WO2001005425A2 - Combined preparations comprising daunorubicin derivatives and her2 antibodies - Google Patents
Combined preparations comprising daunorubicin derivatives and her2 antibodies Download PDFInfo
- Publication number
- WO2001005425A2 WO2001005425A2 PCT/EP2000/006540 EP0006540W WO0105425A2 WO 2001005425 A2 WO2001005425 A2 WO 2001005425A2 EP 0006540 W EP0006540 W EP 0006540W WO 0105425 A2 WO0105425 A2 WO 0105425A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- recombmant
- humanized
- antι
- her2
- antibody
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/32—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against translation products of oncogenes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
Definitions
- the present invention pertains to the field of neoplastic disease therapy.
- this invention provides an antitumor composition comprising an alkylating anthracycline and a recombmant humanized ant ⁇ -HER2 antibody, for example the recombmant humanized monoclonal antibody (rhuMab) anti-
- trastuzumab (HerceptmTM) , having a synergistic or additive antmeoplastic effect.
- the present invention provides, in a first aspect, a pharmaceutical composition for use antmeoplastic therapy m mammals, including humans, comprising
- a recombmant humanized ant ⁇ -HER2 antibody and a pharmaceutically acceptable carrier or excipient .
- the recombmant humanized ant ⁇ -HER2 antibody is preferably, the recombmant humanized monoclonal antibody ant ⁇ -HER2 trastuzumab .
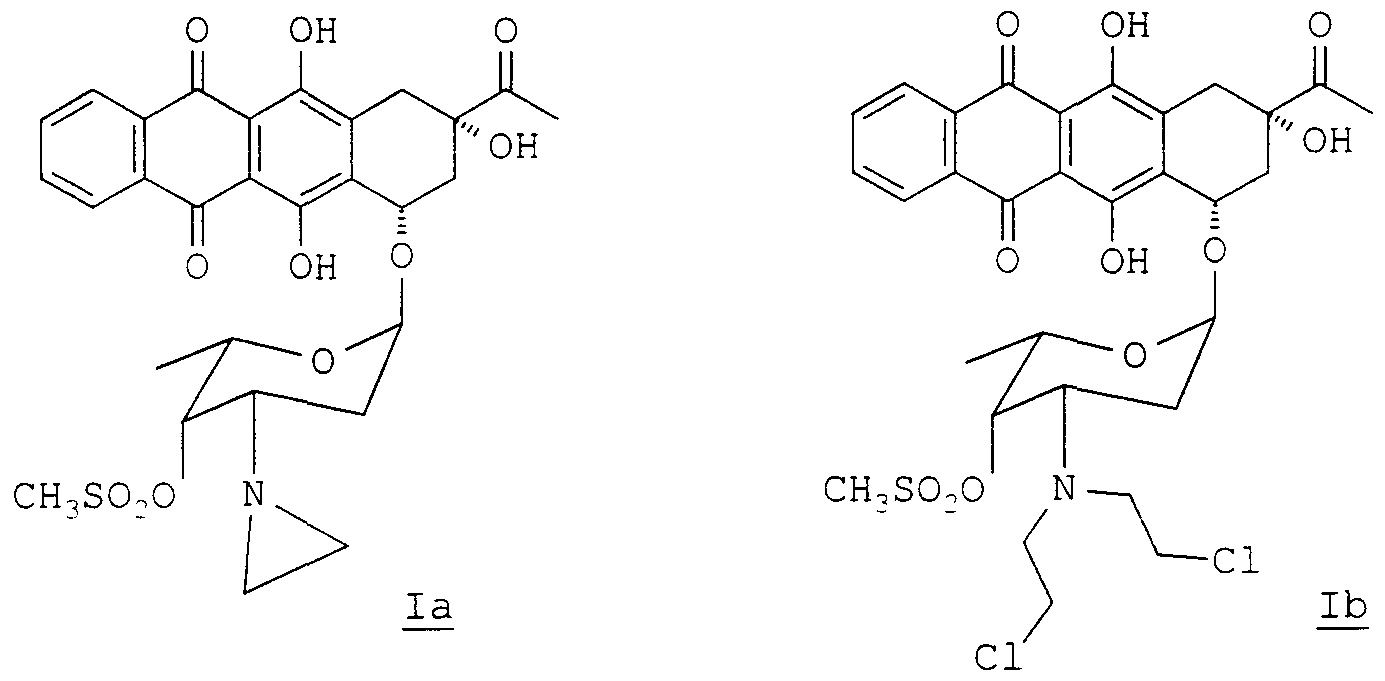
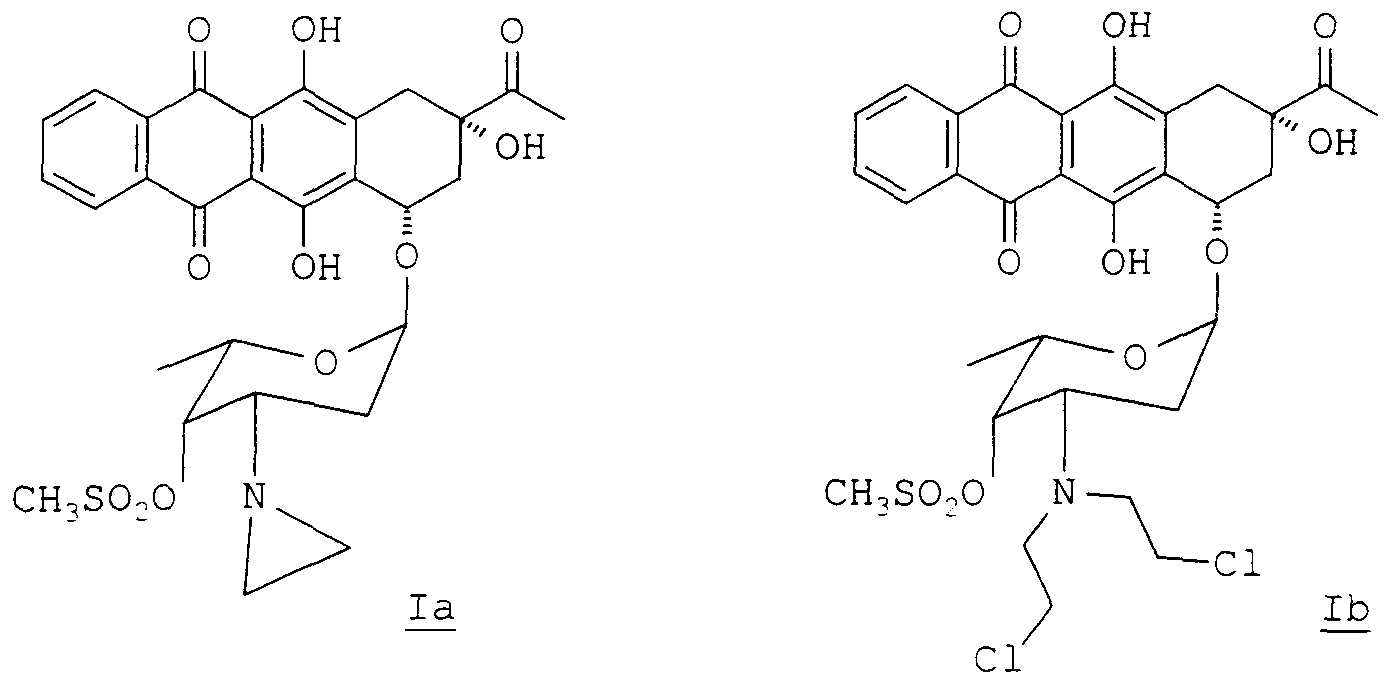
- alkylating anthracyclmes of formula la and lb are 4-demethoxy-3' -deam ⁇ no-3' -az ⁇ r ⁇ dmyl- ' - methansulfonyl daunorubicin (la) and 4-demethoxy-N, N-bis (2- chloroethyl ) -4 ' -methansulfonyl daunorubicin (lb).
- These alkylating anthracyclmes were described in Anticancer Drug Design (1995), vol. 10, 641-653, and claimed respectively in US-A-5, 532, 218 and US-A-5, 496, 800.
