) , 3. 3 8 (1H, dd, J = 2. 1, 14. 5Hz) 3. 47— 3. 52 (1H, m) , 6. 13 (1H, s) , 6. 60 ( 1 H, d, J = 7. 2Hz) , 7. 1 1 - 7. 20 (2H, m) , 7. 25-7. 45 (5H, m) X HNM (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 6Hz), 0. 9 8 (3H, d, J = 6. 6Hz), 1. 72- 1. 85 (1 H , M), 2.79 (3H, s), 3.20 (1H, dd, J = 11.3, 14.5Hz), 3.38 (1H, dd, J = 2.1, 14.5Hz) ) 3.47—3.52 (1H, m), 6.13 (1H, s), 6.60 (1H, d, J = 7.2Hz), 7.11-7.20 (2H, m), 7.25-7.45 (5H, m)
FAB-MS (m/e) : 330 [M + H] + FAB-MS (m / e): 330 [M + H] +
ジァステレオマー b (Ar : 9-Me-Ph ; R1 : H ; R2 : H ; R3 : i一 P r ; R4 : H ; R5 : Ph ; n = 2) Jiasutereoma b (Ar: 9-Me- Ph; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) 6 : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 87 - 1. 99 ( 1 H, m) , 2. 79 (3H, s) , 2. 95-3. 04 (lH, m) , 3. 60— 3. 68 (2H, m) , 5. 64 ( 1 H, s) , 6. 56 - 6. 62 ( 1 H, m) , 7. 16— 7 . 22 (2H, m) , 7. 32— 7. 41 (5H, m) X HNMR (CDC 1 3) 6 : 0. 94 (3H, d, J = 6. 7Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 87 - 1. 99 (1 H , m), 2.79 (3H, s), 2.95-3.04 (lH, m), 3.60—3.68 (2H, m), 5.64 (1H, s), 6 . 56-6.62 (1H, m), 7.16—7.22 (2H, m), 7.32—7.41 (5H, m)
FAB-MS (m/e) : 330 [M + H] + 実施例 79 FAB-MS (m / e): 330 [M + H] + Example 79
化合物例 1169の製造 Preparation of Compound Example 1169
ジァステレオマー a (Ar : T-C l-Ph j R^ H j R^ H j R3 : i—P r ; R4: H; R5: Ph; n=2) Jiasutereoma a (Ar: TC l-Ph j R ^ H j R ^ H j R 3: i-P r; R 4: H; R 5: Ph; n = 2)
^NMR (CDC 13) δ : 0. 99 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 9 Hz) , 1. 73 - 1. 89 ( 1 H, m) , 3. 06 (1H, d d, J =l 1. 4, 14. 3Hz) , 3. 44 (1 H, dd, J = 2 . 2, 14. 3Hz) , 3. 47-3. 55 ( 1 H, m) , 5. 85 (IH, s ) , 6. 67 (IH, d, J = 2. 1Hz) , 7. 38 ( 1 H, dd, J = 2. 1, 8. 4Hz) , 7. 35 - 7. 50 (5H, m) , 8. 09 (IH, d, J =8. 4Hz) ^ NMR (CDC 1 3) δ : 0. 99 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 9 Hz), 1. 73 - 1. 89 (1 H, m), 3.06 (1H, dd, J = l 1.4, 14.3Hz), 3.44 (1H, dd, J = 2.2, 14.3Hz), 3.47-3.55 (1H, m) , 5.85 (IH, s), 6.67 (IH, d, J = 2.1 Hz), 7.38 (1H, dd, J = 2.1, 8.4 Hz), 7.35-7 . 50 (5H, m), 8.09 (IH, d, J = 8.4 Hz)
FAB-MS (m/e) : 350/352 [M + H] + FAB-MS (m / e): 350/352 [M + H] +
ジァステレオマー b (Ar : T-C l -Ph j R^ H j ^ H j R3 : i一 P r ; R4 : H ; R5 : Ph ; n = 2) Jiasutereoma b (Ar: TC l -Ph j R ^ H j ^ H j R 3: i one P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 90 - 2. 03 ( 1 H, m) , 2. 97 X HNMR (CDC 1 3) δ : 0. 95 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 90 - 2. 03 (1 H , m), 2.97
(1H, d d d, J = 3. 7, 5. 7, 8. 9Hz) , 3. 45 (1H, dd, J = 8. 9, 14. 4Hz) , 3. 49 (1H, dd, J = 3. 7, 14. 4H z) , 5. 81 (IH, s) , 6. 88 (1H, d, J = 2. 0Hz) , 7. 3 0— 7. 43 (5H, m) , 7. 45 ( 1 H, d d, J = 2. 0, 8. 4Hz) , 8. 09 (1H, d, J = 8. 4Hz) (1H, ddd, J = 3.7, 5.7, 8.9Hz), 3.45 (1H, dd, J = 8.9, 14.4Hz), 3.49 (1H, dd, J = 3 7, 14.4 Hz), 5.81 (IH, s), 6.88 (1 H, d, J = 2.0 Hz), 7.30—7.43 (5H, m), 7.45 (1H, dd, J = 2.0, 8.4Hz), 8.09 (1H, d, J = 8.4Hz)
FAB-MS (m/e) : 350/352 [M + H] + 実施例 80 FAB-MS (m / e): 350/352 [M + H] + Example 80
化合物例 1 172の製造 Preparation of Compound Example 1 172
ジァステレオマ一 a (Ar : 8— C 1一 Ph R H R^ H R3 : i— P r ; R4: H; R5: Ph; n = 2) Diastereomer a (Ar: 8-C 1 Ph RHR ^ HR 3 : i-P r; R 4 : H; R 5 : Ph; n = 2)
^NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 77— 1. 89 (IH, m) , 3. 08 (IH, dd, J = 1 1. 4, 14. 4Hz) , 3. 44 ( 1 H, dd, J = 2 . 2, 14. 4Hz) , 3. 51 (1H, ddd, J = 2. 2, 4. 8, 1 1. 4Hz) , 5. 82 (IH, s) , 6. 63 (1H, d, J = 8. 3Hz) , 7 . 31 (IH, dd, J = 2. 3, 8. 3Hz) , 7. 32-7. 46 (5H, m) , 8. 13 (IH, d, J = 2. 3Hz) ^ NMR (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 77- 1. 89 (IH, m), 3.08 (IH, dd, J = 1 1.4, 14.4 Hz), 3.44 (1H, dd, J = 2.2, 14.4 Hz), 3.51 (1H, ddd , J = 2.2, 4. 8, 11.4 Hz), 5.82 (IH, s), 6.63 (1H, d, J = 8.3 Hz), 7.31 (IH, dd, J = 2.3, 8.3 Hz), 7.32-7.46 (5H, m), 8.13 (IH, d, J = 2.3 Hz)
FAB-MS ( /e) : 350/352 [M + H] + ジァステレオマー b (Ar : S-C l -Ph j R^ H j R^ H j R3 : i一 P r ; R4 : H ; R5 : Ph ; n = 2) FAB-MS (/ e): 350/352 [M + H] + Diastereomer b (Ar: SC l -Ph j R ^ H j R ^ H j R 3 : i- 1 Pr; R 4 : H; R 5 : Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 92— 2. 03 (1H, m) , 2. 96 (1H, ddd, J = 3. 5, 5. 9, 8. 9Hz) , 3. 46 ( 1 H, dd, J = 8. 9, 14. 4Hz) , 3. 51 ( 1 H, d d, J = 3. 5, 14. 4H z) , 5. 79 (1H, s) , 6. 84 (1H, d, J = 8. 3Hz) , 7. 3 0-7. 42 (5H, m) , 7. 42 ( 1 H, dd, 1 = 2. 2, 8. 3Hz) , 8. 13 (1H, d, J = 2. 2Hz) X HNMR (CDC 1 3) ( 5: 0. 95 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 92- 2. 03 (1H , m), 2.96 (1H, ddd, J = 3.5, 5.9, 8.9Hz), 3.46 (1H, dd, J = 8.9, 14.4Hz), 3.51 (1H, dd, J = 3.5, 14.4Hz), 5.79 (1H, s), 6.84 (1H, d, J = 8.3Hz), 7.30-7.42 (5H, m), 7.42 (1H, dd, 1 = 2.2, 8.3Hz), 8.13 (1H, d, J = 2.2Hz)
FAB-MS (m/e) : 350/352 [M + H] + 実施例 81 FAB-MS (m / e): 350/352 [M + H] + Example 81
化合物例 1 175の製造 Preparation of Compound Example 1 175
ジァステレオマー a (Ar : 9-C 1 -Ph ; R1: H; R2: H; R3: i一 P r ; R4: H; R5: Ph ; n = 2) Jiasutereoma a (Ar: 9-C 1 -Ph; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) δ : 0. 99 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 9Hz) , 1. 75— 1. 89 ( 1 H, m) , 3. 29 (1H, d d, J =l 1. 2, 14. 6Hz) , 3. 45 ( 1 H, dd, J = 2 . 3, 14. 6Hz) , 3. 50 ( 1 H, ddd, J = 2. 3, 4. 9, 1 1. 2Hz) , 6. 1 1 (1H, s) , 6. 69 ( 1 H, dd, J = l. 2, 7. 9 Hz) , 7. 20 (1H, t, J = 7. 9Hz) , 7. 31— 7. 45 (6H, m) X HNMR (CDC 1 3) δ : 0. 99 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 9Hz), 1. 75- 1. 89 (1 H , m), 3.29 (1H, dd, J = l 1.2, 14.6Hz), 3.45 (1H, dd, J = 2.3, 14.6Hz), 3.50 (1H , Ddd, J = 2.3, 4.9, 11.2Hz), 6.11 (1H, s), 6.69 (1H, dd, J = l.2, 7.9Hz), 7.20 (1H, t, J = 7.9Hz), 7.31—7.45 (6H, m)
FAB-MS (m/e) : 350/352 [M + H] + FAB-MS (m / e): 350/352 [M + H] +
ジァステレオマー b (Ar : 9 -C 1 -Ph ; 1 : H ; R2 : H ; R3 : i一 P r ; R4 : H ; 5 : P h ; n= 2) Jiasutereoma b (Ar: 9 -C 1 -Ph ; 1: H; R 2: H; R 3: i one P r; R 4: H; 5: P h; n = 2)
'HNMR (CDC 13) δ : 0. 93 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 88— 2. 02 ( 1 H, m) , 3. 07 (1H, ddd, J = 4. 5, 7. 1, 9. 5Hz) , 3. 67 - 3. 80 (2 H, m) , 5. 49 (1H, s) , 6. 65 ( 1 H, dd, J = 0. 7, 7. 9 Hz) , 7. 19 (1H, t, J = 7. 9Hz) , 7. 26- 7. 45 (6H, m) 'HNMR (CDC 1 3) δ : 0. 93 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 88- 2. 02 (1 H , m), 3.07 (1H, ddd, J = 4.5, 7.1, 9.5 Hz), 3.67-3.80 (2 H, m), 5.49 (1H, s), 6.65 (1 H, dd, J = 0.7, 7. 9 Hz), 7.19 (1H, t, J = 7.9 Hz), 7.26-7.45 (6H, m)
FAB-MS (m/e) : 350Z352 [M + H] + 実施例 82 FAB-MS (m / e): 350Z352 [M + H] + Example 82
化合物例 1178の製造 Preparation of Compound Example 1178
ジァステレオマー a (Ar : 9— C 1一 Ph ; R1: H; R2: H; R3 i一 P r ; R4: H; R5 : 4-C l -Ph ; n=2)Jiasutereoma a (Ar: 9- C 1 one Ph; R 1: H; R 2: H; R 3 i one P r; R 4: H; R 5: 4-C l -Ph; n = 2)
NMR (CDC 13) (5 : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 71— 1. 82 ( 1 H, m) , 3. 26 (1H, dd, J = 11. 5, 14. 8Hz) , 3. 40- 3. 50 (2H, m ) , 6. 13 (1H, s) , 6. 66 (1H, d, J = 7. 9Hz) , 7. 19 -7. 40 (6H, m) NMR (CDC 1 3) (5 : 0. 98 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 71- 1. 82 (1 H , m), 3.26 (1H, dd, J = 11.5, 14.8Hz), 3.40-3.50 (2H, m), 6.13 (1H, s), 6.66 (1H , d, J = 7.9 Hz), 7.19 -7. 40 (6H, m)
FAB-MS (m/e) : 384X386 [M + H] + FAB-MS (m / e): 384X386 [M + H] +
ジァステレオマー b (Ar : 9— C I— Ph '- R H R2 : !! ; !^ : i— P r ; R4 : H ; R5: 4-C 1 -Ph ; n=2) Jiasutereoma b (Ar: 9- CI- Ph ' - RHR 2:! !!; ^: i- P r; R 4: H; R 5: 4-C 1 -Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 92 (3H, d, J = 6. 6Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 80— 1. 95 ( 1 H, m) , 2. 95 一 3. 10 (1H, m) , 3. 60— 3. 80 (2H, m) , 5. 50 (1 H, s) , 6. 62 (1H, d, J = 7. 9Hz) , 7. 15-7. 45 (6H, m ) X HNMR (CDC 1 3) ( 5: 0. 92 (3H, d, J = 6. 6Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 80- 1. 95 (1 H, m), 2.95-3.10 (1H, m), 3.60-3.80 (2H, m), 5.50 (1 H, s), 6.62 (1H, d, J = 7.9Hz), 7.15-7.45 (6H, m)
FAB-MS (m/e) : 384/386 [M + H] + 実施例 83 FAB-MS (m / e): 384/386 [M + H] + Example 83
化合物例 1180の製造 Preparation of Compound Example 1180
ジァステレオマー a (Ar : T-C l -Ph i R^ H j R^ H j R3 : i一 P r ; R4 : H ; R5 : 4-MeO-Ph ; n=2) Jiasutereoma a (Ar: TC l -Ph i R ^ H j R ^ H j R 3: i one P r; R 4: H; R 5: 4-MeO-Ph; n = 2)
XHNM (CDC 13) δ : 0. 98 (3H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 74- 1. 88 (1H, m) , 3. 29 (IH, dd, J =l 1. 2, 14. 8Hz) , 3. 44 (IH, dd, J = 2 . 4, 14. 8Hz) , 3. 49 ( 1 H, ddd, J = 2. 4, 5. 2, 1 1. 2Hz) , 3. 84 (3H, s) , 6. 01 (IH, s) , 6. 73 ( 1 H, d d, J = l. 3, 7. 9Hz) , 6. 94 (2H, d, J = 8. OHz) , 7. 20 (1H, t, J = 7. 9Hz) , 7. 27 (IH, d, J = 8. OHz) , 7. 37 (IH, d d, J = l. 3, 7. 9Hz) X HNM (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 74- 1. 88 (1H, m), 3.29 (IH, dd, J = 1.2, 14.8 Hz), 3.44 (IH, dd, J = 2.4, 14.8 Hz), 3.49 (1 H, ddd, J = 2.4 , 5.2, 11.2Hz), 3.84 (3H, s), 6.01 (IH, s), 6.73 (1H, dd, J = l. 3, 7.9Hz), 6 .94 (2H, d, J = 8. OHz), 7.20 (1H, t, J = 7.9 Hz), 7.27 (IH, d, J = 8. OHz), 7.37 (IH, (dd, J = l. 3, 7.9 Hz)
FAB-MS (m/e) : 380/382 [M + H] + FAB-MS (m / e): 380/382 [M + H] +
ジァステレオマー b ( A r : 9 - C 1 - P h ; R 1 : H ; R 2 : H ; R 3 : i— P r ; R4: H; R5: 4-MeO-Ph; n=2) Jiasutereoma b (A r: 9 - C 1 - P h; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 4-MeO-Ph; n = 2)
iHNMR (CDC 13) (5 : 0. 93 (3H, d, J = 6. 7Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 88-2. 01 (IH, m) , 3. 07 iHNMR (CDC 1 3) (5 :. 0. 93 (3H, d, J = 6. 7Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 88-2 01 (IH, m), 3.07
(IH, ddd, J =4. 6, 7. 0, 9. 1Hz) , 3. 65-3. 84 (2 H, m) , 3. 84 (3H, s) , 5. 45 (IH, s) , 6. 69 (1H, d , 1 =7. 9Hz) , 6. 93 (2H, d, J = 8. 8Hz) , 7. 19 (IH , t, J = 7. 9Hz) , 7. 22 (2H, d, J = 8. 8Hz) , 7. 41 ( IH, d, J = 7. 9Hz) (IH, ddd, J = 4.6, 7.0, 9.1 Hz), 3.65-3.84 (2 H, m), 3.84 (3H, s), 5.45 (IH, s ), 6.69 (1H, d, 1 = 7.9 Hz), 6.93 (2H, d, J = 8.8 Hz), 7.19 (IH, t, J = 7.9 Hz), 7.22 (2H, d, J = 8.8Hz), 7.41 (IH, d, J = 7.9Hz)
FAB-MS (m/e) : 380/382 [M + H] + 実施例 84 FAB-MS (m / e): 380/382 [M + H] + Example 84
化合物例 1 181の製造 (A r : 9 - C 1 - P h; R 1: H; R2: H; R 3 : i -P r ; R4 : H ; R5 : 4-OH-Ph ; n=2) Preparation of compound examples 1 181 (A r: 9 - C 1 - P h; R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: 4-OH-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 92 (3H, d, J = 6. 7Hz) , 0. 9 7 (3H, d, J = 6. 7Hz) , 1. 87— 1. 99 (1H, m) , 3. 00 一 3. 10 (IH, m) , 3. 64— 3. 77 (2H, m) , 5. 43 (1H, s) , 6. 67 - 6. 70 (IH, m) , 6. 84- 6. 89 (2H, m) , 7 . 14-7. 23 (3H, m) , 7. 38- 7. 43 (IH, m) X HNMR (CDC 1 3) δ : 0. 92 (3H, d, J = 6. 7Hz), 0. 9 7 (3H, d, J = 6. 7Hz), 1. 87- 1. 99 (1H, m), 3.00-3.10 (IH, m), 3.64-3.77 (2H, m), 5.43 (1H, s), 6.67-6.70 (IH, m) , 6.84-6.89 (2H, m), 7.14-7.23 (3H, m), 7.38-7.43 (IH, m)
FAB-MS (m/e) : 366/368 [M + H] + 実施例 85 化合物例 1199の製造 FAB-MS (m / e): 366/368 [M + H] + Example 85 Preparation of Compound Example 1199
ジァステレオマー a (A r : 9 -C 1 -P h ; R1 : H; R2 : H; R3 : i—P r ; R4 : H ; R5 : 3-C l -4-Me-Ph ; n = 2) Jiasutereoma a (A r: 9 -C 1 -P h; R 1: H; R 2: H; R 3: i-P r; R 4: H; R 5: 3-C l -4-Me-Ph ; n = 2)
HNMR (CDC 13) δ : 0. 98 (3H, d, 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 74- 1. 85 (1 H, m) , 2. 39 (3H, s) , 3. 40 - 3. 50 (2H, m) , 6. 09 ( 1 H, s) , 6. 7 0 (1H, d, J = 7. 7Hz) , 7. 10 ( 1 H, d, J = 7. 9Hz) , 7. 20 - 7. 27 (2H, m) , 7. 36-7. 41 (2H, m) HNMR (CDC 1 3) δ: 0. 98 (3H, d, 6. 8Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 74- 1. 85 (1 H, m) , 2.39 (3H, s), 3.40-3.50 (2H, m), 6.09 (1 H, s), 6.70 (1H, d, J = 7.7 Hz), 7 . 10 (1 H, d, J = 7.9 Hz), 7.20-7.27 (2H, m), 7.36-7. 41 (2H, m)
FAB-MS (m/e) : 398/400 [M + H] + FAB-MS (m / e): 398/400 [M + H] +
ジァステレオマー b (A r : g-C l -Ph j R^ H i R^ H j R3 : i—P r ; R4 : H ; ( R5 : 3 -C 1—4— Me - Ph; n = 2) Diastereomer b (A r: gC l -Ph j R ^ H i R ^ H j R 3 : i—P r; R 4 : H; ( R 5 : 3 -C 1—4— Me-Ph; n = 2 )
^NMR (CDC 13) (5 : 0. 93 (3H, d, J = 6. 8Hz) , 0. 9 7 (3H, d, J = 6. 6Hz) , 1. 81— 1. 98 ( 1 H, m) , 2. 40 (3H, s) , 3. 00-3. 10 (lH, m) , 3. 67 - 3. 75 (2H, m) , 5. 46 (1H, s) , 6. 66 (1H, d, J = 7. 9Hz) , 7. 0 7 (1H, dd, J = 1. 7, 7. 9Hz) , 7. 21 (1H, d d, J = 6. 9, 7. 9Hz) , 7. 25 ( 1 H, d, J = 7. 9Hz) , 7. 34 ( 1 H, d, J = l. 7Hz) , 7. 42 ( 1 H, d, J = 6. 9Hz) ^ NMR (CDC 1 3) ( 5: 0. 93 (3H, d, J = 6. 8Hz), 0. 9 7 (3H, d, J = 6. 6Hz), 1. 81- 1. 98 (1 H, m), 2.40 (3H, s), 3.00-3.10 (lH, m), 3.67-3.75 (2H, m), 5.46 (1H, s), 6 66 (1H, d, J = 7.9Hz), 7.07 (1H, dd, J = 1.7, 7.9Hz), 7.21 (1H, dd, J = 6.9, 7. 9Hz), 7.25 (1H, d, J = 7.9Hz), 7.34 (1H, d, J = l.7Hz), 7.42 (1H, d, J = 6.9Hz)
FAB-MS (m/e) : 398/400 [M + H] + 実施例 86 FAB-MS (m / e): 398/400 [M + H] + Example 86
化合物例 1201の製造 Preparation of Compound Example 1201
ジァステレオマー a (A r : 9-C 1 -Ph; R1 : Η; R2 : Η; R3 : i—P r ; R4 : H ; R5: 3— F— 4 - F - Ph ; n=2) Jiasutereoma a (A r: 9-C 1 -Ph; R 1: Η; R 2: Η; R 3: i-P r; R 4: H; R 5: 3- F- 4 - F - Ph; n = 2)
'HNMR (CDC 13) d 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 72- 1. 84 ( 1 H, m) , 3. 25 (1H, dd, J = 11. 6, 14. 9Hz) , 3. 40— 3. 50 (2H, m ) , 6. 17 (1H, s) , 6. 66 (1H, d, J = 7. 5Hz) , 7. 00 -7. 10 (1H, m) , 7. 12-7. 30 (3H, m) , 7. 42 (1H, d, J = 8. 1Hz) 'HNMR (CDC 1 3) d 0. 98 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 72- 1. 84 (1 H, m), 3.25 (1H, dd, J = 11.6, 14.9Hz), 3.40—3.50 (2H, m), 6.17 (1H, s), 6.66 (1H, d, J = 7.5 Hz), 7.00 -7.10 (1H, m), 7.12-7.30 (3H, m), 7.42 (1H, d, J = 8.1 Hz)
FAB-MS (m/e) : 386/388 [M + H] + FAB-MS (m / e): 386/388 [M + H] +
ジァステレオマー b ( A r : 9 - C 1 - P h ; R 1 : H ; R 2 : H ; R 3 : i— P r ; R4 : H ; R5: 3 -F-4-F-P h; n= 2) Jiasutereoma b (A r: 9 - C 1 - P h; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 3 -F-4-FP h; n = 2)
iHNMR (CDC 13) <5 : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 82— 1. 95 (lH, m) , 2. 95 一 3. 10 (1H, m) , 3. 60— 3. 78 (2H, m) , 5. 53 (1H, s) , 6. 63 (1H, d, J = 7 · 9Hz) , 7. 00— 7. 09 ( 1 H, m ) , 7. 13-7. 30 (3H, m) , 7. 45 (1H, d, J = 7. 9Hz) FAB-MS (m/e) : 386/388 [M + H] + 実施例 87 iHNMR (CDC 1 3) <5 : 0. 94 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 82- 1. 95 (lH, m), 2.95-3.10 (1H, m), 3.60-3.78 (2H, m), 5.53 (1H, s), 6.63 (1H, d, J = 7 9Hz), 7.00—7.9 (1H, m), 7.13-7.30 (3H, m), 7.45 (1H, d, J = 7.9Hz) FAB-MS (m / e): 386/388 [M + H] + Example 87
化合物例 1208の製造 Preparation of Compound Example 1208
