PYRIMIDINE COUMPOUNDS
The present invention relates to certain novel pyπmidinone compounds, processes for their preparation, intermediates useful in their preparation, pharmaceutical compositions containing them and their use in therapy, in particular in the treatment of atherosclerosis
WO 95/00649 (SmithKline Beecham pic) describe the phospnohpase A2 enzyme Lipoprotein Associated Phospnohpase A . (Lp-PLA7), the sequence, isolation and purification thereof, isolated nucleic acids encoding the enzyme, and recombinant host cells transformed w ith DNA encoding the enzyme Suggested therapeutic uses for inhibitors of the enzyme included atherosclerosis, diabetes, rheumatoid arthritis, stroke, myocardial infarction, reperfusion injury and acute and chronic inflammation A subsequent publication from the same group further describes this enzyme (Tew D et al. Arteπoscler Thromb Vas Biol 1996 16 591 -9) wherein it is referred to as LDL-PLA2 A later patent application (WO 95/09921. Icos Corporation) and a related publication in Nature (Tjoelker et al. vol 374, 6 April 1995, 549) describe the enzyme PAF-AH which has essentially the same sequence as Lp-PLA2 and suggest that it may have potential as a therapeutic protein for regulating pathological inflammatory events
It has been shown that Lp-PLAi is responsible for the conversion of phosphatidylcholine to lysophosphatidylcho ne. during the conversion of low density lipoprotein (LDL) to its oxidised form The enzyme is known to hydrolyse the sn-2 ester of the oxidised phosphatidv lcho ne to give Ivsophosphatidvlcholine and an oxidativeiv modified fatty acid Both products of Lp-PLA2 action are biologically active with I ophosphatidv lcholine. a component of oxidised LDL, known to be a potent chemoattractant for circulating monocytes As such, lysophosphatidylchohne is thought play a significant role in atherosclerosis bv being responsible for the accumulation of cells loaded with cholesterol ester in the aπeπes Inhibition of the Lp-PLAo enzyme would theretore be expected to stop the build up of these macrophage enriched lesions (by inhibition of the formation of
Ivsophosphatidvlcholine and oxidised free tatty acids) and so be useful in the treatment ot atherosclerosis
The increased Ivsophosphatidvlcholine content of oxidativeiv modified LDL is also thought to be responsible for the endothehal dysfunction observed in patients v\ ith atherosclerosis
Inhibitors of Lp-PLAo could therefore prove beneficial in the treatment of this phenomenon An Lp-PLAi inhibitor could also find utility in other disease states that exhibit endothehal dvsfunction including diabetes, hypertension, angina pectoπs and after
ischaemia and repertusion
In addition. Lp-PL.A.2 inhibitors may also have a general application in anv disorder that involves activated monocytes. macrophages or lymphocytes, as all of these cell types express Lp-PLA^ Examples of such disorders include psoriasis
Furthermore, Lp-PLA2 inhibitors may also have a general application in anv disorder that involves lipid oxidation in conjunction with Lp-PLA7 activity to produce the two injurious products, lysophosphatidylchohne and oxidativeiv modified fatty acids Such conditions include the aforementioned conditions atherosclerosis, diabetes, rheumatoid arthritis, stroke, myocardial infarction, reperfusion injury and acute and chronic inflammation Further such conditions include various neuropsychiatπc disorders such as schizophrenia (see Psychopharmacology Bulletin, 31 , 159- 165, 1995)
Patent applications WO 96/12963. WO 96/13484 WO96/19451 WO 97/02242. WO
97/217675, WO 97/217676, WO 96/41098, and WO 97/41099 (SmithKline Beecham pic) disclose inter alia vaπous series of 4-thιonyl/sulfιnyl/sulfonyl azetidinone compounds which are inhibitors of the enzyme Lp-PLA2 T ese are irreversible, acylating inhibitors (Tew et al. Biochemistry, 37, 10087, 1998)
Patent applications WO 99/24420 and WO 00/10980 (SmithKline Beecham pic, published after the priority date of the present application) describe a new class of reversible, non-acylating inhibitors of the enzyme Lp-PLA . in particular a class of pyrimidone compounds The early 2-(alkylthιo)pyπmιdιn-4-one chemical lead is described in Bioorganic and Medicinal Chemistry Letters. 2000 10. 395-8
A further class of pyπmidone compounds has now been identified w hich are inhibitors of the enz me Lp-PLA2
Accordingly, the present invention provides a compound of formula (I)
R is an aryl or heteroaryl group, optionally substituted by 1, 2.3 or 4 substituents which may be the same or different selected from Cn.igvalkyl, C(]_ιg)aikoxy, C(].j3)alkylthιo, arylC( j.jgvalkoxy, hydroxy, halogen, CN. COR4, carboxy, COOR4, CONR7R8. NR4COR5, SO2NR7R8, NR4SO2R5. NR7R8. mono to perfluoro-C( ι_4)alkyl and mono to perfluoro-C( j^valkoxy, or. as a single substituent, optionally in combination with a further substituent as hereinbefore defined. CFtaCOOH or a salt thereof. CH COOR6, CH2CONR7R8, CH2CN. (CH2)pOH. (CH2)pNR7R8 or (CH2)pOR5. and p is an integer from 1 to 3;
7 R
~ is an aryl or heteroaryl group, optionally substituted by 1.2, 3 or 4 substituents which may be the same or different selected from Cn.igvalkyl. C
( i-jg^alkoxy,
hydroxy, halogen. CN, COR , carboxy, COOR
4, CONR
7R
8, NR
4COR
5. SO
2NR
7R
8, NR
4SO R
5, NR
7R
8, mono to perfluoro-C
( 1.
)alkyl. mono to perfluoro-C
( ι_4)alkoxy, and arylO
1. R3 IS selected from hydrogen, C( ι_20)
a'kyl, C( ι_20
) a'kenyl, arylC
( ι_ιo) lkyl. heteroarylC/
j_ιo)alkyl, C^^O^'kylcarbamoyl, Cπ_20)alkylsulphamoyl. C(l-20)
a"y'
sulP
noπy'
anc* ι_20)
acyl
ar>d in which an aryl or heteroaryl group may have one or two substituents selected from Cr
j _ \ gvalkyl, C(
j . \ g^alkoxy , C
( j . | g^alky lthio, arylC
(1.
18)alkoxy, hydroxy, halogen, CN, COR
4. COOR
4, CONR
4R
5, NR
4COR
5, SO
2NR
4R
5, NR
4SO
2R
5, NR
4R
5, mono to perfluoro-C
( 1.
4)alkyl. or a group -R
9-R
10;
R4 and R^ are independently hydrogen or O^O^'kyl. f°r ιr>stance C(i_4)alkyl (e.g. methyl or ethyl),
R" is C(i_4)alkyl or a pharmaceutically acceptable in vivo hydrolysable ester group; R7 and R8 which may be the same or different is each selected from hydrogen,
C( 1 - 12)alky •• CH2R ' ' • HR ' 2CO2H or a salt thereof, or R7 and R8 together with the nitrogen to which they are attached form a 5- to 7 membered ring optionally containing one or more further heteroatoms selected from oxygen, nitrogen and sulphur, and optionally substituted by one or two substituents selected from hydroxy, oxo. C( ι_4)alkyl, C(i_ 4)alkylCO. aryl, e g. phenyl, or aralkyl, e.g benzyl, for instance morpholine or piperazine.
R' is an ar l or a heteroaryl ring optionally substituted by 1.2. or 3 substituents which may be the same or different selected from C
( ]_ιg
)alkyl. C
(
C
( ] . j gvdlky lthio, ary 1C
( j .
hydroxy, halogen, CN. COR
4, carboxy, COOR
4. CONR
7R
8. NR
4COR
5, SO
2NR
7R
8. NR
4SO
2R
5. NR
7R
8. mono to perfluoro-C
( ]_
4)alky! and mono to perfluoro-O
j_4)alkoxy,
R'" is hydrogen or an aryl ring which is further optionally substituted by 1, 2, 3 or 4 substituents which may be the same or different selected from C( j _ j g^alky 1. C( j_]g)alkoxy. C( ] - 1 g)aikylthιo. ar lC( l.igvalkoxy. hydroxy. halogen. CN, COR4,
carboxy. COOR4, CONR4Rλ NR COR5, SO NR4R5, NR4SO2R5. NR4R5, mono to perfluoro-C( ] _4)alkyl and mono to perfluoro-C( i_4)alkoxy;
R 1 1 is COOH or a salt thereof. COOR6, CONR7R8, CN. CH2OR5 or CH2OH;
R '^ is an a ino acid side chain such as CJiJOH from seπne;
n is an integer from 1 to 4. preferably 1 or 3;
X is O or S: and
Z is CR ^R 1 where R and R are each hydrogen or C^ i _4)alkyl. or R ^ and R '4 together with the intervening carbon atom form a Cπ_fj)cycloalkyl πn?-
Compounds of formula (I) are inhibitors of Lp-PLA-> and as such are expected to be of use in treating atherosclerosis and the other disease conditions noted elsewhere. Such compounds are found to act as inhibitors of Lp-PLA in in vitro assays ..
Preferably, Z is CH2.
Preferably R ' is a 5- or 6- membered, monocyclic heteroaryl group containing 1 or 2 nitrogen heteroatoms, more preferably pyridyl, pyrimidyl or pyrazolyl: yet more preferably, pyrimidyl, optionally substituted by 1 or 2 substituents preferably selected from oxo, arylC( ] _4)alkyl (e.g. benzyl), C^ .^-jalkyl (e.g. methyl or ethyl), C(3_6)cycloalkyl hydroxy,
C( ]_4)alkoxy (e.g. methoxy). carboxy
j .
β alkyl, C .6)
a'kylcarboxyC( j_ )alkyl. diC( t
j.gvalkyl (e.g. methyl or ethyl).
Preferably, ZR ' IS pyπmιd-5-ylmethyl. 2-oxo-pyπmιd-5-ylmethyl, 2-methoxypyπmιd-5-yl or l-methylpyrazol-4-yimethyi.
Representative examples of R ' when an aryl group include phenyl and naphthyl.
Representative examples of R- when an aryl group include phenyl and naphthyl
Representative examples of R- when a heteroaryl group include pyridyl, pyπmidin l, pyrazolyl. furanyl, thienyl. thiazolyl. quinolyl.benzothiazolyl. pyπdazolyl and pyrazinyl. Preferably. R- is phenyl optionally substituted by halogen.
Preferably X is S.
Representative examples of R-CHτX include 4-fluorobenzylthio.
Representative examples of
C
].20
) alkylcarbamoyl, C(i_20
) lkvlsulphonyl. in which the alkyl moiety is preferably C
(6_i2
)alkyl, phenylC( ι_4)alkyl. or R"
Representative examples of R" include phenyl optionally substituted by chloro, dichloro, trifluoromethyl or R'^ Representative examples of R'^ include phenyl optionally substituted by halogen, or trifluoromethyl, preferablv at the 4-posιtιon Preferably. R" and R.O together form a 4-(phenvl)-phenvl substituent in which the remote phenyl ring may be optionally substituted bv halogen or trifluoromethyl, preferably at the 4-posιtιon
Preferablv. m is 0
Representative examples of values of pharmaceutically acceptable //; wo hvdrolvsable ester groups for R° include -CH(Ra)O CO Rb.
-CH(Ra)OCOORc,
-CH(Ra)CO NReRf
-RdNReRf,
-CH2ORg.
~CH2 R h
X O- ,o
O R')NH2. and
R
a is hydrogen, (C
j-
fj lkvl in particular methyl
or phenvl. each of which may be optionally substituted,
Rb is (Cι-6)alkyl. (Cι-6)alkox (C
]- )al vl, phenyl. benzyl, (C^Jcycloalkyl
l-amιno(C
|-6)alkyl, or 1-(C
j-6alkvl)amιno(Cι-6)alkvl. each of which may be optionallv substituted, or
Ra and RD together form a 1.2-phenvlene group optionally substituted bv one or two methoxy groups,
Rc is (Cj-6)alkyl, (C3-7)cycloalkyl, (Cι-6)alkvl(C3-7cycloalkyl,
R" is (Cι- )alkylene optionally substituted with a methvl or ethyl group
Re and R^ which may be the same or different is each (C ] -0)alkvl or aryl( C ι -4 alk l. optionally substituted with e.g hydroxy, R§ ιs (C 1 -6)alkyl.
Rn is hydrogen, (C | - )alkyl or phenyl. R1 is hydrogen or phenyl optionally substituted by up to three groups selected from halogen,
(C ι -6)-alkyl. or (C ] - )alkoxy, and γ l is oxygen or NH. for instance (a) acyloxyalkyl groups such as acetoxymethv l. isobutyryloxymethyl, pivaloyloxymethyl, benzoyloxymethyl. α-acetoxyethyl. α-pivaloy loxyethyl, l -(cyclohexylcarbonyloxy)ethyl, ( l-amιnoethyl)carbonyloxymethyl, 2-methoxyprop-2-v lcarbony loxymethy 1, phenylcarbonyloxymethyl and 4-methoxyphenvl-carbonyloxymethyl,
(b) alkoxy/cycloalkoxycarbonyloxyalkyl groups, such as ethox carbonyloxymethyl, t- butyloxycarbonyloxymethyl, cyclohexyloxycarbonyioxymethyl. 1 - methylcyclohexyloxycarbony loxymethy 1 and α-ethoxycarbonyloxyethyl,
(c) dialkylaminoalkyl, especially di-loweralkylamino alkyl groups such as dimethylaminomethyl, dimethylaminoethyl. diethylaminomethyl or diethylaminoethyl.
(d) acetamido groups such as N.N-dimethylaminocarbonylmethyl, N.NJ2- hydroxyethyl)amιnocarbonylmethyl,
(e) lactone groups such as phthahdyl and dimethoxyphtha dyl.
(f) (5-methyl-2-oxo- l J-dιoxolen-4-yl)methyl, and
(g) (2-methoxycarbonyl-£-but-2-en-yl)methyl
Representative examples of pharmaceuticalK acceptable in
o hydrolysable ester groups for R" include
(2-methoxycarbonyl-/f-but-2-en-yl)methyl, isobutyryloxymethv 1. 2-methoxyprop-2- ylcarbonyloxymethyl, phenylcarbonvloxvmeth l, 4-methoxyphenyl-carbonyloxymethy 1, t- butyloxycarbonyloxy methy 1, cyclohexy loxy-carbonyloxymethy 1. 1 - methylcyclohexyloxycarbonyloxymethyl. N.N-dimethylaminocarbon lmethv 1. and (5- methyl-2-oxo- l J-dιoxolen-4-yl)methyl
It will be appreciated that in some instances, compounds of the present invention may include a carboxy group as a substituent Such carboxy groups may be used to form salts. in particular pharmaceutically acceptable salts Pharmaceutically acceptable salts include those described by Berge. Bighley. and Monkhouse, J Pharm Sci , 1977, 66, 1- 19 Preferred salts include alkali metal salts such as the sodium and potassium salts
It will be appreciated that in some instances, compounds of the present invention may include a basic group, such as an ammo group, as a substituent Such basic groups may be used to form acid adddition salts, in particular pharmaceutically acceptable salts. Pharmaceutically acceptable salts include those described by Berge. Bighley. and Monkhouse, J Pharm Set.. 1977, 66. 1J 9
When used herein, the term "alkvl" and similar terms such as 'alkoxv" includes all straight chain and branched isomers Representative examples thereof include methyl, ethyl, n- propyl. .so-propyl. /i-butyl, sec-buty\. / 0-butyl, f-butyl, /.-pentyl and /i-hexyl
When used herein, the term "aryl" refers to, unless otherwise defined, a mono- or bicychc aromatic ring system containing up to 10 carbon atoms in the ring system, for instance phenyl or naphthyl
When used herein, the term "heteroaryl" refers to a mono- or bicychc heteroaromatic ring system comprising up to four, preferably 1 or 2, heteroatoms each selected from oxygen, nitrogen and sulphur Each ring may have from 4 to 7, preferably 5 or 6, ring atoms. A bicychc heteroaromatic ring system may include a carbocyclic ring
When used herein, the terms "halogen ' and "halo" include fluorine, chlorine, bromine and iodine and fluoro, chloro, bromo and lodo, respectively.
