WO1994015624A1 - Use of sulphated polysaccharides for preventing sexually transmitted diseases - Google Patents
Use of sulphated polysaccharides for preventing sexually transmitted diseases Download PDFInfo
- Publication number
- WO1994015624A1 WO1994015624A1 PCT/US1994/000210 US9400210W WO9415624A1 WO 1994015624 A1 WO1994015624 A1 WO 1994015624A1 US 9400210 W US9400210 W US 9400210W WO 9415624 A1 WO9415624 A1 WO 9415624A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sulfated polysaccharide
- composition according
- transmission
- organism
- carrageenan
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/715—Polysaccharides, i.e. having more than five saccharide radicals attached to each other by glycosidic linkages; Derivatives thereof, e.g. ethers, esters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0034—Urogenital system, e.g. vagina, uterus, cervix, penis, scrotum, urethra, bladder; Personal lubricants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
Definitions
- This invention relates to a method and composition for preventing sexual transmission of sexually transmitted diseases, inter alia, the HIV virus, the causative organism of Acquired Immune Deficiency Syndrome (AIDS) , and also Chlamydia infection.
- sexually transmitted diseases inter alia, the HIV virus, the causative organism of Acquired Immune Deficiency Syndrome (AIDS) , and also Chlamydia infection.
- AIDS Acquired Immune Deficiency Syndrome
- AIDS is a catastrophic disease which leads to a failure of the immune system.
- the disease can be transmitted via infected blood and blood products, sexually transmitted, and transmitted through the placenta in utero.
- an HIV infected lymphocyte 1 containing virions V attaches to the surface of an epithelial cell 2 within a cavity 3 such as the vagina, cervix, skin of the penis or penile uretha.
- the virions shed from the lymphocyte 1 fuse with and enter the epithelial cell 2 which thus becomes infected.
- Fig. IB Fig. IB
- the virus then replicates in the epithelial cell 2 while the infected lymphocyte 1 may move on to infect other epithelial cells 2 '. (Fig. 1C) . Finally, the infected epithelial cell 1 sheds virions into the underlying connective tissue 4 where lymphocytes 5 are infected. (Fig. ID) .
- the Chlamydia organism is an obligate intracellular parasite or "quasi bacteria".
- the usual mode of human transmission is heterosexual vaginal intercourse, during which the free organism adheres to and then enters first the vaginal and cervical epithelial cells, and then subsequently the epithelial cells of the upper genital tract i.e. the uterus and Fallopian tubes. It often causes 'silent' infection with no apparent signs or symptoms, but it causes scarring of the Fallopian tubes and is one of the main causes of infertility in the human female.
- sulfated polysaccharides prevent the transmission of the Chlamydia organism.
- cell-to-cell transmission of HIV and thus the sexual transmission of AIDS, is inhibited by the direct application of sulfated polysaccharides to the epithelium of the vaginal mucosa, cervix or penis.
- the sexual transmission of Chlamydia infection is inhibited by the direct application of sulfated polysaccharides to the epithelium of the vaginal mucosa, cervix or penis.
- examples 8 and 11 In vitro studies describing this are set forth in examples 7 and 10.
- sulfated polymers include, but are not limited to, iota carrageenan and dextran sulfate, kappa carrageenan, lambda carrageenan, heparin mimetics, heparan sulfate, pentosan polysulfate, chondroitin sulfate and fucoidan.
- the sulfated polysaccharides may be formulated into a cream, suppository, gel or foam composition, and such compositions are an aspect of the invention.
- Figs. 1A-1D are a schematic representation of cell-to-cell transmission of HIV;
- Fig. 2 shows a segment of an exemplary sulfated polysaccharide;
- Fig. 3 shows the results of a blocking study to evaluate inhibition of HIV-1 infectivity
- Figs. 4-7 show the fluorescence measurements to determine the adhesion blocking ability of various materials.
- Fig. 8 shows cell adhesion results in a test employing HIV infected lymphocytes.
- This invention relates to a method of inhibiting sexually transmitted diseases, inter alia, the acquired immune deficiency syndrome and Chlamydia. It is now recognized that AIDS is caused by two lentiviruses:
- HIV-1 Human Immunodeficiency Virus type 1 (HIV-1) and type 2 (HIV-2) .
- Other names, including LAV and HTLV-III, have been previously used to identify HIV-1.
- Chlamydia infection is caused by an obligate intracellular parasite which passes into the epithelial cells of the female genital tract during vaginal intercourse.
- the invention is based on the observation that HIV is transmitted from infected lymphocytes to epithelial cells which lack the CD4 receptors normally associated with HIV transmission by cell-to-cell transfer, and the discovery that this cell-to-cell transfer can be blocked by sulfated polysaccharides.
- Sulfated polysaccharides for use in the invention are long chain polymers of sugars linked to sulfates and optionally other functional groups.
- Fig. 2 shows a segment of an exemplary sulfated polysaccharide. Sugars other than those shown in Fig. 2, other combinations of ⁇ and ⁇ linkages in the polymer and other substitutions can also be employed.
- sulfated polysaccharides for use in the invention will have a molecular weight of 5000 to 1,000,000, more preferably from 50,000 to 500,000. In addition, from 10 to 100 percent of the available binding sites are sulfated, more preferably from 50 to 100%. This percentage is referred to as the degree of sulfate substitution.
- Particular sulfated polysaccharides useful in the invention include carrageenans, chondroitin sulfate, dextran sulfate, lentinan sulfate, curdlan sulfate, heparin sulfate, and de-N-sulfated heparin. These materials are all available commercially, e.g., from Sigma Chemical Co. of St. Louis, Missouri. In addition, methods for their isolation or synthesis are published. See, e.g., Bray et al., Bioche . J. .38., 142 (1944); Hatanaka et al., J. Med. Chem. 3_0; 810-814 (1987) .
- sulfated polysaccharides are topically applied to the vaginal or rectal mucosa in an amount effective to inhibit virus transmission. Specific amounts may depend on interactions with carrier materials, but in general, application of from approximately 1.0-10.0 grams of sulfated polysaccharide per dosage is preferred.
- the preferred sulfated polysaccharides of the invention are the Carrageenans.
- Iota carrageenan is the most efficacious of the commercially available sulfated polysaccharides in preventing HIV infection and in blocking Chlamydia infection in vitro and in vivo. Because it is a large linear molecule, iota carrageenan forms a gel. Thus, no additives are needed to thicken a formulation of this polysaccharide. The simplicity of this formulation avoids potential problems which may result from interactions between the active ingredient and other components.
- the sulfated polysaccharides of the invention are advantageously formulated with a carrier.
- a carrier may be any one of the numerous creams, gels and foams known for use in vaginal or rectal lubricants or therapeutics.
- the sulfated polysaccharides will generally be present in an amount ranging from 1% to 10% by weight of the carrier.
