WO2001053268A2 - Indazole compounds, pharmaceutical compositions, and their use for mediating or inhibiting cell proliferation - Google Patents
Indazole compounds, pharmaceutical compositions, and their use for mediating or inhibiting cell proliferation Download PDFInfo
- Publication number
- WO2001053268A2 WO2001053268A2 PCT/US2001/001477 US0101477W WO0153268A2 WO 2001053268 A2 WO2001053268 A2 WO 2001053268A2 US 0101477 W US0101477 W US 0101477W WO 0153268 A2 WO0153268 A2 WO 0153268A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- substituted
- heteroaryl
- aryl
- carbocycle
- mmol
- Prior art date
Links
- 0 CC(*1)=NC2=C1CCCC2 Chemical compound CC(*1)=NC2=C1CCCC2 0.000 description 37
- NNIIWWUEQIMBRN-UHFFFAOYSA-N Cc(c(Br)cnc1)c1-c1ccncc1 Chemical compound Cc(c(Br)cnc1)c1-c1ccncc1 NNIIWWUEQIMBRN-UHFFFAOYSA-N 0.000 description 2
- ZUCBIFJLWXIOAO-UHFFFAOYSA-N Cc(c(O)c(CCCO)cc1)c1-c(cc1)cc2c1[nH]nc2-c1nc2ccccc2[nH]1 Chemical compound Cc(c(O)c(CCCO)cc1)c1-c(cc1)cc2c1[nH]nc2-c1nc2ccccc2[nH]1 ZUCBIFJLWXIOAO-UHFFFAOYSA-N 0.000 description 2
- BAFXMNLXDXWMOA-UHFFFAOYSA-N Cc1c(CO)cncc1Br Chemical compound Cc1c(CO)cncc1Br BAFXMNLXDXWMOA-UHFFFAOYSA-N 0.000 description 2
- XXJNYPHVMQLMQO-UHFFFAOYSA-N C(c(c1c2)n[nH]c1ccc2-c1cncc2c1cccc2)=C/c1ccccc1 Chemical compound C(c(c1c2)n[nH]c1ccc2-c1cncc2c1cccc2)=C/c1ccccc1 XXJNYPHVMQLMQO-UHFFFAOYSA-N 0.000 description 1
- FEUCJQGOICDSHT-UHFFFAOYSA-N CC(C)(C)CNCc1c(C)c(-c(cc23)ccc2[nH]nc3-c2nc(cccc3)c3[nH]2)cnc1 Chemical compound CC(C)(C)CNCc1c(C)c(-c(cc23)ccc2[nH]nc3-c2nc(cccc3)c3[nH]2)cnc1 FEUCJQGOICDSHT-UHFFFAOYSA-N 0.000 description 1
- JJNAUDAPHOCCKZ-UHFFFAOYSA-N CC(C)NCc1cncc(-c(cc2)cc3c2[nH]nc3-c2nc(cccc3)c3[nH]2)c1C Chemical compound CC(C)NCc1cncc(-c(cc2)cc3c2[nH]nc3-c2nc(cccc3)c3[nH]2)c1C JJNAUDAPHOCCKZ-UHFFFAOYSA-N 0.000 description 1
- UCKBCYGRSFGTBG-UHFFFAOYSA-N CC(N1c2cncc(Br)c2CCC1)=O Chemical compound CC(N1c2cncc(Br)c2CCC1)=O UCKBCYGRSFGTBG-UHFFFAOYSA-N 0.000 description 1
- MPMBRWOOISTHJV-XVNBXDOJSA-N CC/C=C/c1ccccc1 Chemical compound CC/C=C/c1ccccc1 MPMBRWOOISTHJV-XVNBXDOJSA-N 0.000 description 1
- KVNLRSUGAUVMBQ-UHFFFAOYSA-N CC1(C)C(CNCc2ccccc2)=CN=CC1c(cc12)ccc1[nH]nc2-c1nc2ccccc2[nH]1 Chemical compound CC1(C)C(CNCc2ccccc2)=CN=CC1c(cc12)ccc1[nH]nc2-c1nc2ccccc2[nH]1 KVNLRSUGAUVMBQ-UHFFFAOYSA-N 0.000 description 1
- YBPASEFOMWOPSZ-UHFFFAOYSA-N CC=C=C(C(C=C(CC1)C(C=C2)=C(C)C(CO)=CC2=C)=C1N)C1=C=Cc2ccccc2N1 Chemical compound CC=C=C(C(C=C(CC1)C(C=C2)=C(C)C(CO)=CC2=C)=C1N)C1=C=Cc2ccccc2N1 YBPASEFOMWOPSZ-UHFFFAOYSA-N 0.000 description 1
- VLTVTDFDRZUBNJ-HTXNQAPBSA-N CCCCc(c(C)c1C)ccc1-c1ccc(C)c(C(/C=C/c2ccccc2)=C=C(C)C=C)c1 Chemical compound CCCCc(c(C)c1C)ccc1-c1ccc(C)c(C(/C=C/c2ccccc2)=C=C(C)C=C)c1 VLTVTDFDRZUBNJ-HTXNQAPBSA-N 0.000 description 1
- QAKMXIUXFMQWFO-UHFFFAOYSA-N CCNCc1c(C)c(Br)cnc1 Chemical compound CCNCc1c(C)c(Br)cnc1 QAKMXIUXFMQWFO-UHFFFAOYSA-N 0.000 description 1
- VWMMBEIMPAJTGJ-UHFFFAOYSA-N COc(cc1)ccc1-c(cc1)cc2c1[nH]nc2-c1nc2ccccc2[nH]1 Chemical compound COc(cc1)ccc1-c(cc1)cc2c1[nH]nc2-c1nc2ccccc2[nH]1 VWMMBEIMPAJTGJ-UHFFFAOYSA-N 0.000 description 1
- BSQPDGSSJJZDLU-UHFFFAOYSA-N CS(NC(CC1N)=CC=C1N)(=O)=O Chemical compound CS(NC(CC1N)=CC=C1N)(=O)=O BSQPDGSSJJZDLU-UHFFFAOYSA-N 0.000 description 1
- WZWLRONVXLYRNO-UHFFFAOYSA-N Cc(c(O)cnc1)c1-c(cc1)cc(C(c2nc3ccccc3[nH]2)=N)c1N Chemical compound Cc(c(O)cnc1)c1-c(cc1)cc(C(c2nc3ccccc3[nH]2)=N)c1N WZWLRONVXLYRNO-UHFFFAOYSA-N 0.000 description 1
- IAGXHXLVWOLIHU-UHFFFAOYSA-N Cc1c(C)c(-c(cc2)cc(C(c3nc4ccccc4[nH]3)=N)c2N)cnc1 Chemical compound Cc1c(C)c(-c(cc2)cc(C(c3nc4ccccc4[nH]3)=N)c2N)cnc1 IAGXHXLVWOLIHU-UHFFFAOYSA-N 0.000 description 1
- VMKUWJPBSLTFMK-UHFFFAOYSA-N Cc1c(C=O)cncc1Br Chemical compound Cc1c(C=O)cncc1Br VMKUWJPBSLTFMK-UHFFFAOYSA-N 0.000 description 1
- DOBQWXPAXHCIBM-UHFFFAOYSA-N Cc1c(cccc2)c2ncc1-c(cc12)ccc1[nH]nc2-c1nc(cccc2)c2[nH]1 Chemical compound Cc1c(cccc2)c2ncc1-c(cc12)ccc1[nH]nc2-c1nc(cccc2)c2[nH]1 DOBQWXPAXHCIBM-UHFFFAOYSA-N 0.000 description 1
- WWNHLCQMASFRBX-UHFFFAOYSA-N Cc1cncc(Br)c1C Chemical compound Cc1cncc(Br)c1C WWNHLCQMASFRBX-UHFFFAOYSA-N 0.000 description 1
- FYUDUZRLZITSTF-UHFFFAOYSA-N Nc1cccc(CO)c1N Chemical compound Nc1cccc(CO)c1N FYUDUZRLZITSTF-UHFFFAOYSA-N 0.000 description 1
- PZJYVHVDYTYGJS-UHFFFAOYSA-N OS(c1ccncc1)O Chemical compound OS(c1ccncc1)O PZJYVHVDYTYGJS-UHFFFAOYSA-N 0.000 description 1
- JCCJSXHKZDWKCV-UHFFFAOYSA-N Oc(cc1)ccc1-c(cc1)cc2c1[nH]nc2-c1nc(cccc2)c2[nH]1 Chemical compound Oc(cc1)ccc1-c(cc1)cc2c1[nH]nc2-c1nc(cccc2)c2[nH]1 JCCJSXHKZDWKCV-UHFFFAOYSA-N 0.000 description 1
- KPANNKMXFVCOOV-UHFFFAOYSA-N c1ccc2nc(-c(c3c4)n[nH]c3ccc4-c3c(cccc4)c4cnc3)[nH]c2c1 Chemical compound c1ccc2nc(-c(c3c4)n[nH]c3ccc4-c3c(cccc4)c4cnc3)[nH]c2c1 KPANNKMXFVCOOV-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22D—CASTING OF METALS; CASTING OF OTHER SUBSTANCES BY THE SAME PROCESSES OR DEVICES
- B22D41/00—Casting melt-holding vessels, e.g. ladles, tundishes, cups or the like
- B22D41/50—Pouring-nozzles
- B22D41/56—Means for supporting, manipulating or changing a pouring-nozzle
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/54—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings condensed with carbocyclic rings or ring systems
- C07D231/56—Benzopyrazoles; Hydrogenated benzopyrazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/56—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
- C07F7/1804—Compounds having Si-O-C linkages
Definitions
- This invention is directed to indazole compounds that mediate and/or inhibit cell proliferation, for example, through the inhibition of the activity of protein kinases, such as VEGF, CHK-1, and cyclin-dependent kinases (CDKs), such as CDK1, CDK2, CDK4, and CDK6.
- protein kinases such as VEGF, CHK-1, and cyclin-dependent kinases (CDKs), such as CDK1, CDK2, CDK4, and CDK6.
- CDKs cyclin-dependent kinases
- Uncontrolled cell proliferation is the insignia of cancer.
- Cell proliferation in response to various stimuli is manifested by a deregulation of the cell division cycle, the process by which cells multiply and divide.
- Tumor cells typically have damage to the genes that directly or indirectly regulate progression through the cell division cycle.
- Hyperproliferative disease states including cancer are characterized by cells rampantly winding through the cell cycle with uncontrolled vigor due to, for example, damage to the genes that directly or indirectly regulate progression through the cycle.
- agents that modulate the cell cycle, and thus hyperproliferation could be used to treat various disease states associated with uncontrolled or unwanted cell proliferation.
- cell cycle inhibitors are also proposed as antiparasitics (See, Gray et al., Curr. Med. Chem. 6, 859-875 (1999)) and recently demonstrated as potential antivirals (See, Schang et al., J. Virol. 74, 2107-2120 (2000)).
- cardiovascular maladies such as artherosclerosis or restenosis (See Braun-Dullaeus et al., Circulation, 98, 82-89 (1998)), and states of inflammation, such as arthritis (See, Taniguchi et al., Nature Med., 5, 760-767(1999)) or psoriasis.
- cardiovascular maladies such as artherosclerosis or restenosis (See Braun-Dullaeus et al., Circulation, 98, 82-89 (1998)
- states of inflammation such as arthritis
- psoriasis psoriasis.
- Mechanisms of cell proliferation are under active investigation at cellular and molecular levels. At the cellular level, de-regulation of signaling pathways, loss of cell cycle controls, unbridled angiogenesis or stimulation of inflammatory pathways are under scrutiny, while at the molecular level, these processes are modulated by various proteins, among which protein kinases are prominent suspects. Overall abatement of proliferation may also result from programmed cell death,
- protein kinases are a family of enzymes that catalyze phosphorylation of the hydroxyl group of specific tyrosine, serine, or threonine residues in proteins. Typically, such phosphorylation dramatically perturbs the function of the protein, and thus protein kinases are pivotal in the regulation of a wide variety of cellular processes, including metabolisim, cell proliferation, cell differentiation, and cell survival. Of the many different cellular functions in which the activity of protein kinases is known to be required, some processes represent attractive targets for therapeutic intervention for certain disease states. Two examples are cell-cycle control and angiogenesis, in which protein kinases play a pivotal role; these processes are essential for the growth of solid tumors as well as for other diseases.
- CDKs constitute a class of enzymes that play critical roles in regulating the transitions between different phases of the cell cycle, such as the progression from a quiescent stage in Gi (the gap between mitosis and the onset of DNA replication for a new round of cell division) to S (the period of active DNA synthesis), or the progression from G 2 to M phase, in which active mitosis and cell-division occur.
- Gi the gap between mitosis and the onset of DNA replication for a new round of cell division
- S the period of active DNA synthesis
- G 2 to M phase in which active mitosis and cell-division occur.
- CDK complexes are formed through association of a regulatory cyclin subunit (e.g., cyclin A, B1 , B2, D1 , D2, D3, and E) and a catalytic kinase subunit (e.g., cdc2 (CDK1), CDK2, CDK4, CDK5, and CDK6).
- a regulatory cyclin subunit e.g., cyclin A, B1 , B2, D1 , D2, D3, and E
- a catalytic kinase subunit e.g., cdc2 (CDK1), CDK2, CDK4, CDK5, and CDK6.
- the D cyclins are sensitive to extracellular growth signals and become activated in response to mitogens during the G, phase of the cell cycle.
- CDK4/cyclin D plays an important role in cell cycle progression by phosphorylating, and thereby inactivating, the retinoblastoma protein (Rb). Hypophosphorylated Rb binds to a family of transcriptional regulators, but upon hyperphosphorylation of Rb by CDK4/cyclin D, these transcription factors are released to activate genes whose products are responsible for S phase progression.
- Rb phosphorylation and inactivation by CDK4/cyclin D permit passage of the cell beyond the restriction point of the Gi phase, whereupon sensitivity to extracellular growth or inhibitory signals is lost and the cell is committed to cell division.
- CDK2/cyclin E can also regulate progression into S phase through a parallel pathway that is independent of Rb phosphorylation (see Lukas et al., "Cyclin E-induced S Phase Without Activation of the pRb/E2F Pathway," Genes and Dev., vol. 11 (1997), pp. 1479-1492).
- cyclin E has also been observed in a wide variety of solid tumors, and elevated cyclin E levels have been correlated with poor prognosis.
- the cellular levels of the CDK inhibitor p27 which acts as both a substrate and inhibitor of CDK2/cyclin E, are abnormally low in breast, colon, and prostate cancers, and the expression levels of p27 are inversely correlated with the stage of disease (see Loda et al., "Increased Proteasome-dependent Degradation of the Cyclin-Dependent Kinase Inhibitor p27 in Aggressive Colorectal Carcinomas," Nature Medicine, vol. 3 (1997), pp. 231-234).
- CDK4/cyclin D might sequester p27, as reviewed in Sherr, et al., Genes Dev., Vol. 13 (1999), pp. 1501-1512.
- the p21 proteins also appear to transmit the p53 tumor- suppression signal to the CDKs; thus, the mutation of p53 in approximately 50% of all human cancers may indirectly result in deregulation of CDK activity.
- Flavopiridol displays modest selectivity for inhibition of CDKs over other kinases, but inhibits CDK4, CDK2, and CDK1 equipotently, with IC 50 s in the 0.1 -0.3 M range.
- Flavopiridol is currently in Phase II clinical trials as an oncology chemotherapeutic (Sedlacek et al., "Flavopiridol (L86-8275; NSC 649890), A New Kinase Inhibitor for Tumor Therapy," Int. J. Oncol., vol. 9 (1996), pp. 1143-1168).
- Analogs of flavopiridol are the subject of other publications, for example, U.S. Patent No.
- Benzensulfonamides that block cells in G1 are in development by Eisai, see Owa, et al., J. Med. Chem., Vol. 42 (1999), pp. 3789-3799.
- An oxindole CDK inhibitor is in development by Glaxo-Wellcome, see Luzzio, et al., Proc. Amer. Assoc. Cancer Res., Vol. (1999), Abst. 4102 and WO99/15500.
- Paullones were found in collaboration with the NCI, Schultz, et al., J. Med. Chem., Vol. (1999), pp. 2909-2919.
- Indenopyrazoles are described in WO99/17769 and by Seitz, et al, 218* ACS Natl. Mtg. (Aug. 22-26, 1999, New La), Abst MEDI 316. Aminothiazoles are used in WO99/24416 and W099/21845.
- CHK1 is another protein kinase. CHK 1 plays an important role as a checkpoint in cell cycle progression. Checkpoints are control systems that coordinate cell cycle progression by influencing the formation, activation and subsequent inactivation of the cyclin-dependent kinases. Checkpoints prevent cell cycle progression at inappropriate times, maintain the metabolic balance of cells while the cell is arrested, and in some instances can induce apoptosis (programmed cell death) when the requirements of the checkpoint have not been met.
- CHK1 a kinase, which transduces signals from the DNA- damage sensory complex to inhibit activation of the cyclin B/Cdc2 kinase, which promotes mitotic entry.
- CHK1 a kinase
- Inactivation of CHK1 has been shown to both abrogate G 2 arrest induced by DNA damage inflicted by either anticancer agents or endogenous DNA damage, as well as result in preferential killing of the resulting checkpoint defective cells.
- Tyrosine kinases can be of the receptor type (having extracellular, transmembrane and intracellular domains) or the non-receptor type (being wholly intracellular). At least one of the non-receptor protein tyrosine kinases, namely, LCK, is believed to mediate the transduction in T-cells of a signal from the interaction of a cell-surface protein (Cd4) with a cross-linked anti-Cd4 antibody.
- Cd4 cell-surface protein
- angiogenesis is the mechanism by which new capillaries are formed from existing vessels.
- the vascular system has the potential to generate new capillary networks in order to maintain the proper functioning of tissues and organs.
- angiogenesis is fairly limited, occurring only in the process of wound healing and neovascula zation of the endometrium during menstruation. See Merenmies, J., Parada, L. F., Henkemeyer, M., Cell Growth & Differentiation, 8, 3-10 (1997).
- VEGF-R2 vascular endothelial growth factor receptor 2, also know as KDR (kinase insert domain receptor) and as FLK-1
- FGF-R fibroblast growth factor receptor
- TEK also known as Tie-2
- VEGF-R2 which is expressed only on endothelial cells, binds the potent angiogenic growth factor VEGF and mediates the subsequent signal transduction through activation of its intracellular kinase activity.
- VEGF-R2 direct inhibition of the kinase activity of VEGF-R2 will result in the reduction of angiogenesis even in the presence of exogenous VEGF (see Strawn et al., Cancer Research, 56, 3540-3545 (1996)), as has been shown with mutants of VEGF-R2 which fail to mediate signal transduction. Millauer et al., Cancer Research, 56, 1615-1620 (1996).
- VEGF-R2 appears to have no function in the adult beyond that of mediating the angiogenic activity of VEGF. Therefore, a selective inhibitor of the kinase activity of VEGF-R2 would be expected to exhibit little toxicity.
- FGF-R binds the angiogenic growth factors aFGF and bFGF and mediates subsequent intracellular signal transduction. Recently, it has been suggested that growth factors such as bFGF may play a critical role in inducing angiogenesis in solid tumors that have reached a certain size. Yoshiji et al., Cancer Research, 57, 3924-3928 (1997).
- FGF-R is expressed in a number of different cell types throughout the body and may or may not play important roles in other normal physiological processes in the adult. Nonetheless, systemic administration of a small molecule inhibitor of the kinase activity of FGF-R has been reported to block bFGF-induced angiogenesis in mice without apparent toxicity. Mohammad et al., EMBO Journal, 17, 5996-5904 (1998).
- TEK also known as Tie-2
- Tie-2 is another receptor tyrosine kinase expressed only on endothelial cells which has been shown to play a role in angiogenesis.
- the binding of the factor angiopoietin-1 results in autophosphorylation of the kinase domain of TEK and results in a signal transduction process which appears to mediate the interaction of endothelial cells with peri-endothelial support cells, thereby facilitating the maturation of newly formed blood vessels.
- the factor angiopoietin-2 appears to antagonize the action of angiopoietin-1 on TEK and disrupts angiogenesis. Maisonpierre et al., Science, 277, 55-60 (1997).
- WO 97/34876 discloses certain cinnoline derivatives that are inhibitors of VEGF-R2, which may be used for the treatment of disease states associated with abnormal angiogenesis and/or increased vascular permeability such as cancer, diabetes, psoriosis, rheumatoid arthritis, Kaposi's sarcoma, haemangioma, acute and chronic nephropathies, atheroma, arterial restinosis, autoimmune diseases, acute inflammation and ocular diseases with retinal vessel proliferation.
- CDK4 may serve as a general activator of cell division in most cells, and because complexes of CDK4/cyclin D and CDK2/cyclin E govern the early G-
- An object of the invention is to provide potent anti-proliferative agents. Accordingly, one object of the invention is to attain compounds and drug compositions that inhibit the activity of one or more kinases, such as CDKs, VEGF, and CHK-1 , or cyclin complexes thereof. A further object is to provide an effective method of treating cancer indications through kinases inhibition, such as through inhibition of VEGF,
- Ri is a substituted or unsubstituted alkyl, aryl, heteroaryl, carbocycle, or heterocycle group, or
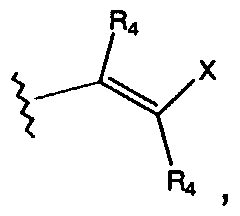
- R 4 is H or lower alkyl, and X is a substituted or unsubstituted alkyl, aryl, heteroaryl, carbocycle, or heterocycle group;
- R 2 is a substituted or unsubstituted alkyl, aryl, heteroaryl, carbocycle, or heterocycle group, or
- R 4 is H or lower alkyl, and X is a substituted or unsubstituted aryl, heteroaryl, carbocycle, or heterocycle group; or a pharmaceutically acceptable salt of a compound of the Formula I; or a prodrug or pharmaceutically active metabolite of a compound of the Formula I, or a pharmaceutically acceptable salt of the prodrug or metabolite.
- R'- is a substituted or unsubstituted alkyl, aryl, heteroaryl,
- each R 4 is individually H or lower alkyl and X is a substituted or unsubstituted alkyl, aryl, heteroaryl, carbocycle, or heterocycle group;
- R' 2 is a substituted or unsubstituted amino, nitro, alkenyl, alkyl, aryl,
- a pharmaceutical composition comprising:
- the invention also provides methods for making compounds of Formula I and II.
