WO1999057246A1 - Animal cell culture media comprising non-animal or plant-derived nutrients - Google Patents
Animal cell culture media comprising non-animal or plant-derived nutrients Download PDFInfo
- Publication number
- WO1999057246A1 WO1999057246A1 PCT/US1999/009574 US9909574W WO9957246A1 WO 1999057246 A1 WO1999057246 A1 WO 1999057246A1 US 9909574 W US9909574 W US 9909574W WO 9957246 A1 WO9957246 A1 WO 9957246A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- animal
- cell
- culture medium
- cell culture
- derived
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/0018—Culture media for cell or tissue culture
- C12N5/0043—Medium free of human- or animal-derived components
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
- C12N2500/10—Metals; Metal chelators
- C12N2500/12—Light metals, i.e. alkali, alkaline earth, Be, Al, Mg
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
- C12N2500/10—Metals; Metal chelators
- C12N2500/20—Transition metals
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/32—Amino acids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/36—Lipids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/40—Nucleotides, nucleosides or bases
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/70—Undefined extracts
- C12N2500/74—Undefined extracts from fungi, e.g. yeasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/70—Undefined extracts
- C12N2500/76—Undefined extracts from plants
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/90—Serum-free medium, which may still contain naturally-sourced components
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/11—Epidermal growth factor [EGF]
Definitions
- the present invention generally relates to cell culture medium formulations. Specifically, the present invention provides cell culture medium formulations which comprise one or more non-animal derived peptides and more specifically one or more plant peptides for facilitating the in viti-o cultivation of animal cells.
- the present invention also relates to media formulations which comprise one or more non-animal derived lipids and/or fatty acids, and more specifically one or more plant lipids and/or fatty acids for cultivation of animal cells in vitro.
- the formulations of the invention may also comprise one or more of such non-animal or plant peptides and one or more of such non-animal or plant lipids and/or fatty acids.
- non-animal or plant-derived peptides and non-animal or plant-derived lipids and/or fatty acids may be used as substitutes for one or more animal-derived culture media components.
- the invention also provides methods for cultivating animal cells using these non-animal or plant nutrient-based culture media.
- the media of the present invention arc particularly suited for virus production in animal cells.
- Cell Culture Media Cell culture media provide the nutrients necessary to maintain and grow cells in a controlled artificial and in vitro environment.
- the characteristics and compositions of the cell culture media vary depending on iho particular cellular requirements. Important parameters include osmolariry, pH, and nutrient formulations.
- Media formulations have been used lo cultivate a number of cell types including animal, plant and bacterial cells.
- Cells cultivated in culture media catabolize available nutrients and produce useful biological substances such as monoclonal antibodies, hormones, growth factors and the like. Such products have therapeutic applications and, with the advent of recombinunt DNA technology, cells can be engineered to produce large quantities of these products.
- Cultured cells arc also routinely used for the isolation, identification and growth of viruses.
- the ability to cultivate cells in vitro is not only important for the study of cell physiology, but is also necessary for the production of useful substances which may not otherwise be obtained by cost-effective means.
- Typical components of cell culture media include amino acids, organic and inorganic salts, vitamins, trace metals, sugars, lipids and nucleic acids, the types and amounts of which may vary depending upon the particular requirements of a given cell or tissue type.
- cell culture media formulations are supplemented with a range of additives, including undefined components such as fetal bovine scrum (FBS) (10-20% v/v) or extracts from animal embryos, organs or glands (0.5-10% v/v). While FBS is the most commonly applied supplement in animal cell culture media, other serum sources are also routinely used, including newborn calf, horse and human. These types of chemically undofi ⁇ cd supplements serve several useful functions in cell culture media (Lambert, K. J. ei al.. In: Animal Cell Biotechnology, Vol. 1, Spier, R. E. et al., Eds., Academic Press New York, pp. 85-122 (1985)).
- FBS fetal bovine scrum
- these supplements provide carriers or chelators for labile or water-insoluble nutrients; bind and neutralize toxic moieties; provide hormones and growth factors, protease inhibitors and essential, of t en unidentified or undefined low molecular weight nutrients; and protect cells from physical stress and 5- damage.
- serum and/or animal extracts are commonly used as relatively low-cost supplements to provide an optimal culture medium for the cultivation of animal cells.
- a major fear is the presence of prions causing spongiform ⁇ cnccphalopathy in humans or animals.
- Cell surface chemistry which is a critical portion of the in vitro microcnvironment for many cell types, can be adversely modified via adsorption or incorporation of scrum or extract proteins.
- undefined components such as serum or animal extracts also prevents the true definition and elucidation of the nutritional and hormonal requirements of the cultured cells, thus eUminating the ability to study, in a controlled way, the effect >0 of specific growth factors or nutrients on cell growth and differentiation in culture.
- undefined supplements prevent the researcher from studying aberrant growth and differentiation and the disease-related changes in cultured cells.
- scrum and animal extract supplementation of culture media can also complicate and increase the costs of purification of the desired substances from the culture media due to nonspecific co- 25 purification of scrum or extract proteins.
- serum-free media which often are specifically formulated to support the culture of a single cell type, incorporate defined quantities of purified growth factors, lipoprotcins and other proteins usually provided by the scrum or extract supplement. Since the components (and concentrations thereof) in such culture media are precisely known, these media are generally referred to as “defined culture media” and often as “serum-free media” or "SFM.” ⁇ number of SFM formulations are commercially available, such as those designed to support the culture of cndothelial cells, keratinocytes, monocytes/macrophagcs, fibroblasts. neurons, lymphocytes, chondrocytes or hepalocytes which arc available from Life Technologies, inc. (Rockvillc, MD).
- SFM generally provide several distinct advantages to the user. For example, the use of SFM facilitates the investigation of the effects of a specific growth factor or other medium component on cellular physiology, which may be masked when the cells are cultivated in serum- or cxtract- containing media. In addition, SFM typically contain much lower quantities of protein (SFM are often called "low protein media") than those containing serum or extracts, rendering purification of biological substances produced by cells cultured in SFM far simpler and more cost-effective.
- SFM small cell culture
- Such media (often called “basal media”), however, arc usually seriously deficient in the nutritional content required by most animal cells.
- SFM incorporate additional components into the basal media to make the media more nutritionally complex, while maintaining the serum-free and low protein content of the media.
- additional components include serum albumin from bovine (BSA) or human (HSA), animal- derived lipids such as human Excylc, sterols, etc., and certain growth factors or hormones derived from natural (animal) or recombinant sources.
- supplements derived from the same species as the cells to be cultured may be used.
- culture of human cells may be facilitated using HSA as a supplement, while media for the culture of bovine cells would instead use BSA.
- his approach runs the risks of introducing contaminants and adventitious pathogens into the culture medium (such as HIV or Hepatitis B virus from HSA preparations, or Bovine Spongiform E ⁇ cephalopathy virus from BSA preparations), which cun obviously negatively impact the use of such media in the preparation of animal and human therapeutics.
- the biotechnology industry anil government agencies are increasingly regulating, discouraging and even forbidding the use of cell culture media containing animal-derived products which may contain such pathogens.
- wheat peptides are known to bo toxic or to induce toxic effects in vitro and in vivo, particularly in the cells and tissues of the gastrointestinal systems of some mammals, including humans (Strober, W., et al., Ann. Int. Med. 55:242-256 (1975); Auricchto, S., et al., Pcdiatr. lies. 22(6) 103-H77 (1987)).
- extracts from certain plants, including wheat, barley, rye and oats have been shown to inhibit protein synthesis in cell-free systems derived from animal cells (Colcman, W.H., and Roberts, W.K..
- a scrum-frcc, low-prntein culture medium suitable for cultivation of animal cells, which is completely devoid of animal or human proteins.
- Such a medium formulation will facilitate the study of the effects of growth factors and other stimuli on cellular physiology, will allow easier and more cost-effective purification of biological substances produced by cultured animal cells in the biotechnology industry, and most importantly, will eliminate the risk of introduction of adventitious animal and human pathogens.
- the present invention provides such an animal cell culture medium formulation
- the present invention provides culture media formulations comprising non-animal or plant peptides that support the culture of animal cells, preferably as a primary protein source.
- the invention provides a cell culture medium capable of supporting the cultivation of an animal cell in vitro, wherein the medium comprises at least one plant peptide which is not derived from wheat and which is most preferably derived from rice.
- the media formulations of the invention comprise at least one non-animal or plant lipid and or fatty acid.
- the media formulations of the invention comprise at least one non-animal or plant peptide and at least " one non-animal or plant lipid and/or fatty acid.
- the invention also provides such media formulations which further comprise an enzymatic digest or extract of yeast cells.
- flic invention also provides a cell culture medium, capable of supporting the cultivation of an animal cell in vitro, comprising an extract of yeast cells, wherein the medium does not further comprise a wheat-derived plant peptide.
- the media of the present invention may be IX formulations, or may be concentrated as 10X- 100X, most preferably as 10X, 20X, 25X, 50X or 100X formulations.
- the basal medium of the present Invention comprises a number of ingredients, including a ino acids, vitamins, organic and inorganic salts, sugars, each ingredient being present in an amount which supports the cultivation of an animal cell in vitro.
- the medium of the invention may be used to culture a variety of animal cells, including insect cells (such as from the Spodoptera or Trichuplusa species), avion cells and mammalian cells (including primary cells, established cell lines, CHO cells, COS cells, VERO ceils, BHK cells and human cells).
- insect cells such as from the Spodoptera or Trichuplusa species
- avion cells including primary cells, established cell lines, CHO cells, COS cells, VERO ceils, BHK cells and human cells.
- the present invention also provides methods of culturing animal and human cells using the culture medium formulations disclosed herein, comprising die steps of (a) contac t ing an animal cell with the cell culture medium or the present invention; and (b) cultivating the animal ceil
- the present invention also relates to methods for replacing or substituting arumal-dcrived products with non-animal or plant-derived peptides, non-animal or plant lipids/fatty acids (or combinations thereoO, ⁇ nd/or enzymatic digests or extracts of yeast cells.