- compositions of the invention may include additional ingredients such as agents which inhibit sperm function (e.g. nonoxynol-9, benzalkonium chloride, chloroquine, and diethyldithiol carbonate) .
- agents which inhibit sperm function e.g. nonoxynol-9, benzalkonium chloride, chloroquine, and diethyldithiol carbonate
- the sulfated polysaccharides of the invention can be used in conjunction with mechanical contraceptives including condoms, diaphragms, sponges and intrauterine devices or NORPLANT®.
- the preservatives that may be used in the formulations include any of the commonly used preservatives.
- the preferred preservative is benzoyl alcohol. Firstly, it is considered to be safe and is the most common preservative in use in OTC vaginal products in the U.S. Also, potential problems may exist when other preservatives are combined with carrageenan.
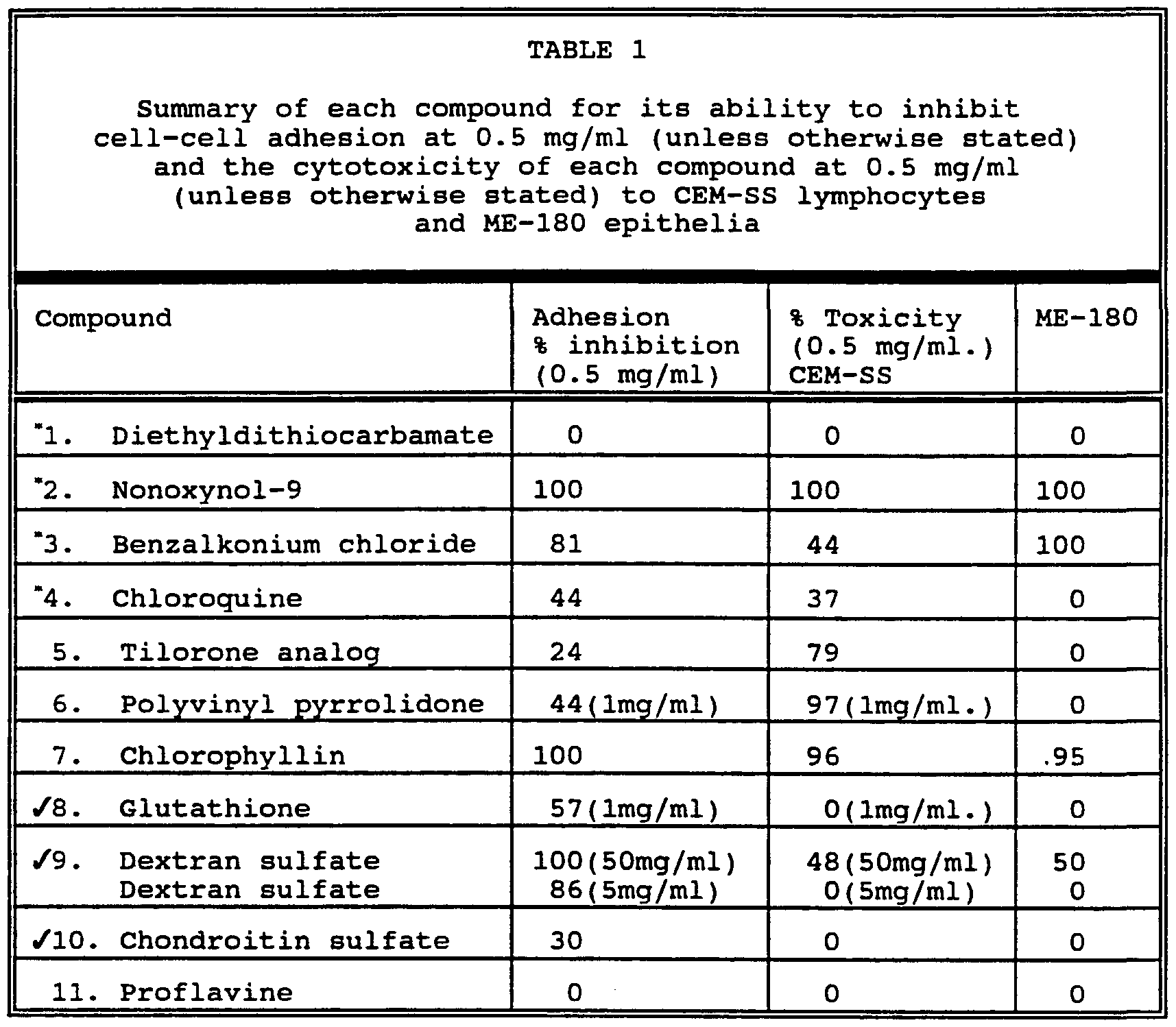
- Example 1 The ability of various sulfated polysaccharides and other materials to block adhesion of lymphocytes to epithelial cells was tested.
- the agents tested included spermicides and other compounds with properties suggesting that they were as follows: Tergitol (NP-9, Sigma Chemical Co., St. Louis, MO), commonly referred to as nonoxynol-9 (N-9) , is a nonionic surfactant employed as the active ingredient in most vaginal spermicides.
- Benzalkonium chloride (B- 1383), Sigma Chemical Co., St. Louis, MO) is a cationic surface-active agent also used in vaginal spermicides.
- Proflavine (3,6-acridinamine, P-4646, Sigma Chemical Co., St.
- Chloroquine is a quinone analog with antimalarial, antiamebic, and antirheumatic properties which is known for its suppressive effect on the immune system in lupus erythematosus.
- Polyvinyl pyrrolidone PVP-l, Sigma Chemical Co., St. Louis, MO
- Diethyldithiocarba ate (C5H10NNaS2) (D- 3506, Sigma Chemical Co., St. Louis, MOS) is a chelating agent with immunomodulating and T-cell specific immunostimulant effects.
- the tilorone analog which we used (T-8014, Sigma Chemical Co., St. Louis, MO) also has immunomodulatory effects, possibly via induction of interferon production.
- Glutathione (G-4251, Sigma Chemical Co., St. Louis, MO) is a reducing agent rich in thiol groups.
- Chlorophyllin (No. 25,828-8, Aldrich Chemical Co., Milwaukee, WI) which is an analog of chlorophyll, is a chelating agent. Chondroitin sulfate A(C-8529, Sigma Chemical Co., St.
- Dextran sulfate (D-6001, Sigma Chemical Co., St. Louis, MO) is a heparin-like sulfated polysaccharide used as an anticoagulant and is also antihyperlipoproteinemic.
- BCECF-AM (No. 1170, Molecular Probes, Eugene, OR) a fluorogenic ester readily passes through the cell membrane of viable cells and is nonfluorescent until hydrolysed by intracellular esterases.
- BCECF-AM was used to label lymphocytes and the fluorescence level was found to be directly proportional to the number of lymphocytes added to an epithelium. This fluorescence was, therefore, used to determine the level of cell adhesion.