Particularly preferred compounds of formula (I) include
1 -(4-(3J-Dιchloropheny l)pιperazιn- 1 -y lcarbony lmethv l)-2-(4-fluorobenzv 1 )thιo-5-(2- methoxypyπmιd-5-> lmethyl)pyπmιdιn-4-one, l-(4-(4-Chlorophenyl)pιperazιn- 1 -y lcarbony lmethyl)-2-(4- fluorobenzyl )thιo-5-(2- methoxypyπmιd-5-ylmethyl)pyπmιdιn-4-one, l-(4-(3J-Dιchlorophenyl)pιρerazιn- l -y lcarbony lmethyl)-2-(4-fluorobenzyl)thιo-5-( I - methylpyrazol-4-ylmethyl)pyπmιdιn-4-one. and l -(3-(4-(4-Tπfluoromethylphenyl)pιperazιn- l -ylcarbonyl)prop- l -yI)-2-(4- fluorobenzyl)thιo-5-(2-methoxypyπmιd-5-ylmethyl)pyπmιdιn-4-one
Since the compounds of the present inv ention, in particular compounds of formula (I), are intended for use in pharmaceutical compositions, it will be understood that they are each provided in substantially pure form, for example at least 50% pure, more suitably at least
75% pure and preferably at least 95% pure (% are on a wt vvt basis) Impure preparations of the compounds of formula (I) may be used for preparing the more pure forms used in the pharmaceutical compositions Although the purity of intermediate compounds of the
present invention is less critical, it will be readilv understood that the substantially pure form is preferred as for the compounds of formula (I) Preferably whenever possible, the compounds of the present invention are obtained in crystalline form
When some of the compounds of this invention are allowed to crv stal se or are recrystalhsed from organic solvents, solvent of crystallisation may be present in the crystalline product This invention includes within its scope such solvates Similarly, some of the compounds of this invention may be crystallised or recrystalhsed from solvents containing water In such cases water of hydration may be formed This invention includes within its scope stoichiometπc hydrates as well as compounds containing v ariable amounts of water that may be produced by processes such as lyophi sation In addition, different crystallisation conditions may lead to the formation of different polymorphic forms of crystalline products This invention includes within its scope all polymorphic forms of the compounds of formula (I)
Compounds of the present invention are inhibitors of the enzyme lipoprotein associated phospnohpase A2 (Lp-PLA2) and as such are expected to be of use in therapy, in particular in the treatment of atherosclerosis In a further aspect therefore the present invention provides a compound of formula (I) for use in therapy
The compounds of formula (I) are inhibitors of lysophosphatidylchohne production by Lp- PLA2 and may therefore also have a general application in any disorder that involves endothehal dysfunction, for example atherosclerosis, diabetes, hypertension, angina pectoris and after ischaemia and reperfusion In addition, compounds of formula (I) av have a general application in any disorder that involves lipid oxidation in conjunction w ith enzyme activity, for example in addition to conditions such as atherosclerosis and diabetes other conditions such as rheumatoid arthritis, stroke, inflammatory conditions of the brain such as Alzheimer's Disease, myocardial infarction, reperfusion injury, sepsis, and acute and chronic inflammation Further such conditions include v arious neuropsvchiatπc disorders such as schizophrenia ( see Psychopharmacology Bulletin, 31. 159- 165. 1995)
Further applications include any disorder that involves activated monocy tes macrophages or lymphocytes, as all of these cell types express Lp-PLA, Examples of such disorders include psoriasis
Accordingl , in a further aspect, the present invention provides for a method of treating a disease state associated with activ itv of the enzyme Lp-PLA which method in olv es treating a patient in need thereof with a therapeuticallv effectiv e amount of an inhibitor of
the enzyme The disease state may be associated with the increased involvement of monocytes. macrophages or lymphocytes, with the formation of lysophosphatidylchohne and oxidised free fatty acids, with lipid oxidation in conjunction with Lp PLA2 activity, or with endothehal dysfunction
Compounds of the present invention may also be of use in treating the above mentioned disease states in combination with anti-hyper pidaemic or anti-atherosclerotic or anti- diabetic or anti-anginal or anti-inflammatory ananti-hypertension agent or an agent for lowering Lp(a) Examples of the above include cholesterol synthesis inhibitors such as statins, anti-oxidants such as probucol, insulin sensmsers, calcium channel antagonists, and anti-inflammatory drugs such as NSAIDs Examples of agents for lowering Lp(a) include the arrunophosphonates described in WO 97/02037, WO 98/28310. WO 98/2831 1 and WO 98/28312 (Symphar S A and SmithKline Beecham)
A preferred combination therapy will be the use of a compound of the present invention and a statin The statins are a well known class of cholesterol lowering agents and include atorvastatin. simvarstatin, pravastatin, ceπvastatin, fluvastatin, lovastatin and ZD 4522 (also referred to as S-4522. Astra Zeneca) The two agents may be administered at substantially the same time or at different times, according to the discretion of the physician
A further preferred combination therapy will be the use of a compound of the present invention and an anti-diabetic agent or an insulin sensitiser, as coronary heart disease is a major cause of death for diabetics Within this class, preferred compounds for use with a compound of the present invention include the PPARgamma activators, for instance G1262570 (Glaxo Wellcome) and also the glitazone class of compounds such as rosig tazone ( Λ.vandιa. SmithKline Beecham). trog tazone and piog tazone
In therapeutic use. the compounds of the present invention are usually administered in a standard pharmaceutical composition The present invention therefore provides, in a further aspect, a pharmaceutical composition comprising a compound of formula (I) and a pharmaceutically acceptable carrier
Suitable pharmaceutical compositions include those w hich are adapted for oral or parenteral administration or as a suppositorv
Suitable pharmaceutical compositions include those which are adapted for oral or parenteral administration or as a suppository Compounds of formula (I) which are active when given orally can be formulated as liquids, for example syrups, suspensions or emulsions, tablets, capsules and lozenges A liquid formulation ill generally consist of a suspension or
solution of the compound or pharmaceutically acceptable salt in a suitable liquid carner( s) for example, ethanol. glycerine, non-aqueous solvent, for example polyethylene glycol. oils, or water with a suspending agent, preservative, flavouring or colouring agent A composition in the form of a tablet can be prepared using anv suitable pharmaceutical carπer(s) routinelv used for preparing solid formulations Examples of such carriers include magnesium stearate. starch, lactose, sucrose and cellulose A. composition in the form of a capsule can be prepared using routine encapsulation procedures For example, pellets containing the active ingredient can be prepared using standard carriers and then filled into a hard gelatin capsule, alternatively, a dispersion or suspension can be prepared using anv suitable pharmaceutical carner(s), for example aqueous gums, celluloses, silicates or oils and the dispersion or suspension then filled into a soft gelatin capsule Typical parenteral compositions consist of a solution or suspension of the compound of formula (I) in a sterile aqueous carrier or parenterally acceptable oil. for example polyethylene glycol. poly vinyl pyrrohdone, lecithin, arachis oil or sesame oil Alternatively , the solution can be lyophi sed and then reconstituted with a suitable solvent just prior to administration A typical suppository formulation comprises a compound of formula (I) which is active when administered in this way, with a binding and/or lubricating agent such as polymeric glycols. gelatins or cocoa butter or other low melting vegetable or synthetic waxes or fats
Preferably the composition is in unit dose form such as a tablet or capsule Each dosage unit for oral administration contains preferably from 1 to 500 mg (and for parenteral administration contains preferably from 0 1 to 25 mg) of a compound of the formula (I) The daily dosage regimen for an adult patient may be, for example, an oral dose of between 1 mg and 1000 mg. preferably between 1 mg and 500 mg, or an intrav enous subcutaneous or intramuscular dose of between 0 1 mg and 100 mg, preferablv between 0 1 mg and 25 mg, of the compound of the formula (I), the compound being administered 1 to 4 times per day Suitably the compounds will be administered for a period of continuous therapy, for example for a week or more
A compound of formula (I) may be prepared by a number of processes which include
(a) reacting a compound of formula (II) o
N Z R1
Ft N
(CH2)n COOH
(ID
1 in which X. Y, Z, R ' and R- are as hereinbefore defined, with a compound of formula (III):
(III) in which m and R^ are as hereinbefore defined, under amide forming conditions;
(b) reacting a compound of formula (IV)
(IV) in which X, Z, R ' and R- are as hereinbefore defined, with a compound of formula (V):
R 1 5-COJCH2)n-L1
(V) in which n is as hereinbefore defined, L ' IS a leaving group such as halogen, for instance bromo or lodo. and R^ is.
in which R-
"1 is as hereinbefore defined, in the presence of a base such as a secondary or tertiary amine. for instance di-isopropyl- ethylamine, in an inert solvent such as dichloromethane.
(c) when X is S. reacting a compound of formula (VI)
(VI) in which n, m. Z. R ' .and R^ are as hereinbefore defined, with a compound of formula (VII):
R^-CFb-L
(VII) in which R- and L 1 are as hereinbefore defined, in the presence of a base such as a secondary or tertiary amine, for instance di-isopropyl- ethylamine, in an inert solvent such as dichloromethane: or
(d) when X is O, reacting a compound of formula (VIII):
(VIII) in which n. m. Z. R
1. and R
J are as hereinbefore defined, and L- is a leaving group such as halogen or alkylthio. for instance methylthio, with a compound of formula (IX)
R2-CH7-OH
(IX) in which R- is as hereinbefore defined.
in the presence of a base such as 4-dιmethylamιnopyπdιne. in a solvent such as pyndine.
Compounds of formulae (II), (IV), (VI) and (VIII) for use in the above processes may be prepared by processes illustrated in the following scheme I:
CICO- C - Z - R L30,C
CH
CH L O-CCHj — Z — R
CH30 (W D
HO (XVIII)
Scheme I in which:
LJ is a C( l -6)alkyl group, for instance methyl.
R '° is a C( i_ )al yl group, for instance methyl, ethyl or .-but l, and L ' , L-, R ' , R-, RJ , m. n, X and Z are as hereinbefore defined
With reference to Scheme I
Amide forming conditions for step (a) are well known in the art Preferablv. the acid of formula (II) is reacted with the amine of formula (III) in an inert solvent, such as dichloromethane at ambient temperature and in the presence of an activating agent such as as l-(3-dιmethvlamιnopropyI)-3-ethylcarbodnmιde plus hydroxybenzotπazole
Alkylation conditions for step (b) include reaction in the presence of a base such as a secondary or tertiary amine, for instance di-.iO-propylethvlamine. in an inert solvent such as dichloromethane, forming an intermediate ester which is converted to the acid of formula (II) by hydrolysis, for instance using aqueous sodium hvdroxide in a solvent such as dioxan or by alternative deprotection, for instance using tπfluoroacetic acid in a solvent such as dichloromethane
Conditions for step (c) include under thioether forming conditions Advantageously, the reaction is carried out in the presence of a base such as sodium ethoxide or potassium carbonate, preferably in a solvent such as ethanol or dimethy l formamide, or a secondary or tertiary amine base such as di-isopropylethyl amine, in solvent such as dichloromethane
In step (d), a compound of formula (XVII) is reacted with thiourea, in the presence of sodium ethoxide (preferably generated in situ from sodium and ethanol)
In step (e), a compound of formula (XVIII) is reacted with ethy l formate in the presence of a base such as sodium hydride or potassium isopropoxide
In step (f). a compound of formula (IV) is reacted with a compound of formula (V) in the presence of a base such as a secondary or tertiary amine, for instance di-isopropyl- ethylamine. in an inert solvent such as dichloromethane
In step (g), a compound of formula (XIII) is reacted with a compound of formula (XIV) in a solvent such as dimethylformamide to form an intermediate thiourea. which is then treated with a base such as sodium methoxide
In step (h). a compound of formula (XVI) is reacted with a metal thiocyanate. for example potassium thiocvanate. in a solvent such as acetonitπle
In step (1), a compound of formula (XVII) is reacted with a methylating agent such as dimethyl sulphate in the presence of a base such as potassium carbonate, followed by hydrolysis of the intermediate ester in conventional manner e g by basic h drolvsis using sodium hydroxide to give the corresponding carboxylic acid which may then be converted into the acv l chloride, for instance by treatment with oxalyl chloride
In step (j), a catalyst such as 4-dιmethylamιnopyπdιne, and a solvent such as pyndine are used
In step (k), a compound of formula (XIII) is reacted with a compound of formula (XV) in a solvent such as dimethvlformamide to form an intermediate thiourea. which is then treated with a base such as sodium methoxide
The present invention will now be illustrated by the following examples
The present invention will now be illustrated by the following examples
Examples
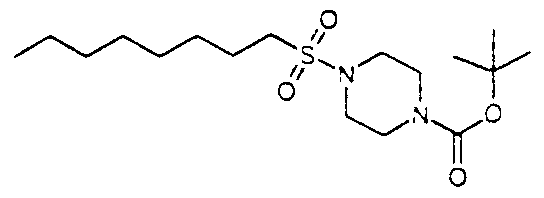
Intermediate Al — l-(Oct-l-ylsulfonyl)-4-tert-butoxycarbonylpiperazine
A solution of 1 -Boc-pιperazιne (5 O l g) and diisopropylethylamine (5.1 1 ml) in dichloromethane (30ml) was added dropwise over lOmin to a solution of 1 -octanesulfonyl chloride (5 26ml) in dichloromethane (30ml). After standing at room temperature overnight, the mixture was washed with aqueous ammonium chloπde, and the organic layer dried and evaporated. Chromatography (silica, 2J hexane/ethyl acetate) gave the required product (9.6g). Η-NMR (CDCI3) δ 3.51 (4H, m), 3.23 (4H, t), 2.89 (2H, m), 1.80 (2H, m),
1.47 (9H. s). 1.38- 1.51 (4H, m). 1.28 (8H, m), 0.89 (3H. m). MS(APCI+) found (M- BOC+H)=263. C 12H26N2O2S requires 262.
Intermediate A2 — l-(Oct-l-ylsulfonyl)piperazine hydrochloride
WOct- l -ylsulfonyl)-4-.er.-butoxycarbonylpiperazιne (Example 1 , 5.08g) was dissolved in
HCl/dioxan (50ml, 4M solution) and stirred at room temperature for 1.5h. The mixture was diluted with diethyl ether, and the solid filtered off (2.9g). ] H-NMR (MeOH-d4) δ 3.58 (4H, m), 3.34 (4H, m), 3.14 (2H, m), 1.80 (2H, ), 1.49 (2H, m), 1.33- 1 40 (8H, m), 0.93 (3H.m); MS(APCI+) found (M+ l )=263, C 12H26N2θ2 requires 262.
Intermediate A3 — l-(Oct-l-vlaminocarbonyl)-4-tert-butoxycarbonyIpiperazine
An equimolar mixture of 1 -Boc-pιperazιne and 1 -octylιsocyanate in dichloromethane was stirred at room temperature for 3h Evapoaration of the solvent gave the desired product as a white solid ' H-NMR (CDCI3) δ 4 42 ( 1 H. m), 3.43 (4H. m). 3.35 (4H. ), 3.20 (2H, m). 1.28- 1.52 (21 H. m), 0.88 (3H. m), MS(APCI-) found (M- l ) = 340. C 18H35N3O3 requires
341.
Intermediate A4 — l-(Oct-l-ylaminocarbonyl)piperazine
Prepared from Intermediate A3 by the method of Intermediate A2 Η-NMR (DMSO) δ 9.30 ( 1 H, br s), 6.69 ( 1 H, t), 3.49 (4H. ), 2.98 (6H, m). 1 15- 1 42 ( 10H. m). 0 86 (3H, m);
MS(APCI÷) found (M+23) = 264. C 13H27N O requires 241
Intermediate A5 — l-(5-Chloropyrid-2-yl)piperazine
Piperazine ( l Og) was heated to 170°C, and 2-bromo-5-chloropyridine (3.56g) added portion wise with stirring over 10 min. Heating was continued for a further 30 min. then the mixture was cooled below 100°C and water (ca 80ml) added. After cooling to room temperature the solution was filtered, extracted with methyl t-butyl ether, and the organic layer dried and evaporated. Chromatography (silica, methanolic ammonia in dichloromethane) gave the desired product as a white solid. H-NMR (CDCI3) δ 2.97 (4H,m). 3.46 (4H,m). 6.57 (2H,d), 7.41 ( l H.dd), 8.1 1 ( lH.d): MS (APCI+) found (M+ l ) = 198/200: C9H 1 2C1N3 requires 197/199.
Intermediate A6 — l-(3,5-Dichloropyrid-2-yl)piperazine
Prepared from 2-bromo-3.5-dichloropyridine by the method of Intermediate A5. Η-NMR (CDCI3) δ 3.02 (4H.m), 3.28 (4H,m), 7.59 (2H.d), 8.12 ( l H.d); MS (APCI+) found (M+ l ) = 232; C9H 1 ι 35Cl2N3 requires 231.
Intermediate A7 — l-(Dec-l-yl)-2-oxo-4-(t-butoxycarbonyi)piperazine
Sodium hydride (OJg, 60% in oil) was washed with pet. ether then suspended in dry DMF (20ml). and 2-oxo-4-(.-butoxycarbonyI)piperazine ( l og) added portionwise. After stirring for 1.5h this gave a clear yellow solution, which was mixed with 1 -bromodecane ( 1.56ml) and stirred at room temperature overnight. Water was added, and the product extracted into pet. ether, dried and evaporated to a crude oil, which was used without further purification. 1 H-NMR (CDCI3) δ 0.88 (3H.t), 1.26 ( 14H.m). 1.47 (9H.s), 1 A- 1 .5 (2H.m). 3.33 (2H,m). 3.40 (2H.m), 3.62 (2H,m). 4.06 (2H.s).
Intermediate A8 — l-(Dec-l-yI)-2-oxopiperazine hydrochloride
Intermediate A7 (total crude) was dissolved in dioxan ( 10ml ). and HCl (->ml. 4M in dioxan) added. After stirring for l h at room temperature the mixture was diluted with ether, and the solid product filtered off and washed with ether. ' H-NMR (DMSO) δ 0.86 (3H,t), 1.25 ( 14H.mj, 1.46 (2H.m), 3.2-3.4 (4H+H2O,m), 3.52 (2H.m). 3.64 (2H.s); MS (APCI+) found
(M+ 1 ) = 241 , (2 +H) = 481 ; C , 4H28N2O requires 240.