ジァステレオマ一 a (Ar : g-C l -Ph j R' i H j R^ H j R3 : i—P r ; R4: H; R5: 3— F— 4— OMe— Ph ; n = 2)Jiasutereoma one a (Ar: gC l -Ph j R 'i H j R ^ H j R 3: i-P r; R 4: H; R 5: 3- F- 4- OMe- Ph; n = 2)
NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 NMR (CDC 1 3) δ: 0. 98 (3H, d, J = 6. 8Hz), 0. 9
8 (3H, d, J = 6. 8Hz) , 1. 72— 1. 84 (1H, m) , 3. 26 (1H, d d, J = 11. 4, 14. 8Hz) , 3. 35— 3. 52 (2H, m8 (3H, d, J = 6.8 Hz), 1.72—1.84 (1H, m), 3.26 (1H, dd, J = 11.4, 14.8 Hz), 3.35—3 . 52 (2H, m
) , 3. 91 (3H, s) , 6. 07 ( 1 H, s) , 6. 72 ( 1 H, d, J = 7. 5Hz) , 6. 92 - 7. 05 (2H, m) , 7. 12-7. 27 (2H, m), 3.91 (3H, s), 6.07 (1H, s), 6.72 (1H, d, J = 7.5Hz), 6.92-7.05 (2H, m), 7.12-7.27 (2H, m
) , 7. 12-7. 27 (2H, m) , 7. 39 ( 1 H, d, J =8. 0Hz) FAB-MS (m/e) : 398/400 [M + H] + ), 7. 12-7.27 (2H, m), 7.39 (1H, d, J = 8.0Hz) FAB-MS (m / e): 398/400 [M + H] +
ジァステレオマー b (Ar : g-C l -Ph j R^ H j R^ H j R3 : i—P r ; R4 : H ; R5 : 3 - F— 4 - OMe— Ph ; n=2)Jiasutereoma b (Ar: gC l -Ph j R ^ H j R ^ H j R 3: i-P r; R 4: H; R 5: 3 - F- 4 - OMe- Ph; n = 2)
NMR (CDC 13) <5 : 0. 93 (3H, d, J = 6. 7Hz) , 0. 9 NMR (CDC 1 3) <5 : 0. 93 (3H, d, J = 6. 7Hz), 0. 9
7 (3H, d, J = 6. 7Hz) , 1. 85— 1. 98 (1H, m) , 3. 00 一 3. 10 (lH, m) , 3. 65 - 3. 75 (2H, m) , 3. 92 (3H, s) , 5. 46 (1H, s) , 6. 68 ( 1 H, d, J = 7. 2Hz) , 6. 97 (3H, d, J = 6.7Hz), 1.85-1.98 (1H, m), 3.00-3.10 (lH, m), 3.65-3.75 (2H, m ), 3.92 (3H, s), 5.46 (1H, s), 6.68 (1H, d, J = 7.2Hz), 6.9
5-7. 12 (2H, m) , 7. 22 ( 1 H, t, J = 7. 9Hz) FAB-MS (m/e) : 398/400 [M + H] + 実施例 88 5-7. 12 (2H, m), 7.22 (1H, t, J = 7.9Hz) FAB-MS (m / e): 398/400 [M + H] + Example 88
化合物例 1219の製造 (Ar : 9— C I— Ph R H R H R3 : i -P r ; R4 : H ; R5 : 3 - 1 , 4-n-P r NHCOCH£Q-P ; n = 2Preparation of compound examples 1219 (Ar: 9- CI- Ph RHRHR 3: i -P r; R 4: H; R 5: 3 - 1, 4-nP r NHCOCH £ QP; n = 2
L L
^NMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 01 (3H, t, J = 7. 4Hz) , 1 . 55 - 1. 70 (2H, m) , 1. 84— 1. 98 (IH, m) , 3. 02— 3. 09 (IH, m) , 3. 37 (IH, t, J = 6. 9Hz) , 3. 39 (1 H, t, J = 6. 9Hz) , 3. 65 - 3. 78 (2H, m) , 4. ·55 (2H , s) , 5. 48 (1H, s) , 6. 64 ( 1 H, d d, J = l. 2, 7. 9H z) , 6. 81 (1H, d, J = 8. 4Hz) , 7. 21 (1H, b r s) , 7 . 22 - 7. 27 (IH, m) , 7. 30 ( 1 H, d d, J = 2. 2, 8. 4H z) , 7. 45 (IH, d d, J = l. 2, 7. 9Hz) , 7. 77 (1 H, d , J = 2. 2Hz) ^ NMR (CDC 1 3) δ : 0. 94 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 01 (3H, t, J = 7.4 Hz), 1.55-1.70 (2H, m), 1.84-1.98 (IH, m), 3.02-3.09 (IH, m), 3.37 (IH, m) t, J = 6.9 Hz), 3.39 (1 H, t, J = 6.9 Hz), 3.65-3.78 (2H, m), 4.55 (2H, s), 5. 48 (1H, s), 6.64 (1H, dd, J = l. 2, 7.9Hz), 6.81 (1H, d, J = 8.4Hz), 7.21 (1H, brs ), 7.22-7.27 (IH, m), 7.30 (1H, dd, J = 2.2, 8.4Hz), 7.45 (IH, dd, J = l.2, 7.9 Hz), 7.77 (1 H, d, J = 2.2 Hz)
FAB-MS (m/e) : 591/593 [M + H] + 実施例 89 FAB-MS (m / e): 591/593 [M + H] + Example 89
化合物例 1220の製造 (Ar : 9_C l— Ph R^ H R2 : ?! ; !^3 : i -P r ; R4 : H ; R5 : 3- 1 -4 -MeO-Ph ; H ; n=2) Preparation of compound examples 1220 (Ar: 9_C l- Ph R ^ HR 2:;?!! ^ 3: i -P r; R 4: H; R 5: 3- 1 -4 -MeO-Ph; H; n = 2)
^NMR (CDC 13) 6 : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 85-2. 00 ( 1 H, m) , 3. 02 一 3. 12 (lH, m) , 3. 68— 3. 72 (2H, m) , 3. 91 (3H, s) , 5. 44 (1H, s) , 6. 65— 6. 69 ( 1 H, m) , 6. 84 (1 H, d, J = 8. 5Hz) , 7. 20 - 7. 27 (2H, m) , 7. 44 (1 H , dd, J = 1. 3, 8. 1Hz) , 7. 76 (IH, d, J = 2. 1Hz) FAB-MS (m/e) : 506/508 [M + H] + 実施例 90 ^ NMR (CDC 1 3) 6 :. 0. 94 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 85-2 00 (1 H , m), 3.02-3.12 (lH, m), 3.68-3.72 (2H, m), 3.91 (3H, s), 5.44 (1H, s), 6. 65-6.69 (1H, m), 6.84 (1H, d, J = 8.5Hz), 7.20-7.27 (2H, m), 7.44 (1H, dd, J = 1.3, 8.1 Hz), 7.76 (IH, d, J = 2.1 Hz) FAB-MS (m / e): 506/508 [M + H] + Example 90
化合物例 1227の製造 Preparation of Compound Example 1227
ジァステレオマー a (Ar : g-F-Ph j R^ H j R^ H i R3 : i一 P r ; R4: H; R5: Ph; n = 2) Jiasutereoma a (Ar: gF-Ph j R ^ H j R ^ H i R 3: i one P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) <5 : 0. 98 (3H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 70- 1. 85 ( 1 H, m) , 3. 00 (1H, d d, J =l 1. 1, 14. 5Hz) , 3. 42 (1 H, d d, J = 2 . 3, 14. 5Hz) , 3. 50 ( 1 H, d d d, 1 = 2. 3, 4. 8, 11. 1Hz) , 5. 99 (1H, s) , 6. 50 ( 1 H, d, J = 7. 9Hz) , 7 . 05 (1H, d d, J = 7. 9, 11. 0Hz) , 7. 28- 7. 45 (6H , m) XHNMR (CDC 1 3) <5 : 0. 98 (3H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 70- 1. 85 (1 H , M), 3.00 (1H, dd, J = l 1.1, 14.5Hz), 3.42 (1H, dd, J = 2.3, 14.5Hz), 3.50 (1H , Ddd, 1 = 2.3, 4. 8, 11.1Hz), 5.99 (1H, s), 6.50 (1H, d, J = 7.9Hz), 7.05 (1H, dd) , J = 7.9, 11.0Hz), 7.28-7.45 (6H, m)
FAB-MS (m/e) : 334 [M + H] + FAB-MS (m / e): 334 [M + H] +
ジァステレオマー b (Ar : 9 -F-Ph; R1 : H ; R2 : H ; R3 : i—P r ; R4 : H ; R 5 : P h ; n = 2 ) Jiasutereoma b (Ar: 9 -F-Ph ; R 1: H; R 2: H; R 3: i-P r; R 4: H; R 5: P h; n = 2)
XHNMR (CDC 13) δ 0. 92 (3H, d, J = 6. 7Hz) , 0. 9 7 (3H, d, J = 6. 7Hz) , 1. 88— 2. 02 (1H, m) , 3. 00 —3. 10 (1H, m) , 3. 60— 3. 75 (2H, m) , 5. 53 (1H, s) , 6. 53 (1H, d, J = 7. 9Hz) , 7. 11 (1 H, dd, J = 7 . 9, 10. 7Hz) , 7. 25- 7. 45 (6H, m) XHNMR (CDC 1 3) δ 0. 92 (3H, d, J = 6. 7Hz), 0. 9 7 (3H, d, J = 6. 7Hz), 1. 88- 2. 02 (1H, m) , 3.00 — 3.10 (1H, m), 3.60 — 3.75 (2H, m), 5.53 (1H, s), 6.53 (1H, d, J = 7.9 Hz) , 7.11 (1 H, dd, J = 7.9, 10.7 Hz), 7.25-7.45 (6H, m)
FAB-MS (m/e) : 334 [M + H] + 実施例 91 FAB-MS (m / e): 334 [M + H] + Example 91
化合物例 1232の製造 (Ar : 9-F-Ph; R 1: H; R 2: H; R 3: i - P r ; R4: H; R5 : 4-MeO-Ph ; n=2) Preparation of compound examples 1232 (Ar: 9-F- Ph; R 1: H; R 2: H; R 3: i - P r; R 4: H; R 5: 4-MeO-Ph; n = 2)
XHNMR (CDC 13) d : 0. 93 (3H, d, J = 6. 7Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 85— 2. 00 ( 1 H, m) , 2. 98 一 3. 11 (lH, m) , 3. 65— 3. 71 (2H, m) , 3. 84 (3 H, s) , 5. 48 (1H, s) , 6. 56 ( 1 H, d, J = 7. 3Hz) , 6. 9 3 (2H, d, J = 8. 5Hz) , 7. 05— 7. 15 (1H, m) , 7. 24 (2H, d, J = 8. 5Hz) , 7. 25— 7. 36 (1H, m) FAB-MS (mZe) : 364 [M + H] + 実施例 92 XHNMR (CDC 1 3) d: 0. 93 (3H, d, J = 6. 7Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 85- 2. 00 (1 H, m), 2.98-3.11 (lH, m), 3.65-3.71 (2H, m), 3.84 (3H, s), 5.48 (1H, s), 6. 56 (1H, d, J = 7.3Hz), 6.93 (2H, d, J = 8.5Hz), 7.05—7.15 (1H, m), 7.24 (2H, d, J = 8.5 Hz), 7.25—7.36 (1H, m) FAB-MS (mZe): 364 [M + H] + Example 92
化合物例 1261の製造 ( A r : 9 - F - P h; R 1: H; R 2: H; R 3: i一 P r ; R4 : H ; R5 : 3- 1 -4 -MeO-P ; n=2) Preparation of compound examples 1261 (A r: 9 - F - P h; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 3- 1 -4 -MeO- P; n = 2)
XHNMR (CDC 13) d : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 X HNMR (CDC 1 3) d : 0. 94 (3H, d, J = 6. 7Hz), 0. 9
7 (3H, d, J = 6. 6Hz) , 1. 85— 1. 98 (lH, m) , 3. 00 一 3. 10 (lH, m) , 3. 63 - 3. 70 (2H, m) , 3. 91 (3H, s) , 5. 49 (1H, s) , 6. 56 ( 1 H, d, J = 7. 9Hz) , 6. 57 (3H, d, J = 6.6 Hz), 1.85-1.98 (lH, m), 3.00-3.10 (lH, m), 3.63-3.70 (2H, m ), 3.91 (3H, s), 5.49 (1H, s), 6.56 (1H, d, J = 7.9Hz), 6.5
8 (1H, d, J = 8. 3Hz) 7. 10— 7. 17 ( 1 H、 dd、 J = 7. 9 , 8. 5Hz) , 7. 20- 7. 40 (2H, m) , 7. 78 ( 1 H, d, J = 2. 2Hz) 8 (1H, d, J = 8.3Hz) 7.10-7.17 (1H, dd, J = 7.9, 8.5Hz), 7.20-7.40 (2H, m), 7 . 78 (1 H, d, J = 2.2 Hz)
FAB-MS (m/e) : 490 [M + H] + 実施例 93 FAB-MS (m / e): 490 [M + H] + Example 93
化合物例 1275の製造 Preparation of Compound Example 1275
ジァステレオマー a (A r : 7— I一 8— n— P rNHCOCH2〇一 Ph^ R1 : H; R2: H; R3: i -P r ; R4: H; R 5: P ; n = 2 ) Jiasutereoma a (A r: 7- I one 8- n- P rNHCOCH 2 〇 one Ph ^ R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: P; n = 2)
XHNMR (CDC 13) (5 : 0. 95 (3H, t, J = 7. 4Hz) , 0. 9 7 (3H, d, J = 7. 5Hz) , 0. 97 (3H, d, J = 7. 5Hz) , 1 . 50- 1. 68 (2H, m) , 1. 73— 1. 88 (1H, m) , 3. 07 ( 1H, d d, J = 11. 0, 14. 1Hz) , 3. 34 (1H, t, J = 6. 7 Hz) , 3. 36 (1 H, t, J = 6. 7Hz) , 3. 43 ( 1 H, d d, J = 2. 0, 14. 1Hz) , 3. 45 - 3. 52 ( 1 H, m) , 4. 59 (2H, s) , 5. 75 (1H, s) , 6. 86 ( 1 H, b r s) , 7. 10 ( 1 H, s ) , 7. 35-7. 48 (5H, m) , 7. 54 ( 1 H, s) XHNMR (CDC 1 3) (5 : 0. 95 (3H, t, J = 7. 4Hz), 0. 9 7 (3H, d, J = 7. 5Hz), 0. 97 (3H, d, J = 7.5 Hz), 1.50- 1.68 (2H, m), 1.73-1.88 (1H, m), 3.07 (1H, dd, J = 11.0, 14.1Hz), 3.34 (1H, t, J = 6.7 Hz), 3.36 (1 H, t, J = 6.7 Hz), 3.43 (1 H, dd, J = 2.0, 14.1 Hz ), 3.45-3.52 (1H, m), 4.59 (2H, s), 5.75 (1H, s), 6.86 (1H, brs), 7.10 (1H , s), 7.35-7.48 (5H, m), 7.54 (1H, s)
FAB-MS (m/e) : 557 [M + H] + FAB-MS (m / e): 557 [M + H] +
ジァステレオマー b (A r : 7— I一 8— n— P rNHC〇CH2〇一 Ph R1 : H ; R2 : H ; R3 : i -P r ; R4 : H ; R5 : P ; n=2) NMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 00 (3H, t, J = 7. 3Hz) , 1 . 55— 1. 70 (2H, m) , 1. 90— 2. 01 (lH, m) , 2. 95 - 3. 01 (lH, m) , 3. 36 (1H, t, J = 6. 7Hz) , 3. 38 (1 H, t, J = 6. 7Hz) , 3. 42 ( 1 H, d d, J = 9. 3, 14. 5Hz ) , 3. 49 (IH, dd, J = 3. 0, 14. 5Hz) , 4. 61 (2H, s ) , 5. 73 (IH, s) , 6. 88 ( 1 H, b r s) , 7. 26- 7. 44 ( 7H, m) , 7. 52 (IH, s) Diastereomer b (Ar: 7—I-1 8—n—P rNHC〇CH 2 PhPh R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: P; n = 2) NMR (CDC 1 3) (5: 0. 95 (3H, d, J = 6 8Hz), 0.98 (3H, d, J = 6.8Hz), 1.00 (3H, t, J = 7.3Hz), 1.55-1.70 (2H, m), 1. 90—2.11 (lH, m), 2.95-3.01 (lH, m), 3.36 (1H, t, J = 6.7Hz), 3.38 (1H, t, J = 6.7Hz), 3.42 (1H, dd, J = 9.3, 14.5Hz), 3.49 (IH, dd, J = 3.0, 14.5Hz), 4.61 (2H, s), 5.73 (IH, s), 6.88 (1H, brs), 7.26-7.44 (7H, m), 7.52 (IH, s)
FAB-MS (m/e) : 557 [M+H] + 実施例 94 FAB-MS (m / e): 557 [M + H] + Example 94
化合物例 1276の製造 Preparation of Compound Example 1276
ジァステレオマー a (Ar : 7- 1 -8 -MeO-Ph j R^ H j R^ H j R 3: i -P r ; R4: H; R5: Ph; n=2) Jiasutereoma a (Ar: 7- 1 -8 -MeO -Ph j R ^ H j R ^ H j R 3: i -P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) (5 : 0. 97 (3H, d, J = 6. 9Hz) , 0. 9 X HNMR (CDC 1 3) ( 5: 0. 97 (3H, d, J = 6. 9Hz), 0. 9
7 (3H, d, J = 6. 9Hz) , 1. 75 - 1. 85 (lH, m) , 3. 08 (IH, dd, J =l 1. 2, 14. 2Hz) , 3. 43 (IH, dd, J = 27 (3H, d, J = 6.9 Hz), 1.75-1.85 (lH, m), 3.08 (IH, dd, J = l 1.2, 14.2 Hz), 3.43 ( IH, dd, J = 2
. 3, 14. 2Hz) , 3. 48 ( 1 H, d d d, J = 2. 3, 5. 0, 11. 2Hz) , 3. 95 (3H, s) , 5. 75 ( 1 H, s) , 7. 08 ( 1 H, s3, 14.2Hz), 3.48 (1H, ddd, J = 2.3, 5.0, 11.2Hz), 3.95 (3H, s), 5.75 (1H, s) , 7. 08 (1 H, s
) , 7. 31-7. 47 (5H, m) , 7. 55 (IH, s) ), 7.31-7.47 (5H, m), 7.55 (IH, s)
FAB-MS (mZe) : 472 [M + H] + FAB-MS (mZe): 472 [M + H] +
ジァステレオマー b (Ar : 7- 1 -8 -MeO-Ph j R^ H j R^ H j RDiastereomer b (Ar: 7-1 -8 -MeO-Ph j R ^ H j R ^ H j R
3 : i -P r ; R4 : H ; R5 : Ph ; n = 2) 3 : i -P r; R 4 : H; R 5 : Ph; n = 2)
NMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 9Hz) , 0. 9 NMR (CDC 1 3) δ: 0. 95 (3H, d, J = 6. 9Hz), 0. 9
8 (3H, d, J = 6. 7Hz) , 1. 88- 1. 99 ( 1 H, m) , 2. 98 (1H, d d d, J = 3. 0, 5. 8, 9. 4Hz) , 3. 41 (IH, dd,8 (3H, d, J = 6.7Hz), 1.88-1.99 (1H, m), 2.98 (1H, ddd, J = 3.0, 5.8, 9.4Hz), 3.41 (IH, dd,
J = 9. 4, 14. 3Hz) , 3. 49 ( 1 H, dd, J = 3. 0, 14. 3H z) , 3. 98 (3H, s) , 5. 71 ( 1 H, s) , 7. 26-7. 41 (5 H, m) , 7. 33 (1H, s) , 7. 54 (1H, s) J = 9.4, 14.3 Hz), 3.49 (1H, dd, J = 3.0, 14.3Hz), 3.98 (3H, s), 5.71 (1H, s) , 7.26-7.41 (5 H, m), 7.33 (1H, s), 7.54 (1H, s)
FAB-MS (m/e) : 472 [M + H] + 実施例 95 FAB-MS (m / e): 472 [M + H] + Example 95
化合物例 1277の製造 (Ar : Q- I-e-MeQ-Ph i R1 : H ; R2 : H ; R3: i -P r ; R4 : H ; R5 : Ph ; n=2) Preparation of compound examples 1277 (Ar: Q- Ie-MeQ -Ph i R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) δ : 0. 96 (3H, d, J =6. 9Hz) , 0. 9 9 (3H, d, J = 6. 9Hz) , 1. 72— 1. 89 (lH,. m) , 3. 10 -3. 33 (3H, m) , 3. 51 (3H, s) , 6. 38 ( 1 H, s) , 7. 18-7. 26 (2H, m) , 7. 26 - 7. 33 (3H, m) , 7. 79 (1 H, d, J = 8. 4Hz) , 7. 99 (1H, d, J = 8. 4Hz) X HNMR (CDC 1 3) δ : 0. 96 (. 3H, d, J = 6 9Hz), 0. 9 9 (3H, d, J = 6. 9Hz), 1. 72- 1. 89 (lH, m), 3.10 -3. 33 (3H, m), 3.51 (3H, s), 6.38 (1H, s), 7.18-7.26 (2H, m), 7 26-7.33 (3H, m), 7.79 (1H, d, J = 8.4Hz), 7.99 (1H, d, J = 8.4Hz)
FAB-MS {m/e) : 472 [M + H] + 実施例 96 FAB-MS (m / e): 472 [M + H] + Example 96
化合物例 1278の製造 (Ar : 6- I -9-QH-P ; R1: H; R2: H; R3: 卜 P r ; R4: H; R5: Ph ; n = 2) Preparation of compound examples 1278 (Ar: 6- I -9- QH-P; R 1: H; R 2: H; R 3: Bok P r; R 4: H; R 5: Ph; n = 2)
XHNMR (CDC 13) δ : 0. 90 (3H, d, J = 6. 6Hz) , 0. 9 6 (3H, d, J = 6. 7Hz) , 1. 85— 1. 95 (1H, m) , 3. 00 — 3. 10 (lH, m) , 3. 64- 3. 7 1 (2H, m) , 4. 07 (3H, s) , 5. 41 (1H, s) , 6. 21 ( 1 H, d, J = 8. 5Hz) , 7. 2 5-7. 45 (5H, m) , 7. 71 (1H, d, J = 8. 5Hz) X HNMR (CDC 1 3) δ : 0. 90 (3H, d, J = 6. 6Hz), 0. 9 6 (3H, d, J = 6. 7Hz), 1. 85- 1. 95 (1H, m), 3.00 — 3.10 (lH, m), 3.64 3.71 (2H, m), 4.07 (3H, s), 5.41 (1H, s), 6. 21 (1H, d, J = 8.5Hz), 7.25-7. 45 (5H, m), 7.71 (1H, d, J = 8.5Hz)
FAB-MS (m/e) : 472 [M + H] + 実施例 97 FAB-MS (m / e): 472 [M + H] + Example 97
化合物例 1093の製造 (5— (4一クロ口— 3—メチルフエニル) 一 3—イソ プロピル一 2, 3, 4, 5ーテトラヒドロー — 1 6, 4—べンゾチアゼピ ンー 1, 1ージオン) Preparation of compound examples 1093 (5- (4 one black port - 3-methylphenyl) Single 3-isopropyl one 2, 3, 4, 5 over tetrahydropyran over - 1 6, 4 base Nzochiazepi Hmm 1, 1-dione)
チォサリチル酸メチルエステル 10. 0 g (59. 5mmo 1 ) を含むメタノ —ル溶液 20mlに、 実施例 1の工程 1で得られた 2—イソプロピルアジリジン 10mlを、 Otにて 10分間かけて滴下した。 反応溶液を室温に戻しー晚攪拌 後、 反応溶液を減圧下に濃縮した。 残査にテトラヒドロフラン溶液 100m lを 加え、 続いてトリェチルァミン 16. 6m l (1 19mmo 1 ) 、 二炭酸— t— ブチル 14. 2 g (65. 4mmo 1) を加え、 2時間攪拌した。 酢酸ェチルェ ステルで反応溶液を抽出し、 有機層を 10%硫酸水素カリウム溶液、 飽和食塩水 溶液で洗浄後、 硫酸マグネシウムで乾燥後、 減圧下に濃縮し、 得られた残渣をシ リカゲルカラムクロマトグラフィー (へキサン:酢酸ェチルエステル =4 : 1) により精製し、 メチル 2— (-2- { ( t—ブトキシカルポニル) アミノ} 一 3—メチルブチル) スルファニル) ベンゾエート 12. 4 g (収率: 59%) を 淡黄色油状物として得た。 2-isopropylaziridine obtained in Step 1 of Example 1 was added to 20 ml of a methanol solution containing 10.0 g (59.5 mmol) of thiosalicylic acid methyl ester. 10 ml was added dropwise over 10 minutes with Ot. After the reaction solution was returned to room temperature and stirred, the reaction solution was concentrated under reduced pressure. To the residue was added 100 ml of a tetrahydrofuran solution, followed by 16.6 ml (119 mmo 1) of triethylamine and 14.2 g (65.4 mmo 1) of t-butyl dicarbonate, followed by stirring for 2 hours. The reaction solution was extracted with ethyl acetate, and the organic layer was washed with a 10% potassium hydrogen sulfate solution and a saturated saline solution, dried over magnesium sulfate, concentrated under reduced pressure, and the resulting residue was subjected to silica gel column chromatography. (Hexane: ethyl acetate = 4: 1) to give methyl 2-(-2-{(t-butoxycarbonyl) amino} -1-methylbutyl) sulfanyl) benzoate 12.4 g (yield: 59%) Was obtained as a pale yellow oil.
