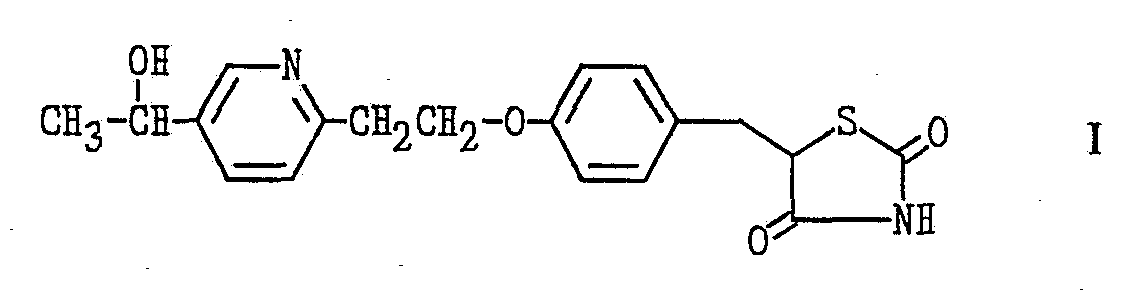WO1993022445A1 - A process for producing pioglitazone metabolite - Google Patents
A process for producing pioglitazone metabolite Download PDFInfo
- Publication number
- WO1993022445A1 WO1993022445A1 PCT/US1993/003569 US9303569W WO9322445A1 WO 1993022445 A1 WO1993022445 A1 WO 1993022445A1 US 9303569 W US9303569 W US 9303569W WO 9322445 A1 WO9322445 A1 WO 9322445A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- microorganism
- thiazolidinedione
- pyridyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*(C(C(Cc(cc1)ccc1O*c1ncc(*)cc1)S1)=O)C1=O Chemical compound C*(C(C(Cc(cc1)ccc1O*c1ncc(*)cc1)S1)=O)C1=O 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P17/00—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms
- C12P17/16—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms containing two or more hetero rings
- C12P17/167—Heterorings having sulfur atoms as ring heteroatoms, e.g. vitamin B1, thiamine nucleus and open chain analogs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/20—Bacteria; Culture media therefor
- C12N1/205—Bacterial isolates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12R—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES C12C - C12Q, RELATING TO MICROORGANISMS
- C12R2001/00—Microorganisms ; Processes using microorganisms
- C12R2001/01—Bacteria or Actinomycetales ; using bacteria or Actinomycetales
- C12R2001/465—Streptomyces
- C12R2001/55—Streptomyces hygroscopicus
Definitions
- Ciglitazone ((+-)-5-[4-[(1-methylcyclohexyl)methoxy]benzyI]-2,4-thiazolidinedione) is characteristic of a class of thiazolidine antidiabetic agents which lower blood glucose in animal models of NIDDM, while actually reducing circulating concentrations of insulin. This is believed to be accomplished by improving the responsiveness of the peripheral tissues to insulin. See, e.g., Chang, et al., Diabetes 32:830-838 (September 1983).
- U.S. Patent 2,602,769 discloses the oxygenation of steroids by mucorales fungi.
- a method of deoxygenating the N-oxide of a pyridine ring and oxygenating the 5-position ethyl side chain of the pyridine ring in a compound of the formula IV to produce a compound of the formula I or II which comprises:
- oral administration preferably, it is administered one to three times a day in a daily dose of 0.05 to 100 mg/kg, and preferably 0.05 to 10 mg/kg.
- a mixture of pioglitazone (48.33 g), chloroform (1.2 1), and methanol (375 ml) is stirred at room temperature.
- m-Chloroperbenzoic acid 41.90 g of 60% technical grade, 25.1 g
- the stirred mixture becomes a clear solution within one hour of the addition of m-chloroperbenzoic acid.
- the solution is stirred overnight at room temperature and then worked up extracting and washing sequentially with saturated sodium bicarbonate (600 ml) plus water (500 ml), with aqueous sodium sulfite (10 g in 200 ml of water), and with water (500 ml).
- NRRL 18975 produced long spore chains of tight spirals. Spore surfaces were rugose. Vegetative hyphae showed no evidence of fragmentation.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- Biotechnology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Virology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
The present invention provides a method for making pharmaceutical compounds. More particularly the present invention provides a new method for making oxygenated pioglitazone derivatives of formulas (I) and (II), which are metabolites of pioglitazone and useful for the treatment of diabetes and as insulin sensitizing agents.
Description
A PROCESS FOR PRODUCING PIOGLITAZONE METABOLITES
FIELD OF INVENTION
The present invention provides a new method of making organic compounds. In particular, the present invention provides a new method for making oxygenated pioglitazone derivatives. These compounds are metabolites of pioglitazone and are useful for the treatment of diabetes and as insulin sensitizing agents.
BACKGROUND
Pioglitazone hydrochloride ((±)-5-[4-[2-(5-ethyl-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione monohydrochloride), a thiazolidinedione derivative, is currently under clinical evaluation and is expected to effectively ameliorate the abnormal glucose and lipid metabolism associated with noninsulin dependent diabetes mellitus (NIDDM) or obesity (cf. Y. Momose et al., Chem. Pharm. Bull., 39:1440 (1991)).
International application, PCT/US92/02566, filed 6 April 1992 (WO 92/18501, published 29 October 1992), discloses thiazolidine derivatives, including 5-[4-[2-(5-acetyI-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione and 5-[4-[2-[5-(1-hydroxyethyl)-2-pyridyl]ethoxy]benzyl]-2,4-thiazolidinedione, which are pioglitazone metabolites.
Another thiazolidine derivative undergoing clinical studies as a hypoglycemic agent is englitazone sodium, 5-([3,4-dihydro-2-(phenylmethyl)-2H-1-benzopyran-6-yl]methyI]-2,4-thiazolidinedione sodium salt) (cf. D.A. Clark et al., J. Med. Chem., 34:319-325 (1991)).
Ciglitazone ((+-)-5-[4-[(1-methylcyclohexyl)methoxy]benzyI]-2,4-thiazolidinedione) is characteristic of a class of thiazolidine antidiabetic agents which lower blood glucose in animal models of NIDDM, while actually reducing circulating concentrations of insulin. This is believed to be accomplished by improving the responsiveness of the peripheral tissues to insulin. See, e.g., Chang, et al., Diabetes 32:830-838 (September 1983).