Intermediate A9 — l-Bromoacetyl-4-(4-chlorophenyi)piperazine
/ — \
Sodium bicarbonate (50.4g) was added portionwise to a suspension of l -(4-chlorophenyl )- piperazine dihydrochloride (50g) in water (270ml), then dichloromethane (400ml) was added and the 2-phase mixture cooled in an ice bath. Bromoacety! bromide ( 16.1 ml) was added, and stirring continued for 1 h before dilution with water and extraction with dichloromethane. Drying and evaporation of the solvent gave the required product (43Jg). Η-NMR (CDC13) δ 3.15 (2H.t). 3.22 (2H.t). 3.67 (2H.t), 3.78 (2H.t), 3.90 (2H.s), 6.85 (2H.m). 7.24 (2H.m).
Intermediate A10 — l-(4-(4-FluorophenyI)phenyl)piperazine
A mixture of l -(4-bromophenyl)piperazine hydrochloride ( 1.39g), 4-fluorobenzeneboronic acid (0.70g), 2M aqueous sodium carbonate (7.5ml), 1 ,2-dimethoxyethane (20ml) and palladium acetate (56mg) was stirred at reflux under argon overnight, then cooled, diluted with water and ethyl acetate, and filtered to remove undissolved solid. The organic layer was separated, dried and evaporated. Chromatography (silica, 5- 10% MeOH/NH3 in
CH2C12) gave the desired product (0.98g) as a pale brown solid. ! H-NMR (CDCI3) δ 2.84 (4H,m), 3.09 (4H.m), 6.98 (2H.d), 7.21 (2H,m), 7.49 (2H,m), 7.62 (2H,m); MS (APCI+) found (M+l ) = 257; C 1 6H 1 7FN2 requires 256.
Intermediate All — l-(4-(4-ChlorophenyI)phenyI)piperazine mXXXXa
Prepared from 4-chlorobenzeneboronic acid by the method of Intermediate A 10. Η-NMR (CDCI3) δ 3.05 (4H.m), 3.18 (4H.m), 6.98 (2H.d). 736 (2HJ), 7J7 (4H.m); MS (APCI+) found (M+ l ) = 273; C 1 6H I 7C1N2 requires 272.
Intermediate A12 — l-(4-(4-Trifluoromethylphenyl)phenyl)piperazine
HN -\ — <\=\ ,) / (x=\ ,) {- F
Prepared from 4-trifluoromethylbenzeneboronic acid by the method of Intermediate A 10. Η-NMR (CDCI3) δ 3.05 (4H.m). 3.22 (4H.m). 7.00 (2H.d). 7.53 (2HJ), 7.65 (4H.s); MS (APCI+) found (M+ l ) = 307; C 17H 17F3 2 requires 306.
Intermediate A13 — l-(4'-Biphenyl )piperazine
/ — /=\ /=\
HN — <• ,) (, ,)
Prepared from benzeneboronic acid by the method of Intermediate A 10. The crude product proved difficult to purify because of poor solubility. H-NMR (CDCI3) δ 2.84 (4H,m). 3.08 (4H.m). 6.99 (2H,m), 7.26 ( l H.m). 7.40 (2H.m). 7.52 (2H,m). 7.58 (2H.mκ MS (APCI+) found (M+ l ) = 239; C | 6H ] gN requires 238.
Intermediate Bl - Ethyl 3-(5-pyrimidinyl)acryiate
A mixture of 5-bromopyπmιdιne (5 93g), ethyl aery late (5 08g), palladium acetate (0 1 12g), tπphenyl phosphine (0 23g) and tπethylamine (4 5g) was stirred at I50°C in a pressure vessel for 6 hours After cooling overnight, water (50ml) was added to the dark residue, and the product was extracted into toluene Drying, charcoaling and evaporation gave a pale oil, which was triturated w ith pet ether to obtain ethyl 3-(5-pyπmιd l)acrylate (4 78g) H-NMR (CDCI3) δ 1 36 (3H.t), 4 27 (2H,q), 6 59 ( lH,d). 7 62 ( l H.d), 8 88 (2H.s), 9 20 ( lH,s)
Intermediate B2 - Ethyl 3-(5-pyrimidyI)propionate
To a solution of ethy l 3-(5-pyπmιdyl)acrylate (4 75g) in ethanol (90ml) as added 5% palladium on charcoal (0 2g) The mixture was hydrogenated at an initial pressure of 50 psi, then filtered to remove catalyst and the solvent evaporated Water was added, and the product extracted into ether Drying, charcoaling and evaporation gave ethyl 3-(5- pyπmidy propionate (2 3g) as a yellow oil ' H-NMR (CDCI3) δ 1 23 (3H.t), 2 69 (2H,t), 2 96 (2H,t), 4 14 (2H,q), 8 635 (2H,s) and 9 09 ( lH,s)
Intermediate B3 - Ethyl 2-formyl-3-(5-pyrimidyl)propionate o
EIO ' HO' .J A mixture of ethyl 3-(5-pyπmιdvl)propιonate (2 2Sg) and ethyl formate ( 1 41 ml) dissolved in dry dimethoxyethane ( 5ml) was added dropwise over 30 min to a suspension of sodium hydride (60% 4 Og) in DME (5 ml) under nitrogen, keeping the temperature below 0°C Stirring was continued for a further 24h, then the mixture was poured onto ice and washed with ether The aqueous layer was adjusted to pH 7 then evaporated and the residue extracted with acetone Filtration and evaporation gave crude product hich was taken up in ethy l acetate, charcoaled, dried and evaporated to give ethyl 2-formv -(5-pvπmιdyl)- propionate Like other compounds of this type, this proved difficult to characterise and was used without further purification
Intermediate B4 -
πmidvl)acrJιc acid
A mixture of 5-bromopyπmιdιne ( lOOg), acrv c acid (48g). tπphenvlphosphine (2 5g) and palladium II acetate (0 l g) was refluxed in tπ-n-butvlamine (260ml ) w ith o erhead stirring for 4 5h at 145- 160°C The mixture was cooled and a 10% solution of potassium carbonate
(2L) added followed by dichloromethane (500mL) The organic layer was separated and the aqueous phase extracted with diethy l ether (3 x 300ml) The aqueous layer was brought to pH with concentrated hydrochloric acid (ice-cooling) and the solid so formed was filtered and dried in vacito to give 3-(5-pyπmιdyl)acrvlιc acid (35g) H-NMR (d^-DMSO) δ 6 82J HJ), 7 60( 1 H,d) and 9 I 5(3H. 2xs)
3-(5-Pyπmιdyl)acrv lιc acid ( lOOg) was added to a mixture of dry methanol (2L) and 4M hydrogen chloride in dioxan (445ml) and allowed to stir at 60°C under argon for 18h The mixture was cooled, and the solvent removed under reduced pressure The residue was partitioned between dichloromethane (500ml) and was washed with saturated sodium bicarbonate (300ml) The aqueous laver was extracted with dichloromethane and the combined dichloromethane layers were dried over magnesium sulfate and evaporated in vacuo to give methyl 3-(5-pyπmιdιnyl)acrylate (85g) H-NMR (d6-DMSO) δ 3 82(3H,s), 7 03( l H,d), 7 75( lH.d) and 9 28(3H,s)
Intermediate B6 - Methyl 3-(5-pyrimidyI)propionate
To a solution of methyl 3-(5-pyπmιdyl)acrylate (85g) in glacial acetic acid was added 10% palladium on charcoal ( 1 1 3g) and ammonium formate (74 2g) under argon The mixture was heated at 1 10°C for 20mιn, cooled and the solvent removed in
The resulting oil was dissolv ed in dichloromethane ( 1 5L) and washed with saturated sodium bicarbonate (750ml) The aqueous lay er was extracted with further dichloromethane (200ml). the organic layers were combined and dried over magnesium sulfate Removal of the solvent under reduced pressure gave an oil This was distilled under reduced pressure to give methyl 3-(5-pyπmιdyl)propιonate (26g) ' H-NMR (CDCI ) δ 2 69(2H.t). 2 97(2H,t),
3 70(3H,s). 8 65(2H,s) and 9 12( l H,s)
Intermediate B7 - Methyl 2-(5-pyπmidyl)meth\l)-3-rnetho\yacr\late
A mixture of methvl 3 (5-pyπmιdyl)propιonate ( 13 l g) and methv l formate (7 1 ml) dissolved in dry dimethoxyethane (20ml) was added portionwise to a suspension of sodium hvdπde (60% 4 Og) in DME ( 10ml ) under argon Reaction initiated rapidly and was stirred for a further 2h diluted with dry diethy l ether (50ml ) and filtered The solid so separated was washed w ith further diethyl ether (50ml) and was dried in \ acito to giv e a solid that was dissolved in dry dimethyl formamide (50ml) and potassium carbonate ( 1 1 3g) added under argon A solution of dimethyl sulfate ( 7 0ml) was then added ov er 1 hour The mixture was
stirred for 18h and the solvent removed in The residue was partitioned between ethyl acetate (200ml) and water ( 100ml) The aqueous layer was re-extracted with ethyl acetate (2 x 100ml ) and the combined ethyl acetate layers washed with brine (50ml) and dried over sodium sulfate The solvent was removed in \acuo to give methyl 2((5- pyπmιdvl)methyl)-3-methoxyacryiate (9 l g)
Intermediate B8 - 2-(5-Pyrimidyl)methyI)-3-metho\yacr\ lie acid
To methyl 2-((5-pyπmιdyl)methyl)-3-methoxyacrylate (9 Og) was added, with stirring, a solution of sodium hydroxide (3 5g) in water (43ml) at RT under argon After 20h, the pH of the solution was brought to 3 5 with concentrated hydrochloric acid Sonication of the oil so formed gave 2-((5-pyrimidyl)methyl)-3-methoxyacryhc acid (5 4g) as a pale yellow solid
Intermediate B9 - 5-(Pyrimid-5-yImeth\I)-2-thiouraciI
Sodium (0 25 g) was dissolved in ethanol (5 ml), thiourea (0 77 g) added, and the mixture stirred under reflux for 1 hour A solution of ethyl 2-formyl-3-(5-pyπmιdyl)propιonate (1 99 g) in ethanol (5 ml) was added slowly, and reflux continued for 18 hours The solvent was evaporated, and the residue taken up in water and washed with dichloromethane The aqueous solution was acidified to pH 5, and the precipitate filtered off. washed with water and dried to obtain 5-(pyπmιd-5-ylmethyl)-2-thιouracιl (0 71 g)
] H-NMR (d6-DMSO) δ
3 58 (2H.s), 7 54 ( lH.s), 8 70 (2H,s) and 9 02 ( 1 H.s) MPt 265-6°C
Intermediate B10 - 2-(4-Fluorobenzylthio)-5-((pyrimid-5-yl)methyl)pyrimidin-4-one
A mixture of intermediate B9 (0 90g), 4-fluorobenzy l bromide (0 51 ml), potassium carbonate ( 1 41 g) and dry DMF ( 1 1 ml) was stirred at 70CC under argon for 5h The DMF was evaporated water added, and the mixture acidified to pH 4 with acetic acid The precipitate was filtered off and purified by chromatography ( silica, 15-20% methanol in ethyl acetate) to give the product as an off-w hite solid (0 13g) Η-NMR (d -DMSO) δ 3 65 (2H,s), 4 38 (2H,s), 7 05-7 2 (2H.m), 7 35-7 5 (2H,m), 7 94 ( 1 H.bs). 8 71 (2H,s), 9 06 ( l H.s). MS (APCI-) found (M- l ) = 327, C ι 6H ] 3FN4OS requires 328
Intermediate Bll - l-Etho\ycarbonvlmethyl-2-(4-πuorobenzyl)thio-5-(pyrimid-5- ylmethyl)pyrimidin-4-one
A mixture of intermediate B I O ( lOg), ethyl bromoacetate (3 38ml), diisopropylethylamine (5 84ml) and dichloromethane (50ml) was stirred overnight, then the solution was washed sequentially with aqueous ammonium chloride and aqueous sodium bicarbonate Chromatography (silica, 5- 10% methanol in ethyl acetate) and tπturation w ith ether gave the desired product (7 02g) ' H-NMR (CDC1 ) δ 1 26 (3H, t), 3 71 (2H, s ). 4 26 (2H. q), 4 46 (2H. s), 4 4S (2H. s), 6 91 ( 1 H, s), 6 98 (2H, m), 7 35 (2H, m), 8 70 (2H, s). 9 09 ( 1H. s), MS(APCI+) M+ 1 =415, C20H 19FN4O3S requires 414 MPt 145 1 °C
Intermediate B12 - l-Carboxymethyl-2-(4-fluorobenzyl)thio-5-(pyrimid-5- ylmethyl)pyrimidin-4-one
0 5M aqueous sodium hydroxide (33 8ml) was added slowly to a solution of Intermediate B l 1 (7 Olg) in dioxan ( 150ml) The mixture was stired for 2 5h at room temperature, then the dioxan was evaporated, water added, and the mixture acidified with aqueous sodium bisulfate The precipitate was filtered off, washed with water and dried to obtain the desired product (6 31g) ! H-NMR (d6-DMSO) δ 3 59 (2H, s), 441 (2H, s), 4 67 (2H, s), 7 1 1 (2H, m), 7 45 (2H, ), 7 72 ( 1H, s). 8 70 (2H, s), 9 03 ( 1H, s). 13 55 ( 1H. bs). MS (APCI-) M- 1=385, C 1 8H 15FN4θ3S requires 386 MPt 206-207°C
Intermediate B13 - l-(3-Ethoxycarbonylprop-l-y.)-5-(pyrimid-5-ylmethyl)-2- thiouracil
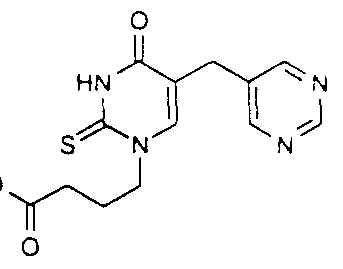
Oxalyl chloride ( 10 5ml) was added slowly to a slurry of Intermediate B8 ( 13 2g) in dichloromethane ( 120ml), and the mixture stirred for a further 3h The solv ent was evaporated, toluene added and re-evaporated to remove v olatile impurities To the crude acid chloride ( 16 9g, as the hydrochloride salt) in dry acetonitπle w as added oven-dried, powdered potassium thiocyanate ( 9 5g), and the mixture was stirred at room temperature overnight Ev aporation of the sovent gave crude acyl isothiocv anate, of w hich a 10% poπion was slurried in dry DMF (22ml) and treated with ethyl 4-amιnobuty rate ( 1 14g) and tπethylamine ( 1 2ml) The mixture was stirred overnight under argon to form the intermediate thiourea, which was then cychsed by addition of sodium ethoxide (4ml, 3M solution in ethanol) and heating to 80°C for 2h The solvent was ev aporated water added, and acidified to pH 4 with acetic acid The resulting precipitate was filtered off, washed with ether and dried to abotain the desired product (0 79g) H-NMR (d^-DMSO) δ
1.17( l H,t). 1.98(2H.m). 2J4(2H.t), 3J9(2H,s), 4.04(2H,q). 4. l 6(2H.t), 7.90( l H.s). 8.75(2H.s) and 9.03( 1 H.s); (ES+) Found (M+ l )=335. C ι
5H ι gN
4O
3S requires 334.
Intermediate B14 - l-(3-Ethoxycarbonylprop-l-yl)-2-(4-fluorobenzyl)thio-5- (pyrimid-5-ylmethyl)pyrimidin-4-one
A mixture of Intermediate B 13 (9.65g), 4-fluorobenzyl bromide (3.61 ml). diisopropyl- ethylamine (6ml) and dry dichloromethane ( 150ml) was stirred under argon for 2 days at room temperature, then washed with aqueous ammonium chloride, dried and applied directly to a silica column. Elution with 5-20% methanol in ethyl acetate gave the desired product as an orange oil (9.08g). ' H-NMR (CDC13) δ 1.26 (3H, t), 2.03 (2H, m), 2.35 (2H, t), 3.70 (2H. s), 3.85 ( 2H, t), 4.12 (2H, m). 4.47 (2H. s), 6.71 -7.07 (3H. m). 7.35-7.41 (2H. m), 8.70 (2H, s), 9.10 ( 1H. s); MS (APCI+) M+ l =443, C22H23FN4O3S requires 442 (orange oil).
Intermediate B15 - l-(3-Carboxyprop-l-yI)-2-(4-fluorobenzyl)thio-5-(pyrimid-5- ylmethyl)pyrimidin-4-one
Prepared from Intermediate B 14 by the method of Intermediate B 12. ' H-NMR (d^-DMSO) δ 1.93 (2H. m), 2.29 (2H, t), 3.58 (2H, s), 3.87 (2H, t), 4.40 (2H, s), 7.14 (2H, m). 7.48 (2H, m), 7.81 ( 1 H, s), 8.70 (2H, s), 9.02 ( 1H, s); MS (APCI-) M- l =413, C20H .9FN4O3S requires 414. MPt 188- 190°C (colourless solid).
