WO1984004745A1 - Novel polypeptides and their use - Google Patents
Novel polypeptides and their use Download PDFInfo
- Publication number
- WO1984004745A1 WO1984004745A1 PCT/JP1983/000174 JP8300174W WO8404745A1 WO 1984004745 A1 WO1984004745 A1 WO 1984004745A1 JP 8300174 W JP8300174 W JP 8300174W WO 8404745 A1 WO8404745 A1 WO 8404745A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- arg
- hours
- human
- ser
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6863—Cytokines, i.e. immune system proteins modifying a biological response such as cell growth proliferation or differentiation, e.g. TNF, CNF, GM-CSF, lymphotoxin, MIF or their receptors
- G01N33/6866—Interferon
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/52—Cytokines; Lymphokines; Interferons
- C07K14/555—Interferons [IFN]
- C07K14/57—IFN-gamma
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
- C07K16/249—Interferons
Definitions
- the present invention relates to novel polypeptides and uses thereof.
- IFN human interferon
- ⁇ r-type interferon
- I-IFN immune interferon
- IFN-r properties include different antigenicity from type a interferon (IFN-a) and type 3 interferon (IFN- / 9), and different types of inducers. It is also known that the stability of foams, acid, and heat is poor (The Interferon System, Sp !; Nanger, New York, 1979).
- IFN is defined as having an anti-viral effect produced by living organisms, but it has also been demonstrated that it has many other biological activities i) (Blood, Vol. 55, p. 71, 1980; Ibid., Vol. 56, p. 875, 1980).
- Methods for suppressing tumor growth include a method of directly suppressing the growth of tumor cells and a method of suppressing the tumor indirectly through the host's immune response.
- a method of directly suppressing the growth of tumor cells for example,
- NK Bacha killer cells
- Activation of macrophages or activation of killer T cells.
- IFN has been proved to have various immunopotentiating activities in addition to its direct action (Biochemica et al., Vol. 516, p.231, 1978).
- FN- r has much higher antitumor activity at INVIT and antitumor activity at in vivo than IF-a and IF! T- / 3 Its importance has been strongly pointed out (Cellular Immunology, Vol. 49, p. 390, 1980).
- IFN-r induced in the mouth is generally low or appropriate. ⁇ ⁇ ⁇ ⁇ Most IFN-r-producing cell lines are difficult to purify due to poor heat and acid stability. Mass production and purification of IFN-T lags far behind IFN-a and IFN-i9 I have.
- the present inventors performed cloning of the IFN- ⁇ gene based on the mBNA of IFiT-, which was derived from human peripheral blood lymphocytes, and clarified its nucleotide sequence.
- a mammal was immunized with the polypeptide or the protein complex obtained here, and the extracted ⁇ ⁇ cells were fused with allogeneic or heterologous lymphoid cells by cell fusion.] The hybridoma was cloned. .
- the obtained hybridoma was inoculated into a mammal to produce a monoclonal antibody, which was collected to produce a monoclonal antibody against the above-mentioned polypeptide.
- a method for purifying human IFN- r from crude human IFN-I content using the nocloclonal antibody obtained here a radioimmunoassay (RIA) method, and a radiobiology enzyme immunoassay (EIA) method
- the present invention provides a novel polypeptide (I), a protein complex thereof, a novel cloned hybridoma, a novel monoclonal antibody, a method for producing the same, and a use of the monoclonal antibody.
- Y is Arg Arg Ala Ser Gin with respect to the polypeptide (I), and!
- X is preferably Lys Arg.
- polypeptide (I) is not a specific number of amino acid residues.
- the peptide can be produced by conventional means of peptide synthesis. Either the solid phase synthesis method or the liquid phase synthesis method may be used, but the liquid phase synthesis method is advantageous in many cases.
- the method for synthesizing such a peptide is described, for example, in “The Peptides-, Vol. 6 6), Schroder and Lbke, Academic Press, New York, USA, or "Peptide Synthesis", Izumiya et al., Maruzen Co., Ltd. (t975), al. , Pp. 207-400 "Haruaki Yajima, Tokyo Kagaku Dojin Co., Ltd. ⁇ 977.
- J Examples include azide method, chloride method, acid anhydride method, mixed acid anhydride method, DCc method, and activity.
- Esthetic method method using wood reagent E, carbodimidazo method, redox method, DC CZ active (eg, HOWS, HOBt, HOSu) method.
- the peptide has a reactive carboxy group corresponding to one of two types of fragments that are bisected at an arbitrary position in the peptide bond, and a reactive amino group corresponding to the other fragment.
- Examples of the protecting group for the amino group of the raw material include canolepobenzoxy, t-butynoleoxycanoleboninole, t-aminole; , 2 black
- the protecting group for the carboxyl group include, for example, akiester (eg, meth, eth, propyl, buty, t-butyl ester group), pendi est group, pout benzene V est group, "0— Methoxy pent diester group, one mouth; 1 pentyl 1 / ester / 1 group, penzhydri ester group, force; bopenzoxy hydrazide group, 1-butoxy carbazyl hydrazide group, triti / hydrazide group Can be
- Protecting groups for the arginine guadino tomb include, for example, nitro, tol, p-methoxybenzenesulfonium, kabobenzoki ⁇ , isoboni, and adaman.
- Tiloxycarb; / t, 4-Methoxy 2,6-dimethylbenzenesulfonol group, pentamethynolensens / U-hole group and the like.
- the guadino group of ⁇ may be protected in the form of an acid (eg, venense; Hongjun, tonosulfonic acid, hydrochloric acid, sulfuric acid, etc.) salt.
- the hydroxyl group of threonine can be protected, for example, by esterification or esterification.
- Suitable groups for this esterification include, for example, groups derived from carbonic acid such as acetyl group, lower alkano group, penzoi group, alkaro group, benzyloxycarbo-nore group, ethynoleoxycanorebo-group and the like.
- groups suitable for 3- or etherification, such as benzyl include benzyl, tetrahydroxy-, and 7-butyl groups.
- Methione may be protected in the form of a sulfoxide.
- Examples of the activated carboxyl group in the raw material include, for example, the corresponding chrysanthemic anhydride, azide, and hot-pottenol (pentachlorophenol, p--trophenol) Nore, IT-Hydroxysuccinimide, N-Hydroxydzintriazole, N-zdoxydoxy 5 -Nonolevo ⁇ nene-2, 3-Zikaboxymidido, etc.) can give.
- the peptide bond formation reaction may be carried out in the presence of a dehydrating agent (eg, a carbodiimide reagent such as dicyclohexylcarbodimid or cabodimidazo).
- the present peptide condensation reaction can be performed in the presence of a solvent.
- the solvent can be appropriately selected from those known to be usable in peptide coupling reactions.
- the reaction temperature is appropriately selected from a range known to be applicable to the peptide bond formation reaction, and is usually about -4 Ot to about 60, preferably about
- -20 is appropriately selected from the range of about 0.
- Such conventional methods include, for example, reductive methods (eg, hydrogenation using a catalyst such as palladium black, reduction with metal sodium in a liquid ammonia), and acid lysis (eg, triflic acid, fluoride).
- reductive methods eg, hydrogenation using a catalyst such as palladium black, reduction with metal sodium in a liquid ammonia
- acid lysis eg, triflic acid, fluoride.
- sulfur-containing compound such as hydrogen, methanesulfonic acid, or thioazole
- the peptide of the present invention produced as described above can be collected from the mixture after completion of the reaction by ordinary means for separating peptides, extraction, distribution, column chromatography, and the like.
- the carrier' and the mixing ratio of carrier and hapten depend on the carrier. If an antibody can be efficiently produced against the hapten immunized by priming, any hapten may be coupled at any ratio.
- any hapten may be coupled at any ratio.
- bovine serum albumin, bovine thyroglobine, etc. may be used in a weight ratio. In this case, a method is used in which a force is applied to the hub ten at a rate of 0.1 to 20, preferably 1 to 5.
- mammals to be immunized include experimental animals such as sheep, goats, rabbits, mo / remots, rats, and mice.
- a rat mouse is preferable.
- immunizing a mouse it is possible to immunize a mouse subcutaneously, intraperitoneally, intravenously, intramuscularly, intradermally, etc.
- inject Prefer to inject.
- the immunity interval, immunity amount, etc. are also highly variable, and various methods are possible.
- immunization is performed 2 to 6 times at 2 week intervals, and 1 to 5 days after the final immunization, preferably 2 to 4 times. A method using cells after days is often used.
- the amount of immunization should be the amount of peptide at a time, per mouse]? 0.1 g or more, preferably! It is preferable to use 0 g to 300 g. Also, before ⁇ the ⁇ , partial blood collection line, after confirming the increase of antibody titer in blood, Shi desirable to perform fusion experiments with ⁇ cells 3
- the above-mentioned saturating fusion between the ferocytic cells and the lymphoid cells can be performed, for example, by excising the extracted mouse cells from hypoxanthine-guanine-phosphoribosyltransferase deficiency (HGBBT ”) or thymidine kinase-deficient (K-).
- HGBBT hypoxanthine-guanine-phosphoribosyltransferase deficiency
- K- thymidine kinase-deficient
- a fusion agent such as lietin glyco-nore (PEG) is used.
- PEG lietin glyco-nore
- PMSO dimethisoxide
- the polymerization degree of PEG is usually 100 to 6000, the time is 0.5 to 30 minutes, and the concentration is 10 ⁇ -80.
- the cells can be fused at a high impeachment rate by treating them for 4 to 10 minutes at ⁇ 55 to 6.
- the fused cells can be grown in a hypoxanthine-aminopterin-thymidine medium (HAT medium; Nayya, Vol. 256, (P. 495, p. 1975) can be used to selectively proliferate.
- HAT medium hypoxanthine-aminopterin-thymidine medium
- the culture supernatant of the cultured cells can be screened for the production of the desired antibody, but the antibody titer can be screened as follows. I can do it. That is, in this case, the presence or absence of antibody production against the immunized peptide can be first examined as a first step by a method such as the BIA method or the EIA method, but various modifications of these methods are also possible. It is possible. As an example of a preferable measurement method, one method using EIA will be described.
- a carrier such as cellulos beads is coupled with, for example, a persimmon anti-Macsimiminoglobulin antibody according to a conventional method, and the measured culture supernatant or mouse serum is added thereto, and the mixture is incubated at a constant temperature for a certain period of time. Less than
- reaction product is thoroughly washed, and an enzyme-labeled peptide (purified after force-purifying the enzyme and the peptide according to a conventional method) is added, and the reaction is carried out at a constant temperature for a certain period of time.
- enzyme substrate is added, and the reaction is allowed to proceed at a constant temperature for a certain period of time. Then, the resulting color product can be measured by absorbance or fluorescence.
- -Capsule is added to a carrier such as beads according to a conventional method, and the hybridoma supernatant or mouse blood to be measured is added thereto and reacted at a constant temperature for a certain period of time. After that, the reaction mixture is added to the reaction solution in a constant amount.
- a recombinant containing IFK-r derived from human peripheral blood lymphocytes with lectin and phorbo este etc.
- ⁇ - IFH-r which can be used as a solution, react at constant temperature for a certain period of time, and then determine the activity of IFr contained in the reaction. In this way, the limb yield of IF-r activity of the target antibody can be measured.
- Hybridomas cloned as described above can be used, for example, in a greedy medium, such as BPMI-164, medium supplemented with 0.1 to 40% bovine serum for 0 days, preferably 3 to 40%. 5 3 times, the culture solution
- a greedy medium such as BPMI-164, medium supplemented with 0.1 to 40% bovine serum for 0 days, preferably 3 to 40%. 5 3 times, the culture solution
- the monoclonal antibody can be obtained from the above, but in addition to the above, by inoculating intraperitoneally into a suitable mammal such as a mouse, and culturing the cell, and collecting ascites, the cell culture can be performed much more. It is possible to efficiently obtain a large amount of crab high titer antibody.
- a suitable mammal such as a mouse
- Also connexion inoculated before Minera old I and the like preferably 5's 1 0 5 - 2 I 0 6 Invite the hive 9-doma into the SC cavity, etc., and collect ascites narrow 7 to 20 days later, preferably 10 to 4 days later.
- Antibodies produced and accumulated in ascites fluid are collected, for example, by Bowan's fraction, DEAE-cellose column chromatography, etc.! ;), The antibody can easily be isolated as pure immunoglobulin.
- the Noclo-na antibody of the present invention has the following properties.
- This monoclonal antibody can be safely manufactured, used and stored O
- the obtained monoclonal antibody can be obtained by utilizing the EIA method or the B.IA method], and the amount of FN in the living body or in a test tube can be increased.
- the production of antibody chromosomes has made natural or genetic
- the resulting IFN-r can be used to purify very efficiently.
- E PN - for detection of r as an example, the measured I 5 1 7 * previously including materials or standard IF N-r, e.g., radioactive ® -?
- the monochrome - By the de or enzyme etc. j labeled the monochrome -After reacting with antibody and then precipitating an immune complex with protein A, for example, and reacting for a certain period of time, measure the radioactivity or Xe activity of the immunoprecipitate to easily reduce the amount of DiFir-r. Can be measured.
- the competition method using an antigen for example, the polypeptide (I) is radioactively or enzymatically labeled, and when a certain amount of the unlabeled monoclonal antibody of the present invention is reacted, the measurement is performed.
- IFN-r co-existing material containing IFN-r was used to precipitate the immune complex.] Or by binding to an anti-mouse antibody! ?, IF5-r can be quantified.
- purified I Fir- r can be used instead of polypeptide (I).
- a fixing method for example, I i ⁇ ⁇ -r or polybutide (I) is added to a phosphoric acid solution containing 0.1 ⁇ weight and nou to 0.,
- the purified antibody is coated on a suitable carrier such as activated agarose beads according to a conventional method, and then applied to a column.
- a suitable carrier such as activated agarose beads according to a conventional method
- Material containing crude I-r such as culture supernatant or disrupted cells is applied to the column, washed after adsorption, and then washed with a pick-up reagent such as ⁇ sc ir or IFN-r
- a pick-up reagent such as ⁇ sc ir or IFN-r
- the antibody column can be prepared, for example, by force-purifying the monoclonal antibody of the present invention purified from ascites or the like inoculated with the hybridoma with an appropriate carrier], and the following methods. .
- Any carrier can be used, as long as IFN-r is specifically and efficiently attached after cou- pling, and then can be appropriately eluted.
- Activated agarose beads such as Affige-10 are conveniently used in the manner described below.
- the reaction between Affige-10 and the antibody is carried out in a buffer solution of 0.001-1 M, preferably 0.1 M, such as biforce-bonate. Reaction conditions are 0 ⁇ ⁇ 20,! Various values of ⁇ ⁇ are possible from 0 minutes to 24 hours, but preferably, the conditions of 4 * C, 4 hours, and pH 3 to 10 are used.
- the amount ratio of Abuigel-10 and antibody to be mixed is such that the amount of antibody
- Antibodies of up to 50 W are more likely to be repelled as much as possible. Therefore, any antibody within this range may be used, but an antibody of 30 or less is preferably used in consideration of the r ratio.
- the antibody-carrier conjugate thus obtained is thoroughly washed with the buffer used for the reaction, and then left for a few days or at a final concentration of 0.05 M.
- O PI Use a method such as adding Tanoamine / hydrochloric acid and reacting with a rock for 1 hour. D. After blocking the remaining unreacted active groups, fill them with an appropriate solution], use as antibody column it can.
- a sample containing human immunogenic protein is dissolved in a buffer solution near neutral, for example, phosphate buffer solution, tris-hydrochloric acid buffer solution, and adsorbed to the antibody column. .
- a buffer solution near neutral for example, phosphate buffer solution, tris-hydrochloric acid buffer solution
- IFN-r is eluted.
- the solution include a weakly acidic solution such as an acetic acid solution, a solution containing polyethylene glycol, a solution containing ⁇ butide, which is more easily D-bound to the antibody than the sample, a high-purity salt solution, and a combination thereof. It is preferably used to promote the degradation of human IFN-IFN]).
- the column eluate is neutralized with a buffer according to a conventional method. If necessary, the above-described purification operation using the antibody force column can be performed again.
- the human I-IFIT protein solution obtained here is subjected to dialysis, and if necessary] this can be made into a powder by freeze-drying].
- human immune Intafe port down protein shows a 1 0 7 UZ more specific activity in WE scan activity measurement by cytopathic effect deter test vesicular stomatitis Huy scan (VSV) on human amnion-derived WISH cells .
- VSV vesicular stomatitis Huy scan
- the method of producing U ZW (cut ⁇ ) as the activity of IFN was performed as follows.
- the international standard IFN- ⁇ and the crude IFN-r derived from leukocytes were determined using the VSV cytopathic effect inhibition test on FL cell line derived from human amniotic membrane, and the titers were compared.
- leukocyte-derived crude IFN-r were determined using the VSV cytopathic effect inhibition test on FL cell line derived from human amniotic membrane, and the titers were compared.
- the efficiency IFN-r was always arranged, and the results were calculated using the WISH-VSV system described above, and the titer was calculated from the ratio.
- the D-purified human immune interface protein can be obtained by conventional methods.
- the human I-IFN protein of the present invention has anti-inoles, anti-tumor, cell growth inhibitory and immunopotentiating effects.
- the human I-IFN protein of the present invention can be mixed with sterilized water, human serum albumin (HSA), physiological saline or other known physiologically acceptable carriers, and can be administered parenterally or topically. it can.
- HSA human serum albumin
- a normal person can receive 100,000 to 100 million ut, preferably 50 to 60 million ut, by intravenous injection or intramuscular injection.
- the preparation containing the human I-IFN protein of the present invention may contain other physiologically acceptable activities such as salts, diluents, adjuvants, other carriers, buffers, binders, surfactants, and preservatives. Components may also be included. Preparations for parenteral administration may be sterile aqueous solutions or suspension ampoules in physiologically acceptable solvents, or sterile powders (usually I-I), which can be used after dilution in physiologically acceptable diluents. It is provided as an amplifier (obtained by freeze-drying FN solution).
- the above-mentioned preparation containing the human immune interferon protein contains 1 to 99 other active ingredients relative to the substance of the present invention, such as IFN- ⁇ or IFN- (9 or interleukin 2 or any other lymphokine). May be.
- a P Adenosine triphosphate
- AeOEt firewood acid
- Fig. 1 shows the elution pattern of the monoclonal antibody of the present invention on the DEAE cellose column
- Fig. 2 shows the elution pattern of the monoclonal antibody of the present invention. 0 indicating the result
- FIG. 3 shows the results of the electrophoresis of the human immunointerferon-feron protein obtained by the purification method of the present invention shown after Example 13, and FIG. 4 shows the molecular weight variation similarly shown after Example i3.
- Figure 5 shows the primary structure (Tong group arrangement) of pHIT 3709 plasmid obtained in Reference Example II (V8)
- Figure 6 shows the reference structure 2)
- Figure 7 shows the reference example 2 (iii) pHIT trp 1101
- Fig. 8 shows the construction of Reference Example 2 (iV) (pEIT cell rp 2101), and Fig.
- FIGS. 10 and 11 show (a) Escherichia coli extract containing or not containing IFN-r and (b) enzyme against the Monica-Nanole antibody according to Examples 20- (i) and 20- (ii), respectively. 1 shows the competition with the paired peptide of the present invention which was recognized. --Best mode for carrying out the invention
- thin-layer chromatography was performed using the following developing solvents, using Mek's Sili-Ige Plate 60 F £ 54 or Funakoshi Pharmaceutical's Cell-Port-Splate, Abysse SF. Was.
- the black was reduced as a catalyst in a stream of hydrogen.
- the catalyst was removed by filtration and the solvent was distilled off.
- Z-A 1 a-S ⁇ rG 1 ⁇ -0 ⁇ u 43 ⁇ 46 was dissolved in methanol 300 W and reduced with palladium black as a catalyst in a hydrogen stream.
- the catalyst was removed by filtration, and the residue obtained by removing the solvent was prepared from Z-Arg (Pme) -OH-CHA 4.549.
- Z-Gl OBu 7 13.0 was converted to MeOH 500: and catalytically reduced in a stream of hydrogen using black as a catalyst. The solvent was separated off, the aqueous solution was decompressed and concentrated and the residue was dissolved in DMF200W.
- Z-Arg (me) -Gly-OB ⁇ 1Q.0 was catalytically reduced in MeOH 500 W, and then transferred to DMF300 ⁇ J. This with Z-: Plie-OH 4.72,
- Boc-Me 7-11 0H.DCEA 3.0 Add the prepared Boc Me Shichiichi OH, H0 1.39 ⁇ , cool on ice, add Dcc 1.59, stir for 15 hours, discriminate 3 folded DCs, concentrate, and concentrate BicQH- AcOE dissolved seven, 1 056 and washed with click E phosphate solution, then dried and concentrated in Na 2 S0 4, E -.. Tenore was ⁇ as crystals by adding yield 6.2 ⁇ (9 3.1%). mp. 1 9 2— ⁇ 95 3 C,
- Z-Ser-iTHlTH-Boc3 was transferred to DMF50O after the thigh was placed in MeOH300. To this, add Z-Arg (Pme) -OH, ⁇ 1.5 prepared from Z-Arg (Pme) -OE-CHA S.29], cool on ice,
- Z-Ar g (Pme) -Ser- ⁇ ⁇ - ⁇ oc 3.9 was subjected to catalytic reduction with MeOH 300 0 and then transferred to DMI “50”.
- Example ⁇ The combination of holibeptide obtained in ( ⁇ ) and Cycloglobulin (hereinafter G-) was performed according to the method of Goodfriend et al. (Science,
- the mixture was freeze-dried to obtain 4.7 W of the protein complex.
- the antigen for EIA was prepared according to the method of Kitagawa et al. (Janab Biochemistry, Vol. 79, p. 233, 1976).
- Antibody injection in serum or hybridoma supernatants immunized with the protein complex obtained in Example 2 was detected by the EIA method (Immunobu-Macologie, Vol. 1, p. 3, 19). 1978) to Wachi, serum some Haiburido - the Ma supernatant buffer - a (20 m M Na 2 HP0 4, 10 OmM -2-JTaCl, 0.1 Na3 ⁇ 4, 1 mM MgCl 2 , pH 7.0), diluted with 100 / ⁇ ])), the e obtained in Example 3. The mixture was thoroughly mixed with the peptide derivative 100, and was subjected to anti-JS at 24 for 24 hours.
- mice Each of 6 6-8 week-old BALBZC female mice was well mixed with Freund's complete adjuvant as a protein, and inoculated subcutaneously (primary immunization). Two weeks after the first immunization, the same amount of antigen was mixed well with Freund's complete adjuvant, and subcutaneously inoculated (secondary immunization). after 6 days of immunization rows Natsuta 3 tertiary immune, blood was partially collected from the mice, was boss measure the anti ⁇ in serum by EIA method real 3 ⁇ 4 example 4 described. Of these, r-2 mice with high antibody titers were inoculated intravenously with 120 g of antigen dissolved in 5 ⁇ of drinking water to achieve final immunization. anti- ⁇ value of each mouse are shown in Table 1 3
- Serum dilution is 1 ZS 300
- Immunization was performed by the method described in Example 5, and 3 days after the final immunization, the spleen was excised from the ⁇ 2 mouse, squeezed with a stainless steel mesh, filtered, and placed in an e-Gluz Minimum 'Etsencia Medium (MEM). The cells were suspended to obtain a cell suspension.
- a cell used for cell fusion BAL BZC mouse myeloma-ma cell P3-63.A 8. ⁇ ( ⁇ 31) was used (current topix-in-micro / kusaiguchi-g-and-immunology).
- spleen cells and 3: 1 each contain serum and are washed three times with MEM and mixed so that the ratio of the number of stored cells to the number of P 3 LM is 5: 1.
- HAT hypoxanthine ⁇ X 1 0 one 4 Micromax, Aminoputeri down 4 X 1 Cf '7 ⁇ , thymidine ⁇ .6 X ⁇ 0 "5) ⁇
- PBMI 1 6 40 - 1 0 FCS medium HA 0? Medium
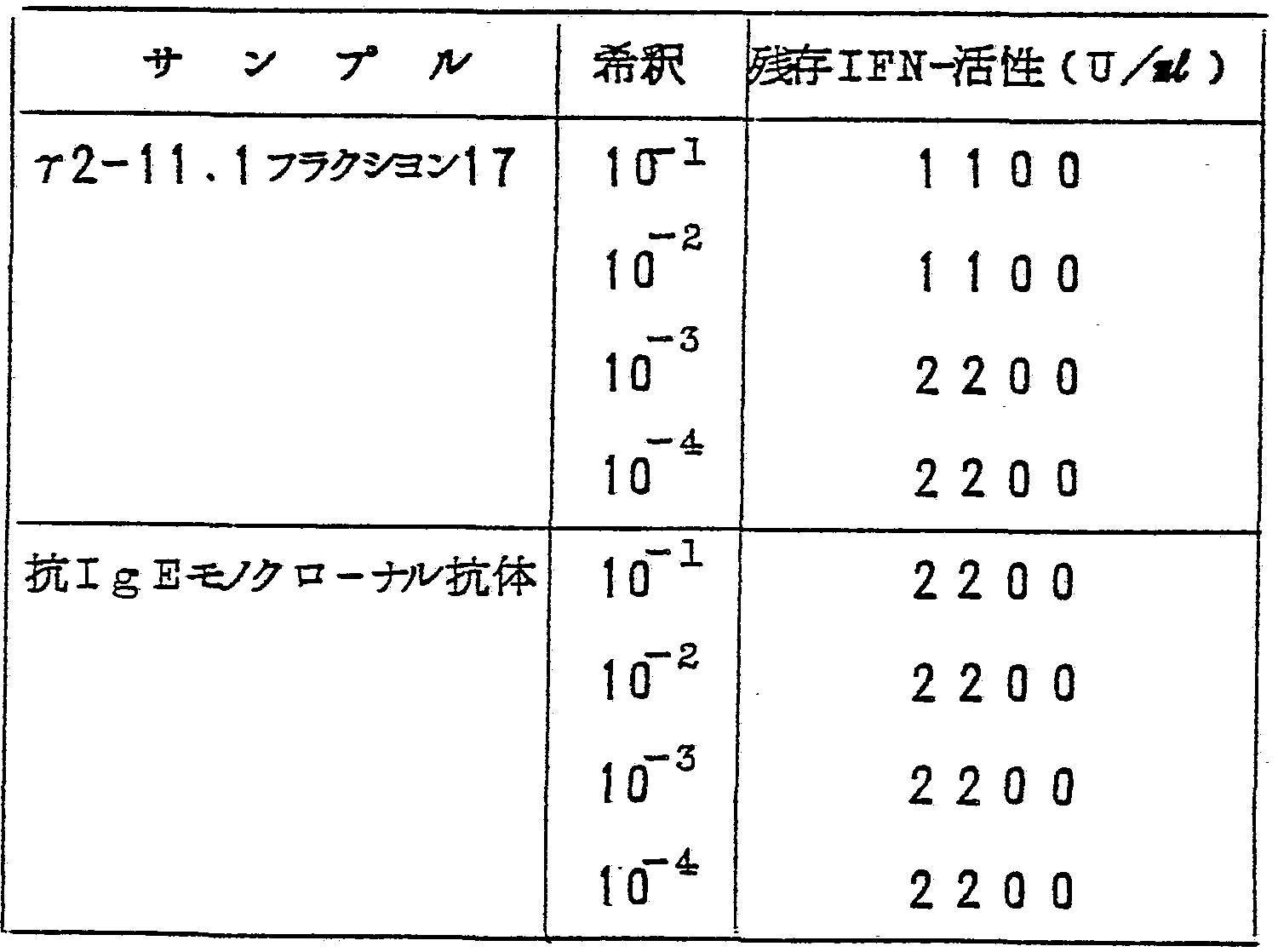
- Each of three hybridomas (a2-! 1, 62, 100) showing positive antibody activity was cloned by limiting dilution. That is, two hybridomas were suspended in HPMI1S40-20FCS so as to reach 2 cells / cell, and the suspension was dispensed in a 96-well microphone plate (manufactured by Nunc Corporation) at a ratio of 0.1 per 1 kg. At the time of segregation, B mouse thymocytes were added as puper cells to make up to 5 x 10 "cells. Thus, after about two weeks, cell proliferation was observed. The supernatant was probed and the presence or absence of the antibody was examined by the EA method described in Example 4. As a result, in r2-11, 8 clones out of 19 clones, r2- In 62, 3 out of 54 clones, and in r2-100, 5 out of 47 clones showed anti-honey injection (Table 2).
- the binding ability of the monoclonal antibody to IFH-r was examined by the following method. That is, a culture of 2 to 3 clones of r2-ll, r2-62 and r2--00 cells, respectively, was added to a three-sense solution 300 bound with a rabbit egan anti-mouse IgG antibody. Add 300 pieces of Kiyoshi at room temperature
- the reaction was performed for 18 to 20 hours. After the reaction, the cells were mixed well with physiological saline, and 550 cells of the IFN-a obtained in the following manner were added, and the mixture was reacted for 3 to 4 hours. After the reaction, the supernatant was collected, and the IFIT-r activity in the supernatant was measured by the cell transfusion effect (c) reading method using a microplate. (Applied Microbiology, Vol. 16, pp. 1706, 1968). That is, 50 ⁇ m was put into all the 96-well microplates (manufactured by Nunc Corporation), and IFFT sample was added to the first layer at 5 ⁇ m, followed by serial 2-fold dilution.
- the FN-r sample used was a human peripheral lymphocyte, which had a naphthalin A 40 / ul and a 12-0-tetradecanoinyl phorbo-1-13 acetate 1 2 hours after stimulation with 5 ng,
- the culture medium contained human cultures (acid, unstable in ⁇ ⁇ processing) and 4400 cuts. -If an antibody capable of binding to ⁇ -r is present in the culture supernatant of the cloned cells, IFIT-r activity added later binds to the antibody on cellose. In addition, the activity in the supernatant is reduced:> Yugo shows relatively strong IN- ⁇ binding ability in the supernatant of r2-111 clone, and the added IP IT -r (550
- BAL BZc mice that have been administered intraperitoneally with 1 x 10 ° clone cells in advance of mineral oil intraperitoneally, producing r 2-1 1.