- a method of using a compound as a cell-cycle control agent for treating a disease or disorder mediated by inhibition of kinase comprising administering to a patient in need thereof, a compound of Formula I or II, or a pharmaceutically acceptable salt of a compound of the Formula I or II; or a prodrug or pharmaceutically active metabolite of a compound of the Formula I or II, or a pharmaceutically acceptable salt of the metabolite or prodrug.
- the invention further provides a method of treating mycotic infection, malignancies or cancer as well as other disease states associated with unwanted angiogenesis and/or cellular proliferation, comprising administering effective amounts of a compound of Formula I or II or a pharmaceutically acceptable salt of a compound of the Formula I or II; or a prodrug or pharmaceutically active metabolite of a compound of the Formula I or II, or a pharmaceutically acceptable salt of the metabolite or prodrug, to a patient in need of such treatment.
- the invention also provides a method of modulating and/or inhibiting kinase activity by administering a compound of the Formula I or II or a pharmaceutically acceptable salt of a compound of the Formula I or II; or a prodrug or pharmaceutically active metabolite of a compound of the Formula I or II, or a pharmaceutically acceptable salt of the metabolite or prodrug, to a patient in need thereof.
- a pharmaceutical composition containing a compound of the Formula I or II or a pharmaceutically acceptable salt of a compound of the Formula I or II; or a prodrug, or pharmaceutically active metabolite of a compound of the Formula I or II, or a pharmaceutically acceptable salt of the metabolite or prodrug, and the therapeutic use of the composition in treating diseases mediated by kinase activity, such as cancer, as well as other disease states associated with unwanted angiogenesis and/or cellular proliferation, such as diabetic retinopathy, neovascular glaucoma, rheumatoid arthritis, and psoriasis.
- diseases mediated by kinase activity such as cancer
- other disease states associated with unwanted angiogenesis and/or cellular proliferation such as diabetic retinopathy, neovascular glaucoma, rheumatoid arthritis, and psoriasis.
- Ri can also be hydrogen, in Formula I and II.
- inventive agents and compositions containing such agents may be useful in treating various disorders or disease states associated with uncontrolled or unwanted cellular proliferation, such as cancer, autoimmune disorders, viral diseases, fungal diseases, neurodegenerative disorders, and cardiovascular diseases.
- the invention is also directed to methods of treating such diseases by administering an effective amount of the inventive agent.
- the compounds and compositions of the present invention are useful as anti-proliferative agents and as inhibitors of mammalian kinase complexes, insect kinase or fungal kinase complexes.
- mammalian kinase complexes for example, VEGF, CHK-1 , and/or CDK complexes can be inhibited.
- Such compounds and compositions are also useful for controlling proliferation, differentiation, and/or apoptosis.
- R 1 t R 2 , R' 1 t and R' 2 preferred in compounds of Formula I or II groups are set forth below:
- Ri and R' ⁇ are:
- Y is CH or N or CR 3
- X is as defined above and R 3 is H, or one or more substituents located on the ring, such as a substituted or unsubstituted alkyl, alkenyl, aryl, heteroaryl, carbocycle, heterocycle, hydroxy, halogen, alkoxy, aryloxy, heteroaryloxy, thioalkyl, thioaryl, thioacyl, thioheteroaryl or amino; or
- Ri or R' ⁇ there can be one or more R 3 substituents on the phenyl ring.
- Ri and R' ⁇ are substituted or unsubstituted
- R 3 groups are as defined above. Also, two R 3 's together with an adjacent nitrogen can form a heteroaryl or heterocycle ring.
- R 2 and R' 2 are unsubstituted or substituted phenyl or
- R 4 is H or lower alkyl
- X is a substituted or unsubstituted group selected from alkyl, aryl, heteroaryl, carbocycle, or heterocycle.
- Other preferred R 2 and R' 2 groups are substituted or unsubstituted heteroaryls such as
- R 3 is as defined above.
- Especially preferred substituents for the phenyl of R 2 include fluorine, chlorine, hydroxyl, or an alkoxy group, such as methoxy. Examples of preferred R groups, X, and Y groups are found in the exemplary compounds that follow. Y is preferably nitrogen.
- X is preferably aryl, heteroaryl, carbocycle, or heterocycle, most preferably phenyl.
- R 2 and R' 2 can also be an amino (-NR'R"), wherein R' and R" are independently as defined for R 3 above, and together with an adjacent nitrogen can form a ring .
- R 4 is preferably hydrogen, or can be a lower alkyl having 1-6 carbon atoms, which may be substituted or unsubstituted.
- the two R 's can be the same or different.
- R ⁇ R 2 , R' ⁇ ,and R' 2 groups are found in the exemplary compounds that follow.
- alkyl group can be used, e.g., as Ri or R 2 or R' ⁇ or R' 2 or R 3 or X.
- the alkyl group can be a straight- or branched-chain alkyl group having one to twelve carbon atoms.
- Exemplary alkyl groups include methyl , ethyl, n-propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, tert-pentyl, hexyl, isohexyl, and the like.
- the alkyl can be substituted or unsubstituted.
- Preferred substituted alkyls include fluoromethyl, difluoromethyl, trifluoromethyl, 2-fluoroethyl, 3-fluoropropyl, hydroxymethyl, 2-hydroxyethyl, 3-hydroxypropyl, and the like.
- Any desired aryl, heteroaryl, carbocycle, or heterocycle group can be used as, e.g., Ri or R 2 or R'i or R' 2 or R 3 or X.
- the groups can be fused or non-fused, monocyclic or polycyclic.
- Preferred aryl and heteroaryl groups include monocyclic and polycyclic unsaturated or aromatic ring structures, with “aryl” referring to those that are carbocycles and “heteroaryl” referring to those that are heterocycles.
- ring structures include phenyl, naphthyl, 1 ,2,3,4-tetrahydronaphthyl, furyl, thienyl, pyrrolyl, pyridyl, pyridinyl, pyrazolyl, imidazolyl, pyrazinyl, pyridazinyl, 1 ,2,3-triazinyl, 1 ,2,4-oxadiazolyl, 1 ,3,4-oxadiazolyl, 1-H-tetrazol-5-yl, indolyl, quinolinyl, benzothiophenyl (thianaphthenyl), furanyl, thiophenyl, imidazolyl, oxazoly
- Preferred carbocyclic groups include those having from three to twelve carbon atoms, including bicyclic and tricyclic cycloalkyl structures.
- Preferred carbocyclic groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and the like.
- Preferred heterocyclic groups include saturated rings containing carbon atoms, for example containing 4 or 5 ring carbon atoms, and at least one heteroatom selected from nitrogen, oxygen and sulfur, and having no unsaturation.
- Preferred heterocyclic groups include pyrrolidinyl, piperidinyl, thiazinyl, and morpholinyl.
- Ri. R 2> R 3 . Y. X > and other R groups can be unsubstituted or substituted with any desired substituent or substituents that do not adversely affect the desired activity of the compound.
- preferred substituents are those found in the exemplary compounds that follows, as well as halogen (chloro, iodo, bromo, or fluoro); C ⁇ . 6 -alkyl; C ⁇ - 6 -alkenyl; C ⁇ . 6 -alkynyl; hydroxyl; C ⁇ .
- the present invention also relates to intermediates useful in the preparation of compounds of Formula I or II.
- a particularly preferred intermediate has the structure
- Another preferred intermediate has the structure
- SEM silyl
- PMB p-methoxybenzyl
- a preferred intermediate has the structure G
- T is a reactive group such as a substituted or unsubstituted boron, halogen, N0 2 , or NH 2 group
- T' is a reactive group such as CHO, C0 2 H, C0 2 R 3 , CONR 3 R 3 , where R 3 groups are as defined above.
- compositions according to the invention may, alternatively or in addition to a compound of the Formula I or II, comprise as an active ingredient a pharmaceutically acceptable salt of a compound of the Formula I or II, or a prodrug or pharmaceutically active metabolite of such a compound or salt or a salt of the prodrug or metabolite.
- a pharmaceutically acceptable salt of a compound of the Formula I or II or a prodrug or pharmaceutically active metabolite of such a compound or salt or a salt of the prodrug or metabolite.
- Such compounds, salts, prodrugs, and metabolites are sometimes referred to herein collectively as "cell-cycle control agents.”
- prodrug refers to a metabolic precursor of a compound of the
- a prodrug may be inactive when administered to a subject but is converted in vivo to an active compound of the Formula I or II.
- active metabolite refers to a metabolic product of a compound of the Formula I or II that is pharmaceutically acceptable and effective.
- Prodrugs and active metabolites of compounds of the Formula I or II may be determined using techniques known in the art. Prodrugs and active metabolites of a compound may be identified using routine techniques known in the art. See, e.g., Bertolini et al., J. Med. Chem., 40,
- inventive compounds may exist as single stereoisomers (i.e., essentially free of other stereoisomers), racemates, and/or mixtures of enantiomers and/or diastereomers. All such single stereoisomers, racemates and mixtures thereof are intended to be within the scope of the present invention.
- inventive compounds that are optically active are used in optically pure form.
- an optically pure compound having one chiral center ⁇ i.e., one asymmetric carbon atom is one that consists essentially of one of the two possible enantiomers (i.e., is enantiomerically pure), and an optically pure compound having more than one chiral center is one that is both diastereomerically pure and enantiomerically pure.
- the compounds of the present invention are used in a form that is at least 90% optically pure, that is, a form that contains at least 90% of a single isomer (80% enantiomeric excess (“e.e.") or diastereomeric excess ("d.e.”)), more preferably at least 95% (90% e.e. or d.e.), even more preferably at least 97.5% (95% e.e. or d.e.), and most preferably at least
- Formulas I and II are intended to cover solvated as well as unsolvated forms of the identified structures.
- Formulas I and II include compounds of the indicated structure in both hydrated and non-hydrated forms.
- Other examples of solvates include the structures in combination with isopropanol, ethanol, methanol, DMSO, ethyl acetate, acetic acid, or ethanolamine.
- a pharmaceutically acceptable salt is intended to mean a salt that retains the biological effectiveness of the free acids and bases of the specified compound and that is not biologically or otherwise undesirable.
- a compound of the invention may possess a sufficiently acidic, a sufficiently basic, or both functional groups, and accordingly react with any of a number of inorganic or organic bases, and inorganic and organic acids, to form a pharmaceutically acceptable salt.
- Exemplary pharmaceutically acceptable salts include those salts prepared by reaction of the compounds of the present invention with a mineral or organic acid or an inorganic base, such as salts including sulfates, pyrosulfates, bisulfates, sulfites, bisulfites, phosphates, monohydrogenphosphates, dihydrogenphosphates, metaphosphates, pyrophosphates, chlorides, bromides, iodides, acetates, propionates, decanoates, caprylates, acrylates, formates, isobutyrates, caproates, heptanoates, propiolates, oxalates, malonates, succinates, suberates, sebacates, fumarates, maleates, butyne- 1 ,4-dioates, hexyne-1 ,6-dioates, benzoates, chlorobenzoates, methylbenzoates, dinitrobenzoates, hydroxybenzoates, methoxy
- the desired pharmaceutically acceptable salt may be prepared by any suitable method available in the art, for example, treatment of the free base with an inorganic acid, such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid and the like, or with an organic acid, such as acetic acid, maleic acid, succinic acid, mandelic acid, fumaric acid, malonic acid, pyrovic acid, oxalic acid, glycolic acid, salicylic acid, a pyranosidyl acid, such as glucuronic acid or galacturonic acid, an alpha- hydroxy acid, such as citric acid or tartaric acid, an amino acid, such as aspartic acid or glutamic acid, an aromatic acid, such as benzoic acid or cinnamic acid, a sulfonic acid, such as p-toluenesulfonic acid or ethanesulfonic acid, or the like.
- an inorganic acid such as hydrochloric acid
- the desired pharmaceutically acceptable salt may be prepared by any suitable method, for example, treatment of the free acid with an inorganic or organic base, such as an amine (primary, secondary or tertiary), an alkali metal hydroxide or alkaline earth metal hydroxide, or the like.
- suitable salts include organic salts derived from amino acids, such as glycine and arginine, ammonia, primary, secondary, and tertiary amines, and cyclic amines, such as pipehdine, morpholine and piperazine, and inorganic salts derived from sodium, calcium, potassium, magnesium, manganese, iron, copper, zinc, aluminum and lithium.
- Cell-cycle control agents in accordance with the invention are useful as pharmaceuticals for treating proliferative disorders in mammals, especially humans, marked by unwanted proliferation of endogenous tissue.
- Compounds of the Formula I or II may be used for treating subjects having a disorder associated with excessive cell proliferation, e.g., cancers, psoriasis, immunological disorders involving undesired proliferation of leukocytes, and restenosis and other smooth-muscle disorders. Furthermore, such compounds may be used to prevent de-differentiation of post- mitotic tissue and/or cells.
- Diseases or disorders associated with uncontrolled or abnormal cellular proliferation include, but are not limited to, the following:
- cancers including, but not limited to, carcinoma, hematopoietic tumors of lymphoid lineage, hematopoietic tumors of myeloid lineage, tumors of mesenchymal origin, tumors of the central and peripheral nervous system and other tumors including melanoma, seminoma and Kaposi's sarcoma and the like.
- a disease process which features abnormal cellular proliferation, e.g., benign prostatic hyperplasia, familial adenomatosis polyposis, neuro- fibromatosis, atherosclerosis, pulmonary fibrosis, arthritis, psoriasis, glomerulonephritis, restenosis following angioplasty or vascular surgery, hypertrophic scar formation, inflammatory bowel disease, transplantation rejection, endotoxic shock, and fungal infections.
- abnormal cellular proliferation e.g., benign prostatic hyperplasia, familial adenomatosis polyposis, neuro- fibromatosis, atherosclerosis, pulmonary fibrosis, arthritis, psoriasis, glomerulonephritis, restenosis following angioplasty or vascular surgery, hypertrophic scar formation, inflammatory bowel disease, transplantation rejection, endotoxic shock, and fungal infections.
- apoptosis-associated conditions such as cancers (including but not limited to those types mentioned hereinabove), viral infections (including but not limited to herpesvirus, poxvirus, Epstein-Barr virus,
- Sindbis virus and adenovirus prevention of AIDS development in HIV- infected individuals, autoimmune diseases (including but not limited to systemic lupus erythematosus, rheumatoid arthritis, psoriasis, autoimmune mediated glomerulonephritis, inflammatory bowel disease and autoimmune diabetes mellitus), neurodegenerative disorders (including but not limited to Alzheimer's disease, amyotrophic lateral sclerosis, retinitis pigmentosa, Parkinson's disease, AIDS-related dementia, spinal muscular atrophy and cerebellar degeneration), myelodysplastic syndromes, aplastic anemia, ischemic injury associated with myocardial infarctions, stroke and reperfusion injury, arrhythmia, atherosclerosis, toxin-induced or alcohol related liver diseases, hematological diseases (including but not limited to chronic anemia and aplastic anemia), degenerative diseases of the musculoskeletal system (including but not limited to osteroporosis and arthritis),
- the active agents of the invention can modulate the level of cellular RNA and DNA synthesis and therefore are expected to be useful in the treatment of viral infections such as HIV, human papilloma virus, herpes virus, Epstein-Barr virus, adenovirus, Sindbis virus, pox virus and the like.
- Compounds and compositions of the invention inhibit the kinase activity of, for example, CDK/cyclin complexes, such as those active in the G 0 or G, stage of the cell cycle, e.g., CDK2, CDK4, and/or CDK6 complexes.
- the specific dosage amount of a cell-cycle control agent being administered to obtain therapeutic or inhibitory effects may be determined in a manner known in the art according to the particular circumstances surrounding the case, including, e.g., the specific agent being administered, the route of administration, the condition being treated, and the subject or host being treated.
- An exemplary total daily dose of a cell- cycle control agent which may be administered in single or multiple doses, contains a dosage level of from about 0.01 mg/kg body weight to about 50 mg/kg body weight.
- the cell-cycle control agents of the invention may be administered by any of a variety of suitable routes, such as orally, rectally, transdermally, subcutaneously, intravenously, intramuscularly, or intranasally.
- the cell-cycle control agents are preferably formulated into compositions suitable for the desired routes before being administered.
- a pharmaceutical composition or preparation according to the invention comprises an effective amount of a cell-cycle control agent, optionally one or more other active agents, and a pharmaceutically acceptable carrier, such as a diluent or excipient for the agent; when the carrier serves as a diluent, it may be a solid, semi- solid, or liquid material acting as a vehicle, excipient, or medium for the active ingredient(s).
- compositions according to the invention may be made by admixing the active ingredient(s) with a carrier, or diluting it with a carrier, or enclosing or encapsulating it within a carrier, which may be in the form of a capsule, sachet, paper container, or the like.
- Exemplary ingredients include Avicel (microcrystalline cellulose), starch, lactose, calcium sulfate dihydrate, terra alba, sucrose, talc, gelatin, agar, pectin, acacia, magnesium stearate, stearic acid, peanut oil, olive oil, glyceryl monostearate, Tween 80 (polysorbate 80), 1 ,3-butanediol, cocoa butter, beeswax, polyethylene glycol, propylene glycol, sorbitan monostearate, polysorbate 60, 2- octyldodecanol, benzyl alcohol, glycine, sorbic acid, potassium sorbate, disodium hydrogen phosphate, sodium chloride, and water.
- Avicel microcrystalline cellulose
- starch lactose
- calcium sulfate dihydrate terra alba
- sucrose talc
- gelatin agar
- pectin acacia
- compositions may be prepared in any of a variety of forms suitable for the desired mode of administration.
- pharmaceutical compositions may be prepared in the form of tablets, pills, powders, lozenges, sachets, cachets, elixirs, suspensions, emulsions, solutions, syrups, aerosols (as solids or in liquid media), ointments (e.g., containing up to 10% by weight of a cell-cycle control agent), soft-gel and hard-gel capsules, suppositories, sterile injectable solutions, sterile packaged powders, and the like.
- the carrier or diluent may include time-delay or time-release material known in the art, such as glyceryl monostearate or glyceryl distearate alone or with a wax, ethylcellulose, hydroxypropylmethylcellulose, methylmethacrylate and the like.
- the preparation can be tableted, placed in a hard gelatin capsule in powder or pellet form or in the form of a troche or lozenge.
- the amount of solid carrier may vary, but generally will be from about 25 mg to about 1 g.
- the preparation can be in the form of syrup, emulsion, soft gelatin capsule, sterile injectable solution or suspension in an ampoule or vial or non-aqueous liquid suspension.
- a pharmaceutically acceptable salt of an inventive agent is dissolved in an aqueous solution of an organic or inorganic acid, such as 0.3M solution of succinic acid or citric acid.
- the agent may be dissolved in a suitable cosolvent or combinations of cosolvents.
- suitable cosolvents include, but are not limited to, alcohol, propylene glycol, polyethylene glycol 300, polysorbate 80, gylcerin and the like in concentrations ranging from 0-60% of the total volume.
- a compound of Formula I or II may be dissolved in DMSO and diluted with water.
- the composition may also be in the form of a solution of a salt form of the active ingredient in an appropriate aqueous vehicle such as water or isotonic saline or dextrose solution.
- compositions of the invention may be manufactured in manners generally known for preparing pharmaceutical compositions, e.g., using conventional techniques such as mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping or lyophilizing.
- Pharmaceutical compositions may be formulated in a conventional manner using one or more physiologically acceptable carriers, which may be selected from excipients and auxiliaries that facilitate processing of the active compounds into preparations which can be used pharmaceutically. Proper formulation is dependent upon the route of administration chosen.
- the agents of the invention may be formulated into aqueous solutions, preferably in physiologically compatible buffers such as Hanks's solution, Ringer's solution, or physiological saline buffer.
- physiologically compatible buffers such as Hanks's solution, Ringer's solution, or physiological saline buffer.
- penetrants appropriate to the barrier to be permeated are used in the formulation. Such penetrants are generally known in the art.
- the compounds can be formulated readily by combining the active compounds with pharmaceutically acceptable carriers known in the art.
- Such carriers enable the compounds of the invention to be formulated as tablets, pills, dragees, capsules, liquids, gels, syrups, slurries, suspensions and the like, for oral ingestion by a patient to be treated.
- Pharmaceutical preparations for oral use can be obtained using a solid excipient in admixture with the active ingredient (agent), optionally grinding the resulting mixture, and processing the mixture of granules after adding suitable auxiliaries, if desired, to obtain tablets or dragee cores.
- Suitable excipients include: fillers such as sugars, including lactose, sucrose, mannitol, or sorbitol; and cellulose preparations, for example, maize starch, wheat starch, rice starch, potato starch, gelatin, gum, hydroxypropylmethyl-cellulose, sodium carboxymethylcellulose, methyl cellulose, or polyvinylpyrrolidone (PVP).
- disintegrating agents may be added, such as crosslinked polyvinyl pyrrolidone, agar, or alginic acid or a salt thereof such as sodium alginate.
- Dragee cores are provided with suitable coatings.
- concentrated sugar solutions may be used, which may optionally contain gum arabic, polyvinyl pyrrolidone, Carbopol gel, polyethylene glycol, and/or titanium dioxide, lacquer solutions, and suitable organic solvents or solvent mixtures.
- Dyestuffs or pigments may be added to the tablets or dragee coatings for identification or to characterize different combinations of active agents.
- compositions which can be used orally include push-fit capsules made of gelatin, as well as soft, sealed capsules made of gelatin and a plasticizer, such as glycerol or sorbitol.
- the push-fit capsules can contain the active ingredients in admixture with fillers such as lactose, binders such as starches, and/or lubricants such as talc or magnesium stearate, and, optionally, stabilizers.
- the active agents may be dissolved or suspended in suitable liquids, such as fatty oils, liquid paraffin, or liquid polyethylene glycols.
- stabilizers may be added. All formulations for oral administration should be in dosages suitable for such administration.
- the compositions may take the form of tablets or lozenges formulated in conventional manner.
- the compounds for use according to the present invention are conveniently delivered in the form of an aerosol spray presentation from pressurized packs or a nebulizer, with the use of a suitable propellant, e.g., dichlorodifluoromethane, trichlorofluoromethane, dichlorotetrafluoroethane, carbon dioxide or other suitable gas.
- a suitable propellant e.g., dichlorodifluoromethane, trichlorofluoromethane, dichlorotetrafluoroethane, carbon dioxide or other suitable gas.
- the dosage unit may be determined by providing a valve to deliver a metered amount.
- Capsules and cartridges of gelatin for use in an inhaler or insufflator and the like may be formulated containing a powder mix of the compound and a suitable powder base such as lactose or starch.
- the compounds may be formulated for parenteral administration by injection, e.g., by bolus injection or continuous infusion.
