WO1994004183A1 - Methods for effecting a prolonged reduction in measurable fat content and medicaments for use therein - Google Patents
Methods for effecting a prolonged reduction in measurable fat content and medicaments for use therein Download PDFInfo
- Publication number
- WO1994004183A1 WO1994004183A1 PCT/NZ1993/000071 NZ9300071W WO9404183A1 WO 1994004183 A1 WO1994004183 A1 WO 1994004183A1 NZ 9300071 W NZ9300071 W NZ 9300071W WO 9404183 A1 WO9404183 A1 WO 9404183A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- animal
- fowl
- growth promotant
- growth
- hormone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/27—Growth hormone [GH], i.e. somatotropin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/05—Phenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/365—Lactones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/565—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/57—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/225—Calcitonin gene related peptide
Definitions
- This invention relates to a method of manipulating body composition and to medicaments suitable for use in such a method.
- the Gardner and Flint paper therefore contains no teaching of raising the levels of growth hormone within an animal early in life to effect a long-term reduction in carcase fat content. It is the object of this invention to provide a method for achieving a prolonged reduction in measurable carcase fat content well beyond the period of hormone treatment or at least to offer the public a useful choice.
- the present invention can be said to consist in a method for effecting a prolonged reduction in the measurable fat content of an animal or fowl consisting of the step of administering a growth promotant to said animal or fowl prior to normal physiological puberty of said animal or fowl.
- prolonged it is meant an effect which continues beyond the time the administered growth promotant remains active or present within the animal or fowl.
- the invention consists in a process for promoting nett protein synthesis in an animal or fowl through effecting a prolonged reduction in the fat content of said animal or fowl, said method comprising the step of administering an effective amount of a growth promotant to the animal or fowl prior to normal physiological puberty of said animal or fowl.
- the invention consists in the use of a growth promotant in the preparation of a medicament for effecting a prolonged reduction in the fat content of an animal or fowl.
- This invention relates generally to manipulating fat deposition. In particular, it has application to the reduction of measurable fat content in a carcase.
- the invention will be more particularly described below in relation to reducing the measurable fat content in lambs and rats, it will be appreciated that it is not limited thereto. Indeed, due to the similarity in physiological action of growth promotants in all animals and birds, (see for example Breier and Gluckman, Livestock Production Science 27: 77-94 (1991), Barkan et al. Journal of Clinical Endocrinology and Metabolism. 67: 69-73 (1988), and Wrav et al. Proceedings. International Symposium on Growth and Development. University of Auckland (1991)), the invention is equally applicable to the manipulation of fat deposition in other livestock (such as porcines, bovines, cervines and caprines), in humans and in domestic fowl.
- livestock such as porcines, bovines, cervines and caprines
- the invention is generally based upon the applicants' surprising finding that the level of growth promotant within an animal or fowl early in life has the capacity to influence the ability of the animal or fowl to deposit fat on a long term basis. While not wishing to be bound by any particular theory, the applicants believe that at a very early time during the life of an animal or fowl, the level of growth promotant can exert a long-term influence on the number, size and metabolic capacity of adipocytes in the animal or fowl.
- growth promotant any compound or agent which exhibits growth-promoting activity in an animal or fowl through altering the function of the somatotrophic axis.
- the term therefore includes but is not limited to somatotrophic hormones and steroidal growth promotants.
- somatotrophic hormone is meant in its most general sense and covers all growth hormones and growth related factors. Examples of such somatotrophic hormones together with sources therefor are as follows: bovine Growth Hormone (bGH) - Eli Lilly ovine Growth Hormone (oGH) - National Institute of Health, USA (NTH) porcine Growth Hormone (pGH) - NTH human Growth Hormone (hGH) - Genentech
- IGF-I Insulin-like Growth Factor-I
- IGF-II Genentech Insulin-like Growth Factor-II
- oPL Ciba Geigy bovine Placental Lactogen
- bPL Monsanto human Placental Lactogen
- hPL - NIH ovine Prolactin
- oPrl - NIH bovine Prolactin
- bPrl NTH human Prolactin
- pPrl - NIH porcine Prolactin
- steroidal growth promotant this primarily covers oestrogens and androgens but is not limited to these.
- steroidal growth promotants such as oestrogens and androgens act by altering function of the somatotrophic axis (S N McCutcheon (1989), "The use of growth-promoting agents", Chapter 9 in Meat Production and Processing, Occasional Publication No 11, New Zealand Society of Animal Production, Ed. R W Purchas, B W Butler-Hogg, A S Davies; Preston R L (1975), Biological responses to estrogen additives in meat- producing cattle and lambs, Journal of Animal Science. 41 : 1414-1430), although the precise mechanism by which they act is not well understood.
- Examples of steroidal growth promotants are as follows:
- growth promotants in accordance with the present invention are compounds or agents which act on the somatotrophic axis to indirectly promote growth. Such compounds include those which upon administration to the animal or fowl cause the animal or fowl to itself produce an increased amount of somatotrophic hormone.
- GRF Growth Hormone Releasing Factor
- such compounds can be those which increase the concentration of active somatotrophic hormone by at least partially inactivating an inhibitory mechanism regulating the secretion or activity of the somatotrophic hormone within the animal or fowl.
- the somatotrophic hormone is somatotropin
- the animal or fowl can be passively or actively immunised against somatostatin to prevent or reduce the inhibitory effect of somatostatin on somatotropin.
- immunisation is performed in accordance with protocols well understood by the skilled worker.
- Still further examples of compounds which act on the somatotrophic axis to indirectly promote growth are those which increase the effective duration (i.e. the physiologically active life) of the somatotrophic hormone.
- Such compounds include antibodies which prolong the action of various somatotrophic hormones and which can be prepared by procedures well understood in the art.
- the somatotrophic hormones are preferred.
- a critical step of the applicants' method is increasing the active concentration or effective duration of a growth promotant as above within the animal or fowl in question. Most conveniently, this increase is achieved through direct administration of the growth promotant to the animal or fowl.
- the growth promotant is a somatotrophic hormone the hormone can be administered parenterally to the subject animal or fowl with or without an appropriate carrier.
- the timing of the administration of growth promotant is also critical to the invention. More particularly, in order to achieve a prolonged reduction in measurable fat content in the animal or fowl, the step of administering the growth promotant must be performed prior to normal physiological puberty of the animal or fowl. Obviously, this will vary depending upon the identity of the animal or fowl in question. For example, in sheep, normal physiological puberty occurs at approximately six months of age. Furthermore, while the administration of the growth promotant will usually be performed subsequent to the birth of the animal or fowl, this is not essential. Indeed, this step can be performed at any time during the last third of the gestation period up to the onset of normal physiological puberty.
- the step of administering the growth promotant will be performed in relation to the neonate. This will mean that the step is performed during the first six months following birth, more preferably within the first three months following birth.
- variables such as the amount of the growth promotant administered and the period of administration will have an influence on the results achieved. Accordingly, while in no way intending to exclude administration of low doses of the growth promotant, or to exclude a single administration step, it is preferred that multiple steps be performed during which relatively large dosages of growth promotants are administered.
- the dosage rate will be in the range of 0.1 to 0.5 mg/kg bodyweight/day over the administration period.