- Such non-a ⁇ ii ⁇ al/pla ⁇ t- dcrivcd products may be substituted for any number of animal-derived products, including but not limited to blood-derived products (e.g., serum, albumin, antibodies, fibrinogen, factor VHI, etc.), tissue or organ extracts and/or hydrolysates (e.g., bovine pituitary extract (BPE), bovine brain extract, chick embryo extract and bovine embryo extract), and animal-derived lipids and fatty acids, peptones, Excyte, slerols (e.g., cholesterol) and lipoproteins (e.g., high-density and low-density lipoproteins (HDLs and LDI ⁇ s, respectively)).
- blood-derived products e.g., serum, albumin, antibodies, fibrinogen, factor VHI, etc.
- tissue or organ extracts and/or hydrolysates e.g., bovine pituitary extract (BPE), bovine brain extract, chic
- compositions comprising the culture media of the present invention and an animal cell, including any of the animal cells described herein.
- FIGURE 1 shows the effect of Plant Powder on the growth of Hybridomu P1F6.
- Figure 2 shows the effect of Plant Powder on IgC production in Hybridoma P1F6.
- Figure 3 shows the effect of Plant Powder on specific productivity of Hybridoma P1F6.
- Cell culture refers to the maintenance of cells in an artificial, in vi t ro environment. It is to be understood, however, that the term “cell culture” is a generic term an d may be used to encompass the cultivation not only of individual cells, but also of tissues, organs, organ systems or whole organisms, for which the terms "tissue culture,” “organ culture,” “organ system culture” or “organotypic culture” may occasionally be used interchangeably with the term “cell culture.”
- culture refers to the maintenance of cells in vitro under conditions favoring growth, differentiation or continued viability, in an active or quiescent state, of the colls. In this sense, “cultivation” may be used interchangeably with “cell culture” or any of its synonyms described above. 5
- culture vessel refers to a glass, plastic, or metal container that can provide an aseptic environment for culturing cells.
- contacting refers to the placing ol cells to be cultivated in vitro into a culture vessel with the medium in which the cells are to be cultivated.
- the term “contacting” encompasses mixing cells with medium, pipetting medium onto cells in a culture vessel, and submerging cells in culture medium.
- combining refers to the mixing or admixing of ingredients in a cell culture medium formulation.
- cytokine refers to a compound that induces a physiological response in a cell, uch as growth, differentiation, senescence, apoplosis, cytotoxicity or antibody secretion. Included 5 in this definition of "cytokine” arc growth factors, interleuki ⁇ s, colony-stimulating factors, interferons and lymphokines.
- enzyme digest refers to a composition comprising a specialized type of extract, namely one prepared by treating the substance to he extracted (e.g., plant components or yeast cells) with at least one enzyme capable of breaking down the components of die substance into simpler -. J forms (e.g., into a preparation comprising mono- or disaccharid s and/or mono-, di- or tripeptides).
- hydrolysate may be us e d interchangeably with the term "enzymatic digest.”
- extract refers to a composition comprising a concentrated preparation of the components of a substance, typically formed by treatment of the substance cither mechanically (e.g., by pressure treatment) or chemically (e.g., by distillation, precipitation, enzymatic action or high salt treatment).
- lipid refers generally to water-insoluble organic molecules that can he extracted from cells and tissues by nonpolar solvents. Lipids are generally classified as complex lipids (which contain fatty acids) or simple lipids (which do not contain fatty acids).
- the complex lipids include acylglycerols, phosphoglyceridcs, sphingolipids and waxes.
- the simple lipids include terpcncs, steroids and prostaglandins. Tcrpc ⁇ es include compounds such as geraniol, limonene, menthol, pinene, camphor and carvone.
- Steroids include the subgroup "sterols,” which are steroid alcohols containing a hydroxyl group and a branched aliphatic chain of eight or more carbon atoms. See generally, Lchningcr, Albert L., Biochemistry, Worth Publishers, Inc. New York, pp. 279-306 (1975), which is herein incorporated by reference.
- the term "ingredient” refers to any compound, whether of chemical or biological origin, that can be used in cell culture media to maintain or promote the growth or proliferation of cells.
- component “nutrient” and “ingredient” can be used interchangeably and are all meant to refer to such compounds.
- Typical ingredients that are ⁇ sed in cell culture media include amino acids, salts, metals, sugars, lipids, nucleic acids, hormones, vitamins, fatty acids, proteins and the like. Other ingredients that promote or maintain cultivation of cells ex vivo can be selected by those of skill in the art in accordance with the particular need.
- non-animal derived refers to the origin of the compound, molecule, peptide, lipid, fatty acid, etc. of interest.
- non-animal sources may include chemical synthesis or synthetic preparations or isolation, preparation or purification of the compound, molecule, peptide, lipid, fatty acid, etc. of interest from bacteria, yeast fungi, and plants.
- a cell culture medium is composed of a number of ingredients and these ingredients vary from one culture medium to another.
- a "IX formulation” refers to any aqueous solution that contains some or all ingredients found in a cell culture medium at working concentrations.
- the "IX formulation” can refer to, for example, the cell culture medium or to any subgroup of ingredients for that medium.
- the concentration of an ingredient in a IX solution is about the same as the concen ⁇ tration of that ingredient found in a cell culture formulation used for maintaining or cultivating cells in vitro.
- a cell culture medium used for the in vitro cultivation of cells is a IX formulation by definition. When a number of ingredients are present, each ingredient in a IX formulation has a concentration about equal to the concentration of those ingredients in a cell culture medium.
- RPMl-1640 culture medium contains, among other ingredients, 0.2 g L L-arginine, 0.05 g/L L-asparagine, and 0.02 g L L-aspartic acid.
- a "IX formulation" of these amino acids contains about the same concentrations of these ingredients in solution.
- each ingredient in solution has the same or about the same concentration as that found in the cell culture medium being described.
- concentrations of ingredients in a IX formulation of cell culture medium are well known to those of ordinary skill in the art. See Allen R. Liss, Methods For Preparation of Media, Supplements and Substrate For Serum-Free Animal Cell Culture, N.Y. (1984), which is incorporated herein by reference in its entirety.
- the osmolarity and or pH may differ in a IX formulation compared to the culture medium, particularly when fewer ingredients are contained in the IX formulation.
- a "10X formulation” refers to a solution wherein each ingredient in that solution is about 10 times more concentrated than the same ingredient in the cell culture medium.
- a 10 formulation of RPMT-1 40 culture medium may contain, among other ingredients, 2.0 g L L- argininc, 0.5 g/L L-asparagine, and 0.2 g/L L-aspartic acid (compare IX formulation, above).
- a "10X formulation” may contain a number of additional ingredients at a concentration about 10 times that found in the IX culture medium.
- "20X formulation,” “25X formulation,” “50X formulation” and “100X formulation” designate solutions that contain ingredients at about 20-, 25-. 50- or 100- fold concentrations, respectively, as compared to a IX cell culture medium. Again, the osmolarity and pH of the media formulation and concentrated solution may vary.
- the cell culture media of the present invention are aqueous-based and comprise a number of ingredients in a solution of dcionizcd, distilled water to form a "basal media.” Any basal medium can be used in accordance with the present invention.
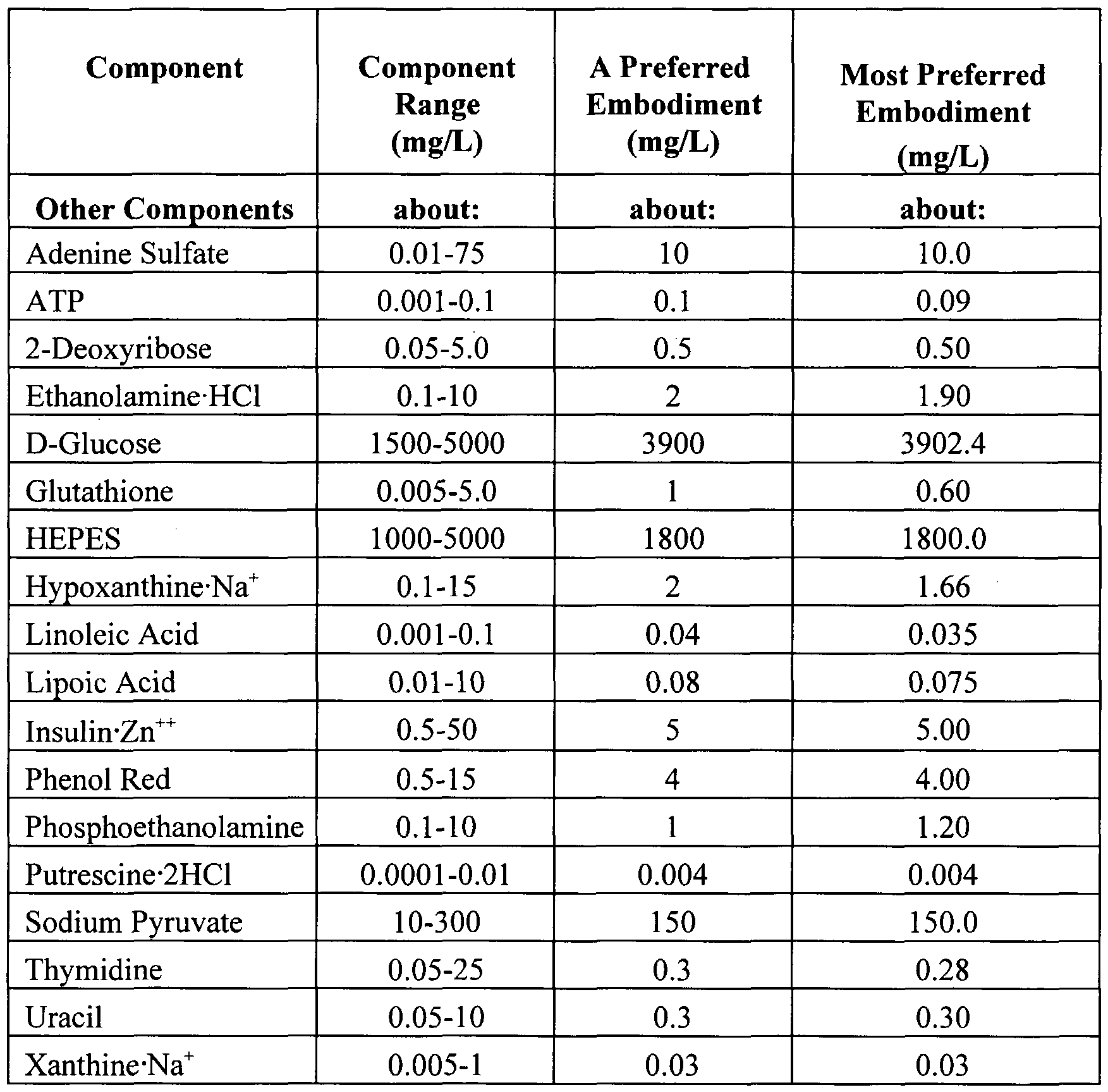
- the basal media of the present invention may - include the following ingredients: ami ⁇ o acids, vitamins, organic and/or inorganic salts, trace elements, buffering salts and sugars.
