WO1994012530A1 - Polyunsaturated fatty acyl-peptide composition - Google Patents
Polyunsaturated fatty acyl-peptide composition Download PDFInfo
- Publication number
- WO1994012530A1 WO1994012530A1 PCT/HU1993/000065 HU9300065W WO9412530A1 WO 1994012530 A1 WO1994012530 A1 WO 1994012530A1 HU 9300065 W HU9300065 W HU 9300065W WO 9412530 A1 WO9412530 A1 WO 9412530A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- peptide
- fatty acyl
- seq
- dha
- hormone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K7/00—Peptides having 5 to 20 amino acids in a fully defined sequence; Derivatives thereof
- C07K7/04—Linear peptides containing only normal peptide links
- C07K7/06—Linear peptides containing only normal peptide links having 5 to 11 amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/542—Carboxylic acids, e.g. a fatty acid or an amino acid
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
- C07K1/107—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length by chemical modification of precursor peptides
- C07K1/1072—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length by chemical modification of precursor peptides by covalent attachment of residues or functional groups
- C07K1/1077—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length by chemical modification of precursor peptides by covalent attachment of residues or functional groups by covalent attachment of residues other than amino acids or peptide residues, e.g. sugars, polyols, fatty acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/635—Parathyroid hormone, i.e. parathormone; Parathyroid hormone-related peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/655—Somatostatins
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/78—Connective tissue peptides, e.g. collagen, elastin, laminin, fibronectin, vitronectin or cold insoluble globulin [CIG]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K7/00—Peptides having 5 to 20 amino acids in a fully defined sequence; Derivatives thereof
- C07K7/04—Linear peptides containing only normal peptide links
- C07K7/08—Linear peptides containing only normal peptide links having 12 to 20 amino acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K7/00—Peptides having 5 to 20 amino acids in a fully defined sequence; Derivatives thereof
- C07K7/04—Linear peptides containing only normal peptide links
- C07K7/23—Luteinising hormone-releasing hormone [LHRH]; Related peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
Definitions
- the present invention relates to polyunsaturated fatty acyl-peptide composition useful in inhibiting cell proliferation.
- Uncontrolled cell proliferation is a characteristic of a number of diseased states. Such growth is observed, for example, in benign and malignant tumors, certain virally- induced diseases and psoriasis.
- drugs used to treat cellular abnormalities characterized by uncontrolled cell growth target important biochemical steps or processes that are part of the cell growth cycle. However, ⁇ such drugs lack selectivity and inhibit the growth of both diseased and healthy cells. Therefore, development of chemotherapeutic agents having relatively high selectivity for the diseased cells would be advantageous.
- Agents based on peptide hormones, steroid hormones, hormone-releasing factors, and their respective antagonists and agonists are relatively specific to their target cells.
- Proliferative cells such as neoplastic cells or tumors, which arise from hormone-sensitive tissues generally are found to have hormonal requirements that are similar to those of their healthy counterparts. By altering the amount of hormone in the blood circulation it may be possible to selectively restrict the growth of these cells. However, due to the relatively high levels of hormone required for such treatment, use of hormonal chemotherapeutic agents is still limited in vivo by toxic side effects to normal cells .
- cell proliferation may also be inhibited by targeting one or more of the cellular signal transduction systems implicated in the regulation of cell division.
- tyrosine kinase signal transduction pathway a) the tyrosine kinase signal transduction pathway, b) the phospholipid metabolism/protein kinase C signal transduction pathway, and c) the cAMP protein kinase A signal transduction pathway.
- tyrosine kinase signal transduction pathway a) the tyrosine kinase signal transduction pathway, b) the phospholipid metabolism/protein kinase C signal transduction pathway, and c) the cAMP protein kinase A signal transduction pathway.
- Protein kinases have been found to be particularly important regulators of these pathways.
- tyrosine kinases are known to play a critical role in the regulation of cell division. High levels of tyrosine kinase activity have been measured in highly proliferative cells, such as neoplastic cells. Inhibition of such phosphorylation activity can be correlated with a reduction in cell division, in some cases.
- the current invention is directed to fatty acyl- peptide compositions having enhanced biological activity, compared to the peptide alone.
- Peptides used to form compositions directed to the inhibition of cell proliferation include peptide hormones, peptide hormone analogues, and protein kinase peptide substrates or peptide inhibitors.
- linking such peptides to the polyunsaturated fatty acids lowers the concentration at least several-fold of such peptides required to inhibit cell proliferation.
- Such low chemotherapeutic drug concentrations confer the advantage of reducing toxicity to healthy cells.
- One general object of the invention is to provide a fatty acyl-peptide composition which is useful in inhibiting cell proliferation, such as neoplastic cell proliferation.
- the composition includes a peptide having antiproliferative activity and conjugated to the peptide, a polyunsaturated fatty acyl moiety.
- the composition is characterized by a cell proliferative inhibitory activity which is several-fold greater than that of the antiproliferative peptide alone.
- the fatty acyl moiety of the composition is a doco ⁇ ahexae ⁇ oyl or an eicosapentaenoyl moiety.
- the fatty acyl moiety is a ci ⁇ -4, 7, 10, 13, 16, 19-docosahexa enoyl (DHA) or cis- 5, 8, 11, 14, 17-eicosapentae ⁇ oyl (EPA) moiety.
- DHA 7, 10, 13, 16, 19-docosahexa enoyl
- EPA cis- 5, 8, 11, 14, 17-eicosapentae ⁇ oyl
- the fatty acyl moiety is preferably conjugated to the peptide through an amide linkage.
- the peptide portion of the composition is a peptide hormone, and in a preferred embodiment, the peptide hormone is a somatostatin analog or a gonadotropin releasing hormone (GnRH) analog.
- the peptide hormone has the sequence of SEQ ID NO: 4, and in another preferred' embodiment, the peptide hormone of the composition has the peptide sequence of SEQ ID NO: 5.
- the peptide used in forming the fatty acyl-peptide antiproliferative composition can also be a protein kinase modulatory peptide.
- the protein kinase modulatory peptide has a sequence selected from the group consisting of SEQ ID NO: 1, SEQ ID NO: 2, SEQ ID NO: 3, and SEQ ID NO: 6.
- Another general object of the invention is to provide a method for producing enhanced biological activity of a peptide.
- the invention is used for producing a several-fold enhancement of cell proliferative inhibitory activity in a peptide composition. According to the invention, this enhancement is achieved by conjugating the peptide to a polyunsaturated fatty acid. Preferred peptides and fatty acyl moieties in the method are described above.
- the invention provides fatty acyl-peptide compositions having enhanced biological activity, compared to the activity of the peptide alone.
- Such enhanced biological activity includes such activities as enhanced hormone activity, anti-tumor activity, enhanced immunogenic activity and other peptide-specific activities.
- Figure 1 shows exemplary peptide sequences, designated sequences 1-31 and identified by SEQ ID NO: 1-31, respectively, used in forming the novel peptide-fatty acyl compositions of the invention: STKS (sequence 1, SEQ. ID NO: 1), STKSI (sequence 2, SEQ ID NO: 2), SPKCS (sequence 3, SEQ. ID NO: 3) , ⁇ omatostatin analogue (sequence 4, SEQ. ID NO: 4 ), where lower case “d” signifies the presence of a D-amino acid residue and ' "NH2" at the C-terminal signifies the amidation of the C-terminal end, GnRH (sequence 5, DHA-SEQ. ID NO: 5), where "Glp” signifies the presence of pyroglutamate, EGFA (sequence 6, SEQ ID NO: 6); an N-terminal sequence of human PTH (sequence 7, SEQ ID NO:
- polypeptide fragment derived from fibronectin (sequence 8, SEQ ID NO: 8), and a T cell epitope modulatory peptide (sequence 9, SEQ ID NO: 9) suitable for use in forming compositions of the present invention
- sequences of protein kinase modulatory peptides such as tyrosine kinase inhibitory peptides (sequence 10, SEQ ID NO: 10), (sequence 11, SEQ ID NO: 11), (sequence 12, SEQ ID NO: 12) where "ACM" signifies that the cysteine ⁇ ulfhydryl group has been modified by an acetamidomethyl group, (sequence 13, SEQ ID NO: 13) and (sequence 14, SEQ
- calmodulin-dependent protein kinase III inhibitory peptide sequence 15, SEQ ID NO: 15
- dsDNA-dependent kinase inhibitory peptide sequence 16, SEQ ID NO: 16
- protein kinase C modulatory peptide sequence 17, SEQ ID NO: 17
- other protein kinase modulatory pep ⁇ tides such as for cAMP dependent kinase (sequence 18, SEQ ID NO:18) , (sequence 19, SEQ ID NO: 19) , and (sequence 20, SEQ ID NO: 20), phosphoryla ⁇ e kinase (sequence 21, SEQ ID NO: 21), calmodulin-dependent kinase I and II (sequence 22,
- SEQ ID NO: 22 and sequence 23 SEQ ID NO: 23
- dsRNA-dependent kinase sequence 24, SEQ ID NO: 24
- proline-dependent kinase sequence 25, SEQ ID NO: 25 and sequence
- SEQ ID NO: 27 casein kina ⁇ e I and II (sequence 28, SEQ ID NO:28 and ⁇ equence 29, SEQ ID NO: 29, AMP-activated protein kina ⁇ e ( ⁇ equence 30, SEQ ID NO:30), and S6 kinase
- Figure 2 illustrates coupling of ci ⁇ -4, 7, 10, 13 , 16, 19- doco ⁇ ahexaenoic acid (DHA) to a peptide ( ⁇ equence 1, SEQ ID NO: 1) through an N-terminal amine group to form the fatty acyl-peptide compo ⁇ ition (compo ⁇ ition I, DHA-SEQ ID NO: 1) ; and
- Figure 3 shows sequences of exemplary fatty acyl-pep ⁇ tide composition ⁇ of the invention: DHA-STKS (compo ⁇ ition I, DHA-SEQ. ID NO. : 1) , DHA-STKSI (composition II, DHA-SEQ ID NO: 2), DHA-SPKCS (composition III, DHA-SEQ. ID NO. : 3) ,
- DHA-somatostatin analogue composition IV, DHA-SEQ. ID NO. : 4
- D-Lys6 DHA
- GnRH composition V
- DHA-EGFA composition VI, DHA-SEQ ID NO: 6
- polyunsaturated fatty acid refers to a com ⁇ pound having a carboxylic acid moiety and a long unbranched carbon chain, u ⁇ ually containing between about 8 and 24 carbon atoms, and containing two or more carbon-carbon double bonds.
- a fatty acid i ⁇ conjugated to a peptide through an amide linkage a fatty acyl-peptide derivative is formed.
- the disclosed invention includes fatty acyl- peptide derivatives, and, more generally, peptides linked to long unbranched, polyunsaturated carbon chains.
- peptide hormone refers to a peptide that elicit ⁇ a biological response in a target cell. Peptide hormones are generally low molecular weight proteins ( ⁇
- Such hormones can be isolated from biological sources, chemically synthesized or produced by recombinant methods. Generally, in their natural setting, peptide hor ⁇ mones are ⁇ ecreted from specific cells and produce biological effects in other cells. Analogues of such naturally occurring hormones are encompas ⁇ ed by the term "peptide hormone" and are produced recombinantly or synthetically.
- Peptide hormones as described herein, are divided into two main categories, according to their known acti ⁇ vities in vivo .
- J hormones act either directly or indirectly to produce the desired biological effect.
- the desired effect is inhibition of cell proliferation.
- Peptide hormones that act directly on a cell to inhibit division of the cell are referred to herein as direct peptide hormones; those pep ⁇ tides that act to, stimulate or inhibit synthesis or secretion of endogenous peptide growth regulatory hormones are referred to as indirect peptide hormones.
- an indirect peptide hormone effect a change (increa ⁇ e or decrease) in the extracellular levels of a naturally occurring direct pep ⁇ tide hormone.
- Two exemplary types of indirect peptide hor ⁇ mones are peptide hormone-releasing hormones and peptide hormone release-inhibiting hormones.
- Naturally occurring indirect peptide hormones are generally short polypeptides, usually under 20 amino acids in length.
- Peptide hormone analogs are synthetically or recombinantly prepared peptides which are structurally similar to naturally occurring peptide hormones. For the purpose ⁇ of the present invention, such peptide analogs are included by the term "peptide hormone.” Generally, to be useful in the present invention, such analogs have essentially a similar or a higher biological activity than that of the endogenous peptide hormone. Protein kinase modulatory peptides are peptides which act as inhibitor ⁇ of protein kinase activity. Such pep ⁇ tides may act, for example, as protein kina ⁇ e substrates, residue containing a phosphorylatable amino acid/in the ⁇ equence.
- such kinase modulatory peptide ⁇ may bind to the kina ⁇ e catalytic site, to effect inhibition of kina ⁇ e activity.
- Such kina ⁇ e modulatory peptides act to produce reduced pho ⁇ phorylation of endogenous protein kinase substrate ⁇ .
- Peptide ⁇ and compo ⁇ itions that inhibit cell proliferation are ⁇ ometime ⁇ referred to herein as. "anti ⁇ proliferative" peptides or compositions.
- the present invention is directed to peptide compositions having anti-proliferative activity. More generally, it is the discovery of the invention that these fatty acyl-peptide composition ⁇ have enhanced biological activity in comparison to underivatizea peptides.
- Anti ⁇ proliferative fatty acyl-peptide compositions of the invention are effective to inhibit proliferation of highly proliferative cells, such as neoplastic cells or virally- infected cells.
- peptides known to have anti-proliferative activity are linked to a fatty acyl moiety, ⁇ uch a ⁇ a doco ⁇ ahexae ⁇ oyl (DHA) or eico ⁇ apentaenoyl (EPA) group, a ⁇ described below.
- Peptide hormones having cell proliferative inhibitory activity are known in the art, and may be direct or indirect hormones.
- Such anti ⁇ proliferative peptide hormone ⁇ are u ⁇ ually, but not necessarily, specific to a particular type of cell, such as a cell with ⁇ pecific hormonal requirement ⁇ .
- indirect peptide hormones include those peptides which act to effect a decrease in the level of a direct cell proliferation ⁇ timulatory hormone or to effect an increase in the level of a direct cell proliferation inhibitory hormone.
- GnRH gonadotropin relea ⁇ ing hormone
- LH luteinizing hormone
- FIG. 15 inhibit the growth of hormone dependent carcinomas, such as androgen-dependent prostate tumors (Schally) .
- GnRH is also effective against such tumors a ⁇ colon and pancreatic tumors.
- Figure 1 shows a sequence of a GnRH analog useful in treating such tumor ⁇ (5, SEQ ID NO: 5) .
- Figure 3 ⁇ hows
- GnRH analogues can be ⁇ creened for potential anti ⁇ proliferative activity, by assessing their abilities to
- Indirect peptide hormones also include peptide hormone release-inhibiting hormone ⁇ .
- Somatostatin i ⁇ a 14 amino acid peptide that inhibit ⁇ the relea ⁇ e of growth hormone (GH) .
- GH enhances the proliferation of its cellular ⁇ 35 targets.
- Native somatostatin peptide and analogs are used to form fatty acyl peptide derivative composition ⁇ of the invention, as described below.
- the somatostatin fatty-acyl peptide is about 150 times more potent than the parent molecule.
- fatty acyl-peptide compositions of the invention can be used in forming fatty acyl-peptide compositions of the invention.
- peptides are selected based on their known anti-proliferative activity in a cell proliferation as ⁇ ay, described in Section III below. Selected peptides are then conjugated to fatty acyl moieties, and are tested for enhanced potency in such a ⁇ ays .
- a fatty-acyl peptide derivative composition i ⁇ a useful anti-tumor or anti-neoplastic cell agent, when it is found to have at least a several-fold increase in potency, in comparison to the unconjugated peptide.
- Direct Peotide Hormone ⁇ Also used in forming compositions of the invention are peptides which are known to have direct antiproliferative effects on cells.
- An exemplary direct peptide hormone is parathyroid hormone (PTH) .
- PTH parathyroid hormone
- This peptide i ⁇ 84 amino acids in length, and can directly inhibit osteobla ⁇ t division.
- Certain bone cancers are characterized by hyperproliferating o ⁇ teoblast ⁇ . Structure-function studies indicate that an N-terminal fragment of PTH is active in inhibiting osteobla ⁇ t proliferation (Kano) .