- the growth promotants used in the present method are naturally present in the animal or fowl, and therefore that administration of further amounts of the growth promotant will merely effect an increase in the normal circulating level of that growth promotant within the animal or fowl. This is particularly the case where, as is preferred, the growth promotant is a somatotrophic hormone.
- the active compound will be administered at regular intervals and in an amount sufficient to increase the active concentration of somatotrophic hormone within the animal or fowl by at least two and more preferably by three to four times.
- the variables of administration necessary to achieve this will be readily determinable by the worker skilled in the art.
- the active compound to be administered is bGH
- the subject is a neonate lamb and the treatment protocol requires weekly administration for a period of from 8 to 14 weeks
- the bGH will be administered at a rate of between 0.1 and 0.5 mg/kg per day, preferably between 0.1 and 0.3 mg/kg per day.
- the compound necessary to achieve the desired increase in concentration or effective duration of somatotrophic hormone within the animal or fowl can be administered by any conventional technique, with the technique selected being dependent upon the compound concerned. However, it remains both usual and preferable for the compound to be administered parenterally.
- parenterally it is meant that the compound is administered by intravenous, intramuscular, intradermal or subcutaneous injection. Most conveniently, the administration is by subcutaneous injection in the neck.
- the invention provides medicaments for use in the above method.
- medicaments will include as their essential component a unit dosage amount of the growth promotant, and can take a number of forms.
- a first and preferred form of medicament comprises an effective amount of a somatotrophic hormone, optionally with a pharmaceutically or veterinarily acceptable carrier or diluent.
- the somatotrophic hormone of the medicament may be any of those somatotrophic hormones described above. It is however preferred that the somatotrophic hormone be either bovine somatotropin or ovine prolactin.
- a second form of the medicament comprises an effective amount of a steroidal growth promotant, optionally with a pharmaceutically or veterinarily acceptable carrier or diluent.
- the steroidal growth promotant may be any of those steroidal growth promotants described above. It is however preferred that the steroidal growth promotant be an oestrogen or androgen, or an oestrogen or androgen analogue.
- a third form of the medicament comprises an effective amount of a compound such as GRF which upon administration to the animal or fowl, causes the animal or fowl to increase its own production of somatotrophic hormone, or which increases the active concentration of somatotrophic hormone.
- the medicament may include a pharmaceutically or veterinarily acceptable carrier.
- the dosage rate of somatotrophic hormone or other compound contained within the medicament as active agent will depend upon variables such as the compound concerned, the desired concentration to be achieved, the treatment protocol and the type and weight of animal or fowl to which the medicament is to be administered. The effect of such variables in determining an appropriate dosage rate will be readily calculable by the ordinary skilled worker in this field.
- a carrier or diluent is desired for inclusion in the medicament, it can be any of those carriers or diluents known in the art.
- examples of such carriers included saponins, glycerol, sorbitol and propylene glycol. It is however particularly preferred that the carrier be one which regulates the rate of release of the active agent.
- the controlled release formulation minus bST Treatment with bST or excipient commenced on the day of birth, the first dose being adjusted in proportion to the number of days between the day of birth and the next Wednesday. Thereafter, animals were treated at 0800 h on each Wednesday. They were weighed immediately prior to injection and doses adjusted to the new liveweight.
- Eight lambs per treatment group were blood-sampled to determine circulating somatotropin levels. Sampling commenced two days before the week 4 injection and continued for 12 days (i.e. until three days after the week 5 injection). Blood samples were taken at 0800 h daily by jugular vempuncture using evacuated blood collection tubes containing 0.1 ml of 15% w/v EDTA as the anticoagulant (Vacutainer, Becton- Dickinson, Rutherford, New Jersey, USA). Blood was stored on ice and within one hour was centrifuged at 3000 g and 4°C for 20 minutes. Plasma was aspirated into duplicate vials and stored at -20°C for subsequent analysis.
- Somatotropin concentrations in plasma were determined using the double-antibody bovine somatotropin radioimmunoassay described by Flux, Mackenzie and Wilson Animal Production. 38: 377-384 (1984).
- Bovine somatotropin was used for iodination (USDA-bGH-Il, 3.3 LU. per mg) and reference standards (USDA-bGH-Bl, 1.9 LU. per mg).
- Intra- and inter- assay coefficients of variation were 8.9% and 14.1% respectively.
- Pituitary derived ovine and bovine somatotropin exhibit greater than 90% cross-reactivity in this assay. Results are shown in Table 1 and Figure 1.
- Fat 1 fat depth at 8 months of age
- Fat 2 fat depth at 13 months of age
- WT 1 weight at 8 months of age
- WT 2 weight at 13 months of age
- GH (ng/ml) mean blood growth hormone level for sub-group of animals sampled by jugular vempuncture
- 1 Least squares means from models including only significant terms, viz: Subcutaneous abdominal - body weight, Utter (cage), sex, treatment Subcutaneous scapular - treatment Genital - bodyweight, cage (litter), treatment Perirenal - litter (cage), sex, treatment ab P ⁇ 0.05
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Endocrinology (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
This invention relates to a method for effecting a prolonged reduction in the measurable fat content of an animal or fowl carcase. The critical step of the method involves the administration of a growth promotant (such as bovine somatotropin) to the animal or fowl prior to normal physiological puberty of the animal or fowl and usually during the first six month period following birth of the animal or fowl. The method provides carcases which have a reduced fat content but which have not been treated with a hormonal growth promotant immediately prior to slaughter.
Description
METHODS FOR EFFECTING A PROLONGED REDUCTION IN MEASURABLE FAT CONTENT AND MEDICAMENTS FOR USE THEREIN
FIELD OF THE INVENTION This invention relates to a method of manipulating body composition and to medicaments suitable for use in such a method.
BACKGROUND OF THE INVENTION
In recent years, there has been an increased emphasis on nutrition and health. At the forefront of this there has been a focus on the body fat content of the individual and the role of fat in coronary disease and a corresponding impetus to reduce the individual's intake of fat, particularly animal fat.
To date there has been no effective programme for achieving a general reduction in the fat content of the carcase of an animal intended for consumption other than by hormonal treatment immediately prior to slaughter. While it has been shown that exogenous bovine growth hormone (bGH) (also known as bovine somatotropin (bST)) administered to the growing or adult animal can decrease carcase fat content and increase protein content (Muir et al. Journal of Animal Science. 56: 1315-1323 (1983), Zainur et al. Australian Journal of Agricultural Research. 40: 195-206 (1989)), these effects are strictly transitory and are the result of lipolytic and anti-lipogenic activity (Boyd et al. Animal Growth Regulation, pp 257-293 (1989)). There is no suggestion in any of these publications that bST or any other somatotrophic hormone can be used to induce a reduction in carcase fat content without continuous administration of the hormone being necessary. A study on the effect on body weight gain of treating rats with antibodies against rat growth hormone is reported by Gardner and Flint (Journal of Endocrinology. 124. 381-386 (1990)). This study inter alia found that while decreasing the level of growth hormone led to increased fat deposition in female rats, a similar effect was not evident in male rats. Further, Gardner and Flint generally characterise the ability of growth hormone to reduce fat deposition as being due to the promotion of lipolysis and to the antagonistic effect on the action of insulin on lipogenesis.