- Antibodies capable of binding to IF ir -r By inoculating intraperitoneally I made it.
- the black - anti ⁇ in the culture supernatant of the emission cells are recognized to ⁇ 0 4-fold dilution, about 1 0 0 0 times antibody activity by to ascites rises.
- OMPI The IgG subclass to which the ⁇ 2- ⁇ -nocchi-null antibody belongs was 10-fold diluted the fraction 17 purified by the method of Example 10 to obtain goat anti-mouse IgG ⁇ , G2a, G2b, Precipitation reaction in agar with G3 antibody (Miles) (Octa-orchid-method: immunologica method., ⁇ ⁇ ? R Ge fusion fusion technique, black black, oxford,
- Supernatant 60 obtained in Reference Example 4 contains 1 mM EDTA and 0.15 M NaCl
- Example 13 The protein obtained in Example 13 was treated with 2-butane ethanol.
- the protein was identified as a single band when subjected to SDS-boriatari / u amide (17.5 *) electrophoresis (15 mV, 6 hours) and stained with Coomassie blue (FIG. 3). From the relationship between the migration distance of the molecular weight marker and the migration distance of the protein, which were simultaneously electrophoresed, the molecular weight of the protein was estimated to be 17,000 soil 1,000 (Fig. 4). On the other hand, another band was detected at a molecular weight of 33,000 ⁇ 2,000 for the protein that was not treated with 2-meadow ethanol. Since this value corresponds to about twice the molecular weight of IFN-r of 17,000 ⁇ 1,000, this band is considered to be derived from the IFN-r dimer.
- Example 13 Transfer the protein obtained in Example 13 to a glass hydrolysis test tube], add 200 volumes (V / W) of constant boiling constant hydrochloric acid containing 4 thioglycolic acid, and seal the tube under reduced pressure. After that, the mixture was hydrolyzed for 24, 48, and 72 hours with 110 watts. After hydrolysis, open the tube, remove the salt under reduced pressure, dissolve the residue in 0.02 N acid, and use Hitachi 8335 high-speed amino acid analyzer to perform the amino acid analysis. Carried out.
- Cystine and cystine were subjected to the formic acid oxidation of the above protein according to the method of Hirs [Methods EnzymoL 11, 197 (1967)], and then hydrolyzed for 24 hours in the same manner as described above to analyze amino acids. By all means]? Quantified as Sistine II. Amino acid analysis values were determined by averaging the values obtained after 24, 48 and 72 hours of hydrolysis. On the other hand, the values of serine, threon, tilidine and tributane were determined by extrapolating the hydrolysis time to 0 hours. Table 6 shows the results.
- Example 5 The protein obtained in Example 5 was used according to the method of Hirs [Methods EnzymoL 11,
- Example 6 Three days after the final immunization described in Example I-4, spleen was excised from the mouse, and cell fusion was performed and HAT selective culture was performed by the method described in Example 6. As a result, in March 43, hybridoma was found to be buried, and the presence or absence of antibodies was searched for by the EIA method described in Jeongseon-jo 4. The result was 2 ⁇ (r3-11, r3—19). In addition, strong antibody activity was observed.
- the binding ability of the monoclonal antibody to IFH-r was examined by the method described in Example 8. However, the 15 ⁇ ⁇ -r sambu used in this example was obtained by combining human IFN-transducer with brassmid and working on E. coli.
- ascites can be created by the method of *! ).
- the ascites obtained was purified by the method described in Example I, Example 0, and the obtained subclass of Monoclonal-anti-Fyle was defeated. .
- the results showed that a significant band was observed between goat anti-mouse Ig (Jl antibody) and that of both 7-3- -1.1 and r3-19-1.20 noclonal antibodies.
- this 100 was dispensed to each tube of a 96-well microphone plate, and reacted at 4 for 24 hours. After the reaction, dissolve 100 ⁇ l / volume of phosphate buffer containing 2 cow beabumin in 100 / ⁇ each to block the excess binding site of the gel, process at 4 C for 24 hours, and use it for ELISA.
- a mixture of the recombinant I PK— r prepared at 2 ⁇ 10 4 and 8 ⁇ 10 4 / W) was allowed to react at 37 ° C. for 3 hours, and reacted at room temperature for 3 hours.
- the well was washed well with phosphate buffer, 100 g of a goat anti-mouse igG antibody labeled with horseradish oxidase was added, and the mixture was reacted at room temperature for 3 hours.
- Example 20-1 Although the principle is the same as that of Example 20-1), a modified method under JK was carried out as a method for automatically and quickly measuring a large number of sambs using a multi-scan.
- test sample or standard IFH- ⁇ is adjusted to an appropriate concentration with buffered narrow A (0.02 M phosphoric acid buffer at pH 7 containing 0.1 M HaCl, 1 mM MgCl 2 , 0.1 Si BSA, 0.1 Na). Dilute and inject 75 into each hole of the plate prepared in (2). Next, 5 ⁇ of the enzyme sample of Example 3 diluted 30 times with buffer A is injected into each well of the plate. The reaction was carried out for 3 hours with the addition of a bulk. The reaction was continued at room temperature for 3 hours. After removing the reaction ⁇ , the plate was washed four times with 0.01 M phosphate buffer ⁇ (pH 7.4) 75.
- buffered narrow A 0.02 M phosphoric acid buffer at pH 7 containing 0.1 M HaCl, 1 mM MgCl 2 , 0.1 Si BSA, 0.1 Na.
- each well of the plate was prepared with buffer ⁇ A in each of the wells of 1.5 4 trofu-1- ⁇ -1D-galactobino s / do (Wako fine, special grade)
- Fig. 11 shows the standard curve of iFir-r.
- Table 10 includes i FH-r
- TSA 12-0-tetradecanoylhobo 13-acetate
- concanavalin A 40 ⁇ gZ *
- the cells were cultured in tfBPMl-l640 medium (including fetal bovine serum of ⁇ 0) for 3 hours to induce. 24 hours later, this induced one! 0 human lymphocytes were converted to a solution of guanidine (5 II guanidine, 5 liters of ethanol), 50 m ⁇ .
- the BU precipitate was dissolved in a 0.25 K-lauroyza / u-sodium sodium cosinate solution and precipitated with ethanol to obtain 8.3 W of BIfA. this! ?
- the NA was adsorbed on an oligo (dT) Senorelose column with a high salt solution [0.5 K NaC, 1 OmM Tris ⁇ EC £ pH 7.6, ImM EDTA, 0.3 SDS].
- a solution of mRNA containing poly (A) in a low salt solution (1 OmM Tr-HCl-pH 7.
- This mENA is further precipitated with ethanol, dissolved in a solution of ⁇ 2 ′′ (1 ⁇ Kris-HC1 pH 7. ⁇ , 2 mMED, 3% SDS), and treated with 65 for 2 minutes to obtain 1 Q to 35 % sucrose density gradient centrifugation (at 2 0 using a rotor of Beckman ST727, 25000rpm-C2 hour centrifugation) by the the child]? to obtain a fractionated by 2 2 fractions.
- a reaction solution of 10 O 5 g of mfNA, 50 ori '(dT), 100 units of reverse transcriptase, 1 dATP, 1 dCTP, dGTP and iTTP, 8 mM MgCl 2l 50 mM PC 1, 1 Q mM dithionite, 50 mM Tris-HC 1 pH 8.3) 42 * C Incubate for 1 hour, then extract with phenol Then, the cells were treated with 0.1 N NaOH at 70'C for 20 minutes to decompose and remove RNA.
- duplex 3 nuclease S 1 5 0 ⁇ a reaction solution NA (duplex]) NA 0.1 M Kokusan sodium ⁇ 4.5, 0.25 M NaCl, 1.521 ZnS0 4, 60 Interview - Tsu City of SI Nuclease) at room temperature for 30 minutes, deproteinize with phenol, precipitate DNA with ethanol, and add terminator transferase to 5 Q fig reaction mixture (double-stranded DMA, 0.14 M codylic acid).
- 0.3MTris (base) pS 7.6, 2 Jichiosureito 'ImM C 0 C1 2, 0.15 mM dCTP, 3 0 Interview - Tsu Tota!
- Bovine serum albumin 20 units of Pstl] was used for 3 hours at 37 t to excise one Ps11 recognition site in PBR322 DNA, deproteinized with phenol, and then terminated with terminal transferase. 5 0 / ⁇ ⁇ of reaction (:. DNA 1 0 a, 0.14M Kakoji acid strength Li, 0.3 M Tris (base) pH 7.6, 2 mM Jichiosurei toe, 1 mM C 0 C1 2 0.15mM dGTP, 3 0 Interview -Butardina transferase) for 3 minutes at 37 t! To extend about 8 deoxyguan chains at both ends of 3T of the above plasmid pBR322 DNA.
- ptrp701 was partially digested with Clal to eliminate one of the two restriction enzyme Clal cleavage sites present in ptrp701 to obtain a fragment in which only one of the two Clal cleavage sites was cleaved. .
- the resulting portion was filled in with DN JL volimase I page fragment and then religated with T4DNA ligase to obtain ptrp771 (FIG. 6).
- pHITtrp2101 was constructed.
- ptrp601 was treated with restriction enzymes Clal and Hpall to obtain a Clal-Hpa! I fragment 0.333 Kb containing a trp promoter. This fragment was cut with Clal and treated with ethanol phosphatase.
- Trp promoter and combined using T 4 DNA ligase to obtain a pHITtr P 2101 entered the two series 1 ((2l ⁇ ).
- Escherichia coli 294 was transformed with this plasmid ⁇ ⁇ trp2101 according to the method of Cohen et al.
- the cells were cultured in a Meyer at 37 t, and when the growth reached KU220, 3 ⁇ -indolino-inorea-clinoleic acid (IAA) was added to 30 S 1 / ⁇ , and the cells were further cultured for 4 hours.
- IAA 3 ⁇ -indolino-inorea-clinoleic acid
- the culture solution 1.2 obtained in Reference Example 3 was centrifuged to collect bacterial cells, and the cells were suspended in 0.05 Tris-HC1 pH 7.6 containing 60i of 10% sucrose.
- Add 2.4 « leave at 0 for 1 hour, treat with 3 7 for 5 minutes, and further apply ARTEK (USA) ultrasonic It was processed at 0'C for 30 seconds with the Xiao Xian.
- the lysate was centrifuged at 105,000x for 1 hour to collect the supernatant 66.
- the novel polypeptide of the present invention can be used to produce a monoclonal antibody that binds to a human interferon.
- the monoclonal antibody is useful for detection and purification of human ⁇ -type interferon.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- Organic Chemistry (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Medicinal Chemistry (AREA)
- Biomedical Technology (AREA)
- Hematology (AREA)
- Cell Biology (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Urology & Nephrology (AREA)
- Pathology (AREA)
- Biotechnology (AREA)
- General Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Microbiology (AREA)
- Food Science & Technology (AREA)
- Toxicology (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Abstract
Description
明 細 書 Specification
, 新規ホ。リベプチドおよびその用途 , New e. Ribeptide and its uses
技術分野 Technical field
本発明は、新規ポリペプチドおよびその用途に関する。 The present invention relates to novel polypeptides and uses thereof.
背景技術 Background art
ケーラーとミ スタイ ンによ 開発され(ネイチヤー ,第 256卷, 495頁, 19 75年)、 近年盛んになって来たハイプリ ドーマを用い たモノクローナル抗体の製造法は、各々の抗原決定基に対し、単一特異 性を示す抗体が得られることや、粗精製標品に対して得られた抗体につ いて吸収操作を必要とし¾いこと ¾どのすぐれた特徴をもっている。 ま たこの他に、抗体取得という面からも、ハイ プリ ドーマの腹水化によ , 高力価の抗体が、 自由に、 多量に、 しかも常に均質 標品を再現性よく 得られる ど多くの利点がある。 この様 意味からハイプリ ドーマによ るモノクローナ 抗体取得の手法は、多方面にわたってその有用性が高 く評価されている。 また、 その使い方も単に抗原検出にとどまらず、抗 体力ヲム作製を通じて、 微盪成分の精製に用いた]) (ネイチヤー,第 Developed by Koehler and Misstein (Nature, Vol. 256, p. 495, 1975), the method of producing monoclonal antibodies using hybridomas, which has become popular in recent years, has been developed for each antigenic determinant. In addition, antibodies that exhibit monospecificity can be obtained, and antibodies obtained from crudely purified samples require an absorption procedure. In addition, from the aspect of antibody acquisition, many advantages such as the ability to obtain high-titer antibodies freely, in large quantities, and with high reproducibility of a homogeneous sample with good reproducibility by ascites of the hybridoma. There is. In this sense, the usefulness of the method for obtaining monoclonal antibodies by hybridomas has been highly evaluated in many fields. In addition, its usage was not limited to antigen detection, and was used to purify the micro-agitated components through the production of an antibody resistance arm.])
' 285卷, 4.46頁, 1 980年)、更には、診断薬,治療薬への応用 ( ョ一口ビアン ジャーナノレ ォブ ィムノロジー ,第 9巻, 94頁, 1979年)も展開されている。 '285, p. 4.46, 1980), and its application to diagnostics and therapeutics (Chapter 9, Bianjar Nanoreoimmunology, Vol. 9, p. 94, 1979).
ヒ トのイ ンターフェロン ( I F N )には、抗原的に異なる a , β , 型の少くとも 3種のタイプが存在することが知られて居る(ネーチヤ一, 第 286卷, 1 10頁, 1980年)。 r型イ ンターフェロン ( I F lf 一 r )については、 マイ ト一ジヱンや抗原刺激によって、主として τリ ンパ球から^されることが判って居]?、 別名免疫インタ一フヱロン (I一 I FN )とも呼ばれている(ザ インターフェロン システム ,スプリンガー社, -ユーヨーク, 1979年)。 I FN— rは生体内で、種々の免疫反応 にともなって産生されることが予想され、免疫調節に重要 役割を果た していると考えられている。 また、 I F N— rの性質としては、 a型ィ ンターフェロン ( I F N— a )や3型ィンターフェロン ( I FN— /9 ) と抗原性が異¾ることや、誘起剤の種寧が異¾ることの泡 、酸や熱に 対する安定性が悪 ことなども判って る(ザ インタ一フエロンシス テム , スプ!;ンガー社,ニュ ヨーク , 1 979年)。 It is known that human interferon (IFN) has at least three types of a, β, and antigenically different a, β, types (Netiya, Vol. 286, 110, 1980, 1980). Year). It has been found that r-type interferon (IFlflfr) is mainly released from τ-lymphocytes by mitogen and antigen stimulation] ?, also known as immune interferon (I-IFN). ) (The Interferon System, Springer, -Eu York, 1979). IFN-r is expected to be produced in vivo in response to various immune responses, and is considered to play an important role in immune regulation. In addition, the properties of IFN-r include different antigenicity from type a interferon (IFN-a) and type 3 interferon (IFN- / 9), and different types of inducers. It is also known that the stability of foams, acid, and heat is poor (The Interferon System, Sp !; Nanger, New York, 1979).
—般的に I F Nは、生体の産生する抗ウィ^ス作用をもつものとして 定義されて るが、 この他に多くの生物活性をもつことが証明されてお i)、特に抗腫 «効果を有する点で注目されている( ブラッ ド , 55卷 71 1頁, 1980年;同誌, 56卷, 875頁, 1980年)。 —Generally, IFN is defined as having an anti-viral effect produced by living organisms, but it has also been demonstrated that it has many other biological activities i) (Blood, Vol. 55, p. 71, 1980; Ibid., Vol. 56, p. 875, 1980).
腫瘍の增殖を抑制する方法として、腫瘍細胞の増殖を直接抑制する方法 と、 宿主の免疫反応を介して、 間接的に腫瘍を抑制する方法が考えられ, 後者の場合、例えばナチュラ キラー細胞( NK )や、マクロファージ の活性化、或いはキラー T細胞の活性化 ¾どが考えられる。 実際、 I F Nには直接作用の他に、 この様 ¾種々の免疫増強活性があることが証明 されて る(バイオケミカ エト バイ才フイジ力 ァクタ ,第 516 卷, 231頁, 1978年)。 ェ F N— rはインビト口でのこれら抗腫 瘍につ ¾がる各種の活性、 およびィンビボに於ける抗腫瘍活性が、 I F — aや I F!T— /3に比べ遙かに高 ことから、その重要性が強く指摘 されて る(セルラーイムノロジー ,第 49巻, 390頁, 1980年)。 然しながら、 イ ンビト口で誘導される I F N— rの力価は一般に低い ことや適切 ¾I F N— r産生株細胞がほとんど こと、 さらに熱や酸 に対する安定性が悪 ため精製がむつかしいこと ¾どのために I F N— Tの大量生産および精製は I F N— aや I F N— i9に比べ大幅に遅れて いる。 Methods for suppressing tumor growth include a method of directly suppressing the growth of tumor cells and a method of suppressing the tumor indirectly through the host's immune response. In the latter case, for example, Natura killer cells (NK ), Activation of macrophages, or activation of killer T cells. In fact, IFN has been proved to have various immunopotentiating activities in addition to its direct action (Biochemica et al., Vol. 516, p.231, 1978). FN- r has much higher antitumor activity at INVIT and antitumor activity at in vivo than IF-a and IF! T- / 3 Its importance has been strongly pointed out (Cellular Immunology, Vol. 49, p. 390, 1980). However, the titer of IFN-r induced in the mouth is generally low or appropriate. ほ と ん ど Most IFN-r-producing cell lines are difficult to purify due to poor heat and acid stability. Mass production and purification of IFN-T lags far behind IFN-a and IFN-i9 I have.
ごぐ最近、 天然の I F IT -ァが単一に出来たという,報告があるが(プ ロシジング ォプ ナショナ アカデミ ー ォブ サイエンス ,第 Very recently, it has been reported that a single natural IF IT-machine was created (Procedures of the National Academy of Sciences, Chapter 1).
79卷, 1 820頁, 1 9 82年)、 活性の回収が大変悪く、 よ]?効果 的 精製法が待望されている。 Vol. 79, p. 1820, 1982), the recovery of the activity is very poor, and an effective purification method is expected.
—方最近に至]?、 ヒト I F N -ァ遺伝子のクロ ""ニングが報告され、 少くとも I F IT -ァの一種として、 〗令 6個のアミ ノ酸から成る約 1 7 キロダ ト ンの分子種が、 大蹑菌で^られる様に ¾つた(ネ -チヤ - , 第 29 5卷, 50 3貞, 1 9 82年;ヌクレイック ァシッズ リサ一 チ ,第 1 0卷, 248 7頁, 1 9 82年)が、 天然に産生されるェ F N -ァについては、 種々の分子量のものが報告されて居]?、その分子種間 の対応関係は未だ不明である。 —How recently?]? Cloning of the human IFN-α gene has been reported, and at least as one type of IF IT-α, a molecular species of about 17 kDa, consisting of six amino acids, has been identified in bacteria. As it is possible, Necchia, Vol. 295, 503 Sada, 1982; Nucleic Acids Lisachi, Vol. 10, pp. 2487, 1982 Various molecular weights of the produced FN-α have been reported.], But the correspondence between the molecular species is still unknown.
この様 視点から、各種の I F IT - 'rに対するモノクロ -ナノレ抗体を 得ることは、 分子種間の対応をつけるのに重要 だけでなく、 天然の、 或いは遺伝子組み換え法による大腸菌などで らせた I F IT - 7·を精製 する上に極めて強力 武器と ¾る3 ごく最近に至]?、 天然の -ァ に対するモノクロ -ナル抗体の取得が報告された(ネ -チヤ-,第 296 巻, 2 5 8頁, 1 9 8 2年)が、 I F N - rが複数の分子種から成って る場合、 遺伝子の組み換え法によ]?クロ-ン化された分子種に対する モノク口-ナル抗体を取得することは極めて重要とるる 3 From this point of view, obtaining monoclonal-nano antibodies against various IF IT-'r is not only important for establishing the correspondence between molecular species, but also using natural or genetically modified E. coli etc. IF iT - 7 · 3 most recently Ru extremely powerful weapon and ¾ on the purification of the optimal '?, natural - monochrome against § - get monoclonal antibody has been reported (Ne - Chiya -, 296, pp. 2 P. 58, 1982), when IFN-r is composed of multiple molecular species, a genetic recombination method was used to obtain monoclonal antibodies against cloned molecular species. It is extremely important to do 3
発明の開示 Disclosure of the invention
本発明者らは、 ヒト末 血リンパ球よ]?誘導した I F iT - の mBNA をもとに、 I F N -ァ遺伝子のクロ -ニングを行い、 その塩基配列を明 The present inventors performed cloning of the IFN-α gene based on the mBNA of IFiT-, which was derived from human peripheral blood lymphocytes, and clarified its nucleotide sequence.
•らかにしたが( P C T ZJ I> 82 /0 CI 0 80明細書参照)、 今回これ をもとに推定されるァミ ノ酸配列の C末端側の部分構造を有する 式 • Although it was clarified (see PCT ZJ I> 82/0 CI 080 specification), it has a partial structure at the C-terminal side of the amino acid sequence deduced based on this. formula
(N) (C) , 、 (N) (C),,
H-X-Lys Arg Ser Gin Met L eu Phe Arg Gl -Y-OH (I) H-X-Lys Arg Ser Gin Met Le eu Phe Arg Gl -Y-OH (I)
A (N) A (N)
〔式中、 X Ji^合手または li e Gin Val Me t Ala Glu Leu (In the formula, X Ji ^ joint hand or lie Gin Val Me t Ala Glu Leu
(C) ― (C) ―
Ser Pro Ala Ala Lys Tiir Gly Lys rgで されるぺブチ ド鎖にお てその C末端から数えて 1〜1 6個のァミノ酸を有するぺブチド Ser Pro Ala Ala Lys Tiir Gly Lys rg ぺ チ 鎖 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1
(N) (C) 一 もしくはアミノ酸残基を示し、 Υは Arg Arg Ala S er Ginで:^さ れるペプチド鎖に てその N末缁から数えて 1〜5個のアミ廣を有する ペプチドもしくはァミノ酸残基を示す〕で表わされるボリペプチドを化 学的に合成し、さらにキヤリヤー蛋白と化学結合させ蛋白複合体とした。 (N) (C) One or amino acid residue, Υ is Arg Arg Ala Ser Gin: Peptide or amino having 1 to 5 amino acids counted from its N-terminal in the peptide chain [The acid residue is shown.] Was chemically synthesized and further chemically bonded to a carrier protein to form a protein complex.
ここで得られたポリぺブチドまたは蛋白複合体で哺乳動物を免疫し、 取 出した陴贜細胞と同種または異種のリンパ球様細胞とを細胞融合に よ]?ハイブリドーマとし、 これをクローン化した。 A mammal was immunized with the polypeptide or the protein complex obtained here, and the extracted 陴 贜 cells were fused with allogeneic or heterologous lymphoid cells by cell fusion.] The hybridoma was cloned. .
ここで得られたハイブリ ドーマを哺乳動物に接種し、モノクローナ 抗体を生成 «せしめ、 これを採取して前記のポリベプチドに对するモ ノクローナ 抗体を製造した。 The obtained hybridoma was inoculated into a mammal to produce a monoclonal antibody, which was collected to produce a monoclonal antibody against the above-mentioned polypeptide.
さらに、 ここで得られる乇ノクローナ 抗体を利用し、粗ヒト I F N 一 Γ含有物からヒト I F N— rを精製する方法およびラジオィムノアツ セィ ( R I A )法るらびにェンザィムィムノアッセィ ( E I A )法によFurther, a method for purifying human IFN- r from crude human IFN-I content using the nocloclonal antibody obtained here, a radioimmunoassay (RIA) method, and a radiobiology enzyme immunoassay (EIA) method By
]?ヒト I N - rを検出する方法を確立し、本発明を完成した。 ]? A method for detecting human I N -r was established, and the present invention was completed.
す ¾わち本発明は、新規ボリべプチド( I ) ,その蛋白複合体,新規 クローン化されたハイブリ ドーマ,新規モノクローナル抗体およびこれ らの製造法、さらに該モノクローナ 抗侔の用途を提供するものである C 前記ポリペプチド( I )に関し、 Yは Arg Arg Ala S er Ginで あることが好ましく、 と!?わけこれに加え Xが Lys Argであることが 好ましい。 また、 ボリペプチド( I )はアミノ酸残基数としてと わけ In other words, the present invention provides a novel polypeptide (I), a protein complex thereof, a novel cloned hybridoma, a novel monoclonal antibody, a method for producing the same, and a use of the monoclonal antibody. It is preferable that Y is Arg Arg Ala Ser Gin with respect to the polypeptide (I), and! In addition, X is preferably Lys Arg. In addition, polypeptide (I) is not a specific number of amino acid residues.
OMPI — b— OMPI — B—
1 5〜25個であることが好まし 。 It is preferably 15 to 25 pieces.
当該ペプチドは、 ペプチド合成の常套手段で製造しうる。 固相合成法, 液相合成法のいずれによってもよいが、液相合成法が有利 場合が多 < » このようるペプチドの合成の手段は、たとえば、 " The Peptides-, 第 1卷( 1 9 6 6 ) , Schroder and L bke 著 , Academic Press , New York, U.S.A. , ある は"ペプチド合成",泉屋 ら著,丸善株式会社( t 9 75年) ,ある は"生化学実験講座,第 1 巻, 20 7頁 - 40 0頁"矢島治明著,東京化学同人株式会社 Π 977 に記載されてお J たとえば、 アジド法,クロライド法,酸無水物法, 混合酸無水 法, D C c法,活性エステ 法,ウッ ドヮ-ド試薬 Eを用 いる方法,カルボジィミダゾ— 法,酸化還元法, D C CZ了ディティ ブ(例、 HOWS, HOBt, HOSu )法 どがあげられる。 The peptide can be produced by conventional means of peptide synthesis. Either the solid phase synthesis method or the liquid phase synthesis method may be used, but the liquid phase synthesis method is advantageous in many cases. The method for synthesizing such a peptide is described, for example, in “The Peptides-, Vol. 6 6), Schroder and Lbke, Academic Press, New York, USA, or "Peptide Synthesis", Izumiya et al., Maruzen Co., Ltd. (t975), al. , Pp. 207-400 "Haruaki Yajima, Tokyo Kagaku Dojin Co., Ltd. Π 977. J Examples include azide method, chloride method, acid anhydride method, mixed acid anhydride method, DCc method, and activity. Esthetic method, method using wood reagent E, carbodimidazo method, redox method, DC CZ active (eg, HOWS, HOBt, HOSu) method.
当該べプチドは、 そのべプチド結合め任意の位置で 2分される 2種の フラグメントの一方に相当する反応性カ ボキシノレ基を有する原锊と、 他方のフラグメントに相当する反応性ァミノ基を有する 料をぺブチド 合成の常套手段で縮合させ、 生成する縮合物が保護基を有する場合、そ の保護基を常套手段で脱離させることによ 製造しうる。 The peptide has a reactive carboxy group corresponding to one of two types of fragments that are bisected at an arbitrary position in the peptide bond, and a reactive amino group corresponding to the other fragment. When the condensate formed is condensed by a conventional method for synthesizing a peptide and the resulting condensate has a protecting group, it can be produced by removing the protecting group by a conventional means.
原料の反応に関与すべきでない官能基の保護および保護基、 るらびに... その保護基の脱離、 反応に関与する官能基の活性化るどもまた公知のも のある は手段から適宜選択しうる 3 Protection of functional groups that should not be involved in the reaction of the raw materials and protective groups, removal of the protective groups, activation of the functional groups involved in the reaction, etc. 3 you can choose
原料のァミノ基の保護基としては、 たとえばカノレポベンゾキシ , t - ブチノレオキシカノレボニノレ , t一アミノレ; キシカルボ-ノレ , イソボノレニノレ キシカノレボ =1ノレ , p -メ ト キシベンジノレオキシカノレボニ , 2一クロ Examples of the protecting group for the amino group of the raw material include canolepobenzoxy, t-butynoleoxycanoleboninole, t-aminole; , 2 black
'ノレ—ぺンジノレ才キシカ ボ =ノレ, ァダマンチノレオキシカノレボニル , ト リ フル才ロアセチノレ , フタ リノレ, ホノレミノレ, ◦ -ニ ト ロフエ二 スノレフエ 'Nore-Zinnore, Chicka Bo-Nore, Adamantinoreoxycanolebonil, Trifle, Loacetinole, Phthalinore, Honoreminore, ◦ -Nitrofenii Snorefe
O PI — a— O PI — A—
ニル, ジフエ-ルホスブイノチオイル 4ーメ トキシー 2 , 3 , 6 ー ト リメチルペンゼンスルホニ どがあげられる。 カ ボキシル基の保護 基としては、 たとえばア キ エステ〃(例、 メチ ,ェチ ,プロビ , ブチ , t -プチ どのエステル基) ,ペンジ エステ 基, p ーュト口べンジ Vエステ 基, "0 —メ トキシペンジ エステル基, 一 ク口; 1ペンジ 1/エステ /1基, ペンズヒ ド リ エステル基, 力; ボペンゾ キシヒ ドラジド基, 一ブチ オキ^カ ボ- ヒ ドラジド基, トリチ / ヒドラジド基 ¾どがあげられる。 Nyl, diphenylphosphinoinoiloyl 4-methoxy2,3,6-trimethylpentenesulfonate and the like. Examples of the protecting group for the carboxyl group include, for example, akiester (eg, meth, eth, propyl, buty, t-butyl ester group), pendi est group, pout benzene V est group, "0— Methoxy pent diester group, one mouth; 1 pentyl 1 / ester / 1 group, penzhydri ester group, force; bopenzoxy hydrazide group, 1-butoxy carbazyl hydrazide group, triti / hydrazide group Can be
アルギ-ンのグァ -ジノ墓の保護基としては、たとえばニトロ基, ト ル基 , p —メ トキシベンゼンスルホ二/基 , カ ボべンゾキ^ , ィソ ボ ニ 才キシカ ボ =/ , ァダマンチルォキシカ ボ; =/t , 4 —メ ト キシー 2 , 6 ージメチルぺンゼンスルホ-ノレ基, ベンタメチノレぺンゼン ス/ Uホ-ル基等が例示される。 また、 ^:のグァ-ジノ基は、 酸(例、 ベ ンゼンス; ホン駿, ト エンス ホン酸,塩酸,硫酸など)塩の形で保 護してもよ 。 Protecting groups for the arginine guadino tomb include, for example, nitro, tol, p-methoxybenzenesulfonium, kabobenzoki ^, isoboni, and adaman. Tiloxycarb; = / t, 4-Methoxy 2,6-dimethylbenzenesulfonol group, pentamethynolensens / U-hole group and the like. In addition, the guadino group of ^: may be protected in the form of an acid (eg, venense; Hongjun, tonosulfonic acid, hydrochloric acid, sulfuric acid, etc.) salt.