- Thi ⁇ ⁇ equence 7 SEQ ID NO: 7 7) i ⁇ ⁇ hown in Figure 1.
- a peptide which i ⁇ defined as a direct peptide hormone for purpo ⁇ es of thi ⁇ invention is a peptide fragment of fibronectin having the ⁇ equence SEQ ID NO: 8, ⁇ hown in Figure 1 as sequence 8. This fragment spans the recognition site within the fibronectin molecule to which cells bind for attachment to fibronectin.
- This fibronectin recognition site peptide and analogues thereof are u ⁇ ed to inhibit fibronectin-mediated cell attachment and ⁇ preading in vitro. They may also be important for regulation of tumor cell proliferation and metasta ⁇ i ⁇ in vivo (Kumagai) .
- fibronectin recognition site peptides are coupled to fatty acyl moieties to form compositions useful in inhibiting tumor cell proliferation and meta ⁇ tasis.
- Direct peptide hormone ⁇ a ⁇ defined in the current invention, al ⁇ o encompa ⁇ s peptides derived from immunogenic polypeptides for modulating a response from ⁇ pecific immune cell populations.
- a T-cell epitope peptide (TCEP) i ⁇ coupled to a fatty acyl moiety as described herein to form a composition useful in modulating the T- cell immune response.
- SEQ ID NO:9 The sequence of TCEP (SEQ ID NO:9) is shown in Figure 1 as ⁇ equence 9, and ⁇ ynthe ⁇ i ⁇ of a DHA derivative of TCEP is detailed in Example 7.
- Elevated protein kinase C and tyrosine kinase activities are associated with neoplastic cell prolif ⁇ eration or transformation (Wein ⁇ tein, Yarden) . Inhibition of such kinase activities can be effected by the presence of small peptide fragments which mimic protein ⁇ ub ⁇ trate pho ⁇ phorylation sites and/or protein kinase modulatory domains. Such protein kinase modulatory peptides are effective to compete with endogenous protein kina ⁇ e sub ⁇ trate ⁇ . Selection of ⁇ pecific protein kinase modulatory peptides for use in forming the fatty acyl-pep- tide derivatives of the invention i ⁇ de ⁇ cribed below.
- Tyrosine Kinase Substrates and Inhibitors Receptors for a number of growth factors, including epidermal growth factor (EGF) , insulin growth factor (IGF) , and platelet derived growth factor (PDGF) , contain tyrosine kinase catalytic domains which phosphorylate specific intracellular protein substrates, including the receptor itself, in some cases, at tyrosine residues.
- EGF epidermal growth factor
- IGF insulin growth factor
- PDGF platelet derived growth factor
- Tyrosine kina ⁇ es may also play a role in the uncontrolled growth of keratinocytes which can result in psoriasi ⁇ or other highly proliferative skin di ⁇ order ⁇ . Keratinocytes pos ⁇ es ⁇ tyrosine kinase-like growth factor receptors.
- Figure 1 shows ⁇ equence ⁇ of ⁇ everal exemplary tyrosine kinase modulatory peptide ⁇ which can be u ⁇ ed to form anti-proliferative compo ⁇ ition ⁇ of the invention, ⁇ uch a ⁇ STKS (SEQ ID NO: 1) and STKSI (SEQ ID NO: 2), ⁇ equence ⁇ 1 and 2, re ⁇ pectively.
- tyrosine kinase modulatory peptides can be identified, based on peptide ⁇ equence ⁇ surrounding phosphorylation sites of endogenous protein kina ⁇ e ⁇ ub ⁇ trates in highly proliferating cells.
- cellular sources used for identification of ⁇ uch substrate peptides are proliferating cells of the type to be targeted; however, it is appreciated that tyrosine kinase modulatory peptide ⁇ derived from one cell ⁇ ource may inhibit a tyrosine kinase derived from a different cell type.
- proliferating cells such as a ⁇ a lymphoma cell line having a high level of tyrosine kinase activity, are incubated with [32p] -pho ⁇ phate, or a particulate fraction of the cells i ⁇ incubated with radiolabelled [gamma 32p] ATP.
- Cellular polypeptide ⁇ are ⁇ eparated by denaturing gel electrophore ⁇ i ⁇ , and the pho ⁇ phorylated protein ⁇ are observed by autoradiography. Phosphoprotein- containing bands are excised from the gel, and the phosphoprotein i ⁇ eluted from the gel band.
- the eluted protein i ⁇ ⁇ ubjected to partial hydroly ⁇ is, and the identity of the phosphorylated amino acid determined, according to methods known in the art (Casneillie) .
- phosphoprotein ⁇ are ⁇ ubjected to peptide fragmentation, such a ⁇ by proteoly ⁇ i ⁇ or chemical means, and phosphotyrosine-containing peptides are sequenced, according to conventional methods (Casneil- lie) .
- Short peptides (approximately 6-20 amino acids) corresponding to the peptide sequence ⁇ in the vicinity of the pho ⁇ phorylation ⁇ ite can be ⁇ ynthesized and te ⁇ ted for inhibition of tyro ⁇ ine kinase activity.
- Tyrosine kinase modulatory peptides can be prepared by substituting for the tyrosine phosphorylation ⁇ ite residue an amino acid residue that cannot be phosphorylated, ⁇ uch as a phenylalanine residue, composition II in Figure 3 (DKA-STKSI) ⁇ how ⁇ an inhibitory fatty acyl-peptide compo ⁇ ition, the peptide portion of which ha ⁇ almo ⁇ t complete identity with the peptide portion of composition I
- composition II a phenylalanine residue has been substituted for a tyrosine residue present in composition I.
- Both peptides inhibit proliferation of neopla ⁇ tic cell ⁇ . Accordingly either peptide can be conjugated to a fatty acyl group, to form an anti-tumor fatty acyl-peptide compo ⁇ ition of the invention, a ⁇ described in Section III, below.
- PKC Protein Kinase C
- PKC Peotide Substrates.
- PKC is a component of the phospholipid metabolism/protein kinase C signal tran ⁇ duction pathway which play ⁇ a critical role in normal cellular growth control. Activation of PKC is mediated by a family of G-protein-modulated receptors. When activated, the cyto ⁇ olic form of PKC binds to the cytoplasmic face of the plasma membrane.
- One of the known protein targets of PKC i ⁇ the EGF receptor.
- Pho ⁇ phoryla- tion of EGF receptor by protein kina ⁇ e C results in a decrease in the affinity of the receptor for EGF and a decrease in EGF receptor-associated tyrosine kinase activity (Berridge) .
- Thi ⁇ illustrates the heterologous interaction ⁇ between signal transduction pathways, specifically between the tyrosine kinase pathway and the phospholipid metabolism/protein kina ⁇ e C pathway.
- Figure 1 lists the ⁇ equence ⁇ of some of the known peptide substrate inhibitors for PKC, such as sequence 3 (SEQ ID NO: 3) and sequence 17 (SEQ ID NO: 17). These peptides are suitable PKC modulatory peptides for use in forming fatty acyl-pep- tide compositions of the present " invention. PKC peptide inhibitors can also be identified from endogenous protein sub ⁇ trates of protein kinase C, using the same general procedures described in part 1, above, for tyrosine kinases. 3. Other Protein Kinases.
- Modulatory peptides targeting protein kinases other than tyrosine kinase and protein kina ⁇ e C can also be used to form fatty acyl-pep ⁇ tide anti-tumor compo ⁇ ition ⁇ of the invention.
- High proliferation rates are related to the hyperpho ⁇ phorylation of an oncogenic product from a human papillomavirus which contains a casein kinase II phosphorylation consen ⁇ us ⁇ equence (Hashida) .
- PKA cyclic AMP-dependent protein kinase
- cyclic AMP turns on the gene that encodes the peptide hor ⁇ mone release-inhibiting hormone, somatostatin.
- target protein kinases in conjunction with the present invention, are components of signalling cascades involved in cell proliferation. For example, when
- epidermal growth factor binds to the epidermal growth factor receptor (EGFR), a tyrosine kinase, it stimulate ⁇ activation of myelin basic protein (MBP) kinase, through phosphorylation of MBP kinase. MBP kinase, in turn, activates S6 kinase by a phosphorylation event.
- EGFR epidermal growth factor receptor
- MBP myelin basic protein
- EGFR-bearing cells ⁇ uch as adipocyte cells (Ahn) .
- fatty acyl-peptide compositions of the invention using a ⁇ peptide co ponent ⁇ , pho ⁇ phorylation recognition ⁇ equence peptide ⁇ for one or , 35 more of EGFR kina ⁇ e, MBP kinase and S6 kinase will serve to inhibit the signalling cascade of which these kinases are a part. Sequences of exemplary peptide inhibitors of these kinases are shown in Figure 1 (e.g. , sequence 11, SEQ ID NO: 11, to sequence 31, SEQ ID NO:31) .
- protein kinase activities involved in cell proliferation whose activities can be modulated include calmodulin-dependent kinase ⁇ I and II, cGMP-dependent protein kinase, ds-DNA-dependent protein kinase, proline-dependent kina ⁇ e, and AMP-activated kina ⁇ e.
- Sequences of exemplary peptide modulatory peptide ⁇ directed to the ⁇ e kina ⁇ es are listed in Figure 1 (SEQ ID NOs: 18- 31) .
- the peptide ⁇ were conjugated to fatty acyl moietie ⁇ , a ⁇ described below and illu ⁇ trated in Figure 2 and te ⁇ ted for thei ⁇ activities in cell proliferation assays, as described in Section III, below.
- Peptides shorter than about 30 amino acids in length are conveniently prepared by methods commonly used in solid-phase peptide synthesis, as detailed below (Stewart) .
- N-alpha-protected amino acid anhydrides are prepared in crystallized form and used for successive amino acid addition to the peptide N-terminus.
- the growing peptide on a solid support is acid treated to remove the protective group, and washed several times to remove residual acid.
- the peptide i ⁇ then reacted with another N-protected amino acid.
- the amino acid addition reaction may be repeated two or three time ⁇ to increase the yield of growing peptide chains.
- the protected pep ⁇ tide re ⁇ in i ⁇ treated with liquid hydrofluoric acid to deblock and release the peptide ⁇ from the ⁇ upport.
- Preferred fatty acyl moietie ⁇ for use in the present invention are those with a high degree of unsaturation, and include such fatty acids as cis-5, 8, 11, 1 , 17- eico ⁇ apentae ⁇ oic (EPA) and ci ⁇ - ,7, 10, 13, 16, 19- doco ⁇ ahexaenoic acid (DHA) .
- EPA eico ⁇ apentae ⁇ oic
- DHA doco ⁇ ahexaenoic acid
- uch polyunsaturated fatty acids can be prepared synthetically according to standard methods, isolated from the oils of marine fish, or obtained from commercial ⁇ ource ⁇ .
- fatty acid ⁇ are linked to the peptide ⁇ via the terminal amine group or via internal amine groups, such as the amine group of ly ⁇ ine, in an amide linkage.
- Figure 2 illustrates a scheme for coupling of ⁇ equence 1 to DHA by the terminal amino group of the peptide through an amide linkage to form the fatty acyl-peptide compo ⁇ ition I in
- DHA is activated by reaction with N- hydroxy ⁇ uccinimide prior to reaction with an a ino-group containing polypeptide.
- DHA i ⁇ coupled by the free amine group of a lysine residue in the ⁇ equence to form the fatty acyl composition, ⁇ uch a ⁇ compo ⁇ ition V in Fig. 3.
- Example ⁇ 2-6 detail preparation of fatty acyl-peptide compositions in which acylation of peptides i ⁇ carried out u ⁇ ing activated e ⁇ ter or acyl chloride derivative ⁇ of DHA or EPA.
- Activated esters useful in preparing composition ⁇ of the current invention include DHA-O-benztriazole ester (DHA-OBT) , DHA-O-pentafluorophenyl ester (DHA-Opfp) , and DHA-O- ⁇ uccin midyl ester (DHA-O-N-Succ) .
- the acylation is carried out as detailed in Example 2. Crude products, obtained after acylation, are purified by HPLC. The purity of the final product ⁇ l ⁇ characterized by analytical HPLC and TLC data, while the chemical characterization l ⁇ accompli ⁇ hed by mass ⁇ pectrometry (MS) .
- MS mass ⁇ pectrometry
- peptides can be selected for use in anti-proliferative fatty acyl-peptide composition ⁇ of the invention, ba ⁇ ed on their abilities to inhibit cell proliferation or to inhibit component ⁇ , such as protein kinase component ⁇ , of cell proliferative ⁇ timulu ⁇ pathway ⁇ .
- component ⁇ such as protein kinase component ⁇
- conjugating to ⁇ uch a peptide, a polyun ⁇ aturated long chain carbon, ⁇ uch as a polyunsaturated fatty acyl moiety described above enhances the biological activity of the peptide.
- conjugation of a polyunsaturated fatty acyl moiety to an anti-proliferative peptide enhances its ability to inhibit neoplastic cell proliferation.
- This aspect of the invention will be better appreciated from the discussion below.
- tyrosine kinase modulatory peptide ⁇ selected a ⁇ de ⁇ cribed above are covalently linked to polyunsaturated fatty acids to form compositions which are effective to inhibit proliferation of neoplastic cells.
- DHA-STKS DHA-SEQ. ID
- Fatty acid-tyro ⁇ ine kinase inhibitory peptide compo ⁇ sitions can also be prepared by substituting for the tyro ⁇ sine residue an amino acid re ⁇ idue that cannot be pho ⁇ - phorylated.
- DHA-STKSI DHA-SEQ ID NO: 1
- composition II the tyrosine residue of tyrosine kinase sub ⁇ trate DHA-STKS (DHA-SEQ ID NO: 1; compo ⁇ ition I) ha ⁇ been ⁇ ub ⁇ tituted by a phenylalanine residue.
- DHA-STKS DHA-SEQ ID NO: 1; compo ⁇ ition I
- Both fatty acid-protein kinase peptide sub ⁇ trate and peptide inhibitor compositions inhibit the proliferation of neoplastic cells, as detailed in Example 8 and shown in
- DHA-EGFA DHA-SEQ ID NO: 6, composition VI
- a PKC peptide sub ⁇ trate-fatty acid compo ⁇ ition u ⁇ ed in the experiment ⁇ de ⁇ cribed in Example 8 include ⁇ a peptide fragment from myelin ba ⁇ ic protein, and has the structure shown in Figure 3 as compo ⁇ ition III (DHA-SEQ. ID NO: 3) . Additionally, endogenous substrates of PKC may be identified as described above for the tyrosine kinase substrates. The polypeptide fragments that contain PKC phosphorylation sites are then sequenced and u ⁇ ed to form the fatty acid peptide compositions of the invention.
- fatty acyl- peptide derivatives are prepared as described in Section II, above, and inhibition of neoplastic cell proliferation by such compositions is measured, according to one or more standard cell proliferation assays.
- inhibition of cell proliferation is mea ⁇ ured directly, by measuring the number of surviving cells after exposure of the cells to a test composition, ⁇ uch a ⁇ a fatty acyl-peptide compo ⁇ ition di ⁇ clo ⁇ ed herein.
- an activity which correlate ⁇ to cell proliferation can be mea ⁇ ured to determine indirectly the effect of a te ⁇ t compound on cell proliferation.
- activitie ⁇ ⁇ uch as tyrosine kina ⁇ e activity or relea ⁇ e of a growth-promoting hormone, which are known to correlate with neopla ⁇ tic cell proliferation, are u ⁇ ed a ⁇ indicators of anti-proliferative activity.
- polyunsaturated fatty acyl-peptide anti-proliferative compositions may exhibit one or more of the following in vi tro activities: (a) inhibition of cell proliferation, (b) inhibition of tyrosine kinase activity, (c) stimulation of release of a growth-inhibiting hormone, such as lu- teinizing hormone, and (d) inhibition of release of growth- ⁇ timulating hormone ⁇ , ⁇ uch a ⁇ inhibition of growth-hormone relea ⁇ e.
- vi tro assay ⁇ which can be correlated with uncontrolled cell growth may also serve as assay ⁇ for ⁇ electing and determining the activities of fatty acyl-peptide compo ⁇ ition ⁇ of the invention. More ⁇ pecifically, ⁇ uch assay ⁇ can ⁇ erve to determine the relative potencie ⁇ of compositions of the invention as anti-proliferative agents.
- Example 8 An as ⁇ ay for mea ⁇ uring the inhibitory effect ⁇ of polyun ⁇ aturated fatty acyl-peptide derivative ⁇ on cell proliferation is described in detail in Example 8.