- the basal media of the invention comprise one or more amino acids, one or more vitamins, one or more inorganic salts, adcnine sulfate, ATP, one or more trace elements, dcoxyribose, ethanolami ⁇ e, D-gluco ⁇ c, glutathione, N-[2-hydroxycthyl]- piperazine-N'-[2-ethancsulfonic acid] (HEPES) or one or more other zwitterion buffers, hypoxanthinc, linoleic acid, lipoic acid, insulin, phenol red, phosphoethanolaminc, putrescine. sodium pyruvalc, thymidi ⁇ e, uracil and xanthine. Each of these ingredients may be obtained commercially, for example from Sigma (Saint Louis, Missouri).
- Amino acid ingredients which may bo included in the media of the present invention include
- L-alanine L-argini ⁇ , L-asparagine, L-aspartic acid, L-cysti ⁇ e, L-cystcinc, L-glutamic acid, L-gluiamine, glycine, L-histidinc, L-isolcucine, L-Ieuci ⁇ e, L-lysinc, L-mcthionine, L-phenylalanine,
- L-proline I ⁇ -serinc, L-thrconine, L-lryptophan, L-tyrosine and L-valinc.
- amino acids may be obtained commercially, for example from Sigma (Saint Louis, Missouri).
- Vitamin ingredients which may be included in the media of the present invention include ascorbic acid magnesium salt, biotin, choli ⁇ e chloride, D-Ca ⁇ -pantothenate, folic acid, Mnositol, menadione, niacinamidc, nicotinic acid, paraaminobenzoic acid (PABA), pyridoxal, pyridoxinc, rihoflavin, t iaminc-HCl, vitamin A acetate, vitamin B
- vitamins may be obtained commercially, for example from Sigma (Saint Louis, Missouri).
- Inorganic salt ingredients which may be used in the media of the present invention include CaCl 2 , KC1, MgCl 2 , MgSO 4 , NaCl, NaHCOj, Na z HP0 4 , a ⁇ PO ⁇ -IIzO and ferric citrate cheiate or ferrous sulfate chelatc. These inorganic salts may be obtained commercially, for example from Sigma (Saint Louis, Missouri).
- Trace elements which may be used in the media of the present invention include ions of barium, bromium, cobalt iodine, manganese, chromium, copper, nickel, selenium, vanadium, titanium, germanium, molybdenum, silicon, iron, fluorine, silver, rubidium, tin, zirconium, cadmium, zinc and aluminum. These ions may be provided, for example, in trace element salts such
- Cyt ⁇ ki ⁇ es which may be used in the media of the present invention include growth factors such as epidermal growth factor (EGF), acidic fibroblast growth factor (aFGF), basic fibroblast growth factor (bFGF), hcpatocyte growth factor (HGF), insulin-like growth factor 1 (IGF-I), insulin- like growth factor 2 (IGF-2), kcratinocyte growth factor (KGF), nerve growth factor (NGF), platelet- derived growth factor (PDGF), transforming growth factor beta (TGF- ⁇ ), vascular endotheiial cell growth factor (VKGF), transferrin, various i ⁇ terleukins (such as IL-1 through IL-18), various colony- stimulating factors (such as granulocyte/macrophage colony-stimulating factor (GM-CSF)), various intcrferons (such as IFN-v) and other cytokines having effects upon hcmatopoletic stem cells such as stem cell factor (SCF) and crythropoictin (
- cytokines may be obtained commercially, for example from Life Technologies, Tnc. (Rockville. Maryland) or R&D Systems (Minneapolis, Minnesota), and may bo citiier natural or rucoinbinant Most preferably, for culture of a wide variety of rruurimalian cells, the basal media will contain EGF at a concentration of about 0.1-100 nanograms/m i 11 i 1 i ter, preferably about 1-10 nanograms/milliliter, and most preferably about 5-10 nanograms per milliliter. Other cytokines, if used, may be added at concentrations that are determined empirically or as guided by the established cytokine art.
- Additional ingredients that may be included in the present media are insulin (especially as insulin»Zn ++ and transferrin. These additional ingredients, available commercially (for example, from Sigma, St. Louis, Missouri), may be formulated into the present media at the concentration ranges and preferred concentrations shown in Table 1.
- An iron salt or chclatc e.g., ferric citrate chclatc or ferrous sulfate
- rccombinant insulin or zinc based salts e.g., ZnCl etc.
- ZnCl zinc based salts
- basal medium When admixed together in solution, form a "basal medium.”
- Other basal media can be cquivalently used in accordance wi ⁇ i the present invention.
- at least one peptide, extract, enzymatic digest or hydrolysate of a non-animal or a plant and particularly of a plant protein, and/or at least one non-animal -derived or plant-derived lipid and/or fatty acid is added to the basal medium to formulate the complete culture media of the present invention.
- Plants suitable as sources of proteins, peptides, lipids and/or fatty acids in formulating the culture media of the present invention include, but are not limited to, rice, soy, potato, corn and aloe vera. Particularly preferred as a source of plant protein i.s rice.
- the use of wheat as a source of plant-derived proteins is specifically excluded from the present invention, as extracts and peptide preparations from wheat have been shown to contain inhibitors of protein synthesis in animal cell systems (Coleman, W.H., and Roberts, W.K., Biochim. Biophys.
- Non-animal or plant peptides for use in formulating the culture media of the present invention may be prepared by digesting non-animal or plant extracts with enzymes such as trypsin or chymotrypsin by methods that are routine in the art.
- enzymes such as trypsin or chymotrypsin
- peptides in the form of enzymatic digests or hydrolysates may be obtained commercially, for example from Quest International (Norwich, New York).
- Non-animal or plant peptides are added to the basal medium at a concentration of about 10-1000 mg/litcr, preferably about 50-500 mg/litcr, and most preferably about 100-200 mg liter.
- Non-animal or plant lipids fatty acids suitable for use in the present culture media may be obtained from any of the above-described plant sources and others that will be familiar to one of ordinary skill, and from bacteria, yeast and fungi, using methods of lipid/fatty acid isolation (for example, extraction, chromatography, particularly HPLC and the like) that arc well-known in the art
- plant as well as non-animal lipids/fatty acids and complexes of lipids and/or fatty acids may be obtained commercially, for example from Matreya. Inc. (Pleasant Gap, Pennsylvania) or Sigma (Saint Louis, Missouri).
- Fatty acids (or combinations thereof) for use in the invention include saturated and unsaturatcd fatty acids.
- Unsaturatcd fatty acids include monoenic acids, dienoic acids, and higher fatty acids (e.g., tri, tetra, penta and hexaenoic acids, etc.). See generally, Ixhningcr, supra.
- Particularly preferred lipids/fatty acids for use in the present culture media include, but are not limited to, palmitate, slearate. oleate. linoleate.
- plant-derived sterols known as phytosterols
- fungi- and yeast-derived sterols known as mycosterols
- mycosterols may be used as the lipid ingredient according to the present invention.
- non-animal derived sterols include, but are not limited to, brassicastcrol, campesterol, desmosterol, ergosterol, fucosterol, lanosterol, stigmastanol ( ⁇ -sitostcrol), sitosterol, stigmasterol and stigmasterol acetate, all of which arc commercially available, for example from Matreya, Inc. (Pleasant Gap, Pennsylvania) or Sigma (Saint Louis, Missouri).
- These lipids/fatty acids may be added individually or as mixtures comprising two or more of the above-described lipids/fatty acids, preferably in specific proportions as described in more detail below.
- non-animal or plant lipids/fatty acids are added to a basal medium a t concentrations of about 0.00001 to about 10,000 ⁇ g/ml, more preferably about 0.0001 to about j 1000 ⁇ g/ml, and most preferably ahout 0.001 to about 100 ⁇ g/ml.
- the basal medium including non-animal or plant-derived peptides and or non- animal or plant-derived lipids/fatty acids formulate complete culture media according to the present invention.
- These complete media are suitable for use in the culture of a variety of animal cells, as described in more detail below. It may be preferable, however, to further enrich the nutritional l ⁇ content of the complete media to support faster growth and enhanced production of biologicals by the cultured cells, and to provide a more suitable environment for the culture of fastidious animal cells. To accomplish such enrichment, one or more additional nutrients derived from non-animal sources may be added to the above-described basal or complete media.
- the additional nutrients added to the basal medium ⁇ or complete medium may comprise extracts of yeast cells (hereinafter “yeast extract” or “YE”), and most preferably ure ultrafiltered YE (hereinafter “yeast extract ultrafiltrate” or “YEU”).
- yeast extract extracts of yeast cells
- YEU ure ultrafiltered YE
- Such extracts may be prepared by methods generally known to those skilled in the art of bacteriological or animal cell culture medium formulation, or may be obtained commercially, for example from Sigma (Saint Louis, Missouri), Difco (Norwood, Massachusetts) or Quest International (Norwich, New :0 York).
- YE or YEU are added to the basal or complete media described above at concentrations of about 10-8000 mg/liter, preferably about 10-100 mg litcr, and most preferably about 50-100 mg/liter.
- YE or YEU may be added to the basal media at these concentrations, in the absence of whcat-dcrivcd plant peptides, enzymatic digests of animal proteins and peptones, to formulate a suitable animal cell culture medium according to the present invention.
- the medium ingredients can be dissolved in a liquid carrier or maintained in dry form.
- the pH of the medium should be adjusted to about 7.0-7.5, preferably about 7.1-7.4, and most preferably about 7.1-7.3.
- the osmolarity of the medium should also be adjusted to about 275-350 mOsm, preferably about 285-325 mOsm, and most preferably about 300-325 uiOsui.
- the 0 type of liquid carrier and the method used to dissolve the ingredients into solution vary and can be determined by one of ordinary skill in the art with no more than routine experimentation. Typically, the medium ingredients can be added in any order.