スレオニンの水酸基は、たとえばエステノレ化またはヱ -テ/レ化によつ て保護することができる。 このエステル化に適する基としてはたとえば ァセチル基るどの低級ァ カノィ 基, ペンゾィ 基 ¾どのァロィ 基, ベンジルォキシカルボ-ノレ基,ェチノレオキシカノレボ - 基などの炭酸か ら誘導される基 ¾どがあげられる 3 またヱ-テル化に適する基としては、 たとえばベンジ 基, テトラヒ ドロ ビラ- 基, 七 ―プチル基などであ る。 しかしるがらスレオニンの水酸基は必ずしも保護する必要は 。 メチォ -ンはスルホキサイ ドの形で保護しておいてもよい。 原料のカル 'ボキシル基の活性化されたものとしては、たとえば対応する謖無水物, アジド ,活注エステノレ(ペンタクロロフエノ ー , p— -トロフエノ ー ノレ , IT一ハイ ドロキシサクシンイ ミ ド , N一ハイ ドロキシぺンズト リア ゾー , N— zヽィ ドロキシー 5 -ノノレボ ^ネン - 2 , 3 -ジカ ボキシ ィ ミ ド¾どとのエステ ) ¾どがあげられる。 ペプチド結合形成反応は 脱水剤(例、 ジシクロへキシルカ〃ボジィミ ド , カ ボジィ ミダゾ一 等のカ ボジィミド試薬)の存在下に実施しうる場合がある。 The hydroxyl group of threonine can be protected, for example, by esterification or esterification. Suitable groups for this esterification include, for example, groups derived from carbonic acid such as acetyl group, lower alkano group, penzoi group, alkaro group, benzyloxycarbo-nore group, ethynoleoxycanorebo-group and the like. Examples of groups suitable for 3- or etherification, such as benzyl, include benzyl, tetrahydroxy-, and 7-butyl groups. However, the hydroxyl group of threonine does not have to be protected. Methione may be protected in the form of a sulfoxide. Examples of the activated carboxyl group in the raw material include, for example, the corresponding chrysanthemic anhydride, azide, and hot-pottenol (pentachlorophenol, p--trophenol) Nore, IT-Hydroxysuccinimide, N-Hydroxydzintriazole, N-zdoxydoxy 5 -Nonolevo ^ nene-2, 3-Zikaboxymidido, etc.) can give. The peptide bond formation reaction may be carried out in the presence of a dehydrating agent (eg, a carbodiimide reagent such as dicyclohexylcarbodimid or cabodimidazo).
本ぺプチド縮合反応は溶媒の存在下に行うことができる。溶媒として は、 ぺプチド耮合反応に使用しうることが知られて るものから適宜選 択されうる。 たとえば無水または含水のジメチ ホ ムアミド, ジメチ スルホキサイ ド , ビリ ジン , クロロホ ム , ジォキサン , ジクロルメ タン , テトラハイ ドロフラン ,酵酸ェチ; U , —メチルビ口リ ドンある はこれらの適宜の混合物るどがあげられる。 The present peptide condensation reaction can be performed in the presence of a solvent. The solvent can be appropriately selected from those known to be usable in peptide coupling reactions. For example, anhydrous or water-containing dimethylformamide, dimethyl sulfoxide, pyridine, chloroform, dioxane, dichloromethane, tetrahydrofuran, acetic acid, U, —methylbilidone, or a mixture thereof. Can be
反応温度はべプチド結合形成反応に 用されうることが知られている 範囲から適宜選択され、 通常約 - 4 O t—約 6 0 ,好ましくは約 The reaction temperature is appropriately selected from a range known to be applicable to the peptide bond formation reaction, and is usually about -4 Ot to about 60, preferably about
- 2 0 一約 0 の範囲から適宜選択される。 -20 is appropriately selected from the range of about 0.
本縮合反応終了後、生成物が保護基を有している場合、それは常法に よ!)離脱できる。 かかる常法としては、 たとえば還元的方法(例、 パラ ジゥム黒等の触媒を用いる水素添加,液体アンモ-ァ中金属ナト リ ゥム による還元) , ァシドリシス(例、 トリフ 才ロ齚酸,フッ化水素,メ タンスルホン酸あるいは、 チオア-ソ -ル等の含硫化合物の存在下、 上 記の酸ある は、その混合物によるァシドリシス ) どがあげられる。 上記のようにして製造された本発明のベプチドは、 反応終了後混合物 から、 通常のぺプチドの分離手段,抽出,分配, カラムクロマ トグラフ ィ一 どによ]?採取できる。 If the product has a protecting group after completion of the condensation reaction, use a standard method! ) You can leave. Such conventional methods include, for example, reductive methods (eg, hydrogenation using a catalyst such as palladium black, reduction with metal sodium in a liquid ammonia), and acid lysis (eg, triflic acid, fluoride). In the presence of a sulfur-containing compound such as hydrogen, methanesulfonic acid, or thioazole, the above-mentioned acids or acid lysis by a mixture thereof are exemplified. The peptide of the present invention produced as described above can be collected from the mixture after completion of the reaction by ordinary means for separating peptides, extraction, distribution, column chromatography, and the like.
' ポリペプチドの蛋白複合体に関し、 キヤリャ' -蛋白の觀 よびキヤ リャ-とハプテン( この場合ペプチド) との混合比は、 キヤリヤ -に力 プリ ングさせて免疫したハプテンに対して抗侔が効率よく出来れば、 ど の様 ものをどの様る比率でカプリ ングさせてもよいが、 例えば牛血清 ア ブミンゃ牛サイログロブ 1Γン等を重量比でハブテン 1に対し 0. 1 - 2 0、 好ましくは 1 ~ 5の割合で力プ させる方法が用いられる。 'In terms of polypeptide-protein complexes, the carrier' and the mixing ratio of carrier and hapten (in this case, peptide) depend on the carrier. If an antibody can be efficiently produced against the hapten immunized by priming, any hapten may be coupled at any ratio.For example, bovine serum albumin, bovine thyroglobine, etc. may be used in a weight ratio. In this case, a method is used in which a force is applied to the hub ten at a rate of 0.1 to 20, preferably 1 to 5.
また、 ハプテンとキヤリヤーの力プリングには、 種々の縮合剤を用 ることが出来るが、 グノレタ ア デヒドゃ力 ボジィミド等が好都合に 用 られる。 In addition, various condensing agents can be used for the hapten and carrier force pulling, and gnoletaldehyde dehydrobodimide and the like are conveniently used.
ポリペプチド( I )または蛋白複合体を用 て免疫するに際し、 免疫 する哺乳動物は、 羊,山羊,兎, モ /レモット ,ラッ ト ,マウス等の実驗 動物が^われるが、 モノクロ—ナ/ u杬体を得るためには、 ラッ ト マウ スが好ましい。 免疫方法は、例えばマウスを免疫する場合、 皮下,腹腔 内 ,静脈内,筋肉内,皮内等のいずれの -トからでも可能であるが、 主として皮下, 内,静脈内に(と け皮下)注入するのが好まし 。 また、 免疫間隔,免疫量等も可変度は高く、氇々の方法が可能であるが、 例えば 2週間隔で 2 ~ 6回免疫し、最終免疫後、 1 ~ 5日、好ましくは 2 - 4日後の鞞钿胞を用いる方法がよぐ用 られる。 免疫量は 1回にベ プチド量として、 マウス当]? 0. 1 g 以上、好ましくは! 0 g ~ 3 0 0 用 ることが望まし 。 又,脬 を擠出する前に、 部分採血を行 、 血中の抗体価の上昇を確認した上で、 ^^細胞を用いる融合実験を行う ことが望まし 3 When immunizing with the polypeptide (I) or the protein complex, mammals to be immunized include experimental animals such as sheep, goats, rabbits, mo / remots, rats, and mice. In order to obtain a u 杬 body, a rat mouse is preferable. For example, when immunizing a mouse, it is possible to immunize a mouse subcutaneously, intraperitoneally, intravenously, intramuscularly, intradermally, etc. Prefer to inject. The immunity interval, immunity amount, etc. are also highly variable, and various methods are possible. For example, immunization is performed 2 to 6 times at 2 week intervals, and 1 to 5 days after the final immunization, preferably 2 to 4 times. A method using cells after days is often used. The amount of immunization should be the amount of peptide at a time, per mouse]? 0.1 g or more, preferably! It is preferable to use 0 g to 300 g. Also, before擠出the脬, partial blood collection line, after confirming the increase of antibody titer in blood, Shi desirable to perform fusion experiments with ^^ cells 3
上記獰藏細胞とリンパ球様細胞との讁飽融合は、 例えば摘出したマゥ スの脬藏細胞をヒポキサンチン一グァニン一ホスホリボシルトランスフ エラーゼ欠損( HGBBT" )や、 チミジンキナ -ゼ欠損( K— )の^ ¾ 'マ-力-を持った適切な同種または異種(好ましくは同種)のミエ口- マ等の、 リンパ球様細胞株との間で融合させる 3 融合には、 センダイゥ - y— The above-mentioned saturating fusion between the ferocytic cells and the lymphoid cells can be performed, for example, by excising the extracted mouse cells from hypoxanthine-guanine-phosphoribosyltransferase deficiency (HGBBT ") or thymidine kinase-deficient (K-). Three fusions to lymphoid-like cell lines, such as appropriate homologous or heterologous (preferably homologous) myeomas with the ability of -y—
イノレス , ホ。リエチングリ コ —ノレ( P E G )等の融合剤が用いられる。 も ちろんジメチ ス ホキシド ( PM S O )その他の融合促進剤を加える ことも可能である。 P E Gの重合度は、ふつう 1 0 0 0 ~ 600 0 ,時 間は 0.5 ~ 3 0分,濃度は 1 0 ^- 80 等が用いられるが、好ましい 条件の"^として、 P E G 60 0 0を 35 ~ 55 ?6で 4~ 1 0分処理す ることによ 、 劾率よく融合させることが出来る。 融合細胞は、 ヒポキ サンチンーァミノプテリ ンーチミ ジン培地 ( H A T培地; ネイチヤー , 第 256卷, 4 9 5頁, 1 9 75年)等を用 て、選択的に増殖させる ことが出来る。 Inores, E. A fusion agent such as lietin glyco-nore (PEG) is used. Of course, it is also possible to add dimethisoxide (PMSO) or other fusion promoters. The polymerization degree of PEG is usually 100 to 6000, the time is 0.5 to 30 minutes, and the concentration is 10 ^ -80. The cells can be fused at a high impeachment rate by treating them for 4 to 10 minutes at ~ 55 to 6. The fused cells can be grown in a hypoxanthine-aminopterin-thymidine medium (HAT medium; Nayya, Vol. 256, (P. 495, p. 1975) can be used to selectively proliferate.
.殖して来た細胞の培養上清は、 目的とする抗体産生があるか否かに つ てスクリ - ングを行うことができるが、 抗体価のスクリ -ニング は次の様に行うことが出来る。 即ち、 この場合には、 まず第 1段階とし て免疫したペプチドに対する抗体産生の有無を、 B I A法または E I A 法等の方法で調べることが出来るが、 これらの方法につ ても種々の変 法が可能である。 好ましい測定法の一例として、 E I Aを用 る一つの 方法について述べる。 セルロ -スビ-ズ等の担体に、 例えばゥサギ抗マ ケスィムノグロプリ ン抗体を常法に従ってカプリ ングさせておき、 これ に測定した 培養上清や、 マウスの血清を加え、 一定時間、定温(以下 The culture supernatant of the cultured cells can be screened for the production of the desired antibody, but the antibody titer can be screened as follows. I can do it. That is, in this case, the presence or absence of antibody production against the immunized peptide can be first examined as a first step by a method such as the BIA method or the EIA method, but various modifications of these methods are also possible. It is possible. As an example of a preferable measurement method, one method using EIA will be described. A carrier such as cellulos beads is coupled with, for example, a persimmon anti-Macsimiminoglobulin antibody according to a conventional method, and the measured culture supernatant or mouse serum is added thereto, and the mixture is incubated at a constant temperature for a certain period of time. Less than
4〜 40てを示す)で反応させる。 この後、 反応物をよく洗った後、 酵 素で標識したペプチド(酵素とベプチドを常法に従い力プリ ングさせた 後精製)を加え、 一定時間、 定温で反応させる。 反応物をよく洗った後、 酵素基質を加え、 一定時間,定温で反応させ、 その後、 生成発色物を吸 光度または螢光度等て測定することが出来る。 4 to 40). After that, the reaction product is thoroughly washed, and an enzyme-labeled peptide (purified after force-purifying the enzyme and the peptide according to a conventional method) is added, and the reaction is carried out at a constant temperature for a certain period of time. After thoroughly washing the reaction product, the enzyme substrate is added, and the reaction is allowed to proceed at a constant temperature for a certain period of time. Then, the resulting color product can be measured by absorbance or fluorescence.
- 選択培地で増殖を示し、 かつ免疫に用いたペプチドに対する抗体活性 のみられたゥェ の細胞は、 限界稀釈^によ]?クロ -ニングを行うこ -Cells that show growth in selective medium and have only antibody activity against the peptide used for immunization should be subjected to limiting dilution].
OMPI — l υ— OMPI — L υ—
とが望ましい。 クロ-ン化された細胞の上清について同様にスクリ - - ングを行い抗体僳の高 ゥエ の釉胞を增やすことによ]?、 免疫したぺ ブチドと反応性を示すモノクロ-ナ 抗体 ハイブリ ドーマクローン が得られる。 Is desirable. Similarly, screening of the cloned cell supernatant is performed to reduce the glaze of the antibody.], A monoclonal antibody that is reactive with the immunized butide A hybridoma clone is obtained.
次にこれらクロ -ンの産生する抗体が免疫に用 Λベプチドだけで く、 I F Ν - r分子そのものと反応性を示すか杳かを靄べる必要がある が、 このためには、標議された I F If - rを用 た!? I A法または Eェ A法を用 て反応性を調べる方法や、 生物活性( Ι Ρ ί - Γ活性) こ の抗体によ]? Κ収されるか否かを見る方法等が用いられる。 後者の場合 は I F IT - rを精製する必要が ¾ ので有和に用 ることが出来る。 有 利に用 られる一例を次に述べるが、 もちろんこの他にもプロテイン A を用 て免疫沈降物を除 た上清中の残存 I F 1ST - Γ活性を測定する方 法等も可能である。 例えばゥサギ抗マウスィムノグロブ ン抗体をセ Next, it is necessary for the antibodies produced by these clones to show reactivity with IF IF-r molecules themselves as well as the peptide used for immunization, or to haze. Used IF If-r! ? A method of examining reactivity using the IA or EA method, a method of checking whether or not biological activity (Ι Ι ί-Γ activity) is detected by this antibody] are used. In the latter case, it is necessary to purify IFIT-r. One example that is advantageously used is described below. Of course, a method of measuring the remaining IF1ST-II activity in the supernatant from which the immunoprecipitate has been removed using protein A is also possible. For example, a heron anti-mouse immunoglobulin antibody
-スビ-ズ等の担体に常法に従 カプリ ングさせておき、 これに測定 したいハイブリド-マ上清またはマウスの血淸を加え、 一定時間、 定温 で反応させる。 この後反応物をよく洙ぃ一定量の I P H -ァを加える。 -Capsule is added to a carrier such as beads according to a conventional method, and the hybridoma supernatant or mouse blood to be measured is added thereto and reacted at a constant temperature for a certain period of time. After that, the reaction mixture is added to the reaction solution in a constant amount.
I i* -ァとして、例えばヒト末梢血リンパ球からレクチンとホルボー エステ 等で誘導した I F K - rを含む培養上淸ゃ組み換え体で大腸 菌等で^らせたェ F IT - rを含む抽出液等を甩いることが出来る β I F H - rを加えた後、 一定時間,定温で反応させた後、反応上淸中に含ま れる I F rの活性を瀾定する。 この様にして目的とする抗体の I F - r活拴の肢収能を測定することが出来る。 For example, as an I i * -a, a recombinant containing IFK-r derived from human peripheral blood lymphocytes with lectin and phorbo este, etc. After adding β- IFH-r, which can be used as a solution, react at constant temperature for a certain period of time, and then determine the activity of IFr contained in the reaction. In this way, the limb yield of IF-r activity of the target antibody can be measured.
とのようにしてクローン化されたハイブリドーマは、例えば、 欲体培 地たとえば B P M I - 1 6 4 0に 0. 1 ~ 4 0 %の牛血淸を加えた培地等 で 0日間、好ましくは 3 ~ 5 3間 ^ることによ 、培養液か Hybridomas cloned as described above can be used, for example, in a greedy medium, such as BPMI-164, medium supplemented with 0.1 to 40% bovine serum for 0 days, preferably 3 to 40%. 5 3 times, the culture solution
OMPI ら該モノク -ナ 抗体を得ることができるが、 との他にマウス等の適 切な哺乳動物の腹腔内に接種し、 細胞 增殖させ、 腹水を採取すること によ 、 細胞培養上淸ょ も遙かに高力価の抗体を、 多量に効率よく取 得することが出来る。 このためには、 例えばマウスの場合、 ミネラ 才 ィ 等を前もつて接種した BALB ZC等のマウスに 1 X 1 04 ~ 1 X 1 07 個、 好ましくは 5ズ 1 05 - 2 I 06 偶のハイブ 9 ド-マを SC 腔内等に接種し、 7~20日後、 好ましくは 1 0~, 4日後に腹水狭等 を採取する。 腹水に生成蓄積した抗体は、 例えば碗安分画, D E A E - セ ロースカラムクロマト等によ!;)、容易に ノク ts -ナ; 1抗体を純粋 ¾免疫グロブリンとして単離することが出来る。 OMPI The monoclonal antibody can be obtained from the above, but in addition to the above, by inoculating intraperitoneally into a suitable mammal such as a mouse, and culturing the cell, and collecting ascites, the cell culture can be performed much more. It is possible to efficiently obtain a large amount of crab high titer antibody. For this purpose, for example, in the case of mouse, 1 X 1 0 4 ~ 1 X 1 0 7 or mice BALB ZC etc. Also connexion inoculated before Minera old I and the like, preferably 5's 1 0 5 - 2 I 0 6 Invite the hive 9-doma into the SC cavity, etc., and collect ascites narrow 7 to 20 days later, preferably 10 to 4 days later. Antibodies produced and accumulated in ascites fluid are collected, for example, by Bowan's fraction, DEAE-cellose column chromatography, etc.! ;), The antibody can easily be isolated as pure immunoglobulin.
本発明の ノクロ -ナ 抗体は、 下記の性伏を有する。 The Noclo-na antibody of the present invention has the following properties.
(1) 免疫に用 たポリペプチド( I )と結合する。 (1) Binds to the polypeptide (I) used for immunization.
(2) I F N- r分子と結合し、 I F N や I F if - とは結合しな (2) Binds to IFN-r molecule and does not bind to IFN or IFif-
(3) ォクタ口-—法による検定によ IgGnbまたは IgGi のサブ クラスの抗体に属する。 (3) It belongs to the IgGnb or IgGi subclass of antibodies by the assay method.
(4) S D S -ポリアクリ アミドゲ 電気泳動において、 標準免疫グ 口ブリンの H鎖および: L鎖に完全に一 る 2本のバンドのみを示 す。 (4) In SDS-polyacrylamide gel electrophoresis, only two bands completely matching the H chain and the: L chain of the standard immunoglobulin are shown.
本モノクロ-ナ〃抗体は安全に製造,使用,保管することができ る O This monoclonal antibody can be safely manufactured, used and stored O
本発明によ]?得られるモノクロ-ナ 抗体は、 E I A法或 は B .I A 法を活用することによ]?、生体中の、 或 は試験管内での徼量のェ F N According to the present invention, the obtained monoclonal antibody can be obtained by utilizing the EIA method or the B.IA method], and the amount of FN in the living body or in a test tube can be increased.
- rの検出に用いることが出来ることの他に、 たとえば抗体力ヲムを作 製することによ 、 今迄大変困難とされていた天然の、 或いは遺伝子組 みかえによ]?作られる I F N- rを、 非常に効率良く精製するのに使用 することが出来る。 -In addition to being able to be used for the detection of r, for example, the production of antibody chromosomes has made natural or genetic The resulting IFN-r can be used to purify very efficiently.
ェ P N - rの検出のためには、 一例として、測定した I 51 7*を含 む資料又は標準 I F N- rを予め、例えば放射性ョ -ド又は酵素等によ j?標識した当該モノクロ-ナ 抗体と反応させ次いで免疫複合体を沈降 させるべく例えばプロテイン Aを¾え、 一定時間反応させた後、 免疫沈 降物の放射活性又は薛素活 を測るととによ DiFir-rの量を簡便に 測定することが出来る。 また、 抗原による競合法を用いる場合は、例え ばポリペプチド( I )を放射性ョ-ド又は酵素標識し、 これと一定量の 無標識の本発明モノクロ -ナ の抗体を反応させる際に、 測定した I F N - rを含む資料を共存させ、 次 で、 免疫複合体を沈降させた]?、 ある は抗マゥス抗体に結合させることによ!?、 i F 5- rを定量する ことができる。 ここで用 る放射性ョ -ドまたは酵素檩議体としては、 ポリペプチド( I )の代 に精製した I Fir- rを使うことも出来る。 また抗体による競合法を用いる場合は、 例えば部分精製された I P IfE PN - for detection of r, as an example, the measured I 5 1 7 * previously including materials or standard IF N-r, e.g., radioactive ® -? By the de or enzyme etc. j labeled the monochrome -After reacting with antibody and then precipitating an immune complex with protein A, for example, and reacting for a certain period of time, measure the radioactivity or Xe activity of the immunoprecipitate to easily reduce the amount of DiFir-r. Can be measured. When the competition method using an antigen is used, for example, the polypeptide (I) is radioactively or enzymatically labeled, and when a certain amount of the unlabeled monoclonal antibody of the present invention is reacted, the measurement is performed. The co-existing material containing IFN-r was used to precipitate the immune complex.], Or by binding to an anti-mouse antibody! ?, IF5-r can be quantified. As the radioactive or enzyme complex used here, purified I Fir- r can be used instead of polypeptide (I). When using the antibody competition method, for example, partially purified IP If
- rまたはポリぺプチド( I )をマイクロトレイ等の固相に固定する。 固定する方法としては、例えば、 0.〗 1ίの重、ノウを含有するリン酸籙衝 液に I i« Ν - rまたはボリぺブチド( I )を 0., -Immobilize r or polypeptide (I) on a solid phase such as a micro tray. As a fixing method, for example, I i Ν Ν -r or polybutide (I) is added to a phosphoric acid solution containing 0.1〗 weight and nou to 0.,
好ましくは 10 ~ 20 に浮遊させこの 100 ^ をマイク p ト レイの各ゥエ に入れ 24時間処理することによ!;可能である。 このゥ エ に本発明モノクロ-ナ 抗佯と測定した i P N- rを含む資料を 37でで!時間又は 4 °Cで 20時間反応させたものを加え、 さらに放射 性ョ-ドまたは酵素標識し ウス ゥサギ抗体等を加えるととによ!) I F rを定量することができる。 これら一連の E I A法 }Cおいて酵 素標識した抗マウス ゥサギ抗侔等を; 3 た場佥は、いわゆる ELISA —丄 — Float it preferably in 10-20, put this 100 ^ in each channel of the microphone p tray and process for 24 hours! It is possible. The material containing iPN-r, which was determined to be the antimonochrome of the present invention, was added to this report at 37! After reacting for 20 hours at 4 ° C, add radioactive or enzyme-labeled mouse heron antibody, and so on! 2.) IF r can be quantified. The enzyme-labeled anti-mouse (eg, heron-antibody, etc.) in these series of EIA methods — 丄 —
( ェンザィム リ ンク ド ィムノ ソ -ベント アツセィ )法によ]?マ チスキャン ( フ口-社)等を用いた迅速アツセィ法が可能となる。 ま た、 抗原認識部位の異なる 2種のハイブリ ド-マクロ- ンの ¾ ^するモ ノク P —ナ 抗体を用いた場合は、 わゆるサン ドウイツチ法による、 よ])簡便な I P iT - rの測定が可能と ¾る。 もちろん、 これらの (By the Enzyme Link Dom-So-Vent Atsay) method]. A quick Atsey method using Machiscan (Fuguchi-sha) is possible. In addition, when the monoclonal P-na antibody of two types of hybrid-macrones having different antigen recognition sites is used, the so-called Sandwich method is used.)) It is possible to measure. Of course, these
B I Aを利用した I F rの定量に適切な抗体さえ得られれば、 種々 の変法が可能であ]?、 上記した方法に限定されるものでは * 。 Various modifications are possible as long as an antibody suitable for the quantification of IFr using BIA can be obtained.], * Not limited to the above method.
I F N - 7"の精製のためには、 精製した当該抗体を例えば活性化した ァガ口 -スゲ〃ビ-ズの様な適切 ¾担体に常法に従ってカブリ ングさせ た後、 カラムに充め、 培養上清或いは破さいした菌体等の粗 I - r を含む資料をカラムにかけ、 吸着さ^ t後、 洗净し、 その後例えば∑s c irの様なカオト口ピック試薬、 或いは I F N - rの失活の¾い程度の 弱酸性条件で溶出させる方法等によ]?、 効率よく精製できる。 In order to purify IFN-7 ", the purified antibody is coated on a suitable carrier such as activated agarose beads according to a conventional method, and then applied to a column. Material containing crude I-r such as culture supernatant or disrupted cells is applied to the column, washed after adsorption, and then washed with a pick-up reagent such as ∑sc ir or IFN-r Depending on the method of elution under weakly acidic conditions with a high degree of inactivation], purification can be performed efficiently.
抗体カラムの作製は、例えばハイブリド-マを接種した腹水等から純 すいに精製した本発明のモノクロ-ナ 抗体を適切な担体と力プリ ング させることによ]?、 以下の様 ¾方法でできる。 The antibody column can be prepared, for example, by force-purifying the monoclonal antibody of the present invention purified from ascites or the like inoculated with the hybridoma with an appropriate carrier], and the following methods. .
用いる担体は、 カブリングの後に I F N - rが特異的に効率よく ¾着 され、 その後適切 ¾溶出が可能 ¾ものであればどの様 ¾ものでもよいが、 —例として蛋白の一級ァミ ンが結合し易い様に活性化されたァガロ -ス ゲ ビ-ズ、例えばァフィゲ - 1 0などが以下に述べる様な方法で好 都合に用いられる。 ァフィゲ - 1 0と抗体との反応は、 0. 0 0 1 ~ 1 M、 好ましくは 0. 1 Mのバイ力-ボネ-ト等の緩衝液中で反応を行なう。 反応条件は 0β~ 2 0 , ! 0分 ~ 2 4時間、種々の ρ Ηが可能であるが、 好ましくは、 4 *C , 4時間, p H 3 ~ 1 0の条件が用いられる。 混合す るアブイゲル- 1 0と抗体の量比は、 アブイゲ に対し抗体量が約 Any carrier can be used, as long as IFN-r is specifically and efficiently attached after cou- pling, and then can be appropriately eluted. Activated agarose beads such as Affige-10 are conveniently used in the manner described below. The reaction between Affige-10 and the antibody is carried out in a buffer solution of 0.001-1 M, preferably 0.1 M, such as biforce-bonate. Reaction conditions are 0 β ~ 20,! Various values of ρ 可能 are possible from 0 minutes to 24 hours, but preferably, the conditions of 4 * C, 4 hours, and pH 3 to 10 are used. The amount ratio of Abuigel-10 and antibody to be mixed is such that the amount of antibody
OMPI 循 — I 4— OMPI circulation — I 4—
5 0 W位迄は多ければ多い程多くの抗佯がつくので、 この範囲内で く らでもよ が、 結^ r率を考慮して 3 0 以下の抗体が好都合に用いら れる。 この様にしてできた抗体-担体結合物は、反応に用いた緩衝液で よく洗った後、数日放置するか、 もしくは最終濩度 0. 0 5 Mのェ Antibodies of up to 50 W are more likely to be repelled as much as possible. Therefore, any antibody within this range may be used, but an antibody of 30 or less is preferably used in consideration of the r ratio. The antibody-carrier conjugate thus obtained is thoroughly washed with the buffer used for the reaction, and then left for a few days or at a final concentration of 0.05 M.
O PI " タノ一 アミン ·塩酸を加え 4わで 1時間反応させる等の方法に.よ D、 残存する未反応の活性基をブロックした後、適切 ¾力ヲムにつめること によ]?、抗体カラムとして使用できる。 O PI " Use a method such as adding Tanoamine / hydrochloric acid and reacting with a rock for 1 hour. D. After blocking the remaining unreacted active groups, fill them with an appropriate solution], use as antibody column it can.
上記した抗体カラムで精製するに際しては、たとえばヒト免疫ィンタ 一フエ口ン蛋白質含有試料を中性附近の緩衝液、たとえばリン酸緩衝液 ゃト リス ·塩酸緩衝液に溶解して抗体カラムに吸着させる。 次にカヲム を同じ緩衝液で洗浄したのち、 I F N - rを溶出する。溶 液としては, 弱酸性溶液たとえば酢酸溶液,ポリエチレングリコ— を含む溶液,試 料にくらべ抗体に、 よ D結合し易いぺブチドを含む溶液,高濮度塩溶液 など よびこれらの組み合せた溶液 どが用いられ、 ヒ ト I一 I F Nの 分解をあま])促進し ¾ ものが好ましい。 When purifying with the antibody column described above, for example, a sample containing human immunogenic protein is dissolved in a buffer solution near neutral, for example, phosphate buffer solution, tris-hydrochloric acid buffer solution, and adsorbed to the antibody column. . After washing the camb with the same buffer, IFN-r is eluted. Examples of the solution include a weakly acidic solution such as an acetic acid solution, a solution containing polyethylene glycol, a solution containing ぺ butide, which is more easily D-bound to the antibody than the sample, a high-purity salt solution, and a combination thereof. It is preferably used to promote the degradation of human IFN-IFN]).