- the recombmant humanized monoclonal antibody ant ⁇ -HER2 trastuzumab (HerceptinTM) is described in various scientific publications, for example Cancer Res., 1998, 58:2825-2831.
- the present invention also provides a product comprising an alkylating anthracycline of formula la or lb as defined above and a recombmant humanized ant ⁇ -HER2 antibody, preferably the recombmant humanized monoclonal antibody ant ⁇ -HER2 trastuzumab, as combined preparation for simultaneous, separate or seguential use m antitumor therapy.
- a further aspect of the present invention is to provide a method of treating a mammal, including a human, suffering from a neoplastic disease comprising administering to said mammal an alkylating anthracycline of formula la or lb as defined above and a recombmant humanized ant ⁇ -HER2 antibody, preferably the recombmant humanized monoclonal antibody anti- HER2 trastuzumab, in amounts effective to produce a synergistic antmeoplastic effect.
- a still further aspect of the present invention is to provide a method for lowering the side effects caused by antmeoplastic therapy with an antmeoplastic agent a mammal, including a human, in need thereof, the method comprising administering to said mammal a combined preparation comprising an alkylating anthracycline of formula la or lb as defined above, and a recombmant humanized ant ⁇ -HER2 antibody, preferably the the recombmant humanized monoclonal antibody ant ⁇ -HER2 trastuzumab, in amounts effective to produce a synerqistic antmeoplastic effect.
- a synergistic antmeoplastic effect as used herein is meant the inhibition of the growth tumor, preferably the complete regression of the tumor, administering an effective amount of the combination of an alkylating anthracycline of formula la or lb as defined above and a recombmant humanized ant ⁇ -HER2 antibody to mammals, including humans.
- parenteral is meant intravenous, subcutaneous and intramuscular administration.
- Oral administration includes administering the costituents of the combined preparation m a suitable oral form such as, e.g., tablets, capsules, suspensions, solutions, emulsions, powders, syrups and the like.
- Parenteral administration includes administering the costituents of the combined preparation by subcutaneous, intravenous or intramuscular injections.
- the actual preferred method and order of administration of the combined preparations of the invention may vary according to, inter alia, the particular pharmaceutical formulation of the alkylating anthracycline of formula la or lb as defined above being utilized, the particular pharmaceutical formulation of the recombmant humanized ant ⁇ -HER2 antibody being utilized, the particular cancer being treated, and the particular patient being treated.
- the dosage ranges for the administration of the combined preparation may vary with the age, condition, sex and extent of the disease in the patient and can be determined by one of skill m the art.
- the dosage regimen must therefore be tailored to the particular of the patient's conditions, response and associate treatments m a manner which is conventional for any therapv, and may need to be adjusted in response to changes in conditions and/or in light of other clinical conditions.
- the alkylating anthracycline may be administered simultaneously with the recombmant humanized ant ⁇ -HER2 antibody, or the compounds may be administered sequentially, in either order.
- the course of therapy generally employed is from about
- the course therapy employed is from about 1 to about 50 mg/m 2 of body surface area.
- the course of therapy generally employed is from about 1 to about 1000 mg/m 2 of body surface area. More preferably, the course therapy employed is from about 50 to about 500 mg/m 2 of body surface area.
- the antmeoplastic therapy of the present invention is, in particular, suitable for treating breast, ovary, lung, colon, kidney, stomach, pancreas, liver, melanoma, leukemia and brain tumors m mammals, including humans.
- an alkylating anthracycline according to the invention and a recombmant humanized ant ⁇ -HER2 antibody for example the recombmant humanized monoclonal antibody anti- HER2 trastuzumab, can be suitable for the treatment of patients with cancers over-expressing the HER2 protein, for example, for patient with metastatic breast cancer over- expressing the HER2 protein.
- the antmeoplastic therapy according to this invention also comprises the prevention and/or treatment of tumor metastasis.
- a still further aspect of the present invention is the use of an alkylating anthracycline of formula la or lb as defined above and a recombmant humanized ant ⁇ -HER2 antibody, preferably the recombmant humanized monoclonal antibody anti- HER2 trastuzumab, for the treatment of tumors by angiogenesis inhibition .