- the solutions comprising ingredients arc more concentrated than the concentration of the same ingredients in a IX media formulation.
- the ingredients can be 10- fold more concentrated (10X formulation), 20-fold more concentrated (20X formulation), 25-fold more concentrated (25X formulation), 50-fold more concentrated (50X concentration), or 100-fold more concentrated (100X formulation). More highly concentrated formulations can be made, provided that the ingredient! remain soluble and stable. Sec U.S. Patent No. 5,474,931, which is directed to methods of solubilizing culture media components at high concentrations. Tf the media ingredients arc prepared as separate concentrated solutions, an appropriate
- the diluent used Is water, but other solutions including aqueous buffers, aqueous saline solution, or other aqueous solutions may be used according to the invention.
- the culture media of the present invention are typically sterilized to prevent unwanted contamination. Sterilization may be accomplished, for example, by filtration through a low protein- binding membrane filter of about 0.1-1.0 ⁇ m pore size (available commercially, for example, from Milliporc, Bedford, Massachusetts) after admixing the concentrated ingredients to produce a sterile culture medium. Alternatively, concentrated subgroups of ingredients may be filter-stcrilizcd and stored as sterile solutions. These sterile concentrates can then be mixed under aseptic conditions with a sterile diluent to produce a concentrated IX sterile medium formulation. Autoclaving or other elevated temperature-based methods of sterilization are not favored, since many of the components of the present culture media arc heat labile and will be irreversibly degraded by temperatures such as those achieved during most heat sterilization methods.
- the optimal concentration ranges for the basal medium ingredients are listed in Table 1. These ingredients can be combined to form the basal animal cell culture medium which is then supplemented with cytokines, non-animal or plant peptides and optionally (but preferably) with YE, YEU and or one or more non-animal or plant lipids/fatty acids (or combinations thereof), to formulate the complete media of the present invention.
- cytokines non-animal or plant peptides
- YE, YEU optionally (but preferably) with YE, YEU and or one or more non-animal or plant lipids/fatty acids (or combinations thereof)
- concentration of a given ingredient can be increased or decreased beyond the range disclosed and the effect of the increased or decreased concentration can be determined using routine experimentation.
- the concentrations of the ingredients of the medium of the present invention are the concentrations listed in the far right column of Table 1, supplemented with cytokines, non-animal or plant peptides and YE, YEU and/or one or more non- animal or plant lipids/fatty acids as described above.
- each of the components of the culture medium may react with one or more other components in the solution.
- the present invention encompasses the formulations disclosed in Table 1, supplemented as described above, as well as any reaction mixture which forms after these ingredients arc combinod.
- the optimization of the present media formulations was carried out using approaches 0 described by Ham (Ham, R.G., Metlwd for Preparation of Media, Supplements and Substrata for Serum-Free Animal Culture, Alan R. Liss, Inc., New York, pp. 3-21 (1984)) and Waymouth (Waymouth, C, Methods for Preparation of Media, Supplements and Substrata fur Serum-Free Animal Culture, Alan R. Liss, Inc., New York, pp. 23-68 (1984)).
- the optimal final concentrations for medium ingredients are typically identified either by empirical studies, in single component (5 titration tudies, or by interpretation of historical and current scientific literature. In single component titration studies, using animal cells, the concentration of a single medium component i.s varied while all other constituents and variables arc kept constant and the effect of the single component on viability, growth or continued health of the animal cells is measured.
- the present invention also relates to methods for replacing or substituting animal-derived 20 products with non-animal or plant peptides, non-animal or plant lipids and/or fatty acids, and/or enzymatic digests or extracts of yeast cells (or combinations thereof).
- Such non-animal-, plant- and or yeast-derived nutrients may be substituted for any number of animal-derived culture medium components or sub tituents, including but not limited to blood-derived products, tissue organ/gland extracts, animal-derived fatty acids and lipids, sterols, and lipoproteins.
- blood-derived 25 products and tissue/organ extracts are substituted in the culture media of the invention using one or more of the above-described non-animal or plant-derived peptides, while animal-derived fatty acids/lipids, sterols and lipoproteins arc preferably substituted with one or more of the above- described non-animal or plant-derived lipids fatty acids.
- Typical blood-derived products that may be replaced in accordance with this aspect of the invention include but are not limited to serum (e.g., 2D fetal bovine serum and calf serum, human scrum, etc.), plasma, albumin (e.g., bovine scrum albumin or human serutn albumin), antibodies, fibrinogen, factor VIU, etc.
- Typical tissuc/organ gland extracts that may be replaced in accordance with this aspect of the invention include but are not limited to bovine pituitary extract (BPE), bovine brain extract, chicken embryo extract and bovine embryo extract.
- BPE bovine pituitary extract
- bovine brain extract bovine brain extract
- chicken embryo extract bovine embryo extract
- bovine embryo extract any animal-derived fatty acid or lipid, including saturated and unsaturated fatly acids lipids that are well-known in the art, may be replaced with one or more of the above-described non-animal or plant-derived lipids/fatty acids.
- animal- derived sterols e.g., cholesterol
- lipoproteins e.g., high- and low-density lipoproteins (HDLs and LDLs, respectively)
- HDLs and LDLs high- and low-density lipoproteins
- animal-derived medium components which may be replaced by one or more non-animal or plant-derived nutrients in accordance with the invention can be easily determined by one of ordinary skill in the art by substituting one or more non-animal or plant lipids/fatty acids, non-animal or plant peptides and/or extracts/digests of yeast (or combinations thereof) and testing the effect of such substitution on cell growth by methods that will be familiar to the ordinarily skilled artisan (such as those methods described in the examples below).
- the present invention further relates to a kit for replacing one or more ariimal-derived ingredients in a cell culture medium, wherein the kit comprises one or more non-ariimal or plant derived peptides and/or one or more non-animal or plant-derived lipids or fatty acids or combinations thereof.
- Cells which can be cultivated in the medium of the present invention are those of animal origin, including but not limited to cells obtained from mammals, birds (avian), insects or fish.
- Mtujimalian cells particularly suitable for cultivation in the present media include those of human origin, which may be primary cells derived from a tissue sample, diploid cell strains, transformed cells or established cell lines (e.g., HeLa), each of which may optionally be diseased or genetically altered.
- Other miunmalian cells uch as hybridomas, CIIO cells, COS cells, VERO cells, IleLa cells, 293 cells, PER-C6 cells, K562 cells, MOLT-4 cells, Ml cells, NS-1 cells.
- COS-7 ceils are also suitable for cultivation in the present media.
- stem cells and cells used in in vitro virus production may be cultivated in the media of the present invention.
- Insect cells particularly suitable for cultivation in the present media include those derived from Spodoptera species (e.g., Sf9 or S/21, derived from Spodopteru frugiperdd) or richoplusa species (e.g., HIGH FIVETM or MG1, derived from Trichoplusa ni).
- Tissues, organs, organ systems and organisms derived from animals or constructed in vitro or in vivo using methods routine in the art may similarly be cultivated in the culture media of the present invention.
- Animal cells for cultiiring by the present invention may be obtained commercially, for example from ATCC (Rockville, Maryland), Cell Systems, Inc. (Kirkland, Washington) or Invitrogcn Corporation (San Diego, California). Alternatively, cells may be isolated directly from samples of animal tissue obtained via biopsy, autopsy, donation or other surgical or medical procedure.
- Tissue should be handled using standard sterile technique and a laminar flow safety cabinet. In the use and p ocessing of all human tissue, the recommendations of the U.S. Department of Health and Human Services Centers for Disease Control and Prevention should be followed (Bios fety in Microbiological tmd Binmedical Laboratories, Richmond, J.Y. et al, Eds., U.S. Government Printing Office, Washington, D.C. 3rd Edition (1993)). The tissue should be cut into small pieces 1 (e.g., 0.5 x 0.5 cm) using sterile urgical instruments.
- the small pieces should be washed twice with sterile saline solution upplemented with antibiotics as above, and then may be optionally treated with an enzymatic solution (e.g., collagenase or trypsin solutions, each available commercially, for example, from Lire Technologies, Inc., Rockville, Maryland) to promote dissociation of cells from the tissue matrix.
- an enzymatic solution e.g., collagenase or trypsin solutions, each available commercially, for example, from Lire Technologies, Inc., Rockville, Maryland
- the mixture of dissociated cells and matrix molecules are washed twice with a suitable physiological saline or tissue culture medium (e.g., Dulbccco' Phosphate Buffered Saline without calcium and magnesium). Between washes, the cells arc ccntrifugcd (e.g., at 200 x g) and then re- suspcnded in serum-free tissue culture medium. Aliquots are counted using an electronic cell counter (such as a Coulter Counter). Alternatively, the cells can be counted manually using a hcmocytometer.
- tissue culture medium e.g., Dulbccco' Phosphate Buffered Saline without calcium and magnesium.
- the isolated cells can be plated according to the experimental conditions determined by the investigator.
- the examples below demonstrate at least one functional set of culture conditions useful for cultivation of certain mammalian cells. It is to be understood, however, that the optimal plating Z and culture conditions for a given animal cell type can be determined by one of ordinary skill in the art using only routine experimentation. For routine culture conditions, using the present invention, cells can be plated onto the surface of culture vessels without attachment factors.
- the vessels can be pre-coatcd with natural, recombinant or synthetic attachment factors or peptide fragments (e.g., collagen or ribronectin, or natural or ynthetic fragments thereof)- Isolated ceils can 0 also be seeded into or onto a natural or syntiietie three-dimensional support matrix such as a preformed collagen gel or a synthetic biopolymeric material, or onto feeder layers of cells.
- attachment factors or a support matrix with the medium of the present invention will enhance cultivation of many attachment-dependent cells In the absence of serum supplementation.
- the cell seeding densities for each experimental condition can be optimized for the specific culture conditions being used. For routine culture in plastic culture vessels, an initial seeding density of O. l-l.O x 10 % cells per cm z or about 1.5X the plating concentration routinely used for the ame cells in serum supplemented media i.s preferable.
- Mammalian cells arc typically cultivated in a cell incubator al about 37"C, while the optimal temperatures for cultivation of avian, nematodc and insect cells are typically somewhat lower and arc 2 0 well-known to those of ordinary skill in the an.