- Formulations for injection may be presented in unit-dosage form, e.g., in ampoules or in multi-dose containers, with an added preservative.
- the compositions may take such forms as suspensions, solutions or emulsions in oily or aqueous vehicles, and may contain formulatory agents such as suspending, stabilizing and/or dispersing agents.
- compositions for parenteral administration include aqueous solutions of the active compounds in water-soluble form. Additionally, suspensions of the active agents may be prepared as appropriate oily injection suspensions. Suitable lipophilic solvents or vehicles include fatty oils such as sesame oil, or synthetic fatty acid esters, such as ethyl oleate or triglycerides, or liposomes. Aqueous injection suspensions may contain substances which increase the viscosity of the suspension, such as sodium carboxymethyl cellulose, sorbitol, or dextran. Optionally, the suspension may also contain suitable stabilizers or agents which increase the solubility of the compounds to allow for the preparation of highly concentrated solutions.
- the active ingredient may be in powder form for constitution with a suitable vehicle, e.g., sterile pyrogen-free water, before use.
- a suitable vehicle e.g., sterile pyrogen-free water
- the compounds may also be formulated in rectal compositions such as suppositories or retention enemas, e.g., containing conventional suppository bases such as cocoa butter or other glycerides.
- the compounds may also be formulated as a depot preparation. Such long- acting formulations may be administered by implantation (for example, subcutaneously or intramuscularly) or by intramuscular injection.
- the compounds may be formulated with suitable polymeric or hydrophobic materials (for example, as an emulsion in an acceptable oil) or ion-exchange resins, or as sparingly soluble derivatives, for example, as a sparingly soluble salt.
- a pharmaceutical carrier for hydrophobic compounds is a cosolvent system comprising benzyl alcohol, a nonpolar surfactant, a water-miscible organic polymer, and an aqueous phase.
- the cosolvent system may be a VPD co-solvent system.
- VPD is a solution of 3% w/v benzyl alcohol, 8% w/v of the nonpolar surfactant polysorbate 80, and 65% w/v polyethylene glycol 300, made up to volume in absolute ethanol.
- the VPD co-solvent system (VPD:5W) contains VPD diluted 1 :1 with a 5% dextrose in water solution. This co-solvent system dissolves hydrophobic compounds well, and itself produces low toxicity upon systemic administration.
- co-solvent system may be varied considerably without destroying its solubility and toxicity characteristics.
- identity of the co-solvent components may be varied: for example, other low-toxicity nonpolar surfactants may be used instead of polysorbate 80; the fraction size of polyethylene glycol may be varied; other biocompatible polymers may replace polyethylene glycol, e.g. polyvinyl pyrrolidone; and other sugars or polysacchahdes may be substituted for dextrose.
- other delivery systems for hydrophobic pharmaceutical compounds may be employed. Liposomes and emulsions are known examples of delivery vehicles or carriers for hydrophobic drugs.
- Certain organic solvents such as dimethylsulfoxide also may be employed, although usually at the cost of greater toxicity.
- the compounds may be delivered using a sustained-release system, such as semipermeable matrices of solid hydrophobic polymers containing the therapeutic agent.
- sustained-release materials have been established and are known by those skilled in the art.
- Sustained-release capsules may, depending on their chemical nature, release the compounds for a few weeks up to over 100 days.
- additional strategies for protein stabilization may be employed.
- the pharmaceutical compositions also may comprise suitable solid- or gel- phase carriers or excipients. Examples of such carriers or excipients include calcium carbonate, calcium phosphate, sugars, starches, cellulose derivatives, gelatin, and polymers such as polyethylene glycols.
- Some of the compounds of the invention may be provided as salts with pharmaceutically compatible counter ions.
- Pharmaceutically compatible salts may be formed with many acids, including hydrochloric, sulfuric, acetic, lactic, tartaric, malic, succinic, etc. Salts tend to be more soluble in aqueous or other protonic solvents than are the corresponding free-base forms.
- a pharmaceutical composition according to the invention comprises a cell- cycle control agent and, optionally, one or more other active ingredients, such as a known antiproliferative agent that is compatible with the cell-cycle control agent and suitable for the indication being treated.
- the compounds are useful as anti-angiogenesis agents and as agents for modulating and/or inhibiting the activity of protein kinases, thus providing treatments for cancer or other diseases associated with cellular proliferation mediated by protein kinases.
- Therapeutically effective amounts of the agents of the invention may be used to treat diseases mediated by modulation or regulation of protein kinases.
- An "effective amount" is intended to mean that amount of an agent that, when administered to a mammal in need of such treatment, is sufficient to effect treatment for a disease mediated by the activity of one or more kinases.
- a therapeutically effective amount of a compound of the Formula I or II, salt, active metabolite or prodrug thereof is a quantity sufficient to modulate, regulate, or inhibit the activity of one or more kinases such that a disease condition which is mediated by that activity is reduced or alleviated.
- 'Treating is intended to mean at least the mitigation of a disease condition in a mammal, such as a human, that is affected, at least in part, by the activity of one or more kinases, and includes: preventing the disease condition from occurring in a mammal, particularly when the mammal is found to be predisposed to having the disease condition but has not yet been diagnosed as having it; modulating and/or inhibiting the disease condition; and/or alleviating the disease condition.
- inventive agents may be prepared using the reaction routes and synthesis schemes as described below, employing the techniques available in the art using starting materials that are readily available.
- a 5-nitro indazole can be halogenated as described above for intermediate A, to afford nitro compound H, by treatment with a suitable base such as sodium hydroxide or potassium hydroxide and elemental halogen such as iodine to yield an intermediate I after standard protection with an appropriate protecting group.
- a suitable base such as sodium hydroxide or potassium hydroxide
- elemental halogen such as iodine
- intermediate I Treatment of intermediate I with an alkyl ditin species, such as hexamethyl ditin, and a suitable Pd catalyst, can afford intermediate J.
- Further reaction of nitro compound J with an alkyl or aryl boronic acid or ester and a suitable Pd catalyst affords the 3-substituted indazole K.
- Reduction of K with a suitable reducing agent, such as hydrogen with palladium catalyst or SnCI 2 affords the amine.
- a suitable reducing agent such as hydrogen with palladium catalyst or SnCI 2
- Diazotization of the resulting 5-amino indazole and treatment of the resulting diazonium salt with a suitable halide salt, such as CuCI or KI affords intermediate halo compound L.
- Reaction of L with an alkyl or aryl boronic acid or ester and a suitable Pd catalyst affords the intermediate M which is deprotected as before to yield final compound F.
- R-, and R 2 are as defined above, and can be R'-, and R' 2 .
- 3-carboxyindazole is activated to provide an active acylating species, such as with carbonyldiimidazole, which is then treated with a suitable alkoxy-alkyl amine, such N,N-dimethylhydroxylamine, to afford the amide A'.
- a suitable alkoxy-alkyl amine such N,N-dimethylhydroxylamine
- Selective halogenation of intermediate A' with elemental halogen such as bromine or iodine and preferably with a catalyst such as bis(trifluoroacetoxy)iodosobenzene or bis(acetoxy) iodosobenzene yields the 5-halo indazole B'. Protection of intermediate B' under standard conditions with a suitable protecting group such as PMB or THP affords protected amide C.
- R 3 is as defined above, and is preferably substituted or unsubstituted alkyl, preferably lower alkyl.
- intermediate D' is reacted with a substituted diamine B" and a suitable oxidizing agent such as sulfur to afford the benzimidazole C".
- Conversion of compound C" to the corresponding borinate ester D" is accomplished by reacting with a suitable diboron species, such as dipinacolatodiboron, or other electrophilic source of boron, with an appropriate palladium catalyst.
- Intermediate D" is further reacted with a halogenated aryl or alkyl halide under palladium catalysis to give 5-substituted indazole intermediate E", which after appropriate deprotection affords the final compound H".
- starting compound D' is reacted with a suitable diboron species, such as bis(pinacolato)diboron, or other suitable electrophilic source of boron, and an appropriate palladium catalyst to give boron ester P.
- a suitable diboron species such as bis(pinacolato)diboron, or other suitable electrophilic source of boron
- palladium catalyst such as palladium
- Another alternative conversion can be accomplished by reacting intermediate aldehyde F with a substituted aryl or alkyl halide to provide R 2 with a palladium catalyst to afford G" which is further reacted with a substituted diamine B" and a suitable oxidizing agent such as sulfur to afford the benzimidazole E". Deprotection as before yields final compound H".
- R 2 is as defined above and can be R' 2 .
- R 3 is as defined above.
- intermediate E can be accomplished by reacting compound such as C" directly with a suitable alkyl borinic acid or ester under suitable palladium catalysis.
- R 3 is as defined above and two R 3 groups can form a ring.
- n 1 ,2 1
- alcohol intermediate X 1 can be activated for example by reaction with a sulfonyl halide such as methanesulfonyl chloride and a suitable base such as triethylamine and this electrophilic species reacted further with a nucleophile such as a substituted amine to afford the intermediate X 2 which is then deprotected under the appropriate conditions.
- R 2 is as defined above, and can be R' 2 .
- R 3 is as defined above.
- the core indazole structure is formed in an annulation of a 2-halo-5-nitrophenyl aryl ketone Y1 with hydrazine to provide the requisite 3-aryl-5-nitroindazole Y2. Subsequent protection and reduction provides the amine Y4.
- diazotization treatment of the diazonium salt with KI, followed by Pd catalyzed coupling of the iodo intermediate with an aryl boronic acid affords the protected 3,5-bisarylindazole intermediate Y6. Standard deprotection then yields the final products.
- Ri and R 2 are as defined above, and can be R' ⁇ and R' 2 .
- THF Tetrahydrofuran
- DMF N, N-dimethylformamide
- the reactions set forth below were done generally under a positive pressure of argon or with a drying tube, at ambient temperature (unless otherwise stated), in anhydrous solvents, and the reaction flasks were fitted with rubber septa for the introduction of substrates and reagents via syringe. Glassware was oven dried and/or heat dried. Analytical thin layer chromatography (TLC) was performed on glass- backed silica gel 60 F 254 plates Analtech (0.25 mm) and eluted with the appropriate solvent ratios (v/v), and are denoted where appropriate. The reactions were assayed by TLC and terminated as judged by the consumption of starting material.
- TLC thin layer chromatography
- NMR spectra were recorded on a Bruker instrument operating at 300 MHz or 500 MHz and 13 C-NMR spectra were recorded operating at 75 MHz.
- NMR spectra were obtained as CDCI 3 solutions (reported in ppm), using chloroform as the reference standard (7.25 ppm and 77.00 ppm) or CD 3 OD (3.4 ppm and 4.8 ppm and 49.3 ppm), or internal tetramethylsilane (0.00 ppm) when appropriate.
- Other NMR solvents were used as needed.
- IR Infrared
- Example 1 5-Phenyl-3-Styryl-1 W-lndazole
- 5-Amino-1 H-indazole (15.41 g, 116 mmol) was suspended in a mixture of water (250 mL), ice (250 mL), and concentrated HCl (100 mL). The mixture was cooled in an ice-salt bath to an internal temperature of -5 °C. To this mixture, was added a solution of sodium nitrite (8.78 g, 127 mmol) in water (75 mL), which had been cooled to 0 °C. The resulting diazonium solution was stirred for 15 minutes at -5 °C.
- 5-Chloro-3-iodo-2-SEM-indazole 1b (553 mg, 1.35 mmol), styryl boronic acid (300 mg, 2.03 mmol), and tetrakis(triphenylphosphine) palladium (78.2 mg, 0.068 mmol) were dissolved in toluene (10 mL) and methanol (1.4 mL). Saturated aqueous sodium bicarbonate solution (1.7 mL) was added, and the mixture heated in a 90 °C oilbath for 3 hours. Slight refluxing was observed. After cooling to room temperature, the solution was diluted with water (15 mL) and extracted with ethyl acetate (4 x 50 mL).
- the resulting diazonium solution was stirred for 10 minutes at -5 °C, then a solution of potassium iodide (15.3 g, 92 mmol) in water (50 mL) was added slowly dropwise. Significant foaming occurred with the first few drops of KI solution, and then a black, tarry gum formed. After the addition was completed, the mixture was heated to 90 °C for 1 hour. The tarry precipitate dissolved and purple vapor was evolved during heating. The reaction was then cooled to room temperature, causing a fine brown precipitate to form.
- Intermediate 2b was prepared by a synthetic method analogous to intermediate 1a synthesis.
- Styryl boronic acid (186 mg, 1.26 mmol) was added to a solution of 2c (210.0 mg, 0.42 mmol) and tetrakis(triphenylphosphine) palladium (48.5 mg, 0.042 mmol) in toluene (3.5 mL) and methanol (0.5 mL).
- Saturated aqueous sodium bicarbonate solution (1.05 mL) was added, and the mixture heated in a 90 °C oilbath (slight reflux) for 4 hours. After cooling to room temperature, the reaction was poured into water (15 mL) and extracted with ethyl acetate (4 x 50 mL).
- Example 3 3-(1H-Benzoimidazol-2-yl)-5-Phenyl-1H-lndazole
- aldehyde 7b' (2.74 g, 6.81 mmol) in DMF (130 mL) were added 1 ,2-phenylenediamine (0.74 g, 6.81 mmol) and elemental sulfur (0.26 g, 8.2 mmol).
- the mixture was heated in a 95 °C oilbath for 14.5 hours, cooled to room temperature, and diluted with ethyl acetate (500 mL).
- the solution was washed with a mixture of saturated aqueous sodium chloride (100 mL) and water (100 mL).
- the reaction was left stirring overnight, gradually equilibrating to room temperature.
- the crude reaction mixture was poured into water (300 mL) and extracted with ethyl acetate (2 x 500 mL). The combined organic extracts were dried over sodium sulfate and concentrated.
- HRMS calculated for C 23 H 20 N 2 O 341.1648 (MH + ), found 341.1638. Anal. (C 23 H 20 N 2 O « 0.2H 2 O) C, H, N.
- Example 17 was prepared similar to intermediate 16. 17a was deprotected to afford 17 in 50% yield as a white solid.
- Anal. C 24 H ⁇ 7 N 3 «1.0 H 2 0) C, H, N.
- Example 18 5-(4-Pyridyl)-3- 2-Pyrrolyl)-1H-lndazole
- the resultant reaction mixture was heated in a 90 °C oil bath for 18 hours, under an argon atmosphere. After cooling to room temperature, the crude reaction mixture was poured into water (100 ml) and extracted with ethyl acetate (2 x 75 ml). The combined organic extracts were washed with water (6 x 75 ml), dried over sodium sulfate and concentrated.
- the mixture was heated in an 80 °C oilbath for 1.5 hours, then cooled and partitioned between ethyl acetate (50 mL) and water (10 mL). The organic layer was dried over magnesium sulfate, filtered, and concentrated.
- the mixture was heated in an 80 °C oilbath for 6 hours, then cooled and partitioned between ethyl acetate (50 mL) and saturated aqueous sodium chloride solution (25 mL). The organic layer was dried over magnesium sulfate, filtered, and concentrated.
- Example 26 3-f3- 1 H-Benzoimidazol
- Methane sulfonyl chloride (119.3 mg, 1.04 mmol) was added dropwise to a solution of 31a (527.5 mg, 1.03 mmol) and diisopropylethyl amine (153.3 mg, 1.19 mmol) in tetrahydrofuran (12.0 mL), cooled to 0 °C in an icebath. After stirring at 0 °C for 2.5 hours, the reaction flask was fitted with a dry ice-cooled cold finger condenser, and dimethyl amine gas was condensed into the reaction solution until the volume had increased by about 5 mL. Stirring was continued at 0 °C for 4 hours, then at room temperature for 15 hours.
- alcohol 31a 512.0 mg, 1.00 mmol
- methanesulfonyl chloride and diisopropylethyl amine at 0 °C for 1 hour.
- Aminomethylcyclopropane (712 mg, 10.0 mmol) was then added, and stirring continued at room temperature for 15 hours.
- alcohol 31a (511.4 mg, 1.00 mmol) was treated with methanesulfonyl chloride and diisopropylethyl amine at 0 °C for 2.5 hours.
- alcohol 31a 371.5 mg, 0.726 mmol
- methanesulfonyl chloride and diisopropylethyl amine at 0°C for 2.5 hours.
- the reaction flask was then fitted with a dry ice-cooled cold finger condenser, and ethylamine gas was condensed into the reaction solution until the volume had increased by about 5 mL. Stirring was continued at room temperature for 15 hours.
- alcohol 31a (623.2 mg, 1.22 mmol) was treated with methanesulfonyl chloride and diisopropylethyl amine at 0 °C for 1 hour.
- te/f-Butylamine (890 mg, 12.2 mmol) was then added, and stirring continued at room temperature for 20 hours.
- alcohol 31a (572.0 mg, 1.12 mmol) was treated with methanesulfonyl chloride and diisopropylethyl amine at 0 °C for 1 hour.
- Imidazole (761 mg, 11.2 mmol) was then added, and stirring was continued at room temperature for 24 hours.
- LDA was prepared by the addition of n-butyllithium (2.5 M in hexanes, 0.95 mL, 2.38 mmol) to a solution of diisopropylamine (0.345 mL, 2.42 mmol) in THF (5 mL) at -20 °C. After 10 minutes, the solution was cooled to -78 °C. 4-Chloro-3-iodo- pyridine (500 mg, 2.09 mmol) in THF (3 mL) was added dropwise, and the reaction stirred for 30 minutes lodoethane (0.2 mL, 2.5 mmol) was added, and the reaction was stirred for 1 hour at -78 °C, then 1 hour while warming to 0 °C.
- Example 50 4-r-3-(1 H-Benzoimidazol-2-ylH H-indazol-5-yll-3,5-dif luoro-phenol and
- Example 51 2-r-3-(1 H-Benzoimidazol-2-yl)-1 H-indazol-5-yl1-3.5-dif luoro- phenol
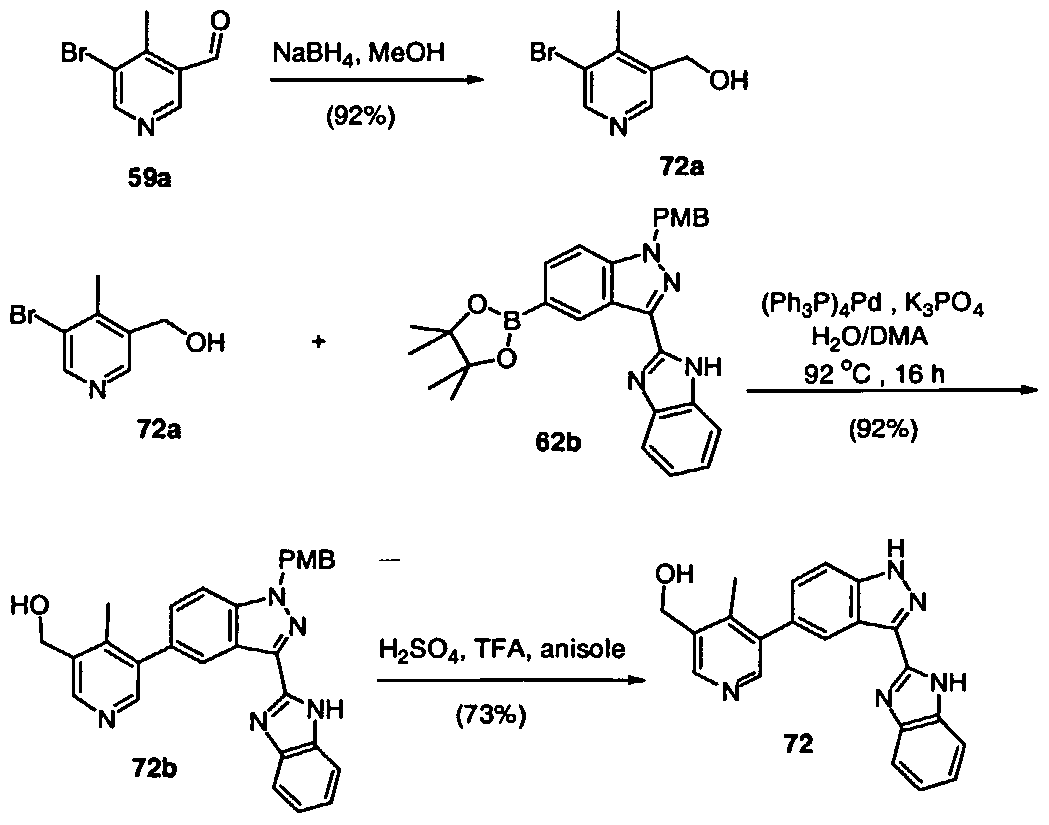
- Example 59 (£)-3-(5-r3-(1 H-Benzoimidazol-2-yl)-1 H-indazol-5-yl1-4-methyl- pyridin-3-yl)-prop-2-en-1-ol and Example 60: (B-3-(5-r3-(1 H-Benzoimidazol-2-ylH H-indazol-5-yll-4-methyl- pyridin-3-yl)-propan-1 -oi
- Tetrakis(triphenylphosphine)palladium (0) (82 mg, 0.07 mmol) was added, and the reaction stirred at 92 °C under argon for 16 hours The solution was diluted with ethyl acetate, washed with H 2 0 and brine, dried over Na 2 S0 4 , and concentrated in vacuo. Purification by silica gel chromatography (75% to 100% ethyl acetate/hexanes) gave 232 mg (60%) of intermediate 61c as a clear oil.
