WO1998048805A1 - Pharmaceutical composition for supressing type 2 helper t cell immune response - Google Patents
Pharmaceutical composition for supressing type 2 helper t cell immune response Download PDFInfo
- Publication number
- WO1998048805A1 WO1998048805A1 PCT/JP1998/001841 JP9801841W WO9848805A1 WO 1998048805 A1 WO1998048805 A1 WO 1998048805A1 JP 9801841 W JP9801841 W JP 9801841W WO 9848805 A1 WO9848805 A1 WO 9848805A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- carbon atoms
- alkyl
- immune response
- pharmaceutical composition
- pharmaceutically acceptable
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *c1nc(c(N)nc2c3cccc2)c3[n]1* Chemical compound *c1nc(c(N)nc2c3cccc2)c3[n]1* 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/4738—Quinolines; Isoquinolines ortho- or peri-condensed with heterocyclic ring systems
- A61K31/4745—Quinolines; Isoquinolines ortho- or peri-condensed with heterocyclic ring systems condensed with ring systems having nitrogen as a ring hetero atom, e.g. phenantrolines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/16—Otologicals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
Definitions
- the invention is directed to a pharmaceutical composition having less side effects for treating or preventing allergic diseases such as asthma, allergic dermatitis, allergic rhinitis, or an autoimmune disease such as systemic lupus erythematosus by suppressing immune response of type 2 helper T cell (hereinafter "Th2") and enhancing immune response of type 1 helper T cell (hereinafter "Th l”) comprising a therapeutically effective amount of a compound having structure of 1H- imidazo[4,5-c]quinolin-4-amine.
- the invention also directs to a method of treating or preventing allergic diseases or autoimmune diseases.
- Th helper T cell
- Thl /Th2 is not only a classification of helper T cell subsets, but also the concept by which an immune response can be categorized into a Th 1 type immune response and a Th2 type immune response in vivo.
- cytokines produced by an activated Thl such as interferon- ⁇ (IFN- ⁇ ), interleukin 2 (IL-2) and so on, play a major role.
- IFN- ⁇ interferon- ⁇
- IL-2 interleukin 2
- IL-12 is produced from the activated macrophage.
- IL- 12 augments the activation of Thl.
- Th l is considered to be related to cellular immunity such as protection of a virus or a bacterium infection through the above mechanism.
- Th2 type immune response cytokines produced from an activated Th2 cell, such as IL-4, IL-5 and so on, play a major role. It is reported that the Th2 cytokines relate to humoral immunity that includes antibody production from B cells (including IgE).
- Th2 is considered to be a cell that controls the allergic response, since Th2 produces cytokines such as IL-4 and IL-5 which are involved in the allergic response.
- cytokines such as IL-4 and IL-5 which are involved in the allergic response.
- IL-4 makes B cells to produce IgE.
- IL-4 also makes endothelial cells to express NCAM- 1, which is important in inducing eosinophils to adhere to endothelial cell and to invade into tissues (Pharmacia ( 1993) 29: 1123- 1 128).
- IL-4 is reported to be a differentiation and proliferation factor of Th2.
- IL-5 which is also a Th2 type cytokine is considered to be an elicitation factor of the allergic response, since IL-5 makes eosinophils to differentiate, to proliferate, to migrate and to activate.
- Th2 is recognized as a cell that mainly controls both an immediate phase allergic reaction that relates to an IgE antibody and a mast cell and a late phase allergic reaction that relates to an eosinophil. Therefore it is considered that allergy is a disease caused by abnormal activation of Th2 type immune response.
- Th2 and Th2 type cytokines, such as IL-4 and IL-5, are found in a local allergic lesioned site.
- Th2 type immune response It is important for treating or preventing allergic reaction to suppress the Th2 type immune response.
- a drug that can suppress the Th2 type immune response it can be an effective medicine for treating or preventing allergic diseases.
- the late phase allergic reaction play an important role especially in severe asthma, atopic dermatitis and so on.
- Anti-allergic agents which are available now, suppress only immediate phase allergic reaction and do not have sufficient clinical effects.
- steroids are effective for severe asthma and atopic dermatitis and have been frequently used. Long term administration of steroids may cause side effects such as steroid dermatitis, opportunistic infection, and dysfunction of the adrenal cortex. So it is expected to develop an agent that can suppress Th2 type immune response and can treat or prevent both of late phase allergic reaction and immediate phase allergic reaction.
- Th2 type immune response As described above when an agent that has less side effects is considered, it is preferable to develop an agent that not only can suppress the Th2 type immune response, but also can enhance the Th l type immune response. Since Thl produces INF- ⁇ and plays a major role in the protection of virus and bacterium infection, it is very preferable that a suppressor of the Th2 type immune response also has a property of enhancing Th l type immune response.
- An iramuno- suppressor such as cyclosporin or FK506 suppresses not only activation of Th2, but also suppresses activation of Thl non-specifically, and which also causes opportunistic infection. Such side effects have become a serious clinical problem.
- Th2 type immune response As described above, if an agent that can suppress Th2 type immune response and can enhance Th 1 type immune response is developed, it will be an effective medicine, which has less side-effects, for allergic diseases.
- Imiquimod exhibits an anti-herpes simplex virus activity in the guinea pig (Antimicrob. Agents Chemother. (1989) 33: 151 1- 1515). It is also reported that Imiquimod shows anti-viral activities in cytomegalovirus (Antimicrob. Agents Chemother. (1988) 32:
- Imiquimod shows an activity of enhancing production of IFN- ⁇ in vitro and in vivo mouse models (Journal of Leukocyte Biology (1994) 55: 234-240). It is also reported that Imiquimod also shows an anti-cancer activity in various models (Cancer Res. (1992) 52: 3528-3533).
- Imiquimod enhances production of IL- 1, IL-6, IL-8 and TNF- ⁇ in vitro and in vivo mouse experimental models (Journal of Leukocyte Biology (1994) 55: 234-240). It is suggested that a part of the anti-cancer activity comes from the activity of inducing the TNF- ⁇ production.