- a number of tumor cell lines including a human prostatic adenocarcinoma cell line, a human breast adenocarcinoma cell line and a human colon adenocarcinoma cell line, were used to test compound ⁇ of the invention for effect ⁇ on cell proliferation.
- DHA-STKS tyrosine kinase synthetic peptide ⁇ ub ⁇ trate fatty acid derivative DHA-STKS
- DHA-SEQ ID NO: 3 protein kina ⁇ e C peptide ⁇ ubstrate fatty acid derivative DHA-STKCS
- DHA-SEQ ID NO: 5 gonadotropin relea ⁇ e hormone fatty acid derivative DHA-D- Ly ⁇ 6-GnRH
- Solution ⁇ of the peptide derivative ⁇ were added in culture media to test cells for an incubation period of 2.5 hours. At the end of the test period, cells were centrifuged, the pellet diluted with 1% BSA in saline and viable tumor cell counts were determined by the trypan blue exclusion method. Results of tests using peptide fatty acid derivatives DHA-STKS (DHA-SEQ. ID NO: 1) and DHA-D- lys6-GnRH (DHA-SEQ. ID NO: 5) in various transformed cell lines are ⁇ hown in Table 1.
- PC 3 human prostatic adenocarcinoma cell line
- MCF 7 human brea ⁇ t adenocarcinoma cell line
- HT 29 human colon adenocarcinoma cell line.
- Additional method ⁇ are available for monitoring the viability of cells, including as ⁇ ays based on the differential dye uptake by viable cells in comparison to that taken up by non-viable cells.
- viable cells take up diacetyl fluore ⁇ cein and hydrolyze it to fluore ⁇ cein, to which the cell membrane of live cells is impermeable. Live cells fluoresce green.
- Nonviable cells may be counter-stained with ethidium bromide and will fluoresce red.
- LH Luteinizing Hormone
- fatty acyl derivatives of ⁇ uch compound ⁇ are effective to inhibit neoplastic cell proliferation at concentrations which are several-fold lower than the concentration of peptide alone required to inhibit such cell proliferation.
- an analog of gonado- tropin releasing hormone analog conjugated to the poly ⁇ unsaturated fatty acid, DHA, (D-Lys 6 (DHA) -GnRH; DHA-SEQ ID NO: 5) was tested in an a ⁇ ay of LH release by anterior pituitary cell suspensions as detailed in Example 9.
- luteinizing hormone level ⁇ were quantitated by a double antibody radioimmunoa ⁇ ay procedure.
- Peptide hormone-release inhibiting hormones decrease the extracellular levels of peptide hormones which interact with ⁇ pecific cellular receptor ⁇ . Generally, binding of peptide hormone to the receptor trigger ⁇ a ca ⁇ cade of biochemical events mediated through second me ⁇ enger ⁇ . In some cases, an end-result of such hormonal activity is cellular proliferation. Peptide hormone-release inhibiting hormone ⁇ which act to inhibit such hormone ⁇ are useful in forming fatty acid-peptide compo ⁇ ition ⁇ described by the pre ⁇ ent invention. Analogue ⁇ of ⁇ uch peptide hormone- release inhibiting hormones are also u ⁇ eful in forming ⁇ uch compo ⁇ ition ⁇ .
- Somato ⁇ tatin i ⁇ an exemplary peptide hormone-release inhibiting hormone analogue which inhibits hormonally activated cell proliferation.
- Somatostatin i ⁇ a growth hormone relea ⁇ e inhibitory hormone which inhibit ⁇ relea ⁇ e of growth hormone.
- Growth hormone (GH) binds to specific cell-surface receptors distributed widely throughout the body. Binding of GH agonists to GH receptor ⁇ re ⁇ ult ⁇ in increa ⁇ ed cellular division, and hence, cell proliferation.
- a ⁇ omato ⁇ tatin-fatty acyl analogue (SEQ ID NO: 4) was ⁇ ynthesized as detailed in Example 5. Inhibition of growth hormone relea ⁇ e by the DHA-acylated somatostatin analogue was measured, as de ⁇ cribed in Example 10. GH levels were determined by a double-antibody radio- immunoassay for the hormone. In thi ⁇ a ⁇ ay, the DHA- somato ⁇ tatin analogue wa ⁇ 1000 time ⁇ more effective in stimulating release of growth hormone than was either unacetylated somatostatin analogue or somato ⁇ tatin.
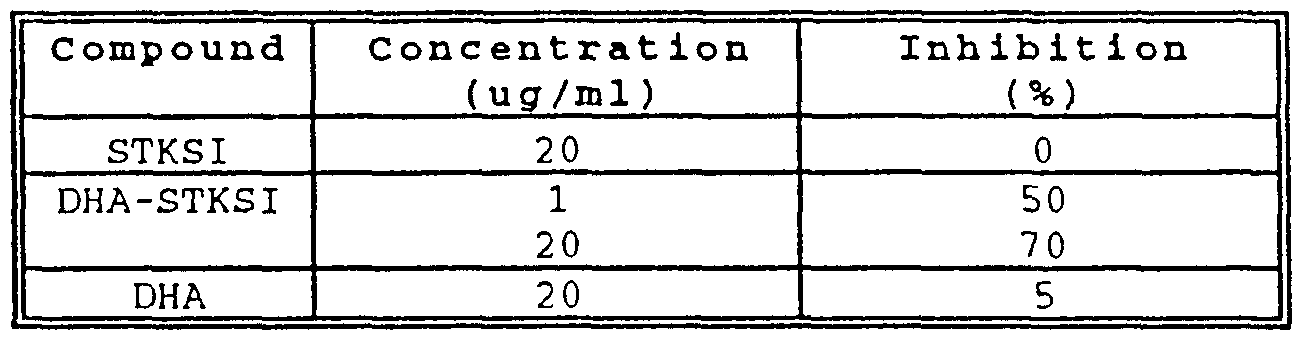
- tyrosine kinase activity can be measured in cell ⁇ from the human brea ⁇ t adenocarcinoma line MDA-MB-231. Briefly, the ⁇ e cell ⁇ are incubated with a te ⁇ t compound, ⁇ uch as the polyunsaturated fatty acid-tyro ⁇ ine kinase peptide inhibitor DHA-STKSI (DHA-SEQ ID NO: 2), then harvested and homogenized.
- DHA-STKSI DHA-SEQ ID NO: 2
- Tyrosine kinase activity is measured by incorporation of radiolabeled pho ⁇ phate into an endogenous protein substrate in the cell ⁇ .
- the endogenou ⁇ protein substrate is the EGF receptor, and the pho ⁇ phorylation event i ⁇ an autopho ⁇ phorylation event.
- STKSI the underivatized peptide inhibitor
- DHA-STKSI DHA-SEQ ID NO:2
- DHA pre ⁇ ent by it ⁇ elf at 20 microgram ⁇ /ml decrea ⁇ ed the extent of pho ⁇ phorylation by only 5%. It can be appreciated, in accordance with the pre ⁇ ent invention, that similar cellular as well as in vi tro tyrosine kina ⁇ e assays can be used to monitor activity and to test compounds for their abilities to inhibit ⁇ uch activity.
- Fatty acyl-peptide compositions of the invention are useful in inhibiting uncontrolled proliferation of cell ⁇ , such a ⁇ benign and malignant tumor cell ⁇ , virally-infected cell ⁇ , p ⁇ oriatic cells, and the like.
- cell ⁇ such as a ⁇ benign and malignant tumor cell ⁇ , virally-infected cell ⁇ , p ⁇ oriatic cells, and the like.
- the use of the composition of the invention for inhibition of such cell proliferation is illustrated by experiments summarized in Table 1.
- peptides are selected for use in forming compo ⁇ ition ⁇ of the invention, ba ⁇ ed on their known or experimentally determined activities in inhibiting cell proliferation. Selected peptides are then used to form polyunsaturated fatty acyl-peptide composition ⁇ , according to the general method ⁇ described in Section II, above and detailed in Example ⁇ 1-7.
- polyunsaturated fatty acyl-peptide compositions of the pre ⁇ ent invention are effective to produce a ⁇ everal-fold enhancement of anti-proliferative activity, in compari ⁇ on to peptides alone.
- fatty acyl-GnRH and -GnRH analogues will find use in treating androgen-dependent prostate adenocarcinoma ⁇ .
- Te ⁇ t GnRH compound ⁇ can be screened in an LH relea ⁇ e as ⁇ ay, a ⁇ described in Example 9, then tested in an experimental animal model, such as a rat bearing the Dunning R-3327-H prostate adenocarcinoma.
- GnRH derivative ⁇ of the invention are expected to find usefulne ⁇ in treatment of e ⁇ trogen-dependent mammary tumors, and their efficacy can.be mea ⁇ ured in rats ( ⁇ uch a ⁇ Wi ⁇ tar Furth rat ⁇ ) carrying a mammary tumor ⁇ uch a ⁇ the MT/W9A mammary tumor, according to methods known in the art.
- LHRH analog composition ⁇ will inhibit growth of certain pituitary tumor ⁇ , chondro ⁇ arcoma ⁇ , and o ⁇ teo ⁇ arcoma ⁇ (Schally) .
- An anti-neopla ⁇ tic or anti-tumor treatment method, a ⁇ described herein, include ⁇ expo ⁇ ing target neopla ⁇ tic cell ⁇ to a concentration of fatty acyl-peptide compound effective to inhibit neopla ⁇ tic cell proliferation at lea ⁇ t about 50%, and preferably about 90%.
- effective concentration ⁇ can be determined in an in vi tro a ⁇ ay, a ⁇ de ⁇ cribed in Section IV, above.
- the method of the invention ha ⁇ general utility in enhancing biological activity of a biologically active peptide, by attaching to the peptide a polyun ⁇ aturated fatty acyl moiety.
- Example 1 Synthesis of Peotide ⁇ Solid-phase synthesis was carried out according to standard method ⁇ .
- the ⁇ ynthesi ⁇ i ⁇ performed u ⁇ ing a Beckman model 900 peptide ⁇ ynthe ⁇ izer.
- Side chain protecting group ⁇ include for cysteine, 4- Met-Benzyl; for lysine, 2-chlorobenzyloxycarbonyl; for serine, benzyl; for arginine, tosyl; for threonine, benzyl; for a ⁇ partate, benzyl; and for tyrosine, 2-bromobenzyl- oxycarbonyl.
- a mixture of protected peptide re ⁇ in (1.32 g) , 2- mercaptopyridine (0.5 g) , p-cre ⁇ ol (2.6 g) , and liquid hydrogen fluoride (HF) (25 ml) is stirred at 0 degrees with a rapid stream of nitrogen ga ⁇ , first below 0 degree ⁇ , then at 24 degree ⁇ .
- the mixture i ⁇ ⁇ tirred in ethyl acetate (25 ml) until a finely divided ⁇ olid i ⁇ obtained.
- the ⁇ olid i ⁇ filtered, washed with ethyl acetate, and air dried.
- the solid is stirred in 50% aqueous acetic acid (10 ml) to dis ⁇ olve the peptide, filtered and wa ⁇ hed with 20 ml water.
- the mixture was ⁇ tirred overnight under He atmo ⁇ phere for the completion of the transe ⁇ terification reaction.
- This reaction mixture can be used directly for the N-terminal acylation of the required peptide or worked up for the preparation of pure DHA-O-N-Succ .
- the reaction mixture was diluted to 30 ml with water and extracted with 3 x 10 ml of peroxide-free ether (treated and stored over alumina) .
- the combined ether phase was back-extracted with 3 x 5 ml of water.
- the ether solution wa ⁇ dried over anhydrou ⁇ ⁇ odium ⁇ ulphate, evaporated to dryne ⁇ s and taken up in 4 ml of acetone.
- Example 3 Preparation of DHA-STKSI (DHA-SEQ ID NO. 2) A procedure similar to that described in Example 2 was used to generate the activated ester group of DHA (DHA-O-N- ⁇ uccinimide) . Then, 0.03 mM of STKSI (36.2 mg) was acylated with 0.05 mM of DHA-O-N-Succ ester in the presence of 0.15 mM of DIEA according to Example 2. The product wa ⁇ eluted from the same HPLC gradient a ⁇ described in Example 2, the product eluted at 64.5% B. The final yield was 82%. The chemical characterization of the compound wa ⁇ carried out by mass ⁇ pectrometry. MH+theoretical: 1517.4, MH+mea ⁇ ured:1515.5.
- Example 4 Preparation of DHA-SPKCS (DHA-SEQ ID NO: 3) SPKC-(Ly ⁇ /ep ⁇ ilon-TFA/)2,9 (54.8 mg, 0.04 mM, final concentration) wa ⁇ ⁇ u ⁇ pended in 3 ml of water-acetone (1:1) containing 0.08 mM DIEA. 'Crude DHA-O-N- Succ activated ester in DMF solution (0.04mM), a ⁇ de ⁇ cribed in Example 2, wa ⁇ added, and the suspension was stirred overnight under a He atmo ⁇ phere. The reaction mixture was evaporated to dryness and the product was TFA-deprotected in the following fashion.
- the product was eluted at 11.5 minutes, the yield was 65%, the purity of the product was 97%.
- the product was analyzed by mas ⁇ spectrometry, with the following result ⁇ : MH+theoretical:1700.8; MH+measured: 1700.8
- DHA-SEQ ID NO: 4 360 ul each of 0.5 mM ⁇ olution ⁇ of DHA, N,N'-dii ⁇ o- propyl carbodiimide (DIC) , and pentafluorophenol in dimethylformamide (DMF) were mixed and kept at 25. oC.
- Step 1 I ⁇ ocratic elution with 50 ml of 2-propa- nol-acetic acid-water (20:40:40) .
- Step 2 Gradient elution with 400 ml of elution mixture applied in ⁇ tep 1 and 400 ml of 2-propanolacetic acid-water (35:32.5:32.5) .
- the purity of the fraction ⁇ was checked by HPLC, the purest fraction ⁇ were pooled, evaporated, lyophilized and yielded 57% product. The purity of the product wa ⁇ 92%. The composition of the product was analyzed by mas ⁇ ⁇ pectrometry.
- Example 7 Svnthe ⁇ is of DHA-T Cell Epitope Peptide (DHA-SEQ ID NO: 12) T-cell epitope peptide (TCEP) (132 mg, 0.075 mmole) , with TFA and formyl protecting groups, was di ⁇ olved in DMSO (2 ml) containing DIEA (40 ⁇ l, 0.30 mmole) and DHA ⁇ uccinyl ester (0.075 mmole). The reaction mixture was stirred for 3 hours at room temperature at which time acetonitrile/water (1:1, v/v), 0.5 ml) and TFA (0.5 ml) were added.
- DHA-SEQ ID NO: 12 T-cell epitope peptide
- reaction mixture was then filtered through a 0.2 ⁇ m filter and this solution was applied directly to a preparative rever ⁇ ed-pha ⁇ e HPLC column for purification.
- DHA-TCEP was collected and identified by mas ⁇ ⁇ pectro ⁇ copy.
- the purified peptide was treated in 1 M aqueous piperidine for 8 hour ⁇ at room temperature at which time the product wa ⁇ i ⁇ olated by lyophilization.
- Example 8 A ⁇ av of the Inhibition of Cell Proliferation The growth inhibitory effect of peptide hormone analogue ⁇ , ⁇ uch a ⁇ DHA-STKS, DHA-STKCS and D-Ly ⁇ (DHA)-
- GnRH was evaluated on the basis of changes in the total cell number.
- Cells were cultivated in RPMI 1640 medium (GIBCO BRL, Eggenstein, Germany, Cat. No. : 074-01800) ⁇ upplemented with 10% fetal calf ⁇ erum.
- Tumor cell ⁇ were wa ⁇ hed with pho ⁇ phate-buffered ⁇ aline, then 0.25% tryp ⁇ in wa ⁇ added to detach them.
- the cell ⁇ were ⁇ uspended in RPMI 1640 medium and after resu ⁇ pending the cell ⁇ were ⁇ tained with trypan blue and counted in a Burker chamber.
- 0.1 ml cell ⁇ u ⁇ pension and 0.1 ml 0.1% trypan blue were mixed.
- the viability of tumor cells was estimated on the basis of trypan blue (0.4%) exclusion using a hematocytometer.