- the effectiveness of an alkylating anthracycline of formula la or lb and a recombmant humanized ant ⁇ -HER2 antibody is significantly increased without a parallel increased toxicity.
- the combined therapy of the present invention enhances the antitumoral effects of the alkylating anthracycline of formula la or lb as defined above and of a recombmant humanized ant ⁇ -HER2 antibody and thus yields the most effective and least toxic treatment for tumors.
- the synergistic action displayed by the combined preparations according to the present invention can be shown, for instance, by testing the activity of the combination m mice bearing human tumor xenografts overexpressmg HER2 protein, following, for example, the method described in Cancer Research, 1998, 58:2825-2831.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Immunology (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Biophysics (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Genetics & Genomics (AREA)
- Biochemistry (AREA)
- Oncology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2001510479A JP2003504413A (en) | 1999-07-20 | 2000-07-10 | Combination preparation containing an antitumor substance |
| AU59839/00A AU5983900A (en) | 1999-07-20 | 2000-07-10 | Combined preparations comprising antitumor agents |
| EP00945903A EP1200098A2 (en) | 1999-07-20 | 2000-07-10 | Combined preparations comprising daunorubicin derivatives and anti her2 antibodies |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB9917012.8 | 1999-07-20 | ||
| GBGB9917012.8A GB9917012D0 (en) | 1999-07-20 | 1999-07-20 | Combined preparations comprising antitumor agents |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2001005425A2 true WO2001005425A2 (en) | 2001-01-25 |
| WO2001005425A3 WO2001005425A3 (en) | 2001-05-17 |
Family
ID=10857596
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2000/006540 Ceased WO2001005425A2 (en) | 1999-07-20 | 2000-07-10 | Combined preparations comprising daunorubicin derivatives and her2 antibodies |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP1200098A2 (en) |
| JP (1) | JP2003504413A (en) |
| AU (1) | AU5983900A (en) |
| GB (1) | GB9917012D0 (en) |
| WO (1) | WO2001005425A2 (en) |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002102817A1 (en) * | 2001-06-15 | 2002-12-27 | Pharmacia Italia S.P.A. | Anti-tumour anthracycline glycoside 14-sulfonic acid derivatives |
| US7485302B2 (en) | 1999-06-25 | 2009-02-03 | Genentech, Inc. | Treatment with anti-ErbB2 antibodies and chemotherapeutic agents |
| WO2010108127A1 (en) | 2009-03-20 | 2010-09-23 | Genentech, Inc. | Bispecific anti-her antibodies |
| WO2010136569A1 (en) | 2009-05-29 | 2010-12-02 | F. Hoffmann-La Roche Ag | Modulators for her2 signaling in her2 expressing patients with gastric cancer |
| US7981418B2 (en) | 2007-03-02 | 2011-07-19 | Genentech, Inc. | Predicting response to a HER inhibitor |
| US7993834B2 (en) | 2000-05-19 | 2011-08-09 | Genentech, Inc. | Detection of ErbB2 gene amplification to increase the likelihood of the effectiveness of ErbB2 antibody breast cancer therapy |
| WO2011103242A1 (en) | 2010-02-18 | 2011-08-25 | Genentech, Inc. | Neuregulin antagonists and use thereof in treating cancer |
| WO2011146568A1 (en) | 2010-05-19 | 2011-11-24 | Genentech, Inc. | Predicting response to a her inhibitor |
| EP2399605A1 (en) | 2005-02-23 | 2011-12-28 | Genentech, Inc. | Extending time to disease progression or survival in cancer patients |