CS-045 is an antidiabetic, thiazolidinedione derivative. Its activity and preparation are described in Drugs Fut. 1991, 16(9).
Also, thiazolidine derivatives useful for the treatment of diabetes are described in U.S. patents 4,287,200; 4,687,777; and 4,572,912. Their effect on insulin resistance are described, e.g., in Chang et al., Diabetes 32:839-845 (1983) and Chang et al., Diabetes 32:830-838 (1983).
The chemical synthesis of several of these thiazolidinedione derivatives have proved problematic and inefficient. In particular, the chemical synthesis of the pioglitazone metabolites includes long, tedious synthetic routes with poor or highly variable yields. What is needed in the art is an easier, more efficient method for performing the synthesis of these compounds.
INFORMATION DISCLOSURE
International application, PCT/US92/02566, filed 6 April 1992 (WO 92/18501, published
29 October 1992), discloses thiazolidine derivatives, including 5-[4-[2-(5-acetyl-2-
pyridyl)ethoxy]benzyI]-2,4-thiazolidinedione and 5-[4-[2-[5-(1-hydroxyethyl)-2-pyridyl]ethoxy]benzyl]-2,4-thiazolidinedione, which are pioglitazone metabolites, and describes their chemical synthesis.
U.S. Patent 4,687,777 and EP 193-256, published 9 March 1986, disclose new 5-((ethyl-2-pyridyl) ethoxy) benzyl thiazolidine-dione compounds, including pioglitazone and
pharmacologically acceptable salts, which have hypoglycaemic and hypolipaemic activity and thus are useful for treating diabetes, and describes the chemical synthesis of these compounds.
European published application 0 508 740 discloses N-oxide thiazolidinedione derivatives, including the compound of formula TV. as shown in the Formula Chart below, having hypoglycemic and hypolipidemic activity.
U.S. Patent 4,444,779 and EP 0 008 203 disclose new thiazolidine-dione compounds, including ciglitazone and pharmacologically acceptable salts thereof, which have hypoglycaemic and hypolipaemic activity, and describes the chemical synthesis of these compounds.
Derwent Abstract 88-057989/09 of EP 257 781 discloses new substituted pyridine compounds used in the preparation of hypoglycaemic and hypolipidaemic pyridyl-ethoxy-methylphenyl thiazolidine-dione compounds.
The use of various microorganisms for the oxygenation of certain organic molecules is known in the art, for example, in the following references: R.A. Johnson, in "Oxidation in Organic Chemistry, Part C," ed. W.S. Trahanovsky, Academic Press, N.Y., 1978. pp. 131-210; H.G. Davies, et al., "Biotransformations in Preparative Organic Chemistry", Academic Press, N.Y., 1989, pp. 157-219.
U.S. Patent 2,602,769 discloses the oxygenation of steroids by mucorales fungi.
In the process of the present invention, a microorganism may be used to deoxygenate and then oxygenate a compound.
Y. Momose et al., Chem. Pharm. Bull., 39:1440 (1991); K. Meguro et al., Japan. Patent
139182 (1988); Chem. Abstr., 109:6504h (1988); and T. Sohda et al., Arzneim.-Forsch./Drug Res. 40 (1), Nr. 1:37 (1990); disclose the chemical synthesis and biological activity of various thiazolidinedione derivatives, including pioglitazone.
D.A. Clark et al., J. Med. Chem., 34:319-325 (1991) discloses the chemical synthesis of substituted dihydrobenzopyran and dihydrobenzofuran thiazolidine-2,4-diones, including englitazone.
Drugs Fut.1991, 16(9) discloses the multistep process, via carbon alkylation, for the preparation of the thiazolidinedione CS-045.
PCT/US92/10329, filed 4 December 1992, discloses a new reduction method for making thiazolidinedione derivatives, particularly ciglitazone, pioglitazone and englitazone.
SUMMARY OF THE INVENTION
The present invention particularly provides:
A process for producing a compound of the formula I or II which comprises:
the biotransformation of a compound of the formula IV using a microorganism in an aqueous nutrient medium containing an assimilable source of carbon and an assimilable source of nitrogen under aerobic conditions;
A biologically pure culture of the microorganism Streptomyces hygroscopicus, characterized as the strain NRRL 18975;
A method of using a compound of the formula IV to produce a compound of the formula I or II which comprises:
adding the compound of formula IV to a metabolizing culture of a microorganism;
A method of using a microorganism Streptomyces hygroscopicus, strain NRRL 18975, or a mutant thereof, to produce a compound of the formula I or II which comprises:
adding a compound of the formula IV to a metabolizing culture of the microorganism; and
A method of deoxygenating the N-oxide of a pyridine ring and oxygenating the 5-position ethyl side chain of the pyridine ring in a compound of the formula IV to produce a compound of the formula I or II which comprises:
adding the compound of formula IV to a metabolizing culture of a microorganism.
By "biotransformation" is meant the use of microorganisms and/or isolated, partially purified enzymes for the conversion of a given substrate into a useful product. H.G. Davies, et al., "Biotransformations in Preparative Organic Chemistry", Academic Press, N.Y., 1989, p. IX.
By "metabolizing" is meant carrying out the processes of metabolism. A definition of metabolism may be found in A.L. Lehninger, "Principles of Biochemistry", Worth Publ., New York, 1982, p. 333.
The pioglitazone metabolites produced by the process of the present invention are 5-[4-
[2-(5-acetyl-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione and 5-[4-[2-[5-(1-hydroxyethyl)-2- ρyridyl]ethoxy]benzyl]-2,4-thiazolidinedione. They are represented as Formulas I and II in the Formula Chart below. These compounds have excellent hypoglycemic and hypolipidemic activity and thus are useful as therapeutic agents to treat diabetes, hyperlipidemia and the like.
These compounds, their pharmacologically acceptable salts or pure stereoisomeric forms can be used for the treatment of diabetes of mammals, including man, as they are or by combining them with known pharmacologically acceptable carriers, excipients, fillers and the like. Normally, such a compound, its pharmacologically acceptable salt or stereoisomeric form can be administered orally in the form of, for example, tablets, capsules including soft capsules and micro capsules, powders, granules or the like. If necessary, it can also be administered parenterally in the form of injectable solutions, suppositories, pellets or the like. In the case of
oral administration, preferably, it is administered one to three times a day in a daily dose of 0.05 to 100 mg/kg, and preferably 0.05 to 10 mg/kg. These pioglitazone metabolites, their biological activity and chemical synthesis is further described in International application,
PCT/US92/02566, filed 6 April 1992 (WO 92/18501, published 29 October 1992), which is hereby incorporated by reference herein.