- Example 84 5-Amino-3
- Benzaldehyde (100 mg, 1 mmol) was added to a solution of 5-amino-3-(2- pyrrolyl)-1 H-indazole 84 (100 mg, 0.5 mmol) in EtOH (100 ml). The resultant solution was stirred for 2 hours at ambient temperature prior to addition of NaBH 3 CN (50 mg, 0.8 mmol) in a single portion as the solid. After stirring for an additional 2 hours, the crude reaction mixture was poured into H 2 0 (200 ml) and extracted with ethyl acetate (2 x 100 ml). The combined organic extracts were dried over sodium sulfate and concentrated.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Veterinary Medicine (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Mechanical Engineering (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
Claims
Priority Applications (21)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR0107783-0A BR0107783A (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, their pharmaceutically acceptable salts, prodrugs or pharmaceutically active metabolites, pharmaceutical compositions, method of treating diseases or dysfunctions mediated by the inhibition of a kinase complex, method of treating mammalian diseases mediated by kinase activity, method of modulation or inhibition of the activity of a protein kinase receptor, method of treatment of unhealthy condition or dysfunction associated with uncontrolled cell proliferation, pharmaceutical composition for the treatment of unhealthy condition associated with uncontrolled cell proliferation and method of treatment of unhealthy condition or associated dysfunction to uncontrolled cell proliferation |
| APAP/P/2002/002564A AP1609A (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation. |
| UA2002086633A UA75880C2 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds as antiproliferative agents, a pharmaceutical composition (variants) and a method for treatment of diseases and disorders (variants) associated wiyh unwanted angiogenesis and/or cellular proliferation |
| PL01357590A PL357590A1 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| SK1005-2002A SK10052002A3 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and their use for mediating or inhibiting cell proliferation |
| AU29539/01A AU785013B2 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| CA002388885A CA2388885A1 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| DZ013301A DZ3301A1 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and their use for mediating or inhibiting cell proliferation |
| MXPA02007058A MXPA02007058A (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation. |
| KR1020027009194A KR20020073505A (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| EEP200200398A EE200200398A (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions and methods for mediating or inhibiting cell proliferation |
| HU0203965A HUP0203965A3 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds and pharmaceutical compositions containing them |
| NZ518531A NZ518531A (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| IL15073001A IL150730A0 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| EA200200768A EA200200768A1 (en) | 2000-01-18 | 2001-01-18 | INDAZOL COMPOUNDS, PHARMACEUTICAL COMPOSITIONS AND METHODS FOR MODULATION OR INHIBITION OF CELLULAR PROLIFERATION |
| JP2001553270A JP2003520273A (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions and methods for inducing or inhibiting cell proliferation |
| EP01942620A EP1250326A2 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and their use for mediating or inhibiting cell proliferation |
| NO20022117A NO20022117L (en) | 2000-01-18 | 2002-05-03 | Indazole Compounds, Pharmaceutical Preparations, and Method of Mediating or Inhibiting Cell Profiling |
| IS6474A IS6474A (en) | 2000-01-18 | 2002-07-16 | Indazol compounds pharmaceutical compositions and a method for controlling or inhibiting cell proliferation |
| BG107011A BG107011A (en) | 2000-01-18 | 2002-08-16 | Indazole compounds, pharmaceutical compositions and methods for mediating or inhibiting cell proliferation |
| HRP20020675 HRP20020675A2 (en) | 2000-01-18 | 2002-08-16 | Indazole compounds, pharmaceutical compositions and methods for mediating or inhibiting cell poliferation |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17648400P | 2000-01-18 | 2000-01-18 | |
| US60/176,484 | 2000-01-18 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2001053268A2 true WO2001053268A2 (en) | 2001-07-26 |
| WO2001053268A3 WO2001053268A3 (en) | 2001-12-27 |
| WO2001053268B1 WO2001053268B1 (en) | 2002-01-24 |
Family
ID=22644536
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2001/001477 WO2001053268A2 (en) | 2000-01-18 | 2001-01-18 | Indazole compounds, pharmaceutical compositions, and their use for mediating or inhibiting cell proliferation |
Country Status (39)
| Country | Link |
|---|---|
| US (4) | US6555539B2 (en) |
| EP (1) | EP1250326A2 (en) |
| JP (1) | JP2003520273A (en) |
| KR (1) | KR20020073505A (en) |
| CN (1) | CN1394205A (en) |
| AP (1) | AP1609A (en) |
| AR (1) | AR032438A1 (en) |
| AU (1) | AU785013B2 (en) |
| BG (1) | BG107011A (en) |
| BR (1) | BR0107783A (en) |
| CA (1) | CA2388885A1 (en) |
| CO (1) | CO5280070A1 (en) |
| CR (1) | CR6630A (en) |
| DO (1) | DOP2001000120A (en) |
| DZ (1) | DZ3301A1 (en) |
| EA (1) | EA200200768A1 (en) |
| EE (1) | EE200200398A (en) |
| GE (1) | GEP20043363B (en) |
| GT (1) | GT200100009A (en) |
| HN (1) | HN2001000007A (en) |
| HR (1) | HRP20020675A2 (en) |
| HU (1) | HUP0203965A3 (en) |
| IL (1) | IL150730A0 (en) |
| IS (1) | IS6474A (en) |
| MA (1) | MA27589A1 (en) |
| MX (1) | MXPA02007058A (en) |
| MY (1) | MY136604A (en) |
| NO (1) | NO20022117L (en) |
| NZ (1) | NZ518531A (en) |
| OA (1) | OA12160A (en) |
| PA (1) | PA8509901A1 (en) |
| PE (1) | PE20011334A1 (en) |
| PL (1) | PL357590A1 (en) |
| SK (1) | SK10052002A3 (en) |
| SV (1) | SV2002000293A (en) |
| UA (1) | UA75880C2 (en) |
| WO (1) | WO2001053268A2 (en) |
| YU (1) | YU54202A (en) |
| ZA (1) | ZA200203040B (en) |
Cited By (153)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002016348A1 (en) * | 2000-08-09 | 2002-02-28 | Astrazeneca Ab | Antiangiogenic bicyclic derivatives |
| WO2002022586A1 (en) * | 2000-09-18 | 2002-03-21 | Bayer Aktiengesellschaft | Indazoles having an action similar to that of a thyroid hormone, method for the production thereof, and their use in medicaments |
| WO2002083648A1 (en) * | 2001-04-16 | 2002-10-24 | Eisai Co., Ltd. | Novel 1h-indazole compound |
| WO2003004488A1 (en) * | 2001-07-03 | 2003-01-16 | Chiron Corporation | Indazole benzimidazole compounds as tyrosine and serine/threonine kinase inhibitors |
| FR2827861A1 (en) * | 2001-07-27 | 2003-01-31 | Aventis Pharma Sa | New aryl-substituted indole, isoindole and indazole compounds, useful for treating cell proliferation disorders e.g. cancer, inhibit polymerization of tubulin |
| WO2003028720A1 (en) * | 2001-09-26 | 2003-04-10 | Pharmacia Italia S.P.A. | Aminoindazole derivatives active as kinase inhibitors, process for their preparation and pharmaceutical compositions containing them |
| WO2003032984A1 (en) * | 2001-10-19 | 2003-04-24 | Ortho-Mcneil Pharmaceutical,Inc. | 2-phenyl benzimidazoles and imidazo-[4,5]-pyridines as cdsi/chk2-inhibitors and adjuvants to chemotherapy or radiation therapy in the treatment of cancer |
| WO2003035065A1 (en) * | 2001-10-26 | 2003-05-01 | Aventis Pharmaceuticals Inc | Benzimidazoles and analogues and their use as protein kinases inhibitors |
| WO2003035644A1 (en) * | 2001-10-26 | 2003-05-01 | Aventis Pharma S.A. | Benzimidazole derivatives and their use as kdr protein kinase inhibitors |
| FR2831537A1 (en) * | 2001-10-26 | 2003-05-02 | Aventis Pharma Sa | Composition useful for treating inflammatory disease, cancer, chronic obstructive pulmonary disease, asthma, allergic rhinitis, or atopic dermatitis comprises benzimidazoles derivatives |
| EP1313711A2 (en) * | 2000-07-31 | 2003-05-28 | Signal Pharmaceuticals, Inc. | Indazole derivatives as jnk inhibitors |
| WO2004014864A1 (en) * | 2002-08-10 | 2004-02-19 | Astex Technology Limited | 1h-indazole-3-carboxamide compounds as cyclin dependent kinases (cdk) inhibitors |
| WO2004014368A1 (en) * | 2002-08-12 | 2004-02-19 | Sugen, Inc. | 3-pyrrolyl-pyridopyrazoles and 3-pyrrolyl-indazoles as novel kinase inhibitors |
| JP2004075673A (en) * | 2002-06-19 | 2004-03-11 | Mitsubishi Chemicals Corp | Compound and organic electroluminescent element using the same |
| WO2004050088A1 (en) * | 2002-12-03 | 2004-06-17 | Kyowa Hakko Kogyo Co., Ltd. | Jnk inhibitor |
| WO2004056806A1 (en) * | 2002-12-19 | 2004-07-08 | Pfizer Inc. | 2-(1h-indazol-6-ylamino)-benzamide compounds as protein kinases inhibitors useful for the treatment of ophtalmic diseases |
| US6770643B2 (en) | 1999-12-24 | 2004-08-03 | Aventis Pharma Limited | Azaindoles |
| WO2004076450A1 (en) * | 2003-02-27 | 2004-09-10 | J. Uriach Y Compañia S.A. | Pyrazolopyridine derivates |
| FR2854159A1 (en) * | 2003-04-25 | 2004-10-29 | Aventis Pharma Sa | New 2-pyrazolyl or indazolyl-indole derivatives, useful for treating e.g. angiogenic or fibrinolytic diseases, act by inhibiting protein kinases |
| WO2005002552A2 (en) | 2003-07-03 | 2005-01-13 | Astex Therapeutics Limited | Benzimidazole derivatives and their use as protein kinases inhibitors |
| EP1496894A1 (en) * | 2002-03-29 | 2005-01-19 | Carlsbad Technology Inc | Angiogenesis inhibitors |
| WO2005007635A2 (en) * | 2003-07-18 | 2005-01-27 | Sigma-Tau Industrie Farmaceutiche Riunite S.P.A. | Combretastatin derivatives with cytotoxic action |
| WO2005009997A1 (en) * | 2003-07-30 | 2005-02-03 | Pfizer Inc. | 3,5 disubstituted indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| WO2005012256A1 (en) | 2003-07-22 | 2005-02-10 | Astex Therapeutics Limited | 3, 4-disubstituted 1h-pyrazole compounds and their use as cyclin dependent kinases (cdk) and glycogen synthase kinase-3 (gsk-3) modulators |
| WO2005014554A1 (en) * | 2003-08-08 | 2005-02-17 | Astex Therapeutics Limited | 1h-indazole-3-carboxamide compounds as mapkap kinase modulators |
| EP1512401A1 (en) * | 2003-09-03 | 2005-03-09 | Yung Shin Pharmaceutical Ind. Co., Ltd. | Enhancement of bone growth and inhibition of bone resorption |
| EP1512400A1 (en) * | 2003-09-03 | 2005-03-09 | Carlsbad Technology Inc | Treatment of neurodegenerative diseases |
| WO2005026175A1 (en) * | 2003-09-08 | 2005-03-24 | Aventis Pharmaceuticals Inc. | Thienopyrazoles |
| WO2005035506A1 (en) * | 2003-10-15 | 2005-04-21 | Ube Industries, Ltd. | Novel indazole derivative |
| US6897207B2 (en) | 2001-06-21 | 2005-05-24 | Aventis Pharmaceuticals Inc. | Azaindoles |
| US6897208B2 (en) | 2001-10-26 | 2005-05-24 | Aventis Pharmaceuticals Inc. | Benzimidazoles |
| WO2005094823A1 (en) * | 2004-03-30 | 2005-10-13 | Kyowa Hakko Kogyo Co., Ltd. | Flt-3 INHIBITOR |
| WO2005103036A1 (en) * | 2004-04-23 | 2005-11-03 | Biofocus Discovery Ltd | Pyrimidin-4-yl-1h-indazol-5yl-amines as chk1 kinases inhibitors |
| EP1640002A1 (en) * | 2004-08-30 | 2006-03-29 | Yung Shin Pharmaceutical Ind. Co., Ltd. | Indazole derivatives for inhibition of angiogenesis |
| WO2006054143A1 (en) * | 2004-11-17 | 2006-05-26 | Pfizer Inc. | Polymorphs of {5-[3-(4,6-difluoro-1h-benzoimidazol-2-yl)-1h-indazol-5-yl]-4-methyl-pyridin-3-ylmethyl}-ethyl-amine |
| WO2006039718A3 (en) * | 2004-10-01 | 2006-07-13 | Amgen Inc | Aryl nitrogen-containing bicyclic compounds and their use as kinase inhibitors |
| WO2006077428A1 (en) | 2005-01-21 | 2006-07-27 | Astex Therapeutics Limited | Pharmaceutical compounds |
| WO2006077424A1 (en) | 2005-01-21 | 2006-07-27 | Astex Therapeutics Limited | Pharmaceutical compounds |
| WO2006086705A1 (en) * | 2005-02-10 | 2006-08-17 | Aventis Pharmaceuticals Inc. | Substituted bis aryl and heteroaryl compounds as selective 5ht2a antagonists |
| WO2006105262A1 (en) | 2005-03-29 | 2006-10-05 | Icos Corporation | HETEROARYL UREA DERIVATIVES USEFUL FOR INHIBITING CHKl |
| US7132440B2 (en) | 2003-04-17 | 2006-11-07 | Janssen Pharmaceutica, N.V. | Substituted benzimidazoles and imidazo-[4,5]-pyridines |
| WO2006125130A1 (en) | 2005-05-17 | 2006-11-23 | Novartis Ag | Methods for synthesizing heterocyclic compounds |
| WO2007058626A1 (en) * | 2005-11-16 | 2007-05-24 | S*Bio Pte Ltd | Indazole compounds |
| WO2007107346A1 (en) * | 2006-03-23 | 2007-09-27 | F. Hoffmann-La Roche Ag | Substituted indazole derivatives, their manufacture and use as pharmaceutical agents |
| CN100354275C (en) * | 2003-02-27 | 2007-12-12 | 帕劳制药股份有限公司 | Pyrazolopyridine derivates |
| WO2008001886A1 (en) | 2006-06-30 | 2008-01-03 | Kyowa Hakko Kirin Co., Ltd. | Aurora inhibitor |
| WO2008001885A1 (en) | 2006-06-30 | 2008-01-03 | Kyowa Hakko Kirin Co., Ltd. | Abl KINASE INHIBITOR |
| US7351729B2 (en) * | 2002-03-08 | 2008-04-01 | Signal Pharmaceuticals, Llc | JNK inhibitors for use in combination therapy for treating or managing proliferative disorders and cancers |
| WO2008044045A1 (en) | 2006-10-12 | 2008-04-17 | Astex Therapeutics Limited | Pharmaceutical combinations |
| WO2008044041A1 (en) | 2006-10-12 | 2008-04-17 | Astex Therapeutics Limited | Pharmaceutical combinations |
| WO2008111441A1 (en) | 2007-03-05 | 2008-09-18 | Kyowa Hakko Kirin Co., Ltd. | Pharmaceutical composition |
| US7429609B2 (en) * | 2002-05-31 | 2008-09-30 | Eisai R & D Management Co., Ltd. | Pyrazole compound and medicinal composition containing the same |
| WO2008154241A1 (en) * | 2007-06-08 | 2008-12-18 | Abbott Laboratories | 5-heteroaryl substituted indazoles as kinase inhibitors |
| US7470807B2 (en) | 2003-08-14 | 2008-12-30 | Asahi Kasei Pharma Corporation | Substituted arylalkanoic acid derivatives and use thereof |
| US7473701B2 (en) | 2002-03-11 | 2009-01-06 | Aventis Pharma S.A. | Substituted indazoles, compositions containing them, method of production and use |
| US7541367B2 (en) | 2005-05-31 | 2009-06-02 | Janssen Pharmaceutica, N.V. | 3-benzoimidazolyl-pyrazolopyridines useful in treating kinase disorders |
| US7598268B2 (en) | 2000-09-11 | 2009-10-06 | Novartis Vaccines & Diagnostics, Inc. | Quinolinone derivatives |
| US7605272B2 (en) | 2005-01-27 | 2009-10-20 | Kyowa Hakko Kirin Co., Ltd. | IGF-1R inhibitor |
| US7642278B2 (en) | 2001-07-03 | 2010-01-05 | Novartis Vaccines And Diagnostics, Inc. | Indazole benzimidazole compounds |
| AU2004260759B2 (en) * | 2003-07-30 | 2010-04-22 | Kyowa Hakko Kirin Co., Ltd. | Protein kinase inhibitors |
| WO2010127329A1 (en) * | 2009-05-01 | 2010-11-04 | Aerie Pharmaceuticals, Inc. | Dual-action inhibitors and methods of using same |
| WO2011057054A1 (en) | 2009-11-06 | 2011-05-12 | Vitae Pharmaceuticals, Inc. | Aryl- and heteroarylcarbonyl derivatives of hexahydroindenopyridine and octahydrobenzoquinoline |
| US8034943B2 (en) | 2005-07-11 | 2011-10-11 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| EP2395000A1 (en) | 2004-12-30 | 2011-12-14 | Astex Therapeutics Limited | Benzimidazole compounds that modulate the activity of CDK, GSK and aurora kinases |
| WO2012061708A1 (en) | 2010-11-05 | 2012-05-10 | Boehringer Ingelheim International Gmbh | Crystalline forms of hydrochloride salt of (4a-r,9a-s) -1- (1h - benzoimidazole- 5 -carbonyl) -2, 3, 4, 4a, 9, 9a - hexahydro -1h- indeno [2, 1 -b] pyridine- 6 -carbonitrile and their use as hsd 1 inhibitors |
| WO2012078777A1 (en) * | 2010-12-09 | 2012-06-14 | Amgen Inc. | Bicyclic compounds as pim inhibitors |
| US8252812B2 (en) | 2009-08-10 | 2012-08-28 | Samumed, Llc | Indazole inhibitors of the WNT signal pathway and therapeutic uses thereof |
| US20120277229A1 (en) * | 2011-04-01 | 2012-11-01 | Bearss David J | Substituted 3-(1h-benzo[d]imidazol-2-yl)-1h-indazole analogs as inhibitors of the pdk1 kinase |
| US8334314B2 (en) | 2008-04-28 | 2012-12-18 | Asahi Kasei Pharma Corporation | Phenylpropionic acid derivative and use thereof |
| US8357699B2 (en) | 2007-01-10 | 2013-01-22 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| WO2013040215A1 (en) | 2011-09-14 | 2013-03-21 | Samumed, Llc | Indazole-3-carboxamides and their use as wnt/b-catenin signaling pathway inhibitors |
| US8450344B2 (en) | 2008-07-25 | 2013-05-28 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US8450340B2 (en) | 2009-12-21 | 2013-05-28 | Samumed, Llc | 1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| US8455514B2 (en) | 2008-01-17 | 2013-06-04 | Aerie Pharmaceuticals, Inc. | 6-and 7-amino isoquinoline compounds and methods for making and using the same |
| WO2013151708A1 (en) | 2012-04-04 | 2013-10-10 | Samumed, Llc | Indazole inhibitors of the wnt signal pathway and therapeutic uses thereof |
| WO2013166396A2 (en) | 2012-05-04 | 2013-11-07 | Samumed, Llc | 1h-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| US8609717B2 (en) | 2010-08-18 | 2013-12-17 | Samumed, Llc | β- and γ-diketones and γ-hydroxyketones as WNT/β-catenin signaling pathway activators |
| US8648069B2 (en) | 2007-06-08 | 2014-02-11 | Abbvie Inc. | 5-substituted indazoles as kinase inhibitors |
| US8658649B2 (en) | 2006-09-11 | 2014-02-25 | Sanofi | Kinase inhibitor |
| WO2014041518A1 (en) * | 2012-09-14 | 2014-03-20 | Glenmark Pharmaceuticals S.A. | Thienopyrrole derivatives as itk inhibitors |
| EP2762475A1 (en) | 2003-11-07 | 2014-08-06 | Novartis Vaccines and Diagnostics, Inc. | Pharmaceutically acceptable salts of quinolinone compounds and their medical use |
| US8809326B2 (en) | 2006-09-20 | 2014-08-19 | Aerie Pharmaceuticals, Inc. | Isoquinolinone Rho kinase inhibitors |
| WO2014130869A1 (en) | 2013-02-22 | 2014-08-28 | Samumed, Llc | Gamma-diketones as wnt/beta -catenin signaling pathway activators |
| EP2772257A1 (en) | 2005-11-29 | 2014-09-03 | Novartis AG | Formulations of quinolinones |
| WO2014134772A1 (en) * | 2013-03-04 | 2014-09-12 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US8999968B2 (en) | 2009-04-06 | 2015-04-07 | University Health Network | Kinase inhibitors and method of treating cancer with same |
| US9139563B2 (en) | 2010-04-06 | 2015-09-22 | University Health Network | Kinase inhibitors and method of treating cancer |
| US9321756B2 (en) | 2011-03-22 | 2016-04-26 | Amgen Inc. | Azole compounds as PIM inhibitors |
| US20160250220A1 (en) | 2013-10-18 | 2016-09-01 | University Health Network | Treatment for pancreatic cancer |
| US9440952B2 (en) | 2013-03-04 | 2016-09-13 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US9469606B2 (en) | 2011-10-25 | 2016-10-18 | The General Hospital Corporation | Wnt/b-catenin inhibitors and methods of use |
| US9493440B2 (en) | 2013-03-04 | 2016-11-15 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US9643927B1 (en) | 2015-11-17 | 2017-05-09 | Aerie Pharmaceuticals, Inc. | Process for the preparation of kinase inhibitors and intermediates thereof |
| US9657016B2 (en) | 2014-09-08 | 2017-05-23 | Samumed, Llc | 3-(1h-benzo[d]imidazol-2-yl)-1h-pyrazolo[3,4-c]pyridine and therapeutic uses thereof |
| US9688654B2 (en) | 2013-03-04 | 2017-06-27 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US9738638B2 (en) | 2014-09-08 | 2017-08-22 | Samumed, Llc | 2-(1H-indazol-3-yl)-3H-imidazo[4,5-B]pyridine and therapeutic uses thereof |