^NMR (CDC 13) δ : 0. 93 (3H, d, J = 6. 7Hz) , 0. 9 7 (3H, d, J = 6. 7Hz) , 1. 42 (9H, s) , 2. 00— 2. 1 0 (lH, m) , 3. 01— 3. 15 (2H, m) , 3. 70— 3. 82 ( 1 H, m) , 3. 9 1 (3H, s) , 4. 71 ( 1 H, b s) , 7. 12-7. 20 ( 1H, m) , 7. 39- 7. 46 (2H, m) , 7. 92 (1H, d, J = 6. 9Hz) ^ NMR (CDC 1 3) δ : 0. 93 (3H, d, J = 6. 7Hz), 0. 9 7 (3H, d, J = 6. 7Hz), 1. 42 (9H, s), 2 00—2.10 (lH, m), 3.01—3.15 (2H, m), 3.70—3.82 (1H, m), 3.91 (3H, s), 4.71 (1H, bs), 7.12-7.20 (1H, m), 7.39-7.46 (2H, m), 7.92 (1H, d, J = 6.9Hz)
FAB-MS (m/e) : 354 [M + H] + FAB-MS (m / e): 354 [M + H] +
工程 1で得られたメチル 2— ( (2— { ( t一ブトキシカルポニル) ァミノ } - 3—メチルブチル) スルファニル) ベンゾエート 12. 4 g (35. lmm o 1 ) のメタノール溶液 (100ml) に、 氷冷下、 2 N水酸化ナトリゥム水溶 液 35ml (70. Ommo 1) を加え、 室温で反応溶液を一晩攪拌した。 反応 溶液を減圧下に濃縮し、 得られた残渣に 1N塩酸水溶液 (40ml) を加えクロ 口ホルムで抽出し、 有機層を飽和食塩水溶液で洗浄し、 硫酸マグネシウムで乾燥 し、 減圧下に濃縮した。 残査に塩化メチレン溶液 50mlを加え、 続いて 1ーヒ ドロキシベンゾトリアゾール 10. 0 g (1 19 mm o 1 ) 、 1— (3—ジメチ ルァミノプロピル) 一 3—ェチルカルポジイミド塩酸塩 10. 0 g、 N, O—ジ メチルヒドロキシルァミン塩酸塩 8. 79 gを加え、 4時間攪拌した。 有機反応 溶液に飽和塩化アンモニゥム水溶液を添加し、 酢酸ェチルエステルで抽出し、 有 機層を 10%硫酸水素カリウム溶液、 飽和食塩水溶液で洗浄後、 硫酸マグネシゥ ムで乾燥し、 減圧下に濃縮した。 得られた残渣をシリカゲルカラムクロマトダラ フィー (へキサン:酢酸ェチルエステル =4 : 1) により精製し、 t一ブチル N- (1- { ( {2- ( {メトキシ (メチル) アミノ} カルボニル) フエ二ル} スルファニル) メチル } 一 2—メチルプロピル) 力ルバメート 8. 15 g (収率 : 60%) を淡黄色油状物として得た。 To a methanol solution (100 ml) of methyl 2-((2-((t-butoxycarbonyl) amino) -3-methylbutyl) sulfanyl) benzoate obtained in Step 1 was added ice-cold solution to 100 ml of methanol. Under cooling, 35 ml of a 2 N aqueous sodium hydroxide solution (70. Ommo 1) was added, and the reaction solution was stirred at room temperature overnight. The reaction solution was concentrated under reduced pressure, a 1N aqueous hydrochloric acid solution (40 ml) was added to the obtained residue, and the mixture was extracted with chloroform. The organic layer was washed with saturated saline solution, dried over magnesium sulfate, and concentrated under reduced pressure. . 50 ml of methylene chloride solution was added to the residue, followed by 1-hydroxybenzotriazole 10.0 g (119 mmo 1), 1- (3-dimethylaminopropyl) -13-ethylcarbodiimide hydrochloride 10. 0 g and 8.79 g of N, O-dimethylhydroxylamine hydrochloride were added, and the mixture was stirred for 4 hours. A saturated aqueous solution of ammonium chloride was added to the organic reaction solution, extracted with ethyl acetate, and the organic layer was washed with a 10% potassium hydrogen sulfate solution and a saturated saline solution. Dried under reduced pressure and concentrated under reduced pressure. The obtained residue was purified by silica gel column chromatography (hexane: ethyl acetate = 4: 1) to give t-butyl N- (1-{({2-({methoxy (methyl) amino} carbonyl) carbonyl) phenyl. 8.15 g (yield: 60%) of the compound {l-sulfanyl) methyl} -1-methylpropyl) caprate were obtained as a pale yellow oil.
XHNMR (CDC 13) 8 : 0. 89 (3H, d, J = 6. 6Hz) , 0. 9 0 (3H, d, J = 6. 8Hz) , 1. 36 (9H, s) , 1. 82-2. 00 (1H, m) , 3. 01-3. 20 (3H, m) , 3. 32 (3H, b s) , 3 . 53 (3H, b s) , 5. 08 (1H, b s) , 7. 21— 7. 27 (2H, m) , 7. 27 - 7. 38 ( 1 H, m) , 7. 54 (1H, d, J = 7. 5Hz ) X HNMR (CDC 1 3) 8 : 0. 89 (3H, d, J = 6. 6Hz), 0. 9 0 (3H, d, J = 6. 8Hz), 1. 36 (9H, s), 1 82-2. 00 (1H, m), 3.01-3. 20 (3H, m), 3.32 (3H, bs), 3.53 (3H, bs), 5.08 (1H, bs) ), 7.21-7.27 (2H, m), 7.27-7.38 (1H, m), 7.54 (1H, d, J = 7.5Hz)
FAB-MS (mZe) : 383 [M + H] + FAB-MS (mZe): 383 [M + H] +
工程 2で得られた t一ブチル N— (1— { ( {2- ( {メトキシ (メチル) アミノ} カルボニル) フエ二ル} スルファニル) メチル } 一 2—メチルプロピル ) 力ルバメート 150mg (0. 39 mm 01 ) を含むテトラヒドロフラン溶液 (2ml ) に、 マグネシウム 117mg (4. 2 mm o 1 ) と 2—クロ口一 5— ブロモトルエン 805mg (3. 9mmo 1) とにより調製したグリニアル試薬 のテトラヒドロフラン溶液 (5ml) を、 窒素雰囲気下、 — 78°Cにて 10分間 かけて滴下した。 反応溶液を— 70°Cで 2. 5時間攪拌後、 反応溶液に飽和塩化 アンモニゥム水溶液を添加した。 酢酸ェチルエステルで反応溶液を抽出し、 有機 層を飽和食塩水溶液で洗浄後、 硫酸マグネシウムで乾燥し、 減圧下に濃縮た。 得 られた残渣をシリカゲルカラムクロマトグラフィー (へキサン:酢酸ェチルエス テル =4: 1) により精製し、 t—ブチル N— (1— { ( {2— (4—クロ口 一 3 _メチルベンゾィル) フエ二ル} スルファニル) メチル } 一 2一メチルプロ ピル) 力ルバメート 107mg (収率: 61%) を淡黄色油状物として得た。 T-Butyl N- (1 — {({2-({methoxy (methyl) amino} carbonyl) phenyl) sulfanyl) methyl} -12-methylpropyl) -capillate obtained in step 2 150 mg (0.39 mm 01) in a tetrahydrofuran solution (2 ml) and magnesium solution 117 mg (4.2 mmo 1) and 2-bromo-1-5-805-bromotoluene 805 mg (3.9 mmo 1) in a tetrahydrofuran solution (5 ml) ) Was added dropwise at −78 ° C. over 10 minutes under a nitrogen atmosphere. After stirring the reaction solution at -70 ° C for 2.5 hours, a saturated aqueous solution of ammonium chloride was added to the reaction solution. The reaction solution was extracted with ethyl acetate, and the organic layer was washed with a saturated saline solution, dried over magnesium sulfate, and concentrated under reduced pressure. The obtained residue was purified by silica gel column chromatography (hexane: ethyl acetate = 4: 1), and t-butyl N— (1 — {({2 -— (4-chloro-1,3-methylbenzoyl) phenyl) {Sulfanyl) methyl} -1-methylpropyl) lubamate 107 mg (yield: 61%) was obtained as a pale yellow oil.
XHNMR (CDC 13) δ : 0. 84 (3Η, d, J = 6. 8Hz) , 0. 8 X HNMR (CDC 1 3) δ : 0. 84 (3Η, d, J = 6. 8Hz), 0. 8
4 (3H, d, J = 6. 8Hz) , 1. 37 (9H, s) , 1. 75— 1. 85 (lH, m) , 2. 29 (3H, s) , 2. 95 - 3. 05 (2H, m) , 3.4 (3H, d, J = 6.8 Hz), 1.37 (9H, s), 1.75—1.85 (lH, m), 2.29 (3H, s), 2.95-3. 05 (2H, m), 3.
50 - 3. 60 (1H, m) , 4. 75 ( 1 H, b s) , 7. 26-7. 31 ( 2H, m) , 7. 39 -7. 59 (4H, m) , 7. 68 (1H, d, J = 1. 9Hz) 50-3.60 (1H, m), 4.75 (1H, bs), 7.26-7.31 ( 2H, m), 7.39 -7.59 (4H, m), 7.68 (1H, d, J = 1.9Hz)
FAB-MS (m/ e ) : 449 [M + H] + FAB-MS (m / e): 449 [M + H] +
工程 3で得られた t—ブチル N— (1一 { ( {2- (4—クロロー 3—メチ ルペンゾィル) フエ二ル} スルファニル) メチル } —2—メチルプロピル) カル バメート 68mg (0. 15 mm o 1 ) に 4N塩化水素 1, 4一ジォキサン溶液 (3. 0ml) を加え室温で一晩攪拌し、 反応溶液を減圧下に濃縮した。 残渣に 2, 6—ルチジン (3. 0ml) を加え、 濃塩酸 (1. 0ml ) を添加した。 添 加終了後、 混合物を 3時間共沸させ、 減圧留去した。 残渣を飽和炭酸水素ナトリ ゥム水溶液で回収し、 溶液を酢酸ェチルで 2回抽出した。 集めた抽出物を飽和食 塩水溶液で洗浄し、 乾燥し、 減圧下に濃縮した。 残渣にトリフルォロ酢酸 (3. 0ml) 溶液を加え、 30 %過酸化水素水 ( 1. 0ml ) を約 0 °Cの温度で添加 した。 添加終了後、 混合物を室温で一晩攪拌し、 10%チォ硫酸ナトリウム水溶 液、 飽和炭酸水素ナトリウム水溶液で処理し、 酢酸ェチルで 2回抽出し、 抽出物 を集めて飽和食塩水溶液で洗浄し、 乾燥し、 減圧下に濃縮した。 残渣に 4 N塩ィ匕 水素 1, 4—ジォキサン溶液 (3. 0ml) を加え、 亜鉛粉末 (200mg) を 徐々に添加した。 混合液を一晩室温で攪拌し、 飽和炭酸水素ナトリウム水溶液で 処理し、 セライトで濾過した。 酢酸ェチルで 2回抽出し、 抽出物を集めて飽和食 塩水溶液で洗浄し、 乾燥し、 減圧下に濃縮した。 残渣をシリカゲル上で、 へキサ ン /酢酸ェチル (4 : 1) を溶離剤とするカラムクロマトグラフィーに付し、 5 一 (4 _クロロー 3—メチルフエニル) 一 3—イソプロピル一 2, 3, 4, 5一 テトラヒドロ— Iff— 1 λ 6, 4 _ベンゾチアゼピン一 1, 1ージオンのジァス テレオマー a (12. 8mg) および b (22. 3mg) を淡黄色の油状生成物 として得た。 T-butyl N- (111-(({2- (4-chloro-3-methylpentenezyl) phenyl} sulfanyl) methyl} —2-methylpropyl) obtained in step 3 Carbamate 68 mg (0.15 mm To 1) was added a 4N hydrogen chloride 1,4-dioxane solution (3.0 ml), the mixture was stirred at room temperature overnight, and the reaction solution was concentrated under reduced pressure. 2,6-Lutidine (3.0 ml) was added to the residue, and concentrated hydrochloric acid (1.0 ml) was added. After the addition was completed, the mixture was azeotropically distilled for 3 hours and distilled under reduced pressure. The residue was collected with a saturated aqueous solution of sodium hydrogen carbonate, and the solution was extracted twice with ethyl acetate. The combined extracts were washed with saturated brine solution, dried and concentrated under reduced pressure. To the residue was added a solution of trifluoroacetic acid (3.0 ml), and 30% aqueous hydrogen peroxide (1.0 ml) was added at a temperature of about 0 ° C. After the addition was completed, the mixture was stirred at room temperature overnight, treated with a 10% aqueous sodium thiosulfate solution and a saturated aqueous sodium hydrogen carbonate solution, extracted twice with ethyl acetate, and the combined extracts were washed with a saturated saline solution. Dry and concentrate under reduced pressure. To the residue was added a 4 N salt / hydrogen 1,4-dioxane solution (3.0 ml), and zinc powder (200 mg) was gradually added. The mixture was stirred overnight at room temperature, treated with saturated aqueous sodium bicarbonate and filtered through celite. The mixture was extracted twice with ethyl acetate, and the combined extracts were washed with a saturated aqueous solution of sodium chloride, dried and concentrated under reduced pressure. The residue was subjected to column chromatography on silica gel using hexane / ethyl acetate (4: 1) as an eluent to give 5- (4-chloro-3-methylphenyl) -13-isopropyl-1,2,3,4, 5 one-tetrahydro - Iff- 1 λ 6, was obtained as a 4 _ benzothiazepine one 1, 1-dione of Jiasu Tereoma a (12. 8 mg) and b (22. 3 mg) as a pale yellow oily product.
ジァステレオマー a Diastereomer a
XHNMR (CDC 13) δ: 0. 98 (3Η, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 75 - 1. 89 ( 1 H, m) , 2. 40 (3H, s) , 3. 05 (1H, dd, J = 11. 3, 14. 2Hz) , 3. 4 4 (1H, dd, J = 2. 3, 14. 2Hz) , 3. 50 ( 1 H, ddd, J = 2. 3, 4. 8, 1 1. 3Hz) , 5. 85 (1H, s) , 7. 1 8 ( 1 H, d d, J = 2. 2, 8. 2Hz) , 7. 26 ( 1 H, d, J = 2. 2Hz) , 7. 36-7. 43 (3H, m) , 8. 13-8. 17 (lH, m) X HNMR (CDC 1 3) δ : 0. 98 (3Η, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 75 - 1. 89 (1 H , m), 2.40 (3H, s), 3.05 (1H, dd, J = 11.3, 14.2 Hz), 3.44 (1H, dd, J = 2.3, 14.2 Hz ), 3.50 (1 H, ddd, J = 2.3, 4.8, 11.3Hz), 5.85 (1H, s), 7.18 (1H, dd, J = 2.2, 8.2Hz), 7.26 (1H , d, J = 2.2 Hz), 7.36-7.43 (3H, m), 8.13-8.17 (lH, m)
FAB-MS (m/e) : 364 [M + H] + FAB-MS (m / e): 364 [M + H] +
ジァステレオマー b Geostereomer b
^NMR (CDC 13) d : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 90— 2. 05 (1H, m) , 2. 37 (3H, s ) , 2. 98 (1 H, d d d, J = 3. 5, 5. 9, 8. 9Hz) , ^ NMR (CDC 1 3) d : 0. 95 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 90- 2. 05 (1H, m), 2.37 (3H, s), 2.98 (1 H, ddd, J = 3.5, 5.9, 8.9 Hz),
3. 43 (1H, dd, J = 8. 9, 14. 3Hz) , 3. 49 ( 1 H, dd, J = 3. 5, 14. 3Hz) , 5. 80 (1H, s) , 6. 89— 6. 94 (13.43 (1H, dd, J = 8.9, 14.3Hz), 3.49 (1H, dd, J = 3.5, 14.3Hz), 5.80 (1H, s), 6. 89— 6. 94 (1
H, m) , 7. 09 (1H, d d, J = 2. 1, 8. 3Hz) , 7. 19 (1H , d, J = 2. 1Hz) , 7. 33 (1H, d, J = 8. 3Hz) , 7. 45- 7. 52 (2H, m) , 8. 13— 8. 18 (1H, m) H, m), 7.09 (1H, dd, J = 2.1, 8.3Hz), 7.19 (1H, d, J = 2.1Hz), 7.33 (1H, d, J = 8 3 Hz), 7.45-7.52 (2H, m), 8.13—8.18 (1H, m)
FAB-MS (m/e) : 364 [M + H] + 以下に実施例 97と同様にして得た化合物の物理定数を示す。 FAB-MS (m / e): 364 [M + H] + The physical constants of the compound obtained in the same manner as in Example 97 are shown below.