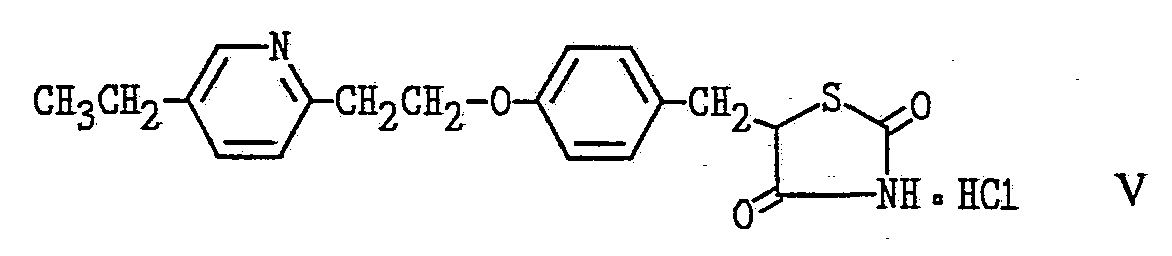
Previously, the chemical synthesis of these pioglitazone metabolites proved problematic and inefficient. In particular, their synthesis includes long, tedious routes with poor or highly variable yields. Surprisingly and unexpectedly, the present invention provides a new, more efficient method for producing these compounds. By using the new method of the present invention, the metabolites are produced by adding pioglitazone of Formula III, pioglitazone hydrochloride of Formula V and/or pioglitazone N-oxide of Formula IV, as shown in the Formula Chart below, to growing cultures of microorganisms. The species Streptomyces hygroscopicus is preferred for effecting this transformation and the novel strain NRRL 18975 is particularly preferred. Pioglitazone hydrochloride and pioglitazone N-oxide are preferred as starting materials, with pioglitazone N-oxide being most preferred as the starting material because of its better solubility characteristics in comparison with pioglitazone. This new method is faster and easier and results in improved yield of the desired metabolites.
For example, Upjohn soil isolate 02179 (UC® 11099) deoxygenates the pyridine-N-oxide of pioglitazone-N-oxide and oxygenates its 5-position ethyl side chain. It also oxygenates the 5-position ethyl side chain of pioglitazone. The culture grows well and produces a white aerial mass which becomes hygroscopic and turns dark gray to black on some media. The mature spore chains are long and form tight spirals. The spore surface is rugose. It has wide range of growth temperature and utilizes most carbon sources. The whole-cell hydrolysate contains LL-diaminopimelic acid. Based on the morphological and cultural characteristics and the whole-cell hydrolysate analysis of the culture, the species classification Streptomyces hygroscopicus has been determined for the culture. The characterization of this microorganism is further described in Example 2 below. A subculture of this microorganism was deposited under the provisions of the Budapest Treaty on 6 May 1992 in the permanent collection of the Northern Region Research Center, ARS; U.S. Dept. of Agriculture; Peoria, Illinois, USA. Its accession number is NRRL 18975.
Table 4 lists other strains of Streptomyces hygroscopicus useful in the process of the present invention. The Upjohn Culture Collection (UC®) Number for these microorganisms is given. Since these microorganisms have been deposited in a permanent collection of a recognized depository, their official accession number or deposit number is also given. The following two microorganisms are also useful in the process of the present invention: Rhizopus Japonicus (UC No. 4842; Deposit No. LFO 4758) and Streptoverticillium sp. (UC No. 2064;
Deposit No. NRRL 21066). The use of these microorganisms in the process of the present invention results in the production of one or both of the compounds of the Formulas I and II.
Pioglitazone metabolites (Formulas I and II) are produced when Streptomyces hygroscopicus is fermented in an aqueous nutrient medium under submerged aerobic conditions in the presence of pioglitazone, pioglitazone hydrochloride and/or pioglitazone N-oxide.
Typically the microorganism is fermented in a nutrient medium containing a carbon source and a proteinaceous material. Preferred carbon sources include glucose, brown sugar, sucrose, glycerol, starch, comstarch, lactose, dextrin, molasses, and the like. Preferred nitrogen sources include cottonseed flour, corn steep liquor, yeast, autolyzed brewer's yeast with milk solids, soybean meal, cottonseed meal, cornmeal, milk solids, pancreatic digest of casein, distillers' solids, animal peptone liquors, meat and bone scraps, and the like. Combinations of these carbon and nitrogen sources can be used advantageously. Trace metals, for example, zinc, magnesium, manganese, cobalt, iron and the like need not be added to the fermentation medium since tap water and unpurified ingredients are used as medium components.
Production of pioglitazone metabolites can be induced at any temperature conducive to satisfactory growth of the microorganism between about 23° and 32° C and preferably at about 28°C. Ordinarily, optimum production of the pioglitazone metabolites is obtained in about 1 to 4 days after addition of the pioglitazone or pioglitazone-N-oxide to the growing culture, and preferably in about 1.5 days. The fermentation broth normally remains weakly basic (pH 7.4 -9.0) during the fermentation. The final pH is dependent, in part, on the buffers present, if any, and in part, on the initial pH of the culture medium. It is advantageously adjusted to about pH 6.5-7.5, and preferably 7.2, prior to sterilization.
When growth is carried out in shake flasks or large vessels and tanks, it is preferable to use the vegetative form, rather than the spore form, of the microorganism for inoculation to avoid a pronounced lag in the production of the pioglitazone metabolites and the attendant inefficient utilization of the equipment. Accordingly, it is desirable to produce a vegetative inoculum in an aqueous nutrient medium by inoculating this medium with an aliquot from a soil or a slant culture. When a young, active vegetative inoculum has thus been secured, it is transferred aseptically to other shake flasks or tanks. The medium in which the vegetative inoculum is produced can be the same as, or different from, that utilized for the production of the antibiotics, as long as it is such that adequate growth of the microorganism is obtained.
A variety of procedures can be employed to isolate and purify the pioglitazone metabolites from the fermentation broth, for example, by chromatographic adsorption procedures followed by elution with a suitable solvent, column chromatography, partition chromatography, and crystallization from solvents and combinations thereof.