- the ability of compounds which block adhesion of HIV infected lymphocytes to inhibit transmission of HIV to epithelial cells was tested using BeWo trophoblast- derived cells.
- the cells were maintained on RPMI-1640 medium, supplemented with 10% heat inactivated fetal bovine serum (FBS) .
- FBS heat inactivated fetal bovine serum
- the cells were plated into 6-well test plates at 10 5 cells/well and incubated in RPMI-1640 for one day. The cells were then incubated with RPMI/FBS medium alone (control), 0.5 or 5 ⁇ g/ml.
- soluble CD4 Pharmacia Piscataway, NJ
- 0.5,5 and 50 ⁇ g/ml. of dextran sulfate Sigma, St. Louis, MO
- Fig. 3 shows the results of the ELISA tests on the BeWo cells from ten sets of tests. Dextran sulfate blocked infection in a dose-dependent manner. Soluble CD4 was not shown to inhibit infection of the BeWo epithelial cells.
- Carrageenan I (CI) is a polymeric material having a molecular weight of 140,000-300,000 daltons and contains predominantly kappa carrageenan. It has the lowest sulfate substitution level of the carrageenan family, about one sulfate in each disaccharide repeating unit.
- Carrageenan II (CII) has the same molecular weight range as Carrageenan I, but contains predominantly iota carrageenan and has the highest sulfate substitution level.
- CI and CII are both non-cytotoxic at levels ⁇ 10 mg/ml.
- carrageenans are approved by the U.S. FDA for use in foods.
- CI, CII, pentosan polysulfate (a sulfated polymer with a molecular weight of 3000-4000 daltons) and mannitol were evaluated for their ability to block adhesion of lymphocytes using the fluorescence assay of Example 1. As shown in Fig. 4, both CI and CII inhibited adhesion at non-toxic concentrations of 2.5 and 5 mg/ml.
- "Co” in Fig. 4 is the control fluorescence measured for untreated cells; "Bgd” reflects the background fluorescence measurement without addition of fluorogenic lymphocytes.
- Example 5 The fluorescence-based adhesion assay of Example 1 was repeated for glutathione, dextran sulfate, Carrageenan I and Carrageenan II using HIV-infected lymphocytes. As shown in Fig. 8, all of the materials were effective inhibitors of cell adhesion, with the Carrageenans being effective at lower concentrations.
- the agents tested are compositions comprising various sulfated polysaccharides including iota carrageenan, kappa carrageenan, lambda carrageenan, dextran sulfate.
- Benzyl alcohol (Sigma, St. Louis, MO) and 6 ml. 0.1 N HCl are added to 93 ml. distilled water in a 250 ml. beaker.
- Two grams of iota carrageenan are added slowly over a 30 minute period while the mixture is continuously stirred with a spatula, at room temperature.
- HlV-infected lymphocytes are contacted with the epithelial cells in an amount effective to cause adhesion to, and transmission of the virus into, the epithelial cells.
- the same amount of HIV-infected lymphocytes together with the sulfated polysaccharide composition of Example 6 is contacted with a further same amount of epithelial cells.
- the amount of sulfated polysaccharide used is an amount effective to inhibit cell adhesion and transmission of the virus.
- Example 1 The fluorescence assay of Example 1 is used to determine the level of cell adhesion. It is found that the sulfated polysaccharide composition prevents adhesion of the HIV-infected lymphocytes to the epithelial cells and transmission of the virus.
- the ability of various sulfated polysaccharides to block adhesion of the Chlamydia organism to epithelial cells is tested.
- the human strain of the Chlamydia organism is introduced into the vagina of each of 100 mice in an amount effective to transmit the organism across the vaginal epithelial cells and infect the mice.
- the same amount of Chlamydia organism together with the sulfated polysaccharide composition described in example 6 is introduced into the vagina of each of another 100 mice.
- the amount of sulfated polysaccharide used is an amount effective to inhibit transmission of the Chlamydia organism. Specific amounts may depend on interactions with carrier materials. It is found that the sulfated polysaccharide composition prevents transmission of the Chlamydia organism. Histology of the ' reproductive tract is used to determine whether or not the animal is infected.
- Example 7 The experiment performed in Example 7 is performed in vitro using epithelial cells from the human cervix.
- the results are similar in that transmission of the Chlamydia organism to the epithelial cells does not occur in the presence of the sulfated polysaccharide composition.
- Example 9 The agents tested are compositions comprising various sulfated polysaccharides and various contraceptive compositions intended for vaginal administration.
- 1.0 ml. of Benzyl alcohol (Sigma, St. Louis, Mo) and 10.0 ml. 0.1 N HCl are added to 88 ml. distilled water in a 250 ml. beaker.
- 1.0 gram of sulfated polystyrene (ORF 13904) is added slowly, with stirring until it is in solution.
- 2.0 grams of iota carrageenan (Sigma, St. Louis, MO) are added slowly over a 30 minute period while the mixture is continuously stirred with a spatula, at room temperature.
- Example 6 The experiment performed in Example 6 is performed using the sulfated polysaccharide composition of Example 9. The results are found to be the same.
- Example 7 The experiment performed in Example 7 is performed using the sulfated polysaccharide composition of Example 9. The results are found to be the same.
- Example 8 The experiment in Example 8 is performed using the sulfated polysaccharide composition of example 9. The results are found to be the same.
- Compounds 1, 2, 3 and 4 exhibit spermicidal activity at 0.6, 0.5-1, 1 and 2.5 mg/ml respectively. Remaining compounds non-spermicidal.
- Non-toxic compounds which inhibit cell-cell adhesion.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Oncology (AREA)
- Virology (AREA)
- Communicable Diseases (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Gynecology & Obstetrics (AREA)
- Reproductive Health (AREA)
- Urology & Nephrology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
Cell-to-cell transmission of HIV, and thus the sexual transmission of AIDS, is inhibited by the direct application of sulfated polysaccharides to the epithelium of the vaginal mucosa, cervix or penis. Sulfated polysaccharides include carrageenan and dextran sulfate, which may be formulated into a cream, suppository, gel or foam composition, and such compositions are an aspect of the invention.
Description
Description
USE OF SULPHATED POLYSACCHARIDES FOR PREVENTING SEXUALLY TRANSMITTED DISEASES
Specification
This is a continuation-in-part of copending U.S. application Serial No. 08/001,795 filed in the United States Patent and Trademark Office on January 8, 1993, which is incorporated by reference herein.
Background of the Invention
This invention relates to a method and composition for preventing sexual transmission of sexually transmitted diseases, inter alia, the HIV virus, the causative organism of Acquired Immune Deficiency Syndrome (AIDS) , and also Chlamydia infection.
AIDS is a catastrophic disease which leads to a failure of the immune system. The disease can be transmitted via infected blood and blood products, sexually transmitted, and transmitted through the placenta in utero.