- the incubator atmosphere should be humidified for cultivation of animal cells, and should contain about 3-10% carbon dioxide in air.
- Culture medium pH should be in the range of about 7.1-7.6, preferably about 7.1-7.4, and most preferably about 7-1- 7.3.
- Cells in closed or batch culture should undergo complete medium exchange (i.e., replacing 2 5 spent media with fresh media) about every 2-3 days, with more or less frequently as required by the specific cell type.
- Cells in pcrfusion culture e.g., in bioreactors or fe ⁇ nentcrs
- the cell culture media of the present invention may also be used to produce cell culture composiiion.s comprising the present media and one or more animal cells.
- Animal cells preferably used in such compositions include, but arc not limited to, cells obtained from mammals, birds (avian), insects or fish.
- Mammalian cells particularly suitable for use in such compositions include those of human origin, which may be primary cells derived from a tissue sample, diploid cell trains, transformed cells or established cell lines (e.g., HeLa), each of which may optionally be diseased or genetically altered.
- mammalian cells such as hybridomas, CHO cells, COS cells, VERO cells, HeLa cells, 293 cells, PER-C6 cells, K562 cells, MOLT-4 cells, Ml cells, NS-1 cells, COS-7 cells, MDBK cells, MDCK cells.
- MRC-5 cells, WI-38 cells, SP2/0 cells, BHK cells (including BHK-21 cells) and derivatives thereof, are also suitable for use in forming the cell culture compositions of the present invention.
- Insect cells particularly suitable for use in forming such compositions include those derived from Sp ⁇ d ⁇ ptera species (e.g., SJ or SJ2I, derived from Spad ⁇ ptcra frugiperda) or Trichoplusa species (e.g., HIGH FIVETM or MG1, derived from Trichoplusa nt). Tissues, organs, organ systems and organisms derived from animals or constructed in vitro or in vivn using methods routine in the art may similarly be used t ⁇ form the cell culture compositions of the present invention. These cell culture compositions may be used in a variety of medical (including diagnostic and therapeutic), industrial, forensic and research applications requiring ready-to-use cultures of " animal cells in scrum-free media.
- Sp ⁇ d ⁇ ptera species e.g., SJ or SJ2I, derived from Spad ⁇ ptcra frugiperda
- Trichoplusa species e.g., HIGH FIVETM or MG1, derived from Trichoplusa nt.
- VERO cultures ATCC were plated in 25 cm 2 ceil culture flasks in duplicate in each medium at about 2.5 x l ⁇ ceils per flask in 5 ml of medium. No attachment factors or coating of the plastic surface are required for the culture of VERO cells. At 3 to 4 days the cells were removed using standard cell culture techniques. The surface of the culture was first washed with Dulbecco's Phosphate Buffered Saline (DPBS) and then 1.0 ml Trypsin-EDTA (Life Technologies, Inc., Rockville, Maryland) was added.
- DPBS Dulbecco's Phosphate Buffered Saline
- the digest was allowed to sit on the cell surface for 3 to 5 minutes or until the cells rounded up and began to detach from the surface of the flask.
- the cells were completely detached by vigorous agitation against the palm of the hand and then 1.5 ml of soybean trypsin inhibitor was added to quickly neutralize enzymatic activity.
- the cells were counted under the microscope using trypan blue straining solution and new cultures plated at 2.5 x 10 5 cells per 25 cm 2 flask. Incubation was at 37°C in 5% CO 2 in air.
- the cultures were passaged for a total of 4 subcultures and the mean cells per subculture determined from the counts of the final 3 subcultures (P2 + P3 + P4 + 3).
- ddH 2 O deionizcd water
- trace elements from lOOOx stock
- L-alaninc (0.22 g)
- L-argini ⁇ e-HCl 9.75 g
- L-asparagine-IICl (1.02 g)
- L-aspartic acid 0.332 g
- L-cysteine-HCI-H 2 O 0.10 g
- L-cystine-HCl (2.87 g)
- glycine (0.188 g)
- L-glutamic acid 0.268 g
- L-histidine-HCl-H 2 O (1.707 g), (4284 g)
- L-leuci ⁇ e (4.511 g)
- L-lysine-HCl 5.640 g)
- L-methionine 1.266
- the solution was gently mixed by magnetic stirring for about 15 minutes.
- the pH of the solution was then adjusted to about 5.50 by adding a sufficient volume (about 20-25 iid) of 5N NaOH.
- NaH*PO rt (7.494 g), NaiHPO 4 (1.175 g) and ascorbic acid Mg salt (1.25 g) were added and the solution was again gently mixed for about 15 minutes.
- the pH was then adjusted to about 6.5 withS aOH.
- ATP (0.025 g), uracil (0.0075 g), PABA (0.0012 g), D-Ca ⁇ -pantothcnatc (0.05 g), riboflavin (0.005 g), NaCi ( 142.50 g), CaCl 2 (3.00 g). MgCl 2 (3.125 g) and EGF (0.00025 g) were then added.
- the solution was again gently mixed for about 15 minules, during which lime a 20 nil volume of absolute ethanol was obtained, to which were added vitamin A acetate (0.0035 g), vitamin D2 (0.0025 g), menadio ⁇ e (0.00025 g), lipoic acid (0.0019 g) and linoleic acid (0.00088 g). After allowing the compounds to dissolve in the ethanol, the ethanol solution was added to the 20 liter medium solution from above, and the medium solution was gently mixed for about 5 minutes.
- Biotin (0.0019 g), folic acid (0.05 g), hypoxanthinc-Na (0.0415 g), xanthine-Na (0.0075 g) and insulin-Zn ++ (0.125 g) were added to a 20 ml volume of ddH 2 O. After allowing the compounds to dissolve in the water, this water solution was added to the 20 liter medium solution from above, and the medium solution was gently mixed for about 5 minutes. The pH r the solution was then adjusted with 5N HC1 or 5N NaOH to about 7.15 ⁇ 0.50.
- VERO cells were cultured in the various medium formulations, and cell counts determined, as described above, and were compared to those obtained in basal medium that was unsupplemented (negative control) or supplemented with 500 mg/litcr human scrum albumin (HSA; positive control). Results shown in Table 2 demonstrate mean cell count per 25 cm 7 flask over 3 subcultures and relative growth efficiency (RGE) for each or the medium formulations. RGE was calculated by dividing the mean cell count for a given medium formulation by that for the HSA control.
- yeast extract as a supplement in the present media preparations of YE or an ultrafiltrate of YE ("YEU") were supplemented into basal media in the presence or absence of 100 mg liler of rice hydrolysate. These media were then used to examine VERO cell growth as described above, Cell counts were compared to those obtained in EMEM supplemented with 5% FBS (positive control). Results shown in Table 5 demonstrate mean cell count and relative growth efficiency (RGE) for each of the medium formulations. RGE was calculated as described in Example 2.
- - rice indicates medium not supplemented with 100 mg/L rice hydrolysate.
- + rice indicates medium supplemented with 100 mg/L rice hydrolysate.
- Example 2 To examine the effect of growth factor concentration on the performance of the culture media, recombinant human EGF was added to the basal media of Example 1 at various concentrations. These media were then used to examine VERO cell growth as described above. Cell counts were compared to those obtained in basal medium that was unsupplemented (negative control) or to EMEM supplemented with 5% FBS (positive control). Results shown in Table 6 demonstrate mean cell count and relative growth efficiency (RGE) for each of the EGF concentrations. RGE was calculated as described in Example 2.
- an optimal culture medium formulation for supporting the cultivation of animal cells is the basal medium formulation shown in Table 1, supplemented with EGF at 5-10 mg/liter, yeast extract (preferably yeast extract ultrafiltrate) at 50-100 mg/liter, and at least one plant peptide (preferably rice peptides or hydrolysate) at 100-200 mg/liter.
- Table 9 shows the use of the medium to grow BHK-21 cells in suspension on a shaker platform.
- 0.2% PLURON1C F68 was added to both the test medium and the EMEM FBS 5% control to reduce shear damage. Counts were made at 72, 96 and 120 hours. As can be scon in Table 9 the test medium performed as well as or better than the control. By 120 hrs the viability began dropping much more rapidly in the control scrum-supplemented medium as compared to this preferred embodiment of the present invention.
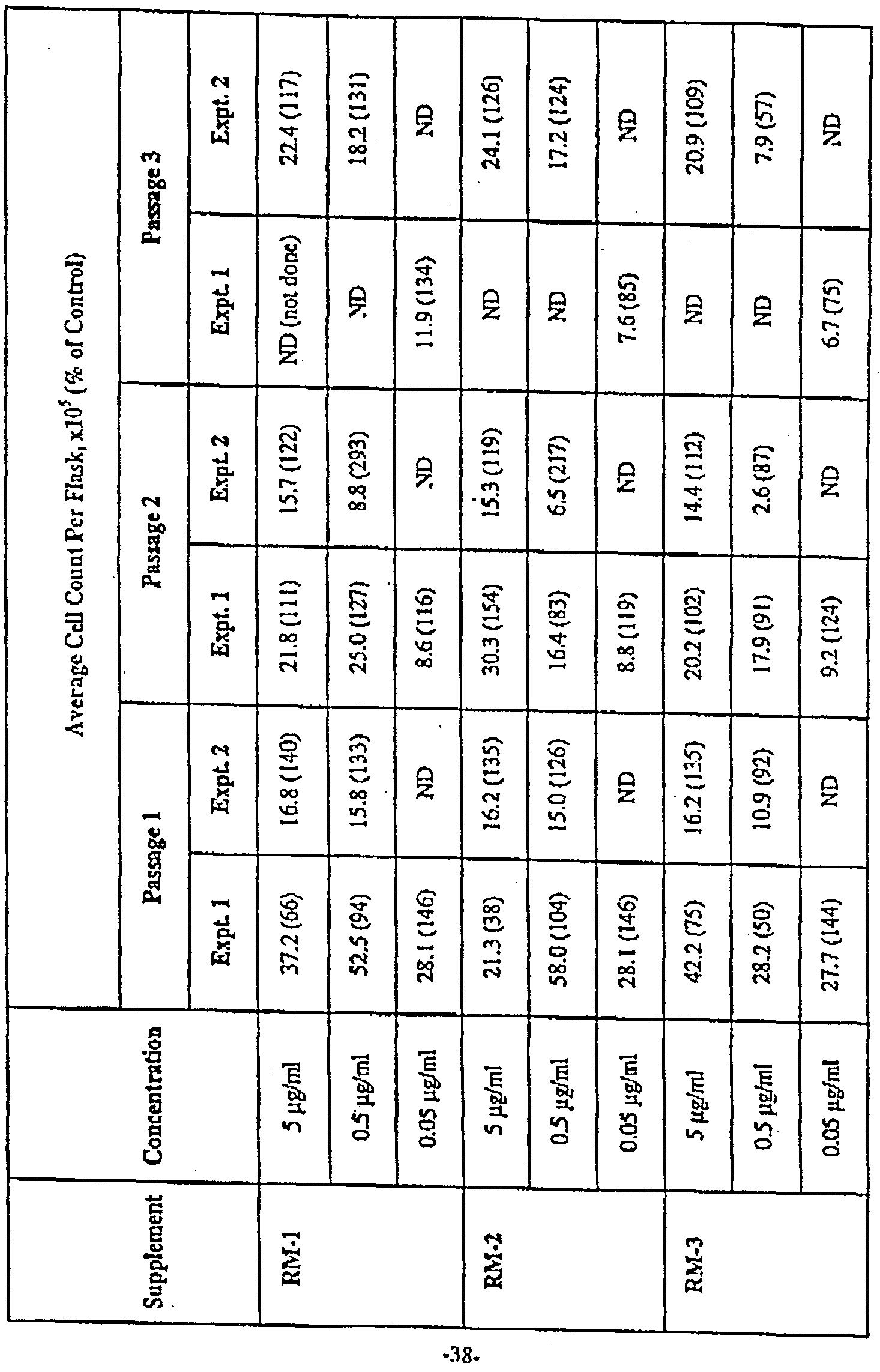
- culture media made as described in Example 1 were used to culture VERO cells.