In the preferred recovery process, the pioglitazone metabolites are extracted from the
whole beer. Column chromatography techniques, preferably over silica gel, are used to perform the initial purification. Final purification of the pioglitazone metabolites is achieved by chromatography and crystallization from organic solvents.
It will be apparent to those skilled in the art that compounds of Formulas I and II contain several asymmetric carbons. All of the enantiomeric and stereoisomeric forms of the compounds of Formulas I and II are included within the scope of the present invention.
DETAILED DESCRIPTION OF THE INVENTION
The present invention is seen more fully by the examples below, but these examples are not to be construed as limiting the scope of this invention.
PREPARATION 1 Preparation of Pioglitazone-N-oxide (also known as (+)-5-[4-[2-(5-ethyl- 2-pyridyl-N-oxide)ethoxy]benzyl]-2,4-thiazolidinedione) Formula IV (Formula Chart)
A mixture of pioglitazone (48.33 g), chloroform (1.2 1), and methanol (375 ml) is stirred at room temperature. m-Chloroperbenzoic acid (41.90 g of 60% technical grade, 25.1 g) is added as the solid in three portions over a period of 45 minutes. The stirred mixture becomes a clear solution within one hour of the addition of m-chloroperbenzoic acid. The solution is stirred overnight at room temperature and then worked up extracting and washing sequentially with saturated sodium bicarbonate (600 ml) plus water (500 ml), with aqueous sodium sulfite (10 g in 200 ml of water), and with water (500 ml). The hazy solution is dried (sodium sulfate), filtered, and concentrated under reduced pressure. The oily, warm residue is immediately dissolved in acetonitrile (350 ml) and crystallization usually begins immediately. Crystallization is allowed to proceed for 24 hours after which colorless crystals of IV (42.11 g) are collected.
Recrystallization from ethyl acetate gives IV as colorless crystals.
Physical characteristics are as follows:
MP. 159-161°C.
% NMR (CD3OD) δ 8.23, 7.49, 7.44, 7.15, 6.86, 4.67, 4.38, 3.35, 3.30, 3.07, 2.67, 1.25.
Anal. Found: C, 60.82; H, 5.69; N, 7.40; S, 8.45.
PREPARATION 2 Protocol for Biotransformation of Pioglitazone (also known as (+)-5-[4- [2-(5-ethyI-2-pyridyl)ethoxy]benzyl]-2,4-thiazoIidinedione) Formula III, pioglitazone hydrochloride (also known as (+)-5-[4-[2-(5-ethyl-2- pyridyl)ethoxy]benzyI]-2,4-thiazolidinedione monohydrochloride) Formula V and/or Pioglitazone N-oxide (also known as (+)-5-[4-[2-(5- ethyl-2-pyridyl-N-oxide)ethoxy]benzyl]-2,4-thiazolidinedione) Formula
IV (Formula Chart)
A. Seed Fermentation
Streptomyces hygroscopicus (NRRL 18975)
Frozen agar plugs of NRRL 18975 (stored over liquid nitrogen) are aseptically transferred into a 100 ml vol of GS-7 (sterile) medium contained in a 500 ml large mouth shake flask. GS-7 is composed of Cerelose and Pharmamedia, each added at 25 g/1 of tap water. The pH of the medium is adjusted to pH 7.2 using ammonium hydroxide. The medium is sterilized by autoclaving for 30 min. Inoculated GS-7 is shaken at 250 rpm at 28° C for 72 hrs. This fermentation is used to inoculate the biotransformation process at a 5% rate.
B. Biotransformation Process
100 ml vols of GS-7 contained in 500 ml large mouth fermentation flasks are inoculated with the 72 hr seed fermentation at a 5% rate. The inoculated GS-7 is shaken at 250 rpm at 28°C for 48 hrs. At this time pioglitazone, pioglitazone hydrochloride or pioglitazone N-oxide dissolved in dimethylformamide (DMF) is added to the fermentation in a range between 20 and 60 mg/flask using no more than 0.3 ml DMF per flask. The fermentations containing
Pioglitazone or Pioglitazone N-oxide are continued as previously indicated for 24-96 hrs.
EXAMPLE 1 Isolation of Metabolites, 5-[4-[2-[5-(1-hydroxyethyl)-2-pyridyl]ethoxy]benzyl]- 2,4-thiazolidinedione and 5-[4-[2-(5-acetyl-2-pyridyl)ethoxy]benzyl]-2,4- thiazolidinedione, Formulas I and II (Formula Chart)
The contents of 120 flasks, each containing 100 ml fermentation as described in the protocol for biotransformation and which has been shaken at 28°C for 24 hrs following addition of pioglitazone-N-oxide (0.050 g per flask, total 6.0 g) in dimethylformamide (0.2 ml per flask, total 24 ml), are pooled in a 20 1 carboy. The mixture is stirred vigorously with methylene chloride (methylene chloride, 7 1) for 3 hrs. The solvent layers separate slowly and after 24 hrs, the methylene chloride layer (1.75 1) is siphoned off under a slight vacuum. Additional methylene chloride (2 1) is added, the mixture is again stirred vigorously and this procedure continues through three more collections of methylene chloride extract (of 15 1 of methylene chloride added to the mixture, about 10 1 are collected). The methylene chloride extracts are dried (sodium sulfate), filtered, and concentrated. The oily residues are placed under a stream of nitrogen for 24-48 hrs, which removes much of the dimethylformamide that is carried into the extract. Upon standing for several days, solids precipitate in the oily residues. These are collected by using small amounts of methylene chloride to wash and transfer during the filtration process. In this way, a yellowish-brown solid (2.27 g) is collected. Examination of this solid by thin layer chromatography (TLC, silica gel plates, 10:1 methylene chloride-methanol; 1:1 acetone-hexane as solvents for TLC) shows the presence of materials with the same Rf as authentic samples of compounds of formulas I and II as well as other components. The components of the solid extract are separated by a series of column chromatographies using a
silica gel-solid extract ratio of 50-125:1. Columns are eluted with either 4-5% methanol in methylene chloride or 30-60% acetone in hexane. From these chromatographic separations, there is isolated 5-[4-[2-(5-acetyl-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione (0.203 g), the compound of formula II, which is recrystallized from acetone-hexane to give colorless needles (0.147 g).
Physical characteristics are as follows:
Mp 113-115°C; 1H NMR spectrum (CDCl3) is identical to that of an authentic sample.