In the case of transmission via administration of HIV infected blood, many researchers agree that infection of helper T-lymphocytes, the cells whose infection by HIV leads to the immune system failure, proceeds when free HIV virions bind to CD-4 receptors of the surface of the cell. It has therefore been proposed that transmission of AIDS through the blood could be prevented by preventing this binding event from occurring.
Among the materials which have been proposed for this purpose are antibodies specific to CD4 receptors. In addition, there have been a number of reports that dextran sulfate and other sulfated polymers are able to
block binding of free HIV virions to CD4 receptors in vitro, Ito et al., Antiviral Research 1_, 361-367 (1987). Mitsuya et al., Science 240. 646-649 (1988); Ueno et al., The Lancet, 1379 (1987); Baba et al., Antimicrobial Agents and Chemotherapy .34., 134-138 (1990); Busso et al. , Antimicrobial Agents and Chemotherapy J34., 1991-1995 (1990); Scholes et al., Virology 175. 5560561 (1990); Aoki et al., AIDS research and Human Retroviruses 2, 409-415 (1991) . Sulfated polysaccharides have also been reported to inhibit other enveloped viruses, including herpes simplex virus. Baba et al., Antimicrobial Agents and Chemotherapy 3_2., 1742-1745 (1988) . Sulfated polysaccharides have also been proposed for oral administration with the hope of slowing the progress of the disease in an infected individual by inhibiting the infection of further lymphocytes. Abrams et al., Ann. Internal Medicine 110. 183-188 (1989); Hanaue et al., Clinical Therapeutics OL; 6140622 (1989) . Prior to the claimed invention it was believed that infection with the HIV virus can occur only via a breach or lesion in the mucous membrane, and that conversely there could be no transmission of virus through an intact mucous membrane. Surprisingly, it has now been found that the virus can be transmitted through an intact mucous membrane.
While the transmission of HIV via blood and blood products can be understood based upon the binding of free virions to CD4 receptors, this same explanation cannot explain sexual and particularly cervical transmission of HIV because the epithelial cells of the mucous membrane lining the vagina and the intestine do not have CD4 receptors. Instead, it now appears that sexual transmission of HIV occurs, to a significant degree, by cell-to-cell infection, via phagocytosis which is a non-specific process that does not involve a
specific HIV receptor. See. Levy, J. Amer. Med. Assn. 259, 3037-8 (1988); Phillips et al., Virology 186. 261-273 (1992) . This mode of transmission is illustrated diagrammatically in Figs IA -ID. As shown in Fig. IA, an HIV infected lymphocyte 1 containing virions V attaches to the surface of an epithelial cell 2 within a cavity 3 such as the vagina, cervix, skin of the penis or penile uretha. The virions shed from the lymphocyte 1 fuse with and enter the epithelial cell 2 which thus becomes infected. (Fig. IB) .
The virus then replicates in the epithelial cell 2 while the infected lymphocyte 1 may move on to infect other epithelial cells 2 '. (Fig. 1C) . Finally, the infected epithelial cell 1 sheds virions into the underlying connective tissue 4 where lymphocytes 5 are infected. (Fig. ID) .
The Chlamydia organism is an obligate intracellular parasite or "quasi bacteria". The usual mode of human transmission is heterosexual vaginal intercourse, during which the free organism adheres to and then enters first the vaginal and cervical epithelial cells, and then subsequently the epithelial cells of the upper genital tract i.e. the uterus and Fallopian tubes. It often causes 'silent' infection with no apparent signs or symptoms, but it causes scarring of the Fallopian tubes and is one of the main causes of infertility in the human female. Until the present invention, it was not known that sulfated polysaccharides prevent the transmission of the Chlamydia organism.
It is the object of the present invention to provide a method and a composition for blocking the cell-to-cell transmission of HIV virus, and also for inhibiting sexual transmission of the Chlamydia organism.
Summary of the Invention
In accordance with the invention, cell-to-cell transmission of HIV, and thus the sexual transmission of AIDS, is inhibited by the direct application of sulfated polysaccharides to the epithelium of the vaginal mucosa, cervix or penis. Also, in accordance with the invention, the sexual transmission of Chlamydia infection, is inhibited by the direct application of sulfated polysaccharides to the epithelium of the vaginal mucosa, cervix or penis. In vitro studies describing this are set forth in examples 8 and 11. In vivo studies describing this are set forth in examples 7 and 10. These sulfated polymers include, but are not limited to, iota carrageenan and dextran sulfate, kappa carrageenan, lambda carrageenan, heparin mimetics, heparan sulfate, pentosan polysulfate, chondroitin sulfate and fucoidan. The sulfated polysaccharides may be formulated into a cream, suppository, gel or foam composition, and such compositions are an aspect of the invention.
Brief Description of the Drawings
Figs. 1A-1D are a schematic representation of cell-to-cell transmission of HIV; Fig. 2 shows a segment of an exemplary sulfated polysaccharide;
Fig. 3 shows the results of a blocking study to evaluate inhibition of HIV-1 infectivity;
Figs. 4-7 show the fluorescence measurements to determine the adhesion blocking ability of various materials; and
Fig. 8 shows cell adhesion results in a test employing HIV infected lymphocytes.
Detailed Description of the Invention
This invention relates to a method of inhibiting sexually transmitted diseases, inter alia, the acquired immune deficiency syndrome and Chlamydia. It is now recognized that AIDS is caused by two lentiviruses:
Human Immunodeficiency Virus type 1 (HIV-1) and type 2 (HIV-2) . Other names, including LAV and HTLV-III, have been previously used to identify HIV-1. Chlamydia infection is caused by an obligate intracellular parasite which passes into the epithelial cells of the female genital tract during vaginal intercourse.
Because it was believed, as discussed above, that there could be no transmission of the HIV virus through an intact mucous membrane, sulfated polysaccharides were not believed to have any effect where HIV viruses were disposed on the epithelial cells of an intact mucous membrane. Surprisingly, sulfated polysaccharides applied topically are now found to be effective in blocking infection of the epithelial cells. Thus, the invention is based on the observation that HIV is transmitted from infected lymphocytes to epithelial cells which lack the CD4 receptors normally associated with HIV transmission by cell-to-cell transfer, and the discovery that this cell-to-cell transfer can be blocked by sulfated polysaccharides.
Sulfated polysaccharides for use in the invention are long chain polymers of sugars linked to sulfates and optionally other functional groups. Fig. 2 shows a segment of an exemplary sulfated polysaccharide. Sugars other than those shown in Fig. 2, other combinations of α and β linkages in the polymer and other substitutions can also be employed.
In general, sulfated polysaccharides for use in the invention will have a molecular weight of 5000 to 1,000,000, more preferably from 50,000 to 500,000. In addition, from 10 to 100 percent of the available
binding sites are sulfated, more preferably from 50 to 100%. This percentage is referred to as the degree of sulfate substitution.