カラム溶出液は、常法によ 緩衝液で中和する。必要によ 再度上記 の抗体力ラムによる精製操作を行 ¾うことができる。 The column eluate is neutralized with a buffer according to a conventional method. If necessary, the above-described purification operation using the antibody force column can be performed again.
ここで得られるヒト I一 I F IT蛋白質溶液は透析に付し、必要によ]? これを凍結乾燥によ])粉末とすることができる。 凍結乾燥に際しては、 ソノレビ トー , マン =1 トーノレ ,デキス トロース , マ/ uトース ,グ'リセ口 一ル どの安定剤を加えることができる。 The human I-IFIT protein solution obtained here is subjected to dialysis, and if necessary] this can be made into a powder by freeze-drying]. For freeze-drying, stabilizers such as sonorebitose, man = 1 tonole, dextrose, ma / u tose, and glucose can be added.
かくして得られるヒト免疫ィンターフェ口ン蛋白質はヒト羊膜由来 W I S H細胞に対する水泡性口内炎ウイ ス( V S V )の細胞変性効果阻 止試験によるウィ ス活性測定において 1 07 UZ 以上の比活性を示す ものである。 Thus obtained human immune Intafe port down protein shows a 1 0 7 UZ more specific activity in WE scan activity measurement by cytopathic effect deter test vesicular stomatitis Huy scan (VSV) on human amnion-derived WISH cells .
おここで I F Nの活性としての U ZW (ュ -ット ^ )の出し方は以 下の様に行った。 ュ=ットの確定した国際標準 I FN— <∑ と白血球由来の 粗 IFN - r をヒ ト羊膜由来 F L細胞株に対する V S Vの細胞変性効果阻 止試験を用 て測定し、その力価の比較から白血球由来粗 I FN— rの力 Here, the method of producing U ZW (cut ^) as the activity of IFN was performed as follows. The international standard IFN- <∑ and the crude IFN-r derived from leukocytes were determined using the VSV cytopathic effect inhibition test on FL cell line derived from human amniotic membrane, and the titers were compared. Of leukocyte-derived crude IFN-r
OMPI 価を決定し IFN— rの檩準品とした。 目的とする资料中の iFii-rの力OMPI The value was determined, and the standard product of IFN-r was used. IFii-r's power in the intended feed
,偭算定のためには、常にこの檁率 IFN-rを並べて前述の W I S H - V S Vの系でアツセィを行 、その比率から力価を算出した。 In order to calculate 偭, the efficiency IFN-r was always arranged, and the results were calculated using the WISH-VSV system described above, and the titer was calculated from the ratio.
本勞明で精製処理されるヒト免疫インターフェロン蛋白質は、たとえ ば一殺式 The human immune interferon protein purified in this study is
(N)H- A-Zx Tyr Z2 Gin Asp Pro Tyr Val Lys Gl Al Gl Asn Leu Lys L73 Tyr Phe Asn Ala Gly His Ser As Yal Ala Asp Aan GI7 Thr Leu Phe Leu Gly lie Leu Lys Asn Trp Lys Glu Glu Ser Asp Arg Lys lie Met Gin Ser Gin lie Val Ser Phe Tyr Pile Lya Leu P e Lys Asn Phe Lys Asp Asp Gin Ser lie Gin Lys Ser Val Glu Thr lie Lys Glu Asp Met Asn Val Lys Phe Plie Asn Ser Asn Lys Lys Lys Arg Asp Asp Phe Glu Lys Leu Thr Asn Tyr Ser Val Thr Asp · Leu Asn Val Gin Arg Lys Ala lie His Glu Leu lie Gin Yal Me Ala Glu Leu Ser Pro Ala Al Lya Thr Gly Lys Arg L s Arg Ser Gin Met Leu Phe Arg Gly Arg Arg Ala Ser Gln-OH(C) ( IE ) (N) H-AZ x Tyr Z 2 Gin Asp Pro Tyr Val Lys Gl Al Gl Asn Leu Lys L73 Tyr Phe Asn Ala Gly His Ser As Yal Ala Asp Aan GI7 Thr Leu Phe Leu Gly lie Leu Lys Asn Trp Lys Glu Glu Ser Asp Arg Lys lie Met Gin Ser Gin lie Val Ser Phe Tyr Pile Lya Leu P e Lys Asn Phe Lys Asp Asp Gin Ser lie Gin Lys Ser Val Glu Thr lie Lys Glu Asp Met Asn Val Lys Phe Plie Asn Ser Asn Lys Lys Lys Arg Asp Asp Phe Glu Lys Leu Thr Asn Tyr Ser Val Thr Asp · Leu Asn Val Gin Arg Lys Ala lie His Glu Leu lie Gin Yal Me Ala Glu Leu Ser Pro Ala Al Lya Thr Gly Lys Arg L s Arg Ser Gin Met Leu Phe Arg Gly Arg Arg Ala Ser Gln-OH (C) (IE)
〔式中、 Aは Metまたは結合手を示し、 Zi, Z2はそれぞれ Cysまたは ½ Cysを示す〕のァミノ酸配列から るポリぺプチドを含有するもので ある。 本発明の精製処理によれば当該ポリペプチドを乾燥品基準で 90 »以上と わけ 95 «1¾上含有するものを得ることができる。 Those containing polypeptide that from wherein, A is Met or indicates a bond, Zi, Z 2 represents a Cys or ½ Cys respectively] Amino acid sequence of. According to the purification treatment of the present invention, it is possible to obtain a polypeptide containing the polypeptide in an amount of 90 or more, especially 95 <1> on a dry matter basis.
本発明の精製法によ!?ヒト^ィン^"フエロン蛋白質を得るととができる。 1) S D S一ポリアクリ ァミ ドゲ ( 17.5 ¾ )電気泳動による分子 量測定で 17,000±1,000を示す β IPO 2) ァ ミノ末端アミノ酸としてシスティンもしくはハーフシスチンまた はメチォ -ンを有する。 According to the purification method of the present invention! ? Human ^ obtains a fin ^ "Feron protein bets can. 1) SDS one polyacrylic § Mi Doge (17.5 ¾) IPO β indicating the molecular weight measured at 17,000 ± 1,000 by electrophoresis 2) It has cysteine or half cystine or methionine as the amino terminal amino acid.
3) H— Lys— Arg— Lys— Arg— S er— Gin— Met— Leu— Phe— Arg— 3) H— Lys— Arg— Lys— Arg— Ser— Gin— Met— Leu— Phe— Arg—
Gly-Arg-Arg-A la-S e r-G ln-ΟΗ に対するモノ クローナ 抗 体と結^る。 Binds to a monoclonal antibody against Gly-Arg-Arg-Ala-Ser-Gln-ΟΗ.
本発明によ D精製されたヒ ト免疫インターフエ口ン蛋白質は従来の方 According to the present invention, the D-purified human immune interface protein can be obtained by conventional methods.
• 法で得られる I一 I FNと同様の目的に同様の用法によ!)使用できるが、 従来品に比し、 夾雑蛋白質,発熱物質が少ないので、 注射剤原体等とし てよ )安全に使用される。 • Use the same usage for the same purpose as the I-I FN obtained by the method! ) It can be used, but it has less contaminating proteins and pyrogens than conventional products, so it can be used safely as an injection drug substance.
本発明のヒ ト I一 I F N蛋白質は抗ゥイノレス,抗腫瘍,細胞増殖抑制お よび免疫増強作用を示す。 本発明のヒト I-IFN 蛋白質は滅菌水 , ヒト 血清アルブミン( H S A ),生理食塩水その他公知の生理学的に許容される 担体と混合することができ、非経口的に又は局所に投与することができ る。 洌えば、正常人 1日当 ]) 1 0万〜 1億ュ-ット、好ましくは 5000万 〜 6000万ュ-ットを静注又は筋注などによ]?投与することができる。 本発明のヒト I— I F N蛋白質を含有する製剤は、塩,希釈剤,アジュ バン ト ,他の担体,バッファー ,結合剤,界面活性剤,保存剤のようる 生理的に許容される他の活性成分も含有して てもよい。 非経口的投与 用製剤は、 滅菌水溶液又は生理学的に許容される溶媒との懸濁液アンプ ル、 または生理学的に許容される希釈液で用事希釈して使用しうる滅菌 粉末(通常 I一 I FN溶液を凍結乾燥して得られる)アンプ として提供 される。 The human I-IFN protein of the present invention has anti-inoles, anti-tumor, cell growth inhibitory and immunopotentiating effects. The human I-IFN protein of the present invention can be mixed with sterilized water, human serum albumin (HSA), physiological saline or other known physiologically acceptable carriers, and can be administered parenterally or topically. it can. In the case of Kiyoshi, a normal person can receive 100,000 to 100 million ut, preferably 50 to 60 million ut, by intravenous injection or intramuscular injection. The preparation containing the human I-IFN protein of the present invention may contain other physiologically acceptable activities such as salts, diluents, adjuvants, other carriers, buffers, binders, surfactants, and preservatives. Components may also be included. Preparations for parenteral administration may be sterile aqueous solutions or suspension ampoules in physiologically acceptable solvents, or sterile powders (usually I-I), which can be used after dilution in physiologically acceptable diluents. It is provided as an amplifier (obtained by freeze-drying FN solution).
さらに上記したヒト免疫インターフェロン蛋白質を含有する製剤は、 I F N— βまたは I FN— (9またはィンタ一ロイキン 2 どのリ ンホカイ ンの よう ¾他の活性成分を本発明物質に対し 1〜9 9 含有していてもよい。 Further, the above-mentioned preparation containing the human immune interferon protein contains 1 to 99 other active ingredients relative to the substance of the present invention, such as IFN-β or IFN- (9 or interleukin 2 or any other lymphokine). May be.
OMPI — I 8— OMPI — I 8—
本明細書にお て、 アミノ酸,ペプチド,保護基,活性基,その池に 関し略号で表示する場合、 それらは I Π P A C— I订 BCGommisaion on Biological Nomenclature による略号あるいは当該分野 に ける慣用略号に基づくものであ]?、その例を次に挙げる。 また、 ァ ミノ酸 どに関し光学異性体があ])うる場合は、特に明示しるければ L 体を示すものとする。 In the present specification, when abbreviations for amino acids, peptides, protecting groups, active groups, and their ponds are indicated by abbreviations, they are based on the abbreviations by IΠPAC—I 订 BCGommisaion on Biological Nomenclature or common abbreviations in the art. The following is an example. When there is an optical isomer with respect to amino acids, etc.]), the L-isomer shall be indicated unless otherwise specified.
DN A: デォキ 5 リホ:孩酸 DN A: Deoki 5 Riho: Baby Acid
A : アデニン ' A: Adenine ''
T ; チミ ン T: Thymin
G -: グァニン G-: Gwanin
C シ ト シン C cytosine
RN A ; リボ核酸 RN A; ribonucleic acid
d A T P; デ才キシアデノ シン三リ ン酸 d A T P; Dexyadenosine triphosphate
d T T P: デォキシチミ ジン三リン酸 d T T P: Deoxythymidine triphosphate
dG T P デ才キシグァノ シン三リ ン酸 dG T P
Π T P ' 亍'ォ J ノ シ ジ- ソニ U ¾s Π T P '亍' o J
A P ; アデノ シン三リ ン酸 A P; Adenosine triphosphate
E D T A ; エチレンジァミ ン四薛酸 E D T A; Ethylenediaminetetrasulfonic acid
S O S ドデシル硫酸ナ ト リ ウム S O S Sodium sodium dodecyl sulfate
G y: グリ シン G y: Glycine
Al a '. - ァラニン Al a '.-Aranin
Vaa : パリ ン Vaa: Paris
L e u ; ロイ シン ' Leu; Leucine '
11 e r イソロイ シン 11 e r isoleucine
S ex : セ !/ ン - 1 y -S ex: Se! / -1 y-
T h r ; ス レ才ニン T h r;
Cy s : システィン Cy s: Sistine
M e t ; メチ才-ン M e t;
Glu: ク"ルタ ミ ン酸 Glu: Klutamic acid
A S J) ァスノ ラギン酸 A S J) Asno laginic acid
L y s リ ジン Lys resin
Ar g アルギニン Ar g Arginine
Hi s : ヒ スチジン Hi s: Histidine
P θ フェニーノレァラニン P θ Pheninolealanine
T 7-r: チロ.シン T 7-r: Chiro. Shin
T r p ト リ フ。ト フア ン T r p Trif. Tohwan
Pro *. フ°口 リ ン Pro *.
A s n · ァスノ ラ ギン A s n · Asno La Gin
Gin ·, グルタ ミ ン Gin
z :カルボベンゾキシ z: Carbobenzoxy
•B o c : t一ブトキシカルボ^: • Boc: t-butoxycarbo ^:
H"br : 4 -メ トキシ— 2 , 3 , 6—ト リメチ ベンゼンス ホニ H "br: 4-Methoxy-2,3,6-trimethylbenzenesoni
Pme:ベンタメチ ぺンゼンス ホ二 ^ Pme : Ventamechi Pengens Hoji ^
ΟΒτι :七ーブチ エステ ΟΒτι : Shichibuchi esthetic
0ΝΒ : 1Τ—ヒ ドロキ、ン— 5ーノ ボ ネンー 2 , 3 -ジカ^ボキシィ 0ΝΒ: 1Τ—Hydroxy, n—5-no Bonen 2,3 -Zika ^ Boxy
ミ ドエステ Midoste
T>C C: ΪΓ , Ν'-ジシク口へキシ' /V力 ボジィミ ド T> C C: ΪΓ, Ν'-disc hex '/ V force Bozimid
D CTJ: ίΓ , ージシクロへキシ ゥレア D CTJ: ίΓ, dicyclohexyl ゥ rare
E0NB : ίΓーヒ ドロキシー 5ーノ〃ボ/ ^ネ ー 2 , 3 -ジカ ボキシィ E0NB: P-Droxy 5-Nobo / ^ Ne 2,3 -Zika Boxy
Ο ΪΙ HOB : U—ヒ ドロキシペンゾト リアゾ一 Ο ΪΙ HOB: U—Hydroxypenzoto Liazoi
CHA : シク口へキ^ ァミン CHA: Awesome mouth
DCE A:ジ ク口へキシ ァミ ン DCE A: Exit mouth
TEA : ト リエチルァミン TEA: Triethylamine
TFA :ト!; 7 ォロ静醭 TFA: G! ;
USA :メタンス ホン酸 USA: Methanesulfonic acid
THF :テトヲヒ ト,ロブラン THF: Tetohito, Roblanc
DMF :ジメチ ホ ムアミ ド DMF: Dimethiformamide
MeOE:メタノー MeOE: Methaneau
AeOEt:薪酸ェチ AeOEt: firewood acid
図面の簡単 ¾睇明 Easy drawing
図 1は実旌例 1 0 *^る本発明のモノク -ナル抗体の DE AEセ ロースカラムに ける溶出バタ -ンを示し、図 2は実施倒 1 0に け るモノタローナ 抗体の ¾ ^泳動の結杲を示す 0 Fig. 1 shows the elution pattern of the monoclonal antibody of the present invention on the DEAE cellose column, and Fig. 2 shows the elution pattern of the monoclonal antibody of the present invention. 0 indicating the result
図 3は実施例 1 3の後に示した本発明の精製法で得られるヒト免疫ィ ンタ -フェロン蛋白質の鼋気泳動の結果を、図 4は実施例 i 3の後に同 様示した分子量瀾定の結果を示す。 図 5は参考例〗 (V8)で得られた pHIT 3709 プラスミ ドの一次構造(潼基配列)を、 図 6は参考倒 2 Π),《)の ptrp 701および ptrp 771 ,図 7は参考例 2 (iii )の pHIT trp 1101 ,図 8は参考例 2 (iV) ( pEITセ rp 2101 の構築 図を、 図 9は実旅例 1 9によるモノクロ—ナ 抗体に対する ωリコンビ ナント i FN-rサンブ と fc)固相に固定された I FIT- rとの競合を 示す。 図 1 0および 1 1はそれぞれ実施例 2 0 -(i) , 2 0— (ii)によるモ ノク口—ナノレ抗体に対する (a)IFN— r含有又は不含大腸菌抽出液と (b)酵 素壤識された本発明のペアチドとの競合を示す。 - - 発明を実施するための最良の形態 FIG. 3 shows the results of the electrophoresis of the human immunointerferon-feron protein obtained by the purification method of the present invention shown after Example 13, and FIG. 4 shows the molecular weight variation similarly shown after Example i3. The result is shown. Figure 5 shows the primary structure (Tong group arrangement) of pHIT 3709 plasmid obtained in Reference Example II (V8), Figure 6 shows the reference structure 2), ptrp 701 and ptrp 771 in <<), and Figure 7 shows the reference example 2 (iii) pHIT trp 1101 and Fig. 8 shows the construction of Reference Example 2 (iV) (pEIT cell rp 2101), and Fig. 9 shows the ω recombinant i FN-r And fc) show competition with I FIT-r immobilized on the solid phase. FIGS. 10 and 11 show (a) Escherichia coli extract containing or not containing IFN-r and (b) enzyme against the Monica-Nanole antibody according to Examples 20- (i) and 20- (ii), respectively. 1 shows the competition with the paired peptide of the present invention which was recognized. --Best mode for carrying out the invention
以下の実施例に いて、 薄層クロマトグラフィ ーは、 メ ク社製シリ 力ゲ プレー ト 60 F £54 又は、 フナコシ薬品社製セル口 -スプレ - ト ,アビセ S Fを用い、 下記の展開溶媒を用いた。 In the following examples, thin-layer chromatography was performed using the following developing solvents, using Mek's Sili-Ige Plate 60 F £ 54 or Funakoshi Pharmaceutical's Cell-Port-Splate, Abysse SF. Was.
Ef1 : クロロホノレム : メタノ -〃:鏵酸- 9 : 1 : 0.5 Ef 1 : Chlorophonorem: Methano -〃: 鏵 acid-9: 1: 0.5
Bf2:醉酸ェチ : ピリ ジン :酢酸:水- 30 : 1 0 : 3 : 5 Bf 2:醉酸E Chi: pyridinium Jin: acetic acid: water - 30: 1 0: 3: 5
Bf^ : ク口 ロホノレム : メタノ ー : k = 7 : 3 : 0.5 Bf ^: Kuho Rohonolem: Methanol: k = 7: 3: 0.5
Pf : n -ブタノ 一 : ビリ ジン : 酸:水- 3 0 : 20 : 6 : 24 Pf: n-butanoyl: viridine: acid: water-30: 20: 6: 24
Bf5:齚酸ェチ : n—ブタノ - :齚駿:水 = 1 : 1 : 1 : 1 Bf 5 : 齚 酸 チ: n —butano-: 齚 jung: water = 1: 1: 1: 1: 1
実施例 1 Example 1
( ί ) Z- Ser- Gin - OBu七 の製造 (ί) Manufacture of Z- Ser- Gin-OBu 7
Z-Gln-OBu* 1 0.! をメタノ -ノレ 50 0 βに溶解し、パラジウム Z-Gln-OBu * 10! Is dissolved in methano
黒を触媒に水素気流中で還元した。 触媒をろ去し、 溶媒を留去した残留 The black was reduced as a catalyst in a stream of hydrogen. The catalyst was removed by filtration and the solvent was distilled off.
¾Jを Z— Ser-OH 7.5 ^ , 30273 6.8 とともに D M F 250 Wに溶 解し、 氷冷した。 永冷下に!) C C 7. 1 5?を加え、 0 で 4時間室温で ¾J was dissolved together with Z-Ser-OH 7.5 ^, 30273 6.8 in DMF 250 W and cooled on ice. Under eternal cooling! ) C C 7. 15? And add 0 at room temperature for 4 hours
1 2時間攪拌した。 析出した D C ϋを 去し、 溶媒を留去ののち残留物 を AcOUt 3 0 0 Wに抽出し、 4 % aHC03水, 0.2 IT塩酸,水で洗い、 無水硫酸ナ ト リ ウムで乾燥した。 溶媒を留去し、 析出した結晶をエ-テ ルでろ取し、 乾燥ののち c¾5 Ci『 よ ]3再結晶した。 収量 6.5 ,収率 Stirred for 12 hours. Were removed and precipitated DC Y, the residue after the evaporation of the solvent was extracted into AcOUt 3 0 0 W, 4% aHC0 3 water, 0.2 IT hydrochloride, washed with water and dried over anhydrous sulfate Na Application Benefits um. The solvent was distilled off, and the precipitated crystals were collected by filtration with an ether. After drying, the crystals were recrystallized from c 『5Ci. Yield 6.5, Yield
O PIO PI
、 5 1.2 ¾ , Rf 1 0.6 4 mp. 9 7 - 1 0 0わ , 〔 a〕¾5 - 24.1。 , 5 1.2 ¾, Rf 1 0.6 4 mp 9 7 -. 1 0 0 Wa, [a] ¾ 5 - 24.1.
( c = 0.4 0 , メタノール) (c = 0.40, methanol)
元素分析 C2oS2907li3 として Elemental analysis as C 2 oS 29 0 7 li3
計算値: C, 56. 72 ; H.6. 90; N , 9. 92 Calculated: C, 56.72; H.6.90; N, 9.92
実験値: C,56. 21 ; H,6. 72; N,9. 76 Experimental values: C, 56.21; H, 6.72; N, 9.76
( β ) Z-Ala-Ser-Gln-OB * の製造 Production of (β) Z-Ala-Ser-Gln-OB *
Z-Ser-Gln-OBu11 4.2 3 をメタノール 3 0 に溶解し、ノヽ。 ラジウム黒を触媒に水素気流中で還元した。触媒をろ去し、溶媒を留去 した残留物を Z— Ala— OH 2.3 4 ? , HONB 2·2 7 ?とともに、 Dissolve Z-Ser-Gln-OBu 11 4.2 3 in methanol 30 and mix. Radium black was reduced as a catalyst in a stream of hydrogen. The catalyst was removed by filtration, and the residue obtained by evaporating the solvent was purified to give Z—Ala—OH 2.34? , HONB 2.2
AcOEt 2 0 0* とジォキサン 2 0 0» , D M F 1 0 0» の混合溶媒に 溶解し、氷冷した。冷却下に! C C 2.38 ?を加え 0 で 4時間、室温 で 1 2時間攪拌ののち、 D C Uをろ去し、溶媒を留去した。残留物に CH3C とエーテ を加え結晶としろ取、乾燥ののち CH3CN よ ])再結 晶した。収量 3.7 2 ,収率 7 5.3 ¾ , Rf1 0.6 0 , mp. 1 6 5 — 1 7 0 t , 〔 a〕¾a—4 1.2 ° ( c = 0.5 0 , メタノー〃) It was dissolved in a mixed solvent of AcOEt 200 *, dioxane 200 », and DMF 100» and cooled with ice. Under cooling! CC 2.38? After stirring at 0 for 4 hours and at room temperature for 12 hours, the DCU was filtered off and the solvent was distilled off. CH 3 C and ether were added to the residue, and the crystals were collected by filtration, dried, and then recrystallized from CH 3 CN. Yield 3.7 2, Yield 75.3 R, Rf 1 0.60, mp. 16 5 — 17 0 t, [a] ¾ a —4 1.2 ° (c = 0.50, Methanol)
元素分析 C23H3408N4 として Elemental analysis as C 23 H 34 0 8 N 4
計算値: C,55. 86; H,6. 93; N.11. 33 Calculated: C, 55.86; H, 6.93; N.11.33
実験値: C, 55. 58; Η,6· 74 ; Ν,11· 12 Experimental values: C, 55.58; Η, 6 · 74; Ν, 11 · 12
( 1 ) Z-Arg(Pme)-Ala-Ser-Gln-OBut の製造 (1) Production of Z-Arg (Pme) -Ala- Ser-Gln-OBu t
Z- A 1 a- S β r-G 1 η-0 Β u ¾ 3· 4 6 をメタノール 3 0 0 Wに溶解し, パラジウム黒を触媒に水素気流中で還元した。 触媒をろ去し、溶媒を留 去した残留物を、 Z-Arg(Pme)-OH- CHA 4.5 49から調製した Z-A 1 a-S β rG 1 η-0 Βu 43 · 46 was dissolved in methanol 300 W and reduced with palladium black as a catalyst in a hydrogen stream. The catalyst was removed by filtration, and the residue obtained by removing the solvent was prepared from Z-Arg (Pme) -OH-CHA 4.549.
Z-Arg(Pme)-OHf HOB 1.9 fとともに D M F 1 5 0 に溶解し 永冷した。 冷却下に D C C 1.9 を加え、 0 で 4時間、室温で 1 5時 間攪拌した。 D C ϋをろ去し、溶媒を留去し残留物に AcOEt を加えゲ - - ル伏の沈澱を得、 ろ取した。 乾燥後、 メタノ - と AcOEt よ 再沈澱 した。 収量 4.5 5 ,収率 75.5 % , Si"1 0.5 7 , mp . 1 3 0 - 1 34°C , 〔 〕 5— 24. Γ ( c = 0.26 , メタノー〃) With Z-Arg (Pme) -OH f HOB 1.9 f was cooled and dissolved in DMF 1 5 0 permanent. Under cooling, DCC 1.9 was added, and the mixture was stirred at 0 for 4 hours and at room temperature for 15 hours. DC DC was removed by filtration, the solvent was distilled off, AcOEt was added to the residue, and --A precipitate was obtained and filtered off. After drying, it was reprecipitated with methano- and AcOEt. Yield 4.5 5 75.5% yield, Si "1 0.5 7, mp 1 3 0 -. 1 34 ° C, [] 5 - 24. Γ (c = 0.26 , methanol 〃)
元素分析 C40 H6001]LN8 S - H20として Elemental analysis as C 40 H 60 0 1] L N 8 S-H 20
計算値: c, 55.22; Η,7·0了; if, 12.88; S,3,69 Calculated: c, 55.22; Η, 7.00; if, 12.88; S, 3,69
実験値: c, 55.23; H,6.93 ; 12.54; s, 3.48 Experimental values: c, 55.23; H, 6.93; 12.54; s, 3.48
( iV ) Z-Arg (: Pme)-Arg (: Pme ) - Ala— Ser - Gln-OBuセ の製造 (iV) Z-Arg (: Pme) -Arg (: Pme)-Ala— Ser-Gln-OBu
Z— Arg (: Pme)— Ala— Ser— (Jin— OBu七 4.3 をメタノー 40 0 Z— Arg (: Pme) — Ala— Ser— (Jin— OBu 7 4.3
Wに溶解し、 パラジウム黒を に水素気流中で還元した。 ^^をろ去 し、溶媒を留去した残留物を、 Z-Arg(Pme)-OH-CHA 3.24ダよ It was dissolved in W and the palladium black was reduced in a stream of hydrogen. The ^^ was removed by filtration, and the residue obtained by evaporating the solvent was purified using Z-Arg (Pme) -OH-CHA 3.24.
調製した Z— Arg(Pme)— OH, EOB七 1.42 とともに nt Prepared Z—Arg (Pme) —OH, EOB with 1.42 nt
に溶解し永冷した。 冷却下に ID C C 1.4 1 を加え 0 で 4時間、 室温 And cooled forever. Add ID C C 1.4 1 under cooling at 0 for 4 hours at room temperature
で 2 0時間攪拌した。 D C Uをろ去し、'溶媒を留去した残留称 i ^cOE七 For 20 hours. The DCU was removed by filtration and the solvent was distilled off.
を加え沈澱を得、 ろ取した。 乾燥後、 メタノ - と AcOEセ よ]?、再沈 澱した。 iKfi4,4 ,収率 7 1.7 , Ef1 0.63 , mp. 1 25— Was added to obtain a precipitate, which was collected by filtration. After drying, it was reprecipitated. IKfi4,4, yield 7 1.7, Ef 1 0.63, mp . 1 25-
1 3 0 , 〔 〕^3 - 1 8.0 ( c = 0.4 0 , メタノー ) 1 3 0, [] ^ 3 - 1 8.0 (c = 0.4 0, methanol)
元素分析 C57H860i42Ti2 S2 - H2 0として Elemental analysis C 5 7H 86 0 i 42 Ti 2 S 2 -H 20
計算値: c,54.9S; H,7.12; N,t3.50;S,5.15 Calculated values: c, 54.9S; H, 7.12; N, t3.50; S, 5.15
実験値: C, 54.66 ; H, 6.92; N, 13.32 ; S , 5.34 Experimental values: C, 54.66; H, 6.92; N, 13.32; S, 5.34
( V ) Z-Arg ( Pme ) -Gly-OBu の製造 (V) Manufacture of Z-Arg (Pme) -Gly-OBu
Z— Glァー OBu七 1 3.0 を MeOH 5 0 0 にと:^し、 黒を触媒 として、 水素気流中、 接触還元した。 蝕媒を泸別し、 庐液を滅圧濃縮し、 残留物を; D M F 2 0 0 Wにとかした。 これに、 Z-Arg(Pme)— 0Η· Z-Gl OBu 7 13.0 was converted to MeOH 500: and catalytically reduced in a stream of hydrogen using black as a catalyst. The solvent was separated off, the aqueous solution was decompressed and concentrated and the residue was dissolved in DMF200W. In addition, Z-Arg (Pme) — 0Η ·
■CHA 2 0.0 ^よ]?調製した Z— Arg(Pme)— OH, HOB七 5.4 ^を加 えて氷冷し、 D C C 8.2 を加えて 4 8時間かき混ぜた。 析出した DC ■ CHA 20.0 ^ Y]? The prepared Z-Arg (Pme) -OH, HOB 75.4 ^ was added, ice-cooled, and DCC8.2 was added, followed by stirring for 48 hours. Precipitated DC
O P を泸別し、濃縮後、 AcOEt 50 にとかした。 AcOEt 層を 4 HaHC03 水, 1 0 ?6クェン酸水で洗净後、 Ha2S04 で乾燥した ^縮 後、石油ペンジンを加えて沈凝として 取した。 収量 1 9.8 (96. 8 )。 mp. 了 1 - 73 , 〔 〕23 + 0.2。 ( c = 0.9 , D M P ) , Bf 1 0.62 OP After concentration, the mixture was concentrated and dissolved in AcOEt 50. The AcOEt layer 4 HaHC0 3 water, 1 0? 6 Kuen acid water in washing净後, Ha 2 and dried S0 4 ^ After condensation, was collected as沈凝added petroleum Penjin. Yield 19.8 (96.8). mp. end 1-73, [] 23 + 0.2. (c = 0.9, DMP), Bf 1 0.62
元素分折 (33ιΗ45 θ7 Ή5 S として Elemental analysis (as 3 3 ιΗ45 θ 7 Ή 5 S
計算値: c,58.93; H,7.18; ir, 11.03; s,5.08 Calculated: c, 58.93; H, 7.18; ir, 11.03; s, 5.08
実験値: C,53*42i H,了.58; HO.95; S,4.84 Experimental value: C, 53 * 42i H, R. 58; HO. 95; S, 4.84
( Vi ) Z-Piie-Ar CPme -Gl -OBu11 の製造 (Vi) Manufacture of Z-Piie-Ar CPme -Gl -OBu 11
Z-Arg ( me ) -Gly-OB^ 1 Q.0 を MeOH 50 0 W中、 接触還 元したのち、 D M F 30 0 «Jに転溶した。 これに Z -: Plie - OH 4.72 , Z-Arg (me) -Gly-OB ^ 1Q.0 was catalytically reduced in MeOH 500 W, and then transferred to DMF300 <J. This with Z-: Plie-OH 4.72,
HOBt ZZ 59を加えて氷冷し、 D C C 3« 5 9 を加えて、 ! 5 時間 かきまぜた。 折出した D C ϋを泸別し、'庐液を濃縮後、残留 を AcOEtAdd HOBt ZZ 59, cool with ice, add D C C 3 «59, and! Stir for 5 hours. The separated DC was separated and the solution was concentrated.