Also isolated is 5-[4-[2-[5-(I-hydroxyethyl)-2-pyridyl]ethoxy]benzyl]-2,4- thiazolidinedione (0.756 g), the compound of formula I, which is recrystallized from acetone-hexane to give colorless crystals (0.680 g).
Physical characteristics are as follows:
Mp 154-155°C; [α]D 25.5 -12.4°(C, 1.18 in ethanol); % NMR spectrum (CDCI3) is identical with the spectrum of an authentic sample.
The following similar experimental procedure resulted in improved yields of the compounds of formulas I and II:
A solution of pioglitazone-N-oxide (6.0 g.) in dimethylformamide (total volume of solvent and substrate, 24 mL) is divided equally among 120 flasks each containing 100 mL of medium and growing culture and shaking at 28°C is continued for 24 hrs. The contents of the flasks are pooled in a 20 L glass carboy, methylene chloride (6 L) is added, and the two-phase system is stirred vigorously with an air driven motor for 2 hrs. The solvent layers separate slowly and after about 24 hrs, the methylene chloride layer is siphoned off under a slight vacuum. The extraction process is repeated by adding fresh methylene chloride in the amount removed to the pooled fermentation, stirring vigorously, and removing methylene chloride by siphon. This extraction process is done a total of ten times. Extractions 1-7 (total volume 18.2 L) are combined and extractions 8-10 (total volume 12.2 L) are combined.
Combined extracts 1-7 are dried (sodium sulfate), filtered, and concentrated. The residual material is chromatographed over silica gel (40-63 μm, 400 g, preconditioned with i-propyl alcohol/ethyl acetate/hexane/glacial acetic acid (5/65/29.5/0.5)) packed in a 4.7 × 45 cm Michel-Miller column. The residue is applied to the column by first dissolving 1:1 chloroformmethanol, mixing with gravity grade silica gel (63-200 μm, 35 g), and removing the solvent on the rotary evaporator under reduced pressure. The dry solid is placed in a second 4.7 × 45 cm Michel-Miller column held in a vertical position. The empty space in the column is filled with 40-63 μm silica gel and the column is eluted with the same solvent mixture used to precondition the column. Fractions of 50 mL volume are collected and are pooled based on the results of tlc analysis.
Fractions 21-33 are pooled and contain ketone of formula II and a small amount of
pioglitazone of formula III (total, 1.20 g). This material is chromatographed over 200 g of 40-63μm silica gel using ethyl acetate to apply the material to the column and i-propyl
alcohol/ethyl acetate/hexane/ glacial acetic acid (5/65/29.5/0.5) to elute the column. Fractions of 30 mL volume are collected with fractions 11-15 containing the desired ketone of formula II (0.955 g) still mixed with pioglitazone of formula III. The mixture is again chromatographed over 200 g of 40-63um silica gel, eluting with chloroform/i-propyl alcohol/acetic acid (94/5/1) and collecting 40 mL fractions. Fractions 12-15 contain crystalline ketone of formula II (0.602 g) and fractions 19-26 contain impure pioglitazone of formula III (0.184 g). The combined fractions 12-15 are recrystallized from acetone-hexane, giving the ketone of formula II (0.511 g).
Physical characteristics are as follows:
Mp 113-115°C.
The 1H NMR spectrum in CDCl3 of the sample of ketone of formula II is identical to the spectrum of an authentic sample of ketone of formula II.
Fractions 34-40 are pooled and contain a mixture of ketone of formula II and alcohol of formula I (total, 0.41 g). This mixture is plated onto 8 g of silica gel and placed in line in front of a column of 200 g of 40-63 μm silica gel. The system is eluted with chloroform/i-propyl alcohol/acetic acid (94/5/1) and fractions of 45 mL volume are collected. Fractions 15-20 contain pure ketone of formula II (0.082 g, total 0.684 g). Fractions 21-23 contain impure ketone of formula II (0.030 g). Fractions 191-196 contain pure alcohol of formula I (0.151 g).
Fractions 41-63 are pooled and contain primarily alcohol of formula I as a yellow solid (1.34 g). The solid is dissolved in methanol-ethyl acetate and while hot is decolorized with activated charcoal. After filtration and removal of solvent, an off-white solid (1.26 g) remains and is chromatographed over 200 g of 40-63 μm silica gel. Fractions of 50 mL volume are collected and fractions 85-106 contain pure crystalline alcohol of formula I (1.0 g).
Recrystallization from acetone-hexane gives alcohol of formula I (0.884 g).
Physical characteristics are as follows:
Mp 154.5-155.5°C.
[α]D -8.1° (c, 0.620, ethanol).
Fractions 64-77 are pooled and contain a mixture of alcohol of formula I and pioglitazone-N-oxide of formula IV (total, 0.11 g).
Fractions 78-120 are pooled and contain primarily pioglitazone-N-oxide of formula IV (0.282 g). The material is plated onto silica gel (7 g) and placed in line in front of a column of 200 g of 40-63 μm silica gel. The system is eluted with chloroform/i-propyl alcohol/acetic acid (94/6/1) (2 L) followed by CHCl3/i-propyl alcohol/acetic acid (88/10/2). Pure pioglitazone-N-oxide of formula IV (0.186 g) is eluted in fractions 33-42 (45 mL volume).
Combined extracts 8-10 are dried (sodium sulfate), filtered, and concentrated. The residue is plated onto silica gel (25 g, 40-63 μm), which is placed ahead of a column of 200 g of 40-63 μm silica gel and eluted with i-propyl alcohol/ethyl acetate/hexane/acetic acid
(5/65/29.5/0.5). Fractions (45 mL volume) 29-40 contain impure alcohol of formula i (0.295 g), which is rechromatographed over 200 g of 40-63 μm silica gel using 5 L of chloroform/i-propyl alcohol/acetic acid (94/5/1) followed by a 88/10/2 mixture of the same. Fractions (45 mL volume) 156-176 contain pure alcohol of formula I [0.201 g, total (using the 1.0 g weight from fractions 64-77, above)] 1.35 g.
Physical characteristics are as follows:
[α]D -6.7° (c, 0.780, ethanol).
The 1H NMR spectrum of the alcohol of formula I is identical with that of an authentic sample of formula I
EXAMPLE 2 Characterization of Isolate 02179 (UC 11099) (NRRL 18975)
Materials and Methods:
Microbial Strain: UC 11099 was isolated from a soil sample collected in Tennessee.