- Cells were plated in T-25 flasks into culture media that were not supplemented with plant-derived lipids or fatty acids (control) or that were supplemented with 5 ⁇ g ml, 0.5 ⁇ g/ml or 0.05 ⁇ g/ml of one of the following plant lipid or fatty acid mixes obtained from Matreya Inc. (Pleasant Gap, Pennsylvania), having constituents present in the indicated percentages:
- RM-1 palmitate (6.0%), stcarate (3.0%), oleate (35.0%), linoleate (50.0%), linolenate (3.0%), arachidate (3.0%)
- RM-2 palmitate (7.0%), stcarate (5.0%), oleate (18.0%), linoleate (36.0%), linolenate (34.0%)
- RM-3 myrlstate (11.0%), palmitate (4.0%), siearate (3.0%), oleate (45.0%), linoleate (15.0%), linolenate (3.0%), arachidate (3.0%), behenatc (3.0%), erucate (20.0%), lignocer te (3.0%)
- RM-5 caprylate (7.0%), caprate (5.0%), laurate (48.0%), myristatc (15.0%), palmitate (7.0%), stcarate (3.0%), oleate (12.0%), linoleate (3.0%)
- RM-6 myristate (2.0%), palmitate (30.0%), palmitolcatc (3.0%), stcarate (14.0%), oleate ( 1 .0%), linoleate (7.0%), linolenate (3.0%)
- the flasks were placed in a 37°C incubator in a humidified atmosphere of 8% C ⁇ 2 in air and subcultured every 2 to 3 days for 3 passages.
- replicate T-75 flasks were set up and samples were taken on days 3, 4 and 5 post planting for determination of total cell density, viable cell density and IgG expression.
- the samples that were used for IgG determination were oentrifuged and the supcrnatants stored al -20°C until assayed. IgG levels were estimated by ELISA using one sample from each of the replicate samples.
- the Plant Powder had a positive effect on viable cell density in a dose- depcndcnt manner. Additionally, IgG expression was also improved by addition of the Plant Powder in a dose-dependent manner as seen in Figure 2. Furthermore, as seen in Figure 3, the specific productivity ( ⁇ g IgG/10 6 total cells) was slightly increased in the Plant Powder cultures, with the effect most pronounced on day 5 post planting.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Cell Biology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
Claims
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US7080798A | 1998-05-01 | 1998-05-01 | |
| US09/070,807 | 1998-05-01 | ||
| US30295399A | 1999-04-30 | 1999-04-30 | |
| US09/302,953 | 1999-04-30 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO1999057246A1 true WO1999057246A1 (en) | 1999-11-11 |
| WO1999057246A8 WO1999057246A8 (en) | 2000-10-12 |
| WO1999057246A9 WO1999057246A9 (en) | 2001-06-21 |
Family
ID=26751514
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1999/009574 Ceased WO1999057246A1 (en) | 1998-05-01 | 1999-05-03 | Animal cell culture media comprising non-animal or plant-derived nutrients |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO1999057246A1 (en) |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1181865A1 (en) * | 2000-08-23 | 2002-02-27 | Universite Catholique De Louvain | Cryoprotective solutions |
| WO2001066560A3 (en) * | 2000-03-07 | 2002-03-28 | Forbes Medi Tech Inc | Novel derivatives comprising phytosterols and/or phytostanols and alpha-lipoic and use thereof in treating or preventing cardiovascular disease, its underlying conditions and other disorders |
| WO2004078955A1 (en) * | 2003-03-03 | 2004-09-16 | Glaxosmithkline Biologicals S.A. | Animal-free cell culture method |
| FR2879214A1 (en) * | 2004-12-14 | 2006-06-16 | Pierre Fabre Medicament Sa | PEPTIDE FRACTIONS PROMOTING THE GROWTH AND SYNTHESIS OF PRODUCT (S) OF INTEREST IN CELL CULTURE AND / OR TISSUE |
| NL1029059C2 (en) * | 2005-05-17 | 2006-11-20 | Noord Nl Oliemolen Holding B V | Peptide preparation for growing and / or growing microorganisms and / or cells. |
| EP2064314A4 (en) * | 2006-09-13 | 2009-12-30 | Abbott Lab | Cell culture improvements |
| US7754484B2 (en) * | 2005-11-16 | 2010-07-13 | Cognis Ip Management Gmbh | Use of esters of unsaturated, physiologically active fatty acids as nutrient media for cell cultures |
| US7955833B2 (en) | 2002-07-09 | 2011-06-07 | Baxter International Inc. | Animal protein free media for cultivation of cells |
| US8021881B2 (en) | 1999-09-28 | 2011-09-20 | Baxter Innovations Gmbh | Medium for the protein-free and serum-free cultivation of cells |
| US8080414B2 (en) | 1997-06-20 | 2011-12-20 | Baxter Innovations Gmbh | Recombinant cell clones having increased stability and methods of making and using the same |
| EP2500416A1 (en) * | 2006-09-13 | 2012-09-19 | Abbott Laboratories | Cell culture improvements |
| US8440408B2 (en) | 2004-10-29 | 2013-05-14 | Baxter International Inc. | Animal protein-free media for cultivation of cells |
| US8911964B2 (en) | 2006-09-13 | 2014-12-16 | Abbvie Inc. | Fed-batch method of making human anti-TNF-alpha antibody |
| US8921526B2 (en) | 2013-03-14 | 2014-12-30 | Abbvie, Inc. | Mutated anti-TNFα antibodies and methods of their use |
| US8946395B1 (en) | 2013-10-18 | 2015-02-03 | Abbvie Inc. | Purification of proteins using hydrophobic interaction chromatography |
| US9017687B1 (en) | 2013-10-18 | 2015-04-28 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same using displacement chromatography |
| US9062106B2 (en) | 2011-04-27 | 2015-06-23 | Abbvie Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| US9067990B2 (en) | 2013-03-14 | 2015-06-30 | Abbvie, Inc. | Protein purification using displacement chromatography |
| US9085618B2 (en) | 2013-10-18 | 2015-07-21 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US9150645B2 (en) | 2012-04-20 | 2015-10-06 | Abbvie, Inc. | Cell culture methods to reduce acidic species |
| US9181572B2 (en) | 2012-04-20 | 2015-11-10 | Abbvie, Inc. | Methods to modulate lysine variant distribution |
| US9181337B2 (en) | 2013-10-18 | 2015-11-10 | Abbvie, Inc. | Modulated lysine variant species compositions and methods for producing and using the same |
| US9193787B2 (en) | 2012-04-20 | 2015-11-24 | Abbvie Inc. | Human antibodies that bind human TNF-alpha and methods of preparing the same |
| US9206390B2 (en) | 2012-09-02 | 2015-12-08 | Abbvie, Inc. | Methods to control protein heterogeneity |
| US9234033B2 (en) | 2012-09-02 | 2016-01-12 | Abbvie, Inc. | Methods to control protein heterogeneity |
| US9249182B2 (en) | 2012-05-24 | 2016-02-02 | Abbvie, Inc. | Purification of antibodies using hydrophobic interaction chromatography |
| US9499614B2 (en) | 2013-03-14 | 2016-11-22 | Abbvie Inc. | Methods for modulating protein glycosylation profiles of recombinant protein therapeutics using monosaccharides and oligosaccharides |
| US9550826B2 (en) | 2013-11-15 | 2017-01-24 | Abbvie Inc. | Glycoengineered binding protein compositions |
| US9598667B2 (en) | 2013-10-04 | 2017-03-21 | Abbvie Inc. | Use of metal ions for modulation of protein glycosylation profiles of recombinant proteins |
| US9758568B2 (en) | 2006-01-04 | 2017-09-12 | Baxalta GmbH | Oligopeptide-free cell culture media |
| CN110257325A (en) * | 2019-05-31 | 2019-09-20 | 苏州智享众创孵化管理有限公司 | A method of the in vitro culture self-healing cell based on aloe extract |
| WO2020127941A2 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Serum free intracellular pathogen vaccine |
| WO2021123099A1 (en) | 2019-12-20 | 2021-06-24 | Intervet International B.V. | Reassorted isa virus |
| US20220186191A1 (en) * | 2019-01-17 | 2022-06-16 | Boehringer Ingelheim Animal Health USA Inc. | Serum-free medium for avian vaccine production and uses thereof |
| US12349704B2 (en) | 2018-11-15 | 2025-07-08 | Aleph Farms Ltd. | High quality cultured meat, compositions and methods for producing same |
| US12408684B2 (en) | 2019-12-31 | 2025-09-09 | Air Protein, Inc. | High protein food compositions |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5607852A (en) * | 1992-06-04 | 1997-03-04 | Merch & Co., Inc. | Process for attenuated varicella zoster virus vaccine production |
| US5633162A (en) * | 1990-10-17 | 1997-05-27 | Glaxo Wellcome Inc. | Method for culturing Chinese hamster ovary cells |
-
1999
- 1999-05-03 WO PCT/US1999/009574 patent/WO1999057246A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5633162A (en) * | 1990-10-17 | 1997-05-27 | Glaxo Wellcome Inc. | Method for culturing Chinese hamster ovary cells |
| US5607852A (en) * | 1992-06-04 | 1997-03-04 | Merch & Co., Inc. | Process for attenuated varicella zoster virus vaccine production |
Non-Patent Citations (5)
| Title |
|---|