実施例 98 Example 98
化合物例 1008の製造 (Ar : ?!! ; !^1 : !! ; !^2 : !^ ; !^3 : i— P r ; R4 : H; R5: 4-Py r i dy 1 ; n=0) Production of Compound Example 1008 (Ar:? !!;! ^ 1 : !!;! ^ 2 :! ^;! ^ 3 : i—P r; R 4 : H; R 5 : 4-Pyri dy 1; n = 0)
'HNMR (CDC 13) δ : 0. 95. (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, 1 = 6. 6Hz) , 1. 63- 1. 80 (lH, m) , 2. 41 (1H, d d, J = 10. 9, 14. 0Hz) , 2. 91 ( 1 H, dd, J = 2 . 2, 14. 0Hz) , 2. 96 - 3. 04 (1H, m) , 5. 75 ( 1 H, s ) , 6. 5 1— 6. 54 (1H, m) , 7. 15-7. 19 (2H, m) , 7. 41 (2H, d, J = 6. 1Hz) , 7. 67-7. 71 ( 1 H, m) , 8. 6 1 (2H, d, J = 6. 1Hz) 'HNMR (CDC 1 3) δ : 0. 95. (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, 1 = 6. 6Hz), 1. 63- 1. 80 (lH , M), 2.41 (1H, dd, J = 10.9, 14.0 Hz), 2.91 (1H, dd, J = 2.2, 14.0 Hz), 2.96-3.04 (1H, m), 5.75 (1H, s), 6.5 1—6.54 (1H, m), 7.15-7.19 (2H, m), 7.41 (2H, d , J = 6.1 Hz), 7.67-7.71 (1H, m), 8.61 (2H, d, J = 6.1Hz)
FAB-MS (m/e) : 285 [M + H] + 実施例 99 化合物例 1018の製造 FAB-MS (m / e): 285 [M + H] + Example 99 Preparation of Compound Example 1018
ジァステレオマー a (Ar i Ph i R^ H ^^ H ,^3 -P r : R4 : H ; R5: 4-Me -Ph; n = 2) Diastereomer a (Ar i Ph i R ^ H ^^ H, ^ 3 -P r: R 4 : H; R 5 : 4-Me -Ph; n = 2)
^NMR (CDC 13) (5 : 0. 97 (3H, d, J = 6. 8Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 70 - 1. 87 (lH, m) , 2. 40 (3H, s) , 3. 07 (1H, dd, J = 11. 4, 14. 3Hz) , 3. 4 2 (1H, dd, J = 2. 3, 14. 3Hz) , 3. 52 (1H, ddd, J = ^ NMR (CDC 1 3) ( 5: 0. 97 (3H, d, J = 6. 8Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 70 - 1. 87 (lH , M), 2.40 (3H, s), 3.07 (1H, dd, J = 11.4, 14.3 Hz), 3.42 (1H, dd, J = 2.3, 14.3 Hz ), 3.52 (1H, ddd, J =
2, 3, 4. 9, 11. 4Hz) , 5. 82 ( 1 H, s) , 6. 71-6. 75 (1H, m) , 7. 21-7. 30 (4H, m) , 7. 32-7. 41 (2H, m) , 8. 12-8. 16 ( 1 H, m) 2, 3, 4.9, 11.4 Hz), 5.82 (1H, s), 6.71-6.75 (1H, m), 7.21-7.30 (4H, m), 7 32-7. 41 (2H, m), 8.12-8. 16 (1H, m)
FAB-MS (m/e) : 330 [M + H] + FAB-MS (m / e): 330 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i— P r ; R4 : H ; R5: 4— Me - Ph; n=2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i- P r; R 4: H; R 5: 4- Me - Ph; n = 2)
XHNMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 93 - 2. 00 ( 1 H, m) , 2. 37 (3H, s) , 2. 96 (1H, ddd, J = 3. 6, 5. 7, 8. 9Hz) , XHNMR (CDC 1 3) δ: 0. 96 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 93 - 2. 00 (1 H, m), 2.37 (3H, s), 2.96 (1H, ddd, J = 3.6, 5.7, 8.9 Hz),
3. 44 (1 H, dd, J = 8. 9, 14. 3Hz) , 3. 47 (1H, dd, J = 3. 6, 14. 3Hz) , 5. 80 ( 1 H, s) , 6. 91-6. 95 (1 H, m) , 7. 16-7. 23 (4H, m) , 7. 43- 7. 49 (2H, m) , 8. 13-8. 17 (1H, m) 3.44 (1H, dd, J = 8.9, 14.3Hz), 3.47 (1H, dd, J = 3.6, 14.3Hz), 5.80 (1H, s), 6 91-6. 95 (1 H, m), 7.16-7.23 (4H, m), 7.43-7.49 (2H, m), 8.13-8.17 (1H, m) )
FAB-MS (m/e) : 330 [M + H] + 実施例 100 FAB-MS (m / e): 330 [M + H] + Example 100
化合物例 1019の製造 Preparation of Compound Example 1019
ジァステレオマ一 a (A r : Ph R H ; 2 : H : R 3 : i -P r ; R4 : H ; R5: 3 -Me -Ph; n = 2) Diastereomer a (A r: Ph RH; 2 : H: R 3 : i -P r; R 4 : H; R 5 : 3 -Me -Ph; n = 2)
XHNMR (CDC 13) (5 : 0 97 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 73 - 1. 88 (1H, m) , 2. 38 XHNMR (CDC 1 3) (5 : 0 97 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 73 - 1. 88 (1H, m ), 2.38
(3H, s) , 3. 07 (1H, dd, J =l 1. 4, 14. 3Hz) , 3. 4 2 (1H, dd, J = 2. 2, 14. 3Hz) , 3. 52 (1H, d d d, J =(3H, s), 3.07 (1H, dd, J = l 1.4, 14.3 Hz), 3.4 2 (1H, dd, J = 2.2, 14.3Hz), 3.52 (1H, ddd, J =
2, 2, 4. 9, 1 1. 4Hz) , 5. 83 ( 1 H, s) , 6. 70-6. 74 (lH, m) , 7. 12-7. 22 (3H, m) , 7. 27-7. 41 (3H, m) , 8. 12-8. 16 ( 1 H, m) 2, 2, 4. 9, 1 1.4 Hz), 5.83 (1 H, s), 6.70-6.74 (lH, m), 7.12-7.22 (3H, m), 7.27-7. 41 (3H, m), 8.12-8.16 (1H, m)
FAB-MS (m/e) : 330 [M + H] + FAB-MS (m / e): 330 [M + H] +
ジァステレオマー b ( A r : P h ; R 1 : H ; R 2 : H ; R 3 : i一 P r ; R4 : H ; R5: 3 - Me— Ph ; n = 2) Jiasutereoma b (A r: P h; R 1: H; R 2: H; R 3: i one P r; R 4: H; R 5: 3 - Me- Ph; n = 2)
^NMR (CDC 13) 6 : 0. 95 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 92— 2. 04 ( 1 H, m) , 2. 36 (3H, s) , 2. 96 (1H, ddd, J = 3. 7, 5. 9, 8. 9Hz) , ^ NMR (CDC 1 3) 6 : 0. 95 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 92- 2. 04 (1 H , M), 2.36 (3H, s), 2.96 (1H, ddd, J = 3. 7, 5. 9, 8.9 Hz),
3. 46 (1H, d d, J = 8. 9, 14. 3Hz) , 3. 50 ( 1 H, d d, J = 3. 7, 14. 3Hz) , 5. 80 ( 1 H, s) , 6. 88-6. 92 (1 H, m) , 7. 11-7. 17 (3H, m) , 7. 25— 7. 31 (lH, m)3.46 (1H, dd, J = 8.9, 14.3Hz), 3.50 (1H, dd, J = 3.7, 14.3Hz), 5.80 (1H, s), 6 88-6. 92 (1 H, m), 7.11-7. 17 (3H, m), 7.25—7.31 (lH, m)
, 7. 43-7. 50 (2H, m) , 8. 12-8. 16 (lH, m) , 7.43-7.50 (2H, m), 8.12-8.16 (lH, m)
FAB-MS (m/e) : 330 [M + H] + 実施例 101 FAB-MS (m / e): 330 [M + H] + Example 101
化合物例 1024の製造 Preparation of Compound Example 1024
ジァステレオマー a (Ar : Ph; R1: H; R2: H; R3: i -P r ; R4: H ; R5: 3-Ph-Ph ; n = 2) Jiasutereoma a (Ar: Ph; R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: 3-Ph-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 99 (3Η, d, J = 6. 6Hz) , 0. 9 9 (3H, d, J = 6. 6Hz) , 1. 75— 1. 85 ( 1 H, m) , 3. 10 (1H, d d, J =l 1. 7, 14. 1Hz) , 3. 45 ( 1 H, dd, J = 2 . 1, 14. 1Hz) , 3. 51-3. 58 (1H, m) , 5. 97 ( 1 H, s ) , 6. 78 (1H, d d, J = 2. 1, 6. 3Hz) , 7. 30— 7. 70 ( 11 H, m) , 8. 10-8. 20 ( 1 H, m) X HNMR (CDC 1 3) δ : 0. 99 (3Η, d, J = 6. 6Hz), 0. 9 9 (3H, d, J = 6. 6Hz), 1. 75- 1. 85 (1 H , m), 3.10 (1H, dd, J = 1.17, 14.1 Hz), 3.45 (1H, dd, J = 2.1, 14.1 Hz), 3.51-3. 58 (1H, m), 5.97 (1H, s), 6.78 (1H, dd, J = 2.1, 6.3Hz), 7.30—7.70 (11H, m), 8. 10-8. 20 (1 H, m)
FAB-MS (m/e) : 392 [M + H] + FAB-MS (m / e): 392 [M + H] +
ジァステレオマー b (Ar : Ph ; R1: H ; R2 : H ; R3 : ί -P r ; R4 : H ; R5 : 3-Ph-Ph ; n = 2) XHNMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 9Hz) , 1. 0 1 (3H, d, J = 6. 5Hz) , 1. 95— 2. 03 ( 1 H, m) , 2. 98 -3. 18 (IH, m) , 3. 45- 3. 56 (2H, m) , 5. 92 (IH, s) , 6. 96- 6. 99 (IH, dd, J = 3. 6, 5. 5Hz) , 7. 28 -7. 60 (11 H, m) , 8. 16 (1H, dd, J = 3. 5, 5. 5Hz)Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: ί -P r; R 4: H; R 5: 3-Ph-Ph; n = 2) X HNMR (CDC 1 3) δ : 0. 97 (3H, d, J = 6. 9Hz), 1. 0 1 (3H, d, J = 6. 5Hz), 1. 95- 2. 03 (1 H , M), 2.98-3.18 (IH, m), 3.45-3.56 (2H, m), 5.92 (IH, s), 6.96-6.99 (IH, dd , J = 3.6, 5.5 Hz), 7.28-7.60 (11 H, m), 8.16 (1H, dd, J = 3.5, 5.5 Hz)
FAB-MS (m/e) : 392 [M + H] + 実施例 102 FAB-MS (m / e): 392 [M + H] + Example 102
化合物例 1026の製造 Preparation of Compound Example 1026
ジァステレオマー a (Ar r Ph j R^ H j R^ H j R3 一 P r ; R4: H ; R5 : 4-C l -Ph ; n = 2) Jiasutereoma a (Ar r Ph j R ^ H j R ^ H j R 3 one P r; R 4: H; R 5: 4-C l -Ph; n = 2)
iHNMR (CDC 13) d : 0. 98 (3H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 73— 1. 88 ( 1 H, m) , 3. 05 (IH, d d, J =l 1. 6, 14. 5Hz) , 3. 45 ( 1 H, d d, J = 2 . 3, 14. 5Hz) , 3. 49 ( 1 H, d d d, J = 2, 3, 4. 9, 11. 6Hz) , 5. 91 (1H, s) , 6. 60-6. 70 ( 1 H, m) , 7. 31 -7. 42 (6H, m) , 8. 12— 8. 17 (lH, m) iHNMR (CDC 1 3) d: 0. 98 (3H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 73- 1. 88 (1 H, m), 3.05 (IH, dd, J = l 1.6, 14.5Hz), 3.45 (1H, dd, J = 2.3, 14.5Hz), 3.49 (1H, ddd, J = 2, 3, 4.9, 11.6 Hz), 5.91 (1H, s), 6.60-6.70 (1H, m), 7.31-7.42 (6H, m), 8.12—8.17 (lH, m)
FAB-MS (m/e) : 350/352 [M + H] + FAB-MS (m / e): 350/352 [M + H] +
ジァステレオマ一 b Ge stereomer b
(Ar : Ph ; Rx : H ; R2 H ; R3 : i -P r ; R4 : H ; R5 : 4-C 1 - Ph ; n=2) (Ar: Ph; R x : H; R 2 H; R 3 : i -P r; R 4 : H; R 5 : 4-C 1-Ph; n = 2)
iHNMR (CDC 13) δ : 0 95 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 88 -2. 00 (IH, m) , 3. 00 (IH, d d d, J = 3. 5, 5. 6, 8. 9Hz) , 3. 41 ( 1 H, d d, JJ == 88.. 99,, 1144.. 33HHzz)) ,, 33.. 44 ( 1 H, d d, J = 3. 5, 14. 3H z) , 5. 83 (1H, s) , 6. 90-6. 94 (IH, m) , 7. 26 (2 H, d, J = 8. 6Hz) , 7. 34 (2H, d, J = 8. 6Hz) , 7. 46 -7. 53 (2H, m) , 8. 14-8. 18 (lH, m) iHNMR (CDC 1 3) δ: . 0 95 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 88 -2 00 (IH, m) , 3.00 (IH, ddd, J = 3.5, 5.6, 8.9 Hz), 3.41 (1H, dd, JJ == 88..99 ,, 1144 .. 33HHzz)), 33..44 (1H, dd, J = 3.5, 14.3Hz), 5.83 (1H, s), 6.90-6.94 (IH, m), 7.26 (2H , d, J = 8.6 Hz), 7.34 (2H, d, J = 8.6 Hz), 7.46 -7.53 (2H, m), 8.14-8.18 (lH, m)
FAB-MS (m/e) : 350/352 [M + H] + 実施例 103 FAB-MS (m / e): 350/352 [M + H] + Example 103
化合物例 1083の製造 Preparation of Compound Example 1083
ジァステレオマー a (Ar : P h; R 1: H; R 2 H; R: i -P r ; R4 : H ; R5: 3-Me-4-Me-Ph; n=2) Jiasutereoma a (Ar: P h; R 1: H; R 2 H; R: i -P r; R 4: H; R 5: 3-Me-4-Me-Ph; n = 2)
XHNMR (CDC 13) d : 0. 98 (3H, d, J = 6. 9Hz) , 0. 9 8 (3H, d, J = 6. 9Hz) , 1. 72 - 1. 90 (1H, m) , 1. 95 (3H, s) , 2. 30 (3H, s) , 3. 06 (1H, dd, J =l 1. 4, 14. 3Hz) , 3. 44 ( 1 H, dd, J = 2. 2, 14. 3Hz) , 3. 5 3 (1 H, dd d, J = 2, 2, 4. 9, 1 1. Hz) , 6. 09 ( 1 H, s ) , 6. 60- 6. 64 (1H, m) , 7. 16— 7. 26 (2H, m) , 7. 31 -7. 42 (2H, m) , 7. 46 (1H, d, J = 7. 7Hz) , 8. 1 2-8. 16 (1H, m) X HNMR (CDC 1 3) d : 0. 98 (3H, d, J = 6. 9Hz), 0. 9 8 (3H, d, J = 6. 9Hz), 1. 72 - 1. 90 (1H, m), 1.95 (3H, s), 2.30 (3H, s), 3.06 (1H, dd, J = l1.4, 14.3Hz), 3.44 (1H, dd, J = 2.2, 14.3 Hz), 3.53 (1 H, dd d, J = 2, 2, 4. 9, 11.1 Hz), 6.09 (1 H, s), 6. 60- 6.64 (1H, m), 7.16—7.26 (2H, m), 7.31 -7.42 (2H, m), 7.46 (1H, d, J = 7.7Hz ), 8. 1 2-8. 16 (1H, m)
FAB-MS (m/e) : 344 [M + H] + FAB-MS (m / e): 344 [M + H] +
ジァステレオマー b (Ar Ph j R^ H j R^ H j R3 : i一 P r ; R4 : H ; R5: 3-Me-4-Me-Ph; n = 2) Diastereomer b (Ar Ph j R ^ H j R ^ H j R 3 : i-Pr; R 4 : H; R 5 : 3-Me-4-Me-Ph; n = 2)
HNMR (CDC 13) δ : 0. 95 (3H, d 6. 8Hz) , 0. 9 HNMR (CDC 1 3) δ: 0. 95 (3H, d 6. 8Hz), 0. 9
8 (3H, d, J = 6. 8Hz) , 1. 96 (3H, s) , 1. 96-2. 1 1 (1 H, m) , 2. 29 (3H, s) , 2. 77 - 2. 90 ( 1 H, m) , 3. 56— 3. 68 (2H, m) , 6. 04 (1H, s) , 6. 69 - 6. 73 (1 H, m) , 7. 19-7. 26 (3H, m) , 7. 38— 7. 5 1 (2H, m) , 8. 08-8. 12 (1H, m) 8 (3H, d, J = 6.8 Hz), 1.96 (3H, s), 1.96-2.1 1 (1 H, m), 2.29 (3H, s), 2.77- 2.90 (1H, m), 3.56—3.68 (2H, m), 6.04 (1H, s), 6.69-6.73 (1H, m), 7.19- 7.26 (3H, m), 7.38—7.51 (2H, m), 8.08-8.12 (1H, m)
FAB-MS (m/e) : 344 [M + H] + 実施例 104 FAB-MS (m / e): 344 [M + H] + Example 104
化合物例 1084の製造 Preparation of Compound Example 1084
ジァステレオマー a (Ar ^h j R^ H i R' r H j R3 i -P r ; R4 : H ; R5: 2— Me— 4— Me— Ph; n = 2)Jiasutereoma a (Ar ^ hj R ^ H i R 'r H j R 3 i -P r; R 4: H; R 5: 2- Me- 4- Me- Ph; n = 2)
NMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 76 - 1. 83 (lH, m) , 2. 03 (3H, s) , 2. 37 (3H, s) , 3. 05 ( 1 H, d d, J =l 1. 5, 14. 3Hz) , 3. 45 ( 1 H, d d, J = 2. 1, 14. 3Hz) , 3. 5 2 (IH, ddd, J = 2, 1, 4. 5, 11. 5Hz) , 5. 99 ( 1 H, s ) , 6. 63- 6. 66 (lH, m) , 7. 02 ( 1 H, s) 7. 14 (IH , d, J = 8. 0Hz) , 7. 31— 7. 42 (2H, m) 7 45 (IH, d , J = 8. 0Hz) , 8. 12-8. 15 (lH, m) NMR (CDC 1 3) δ: 0. 98 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6.8 Hz), 1.76-1.83 (lH, m), 2.03 (3H, s), 2.37 (3H, s), 3.05 (1H , Dd, J = l 1.5, 14.3Hz), 3.45 (1H, dd, J = 2.1, 14.3Hz), 3.52 (IH, ddd, J = 2, 1, 4.5, 11.5Hz), 5.99 (1H, s), 6.63-6.66 (lH, m), 7.02 (1H, s) 7.14 (IH, d, J = 8.0Hz), 7.31-7.42 (2H, m) 745 (IH, d, J = 8.0Hz), 8.12-8.15 (lH, m)
FAB-MS (mZe) : 344 [M + H] + FAB-MS (mZe): 344 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i— P r ; R4 : H ; R5 : 2 -Me -4 -Me -Ph; n=2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i- P r; R 4: H; R 5: 2 -Me -4 -Me -Ph; n = 2)
^NMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 8Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 98-2. 10 (lH, m) , 2. 20 5 (3H, s) , 2. 36 (3H, s) , 2. 76 - 2. 83 ( 1 H, m) , 3 . 55 - 3. 59 (2H, m) , 5. 95 (IH, s) , 6. 71-6. 75 ( 1H, m) , 7. 03 (IH, s) , 7. 12 (1 H, d, J = 8. 2Hz) , 7. 38-7. 47 (2H, m) , 7. 44 ( 1 H, d, J = 8. 2Hz) , 8 . 08— 8. 12 (IH, m) ^ NMR (CDC 1 3) δ :. 0. 94 (3H, d, J = 6. 8Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 98-2 10 (lH, m), 2.205 (3H, s), 2.36 (3H, s), 2.76-2.83 (1H, m), 3.55-3.59 (2H, m), 5 95 (IH, s), 6.71-6.75 (1H, m), 7.03 (IH, s), 7.12 (1H, d, J = 8.2Hz), 7.38- 7.47 (2H, m), 7.44 (1H, d, J = 8.2Hz), 8.08—8.12 (IH, m)
FAB -MS (m/e) : 344 [M + H] + 実施例 105 FAB-MS (m / e): 344 [M + H] + Example 105
化合物例 1085の製造 Preparation of Compound Example 1085
ジァステレオマー a (Ar : Ph; R1 : H; R2: H; R3: i一 P r ; R4: H ; R5: 2- e-5-Me-Ph; n=2) Jiasutereoma a (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4: H; R 5: 2- e-5-Me-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 99 (3H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 9Hz) , 1. 77 - 1. 90 (lH, m) , 2. 02 (3H, s) , 2. 40 (3H, s) , 3. 05 ( 1 H, dd, J =l 1. 5, 14. 4Hz) , 3. 47 ( 1 H, d d, J = 2. 2, 14. 4Hz) , 3. 5 3 (IH, d d d, J = 2, 2, 4. 9, 11. 5Hz) , 6. 00 ( 1 H, s ) , 6. 61-6. 65 (IH, m) , 7. 07 (1H, d, J = 1. 1Hz) , 7. 07 (1H, d, J = 1. lHz) , 7. 32-7. 42 (2H, m) , 7. 37 (1 H, d, J = 1. 1Hz) , 8. 12-8. 16 (1H, m) XHNMR (CDC 1 3) δ: 0. 99 (3H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 9Hz), 1. 77 - 1. 90 (lH, m ), 2.02 (3H, s), 2.40 (3H, s), 3.05 (1H, dd, J = l 1.5,14.4Hz), 3.47 (1H, dd, J = 2.2, 14.4Hz), 3.53 (IH, ddd, J = 2, 2, 4. 9, 11.5Hz), 6.00 (1H, s), 6.61-6 . 65 (IH, m), 7.07 (1H, d, J = 1.1 Hz) , 7.07 (1H, d, J = 1. lHz), 7.32-7.42 (2H, m), 7.37 (1H, d, J = 1.1 Hz), 8.12-8 . 16 (1H, m)