Intermediate B16 - Ethyl 3-(2-methoxypyrimidin-5-yl)acrylate
A mixture of 2-methoxy-5-bromopyrimidine (75.43 g. 0.399 mol), ethyl aery late (47.5 ml. 0J39 mol ). palladium (II) acetate ( 1.07 g, 0.0048 mol), tri-o-tolylphosphine (2.92 g. 0.0096 mol) and triethylamine (84 ml ) were heated at 135°C with stirring under argon for 12 h.
After allo ing to cool the solid mass was dissolved in water and ethyl acetate, filtered, and the aqueous phase separated and further extracted with ethyl acetate. The combined extracts ere washed with saturated aqueous ammonium chloride, dried (MgSO4) and evaporated. The solid thus obtained was triturated with ether/light petrol ( 1 :3, 350 ml), filtered, w ashed and dried, yield 52.41 g (63%). ] H-NMR (CDCI3) δ 1.33 (3H, t). 4.06
(3H. s), 4.28 (2H, q), 6.45 ( 1H, d). 7.58 ( 1H, d), 8.67 (2H, s); MS (APCI+) found (M+H) = 209; C
10H
1 2N2θ3 requires 208.
Intermediate B 17 - Ethyl
πmidin-5-yl)propanoate
A suspension of ethyl 3-(2-methoxypyπmιdιn-5-yl)acrylate (52 4 g, 0 252 mol) in ethanol (400 ml) and tπethy lamine (50 ml) was treated with 10% palladium on carbon (3 g) and hydrogenated at 50 psi for 1 75 h The catalyst was filtered off through hyflo and the filtrate evaporated The residue was dissolved in dichloromethane washed twice with saturated aqueous ammonium chloπde, dried (MgSU4) and evaporated to an oil. yield 41 2 g (78%) ! H-NMR (CDC13) δ 1 23 (3H, t). 2 61 (2H. t), 2 87 (2H, t). 3 99 (3H. s), 4 13 (2H. q), 8 39 (2H, s). MS (APCI+) found (M+H) = 21 1 . C 10H 14N2O3 requires 210
Intermediate B18 - 2-(Metho\ymethylene)-3-(2-metho\ypyrimidin-5-yI)propιonic acid, mixed methyl/ethyl esters
To a stirring suspension of sodium hydride (0 83 g of a 60% dispersion in oil ) in anhydrous 1 , 2-dιmethoxyethane (6 ml) was added dropwise a solution of methyl formate ( 1 54 ml) and ethyl 3-(2-methoxypyπmιd-5-yl)propιonate (3 5 g) in anhydrous 1 ,2-dιmethoxyethane
(6 ml) at such a rate as to maintain the reaction temperature at 25-30°C After 1 h, ether was added and the precipitated oil allowed to settle The solution was decanted off and replaced with fresh ether, and the oil slowly solidified The solid 2-(hydroxymethylene) derivative was filtered, washed and dried, yield 3 8 g A 1 33 g portion was suspended in dimethyl formamide ( 10 ml) together with anhydrous potassium carbonate ( 1 15 g), and a solution of dimethyl sulphate (0 48 ml) in dimethvlformamide ( 10 ml) was added dropwise with stirring over 30 min After 16 h the solvent was evaporated and the residue treated with water and extracted w ith ethyl acetate The extracts were washed with water, dried (MgSO4) and evaporated to give the product as an oil y ield 0 91 g Η-NMR (CDCI3) δ 1 23 (3H. t), 3 46 (2H, s). 3 69 (3H, s. methvl ester), 3 88 (3H, s), 3 97 (3H, s), 4 16 (2H, q),
7 39 ( 1 H. s), 8 40 (2H, s) 3 2 ratio of methyl ethyl esters MS ( APCI+) found (M+ 1 ) = 253, 239 (ethyl and methv l esters) C 1 H 16N2O4 requires 252 C ] 1 H 14N2O4 requires 238
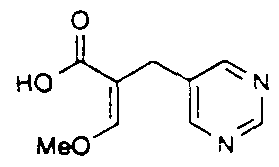
Intermediate B19 - 2-(Metho\ymethyIene)-3-(2-metho\ypyπmidin-5-yl)propιonic acid
A suspension of the mixed esters of Intermediate B 18 (0 9 g) in 2M aqueous sodium hydroxide (3 6 ml) was stirred at ambient temperature for 16 h to give a clear solution This was diluted with water, extracted with dichloromethane and evaporated to about half volume, then acidified to pH 3-4 (2M hydrochloric acid) when the product crystallised out The white solid was filtered, washed with ice-cold water and dried v ιeld 0 46 g H-NMR
(CDCI3) δ 3 43 (2H, s) 3 91 (3H, s), 3 99 (3H. s,), 7 49 ( 1 H. s). 8 42 (2H. s), MS (APCI+)
found (M+ l ) = 225. C 1oH 1 2N2θ4 requires 224.
Intermediate B20 - 5-(2-Methoxypyrimidin-5-y methyi)-2-thiouracil
To an ice cooled solution of potassium t-butoxide (7.83 g, 0.07 mol) in anhydrous THF (60 ml) was added dropwise with stirring under argon over 1 hour to a solution of ethyl 3-(2- methoxypyrimidin-5-yl) propionate (5.87 g. 0.028 mol) and methyl formate (3.6 ml, 0.059 mol) in anhydrous ether (70 ml). After stirring for 16 h, the solvents were evaporated, thiourea (4.25 g, 0.056 mol) and propan-2-ol (80 ml) added and the mixture refluxed for 5 h. The solvent was evaporated and the residue dissolved in water, extracted twice with ether and acidified to pH 4.5 with acetic acid. The solid which precipitated was filtered, washed well with water and dried, yield 5.57 g (80%). ] H-NMR (d6-DMSO) δ 3.47 (2H, s). 3.85 (3H, s), 7J3 ( 1 H, s). 8.48 (2H. s). 12.25 ( 1 H. br s), 12.46 ( 1 H. br s): MS (APCI+) found (M+H) = 25 1 ; C 10H 10N4O2S requires 250.
Intermediate B21 - 2-(4-Fluorobenzylthio)-5-((2-methoxypyrirnid-5- yl)methyl)pyrimidin-4-one
Prepared from Intermediate B20 by the method of Intermediate B 10. ' H-NMR (dg DMSO) δ 3.55 (2H,s), 3.85 (3H,s), 436 (2H,s), 7.0-7.2 (2H,m), 7.35-7.5 (2H,m), 7.86 ( I H.bs), 8.48 (2H,s) 12.81 ( l H.b): MS (APCI+) found (M+ l ) = 359: C 17H 15FN4O2S requires 358.
Intermediate B22 - l-(Ethoxycarbonylmethyl)-2-(4-fiuorobenzyl)thio-5-(2- methoxypyrimid-5-ylrnethyI)pyrimidin-4-one
Prepared from Intermediate B21 by the method of Intermediate B l l . H-NMR (CDC13) δ 1.25 (3H. t), 3.65 (2H. s), 3.97 (3H, s), 4.23 (2H, q), 4.47 (2H. s), 4.51 (2H, s), 6.94-7.01 (3H, m). 7.35 (2H. m). 8.42 (2H. s); MS (APCI+) found (M+ l ) = 445: C21 H21 FN4O4S requires 444.
Intermediate B23 — l-Carboxymethyl-2-(4-Fluorobenzylthio)-5-(2-rnethoxypyrimid- 5-ylmethyl)pyrimidin-4-one
Prepared from Intermediate B22 by the method of Intermediate B 12 ' H-NMR (d6-DMSO) δ 3.51 (2H, s), 3 88(3H, s). 4 41 (2H, s), 4 63 (2H, s), 7 12 (2H, m). 7 45 (2H. m), 7 62 ( 1 H. s) 8.48 (2H, s), MS (APCI+) found (M+ l ) = 417, C 19H 17FN4O4S requires 416
Intermediate B24 - l-(3-EthoxycarbonyIprop-l-yl)-5-(2-methoxypyrirnid-5- ylmethyl)-2-thiouracil
Prepared from Intermediate B 19 by the method of Intermediate B 13, except using sodium methoxide in place of ethoxide Some ether exchange took place during the reaction, and the title compound was obtained mixed with ca 30% of the corresponding 2- ethoxypynmidine ! H-NMR (DMSO-d6) δ 1 17 (3H, t), 1 31 (t, ethoxy). 1 97 (2H, t), 2 36
(2H, t), 3 49 (s, ethoxy), 3 51 (s. methoxy), 3 88 (s. methoxy) 4 04 (2H. q ). 4 16 (2H, t), 4 31 (q, ethoxy), 7 81 ( 1 H. s), 8 48 (s. ethoxy), 8 50 (s, methoxy). 12 50 (bs. NH) (light brown solid)
Intermediate B25 - l-(3-Ethoxycarbonylprop-l-yl)-2-(4-fluorobenzyl)thio-5-(2- methoxypyrimid-5-ylmethyl)py rimidin-4-oπe
Prepared from Intermediate B24 by the method of Intermediate B 14 Η-NMR (CDCI3) δ 1.25 (3H, t), 2 03 (2H. ), 2.33 (2H, t), 3.64 (2H, s), 3 83 (2H, m). 3 99 (3H, s), 4 12 (2H. q). 4 47 (2H, s). 6 98 (3H, m), 7 37 (2H, m), 8 46 (2H. s), MS (APCI+) M+ l =473, C23H25FN4O4S requires 472 (waxy cream solid)
Intermediate B26 - l-(3-Carboxyprop-l-yl)-2-(4-fluorobenzyl)thio-5-(2-methoxy- pyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from Intermediate B25 by the method of Intermediate B 12 Η-NMR ( DMSO-d£) δ 1 89 (2H, m), 2 28 (2H, t), 3 50 (2H, s), 3 86 (5H. ), 4 40 (2H. sm), 7 13 (2H. m), 7 47
(2H, m).7 72 ( 1 H, s), 8 50 (2H. s ) MS (APCI+) M+ 1 =445. C21 H21 FN4O4S requires 444 (colourless solid)
Intermediate B27 - 3-(l-Methylpyrazol-4-yl)acrylic acid
A mixture of i -methylpyrazole-4-carboxaldehyde (J Chem Soc 1957. 3314) ( 18 1 g), malonic acid ( 17 1 g), pyndine ( 15 ml) and piperidine (0 2 ml ) was heated to 100°C for 1 hour After cooling, water was added, followed by aqueous ammonia to obtain a clear solution, which was acidified to pH5 with hydrochloric acid The resulting solid was filtered off, washed with water and dried to obtain 3-( l-methylpyrazoI-4-yl)acryhc acid ( 18 9 g) ' H-NMR (d
6-DMSO) δ 3 83 (3H,s), 6 18 ( lH.d). 7 44 ( l H.d). 7 83 ( l H.s), 8 07 ( l H,s) (APCI) found (M+H)=153 C7H8N2O2 requires 152
Intermediate B28 - Methy l 3-(l-methylpyrazol-4-yI)acrylate o
-N 3-( l-Methylpyrazol-4-yl)acryhc acid ( 18 86 g) was added to a solution of sulphuric acid ( 15 ml) in methanol ( 150 ml), and the mixture refluxed for 2 hours, cooled, and poured onto ice The acid was neutralised with solid sodium carbonate and the product extracted into dichloromethane, which was dried and evaporated Crystallisation from ether/petrol gave methyl 3-( 1 -methylpyrazol-4-yl)acrvlate ( 16 0 g) ' H-NMR (d6-DMSO) δ 3 77 (3H.s), 3 91 (3H.s), 6 16 ( l H,d), 7 54 ( lH.s), 7 56 (lH.d), 7 69 ( lH,s) (APCI) found (M+H)=167
CgH ιoN2θ2 requires 166
Intermediate B29 - Methyl 3-(l-methyIpyrazol-4-yi)propionate
Prepared from Intermediate B28 by the method of Intermediate B2, except that the reaction was run at 50°C ] H-NMR (d6-DMSO) δ 2 56 (2H,t), 2 79 (2H,t), 3 67 (3H,s). 3 85 (3H,s),
7 17 (l H,s), 7 31 (3H.s) (APCI) M+H=169 CgHi2 2θ2 requires 168
Intermediate B30 - Methyl 2-formyl-3-(l-meth\ lpy razol-4-yl)propιonate. sodium salt
Sodium hydride (2 62 g, 60% in oil) was washed with petrol and suspended in drv dimethoxyethane (20 ml ) Methyl 3-( l -methylpyrazol-4-yl)propιonate (8 8 g) and methyl formate (4 87 ml) were dissolved in DME (20 ml), and a few drops of the mixture added to the sodium hydride suspension which was warmed briefly to initiate the reaction before continuing dropwise addition at a rate which sustained controlled ev olution of hvdrogen The mixture was stirred for a further 16 hours at room temperature then diluted w ith ether The solid was filtered off washed with ether and immediately dried, and w as used promptly without further purification
Intermediate B31 - 5-((l-MethylpyrazoI-4-yl)methyl)-2-thiouracil
Prepared from Intermediate B30 analogously to Intermediate B9, except using the preformed sodium salt instead of adding sodium ethoxide. H-NMR (d0-DMSO) δ 3.33 (3H.s).3.75 (3H,s),7J5 (lH.s).7.23 (lH.s).7J6 (lH.s).12.2 (lH.br s), 12J(lH.brs). (APCI) M+H=223. C9H10N4OS requires 222.
Intermediate B32 - 2-(4-Fluorobenzylthio)-5-((l-methylpyrazol-4- yl)methyI)pyrimidin-4-one
Prepared from Intermediate B31 by the method of Intermediate BIO. H-NMR (d6-DMSO) δ 3.47 (2H. s).3.81 (3H, s).4J1 (2H. s), 7J9 (2H. s), 7.29 (1H, s), 7.48 (3H, m), 7.84 (1H, s).12.74 (1H. br.s); MS (APCI+) found (M+l) = 331; Cι6Hι5FN4OS requires 330.
Intermediate B33 - lJEthoxycarbonylmethyl)-2-(4-fiuorobenzylthio)-5-((l-methyI- pyrazol-4-yl)methyl)pyrimidin-4-one
Prepared from Intermediate B32 by the method of Intermediate Bl 1. Η-NMR (CDCI3) δ
1.22 (3H, s), 3.60 (2H. s), 3.87 (3H, s), 4.23 (2H, q), 4.42 (2H. s), 4.50 (2H, s), 6.77 (1H, s), 6.96 (2H. m), 7.25-7.39 (4H. m): MS (APCI+) found (M+l) = 417; C20H21FN4O3S requires 416.
Intermediate B34 - l-(carboxymethyl)-2-(4-fluorobenzylthio)-5-((l-methylpyrazol-4- yi)methyI)pyrimidin-4-one
Prepared from Intermediate B33 by the method of Intermediate B 1 . ' H-NMR (d6-DMSO) δ 3.36 (2H. s).3.76 (3H. s), 4.41 (2H. s).4.67 (2H. s), 7.14 (2H, m).7.23 ( 1H, s).7.43-7.49 (4H. m): MS (APCI+) found (M+l) = 389: CigH|7FN4O3S requires 388.
Intermediate B35 - Ethyl 2-(methoxymethylene)-3-(l-methyIpyrazoI-4-yl)propionate
Prepared from Intermediate B30 by the method of Intermediate B7. ' HNMR (CDCI3) inter
alia 3.37 (2H. s ). 3.70 (3H, s). 3.81 (3H. s), 3.85 (3H, s), 7.17 ( 1 H. s). 7.30 ( 1 H. s) and 7.31 ( 1 H, s). MS( APCI+) MH+ 21 1 ; C 10H j 4N2O3 requires M+ 210.
Intermediate B36 - 2-(Methoxymethylene)-3-(l-rnethylpyrazoi-4-yI)propionic acid
Prepared from Intermediate B35 by the method of Intermediate B8. ' HNMR (CDCI3) 3.36 (2H. s), 3.82 (3H. s). 3.88 (3H, s), 7.18 ( 1H. s). 7.33 ( 1H, s) and 7.40 OH. s). MS(APCI+) MH+ 197; C9H 12N2O3 requires M+ 196.
Intermediate B37 - l-(3-Ethoxycarbonylprop-l-yl)-5-(l-methylpyrazol-4-ylmethyI)-2- thiouracil
Prepared from Intermediate B36 by the method of Intermediate B 13. ' HNMR (CDCI3) 1.27 (3H,t,J7.1Hz). 2.07 (2H,m), 238 (2H,t,76.9Hz), 3.52 (2H,s), 3.88 (3H.s), 4.15 (4H,m), 7.05 (lH.s), 7.30 ( lH.s) and 7.35 ( lH.s). MS(APCI+) MH+ 337; C15H20N4O3S requires M+ 336.
15 Intermediate B38 - l-(3-Ethoxycarbonylprop-l-yl)-2-(4-fluorobenzy )thio-5-(l- methyIpyrazoI-4-ylmethyl)pyrimidin-4-one
Prepared from Intermediate B37 by the method of Intermediate B 14. ' HNMR (CDCI3) 1.24 (3H. /=7.2Hz), 2.00 (2H,m). 2.32 (2H.t.6.8Hz). 3.58 (2H.s), 3.79 (2H.tJ=7.4Hz), 20 3.87 (3H,s), 4.12 (2H.q,7.2Hz), 4.49 (2H.s), 6.89 ( lH.s). 7.00 (2H.m) and 7.37 (4H,m).