The culture was assigned the Isolate number 02179 in November of 1990, and later it was deposited in the Upjohn Culture (UC) Collection as UC 11099. It was also deposited on 6 May 1992 in the permanent collection of the Northern Research Center, U.S. Dept of Agriculture, Peoria, IL, under the accession number NRRL 18975.
Morphological Characterization: The aerial mycelia of the culture grown on ISP media
2, 3, 4, and 5 (Difco) for 14 days at 28°C were examined directly by light microscopy. Spore surfaces were examined by scanning electron microscopy according to the methods described in A. Dietz, and J. Mathews. 1969. Scanning electron microscopy of selected members of the Streptomyces hygroscopicus group. Appl. Microbiol. 18: 694-696.
Cultural and Physiological Characterization: Methods described in the following references were employed: E. B. Shirling and D. Gottlieb. 1966. Methods for characterization of Streptomyces species. Int. J. Syst. Bacteriol. 16: 313-340; and A. Dietz and D. W. Thayer (ed.). 1980. Actinomycete Taxonomy (Procedures for studying aerobic actinomycetes with emphasis on the strεptomycetes). SIM special publication No. 6. Soc. for Indus. Microbiol. Arlington, VA. Observations were made after 14 days incubation at 28°C. Growth temperature range was determined on ISP medium 2.
Cell-wall and Sugar Analysis: Methods described in J.L. Staneck and G.D. Roberts, 1974, "Simplified Approach to Identification of Aerobic Actinomycetes by Thin Layer
Chromatography," Applied Microbiology, 28:226-231, or methods analogous thereto, were followed to prepare whole cell hydrolysates and to analyze these hydrolysates for amino acid composition and sugar analysis by thin-layer chromatography.
Results:
Morphological Characterization: NRRL 18975 produced long spore chains of tight spirals. Spore surfaces were rugose. Vegetative hyphae showed no evidence of fragmentation.
Cultural and Physiological Characterization: The cultural and physiological characteristics of NRRL 18975 on various media are shown in Tables 1 and 2 below. The isolate grew well and produced white aerial mass which became hygroscopic and turned dark gray to black on most media as the incubation time prolonged beyond two weeks. The reverse side of the colony was yellowish brown to yellowish gray. Melanoid pigment was not produced. The culture strongly solubilized tyrosine and weakly solubilized casein. It did not solubilize calcium malate or xanthine. It hydrolyzed starch on ISP medium 4. Nitrate was not reduced to nitrite.
Utilization of carbon sources by NRRL 18975 is shown in Table 3 below.
The isolate grew well and produced white aerial mass which turned dark gray on the positive control (ISP medium 9 plus glucose). A scant black growth was observed on the negative control (ISP medium 9 plus no sugar). The isolate utilized a wide range of sugars, including glucose, arabinose, xylose, mannitol, fructose, rhamnose and raffinose. It did not utilize sucrose or cellulose. Utilization of inositol was questionable.
The isolate grew in the temperature range of 24-45°C with the optimum range of 28-37°C. It did not grow at 55°C.
Cell-wall and Sugar Analysis: The cell-wall of NRRL 18975 contained LL-diaminopimelic acid; no meso isomer was detected. The whole-cell hydrolysate contained galactose.
Conclusion
Isolate 02179 (UC 11099) (NRRL 18975) has been studied and found to have the macroscopic, micro-scopic and whole-cell hydrolysate properties of the genus Streptomyces. The hygroscopic colony appearance and the rugose surfaced spores further place the culture in the species hygroscopicus. Therefore, Streptomyces hygroscopicus strain 02179 has been determined for NRRL 18975.
Claims
1. A process for producing a compound of the formula I or II:
which comprises:
the biotransformation of a compound of the formula IV:
2. The process of Claim 1 wherein the microorganism is a strain of Streptomyces hygroscopicus or a mutant thereof.
3. The process of Claim 2 wherein the strain is NRRL 18975 or a mutant thereof.
4. The process of Claim 3 which further comprises the biotransformation of a compound of the formula III:
5. The process of Claim 4 which further comprises isolating the compounds of formulas I and II which are 5-[4-[2-[5-(1-hydroxyethyl)-2-pyridyl]ethoxy]benzyl]-2,4-thiazolidinedione and 5-[4-[2-(5-acetyl-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione.
6. The process of Claim 1 wherein the biotransformation is conducted at a temperature of from about 23°C to about 32°C.
7. The process of Claim 6 wherein the temperature is about 28°C.
8. The process of Claim 1 wherein the biotransformation is conducted at a pH of from about 6.5 to about 7.5.
9. The process of Claim 8 wherein the pH is about 7.2.
10. The process of Claim 1 wherein the nutrient medium contains carbon sources of from about 1 to about 5% by weight and nitrogen sources of from about 1 to about 5% by weight.
11. The process of Claim 4 wherein the biotransformation is complete in about 1 to 4 days after the addition of the compound of formula III or the compound of formula IV to a growing culture of the microorganism.
12. The process of Claim 11 wherein the biotransformation is complete in about 1.5 days.
13. A biologically pure culture of the microorganism Streptomyces hygroscopicus, characterized as the strain NRRL 18975.
to produce a compound of the formula I or II:
adding the compound of formula IV to a metabolizing culture of a microorganism.
15. The method of Claim 14 wherein the microorganism is Streptomyces hygroscopicus, strain NRRL 18975, or a mutant thereof.
16. The method of Claim 15 which further comprises isolating the compounds of formulas I and II which are 5-[4-[2-[5-(1-hydroxyethyl)-2-pyridyl]ethoxy]benzyl]-2,4-thiazolidinedione and 5-[4-[2-(5-acetyI-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione.
17. A method of using a microorganism Streptomyces hygroscopicus, strain NRRL 18975, or a mutant thereof, to produce a compound of the formula I or II:
which comprises: adding a compound of the formula IV:
to a metabolizing culture of the microorganism.
18. The method of Claim 17 which further comprises isolating the compounds of formulas I and II which are 5-[4-[2-[5-(1-hydroxyethyl)-2-pyridyl]ethoxylbenzyl]-2,4-thiazolidinedione and 5-[4-[2-(5-acetyl-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione.
19. A method of deoxygenating the N-oxide of a pyridine ring and oxygenating the 5-position ethyl side chain of the pyridine ring in a compound of the formula IV:
to produce a compound of the formula I or II:
adding the compound of formula IV to a metabolizing culture of a microorganism.