| AURICCHIO S, ET AL.: "PREVENTION BY MANNAN AND OTHER SUGARS OF IN VITRO DAMAGE OF RAT FETAL SMALL INTESTINE INDUCED BY CEREAL PROLAMIN PEPTIDES TOXIC FORHUMAN CELIAC INTESTINE", PEDIATRIC RESEARCH, LIPPINCOTT WILLIAMS & WILKINS, NEW YORK, US, vol. 22, no. 06, 1 January 1987 (1987-01-01), US, pages 703 - 707, XP002919505, ISSN: 0031-3998 * |
| COLEMAN W H, ROBERTS W K: "INHIBITORS OF ANIMAL CELL-FREE PROTEIN SYNTHESIS FROM GRAINS", BIOCHIMICA ET BIOPHYSICA ACTA., ELSEVIER, NL, vol. 696, 1 January 1982 (1982-01-01), NL, pages 239 - 244, XP002919508, ISSN: 0006-3002 * |
| FRESHNEY R. I.: "CULTURE OF ANIMAL CELLS. MANUAL OF BASIC TECHNIQUE.", 1 January 1987, NEW YORK, ALAN R. LISS., US, article FRESHNEY R I: "CULTURE OF ANIMAL CELLS A MANUAL OF BASIC TECHNIQUE", pages: 74 - 84 + A, XP002919507, 015492 * |
| KEAY L: "AUTOCLAVABLE LOW COST SERUM-FREE CELL CULTURE MEDIA. THE GROWTH OF L CELLS AND BHK CELLS ON PEPTONES", BIOTECHNOLOGY AND BIOENGINEERING., WILEY & SONS, HOBOKEN, NJ., US, vol. 17, 1 January 1975 (1975-01-01), US, pages 745 - 764, XP002919506, ISSN: 0006-3592, DOI: 10.1002/bit.260170510 * |
| MURAKAMI H, MASUI H, SATO G H: "SUSPENSION CULTURE OF HYBRIDOMA CELLS IN SERUM-FREE MEDIUM: SOYBEANPHOSPHOLIPIDS AS THE ESSENTIAL COMPONENTS", COLD SPRING HARBOR MONOGRAPH SERIES., COLD SPRING HARBOR LABORATORY, COLD SPRING HARBOR, NY., US, vol. 09, 1 January 1982 (1982-01-01), US, pages 711 - 715, XP002919509, ISSN: 0270-1847 * |
Cited By (101)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8080414B2 (en) | 1997-06-20 | 2011-12-20 | Baxter Innovations Gmbh | Recombinant cell clones having increased stability and methods of making and using the same |
| USRE46745E1 (en) | 1997-06-20 | 2018-03-06 | Baxalta Incorporated | Recombinant cell clones having increased stability and methods of making and using the same |
| USRE46860E1 (en) | 1997-06-20 | 2018-05-22 | Baxalta Incorporated | Recombinant cell clones having increased stability and methods of making and using the same |
| USRE46897E1 (en) | 1997-06-20 | 2018-06-19 | Baxalta Incorporated | Recombinant cell clones having increased stability and methods of making and using the same |
| US8329465B2 (en) | 1997-06-20 | 2012-12-11 | Baxter Innovations Gmbh | Recombinant cell clones having increased stability and methods of making and using the same |
| US8084251B2 (en) | 1997-06-20 | 2011-12-27 | Baxter Innovations Gmbh | Recombinant cell clones having increased stability and methods of making and using the same |
| US8084252B2 (en) | 1997-06-20 | 2011-12-27 | Baxter Innovations Gmbh | Recombinant cell clones having increased stability and methods of making and using the same |
| US9982286B2 (en) | 1999-09-28 | 2018-05-29 | Baxalta Incorporated | Medium for the protein-free and serum-free cultivation of cells |
| US9441203B2 (en) | 1999-09-28 | 2016-09-13 | Baxalta Innovations Gmbh | Medium for the protein-free and serum-free cultivation of cells |
| US8722406B2 (en) | 1999-09-28 | 2014-05-13 | Baxter Innovations Gmbh | Medium for the protein-free and serum-free cultivation of cells |
| US8021881B2 (en) | 1999-09-28 | 2011-09-20 | Baxter Innovations Gmbh | Medium for the protein-free and serum-free cultivation of cells |
| WO2001066560A3 (en) * | 2000-03-07 | 2002-03-28 | Forbes Medi Tech Inc | Novel derivatives comprising phytosterols and/or phytostanols and alpha-lipoic and use thereof in treating or preventing cardiovascular disease, its underlying conditions and other disorders |
| EP1181865A1 (en) * | 2000-08-23 | 2002-02-27 | Universite Catholique De Louvain | Cryoprotective solutions |
| US8524497B2 (en) | 2002-07-09 | 2013-09-03 | Baxter International Inc. | Animal protein free media for cultivation of cells |
| US9163211B2 (en) | 2002-07-09 | 2015-10-20 | Baxter International Inc. | Animal protein free media for cultivation of cells |
| US7955833B2 (en) | 2002-07-09 | 2011-06-07 | Baxter International Inc. | Animal protein free media for cultivation of cells |
| AU2004217807B2 (en) * | 2003-03-03 | 2009-06-25 | Glaxosmithkline Biologicals S.A. | Animal-free cell culture method |
| US8034617B2 (en) | 2003-03-03 | 2011-10-11 | Glaxosmithkline Biologicals S.A. | Cell culture in culture media free of components of animal origin |
| CN1756837B (en) * | 2003-03-03 | 2010-05-26 | 葛兰素史密丝克莱恩生物有限公司 | Animal cell-free culture method |
| EP2192175A1 (en) * | 2003-03-03 | 2010-06-02 | GlaxoSmithKline Biologicals S.A. | Animal-free cell culture method |
| EP2186885A1 (en) * | 2003-03-03 | 2010-05-19 | GlaxoSmithKline Biologicals S.A. | Animal-free cell culture method |
| WO2004078955A1 (en) * | 2003-03-03 | 2004-09-16 | Glaxosmithkline Biologicals S.A. | Animal-free cell culture method |
| US8748156B2 (en) | 2004-10-29 | 2014-06-10 | Baxter International Inc. | Animal protein-free media for cultivation of cells |
| US9809796B2 (en) | 2004-10-29 | 2017-11-07 | Baxalta GmbH | Animal protein-free media for cultivation of cells |
| US10655099B2 (en) | 2004-10-29 | 2020-05-19 | Baxalta Incorporated | Animal protein-free media for cultivation of cells |
| US10138461B2 (en) | 2004-10-29 | 2018-11-27 | Baxalta GmbH | Animal protein-free media for cultivation of cells |
| US8440408B2 (en) | 2004-10-29 | 2013-05-14 | Baxter International Inc. | Animal protein-free media for cultivation of cells |
| US9714411B2 (en) | 2004-10-29 | 2017-07-25 | Baxalta GmbH | Animal protein-free media for cultivation of cells |
| US9222075B2 (en) | 2004-10-29 | 2015-12-29 | Baxalta Incorporated | Animal protein-free media for cultivation of cells |
| FR2879214A1 (en) * | 2004-12-14 | 2006-06-16 | Pierre Fabre Medicament Sa | PEPTIDE FRACTIONS PROMOTING THE GROWTH AND SYNTHESIS OF PRODUCT (S) OF INTEREST IN CELL CULTURE AND / OR TISSUE |
| US7993923B2 (en) | 2004-12-14 | 2011-08-09 | Pierre Fabre Medicament | Peptide fractions promoting growth and synthesis of desired product(S) into cell and/or tissue culture |
| WO2006064020A1 (en) * | 2004-12-14 | 2006-06-22 | Pierre Fabre Medicament | Peptide fractions promoting growth and synthesis of desired product(s) into cell and/or tissue culture |
| WO2006123926A3 (en) * | 2005-05-17 | 2007-07-12 | Noord Nl Oliemolen Holding B V | Peptide composition for growing and/or culturing micro-organisms and/or cells |
| NL1029059C2 (en) * | 2005-05-17 | 2006-11-20 | Noord Nl Oliemolen Holding B V | Peptide preparation for growing and / or growing microorganisms and / or cells. |
| US7754484B2 (en) * | 2005-11-16 | 2010-07-13 | Cognis Ip Management Gmbh | Use of esters of unsaturated, physiologically active fatty acids as nutrient media for cell cultures |
| US9758568B2 (en) | 2006-01-04 | 2017-09-12 | Baxalta GmbH | Oligopeptide-free cell culture media |
| US8663945B2 (en) | 2006-09-13 | 2014-03-04 | Abbvie Inc | Methods of producing anti-TNF-alpha antibodies in mammalian cell culture |
| EP2064314A4 (en) * | 2006-09-13 | 2009-12-30 | Abbott Lab | Cell culture improvements |
| US8906646B2 (en) | 2006-09-13 | 2014-12-09 | Abbvie Inc. | Fed-batch method of making human anti-TNF-alpha antibody |
| US8911964B2 (en) | 2006-09-13 | 2014-12-16 | Abbvie Inc. | Fed-batch method of making human anti-TNF-alpha antibody |
| AU2007294731B2 (en) * | 2006-09-13 | 2014-04-17 | Abbvie Inc. | Cell culture improvements |
| JP2013230151A (en) * | 2006-09-13 | 2013-11-14 | Abbott Lab | Cell culture improvement |
| US10119118B2 (en) | 2006-09-13 | 2018-11-06 | Abbvie Inc. | Modified serum-free cell culture medium |
| RU2518289C2 (en) * | 2006-09-13 | 2014-06-10 | Эббви Инк, | Method for preparing fed-batch antibody or its fragment (versions) |
| EP2532737A3 (en) * | 2006-09-13 | 2013-03-27 | Abbott Laboratories | Cell culture improvements |
| US9284371B2 (en) | 2006-09-13 | 2016-03-15 | Abbvie Inc. | Methods of producing adalimumab |
| US9090867B2 (en) | 2006-09-13 | 2015-07-28 | Abbvie Inc. | Fed-batch method of making anti-TNF-alpha antibody |
| JP2016000030A (en) * | 2006-09-13 | 2016-01-07 | アッヴィ・インコーポレイテッド | Cell culture improvements |
| EP2527425A1 (en) * | 2006-09-13 | 2012-11-28 | Abbott Laboratories | Cell culture improvements |
| EP2500415A1 (en) * | 2006-09-13 | 2012-09-19 | Abbott Laboratories | Cell culture improvements |
| EP2500414A1 (en) * | 2006-09-13 | 2012-09-19 | Abbott Laboratories | Cell culture improvements |
| EP2500413A1 (en) * | 2006-09-13 | 2012-09-19 | Abbott Laboratories | Cell culture improvements |