MS(APCI+) MH+ 445; C22H25FN4O3S requires M+ 444.
Intermediate B39 - l-(3-Carboxyprop-l-yl)-2-(4-fluorobenzyi)thio-5-( l-methyl- pyrazol-4-ylmethyl)pyrimidin-4-oπe
25 Prepared from Intermediate B38 by the method of Intermediate B 12. ' HNMR (CDCI3)
2.02 (2H,m). 2.35 (2H.t 6.5Hz), 3.57 (2H.s), 3.84 and 3.85 (5H.m and s). 4.46 (2H.s), 6.98 (3H.m) and 7.35 (4H.m). MS(APCI+) MH+ 417, C20H21 FN4O3S requires M+ 416.
Intermediate B40 - 2-methyl-5-bromopyrimidine-4-carboxylic acid
Sodium ethoxide ( 100ml of a 21 wt% solution in ethanol) was added to a stirred suspension of acetamidine hydrochloride (25.6g), in ethanol (30ml). The mixture was warmed to 50°C, then the heating bath was removed and a solution of mucobromic acid (29 4g), in ethanol (50ml) added dropwise at a rate which maintained a constant temperature ( ca 30mιn addition), followed by a further 60ml of sodium ethoxide solution. After cooling, the mixture was filtered and the filtrate evaporated to a residue which was shaken vigorously with 2M hydrochloric acid (70ml). The brown precipitate was filtered off. washed with a small amount of ice cold water, and dried. ' H-NMR (d^-DMSO) δ 2.62 (3H,s), 9.03 (lH,s); MS(APCI+) found (M+H) = 217/219, C6H5BrN2θ2 requires 216/218.
Intermediate B41 - 2-methyl-5-bromopyrimidine
A slurry of Intermediate B40 (8.13g) in xylene ( 1 10ml) was heated to reflux for 4h. After cooling, the mixture was applied directly to a silica column, which was eluted with pentane to remove the xylene, then ether to obtain the desired product as a pale brown solid after evaporation (4.4g). ' H-NMR (CDC13) δ 2.69 (3H.s). 8.69 (2H,s), MS(APCI+) found (M+H) = 173/175, C5H5BrN2 requires 172/174.
Intermediate B42 - Ethyl 3-(2-methylpyrimidin-5-yl)acrylate
Prepared from Intermediate B41 by the method of intermediate B 16 ' H-NMR (CDCI3) δ
1.35 (3H,t). 2 76 (3H.s). 4.29(2H,q). 6 54 ( l H.d,J= 16Hz), 7.59 ( I H.d.J= ! 6Hz). 8 78 (2H.s ). MS(APCI+) found (M+H) = 195. C ι0H ] 4N2θ2 requires 194
Intermediate B43 - Ethyl 3-(2-methylpyrimidin-5-yl)propanoate
Prepared from Intermediate B42 by the method of lntermediate B 17 ' H-NMR (CDCI3) δ
1.24 (3H,t), 2.63 (2H. . 2.71 (3H.s). 2 91 (2H.t), 4.13 (2H,q), 8 5 1 (2H.s ). MS(APCI+) found (M+H) = 195, C ι oH ] 4Nτ2θ2 requires 194
Intermediate B44 - 5-(2-Methylpyrimidin-5-ylmethyI)-2-thiouracil
Prepared from Intermediate B43 by the method of Intermediate B20 ' H-NMR (DMSO) δ
2.57 (3H.s).3.53 (2H.s), 7.50 (lH.d).8.58 (2H.s), 12.3 (lH.br s).
Intermediate B45 - 2-(4-FIuorobenzylthio)-5-((2-methylpyrimid-5- yl)methyl)pyrimidin-4-one
Prepared from Intermediate B44 by the method of Intermediate BIO. ' H-NMR (DMSO) δ 2.56 (3H.s).3.59 (2H.s), 4.37 (2H.s).7.13 (2H.m).7.44 (2H,m), 7.90 (lH.br s), 8.58 (2H.s), 12.9 (lH.br s); MS(APCI-) found (M-H) = 341. C17H15FN OS requires 342.
Intermediate B46 - 2-Cyclopropyl-5-bromopyrimidine-4-carboxylic acid
Prepared from cyclopropylcarbamidine hydrochloride by the method of Intermediate B40.
!H-NMR (DMSO) δ 8.72 (1H, s), 2.00 (1H, m), 0.78-0.93 (4H. m): MS(APCI+) found (M- 41) = 201, CgH7N2O279Br requires 242.
Intermediate B47 - 2-Cyclopropyl-5-bromopyrimidine
Prepared from Intermediate B46 by the method of Intermediate B41. ' H-NMR (CDCI3) δ
8.38 (2H. s), 1.99 (1H, m), 0.87 (4H. ); MS(APCI+) found (M+l) = 199. C7H7N2 79Br requires 198.
Intermediate B48 - Ethyl 3-(2-cyclopropylpyrimidin-5-yl)acrylate
Prepared from Intermediate B47 by the method of Intermediate B 16. ' H-NMR (CDCI3) δ
8.62 (2H. s), 7.49 (1H. d).6.42 (1H. d), 4.22 (2H. q).2.21 (1H, m), 1.27 (3H. t).1.04-1 JO (4H. m); MS(APCI+) found (M+l) = 219, C12H14 2O2 requires 218.
-cvclopropylpvrimidin-5-
Prepared from Intermediate B48 by the method of Intermediate B17. Η-NMR (CDCI3) δ
8.43 (2H. s).4.12 (2H. q).2.S7 (2H, t), 2.62 (2H, t).2.21 (1H, m).1.23 (3H. t).1.02-1.26
(4H. m): MS(APCI+) found (M+ l ) = 221. C I 2H ι6N2θ2 requires 220.
Intermediate B50 - 5-(2-CycIopropylpyrimidin-5-ylmethyl)-2-thiouracil
Prepared from Intermediate B49 by the method of Intermediate B20. ' H-NMR (DMSO) δ 8.73 (2H, s), 7.69 ( I H, s), 3.72 (2H. s), 238 ( IH. m), 1.13- 1 .26 (4H. m): MS(APCI+) found (M+ 1 ) = 261 , C j 2H - 2N4OS requires 260.
Intermediate B51 - 2-(4-FIuorobenzylthio)-5-((2-cyclopropylpyrimid-5- yl)methyl)pyrimidin-4-one
Prepared from Intermediate B50 by the method of Intermediate B 10. Η-NMR (CDCI3) δ
1 1.48 ( IH. s), 8.50 (2H, s), 7.73 ( IH, s), 7.35 (2H, m), 6.98 (2H. m), 4.38 (2H. s), 3.63 (2H, s), 2.21 ( I H. m), 1.10 (4H, m).
Intermediate B52 - 2-(lJ-DimethylethyI)-5-bromopyrimidine-4-carboxylic acid
Prepared from tert-butylcarbamidine hydrochloride by the method of Intermediate B40. Light brown precipitate. Η-NMR (CDCI3) δ 1 .34 (9H. s), 9.09 ( I H. s); MS (APCI-) found (M- l ) =257/259; C9H 1 ] BrN2O2 requires 258/260.
Intermediate B53 - 2-(lJ-Dimethylethyl)-5-bromopyrimidine
Prepared from Intermediate B52 by the method of Intermediate B41. Light brown solid. H-NMR (CDCI3) δ 1.40 (9H. s). 8.71 (2H. s); MS (APCI+) found (M+ l ) =215; CgH 1
j N Br requires 215/217.
Intermediate B54 - Ethyl 3-(2-( lJ-dimethylethyl)pyrimidin-5-yl)acrylate
Prepared from Intermediate B53 by the method of Intermediate B 16. Brown oil. H-NMR
(CDCI3) δ 1 .30- 1 .47 ( 12H. m). 4.23-437 (2H, q). 6.47-6.58 ( 1 H. 2.xs ). 7.54-7.65 ( 1 H. 2xs),
8 80 (2H. s), ), MS (APCI+) found (M+ l ) = 235, C i3H i N2O2 requires 234
-(lJ-dimethylethyI)pyrimidin-5-
Prepared from Intermediate B54 by the method of Intermediate B 17 Orange oil H-NMR (CDC1 ) δ 1 19- 1 30 (3H, t), 1 40 (9H. s), 2 58-2 68 (2H. t), 2 85-2 97 (2H. t), 4 07-4 20 (2H, q), 8 55 (2H, s). ), MS (APCI+) found (M+ l ) = 237, C 1 H2o 2θ2 requires 236
Intermediate B56 - 5-(2-(lJ-Dimethylethyl)pyrimidin-5-ylmethyI)-2-thiouracil
Prepared from Intermediate B55 by the method of Intermediate B20 Brown solid H- NMR (DMSO) δ 1 33 (9H. s), 3 54 (2H. s), 7 52 ( IH, s), 8 62 (2H, s), 12 27- 12 37 ( IH, br s), 12 52 ( I H, s), ), MS (APCI-) found (M- l ) = 275. C I 3H ] 6N4OS requires 276
Intermediate B57 - 2-(4-FIuorobenzylthio)-5-((2-(lJ-dimethylethyl)pyrimid-5- yl)methyl)pyrimidin-4-one
Prepared from Intermediate B56 by the method of Intermediate B 10 Light brown solid
J-I-NMR (CDCI3) δ 1 38 (9H, s), 3 68 (2H. s), 4 37 (2H, s), 6 93-7 05 (2H, t). 7 30-7 42 (2H, ). 7 78 ( IH, s), 8 62 (2H, s). MS (APCI+) found (M+l ) = 385 C20H21N4OSF requires 384
Intermediate B58 — Ethyl 2-trifluoromethyl-4-oxopyrimidine-5-carboxylate
To a solution of tnfluoroacetamidine ( 36 5 g) and diethv 1 ethoxymalonate ( 71 g) in ethanol (300 ml) was added a solution of sodium ethoxide in ethanol (21 wt %. 109 5 ml ) over 5 min The mixture was heated at reflux for 6 h. then cooled, concentrated and water (200 ml) added The resulting solid was collected by filtration, washed with cold water (50 ml) and diethyl ether (2 x 100 ml) and then suspended in water (400 ml) Dichloromethane
(300 ml) was added and the mixture acidified with dilute HCl (2 5 M. 125 ml) The organic extract was washed with water, dried (MgSO4) and ev aporated to giv e the title compound as a buff solid (53 g, 68%) • H-NMR (CDCI3) δ 12 00 ( I H, br «,), 9 18 ( I H, s), 4 57 (2H, q), 1 49 (3H, t) C-NMR (CDCI3) δ 171 4 167 3, 160 4 160 2 (q. J = 38 Hz). 1 18 8 (q. 7
= 276 Hz), 109.2. 63.8. 14 0. MS (APCI+) found (M+ l ) = 237, C8H7F3N2O3 requires 236
Intermediate B59 — Ethyl 2-trifluoromethyI-4-chIoropyrimidine-5-carboxylate
To a solution of ethyl 4-hydroxy -2-tnfluoromethylpyrιmιdιne-5-carboxylate (51 8 g) in dichloromethane (600 ml) cooled in an ice bath was added oxalyl chloride (57 4 ml) followed by dimethvlformamide (0.2 ml). The mixture was stirred at room temperature for 16 h and then evaporated Toluene was added and evaporated. The residue was dissolved in dichloromethane, washed with water, dπed (MgSO4) and evaporated to give the title compound as an orange oil (55 7 g, 100%) ! H-NMR (CDCI3) δ 9 25 ( I H. s). 4 51 (2H. qj. 1.46 (3H, t); 1 3C-NMR (CDCI3) δ 162.0 (2C), 161.1. 158.1 (q, 7 = 39 Hz), 127.0. 1 18.9 (q.
J = 276 Hz), 63.5, 14 4
Intermediate B60 — Ethyl 2-trifluoromethylpyrimidine-5-carboxylate
A mixture of ethyl 4-chloro-2-trιfluorornethylpyπmιdιne-5-carboxylate (55 7 g), 10% palladium on carbon (0.3 g), ethanol ( 1000 ml) and /V./V-diisopropylethylamine (90 ml) was shaken under hydrogen pressure maintained at 1 atmosphere for 2 h. The catalyst was then filtered off and the solvents evaporated. The residue was dissolved in dichloromethane, washed with ammonium chloride solution, then water, dned (MgSO4) and evaporated to give the title compound as a buff solid (48 g, 100%) ] H-NMR (CDCI3) δ 942 (2H, s), 4.51 (2H, q), 1.45 (3H, t), 1 3C-NMR (CDCI3) δ 162.7, 159 4 (2C). 159.3 (q, = 37 Hz),
126.3. 1 19.6 (q, J = 275 Hz). 62.9, 14 4
Intermediate B61 — 2-trifluoromethyl-5-formylpyrimidine
To a solution of ethyl 2-trιfluoromethylpyπmιdιne-5-carboxylate (5 17 g) in toluene ( 120 ml) cooled in dry ice/acetone was added a solution of dnsobutylaluminium hydride (25 wt
%. 31 ml) over 15 min The mixture was stirred at -78°C for 45 min. then dilute HCl (2M.
120 ml) was added cautiously After allowing the mixture to warm to room temperature diethyl ether was added The organic phase was separated, washed with water, then brine. dried (MgSO4) and evaporated to give the title compound as a colourless solid (3 46 g, 84%). ' H-NMR (CDCl3) δ 10.29 ( I H, s). 937 (2H, s), 1 3C-NMR (CDCI3) δ 187 7. 159 6
(q, = 38 Hz), 159.1 (2C) 129 6. 1 19.2 (q, J = 276 Hz)
Intermediate B62 - 3-(2-trifiuoromethylpyrimidin-5-yl)acrylic acid
A mixture of 5-formyl-2-trifluoromethylpyrimidine ( 13.15 g], malonic acid (7.77 g), pyndine (45 ml) and piperidine (0.1 ml) were heated at 1 10°C for 2 h. After cooling, the solvent was evaporated and the residue was suspended in water. Ammonium hydroxide was added to effect solution, then the mixture was acidified with cHCl. The resulting solid was collected by filtration, washed with water and dried under vacuum to give the title compound as a buff solid ( 12.7 g, 78%). ] H-NMR (DMSO-d6) δ 12.8 ( IH, br s), 9.40 (2H, s), 7.70 (2H. d), 6.71 (2H, d); I 3C-NMR (DMSO-d6) δ 167.0, 157.7 (2C), 155.1 (q, J = 36 Hz), 136.5, 131.1 , 126.0, 1 19.9 (q, J = 275 Hz): MS (APCI-) found (M- l ) = 217, C H5F3N2O2 requires 218.
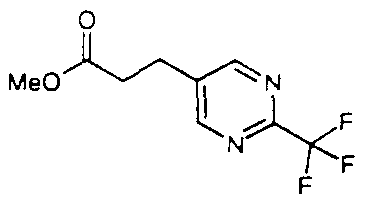
Intermediate B63 - Methyl 3-(2-trifluoromethylpyrimidin-5-yl)acrylate
A mixture of (E)-3-(2-trifluoromethylpyrimidin-5-yl)propenoic acid ( 12.7 g), methanol (80 ml) and concentrated sulphuric acid (4 ml) was heated under reflux for 4 h. After cooling, the solvent was evaporated and the residue partitioned between dichloromethane and water.
The organic phase was separated, washed with sodium bicarbonate solution, dried (MgSO4) and evaporated to give the title compound as an off-white solid ( 10.12 g, 75%). I H-NMR (CDC13) δ 9.04 (2H, s), 7.69 (2H, d), 6.71 (2H, d), 3.87 (3H. s); 13C-NMR (CDCI3) δ 165.9, 157.2 (q, 7 = 37 Hz), 156.8 (2C), 136J, 130.5, 124.7, 120.6, (q, 7 = 275 Hz). 52.6; MS (APCI+) found (M+ 1 ) = 233, C9H7F3N2O2 requires 232.
Intermediate B64 - Methyl 3-(2-trifluoromethylpyrimidin-5-yl)propanoate
Prepared from Intermediate B63 by the method of Intermediate B 17. ' H-NMR (CDCI3) δ 9.23 (2H, s). 3.78 (3H. s). 3.14 (2H. t), 2.81 (2H. t); MS (APCI+) found ( M+ l ) = 235. C9H9F3N2O2 requires 234.
Intermediate B65 - 5-(2-Trifluoromethylpyrimidin-5-yImethyl)-2-thiouraciI
Prepared from Intermediate B64 by the method of Intermediate B20. ' H-NMR ( DMSO-d6) δ 12.41 ( 1 H br s), 8.96 (2H, s), 7.58 ( 1 H. s), 3.70 (2H. s); MS (APCI-) found (M- 1 ) = 287, C I 0H7F3N4OS requires 288.
Intermediate B66 - 2-(4-Fluorobenzylthio)-5-((2-trifluoromethylpyrimid-5- yl)methyl)pyrimidin-4-one
Prepared from Intermediate B65 by the method of Intermediate B IO. ' H-NMR (CDCI3) δ 8.91 (2H. s), 7.95 ( IH. s), 7.45-740 (2H, m). 7.05 (2H, t), 4.44 (2H. s). 3.82 (2H. s); MS
(APCI+) found (M+ l ) = 397, C 17H 12F4N4OS requires 396.