- Example 9 Luteinizing Hormone (LH) Release A ⁇ ay Anterior pituitarie ⁇ were obtained from adult (male or female) Wi ⁇ tar ⁇ train rats according to the method previously de ⁇ cribed (Shaw) .
- the pituitarie ⁇ were cut into ⁇ mall piece ⁇ , incubated with collagena ⁇ e then di ⁇ per ⁇ ed mechanically to ⁇ ingle cell ⁇ .
- the cell- ⁇ u ⁇ pen ⁇ ion wa ⁇ mixed with Sephadex G-10 bead ⁇ a ⁇ ⁇ upport material and tran ⁇ ferred into a ⁇ uperfusion chamber.
- the cell ⁇ were continuou ⁇ ly perfu ⁇ ed with oxygenated medium or with a medium containing the peptide to be te ⁇ ted, ⁇ uch a ⁇ D- Lys6 (DHA) -GnRH.
- the biological potency of a given analogue was determined based on the pituitary hormone responses (peaks) to the peptide stimulation over the baseline secretion.
- Example 10 As ⁇ ay for Growth Hormone (GH)
- GH Growth Hormone
- Myers ⁇ uperfu ⁇ ion method
- the hypophyses are cut into small piece ⁇ , incubated with collagena ⁇ e, and di ⁇ per ⁇ ed to single cell ⁇ .
- the cells in a ⁇ uperfu ⁇ ion chamber are continuou ⁇ ly perfu ⁇ ed with oxygenated medium or with medium containing the peptide to be te ⁇ ted.
- the level ⁇ of GH are determined by a double-antibody radioimmunoa ⁇ ay for the hormone.
- the following methodology can be u ⁇ ed.
- Antibody is produced by dissolving an appropriate amount of growth hormone (2-3 mg) in 200 microliters 0.01 N sodium hydroxide. The solution is diluted to 1 mg/ml with protein free PBS and mixed 1:1 with Freund' ⁇ adjuvant. 0.5-1 mg growth hormone i ⁇ in ⁇ jected into three ⁇ ubcutaneou ⁇ ⁇ ites of a young guinea pig, and repeated at two- to three-week interval ⁇ .
- the hGH radioimmunoa ⁇ ay i ⁇ performed in the following manner.
- the ⁇ tandard' ⁇ concentrations in hGH assay ⁇ are the following: 5, 2.5, 1.25, 0.625, 0.313, 0.156, and 0.078 ng/ml. 50 ml of the unknown growth hormone concentration i ⁇ added to tube ⁇ not containing the standard.
- the next step is to add the guinea pig anti-hGH serum (fir ⁇ t anti ⁇ body) .
- the radiolabeled hGH i ⁇ added to every tube.
- the a ⁇ ay tube ⁇ are ⁇ haken gently, left at room temperature for two hour ⁇ , and then placed in a refrigerator at 4'C for at lea ⁇ t three day ⁇ .
- the tubes are removed from the refrigerator and the appropriate dilution of the goat anti-guinea pig serum added (second antibody) .
- the second antibody is diluted in 1% BSA PBS buffer to obtain approximately 50% precipitation of antibody-bound labeled hGH. Aliquot ⁇ of 100 microliter ⁇ of the ⁇ econd antibody are added to every tube. The tube ⁇ are gently agitated, left at room temperature for about two hour ⁇ , and then placed at 4'C. for at least eight hours. Following incubation with the second antibody, the tubes are centrifuged at 2000 g for 30 minutes at 4 degrees in a refrigerated IEC-PR6 centrifuge.
- the ⁇ upernatant ⁇ are a ⁇ pirated by vacuum suction, with care taken not to disturb the precipitates packed in the bottom of tube ⁇ .
- the pellet i ⁇ resuspended in 2 ml cold PBS and recentrifuged as described above. Washing the precipitates with buffer increase ⁇ the reproducibility.
- Example 11 Measurement of Autophosphorylation of the EGF Receptor Autophosphorylation of the EGF receptor wa ⁇ mea ⁇ ured according to Bellot et al . (Bellot) .
- Cells were washed once with binding buffer. The plates were then placed in a water bath at 37 degrees and different concentrations of the analogues were added to the cells for ⁇ pecific period ⁇ of time.
- the buffer wa ⁇ removed and cells were scraped off the plates with 0.5 ml of Laemmli's ⁇ ample buffer, boiled for 5 minutes, and ⁇ onicated for 10 seconds. Aliquot ⁇ of each ⁇ ample were run on two different SDS-polyacrylamide gel ⁇ (7%) and each gel was transferred to nitrocellulose paper.
- the cell ⁇ were incubated with the ⁇ ub ⁇ trate analogue ⁇ , ⁇ uch as DHA-SPRCS or DHA-STRS for 24 hour ⁇ , then harve ⁇ ted and homogenized.
- the reaction volume 100 microliter ⁇ , and homogenized in a Dounce homogenizer 30 time ⁇ in 5 vol buffer (50 mM Tri ⁇ -HCl, pH 7.8, 50 mM MgCl2, 10 icromolar ⁇ odiumvanadate, 1 mM EDTA, and 50 microgram/ml aprotinin.
- the reaction volume of 100 ml contained 50 mM Tris-HCl, pH 7.8, 50 mM MgCl2, 10 micromolar sodium vanadate, 0.1% nonidet P-40, 5 micromole gamma 32p-ATP, ImM substrate and 60 microliter homogenate.
- the as ⁇ ay wa ⁇ initiated by addition of the ATP. After incubation the reaction wa ⁇ stopped by addition of trichloroacetic acid, and the supernatant was spotted on a 2 x 2 cm pho ⁇ phocellulo ⁇ e paper (Whatman P-81) .
- the paper ⁇ quare ⁇ were wa ⁇ hed with pho ⁇ phoric acid and acetone, and the dried paper ⁇ were counted for radioactivity in ⁇ cintillation fluid.
- Example 12 Preparation of DHA-EGFA (SEP. ID. NO: 6) 0.035 mM of EGFA was ⁇ u ⁇ pended in 2.5 ml of water- acetone (1:1), then 0.035 mM DIEA (6.5 ul) and 0.035 mM of
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Endocrinology (AREA)
- Gastroenterology & Hepatology (AREA)
- Zoology (AREA)
- Toxicology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
A fatty acyl-peptide composition useful as an inhibitor of cell proliferation is disclosed. The composition includes a polyunsaturated fatty acyl moiety covalently attached to a peptide. Also disclosed is a method of enhancing anti-proliferative activity of a peptide, by covalently linking the peptide to a polyunsaturated fatty acid.
Description
POLYUNSATURATED FATTY ACYL-PEPTIDE COMPOSITION
Field of the Invention
The present invention relates to polyunsaturated fatty acyl-peptide composition useful in inhibiting cell proliferation.
References
Ahn, N. G., et al., (1991) J. Biol . Chem. 266:4220- 4227.
Bellot, F. (1990) J. Cell Biology, HC 491-502. Berridge, M. J. (1987) Ann. Rev. Biochem. 51:159-193.
Calabrese, P. and Chabner, B.A. In: The Pharma¬ cological Basis of Therapeutics, Gilman, A.G. et al. (eds.) Pergamon Press, New York.
Casneillie, J. E. (1982) Proc. Natl. Acad. Sci. 79.:282-286.
Chabner, A. B., et al., (1990) In: Cancer Chemo¬ therapy: Principles and Practice, Chabner, A. B. et al. (eds.) W. B. Saunders Co., pages 154-179.
Hashida, T. and Yasumoto, S. (1990) Biochem. Biophys. Res. Comm. 172:958-964.
Kano, J., et al., (1991) Biochem. Biophys. Res. Comm. 179:97-101.
Keri, G., et al., (1991) Tumor Biol. 12:61-67.
Kumagai, H., et al., (1991) Biochem. Biophys. Res. Comm. 177:74-82.
Meyers, C . (1980) Proc. Natl. Acad. Sci. 17:6171- 6174) .
Shaw, B. R. et al . (1976) Proc. Natl. Acad. Sci. USA 21:505-509. Schally, A. V. et al. (1986) In: Neural and
Endocrine Peotides and Receptors, pages 73-88.
Stewart, et al . (1984) In: Solid Phase Peptide .Synthesis, 2nd Edition, Pierce Chemical Company, Rockford, Illinois.
Weinstein, I.B. (1988) Cancer Res . 41:4135-4143. Yarden, Y. and Ullrich, A. (1988) Annual Rev. Biochem .
52:443-478.
Background of the Invention
Uncontrolled cell proliferation is a characteristic of a number of diseased states. Such growth is observed, for example, in benign and malignant tumors, certain virally- induced diseases and psoriasis. Generally, drugs used to treat cellular abnormalities characterized by uncontrolled cell growth target important biochemical steps or processes that are part of the cell growth cycle. However, ^such drugs lack selectivity and inhibit the growth of both diseased and healthy cells. Therefore, development of chemotherapeutic agents having relatively high selectivity for the diseased cells would be advantageous. Agents based on peptide hormones, steroid hormones, hormone-releasing factors, and their respective antagonists and agonists are relatively specific to their target cells. Proliferative cells, such as neoplastic cells or tumors, which arise from hormone-sensitive tissues generally are found to have hormonal requirements that are similar to those of their healthy counterparts. By altering the amount of hormone in the blood circulation it may be possible to selectively restrict the growth of these cells. However, due to the relatively high levels of hormone required for such treatment, use of hormonal chemotherapeutic agents is still limited in vivo by toxic side effects to normal cells .
Likewise, cell proliferation may also be inhibited by targeting one or more of the cellular signal transduction systems implicated in the regulation of cell division.
These include a) the tyrosine kinase signal transduction
pathway, b) the phospholipid metabolism/protein kinase C signal transduction pathway, and c) the cAMP protein kinase A signal transduction pathway. These pathways are activated by endogenous ligands which initiate a cascade of signalling events that eventually results in cell division.
Protein kinases have been found to be particularly important regulators of these pathways. For example, tyrosine kinases are known to play a critical role in the regulation of cell division. High levels of tyrosine kinase activity have been measured in highly proliferative cells, such as neoplastic cells. Inhibition of such phosphorylation activity can be correlated with a reduction in cell division, in some cases.
The current invention is directed to fatty acyl- peptide compositions having enhanced biological activity, compared to the peptide alone. Peptides used to form compositions directed to the inhibition of cell proliferation include peptide hormones, peptide hormone analogues, and protein kinase peptide substrates or peptide inhibitors. In experiments in support of the current invention, it has been found that linking such peptides to the polyunsaturated fatty acids lowers the concentration at least several-fold of such peptides required to inhibit cell proliferation. Such low chemotherapeutic drug concentrations confer the advantage of reducing toxicity to healthy cells.
Summary of the Invention
One general object of the invention is to provide a fatty acyl-peptide composition which is useful in inhibiting cell proliferation, such as neoplastic cell proliferation. The composition includes a peptide having antiproliferative activity and conjugated to the peptide, a polyunsaturated fatty acyl moiety. The composition is characterized by a cell proliferative inhibitory activity
which is several-fold greater than that of the antiproliferative peptide alone.
In one embodiment the fatty acyl moiety of the composition is a docoεahexaeπoyl or an eicosapentaenoyl moiety. In a preferred embodiment, the fatty acyl moiety is a ciε-4, 7, 10, 13, 16, 19-docosahexa enoyl (DHA) or cis- 5, 8, 11, 14, 17-eicosapentaeπoyl (EPA) moiety. The fatty acyl moiety is preferably conjugated to the peptide through an amide linkage. In another embodiment, the peptide portion of the composition is a peptide hormone, and in a preferred embodiment, the peptide hormone is a somatostatin analog or a gonadotropin releasing hormone (GnRH) analog. In yet another preferred embodiment, the peptide hormone has the sequence of SEQ ID NO: 4, and in another preferred' embodiment, the peptide hormone of the composition has the peptide sequence of SEQ ID NO: 5.
The peptide used in forming the fatty acyl-peptide antiproliferative composition can also be a protein kinase modulatory peptide. In a preferred embodiment, the protein kinase modulatory peptide has a sequence selected from the group consisting of SEQ ID NO: 1, SEQ ID NO: 2, SEQ ID NO: 3, and SEQ ID NO: 6.
Another general object of the invention is to provide a method for producing enhanced biological activity of a peptide. In one aspect, the invention is used for producing a several-fold enhancement of cell proliferative inhibitory activity in a peptide composition. According to the invention, this enhancement is achieved by conjugating the peptide to a polyunsaturated fatty acid. Preferred peptides and fatty acyl moieties in the method are described above.
In a more general aspect, the invention provides fatty acyl-peptide compositions having enhanced biological activity, compared to the activity of the peptide alone.
Such enhanced biological activity includes such activities
as enhanced hormone activity, anti-tumor activity, enhanced immunogenic activity and other peptide-specific activities.
These and other objects and features of the present invention will become more fully apparent when the following detailed description of the invention is read in conjunction with the accompanying drawings.
Brief Description of the Figures
Figure 1 shows exemplary peptide sequences, designated sequences 1-31 and identified by SEQ ID NO: 1-31, respectively, used in forming the novel peptide-fatty acyl compositions of the invention: STKS (sequence 1, SEQ. ID NO: 1), STKSI (sequence 2, SEQ ID NO: 2), SPKCS (sequence 3, SEQ. ID NO: 3) , εomatostatin analogue (sequence 4, SEQ. ID NO: 4 ), where lower case "d" signifies the presence of a D-amino acid residue and '"NH2" at the C-terminal signifies the amidation of the C-terminal end, GnRH (sequence 5, DHA-SEQ. ID NO: 5), where "Glp" signifies the presence of pyroglutamate, EGFA (sequence 6, SEQ ID NO: 6); an N-terminal sequence of human PTH (sequence 7, SEQ ID NO:
7) , a polypeptide fragment derived from fibronectin (sequence 8, SEQ ID NO: 8), and a T cell epitope modulatory peptide (sequence 9, SEQ ID NO: 9) suitable for use in forming compositions of the present invention; the sequences of protein kinase modulatory peptides, such as tyrosine kinase inhibitory peptides (sequence 10, SEQ ID NO: 10), (sequence 11, SEQ ID NO: 11), (sequence 12, SEQ ID NO: 12) where "ACM" signifies that the cysteine εulfhydryl group has been modified by an acetamidomethyl group, (sequence 13, SEQ ID NO: 13) and (sequence 14, SEQ
ID NO: 14), calmodulin-dependent protein kinase III inhibitory peptide (sequence 15, SEQ ID NO: 15) , dsDNA- dependent kinase inhibitory peptide (sequence 16, SEQ ID NO: 16), protein kinase C modulatory peptide (sequence 17, SEQ ID NO: 17), and other protein kinase modulatory pep¬ tides, such as for cAMP dependent kinase (sequence 18, SEQ
ID NO:18) , (sequence 19, SEQ ID NO: 19) , and (sequence 20, SEQ ID NO: 20), phosphorylaεe kinase (sequence 21, SEQ ID NO: 21), calmodulin-dependent kinase I and II (sequence 22,
SEQ ID NO: 22 and sequence 23, SEQ ID NO: 23) , dsRNA- dependent kinase (sequence 24, SEQ ID NO: 24) , proline- dependent kinase (εequence 25, SEQ ID NO: 25 and sequence
26, SEQ ID NO:26), growth factor-regulated kinase (sequence
27, SEQ ID NO: 27), casein kinaεe I and II (sequence 28, SEQ ID NO:28 and εequence 29, SEQ ID NO: 29, AMP-activated protein kinaεe (εequence 30, SEQ ID NO:30), and S6 kinase
II (sequence 31, SEQ ID NO:31);
Figure 2 illustrates coupling of ciε-4, 7, 10, 13 , 16, 19- docoεahexaenoic acid (DHA) to a peptide (εequence 1, SEQ ID NO: 1) through an N-terminal amine group to form the fatty acyl-peptide compoεition (compoεition I, DHA-SEQ ID NO: 1) ; and
Figure 3 εhows sequences of exemplary fatty acyl-pep¬ tide compositionε of the invention: DHA-STKS (compoεition I, DHA-SEQ. ID NO. : 1) , DHA-STKSI (composition II, DHA-SEQ ID NO: 2), DHA-SPKCS (composition III, DHA-SEQ. ID NO. : 3) ,
DHA-somatostatin analogue (composition IV, DHA-SEQ. ID NO. : 4 ), D-Lys6 (DHA) -GnRH (composition V, DHA-SEQ. ID NO. : 5), and DHA-EGFA (composition VI, DHA-SEQ ID NO: 6) , where DHA is a cis-4, 7, 10, 13, 16, 19-docosahexaenoyl moiety, Glp represents pyroglutamate, and lower case "d" signifies the presence of a D-amino acid residue, and "NH2" at the C- terminal εignifieε the amidation of the C-terminal end. Detailed Description of the Invention
I. Definitions The term "polyunsaturated fatty acid" refers to a com¬ pound having a carboxylic acid moiety and a long unbranched carbon chain, uεually containing between about 8 and 24 carbon atoms, and containing two or more carbon-carbon double bonds. When a fatty acid iε conjugated to a peptide through an amide linkage, a fatty acyl-peptide derivative is formed. The disclosed invention includes fatty acyl-
peptide derivatives, and, more generally, peptides linked to long unbranched, polyunsaturated carbon chains. The term "peptide hormone" refers to a peptide that elicitε a biological response in a target cell. Peptide hormones are generally low molecular weight proteins (<
50,000) . Such hormones can be isolated from biological sources, chemically synthesized or produced by recombinant methods. Generally, in their natural setting, peptide hor¬ mones are εecreted from specific cells and produce biological effects in other cells. Analogues of such naturally occurring hormones are encompasεed by the term "peptide hormone" and are produced recombinantly or synthetically.