| WO2012069466A1 (en) | 2010-11-24 | 2012-05-31 | Novartis Ag | Multispecific molecules |
| WO2012085111A1 (en) | 2010-12-23 | 2012-06-28 | F. Hoffmann-La Roche Ag | Polypeptide-polynucleotide-complex and its use in targeted effector moiety delivery |
| WO2013025853A1 (en) | 2011-08-17 | 2013-02-21 | Genentech, Inc. | Neuregulin antibodies and uses thereof |
| EP2592156A2 (en) | 2007-06-08 | 2013-05-15 | Genentech, Inc. | Gene expression markers of tumor resistance to HER2 inhibitor treatment |
| WO2013081645A2 (en) | 2011-11-30 | 2013-06-06 | Genentech, Inc. | Erbb3 mutations in cancer |
| WO2013083810A1 (en) | 2011-12-09 | 2013-06-13 | F. Hoffmann-La Roche Ag | Identification of non-responders to her2 inhibitors |
| WO2013148315A1 (en) | 2012-03-27 | 2013-10-03 | Genentech, Inc. | Diagnosis and treatments relating to her3 inhibitors |
| WO2014083178A1 (en) | 2012-11-30 | 2014-06-05 | F. Hoffmann-La Roche Ag | Identification of patients in need of pd-l1 inhibitor cotherapy |
| US9551033B2 (en) | 2007-06-08 | 2017-01-24 | Genentech, Inc. | Gene expression markers of tumor resistance to HER2 inhibitor treatment |
| WO2017194554A1 (en) | 2016-05-10 | 2017-11-16 | Inserm (Institut National De La Sante Et De La Recherche Medicale) | Combinations therapies for the treatment of cancer |
| US10167342B2 (en) | 2016-08-29 | 2019-01-01 | Fazel Shokri | Production of hersintuzumab: a new humanized antibody against HER2 for cancer treatment |
| WO2019147152A1 (en) | 2018-01-26 | 2019-08-01 | Universidade Nova De Lisboa | L2a5 antibody or functional fragment thereof against tumour antigens |
| US10689457B2 (en) | 2008-06-16 | 2020-06-23 | Genentech, Inc. | Treatment of metastatic breast cancer |
| EP3698807A1 (en) | 2005-01-21 | 2020-08-26 | Genentech, Inc. | Fixed dosing of her antibodies |
| WO2022074206A1 (en) | 2020-10-08 | 2022-04-14 | Affimed Gmbh | Trispecific binders |
| WO2023007023A1 (en) | 2021-07-30 | 2023-02-02 | Affimed Gmbh | Duplexbodies |
| WO2023078968A1 (en) | 2021-11-03 | 2023-05-11 | Affimed Gmbh | Bispecific cd16a binders |
| WO2025257588A1 (en) | 2024-06-10 | 2025-12-18 | Affimed Gmbh | Cd16a/tumor antigen polyspecific binder for use in the treatment of immune checkpoint inhibitor resistance |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7041292B1 (en) | 1999-06-25 | 2006-05-09 | Genentech, Inc. | Treating prostate cancer with anti-ErbB2 antibodies |
| WO2013063229A1 (en) | 2011-10-25 | 2013-05-02 | The Regents Of The University Of Michigan | Her2 targeting agent treatment in non-her2-amplified cancers having her2 expressing cancer stem cells |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1989006692A1 (en) * | 1988-01-12 | 1989-07-27 | Genentech, Inc. | Method of treating tumor cells by inhibiting growth factor receptor function |
| IL106992A (en) * | 1988-02-11 | 1994-06-24 | Bristol Myers Squibb Co | Acylhydrazone derivatives of anthracycline and methods for their preparation |
| US5705157A (en) * | 1989-07-27 | 1998-01-06 | The Trustees Of The University Of Pennsylvania | Methods of treating cancerous cells with anti-receptor antibodies |
| ZA9811162B (en) * | 1997-12-12 | 2000-06-07 | Genentech Inc | Treatment with anti-ERBB2 antibodies. |
| CA2357525A1 (en) * | 1999-01-27 | 2000-08-03 | Cornell Research Foundation, Inc. | Treating cancers associated with overexpression of her-2/neu |
| US6333348B1 (en) * | 1999-04-09 | 2001-12-25 | Aventis Pharma S.A. | Use of docetaxel for treating cancers |