The Gardner and Flint paper therefore contains no teaching of raising the levels of growth hormone within an animal early in life to effect a long-term reduction in carcase fat content. It is the object of this invention to provide a method for achieving a prolonged reduction in measurable carcase fat content well beyond the period of hormone treatment or at least to offer the public a useful choice.
SUBSTITUTE SHEET
SUMMARY OF THE INVENTION
Accordingly, in one aspect the present invention can be said to consist in a method for effecting a prolonged reduction in the measurable fat content of an animal or fowl consisting of the step of administering a growth promotant to said animal or fowl prior to normal physiological puberty of said animal or fowl.
By "prolonged" it is meant an effect which continues beyond the time the administered growth promotant remains active or present within the animal or fowl.
In a further aspect, the invention consists in a process for promoting nett protein synthesis in an animal or fowl through effecting a prolonged reduction in the fat content of said animal or fowl, said method comprising the step of administering an effective amount of a growth promotant to the animal or fowl prior to normal physiological puberty of said animal or fowl.
In yet a further aspect, the invention consists in the use of a growth promotant in the preparation of a medicament for effecting a prolonged reduction in the fat content of an animal or fowl.
SUMMARY OF THE DRAWING
Although the present invention is broadly as defined above, it will be appreciated by those persons skilled in the art that it is not limited thereto but also includes embodiments of which the following description provides examples. In addition, the invention will be described in relation to the accompanying drawing designated Figure 1 which is a graph showing plasma concentrations of immunoreactive somatotropin in treated lambs.
DETAILED DESCRIPTION OF THE INVENTION
This invention relates generally to manipulating fat deposition. In particular, it has application to the reduction of measurable fat content in a carcase.
While the invention will be more particularly described below in relation to reducing the measurable fat content in lambs and rats, it will be appreciated that it is not limited thereto. Indeed, due to the similarity in physiological action of growth promotants in all animals and birds, (see for example Breier and Gluckman, Livestock Production Science 27: 77-94 (1991), Barkan et al. Journal of Clinical Endocrinology and Metabolism. 67: 69-73 (1988), and Wrav et al. Proceedings. International Symposium on Growth and Development. University of Auckland (1991)), the invention is equally applicable to the manipulation of fat deposition in other livestock (such as porcines, bovines, cervines and caprines), in humans and in domestic fowl.
The invention is generally based upon the applicants' surprising finding that the level of growth promotant within an animal or fowl early in life has the capacity to influence the ability of the animal or fowl to deposit fat on a long term basis. While not wishing to be bound by any particular theory, the applicants believe that at a very early time during the life of an animal or fowl, the level of growth promotant can exert a long-term influence on the number, size and metabolic capacity of adipocytes in the animal or fowl.
By "growth promotant", the applicants mean any compound or agent which exhibits growth-promoting activity in an animal or fowl through altering the function of the somatotrophic axis. The term therefore includes but is not limited to somatotrophic hormones and steroidal growth promotants.
As used herein, the term "somatotrophic hormone" is meant in its most general sense and covers all growth hormones and growth related factors. Examples of such somatotrophic hormones together with sources therefor are as follows: bovine Growth Hormone (bGH) - Eli Lilly ovine Growth Hormone (oGH) - National Institute of Health, USA (NTH) porcine Growth Hormone (pGH) - NTH human Growth Hormone (hGH) - Genentech
Insulin-like Growth Factor-I (IGF-I) - Genentech Insulin-like Growth Factor-II (IGF-II) - Genentech ovine Placental Lactogen (oPL) - Ciba Geigy bovine Placental Lactogen (bPL) - Monsanto human Placental Lactogen (hPL) - NIH ovine Prolactin (oPrl) - NIH bovine Prolactin (bPrl) - NTH human Prolactin (hPrl) - NIH porcine Prolactin (pPrl) - NIH
The above list is of course not intended to be exhaustive. Regarding the term "steroidal growth promotant", this primarily covers oestrogens and androgens but is not limited to these. There is evidence that steroidal growth promotants such as oestrogens and androgens act by altering function of the somatotrophic axis (S N McCutcheon (1989), "The use of growth-promoting agents", Chapter 9 in Meat Production and Processing, Occasional Publication No 11, New Zealand Society of Animal Production, Ed. R W Purchas, B W Butler-Hogg, A S Davies; Preston R L (1975), Biological responses to estrogen additives in meat-
producing cattle and lambs, Journal of Animal Science. 41 : 1414-1430), although the precise mechanism by which they act is not well understood. Examples of steroidal growth promotants are as follows:
Other "growth promotants" in accordance with the present invention are compounds or agents which act on the somatotrophic axis to indirectly promote growth. Such compounds include those which upon administration to the animal or fowl cause the animal or fowl to itself produce an increased amount of somatotrophic hormone. An example of such a compound is Growth Hormone Releasing Factor (GRF), obtainable from American Cyanamid.
Alternatively, such compounds can be those which increase the concentration of active somatotrophic hormone by at least partially inactivating an inhibitory mechanism regulating the secretion or activity of the somatotrophic hormone within the animal or fowl. For example, where the somatotrophic hormone is somatotropin, the animal or fowl can be passively or actively immunised against somatostatin to prevent or reduce the inhibitory effect of somatostatin on somatotropin. Such immunisation is performed in accordance with protocols well understood by the skilled worker.
Still further examples of compounds which act on the somatotrophic axis to indirectly promote growth are those which increase the effective duration (i.e. the physiologically active life) of the somatotrophic hormone. Such compounds include antibodies which prolong the action of various somatotrophic hormones and which can be prepared by procedures well understood in the art.
Of the above growth promotants, the somatotrophic hormones are preferred. A critical step of the applicants' method is increasing the active concentration or effective duration of a growth promotant as above within the animal or fowl in question. Most conveniently, this increase is achieved through direct administration of the growth promotant to the animal or fowl. For example, where as is preferred the growth promotant is a somatotrophic hormone the hormone can be administered parenterally to the subject animal or fowl with or without an appropriate carrier.
The timing of the administration of growth promotant is also critical to the invention. More particularly, in order to achieve a prolonged reduction in measurable fat content in the animal or fowl, the step of administering the growth promotant must be performed prior to normal physiological puberty of the animal or fowl. Obviously, this will vary depending upon the identity of the animal or fowl in question. For example, in sheep, normal physiological puberty occurs at approximately six months of age. Furthermore, while the administration of the growth promotant will usually be performed subsequent to the birth of the animal or fowl, this is not essential. Indeed, this step can be performed at any time during the last third of the gestation period up to the onset of normal physiological puberty.
It is however preferred that the step of administering the growth promotant will be performed in relation to the neonate. This will mean that the step is performed during the first six months following birth, more preferably within the first three months following birth.
It will further be appreciated that variables such as the amount of the growth promotant administered and the period of administration will have an influence on the results achieved. Accordingly, while in no way intending to exclude administration of low doses of the growth promotant, or to exclude a single administration step, it is preferred that multiple steps be performed during which relatively large dosages of growth promotants are administered.