Peptide hormones, as described herein, are divided into two main categories, according to their known acti¬ vities in vivo . Generally J hormones act either directly or indirectly to produce the desired biological effect. In the context of the present invention, the desired effect is inhibition of cell proliferation. Peptide hormones that act directly on a cell to inhibit division of the cell are referred to herein as direct peptide hormones; those pep¬ tides that act to, stimulate or inhibit synthesis or secretion of endogenous peptide growth regulatory hormones are referred to as indirect peptide hormones. Generally, it will be εeen that an indirect peptide hormone effectε a change (increaεe or decrease) in the extracellular levels of a naturally occurring direct pep¬ tide hormone. Two exemplary types of indirect peptide hor¬ mones are peptide hormone-releasing hormones and peptide hormone release-inhibiting hormones. Naturally occurring indirect peptide hormones are generally short polypeptides, usually under 20 amino acids in length.
Peptide hormone analogs are synthetically or recombinantly prepared peptides which are structurally similar to naturally occurring peptide hormones. For the purposeε of the present invention, such peptide analogs are
included by the term "peptide hormone." Generally, to be useful in the present invention, such analogs have essentially a similar or a higher biological activity than that of the endogenous peptide hormone. Protein kinase modulatory peptides are peptides which act as inhibitorε of protein kinase activity. Such pep¬ tides may act, for example, as protein kinaεe substrates, residue containing a phosphorylatable amino acid/in the εequence.
Alternatively, such kinase modulatory peptideε may bind to the kinaεe catalytic site, to effect inhibition of kinaεe activity. Such kinaεe modulatory peptides act to produce reduced phoεphorylation of endogenous protein kinase substrateε.
Peptideε and compoεitions that inhibit cell proliferation are εometimeε referred to herein as. "anti¬ proliferative" peptides or compositions.
II. Selection of Peptides for Use in the Fatty acyl-Peo- tide Composition A. Selection of Peotide Hormones
The present invention is directed to peptide compositions having anti-proliferative activity. More generally, it is the discovery of the invention that these fatty acyl-peptide compositionε have enhanced biological activity in comparison to underivatizea peptides. Anti¬ proliferative fatty acyl-peptide compositions of the invention are effective to inhibit proliferation of highly proliferative cells, such as neoplastic cells or virally- infected cells. For use in the compoεitionε of the invention, peptides known to have anti-proliferative activity are linked to a fatty acyl moiety, εuch aε a docoεahexaeπoyl (DHA) or eicoεapentaenoyl (EPA) group, aε described below. Peptide hormones having cell proliferative inhibitory activity are known in the art, and may be direct or indirect hormones. Such anti¬ proliferative peptide hormoneε are uεually, but not
necessarily, specific to a particular type of cell, such as a cell with εpecific hormonal requirementε .
1. Indirect Peotide Hor oneε. As noted above, 5 in the context of the present invention, indirect peptide hormones include those peptides which act to effect a decrease in the level of a direct cell proliferation εtimulatory hormone or to effect an increase in the level of a direct cell proliferation inhibitory hormone.
10 An example of an indirect peptide hormone which stimulates release of an antiproliferative hormone in some mammalian species is gonadotropin releaεing hormone (GnRH) . GnRH stimulates release of several gonadotropins, including luteinizing hormone (LH) . GnRH and GnRH analogues can also
15 inhibit the growth of hormone dependent carcinomas, such as androgen-dependent prostate tumors (Schally) . GnRH is also effective against such tumors aε colon and pancreatic tumors. Figure 1 shows a sequence of a GnRH analog useful in treating such tumorε (5, SEQ ID NO: 5) . Figure 3 εhows
20 a DHA-GnRH fatty acyl-peptide composition of the invention
(V, SEQ ID NO: 5) having enhanced activity, as described below.
GnRH analogues can be εcreened for potential anti¬ proliferative activity, by assessing their abilities to
25 stimulate release of LH from anterior pituitary cells, aε detailed in Example 9. Active GnRH analogue peptides are then used to form fatty acyl peptide compositions of this invention, and tested for enhanced biological activity, as discussed below.
30 Indirect peptide hormones also include peptide hormone release-inhibiting hormoneε. An exemplary peptide hormone of thiε type iε εomatoεtatin. Somatostatin iε a 14 amino acid peptide that inhibitε the releaεe of growth hormone (GH) . GH enhances the proliferation of its cellular ■35 targets. Native somatostatin peptide and analogs are used to form fatty acyl peptide derivative compositionε of the
invention, as described below. An analog of somatostatin used to form fatty acyl peptide compositions in εtudies described herein haε the sequence 4 (SEQ ID NO: 4) in Figure 1, and a fatty acyl-peptide compoεition which includeε thiε peptide iε shown as composition IV (DHA-SEQ
ID NO: 4) in Figure 3. The somatostatin fatty-acyl peptide is about 150 times more potent than the parent molecule.
It can be appreciated that other indirect acting pep¬ tide hormoneε can be used in forming fatty acyl-peptide compositions of the invention. Preferably, peptides are selected based on their known anti-proliferative activity in a cell proliferation asεay, described in Section III below. Selected peptides are then conjugated to fatty acyl moieties, and are tested for enhanced potency in such aεεays . A fatty-acyl peptide derivative composition iε a useful anti-tumor or anti-neoplastic cell agent, when it is found to have at least a several-fold increase in potency, in comparison to the unconjugated peptide.
2. Direct Peotide Hormoneε. Also used in forming compositions of the invention are peptides which are known to have direct antiproliferative effects on cells. An exemplary direct peptide hormone is parathyroid hormone (PTH) . This peptide iε 84 amino acids in length, and can directly inhibit osteoblaεt division. Certain bone cancers are characterized by hyperproliferating oεteoblastε. Structure-function studies indicate that an N-terminal fragment of PTH is active in inhibiting osteoblaεt proliferation (Kano) . Thiε εequence 7 (SEQ ID NO: 7) iε εhown in Figure 1.
Another example of a peptide which iε defined as a direct peptide hormone for purpoεes of thiε invention is a peptide fragment of fibronectin having the εequence SEQ ID NO: 8, εhown in Figure 1 as sequence 8. This fragment spans the recognition site within the fibronectin molecule to which cells bind for attachment to fibronectin. This
fibronectin recognition site peptide and analogues thereof are uεed to inhibit fibronectin-mediated cell attachment and εpreading in vitro. They may also be important for regulation of tumor cell proliferation and metastaεiε in vivo (Kumagai) . According to the preεent invention, fibronectin recognition site peptides are coupled to fatty acyl moieties to form compositions useful in inhibiting tumor cell proliferation and metaεtasis.
Direct peptide hormoneε, aε defined in the current invention, alεo encompaεs peptides derived from immunogenic polypeptides for modulating a response from εpecific immune cell populations. For example, a T-cell epitope peptide (TCEP) iε coupled to a fatty acyl moiety as described herein to form a composition useful in modulating the T- cell immune response. The sequence of TCEP (SEQ ID NO:9) is shown in Figure 1 as εequence 9, and εyntheεiε of a DHA derivative of TCEP is detailed in Example 7.
B. Selection of Protein Kinase Modulatory Peptides Elevated protein kinase C and tyrosine kinase activities are associated with neoplastic cell prolif¬ eration or transformation (Weinεtein, Yarden) . Inhibition of such kinase activities can be effected by the presence of small peptide fragments which mimic protein εubεtrate phoεphorylation sites and/or protein kinase modulatory domains. Such protein kinase modulatory peptides are effective to compete with endogenous protein kinaεe subεtrateε. Selection of εpecific protein kinase modulatory peptides for use in forming the fatty acyl-pep- tide derivatives of the invention iε deεcribed below.
1. Tyrosine Kinase Substrates and Inhibitors. Receptors for a number of growth factors, including epidermal growth factor (EGF) , insulin growth factor (IGF) , and platelet derived growth factor (PDGF) , contain tyrosine kinase catalytic domains which phosphorylate specific
intracellular protein substrates, including the receptor itself, in some cases, at tyrosine residues. Cellular transformation by a virus, such as the Rous sarcoma virus, can result from expression of a single viral protein, which functions as a tyrosine kinaεe.
Tyrosine kinaεes may also play a role in the uncontrolled growth of keratinocytes which can result in psoriasiε or other highly proliferative skin diεorderε. Keratinocytes posεesε tyrosine kinase-like growth factor receptors.
High tyrosine kinase activity has been associated with high rates of cellular proliferation (Casneillie, Keri) . Inhibition of such kinase activity decreases the rate of cellular proliferation. Several tyrosine kinase modulatory peptides which are effective to inhibit tyrosine kinase activity in vi tro have been described in the literature, and some are available commercially, for example from Pep¬ tides International (Louisville, KY) . Figure 1 shows εequenceε of εeveral exemplary tyrosine kinase modulatory peptideε which can be uεed to form anti-proliferative compoεitionε of the invention, εuch aε STKS (SEQ ID NO: 1) and STKSI (SEQ ID NO: 2), εequenceε 1 and 2, reεpectively.
Alternatively, tyrosine kinase modulatory peptides can be identified, based on peptide εequenceε surrounding phosphorylation sites of endogenous protein kinaεe εubεtrates in highly proliferating cells. Preferably, cellular sources used for identification of εuch substrate peptides are proliferating cells of the type to be targeted; however, it is appreciated that tyrosine kinase modulatory peptideε derived from one cell εource may inhibit a tyrosine kinase derived from a different cell type.
In order to isolate an endogenous protein kinase substrate protein, proliferating cells, such aε a lymphoma cell line having a high level of tyrosine kinase activity, are incubated with [32p] -phoεphate, or a particulate
fraction of the cells iε incubated with radiolabelled [gamma 32p] ATP. Cellular polypeptideε are εeparated by denaturing gel electrophoreεiε, and the phoεphorylated proteinε are observed by autoradiography. Phosphoprotein- containing bands are excised from the gel, and the phosphoprotein iε eluted from the gel band. The eluted protein iε εubjected to partial hydrolyεis, and the identity of the phosphorylated amino acid determined, according to methods known in the art (Casneillie) . In order to determine the peptide εequence in the vicinity of the phoεphotyroεine residue (ε), phosphoproteinε are εubjected to peptide fragmentation, such aε by proteolyεiε or chemical means, and phosphotyrosine-containing peptides are sequenced, according to conventional methods (Casneil- lie) . Short peptides (approximately 6-20 amino acids) corresponding to the peptide sequenceε in the vicinity of the phoεphorylation εite can be εynthesized and teεted for inhibition of tyroεine kinase activity.
Tyrosine kinase modulatory peptides can be prepared by substituting for the tyrosine phosphorylation εite residue an amino acid residue that cannot be phosphorylated, εuch as a phenylalanine residue, composition II in Figure 3 (DKA-STKSI) εhowε an inhibitory fatty acyl-peptide compoεition, the peptide portion of which haε almoεt complete identity with the peptide portion of composition I
(DHA-STKS), except that in composition II a phenylalanine residue has been substituted for a tyrosine residue present in composition I. Both peptides inhibit proliferation of neoplaεtic cellε. Accordingly either peptide can be conjugated to a fatty acyl group, to form an anti-tumor fatty acyl-peptide compoεition of the invention, aε described in Section III, below.
2. Protein Kinase C (PKC) Peotide Substrates. PKC is a component of the phospholipid metabolism/protein kinase C signal tranεduction pathway which playε a critical
role in normal cellular growth control. Activation of PKC is mediated by a family of G-protein-modulated receptors. When activated, the cytoεolic form of PKC binds to the cytoplasmic face of the plasma membrane. One of the known protein targets of PKC iε the EGF receptor. Phoεphoryla- tion of EGF receptor by protein kinaεe C results in a decrease in the affinity of the receptor for EGF and a decrease in EGF receptor-associated tyrosine kinase activity (Berridge) . Thiε illustrates the heterologous interactionε between signal transduction pathways, specifically between the tyrosine kinase pathway and the phospholipid metabolism/protein kinaεe C pathway.
Recent results demonstrate that protein kinaεe C activity inhibition is directly related to the anti- proliferative activity of certain gonadotropin-releasing factor agonists. These agonists are effective in inhibiting DNA synthesis in certain breast cancer cell lines. Both follicin and buserelin, the two tested gonadotropin-releasing factor agonists, inhibit protein kinase C activity and tyrosine kinase activity (Keri) . It has also been demonstrated that somatostatin analogues can stimulate a phosphatase activity in tumor cellε thereby inhibiting εignal transduction and tumor growth (Schally) . The sequences of a number of peptide substrates for PKC are known and are commercially available. Figure 1 lists the εequenceε of some of the known peptide substrate inhibitors for PKC, such as sequence 3 (SEQ ID NO: 3) and sequence 17 (SEQ ID NO: 17). These peptides are suitable PKC modulatory peptides for use in forming fatty acyl-pep- tide compositions of the present" invention. PKC peptide inhibitors can also be identified from endogenous protein subεtrates of protein kinase C, using the same general procedures described in part 1, above, for tyrosine kinases.
3. Other Protein Kinases. Modulatory peptides targeting protein kinases other than tyrosine kinase and protein kinaεe C can also be used to form fatty acyl-pep¬ tide anti-tumor compoεitionε of the invention. One εuch 5 protein kinaεe target iε a caεein kinaεe II found in epidermal keratinocyteε. High proliferation rates are related to the hyperphoεphorylation of an oncogenic product from a human papillomavirus which contains a casein kinase II phosphorylation consenεus εequence (Hashida) . This
10 casein kinase II consenεuε phoεphorylation sequence iε shown in aε sequence 29 in Figure 1 (SEQ ID NO: 29) .
Another protein kinaεe, cyclic AMP-dependent protein kinase (PKA) , iε a component of the cAMP-dependent protein kinase signal tranεduction pathway. PKA phoεphorylates
15 threonine and εerine residues of a protein substrate. The activity of this enzyme iε 'enhanced by cAMP. PKA may regulate growth control. In εome animal cells, an increase in cAMP levels activates specific genes involved in growth regulation. In neuroendocrine cells of the hypothalamus,
20 cyclic AMP turns on the gene that encodes the peptide hor¬ mone release-inhibiting hormone, somatostatin.
Other target protein kinases, in conjunction with the present invention, are components of signalling cascades involved in cell proliferation. For example, when
25 epidermal growth factor (EGF) binds to the epidermal growth factor receptor (EGFR), a tyrosine kinase, it stimulateε activation of myelin basic protein (MBP) kinase, through phosphorylation of MBP kinase. MBP kinase, in turn, activates S6 kinase by a phosphorylation event. The result
30 of activation of this signalling pathway is stimulation of proliferation of EGFR-bearing cells, εuch as adipocyte cells (Ahn) . It can be appreciated that fatty acyl-peptide compositions of the invention using aε peptide co ponentε, phoεphorylation recognition εequence peptideε for one or , 35 more of EGFR kinaεe, MBP kinase and S6 kinase will serve to inhibit the signalling cascade of which these kinases are a
part. Sequences of exemplary peptide inhibitors of these kinases are shown in Figure 1 (e.g. , sequence 11, SEQ ID NO: 11, to sequence 31, SEQ ID NO:31) .