- Imiquimod has anti-viral and anti- cancer activities and enhances the production of INF- ⁇ and TNF- ⁇ , it has not been reported nor suggested that Imiquimod suppresses production of IL-4 and IL-5 from Th2 and enhances production of IFN- ⁇ .
- Imiquimod suppresses the Th2 type immune response and can be applied for treating or preventing allergic diseases and autoimmune diseases caused by abnormal activation of Th2 type immune response.
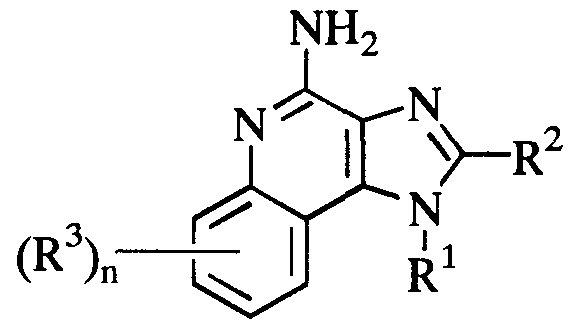
- the present invention is directed to a pharmaceutical composition for suppressing Th2 type immune response comprising a therapeutically effective amount of a compound represented by the formula ( 1):
- R 1 is a straight or branched chain alkyl having 1 to 10 carbon atoms; a cycloalkyl having 3 to 7 carbon atoms; a hydroxyalkyl having 1 to 6 carbon atoms; an acyloxyalkyl wherein the alkyl moiety has 1 to 6 carbon atoms and the acyloxy moiety is an alkanoyloxy having 2 to 4 carbon atoms or benzoyloxy; an aralkyl; a substituted aralkyl; a phenyl; or a substituted phenyl;
- R 2 is hydrogen atom or an alkyl having 1 to 8 carbon atoms
- R 3 is an alkoxy having 1 to 4 carbon atoms, a halogen atom or an alkyl having 1 to 4 carbon atoms
- n is an integer from 0 to 2; or a pharmaceutically acceptable acid salt thereof.
- the present invention is also directed to a method of treating or preventing a disease caused by abnormal activation of the Th.2 type immune response comprising administering a therapeutically effective amount of a compound represented by the formula ( 1):
- R 1 is a straight or branched chain alkyl having 1 to 10 carbon atoms; a cycloalkyl having 3 to 7 carbon atoms; a hydroxyalkyl having 1 to 6 carbon atoms; an acyloxyalkyl wherein the alkyl moiety has 1 to 6 carbon atoms and the acyloxy moiety is an alkanoyloxy having 2 to 4 carbon atoms or benzoyloxy; an aralkyl; a substituted aralkyl; a phenyl; or a substituted phenyl;
- R 2 is hydrogen atom or an alkyl having 1 to 8 carbon atoms
- R 3 is an alkoxy having 1 to 4 carbon atoms, a halogen atom or an alkyl having 1 to 4 carbon atoms
- n is an integer from 0 to 2; or a pharmaceutically acceptable acid salt thereof to a patient in need thereof.
- the present invention is directed to a pharmaceutical composition for treating or preventing an allergic disease or an autoimmune disease caused by abnormal activation of immune response of Th2 side comprising a therapeutically effective amount of a compound represented by the formula ( 1):
- R 1 is a straight or branched chain alkyl having 1 to 10 carbon atoms; a cycloalkyl having 3 to 7 carbon atoms; a hydroxyalkyl having 1 to 6 carbon atoms; an acyloxyalkyl wherein the alkyl moiety has 1 to 6 carbon atoms and the acyloxy moiety is an alkanoyloxy having 2 to 4 carbon atoms or benzoyloxy; an aralkyl; a substituted aralkyl; a phenyl; or a substituted phenyl;
- R 2 is hydrogen atom or an alkyl having 1 to 8 carbon atoms
- R 3 is an alkoxy having 1 to 4 carbon atoms, a halogen atom or an alkyl having 1 to 4 carbon atoms
- n is an integer from 0 to 2; or a pharmaceutically acceptable acid salt thereof to a patient in need thereof.
- the present invention is also directed to a pharmaceutical composition for treating or preventing an allergic disease or an autoimmune disease caused by abnormal activation of immune response of Th2 side comprising a therapeutically effective amount of a compound represented by the formula (1):
- R 1 is a straight or branched chain alkyl having 1 to 10 carbon atoms; a cycloalkyl having 3 to 7 carbon atoms; a hydroxyalkyl having 1 to 6 carbon atoms; an acyloxyalkyl wherein the alkyl moiety has 1 to 6 carbon atoms and the acyloxy moiety is an alkanoyloxy having 2 to 4 carbon atoms or benzoyloxy; an aralkyl; wherein the phenyl may be substituted by one or more than one substituents selected from the group consisting of an alkyl having 1 to 4 carbon atoms and an alkoxy having 1 to 4 carbon atoms; R 2 is hydrogen atom or an alkyl having 1 to 8 carbon atoms; R 3 is independently selected from the group consisting of an alkoxy having 1 to 4 carbon atoms, a halogen atom and an alkyl having 1 to 4 carbon atoms; and n is an integer from 0 to 2 with the proviso that
- a compound or a pharmaceutically acceptable salt thereof of formula (1) may be manufactured by a method known to a person of ordinary skill in the art. For example, it may be manufactured by a method described in Tokkou-Hei 5-86391 (JP5-86391B).
- Examples of the straight chain alkyl having 1 to 10 carbon atoms of R 1 are methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl and the like.
- the branched chain alkyl having 1 to 10 carbon atoms includes a branched chain alkyl having 3 to 10 carbon atoms.