For most animals or fowls, where as is preferred the growth promotant is a somatotrophic hormone, the dosage rate will be in the range of 0.1 to 0.5 mg/kg bodyweight/day over the administration period.
It will of course be appreciated that a number of the growth promotants used in the present method are naturally present in the animal or fowl, and therefore that administration of further amounts of the growth promotant will merely effect an increase in the normal circulating level of that growth promotant within the animal or fowl. This
is particularly the case where, as is preferred, the growth promotant is a somatotrophic hormone.
In the presently preferred embodiment, where the growth promotant is a somatotrophic hormone, the active compound will be administered at regular intervals and in an amount sufficient to increase the active concentration of somatotrophic hormone within the animal or fowl by at least two and more preferably by three to four times. The variables of administration necessary to achieve this will be readily determinable by the worker skilled in the art. For example, where the active compound to be administered is bGH, the subject is a neonate lamb and the treatment protocol requires weekly administration for a period of from 8 to 14 weeks, the bGH will be administered at a rate of between 0.1 and 0.5 mg/kg per day, preferably between 0.1 and 0.3 mg/kg per day.
The compound necessary to achieve the desired increase in concentration or effective duration of somatotrophic hormone within the animal or fowl can be administered by any conventional technique, with the technique selected being dependent upon the compound concerned. However, it remains both usual and preferable for the compound to be administered parenterally. By "parenterally", it is meant that the compound is administered by intravenous, intramuscular, intradermal or subcutaneous injection. Most conveniently, the administration is by subcutaneous injection in the neck.
In its further aspects, the invention provides medicaments for use in the above method. Such medicaments will include as their essential component a unit dosage amount of the growth promotant, and can take a number of forms.
A first and preferred form of medicament comprises an effective amount of a somatotrophic hormone, optionally with a pharmaceutically or veterinarily acceptable carrier or diluent. The somatotrophic hormone of the medicament may be any of those somatotrophic hormones described above. It is however preferred that the somatotrophic hormone be either bovine somatotropin or ovine prolactin.
A second form of the medicament comprises an effective amount of a steroidal growth promotant, optionally with a pharmaceutically or veterinarily acceptable carrier or diluent. The steroidal growth promotant may be any of those steroidal growth promotants described above. It is however preferred that the steroidal growth promotant be an oestrogen or androgen, or an oestrogen or androgen analogue.
A third form of the medicament comprises an effective amount of a compound such as GRF which upon administration to the animal or fowl, causes the animal or fowl to increase its own production of somatotrophic hormone, or which increases the active
concentration of somatotrophic hormone. Again, the medicament may include a pharmaceutically or veterinarily acceptable carrier.
As discussed above, the dosage rate of somatotrophic hormone or other compound contained within the medicament as active agent will depend upon variables such as the compound concerned, the desired concentration to be achieved, the treatment protocol and the type and weight of animal or fowl to which the medicament is to be administered. The effect of such variables in determining an appropriate dosage rate will be readily calculable by the ordinary skilled worker in this field.
Where a carrier or diluent is desired for inclusion in the medicament, it can be any of those carriers or diluents known in the art. Examples of such carriers included saponins, glycerol, sorbitol and propylene glycol. It is however particularly preferred that the carrier be one which regulates the rate of release of the active agent.