Particular sulfated polysaccharides useful in the invention include carrageenans, chondroitin sulfate, dextran sulfate, lentinan sulfate, curdlan sulfate, heparin sulfate, and de-N-sulfated heparin. These materials are all available commercially, e.g., from Sigma Chemical Co. of St. Louis, Missouri. In addition, methods for their isolation or synthesis are published. See, e.g., Bray et al., Bioche . J. .38., 142 (1944); Hatanaka et al., J. Med. Chem. 3_0; 810-814 (1987) .
In accordance with the invention, sulfated polysaccharides are topically applied to the vaginal or rectal mucosa in an amount effective to inhibit virus transmission. Specific amounts may depend on interactions with carrier materials, but in general, application of from approximately 1.0-10.0 grams of sulfated polysaccharide per dosage is preferred.
The preferred sulfated polysaccharides of the invention are the Carrageenans. Iota carrageenan is the most efficacious of the commercially available sulfated polysaccharides in preventing HIV infection and in blocking Chlamydia infection in vitro and in vivo. Because it is a large linear molecule, iota carrageenan forms a gel. Thus, no additives are needed to thicken a formulation of this polysaccharide. The simplicity of this formulation avoids potential problems which may result from interactions between the active ingredient and other components.
In the formulation used in Examples 6-8, a concentration of about 2% is chosen, as at this concentration it compares favorably with commercially available sexual lubricants such as K-Y jelly.
A great deal of research has been done on carrageenan and it has been found to be remarkably safe with few, if any, side-effects. It is FDA-approved for use in foods and cosmetics, and is not absorbed by the epithelium of the gastro-intestinal tract. Carrageenan is a reasonably strong buffer, and the pH can be adjusted without the use of an additional buffer in formulations containing carrageenan. Although the pH of the human vagina is usually 4-5, a pH of 6 is chosen as carrageenan is unstable and therefore less efficacious at a lower pH.
The sulfated polysaccharides of the invention are advantageously formulated with a carrier. Such a carrier may be any one of the numerous creams, gels and foams known for use in vaginal or rectal lubricants or therapeutics. The sulfated polysaccharides will generally be present in an amount ranging from 1% to 10% by weight of the carrier.
The advantage of a composition comprising a sulfated polysaccharide and a contraceptive is the prevention of conception while simultaneously inhibiting infection due to sexually transmitted diseases. Thus, the compositions of the invention may include additional ingredients such as agents which inhibit sperm function (e.g. nonoxynol-9, benzalkonium chloride, chloroquine, and diethyldithiol carbonate) . In addition, the sulfated polysaccharides of the invention can be used in conjunction with mechanical contraceptives including condoms, diaphragms, sponges and intrauterine devices or NORPLANT®.
The preservatives that may be used in the formulations include any of the commonly used preservatives. The preferred preservative is benzoyl alcohol. Firstly, it is considered to be safe and is the most common preservative in use in OTC vaginal products in the U.S. Also, potential problems may
exist when other preservatives are combined with carrageenan.
Example 1 The ability of various sulfated polysaccharides and other materials to block adhesion of lymphocytes to epithelial cells was tested. The agents tested included spermicides and other compounds with properties suggesting that they were as follows: Tergitol (NP-9, Sigma Chemical Co., St. Louis, MO), commonly referred to as nonoxynol-9 (N-9) , is a nonionic surfactant employed as the active ingredient in most vaginal spermicides. Benzalkonium chloride (B- 1383), Sigma Chemical Co., St. Louis, MO) is a cationic surface-active agent also used in vaginal spermicides. Proflavine (3,6-acridinamine, P-4646, Sigma Chemical Co., St. Louis, MO), which is used as a topical antiseptic, has been known to terminate DNA synthesis. Chloroquine is a quinone analog with antimalarial, antiamebic, and antirheumatic properties which is known for its suppressive effect on the immune system in lupus erythematosus. Polyvinyl pyrrolidone (PVP-l, Sigma Chemical Co., St. Louis, MO) is used as an antiseptic. Diethyldithiocarba ate (C5H10NNaS2) (D- 3506, Sigma Chemical Co., St. Louis, MOS) is a chelating agent with immunomodulating and T-cell specific immunostimulant effects. It is also used as an antidote to nickel and cadmium poisoning. The tilorone analog which we used (T-8014, Sigma Chemical Co., St. Louis, MO) also has immunomodulatory effects, possibly via induction of interferon production. Glutathione (G-4251, Sigma Chemical Co., St. Louis, MO) is a reducing agent rich in thiol groups. Chlorophyllin (No. 25,828-8, Aldrich Chemical Co., Milwaukee, WI) which is an analog of chlorophyll, is a chelating agent. Chondroitin sulfate A(C-8529, Sigma
Chemical Co., St. Louis, Mo) is a highly viscous mucopolysaccharide which occurs in both skeletal and soft tissue and has antiihyperlipoproteinemic activity. Dextran sulfate (D-6001, Sigma Chemical Co., St. Louis, MO) is a heparin-like sulfated polysaccharide used as an anticoagulant and is also antihyperlipoproteinemic.
BCECF-AM, (No. 1170, Molecular Probes, Eugene, OR) a fluorogenic ester readily passes through the cell membrane of viable cells and is nonfluorescent until hydrolysed by intracellular esterases. BCECF-AM was used to label lymphocytes and the fluorescence level was found to be directly proportional to the number of lymphocytes added to an epithelium. This fluorescence was, therefore, used to determine the level of cell adhesion. These results are summarized in Table 1. As can be seen, dextran sulfate and chondroitin sulfate, as well as glutathione, inhibited adhesion at nontoxic levels.
Example 2
The ability of compounds which block adhesion of HIV infected lymphocytes to inhibit transmission of HIV to epithelial cells was tested using BeWo trophoblast- derived cells. The cells were maintained on RPMI-1640 medium, supplemented with 10% heat inactivated fetal bovine serum (FBS) . The cells were plated into 6-well test plates at 105 cells/well and incubated in RPMI-1640 for one day. The cells were then incubated with RPMI/FBS medium alone (control), 0.5 or 5μg/ml. soluble CD4 (Pharmacia Piscataway, NJ) or 0.5,5 and 50μg/ml. of dextran sulfate (Sigma, St. Louis, MO) for 1 hour. 10° HIV-infected MOLT-4 lymphocytes, pretreated with 150/xg/ml. mitomycin C (Sigma, St. Louis, MO) , were then added to each well. After three hours, the lymphocytes were removed by repeated washings. The BeWo cells were then grown for 8 days, changing the medium daily. On
the eighth day, the supernatants were tested for HIV by ELISA using an HIV-1 p24 antigen detection kit (Coulter, Hialeah, FL) .
Fig. 3 shows the results of the ELISA tests on the BeWo cells from ten sets of tests. Dextran sulfate blocked infection in a dose-dependent manner. Soluble CD4 was not shown to inhibit infection of the BeWo epithelial cells.