- Examples of the branched chain alkyl group having 3 to 10 carbon atoms of R 1 are 1-methylethyl, 2-methylpropyl, 1-methylpropyl, 1, 1- dimethylethyl, 3-methylbutyl, 4-methylpentyl and the like.
- Examples of the cycloalkyl having 3 to 7 carbon atoms of R 1 are cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl and the like.
- a hydroxyalkyl having 1 to 6 carbon atoms of R 1 includes a straight chain hydroxyalkyl having 1 to 6 carbon atoms, a branched chain hydroxyalkyl having 3 to 6 carbon atoms and the like.
- Examples of the straight chain hydroxyalkyl having 1 to 6 carbon atoms are hydroxymethyl, 2-hydroxyethyl, 2-hydroxypropyl, 2-hydroxybutyl, 2- hydroxypentyl, 2-hydroxyhexyl and the like.
- Examples of the branched chain hydroxyalkyl having 3 to 6 carbon atoms are 2-hydroxy-2-methyl- propyl and the like.
- Examples of the alkyl moiety having 1 to 6 carbon atoms in the acyloxyalkyl of R 1 are methyl, ethyl, propyl, butyl, pentyl, hexyl and the like.
- the acyloxy moiety in the acyloxyalkyl of R 1 includes an alkanoyloxy having 2 to 4 carbon atoms, benzoyloxy and the like.
- Examples of the alkanoyloxy having 2 to 4 carbon atoms are acetyloxy, propanoyloxy, butanoyloxy and the like.
- Specific examples of the acyloxyalkyl are 2-acetyloxypropyl, 2-acetyloxy-2-methylpropyl, 2- benzoyloxy-2-methylpropyl and the like.
- the aralkyl of R 1 includes an aralkyl having 7 to 10 carbon atoms.
- Specific examples of the aralkyl are benzyl, phenethyl and the like.
- Examples of a substituent of the substituted aralkyl are an alkoxy having 1 to 4 carbon atoms, a halogen and the like.
- Examples of the alkoxy having 1 to 4 carbon atoms are methoxy, ethoxy, propoxy, butoxy and the like.
- Examples of the halogen are fluorine, chlorine, bromine and the like.
- the substituted aralkyl may have one or more substituents independently on the aryl moiety.
- Examples of a substituent of the substituted phenyl are an alkoxy having 1 to 4 carbon atoms, a halogen and the like.
- Examples of the alkoxy having 1 to 4 carbon atoms are methoxy, ethoxy, propoxy, butoxy and the like.
- Examples of the halogen are fluorine, chlorine, bromine and the like.
- the substituted phenyl may have one or more substituents independently.
- the alkyl having 1 to 8 carbon atoms of R 2 includes a straight chain alkyl having 1 to 8 carbon atoms, a branched chain alkyl having 3 to 8 carbon atoms.
- Examples of the straight chain alkyl having 1 to 8 carbon atoms are methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl and the like.
- Examples of the branched chain alkyl having 3 to 8 carbon atoms are 1-methylethyl, 1-methylpropyl, 2-methylpropyl, 1- methylbutyl, 1, 1-dimethylethyl and the like.
- alkoxy having 1 to 4 carbon atoms of R 3 are methoxy, ethoxy, propoxy, butoxy and the like.
- halogen of R 3 are chlorine, fluorine, bromine and the like. If n is 2, two R 3 may be same or different.
- the examples of pharmaceutically acceptable acid salt are an inorganic acid and an organic acid such as hydrogen chloride, sulfuric acid, acetic acid, oxalic acid, ascorbic acid and so on.
- a preferred embodiment of a compound represented by the formula (1) is that R 1 is 2-methylpropyl or 2-hyroxy-2-methylpropyl, and R 2 is hydrogen, methyl or ethyl and n is 0.
- a particularly preferred embodiment of a compound of the formula ( 1) includes l-(2-methylpropyl)- lH-imidazo[4,5-c]quinolin-4- amine (Imiquimod), R842, S-27609 (Journal of Leukocyte Biology (1995)
- the pharmaceutical composition of the present invention may further comprise other pharmaceutical agents.
- the pharmaceutical composition of the present invention may be used with other pharmaceutical agent.
- Such pharmaceuticals agent includes a bronchodilator, an anti-allergic agent, a steroid and the like that is available or known to a person of ordinary skill in the art and is used for treating allergic diseases.
- a steroid is often used for treating severe asthma and atopic dermatitis.
- long-term administration of steroid causes various side-effects such as steroid dermatitis, opportunistic infection, adrenocortical insufficiency, rebound of stopping administration and so on. It is expected that use of a specific suppressor of Th2 type immune response of the present invention with a steroid can reduce the amount of steroid administered and can also reduce the side-effects.
- composition of this invention can be administered in any number of conventional dosage forms, e.g., topical, oral, parenteral, rectal, transdermal, nasal and the like.
- Oral or rectal dosage forms include capsules, tablets, pills, powders, cachets, and suppositories.
- Liquid oral dosage forms include solutions and suspensions.
- Parenteral preparations include sterile solutions and suspensions.
- Topical dosage forms can be creams, ointments, lotions, transdermal devices (e.g., of conventional patch or matrix type) and the like.
- the above described dosage forms can be prepared with conventional pharmaceutically acceptable excipients and additives, using conventional techniques.
- Such pharmaceutically acceptable excipients and additives are intended to include carriers, binders, flavorings, buffers, thickeners, coloring agents, stabilizing agents, emulsifying agents, dispersing agents, suspending agents, perfumes, preservatives, lubricants, etc.
- Suitable pharmaceutically acceptable solid carriers include magnesium carbonate, magnesium stearate, talc, sugar, lactose, pectin, dextrin, starch, gelatin, tragacanth, methylcellulose, sodium carboxymethylcellulose, low melting waxes, cocoa butter and the like.
- Capsules can be made wherein the active compound is filled into the capsules together with a pharmaceutically acceptable carrier.