The present invention will now be illustrated by the following non-Umiting examples.
EXAMPLE 1
A. Animals and treatments
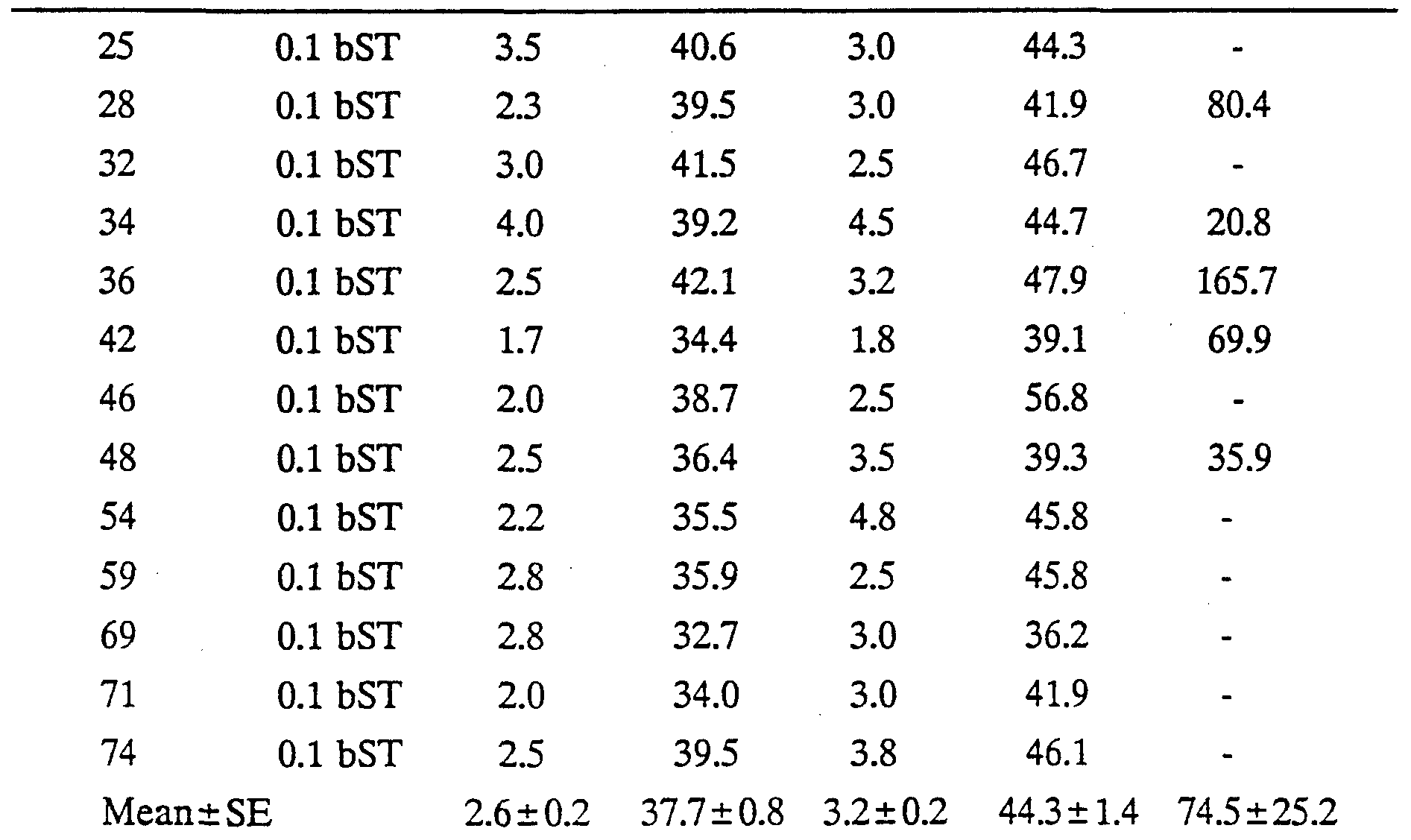
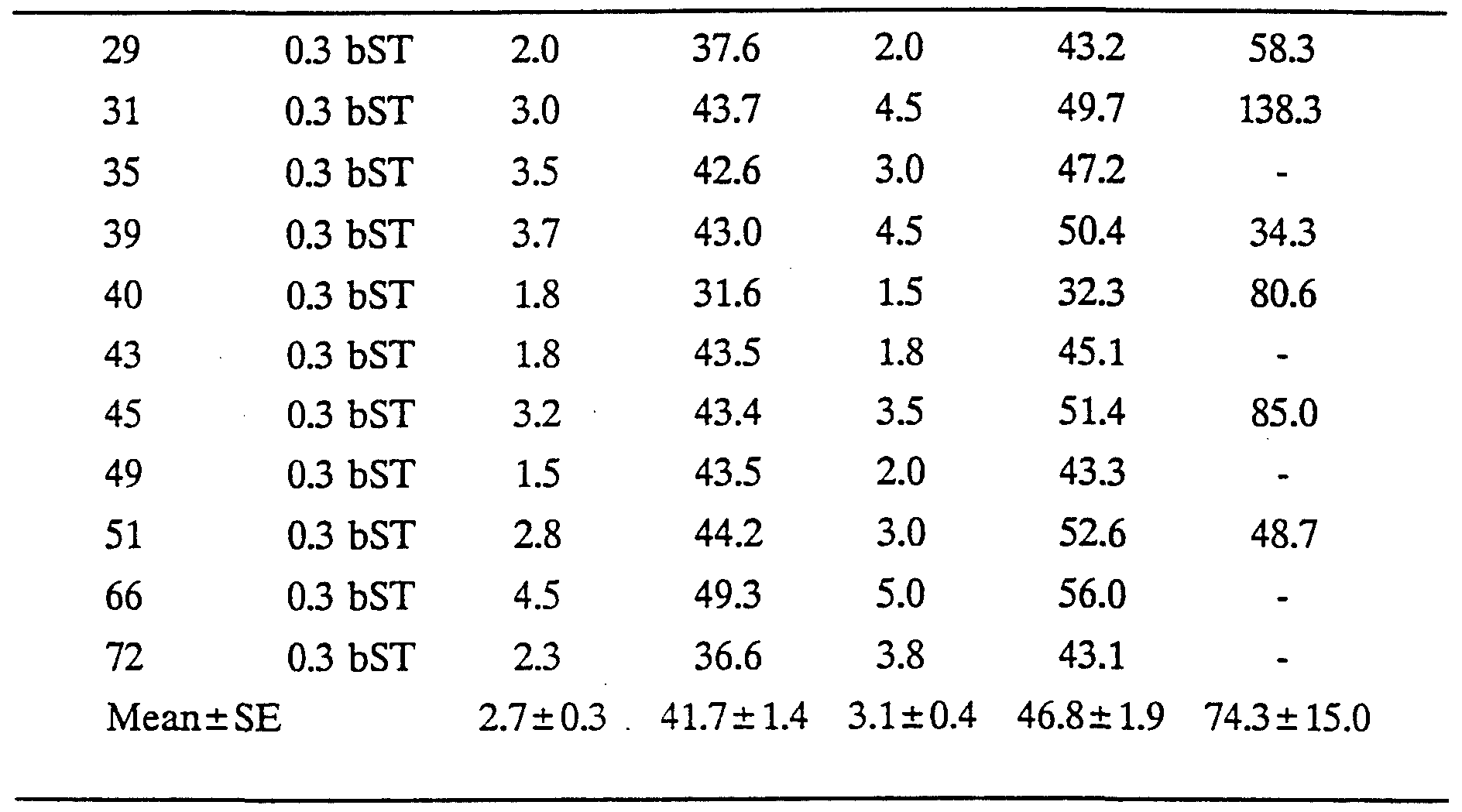
The study involved 48 Romney x (Border Leicester x Romney) lambs which were assigned at random to three groups of 16 lambs balanced for sex, birth rank (singles versus multiples) and date of birth. The first group, "low dose bST', received recombinantly derived bovine somatotropin (bST) at a daily dose equivalent to 0.1 mg per kg liveweight, while the second group, "high dose bST', received bST at a daily dose equivalent to 0.3 mg per kg liveweight. The bST was administered by subcutaneous injection (in the neck) of a slow release formulation (Lilly Research Laboratories, Greenfield, Indiana, USA). The third group, "control", received subcutaneous injections of excipient (i.e. the controlled release formulation minus bST). Treatment with bST or excipient commenced on the day of birth, the first dose being adjusted in proportion to the number of days between the day of birth and the next Wednesday. Thereafter, animals were treated at 0800 h on each Wednesday. They were weighed immediately prior to injection and doses adjusted to the new liveweight.
Weekly treatment with bST or excipient continued until the last injection was given at 11 weeks of age. During this time the lambs were run with their dams on mixed ryegrass/white clover pasture. Lambs were weaned at 12 weeks of age and grazed on pasture until the experiment concluded when they were 13 months old. Male lambs were castrated when they were 10 weeks old and all the animals were shorn at an average 4 months of age.
B. Blood sampling and hormone measurements
Eight lambs per treatment group were blood-sampled to determine circulating somatotropin levels. Sampling commenced two days before the week 4 injection and continued for 12 days (i.e. until three days after the week 5 injection). Blood samples were taken at 0800 h daily by jugular vempuncture using evacuated blood collection tubes containing 0.1 ml of 15% w/v EDTA as the anticoagulant (Vacutainer, Becton- Dickinson, Rutherford, New Jersey, USA). Blood was stored on ice and within one hour was centrifuged at 3000 g and 4°C for 20 minutes. Plasma was aspirated into duplicate vials and stored at -20°C for subsequent analysis. Somatotropin concentrations in plasma were determined using the double-antibody bovine somatotropin radioimmunoassay described by Flux, Mackenzie and Wilson Animal Production. 38: 377-384 (1984). Bovine somatotropin was used for iodination (USDA-bGH-Il, 3.3 LU. per mg) and reference standards (USDA-bGH-Bl, 1.9 LU. per mg). Intra- and inter- assay coefficients of variation were 8.9% and 14.1% respectively. Pituitary derived ovine and bovine somatotropin exhibit greater than 90% cross-reactivity in this assay. Results are shown in Table 1 and Figure 1.
Following covariate adjustment of the raw data given in Table 1, mean (±s.e.) plasma concentrations of immunoreactive somatotropin over the 12 days of blood sampling were 19.2 ±2.1, 65.2 ±2.8 and 74.7 ±4.8 ng/ml in the control, low dose bST and high dose bST groups, respectively. Concentrations were significantly (P<0.01) higher in the bST-treated lambs than in the controls but did not differ significantly between the low and high dose bST groups (P>0.10). There was no apparent peak value of bST concentrations in plasma after injection and the values over the 12-day sampling interval were relatively stable (Figure 1), the interaction between treatment and sampling time being non-significant (P>0.10).
C. Backfat depth
Backfat depth was assessed ultrasonically at average ages of 8 and 13 months using the procedures outlined by Purchas and Beach New Zealand Journal of Experimental Agriculture. 9: 213-220 (19811
Results are shown in Table 1.