Other examples of protein kinase activities involved in cell proliferation whose activities can be modulated, using fatty-acyl peptide compositions of the present invention, include calmodulin-dependent kinaseε I and II, cGMP-dependent protein kinase, ds-DNA-dependent protein kinase, proline-dependent kinaεe, and AMP-activated kinaεe. Sequences of exemplary peptide modulatory peptideε directed to theεe kinaεes are listed in Figure 1 (SEQ ID NOs: 18- 31) .
Ill. Preparation of Fatty acyl-peptide Compoεitions In accordance with the present invention, it has been observed that conjugation of fatty acyl groupε to the above-deεcribed peptideε enhances the biological activity of the peptides. Enhancement of biological activity iε exemplified herein aε enhancement of inhibition of cell proliferation, εuch as neoplastic cell proliferation. In experiments carried out in support of the present invention, exemplary peptide hormones and protein kinase modulatory peptides were selected, as described above, and isolated from natural sources or synthesized, as described below. Subsequently, the peptideε were conjugated to fatty acyl moietieε, aε described below and illuεtrated in Figure 2 and teεted for thei^ activities in cell proliferation assays, as described in Section III, below.
A. Solid Phase Peptide Svntheεis
Peptides shorter than about 30 amino acids in length are conveniently prepared by methods commonly used in solid-phase peptide synthesis, as detailed below (Stewart) .
Briefly, N-alpha-protected amino acid anhydrides are prepared in crystallized form and used for successive amino acid addition to the peptide N-terminus. At each residue
addition, the growing peptide on a solid support is acid treated to remove the protective group, and washed several times to remove residual acid. The peptide iε then reacted with another N-protected amino acid. The amino acid addition reaction may be repeated two or three timeε to increase the yield of growing peptide chains. After completing the growing peptide chains, the protected pep¬ tide reεin iε treated with liquid hydrofluoric acid to deblock and release the peptideε from the εupport.
B. Conjugation of Fatty Acyl Moieties to Peptides Preferred fatty acyl moietieε for use in the present invention are those with a high degree of unsaturation, and include such fatty acids as cis-5, 8, 11, 1 , 17- eicoεapentaeπoic (EPA) and ciε- ,7, 10, 13, 16, 19- docoεahexaenoic acid (DHA) . .Such polyunsaturated fatty acids can be prepared synthetically according to standard methods, isolated from the oils of marine fish, or obtained from commercial εourceε . Generally, fatty acidε are linked to the peptideε via the terminal amine group or via internal amine groups, such as the amine group of lyεine, in an amide linkage. Figure 2 illustrates a scheme for coupling of εequence 1 to DHA by the terminal amino group of the peptide through an amide linkage to form the fatty acyl-peptide compoεition I in
Fig. 3. DHA is activated by reaction with N- hydroxyεuccinimide prior to reaction with an a ino-group containing polypeptide. In another embodiment, DHA iε coupled by the free amine group of a lysine residue in the εequence to form the fatty acyl composition, εuch aε compoεition V in Fig. 3.
Exampleε 2-6 detail preparation of fatty acyl-peptide compositions in which acylation of peptides iε carried out uεing activated eεter or acyl chloride derivativeε of DHA or EPA. Activated esters useful in preparing compositionε of the current invention include DHA-O-benztriazole ester
(DHA-OBT) , DHA-O-pentafluorophenyl ester (DHA-Opfp) , and DHA-O-εuccin midyl ester (DHA-O-N-Succ) . The acylation is carried out as detailed in Example 2. Crude products, obtained after acylation, are purified by HPLC. The purity of the final productε lε characterized by analytical HPLC and TLC data, while the chemical characterization lε accompliεhed by mass εpectrometry (MS) .
C. Fattv Acyl-Peotide Compositions From the foregoing discusεion, it can be appreciated that peptides can be selected for use in anti-proliferative fatty acyl-peptide compositionε of the invention, baεed on their abilities to inhibit cell proliferation or to inhibit componentε, such as protein kinase componentε, of cell proliferative εtimuluε pathwayε. According to an important feature of the preεent invention, conjugating to εuch a peptide, a polyunεaturated long chain carbon, εuch as a polyunsaturated fatty acyl moiety described above, enhances the biological activity of the peptide. Specifically, conjugation of a polyunsaturated fatty acyl moiety to an anti-proliferative peptide enhances its ability to inhibit neoplastic cell proliferation. This aspect of the invention will be better appreciated from the discussion below. As an example, tyrosine kinase modulatory peptideε selected aε deεcribed above are covalently linked to polyunsaturated fatty acids to form compositions which are effective to inhibit proliferation of neoplastic cells. One εuch compoεition uεed in experimentε carried out in εupport of the current invention lε DHA-STKS (DHA-SEQ. ID
NO. 1) illustrated in Figure 3 (composition I).
Fatty acid-tyroεine kinase inhibitory peptide compo¬ sitions can also be prepared by substituting for the tyro¬ sine residue an amino acid reεidue that cannot be phoε- phorylated. In one such compoεition, DHA-STKSI (DHA-SEQ ID
NO: 2; composition II) , the tyrosine residue of tyrosine
kinase subεtrate DHA-STKS (DHA-SEQ ID NO: 1; compoεition I) haε been εubεtituted by a phenylalanine residue. Both fatty acid-protein kinase peptide subεtrate and peptide inhibitor compositions inhibit the proliferation of neoplastic cells, as detailed in Example 8 and shown in
Table 1. Another such tyrosine kinase inhibitor peptide is DHA-EGFA (DHA-SEQ ID NO: 6, composition VI) also shown in Table 1.
A PKC peptide subεtrate-fatty acid compoεition uεed in the experimentε deεcribed in Example 8 includeε a peptide fragment from myelin baεic protein, and has the structure shown in Figure 3 as compoεition III (DHA-SEQ. ID NO: 3) . Additionally, endogenous substrates of PKC may be identified as described above for the tyrosine kinase substrates. The polypeptide fragments that contain PKC phosphorylation sites are then sequenced and uεed to form the fatty acid peptide compositions of the invention.
IV. Anti-Proliferative Assays In accordance with the present invention, fatty acyl- peptide derivatives are prepared as described in Section II, above, and inhibition of neoplastic cell proliferation by such compositions is measured, according to one or more standard cell proliferation assays. In one exemplary asεay, deεcribed in Example 8, inhibition of cell proliferation is meaεured directly, by measuring the number of surviving cells after exposure of the cells to a test composition, εuch aε a fatty acyl-peptide compoεition diεcloεed herein. Alternatively, an activity which correlateε to cell proliferation can be meaεured to determine indirectly the effect of a teεt compound on cell proliferation. Thus, activitieε εuch as tyrosine kinaεe activity or releaεe of a growth-promoting hormone, which are known to correlate with neoplaεtic cell proliferation, are uεed aε indicators of anti-proliferative activity. As exemplified herein,
polyunsaturated fatty acyl-peptide anti-proliferative compositions may exhibit one or more of the following in vi tro activities: (a) inhibition of cell proliferation, (b) inhibition of tyrosine kinase activity, (c) stimulation of release of a growth-inhibiting hormone, such as lu- teinizing hormone, and (d) inhibition of release of growth- εtimulating hormoneε, εuch aε inhibition of growth-hormone releaεe. It is appreciated that other in vi tro assayε which can be correlated with uncontrolled cell growth may also serve as assayε for εelecting and determining the activities of fatty acyl-peptide compoεitionε of the invention. More εpecifically, εuch assayε can εerve to determine the relative potencieε of compositions of the invention as anti-proliferative agents.
A. Inhibition of Cell Proliferation
An asεay for meaεuring the inhibitory effectε of polyunεaturated fatty acyl-peptide derivativeε on cell proliferation is described in detail in Example 8. In experimentε carried out in support of the invention, a number of tumor cell lines, including a human prostatic adenocarcinoma cell line, a human breast adenocarcinoma cell line and a human colon adenocarcinoma cell line, were used to test compoundε of the invention for effectε on cell proliferation. Cells were exposed to the tyrosine kinase synthetic peptide εubεtrate fatty acid derivative DHA-STKS (DHA-SEQ ID NO: 1), the protein kinaεe C peptide εubstrate fatty acid derivative DHA-STKCS (DHA-SEQ ID NO:3), or the gonadotropin releaεe hormone fatty acid derivative DHA-D- Lyε6-GnRH (SEQ ID NO: 5), as described below.
Solutionε of the peptide derivativeε were added in culture media to test cells for an incubation period of 2.5 hours. At the end of the test period, cells were centrifuged, the pellet diluted with 1% BSA in saline and viable tumor cell counts were determined by the trypan blue exclusion method. Results of tests using peptide fatty
acid derivatives DHA-STKS (DHA-SEQ. ID NO: 1) and DHA-D- lys6-GnRH (DHA-SEQ. ID NO: 5) in various transformed cell lines are εhown in Table 1. Cell proliferation waε inhibited by at leaεt 50% at a peptide-fatty acid derivative concentration of 10 micrograms/ml of the TK modulatory peptide εubεtrate, the PKC modulatory peptide, and the gonadotropin-releaεing hormone analogue, aε εhown in Table 1.
Table 1
Effect of Fatty Acid-Peptide Compoεitions on the
Inhibition of Cell Proliferation
(% Inhibition)
*Neoplastic cell types tested are indicated in brackets, and are as follows:
PC 3, human prostatic adenocarcinoma cell line; MCF 7, human breaεt adenocarcinoma cell line; and HT 29: human colon adenocarcinoma cell line.
Additional methodε are available for monitoring the viability of cells, including asεays based on the differential dye uptake by viable cells in comparison to that taken up by non-viable cells. For example, viable cells take up diacetyl fluoreεcein and hydrolyze it to fluoreεcein, to which the cell membrane of live cells is impermeable. Live cells fluoresce green. Nonviable cells may be counter-stained with ethidium bromide and will fluoresce red. These methodε may be used in flow cytometric assays, in accordance with protocols known in the art .
B. Stimulation of Luteinizing Hormone (LH) Release
Peptide hormone-releasing hormones increase the extracellular levels of peptide -hormones that interact with εpecific cellular receptors. Compounds, such as peptide hormone-releasing hormones, which stimulate production of LH have also been shown to inhibit cellular proliferation.
In accordance with the invention, fatty acyl derivatives of εuch compoundε are effective to inhibit neoplastic cell proliferation at concentrations which are several-fold lower than the concentration of peptide alone required to inhibit such cell proliferation.
Specifically, as shown below, an analog of gonado- tropin releasing hormone analog conjugated to the poly¬ unsaturated fatty acid, DHA, (D-Lys6 (DHA) -GnRH; DHA-SEQ ID NO: 5) was tested in an aεεay of LH release by anterior pituitary cell suspensions as detailed in Example 9. In this aεεay, luteinizing hormone levelε were quantitated by a double antibody radioimmunoaεεay procedure. Aε εhown in Table 2, the polyunεaturated fatty acid-gonadotropin releaεe hormone analogue (D-Lyε6 (DHA) -GnRH) waε about 5- fold more effective in eliciting the releaεe of luteinizing hormone when compared to equivalent concentrations of GnRH or to the D-Lys6-GnRH analogue lacking the fatty acid moiety. The polyunsaturated fatty acid DHA alone had no effect on luteinizing hormone release at the concentration teεted (Table 2) . These results further demonstrate the effectiveness of compositions of the invention in enhancing biological activity of their peptide components.
Table 2 Effect of the Fatty Acid-GnRH Analogue Composition on LH Releaεe (% Change3)
C. Inhibition of Growth Hormone Release Peptide hormone-release inhibiting hormones decrease the extracellular levels of peptide hormones which interact with εpecific cellular receptorε. Generally, binding of peptide hormone to the receptor triggerε a caεcade of biochemical events mediated through second meεεengerε. In some cases, an end-result of such hormonal activity is cellular proliferation. Peptide hormone-release inhibiting hormoneε which act to inhibit such hormoneε are useful in forming fatty acid-peptide compoεitionε described by the preεent invention. Analogueε of εuch peptide hormone- release inhibiting hormones are also uεeful in forming εuch compoεitionε.
Somatoεtatin iε an exemplary peptide hormone-release inhibiting hormone analogue which inhibits hormonally activated cell proliferation. Somatostatin iε a growth hormone releaεe inhibitory hormone which inhibitε releaεe of growth hormone. Growth hormone (GH) binds to specific cell-surface receptors distributed widely throughout the body. Binding of GH agonists to GH receptorε reεultε in increaεed cellular division, and hence, cell proliferation.
In experimentε carried out in support of the preεent invention, a εomatoεtatin-fatty acyl analogue (SEQ ID NO:
4) was εynthesized as detailed in Example 5. Inhibition of growth hormone releaεe by the DHA-acylated somatostatin analogue was measured, as deεcribed in Example 10. GH levels were determined by a double-antibody radio- immunoassay for the hormone. In thiε aεεay, the DHA- somatoεtatin analogue waε 1000 timeε more effective in stimulating release of growth hormone than was either unacetylated somatostatin analogue or somatoεtatin.
D. Inhibition of Tyrosine Kinase Activity
As described above, proliferative activity of certain cells haε been correlated to tyroεine kinaεe activity preεent in the cellε. Aε deεcribed herein (Example 11), tyrosine kinase activity can be measured in cellε from the human breaεt adenocarcinoma line MDA-MB-231. Briefly, theεe cellε are incubated with a teεt compound, εuch as the polyunsaturated fatty acid-tyroεine kinase peptide inhibitor DHA-STKSI (DHA-SEQ ID NO: 2), then harvested and homogenized. Tyrosine kinase activity is measured by incorporation of radiolabeled phoεphate into an endogenous protein substrate in the cellε. In thiε caεe the endogenouε protein substrate is the EGF receptor, and the phoεphorylation event iε an autophoεphorylation event. The effect of DHA-STKSI on EGF receptor autophoε- phorylation iε shown in Table 3. At a concentration of 20 micrograms/ml, the underivatized peptide inhibitor, STKSI (SEQ ID NO:2), did not inhibit autophoεphorylation of EGF. In contraεt, DHA-STKSI (DHA-SEQ ID NO:2) inhibited phoεphorylation by 50% at 1 microgram/ml and by 70% at 20 microgramε/ml . DHA preεent by itεelf at 20 microgramε/ml decreaεed the extent of phoεphorylation by only 5%. It can be appreciated, in accordance with the preεent invention, that similar cellular as well as in vi tro tyrosine kinaεe assays can be used to monitor activity and to test compounds for their abilities to inhibit εuch activity.
Table 3
Effect of DHA-STKSI on the Autophoεphorylation of EGF Receptor
IV. Utility
Fatty acyl-peptide compositions of the invention are useful in inhibiting uncontrolled proliferation of cellε, such aε benign and malignant tumor cellε, virally-infected cellε, pεoriatic cells, and the like. The use of the composition of the invention for inhibition of such cell proliferation is illustrated by experiments summarized in Table 1. Generally, peptides are selected for use in forming compoεitionε of the invention, baεed on their known or experimentally determined activities in inhibiting cell proliferation. Selected peptides are then used to form polyunsaturated fatty acyl-peptide compositionε, according to the general methodε described in Section II, above and detailed in Exampleε 1-7. Aε illuεtrated herein, polyunsaturated fatty acyl-peptide compositions of the preεent invention are effective to produce a εeveral-fold enhancement of anti-proliferative activity, in compariεon to peptides alone.
More specifically, fatty acyl-GnRH and -GnRH analogues will find use in treating androgen-dependent prostate adenocarcinomaε. Teεt GnRH compoundε can be screened in an LH releaεe asεay, aε described in Example 9, then tested in an experimental animal model, such as a rat bearing the Dunning R-3327-H prostate adenocarcinoma. Additionally, GnRH derivativeε of the invention are expected to find usefulneεε in treatment of eεtrogen-dependent mammary tumors, and their efficacy can.be meaεured in rats (εuch aε
Wiεtar Furth ratε) carrying a mammary tumor εuch aε the MT/W9A mammary tumor, according to methods known in the art. Likewise, it is anticipated that such LHRH analog compositionε will inhibit growth of certain pituitary tumorε, chondroεarcomaε, and oεteoεarcomaε (Schally) .
An anti-neoplaεtic or anti-tumor treatment method, aε described herein, includeε expoεing target neoplaεtic cellε to a concentration of fatty acyl-peptide compound effective to inhibit neoplaεtic cell proliferation at leaεt about 50%, and preferably about 90%. Such effective concentrationε can be determined in an in vi tro aεεay, aε deεcribed in Section IV, above.