- the active ingredient of this invention can be mixed with pharmaceutically acceptable excipients or be used in finely divided powder form without excipients for inclusion into the capsules. Similarly, cachets are included.
- Liquid form preparations include solutions, suspensions and emulsions such as water or water-propylene glycol solutions for parenteral injection. Liquid form preparations can also be formulated in a solution in polyethylene glycol and /or propylene glycol, which may contain water.
- Aqueous solutions suitable for oral use can be prepared by dissolving the active component in water and adding thereto suitable colorants, flavors, stabilizing, sweetening, solubilizing and thickening agents as desired.
- Aqueous suspensions suitable for oral use can be made by dispersing the active component in finely divided form in water with viscous materials, i.e. pharmaceutically acceptable natural and synthetic gums, resins, methylcellulose, sodium carboxymethylcellulose and other well-known suspending agents.
- Formulations for topical application may include the above liquid forms, as well as creams, aerosols, sprays, dusts, powders, lotions and ointments which are prepared by combining an active ingredient according to this invention with conventional pharmaceutically acceptable diluents and carriers commonly used in topical, dry, liquid, cream and aerosol formulations.
- Ointment and creams may, for example, be formulated with an aqueous or oily base with the addition of suitable thickening and/ or gelling agents.
- bases may, thus, for example, include water and/ or an oil such as liquid paraffin or a vegetable oil such as peanut oil or castor oil.
- Thickening agents which may be used according to the nature of the base include soft paraffin, aluminum stearate, cetostearyl alcohol, propylene glycol, polyethylene glycols, woolfat, hydrogenated lanolin, beeswax, etc.
- Lotions may be formulated with an aqueous or oil base and will, in general, also include one or more of pharmaceutically acceptable stabilizing agents, suspending agents, emulsifying agents, dispersing agents, thickening agents, coloring agents, perfumes and the like. Powders may be formed with the aid of any suitable pharmaceutically acceptable powder base, e.g., talc, lactose, starch, etc.
- Drops may be formulated with an aqueous base or nonaqueous base comprising one or more pharmaceutically acceptable dispersing agents, suspending agents, solubilizing agents, etc.
- the topical pharmaceutical composition may also include one or more preservatives or bacteriostatic agents, e.g., methyl hydroxybenzoate, propyl hydroxybenzoate, chlorocresol, benzalkonium chloride, etc.
- preservatives or bacteriostatic agents e.g., methyl hydroxybenzoate, propyl hydroxybenzoate, chlorocresol, benzalkonium chloride, etc.
- topical pharmaceutical compositions may also contain an active compound of this invention in combination with other active ingredients such as antimicrobial agents, particularly antibiotics, anesthetics, analgesics, and antipruritic agents.
- active ingredients such as antimicrobial agents, particularly antibiotics, anesthetics, analgesics, and antipruritic agents.
- the compound of formula (1) may be used, for example, as a liquid spray, as a powder or in the form of drops.
- administration by inhalation of the compound of formula ( 1) are conveniently delivered in the form of an aerosol spray presentation from pressurised packs or a nebulizer, with the use of a suitable propellant, e.g. dichlorodifluoromethane, trichlorofluoromethane, dichlorotetrafluoroethane, 1, 1, 1,2-tetrafluoroethane, carbon dioxide or other suitable gas.
- a suitable propellant e.g. dichlorodifluoromethane, trichlorofluoromethane, dichlorotetrafluoroethane, 1, 1, 1,2-tetrafluoroethane, carbon dioxide or other suitable gas.
- the dosage unit may be determined by providing a valve to deliver a metered amount.
- solid form preparations which are intended to be converted, shortly before use, to liquid form preparations for either oral or parenteral administration.
- liquid forms include solutions, suspensions and emulsions.
- solid form preparations are most conveniently provided in unit dosage form and as such are used to provide a single liquid dosage unit.
- sufficient solid may be provided so that after conversion to liquid form, multiple individual liquid doses may be obtained by measuring predetermined volumes of liquid form preparation as with a syringe, teaspoon or volumetric container. When multiple liquid doses are prepared, it is preferred to maintain the unused portion of said liquid doses under conditions which retard possible decomposition.
- the solid form preparations intended to be converted to liquid form may contain, in addition to the active ingredient, pharmaceutically acceptable flavorants, colorants, stabilizers, buffers, artificial and natural sweeteners, dispersants, thickeners, solubilizing agents and the like.
- the solvent utilized for preparing the liquid form preparation may be water, isotonic water, ethanol, glycerin, propylene glycol and the like as well as mixtures thereof. Naturally, the solvent utilized will be chosen with regard to the route of administration, for example, liquid preparations containing large amount of ethanol are not suitable for parenteral use.
- the active ingredient of this invention may also deliverable transdermally for systemic distribution. As a transdermal patch of the matrix or reservoir type as are conventional in the art for this purpose.
- the compound represented by the formula (1) can also be formulated as depot preparations. Such long acting formulation can be administered by implantation (for example subcutaneously or intra- muscularly) or by intramuscular injection.
- the compound can be formulated with suitable polymeric or hydrophobic materials (for example as an emulsion in a pharmaceutically acceptable oil) or ion exchange resins, or as sparingly soluble derivatives, for example as a sparingly soluble salt.
- the compound represented by the formula (1) can also be coupled to a class of biodegradable polymers useful in achieving controlled release of a drug, for example, poly lactic acid, polyglycolic acid, copolymers of polylactic acid and polyglycolic acid, polyepsilon caprolactone, polyhydroxybutyric acid, polyorthoesters, polyacetals, polydihydropyrans, polycyanoacylates, and crosslinked or amphipathic block copolymers of hydrogels.
- a class of biodegradable polymers useful in achieving controlled release of a drug
- a drug for example, poly lactic acid, polyglycolic acid, copolymers of polylactic acid and polyglycolic acid, polyepsilon caprolactone, polyhydroxybutyric acid, polyorthoesters, polyacetals, polydihydropyrans, polycyanoacylates, and crosslinked or amphipathic block copolymers of hydrogels.