Intermediate B67 - 3-(l-Ethylpyrazol-4-yl)acrylic acid
Prepared from l -ethylpyrazole-4-carboxaldehyde by the method of Intermediate B27. H- NMR (CDCl3) δ 1.5 1 (3H.t). 4.19 (2H,q), 6J6 ( l H.d). 7.61 ( l H.s), 7.66 ( l H.d), 7.73
( lH.s).
Intermediate B68 - Methyl 3-(l-ethylpyrazol-4-yl)acrylate
Prepared from Intermediate B67 by the method of Intermediate B28. ' H-NMR (CDCI3) δ 1.50 (3H,t), 3.77 (3H,s), 4.17 (2H,q). 6.16 ( lH.d), 7.56 ( lH,d), 7.57 ( lH,s), 7.69 ( l H,s).
Intermediate B69 - Methyl 3-(l-ethylpyrazol-4-yl)proρionate
Prepared from Intermediate B68 by the method of Intermediate B2. except that the reaction was run at 50°C. ' H-NMR (d6-DMSO) δ 1.47 (3H,t). 2.56 (2H.t). 2.79 (2H,t), 3.67 (3H,s), 4J 1 (2H,q), 7.21 ( l H,s), 7.32 ( 1 H.S).
Intermediate B70 - Methyl 2-formyl-3-(l-ethylpyrazol-4-yl)propionate, sodium salt
Prepared from Intermediate B69 by the method of Intermediate B30, and used immediately wthout purification.
Intermediate B71 - 5-((l-Ethylpyrazol-4-yl)methyl)-2-thiouracil
Prepared from Intermediate B70 analogously to Intermediate B9. except using the
preformed sodium salt instead of adding sodium ethoxide. ' H-NMR (dζ-DMSO) δ 1.31 (3H,t), 3.34 (2H,s), 4.04 (2H,q), 7.15 (lH.d).7.25 (lH,s), 7.50 (lH.s).12.2 (lH.br s), 12.4 (lH.brs). (APCI) M+H=237. C10H12N4OS requires 236.
Intermediate B72 - 2-(4-Fluorobenzylthio)-5-((l-ethyIpyrazoI-4-yl)methyl)pyrimidin- 4-one
Prepared from Intermediate B71 by the method of Intermediate B10. ' H-NMR (d6-DMSO) δ 1.32 (3H,t), 3.42 (2H, s), 4.04 (2H. q).4.36 (2H, s), 7.14 (2H, m), 7.25 (IH. s), 7.44 (2H, m), 7.49 (IH, s).1.1 (IH. br s), 12.8 (IH, br s); MS (APCI+) found (M+l) = 345; C17H17FN4OS requires 344.
Intermediate B73 - l-(Ethoxycarbonylmethyl)-2-(4-fluorobenzylthio)-5-((l-ethyl- pyrazoI-4-yl)methyl)pyrimidin-4-one
Prepared from Intermediate B72 by the method of Intermediate B 11. ' H-NMR (d6-DMSO) δ 1J7 (3H, t), 1.32 (3H, t), 3.37 (2H, s), 4.03 (2H, q), 4.15 (2H, q), 4.42 (2H, s), 4.78 (2H, s), 7.14 (2H, ), 7.25 (IH, s), 7.4-7.5 (4H, ); MS (APCI+) found (M+l) = 431; C21H23FN4O3S requires 430.
Intermediate B74 - l-(carboxymethyI)-2-(4-fluorobenzylthio)-5-((l-ethyIpyrazol-4- yl)methyI)pyrimidin-4-one
Prepared from Intermediate B73 by the method of Intermediate B 12. ' H-NMR (d6-DMSO) δ 1.33, (3H.t), 3.37 (2H, s), 4.05 (2H, q), 4.41 (2H. s), 4.65 (2H, s), 7.14 (2H. m).7.25 (IH, s), 744-7.50 (4H, m); MS (APCI+) found (M+l) = 403; C19H19FN4O3S requires 402.
Intermediate B75 — Ethyl 3-[2-(l-morpholino)-5-pyrimidy ]acrylate
Prepared from 2-( l-morpholino)-5-bromopyrimidine by the method of Intermediate B16. 'HNMR (CDCI3) δ 1.33 (t.3H), 3.78 (m, 4H), 3.88 (m.4H).4.25 (q.2H).6.3 (d, IH), 7.5 (d, IH), 8.50 (s, 2H). MS (APCI+) Found (M+l) = 264: C13H17N3O requires 263.
Intermediate B76 — Ethyl 3-[2-(l-morpholino)-5-pyrimidyl]propionate
Prepared from Intermediate B75 by the method of Intermediate B 17. Η NMR (CDCI3) δ 1.26 (t. 3H). 2.55 (t, 2H), 2.77 (t, 2H), 3.76 (s, 8H), 4.13 (q, 2H). 8.20 (s, 2H). MS (APCI+) Found (M+ l ) = 266; C
13H
19N
3O3 requires 265.
Intermediate B77 — 5-(2-(l-Morpholino)pyrimid-5-ylmethyl)-2-thiouracil
Prepared from Intermediate B76 by the method of Intermediate B20. Η NMR (DMSO-d ) δ 336 (s, 2H), 3.63 (s, 8H), 7.37 (s, I H), 8.28 (s, 2H), 12.25 (br s, 2H). MS (ES+) Found (M+ l ) = 306; C ] 3H i5N5O2S requires 305.
Intermediate B78 — 2-(4-FluorobenzyI)thio-5-(2-(l-morpholino)pyrimid-5- ylmethyl)pyrimidin-4-one
Prepared from Intermediate B77 by the method of Intermediate B 10. Η NMR (DMSO-dg) δ 3.44 (s, 2H), 3.63 (s, 8H), 4.37 (s, 2H), 7.13 (t, 2H), 7.43 ( , 2H), 7.80 (br s, IH), 8.30 (s,
2H), 12.75 (br s, I H). MS (ES+) Found (M+ l ) = 414; C2oH2θF 5O2S requires 413.
Intermediate B79 — Ethyl 3-(2-dimethylamino-5-pyrimidyl)acrylate
Prepared from 2-dimethylamino-5-bromopyrimidine by the method of Intermediate B 16. ' H NMR (CDCI3) δ: 1.33 (t. 3H), 3.24 (s, 6H), 4.25 (q. 2H). 6.3 (d. IH). 7.5 (d. IH), 8.49
(s, 2H). MS (APCI+) Found (M+ 1 ) = 222; C 1 H j 5N3O2 requires 221.
Intermediate B80 — Ethyl 3-(2-dimethylamino-5-pyrimidyl)propionate
Prepared from Intermediate B79 by the method of Intermediate B 17. Η NMR (CDCI3) δ: 1.24 (t, 3H), 2.53 (t, 2H), 2.75 (t, 2H), 3J6 (s, 6H), 4.13 (q, 2H), 8.27 (s. 2H). MS (APCI+)
Found (M+ l ) = 224; C j 1 H 17N3O2 requires 223.
Intermediate B81 — 5-(2-Dimethylaminopyrimid-5-ylmethyl)-2-thiouracil
Prepared from Intermediate B80 by the method of Intermediate B20. ' H NMR (DMSO-d£) δ: 3.07 (s. 6H), 334 (s, 2H). 7.33 (s, IH), 8.23 (s, 2H), 12.25 (br s. 2H). MS (ES+) Found (M+l ) = 264; C
{ ] H
1 3N5OS requires 263.
Intermediate B82 — 2-(4-FluorobenzyI)thio-5-(2-dimethylaminopyrimid-5- ylmethyl)pyrimidin-4-one
Prepared from Intermediate B81 by the method of Intermediate B IO. Η NMR (CDCI3) δ: 3.15 (s. 6H). 3.51 (s, 2H). 4.36 (s, 2H), 7.0 (m, 2H), 7.35 (m, 2H), 7.66 (br s, IH). 8.23 (s,
2H). MS (ES+) Found (M+ l ) = 372; C 1 8H1 8FN5OS requires 371.
Intermediate B83 — 2-(Bis(2-methoxyethyl)amino)-5-bromopyrimidine
5-Bromo-2-chloropyrimidine (7.5g) was added poπionwise over 15mιn to a solution of bis(2-methoxyethyl)amιne (20.7g) in methanol (200ml), then the solution was stirred at room temperature for 7h. The solvent was evaporated, water was added, and the product extracted into ether. Drying and evaporation of the solvent, followed by chromatography (silica. 1 • 1 ether/hexane) gave a pale oil (7.9g). ' H NMR (CDCI3) δ: 3.35 (s, 6H), 3.57 (t, 4H), 3.81 (t. 4H), 8.27 (s, 2H). MS (APCI+) Found (M+ l ) = 290/292. C ] 0H16BrN3O2 requires 290.
Intermediate B84 — Ethyl 3-(2-di(2-methoxyethyl)amino-5-pyrimidy )acrylate
A mixture of Intermediate B83 (6.09g), ethyl acrylate (24g), dιchlorobιs(tπphenyl- phosphιne)palladium(II) (0.73g), cuprous iodide (040g) and tnethylamine (7ml) was heated at reflux for 1 8h. Ethyl acetate and aqueous ammonium chloride were added, the mixture was filtered to remove insoluble solid, and the organic layer was separated, dried and evaporated. The crude product was purified by chromatography (silica, 50-757c ether in hexane) to yield a pale solid. Η NMR (CDCI3) δ: 1.33 (t, 3H), 3.35 (s. 6H), 3.60 (t, 4H), 3.89 (t. 4H), 4.25 (q, 2H). 6.3 (d, I H), 7.5 (d, I H), 8.48 (s, 2H). MS (APCI+) Found (M+ l ) = 310; C 15H23N3O4 requires 309; m.p. 54-56°C.
Intermediate B85 — Ethyl 3-(2-bis(2-methoxyethyl)amino-5-pyrimidyl)propionate
Prepared from Intermediate B84 by the method of Intermediate B 17. ' H NMR (CDCI3) δ: 1.25 (t, 3H). 2.54 (t, 2H), 2.75 (t, 2H), 3.35 (s. 6H). 3.60 (t, 4H), 3.83 (t, 4H). 4.13 (q, 2H), 8.27 (s, 2H). MS (APCI+) Found (M+ l ) = 312; C 15H25N3O4 requires 31 1.
Intermediate B86 — 5-(2-Bis(2-methoxyethyl)aminopyrimid-5-ylmethyl)-2-thiouracil
Prepared from Intermediate B85 by the method of Intermediate B20. Η NMR (CDCI3) δ: 3.36 (s. 6H), 3.43 (s, 2H). 3.59 (t. 4H), 3.82 (t, 4H), 6.94 (s, I H), 8.27 (s. 2H). MS (APCI+) Found (M+ l ) = 352; C 15H21 N5O3S requires 351.
Intermediate B87 — 2-(4-Fluorobenzyl)thio-5-(2-bis(2-methoxyethyl)aminopyrimid- 5-ylmethyI)pyrimidin-4-one
Prepared from Intermediate B86 by the method of Intermediate B IO. ' H NMR (CDCI3) δ: 3.34 (s, 6H), 3.51 (s, 2H), 3.57 (t, 4H), 3.81 (t, 4H), 4.38 (s, 2H), 6.99 (t, 2H), 7.35 (m, 2H),
7.69 (s, 1 H), 8.23 (s. 2H) 1 1.95 (br s. 1 H). MS (APCI+) Found (M+ 1 ) = 460; C22H26FN5O3S requires 459.
Example 1 — l-(3-(4-(4-Chlorophenyl)piperazin-l-yIcarbonyl)prop-l-yl)-2-(4- fluorobenzyl)thio-5-(2-methoxypyrimid-5-ylmethyl)pyrimidin-4-one
A mixture of l-(3-carboxyprop- l -yl)-2-(4-fluorobenzyl)thio-5-(2-methoxypyrimid-5-yl- methyl)pyrimidin-4-one (Intermediate B26, 0.29g. 1 equiv). l -(4-chlorophenyl)piperazine dihydrochloride (0.176g, 1 equiv), hydroxvbenzotriazole (0.01 g, 0.1 equiv). l-(3-dimethyl- aminopropyl)-3-ethylcarbodiimide (0.128g, 1.03 equiv), diisopropylethylamine (0.33ml, 2 equiv) and dichloromethane (5 ml) was stirred at room temperature under argon overnight, then diluted with dichloromethane and washed successively with aqueous ammonium chloride, water, aqueous sodium bicarbonate and brine. After drying and Filtration, the crude solution was applied directly to a lOg silica cartridge and the product eluted with 0- 5% methanol in dichloromethane. Evaporation of product fractions gave a white solid (0.40g). Η-NMR (CDC13) δ 1.95-2J5 (2H, m), 230-245 (2H,t), 3.00-3.20 (4H,m), 3.47- 3.59 (2H, t), 3.67 (2H, s), 3.69-3.8 (2H, t), 3.84-3.96 (2H, t), 3.99 (3H, s), 4.47 (2H, s), 6.75- 6.90 (2H, d), 6.91-7.05 (2H, t), 7J0 ( IH, s), 7.19-743 (4H, m), 8.48 (2H, s); MS (APCI+) found (M+l ) = 623; C3 1 H32N6O3SFCl requires 622.
Example 2 — l-(3-(4-(3,4-Dichlorophenyl)piperazin-l-ylcarbonyI)prop-l-yl)-2-(4- fluorobenzyl)thio-5-(2-methoxypyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B26 and l -(3,4-dichlorophenyl)piperazine by the method of
Example 1 . ' H NMR (CDCI3) δ 2.09 (2H. t); 236 (2H. t); 3.12 (4H, m): 3.51 (2H, m); 3.63 (2H. s): 3.72 (2H. m); 3.90 (2H. t); 3.99 (3H, s); 4.46 (2H, s): 6.72 ( I H. d): 6.95-6.99 (3H. m); 7.08 ( I H, s); 7.31 ( I H, d); 734-738 (2H. m); 8.46 (2H. s).
Example 3 — l-(3-(4-(4-TrifluoromethylphenyI)piperazin-l-ylcarbonyl)prop-l-yI)-2- (4-fluorobenzyl)thio-5-(2-methoxypyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B26 and l -(4-trifluoromethylphenyl)piperazιne by the method of Example 1. 1H NMR (CDCI3) δ 2.07 (2H, t); 2.36 (2H, t); 3.22-3.27 (4H. m); 3.52-3.55 (2H, m); 3.63 (2H, s); 3.73-3.76 (2H, m); 3.90 (2H. t); 3.99 (3H. s); 4.46 (2H, s): 6.90-6.99 (4H, m); 7.08 ( I H, s); 734-738 (2H, m); 7.50-7.53 (2H. d); 8.46 (2H. s).
Example 4 — l-(3-(4-(3J-Dichlorophenyl)piperazin-l-ylcarbonyl)prop-l-yl)-2-(4- fluorobenzyl)thio-5-(2-oxopyrimid-5-ylmethyl)pyrimidin-4-one
l-(3-(4-(3,4-dichlorophenyl)piperazin- l -ylcarbonyl)prop- l -yl)-2-(4-fluorobenzyl)thio-5-(2- methoxypyrirnid-5-ylrnethyl)pyrimidin-4-one (Example 2, 6.90g) was dissolved in dry dichloromethane (200ml), and bromocatechloborane (842g, 4 equiv) added. The resulting yellow suspension was stirred at reflux under argon for 24h. After cooling, water was added and stirring continued at room temperature for 30min. The organic layer was separated, dried and filtered, then applied to a silica column and the product eluted with 0- 25% methanol in dichloromethane. Product fractions were evaporated to a pale yellow solid (3.45g). ' H-NMR (CDCI3) δ 2.0-2.28 (2H. br.d), 2.23-2.58 (2H.br. t), 2.98-3.20 (4H, br.t), 335-3.60 (4H. br.m), 3.60-3.76 (2H, br.s), 3.90-4.15 (2H. br.t). 430-447 (2H. br.s), 6.62-6.11 ( I H, d), 6.80-7.00 (3H, t), 7.15-738 (3H, m), 7.84-8.03 ( I H, br.s), 8.29-8.60 (2H, br.s); MS (APCI+) found (M+ l ) = 644; C30H29N6O3SFCI2 requires 643.