FAB-MS (m/e) : 344 [M + H] + FAB-MS (m / e): 344 [M + H] +
ジァステレオマー b (Ar i Ph j R^ H j R^ H i R3 : i一 P r ; R4 : H ; R5: 2-Me-5-Me-Ph; n=2) Diastereomer b (Ar i Ph j R ^ H j R ^ H i R 3 : i- 1 Pr; R 4 : H; R 5 : 2-Me-5-Me-Ph; n = 2)
^NMR (CDC 13) δ ·· 0. 96 (3H, d, J = 6. 9Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 2. 00— 2. 12 ( 1 H, m) , 2. 03 (3H, s) , 2. 39 (3H, s) , 2. 81 ( 1 H, d d d, J = 3. 8, 6. 0, 8. 4Hz) , 3. 56 ( 1 H, d d, J = 8. 4, 14. 1Hz) , 3. 59 (1H, dd, J = 3. 8, 14. 1Hz) , 5. 95 ( 1 H, s) , 6. 70-6. 74 ( 1 H, m) , 7. 08 (2H, s) , 7. 36 (1H, s ) , 7. 38 -7. 48 (2H, m) , 8. 08-8. 12 ( 1 H, m) ^ NMR (CDC 1 3) δ ·· 0. 96 (3H, d, J = 6. 9Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 2. 00- 2. 12 (1 H, m), 2.03 (3H, s), 2.39 (3H, s), 2.81 (1H, ddd, J = 3.8, 6.0, 8.4 Hz), 3.56 (1H, dd, J = 8.4, 14.1Hz), 3.59 (1H, dd, J = 3.8, 14.1Hz), 5.95 (1H, s), 6.70- 6.74 (1H, m), 7.08 (2H, s), 7.36 (1H, s), 7.38-7.48 (2H, m), 8.08-8.12 (1 H, m)
FAB-MS (m/e) : 344 [M + H] + 実施例 106 FAB-MS (m / e): 344 [M + H] + Example 106
化合物例 1087の製造 Preparation of Compound Example 1087
ジァステレオマー a (Ar : P h; R1: H; R2: H; R3: i— P r ; R4: H ; R5: 3-Me-4-Me-Ph; n=2) Jiasutereoma a (Ar: P h; R 1: H; R 2: H; R 3: i- P r; R 4: H; R 5: 3-Me-4-Me-Ph; n = 2)
^NMR (CDC 13) (5 : 0. 97 (3H, d, J = 6. 9Hz) , 0. 9 7 (3H, d, J = 6. 9Hz) , 1. 75 - 1. 89 (1H, m) , 2. 29 (3H, s) , 2. 30 (3H, s) , 3. 07 ( 1 H, d d, J =l 1. 4, 14. 3Hz) , 3. 42 ( 1 H, dd, J = 2. 2, 14. 3Hz) , 3. 5 1 (1H, ddd, J = 2, 2, 4. 9, 11. 4Hz) , 5. 78 (1H, s ) , 6. 73 - 6. 77 ( 1 H, m) , 7. 11-7. 21 (3H, m) , 7. 33 - 7. 40 (2H, m) , 8. 11 -8. 15 (1H, m) ^ NMR (CDC 1 3) ( 5: 0. 97 (3H, d, J = 6. 9Hz), 0. 9 7 (3H, d, J = 6. 9Hz), 1. 75 - 1. 89 (1H , m), 2.29 (3H, s), 2.30 (3H, s), 3.07 (1 H, dd, J = l 1.4, 14.3 Hz), 3.42 (1 H, s) dd, J = 2.2, 14.3 Hz), 3.51 (1H, ddd, J = 2, 2, 4.9, 11.4 Hz), 5.78 (1H, s), 6.73- 6.77 (1H, m), 7.11-7.21 (3H, m), 7.33-7.40 (2H, m), 8.11-8.15 (1H, m)
FAB-MS (m/e) : 344 [M + H] + FAB-MS (m / e): 344 [M + H] +
ジァステレオマ一 b (Ar : P ; R1 : H ; R2 : H ; R3: i - P r ; 4 : H ; R5: 3— Me - 4一 Me— Ph; n = 2) Jiasutereoma one b (Ar: P; R 1 : H; R 2: H; R 3: i - P r; 4: H; R 5: 3- Me - 4 one Me- Ph; n = 2)
^NMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 93— 2. 06 (lH, m) , 2. 26 (3H, s) , 2. 28 (3H, s) , 2. 96 ( 1 H, ddd, J = 3. 6, ^ NMR (CDC 1 3) δ : 0. 95 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6.8 Hz), 1.93-2.06 (lH, m), 2.26 (3H, s), 2.28 (3H, s), 2.96 (1H , Ddd, J = 3.6,
5. 8, 9. 0Hz) , 3. 45 ( 1 H, d d, J = 9. 0, 14. 5Hz) , 3. 51 (IH, d d, J = 3. 6, 14. 5Hz) , 5. 76 (1H, s) ,5.8, 9.0Hz), 3.45 (1H, dd, J = 9.0, 14.5Hz), 3.51 (IH, dd, J = 3.6, 14.5Hz), 5. 76 (1H, s),
6. 91-6. 94 (1H, m) , 7. 04 ( 1 H, dd, J一 1. 6, 7. 8 Hz) , 7. 10 (1H, s) , 7. 14 ( 1 H, d, J = 7. 8Hz) , 7. 42-7. 49 (2H, m) , 8. 12— 8. 16 ( 1 H, m) 6.91-6.94 (1H, m), 7.04 (1H, dd, J1.6, 7.8 Hz), 7.10 (1H, s), 7.14 (1H, d, J = 7.8Hz), 7.42-7.49 (2H, m), 8.12—8.16 (1H, m)
FAB-MS (mZe) : 344 [M + H] + 実施例 107 FAB-MS (mZe): 344 [M + H] + Example 107
化合物例 1090の製造 Preparation of Compound Example 1090
ジァステレオマー a (Ar i Ph j R^ H j R' i H j R3 : i - P r ; R4 : H ; R5: 3 -F, 4-Me-Ph; n=2) Diastereomer a (Ar i Ph j R ^ H j R 'i H j R 3 : i-P r; R 4 : H; R 5 : 3 -F, 4-Me-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 70— 1. 90 (lH, m) , 2. 31 (3H, s) , 3. 05 (IH, dd, J =l 1. 4, 14. 4Hz) , 3. 4 3 (1H, d d, J = 2. 3, 14. 4Hz) , 3. 47 - 3. 55 ( 1 H, m ) , 5. 88 (1H, s) , 6. 71-6. 74 ( 1 H, m) , 7. 03 (1 H , dd, J = 1. 5, 7. 7Hz) , 7. 11 ( 1 H, d d, J = l. 5, 10 . 6Hz) , 7. 20 (IH, dd, J = 7. 7, 10. 6Hz) , 7. 37— 7. 41 (2H, m) , 8. 12-8. 16 (lH, m) XHNMR (CDC 1 3) δ: 0. 97 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 70- 1. 90 (lH, m ), 2.31 (3H, s), 3.05 (IH, dd, J = l 1.4, 14.4Hz), 3.43 (1H, dd, J = 2.3, 14.4Hz) , 3.47-3.55 (1H, m), 5.88 (1H, s), 6.71-6.74 (1H, m), 7.03 (1H, dd, J = 1 5, 7.7 Hz), 7.11 (1H, dd, J = l. 5, 10.6Hz), 7.20 (IH, dd, J = 7.7, 10.6Hz), 7.37 — 7.41 (2H, m), 8.12-8. 16 (lH, m)
FAB-MS (m/e) : 348 [M + H] + FAB-MS (m / e): 348 [M + H] +
ジァステレオマー b (Ar r Ph j R^ H j R^ H i R3 : i一 P r ; R4 : H ; R5: 3— F, 4-Me-Ph; n=2) Diastereomer b (Ar r Ph j R ^ H j R ^ H i R 3 : i-Pr; R 4 : H; R 5 : 3-F, 4-Me-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 7Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 90— 2. 11 ( 1 H, m) , 2. 28 (3H, s) , 2. 97 - 3. 05 ( 1 H, m) , 3. 35— 3. 50 (2H, m) , 5. 81 (IH, s) , 6. 92 - 7. 00 (2H, m) , 7. 03 (1 H, d, J = 10. 8Hz) , 7. 16 (1H, dd, J = 7. 7, 10. 8H z) , 7. 49-7. 51 (2H, m) , 8. 14-8. 17 ( 1 H, m) FAB-MS (mZe) : 348 [M + H] + 実施例 108 XHNMR (CDC 1 3) δ: 0. 95 (3H, d, J = 6. 7Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 90- 2. 11 (1 H, m), 2.28 (3H, s), 2.97-3.05 (1H, m), 3.35—3.50 (2H, m), 5.81 (IH, s), 6. 92-7.00 (2H, m), 7.03 (1H, d, J = 10.8 Hz), 7.16 (1H, dd, J = 7.7, 10.8H z), 7.49-7.51 (2H, m), 8.14-8.17 (1H, m) FAB-MS (mZe): 348 [M + H] + Example 108
化合物例 1092の製造 Preparation of Compound Example 1092
ジァステレオマ一 a (Ar : Ph; R1: H; R2: H; R3: i - P r ; R4: H ; R5: 4-F-3-Me-Ph; n = 2) Jiasutereoma one a (Ar: Ph; R 1 : H; R 2: H; R 3: i - P r; R 4: H; R 5: 4-F-3-Me-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 97 (3H, d, J = 6. 9Hz) , 0. 9 7 (3H, d, J = 6. 9Hz) , 1. 75— 1. 85 ( 1 H, m) , 2. 30 (3H, d, J f_H= 1 - 9Hz) , 3. 06 ( 1 H, d d, J = 11. 3, 1 4. 2Hz) , 3. 44 (1H, dd, J = 2. 3, 14. 2Hz) , 3. 49 (1H, ddd, J = 2, 3, 4. 9, 11. 3Hz) , 5. 83 (1H, s) , 6. 69 -6. 74 (1H, m) , 7. 01-7. 08 (lH, m) , 7. 1 5-7. 22 (2H, m) , 7. 36-7. 43 (2H, m) , 8. 12— 8. 16 (1 H, m) X HNMR (CDC 1 3) δ : 0. 97 (3H, d, J = 6. 9Hz), 0. 9 7 (3H, d, J = 6. 9Hz), 1. 75- 1. 85 (1 H , m), 2.30 (3H, d, J f _H = 1-9 Hz), 3.06 (1 H, dd, J = 11.3, 14.2 Hz), 3.44 (1H, dd, J = 2.3, 14.2 Hz), 3.49 (1H, ddd, J = 2, 3, 4. 9, 11.3 Hz), 5.83 (1H, s), 6.69 -6.74 (1H, m), 7.01-7.08 (lH, m), 7.15-7.22 (2H, m), 7.36-7.43 (2H, m), 8.12— 8.16 (1 H, m)
FAB-MS (m/e) : 348 [M + H] + FAB-MS (m / e): 348 [M + H] +
ジァステレオマ一 b (Ar : P j R^ H i R' H j R3 : i— P r ; R4 : H ; R5 : 4-F-3-Me-Ph; n = 2) Jiasutereoma one b (Ar: P j R ^ H i R 'H j R 3: i- P r; R 4: H; R 5: 4-F-3-Me-Ph; n = 2)
^NMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 88-2. 03 ( 1 H, m) , 2. 27 (3H, d, J p_H= 1. 9Hz) , 2. 97 ( 1 H, ddd, J = 3. 5, 5 . 7, 9. 0Hz) , 3. 43 ( 1 H, dd, J = 9. 0, 14. 3Hz) , 3 . 47 (1 H, d d, J = 3. 5, 14. 3Hz) , 5. 79 ( 1 H, s) , 6 . 89-6. 91 (lH, m) , 6. 91— 7. 03 (lH, m) , 7. 07— 7. 17 (2H, m) , 7. 45-7. 51 (2H, m) , 8. 13— 8. 17 (1H, m) ^ NMR (CDC 1 3) ( 5:. 0. 95 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 88-2 03 (1 H, m), 2.27 (3H, d, J p_H = 1.9 Hz), 2.97 (1 H, ddd, J = 3.5, 5.7, 9.0 Hz), 3.43 (1 H, dd, J = 9.0, 14.3 Hz), 3.47 (1 H, dd, J = 3.5, 14.3 Hz), 5.79 (1 H, s), 6.89-6 .91 (lH, m), 6.91-7.03 (lH, m), 7.07-7.17 (2H, m), 7.45-7.51 (2H, m), 8.13 — 8. 17 (1H, m)
FAB-MS (mZe) : 348 [M + H] + 実施例 109 " 化合物例 1094の製造 FAB-MS (mZe): 348 [M + H] + Example 109 " Preparation of Compound Example 1094
ジァステレオマー a (Ar : Ph i R^ H j R^ H j R3 : i -P r ; R4: H ; R5: 3 -F, 4-F-Ph; n = 2) Jiasutereoma a (Ar: Ph i R ^ H j R ^ H j R 3: i -P r; R 4: H; R 5: 3 -F, 4-F-Ph; n = 2)
XHNMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 4Hz) , 0. 9 9 (3H, d, J = 6. 4Hz) , 1. 75 - 1. 85 (lH, m) , 3. 04 (1H, dd, J =l 1. 9, 14. 2Hz) , 3. 46 (1H, dd, J = 2 . 2, 14. 2Hz) , 3. 45-3. 54 ( 1 H, m) , 5. 93 (1H, s ) , 6. 65 - 6. 90 (2H, m) , 7. 05 - 7. 20 (2H, m) , 7. 39— 7. 48 (2H, m) , 8. 13-8. 20 ( 1 H, m) X HNMR (CDC 1 3) δ : 0. 98 (3H, d, J = 6. 4Hz), 0. 9 9 (3H, d, J = 6. 4Hz), 1. 75 - 1. 85 (lH, m), 3.04 (1H, dd, J = l 1.9, 14.2Hz), 3.46 (1H, dd, J = 2.2, 14.2Hz), 3.45-3.54 ( 1 H, m), 5.93 (1H, s), 6.65-6.90 (2H, m), 7.05-7.20 (2H, m), 7.39-7.48 (2H , M), 8.13-8.20 (1 H, m)
FAB-MS (m/e) : 352 [M + H] + FAB-MS (m / e): 352 [M + H] +
ジァステレオマー b (Ar Ph j R^ H j R' r H j R3 : i -P r ; R4 : H ; R5 : 3 -F, 4-F-Ph; n=2) Diastereomer b (Ar Ph j R ^ H j R 'r H j R 3 : i -P r; R 4 : H; R 5 : 3 -F, 4-F-Ph; n = 2)
HNMR (CDC 1 J 6 : 0. 96 (3H, d, J = 6. 9Hz) 0 HNMR (CDC 1 J 6: 0.96 (3H, d, J = 6.9 Hz) 0
1 (3H, d, J = 6. 4Hz) , 1. 88— 1. 99 (1H, m) , 2. 97 一 3. 05 (1H, m) , 3. 35— 3. 42 (2H, m) , 5. 83 (1H, s) , 6. 95-7. 01 (2H, m) , 7. 05 - 7. 25 (2H, m) , 7 . 50 - 7. 55 (2H, m) , 8. 15-8. 20 ( 1 H, m) 1 (3H, d, J = 6.4Hz), 1.88—1.99 (1H, m), 2.97 1 3.05 (1H, m), 3.35—3.42 (2H, m ), 5.83 (1H, s), 6.95-7.01 (2H, m), 7.05-7.25 (2H, m), 7.50-7.55 (2H, m), 8. 15-8. 20 (1 H, m)
FAB-MS (m/e) : 352 [M + H] + 実施例 110 FAB-MS (m / e): 352 [M + H] + Example 110
化合物例 1095の製造 Preparation of Compound Example 1095
ジァステレオマー a (Ar i Ph j R^ H ^' r H ^3 : i -P r ; R4 : H ; R5: 3 -F, 4-C 1 -Ph ; n = 2) Diastereomer a (Ar i Ph j R ^ H ^ 'r H ^ 3 : i -P r; R 4 : H; R 5 : 3 -F, 4-C 1 -Ph; n = 2)
XHNMR (CDC 13) δ : 0. 99 (3H, d, J = 7. 0Hz) , 1. 0 0 (3H, d, J = 7. 0Hz) , 1. 75 - 1. 90 (1H, m) , 3. 05 (1H, dd, J =l 1. 4, 14. 8Hz) , 3. 46 (1H, dd, 1=2 . 3, 14. 8Hz) , 3. 45 -3. 52 (lH, m) , 5. 96 ( 1 H, s ) , 6. 70 - 6. 77 (1H, m) , 7. 11 (1H, d, J = 8. 3Hz) , 7. 30 (1H, dd, J = l. 7, 8. 3Hz) , 7. 39-7. 47 (3 H, m) , 8. 14-8. 17 (1H, m) XHNMR (CDC 1 3) δ: 0. 99 (3H, d, J = 7. 0Hz), 1. 0 0 (3H, d, J = 7. 0Hz), 1. 75 - 1. 90 (1H, m ), 3.05 (1H, dd, J = l 1.4, 14.8 Hz), 3.46 (1H, dd, 1 = 2.3, 14.8 Hz), 3.45-3.52 (lH , M), 5.96 (1H, s), 6.70-6.77 (1H, m), 7.11 (1H, d, J = 8.3Hz), 7.30 (1H, dd, J = l. 7, 8.3 Hz), 7.39-7.47 (3 H, m), 8.14-8.17 (1H, m)
FAB-MS (mZe) : 368/370 [M + H] + FAB-MS (mZe): 368/370 [M + H] +
ジァステレオマー b (Ar i Ph j R^ H j R^ H j R3 i -P r ; R4 : HDiastereomer b (Ar i Ph j R ^ H j R ^ H j R 3 i -P r; R 4 : H
; R5: 3 -F, 4-C 1 -Ph ; n = 2) R 5 : 3 -F, 4-C 1 -Ph; n = 2)
'HNMR (CDC 13) (5 : 0. 96 (3H, d, J = 7. 0Hz) , 0 1 (3H, d, J = 7. 0Hz) , 1. 90- 2. 00 ( 1 H, m) , 2. 98 -3. 07 (lH, m) , 3. 35— 3. 42 (2H, m) , 5. 80 (1H, s) , 6. 92 - 6. 97 (1 H, m) , 6. 99 ( 1 H, d, J = 8. 2Hz ) , 7. 18 (1H, d, J = 10. 2Hz) , 7. 36 ( 1 H, d d, J = 8 . 2, 10. 2Hz) , 7. 48-7. 55 (2H, m) , 8. 12-8. 20'HNMR (CDC 1 3 ) (5: 0.96 (3H, d, J = 7.0 Hz), 0 1 (3H, d, J = 7.0 Hz), 1.90-2.00 (1 H, m), 2.98 -3.07 (lH, m), 3.35—3.42 (2H, m), 5.80 (1H, s), 6.92-6.97 (1H, m ), 6.99 (1H, d, J = 8.2Hz), 7.18 (1H, d, J = 10.2Hz), 7.36 (1H, dd, J = 8.2, 10. 2Hz), 7.48-7.55 (2H, m), 8.12-8.20
(1H, m) (1H, m)
FAB-MS (mZe) : 368/370 [M + H] + 実施例 1 1 1 FAB-MS (mZe): 368/370 [M + H] + Example 1 1 1
化合物例 1096の製造 Preparation of Compound Example 1096
ジァステレオマー a (Ar : P j R^ H j R' i H j R3 : i— P r ; R4 : HJiasutereoma a (Ar: P j R ^ H j R 'i H j R 3: i- P r; R 4: H
; R5: 3-C l -4-C l -Ph ; n=2) R 5 : 3-Cl-4-Cl-Ph; n = 2)
iHNMR (CDC 13) δ : 0. 98 (3H, d, J = 6. 9Hz) , 0. 9 iHNMR (CDC 1 3) δ: 0. 98 (3H, d, J = 6. 9Hz), 0. 9
9 (3H, d, J = 6. 6Hz) , 1. 77— 1. 84 (1H, m) , 3. 04 (1H, d d, J = 1 1. 8, 14. 6Hz) , 3. 46 ( 1 H, dd, J = 29 (3H, d, J = 6.6 Hz), 1.77—1.84 (1H, m), 3.04 (1H, dd, J = 1 1.8, 14.6 Hz), 3.46 ( 1 H, dd, J = 2
. 3, 14. 6Hz) , 3. 45-3. 52 (lH, m) , 5. 94 ( 1 H, s3, 14.6 Hz), 3.45-3.52 (lH, m), 5.94 (1H, s
) , 6. 69 - 6. 73 (1H, m) , 7. 22 (1H, dd, J = l. 9, 8), 6.69-6.73 (1H, m), 7.22 (1H, dd, J = l. 9, 8
. 3Hz) , 7. 39 - 7. 44 (2H, m) , 7. 48 ( 1 H, d, J = 8.3 Hz), 7.39-7.44 (2H, m), 7.48 (1H, d, J = 8.
3Hz) , 7. 56 (1 H, d, J = l. 9Hz) , 8. 14-8. 18 (1 H , m) 3Hz), 7.56 (1H, d, J = l. 9Hz), 8.14-8.18 (1H, m)
FAB-MS (m/e) : 384/386 [M + H] + FAB-MS (m / e): 384/386 [M + H] +
ジァステレオマー b (Ar : P ; Rx : H ; R2 : H ; R3 : i -P r ; R4 : H ; R5: 3-C 1 -4-C 1 -Ph ; n = 2)Jiasutereoma b (Ar: P; R x : H; R 2: H; R 3: i -P r; R 4: H; R 5: 3-C 1 -4-C 1 -Ph; n = 2)
NMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 9Hz) , 1. 0 1 (3H, d, J =6. 8Hz) 1. 87- 1. 98 (1 H, m) , 3. 02 (IH, d dd, J = 3. 6, 5 5, 8. 9Hz) , 3. 38 ( 1 H, d d, J = 8. 9, 14. 4Hz) , 3 43 (IH, dd, J = 3. 6, 14. 4H z) , 5. 83 (IH, s) , 6 93 - 6. 97 (1H, m) , 7. 08 (1 H, dd, J = 1. 9, 8. 3Hz) , 7. 42 ( 1 H, d, J = 8. 3Hz) , 7. 48 (IH, d, J = l. 9Hz) , 7. 50 - 7. 56 (2H, m) , 8. 1 5— 8. 1 9 (IH, m) NMR (CDC 1 3) δ: 0. 96 (3H, d, J = 6. 9Hz), 1. 0 1 (3H, d, J = 6.8 Hz) 1.87- 1.98 (1 H, m), 3.02 (IH, d dd, J = 3.6, 5, 5, 8.9 Hz), 3 38 (1H, dd, J = 8.9, 14.4Hz), 343 (IH, dd, J = 3.6, 14.4Hz), 5.83 (IH, s), 693- 6.97 (1H, m), 7.08 (1H, dd, J = 1.9, 8.3Hz), 7.42 (1H, d, J = 8.3Hz), 7.48 (IH , d, J = l. 9Hz), 7.50-7.56 (2H, m), 8.15-5.19 (IH, m)
FAB-MS (m/e) : 384/386 [M + H] + 実施例 112 FAB-MS (m / e): 384/386 [M + H] + Example 112
化合物例 1 100の製造 Preparation of Compound Example 1 100
ジァステレオマー a (Ar : P h; R 1: H; R 2: H; R 3 i -P r ; R4 : H ; R5: 3 -Me, 4-Me O-P h; n = 2)Jiasutereoma a (Ar: P h; R 1: H; R 2: H; R 3 i -P r; R 4: H; R 5: 3 -Me, 4-Me OP h; n = 2)