Example 3
Carrageenan I (CI) is a polymeric material having a molecular weight of 140,000-300,000 daltons and contains predominantly kappa carrageenan. It has the lowest sulfate substitution level of the carrageenan family, about one sulfate in each disaccharide repeating unit.
Carrageenan II (CII) has the same molecular weight range as Carrageenan I, but contains predominantly iota carrageenan and has the highest sulfate substitution level.
CI and CII are both non-cytotoxic at levels <10 mg/ml. In fact, carrageenans are approved by the U.S. FDA for use in foods.
CI, CII, pentosan polysulfate (a sulfated polymer with a molecular weight of 3000-4000 daltons) and mannitol were evaluated for their ability to block adhesion of lymphocytes using the fluorescence assay of Example 1. As shown in Fig. 4, both CI and CII inhibited adhesion at non-toxic concentrations of 2.5 and 5 mg/ml. "Co" in Fig. 4 is the control fluorescence measured for untreated cells; "Bgd" reflects the background fluorescence measurement without addition of fluorogenic lymphocytes.
Example 4
Fluorescence-based adhesion assays were conducted on fucoidan (MW=1000,000 Da), heparin (MW=16,000), heparin sulfate (MW=16,000 Da), De-N-sulfated heparin (MW=15,000-18,000 Da) and chondroitin sulfate. Fucoidan and heparin did not inhibit adhesion at concentrations as high as 5.0 mg/ml. Heparin sulfate inhibited cell adhesion by 42% at a concentration of .25 mg/ml. (Fig. 5). De-N-sulfated heparin inhibited cell-cell adhesion by 52% at a concentration of 6.25 mg/ml. (Fig. 6). Chondroitin sulfate inhibited cell- cell adhesion by 30% at 4 mg/ml. (Fig. 7) .
Example 5 The fluorescence-based adhesion assay of Example 1 was repeated for glutathione, dextran sulfate, Carrageenan I and Carrageenan II using HIV-infected lymphocytes. As shown in Fig. 8, all of the materials were effective inhibitors of cell adhesion, with the Carrageenans being effective at lower concentrations.
Example 6
The ability of various sulfated polysaccharides to block adhesion of lymphocytes to epithelial cells are tested, using HIV-infected lymphocytes and epithelial cells from the human cervix.
The agents tested are compositions comprising various sulfated polysaccharides including iota carrageenan, kappa carrageenan, lambda carrageenan, dextran sulfate.
In a preferred formulation, 1 ml. Benzyl alcohol (Sigma, St. Louis, MO) and 6 ml. 0.1 N HCl are added to 93 ml. distilled water in a 250 ml. beaker. Two grams of iota carrageenan are added slowly over a 30 minute period while the mixture is continuously stirred with a spatula, at room temperature.
HlV-infected lymphocytes are contacted with the epithelial cells in an amount effective to cause adhesion to, and transmission of the virus into, the epithelial cells. These preparations serve as controls. The same amount of HIV-infected lymphocytes together with the sulfated polysaccharide composition of Example 6 is contacted with a further same amount of epithelial cells. The amount of sulfated polysaccharide used is an amount effective to inhibit cell adhesion and transmission of the virus.
The fluorescence assay of Example 1 is used to determine the level of cell adhesion. It is found that the sulfated polysaccharide composition prevents adhesion of the HIV-infected lymphocytes to the epithelial cells and transmission of the virus.
Example 7
The ability of various sulfated polysaccharides to block adhesion of the Chlamydia organism to epithelial cells is tested. In controls, the human strain of the Chlamydia organism is introduced into the vagina of each of 100 mice in an amount effective to transmit the organism across the vaginal epithelial cells and infect the mice. The same amount of Chlamydia organism together with the sulfated polysaccharide composition described in example 6 is introduced into the vagina of each of another 100 mice. The amount of sulfated polysaccharide used is an amount effective to inhibit transmission of the Chlamydia organism. Specific amounts may depend on interactions with carrier materials. It is found that the sulfated polysaccharide composition prevents transmission of the Chlamydia organism. Histology of the' reproductive tract is used to determine whether or not the animal is infected.
Example 8
The experiment performed in Example 7 is performed in vitro using epithelial cells from the human cervix.
The results are similar in that transmission of the Chlamydia organism to the epithelial cells does not occur in the presence of the sulfated polysaccharide composition.
Example 9 The agents tested are compositions comprising various sulfated polysaccharides and various contraceptive compositions intended for vaginal administration. 1.0 ml. of Benzyl alcohol (Sigma, St. Louis, Mo) and 10.0 ml. 0.1 N HCl are added to 88 ml. distilled water in a 250 ml. beaker. 1.0 gram of sulfated polystyrene (ORF 13904) is added slowly, with stirring until it is in solution. 2.0 grams of iota carrageenan (Sigma, St. Louis, MO) are added slowly over a 30 minute period while the mixture is continuously stirred with a spatula, at room temperature.
The experiment performed in Example 6 is performed using the sulfated polysaccharide composition of Example 9. The results are found to be the same.
Example 10
The experiment performed in Example 7 is performed using the sulfated polysaccharide composition of Example 9. The results are found to be the same.
Example 11
The experiment in Example 8 is performed using the sulfated polysaccharide composition of example 9. The results are found to be the same.
Compounds 1, 2, 3 and 4 exhibit spermicidal activity at 0.6, 0.5-1, 1 and 2.5 mg/ml respectively. Remaining compounds non-spermicidal.
Non-toxic compounds which inhibit cell-cell adhesion.
Claims
1. A method for inhibiting sexual transmission of the virus causing acquired immunodeficiency syndrome comprising topically applying to the vagina or penis a sulfated polysaccharide that blocks adhesion of lymphocytes to epithelial cells of a mucosa in a region of sexual contact in an amount effective to inhibit transmission of the virus.
2. A method according to claim 1 or 19, wherein the sulfated polysaccharide has a molecular weight of from 5,000 to 1,000,000 and a degree of sulfate substitution of from 10 to 100%.
3. A method according to claim 1 or 19, wherein the sulfated polysaccharide is a carrageenan.
4. A method according to claim 1 or 19, wherein the sulfated polysaccharide is dextran sulfate.
5. A composition for inhibiting transmission of the virus causing acquired immunodeficiency syndrome comprising: (a) a sulfated polysaccharide that blocks adhesion of lymphocytes to epithelial cells; and (b) a carrier selected from the group consisting of cremes, gels, foams and foaming suppositories.
6. A composition according to claim 5 or 20, wherein the carrier is a foaming suppository.
7. A composition according to claim 5 or 20, wherein the composition contains from 1 to 10% by weight of the sulfated polysaccharide.
8. A composition according to claim 5 or 20, wherein the sulfated polysaccharide is a carrageenan.
9. A composition according to claim 5 or 20, wherein the sulfated polysaccharide is dextran sulfate.
10. A composition according to claim 5 or 20, wherein the sulfated polysaccharide has a molecular weight of from 5,000 to 1,000,000 and a degree of sulfate substitution of from 10 to 100%.