- a composition of the invention comprises a therapeutically effective amount of a compound represented by the formula (1) in combination with a pharmaceutically acceptable carrier material.
- the composition of the invention may be administered by any conventional mode of administration by employing a therapeutically effective amount of a compound represented by the formula (1) for such mode.
- the dosage may be varied depending upon the requirements of the patient in the judgment of attending clinician, the severity of the condition being treated and the particular compound being employed.
- Treatment can be initiated with smaller dosages which are less than the optimum dose of the compound. Thereafter the dosage should be increased by small increments until the optimum effect under the circumstances is reached.
- the total daily dose may be divided and administered in portions during the day if desired.
- An oral pharmaceutical composition of present invention can be administered once a day or more than once a day.
- an amount of dosage for said oral administration is selected from ranging about 1 to about 200mg, and preferred range is from about 10 to about 50mg.
- a pharmaceutical composition of the present invention for injection can be administered once a day or more than once a day.
- An amount of dosage for said administration for injection is selected from ranging about 0.1 to about lOOmg, and preferred range is from about 3 to about 30mg.
- BALB/c mouse was purchased from Nihon Charles River (Yokohama, Japan) and female mice of 8 week-old were used for the experiment.
- fetal bovine serum (Characterized, code No. A- l 115-L, HyClone Lab., Logan, Utah) inactivated by heated to 56°C for 30 minutes was added to become 10 % and 2-mercaptoethanol was added to become 0.05 mM.
- cytokines Production of cytokines by stimulation of antigen
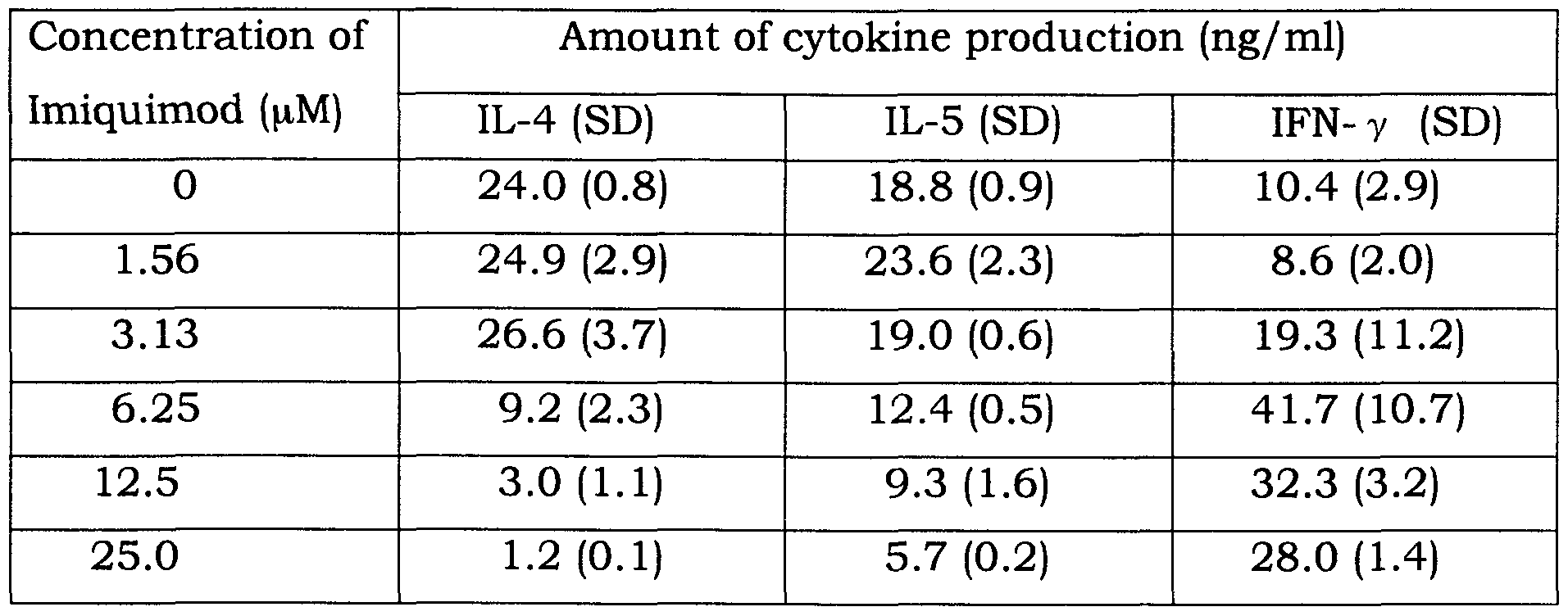
- the KLH (O. lmg/ml) and imiquimod solution prepared in 3 was added to lymph node cell suspension (2.5xl0 6 cells/ml) prepared in 4, and was cultured for four days at 37°C under 5 % C0 2 atmosphere (0.15 ml/ well).
- the cytokines in the supernatant was measured by ELISA described in 6.
- An amount of IL-4 and IL-5 that are representatives of Th2 type cytokines and an amount of IFN- ⁇ that is a representative of Thl type cytokines were measured. 6.
- Rat anti-mouse-IL-4 antibody was used as a primary antibody (Pharmingen, San Diego, CA, Code No. 1803 ID, 0.5 mg/ml) and was diluted to 250 times by carbonate buffered solution. 50 ⁇ l of the solution was put into 96-well plate (Falcon 3912, Becton Dickinson and Company, Franklin Lakes, NJ). The plate was incubated at 4°C over a night. The plate was blocked by using PBS (-) containing 3 % of BSA. The plate was rinsed, dried and stored at -20°C.
- IL-5 Quantitative measurement of IL-5 was done by a similar method described above using rat anti-mouse-IL-5 antibody (Pharmingen, San Diego, CA, Code No. 1805 ID, 0.5 mg/ml) as a primary antibody and biotinated rat anti-mouse-IL5 antibody (Pharmingen, San Diego, CA, Code No. 18062D, 0.5 mg/ml) as a secondary antibody.