NMR (CDC 13) (5 : 0. 97 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, 1 = 6. 8Hz) , 1. 75 - 1. 85 ( 1 H, m) , 2. 24 (3H, s) , 3. 07 (1H, dd, J = 1 1. , 14. 3Hz) , 3. 4 2 (IH, dd, J =2. 2, 14. 3 Hz) , 3. 50 ( 1 H, d d d, 2. 2, 4. 8, 1 1. 4Hz) , 3. 86 (3H, s) , 5. 75 ( 1 H, s) , 6. 75 - 6. 78 (1H, m) , 6. 86 (IH, d, J = 8. 4Hz) , 7 . 14 (1H, d, J = 2. 0Hz) , 7. 19 ( 1 H, d d, J = 2. 0, 8 • 4Hz) , 7. 35- 7. 38 (2H, m) , 8. 1 1— 8. 14 ( 1 H, m ) NMR (CDC 1 3) (5 : 0. 97 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, 1 = 6. 8Hz), 1. 75 - 1. 85 (1 H , m), 2.24 (3H, s), 3.07 (1H, dd, J = 1 1., 14.3 Hz), 3.42 (IH, dd, J = 2.2, 14.3 Hz), 3.50 (1H, ddd, 2.2, 4.8, 11.4Hz), 3.86 (3H, s), 5.75 (1H, s), 6.75-6 78 (1H, m), 6.86 (IH, d, J = 8.4 Hz), 7.14 (1H, d, J = 2.0 Hz), 7.19 (1H, dd, J = 2 0, 8 • 4Hz), 7.35-7.38 (2H, m), 8.11-8.14 (1H, m)
FAB-MS (m/e) 360 [M + H] + FAB-MS (m / e) 360 [M + H] +
ジァステレオマー b (A r P ; R 1 : H ; R 2 : H ; R 3 : i -P r ; R4 : H ; R5 : 3— Me, 4-MeO-Ph; n = 2) Jiasutereoma b (A r P; R 1 : H; R 2: H; R 3: i -P r; R 4: H; R 5: 3- Me, 4-MeO-Ph; n = 2)
XHNMR (CDC 13) 6 : 0. 95 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 8Hz) , 1. 95 - 2. 05 (lH, m) , 2. 2 1 (3H, s) , 2. 92-3. 01 (lH, m) , 3. 40— 3. 55 (2H, m) , 3. 84 (3H, s) , 5. 76 (IH, s) , 6. 82 ( 1 H, d, J =8. 3Hz) , 6. 92 - 6. 96 (1 H, m) , 7. 08 (IH, d d, J =2. 3, 8. 3Hz) , 7. 12 (1H, d, J = 2. 3Hz) , 7. 44一 7. 47 (2H, m) , 8. 12— 8. 15 (lH, m) X HNMR (CDC 1 3) 6 : 0. 95 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 8Hz), 1. 95 - 2. 05 (lH, m), 2.21 (3H, s), 2.92-3. 01 (lH, m), 3.40—3.55 (2H, m), 3.84 (3H, s), 5. 76 (IH, s), 6.82 (1 H, d, J = 8.3 Hz), 6.92-6.96 (1 H, m), 7.08 (IH, dd, J = 2.3, 8.3 Hz), 7.12 (1H, d, J = 2 3 Hz), 7.44-1 7.47 (2H, m), 8.12—8.15 (lH, m)
FAB-MS (m/e) : 360 [M + H] + 実施例 113 FAB-MS (m / e): 360 [M + H] + Example 113
化合物例 1 103の製造 Preparation of Compound Example 1 103
ジァステレオマー a (Ar i Ph i R^ H j R^ H i R3 i -P r ; R' H ; R5: 3-MeO-4-MeO-Ph; n=2) Jiasutereoma a (Ar i Ph i R ^ H j R ^ H i R 3 i -P r; R 'H; R 5: 3-MeO-4-MeO-Ph; n = 2)
^NMR (CDC 13) (5 : 0. 99 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 72 - 1. 87 (lH, m) , 3. 08 (1H, dd, J = 11. 4, 14. 3Hz) , 3. 44 (IH, dd, J = 2 . 0, 14. 3Hz) , 3. 52 ( 1 H, ddd, J = 2, 0, 5. 0, 11. 4Hz) , 3. 86 (3H, s) , 3. 93 (3H, s) , 5. 80 (1H, s ) , 6. 75 - 6. 80 (1H, m) , 6. 89 ( 1 H, d, J = 1. 7Hz) , 6. 93 (IH, d, J = 8. 3Hz) , 7. 00 (1H, dd, J = 1. 7 , 8. 3Hz) , 7. 38-7. 43 (2H, m) , 8. 13— 8. 16 (1 H , m) ^ NMR (CDC 1 3) ( 5: 0. 99 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 72 - 1. 87 (lH , M), 3.08 (1H, dd, J = 11.4, 14.3Hz), 3.44 (IH, dd, J = 2.0, 14.3Hz), 3.52 (1H, ddd , J = 2, 0, 5.0, 11.4 Hz), 3.86 (3H, s), 3.93 (3H, s), 5.80 (1H, s), 6.75-6.80 (1H, m), 6.89 (1H, d, J = 1.7Hz), 6.93 (IH, d, J = 8.3Hz), 7.00 (1H, dd, J = 1.7) , 8.3 Hz), 7.38-7.43 (2H, m), 8.13—8.16 (1H, m)
FAB-MS (m/e) : 376 [M + H] + FAB-MS (m / e): 376 [M + H] +
ジァステレオマー b (Ar i Ph i R H j R^ H j R3 : i -P r ; R4 : H ; R5: 3-MeO-4-MeO-Ph; n=2) Diastereomer b (Ar i Ph i RH j R ^ H j R 3 : i -P r; R 4 : H; R 5 : 3-MeO-4-MeO-Ph; n = 2)
^NMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 8Hz) , 1. 0 0 (3H, d, J = 6. 8Hz) , 1. 92- 2. 07 ( 1 H, m) , 2. 98 ^ NMR (CDC 1 3) δ : 0. 96 (3H, d, J = 6. 8Hz), 1. 0 0 (3H, d, J = 6. 8Hz), 1. 92- 2. 07 (1 H , m), 2.98
(1H, ddd, J = 3. 5, 5 7, 9. 2Hz) , 3. 45 ( 1 H, d d, J = 9. 2, 14. 4Hz) , 3 51 (IH, d d, J = 3. 5, 14. 4H z) , 3. 84 (3H, s) , 3 90 (3H, s) , 5. 80 (IH, s) , 6. 83 (1H, d d, J = l. 7, 8. 4Hz) , 6. 87 (1H, d, J = 8. 4Hz) , 6. 92 (1H, d, J = 1. 7Hz) , 6. 93 - 6. 97 ( IH, m) , 7. 44-7. 51 (2H, m) , 8. 13-8. 17 ( 1 H, m FAB-MS (m/e) : 376 [M + H] + 実施例 114 (1H, ddd, J = 3.5, 5, 7, 9.2Hz), 3.45 (1H, dd, J = 9.2, 14.4.4Hz), 351 (IH, dd, J = 3. 5, 14.4Hz), 3.84 (3H, s), 390 (3H, s), 5.80 (IH, s), 6.83 (1H, dd, J = l. 7, 8. 4Hz), 6.87 (1H, d, J = 8.4Hz), 6.92 (1H, d, J = 1.7Hz), 6.93-6.97 (IH, m), 7.44- 7.51 (2H, m), 8.13-8.17 (1H, m FAB-MS (m / e): 376 [M + H] + Example 114
化合物例 1 104の製造 Preparation of Compound Example 1 104
ジァステレオマー a (Ar : P h; R 1: H; R 2: H; 3: i一 P r ; R 4 : H ; R5: 3, 4-Me t hy 1 e ne d i oxy 1 -Ph; n = 2) R 1 : H; R 2 : H; 3 : i-Pr; R 4 : H; R 5 : 3, 4-Methylene dioxy 1 -Ph; n = 2)
^NMR (CDC 13) (5 : 0. 97 (3H, d, J = 6. 8Hz) , 0. 9 7 (3H, d, J = 6. 8Hz) , 1. 73— 1. 88 ( 1 H, m) , 3. 05 (1H, d d, J = 11. 4, 14. 3Hz) , 3. 43 ( 1 H, d d, J = 2 . 2, 14. 3Hz) , 3. 46- 3. 53 (lH, m) , 5. 79 ( 1 H, s ) , 6. 00 (2H, s) , 6. 78-6. 83 (lH, m) , 6. 84—6. 90 (3H, m) , 7. 37— 7. 41 (2H, m) , 8. 11— 8. 15 (1 H, m) ^ NMR (CDC 1 3) ( 5: 0. 97 (3H, d, J = 6. 8Hz), 0. 9 7 (3H, d, J = 6. 8Hz), 1. 73- 1. 88 (1 H, m), 3.05 (1H, dd, J = 11.4, 14.3Hz), 3.43 (1H, dd, J = 2.2, 14.3Hz), 3.46-3. 53 (lH, m), 5.79 (1H, s), 6.00 (2H, s), 6.78-6.83 (lH, m), 6.84—6.90 (3H, m ), 7.37—7.41 (2H, m), 8.11—8.15 (1H, m)
FAB-MS (m/e) : 360 [M + H] + FAB-MS (m / e): 360 [M + H] +
ジァステレオマー b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i一 P r ; R4: H ; R5 : 3, 4-Me t hy 1 e n e d i oxy 1 - Ph; n = 2) Jiasutereoma b (Ar: Ph; R 1 : H; R 2: H; R 3: i one P r; R 4: H; R 5: 3, 4-Me t hy 1 enedi oxy 1 - Ph; n = 2 )
XHNMR (CDC 13) 6 : 0. 96 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 8Hz) , 1. 90— 2. 04 (lH, m) , 2. 98 (1H, d d d, J = 3. 5, 5. 7, 9. 1Hz) , 3. 43 ( 1 H, dd, J = 9. 1, 14. 1Hz) , 3. 48. (1H, d d, J = 3. 5, 14. 1 H z) , 5. 77 (1H, s) , 5. 99 (2H, s) , 6. 75 ( 1 H, dd, J = 1. 5, 8. 0Hz) , 6. 80 ( 1 H, d, J = 8. 0Hz) , 6. 87 (1H, d, J = l. 5Hz) , 6. 95 - 6. 99 (lH, m) , 7. 46- 7. 51 (2H, m) , 8. 12-8. 16 (1H, m) X HNMR (CDC 1 3) 6 : 0. 96 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 8Hz), 1. 90- 2. 04 (lH, m), 2.98 (1H, ddd, J = 3.5, 5. 7, 9.1Hz), 3.43 (1H, dd, J = 9.1, 14.1Hz), 3.48. (1H, dd, J = 3.5, 14.1 Hz), 5.77 (1H, s), 5.99 (2H, s), 6.75 (1H, dd, J = 1.5 , 8.0Hz), 6.80 (1H, d, J = 8.0Hz), 6.87 (1H, d, J = l. 5Hz), 6.95-6.99 (lH, m), 7.46-7.51 (2H, m), 8.12-8.16 (1H, m)
FAB-MS (m/e) : 360 [M+H] + 実施例 115 FAB-MS (m / e): 360 [M + H] + Example 115
化合物例 1105の製造 ジァステレオマ一 a (Ar : Ph j R^ H i R' i H j R3 : i一 P r ; R4 : H ; R5 : 3, 4-E t hy l en e d i oxy l -Ph ; n = 2) Preparation of Compound Example 1105 Jiasutereoma one a (Ar: Ph j R ^ H i R 'i H j R 3: i one P r; R 4: H; R 5: 3, 4-E t hy l en edi oxy l -Ph; n = 2)
XHNMR (CDC 13) δ 0. 97 (3H, d, J = 6. 9Hz) , 0. 9 XHNMR (CDC 1 3) δ 0. 97 (3H, d, J = 6. 9Hz), 0. 9
7 (3H, d, J = 6. 9Hz) , 1. 70— 1. 85 ( 1 H, m) , 3. 05 (1H, dd, J = 1 1. 5, 14. 3Hz) , 3. 41 (IH, dd, J = 27 (3H, d, J = 6.9 Hz), 1.70—1.85 (1 H, m), 3.05 (1H, dd, J = 1 1.5, 14.3 Hz), 3.41 (IH, dd, J = 2
. 2, 14. 3Hz) , 3. 50 ( 1 H, ddd, J = 2, 2, 4. 8, 1 1. 5Hz) , 4. 30 (4H, s) , 5. 76 ( 1 H, s) , 6. 78-6. 83 (lH, m) , 6. 83 (IH, dd, J = l. 9, 8. 3Hz) , 7. 35— 7. 42 (2H, m) , 8. 1 1— 8. 1 5 ( 1 H, m) .2, 14.3Hz), 3.50 (1H, ddd, J = 2, 2, 4.8, 11.5Hz), 4.30 (4H, s), 5.76 (1H, s) ), 6.78-6.83 (lH, m), 6.83 (IH, dd, J = l. 9, 8.3Hz), 7.35—7.42 (2H, m), 8.1 1—8.15 (1H, m)
FAB-MS i /e) : 374 [M + H] + FAB-MS i / e): 374 [M + H] +
ジァステレオマ一 b (Ar : Ph ; R1 : H ; R2 : H ; R3 : i— P r ; R4 : H ; R5 : 3, 4-E t hy l en e d i oxy l -P ; n=2) Jiasutereoma one b (Ar: Ph; R 1 : H; R 2: H; R 3: i- P r; R 4: H; R 5: 3, 4-E t hy l en edi oxy l -P; n = 2)
XHNMR (CDC 13) 5 : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 XHNMR (CDC 1 3) 5: 0. 95 (3H, d, J = 6. 8Hz), 0. 9
8 (3H, d, J = 6. 8Hz) , 1. 90 - 2. 03 (lH, m) , 2. 97 (IH, dd d, J = 3. 5, 5. 7, 9. OHz) , 3. 42 (IH, dd,8 (3H, d, J = 6.8 Hz), 1.90-2.03 (lH, m), 2.97 (IH, dd d, J = 3.5, 5. 7, 9. OHz), 3.42 (IH, dd,
J = 9. 0, 14. 3Hz) , 3. 48 ( 1 H, dd, J = 3. 5, 14. 3H z) , 4. 27 (4H, s) , 5. 73 ( 1 H, s) , 6. 77 ( 1 H, dd, J = 1. 9, 8. 6Hz) , 6. 85 ( 1 H, d, J = 8. 6Hz) , 6. 87 (IH, d, J = 1. 9Hz) , 7. 42-7. 51 (2H, m) , 8. 1 1一 8. 1 5 (1 H, m) J = 9.0, 14.3Hz), 3.48 (1H, dd, J = 3.5, 14.3Hz), 4.27 (4H, s), 5.73 (1H, s) , 6.77 (1H, dd, J = 1.9, 8.6Hz), 6.85 (1H, d, J = 8.6Hz), 6.87 (IH, d, J = 1.9Hz) ), 7.42-7.51 (2H, m), 8.11 1 8.15 (1H, m)
FAB-MS (mZe) : 374 [M + H] + 実施例 1 16 FAB-MS (mZe): 374 [M + H] + Example 1 16
化合物例 1 1 1 1の製造 (Ai^ Pt^ R^ H R H R3 : i— P r ; R4 : H ; R5: 3— F, 4一 Me〇— P h ; n = 2) Preparation of compound examples 1 1 1 1 (Ai ^ Pt ^ R ^ HRHR 3: i- P r; R 4: H; R 5: 3- F, 4 one Me_〇- P h; n = 2)
XHNMR (CDC 13) (3 : 0. 96 (3H, d, J = 6. 8Hz) , 0. 9 9 (3H, d, J = 6. 7Hz) , 1. 90- 2. 00 (IH, m) , 2. 98 一 3. 04 (IH, m) , 3. 35— 3. 50 (2H, m) , 3. 90 (3H, s) , 5. 79 (IH, s) , 6. 92 - 6. 97 (2H, m) , 7. 15 ( 1 H, d d, J = l. 3、 1 1. 6Hz) , 7. 48-7. 51 (2H, m) , 8 . 14-8. 17 (1H, m) X HNMR (CDC 1 3) ( 3: 0. 96 (3H, d, J = 6. 8Hz), 0. 9 9 (3H, d, J = 6. 7Hz), 1. 90- 2. 00 (IH , m), 2.98-3.04 (IH, m), 3.35-3.50 (2H, m), 3.90 (3H, s), 5.79 (IH, s), 6. 92-6.97 (2H, m), 7.15 (1 H, dd, J = l. 3, 11.6 Hz), 7.48-7.51 (2H, m), 8.14-8.17 (1H, m)
FAB-MS (m/e) : 364 [M + H] + 実施例 117 FAB-MS (m / e): 364 [M + H] + Example 117
化合物例 1 1 58の製造 (3—イソプロピル一 7—ニトロ一 5—フエニル 2, 3 , 4, 5, ーテトラヒドロ— 1 一 1 Λ 6, 4一べンゾチアゼピン— 1, 1ージ オン) Preparation of compound examples 1 1 58 (3-isopropyl one 7-nitro-one 5-phenyl 2, 3, 4, 5, Tetorahidoro - 1 one 1 lambda 6, 4 one base Nzochiazepin - 1, -1-di-one)
2—プロモー 5—二卜ロー安息香酸 10. 0 g (40. 6 mm o 1 ) に塩化チ ォニル 6. 0mlを加え、 120°Cで 3時間加熱還流した後、 反応溶液を減圧下 に濃縮した。 残查にァ二ソールを 30. 0ml加え、 続いて塩ィ匕アルミニウム 6 . 8 g (48. 7mmo 1 ) を加え、 160でで 5時間加熱攪拌した。 飽和塩 化アンモニゥム水溶液を添加し、 酢酸ェチルエステルで反応溶液を抽出し、 有機 層を飽和炭酸水素ナトリウム水溶液、 飽和食塩水溶液で洗浄後、 硫酸マグネシゥ ムで乾燥後、 減圧下に濃縮し、 得られた残渣をシリカゲルカラムクロマトグラフ ィー (へキサン:酢酸ェチルエステル =4: 1) により精製し、 (2—プロモー 5—二トロフエニル) (フエニル) メタノン 8. 3 g (収率: 6 1%) を淡黄色 油状物として得た。 2-Promo 5-2-nitrobenzoic acid 10.0 mL (40.6 mmo 1) was added with 6.0 mL of thionyl chloride, and the mixture was heated under reflux at 120 ° C for 3 hours. The reaction solution was concentrated under reduced pressure. did. 30.0 ml of ethanol was added to the residue, followed by 6.8 g (48.7 mmo 1) of aluminum salt, and the mixture was heated and stirred at 160 for 5 hours. A saturated aqueous solution of ammonium chloride was added, the reaction solution was extracted with ethyl acetate, and the organic layer was washed with a saturated aqueous solution of sodium bicarbonate and a saturated aqueous solution of sodium chloride, dried over magnesium sulfate, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (hexane: ethyl acetate = 4: 1), and 8.3 g (yield: 61%) of (2-promo-5-ditrophenyl) (phenyl) methanone was obtained. Obtained as a yellow oil.
^NMR (CDC 13) δ : 3. 90 (3Η, s) , 6. 97 (2H, d, J =9. 0Hz) , 7. 77 (2H, d, J = 9. 0Hz) , 7. 85 (2H, d , J = 9. 5Hz) , 8. 16— 8. 20 (2H, m) ^ NMR (CDC 1 3) δ : 3. 90 (3Η, s), 6. 97 (. 2H, d, J = 9 0Hz), 7. 77 (2H, d, J = 9. 0Hz), 7. 85 (2H, d, J = 9.5Hz), 8.16—8.20 (2H, m)
FAB-MS ( /e) : 336/338 [M + H] + FAB-MS (/ e): 336/338 [M + H] +
工程 1で得られた (2—プロモー 5—二トロフエニル) (フエニル) メタノン 2. 60 g (7. 73mmo 1 ) のジメチルホルムアミド溶液 ( 30 m 1 ) に、 t—ブチル N— { 2—メチル— 1一 (スルファニルメチル) プロピル } 力ルバ メート 3. 0 g (1 1. 4mmo 1) 炭酸カリウム 2. 09 g (15. lmmo 1) を加え、 120°Cで 2時間加熱攪拌した。 飽和塩化アンモニゥム水溶液を添 加し、 酢酸ェチルエステルで反応溶液を抽出し、 有機層を 1 0%硫酸水素力リウ ム溶液、 飽和食塩水溶液で洗浄後、 硫酸マグネシウムで乾燥後、 減圧下に濃縮し た。 残査に 4N塩化水素 1, 4一ジォキサン溶液 (10. 0ml) を加え室温で 一晩攪拌し、 反応溶液を減圧下に濃縮した。 残渣に 2, 6—ルチジン (20. 0 ml) を加え、 濃塩酸 (10. 0 ml) を添加した。 添加終了後、 混合物を 3時 間共沸させ、 減圧留去した。 残渣を飽和炭酸水素ナトリウム水溶液で回収し、 溶 液を酢酸ェチルで 2回抽出した。 集めた抽出物を飽和食塩水溶液で洗浄し、 硫酸 マグネシウムで乾燥後、 減圧下に濃縮した。 得られた残渣をシリカゲルカラムク 口マトグラフィー (へキサン:酢酸ェチルエステル =4 : 1) により精製し、 3 一イソプロピル一7—ニトロ— 5—フエ二ルー 2, 3—ジヒドロー 1, 4一ベン ゾチアゼピン 1. 65 g (収率: 60%) を淡黄色油状物として得た。 To a solution of 2.60 g (7.73 mmo 1) of (2-promo 5-ditrophenyl) methanone obtained in step 1 in dimethylformamide (30 m 1), t-butyl N— {2-methyl— To the mixture was added 3.0 g (11.4 mmo 1) of potassium carbonate and 2.09 g (15. lmmo 1) of potassium carbonate, and the mixture was heated with stirring at 120 ° C. for 2 hours. A saturated aqueous solution of ammonium chloride was added, and the reaction solution was extracted with ethyl acetate.The organic layer was washed with a 10% aqueous solution of hydrogen hydrogen sulfate and a saturated aqueous solution of sodium chloride, dried over magnesium sulfate, and concentrated under reduced pressure. Was. To the residue was added a 4N hydrogen chloride 1,4-dioxane solution (10.0 ml), the mixture was stirred at room temperature overnight, and the reaction solution was concentrated under reduced pressure. 2,6-Lutidine (20.0 ml) was added to the residue, and concentrated hydrochloric acid (10.0 ml) was added. After the addition was completed, the mixture was azeotroped for 3 hours and distilled under reduced pressure. The residue was collected with a saturated aqueous sodium hydrogen carbonate solution, and the solution was extracted twice with ethyl acetate. The collected extract was washed with a saturated saline solution, dried over magnesium sulfate, and concentrated under reduced pressure. The obtained residue was purified by silica gel column chromatography (hexane: ethyl acetate = 4: 1) to give 3-isopropyl-17-nitro-5-phenyl-2,3-dihydro-1,4-benzothiazepine. 1.65 g (yield: 60%) was obtained as a pale yellow oil.
^NMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 7Hz) , 1. 0 5 (3H, d, J = 6. 6Hz) , 1. 95-2. 10 ( 1 H, m) , 2. 89 -2. 99 (1H, m) , 3. 42 (1H, dd, J =l 1. 2, 12. 0Hz ) , 3. 62 (1H, d d, J = 3. 7, 1 1. 2Hz) , 3. 84 (3H, s ) , 6. 90 (2H, d, J = 8. 9Hz) , 7. 52 (2H, d, J = 8. 9 Hz) , 7. 76 (1H, d, J = 8. 3Hz) , 7. 99 (1H, d, J = 2 . 6Hz) , 8. 19 (1H, dd, J = 2. 6, 8. 3Hz) ^ NMR (CDC 1 3) δ :. 0. 95 (3H, d, J = 6. 7Hz), 1. 0 5 (3H, d, J = 6. 6Hz), 1. 95-2 10 (1 H , M), 2.89-2.99 (1H, m), 3.42 (1H, dd, J = l 1.2, 12.0 Hz), 3.62 (1H, dd, J = 3.7 , 11.2Hz), 3.84 (3H, s), 6.90 (2H, d, J = 8.9Hz), 7.52 (2H, d, J = 8.9Hz), 7.76 (1H, d, J = 8.3Hz), 7.99 (1H, d, J = 2.6Hz), 8.19 (1H, dd, J = 2.6, 8.3Hz)
FAB-MS (m/e) : 357 [M + H] + FAB-MS (m / e): 357 [M + H] +
工程 2で得られた 3—イソプロピル— 7—二トロー 5—フエ二ルー 2, 3—ジ ヒドロ— 1, 4一べンゾチアゼピン 1. 58 g (4. 43 mm o 1 ) のテトラ ヒドロフラン溶液 10m 1にトリフルォロ酢酸 (3. 0ml) 溶液を加え、 30 %過酸化水素水 (1. 0ml) を約 O :の温度で添加した。 添加終了後、 混合物 を室温で一晩攪拌し、 10 %チォ硫酸ナトリゥム水溶液、 飽和炭酸水素ナトリウ ム水溶液で処理し、 酢酸ェチルで 2回抽出し、 抽出物を集めて飽和食塩水溶液で 洗浄し、 乾燥し、 減圧下に濃縮した。 残渣に 4N塩化水素 1, 4一ジォキサン溶 液 (10. 0ml) を加え、 亜鉛粉末 288 mgを約 O の温度で徐々に添加し た。 混合液を一晩室温で攪拌し、 飽和炭酸水素ナトリウム水溶液で処理し、 セラ イトで濾過した。 酢酸ェチルで 2回抽出し、 抽出物を集めて飽和食塩水溶液で洗 浄し、 硫酸マグネシウムで乾燥し、 減圧下に濃縮た。 得られた残渣をシリカゲル 3-58- (2-nitro) -5-phenyl-2,3-dihydro-1,4-monobenzothiazepine obtained in Step 2 1.58 g (4.43 mmo 1) of tetrahydrofuran solution in 10 m 1 To this was added a solution of trifluoroacetic acid (3.0 ml), and 30% aqueous hydrogen peroxide (1.0 ml) was added at a temperature of about O :. After the addition was completed, the mixture was stirred at room temperature overnight, treated with a 10% aqueous sodium thiosulfate solution and a saturated aqueous sodium hydrogen carbonate solution, extracted twice with ethyl acetate, and the combined extracts were washed with a saturated aqueous sodium chloride solution. Dry and concentrate under reduced pressure. 4N Hydrogen chloride 1,4-dioxane solution (10.0 ml) was added to the residue, and 288 mg of zinc powder was gradually added at a temperature of about O. The mixture was stirred overnight at room temperature, treated with saturated aqueous sodium bicarbonate and filtered through celite. The mixture was extracted twice with ethyl acetate, and the combined extracts were washed with a saturated saline solution, dried over magnesium sulfate, and concentrated under reduced pressure. Silica gel obtained residue
- (へキサン:酢酸ェチルエステル = 1 : 1) により精 製し、 3—イソプロピル一 7—ニトロ一 5—フエニル 2, 3, 4, 5, ーテトラ ヒドロー Iff一 1 λ 6, 4—ベンゾチアゼピン— 1, 1—ジオンのジァステレオ マー a (408mg) および b (53 Omg) を淡黄色の油状生成物として得た ジァステレオマー a -(Hexane: ethyl acetate = 1: 1) Papermaking, 3-isopropyl one 7-nitro-one 5-phenyl 2, 3, 4, 5, Tetora Hidoro Iff one 1 lambda 6, 4-benzothiazepine - 1, 1-dione Jiasutereo mer a (408 mg) and b (53 Omg) as pale yellow oily product diastereomer a
XHNMR (CDC 13) δ : 0. 95 (3Η, d, J =6. 8Hz) , 0. 9 5 (3H, d, J = 6. 8Hz) , 1. 70- 1. 81 (1 H, m) , 3. 01 (1H, d d, J = 11. 4, 14. OHz) , 3. 37 (1H, dd, J = 2 . 3, 14. OHz) , 3. 44 (1H, d d d, J = 2. 2, 5. 0, 11. 4Hz) , 3. 85 (3H, s) , 5. 71 (1H, s) , 5. 88 ( 1 H, d , J = 2. 3Hz) , 6. 51 (1H, d d, J = 2. 3, 8. 4Hz) , 6. 94 (2H, d, J = 8. 6Hz) , 7. 31 (1H, d, J = 8. 6Hz) , 7. 88 (1H, d, J = 8. 4Hz) X HNMR (CDC 1 3) δ : 0. 95 (. 3Η, d, J = 6 8Hz), 0. 9 5 (3H, d, J = 6. 8Hz), 1. 70- 1. 81 (1 H , m), 3.01 (1H, dd, J = 11.4, 14.OHz), 3.37 (1H, dd, J = 2.3, 14.OHz), 3.44 (1H, ddd, J = 2.2, 5.0, 11.4 Hz), 3.85 (3H, s), 5.71 (1H, s), 5.88 (1H, d, J = 2.3 Hz), 6 .51 (1H, dd, J = 2.3, 8.4 Hz), 6.94 (2H, d, J = 8.6 Hz), 7.31 (1H, d, J = 8.6 Hz), 7. 88 (1H, d, J = 8.4Hz)
FAB-MS (m/e) : 361 [M + H] + FAB-MS (m / e): 361 [M + H] +
ジァステレオマ一 b Ge stereomer b
XHNMR (CDC 13) 6 : 0. 93 (3H, d, J = 6. 8Hz) , 0. 9 7 (3H, d, J = 6. 7Hz) , 1. 85 - 1. 99 (1H, m) , 2. 90 —3. 00 (lH, m) , 3. 30 -3. 40 (2H, m) , 3. 82 (3H, s) , 5. 73 (1H, s) , 6. 08 ( 1 H, d, J = 2. 4Hz) , 6. 6 1 (1H, d d, J = 2. 4, 8. 5Hz) , 6. 90 (2H, d, J = 8. 6 Hz) , 7. 26 (1H, d, J = 8. 6Hz) , 7. 89 ( 1 H, d, J = 8 . 5Hz) X HNMR (CDC 1 3) 6 : 0. 93 (3H, d, J = 6. 8Hz), 0. 9 7 (3H, d, J = 6. 7Hz), 1. 85 - 1. 99 (1H, m), 2.90—3.00 (lH, m), 3.30-3.40 (2H, m), 3.82 (3H, s), 5.73 (1H, s), 6.08 (1H, d, J = 2.4Hz), 6.61 (1H, dd, J = 2.4, 8.5Hz), 6.90 (2H, d, J = 8.6Hz), 7 26 (1H, d, J = 8.6Hz), 7.89 (1H, d, J = 8.5Hz)
FAB-MS (m/e) : 361 [M + H] + FAB-MS (m / e): 361 [M + H] +
以下に実施例 117と同様にして得た化合物の物理定数を示す。 実施例 118 The physical constants of the compound obtained in the same manner as in Example 117 are shown below. Example 118
化合物例 1157の製造 Preparation of Compound Example 1157
ジァステレオマー a (A r : 7-N02-Ph ; R1 : H ; R2 : H ; R3 P r ; R 4 : H ; R5: 4-MeO-Ph ; n=0) ^NMR (CDC 13) δ : 0. 95 (3H, d, J = 6. 8Hz) , 0. 9 6 (3H, d, J = 6. 8Hz) , 1. 65 - 1. 78 (lH, m) , 2. 49 (IH, dd, J = 10. 3, 13. 9Hz) , 2. 98 (1H, dd, J = 2 . 2, 13. 9Hz) , 3. 06 ( 1 H, m) , 3. 86 (3H, s) , 5. 4 8 (IH, s) , 6. 97 (2H, d, J = 8. 8Hz) , 7. 33 (2H, d , J = 8. 8Hz) , 7. 49 (1H, d, J = 2. 5Hz) , 7. 75 (1 H , d, J = 8. 4Hz) , 7. 92 (IH, dd, J = 2. 5, 8. 4Hz)Diastereomer a (A r: 7-N02-Ph; R 1 : H; R 2 : H; R 3 P r; R 4 : H; R 5 : 4-MeO-Ph; n = 0) ^ NMR (CDC 1 3) δ : 0. 95 (3H, d, J = 6. 8Hz), 0. 9 6 (3H, d, J = 6. 8Hz), 1. 65 - 1. 78 (lH, m), 2.49 (IH, dd, J = 10.3, 13.9 Hz), 2.98 (1H, dd, J = 2.2, 13.9 Hz), 3.06 (1 H, m) , 3.86 (3H, s), 5.48 (IH, s), 6.97 (2H, d, J = 8.8 Hz), 7.33 (2H, d, J = 8.8 Hz), 7.49 (1H, d, J = 2.5Hz), 7.75 (1H, d, J = 8.4Hz), 7.92 (IH, dd, J = 2.5, 8.4Hz)
FAB-MS (m/e) : 359 [M + H] + FAB-MS (m / e): 359 [M + H] +
ジァステレオマー b (A r : 7-N02-Ph; RX : H ; R2 : H ; : l ― P r ; R 4 : H ; R5 : 4-MeO-P ; n=0) Jiasutereoma b (A r: 7-N02 -Ph; R X: H; R 2: H;: l - P r; R 4: H; R 5: 4-MeO-P; n = 0)
aHNMR (CDC 13) (5 : 0. 93 (3H, d, J = 6. 7Hz) , 0. 9 5 (3H, d, J =6. 7Hz) , 1. 90— 2. 10 ( 1 H, m) , 2. 75 一 2. 85 (IH, m) , 2. 90- 3. 05 (2H, m) , 3. 84 (3H, s) , 5. 64 (IH, s) , 6. 93 (2H, d, J = 8. 8Hz) , 7. 2 5 (2H, d, J = 8. 8Hz) , 7. 58 (IH, d, J = 8. 5Hz) , 7 . 63 (IH, d, J = 2. 3Hz) , 7. 93 ( 1 H, dd, J = 2. 3, 8 . 5Hz) aHNMR (CDC 1 3) (5 :. 0. 93 (3H, d, J = 6. 7Hz), 0. 9 5 (3H, d, J = 6 7Hz), 1. 90- 2. 10 (1 H , m), 2.75-2.85 (IH, m), 2.90-3.05 (2H, m), 3.84 (3H, s), 5.64 (IH, s), 6. 93 (2H, d, J = 8.8 Hz), 7.25 (2H, d, J = 8.8 Hz), 7.58 (IH, d, J = 8.5 Hz), 7.63 (IH, d, J = 2.3Hz), 7.93 (1H, dd, J = 2.3, 8.5Hz)
FAB-MS (m/e) : 359 [M + H] + 実施例 119 FAB-MS (m / e): 359 [M + H] + Example 119
化合物例 1159の製造 Preparation of Compound Example 1159
ジァステレオマー a (Ar : 7— N3— Pt R^ H R^ H R3 : i -P r ; R4 : H ; R5: 4-MeO-Ph; n=2) Jiasutereoma a (Ar: 7- N3- Pt R ^ HR ^ HR 3: i -P r; R 4: H; R 5: 4-MeO-Ph; n = 2)
XHNMR (CDC 13) δ 0. 96 ( 3 H, d, J = 6. 8Hz) , 0. 9 6 (3H, d, J = 6. 8Hz) , 1. 70— 1. 82 ( 1 H, m) , 3. 05 (IH, d d, J = 11. 8, 14. OHz) , 3. 41 (1H, dd, J = 2 . 0, 14. OHz) , 3. 48 ( 1 H, ddd, J = 2. 0, 5. 0, 11. 8Hz) , 3. 85 (3H, s) , 5. 73 (1H, s) , 6. 31 (IH, d , J = 2. 4Hz) , 6. 96 (2H, d, J = 8. 5Hz) , 7. 03 (2H , d d, J = 2. 4, 8. 4Hz) , 7. 29 (2H, d, J = 8. 5Hz) , 8. 12 (1H, d, J = 8. 4Hz) XHNMR (CDC 1 3) δ 0. 96 (3 H, d, J = 6. 8Hz), 0. 9 6 (3H, d, J = 6. 8Hz), 1. 70- 1. 82 (1 H, m), 3.05 (IH, dd, J = 11.8, 14.OHz), 3.41 (1H, dd, J = 2.0, 14.OHz), 3.48 (1H, ddd, J = 2.0, 5.0, 11.8 Hz), 3.85 (3H, s), 5.73 (1H, s), 6.31 (IH, d, J = 2.4 Hz), 6. 96 (2H, d, J = 8.5Hz), 7.03 (2H , dd, J = 2.4, 8.4Hz), 7.29 (2H, d, J = 8.5Hz), 8.12 (1H, d, J = 8.4Hz)
FAB-MS (m/e) : 384 [M + H] + FAB-MS (m / e): 384 [M + H] +
ジァステレオマー b (Ar : T-NS-P i R^ H j R' i H j R3 : i一 P r ; R4 : H ; R5: 4-MeO-Ph; n=2) Jiasutereoma b (Ar: T-NS- P i R ^ H j R 'i H j R 3: i one P r; R 4: H; R 5: 4-MeO-Ph; n = 2)
:HNMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 7Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 90— 2. 05 (lH, m) , 2. 95 —3. 04 (lH, m) , 3. 35- 3. 50 (2H, m) , 3. 82 (3H, s) , 5. 76 (1H, s) , 6. 54 ( 1 H, d, J = 2. 2Hz) , 6. 9 2 (2H, d, J = 8. 6Hz) , 7. 09 ( 1 H, d d, J = 2. 2, 8. 5 Hz) , 7. 23 (2H, d, J = 8. 6Hz) , 8. 1 3 (1H, d, J = 8 . 5Hz) : HNMR (CDC 1 3) δ : 0. 94 (3H, d, J = 6. 7Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 90- 2. 05 (lH, m), 2.95 — 3.04 (lH, m), 3.35-3.50 (2H, m), 3.82 (3H, s), 5.76 (1H, s), 6.54 (1H, d, J = 2.2Hz), 6.92 (2H, d, J = 8.6Hz), 7.09 (1H, dd, J = 2.2, 8.5Hz), 7.23 (2H, d, J = 8.6Hz), 8.13 (1H, d, J = 8.5Hz)
FAB-MS (m/e) : 387 [M + H] + 実施例 120 FAB-MS (m / e): 387 [M + H] + Example 120
化合物例 1 164の製造 (Ar : y-MeO-P i R^ H ^^ H i R3 : i -P r ; R4 : H ; R5 : Ph ; n=0) Preparation of compound examples 1 164 (Ar: y-MeO -P i R ^ H ^^ H i R 3: i -P r; R 4: H; R 5: Ph; n = 0)
XHNMR (CDC 13) (5 : 0. 94 (3H, d, J = 6. 8Hz) , 0. 9 5 (3H, d, J = 6. 8Hz) , 1. 55— 1. 75 ( 1 H, m) , 2. 38 (1H, d d, J = 10. 8, 13. 8Hz) , 2. 84 (1H, dd, J = 2 . 1, 13. 8Hz) , 3. 00 - 3. 08 ( 1 H, m) , 3. 58 (3H, s ) , 5. 66 (1H, s) , 6. 14 ( 1 H, d, J = 2. 7Hz) , 6. 62 (1H, dd, J = 2. 7, 8. 3Hz) , 7. 25-7 45 (5H, m) , 7. 56 (1H, d, J = 8. 3Hz) X HNMR (CDC 1 3) ( 5: 0. 94 (3H, d, J = 6. 8Hz), 0. 9 5 (3H, d, J = 6. 8Hz), 1. 55- 1. 75 (1 H, m), 2.38 (1H, dd, J = 10.8, 13.8 Hz), 2.84 (1H, dd, J = 2.1, 13.8 Hz), 3.00-3.08 (1H, m), 3.58 (3H, s), 5.66 (1H, s), 6.14 (1H, d, J = 2.7Hz), 6.62 (1H, dd, J = 2.7, 8.3Hz), 7.25-745 (5H, m), 7.56 (1H, d, J = 8.3Hz)
FAB-MS (m/e) : 3 14 [M + H] + 実施例 121 FAB-MS (m / e): 3 14 [M + H] + Example 121
化合物例 1279の製造 Preparation of Compound Example 1279
ジァステレオマー a (Ar : 3, 4-Py r i dy l ; R^ H J R^ H J R3 : i -P r ; R4 : H ; R5 : Ph ; n = 0) Diastereomer a (Ar: 3, 4-Py ri dy l; R ^ HJR ^ HJR 3 : I -P r; R 4 : H; R 5 : Ph; n = 0)
iHNMR (CDC 13) δ : 0. 96 (3H, d, J = 6. 8Hz) , 0. 9 7 (3H, d, J = 6. 7Hz) , 1. 67— 1. 80 (1 H, m) , 2. 46 (1H, dd, J = 10. 5, 14. 1Hz) , 2. 98 ( 1 H, dd, J = 2 . 1, 14. 1 Hz) , 3. 10 ( 1 H, ddd, J = l. 9, 4. 9, 10. 5Hz) , 5. 54 (1H, s) , 6. 64 ( 1 H, d, J = 6. 0Hz) , 7 . 35 -7. 46 (5H, m) , 8. 21 ( 1 H, d, J = 6. OHz) , 8. 78 (1H, s) iHNMR (CDC 1 3) δ: 0. 96 (3H, d, J = 6. 8Hz), 0. 9 7 (3H, d, J = 6. 7Hz), 1. 67- 1. 80 (1 H, m), 2.46 (1H, dd, J = 10.5, 14.1 Hz), 2.98 (1H, dd, J = 2.1, 14.1 Hz), 3.10 (1H, ddd, J = l. 9, 4. 9, 10.5 Hz), 5.54 (1H, s), 6.64 (1 H, d, J = 6.0 Hz), 7.35 -7.46 ( 5H, m), 8.21 (1H, d, J = 6. OHz), 8.78 (1H, s)
FAB-MS (mZe) : 285 [M + H] + FAB-MS (mZe): 285 [M + H] +
ジァステレオマー b (Ar : 3, 4-Py r i dy l ; R1 : H ; R2 : H ; R3 : i -P r ; R4 : H ; R5 : Ph ; n=0) Jiasutereoma b (Ar: 3, 4- Py ri dy l; R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: Ph; n = 0)
XHNMR (CDC 13) δ : 0. 94 (3H, d, J = 6. 8Hz) , 0. 9 X HNMR (CDC 1 3) δ : 0. 94 (3H, d, J = 6. 8Hz), 0. 9
5 (3H, d, J = 6. 6Hz) , 2. 00-2. 12 ( 1 H, m) , 2. 75 -2. 85 (1H, m) , 2. 95— 3. 00 (2H, m) , 5. 64 (1H, s) , 6. 80 (1H, d, J = 5. 9Hz) , 7. 28- 7. 43 ( 5 H, m ) , 8. 23 (1H, d, J = 5. 9Hz) , 8. 66 ( 1 H, s) 5 (3H, d, J = 6.6Hz), 2.00-2.12 (1H, m), 2.75-2.85 (1H, m), 2.95—3.00 (2H, m), 5.64 (1H, s), 6.80 (1H, d, J = 5.9 Hz), 7.28-7.43 (5H, m), 8.23 (1H, d, J = 5.9Hz), 8.66 (1 H, s)
FAB-MS (m/e) : 285 [M + H] + 実施例 122 FAB-MS (m / e): 285 [M + H] + Example 122
化合物例 128 1の製造 Preparation of Compound Example 128 1
ジァステレオマー a (Ar : 3, 4-Py r i dy l ; R1 : H ; R2 : H ; R3 : i -P r ; R4 : H ; R5 : 4-PhCH2O-P ; n = 0) Jiasutereoma a (Ar: 3, 4- Py ri dy l; R 1: H; R 2: H; R 3: i -P r; R 4: H; R 5: 4-PhCH2O-P; n = 0)
XHNMR (CDC 13) (5 : 0. 95 (3H, d, J = 6. 9Hz) , 0. 9 XHNMR (CDC 1 3) (5 : 0. 95 (3H, d, J = 6. 9Hz), 0. 9
6 (3H, d, J = 6. 9Hz) , 1. 65— 1. 80 (lH, m) , 2. 45 (1H, dd, J = 10. 4, 14. OHz) , 2. 94 ( 1 H, dd, J = 26 (3H, d, J = 6.9 Hz), 1.65-1.80 (lH, m), 2.45 (1H, dd, J = 10.4, 14. OHz), 2.94 (1 H, dd, J = 2
. 0, 14. OHz) , 3. 10 (1H, ddd, J = 2. 0, 4. 9, 10. 4Hz) , 5. 10 (2H, s) , 5. 46 ( 1 H, s) , 6. 69 ( 1 H, d , J = 5. 9Hz) , 7. 04 (1H, d, J = 8. 7Hz) , 7. 29— 7. 47 (5H, m) , 7. 28 ( 1 H, d, J = 8. 7Hz) , 8. 22 (1H, d, J = 5. 9Hz) , 8. 76 (1H, s) 0, 14. OHz), 3.10 (1H, ddd, J = 2.0, 4.9, 10.4Hz), 5.10 (2H, s), 5.46 (1H, s), 6.69 (1H, d, J = 5.9Hz), 7.04 (1H, d, J = 8.7Hz), 7.29—7.47 (5H, m), 7.28 (1H , D, J = 8.7 Hz), 8.22 (1H, d, J = 5.9 Hz), 8.76 (1H, s)
FAB-MS (m/e) : 391 [M + H] + FAB-MS (m / e): 391 [M + H] +
ジァステレオマ一 b (Ar : 3, 4-Py r i dy 1 ; R 1 : H ; R 2 : H ; R 3 : i -P r ; R4 : H ; R5 : 4-PhCH20-Ph ; n=0) Diastereomer b (Ar: 3, 4-Py ri dy 1; R 1 : H; R 2 : H; R 3 : i-P r; R 4 : H; R 5 : 4-PhCH20-Ph; n = 0 )
XHNMR (CDC 13) d : 0. 95 (3H, d, J = 5. 7Hz) , 0. 9 XHNMR (CDC 1 3) d: 0. 95 (3H, d, J = 5. 7Hz), 0. 9
5 (3H, d, J = 5. 7Hz) , 1. 98— 2. 10 ( 1 H, m) , 2. 71 -2. 82 (lH, m) , 2. 95— 3. 00 (2H, m) , 5. 08 (2H, s) , 5. 58 (1H, s) , 6. 82 (1H, d, J = 5. 8Hz) , 7. 0 0 (2H, d, J = 8. 7Hz) , 7. 10 (2H, d, J = 8. 7Hz) , 7 . 25- 7. 43 (5H, m) , 8. 23 ( 1 H, d, J = 5. 8Hz) , 8.5 (3H, d, J = 5.7 Hz), 1.98—2.10 (1H, m), 2.71 -2.82 (lH, m), 2.95—3.00 (2H, m), 5.08 (2H, s), 5.58 (1H, s), 6.82 (1H, d, J = 5.8 Hz), 7.00 (2H, d, J = 8.7 Hz) ), 7.10 (2H, d, J = 8.7Hz), 7.25-7.43 (5H, m), 8.23 (1H, d, J = 5.8Hz), 8.
65 (1H, s) 65 (1H, s)
FAB-MS (m/e) : 39 1 [M + H] + 実施例 123 FAB-MS (m / e): 39 1 [M + H] + Example 123
化合物例 1288の製造 Preparation of Compound Example 1288
ジァステレオマー a (Ar : 2, 3-Py r i dy l ; R^ H j R' r H j R3 : i - P r ; R 4: H; R5 : P ; n= 0)Jiasutereoma a (Ar: 2, 3- Py ri dy l; R ^ H j R 'r H j R 3: i - P r; R 4: H; R 5: P; n = 0)
NMR (CDC 13) 6 : 0. 97 (3H, d, J = 6. 4Hz) , 0. 9 NMR (CDC 1 3) 6: 0. 97 (3H, d, J = 6. 4Hz), 0. 9
7 (3H, d, J = 6. 4Hz) , 1. 70- 1. 80 (1 H, m) , 2. 64 (1 H, dd, J = 9. 8, 14. 1Hz) , 3. 01 (1H, dd, J = 2.7 (3H, d, J = 6.4Hz), 1.70-1.80 (1H, m), 2.64 (1H, dd, J = 9.8, 14.1Hz), 3.01 (1H, dd, J = 2.
1, 14. 1Hz) , 3. 02-3. 12 (1H, m) , 5. 44 ( 1 H, s) , 6. 85 (1H, dd, J = 2. 0, 7. 9Hz) , 6. 95 (1H, d d, J =4. 8, 7. 9Hz) , 7. 30-7. 43 (5H, m) , 8. 29 (1H , dd, J = 2. 0, 4. 8Hz) 1, 14.1 Hz), 3.02-3.12 (1H, m), 5.44 (1H, s), 6.85 (1H, dd, J = 2.0, 7.9Hz), 6 95 (1H, dd, J = 4.8, 7.9Hz), 7.30-7.43 (5H, m), 8.29 (1H, dd, J = 2.0, 4.8Hz)
FAB-MS (m/e) : 285 [M + H] + FAB-MS (m / e): 285 [M + H] +
ジァステレオマー b (Ar : 2, 3-Py r i dy l ; R^ H J R' I H I R3 : i -P r ; R4 : H ; R5 : P h ; n= 0) Jiasutereoma b (Ar: 2, 3- Py ri dy l; R ^ HJR 'IHIR 3: i -P r; R 4: H; R 5: P h; n = 0)
XHNMR (CDC 13) 6 : 0. 94 (3H, d, J = 6. 2Hz) , 0. 9 6 (3H, d, J = 6. 6Hz) , 1. 86— 2. 01 ( 1 H, m) , 2. 80 —2. 90 (1H, m) , 3. 05-3. 11 (2H, m) , 5. 56 (1H, s) , 6. 92 - 6. 96 (2H, m) , 7. 28- 7. 42 (5H, m) , 8 . 28 - 8. 30 (1H, m) X HNMR (CDC 1 3) 6 : 0. 94 (3H, d, J = 6. 2Hz), 0. 9 6 (3H, d, J = 6. 6Hz), 1. 86- 2. 01 (1 H , m), 2.80 — 2.90 (1H, m), 3.05-3.11 (2H, m), 5.56 (1H, s), 6.92-6.96 (2H, m), 7.28-7 . 42 (5H, m), 8.28-8.30 (1H, m)
FAB-MS (m/e) : 285 [M + H] + 実施例 124 FAB-MS (m / e): 285 [M + H] + Example 124
化合物例 1291の製造 (Ar : 5, e-Py r i dy ^ R^. H R^ H ; R3: i - P r ; R4 : H ; R5 : Ph ; n=0) Preparation of compound examples 1291 (Ar: 5, e- Py ri dy ^ R ^ HR ^ H; R 3:. I - P r; R 4: H; R 5: Ph; n = 0)
^NMR (CDC 13) (5 : 0. 97 (3H, d, J = 6. 8Hz) , 0. 9 8 (3H, d, J = 6. 7Hz) , 1. 65- 1. 80 ( 1 H, m) , 2. 48 (1H, d d, J =l 0. 8, 14. 1Hz) , 2. 95— 3. 05 (2H, m ) , 5. 68 (1H, s) , 7. 02 ( 1 H, dd, J =4. 7, 7. 6Hz) , 7. 27 -7. 46 (5H, m) , 7. 88 ( 1 H, dd, J = 1. 7, 7. 7Hz) , 8. 32 (1H, dd, J = l. 7, 4. 8Hz) ^ NMR (CDC 1 3) ( 5: 0. 97 (3H, d, J = 6. 8Hz), 0. 9 8 (3H, d, J = 6. 7Hz), 1. 65- 1. 80 (1 H, m), 2.48 (1H, dd, J = l 0.8, 14.1 Hz), 2.95—3.05 (2H, m), 5.68 (1H, s), 7.02 (1H, dd, J = 4.7, 7.6Hz), 7.27 -7. 46 (5H, m), 7.88 (1H, dd, J = 1. 7, 7.7Hz), 8.32 (1H, dd, J = l. 7, 4.8Hz)
FAB-MS (mZe) : 285 [M + H] + 製剤化例 FAB-MS (mZe): 285 [M + H] + Formulation example
以下に本発明の化合物の製剤化例を示すが、 本発明の化合物の製剤化は、 本製 剤化例に限定されるものではない。 Formulation examples of the compound of the present invention are shown below, but the formulation of the compound of the present invention is not limited to the present formulation example.