TABLE 1
No Treatment Fat 1 T1 Fat 2 WT2 GH
No Treatment Fat 1 WT1 Fat 2 WT2 GH
Key: (a) No = Tag number of lamb
(b) Treatment C = control group (treated with excipient)
0.1 bST = 0.1 mg bST/kg liveweight/day 0.3 bST = 0.3 mg bST/kg liveweight/day
(c) Fat depths (mm)
Fat 1 = fat depth at 8 months of age Fat 2 = fat depth at 13 months of age
(d) Weight (kg)
WT 1 = weight at 8 months of age WT 2 = weight at 13 months of age (e) GH (ng/ml) = mean blood growth hormone level for sub-group of animals sampled by jugular vempuncture
When the data above is covariately adjusted, ultrasonic backfat depth at 8 months of age was significantly (P<0.05) decreased by bST treatment, being 3.4 ±0.3, 2.8 ±0.3 and 2.5 ±0.3 mm in the control, low dose and high dose bST groups, respectively. The difference between the two doses of bST was not significant. However, when the animals were 13 months of age, the effect of bST treatment on backfat depth was no longer as significant (control group, 3.6 ±0.3 mm; low dose, 3.4 ±0.3 mm; high dose 3.1 ±0.4 mm; P>0.10) although still 6-9% lower than the control group.
EXAMPLE 2
Female white rats were mated to males of the same strain and offered an ad libitum complete pelleted diet throughout pregnancy and lactation. At birth, the newborn pups were individually identified and divided at random into five groups balanced, as far as possible, for litter (of origin) and sex. Pups in each group were treated by twice daily (subcutaneous) injection with saline, recombinantly derived bovine somatotropin (0.2 or 0.4 μg/g bodyweight/day) or pituitary-derived ovine prolactin (0.2 or 0.4 μg/g bodyweight/day).
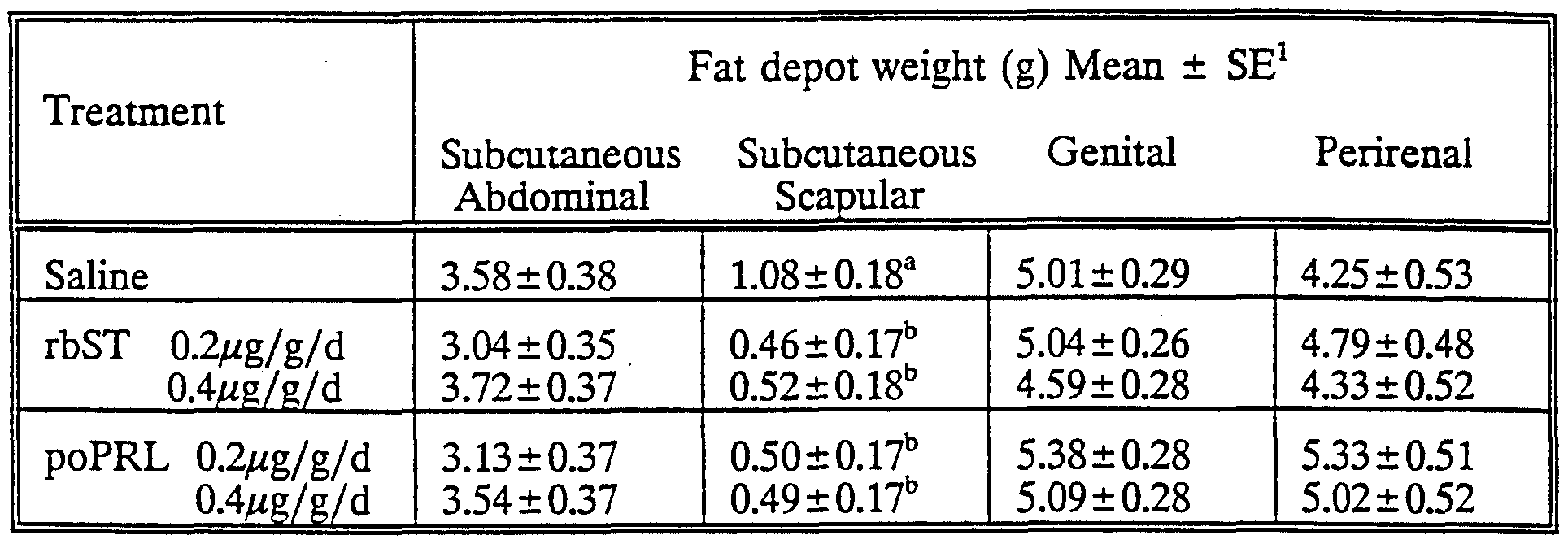
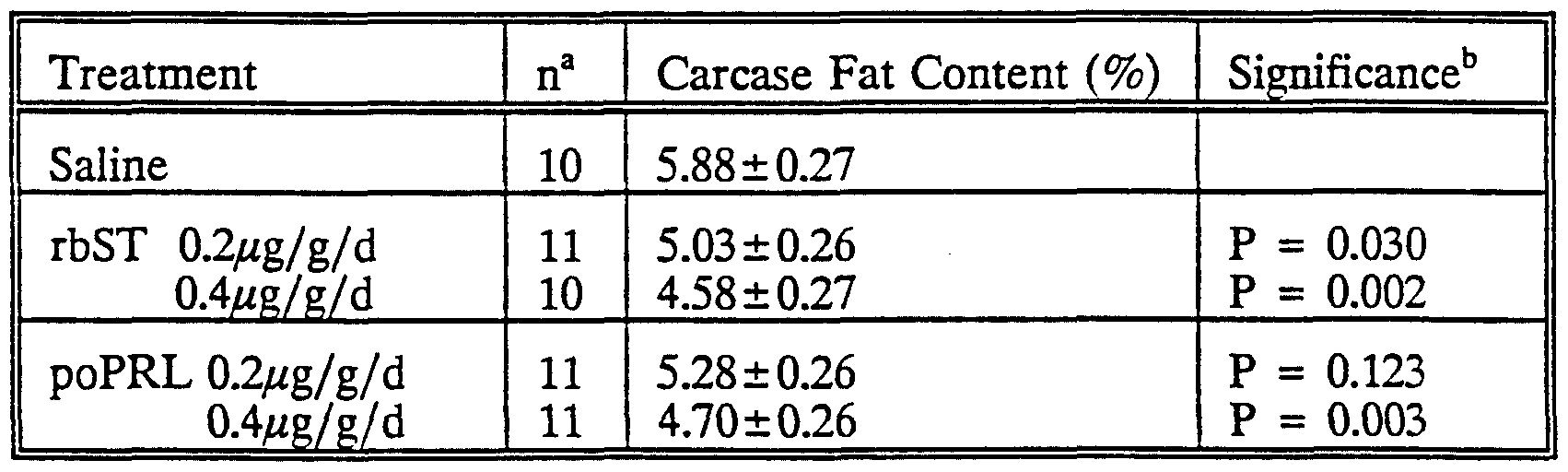
Treatment continued until day 21 of life when the young were weaned from their dams. Thereafter, the young were separated by sex and maintained in rearing cages on an ad libitum complete pelleted diet until 120 days of age. At that age, the experimental animals were slaughtered, the head, feet, tail, skin and visceral contents removed, and four fat depots - subcutaneous abdominal, subcutaneous scapular, genital and perirenal - excised from the carcass and weighed. The remaining carcass was then frozen, sliced, finely ground, mixed thoroughly and a sub-sample taken for the determination of fat content by soxhlet extraction.