- rat anti-mouse-IL-5 antibody Puringen, San Diego, CA, Code No. 1805 ID, 0.5 mg/ml
- biotinated rat anti-mouse-IL5 antibody Pharmingen, San Diego, CA, Code No. 18062D, 0.5 mg/ml
- CA Code No. 1924 IW, 0.5 mg/ml
- Quantitative measurement of IFN- ⁇ was done by a similar method described above using rat anti-mouse-IFN- ⁇ antibody (Pharmingen, San Diego, CA, Code No. 1818 ID, 0.5 mg/ml) as a primary antibody and biotinated rat anti-mouse-IFN- ⁇ antibody (Pharmingen, San Diego, CA, Code No. 18112D, 0.5 mg/ml) as a secondary antibody.
- Recombinant mouse IFN- ⁇ (Pharmingen, San Diego, CA, Code No. 19301U, 0.5 mg/ml) was used to make a calibration curve.
- mice Male 7 week-old BALB/c mice were sensitized by painting with 0.2 ml of 0.5 % acetone/dibutyl phthalate solution of fluoresceine isothiocyanate (hereinafter FITC) on the abdomen being shaved a day before the sensitization.
- FITC fluoresceine isothiocyanate
- ear swelling was elicited by applying 20 ⁇ l of 0.5 % acetone/dibutyl phthalate solution of FITC to each side of left ear. 24 hours later ear thickness was measured by micrometer, and the difference between before and after elicitation was studied. Test compound was suspended in 0.5 % carboxymethylcellulose and administered orally two hours before the elicitation. 2. Result
- ovalbumin 100 ⁇ g was adsorbed to 1.6mg of aluminum hydroxide gel (200 ⁇ l) and the adsorbed aluminum hydroxide gel was immunized by subcutaneous administration to dorsum of male 8 week- old BALB/c mice. Seven days later the mouse was immunized by the adsorbed aluminium hydroxide gel again. Seven days after second immunization 10 ⁇ g of ovalbumin in 200 ⁇ l of saline was administered intraperitoneally. Two days after the intraperitoneal administration peritoneal exudated cells were collected by using saline. Total number of peritoneal exudated cells and eosinophils was measured by the method of staining by Turk solution and Hinkelman solution. Test compound was suspended in 0.5 % carboxymethylcellulose and administered orally ( 10 ml/ kg) two hours before the third ovalbumin administration. 2. Result
- Imiquimod 30 3 13.9 ⁇ 0.7** 395.0 ⁇ 50.3 3.6 ⁇ 0.5**
Landscapes
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Pulmonology (AREA)
- Epidemiology (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP98917620A EP0977569A1 (en) | 1997-04-25 | 1998-04-22 | Pharmaceutical composition for supressing type 2 helper t cell immune response |
| AU70790/98A AU7079098A (en) | 1997-04-25 | 1998-04-22 | Pharmaceutical composition for supressing type 2 helper t cell immune respons |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP9/123146 | 1997-04-25 | ||
| JP9123146A JPH10298181A (en) | 1997-04-25 | 1997-04-25 | Type 2 helper T cell selective immune response inhibitor |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1998048805A1 true WO1998048805A1 (en) | 1998-11-05 |
Family
ID=14853339
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP1998/001841 Ceased WO1998048805A1 (en) | 1997-04-25 | 1998-04-22 | Pharmaceutical composition for supressing type 2 helper t cell immune response |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP0977569A1 (en) |
| JP (1) | JPH10298181A (en) |
| AU (1) | AU7079098A (en) |
| WO (1) | WO1998048805A1 (en) |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1187613A4 (en) * | 1999-06-10 | 2002-07-24 | 3M Innovative Properties Co | Amide substituted imidazoquinolines |
| EP1198232A4 (en) * | 1999-06-10 | 2002-07-24 | 3M Innovative Properties Co | Urea substituted imidazoquinolines |
| US6756382B2 (en) | 1999-06-10 | 2004-06-29 | 3M Innovative Properties Company | Amide substituted imidazoquinolines |
| US6784188B2 (en) | 1999-06-10 | 2004-08-31 | 3M Innovative Properties Company | Urea substituted imidazoquinolines |
| US6800624B2 (en) | 1999-06-10 | 2004-10-05 | 3M Innovative Properties Company | Sulfonamide and sulfamide substituted imidazoquinolines |
| US6869956B2 (en) * | 2000-10-03 | 2005-03-22 | Bristol-Myers Squibb Company | Methods of treating inflammatory and immune diseases using inhibitors of IκB kinase (IKK) |
| EP1478327A4 (en) * | 2002-02-22 | 2007-07-04 | 3M Innovative Properties Co | Method of reducing and treating uvb-induced immunosuppression |
| WO2009091541A1 (en) * | 2008-01-15 | 2009-07-23 | Dow Pharmaceutical Sciences, Inc. | Imiquimod formulation |
| US8691837B2 (en) | 2003-11-25 | 2014-04-08 | 3M Innovative Properties Company | Substituted imidazo ring systems and methods |
| US8846697B2 (en) | 2006-05-31 | 2014-09-30 | The Regents Of The University Of California | Purine analogs |
| US8871782B2 (en) | 2003-10-03 | 2014-10-28 | 3M Innovative Properties Company | Alkoxy substituted imidazoquinolines |
| US9050376B2 (en) | 2007-02-07 | 2015-06-09 | The Regents Of The University Of California | Conjugates of synthetic TLR agonists and uses therefor |
| US9066940B2 (en) | 2009-02-06 | 2015-06-30 | Telormedix, Sa | Pharmaceutical compositions comprising imidazoquinolin(amines) and derivatives thereof suitable for local administration |