Example s — l-(3-(4-(3,4-Dichiorophenyl)piperazin-l-yIcarbonyl)prop-l-yl)-2-(4- fluorobenzyl)thio-5-(l-methyl-2-oxopyrimid-5-ylmethyl)pyrimidin-4-one
A mixture of l -(3-(4-(3,4-dιchlorophenyl)pιperazιn- l-ylcarbonyl)prop- l-yl)-2-(4- fluorobenzyl)thιo-5-(2-oxopyπmιd-5-ylmethyl)pyrιmιdιn-4-one (Example 4, 0 50g, 0 78 mmol), methyl iodide (0 049ml, 0 78 mmol), potassium carbonate (0 24g, 1 73 mmol) and dry DMF ( lOml) was stirred under argon at room temperature for 2 days The solvent was evaporated, the residue dissolved in dichloromethane, and this solution washed with aquous potassium carbonate, dried and evaporated before applying to a lOg silica cartridge and eluting with 0-87c methanol in dichloromethane Product fractions were evaporated to a white solid (0 35g, 687c) ' H-NMR (CDCI3) 5 2 00-2 20 (2H, m). 2 33-2 49 (2H, m), 3 05- 3 24 (4H, m), 3 45 (2H, s), 3 57 (5H, s), 3 67-3 82 (2H, m), 3 87-4 05 (2H, t), 4 45 (2H, s),
6 68-6 83 ( I H, m), 6 88-7 08 (3H, m), 7 20-7 45 (4H, ), 7 94-8 04 ( I H, d), 8 40-8 52 ( IH, d), MS (APCI+) found (M+ l ) =657, C31H3 1N6O3SFCl2 requires 656
Example 6 — l-(3-(4-(3,4-DichlorophenyI)piperazin-l-ylcarbonyl)prop-l-yl)-2-(4- fluorobenzyl)thio-5-(l-ethoxycarbonylmethyI-2-oxopyrimid-5-ylmethyl)pyrimidin-4- one
Prepared by the method of Example 5. using ethyl bromoacetate in place of methy l iodide and carrying out the reaction at 40°C overnight White solid ' H-NMR (CDCI3) δ 1 22- 1 38 (3H, t), 2 03-2 22 (2H, m). 2 35-2 50 (2H, t), 3 02-3 23 (4H m), 3 37-3 63 (4H, ), 3 65-3 83 (2H, m), 3 87-4 06 (2H. t), 4 17-4 35 (2H, q), 4 45 (2H. s) 4 59 (2H. s). 6 68-6 81
( IH. m), 6 88-7 07 (3H, m), 7 20-7 43 (4H, m), 7 87-7 95 ( 1 H, d). 8 47-8 57 ( 1 H. d), MS (APCI+) found (M+ l ) = 729 C34H 5N6O5SFCl2 requires 728
Example 7 — l-(3-(4-(3,4-Dichlorophenyl)piperazin-l-ylcarbonyl)prop-l-yl)-2-(4- fluorobenzyl)thio-5-(l-carboxymethyI-2-oxopyrimid-5-yImethyl)pyrιmidin-4-one
l-(3-(4-(3,4-dιchlorophenyl)pιperazιn- l-y lcarbony l)prop- i -yl)-2-(4-fluorobenzyl)thιo-5-( l- ethoxycarbonylmethyl-2-oxopynmιd-5-ylmethyl)pynmιdιn-4-one (Example 6. 1 66g, 1 equiv) was dissolved in dioxan (22ml) and a solution of sodium hydroxide (87mg, 0 92 equiv) in water (5ml) was added The mixture was stirred overnight, then evaporated The residue was taken up in water, washed with ethyl acetate, then acidified to pH 4 with sodium bisulfate The precipitate was filtered off and washed with ethyl acetate and water to give the product as a white solid (0 91 g) ' H-NMR (DMSO) δ 1 83-2 03 (2H, br ), 2 34-2 58 (2H, br t), 3 06-3 25 (4H. br m), 3 37 (2H, s). 3 43-3 60 (4H. br m) 3 82-3 98 (2H, br t), 4 39 (2H, s), 4 55 (2H, s), 6 90-7 02 ( IH, dd), 7 04-7 20 (3H, m), 7 36-7 55 (3H, m), 7 75 ( I H, s), 8 00-8 07 ( I H, d), 8 54-8 63 ( IH, d), 13 00- 13 30 ( I H. br s), ), MS (APCI+) found (M+l ) = 701 , C32H
3 1N
6O
5SFCl2 requires 700
Example 8 — l-(3-(4-(4-Chlorophenyl)piperazin-l-ylcarbonyl)prop-l-yl)-2-(4- fluorobenzyl)thio-5-(2-methoxypyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B39 and l-(4-chlorophenyl)pιperazιne by the method of Example 1 ' H-NMR (CDC13) δ 2 04 (2H.m), 2 34 (2H,m), 3 0-3 15 (4H.m), 3 45-3 55 (2H,m), 3 57 (2H,s), 3 65-3 75 (2H,m), 3 86 (3H,s), 3 8-3 9 (2H,m). 4 48 (2H.s). 6 75-6 85 (2H,m), 6 9-7 05 (3H,m), 7 2-7 3 (2H,m), 7 3-7 4 (4H,m) MS (APCI+) found (M+l ) = 595, C30H32C1FN6O2S requires 594
Example 9 — l-(3-(4-(4-Trifluoromethylphenyl)piperazin-l-ylcarbonyl)prop-l-yl)-2- (4-fluorobenzyl)thio-5-(2-methoxypyrimid-5-ylmeth l)pyrimidin-4-one
Prepared from intermediate B39 and l-(4-trifluoromethylphenyl)piperazιne by the method of Example 1 . Η-NMR (CDC13) δ 1.95-2.1 (2H,m), 235 (2H,m), 3.15-33 (4H,m). 3.45- 3.55 (2H,m), 3.57 (2H.s), 3.7-3.8 (2H,m), 3.8-3.95 (5H.m), 4.48 (2H,s), 6.85-7.0 (5H,s), 7.3-7.4 (4H.m), 745-7.55 (2H,m); MS (APCI+) found (M+l ) = 629: C31 H32F4N6O2S requires 628.
Example 10 — l-(4-Benzylpiperazin-l-ylcarbonylmethyl)-2-(4-fluorobenzyl)thio-5-(l- methy pyrazol-4-yImethyl)pyrimidin-4-one
Prepared from intermediate B 12 and 1-benzylpiperazine by the method of Example 1. H-
NMR (CDCI3) δ 2.46 (4H, ), 3.40 (2H, m), 3.52 (2H, s), 3.63 (2H, ), 3.72 (2H. s), 4.48 (2H. s), 4.54 (2H. s). 6.88 ( IH, s), 6.99 (2H, m), 7.24-738 (7H. m). 8.70 (2H. s). 9.09 ( IH. s): MS (APCI+) found (M+ l ) = 545: C29H29FN6O2S requires 544.
Example 11 — l-(4-(Oct-l-yl)piperazin-l-ylcarbonylmethyl)-2-(4-fluorobenzyl)thio- 5-(l-methylpyrazol-4-y methyl)pyrimidin-4-one
Prepared from intermediate B34 and 1 -octylpiperazine by the method of Example 1. H- NMR (CDCI3) δ 0.80-0.95 (3H. t), 1.15- 1 42 ( 1 OH, br.s), 1.40- 1.63 (2H, br.s). 230-2.70 (6H. br.m). 335-3.55 (2H. br.s), 3.60 (2H, s), 3.62-3.77 (2H. br.s), 3.85 (3H. s). 4.50 (2H. s), 4.52 (2H. s), 6.78 ( 1 H. s), 6.92 7.07 (2H. t). 7.28-7.42 (4H. m ); MS ( APCI+) found
(M+ 1 ) = 569; C30H4 ] N6O SF requires 568.
Example 12 — l-(4-(Oct-l-ylaminocarbonyl)piperazin- l-y carbonylmethyl)-2-(4- fluorobenzyl)thio-5-(l-methylpyrazol-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates A4 and B34 by the method of Example 1. ' H-NMR (CDCI3) δ 7.20 (4H. m). 6.83 (2H. t), 6.56 ( IH. s), 4.34 (4H, s), 4.24 ( IH, s), 3.69 (3H, s), 3.46 (2H, ), 343 (2H. s), 335 (2H, m). 3.25 (2H. s), 3.16 (2H. m), 3.05 (2H. m), 1.32 (2H, m), 1.1 1 ( 10H, m), 0.71 (3H, t); MS(APCI+) found (M+l ) = 612. C31 H42FN7O3S requires 61 1.
Example 13 — l-(4-(Oct-l-ylsulfonyl)piperazin-l-yIcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-( l-methylpyrazol-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates A2 and B34 by the method of Example 1. Η-NMR (CDCI3) δ θ.88 ( 3H, t), 1.18- 147 ( 10H, ), 1.73- 1.85 (2H. m), 2.89 (2H, m), 3.31 (4H, m), 349
(2H, m), 3.60 (2H. s). 3.70 (2H. ), 3.86 (3H, s). 4.50 (2H. s), 4.53 (2H. s), 6.73OH, s),
6.99 (2H, m). 7.26-738 (4H, m); MS (APCI+) found (M+ l ) = 633; C30H41 FN6O4S2 requires 632.
Example 14 — l-(4-(4-ChlorophenyI)piperazin-l-ylcarbonylmethyI)-2-(4- fluorobenzyI)thio-5-(pyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B 12 and l -(4-chlorophenyl)piperazιne by the method of Example 1 . Pale yellow solid. Η-NMR (CDCI3) δ 3.05-3.27 (4H. m), 3.50-3.64 (2H. br.t),
3.69-3.88(4H. m), 4.46 (2H, s), 4.62 (2H. s), 6.78-6.88 (2H. d), 6.89-7.06 (3H, m), 7.16- 7.43 (4H. m). 8.70 (2H. s), 9.08 ( I H. s): MS (APCI+) found (M+ l ) = 565:
C2 H26 lFN( θ2S requires 564.
Example 15 — l-(4-(3J-DichlorophenyI)piperazin-l-yIcarbonyimethyl)-2-(4- fluorobenzyl)thio-5-(pyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B 12 and l-(3,4-dichlorophenyl)piperazine by the method of
Example 1. Η-NMR (CDC13) δ 3.05-33 (4H,m), 345-3.65 (2H.m), 3.65-3.85 (4H,m), 4.48 (2H.s). 4.61 (2H,s), 6.65-6.8 ( l H. ), 6.85-7.1 (4H,m), 7.25-745 (3H.m), 8.70 (2H,s), 9J0 ( l H.s): MS (APCI+) found (M+ l ) = 599; C2gH25Cl2FN6O2S requires 598.
Example 16 — l-(4-(3-TrifluoromethyIphenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(pyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B 12 and l -(3-trifluoromethylphenyl)pιperazine by the method of Example 1. ' H-NMR (CDCI3) δ 3.26 ( 4H. ), 3.60 (2H. m). 3.73 (2H. s). 3.80 (2H,m), 4.87 (2H. s ). 4.62 (2H, s), 6.90-743 (9H, m), 8.70 (2H, s), 9.10 ( I H. s); MS (APCI+) found (M+l ) = 599: C29H26F4N6O2S requires 598.
Example 17 — l-(4-(4-Trifluoromethy pheny )piperazin-l-ylcarbonylmethyI)-2-(4- fluorobenzyl)thio-5-(pyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B 12 and l -(4-trifluoromethylphenyl)piperazine by the method of Example 1. ' H-NMR (CDCI3) δ 3.30 (4H, m), 3.60 (2H, m). 3.73 (2H. s). 3.79 (2H, m), 4.48 (2H. s). 4.62 (2H, s), 6.90-7.02 (5H, m), 730-737 (2H, m), 7.52 (2H. m). 8.70 (2H, s) 9.09 ( IH, s); MS (APCI+) found (M+l ) = 599; C29H26F4 6O2S requires 598.
Example 18 — l-(4-(3,4-Dichlorophenyi)piperazin-l-yIcarbonyimethyϊ)-2-(4- fluorobenzyl)thio-5-(2-methoxypyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B23 and l -(3,4-dichlorophenyl)piperazine by the method of Example 1 . White solid. Η-NMR (CDCI3) δ 3.05-3.25 (4H. br.s), 347-3.60 (2H. br.d), 3.65 (2H, s), 3.70-3.85 (2H, br.d), 3.97 (3H, s), 447 (2H, s), 4.56 (2H, s), 6.64-6.84 (2H, m), 6.88-7.06 (3H, m), 7.20-742 (3H, m), 8.43 (2H, s); MS (APCI+) found (M+ l ) = 629; C29H27FCI2N5O3S requires 628.
Example 19 — l-(4-(4-Chlorophenyl)piperazin-l-ylcarbonylmethyI)-2-(4- fluorobenzyl)thio-5-(2-methoxypyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate B23 and l -(4-chlorophenyl (piperazine by the method of Example 1. Pale yellow solid. Η-NMR (CDCI3) δ 3.07-3.28 (4H, br.t), 3.51 -3.62 (2H. br.t). 3.65 (2H, s), 3.70-3.85 (2H, br.t),3.98 (3H, s), 4.48 (2H, s), 4.61 (2H. s), 6.75-6.90 (3H, m). 6.91-7.05 (2H. t). 7.17-742 (4H, m). 8.42 (2H, s): MS (APCI-) found (M- I ) = 593 C29H28N6O3SFC1 requires 594.
Example 20 — l-(4-(4-Chlorophenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyI)thio-5-(2-methylpyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediate A9 and B45 by the method of Intermediate B 1 1 . ' H-NMR (CDC13) δ 8.58 (2H. s), 7.35 (2H. m), 7.25 (2H, m), 6.96 (2H, t), 6.81 (3H. m). 4.57 (2H, s), 4.49 (2H. s), 3.77 (2H. m), 3.69 (2H, s). 3.56 (2H, m), 3.15 (4H, m), 2.71 (3H, s), MS(APCI+) found (M+l ) = 579. C29H2 ClFN6O2S requires 578.
Example 21 — l-(4-(4-Chlorophenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyI)thio-5-(2-cyclopropylpyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediates A9 and B51 by the method of Intermediate B 1 I . ' H-NMR (DMSO) 8.33 (2H, s). 7.38 ( I H, s), 7.27 (2H, dd), 7.08 (2H, d), 6.94 (2H. d), 6.79 (2H, d),
4.77 (2H, s), 4.21 (2H. s). 3.40 (4H. m). 3.34 (2H, s). 3.02 (2H, m). 2.94 (2H. ). 1.97 ( I H, m), 0.79 (4H,m).
Example 22 — l-(4-(4-ChlorophenyI)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(2-(lJ-dimethylethyl)pyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediates A9 and B57 by the method of Intermediate B 1 1 . White solid. 1 H-NMR (DMSO) δ 133 (9H. s), 3.05-3.26 (4H, br.m). 348-3.70 (6H. m), 4.39 (2H, s), 4.66 (2H, s). 6.93-7.05 (2H. d). 7.06-7.20 (2H. t), 7.22-732 (2H. d ). 740-7.52 (2H. t). 7.18 ( I H, s), 8.63 (2H. s); MS (APCI+) found (M+ l ) = 621 ; C32H34CIFN6O2S requires 620.
Example 23 — l-(4-(4-Chlorophenyl)piperazin-l-yIcarbonylmethy )-2-(4- πuorobenzyl)thio-5-(2-trifluoromethylpyrimid-5-ylmethyI)pyrimidin-4-one
Prepared from intermediates A9 and B66 by the method of Intermediate B 1 1. ' H-NMR (CDC1 ) δ 8.80 (2H, s). 1.29-1.26 (2H, m), 7.19-7.16 (2H, m), 6.95 ( I H. s), 6.91 (2H, t), 6.76 (2H. d), 4.55 (2H, s), 4.42 (2H, s), 3.72-3.70 (4H, m), 3.53-3. 1 (2H, m), 3.13-3.07 (4H, m); MS (APCI+) found (M+ l ) = 633, C29H2535ClF4N6O2S requires 632.
Example 24 — l-(4-(4-Chlorophenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(2-(l-morpholino)pyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediates A9 and B78 by the method of Intermediate B 1 1. ' H NMR (DMSO-d6) δ: 3.12 (m. 2H), 3.17 (m. 2H). 3.28 (s, I H), 3.40 (s. I H). 3.58 (m, 4H), 3.63 (s. 8H). 4.39 (s. 2H). 4.95 (s, 2H). 6.97 (d, 2H), 7.12 (t, 2H), 7.25 (d. 2H), 7.45 (m. 3H). 8.28 (s, 2H). MS (ES+) Found (M+ l ) = 650; C32H33CIFN7O3S requires 649.
Example 25 — l-(4-(4-Chlorophenyl)piperazin-l-ylcarbonylmethyI)-2-(4- fluorobenzyl)thio-5-(2-dimethylaminopyrimid-5-y methyl)pyrimidin-4-one
Prepared from intermediates A9 and B82 by the method of Intermediate B 1 1. ' H NMR (DMSO-d6) δ: 3.08 (s, 6H), 3.12 (m, 2H), 3.19 < m, 2H). 3.28 (s. I H). 338 (s. IH). 3.58 (m. 4H), 4.39 (s. 2H), 4.94 (s. 2H), 6.97 (d, 2H), 7.12 (t. 2H). 7.26 ( d. 2H). 7.44 (m. 3H). 8.23
(s, 2H). MS (ES+) Found (M+ 1 ) = 608; C30H3 1 CIFN7O2S requires 607.
Example 26 — l-(4-(4-Chlorophenyl)piperazin-l-yIcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(2-di(2-methoxyethyl)aminopyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from intermediates A9 and B87 by the method of Intermediate B 1 1. ' H NMR
(DMSO-d6) δ: 3.12 (m. 2H). 3.20 (m. 2H), 3.24 (s, 6H), 3.38 (s, 2H). 3.47 (t, 4H). 3.59 ( , 4H), 3.71 (t, 4H). 4.39 (s, 2H), 4.95 (s, 2H), 6.97 (d. 2H). 7.12 (t. 2H), 7.26 (d. 2H), 7.45 (m, 2H), 7.52 (s, I H), 833 (s. 2H). MS (APCI+) Found (M+l ) = 696; C34H39CIFN7O4S requires 695.