11. A composition according to claim 5 or 20, further comprising an agent which inhibits sperm function.
12. A composition according to claim 7, wherein the sulfated polysaccharide is a carrageenan.
13. A composition according to claim 7, wherein the sulfated polysaccharide is dextran sulfate.
14. A composition according to claim 7, wherein the sulfated polysaccharide has a molecular weight of from 5,000 to 1,000,000 and a degree of sulfate substitution of from 10 to 100%.
15. A composition according to claim 7, further comprising an agent that inhibits sperm function.
16. A composition according to claim 11, wherein the sulfated polysaccharide is a carrageenan.
17. A composition according to claim 11, wherein the sulfated polysaccharide is dextran sulfate.
18. A composition according to claim 11, wherein the sulfated polysaccharide has a molecular weight of from 5,000 to 1,000,000 and a degree of sulfate substitution of from 10 to 100%.
19. A method for inhibiting sexual transmission of the organism causing Chlamydia infection comprising topically applying a sulfated polysaccharide that blocks adhesion of the organism to, and entry of the organism into, epithelial cells of a mucosa in a region of sexual contact in an amount effective to inhibit transmission of the organism.
20. A composition of inhibiting transmission of the Chlamydia organism comprising: (a) a sulfated polysaccharide that blocks adhesion of the organism to, and entry of the organism into, epithelial cells; and (b) a carrier selected from the group consisting of creams, gels, foams and foaming suppositories.
21. A composition according to Claim 20, wherein the sulfated polysaccharide is a carrageenan.
22. A composition according to Claim 20 or 21, further comprising an agent that inhibits sperm function.
23. A composition according to Claim 20, wherein the sulfated polysaccharide has a molecular weight of from 5,000 to 1,000,000 and a degree of sulfate substitution of from 10 to 100%.
24. A composition according to Claim 22, wherein the sulfated polysaccharide has a molecular weight of from 5,000 to 1,000,000 and a degree of sulfate substitution of from 10 to 100%.
25. A method according to claim 1 or 19, wherein the mucosa is intact.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP94909436A EP0678030A1 (en) | 1993-01-08 | 1994-01-06 | Use of sulphated polysaccharides for preventing sexually transmitted diseases |
| JP6516216A JPH08506570A (en) | 1993-01-08 | 1994-01-06 | Use of sulfated polysaccharides to prevent sexually transmitted diseases |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US179593A | 1993-01-08 | 1993-01-08 | |
| US08/001,795 | 1993-01-08 | ||
| US17379093A | 1993-12-27 | 1993-12-27 | |
| US08/173,790 | 1993-12-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1994015624A1 true WO1994015624A1 (en) | 1994-07-21 |
Family
ID=26669479
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1994/000210 Ceased WO1994015624A1 (en) | 1993-01-08 | 1994-01-06 | Use of sulphated polysaccharides for preventing sexually transmitted diseases |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP0678030A1 (en) |
| JP (1) | JPH08506570A (en) |
| WO (1) | WO1994015624A1 (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5658893A (en) * | 1995-03-29 | 1997-08-19 | Abbott Laboratories | Method for inhibition of rotavirus infection with carrageenan |
| WO1998019663A1 (en) * | 1996-11-04 | 1998-05-14 | Apr Applied Pharma Research S.A. | Compositions for topical use containing polysaccharide sulphates |
| WO2000006133A3 (en) * | 1998-07-28 | 2000-08-17 | Neurochem Inc | Compositions for treating diseases associated with glycosaminoglycan-associated molecular interactions |
| WO2003055498A1 (en) * | 2001-12-24 | 2003-07-10 | Ml Laboratories Plc | Composition comprising dextrinsulfate for the treatment of sexual transmitted diseases (std) |
| EP1609467A1 (en) * | 1998-07-28 | 2005-12-28 | Neurochem (International) Limited | Compositions for treating diseases associated with glycosaminoglycan-associated molecular interactions |
| EP1764088A1 (en) * | 2005-08-24 | 2007-03-21 | Stephen B. Roman | Thixotropic personal lubricant containing carageenan |
| CN100494222C (en) * | 2006-04-30 | 2009-06-03 | 华南理工大学 | Preparation method of beta-1,4-glucan-6,2,3-sulfate |
| US20110059919A1 (en) * | 2007-08-24 | 2011-03-10 | Marinomed Biotechnologie Gmbh | Antiviral composition comprising a sulfated polysaccharide |
| WO2011044034A1 (en) | 2009-10-06 | 2011-04-14 | The Population Council, Inc. | Carrageenan-containing aqueous antimicrobial compositions |
| US8232371B2 (en) | 2004-02-02 | 2012-07-31 | Ambrx, Inc. | Modified human interferon polypeptides and their uses |
| WO2014123880A1 (en) | 2013-02-05 | 2014-08-14 | The Population Council, Inc. | Intravaginal ring for the delivery of unique combinations of antimicrobial compositions |
| US8999345B2 (en) | 1992-07-24 | 2015-04-07 | Vical Incorporated | Compositions and methods for enhancing immune responses to vaccines |
| WO2015116375A1 (en) | 2014-01-28 | 2015-08-06 | The Population Council, Inc. | Combination product for the prevention of sexually transmitted infections |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4633233B2 (en) * | 2000-06-29 | 2011-02-16 | 生化学工業株式会社 | Chlamydia infection treatment |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0255902A1 (en) * | 1986-08-07 | 1988-02-17 | Tofco Sa | Composition for topical use |
| JPH02178230A (en) * | 1988-12-28 | 1990-07-11 | Ueno Seiyaku Oyo Kenkyusho:Kk | Suppository |
-
1994
- 1994-01-06 WO PCT/US1994/000210 patent/WO1994015624A1/en not_active Ceased
- 1994-01-06 EP EP94909436A patent/EP0678030A1/en not_active Withdrawn
- 1994-01-06 JP JP6516216A patent/JPH08506570A/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0255902A1 (en) * | 1986-08-07 | 1988-02-17 | Tofco Sa | Composition for topical use |
| JPH02178230A (en) * | 1988-12-28 | 1990-07-11 | Ueno Seiyaku Oyo Kenkyusho:Kk | Suppository |
Non-Patent Citations (5)
| Title |
|---|
| DATABASE WPI Week 8807, Derwent World Patents Index; AN 88-043995 * |
| DATABASE WPI Week 9034, Derwent World Patents Index; AN 90-256414 * |
| PEARCE-PRATT, R. ET AL: "STUDIES OF ADHESION OF LYMPHOCYTIC CELLS: IMPLICATIONS FOR SEXUAL TRANSMISSION OF HUMAN IMMUNODEFICIENCY VIRUS", BIOLOGY OF REPRODUCTION, vol. 48, no. 3, March 1993 (1993-03-01), pages 431 - 445 * |