40 にとかした。 AcOEt 層は、 4HTaHC0 水, 1 0 $クェン 酸水で洗浄し、 ifa2 SO で乾燥した。 濃縮後、 ヱ-テ^を加えて結晶 として庐取し、 MeOE —ヱ -テ よ 再結晶した。 収量 1 G.5 ^40. The AcOEt layer was washed with 4HTaHC0 water, 10 $ citric acid water, and dried with ifa 2 SO. After concentration, -te ^ was added and the mixture was collected as crystals and recrystallized from MeOE- ヱ -te. Yield 1 G.5 ^
( 85.3 )。 mp . 1 Q 1 - 1 0 3 °C , C « ^° - 8.3°( c = 0.9 ,(85.3). mp .1 Q 1-103 ° C, C «^ °-8.3 ° (c = 0.9,
D M F ) , Ε Q.64 D M F), Ε Q.64
元素分析 C40H5408 if6 S として Elemental analysis C 40 H 54 0 8 as 6 S
計算値: c,61.67; H,6.93; N, 10.79; s,4.12 Calculated: c, 61.67; H, 6.93; N, 10.79; s, 4.12.
実験値: c, 61.66; : s, 6.56; 10.93; s,4.14 Experimental values: c, 61.66;: s, 6.56; 10.93; s, 4.14
( Vii ) Z-Le -Bhe-Arg(Pme )-Gl -OBu* の製造 (Vii) Production of Z-Le -Bhe-Arg (Pme) -Gl -OBu *
Z-Phe-Arg(Pme )-Gl -0But 5.5 を MeOH 3 0 0 *?中、接 触還元したのち、 ; D MF 3 0 0 Wに転溶した これ 、 Z-Le -OH DCHA 3.3 1 ^よ]?調製し 7 Z— Leu— OH, HOiiB 1.47 を で — 20— Z-Phe-Arg (Pme) -Gl-0 But 5.5 was contact-reduced in MeOH 300 * ?; then, it was transferred to DMF 300 W. This Z-Le-OH DCHA 3.3 1 ^ 7] Z-Leu-OH, HOiiB 1.47 — 20—
冷し、 D C C 1, 6 8 を加えて 48時閬かきまぜた。 析出した DC ϋを 泸別し、 濃縮後、 AcOEt 3 0 0 にとかした oAcOEt 層は、 4 % HaHC03 水, 〗 0 クェン酸水で诜狰し、 Na2S04で乾燥した。 濃 縮後、 エーテ を加えて粉末として庐取した 3 収量 &2 f ( 98.3^) mP , 1 7 1 - 1 7 3 Ό , C « ¾6 - 1 5.5 ( c = 0.9 , D M F ) , Bf 1 0.64 Cool, add DCC 1,6 8 and stir for 48 hours. The precipitated DC Y and泸別, concentrated, OAcOEt was dissolved in AcOEt 3 0 0 layer was诜狰with 4% HaHC0 3 water〗 0 Kuen acid solution and dried with Na 2 S0 4. After enrichment, 3 yield was庐取as a powder by adding ether & 2 f (98.3 ^) m P, 1 7 1 - 1 7 3 Ό, C «¾ 6 - 1 5.5 (c = 0.9, DMF), Bf 1 0.64
元素分拆 C 6H6509 2T7, S Element removal C 6 H 65 0 9 2T 7 , S
計算値: C, 61.93; H,7.34; iT, 10.99; S,3.59 Calculated: C, 61.93; H, 7.34; iT, 10.99; S, 3.59
実験値: C, 62.(32; H,7.37; 11.08; s,3.59 Experimental value: C, 62. (32; H, 7.37; 11.08; s, 3.59
( Vfii ) Boc— Me七一: Leu— Phe— Arg(Piae )— illy— OBu七 の製造 (Vfii) Boc—Michiichi: Production of Leu—Phe—Arg (Piae) —illy—OBu
Z— Leu— Phe— Arg(Pnie)— Gly— OBu七 6.0 を MeOH 200 W中、 接蝕還元したのち、 D MF 1 50"に転溶した これに、 Boc-Me七 一 0H.DCEA 3.0 よ]?調製した Boc Me七一 OH, H0 1.3 9 ^ を 加えて氷冷し、 D c c 1.5 9 を加えて 1 5時間かきまぜた 3 折出 し た D C ϋを泸別し、 濃縮後、 n - BicQH- AcOE七にとかし、 1 056 ク ェン酸水で洗浄し、 Na2S04 で乾燥した。 濃縮後、 ェ-テノレを加えて 結晶として泸取した。 収量 6.2 ^ ( 9 3.1 % ) 。 mp. 1 9 2— ί 953C,Z—Leu—Phe—Arg (Pnie) —Gly—OBu 76.0 was reduced by corrosion in 200 W of MeOH and then transferred to DMF 150 ”. Boc-Me 7-11 0H.DCEA 3.0 ] Add the prepared Boc Me Shichiichi OH, H0 1.39 ^, cool on ice, add Dcc 1.59, stir for 15 hours, discriminate 3 folded DCs, concentrate, and concentrate BicQH- AcOE dissolved seven, 1 056 and washed with click E phosphate solution, then dried and concentrated in Na 2 S0 4, E -.. Tenore was泸取as crystals by adding yield 6.2 ^ (9 3.1%). mp. 1 9 2— ί 95 3 C,
〔 ¾ 一 1 9. ( c = 1.0 , D M F ) 5 Rf1 0.6 4 〔¾1 9. (c = 1.0, DMF) 5 Rf 1 0.6 4
元素分析 C48H76010 W8 S2 として Elemental analysis C 48 H 76 0 10 W 8 S 2
計雄: c,5.8.27; H, 7.74; I .33; s, 6.48 Total male: c, 5.8.27; H, 7.74; I.33; s, 6.48
実験値: c, 58.48; H,7.79 ; 1 ] .-34; S,5.98 Experimental values: c, 58.48; H, 7.79; 1] .- 34; S, 5.98
( IX ) Boc-Gln-Met-Le -Phe-Arg(Pme)-Gl7-0H の製造 (IX) Production of Boc-Gln-Met-Le-Phe-Arg (Pme) -Gl7-0H
Boc-Met-Le -Phe-Arg(Pme)-Gl7-0But 5, 5 ^に T F A Boc-Met-Le -Phe-Arg (Pme) -Gl7-0Bu t 5,5 ^ TFA
を加え、 室温で〗 0分間振]?まぜたのち濃縮し、 ェ-テルを加え て庐取し乾燥した。 これを、 D M F 5 にとかして氷冷し、 T E A 一 2 O — Was added and shaken at room temperature for about 0 minutes, followed by concentration, addition of ether, and extraction and drying. This was dissolved in DMF 5 and cooled on ice. One 2 O —
1.8*を加えた。 これに; Boc— Gln-OH 1.85 H0 1.49 , D C C 1.9 0 よ]?調製した Boc-Gln-Ol を加え、 1 5時 ffi^き まぜた。 濃縮後、 AcOHを加え、 AcOEt を加えて沈澱として^取し Tto 収量 5.3 ^ ( 89.8 % )。 mp. 1 8 1 - 1 83X: (分解) , !: a〕 一 1 9. S ( c = 1.0 , D H F ) , Ήί丄 0.30 1.8 * was added. To this; Boc-Gln-OH 1.85 H0 1.49, DCC 1.90]? The prepared Boc-Gln-Ol was added and mixed at 15 o'clock. After concentration, AcOH was added, and AcOEt was added to obtain a precipitate, and the Tto yield was 5.3 ^ (89.8%). mp. 181-183X: (decomposition),! : A] 1 1 9. S (c = 1.0, DHF), Ήί 丄 0.30
元素分析 C49 Η76012 Κ 10 S2 として Elemental analysis C49 Η 7 6012 Κ 10 S 2
計算値: C, 55.45; H,7.22; IT, 13.20; s,6.04 Calculated: C, 55.45; H, 7.22; IT, 13.20; s, 6.04
実験値: C,55.33; H,7.17i ir, 13.34; S,6.20 Experimental value: C, 55.33; H, 7.17i ir, 13.34; S, 6.20
( X ) Z-Ser-iTHliH-Boc の製造 (X) Production of Z-Ser-iTHliH-Boc
Z— Ser— OH 1 0.0 , セ一プチ 力 バゼー ト 6.2 yを Dlii" Z— Ser— OH 1 0.0, septum force bathet 6.2 y Dlii "
1 5 にとかして永冷し、, Η01 Β 8.0 , D C C 9.3 を加えて15 Cool forever, add Η01 Β 8.0 and DCC 9.3
1 5時間かきまぜた。 折出した D C ϋを庐別し、 濃縮後、 AcOEt に転 溶した。 AcOEt層は、 4 % NaHC03 ' , 1 0 %クェン酸水で洗铮し、 li s 304 で乾燥した。 瀵縮後、 エ-テ を加えて結晶として泸取し Tto Stir for 15 hours. The separated DCs were separated, concentrated, and then dissolved in AcOEt. AcOEt layer, 4% NaHC0 3 ', and washed铮1 0% Kuen acid solution, and dried over li s 304. After contraction, add ether and extract as crystals.
OR o 収量了.3 ( 50.45S ) mp . 9 5 - 9 8 Ό , C « - 6.2 ( c = 0.8 i D MF ) , Ef1 0.62 OR o Yield. 3 (50.45S) mp. 95-98 Ό, C «-6.2 (c = 0.8 i D MF), Ef 1 0.62
元素分折 C16H230s 3 として Elemental analysis C 16 H 23 0 s 3
計算値: c,54.38; Η,6.56; ίΓ, Π .89 Calculated: c, 54.38; Η, 6.56; ίΓ, Π.89
実験値: C, 54.77; H,6.88; 12.29 Experimental values: C, 54.77; H, 6.88; 12.29
( Χί ) Z-Arg(Pme)-Ser-S"HNH-Boc の製造 (Χί) Production of Z-Arg (Pme) -Ser-S "HNH-Boc
Z-Ser-iTHlTH-Boc 3. を MeOH 3 0 0 中接腿元したのち、 DM F 5 0 に転溶した。 これに、 Z-Arg(Pme)-OE-CHA S.29 よ ]J調製した Z-Arg(Pme)-OH, ΗΟΒΐ 1.5 を加えて氷冷し、 Z-Ser-iTHlTH-Boc3 was transferred to DMF50O after the thigh was placed in MeOH300. To this, add Z-Arg (Pme) -OH, ΗΟΒΐ1.5 prepared from Z-Arg (Pme) -OE-CHA S.29], cool on ice,
D C C 2.3 を加えて 1 5時間かきまぜた。 析出した D C を泸別し、 濃'縮後、 AcOE にと:^した。 AeOE 層は、 4 % iTaHC03 水, 1 0 — ' — クニン酸水で洗净し、: iTa2S04で乾燥した。 濃縮後ェ-テノレを加えて, 沈澱として泸取する。 収量 7.45 ( 93.7 )。 mp. 100 - 1 (Π XT, C « D - 0.9°C c = 1.2 , D M F ) , Bf 1 0.5 1 元素分析 CssH^^O^I^ S として Add DCC 2.3 and stir for 15 hours. The precipitated DC was separated, concentrated and reduced to AcOE. AeOE layer, 4% iTaHC0 3 water, 1 0 - '- and it washed净in Knin acid water: dried with ITA 2 S0 4. After concentration, add ethanol and collect as a precipitate. Yield 7.45 (93.7). mp. 100-1 (Π XT, C « D -0.9 ° C c = 1.2, DMF), Bf 1 0.5 1 Elemental analysis As CssH ^^ O ^ I ^ S
計'算値: C, 55.(36 ; Η,6·86; H3.62; S,4.46 実験値: c, 55.49; H,6.94; 13.14; S,3.86 ( fl ) Z-L3TS(li" r)-Arg(Pme)—Ser-3iH:ErH-:Boc の製造 Calculated value: C, 55. (36; Η, 6.86; H3.62; S, 4.46 Experimental value: c, 55.49; H, 6.94; 13.14; S, 3.86 (fl) Z-L3TS (li " r) -Arg (Pme) —Production of Ser-3iH: ErH-: Boc
Z-Ar g ( Pme ) - S e r-Ι ΕΙΓΗ-Β o c 3.9 を MeOH 30 0 かし、 接触還元したのち、 : DM I" 50"に転溶した。 これに、 Z - I rsOi七 r) - 0 H · D C H A 3'4 ょ D調製した Z— I rs(M" r)— 0H, HOB七 Z-Ar g (Pme) -Ser-Ι ΕΙΓΗ-Β oc 3.9 was subjected to catalytic reduction with MeOH 300 0 and then transferred to DMI “50”. Z-I rs (M "r) -0H, HOB
0.8 8 を加えて氷冷し、 : D e c 1, 34 を加えて 20時間かきまぜ た。 析出した D C 13:を庐別し、 濃縮後、 AcOE を加えて粉末として庐 取し、 MeOH-AcOEtよ 再結晶した。 収量 5.1 ^ ( 9 3.4 $6 )After adding 0.88, the mixture was cooled on ice, and: Dec1, 34 was added and stirred for 20 hours. Precipitated DC13: was separated and concentrated, and then AcOE was added to obtain a powder, which was recrystallized from MeOH-AcOEt. Yield 5.1 ^ (9 3.4 $ 6)
g g
mp. 1 0 3 - 0 5 , 〔 a - 7.2° ( c = 0.8 , D M F ) , Bf1 0.57 元素分析 C49H 73013 Hg S2 として mp. 10 3-0 5, [a-7.2 ° (c = 0.8, DMF), Bf 1 0.57 Elemental analysis C 49 H 73 0 13 H g S 2
計算値: 55.50; H,6.94; if, 11.89; s,6.05 実験値: c, 55.70; H,7.15; IT, Π .60; s,5.63 Calculated: 55.50; H, 6.94; if, 11.89; s, 6.05 Experimental: c, 55.70; H, 7.15; IT, Π.60; s, 5.63
(Xiil) Z-Arg ( Pme ) -Lys ( M七 r ) -Arg ( Pme ) - Ser - NMTH -: Bocの製 造 (Xiil) Z-Arg (Pme) -Lys (M7r) -Arg (Pme)-Ser-NMTH-: Boc
Z-Lys (Mtr)-Arg(Pme )-Ser-HHiJ"H-Boc 4.8 f を MeOH 3 0 0 «fにとかし、 接触還元したのち、 DM F 7 に転溶した。 こ れに、 Z-Arg(Pme)-OH'CHA 2.8 ょ 調製した Z— Arg ( Pme )— '0Η, HOBt 0.74 ^を加えて氷冷し、 : D C C U 2 ^を加えて 1 5 時間かきまぜ 。 析出した D C ϋを泸別し濃縮後、 AcOEセ にとかし、 Z-Lys (Mtr) -Arg (Pme) -Ser-HHiJ "H-Boc 4.8 f was dissolved in MeOH 300 f and subjected to catalytic reduction, and then dissolved in DMF7. Arg (Pme) -OH'CHA 2.8 Add prepared Z—Arg (Pme) — '0Η, add HOBt 0.74 ^, cool on ice, add: DCCU 2 ^, and stir for 15 hours. Separately concentrate and dissolve in AcOE
OMPI -2B- AcOEt 層を 4 %lTaHC03 永, 1 0 クェン酸 7_Rで 净し、 Na2S04 で乾燥した。 濃縮後、 エーテ を加えて沈鏺として泸取し、 MeOH -ェ 一テ よ 再沈澱した。 収量 5.8 f ( 89·8 )。 mp.136- 137 ,OMPI -2B- AcOEt layer 4% lTaHC0 3-permanent, and净1 0 Kuen acid 7_R, and dried over Na2S0 4. After concentration, ether was added and the precipitate was collected and reprecipitated from MeOH. Yield 5.8 f (89 · 8). mp.136-137,
〔 a 〕2S - 6.6°( c = 1.0 , DM F ) , Ef1 0.58 (A) 2S -6.6 ° (c = 1.0, DMF), Ef 1 0.58
D D
元素分折 C66H990ェ 6ίί13 S3 として 計算値: C, 55.56; Η36.99; ΐί, 12.78; S,6.了 4 実験値: C,55.34; H,7.Q7; ir,12.50; S,6.59 Calculated elemental folding C 6 6H 99 0 E 6 ίί 13 S 3:. C , 55.56; Η 3 6.99; ΐί, 12.78; S, 6 Ryo 4 Found: C, 55.34; H, 7.Q7 ; ir , 12.50; S, 6.59
( XiV ) Z-L73 (Mtr ) -ArgCPme )一 1«ァ3 C M r )一 Arg C Pme )一 Ser— KH H-Boc の製造 Z-Arg ( Pme ) -Lys ( r ) -Arg ( Pme ) -S er-UHiTH-B o c 3.0 を] ieOH 1 5 にとかし、接敏還元したのち、 DMF 60 に 溶した。 これに、 -Lys(Mtr)-OH-DCHA 1.54 ょ ]?調製した Z—I^sCMtr -aE, HOBt 0.3 1 を加えて氷冷し、 D C C 0.57 ^を加えて 1 5時間かきまぜた。 折出した D C ϋを泸別し、濃縮後、 AcOEt にと し、 AcOEt層を 4 % aHCO 水, 1 0 $クェン酸水で 洗净し、 Nan S04で乾燥した。 濃縮後、 ヱ -テルを加えて結晶として泸 取し、 AcOEt よ]?再結晶した。 収量 2.70 ( 72.8 $ )(XiV) Z-L73 (Mtr) -ArgCPme) 1 11 ァ 3CM r) 1 Arg C Pme) 1 Ser-- Production of KH H-Boc Z-Arg (Pme) -Lys (r) -Arg (Pme) -Ser-UHiTH-Boc 3.0] was dissolved in ieOH 15, reduced by sensitization, and then dissolved in DMF60. To this, -Lys (Mtr) -OH-DCHA 1.54]? Prepared Z-I ^ sCMtr -aE, HOBt 0.31 was added, ice-cooled, DCC 0.57 ^ was added, and the mixture was stirred for 15 hours. And泸別the DC Y that issued folding, after concentration, and in AcOEt, and CLEANING The AcOEt layer 4% aHCO water, 1 0 $ Kuen acid solution and dried with Na n S0 4. After concentration, ヱ -tel was added thereto to collect crystals, which were recrystallized from AcOEt. Yield 2.70 (72.8 $)
P. 1 23— 1 25 , 〔な 6 - 5.9。 ( c = 1.1 , D M F ) , Ef1 0.57 元素分析 c82 3o20iri5 s4 として 計算値: c, 55.73 Η,7·02; N, 11.89; s,7.26 実験値: c,55.89; H,7.3Q; u, 11.72; S,7.Q8 P. 1 23- 1 25, [of 6 - 5.9. (c = 1.1, DMF), Ef 1 0.57 Elemental analysis c 82 3 o 20 iri 5 s 4 Calculated: c, 55.73 Η, 7.02; N, 11.89; s, 7.26 Experimental: c, 55.89; H , 7.3Q; u, 11.72; S, 7.Q8
( XV ) Bo c Gin - Met— Leu -: Phe - Arg ( Pme )一 Gly - Arg ( Pme )一 ArgC me )一 Ala - Ser - Gin - OBu七 の製造 Z-Arg(Pme )-Arg(Pme )-Ala-Ser-G-ln-OBut 1. 0 を - - MeOH 1 0 0 にとかし、 接触還元したのち D M F 2 flWに転溶し た。 これに、 Bo c-Gln-Me t-Leu-Phe-Arg ( Pme ) --(JI7-OH 0.8 5 , HOBt 1 3 5 ¾^を加えて氷冷し、 : D C C 2 1 0 ^を加え て 20時間かきまぜた。 折出した D C TJを庐別し、濃縮後、 MeOHを 加えて沈嚴として^取した。 収量 1.5 0 f ( 87.4 % )。 (XV) Bo c Gin-Met— Leu-: Phe-Arg (Pme)-Gly-Arg (Pme)-ArgC me)-Ala-Ser-Gin-OBu 7 Production Z-Arg (Pme) -Arg (Pme ) -Ala-Ser-G-ln-OBu t 1.0 --Dissolved in MeOH 100, catalytically reduced, and then dissolved in DMF 2 flW. Then, add Boc-Gln-Met-Leu-Phe-Arg (Pme)-(JI7-OH 0.85, HOBt135 5 ^), cool with ice, and add: DCC210 ^ The obtained DC TJ was separated and concentrated, and then MeOH was added to the precipitate to remove the precipitate, yielding 1.50 f (87.4%).
mP. 2 1 6 - 2 1 了。 C (分解) , 〔 な : ¾6 - 1 1.8。 ( c - 1.0 ,m P. 2 16-21 C (decomposition), [Do: ¾ 6 - 1 1.8. (C-1.0,
D M F ) , Bf 1 0.4 3 DMF), Bf 1 0.4 3
元素分析 C98 Hi54 023 if 22 として Elemental analysis C 98 Hi54 023 as 22
計算値: c, 55.09; H,7.27i , 14.42; S,6,00 Calculated values: c, 55.09; H, 7.27i, 14.42; S, 6,00
実験値: C, 54.81 ; H.7.33; U, 14.23; S,5.79 Found: C, 54.81; H.7.33; U, 14.23; S, 5.79
(XVDH-Ljs-Arg-Lys—Arg— Ser— Gln-Me t-Leu-Phe-Arg— Gl -Arg-Arg-Ala-Ser-Gln-OHの製造 (Production of XVDH-Ljs-Arg-Lys—Arg—Ser—Gln-Met-Leu-Phe-Arg—Gl-Arg-Arg-Ala-Ser-Gln-OH
Boc -Gln-Me ~LeU'— Phe -Arg ( me ) -Glj-Arg (Pme ) -Arg (Pme)-Ala-Ser-Gln-OBut ]. Q 9 ΚΤ Έ A 1 0露を加免、 室 温で 5 0分間振]?混ぜたのち、 濃縮し、 ェ-テ を加えて沈澱として泸 取し、 乾燥した 3 Boc -Gln-Me ~ LeU'— Phe -Arg (me) -Glj-Arg (Pme) -Arg (Pme) -Ala-Ser-Gln-OBut]. Q 9 ΚΤ Έ A10 After 5 vibration 0 min] was mixed with warm, concentrated, E -? it was collected泸a precipitate by addition of Te and dried 3
一方、 -Lya、 tr)-Ar ( Pme )-L s (M七 r ) -Ar ( Pme )— Ser -ITHITH-Boc 0.8 3 ^に T F A | 0 «fを加えて室温で 1 0分間振 ]?混ぜたのち、 濃縮し、 ェ -テ を加えて沈澱として泸取し乾燥した。 これを: D M F にとかし、 ドライアイス -アセ トンで冷却後、 On the other hand, add TFA | 0 «f to -Lya, tr) -Ar (Pme) -Ls (M7r) -Ar (Pme)-Ser-ITHITH-Boc 0.83 ^ and shake at room temperature for 10 minutes. After mixing, the mixture was concentrated, added with ether and collected as a precipitate, and dried. Combine this: DMF, cool with dry ice-acetone,
6.2 N HCi /AcOEt 0.2 3 W ,亜硝酸イソァミル( 0.0 7 5 W )を 加え、 - 2 5〜- 2 0 で 2 0分間保った( ヒ ドラジンテス ト :陰挫)。 これを再びドライアイス—アセ ト ンで冷却し、 T E A 0.2 5 «fを加え て中和した。 ァミ ン成分を D M 5 4 0 βにとかし、 氷冷し T E A 0.16 ^を加えたのち、 先に調製したアジド溶液を加えて、 4てで 72時間か 6.2 N HCi / AcOEt 0.23 W and isoamyl nitrite (0.075 W) were added, and kept at −25 to −20 for 20 minutes (hydrazine test: insult). This was again cooled with dry ice-aceton and neutralized by adding TEA 0.25 «f. Dissolve the amine component in DM540β, cool on ice, add TEA 0.16 ^, add the azide solution prepared earlier, and allow 4 hours for 72 hours.
OMPI きまぜた。反応液を、希酢酸水にそそぎ、 折出したゲ を 取し、含水OMPI I mixed. The reaction solution was poured into dilute acetic acid water, and the deposit was removed.
CH3 cirで洗浄した。 収量 ί.40 ( 8 1.5 ^ ) , Bf1 0.0 9。 この うち 40 OW^rO.1 5 M M s Aを舍む τ F A—チオア- y-〃一メチ ス フイド( 8 : ί ·· 1 ) 60 にとかし、 室温で 2時間振]?まぜた のち、 AcOiT¾ 40 0^を加えて濃縮し、 エーテルを加えて沈繫とし て泸取し、乾燥した。 これを少量の 1 AcOHにとかしセフアデック ス G— 25'のカラム ( 2.2 X 1 2 Ocw )に付した。 1 H AcOE で溶 出し 1 60言 - 2 8 の分画を集めて凍結乾燥し、つ でアンバーラ イト I B A - 4 1 0 (薛麵)のカラムを通し、凍結乾燥した。 匕れを. さら-に: E S E - :L S - 4 1 0のカラム ( 2J 4 7.5 c»+ 2.1 4 30 )を用いる H P L Cで精製し、 目的 を得た。 収量 45 ^ Washed with CH 3 cir. Yield ί.40 (8 1.5 ^), Bf 1 0.0 9. Among them, 40 OW ^ rO.15 5 MM s A is melted into τ FA—thioa-y-〃-one-methyl sulfide (8: ί ·· 1) 60, shaken at room temperature for 2 hours, and mixed. AcOiT¾400 ^ was added, and the mixture was concentrated. Ether was added, the precipitate was collected and dried. This was dissolved in a small amount of 1 AcOH and applied to a column of Sephadex G-25 '(2.2 X 12 Ocw). The fraction was eluted with 1 H AcOE, and the fractions of 160 words-28 were collected and freeze-dried, then passed through a column of Amberlite IBA-410 (Seul) and freeze-dried. Furthermore, purification was performed by HPLC using a column of ESE-: LS-410 (2J47.5c + 2.1430) to obtain the objective. Yield 45 ^
、24 o , 24 o
〔 な : ζ— 45.9 ( c = 0.7 , 0.1 N AcOH) [N: ζ— 45.9 (c = 0.7, 0.1 N AcOH)
Bf4 (cellulose) 0.20 ' Bf 4 (cellulose) 0.20 '
アミノ酸分折: I rs 1 ,71 , Arg 4.71 , Ser 1.69, Glu Amino acid analysis: Irs1, 71, Arg 4.71, Ser 1.69, Glu
2.12, Gly 1.00, Ala 1.03, Met 0.32, Leu 1.30, 2.12, Gly 1.00, Ala 1.03, Met 0.32, Leu 1.30,
Phe 1.08 (平均回収率 77 ) Phe 1.08 (average recovery rate 77)
実施例 2 - キヤ リヤ-蛋白とポリべプチド複合钵の合成 Example 2-Synthesis of Carrier-protein and Polypeptide Complex II
実施例 · (χνί) で得たホ°リベプチドとサイログロブリ ン(以下 G- ) の結合を、 グットフレンドらの方法に準じて行 ¾つた(サイエンス , Example · The combination of holibeptide obtained in (χνί) and Cycloglobulin (hereinafter G-) was performed according to the method of Goodfriend et al. (Science,
1 44巻, 1 3 34頁, 1 9 6 4年)。 即ち、 当該ホ。リペプチド 2.5 を T G 3.75^と混合し、 2 の 50 m Mのフォスフエ一トバッファー を加え、氷水中でよく攪拌した。 これにカノレボジイミド塩酸 ¾3 0.4 ¾^ を蒸留水 20 0 に溶かしたものを 1滴ずつゆつく Dと加えた後、 3 時間氷水中で魇拌し がら反応させた。 反応後、 蒸留水で透折を充分に 144, 1334, 1964). That is, the e. Lipeptide 2.5 was mixed with TG 3.75 ^, 2 50 mM phosphate buffer was added, and the mixture was stirred well in ice water. To this, a solution prepared by dissolving canolebodiimide hydrochloride ¾30.4¾ in distilled water 200 was added drop by drop to D. Then, the mixture was reacted for 3 hours while stirring in ice water. After the reaction, make sure that it is transparent with distilled water.
OMPI 一 一 OMPI One one
行 ¾ 、 凍結乾燥を行 つて蛋白複合体 4.7 Wを得た。 The mixture was freeze-dried to obtain 4.7 W of the protein complex.
実施例 3 . Example 3.