20. The method of Claim 19 wherein the microorganism is Streptomyces hygroscopicus, strain NRRL 18975, or a mutant thereof.
21. The method of Claim 20 which further comprises isolating the compounds of formula I and II which are 5-[4-[2-[5-(1-hydroxyethyl)-2-pyridyl]ethoxy]benzyl]-2,4-thiazolidinedione and 5-[4-[2-(5-acetyl-2-pyridyl)ethoxy]benzyl]-2,4-thiazolidinedione.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US87895292A | 1992-05-05 | 1992-05-05 | |
| US07/878,952 | 1992-05-05 | ||
| US88205692A | 1992-05-13 | 1992-05-13 | |
| US07/882,056 | 1992-05-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1993022445A1 true WO1993022445A1 (en) | 1993-11-11 |
Family
ID=27128514
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1993/003569 Ceased WO1993022445A1 (en) | 1992-05-05 | 1993-04-21 | A process for producing pioglitazone metabolite |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU4104593A (en) |
| WO (1) | WO1993022445A1 (en) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1999061660A1 (en) * | 1998-05-22 | 1999-12-02 | Creatogen Aktiengesellschaft | Demonstrating resistance to antibiotics in microorganisms |
| WO2004007490A3 (en) * | 2002-07-16 | 2004-03-25 | Cadila Healthcare Ltd | A process to prepare pioglitazone via several intermediates. |
| BG65351B1 (en) * | 1998-11-12 | 2008-03-31 | Smithkline Beecham P.L.C. | Modified release pharmaceutical composition |
| EP1917962A1 (en) | 1998-11-12 | 2008-05-07 | Smithkline Beecham Plc | Pharmaceutical composition for modified release insulin sensitiser |
| EP2036554A1 (en) | 2000-06-19 | 2009-03-18 | SmithKline Beecham plc | Combinations of dipeptidyl peptidase iv inhibitors and other antidiabetic agents for the treatment for diabetes mellitus |
| EP2266576A1 (en) | 1997-06-18 | 2010-12-29 | SmithKline Beecham Limited | Treatment of diabetes with thiazolidinedione and sulphonylurea |
| US7976871B2 (en) | 2002-08-05 | 2011-07-12 | Torrent Pharmaceuticals Limited | Modified release composition of highly soluble drugs |
| US8268352B2 (en) | 2002-08-05 | 2012-09-18 | Torrent Pharmaceuticals Limited | Modified release composition for highly soluble drugs |
| US8993773B2 (en) | 2002-07-16 | 2015-03-31 | Cadila Healthcare Limited | Process to prepare pioglitazone via several novel intermediates |
| WO2016071727A1 (en) | 2014-11-04 | 2016-05-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for the prevention and the treatment of rapidly progressive glomerulonephritis |
| US9579335B2 (en) | 2010-10-19 | 2017-02-28 | Indiana University Research And Technology Corporation | Treatment of cystic diseases |
| CN106470991A (en) * | 2014-04-02 | 2017-03-01 | 米尼奥尔克斯治疗有限公司 | 2,4‑Thiazolidinedione derivatives in the treatment of central nervous system disorders |
| CN106478620A (en) * | 2016-09-18 | 2017-03-08 | 浙江华海药业股份有限公司 | A kind of preparation method of pioglitazone impurity |
| US11938122B2 (en) | 2016-12-01 | 2024-03-26 | Minoryx Therapeutics S.L. | 5-[[4-[2-[5-(1-hydroxyethyl)pyridin-2-yl]ethoxy]phenyl]methyl]-1,3- thiazolidine-2,4-dione for treating nonalcoholic fatty liver disease |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR1365278A (en) * | 1962-08-09 | 1964-07-03 | Roussel Uclaf | New microbiological alkaloid oxidation process |
| EP0257781A1 (en) * | 1986-07-24 | 1988-03-02 | Takeda Chemical Industries, Ltd. | A method for producing thiazolidinedione derivatives |
| EP0508740A1 (en) * | 1991-04-11 | 1992-10-14 | Takeda Chemical Industries, Ltd. | Thiazolidinedione derivatives, their production and their use |
| WO1992018501A1 (en) * | 1991-04-11 | 1992-10-29 | The Upjohn Company | Thiazolidinedione derivatives, production and use thereof |
-
1993
- 1993-04-21 WO PCT/US1993/003569 patent/WO1993022445A1/en not_active Ceased
- 1993-04-21 AU AU41045/93A patent/AU4104593A/en not_active Abandoned
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR1365278A (en) * | 1962-08-09 | 1964-07-03 | Roussel Uclaf | New microbiological alkaloid oxidation process |
| EP0257781A1 (en) * | 1986-07-24 | 1988-03-02 | Takeda Chemical Industries, Ltd. | A method for producing thiazolidinedione derivatives |
| EP0508740A1 (en) * | 1991-04-11 | 1992-10-14 | Takeda Chemical Industries, Ltd. | Thiazolidinedione derivatives, their production and their use |
| WO1992018501A1 (en) * | 1991-04-11 | 1992-10-29 | The Upjohn Company | Thiazolidinedione derivatives, production and use thereof |
Non-Patent Citations (2)
| Title |
|---|
| ARZNEIMITTEL FORSCHUNG. DRUG RESEARCH vol. 40, no. 1, 1990, AULENDORF DE pages 37 - 42 T. SOHDA ET AL. 'Studies on antidiabetic agents. Synthesis and hypoglycemic activity of 5-[4-(pyridylalkoxy)benzyl]-2, 4-thiazolidinediones.' * |
| DATABASE WPI Section Ch, Week 8737, Derwent Publications Ltd., London, GB; Class B02, AN 87-260936 * |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2266576A1 (en) | 1997-06-18 | 2010-12-29 | SmithKline Beecham Limited | Treatment of diabetes with thiazolidinedione and sulphonylurea |
| WO1999061660A1 (en) * | 1998-05-22 | 1999-12-02 | Creatogen Aktiengesellschaft | Demonstrating resistance to antibiotics in microorganisms |
| US7005257B1 (en) | 1998-05-22 | 2006-02-28 | Seapro Theranostics International | Detection of antibiotic resistance in microorganisms |
| BG65351B1 (en) * | 1998-11-12 | 2008-03-31 | Smithkline Beecham P.L.C. | Modified release pharmaceutical composition |
| EP1917962A1 (en) | 1998-11-12 | 2008-05-07 | Smithkline Beecham Plc | Pharmaceutical composition for modified release insulin sensitiser |