| US9758531B2 (en) | 2014-09-08 | 2017-09-12 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridine and therapeutic uses thereof |
| US9763951B2 (en) | 2014-09-08 | 2017-09-19 | Samumed, Llc | 3-(3H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US9795550B2 (en) | 2014-08-20 | 2017-10-24 | Samumed, Llc | Gamma-diketones for treatment and prevention of aging skin and wrinkles |
| WO2017189823A2 (en) | 2016-04-27 | 2017-11-02 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| WO2017189829A1 (en) | 2016-04-27 | 2017-11-02 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US9844536B2 (en) | 2014-09-08 | 2017-12-19 | Samumed, Llc | 3-(1H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US9849122B2 (en) | 2013-03-15 | 2017-12-26 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US9889140B2 (en) | 2014-09-08 | 2018-02-13 | Samumed, Llc | 3-(1H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridine and therapeutic uses thereof |
| US9908867B2 (en) | 2013-01-08 | 2018-03-06 | Samumed, Llc | 3-(benzoimidazol-2-yl)-indazole inhibitors of the Wnt signaling pathway and therapeutic uses thereof |
| CN107922349A (en) * | 2015-08-07 | 2018-04-17 | 哈尔滨珍宝制药有限公司 | Vinyl compound as FGFR and VEGFR inhibitor |
| WO2018075858A1 (en) | 2016-10-21 | 2018-04-26 | Samumed, Llc | Methods of using indazole-3-carboxamides and their use as wnt/b-catenin signaling pathway inhibitors |
| US10072004B2 (en) | 2016-06-01 | 2018-09-11 | Samumed, Llc | Process for preparing N-(5-(3-(7-(3-fluorophenyl)-3H-imidazo [4,5-C]pyridin-2-yl)-1H-indazol-5-yl)pyridin-3-yl)-3-methylbutanamide |
| US10081631B2 (en) | 2014-09-08 | 2018-09-25 | Samumed, Llc | 2-(1H-indazol-3-yl)-1H-imidazo[4,5-C]pyridine and therapeutic uses thereof |
| US10118920B2 (en) | 2015-04-20 | 2018-11-06 | Cellcentric Ltd | Isoxazolyl substituted benzimidazoles |
| US10166218B2 (en) | 2015-08-03 | 2019-01-01 | Samumed, Llc | 3-(1H-indol-2-yl)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10188634B2 (en) | 2015-08-03 | 2019-01-29 | Samumed, Llc | 3-(3H-imidazo[4,5-C]pyridin-2-yl)-1 H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10195185B2 (en) | 2015-08-03 | 2019-02-05 | Samumed, Llc | 3-(1H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10206908B2 (en) | 2015-08-03 | 2019-02-19 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-YL)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10206909B2 (en) | 2015-08-03 | 2019-02-19 | Samumed, Llc | 3-(1H-pyrrolo[2,3-B]pyridin-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10226448B2 (en) | 2015-08-03 | 2019-03-12 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridines and therapeutic uses thereof |
| US10226453B2 (en) | 2015-08-03 | 2019-03-12 | Samumed, Llc | 3-(1H-indol-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10231956B2 (en) | 2015-08-03 | 2019-03-19 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-YL)-1 H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10239864B2 (en) | 2014-07-24 | 2019-03-26 | Beta Pharma, Inc. | 2-H-indazole derivatives as cyclin-dependent kinase (CDK) inhibitors and therapeutic uses thereof |
| WO2019079626A1 (en) | 2017-10-19 | 2019-04-25 | Samumed, Llc | 6-(5-membered heteroaryl)isoquinolin-3-yl carboxamides and preparation and use thereof |
| WO2019084497A1 (en) | 2017-10-27 | 2019-05-02 | Samumed, Llc | 6-(6-membered heteroaryl & aryl)isoquinolin-3-yl carboxamides and preparation and use thereof |
| WO2019084496A1 (en) | 2017-10-27 | 2019-05-02 | Samumed, Llc | 6-(5-membered heteroaryl)isoquinolin-3-yl-(5-membered heteroaryl) carboxamides and preparation and use thereof |
| WO2019089835A1 (en) | 2017-10-31 | 2019-05-09 | Samumed, Llc | Diazanaphthalen-3-yl carboxamides and preparation and use thereof |
| US10285982B2 (en) | 2015-08-03 | 2019-05-14 | Samumed, Llc | 3-(1H-pyrrolo[2,3-B]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10285983B2 (en) | 2015-08-03 | 2019-05-14 | Samumed, Llc | 3-(1H-pyrrolo[2,3-B]pyridin-2-yl)-1H-pyrazolo[3,4-B] pyridines and therapeutic uses thereof |
| EP3483142A4 (en) * | 2016-07-05 | 2019-05-15 | Guangzhou Maxinovel Pharmaceuticals Co., Ltd. | Aromatic acetylene or aromatic ethylene compound, intermediate, preparation method, pharmaceutical composition and use thereof |
| US10329309B2 (en) | 2015-08-03 | 2019-06-25 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10350199B2 (en) | 2015-08-03 | 2019-07-16 | Samumed, Llc | 3-(1h-pyrrolo[2,3-b]pyridin-2-yl)-1h-indazoles and therapeutic uses thereof |
| US10383861B2 (en) | 2015-08-03 | 2019-08-20 | Sammumed, LLC | 3-(1H-pyrrolo[2,3-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10392383B2 (en) | 2015-08-03 | 2019-08-27 | Samumed, Llc | 3-(1H-benzo[d]imidazol-2-yl)-1H-pyrazolo[4,3-b]pyridines and therapeutic uses thereof |
| WO2019165192A1 (en) | 2018-02-23 | 2019-08-29 | Samumed, Llc | 5-heteroaryl substituted indazole-3-carboxamides and preparation and use thereof |
| US10428065B2 (en) | 2015-04-20 | 2019-10-01 | Cellcentric Ltd | Isoxazolyl substituted imidazopyridines |
| US10463651B2 (en) | 2015-08-03 | 2019-11-05 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-YL)-1H-indazoles and therapeutic uses thereof |
| WO2019241540A1 (en) | 2018-06-15 | 2019-12-19 | Samumed, Llc | Indazole containing macrocycles and therapeutic uses thereof |
| US10519169B2 (en) | 2015-08-03 | 2019-12-31 | Samumed, Llc | 3-(1H-pyrrolo[2,3-C]pyridin-2-yl)-1 H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10533020B2 (en) | 2014-09-08 | 2020-01-14 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1 H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US10544139B2 (en) | 2015-11-06 | 2020-01-28 | Samumed, Llc | Treatment of osteoarthritis |
| US10550087B2 (en) | 2015-11-17 | 2020-02-04 | Aerie Pharmaceuticals, Inc. | Process for the preparation of kinase inhibitors and intermediates thereof |
| US10604512B2 (en) | 2015-08-03 | 2020-03-31 | Samumed, Llc | 3-(1H-indol-2-yl)-1H-indazoles and therapeutic uses thereof |
| WO2020150545A1 (en) | 2019-01-17 | 2020-07-23 | Samumed, Llc | Pyrazole derivatives as modulators of the wnt/b-catenin signaling pathway |
| US10758523B2 (en) | 2016-11-07 | 2020-09-01 | Samumed, Llc | Single-dose, ready-to-use injectable formulations |
| US10858339B2 (en) | 2017-03-31 | 2020-12-08 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| CN112724134A (en) * | 2021-01-14 | 2021-04-30 | 复旦大学 | Azaindazole bipyridine derivative myeloid cell proliferation inhibitor, preparation method and application thereof in pharmacy |
| WO2021125229A1 (en) * | 2019-12-17 | 2021-06-24 | 富士フイルム株式会社 | Indazole compound or salt thereof, and pharmaceutical composition |
| WO2021121420A1 (en) * | 2019-12-20 | 2021-06-24 | 江苏凯迪恩医药科技有限公司 | Benzopyrazole compound and intermediate, preparation method, and application thereof |
| WO2021168341A1 (en) | 2020-02-21 | 2021-08-26 | Samumed, Llc | Solid forms of 1-(5-(3-(7-(3-fluorophenyl)-3h-imidazo[4,5-c]pyridin-2-yl)-1h-pyrazolo[3,4-b]pyridin-5-yl)pyridin-3-yl)-n,n-dimethylmethanamine |
| US11384048B2 (en) | 2017-12-29 | 2022-07-12 | Guangzhou Maxinovel Pharmaceuticals Co., Ltd. | Aromatic vinyl or aromatic ethyl derivative, preparation method therefor, intermediate, pharmaceutical composition, and application |
| US11389441B2 (en) | 2016-08-31 | 2022-07-19 | Aerie Pharmaceuticals, Inc. | Ophthalmic compositions |
| US11427563B2 (en) | 2018-09-14 | 2022-08-30 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| WO2023240138A1 (en) * | 2022-06-08 | 2023-12-14 | Blossomhill Therapeutics, Inc. | Indazole containing macrocycles and their use |
| EP4056558A4 (en) * | 2019-11-06 | 2023-12-27 | Jinan University | INDAZOLE COMPOUND, PHARMACEUTICAL COMPOSITION THEREOF AND ITS APPLICATIONS |
| US12071428B2 (en) | 2020-12-30 | 2024-08-27 | Tyra Biosciences, Inc. | Indazole compounds as kinase inhibitors |
| US12264149B2 (en) | 2021-12-30 | 2025-04-01 | Tyra Biosciences, Inc. | Indazole compounds as kinase inhibitors |
Families Citing this family (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020150921A1 (en) * | 1996-02-09 | 2002-10-17 | Francis Barany | Detection of nucleic acid sequence differences using the ligase detection reaction with addressable arrays |
| YU54202A (en) * | 2000-01-18 | 2006-01-16 | Agouron Pharmaceuticals Inc. | Indazole compounds,pharmaceutical compositions,and methods for mediating or inhibiting cell proliferation |
| US7211594B2 (en) | 2000-07-31 | 2007-05-01 | Signal Pharmaceuticals, Llc | Indazole compounds and compositions thereof as JNK inhibitors and for the treatment of diseases associated therewith |
| US20050009876A1 (en) * | 2000-07-31 | 2005-01-13 | Bhagwat Shripad S. | Indazole compounds, compositions thereof and methods of treatment therewith |
| US7074805B2 (en) * | 2002-02-20 | 2006-07-11 | Abbott Laboratories | Fused azabicyclic compounds that inhibit vanilloid receptor subtype 1 (VR1) receptor |
| US6867320B2 (en) * | 2002-02-21 | 2005-03-15 | Asahi Kasei Pharma Corporation | Substituted phenylalkanoic acid derivatives and use thereof |
| WO2003102151A2 (en) * | 2002-05-30 | 2003-12-11 | Celgene Corporation | Modulating cell differentiation and treating myeloprolifertive disorders with jnk/mkk inhibitors |
| US20040087642A1 (en) * | 2002-10-24 | 2004-05-06 | Zeldis Jerome B. | Methods of using and compositions comprising a JNK inhibitor for the treatment, prevention, management and/or modification of pain |
| US20050019366A1 (en) * | 2002-12-31 | 2005-01-27 | Zeldis Jerome B. | Drug-coated stents and methods of use therefor |
| US6933311B2 (en) * | 2003-02-11 | 2005-08-23 | Abbott Laboratories | Fused azabicyclic compounds that inhibit vanilloid receptor subtype 1 (VR1) receptor |
| BRPI0407618A (en) * | 2003-02-21 | 2006-02-21 | Pfizer | cycloalkyl-substituted amino thiazole derivatives containing n and pharmaceutical compositions for inhibiting cell proliferation and methods for their use |
| US7015233B2 (en) * | 2003-06-12 | 2006-03-21 | Abbott Laboratories | Fused compounds that inhibit vanilloid subtype 1 (VR1) receptor |
| MXPA05011420A (en) * | 2003-07-30 | 2005-12-12 | Kyowa Hakko Kogyo Kk | Indazole derivatives. |
| US20050090529A1 (en) * | 2003-07-31 | 2005-04-28 | Pfizer Inc | 3,5 Disubstituted indazole compounds with nitrogen-bearing 5-membered heterocycles, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| CN1829709A (en) * | 2003-08-01 | 2006-09-06 | 健亚生物科技公司 | Bicyclic imidazol derivatives against flaviviridae |
| US20050113576A1 (en) * | 2003-08-05 | 2005-05-26 | Chih-Hung Lee | Fused azabicyclic compounds that inhibit vanilloid receptor subtype 1 (VR1) receptor |
| US6984652B2 (en) * | 2003-09-05 | 2006-01-10 | Warner-Lambert Company Llc | Gyrase inhibitors |
| KR20060124610A (en) * | 2003-11-06 | 2006-12-05 | 셀진 코포레이션 | Methods of using uncN inhibitors to treat and manage asbestos-related diseases or disorders and compositions comprising the materials |
| US20060179711A1 (en) * | 2003-11-17 | 2006-08-17 | Aerogrow International, Inc. | Devices and methods for growing plants |
| EP1692128A1 (en) * | 2003-11-19 | 2006-08-23 | Signal Pharmaceuticals LLC | Indazole compounds and methods of use thereof as protein kinase inhibitors |
| US7488817B2 (en) * | 2004-02-02 | 2009-02-10 | The Trustees Of The University Of Pennsylvania | Metal complex protein kinase inhibitors |
| WO2005094830A1 (en) * | 2004-03-30 | 2005-10-13 | Pfizer Products Inc. | Combinations of signal transduction inhibitors |
| CN101027053A (en) * | 2004-07-27 | 2007-08-29 | 诺瓦提斯公司 | Inhibitors of HSP90 |
| FR2876584B1 (en) * | 2004-10-15 | 2007-04-13 | Centre Nat Rech Scient Cnrse | USE OF KENPAULLONE FOR THE PRODUCTION OF MEDICAMENTS FOR THE TREATMENT OF MUCOVISCIDOSIS AND DISEASES ASSOCIATED WITH A DEFECT OF ADDRESSING PROTEINS IN CELLS |
| US20060116519A1 (en) * | 2004-11-17 | 2006-06-01 | Agouron Pharmaceuticals, Inc. | Synthesis of 5-bromo-4-methyl-pyridin-3-ylmethyl)-ethyl-carbamic acid tert-butyl ester |
| EP1827422A2 (en) * | 2004-11-23 | 2007-09-05 | Celgene Corporation | Jnk inhibitors for the treatment of cns injury |
| US20070004777A1 (en) * | 2005-03-23 | 2007-01-04 | Bhagwat Shripad S | Methods for treating or preventing acute myelogenous leukemia |
| WO2006130297A2 (en) * | 2005-04-29 | 2006-12-07 | Celgene Corporation | Solid forms of 1-( 5-(ih-i , 2 , 4 -triazol- 5 -yl)(1h-indazol-3-yl))-3-(2-piperidylethoxy)benzene |
| KR20070077468A (en) * | 2006-01-23 | 2007-07-26 | 크리스탈지노믹스(주) | Imidazopyridine derivatives that inhibit protein kinase activity, preparation method thereof, and pharmaceutical composition containing same |
| AU2007213452A1 (en) * | 2006-02-10 | 2007-08-16 | Biomarin Iga Limited | Treatment of Duchenne muscular dystrophy |
| JPWO2008020606A1 (en) * | 2006-08-16 | 2010-01-07 | 協和発酵キリン株式会社 | Angiogenesis inhibitor |
| MX2009002018A (en) * | 2006-08-25 | 2009-03-05 | Abbott Lab | Indazole derivatives that inhibit trpv1 and uses thereof. |
| US20100022517A1 (en) * | 2006-12-18 | 2010-01-28 | Richards Lori A | Ophthalmic formulation of rho kinase inhibitor compound |
| US8071779B2 (en) | 2006-12-18 | 2011-12-06 | Inspire Pharmaceuticals, Inc. | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
| WO2008079683A2 (en) * | 2006-12-20 | 2008-07-03 | Abbott Laboratories | N- (5, 6, 7, 8-tetrahydronaphthalen-1-yl) urea derivatives and related compounds as trpv1 vanilloid receptor antagonists for the treatment of pain |
| US8063089B2 (en) * | 2007-02-28 | 2011-11-22 | Arizona Board Of Regents On Behalf Of The University Of Arizona | Inhibitors of nucleophosmin (NPM) and methods for inducing apoptosis |
| ES2439705T3 (en) * | 2007-10-25 | 2014-01-24 | Genentech, Inc. | Process for the preparation of thienopyrimidine compounds |
| JP2011515414A (en) * | 2008-03-20 | 2011-05-19 | アボット・ラボラトリーズ | Methods of making central nervous system drugs that are TRPV1 antagonists |
| US8106039B2 (en) * | 2008-04-30 | 2012-01-31 | The Trustees Of The University Of Pennsylvania | Metal complex phosphatidyl-inositol-3-kinase inhibitors |
| CA2770320A1 (en) * | 2009-08-10 | 2011-02-17 | Epitherix, Llc | Indazoles as wnt/b-catenin signaling pathway inhibitors and therapeutic uses thereof |
| WO2012020725A1 (en) * | 2010-08-10 | 2012-02-16 | 塩野義製薬株式会社 | Heterocyclic derivative having npy y5 receptor antagonism |
| CN104370889A (en) * | 2013-01-24 | 2015-02-25 | 韩冰 | Intraocular tension reducing compound and preparation method and use thereof |
| US9737510B2 (en) | 2013-07-31 | 2017-08-22 | Council Of Scientific & Industrial Research | Indazole compounds and a process for the preparation thereof |
| GB201511382D0 (en) | 2015-06-29 | 2015-08-12 | Imp Innovations Ltd | Novel compounds and their use in therapy |
| WO2018013430A2 (en) | 2016-07-12 | 2018-01-18 | Arisan Therapeutics Inc. | Heterocyclic compounds for the treatment of arenavirus infection |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1376600A (en) * | 1971-02-15 | 1974-12-04 | Agfa Gevaert | Photographic developer compositions |
| US3994890A (en) * | 1974-01-31 | 1976-11-30 | Chugai Seiyaku Kabushiki Kaisha | 1-Aminoalkyl, 3-phenyl indazoles |
| US4051145A (en) * | 1974-03-29 | 1977-09-27 | Produits Chimiques Ugine Kuhlmann | Fluorinated indazole derivatives and a process for their preparation |
| EP0023633A1 (en) * | 1979-07-16 | 1981-02-11 | Chugai Seiyaku Kabushiki Kaisha | Pyrazoloindazole derivatives and process for preparing the same and pharmaceutical compositions |
| US4978603A (en) * | 1987-12-14 | 1990-12-18 | Fuji Photo Film Co., Ltd. | Image forming process comprising developing fine grain silver halide emulsion with a hydroquinone developer |
| EP0494774A1 (en) * | 1991-01-11 | 1992-07-15 | MERCK SHARP & DOHME LTD. | Indazole-substituted fivemembered heteroaromatic compounds |
| WO1996020192A1 (en) * | 1994-12-23 | 1996-07-04 | Glaxo Group Limited | Piperidineacetic acid derivatives useful as fibrinogen antagonist agent |
| US5760028A (en) * | 1995-12-22 | 1998-06-02 | The Dupont Merck Pharmaceutical Company | Integrin receptor antagonists |
| WO1998043969A1 (en) * | 1997-03-31 | 1998-10-08 | Dupont Pharmaceuticals Company | Indazoles of cyclic ureas useful as hiv protease inhibitors |
| EP0904769A1 (en) * | 1997-08-25 | 1999-03-31 | L'oreal | Keratin fibre dye compositions containing Indazole amine derivatives and dyeing method |
| WO1999015500A1 (en) * | 1997-09-05 | 1999-04-01 | Glaxo Group Limited | Substituted oxindole derivatives as protein tyrosine kinase and as protein serine/threonine kinase inhibitors |
| WO1999017769A1 (en) * | 1997-10-06 | 1999-04-15 | Basf Aktiengesellschaft | INDENO[1,2-c]-, NAPHTHO[1,2-c]- AND BENZO[6,7]CYCLOHEPTA[1,2-c]PYRAZOLE DERIVATIVES |
Family Cites Families (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH575397A5 (en) | 1971-11-09 | 1976-05-14 | Basf Ag | |
| ATE36787T1 (en) | 1982-09-17 | 1988-09-15 | Kudelski Sa | CONTROL DEVICE FOR AN ELECTRIC MOTOR. |
| JPS59228248A (en) | 1983-06-08 | 1984-12-21 | Konishiroku Photo Ind Co Ltd | Posi-posi silver halide photosensitive material |
| JPS604184A (en) * | 1983-06-23 | 1985-01-10 | Chugai Pharmaceut Co Ltd | Pyrazoloindazole derivative |
| JPH083564B2 (en) | 1986-07-25 | 1996-01-17 | 三菱化学株式会社 | Polarizing film |
| DE69024215T2 (en) | 1990-09-24 | 1996-07-18 | Agfa Gevaert Nv | Photographic direct positive silver halide material that can be handled in room lighting |
| US5208248A (en) * | 1991-01-11 | 1993-05-04 | Merck Sharpe & Dohme, Ltd. | Indazole-substituted five-membered heteroaromatic compounds |
| US5612360A (en) | 1992-06-03 | 1997-03-18 | Eli Lilly And Company | Angiotensin II antagonists |
| EP0674627A1 (en) | 1992-12-18 | 1995-10-04 | The Wellcome Foundation Limited | Pyrimidine, pyridine, pteridinone and indazole derivatives as enzyme inhibitors |
| US5625031A (en) | 1994-02-08 | 1997-04-29 | Bristol-Myers Squibb Company | Peptide inhibitors of the p33cdk2 and p34cdc2 cell cycle regulatory kinases and human papillomavirus E7 oncoprotein |
| US5631156A (en) | 1994-06-21 | 1997-05-20 | The University Of Michigan | DNA encoding and 18 KD CDK6 inhibiting protein |
| CN1165482A (en) | 1994-11-10 | 1997-11-19 | 科西雷佩蒂斯公司 | Pharmaceutical pyrazole compositions useful as inhibitors of protein kinases |
| TR199700749T1 (en) | 1995-02-02 | 1998-02-21 | Smithkline Beecham P.L.C. | Indole derivatives as 5-HT receptor antagonists. |
| WO1997003967A1 (en) | 1995-07-22 | 1997-02-06 | Rhone-Poulenc Rorer Limited | Substituted aromatic compounds and their pharmaceutical use |
| US5733920A (en) | 1995-10-31 | 1998-03-31 | Mitotix, Inc. | Inhibitors of cyclin dependent kinases |
| US6495526B2 (en) | 1996-01-23 | 2002-12-17 | Gpc Biotech, Inc. | Inhibitors of cell-cycle progression and uses related thereto |
| WO1997034876A1 (en) | 1996-03-15 | 1997-09-25 | Zeneca Limited | Cinnoline derivatives and use as medicine |
| GB9608435D0 (en) | 1996-04-24 | 1996-06-26 | Celltech Therapeutics Ltd | Chemical compounds |
| US5849733A (en) | 1996-05-10 | 1998-12-15 | Bristol-Myers Squibb Co. | 2-thio or 2-oxo flavopiridol analogs |
| US5821243A (en) | 1996-07-22 | 1998-10-13 | Viropharma Incorporated | Compounds compositions and methods for treating influenza |