抗体検出のための E I A用抗原の調製 Preparation of antigen for EIA for antibody detection
E I A用抗原の調製は北川らの方法( ジャ-ナ ォブ バイオケミ ストリー,第 7 9巻 23 3頁, 1 9 76年)の方法に準じて行った。 The antigen for EIA was prepared according to the method of Kitagawa et al. (Janab Biochemistry, Vol. 79, p. 233, 1976).
Π) ポリぺプチドへのマレイミ ド基の導入 Ii) Introduction of maleimide groups into polypeptides
実施例 1 (XVI)で得たホ。リベプチド( 3.50 n moles)を の E obtained in Example 1 (XVI). Rebeptide (3.50 nmoles)
1 O O mMフォスフェートバッファー pH 6.8 に溶解し、 一(4 力 ボキシシク口へキシ メチ /V )マレイミドの ITーヒドロキシサクシ- ミ ドエステ 5 85 g ( 1.了 5 moles) 70 « の —ジメチ クロマミド溶液に加え混合し、 3 0 で 3 0分攪拌して反応後、 セフ アデックス G - 25カラムで分画を行 いマレイミド基の導入されたホ° リペプチド分画 1 8 5 n moles を得 ¾。 1 Dissolve in OO mM phosphate buffer pH 6.8, and add IT-hydroxysuccinate-midoester of 1- (4-forced hexoxime / V) maleimide 5 85 g (1. 5 moles) 70 «of — dimethyl chromamide solution After stirring for 30 minutes at 30, the mixture was fractionated using a Sephadex G-25 column to obtain a polypeptide fraction having a maleimide group introduced therein, and thus yielded 185 nmoles.
0Ό マレイミ ド基導入ポリペプチドと /9 - Dガラク トシダ—ゼとの結合 実施例 3 Π)で得たマレイミド基導入ポリペプチド ί 6.511 moles と β — T) 一力 'ラウ トシダーゼ 3.3 n moles を混合し、 4 で 1 8時間反 応後、 4 1 2.5 n moles の^ S メノレカプトェタノ— で反応を俘止さ せた。 /? - D -ガラク トシダ -ゼと結合したホ。リベプチドは、 セファロ -ス 6 Bカラムで分画を行¾い、 以下の実験に使用した。 0Ό Binding of maleimide-introduced polypeptide to / 9-D galactosidase Example 3 Maleimide-introduced polypeptide obtained in Π) ί 6.511 moles and β-T) Combined with 3.3 nmoles of lautosidase After 4 hours of reaction at 4 hours, the reaction was stopped with 4 12.5 nmoles of ^ S menolecaptoetano. /?-D-Galacto combined with Toshida-ze. Ribeptide was fractionated on a Sepharose 6B column and used in the following experiments.
実施例 4 · Example 4
E I A法 EIA method
実施例 2で得た蛋白複合体で免疫したマウス血清あるいはハイブリ ド -マ上清中の抗体活注の検出は E I A法を用 て行なった(ィムノブァ -マコロジ— 第 1卷, 3頁, 1 9 78年) す わち、血清ある は ハィブリド -マ上清をバッファ - A ( 20 m M Na2 HP04 , 10 OmM - 2- JTaCl, 0.1 Na¾ , 1 mM MgCl2, pH7.0 ) で希 し、 この 1 00 /^をと])、 実施例 3で得たホ。リぺプチド誘導体 1 0 0 とよ く混合し、 24でで 24時間反 JSさせた。反応後、 ゥサギ抗マウス IgG を結合した 3 セ ロース 1 0 0 を加えて 24 , 4時間反応させ た。 反応後、 セ ースを 0.5 %ツイーン 20をきんだパッファー Aで よく洗淨し、 4ーメチレンペリ フェリ 一 j3一: D—ガラク トシド 20 Antibody injection in serum or hybridoma supernatants immunized with the protein complex obtained in Example 2 was detected by the EIA method (Immunobu-Macologie, Vol. 1, p. 3, 19). 1978) to Wachi, serum some Haiburido - the Ma supernatant buffer - a (20 m M Na 2 HP0 4, 10 OmM -2-JTaCl, 0.1 Na¾, 1 mM MgCl 2 , pH 7.0), diluted with 100 / ^])), the e obtained in Example 3. The mixture was thoroughly mixed with the peptide derivative 100, and was subjected to anti-JS at 24 for 24 hours. After the reaction, 3 celloses 100 conjugated with egrat anti-mouse IgG were added, and the mixture was reacted for 24 or 4 hours. After the reaction, the case was thoroughly washed with Puffer A containing 0.5% Tween 20, and 4-methyleneperi- f j-1: D-galactoside 20
を 5 (3 0 ^ 加え、 3了 で 2時間反応後、 l O O mM力- ' ネートバッファ - C PH 10.5 )を 3 W加えて反応を停止し、上清中の 螢光強度を螢光計で測定した(ェキサイテ-シヨ ン 3 65 nm , シ ヨン 450 nm )。 5 (3 0 ^ added and after 2 hours at 3 Ryo, l OO mM force - 'sulfonate buffer - C P H 10.5) Stop W 3 W addition reaction, fluorescence and fluorescence intensity in the supernatant (Excitation-365 nm, Exciton 450 nm).
実施例 5 Example 5
免 2¾ Exemption 2¾
7 ~ 8週令の BALBZC雌マウス 6 各々に抗原として実施例 2で 得た蛋白複合佯の、 タンパク量として、 40 をフロインドコンプリ - ト ァジュバントとよく混合し、 皮下に接種した(初回免疫)。 初回 免疫の 2週後、 同量の抗原をフロイ ン ドイ ンコ ンプリート ァジュバント とよく混合し、 皮下に接種した(二次免疫)つ さらに、その 2遷後、—二 次免疫と同様の方法で三次免疫を行なつた3 三次免疫の 6日後、 マウス から血液を部分採取し、血清中の抗钵価を実 ¾例 4記載の E I A法で測 定した。 このうち、 高い抗体価を示した r - 2マウスに対して 1 20 g の抗原を 5 ^の食 ¾水に溶解させたものを静脈内に接種することによ 1?最終免疫を行るつた 各マウスの抗^価は表 1に示した 3 Each of 6 6-8 week-old BALBZC female mice was well mixed with Freund's complete adjuvant as a protein, and inoculated subcutaneously (primary immunization). Two weeks after the first immunization, the same amount of antigen was mixed well with Freund's complete adjuvant, and subcutaneously inoculated (secondary immunization). after 6 days of immunization rows Natsuta 3 tertiary immune, blood was partially collected from the mice, was boss measure the anti钵価in serum by EIA method real ¾ example 4 described. Of these, r-2 mice with high antibody titers were inoculated intravenously with 120 g of antigen dissolved in 5 ^ of drinking water to achieve final immunization. anti-^ value of each mouse are shown in Table 1 3
OMPI - - 免疫し ftマゥスの抗ぺプチド抗体価 OMPI --Immunized ft-mass anti-peptide antibody titer
( % ) (%)
1 ) 血清の希釈は 1 Z 1 000 1) Serum dilution is 1 Z 1 000
2) 血清の希釈は 1 ZS 300 2) Serum dilution is 1 ZS 300
3) 血清の希釈は 1 Ζ了 800 3) Serum dilution is 1 end 800
4) 一 :検出不可能 4) I: Undetectable
5) '検索^ f 5) 'Search ^ f
BX : (結合した酵素活性 Z加えた全酵素活性) X 1 00 実施例 6 BX: (Bound enzyme activity Z plus total enzyme activity) X 100 Example 6
細胞融合 Cell fusion
実施例 5記載の方法で免疫を行るい、 最終免疫の 3日後の^ 2マウス から脾藏を摘出し、 ステンレスメッシュで圧迫,炉過し、 ィ -グルズ · ミニマム 'エツセ ンシャ メディウム ( M E M )に浮遊させ、 脬藏細胞 浮遊液を得た。 細胞融合に用 る細胞として、 ; B A L BZCマウス由来 ミエ口 -マ細胞 P 3 - 63 . A 8. ΐ (Ρ3 1 ) を用いた(カレ ント ト ピックス イ ン マイクロ/くィ才口ジー アンド ィムノロジ Immunization was performed by the method described in Example 5, and 3 days after the final immunization, the spleen was excised from the ^ 2 mouse, squeezed with a stainless steel mesh, filtered, and placed in an e-Gluz Minimum 'Etsencia Medium (MEM). The cells were suspended to obtain a cell suspension. A cell used for cell fusion: BAL BZC mouse myeloma-ma cell P3-63.A 8. ΐ (Ρ31) was used (current topix-in-micro / kusaiguchi-g-and-immunology).
' ' 翥 8 1卷 1頁, 1 3了 8年)。飆胞融合は、 原法( ネィチヤ- , 256'卷, 495頁, 1 975年)に準じて行 ¾つた。 即ち、脾藏細胞 およぴ: Ρ 3び 1をそれぞれ血清を含有し ¾い MEMで 3度洗淨し、脬蔵 細胞と P 3 LM数の比率を 5 : 1に ¾るよう混合して、 800回転で 1 5分間遠心を行 つて細胞を沈殿させ 3 上清を充分に除去した後、 沈殿 ¾:籙くほぐし、 45%ポリエチレングリコー〃( : P E G )6000一 ( コッホライト社製)を 0.3 «f加え、 37 温水槽中で了分間静置して 融合を行 つた。 融合後钿胞に毎分 2 «fの割合で H E Mを添加し、合計 1 2*?の3«112£を加ぇた後600回転 1 5分閭遠心して-上清を除去し この細胞沈環 を 1 0 %牛胎 血清を含有する Ε Ρϋ ΐ 1 δ 40メディ ゥム ( Β Ρ ΐί Ι 1 640— 1 0 F C S )に Ρ 31Μが ί W当 2 ifl。 個に ¾るよう浮遊し.、 24穴マ チディ、ンュ( リンブ 社製)に 1ゥェ \ "ずつ I 44ゥエルに播種した。 氇後、細胞を 37 で 5 炭酸 ガスフラン器中培養した。 24時間後、 H A T ( ヒポキサンチン ί X 1 0一4 Μ ,ァミノプテリ ン 4 X 1 Cf'7 Μ ,チミジン ί.6 X ί 0" 5 )ί ) を含んだ P B M I 1 6 40 - 1 0 F C S培地( H A 0?培地)を!ゥェ 当 1M Λ?ずつ添加することによ]?、 H A Τ選択培養を開始した。 HA T 選択培養は、培養開始 3 , 5 , 7日後に旧液を 1 s捨てたあと、 ί の'' 翥 81 1st page, 1st, 13 years 8 years). The vesicle fusion was carried out according to the original method (Neichia, 256 ', p. 495, 1975). That is, spleen cells and 3: 1 each contain serum and are washed three times with MEM and mixed so that the ratio of the number of stored cells to the number of P 3 LM is 5: 1. after thoroughly removing the 3 supernatant centrifuged for 15 minutes to precipitate the means pursuant cells 800 rpm, precipitated ¾: disentangling籙Ku, 45% polyethylene glycol 〃 (: PEG) 6000 scratch (Koch light Co.) The fusion was performed by adding 0.3 «f and leaving it to stand in a warm water bath for 30 minutes. After fusion, HEM was added to the cells at a rate of 2 、 f per minute, and a total of 12? Of 3 112 112 ぇ was added, followed by centrifugation at 600 rpm for 15 minutes, and the supernatant was removed.環 Μ に 1 δ40 medium containing 10% fetal calf serum (Β Ρ ΐί Ι 1 640—10 FCS) Ρ 31 Μ ί W 640 2 ifl. The cells were inoculated into a 24-well multi-layered nut, Nu (manufactured by Limbu), and seeded in I-44 wells at a time. After the incubation, the cells were cultured at 37 ° C. in 5 CO 2 -furan apparatus. after time, HAT (hypoxanthine ί X 1 0 one 4 Micromax, Aminoputeri down 4 X 1 Cf '7 Μ, thymidine ί.6 X ί 0 "5) ί ) containing PBMI 1 6 40 - 1 0 FCS medium ( HA 0? Medium)! By adding each 1M solution, the selective culture of HA Τ was started. In the HAT selective culture, the old solution was discarded for 1 s after 3, 5, and 7 days from the start of culture, and
HA T培地を添加することによ U縫続した。 ハイプリ ド-マの増殖は、 細跑融合後 1 0~ 1 4日で認められ、培養液が黄変したとき(約 1 U suturing was performed by adding HAT medium. Proliferation of hybridomas was observed 10 to 14 days after fusion, and when the culture broth turned yellow (about 1 to 14 days).
1 06 ) 上清を採取し、 E I A法で、 抗体の有無を検索した。 このようにして、 ハイブリ ドーマの増殖が認められた 1 41 ゥエルの上 清を調べたところ、 2ゥェル(ァ 2— 1 し r 2 - ! 00 )に強 抗体 活性, 2ウエノレ( r 2 - 62 , r 2 - 70 )に弱 活注を認めた 3 実議 7 - - クロー: =:ング 1 0 6) supernatant was collected, in the EIA method, a search for the presence or absence of antibody. In this way, when the supernatant of 141 wells in which the growth of the hybridoma was recognized was examined, the antibody activity was strong in 2 wells (a 2-1 and r 2-! 00), and 2 wells (r 2 -62). , r 2 - 70) 3 real discussions showed a weak Katsuchu 7 --Claw: =: Ng
抗体活 が陽性を示した 3ゥェ ( ァ 2 -! 1 , 62 , 1 00 )の各 ハイプリ ドーマ、を、限界希釈法によってクロ- -ングを行るつた。 即ち ハイブリドーマが 2個/ に ¾るよう H PM I 1 S 40 - 20 F C Sに 浮遊させ、 96穴マイク プレート (ヌンク社製)に 1ゥェ 当 0.1 分注した。 分住する際、 プィ —ダ—細胞として B マウ スの胸腺細胞をゥエル当 j? 5 X 1 0" 個にるるよう加えた。 このように して、約 2週間後には細胞の増殖が認められるように ]?、上清を探取 して、 抗体の有無を実施例 4記載の Eェ A法で調べた。その結果、 r 2 - 1 1では 1 9クローン中 8クロー ン , r2 - 62では 54クローン中 3クローン, r 2 - 1 00では 47クローン中 5クローンに抗佯活注を 認めた(表 2 )。 Each of three hybridomas (a2-! 1, 62, 100) showing positive antibody activity was cloned by limiting dilution. That is, two hybridomas were suspended in HPMI1S40-20FCS so as to reach 2 cells / cell, and the suspension was dispensed in a 96-well microphone plate (manufactured by Nunc Corporation) at a ratio of 0.1 per 1 kg. At the time of segregation, B mouse thymocytes were added as puper cells to make up to 5 x 10 "cells. Thus, after about two weeks, cell proliferation was observed. The supernatant was probed and the presence or absence of the antibody was examined by the EA method described in Example 4. As a result, in r2-11, 8 clones out of 19 clones, r2- In 62, 3 out of 54 clones, and in r2-100, 5 out of 47 clones showed anti-honey injection (Table 2).
OMPI 3δ - 表 2 OMPI 3δ-Table 2
クローン化されたハイ _ブリ ドー マの抗べプチド抗侔活拴 Anti-peptide activity of cloned hybridoma
実施例 8 一 — Example 8 One —
モノクローナ 抗钵のェ F ίΓ -ァに^ る結合能 Monoclonal Antibody F F
モノクロ -ナル抗体の I F H - rに対する結合能は、 次の方法で検討 した。 即ち、 ゥサギ抗マウス IgG 抗体を結合させた 3 セ ース溶 液 300 に、 r 2— l l , r2— 62 , r2— ! 00由来のそれぞ れ 2~3種のク -ン細胞の培養上清を各々 300 加え、 室温で The binding ability of the monoclonal antibody to IFH-r was examined by the following method. That is, a culture of 2 to 3 clones of r2-ll, r2-62 and r2--00 cells, respectively, was added to a three-sense solution 300 bound with a rabbit egan anti-mouse IgG antibody. Add 300 pieces of Kiyoshi at room temperature
1 8 ~ 20時間反応させた。 反応後セ -スを生理食塩水でよく珠^ し、 550 ひ の下記によ 2?得た I FN -アを加え、 3 ~ 4時.間反応 さぜた。反応後、上清を採取し上清中の IF IT- r活性をマイクロプレ 一トを用いた細胞変注効果( c ) リ -デング法で測定し 。 (ァプ ライ ド マイクロバイオロジー , 1 6卷, 1 706頁, 1 968年)。 す わち、 96穴マイクロプレー ト (ヌンク社製)全てのクェ に 50 の Μ を入れ、最初のゥェ に I FfTサンプルを 5 Ομ^加えて、 連続的に 2倍希釈を行るつた。 このよ にした各ゥエ に、 W I S H細 胞を 20 ^ F C S含有 M E Mに ί W当 4 1 05 個に るよう調整し た細胞浮遊液 50 を加え、 24時間, 37 ,炭酸ガスフラン器で 培養した。 培養後、 k泡性口内炎ウイルス(二ユ-ジャ -シ -株)を The reaction was performed for 18 to 20 hours. After the reaction, the cells were mixed well with physiological saline, and 550 cells of the IFN-a obtained in the following manner were added, and the mixture was reacted for 3 to 4 hours. After the reaction, the supernatant was collected, and the IFIT-r activity in the supernatant was measured by the cell transfusion effect (c) reading method using a microplate. (Applied Microbiology, Vol. 16, pp. 1706, 1968). That is, 50 μm was put into all the 96-well microplates (manufactured by Nunc Corporation), and IFFT sample was added to the first layer at 5 μm, followed by serial 2-fold dilution. Each © e you this Yo, the cell suspension 50 that so that adjusting the WISH cells in I W those 4 1 0 5 to 20 ^ FCS-containing MEM was added, 24 hours, 37, cultured in a carbonate Gasufuran unit did. After culturing, the vesicular stomatitis virus (Niyuja-shi-strain)
20 0 0 T C I D50 ( テイツ シュ-カ チュアイ ンフエクティングド— ズ 50 )になるよう M E Mで調整し、 その 50 を各々のゥェ に加 え、 3 7 °C,炭酸ガスフラ ン器内で培養した。 約 3 5時間後、 i F irサ ンプ を加えているいゥエルの細胞が I 0 0 C P Eを起こした時点で、 各ゥエルの C P Eを頭徵鏡で観察し、 50 %の C: P Eを起こしているゥ エルのェ ϋ■>サンプルの希釈数の逆数をもって I F ifの力価とした。 Adjust with MEM to 200 TCID 50 (Tissues-Cuche Infection Dose 50), add 50 to each well, and incubate at 37 ° C in a CO 2 gas flannel. did. Approximately 35 hours later, when the cells in the wells to which the iFir sample was added developed I00 CPE, the CPE in each well was observed with a cranioscope and 50% C: PE was developed. The IF if titer was the reciprocal of the dilution number of the sample.
ェ F N - rサンプルとして用いたものは、 ヒ ト末梢^リ ンパ球を、 コ ン力ナパリ ン A 40 /ulと 1 2 - 0ーテトラデカノィノレホルボ一ノレ - 1 3ァセテ " ト 1 5 ng で刺激して了 2時間後に採取した上清で、 The FN-r sample used was a human peripheral lymphocyte, which had a naphthalin A 40 / ul and a 12-0-tetradecanoinyl phorbo-1-13 acetate 1 2 hours after stimulation with 5 ng,
O PI — ΰ δ— O PI — Ϋ́ δ—
この培 ¾_b清 1 *中にはヒト —ァ(酸, ρ Ε処理に不安定)ま' 4400ュ-ット含有して た。 - じ、 クローン細胞の培養上清中に ιΐΊΤ-rに対して結合能を持つ抗体が存在して れば、後で加えた I F IT - r活 は、 セ ロ-ス上の抗体に結合し、上清中の活性は低 減される箬である: > 結杲は、 r 2 - 1 1のクロ-ンの上清中に比較的 強 I N - τ結合能が認められ、加えた I P IT - r ( 550 The culture medium contained human cultures (acid, unstable in ρ Ε processing) and 4400 cuts. -If an antibody capable of binding to ιΐΊΤ-r is present in the culture supernatant of the cloned cells, IFIT-r activity added later binds to the antibody on cellose. In addition, the activity in the supernatant is reduced:> Yugo shows relatively strong IN-τ binding ability in the supernatant of r2-111 clone, and the added IP IT -r (550
の 50 ~ 7556が抗体に結合した(表 3 )。 Of the antibodies bound to the antibody (Table 3).
表 3 Table 3
モノク ーナ 抗体による IF - T活拴の吸収能 Absorption ability of IF-T activity by Monoclonal antibody
実施例 9 Example 9
モノクロ一ナル抗体産生ハイブリ ド一マの腹水化 Ascites in hybridomas producing monoclonal antibodies
I F ir - rに対して結合能を示す抗体を産生する r 2 - 1 1 . 〗クロ 一ン細胞 1 X 10° 個を、 あらかじめ のミ.ネラルオイルを腹腔内 に投与しておいた B A L BZcマウスの腹腔内に接種することによ 腹 ^化を行った。 ハイブリ ド-マを腹腔に投与して〗 0日後、 腹水を採取 して抗钵価を E I A法で測定したところ、 ί 07 倍希釈まで抗体活性を 示した。 、 当該クロ-ン細胞の培養上清中の抗钵活拴は、 〗 04倍 希釈まで認められているが、 腹水化することによ 約 1 0 0 0倍程度抗 体活性が上昇した。 BAL BZc mice that have been administered intraperitoneally with 1 x 10 ° clone cells in advance of mineral oil intraperitoneally, producing r 2-1 1. Antibodies capable of binding to IF ir -r By inoculating intraperitoneally I made it. Hybrid -〗 0 days after the administration of Ma intraperitoneally, where the anti钵価was collected ascites was measured by EIA method, showed antibody activity up to I 0 7-fold dilution. , The black - anti钵活拴in the culture supernatant of the emission cells are recognized to〗 0 4-fold dilution, about 1 0 0 0 times antibody activity by to ascites rises.
実施例.10 Example 10
モノクロ -ナ^体の精製 Monochrome-Purification of Na ^ body
実施例 9で得られた腹永 4 Wを出発材耔として、 ステーリンら(ジャ ーナ〃 ォブ バイオ ジカ ケミス ト リー , 25 6巻, 975 0頁, 1 3.8 1年)の方法に準じてモノクロ-ナ 抗体を賴 した。 まず腹水 からフイブリ ン様称質を除去するため 1 0 , 0 0 0回転 1 5分間遠心し た後、 リン酸緩衝液一食塩水( P B S : 8, ί m -lTaH2P04 , 1.5m3i ΕΗ2Ρ04,2.7ηιΜ KCl, 137mM kaCl, H 7.2 ) 2 80 n m <D 紫 都吸収 ( A 280 ) が 1 2 ~! ½を示す漢度に希釈した = 希釈後 サンプノレに飽和硫酸アンモ-ゥム溶液を 47 %の濃度にるるように加え、 4 °Cで攙拌しながら 6 0分間塩柝を行 、その後遠心( 1 G,C) (3 0回 転, 1 5分間)を行るつて沈澱^を得た。 沈澱物を 5 0 mM liaCl含有 2 0 m Mトリス緩衝溶液( p H 7.9 )に溶遊し、 同溶液 2 に対して透 折を行 つた。 2時間後、 2 の新しい同じ透析液に換 、 さらに ί 5 時間透析を行 つた。 透析後、 沈殿を除去するため 1 0 , 0 0 0回転 Using 4 W of the peristalsis obtained in Example 9 as a starting material, according to the method of Stalin et al. (Jana Biob Zika Chemistry, 256, 9975, 13.81). Monoclonal antibodies were tested. First After centrifugation 1 0, 0 0 0 Rotation 1 for 5 minutes to remove Fuiburi down like universal quality from ascites, phosphate buffer one meal brine (PBS: 8, ί m -lTaH 2 P0 4, 1.5m3i ΕΗ 2 Ρ0 4, 2.7ηιΜ KCl, 137mM kaCl, H 7.2) 2 80 nm <D purple Metropolitan absorption (a 280) is 1 2 ~! Diluted to a degree that indicates 度 = After dilution Add saturated ammonium sulfate solution to Sampnole to a concentration of 47%, perform stirring for 60 minutes while stirring at 4 ° C, and then centrifuge ( 1 G, C) (30 rotations, 15 minutes) to obtain a precipitate ^. The precipitate was dissolved in 20 mM Tris buffer solution (pH 7.9) containing 50 mM liaCl, and the solution 2 was subjected to filtration. Two hours later, two new same dialysates were used and dialysis was performed for another 5 hours. After dialysis, 100,000 rotations to remove precipitate
1 5分間遠心を行ない、上清を Α280 の値が 2 0 ~ 3 0の濃度になる ように調整した。 このサン: 7 レを充分量の 5 0 m Μ - ITaCl含有トリス 緩^^液で》| (ヒした 8 atの セノレ口 -スカラム ( ワッ トマン Centrifuge for 15 minutes, was adjusted so that the value of the supernatant Alpha 280 is a concentration of 2 0-3 0. This sun: 7 m with a sufficient amount of 50 m Μ-Tris buffer containing ITaCl >> |
' D Ξ52)にかけ、 5 0 m M NaCl含有トリス緩衝溶液を用 て 1.5 a 分の流出速度で分画を行 つた。 この条件下では、 抗体活性は主として 'DΞ52), and fractionation was carried out using a Tris buffer solution containing 50 mM NaCl at an outflow rate of 1.5 a. Under these conditions, antibody activity is primarily
O PI 素通]?分画に認められた(図〗 )。 抗体の確認にはラエムリらの方法 (ネイチヤー,第 227卷, 680頁, ί 970年)に準じて S D S - ポリアクリ〃ァミ ドゲ 電気泳動( S S -PAGE )法を用 た。す ¾わち、 硫安^; 一 セ ロースカラムで分画したいぐつかの フラクションを、 各々 2 -メ カブトェタノ一 で還元を行¾ 1 7 % S D Sゲル ;30ボルト, 24時間泳動を行 ¾つた。その結杲、抗佯活性に —致して、分子量約 55キロダ トン前後に E鎖、約 28キロダ〃トン 前後に I·鎖の 2つのバンドが認められた(図 2 )。 のように精製され た抗体フラクション 1 了が — rに对して、 結合能を^^どうか を実施例 8の方法に従って I F - r 2200 σ を加えて検索し たところ、 ェ - rの約 50 が抗体に結合 &を示すことが判明した (表 4 )。 表 4 O PI 通] was recognized in the fractionation (Fig.〗). Antibodies were confirmed by SDS-polyacrylamide gel electrophoresis (SS-PAGE) according to the method of Laemli et al. (Naturia, Vol. 227, p. 680, ί970). That is, several fractions fractionated on a cellulose ammonium sulfate column were reduced with 2-methabetethanol, respectively, and subjected to 17% SDS gel; 30 volts, 24 hours electrophoresis. As a result, two bands, an E-chain and a I-chain, were found at a molecular weight of about 55 kDa and about 28 kDa, respectively (Fig. 2). The antibody fraction purified as described above was compared with — r , and the binding ability was determined by adding IF-r 2200 σ according to the method of Example 8 to find that the binding ability was about 50%. Showed binding & to the antibody (Table 4). Table 4
•実施例 11 Example 11
モノクローナル抗俘の属するサブクラス Subclass to which monoclonal antibodies belong
OMPI - - τ2-\し ί¾ノク口-ナル抗体の属する IgG サブクラスは、実施例 1 0の方法で精製したフラクション 1 7を 1 0倍^希釈し、 ャギ抗マウ ス IgG〗, G2a,G2b , G3 抗体(マイルス社)との寒天内沈降反応 法(ォクタ口-一法:ィムノロジカ メソッ ド .,{ γ? r ゲ 一ディ フュージョン テクニック , ブラックゥェ , オックスフォー ド ,OMPI --The IgG subclass to which the τ2-\-nocchi-null antibody belongs was 10-fold diluted the fraction 17 purified by the method of Example 10 to obtain goat anti-mouse IgG〗, G2a, G2b, Precipitation reaction in agar with G3 antibody (Miles) (Octa-orchid-method: immunologica method., { Γ? R Ge fusion fusion technique, black black, oxford,
1 9 64年)で検討した。 結果は、 モノクロ -ナ 抗体とャギ抗マウス1964). The result was a monoclonal antibody and goat anti-mouse.
IgG2b 抗体との間に著明な 1つのバンドが認められ、 他の抗抗体との 間には、 バンドの彤成はみられ かった。 従って、当該モノクロ ナ 抗体は、 IgG2bに属するものであることが判明した('表 5 )。 One prominent band was observed with the IgG2b antibody, and no band formation was observed with the other anti-antibodies. Therefore, the monoclonal antibody was found to belong to IgG2b ('Table 5).
表 5 Table 5
モノクロ一ナ;^抗侔の属するサプクラス Monochrome; ^ class belonging to ^ 侔
実施例 12 , Example 12
実施例.10の要領で精製された素通]?分画のモノ ク ローナル抗体 2 5 I ( 6 5.3 ^ )を 0.1 M NaHC03 , pH 8: 3 溶液に対して一晚透析を行つ た。 —方、 アブイゲル一 1 0 (バイオ ' ラ ド社) 2 5 をグラスフィル ターを用いて充分水洗を行った後、 0.1 M NafiCO3 p H 8.3溶液に浮 遊させ前記抗体とを混合し、 4 'Cで 4時間、 ゆつく ]?攪拌し がら反応 させた。 その後、 4 Cで -晚放置した後、 グラス フィルターを用いて ? Example Motodori purified in the manner of .10] fractions mono click polyclonal antibodies 2 5 I (6 5.3 ^) the 0.1 M NaHC0 3, pH 8: having conducted an晚dialyzed against 3 solution . After washing the Abuigel 10 (Bio'Rad) 25 thoroughly with a glass filter, float it in a 0.1 M NafiCO 3 pH 8.3 solution and mix with the antibody. 4 hours at 'C.] The reaction was carried out with stirring. After that, let it stand at 4 C for-晚 and then use a glass filter
O PI 0.1 M liaHCOg pH 8.3溶液でよく洗净した。 反応させたゲ に 0.1 Mエタノールァミン, 0.1 5 M NaClを含む溶液( H 8.0 ) ¾: 25 ^ 加え、 4 , 1時間振とうし、残存するかも知れな 未反応の活性基を ブロックした。 その後ゲルを P B Sでよく洗浄し、 0. l «HaN3を含む P B S 25»に懸濁し、 4 で保存した。 加えた抗体量と回収された泸 液中の抗体量から、 ゲ 1*当!) 2.35« の抗体が結合して ることが 判明した。 この様にして得た反応物をカラムに充め、抗体カラムとして 用した。 O PI The well was washed well with a 0.1 M liaHCOg pH 8.3 solution. A solution containing 0.1 M ethanolamine and 0.15 M NaCl (H 8.0) ¾: 25 ^ was added to the reacted gel, and shaken for 4.1 hour to block unreacted active groups that might remain. Thereafter, the gel was thoroughly washed with PBS, suspended in 0.1 l PBS 25 containing HaN 3 and stored in 4. From the amount of antibody added and the amount of antibody in the recovered solution, 1 * equivalent! It turned out that 2.35% of the antibodies were bound. The reaction product thus obtained was packed in a column and used as an antibody column.