| AU782325B2 (en) * | 1999-05-14 | 2005-07-21 | Genentech Inc. | Treatment with anti-ErbB2 antibodies |
-
1999
- 1999-07-20 GB GBGB9917012.8A patent/GB9917012D0/en not_active Ceased
-
2000
- 2000-07-10 EP EP00945903A patent/EP1200098A2/en not_active Withdrawn
- 2000-07-10 WO PCT/EP2000/006540 patent/WO2001005425A2/en not_active Ceased
- 2000-07-10 JP JP2001510479A patent/JP2003504413A/en not_active Withdrawn
- 2000-07-10 AU AU59839/00A patent/AU5983900A/en not_active Abandoned
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7485302B2 (en) | 1999-06-25 | 2009-02-03 | Genentech, Inc. | Treatment with anti-ErbB2 antibodies and chemotherapeutic agents |
| US7537931B2 (en) | 1999-06-25 | 2009-05-26 | Genentech, Inc. | Humanized anti-ERBB2 antibodies and treatment with anti-ERBB2 antibodies |
| US7993834B2 (en) | 2000-05-19 | 2011-08-09 | Genentech, Inc. | Detection of ErbB2 gene amplification to increase the likelihood of the effectiveness of ErbB2 antibody breast cancer therapy |
| US8076066B2 (en) | 2000-05-19 | 2011-12-13 | Genentech, Inc. | Gene detection assay for improving the likelihood of an effective response to a HER2 antibody cancer therapy |
| WO2002102817A1 (en) * | 2001-06-15 | 2002-12-27 | Pharmacia Italia S.P.A. | Anti-tumour anthracycline glycoside 14-sulfonic acid derivatives |
| EP3698807A1 (en) | 2005-01-21 | 2020-08-26 | Genentech, Inc. | Fixed dosing of her antibodies |
| US8691232B2 (en) | 2005-02-23 | 2014-04-08 | Genentech, Inc. | Extending time to disease progression or survival in cancer patients |
| EP2399605A1 (en) | 2005-02-23 | 2011-12-28 | Genentech, Inc. | Extending time to disease progression or survival in cancer patients |
| US8940302B2 (en) | 2007-03-02 | 2015-01-27 | Genentech, Inc. | Predicting response to a HER inhibitor |
| EP2899541A1 (en) | 2007-03-02 | 2015-07-29 | Genentech, Inc. | Predicting response to a HER dimerisation inhbitor based on low HER3 expression |
| US7981418B2 (en) | 2007-03-02 | 2011-07-19 | Genentech, Inc. | Predicting response to a HER inhibitor |
| EP2592156A2 (en) | 2007-06-08 | 2013-05-15 | Genentech, Inc. | Gene expression markers of tumor resistance to HER2 inhibitor treatment |
| US10385405B2 (en) | 2007-06-08 | 2019-08-20 | Genentech, Inc. | Gene expression markers of tumor resistance to HER2 inhibitor treatment |
| US9551033B2 (en) | 2007-06-08 | 2017-01-24 | Genentech, Inc. | Gene expression markers of tumor resistance to HER2 inhibitor treatment |
| US11655305B2 (en) | 2008-06-16 | 2023-05-23 | Genentech, Inc. | Treatment of metastatic breast cancer |
| US10689457B2 (en) | 2008-06-16 | 2020-06-23 | Genentech, Inc. | Treatment of metastatic breast cancer |
| EP3088420A1 (en) | 2009-03-20 | 2016-11-02 | F. Hoffmann-La Roche AG | Bispecific anti-her antibodies |
| WO2010108127A1 (en) | 2009-03-20 | 2010-09-23 | Genentech, Inc. | Bispecific anti-her antibodies |
| WO2010136569A1 (en) | 2009-05-29 | 2010-12-02 | F. Hoffmann-La Roche Ag | Modulators for her2 signaling in her2 expressing patients with gastric cancer |
| WO2011103242A1 (en) | 2010-02-18 | 2011-08-25 | Genentech, Inc. | Neuregulin antagonists and use thereof in treating cancer |
| WO2011146568A1 (en) | 2010-05-19 | 2011-11-24 | Genentech, Inc. | Predicting response to a her inhibitor |
| WO2012069466A1 (en) | 2010-11-24 | 2012-05-31 | Novartis Ag | Multispecific molecules |
| WO2012085111A1 (en) | 2010-12-23 | 2012-06-28 | F. Hoffmann-La Roche Ag | Polypeptide-polynucleotide-complex and its use in targeted effector moiety delivery |