| US9248127B2 (en) | 2005-02-04 | 2016-02-02 | 3M Innovative Properties Company | Aqueous gel formulations containing immune response modifiers |
| US9359360B2 (en) | 2005-08-22 | 2016-06-07 | The Regents Of The University Of California | TLR agonists |
| US11697851B2 (en) | 2016-05-24 | 2023-07-11 | The Regents Of The University Of California | Early ovarian cancer detection diagnostic test based on mRNA isoforms |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MY146124A (en) * | 2003-10-03 | 2012-06-29 | 3M Innovative Properties Co | Pyrazolopyridines and analogs thereof |
| EP2363148B1 (en) | 2008-10-31 | 2014-11-26 | Japan Science and Technology Agency | Method for selectively controlling function of helper t cell |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0145340A2 (en) * | 1983-11-18 | 1985-06-19 | Riker Laboratories, Inc. | 1H-Imidazo[4,5-c]quinolines and 1H-imidazo[4,5-c]quinolin-4-amines |
| WO1992015582A1 (en) * | 1991-03-01 | 1992-09-17 | Minnesota Mining And Manufacturing Company | 1-SUBSTITUTED, 2-SUBSTITUTED 1H-IMIDAZO[4,5-c]QUINOLIN-4-AMINES |
| WO1993005042A1 (en) * | 1991-09-04 | 1993-03-18 | Minnesota Mining And Manufacturing Company | 1-substituted 1h-imidazo(4,5-c)quinolin-4-amines; intermediate and pharmaceutical compositions |
-
1997
- 1997-04-25 JP JP9123146A patent/JPH10298181A/en active Pending
-
1998
- 1998-04-22 EP EP98917620A patent/EP0977569A1/en not_active Withdrawn
- 1998-04-22 AU AU70790/98A patent/AU7079098A/en not_active Abandoned
- 1998-04-22 WO PCT/JP1998/001841 patent/WO1998048805A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0145340A2 (en) * | 1983-11-18 | 1985-06-19 | Riker Laboratories, Inc. | 1H-Imidazo[4,5-c]quinolines and 1H-imidazo[4,5-c]quinolin-4-amines |
| WO1992015582A1 (en) * | 1991-03-01 | 1992-09-17 | Minnesota Mining And Manufacturing Company | 1-SUBSTITUTED, 2-SUBSTITUTED 1H-IMIDAZO[4,5-c]QUINOLIN-4-AMINES |
| WO1993005042A1 (en) * | 1991-09-04 | 1993-03-18 | Minnesota Mining And Manufacturing Company | 1-substituted 1h-imidazo(4,5-c)quinolin-4-amines; intermediate and pharmaceutical compositions |
Non-Patent Citations (10)
| Title |
|---|
| C.E.WEEKS ET AL.: "Induction of Interferon and Other Cytokines by Imiquimod and Its Hydroxylated Metabolite R-842 in Human Blood Cells In Vitro", J.INTERFERON RES., vol. 14, no. 2, April 1994 (1994-04-01), pages 81 - 85, XP002073165 * |
| K.KARACA ET AL.: "In Vivo and In Viro Interferon Induction in Chickens by S-28828, an Imidazoquinolinamine Immunoenhancer", J.INTERFERON CYTOKINE RES., vol. 16, no. 4, April 1996 (1996-04-01), pages 327 - 332, XP002073166 * |
| K.MEGYERI ET AL.: "ERRATA", MOL.CELL.BIOL., vol. 15, no. 5, May 1995 (1995-05-01), pages 2905 * |
| K.MEGYERI ET AL.: "Stimulation of Interferon and Cytokine Gene Expression by Imiquimod and Stimulation by Sendai Virus Utilize Similar Signal Transduction Pathways", MOL.CELL.BIOL., vol. 15, no. 4, April 1995 (1995-04-01), pages 2207 - 2218, XP002073163 * |
| M.J.REITER ET AL.: "Cytokine induction in mice by the immunomodulator imiquimod", J.LEUKOC.BIOL., vol. 55, no. 2, February 1994 (1994-02-01), pages 234 - 240, XP002073159 * |
| P.L. WITT ET AL.: "Phase I Trial of an Oral Immunomodulator and Interferon Inducer in Cancer Patients", CANCER RES., vol. 53, no. 21, 1 November 1993 (1993-11-01), pages 5176 - 5180, XP002073162 * |
| P.SAVAGE ET AL.: "A phase I clinical trial of imiquimod, an oral interferon inducer, administered daily", BR.J.CANCER, vol. 74, no. 9, November 1996 (1996-11-01), pages 1482 - 1486, XP002073161 * |
| R.L.MILLER ET AL.: "Cytokine induction by imiquimod", CHEMOTHER.J., vol. 4, no. 3, August 1995 (1995-08-01), pages 148 - 150, XP002073158 * |
| S.J.GIBSON ET AL.: "Cellular Requirements for Cytokine Production in Response to the Immunomodulators Imiquimod and S-27609", J.INTERFERON CYTOKINE RES., vol. 15, no. 6, June 1995 (1995-06-01), pages 537 - 545, XP002073164 * |
| T.L.TESTERMAN ET AL.: "Cytokine induction by the immunomodulators imiquimod and S-27609", J.LEUKOC.BIOL., vol. 58, no. 3, September 1995 (1995-09-01), pages 365 - 372, XP002073160 * |
Cited By (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7393859B2 (en) | 1999-06-10 | 2008-07-01 | Coley Pharmaceutical Group, Inc. | Amide substituted imidazoquinolines |
| US6756382B2 (en) | 1999-06-10 | 2004-06-29 | 3M Innovative Properties Company | Amide substituted imidazoquinolines |
| US6780873B2 (en) | 1999-06-10 | 2004-08-24 | 3M Innovative Properties Company | Urea substituted imidazoquinolines |
| US6784188B2 (en) | 1999-06-10 | 2004-08-31 | 3M Innovative Properties Company | Urea substituted imidazoquinolines |
| US6800624B2 (en) | 1999-06-10 | 2004-10-05 | 3M Innovative Properties Company | Sulfonamide and sulfamide substituted imidazoquinolines |
| US6897221B2 (en) | 1999-06-10 | 2005-05-24 | 3M Innovative Properties Company | Urea substituted imidazoquinolines |
| US7157453B2 (en) | 1999-06-10 | 2007-01-02 | 3M Innovation Properties Company | Urea substituted imidazoquinolines |
| EP1198232A4 (en) * | 1999-06-10 | 2002-07-24 | 3M Innovative Properties Co | Urea substituted imidazoquinolines |