実施例 13. Example 13.
参考例 4で得た上清 60 を 1 m M E D T A , 0.1 5 M NaClを含む Supernatant 60 obtained in Reference Example 4 contains 1 mM EDTA and 0.15 M NaCl
20 m Tris -HC1 , pH 7.6 ( T E H )で 1 5 に稀釈したのち、 実施例 1 2の方法で調製した抗 I F N— r抗体カラム( にかけた。 After dilution to 15 with 20 m Tris-HC1, pH 7.6 (TEH), the mixture was applied to an anti-IFN-r antibody column (prepared by the method of Example 12).
T E Nで十分洗浄したのち、 さらに 0.01 «ノ -デット P— 40 After thorough washing with TEN, an additional 0.01 «
( Shell社製) 0.5 M NaClを含む T E Nで洗浄した。次に 0.25 M NaClを含む 0.1 M酢醸で I F N— rを溶出し、溶出液はただちに 1 M トリス HC1 pH 7.6で中和した。得られた溶液を蒸留水に対し 4わ, 1 6時間透析し、 これをァセト ン一ドライアイスで凍結後凍結乾燥に付 し粉末とし 。 (Shell) Washed with TEN containing 0.5 M NaCl. Next, IFN-r was eluted with 0.1 M vinegar containing 0.25 M NaCl, and the eluate was immediately neutralized with 1 M Tris HC1 pH 7.6. The resulting solution was dialyzed against distilled water for 4 hours and 16 hours, frozen with acetone-dry ice, and freeze-dried to obtain a powder.
結果は iiTFの通]?であった。 The result was iiTF?].
OMPI ― ― OMPI ― ―
蛋白質 (W) 全活性(ϋ) 比活性 (Ιί/^) 回収率 ( 溶菌液上清 250.8 4 XI 08 1.6 x106 一 抗体カラム処理 2.0 1.5 x108 7.5 X107 37.5 ここで得られた最終のヒト免疫ィンターフェロン蛋白質の比活性( W I S H細胞に対する V S Vの細胞変性効果阻止試験によるウィルス活性 測定による(前出) )は 7.5 X 1 07 Xi/ であった。 この蛋白質の性 状は下記のとお]?であった。 Protein (W) Total activity (ϋ) Specific activity (Ιί / ^) Recovery (Lysate supernatant 250.8 4 XI 0 8 1.6 x10 6 1 Antibody column treatment 2.0 1.5 x10 8 7.5 X10 7 37.5 (by viral activity measurement by cytopathic effect inhibition test of VSV for WISH cells (supra)) the specific activity of the human immunodeficiency I centers Feron protein was 7.5 X 1 0 7 Xi /. of the protein sex shape below Too]?
ヒト免疫ィンターフェロン蛋白質の性状 Properties of human immune interferon protein
(0 分子量 (0 molecular weight
実施例 1 3で得られた蛋白質を 2—メ カブトエタノー で処理して The protein obtained in Example 13 was treated with 2-butane ethanol.
S D S—ボリアタリ/ uアミドゲ ( 1 7.5 * )電気泳動( 1 5 mV , 6 時間)にかけ、 クマジーブルーで染色したところ、 蛋白質は単一のパン ドとして確認できた(図 3 )。 お同時に泳動させた分子量マーカーの 泳動距離と蛋白質の泳動距離との闋係から蛋白質の分子量は 17 , 000土 1,000 と推定された(図 4 )。 一方、 2—メ カブトエタノー で処 理しなかった蛋白質は分子量 33, 000 ± 2,000の位置にもう一本のバ ンドが検出された。 この値は I F N— rの分子量 17,000±1 ,000 の 約 2倍に相当することから、 このバン ドは I F N— rの二量体に由来す ると考えられる。 The protein was identified as a single band when subjected to SDS-boriatari / u amide (17.5 *) electrophoresis (15 mV, 6 hours) and stained with Coomassie blue (FIG. 3). From the relationship between the migration distance of the molecular weight marker and the migration distance of the protein, which were simultaneously electrophoresed, the molecular weight of the protein was estimated to be 17,000 soil 1,000 (Fig. 4). On the other hand, another band was detected at a molecular weight of 33,000 ± 2,000 for the protein that was not treated with 2-meadow ethanol. Since this value corresponds to about twice the molecular weight of IFN-r of 17,000 ± 1,000, this band is considered to be derived from the IFN-r dimer.
(B) アミノ酸分析 (B) Amino acid analysis
実施例 1 3で得られた蛋白質をガラス製加水分解用試験管にと]?、 2 0 0倍量( V/W )の 4 チォグリコー 酸を含む定沸定塩酸を加え て 減圧下に封管したのち、 1 1 0わで 2 4 , 4 8 , 7 2時間加水分解 した。 加水分解後、開管し、塩黢を滅圧下で除去し、残渣を 0.0 2 N¾ 酸に溶解して日立製 8 3 5型高速アミノ酸分析計によ])アミノ酸分析を 実施した。 Transfer the protein obtained in Example 13 to a glass hydrolysis test tube], add 200 volumes (V / W) of constant boiling constant hydrochloric acid containing 4 thioglycolic acid, and seal the tube under reduced pressure. After that, the mixture was hydrolyzed for 24, 48, and 72 hours with 110 watts. After hydrolysis, open the tube, remove the salt under reduced pressure, dissolve the residue in 0.02 N acid, and use Hitachi 8335 high-speed amino acid analyzer to perform the amino acid analysis. Carried out.
シスチンおよびシスティンは、 Hi rsの方法〔 Methods EnzymoL 1 1 , 1 9 7 ( 1 9 67 )〕に従い、上記蛋白質を過ギ酸酸化したのち、上記 と同様に 2 4時間加水分解して、 ァミノ酸分析計によ]?システィン黢と して定量した。 アミノ酸分析値は、 2 4 , 4 8および 7 2時間の加水分 解で得られた値を平均して求めた。伹し、セリ ン , スレオ-ン,チロジ ンぉよびトリブトブァンの値は加水分解時間を 0時間に外挿して求めた。 その結果を第 6表に示す。 第 6 表 検出アミノ酸 モ ) 検出アミノ酸 モ « ァスパラギン酸 13. 4 メチ才ニン 2. 9 スレ才ニン 3. 6 イソロイシン 4. 8 セリ ン 7. 4 ロイ シン 6. 8 グ タミ ン酸 12. 5 チロジン 3. 4 プ Π リ ン • :| フエ = ヲニン 6. 7 グリ ^ン 3. 8 :: リジン 13. 5 ァラ =ン 5. 3 ヒスチジン 1. 5 システィン酸 1. 3 アルギニン 5. 4 パリ ン 5. 7 ト リ プトファン 0. 9 Cystine and cystine were subjected to the formic acid oxidation of the above protein according to the method of Hirs [Methods EnzymoL 11, 197 (1967)], and then hydrolyzed for 24 hours in the same manner as described above to analyze amino acids. By all means]? Quantified as Sistine II. Amino acid analysis values were determined by averaging the values obtained after 24, 48 and 72 hours of hydrolysis. On the other hand, the values of serine, threon, tilidine and tributane were determined by extrapolating the hydrolysis time to 0 hours. Table 6 shows the results. Table 6 Detected Amino Acids) Detected Amino Acids 1 .3 Aspartic acid 13.4 Methine 2.9 Sleinine 3.6 Isoleucine 4.8 Serine 7.4 Leucine 6.8 Gutamate 12.5 Tyrosine 3.4 Purin •: | Hue = Panin 6.7 Glyin 3.8: Lysine 13.5 Phala = 5.3 5.3 Histidine 1.5 Cysteinic acid 1.3 Arginine 5.4 Parin 5.7 Tryptophan 0.9
ΟΜΡΙ 一 b— (画) ァミノ末端アミノ酸分析 ΟΜΡΙ One b— (Picture) Amino terminal amino acid analysis
実施例 5で得られた蛋白 ¾を Hirsの方法〔 Methods EnzymoL 11, The protein obtained in Example 5 was used according to the method of Hirs [Methods EnzymoL 11,
197(1967) 〕に従って過ギ酸酸化したのち、 エ ドマン分解法の岩永 らによる変法〔 Eur. J. Bioohem. , 8 , 189 ( 1969) 〕によ]?その ァミノ末端アミノ酸分析を実施した。 生成したフエ- チオヒダン トイ ンーアミノ酸( P T H—アミノ酸)はウルトヲスフェアー 0 D Sカラム 〔ア テックス社製( U. S. A. ) . 46 X 2 50 ram ,粒子怪 5 〕を 用 、 Archerの方法〔 Altex Chromatogram, 3 , 8 ( 1980 ) 〕に よ])、バリアン製高速液体クロマ トグラフ 5 040型( U. S. )で同 定,定量した。 その結果、 P T H—メチォ -ンスルホンおよび P T H一 システィ ン酸が検出された。 上記実施例よ]?明らか ように本発明の精製法によれば、 7.5x107 以上の比活性を有する実質的に純粋るヒ ト免疫ィ ンターフェロン 蛋白質を製造することができる。 197 (1967)], and the amino acid amino acid analysis of the Edman degradation method by Iwanaga et al. [Eur. J. Bioohem., 8, 189 (1969)] was performed. The produced thio-thiohydantoin-amino acid (PTH-amino acid) was purified using an Ultrasphere 0 DS column (manufactured by Atex (USA). 46 x 250 ram, particle phantom 5) using the Archer method [Altex Chromatogram, 3 , 8 (1980)]]), and were quantified using a Varian high-performance liquid chromatograph Model 5040 (US). As a result, PTH-methionone sulfone and PTH monocystic acid were detected. As is clear from the above examples, according to the purification method of the present invention, a substantially pure human immunointerferon protein having a specific activity of 7.5 × 10 7 or more can be produced.
OMPI IFO 以下に r- 3マウスよ]?得た ノクローナ〃抗佯を用いた成績を示す。 突翻 1 OMPI IFO The following shows the results obtained using the Noclona II anti-fly. Abrupt 1
免疫 Immunity
実施例 5の表 1に記载の r - 3マウスに、二次,三次と同様の方法で 四次免疫を行 ¾い四次免疫の 2逦後、 実 ½ 5と同様に 120 igの抗 原を 0.5 の食 水に溶解させたものを静腺内に接種するととによ]?最 終免瘐を行なった。 Fourth immunization was performed on the r-3 mice described in Table 1 of Example 5 in the same manner as in the second and third immunizations, two days after the fourth immunization, and 120 ig as in the fifth example. It was inoculated in the thymus with a solution prepared by dissolving the raw material in 0.5% saline.].
実施例 15 Example 15
細胞融合 Cell fusion
実施例〗 4に記載した最終免疫の 3日後のァ - 3マウスから脾醭を摘 出し、 実施例 6記載の方法で細胞融合 ¾らびに H A T選択培養を行 ¾つ た。その結果 43ゥェ に、 ハイブリ ド-マの增殯を認め、実旌例 4に 記載の E I A法で抗体の有無を検索したところ、 2ゥェ ( r 3 - 11, r 3— 1 9 )に強 抗体活性を認めた。 Three days after the final immunization described in Example I-4, spleen was excised from the mouse, and cell fusion was performed and HAT selective culture was performed by the method described in Example 6. As a result, in March 43, hybridoma was found to be buried, and the presence or absence of antibodies was searched for by the EIA method described in Jeongseon-jo 4. The result was 2 ゥ (r3-11, r3—19). In addition, strong antibody activity was observed.
実施例 16 Example 16
ク 13 _ =ング 13_
抗体活拴が強陽性を示した 2ゥェ〃( r 3 - 1 ! ,ァ 3- ! 9 )の各 ハイブリド-マを、 実 例 7記載の方法でクロー-ングを行 ¾つた。そ の結果、 7* 3 - 1 1では 21クローン中 9クロ -ン ,ァ 3-, 9では 25クローン中〗 5クロ-ンに抗侓活性を認めた(表 7 )。 — — Each hybridoma (r3-1 !, a3-! 9) showing strongly positive antibody activity was cloned by the method described in Example 7. As a result, 9 * clones out of 21 clones in 7 * 3-11, and〗 5 clones out of 25 clones in 3-3- and 99 showed anti-tumor activity (Table 7). — —
クロ-ン化されたハイグリ ドーマの Of cloned highgly dorma
抗ペプチド抗体活性 ノヽイブリ K - -マ ot B / T ( ) Anti-peptide antibody activity Noiburi K--ma ot B / T ()
o o
T 3 1 I I T 3 1 I I
7 7 7 7
0 1 I 0 1 I
Λ Λ
C 7 C 7
0 o 0 o
7 7 f g Q 7 7 f g Q
8 1 9 8 1 9
H 78 H 78
1 7 1 1 I 1 7 1 1 I
16 8 1 16 8 1
r 3 - 1 9 2 85 r 3-1 9 2 85
3 88 3 88
5 83 5 83
7 80 7 80
3 . 77 3. 77
12 75 12 75
13 T 3 13 T 3
1 76 1 76
16 80 16 80
17 8 1 17 8 1
18 90 18 90
19 85 19 85
20 9 1 20 9 1
23 8 t 23 8 t
24 82 24 82
i0 実施例 17 i 0 Example 17
モノクロ-ナ 抗侔の I F 21 -ァに^ fる結合能 Monoclonal anti-IF 21-A binding ability
モノク -ナ 抗体の I F H - rに対する結合能は、実施倒 8に記載 の方法で検討した。 但し、本実施例で用いた 15· κ-rサンブ は、 ヒ ト I F N -ァ遣伝子をブラスミドに組み で大腸菌で努現させたもの The binding ability of the monoclonal antibody to IFH-r was examined by the method described in Example 8. However, the 15 · κ-r sambu used in this example was obtained by combining human IFN-transducer with brassmid and working on E. coli.
( リ コンビナント i Fir - r ;参考例 4参照)で、 1 1 00 uZWに ¾ るよう調製した。結果は、 Γ 3- 1 !.1 , Γ 3- 1 9·20ク -ンの上 清中に強 I Fir - r結合能が認められ、 加えた I ϊ·ΐϊ- 7·の約 90 が抗体に結合した(表 8 )。 表 8 モノクロ -ナ〃抗体によるリコンビナン (Recombinant i Fir -r; see Reference Example 4). As a result, strong I Fir -r binding ability was observed during the purification of Γ3-1-1..1 and Γ3-192 Bound to the antibody (Table 8). Table 8 Recombination with Monoclonal Antibody
i F ir- r活性の Κ収能 iFir-r activity
実施例 18 Example 18
モノクロ -ナ 抗体の属するサブクラス Monoclonal-Subclass to which the antibody belongs
ァ 3- 1 ,.1 ,ァ 3- 13.20ク -ン钿跑を、 実施例 9に記 *の方 法で腹水化を図!)、 得られた腹水につき、 実施例〗 0記 «の方法で精製 を行 ¾つた, 得られたモノクロ -ナ 抗佯のサブクラスは、実施倒, 1 記¾の寒天内 2»反応法 討した。 結果は、 7· 3 - ί 1.1 , r 3 - 1 9.20何れの ノクロ-ナ 抗体とも、 ャギ抗マウス Ig(Jl抗体との 間に著明なバンドが認められ、池の抗抗体との間には、 パンドの彤成は As shown in Example 9 for Asa-1, .1, Asa 13.20 钿 跑, ascites can be created by the method of *! ), The ascites obtained was purified by the method described in Example I, Example 0, and the obtained subclass of Monoclonal-anti-Fyle was defeated. . The results showed that a significant band was observed between goat anti-mouse Ig (Jl antibody) and that of both 7-3- -1.1 and r3-19-1.20 noclonal antibodies. The formation of Pando
OMPI -4 - みられなかった。 従って、 r 3 - 1 1.1 r 3 - f 9.20モノクローナ 〃抗体は、 igG lに属するものであることが判明した(表 9 )。 表 9 OMPI -4-I didn't see it. Therefore, the r3-11.1 r3-f9.20 monoclonal antibody was found to belong to igGl (Table 9). Table 9
モノクロ -ナ 抗体の属するサブクラス Monoclonal-Subclass to which the antibody belongs
実施例 19 Example 19
E I A法による I F N - Γの定量法 I Determination of IFN-Γ by EIA method I
モノクロ-ナ 抗体に対する fej I F rと (b)固相に固定された I F W - Γとの競合による定量法 Quantitative method by competition between fej I Fr for monoclonal antibody and (b) I FW-Γ immobilized on solid phase
実施例 1 3で示された大腸菌で発現させえ後精製した i F K- ^ 1 5 gZWになるよう 0.1 M重炭駿ナトリウムを含有したリン酸緩衝 Phosphate buffer containing 0.1 M sodium bicarbonate to express i F K- ^ 15 gZW expressed in E. coli as shown in Example 13
( p H 8.0 )に浮遊させ、 この 1 00 を 96ウェ マイク ブレ -トの各ゥエ に分注し、 4 で 24時間反応させた。 反応後、 ゥェ の余剰の結合部位をふさぐため 2 牛愈澝ァ ブミ ン含有リン酸锾衝液 を 1 0 0 /< ずつ分注し、 4 Cで 24時間 理し、 E L I S Aに使用す (pH 8.0), this 100 was dispensed to each tube of a 96-well microphone plate, and reacted at 4 for 24 hours. After the reaction, dissolve 100 μl / volume of phosphate buffer containing 2 cow beabumin in 100 / <each to block the excess binding site of the gel, process at 4 C for 24 hours, and use it for ELISA.
O ?I るブレ-トを侏製し 。以上のよ に調製したブレートに、 最大量 する抗体の約 50 量の抗体を含む 1 00 ^ の抗体 r 3 - 1 1.1 ) 溶液と種々の濃度( 75 , 3 X! 02 , 1.25 103 , 5 1 03 ,O? I Made a brat. More in Bureto, prepared Yo, 1 contains about 50 amount of the antibody of an antibody that maximum amount 00 ^ antibody r 3 -! 1 1.1) solution with various concentrations (75, 3 X 0 2, 1.25 10 3, 5 1 0 3 ,
2 1 04 , 8 x 1 04 /W)に調製したリコンビナント I PK— r を 37 で〗時閭反応させた混合欲を加え、 室温で 3時間反応させた。 反応後リン酸緩衝欲でよく洗浄し、 ホースラデイシュペ オキ^ダ-ゼ でラベ したャギ抗マウス igG抗体を 1 00 加え、 室温で 3時間 反応させた。反応終了後、 ゥェ をリン酸 3»銜液でよく洗浄し、 1 の 0, 1 ϋクェン酸緩衝液に 22 «5?の才 ソプェ-レンジァミ ン , 1 の過酸化水素を加えた基質溶欲 1 00 を加えて酵素反 を室温で 1 5分行 、 4規定硫酸で反応を浮止させた。 俘止後、 マ チスキヤ ン(タイ ·ターテック社製)を用いて波長 492 nmで色素量を定量し た。 図 9は、その結果を示したもので、本 E L I S A法を用 ることに よ 3ズ , 02 ~5ズ 1 03 F ΙΪ - r含有液中の I F ir - r が ¾aされることが判明した。 A mixture of the recombinant I PK— r prepared at 2 × 10 4 and 8 × 10 4 / W) was allowed to react at 37 ° C. for 3 hours, and reacted at room temperature for 3 hours. After the reaction, the well was washed well with phosphate buffer, 100 g of a goat anti-mouse igG antibody labeled with horseradish oxidase was added, and the mixture was reacted at room temperature for 3 hours. After the completion of the reaction, wash the well with phosphoric acid 3 »mouth liquid, and add a substrate solution obtained by adding 22 5? -Year-old soap-diamine and 1 hydrogen peroxide to 1 10,1 1-citrate buffer. After adding greed 100, the enzyme reaction was carried out at room temperature for 15 minutes, and the reaction was suspended with 4N sulfuric acid. After detention, the amount of dye was quantified at 492 nm using Matiskian (Tertec, Thailand). Figure 9 shows the results. Using this ELISA method, 3 and 0 2 to 5 10 3 It was found that IF ir -r in the F ΙΪ -r-containing solution was ¾a.
実細 20 Real 20
E I A法による I P N一 rの定量法 H Quantitative method of IPN-r by EIA method H
モノクロ -ナ 抗佯に対する fe) I F 2i-rサンブ と fc)薛素標識した 本発明ぺブチドとの銃合による定量法 Monoclonal-na Quantitative determination of fe) IF 2i-r sambu and fc) Xuesin-labeled ぺ -butide for anti-honey
実旌例 20 -Π) Jeongjeong example 20 -Π)
参考例 4で得られたリコンビナント Ι ΡΪΤ-Τを種々の ¾度( 64 , The recombinant Ι ΡΪΤ-Τ obtained in Reference Example 4 was converted to various degrees (64,
320 , 1.6 103 , 8 1 03 , 5 x 104 , 2 105 S/ml) に調製し、 この 1 00 * を一定量の r 3 - 1 1.1 , r 3 S.20 抗 体含有液 100 と室温で 2時間反応させた。 これに実旌例 3で得ら れた酵素結合ボリぺブチド薛導体を 1 00 加え、 4 で 48時間反 一 - 応させた。 反応終了後、 この混合物を、 ゥサギ抗マウス I g G抗体を結 合させた 3 ίセ ロース溶 ¾Π 00 に加え、 室温で 4時間反応させ た。 反応後、 実施例 4で記載したのと同様の方法で ^反おを行¾ 、 螢光強度を測定した。 その結果、 モノクロ -ナ 抗体( r 3 - 1 1.1 , r 3 - 19.20 )への酵素結合ボリぺ チド靜導体の結合は、 320TT の I P N - 存在下でわずかに阻害され、 8 1 03 u 存在下では、 ほぽ完全に阻害された(図 ί 0 )。 従って本方法を^ するととによ]?、 320 - 8 x 1 03 S /miの τ i* if -ァ含有液中の I i1 rの定量が 可能と ¾つた。 320, 1.6 10 3, 8 1 0 3, 5 x 10 4, 2 10 5 S / ml) to prepare, the 1 00 * a certain amount of r 3 - 1 1.1, r 3 S.20 antibody-containing solution The reaction was carried out with 100 at room temperature for 2 hours. The enzyme-conjugated polysaccharide obtained in Example 3 was added to this, and added for 4 hours. I responded. After the completion of the reaction, this mixture was added to 3ί cellulose solution 00 conjugated with a perforated anti-mouse IgG antibody, and reacted at room temperature for 4 hours. After the reaction, the reaction was repeated in the same manner as described in Example 4, and the fluorescence intensity was measured. As a result, monochrome - Na antibodies (r 3 - 1 1.1, r 3 - 19.20) binding enzyme-linked Boripe tides quietness conductor to the, IPN of 320TT - slightly inhibited in the presence, 8 1 0 3 u presence Below, it was almost completely inhibited (Figure 0). Thus by the present method ^ Then door] ?, 320 - 8 x 1 0 3 S / mi of τ i * if - possible the quantification of I i 1 r of § containing solution and ¾ ivy.
実施例 20 -ίϊ) (Example 20 -ίϊ)
実施例 20一 Π)と原理的には同じであるが、 マ チスキヤ ンを用いて、 自動的に迅速に多数 サンブ を測定出来る方法として JK下の変法を行 づ ito Although the principle is the same as that of Example 20-1), a modified method under JK was carried out as a method for automatically and quickly measuring a large number of sambs using a multi-scan.
(1)マウス I s G吸着ブレー トの作製 (1) Preparation of mouse I sG adsorption plate
9 D穴の microtest 用プレー ト I Nunc一 Imm noplate I: ヌンク社( デンマーク製)の各穴にマウス I g G溶液( 1 Q μ SZmi 0.0 1 Mホウ酸瑷衝液 p H ) 20 0 を注入し 4 で〗晚放置し た。 液を除 た後、 1 の B S Aを含む 0.02 Mリ ン黢緩衝液( pH9 D play for microtest holes bets I Nunc one Imm noplate I: Nunc mouse to each well of (made in Denmark) I g G solution (1 Q μ S Zmi 0.0 1 M borate瑷衝solution p H) injected 20 0 Then left for 4 minutes. After removing the solution, a 0.02 M buffer containing 1 BSA (pH
7.4 ) 300 を注入し、 室瀘で 3時間放置後、 液を除去した。 7.4) 300 was injected and left for 3 hours in a chamber filter, and the liquid was removed.
(2)¾ノクロ -ナ 抗体 r 3 - I 1.1吸着ブレ -トの作製 (2) Preparation of Anticrona antibody r 3-I 1.1 adsorption plate
0.1 % B S Aを含む 0.02 Mリン酸緩衝 ¾( pH 7 )で r 3 - 〗 1. 1 の 350000倍希釈狭を調製し、(1)で作製したマウス I g (J吸着ブレ -トの各穴に, 50 ί 注入した。 4てで 1晚放置後、 狭を除去し、 Prepare a 350,000-fold dilution of r3-〗 1.1 in 0.02 M phosphate buffer 0.1 (pH 7) containing 0.1% BSA, and use the mouse Ig prepared in (1) (each well of the J adsorption plate). Then, after injecting 50ί, leave it at 1 て for 1 て.
0.01 Μリン黢緩衝液( Ρ Η 7.4 )で 3回洗浄し、 抗体 ¾着ブレートを 製しえ。 Wash three times with 0.01 μl buffer ( Ρ 7.4) to produce antibody-adhered plate.
Ο ΡΙ -¾2- (3) 操作法 Ο ΡΙ -¾2- (3) Operation
被検試料ある は標準 I F H - Γを緩衝狭 A ( 0.1 M HaCl, 1 m M MgCl2, 0.1 Si B S A , 0.1 Na 。を含む p H 7の 0.02 M リ ン酸瑷衝液)で適当な濃度に希釈し、各々を (2)で 製したブレ-トの各 穴に 75 ずつ注入する。 次に緩衝液 Aで〗 30倍に希^した実施例 3の酵素標議体の 5 ^ をブレ-トの各穴に注入する。 バラフィ ムを かけ 3 7て〖時閩反応 .引き続き室温で 3時間反応させた。反応硖を除 去後、 0.0 1 Mリン酸緩衝硖( p H 7.4 ) 75 で 4回洗浄した。 The test sample or standard IFH-Γ is adjusted to an appropriate concentration with buffered narrow A (0.02 M phosphoric acid buffer at pH 7 containing 0.1 M HaCl, 1 mM MgCl 2 , 0.1 Si BSA, 0.1 Na). Dilute and inject 75 into each hole of the plate prepared in (2). Next, 5 ^ of the enzyme sample of Example 3 diluted 30 times with buffer A is injected into each well of the plate. The reaction was carried out for 3 hours with the addition of a bulk. The reaction was continued at room temperature for 3 hours. After removing the reaction 硖, the plate was washed four times with 0.01 M phosphate buffer 硖 (pH 7.4) 75.
発色反応のため、 ブレ-トの各穴に緩衝硖 Aで調製した 1.5 の 4 トロフエ- 一 β一 D—ガヲク トビヲノ s/ド(和光繊 ,特級) For the color development reaction, each well of the plate was prepared with buffer 硖 A in each of the wells of 1.5 4 trofu-1-β-1D-galactobino s / do (Wako fine, special grade)
1 50 /^を注入して、 室温で!晚反応させた。 0.4 ΐί炭酸耰街欲(ρΗ 1 0.5 ) 1 0 0 /* を各穴に加えて、 発色反応を伴止し、 405 nmの 吸光度をマイクロタイ トレーシ ¾ンブレート用光度計 Titertek Inject 1 50 / ^ at room temperature!晚 Reacted. 0.4 Ii carbonate耰街greed (ρΗ 1 0.5) 1 0 0 / * was added to each well, sealed accompanied color reaction, 405 nm of absorbance microtiter Tracey ¾ Nbureto for photometer Titertek
Multiskan (フ -一社(米国)製)で測定した。 It was measured by Multiskan (manufactured by U-One (USA)).
図 1 1に i F ir- rの標準曲線を示した。 表 1 0で i FH - rを含む Fig. 11 shows the standard curve of iFir-r. Table 10 includes i FH-r
S種類の被検試料について、本法で得られた結果と抗ウイ ス活性( A V A )とを比較した。 又、 表 1 1に本法の日間変動を示したが、変動係 数 7* 4 と良好 ¾結果を与えた。 For S test samples, the results obtained by this method were compared with the antiviral activity (AVA). Table 11 shows the daily variation of this method. The variation coefficient was 7 * 4, giving good results.
一 OM—PI一 1 One OM—PI one 1
表 1 0 Table 10
10 Ten
表 1 1 Table 11
15 Fifteen
30 30
25 実施例 1 3 , 1 9および 20(i),(ii)で使用した粗 I FN -ァ含有上淸は 以下の参考例で示す方法によ!?得ちれた。 twenty five The crude I FN -α content used in Examples 13, 19 and 20 (i), (ii) was determined by the method shown in the following Reference Example! ? I got it.