| GB9621757D0 (en) | 1996-10-18 | 1996-12-11 | Ciba Geigy Ag | Phenyl-substituted bicyclic heterocyclyl derivatives and their use |
| DE69839338T2 (en) | 1997-02-05 | 2008-07-10 | Warner-Lambert Company Llc | PYRIDO (2,3-D) PYRIMIDINE AND 4-AMINO-PYRIMIDINE AS INHIBITORS OF CELLULAR PROLIFERATION |
| WO1998038168A1 (en) | 1997-02-27 | 1998-09-03 | Tanabe Seiyaku Co., Ltd. | Isoquinolinone derivatives, process for preparing the same, and their use as phosphodiesterase inhibitors |
| DE69832715T2 (en) | 1997-07-12 | 2007-01-11 | Cancer Research Technology Ltd. | CYCLINE-DEPENDENT KINASE INHIBITING PURE DERIVATIVES |
| GB9716324D0 (en) | 1997-08-01 | 1997-10-08 | Mead Corp | Packaging machine and method of carton set up |
| FR2767827A1 (en) | 1997-09-03 | 1999-02-26 | Adir | NOVEL INDOLE AND INDAZOLE DERIVATIVES, PROCESS FOR PREPARING THEM AND PHARMACEUTICAL COMPOSITIONS CONTAINING THE SAME |
| DE19744026A1 (en) * | 1997-10-06 | 1999-04-08 | Hoechst Marion Roussel De Gmbh | Pyrazole derivatives, their preparation and their use in medicinal products |
| US6569878B1 (en) | 1997-10-27 | 2003-05-27 | Agouron Pharmaceuticals Inc. | Substituted 4-amino-thiazol-2-yl compounds as cyclin-dependent kinase inhibitors |
| EA200000385A1 (en) | 1997-11-04 | 2000-10-30 | Пфайзер Продактс Инк. | THERAPEUTICALLY ACTIVE CONNECTIONS ON THE BASIS OF INDASOLIC BIOISOSTERIC SUBSTITUTION OF CATEHIN IN TYPE VI PHOSPHODYESTERASE INHIBITORS (PDE4) |
| BR9813926A (en) | 1997-11-04 | 2000-09-19 | Pfizer Prod Inc | Replacement of catechol bioisomer with indazole in therapeutically active compounds |
| US6040321A (en) | 1997-11-12 | 2000-03-21 | Bristol-Myers Squibb Company | Aminothiazole inhibitors of cyclin dependent kinases |
| US6479487B1 (en) | 1998-02-26 | 2002-11-12 | Aventis Pharmaceuticals Inc. | 6, 9-disubstituted 2-[trans-(4-aminocyclohexyl)amino] purines |
| PL344021A1 (en) | 1998-02-26 | 2001-09-24 | Aventis Pharma Inc | 6,9-disubstituted 2-[trans-(4- aminocyclohexyl) amino]purines |
| NZ504769A (en) | 1998-02-27 | 2005-04-29 | Pfizer Prod Inc | N-[(substituted five-membered di- or triaza diunsaturated ring) carbonyl] guanidine derivatives for the treatment of ischemia |
| BR9909597A (en) | 1998-04-21 | 2001-10-02 | Du Pont Pharm Co | Compound, pharmaceutical composition and method of treating cancer and proliferative diseases |
| YU54202A (en) * | 2000-01-18 | 2006-01-16 | Agouron Pharmaceuticals Inc. | Indazole compounds,pharmaceutical compositions,and methods for mediating or inhibiting cell proliferation |
| US6897231B2 (en) * | 2000-07-31 | 2005-05-24 | Signal Pharmaceuticals, Inc. | Indazole derivatives as JNK inhibitors and compositions and methods related thereto |
| AU2003259749A1 (en) * | 2002-08-12 | 2004-02-25 | Sugen, Inc. | 3-pyrrolyl-pyridopyrazoles and 3-pyrrolyl-indazoles as novel kinase inhibitors |
-
2000
- 2000-01-18 YU YU54202A patent/YU54202A/en unknown
-
2001
- 2001-01-17 MY MYPI20010207A patent/MY136604A/en unknown
- 2001-01-17 DO DO2001000120A patent/DOP2001000120A/en unknown
- 2001-01-17 PE PE2001000043A patent/PE20011334A1/en not_active Application Discontinuation
- 2001-01-18 AU AU29539/01A patent/AU785013B2/en not_active Ceased
- 2001-01-18 US US09/761,656 patent/US6555539B2/en not_active Expired - Fee Related
- 2001-01-18 AP APAP/P/2002/002564A patent/AP1609A/en active
- 2001-01-18 IL IL15073001A patent/IL150730A0/en unknown
- 2001-01-18 KR KR1020027009194A patent/KR20020073505A/en active IP Right Grant
- 2001-01-18 WO PCT/US2001/001477 patent/WO2001053268A2/en active IP Right Grant
- 2001-01-18 PA PA20018509901A patent/PA8509901A1/en unknown
- 2001-01-18 CO CO01003496A patent/CO5280070A1/en not_active Application Discontinuation
- 2001-01-18 HN HN2001000007A patent/HN2001000007A/en unknown
- 2001-01-18 DZ DZ013301A patent/DZ3301A1/en active
- 2001-01-18 NZ NZ518531A patent/NZ518531A/en unknown
- 2001-01-18 PL PL01357590A patent/PL357590A1/en not_active Application Discontinuation
- 2001-01-18 GT GT200100009A patent/GT200100009A/en unknown
- 2001-01-18 GE GE4848A patent/GEP20043363B/en unknown
- 2001-01-18 SK SK1005-2002A patent/SK10052002A3/en not_active Application Discontinuation
- 2001-01-18 MX MXPA02007058A patent/MXPA02007058A/en active IP Right Grant
- 2001-01-18 HU HU0203965A patent/HUP0203965A3/en unknown
- 2001-01-18 AR ARP010100234A patent/AR032438A1/en unknown
- 2001-01-18 OA OA1200200212A patent/OA12160A/en unknown
- 2001-01-18 CN CN01803552A patent/CN1394205A/en active Pending
- 2001-01-18 UA UA2002086633A patent/UA75880C2/en unknown
- 2001-01-18 CA CA002388885A patent/CA2388885A1/en not_active Abandoned
- 2001-01-18 EP EP01942620A patent/EP1250326A2/en not_active Withdrawn
- 2001-01-18 JP JP2001553270A patent/JP2003520273A/en not_active Abandoned
- 2001-01-18 EE EEP200200398A patent/EE200200398A/en unknown
- 2001-01-18 BR BR0107783-0A patent/BR0107783A/en not_active IP Right Cessation
- 2001-01-18 EA EA200200768A patent/EA200200768A1/en unknown
- 2001-01-22 SV SV2001000293A patent/SV2002000293A/en not_active Application Discontinuation
-
2002
- 2002-04-17 ZA ZA200203040A patent/ZA200203040B/en unknown
- 2002-04-25 CR CR6630A patent/CR6630A/en not_active Application Discontinuation
- 2002-05-03 NO NO20022117A patent/NO20022117L/en unknown
- 2002-07-16 IS IS6474A patent/IS6474A/en unknown
- 2002-08-16 HR HRP20020675 patent/HRP20020675A2/en not_active Application Discontinuation
- 2002-08-16 BG BG107011A patent/BG107011A/en unknown
- 2002-08-16 MA MA26785A patent/MA27589A1/en unknown
- 2002-11-08 US US10/291,158 patent/US6919461B2/en not_active Expired - Fee Related
-
2005
- 2005-04-22 US US11/112,423 patent/US7232912B2/en not_active Expired - Fee Related
-
2006
- 2006-01-10 US US11/329,303 patent/US20060111322A1/en not_active Abandoned
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1376600A (en) * | 1971-02-15 | 1974-12-04 | Agfa Gevaert | Photographic developer compositions |
| US3994890A (en) * | 1974-01-31 | 1976-11-30 | Chugai Seiyaku Kabushiki Kaisha | 1-Aminoalkyl, 3-phenyl indazoles |
| US4051145A (en) * | 1974-03-29 | 1977-09-27 | Produits Chimiques Ugine Kuhlmann | Fluorinated indazole derivatives and a process for their preparation |
| EP0023633A1 (en) * | 1979-07-16 | 1981-02-11 | Chugai Seiyaku Kabushiki Kaisha | Pyrazoloindazole derivatives and process for preparing the same and pharmaceutical compositions |
| US4978603A (en) * | 1987-12-14 | 1990-12-18 | Fuji Photo Film Co., Ltd. | Image forming process comprising developing fine grain silver halide emulsion with a hydroquinone developer |
| EP0494774A1 (en) * | 1991-01-11 | 1992-07-15 | MERCK SHARP & DOHME LTD. | Indazole-substituted fivemembered heteroaromatic compounds |
| WO1996020192A1 (en) * | 1994-12-23 | 1996-07-04 | Glaxo Group Limited | Piperidineacetic acid derivatives useful as fibrinogen antagonist agent |
| US5760028A (en) * | 1995-12-22 | 1998-06-02 | The Dupont Merck Pharmaceutical Company | Integrin receptor antagonists |
| WO1998043969A1 (en) * | 1997-03-31 | 1998-10-08 | Dupont Pharmaceuticals Company | Indazoles of cyclic ureas useful as hiv protease inhibitors |
| EP0904769A1 (en) * | 1997-08-25 | 1999-03-31 | L'oreal | Keratin fibre dye compositions containing Indazole amine derivatives and dyeing method |
| WO1999015500A1 (en) * | 1997-09-05 | 1999-04-01 | Glaxo Group Limited | Substituted oxindole derivatives as protein tyrosine kinase and as protein serine/threonine kinase inhibitors |
| WO1999017769A1 (en) * | 1997-10-06 | 1999-04-15 | Basf Aktiengesellschaft | INDENO[1,2-c]-, NAPHTHO[1,2-c]- AND BENZO[6,7]CYCLOHEPTA[1,2-c]PYRAZOLE DERIVATIVES |
Non-Patent Citations (7)
| Title |
|---|
| C WENTRUP ET AL: JOURNAL OF ORGANIC CHEMISTRY, vol. 43, no. 10, 1978, pages 2037-41, XP002172436 * |
| CHEMICAL ABSTRACTS, vol. 107, no. 21, 23 November 1987 (1987-11-23) Columbus, Ohio, US; abstract no. 198159c, page 749; XP002172438 & Y FUJIMURA ET AL: YAKUGAKU ZASSHI, vol. 106, no. 11, 1986, pages 1002-7, * |
| D R STOVER ET AL: CURRENT OPINION IN DRUG DISCOVERY & DEVELOPMENT, vol. 2, no. 4, 1999, pages 274-85, XP000926237 cited in the application * |
| G V DE LUCCA ET AL: JOURNAL OF MEDICINAL CHEMISTRY, vol. 42, no. 1, 1999, pages 135-52, XP002172435 * |
| K R WEBSTER: EXP. OPIN. INVEST. DRUGS, vol. 7, no. 6, 1998, pages 865-87, XP000926235 cited in the application * |
| PATENT ABSTRACTS OF JAPAN vol. 009, no. 112 (C-281), 16 May 1985 (1985-05-16) & JP 60 004184 A (CHUGAI SEIYAKU KK), 10 January 1985 (1985-01-10) * |
| T OWA ET AL: JOURNAL OF MEDICINAL CHEMISTRY, vol. 42, no. 19, 1999, pages 3789-99, XP002172437 cited in the application * |
Cited By (341)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7227020B2 (en) | 1999-12-24 | 2007-06-05 | Aventis Pharma Limited | Azaindoles |
| US6770643B2 (en) | 1999-12-24 | 2004-08-03 | Aventis Pharma Limited | Azaindoles |
| EP1313711A2 (en) * | 2000-07-31 | 2003-05-28 | Signal Pharmaceuticals, Inc. | Indazole derivatives as jnk inhibitors |
| WO2002016348A1 (en) * | 2000-08-09 | 2002-02-28 | Astrazeneca Ab | Antiangiogenic bicyclic derivatives |
| US7598268B2 (en) | 2000-09-11 | 2009-10-06 | Novartis Vaccines & Diagnostics, Inc. | Quinolinone derivatives |
| WO2002022586A1 (en) * | 2000-09-18 | 2002-03-21 | Bayer Aktiengesellschaft | Indazoles having an action similar to that of a thyroid hormone, method for the production thereof, and their use in medicaments |
| US6608049B2 (en) | 2000-09-18 | 2003-08-19 | Bayer Aktiengesellschaft | Indazoles |
| WO2002083648A1 (en) * | 2001-04-16 | 2002-10-24 | Eisai Co., Ltd. | Novel 1h-indazole compound |
| US7541376B2 (en) | 2001-04-16 | 2009-06-02 | Eisai R&D Management Co., Ltd. | 1H-indazole compounds |
| US7776890B2 (en) | 2001-04-16 | 2010-08-17 | Eisai R&D Management Co., Ltd. | 1H-indazole compounds |
| US6982274B2 (en) | 2001-04-16 | 2006-01-03 | Eisai Co., Ltd. | 1H-indazole compound |
| US6897207B2 (en) | 2001-06-21 | 2005-05-24 | Aventis Pharmaceuticals Inc. | Azaindoles |
| US7943616B2 (en) | 2001-06-21 | 2011-05-17 | Aventis Pharmaceuticals Inc. | Azaindoles for inhibiting aurora2 and other kinases |
| US7642278B2 (en) | 2001-07-03 | 2010-01-05 | Novartis Vaccines And Diagnostics, Inc. | Indazole benzimidazole compounds |
| US7064215B2 (en) | 2001-07-03 | 2006-06-20 | Chiron Corporation | Indazole benzimidazole compounds |
| WO2003004488A1 (en) * | 2001-07-03 | 2003-01-16 | Chiron Corporation | Indazole benzimidazole compounds as tyrosine and serine/threonine kinase inhibitors |
| JP2004536113A (en) * | 2001-07-03 | 2004-12-02 | カイロン コーポレイション | Indazole benzimidazole compounds as inhibitors of tyrosine kinase and serine / threonine kinase |
| US7119115B2 (en) | 2001-07-27 | 2006-10-10 | Aventis Pharma S.A. | Indazole or indole derivatives, and use thereof in human medicine and more particularly in oncology |
| WO2003011833A1 (en) * | 2001-07-27 | 2003-02-13 | Aventis Pharma S.A. | Indazole or indole derivatives, their use in human medicine and more particularly in cancerology |
| FR2827861A1 (en) * | 2001-07-27 | 2003-01-31 | Aventis Pharma Sa | New aryl-substituted indole, isoindole and indazole compounds, useful for treating cell proliferation disorders e.g. cancer, inhibit polymerization of tubulin |
| EA007430B1 (en) * | 2001-09-26 | 2006-10-27 | Фармация Италия С.П.А. | Aminoindazole derivatives active as kinase inhibitors, process for their preparation and pharmaceutical compositions containing them |
| WO2003028720A1 (en) * | 2001-09-26 | 2003-04-10 | Pharmacia Italia S.P.A. | Aminoindazole derivatives active as kinase inhibitors, process for their preparation and pharmaceutical compositions containing them |
| US7511136B2 (en) | 2001-09-26 | 2009-03-31 | Pfizer Italia S.R.L. | Aminoindazole derivatives active as kinase inhibitors |
| EP1820503A3 (en) * | 2001-09-26 | 2008-05-21 | Pfizer Italia S.r.l. | Aminoindazole derivatives active as kinase inhibitors, process for their preparation and pharmaceutical compositions containing them |
| US7271261B2 (en) | 2001-10-19 | 2007-09-18 | Ortho-Mcneil Pharmaceutical, Inc. | Substituted benzimidazoles and imidazo-[4,5]-pyridines |
| WO2003032984A1 (en) * | 2001-10-19 | 2003-04-24 | Ortho-Mcneil Pharmaceutical,Inc. | 2-phenyl benzimidazoles and imidazo-[4,5]-pyridines as cdsi/chk2-inhibitors and adjuvants to chemotherapy or radiation therapy in the treatment of cancer |
| JP2005506349A (en) * | 2001-10-19 | 2005-03-03 | オーソ−マクニール・フアーマシユーチカル・インコーポレーテツド | 2-phenylbenzimidazole and imidazo- [4,5] -pyridine as CDS1 / CHK2 inhibitors and adjuvants for chemotherapy or radiotherapy in cancer treatment |
| US7504414B2 (en) | 2001-10-19 | 2009-03-17 | Ortho-Mcneil Pharmaceutical, Inc. | Substituted benzimidazoles and imidazo-[4,5]-pyridines |
| US6897208B2 (en) | 2001-10-26 | 2005-05-24 | Aventis Pharmaceuticals Inc. | Benzimidazoles |
| FR2831536A1 (en) * | 2001-10-26 | 2003-05-02 | Aventis Pharma Sa | NOVEL BENZIMIDAZOLE DERIVATIVES, PROCESS FOR THEIR PREPARATION, THEIR USE AS MEDICAMENTS, PHARMACEUTICAL COMPOSITIONS AND NOVEL USE IN PARTICULAR AS KDR INHIBITORS |
| AU2002334217B2 (en) * | 2001-10-26 | 2008-07-03 | Aventis Pharmaceuticals Inc. | Benzimidazoles and analogues and their use as protein kinases inhibitors |
| WO2003035644A1 (en) * | 2001-10-26 | 2003-05-01 | Aventis Pharma S.A. | Benzimidazole derivatives and their use as kdr protein kinase inhibitors |
| US8288425B2 (en) | 2001-10-26 | 2012-10-16 | Aventis Pharmaceuticals Inc. | Benzimidazoles |
| WO2003035065A1 (en) * | 2001-10-26 | 2003-05-01 | Aventis Pharmaceuticals Inc | Benzimidazoles and analogues and their use as protein kinases inhibitors |
| FR2831537A1 (en) * | 2001-10-26 | 2003-05-02 | Aventis Pharma Sa | Composition useful for treating inflammatory disease, cancer, chronic obstructive pulmonary disease, asthma, allergic rhinitis, or atopic dermatitis comprises benzimidazoles derivatives |
| US7351729B2 (en) * | 2002-03-08 | 2008-04-01 | Signal Pharmaceuticals, Llc | JNK inhibitors for use in combination therapy for treating or managing proliferative disorders and cancers |
| US7473701B2 (en) | 2002-03-11 | 2009-01-06 | Aventis Pharma S.A. | Substituted indazoles, compositions containing them, method of production and use |
| AU2003222148C1 (en) * | 2002-03-29 | 2009-05-21 | Carlsbad Technology, Inc. | Angiogenesis inhibitors |
| EP1496894A1 (en) * | 2002-03-29 | 2005-01-19 | Carlsbad Technology Inc | Angiogenesis inhibitors |
| US7166293B2 (en) | 2002-03-29 | 2007-01-23 | Carlsbad Technology, Inc. | Angiogenesis inhibitors |
| EP1496894A4 (en) * | 2002-03-29 | 2007-06-06 | Carlsbad Technology Inc | Angiogenesis inhibitors |
| AU2003222148B2 (en) * | 2002-03-29 | 2008-10-09 | Carlsbad Technology, Inc. | Angiogenesis inhibitors |
| US7429609B2 (en) * | 2002-05-31 | 2008-09-30 | Eisai R & D Management Co., Ltd. | Pyrazole compound and medicinal composition containing the same |
| JP2004075673A (en) * | 2002-06-19 | 2004-03-11 | Mitsubishi Chemicals Corp | Compound and organic electroluminescent element using the same |
| WO2004014864A1 (en) * | 2002-08-10 | 2004-02-19 | Astex Technology Limited | 1h-indazole-3-carboxamide compounds as cyclin dependent kinases (cdk) inhibitors |
| WO2004014368A1 (en) * | 2002-08-12 | 2004-02-19 | Sugen, Inc. | 3-pyrrolyl-pyridopyrazoles and 3-pyrrolyl-indazoles as novel kinase inhibitors |
| US7186716B2 (en) | 2002-08-12 | 2007-03-06 | Sugen, Inc. | 3-Pyrrol-pyridopyrazoles and 3-pyrrolyl-indazoles as novel kinase inhibitors |
| WO2004050088A1 (en) * | 2002-12-03 | 2004-06-17 | Kyowa Hakko Kogyo Co., Ltd. | Jnk inhibitor |
| EA008501B1 (en) * | 2002-12-19 | 2007-06-29 | Пфайзер Инк. | 2-(1h-indazol-6-ylamino)-benzamide compounds as protein kinases inhibitors useful for the treatment of ophtalmic diseases |
| US7053107B2 (en) | 2002-12-19 | 2006-05-30 | Agouron Pharmaceuticals, Inc. | Indazole compounds and pharmaceutical compositions for inhibiting protein kinases, and methods for their use |
| KR100730267B1 (en) * | 2002-12-19 | 2007-06-20 | 화이자 인코포레이티드 | 2-1h-indazol-6-ylamino-benzamide compounds as protein kinases inhibitors useful for the treatment of ophtalmic diseases |
| WO2004056806A1 (en) * | 2002-12-19 | 2004-07-08 | Pfizer Inc. | 2-(1h-indazol-6-ylamino)-benzamide compounds as protein kinases inhibitors useful for the treatment of ophtalmic diseases |
| NL1025073C2 (en) * | 2002-12-19 | 2005-02-01 | Pfizer | Indazole compounds and pharmaceutical compositions for inhibiting protein kinases, and methods for their use. |
| WO2004076450A1 (en) * | 2003-02-27 | 2004-09-10 | J. Uriach Y Compañia S.A. | Pyrazolopyridine derivates |
| KR101312736B1 (en) * | 2003-02-27 | 2013-09-27 | 팔라우 파르마 에스에이 | Pyrazolopyridine derivates |
| CN100354275C (en) * | 2003-02-27 | 2007-12-12 | 帕劳制药股份有限公司 | Pyrazolopyridine derivates |
| US8536194B2 (en) | 2003-02-27 | 2013-09-17 | Palau Pharma, S.A. | Pyrazolopyridine derivates |
| US7468376B2 (en) | 2003-02-27 | 2008-12-23 | Palau Pharma, S.A. | Pyrazolopyridine derivates |
| US7132440B2 (en) | 2003-04-17 | 2006-11-07 | Janssen Pharmaceutica, N.V. | Substituted benzimidazoles and imidazo-[4,5]-pyridines |
| US7687639B2 (en) | 2003-04-17 | 2010-03-30 | Janssen Pharmaceutica N.V. | Substituted benzimidazoles and imidazo-[4,5]-pyridines |
| FR2854159A1 (en) * | 2003-04-25 | 2004-10-29 | Aventis Pharma Sa | New 2-pyrazolyl or indazolyl-indole derivatives, useful for treating e.g. angiogenic or fibrinolytic diseases, act by inhibiting protein kinases |
| WO2004096792A2 (en) * | 2003-04-25 | 2004-11-11 | Aventis Pharma S.A. | Novel indole derivatives, method of preparing same in the form of medicaments, pharmaceutical compositions and, in particular, kdr inhibitors |
| WO2004096792A3 (en) * | 2003-04-25 | 2005-09-15 | Aventis Pharma Sa | Novel indole derivatives, method of preparing same in the form of medicaments, pharmaceutical compositions and, in particular, kdr inhibitors |
| JP2006524668A (en) * | 2003-04-25 | 2006-11-02 | アヴェンティス ファーマ エス.エー. | Novel indole derivatives and pharmaceutical substances and pharmaceutical compositions, in particular their preparation as KDR inhibitors |
| EP2305250A1 (en) | 2003-07-03 | 2011-04-06 | Astex Therapeutics Limited | Benzimidazole derivatives and their use as protein kinases inhibitors |
| WO2005002552A2 (en) | 2003-07-03 | 2005-01-13 | Astex Therapeutics Limited | Benzimidazole derivatives and their use as protein kinases inhibitors |
| WO2005007635A3 (en) * | 2003-07-18 | 2005-08-11 | Sigma Tau Ind Farmaceuti | Combretastatin derivatives with cytotoxic action |
| WO2005007635A2 (en) * | 2003-07-18 | 2005-01-27 | Sigma-Tau Industrie Farmaceutiche Riunite S.P.A. | Combretastatin derivatives with cytotoxic action |
| EP2256106A1 (en) | 2003-07-22 | 2010-12-01 | Astex Therapeutics Limited | 3,4-disubstituted 1H-pyrazole compounds and their use as cyclin dependent kinases (CDK) and glycogen synthase kinase-3 (GSK-3) modulators |
| WO2005012256A1 (en) | 2003-07-22 | 2005-02-10 | Astex Therapeutics Limited | 3, 4-disubstituted 1h-pyrazole compounds and their use as cyclin dependent kinases (cdk) and glycogen synthase kinase-3 (gsk-3) modulators |
| AU2004260759B2 (en) * | 2003-07-30 | 2010-04-22 | Kyowa Hakko Kirin Co., Ltd. | Protein kinase inhibitors |