Data (fat depot weights or carcass fat content) were subjected to analysis of covariance to test the effects of hormone treatment after adjustment for appropriate covariates or other main effects. Only those covariates or main effects (other than treatment) found to be significant in preliminary analyses were fitted to the final statistical models. Thus the subcutaneous abdominal fat weight was adjusted for bodyweight, litter and sex prior to fitting treatment; the genital fat depot weight was adjusted for bodyweight and litter, the perirenal fat depot weight was adjusted for litter and sex; but the subcutaneous scapular fat depot weight and the carcass fat content were not adjusted for covariates or other main effects as none of these was significant. Comparisons between the four hormone treatments and the saline (control) treatment were made by orthogonal contrasts. Data are expressed as least squares means and standard errors derived from the appropriate statistical models (as described above). The results are given in Tables 2 and 3 below.
TABLE 2: Effect of recombinantlv derived bovine somatotropin (rbST) and pituitary- derived ovine prolactin (poPRL) treatment during days 1 to 21 of life on weights of fat depots at 120 days of age in rats
1 Least squares means from models including only significant terms, viz: Subcutaneous abdominal - body weight, Utter (cage), sex, treatment Subcutaneous scapular - treatment Genital - bodyweight, cage (litter), treatment Perirenal - litter (cage), sex, treatment ab P<0.05
TABLE 3: Effect of recombinantly derived bovine somatotropin (rbST) and pituitary-derived ovine prolactin (poPRL) treatment during days 1 to 21 of life on fat composition (%) of the carcass at 120 days of age in rats.
"Significance of difference from saline group
These results clearly show that the administered growth promotants, at least at a dosage of 0.4μg/g/day, are capable of reducing a substantial, statistically significant reduction in fat content of the treated animals.
INDUSTRIAL APPLICATION
Thus, in accordance with the present invention there is provided a method of effecting a prolonged reduction in measurable fat content in animals or fowls. As such,
the invention will have particular application to reducing fat levels in animals and fowls intended for slaughter and subsequent consumption.
The fact that the growth promotant is administered only in the very early stages of life of the animal or fowl is also highly significant. As will be appreciated, animals or fowls treated in accordance with this method will harbour no residue of the administered growth promotant at the time of slaughter. This means that the current concerns over the administration of growth promotants such as recombinantly produced growth hormones will not apply to the reduced fat animal or fowl carcases produced using the method of this invention. It will be appreciated by those persons skilled in the art that the above description is provided by way of example only and that the invention is not limited in scope thereto.
Claims
1. A method for effecting a prolonged reduction in the fat content of an animal or fowl consisting of the step of administering a growth promotant to said animal or fowl prior to normal physiological puberty of said animal or fowl.
2. A method as defined in claim 1 wherein the growth promotant is a somatotrophic hormone.
3. A method as defined in claim 2 wherein said somatotrophic hormone is selected from bovine somatotropin and ovine prolactin.
4. A method as defined in claim 3 wherein the growth promotant is a steroidal growth promotant.
5. A method as defined in claim 1 wherein the growth promotant is a compound which upon administration to the animal or fowl causes an increased production of somatotrophic hormone within said animal or fowl.
6. A method as defined in claim 1 wherein the growth promotant is a compound which upon administration to the animal or fowl, at least partially inactivates an inhibitory mechanism which suppresses production of somatotrophic hormone within the animal or fowl.
7. A method as defined in any one of the preceding claims wherein the growth promotant is administered to a bovine, ovine, porcine or caprine animal.
8. A method as defined in any one of the preceding claims wherein the growth promotant is administered during the first six month period following birth of the animal or fowl.
9. A method as defined in any one of claims 1 to 7 wherein the growth promotant is administered during the first three month period following birth of the animal or fowl.
10. A method as claimed in claim 1 wherein the growth promotant is a somatotrophic hormone and is administered in an amount sufficient to increase the normal physiological level of somatotrophic hormone within the animal or fowl by at least a factor of 2.
11. A method as claimed in claim 1 wherein the growth promotant is a somatotrophic hormone and is administered in an amount sufficient to increase the normal physiological level of somatotrophic hormone within the animal or fowl by at least a factor of 3.
12. A method as defined in claim 1 wherein the growth promotant is a somatotrophic hormone and is administered to said animal or fowl in an amount of from 0.1 to 0.5 mg/kg/day.
13. A method as defined in claim 1 wherein the animal is a bovine, ovine, porcine, cervine or caprine animal and wherein a somatotrophic hormone is administered in an amount of from 0.1 to 0.5 mg/kg/day for from 8 to 14 weeks during the first six month period following birth.
14. A method as defined in claim 13 wherein said animal is a lamb.
15. An animal or fowl having a reduced amount of fat which is the product of a method as defined in any one of claims 1 to 14.
16. A process for promoting nett protein synthesis in an animal or fowl through effecting a prolonged reduction in the fat content of said animal or fowl, said process consisting of the step of administering an effective amount of a growth promotant to the animal or fowl prior to normal physiological puberty of said animal or fowl.
17. The use of a growth promotant in the preparation of a medicament for effecting a prolonged reduction in the fat content of an animal or fowl.
18. The use of claim 17 wherein the growth promotant is a somatotrophic hormone.
19. The use of claim 18 wherein said somatotrophic hormone is selected from bovine somatotropin and ovine prolactin.
20. The use of claim 17 wherein the growth promotant is a steroidal growth promotant.
21. The use of claim 17 wherein the growth promotant is a compound which upon administration to the animal or fowl causes an increased production of somatotrophic hormone within said animal or fowl.
22. The use of claim 17 wherein the growth promotant is a compound which upon administration to the animal or fowl at least partially inactivates an inhibitory mechanism which represses production of somatotrophic hormone within the animal or fowl.