製剤化例 1 Formulation example 1
化合物例 1116の化合物 45 (部) Compound Example 1116 Compound 45 (parts)
重質酸化マグネシウム 15 Heavy magnesium oxide 15
乳糖 75 Lactose 75
を均一に混合して 350 m以下の粉末状又は細粒状の散剤とする。 この散剤を 力プセル容器に入れて力プセフレ剤とした。 製剤化例 2 Into a powder or fine-grained powder of 350 m or less. This powder was placed in a forcepsell container to form a forcepsefle. Formulation example 2
化合物例 1116の化合物 45 (部) Compound Example 1116 Compound 45 (parts)
澱粉 15 乳糖 1 6 Starch 15 Lactose 1 6
結晶性セルロース 2 1 Crystalline cellulose 2 1
ポリビニルアルコール 3 Polyvinyl alcohol 3
蒸留水 3 0 Distilled water 3 0
を均一に混合した後、 破碎造粒して乾燥し、 次いで篩別して 1 4 1〜 1 7 7 ΠΙ の大きさの顆粒剤とした。 製剤化例 3 Was uniformly mixed, crushed and granulated, dried, and then sieved to obtain granules having a size of 141 to 1177ΠΙ. Formulation example 3
製剤化例 2と同様の方法で顆粒剤を作った後、 この顆粒剤 9 6部に対してステ アリン酸カルシウム 4部を加えて圧縮成形し、 直径 1 0 mmの錠剤を作製した。 製剤化例 4 After preparing granules in the same manner as in Formulation Example 2, 96 parts of the granules were added with 4 parts of calcium stearate and compression-molded to prepare tablets having a diameter of 10 mm. Formulation example 4
製剤化例 2の方法で得られた顆粒剤の 9 0部に対して結晶性セルロース 1 0部 及びステアリン酸カルシウム 3部を加えて圧縮成形し、 直径 8 mmの錠剤とした 後、 これにシロップゼラチン、 沈降性炭酸カルシウム混合懸濁液を加えて糖衣錠 を作製した。 産業上の利用可能性 To 90 parts of the granules obtained by the method of Formulation Example 2, 10 parts of crystalline cellulose and 3 parts of calcium stearate were added, and the mixture was compression-molded into tablets having a diameter of 8 mm. A sugar-coated tablet was prepared by adding a precipitated calcium carbonate mixed suspension. Industrial applicability
本発明化合物のベンゾチアゼピン誘導体は、 高い血中 G L P— 1濃度を呈する 活性を示していることから、 糖尿病治療剤、 糖尿病の慢性合併症の予防剤又は抗 肥満薬として有用である。 Since the benzothiazepine derivative of the compound of the present invention exhibits an activity of exhibiting a high blood GLP-1 concentration, it is useful as a therapeutic agent for diabetes, a preventive agent for chronic complications of diabetes, or an antiobesity agent.
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2000-397400 | 2000-12-27 | ||
| JP2000397400 | 2000-12-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2002053548A1 true WO2002053548A1 (en) | 2002-07-11 |
Family
ID=18862526
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2001/011267 Ceased WO2002053548A1 (en) | 2000-12-27 | 2001-12-21 | Benzothiazepine derivatives |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2002053548A1 (en) |
Cited By (50)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004020421A1 (en) * | 2002-08-28 | 2004-03-11 | Asahi Kasei Pharma Corporation | Novel quaternary ammonium compounds |
| WO2004076430A1 (en) * | 2003-02-25 | 2004-09-10 | Astrazeneca Ab | Benzothiazepine and benzothiepine derivatives |
| US7125864B2 (en) | 2001-09-07 | 2006-10-24 | Astrazeneca Ab | Benzothiazepine derivatives for the treatment of hyperlipidemia |
| US7132416B2 (en) | 2001-09-08 | 2006-11-07 | Astrazeneca Ab | Benzothiazepine and benzothiazepine derivatives with ileal bile acid transport (IBAT) inhibotory activity for the treatment hyperlipidaemia |
| US7192945B2 (en) | 2000-12-21 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7192946B2 (en) | 2001-09-04 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7192947B2 (en) | 2002-06-14 | 2007-03-20 | Astrazeneca Ab | Peptides derivatives comprising thiazepine group for the treatment of hyperlipidemic conditions |
| US7226943B2 (en) | 2001-09-07 | 2007-06-05 | Astrazeneca Ab | Benzothiepine ileal bile acid transport inhibitors |
| WO2007072083A1 (en) | 2005-12-23 | 2007-06-28 | Prosidion Limited | Treatment of type 2 diabetes with a combination of dpiv inhibitor and metformin or thiazolidinedione |
| US7238684B2 (en) | 2002-04-25 | 2007-07-03 | Astrazeneca Ab | Benzothiadiazepine derivatives, processes for their preparation and pharmaceutical compositions containing them |
| US7312044B2 (en) | 2003-03-07 | 2007-12-25 | The Trustees Of Columbia University In The City Of New York | Type 1 ryanodine receptor-based methods |
| US7393652B2 (en) | 2000-05-10 | 2008-07-01 | The Trustees Of Columbia University In The City Of New York | Methods for identifying a chemical compound that directly enhances binding of FKBP12.6 to PKA-phosphorylated type 2 ryanodine receptor (RyR2) |
| US7514421B2 (en) | 2003-04-05 | 2009-04-07 | Albireo Ab | Use of an IBAT inhibitor for the treatment of constipation |
| US7544678B2 (en) | 2002-11-05 | 2009-06-09 | The Trustees Of Columbia University In The City Of New York | Anti-arrythmic and heart failure drugs that target the leak in the ryanodine receptor (RyR2) |
| EP2165703A2 (en) | 2004-01-20 | 2010-03-24 | Novartis Pharma AG. | Direct compression formulation and process |
| US7973030B2 (en) | 2004-02-27 | 2011-07-05 | Asahi Kasei Pharma Corporation | Benzothiazepine and benzothiepine compounds |
| JP2013525444A (en) * | 2010-04-27 | 2013-06-20 | グラクソスミスクライン エルエルシー | Compound |
| US8710045B2 (en) | 2004-01-22 | 2014-04-29 | The Trustees Of Columbia University In The City Of New York | Agents for preventing and treating disorders involving modulation of the ryanodine receptors |
| EP2839832A2 (en) | 2003-11-17 | 2015-02-25 | Novartis AG | Use of dipeptidyl peptidase IV inhibitors |
| US9409875B2 (en) | 2013-04-26 | 2016-08-09 | Elobix Ab | Crystal modifications of elobixibat |
| US9688720B2 (en) | 2010-11-04 | 2017-06-27 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| WO2018162722A1 (en) | 2017-03-09 | 2018-09-13 | Deutsches Institut Für Ernährungsforschung Potsdam-Rehbrücke | Dpp-4 inhibitors for use in treating bone fractures |
| US10183920B2 (en) | 2014-10-24 | 2019-01-22 | Elobix Ab | Crystal modifications of elobixibat |
| US10441605B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10441604B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Cholestyramine pellets and methods for preparation thereof |
| EP2367554A4 (en) * | 2008-11-26 | 2020-03-25 | Satiogen Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of obesity and diabetes |
| US10709755B2 (en) | 2014-06-25 | 2020-07-14 | Elobix Ab | Solid formulation and method for preventing or reducing coloration thereof |
| US10722457B2 (en) | 2018-08-09 | 2020-07-28 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10786529B2 (en) | 2016-02-09 | 2020-09-29 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10793534B2 (en) | 2018-06-05 | 2020-10-06 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US10881685B2 (en) | 2017-08-09 | 2021-01-05 | Albireo Ab | Cholestyramine granules, oral cholestyramine formulations and use thereof |
| US10941127B2 (en) | 2019-02-06 | 2021-03-09 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US10975045B2 (en) | 2019-02-06 | 2021-04-13 | Aibireo AB | Benzothiazepine compounds and their use as bile acid modulators |
| US10975046B2 (en) | 2018-06-20 | 2021-04-13 | Albireo Ab | Crystal modifications of odevixibat |
| US11007142B2 (en) | 2018-08-09 | 2021-05-18 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US11014898B1 (en) | 2020-12-04 | 2021-05-25 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US11111224B2 (en) | 2019-12-04 | 2021-09-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11180465B2 (en) | 2019-12-04 | 2021-11-23 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11225466B2 (en) | 2019-12-04 | 2022-01-18 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US11260053B2 (en) | 2010-05-26 | 2022-03-01 | Satiogen Pharmaceuticals, Inc. | Bile acid recycling inhibitors and satiogens for treatment of diabetes, obesity, and inflammatory gastrointestinal conditions |
| US11267794B2 (en) | 2019-12-04 | 2022-03-08 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11306064B2 (en) | 2018-06-05 | 2022-04-19 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11377429B2 (en) | 2020-08-03 | 2022-07-05 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11549878B2 (en) | 2018-08-09 | 2023-01-10 | Albireo Ab | In vitro method for determining the adsorbing capacity of an insoluble adsorbant |
| US11572350B1 (en) | 2020-12-04 | 2023-02-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11583539B2 (en) | 2020-11-12 | 2023-02-21 | Albireo Ab | Treating progressive familial intrahepatic cholestasis (PFIC) with IBAT inhibitors |
| US11801226B2 (en) | 2018-06-20 | 2023-10-31 | Albireo Ab | Pharmaceutical formulation of odevixibat |
| US12187690B2 (en) | 2019-02-06 | 2025-01-07 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US12365658B2 (en) | 2021-06-03 | 2025-07-22 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US12545705B2 (en) | 2010-11-08 | 2026-02-10 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996005188A1 (en) * | 1994-08-10 | 1996-02-22 | The Wellcome Foundation Limited | Hypolipidemic 1,4-benzothiazepine-1,1-dioxides |
-
2001
- 2001-12-21 WO PCT/JP2001/011267 patent/WO2002053548A1/en not_active Ceased
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996005188A1 (en) * | 1994-08-10 | 1996-02-22 | The Wellcome Foundation Limited | Hypolipidemic 1,4-benzothiazepine-1,1-dioxides |
Non-Patent Citations (1)
| Title |
|---|
| JANOS SZABO ET AL.: "Synthesis and spectroscopic investigations of 1,4-benzothiazepine derivatives", CAN. J. CHEM., vol. 65, no. 1, 1987, pages 175 - 181, XP002950309 * |
Cited By (92)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7393652B2 (en) | 2000-05-10 | 2008-07-01 | The Trustees Of Columbia University In The City Of New York | Methods for identifying a chemical compound that directly enhances binding of FKBP12.6 to PKA-phosphorylated type 2 ryanodine receptor (RyR2) |
| US7192945B2 (en) | 2000-12-21 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7192946B2 (en) | 2001-09-04 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7226943B2 (en) | 2001-09-07 | 2007-06-05 | Astrazeneca Ab | Benzothiepine ileal bile acid transport inhibitors |
| US7125864B2 (en) | 2001-09-07 | 2006-10-24 | Astrazeneca Ab | Benzothiazepine derivatives for the treatment of hyperlipidemia |
| US7132416B2 (en) | 2001-09-08 | 2006-11-07 | Astrazeneca Ab | Benzothiazepine and benzothiazepine derivatives with ileal bile acid transport (IBAT) inhibotory activity for the treatment hyperlipidaemia |
| US7238684B2 (en) | 2002-04-25 | 2007-07-03 | Astrazeneca Ab | Benzothiadiazepine derivatives, processes for their preparation and pharmaceutical compositions containing them |
| US7192947B2 (en) | 2002-06-14 | 2007-03-20 | Astrazeneca Ab | Peptides derivatives comprising thiazepine group for the treatment of hyperlipidemic conditions |
| WO2004020421A1 (en) * | 2002-08-28 | 2004-03-11 | Asahi Kasei Pharma Corporation | Novel quaternary ammonium compounds |
| KR100990524B1 (en) | 2002-08-28 | 2010-10-29 | 아사히 가세이 파마 가부시키가이샤 | Novel Quaternary Ammonium Compounds |
| CN100494185C (en) * | 2002-08-28 | 2009-06-03 | 旭化成制药株式会社 | Novel quaternary ammonium compounds |
| JPWO2004020421A1 (en) * | 2002-08-28 | 2005-12-15 | 旭化成ファーマ株式会社 | New quaternary ammonium compounds |
| US7544678B2 (en) | 2002-11-05 | 2009-06-09 | The Trustees Of Columbia University In The City Of New York | Anti-arrythmic and heart failure drugs that target the leak in the ryanodine receptor (RyR2) |
| WO2004076430A1 (en) * | 2003-02-25 | 2004-09-10 | Astrazeneca Ab | Benzothiazepine and benzothiepine derivatives |
| JP2006518728A (en) * | 2003-02-25 | 2006-08-17 | アストラゼネカ アクチボラグ | Benzothiazepine and benzothiepine derivatives |
| US8067584B2 (en) | 2003-02-25 | 2011-11-29 | Albireo Ab | Benzothiazepine derivatives |
| US7312044B2 (en) | 2003-03-07 | 2007-12-25 | The Trustees Of Columbia University In The City Of New York | Type 1 ryanodine receptor-based methods |
| US7514421B2 (en) | 2003-04-05 | 2009-04-07 | Albireo Ab | Use of an IBAT inhibitor for the treatment of constipation |
| EP2839832A2 (en) | 2003-11-17 | 2015-02-25 | Novartis AG | Use of dipeptidyl peptidase IV inhibitors |
| EP3738585A1 (en) | 2004-01-20 | 2020-11-18 | Novartis Ag | Direct compression formulation and process |
| EP2165703A2 (en) | 2004-01-20 | 2010-03-24 | Novartis Pharma AG. | Direct compression formulation and process |
| EP3366283A1 (en) | 2004-01-20 | 2018-08-29 | Novartis AG | Direct compression formulation and process |
| EP3023095A1 (en) | 2004-01-20 | 2016-05-25 | Novartis AG | Direct compression formulation and process |
| US8710045B2 (en) | 2004-01-22 | 2014-04-29 | The Trustees Of Columbia University In The City Of New York | Agents for preventing and treating disorders involving modulation of the ryanodine receptors |
| US7973030B2 (en) | 2004-02-27 | 2011-07-05 | Asahi Kasei Pharma Corporation | Benzothiazepine and benzothiepine compounds |
| WO2007072083A1 (en) | 2005-12-23 | 2007-06-28 | Prosidion Limited | Treatment of type 2 diabetes with a combination of dpiv inhibitor and metformin or thiazolidinedione |
| EP2367554A4 (en) * | 2008-11-26 | 2020-03-25 | Satiogen Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of obesity and diabetes |
| US9040518B2 (en) | 2010-04-27 | 2015-05-26 | Glaxosmithkline Llc | Chemical compounds |
| JP2013525444A (en) * | 2010-04-27 | 2013-06-20 | グラクソスミスクライン エルエルシー | Compound |
| EP2563122A4 (en) * | 2010-04-27 | 2013-09-11 | Glaxosmithkline Llc | CHEMICAL COMPOUNDS |
| US11260053B2 (en) | 2010-05-26 | 2022-03-01 | Satiogen Pharmaceuticals, Inc. | Bile acid recycling inhibitors and satiogens for treatment of diabetes, obesity, and inflammatory gastrointestinal conditions |
| US11732006B2 (en) | 2010-11-04 | 2023-08-22 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US9694018B1 (en) | 2010-11-04 | 2017-07-04 | Albireo Ab | IBAT inhibitors for the treatment of liver disease |
| US10011633B2 (en) | 2010-11-04 | 2018-07-03 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10981952B2 (en) | 2010-11-04 | 2021-04-20 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US11261212B2 (en) | 2010-11-04 | 2022-03-01 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10093697B2 (en) | 2010-11-04 | 2018-10-09 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US9688720B2 (en) | 2010-11-04 | 2017-06-27 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10221212B2 (en) | 2010-11-04 | 2019-03-05 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US12187812B2 (en) | 2010-11-04 | 2025-01-07 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10000528B2 (en) | 2010-11-04 | 2018-06-19 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10487111B2 (en) | 2010-11-04 | 2019-11-26 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US12545705B2 (en) | 2010-11-08 | 2026-02-10 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US9701649B2 (en) | 2013-04-26 | 2017-07-11 | Elobix Ab | Crystal modifications of elobixibat |
| US9409875B2 (en) | 2013-04-26 | 2016-08-09 | Elobix Ab | Crystal modifications of elobixibat |
| US9745276B2 (en) | 2013-04-26 | 2017-08-29 | Elobix Ab | Crystal modifications of elobixibat |
| US11844822B2 (en) | 2014-06-25 | 2023-12-19 | Elobix Ab | Solid formulation and method for preventing or reducing coloration thereof |
| US10709755B2 (en) | 2014-06-25 | 2020-07-14 | Elobix Ab | Solid formulation and method for preventing or reducing coloration thereof |
| US10519120B2 (en) | 2014-10-24 | 2019-12-31 | Elobix Ab | Crystal modifications of elobixibat |
| US10183920B2 (en) | 2014-10-24 | 2019-01-22 | Elobix Ab | Crystal modifications of elobixibat |
| US10610543B2 (en) | 2016-02-09 | 2020-04-07 | Albireo Ab | Cholestyramine pellets and methods for preparation thereof |
| US10441605B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10864228B2 (en) | 2016-02-09 | 2020-12-15 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10493096B2 (en) | 2016-02-09 | 2019-12-03 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10441604B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Cholestyramine pellets and methods for preparation thereof |
| US10799527B2 (en) | 2016-02-09 | 2020-10-13 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10786529B2 (en) | 2016-02-09 | 2020-09-29 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10758563B2 (en) | 2016-02-09 | 2020-09-01 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| WO2018162722A1 (en) | 2017-03-09 | 2018-09-13 | Deutsches Institut Für Ernährungsforschung Potsdam-Rehbrücke | Dpp-4 inhibitors for use in treating bone fractures |
| US10881685B2 (en) | 2017-08-09 | 2021-01-05 | Albireo Ab | Cholestyramine granules, oral cholestyramine formulations and use thereof |
| US10793534B2 (en) | 2018-06-05 | 2020-10-06 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11306064B2 (en) | 2018-06-05 | 2022-04-19 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US10975046B2 (en) | 2018-06-20 | 2021-04-13 | Albireo Ab | Crystal modifications of odevixibat |
| US11802115B2 (en) | 2018-06-20 | 2023-10-31 | Albireo Ab | Pharmaceutical formulation of odevixibat |
| US12508234B2 (en) | 2018-06-20 | 2025-12-30 | Albireo Ab | Pharmaceutical formulation of odevixibat |
| US11801226B2 (en) | 2018-06-20 | 2023-10-31 | Albireo Ab | Pharmaceutical formulation of odevixibat |
| US12091394B2 (en) | 2018-06-20 | 2024-09-17 | Albireo Ab | Crystal modifications of odevixibat |
| US11365182B2 (en) | 2018-06-20 | 2022-06-21 | Albireo Ab | Crystal modifications of odevixibat |
| US11007142B2 (en) | 2018-08-09 | 2021-05-18 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US11549878B2 (en) | 2018-08-09 | 2023-01-10 | Albireo Ab | In vitro method for determining the adsorbing capacity of an insoluble adsorbant |
| US10722457B2 (en) | 2018-08-09 | 2020-07-28 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US11773071B2 (en) | 2019-02-06 | 2023-10-03 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US10975045B2 (en) | 2019-02-06 | 2021-04-13 | Aibireo AB | Benzothiazepine compounds and their use as bile acid modulators |
| US12187690B2 (en) | 2019-02-06 | 2025-01-07 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US11603359B2 (en) | 2019-02-06 | 2023-03-14 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US10941127B2 (en) | 2019-02-06 | 2021-03-09 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US12024495B2 (en) | 2019-12-04 | 2024-07-02 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US11267794B2 (en) | 2019-12-04 | 2022-03-08 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11111224B2 (en) | 2019-12-04 | 2021-09-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US12202809B2 (en) | 2019-12-04 | 2025-01-21 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11891368B2 (en) | 2019-12-04 | 2024-02-06 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11180465B2 (en) | 2019-12-04 | 2021-11-23 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US12060338B2 (en) | 2019-12-04 | 2024-08-13 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11708340B2 (en) | 2019-12-04 | 2023-07-25 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11225466B2 (en) | 2019-12-04 | 2022-01-18 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US11377429B2 (en) | 2020-08-03 | 2022-07-05 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US12447156B2 (en) | 2020-11-12 | 2025-10-21 | Albireo Ab | Treating progressive familial intrahepatic cholestasis (PFIC) with IBAT inhibitors |
| US11583539B2 (en) | 2020-11-12 | 2023-02-21 | Albireo Ab | Treating progressive familial intrahepatic cholestasis (PFIC) with IBAT inhibitors |
| US12134606B2 (en) | 2020-12-04 | 2024-11-05 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11572350B1 (en) | 2020-12-04 | 2023-02-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11014898B1 (en) | 2020-12-04 | 2021-05-25 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US12365658B2 (en) | 2021-06-03 | 2025-07-22 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2002053548A1 (en) | Benzothiazepine derivatives | |
| KR102429220B1 (en) | Substituted 4-phenylpiperidines, their preparation and use | |
| CN106535901B (en) | Mu-opioid receptor agonists | |
| ES2716151T3 (en) | Compound that has somatostatin receptor agonist activity and pharmaceutical use thereof | |
| WO2011123947A1 (en) | Synthesis of chiral 2-(1h-indazol-6-yl)-spiro[cyclopropane-1,3'- indolin]-2'-ones | |
| CA2896871A1 (en) | Heterocyclic compounds and methods of use thereof | |
| CN103180326A (en) | Heterocyclic compounds and their uses | |
| KR20150036223A (en) | Nitrogenous heterocyclic derivatives and their application in drugs | |
| TWI856012B (en) | Novel functionalized lactams as modulators of the 5-hydroxytryptamine receptor 7 and their method of use | |
| CN112823157B (en) | Spirochroman derivatives | |
| CN114710956A (en) | Perk-inhibiting pyrrolopyrimidine compounds | |
| JP2024516518A (en) | SOS1 decomposition agent, its manufacturing method and application | |
| CN115916776A (en) | Collagen 1 translation inhibitors and methods of use thereof | |
| JP2023178455A (en) | Novel heterocyclic compounds | |
| WO2001014386A1 (en) | Novel isoindole derivatives | |
| CN103864754A (en) | Five-membered azole heterocycle compound, preparation method, drug composition and use thereof | |
| CN107286166B (en) | Substituted 1,3,4, 5-tetrahydro-6H-pyrrolo [4,3,2-EF ] [2] benzazepin-6-one derivatives | |
| JP2015531398A (en) | Indoline compounds as aldosterone synthase inhibitor related applications | |
| JP2024516592A (en) | FXR Modulators, Methods of Preparation, Pharmaceutical Compositions and Uses Thereof | |
| CN103097391B (en) | Novel spiroheterocyclic compounds as mGlu5 antagonists | |
| WO2002066479A1 (en) | Novel isoindole derivatives | |
| WO2002067918A1 (en) | Novel diallylmethylamine derivative | |
| CN119462647A (en) | Aromatic linked fused ring compounds, pharmaceutical compositions containing the same and applications thereof | |
| WO2024140657A1 (en) | Substituted phenol derivative and use thereof | |
| AU2014365915A1 (en) | Tetrahydro-tetrazolo[1,5-a]pyrazines as ROR-gamma inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| 122 | Ep: pct application non-entry in european phase |