| PHILLIPS, D.M. ET AL: "MECHANISM OF HIV SPREAD FROM LYMPHOCYTES TO EPITHELIA", VIROLOGY, vol. 186, no. 1, January 1992 (1992-01-01), pages 261 - 273 * |
| SCMIEL, D.H. ET AL: "RECOMBINANT ESCHERICHIA COLI CLONES EXPRESSING CHLAMYDIA TRACHOMATIS GENE PRODUCTS ATTACH TO HUMAN ENDOMETRIAL EPITHELIAL CELLS", INFECTION AND IMMUNITY, vol. 59, no. 11, November 1991 (1991-11-01), pages 4001 - 4012 * |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8999345B2 (en) | 1992-07-24 | 2015-04-07 | Vical Incorporated | Compositions and methods for enhancing immune responses to vaccines |
| US9102950B2 (en) | 1992-07-24 | 2015-08-11 | Vical Incorporated | Compositions and methods for enhancing immune responses to vaccines |
| US5658893A (en) * | 1995-03-29 | 1997-08-19 | Abbott Laboratories | Method for inhibition of rotavirus infection with carrageenan |
| WO1998019663A1 (en) * | 1996-11-04 | 1998-05-14 | Apr Applied Pharma Research S.A. | Compositions for topical use containing polysaccharide sulphates |
| WO2000006133A3 (en) * | 1998-07-28 | 2000-08-17 | Neurochem Inc | Compositions for treating diseases associated with glycosaminoglycan-associated molecular interactions |
| US6310073B1 (en) | 1998-07-28 | 2001-10-30 | Queen's University At Kingston | Methods and compositions to treat glycosaminoglycan-associated molecular interactions |
| EP1609467A1 (en) * | 1998-07-28 | 2005-12-28 | Neurochem (International) Limited | Compositions for treating diseases associated with glycosaminoglycan-associated molecular interactions |
| WO2003055498A1 (en) * | 2001-12-24 | 2003-07-10 | Ml Laboratories Plc | Composition comprising dextrinsulfate for the treatment of sexual transmitted diseases (std) |
| US8367098B2 (en) | 2002-04-30 | 2013-02-05 | The Population Council, Inc. | Unique combinations of antimicrobial compositions |
| US8232371B2 (en) | 2004-02-02 | 2012-07-31 | Ambrx, Inc. | Modified human interferon polypeptides and their uses |
| EP1764088A1 (en) * | 2005-08-24 | 2007-03-21 | Stephen B. Roman | Thixotropic personal lubricant containing carageenan |
| CN100494222C (en) * | 2006-04-30 | 2009-06-03 | 华南理工大学 | Preparation method of beta-1,4-glucan-6,2,3-sulfate |
| US20110059919A1 (en) * | 2007-08-24 | 2011-03-10 | Marinomed Biotechnologie Gmbh | Antiviral composition comprising a sulfated polysaccharide |
| US10342820B2 (en) | 2007-08-24 | 2019-07-09 | Marinomed Biotech Ag | Antiviral composition comprising a sulfated polysaccharide |
| WO2011044034A1 (en) | 2009-10-06 | 2011-04-14 | The Population Council, Inc. | Carrageenan-containing aqueous antimicrobial compositions |
| WO2014123880A1 (en) | 2013-02-05 | 2014-08-14 | The Population Council, Inc. | Intravaginal ring for the delivery of unique combinations of antimicrobial compositions |
| WO2015116375A1 (en) | 2014-01-28 | 2015-08-06 | The Population Council, Inc. | Combination product for the prevention of sexually transmitted infections |
| US10143721B2 (en) | 2014-01-28 | 2018-12-04 | The Population Council, Inc. | Combination product for the prevention of sexually transmitted infections |
| US11331370B2 (en) | 2014-01-28 | 2022-05-17 | The Population Council, Inc. | Combination product for the prevention of sexually transmitted infections |
Also Published As
| Publication number | Publication date |
|---|---|
| JPH08506570A (en) | 1996-07-16 |
| EP0678030A1 (en) | 1995-10-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2305331C (en) | Methods for decreasing the frequency of transmission of viral and for preventing and treating bacterial infections using cellulose acetate phthalate or hyroxypropyl methylcellulose phthalate excipients | |
| WO1994015624A1 (en) | Use of sulphated polysaccharides for preventing sexually transmitted diseases | |
| US5545401A (en) | Antiviral, spermicidal vaginal gel and foam containing low molecular weight povidone-iodine | |
| ES2357474T3 (en) | COMPOSITIONS AND ITS USE TO CATCH AND INACTIVATE PATHOGEN MICROBES AND SpermATOZIODS. | |
| Zaneveld et al. | Efficacy and safety of a new vaginal contraceptive antimicrobial formulation containing high molecular weight poly (sodium 4-styrenesulfonate) | |
| US8552064B2 (en) | Suramin and derivatives thereof as topical microbicide and contraceptive | |
| ES2550815T3 (en) | Aqueous antimicrobial compositions containing carrageenans | |
| EA011298B1 (en) | Use of microbicidal pyrimidines for preventing sexual hiv transmission and pharmaceutical compositions based thereon | |
| JP2004506482A (en) | Non-hormonal vaginal contraceptive device | |
| Uckun et al. | Prophylactic contraceptives for HIV/AIDS | |
| US20020151521A1 (en) | Universal antiviral composition | |
| Haineault et al. | Thermoreversible gel formulation containing sodium lauryl sulfate as a potential contraceptive device | |
| Anderson et al. | Contraception by Ushercell™(cellulose sulfate) in formulation: duration of effect and dose effectiveness | |
| Carlin et al. | Women, contraception and STDs including HIV | |
| US8518434B2 (en) | Antiseptic spermicidal composition and means for its application | |
| Cohen | Women: absent term in the AIDS research equation | |
| Mauck et al. | An update on vaginal microbicides | |
| Ladipo et al. | A new vaginal antimicrobial contraceptive formulation: phase I clinical pilot studies | |
| Malcolm et al. | Vaginal microbicides for the prevention of HIV transmission | |
| AP868A (en) | Pharmaceutical compositions for the prevention or treatment of retrovirus infections. | |
| JP4350805B2 (en) | Condensation polymer composition for contraceptive use | |
| Scholand et al. | Anti-HIV-1 microbicides-'Chemical condoms' designed to limit the scourge of the HIV-1 pandemic | |
| Sitruk-Ware et al. | Pharmacokinetic study to compare the absorption and tolerability of two doses of levonorgestrel following single vaginal administration of levonorgestrel in Carraguard® gel: a new formulation for “dual protection” contraception | |
| JPH11503745A (en) | 2,4-Dichlorobenzyl alcohol and amyl methcresol for HIV infection | |
| Bassol et al. | Comparative trial between two soft jelly capsules containing nonoxynol as spermicidal contraceptives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1994909436 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1994909436 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1994909436 Country of ref document: EP |