| WO2005009997A1 (en) * | 2003-07-30 | 2005-02-03 | Pfizer Inc. | 3,5 disubstituted indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| US7008953B2 (en) | 2003-07-30 | 2006-03-07 | Agouron Pharmaceuticals, Inc. | 3, 5 Disubstituted indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| WO2005014554A1 (en) * | 2003-08-08 | 2005-02-17 | Astex Therapeutics Limited | 1h-indazole-3-carboxamide compounds as mapkap kinase modulators |
| US7470807B2 (en) | 2003-08-14 | 2008-12-30 | Asahi Kasei Pharma Corporation | Substituted arylalkanoic acid derivatives and use thereof |
| EP1512400A1 (en) * | 2003-09-03 | 2005-03-09 | Carlsbad Technology Inc | Treatment of neurodegenerative diseases |
| EP1512401A1 (en) * | 2003-09-03 | 2005-03-09 | Yung Shin Pharmaceutical Ind. Co., Ltd. | Enhancement of bone growth and inhibition of bone resorption |
| US7160916B2 (en) | 2003-09-03 | 2007-01-09 | Yung Shin Pharmaceutical Ind. Co., Ltd. | Method of increasing bone density or treating osteoporosis |
| WO2005026175A1 (en) * | 2003-09-08 | 2005-03-24 | Aventis Pharmaceuticals Inc. | Thienopyrazoles |
| AU2004272507B2 (en) * | 2003-09-08 | 2011-05-12 | Aventis Pharmaceuticals Inc. | Thienopyrazoles |
| WO2005035506A1 (en) * | 2003-10-15 | 2005-04-21 | Ube Industries, Ltd. | Novel indazole derivative |
| EP2762475A1 (en) | 2003-11-07 | 2014-08-06 | Novartis Vaccines and Diagnostics, Inc. | Pharmaceutically acceptable salts of quinolinone compounds and their medical use |
| WO2005094823A1 (en) * | 2004-03-30 | 2005-10-13 | Kyowa Hakko Kogyo Co., Ltd. | Flt-3 INHIBITOR |
| WO2005103036A1 (en) * | 2004-04-23 | 2005-11-03 | Biofocus Discovery Ltd | Pyrimidin-4-yl-1h-indazol-5yl-amines as chk1 kinases inhibitors |
| EP1640002A1 (en) * | 2004-08-30 | 2006-03-29 | Yung Shin Pharmaceutical Ind. Co., Ltd. | Indazole derivatives for inhibition of angiogenesis |
| WO2006039718A3 (en) * | 2004-10-01 | 2006-07-13 | Amgen Inc | Aryl nitrogen-containing bicyclic compounds and their use as kinase inhibitors |
| WO2006054143A1 (en) * | 2004-11-17 | 2006-05-26 | Pfizer Inc. | Polymorphs of {5-[3-(4,6-difluoro-1h-benzoimidazol-2-yl)-1h-indazol-5-yl]-4-methyl-pyridin-3-ylmethyl}-ethyl-amine |
| EP2395000A1 (en) | 2004-12-30 | 2011-12-14 | Astex Therapeutics Limited | Benzimidazole compounds that modulate the activity of CDK, GSK and aurora kinases |
| WO2006077428A1 (en) | 2005-01-21 | 2006-07-27 | Astex Therapeutics Limited | Pharmaceutical compounds |
| WO2006077424A1 (en) | 2005-01-21 | 2006-07-27 | Astex Therapeutics Limited | Pharmaceutical compounds |
| US7605272B2 (en) | 2005-01-27 | 2009-10-20 | Kyowa Hakko Kirin Co., Ltd. | IGF-1R inhibitor |
| WO2006086705A1 (en) * | 2005-02-10 | 2006-08-17 | Aventis Pharmaceuticals Inc. | Substituted bis aryl and heteroaryl compounds as selective 5ht2a antagonists |
| US7625889B2 (en) | 2005-02-10 | 2009-12-01 | Aventis Pharmaceuticals Inc. | Substituted bis aryl and heteroaryl compounds as selective 5HT2A antagonists |
| EP2330105A1 (en) | 2005-03-29 | 2011-06-08 | ICOS Corporation | Heteroaryl urea derivatives useful for inhibiting CHK1 |
| WO2006105262A1 (en) | 2005-03-29 | 2006-10-05 | Icos Corporation | HETEROARYL UREA DERIVATIVES USEFUL FOR INHIBITING CHKl |
| EP2465857A1 (en) | 2005-05-17 | 2012-06-20 | Novartis AG | Methods for synthesizing heterocyclic compounds |
| WO2006125130A1 (en) | 2005-05-17 | 2006-11-23 | Novartis Ag | Methods for synthesizing heterocyclic compounds |
| US8222413B2 (en) | 2005-05-17 | 2012-07-17 | Novartis Ag | Methods for synthesizing heterocyclic compounds |
| US7541367B2 (en) | 2005-05-31 | 2009-06-02 | Janssen Pharmaceutica, N.V. | 3-benzoimidazolyl-pyrazolopyridines useful in treating kinase disorders |
| US8455647B2 (en) | 2005-07-11 | 2013-06-04 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| US8034943B2 (en) | 2005-07-11 | 2011-10-11 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| WO2007058626A1 (en) * | 2005-11-16 | 2007-05-24 | S*Bio Pte Ltd | Indazole compounds |
| EP2772257A1 (en) | 2005-11-29 | 2014-09-03 | Novartis AG | Formulations of quinolinones |
| WO2007107346A1 (en) * | 2006-03-23 | 2007-09-27 | F. Hoffmann-La Roche Ag | Substituted indazole derivatives, their manufacture and use as pharmaceutical agents |
| WO2008001886A1 (en) | 2006-06-30 | 2008-01-03 | Kyowa Hakko Kirin Co., Ltd. | Aurora inhibitor |
| WO2008001885A1 (en) | 2006-06-30 | 2008-01-03 | Kyowa Hakko Kirin Co., Ltd. | Abl KINASE INHIBITOR |
| US8658649B2 (en) | 2006-09-11 | 2014-02-25 | Sanofi | Kinase inhibitor |
| US8809326B2 (en) | 2006-09-20 | 2014-08-19 | Aerie Pharmaceuticals, Inc. | Isoquinolinone Rho kinase inhibitors |
| US10624882B2 (en) | 2006-09-20 | 2020-04-21 | Aerie Pharmaceuticals, Inc. | Rho kinase inhibitors |
| WO2008044041A1 (en) | 2006-10-12 | 2008-04-17 | Astex Therapeutics Limited | Pharmaceutical combinations |
| WO2008044045A1 (en) | 2006-10-12 | 2008-04-17 | Astex Therapeutics Limited | Pharmaceutical combinations |
| US9365518B2 (en) | 2007-01-10 | 2016-06-14 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| US8455513B2 (en) | 2007-01-10 | 2013-06-04 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| US8357699B2 (en) | 2007-01-10 | 2013-01-22 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| US10899714B2 (en) | 2007-01-10 | 2021-01-26 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| US10472327B2 (en) | 2007-01-10 | 2019-11-12 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| US8921392B2 (en) | 2007-01-10 | 2014-12-30 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| US9890123B2 (en) | 2007-01-10 | 2018-02-13 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| WO2008111441A1 (en) | 2007-03-05 | 2008-09-18 | Kyowa Hakko Kirin Co., Ltd. | Pharmaceutical composition |
| RU2487873C2 (en) * | 2007-06-08 | 2013-07-20 | Эбботт Лэборетриз | 5-substituted indazole as kinase inhibitors |
| US9163007B2 (en) | 2007-06-08 | 2015-10-20 | Abbvie Inc. | 5-substituted indazoles as kinase inhibitors |
| WO2008154241A1 (en) * | 2007-06-08 | 2008-12-18 | Abbott Laboratories | 5-heteroaryl substituted indazoles as kinase inhibitors |
| US8648069B2 (en) | 2007-06-08 | 2014-02-11 | Abbvie Inc. | 5-substituted indazoles as kinase inhibitors |
| JP2014074057A (en) * | 2007-06-08 | 2014-04-24 | Abbvie Inc | 5-heteroaryl substituted indazoles as kinase inhibitors |
| US8455514B2 (en) | 2008-01-17 | 2013-06-04 | Aerie Pharmaceuticals, Inc. | 6-and 7-amino isoquinoline compounds and methods for making and using the same |
| US8871757B2 (en) | 2008-01-17 | 2014-10-28 | Aerie Pharmaceuticals, Inc. | 6-and 7-amino isoquinoline compounds and methods for making and using the same |
| US8334314B2 (en) | 2008-04-28 | 2012-12-18 | Asahi Kasei Pharma Corporation | Phenylpropionic acid derivative and use thereof |
| US8772327B2 (en) | 2008-04-28 | 2014-07-08 | Asahi Kasei Pharma Corporation | Phenylpropionic acid derivative and use thereof |
| US8450344B2 (en) | 2008-07-25 | 2013-05-28 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US9096569B2 (en) | 2008-07-25 | 2015-08-04 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US9884840B2 (en) | 2008-07-25 | 2018-02-06 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US10882840B2 (en) | 2008-07-25 | 2021-01-05 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US9512101B2 (en) | 2008-07-25 | 2016-12-06 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US10532993B2 (en) | 2008-07-25 | 2020-01-14 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US11021456B2 (en) | 2008-07-25 | 2021-06-01 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US10112920B2 (en) | 2008-07-25 | 2018-10-30 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US8759388B2 (en) | 2008-07-25 | 2014-06-24 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| USRE47731E1 (en) | 2009-04-06 | 2019-11-19 | University Health Network | Kinase inhibitors and method of treating cancer with same |
| US8999968B2 (en) | 2009-04-06 | 2015-04-07 | University Health Network | Kinase inhibitors and method of treating cancer with same |
| US11618748B2 (en) | 2009-05-01 | 2023-04-04 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| WO2010127329A1 (en) * | 2009-05-01 | 2010-11-04 | Aerie Pharmaceuticals, Inc. | Dual-action inhibitors and methods of using same |
| US8716310B2 (en) | 2009-05-01 | 2014-05-06 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| US8394826B2 (en) | 2009-05-01 | 2013-03-12 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| US10174017B2 (en) | 2009-05-01 | 2019-01-08 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| WO2010127330A1 (en) * | 2009-05-01 | 2010-11-04 | Aerie Phamaceuticals, Inc. | Dual-action inhibitors and methods of using same |
| US10316029B2 (en) | 2009-05-01 | 2019-06-11 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| US10654844B2 (en) | 2009-05-01 | 2020-05-19 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| US11028081B2 (en) | 2009-05-01 | 2021-06-08 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| US9951059B2 (en) | 2009-05-01 | 2018-04-24 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| US8703794B2 (en) | 2009-08-10 | 2014-04-22 | Samumed, Llc | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| EP2987487A1 (en) | 2009-08-10 | 2016-02-24 | Samumed, LLC | Indazole inhibitors of the wnt signal pathway and therapeutic uses thereof |
| US8604052B2 (en) | 2009-08-10 | 2013-12-10 | Samumed, Llc | Indazole inhibitors of the WNT signal pathway and therapeutic uses thereof |
| US9763927B2 (en) | 2009-08-10 | 2017-09-19 | Samumed, Llc | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| US9090613B2 (en) | 2009-08-10 | 2015-07-28 | Samumed, Llc | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| US10016406B2 (en) | 2009-08-10 | 2018-07-10 | Samumed, Llc | Indazole inhibitors of the WNT signal pathway and therapeutic uses thereof |
| US8252812B2 (en) | 2009-08-10 | 2012-08-28 | Samumed, Llc | Indazole inhibitors of the WNT signal pathway and therapeutic uses thereof |
| WO2011057054A1 (en) | 2009-11-06 | 2011-05-12 | Vitae Pharmaceuticals, Inc. | Aryl- and heteroarylcarbonyl derivatives of hexahydroindenopyridine and octahydrobenzoquinoline |
| US9855272B2 (en) | 2009-12-21 | 2018-01-02 | Samumed, Llc | 1H-pyrazolo[3,4-B]pyridines and therapeutic uses thereof |
| US10105370B2 (en) | 2009-12-21 | 2018-10-23 | Samumed, Llc | 1H-pyrazolo[3,4-B]pyridines and therapeutic uses thereof |
| US9067939B2 (en) | 2009-12-21 | 2015-06-30 | Samumed, Llc | 1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| EP3305073A1 (en) | 2009-12-21 | 2018-04-11 | Samumed, LLC | 1h-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| EP3001903A1 (en) | 2009-12-21 | 2016-04-06 | Samumed, LLC | 1h-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| US8450340B2 (en) | 2009-12-21 | 2013-05-28 | Samumed, Llc | 1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| US9579327B2 (en) | 2010-04-06 | 2017-02-28 | University Health Network | Synthesis of chiral 2-(1H-indazol-6-yl)-spiro[cyclopropane-1,3′-indolin]-2′-ones |
| US10077255B2 (en) | 2010-04-06 | 2018-09-18 | University Health Network | Synthesis of chiral 2-(1H-indazol-6-yl)-spiro[cyclopropane-1,3′-indolin]-2′-ones |
| US9139563B2 (en) | 2010-04-06 | 2015-09-22 | University Health Network | Kinase inhibitors and method of treating cancer |
| US10358436B2 (en) | 2010-04-06 | 2019-07-23 | University Health Network | Kinase inhibitors and method of treating cancer |
| US9907800B2 (en) | 2010-04-06 | 2018-03-06 | University Health Network | Kinase inhibitors and method of treating cancer |
| US9796703B2 (en) | 2010-04-06 | 2017-10-24 | University Health Network | Synthesis of chiral 2-(1H-indazol-6-yl)-spiro[cyclopropane-1,3′-indolin]-2′-ones |
| US9303010B2 (en) | 2010-08-18 | 2016-04-05 | Samumed, Llc | β- and γ-diketones and γ-hydroxyketones as Wnt/β-catenin signaling pathway activators |
| US8609717B2 (en) | 2010-08-18 | 2013-12-17 | Samumed, Llc | β- and γ-diketones and γ-hydroxyketones as WNT/β-catenin signaling pathway activators |
| EP3915367A1 (en) | 2010-08-18 | 2021-12-01 | BioSplice Therapeutics, Inc. | Diketones and hydroxyketones as catenin signaling pathway activators |
| EP3311666A1 (en) | 2010-08-18 | 2018-04-25 | Samumed, LLC | Diketones and hydroxyketones as catenin signaling pathway activators |
| US8921413B2 (en) | 2010-08-18 | 2014-12-30 | Samumed, Llc | β- and γ-diketones and γ-hydroxyketones as WNT/ β-catenin signaling pathway activators |
| US10314832B2 (en) | 2010-08-18 | 2019-06-11 | Samumed, Llc | β- and γ-diketones and γ-hydroxyketones as Wnt/β-catenin signaling pathway activators |
| US9884053B2 (en) | 2010-08-18 | 2018-02-06 | Samumed, Llc | β- and γ-diketones and γ-hydroxyketones as WNT/β-catenin signaling pathway activators |
| US8629176B1 (en) | 2010-08-18 | 2014-01-14 | Samumed, Llc | β- and γ-diketones and γ-hydroxyketones as WNT/ β-catenin signaling pathway activators |
| WO2012061708A1 (en) | 2010-11-05 | 2012-05-10 | Boehringer Ingelheim International Gmbh | Crystalline forms of hydrochloride salt of (4a-r,9a-s) -1- (1h - benzoimidazole- 5 -carbonyl) -2, 3, 4, 4a, 9, 9a - hexahydro -1h- indeno [2, 1 -b] pyridine- 6 -carbonitrile and their use as hsd 1 inhibitors |
| US9090593B2 (en) | 2010-12-09 | 2015-07-28 | Amgen Inc. | Bicyclic compounds as Pim inhibitors |
| WO2012078777A1 (en) * | 2010-12-09 | 2012-06-14 | Amgen Inc. | Bicyclic compounds as pim inhibitors |
| US9321756B2 (en) | 2011-03-22 | 2016-04-26 | Amgen Inc. | Azole compounds as PIM inhibitors |
| US8569511B2 (en) * | 2011-04-01 | 2013-10-29 | University Of Utah Research Foundation | Substituted 3-(1H-benzo[d]imidazol-2-yl)-1H-indazole analogs as inhibitors of the PDK1 kinase |
| US20120277229A1 (en) * | 2011-04-01 | 2012-11-01 | Bearss David J | Substituted 3-(1h-benzo[d]imidazol-2-yl)-1h-indazole analogs as inhibitors of the pdk1 kinase |
| US9802916B2 (en) | 2011-09-14 | 2017-10-31 | Samumed, Llc | Indazole-3-carboxamides and their use as Wnt/beta-catenin signaling pathway inhibitors |
| WO2013040215A1 (en) | 2011-09-14 | 2013-03-21 | Samumed, Llc | Indazole-3-carboxamides and their use as wnt/b-catenin signaling pathway inhibitors |
| US11780823B2 (en) | 2011-09-14 | 2023-10-10 | Biosplice Therapeutics, Inc. | Indazole-3-carboxamides and their use as Wnt/β-catenin signaling pathway inhibitors |
| US10464924B2 (en) | 2011-09-14 | 2019-11-05 | Samumed, Llc | Indazole-3-carboxamides and their use as Wnt/β-catenin signaling pathway inhibitors |
| EP3473099A1 (en) | 2011-09-14 | 2019-04-24 | Samumed, LLC | Indazole-3-carboxamides and their use as wnt/b-catenin signaling pathway inhibitors |
| US9221793B2 (en) | 2011-09-14 | 2015-12-29 | Samumed, Llc | Indazole-3-carboxamides and their use as Wnt/β-catenin signaling pathway inhibitors |
| US11066388B2 (en) | 2011-09-14 | 2021-07-20 | Biosplice Therapeutics, Inc. | Indazole-3-carboxamides and their use as WNT/B-catenin signaling pathway inhibitors |
| US9469606B2 (en) | 2011-10-25 | 2016-10-18 | The General Hospital Corporation | Wnt/b-catenin inhibitors and methods of use |
| EP3284744A1 (en) | 2012-04-04 | 2018-02-21 | Samumed, LLC | Indazole inhibitors of the wnt signal pathway and therapeutic uses thereof |
| US8664241B2 (en) | 2012-04-04 | 2014-03-04 | Samumed, Llc | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| EP3915988A1 (en) | 2012-04-04 | 2021-12-01 | BioSplice Therapeutics, Inc. | Indazole inhibitors of the wnt signal pathway and therapeutic uses thereof |
| US10407425B2 (en) | 2012-04-04 | 2019-09-10 | Samumed, Llc | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| WO2013151708A1 (en) | 2012-04-04 | 2013-10-10 | Samumed, Llc | Indazole inhibitors of the wnt signal pathway and therapeutic uses thereof |
| US8673936B2 (en) | 2012-04-04 | 2014-03-18 | Samumed, Llc | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| US10947228B2 (en) | 2012-04-04 | 2021-03-16 | Samumed, Llc | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| US11697649B2 (en) | 2012-04-04 | 2023-07-11 | Biosplice Therapeutics, Inc. | Indazole inhibitors of the Wnt signal pathway and therapeutic uses thereof |
| US9994563B2 (en) | 2012-04-04 | 2018-06-12 | Samumed, Llc | Indazole inhibitors of the wnt signal pathway and therapeutic uses thereof |
| US9012472B2 (en) | 2012-05-04 | 2015-04-21 | Samumed, Llc | 1H-pyrazolo[3,4-B]pyridines and therapeutic uses thereof |
| US10071086B2 (en) | 2012-05-04 | 2018-09-11 | Samumed, Llc | 1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| US8883822B2 (en) | 2012-05-04 | 2014-11-11 | Samumed, Llc | 1H-pyrazolo[3,4-B]pyridines and therapeutic uses thereof |
| WO2013166396A2 (en) | 2012-05-04 | 2013-11-07 | Samumed, Llc | 1h-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| EP3593800A1 (en) | 2012-05-04 | 2020-01-15 | Samumed, LLC | 1h-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| US10342788B2 (en) | 2012-05-04 | 2019-07-09 | Samumed, Llc | 1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| US8618128B1 (en) | 2012-05-04 | 2013-12-31 | Samumed, Llc | 1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof |
| WO2014041518A1 (en) * | 2012-09-14 | 2014-03-20 | Glenmark Pharmaceuticals S.A. | Thienopyrrole derivatives as itk inhibitors |
| US9908867B2 (en) | 2013-01-08 | 2018-03-06 | Samumed, Llc | 3-(benzoimidazol-2-yl)-indazole inhibitors of the Wnt signaling pathway and therapeutic uses thereof |
| US10654832B2 (en) | 2013-01-08 | 2020-05-19 | Samumed, Llc | 3-(benzoimidazol-2-YL)-indazole inhibitors of the Wnt signaling pathway and therapeutic uses thereof |
| US10183929B2 (en) | 2013-01-08 | 2019-01-22 | Samumed, Llc | 3-(benzoimidazol-2-yl)-indazole inhibitors of the Wnt signaling pathway and therapeutic uses thereof |
| US11034682B2 (en) | 2013-02-22 | 2021-06-15 | Samumed, Llc | Gamma-diketones as wnt/β-catenin signaling pathway activators |
| US10457672B2 (en) | 2013-02-22 | 2019-10-29 | Samumed, Llc | γ-diketones as Wnt/β-catenin signaling pathway activators |
| EP3456325A2 (en) | 2013-02-22 | 2019-03-20 | Samumed, LLC | Gamma-diketones as wnt/beta-catenin signalling pathway activators |
| US11673885B2 (en) | 2013-02-22 | 2023-06-13 | Biosplice Therapeutics, Inc. | γ-diketones as Wnt/β-catenin signaling pathway activators |
| WO2014130869A1 (en) | 2013-02-22 | 2014-08-28 | Samumed, Llc | Gamma-diketones as wnt/beta -catenin signaling pathway activators |
| US9951053B2 (en) | 2013-02-22 | 2018-04-24 | Samumed, Llc | γ-diketones as Wnt/β-catenin signaling pathway activators |
| US9493440B2 (en) | 2013-03-04 | 2016-11-15 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US9440952B2 (en) | 2013-03-04 | 2016-09-13 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US9416126B2 (en) | 2013-03-04 | 2016-08-16 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US9688654B2 (en) | 2013-03-04 | 2017-06-27 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| WO2014134772A1 (en) * | 2013-03-04 | 2014-09-12 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| US10588901B2 (en) | 2013-03-15 | 2020-03-17 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US10568878B2 (en) | 2013-03-15 | 2020-02-25 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US11197853B2 (en) | 2013-03-15 | 2021-12-14 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US11020385B2 (en) | 2013-03-15 | 2021-06-01 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US9849122B2 (en) | 2013-03-15 | 2017-12-26 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US9931336B2 (en) | 2013-03-15 | 2018-04-03 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US9993470B2 (en) | 2013-03-15 | 2018-06-12 | Aerie Pharmaceuticals, Inc. | Combination therapy |