| EP1992337A1 (en) | 1998-11-12 | 2008-11-19 | Smithkline Beecham Plc | Novel modified release composition and use |
| EP2036554A1 (en) | 2000-06-19 | 2009-03-18 | SmithKline Beecham plc | Combinations of dipeptidyl peptidase iv inhibitors and other antidiabetic agents for the treatment for diabetes mellitus |
| US8173816B2 (en) | 2002-07-16 | 2012-05-08 | Cadila Healthcare Limited | Process to prepare pioglitazone via several novel intermediates |
| US8993773B2 (en) | 2002-07-16 | 2015-03-31 | Cadila Healthcare Limited | Process to prepare pioglitazone via several novel intermediates |
| US7465801B2 (en) | 2002-07-16 | 2008-12-16 | Cadila Healthcare Limited | Process to prepare pioglitazone via several novel intermediates |
| US7863300B2 (en) | 2002-07-16 | 2011-01-04 | Cadila Healthcare Limited | Process to prepare pioglitazone via several novel intermediates |
| US7671207B2 (en) | 2002-07-16 | 2010-03-02 | Cadila Healthcare Limited | Process to prepare pioglitazone via several novel intermediates |
| WO2004007490A3 (en) * | 2002-07-16 | 2004-03-25 | Cadila Healthcare Ltd | A process to prepare pioglitazone via several intermediates. |
| US8445685B2 (en) | 2002-07-16 | 2013-05-21 | Cadila Healthcare Limited | Process to prepare pioglitazone via several novel intermediates |
| US8268352B2 (en) | 2002-08-05 | 2012-09-18 | Torrent Pharmaceuticals Limited | Modified release composition for highly soluble drugs |
| US8216609B2 (en) | 2002-08-05 | 2012-07-10 | Torrent Pharmaceuticals Limited | Modified release composition of highly soluble drugs |
| US7976871B2 (en) | 2002-08-05 | 2011-07-12 | Torrent Pharmaceuticals Limited | Modified release composition of highly soluble drugs |
| US9579335B2 (en) | 2010-10-19 | 2017-02-28 | Indiana University Research And Technology Corporation | Treatment of cystic diseases |
| CN106470991A (en) * | 2014-04-02 | 2017-03-01 | 米尼奥尔克斯治疗有限公司 | 2,4‑Thiazolidinedione derivatives in the treatment of central nervous system disorders |
| US9782395B2 (en) | 2014-04-02 | 2017-10-10 | Minoryx Therapeutics S.L. | 2,4-thiazolidinedione derivatives in the treatment of central nervous system disorders |
| US10179126B2 (en) | 2014-04-02 | 2019-01-15 | Minoryx Therapeutics S.L. | 2,4-thiazolidinedione derivatives in the treatment of central nervous system disorders |
| CN106470991B (en) * | 2014-04-02 | 2020-07-31 | 米尼奥尔克斯治疗有限公司 | 2, 4-thiazolidinedione derivatives in the treatment of central nervous system disorders |
| WO2016071727A1 (en) | 2014-11-04 | 2016-05-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for the prevention and the treatment of rapidly progressive glomerulonephritis |
| CN106478620A (en) * | 2016-09-18 | 2017-03-08 | 浙江华海药业股份有限公司 | A kind of preparation method of pioglitazone impurity |
| US11938122B2 (en) | 2016-12-01 | 2024-03-26 | Minoryx Therapeutics S.L. | 5-[[4-[2-[5-(1-hydroxyethyl)pyridin-2-yl]ethoxy]phenyl]methyl]-1,3- thiazolidine-2,4-dione for treating nonalcoholic fatty liver disease |
Also Published As
| Publication number | Publication date |
|---|---|
| AU4104593A (en) | 1993-11-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR930010705B1 (en) | Method of manufacturing FR-900520 material | |
| EP2514828B1 (en) | Process for preparing purified ansamitocins | |
| TWI291464B (en) | Methods for the preparation, isolation and purification of epothilone B, and X-ray crystal structures of epothilone B | |
| WO1993022445A1 (en) | A process for producing pioglitazone metabolite | |
| JPH03201992A (en) | Novel method of preparing immunosuppressive agent | |
| JPH06339390A (en) | BU-4164E-A and B, prolyl endopeptidase inhibitors | |
| JPH0216756B2 (en) | ||
| JP4521145B2 (en) | Antibiotic caprazomycin and its production | |
| US5290772A (en) | Immunosuppressant agent | |
| EP0253413B1 (en) | New antibiotics called "mureidomycins a, b, c and d" a process for their preparation and their therapeutic use | |
| US5189150A (en) | Oasomycins | |
| EP1372640B1 (en) | Use of thiolutin dioxide and its derivatives in the manufacture of a medicament for the treatment of cns disorders | |
| US4835141A (en) | Neutral macrolide antibiotics from Streptomyces | |
| Johnson et al. | Microbiological N-deoxygenation and C-oxygenation of pioglitazone-N-oxide in a single fermentation | |
| AU2012201869B2 (en) | Methods for the production of ansamitocins | |
| JPH05331171A (en) | New metabolic product obtained from pseurochin f1/f2 aspergillus fumigatus, preparation thereof and its use | |
| US4753959A (en) | Antibiotic lactone compound | |
| JPH08239379A (en) | Kr 2827 derivative as new substance, its production and use | |
| US4540575A (en) | Choline oxidase inhibitor | |
| JP2716728B2 (en) | Novel microorganism and method for producing 9-keto 16-membered macrolide antibiotics | |
| JPH0710857A (en) | Novel aldose reductase inhibitor | |
| JPH08176157A (en) | Novel bioactive substance epostatin, method for producing the same and use thereof | |
| JPH0525150A (en) | New decaloess tricutins and related compound, preparation thereof and use thereof | |
| CA2007679A1 (en) | Microbial transformation product | |
| JPH06256275A (en) | Antiplatelet drug |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AT AU BB BG BR CA CH CZ DE DK ES FI GB HU JP KP KR LK LU MG MN MW NL NO NZ PL PT RO RU SD SE SK UA US VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| 122 | Ep: pct application non-entry in european phase | ||
| NENP | Non-entry into the national phase |
Ref country code: CA |