23. A method of effecting a prolonged reduction in fat content in an animal or fowl substantially as herein described.
24. The use of a growth promotant in the preparation of a medicament for effecting a prolonged reduction in the fat content of an animal or fowl substantially as herein described.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU49863/93A AU4986393A (en) | 1992-08-19 | 1993-08-19 | Methods for effecting a prolonged reduction in measurable fat content and medicaments for use therein |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| NZ24401792 | 1992-08-19 | ||
| NZ244017 | 1992-08-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1994004183A1 true WO1994004183A1 (en) | 1994-03-03 |
Family
ID=19924066
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/NZ1993/000071 Ceased WO1994004183A1 (en) | 1992-08-19 | 1993-08-19 | Methods for effecting a prolonged reduction in measurable fat content and medicaments for use therein |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU4986393A (en) |
| WO (1) | WO1994004183A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5595752A (en) * | 1994-07-01 | 1997-01-21 | Monsanto Company | Increasing dressing percentage and carcass weight in finishing beef cattle |
| US5670162A (en) * | 1994-07-01 | 1997-09-23 | Monsanto Company | Method and device for implantation of large diameter objects in bovines |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1989000166A2 (en) * | 1987-07-06 | 1989-01-12 | Peptide Technology Ltd | Growth hormone related peptide |
| WO1989008458A1 (en) * | 1988-03-18 | 1989-09-21 | University Of Delaware | Endocrine manipulation to improve body composition of poultry |
| WO1990009190A1 (en) * | 1989-02-08 | 1990-08-23 | The Texas A&M University System | In ovo administration of growth factors alters physiological development and response of poultry |
| WO1992007556A1 (en) * | 1990-10-31 | 1992-05-14 | Amgen Inc. | Polyvinyl alcohol coated pellet of growth hormone |
| US5122513A (en) * | 1991-03-19 | 1992-06-16 | Monsanto Company | Fish growth using bovine placental lactogen |
-
1993
- 1993-08-19 AU AU49863/93A patent/AU4986393A/en not_active Abandoned
- 1993-08-19 WO PCT/NZ1993/000071 patent/WO1994004183A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1989000166A2 (en) * | 1987-07-06 | 1989-01-12 | Peptide Technology Ltd | Growth hormone related peptide |
| WO1989008458A1 (en) * | 1988-03-18 | 1989-09-21 | University Of Delaware | Endocrine manipulation to improve body composition of poultry |
| WO1990009190A1 (en) * | 1989-02-08 | 1990-08-23 | The Texas A&M University System | In ovo administration of growth factors alters physiological development and response of poultry |
| WO1992007556A1 (en) * | 1990-10-31 | 1992-05-14 | Amgen Inc. | Polyvinyl alcohol coated pellet of growth hormone |
| US5122513A (en) * | 1991-03-19 | 1992-06-16 | Monsanto Company | Fish growth using bovine placental lactogen |
Non-Patent Citations (6)
| Title |
|---|
| CHEMICAL ABSTRACTS, vol. 79, no. 15, 15 October 1973, Columbus, Ohio, US; abstract no. 90880v, LEMONNIER ET AL: "LONG-TERM EFFECT OF MOUSE NEONATE FOOD INTAKE ON ADULT BODY COMPOSITION,INSULIN,AND GLUCOSE SERUM LEVELS" page 332; column 2; * |
| FILE SERVER STN KARLSRUHE,FILE BIOSIS ABSTRACT NO.90:24025 & J ANIM SCI 67 (10) 1989 2655-2660 * |
| FILE SERVER STN KARLSRUHE,FILE BIOSIS ABSTRACT NO.92:210118 & ACTA ENDOCRINOL 126 (2). 1992. 155-161 * |
| FILE SERVER STN KARLSRUHE,FILE BIOSIS ABSTRACT NO.93:450016 & ACTA AGRIC SCAND SECT A ANIM SCI 43 (3) 1993 165-172 * |
| FILE SERVER STN KARLSRUHE,FILE MEDLINE ABSTRACT NO.86167851 & J ANIM SCI (1986 FEB) 62 (2) 279-89 * |
| HORM.METAB.RES., vol. 5, no. 3, 1973, pages 223 - 224 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5595752A (en) * | 1994-07-01 | 1997-01-21 | Monsanto Company | Increasing dressing percentage and carcass weight in finishing beef cattle |
| US5670162A (en) * | 1994-07-01 | 1997-09-23 | Monsanto Company | Method and device for implantation of large diameter objects in bovines |
| US5672357A (en) * | 1994-07-01 | 1997-09-30 | Monsanto Company | Method and device for implantation of large diameter objects in bovines |
Also Published As
| Publication number | Publication date |
|---|---|
| AU4986393A (en) | 1994-03-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Reubi | A somatostatin analogue inhibits chondrosarcoma and insulinoma tumour growth | |
| Varner et al. | Temporal serum concentrations of growth hormone, thyrotropin, insulin, and glucagon in sheep immunized against somatostatin | |
| Thomas et al. | Effect of restricted feeding on the relationship between hypophysial portal concentrations of growth hormone (GH)-releasing factor and somatostatin, and jugular concentrations of GH in ovariectomized ewes | |
| Enright et al. | Effects of long-term administration of pituitary-derived bovine growth hormone and estradiol on growth in steers | |
| Davis et al. | Episodic growth hormone secretory patterns in sheep: relationship to gonadal steroid hormones. | |
| Bines et al. | Endocrine control of energy metabolism in the cow: the effect on milk yield and levels of some blood constituents of injecting growth hormone and growth hormone fragments | |
| Baker et al. | Long-term peripheral treatment of immature coho salmon (Oncorhynchus kisutch) with human leptin has no clear physiologic effect | |
| Beermann et al. | Exogenous human growth hormone-releasing factor and ovine somatotropin improve growth performance and composition of gain in lambs | |
| US5134120A (en) | Use of growth hormone to enhance porcine weight gain | |
| Gatford et al. | Sexual dimorphism of circulating somatotropin, insulin-like growth factor I and II, insulin-like growth factor binding proteins, and insulin: relationships to growth rate and carcass characteristics in growing lambs | |
| Cottam et al. | Body growth, carcass composition, and endocrine changes in lambs chronically treated with recombinantly derived insulin-like growth factor-I. | |
| Sandles et al. | Growth and carcass composition of pre-pubertal dairy heifers treated with bovine growth hormone | |
| Hart et al. | Effect of different growth hormone-releasing factors on the concentrations of growth hormone, insulin and metabolites in the plasma of sheep maintained in positive and negative energy balance | |
| Sechen et al. | Response to metabolic challenges in early lactation dairy cows during treatment with bovine somatotropin | |
| Hufstedler et al. | Physiological and hormonal responses of lambs repeatedly implanted with zeranol and provided two levels of feed intake | |
| Gopinath et al. | Effects of porcine growth hormone on glucose metabolism of pigs: I. Acute and chronic effects on plasma glucose and insulin status | |
| Gulay et al. | Responses of Holstein cows to a low dose of somatotropin (bST) prepartum and postpartum | |
| Vanderwal et al. | Effect of anabolic agents on performance of calves | |
| EP0146703B1 (en) | Use of growth hormone to enhance ruminant mammary development | |
| Allen et al. | Changes in pancreatic hormones associated with coccidiosis | |
| WO1989008458A1 (en) | Endocrine manipulation to improve body composition of poultry | |
| Zainur et al. | Recombinant growth hormone in growing lambs: effects on growth, feed utilization, body and carcass characteristics and on wool growth | |
| Moseley et al. | Serum growth hormone and nitrogen metabolism responses in young bull calves infused with growth hormone-releasing factor for 20 days | |
| Fox et al. | Ostertagia ostertagi infection in the calf: effects of a trickle challenge on the hormonal control of digestive and metabolic function | |
| WO1994004183A1 (en) | Methods for effecting a prolonged reduction in measurable fat content and medicaments for use therein |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AT AU BB BG BR BY CA CH CZ DE DK ES FI GB HU JP KP KR KZ LK LU MG MN MW NL NO NZ PL PT RO RU SD SE SK UA US VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| 122 | Ep: pct application non-entry in european phase | ||
| NENP | Non-entry into the national phase |
Ref country code: CA |