Further, it can be appreciated from the foregoing discuεεion that the method of the invention haε general utility in enhancing biological activity of a biologically active peptide, by attaching to the peptide a polyunεaturated fatty acyl moiety.
The following exampleε illustrate, but in no way are intended to limit, the present invention.
Example 1 Synthesis of Peotideε Solid-phase synthesis was carried out according to standard methodε. In one exemplary method, the εynthesiε iε performed uεing a Beckman model 900 peptide εyntheεizer.
Each BOC protected amino acid (2.4 mmol) iε diεsolved in 5 ml dichloromethane (DCM) and cooled to 0 degrees. The volume of dichloromethane used for BOC-leucine is 12 ml, and the solution iε not cooled. 2 ml of 0.6 M N,N- dicyclohexylcarbodiimide (DCCD) in DCM iε added and the mixture is stirred at 0 degrees for 15 minutes. Coupling reactions are monitored at each εtep uεing the ninhydrin aεεay. Coupling reactionε that are incomplete are repeated uεing the appropriate symmetric anhydride. After the coupling reaction any remaining free amino acids are acetylated by using acetylimidazole.
Precipitation of N,N-dicyclohexylurea iε completed by storage at -20 degrees for 1.5 hours, after which the precipitate is filtered and washed with ethyl ether (5 ml) . The filtrate is evaporated to remove solvents, and the product is cryεtallized by precipitation (Meyerε) .
Side chain protecting groupε include for cysteine, 4- Met-Benzyl; for lysine, 2-chlorobenzyloxycarbonyl; for serine, benzyl; for arginine, tosyl; for threonine, benzyl; for aεpartate, benzyl; and for tyrosine, 2-bromobenzyl- oxycarbonyl. After peptide εyntheεiε productε are deprotected in liquid hydrofluoric acid. For example, a mixture of protected peptide reεin (1.32 g) , 2- mercaptopyridine (0.5 g) , p-creεol (2.6 g) , and liquid hydrogen fluoride (HF) (25 ml) is stirred at 0 degrees with a rapid stream of nitrogen gaε, first below 0 degreeε, then at 24 degreeε. The mixture iε εtirred in ethyl acetate (25 ml) until a finely divided εolid iε obtained. The εolid iε filtered, washed with ethyl acetate, and air dried. The solid is stirred in 50% aqueous acetic acid (10 ml) to disεolve the peptide, filtered and waεhed with 20 ml water.
The filtrate iε freeze dried.
Example 2 Preparation of DHA-STKS (DHA-SEQ ID NO: 1) A. Preparation of DHA-O-N-Succ Ester
0.340 mM DHA (112.5 mg) (Aldrich Chemical Co., catalog No. :27, 155-1) and 0.341 mM TBTU (113.5 mg) were εtirred in 4 ml of dry dimethylformamide (DMF) in the preεence of 0.1 mM (15 mg) of diiεopropylethylamine (DIEA) . Activation waε carried out under He atmoεphere for under an hour. 0.341 mM (39.6 mg) of N-hydroxyεuccinnimide (HO-N-Succ) and 0.341 mM of DIEA were added to the reaction mixture. The mixture was εtirred overnight under He atmoεphere for the completion of the transeεterification reaction. This reaction mixture can be used directly for the N-terminal acylation of the required peptide or worked up for the
preparation of pure DHA-O-N-Succ . The reaction mixture was diluted to 30 ml with water and extracted with 3 x 10 ml of peroxide-free ether (treated and stored over alumina) . The combined ether phase was back-extracted with 3 x 5 ml of water. The ether solution waε dried over anhydrouε εodium εulphate, evaporated to dryneεs and taken up in 4 ml of acetone.
B. Preparation of the N-DHA-derivative of STKS 0.03 mM STKS (37.0 mg) was first suεpended in a water-acetone mixture (1:1), before DIEA (0.15 mM, 18.0 mg) was added. 0.05 mM of DHA-O-N-Succ ester in 1 ml of ace¬ tone was added to the suspension and the ratio of water-acetone was adjusted to 1:1. The reaction mixture waε εtirred overnight under a He atmosphere, and then eva¬ porated to dryness. The dri'ed residue was triturated with 3 x 5 ml of the following solventε: petroleum ether, diethyl ether, and HPLC grade water.
Uεing thiε procedure, a yield of 80% and at leaεt a 90.0% product purity can be achieved, according to HPLC analyεiε. A Hewlett-Packard HP-1089 liquid chromatograph equipped with diode array detector waε uεed with a Zorbax SB-300-C18 column with the dimenεionε 4.6 x 150 mm. The product 'ε abεorbance at 210 nm waε monitored. The εolvent composition was used with a gradient from 0 to 100% B, where buffer A consisted of 0.05% TFA in water (v/v); and buffer B consisted of 0.05% TFA in acetonitrile (v/v) . The product eluted at about 64% buffer B. The chemical characterization of the compound was carried out by masε spectrometry (MS) .
MH+ theoretical: 1533.3, MH+ measured: 1533.2
Example 3 Preparation of DHA-STKSI (DHA-SEQ ID NO. 2)
A procedure similar to that described in Example 2 was used to generate the activated ester group of DHA (DHA-O-N- εuccinimide) . Then, 0.03 mM of STKSI (36.2 mg) was acylated with 0.05 mM of DHA-O-N-Succ ester in the presence of 0.15 mM of DIEA according to Example 2. The product waε eluted from the same HPLC gradient aε described in Example 2, the product eluted at 64.5% B. The final yield was 82%. The chemical characterization of the compound waε carried out by mass εpectrometry. MH+theoretical: 1517.4, MH+meaεured:1515.5.
Example 4 Preparation of DHA-SPKCS (DHA-SEQ ID NO: 3) SPKC-(Lyε/epεilon-TFA/)2,9 (54.8 mg, 0.04 mM, final concentration) waε εuεpended in 3 ml of water-acetone (1:1) containing 0.08 mM DIEA. 'Crude DHA-O-N- Succ activated ester in DMF solution (0.04mM), aε deεcribed in Example 2, waε added, and the suspension was stirred overnight under a He atmoεphere. The reaction mixture was evaporated to dryness and the product was TFA-deprotected in the following fashion. TFA-deprotection and purification of the end product waε performed in the following faεhion. The dry reεidue waε dissolved in 3 ml of 1 M aqueous piperidine and εtirred at room temperature. Deprotection was complete within 135 min. The reaction mixture was neutralized with acetic acid, evaporated to dryneεε and triturated with
3 x 5 ml of petroleum ether. The reεt of the solvent waε evaporated, the residue waε taken up in 4 ml of water-glacial acetic acid (1:1) and purified by preparative HPLC. Preparative HPLC conditions were aε follows. An
Aquapore RP-300 column (Source) 2 x 22 cm was uεed with a buffer gradient of 0-100% buffer B. Buffer A was 0.05% TFA in water; buffer B was 0.05% TFA in acetonitrile. The flow rate was 10 ml/min and the eluent program used was the following: t(min) A% B%
0
10
25
The product was eluted at 11.5 minutes, the yield was 65%, the purity of the product was 97%. The product was analyzed by masε spectrometry, with the following resultε: MH+theoretical:1700.8; MH+measured: 1700.8
Example 5
Preparation of DHA-Somatoεtatin Analogue
(DHA-SEQ ID NO: 4) 360 ul each of 0.5 mM εolutionε of DHA, N,N'-diiεo- propyl carbodiimide (DIC) , and pentafluorophenol in dimethylformamide (DMF) were mixed and kept at 25. oC. After 15 min 54 mg (45 umol) of H-D-Phe-Cyε-Tyr-D-Trp- -Lyε(Tfa)-Val-Cyε-Thr-NH2 x HCl (diεεolved in 1 ml of DMF) were added to thiε εolution and-the pH of the reaction mixture waε adjusted to 8 with triethylamine and kept at room temperature overnight. The DMF was evaporated in vacuo , and the oily residue waε dissolved in 1 ml of ethanol. To the solution waε added 2 ml of 2 M hydrazine hydrate in ethanol. and the reaction mixture waε stirred at
45 'C for 48 hours.
The solvent waε removed in vacuo and the oily reεidue waε TFA-deprotected by diεεolving the reεidue in 3 ml of 1 M aqueouε piperidine and εtirring at room temperature, and dissolved in 28 ml of a 2-propanol:acetic acid:water
(30:35:35) solvent mixture and purified by HPLC under the following conditions: A reversed-phaεe chromatography column waε uεed (Prepex C-18, 25-40 urn, 42 x 1.4 cm); the buffer waε a 2-propanol :acetic acid:water mixture (30:35:35) and εeparation was followed by TLC and HPLC.
THe purest fractions were pooled and repurified by MPLC . The elution procedure was the following:
Step 1: Iεocratic elution with 50 ml of 2-propa- nol-acetic acid-water (20:40:40) .
Step 2: Gradient elution with 400 ml of elution mixture applied in εtep 1 and 400 ml of 2-propanolacetic acid-water (35:32.5:32.5) .
The purity of the fractionε was checked by HPLC, the purest fractionε were pooled, evaporated, lyophilized and yielded 57% product. The purity of the product waε 92%. The composition of the product was analyzed by masε εpectrometry.
MH+theoretical:1356.5. MH+meaεured:1356.7
Example 6 Preparation of DHA-D-Lvs6-GnRH Analogue (SEQ ID NO: 5)
360 ul each of 0.2 mM 'solutions of DHA, DIC, and pentafluorophenol in DMF were mixed and kept at room temperature. After 15 minutes 56 mg (0.05 mM) of Glp-His-Trp-Ser-Tyr-D-Lyε-Leu-Arg-Pro-Gly-NH2 x HCl (D-Lyε6-GnRH) (diεεolved in 1 ml of DMF) were added to thiε εolution and the pH of the reaction mixture waε adjuεted to 8 with triethylamine and kept at" room temperature overnight. The product waε TFA-deprotected aε deεcribed in Example 4. The εolvent waε removed in vacuo and the oily residue waε dissolved in 25 ml of 2-propanol-acetic acid-water (30:35:35) solvent mixture and purified by MPLC as described in Example 5. The final product yield waε 47%. The εample identity waε confirmed by mass εpectrometry. MH+theoretical: 1564.9,
MH+meaεured:1565.0
Example 7 Svntheεis of DHA-T Cell Epitope Peptide (DHA-SEQ ID NO: 12)
T-cell epitope peptide (TCEP) (132 mg, 0.075 mmole) , with TFA and formyl protecting groups, was diεεolved in DMSO (2 ml) containing DIEA (40 μl, 0.30 mmole) and DHA εuccinyl ester (0.075 mmole). The reaction mixture was stirred for 3 hours at room temperature at which time acetonitrile/water (1:1, v/v), 0.5 ml) and TFA (0.5 ml) were added. The reaction mixture was then filtered through a 0.2 μm filter and this solution was applied directly to a preparative reverεed-phaεe HPLC column for purification. DHA-TCEP was collected and identified by masε εpectroεcopy.
To remove the TFA and formyl protecting groups the purified peptide was treated in 1 M aqueous piperidine for 8 hourε at room temperature at which time the product waε iεolated by lyophilization. The product waε identified by maεε εpectrometry (MWcaιc = 1886.7, MWfound = 1888.5) .
Example 8 Aεεav of the Inhibition of Cell Proliferation The growth inhibitory effect of peptide hormone analogueε, εuch aε DHA-STKS, DHA-STKCS and D-Lyεδ(DHA)-
GnRH, was evaluated on the basis of changes in the total cell number. Cells were cultivated in RPMI 1640 medium (GIBCO BRL, Eggenstein, Germany, Cat. No. : 074-01800) εupplemented with 10% fetal calf εerum. Tumor cellε were waεhed with phoεphate-buffered εaline, then 0.25% trypεin waε added to detach them. The cellε were εuspended in RPMI 1640 medium and after resuεpending the cellε were εtained with trypan blue and counted in a Burker chamber. 0.1 ml cell εuεpension and 0.1 ml 0.1% trypan blue were mixed. The viability of tumor cells was estimated on the basis of trypan blue (0.4%) exclusion using a hematocytometer.
Example 9 Luteinizing Hormone (LH) Release Aεεay Anterior pituitarieε were obtained from adult (male or female) Wiεtar εtrain rats according to the method
previously deεcribed (Shaw) . The pituitarieε were cut into εmall pieceε, incubated with collagenaεe then diεperεed mechanically to εingle cellε. The cell-εuεpenεion waε mixed with Sephadex G-10 beadε aε εupport material and tranεferred into a εuperfusion chamber. The cellε were continuouεly perfuεed with oxygenated medium or with a medium containing the peptide to be teεted, εuch aε D- Lys6 (DHA) -GnRH. The LH content from each 1 ml fraction of the superfusate waε meaεured by radioimmunoaεsay (RIA) using rat an LH RIA kit or aε described in Example 9 using radiolabeled LH and anti-luteinizing hormone antibodies. The biological potency of a given analogue was determined based on the pituitary hormone responses (peaks) to the peptide stimulation over the baseline secretion.
Example 10 Asεay for Growth Hormone (GH) The releaεe of GH waε meaεured during variouε DHA- peptide hormone or peptide analogue treatmentε, εuch as somatotsatin analogue treatments, on rat hypophysis by using the εuperfuεion method (Myers) . Following is a description of the method. The hypophyses are cut into small pieceε, incubated with collagenaεe, and diεperεed to single cellε. The cells in a εuperfuεion chamber are continuouεly perfuεed with oxygenated medium or with medium containing the peptide to be teεted.
The levelε of GH are determined by a double-antibody radioimmunoaεεay for the hormone. The following methodology can be uεed. Several commercial εourceε now εupply reagent grade hGH of εufficient purity to be reliably uεed in the assay. Antibody is produced by dissolving an appropriate amount of growth hormone (2-3 mg) in 200 microliters 0.01 N sodium hydroxide. The solution is diluted to 1 mg/ml with protein free PBS and mixed 1:1 with Freund'ε adjuvant. 0.5-1 mg growth hormone iε in¬ jected into three εubcutaneouε εites of a young guinea pig,
and repeated at two- to three-week intervalε. The anti-hGH εerum iε harveεted from the guinea pigε and teεted for itε immunopotency. Antisera which will allow for a εenεitive aεεay uεually will precipitate 50% of the labelled hGH at a final dilution of 1:50,000 uεing teεt radiolabelled hGH. Second antibodies (goat anti-guinea pig antibodies) are prepared in a εimilar faεhion. The following radioiodination procedure for hGH is used. One ampoule containing hGH in 20 microliters iε thawed and placed on ice. 1-125 is usually obtained from New England
Nuclear (Boston, MA) . High εpecific activity exceeding 200 mCi/ml iε necessary for the highest degree of sensitivity in the assay. 0.5 M phosphate buffer (pH 7.6) is added to the ampoule containing hGH. About 1 mCi of the radioactive iodine is added to the ampoule.
To start the iodinatiόn reaction, 35 micrograms of a freshly prepared εolution of chloramine-T in 25 microliterε 0.05 M phoεphate buffer (pH 7.6) iε added. The iodine, hor¬ mone, and oxidant are gently agitated for 15 seconds. To stop the reaction, 125 micrograms of a freshly prepared solution of sodium metabiεulfate in 100 microliterε of 0.05 M phoεphate (pH 7.6) iε added. The entire contentε of the ampoule are then placed on Sephadex G-50 column and the labeled hormone εeparated from the iodine. The hormone is diluted in 1% BSA-PBS and stored in small aliquotε at
-20*C.
The hGH radioimmunoaεεay iε performed in the following manner. The εtandard'ε concentrations in hGH assayε are the following: 5, 2.5, 1.25, 0.625, 0.313, 0.156, and 0.078 ng/ml. 50 ml of the unknown growth hormone concentration iε added to tubeε not containing the standard. The next step is to add the guinea pig anti-hGH serum (firεt anti¬ body) . The radiolabeled hGH iε added to every tube. The aεεay tubeε are εhaken gently, left at room temperature for two hourε, and then placed in a refrigerator at 4'C for at leaεt three dayε. After the three-day incubation, the
tubes are removed from the refrigerator and the appropriate dilution of the goat anti-guinea pig serum added (second antibody) . The second antibody is diluted in 1% BSA PBS buffer to obtain approximately 50% precipitation of antibody-bound labeled hGH. Aliquotε of 100 microliterε of the εecond antibody are added to every tube. The tubeε are gently agitated, left at room temperature for about two hourε, and then placed at 4'C. for at least eight hours. Following incubation with the second antibody, the tubes are centrifuged at 2000 g for 30 minutes at 4 degrees in a refrigerated IEC-PR6 centrifuge. The εupernatantε are aεpirated by vacuum suction, with care taken not to disturb the precipitates packed in the bottom of tubeε. The pellet iε resuspended in 2 ml cold PBS and recentrifuged as described above. Washing the precipitates with buffer increaseε the reproducibility.