| EP2500416A1 (en) * | 2006-09-13 | 2012-09-19 | Abbott Laboratories | Cell culture improvements |
| CN102337243A (en) * | 2006-09-13 | 2012-02-01 | 雅培制药有限公司 | Cell culture improvements |
| US8093045B2 (en) | 2006-09-13 | 2012-01-10 | Abbott Laboratories | Fed-batch cell culture methods using non-animal-based hydrolysates |
| US9234032B2 (en) | 2006-09-13 | 2016-01-12 | Abbvie Inc. | Fed-batch methods for producing adalimumab |
| JP2010503397A (en) * | 2006-09-13 | 2010-02-04 | アボット・ラボラトリーズ | Improved cell culture |
| US9062106B2 (en) | 2011-04-27 | 2015-06-23 | Abbvie Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| US9505834B2 (en) | 2011-04-27 | 2016-11-29 | Abbvie Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| US9090688B2 (en) | 2011-04-27 | 2015-07-28 | Abbvie Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| US9365645B1 (en) | 2011-04-27 | 2016-06-14 | Abbvie, Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| US9255143B2 (en) | 2011-04-27 | 2016-02-09 | Abbvie Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| US9181572B2 (en) | 2012-04-20 | 2015-11-10 | Abbvie, Inc. | Methods to modulate lysine variant distribution |
| US9193787B2 (en) | 2012-04-20 | 2015-11-24 | Abbvie Inc. | Human antibodies that bind human TNF-alpha and methods of preparing the same |
| US9708400B2 (en) | 2012-04-20 | 2017-07-18 | Abbvie, Inc. | Methods to modulate lysine variant distribution |
| US9150645B2 (en) | 2012-04-20 | 2015-10-06 | Abbvie, Inc. | Cell culture methods to reduce acidic species |
| US9334319B2 (en) | 2012-04-20 | 2016-05-10 | Abbvie Inc. | Low acidic species compositions |
| US9346879B2 (en) | 2012-04-20 | 2016-05-24 | Abbvie Inc. | Protein purification methods to reduce acidic species |
| US9359434B2 (en) | 2012-04-20 | 2016-06-07 | Abbvie, Inc. | Cell culture methods to reduce acidic species |
| US9505833B2 (en) | 2012-04-20 | 2016-11-29 | Abbvie Inc. | Human antibodies that bind human TNF-alpha and methods of preparing the same |
| US9683033B2 (en) | 2012-04-20 | 2017-06-20 | Abbvie, Inc. | Cell culture methods to reduce acidic species |
| US9957318B2 (en) | 2012-04-20 | 2018-05-01 | Abbvie Inc. | Protein purification methods to reduce acidic species |
| US9249182B2 (en) | 2012-05-24 | 2016-02-02 | Abbvie, Inc. | Purification of antibodies using hydrophobic interaction chromatography |
| US9234033B2 (en) | 2012-09-02 | 2016-01-12 | Abbvie, Inc. | Methods to control protein heterogeneity |
| US9206390B2 (en) | 2012-09-02 | 2015-12-08 | Abbvie, Inc. | Methods to control protein heterogeneity |
| US9512214B2 (en) | 2012-09-02 | 2016-12-06 | Abbvie, Inc. | Methods to control protein heterogeneity |
| US9290568B2 (en) | 2012-09-02 | 2016-03-22 | Abbvie, Inc. | Methods to control protein heterogeneity |
| US9067990B2 (en) | 2013-03-14 | 2015-06-30 | Abbvie, Inc. | Protein purification using displacement chromatography |
| US9499614B2 (en) | 2013-03-14 | 2016-11-22 | Abbvie Inc. | Methods for modulating protein glycosylation profiles of recombinant protein therapeutics using monosaccharides and oligosaccharides |
| US8921526B2 (en) | 2013-03-14 | 2014-12-30 | Abbvie, Inc. | Mutated anti-TNFα antibodies and methods of their use |
| US9708399B2 (en) | 2013-03-14 | 2017-07-18 | Abbvie, Inc. | Protein purification using displacement chromatography |
| US9598667B2 (en) | 2013-10-04 | 2017-03-21 | Abbvie Inc. | Use of metal ions for modulation of protein glycosylation profiles of recombinant proteins |
| US9315574B2 (en) | 2013-10-18 | 2016-04-19 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US9017687B1 (en) | 2013-10-18 | 2015-04-28 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same using displacement chromatography |
| US9200069B2 (en) | 2013-10-18 | 2015-12-01 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US9499616B2 (en) | 2013-10-18 | 2016-11-22 | Abbvie Inc. | Modulated lysine variant species compositions and methods for producing and using the same |
| US9181337B2 (en) | 2013-10-18 | 2015-11-10 | Abbvie, Inc. | Modulated lysine variant species compositions and methods for producing and using the same |
| US9522953B2 (en) | 2013-10-18 | 2016-12-20 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US9688752B2 (en) | 2013-10-18 | 2017-06-27 | Abbvie Inc. | Low acidic species compositions and methods for producing and using the same using displacement chromatography |
| US9085618B2 (en) | 2013-10-18 | 2015-07-21 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US9266949B2 (en) | 2013-10-18 | 2016-02-23 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US9200070B2 (en) | 2013-10-18 | 2015-12-01 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US8946395B1 (en) | 2013-10-18 | 2015-02-03 | Abbvie Inc. | Purification of proteins using hydrophobic interaction chromatography |
| US9550826B2 (en) | 2013-11-15 | 2017-01-24 | Abbvie Inc. | Glycoengineered binding protein compositions |
| US12349704B2 (en) | 2018-11-15 | 2025-07-08 | Aleph Farms Ltd. | High quality cultured meat, compositions and methods for producing same |
| WO2020127941A2 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Serum free intracellular pathogen vaccine |
| US20220186191A1 (en) * | 2019-01-17 | 2022-06-16 | Boehringer Ingelheim Animal Health USA Inc. | Serum-free medium for avian vaccine production and uses thereof |
| US12441989B2 (en) * | 2019-01-17 | 2025-10-14 | Boehringer Ingelheim Vetmedica Gmbh | Serum-free medium for avian vaccine production and uses thereof |
| CN110257325A (en) * | 2019-05-31 | 2019-09-20 | 苏州智享众创孵化管理有限公司 | A method of the in vitro culture self-healing cell based on aloe extract |
| WO2021123099A1 (en) | 2019-12-20 | 2021-06-24 | Intervet International B.V. | Reassorted isa virus |
| US12408684B2 (en) | 2019-12-31 | 2025-09-09 | Air Protein, Inc. | High protein food compositions |
Also Published As
| Publication number | Publication date |
|---|---|
| WO1999057246A8 (en) | 2000-10-12 |
| WO1999057246A9 (en) | 2001-06-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1999057246A1 (en) | Animal cell culture media comprising non-animal or plant-derived nutrients | |
| US6103529A (en) | Animal cell culture media comprising peptides derived from rice | |
| US20090061516A1 (en) | Animal cell culture media comprising non animal or plant derived nutrients | |
| US20040171152A1 (en) | Animal cell culture media comprising non-animal or plant-derived nutrients | |
| US6692961B1 (en) | Defined systems for epithelial cell culture and use thereof | |
| US20050227355A1 (en) | Serum-free cell culture media | |
| US20110183375A1 (en) | Metal binding compounds and their use in cell culture medium compositions | |
| US20200392461A1 (en) | In vitro avian food product | |
| EP1210410B1 (en) | Metal binding compounds and their use in cell culture medium compositions | |
| US6833271B2 (en) | Serum-free cell culture media | |
| DE69737455T2 (en) | DEFINED SYSTEMS FOR CULTURING EPITHELIAL CELLS AND APPLYING THE SAME | |
| KR20110138269A (en) | Improved Single Cell Cloning Method | |
| US20110143432A1 (en) | Use of extract of selenium-enriched yeast (se-ye) in mammalian cell culture media formulations | |
| DK2218775T3 (en) | A method of producing a recombinant polypeptide in vitro CHO cell in a serum-free and protein-free culture medium | |
| WO2012008920A1 (en) | Materials and methods for enhanced iron uptake in cell culture |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: C1 Designated state(s): JP |
|
| AL | Designated countries for regional patents |
Kind code of ref document: C1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| CFP | Corrected version of a pamphlet front page | ||
| CR1 | Correction of entry in section i |
Free format text: PAT. BUL. 45/99 UNDER (30) REPLACE "NOT FURNISHED" BY "09/302953" |
|
| AK | Designated states |
Kind code of ref document: C2 Designated state(s): JP |
|
| AL | Designated countries for regional patents |
Kind code of ref document: C2 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| COP | Corrected version of pamphlet |
Free format text: PAGES 1-41, DESCRIPTION, REPLACED BY NEW PAGES 1-43; PAGES 42-45, CLAIMS, REPLACED BY NEW PAGES 44-48; PAGES 1/3-3/3, DRAWINGS, REPLACED BY NEW PAGES 1/3-3/3; DUE TO LATE TRANSMITTAL BY THE RECEIVING OFFICE |
|
| 122 | Ep: pct application non-entry in european phase |