Example 27 — l-(4-(4-ChlorophenyI)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(2-oxopyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from Example 19 by the method of Example 4. ' H-NMR (DMSO) δ 3.00-3.27 (4H, m), 3.32 (2H. s). 345-3.75 (4H, br.s). 4.38 (2H, s), 4.95 (2H. s), 6.90-7.05 (2H, d), 7.07-7.20 (2H. t), 7.21 -7.32 (2H, d). 738-7.55 (3H, mj. 7.94-836 (2H. br.s): MS (APCI-) found (MJ ) = 579; C28H26N6O3SFCI requires 580.
Example 28 — l-(4-(3J-Dichiorophenyl)piperazin-l-ylcarbonylmethyI)-2-(4- fluorobeπzyl)thio-5-(2-oxopyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from Example 18 by the method of Example 4. Light brown solid. ' H-NMR (DMSO) δ 3.08-3.44 (6H, m), 345-3.68 (4H. s), 4.38 (2H. s), 4.93 (2H. s). 6.90-7.01 ( I H, d), 7.05-7.22 (3H. ), 735-7.53 (4H. m), 7.80-8.50 (2H. br.s); ); MS (APCI+) found (M+l ) = 615; C2 H25N6θ3SFCh requires 614.
Example 29 — l-(4-(4-Chlorophenyl)piperazin-l-yIcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(l-methyl-2-oxopyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from Example 27 by the method of Example 5. White solid. Η-NMR (CDCI3) δ 3.05-330 (4H, ). 3.43 (2H, s). 3.49 (3H, s), 3.56-3.69 (2H, br.t). 3.70-3.85 (2H. br.t), 4.45 (2H, s). 4.89 (2H, s), 6.75-6.90 (2H, d), 6.91-7.06 (2H, t), 7J6-740 (4H, m), 7.45 ( IH, s), 7.93-8.02 ( IH, d), 832-8.43 ( IH, d); ); MS (APCI+) found (M+l) = 595; C29H28N6°3SFC1 requires 594.
Example 30 — l-(4-(4-Chlorophenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyI)thio-5-(l-ethyl-2-oxopyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from Example 27 by the method of Example 4. except using ethyl iodide in place of methyl iodide. Pale yellow solid. Η-NMR (CDCI3) δ 1 .29- 146 (3H. t). 3.01-330 (4H. m), 3.46 (2H. s), 3.55-3.70 (2H. br.t), 3.70-3.81 (2H. br.t), 3.84-4.02 (2H. q),446 (2H. s),4.86 (2H. s), 6.75-6.89 (2H. d). 6.91 -7.05 (2H, t). 7.02-747 (5H. m), 7.89-8.05 ( I H. d), 830-843 ( I H. s); MS (APCI+) found (M+ l ) = 609; C30H30N6O3SFC1 requires 608.
Example 31 — l-(4-(3J-DichlorophenyI)piperazin-l-ylcarbonylmethyI)-2-(4- fluorobenzy )thio-5-(l-ethyl-2-oxopyrimid-5-ylmethyl)pyrimidin-4-one
Prepared from Example 28 by the method of Example 4, except using ethyl iodide in place of methyl iodide. ' H-NMR (CDC1
3) δ 132- 145 (3H, t). 3.08-331 (4H. m). 3.45 (2H, s), 347-3.69 (2H. br.t). 3.71 -3.84 (2H, br.t), 3.85-4.00 (2H, q), 4.48 (2H. s), 4.83 (2H, s). 6.67- 6.79 ( IH, d), 6.90-7.05 (3H. t), 7.20-741 (4H, m), 7.92-8.00 ( IH, d), 834-841 ( I H. d); MS (APCI+) found (M+ l ) = 643: C30H29N6O3SFCI2 requires 642.
Example 32 — l-(4-(34-Dichlorophenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(l-(cyclopropylmethyl)-2-oxopyrimid-5-ylmethyI)pyrimidin-4-one
Prepared from Example 28 by the method of Example 4. except using bromomethylcyclo- propane in place of methyl iodide and carrying out the reaction at 40°C overnight. H- NMR (CDCI3) δ 030-047 (2H. m), 0.58 (2H. m), 1.18- 140 ( I H, m), 3.01 -332 (4H. m). 3.48 (2H. s). 3.57-3.88 (6H, m). 4.47 (2H, s). 4.93 (2H, s). 6.65-6.82 ( I H. d). 6.84-7.07 (3H. m), 7.19-740 (3H. m), 7.45 ( IH, s), 8.05-8.19 ( I H, d). 8.27-S39 ( I H, d): MS (APCI+) found (M+ 1 ) = 669; C
3 H
3 j N
6O
3SFCh requires 668.
Example 33 — l-(4-(3J-DichlorophenyI)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(l-(ethoxycarbonylmethyI)-2-oxopyrimid-5-ylmethyI)pyrimidin-4- one
Prepared from Example 28 by the method of Example 4, except using ethyl bromoacetate in
place of methyl iodide and carrying out the reaction at 40°C overnight. ' H-NMR (CDCI3) δ 1.23- 147 (3H, t), 3.08-3.29 (4H, br.m), 3.48 (2H, s), 3.51 -3.66 (2H. br.t). 3.69-3.82 (2H, br.t), 4.03-4.28 (2H. q), 4.49 (2H, s), 4.56 (2H. s), 4.85 (2H. s), 6.68-6.78 ( I H. d), 6.88-7.07 (3H, m), 7.28-740 (4H, m), 7.84-7.92 ( 1H, d), 836-848 ( IH, d); ); MS (APCI+) found (M+ 1 ) = 701 ; C
3 H
31 N
6O
5SFCl2 requires 700.
Example 34 — l-(4-(3J-DichlorophenyI)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(l-(dimethylaminocarbonylmethyl)-2-oxopyrimid-5- ylmethyl)pyrimidin-4-one
Prepared from Example 33 by hydrolysis to the acid using the method of Example 7, followed by reaction with dimethylamine hydrochloride using the method of Example 1. 'H-NMR (CDCI3) δ 2.94 (3H, s), 3.07 (3H, s), 3.10-330 (4H. m), 3.48 (2H, s), 3.54-3.67 (2H, br.s). 3.68-3.83 (2H, br.s), 4.48 (2H, s), 4.70 (2H, s), 4.85 (2H, s), 6.67-6.78 ( IH, m), 6.88-7.04 (3H, ), 7.20-744 (4H, m), 7.74-7.83 ( IH, d), 835-845 ( I H. d): MS (APCI+) found (M+ l ) = 701 ; C32H32N7O4SFCI2 requires 699.
Example 35 — l-(4-(4-ChIorophenyl)piperazin-l-ylcarbonylmeth\I)-2-(4- fluorobenzyl)thio-5-(l-methylpyrazoI-4-yImethyI)pyrimidin-4-one
Prepared from intermediate B34 and l -(4-chlorophenyl)piperazιne by the method of Example 1 . Η-NMR (CDCI3) δ 3.15 (4H, m), 3.56 (2H, ), 3.60 ( 2H. s). 3.76 (2H, m).
3.85 (3H. m). 4.50 (2H, s). 4.55 (2H. s), 6.81 (3H, ). 6.97 (2H. m ). 7.22-738 (6H. m ): MS (APCI+) found (M+ l ) = 567; C2gH2gClFN6O2S requires 566.
Example 36 — l-(4-(3J-Dichlorophenyl)piperazin-l-yIcarbonylrnethyI)-2-(4- fluorobenzyl)thio-5-(l-methylpyrazol-4-ylmethyI)pyrimidin-4-one
Prepared from intermediate B34 and l-(3,4-dichlorophenyI)piperazine by the method of Example 1 . Η-NMR (CDC1 ) δ 3.18 (4H, m), 3.56 (2H, m), 3.60 (2H, s). 3.76 (2H. m), 3.86 (3H, s), 4.50 (2H, s), 4.56 (2H. s). 6.72 (2H. m), 6.96 (3H. m), 7.32 (5H, m); MS (APCI+) found (M+ l ) = 601 ; C2 H27Cl2FN6O2S requires 600.
Example 37 — l-(4-(2,4-Dichlorophenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzy )thio-5-(l-methylpyrazol-4-ylmethyl)pyrimidin-4-one
Prepared from intermediate B34 and l -(2,4-dichlorophenyl)piperazine by the method of Example 1. ' H-NMR (CDC13) δ 2.9-3.1 (4H,m), 3.5-3,7 (4H,m), 3.75-3.85 (2H,m), 3.86
(3H,s), 4.51 (2H,s), 4.56 (2H.s). 6.77 ( lH.s), 6.89 ( l H,d), 6.99 (2H,bt), 7.15-745 (6H,m); MS (APCI+) found (M+l ) = 601 : C2gH27Cl2FN6O2S requires 600.
Example 38 — l-(4-(4-TrifluoromethylphenyI)piperazin-l-ylcarbony rnethyl)-2-(4- fluorobenzyl)thio-5-(l-methylpyrazol-4-ylmethy )pyrimidin-4-one
Prepared from intermediate B34 and l -(4-trifluoromethylphenyl)piperazine by the method of Example 1. Η-NMR (CDCI3) δ 3.30 (4H, m), 3.67 (4H, m). 3.78 (2H. ), 3.85 (3H. s) 4.50 (2H. s). 4.57 (2H,s). 6.78 ( I H, s), 6.89-7.00 (4H. ). 7.26-7.37 (4H, m) 7.68 (2H, m); MS (APCI+) found (M+ l ) = 601 C29H28F4N6O2S requires 600.
Example 39 — l-(4-(4-Chlorophenyl)piperazin-l-ylcarbonyimethyl)-2-(4- fiuorobenzyl)thio-5-(l-ethylpyrazol-4-ylmethyl)pyrimidin-4-one
Prepared from intermediate B74 and l-(4-chlorophenyl)piperazine by the method of Example 1. 1 H-NMR (CDC13) δ, 737-734 (4H, m), 7.26-7.23 (2H. m), 6.98 (2H, t), 6.81-
6.77 (3H. m), 4.55 (2H, s), 4.5 1 (2H, s), 4J2 (2H, q), 3.77-3.75 (2H, m), 3.61 (2H. m), 3.57-3.54 (2H, m), 3J9-3J2 (2H, m), 1.47 (3H, t); MS (ES+) found (M+ l ) = 581 , c29H30 5ciFN6°2s requires 580.
Example 40 — l-(4-(5-TrifluoromethyIpyrid-2-yl)piperazin-l-y carbonylmethyl)-2- (4-fluorobenzyI)thio-5-(l-methylpyrazoI-4-ylmethyl)pyrimidin-4-one
Prepared from intermediate B34 and l -(5-trifluoromethyl-2-pyridyl)piperazine by the method of Example 1. Η-NMR (CDC13) δ 345-3.7 (6H.m), 3.7-3.9 (7H.m). 4.48 (2H.s), 4.61 (2H.s), 6.65 ( lH.bd), 6.83 ( l H.s), 6.98 (2H,bt). 7.25-74 (4H,m). 7.65-7.75 ( lH,m), 841 ( I H.bs); MS (APCI+) found (M+ I ) = 602; C28H27F4N7O2S requires 601.
Example 41 — l-(4-(5-Chloropyrid-2-yl)piperazin-l-yIcarbonyimethyI)-2-(4- fiuorobenzyl)thio-5-(l-methylpyrazoI-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates A5 and B34 by the method of Example 1 . ' H-NMR (CDC13 )
δ 34-3.45 (4H.m). 3.61 (2H,s), 3.61 -3.8 (4H.m). 3.S6 (3H.s). 4.51 (2H.s), 4.55 (2H.s), 6.59 ( l H,d), 636 ( l H.s), 6.97 (2H,t). 7.25-74 (4H,m). 745-7.5 ( I H.m), 8.13 ( l H.m): MS (APCI+) found (M+ l ) = 568; C27H27CIFN7O2S requires 567.
Example 42 — l-(4-(3,5-Dichloropyrid-2-yl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(l-methylpyrazol-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates A6 and B34 by the method of Example 1 . ' H-NMR (CDCI3) δ 3.25-345 (4H,m), 3.5-3.65 (4H.m). 3.7-3.85 (2H.m). 3.86 ( 3H.s), 4.5 1 (2H.s), 4.56 (2H,s), 6.78 ( lH.s), 6.98 (2H.bt), 7.25-74 (4H,m), 7.64 ( I H.bs), 8.13 ( I H.bs); MS (APCI+) found (M+l ) = 602; C27H26CI2FN7O2S requires 601.
Example 43 — l-(4-(3-Chloro-5-trifluoromethylpyrid-2-yl)piperazin-l- ylcarbonylmethyl)-2-(4-fluorobenzyl)thio-5-(l-methylpyrazol-4-ylmethyl)pyrimidin-4-
Prepared from intermediate B34 and l -(3-chloro-5-trifluoromethyl-2-pyridyl)piperazine by the method of Example 1 . Η-NMR (CDCI3) δ 345-3.65 (8H.m). 3.7-3.8 (2H.m). 3.86 (3H,s), 4.5 1 (2H,s). 4.57 (2H,s), 6.77 ( lH,s), 6.98 (2H.bt). 7.25-74 (4H.m), 7.81 ( I H.bs), 8.41 ( I H.bs): MS (APCI+) found (M+ l ) = 636; C
2gH
26ClF
4N7θ2S requires 635.
Example 44 — l-(3-Oxo-4-dec-l-ylpiperazin-l-ylcarbonylmethyI)-2-(4- fluorobenzyl)thio-5-(l-methylpyrazol-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates AS and B34 by the method of Example 1. ' H-NMR (CDCI3) δ 0.88 (3H, ). 1.26 ( 14H, m). 145- 1.12 (2H. m), 3.38 (4H, m), 3.60 (2H, s). 3.81 (2H, m), 3.87 (3H, s), 4.06 and 4.20 (2H. 2x s), 4.51 (4H, ), 632 ( IH, s). 6.99 (2H. m), 732 (4H, m); MS (APCI+) found (M+ l ) = 61 1 ; C32H43FN6O3S requires 610.
Example 45 — l-(4-(4-(4-FIuorophenyl)phenyl)piperazin-l-ylcarbonylmethyl)-2-(4- fluorobenzyl)thio-5-(l-methylpyrazoI-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates A 10 and B34 by the method of Example 1 . Η-NMR (CDCI3) δ 3.2-33 (4H.m), 3.58 (2H,m), 3.61 (2H.s), 3.80 (2H,m), 3.86 (3H.s). 4.51 (2H,s), 4.57
(2H,s), 6.78 ( I H.s), 6.96 (4H.m), 7.10 (2H,m), 7.31 ( lH,s), 7.35 (3H.m), 7.4-7.5 (4H.m); MS (APCI+) found (M+ l ) = 627; C34H32F2N6O2S requires 626.
Example 46 — l-(4-(4-(4-Chlorophenyl)phenyl)piperazin-l-ylcarbonylmethyI)-2-(4- fluorobenzyI)thio-5-(l-methyIpyrazoI-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates Al 1 and B34 by the method of Example 1. ' H-NMR
Example 47 — l-(4-(4-(4-Trifluoromethylphenyl)phenyl)piperazin-l-ylcarbonyI- methyl)-2-(4-fluorobenzyl)thio-5-(l-methylpyrazol-4-ylmethy )pyrimidin-4-one
Prepared from intermediates A 12 and B34 by the method of Example 1. ' H-NMR (d6~
DMSO) δ 3.23 (2H,m), 3.29 (2H.m). 338 (2H.s), 3.61 (4H,m), 3.76 (3H.s), 4.41 (2H,s). 4.96 (2H.s), 7.08 ( I H.d), 7. 14 ( I H.m), 7.25 ( I H.s), 7.36 ( I H.s). 74-7.5 ( I H.m), 7.64 ( I H.d), 7.75 ( I H.d), 7.84 ( I H.d); MS (APCI+) found (M+ l ) = 677; C35H 2F4N6O2S requires 676.
Example 48 — l-(4-(4'-Biphenyl)piperazin-l-ylcarbonylmethyl)-2-(4-πuorobenzyl)- thio-5-(l-methyIpyrazol-4-ylmethyl)pyrimidin-4-one
Prepared from intermediates A 13 and B34 by the method of Example 1. ' H-NMR
39
Biological Data
1. Screen for Lp-PLA2 inhibition.
Enzyme activity was determined by measuring the rate of turnover of the artificial substrate (A) at 37 C in 50mM HEPES (N-2-hydroxyethylpiperazine-N'-2-ethanesulphonic acid) buffer containing 150mM NaCl, pH 7.4.
(A)
Assays were performed in 96 well titre plates.
Recombinant LpPLA2 was purified to homogeneity from baculovirus infected Sf9 cells, using a zinc chelating column, blue sepharose affinity chromatography and an anion exchange column. Following purification and ultrafiltration, the enzyme was stored at
6mg/ml at 4°C. Assay plates of compound or vehicle plus buffer were set up using automated robotics to a volume of 170μl. The reaction was initiated by the addition of 20μl of lOx substrate (A) to give a final substrate concentration of 20μM and 10 μl of diluted enzyme to a final 0.2nM LpPLA2. The reaction was followed at 405 nm and 37 °C for 20 minutes using a plate reader with automatic mixing. The rate of reaction was measured as the rate of change of absorbance.
Results
The compounds described in Examples 3, 18, 19 and 36 were tested as described above and had respective IC5 values of 2, 8, 20 and 12 nM.