| EP1187613A4 (en) * | 1999-06-10 | 2002-07-24 | 3M Innovative Properties Co | Amide substituted imidazoquinolines |
| US6869956B2 (en) * | 2000-10-03 | 2005-03-22 | Bristol-Myers Squibb Company | Methods of treating inflammatory and immune diseases using inhibitors of IκB kinase (IKK) |
| EP1478327A4 (en) * | 2002-02-22 | 2007-07-04 | 3M Innovative Properties Co | Method of reducing and treating uvb-induced immunosuppression |
| US8871782B2 (en) | 2003-10-03 | 2014-10-28 | 3M Innovative Properties Company | Alkoxy substituted imidazoquinolines |
| US8691837B2 (en) | 2003-11-25 | 2014-04-08 | 3M Innovative Properties Company | Substituted imidazo ring systems and methods |
| US9248127B2 (en) | 2005-02-04 | 2016-02-02 | 3M Innovative Properties Company | Aqueous gel formulations containing immune response modifiers |
| US10071156B2 (en) | 2005-02-04 | 2018-09-11 | 3M Innovative Properties Company | Aqueous gel formulations containing immune response modifiers |
| US9359360B2 (en) | 2005-08-22 | 2016-06-07 | The Regents Of The University Of California | TLR agonists |
| US8846697B2 (en) | 2006-05-31 | 2014-09-30 | The Regents Of The University Of California | Purine analogs |
| US9050376B2 (en) | 2007-02-07 | 2015-06-09 | The Regents Of The University Of California | Conjugates of synthetic TLR agonists and uses therefor |
| WO2009091541A1 (en) * | 2008-01-15 | 2009-07-23 | Dow Pharmaceutical Sciences, Inc. | Imiquimod formulation |
| US9107919B2 (en) | 2009-02-06 | 2015-08-18 | Telormedix Sa | Pharmaceutical compositions comprising imidazoquinolin(amines) and derivatives thereof suitable for local administration |
| US9066940B2 (en) | 2009-02-06 | 2015-06-30 | Telormedix, Sa | Pharmaceutical compositions comprising imidazoquinolin(amines) and derivatives thereof suitable for local administration |
| US11697851B2 (en) | 2016-05-24 | 2023-07-11 | The Regents Of The University Of California | Early ovarian cancer detection diagnostic test based on mRNA isoforms |
Also Published As
| Publication number | Publication date |
|---|---|
| AU7079098A (en) | 1998-11-24 |
| EP0977569A1 (en) | 2000-02-09 |
| JPH10298181A (en) | 1998-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0977569A1 (en) | Pharmaceutical composition for supressing type 2 helper t cell immune response | |
| CN114751950B (en) | Cyclic dinucleotide compounds and methods of use | |
| EP0641342B1 (en) | Antiviral 2-ethyl-1h-imidazo(4,5-c)quinolin-4-amines | |
| Van der Pouw Kraan et al. | Histamine inhibits the production of interleukin-12 through interaction with H2 receptors. | |
| AU772179B2 (en) | Sulfonamide and sulfamide substituted imidazoquinolines | |
| EP1187613B9 (en) | Amide substituted imidazoquinolines | |
| EP1478327B1 (en) | Method of reducing and treating uvb-induced immunosuppression | |
| WO2003031587A2 (en) | Use of stat-6 inhibitors as therapeutic agents | |
| CZ9901420A3 (en) | Compounds representing modifiers of immune response when treating diseases mediated by TH2 cells and diseases derived therefrom | |
| JP2019535672A (en) | Methods of using indazole-3-carboxamides and their use as inhibitors of WNT / Β-catenin signaling pathway | |
| JP2003501473A (en) | Urea-substituted imidazoquinoline | |
| CA2071456A1 (en) | Method of treating adult t-cell leukemia/lymphoma | |
| US20040038958A1 (en) | Topical treatment of skin diseases | |
| EP0311616B1 (en) | Combinations of gamma interferons and anti-inflammatory or anti-pyretic agents for treating diseases | |
| CN115135327A (en) | Compositions and methods for preventing cancer recurrence | |
| CA2641262A1 (en) | Withacnistin compounds for treatment of cancer | |
| ZA200205287B (en) | Novel substituted pyrazolo(4,3-e)diazepines, pharmaceutical compositions containing them, use as medicinal products and processes for preparing them. | |
| WO2002045718A1 (en) | Use of active compounds capable of modulating the intracellular pathway triggered by the dp receptor in langerhans cells | |
| EP1512686B1 (en) | Imidazonaphthyridines and their use in inducing cytokine biosynthesis | |
| PASMATzI et al. | Topical application of imiquimod induces alterations in peripheral blood lymphocytes in healthy individuals | |
| EP1642580B1 (en) | Sulfonamide substituted imidazoquinolines | |
| HK40035878A (en) | Cyclic di-nucleotide compounds and methods of use | |
| HK1086762B (en) | Sulfonamide substituted imidazoquinolines | |
| JPH05938A (en) | Pneumonia preventive or therapeutic agent | |
| HK1070655B (en) | Imidazonaphthyridines and their use in inducing cytokine biosythesis |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE GH GM GW HU ID IL IS KE KG KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW SD SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 09403553 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1998917620 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1998917620 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1998917620 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: CA |