| US11185538B2 (en) | 2013-03-15 | 2021-11-30 | Aerie Pharmaceuticals, Inc. | Compositions for treating glaucoma or reducing intraocular pressure |
| US20160250220A1 (en) | 2013-10-18 | 2016-09-01 | University Health Network | Treatment for pancreatic cancer |
| US9642856B2 (en) | 2013-10-18 | 2017-05-09 | University Health Network | Treatment for pancreatic cancer |
| US10239864B2 (en) | 2014-07-24 | 2019-03-26 | Beta Pharma, Inc. | 2-H-indazole derivatives as cyclin-dependent kinase (CDK) inhibitors and therapeutic uses thereof |
| US11077046B2 (en) | 2014-08-20 | 2021-08-03 | Biosplice Therapeutics, Inc. | Gamma-diketones for treatment and prevention of aging skin and wrinkles |
| US11839679B2 (en) | 2014-08-20 | 2023-12-12 | Biosplice Therapeutics, Inc. | Gamma-diketones for treatment and prevention of aging skin and wrinkles |
| US9795550B2 (en) | 2014-08-20 | 2017-10-24 | Samumed, Llc | Gamma-diketones for treatment and prevention of aging skin and wrinkles |
| US10434052B2 (en) | 2014-08-20 | 2019-10-08 | Samumed, Llc | Gamma-diketones for treatment and prevention of aging skin and wrinkles |
| US9738638B2 (en) | 2014-09-08 | 2017-08-22 | Samumed, Llc | 2-(1H-indazol-3-yl)-3H-imidazo[4,5-B]pyridine and therapeutic uses thereof |
| US10280166B2 (en) | 2014-09-08 | 2019-05-07 | Samumed, Llc | 2-(1H-indazol-3-yl)-3H-imidazo[4,5-B]pyridine and therapeutic uses thereof |
| US9889140B2 (en) | 2014-09-08 | 2018-02-13 | Samumed, Llc | 3-(1H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridine and therapeutic uses thereof |
| US10533020B2 (en) | 2014-09-08 | 2020-01-14 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1 H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US10596154B2 (en) | 2014-09-08 | 2020-03-24 | Samumed, Llc | 3-(1H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US9758531B2 (en) | 2014-09-08 | 2017-09-12 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridine and therapeutic uses thereof |
| US10131677B2 (en) | 2014-09-08 | 2018-11-20 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridine and therapeutic uses thereof |
| US10526347B2 (en) | 2014-09-08 | 2020-01-07 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridine and therapeutic uses thereof |
| US9763951B2 (en) | 2014-09-08 | 2017-09-19 | Samumed, Llc | 3-(3H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US10081631B2 (en) | 2014-09-08 | 2018-09-25 | Samumed, Llc | 2-(1H-indazol-3-yl)-1H-imidazo[4,5-C]pyridine and therapeutic uses thereof |
| US10023572B2 (en) | 2014-09-08 | 2018-07-17 | Samumed, Llc | 2-(1h-indazol-3-yl)-3h-imidazo[4,5-b]pyridine and therapeutic uses thereof |
| US9657016B2 (en) | 2014-09-08 | 2017-05-23 | Samumed, Llc | 3-(1h-benzo[d]imidazol-2-yl)-1h-pyrazolo[3,4-c]pyridine and therapeutic uses thereof |
| US9844536B2 (en) | 2014-09-08 | 2017-12-19 | Samumed, Llc | 3-(1H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US10052331B2 (en) | 2014-09-08 | 2018-08-21 | Samumed, Llc | 3-(3H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US10206929B2 (en) | 2014-09-08 | 2019-02-19 | Samumed, Llc | 3-(1H-imidazo[4,5-c]pyridin-2-yl)-1H-pyrazolo[3,4-b]pyridine and therapeutic uses thereof |
| US10202377B2 (en) | 2014-09-08 | 2019-02-12 | Samumed, Llc | 3-(1H-benzo[D]imidazol-2-yl)-1H-pyrazolo[3,4-C]pyridine and therapeutic uses thereof |
| US10428065B2 (en) | 2015-04-20 | 2019-10-01 | Cellcentric Ltd | Isoxazolyl substituted imidazopyridines |
| US10118920B2 (en) | 2015-04-20 | 2018-11-06 | Cellcentric Ltd | Isoxazolyl substituted benzimidazoles |
| US10329309B2 (en) | 2015-08-03 | 2019-06-25 | Samumed, Llc | 3-(3H-imidazo[4,5-B]pyridin-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10285983B2 (en) | 2015-08-03 | 2019-05-14 | Samumed, Llc | 3-(1H-pyrrolo[2,3-B]pyridin-2-yl)-1H-pyrazolo[3,4-B] pyridines and therapeutic uses thereof |
| US10392383B2 (en) | 2015-08-03 | 2019-08-27 | Samumed, Llc | 3-(1H-benzo[d]imidazol-2-yl)-1H-pyrazolo[4,3-b]pyridines and therapeutic uses thereof |
| US10383861B2 (en) | 2015-08-03 | 2019-08-20 | Sammumed, LLC | 3-(1H-pyrrolo[2,3-C]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10350199B2 (en) | 2015-08-03 | 2019-07-16 | Samumed, Llc | 3-(1h-pyrrolo[2,3-b]pyridin-2-yl)-1h-indazoles and therapeutic uses thereof |
| US10231956B2 (en) | 2015-08-03 | 2019-03-19 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-YL)-1 H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10166218B2 (en) | 2015-08-03 | 2019-01-01 | Samumed, Llc | 3-(1H-indol-2-yl)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10226453B2 (en) | 2015-08-03 | 2019-03-12 | Samumed, Llc | 3-(1H-indol-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10519169B2 (en) | 2015-08-03 | 2019-12-31 | Samumed, Llc | 3-(1H-pyrrolo[2,3-C]pyridin-2-yl)-1 H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10604512B2 (en) | 2015-08-03 | 2020-03-31 | Samumed, Llc | 3-(1H-indol-2-yl)-1H-indazoles and therapeutic uses thereof |
| US10463651B2 (en) | 2015-08-03 | 2019-11-05 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-YL)-1H-indazoles and therapeutic uses thereof |
| US10285982B2 (en) | 2015-08-03 | 2019-05-14 | Samumed, Llc | 3-(1H-pyrrolo[2,3-B]pyridin-2-yl)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10188634B2 (en) | 2015-08-03 | 2019-01-29 | Samumed, Llc | 3-(3H-imidazo[4,5-C]pyridin-2-yl)-1 H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10195185B2 (en) | 2015-08-03 | 2019-02-05 | Samumed, Llc | 3-(1H-imidazo[4,5-C]pyridin-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| US10206908B2 (en) | 2015-08-03 | 2019-02-19 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-YL)-1H-pyrazolo[3,4-C]pyridines and therapeutic uses thereof |
| US10226448B2 (en) | 2015-08-03 | 2019-03-12 | Samumed, Llc | 3-(1H-pyrrolo[3,2-C]pyridin-2-yl)-1H-pyrazolo[3,4-B]pyridines and therapeutic uses thereof |
| US10206909B2 (en) | 2015-08-03 | 2019-02-19 | Samumed, Llc | 3-(1H-pyrrolo[2,3-B]pyridin-2-yl)-1H-pyrazolo[4,3-B]pyridines and therapeutic uses thereof |
| CN107922349B (en) * | 2015-08-07 | 2021-05-07 | 哈尔滨珍宝制药有限公司 | Vinyl compounds as FGFR and VEGFR inhibitors |
| US10519133B2 (en) | 2015-08-07 | 2019-12-31 | Medshine Discovery Inc. | Vinyl compounds as FGFR and VEGFR inhibitors |
| EP3333157A4 (en) * | 2015-08-07 | 2018-10-17 | Harbin Zhenbao Pharmaceutical Co., Ltd. | Vinyl compounds as fgfr and vegfr inhibitors |
| CN107922349A (en) * | 2015-08-07 | 2018-04-17 | 哈尔滨珍宝制药有限公司 | Vinyl compound as FGFR and VEGFR inhibitor |
| US10544139B2 (en) | 2015-11-06 | 2020-01-28 | Samumed, Llc | Treatment of osteoarthritis |
| US10882860B2 (en) | 2015-11-06 | 2021-01-05 | Samumed, Llc | Treatment of osteoarthritis |
| US10899757B2 (en) | 2015-11-06 | 2021-01-26 | Samumed, Llc | 2-(1H-indazol-3-yl)-3H-imidazo[4,5-C]pyridines and their anti-inflammatory uses thereof |
| US11560378B2 (en) | 2015-11-06 | 2023-01-24 | Biosplice Therapeutics, Inc. | Treatment of osteoarthritis |
| US11667632B2 (en) | 2015-11-06 | 2023-06-06 | Biosplice Therapeutics, Inc. | 2-(1H-indazol-3-yl)-3H-imidazo[4,5-C]pyridines and their anti-inflammatory uses thereof |
| US10550087B2 (en) | 2015-11-17 | 2020-02-04 | Aerie Pharmaceuticals, Inc. | Process for the preparation of kinase inhibitors and intermediates thereof |
| US9643927B1 (en) | 2015-11-17 | 2017-05-09 | Aerie Pharmaceuticals, Inc. | Process for the preparation of kinase inhibitors and intermediates thereof |
| EP3892276A1 (en) | 2016-04-27 | 2021-10-13 | BioSplice Therapeutics, Inc. | Isoquinolin-3-yl carboxamides as wnt inhibitors |
| US10556885B2 (en) | 2016-04-27 | 2020-02-11 | Samumed, Llc | Isoquinolin-3-YL carboxamides and preparation and use thereof |
| US11174244B2 (en) | 2016-04-27 | 2021-11-16 | Biosplice Therapeutics, Inc. | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| WO2017189823A2 (en) | 2016-04-27 | 2017-11-02 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10100038B2 (en) | 2016-04-27 | 2018-10-16 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10508099B2 (en) | 2016-04-27 | 2019-12-17 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10106527B2 (en) | 2016-04-27 | 2018-10-23 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| EP3943086A1 (en) | 2016-04-27 | 2022-01-26 | BioSplice Therapeutics, Inc. | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10287267B2 (en) | 2016-04-27 | 2019-05-14 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US11548872B2 (en) | 2016-04-27 | 2023-01-10 | Biosplice Therapeutics, Inc. | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10947217B2 (en) | 2016-04-27 | 2021-03-16 | Samumed, Llc | Isoquinolin-3-YL carboxamides and preparation and use thereof |
| US9951048B1 (en) | 2016-04-27 | 2018-04-24 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| WO2017189829A1 (en) | 2016-04-27 | 2017-11-02 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US11673881B2 (en) | 2016-04-27 | 2023-06-13 | Biosplice Therapeutics, Inc. | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10544128B2 (en) | 2016-04-27 | 2020-01-28 | Samumed, Llc | Isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10072004B2 (en) | 2016-06-01 | 2018-09-11 | Samumed, Llc | Process for preparing N-(5-(3-(7-(3-fluorophenyl)-3H-imidazo [4,5-C]pyridin-2-yl)-1H-indazol-5-yl)pyridin-3-yl)-3-methylbutanamide |
| US12012401B2 (en) | 2016-06-01 | 2024-06-18 | Biosplice Therapeutics, Inc. | Process for preparing N-(5-(3-(7-(3-fluorophenyl)-3H-imidazo[4,5-c]pyridin-2-yl)-1H-indazol-5-yl)pyridin-3-yl)-3-methylbutanamide |
| US10633380B2 (en) | 2016-06-01 | 2020-04-28 | Samumed, Llc | Process for preparing N-(5-(3-(7-(3-fluorophenyl)-3H-imidazo[4,5-C]pyridin-2-yl)-1H-indazol-5-yl)pyridin-3-yl)-3-methylbutanamide |
| EP3483142A4 (en) * | 2016-07-05 | 2019-05-15 | Guangzhou Maxinovel Pharmaceuticals Co., Ltd. | Aromatic acetylene or aromatic ethylene compound, intermediate, preparation method, pharmaceutical composition and use thereof |
| US11078192B2 (en) | 2016-07-05 | 2021-08-03 | Guangzhou Maxinovel Pharmaceuticals Co., Ltd. | Aromatic acetylene or aromatic ethylene compound, intermediate, preparation method, pharmaceutical composition and use thereof |
| AU2017294231B2 (en) * | 2016-07-05 | 2021-09-09 | Guangzhou Maxinovel Pharmaceuticals Co., Ltd | Aromatic acetylene or aromatic ethylene compound, intermediate, preparation method, pharmaceutical composition and use thereof |
| US11389441B2 (en) | 2016-08-31 | 2022-07-19 | Aerie Pharmaceuticals, Inc. | Ophthalmic compositions |
| US11707460B2 (en) | 2016-08-31 | 2023-07-25 | Aerie Pharmaceuticals, Inc. | Ophthalmic compositions |
| WO2018075858A1 (en) | 2016-10-21 | 2018-04-26 | Samumed, Llc | Methods of using indazole-3-carboxamides and their use as wnt/b-catenin signaling pathway inhibitors |
| US10806726B2 (en) | 2016-10-21 | 2020-10-20 | Samumed, Llc | Methods of using indazole-3-carb oxamides and their use as Wnt/B-catenin signaling pathway inhibitors |
| US11684615B2 (en) | 2016-10-21 | 2023-06-27 | Biosplice Therapeutics, Inc. | Methods of using indazole-3-carboxamides and their use as Wnt/β-catenin signaling pathway inhibitors |
| US11446288B2 (en) | 2016-11-07 | 2022-09-20 | Biosplice Therapeutics, Inc. | Single-dose, ready-to-use injectable formulations |
| US11819499B2 (en) | 2016-11-07 | 2023-11-21 | Biosplice Therapeutics, Inc. | Single-dose, ready-to-use injectable formulations |
| US10758523B2 (en) | 2016-11-07 | 2020-09-01 | Samumed, Llc | Single-dose, ready-to-use injectable formulations |
| US11312700B2 (en) | 2017-03-31 | 2022-04-26 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| US10858339B2 (en) | 2017-03-31 | 2020-12-08 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| US12018012B2 (en) | 2017-03-31 | 2024-06-25 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| US10604514B2 (en) | 2017-10-19 | 2020-03-31 | Samumed, Llc | 6-(5-membered heteroaryl)isoquinolin-3-yl carboxamides and preparation and use thereof |
| WO2019079626A1 (en) | 2017-10-19 | 2019-04-25 | Samumed, Llc | 6-(5-membered heteroaryl)isoquinolin-3-yl carboxamides and preparation and use thereof |
| WO2019084496A1 (en) | 2017-10-27 | 2019-05-02 | Samumed, Llc | 6-(5-membered heteroaryl)isoquinolin-3-yl-(5-membered heteroaryl) carboxamides and preparation and use thereof |
| US10413537B2 (en) | 2017-10-27 | 2019-09-17 | Samumed, Llc | 6-(5-membered heteroaryl)isoquinolin-3-yl-(5-membered heteroaryl) carboxamides and preparation and use thereof |
| WO2019084497A1 (en) | 2017-10-27 | 2019-05-02 | Samumed, Llc | 6-(6-membered heteroaryl & aryl)isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10653688B2 (en) | 2017-10-27 | 2020-05-19 | Samumed, Llc | 6-(6-membered heteroaryl and aryl)isoquinolin-3-yl carboxamides and preparation and use thereof |
| US10744128B2 (en) | 2017-10-27 | 2020-08-18 | Samumed, Llc | 6-(5-membered heteroaryl)isoquinolin-3-yl-(5-membered heteroaryl) carboxamides and preparation and use thereof |
| US10703748B2 (en) | 2017-10-31 | 2020-07-07 | Samumed, Llc | Diazanaphthalen-3-yl carboxamides and preparation and use thereof |
| WO2019089835A1 (en) | 2017-10-31 | 2019-05-09 | Samumed, Llc | Diazanaphthalen-3-yl carboxamides and preparation and use thereof |
| US11384048B2 (en) | 2017-12-29 | 2022-07-12 | Guangzhou Maxinovel Pharmaceuticals Co., Ltd. | Aromatic vinyl or aromatic ethyl derivative, preparation method therefor, intermediate, pharmaceutical composition, and application |
| WO2019165192A1 (en) | 2018-02-23 | 2019-08-29 | Samumed, Llc | 5-heteroaryl substituted indazole-3-carboxamides and preparation and use thereof |
| US10934297B2 (en) | 2018-02-23 | 2021-03-02 | Samumed, Llc | 5-heteroaryl substituted indazole-3-carboxamides and preparation and use thereof |
| US11713320B2 (en) | 2018-02-23 | 2023-08-01 | Biosplice Therapeutics, Inc. | 5-heteroaryl substituted indazole-3-carboxamides and preparation and use thereof |
| WO2019241540A1 (en) | 2018-06-15 | 2019-12-19 | Samumed, Llc | Indazole containing macrocycles and therapeutic uses thereof |
| US11427563B2 (en) | 2018-09-14 | 2022-08-30 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| US11891376B2 (en) | 2018-09-14 | 2024-02-06 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| WO2020150545A1 (en) | 2019-01-17 | 2020-07-23 | Samumed, Llc | Pyrazole derivatives as modulators of the wnt/b-catenin signaling pathway |
| EP4056558A4 (en) * | 2019-11-06 | 2023-12-27 | Jinan University | INDAZOLE COMPOUND, PHARMACEUTICAL COMPOSITION THEREOF AND ITS APPLICATIONS |
| WO2021125229A1 (en) * | 2019-12-17 | 2021-06-24 | 富士フイルム株式会社 | Indazole compound or salt thereof, and pharmaceutical composition |
| WO2021121420A1 (en) * | 2019-12-20 | 2021-06-24 | 江苏凯迪恩医药科技有限公司 | Benzopyrazole compound and intermediate, preparation method, and application thereof |
| WO2021168341A1 (en) | 2020-02-21 | 2021-08-26 | Samumed, Llc | Solid forms of 1-(5-(3-(7-(3-fluorophenyl)-3h-imidazo[4,5-c]pyridin-2-yl)-1h-pyrazolo[3,4-b]pyridin-5-yl)pyridin-3-yl)-n,n-dimethylmethanamine |
| US12071428B2 (en) | 2020-12-30 | 2024-08-27 | Tyra Biosciences, Inc. | Indazole compounds as kinase inhibitors |
| CN112724134A (en) * | 2021-01-14 | 2021-04-30 | 复旦大学 | Azaindazole bipyridine derivative myeloid cell proliferation inhibitor, preparation method and application thereof in pharmacy |
| CN112724134B (en) * | 2021-01-14 | 2022-04-01 | 复旦大学 | Azaindazole bipyridine derivative myeloid cell proliferation inhibitor, preparation method and application thereof in pharmacy |
| US12264149B2 (en) | 2021-12-30 | 2025-04-01 | Tyra Biosciences, Inc. | Indazole compounds as kinase inhibitors |
| WO2023240138A1 (en) * | 2022-06-08 | 2023-12-14 | Blossomhill Therapeutics, Inc. | Indazole containing macrocycles and their use |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6555539B2 (en) | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation | |
| US6462069B2 (en) | Compounds, pharmaceutical compositions, and methods for inhibiting protein kinases | |
| JP4762543B2 (en) | Indazole carboxamide derivatives, processes for their preparation and use as CDK1, CDK2 and CDK4 inhibitors | |
| CA2542609C (en) | Novel indazole derivatives | |
| AU777701B2 (en) | Indazole compounds and pharmaceutical compositions for inhibiting protein kinases, and methods for their use | |
| HRP20020008A2 (en) | Thiazole compounds and pharmaceutical compositions for inhibiting protein kinases and methods for their use | |
| CN105073742A (en) | Flap modulators | |
| WO2011138657A1 (en) | Aryl substituted olefinic compounds as pde10a inhibitors | |
| US6566363B2 (en) | Pyrazole-thiazole compounds, pharmaceutical compositions containing them | |
| JPH10291988A (en) | Heterocyclic compound |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: P-542/02 Country of ref document: YU |
|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| AK | Designated states |
Kind code of ref document: B1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: B1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| B | Later publication of amended claims | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 200203040 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2388885 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 518531 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IN/PCT/2002/00589/MU Country of ref document: IN Ref document number: 29539/01 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2001942620 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PV2002-2391 Country of ref document: CZ Ref document number: 018035523 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10052002 Country of ref document: SK |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 150730 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020027009194 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: 2001 553270 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2002/007058 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 20020570 Country of ref document: UZ Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2001 107011 Country of ref document: BG Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: P20020675A Country of ref document: HR Ref document number: 1200200748 Country of ref document: VN Ref document number: 200200768 Country of ref document: EA |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020027009194 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2001942620 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: PV2002-2391 Country of ref document: CZ |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 518531 Country of ref document: NZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 518531 Country of ref document: NZ |