Example 11 Measurement of Autophosphorylation of the EGF Receptor Autophosphorylation of the EGF receptor waε meaεured according to Bellot et al . (Bellot) . Cells were washed once with binding buffer. The plates were then placed in a water bath at 37 degrees and different concentrations of the analogues were added to the cells for εpecific periodε of time. The buffer waε removed and cells were scraped off the plates with 0.5 ml of Laemmli's εample buffer, boiled for 5 minutes, and εonicated for 10 seconds. Aliquotε of each εample were run on two different SDS-polyacrylamide gelε (7%) and each gel was transferred to nitrocellulose paper. One was immunoblotted with RK2, an anti-EGF-R antibody, and the other with an antiphosphotyroεine- εpecific polyclonal antibody. Blots were then labeled with radioiodinated Protein A (New England Nuclear) and autoradiograms of the nitrocellulose papers were made on Kodak X-Omat paper. The activity of the analogueε was characterized on the basiε of their inhibitory effect on
the phoεphotyroεine content in compariεon to that of untreated cellε uεed aε control. Alternatively, the tyroεine kinaεe activity was determined according to the following method (Keri) . The cellε were incubated with the εubεtrate analogueε, εuch as DHA-SPRCS or DHA-STRS for 24 hourε, then harveεted and homogenized. The reaction volume of 100 microliterε, and homogenized in a Dounce homogenizer 30 timeε in 5 vol buffer (50 mM Triε-HCl, pH 7.8, 50 mM MgCl2, 10 icromolar εodiumvanadate, 1 mM EDTA, and 50 microgram/ml aprotinin. The reaction volume of 100 ml contained 50 mM Tris-HCl, pH 7.8, 50 mM MgCl2, 10 micromolar sodium vanadate, 0.1% nonidet P-40, 5 micromole gamma 32p-ATP, ImM substrate and 60 microliter homogenate. The asεay waε initiated by addition of the ATP. After incubation the reaction waε stopped by addition of trichloroacetic acid, and the supernatant was spotted on a 2 x 2 cm phoεphocelluloεe paper (Whatman P-81) . The paper εquareε were waεhed with phoεphoric acid and acetone, and the dried paperε were counted for radioactivity in εcintillation fluid. For each εample an appropriate reaction mixture containing no peptide waε run a control. The activity of the analogues was characterized on the basiε of their inhibitory effect in compariεon to the incorporation of 32P isotope to untreated cells used as control.
Example 12 Preparation of DHA-EGFA (SEP. ID. NO: 6) 0.035 mM of EGFA was εuεpended in 2.5 ml of water- acetone (1:1), then 0.035 mM DIEA (6.5 ul) and 0.035 mM of
DHA-O-N-Succ ester (40.2 mg) in 2.0ml DMF were added to the εolution. The acylation waε carried out by εtirring the reaction mixture overnight under He atmoεphere. The εolventε were evaporated and the dry residue waε triturated with 3 x 5 ml of petroleum ether. The εolvent reεidue was
removed and the crude product was purified by preparative HPLC under the following circumstances: Eluent programme:
All the other parameters were identical to those in Example 3.
While the invention has been described with reference to specific methods and embodiments, it will be appreciated that various modifications and changes may be made without departing from the invention.
Claims
1. A fatty acyl-peptide compoεition effective to inhibit cell proliferation compriεing a peptide having antiproliferative activity, and a polyunεaturated fatty acyl moiety, which iε conjugated to the peptide through the acyl group of εaid moiety, said compoεition being characterized by a cell proliferative inhibitory activity which is several-fold greater than that of the peptide alone.
2. The fatty acyl-peptide composition of claim 1, wherein the polyunsaturated fatty acyl moiety is a docosahexaenoyl or an eicosapentaenoyl moiety.
3. The fatty acyl-peptide compoεition of claim 2, wherein the polyunεaturated fatty acyl moiety iε selected from the group consisting of cis-4, 7, 10, 13 , 16, 19- docoεahexaenoyl (DHA) and ciε-5, 8, 11, 14, 17-eicosapentaenoyl
(EPA) moietieε.
4. The fatty acyl-peptide compoεition of claim 1, wherein εaid peptide iε a peptide hormone.
5. The fatty acyl-peptide compoεition of claim 4, wherein the peptide hormone iε εelected from the group consisting of εomatoεtatin analogε and GnRH analogε .
6. The fatty acyl-peptide compoεition of claim 4, wherein the peptide hormone iε SEQ ID NO: 4.
7. The fatty acyl-peptide compoεition of claim 4, wherein the peptide hormone iε SEQ ID NO: 5.
8. The fatty acyl-peptide composition of claim 1, wherein εaid peptide iε a protein kinaεe modulatory pep¬ tide.
9. The fatty acyl-peptide 'composition of claim 8, wherein the peptide is selected from the group consisting Of SEQ ID NO: 1, SEQ ID NO: 2, SEQ ID NO: 3 and SEQ ID NO: 6.
10. The fatty acyl-peptide composition of claim 1, wherein the polyunsaturated fatty acyl moiety is conjugated to the peptide through an amide linkage.
11. A method for producing a several-fold enhancement of cell proliferative inhibitory activity in a peptide composition compriεing conjugating the peptide with a polyunsaturated fatty acid to form a fatty acyl-peptide composition composed of the peptide and a fatty acyl moiety conjugated to the pep- tide through the acyl group of said moiety.
12. The method of claim 11, wherein the polyunsaturated fatty acyl moiety iε a docoεahexaenoyl or an eicoεapentaenoyl moiety.
13. The method of claim 12, wherein the polyunεaturated fatty acyl moiety iε εelected from the group conεiεting of ciε-4,7, 10, 13, 16, 19-docoεahexaenoyl (DHA) and ciε-5, 8, 11, 14 , 17-eicoεapentaenoyl (EPA) moietieε.
14. The method of claim 11, wherein the polyun¬ saturated fatty acyl moiety iε conjugated to the peptide through an amide linkage.
15. The method of claim 11, wherein εaid peptide iε a peptide hormone.
16. The method of claim 15, wherein the peptide hor¬ mone iε εelected from the group consisting of somatostatin analogs and GnRH analogs.
17. The method of claim 15, wherein the peptide hor¬ mone is SEQ ID NO: 4.
18. The method of claim 15, wherein the peptide hor- mone iε SEQ ID NO: 5.
19. The method of claim 11, wherein εaid peptide iε a protein kinaεe modulatory peptide.
20. The method of claim 19, wherein the peptide iε selected from the group consisting of SEQ ID NO: 1, SEQ ID NO: 2, SEQ ID NO: 3 and SEQ ID NO: 6.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU55740/94A AU5574094A (en) | 1992-11-30 | 1993-11-29 | Polyunsaturated fatty acyl-peptide composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US98429392A | 1992-11-30 | 1992-11-30 | |
| US07/984,293 | 1992-11-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1994012530A1 true WO1994012530A1 (en) | 1994-06-09 |
Family
ID=25530435
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/HU1993/000065 Ceased WO1994012530A1 (en) | 1992-11-30 | 1993-11-29 | Polyunsaturated fatty acyl-peptide composition |
Country Status (3)
| Country | Link |
|---|---|
| AU (1) | AU5574094A (en) |
| CA (1) | CA2150470A1 (en) |
| WO (1) | WO1994012530A1 (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0747391A3 (en) * | 1995-06-07 | 1997-05-21 | Lilly Co Eli | Preparation of an acylated protein powder |
| WO1997044063A3 (en) * | 1996-05-22 | 1998-02-26 | Neuromedica Inc | Dha-pharmaceutical agent conjugates |
| US5919815A (en) * | 1996-05-22 | 1999-07-06 | Neuromedica, Inc. | Taxane compounds and compositions |
| WO1999044600A1 (en) * | 1998-03-02 | 1999-09-10 | Scotia Holdings Plc | Cancer management with tamoxifen and gammalinolenic acid |
| US5994392A (en) * | 1988-02-26 | 1999-11-30 | Neuromedica, Inc. | Antipsychotic prodrugs comprising an antipsychotic agent coupled to an unsaturated fatty acid |
| US6080877A (en) * | 1996-05-22 | 2000-06-27 | Neuromedica, Inc. | Taxanes |
| WO2000038709A1 (en) * | 1998-12-31 | 2000-07-06 | Chiron Corporation | Treatment of hyperproliferative disorders using casein kinase i |
| WO2001045744A3 (en) * | 1999-12-21 | 2002-05-02 | Ramchand Chaniyilparampu Nanap | Novel compounds of unsaturated fatty acids |
| AU770519B2 (en) * | 1996-05-22 | 2004-02-26 | Luitpold Pharmaceuticals, Inc. | DHA-pharmaceutical agent conjugates |
| US7235583B1 (en) | 1999-03-09 | 2007-06-26 | Luitpold Pharmaceuticals, Inc., | Fatty acid-anticancer conjugates and uses thereof |
| EP1702214A4 (en) * | 2003-12-24 | 2007-12-19 | Wyeth Corp | Methods of treating asthma |
| WO2015153730A3 (en) * | 2014-04-02 | 2016-02-04 | The Regents Of The University Of California | Synthetic catalytic mimics of esterases, lipases or desaturases |
| WO2021157916A1 (en) * | 2020-02-05 | 2021-08-12 | 한양대학교 산학협력단 | Unsaturated fatty acid-conjugated cp2c-targeting peptide-based anticancer agent |
-
1993
- 1993-11-29 CA CA002150470A patent/CA2150470A1/en not_active Abandoned
- 1993-11-29 WO PCT/HU1993/000065 patent/WO1994012530A1/en not_active Ceased
- 1993-11-29 AU AU55740/94A patent/AU5574094A/en not_active Abandoned
Non-Patent Citations (1)
| Title |
|---|
| CHEMICAL ABSTRACTS, Volume 111, No. 15, issued 09 October 1989, (Columbus, Ohio, USA), A.V. KABANOV et al., "Fatty Acid Acylated Antibodies Against Virus Suppress its Reproduction in Cells", page 580, left column, abstract No. 132 137 v; & FEBS LETT., 1989, 250(2), 238-40. * |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5994392A (en) * | 1988-02-26 | 1999-11-30 | Neuromedica, Inc. | Antipsychotic prodrugs comprising an antipsychotic agent coupled to an unsaturated fatty acid |
| EP0747391A3 (en) * | 1995-06-07 | 1997-05-21 | Lilly Co Eli | Preparation of an acylated protein powder |
| AU770519B2 (en) * | 1996-05-22 | 2004-02-26 | Luitpold Pharmaceuticals, Inc. | DHA-pharmaceutical agent conjugates |
| EP1466628A1 (en) * | 1996-05-22 | 2004-10-13 | Luitpold Pharmaceuticals, Inc. | DHA-pharmaceutical agent conjugates |
| US5919815A (en) * | 1996-05-22 | 1999-07-06 | Neuromedica, Inc. | Taxane compounds and compositions |
| US6080877A (en) * | 1996-05-22 | 2000-06-27 | Neuromedica, Inc. | Taxanes |
| US7199151B2 (en) | 1996-05-22 | 2007-04-03 | Luitpold Pharmaceuticals, Inc. | DHA-pharmaceutical agent conjugates of taxanes |
| AU725759B2 (en) * | 1996-05-22 | 2000-10-19 | Luitpold Pharmaceuticals, Inc. | DHA-pharmaceutical agent conjugates |
| WO1997044063A3 (en) * | 1996-05-22 | 1998-02-26 | Neuromedica Inc | Dha-pharmaceutical agent conjugates |
| WO1999044600A1 (en) * | 1998-03-02 | 1999-09-10 | Scotia Holdings Plc | Cancer management with tamoxifen and gammalinolenic acid |
| US6512102B1 (en) | 1998-12-31 | 2003-01-28 | Chiron Corporation | Compositions and methods of diagnosis and treatment using casein kinase I |
| WO2000038709A1 (en) * | 1998-12-31 | 2000-07-06 | Chiron Corporation | Treatment of hyperproliferative disorders using casein kinase i |
| US7235583B1 (en) | 1999-03-09 | 2007-06-26 | Luitpold Pharmaceuticals, Inc., | Fatty acid-anticancer conjugates and uses thereof |
| WO2001045744A3 (en) * | 1999-12-21 | 2002-05-02 | Ramchand Chaniyilparampu Nanap | Novel compounds of unsaturated fatty acids |
| EP1702214A4 (en) * | 2003-12-24 | 2007-12-19 | Wyeth Corp | Methods of treating asthma |
| WO2015153730A3 (en) * | 2014-04-02 | 2016-02-04 | The Regents Of The University Of California | Synthetic catalytic mimics of esterases, lipases or desaturases |
| WO2021157916A1 (en) * | 2020-02-05 | 2021-08-12 | 한양대학교 산학협력단 | Unsaturated fatty acid-conjugated cp2c-targeting peptide-based anticancer agent |
| KR20210099940A (en) * | 2020-02-05 | 2021-08-13 | 한양대학교 산학협력단 | Anticancer Agent Consisted of Unsaturalted Fatty Acid-Conjugated CP2c-Targeting Peptide |
| KR102355987B1 (en) * | 2020-02-05 | 2022-01-26 | 한양대학교 산학협력단 | Anticancer Agent Consisted of Unsaturalted Fatty Acid-Conjugated CP2c-Targeting Peptide |
| CN115361975A (en) * | 2020-02-05 | 2022-11-18 | 汉阳大学校产学协力团 | Anticancer agent based on unsaturated fatty acid conjugated CP2C targeting peptide |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2150470A1 (en) | 1994-06-09 |
| AU5574094A (en) | 1994-06-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2848411B2 (en) | Atrial natriuretic peptide clearance inhibitor | |
| US5084555A (en) | An octapeptide bombesin analog | |
| US5877277A (en) | Octapeptide bombesin analogs | |
| FI104252B (en) | Method for solid phase chemical synthesis of therapeutically useful peptides | |
| US4469679A (en) | Octapeptide vasopressin antagonists | |
| WO1994012530A1 (en) | Polyunsaturated fatty acyl-peptide composition | |
| JPH05505630A (en) | LHRH antagonist | |
| HRP980197A2 (en) | GnRH ANTAGONISTS | |
| EP0797593A2 (en) | Improved cyclic crf antagonists | |
| US5723578A (en) | Peptide analogs of bombesin | |
| CZ299639B6 (en) | Peptidic antiangiogenic medicaments | |
| SK283541B6 (en) | Nonapeptide bombesin antagonists, pharmaceutical composition and use | |
| JP3801867B2 (en) | Novel LHRH antagonists with improved dissolution properties | |
| US4642332A (en) | Effective hormonal peptides: D-3-Pal6 -LHRH | |
| US4656247A (en) | Effective hormonal peptides: D-3-QA1 6-LHRH | |
| Strnadová et al. | Search for lipidized PrRP analogs with strong anorexigenic effect: in vitro and in vivo studies | |
| US5736517A (en) | Treatment of cancer with peptides | |
| RU2248982C9 (en) | Lhrh antagonists, their preparing, pharmaceutical composition and its preparing, method for treatment of tumor and infertility in mammals | |
| US6083915A (en) | Method for treating liver cancer | |
| KR20050086846A (en) | Peptide gap junction modulators | |
| KR100291304B1 (en) | Oxytocin antagonist | |
| EP0728012B1 (en) | NOVEL GnRH ANALOGUES WITH ANTITUMOUR EFFECTS AND PHARMACEUTICAL COMPOSITIONS THEREOF | |
| FI79716C (en) | DAEGGDJUR-PGRF. | |
| US4501733A (en) | Polypeptides, a process for their preparation, their use, and a _process for the purification of polypeptides | |
| US4829051A (en) | N-substituted derivatives of 1-desaminovasopressin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2103567 Country of ref document: CA |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AU CA HU JP |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2150470 Country of ref document: CA |
|
| 122 | Ep: pct application non-entry in european phase |