参考例〗 Reference example〗
(\) ヒト I - I F ΪΓをコードする mBlTAの分離 (\) Isolation of mBlTA encoding human I-IF F
ヒト末捎血よ])調製したリ ンパ球を 1 2 - 0 -テトラデカノィルホ ボー 一 1 3ーアセテー ト ( T S A ) ( 1 5 a g /ml )とコンカナバ リ ン A ( 40 ^ gZ* ) ¾ tfBPMl- l 640培地( ί 0 の牛胎児血 清を含む)で 3了て培養し、 を誘導させた。 24時間後、 こ の誘導した 1 ! 0 個のヒトリンパ球をチォグァ-ジン変拴溶液( 5 IIグァ二ジンチ才シァネー ト、 5多メ 力ブトエタズ - 、 50 m Μ Human lymphocytes]) prepared lymphocytes were treated with 12-0-tetradecanoylhobo 13-acetate (TSA) (15 ag / ml) and concanavalin A (40 ^ gZ *). The cells were cultured in tfBPMl-l640 medium (including fetal bovine serum of ί0) for 3 hours to induce. 24 hours later, this induced one! 0 human lymphocytes were converted to a solution of guanidine (5 II guanidine, 5 liters of ethanol), 50 mΜ.
Tris · 玨 H 7.6 , ί OmM EDT A )中でテフロンホ乇ゲナイザ 一によつて破壊変性した後 N -ラウ口イリ ザ コシン酸ナト リウムを に ¾るように加え、 質化した混合物を 5,7 M¾化セシウム溶欲After destruction and denaturation with a Teflon homogenizer in Tris (玨 H7.6, ίOmM EDT A), N-lau mouth iriza sodium succinate was added to the mixture, and the quenched mixture was added to 5,7 Cesium cesium lust
〔 5.7 M塩化セシゥム、 0.1 Mエチレンジアミン四齚酸( SDTA ) QMI 上に重層し、 ベックマン S W 27のロータ-を用 て ί 5 で 24000 rpm 30時間遠心処理を行い、 S NA沈嚴を得た。 [Layered on 5.7 M cesium chloride, 0.1 M ethylenediaminetetraacetic acid (SDTA) QMI, and centrifuged at 4,000 rpm for 30 hours at ί5 using a Beckman SW 27 rotor to obtain SNA strain.
この B U A沈殿を 0.25 K -ラウロイ ザ/ uコ シン酸ナト リウム溶液 にとかした後、 エタノールで沈殿させ、 8.3 Wの B If Aを得た。 この!? N Aを高塩溶液〔 0.5 K NaC , 1 OmM Tris · EC£ pH 7. 6 , I mM EDT A , 0.3 SDS〕甲でオリゴ(dT )セノレロースカラムに吸着させ、 The BU precipitate was dissolved in a 0.25 K-lauroyza / u-sodium sodium cosinate solution and precipitated with ethanol to obtain 8.3 W of BIfA. this! ? The NA was adsorbed on an oligo (dT) Senorelose column with a high salt solution [0.5 K NaC, 1 OmM Tris · EC £ pH 7.6, ImM EDTA, 0.3 SDS].
OMPI 一 ー OMPI One
ポリ ( A)を含む mRN Aを低塩溶液( 1 OmM Tr -HCl-pH 7. ό . A solution of mRNA containing poly (A) in a low salt solution (1 OmM Tr-HCl-pH 7.
1 m EDTAjO.3 % SDS )で溶出させることによ]?、 ホ°リ (A ) む mSNA 70 D z を分取した。 By eluting with 1 m EDTAjO.3% SDS), mSNA 70 Dz was collected from the solution (A).
この mEN Aを更にエタノールで沈澱させ、 α2 "の溶液( 1 ΟιαΜ Kris - H C 1 pH 7. ό , 2 mMED , 3 % S D S )に溶カし、 65でで 2 分間処理して 1 Q〜35%庶糖密度勾配遠心処理(ベックマン ST727 のローターを用いて 20で, 25000rpm-C2 時間遠心分離)するこ とによ]?分画化して 2 2分画を得た。 この各分画につき ΉΝ Aの一部 ずつを、 アフ リカッメガエルの卵母細胞に注入し、 合成 れる蛋 0質 中のイ ンタ一フエロ ン活性〔 ヒ ト羊膜由来 ェ SH細胞に対する水泡 性口内炎ウィルス ( VS T)の細胞変性効果阻止試験を用いて測定 : 抗ゥイノレス活性 ( Stewart . W. Ξ . THe interferon -Sys em , (This mENA is further precipitated with ethanol, dissolved in a solution of α2 ″ (1Οαα Kris-HC1 pH 7.ό, 2 mMED, 3% SDS), and treated with 65 for 2 minutes to obtain 1 Q to 35 % sucrose density gradient centrifugation (at 2 0 using a rotor of Beckman ST727, 25000rpm-C2 hour centrifugation) by the the child]? to obtain a fractionated by 2 2 fractions. EN per each fraction A portion of A is injected into oocytes of African frogs, and the interferon activity in the synthesized protein (cytopathic vesicular stomatitis virus (VST) on SH cells derived from human amniotic membrane) Measured using an effect inhibition test: Anti-inoles activity (Stewart. W. Ξ. THe interferon -Sys em, (
Springer Berlinl 97 を測定し、分画 12 (沈降定数 S値は 1 2— 1 4 Sを示した)に 1 ?5ュニフ トノ gENA の活性を検出し ' た。 このようにして得た分画 1 2の mRNAは約 2 0 であった。 Springer Berlinl 97 was measured, and the activity of 1-5 juniftono gENA was detected in fraction 12 (sedimentation constant S value showed 12-14 S). The mRNA of the fraction 12 thus obtained was about 20.
' (ϋ) 単鎖 DN Aの合成 '' (ϋ) Synthesis of single-chain DNA
上記で得た mHN Aおよび逆転写酵素を用い、 1 0 O の反応液( 5 gの mf N A、 50 ォリコ'( d T ) , 1 00ユニットの逆 写酵 素、 1 ずつの dATP ,dCTP , dGTPおよび iTTP , 8 mM Mg Cl2l 50 mM P C 1, 1 Q mMジチ才スレイ トーノレ、 50 mM Tr i s— HC 1 pH 8.3 )中で 42 *C 1時間ィンキュベ一トした後に、 フエノールで涂蛋 白し、 0.1 Nの Na 0Hで 70 'C,20分処理して R N Aを分解除去した。 Using the mHNA and reverse transcriptase obtained above, a reaction solution of 10 O (5 g of mfNA, 50 ori '(dT), 100 units of reverse transcriptase, 1 dATP, 1 dCTP, dGTP and iTTP, 8 mM MgCl 2l 50 mM PC 1, 1 Q mM dithionite, 50 mM Tris-HC 1 pH 8.3) 42 * C Incubate for 1 hour, then extract with phenol Then, the cells were treated with 0.1 N NaOH at 70'C for 20 minutes to decompose and remove RNA.
(iii) 二重鎖 DN Aの合成 _ (iii) Synthesis of double-stranded DNA_
ここで合成された単鎖の相補 DN Aを50^ の反応液(mENAとォリ Complementary DN A 5 0 ^ of the reaction solution of the single chain synthesized here (Mena and O Li
OMPI ゴ d Tを含ま い 外は上記と同じ反応液)中で 4 2 2時間反応させ ることによ 二重鎖 D 1ί Aを合成した。 OMPI The reaction was carried out for 42 hours in the same reaction solution as described above except that it contained no dT, thereby synthesizing a double-stranded D1A.
(iV) d Cティルの付加 (iV) Add d C tile
この二重鎖 3) N Aにヌクレアーゼ S 1を 5 0 < の反応液(二重鎖]) N A 0.1 M酷酸ナト リウム ρΗ4.5 , 0.25 M NaCl , 1.521 ZnS04 ,60 ュ-ッ トの S Iヌクレアーゼ)中で室温 3 0分間作用させ、 フエノー で除蛋白し、 エタノー で D N Aを沈澱させた後、 これにターミナ ト ランスフェラーゼを 5 Q fig の反応液(二重鎖 D M A , 0.14M力コジ 酸カリ , 0.3MTris (塩基) pS 7.6 , 2 ジチオスレィトー ' ImM C0C12 , 0.15 mM dCTP , 3 0ュ-ッ トタ—ミナ トランスフェラー ゼ)中で 3分間 3 7 で ^用させ二重鎖!) N Aの 3^端に約 2 0個のデ ォキシ 'ンチジン鎖を伸長させた。 これらの一連の反応で約 3 0 0 ngの デォキシシチジン鎖をもった二重鎖 D N Aを得た。. The duplex 3) nuclease S 1 5 0 <a reaction solution NA (duplex]) NA 0.1 M Kokusan sodium ρΗ4.5, 0.25 M NaCl, 1.521 ZnS0 4, 60 Interview - Tsu City of SI Nuclease) at room temperature for 30 minutes, deproteinize with phenol, precipitate DNA with ethanol, and add terminator transferase to 5 Q fig reaction mixture (double-stranded DMA, 0.14 M codylic acid). Cali, 0.3MTris (base) pS 7.6, 2 Jichiosureito 'ImM C 0 C1 2, 0.15 mM dCTP, 3 0 Interview - Tsu Tota! - Mina transferase peptidase) by use ^ 3 min 3 7 in duplex) NA Approximately 20 dextrin 'chains were extended at the 3 ^ end of the DNA. In these series of reactions, about 300 ng of double-stranded DNA having a deoxycytidine chain was obtained. .
(V) 大腸菌ブラスミ ドの開裂 ¾らびに d (3ティ の付加 (V) Cleavage of Escherichia coli plasmid
一方、 1 0 /« の大腸菌プラスミ ド pBR 322 D N Aに制限酵素 Pst ェを 5 0 の反応液 (: DNA , 50mM NaCl , 6mM Tris-HCl (pH 7.4 ) , 6 mM MgClo . 6 mM 2—メ カプトエタノール, 100 On the other hand, a restriction enzyme Pste was added to a reaction mixture of 50/50 (NaOH, 6 mM Tris-HCl (pH 7.4), 6 mM MgClo. 6 mM 2-mcapto) with 10 / «of E. coli plasmid pBR322 DNA. Ethanol, 100
牛血清ア ブミン, 2 0ユニットの Pstl〕中で 3時間 3 7 t で 用させて PBR322 D N A中に 1ケ所存在する P s 11認識部位を切 靳し、 フエノールで除蛋白した後、 ターミナ トランスフェラーゼを 5 0 /ί^ の反応液 (: D N A 1 0 A 、 0.14Mカコジ 酸力リ、 0.3M Tris (塩基) pH 7.6 , 2 mMジチオスレィ トー 、 1 mM C0C12. 0.15mM dGTP , 3 0ュ -ッ トターミナ トランスフェラーゼ〕中で 3分間 3 7 t!で ^用させ上記ブラスミド pBR322 D N Aの 3T両端に約 8個のデォキシグァ-ン鎖を延長させた„ Bovine serum albumin, 20 units of Pstl] was used for 3 hours at 37 t to excise one Ps11 recognition site in PBR322 DNA, deproteinized with phenol, and then terminated with terminal transferase. 5 0 / ί ^ of reaction (:. DNA 1 0 a, 0.14M Kakoji acid strength Li, 0.3 M Tris (base) pH 7.6, 2 mM Jichiosurei toe, 1 mM C 0 C1 2 0.15mM dGTP, 3 0 Interview -Butardina transferase) for 3 minutes at 37 t! To extend about 8 deoxyguan chains at both ends of 3T of the above plasmid pBR322 DNA.
Ο ΡΙ - - (Vi) C D N Aの会合るらびに大腸菌の形質変換 Ο ΡΙ --(Vi) CDNA association and transformation of E. coli
このようにして得られた合成! 重鎖 DN A 0.1 μ 9 と上記ブラスミ ド pBR 322 0.5 μ9を 0.1 NaCl , 50 mMTris -HC1 pH7.6 , ImM E D T Aよ] る溶液中で 65 2分間、 4 5 2時間加熱しその後除 冷して会合させ Eneaらの方法〔 X oL BioL 96 t 495(1975) 〕 に従って大腸菌 1 1 7 76 を形質転換させた。 The synthesis obtained in this way! 0.1 μ9 of heavy chain DNA and 0.5 μ9 of the above plasmid pBR322 are heated in a solution of 0.1 NaCl, 50 mM Tris-HC1 pH 7.6, ImM EDTA for 65 2 minutes and 45 2 hours, and then cooled. E. coli 117776 was transformed according to the method of Enea et al. [XOL BioL 96 t 495 (1975)].
(Vii) C D N λ含有ブラスミドの単離 (Vii) Isolation of CDNλ-containing brasmid
こうして約 8500のテトラサイクリン耐性株が単離され、 とれら各々 の D Ν Αを-トロセ ロースフィルターの上に固定した。 ( Grunstein ML and flogness,D. S. Proc. NatL Acad. Sci. USA 72, 3961(1975)) —方、 D.7. Goeddel らの報告〔Nature,295.503(1982)〕の:!:—IF のァミノ酸配列をもとにしてアミノ酸 NO. l-5(Cys-Tyr-Cys.Gln- Asp)及びアミノ酸 NO. 77-82(Lya - Gin- Asp-Met-Asn-Val) よ]? 推測できる植基郵列(それぞれぎ TCCTGGCA TA C3'および 5' )をト リ: cステノレ法〔 Crea. R.ら、 Proc.Approximately 8,500 tetracycline-resistant strains were isolated in this way, and each of these DΝDΝ was immobilized on a -Troserose filter. (Grunstein ML and flogness, DS Proc. NatL Acad. Sci. USA 72, 3961 (1975)) —D.7. Goeddel et al.'S report [Nature, 295.503 (1982)]:!: — IF amino acids Amino acid NO. L-5 (Cys-Tyr-Cys.Gln-Asp) and amino acid NO. 77-82 (Lya-Gin-Asp-Met-Asn-Val) based on the sequence]? Postal (TCCTGGCA TA C 3 'and 5' respectively) ) To Tri: c Stenole method [Crea. R. et al., Proc.
Natl. Acad. Soi. USA, , 5765 ( 1978 )〕を用いて化学合成した。 こ の才リゴヌクレオチドに対して T 4ホ'リヌクレオチドカイネースを用い て 50 β の反応液(ォリゴヌクレオチド 0.2 «", 5 OmM Tris 'HC1 pH , 1 OmM MgCl2 , 10 mMメルカプトエタノー 、 50μαγ- 3c,P ATP , 3^=·ット T4 ポリヌクレ才チドカイネース )中で 1時間 37 で反応させ、 5'末端を32 Ρで標識した。 この標識されたオリゴヌクレ 才チドをプローブとして Lsuwnらの方法〔 Nucleic Acids Res. , 9, 6103 (1981 ) 〕に従って上記の-トロセルロースフィルター上に固 定した D Ν Αに会合させ、 ォー トラジオグラフィーによって上記二種類 のオリゴヌクレオチドブローブに反応する菌株を 4個単離した。 -S8- これらの菌株の各々の菌体からプラスミド D N Aをァノレカリ法〔Birn— boim Η· C. & Doly,;r. Nucleic Acids Res..7, 1513(1979)〕によ つて単離した。 次にプラスミ ド D N Aの挿入部を制限酵素 Pstlによ]) 切])出し、分離したブヲスミドのうちでその挿入部の長さの最も長 断 片を含むものをえらび、 とのプラスミ ドを PHIT3709と名ずけた。 次にこの pSIT 3709ブラスミドに揷入された mRliA¾列の一次構造 (塩基 列)をジヌクレオチド合成鎖停止法と Maxam-Gilbertの方法 によって決定した。その一次構造は第 2図の通 であった。 Natl. Acad. Soi. USA, 5765 (1978)]. A reaction solution of 50 β was prepared using the T4 oligonucleotide (Kinase) for this lignonucleotide (oligonucleotide 0.2 «, 5 OmM Tris' HC1 pH, 1 OmM MgCl 2 , 10 mM mercaptoethanol, 50 μαγ -3 c, P ATP, 3 ^ = · T4 Polynucleotide (1) 37 hours, 37 ', and 5'-end was labeled with 32 。. Using this labeled oligonucleotide as a probe, Lsuwn et al. A strain that is associated with the D- し た fixed on the above-mentioned trocellulose filter according to the method [Nucleic Acids Res., 9, 6103 (1981)] and reacts with the two oligonucleotide probes by autoradiography. Were isolated. -S8- Plasmid DNA was isolated from the cells of each of these strains by the Anorecali method [Birn-boim II C. & Doly, r. Nucleic Acids Res .. 7, 1513 (1979)]. Next, insert the plasmid DNA insert with the restriction enzyme Pstl]), cut out)), select the separated plasmids containing the longest fragment of the insert, and replace the plasmid with PHIT3709. It was named. Next, the primary structure (base sequence) of the mRliA sequence inserted into this pSIT3709 plasmid was determined by the dinucleotide synthesis chain termination method and the Maxam-Gilbert method. Its primary structure is shown in Figure 2.
参考例 2 Reference example 2
(I) 発現プラス ミ ドとして大腸蕾のトリブトファン合成のプロモーター 部分〔プロモータ一、 オペレーターを含む D If A断片、 276塩基対、 Bennett,G. Ν·ら, J. Μοΐ· Biol., 121 , 113(1978) 〕を含むブラス ミ ド ptrp601 (ベクターは pBR 322 ) を構築した。 (I) The promoter part of tributophan synthesis in the colon bud [Expression plasmid] [Promoter, D If A fragment including operator, 276 base pairs, Bennett, G. Ν · et al., J. Μοΐ · Biol., 121, 113 (1978)] was constructed to construct a plasmid ptrp601 (vector is pBR322).
—方、 プラスミ ド PBR322を制限酵素 EcoRI および制限酵素 Ava —Plasmid PBR322 with restriction enzymes EcoRI and Ava
Iで切断し、得られたテトヲサイクリ ン耐性遺伝子を含む EcoRI— Ava I断片のの]?しろ部分を D N Aホ。リメラーゼ Iラージフヲグメントでう めた。 この断片を T 4 D N Aリガ—ゼを用いて ptrp601の PvuIIの切 断部位に結合させ ptrp701を構築した(図 6 )。 Of the EcoRI-Ava I fragment containing the Tet-cyclin resistance gene The white part is DNA. I chose Limerase I large fragment. This fragment was ligated to the cleavage site of PvuII of ptrp601 using T4DNA ligase to construct ptrp701 (FIG. 6).
(B) 次に ptrp701に 2ケ所存在する制限酵素 Clal切断部位の一つを 消去するため、 ptrp701を Clalで部分分解して二つの Clal切断部 位のうち一方のみが切断されたものを得た。生じたの]?しろ部分を D N JLボリメラーゼ Iヲージフラグメ ントでうめたのち、 T 4 D N Aリガー ゼで再度結合させて ptrp771を得た(図 6 )。 (B) Next, ptrp701 was partially digested with Clal to eliminate one of the two restriction enzyme Clal cleavage sites present in ptrp701 to obtain a fragment in which only one of the two Clal cleavage sites was cleaved. . The resulting portion was filled in with DN JL volimase I page fragment and then religated with T4DNA ligase to obtain ptrp771 (FIG. 6).
(Hi) pHIT3709を制限酵素 Pst Iで切断して I FN- rの構造遺伝子を - - 含む Pstl断片を得た。 この断片を制限酵素 BstNI で部分分解し、 I FN- 構造遺伝子内にある BstNI 部位の切断された BstNI -: Pstl断 片を得た。 BstNI 切断部位のの j)しろ部分を D N Aポリメ ラーゼ Iラ —ジフラグメ ントでうめたのち、 前述したト リ エステル法によって化学 合成した蛋白合成開始コ ドン A T Gを含むォリゴヌク レ才チ ドアダプタ 一 C G AT A A TGTG T A C T G C C (Hi) pHIT3709 is cut with the restriction enzyme Pst I to obtain the IFN-r structural gene. --The obtained Pstl fragment was obtained. This fragment was partially digested with the restriction enzyme BstNI to obtain a BstNI-: Pstl fragment in which the BstNI site in the IFN- structural gene was cut. After filling the j) margin of the BstNI cleavage site with DNA polymerase I difragment, an oligonucleoside adapter containing a protein synthesis initiation codon ATG chemically synthesized by the above-mentioned triester method CG AT AA TGTG TACTGCC
TA TT AC A C AA TG A C GG TA TT AC A C AA TG A C GG
を T 4 D N Aリガ―ゼで結合させた。 Was ligated with T4DNA ligase.
一方、 上記アダプタ—を結合させた I FN- Γ遺伝子を ptrp771を制 限酵素 Ps tlと制限酵素 Clalで切断して得た断片のト リ プト フアンプ 口モータ一の下流に揷入して T 4 D N Aリガーゼを用いて結合させ、 ェ Ffi- r発現プラスミ ド pHIT trpl lOlを構築した(.12 ワ )。 On the other hand, a fragment obtained by cutting the ptrp771 with the restriction enzyme Pstl and the restriction enzyme Clal into the IFN-Γ gene to which the above-mentioned adapter was bound was inserted into the downstream of one of the motors of T4 and T4. The DNA was ligated using DNA ligase to construct an Ffi-r expression plasmid pHIT trplIOl (.12 W).
(ίν) pHITtrpl lOl をさらに改良して次の通]) pHITtrp2101 を構 築した。 (ίν) pHITtrplOl was further improved and the following passage was established.)) pHITtrp2101 was constructed.
まず ptrp601を制限酵素 Clalおよび制限酵素 Hpallで処理して trpプロモーターを含む Clal— Hpa!I断片 0· 3 3 Kb を得た。 この断 片を、 Clalで切断しァノレ力 リ ホスファタ—ゼ処理した pHITtrpl lO First, ptrp601 was treated with restriction enzymes Clal and Hpall to obtain a Clal-Hpa! I fragment 0.333 Kb containing a trp promoter. This fragment was cut with Clal and treated with ethanol phosphatase.
1に T 4 D N Aリガーゼを用いて結合させ trpプロモーターが二つ直列 に入った pHITtrP 2101 を得た( (2l ^ )。 Trp promoter and combined using T 4 DNA ligase to obtain a pHITtr P 2101 entered the two series 1 ((2l ^).
このプラスミ ド ρΗΙΤ · trp2101 を用いて Cohen らの方法〔前出〕 に従って、大腸菌 2 9 4を形質転換させ、 このプラスミ ドを含む菌株 Escherichia coli 294 was transformed with this plasmid ρΗΙΤ · trp2101 according to the method of Cohen et al.
E. 'coli294ZpHIT · trp2101を得た。 E. 'coli294ZpHIT · trp2101 was obtained.
蔘考例 ¾ Ginseng example ¾
- E. coli294ZpHITtrp2101を 8 μ9/ηί€>亍 ト ラサイ ク リ ン, 0.4 % カザミ ノ酸,.1 %グルコースを含む Μ 9培地 2 0 0 Wを分注した 1 -E. coli 294ZpHITtrp2101 containing 8 μ9 / ηί >> tracycline, 0.4% casamino acid, and 0.1% glucose.
Ο ΡΙΟ ΡΙ
WIPO , マイヤー中で 3 7 tで培養し、生育が K U 220に達した時に 3 β一 イ ンドリ一ノレアクリノレ酸( I A A )を 3 0 S1/^に るように加えて更 に 4時間培養した。 WIPO, The cells were cultured in a Meyer at 37 t, and when the growth reached KU220, 3β-indolino-inorea-clinoleic acid (IAA) was added to 30 S 1 / ^, and the cells were further cultured for 4 hours.
参考例 4 Reference example 4
参考例 3で得られた培養液 1. 2 を遠心分離して菌体を集め、 これ 6 0 i の 1 0 ¾ シュクロースを含む 0.0 5 Tris -HC1 pH 7.6 に懸濁した。 この菌体懸濁液に 0.2 Mフエニノレーメチノレース レフォニ フノレオラィ ト、、( P S F ) 0. 3 m , 5 M NaCl 2.4 nl , 0.2 Μェチ レンジア ミ ンテ ト ラアセテー ト ( Ε D Τ A ) 2.4 Λ , 1 Mスぺノレミ ジ ン 2:4 W, 5^Z リゾチーム. 2.4 « を加え 0 で 1時間放置した のち、 3 7 で 5分処理し、 れを更に ARTEK社(米国)製超音波破 薛器で 0 'C 3 0秒処理した。 The culture solution 1.2 obtained in Reference Example 3 was centrifuged to collect bacterial cells, and the cells were suspended in 0.05 Tris-HC1 pH 7.6 containing 60i of 10% sucrose. 0.2 M feninole methinolace refoni phnoleorate, (PSF) 0.3 m, 5 M NaCl 2.4 nl, 0.2 チ レ ン ジ ア ア ミ ミ ア ((((Ε D Ε A) 2.4 M, 1 M Sunoremidin 2: 4 W, 5 ^ Z lysozyme. Add 2.4 «, leave at 0 for 1 hour, treat with 3 7 for 5 minutes, and further apply ARTEK (USA) ultrasonic It was processed at 0'C for 30 seconds with the Xiao Xian.
この溶菌液を 105 ,000x? で 1時間遠心分離して上清 6 6 を集 めた。 産業上の利用可能性 The lysate was centrifuged at 105,000x for 1 hour to collect the supernatant 66. Industrial applicability
本発明の新規ポリぺプチドを使用してヒ ト ァ型イ ンタ -フエ口ンに結 合するモノクロ-ナル抗体を製造することができる。 該モノクロ -ナ 抗体は、 ヒ ト Γ型イ ンタ - フエロ ンの検出,精製に有用である, The novel polypeptide of the present invention can be used to produce a monoclonal antibody that binds to a human interferon. The monoclonal antibody is useful for detection and purification of human Γ-type interferon.
O PI O PI
V/IPO V / IPO
Claims
Priority Applications (13)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP1983/000174 WO1984004745A1 (en) | 1983-05-31 | 1983-05-31 | Novel polypeptides and their use |
| US06/534,091 US4681848A (en) | 1982-09-22 | 1983-09-20 | Novel peptide and use thereof |
| EP83109402A EP0103898B1 (en) | 1982-09-22 | 1983-09-21 | Novel peptide and use thereof |
| NZ205666A NZ205666A (en) | 1982-09-22 | 1983-09-21 | Peptide derived from c-terminal region of human immune interferon,and conjugate with carrier protein;hybridomas and production of monoclonal antibodies against human immune interferon;purification or detection of human immune interferon |
| CA000437187A CA1216807A (en) | 1982-09-22 | 1983-09-21 | Peptide and use thereof |
| IE2215/83A IE56003B1 (en) | 1982-09-22 | 1983-09-21 | Novel peptide and use thereof |
| IL69790A IL69790A (en) | 1982-09-22 | 1983-09-21 | Peptides consisting of a sequence of human ifn-ypsilon and use thereof |
| DE8383109402T DE3362936D1 (en) | 1982-09-22 | 1983-09-21 | Novel peptide and use thereof |
| DK430583A DK430583A (en) | 1982-09-22 | 1983-09-21 | POLYPEPTIDES AND THEIR PREPARATION AND USE |
| PH29569A PH22559A (en) | 1982-09-22 | 1983-09-21 | Monoclonal antibody and method for purifying human gamma interferon |
| AT83109402T ATE19088T1 (en) | 1982-09-22 | 1983-09-21 | PEPTIDE AND ITS USE. |
| AU19345/83A AU564620B2 (en) | 1982-09-22 | 1983-09-21 | Peptides and protein-peptide complexes |
| KR1019830004438A KR910001726B1 (en) | 1982-09-22 | 1983-09-22 | Process for preparation of novel peptide |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP1983/000174 WO1984004745A1 (en) | 1983-05-31 | 1983-05-31 | Novel polypeptides and their use |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1984004745A1 true WO1984004745A1 (en) | 1984-12-06 |
Family
ID=13789993
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP1983/000174 Ceased WO1984004745A1 (en) | 1982-09-22 | 1983-05-31 | Novel polypeptides and their use |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO1984004745A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7201897B2 (en) | 1996-05-31 | 2007-04-10 | Hoffmann-La Roche Inc. | Interferon conjugates |
-
1983
- 1983-05-31 WO PCT/JP1983/000174 patent/WO1984004745A1/en not_active Ceased
Non-Patent Citations (2)
| Title |
|---|
| Miklos Bodanszky et al: "Peptide Sythesis (Second Edition)", (1976) John Wiley & Sons (New York) * |
| Nature, Vol. 295, "February 11, 1982" Go (11.02.82) p. 503-508 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7201897B2 (en) | 1996-05-31 | 2007-04-10 | Hoffmann-La Roche Inc. | Interferon conjugates |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| FI90983B (en) | Process for producing hybrid interferon polypeptides | |
| KR910001726B1 (en) | Process for preparation of novel peptide | |
| JP3375949B2 (en) | Antibodies to cytotoxic lymphocyte maturation factor | |
| FI81832C (en) | FOERFARANDE FOER FRAMSTAELLNING AV YTTERST RENT REKOMBINANT HUMANT IMMUN INTERFERONPROTEIN. | |
| JPH0794480B2 (en) | Antibody and method for producing the same | |
| JPH07501698A (en) | Bivalent specific heterodimer | |
| JPH11505132A (en) | Hybrid of interferon-α and immunoglobulin bound via non-immunogenic peptide | |
| JPH03204896A (en) | Extracellular fragment of interferon receptor | |
| JP3784078B2 (en) | Soluble LDL receptor | |
| CN101797381A (en) | Compound of hepatitis B virus antigen and lewis oligosaccharide and preparation method and application thereof | |
| WO1984004745A1 (en) | Novel polypeptides and their use | |
| CN86108955A (en) | New cell growth regulator | |
| WO2023016564A1 (en) | GPC3-TARGETED ANTIBODY INTERFERON α FUSION PROTEIN AND USE THEREOF | |
| CN115125215B (en) | Hybridoma cell strain secreting porcine IFN-lambda 4 monoclonal antibody, monoclonal antibody secreted by hybridoma cell strain and application of hybridoma cell strain | |
| JPS6034994A (en) | Novel polypeptide and its use | |
| CN114957477B (en) | Porcine RIG-I-like receptor RIG-I specific monoclonal antibodies and applications | |
| JPH0646959B2 (en) | Novel polypeptide and its use | |
| CN116769013A (en) | Interleukin 2 mutant and complex containing same | |
| CN117659158A (en) | IL-2 derivative and preparation method thereof | |
| WO1984002130A1 (en) | Novel polypeptides and their use | |
| Wang et al. | Prokaryotic-expressed porcine IFNα1-THYα1 fusion proteins exert IFN and THY activities in vitro | |
| JPH0653067B2 (en) | Novel hybridoma and method for producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Designated state(s): MC |