| WO2013025853A1 (en) | 2011-08-17 | 2013-02-21 | Genentech, Inc. | Neuregulin antibodies and uses thereof |
| WO2013081645A2 (en) | 2011-11-30 | 2013-06-06 | Genentech, Inc. | Erbb3 mutations in cancer |
| WO2013083810A1 (en) | 2011-12-09 | 2013-06-13 | F. Hoffmann-La Roche Ag | Identification of non-responders to her2 inhibitors |
| WO2013148315A1 (en) | 2012-03-27 | 2013-10-03 | Genentech, Inc. | Diagnosis and treatments relating to her3 inhibitors |
| EP3511718A1 (en) | 2012-11-30 | 2019-07-17 | F. Hoffmann-La Roche AG | Pd-l1 inhibitor |
| WO2014083178A1 (en) | 2012-11-30 | 2014-06-05 | F. Hoffmann-La Roche Ag | Identification of patients in need of pd-l1 inhibitor cotherapy |
| WO2017194554A1 (en) | 2016-05-10 | 2017-11-16 | Inserm (Institut National De La Sante Et De La Recherche Medicale) | Combinations therapies for the treatment of cancer |
| US10167342B2 (en) | 2016-08-29 | 2019-01-01 | Fazel Shokri | Production of hersintuzumab: a new humanized antibody against HER2 for cancer treatment |
| WO2019147152A1 (en) | 2018-01-26 | 2019-08-01 | Universidade Nova De Lisboa | L2a5 antibody or functional fragment thereof against tumour antigens |
| WO2022074206A1 (en) | 2020-10-08 | 2022-04-14 | Affimed Gmbh | Trispecific binders |
| WO2023007023A1 (en) | 2021-07-30 | 2023-02-02 | Affimed Gmbh | Duplexbodies |
| WO2023078968A1 (en) | 2021-11-03 | 2023-05-11 | Affimed Gmbh | Bispecific cd16a binders |
| WO2025257588A1 (en) | 2024-06-10 | 2025-12-18 | Affimed Gmbh | Cd16a/tumor antigen polyspecific binder for use in the treatment of immune checkpoint inhibitor resistance |
Also Published As
| Publication number | Publication date |
|---|---|
| AU5983900A (en) | 2001-02-05 |
| JP2003504413A (en) | 2003-02-04 |
| WO2001005425A3 (en) | 2001-05-17 |
| EP1200098A2 (en) | 2002-05-02 |
| GB9917012D0 (en) | 1999-09-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2001005425A2 (en) | Combined preparations comprising daunorubicin derivatives and her2 antibodies | |
| EP1165069B1 (en) | Antitumour synergistic composition | |
| Seidman et al. | Paclitaxel as second and subsequent therapy for metastatic breast cancer: activity independent of prior anthracycline response. | |
| EP2127652B1 (en) | Method for treating cancer using anticancer agent in combination | |
| US6403563B1 (en) | Antitumor composition containing a synergistic combination of an anthracycline derivative with a camptothecin derivate | |
| EP1323423B1 (en) | Combined preparation comprising a morpholinyl anthracycline derivative and a topoisomerase II inhibitor | |
| CZ296373B6 (en) | Pharmaceutical composition | |
| AU2024201856A1 (en) | Combination therapy using a LIV1-ADC and a chemotherapeutic | |
| Harvey et al. | Phase II study of the amsacrine analogue CI-921 (NSC 343499) in non-small cell lung cancer | |
| US6593303B1 (en) | Anti-tumor synergetic composition | |
| EP0503484B1 (en) | Monoclonal antibodies as antidotes | |
| CN121154844A (en) | Use of pharmaceutical combination containing anti-CD 20 antibody drug conjugate in preparation of drugs for treating NHL | |
| WO2004073719A1 (en) | A combined therapy comprising an indolopyrrolocarbazole derivative and another antitumor agent | |
| MXPA01008646A (en) | Antitumour synergistic composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2000945903 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2000945903 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2000945903 Country of ref document: EP |